User login
Bringing you the latest news, research and reviews, exclusive interviews, podcasts, quizzes, and more.
div[contains(@class, 'header__large-screen')]
div[contains(@class, 'read-next-article')]
div[contains(@class, 'nav-primary')]
nav[contains(@class, 'nav-primary')]
section[contains(@class, 'footer-nav-section-wrapper')]
footer[@id='footer']
div[contains(@class, 'main-prefix')]
section[contains(@class, 'nav-hidden')]
div[contains(@class, 'ce-card-content')]
nav[contains(@class, 'nav-ce-stack')]
COVID-19 vaccinations lag in youngest children
Case: A 3-year-old girl presented to the emergency department after a brief seizure at home. She looked well on physical exam except for a fever of 103° F and thick rhinorrhea.
The intern on duty methodically worked through the standard list of questions. “Immunizations up to date?” she asked.
“Absolutely,” the child’s mom responded. “She’s had everything that’s recommended.”
“Including COVID-19 vaccine?” the intern prompted.
“No.” The mom responded with a shake of her head. “We don’t do that vaccine.”
That mom is not alone.
COVID-19 vaccines for children as young as 6 months were given emergency-use authorization by the Food and Drug Administration in June 2022 and in February 2023, the Advisory Committee on Immunization Practices included COVID-19 vaccine on the routine childhood immunization schedule.
COVID-19 vaccines are safe in young children, and they prevent the most severe outcomes associated with infection, including hospitalization. Newly released data confirm that the COVID-19 vaccines produced by Moderna and Pfizer also provide protection against symptomatic infection for at least 4 months after completion of the monovalent primary series.
In a Morbidity and Mortality Weekly Report released on Feb. 17, 2023, the Centers for Disease Control and Prevention reported the results of a test-negative design case-control study that enrolled symptomatic children tested for SARS-CoV-2 infection through Feb. 5, 2023, as part of the Increasing Community Access to Testing (ICATT) program.1 ICATT provides SARS-CoV-2 testing to persons aged at least 3 years at pharmacy and community-based testing sites nationwide.
Two doses of monovalent Moderna vaccine (complete primary series) was 60% effective against symptomatic infection (95% confidence interval, 49%-68%) 2 weeks to 2 months after receipt of the second dose. Vaccine effectiveness dropped to 36% (95% CI, 15%-52%) 3-4 months after the second dose. Three doses of monovalent Pfizer-BioNTech vaccine (complete primary series) was 31% effective (95% CI, 7%-49%) at preventing symptomatic infection 2 weeks to 4 months after receipt of the third dose. A bivalent vaccine dose for eligible children is expected to provide more protection against currently circulating SARS-CoV-2 variants.
Despite evidence of vaccine efficacy, very few parents are opting to protect their young children with the COVID-19 vaccine. The CDC reports that, as of March 1, 2023, only 8% of children under 2 years and 10.5% of children aged 2-4 years have initiated a COVID vaccine series. The American Academy of Pediatrics has emphasized that 15.0 million children between the ages of 6 months and 4 years have not yet received their first COVID-19 vaccine dose.
While the reasons underlying low COVID-19 vaccination rates in young children are complex, themes emerge. Socioeconomic disparities contributing to low vaccination rates in young children were highlighted in another recent MMWR article.2 Through Dec. 1, 2022, vaccination coverage was lower in rural counties (3.4%) than in urban counties (10.5%). Rates were lower in Black and Hispanic children than in White and Asian children.
According to the CDC, high rates of poverty in Black and Hispanic communities may affect vaccination coverage by affecting caregivers’ access to vaccination sites or ability to leave work to take their child to be vaccinated. Pediatric care providers have repeatedly been identified by parents as a source of trusted vaccine information and a strong provider recommendation is associated with vaccination, but not all families are receiving vaccine advice. In a 2022 Kaiser Family Foundation survey, parents of young children with annual household incomes above $90,000 were more likely to talk to their pediatrician about a COVID-19 vaccine than families with lower incomes.3Vaccine hesitancy, fueled by general confusion and skepticism, is another factor contributing to low vaccination rates. Admittedly, the recommendations are complex and on March 14, 2023, the FDA again revised the emergency-use authorization for young children. Some caregivers continue to express concerns about vaccine side effects as well as the belief that the vaccine won’t prevent their child from getting sick.
Kendall Purcell, MD, a pediatrician with Norton Children’s Medical Group in Louisville, Ky., recommends COVID-19 vaccination for her patients because it reduces the risk of severe disease. That factored into her own decision to vaccinate her 4-year-old son and 1-year-old daughter, but she hasn’t been able to convince the parents of all her patients. “Some feel that COVID-19 is not as severe for children, so the risks don’t outweigh the benefits when it comes to vaccinating their children.” Back to our case: In the ED the intern reviewed the laboratory testing she had ordered. She then sat down with the mother of the 3-year-old girl to discuss the diagnosis: febrile seizure associated with COVID-19 infection. Febrile seizures are a well-recognized but uncommon complication of COVID-19 in children. In a retrospective cohort study using electronic health record data, febrile seizures occurred in 0.5% of 8,854 children aged 0-5 years with COVID-19 infection.4 About 9% of these children required critical care services. In another cohort of hospitalized children, neurologic complications occurred in 7% of children hospitalized with COVID-19.5 Febrile and nonfebrile seizures were most commonly observed.
“I really thought COVID-19 was no big deal in young kids,” the mom said. “Parents need the facts.”
The facts are these: Through Dec. 2, 2022, more than 3 million cases of COVID-19 have been reported in children aged younger than 5 years. While COVID is generally less severe in young children than older adults, it is difficult to predict which children will become seriously ill. When children are hospitalized, one in four requires intensive care. COVID-19 is now a vaccine-preventable disease, but too many children remain unprotected.
Dr. Bryant is a pediatrician specializing in infectious diseases at the University of Louisville (Ky.) and Norton Children’s Hospital, also in Louisville. She is a member of the AAP’s Committee on Infectious Diseases and one of the lead authors of the AAP’s Recommendations for Prevention and Control of Influenza in Children, 2022-2023. The opinions expressed in this article are her own. Dr. Bryant discloses that she has served as an investigator on clinical trials funded by Pfizer, Enanta, and Gilead. Email her at [email protected]. Ms. Ezell is a recent graduate from Indiana University Southeast with a Bachelor of Arts in English. They have no conflicts of interest.
References
1. Fleming-Dutra KE et al. Morb Mortal Wkly Rep. 2023;72:177-182.
2. Murthy BP et al. Morb Mortal Wkly Rep. 2023;72:183-9.
3. Lopes L et al. KFF COVID-19 vaccine monitor: July 2022. San Francisco: Kaiser Family Foundation, 2022.
4. Cadet K et al. J Child Neurol. 2022 Apr;37(5):410-5.
5. Antoon JW et al. Pediatrics. 2022 Nov 1;150(5):e2022058167.
Case: A 3-year-old girl presented to the emergency department after a brief seizure at home. She looked well on physical exam except for a fever of 103° F and thick rhinorrhea.
The intern on duty methodically worked through the standard list of questions. “Immunizations up to date?” she asked.
“Absolutely,” the child’s mom responded. “She’s had everything that’s recommended.”
“Including COVID-19 vaccine?” the intern prompted.
“No.” The mom responded with a shake of her head. “We don’t do that vaccine.”
That mom is not alone.
COVID-19 vaccines for children as young as 6 months were given emergency-use authorization by the Food and Drug Administration in June 2022 and in February 2023, the Advisory Committee on Immunization Practices included COVID-19 vaccine on the routine childhood immunization schedule.
COVID-19 vaccines are safe in young children, and they prevent the most severe outcomes associated with infection, including hospitalization. Newly released data confirm that the COVID-19 vaccines produced by Moderna and Pfizer also provide protection against symptomatic infection for at least 4 months after completion of the monovalent primary series.
In a Morbidity and Mortality Weekly Report released on Feb. 17, 2023, the Centers for Disease Control and Prevention reported the results of a test-negative design case-control study that enrolled symptomatic children tested for SARS-CoV-2 infection through Feb. 5, 2023, as part of the Increasing Community Access to Testing (ICATT) program.1 ICATT provides SARS-CoV-2 testing to persons aged at least 3 years at pharmacy and community-based testing sites nationwide.
Two doses of monovalent Moderna vaccine (complete primary series) was 60% effective against symptomatic infection (95% confidence interval, 49%-68%) 2 weeks to 2 months after receipt of the second dose. Vaccine effectiveness dropped to 36% (95% CI, 15%-52%) 3-4 months after the second dose. Three doses of monovalent Pfizer-BioNTech vaccine (complete primary series) was 31% effective (95% CI, 7%-49%) at preventing symptomatic infection 2 weeks to 4 months after receipt of the third dose. A bivalent vaccine dose for eligible children is expected to provide more protection against currently circulating SARS-CoV-2 variants.
Despite evidence of vaccine efficacy, very few parents are opting to protect their young children with the COVID-19 vaccine. The CDC reports that, as of March 1, 2023, only 8% of children under 2 years and 10.5% of children aged 2-4 years have initiated a COVID vaccine series. The American Academy of Pediatrics has emphasized that 15.0 million children between the ages of 6 months and 4 years have not yet received their first COVID-19 vaccine dose.
While the reasons underlying low COVID-19 vaccination rates in young children are complex, themes emerge. Socioeconomic disparities contributing to low vaccination rates in young children were highlighted in another recent MMWR article.2 Through Dec. 1, 2022, vaccination coverage was lower in rural counties (3.4%) than in urban counties (10.5%). Rates were lower in Black and Hispanic children than in White and Asian children.
According to the CDC, high rates of poverty in Black and Hispanic communities may affect vaccination coverage by affecting caregivers’ access to vaccination sites or ability to leave work to take their child to be vaccinated. Pediatric care providers have repeatedly been identified by parents as a source of trusted vaccine information and a strong provider recommendation is associated with vaccination, but not all families are receiving vaccine advice. In a 2022 Kaiser Family Foundation survey, parents of young children with annual household incomes above $90,000 were more likely to talk to their pediatrician about a COVID-19 vaccine than families with lower incomes.3Vaccine hesitancy, fueled by general confusion and skepticism, is another factor contributing to low vaccination rates. Admittedly, the recommendations are complex and on March 14, 2023, the FDA again revised the emergency-use authorization for young children. Some caregivers continue to express concerns about vaccine side effects as well as the belief that the vaccine won’t prevent their child from getting sick.
Kendall Purcell, MD, a pediatrician with Norton Children’s Medical Group in Louisville, Ky., recommends COVID-19 vaccination for her patients because it reduces the risk of severe disease. That factored into her own decision to vaccinate her 4-year-old son and 1-year-old daughter, but she hasn’t been able to convince the parents of all her patients. “Some feel that COVID-19 is not as severe for children, so the risks don’t outweigh the benefits when it comes to vaccinating their children.” Back to our case: In the ED the intern reviewed the laboratory testing she had ordered. She then sat down with the mother of the 3-year-old girl to discuss the diagnosis: febrile seizure associated with COVID-19 infection. Febrile seizures are a well-recognized but uncommon complication of COVID-19 in children. In a retrospective cohort study using electronic health record data, febrile seizures occurred in 0.5% of 8,854 children aged 0-5 years with COVID-19 infection.4 About 9% of these children required critical care services. In another cohort of hospitalized children, neurologic complications occurred in 7% of children hospitalized with COVID-19.5 Febrile and nonfebrile seizures were most commonly observed.
“I really thought COVID-19 was no big deal in young kids,” the mom said. “Parents need the facts.”
The facts are these: Through Dec. 2, 2022, more than 3 million cases of COVID-19 have been reported in children aged younger than 5 years. While COVID is generally less severe in young children than older adults, it is difficult to predict which children will become seriously ill. When children are hospitalized, one in four requires intensive care. COVID-19 is now a vaccine-preventable disease, but too many children remain unprotected.
Dr. Bryant is a pediatrician specializing in infectious diseases at the University of Louisville (Ky.) and Norton Children’s Hospital, also in Louisville. She is a member of the AAP’s Committee on Infectious Diseases and one of the lead authors of the AAP’s Recommendations for Prevention and Control of Influenza in Children, 2022-2023. The opinions expressed in this article are her own. Dr. Bryant discloses that she has served as an investigator on clinical trials funded by Pfizer, Enanta, and Gilead. Email her at [email protected]. Ms. Ezell is a recent graduate from Indiana University Southeast with a Bachelor of Arts in English. They have no conflicts of interest.
References
1. Fleming-Dutra KE et al. Morb Mortal Wkly Rep. 2023;72:177-182.
2. Murthy BP et al. Morb Mortal Wkly Rep. 2023;72:183-9.
3. Lopes L et al. KFF COVID-19 vaccine monitor: July 2022. San Francisco: Kaiser Family Foundation, 2022.
4. Cadet K et al. J Child Neurol. 2022 Apr;37(5):410-5.
5. Antoon JW et al. Pediatrics. 2022 Nov 1;150(5):e2022058167.
Case: A 3-year-old girl presented to the emergency department after a brief seizure at home. She looked well on physical exam except for a fever of 103° F and thick rhinorrhea.
The intern on duty methodically worked through the standard list of questions. “Immunizations up to date?” she asked.
“Absolutely,” the child’s mom responded. “She’s had everything that’s recommended.”
“Including COVID-19 vaccine?” the intern prompted.
“No.” The mom responded with a shake of her head. “We don’t do that vaccine.”
That mom is not alone.
COVID-19 vaccines for children as young as 6 months were given emergency-use authorization by the Food and Drug Administration in June 2022 and in February 2023, the Advisory Committee on Immunization Practices included COVID-19 vaccine on the routine childhood immunization schedule.
COVID-19 vaccines are safe in young children, and they prevent the most severe outcomes associated with infection, including hospitalization. Newly released data confirm that the COVID-19 vaccines produced by Moderna and Pfizer also provide protection against symptomatic infection for at least 4 months after completion of the monovalent primary series.
In a Morbidity and Mortality Weekly Report released on Feb. 17, 2023, the Centers for Disease Control and Prevention reported the results of a test-negative design case-control study that enrolled symptomatic children tested for SARS-CoV-2 infection through Feb. 5, 2023, as part of the Increasing Community Access to Testing (ICATT) program.1 ICATT provides SARS-CoV-2 testing to persons aged at least 3 years at pharmacy and community-based testing sites nationwide.
Two doses of monovalent Moderna vaccine (complete primary series) was 60% effective against symptomatic infection (95% confidence interval, 49%-68%) 2 weeks to 2 months after receipt of the second dose. Vaccine effectiveness dropped to 36% (95% CI, 15%-52%) 3-4 months after the second dose. Three doses of monovalent Pfizer-BioNTech vaccine (complete primary series) was 31% effective (95% CI, 7%-49%) at preventing symptomatic infection 2 weeks to 4 months after receipt of the third dose. A bivalent vaccine dose for eligible children is expected to provide more protection against currently circulating SARS-CoV-2 variants.
Despite evidence of vaccine efficacy, very few parents are opting to protect their young children with the COVID-19 vaccine. The CDC reports that, as of March 1, 2023, only 8% of children under 2 years and 10.5% of children aged 2-4 years have initiated a COVID vaccine series. The American Academy of Pediatrics has emphasized that 15.0 million children between the ages of 6 months and 4 years have not yet received their first COVID-19 vaccine dose.
While the reasons underlying low COVID-19 vaccination rates in young children are complex, themes emerge. Socioeconomic disparities contributing to low vaccination rates in young children were highlighted in another recent MMWR article.2 Through Dec. 1, 2022, vaccination coverage was lower in rural counties (3.4%) than in urban counties (10.5%). Rates were lower in Black and Hispanic children than in White and Asian children.
According to the CDC, high rates of poverty in Black and Hispanic communities may affect vaccination coverage by affecting caregivers’ access to vaccination sites or ability to leave work to take their child to be vaccinated. Pediatric care providers have repeatedly been identified by parents as a source of trusted vaccine information and a strong provider recommendation is associated with vaccination, but not all families are receiving vaccine advice. In a 2022 Kaiser Family Foundation survey, parents of young children with annual household incomes above $90,000 were more likely to talk to their pediatrician about a COVID-19 vaccine than families with lower incomes.3Vaccine hesitancy, fueled by general confusion and skepticism, is another factor contributing to low vaccination rates. Admittedly, the recommendations are complex and on March 14, 2023, the FDA again revised the emergency-use authorization for young children. Some caregivers continue to express concerns about vaccine side effects as well as the belief that the vaccine won’t prevent their child from getting sick.
Kendall Purcell, MD, a pediatrician with Norton Children’s Medical Group in Louisville, Ky., recommends COVID-19 vaccination for her patients because it reduces the risk of severe disease. That factored into her own decision to vaccinate her 4-year-old son and 1-year-old daughter, but she hasn’t been able to convince the parents of all her patients. “Some feel that COVID-19 is not as severe for children, so the risks don’t outweigh the benefits when it comes to vaccinating their children.” Back to our case: In the ED the intern reviewed the laboratory testing she had ordered. She then sat down with the mother of the 3-year-old girl to discuss the diagnosis: febrile seizure associated with COVID-19 infection. Febrile seizures are a well-recognized but uncommon complication of COVID-19 in children. In a retrospective cohort study using electronic health record data, febrile seizures occurred in 0.5% of 8,854 children aged 0-5 years with COVID-19 infection.4 About 9% of these children required critical care services. In another cohort of hospitalized children, neurologic complications occurred in 7% of children hospitalized with COVID-19.5 Febrile and nonfebrile seizures were most commonly observed.
“I really thought COVID-19 was no big deal in young kids,” the mom said. “Parents need the facts.”
The facts are these: Through Dec. 2, 2022, more than 3 million cases of COVID-19 have been reported in children aged younger than 5 years. While COVID is generally less severe in young children than older adults, it is difficult to predict which children will become seriously ill. When children are hospitalized, one in four requires intensive care. COVID-19 is now a vaccine-preventable disease, but too many children remain unprotected.
Dr. Bryant is a pediatrician specializing in infectious diseases at the University of Louisville (Ky.) and Norton Children’s Hospital, also in Louisville. She is a member of the AAP’s Committee on Infectious Diseases and one of the lead authors of the AAP’s Recommendations for Prevention and Control of Influenza in Children, 2022-2023. The opinions expressed in this article are her own. Dr. Bryant discloses that she has served as an investigator on clinical trials funded by Pfizer, Enanta, and Gilead. Email her at [email protected]. Ms. Ezell is a recent graduate from Indiana University Southeast with a Bachelor of Arts in English. They have no conflicts of interest.
References
1. Fleming-Dutra KE et al. Morb Mortal Wkly Rep. 2023;72:177-182.
2. Murthy BP et al. Morb Mortal Wkly Rep. 2023;72:183-9.
3. Lopes L et al. KFF COVID-19 vaccine monitor: July 2022. San Francisco: Kaiser Family Foundation, 2022.
4. Cadet K et al. J Child Neurol. 2022 Apr;37(5):410-5.
5. Antoon JW et al. Pediatrics. 2022 Nov 1;150(5):e2022058167.
Older men more at risk as dangerous falls rise for all seniors
When Senate Minority Leader Mitch McConnell (R-Ky.) fell recently at a dinner event in Washington, he unfortunately joined a large group of his senior citizen peers.
This wasn’t the first tumble the 81-year-old has taken. In 2019, he fell in his home, fracturing his shoulder. This time, he got a concussion and was recently released to an in-patient rehabilitation facility. While Sen. McConnell didn’t fracture his skull, in falling and hitting his head, he became part of an emerging statistic: One that reveals falls are more dangerous for senior men than senior women.
This new research, which appeared in the American Journal of Emergency Medicine, came as a surprise to lead researcher Scott Alter, MD, associate professor of emergency medicine at the Florida Atlantic University, Boca Raton.
“We always hear about lower bone density rates among females, so we didn’t expect to see males with more skull fractures,” he said.
Dr. Alter said that as a clinician in a southern Florida facility, his emergency department was the perfect study grounds to evaluate incoming geriatric patients due to falls. Older “patients are at higher risk of skull fractures and intercranial bleeding, and we wanted to look at any patient presenting with a head injury. Some 80% were fall related, however.”
The statistics bear out the fact that falls of all types are common among the elderly: Some 800,000 seniors wind up in the hospital each year because of falls.
The numbers show death rates from falls are on the rise in the senior citizen age group, too, up 30% from 2007 to 2016. Falls account for 70% of accidental deaths in people 75 and older. They are the leading cause of injury-related visits to emergency departments in the country, too.
Jennifer Stevens, MD, a gerontologist and executive director at Florida-based Abbey Delray South, is aware of the dire numbers and sees their consequences regularly. “The reasons seniors are at a high fall risk are many,” she said. “They include balance issues, declining strength, diseases like Parkinson’s and Alzheimer’s, side effects of their medications, and more.”
In addition, many seniors live in spaces that are not necessarily equipped for their limitations, and hazards exist all over their homes. Put together, and the risks for falls are everywhere. But there are steps seniors, their families, and even middle-aged people can take to mitigate and hopefully prevent dangerous falls.
Starting early
While in many cases the journey to lessen fall risks begins after a fall, the time to begin addressing the issue is long before you hit your senior years. Mary Therese Cole, a physical therapist and certified dementia practitioner at Manual Edge Physical Therapy in Colorado Springs, Colo., says that age 50 is a good time to start paying attention and addressing physical declines.
“This is an age where your vision might begin deteriorating,” she said. “It’s a big reason why elderly people trip and fall.”
As our brains begin to age in our middle years, the neural pathways from brain to extremities start to decline, too. The result is that many people stop picking up their feet as well as they used to do, making them more likely to trip.
“You’re not elderly yet, but you’re not a spring chicken, either,” Ms. Cole said. “Any issues you have now will only get worse if you’re not working on them.”
A good starting point in middle age, then, is to work on both strength training and balance exercises. A certified personal trainer or physical therapist can help get you on a program to ward off many of these declines.
If you’ve reached your later years, however, and are experiencing physical declines, it’s smart to check in with your primary care doctor for an assessment. “He or she can get your started on regular PT to evaluate any shortcomings and then address them,” Ms. Cole said.
She noted that when she’s working with senior patients, she’ll test their strength getting into and out of a chair, do a manual strength test to check on lower extremities, check their walking stride, and ask about conditions such as diabetes, former surgeries, and other conditions.
From there, Ms. Cole said she can write up a plan for the patient. Likewise, Dr. Stevens uses a program called Be Active that allows her to test seniors on a variety of measurements, including flexibility, balance, hand strength, and more.
“Then we match them with classes to address their shortcomings,” she said. “It’s critical that seniors have the ability to recover and not fall if they get knocked off balance.”
Beyond working on your physical limitations, taking a good look at your home is essential, too. “You can have an occupational therapist come to your home and do an evaluation,” Dr. Stevens said. “They can help you rearrange and reorganize for a safer environment.”
Big, common household fall hazards include throw rugs, lack of nightlights for middle-of-the-night visits to the bathroom, a lack of grab bars in the shower/bathtub, and furniture that blocks pathways.
For his part, Dr. Alter likes to point seniors and their doctors to the CDC’s STEADI program, which is aimed at stopping elderly accidents, deaths, and injuries.
“It includes screening for fall risk, assessing factors you can modify or improve, and more tools,” he said.
Dr. Alter also recommended seniors talk to their doctors about medications, particularly blood thinners.
“At a certain point, you need to weigh the benefits of disease prevention with the risk of injury if you fall,” he said. “The bleeding risk might be too high if the patient is at a high risk of falls.”
A version of this article originally appeared on WebMD.com.
When Senate Minority Leader Mitch McConnell (R-Ky.) fell recently at a dinner event in Washington, he unfortunately joined a large group of his senior citizen peers.
This wasn’t the first tumble the 81-year-old has taken. In 2019, he fell in his home, fracturing his shoulder. This time, he got a concussion and was recently released to an in-patient rehabilitation facility. While Sen. McConnell didn’t fracture his skull, in falling and hitting his head, he became part of an emerging statistic: One that reveals falls are more dangerous for senior men than senior women.
This new research, which appeared in the American Journal of Emergency Medicine, came as a surprise to lead researcher Scott Alter, MD, associate professor of emergency medicine at the Florida Atlantic University, Boca Raton.
“We always hear about lower bone density rates among females, so we didn’t expect to see males with more skull fractures,” he said.
Dr. Alter said that as a clinician in a southern Florida facility, his emergency department was the perfect study grounds to evaluate incoming geriatric patients due to falls. Older “patients are at higher risk of skull fractures and intercranial bleeding, and we wanted to look at any patient presenting with a head injury. Some 80% were fall related, however.”
The statistics bear out the fact that falls of all types are common among the elderly: Some 800,000 seniors wind up in the hospital each year because of falls.
The numbers show death rates from falls are on the rise in the senior citizen age group, too, up 30% from 2007 to 2016. Falls account for 70% of accidental deaths in people 75 and older. They are the leading cause of injury-related visits to emergency departments in the country, too.
Jennifer Stevens, MD, a gerontologist and executive director at Florida-based Abbey Delray South, is aware of the dire numbers and sees their consequences regularly. “The reasons seniors are at a high fall risk are many,” she said. “They include balance issues, declining strength, diseases like Parkinson’s and Alzheimer’s, side effects of their medications, and more.”
In addition, many seniors live in spaces that are not necessarily equipped for their limitations, and hazards exist all over their homes. Put together, and the risks for falls are everywhere. But there are steps seniors, their families, and even middle-aged people can take to mitigate and hopefully prevent dangerous falls.
Starting early
While in many cases the journey to lessen fall risks begins after a fall, the time to begin addressing the issue is long before you hit your senior years. Mary Therese Cole, a physical therapist and certified dementia practitioner at Manual Edge Physical Therapy in Colorado Springs, Colo., says that age 50 is a good time to start paying attention and addressing physical declines.
“This is an age where your vision might begin deteriorating,” she said. “It’s a big reason why elderly people trip and fall.”
As our brains begin to age in our middle years, the neural pathways from brain to extremities start to decline, too. The result is that many people stop picking up their feet as well as they used to do, making them more likely to trip.
“You’re not elderly yet, but you’re not a spring chicken, either,” Ms. Cole said. “Any issues you have now will only get worse if you’re not working on them.”
A good starting point in middle age, then, is to work on both strength training and balance exercises. A certified personal trainer or physical therapist can help get you on a program to ward off many of these declines.
If you’ve reached your later years, however, and are experiencing physical declines, it’s smart to check in with your primary care doctor for an assessment. “He or she can get your started on regular PT to evaluate any shortcomings and then address them,” Ms. Cole said.
She noted that when she’s working with senior patients, she’ll test their strength getting into and out of a chair, do a manual strength test to check on lower extremities, check their walking stride, and ask about conditions such as diabetes, former surgeries, and other conditions.
From there, Ms. Cole said she can write up a plan for the patient. Likewise, Dr. Stevens uses a program called Be Active that allows her to test seniors on a variety of measurements, including flexibility, balance, hand strength, and more.
“Then we match them with classes to address their shortcomings,” she said. “It’s critical that seniors have the ability to recover and not fall if they get knocked off balance.”
Beyond working on your physical limitations, taking a good look at your home is essential, too. “You can have an occupational therapist come to your home and do an evaluation,” Dr. Stevens said. “They can help you rearrange and reorganize for a safer environment.”
Big, common household fall hazards include throw rugs, lack of nightlights for middle-of-the-night visits to the bathroom, a lack of grab bars in the shower/bathtub, and furniture that blocks pathways.
For his part, Dr. Alter likes to point seniors and their doctors to the CDC’s STEADI program, which is aimed at stopping elderly accidents, deaths, and injuries.
“It includes screening for fall risk, assessing factors you can modify or improve, and more tools,” he said.
Dr. Alter also recommended seniors talk to their doctors about medications, particularly blood thinners.
“At a certain point, you need to weigh the benefits of disease prevention with the risk of injury if you fall,” he said. “The bleeding risk might be too high if the patient is at a high risk of falls.”
A version of this article originally appeared on WebMD.com.
When Senate Minority Leader Mitch McConnell (R-Ky.) fell recently at a dinner event in Washington, he unfortunately joined a large group of his senior citizen peers.
This wasn’t the first tumble the 81-year-old has taken. In 2019, he fell in his home, fracturing his shoulder. This time, he got a concussion and was recently released to an in-patient rehabilitation facility. While Sen. McConnell didn’t fracture his skull, in falling and hitting his head, he became part of an emerging statistic: One that reveals falls are more dangerous for senior men than senior women.
This new research, which appeared in the American Journal of Emergency Medicine, came as a surprise to lead researcher Scott Alter, MD, associate professor of emergency medicine at the Florida Atlantic University, Boca Raton.
“We always hear about lower bone density rates among females, so we didn’t expect to see males with more skull fractures,” he said.
Dr. Alter said that as a clinician in a southern Florida facility, his emergency department was the perfect study grounds to evaluate incoming geriatric patients due to falls. Older “patients are at higher risk of skull fractures and intercranial bleeding, and we wanted to look at any patient presenting with a head injury. Some 80% were fall related, however.”
The statistics bear out the fact that falls of all types are common among the elderly: Some 800,000 seniors wind up in the hospital each year because of falls.
The numbers show death rates from falls are on the rise in the senior citizen age group, too, up 30% from 2007 to 2016. Falls account for 70% of accidental deaths in people 75 and older. They are the leading cause of injury-related visits to emergency departments in the country, too.
Jennifer Stevens, MD, a gerontologist and executive director at Florida-based Abbey Delray South, is aware of the dire numbers and sees their consequences regularly. “The reasons seniors are at a high fall risk are many,” she said. “They include balance issues, declining strength, diseases like Parkinson’s and Alzheimer’s, side effects of their medications, and more.”
In addition, many seniors live in spaces that are not necessarily equipped for their limitations, and hazards exist all over their homes. Put together, and the risks for falls are everywhere. But there are steps seniors, their families, and even middle-aged people can take to mitigate and hopefully prevent dangerous falls.
Starting early
While in many cases the journey to lessen fall risks begins after a fall, the time to begin addressing the issue is long before you hit your senior years. Mary Therese Cole, a physical therapist and certified dementia practitioner at Manual Edge Physical Therapy in Colorado Springs, Colo., says that age 50 is a good time to start paying attention and addressing physical declines.
“This is an age where your vision might begin deteriorating,” she said. “It’s a big reason why elderly people trip and fall.”
As our brains begin to age in our middle years, the neural pathways from brain to extremities start to decline, too. The result is that many people stop picking up their feet as well as they used to do, making them more likely to trip.
“You’re not elderly yet, but you’re not a spring chicken, either,” Ms. Cole said. “Any issues you have now will only get worse if you’re not working on them.”
A good starting point in middle age, then, is to work on both strength training and balance exercises. A certified personal trainer or physical therapist can help get you on a program to ward off many of these declines.
If you’ve reached your later years, however, and are experiencing physical declines, it’s smart to check in with your primary care doctor for an assessment. “He or she can get your started on regular PT to evaluate any shortcomings and then address them,” Ms. Cole said.
She noted that when she’s working with senior patients, she’ll test their strength getting into and out of a chair, do a manual strength test to check on lower extremities, check their walking stride, and ask about conditions such as diabetes, former surgeries, and other conditions.
From there, Ms. Cole said she can write up a plan for the patient. Likewise, Dr. Stevens uses a program called Be Active that allows her to test seniors on a variety of measurements, including flexibility, balance, hand strength, and more.
“Then we match them with classes to address their shortcomings,” she said. “It’s critical that seniors have the ability to recover and not fall if they get knocked off balance.”
Beyond working on your physical limitations, taking a good look at your home is essential, too. “You can have an occupational therapist come to your home and do an evaluation,” Dr. Stevens said. “They can help you rearrange and reorganize for a safer environment.”
Big, common household fall hazards include throw rugs, lack of nightlights for middle-of-the-night visits to the bathroom, a lack of grab bars in the shower/bathtub, and furniture that blocks pathways.
For his part, Dr. Alter likes to point seniors and their doctors to the CDC’s STEADI program, which is aimed at stopping elderly accidents, deaths, and injuries.
“It includes screening for fall risk, assessing factors you can modify or improve, and more tools,” he said.
Dr. Alter also recommended seniors talk to their doctors about medications, particularly blood thinners.
“At a certain point, you need to weigh the benefits of disease prevention with the risk of injury if you fall,” he said. “The bleeding risk might be too high if the patient is at a high risk of falls.”
A version of this article originally appeared on WebMD.com.
The human-looking robot therapist will coach your well-being now
Do android therapists dream of electric employees?
Robots. It can be tough to remember that, when they’re not dooming humanity to apocalypse or just telling you that you’re doomed, robots have real-world uses. There are actual robots in the world, and they can do things beyond bend girders, sing about science, or run the navy.
Look, we’ll stop with the pop-culture references when pop culture runs out of robots to reference. It may take a while.
Robots are indelibly rooted in the public consciousness, and that plays into our expectations when we encounter a real-life robot. This leads us into a recent study conducted by researchers at the University of Cambridge, who developed a robot-led mental well-being program that a tech company utilized for 4 weeks. Why choose a robot? Well, why spring for a qualified therapist who requires a salary when you could simply get a robot to do the job for free? Get with the capitalist agenda here. Surely it won’t backfire.
The 26 people enrolled in the study received coaching from one of two robots, both programmed identically to act like mental health coaches, based on interviews with human therapists. Both acted identically and had identical expressions. The only difference between the two was their appearance. QTRobot was nearly a meter tall and looked like a human child; Misty II was much smaller and looked like a toy.
People who received coaching from Misty II were better able to connect and had a better experience than those who received coaching from QTRobot. According to those in the QTRobot group, their expectations didn’t match reality. The robots are good coaches, but they don’t act human. This wasn’t a problem for Misty II, since it doesn’t look human, but for QTRobot, the participants were expecting “to hell with our orders,” but received “Daisy, Daisy, give me your answer do.” When you’ve been programmed to think of robots as metal humans, it can be off-putting to see them act as, well, robots.
That said, all participants found the exercises helpful and were open to receiving more robot-led therapy in the future. And while we’re sure the technology will advance to make robot therapists more empathetic and more human, hopefully scientists won’t go too far. We don’t need depressed robots.
Birthing experience is all in the mindset
Alexa, play Peer Gynt Suite No. 1, Op. 46 - I. Morning Mood.
Birth.
Giving birth is a common experience for many, if not most, female mammals, but wanting it to be a pleasurable one seems distinctly human. There are many methods and practices that may make giving birth an easier and enjoyable experience for the mother, but a new study suggests that the key could be in her mind.
The mindset of the expectant mother during pregnancy, it seems, has some effect on how smooth or intervention-filled delivery is. If the mothers saw their experience as a natural process, they were less likely to need pain medication or a C-section, but mothers who viewed the experience as more of a “medical procedure” were more likely to require more medical supervision and intervention, according to investigators from the University of Bonn (Germany).
Now, the researchers wanted to be super clear in saying that there’s no right or wrong mindset to have. They just focused on the outcomes of those mindsets and whether they actually do have some effect on occurrences.
Apparently, yes.
“Mindsets can be understood as a kind of mental lense that guide our perception of the world around us and can influence our behavior,” Dr. Lisa Hoffmann said in a statement from the university. “The study highlights the importance of psychological factors in childbirth.”
The researchers surveyed 300 women with an online tool before and after delivery and found the effects of the natural process mindset lingered even after giving birth. They had lower rates of depression and posttraumatic stress, which may have a snowballing effect on mother-child bonding after childbirth.
Preparation for the big day, then, should be about more than gathering diapers and shopping for car seats. Women should prepare their minds as well. If it’s going to make giving birth better, why not?
Becoming a parent is going to create a psychological shift, no matter how you slice it.
Giant inflatable colon reported in Utah
Do not be alarmed! Yes, there is a giant inflatable colon currently at large in the Beehive State, but it will not harm you. The giant inflatable colon is in Utah as part of Intermountain Health’s “Let’s get to the bottom of colon cancer tour” and he only wants to help you.
The giant inflatable colon, whose name happens to be Collin, is 12 feet long and weighs 113 pounds. March is Colon Cancer Awareness Month, so Collin is traveling around Utah and Idaho to raise awareness about colon cancer and the various screening options. He is not going to change local weather patterns, eat small children, or take over local governments and raise your taxes.
Instead, Collin is planning to display “portions of a healthy colon, polyps or bumps on the colon, malignant polyps which look more vascular and have more redness, cancerous cells, advanced cancer cells, and Crohn’s disease,” KSL.com said.
Collin the colon is on loan to Intermountain Health from medical device manufacturer Boston Scientific and will be traveling to Spanish Fork, Provo, and Ogden, among other locations in Utah, as well as Burley and Meridian, Idaho, in the coming days.
Collin the colon’s participation in the tour has created some serious buzz in the Colin/Collin community:
- Colin Powell (four-star general and Secretary of State): “Back then, the second-most important topic among the Joint Chiefs of Staff was colon cancer screening. And the Navy guy – I can’t remember his name – was a huge fan of giant inflatable organs.”
- Colin Jost (comedian and Saturday Night Live “Weekend Update” cohost): “He’s funnier than Tucker Carlson and Pete Davidson combined.”
Do android therapists dream of electric employees?
Robots. It can be tough to remember that, when they’re not dooming humanity to apocalypse or just telling you that you’re doomed, robots have real-world uses. There are actual robots in the world, and they can do things beyond bend girders, sing about science, or run the navy.
Look, we’ll stop with the pop-culture references when pop culture runs out of robots to reference. It may take a while.
Robots are indelibly rooted in the public consciousness, and that plays into our expectations when we encounter a real-life robot. This leads us into a recent study conducted by researchers at the University of Cambridge, who developed a robot-led mental well-being program that a tech company utilized for 4 weeks. Why choose a robot? Well, why spring for a qualified therapist who requires a salary when you could simply get a robot to do the job for free? Get with the capitalist agenda here. Surely it won’t backfire.
The 26 people enrolled in the study received coaching from one of two robots, both programmed identically to act like mental health coaches, based on interviews with human therapists. Both acted identically and had identical expressions. The only difference between the two was their appearance. QTRobot was nearly a meter tall and looked like a human child; Misty II was much smaller and looked like a toy.
People who received coaching from Misty II were better able to connect and had a better experience than those who received coaching from QTRobot. According to those in the QTRobot group, their expectations didn’t match reality. The robots are good coaches, but they don’t act human. This wasn’t a problem for Misty II, since it doesn’t look human, but for QTRobot, the participants were expecting “to hell with our orders,” but received “Daisy, Daisy, give me your answer do.” When you’ve been programmed to think of robots as metal humans, it can be off-putting to see them act as, well, robots.
That said, all participants found the exercises helpful and were open to receiving more robot-led therapy in the future. And while we’re sure the technology will advance to make robot therapists more empathetic and more human, hopefully scientists won’t go too far. We don’t need depressed robots.
Birthing experience is all in the mindset
Alexa, play Peer Gynt Suite No. 1, Op. 46 - I. Morning Mood.
Birth.
Giving birth is a common experience for many, if not most, female mammals, but wanting it to be a pleasurable one seems distinctly human. There are many methods and practices that may make giving birth an easier and enjoyable experience for the mother, but a new study suggests that the key could be in her mind.
The mindset of the expectant mother during pregnancy, it seems, has some effect on how smooth or intervention-filled delivery is. If the mothers saw their experience as a natural process, they were less likely to need pain medication or a C-section, but mothers who viewed the experience as more of a “medical procedure” were more likely to require more medical supervision and intervention, according to investigators from the University of Bonn (Germany).
Now, the researchers wanted to be super clear in saying that there’s no right or wrong mindset to have. They just focused on the outcomes of those mindsets and whether they actually do have some effect on occurrences.
Apparently, yes.
“Mindsets can be understood as a kind of mental lense that guide our perception of the world around us and can influence our behavior,” Dr. Lisa Hoffmann said in a statement from the university. “The study highlights the importance of psychological factors in childbirth.”
The researchers surveyed 300 women with an online tool before and after delivery and found the effects of the natural process mindset lingered even after giving birth. They had lower rates of depression and posttraumatic stress, which may have a snowballing effect on mother-child bonding after childbirth.
Preparation for the big day, then, should be about more than gathering diapers and shopping for car seats. Women should prepare their minds as well. If it’s going to make giving birth better, why not?
Becoming a parent is going to create a psychological shift, no matter how you slice it.
Giant inflatable colon reported in Utah
Do not be alarmed! Yes, there is a giant inflatable colon currently at large in the Beehive State, but it will not harm you. The giant inflatable colon is in Utah as part of Intermountain Health’s “Let’s get to the bottom of colon cancer tour” and he only wants to help you.
The giant inflatable colon, whose name happens to be Collin, is 12 feet long and weighs 113 pounds. March is Colon Cancer Awareness Month, so Collin is traveling around Utah and Idaho to raise awareness about colon cancer and the various screening options. He is not going to change local weather patterns, eat small children, or take over local governments and raise your taxes.
Instead, Collin is planning to display “portions of a healthy colon, polyps or bumps on the colon, malignant polyps which look more vascular and have more redness, cancerous cells, advanced cancer cells, and Crohn’s disease,” KSL.com said.
Collin the colon is on loan to Intermountain Health from medical device manufacturer Boston Scientific and will be traveling to Spanish Fork, Provo, and Ogden, among other locations in Utah, as well as Burley and Meridian, Idaho, in the coming days.
Collin the colon’s participation in the tour has created some serious buzz in the Colin/Collin community:
- Colin Powell (four-star general and Secretary of State): “Back then, the second-most important topic among the Joint Chiefs of Staff was colon cancer screening. And the Navy guy – I can’t remember his name – was a huge fan of giant inflatable organs.”
- Colin Jost (comedian and Saturday Night Live “Weekend Update” cohost): “He’s funnier than Tucker Carlson and Pete Davidson combined.”
Do android therapists dream of electric employees?
Robots. It can be tough to remember that, when they’re not dooming humanity to apocalypse or just telling you that you’re doomed, robots have real-world uses. There are actual robots in the world, and they can do things beyond bend girders, sing about science, or run the navy.
Look, we’ll stop with the pop-culture references when pop culture runs out of robots to reference. It may take a while.
Robots are indelibly rooted in the public consciousness, and that plays into our expectations when we encounter a real-life robot. This leads us into a recent study conducted by researchers at the University of Cambridge, who developed a robot-led mental well-being program that a tech company utilized for 4 weeks. Why choose a robot? Well, why spring for a qualified therapist who requires a salary when you could simply get a robot to do the job for free? Get with the capitalist agenda here. Surely it won’t backfire.
The 26 people enrolled in the study received coaching from one of two robots, both programmed identically to act like mental health coaches, based on interviews with human therapists. Both acted identically and had identical expressions. The only difference between the two was their appearance. QTRobot was nearly a meter tall and looked like a human child; Misty II was much smaller and looked like a toy.
People who received coaching from Misty II were better able to connect and had a better experience than those who received coaching from QTRobot. According to those in the QTRobot group, their expectations didn’t match reality. The robots are good coaches, but they don’t act human. This wasn’t a problem for Misty II, since it doesn’t look human, but for QTRobot, the participants were expecting “to hell with our orders,” but received “Daisy, Daisy, give me your answer do.” When you’ve been programmed to think of robots as metal humans, it can be off-putting to see them act as, well, robots.
That said, all participants found the exercises helpful and were open to receiving more robot-led therapy in the future. And while we’re sure the technology will advance to make robot therapists more empathetic and more human, hopefully scientists won’t go too far. We don’t need depressed robots.
Birthing experience is all in the mindset
Alexa, play Peer Gynt Suite No. 1, Op. 46 - I. Morning Mood.
Birth.
Giving birth is a common experience for many, if not most, female mammals, but wanting it to be a pleasurable one seems distinctly human. There are many methods and practices that may make giving birth an easier and enjoyable experience for the mother, but a new study suggests that the key could be in her mind.
The mindset of the expectant mother during pregnancy, it seems, has some effect on how smooth or intervention-filled delivery is. If the mothers saw their experience as a natural process, they were less likely to need pain medication or a C-section, but mothers who viewed the experience as more of a “medical procedure” were more likely to require more medical supervision and intervention, according to investigators from the University of Bonn (Germany).
Now, the researchers wanted to be super clear in saying that there’s no right or wrong mindset to have. They just focused on the outcomes of those mindsets and whether they actually do have some effect on occurrences.
Apparently, yes.
“Mindsets can be understood as a kind of mental lense that guide our perception of the world around us and can influence our behavior,” Dr. Lisa Hoffmann said in a statement from the university. “The study highlights the importance of psychological factors in childbirth.”
The researchers surveyed 300 women with an online tool before and after delivery and found the effects of the natural process mindset lingered even after giving birth. They had lower rates of depression and posttraumatic stress, which may have a snowballing effect on mother-child bonding after childbirth.
Preparation for the big day, then, should be about more than gathering diapers and shopping for car seats. Women should prepare their minds as well. If it’s going to make giving birth better, why not?
Becoming a parent is going to create a psychological shift, no matter how you slice it.
Giant inflatable colon reported in Utah
Do not be alarmed! Yes, there is a giant inflatable colon currently at large in the Beehive State, but it will not harm you. The giant inflatable colon is in Utah as part of Intermountain Health’s “Let’s get to the bottom of colon cancer tour” and he only wants to help you.
The giant inflatable colon, whose name happens to be Collin, is 12 feet long and weighs 113 pounds. March is Colon Cancer Awareness Month, so Collin is traveling around Utah and Idaho to raise awareness about colon cancer and the various screening options. He is not going to change local weather patterns, eat small children, or take over local governments and raise your taxes.
Instead, Collin is planning to display “portions of a healthy colon, polyps or bumps on the colon, malignant polyps which look more vascular and have more redness, cancerous cells, advanced cancer cells, and Crohn’s disease,” KSL.com said.
Collin the colon is on loan to Intermountain Health from medical device manufacturer Boston Scientific and will be traveling to Spanish Fork, Provo, and Ogden, among other locations in Utah, as well as Burley and Meridian, Idaho, in the coming days.
Collin the colon’s participation in the tour has created some serious buzz in the Colin/Collin community:
- Colin Powell (four-star general and Secretary of State): “Back then, the second-most important topic among the Joint Chiefs of Staff was colon cancer screening. And the Navy guy – I can’t remember his name – was a huge fan of giant inflatable organs.”
- Colin Jost (comedian and Saturday Night Live “Weekend Update” cohost): “He’s funnier than Tucker Carlson and Pete Davidson combined.”
High school athletes sustaining worse injuries
High school students are injuring themselves more severely even as overall injury rates have declined, according to a new study presented at the annual meeting of the American Academy of Orthopaedic Surgeons.
The study compared injuries from a 4-year period ending in 2019 to data from 2005 and 2006. The overall rate of injuries dropped 9%, from 2.51 injuries per 1,000 athletic games or practices to 2.29 per 1,000; injuries requiring less than 1 week of recovery time fell by 13%. But, the number of head and neck injuries increased by 10%, injuries requiring surgery increased by 1%, and injuries leading to medical disqualification jumped by 11%.
“It’s wonderful that the injury rate is declining,” said Jordan Neoma Pizzarro, a medical student at George Washington University, Washington, who led the study. “But the data does suggest that the injuries that are happening are worse.”
The increases may also reflect increased education and awareness of how to detect concussions and other injuries that need medical attention, said Micah Lissy, MD, MS, an orthopedic surgeon specializing in sports medicine at Michigan State University, East Lansing. Dr. Lissy cautioned against physicians and others taking the data at face value.
“We need to be implementing preventive measures wherever possible, but I think we can also consider that there may be some confounding factors in the data,” Dr. Lissy told this news organization.
Ms. Pizzarro and her team analyzed data collected from athletic trainers at 100 high schools across the country for the ongoing National Health School Sports-Related Injury Surveillance Study.
Athletes participating in sports such as football, soccer, basketball, volleyball, and softball were included in the analysis. Trainers report the number of injuries for every competition and practice, also known as “athletic exposures.”
Boys’ football carried the highest injury rate, with 3.96 injuries per 1,000 AEs, amounting to 44% of all injuries reported. Girls’ soccer and boys’ wrestling followed, with injury rates of 2.65 and 1.56, respectively.
Sprains and strains accounted for 37% of injuries, followed by concussions (21.6%). The head and/or face was the most injured body site, followed by the ankles and/or knees. Most injuries took place during competitions rather than in practices (relative risk, 3.39; 95% confidence interval, 3.28-3.49; P < .05).
Ms. Pizzarro said that an overall increase in intensity, physical contact, and collisions may account for the spike in more severe injuries.
“Kids are encouraged to specialize in one sport early on and stick with it year-round,” she said. “They’re probably becoming more agile and better athletes, but they’re probably also getting more competitive.”
Dr. Lissy, who has worked with high school athletes as a surgeon, physical therapist, athletic trainer, and coach, said that some of the increases in severity of injuries may reflect trends in sports over the past two decades: Student athletes have become stronger and faster and have put on more muscle mass.
“When you have something that’s much larger, moving much faster and with more force, you’re going to have more force when you bump into things,” he said. “This can lead to more significant injuries.”
The study was independently supported. Study authors report no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
High school students are injuring themselves more severely even as overall injury rates have declined, according to a new study presented at the annual meeting of the American Academy of Orthopaedic Surgeons.
The study compared injuries from a 4-year period ending in 2019 to data from 2005 and 2006. The overall rate of injuries dropped 9%, from 2.51 injuries per 1,000 athletic games or practices to 2.29 per 1,000; injuries requiring less than 1 week of recovery time fell by 13%. But, the number of head and neck injuries increased by 10%, injuries requiring surgery increased by 1%, and injuries leading to medical disqualification jumped by 11%.
“It’s wonderful that the injury rate is declining,” said Jordan Neoma Pizzarro, a medical student at George Washington University, Washington, who led the study. “But the data does suggest that the injuries that are happening are worse.”
The increases may also reflect increased education and awareness of how to detect concussions and other injuries that need medical attention, said Micah Lissy, MD, MS, an orthopedic surgeon specializing in sports medicine at Michigan State University, East Lansing. Dr. Lissy cautioned against physicians and others taking the data at face value.
“We need to be implementing preventive measures wherever possible, but I think we can also consider that there may be some confounding factors in the data,” Dr. Lissy told this news organization.
Ms. Pizzarro and her team analyzed data collected from athletic trainers at 100 high schools across the country for the ongoing National Health School Sports-Related Injury Surveillance Study.
Athletes participating in sports such as football, soccer, basketball, volleyball, and softball were included in the analysis. Trainers report the number of injuries for every competition and practice, also known as “athletic exposures.”
Boys’ football carried the highest injury rate, with 3.96 injuries per 1,000 AEs, amounting to 44% of all injuries reported. Girls’ soccer and boys’ wrestling followed, with injury rates of 2.65 and 1.56, respectively.
Sprains and strains accounted for 37% of injuries, followed by concussions (21.6%). The head and/or face was the most injured body site, followed by the ankles and/or knees. Most injuries took place during competitions rather than in practices (relative risk, 3.39; 95% confidence interval, 3.28-3.49; P < .05).
Ms. Pizzarro said that an overall increase in intensity, physical contact, and collisions may account for the spike in more severe injuries.
“Kids are encouraged to specialize in one sport early on and stick with it year-round,” she said. “They’re probably becoming more agile and better athletes, but they’re probably also getting more competitive.”
Dr. Lissy, who has worked with high school athletes as a surgeon, physical therapist, athletic trainer, and coach, said that some of the increases in severity of injuries may reflect trends in sports over the past two decades: Student athletes have become stronger and faster and have put on more muscle mass.
“When you have something that’s much larger, moving much faster and with more force, you’re going to have more force when you bump into things,” he said. “This can lead to more significant injuries.”
The study was independently supported. Study authors report no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
High school students are injuring themselves more severely even as overall injury rates have declined, according to a new study presented at the annual meeting of the American Academy of Orthopaedic Surgeons.
The study compared injuries from a 4-year period ending in 2019 to data from 2005 and 2006. The overall rate of injuries dropped 9%, from 2.51 injuries per 1,000 athletic games or practices to 2.29 per 1,000; injuries requiring less than 1 week of recovery time fell by 13%. But, the number of head and neck injuries increased by 10%, injuries requiring surgery increased by 1%, and injuries leading to medical disqualification jumped by 11%.
“It’s wonderful that the injury rate is declining,” said Jordan Neoma Pizzarro, a medical student at George Washington University, Washington, who led the study. “But the data does suggest that the injuries that are happening are worse.”
The increases may also reflect increased education and awareness of how to detect concussions and other injuries that need medical attention, said Micah Lissy, MD, MS, an orthopedic surgeon specializing in sports medicine at Michigan State University, East Lansing. Dr. Lissy cautioned against physicians and others taking the data at face value.
“We need to be implementing preventive measures wherever possible, but I think we can also consider that there may be some confounding factors in the data,” Dr. Lissy told this news organization.
Ms. Pizzarro and her team analyzed data collected from athletic trainers at 100 high schools across the country for the ongoing National Health School Sports-Related Injury Surveillance Study.
Athletes participating in sports such as football, soccer, basketball, volleyball, and softball were included in the analysis. Trainers report the number of injuries for every competition and practice, also known as “athletic exposures.”
Boys’ football carried the highest injury rate, with 3.96 injuries per 1,000 AEs, amounting to 44% of all injuries reported. Girls’ soccer and boys’ wrestling followed, with injury rates of 2.65 and 1.56, respectively.
Sprains and strains accounted for 37% of injuries, followed by concussions (21.6%). The head and/or face was the most injured body site, followed by the ankles and/or knees. Most injuries took place during competitions rather than in practices (relative risk, 3.39; 95% confidence interval, 3.28-3.49; P < .05).
Ms. Pizzarro said that an overall increase in intensity, physical contact, and collisions may account for the spike in more severe injuries.
“Kids are encouraged to specialize in one sport early on and stick with it year-round,” she said. “They’re probably becoming more agile and better athletes, but they’re probably also getting more competitive.”
Dr. Lissy, who has worked with high school athletes as a surgeon, physical therapist, athletic trainer, and coach, said that some of the increases in severity of injuries may reflect trends in sports over the past two decades: Student athletes have become stronger and faster and have put on more muscle mass.
“When you have something that’s much larger, moving much faster and with more force, you’re going to have more force when you bump into things,” he said. “This can lead to more significant injuries.”
The study was independently supported. Study authors report no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
What do I have? How to tell patients you’re not sure
Physicians often struggle with telling patients when they are unsure about a diagnosis. In the absence of clarity, doctors may fear losing a patient’s trust by appearing unsure.
Yet diagnostic uncertainty is an inevitable part of medicine.
“It’s often uncertain what is really going on. People have lots of unspecific symptoms,” said Gordon D. Schiff, MD, a patient safety researcher at Harvard Medical School and Brigham and Women’s Hospital in Boston.
By one estimate, more than one-third of patients are discharged from an emergency department without a clear diagnosis. Physicians may order more tests to try to resolve uncertainty, but this method is not foolproof and may lead to increased health care costs. Physicians can use an uncertain diagnosis as an opportunity to improve conversations with patients, Dr. Schiff said.
“How do you talk to patients about that? How do you convey that?” Dr. Schiff asked.
To begin to answer these questions, The scenarios included an enlarged lymph node in a patient in remission for lymphoma, which could suggest recurrence of the disease but not necessarily; a patient with a new-onset headache; and another patient with an unexplained fever and a respiratory tract infection.
For each vignette, the researchers also asked patient advocates – many of whom had experienced receiving an incorrect diagnosis – for their thoughts on how the conversation should go.
Almost 70 people were consulted (24 primary care physicians, 40 patients, and five experts in informatics and quality and safety). Dr. Schiff and his colleagues produced six standardized elements that should be part of a conversation whenever a diagnosis is unclear.
- The most likely diagnosis, along with any alternatives if this isn’t certain, with phrases such as, “Sometimes we don’t have the answers, but we will keep trying to figure out what is going on.”
- Next steps – lab tests, return visits, etc.
- Expected time frame for patient’s improvement and recovery.
- Full disclosure of the limitations of the physical examination or any lab tests.
- Ways to contact the physician going forward.
- Patient insights on their experience and reaction to what they just heard.
The researchers, who published their findings in JAMA Network Open, recommend that the conversation be transcribed in real time using voice recognition software and a microphone, and then printed for the patient to take home. The physician should make eye contact with the patient during the conversation, they suggested.
“Patients felt it was a conversation, that they actually understood what was said. Most patients felt like they were partners during the encounter,” said Maram Khazen, PhD, a coauthor of the paper, who studies communication dynamics. Dr. Khazen was a visiting postdoctoral fellow with Dr. Schiff during the study, and is now a lecturer at the Max Stern Yezreel Valley College in Israel.
Hardeep Singh, MD, MPH, a patient safety researcher at the Michael E. DeBakey Veterans Affairs Medical Center and Baylor College of Medicine in Houston, called the new work “a great start,” but said that the complexity of the field warrants more research into the tool. Dr. Singh was not involved in the study.
Dr. Singh pointed out that many of the patient voices came from spokespeople for advocacy groups, and that these participants are not necessarily representative of actual people with unclear diagnoses.
“The choice of words really matters,” said Dr. Singh, who led a 2018 study that showed that people reacted more negatively when physicians bluntly acknowledged uncertainty than when they walked patients through different possible diagnoses. Dr. Schiff and Dr. Khazen’s framework offers good principles for discussing uncertainty, he added, but further research is needed on the optimal language to use during conversations.
“It’s really encouraging that we’re seeing high-quality research like this, that leverages patient engagement principles,” said Dimitrios Papanagnou, MD, MPH, an emergency medicine physician and vice dean of medicine at Thomas Jefferson University in Philadelphia.
Dr. Papanagnou, who was not part of the study, called for diverse patients to be part of conversations about diagnostic uncertainty.
“Are we having patients from diverse experiences, from underrepresented groups, participate in this kind of work?” Dr. Papanagnou asked. Dr. Schiff and Dr. Khazen said they agree that the tool needs to be tested in larger samples of diverse patients.
Some common themes about how to communicate diagnostic uncertainty are emerging in multiple areas of medicine. Dr. Papanagnou helped develop an uncertainty communication checklist for discharging patients from an emergency department to home, with principles similar to those that Dr. Schiff and Dr. Khazen recommend for primary care providers.
The study was funded by Harvard Hospitals’ malpractice insurer, the Controlled Risk Insurance Company. The authors disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Physicians often struggle with telling patients when they are unsure about a diagnosis. In the absence of clarity, doctors may fear losing a patient’s trust by appearing unsure.
Yet diagnostic uncertainty is an inevitable part of medicine.
“It’s often uncertain what is really going on. People have lots of unspecific symptoms,” said Gordon D. Schiff, MD, a patient safety researcher at Harvard Medical School and Brigham and Women’s Hospital in Boston.
By one estimate, more than one-third of patients are discharged from an emergency department without a clear diagnosis. Physicians may order more tests to try to resolve uncertainty, but this method is not foolproof and may lead to increased health care costs. Physicians can use an uncertain diagnosis as an opportunity to improve conversations with patients, Dr. Schiff said.
“How do you talk to patients about that? How do you convey that?” Dr. Schiff asked.
To begin to answer these questions, The scenarios included an enlarged lymph node in a patient in remission for lymphoma, which could suggest recurrence of the disease but not necessarily; a patient with a new-onset headache; and another patient with an unexplained fever and a respiratory tract infection.
For each vignette, the researchers also asked patient advocates – many of whom had experienced receiving an incorrect diagnosis – for their thoughts on how the conversation should go.
Almost 70 people were consulted (24 primary care physicians, 40 patients, and five experts in informatics and quality and safety). Dr. Schiff and his colleagues produced six standardized elements that should be part of a conversation whenever a diagnosis is unclear.
- The most likely diagnosis, along with any alternatives if this isn’t certain, with phrases such as, “Sometimes we don’t have the answers, but we will keep trying to figure out what is going on.”
- Next steps – lab tests, return visits, etc.
- Expected time frame for patient’s improvement and recovery.
- Full disclosure of the limitations of the physical examination or any lab tests.
- Ways to contact the physician going forward.
- Patient insights on their experience and reaction to what they just heard.
The researchers, who published their findings in JAMA Network Open, recommend that the conversation be transcribed in real time using voice recognition software and a microphone, and then printed for the patient to take home. The physician should make eye contact with the patient during the conversation, they suggested.
“Patients felt it was a conversation, that they actually understood what was said. Most patients felt like they were partners during the encounter,” said Maram Khazen, PhD, a coauthor of the paper, who studies communication dynamics. Dr. Khazen was a visiting postdoctoral fellow with Dr. Schiff during the study, and is now a lecturer at the Max Stern Yezreel Valley College in Israel.
Hardeep Singh, MD, MPH, a patient safety researcher at the Michael E. DeBakey Veterans Affairs Medical Center and Baylor College of Medicine in Houston, called the new work “a great start,” but said that the complexity of the field warrants more research into the tool. Dr. Singh was not involved in the study.
Dr. Singh pointed out that many of the patient voices came from spokespeople for advocacy groups, and that these participants are not necessarily representative of actual people with unclear diagnoses.
“The choice of words really matters,” said Dr. Singh, who led a 2018 study that showed that people reacted more negatively when physicians bluntly acknowledged uncertainty than when they walked patients through different possible diagnoses. Dr. Schiff and Dr. Khazen’s framework offers good principles for discussing uncertainty, he added, but further research is needed on the optimal language to use during conversations.
“It’s really encouraging that we’re seeing high-quality research like this, that leverages patient engagement principles,” said Dimitrios Papanagnou, MD, MPH, an emergency medicine physician and vice dean of medicine at Thomas Jefferson University in Philadelphia.
Dr. Papanagnou, who was not part of the study, called for diverse patients to be part of conversations about diagnostic uncertainty.
“Are we having patients from diverse experiences, from underrepresented groups, participate in this kind of work?” Dr. Papanagnou asked. Dr. Schiff and Dr. Khazen said they agree that the tool needs to be tested in larger samples of diverse patients.
Some common themes about how to communicate diagnostic uncertainty are emerging in multiple areas of medicine. Dr. Papanagnou helped develop an uncertainty communication checklist for discharging patients from an emergency department to home, with principles similar to those that Dr. Schiff and Dr. Khazen recommend for primary care providers.
The study was funded by Harvard Hospitals’ malpractice insurer, the Controlled Risk Insurance Company. The authors disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Physicians often struggle with telling patients when they are unsure about a diagnosis. In the absence of clarity, doctors may fear losing a patient’s trust by appearing unsure.
Yet diagnostic uncertainty is an inevitable part of medicine.
“It’s often uncertain what is really going on. People have lots of unspecific symptoms,” said Gordon D. Schiff, MD, a patient safety researcher at Harvard Medical School and Brigham and Women’s Hospital in Boston.
By one estimate, more than one-third of patients are discharged from an emergency department without a clear diagnosis. Physicians may order more tests to try to resolve uncertainty, but this method is not foolproof and may lead to increased health care costs. Physicians can use an uncertain diagnosis as an opportunity to improve conversations with patients, Dr. Schiff said.
“How do you talk to patients about that? How do you convey that?” Dr. Schiff asked.
To begin to answer these questions, The scenarios included an enlarged lymph node in a patient in remission for lymphoma, which could suggest recurrence of the disease but not necessarily; a patient with a new-onset headache; and another patient with an unexplained fever and a respiratory tract infection.
For each vignette, the researchers also asked patient advocates – many of whom had experienced receiving an incorrect diagnosis – for their thoughts on how the conversation should go.
Almost 70 people were consulted (24 primary care physicians, 40 patients, and five experts in informatics and quality and safety). Dr. Schiff and his colleagues produced six standardized elements that should be part of a conversation whenever a diagnosis is unclear.
- The most likely diagnosis, along with any alternatives if this isn’t certain, with phrases such as, “Sometimes we don’t have the answers, but we will keep trying to figure out what is going on.”
- Next steps – lab tests, return visits, etc.
- Expected time frame for patient’s improvement and recovery.
- Full disclosure of the limitations of the physical examination or any lab tests.
- Ways to contact the physician going forward.
- Patient insights on their experience and reaction to what they just heard.
The researchers, who published their findings in JAMA Network Open, recommend that the conversation be transcribed in real time using voice recognition software and a microphone, and then printed for the patient to take home. The physician should make eye contact with the patient during the conversation, they suggested.
“Patients felt it was a conversation, that they actually understood what was said. Most patients felt like they were partners during the encounter,” said Maram Khazen, PhD, a coauthor of the paper, who studies communication dynamics. Dr. Khazen was a visiting postdoctoral fellow with Dr. Schiff during the study, and is now a lecturer at the Max Stern Yezreel Valley College in Israel.
Hardeep Singh, MD, MPH, a patient safety researcher at the Michael E. DeBakey Veterans Affairs Medical Center and Baylor College of Medicine in Houston, called the new work “a great start,” but said that the complexity of the field warrants more research into the tool. Dr. Singh was not involved in the study.
Dr. Singh pointed out that many of the patient voices came from spokespeople for advocacy groups, and that these participants are not necessarily representative of actual people with unclear diagnoses.
“The choice of words really matters,” said Dr. Singh, who led a 2018 study that showed that people reacted more negatively when physicians bluntly acknowledged uncertainty than when they walked patients through different possible diagnoses. Dr. Schiff and Dr. Khazen’s framework offers good principles for discussing uncertainty, he added, but further research is needed on the optimal language to use during conversations.
“It’s really encouraging that we’re seeing high-quality research like this, that leverages patient engagement principles,” said Dimitrios Papanagnou, MD, MPH, an emergency medicine physician and vice dean of medicine at Thomas Jefferson University in Philadelphia.
Dr. Papanagnou, who was not part of the study, called for diverse patients to be part of conversations about diagnostic uncertainty.
“Are we having patients from diverse experiences, from underrepresented groups, participate in this kind of work?” Dr. Papanagnou asked. Dr. Schiff and Dr. Khazen said they agree that the tool needs to be tested in larger samples of diverse patients.
Some common themes about how to communicate diagnostic uncertainty are emerging in multiple areas of medicine. Dr. Papanagnou helped develop an uncertainty communication checklist for discharging patients from an emergency department to home, with principles similar to those that Dr. Schiff and Dr. Khazen recommend for primary care providers.
The study was funded by Harvard Hospitals’ malpractice insurer, the Controlled Risk Insurance Company. The authors disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FROM JAMA NETWORK OPEN
Children and COVID: A look back as the fourth year begins
With 3 years of the COVID-19 experience now past, it’s safe to say that SARS-CoV-2 changed American society in ways that could not have been predicted when the first U.S. cases were reported in January of 2020.
Who would have guessed back then that not one but two vaccines would be developed, approved, and widely distributed before the end of the year? Or that those vaccines would be rejected by large segments of the population on ideological grounds? Could anyone have predicted in early 2020 that schools in 21 states would be forbidden by law to require COVID-19 vaccination in students?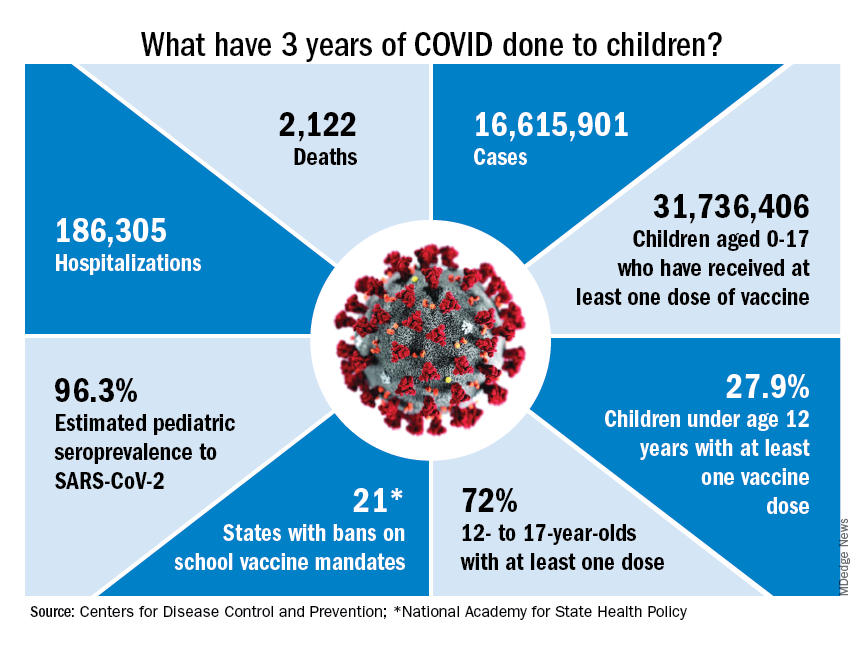
Vaccination is generally considered to be an activity of childhood, but that practice has been turned upside down with COVID-19. Among Americans aged 65 years and older, 95% have received at least one dose of vaccine, versus 27.9% of children younger than 12 years old, according to the Centers for Disease Control and Prevention.
The vaccine situation for children mirrors that of the population as a whole. The oldest children have the highest vaccination rates, and the rates decline along with age: 72.0% of those aged 12-17 years have received at least one dose, compared with 39.8% of 5- to 11-year-olds, 10.5% of 2- to 4-year-olds, and 8.0% of children under age 2, the CDC said on its COVID Data Tracker.
The youngest children were, of course, the last ones to be eligible for the vaccine, but their uptake has been much slower since emergency use was authorized in June of 2022. In the nearly 9 months since then, 9.5% of children aged 4 and under have received at least one dose, versus 66% of children aged 12-15 years in the first 9 months (May 2021 to March 2022).
Altogether, a total of 31.7 million, or 43%, of all children under age 18 had received at least one dose of COVID-19 vaccine as of March 8, 2023, according to the most recent CDC data.
Incidence: Counting COVID
Vaccination and other prevention efforts have tried to stem the tide, but what has COVID actually done to children since the Trump administration declared a nationwide emergency on March 13, 2020?
- 16.6 million cases.
- 186,035 new hospital admissions.
- 2,122 deaths.
Even the proportion of total COVID cases in children, 17.2%, is less than might be expected, given their relatively undervaccinated status.
Seroprevalence estimates seem to support the undercounting of pediatric cases. A survey of commercial laboratories working with the CDC put the seroprevalance of SARS-CoV-2 antibodies in children at 96.3% as of late 2022, based on tests of almost 27,000 specimens performed over an 8-week period from mid-October to mid-December. That would put the number of infected children at 65.7 million children.
Since Omicron
There has not been another major COVID-19 surge since the winter of 2021-2022, when the weekly rate of new cases reached 1,900 per 100,000 population in children aged 16-17 years in early January 2022 – the highest seen among children of any of the CDC’s age groups (0-4, 5-11, 12-15, 16-17) during the entire pandemic. Since the Omicron surge, the highest weekly rate was 221 per 100,000 during the week of May 15-21, again in 16- to 17-year-olds, the CDC reports.
The widely anticipated surge of COVID in the fall and winter of 2022 and 2023 – the so-called “tripledemic” involving influenza and respiratory syncytial virus – did not occur, possibly because so many Americans were vaccinated or previously infected, experts suggested. New-case rates, emergency room visits, and hospitalizations in children have continued to drop as winter comes to a close, CDC data show.
With 3 years of the COVID-19 experience now past, it’s safe to say that SARS-CoV-2 changed American society in ways that could not have been predicted when the first U.S. cases were reported in January of 2020.
Who would have guessed back then that not one but two vaccines would be developed, approved, and widely distributed before the end of the year? Or that those vaccines would be rejected by large segments of the population on ideological grounds? Could anyone have predicted in early 2020 that schools in 21 states would be forbidden by law to require COVID-19 vaccination in students?
Vaccination is generally considered to be an activity of childhood, but that practice has been turned upside down with COVID-19. Among Americans aged 65 years and older, 95% have received at least one dose of vaccine, versus 27.9% of children younger than 12 years old, according to the Centers for Disease Control and Prevention.
The vaccine situation for children mirrors that of the population as a whole. The oldest children have the highest vaccination rates, and the rates decline along with age: 72.0% of those aged 12-17 years have received at least one dose, compared with 39.8% of 5- to 11-year-olds, 10.5% of 2- to 4-year-olds, and 8.0% of children under age 2, the CDC said on its COVID Data Tracker.
The youngest children were, of course, the last ones to be eligible for the vaccine, but their uptake has been much slower since emergency use was authorized in June of 2022. In the nearly 9 months since then, 9.5% of children aged 4 and under have received at least one dose, versus 66% of children aged 12-15 years in the first 9 months (May 2021 to March 2022).
Altogether, a total of 31.7 million, or 43%, of all children under age 18 had received at least one dose of COVID-19 vaccine as of March 8, 2023, according to the most recent CDC data.
Incidence: Counting COVID
Vaccination and other prevention efforts have tried to stem the tide, but what has COVID actually done to children since the Trump administration declared a nationwide emergency on March 13, 2020?
- 16.6 million cases.
- 186,035 new hospital admissions.
- 2,122 deaths.
Even the proportion of total COVID cases in children, 17.2%, is less than might be expected, given their relatively undervaccinated status.
Seroprevalence estimates seem to support the undercounting of pediatric cases. A survey of commercial laboratories working with the CDC put the seroprevalance of SARS-CoV-2 antibodies in children at 96.3% as of late 2022, based on tests of almost 27,000 specimens performed over an 8-week period from mid-October to mid-December. That would put the number of infected children at 65.7 million children.
Since Omicron
There has not been another major COVID-19 surge since the winter of 2021-2022, when the weekly rate of new cases reached 1,900 per 100,000 population in children aged 16-17 years in early January 2022 – the highest seen among children of any of the CDC’s age groups (0-4, 5-11, 12-15, 16-17) during the entire pandemic. Since the Omicron surge, the highest weekly rate was 221 per 100,000 during the week of May 15-21, again in 16- to 17-year-olds, the CDC reports.
The widely anticipated surge of COVID in the fall and winter of 2022 and 2023 – the so-called “tripledemic” involving influenza and respiratory syncytial virus – did not occur, possibly because so many Americans were vaccinated or previously infected, experts suggested. New-case rates, emergency room visits, and hospitalizations in children have continued to drop as winter comes to a close, CDC data show.
With 3 years of the COVID-19 experience now past, it’s safe to say that SARS-CoV-2 changed American society in ways that could not have been predicted when the first U.S. cases were reported in January of 2020.
Who would have guessed back then that not one but two vaccines would be developed, approved, and widely distributed before the end of the year? Or that those vaccines would be rejected by large segments of the population on ideological grounds? Could anyone have predicted in early 2020 that schools in 21 states would be forbidden by law to require COVID-19 vaccination in students?
Vaccination is generally considered to be an activity of childhood, but that practice has been turned upside down with COVID-19. Among Americans aged 65 years and older, 95% have received at least one dose of vaccine, versus 27.9% of children younger than 12 years old, according to the Centers for Disease Control and Prevention.
The vaccine situation for children mirrors that of the population as a whole. The oldest children have the highest vaccination rates, and the rates decline along with age: 72.0% of those aged 12-17 years have received at least one dose, compared with 39.8% of 5- to 11-year-olds, 10.5% of 2- to 4-year-olds, and 8.0% of children under age 2, the CDC said on its COVID Data Tracker.
The youngest children were, of course, the last ones to be eligible for the vaccine, but their uptake has been much slower since emergency use was authorized in June of 2022. In the nearly 9 months since then, 9.5% of children aged 4 and under have received at least one dose, versus 66% of children aged 12-15 years in the first 9 months (May 2021 to March 2022).
Altogether, a total of 31.7 million, or 43%, of all children under age 18 had received at least one dose of COVID-19 vaccine as of March 8, 2023, according to the most recent CDC data.
Incidence: Counting COVID
Vaccination and other prevention efforts have tried to stem the tide, but what has COVID actually done to children since the Trump administration declared a nationwide emergency on March 13, 2020?
- 16.6 million cases.
- 186,035 new hospital admissions.
- 2,122 deaths.
Even the proportion of total COVID cases in children, 17.2%, is less than might be expected, given their relatively undervaccinated status.
Seroprevalence estimates seem to support the undercounting of pediatric cases. A survey of commercial laboratories working with the CDC put the seroprevalance of SARS-CoV-2 antibodies in children at 96.3% as of late 2022, based on tests of almost 27,000 specimens performed over an 8-week period from mid-October to mid-December. That would put the number of infected children at 65.7 million children.
Since Omicron
There has not been another major COVID-19 surge since the winter of 2021-2022, when the weekly rate of new cases reached 1,900 per 100,000 population in children aged 16-17 years in early January 2022 – the highest seen among children of any of the CDC’s age groups (0-4, 5-11, 12-15, 16-17) during the entire pandemic. Since the Omicron surge, the highest weekly rate was 221 per 100,000 during the week of May 15-21, again in 16- to 17-year-olds, the CDC reports.
The widely anticipated surge of COVID in the fall and winter of 2022 and 2023 – the so-called “tripledemic” involving influenza and respiratory syncytial virus – did not occur, possibly because so many Americans were vaccinated or previously infected, experts suggested. New-case rates, emergency room visits, and hospitalizations in children have continued to drop as winter comes to a close, CDC data show.
Factors linked with increased VTE risk in COVID outpatients
Though VTE risk is well studied and significant in those hospitalized with COVID, little is known about the risk in the outpatient setting, said the authors of the new research published online in JAMA Network Open.
The study was conducted at two integrated health care delivery systems in northern and southern California. Data were gathered from the Kaiser Permanente Virtual Data Warehouse and electronic health records.
Nearly 400,000 patients studied
Researchers, led by Margaret Fang, MD, with the division of hospital medicine, University of California, San Francisco, identified 398,530 outpatients with COVID-19 from Jan. 1, 2020, through Jan. 31, 2021.
VTE risk was low overall for ambulatory COVID patients.
“It is a reassuring study,” Dr. Fang said in an interview.
The researchers found that the risk is highest in the first 30 days after COVID-19 diagnosis (unadjusted rate, 0.58; 95% confidence interval, 0.51-0.67 per 100 person-years vs. 0.09; 95% CI, 0.08-0.11 per 100 person-years after 30 days).
Factors linked with high VTE risk
They also found that several factors were linked with a higher risk of blood clots in the study population, including being at least 55 years old; being male; having a history of blood clots or thrombophilia; and a body mass index (BMI) of at least 30 kg/m2.
The authors write, “These findings may help identify subsets of patients with COVID-19 who could benefit from VTE preventive strategies and more intensive short-term surveillance.”
Are routine anticoagulants justified?
Previously, randomized clinical trials have found that hospitalized patients with moderate COVID-19 may benefit from therapeutically dosed heparin anticoagulants but that therapeutic anticoagulation had no net benefit – and perhaps could even harm – patients who were critically ill with COVID.
“[M]uch less is known about the optimal thromboprophylaxis strategy for people with milder presentations of COVID-19 who do not require hospitalization,” they write.
Mild COVID VTE risk similar to general population
The authors note that rates of blood clots linked with COVID-19 are not much higher than the average blood clot rate in the general population, which is about 0.1-0.2 per 100 person-years.
Therefore, the results don’t justify routine administration of anticoagulation given the costs, inconvenience, and bleeding risks, they acknowledge.
Dr. Fang told this publication that it’s hard to know what to tell patients, given the overall low VTE risk. She said their study wasn’t designed to advise when to give prophylaxis.
Physicians should inform patients of their higher risk
“We should tell our patients who fall into these risk categories that blood clot is a concern after the development of COVID, especially in those first 30 days. And some people might benefit from increased surveillance,” Dr. Fang said.
”I think this study would support ongoing studies that look at whether selected patients benefit from VTE prophylaxis, for example low-dose anticoagulants,” she said.
Dr. Fang said the subgroup factors they found increased risk of blood clots for all patients, not just COVID-19 patients. It’s not clear why factors such as being male may increase blood clot risk, though that is consistent with previous literature, but higher risk with higher BMI might be related to a combination of inflammation or decreased mobility, she said.
Unanswered questions
Robert H. Hopkins Jr., MD, says the study helps answer a couple of important questions – that the VTE risk in nonhospitalized COVID-19 patients is low and when and for which patients risk may be highest.
However, there are several unanswered questions that argue against routine initiation of anticoagulants, notes the professor of internal medicine and pediatrics chief, division of general internal medicine, at University of Arkansas for Medical Sciences, Little Rock.
One is the change in the COVID variant landscape.
“We do not know whether rates of VTE are same or lower or higher with current circulating variants,” Dr. Hopkins said.
The authors acknowledge this as a limitation. Study data predate Omicron and subvariants, which appear to lower clinical severity, so it’s unclear whether VTE risk is different in this Omicron era.
Dr. Hopkins added another unknown: “We do not know whether vaccination affects rates of VTE in ambulatory breakthrough infection.”
Dr. Hopkins and the authors also note the lack of a control group in the study, to better compare risk.
Coauthor Dr. Prasad reports consultant fees from EpiExcellence LLC outside the submitted work. Coauthor Dr. Go reports grants paid to the division of research, Kaiser Permanente Northern California, from CSL Behring, Novartis, Bristol Meyers Squibb/Pfizer Alliance, and Janssen outside the submitted work.
The research was funded through Patient-Centered Outcomes Research Institute.
Dr. Hopkins reports no relevant financial relationships.
Though VTE risk is well studied and significant in those hospitalized with COVID, little is known about the risk in the outpatient setting, said the authors of the new research published online in JAMA Network Open.
The study was conducted at two integrated health care delivery systems in northern and southern California. Data were gathered from the Kaiser Permanente Virtual Data Warehouse and electronic health records.
Nearly 400,000 patients studied
Researchers, led by Margaret Fang, MD, with the division of hospital medicine, University of California, San Francisco, identified 398,530 outpatients with COVID-19 from Jan. 1, 2020, through Jan. 31, 2021.
VTE risk was low overall for ambulatory COVID patients.
“It is a reassuring study,” Dr. Fang said in an interview.
The researchers found that the risk is highest in the first 30 days after COVID-19 diagnosis (unadjusted rate, 0.58; 95% confidence interval, 0.51-0.67 per 100 person-years vs. 0.09; 95% CI, 0.08-0.11 per 100 person-years after 30 days).
Factors linked with high VTE risk
They also found that several factors were linked with a higher risk of blood clots in the study population, including being at least 55 years old; being male; having a history of blood clots or thrombophilia; and a body mass index (BMI) of at least 30 kg/m2.
The authors write, “These findings may help identify subsets of patients with COVID-19 who could benefit from VTE preventive strategies and more intensive short-term surveillance.”
Are routine anticoagulants justified?
Previously, randomized clinical trials have found that hospitalized patients with moderate COVID-19 may benefit from therapeutically dosed heparin anticoagulants but that therapeutic anticoagulation had no net benefit – and perhaps could even harm – patients who were critically ill with COVID.
“[M]uch less is known about the optimal thromboprophylaxis strategy for people with milder presentations of COVID-19 who do not require hospitalization,” they write.
Mild COVID VTE risk similar to general population
The authors note that rates of blood clots linked with COVID-19 are not much higher than the average blood clot rate in the general population, which is about 0.1-0.2 per 100 person-years.
Therefore, the results don’t justify routine administration of anticoagulation given the costs, inconvenience, and bleeding risks, they acknowledge.
Dr. Fang told this publication that it’s hard to know what to tell patients, given the overall low VTE risk. She said their study wasn’t designed to advise when to give prophylaxis.
Physicians should inform patients of their higher risk
“We should tell our patients who fall into these risk categories that blood clot is a concern after the development of COVID, especially in those first 30 days. And some people might benefit from increased surveillance,” Dr. Fang said.
”I think this study would support ongoing studies that look at whether selected patients benefit from VTE prophylaxis, for example low-dose anticoagulants,” she said.
Dr. Fang said the subgroup factors they found increased risk of blood clots for all patients, not just COVID-19 patients. It’s not clear why factors such as being male may increase blood clot risk, though that is consistent with previous literature, but higher risk with higher BMI might be related to a combination of inflammation or decreased mobility, she said.
Unanswered questions
Robert H. Hopkins Jr., MD, says the study helps answer a couple of important questions – that the VTE risk in nonhospitalized COVID-19 patients is low and when and for which patients risk may be highest.
However, there are several unanswered questions that argue against routine initiation of anticoagulants, notes the professor of internal medicine and pediatrics chief, division of general internal medicine, at University of Arkansas for Medical Sciences, Little Rock.
One is the change in the COVID variant landscape.
“We do not know whether rates of VTE are same or lower or higher with current circulating variants,” Dr. Hopkins said.
The authors acknowledge this as a limitation. Study data predate Omicron and subvariants, which appear to lower clinical severity, so it’s unclear whether VTE risk is different in this Omicron era.
Dr. Hopkins added another unknown: “We do not know whether vaccination affects rates of VTE in ambulatory breakthrough infection.”
Dr. Hopkins and the authors also note the lack of a control group in the study, to better compare risk.
Coauthor Dr. Prasad reports consultant fees from EpiExcellence LLC outside the submitted work. Coauthor Dr. Go reports grants paid to the division of research, Kaiser Permanente Northern California, from CSL Behring, Novartis, Bristol Meyers Squibb/Pfizer Alliance, and Janssen outside the submitted work.
The research was funded through Patient-Centered Outcomes Research Institute.
Dr. Hopkins reports no relevant financial relationships.
Though VTE risk is well studied and significant in those hospitalized with COVID, little is known about the risk in the outpatient setting, said the authors of the new research published online in JAMA Network Open.
The study was conducted at two integrated health care delivery systems in northern and southern California. Data were gathered from the Kaiser Permanente Virtual Data Warehouse and electronic health records.
Nearly 400,000 patients studied
Researchers, led by Margaret Fang, MD, with the division of hospital medicine, University of California, San Francisco, identified 398,530 outpatients with COVID-19 from Jan. 1, 2020, through Jan. 31, 2021.
VTE risk was low overall for ambulatory COVID patients.
“It is a reassuring study,” Dr. Fang said in an interview.
The researchers found that the risk is highest in the first 30 days after COVID-19 diagnosis (unadjusted rate, 0.58; 95% confidence interval, 0.51-0.67 per 100 person-years vs. 0.09; 95% CI, 0.08-0.11 per 100 person-years after 30 days).
Factors linked with high VTE risk
They also found that several factors were linked with a higher risk of blood clots in the study population, including being at least 55 years old; being male; having a history of blood clots or thrombophilia; and a body mass index (BMI) of at least 30 kg/m2.
The authors write, “These findings may help identify subsets of patients with COVID-19 who could benefit from VTE preventive strategies and more intensive short-term surveillance.”
Are routine anticoagulants justified?
Previously, randomized clinical trials have found that hospitalized patients with moderate COVID-19 may benefit from therapeutically dosed heparin anticoagulants but that therapeutic anticoagulation had no net benefit – and perhaps could even harm – patients who were critically ill with COVID.
“[M]uch less is known about the optimal thromboprophylaxis strategy for people with milder presentations of COVID-19 who do not require hospitalization,” they write.
Mild COVID VTE risk similar to general population
The authors note that rates of blood clots linked with COVID-19 are not much higher than the average blood clot rate in the general population, which is about 0.1-0.2 per 100 person-years.
Therefore, the results don’t justify routine administration of anticoagulation given the costs, inconvenience, and bleeding risks, they acknowledge.
Dr. Fang told this publication that it’s hard to know what to tell patients, given the overall low VTE risk. She said their study wasn’t designed to advise when to give prophylaxis.
Physicians should inform patients of their higher risk
“We should tell our patients who fall into these risk categories that blood clot is a concern after the development of COVID, especially in those first 30 days. And some people might benefit from increased surveillance,” Dr. Fang said.
”I think this study would support ongoing studies that look at whether selected patients benefit from VTE prophylaxis, for example low-dose anticoagulants,” she said.
Dr. Fang said the subgroup factors they found increased risk of blood clots for all patients, not just COVID-19 patients. It’s not clear why factors such as being male may increase blood clot risk, though that is consistent with previous literature, but higher risk with higher BMI might be related to a combination of inflammation or decreased mobility, she said.
Unanswered questions
Robert H. Hopkins Jr., MD, says the study helps answer a couple of important questions – that the VTE risk in nonhospitalized COVID-19 patients is low and when and for which patients risk may be highest.
However, there are several unanswered questions that argue against routine initiation of anticoagulants, notes the professor of internal medicine and pediatrics chief, division of general internal medicine, at University of Arkansas for Medical Sciences, Little Rock.
One is the change in the COVID variant landscape.
“We do not know whether rates of VTE are same or lower or higher with current circulating variants,” Dr. Hopkins said.
The authors acknowledge this as a limitation. Study data predate Omicron and subvariants, which appear to lower clinical severity, so it’s unclear whether VTE risk is different in this Omicron era.
Dr. Hopkins added another unknown: “We do not know whether vaccination affects rates of VTE in ambulatory breakthrough infection.”
Dr. Hopkins and the authors also note the lack of a control group in the study, to better compare risk.
Coauthor Dr. Prasad reports consultant fees from EpiExcellence LLC outside the submitted work. Coauthor Dr. Go reports grants paid to the division of research, Kaiser Permanente Northern California, from CSL Behring, Novartis, Bristol Meyers Squibb/Pfizer Alliance, and Janssen outside the submitted work.
The research was funded through Patient-Centered Outcomes Research Institute.
Dr. Hopkins reports no relevant financial relationships.
FROM JAMA NETWORK OPEN
Opioid overdose is an important cause of postpartum death
(OUD), according to research published in Obstetrics and Gynecology.
Opioid overdose deaths account for up to 10% of pregnancy-associated deaths in the United States, and 75% of the deliveries of women with OUD are covered by Medicaid, according to lead author Elizabeth Suarez, PhD, MPH, with the division of pharmacoepidemiology and pharmacoeconomics at Brigham and Women’s Hospital and Harvard Medical School in Boston, and colleagues.
Nearly 5 million deliveries studied
Researchers studied claims data from Medicaid and the National Death Index database in the United States from 2006 to 2013 for 4,972,061 deliveries. They also identified a subgroup of women with a documented history of OUD in the 3 months before delivery.
They found the incidence of postpartum opioid overdose deaths was 5.4 per 100,000 deliveries (95% confidence interval, 4.5-6.4) among all in the study and 118 per 100,000 (95% CI, 84-163) among individuals with OUD.
Incidence of all-cause postpartum death was six times higher in women with OUD than in all the women studied. Common causes of death of those with OUD were other drug- and alcohol-related deaths (47/100,000); suicide (26/100,000); and other injuries, including accidents and falls (33/100,000).
Risk factors strongly linked with postpartum opioid overdose death included mental health and other substance use disorders.
Medication significantly lowers death risk
The authors also documented the benefit of buprenorphine or methadone for OUD.
For women with OUD who used medication to treat OUD post partum, odds of opioid overdose death were 60% lower (odds ratio, 0.4; 95% CI 0.1-0.9).
As important as use of medication, Marcela Smid, MD, MS, writes in an accompanying editorial, is noting that 80% of the women in this study who died of opioid overdoses had contact with a health care provider before death.
“Both of these results indicate that we have the means and opportunity to prevent these deaths,” writes Dr. Smid, with the division of maternal fetal medicine, University of Utah Health in Salt Lake City.
Dismal numbers on ob.gyns. trained to prescribe medications
She points out some barriers, however. Most clinicians, she notes, lack time and training to prescribe buprenorphine, and in 2019, fewer than 2% of ob.gyns. who accept Medicaid were able to prescribe it.
Her charge to ob.gyns.: “We need to help identify individuals who are at high risk of OUD or opioid overdose by screening.” A validated screening tool should be used at prenatal and postpartum appointments.
On a bigger scale, she urges Medicaid to be expanded for a full year post partum through the American Rescue Act’s State Plan Amendment, something only 28 states and Washington, D.C., have done so far.
Dr. Smid points out some good news, however: President Joe Biden signed the Consolidated Appropriations Act 2023, which eliminated the “X” waiver.
Now all clinicians who have a Drug Enforcement Administration registration that includes Schedule III authority can prescribe buprenorphine for OUD if applicable state law allows it.
But that calls for medical schools and residency programs to prioritize addiction medicine as a core competency, Dr. Smid says.
Getting naloxone to patients, families
One of the potential interventions the study authors suggest is providing naloxone prescriptions and training to pregnant and postpartum women who have a substance use history and to their partners and significant others.
However, Mishka Terplan, MD, MPH, told this publication, “It’s one thing to write a prescription; it’s another thing for the person to actually get the medication.” He is medical director of the Friends Research Institute in Baltimore, an ob.gyn. who specializes in addiction medicine.
“What can we do?” We can think about how to get naloxone into people’s hands at discharge from the hospital after they give birth, instead of prescribing. That would mean that health systems need to prioritize this, he said. “We give people discharge medications all the time.”
Still, naloxone can’t be seen as the answer, he said.
He compares it to defibrillators in public places, which are for rescues, not reversing a population problem.
“Some people think that naloxone reversals are doing something about OUD. It’s doing about as much about OUD as defibrillators do for cardiovascular disease,” he said.
The best help, he says, will be continuation of treatment.
“Addiction is a chronic condition,” he says, “but often we only provide episodic care. We see that particularly in pregnancy. Once the pregnancy is finished, there’s not categorical continuation of insurance.”
Even if you do have insurance, it’s hard to find a clinic that’s family friendly, he notes. “You might not feel comfortable taking your newborn and standing in line in the morning to get your daily methodone dose. We have to make those environments more welcoming.”
Problem probably understated
He also says that though the study was well done given the data available, he’s frustrated that researchers still have to depend on billing data and can’t capture factors such as child care availability, living wages, and continuation of health insurance. Additionally, not everyone is coded correctly for OUD.
“It’s all Medicaid, so it’s only people who continued with care,” he pointed out. That means these numbers may actually underrepresent the problem.
Still, he says it’s important to realize the magnitude of deaths this study does highlight in this population.
In people with OUD in the postpartum period, the deaths are more than 1 in 1,000.
“That should be alarming,” Dr. Terplan said. “That’s a very big number from a public health perspective.”
Coauthor Kathryn J. Gray received payment from Aetion Inc., Roche, and BillionToOne. Funds were paid to the University of Utah for Dr. Smid from Alydia Inc. for being the site principal investigator for a study of the JADA device, and from Gilead for Dr. Smid’s study of hepatitis C in pregnancy; she was also a consultant for Organon and Rhia Ventures. Dr. Terplan reports no relevant financial relationships.
(OUD), according to research published in Obstetrics and Gynecology.
Opioid overdose deaths account for up to 10% of pregnancy-associated deaths in the United States, and 75% of the deliveries of women with OUD are covered by Medicaid, according to lead author Elizabeth Suarez, PhD, MPH, with the division of pharmacoepidemiology and pharmacoeconomics at Brigham and Women’s Hospital and Harvard Medical School in Boston, and colleagues.
Nearly 5 million deliveries studied
Researchers studied claims data from Medicaid and the National Death Index database in the United States from 2006 to 2013 for 4,972,061 deliveries. They also identified a subgroup of women with a documented history of OUD in the 3 months before delivery.
They found the incidence of postpartum opioid overdose deaths was 5.4 per 100,000 deliveries (95% confidence interval, 4.5-6.4) among all in the study and 118 per 100,000 (95% CI, 84-163) among individuals with OUD.
Incidence of all-cause postpartum death was six times higher in women with OUD than in all the women studied. Common causes of death of those with OUD were other drug- and alcohol-related deaths (47/100,000); suicide (26/100,000); and other injuries, including accidents and falls (33/100,000).
Risk factors strongly linked with postpartum opioid overdose death included mental health and other substance use disorders.
Medication significantly lowers death risk
The authors also documented the benefit of buprenorphine or methadone for OUD.
For women with OUD who used medication to treat OUD post partum, odds of opioid overdose death were 60% lower (odds ratio, 0.4; 95% CI 0.1-0.9).
As important as use of medication, Marcela Smid, MD, MS, writes in an accompanying editorial, is noting that 80% of the women in this study who died of opioid overdoses had contact with a health care provider before death.
“Both of these results indicate that we have the means and opportunity to prevent these deaths,” writes Dr. Smid, with the division of maternal fetal medicine, University of Utah Health in Salt Lake City.
Dismal numbers on ob.gyns. trained to prescribe medications
She points out some barriers, however. Most clinicians, she notes, lack time and training to prescribe buprenorphine, and in 2019, fewer than 2% of ob.gyns. who accept Medicaid were able to prescribe it.
Her charge to ob.gyns.: “We need to help identify individuals who are at high risk of OUD or opioid overdose by screening.” A validated screening tool should be used at prenatal and postpartum appointments.
On a bigger scale, she urges Medicaid to be expanded for a full year post partum through the American Rescue Act’s State Plan Amendment, something only 28 states and Washington, D.C., have done so far.
Dr. Smid points out some good news, however: President Joe Biden signed the Consolidated Appropriations Act 2023, which eliminated the “X” waiver.
Now all clinicians who have a Drug Enforcement Administration registration that includes Schedule III authority can prescribe buprenorphine for OUD if applicable state law allows it.
But that calls for medical schools and residency programs to prioritize addiction medicine as a core competency, Dr. Smid says.
Getting naloxone to patients, families
One of the potential interventions the study authors suggest is providing naloxone prescriptions and training to pregnant and postpartum women who have a substance use history and to their partners and significant others.
However, Mishka Terplan, MD, MPH, told this publication, “It’s one thing to write a prescription; it’s another thing for the person to actually get the medication.” He is medical director of the Friends Research Institute in Baltimore, an ob.gyn. who specializes in addiction medicine.
“What can we do?” We can think about how to get naloxone into people’s hands at discharge from the hospital after they give birth, instead of prescribing. That would mean that health systems need to prioritize this, he said. “We give people discharge medications all the time.”
Still, naloxone can’t be seen as the answer, he said.
He compares it to defibrillators in public places, which are for rescues, not reversing a population problem.
“Some people think that naloxone reversals are doing something about OUD. It’s doing about as much about OUD as defibrillators do for cardiovascular disease,” he said.
The best help, he says, will be continuation of treatment.
“Addiction is a chronic condition,” he says, “but often we only provide episodic care. We see that particularly in pregnancy. Once the pregnancy is finished, there’s not categorical continuation of insurance.”
Even if you do have insurance, it’s hard to find a clinic that’s family friendly, he notes. “You might not feel comfortable taking your newborn and standing in line in the morning to get your daily methodone dose. We have to make those environments more welcoming.”
Problem probably understated
He also says that though the study was well done given the data available, he’s frustrated that researchers still have to depend on billing data and can’t capture factors such as child care availability, living wages, and continuation of health insurance. Additionally, not everyone is coded correctly for OUD.
“It’s all Medicaid, so it’s only people who continued with care,” he pointed out. That means these numbers may actually underrepresent the problem.
Still, he says it’s important to realize the magnitude of deaths this study does highlight in this population.
In people with OUD in the postpartum period, the deaths are more than 1 in 1,000.
“That should be alarming,” Dr. Terplan said. “That’s a very big number from a public health perspective.”
Coauthor Kathryn J. Gray received payment from Aetion Inc., Roche, and BillionToOne. Funds were paid to the University of Utah for Dr. Smid from Alydia Inc. for being the site principal investigator for a study of the JADA device, and from Gilead for Dr. Smid’s study of hepatitis C in pregnancy; she was also a consultant for Organon and Rhia Ventures. Dr. Terplan reports no relevant financial relationships.
(OUD), according to research published in Obstetrics and Gynecology.
Opioid overdose deaths account for up to 10% of pregnancy-associated deaths in the United States, and 75% of the deliveries of women with OUD are covered by Medicaid, according to lead author Elizabeth Suarez, PhD, MPH, with the division of pharmacoepidemiology and pharmacoeconomics at Brigham and Women’s Hospital and Harvard Medical School in Boston, and colleagues.
Nearly 5 million deliveries studied
Researchers studied claims data from Medicaid and the National Death Index database in the United States from 2006 to 2013 for 4,972,061 deliveries. They also identified a subgroup of women with a documented history of OUD in the 3 months before delivery.
They found the incidence of postpartum opioid overdose deaths was 5.4 per 100,000 deliveries (95% confidence interval, 4.5-6.4) among all in the study and 118 per 100,000 (95% CI, 84-163) among individuals with OUD.
Incidence of all-cause postpartum death was six times higher in women with OUD than in all the women studied. Common causes of death of those with OUD were other drug- and alcohol-related deaths (47/100,000); suicide (26/100,000); and other injuries, including accidents and falls (33/100,000).
Risk factors strongly linked with postpartum opioid overdose death included mental health and other substance use disorders.
Medication significantly lowers death risk
The authors also documented the benefit of buprenorphine or methadone for OUD.
For women with OUD who used medication to treat OUD post partum, odds of opioid overdose death were 60% lower (odds ratio, 0.4; 95% CI 0.1-0.9).
As important as use of medication, Marcela Smid, MD, MS, writes in an accompanying editorial, is noting that 80% of the women in this study who died of opioid overdoses had contact with a health care provider before death.
“Both of these results indicate that we have the means and opportunity to prevent these deaths,” writes Dr. Smid, with the division of maternal fetal medicine, University of Utah Health in Salt Lake City.
Dismal numbers on ob.gyns. trained to prescribe medications
She points out some barriers, however. Most clinicians, she notes, lack time and training to prescribe buprenorphine, and in 2019, fewer than 2% of ob.gyns. who accept Medicaid were able to prescribe it.
Her charge to ob.gyns.: “We need to help identify individuals who are at high risk of OUD or opioid overdose by screening.” A validated screening tool should be used at prenatal and postpartum appointments.
On a bigger scale, she urges Medicaid to be expanded for a full year post partum through the American Rescue Act’s State Plan Amendment, something only 28 states and Washington, D.C., have done so far.
Dr. Smid points out some good news, however: President Joe Biden signed the Consolidated Appropriations Act 2023, which eliminated the “X” waiver.
Now all clinicians who have a Drug Enforcement Administration registration that includes Schedule III authority can prescribe buprenorphine for OUD if applicable state law allows it.
But that calls for medical schools and residency programs to prioritize addiction medicine as a core competency, Dr. Smid says.
Getting naloxone to patients, families
One of the potential interventions the study authors suggest is providing naloxone prescriptions and training to pregnant and postpartum women who have a substance use history and to their partners and significant others.
However, Mishka Terplan, MD, MPH, told this publication, “It’s one thing to write a prescription; it’s another thing for the person to actually get the medication.” He is medical director of the Friends Research Institute in Baltimore, an ob.gyn. who specializes in addiction medicine.
“What can we do?” We can think about how to get naloxone into people’s hands at discharge from the hospital after they give birth, instead of prescribing. That would mean that health systems need to prioritize this, he said. “We give people discharge medications all the time.”
Still, naloxone can’t be seen as the answer, he said.
He compares it to defibrillators in public places, which are for rescues, not reversing a population problem.
“Some people think that naloxone reversals are doing something about OUD. It’s doing about as much about OUD as defibrillators do for cardiovascular disease,” he said.
The best help, he says, will be continuation of treatment.
“Addiction is a chronic condition,” he says, “but often we only provide episodic care. We see that particularly in pregnancy. Once the pregnancy is finished, there’s not categorical continuation of insurance.”
Even if you do have insurance, it’s hard to find a clinic that’s family friendly, he notes. “You might not feel comfortable taking your newborn and standing in line in the morning to get your daily methodone dose. We have to make those environments more welcoming.”
Problem probably understated
He also says that though the study was well done given the data available, he’s frustrated that researchers still have to depend on billing data and can’t capture factors such as child care availability, living wages, and continuation of health insurance. Additionally, not everyone is coded correctly for OUD.
“It’s all Medicaid, so it’s only people who continued with care,” he pointed out. That means these numbers may actually underrepresent the problem.
Still, he says it’s important to realize the magnitude of deaths this study does highlight in this population.
In people with OUD in the postpartum period, the deaths are more than 1 in 1,000.
“That should be alarming,” Dr. Terplan said. “That’s a very big number from a public health perspective.”
Coauthor Kathryn J. Gray received payment from Aetion Inc., Roche, and BillionToOne. Funds were paid to the University of Utah for Dr. Smid from Alydia Inc. for being the site principal investigator for a study of the JADA device, and from Gilead for Dr. Smid’s study of hepatitis C in pregnancy; she was also a consultant for Organon and Rhia Ventures. Dr. Terplan reports no relevant financial relationships.
FROM OBSTETRICS AND GYNECOLOGY
The 2023 ‘Meddy’ awards
Without further ado (or comedy skits or musical numbers or extended tributes or commercials), the Meddys go to ...
Best depiction of emergency medicine’s rollercoaster
M*A*S*H (1970)
The original film, not the TV show, jumps from Frank Burns being hauled away in a straitjacket to a soldier’s spurting neck wound. Hawkeye Pierce calmly steps in and we see the entire sequence of him applying pressure, then stepping back to gown-and-glove (“it’s going to spurt a bit”), then jumping back in with arterial sutures, quipping, “Baby, we’re gonna see some stitchin’ like you never saw before.” After that, cocktail hour. Yes, medicine in Hollywood can be overdramatized and even inaccurate, but Robert Altman’s take on the novel by former U.S. Army surgeon Richard Hooker still stands tall for just how crazy emergency medicine can be.
Best ‘is there a doctor in the house?’ moment
Field of Dreams (1989)
When Ray Kinsella’s daughter gets knocked off the back of the bleachers, everything stops. No one knows what to do … except Doc “Moonlight” Graham, who gives up his life’s (and afterlife’s) dream to step off the field and save the girl from choking to death. Burt Lancaster, in his final movie role, embodies everything people wish a doctor to be: Calm, kind, and able to offer a quick, effective solution to a crisis. “Hey rookie! You were good.” Yes, he sure was.
Most unethical doctor
Elvis (2022)
No doctor wants to be remembered as the guy who killed Elvis. But that legacy clings to Dr. George Nichopoulos, Elvis’s personal physician in the 1970s. In Elvis, Dr. Nichopoulos, played by Tony Nixon, hovers in the background, enabling the King’s worsening addictions. Taking late-night calls for narcotics and injecting the unconscious star with stimulants, “unethical” is an understatement for the fictional “Dr. Nick.” The real Dr. Nichopoulos was acquitted of wrongdoing in Elvis’ death, although there is little doubt that the thousands of medication doses he prescribed played a role. When his license was finally revoked for overprescribing in the 1990s, the obliging doc reportedly claimed, “I cared too much.”
Best self-use of a defibrillator
Casino Royale (2006)
We expect backlash in the post-award press conference since James Bond technically only attempted to self-defibrillate in the passenger seat of his car. He never attached the device to the leads. Vesper Lynd had to pick up his slack and save the day. Also, supporters of fellow self-defibrillating nominee Jason Statham in Crank will no doubt raise a stink on Twitter. But we stand by our choice because it was such an, ahem, heart-stopper of a scene.
Best worst patient lying about an injury
Tár (2022)
Love it or hate it, few recent movies have been as polarizing as Tár. Cate Blanchett’s portrayal of a musical genius might be toweringly brilliant or outrageously offensive (or both) depending on whom you ask. But clearly the character has a loose relationship with facts. More than a few doctors might have raised an eyebrow had Lydia Tár appeared with injuries to her face, claiming to have been attacked in a mugging. In reality, Lydia tripped and fell while pursuing an attractive young cellist into a hazardous basement. Did she lie to protect her image, preserve her marriage, or – like many patients – avoid a lecture on unhealthy behavior? We pick D, all of the above.
Best therapy for a speech disorder
The King’s Speech (2010)
Public speaking might cause anxiety for many of us, but how about doing it in front of a global radio audience while wrestling with a speech disorder? Based on a true story, The King’s Speech revealed that terrifying experience for England’s King George VI. Enter Lionel Logue, played by Geoffrey Rush. Irreverent, unconventional, and untrained, the Australian pioneer in speech and language therapy uses a range of strategies – some of which are still used today – to help the royal find his voice. But when singing, shouting swear words, and provoking rage don’t do the trick, Mr. Logue turns to psychotherapy to unearth the childhood traumas at the root of the king’s disability. Experience, as Mr. Logue tells his patient, matters just as much as “letters after your name.”
A version of this article first appeared on Medscape.com.
Without further ado (or comedy skits or musical numbers or extended tributes or commercials), the Meddys go to ...
Best depiction of emergency medicine’s rollercoaster
M*A*S*H (1970)
The original film, not the TV show, jumps from Frank Burns being hauled away in a straitjacket to a soldier’s spurting neck wound. Hawkeye Pierce calmly steps in and we see the entire sequence of him applying pressure, then stepping back to gown-and-glove (“it’s going to spurt a bit”), then jumping back in with arterial sutures, quipping, “Baby, we’re gonna see some stitchin’ like you never saw before.” After that, cocktail hour. Yes, medicine in Hollywood can be overdramatized and even inaccurate, but Robert Altman’s take on the novel by former U.S. Army surgeon Richard Hooker still stands tall for just how crazy emergency medicine can be.
Best ‘is there a doctor in the house?’ moment
Field of Dreams (1989)
When Ray Kinsella’s daughter gets knocked off the back of the bleachers, everything stops. No one knows what to do … except Doc “Moonlight” Graham, who gives up his life’s (and afterlife’s) dream to step off the field and save the girl from choking to death. Burt Lancaster, in his final movie role, embodies everything people wish a doctor to be: Calm, kind, and able to offer a quick, effective solution to a crisis. “Hey rookie! You were good.” Yes, he sure was.
Most unethical doctor
Elvis (2022)
No doctor wants to be remembered as the guy who killed Elvis. But that legacy clings to Dr. George Nichopoulos, Elvis’s personal physician in the 1970s. In Elvis, Dr. Nichopoulos, played by Tony Nixon, hovers in the background, enabling the King’s worsening addictions. Taking late-night calls for narcotics and injecting the unconscious star with stimulants, “unethical” is an understatement for the fictional “Dr. Nick.” The real Dr. Nichopoulos was acquitted of wrongdoing in Elvis’ death, although there is little doubt that the thousands of medication doses he prescribed played a role. When his license was finally revoked for overprescribing in the 1990s, the obliging doc reportedly claimed, “I cared too much.”
Best self-use of a defibrillator
Casino Royale (2006)
We expect backlash in the post-award press conference since James Bond technically only attempted to self-defibrillate in the passenger seat of his car. He never attached the device to the leads. Vesper Lynd had to pick up his slack and save the day. Also, supporters of fellow self-defibrillating nominee Jason Statham in Crank will no doubt raise a stink on Twitter. But we stand by our choice because it was such an, ahem, heart-stopper of a scene.
Best worst patient lying about an injury
Tár (2022)
Love it or hate it, few recent movies have been as polarizing as Tár. Cate Blanchett’s portrayal of a musical genius might be toweringly brilliant or outrageously offensive (or both) depending on whom you ask. But clearly the character has a loose relationship with facts. More than a few doctors might have raised an eyebrow had Lydia Tár appeared with injuries to her face, claiming to have been attacked in a mugging. In reality, Lydia tripped and fell while pursuing an attractive young cellist into a hazardous basement. Did she lie to protect her image, preserve her marriage, or – like many patients – avoid a lecture on unhealthy behavior? We pick D, all of the above.
Best therapy for a speech disorder
The King’s Speech (2010)
Public speaking might cause anxiety for many of us, but how about doing it in front of a global radio audience while wrestling with a speech disorder? Based on a true story, The King’s Speech revealed that terrifying experience for England’s King George VI. Enter Lionel Logue, played by Geoffrey Rush. Irreverent, unconventional, and untrained, the Australian pioneer in speech and language therapy uses a range of strategies – some of which are still used today – to help the royal find his voice. But when singing, shouting swear words, and provoking rage don’t do the trick, Mr. Logue turns to psychotherapy to unearth the childhood traumas at the root of the king’s disability. Experience, as Mr. Logue tells his patient, matters just as much as “letters after your name.”
A version of this article first appeared on Medscape.com.
Without further ado (or comedy skits or musical numbers or extended tributes or commercials), the Meddys go to ...
Best depiction of emergency medicine’s rollercoaster
M*A*S*H (1970)
The original film, not the TV show, jumps from Frank Burns being hauled away in a straitjacket to a soldier’s spurting neck wound. Hawkeye Pierce calmly steps in and we see the entire sequence of him applying pressure, then stepping back to gown-and-glove (“it’s going to spurt a bit”), then jumping back in with arterial sutures, quipping, “Baby, we’re gonna see some stitchin’ like you never saw before.” After that, cocktail hour. Yes, medicine in Hollywood can be overdramatized and even inaccurate, but Robert Altman’s take on the novel by former U.S. Army surgeon Richard Hooker still stands tall for just how crazy emergency medicine can be.
Best ‘is there a doctor in the house?’ moment
Field of Dreams (1989)
When Ray Kinsella’s daughter gets knocked off the back of the bleachers, everything stops. No one knows what to do … except Doc “Moonlight” Graham, who gives up his life’s (and afterlife’s) dream to step off the field and save the girl from choking to death. Burt Lancaster, in his final movie role, embodies everything people wish a doctor to be: Calm, kind, and able to offer a quick, effective solution to a crisis. “Hey rookie! You were good.” Yes, he sure was.
Most unethical doctor
Elvis (2022)
No doctor wants to be remembered as the guy who killed Elvis. But that legacy clings to Dr. George Nichopoulos, Elvis’s personal physician in the 1970s. In Elvis, Dr. Nichopoulos, played by Tony Nixon, hovers in the background, enabling the King’s worsening addictions. Taking late-night calls for narcotics and injecting the unconscious star with stimulants, “unethical” is an understatement for the fictional “Dr. Nick.” The real Dr. Nichopoulos was acquitted of wrongdoing in Elvis’ death, although there is little doubt that the thousands of medication doses he prescribed played a role. When his license was finally revoked for overprescribing in the 1990s, the obliging doc reportedly claimed, “I cared too much.”
Best self-use of a defibrillator
Casino Royale (2006)
We expect backlash in the post-award press conference since James Bond technically only attempted to self-defibrillate in the passenger seat of his car. He never attached the device to the leads. Vesper Lynd had to pick up his slack and save the day. Also, supporters of fellow self-defibrillating nominee Jason Statham in Crank will no doubt raise a stink on Twitter. But we stand by our choice because it was such an, ahem, heart-stopper of a scene.
Best worst patient lying about an injury
Tár (2022)
Love it or hate it, few recent movies have been as polarizing as Tár. Cate Blanchett’s portrayal of a musical genius might be toweringly brilliant or outrageously offensive (or both) depending on whom you ask. But clearly the character has a loose relationship with facts. More than a few doctors might have raised an eyebrow had Lydia Tár appeared with injuries to her face, claiming to have been attacked in a mugging. In reality, Lydia tripped and fell while pursuing an attractive young cellist into a hazardous basement. Did she lie to protect her image, preserve her marriage, or – like many patients – avoid a lecture on unhealthy behavior? We pick D, all of the above.
Best therapy for a speech disorder
The King’s Speech (2010)
Public speaking might cause anxiety for many of us, but how about doing it in front of a global radio audience while wrestling with a speech disorder? Based on a true story, The King’s Speech revealed that terrifying experience for England’s King George VI. Enter Lionel Logue, played by Geoffrey Rush. Irreverent, unconventional, and untrained, the Australian pioneer in speech and language therapy uses a range of strategies – some of which are still used today – to help the royal find his voice. But when singing, shouting swear words, and provoking rage don’t do the trick, Mr. Logue turns to psychotherapy to unearth the childhood traumas at the root of the king’s disability. Experience, as Mr. Logue tells his patient, matters just as much as “letters after your name.”
A version of this article first appeared on Medscape.com.
A surfing PA leads an intense beach rescue
There’s a famous surf spot called Old Man’s on San Onofre beach in north San Diego County. It has nice, gentle waves that people say are similar to Waikiki in Hawaii. Since the waves are so forgiving, a lot of older people surf there. I taught my boys and some friends how to surf there. Everyone enjoys the water. It’s just a really fun vibe.
In September of 2008, I was at Old Man’s surfing with friends. After a while, I told them I was going to catch the next wave in. When I rode the wave to the beach, I saw an older guy waving his arms above his head, trying to get the lifeguard’s attention. His friend was lying on the sand at the water’s edge, unconscious. The lifeguards were about 200 yards away in their truck. Since it was off-season, they weren’t in the nearby towers.
I threw my board down on the sand and ran over. The guy was blue in the face and had some secretions around his mouth. He wasn’t breathing and had no pulse. I told his friend to get the lifeguards.
I gave two rescue breaths, and then started CPR. The waves were still lapping against his feet. I could sense people gathering around, so I said, “Okay, we’re going to be hooking him up to electricity, let’s get him out of the water.” I didn’t want him in contact with the water that could potentially transmit that electricity to anyone else.
Many hands reached in and we dragged him up to dry sand. When we pulled down his wetsuit, I saw an old midline sternotomy incision on his chest and I thought: “Oh man, he’s got a cardiac history.” I said, “I need a towel,” and suddenly there was a towel in my hand. I dried him off and continued doing CPR.
The lifeguard truck pulled up and in my peripheral vision I saw two lifeguards running over with their first aid kit. While doing compressions, I yelled over my shoulder: “Bring your AED! Get your oxygen!” They ran back to the truck.
At that point, a young woman came up and said: “I’m a nuclear medicine tech. What can I do?” I asked her to help me keep his airway open. I positioned her at his head, and she did a chin lift.
The two lifeguards came running back. One was very experienced, and he started getting the AED ready and putting the pads on. The other lifeguard was younger. He was nervous and shaking, trying to figure out how to turn on the oxygen tank. I told him: “Buddy, you better figure that out real fast.”
The AED said there was a shockable rhythm so it delivered a shock. I started compressions again. The younger lifeguard finally figured out how to turn on the oxygen tank. Now we had oxygen, a bag valve mask, and an AED. We let our training take over and quickly melded together as an efficient team.
Two minutes later the AED analyzed the rhythm and administered another shock. More compressions. Then another shock and compressions. I had so much adrenaline going through my body that I wasn’t even getting tired.
By then I had been doing compressions for a good 10 minutes. Finally, I asked: “Hey, when are the paramedics going to get here?” And the lifeguard said: “They’re on their way.” But we were all the way down on a very remote section of beach.
We did CPR on him for what seemed like eternity, probably only 15-20 minutes. Sometimes he would get a pulse back and pink up, and we could stop and get a break. But then I would see him become cyanotic. His pulse would become thready, so I would start again.
The paramedics finally arrived and loaded him into the ambulance. He was still blue in the face, and I honestly thought he would probably not survive. I said a quick prayer for him as they drove off.
For the next week, I wondered what happened to him. The next time I was at the beach, I approached some older guys and said: “Hey, I was doing CPR on a guy here last week. Do you know what happened to him?” They gave me a thumbs up sign and said: “He’s doing great!” I was amazed!
While at the beach, I saw the nuclear med tech who helped with the airway and oxygen. She told me she’d called her hospital after the incident and asked if they had received a full arrest from the beach. They said: “Yes, he was sitting up, awake and talking when he came through the door.”
A few weeks later, the local paper called and wanted to do an interview and get some photos on the beach. We set up a time to meet, and I told the reporter that if he ever found out who the guy was, I would love to meet him. I had two reasons: First, because I had done mouth-to-mouth on him and I wanted to make sure he didn’t have any communicable diseases. Second, and this is a little weirder, I wanted to find out if he had an out-of-body experience. They fascinate me.
The reporter called back a few minutes later and said: “You’ll never believe this – while I was talking to you, my phone beeped with another call. The person left a message, and it was the guy. He wants to meet you.” I was amazed at the coincidence that he would call at exactly the same time.
Later that day, we all met at the beach. I gave him a big hug and told him he looked a lot better than the last time I saw him. He now had a pacemaker/defibrillator. I found out he was married and had three teenage boys (who still have a father). He told me on the day of the incident he developed chest pain, weakness, and shortness of breath while surfing, so he came in and sat down at the water’s edge to catch his breath. That was the last thing he remembered.
When I told him I did mouth-to-mouth on him, he laughed and reassured me that he didn’t have any contagious diseases. Then I asked him about an out-of-body experience, like hovering above his body and watching the CPR. “Did you see us doing that?” I asked. He said: “No, nothing but black. The next thing I remember is waking up in the back of the ambulance, and the paramedic asked me, ‘how does it feel to come back from the dead?’ ” He answered: “I think I have to throw up.”
He was cleared to surf 6 weeks later, and I thought it would be fun to surf with him. But when he started paddling out, he said his defibrillator went off, so he has now retired to golf.
I’ve been a PA in the emergency room for 28 years. I’ve done CPR for so long it’s instinctive for me. It really saves lives, especially with the AED. When people say: “You saved his life,” I say: “No, I didn’t. I just kept him alive and let the AED do its job.”
Ms. Westbrook-May is an emergency medicine physician assistant in Newport Beach, Calif.
A version of this article first appeared on Medscape.com.
There’s a famous surf spot called Old Man’s on San Onofre beach in north San Diego County. It has nice, gentle waves that people say are similar to Waikiki in Hawaii. Since the waves are so forgiving, a lot of older people surf there. I taught my boys and some friends how to surf there. Everyone enjoys the water. It’s just a really fun vibe.
In September of 2008, I was at Old Man’s surfing with friends. After a while, I told them I was going to catch the next wave in. When I rode the wave to the beach, I saw an older guy waving his arms above his head, trying to get the lifeguard’s attention. His friend was lying on the sand at the water’s edge, unconscious. The lifeguards were about 200 yards away in their truck. Since it was off-season, they weren’t in the nearby towers.
I threw my board down on the sand and ran over. The guy was blue in the face and had some secretions around his mouth. He wasn’t breathing and had no pulse. I told his friend to get the lifeguards.
I gave two rescue breaths, and then started CPR. The waves were still lapping against his feet. I could sense people gathering around, so I said, “Okay, we’re going to be hooking him up to electricity, let’s get him out of the water.” I didn’t want him in contact with the water that could potentially transmit that electricity to anyone else.
Many hands reached in and we dragged him up to dry sand. When we pulled down his wetsuit, I saw an old midline sternotomy incision on his chest and I thought: “Oh man, he’s got a cardiac history.” I said, “I need a towel,” and suddenly there was a towel in my hand. I dried him off and continued doing CPR.
The lifeguard truck pulled up and in my peripheral vision I saw two lifeguards running over with their first aid kit. While doing compressions, I yelled over my shoulder: “Bring your AED! Get your oxygen!” They ran back to the truck.
At that point, a young woman came up and said: “I’m a nuclear medicine tech. What can I do?” I asked her to help me keep his airway open. I positioned her at his head, and she did a chin lift.
The two lifeguards came running back. One was very experienced, and he started getting the AED ready and putting the pads on. The other lifeguard was younger. He was nervous and shaking, trying to figure out how to turn on the oxygen tank. I told him: “Buddy, you better figure that out real fast.”
The AED said there was a shockable rhythm so it delivered a shock. I started compressions again. The younger lifeguard finally figured out how to turn on the oxygen tank. Now we had oxygen, a bag valve mask, and an AED. We let our training take over and quickly melded together as an efficient team.
Two minutes later the AED analyzed the rhythm and administered another shock. More compressions. Then another shock and compressions. I had so much adrenaline going through my body that I wasn’t even getting tired.
By then I had been doing compressions for a good 10 minutes. Finally, I asked: “Hey, when are the paramedics going to get here?” And the lifeguard said: “They’re on their way.” But we were all the way down on a very remote section of beach.
We did CPR on him for what seemed like eternity, probably only 15-20 minutes. Sometimes he would get a pulse back and pink up, and we could stop and get a break. But then I would see him become cyanotic. His pulse would become thready, so I would start again.
The paramedics finally arrived and loaded him into the ambulance. He was still blue in the face, and I honestly thought he would probably not survive. I said a quick prayer for him as they drove off.
For the next week, I wondered what happened to him. The next time I was at the beach, I approached some older guys and said: “Hey, I was doing CPR on a guy here last week. Do you know what happened to him?” They gave me a thumbs up sign and said: “He’s doing great!” I was amazed!
While at the beach, I saw the nuclear med tech who helped with the airway and oxygen. She told me she’d called her hospital after the incident and asked if they had received a full arrest from the beach. They said: “Yes, he was sitting up, awake and talking when he came through the door.”
A few weeks later, the local paper called and wanted to do an interview and get some photos on the beach. We set up a time to meet, and I told the reporter that if he ever found out who the guy was, I would love to meet him. I had two reasons: First, because I had done mouth-to-mouth on him and I wanted to make sure he didn’t have any communicable diseases. Second, and this is a little weirder, I wanted to find out if he had an out-of-body experience. They fascinate me.
The reporter called back a few minutes later and said: “You’ll never believe this – while I was talking to you, my phone beeped with another call. The person left a message, and it was the guy. He wants to meet you.” I was amazed at the coincidence that he would call at exactly the same time.
Later that day, we all met at the beach. I gave him a big hug and told him he looked a lot better than the last time I saw him. He now had a pacemaker/defibrillator. I found out he was married and had three teenage boys (who still have a father). He told me on the day of the incident he developed chest pain, weakness, and shortness of breath while surfing, so he came in and sat down at the water’s edge to catch his breath. That was the last thing he remembered.
When I told him I did mouth-to-mouth on him, he laughed and reassured me that he didn’t have any contagious diseases. Then I asked him about an out-of-body experience, like hovering above his body and watching the CPR. “Did you see us doing that?” I asked. He said: “No, nothing but black. The next thing I remember is waking up in the back of the ambulance, and the paramedic asked me, ‘how does it feel to come back from the dead?’ ” He answered: “I think I have to throw up.”
He was cleared to surf 6 weeks later, and I thought it would be fun to surf with him. But when he started paddling out, he said his defibrillator went off, so he has now retired to golf.
I’ve been a PA in the emergency room for 28 years. I’ve done CPR for so long it’s instinctive for me. It really saves lives, especially with the AED. When people say: “You saved his life,” I say: “No, I didn’t. I just kept him alive and let the AED do its job.”
Ms. Westbrook-May is an emergency medicine physician assistant in Newport Beach, Calif.
A version of this article first appeared on Medscape.com.
There’s a famous surf spot called Old Man’s on San Onofre beach in north San Diego County. It has nice, gentle waves that people say are similar to Waikiki in Hawaii. Since the waves are so forgiving, a lot of older people surf there. I taught my boys and some friends how to surf there. Everyone enjoys the water. It’s just a really fun vibe.
In September of 2008, I was at Old Man’s surfing with friends. After a while, I told them I was going to catch the next wave in. When I rode the wave to the beach, I saw an older guy waving his arms above his head, trying to get the lifeguard’s attention. His friend was lying on the sand at the water’s edge, unconscious. The lifeguards were about 200 yards away in their truck. Since it was off-season, they weren’t in the nearby towers.
I threw my board down on the sand and ran over. The guy was blue in the face and had some secretions around his mouth. He wasn’t breathing and had no pulse. I told his friend to get the lifeguards.
I gave two rescue breaths, and then started CPR. The waves were still lapping against his feet. I could sense people gathering around, so I said, “Okay, we’re going to be hooking him up to electricity, let’s get him out of the water.” I didn’t want him in contact with the water that could potentially transmit that electricity to anyone else.
Many hands reached in and we dragged him up to dry sand. When we pulled down his wetsuit, I saw an old midline sternotomy incision on his chest and I thought: “Oh man, he’s got a cardiac history.” I said, “I need a towel,” and suddenly there was a towel in my hand. I dried him off and continued doing CPR.
The lifeguard truck pulled up and in my peripheral vision I saw two lifeguards running over with their first aid kit. While doing compressions, I yelled over my shoulder: “Bring your AED! Get your oxygen!” They ran back to the truck.
At that point, a young woman came up and said: “I’m a nuclear medicine tech. What can I do?” I asked her to help me keep his airway open. I positioned her at his head, and she did a chin lift.
The two lifeguards came running back. One was very experienced, and he started getting the AED ready and putting the pads on. The other lifeguard was younger. He was nervous and shaking, trying to figure out how to turn on the oxygen tank. I told him: “Buddy, you better figure that out real fast.”
The AED said there was a shockable rhythm so it delivered a shock. I started compressions again. The younger lifeguard finally figured out how to turn on the oxygen tank. Now we had oxygen, a bag valve mask, and an AED. We let our training take over and quickly melded together as an efficient team.
Two minutes later the AED analyzed the rhythm and administered another shock. More compressions. Then another shock and compressions. I had so much adrenaline going through my body that I wasn’t even getting tired.
By then I had been doing compressions for a good 10 minutes. Finally, I asked: “Hey, when are the paramedics going to get here?” And the lifeguard said: “They’re on their way.” But we were all the way down on a very remote section of beach.
We did CPR on him for what seemed like eternity, probably only 15-20 minutes. Sometimes he would get a pulse back and pink up, and we could stop and get a break. But then I would see him become cyanotic. His pulse would become thready, so I would start again.
The paramedics finally arrived and loaded him into the ambulance. He was still blue in the face, and I honestly thought he would probably not survive. I said a quick prayer for him as they drove off.
For the next week, I wondered what happened to him. The next time I was at the beach, I approached some older guys and said: “Hey, I was doing CPR on a guy here last week. Do you know what happened to him?” They gave me a thumbs up sign and said: “He’s doing great!” I was amazed!
While at the beach, I saw the nuclear med tech who helped with the airway and oxygen. She told me she’d called her hospital after the incident and asked if they had received a full arrest from the beach. They said: “Yes, he was sitting up, awake and talking when he came through the door.”
A few weeks later, the local paper called and wanted to do an interview and get some photos on the beach. We set up a time to meet, and I told the reporter that if he ever found out who the guy was, I would love to meet him. I had two reasons: First, because I had done mouth-to-mouth on him and I wanted to make sure he didn’t have any communicable diseases. Second, and this is a little weirder, I wanted to find out if he had an out-of-body experience. They fascinate me.
The reporter called back a few minutes later and said: “You’ll never believe this – while I was talking to you, my phone beeped with another call. The person left a message, and it was the guy. He wants to meet you.” I was amazed at the coincidence that he would call at exactly the same time.
Later that day, we all met at the beach. I gave him a big hug and told him he looked a lot better than the last time I saw him. He now had a pacemaker/defibrillator. I found out he was married and had three teenage boys (who still have a father). He told me on the day of the incident he developed chest pain, weakness, and shortness of breath while surfing, so he came in and sat down at the water’s edge to catch his breath. That was the last thing he remembered.
When I told him I did mouth-to-mouth on him, he laughed and reassured me that he didn’t have any contagious diseases. Then I asked him about an out-of-body experience, like hovering above his body and watching the CPR. “Did you see us doing that?” I asked. He said: “No, nothing but black. The next thing I remember is waking up in the back of the ambulance, and the paramedic asked me, ‘how does it feel to come back from the dead?’ ” He answered: “I think I have to throw up.”
He was cleared to surf 6 weeks later, and I thought it would be fun to surf with him. But when he started paddling out, he said his defibrillator went off, so he has now retired to golf.
I’ve been a PA in the emergency room for 28 years. I’ve done CPR for so long it’s instinctive for me. It really saves lives, especially with the AED. When people say: “You saved his life,” I say: “No, I didn’t. I just kept him alive and let the AED do its job.”
Ms. Westbrook-May is an emergency medicine physician assistant in Newport Beach, Calif.
A version of this article first appeared on Medscape.com.






