User login
Bringing you the latest news, research and reviews, exclusive interviews, podcasts, quizzes, and more.
div[contains(@class, 'header__large-screen')]
div[contains(@class, 'read-next-article')]
div[contains(@class, 'nav-primary')]
nav[contains(@class, 'nav-primary')]
section[contains(@class, 'footer-nav-section-wrapper')]
footer[@id='footer']
div[contains(@class, 'main-prefix')]
section[contains(@class, 'nav-hidden')]
div[contains(@class, 'ce-card-content')]
nav[contains(@class, 'nav-ce-stack')]
Scurvy: A Diagnosis Still Relevant Today
“Petechial rash often prompts further investigation into hematological, dermatological, or vasculitis causes. However, if the above investigations are negative and skin biopsy has not revealed a cause, there is a Renaissance-era diagnosis that is often overlooked but is easily investigated and treated,” wrote Andrew Dermawan, MD, and colleagues from Sir Charles Gairdner Hospital in Nedlands, Australia, in BMJ Case Reports. The diagnosis they highlight is scurvy, a disease that has faded from common medical concern but is reemerging, partly because of the rise in bariatric surgery.
Diagnosing Scurvy in the 2020s
In their article, Dermawan and colleagues present the case of a 50-year-old man with a bilateral petechial rash on his lower limbs, without any history of trauma. The patient, who exhibited no infectious symptoms, also had gross hematuria, microcytic anemia, mild neutropenia, and lymphopenia. Tests for autoimmune and hematological diseases were negative, as were abdominal and leg CT scans, ruling out abdominal hemorrhage and vasculitis. Additionally, a skin biopsy showed no causative findings.
The doctors noted that the patient had undergone sleeve gastrectomy, prompting them to inquire about his diet. They discovered that, because of financial difficulties, his diet primarily consisted of processed foods with little to no fruits or vegetables, and he had stopped taking supplements recommended by his gastroenterologist. Further tests revealed a vitamin D deficiency and a severe deficiency in vitamin C. With the diagnosis of scurvy confirmed, the doctors treated the patient with 1000 mg of ascorbic acid daily, along with cholecalciferol, folic acid, and a multivitamin complex, leading to a complete resolution of his symptoms.
Risk Factors Then and Now
It can cause mucosal and gastric hemorrhages, and if left untreated, it can lead to fatal bleeding.
Historically known as “sailors’ disease,” scurvy plagued men on long voyages who lacked access to fresh fruits or vegetables and thus did not get enough vitamin C. In 1747, James Lind, a British physician in the Royal Navy, demonstrated that the consumption of oranges and lemons could combat scurvy.
Today’s risk factors for scurvy include malnutrition, gastrointestinal disorders (eg, chronic inflammatory bowel diseases), alcohol and tobacco use, eating disorders, psychiatric illnesses, dialysis, and the use of medications that reduce the absorption of ascorbic acid (such as corticosteroids and proton pump inhibitors).
Scurvy remains more common among individuals with unfavorable socioeconomic conditions. The authors of the study emphasize how the rising cost of living — specifically in Australia but applicable elsewhere — is changing eating habits, leading to a high consumption of low-cost, nutritionally poor foods.
Poverty has always been a risk factor for scurvy, but today there may be an additional cause: bariatric surgery. Patients undergoing these procedures are at a risk for deficiencies in fat-soluble vitamins A, D, E, and K, and if their diet is inadequate, they may also experience a vitamin C deficiency. Awareness of this can facilitate the timely diagnosis of scurvy in these patients.
This story was translated from Univadis Italy using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article appeared on Medscape.com.
“Petechial rash often prompts further investigation into hematological, dermatological, or vasculitis causes. However, if the above investigations are negative and skin biopsy has not revealed a cause, there is a Renaissance-era diagnosis that is often overlooked but is easily investigated and treated,” wrote Andrew Dermawan, MD, and colleagues from Sir Charles Gairdner Hospital in Nedlands, Australia, in BMJ Case Reports. The diagnosis they highlight is scurvy, a disease that has faded from common medical concern but is reemerging, partly because of the rise in bariatric surgery.
Diagnosing Scurvy in the 2020s
In their article, Dermawan and colleagues present the case of a 50-year-old man with a bilateral petechial rash on his lower limbs, without any history of trauma. The patient, who exhibited no infectious symptoms, also had gross hematuria, microcytic anemia, mild neutropenia, and lymphopenia. Tests for autoimmune and hematological diseases were negative, as were abdominal and leg CT scans, ruling out abdominal hemorrhage and vasculitis. Additionally, a skin biopsy showed no causative findings.
The doctors noted that the patient had undergone sleeve gastrectomy, prompting them to inquire about his diet. They discovered that, because of financial difficulties, his diet primarily consisted of processed foods with little to no fruits or vegetables, and he had stopped taking supplements recommended by his gastroenterologist. Further tests revealed a vitamin D deficiency and a severe deficiency in vitamin C. With the diagnosis of scurvy confirmed, the doctors treated the patient with 1000 mg of ascorbic acid daily, along with cholecalciferol, folic acid, and a multivitamin complex, leading to a complete resolution of his symptoms.
Risk Factors Then and Now
It can cause mucosal and gastric hemorrhages, and if left untreated, it can lead to fatal bleeding.
Historically known as “sailors’ disease,” scurvy plagued men on long voyages who lacked access to fresh fruits or vegetables and thus did not get enough vitamin C. In 1747, James Lind, a British physician in the Royal Navy, demonstrated that the consumption of oranges and lemons could combat scurvy.
Today’s risk factors for scurvy include malnutrition, gastrointestinal disorders (eg, chronic inflammatory bowel diseases), alcohol and tobacco use, eating disorders, psychiatric illnesses, dialysis, and the use of medications that reduce the absorption of ascorbic acid (such as corticosteroids and proton pump inhibitors).
Scurvy remains more common among individuals with unfavorable socioeconomic conditions. The authors of the study emphasize how the rising cost of living — specifically in Australia but applicable elsewhere — is changing eating habits, leading to a high consumption of low-cost, nutritionally poor foods.
Poverty has always been a risk factor for scurvy, but today there may be an additional cause: bariatric surgery. Patients undergoing these procedures are at a risk for deficiencies in fat-soluble vitamins A, D, E, and K, and if their diet is inadequate, they may also experience a vitamin C deficiency. Awareness of this can facilitate the timely diagnosis of scurvy in these patients.
This story was translated from Univadis Italy using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article appeared on Medscape.com.
“Petechial rash often prompts further investigation into hematological, dermatological, or vasculitis causes. However, if the above investigations are negative and skin biopsy has not revealed a cause, there is a Renaissance-era diagnosis that is often overlooked but is easily investigated and treated,” wrote Andrew Dermawan, MD, and colleagues from Sir Charles Gairdner Hospital in Nedlands, Australia, in BMJ Case Reports. The diagnosis they highlight is scurvy, a disease that has faded from common medical concern but is reemerging, partly because of the rise in bariatric surgery.
Diagnosing Scurvy in the 2020s
In their article, Dermawan and colleagues present the case of a 50-year-old man with a bilateral petechial rash on his lower limbs, without any history of trauma. The patient, who exhibited no infectious symptoms, also had gross hematuria, microcytic anemia, mild neutropenia, and lymphopenia. Tests for autoimmune and hematological diseases were negative, as were abdominal and leg CT scans, ruling out abdominal hemorrhage and vasculitis. Additionally, a skin biopsy showed no causative findings.
The doctors noted that the patient had undergone sleeve gastrectomy, prompting them to inquire about his diet. They discovered that, because of financial difficulties, his diet primarily consisted of processed foods with little to no fruits or vegetables, and he had stopped taking supplements recommended by his gastroenterologist. Further tests revealed a vitamin D deficiency and a severe deficiency in vitamin C. With the diagnosis of scurvy confirmed, the doctors treated the patient with 1000 mg of ascorbic acid daily, along with cholecalciferol, folic acid, and a multivitamin complex, leading to a complete resolution of his symptoms.
Risk Factors Then and Now
It can cause mucosal and gastric hemorrhages, and if left untreated, it can lead to fatal bleeding.
Historically known as “sailors’ disease,” scurvy plagued men on long voyages who lacked access to fresh fruits or vegetables and thus did not get enough vitamin C. In 1747, James Lind, a British physician in the Royal Navy, demonstrated that the consumption of oranges and lemons could combat scurvy.
Today’s risk factors for scurvy include malnutrition, gastrointestinal disorders (eg, chronic inflammatory bowel diseases), alcohol and tobacco use, eating disorders, psychiatric illnesses, dialysis, and the use of medications that reduce the absorption of ascorbic acid (such as corticosteroids and proton pump inhibitors).
Scurvy remains more common among individuals with unfavorable socioeconomic conditions. The authors of the study emphasize how the rising cost of living — specifically in Australia but applicable elsewhere — is changing eating habits, leading to a high consumption of low-cost, nutritionally poor foods.
Poverty has always been a risk factor for scurvy, but today there may be an additional cause: bariatric surgery. Patients undergoing these procedures are at a risk for deficiencies in fat-soluble vitamins A, D, E, and K, and if their diet is inadequate, they may also experience a vitamin C deficiency. Awareness of this can facilitate the timely diagnosis of scurvy in these patients.
This story was translated from Univadis Italy using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article appeared on Medscape.com.
Plasma Omega-6 and Omega-3 Fatty Acids Inversely Associated With Cancer
TOPLINE:
Higher plasma levels of omega-6 and omega-3 fatty acids are associated with a lower incidence of cancer. However, omega-3 fatty acids are linked to an increased risk for prostate cancer, specifically.
METHODOLOGY:
- Researchers looked for associations of plasma omega-3 and omega-6 polyunsaturated fatty acids (PUFAs) with the incidence of cancer overall and 19 site-specific cancers in the large population-based prospective UK Biobank cohort.
- They included 253,138 participants aged 37-73 years who were followed for an average of 12.9 years, with 29,838 diagnosed with cancer.
- Plasma levels of omega-3 and omega-6 fatty acids were measured using nuclear magnetic resonance and expressed as percentages of total fatty acids.
- Participants with cancer diagnoses at baseline, those who withdrew from the study, and those with missing data on plasma PUFAs were excluded.
- The study adjusted for multiple covariates, including age, sex, ethnicity, socioeconomic status, lifestyle behaviors, and family history of diseases.
TAKEAWAY:
- Higher plasma levels of omega-6 and omega-3 fatty acids were associated with a 2% and 1% reduction in overall cancer risk per SD increase, respectively (P = .001 and P = .03).
- Omega-6 fatty acids were inversely associated with 14 site-specific cancers, whereas omega-3 fatty acids were inversely associated with five site-specific cancers.
- Prostate cancer was positively associated with omega-3 fatty acids, with a 3% increased risk per SD increase (P = .049).
- A higher omega-6/omega-3 ratio was associated with an increased risk for overall cancer, and three site-specific cancers showed positive associations with the ratio. “Each standard deviation increase, corresponding to a 13.13 increase in the omega ratio, was associated with a 2% increase in the risk of rectum cancer,” for example, the authors wrote.
IN PRACTICE:
“Overall, our findings provide support for possible small net protective roles of omega-3 and omega-6 PUFAs in the development of new cancer incidence. Our study also suggests that the usage of circulating blood biomarkers captures different aspects of dietary intake, reduces measurement errors, and thus enhances statistical power. The differential effects of omega-6% and omega-3% in age and sex subgroups warrant future investigation,” wrote the authors of the study.
SOURCE:
The study was led by Yuchen Zhang of the University of Georgia in Athens, Georgia. It was published online in the International Journal of Cancer.
LIMITATIONS:
The study’s potential for selective bias persists due to the participant sample skewing heavily toward European ancestry and White ethnicity. The number of events was small for some specific cancer sites, which may have limited the statistical power. The study focused on total omega-3 and omega-6 PUFAs, with only two individual fatty acids measured. Future studies are needed to examine the roles of other individual PUFAs and specific genetic variants.
DISCLOSURES:
This study was supported by grants from the National Institute of General Medical Sciences of the National Institutes of Health. No relevant conflicts of interest were disclosed by the authors.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article appeared on Medscape.com.
TOPLINE:
Higher plasma levels of omega-6 and omega-3 fatty acids are associated with a lower incidence of cancer. However, omega-3 fatty acids are linked to an increased risk for prostate cancer, specifically.
METHODOLOGY:
- Researchers looked for associations of plasma omega-3 and omega-6 polyunsaturated fatty acids (PUFAs) with the incidence of cancer overall and 19 site-specific cancers in the large population-based prospective UK Biobank cohort.
- They included 253,138 participants aged 37-73 years who were followed for an average of 12.9 years, with 29,838 diagnosed with cancer.
- Plasma levels of omega-3 and omega-6 fatty acids were measured using nuclear magnetic resonance and expressed as percentages of total fatty acids.
- Participants with cancer diagnoses at baseline, those who withdrew from the study, and those with missing data on plasma PUFAs were excluded.
- The study adjusted for multiple covariates, including age, sex, ethnicity, socioeconomic status, lifestyle behaviors, and family history of diseases.
TAKEAWAY:
- Higher plasma levels of omega-6 and omega-3 fatty acids were associated with a 2% and 1% reduction in overall cancer risk per SD increase, respectively (P = .001 and P = .03).
- Omega-6 fatty acids were inversely associated with 14 site-specific cancers, whereas omega-3 fatty acids were inversely associated with five site-specific cancers.
- Prostate cancer was positively associated with omega-3 fatty acids, with a 3% increased risk per SD increase (P = .049).
- A higher omega-6/omega-3 ratio was associated with an increased risk for overall cancer, and three site-specific cancers showed positive associations with the ratio. “Each standard deviation increase, corresponding to a 13.13 increase in the omega ratio, was associated with a 2% increase in the risk of rectum cancer,” for example, the authors wrote.
IN PRACTICE:
“Overall, our findings provide support for possible small net protective roles of omega-3 and omega-6 PUFAs in the development of new cancer incidence. Our study also suggests that the usage of circulating blood biomarkers captures different aspects of dietary intake, reduces measurement errors, and thus enhances statistical power. The differential effects of omega-6% and omega-3% in age and sex subgroups warrant future investigation,” wrote the authors of the study.
SOURCE:
The study was led by Yuchen Zhang of the University of Georgia in Athens, Georgia. It was published online in the International Journal of Cancer.
LIMITATIONS:
The study’s potential for selective bias persists due to the participant sample skewing heavily toward European ancestry and White ethnicity. The number of events was small for some specific cancer sites, which may have limited the statistical power. The study focused on total omega-3 and omega-6 PUFAs, with only two individual fatty acids measured. Future studies are needed to examine the roles of other individual PUFAs and specific genetic variants.
DISCLOSURES:
This study was supported by grants from the National Institute of General Medical Sciences of the National Institutes of Health. No relevant conflicts of interest were disclosed by the authors.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article appeared on Medscape.com.
TOPLINE:
Higher plasma levels of omega-6 and omega-3 fatty acids are associated with a lower incidence of cancer. However, omega-3 fatty acids are linked to an increased risk for prostate cancer, specifically.
METHODOLOGY:
- Researchers looked for associations of plasma omega-3 and omega-6 polyunsaturated fatty acids (PUFAs) with the incidence of cancer overall and 19 site-specific cancers in the large population-based prospective UK Biobank cohort.
- They included 253,138 participants aged 37-73 years who were followed for an average of 12.9 years, with 29,838 diagnosed with cancer.
- Plasma levels of omega-3 and omega-6 fatty acids were measured using nuclear magnetic resonance and expressed as percentages of total fatty acids.
- Participants with cancer diagnoses at baseline, those who withdrew from the study, and those with missing data on plasma PUFAs were excluded.
- The study adjusted for multiple covariates, including age, sex, ethnicity, socioeconomic status, lifestyle behaviors, and family history of diseases.
TAKEAWAY:
- Higher plasma levels of omega-6 and omega-3 fatty acids were associated with a 2% and 1% reduction in overall cancer risk per SD increase, respectively (P = .001 and P = .03).
- Omega-6 fatty acids were inversely associated with 14 site-specific cancers, whereas omega-3 fatty acids were inversely associated with five site-specific cancers.
- Prostate cancer was positively associated with omega-3 fatty acids, with a 3% increased risk per SD increase (P = .049).
- A higher omega-6/omega-3 ratio was associated with an increased risk for overall cancer, and three site-specific cancers showed positive associations with the ratio. “Each standard deviation increase, corresponding to a 13.13 increase in the omega ratio, was associated with a 2% increase in the risk of rectum cancer,” for example, the authors wrote.
IN PRACTICE:
“Overall, our findings provide support for possible small net protective roles of omega-3 and omega-6 PUFAs in the development of new cancer incidence. Our study also suggests that the usage of circulating blood biomarkers captures different aspects of dietary intake, reduces measurement errors, and thus enhances statistical power. The differential effects of omega-6% and omega-3% in age and sex subgroups warrant future investigation,” wrote the authors of the study.
SOURCE:
The study was led by Yuchen Zhang of the University of Georgia in Athens, Georgia. It was published online in the International Journal of Cancer.
LIMITATIONS:
The study’s potential for selective bias persists due to the participant sample skewing heavily toward European ancestry and White ethnicity. The number of events was small for some specific cancer sites, which may have limited the statistical power. The study focused on total omega-3 and omega-6 PUFAs, with only two individual fatty acids measured. Future studies are needed to examine the roles of other individual PUFAs and specific genetic variants.
DISCLOSURES:
This study was supported by grants from the National Institute of General Medical Sciences of the National Institutes of Health. No relevant conflicts of interest were disclosed by the authors.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article appeared on Medscape.com.
Digital Danger: How Cyberattacks Put Patients at Risk
On September 27, 2024, UMC Health System in Lubbock, Texas, experienced an IT outage because of a cybersecurity incident that temporarily diverted patients to other healthcare facilities. So far, in 2024, there have been 386 cyberattacks on healthcare organizations. These high-impact ransomware attacks disrupt and delay patient care.
In recent years, many healthcare systems, including Scripps Health, Universal Health Services, Vastaamo, Sky Lakes, and the University of Vermont, have paid millions — even tens of millions — to recover data after a cyberattack or data breach. When healthcare systems come under cyber fire, the impact extends far past disrupting workflows and compromising data, patient safety can be also be compromised, vital information may be lost, and imaging and lab results can go missing or be held for ransom, making physicians’ job difficult or impossible.
In fact, cyberattacks on hospitals are far more common than you may realize. A new report issued by Ponemon and Proofpoint found that 92% of healthcare organizations have experienced a cyberattack in the past 12 months. Even more sobering is that about half of the organizations affected suffered disruptions in patient care.
Healthcare Systems = ‘Soft Targets’
Healthcare systems are a “soft target” for hackers for several reasons, pointed out Matthew Radolec, vice president, incident response and cloud operations at Varonis, a data security company. “One, they’re usually an amalgamation of many healthcare systems that are interconnected,” said Radolec. “A lot of hospitals are connected to other hospitals or connected to educational institutions, which means their computer vulnerabilities are shared ... and if they have an issue, it could very easily spread to your network.”
Another factor is the cost of securing data. “[With hospitals], they’ll say that a dollar spent on security is a dollar not spent on patient care,” said Radolec. “So the idea of investing in security is really tough from a budget standpoint…they’re choosing between a new MRI machine or better antivirus, backups, or data security.”
Because of the wealth of private data and healthcare information they maintain, hospitals are considered “high impact” for cybercriminals. Attackers know that if they get a foothold in a hospital, it’s more likely to pay — and pay quickly, Radolec told this news organization. Hospitals are also likely to have cyber insurance to help cover the cost of having their data stolen, encrypted, and ransomed.
The 2024 Microsoft Digital Defense Report also found that the bad actors are more sophisticated and better resourced and can challenge even the best cybersecurity. Improved defenses may not be good enough, and the sheer volume of attacks must be met with effective deterrence and government solutions that impose consequences for cybercriminals.
Vulnerable Users
Whether through a phishing email or text, password attack, or web attack, “the moment a ‘threat actor’ gets into your institution and gets credentials ... that’s the Nirvana state of a threat actor,” warned Ryan Witt, chair of the healthcare customer advisory board and vice president of Industry Solutions at Proofpoint, a cybersecurity platform. “They have those credentials and will go into deep reconnaissance mode. It often takes healthcare up to 6 months to even ascertain whether somebody’s actually in the network.” During that time, the hacker is learning how the institution works, what job functions matter, and how best to plan their attack.
“Attackers are getting in because they’re buying databases of usernames and passwords. And they’re trying them by the millions,” added Radolec. “For a sophisticated actor, all it takes is time and motivation. They have the skills. It’s just a matter of how persistent they want to be.”
Certain hospital staff are also more likely to be targeted by cyberhackers than others. “About 10% of a healthcare organization’s user base is much more vulnerable for all sorts of reasons — how they work, the value of their job title and job function, and therefore their access to systems,” said Witt.
High-profile staff are more likely to be targeted than those in lower-level positions; the so-called “CEO attack” is typical. However, staff in other hospital departments are also subject to cybercriminals, including hospice departments/hospice organizations and research arms of hospitals.
The Impact of Cyberattacks on Patients
Physicians and healthcare execs may have considered cybersecurity more of a compliance issue than a true threat to patients in the past. But this attitude is rapidly changing. “We are starting to see a very clear connection between a cyber event and how it can impact patient care and patient safety,” said Witt.
According to the Proofpoint report, cyber breaches can severely affect patient care. In 2024:
- 56% of respondents saw a delay in patient tests/procedures
- 53% experienced increased patient complications from medical procedures
- 52% noted a longer patient length of stay
- 44% saw an increase in patient transfers to other facilities
- 28% had an increase in mortality rate
What Hospitals and Physicians Can Do
Fortunately, hospitals can take measures to better protect their data and their patients. One strategy is segmenting networks to reduce the amount of data or systems one person or system can access. Educating staff about the dangers of phishing and spoofing emails also help protect organizations from ransomware attacks. Having staff avoid reusing passwords and updating logins and passwords frequently helps.
Most hospitals also need more robust security controls. Physicians and healthcare facilities must also embrace the cybersecurity controls found in other industries, said Witt. “Multifactor authentication is one of those things that can cause us frustration,” he said. “The controls can seem onerous, but they’re really valuable overall…and should become standard practice.”
Doctors can also prepare for a ransomware attack and protect patients by practicing some “old-school” medicine, like using paper systems and maintaining good patient notes — often, those notes are synced locally as well as offsite, so you’d be able to access them even during a data breach. “It’s smart to write prescriptions on pads sometimes,” said Radolec. “Don’t forget how to do those things because that will make you more resilient in the event of a ransomware attack.”
A Continuing Threat
Cyberattacks will continue. “When you look at the high likelihood [of success] and the soft target, you end up with ... a perfect storm,” said Radolec. “Hospitals have a lot of vulnerabilities. They have to keep operations going just to receive income, but also to deliver care to people.”
That means that the burden is on healthcare organizations — including physicians, nurses, staff, and C-level execs — to help keep the “security” in cybersecurity. “We are all part of the cybersecurity defense,” said Witt. Helping to maintain that defense has become a critical aspect of caring for patients.
A version of this article first appeared on Medscape.com.
On September 27, 2024, UMC Health System in Lubbock, Texas, experienced an IT outage because of a cybersecurity incident that temporarily diverted patients to other healthcare facilities. So far, in 2024, there have been 386 cyberattacks on healthcare organizations. These high-impact ransomware attacks disrupt and delay patient care.
In recent years, many healthcare systems, including Scripps Health, Universal Health Services, Vastaamo, Sky Lakes, and the University of Vermont, have paid millions — even tens of millions — to recover data after a cyberattack or data breach. When healthcare systems come under cyber fire, the impact extends far past disrupting workflows and compromising data, patient safety can be also be compromised, vital information may be lost, and imaging and lab results can go missing or be held for ransom, making physicians’ job difficult or impossible.
In fact, cyberattacks on hospitals are far more common than you may realize. A new report issued by Ponemon and Proofpoint found that 92% of healthcare organizations have experienced a cyberattack in the past 12 months. Even more sobering is that about half of the organizations affected suffered disruptions in patient care.
Healthcare Systems = ‘Soft Targets’
Healthcare systems are a “soft target” for hackers for several reasons, pointed out Matthew Radolec, vice president, incident response and cloud operations at Varonis, a data security company. “One, they’re usually an amalgamation of many healthcare systems that are interconnected,” said Radolec. “A lot of hospitals are connected to other hospitals or connected to educational institutions, which means their computer vulnerabilities are shared ... and if they have an issue, it could very easily spread to your network.”
Another factor is the cost of securing data. “[With hospitals], they’ll say that a dollar spent on security is a dollar not spent on patient care,” said Radolec. “So the idea of investing in security is really tough from a budget standpoint…they’re choosing between a new MRI machine or better antivirus, backups, or data security.”
Because of the wealth of private data and healthcare information they maintain, hospitals are considered “high impact” for cybercriminals. Attackers know that if they get a foothold in a hospital, it’s more likely to pay — and pay quickly, Radolec told this news organization. Hospitals are also likely to have cyber insurance to help cover the cost of having their data stolen, encrypted, and ransomed.
The 2024 Microsoft Digital Defense Report also found that the bad actors are more sophisticated and better resourced and can challenge even the best cybersecurity. Improved defenses may not be good enough, and the sheer volume of attacks must be met with effective deterrence and government solutions that impose consequences for cybercriminals.
Vulnerable Users
Whether through a phishing email or text, password attack, or web attack, “the moment a ‘threat actor’ gets into your institution and gets credentials ... that’s the Nirvana state of a threat actor,” warned Ryan Witt, chair of the healthcare customer advisory board and vice president of Industry Solutions at Proofpoint, a cybersecurity platform. “They have those credentials and will go into deep reconnaissance mode. It often takes healthcare up to 6 months to even ascertain whether somebody’s actually in the network.” During that time, the hacker is learning how the institution works, what job functions matter, and how best to plan their attack.
“Attackers are getting in because they’re buying databases of usernames and passwords. And they’re trying them by the millions,” added Radolec. “For a sophisticated actor, all it takes is time and motivation. They have the skills. It’s just a matter of how persistent they want to be.”
Certain hospital staff are also more likely to be targeted by cyberhackers than others. “About 10% of a healthcare organization’s user base is much more vulnerable for all sorts of reasons — how they work, the value of their job title and job function, and therefore their access to systems,” said Witt.
High-profile staff are more likely to be targeted than those in lower-level positions; the so-called “CEO attack” is typical. However, staff in other hospital departments are also subject to cybercriminals, including hospice departments/hospice organizations and research arms of hospitals.
The Impact of Cyberattacks on Patients
Physicians and healthcare execs may have considered cybersecurity more of a compliance issue than a true threat to patients in the past. But this attitude is rapidly changing. “We are starting to see a very clear connection between a cyber event and how it can impact patient care and patient safety,” said Witt.
According to the Proofpoint report, cyber breaches can severely affect patient care. In 2024:
- 56% of respondents saw a delay in patient tests/procedures
- 53% experienced increased patient complications from medical procedures
- 52% noted a longer patient length of stay
- 44% saw an increase in patient transfers to other facilities
- 28% had an increase in mortality rate
What Hospitals and Physicians Can Do
Fortunately, hospitals can take measures to better protect their data and their patients. One strategy is segmenting networks to reduce the amount of data or systems one person or system can access. Educating staff about the dangers of phishing and spoofing emails also help protect organizations from ransomware attacks. Having staff avoid reusing passwords and updating logins and passwords frequently helps.
Most hospitals also need more robust security controls. Physicians and healthcare facilities must also embrace the cybersecurity controls found in other industries, said Witt. “Multifactor authentication is one of those things that can cause us frustration,” he said. “The controls can seem onerous, but they’re really valuable overall…and should become standard practice.”
Doctors can also prepare for a ransomware attack and protect patients by practicing some “old-school” medicine, like using paper systems and maintaining good patient notes — often, those notes are synced locally as well as offsite, so you’d be able to access them even during a data breach. “It’s smart to write prescriptions on pads sometimes,” said Radolec. “Don’t forget how to do those things because that will make you more resilient in the event of a ransomware attack.”
A Continuing Threat
Cyberattacks will continue. “When you look at the high likelihood [of success] and the soft target, you end up with ... a perfect storm,” said Radolec. “Hospitals have a lot of vulnerabilities. They have to keep operations going just to receive income, but also to deliver care to people.”
That means that the burden is on healthcare organizations — including physicians, nurses, staff, and C-level execs — to help keep the “security” in cybersecurity. “We are all part of the cybersecurity defense,” said Witt. Helping to maintain that defense has become a critical aspect of caring for patients.
A version of this article first appeared on Medscape.com.
On September 27, 2024, UMC Health System in Lubbock, Texas, experienced an IT outage because of a cybersecurity incident that temporarily diverted patients to other healthcare facilities. So far, in 2024, there have been 386 cyberattacks on healthcare organizations. These high-impact ransomware attacks disrupt and delay patient care.
In recent years, many healthcare systems, including Scripps Health, Universal Health Services, Vastaamo, Sky Lakes, and the University of Vermont, have paid millions — even tens of millions — to recover data after a cyberattack or data breach. When healthcare systems come under cyber fire, the impact extends far past disrupting workflows and compromising data, patient safety can be also be compromised, vital information may be lost, and imaging and lab results can go missing or be held for ransom, making physicians’ job difficult or impossible.
In fact, cyberattacks on hospitals are far more common than you may realize. A new report issued by Ponemon and Proofpoint found that 92% of healthcare organizations have experienced a cyberattack in the past 12 months. Even more sobering is that about half of the organizations affected suffered disruptions in patient care.
Healthcare Systems = ‘Soft Targets’
Healthcare systems are a “soft target” for hackers for several reasons, pointed out Matthew Radolec, vice president, incident response and cloud operations at Varonis, a data security company. “One, they’re usually an amalgamation of many healthcare systems that are interconnected,” said Radolec. “A lot of hospitals are connected to other hospitals or connected to educational institutions, which means their computer vulnerabilities are shared ... and if they have an issue, it could very easily spread to your network.”
Another factor is the cost of securing data. “[With hospitals], they’ll say that a dollar spent on security is a dollar not spent on patient care,” said Radolec. “So the idea of investing in security is really tough from a budget standpoint…they’re choosing between a new MRI machine or better antivirus, backups, or data security.”
Because of the wealth of private data and healthcare information they maintain, hospitals are considered “high impact” for cybercriminals. Attackers know that if they get a foothold in a hospital, it’s more likely to pay — and pay quickly, Radolec told this news organization. Hospitals are also likely to have cyber insurance to help cover the cost of having their data stolen, encrypted, and ransomed.
The 2024 Microsoft Digital Defense Report also found that the bad actors are more sophisticated and better resourced and can challenge even the best cybersecurity. Improved defenses may not be good enough, and the sheer volume of attacks must be met with effective deterrence and government solutions that impose consequences for cybercriminals.
Vulnerable Users
Whether through a phishing email or text, password attack, or web attack, “the moment a ‘threat actor’ gets into your institution and gets credentials ... that’s the Nirvana state of a threat actor,” warned Ryan Witt, chair of the healthcare customer advisory board and vice president of Industry Solutions at Proofpoint, a cybersecurity platform. “They have those credentials and will go into deep reconnaissance mode. It often takes healthcare up to 6 months to even ascertain whether somebody’s actually in the network.” During that time, the hacker is learning how the institution works, what job functions matter, and how best to plan their attack.
“Attackers are getting in because they’re buying databases of usernames and passwords. And they’re trying them by the millions,” added Radolec. “For a sophisticated actor, all it takes is time and motivation. They have the skills. It’s just a matter of how persistent they want to be.”
Certain hospital staff are also more likely to be targeted by cyberhackers than others. “About 10% of a healthcare organization’s user base is much more vulnerable for all sorts of reasons — how they work, the value of their job title and job function, and therefore their access to systems,” said Witt.
High-profile staff are more likely to be targeted than those in lower-level positions; the so-called “CEO attack” is typical. However, staff in other hospital departments are also subject to cybercriminals, including hospice departments/hospice organizations and research arms of hospitals.
The Impact of Cyberattacks on Patients
Physicians and healthcare execs may have considered cybersecurity more of a compliance issue than a true threat to patients in the past. But this attitude is rapidly changing. “We are starting to see a very clear connection between a cyber event and how it can impact patient care and patient safety,” said Witt.
According to the Proofpoint report, cyber breaches can severely affect patient care. In 2024:
- 56% of respondents saw a delay in patient tests/procedures
- 53% experienced increased patient complications from medical procedures
- 52% noted a longer patient length of stay
- 44% saw an increase in patient transfers to other facilities
- 28% had an increase in mortality rate
What Hospitals and Physicians Can Do
Fortunately, hospitals can take measures to better protect their data and their patients. One strategy is segmenting networks to reduce the amount of data or systems one person or system can access. Educating staff about the dangers of phishing and spoofing emails also help protect organizations from ransomware attacks. Having staff avoid reusing passwords and updating logins and passwords frequently helps.
Most hospitals also need more robust security controls. Physicians and healthcare facilities must also embrace the cybersecurity controls found in other industries, said Witt. “Multifactor authentication is one of those things that can cause us frustration,” he said. “The controls can seem onerous, but they’re really valuable overall…and should become standard practice.”
Doctors can also prepare for a ransomware attack and protect patients by practicing some “old-school” medicine, like using paper systems and maintaining good patient notes — often, those notes are synced locally as well as offsite, so you’d be able to access them even during a data breach. “It’s smart to write prescriptions on pads sometimes,” said Radolec. “Don’t forget how to do those things because that will make you more resilient in the event of a ransomware attack.”
A Continuing Threat
Cyberattacks will continue. “When you look at the high likelihood [of success] and the soft target, you end up with ... a perfect storm,” said Radolec. “Hospitals have a lot of vulnerabilities. They have to keep operations going just to receive income, but also to deliver care to people.”
That means that the burden is on healthcare organizations — including physicians, nurses, staff, and C-level execs — to help keep the “security” in cybersecurity. “We are all part of the cybersecurity defense,” said Witt. Helping to maintain that defense has become a critical aspect of caring for patients.
A version of this article first appeared on Medscape.com.
Disparities in Skin Cancer Outcomes in the Latine/Hispanic Population
The Latine/Hispanic population in the United States comprises one of the largest and youngest skin of color communities.1,2 In 2020, this group accounted for 19% of all Americans—a percentage expected to increase to more than 25% by 2060.3
It must be emphasized that the Latine/Hispanic community in the United States is incredibly diverse.4 Approximately one-third of individuals in this group are foreign-born, and this community is made up of people from all racialized groups, religions, languages, and cultural identities.2 The heterogeneity of the Latine/Hispanic population translates into a wide representation of skin tones, reflecting a rich range of ancestries, ethnicities, and cultures. The percentage of individuals from each origin group may differ according to where they live in the United States; for instance, individuals who identify as Mexican comprise more than 80% of the Latine/Hispanic population in both Texas and California but only 17% in Florida, where more than half of Latine/Hispanic people identify as Cuban or Puerto Rican.4,5 As a result, when it comes to skin cancer epidemiology, variations in incidence and mortality may exist within each of these subgroups who identify as part of the Latine/Hispanic community, as reported for other cancers.6,7 Further research is needed to investigate these potential differences.Unfortunately, considerable health disparities persist among this rapidly growing population, including increased morbidity and mortality from melanoma and keratinocyte carcinomas (KCs) despite overall low lifetime incidence.8,9 In this review, the epidemiology, clinical manifestation, and ethnic disparities for skin cancer among the US Latine/Hispanic population are summarized; other factors impacting overall health and health care, including sociocultural factors, also are briefly discussed.
Terminology
Before a meaningful dialogue can be had about skin cancer in the Latine/Hispanic population, it is important to contextualize the terms used to identify this patient population, including Latino/Latine and Hispanic. In the early 1970s, the United States adopted the term Hispanic as a way of conglomerating Spanish-speaking individuals from Spain, the Caribbean, and Central and South America. The goal was to implement a common identifier that enabled the US government to study the economic and social development of these groups.10 Nevertheless, considerable differences (eg, variations in skin pigmentation, sun sensitivity) exist among Hispanic communities, with some having stronger European, African, or Amerindian influences due to colonization of their distinct countries.11
In contrast, Latino is a geographic term and refers to people with roots in Latin America and the Caribbean (Table 1).12,13 For example, a person from Brazil may be considered Latino but not Hispanic as Brazilians speak Portuguese; alternatively, Spaniards (who are considered Hispanic) are not Latino because Spain is not a Latin American country. A person from Mexico would be considered both Latino and Hispanic.13

More recently, the term Latine has been introduced as an alternative to the gender binary inherent in the Spanish language.12 For the purposes of this article, the terms Latine and Hispanic will be used interchangeably (unless otherwise specified) depending on how they are cited in the existing literature. Furthermore, the term non-Hispanic White (NHW) will be used to refer to individuals who have been socially ascribed or who self-identify as White in terms of race or ethnicity.
Melanoma
Melanoma, the deadliest form of skin cancer, is more likely to metastasize compared to other forms of skin cancer, including basal cell carcinoma (BCC) and squamous cell carcinoma (SCC). For Latine/Hispanic individuals living in the United States, the lifetime risk for melanoma is 1 in 200 compared to 1 in 33 for NHW individuals.14 While the lifetime risk for melanoma is low for the Latine/Hispanic population, Hispanic individuals are diagnosed with melanoma at an earlier age (mean, 56 years), and the rate of new cases is marginally higher for women (4.9 per 100,000) compared to men (4.8 per 100,000).15,16
Typical sites of melanoma manifestation in Latine/Hispanic individuals include the torso (most common site in Hispanic men), lower extremities (most common site in Hispanic women), and acral sites (palms, soles, and nails).9,16,17 Anatomic location also can vary according to age for both men and women. For men, the incidence of melanoma on the trunk appears to decrease with age, while the incidence on the head and neck may increase. For women, the incidence of melanoma on the lower extremities and hip increases with age. Cutaneous melanoma may manifest as a lesion with asymmetry, irregular borders, variation in pigmentation, large diameter (>6 mm), and evolution over time. In patients with skin of color, melanoma easily can be missed, as it also typically mimics more benign skin conditions and may develop from an existing black- or dark brown–pigmented macule.18 The most common histologic subtype reported among Latine/Hispanic individuals in the United States is superficial spreading melanoma (20%–23%) followed by nodular melanoma and acral lentiginous melanoma.16,19 Until additional risk factors associated with melanoma susceptibility in Hispanic/Latine people are better elucidated, it may be appropriate to use an alternative acronym, such as CUBED (Table 2), in addition to the standard ABCDE system to help recognize potential melanoma on acral sites.18

Although the lifetime risk for melanoma among Hispanic individuals in the United States is lower than that for NHW individuals, Hispanic patients who are diagnosed with melanoma are more likely to present with increased tumor thickness and later-stage diagnosis compared to NHW individuals.8,16,20 In a recent study by Qian et al,8 advanced stage melanoma—defined as regional or distant stage disease—was present in 12.6% of NHW individuals. In contrast, the percentage of Hispanics with advanced disease was higher at 21%.8 Even after controlling for insurance and poverty status, Hispanic individuals were at greater risk than NHW individuals for late-stage diagnosis.16,20
Morbidity and mortality also have been shown to be higher in Hispanic patients with cutaneous melanoma.9,17 Reasons for this are multifactorial, with studies specific to melanoma citing challenges associated with early detection in individuals with deeply pigmented skin, a lack of awareness and knowledge about skin cancer among Latine/Hispanic patients, and treatment disparities.21-23 Moreover, very few studies have reported comprehensive data on patients from Africa and Latin America. Studies examining the role of genetic ancestry, epigenetic variants, and skin pigmentation and the risk for melanoma among the Latine/Hispanic population therefore are much needed.24
Keratinocyte Carcinomas
Keratinocyte carcinomas, also known as nonmelanoma skin cancers, include BCC and SCC. In comparison to the high-quality data available for melanoma from cancer registries, there are less reliable incidence data for KCs, especially among individuals with skin of color.25 As a result, KC epidemiology in the United States is drawn largely from case series (especially for individuals with skin of color) or claims data from small data sets often from geographically restricted regions within the United States.25,26
Basal Cell Carcinoma—Basal cell carcinoma is the most common malignant skin cancer in Latine/Hispanic individuals. Among those with lighter skin tones, the lifetime risk for BCC is about 30%.27,28 Men typically are affected at a higher rate than women, and the median age for diagnosis is 68 years.29 The development of BCC primarily is linked to lifetime accumulated UV radiation exposure. Even though BCC has a low mortality rate, it can lead to substantial morbidity due to factors such as tumor location, size, and rate of invasion, resulting in cosmetic and functional issues. Given its low metastatic potential, treatment of BCC typically is aimed at local control.30 Options for treatment include Mohs micrographic surgery (MMS), curettage and electrodessication, cryosurgery, photodynamic therapy, radiation therapy, and topical therapies. Systemic therapies are reserved for patients with locally advanced or metastatic disease.30
Latine/Hispanic patients characteristically present with BCCs on sun-exposed areas of the skin such as the head and neck region. In most patients, BCC manifests as a translucent pearly nodule with superficial telangiectasias and/or a nonhealing ulcer with a central depression and rolled nontender borders. However, in patients with skin of color, 66% of BCCs manifest with pigmentation; in fact, pigmented BCC (a subtype of BCC) has been shown to have a higher prevalence among Hispanic individuals, with an incidence twice as frequent as in NHW individuals.31 In addition, there are reports of increased tendency among Latine/Hispanic individuals to develop multiple BCCs.32,33
The relationship between UV exposure and KCs could explain the relatively higher incidence in populations with skin of color living in warmer climates, including Hispanic individuals.34 Even so, the development of BCCs appears to correlate directly with the degree of pigmentation in the skin, as it is most common in individuals with lighter skin tones within the Hispanic population.25,34,35 Other risk factors associated with BCC development include albinism, arsenic ingestion, chronic infections, immunosuppression, history of radiation treatment, and history of scars or ulcers due to physical/thermal trauma.35-37
Squamous Cell Carcinoma—Squamous cell carcinoma is the second most common skin cancer among Latine/Hispanic patients. In contrast with NHW patients, evidence supporting the role of UV exposure as a primary risk factor for SCC in patients with skin of color remains limited.25,38 Reports linking UV exposure and KCs in Hispanic and Black individuals predominantly include case series or population-based studies that do not consider levels of UV exposure.25
More recently, genetic ancestry analyses of a large multiethnic cohort found an increased risk for cutaneous SCC among Latine/Hispanic individuals with European ancestry compared to those with Native American or African ancestry; however, these genetic ancestry associations were attenuated (although not eliminated) after considering skin pigmentation (using loci associated with skin pigmentation), history of sun exposure (using actinic keratoses as a covariate for chronic sun exposure), and sun-protected vs sun-exposed anatomic sites, supporting the role of other environmental or sociocultural factors in the development of SCC.39 Similar to BCCs, immunosuppression, chronic scarring, skin irritation, and inflammatory disease also are documented risk factors.9,32
Among NHW individuals with lighter skin tones, SCC characteristically manifests on sun-exposed areas of the skin such as the head and neck region. Typically, a lesion may appear as a scaly erythematous papule or plaque that may be verrucous in nature or a nonhealing bleeding ulcer. In patients with more deeply pigmented skin, SCC tends to develop in the perianal region and on the penis and lower legs; pigmented lesions also may be present (as commonly reported in BCCs).9,32,36
Unfortunately, the lower incidence of KCs and lack of surveillance in populations with skin of color result in a low index of clinical suspicion, leading to delayed diagnoses and increased morbidity.40 Keratinocyte carcinomas are more costly to treat and require more health care resources for Latine/Hispanic and Black patients compared to their NHW counterparts; for example, KCs are associated with more ambulatory visits, more prescription medications, and greater cost on a per-person, per-year basis in Latine/Hispanic and Black patients compared with NHW patients.41 Moreover, a recent multicenter retrospective study found Hispanic patients had 17% larger MMS defects following treatment for KCs compared to NHW patients after adjustment for age, sex, and insurance type.42
Hispanic patients tend to present initially with SCCs in areas associated with advanced disease, such as the anogenital region, penis, and the lower extremities. Latine and Black men have the highest incidence of penile SCC, which is rare with high morbidity and mortality.32,43,44 The higher incidence of penile SCC among Hispanic individuals living in southern states could correspond to circumcision or HPV infection rates,44 ultimately impacting incidence.45
Dermatofibrosarcoma Protuberans
Dermatofibrosarcoma protuberans (DFSP) is a rare locally aggressive cutaneous sarcoma. According to population studies, overall incidence of DFSP is around 4.1 to 4.2 per million in the United States. Population-based studies on DFSP are limited, but available data suggest that Black patients as well as women have the highest incidence.46
Dermatofibrosarcoma protuberans is characterized by its capacity to invade surrounding tissues in a tentaclelike pattern.47 This characteristic often leads to inadequate initial resection of the lesion as well as a high recurrence rate despite its low metastatic potential.48 In early stages, DFSP typically manifests as an asymptomatic plaque with a slow growth rate. The color of the lesion ranges from reddish brown to flesh colored. The pigmented form of DFSP, known as Bednar tumor, is the most common among Black patients.47 As the tumor grows, it tends to become firm and nodular. The most common location for
Although current guidelines designate MMS as the first-line treatment for DFSP, the procedure may be inaccessible for certain populations.49 Patients with skin of color are more likely to undergo wide local excision (WLE) than MMS; however, WLE is less effective, with a recurrence rate of 30% compared with 3% in those treated with MMS.50 A retrospective cohort study of more than 2000 patients revealed that Hispanic and Black patients were less likely to undergo MMS. In addition, the authors noted that WLE recipients more commonly were deceased at the end of the study.51
Despite undergoing treatment for a primary DFSP, Hispanic patients also appear to be at increased risk for a second surgery.52 Additional studies are needed to elucidate the reasons behind higher recurrence rates in Latine/Hispanic patients compared to NHW individuals.
Factors Influencing Skin Cancer Outcomes
In recent years, racial and ethnic disparities in health care use, medical treatment, and quality of care among minoritized populations (including Latine/Hispanic groups) have been documented in the medical literature.53,54 These systemic inequities, which are rooted in structural racism,55 have contributed to poorer health outcomes, worse health status, and lower-quality care for minoritized patients living in the United States, including those impacted by dermatologic conditions.8,43,55-57 Becoming familiar with the sociocultural factors influencing skin cancer outcomes in the Latine/Hispanic community (including the lack of or inadequate health insurance, medical mistrust, language, and other cultural elements) and the paucity of research in this domain could help eliminate existing health inequities in this population.
Health Insurance Coverage—Although the uninsured rates in the Latine population have decreased since the passage of the Affordable Care Act (from 30% in 2013 to a low of 19% in 2017),58 inadequate health insurance coverage remains one of the largest barriers to health care access and a contributor to health disparities among the Latine community. Nearly 1 in 5 Latine individuals in the United States are uninsured compared to 8% of NHW individuals.58 Even though Latine individuals are more likely than non-Latine individuals to be part of the workforce, Latine employees are less likely to receive employer-sponsored coverage (27% vs 53% for NHW individuals).59
Not surprisingly, noncitizens are far less likely to be insured; this includes lawfully present immigrants (ie, permanent residents or green card holders, refugees, asylees, and others who are authorized to live in the United States temporarily or permanently) and undocumented immigrants (including individuals who entered the country without authorization and individuals who entered the country lawfully and stayed after their visa or status expired). The higher uninsured rate among noncitizens reflects not only limited access to employer-sponsored coverage but includes immigrant eligibility restrictions for federal programs such as Medicaid, the Children’s Health Insurance Program, and the Affordable Care Act Marketplace coverage.60
With approximately 9 million Americans living in mixed-status families (and nearly 10% of babies born each year with at least one undocumented parent), restrictive federal or state health care policies may extend beyond their stated target and impact both Latine citizens and noncitizens.61-65 For instance, Vargas et al64 found that both Latine citizens and noncitizens who lived in states with a high number of immigration-related laws had decreased odds of reporting optimal health as compared to Latine respondents in states with fewer immigration-related laws. Other barriers to enrollment include fears and confusion about program qualification, even if eligible.58
Medical Mistrust and Unfamiliarity—Mistrust of medical professionals has been shown to reduce patient adherence to treatment as prescribed by their medical provider and can negatively influence health outcomes.53 For racial/ethnic minoritized groups (including Latine/Hispanic patients), medical mistrust may be rooted in patients’ experience of discrimination in the health care setting. In a recent cross-sectional study, results from a survey of California adults (including 704 non-Hispanic Black, 711 Hispanic, and 913 NHW adults) found links between levels of medical mistrust and perceived discrimination based on race/ethnicity and language as well as perceived discrimination due to income level and type or lack of insurance.53 Interestingly, discrimination attributed to income level and insurance status remained after controlling for race/ethnicity and language. As expected, patients reliant on public insurance programs such as Medicare have been reported to have greater medical mistrust and suspicion compared with private insurance holders.65 Together, these findings support the notion that individuals who have low socioeconomic status and lack insurance coverage—disproportionately historically marginalized populations—are more likely to perceive discrimination in health care settings, have greater medical mistrust, and experience poorer health outcomes.53
It also is important for health care providers to consider that the US health care system is unfamiliar to many Latine/Hispanic individuals. Costs of medical services tend to be substantially higher in the United States, which can contribute to mistrust in the system.66 In addition, unethical medical experimentations have negatively affected both Latine and especially non-Hispanic Black populations, with long-lasting perceptions of deception and exploitation.67 These beliefs have undermined the trust that these populations have in clinicians and the health care system.54,67
Language and Other Cultural Elements—The inability to effectively communicate with health care providers could contribute to disparities in access to and use of health care services among Latine/Hispanic individuals. In a Medical Expenditure Panel Survey analysis, half of Hispanic patients with limited comfort speaking English did not have a usual source of care, and almost 90% of those with a usual source of care had a provider who spoke Spanish or used interpreters—indicating that few Hispanic individuals with limited comfort speaking English selected a usual source of care without language assistance.68,69 In other examples, language barriers contributed to disparities in cancer screening, and individuals with limited English proficiency were more likely to have difficulty understanding their physician due to language barriers.68,70
Improving cultural misconceptions regarding skin conditions, especially skin cancer, is another important consideration in the Latine/Hispanic community. Many Latine/Hispanic individuals wrongly believe they cannot develop skin cancer due to their darker skin tones and lack of family history.26 Moreover, multiple studies assessing melanoma knowledge and perception among participants with skin of color (including one with an equal number of Latine/Hispanic, Black/African American, and Asian individuals for a total of 120 participants) revealed that many were unaware of the risk for melanoma on acral sites.71 Participants expressed a need for more culturally relevant content from both clinicians and public materials (eg, images of acral melanoma in a person with skin of color).71-73
Paucity of Research—There is limited research regarding skin cancer risks and methods of prevention for patients with skin of color, including the Latine/Hispanic population. Efforts to engage and include patients from these communities, as well as clinicians or investigators from similar backgrounds, in clinical studies are desperately needed. It also is important that clinical studies collect data beyond population descriptors to account for both clinical and genetic variations observed in the Latine/Hispanic population.
Latine/Hispanic individuals are quite diverse with many variable factors that may influence skin cancer outcomes. Often, cancer surveillance data are available in aggregate only, which could mask this heterogeneity.74 Rigorous studies that collect more granular data, including objective measures of skin pigmentation beyond self-reported Fitzpatrick skin type, culture/beliefs, lifestyle/behavior, geographic location, socioeconomic status, genetics, or epigenetics could help fully elucidate skin cancer risks and mitigate health disparities among individuals who identify as part of this population.
Final Thoughts
The Latine/Hispanic community—the largest ethnic minoritized group in the United States—is disproportionately affected by dermatologic health disparities. We hope this review helps to increase recognition of the clinical manifestations of skin cancer in Latine/Hispanic patients. Other factors that may impact skin cancer outcomes in this population include (but are not limited to) lack of or inadequate health insurance, medical mistrust, linguistic barriers and/or individual/cultural perspectives, along with limited research. Recognizing and addressing these (albeit complex) barriers that contribute to the inequitable access to health care in this population remains a critical step toward improving skin cancer outcomes.
- Noe-Bustamnate L, Lopez MH, Krogstad JM. US Hispanic population surpassed 60 million in 2019, but growth has slowed. July 7, 2020. Accessed September 3, 2024. https://www.pewresearch.org/short-reads/2020/07/07/u-s-hispanic-population-surpassed-60-million-in-2019-but-growth-has-slowed/
- Frank C, Lopez MH. Hispanic Americans’ trust in and engagement with science. Pew Research Center. June 14, 2022. Accessed September 3, 2024. https://www.pewresearch.org/wp-content/uploads/sites/20/2022/06/PS_2022.06.14_hispanic-americans-science_REPORT.pdf
- US Census Bureau. Projections of the size and composition of the US population: 2014 to 2060. US Government Printing Office; 2015. Accessed September 5, 2024. https://www.census.gov/content/dam/Census/library/publications/2015/demo/p25-1143.pdf
- Zong J. A mosaic, not a monolith: a profile of the U.S. Latino population, 2000-2020. October 26, 2022. Accessed September 3, 2024. https://latino.ucla.edu/research/latino-population-2000-2020/
- Latinos in California, Texas, New York, Florida and New Jersey. Pew Research Center. March 19, 2004. Accessed September 3, 2024. https://www.pewresearch.org/hispanic/2004/03/19/latinos-in-california-texas-new-york-florida-and-new-jersey/
- Pinheiro PS, Sherman RL, Trapido EJ, et al. Cancer incidence in first generation US Hispanics: Cubans, Mexicans, Puerto Ricans, and new Latinos. Cancer Epidemiol Biomarkers Prev. 2009;18:2162-2169.
- Pinheiro PS, Callahan KE, Kobetz EN. Disaggregated Hispanic groups and cancer: importance, methodology, and current knowledge. In: Ramirez AG, Trapido EJ, eds. Advancing the Science of Cancer in Latinos. Springer; 2020:17-34.
- Qian Y, Johannet P, Sawyers A, et al. The ongoing racial disparities in melanoma: an analysis of the Surveillance, Epidemiology, and End Results database (1975-2016). J Am Acad Dermatol. 2021;84:1585-1593.
- Hogue L, Harvey VM. Basal cell carcinoma, squamous cell carcinoma, and cutaneous melanoma in skin of color patients. Dermatol Clin. 2019;37:519-526.
- Cruzval-O’Reilly E, Lugo-Somolinos A. Melanoma in Hispanics: we may have it all wrong. Cutis. 2020;106:28-30.
- Borrell LN, Elhawary JR, Fuentes-Afflick E, et al. Race and genetic ancestry in medicine—a time for reckoning with racism. N Engl J Med. 2021;384:474-480.
- Lopez MH, Krogstad JM, Passel JS. Who is Hispanic? September 5, 2023. Accessed September 3, 2024. https://www.pewresearch.org/short-reads/2023/09/05/who-is-hispanic/
- Carrasquillo OY, Lambert J, Merritt BG. Comment on “Disparities in nonmelanoma skin cancer in Hispanic/Latino patients based on Mohs micrographic surgery defect size: a multicenter retrospective study.”J Am Acad Dermatol. 2022;87:E129-E130.
- American Cancer Society. Key statistics for melanoma skin cancer. Updated January 17, 2024. Accessed September 3, 2024. https://www.cancer.org/cancer/types/melanoma-skin-cancer/about/key-statistics.html
- National Cancer Institute. Melanoma of the skin: recent trends in SEER age-adjusted incidence rates, 2000-2021. Updated June 27, 2024. Accessed September 3, 2024. https://seer.cancer.gov/statistics-network/explorer/application.htmlsite=53&data_type=1&graph_type=2&compareBy=sex&chk_sex_3=3&chk_sex_2=2&rate_type=2&race=6&age_range=1&stage=101&advopt_precision=1&advopt_show_ci=on&hdn_view=0&advopt_display=2
- Garnett E, Townsend J, Steele B, et al. Characteristics, rates, and trends of melanoma incidence among Hispanics in the USA. Cancer Causes Control. 2016;27:647-659.
- Higgins S, Nazemi A, Feinstein S, et al. Clinical presentations of melanoma in African Americans, Hispanics, and Asians. Dermatol Surg. 2019;45:791-801.
- Bristow IR, de Berker DA, Acland KM, et al. Clinical guidelines for the recognition of melanoma of the foot and nail unit. J Foot Ankle Res. 2010;3:25.
- Fernandez JM, Mata EM, Behbahani S, et al. Survival of Hispanic patients with cutaneous melanoma: a retrospective cohort analysis of 6016 cases from the National Cancer Database. J Am Acad Dermatol. 2023;88:1135-1138.
- Hu S, Sherman R, Arheart K, et al. Predictors of neighborhood risk for late-stage melanoma: addressing disparities through spatial analysis and area-based measures. J Investigative Dermatol. 2014;134:937-945.
- Buster KJ, You Z, Fouad M, et al. Skin cancer risk perceptions: a comparison across ethnicity, age, education, gender, and income. J Am Acad Dermatol. 2012;66:771-779.
- Halpern MT, Ward EM, Pavluck AL, et al. Association of insurance status and ethnicity with cancer stage at diagnosis for 12 cancer sites: a retrospective analysis. Lancet Oncology. 2008;9:222-231.
- Weiss J, Kirsner RS, Hu S. Trends in primary skin cancer prevention among US Hispanics: a systematic review. J Drugs Dermatol. 2012;11:580-586.
- Carvalho LAD, Aguiar FC, Smalley KSM, et al. Acral melanoma: new insights into the immune and genomic landscape. Neoplasia. 2023;46:100947.
- Kolitz E, Lopes F, Arffa M, et al. UV Exposure and the risk of keratinocyte carcinoma in skin of color: a systematic review. JAMA Dermatol. 2022;158:542-546.
- Lukowiak TM, Aizman L, Perz A, et al. Association of age, sex, race, and geographic region with variation of the ratio of basal cell to cutaneous squamous cell carcinomas in the United States. JAMA Dermatol. 2020;156:1192-1198.
- Basset-Seguin N, Herms F. Update in the management of basal cell carcinoma. Acta Derm Venereol. 2020;100:adv00140.
- McDaniel B, Badri T, Steele RB. Basal cell carcinoma. StatPearls [Internet]. Updated March 13, 2024. Accessed September 3, 2024. https://www.ncbi.nlm.nih.gov/books/NBK482439/
- Dessinioti C, Antoniou C, Katsambas A, et al. Basal cell carcinoma: what’s new under the sun. Photochem Photobiol. 2010;86:481-491.
- Kim DP, Kus KJB, Ruiz E. Basal cell carcinoma review. Hematol Oncol Clin North Am. 2019;33:13-24.
- Bigler C, Feldman J, Hall E, et al. Pigmented basal cell carcinoma in Hispanics. J Am Acad Dermatol. 1996;34(5 pt 1):751-752.
- Higgins S, Nazemi A, Chow M, et al. Review of nonmelanoma skin cancer in African Americans, Hispanics, and Asians. Dermatol Surg. 2018;44:903-910.
- Byrd-Miles K, Toombs EL, Peck GL. Skin cancer in individuals of African, Asian, Latin-American, and American-Indian descent: differences in incidence, clinical presentation, and survival compared to Caucasians. J Drugs Dermatol. 2007;6:10-16.
- Rivas M, Rojas E, Calaf GM, et al. Association between non-melanoma and melanoma skin cancer rates, vitamin D and latitude. Oncol Lett. 2017;13:3787-3792.
- Bradford PT. Skin cancer in skin of color. Dermatol Nurs. 2009;21:170-177, 206.
- Davis DS, Robinson C, Callender VD. Skin cancer in women of color: epidemiology, pathogenesis and clinical manifestations. Int J Womens Dermatol. 2021;7:127-134.
- Maafs E, De la Barreda F, Delgado R, et al. Basal cell carcinoma of trunk and extremities. Int J Dermatol. 1997;36:622-628.
- Munjal A, Ferguson N. Skin cancer in skin of color. Dermatol Clin. 2023;41:481-489.
- Jorgenson E, Choquet H, Yin J, et al. Genetic ancestry, skin pigmentation, and the risk of cutaneous squamous cell carcinoma in Hispanic/Latino and non-Hispanic white populations. Commun Biol. 2020;3:765.
- Soliman YS, Mieczkowska K, Zhu TR, et al. Characterizing basal cell carcinoma in Hispanic individuals undergoing Mohs micrographic surgery: a 7-year retrospective review at an academic institution in the Bronx. Brit J Dermatol. 2022;187:597-599.
- Sierro TJ, Blumenthal LY, Hekmatjah J, et al. Differences in health care resource utilization and costs for keratinocyte carcinoma among racioethnic groups: a population-based study. J Am Acad Dermatol. 2022;86:373-378.
- Blumenthal LY, Arzeno J, Syder N, et al. Disparities in nonmelanoma skin cancer in Hispanic/Latino patients based on Mohs micrographic surgery defect size: a multicenter retrospective study. J Am Acad Dermatol. 2022;86:353-358.
- Slopnick EA, Kim SP, Kiechle JE, et al. Racial disparities differ for African Americans and Hispanics in the diagnosis and treatment of penile cancer. Urology. 2016;96:22-28.
- Goodman MT, Hernandez BY, Shvetsov YB. Demographic and pathologic differences in the incidence of invasive penile cancer in the United States, 1995-2003. Cancer Epidemiol Biomarkers Prev. 2007;16:1833-1839.
- Thompson EL, Rosen BL, Maness SB. Social determinants of health and human papillomavirus vaccination among young adults, National Health Interview Survey 2016. J Community Health. 2019;44:149-158.
- Hao X, Billings SD, Wu F, et al. Dermatofibrosarcoma protuberans: update on the diagnosis and treatment. J Clin Med. 2020;9:1752.
- Mosallaei D, Lee EB, Lobl M, et al. Rare cutaneous malignancies in skin of color. Dermatol Surg. 2022;48:606-612.
- Criscito MC, Martires KJ, Stein JA. Prognostic factors, treatment, and survival in dermatofibrosarcoma protuberans. JAMA Dermatol. 2016;152:1365-1371.
- Orenstein LAV, Nelson MM, Wolner Z, et al. Differences in outpatient dermatology encounter work relative value units and net payments by patient race, sex, and age. JAMA Dermatol. 2021;157:406-412.
- Lowe GC, Onajin O, Baum CL, et al. A comparison of Mohs micrographic surgery and wide local excision for treatment of dermatofibrosarcoma protuberans with long-term follow-up: the Mayo Clinic experience. Dermatol Surg. 2017;43:98-106.
- Moore KJ, Chang MS, Weiss J, et al. Racial and ethnic differences in the surgical treatment of dermatofibrosarcoma protuberans: a retrospective cohort analysis. J Am Acad Dermatol. 2022;87:245-247.
- Trofymenko O, Bordeaux JS, Zeitouni NC. Survival in patients with primary dermatofibrosarcoma protuberans: National Cancer Database analysis. J Am Acad Dermatol. 2018;78:1125-1134.
- Bazargan M, Cobb S, Assari S. Discrimination and medical mistrust in a racially and ethnically diverse sample of California adults. Ann Fam Med. 2021;19:4-15.
- Smedley BD, Stith AY, Nelson AR, eds. Unequal Treatment: Confronting Racial and Ethnic Disparities in Health Care. Washington, DC; 2003.
- Bailey ZD, Krieger N, Agenor M, et al. Structural racism and health inequities in the USA: evidence and interventions. Lancet. 2017;389:1453-1463.
- Tackett KJ, Jenkins F, Morrell DS, et al. Structural racism and its influence on the severity of atopic dermatitis in African American children. Pediatric Dermatol. 2020;37:142-146.
- Greif C, Srivastava D, Nijhawan RI. A retrospective cohort study of dermatofibrosarcoma protuberans at a large metropolitan academic center. JAAD Int. 2022;6:104-106.
- Office of the Assistant Secretary for Planning and Evaluation. Health insurance coverage and access to care among Latinos: recent rrends and key challenges (HP-2021-22). October 8, 2021. Accessed September 3, 2024. https://aspe.hhs.gov/reports/health-insurance-coverage-access-care-among-latinos
- Keisler-Starkey K, Bunch LN. Health insurance coverage in the United States: 2020 (Current Population Reports No. P60-274). US Census Bureau; 2021. https://www.census.gov/content/dam/Census/library/publications/2021/demo/p60-274.pdf
- Kaiser Family Foundation. Key facts on health coverage of immigrants. Updated June 26, 2024. Accessed September 3, 2024. https://www.kff.org/racial-equity-and-health-policy/fact-sheet/key-facts-on-health-coverage-of-immigrants/
- Pew Research Center. Unauthorized immigrants: length of residency, patterns of parenthood. Published December 1, 2011. Accessed October 28, 2024. https://www.pewresearch.org/race-and-ethnicity/2011/12/01/unauthorized-immigrants-length-of-residency-patterns-of-parenthood/
- Schneider J, Schmitt M. Understanding the relationship between racial discrimination and mental health among African American adults: a review. SAGE Open. 2015;5:1-10.
- Philbin MM, Flake M, Hatzenbuehler ML, et al. State-level immigration and immigrant-focused policies as drivers of Latino health disparities in the United States. Soc Sci Med. 2018;199:29-38.
- Vargas ED, Sanchez GR, Juarez M. The impact of punitive immigrant laws on the health of Latina/o Populations. Polit Policy. 2017;45:312-337.
- Sutton AL, He J, Edmonds MC, et al. Medical mistrust in Black breast cancer patients: acknowledging the roles of the trustor and the trustee. J Cancer Educ. 2019;34:600-607.
- Jacobs J. An overview of Latin American healthcare systems. Pacific Prime Latin America. July 31, 2023. Accessed September 3, 2024. https://www.pacificprime.lat/blog/an-overview-of-latin-american-healthcare-systems/
- CDC. Unfair and unjust practices and conditions harm Hispanic and Latino people and drive health disparities. May 15, 2024. Accessed September 3, 2024. https://www.cdc.gov/tobacco-health-equity/collection/hispanic-latino-unfair-and-unjust.html
- Hall IJ, Rim SH, Dasari S. Preventive care use among Hispanic adults with limited comfort speaking English: an analysis of the Medical Expenditure Panel Survey data. Prev Med. 2022;159:107042.
- Brach C, Chevarley FM. Demographics and health care access and utilization of limited-English-proficient and English-proficient Hispanics. Agency for Healthcare Research and Quality. February 2008. http://meps.ahrq.gov/mepsweb/data_files/publications//rf28/rf28.pdf
- Berdahl TA, Kirby JB. Patient-provider communication disparities by limited English proficiency (LEP): trends from the US Medical Expenditure Panel Survey, 2006-2015. J General Intern Med. 2019;34:1434-1440.
- Robinson JK, Joshi KM, Ortiz S, et al. Melanoma knowledge, perception, and awareness in ethnic minorities in Chicago: recommendations regarding education. Psychooncology. 2011;20:313-320.
- Robinson JK, Nodal M, Chavez L, et al. Enhancing the relevance of skin self-examination for Latinos. JAMA Dermatol. 2017;153:717-718.
- Buchanan Lunsford N, Berktold J, Holman DM, et al. Skin cancer knowledge, awareness, beliefs and preventive behaviors among black and hispanic men and women. Prev Med Rep. 2018;12:203-209.
- Madrigal JM, Correa-Mendez M, Arias JD, et al. Hispanic, Latino/a, Latinx, Latine: disentangling the identities of Hispanic/Latino Americans. National Cancer Institute Division of Cancer Epidemiology & Genetics. October 20, 2022. Accessed September 3, 2024. https://dceg.cancer.gov/about/diversity-inclusion/inclusivity-minute/2022/disentangling-identities-hispanic-latino-americans
The Latine/Hispanic population in the United States comprises one of the largest and youngest skin of color communities.1,2 In 2020, this group accounted for 19% of all Americans—a percentage expected to increase to more than 25% by 2060.3
It must be emphasized that the Latine/Hispanic community in the United States is incredibly diverse.4 Approximately one-third of individuals in this group are foreign-born, and this community is made up of people from all racialized groups, religions, languages, and cultural identities.2 The heterogeneity of the Latine/Hispanic population translates into a wide representation of skin tones, reflecting a rich range of ancestries, ethnicities, and cultures. The percentage of individuals from each origin group may differ according to where they live in the United States; for instance, individuals who identify as Mexican comprise more than 80% of the Latine/Hispanic population in both Texas and California but only 17% in Florida, where more than half of Latine/Hispanic people identify as Cuban or Puerto Rican.4,5 As a result, when it comes to skin cancer epidemiology, variations in incidence and mortality may exist within each of these subgroups who identify as part of the Latine/Hispanic community, as reported for other cancers.6,7 Further research is needed to investigate these potential differences.Unfortunately, considerable health disparities persist among this rapidly growing population, including increased morbidity and mortality from melanoma and keratinocyte carcinomas (KCs) despite overall low lifetime incidence.8,9 In this review, the epidemiology, clinical manifestation, and ethnic disparities for skin cancer among the US Latine/Hispanic population are summarized; other factors impacting overall health and health care, including sociocultural factors, also are briefly discussed.
Terminology
Before a meaningful dialogue can be had about skin cancer in the Latine/Hispanic population, it is important to contextualize the terms used to identify this patient population, including Latino/Latine and Hispanic. In the early 1970s, the United States adopted the term Hispanic as a way of conglomerating Spanish-speaking individuals from Spain, the Caribbean, and Central and South America. The goal was to implement a common identifier that enabled the US government to study the economic and social development of these groups.10 Nevertheless, considerable differences (eg, variations in skin pigmentation, sun sensitivity) exist among Hispanic communities, with some having stronger European, African, or Amerindian influences due to colonization of their distinct countries.11
In contrast, Latino is a geographic term and refers to people with roots in Latin America and the Caribbean (Table 1).12,13 For example, a person from Brazil may be considered Latino but not Hispanic as Brazilians speak Portuguese; alternatively, Spaniards (who are considered Hispanic) are not Latino because Spain is not a Latin American country. A person from Mexico would be considered both Latino and Hispanic.13

More recently, the term Latine has been introduced as an alternative to the gender binary inherent in the Spanish language.12 For the purposes of this article, the terms Latine and Hispanic will be used interchangeably (unless otherwise specified) depending on how they are cited in the existing literature. Furthermore, the term non-Hispanic White (NHW) will be used to refer to individuals who have been socially ascribed or who self-identify as White in terms of race or ethnicity.
Melanoma
Melanoma, the deadliest form of skin cancer, is more likely to metastasize compared to other forms of skin cancer, including basal cell carcinoma (BCC) and squamous cell carcinoma (SCC). For Latine/Hispanic individuals living in the United States, the lifetime risk for melanoma is 1 in 200 compared to 1 in 33 for NHW individuals.14 While the lifetime risk for melanoma is low for the Latine/Hispanic population, Hispanic individuals are diagnosed with melanoma at an earlier age (mean, 56 years), and the rate of new cases is marginally higher for women (4.9 per 100,000) compared to men (4.8 per 100,000).15,16
Typical sites of melanoma manifestation in Latine/Hispanic individuals include the torso (most common site in Hispanic men), lower extremities (most common site in Hispanic women), and acral sites (palms, soles, and nails).9,16,17 Anatomic location also can vary according to age for both men and women. For men, the incidence of melanoma on the trunk appears to decrease with age, while the incidence on the head and neck may increase. For women, the incidence of melanoma on the lower extremities and hip increases with age. Cutaneous melanoma may manifest as a lesion with asymmetry, irregular borders, variation in pigmentation, large diameter (>6 mm), and evolution over time. In patients with skin of color, melanoma easily can be missed, as it also typically mimics more benign skin conditions and may develop from an existing black- or dark brown–pigmented macule.18 The most common histologic subtype reported among Latine/Hispanic individuals in the United States is superficial spreading melanoma (20%–23%) followed by nodular melanoma and acral lentiginous melanoma.16,19 Until additional risk factors associated with melanoma susceptibility in Hispanic/Latine people are better elucidated, it may be appropriate to use an alternative acronym, such as CUBED (Table 2), in addition to the standard ABCDE system to help recognize potential melanoma on acral sites.18

Although the lifetime risk for melanoma among Hispanic individuals in the United States is lower than that for NHW individuals, Hispanic patients who are diagnosed with melanoma are more likely to present with increased tumor thickness and later-stage diagnosis compared to NHW individuals.8,16,20 In a recent study by Qian et al,8 advanced stage melanoma—defined as regional or distant stage disease—was present in 12.6% of NHW individuals. In contrast, the percentage of Hispanics with advanced disease was higher at 21%.8 Even after controlling for insurance and poverty status, Hispanic individuals were at greater risk than NHW individuals for late-stage diagnosis.16,20
Morbidity and mortality also have been shown to be higher in Hispanic patients with cutaneous melanoma.9,17 Reasons for this are multifactorial, with studies specific to melanoma citing challenges associated with early detection in individuals with deeply pigmented skin, a lack of awareness and knowledge about skin cancer among Latine/Hispanic patients, and treatment disparities.21-23 Moreover, very few studies have reported comprehensive data on patients from Africa and Latin America. Studies examining the role of genetic ancestry, epigenetic variants, and skin pigmentation and the risk for melanoma among the Latine/Hispanic population therefore are much needed.24
Keratinocyte Carcinomas
Keratinocyte carcinomas, also known as nonmelanoma skin cancers, include BCC and SCC. In comparison to the high-quality data available for melanoma from cancer registries, there are less reliable incidence data for KCs, especially among individuals with skin of color.25 As a result, KC epidemiology in the United States is drawn largely from case series (especially for individuals with skin of color) or claims data from small data sets often from geographically restricted regions within the United States.25,26
Basal Cell Carcinoma—Basal cell carcinoma is the most common malignant skin cancer in Latine/Hispanic individuals. Among those with lighter skin tones, the lifetime risk for BCC is about 30%.27,28 Men typically are affected at a higher rate than women, and the median age for diagnosis is 68 years.29 The development of BCC primarily is linked to lifetime accumulated UV radiation exposure. Even though BCC has a low mortality rate, it can lead to substantial morbidity due to factors such as tumor location, size, and rate of invasion, resulting in cosmetic and functional issues. Given its low metastatic potential, treatment of BCC typically is aimed at local control.30 Options for treatment include Mohs micrographic surgery (MMS), curettage and electrodessication, cryosurgery, photodynamic therapy, radiation therapy, and topical therapies. Systemic therapies are reserved for patients with locally advanced or metastatic disease.30
Latine/Hispanic patients characteristically present with BCCs on sun-exposed areas of the skin such as the head and neck region. In most patients, BCC manifests as a translucent pearly nodule with superficial telangiectasias and/or a nonhealing ulcer with a central depression and rolled nontender borders. However, in patients with skin of color, 66% of BCCs manifest with pigmentation; in fact, pigmented BCC (a subtype of BCC) has been shown to have a higher prevalence among Hispanic individuals, with an incidence twice as frequent as in NHW individuals.31 In addition, there are reports of increased tendency among Latine/Hispanic individuals to develop multiple BCCs.32,33
The relationship between UV exposure and KCs could explain the relatively higher incidence in populations with skin of color living in warmer climates, including Hispanic individuals.34 Even so, the development of BCCs appears to correlate directly with the degree of pigmentation in the skin, as it is most common in individuals with lighter skin tones within the Hispanic population.25,34,35 Other risk factors associated with BCC development include albinism, arsenic ingestion, chronic infections, immunosuppression, history of radiation treatment, and history of scars or ulcers due to physical/thermal trauma.35-37
Squamous Cell Carcinoma—Squamous cell carcinoma is the second most common skin cancer among Latine/Hispanic patients. In contrast with NHW patients, evidence supporting the role of UV exposure as a primary risk factor for SCC in patients with skin of color remains limited.25,38 Reports linking UV exposure and KCs in Hispanic and Black individuals predominantly include case series or population-based studies that do not consider levels of UV exposure.25
More recently, genetic ancestry analyses of a large multiethnic cohort found an increased risk for cutaneous SCC among Latine/Hispanic individuals with European ancestry compared to those with Native American or African ancestry; however, these genetic ancestry associations were attenuated (although not eliminated) after considering skin pigmentation (using loci associated with skin pigmentation), history of sun exposure (using actinic keratoses as a covariate for chronic sun exposure), and sun-protected vs sun-exposed anatomic sites, supporting the role of other environmental or sociocultural factors in the development of SCC.39 Similar to BCCs, immunosuppression, chronic scarring, skin irritation, and inflammatory disease also are documented risk factors.9,32
Among NHW individuals with lighter skin tones, SCC characteristically manifests on sun-exposed areas of the skin such as the head and neck region. Typically, a lesion may appear as a scaly erythematous papule or plaque that may be verrucous in nature or a nonhealing bleeding ulcer. In patients with more deeply pigmented skin, SCC tends to develop in the perianal region and on the penis and lower legs; pigmented lesions also may be present (as commonly reported in BCCs).9,32,36
Unfortunately, the lower incidence of KCs and lack of surveillance in populations with skin of color result in a low index of clinical suspicion, leading to delayed diagnoses and increased morbidity.40 Keratinocyte carcinomas are more costly to treat and require more health care resources for Latine/Hispanic and Black patients compared to their NHW counterparts; for example, KCs are associated with more ambulatory visits, more prescription medications, and greater cost on a per-person, per-year basis in Latine/Hispanic and Black patients compared with NHW patients.41 Moreover, a recent multicenter retrospective study found Hispanic patients had 17% larger MMS defects following treatment for KCs compared to NHW patients after adjustment for age, sex, and insurance type.42
Hispanic patients tend to present initially with SCCs in areas associated with advanced disease, such as the anogenital region, penis, and the lower extremities. Latine and Black men have the highest incidence of penile SCC, which is rare with high morbidity and mortality.32,43,44 The higher incidence of penile SCC among Hispanic individuals living in southern states could correspond to circumcision or HPV infection rates,44 ultimately impacting incidence.45
Dermatofibrosarcoma Protuberans
Dermatofibrosarcoma protuberans (DFSP) is a rare locally aggressive cutaneous sarcoma. According to population studies, overall incidence of DFSP is around 4.1 to 4.2 per million in the United States. Population-based studies on DFSP are limited, but available data suggest that Black patients as well as women have the highest incidence.46
Dermatofibrosarcoma protuberans is characterized by its capacity to invade surrounding tissues in a tentaclelike pattern.47 This characteristic often leads to inadequate initial resection of the lesion as well as a high recurrence rate despite its low metastatic potential.48 In early stages, DFSP typically manifests as an asymptomatic plaque with a slow growth rate. The color of the lesion ranges from reddish brown to flesh colored. The pigmented form of DFSP, known as Bednar tumor, is the most common among Black patients.47 As the tumor grows, it tends to become firm and nodular. The most common location for
Although current guidelines designate MMS as the first-line treatment for DFSP, the procedure may be inaccessible for certain populations.49 Patients with skin of color are more likely to undergo wide local excision (WLE) than MMS; however, WLE is less effective, with a recurrence rate of 30% compared with 3% in those treated with MMS.50 A retrospective cohort study of more than 2000 patients revealed that Hispanic and Black patients were less likely to undergo MMS. In addition, the authors noted that WLE recipients more commonly were deceased at the end of the study.51
Despite undergoing treatment for a primary DFSP, Hispanic patients also appear to be at increased risk for a second surgery.52 Additional studies are needed to elucidate the reasons behind higher recurrence rates in Latine/Hispanic patients compared to NHW individuals.
Factors Influencing Skin Cancer Outcomes
In recent years, racial and ethnic disparities in health care use, medical treatment, and quality of care among minoritized populations (including Latine/Hispanic groups) have been documented in the medical literature.53,54 These systemic inequities, which are rooted in structural racism,55 have contributed to poorer health outcomes, worse health status, and lower-quality care for minoritized patients living in the United States, including those impacted by dermatologic conditions.8,43,55-57 Becoming familiar with the sociocultural factors influencing skin cancer outcomes in the Latine/Hispanic community (including the lack of or inadequate health insurance, medical mistrust, language, and other cultural elements) and the paucity of research in this domain could help eliminate existing health inequities in this population.
Health Insurance Coverage—Although the uninsured rates in the Latine population have decreased since the passage of the Affordable Care Act (from 30% in 2013 to a low of 19% in 2017),58 inadequate health insurance coverage remains one of the largest barriers to health care access and a contributor to health disparities among the Latine community. Nearly 1 in 5 Latine individuals in the United States are uninsured compared to 8% of NHW individuals.58 Even though Latine individuals are more likely than non-Latine individuals to be part of the workforce, Latine employees are less likely to receive employer-sponsored coverage (27% vs 53% for NHW individuals).59
Not surprisingly, noncitizens are far less likely to be insured; this includes lawfully present immigrants (ie, permanent residents or green card holders, refugees, asylees, and others who are authorized to live in the United States temporarily or permanently) and undocumented immigrants (including individuals who entered the country without authorization and individuals who entered the country lawfully and stayed after their visa or status expired). The higher uninsured rate among noncitizens reflects not only limited access to employer-sponsored coverage but includes immigrant eligibility restrictions for federal programs such as Medicaid, the Children’s Health Insurance Program, and the Affordable Care Act Marketplace coverage.60
With approximately 9 million Americans living in mixed-status families (and nearly 10% of babies born each year with at least one undocumented parent), restrictive federal or state health care policies may extend beyond their stated target and impact both Latine citizens and noncitizens.61-65 For instance, Vargas et al64 found that both Latine citizens and noncitizens who lived in states with a high number of immigration-related laws had decreased odds of reporting optimal health as compared to Latine respondents in states with fewer immigration-related laws. Other barriers to enrollment include fears and confusion about program qualification, even if eligible.58
Medical Mistrust and Unfamiliarity—Mistrust of medical professionals has been shown to reduce patient adherence to treatment as prescribed by their medical provider and can negatively influence health outcomes.53 For racial/ethnic minoritized groups (including Latine/Hispanic patients), medical mistrust may be rooted in patients’ experience of discrimination in the health care setting. In a recent cross-sectional study, results from a survey of California adults (including 704 non-Hispanic Black, 711 Hispanic, and 913 NHW adults) found links between levels of medical mistrust and perceived discrimination based on race/ethnicity and language as well as perceived discrimination due to income level and type or lack of insurance.53 Interestingly, discrimination attributed to income level and insurance status remained after controlling for race/ethnicity and language. As expected, patients reliant on public insurance programs such as Medicare have been reported to have greater medical mistrust and suspicion compared with private insurance holders.65 Together, these findings support the notion that individuals who have low socioeconomic status and lack insurance coverage—disproportionately historically marginalized populations—are more likely to perceive discrimination in health care settings, have greater medical mistrust, and experience poorer health outcomes.53
It also is important for health care providers to consider that the US health care system is unfamiliar to many Latine/Hispanic individuals. Costs of medical services tend to be substantially higher in the United States, which can contribute to mistrust in the system.66 In addition, unethical medical experimentations have negatively affected both Latine and especially non-Hispanic Black populations, with long-lasting perceptions of deception and exploitation.67 These beliefs have undermined the trust that these populations have in clinicians and the health care system.54,67
Language and Other Cultural Elements—The inability to effectively communicate with health care providers could contribute to disparities in access to and use of health care services among Latine/Hispanic individuals. In a Medical Expenditure Panel Survey analysis, half of Hispanic patients with limited comfort speaking English did not have a usual source of care, and almost 90% of those with a usual source of care had a provider who spoke Spanish or used interpreters—indicating that few Hispanic individuals with limited comfort speaking English selected a usual source of care without language assistance.68,69 In other examples, language barriers contributed to disparities in cancer screening, and individuals with limited English proficiency were more likely to have difficulty understanding their physician due to language barriers.68,70
Improving cultural misconceptions regarding skin conditions, especially skin cancer, is another important consideration in the Latine/Hispanic community. Many Latine/Hispanic individuals wrongly believe they cannot develop skin cancer due to their darker skin tones and lack of family history.26 Moreover, multiple studies assessing melanoma knowledge and perception among participants with skin of color (including one with an equal number of Latine/Hispanic, Black/African American, and Asian individuals for a total of 120 participants) revealed that many were unaware of the risk for melanoma on acral sites.71 Participants expressed a need for more culturally relevant content from both clinicians and public materials (eg, images of acral melanoma in a person with skin of color).71-73
Paucity of Research—There is limited research regarding skin cancer risks and methods of prevention for patients with skin of color, including the Latine/Hispanic population. Efforts to engage and include patients from these communities, as well as clinicians or investigators from similar backgrounds, in clinical studies are desperately needed. It also is important that clinical studies collect data beyond population descriptors to account for both clinical and genetic variations observed in the Latine/Hispanic population.
Latine/Hispanic individuals are quite diverse with many variable factors that may influence skin cancer outcomes. Often, cancer surveillance data are available in aggregate only, which could mask this heterogeneity.74 Rigorous studies that collect more granular data, including objective measures of skin pigmentation beyond self-reported Fitzpatrick skin type, culture/beliefs, lifestyle/behavior, geographic location, socioeconomic status, genetics, or epigenetics could help fully elucidate skin cancer risks and mitigate health disparities among individuals who identify as part of this population.
Final Thoughts
The Latine/Hispanic community—the largest ethnic minoritized group in the United States—is disproportionately affected by dermatologic health disparities. We hope this review helps to increase recognition of the clinical manifestations of skin cancer in Latine/Hispanic patients. Other factors that may impact skin cancer outcomes in this population include (but are not limited to) lack of or inadequate health insurance, medical mistrust, linguistic barriers and/or individual/cultural perspectives, along with limited research. Recognizing and addressing these (albeit complex) barriers that contribute to the inequitable access to health care in this population remains a critical step toward improving skin cancer outcomes.
The Latine/Hispanic population in the United States comprises one of the largest and youngest skin of color communities.1,2 In 2020, this group accounted for 19% of all Americans—a percentage expected to increase to more than 25% by 2060.3
It must be emphasized that the Latine/Hispanic community in the United States is incredibly diverse.4 Approximately one-third of individuals in this group are foreign-born, and this community is made up of people from all racialized groups, religions, languages, and cultural identities.2 The heterogeneity of the Latine/Hispanic population translates into a wide representation of skin tones, reflecting a rich range of ancestries, ethnicities, and cultures. The percentage of individuals from each origin group may differ according to where they live in the United States; for instance, individuals who identify as Mexican comprise more than 80% of the Latine/Hispanic population in both Texas and California but only 17% in Florida, where more than half of Latine/Hispanic people identify as Cuban or Puerto Rican.4,5 As a result, when it comes to skin cancer epidemiology, variations in incidence and mortality may exist within each of these subgroups who identify as part of the Latine/Hispanic community, as reported for other cancers.6,7 Further research is needed to investigate these potential differences.Unfortunately, considerable health disparities persist among this rapidly growing population, including increased morbidity and mortality from melanoma and keratinocyte carcinomas (KCs) despite overall low lifetime incidence.8,9 In this review, the epidemiology, clinical manifestation, and ethnic disparities for skin cancer among the US Latine/Hispanic population are summarized; other factors impacting overall health and health care, including sociocultural factors, also are briefly discussed.
Terminology
Before a meaningful dialogue can be had about skin cancer in the Latine/Hispanic population, it is important to contextualize the terms used to identify this patient population, including Latino/Latine and Hispanic. In the early 1970s, the United States adopted the term Hispanic as a way of conglomerating Spanish-speaking individuals from Spain, the Caribbean, and Central and South America. The goal was to implement a common identifier that enabled the US government to study the economic and social development of these groups.10 Nevertheless, considerable differences (eg, variations in skin pigmentation, sun sensitivity) exist among Hispanic communities, with some having stronger European, African, or Amerindian influences due to colonization of their distinct countries.11
In contrast, Latino is a geographic term and refers to people with roots in Latin America and the Caribbean (Table 1).12,13 For example, a person from Brazil may be considered Latino but not Hispanic as Brazilians speak Portuguese; alternatively, Spaniards (who are considered Hispanic) are not Latino because Spain is not a Latin American country. A person from Mexico would be considered both Latino and Hispanic.13

More recently, the term Latine has been introduced as an alternative to the gender binary inherent in the Spanish language.12 For the purposes of this article, the terms Latine and Hispanic will be used interchangeably (unless otherwise specified) depending on how they are cited in the existing literature. Furthermore, the term non-Hispanic White (NHW) will be used to refer to individuals who have been socially ascribed or who self-identify as White in terms of race or ethnicity.
Melanoma
Melanoma, the deadliest form of skin cancer, is more likely to metastasize compared to other forms of skin cancer, including basal cell carcinoma (BCC) and squamous cell carcinoma (SCC). For Latine/Hispanic individuals living in the United States, the lifetime risk for melanoma is 1 in 200 compared to 1 in 33 for NHW individuals.14 While the lifetime risk for melanoma is low for the Latine/Hispanic population, Hispanic individuals are diagnosed with melanoma at an earlier age (mean, 56 years), and the rate of new cases is marginally higher for women (4.9 per 100,000) compared to men (4.8 per 100,000).15,16
Typical sites of melanoma manifestation in Latine/Hispanic individuals include the torso (most common site in Hispanic men), lower extremities (most common site in Hispanic women), and acral sites (palms, soles, and nails).9,16,17 Anatomic location also can vary according to age for both men and women. For men, the incidence of melanoma on the trunk appears to decrease with age, while the incidence on the head and neck may increase. For women, the incidence of melanoma on the lower extremities and hip increases with age. Cutaneous melanoma may manifest as a lesion with asymmetry, irregular borders, variation in pigmentation, large diameter (>6 mm), and evolution over time. In patients with skin of color, melanoma easily can be missed, as it also typically mimics more benign skin conditions and may develop from an existing black- or dark brown–pigmented macule.18 The most common histologic subtype reported among Latine/Hispanic individuals in the United States is superficial spreading melanoma (20%–23%) followed by nodular melanoma and acral lentiginous melanoma.16,19 Until additional risk factors associated with melanoma susceptibility in Hispanic/Latine people are better elucidated, it may be appropriate to use an alternative acronym, such as CUBED (Table 2), in addition to the standard ABCDE system to help recognize potential melanoma on acral sites.18

Although the lifetime risk for melanoma among Hispanic individuals in the United States is lower than that for NHW individuals, Hispanic patients who are diagnosed with melanoma are more likely to present with increased tumor thickness and later-stage diagnosis compared to NHW individuals.8,16,20 In a recent study by Qian et al,8 advanced stage melanoma—defined as regional or distant stage disease—was present in 12.6% of NHW individuals. In contrast, the percentage of Hispanics with advanced disease was higher at 21%.8 Even after controlling for insurance and poverty status, Hispanic individuals were at greater risk than NHW individuals for late-stage diagnosis.16,20
Morbidity and mortality also have been shown to be higher in Hispanic patients with cutaneous melanoma.9,17 Reasons for this are multifactorial, with studies specific to melanoma citing challenges associated with early detection in individuals with deeply pigmented skin, a lack of awareness and knowledge about skin cancer among Latine/Hispanic patients, and treatment disparities.21-23 Moreover, very few studies have reported comprehensive data on patients from Africa and Latin America. Studies examining the role of genetic ancestry, epigenetic variants, and skin pigmentation and the risk for melanoma among the Latine/Hispanic population therefore are much needed.24
Keratinocyte Carcinomas
Keratinocyte carcinomas, also known as nonmelanoma skin cancers, include BCC and SCC. In comparison to the high-quality data available for melanoma from cancer registries, there are less reliable incidence data for KCs, especially among individuals with skin of color.25 As a result, KC epidemiology in the United States is drawn largely from case series (especially for individuals with skin of color) or claims data from small data sets often from geographically restricted regions within the United States.25,26
Basal Cell Carcinoma—Basal cell carcinoma is the most common malignant skin cancer in Latine/Hispanic individuals. Among those with lighter skin tones, the lifetime risk for BCC is about 30%.27,28 Men typically are affected at a higher rate than women, and the median age for diagnosis is 68 years.29 The development of BCC primarily is linked to lifetime accumulated UV radiation exposure. Even though BCC has a low mortality rate, it can lead to substantial morbidity due to factors such as tumor location, size, and rate of invasion, resulting in cosmetic and functional issues. Given its low metastatic potential, treatment of BCC typically is aimed at local control.30 Options for treatment include Mohs micrographic surgery (MMS), curettage and electrodessication, cryosurgery, photodynamic therapy, radiation therapy, and topical therapies. Systemic therapies are reserved for patients with locally advanced or metastatic disease.30
Latine/Hispanic patients characteristically present with BCCs on sun-exposed areas of the skin such as the head and neck region. In most patients, BCC manifests as a translucent pearly nodule with superficial telangiectasias and/or a nonhealing ulcer with a central depression and rolled nontender borders. However, in patients with skin of color, 66% of BCCs manifest with pigmentation; in fact, pigmented BCC (a subtype of BCC) has been shown to have a higher prevalence among Hispanic individuals, with an incidence twice as frequent as in NHW individuals.31 In addition, there are reports of increased tendency among Latine/Hispanic individuals to develop multiple BCCs.32,33
The relationship between UV exposure and KCs could explain the relatively higher incidence in populations with skin of color living in warmer climates, including Hispanic individuals.34 Even so, the development of BCCs appears to correlate directly with the degree of pigmentation in the skin, as it is most common in individuals with lighter skin tones within the Hispanic population.25,34,35 Other risk factors associated with BCC development include albinism, arsenic ingestion, chronic infections, immunosuppression, history of radiation treatment, and history of scars or ulcers due to physical/thermal trauma.35-37
Squamous Cell Carcinoma—Squamous cell carcinoma is the second most common skin cancer among Latine/Hispanic patients. In contrast with NHW patients, evidence supporting the role of UV exposure as a primary risk factor for SCC in patients with skin of color remains limited.25,38 Reports linking UV exposure and KCs in Hispanic and Black individuals predominantly include case series or population-based studies that do not consider levels of UV exposure.25
More recently, genetic ancestry analyses of a large multiethnic cohort found an increased risk for cutaneous SCC among Latine/Hispanic individuals with European ancestry compared to those with Native American or African ancestry; however, these genetic ancestry associations were attenuated (although not eliminated) after considering skin pigmentation (using loci associated with skin pigmentation), history of sun exposure (using actinic keratoses as a covariate for chronic sun exposure), and sun-protected vs sun-exposed anatomic sites, supporting the role of other environmental or sociocultural factors in the development of SCC.39 Similar to BCCs, immunosuppression, chronic scarring, skin irritation, and inflammatory disease also are documented risk factors.9,32
Among NHW individuals with lighter skin tones, SCC characteristically manifests on sun-exposed areas of the skin such as the head and neck region. Typically, a lesion may appear as a scaly erythematous papule or plaque that may be verrucous in nature or a nonhealing bleeding ulcer. In patients with more deeply pigmented skin, SCC tends to develop in the perianal region and on the penis and lower legs; pigmented lesions also may be present (as commonly reported in BCCs).9,32,36
Unfortunately, the lower incidence of KCs and lack of surveillance in populations with skin of color result in a low index of clinical suspicion, leading to delayed diagnoses and increased morbidity.40 Keratinocyte carcinomas are more costly to treat and require more health care resources for Latine/Hispanic and Black patients compared to their NHW counterparts; for example, KCs are associated with more ambulatory visits, more prescription medications, and greater cost on a per-person, per-year basis in Latine/Hispanic and Black patients compared with NHW patients.41 Moreover, a recent multicenter retrospective study found Hispanic patients had 17% larger MMS defects following treatment for KCs compared to NHW patients after adjustment for age, sex, and insurance type.42
Hispanic patients tend to present initially with SCCs in areas associated with advanced disease, such as the anogenital region, penis, and the lower extremities. Latine and Black men have the highest incidence of penile SCC, which is rare with high morbidity and mortality.32,43,44 The higher incidence of penile SCC among Hispanic individuals living in southern states could correspond to circumcision or HPV infection rates,44 ultimately impacting incidence.45
Dermatofibrosarcoma Protuberans
Dermatofibrosarcoma protuberans (DFSP) is a rare locally aggressive cutaneous sarcoma. According to population studies, overall incidence of DFSP is around 4.1 to 4.2 per million in the United States. Population-based studies on DFSP are limited, but available data suggest that Black patients as well as women have the highest incidence.46
Dermatofibrosarcoma protuberans is characterized by its capacity to invade surrounding tissues in a tentaclelike pattern.47 This characteristic often leads to inadequate initial resection of the lesion as well as a high recurrence rate despite its low metastatic potential.48 In early stages, DFSP typically manifests as an asymptomatic plaque with a slow growth rate. The color of the lesion ranges from reddish brown to flesh colored. The pigmented form of DFSP, known as Bednar tumor, is the most common among Black patients.47 As the tumor grows, it tends to become firm and nodular. The most common location for
Although current guidelines designate MMS as the first-line treatment for DFSP, the procedure may be inaccessible for certain populations.49 Patients with skin of color are more likely to undergo wide local excision (WLE) than MMS; however, WLE is less effective, with a recurrence rate of 30% compared with 3% in those treated with MMS.50 A retrospective cohort study of more than 2000 patients revealed that Hispanic and Black patients were less likely to undergo MMS. In addition, the authors noted that WLE recipients more commonly were deceased at the end of the study.51
Despite undergoing treatment for a primary DFSP, Hispanic patients also appear to be at increased risk for a second surgery.52 Additional studies are needed to elucidate the reasons behind higher recurrence rates in Latine/Hispanic patients compared to NHW individuals.
Factors Influencing Skin Cancer Outcomes
In recent years, racial and ethnic disparities in health care use, medical treatment, and quality of care among minoritized populations (including Latine/Hispanic groups) have been documented in the medical literature.53,54 These systemic inequities, which are rooted in structural racism,55 have contributed to poorer health outcomes, worse health status, and lower-quality care for minoritized patients living in the United States, including those impacted by dermatologic conditions.8,43,55-57 Becoming familiar with the sociocultural factors influencing skin cancer outcomes in the Latine/Hispanic community (including the lack of or inadequate health insurance, medical mistrust, language, and other cultural elements) and the paucity of research in this domain could help eliminate existing health inequities in this population.
Health Insurance Coverage—Although the uninsured rates in the Latine population have decreased since the passage of the Affordable Care Act (from 30% in 2013 to a low of 19% in 2017),58 inadequate health insurance coverage remains one of the largest barriers to health care access and a contributor to health disparities among the Latine community. Nearly 1 in 5 Latine individuals in the United States are uninsured compared to 8% of NHW individuals.58 Even though Latine individuals are more likely than non-Latine individuals to be part of the workforce, Latine employees are less likely to receive employer-sponsored coverage (27% vs 53% for NHW individuals).59
Not surprisingly, noncitizens are far less likely to be insured; this includes lawfully present immigrants (ie, permanent residents or green card holders, refugees, asylees, and others who are authorized to live in the United States temporarily or permanently) and undocumented immigrants (including individuals who entered the country without authorization and individuals who entered the country lawfully and stayed after their visa or status expired). The higher uninsured rate among noncitizens reflects not only limited access to employer-sponsored coverage but includes immigrant eligibility restrictions for federal programs such as Medicaid, the Children’s Health Insurance Program, and the Affordable Care Act Marketplace coverage.60
With approximately 9 million Americans living in mixed-status families (and nearly 10% of babies born each year with at least one undocumented parent), restrictive federal or state health care policies may extend beyond their stated target and impact both Latine citizens and noncitizens.61-65 For instance, Vargas et al64 found that both Latine citizens and noncitizens who lived in states with a high number of immigration-related laws had decreased odds of reporting optimal health as compared to Latine respondents in states with fewer immigration-related laws. Other barriers to enrollment include fears and confusion about program qualification, even if eligible.58
Medical Mistrust and Unfamiliarity—Mistrust of medical professionals has been shown to reduce patient adherence to treatment as prescribed by their medical provider and can negatively influence health outcomes.53 For racial/ethnic minoritized groups (including Latine/Hispanic patients), medical mistrust may be rooted in patients’ experience of discrimination in the health care setting. In a recent cross-sectional study, results from a survey of California adults (including 704 non-Hispanic Black, 711 Hispanic, and 913 NHW adults) found links between levels of medical mistrust and perceived discrimination based on race/ethnicity and language as well as perceived discrimination due to income level and type or lack of insurance.53 Interestingly, discrimination attributed to income level and insurance status remained after controlling for race/ethnicity and language. As expected, patients reliant on public insurance programs such as Medicare have been reported to have greater medical mistrust and suspicion compared with private insurance holders.65 Together, these findings support the notion that individuals who have low socioeconomic status and lack insurance coverage—disproportionately historically marginalized populations—are more likely to perceive discrimination in health care settings, have greater medical mistrust, and experience poorer health outcomes.53
It also is important for health care providers to consider that the US health care system is unfamiliar to many Latine/Hispanic individuals. Costs of medical services tend to be substantially higher in the United States, which can contribute to mistrust in the system.66 In addition, unethical medical experimentations have negatively affected both Latine and especially non-Hispanic Black populations, with long-lasting perceptions of deception and exploitation.67 These beliefs have undermined the trust that these populations have in clinicians and the health care system.54,67
Language and Other Cultural Elements—The inability to effectively communicate with health care providers could contribute to disparities in access to and use of health care services among Latine/Hispanic individuals. In a Medical Expenditure Panel Survey analysis, half of Hispanic patients with limited comfort speaking English did not have a usual source of care, and almost 90% of those with a usual source of care had a provider who spoke Spanish or used interpreters—indicating that few Hispanic individuals with limited comfort speaking English selected a usual source of care without language assistance.68,69 In other examples, language barriers contributed to disparities in cancer screening, and individuals with limited English proficiency were more likely to have difficulty understanding their physician due to language barriers.68,70
Improving cultural misconceptions regarding skin conditions, especially skin cancer, is another important consideration in the Latine/Hispanic community. Many Latine/Hispanic individuals wrongly believe they cannot develop skin cancer due to their darker skin tones and lack of family history.26 Moreover, multiple studies assessing melanoma knowledge and perception among participants with skin of color (including one with an equal number of Latine/Hispanic, Black/African American, and Asian individuals for a total of 120 participants) revealed that many were unaware of the risk for melanoma on acral sites.71 Participants expressed a need for more culturally relevant content from both clinicians and public materials (eg, images of acral melanoma in a person with skin of color).71-73
Paucity of Research—There is limited research regarding skin cancer risks and methods of prevention for patients with skin of color, including the Latine/Hispanic population. Efforts to engage and include patients from these communities, as well as clinicians or investigators from similar backgrounds, in clinical studies are desperately needed. It also is important that clinical studies collect data beyond population descriptors to account for both clinical and genetic variations observed in the Latine/Hispanic population.
Latine/Hispanic individuals are quite diverse with many variable factors that may influence skin cancer outcomes. Often, cancer surveillance data are available in aggregate only, which could mask this heterogeneity.74 Rigorous studies that collect more granular data, including objective measures of skin pigmentation beyond self-reported Fitzpatrick skin type, culture/beliefs, lifestyle/behavior, geographic location, socioeconomic status, genetics, or epigenetics could help fully elucidate skin cancer risks and mitigate health disparities among individuals who identify as part of this population.
Final Thoughts
The Latine/Hispanic community—the largest ethnic minoritized group in the United States—is disproportionately affected by dermatologic health disparities. We hope this review helps to increase recognition of the clinical manifestations of skin cancer in Latine/Hispanic patients. Other factors that may impact skin cancer outcomes in this population include (but are not limited to) lack of or inadequate health insurance, medical mistrust, linguistic barriers and/or individual/cultural perspectives, along with limited research. Recognizing and addressing these (albeit complex) barriers that contribute to the inequitable access to health care in this population remains a critical step toward improving skin cancer outcomes.
- Noe-Bustamnate L, Lopez MH, Krogstad JM. US Hispanic population surpassed 60 million in 2019, but growth has slowed. July 7, 2020. Accessed September 3, 2024. https://www.pewresearch.org/short-reads/2020/07/07/u-s-hispanic-population-surpassed-60-million-in-2019-but-growth-has-slowed/
- Frank C, Lopez MH. Hispanic Americans’ trust in and engagement with science. Pew Research Center. June 14, 2022. Accessed September 3, 2024. https://www.pewresearch.org/wp-content/uploads/sites/20/2022/06/PS_2022.06.14_hispanic-americans-science_REPORT.pdf
- US Census Bureau. Projections of the size and composition of the US population: 2014 to 2060. US Government Printing Office; 2015. Accessed September 5, 2024. https://www.census.gov/content/dam/Census/library/publications/2015/demo/p25-1143.pdf
- Zong J. A mosaic, not a monolith: a profile of the U.S. Latino population, 2000-2020. October 26, 2022. Accessed September 3, 2024. https://latino.ucla.edu/research/latino-population-2000-2020/
- Latinos in California, Texas, New York, Florida and New Jersey. Pew Research Center. March 19, 2004. Accessed September 3, 2024. https://www.pewresearch.org/hispanic/2004/03/19/latinos-in-california-texas-new-york-florida-and-new-jersey/
- Pinheiro PS, Sherman RL, Trapido EJ, et al. Cancer incidence in first generation US Hispanics: Cubans, Mexicans, Puerto Ricans, and new Latinos. Cancer Epidemiol Biomarkers Prev. 2009;18:2162-2169.
- Pinheiro PS, Callahan KE, Kobetz EN. Disaggregated Hispanic groups and cancer: importance, methodology, and current knowledge. In: Ramirez AG, Trapido EJ, eds. Advancing the Science of Cancer in Latinos. Springer; 2020:17-34.
- Qian Y, Johannet P, Sawyers A, et al. The ongoing racial disparities in melanoma: an analysis of the Surveillance, Epidemiology, and End Results database (1975-2016). J Am Acad Dermatol. 2021;84:1585-1593.
- Hogue L, Harvey VM. Basal cell carcinoma, squamous cell carcinoma, and cutaneous melanoma in skin of color patients. Dermatol Clin. 2019;37:519-526.
- Cruzval-O’Reilly E, Lugo-Somolinos A. Melanoma in Hispanics: we may have it all wrong. Cutis. 2020;106:28-30.
- Borrell LN, Elhawary JR, Fuentes-Afflick E, et al. Race and genetic ancestry in medicine—a time for reckoning with racism. N Engl J Med. 2021;384:474-480.
- Lopez MH, Krogstad JM, Passel JS. Who is Hispanic? September 5, 2023. Accessed September 3, 2024. https://www.pewresearch.org/short-reads/2023/09/05/who-is-hispanic/
- Carrasquillo OY, Lambert J, Merritt BG. Comment on “Disparities in nonmelanoma skin cancer in Hispanic/Latino patients based on Mohs micrographic surgery defect size: a multicenter retrospective study.”J Am Acad Dermatol. 2022;87:E129-E130.
- American Cancer Society. Key statistics for melanoma skin cancer. Updated January 17, 2024. Accessed September 3, 2024. https://www.cancer.org/cancer/types/melanoma-skin-cancer/about/key-statistics.html
- National Cancer Institute. Melanoma of the skin: recent trends in SEER age-adjusted incidence rates, 2000-2021. Updated June 27, 2024. Accessed September 3, 2024. https://seer.cancer.gov/statistics-network/explorer/application.htmlsite=53&data_type=1&graph_type=2&compareBy=sex&chk_sex_3=3&chk_sex_2=2&rate_type=2&race=6&age_range=1&stage=101&advopt_precision=1&advopt_show_ci=on&hdn_view=0&advopt_display=2
- Garnett E, Townsend J, Steele B, et al. Characteristics, rates, and trends of melanoma incidence among Hispanics in the USA. Cancer Causes Control. 2016;27:647-659.
- Higgins S, Nazemi A, Feinstein S, et al. Clinical presentations of melanoma in African Americans, Hispanics, and Asians. Dermatol Surg. 2019;45:791-801.
- Bristow IR, de Berker DA, Acland KM, et al. Clinical guidelines for the recognition of melanoma of the foot and nail unit. J Foot Ankle Res. 2010;3:25.
- Fernandez JM, Mata EM, Behbahani S, et al. Survival of Hispanic patients with cutaneous melanoma: a retrospective cohort analysis of 6016 cases from the National Cancer Database. J Am Acad Dermatol. 2023;88:1135-1138.
- Hu S, Sherman R, Arheart K, et al. Predictors of neighborhood risk for late-stage melanoma: addressing disparities through spatial analysis and area-based measures. J Investigative Dermatol. 2014;134:937-945.
- Buster KJ, You Z, Fouad M, et al. Skin cancer risk perceptions: a comparison across ethnicity, age, education, gender, and income. J Am Acad Dermatol. 2012;66:771-779.
- Halpern MT, Ward EM, Pavluck AL, et al. Association of insurance status and ethnicity with cancer stage at diagnosis for 12 cancer sites: a retrospective analysis. Lancet Oncology. 2008;9:222-231.
- Weiss J, Kirsner RS, Hu S. Trends in primary skin cancer prevention among US Hispanics: a systematic review. J Drugs Dermatol. 2012;11:580-586.
- Carvalho LAD, Aguiar FC, Smalley KSM, et al. Acral melanoma: new insights into the immune and genomic landscape. Neoplasia. 2023;46:100947.
- Kolitz E, Lopes F, Arffa M, et al. UV Exposure and the risk of keratinocyte carcinoma in skin of color: a systematic review. JAMA Dermatol. 2022;158:542-546.
- Lukowiak TM, Aizman L, Perz A, et al. Association of age, sex, race, and geographic region with variation of the ratio of basal cell to cutaneous squamous cell carcinomas in the United States. JAMA Dermatol. 2020;156:1192-1198.
- Basset-Seguin N, Herms F. Update in the management of basal cell carcinoma. Acta Derm Venereol. 2020;100:adv00140.
- McDaniel B, Badri T, Steele RB. Basal cell carcinoma. StatPearls [Internet]. Updated March 13, 2024. Accessed September 3, 2024. https://www.ncbi.nlm.nih.gov/books/NBK482439/
- Dessinioti C, Antoniou C, Katsambas A, et al. Basal cell carcinoma: what’s new under the sun. Photochem Photobiol. 2010;86:481-491.
- Kim DP, Kus KJB, Ruiz E. Basal cell carcinoma review. Hematol Oncol Clin North Am. 2019;33:13-24.
- Bigler C, Feldman J, Hall E, et al. Pigmented basal cell carcinoma in Hispanics. J Am Acad Dermatol. 1996;34(5 pt 1):751-752.
- Higgins S, Nazemi A, Chow M, et al. Review of nonmelanoma skin cancer in African Americans, Hispanics, and Asians. Dermatol Surg. 2018;44:903-910.
- Byrd-Miles K, Toombs EL, Peck GL. Skin cancer in individuals of African, Asian, Latin-American, and American-Indian descent: differences in incidence, clinical presentation, and survival compared to Caucasians. J Drugs Dermatol. 2007;6:10-16.
- Rivas M, Rojas E, Calaf GM, et al. Association between non-melanoma and melanoma skin cancer rates, vitamin D and latitude. Oncol Lett. 2017;13:3787-3792.
- Bradford PT. Skin cancer in skin of color. Dermatol Nurs. 2009;21:170-177, 206.
- Davis DS, Robinson C, Callender VD. Skin cancer in women of color: epidemiology, pathogenesis and clinical manifestations. Int J Womens Dermatol. 2021;7:127-134.
- Maafs E, De la Barreda F, Delgado R, et al. Basal cell carcinoma of trunk and extremities. Int J Dermatol. 1997;36:622-628.
- Munjal A, Ferguson N. Skin cancer in skin of color. Dermatol Clin. 2023;41:481-489.
- Jorgenson E, Choquet H, Yin J, et al. Genetic ancestry, skin pigmentation, and the risk of cutaneous squamous cell carcinoma in Hispanic/Latino and non-Hispanic white populations. Commun Biol. 2020;3:765.
- Soliman YS, Mieczkowska K, Zhu TR, et al. Characterizing basal cell carcinoma in Hispanic individuals undergoing Mohs micrographic surgery: a 7-year retrospective review at an academic institution in the Bronx. Brit J Dermatol. 2022;187:597-599.
- Sierro TJ, Blumenthal LY, Hekmatjah J, et al. Differences in health care resource utilization and costs for keratinocyte carcinoma among racioethnic groups: a population-based study. J Am Acad Dermatol. 2022;86:373-378.
- Blumenthal LY, Arzeno J, Syder N, et al. Disparities in nonmelanoma skin cancer in Hispanic/Latino patients based on Mohs micrographic surgery defect size: a multicenter retrospective study. J Am Acad Dermatol. 2022;86:353-358.
- Slopnick EA, Kim SP, Kiechle JE, et al. Racial disparities differ for African Americans and Hispanics in the diagnosis and treatment of penile cancer. Urology. 2016;96:22-28.
- Goodman MT, Hernandez BY, Shvetsov YB. Demographic and pathologic differences in the incidence of invasive penile cancer in the United States, 1995-2003. Cancer Epidemiol Biomarkers Prev. 2007;16:1833-1839.
- Thompson EL, Rosen BL, Maness SB. Social determinants of health and human papillomavirus vaccination among young adults, National Health Interview Survey 2016. J Community Health. 2019;44:149-158.
- Hao X, Billings SD, Wu F, et al. Dermatofibrosarcoma protuberans: update on the diagnosis and treatment. J Clin Med. 2020;9:1752.
- Mosallaei D, Lee EB, Lobl M, et al. Rare cutaneous malignancies in skin of color. Dermatol Surg. 2022;48:606-612.
- Criscito MC, Martires KJ, Stein JA. Prognostic factors, treatment, and survival in dermatofibrosarcoma protuberans. JAMA Dermatol. 2016;152:1365-1371.
- Orenstein LAV, Nelson MM, Wolner Z, et al. Differences in outpatient dermatology encounter work relative value units and net payments by patient race, sex, and age. JAMA Dermatol. 2021;157:406-412.
- Lowe GC, Onajin O, Baum CL, et al. A comparison of Mohs micrographic surgery and wide local excision for treatment of dermatofibrosarcoma protuberans with long-term follow-up: the Mayo Clinic experience. Dermatol Surg. 2017;43:98-106.
- Moore KJ, Chang MS, Weiss J, et al. Racial and ethnic differences in the surgical treatment of dermatofibrosarcoma protuberans: a retrospective cohort analysis. J Am Acad Dermatol. 2022;87:245-247.
- Trofymenko O, Bordeaux JS, Zeitouni NC. Survival in patients with primary dermatofibrosarcoma protuberans: National Cancer Database analysis. J Am Acad Dermatol. 2018;78:1125-1134.
- Bazargan M, Cobb S, Assari S. Discrimination and medical mistrust in a racially and ethnically diverse sample of California adults. Ann Fam Med. 2021;19:4-15.
- Smedley BD, Stith AY, Nelson AR, eds. Unequal Treatment: Confronting Racial and Ethnic Disparities in Health Care. Washington, DC; 2003.
- Bailey ZD, Krieger N, Agenor M, et al. Structural racism and health inequities in the USA: evidence and interventions. Lancet. 2017;389:1453-1463.
- Tackett KJ, Jenkins F, Morrell DS, et al. Structural racism and its influence on the severity of atopic dermatitis in African American children. Pediatric Dermatol. 2020;37:142-146.
- Greif C, Srivastava D, Nijhawan RI. A retrospective cohort study of dermatofibrosarcoma protuberans at a large metropolitan academic center. JAAD Int. 2022;6:104-106.
- Office of the Assistant Secretary for Planning and Evaluation. Health insurance coverage and access to care among Latinos: recent rrends and key challenges (HP-2021-22). October 8, 2021. Accessed September 3, 2024. https://aspe.hhs.gov/reports/health-insurance-coverage-access-care-among-latinos
- Keisler-Starkey K, Bunch LN. Health insurance coverage in the United States: 2020 (Current Population Reports No. P60-274). US Census Bureau; 2021. https://www.census.gov/content/dam/Census/library/publications/2021/demo/p60-274.pdf
- Kaiser Family Foundation. Key facts on health coverage of immigrants. Updated June 26, 2024. Accessed September 3, 2024. https://www.kff.org/racial-equity-and-health-policy/fact-sheet/key-facts-on-health-coverage-of-immigrants/
- Pew Research Center. Unauthorized immigrants: length of residency, patterns of parenthood. Published December 1, 2011. Accessed October 28, 2024. https://www.pewresearch.org/race-and-ethnicity/2011/12/01/unauthorized-immigrants-length-of-residency-patterns-of-parenthood/
- Schneider J, Schmitt M. Understanding the relationship between racial discrimination and mental health among African American adults: a review. SAGE Open. 2015;5:1-10.
- Philbin MM, Flake M, Hatzenbuehler ML, et al. State-level immigration and immigrant-focused policies as drivers of Latino health disparities in the United States. Soc Sci Med. 2018;199:29-38.
- Vargas ED, Sanchez GR, Juarez M. The impact of punitive immigrant laws on the health of Latina/o Populations. Polit Policy. 2017;45:312-337.
- Sutton AL, He J, Edmonds MC, et al. Medical mistrust in Black breast cancer patients: acknowledging the roles of the trustor and the trustee. J Cancer Educ. 2019;34:600-607.
- Jacobs J. An overview of Latin American healthcare systems. Pacific Prime Latin America. July 31, 2023. Accessed September 3, 2024. https://www.pacificprime.lat/blog/an-overview-of-latin-american-healthcare-systems/
- CDC. Unfair and unjust practices and conditions harm Hispanic and Latino people and drive health disparities. May 15, 2024. Accessed September 3, 2024. https://www.cdc.gov/tobacco-health-equity/collection/hispanic-latino-unfair-and-unjust.html
- Hall IJ, Rim SH, Dasari S. Preventive care use among Hispanic adults with limited comfort speaking English: an analysis of the Medical Expenditure Panel Survey data. Prev Med. 2022;159:107042.
- Brach C, Chevarley FM. Demographics and health care access and utilization of limited-English-proficient and English-proficient Hispanics. Agency for Healthcare Research and Quality. February 2008. http://meps.ahrq.gov/mepsweb/data_files/publications//rf28/rf28.pdf
- Berdahl TA, Kirby JB. Patient-provider communication disparities by limited English proficiency (LEP): trends from the US Medical Expenditure Panel Survey, 2006-2015. J General Intern Med. 2019;34:1434-1440.
- Robinson JK, Joshi KM, Ortiz S, et al. Melanoma knowledge, perception, and awareness in ethnic minorities in Chicago: recommendations regarding education. Psychooncology. 2011;20:313-320.
- Robinson JK, Nodal M, Chavez L, et al. Enhancing the relevance of skin self-examination for Latinos. JAMA Dermatol. 2017;153:717-718.
- Buchanan Lunsford N, Berktold J, Holman DM, et al. Skin cancer knowledge, awareness, beliefs and preventive behaviors among black and hispanic men and women. Prev Med Rep. 2018;12:203-209.
- Madrigal JM, Correa-Mendez M, Arias JD, et al. Hispanic, Latino/a, Latinx, Latine: disentangling the identities of Hispanic/Latino Americans. National Cancer Institute Division of Cancer Epidemiology & Genetics. October 20, 2022. Accessed September 3, 2024. https://dceg.cancer.gov/about/diversity-inclusion/inclusivity-minute/2022/disentangling-identities-hispanic-latino-americans
- Noe-Bustamnate L, Lopez MH, Krogstad JM. US Hispanic population surpassed 60 million in 2019, but growth has slowed. July 7, 2020. Accessed September 3, 2024. https://www.pewresearch.org/short-reads/2020/07/07/u-s-hispanic-population-surpassed-60-million-in-2019-but-growth-has-slowed/
- Frank C, Lopez MH. Hispanic Americans’ trust in and engagement with science. Pew Research Center. June 14, 2022. Accessed September 3, 2024. https://www.pewresearch.org/wp-content/uploads/sites/20/2022/06/PS_2022.06.14_hispanic-americans-science_REPORT.pdf
- US Census Bureau. Projections of the size and composition of the US population: 2014 to 2060. US Government Printing Office; 2015. Accessed September 5, 2024. https://www.census.gov/content/dam/Census/library/publications/2015/demo/p25-1143.pdf
- Zong J. A mosaic, not a monolith: a profile of the U.S. Latino population, 2000-2020. October 26, 2022. Accessed September 3, 2024. https://latino.ucla.edu/research/latino-population-2000-2020/
- Latinos in California, Texas, New York, Florida and New Jersey. Pew Research Center. March 19, 2004. Accessed September 3, 2024. https://www.pewresearch.org/hispanic/2004/03/19/latinos-in-california-texas-new-york-florida-and-new-jersey/
- Pinheiro PS, Sherman RL, Trapido EJ, et al. Cancer incidence in first generation US Hispanics: Cubans, Mexicans, Puerto Ricans, and new Latinos. Cancer Epidemiol Biomarkers Prev. 2009;18:2162-2169.
- Pinheiro PS, Callahan KE, Kobetz EN. Disaggregated Hispanic groups and cancer: importance, methodology, and current knowledge. In: Ramirez AG, Trapido EJ, eds. Advancing the Science of Cancer in Latinos. Springer; 2020:17-34.
- Qian Y, Johannet P, Sawyers A, et al. The ongoing racial disparities in melanoma: an analysis of the Surveillance, Epidemiology, and End Results database (1975-2016). J Am Acad Dermatol. 2021;84:1585-1593.
- Hogue L, Harvey VM. Basal cell carcinoma, squamous cell carcinoma, and cutaneous melanoma in skin of color patients. Dermatol Clin. 2019;37:519-526.
- Cruzval-O’Reilly E, Lugo-Somolinos A. Melanoma in Hispanics: we may have it all wrong. Cutis. 2020;106:28-30.
- Borrell LN, Elhawary JR, Fuentes-Afflick E, et al. Race and genetic ancestry in medicine—a time for reckoning with racism. N Engl J Med. 2021;384:474-480.
- Lopez MH, Krogstad JM, Passel JS. Who is Hispanic? September 5, 2023. Accessed September 3, 2024. https://www.pewresearch.org/short-reads/2023/09/05/who-is-hispanic/
- Carrasquillo OY, Lambert J, Merritt BG. Comment on “Disparities in nonmelanoma skin cancer in Hispanic/Latino patients based on Mohs micrographic surgery defect size: a multicenter retrospective study.”J Am Acad Dermatol. 2022;87:E129-E130.
- American Cancer Society. Key statistics for melanoma skin cancer. Updated January 17, 2024. Accessed September 3, 2024. https://www.cancer.org/cancer/types/melanoma-skin-cancer/about/key-statistics.html
- National Cancer Institute. Melanoma of the skin: recent trends in SEER age-adjusted incidence rates, 2000-2021. Updated June 27, 2024. Accessed September 3, 2024. https://seer.cancer.gov/statistics-network/explorer/application.htmlsite=53&data_type=1&graph_type=2&compareBy=sex&chk_sex_3=3&chk_sex_2=2&rate_type=2&race=6&age_range=1&stage=101&advopt_precision=1&advopt_show_ci=on&hdn_view=0&advopt_display=2
- Garnett E, Townsend J, Steele B, et al. Characteristics, rates, and trends of melanoma incidence among Hispanics in the USA. Cancer Causes Control. 2016;27:647-659.
- Higgins S, Nazemi A, Feinstein S, et al. Clinical presentations of melanoma in African Americans, Hispanics, and Asians. Dermatol Surg. 2019;45:791-801.
- Bristow IR, de Berker DA, Acland KM, et al. Clinical guidelines for the recognition of melanoma of the foot and nail unit. J Foot Ankle Res. 2010;3:25.
- Fernandez JM, Mata EM, Behbahani S, et al. Survival of Hispanic patients with cutaneous melanoma: a retrospective cohort analysis of 6016 cases from the National Cancer Database. J Am Acad Dermatol. 2023;88:1135-1138.
- Hu S, Sherman R, Arheart K, et al. Predictors of neighborhood risk for late-stage melanoma: addressing disparities through spatial analysis and area-based measures. J Investigative Dermatol. 2014;134:937-945.
- Buster KJ, You Z, Fouad M, et al. Skin cancer risk perceptions: a comparison across ethnicity, age, education, gender, and income. J Am Acad Dermatol. 2012;66:771-779.
- Halpern MT, Ward EM, Pavluck AL, et al. Association of insurance status and ethnicity with cancer stage at diagnosis for 12 cancer sites: a retrospective analysis. Lancet Oncology. 2008;9:222-231.
- Weiss J, Kirsner RS, Hu S. Trends in primary skin cancer prevention among US Hispanics: a systematic review. J Drugs Dermatol. 2012;11:580-586.
- Carvalho LAD, Aguiar FC, Smalley KSM, et al. Acral melanoma: new insights into the immune and genomic landscape. Neoplasia. 2023;46:100947.
- Kolitz E, Lopes F, Arffa M, et al. UV Exposure and the risk of keratinocyte carcinoma in skin of color: a systematic review. JAMA Dermatol. 2022;158:542-546.
- Lukowiak TM, Aizman L, Perz A, et al. Association of age, sex, race, and geographic region with variation of the ratio of basal cell to cutaneous squamous cell carcinomas in the United States. JAMA Dermatol. 2020;156:1192-1198.
- Basset-Seguin N, Herms F. Update in the management of basal cell carcinoma. Acta Derm Venereol. 2020;100:adv00140.
- McDaniel B, Badri T, Steele RB. Basal cell carcinoma. StatPearls [Internet]. Updated March 13, 2024. Accessed September 3, 2024. https://www.ncbi.nlm.nih.gov/books/NBK482439/
- Dessinioti C, Antoniou C, Katsambas A, et al. Basal cell carcinoma: what’s new under the sun. Photochem Photobiol. 2010;86:481-491.
- Kim DP, Kus KJB, Ruiz E. Basal cell carcinoma review. Hematol Oncol Clin North Am. 2019;33:13-24.
- Bigler C, Feldman J, Hall E, et al. Pigmented basal cell carcinoma in Hispanics. J Am Acad Dermatol. 1996;34(5 pt 1):751-752.
- Higgins S, Nazemi A, Chow M, et al. Review of nonmelanoma skin cancer in African Americans, Hispanics, and Asians. Dermatol Surg. 2018;44:903-910.
- Byrd-Miles K, Toombs EL, Peck GL. Skin cancer in individuals of African, Asian, Latin-American, and American-Indian descent: differences in incidence, clinical presentation, and survival compared to Caucasians. J Drugs Dermatol. 2007;6:10-16.
- Rivas M, Rojas E, Calaf GM, et al. Association between non-melanoma and melanoma skin cancer rates, vitamin D and latitude. Oncol Lett. 2017;13:3787-3792.
- Bradford PT. Skin cancer in skin of color. Dermatol Nurs. 2009;21:170-177, 206.
- Davis DS, Robinson C, Callender VD. Skin cancer in women of color: epidemiology, pathogenesis and clinical manifestations. Int J Womens Dermatol. 2021;7:127-134.
- Maafs E, De la Barreda F, Delgado R, et al. Basal cell carcinoma of trunk and extremities. Int J Dermatol. 1997;36:622-628.
- Munjal A, Ferguson N. Skin cancer in skin of color. Dermatol Clin. 2023;41:481-489.
- Jorgenson E, Choquet H, Yin J, et al. Genetic ancestry, skin pigmentation, and the risk of cutaneous squamous cell carcinoma in Hispanic/Latino and non-Hispanic white populations. Commun Biol. 2020;3:765.
- Soliman YS, Mieczkowska K, Zhu TR, et al. Characterizing basal cell carcinoma in Hispanic individuals undergoing Mohs micrographic surgery: a 7-year retrospective review at an academic institution in the Bronx. Brit J Dermatol. 2022;187:597-599.
- Sierro TJ, Blumenthal LY, Hekmatjah J, et al. Differences in health care resource utilization and costs for keratinocyte carcinoma among racioethnic groups: a population-based study. J Am Acad Dermatol. 2022;86:373-378.
- Blumenthal LY, Arzeno J, Syder N, et al. Disparities in nonmelanoma skin cancer in Hispanic/Latino patients based on Mohs micrographic surgery defect size: a multicenter retrospective study. J Am Acad Dermatol. 2022;86:353-358.
- Slopnick EA, Kim SP, Kiechle JE, et al. Racial disparities differ for African Americans and Hispanics in the diagnosis and treatment of penile cancer. Urology. 2016;96:22-28.
- Goodman MT, Hernandez BY, Shvetsov YB. Demographic and pathologic differences in the incidence of invasive penile cancer in the United States, 1995-2003. Cancer Epidemiol Biomarkers Prev. 2007;16:1833-1839.
- Thompson EL, Rosen BL, Maness SB. Social determinants of health and human papillomavirus vaccination among young adults, National Health Interview Survey 2016. J Community Health. 2019;44:149-158.
- Hao X, Billings SD, Wu F, et al. Dermatofibrosarcoma protuberans: update on the diagnosis and treatment. J Clin Med. 2020;9:1752.
- Mosallaei D, Lee EB, Lobl M, et al. Rare cutaneous malignancies in skin of color. Dermatol Surg. 2022;48:606-612.
- Criscito MC, Martires KJ, Stein JA. Prognostic factors, treatment, and survival in dermatofibrosarcoma protuberans. JAMA Dermatol. 2016;152:1365-1371.
- Orenstein LAV, Nelson MM, Wolner Z, et al. Differences in outpatient dermatology encounter work relative value units and net payments by patient race, sex, and age. JAMA Dermatol. 2021;157:406-412.
- Lowe GC, Onajin O, Baum CL, et al. A comparison of Mohs micrographic surgery and wide local excision for treatment of dermatofibrosarcoma protuberans with long-term follow-up: the Mayo Clinic experience. Dermatol Surg. 2017;43:98-106.
- Moore KJ, Chang MS, Weiss J, et al. Racial and ethnic differences in the surgical treatment of dermatofibrosarcoma protuberans: a retrospective cohort analysis. J Am Acad Dermatol. 2022;87:245-247.
- Trofymenko O, Bordeaux JS, Zeitouni NC. Survival in patients with primary dermatofibrosarcoma protuberans: National Cancer Database analysis. J Am Acad Dermatol. 2018;78:1125-1134.
- Bazargan M, Cobb S, Assari S. Discrimination and medical mistrust in a racially and ethnically diverse sample of California adults. Ann Fam Med. 2021;19:4-15.
- Smedley BD, Stith AY, Nelson AR, eds. Unequal Treatment: Confronting Racial and Ethnic Disparities in Health Care. Washington, DC; 2003.
- Bailey ZD, Krieger N, Agenor M, et al. Structural racism and health inequities in the USA: evidence and interventions. Lancet. 2017;389:1453-1463.
- Tackett KJ, Jenkins F, Morrell DS, et al. Structural racism and its influence on the severity of atopic dermatitis in African American children. Pediatric Dermatol. 2020;37:142-146.
- Greif C, Srivastava D, Nijhawan RI. A retrospective cohort study of dermatofibrosarcoma protuberans at a large metropolitan academic center. JAAD Int. 2022;6:104-106.
- Office of the Assistant Secretary for Planning and Evaluation. Health insurance coverage and access to care among Latinos: recent rrends and key challenges (HP-2021-22). October 8, 2021. Accessed September 3, 2024. https://aspe.hhs.gov/reports/health-insurance-coverage-access-care-among-latinos
- Keisler-Starkey K, Bunch LN. Health insurance coverage in the United States: 2020 (Current Population Reports No. P60-274). US Census Bureau; 2021. https://www.census.gov/content/dam/Census/library/publications/2021/demo/p60-274.pdf
- Kaiser Family Foundation. Key facts on health coverage of immigrants. Updated June 26, 2024. Accessed September 3, 2024. https://www.kff.org/racial-equity-and-health-policy/fact-sheet/key-facts-on-health-coverage-of-immigrants/
- Pew Research Center. Unauthorized immigrants: length of residency, patterns of parenthood. Published December 1, 2011. Accessed October 28, 2024. https://www.pewresearch.org/race-and-ethnicity/2011/12/01/unauthorized-immigrants-length-of-residency-patterns-of-parenthood/
- Schneider J, Schmitt M. Understanding the relationship between racial discrimination and mental health among African American adults: a review. SAGE Open. 2015;5:1-10.
- Philbin MM, Flake M, Hatzenbuehler ML, et al. State-level immigration and immigrant-focused policies as drivers of Latino health disparities in the United States. Soc Sci Med. 2018;199:29-38.
- Vargas ED, Sanchez GR, Juarez M. The impact of punitive immigrant laws on the health of Latina/o Populations. Polit Policy. 2017;45:312-337.
- Sutton AL, He J, Edmonds MC, et al. Medical mistrust in Black breast cancer patients: acknowledging the roles of the trustor and the trustee. J Cancer Educ. 2019;34:600-607.
- Jacobs J. An overview of Latin American healthcare systems. Pacific Prime Latin America. July 31, 2023. Accessed September 3, 2024. https://www.pacificprime.lat/blog/an-overview-of-latin-american-healthcare-systems/
- CDC. Unfair and unjust practices and conditions harm Hispanic and Latino people and drive health disparities. May 15, 2024. Accessed September 3, 2024. https://www.cdc.gov/tobacco-health-equity/collection/hispanic-latino-unfair-and-unjust.html
- Hall IJ, Rim SH, Dasari S. Preventive care use among Hispanic adults with limited comfort speaking English: an analysis of the Medical Expenditure Panel Survey data. Prev Med. 2022;159:107042.
- Brach C, Chevarley FM. Demographics and health care access and utilization of limited-English-proficient and English-proficient Hispanics. Agency for Healthcare Research and Quality. February 2008. http://meps.ahrq.gov/mepsweb/data_files/publications//rf28/rf28.pdf
- Berdahl TA, Kirby JB. Patient-provider communication disparities by limited English proficiency (LEP): trends from the US Medical Expenditure Panel Survey, 2006-2015. J General Intern Med. 2019;34:1434-1440.
- Robinson JK, Joshi KM, Ortiz S, et al. Melanoma knowledge, perception, and awareness in ethnic minorities in Chicago: recommendations regarding education. Psychooncology. 2011;20:313-320.
- Robinson JK, Nodal M, Chavez L, et al. Enhancing the relevance of skin self-examination for Latinos. JAMA Dermatol. 2017;153:717-718.
- Buchanan Lunsford N, Berktold J, Holman DM, et al. Skin cancer knowledge, awareness, beliefs and preventive behaviors among black and hispanic men and women. Prev Med Rep. 2018;12:203-209.
- Madrigal JM, Correa-Mendez M, Arias JD, et al. Hispanic, Latino/a, Latinx, Latine: disentangling the identities of Hispanic/Latino Americans. National Cancer Institute Division of Cancer Epidemiology & Genetics. October 20, 2022. Accessed September 3, 2024. https://dceg.cancer.gov/about/diversity-inclusion/inclusivity-minute/2022/disentangling-identities-hispanic-latino-americans
Practice Points
- The Latine/Hispanic community—the largest ethnic minoritized group in the United States—is disproportionately affected by disparities in skin cancer outcomes.
- Factors influencing skin cancer outcomes in Latine/Hispanic patients in the United States are complex and multidimensional, including lack of familiarity among dermatologists with skin cancer manifestation in this population compared to non-Hispanic White individuals as well as limited data elucidating risk factors for skin cancer in patients with skin of color and sociocultural factors.
Pinto Bean Pressure Wraps: A Novel Approach to Treating Digital Warts
Practice Gap
Verruca vulgaris is a common dermatologic challenge due to its high prevalence and tendency to recur following routinely employed destructive modalities (eg, cryotherapy, electrosurgery), which can incur a considerable amount of pain and some risk for scarring.1,2 Other treatment methods for warts such as topical salicylic acid preparations, topical immunotherapy, or intralesional allergen injections often require multiple treatment sessions.3,4 Furthermore, the financial burden of traditional wart treatment can be substantial.4 Better techniques are needed to improve the clinician’s approach to treating warts. We describe a home-based technique to treat common digital warts using pinto bean pressure wraps to induce ischemic changes in wart tissue with similar response rates to commonly used modalities.
Technique
Our technique utilizes a small, hard, convex object that is applied directly over the digital wart. A simple self-adhesive wrap is used to cover the object and maintain constant pressure on the wart overnight. We typically use a dried pinto bean (a variety of the common bean Phaseolus vulgaris) acquired from a local grocery store due to its ideal size, hard surface, and convex shape (Figure 1). The bean is taped in place directly overlying the wart and covered with a self-adhesive wrap overnight. The wrap is removed in the morning, and often no further treatment is needed. The ischemic wart tissue is allowed to slough spontaneously over 1 to 2 weeks. No wound care or dressing is necessary (Figure 2). Larger warts may require application of the pressure wraps for 2 to 3 additional nights. While most warts resolve with this technique, we have observed a recurrence rate similar to that for cryotherapy. Patients are advised that any recurrent warts can be re-treated monthly, if needed, until resolution.
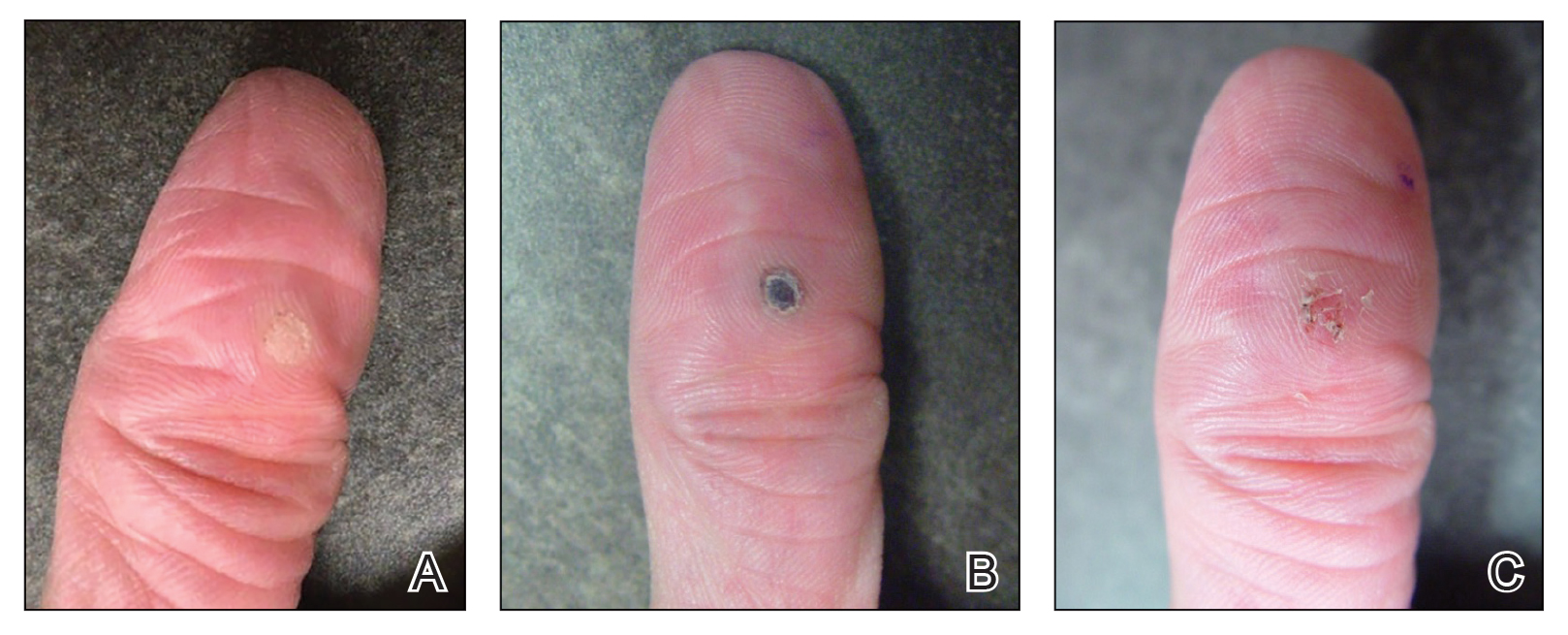
What to Use and How to Prepare—Any small, hard, convex object can be used for the pressure wrap; we also have used appropriately sized and shaped plastic shirt buttons with similar results. Home kits can be assembled in advance and provided to patients at their initial visit along with appropriate instructions (Figure 1A).
Effects on the Skin and Distal Digit—Application of pressure wraps does not harm normal skin; however, care should be taken when the self-adherent wrap is applied so as not to induce ischemia of the distal digit. The wrap should be applied using gentle pressure with patients experiencing minimal discomfort from the overnight application.
Indications—This pressure wrap technique can be employed on most digital warts, including periungual warts, which can be difficult to treat by other means. However, in our experience this technique is not effective for nondigital warts, likely due to the inability to maintain adequate pressure with the overlying dressing. Patients at risk for compromised digital perfusion, such as those with Raynaud phenomenon or systemic sclerosis, should not be treated with pressure wraps due to possible digital ischemia.
Precautions—Patients should be advised that the pinto bean should only be used if dry and should not be ingested. The bean can be a choking hazard for small children, therefore appropriate precautions should be used. Allergic contact dermatitis to the materials used in this technique is possible, but we have never observed this. The pinto bean can be reused for future application as long as it remains dry and provides a hard convex surface.
Practice Implications
The probable mechanism of the ischemic changes to the wart tissue likely is the occlusion of tortuous blood vessels in the dermal papillae, which are intrinsic to wart tissue and absent in normal skin.1 This pressure-induced ischemic injury allows for selective destruction of the wart tissue with sparing of the normal skin. Our technique is fairly novel, although at least one report in the literature has described the use of a mechanical device to induce ischemic changes in skin tags.5
The use of pinto bean pressure wraps to induce ischemic change in digital warts provides a low-risk and nearly pain-free alternative to more expensive and invasive treatment methods. Moreover, this technique allows for a low-cost home-based therapy that can be repeated easily for other digital sites or if recurrence is noted.
- Cardoso J, Calonje E. Cutaneous manifestations of human papillomaviruses: a review. Acta Dermatovenerol Alp Pannonica Adriat. 2011;20:145-154.
- Lipke M. An armamentarium of wart treatments. Clin Med Res. 2006;4:273-293. doi:10.3121/cmr.4.4.273
- Muse M, Stiff K, Glines K, et al. A review of intralesional wart therapy. Dermatol Online J. 2020;26:2. doi:10.5070/D3263048027
- Berna R, Margolis D, Barbieri J. Annual health care utilization and costs for treatment of cutaneous and anogenital warts among a commercially insured population in the US, 2017-2019. JAMA Dermatol. 2022;158:695-697. doi:10.1001/jamadermatol.2022.0964
- Fredriksson C, Ilias M, Anderson C. New mechanical device for effective removal of skin tags in routine health care. Dermatol Online J. 2009;15:9. doi:10.5070/D37tj2800k
Practice Gap
Verruca vulgaris is a common dermatologic challenge due to its high prevalence and tendency to recur following routinely employed destructive modalities (eg, cryotherapy, electrosurgery), which can incur a considerable amount of pain and some risk for scarring.1,2 Other treatment methods for warts such as topical salicylic acid preparations, topical immunotherapy, or intralesional allergen injections often require multiple treatment sessions.3,4 Furthermore, the financial burden of traditional wart treatment can be substantial.4 Better techniques are needed to improve the clinician’s approach to treating warts. We describe a home-based technique to treat common digital warts using pinto bean pressure wraps to induce ischemic changes in wart tissue with similar response rates to commonly used modalities.
Technique
Our technique utilizes a small, hard, convex object that is applied directly over the digital wart. A simple self-adhesive wrap is used to cover the object and maintain constant pressure on the wart overnight. We typically use a dried pinto bean (a variety of the common bean Phaseolus vulgaris) acquired from a local grocery store due to its ideal size, hard surface, and convex shape (Figure 1). The bean is taped in place directly overlying the wart and covered with a self-adhesive wrap overnight. The wrap is removed in the morning, and often no further treatment is needed. The ischemic wart tissue is allowed to slough spontaneously over 1 to 2 weeks. No wound care or dressing is necessary (Figure 2). Larger warts may require application of the pressure wraps for 2 to 3 additional nights. While most warts resolve with this technique, we have observed a recurrence rate similar to that for cryotherapy. Patients are advised that any recurrent warts can be re-treated monthly, if needed, until resolution.


What to Use and How to Prepare—Any small, hard, convex object can be used for the pressure wrap; we also have used appropriately sized and shaped plastic shirt buttons with similar results. Home kits can be assembled in advance and provided to patients at their initial visit along with appropriate instructions (Figure 1A).
Effects on the Skin and Distal Digit—Application of pressure wraps does not harm normal skin; however, care should be taken when the self-adherent wrap is applied so as not to induce ischemia of the distal digit. The wrap should be applied using gentle pressure with patients experiencing minimal discomfort from the overnight application.
Indications—This pressure wrap technique can be employed on most digital warts, including periungual warts, which can be difficult to treat by other means. However, in our experience this technique is not effective for nondigital warts, likely due to the inability to maintain adequate pressure with the overlying dressing. Patients at risk for compromised digital perfusion, such as those with Raynaud phenomenon or systemic sclerosis, should not be treated with pressure wraps due to possible digital ischemia.
Precautions—Patients should be advised that the pinto bean should only be used if dry and should not be ingested. The bean can be a choking hazard for small children, therefore appropriate precautions should be used. Allergic contact dermatitis to the materials used in this technique is possible, but we have never observed this. The pinto bean can be reused for future application as long as it remains dry and provides a hard convex surface.
Practice Implications
The probable mechanism of the ischemic changes to the wart tissue likely is the occlusion of tortuous blood vessels in the dermal papillae, which are intrinsic to wart tissue and absent in normal skin.1 This pressure-induced ischemic injury allows for selective destruction of the wart tissue with sparing of the normal skin. Our technique is fairly novel, although at least one report in the literature has described the use of a mechanical device to induce ischemic changes in skin tags.5
The use of pinto bean pressure wraps to induce ischemic change in digital warts provides a low-risk and nearly pain-free alternative to more expensive and invasive treatment methods. Moreover, this technique allows for a low-cost home-based therapy that can be repeated easily for other digital sites or if recurrence is noted.
Practice Gap
Verruca vulgaris is a common dermatologic challenge due to its high prevalence and tendency to recur following routinely employed destructive modalities (eg, cryotherapy, electrosurgery), which can incur a considerable amount of pain and some risk for scarring.1,2 Other treatment methods for warts such as topical salicylic acid preparations, topical immunotherapy, or intralesional allergen injections often require multiple treatment sessions.3,4 Furthermore, the financial burden of traditional wart treatment can be substantial.4 Better techniques are needed to improve the clinician’s approach to treating warts. We describe a home-based technique to treat common digital warts using pinto bean pressure wraps to induce ischemic changes in wart tissue with similar response rates to commonly used modalities.
Technique
Our technique utilizes a small, hard, convex object that is applied directly over the digital wart. A simple self-adhesive wrap is used to cover the object and maintain constant pressure on the wart overnight. We typically use a dried pinto bean (a variety of the common bean Phaseolus vulgaris) acquired from a local grocery store due to its ideal size, hard surface, and convex shape (Figure 1). The bean is taped in place directly overlying the wart and covered with a self-adhesive wrap overnight. The wrap is removed in the morning, and often no further treatment is needed. The ischemic wart tissue is allowed to slough spontaneously over 1 to 2 weeks. No wound care or dressing is necessary (Figure 2). Larger warts may require application of the pressure wraps for 2 to 3 additional nights. While most warts resolve with this technique, we have observed a recurrence rate similar to that for cryotherapy. Patients are advised that any recurrent warts can be re-treated monthly, if needed, until resolution.


What to Use and How to Prepare—Any small, hard, convex object can be used for the pressure wrap; we also have used appropriately sized and shaped plastic shirt buttons with similar results. Home kits can be assembled in advance and provided to patients at their initial visit along with appropriate instructions (Figure 1A).
Effects on the Skin and Distal Digit—Application of pressure wraps does not harm normal skin; however, care should be taken when the self-adherent wrap is applied so as not to induce ischemia of the distal digit. The wrap should be applied using gentle pressure with patients experiencing minimal discomfort from the overnight application.
Indications—This pressure wrap technique can be employed on most digital warts, including periungual warts, which can be difficult to treat by other means. However, in our experience this technique is not effective for nondigital warts, likely due to the inability to maintain adequate pressure with the overlying dressing. Patients at risk for compromised digital perfusion, such as those with Raynaud phenomenon or systemic sclerosis, should not be treated with pressure wraps due to possible digital ischemia.
Precautions—Patients should be advised that the pinto bean should only be used if dry and should not be ingested. The bean can be a choking hazard for small children, therefore appropriate precautions should be used. Allergic contact dermatitis to the materials used in this technique is possible, but we have never observed this. The pinto bean can be reused for future application as long as it remains dry and provides a hard convex surface.
Practice Implications
The probable mechanism of the ischemic changes to the wart tissue likely is the occlusion of tortuous blood vessels in the dermal papillae, which are intrinsic to wart tissue and absent in normal skin.1 This pressure-induced ischemic injury allows for selective destruction of the wart tissue with sparing of the normal skin. Our technique is fairly novel, although at least one report in the literature has described the use of a mechanical device to induce ischemic changes in skin tags.5
The use of pinto bean pressure wraps to induce ischemic change in digital warts provides a low-risk and nearly pain-free alternative to more expensive and invasive treatment methods. Moreover, this technique allows for a low-cost home-based therapy that can be repeated easily for other digital sites or if recurrence is noted.
- Cardoso J, Calonje E. Cutaneous manifestations of human papillomaviruses: a review. Acta Dermatovenerol Alp Pannonica Adriat. 2011;20:145-154.
- Lipke M. An armamentarium of wart treatments. Clin Med Res. 2006;4:273-293. doi:10.3121/cmr.4.4.273
- Muse M, Stiff K, Glines K, et al. A review of intralesional wart therapy. Dermatol Online J. 2020;26:2. doi:10.5070/D3263048027
- Berna R, Margolis D, Barbieri J. Annual health care utilization and costs for treatment of cutaneous and anogenital warts among a commercially insured population in the US, 2017-2019. JAMA Dermatol. 2022;158:695-697. doi:10.1001/jamadermatol.2022.0964
- Fredriksson C, Ilias M, Anderson C. New mechanical device for effective removal of skin tags in routine health care. Dermatol Online J. 2009;15:9. doi:10.5070/D37tj2800k
- Cardoso J, Calonje E. Cutaneous manifestations of human papillomaviruses: a review. Acta Dermatovenerol Alp Pannonica Adriat. 2011;20:145-154.
- Lipke M. An armamentarium of wart treatments. Clin Med Res. 2006;4:273-293. doi:10.3121/cmr.4.4.273
- Muse M, Stiff K, Glines K, et al. A review of intralesional wart therapy. Dermatol Online J. 2020;26:2. doi:10.5070/D3263048027
- Berna R, Margolis D, Barbieri J. Annual health care utilization and costs for treatment of cutaneous and anogenital warts among a commercially insured population in the US, 2017-2019. JAMA Dermatol. 2022;158:695-697. doi:10.1001/jamadermatol.2022.0964
- Fredriksson C, Ilias M, Anderson C. New mechanical device for effective removal of skin tags in routine health care. Dermatol Online J. 2009;15:9. doi:10.5070/D37tj2800k
Alopecia Induced by Poly-L-Lactic Acid Injection
Cosmetic procedures carry inherent risks of adverse events. Transient and permanent alopecia are rare complications of these procedures. Although they have not been fully elucidated, several pathologic mechanisms for hair loss following cosmetic procedures have been proposed, including extravascular compression (a phenomenon that has been well documented in bedridden patients) as well as intravascular occlusion leading to inflammation and necrosis, which has been associated with hyaluronic acid (HA) fillers.¹ Cases of alopecia also have been reported following mesotherapy and calcium hydroxyapatite, deoxycholic acid, and botulinum toxin injections.² We report a case of alopecia resulting from poly-L-lactic acid (PLLA) injection in a 35-year-old woman with the intent to raise awareness of this rare adverse event.
Case Report
A healthy 35-year-old woman received aesthetic PLLA injections on the face and frontal hairline performed by an outside dermatologist using the vector technique. During the procedure, the patient experienced intense itchiness at the right temporal artery vascular territory and reported a substantial headache the next day. She also presented with erythema and edema of the frontal and right parietal scalp with a well-delimited livedoid vascular area along the temporal artery territory on the right side of the head 1 day after the procedure (Figure 1). These signs were reported to the outside dermatologist who performed the procedure, but they were not assumed to be adverse events at that time.
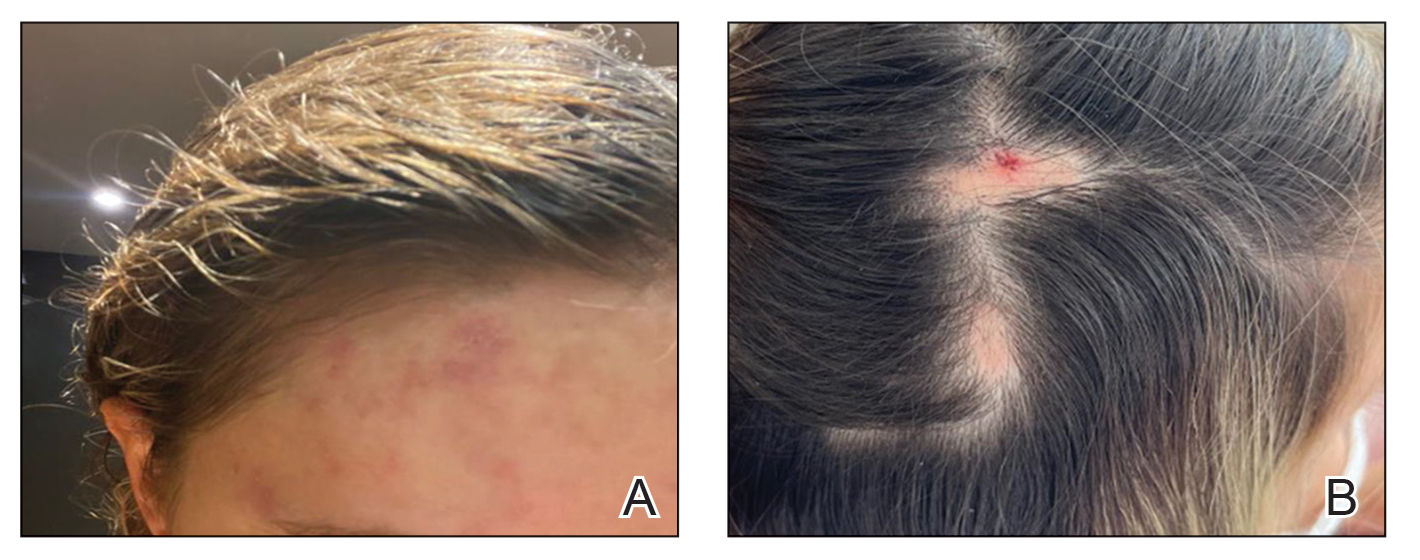
The condition persisted for 4 days followed by the development of an irregular 3×2-cm patch of alopecia on the right parietal scalp. A 3-day course of self-administered oral prednisolone 0.2 mg/kg/d was prescribed.
Twenty-seven days after the procedure, the patient presented to our trichology clinic for evaluation of a single patch of nonscarring alopecia on the right parietal scalp. Trichoscopy showed multiple yellow and black dots, broken hairs, pigment deposits, and an erythematous background mainly composed of linear telangiectatic vessels (Figure 2). Histopathologic analysis revealed a lymphocytic inflammatory infiltrate surrounding the follicular units that was compatible with an alopecia areata–like pattern as well as PLLA deposits in the subcutaneous tissue forming foreign body granulomas (Figure 3). The diagnosis of PLLA-induced alopecia was made based on the detection of PLLA at the biopsy site within the patchy alopecia.
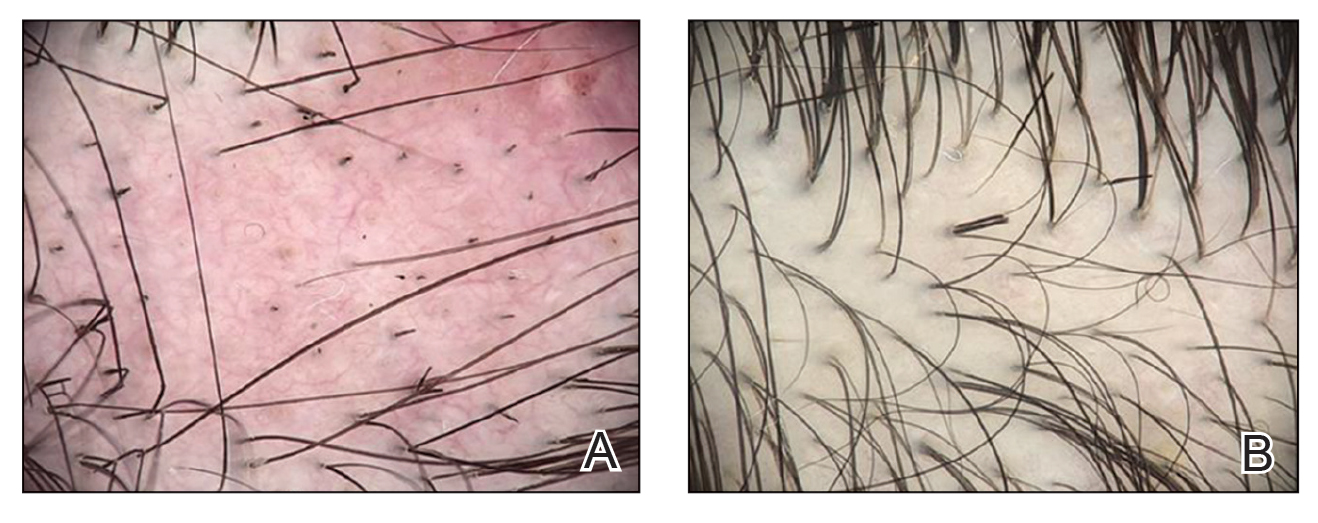
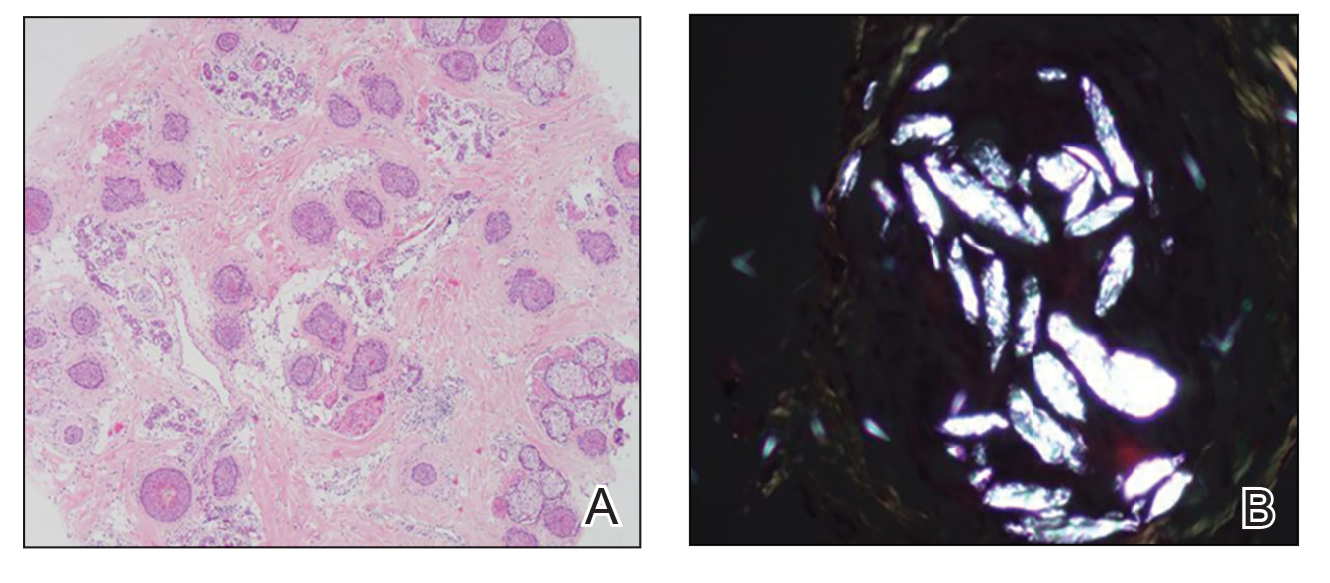
Intralesional triamcinolone acetonide 5 mg/mL was administered at 1-cm intervals in the subdermal space (0.1 mL/puncture site). After 14 days, the patient developed an additional patch of alopecia in the same vascular territory as the right temporal artery, positioned just beneath the initial patch, with similar trichoscopy findings. The patches were treated with intralesional triamcinolone acetonide for 3 additional sessions, administered every 4 weeks. Long-term monitoring of the patient revealed regrowth with comparable hair count to the unaffected contralateral scalp, indicative of a nonscarring alopecia.
Comment
Poly-L-lactic acid is a biostimulator synthesized from the α-hydroxy acid family in 1954 that has been safely used in suture materials, resorbable plates, and orthopedic screws.4 Alopecia has been reported as a systemic allergic reaction to biodegradable screws following an orthopedic procedure.5 Prior reports of embolization and retinal ischemia with PLLA have raised concerns regarding its occlusive potential.6-9
Approved by the US Food and Drug Administration in 2004 for soft tissue restoration in HIV-related lipoatrophy, PLLA was expanded to cosmetic applications in 2009. As previously reported with HA fillers, we hypothesize that extravascular compression resulting from the placement of the filler material (due to the volume injected in the scalp area) contributes to the development of alopecia plus PLLA embolism–induced ischemic alopecia in the affected areas.10 In our case, the diagnosis of PLLA-induced alopecia was confirmed based on the finding of the filler material in the subcutaneous tissue on histopathology, probably due to embolization. Moreover, trichoscopic findings were all similar to those described after HA embolization.11 The features found in our patient due to the PLLA local reaction were similar to those seen in other conditions such as alopecia areata, pressure alopecia, and chemotherapy-induced alopecia; therefore, histopathology confirmation is mandatory in cases of hair loss associated with PLLA.
The emergence of a secondary patch of alopecia prompts consideration of an intrinsic late inflammatory propensity of PLLA. Immune cells recognize PLLA as a foreign body, and subclinical inflammatory foreign body reactions can cause PLLA-induced collagen synthesis.12 This phenomenon underscores the need for further investigation into the immunologic implications of PLLA in alopecia pathogenesis.
The angiogenic properties of the anagen phase require an adequate blood supply for effective hair growth; therefore, the lack of blood and nutrient supply to the hair bulb triggers miniaturization, a possible explanation for the hair thinning found in the alopecic patch.13
Conclusion
Alopecia as an adverse effect of cosmetic procedures can be distressing for patients, even when reversible. A detailed understanding of scalp anatomy is critical for satisfactory outcomes with aesthetic procedures. Physicians must pay attention to the amount and area of material injected in order to avoid possible mechanisms of ischemia—embolization and/or extravascular compression—especially in highly vascularized areas.
We present a rare report of alopecia as an adverse event of PLLA injection. Dermatologists must be aware of this rare condition, and trichoscopy combined with histopathologic analysis are encouraged for early recognition and proper management.
- Issa NT, Kaiser M, Martinez-Velasco A, et al. Alopecia after cosmetic injection procedures: a review. Dermatol Surg. 2022;48:855-861.
- Alopecia with foreign body granulomas induced by Radiesse injection: a case report. J Cosmet Laser Ther. 2018;20:462-464.
- Munia C, Parada M, de Alvarenga Morais MH. Changes in facial morphology using poly-L-lactic acid application according to vector technique: a case series. J Clin Aesthet Dermatol. 2022;15:38-42.
- Attenello NH, Maas CS. Injectable fillers: review of material and properties. Facial Plast Surg. 2015;31:29-34.
- Mastrokalos DS, Paessler HH. Allergic reaction to biodegradable interference poly-L-lactic acid screws after anterior cruciate ligament reconstruction with bone-patellar tendon-bone graft. Arthroscopy. 2008;24:732-733.
- Wu CW, Wu HJ. Retinal artery occlusion following cosmetic injection of poly-L-lactic acid. Taiwan J Ophthalmol. 2021;11:317-320.
- Yuan JT, Chang TW, Yu SS, et al. Mental artery occlusion from poly-L-lactic acid injection at the lateral chin. Dermatol Surg. 2017;43:1402-1405.
- Ragam A, Agemy SA, Dave SB, et al. Ipsilateral ophthalmic and cerebral infarctions after cosmetic polylactic acid injection into the forehead. J Neuroophthalmol. 2017;37:77-80.
- Witmanowski H, Błochowiak K. Another face of dermal fillers. Postepy Dermatol Alergol. 2020;37:651-659.
- Yang Q, Qiu L, Yi C, et al. Reversible alopecia with localized scalp necrosis after accidental embolization of the parietal artery with hyaluronic acid. Aesthetic Plast Surg. 2017;41:695-699.
- Asz-Sigall D, Iñigo-Gomez K, Ortega-Springall MF, et al. Alopecia secondary to hyaluronic acid embolization: trichoscopic findings. Skin Appendage Disord. 2019;5:396-400.
- Oh S, Lee JH, Kim HM, et al. Poly-L-lactic acid fillers improved dermal collagen synthesis by modulating M2 macrophage polarization in aged animal skin. Cells. 2023;12:1320. doi:10.3390/cells12091320
- Natarelli N, Gahoonia N, Sivamani RK. Integrative and mechanistic approach to the hair growth cycle and hair loss. J Clin Med. 2023;12:893.2. Liu RF, Kuo TT, Chao YY, et al.
Cosmetic procedures carry inherent risks of adverse events. Transient and permanent alopecia are rare complications of these procedures. Although they have not been fully elucidated, several pathologic mechanisms for hair loss following cosmetic procedures have been proposed, including extravascular compression (a phenomenon that has been well documented in bedridden patients) as well as intravascular occlusion leading to inflammation and necrosis, which has been associated with hyaluronic acid (HA) fillers.¹ Cases of alopecia also have been reported following mesotherapy and calcium hydroxyapatite, deoxycholic acid, and botulinum toxin injections.² We report a case of alopecia resulting from poly-L-lactic acid (PLLA) injection in a 35-year-old woman with the intent to raise awareness of this rare adverse event.
Case Report
A healthy 35-year-old woman received aesthetic PLLA injections on the face and frontal hairline performed by an outside dermatologist using the vector technique. During the procedure, the patient experienced intense itchiness at the right temporal artery vascular territory and reported a substantial headache the next day. She also presented with erythema and edema of the frontal and right parietal scalp with a well-delimited livedoid vascular area along the temporal artery territory on the right side of the head 1 day after the procedure (Figure 1). These signs were reported to the outside dermatologist who performed the procedure, but they were not assumed to be adverse events at that time.

The condition persisted for 4 days followed by the development of an irregular 3×2-cm patch of alopecia on the right parietal scalp. A 3-day course of self-administered oral prednisolone 0.2 mg/kg/d was prescribed.
Twenty-seven days after the procedure, the patient presented to our trichology clinic for evaluation of a single patch of nonscarring alopecia on the right parietal scalp. Trichoscopy showed multiple yellow and black dots, broken hairs, pigment deposits, and an erythematous background mainly composed of linear telangiectatic vessels (Figure 2). Histopathologic analysis revealed a lymphocytic inflammatory infiltrate surrounding the follicular units that was compatible with an alopecia areata–like pattern as well as PLLA deposits in the subcutaneous tissue forming foreign body granulomas (Figure 3). The diagnosis of PLLA-induced alopecia was made based on the detection of PLLA at the biopsy site within the patchy alopecia.


Intralesional triamcinolone acetonide 5 mg/mL was administered at 1-cm intervals in the subdermal space (0.1 mL/puncture site). After 14 days, the patient developed an additional patch of alopecia in the same vascular territory as the right temporal artery, positioned just beneath the initial patch, with similar trichoscopy findings. The patches were treated with intralesional triamcinolone acetonide for 3 additional sessions, administered every 4 weeks. Long-term monitoring of the patient revealed regrowth with comparable hair count to the unaffected contralateral scalp, indicative of a nonscarring alopecia.
Comment
Poly-L-lactic acid is a biostimulator synthesized from the α-hydroxy acid family in 1954 that has been safely used in suture materials, resorbable plates, and orthopedic screws.4 Alopecia has been reported as a systemic allergic reaction to biodegradable screws following an orthopedic procedure.5 Prior reports of embolization and retinal ischemia with PLLA have raised concerns regarding its occlusive potential.6-9
Approved by the US Food and Drug Administration in 2004 for soft tissue restoration in HIV-related lipoatrophy, PLLA was expanded to cosmetic applications in 2009. As previously reported with HA fillers, we hypothesize that extravascular compression resulting from the placement of the filler material (due to the volume injected in the scalp area) contributes to the development of alopecia plus PLLA embolism–induced ischemic alopecia in the affected areas.10 In our case, the diagnosis of PLLA-induced alopecia was confirmed based on the finding of the filler material in the subcutaneous tissue on histopathology, probably due to embolization. Moreover, trichoscopic findings were all similar to those described after HA embolization.11 The features found in our patient due to the PLLA local reaction were similar to those seen in other conditions such as alopecia areata, pressure alopecia, and chemotherapy-induced alopecia; therefore, histopathology confirmation is mandatory in cases of hair loss associated with PLLA.
The emergence of a secondary patch of alopecia prompts consideration of an intrinsic late inflammatory propensity of PLLA. Immune cells recognize PLLA as a foreign body, and subclinical inflammatory foreign body reactions can cause PLLA-induced collagen synthesis.12 This phenomenon underscores the need for further investigation into the immunologic implications of PLLA in alopecia pathogenesis.
The angiogenic properties of the anagen phase require an adequate blood supply for effective hair growth; therefore, the lack of blood and nutrient supply to the hair bulb triggers miniaturization, a possible explanation for the hair thinning found in the alopecic patch.13
Conclusion
Alopecia as an adverse effect of cosmetic procedures can be distressing for patients, even when reversible. A detailed understanding of scalp anatomy is critical for satisfactory outcomes with aesthetic procedures. Physicians must pay attention to the amount and area of material injected in order to avoid possible mechanisms of ischemia—embolization and/or extravascular compression—especially in highly vascularized areas.
We present a rare report of alopecia as an adverse event of PLLA injection. Dermatologists must be aware of this rare condition, and trichoscopy combined with histopathologic analysis are encouraged for early recognition and proper management.
Cosmetic procedures carry inherent risks of adverse events. Transient and permanent alopecia are rare complications of these procedures. Although they have not been fully elucidated, several pathologic mechanisms for hair loss following cosmetic procedures have been proposed, including extravascular compression (a phenomenon that has been well documented in bedridden patients) as well as intravascular occlusion leading to inflammation and necrosis, which has been associated with hyaluronic acid (HA) fillers.¹ Cases of alopecia also have been reported following mesotherapy and calcium hydroxyapatite, deoxycholic acid, and botulinum toxin injections.² We report a case of alopecia resulting from poly-L-lactic acid (PLLA) injection in a 35-year-old woman with the intent to raise awareness of this rare adverse event.
Case Report
A healthy 35-year-old woman received aesthetic PLLA injections on the face and frontal hairline performed by an outside dermatologist using the vector technique. During the procedure, the patient experienced intense itchiness at the right temporal artery vascular territory and reported a substantial headache the next day. She also presented with erythema and edema of the frontal and right parietal scalp with a well-delimited livedoid vascular area along the temporal artery territory on the right side of the head 1 day after the procedure (Figure 1). These signs were reported to the outside dermatologist who performed the procedure, but they were not assumed to be adverse events at that time.

The condition persisted for 4 days followed by the development of an irregular 3×2-cm patch of alopecia on the right parietal scalp. A 3-day course of self-administered oral prednisolone 0.2 mg/kg/d was prescribed.
Twenty-seven days after the procedure, the patient presented to our trichology clinic for evaluation of a single patch of nonscarring alopecia on the right parietal scalp. Trichoscopy showed multiple yellow and black dots, broken hairs, pigment deposits, and an erythematous background mainly composed of linear telangiectatic vessels (Figure 2). Histopathologic analysis revealed a lymphocytic inflammatory infiltrate surrounding the follicular units that was compatible with an alopecia areata–like pattern as well as PLLA deposits in the subcutaneous tissue forming foreign body granulomas (Figure 3). The diagnosis of PLLA-induced alopecia was made based on the detection of PLLA at the biopsy site within the patchy alopecia.


Intralesional triamcinolone acetonide 5 mg/mL was administered at 1-cm intervals in the subdermal space (0.1 mL/puncture site). After 14 days, the patient developed an additional patch of alopecia in the same vascular territory as the right temporal artery, positioned just beneath the initial patch, with similar trichoscopy findings. The patches were treated with intralesional triamcinolone acetonide for 3 additional sessions, administered every 4 weeks. Long-term monitoring of the patient revealed regrowth with comparable hair count to the unaffected contralateral scalp, indicative of a nonscarring alopecia.
Comment
Poly-L-lactic acid is a biostimulator synthesized from the α-hydroxy acid family in 1954 that has been safely used in suture materials, resorbable plates, and orthopedic screws.4 Alopecia has been reported as a systemic allergic reaction to biodegradable screws following an orthopedic procedure.5 Prior reports of embolization and retinal ischemia with PLLA have raised concerns regarding its occlusive potential.6-9
Approved by the US Food and Drug Administration in 2004 for soft tissue restoration in HIV-related lipoatrophy, PLLA was expanded to cosmetic applications in 2009. As previously reported with HA fillers, we hypothesize that extravascular compression resulting from the placement of the filler material (due to the volume injected in the scalp area) contributes to the development of alopecia plus PLLA embolism–induced ischemic alopecia in the affected areas.10 In our case, the diagnosis of PLLA-induced alopecia was confirmed based on the finding of the filler material in the subcutaneous tissue on histopathology, probably due to embolization. Moreover, trichoscopic findings were all similar to those described after HA embolization.11 The features found in our patient due to the PLLA local reaction were similar to those seen in other conditions such as alopecia areata, pressure alopecia, and chemotherapy-induced alopecia; therefore, histopathology confirmation is mandatory in cases of hair loss associated with PLLA.
The emergence of a secondary patch of alopecia prompts consideration of an intrinsic late inflammatory propensity of PLLA. Immune cells recognize PLLA as a foreign body, and subclinical inflammatory foreign body reactions can cause PLLA-induced collagen synthesis.12 This phenomenon underscores the need for further investigation into the immunologic implications of PLLA in alopecia pathogenesis.
The angiogenic properties of the anagen phase require an adequate blood supply for effective hair growth; therefore, the lack of blood and nutrient supply to the hair bulb triggers miniaturization, a possible explanation for the hair thinning found in the alopecic patch.13
Conclusion
Alopecia as an adverse effect of cosmetic procedures can be distressing for patients, even when reversible. A detailed understanding of scalp anatomy is critical for satisfactory outcomes with aesthetic procedures. Physicians must pay attention to the amount and area of material injected in order to avoid possible mechanisms of ischemia—embolization and/or extravascular compression—especially in highly vascularized areas.
We present a rare report of alopecia as an adverse event of PLLA injection. Dermatologists must be aware of this rare condition, and trichoscopy combined with histopathologic analysis are encouraged for early recognition and proper management.
- Issa NT, Kaiser M, Martinez-Velasco A, et al. Alopecia after cosmetic injection procedures: a review. Dermatol Surg. 2022;48:855-861.
- Alopecia with foreign body granulomas induced by Radiesse injection: a case report. J Cosmet Laser Ther. 2018;20:462-464.
- Munia C, Parada M, de Alvarenga Morais MH. Changes in facial morphology using poly-L-lactic acid application according to vector technique: a case series. J Clin Aesthet Dermatol. 2022;15:38-42.
- Attenello NH, Maas CS. Injectable fillers: review of material and properties. Facial Plast Surg. 2015;31:29-34.
- Mastrokalos DS, Paessler HH. Allergic reaction to biodegradable interference poly-L-lactic acid screws after anterior cruciate ligament reconstruction with bone-patellar tendon-bone graft. Arthroscopy. 2008;24:732-733.
- Wu CW, Wu HJ. Retinal artery occlusion following cosmetic injection of poly-L-lactic acid. Taiwan J Ophthalmol. 2021;11:317-320.
- Yuan JT, Chang TW, Yu SS, et al. Mental artery occlusion from poly-L-lactic acid injection at the lateral chin. Dermatol Surg. 2017;43:1402-1405.
- Ragam A, Agemy SA, Dave SB, et al. Ipsilateral ophthalmic and cerebral infarctions after cosmetic polylactic acid injection into the forehead. J Neuroophthalmol. 2017;37:77-80.
- Witmanowski H, Błochowiak K. Another face of dermal fillers. Postepy Dermatol Alergol. 2020;37:651-659.
- Yang Q, Qiu L, Yi C, et al. Reversible alopecia with localized scalp necrosis after accidental embolization of the parietal artery with hyaluronic acid. Aesthetic Plast Surg. 2017;41:695-699.
- Asz-Sigall D, Iñigo-Gomez K, Ortega-Springall MF, et al. Alopecia secondary to hyaluronic acid embolization: trichoscopic findings. Skin Appendage Disord. 2019;5:396-400.
- Oh S, Lee JH, Kim HM, et al. Poly-L-lactic acid fillers improved dermal collagen synthesis by modulating M2 macrophage polarization in aged animal skin. Cells. 2023;12:1320. doi:10.3390/cells12091320
- Natarelli N, Gahoonia N, Sivamani RK. Integrative and mechanistic approach to the hair growth cycle and hair loss. J Clin Med. 2023;12:893.2. Liu RF, Kuo TT, Chao YY, et al.
- Issa NT, Kaiser M, Martinez-Velasco A, et al. Alopecia after cosmetic injection procedures: a review. Dermatol Surg. 2022;48:855-861.
- Alopecia with foreign body granulomas induced by Radiesse injection: a case report. J Cosmet Laser Ther. 2018;20:462-464.
- Munia C, Parada M, de Alvarenga Morais MH. Changes in facial morphology using poly-L-lactic acid application according to vector technique: a case series. J Clin Aesthet Dermatol. 2022;15:38-42.
- Attenello NH, Maas CS. Injectable fillers: review of material and properties. Facial Plast Surg. 2015;31:29-34.
- Mastrokalos DS, Paessler HH. Allergic reaction to biodegradable interference poly-L-lactic acid screws after anterior cruciate ligament reconstruction with bone-patellar tendon-bone graft. Arthroscopy. 2008;24:732-733.
- Wu CW, Wu HJ. Retinal artery occlusion following cosmetic injection of poly-L-lactic acid. Taiwan J Ophthalmol. 2021;11:317-320.
- Yuan JT, Chang TW, Yu SS, et al. Mental artery occlusion from poly-L-lactic acid injection at the lateral chin. Dermatol Surg. 2017;43:1402-1405.
- Ragam A, Agemy SA, Dave SB, et al. Ipsilateral ophthalmic and cerebral infarctions after cosmetic polylactic acid injection into the forehead. J Neuroophthalmol. 2017;37:77-80.
- Witmanowski H, Błochowiak K. Another face of dermal fillers. Postepy Dermatol Alergol. 2020;37:651-659.
- Yang Q, Qiu L, Yi C, et al. Reversible alopecia with localized scalp necrosis after accidental embolization of the parietal artery with hyaluronic acid. Aesthetic Plast Surg. 2017;41:695-699.
- Asz-Sigall D, Iñigo-Gomez K, Ortega-Springall MF, et al. Alopecia secondary to hyaluronic acid embolization: trichoscopic findings. Skin Appendage Disord. 2019;5:396-400.
- Oh S, Lee JH, Kim HM, et al. Poly-L-lactic acid fillers improved dermal collagen synthesis by modulating M2 macrophage polarization in aged animal skin. Cells. 2023;12:1320. doi:10.3390/cells12091320
- Natarelli N, Gahoonia N, Sivamani RK. Integrative and mechanistic approach to the hair growth cycle and hair loss. J Clin Med. 2023;12:893.2. Liu RF, Kuo TT, Chao YY, et al.
Practice Points
- Alopecia is a potential adverse event of poly-L-lactic acid (PLLA) injection, and prior reports of embolization and retinal ischemia with PLLA use raise the concern of its occlusive potential.
- The combination of extravascular compression due to the presence of the filler material in the subcutaneous tissue as well as intravascular PLLA embolism may contribute to tissue ischemia–induced alopecia in the affected areas.
- Poly-L-lactic acid also may cause a local inflammatory reaction that is alopecia areata–like, which would explain its similar trichoscopy findings.
Hospital Dermatology: Review of Research in 2023-2024
Inpatient consultative dermatology has advanced as a subspecialty and increasingly gained recognition in recent years. Since its founding in 2009, the Society of Dermatology Hospitalists has fostered research and education in hospital dermatology. Last year, we reviewed the 2022-2023 literature with a focus on developments in severe cutaneous adverse reactions, supportive oncodermatology, cost of inpatient services, and teledermatology.1 In this review, we highlight 3 areas of interest from the 2023-2024 literature: severe cutaneous adverse drug reactions, skin and soft tissue infections, and autoimmune blistering diseases (AIBDs).
Severe Cutaneous Adverse Drug Reactions
Adverse drug reactions are among the most common diagnoses encountered by inpatient dermatology consultants.2,3 Severe cutaneous adverse drug reactions are associated with substantial morbidity and mortality. Efforts to characterize these conditions and standardize their diagnosis and management continue to be a major focus of ongoing research.
A single-center retrospective analysis of 102 cases of drug reaction with eosinophilia and systemic symptoms (DRESS) syndrome evaluated differences in clinical manifestations depending on the culprit drug, offering insights into the heterogeneity of DRESS syndrome and the potential for diagnostic uncertainty.4 The shortest median latency was observed in a case caused by penicillin and cephalosporins (12 and 18 days, respectively), while DRESS syndrome secondary to allopurinol had the longest median latency (36 days). Nonsteroidal anti-inflammatory drug–induced DRESS syndrome was associated with the shortest hospital stay (6.5 days), while cephalosporin and vancomycin cases had the highest mortality rates.4
In the first international Delphi consensus study on the diagnostic workup, severity assessment, and management of DRESS syndrome, 54 dermatology and/or allergy experts reached consensus on 93 statements.5 Specific recommendations included basic evaluation with complete blood count with differential, kidney and liver function parameters, and electrocardiogram for all patients with suspected DRESS syndrome, with additional complementary workup considered in patients with evidence of specific organ damage and/or severe disease. In the proposed DRESS syndrome severity grading scheme, laboratory values that reached consensus for inclusion were hemoglobin, neutrophil, and platelet counts and creatinine, transaminases, and alkaline phosphatase levels. Although treatment of DRESS syndrome should be based on assessed disease severity, treatment with corticosteroids should be initiated in all patients with confirmed DRESS syndrome. Cyclosporine, antibodies interfering with the IL-5 axis, and intravenous immunoglobulins can be considered in patients with corticosteroid-refractory DRESS syndrome, and antiviral treatment can be considered in patients with a high serum cytomegalovirus viral load. Regularly following up with laboratory evaluation of involved organs; screening for autoantibodies, thyroid dysfunction, and steroid adverse effects; and offering of psychological support also were consensus recommendations.5
Identifying causative agents in drug hypersensitivity reactions remains challenging. A retrospective cohort study of 48 patients with Stevens-Johnson syndrome (SJS)/toxic epidermal necrolysis (TEN) highlighted the need for a systematic unbiased approach to identifying culprit drugs. Using the RegiSCAR database and algorithm for drug causality for epidermal necrolysis to analyze the cohort, more than half of causative agents were determined to be different from those initially identified by the treating physicians. Nine additional suspected culprit drugs were identified, while 43 drugs initially identified as allergens were exonerated.6
Etiology-associated definitions for blistering reactions in children have been proposed to replace the existing terms Stevens-Johnson syndrome, toxic epidermal necrolysis, and others.7 Investigators in a recent study reclassified cases of SJS and TEN as reactive infectious mucocutaneous eruption (RIME) or drug-induced epidermal necrolysis (DEN), respectively. In RIME cases, Mycoplasma pneumoniae was the most commonly identified trigger, and in DEN cases, anticonvulsants were the most common class of culprit medications. Cases of RIME were less severe and were most often treated with antibiotics, whereas patients with DEN were more likely to receive supportive care, corticosteroids, intravenous immunoglobulins, and other immunosuppressive therapies.7
In addition to causing acute devastating mucocutaneous complications, SJS and TEN have long-lasting effects that require ongoing care. In a cohort of 6552 incident SJS/TEN cases over an 11-year period, survivors of SJS/TEN endured a mean loss of 9.4 years in life expectancy and excess health care expenditures of $3752 per year compared with age- and sex-matched controls. Patients with more severe disease, comorbid malignancy, diabetes, end-stage renal disease, or SJS/TEN sequelae experienced greater loss in life expectancy and lifetime health care expenditures.8 Separately, a qualitative study investigating the psychological impact of SJS/TEN in pediatric patients described sequelae including night terrors, posttraumatic stress disorder, depression, and anxiety for many years after the acute phase. Many patients reported a desire for increased support for their physical and emotional needs following hospital discharge.9
Skin and Soft Tissue Infections: Diagnosis, Management, and Prevention
Dermatology consultation has been shown to be a cost-effective intervention to improve outcomes in hospitalized patients with skin and soft tissue infections.10,11 In particular, cellulitis frequently is misdiagnosed, leading to unnecessary antibiotic use, hospitalizations, and major health care expenditures.12 Recognizing this challenge, researchers have worked to develop objective tools to improve diagnostic accuracy. In a large prospective prognostic validation study, Pulia et al13 found that thermal imaging alone or in combination with the ALT-70 prediction model (asymmetry, leukocytosis, tachycardia, and age ≥70 years) could be used successfully to reduce overdiagnosis of cellulitis. Both thermal imaging and the ALT-70 prediction model demonstrated robust sensitivity (93.5% and 98.8%, respectively) but low specificity (38.4% and 22.0%, respectively, and 53.9% when combined).13
In a systematic review, Kovacs et al14 analyzed case reports of pseudocellulitis caused by chemotherapeutic medications. Of the 81 cases selected, 58 (71.6%) were associated with gemcitabine, with the remaining 23 (28.4%) attributed to pemetrexed. Within this group, two-thirds of the patients received antibiotic treatment prior to receiving the correct diagnosis, and 36% experienced interruptions to their oncologic therapies. In contrast to infectious cellulitis, which tends to be unilateral and associated with elevated erythrocyte sedimentation rate or C-reactive protein, most chemotherapy-induced pseudocellulitis cases occurred bilaterally on the lower extremities, while erythrocyte sedimentation rate and C-reactive protein seldom were elevated.14
Necrotizing soft tissue infections (NSTIs) are severe life-threatening conditions characterized by widespread tissue destruction, signs of systemic toxicity, hemodynamic collapse, organ failure, and high mortality. Surgical inspection along with intraoperative tissue culture is the gold standard for diagnosis. Early detection, prompt surgical intervention, and appropriate antibiotic treatment are essential to reduce mortality and improve outcomes.15 A retrospective study of patients with surgically confirmed NSTIs assessed the incidence and risk factors for recurrence within 1 year following an initial NSTI of the lower extremity. Among 93 included patients, 32 (34.4%) had recurrence within 1 year, and more than half of recurrences occurred in the first 3 months (median, 66 days). The comparison of patients with and without recurrence showed similar proportions of antibiotic prophylaxis use after the first NSTI. There was significantly less compression therapy use (33.3% vs 62.3%; P=.13) and more negative pressure wound therapy use (83.3% vs 63.3%; P=.03) in the recurrence group, though the authors acknowledged that factors such as severity of pain and size of soft tissue defect may have affected the decisions for compression and negative pressure wound therapy.16
Residents of nursing homes are a particularly vulnerable population at high risk for health care–associated infections due to older age and a higher likelihood of having wounds, indwelling medical devices, and/or coexisting conditions.17 One cluster-randomized trial compared universal decolonization with routine-care bathing practices in nursing homes (N=28,956 residents). Decolonization entailed the use of chlorhexidine for all routine bathing and showering and administration of nasal povidone-iodine twice daily for the first 5 days after admission and then twice daily for 5 days every other week. Transfer to a hospital due to infection decreased from 62.9% to 52.2% with decolonization, for a difference in risk ratio of 16.6% (P<.001) compared with routine care. Additionally, the difference in risk ratio of the secondary end point (transfer to a hospital for any reason) was 14.6%. The number needed to treat was 9.7 to prevent 1 infection-related hospitalization and 8.9 to prevent 1 hospitalization for any reason.17
Autoimmune Blistering Diseases
Although rare, AIBDs are potentially life-threatening cutaneous diseases that often require inpatient management. While corticosteroids remain the mainstay of initial AIBD management, rituximab is now well recognized as the steroid-sparing treatment of choice for patients with moderate to severe pemphigus. In a long-term follow-up study of Ritux 318—the trial that led to the US Food and Drug Administration approval of rituximab in the treatment of moderate to severe pemphigus vulgaris—researchers assessed the long-term efficacy and safety of rituximab as a first-line treatment in patients with pemphigus.19 The 5- and 7-year disease-free survival rates without corticosteroid therapy for patients treated with rituximab were 76.7% and 72.1%, respectively, compared with 35.3% and 35.3% in those treated with prednisone alone (P<.001). Fewer serious adverse events were reported in those treated with rituximab plus prednisone compared with those treated with prednisone alone. None of the patients who maintained complete remission off corticosteroid therapy received any additional maintenance infusions of rituximab after the end of the Ritux 3 regimen (1 g of rituximab at day 0 and day 14, then 500 mg at months 12 and 18).19
By contrast, treatment of severe bullous pemphigoid (BP) often is less clear-cut, as no single therapeutic option has been shown to be superior to other immunomodulatory and immunosuppressive regimens, and the medical comorbidities of elderly patients with BP can be limiting. Fortunately, newer therapies with favorable safety profiles have emerged in recent years. In a multicenter retrospective study, 100 patients with BP received omalizumab after previously failing to respond to at least one alternative therapy. Disease control was obtained after a median of 10 days, and complete remission was achieved in 77% of patients in a median time of 3 months.20 In a multicenter retrospective cohort study of 146 patients with BP treated with dupilumab following the atopic dermatitis dosing schedule (one 600-mg dose followed by 300 mg every 2 weeks), disease control was achieved in a median of 14 days, while complete remission was achieved in 35.6% of patients, with 8.9% relapsing during the observation period.21 A retrospective case series of 30 patients with BP treated with dupilumab with maintenance dosing frequency tailored to individual patient response showed complete remission or marked response in 76.7% (23/30) of patients.22 A phase 2/3 randomized controlled trial of dupilumab in BP is currently ongoing (ClinicalTrials.gov identifier NCT04206553).
Pemphigoid gestationis is a rare autoimmune subepidermal bullous dermatosis of pregnancy that may be difficult to distinguish clinically from polymorphic eruption of pregnancy but confers notably different maternal and fetal risks. Researchers developed and validated a scoring system using clinical factors—history of pemphigoid gestationis, primigravidae, timing of rash onset, and specific clinical examination findings—that was able to differentiate between the 2 diseases with 79% sensitivity, 95% specificity, and an area under the curve of 0.93 without the need for advanced immunologic testing.23
Final Thoughts
Highlights of the literature from 2023-2024 demonstrate advancements in hospital-based dermatology as well as ongoing challenges. This year’s review emphasizes key developments in severe cutaneous adverse drug reactions, skin and soft tissue infections, and AIBDs. Continued expansion of knowledge in these areas and others informs patient care and demonstrates the value of dermatologic expertise in the inpatient setting.
- Berk-Krauss J, Micheletti RG. Hospital dermatology: review of research in 2022-2023. Cutis. 2023;112:236-239.
- Falanga V, Schachner LA, Rae V, et al. Dermatologic consultations in the hospital setting. Arch Dermatol. 1994;130:1022-1025.
- Kroshinsky D, Cotliar J, Hughey LC, et al. Association of dermatology consultation with accuracy of cutaneous disorder diagnoses in hospitalized patients: a multicenter analysis. JAMA Dermatol. 2016;152:477-480.
- Blumenthal KG, Alvarez-Arango S, Kroshinsky D, et al. Drug reaction eosinophilia and systemic symptoms: clinical phenotypic patterns according to causative drug. J Am Acad Dermatol. 2024;90:1240-1242.
- Brüggen MC, Walsh S, Ameri MM, et al. Management of adult patients with drug reaction with eosinophilia and systemic symptoms: a Delphi-based international consensus. JAMA Dermatol. 2024;160:37-44.
- Li DJ, Velasquez GA, Romar GA, et al. Assessment of need for improved identification of a culprit drug in Stevens-Johnson syndrome/toxic epidermal necrolysis. JAMA Dermatol. 2023;159:830-836.
- Martinez-Cabriales S, Coulombe J, Aaron M, et al. Preliminary summary and reclassification of cases from the Pediatric Research of Management in Stevens-Johnson syndrome and Epidermonecrolysis (PROMISE) study: a North American, multisite retrospective cohort. J Am Acad Dermatol. 2024;90:635-637.
- Chiu YM, Chiu HY. Lifetime risk, life expectancy, loss-of-life expectancy and lifetime healthcare expenditure for Stevens-Johnson syndrome/toxic epidermal necrolysis in Taiwan: follow-up of a nationwide cohort from 2008 to 2019. Br J Dermatol. 2023;189:553-560.
- Phillips C, Russell E, McNiven A, et al. A qualitative study of psychological morbidity in paediatric survivors of Stevens-Johnson syndrome/toxic epidermal necrolysis. Br J Dermatol. 2024;191:293-295.
- Li DG, Xia FD, Khosravi H, et al. Outcomes of early dermatology consultation for inpatients diagnosed with cellulitis. JAMA Dermatol. 2018;154:537-543.
- Milani-Nejad N, Zhang M, Kaffenberger BH. Association of dermatology consultations with patient care outcomes in hospitalized patients with inflammatory skin diseases. JAMA Dermatol. 2017;153:523-528.
- Weng QY, Raff AB, Cohen JM, et al. Costs and consequences associated with misdiagnosed lower extremity cellulitis. JAMA Dermatol. 2017;153:141-146.
- Pulia MS, Schwei RJ, Alexandridis R, et al. Validation of thermal imaging and the ALT-70 prediction model to differentiate cellulitis from pseudocellulitis. JAMA Dermatol. 2024;160:511-517.
- Kovacs LD, O’Donoghue M, Cogen AL. Chemotherapy-induced pseudocellulitis without prior radiation exposure: a systematic review. JAMA Dermatol. 2023;159:870-874.
- Yildiz H, Yombi JC. Necrotizing soft-tissue infections. comment. N Engl J Med. 2018;378:970.
- Traineau H, Charpentier C, Lepeule R, et al. First-year recurrence rate of skin and soft tissue infections following an initial necrotizing soft tissue infection of the lower extremities: a retrospective cohort study of 93 patients. J Am Acad Dermatol. 2023;88:1360-1363.
- Miller LG, McKinnell JA, Singh RD, et al. Decolonization in nursing homes to prevent infection and hospitalization. N Engl J Med. 2023;389:1766-1777.
- Joly P, Maho-Vaillant M, Prost-Squarcioni C, et al; French Study Group on Autoimmune Bullous Skin Diseases. First-line rituximab combined with short-term prednisone versus prednisone alone for the treatment of pemphigus (Ritux 3): a prospective, multicentre, parallel-group, open-label randomised trial. Lancet. 2017;389:2031-2040.
- Tedbirt B, Maho-Vaillant M, Houivet E, et al; French Reference Center for Autoimmune Blistering Diseases MALIBUL. Sustained remission without corticosteroids among patients with pemphigus who had rituximab as first-line therapy: follow-up of the Ritux 3 Trial. JAMA Dermatol. 2024;160:290-296.
- Chebani R, Lombart F, Chaby G, et al; French Study Group on Autoimmune Bullous Diseases. Omalizumab in the treatment of bullous pemphigoid resistant to first-line therapy: a French national multicentre retrospective study of 100 patients. Br J Dermatol. 2024;190:258-265.
- Zhao L, Wang Q, Liang G, et al. Evaluation of dupilumab in patients with bullous pemphigoid. JAMA Dermatol. 2023;159:953-960.
- Miller AC, Temiz LA, Adjei S, et al. Treatment of bullous pemphigoid with dupilumab: a case series of 30 patients. J Drugs Dermatol. 2024;23:E144-E148.
- Xie F, Davis DMR, Baban F, et al. Development and multicenter international validation of a diagnostic tool to differentiate between pemphigoid gestationis and polymorphic eruption of pregnancy. J Am Acad Dermatol. 2023;89:106-113.
Inpatient consultative dermatology has advanced as a subspecialty and increasingly gained recognition in recent years. Since its founding in 2009, the Society of Dermatology Hospitalists has fostered research and education in hospital dermatology. Last year, we reviewed the 2022-2023 literature with a focus on developments in severe cutaneous adverse reactions, supportive oncodermatology, cost of inpatient services, and teledermatology.1 In this review, we highlight 3 areas of interest from the 2023-2024 literature: severe cutaneous adverse drug reactions, skin and soft tissue infections, and autoimmune blistering diseases (AIBDs).
Severe Cutaneous Adverse Drug Reactions
Adverse drug reactions are among the most common diagnoses encountered by inpatient dermatology consultants.2,3 Severe cutaneous adverse drug reactions are associated with substantial morbidity and mortality. Efforts to characterize these conditions and standardize their diagnosis and management continue to be a major focus of ongoing research.
A single-center retrospective analysis of 102 cases of drug reaction with eosinophilia and systemic symptoms (DRESS) syndrome evaluated differences in clinical manifestations depending on the culprit drug, offering insights into the heterogeneity of DRESS syndrome and the potential for diagnostic uncertainty.4 The shortest median latency was observed in a case caused by penicillin and cephalosporins (12 and 18 days, respectively), while DRESS syndrome secondary to allopurinol had the longest median latency (36 days). Nonsteroidal anti-inflammatory drug–induced DRESS syndrome was associated with the shortest hospital stay (6.5 days), while cephalosporin and vancomycin cases had the highest mortality rates.4
In the first international Delphi consensus study on the diagnostic workup, severity assessment, and management of DRESS syndrome, 54 dermatology and/or allergy experts reached consensus on 93 statements.5 Specific recommendations included basic evaluation with complete blood count with differential, kidney and liver function parameters, and electrocardiogram for all patients with suspected DRESS syndrome, with additional complementary workup considered in patients with evidence of specific organ damage and/or severe disease. In the proposed DRESS syndrome severity grading scheme, laboratory values that reached consensus for inclusion were hemoglobin, neutrophil, and platelet counts and creatinine, transaminases, and alkaline phosphatase levels. Although treatment of DRESS syndrome should be based on assessed disease severity, treatment with corticosteroids should be initiated in all patients with confirmed DRESS syndrome. Cyclosporine, antibodies interfering with the IL-5 axis, and intravenous immunoglobulins can be considered in patients with corticosteroid-refractory DRESS syndrome, and antiviral treatment can be considered in patients with a high serum cytomegalovirus viral load. Regularly following up with laboratory evaluation of involved organs; screening for autoantibodies, thyroid dysfunction, and steroid adverse effects; and offering of psychological support also were consensus recommendations.5
Identifying causative agents in drug hypersensitivity reactions remains challenging. A retrospective cohort study of 48 patients with Stevens-Johnson syndrome (SJS)/toxic epidermal necrolysis (TEN) highlighted the need for a systematic unbiased approach to identifying culprit drugs. Using the RegiSCAR database and algorithm for drug causality for epidermal necrolysis to analyze the cohort, more than half of causative agents were determined to be different from those initially identified by the treating physicians. Nine additional suspected culprit drugs were identified, while 43 drugs initially identified as allergens were exonerated.6
Etiology-associated definitions for blistering reactions in children have been proposed to replace the existing terms Stevens-Johnson syndrome, toxic epidermal necrolysis, and others.7 Investigators in a recent study reclassified cases of SJS and TEN as reactive infectious mucocutaneous eruption (RIME) or drug-induced epidermal necrolysis (DEN), respectively. In RIME cases, Mycoplasma pneumoniae was the most commonly identified trigger, and in DEN cases, anticonvulsants were the most common class of culprit medications. Cases of RIME were less severe and were most often treated with antibiotics, whereas patients with DEN were more likely to receive supportive care, corticosteroids, intravenous immunoglobulins, and other immunosuppressive therapies.7
In addition to causing acute devastating mucocutaneous complications, SJS and TEN have long-lasting effects that require ongoing care. In a cohort of 6552 incident SJS/TEN cases over an 11-year period, survivors of SJS/TEN endured a mean loss of 9.4 years in life expectancy and excess health care expenditures of $3752 per year compared with age- and sex-matched controls. Patients with more severe disease, comorbid malignancy, diabetes, end-stage renal disease, or SJS/TEN sequelae experienced greater loss in life expectancy and lifetime health care expenditures.8 Separately, a qualitative study investigating the psychological impact of SJS/TEN in pediatric patients described sequelae including night terrors, posttraumatic stress disorder, depression, and anxiety for many years after the acute phase. Many patients reported a desire for increased support for their physical and emotional needs following hospital discharge.9
Skin and Soft Tissue Infections: Diagnosis, Management, and Prevention
Dermatology consultation has been shown to be a cost-effective intervention to improve outcomes in hospitalized patients with skin and soft tissue infections.10,11 In particular, cellulitis frequently is misdiagnosed, leading to unnecessary antibiotic use, hospitalizations, and major health care expenditures.12 Recognizing this challenge, researchers have worked to develop objective tools to improve diagnostic accuracy. In a large prospective prognostic validation study, Pulia et al13 found that thermal imaging alone or in combination with the ALT-70 prediction model (asymmetry, leukocytosis, tachycardia, and age ≥70 years) could be used successfully to reduce overdiagnosis of cellulitis. Both thermal imaging and the ALT-70 prediction model demonstrated robust sensitivity (93.5% and 98.8%, respectively) but low specificity (38.4% and 22.0%, respectively, and 53.9% when combined).13
In a systematic review, Kovacs et al14 analyzed case reports of pseudocellulitis caused by chemotherapeutic medications. Of the 81 cases selected, 58 (71.6%) were associated with gemcitabine, with the remaining 23 (28.4%) attributed to pemetrexed. Within this group, two-thirds of the patients received antibiotic treatment prior to receiving the correct diagnosis, and 36% experienced interruptions to their oncologic therapies. In contrast to infectious cellulitis, which tends to be unilateral and associated with elevated erythrocyte sedimentation rate or C-reactive protein, most chemotherapy-induced pseudocellulitis cases occurred bilaterally on the lower extremities, while erythrocyte sedimentation rate and C-reactive protein seldom were elevated.14
Necrotizing soft tissue infections (NSTIs) are severe life-threatening conditions characterized by widespread tissue destruction, signs of systemic toxicity, hemodynamic collapse, organ failure, and high mortality. Surgical inspection along with intraoperative tissue culture is the gold standard for diagnosis. Early detection, prompt surgical intervention, and appropriate antibiotic treatment are essential to reduce mortality and improve outcomes.15 A retrospective study of patients with surgically confirmed NSTIs assessed the incidence and risk factors for recurrence within 1 year following an initial NSTI of the lower extremity. Among 93 included patients, 32 (34.4%) had recurrence within 1 year, and more than half of recurrences occurred in the first 3 months (median, 66 days). The comparison of patients with and without recurrence showed similar proportions of antibiotic prophylaxis use after the first NSTI. There was significantly less compression therapy use (33.3% vs 62.3%; P=.13) and more negative pressure wound therapy use (83.3% vs 63.3%; P=.03) in the recurrence group, though the authors acknowledged that factors such as severity of pain and size of soft tissue defect may have affected the decisions for compression and negative pressure wound therapy.16
Residents of nursing homes are a particularly vulnerable population at high risk for health care–associated infections due to older age and a higher likelihood of having wounds, indwelling medical devices, and/or coexisting conditions.17 One cluster-randomized trial compared universal decolonization with routine-care bathing practices in nursing homes (N=28,956 residents). Decolonization entailed the use of chlorhexidine for all routine bathing and showering and administration of nasal povidone-iodine twice daily for the first 5 days after admission and then twice daily for 5 days every other week. Transfer to a hospital due to infection decreased from 62.9% to 52.2% with decolonization, for a difference in risk ratio of 16.6% (P<.001) compared with routine care. Additionally, the difference in risk ratio of the secondary end point (transfer to a hospital for any reason) was 14.6%. The number needed to treat was 9.7 to prevent 1 infection-related hospitalization and 8.9 to prevent 1 hospitalization for any reason.17
Autoimmune Blistering Diseases
Although rare, AIBDs are potentially life-threatening cutaneous diseases that often require inpatient management. While corticosteroids remain the mainstay of initial AIBD management, rituximab is now well recognized as the steroid-sparing treatment of choice for patients with moderate to severe pemphigus. In a long-term follow-up study of Ritux 318—the trial that led to the US Food and Drug Administration approval of rituximab in the treatment of moderate to severe pemphigus vulgaris—researchers assessed the long-term efficacy and safety of rituximab as a first-line treatment in patients with pemphigus.19 The 5- and 7-year disease-free survival rates without corticosteroid therapy for patients treated with rituximab were 76.7% and 72.1%, respectively, compared with 35.3% and 35.3% in those treated with prednisone alone (P<.001). Fewer serious adverse events were reported in those treated with rituximab plus prednisone compared with those treated with prednisone alone. None of the patients who maintained complete remission off corticosteroid therapy received any additional maintenance infusions of rituximab after the end of the Ritux 3 regimen (1 g of rituximab at day 0 and day 14, then 500 mg at months 12 and 18).19
By contrast, treatment of severe bullous pemphigoid (BP) often is less clear-cut, as no single therapeutic option has been shown to be superior to other immunomodulatory and immunosuppressive regimens, and the medical comorbidities of elderly patients with BP can be limiting. Fortunately, newer therapies with favorable safety profiles have emerged in recent years. In a multicenter retrospective study, 100 patients with BP received omalizumab after previously failing to respond to at least one alternative therapy. Disease control was obtained after a median of 10 days, and complete remission was achieved in 77% of patients in a median time of 3 months.20 In a multicenter retrospective cohort study of 146 patients with BP treated with dupilumab following the atopic dermatitis dosing schedule (one 600-mg dose followed by 300 mg every 2 weeks), disease control was achieved in a median of 14 days, while complete remission was achieved in 35.6% of patients, with 8.9% relapsing during the observation period.21 A retrospective case series of 30 patients with BP treated with dupilumab with maintenance dosing frequency tailored to individual patient response showed complete remission or marked response in 76.7% (23/30) of patients.22 A phase 2/3 randomized controlled trial of dupilumab in BP is currently ongoing (ClinicalTrials.gov identifier NCT04206553).
Pemphigoid gestationis is a rare autoimmune subepidermal bullous dermatosis of pregnancy that may be difficult to distinguish clinically from polymorphic eruption of pregnancy but confers notably different maternal and fetal risks. Researchers developed and validated a scoring system using clinical factors—history of pemphigoid gestationis, primigravidae, timing of rash onset, and specific clinical examination findings—that was able to differentiate between the 2 diseases with 79% sensitivity, 95% specificity, and an area under the curve of 0.93 without the need for advanced immunologic testing.23
Final Thoughts
Highlights of the literature from 2023-2024 demonstrate advancements in hospital-based dermatology as well as ongoing challenges. This year’s review emphasizes key developments in severe cutaneous adverse drug reactions, skin and soft tissue infections, and AIBDs. Continued expansion of knowledge in these areas and others informs patient care and demonstrates the value of dermatologic expertise in the inpatient setting.
Inpatient consultative dermatology has advanced as a subspecialty and increasingly gained recognition in recent years. Since its founding in 2009, the Society of Dermatology Hospitalists has fostered research and education in hospital dermatology. Last year, we reviewed the 2022-2023 literature with a focus on developments in severe cutaneous adverse reactions, supportive oncodermatology, cost of inpatient services, and teledermatology.1 In this review, we highlight 3 areas of interest from the 2023-2024 literature: severe cutaneous adverse drug reactions, skin and soft tissue infections, and autoimmune blistering diseases (AIBDs).
Severe Cutaneous Adverse Drug Reactions
Adverse drug reactions are among the most common diagnoses encountered by inpatient dermatology consultants.2,3 Severe cutaneous adverse drug reactions are associated with substantial morbidity and mortality. Efforts to characterize these conditions and standardize their diagnosis and management continue to be a major focus of ongoing research.
A single-center retrospective analysis of 102 cases of drug reaction with eosinophilia and systemic symptoms (DRESS) syndrome evaluated differences in clinical manifestations depending on the culprit drug, offering insights into the heterogeneity of DRESS syndrome and the potential for diagnostic uncertainty.4 The shortest median latency was observed in a case caused by penicillin and cephalosporins (12 and 18 days, respectively), while DRESS syndrome secondary to allopurinol had the longest median latency (36 days). Nonsteroidal anti-inflammatory drug–induced DRESS syndrome was associated with the shortest hospital stay (6.5 days), while cephalosporin and vancomycin cases had the highest mortality rates.4
In the first international Delphi consensus study on the diagnostic workup, severity assessment, and management of DRESS syndrome, 54 dermatology and/or allergy experts reached consensus on 93 statements.5 Specific recommendations included basic evaluation with complete blood count with differential, kidney and liver function parameters, and electrocardiogram for all patients with suspected DRESS syndrome, with additional complementary workup considered in patients with evidence of specific organ damage and/or severe disease. In the proposed DRESS syndrome severity grading scheme, laboratory values that reached consensus for inclusion were hemoglobin, neutrophil, and platelet counts and creatinine, transaminases, and alkaline phosphatase levels. Although treatment of DRESS syndrome should be based on assessed disease severity, treatment with corticosteroids should be initiated in all patients with confirmed DRESS syndrome. Cyclosporine, antibodies interfering with the IL-5 axis, and intravenous immunoglobulins can be considered in patients with corticosteroid-refractory DRESS syndrome, and antiviral treatment can be considered in patients with a high serum cytomegalovirus viral load. Regularly following up with laboratory evaluation of involved organs; screening for autoantibodies, thyroid dysfunction, and steroid adverse effects; and offering of psychological support also were consensus recommendations.5
Identifying causative agents in drug hypersensitivity reactions remains challenging. A retrospective cohort study of 48 patients with Stevens-Johnson syndrome (SJS)/toxic epidermal necrolysis (TEN) highlighted the need for a systematic unbiased approach to identifying culprit drugs. Using the RegiSCAR database and algorithm for drug causality for epidermal necrolysis to analyze the cohort, more than half of causative agents were determined to be different from those initially identified by the treating physicians. Nine additional suspected culprit drugs were identified, while 43 drugs initially identified as allergens were exonerated.6
Etiology-associated definitions for blistering reactions in children have been proposed to replace the existing terms Stevens-Johnson syndrome, toxic epidermal necrolysis, and others.7 Investigators in a recent study reclassified cases of SJS and TEN as reactive infectious mucocutaneous eruption (RIME) or drug-induced epidermal necrolysis (DEN), respectively. In RIME cases, Mycoplasma pneumoniae was the most commonly identified trigger, and in DEN cases, anticonvulsants were the most common class of culprit medications. Cases of RIME were less severe and were most often treated with antibiotics, whereas patients with DEN were more likely to receive supportive care, corticosteroids, intravenous immunoglobulins, and other immunosuppressive therapies.7
In addition to causing acute devastating mucocutaneous complications, SJS and TEN have long-lasting effects that require ongoing care. In a cohort of 6552 incident SJS/TEN cases over an 11-year period, survivors of SJS/TEN endured a mean loss of 9.4 years in life expectancy and excess health care expenditures of $3752 per year compared with age- and sex-matched controls. Patients with more severe disease, comorbid malignancy, diabetes, end-stage renal disease, or SJS/TEN sequelae experienced greater loss in life expectancy and lifetime health care expenditures.8 Separately, a qualitative study investigating the psychological impact of SJS/TEN in pediatric patients described sequelae including night terrors, posttraumatic stress disorder, depression, and anxiety for many years after the acute phase. Many patients reported a desire for increased support for their physical and emotional needs following hospital discharge.9
Skin and Soft Tissue Infections: Diagnosis, Management, and Prevention
Dermatology consultation has been shown to be a cost-effective intervention to improve outcomes in hospitalized patients with skin and soft tissue infections.10,11 In particular, cellulitis frequently is misdiagnosed, leading to unnecessary antibiotic use, hospitalizations, and major health care expenditures.12 Recognizing this challenge, researchers have worked to develop objective tools to improve diagnostic accuracy. In a large prospective prognostic validation study, Pulia et al13 found that thermal imaging alone or in combination with the ALT-70 prediction model (asymmetry, leukocytosis, tachycardia, and age ≥70 years) could be used successfully to reduce overdiagnosis of cellulitis. Both thermal imaging and the ALT-70 prediction model demonstrated robust sensitivity (93.5% and 98.8%, respectively) but low specificity (38.4% and 22.0%, respectively, and 53.9% when combined).13
In a systematic review, Kovacs et al14 analyzed case reports of pseudocellulitis caused by chemotherapeutic medications. Of the 81 cases selected, 58 (71.6%) were associated with gemcitabine, with the remaining 23 (28.4%) attributed to pemetrexed. Within this group, two-thirds of the patients received antibiotic treatment prior to receiving the correct diagnosis, and 36% experienced interruptions to their oncologic therapies. In contrast to infectious cellulitis, which tends to be unilateral and associated with elevated erythrocyte sedimentation rate or C-reactive protein, most chemotherapy-induced pseudocellulitis cases occurred bilaterally on the lower extremities, while erythrocyte sedimentation rate and C-reactive protein seldom were elevated.14
Necrotizing soft tissue infections (NSTIs) are severe life-threatening conditions characterized by widespread tissue destruction, signs of systemic toxicity, hemodynamic collapse, organ failure, and high mortality. Surgical inspection along with intraoperative tissue culture is the gold standard for diagnosis. Early detection, prompt surgical intervention, and appropriate antibiotic treatment are essential to reduce mortality and improve outcomes.15 A retrospective study of patients with surgically confirmed NSTIs assessed the incidence and risk factors for recurrence within 1 year following an initial NSTI of the lower extremity. Among 93 included patients, 32 (34.4%) had recurrence within 1 year, and more than half of recurrences occurred in the first 3 months (median, 66 days). The comparison of patients with and without recurrence showed similar proportions of antibiotic prophylaxis use after the first NSTI. There was significantly less compression therapy use (33.3% vs 62.3%; P=.13) and more negative pressure wound therapy use (83.3% vs 63.3%; P=.03) in the recurrence group, though the authors acknowledged that factors such as severity of pain and size of soft tissue defect may have affected the decisions for compression and negative pressure wound therapy.16
Residents of nursing homes are a particularly vulnerable population at high risk for health care–associated infections due to older age and a higher likelihood of having wounds, indwelling medical devices, and/or coexisting conditions.17 One cluster-randomized trial compared universal decolonization with routine-care bathing practices in nursing homes (N=28,956 residents). Decolonization entailed the use of chlorhexidine for all routine bathing and showering and administration of nasal povidone-iodine twice daily for the first 5 days after admission and then twice daily for 5 days every other week. Transfer to a hospital due to infection decreased from 62.9% to 52.2% with decolonization, for a difference in risk ratio of 16.6% (P<.001) compared with routine care. Additionally, the difference in risk ratio of the secondary end point (transfer to a hospital for any reason) was 14.6%. The number needed to treat was 9.7 to prevent 1 infection-related hospitalization and 8.9 to prevent 1 hospitalization for any reason.17
Autoimmune Blistering Diseases
Although rare, AIBDs are potentially life-threatening cutaneous diseases that often require inpatient management. While corticosteroids remain the mainstay of initial AIBD management, rituximab is now well recognized as the steroid-sparing treatment of choice for patients with moderate to severe pemphigus. In a long-term follow-up study of Ritux 318—the trial that led to the US Food and Drug Administration approval of rituximab in the treatment of moderate to severe pemphigus vulgaris—researchers assessed the long-term efficacy and safety of rituximab as a first-line treatment in patients with pemphigus.19 The 5- and 7-year disease-free survival rates without corticosteroid therapy for patients treated with rituximab were 76.7% and 72.1%, respectively, compared with 35.3% and 35.3% in those treated with prednisone alone (P<.001). Fewer serious adverse events were reported in those treated with rituximab plus prednisone compared with those treated with prednisone alone. None of the patients who maintained complete remission off corticosteroid therapy received any additional maintenance infusions of rituximab after the end of the Ritux 3 regimen (1 g of rituximab at day 0 and day 14, then 500 mg at months 12 and 18).19
By contrast, treatment of severe bullous pemphigoid (BP) often is less clear-cut, as no single therapeutic option has been shown to be superior to other immunomodulatory and immunosuppressive regimens, and the medical comorbidities of elderly patients with BP can be limiting. Fortunately, newer therapies with favorable safety profiles have emerged in recent years. In a multicenter retrospective study, 100 patients with BP received omalizumab after previously failing to respond to at least one alternative therapy. Disease control was obtained after a median of 10 days, and complete remission was achieved in 77% of patients in a median time of 3 months.20 In a multicenter retrospective cohort study of 146 patients with BP treated with dupilumab following the atopic dermatitis dosing schedule (one 600-mg dose followed by 300 mg every 2 weeks), disease control was achieved in a median of 14 days, while complete remission was achieved in 35.6% of patients, with 8.9% relapsing during the observation period.21 A retrospective case series of 30 patients with BP treated with dupilumab with maintenance dosing frequency tailored to individual patient response showed complete remission or marked response in 76.7% (23/30) of patients.22 A phase 2/3 randomized controlled trial of dupilumab in BP is currently ongoing (ClinicalTrials.gov identifier NCT04206553).
Pemphigoid gestationis is a rare autoimmune subepidermal bullous dermatosis of pregnancy that may be difficult to distinguish clinically from polymorphic eruption of pregnancy but confers notably different maternal and fetal risks. Researchers developed and validated a scoring system using clinical factors—history of pemphigoid gestationis, primigravidae, timing of rash onset, and specific clinical examination findings—that was able to differentiate between the 2 diseases with 79% sensitivity, 95% specificity, and an area under the curve of 0.93 without the need for advanced immunologic testing.23
Final Thoughts
Highlights of the literature from 2023-2024 demonstrate advancements in hospital-based dermatology as well as ongoing challenges. This year’s review emphasizes key developments in severe cutaneous adverse drug reactions, skin and soft tissue infections, and AIBDs. Continued expansion of knowledge in these areas and others informs patient care and demonstrates the value of dermatologic expertise in the inpatient setting.
- Berk-Krauss J, Micheletti RG. Hospital dermatology: review of research in 2022-2023. Cutis. 2023;112:236-239.
- Falanga V, Schachner LA, Rae V, et al. Dermatologic consultations in the hospital setting. Arch Dermatol. 1994;130:1022-1025.
- Kroshinsky D, Cotliar J, Hughey LC, et al. Association of dermatology consultation with accuracy of cutaneous disorder diagnoses in hospitalized patients: a multicenter analysis. JAMA Dermatol. 2016;152:477-480.
- Blumenthal KG, Alvarez-Arango S, Kroshinsky D, et al. Drug reaction eosinophilia and systemic symptoms: clinical phenotypic patterns according to causative drug. J Am Acad Dermatol. 2024;90:1240-1242.
- Brüggen MC, Walsh S, Ameri MM, et al. Management of adult patients with drug reaction with eosinophilia and systemic symptoms: a Delphi-based international consensus. JAMA Dermatol. 2024;160:37-44.
- Li DJ, Velasquez GA, Romar GA, et al. Assessment of need for improved identification of a culprit drug in Stevens-Johnson syndrome/toxic epidermal necrolysis. JAMA Dermatol. 2023;159:830-836.
- Martinez-Cabriales S, Coulombe J, Aaron M, et al. Preliminary summary and reclassification of cases from the Pediatric Research of Management in Stevens-Johnson syndrome and Epidermonecrolysis (PROMISE) study: a North American, multisite retrospective cohort. J Am Acad Dermatol. 2024;90:635-637.
- Chiu YM, Chiu HY. Lifetime risk, life expectancy, loss-of-life expectancy and lifetime healthcare expenditure for Stevens-Johnson syndrome/toxic epidermal necrolysis in Taiwan: follow-up of a nationwide cohort from 2008 to 2019. Br J Dermatol. 2023;189:553-560.
- Phillips C, Russell E, McNiven A, et al. A qualitative study of psychological morbidity in paediatric survivors of Stevens-Johnson syndrome/toxic epidermal necrolysis. Br J Dermatol. 2024;191:293-295.
- Li DG, Xia FD, Khosravi H, et al. Outcomes of early dermatology consultation for inpatients diagnosed with cellulitis. JAMA Dermatol. 2018;154:537-543.
- Milani-Nejad N, Zhang M, Kaffenberger BH. Association of dermatology consultations with patient care outcomes in hospitalized patients with inflammatory skin diseases. JAMA Dermatol. 2017;153:523-528.
- Weng QY, Raff AB, Cohen JM, et al. Costs and consequences associated with misdiagnosed lower extremity cellulitis. JAMA Dermatol. 2017;153:141-146.
- Pulia MS, Schwei RJ, Alexandridis R, et al. Validation of thermal imaging and the ALT-70 prediction model to differentiate cellulitis from pseudocellulitis. JAMA Dermatol. 2024;160:511-517.
- Kovacs LD, O’Donoghue M, Cogen AL. Chemotherapy-induced pseudocellulitis without prior radiation exposure: a systematic review. JAMA Dermatol. 2023;159:870-874.
- Yildiz H, Yombi JC. Necrotizing soft-tissue infections. comment. N Engl J Med. 2018;378:970.
- Traineau H, Charpentier C, Lepeule R, et al. First-year recurrence rate of skin and soft tissue infections following an initial necrotizing soft tissue infection of the lower extremities: a retrospective cohort study of 93 patients. J Am Acad Dermatol. 2023;88:1360-1363.
- Miller LG, McKinnell JA, Singh RD, et al. Decolonization in nursing homes to prevent infection and hospitalization. N Engl J Med. 2023;389:1766-1777.
- Joly P, Maho-Vaillant M, Prost-Squarcioni C, et al; French Study Group on Autoimmune Bullous Skin Diseases. First-line rituximab combined with short-term prednisone versus prednisone alone for the treatment of pemphigus (Ritux 3): a prospective, multicentre, parallel-group, open-label randomised trial. Lancet. 2017;389:2031-2040.
- Tedbirt B, Maho-Vaillant M, Houivet E, et al; French Reference Center for Autoimmune Blistering Diseases MALIBUL. Sustained remission without corticosteroids among patients with pemphigus who had rituximab as first-line therapy: follow-up of the Ritux 3 Trial. JAMA Dermatol. 2024;160:290-296.
- Chebani R, Lombart F, Chaby G, et al; French Study Group on Autoimmune Bullous Diseases. Omalizumab in the treatment of bullous pemphigoid resistant to first-line therapy: a French national multicentre retrospective study of 100 patients. Br J Dermatol. 2024;190:258-265.
- Zhao L, Wang Q, Liang G, et al. Evaluation of dupilumab in patients with bullous pemphigoid. JAMA Dermatol. 2023;159:953-960.
- Miller AC, Temiz LA, Adjei S, et al. Treatment of bullous pemphigoid with dupilumab: a case series of 30 patients. J Drugs Dermatol. 2024;23:E144-E148.
- Xie F, Davis DMR, Baban F, et al. Development and multicenter international validation of a diagnostic tool to differentiate between pemphigoid gestationis and polymorphic eruption of pregnancy. J Am Acad Dermatol. 2023;89:106-113.
- Berk-Krauss J, Micheletti RG. Hospital dermatology: review of research in 2022-2023. Cutis. 2023;112:236-239.
- Falanga V, Schachner LA, Rae V, et al. Dermatologic consultations in the hospital setting. Arch Dermatol. 1994;130:1022-1025.
- Kroshinsky D, Cotliar J, Hughey LC, et al. Association of dermatology consultation with accuracy of cutaneous disorder diagnoses in hospitalized patients: a multicenter analysis. JAMA Dermatol. 2016;152:477-480.
- Blumenthal KG, Alvarez-Arango S, Kroshinsky D, et al. Drug reaction eosinophilia and systemic symptoms: clinical phenotypic patterns according to causative drug. J Am Acad Dermatol. 2024;90:1240-1242.
- Brüggen MC, Walsh S, Ameri MM, et al. Management of adult patients with drug reaction with eosinophilia and systemic symptoms: a Delphi-based international consensus. JAMA Dermatol. 2024;160:37-44.
- Li DJ, Velasquez GA, Romar GA, et al. Assessment of need for improved identification of a culprit drug in Stevens-Johnson syndrome/toxic epidermal necrolysis. JAMA Dermatol. 2023;159:830-836.
- Martinez-Cabriales S, Coulombe J, Aaron M, et al. Preliminary summary and reclassification of cases from the Pediatric Research of Management in Stevens-Johnson syndrome and Epidermonecrolysis (PROMISE) study: a North American, multisite retrospective cohort. J Am Acad Dermatol. 2024;90:635-637.
- Chiu YM, Chiu HY. Lifetime risk, life expectancy, loss-of-life expectancy and lifetime healthcare expenditure for Stevens-Johnson syndrome/toxic epidermal necrolysis in Taiwan: follow-up of a nationwide cohort from 2008 to 2019. Br J Dermatol. 2023;189:553-560.
- Phillips C, Russell E, McNiven A, et al. A qualitative study of psychological morbidity in paediatric survivors of Stevens-Johnson syndrome/toxic epidermal necrolysis. Br J Dermatol. 2024;191:293-295.
- Li DG, Xia FD, Khosravi H, et al. Outcomes of early dermatology consultation for inpatients diagnosed with cellulitis. JAMA Dermatol. 2018;154:537-543.
- Milani-Nejad N, Zhang M, Kaffenberger BH. Association of dermatology consultations with patient care outcomes in hospitalized patients with inflammatory skin diseases. JAMA Dermatol. 2017;153:523-528.
- Weng QY, Raff AB, Cohen JM, et al. Costs and consequences associated with misdiagnosed lower extremity cellulitis. JAMA Dermatol. 2017;153:141-146.
- Pulia MS, Schwei RJ, Alexandridis R, et al. Validation of thermal imaging and the ALT-70 prediction model to differentiate cellulitis from pseudocellulitis. JAMA Dermatol. 2024;160:511-517.
- Kovacs LD, O’Donoghue M, Cogen AL. Chemotherapy-induced pseudocellulitis without prior radiation exposure: a systematic review. JAMA Dermatol. 2023;159:870-874.
- Yildiz H, Yombi JC. Necrotizing soft-tissue infections. comment. N Engl J Med. 2018;378:970.
- Traineau H, Charpentier C, Lepeule R, et al. First-year recurrence rate of skin and soft tissue infections following an initial necrotizing soft tissue infection of the lower extremities: a retrospective cohort study of 93 patients. J Am Acad Dermatol. 2023;88:1360-1363.
- Miller LG, McKinnell JA, Singh RD, et al. Decolonization in nursing homes to prevent infection and hospitalization. N Engl J Med. 2023;389:1766-1777.
- Joly P, Maho-Vaillant M, Prost-Squarcioni C, et al; French Study Group on Autoimmune Bullous Skin Diseases. First-line rituximab combined with short-term prednisone versus prednisone alone for the treatment of pemphigus (Ritux 3): a prospective, multicentre, parallel-group, open-label randomised trial. Lancet. 2017;389:2031-2040.
- Tedbirt B, Maho-Vaillant M, Houivet E, et al; French Reference Center for Autoimmune Blistering Diseases MALIBUL. Sustained remission without corticosteroids among patients with pemphigus who had rituximab as first-line therapy: follow-up of the Ritux 3 Trial. JAMA Dermatol. 2024;160:290-296.
- Chebani R, Lombart F, Chaby G, et al; French Study Group on Autoimmune Bullous Diseases. Omalizumab in the treatment of bullous pemphigoid resistant to first-line therapy: a French national multicentre retrospective study of 100 patients. Br J Dermatol. 2024;190:258-265.
- Zhao L, Wang Q, Liang G, et al. Evaluation of dupilumab in patients with bullous pemphigoid. JAMA Dermatol. 2023;159:953-960.
- Miller AC, Temiz LA, Adjei S, et al. Treatment of bullous pemphigoid with dupilumab: a case series of 30 patients. J Drugs Dermatol. 2024;23:E144-E148.
- Xie F, Davis DMR, Baban F, et al. Development and multicenter international validation of a diagnostic tool to differentiate between pemphigoid gestationis and polymorphic eruption of pregnancy. J Am Acad Dermatol. 2023;89:106-113.
Practice Points
- An international Delphi study reached consensus on 93 statements regarding workup, severity assessment, and management of DRESS syndrome.
- In nursing homes, universal decolonization with chlorhexidine and nasal iodophor greatly reduced the risk for hospital transfers due to infection compared to routine care.
- Rituximab as the first-line therapy for pemphigus vulgaris is associated with long-term sustained complete remission without corticosteroid therapy.
- Dupilumab and omalizumab are emerging safe and effective treatment options for bullous pemphigoid.
Emerging Insights in Keloid Pathogenesis and Therapeutics
Keloids are fibroproliferative lesions caused by aberrant wound healing in predisposed individuals.1 While keloids have been reported in patients of all races and ethnicities, they most commonly develop in individuals of African or Asian descent.2 Often associated with symptoms such as pain and itching, keloids can be disfiguring and result in poorer quality of life.3 There is a paucity of research on keloid pathogenesis and efficacious therapeutics, particularly in patients with skin of color (SOC). Herein, we outline the current research on keloid treatment and highlight promising new therapies ranging from innovative intralesional techniques to advanced laser-based and biologic therapies.
Deficiencies in Skin of Color Research
Although keloids are 17 times more prevalent in patients with SOC,4 there is a considerable lack of focus on this population in the literature.5 Studies on keloids that include individuals with SOC often group patients of all skin types together, and subgroup analyses are not always performed.6,7 As a result, dermatologists may face considerable challenges in providing effective treatments for keloids in patients with SOC. With few evidence-based options available, patients with SOC who have keloids continue to experience impairments in quality of life.
Common Keloid Therapies
There currently is no gold-standard treatment for keloids. Common therapeutic modalities include intralesional corticosteroids (ILCs), antineoplastic agents and neuromodulators, laser-based devices, and surgical therapies (eg, excision), as well as combined medical and surgical techniques.8
Intralesional Corticosteroids—Minimally invasive ILCs are the first-line treatment in all patients with keloids, regardless of skin phototype. Because keloid formation results from trauma to the skin, ILCs often are recommended to minimize further skin damage.5 One meta-analysis found that ILCs have demonstrated success rates of 50% to 100%9; however, these studies frequently combine ILCs with other treatment modalities, and few studies have focused on the efficacy of ILC monotherapy in patients with SOC.6,10-13
Antineoplastic Agents and Neuromodulators—Certain antineoplastic agents (eg, 5-fluorouracil [5-FU] and bleomycin) and neuromodulators (eg, botulinum toxin A [BTA]) also have been studied in keloid management.8
5-Fluorouracil frequently is combined with ILCs such as triamcinolone (TAC). Combined therapy is more effective than TAC monotherapy in scar height reduction.14,15 Rates of adverse events such as dyspigmentation, atrophy, and telangiectasias also were lower in patients who received combined therapy.14,15 A systematic review found that intralesional bleomycin may be more effective than TAC alone, 5-FU alone, TAC combined with 5-FU, and TAC combined with cryotherapy; however, hyperpigmentation was a common adverse event, occurring in roughly 70% (42/60) of patients.16,17 Additionally, a 2024 meta-analysis evaluated 20 randomized controlled trials comprising 1114 patients treated with intralesional TAC, 5-FU, BTA, verapamil, and/or bleomycin. Botulinum toxin A and TAC plus 5-FU were found to have outstanding therapeutic efficacy for keloids, and rates of adverse events were similar among users of TAC, 5-FU, BTA, and TAC plus 5-FU.18
While antineoplastic agents and BTA may be promising keloid therapies, further studies demonstrating their efficacy and safety profiles are necessary, particularly regarding dyspigmentation as a potential adverse event, as this may be of concern in patients with darker phototypes.
Laser Therapies—Of all treatment modalities, laser-based keloid therapies have been the most robustly studied in SOC. The 2 main types are ablative (eg, CO2, Er:YAG) and nonablative (eg, pulsed dye, Nd:YAG) lasers. Ablative lasers rapidly heat water molecules within the skin, thereby vaporizing the skin cells in a controlled precise process that reduces scar tissue by removing layers of skin. Nonablative lasers target hemoglobin in blood vessels, reducing oxygen supply and inducing collagen remodeling without damaging the epidermis.19
For patients with SOC, lasers carry a risk for postinflammatory hyperpigmentation.20 To address this risk, recent advancements in laser technology and procedural protocols have aimed to minimize the number of passes and utilize cooling devices21; however, many of these recommendations are based on retrospective reviews and small case series. A 2024 meta-analysis comprising 550 patients found that the combination of fractional CO2 laser therapy and 5-FU was the most effective intervention, markedly reducing Vancouver Scar Scale and pliability scores as well as keloid thickness.22 Conversely, pulsed dye lasers were the least effective in terms of improving scar thickness, pigmentation, and pliability when compared to other treatments.
Randomized controlled trials of laser-based therapies in patients with SOC are lacking in the literature. Future studies should focus on calibrating laser-based therapies for those with darker skin tones and examine the efficacy and adverse effects of ablative and nonablative lasers in patients with SOC.
Promising New Keloid Therapies
Keloid disease pathogenesis is incompletely understood, but several new therapeutic targets have been highlighted in the literature, including dupilumab, pentoxifylline, sirtuin 6 (SIRT6) modulators, remdesivir, and needle-assisted electrocoagulation plus pharmacotherapy.
Dupilumab—An anti–IL-4 and IL-13 monoclonal antibody, dupilumab was first approved for the treatment of severe atopic dermatitis. Its use has broadened since its approval, and keloids have been identified as a potential therapeutic target. A 2019 case study described a 53-year-old Black man with severe atopic dermatitis and chronic keloids that regressed with systemic dupilumab therapy.23 This prompted a follow-up case-control study using real-time polymerase chain reaction testing to evaluate Th2 gene expression (IL-4R, IL-13, and CCL18) of lesional and nonlesional tissue in 3 Black patients with chronic keloids and no concurrent atopic dermatitis vs 5 healthy Black controls. Despite the limited sample size, a significant increase in IL-13 and the Th2 chemokine CCL18 was found in patients with keloids compared to controls (P<.05), suggesting that the entire integument of patients with severe keloids is abnormal.23 This finding supports the use of systemic treatments for chronic and multifocal keloid disease. Several subsequent case reports have corroborated the efficacy of systemic and/or intralesional dupilumab.24,25 However, some studies have reported contradictory findings, suggesting the need for high-quality clinical trials.26,27
Pentoxifylline—Pentoxifylline is a methylated xanthine derivative and a nonspecific phosphodiesterase inhibitor used to treat claudication from peripheral artery disease. It also inhibits the proliferation and rate of collagen synthesis of fibroblasts from keloids in vitro.28,29 A 2019 retrospective, open-label pilot study analyzed postsurgical keloid recurrence in 45 patients with 67 unique keloids that were stratified into low- and high-risk groups based on clinical factors including multiple symptomatic keloids, history of recurrence, and family history.30 Both the low- and the high-risk groups were treated with 40 mg/mL intralesional triamcinolone acetonide monthly for 6 months; however, some of the high-risk keloids also received pentoxifylline 300 mg 3 times daily for 6 months. There was a statistically significant decrease in keloid recurrence rate between the high-risk group treated with pentoxifylline and the low-risk group for whom pentoxifylline was not prescribed (P=.015).
Similarly, a randomized clinical trial comparing the efficacy of combination intralesional pentoxifylline and intralesional triamcinolone vs monotherapy with pentoxifylline or triamcinolone found the most significant improvement in the combination cohort with reduction in keloid height (P=.04), pliability (P=.003), and vascularity (P=.05).31 These findings highlight the need for supplementary studies on the use of pentoxifylline for keloid therapy.
SIRT6 Modulators—SIRT6 modulators are an exciting future therapeutic target. In a recent case-control study evaluating the histologic milieu of keloid tissue vs normal skin specimens, the researchers found that selective overexpression of SIRT6 via the use of a recombinant adenovirus in keloid fibroblasts attenuated proliferation, invasion, and collagen synthesis while fostering apoptosis, likely through the suppression of MAPK/ERK pathway activity.32
Remdesivir—The antiviral drug remdesivir has been reported to have pharmacologic activities in a wide range of fibrotic diseases, including keloids. A 2024 study explored the potential effect and mechanisms of remdesivir on skin fibrosis both in vitro and in rodents.33 Remdesivir was found to decrease skin fibrosis and attenuate the gross weight of keloid tissues in vivo, suppress fibroblast activation and autophagy both in vivo and in vitro, dampen fibroblast activation by the TGF-β1/Smad signaling pathway, and inhibit fibroblasts autophagy by the PI3K/Akt/mTOR signaling pathway. These results demonstrate the therapeutic potential of remdesivir for keloid management.
Needle-Assisted Electrocoagulation Plus Pharmacotherapy—A novel needle-assisted electrocoagulation technique combined with pharmacotherapy (corticosteroid and 5-FU injections) was effective in a Chinese clinical trial involving 6 patients with keloids.34 Investigators used Vancouver Scar Scale and both Patient and Observer Scar Assessment Scale scores to grade patients’ scars before treatment and 1 month after the first treatment cycle. They found that ablation combined with pharmacotherapy significantly reduced all 3 scores without any obvious adverse events (P=.004, P=.006, and P=.017, respectively). This novel combination treatment may serve as a safe and effective therapeutic approach for keloid removal.
Final Thoughts
Emerging treatments offer promising new horizons in keloid management; however, the lack of robust, high-quality clinical trials, especially those focusing on
- Téot L, Mustoe TA, Middelkoop E, eds. Textbook on Scar Management: State of the Art Management and Emerging Technologies. Springer; 2020.
- Davis SA, Feldman SR, McMichael AJ. Management of keloids in the United States, 1990-2009: an analysis of the National Ambulatory Medical Care Survey. Dermatol Surg. 2013;39:988-994. doi:10.1111/dsu.12182
- Kassi K, Kouame K, Kouassi A, et al. Quality of life in black African patients with keloid scars. Dermatol Reports. 2020;12:8312. doi:10.4081/dr.2020.8312
- Delaleu J, Charvet E, Petit A. Keloid disease: review with clinical atlas. part I: definitions, history, epidemiology, clinics and diagnosis. Ann Dermatol Venereol. 2023;150:3-15.
doi:10.1016/j.annder.2022.08.010 - Bronte J, Zhou C, Vempati A, et al. A comprehensive review of non-surgical treatments for hypertrophic and keloid scars in skin of color. Clin Cosmet Investig Dermatol.
2024;17:1459-1469. doi:10.2147/CCID.S470997 - Davison SP, Dayan JH, Clemens MW, et al. Efficacy of intralesional 5-fluorouracil and triamcinolone in the treatment of keloids. Aesthet Surg J. 2009;29:40-46. doi:10.1016/j.asj.2008.11.006
- Azzam OA, Bassiouny DA, El-Hawary MS, et al. Treatment of hypertrophic scars and keloids by fractional carbon dioxide laser: a clinical, histological, and immunohistochemical study. Lasers Med Sci. 2016;31:9-18. doi:10.1007/s10103-015-1824-4
- Ekstein SF, Wyles SP, Moran SL, et al. Keloids: a review of therapeutic management. Int J Dermatol. 2021;60:661-671. doi:10.1111/ijd.15159
- Morelli Coppola M, Salzillo R, Segreto F, et al. Triamcinolone acetonide intralesional injection for the treatment of keloid scars: patient selection and perspectives. Clin Cosmet Investig Dermatol. 2018;11:387-396. doi:10.2147/CCID.S133672
- Kant SB, van den Kerckhove E, Colla C, et al. A new treatment of hypertrophic and keloid scars with combined triamcinolone and verapamil: a retrospective study. Eur J Plast Surg. 2018;41:69-80. doi:10.1007/s00238-017-1322-y
- Cohen AJ, Talasila S, Lazarevic B, et al. Combination cryotherapy and intralesional corticosteroid versus steroid monotherapy in the treatment of keloids. J Cosmet Dermatol. 2023;22:932-936. doi:10.1111/jocd.15520
- Tawaranurak N, Pliensiri P, Tawaranurak K. Combination of fractional carbon dioxide laser and topical triamcinolone vs intralesional triamcinolone for keloid treatment: a randomised clinical trial. Int Wound J. 2022;19:1729-1735. doi:10.1111/iwj.13775
- Belie O, Ugburo AO, Mofikoya BO, et al. A comparison of intralesional verapamil and triamcinolone monotherapy in the treatment of keloids in an African population. Niger J Clin Pract. 2021;24:986-992. doi:10.4103/njcp.njcp_474_20
- Khalid FA, Mehrose MY, Saleem M, et al. Comparison of efficacy and safety of intralesional triamcinolone and combination of triamcinolone with 5-fluorouracil in the treatment of keloids and hypertrophic scars: randomised control trial. Burns. 2019;45:69-75. doi:10.1016/j.burns.2018.08.011
- Asilian A, Darougheh A, Shariati F. New combination of triamcinolone, 5-Fluorouracil, and pulsed-dye laser for treatment of keloid and hypertrophic scars. Dermatol Surg. 2006;32:907-915. doi:10.1111/j.1524-4725.2006.32195.x
- Kim WI, Kim S, Cho SW, et al. The efficacy of bleomycin for treating keloid and hypertrophic scar: a systematic review and meta-analysis. J Cosmet Dermatol. 2020;19:3357-3366. doi:10.1111/jocd.13390
- Kabel A, Sabry H, Sorour N, et al. Comparative study between intralesional injection of bleomycin and 5-fluorouracil in the treatment of keloids and hypertrophic scars. J Dermatol Dermatol Surg. 2016;20:32-38.
- Yang HA, Jheng WL, Yu J, et al. Comparative efficacy of drug interventions for keloids: a network meta-analysis. Ann Plast Surg. 2024;92(1S suppl 1
):S52-S59. doi:10.1097/SAP.0000000000003759 - Preissig J, Hamilton K, Markus R. Current laser resurfacing technologies: a review that delves beneath the surface. Semin Plast Surg. 2012;26:109-116. doi:10.1055/s-0032-1329413
- Bin Dakhil A, Shadid A, Altalhab S. Post-inflammatory hyperpigmentation after carbon dioxide laser: review of prevention and risk factors. Dermatol Reports. 2023;15:9703. doi:10.4081/dr.2023.9703
- Kaushik SB, Alexis AF. Nonablative fractional laser resurfacing in skin of color: evidence-based review. J Clin Aesthet Dermatol. 2017;10:51-67.
- Foppiani JA, Khaity A, Al-Dardery NM, et al. Laser therapy in hypertrophic and keloid scars: a systematic review and network meta-analysis. Aesthetic Plast Surg. Published May 17, 2024. doi:10.1007/s00266-024-04027-9
- Diaz A, Tan K, He H, et al. Keloid lesions show increased IL-4/IL-13 signaling and respond to Th2-targeting dupilumab therapy. J Eur Acad Dermatol Venereol. 2020;34:E161-E164. doi:10.1111/jdv.16097
- Min MS,
Mazori DR, Lee MS, et al. Successful treatment of keloids and hypertrophic scars with systemic and intralesional dupilumab. J Drugs Dermatol. 2023;22:1220-1222. doi:10.36849/JDD.6385 - Wittmer A, Finklea L, Joseph J. Effects of dupilumab on keloid stabilization and prevention. JAAD Case Rep. 2023;37:103-105. doi:10.1016/j.jdcr.2023.05.001
- Luk K, Fakhoury J, Ozog D. Nonresponse and progression of diffuse keloids to dupilumab therapy. J Drugs Dermatol. 2022;21:197-199. doi:10.36849/jdd.6252
- Tirgan MH, Uitto J. Lack of efficacy of dupilumab in the treatment of keloid disorder. J Eur Acad Dermatol Venereol. 2022;36:E120-E122. doi:10.1111/jdv.17669
- Berman B, Duncan MR. Pentoxifylline inhibits the proliferation of human fibroblasts derived from keloid, scleroderma and morphoea skin and their production of collagen, glycosaminoglycans and fibronectin. Br J Dermatol. 1990;123:339-346. doi:10.1111/j.1365-
2133.1990.tb06294.x - Berman B, Duncan MR. Pentoxifylline inhibits normal human dermal fibroblast in vitro proliferation, collagen, glycosaminoglycan, and fibronectin production, and increases collagenase activity. J Invest Dermatol. 1989;92:605-610.
- Tan A, Martinez Luna O, Glass DA 2nd. Pentoxifylline for the prevention of postsurgical keloid recurrence. Dermatol Surg. 2020;46:1353-1356. doi:10.1097/DSS.0000000000002090
- Serag-Eldin YMA, Mahmoud WH, Gamea MM, et al. Intralesional pentoxifylline, triamcinolone acetonide, and their combination for treatment of keloid scars. J Cosmet Dermatol. 2021;20:3330-3340. doi:10.1111/jocd.14305
- Zhou T, Chen Y, Wang C, et al. SIRT6 inhibits the proliferation and collagen synthesis of keloid fibroblasts through MAPK/ERK pathway. Discov Med. 2024;36:1430-1440. doi:10.24976/Discov.Med.202436186.133
- Zhang J, Zhang X, Guo X, et al. Remdesivir alleviates skin fibrosis by suppressing TGF-β1 signaling pathway. PLoS One. 2024;19:E0305927.
doi:10.1371/journal.pone.0305927 - Zhao J, Zhai X, Xu Z, et al. Novel needle-type electrocoagulation and combination pharmacotherapy: basic and clinical studies on efficacy and safety in treating keloids. J Cosmet Dermatol. doi:10.1111/jocd.16453
Keloids are fibroproliferative lesions caused by aberrant wound healing in predisposed individuals.1 While keloids have been reported in patients of all races and ethnicities, they most commonly develop in individuals of African or Asian descent.2 Often associated with symptoms such as pain and itching, keloids can be disfiguring and result in poorer quality of life.3 There is a paucity of research on keloid pathogenesis and efficacious therapeutics, particularly in patients with skin of color (SOC). Herein, we outline the current research on keloid treatment and highlight promising new therapies ranging from innovative intralesional techniques to advanced laser-based and biologic therapies.
Deficiencies in Skin of Color Research
Although keloids are 17 times more prevalent in patients with SOC,4 there is a considerable lack of focus on this population in the literature.5 Studies on keloids that include individuals with SOC often group patients of all skin types together, and subgroup analyses are not always performed.6,7 As a result, dermatologists may face considerable challenges in providing effective treatments for keloids in patients with SOC. With few evidence-based options available, patients with SOC who have keloids continue to experience impairments in quality of life.
Common Keloid Therapies
There currently is no gold-standard treatment for keloids. Common therapeutic modalities include intralesional corticosteroids (ILCs), antineoplastic agents and neuromodulators, laser-based devices, and surgical therapies (eg, excision), as well as combined medical and surgical techniques.8
Intralesional Corticosteroids—Minimally invasive ILCs are the first-line treatment in all patients with keloids, regardless of skin phototype. Because keloid formation results from trauma to the skin, ILCs often are recommended to minimize further skin damage.5 One meta-analysis found that ILCs have demonstrated success rates of 50% to 100%9; however, these studies frequently combine ILCs with other treatment modalities, and few studies have focused on the efficacy of ILC monotherapy in patients with SOC.6,10-13
Antineoplastic Agents and Neuromodulators—Certain antineoplastic agents (eg, 5-fluorouracil [5-FU] and bleomycin) and neuromodulators (eg, botulinum toxin A [BTA]) also have been studied in keloid management.8
5-Fluorouracil frequently is combined with ILCs such as triamcinolone (TAC). Combined therapy is more effective than TAC monotherapy in scar height reduction.14,15 Rates of adverse events such as dyspigmentation, atrophy, and telangiectasias also were lower in patients who received combined therapy.14,15 A systematic review found that intralesional bleomycin may be more effective than TAC alone, 5-FU alone, TAC combined with 5-FU, and TAC combined with cryotherapy; however, hyperpigmentation was a common adverse event, occurring in roughly 70% (42/60) of patients.16,17 Additionally, a 2024 meta-analysis evaluated 20 randomized controlled trials comprising 1114 patients treated with intralesional TAC, 5-FU, BTA, verapamil, and/or bleomycin. Botulinum toxin A and TAC plus 5-FU were found to have outstanding therapeutic efficacy for keloids, and rates of adverse events were similar among users of TAC, 5-FU, BTA, and TAC plus 5-FU.18
While antineoplastic agents and BTA may be promising keloid therapies, further studies demonstrating their efficacy and safety profiles are necessary, particularly regarding dyspigmentation as a potential adverse event, as this may be of concern in patients with darker phototypes.
Laser Therapies—Of all treatment modalities, laser-based keloid therapies have been the most robustly studied in SOC. The 2 main types are ablative (eg, CO2, Er:YAG) and nonablative (eg, pulsed dye, Nd:YAG) lasers. Ablative lasers rapidly heat water molecules within the skin, thereby vaporizing the skin cells in a controlled precise process that reduces scar tissue by removing layers of skin. Nonablative lasers target hemoglobin in blood vessels, reducing oxygen supply and inducing collagen remodeling without damaging the epidermis.19
For patients with SOC, lasers carry a risk for postinflammatory hyperpigmentation.20 To address this risk, recent advancements in laser technology and procedural protocols have aimed to minimize the number of passes and utilize cooling devices21; however, many of these recommendations are based on retrospective reviews and small case series. A 2024 meta-analysis comprising 550 patients found that the combination of fractional CO2 laser therapy and 5-FU was the most effective intervention, markedly reducing Vancouver Scar Scale and pliability scores as well as keloid thickness.22 Conversely, pulsed dye lasers were the least effective in terms of improving scar thickness, pigmentation, and pliability when compared to other treatments.
Randomized controlled trials of laser-based therapies in patients with SOC are lacking in the literature. Future studies should focus on calibrating laser-based therapies for those with darker skin tones and examine the efficacy and adverse effects of ablative and nonablative lasers in patients with SOC.
Promising New Keloid Therapies
Keloid disease pathogenesis is incompletely understood, but several new therapeutic targets have been highlighted in the literature, including dupilumab, pentoxifylline, sirtuin 6 (SIRT6) modulators, remdesivir, and needle-assisted electrocoagulation plus pharmacotherapy.
Dupilumab—An anti–IL-4 and IL-13 monoclonal antibody, dupilumab was first approved for the treatment of severe atopic dermatitis. Its use has broadened since its approval, and keloids have been identified as a potential therapeutic target. A 2019 case study described a 53-year-old Black man with severe atopic dermatitis and chronic keloids that regressed with systemic dupilumab therapy.23 This prompted a follow-up case-control study using real-time polymerase chain reaction testing to evaluate Th2 gene expression (IL-4R, IL-13, and CCL18) of lesional and nonlesional tissue in 3 Black patients with chronic keloids and no concurrent atopic dermatitis vs 5 healthy Black controls. Despite the limited sample size, a significant increase in IL-13 and the Th2 chemokine CCL18 was found in patients with keloids compared to controls (P<.05), suggesting that the entire integument of patients with severe keloids is abnormal.23 This finding supports the use of systemic treatments for chronic and multifocal keloid disease. Several subsequent case reports have corroborated the efficacy of systemic and/or intralesional dupilumab.24,25 However, some studies have reported contradictory findings, suggesting the need for high-quality clinical trials.26,27
Pentoxifylline—Pentoxifylline is a methylated xanthine derivative and a nonspecific phosphodiesterase inhibitor used to treat claudication from peripheral artery disease. It also inhibits the proliferation and rate of collagen synthesis of fibroblasts from keloids in vitro.28,29 A 2019 retrospective, open-label pilot study analyzed postsurgical keloid recurrence in 45 patients with 67 unique keloids that were stratified into low- and high-risk groups based on clinical factors including multiple symptomatic keloids, history of recurrence, and family history.30 Both the low- and the high-risk groups were treated with 40 mg/mL intralesional triamcinolone acetonide monthly for 6 months; however, some of the high-risk keloids also received pentoxifylline 300 mg 3 times daily for 6 months. There was a statistically significant decrease in keloid recurrence rate between the high-risk group treated with pentoxifylline and the low-risk group for whom pentoxifylline was not prescribed (P=.015).
Similarly, a randomized clinical trial comparing the efficacy of combination intralesional pentoxifylline and intralesional triamcinolone vs monotherapy with pentoxifylline or triamcinolone found the most significant improvement in the combination cohort with reduction in keloid height (P=.04), pliability (P=.003), and vascularity (P=.05).31 These findings highlight the need for supplementary studies on the use of pentoxifylline for keloid therapy.
SIRT6 Modulators—SIRT6 modulators are an exciting future therapeutic target. In a recent case-control study evaluating the histologic milieu of keloid tissue vs normal skin specimens, the researchers found that selective overexpression of SIRT6 via the use of a recombinant adenovirus in keloid fibroblasts attenuated proliferation, invasion, and collagen synthesis while fostering apoptosis, likely through the suppression of MAPK/ERK pathway activity.32
Remdesivir—The antiviral drug remdesivir has been reported to have pharmacologic activities in a wide range of fibrotic diseases, including keloids. A 2024 study explored the potential effect and mechanisms of remdesivir on skin fibrosis both in vitro and in rodents.33 Remdesivir was found to decrease skin fibrosis and attenuate the gross weight of keloid tissues in vivo, suppress fibroblast activation and autophagy both in vivo and in vitro, dampen fibroblast activation by the TGF-β1/Smad signaling pathway, and inhibit fibroblasts autophagy by the PI3K/Akt/mTOR signaling pathway. These results demonstrate the therapeutic potential of remdesivir for keloid management.
Needle-Assisted Electrocoagulation Plus Pharmacotherapy—A novel needle-assisted electrocoagulation technique combined with pharmacotherapy (corticosteroid and 5-FU injections) was effective in a Chinese clinical trial involving 6 patients with keloids.34 Investigators used Vancouver Scar Scale and both Patient and Observer Scar Assessment Scale scores to grade patients’ scars before treatment and 1 month after the first treatment cycle. They found that ablation combined with pharmacotherapy significantly reduced all 3 scores without any obvious adverse events (P=.004, P=.006, and P=.017, respectively). This novel combination treatment may serve as a safe and effective therapeutic approach for keloid removal.
Final Thoughts
Emerging treatments offer promising new horizons in keloid management; however, the lack of robust, high-quality clinical trials, especially those focusing on
Keloids are fibroproliferative lesions caused by aberrant wound healing in predisposed individuals.1 While keloids have been reported in patients of all races and ethnicities, they most commonly develop in individuals of African or Asian descent.2 Often associated with symptoms such as pain and itching, keloids can be disfiguring and result in poorer quality of life.3 There is a paucity of research on keloid pathogenesis and efficacious therapeutics, particularly in patients with skin of color (SOC). Herein, we outline the current research on keloid treatment and highlight promising new therapies ranging from innovative intralesional techniques to advanced laser-based and biologic therapies.
Deficiencies in Skin of Color Research
Although keloids are 17 times more prevalent in patients with SOC,4 there is a considerable lack of focus on this population in the literature.5 Studies on keloids that include individuals with SOC often group patients of all skin types together, and subgroup analyses are not always performed.6,7 As a result, dermatologists may face considerable challenges in providing effective treatments for keloids in patients with SOC. With few evidence-based options available, patients with SOC who have keloids continue to experience impairments in quality of life.
Common Keloid Therapies
There currently is no gold-standard treatment for keloids. Common therapeutic modalities include intralesional corticosteroids (ILCs), antineoplastic agents and neuromodulators, laser-based devices, and surgical therapies (eg, excision), as well as combined medical and surgical techniques.8
Intralesional Corticosteroids—Minimally invasive ILCs are the first-line treatment in all patients with keloids, regardless of skin phototype. Because keloid formation results from trauma to the skin, ILCs often are recommended to minimize further skin damage.5 One meta-analysis found that ILCs have demonstrated success rates of 50% to 100%9; however, these studies frequently combine ILCs with other treatment modalities, and few studies have focused on the efficacy of ILC monotherapy in patients with SOC.6,10-13
Antineoplastic Agents and Neuromodulators—Certain antineoplastic agents (eg, 5-fluorouracil [5-FU] and bleomycin) and neuromodulators (eg, botulinum toxin A [BTA]) also have been studied in keloid management.8
5-Fluorouracil frequently is combined with ILCs such as triamcinolone (TAC). Combined therapy is more effective than TAC monotherapy in scar height reduction.14,15 Rates of adverse events such as dyspigmentation, atrophy, and telangiectasias also were lower in patients who received combined therapy.14,15 A systematic review found that intralesional bleomycin may be more effective than TAC alone, 5-FU alone, TAC combined with 5-FU, and TAC combined with cryotherapy; however, hyperpigmentation was a common adverse event, occurring in roughly 70% (42/60) of patients.16,17 Additionally, a 2024 meta-analysis evaluated 20 randomized controlled trials comprising 1114 patients treated with intralesional TAC, 5-FU, BTA, verapamil, and/or bleomycin. Botulinum toxin A and TAC plus 5-FU were found to have outstanding therapeutic efficacy for keloids, and rates of adverse events were similar among users of TAC, 5-FU, BTA, and TAC plus 5-FU.18
While antineoplastic agents and BTA may be promising keloid therapies, further studies demonstrating their efficacy and safety profiles are necessary, particularly regarding dyspigmentation as a potential adverse event, as this may be of concern in patients with darker phototypes.
Laser Therapies—Of all treatment modalities, laser-based keloid therapies have been the most robustly studied in SOC. The 2 main types are ablative (eg, CO2, Er:YAG) and nonablative (eg, pulsed dye, Nd:YAG) lasers. Ablative lasers rapidly heat water molecules within the skin, thereby vaporizing the skin cells in a controlled precise process that reduces scar tissue by removing layers of skin. Nonablative lasers target hemoglobin in blood vessels, reducing oxygen supply and inducing collagen remodeling without damaging the epidermis.19
For patients with SOC, lasers carry a risk for postinflammatory hyperpigmentation.20 To address this risk, recent advancements in laser technology and procedural protocols have aimed to minimize the number of passes and utilize cooling devices21; however, many of these recommendations are based on retrospective reviews and small case series. A 2024 meta-analysis comprising 550 patients found that the combination of fractional CO2 laser therapy and 5-FU was the most effective intervention, markedly reducing Vancouver Scar Scale and pliability scores as well as keloid thickness.22 Conversely, pulsed dye lasers were the least effective in terms of improving scar thickness, pigmentation, and pliability when compared to other treatments.
Randomized controlled trials of laser-based therapies in patients with SOC are lacking in the literature. Future studies should focus on calibrating laser-based therapies for those with darker skin tones and examine the efficacy and adverse effects of ablative and nonablative lasers in patients with SOC.
Promising New Keloid Therapies
Keloid disease pathogenesis is incompletely understood, but several new therapeutic targets have been highlighted in the literature, including dupilumab, pentoxifylline, sirtuin 6 (SIRT6) modulators, remdesivir, and needle-assisted electrocoagulation plus pharmacotherapy.
Dupilumab—An anti–IL-4 and IL-13 monoclonal antibody, dupilumab was first approved for the treatment of severe atopic dermatitis. Its use has broadened since its approval, and keloids have been identified as a potential therapeutic target. A 2019 case study described a 53-year-old Black man with severe atopic dermatitis and chronic keloids that regressed with systemic dupilumab therapy.23 This prompted a follow-up case-control study using real-time polymerase chain reaction testing to evaluate Th2 gene expression (IL-4R, IL-13, and CCL18) of lesional and nonlesional tissue in 3 Black patients with chronic keloids and no concurrent atopic dermatitis vs 5 healthy Black controls. Despite the limited sample size, a significant increase in IL-13 and the Th2 chemokine CCL18 was found in patients with keloids compared to controls (P<.05), suggesting that the entire integument of patients with severe keloids is abnormal.23 This finding supports the use of systemic treatments for chronic and multifocal keloid disease. Several subsequent case reports have corroborated the efficacy of systemic and/or intralesional dupilumab.24,25 However, some studies have reported contradictory findings, suggesting the need for high-quality clinical trials.26,27
Pentoxifylline—Pentoxifylline is a methylated xanthine derivative and a nonspecific phosphodiesterase inhibitor used to treat claudication from peripheral artery disease. It also inhibits the proliferation and rate of collagen synthesis of fibroblasts from keloids in vitro.28,29 A 2019 retrospective, open-label pilot study analyzed postsurgical keloid recurrence in 45 patients with 67 unique keloids that were stratified into low- and high-risk groups based on clinical factors including multiple symptomatic keloids, history of recurrence, and family history.30 Both the low- and the high-risk groups were treated with 40 mg/mL intralesional triamcinolone acetonide monthly for 6 months; however, some of the high-risk keloids also received pentoxifylline 300 mg 3 times daily for 6 months. There was a statistically significant decrease in keloid recurrence rate between the high-risk group treated with pentoxifylline and the low-risk group for whom pentoxifylline was not prescribed (P=.015).
Similarly, a randomized clinical trial comparing the efficacy of combination intralesional pentoxifylline and intralesional triamcinolone vs monotherapy with pentoxifylline or triamcinolone found the most significant improvement in the combination cohort with reduction in keloid height (P=.04), pliability (P=.003), and vascularity (P=.05).31 These findings highlight the need for supplementary studies on the use of pentoxifylline for keloid therapy.
SIRT6 Modulators—SIRT6 modulators are an exciting future therapeutic target. In a recent case-control study evaluating the histologic milieu of keloid tissue vs normal skin specimens, the researchers found that selective overexpression of SIRT6 via the use of a recombinant adenovirus in keloid fibroblasts attenuated proliferation, invasion, and collagen synthesis while fostering apoptosis, likely through the suppression of MAPK/ERK pathway activity.32
Remdesivir—The antiviral drug remdesivir has been reported to have pharmacologic activities in a wide range of fibrotic diseases, including keloids. A 2024 study explored the potential effect and mechanisms of remdesivir on skin fibrosis both in vitro and in rodents.33 Remdesivir was found to decrease skin fibrosis and attenuate the gross weight of keloid tissues in vivo, suppress fibroblast activation and autophagy both in vivo and in vitro, dampen fibroblast activation by the TGF-β1/Smad signaling pathway, and inhibit fibroblasts autophagy by the PI3K/Akt/mTOR signaling pathway. These results demonstrate the therapeutic potential of remdesivir for keloid management.
Needle-Assisted Electrocoagulation Plus Pharmacotherapy—A novel needle-assisted electrocoagulation technique combined with pharmacotherapy (corticosteroid and 5-FU injections) was effective in a Chinese clinical trial involving 6 patients with keloids.34 Investigators used Vancouver Scar Scale and both Patient and Observer Scar Assessment Scale scores to grade patients’ scars before treatment and 1 month after the first treatment cycle. They found that ablation combined with pharmacotherapy significantly reduced all 3 scores without any obvious adverse events (P=.004, P=.006, and P=.017, respectively). This novel combination treatment may serve as a safe and effective therapeutic approach for keloid removal.
Final Thoughts
Emerging treatments offer promising new horizons in keloid management; however, the lack of robust, high-quality clinical trials, especially those focusing on
- Téot L, Mustoe TA, Middelkoop E, eds. Textbook on Scar Management: State of the Art Management and Emerging Technologies. Springer; 2020.
- Davis SA, Feldman SR, McMichael AJ. Management of keloids in the United States, 1990-2009: an analysis of the National Ambulatory Medical Care Survey. Dermatol Surg. 2013;39:988-994. doi:10.1111/dsu.12182
- Kassi K, Kouame K, Kouassi A, et al. Quality of life in black African patients with keloid scars. Dermatol Reports. 2020;12:8312. doi:10.4081/dr.2020.8312
- Delaleu J, Charvet E, Petit A. Keloid disease: review with clinical atlas. part I: definitions, history, epidemiology, clinics and diagnosis. Ann Dermatol Venereol. 2023;150:3-15.
doi:10.1016/j.annder.2022.08.010 - Bronte J, Zhou C, Vempati A, et al. A comprehensive review of non-surgical treatments for hypertrophic and keloid scars in skin of color. Clin Cosmet Investig Dermatol.
2024;17:1459-1469. doi:10.2147/CCID.S470997 - Davison SP, Dayan JH, Clemens MW, et al. Efficacy of intralesional 5-fluorouracil and triamcinolone in the treatment of keloids. Aesthet Surg J. 2009;29:40-46. doi:10.1016/j.asj.2008.11.006
- Azzam OA, Bassiouny DA, El-Hawary MS, et al. Treatment of hypertrophic scars and keloids by fractional carbon dioxide laser: a clinical, histological, and immunohistochemical study. Lasers Med Sci. 2016;31:9-18. doi:10.1007/s10103-015-1824-4
- Ekstein SF, Wyles SP, Moran SL, et al. Keloids: a review of therapeutic management. Int J Dermatol. 2021;60:661-671. doi:10.1111/ijd.15159
- Morelli Coppola M, Salzillo R, Segreto F, et al. Triamcinolone acetonide intralesional injection for the treatment of keloid scars: patient selection and perspectives. Clin Cosmet Investig Dermatol. 2018;11:387-396. doi:10.2147/CCID.S133672
- Kant SB, van den Kerckhove E, Colla C, et al. A new treatment of hypertrophic and keloid scars with combined triamcinolone and verapamil: a retrospective study. Eur J Plast Surg. 2018;41:69-80. doi:10.1007/s00238-017-1322-y
- Cohen AJ, Talasila S, Lazarevic B, et al. Combination cryotherapy and intralesional corticosteroid versus steroid monotherapy in the treatment of keloids. J Cosmet Dermatol. 2023;22:932-936. doi:10.1111/jocd.15520
- Tawaranurak N, Pliensiri P, Tawaranurak K. Combination of fractional carbon dioxide laser and topical triamcinolone vs intralesional triamcinolone for keloid treatment: a randomised clinical trial. Int Wound J. 2022;19:1729-1735. doi:10.1111/iwj.13775
- Belie O, Ugburo AO, Mofikoya BO, et al. A comparison of intralesional verapamil and triamcinolone monotherapy in the treatment of keloids in an African population. Niger J Clin Pract. 2021;24:986-992. doi:10.4103/njcp.njcp_474_20
- Khalid FA, Mehrose MY, Saleem M, et al. Comparison of efficacy and safety of intralesional triamcinolone and combination of triamcinolone with 5-fluorouracil in the treatment of keloids and hypertrophic scars: randomised control trial. Burns. 2019;45:69-75. doi:10.1016/j.burns.2018.08.011
- Asilian A, Darougheh A, Shariati F. New combination of triamcinolone, 5-Fluorouracil, and pulsed-dye laser for treatment of keloid and hypertrophic scars. Dermatol Surg. 2006;32:907-915. doi:10.1111/j.1524-4725.2006.32195.x
- Kim WI, Kim S, Cho SW, et al. The efficacy of bleomycin for treating keloid and hypertrophic scar: a systematic review and meta-analysis. J Cosmet Dermatol. 2020;19:3357-3366. doi:10.1111/jocd.13390
- Kabel A, Sabry H, Sorour N, et al. Comparative study between intralesional injection of bleomycin and 5-fluorouracil in the treatment of keloids and hypertrophic scars. J Dermatol Dermatol Surg. 2016;20:32-38.
- Yang HA, Jheng WL, Yu J, et al. Comparative efficacy of drug interventions for keloids: a network meta-analysis. Ann Plast Surg. 2024;92(1S suppl 1
):S52-S59. doi:10.1097/SAP.0000000000003759 - Preissig J, Hamilton K, Markus R. Current laser resurfacing technologies: a review that delves beneath the surface. Semin Plast Surg. 2012;26:109-116. doi:10.1055/s-0032-1329413
- Bin Dakhil A, Shadid A, Altalhab S. Post-inflammatory hyperpigmentation after carbon dioxide laser: review of prevention and risk factors. Dermatol Reports. 2023;15:9703. doi:10.4081/dr.2023.9703
- Kaushik SB, Alexis AF. Nonablative fractional laser resurfacing in skin of color: evidence-based review. J Clin Aesthet Dermatol. 2017;10:51-67.
- Foppiani JA, Khaity A, Al-Dardery NM, et al. Laser therapy in hypertrophic and keloid scars: a systematic review and network meta-analysis. Aesthetic Plast Surg. Published May 17, 2024. doi:10.1007/s00266-024-04027-9
- Diaz A, Tan K, He H, et al. Keloid lesions show increased IL-4/IL-13 signaling and respond to Th2-targeting dupilumab therapy. J Eur Acad Dermatol Venereol. 2020;34:E161-E164. doi:10.1111/jdv.16097
- Min MS,
Mazori DR, Lee MS, et al. Successful treatment of keloids and hypertrophic scars with systemic and intralesional dupilumab. J Drugs Dermatol. 2023;22:1220-1222. doi:10.36849/JDD.6385 - Wittmer A, Finklea L, Joseph J. Effects of dupilumab on keloid stabilization and prevention. JAAD Case Rep. 2023;37:103-105. doi:10.1016/j.jdcr.2023.05.001
- Luk K, Fakhoury J, Ozog D. Nonresponse and progression of diffuse keloids to dupilumab therapy. J Drugs Dermatol. 2022;21:197-199. doi:10.36849/jdd.6252
- Tirgan MH, Uitto J. Lack of efficacy of dupilumab in the treatment of keloid disorder. J Eur Acad Dermatol Venereol. 2022;36:E120-E122. doi:10.1111/jdv.17669
- Berman B, Duncan MR. Pentoxifylline inhibits the proliferation of human fibroblasts derived from keloid, scleroderma and morphoea skin and their production of collagen, glycosaminoglycans and fibronectin. Br J Dermatol. 1990;123:339-346. doi:10.1111/j.1365-
2133.1990.tb06294.x - Berman B, Duncan MR. Pentoxifylline inhibits normal human dermal fibroblast in vitro proliferation, collagen, glycosaminoglycan, and fibronectin production, and increases collagenase activity. J Invest Dermatol. 1989;92:605-610.
- Tan A, Martinez Luna O, Glass DA 2nd. Pentoxifylline for the prevention of postsurgical keloid recurrence. Dermatol Surg. 2020;46:1353-1356. doi:10.1097/DSS.0000000000002090
- Serag-Eldin YMA, Mahmoud WH, Gamea MM, et al. Intralesional pentoxifylline, triamcinolone acetonide, and their combination for treatment of keloid scars. J Cosmet Dermatol. 2021;20:3330-3340. doi:10.1111/jocd.14305
- Zhou T, Chen Y, Wang C, et al. SIRT6 inhibits the proliferation and collagen synthesis of keloid fibroblasts through MAPK/ERK pathway. Discov Med. 2024;36:1430-1440. doi:10.24976/Discov.Med.202436186.133
- Zhang J, Zhang X, Guo X, et al. Remdesivir alleviates skin fibrosis by suppressing TGF-β1 signaling pathway. PLoS One. 2024;19:E0305927.
doi:10.1371/journal.pone.0305927 - Zhao J, Zhai X, Xu Z, et al. Novel needle-type electrocoagulation and combination pharmacotherapy: basic and clinical studies on efficacy and safety in treating keloids. J Cosmet Dermatol. doi:10.1111/jocd.16453
- Téot L, Mustoe TA, Middelkoop E, eds. Textbook on Scar Management: State of the Art Management and Emerging Technologies. Springer; 2020.
- Davis SA, Feldman SR, McMichael AJ. Management of keloids in the United States, 1990-2009: an analysis of the National Ambulatory Medical Care Survey. Dermatol Surg. 2013;39:988-994. doi:10.1111/dsu.12182
- Kassi K, Kouame K, Kouassi A, et al. Quality of life in black African patients with keloid scars. Dermatol Reports. 2020;12:8312. doi:10.4081/dr.2020.8312
- Delaleu J, Charvet E, Petit A. Keloid disease: review with clinical atlas. part I: definitions, history, epidemiology, clinics and diagnosis. Ann Dermatol Venereol. 2023;150:3-15.
doi:10.1016/j.annder.2022.08.010 - Bronte J, Zhou C, Vempati A, et al. A comprehensive review of non-surgical treatments for hypertrophic and keloid scars in skin of color. Clin Cosmet Investig Dermatol.
2024;17:1459-1469. doi:10.2147/CCID.S470997 - Davison SP, Dayan JH, Clemens MW, et al. Efficacy of intralesional 5-fluorouracil and triamcinolone in the treatment of keloids. Aesthet Surg J. 2009;29:40-46. doi:10.1016/j.asj.2008.11.006
- Azzam OA, Bassiouny DA, El-Hawary MS, et al. Treatment of hypertrophic scars and keloids by fractional carbon dioxide laser: a clinical, histological, and immunohistochemical study. Lasers Med Sci. 2016;31:9-18. doi:10.1007/s10103-015-1824-4
- Ekstein SF, Wyles SP, Moran SL, et al. Keloids: a review of therapeutic management. Int J Dermatol. 2021;60:661-671. doi:10.1111/ijd.15159
- Morelli Coppola M, Salzillo R, Segreto F, et al. Triamcinolone acetonide intralesional injection for the treatment of keloid scars: patient selection and perspectives. Clin Cosmet Investig Dermatol. 2018;11:387-396. doi:10.2147/CCID.S133672
- Kant SB, van den Kerckhove E, Colla C, et al. A new treatment of hypertrophic and keloid scars with combined triamcinolone and verapamil: a retrospective study. Eur J Plast Surg. 2018;41:69-80. doi:10.1007/s00238-017-1322-y
- Cohen AJ, Talasila S, Lazarevic B, et al. Combination cryotherapy and intralesional corticosteroid versus steroid monotherapy in the treatment of keloids. J Cosmet Dermatol. 2023;22:932-936. doi:10.1111/jocd.15520
- Tawaranurak N, Pliensiri P, Tawaranurak K. Combination of fractional carbon dioxide laser and topical triamcinolone vs intralesional triamcinolone for keloid treatment: a randomised clinical trial. Int Wound J. 2022;19:1729-1735. doi:10.1111/iwj.13775
- Belie O, Ugburo AO, Mofikoya BO, et al. A comparison of intralesional verapamil and triamcinolone monotherapy in the treatment of keloids in an African population. Niger J Clin Pract. 2021;24:986-992. doi:10.4103/njcp.njcp_474_20
- Khalid FA, Mehrose MY, Saleem M, et al. Comparison of efficacy and safety of intralesional triamcinolone and combination of triamcinolone with 5-fluorouracil in the treatment of keloids and hypertrophic scars: randomised control trial. Burns. 2019;45:69-75. doi:10.1016/j.burns.2018.08.011
- Asilian A, Darougheh A, Shariati F. New combination of triamcinolone, 5-Fluorouracil, and pulsed-dye laser for treatment of keloid and hypertrophic scars. Dermatol Surg. 2006;32:907-915. doi:10.1111/j.1524-4725.2006.32195.x
- Kim WI, Kim S, Cho SW, et al. The efficacy of bleomycin for treating keloid and hypertrophic scar: a systematic review and meta-analysis. J Cosmet Dermatol. 2020;19:3357-3366. doi:10.1111/jocd.13390
- Kabel A, Sabry H, Sorour N, et al. Comparative study between intralesional injection of bleomycin and 5-fluorouracil in the treatment of keloids and hypertrophic scars. J Dermatol Dermatol Surg. 2016;20:32-38.
- Yang HA, Jheng WL, Yu J, et al. Comparative efficacy of drug interventions for keloids: a network meta-analysis. Ann Plast Surg. 2024;92(1S suppl 1
):S52-S59. doi:10.1097/SAP.0000000000003759 - Preissig J, Hamilton K, Markus R. Current laser resurfacing technologies: a review that delves beneath the surface. Semin Plast Surg. 2012;26:109-116. doi:10.1055/s-0032-1329413
- Bin Dakhil A, Shadid A, Altalhab S. Post-inflammatory hyperpigmentation after carbon dioxide laser: review of prevention and risk factors. Dermatol Reports. 2023;15:9703. doi:10.4081/dr.2023.9703
- Kaushik SB, Alexis AF. Nonablative fractional laser resurfacing in skin of color: evidence-based review. J Clin Aesthet Dermatol. 2017;10:51-67.
- Foppiani JA, Khaity A, Al-Dardery NM, et al. Laser therapy in hypertrophic and keloid scars: a systematic review and network meta-analysis. Aesthetic Plast Surg. Published May 17, 2024. doi:10.1007/s00266-024-04027-9
- Diaz A, Tan K, He H, et al. Keloid lesions show increased IL-4/IL-13 signaling and respond to Th2-targeting dupilumab therapy. J Eur Acad Dermatol Venereol. 2020;34:E161-E164. doi:10.1111/jdv.16097
- Min MS,
Mazori DR, Lee MS, et al. Successful treatment of keloids and hypertrophic scars with systemic and intralesional dupilumab. J Drugs Dermatol. 2023;22:1220-1222. doi:10.36849/JDD.6385 - Wittmer A, Finklea L, Joseph J. Effects of dupilumab on keloid stabilization and prevention. JAAD Case Rep. 2023;37:103-105. doi:10.1016/j.jdcr.2023.05.001
- Luk K, Fakhoury J, Ozog D. Nonresponse and progression of diffuse keloids to dupilumab therapy. J Drugs Dermatol. 2022;21:197-199. doi:10.36849/jdd.6252
- Tirgan MH, Uitto J. Lack of efficacy of dupilumab in the treatment of keloid disorder. J Eur Acad Dermatol Venereol. 2022;36:E120-E122. doi:10.1111/jdv.17669
- Berman B, Duncan MR. Pentoxifylline inhibits the proliferation of human fibroblasts derived from keloid, scleroderma and morphoea skin and their production of collagen, glycosaminoglycans and fibronectin. Br J Dermatol. 1990;123:339-346. doi:10.1111/j.1365-
2133.1990.tb06294.x - Berman B, Duncan MR. Pentoxifylline inhibits normal human dermal fibroblast in vitro proliferation, collagen, glycosaminoglycan, and fibronectin production, and increases collagenase activity. J Invest Dermatol. 1989;92:605-610.
- Tan A, Martinez Luna O, Glass DA 2nd. Pentoxifylline for the prevention of postsurgical keloid recurrence. Dermatol Surg. 2020;46:1353-1356. doi:10.1097/DSS.0000000000002090
- Serag-Eldin YMA, Mahmoud WH, Gamea MM, et al. Intralesional pentoxifylline, triamcinolone acetonide, and their combination for treatment of keloid scars. J Cosmet Dermatol. 2021;20:3330-3340. doi:10.1111/jocd.14305
- Zhou T, Chen Y, Wang C, et al. SIRT6 inhibits the proliferation and collagen synthesis of keloid fibroblasts through MAPK/ERK pathway. Discov Med. 2024;36:1430-1440. doi:10.24976/Discov.Med.202436186.133
- Zhang J, Zhang X, Guo X, et al. Remdesivir alleviates skin fibrosis by suppressing TGF-β1 signaling pathway. PLoS One. 2024;19:E0305927.
doi:10.1371/journal.pone.0305927 - Zhao J, Zhai X, Xu Z, et al. Novel needle-type electrocoagulation and combination pharmacotherapy: basic and clinical studies on efficacy and safety in treating keloids. J Cosmet Dermatol. doi:10.1111/jocd.16453
When Your Malpractice Insurer Investigates You: What to Know
When psychiatrist Paul Sartain, MD (not his real name), received a letter from his state’s medical board, he was concerned. A patient’s family complained that he made sexual advances to a young woman he treated for psychotic depression.
“There was absolutely no evidence, and the claims were vague,” he said. “I think the family was angry at me and with the system — the woman had not gotten better.” Sartain reviewed his medical records and then called his malpractice insurer.
The insurer asked about his involvement with the patient’s case, if there was anything credible to the patient’s complaint, and if he had thorough documentation. Then, the carrier offered Sartain his choice of several attorneys who could represent him. The medical board ultimately closed the case with no findings against him, and the patient’s family never sued him.
“If I’m wrongly accused, I’m defended (by the carrier). If I had stolen money or had a sexual relationship with the patient, then you’re acting outside the bounds of what is protected (by the carrier),” he said.
How Medical Board and Malpractice Insurer Investigations Differ
Medical board complaints differ from malpractice claims, in which patients seek damages. The investigation process also varies.
When a patient reports a doctor to a state medical board, they may also sue the doctor for monetary damages in civil court. The medical board responds to patient complaints made directly to them, but it also may also initiate its own investigations. Those can be prompted by a malpractice claim resolution, with a court verdict against the doctor, or a settlement recorded in the National Practitioner Data Bank.
Malpractice insurers may offer limited legal representation for medical board investigations, requiring the doctor to report the medical board issue to them before the doctor takes any action. Often, they will cover up to $50,000 in defense costs but not cover any subsequent medical board fines or required classes or medical board fees.
When a doctor contacts the carrier about a medical board investigation, the carrier may ask for the medical board document and the medical records, said Alex Keoskey, a partner in Frier Levitt’s life sciences group.
The carrier may want to ask about the patient, staff members involved, the doctor’s background, if there have been previous medical board investigations or lawsuits against this doctor, and the doctor’s opinion of the allegations. The doctor should be transparent with the carrier, Keoskey said.
Some carriers conduct more in-depth investigations, examining record-keeping, prescription practices, patient consent processes, and continuing medical education status. That’s because the medical board may inquire about these as well should its own investigation expand.
Not all carriers explore cases like these, even if reimbursing for defense costs, said Karen Frisella, director of professional liability claims at BETA Healthcare Group in California. In her experience, a licensing investigation usually follows a claim resolution that was already worked up by the carrier. If a complaint was made directly to the licensing board without an accompanying liability claim, the carrier’s ability to initiate an investigation on the incident depends on the policy terms or coverage available.
“Typically, a professional liability policy requires that the insured report a claim to trigger coverage. The carrier can’t unilaterally decide to open a claim,” she said. A licensing board investigation is not a claim by definition and therefore does not provide a mechanism for the carrier to open a liability claim file, she added.
If the medical board ultimately restricts the doctor’s license or puts the doctor on probation, that becomes public, and the underwriting department may then look into it.
Malpractice insurers routinely monitor licensing board discipline notices. A reprimand or restrictions on a doctor’s license could trigger a review of the physician’s future insurability and lead to higher premiums or even nonrenewal, Frisella said.
If a carrier investigates a reported claim and determines there are issues with the care rendered, whether there is an accompanying medical board action, that also can affect underwriting decisions, Frisella said.
Who Is Your Attorney Really Working for?
The doctor should understand whose interests the attorney represents. In a medical board claim, the attorney — even if defense is paid by the carrier — represents the doctor.
Frisella said her organization provides pass-through coverage, meaning it reimburses the doctor for medical board defense costs. “Because the carrier isn’t directing the medical board defense, it is not generally privy to the work product.”
If a patient files a malpractice claim, however, the attorney ultimately represents the insurance company.
“The panel counsel who works for the insurer does not work for the doctor, and that’s always important to remember,” Keoskey said. While the attorney will do their best to aggressively defend the doctor, “he’s going to protect the insurer’s interest before the doctor’s.”
Physicians who find any conflict of interest with their insurer should seek counsel.
Such conflicts could include:
- Disagreements over the case’s ultimate worth. For example, a physician might want a case to settle for less than their carrier is willing to pay.
- The legal judgment may exceed the carrier’s policy limits, or there are punitive damages or allegations of criminal acts that the insurer does not cover.
In these cases, the insurance company should recommend the doctor get personal counsel. They will send a reservation of rights letter saying they will defend the doctor for now, but if the facts show the doctor committed some type of misconduct, they may decline coverage, said Keoskey. Some states, including California, require that the carrier pay for this independent counsel.
Unless there is a conflict of interest, though, having a personal attorney just makes the situation more complicated, said Frisella.
A version of this article first appeared on Medscape.com.
When psychiatrist Paul Sartain, MD (not his real name), received a letter from his state’s medical board, he was concerned. A patient’s family complained that he made sexual advances to a young woman he treated for psychotic depression.
“There was absolutely no evidence, and the claims were vague,” he said. “I think the family was angry at me and with the system — the woman had not gotten better.” Sartain reviewed his medical records and then called his malpractice insurer.
The insurer asked about his involvement with the patient’s case, if there was anything credible to the patient’s complaint, and if he had thorough documentation. Then, the carrier offered Sartain his choice of several attorneys who could represent him. The medical board ultimately closed the case with no findings against him, and the patient’s family never sued him.
“If I’m wrongly accused, I’m defended (by the carrier). If I had stolen money or had a sexual relationship with the patient, then you’re acting outside the bounds of what is protected (by the carrier),” he said.
How Medical Board and Malpractice Insurer Investigations Differ
Medical board complaints differ from malpractice claims, in which patients seek damages. The investigation process also varies.
When a patient reports a doctor to a state medical board, they may also sue the doctor for monetary damages in civil court. The medical board responds to patient complaints made directly to them, but it also may also initiate its own investigations. Those can be prompted by a malpractice claim resolution, with a court verdict against the doctor, or a settlement recorded in the National Practitioner Data Bank.
Malpractice insurers may offer limited legal representation for medical board investigations, requiring the doctor to report the medical board issue to them before the doctor takes any action. Often, they will cover up to $50,000 in defense costs but not cover any subsequent medical board fines or required classes or medical board fees.
When a doctor contacts the carrier about a medical board investigation, the carrier may ask for the medical board document and the medical records, said Alex Keoskey, a partner in Frier Levitt’s life sciences group.
The carrier may want to ask about the patient, staff members involved, the doctor’s background, if there have been previous medical board investigations or lawsuits against this doctor, and the doctor’s opinion of the allegations. The doctor should be transparent with the carrier, Keoskey said.
Some carriers conduct more in-depth investigations, examining record-keeping, prescription practices, patient consent processes, and continuing medical education status. That’s because the medical board may inquire about these as well should its own investigation expand.
Not all carriers explore cases like these, even if reimbursing for defense costs, said Karen Frisella, director of professional liability claims at BETA Healthcare Group in California. In her experience, a licensing investigation usually follows a claim resolution that was already worked up by the carrier. If a complaint was made directly to the licensing board without an accompanying liability claim, the carrier’s ability to initiate an investigation on the incident depends on the policy terms or coverage available.
“Typically, a professional liability policy requires that the insured report a claim to trigger coverage. The carrier can’t unilaterally decide to open a claim,” she said. A licensing board investigation is not a claim by definition and therefore does not provide a mechanism for the carrier to open a liability claim file, she added.
If the medical board ultimately restricts the doctor’s license or puts the doctor on probation, that becomes public, and the underwriting department may then look into it.
Malpractice insurers routinely monitor licensing board discipline notices. A reprimand or restrictions on a doctor’s license could trigger a review of the physician’s future insurability and lead to higher premiums or even nonrenewal, Frisella said.
If a carrier investigates a reported claim and determines there are issues with the care rendered, whether there is an accompanying medical board action, that also can affect underwriting decisions, Frisella said.
Who Is Your Attorney Really Working for?
The doctor should understand whose interests the attorney represents. In a medical board claim, the attorney — even if defense is paid by the carrier — represents the doctor.
Frisella said her organization provides pass-through coverage, meaning it reimburses the doctor for medical board defense costs. “Because the carrier isn’t directing the medical board defense, it is not generally privy to the work product.”
If a patient files a malpractice claim, however, the attorney ultimately represents the insurance company.
“The panel counsel who works for the insurer does not work for the doctor, and that’s always important to remember,” Keoskey said. While the attorney will do their best to aggressively defend the doctor, “he’s going to protect the insurer’s interest before the doctor’s.”
Physicians who find any conflict of interest with their insurer should seek counsel.
Such conflicts could include:
- Disagreements over the case’s ultimate worth. For example, a physician might want a case to settle for less than their carrier is willing to pay.
- The legal judgment may exceed the carrier’s policy limits, or there are punitive damages or allegations of criminal acts that the insurer does not cover.
In these cases, the insurance company should recommend the doctor get personal counsel. They will send a reservation of rights letter saying they will defend the doctor for now, but if the facts show the doctor committed some type of misconduct, they may decline coverage, said Keoskey. Some states, including California, require that the carrier pay for this independent counsel.
Unless there is a conflict of interest, though, having a personal attorney just makes the situation more complicated, said Frisella.
A version of this article first appeared on Medscape.com.
When psychiatrist Paul Sartain, MD (not his real name), received a letter from his state’s medical board, he was concerned. A patient’s family complained that he made sexual advances to a young woman he treated for psychotic depression.
“There was absolutely no evidence, and the claims were vague,” he said. “I think the family was angry at me and with the system — the woman had not gotten better.” Sartain reviewed his medical records and then called his malpractice insurer.
The insurer asked about his involvement with the patient’s case, if there was anything credible to the patient’s complaint, and if he had thorough documentation. Then, the carrier offered Sartain his choice of several attorneys who could represent him. The medical board ultimately closed the case with no findings against him, and the patient’s family never sued him.
“If I’m wrongly accused, I’m defended (by the carrier). If I had stolen money or had a sexual relationship with the patient, then you’re acting outside the bounds of what is protected (by the carrier),” he said.
How Medical Board and Malpractice Insurer Investigations Differ
Medical board complaints differ from malpractice claims, in which patients seek damages. The investigation process also varies.
When a patient reports a doctor to a state medical board, they may also sue the doctor for monetary damages in civil court. The medical board responds to patient complaints made directly to them, but it also may also initiate its own investigations. Those can be prompted by a malpractice claim resolution, with a court verdict against the doctor, or a settlement recorded in the National Practitioner Data Bank.
Malpractice insurers may offer limited legal representation for medical board investigations, requiring the doctor to report the medical board issue to them before the doctor takes any action. Often, they will cover up to $50,000 in defense costs but not cover any subsequent medical board fines or required classes or medical board fees.
When a doctor contacts the carrier about a medical board investigation, the carrier may ask for the medical board document and the medical records, said Alex Keoskey, a partner in Frier Levitt’s life sciences group.
The carrier may want to ask about the patient, staff members involved, the doctor’s background, if there have been previous medical board investigations or lawsuits against this doctor, and the doctor’s opinion of the allegations. The doctor should be transparent with the carrier, Keoskey said.
Some carriers conduct more in-depth investigations, examining record-keeping, prescription practices, patient consent processes, and continuing medical education status. That’s because the medical board may inquire about these as well should its own investigation expand.
Not all carriers explore cases like these, even if reimbursing for defense costs, said Karen Frisella, director of professional liability claims at BETA Healthcare Group in California. In her experience, a licensing investigation usually follows a claim resolution that was already worked up by the carrier. If a complaint was made directly to the licensing board without an accompanying liability claim, the carrier’s ability to initiate an investigation on the incident depends on the policy terms or coverage available.
“Typically, a professional liability policy requires that the insured report a claim to trigger coverage. The carrier can’t unilaterally decide to open a claim,” she said. A licensing board investigation is not a claim by definition and therefore does not provide a mechanism for the carrier to open a liability claim file, she added.
If the medical board ultimately restricts the doctor’s license or puts the doctor on probation, that becomes public, and the underwriting department may then look into it.
Malpractice insurers routinely monitor licensing board discipline notices. A reprimand or restrictions on a doctor’s license could trigger a review of the physician’s future insurability and lead to higher premiums or even nonrenewal, Frisella said.
If a carrier investigates a reported claim and determines there are issues with the care rendered, whether there is an accompanying medical board action, that also can affect underwriting decisions, Frisella said.
Who Is Your Attorney Really Working for?
The doctor should understand whose interests the attorney represents. In a medical board claim, the attorney — even if defense is paid by the carrier — represents the doctor.
Frisella said her organization provides pass-through coverage, meaning it reimburses the doctor for medical board defense costs. “Because the carrier isn’t directing the medical board defense, it is not generally privy to the work product.”
If a patient files a malpractice claim, however, the attorney ultimately represents the insurance company.
“The panel counsel who works for the insurer does not work for the doctor, and that’s always important to remember,” Keoskey said. While the attorney will do their best to aggressively defend the doctor, “he’s going to protect the insurer’s interest before the doctor’s.”
Physicians who find any conflict of interest with their insurer should seek counsel.
Such conflicts could include:
- Disagreements over the case’s ultimate worth. For example, a physician might want a case to settle for less than their carrier is willing to pay.
- The legal judgment may exceed the carrier’s policy limits, or there are punitive damages or allegations of criminal acts that the insurer does not cover.
In these cases, the insurance company should recommend the doctor get personal counsel. They will send a reservation of rights letter saying they will defend the doctor for now, but if the facts show the doctor committed some type of misconduct, they may decline coverage, said Keoskey. Some states, including California, require that the carrier pay for this independent counsel.
Unless there is a conflict of interest, though, having a personal attorney just makes the situation more complicated, said Frisella.
A version of this article first appeared on Medscape.com.
The Rise of Sham Peer Reviews
While a medical peer review occurs once a patient, fellow doctor, or staff member reports that a physician failed to treat a patient up to standards or acted improperly, a “sham peer review” is undertaken for ulterior motives.
They’re typically undertaken due to professional competition or institutional politics rather than to promote quality care or uphold professional standards.
Physicians should be concerned. In a soon-to-be-published Medscape report on peer reviews, 56% of US physicians surveyed expressed higher levels of concern that a peer review could be misused to punish a physician for reasons unrelated to the matter being reviewed.
This is a troublesome issue, and many doctors may not be aware of it or how often it occurs.
“The biggest misconception about sham peer reviews is a denial of how pervasive they are,” said Andy Schlafly, general counsel for the Association of American Physicians and Surgeons (AAPS), which offers a free legal consultation service for physicians facing a sham peer review. “Many hospital administrations are as dangerous to good physicians as street gangs can be in a crime-ridden neighborhood.”
“Physicians should become aware of whether sham peer reviews are prevalent at their hospital and, if so, those physicians should look to practice somewhere else,” Schlafly said in an interview.
Unfortunately, there are limited data on how often this happens. When it does, it can be a career killer, said Lawrence Huntoon, MD, PhD, who has run the AAPS sham peer review hotline for over 20 years.
The physicians at the most risk for a sham peer review tend to be those who work for large hospital systems — as this is one way for hospitals to get rid of the doctors they don’t want to retain on staff, Huntoon said.
“Hospitals want a model whereby every physician on the medical staff is an employee,” Huntoon added. “This gives them complete power and control over these physicians, including the way they practice and how many patients they see per day, which, for some, is 20-50 a day to generate sufficient revenue.”
Complaints are generally filed via incident reporting software.
“The complaint could be that the physician is ‘disruptive,’ which can include facial expression, tone of voice, and body language — for example, ‘I found his facial expression demeaning’ or ‘I found her tone condescending’ — and this can be used to prosecute a doctor,” Huntoon said.
After the complaint is filed, the leaders of a hospital’s peer review committee meet to discuss the incident, followed by a panel of fellow physicians convened to review the matter. Once the date for a meeting is set, the accused doctor is allowed to testify, offer evidence, and have attorney representation.
The entire experience can take a physician by surprise.
“A sham peer review is difficult to prepare for because no physician thinks this is going to happen to them,” said Laurie L. York, a medical law attorney in Austin, Texas.
York added that there may also be a misperception of what is actually happening.
“When a physician becomes aware of an investigation, it initially may look like a regular peer review, and the physician may feel there has been a ‘misunderstanding’ that they can make right by explaining things,” York said. “The window of opportunity to shut down a sham peer review happens quickly. That’s why the physician needs the help of an experienced attorney as early in the process as possible.”
If You’re a Victim of a Sham Peer Review
Be vigilant. The most important thing you should think about when it comes to sham peer reviews is that this can, indeed, happen to you, Huntoon said. “I’ve written articles to help educate physicians about the tactics that are used,” he said. “You need to be educated and read medical staff bylaws to know your rights before something bad happens.”
Stay in your job. No matter what, if you’re under review, do not resign your position, no matter how difficult this may be. “A resignation during a sham peer review triggers an adverse report to the National Practitioner Data Bank [NPDB],” Schlafly said. The NPDB is a flagging system created by Congress to improve healthcare quality and reduce healthcare fraud and abuse. “A resignation also waives the physician’s right to contest the unfair review. In addition, leverage to negotiate a favorable settlement is lost if the physician simply resigns.”
Get a lawyer on board early. This is the only way to protect your rights. “Don’t wait a year to get an attorney involved,” Huntoon said. But this also can’t be any lawyer. It’s critical to find someone who specializes in sham peer reviews, so be sure to ask about their experience in handling peer review matters in hospitals and how knowledgeable they are about databank reporting requirements. “Sometimes, doctors will hire a malpractice attorney with no knowledge of what happens with sham peer reviews, and they may give bad advice,” he said. “Others may hire an employment attorney and that attorney will be up on employment law but has no experience with peer review matters in hospitals.”
Given the seriousness of a sham peer review, following these guidelines can help.
Contact the AAPA right away. There are things that can be done early on like getting a withdrawal of the request for corrective action as well as obtaining a preliminary injunction. Preparing for the fallout that may occur can be just as challenging.
“After this situation, the doctor is damaged goods,” Huntoon said. “What hospital will want to hire damaged goods to be part of their medical staff? Finding employment is going to be challenging and opening your own practice may also be difficult because the insurers have access to data bank reports.”
Ultimately, the best advice Huntoon can offer is to do your best to stay one step ahead of any work issues that could even lead to a sham peer review.
“Try and shield yourself from a sham peer review and be prepared should it happen,” he said. “I’ve seen careers end in the blink of an eye — wrongfully.”
A version of this article first appeared on Medscape.com.
While a medical peer review occurs once a patient, fellow doctor, or staff member reports that a physician failed to treat a patient up to standards or acted improperly, a “sham peer review” is undertaken for ulterior motives.
They’re typically undertaken due to professional competition or institutional politics rather than to promote quality care or uphold professional standards.
Physicians should be concerned. In a soon-to-be-published Medscape report on peer reviews, 56% of US physicians surveyed expressed higher levels of concern that a peer review could be misused to punish a physician for reasons unrelated to the matter being reviewed.
This is a troublesome issue, and many doctors may not be aware of it or how often it occurs.
“The biggest misconception about sham peer reviews is a denial of how pervasive they are,” said Andy Schlafly, general counsel for the Association of American Physicians and Surgeons (AAPS), which offers a free legal consultation service for physicians facing a sham peer review. “Many hospital administrations are as dangerous to good physicians as street gangs can be in a crime-ridden neighborhood.”
“Physicians should become aware of whether sham peer reviews are prevalent at their hospital and, if so, those physicians should look to practice somewhere else,” Schlafly said in an interview.
Unfortunately, there are limited data on how often this happens. When it does, it can be a career killer, said Lawrence Huntoon, MD, PhD, who has run the AAPS sham peer review hotline for over 20 years.
The physicians at the most risk for a sham peer review tend to be those who work for large hospital systems — as this is one way for hospitals to get rid of the doctors they don’t want to retain on staff, Huntoon said.
“Hospitals want a model whereby every physician on the medical staff is an employee,” Huntoon added. “This gives them complete power and control over these physicians, including the way they practice and how many patients they see per day, which, for some, is 20-50 a day to generate sufficient revenue.”
Complaints are generally filed via incident reporting software.
“The complaint could be that the physician is ‘disruptive,’ which can include facial expression, tone of voice, and body language — for example, ‘I found his facial expression demeaning’ or ‘I found her tone condescending’ — and this can be used to prosecute a doctor,” Huntoon said.
After the complaint is filed, the leaders of a hospital’s peer review committee meet to discuss the incident, followed by a panel of fellow physicians convened to review the matter. Once the date for a meeting is set, the accused doctor is allowed to testify, offer evidence, and have attorney representation.
The entire experience can take a physician by surprise.
“A sham peer review is difficult to prepare for because no physician thinks this is going to happen to them,” said Laurie L. York, a medical law attorney in Austin, Texas.
York added that there may also be a misperception of what is actually happening.
“When a physician becomes aware of an investigation, it initially may look like a regular peer review, and the physician may feel there has been a ‘misunderstanding’ that they can make right by explaining things,” York said. “The window of opportunity to shut down a sham peer review happens quickly. That’s why the physician needs the help of an experienced attorney as early in the process as possible.”
If You’re a Victim of a Sham Peer Review
Be vigilant. The most important thing you should think about when it comes to sham peer reviews is that this can, indeed, happen to you, Huntoon said. “I’ve written articles to help educate physicians about the tactics that are used,” he said. “You need to be educated and read medical staff bylaws to know your rights before something bad happens.”
Stay in your job. No matter what, if you’re under review, do not resign your position, no matter how difficult this may be. “A resignation during a sham peer review triggers an adverse report to the National Practitioner Data Bank [NPDB],” Schlafly said. The NPDB is a flagging system created by Congress to improve healthcare quality and reduce healthcare fraud and abuse. “A resignation also waives the physician’s right to contest the unfair review. In addition, leverage to negotiate a favorable settlement is lost if the physician simply resigns.”
Get a lawyer on board early. This is the only way to protect your rights. “Don’t wait a year to get an attorney involved,” Huntoon said. But this also can’t be any lawyer. It’s critical to find someone who specializes in sham peer reviews, so be sure to ask about their experience in handling peer review matters in hospitals and how knowledgeable they are about databank reporting requirements. “Sometimes, doctors will hire a malpractice attorney with no knowledge of what happens with sham peer reviews, and they may give bad advice,” he said. “Others may hire an employment attorney and that attorney will be up on employment law but has no experience with peer review matters in hospitals.”
Given the seriousness of a sham peer review, following these guidelines can help.
Contact the AAPA right away. There are things that can be done early on like getting a withdrawal of the request for corrective action as well as obtaining a preliminary injunction. Preparing for the fallout that may occur can be just as challenging.
“After this situation, the doctor is damaged goods,” Huntoon said. “What hospital will want to hire damaged goods to be part of their medical staff? Finding employment is going to be challenging and opening your own practice may also be difficult because the insurers have access to data bank reports.”
Ultimately, the best advice Huntoon can offer is to do your best to stay one step ahead of any work issues that could even lead to a sham peer review.
“Try and shield yourself from a sham peer review and be prepared should it happen,” he said. “I’ve seen careers end in the blink of an eye — wrongfully.”
A version of this article first appeared on Medscape.com.
While a medical peer review occurs once a patient, fellow doctor, or staff member reports that a physician failed to treat a patient up to standards or acted improperly, a “sham peer review” is undertaken for ulterior motives.
They’re typically undertaken due to professional competition or institutional politics rather than to promote quality care or uphold professional standards.
Physicians should be concerned. In a soon-to-be-published Medscape report on peer reviews, 56% of US physicians surveyed expressed higher levels of concern that a peer review could be misused to punish a physician for reasons unrelated to the matter being reviewed.
This is a troublesome issue, and many doctors may not be aware of it or how often it occurs.
“The biggest misconception about sham peer reviews is a denial of how pervasive they are,” said Andy Schlafly, general counsel for the Association of American Physicians and Surgeons (AAPS), which offers a free legal consultation service for physicians facing a sham peer review. “Many hospital administrations are as dangerous to good physicians as street gangs can be in a crime-ridden neighborhood.”
“Physicians should become aware of whether sham peer reviews are prevalent at their hospital and, if so, those physicians should look to practice somewhere else,” Schlafly said in an interview.
Unfortunately, there are limited data on how often this happens. When it does, it can be a career killer, said Lawrence Huntoon, MD, PhD, who has run the AAPS sham peer review hotline for over 20 years.
The physicians at the most risk for a sham peer review tend to be those who work for large hospital systems — as this is one way for hospitals to get rid of the doctors they don’t want to retain on staff, Huntoon said.
“Hospitals want a model whereby every physician on the medical staff is an employee,” Huntoon added. “This gives them complete power and control over these physicians, including the way they practice and how many patients they see per day, which, for some, is 20-50 a day to generate sufficient revenue.”
Complaints are generally filed via incident reporting software.
“The complaint could be that the physician is ‘disruptive,’ which can include facial expression, tone of voice, and body language — for example, ‘I found his facial expression demeaning’ or ‘I found her tone condescending’ — and this can be used to prosecute a doctor,” Huntoon said.
After the complaint is filed, the leaders of a hospital’s peer review committee meet to discuss the incident, followed by a panel of fellow physicians convened to review the matter. Once the date for a meeting is set, the accused doctor is allowed to testify, offer evidence, and have attorney representation.
The entire experience can take a physician by surprise.
“A sham peer review is difficult to prepare for because no physician thinks this is going to happen to them,” said Laurie L. York, a medical law attorney in Austin, Texas.
York added that there may also be a misperception of what is actually happening.
“When a physician becomes aware of an investigation, it initially may look like a regular peer review, and the physician may feel there has been a ‘misunderstanding’ that they can make right by explaining things,” York said. “The window of opportunity to shut down a sham peer review happens quickly. That’s why the physician needs the help of an experienced attorney as early in the process as possible.”
If You’re a Victim of a Sham Peer Review
Be vigilant. The most important thing you should think about when it comes to sham peer reviews is that this can, indeed, happen to you, Huntoon said. “I’ve written articles to help educate physicians about the tactics that are used,” he said. “You need to be educated and read medical staff bylaws to know your rights before something bad happens.”
Stay in your job. No matter what, if you’re under review, do not resign your position, no matter how difficult this may be. “A resignation during a sham peer review triggers an adverse report to the National Practitioner Data Bank [NPDB],” Schlafly said. The NPDB is a flagging system created by Congress to improve healthcare quality and reduce healthcare fraud and abuse. “A resignation also waives the physician’s right to contest the unfair review. In addition, leverage to negotiate a favorable settlement is lost if the physician simply resigns.”
Get a lawyer on board early. This is the only way to protect your rights. “Don’t wait a year to get an attorney involved,” Huntoon said. But this also can’t be any lawyer. It’s critical to find someone who specializes in sham peer reviews, so be sure to ask about their experience in handling peer review matters in hospitals and how knowledgeable they are about databank reporting requirements. “Sometimes, doctors will hire a malpractice attorney with no knowledge of what happens with sham peer reviews, and they may give bad advice,” he said. “Others may hire an employment attorney and that attorney will be up on employment law but has no experience with peer review matters in hospitals.”
Given the seriousness of a sham peer review, following these guidelines can help.
Contact the AAPA right away. There are things that can be done early on like getting a withdrawal of the request for corrective action as well as obtaining a preliminary injunction. Preparing for the fallout that may occur can be just as challenging.
“After this situation, the doctor is damaged goods,” Huntoon said. “What hospital will want to hire damaged goods to be part of their medical staff? Finding employment is going to be challenging and opening your own practice may also be difficult because the insurers have access to data bank reports.”
Ultimately, the best advice Huntoon can offer is to do your best to stay one step ahead of any work issues that could even lead to a sham peer review.
“Try and shield yourself from a sham peer review and be prepared should it happen,” he said. “I’ve seen careers end in the blink of an eye — wrongfully.”
A version of this article first appeared on Medscape.com.