User login
AVAHO
div[contains(@class, 'header__large-screen')]
div[contains(@class, 'read-next-article')]
div[contains(@class, 'nav-primary')]
nav[contains(@class, 'nav-primary')]
section[contains(@class, 'footer-nav-section-wrapper')]
footer[@id='footer']
div[contains(@class, 'main-prefix')]
section[contains(@class, 'nav-hidden')]
div[contains(@class, 'ce-card-content')]
nav[contains(@class, 'nav-ce-stack')]


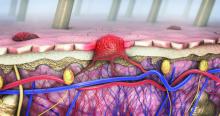
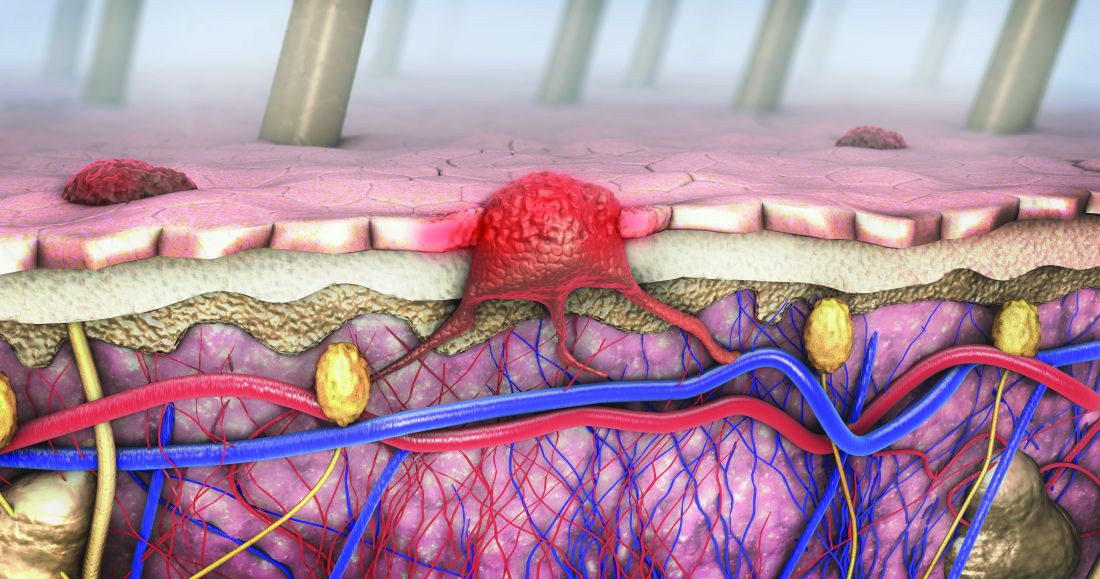
More than 97K new cutaneous melanoma diagnoses expected in 2023
SAN DIEGO – , following cancer of the colorectal area, lung and bronchus, prostate, and breast.
“The incidence of melanoma seems to have continued to go up since the early 1990s,” David E. Kent, MD, a dermatologist who practices in Macon, Ga., said at the annual Cutaneous Malignancy Update. “The death rates have been flat and may have slightly decreased.”
In 2023, the ACS estimates that about 97,610 new melanomas will be diagnosed in the United States (58,120 men and 39,490 women), and about 7,990 people are expected to die of melanoma (5,420 men and 2,570 women). In addition, ACS data from 2017-2019 project that about 2.1% of men and women will be diagnosed with cutaneous melanoma in their lifetime. To date, more than 1.3 million people in the United States live with cutaneous melanoma, and the overall 5-year survival is 93.7%.
Epidemiologic studies show an increase in melanoma incidence, primarily among White populations. “This is believed to be due primarily to sun exposure and to changing recreational behaviors and tanning bed exposures,” said Dr. Kent, who holds a faculty position in the department of dermatology at the Medical College of Georgia, Augusta. Increased surveillance and diagnosis also play a role. In the medical literature, annual increases in melanoma incidence vary from 3% to 7% per year, “which translates into a doubling of rates every 10-20 years,” he said, noting that annual melanoma costs are approximately $3.3 billion.
While incidence rates are lower in non-White, non-Hispanic populations, poor outcomes are disproportionately higher in persons of color. Blacks present at diagnosis with more advanced stage disease and are 1.5 times more likely to die from melanoma, he said, while Hispanics are 2.4 times more likely to present with stage III disease and 3.6 times more likely to have distant metastases. Persons of color also have higher rates of mucosal, acral lentiginous, and subungual melanoma.
Known genetic risk factors for melanoma include having skin types I and II, particularly those with light hair, light eyes, and freckling, and those with a family history have a twofold increased risk. Also, up to 40% of genetic cases are from inherited mutations in CDKN2A, CDK4, BAP1, and MCR1. Other genetic-related risk factors include the number and size of nevi, having atypical nevus syndrome, DNA repair defects, large congenital nevi, and a personal history of melanoma.
The main environmental risk factor for melanoma is exposure to ultraviolet radiation. “You can break it down in terms of whether this exposure is lifetime, intermittent intense UV exposure, from the use of tanning beds, or due to sunburns during childhood,” Dr. Kent said at the meeting, which was hosted by Scripps MD Anderson Cancer Center. Other environmental risk factors include distance from the equator, having a high socioeconomic status, being immunosuppressed, and exposure to heavy metals, insecticides, or hormones.
In a recently published study, researchers investigated the risk factors associated with first and second primary melanomas in 38,845 patients who were followed in Australia between 2011 and 2018. During a median follow-up of 7.4 years, 1,212 patients (3.1%) had a single primary melanoma diagnosis and 245 (0.6%) had a secondary primary melanoma diagnosis. The researchers found that second melanomas were more likely than were first melanomas to be in situ; for invasive tumors, second melanomas were more likely to be thin (defined as 1 mm or less) than were first melanomas.
In addition, having many self-reported moles at age 21 years was more strongly associated with second melanomas compared with first melanomas (hazard ratio [HR], 6.36 vs. 3.46, respectively; P = .01), as was having a high genetic predisposition (HR, 3.28 vs. 2.06; P = .03).
Second melanomas were also more strongly associated with a history of multiple skin cancer excisions than were first melanomas (HR, 2.63 vs. 1.86; P = .05). “Interestingly, there were no differences in UV exposure between the first primary and second primary melanoma groups,” said Dr. Kent, who was not involved with the study.
He noted that while sunscreen use protects against melanoma, a National Ambulatory Medical Care Survey (NAMCS) found that internists and pediatricians mentioned sunscreen at fewer than 0.1% of visits – even those with patients who have a diagnosis of skin disease. “Physicians need to do better,” he said. “We as dermatologists have work to do to help educate them.”
Dr. Kent reported having no relevant disclosures.
SAN DIEGO – , following cancer of the colorectal area, lung and bronchus, prostate, and breast.
“The incidence of melanoma seems to have continued to go up since the early 1990s,” David E. Kent, MD, a dermatologist who practices in Macon, Ga., said at the annual Cutaneous Malignancy Update. “The death rates have been flat and may have slightly decreased.”
In 2023, the ACS estimates that about 97,610 new melanomas will be diagnosed in the United States (58,120 men and 39,490 women), and about 7,990 people are expected to die of melanoma (5,420 men and 2,570 women). In addition, ACS data from 2017-2019 project that about 2.1% of men and women will be diagnosed with cutaneous melanoma in their lifetime. To date, more than 1.3 million people in the United States live with cutaneous melanoma, and the overall 5-year survival is 93.7%.
Epidemiologic studies show an increase in melanoma incidence, primarily among White populations. “This is believed to be due primarily to sun exposure and to changing recreational behaviors and tanning bed exposures,” said Dr. Kent, who holds a faculty position in the department of dermatology at the Medical College of Georgia, Augusta. Increased surveillance and diagnosis also play a role. In the medical literature, annual increases in melanoma incidence vary from 3% to 7% per year, “which translates into a doubling of rates every 10-20 years,” he said, noting that annual melanoma costs are approximately $3.3 billion.
While incidence rates are lower in non-White, non-Hispanic populations, poor outcomes are disproportionately higher in persons of color. Blacks present at diagnosis with more advanced stage disease and are 1.5 times more likely to die from melanoma, he said, while Hispanics are 2.4 times more likely to present with stage III disease and 3.6 times more likely to have distant metastases. Persons of color also have higher rates of mucosal, acral lentiginous, and subungual melanoma.
Known genetic risk factors for melanoma include having skin types I and II, particularly those with light hair, light eyes, and freckling, and those with a family history have a twofold increased risk. Also, up to 40% of genetic cases are from inherited mutations in CDKN2A, CDK4, BAP1, and MCR1. Other genetic-related risk factors include the number and size of nevi, having atypical nevus syndrome, DNA repair defects, large congenital nevi, and a personal history of melanoma.
The main environmental risk factor for melanoma is exposure to ultraviolet radiation. “You can break it down in terms of whether this exposure is lifetime, intermittent intense UV exposure, from the use of tanning beds, or due to sunburns during childhood,” Dr. Kent said at the meeting, which was hosted by Scripps MD Anderson Cancer Center. Other environmental risk factors include distance from the equator, having a high socioeconomic status, being immunosuppressed, and exposure to heavy metals, insecticides, or hormones.
In a recently published study, researchers investigated the risk factors associated with first and second primary melanomas in 38,845 patients who were followed in Australia between 2011 and 2018. During a median follow-up of 7.4 years, 1,212 patients (3.1%) had a single primary melanoma diagnosis and 245 (0.6%) had a secondary primary melanoma diagnosis. The researchers found that second melanomas were more likely than were first melanomas to be in situ; for invasive tumors, second melanomas were more likely to be thin (defined as 1 mm or less) than were first melanomas.
In addition, having many self-reported moles at age 21 years was more strongly associated with second melanomas compared with first melanomas (hazard ratio [HR], 6.36 vs. 3.46, respectively; P = .01), as was having a high genetic predisposition (HR, 3.28 vs. 2.06; P = .03).
Second melanomas were also more strongly associated with a history of multiple skin cancer excisions than were first melanomas (HR, 2.63 vs. 1.86; P = .05). “Interestingly, there were no differences in UV exposure between the first primary and second primary melanoma groups,” said Dr. Kent, who was not involved with the study.
He noted that while sunscreen use protects against melanoma, a National Ambulatory Medical Care Survey (NAMCS) found that internists and pediatricians mentioned sunscreen at fewer than 0.1% of visits – even those with patients who have a diagnosis of skin disease. “Physicians need to do better,” he said. “We as dermatologists have work to do to help educate them.”
Dr. Kent reported having no relevant disclosures.
SAN DIEGO – , following cancer of the colorectal area, lung and bronchus, prostate, and breast.
“The incidence of melanoma seems to have continued to go up since the early 1990s,” David E. Kent, MD, a dermatologist who practices in Macon, Ga., said at the annual Cutaneous Malignancy Update. “The death rates have been flat and may have slightly decreased.”
In 2023, the ACS estimates that about 97,610 new melanomas will be diagnosed in the United States (58,120 men and 39,490 women), and about 7,990 people are expected to die of melanoma (5,420 men and 2,570 women). In addition, ACS data from 2017-2019 project that about 2.1% of men and women will be diagnosed with cutaneous melanoma in their lifetime. To date, more than 1.3 million people in the United States live with cutaneous melanoma, and the overall 5-year survival is 93.7%.
Epidemiologic studies show an increase in melanoma incidence, primarily among White populations. “This is believed to be due primarily to sun exposure and to changing recreational behaviors and tanning bed exposures,” said Dr. Kent, who holds a faculty position in the department of dermatology at the Medical College of Georgia, Augusta. Increased surveillance and diagnosis also play a role. In the medical literature, annual increases in melanoma incidence vary from 3% to 7% per year, “which translates into a doubling of rates every 10-20 years,” he said, noting that annual melanoma costs are approximately $3.3 billion.
While incidence rates are lower in non-White, non-Hispanic populations, poor outcomes are disproportionately higher in persons of color. Blacks present at diagnosis with more advanced stage disease and are 1.5 times more likely to die from melanoma, he said, while Hispanics are 2.4 times more likely to present with stage III disease and 3.6 times more likely to have distant metastases. Persons of color also have higher rates of mucosal, acral lentiginous, and subungual melanoma.
Known genetic risk factors for melanoma include having skin types I and II, particularly those with light hair, light eyes, and freckling, and those with a family history have a twofold increased risk. Also, up to 40% of genetic cases are from inherited mutations in CDKN2A, CDK4, BAP1, and MCR1. Other genetic-related risk factors include the number and size of nevi, having atypical nevus syndrome, DNA repair defects, large congenital nevi, and a personal history of melanoma.
The main environmental risk factor for melanoma is exposure to ultraviolet radiation. “You can break it down in terms of whether this exposure is lifetime, intermittent intense UV exposure, from the use of tanning beds, or due to sunburns during childhood,” Dr. Kent said at the meeting, which was hosted by Scripps MD Anderson Cancer Center. Other environmental risk factors include distance from the equator, having a high socioeconomic status, being immunosuppressed, and exposure to heavy metals, insecticides, or hormones.
In a recently published study, researchers investigated the risk factors associated with first and second primary melanomas in 38,845 patients who were followed in Australia between 2011 and 2018. During a median follow-up of 7.4 years, 1,212 patients (3.1%) had a single primary melanoma diagnosis and 245 (0.6%) had a secondary primary melanoma diagnosis. The researchers found that second melanomas were more likely than were first melanomas to be in situ; for invasive tumors, second melanomas were more likely to be thin (defined as 1 mm or less) than were first melanomas.
In addition, having many self-reported moles at age 21 years was more strongly associated with second melanomas compared with first melanomas (hazard ratio [HR], 6.36 vs. 3.46, respectively; P = .01), as was having a high genetic predisposition (HR, 3.28 vs. 2.06; P = .03).
Second melanomas were also more strongly associated with a history of multiple skin cancer excisions than were first melanomas (HR, 2.63 vs. 1.86; P = .05). “Interestingly, there were no differences in UV exposure between the first primary and second primary melanoma groups,” said Dr. Kent, who was not involved with the study.
He noted that while sunscreen use protects against melanoma, a National Ambulatory Medical Care Survey (NAMCS) found that internists and pediatricians mentioned sunscreen at fewer than 0.1% of visits – even those with patients who have a diagnosis of skin disease. “Physicians need to do better,” he said. “We as dermatologists have work to do to help educate them.”
Dr. Kent reported having no relevant disclosures.
AT MELANOMA 2023
‘Valid option’ for partial breast irradiation in breast cancer
The study covered in this summary was published on researchsquare.com as a preprint and has not yet been peer reviewed.
Key takeaway
Why this matters
- According to numerous guidelines, partial breast irradiation after lumpectomy is a sound approach for early-stage breast cancer, but there is a lack of consensus about treatment schedules.
- The investigators suggest that 30 Gy in five daily fractions is a “valid option” for these patients in a field that lacks consensus.
Study design
- The team reviewed 381 women with early breast cancer treated with this approach (30 Gy in five daily fractions) at their center from 2013 to 2022.
- Half of patients had left-sided tumors, 94.5% had invasive ductal carcinomas, 96.6% had grade 1 or grade 2 disease, and tumors were luminal like in 99.2% of patients.
- Following lumpectomy, women underwent partial breast irradiation to the tumor bed plus 15 mm of isometric expansion beyond it.
- Follow-up was a median of 28 months.
Key results
- Seven patients (2%) had a local recurrence, of which two were in the treatment field.
- Three-year local control, disease-free survival, and overall survival were high (97.5%, 95.7%, and 96.9%, respectively).
- Nearly 90% of patients and 97% of physicians reported good or excellent cosmesis.
- Ten patients (2.9%) had grade 2 late toxicities, including edema, asthenia, and fibrosis; there were no grade 3 or higher adverse events.
- Five patients (1.5%) had late cardiac major events, four of whom were treated on the right breast; three patients (0.9%) had late pulmonary fibrosis.
- The safety and efficacy outcomes are in line with previous reports, including those that used different dosage and/or fractionation schedules.
Limitations
- The study was retrospective, with a relatively short follow-up.
- Quality of life was not assessed.
- There was no objective baseline measure of cosmesis against which to compare cosmetic results.
Disclosures
- There was no funding for the study, and the investigators didn’t have any conflicts of interest to report.
This is a summary of a preprint research study, “One-Week External Beam Partial Breast Irradiation: Survival and Toxicity Outcomes,” led by Riccardo Ray Colciago from the Fondazione IRCCS Istituto Nazionale dei Tumori, Milan. The study has not been peer reviewed. The full text can be found at researchsquare.com.
A version of this article first appeared on Medscape.com.
The study covered in this summary was published on researchsquare.com as a preprint and has not yet been peer reviewed.
Key takeaway
Why this matters
- According to numerous guidelines, partial breast irradiation after lumpectomy is a sound approach for early-stage breast cancer, but there is a lack of consensus about treatment schedules.
- The investigators suggest that 30 Gy in five daily fractions is a “valid option” for these patients in a field that lacks consensus.
Study design
- The team reviewed 381 women with early breast cancer treated with this approach (30 Gy in five daily fractions) at their center from 2013 to 2022.
- Half of patients had left-sided tumors, 94.5% had invasive ductal carcinomas, 96.6% had grade 1 or grade 2 disease, and tumors were luminal like in 99.2% of patients.
- Following lumpectomy, women underwent partial breast irradiation to the tumor bed plus 15 mm of isometric expansion beyond it.
- Follow-up was a median of 28 months.
Key results
- Seven patients (2%) had a local recurrence, of which two were in the treatment field.
- Three-year local control, disease-free survival, and overall survival were high (97.5%, 95.7%, and 96.9%, respectively).
- Nearly 90% of patients and 97% of physicians reported good or excellent cosmesis.
- Ten patients (2.9%) had grade 2 late toxicities, including edema, asthenia, and fibrosis; there were no grade 3 or higher adverse events.
- Five patients (1.5%) had late cardiac major events, four of whom were treated on the right breast; three patients (0.9%) had late pulmonary fibrosis.
- The safety and efficacy outcomes are in line with previous reports, including those that used different dosage and/or fractionation schedules.
Limitations
- The study was retrospective, with a relatively short follow-up.
- Quality of life was not assessed.
- There was no objective baseline measure of cosmesis against which to compare cosmetic results.
Disclosures
- There was no funding for the study, and the investigators didn’t have any conflicts of interest to report.
This is a summary of a preprint research study, “One-Week External Beam Partial Breast Irradiation: Survival and Toxicity Outcomes,” led by Riccardo Ray Colciago from the Fondazione IRCCS Istituto Nazionale dei Tumori, Milan. The study has not been peer reviewed. The full text can be found at researchsquare.com.
A version of this article first appeared on Medscape.com.
The study covered in this summary was published on researchsquare.com as a preprint and has not yet been peer reviewed.
Key takeaway
Why this matters
- According to numerous guidelines, partial breast irradiation after lumpectomy is a sound approach for early-stage breast cancer, but there is a lack of consensus about treatment schedules.
- The investigators suggest that 30 Gy in five daily fractions is a “valid option” for these patients in a field that lacks consensus.
Study design
- The team reviewed 381 women with early breast cancer treated with this approach (30 Gy in five daily fractions) at their center from 2013 to 2022.
- Half of patients had left-sided tumors, 94.5% had invasive ductal carcinomas, 96.6% had grade 1 or grade 2 disease, and tumors were luminal like in 99.2% of patients.
- Following lumpectomy, women underwent partial breast irradiation to the tumor bed plus 15 mm of isometric expansion beyond it.
- Follow-up was a median of 28 months.
Key results
- Seven patients (2%) had a local recurrence, of which two were in the treatment field.
- Three-year local control, disease-free survival, and overall survival were high (97.5%, 95.7%, and 96.9%, respectively).
- Nearly 90% of patients and 97% of physicians reported good or excellent cosmesis.
- Ten patients (2.9%) had grade 2 late toxicities, including edema, asthenia, and fibrosis; there were no grade 3 or higher adverse events.
- Five patients (1.5%) had late cardiac major events, four of whom were treated on the right breast; three patients (0.9%) had late pulmonary fibrosis.
- The safety and efficacy outcomes are in line with previous reports, including those that used different dosage and/or fractionation schedules.
Limitations
- The study was retrospective, with a relatively short follow-up.
- Quality of life was not assessed.
- There was no objective baseline measure of cosmesis against which to compare cosmetic results.
Disclosures
- There was no funding for the study, and the investigators didn’t have any conflicts of interest to report.
This is a summary of a preprint research study, “One-Week External Beam Partial Breast Irradiation: Survival and Toxicity Outcomes,” led by Riccardo Ray Colciago from the Fondazione IRCCS Istituto Nazionale dei Tumori, Milan. The study has not been peer reviewed. The full text can be found at researchsquare.com.
A version of this article first appeared on Medscape.com.
Radiotherapy for early breast cancer: Sharp cutoff at age 70
say researchers reporting new data showing a sharp cut-off at age 70.
“In our study, one of the most significant variables in determining whether breast cancer patients who are close their 70th birthday are recommended standard-of-care radiation or de-escalated treatment is whether they show up a few months before or a few months after that 70th birthday,” commented study author Wesley J. Talcott, MD, of the department of therapeutic radiology at the Yale School of Medicine, New Haven, Conn.
The results show a trend in which radiation therapy is 50% less likely to be prescribed for patients age 70 and older with early-stage breast cancer, even when controlling for population size, patient demographics, and disease specific variables.
This suggests that oncologists are weighing the variable of age too heavily when deciding on adjuvant treatments, the authors suggest.
“In certain circumstances, breast cancer oncology providers are treating age like a binary categorical variable when selecting patients for treatments or diagnostic procedures, rather than the continuous variable that it is,” Dr. Talcott commented.
The study was published online in the International Journal of Radiation Oncology: Biology, Physics.
Approached for comment, Casey Chollet-Lipscomb, MD, radiation oncologist with Tennessee Oncology, Nashville, who was not associated with the study, agreed with its main finding.
“The study helps emphasize the importance of individualized care,” she said. “Increasing age is the most common risk factor for breast cancer, but breast cancer is an incredibly diverse disease. While you can observe trends based on age, every patient is unique, and they can’t be lumped into one bucket and prescribed treatment based on a strict age cutoff.”
The retrospective study included two cohorts of women identified in the National Cancer Data Base (2004-2017) all of whom underwent lumpectomy for early-stage breast cancer. All patients had “strong indications” for adjuvant treatment.
Patients in cohort 1 (n = 160,990) included women with estrogen-receptor negative cancer, tumor size greater than 3 cm, who were determined to be “appropriate” for radiation therapy.
Patients in cohort 2 (n = 394,946) had hormone-receptor positive cancer, tumor size greater than 5 mm, and were considered to be “appropriate” candidates for endocrine therapy.
Multivariable analysis was performed to control for comorbidity burden (measured by the Charlson-Deyo Comorbidity Index), race and ethnicity, insurance status, academic versus non-academic treatment center, median annual income of a patient’s area of residence, distance from the site of treatment, and pathology variables including number of lymph nodes sampled, histologic grade, and genomic risk score.
In cohort 1, radiation was recommended for 90%-92% of patients between the ages of 50-69; this dropped to 81% for those aged 70.
After MVA, it was determined that age difference was an independent predictor for adjuvant radiation recommendation only at age 70 versus 69 (odds ratio, 0.47; 95% confidence interval 0.39-0.57, P < .001).
For cohort 2, year-over-year age difference predicted endocrine therapy recommendation only at the juncture between age 70 versus 69 (OR, 0.86, 95% CI 0.74-0.99, P = .001).
“Our results don’t say that we should be increasing the amount of treatment for patients over the age [of] 70 or decreasing that patient treatment for patients younger than age 70. What we believe is that we need to be assessing physiologic age of our patients when treating patients,” Dr. Talcott said.
“We would do this by looking at not just how many years a patient has been on this Earth but also what their current health status is, how many good quality-of-life years they might have after treatment or without it, and what the patient wants in terms of burden of treatment. This is a much more valuable way to approach the allocation of treatments than using age alone,” he added.
Both Dr. Talcott and Dr. Chollet-Lipscomb agreed that a limitation of the study was a lack of data on how physicians decided on a specific treatment in each individual case, but they agree that even without this information the results were “significant.”
Dr. Chollet-Lipscomb also highlighted the factors other than age she would use to determine the best adjuvant treatment for a patient with early stage breast cancer, including the individual features of the tumor, how aggressive it looks under the microscope, what the receptor status is, and a patient’s overall performance status and comorbidities.
Dr. Talcott and Dr. Chollet-Lipscomb report no relevant financial relationships. The authors had no acknowledgement of research support for this study.
A version of this article first appeared on Medscape.com.
say researchers reporting new data showing a sharp cut-off at age 70.
“In our study, one of the most significant variables in determining whether breast cancer patients who are close their 70th birthday are recommended standard-of-care radiation or de-escalated treatment is whether they show up a few months before or a few months after that 70th birthday,” commented study author Wesley J. Talcott, MD, of the department of therapeutic radiology at the Yale School of Medicine, New Haven, Conn.
The results show a trend in which radiation therapy is 50% less likely to be prescribed for patients age 70 and older with early-stage breast cancer, even when controlling for population size, patient demographics, and disease specific variables.
This suggests that oncologists are weighing the variable of age too heavily when deciding on adjuvant treatments, the authors suggest.
“In certain circumstances, breast cancer oncology providers are treating age like a binary categorical variable when selecting patients for treatments or diagnostic procedures, rather than the continuous variable that it is,” Dr. Talcott commented.
The study was published online in the International Journal of Radiation Oncology: Biology, Physics.
Approached for comment, Casey Chollet-Lipscomb, MD, radiation oncologist with Tennessee Oncology, Nashville, who was not associated with the study, agreed with its main finding.
“The study helps emphasize the importance of individualized care,” she said. “Increasing age is the most common risk factor for breast cancer, but breast cancer is an incredibly diverse disease. While you can observe trends based on age, every patient is unique, and they can’t be lumped into one bucket and prescribed treatment based on a strict age cutoff.”
The retrospective study included two cohorts of women identified in the National Cancer Data Base (2004-2017) all of whom underwent lumpectomy for early-stage breast cancer. All patients had “strong indications” for adjuvant treatment.
Patients in cohort 1 (n = 160,990) included women with estrogen-receptor negative cancer, tumor size greater than 3 cm, who were determined to be “appropriate” for radiation therapy.
Patients in cohort 2 (n = 394,946) had hormone-receptor positive cancer, tumor size greater than 5 mm, and were considered to be “appropriate” candidates for endocrine therapy.
Multivariable analysis was performed to control for comorbidity burden (measured by the Charlson-Deyo Comorbidity Index), race and ethnicity, insurance status, academic versus non-academic treatment center, median annual income of a patient’s area of residence, distance from the site of treatment, and pathology variables including number of lymph nodes sampled, histologic grade, and genomic risk score.
In cohort 1, radiation was recommended for 90%-92% of patients between the ages of 50-69; this dropped to 81% for those aged 70.
After MVA, it was determined that age difference was an independent predictor for adjuvant radiation recommendation only at age 70 versus 69 (odds ratio, 0.47; 95% confidence interval 0.39-0.57, P < .001).
For cohort 2, year-over-year age difference predicted endocrine therapy recommendation only at the juncture between age 70 versus 69 (OR, 0.86, 95% CI 0.74-0.99, P = .001).
“Our results don’t say that we should be increasing the amount of treatment for patients over the age [of] 70 or decreasing that patient treatment for patients younger than age 70. What we believe is that we need to be assessing physiologic age of our patients when treating patients,” Dr. Talcott said.
“We would do this by looking at not just how many years a patient has been on this Earth but also what their current health status is, how many good quality-of-life years they might have after treatment or without it, and what the patient wants in terms of burden of treatment. This is a much more valuable way to approach the allocation of treatments than using age alone,” he added.
Both Dr. Talcott and Dr. Chollet-Lipscomb agreed that a limitation of the study was a lack of data on how physicians decided on a specific treatment in each individual case, but they agree that even without this information the results were “significant.”
Dr. Chollet-Lipscomb also highlighted the factors other than age she would use to determine the best adjuvant treatment for a patient with early stage breast cancer, including the individual features of the tumor, how aggressive it looks under the microscope, what the receptor status is, and a patient’s overall performance status and comorbidities.
Dr. Talcott and Dr. Chollet-Lipscomb report no relevant financial relationships. The authors had no acknowledgement of research support for this study.
A version of this article first appeared on Medscape.com.
say researchers reporting new data showing a sharp cut-off at age 70.
“In our study, one of the most significant variables in determining whether breast cancer patients who are close their 70th birthday are recommended standard-of-care radiation or de-escalated treatment is whether they show up a few months before or a few months after that 70th birthday,” commented study author Wesley J. Talcott, MD, of the department of therapeutic radiology at the Yale School of Medicine, New Haven, Conn.
The results show a trend in which radiation therapy is 50% less likely to be prescribed for patients age 70 and older with early-stage breast cancer, even when controlling for population size, patient demographics, and disease specific variables.
This suggests that oncologists are weighing the variable of age too heavily when deciding on adjuvant treatments, the authors suggest.
“In certain circumstances, breast cancer oncology providers are treating age like a binary categorical variable when selecting patients for treatments or diagnostic procedures, rather than the continuous variable that it is,” Dr. Talcott commented.
The study was published online in the International Journal of Radiation Oncology: Biology, Physics.
Approached for comment, Casey Chollet-Lipscomb, MD, radiation oncologist with Tennessee Oncology, Nashville, who was not associated with the study, agreed with its main finding.
“The study helps emphasize the importance of individualized care,” she said. “Increasing age is the most common risk factor for breast cancer, but breast cancer is an incredibly diverse disease. While you can observe trends based on age, every patient is unique, and they can’t be lumped into one bucket and prescribed treatment based on a strict age cutoff.”
The retrospective study included two cohorts of women identified in the National Cancer Data Base (2004-2017) all of whom underwent lumpectomy for early-stage breast cancer. All patients had “strong indications” for adjuvant treatment.
Patients in cohort 1 (n = 160,990) included women with estrogen-receptor negative cancer, tumor size greater than 3 cm, who were determined to be “appropriate” for radiation therapy.
Patients in cohort 2 (n = 394,946) had hormone-receptor positive cancer, tumor size greater than 5 mm, and were considered to be “appropriate” candidates for endocrine therapy.
Multivariable analysis was performed to control for comorbidity burden (measured by the Charlson-Deyo Comorbidity Index), race and ethnicity, insurance status, academic versus non-academic treatment center, median annual income of a patient’s area of residence, distance from the site of treatment, and pathology variables including number of lymph nodes sampled, histologic grade, and genomic risk score.
In cohort 1, radiation was recommended for 90%-92% of patients between the ages of 50-69; this dropped to 81% for those aged 70.
After MVA, it was determined that age difference was an independent predictor for adjuvant radiation recommendation only at age 70 versus 69 (odds ratio, 0.47; 95% confidence interval 0.39-0.57, P < .001).
For cohort 2, year-over-year age difference predicted endocrine therapy recommendation only at the juncture between age 70 versus 69 (OR, 0.86, 95% CI 0.74-0.99, P = .001).
“Our results don’t say that we should be increasing the amount of treatment for patients over the age [of] 70 or decreasing that patient treatment for patients younger than age 70. What we believe is that we need to be assessing physiologic age of our patients when treating patients,” Dr. Talcott said.
“We would do this by looking at not just how many years a patient has been on this Earth but also what their current health status is, how many good quality-of-life years they might have after treatment or without it, and what the patient wants in terms of burden of treatment. This is a much more valuable way to approach the allocation of treatments than using age alone,” he added.
Both Dr. Talcott and Dr. Chollet-Lipscomb agreed that a limitation of the study was a lack of data on how physicians decided on a specific treatment in each individual case, but they agree that even without this information the results were “significant.”
Dr. Chollet-Lipscomb also highlighted the factors other than age she would use to determine the best adjuvant treatment for a patient with early stage breast cancer, including the individual features of the tumor, how aggressive it looks under the microscope, what the receptor status is, and a patient’s overall performance status and comorbidities.
Dr. Talcott and Dr. Chollet-Lipscomb report no relevant financial relationships. The authors had no acknowledgement of research support for this study.
A version of this article first appeared on Medscape.com.
FROM THE INTERNATIONAL JOURNAL OF RADIATION ONCOLOGY: BIOLOGY, PHYSICS
Less invasive NSCLC surgery does not compromise survival
suggest results from the CALGB 140503 trial, although strict patient selection remains key.
These new results contrast with those from a previous study from 1995, which found that local recurrence was three times higher and cancer mortality was twice as high with the less invasive procedure.
Those results from nearly 30 years ago established lobectomy as the standard of surgical care in this patient population, but since then advances in imaging and staging have allowed the detection of smaller and earlier tumors, which has “rekindled interest in sublobar resection,” the authors comment.
Hence, they conducted the new trial, which involved almost 700 U.S. patients with clinical T1aN0 NSCLC and a tumor size up to 2 cm, who were randomly assigned to lobar or sublobar tumor resection, and followed for 7 years.
The rates of both disease-free and overall survival were similar between the two groups, with no significant differences observed. There were also no substantial differences in rates of distant and locoregional recurrence.
In addition, there was a suggestion of less reduction in pulmonary function following the less invasive procedure.
“These findings affirm that sublobar resection ... is an effective management approach for this subgroup of patients with NSCLC,” says lead author Nasser Altorki, MD, Weill Cornell Medicine, NewYork–Presbyterian Hospital, New York.
“It is important that these results are interpreted strictly within the constraints of the eligibility criteria mandated by the trial, he emphasizes. “Specifically, the results are applicable only to a highly selected group of patients ... in whom the absence of metastases to hilar and mediastinal lymph nodes is pathologically confirmed.”
Nevertheless, Dr. Altorki said that “these results will become increasingly relevant as the proportion of patients with early-stage lung cancer increases with expanded implementation of lung cancer screening, and as the number of older persons with early-stage disease in whom sublobar resection may be the preferred surgical option increases.”
The study was published online in the New England Journal of Medicine.
In an accompanying editorial, Valerie W. Rusch, MD, Thoracic Service, Memorial Sloan Kettering Cancer Center, New York, agrees. “As CT screening becomes more widespread, this patient population will increase in clinical practice,” she explains.
However, Dr. Rusch also urges caution around patient selection, underlining that the results do not “provide a license for suboptimal surgical care.”
She says that “safeguards” such as the meticulous and strict patient criteria used in the trial “must be preserved in routine practice.”
“Thoracic surgeons will need to expand their expertise in sublobar resections, especially complex segmentectomies, and will need to collaborate closely with pathologists in assessing margins of resection, adequacy of lymph-node staging, and tumor characteristics that may predict recurrence.”
While emphasizing that lobectomy should still be performed when appropriate, Dr. Rusch nevertheless says: “The era of ‘precision’ surgery for NSCLC has arrived.”
Consistent with Japanese results
The investigators also point out that their findings are “consistent” with those of a recent Japanese study that compared lobectomy with anatomical segmentectomy, which found that the 5-year overall survival was 91.1% for lobectomy and 94.3% for segmentectomy.
The authors suggest that the difference in overall survival rates between the two trials might be due to anatomical segmentectomy being “considered by most surgeons to be more oncologically sound than wedge resection.”
In the current trial, wedge resection was allowed, however, “because it is the most frequently practiced method of sublobar resection in North America and Europe; thus, its inclusion would make the trial more representative of a ‘real world’ setting.”
Another important difference could be that more than 90% of the patients in the Japanese trial had adenocarcinoma, 45% with an associated ground-glass component, which is associated with better survival than a completely solid adenocarcinoma.
Dr. Rusch agrees that there are likely to be various factors related to the survival differences between the two trials, including patient selection, intraoperative management, and tumor characteristics.
“However, these two landmark trials are practice-changing because they establish sublobar resection as the standard of care for a select group of patients with NSCLC,” Dr. Rusch concluded.
Study details
Dr. Altorki and colleagues conducted the multicenter, international, randomized, noninferiority, phase 3 trial in patients with clinically staged T1aN0 NSCLC from 83 academic and community-based institutions in the United States, Canada, and Australia.
Patients were required to have a peripheral lung nodule with a solid component of up to 2 cm on preoperative CT, a tumor center in the outer third of the lung, and a tumor location amenable to sublobar resection, whether wedge or segment, or lobar resection, among other criteria.
In all, 697 patients were randomly assigned to undergo either lobar resection or sublobar resection, of whom 59.1% had wedge resection and 37.9% anatomical segmental resection. The median age was 67.9 years, and 57.4% were female. The vast majority (90%) were White.
After a median follow-up of 7 years, the 5-year disease-free survival was 63.6% with sublobar resection and 64.1% following lobar resection.
The team found that sublobar resection was not inferior to lobectomy for disease-free survival, at a hazard ratio for disease recurrence or death of 1.01 (90% confidence interval, 0.83-1.24), which adjusted to 0.99 after taking into account the site where the patient was treated.
The 5-year overall survival rate was 80.3% after sublobar resection, and 78.9% following lobar resection, at a hazard ratio for death of 0.95 (95% CI, 0.72-1.26).
The results were “generally consistent” when accounting for factors such as age group, sex, tumor location, histologic type, smoking history, tumor size, and ECOG performance status, the team says.
Turning to recurrence, they showed that, among 687 patients eligible for assessment, 30.4% of those in the sublobar resection group and 29.3% of those assigned to lobar resection experienced disease recurrence, with 13.4% and 10%, respectively, having locoregional recurrence.
An exploratory analysis indicated that 5-year recurrence-free survival was similar in the two groups, at 70.2% vs. 71.2% or a hazard ratio for recurrence of 1.05 (95% CI, 0.80-1.39). The cumulative incidence of death was also similar.
It was also notable that reduction in predictive forced expiratory volume in 1 second from baseline was lower with sublobar than lobar resection, at –4.0 vs. –6.0, as was the reduction in predicted forced vital capacity, at –3.0 vs. –5.0.
“Although this difference is arguably not clinically meaningful in this patient population with normal baseline pulmonary functions,” the team writes, “it may be more clinically relevant in patients with compromised pulmonary functions, or in those with lower-lobe disease in whom lobar resection may be associated with greater impairment of pulmonary function.”
Dr. Rusch suggests that “more sensitive or functional assessments” of pulmonary function might include “diffusion capacity and 6-minute walk tests,” although she noted that even short-term differences in pulmonary function “may affect perioperative and functional outcomes, especially for tumors in the lower lobe.”
The study was supported by the National Cancer Institute of the National Institutes of Health, including via grants to the Alliance for Clinical Trials in Oncology and the Canadian Cancer Trials Group, and supported in part by Covidien and Ethicon.
Dr. Altorki reports relationships with AstraZeneca, Genentech, Johnson & Johnson, and Regeneron. Dr. Rusch reports relationships with Cancer Research UK, Genentech, and the National Cancer Institute.
A version of this article first appeared on Medscape.com.
suggest results from the CALGB 140503 trial, although strict patient selection remains key.
These new results contrast with those from a previous study from 1995, which found that local recurrence was three times higher and cancer mortality was twice as high with the less invasive procedure.
Those results from nearly 30 years ago established lobectomy as the standard of surgical care in this patient population, but since then advances in imaging and staging have allowed the detection of smaller and earlier tumors, which has “rekindled interest in sublobar resection,” the authors comment.
Hence, they conducted the new trial, which involved almost 700 U.S. patients with clinical T1aN0 NSCLC and a tumor size up to 2 cm, who were randomly assigned to lobar or sublobar tumor resection, and followed for 7 years.
The rates of both disease-free and overall survival were similar between the two groups, with no significant differences observed. There were also no substantial differences in rates of distant and locoregional recurrence.
In addition, there was a suggestion of less reduction in pulmonary function following the less invasive procedure.
“These findings affirm that sublobar resection ... is an effective management approach for this subgroup of patients with NSCLC,” says lead author Nasser Altorki, MD, Weill Cornell Medicine, NewYork–Presbyterian Hospital, New York.
“It is important that these results are interpreted strictly within the constraints of the eligibility criteria mandated by the trial, he emphasizes. “Specifically, the results are applicable only to a highly selected group of patients ... in whom the absence of metastases to hilar and mediastinal lymph nodes is pathologically confirmed.”
Nevertheless, Dr. Altorki said that “these results will become increasingly relevant as the proportion of patients with early-stage lung cancer increases with expanded implementation of lung cancer screening, and as the number of older persons with early-stage disease in whom sublobar resection may be the preferred surgical option increases.”
The study was published online in the New England Journal of Medicine.
In an accompanying editorial, Valerie W. Rusch, MD, Thoracic Service, Memorial Sloan Kettering Cancer Center, New York, agrees. “As CT screening becomes more widespread, this patient population will increase in clinical practice,” she explains.
However, Dr. Rusch also urges caution around patient selection, underlining that the results do not “provide a license for suboptimal surgical care.”
She says that “safeguards” such as the meticulous and strict patient criteria used in the trial “must be preserved in routine practice.”
“Thoracic surgeons will need to expand their expertise in sublobar resections, especially complex segmentectomies, and will need to collaborate closely with pathologists in assessing margins of resection, adequacy of lymph-node staging, and tumor characteristics that may predict recurrence.”
While emphasizing that lobectomy should still be performed when appropriate, Dr. Rusch nevertheless says: “The era of ‘precision’ surgery for NSCLC has arrived.”
Consistent with Japanese results
The investigators also point out that their findings are “consistent” with those of a recent Japanese study that compared lobectomy with anatomical segmentectomy, which found that the 5-year overall survival was 91.1% for lobectomy and 94.3% for segmentectomy.
The authors suggest that the difference in overall survival rates between the two trials might be due to anatomical segmentectomy being “considered by most surgeons to be more oncologically sound than wedge resection.”
In the current trial, wedge resection was allowed, however, “because it is the most frequently practiced method of sublobar resection in North America and Europe; thus, its inclusion would make the trial more representative of a ‘real world’ setting.”
Another important difference could be that more than 90% of the patients in the Japanese trial had adenocarcinoma, 45% with an associated ground-glass component, which is associated with better survival than a completely solid adenocarcinoma.
Dr. Rusch agrees that there are likely to be various factors related to the survival differences between the two trials, including patient selection, intraoperative management, and tumor characteristics.
“However, these two landmark trials are practice-changing because they establish sublobar resection as the standard of care for a select group of patients with NSCLC,” Dr. Rusch concluded.
Study details
Dr. Altorki and colleagues conducted the multicenter, international, randomized, noninferiority, phase 3 trial in patients with clinically staged T1aN0 NSCLC from 83 academic and community-based institutions in the United States, Canada, and Australia.
Patients were required to have a peripheral lung nodule with a solid component of up to 2 cm on preoperative CT, a tumor center in the outer third of the lung, and a tumor location amenable to sublobar resection, whether wedge or segment, or lobar resection, among other criteria.
In all, 697 patients were randomly assigned to undergo either lobar resection or sublobar resection, of whom 59.1% had wedge resection and 37.9% anatomical segmental resection. The median age was 67.9 years, and 57.4% were female. The vast majority (90%) were White.
After a median follow-up of 7 years, the 5-year disease-free survival was 63.6% with sublobar resection and 64.1% following lobar resection.
The team found that sublobar resection was not inferior to lobectomy for disease-free survival, at a hazard ratio for disease recurrence or death of 1.01 (90% confidence interval, 0.83-1.24), which adjusted to 0.99 after taking into account the site where the patient was treated.
The 5-year overall survival rate was 80.3% after sublobar resection, and 78.9% following lobar resection, at a hazard ratio for death of 0.95 (95% CI, 0.72-1.26).
The results were “generally consistent” when accounting for factors such as age group, sex, tumor location, histologic type, smoking history, tumor size, and ECOG performance status, the team says.
Turning to recurrence, they showed that, among 687 patients eligible for assessment, 30.4% of those in the sublobar resection group and 29.3% of those assigned to lobar resection experienced disease recurrence, with 13.4% and 10%, respectively, having locoregional recurrence.
An exploratory analysis indicated that 5-year recurrence-free survival was similar in the two groups, at 70.2% vs. 71.2% or a hazard ratio for recurrence of 1.05 (95% CI, 0.80-1.39). The cumulative incidence of death was also similar.
It was also notable that reduction in predictive forced expiratory volume in 1 second from baseline was lower with sublobar than lobar resection, at –4.0 vs. –6.0, as was the reduction in predicted forced vital capacity, at –3.0 vs. –5.0.
“Although this difference is arguably not clinically meaningful in this patient population with normal baseline pulmonary functions,” the team writes, “it may be more clinically relevant in patients with compromised pulmonary functions, or in those with lower-lobe disease in whom lobar resection may be associated with greater impairment of pulmonary function.”
Dr. Rusch suggests that “more sensitive or functional assessments” of pulmonary function might include “diffusion capacity and 6-minute walk tests,” although she noted that even short-term differences in pulmonary function “may affect perioperative and functional outcomes, especially for tumors in the lower lobe.”
The study was supported by the National Cancer Institute of the National Institutes of Health, including via grants to the Alliance for Clinical Trials in Oncology and the Canadian Cancer Trials Group, and supported in part by Covidien and Ethicon.
Dr. Altorki reports relationships with AstraZeneca, Genentech, Johnson & Johnson, and Regeneron. Dr. Rusch reports relationships with Cancer Research UK, Genentech, and the National Cancer Institute.
A version of this article first appeared on Medscape.com.
suggest results from the CALGB 140503 trial, although strict patient selection remains key.
These new results contrast with those from a previous study from 1995, which found that local recurrence was three times higher and cancer mortality was twice as high with the less invasive procedure.
Those results from nearly 30 years ago established lobectomy as the standard of surgical care in this patient population, but since then advances in imaging and staging have allowed the detection of smaller and earlier tumors, which has “rekindled interest in sublobar resection,” the authors comment.
Hence, they conducted the new trial, which involved almost 700 U.S. patients with clinical T1aN0 NSCLC and a tumor size up to 2 cm, who were randomly assigned to lobar or sublobar tumor resection, and followed for 7 years.
The rates of both disease-free and overall survival were similar between the two groups, with no significant differences observed. There were also no substantial differences in rates of distant and locoregional recurrence.
In addition, there was a suggestion of less reduction in pulmonary function following the less invasive procedure.
“These findings affirm that sublobar resection ... is an effective management approach for this subgroup of patients with NSCLC,” says lead author Nasser Altorki, MD, Weill Cornell Medicine, NewYork–Presbyterian Hospital, New York.
“It is important that these results are interpreted strictly within the constraints of the eligibility criteria mandated by the trial, he emphasizes. “Specifically, the results are applicable only to a highly selected group of patients ... in whom the absence of metastases to hilar and mediastinal lymph nodes is pathologically confirmed.”
Nevertheless, Dr. Altorki said that “these results will become increasingly relevant as the proportion of patients with early-stage lung cancer increases with expanded implementation of lung cancer screening, and as the number of older persons with early-stage disease in whom sublobar resection may be the preferred surgical option increases.”
The study was published online in the New England Journal of Medicine.
In an accompanying editorial, Valerie W. Rusch, MD, Thoracic Service, Memorial Sloan Kettering Cancer Center, New York, agrees. “As CT screening becomes more widespread, this patient population will increase in clinical practice,” she explains.
However, Dr. Rusch also urges caution around patient selection, underlining that the results do not “provide a license for suboptimal surgical care.”
She says that “safeguards” such as the meticulous and strict patient criteria used in the trial “must be preserved in routine practice.”
“Thoracic surgeons will need to expand their expertise in sublobar resections, especially complex segmentectomies, and will need to collaborate closely with pathologists in assessing margins of resection, adequacy of lymph-node staging, and tumor characteristics that may predict recurrence.”
While emphasizing that lobectomy should still be performed when appropriate, Dr. Rusch nevertheless says: “The era of ‘precision’ surgery for NSCLC has arrived.”
Consistent with Japanese results
The investigators also point out that their findings are “consistent” with those of a recent Japanese study that compared lobectomy with anatomical segmentectomy, which found that the 5-year overall survival was 91.1% for lobectomy and 94.3% for segmentectomy.
The authors suggest that the difference in overall survival rates between the two trials might be due to anatomical segmentectomy being “considered by most surgeons to be more oncologically sound than wedge resection.”
In the current trial, wedge resection was allowed, however, “because it is the most frequently practiced method of sublobar resection in North America and Europe; thus, its inclusion would make the trial more representative of a ‘real world’ setting.”
Another important difference could be that more than 90% of the patients in the Japanese trial had adenocarcinoma, 45% with an associated ground-glass component, which is associated with better survival than a completely solid adenocarcinoma.
Dr. Rusch agrees that there are likely to be various factors related to the survival differences between the two trials, including patient selection, intraoperative management, and tumor characteristics.
“However, these two landmark trials are practice-changing because they establish sublobar resection as the standard of care for a select group of patients with NSCLC,” Dr. Rusch concluded.
Study details
Dr. Altorki and colleagues conducted the multicenter, international, randomized, noninferiority, phase 3 trial in patients with clinically staged T1aN0 NSCLC from 83 academic and community-based institutions in the United States, Canada, and Australia.
Patients were required to have a peripheral lung nodule with a solid component of up to 2 cm on preoperative CT, a tumor center in the outer third of the lung, and a tumor location amenable to sublobar resection, whether wedge or segment, or lobar resection, among other criteria.
In all, 697 patients were randomly assigned to undergo either lobar resection or sublobar resection, of whom 59.1% had wedge resection and 37.9% anatomical segmental resection. The median age was 67.9 years, and 57.4% were female. The vast majority (90%) were White.
After a median follow-up of 7 years, the 5-year disease-free survival was 63.6% with sublobar resection and 64.1% following lobar resection.
The team found that sublobar resection was not inferior to lobectomy for disease-free survival, at a hazard ratio for disease recurrence or death of 1.01 (90% confidence interval, 0.83-1.24), which adjusted to 0.99 after taking into account the site where the patient was treated.
The 5-year overall survival rate was 80.3% after sublobar resection, and 78.9% following lobar resection, at a hazard ratio for death of 0.95 (95% CI, 0.72-1.26).
The results were “generally consistent” when accounting for factors such as age group, sex, tumor location, histologic type, smoking history, tumor size, and ECOG performance status, the team says.
Turning to recurrence, they showed that, among 687 patients eligible for assessment, 30.4% of those in the sublobar resection group and 29.3% of those assigned to lobar resection experienced disease recurrence, with 13.4% and 10%, respectively, having locoregional recurrence.
An exploratory analysis indicated that 5-year recurrence-free survival was similar in the two groups, at 70.2% vs. 71.2% or a hazard ratio for recurrence of 1.05 (95% CI, 0.80-1.39). The cumulative incidence of death was also similar.
It was also notable that reduction in predictive forced expiratory volume in 1 second from baseline was lower with sublobar than lobar resection, at –4.0 vs. –6.0, as was the reduction in predicted forced vital capacity, at –3.0 vs. –5.0.
“Although this difference is arguably not clinically meaningful in this patient population with normal baseline pulmonary functions,” the team writes, “it may be more clinically relevant in patients with compromised pulmonary functions, or in those with lower-lobe disease in whom lobar resection may be associated with greater impairment of pulmonary function.”
Dr. Rusch suggests that “more sensitive or functional assessments” of pulmonary function might include “diffusion capacity and 6-minute walk tests,” although she noted that even short-term differences in pulmonary function “may affect perioperative and functional outcomes, especially for tumors in the lower lobe.”
The study was supported by the National Cancer Institute of the National Institutes of Health, including via grants to the Alliance for Clinical Trials in Oncology and the Canadian Cancer Trials Group, and supported in part by Covidien and Ethicon.
Dr. Altorki reports relationships with AstraZeneca, Genentech, Johnson & Johnson, and Regeneron. Dr. Rusch reports relationships with Cancer Research UK, Genentech, and the National Cancer Institute.
A version of this article first appeared on Medscape.com.
FROM NEW ENGLAND JOURNAL OF MEDICINE
Service-Related Toxin Exposure and Acute Myeloid Leukemia in Veterans
Members of the United States military who served in Vietnam between 1961 and 1971 risked exposure to the weaponized chemical defoliant known as Agent Orange. Among the components of Agent Orange, benzene and tetrachlorodibenzo-p-dioxin (TCDD) are known carcinogens linked to several cancers. They include multiple myeloma, Hodgkin and non-Hodgkin lymphoma, as well as bladder, prostate, and lung cancer.
In this ReCAP, Dr Timothy O'Brien, section chief of hematology at the Louis Stokes Cleveland VA Medical Center, examines the evidence that suggests a link between service-related Agent Orange exposure and acute myeloid leukemia (AML). He discusses preclinical models that show a relationship between benzene and TCDD exposure and the development of AML.
Dr O'Brien also explains the factors that have limited researchers' ability to positively connect Agent Orange and AML. For example, there is a dwindling cohort of affected patients to study because dioxins can lie latent in fat cells for more than a decade, delaying the development of AML. During that time, many veterans will have died from unrelated causes.
More research is needed for veterans to receive service-connected benefits for AML diagnoses. However, as Dr O'Brien notes, the PACT Act provides coverage for veterans who developed AML after exposure to benzene-contaminated water at Camp Lejeune.
--
Timothy O'Brien, MD, Associate Professor, Case Western Reserve University School of Medicine; Chief of Hematology, Louis Stokes Cleveland VA Medical Center, Cleveland, Ohio
Timothy O'Brien, MD, has disclosed no relevant financial relationships.
Members of the United States military who served in Vietnam between 1961 and 1971 risked exposure to the weaponized chemical defoliant known as Agent Orange. Among the components of Agent Orange, benzene and tetrachlorodibenzo-p-dioxin (TCDD) are known carcinogens linked to several cancers. They include multiple myeloma, Hodgkin and non-Hodgkin lymphoma, as well as bladder, prostate, and lung cancer.
In this ReCAP, Dr Timothy O'Brien, section chief of hematology at the Louis Stokes Cleveland VA Medical Center, examines the evidence that suggests a link between service-related Agent Orange exposure and acute myeloid leukemia (AML). He discusses preclinical models that show a relationship between benzene and TCDD exposure and the development of AML.
Dr O'Brien also explains the factors that have limited researchers' ability to positively connect Agent Orange and AML. For example, there is a dwindling cohort of affected patients to study because dioxins can lie latent in fat cells for more than a decade, delaying the development of AML. During that time, many veterans will have died from unrelated causes.
More research is needed for veterans to receive service-connected benefits for AML diagnoses. However, as Dr O'Brien notes, the PACT Act provides coverage for veterans who developed AML after exposure to benzene-contaminated water at Camp Lejeune.
--
Timothy O'Brien, MD, Associate Professor, Case Western Reserve University School of Medicine; Chief of Hematology, Louis Stokes Cleveland VA Medical Center, Cleveland, Ohio
Timothy O'Brien, MD, has disclosed no relevant financial relationships.
Members of the United States military who served in Vietnam between 1961 and 1971 risked exposure to the weaponized chemical defoliant known as Agent Orange. Among the components of Agent Orange, benzene and tetrachlorodibenzo-p-dioxin (TCDD) are known carcinogens linked to several cancers. They include multiple myeloma, Hodgkin and non-Hodgkin lymphoma, as well as bladder, prostate, and lung cancer.
In this ReCAP, Dr Timothy O'Brien, section chief of hematology at the Louis Stokes Cleveland VA Medical Center, examines the evidence that suggests a link between service-related Agent Orange exposure and acute myeloid leukemia (AML). He discusses preclinical models that show a relationship between benzene and TCDD exposure and the development of AML.
Dr O'Brien also explains the factors that have limited researchers' ability to positively connect Agent Orange and AML. For example, there is a dwindling cohort of affected patients to study because dioxins can lie latent in fat cells for more than a decade, delaying the development of AML. During that time, many veterans will have died from unrelated causes.
More research is needed for veterans to receive service-connected benefits for AML diagnoses. However, as Dr O'Brien notes, the PACT Act provides coverage for veterans who developed AML after exposure to benzene-contaminated water at Camp Lejeune.
--
Timothy O'Brien, MD, Associate Professor, Case Western Reserve University School of Medicine; Chief of Hematology, Louis Stokes Cleveland VA Medical Center, Cleveland, Ohio
Timothy O'Brien, MD, has disclosed no relevant financial relationships.

Renewed calls for fallopian tube removal to avoid ovarian cancer
All women, regardless of their risk profile, should consider prophylactic removal of the fallopian tubes at the same time as other pelvic surgery once they are finished having children, the Ovarian Cancer Research Alliance has advised.
The recommendation, announced Feb. 1, replaces the decades-old focus on symptom awareness and early detection and follows “sobering and deeply disappointing” results from a large U.K. study published 2 years ago, the organization said.
That was the UK Collaborative Trial of Ovarian Cancer Screening published in The Lancet in 2021, which followed more than 200,000 women for a median 16 years. It showed that screening average-risk women with a CA-125 blood test and ultrasound does not reduce deaths from the disease, as reported at the time by this news organization.
“We all hoped that the trial would show that early detection was effective in changing mortality rates. When the results came out, it was very hard to accept,” Audra Moran, OCRA president and CEO, said in an interview.
“We have an obligation to let people know that symptom awareness and early detection will not save lives” but considering opportunistic salpingectomy “absolutely will,” said Ms. Moran. Hence the renewed call for women to consider having their fallopian tubes removed.
What sounds new about this call is that the group is directing fallopian tube removal to all women “who are undergoing pelvic surgeries for benign conditions,” irrespective of what perceived risk they have of developing ovarian cancer (for example, based on family history).
But this advice has been in place for years for women who are known to be at higher risk for the disease.
For instance, women at high risk for ovarian cancer based on Hereditary Breast and Ovarian Cancer Syndrome (HBOC) have long been recommended to undergo surgery to remove ovaries and fallopian tubes (risk-reducing bilateral salpingo-oophorectomy or RRBSO) once there is no longer a desire for pregnancy.
Approached for comment about the new messaging, Stephanie V. Blank, MD, president of the Society of Gynecologic Oncology, says that the new recommendation – that all women who are finished childbearing consider opportunistic salpingectomy at the time of other pelvic surgery for benign conditions – is “not aggressive.”
“It’s reasonable and makes sense,” Dr. Blank said in an interview.
And she pointed out that it’s actually not “new”; it is, however, getting “new attention” based on the disappointing U.K. screening study, said Dr. Blank, director of gynecologic oncology for the Mount Sinai Health System in New York and professor of gynecologic oncology at Icahn School of Medicine at Mount Sinai.
She noted that the procedure of opportunistic salpingectomy has been endorsed by SGO since 2013 and by the American College of Obstetricians and Gynecologists since 2015.
There is increasing evidence that most high-grade serous ovarian cancers arise from cells in the fallopian tubes, William Dahut, MD, chief scientific officer for the American Cancer Society, told this news organization.
“Indirect evidence suggests a fairly strong degree of risk reduction associated with opportunistic salpingectomy for the most prevalent type of ovarian cancer (serous), and some risk reduction of epithelial ovarian cancer. At this time, these discussions seem warranted,” Dr. Dahut said.
At this point, however, the fact that leading organizations advise “consideration” means that the evidence base has “not been judged to be sufficiently strong (in terms of what we can say about benefits and harms) to advise a direct recommendation for opportunistic salpingectomy,” Dr. Dahut added.
There is no current recommendation to have fallopian tubes removed as a stand-alone procedure, he pointed out. However, he commented that “the occasion of scheduled gynecologic surgery presents an opportunity to possibly reduce the risk of ovarian cancer without known adverse effects in women who have completed childbearing. Having the discussion seems to be justified by the current evidence,” Dr. Dahut said.
Deanna Gerber, MD, a gynecologic oncologist at NYU Langone Perlmutter Cancer Center-Long Island, agrees. “In women who are scheduled to have a gynecologic or pelvic procedure, clinicians should discuss the possibility of removing the fallopian tubes at that time. A salpingectomy is a relatively low-risk procedure and adds little time to the surgery,” Dr. Gerber said in an interview.
“Women should understand that there is still ongoing research on this topic, but this low-risk procedure may reduce their risk of developing an ovarian or fallopian tube cancer,” Dr. Gerber said.
OCRA also encourages all women (or anyone born with ovaries) to know their risk for ovarian cancer. To that end, the organization has launched a pilot program offering free, at-home genetic testing kits to people with a personal or family history of breast, ovarian, uterine, or colorectal cancer.
Ms. Moran, Dr. Blank, Dr. Dahut, and Dr. Gerber report no relevant financial relationships.
A version of this article first appeared on Medscape.com.
All women, regardless of their risk profile, should consider prophylactic removal of the fallopian tubes at the same time as other pelvic surgery once they are finished having children, the Ovarian Cancer Research Alliance has advised.
The recommendation, announced Feb. 1, replaces the decades-old focus on symptom awareness and early detection and follows “sobering and deeply disappointing” results from a large U.K. study published 2 years ago, the organization said.
That was the UK Collaborative Trial of Ovarian Cancer Screening published in The Lancet in 2021, which followed more than 200,000 women for a median 16 years. It showed that screening average-risk women with a CA-125 blood test and ultrasound does not reduce deaths from the disease, as reported at the time by this news organization.
“We all hoped that the trial would show that early detection was effective in changing mortality rates. When the results came out, it was very hard to accept,” Audra Moran, OCRA president and CEO, said in an interview.
“We have an obligation to let people know that symptom awareness and early detection will not save lives” but considering opportunistic salpingectomy “absolutely will,” said Ms. Moran. Hence the renewed call for women to consider having their fallopian tubes removed.
What sounds new about this call is that the group is directing fallopian tube removal to all women “who are undergoing pelvic surgeries for benign conditions,” irrespective of what perceived risk they have of developing ovarian cancer (for example, based on family history).
But this advice has been in place for years for women who are known to be at higher risk for the disease.
For instance, women at high risk for ovarian cancer based on Hereditary Breast and Ovarian Cancer Syndrome (HBOC) have long been recommended to undergo surgery to remove ovaries and fallopian tubes (risk-reducing bilateral salpingo-oophorectomy or RRBSO) once there is no longer a desire for pregnancy.
Approached for comment about the new messaging, Stephanie V. Blank, MD, president of the Society of Gynecologic Oncology, says that the new recommendation – that all women who are finished childbearing consider opportunistic salpingectomy at the time of other pelvic surgery for benign conditions – is “not aggressive.”
“It’s reasonable and makes sense,” Dr. Blank said in an interview.
And she pointed out that it’s actually not “new”; it is, however, getting “new attention” based on the disappointing U.K. screening study, said Dr. Blank, director of gynecologic oncology for the Mount Sinai Health System in New York and professor of gynecologic oncology at Icahn School of Medicine at Mount Sinai.
She noted that the procedure of opportunistic salpingectomy has been endorsed by SGO since 2013 and by the American College of Obstetricians and Gynecologists since 2015.
There is increasing evidence that most high-grade serous ovarian cancers arise from cells in the fallopian tubes, William Dahut, MD, chief scientific officer for the American Cancer Society, told this news organization.
“Indirect evidence suggests a fairly strong degree of risk reduction associated with opportunistic salpingectomy for the most prevalent type of ovarian cancer (serous), and some risk reduction of epithelial ovarian cancer. At this time, these discussions seem warranted,” Dr. Dahut said.
At this point, however, the fact that leading organizations advise “consideration” means that the evidence base has “not been judged to be sufficiently strong (in terms of what we can say about benefits and harms) to advise a direct recommendation for opportunistic salpingectomy,” Dr. Dahut added.
There is no current recommendation to have fallopian tubes removed as a stand-alone procedure, he pointed out. However, he commented that “the occasion of scheduled gynecologic surgery presents an opportunity to possibly reduce the risk of ovarian cancer without known adverse effects in women who have completed childbearing. Having the discussion seems to be justified by the current evidence,” Dr. Dahut said.
Deanna Gerber, MD, a gynecologic oncologist at NYU Langone Perlmutter Cancer Center-Long Island, agrees. “In women who are scheduled to have a gynecologic or pelvic procedure, clinicians should discuss the possibility of removing the fallopian tubes at that time. A salpingectomy is a relatively low-risk procedure and adds little time to the surgery,” Dr. Gerber said in an interview.
“Women should understand that there is still ongoing research on this topic, but this low-risk procedure may reduce their risk of developing an ovarian or fallopian tube cancer,” Dr. Gerber said.
OCRA also encourages all women (or anyone born with ovaries) to know their risk for ovarian cancer. To that end, the organization has launched a pilot program offering free, at-home genetic testing kits to people with a personal or family history of breast, ovarian, uterine, or colorectal cancer.
Ms. Moran, Dr. Blank, Dr. Dahut, and Dr. Gerber report no relevant financial relationships.
A version of this article first appeared on Medscape.com.
All women, regardless of their risk profile, should consider prophylactic removal of the fallopian tubes at the same time as other pelvic surgery once they are finished having children, the Ovarian Cancer Research Alliance has advised.
The recommendation, announced Feb. 1, replaces the decades-old focus on symptom awareness and early detection and follows “sobering and deeply disappointing” results from a large U.K. study published 2 years ago, the organization said.
That was the UK Collaborative Trial of Ovarian Cancer Screening published in The Lancet in 2021, which followed more than 200,000 women for a median 16 years. It showed that screening average-risk women with a CA-125 blood test and ultrasound does not reduce deaths from the disease, as reported at the time by this news organization.
“We all hoped that the trial would show that early detection was effective in changing mortality rates. When the results came out, it was very hard to accept,” Audra Moran, OCRA president and CEO, said in an interview.
“We have an obligation to let people know that symptom awareness and early detection will not save lives” but considering opportunistic salpingectomy “absolutely will,” said Ms. Moran. Hence the renewed call for women to consider having their fallopian tubes removed.
What sounds new about this call is that the group is directing fallopian tube removal to all women “who are undergoing pelvic surgeries for benign conditions,” irrespective of what perceived risk they have of developing ovarian cancer (for example, based on family history).
But this advice has been in place for years for women who are known to be at higher risk for the disease.
For instance, women at high risk for ovarian cancer based on Hereditary Breast and Ovarian Cancer Syndrome (HBOC) have long been recommended to undergo surgery to remove ovaries and fallopian tubes (risk-reducing bilateral salpingo-oophorectomy or RRBSO) once there is no longer a desire for pregnancy.
Approached for comment about the new messaging, Stephanie V. Blank, MD, president of the Society of Gynecologic Oncology, says that the new recommendation – that all women who are finished childbearing consider opportunistic salpingectomy at the time of other pelvic surgery for benign conditions – is “not aggressive.”
“It’s reasonable and makes sense,” Dr. Blank said in an interview.
And she pointed out that it’s actually not “new”; it is, however, getting “new attention” based on the disappointing U.K. screening study, said Dr. Blank, director of gynecologic oncology for the Mount Sinai Health System in New York and professor of gynecologic oncology at Icahn School of Medicine at Mount Sinai.
She noted that the procedure of opportunistic salpingectomy has been endorsed by SGO since 2013 and by the American College of Obstetricians and Gynecologists since 2015.
There is increasing evidence that most high-grade serous ovarian cancers arise from cells in the fallopian tubes, William Dahut, MD, chief scientific officer for the American Cancer Society, told this news organization.
“Indirect evidence suggests a fairly strong degree of risk reduction associated with opportunistic salpingectomy for the most prevalent type of ovarian cancer (serous), and some risk reduction of epithelial ovarian cancer. At this time, these discussions seem warranted,” Dr. Dahut said.
At this point, however, the fact that leading organizations advise “consideration” means that the evidence base has “not been judged to be sufficiently strong (in terms of what we can say about benefits and harms) to advise a direct recommendation for opportunistic salpingectomy,” Dr. Dahut added.
There is no current recommendation to have fallopian tubes removed as a stand-alone procedure, he pointed out. However, he commented that “the occasion of scheduled gynecologic surgery presents an opportunity to possibly reduce the risk of ovarian cancer without known adverse effects in women who have completed childbearing. Having the discussion seems to be justified by the current evidence,” Dr. Dahut said.
Deanna Gerber, MD, a gynecologic oncologist at NYU Langone Perlmutter Cancer Center-Long Island, agrees. “In women who are scheduled to have a gynecologic or pelvic procedure, clinicians should discuss the possibility of removing the fallopian tubes at that time. A salpingectomy is a relatively low-risk procedure and adds little time to the surgery,” Dr. Gerber said in an interview.
“Women should understand that there is still ongoing research on this topic, but this low-risk procedure may reduce their risk of developing an ovarian or fallopian tube cancer,” Dr. Gerber said.
OCRA also encourages all women (or anyone born with ovaries) to know their risk for ovarian cancer. To that end, the organization has launched a pilot program offering free, at-home genetic testing kits to people with a personal or family history of breast, ovarian, uterine, or colorectal cancer.
Ms. Moran, Dr. Blank, Dr. Dahut, and Dr. Gerber report no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Dermoscopy, other modalities for improving melanoma diagnoses reviewed
San Diego – .
“I don’t think that’s going to change in the short term,” Travis W. Blalock, MD, director of dermatologic surgery, Mohs micrographic surgery, and cutaneous oncology at Emory University, Atlanta, said at the annual Cutaneous Malignancy Update. “But I do think we can supplement that with other modalities that will improve the clinical examination and help dermatopathologists as they assess and evaluate these lesions,” he said, adding: “The reality is, histopathology, while it may be the gold standard, is not necessarily a consistently reproducible evaluation. That raises the question: What can we do better?”
According to Dr. Blalock, the future may include more routine use of noninvasive genetic molecular assays to assist with the diagnostics challenges linked to the visual image and pattern recognition approach of detecting cutaneous melanoma. For example, a two-gene classification method based on LINC00518 and preferentially expressed antigen in melanoma (PRAME) gene expression was evaluated and validated in 555 pigmented lesions obtained noninvasively via adhesive patch biopsy.
“Today, you can pick up a kit from your local pharmacy that can tell you a bit about broad genetic susceptibilities,” he said at the meeting, which was hosted by Scripps MD Anderson Cancer Center. He predicted that using adhesive patch biopsies to assess suspicious melanocytic lesions “is likely the wave of the future.” This may increase patient understanding “as to the types of risks they have, the different lesions they have, and minimize invasive disease, but it also will pose different challenges for us when it comes to deploying patient-centered health care. For example, in a patient with multiple different lesions, how are you going to keep track of them all?”
Dermoscopy
In Dr. Blalock’s clinical opinion, dermoscopy improves the sensitivity of human visual detection of melanoma and may allow detection before a lesion displays classical features described with the “ABCDE rule.” However, the learning curve for dermoscopy is steep, he added, and whether the technique should be considered a first-line tool or as a supplement to other methods of examining cutaneous lesions remains a matter of debate.
“Dermoscopy is our version of the stethoscope,” he said. “We need to figure out when we’re going to use it. Should we be using it all of the time or only some of the time? Based on the clinical setting, maybe it’s a personal choice, but this can be a helpful skill and art in your practice if you’re willing to take the time to learn.”
In 2007, the International Dermoscopy Society (IDS) established a proposal for the standardization and recommended criteria necessary to effectively convey dermoscopic findings to consulting physicians and colleagues. The document includes 10 points categorized as either recommended or optional for a standardized dermoscopy report.
“The first step is to assess the lesion to determine whether or not it’s melanocytic in the first place,” said Dr. Blalock. “There are many different features – the mile-high [global features] evaluation of the lesions – then more specific local features that may clue you in to specific diagnoses,” he noted. “Once we get past that first step of determining that a lesion is melanocytic, it’s not enough to stop there, because we don’t want to biopsy every single lesion that’s melanocytic,” so there is a need to determine which ones require intervention, which is where dermoscopy “gets trickier and a little more challenging.”
According to the IDS, a standard dermoscopy report should include the patient’s age, relevant history pertaining to the lesion, pertinent personal and family history (recommended); clinical description of the lesion (recommended); the two-step method of dermoscopy differentiating melanocytic from nonmelanocytic tumors (recommended); and the use of standardized terms to describe structures as defined by the Dermoscopy Consensus Report published in 2003.
For new terms, the document states, “it would be helpful” for the physician to provide a working definition (recommended); the dermoscopic algorithm used should be mentioned (optional); information on the imaging equipment and magnification (recommended); clinical and dermoscopic images of the tumor (recommended); a diagnosis or differential diagnosis (recommended); decision concerning management (recommended), and specific comments for the pathologist when excision and histopathologic examination are recommended (optional).
The 2007 IDS document also includes a proposed seven-point checklist to differentiate between benign and melanocytic lesions on dermoscopy. Three major criteria are worth two points each: The presence of an atypical pigment network, gray-blue areas (commonly known as the veil), and an atypical vascular pattern. Four minor criteria are worth one point each: Irregular streaks, irregular dots/globules, irregular pigmentation, and regression structures. A minimum total score of 3 is required to establish a diagnosis of melanoma.
Another diagnostic technique, digital mole mapping, involves the use of photography to detect new or changing lesions. Dr. Blalock described this approach as rife with limitations, including variations in quality, challenges of storing and maintaining records, cost, time required to evaluate them, and determining which patients are appropriate candidates.
Other techniques being evaluated include computer algorithms to help dermatologists determine the diagnosis of melanoma from dermoscopic images, electrical impedance spectroscopy for noninvasive evaluation of atypical pigmented lesions, and ultrasound for staging of cutaneous malignant tumors.
Ultimately, “I think we’ll have multiple tools in our belt,” Dr. Blalock said, adding, “How do we pull them out at the right time to improve the lives of our patients? Are we going to use ultrasound? Dermoscopy? Integrate them with some of the genetic findings?”
Dr. Blalock disclosed that he has served as a principal investigator for Castle Biosciences.
San Diego – .
“I don’t think that’s going to change in the short term,” Travis W. Blalock, MD, director of dermatologic surgery, Mohs micrographic surgery, and cutaneous oncology at Emory University, Atlanta, said at the annual Cutaneous Malignancy Update. “But I do think we can supplement that with other modalities that will improve the clinical examination and help dermatopathologists as they assess and evaluate these lesions,” he said, adding: “The reality is, histopathology, while it may be the gold standard, is not necessarily a consistently reproducible evaluation. That raises the question: What can we do better?”
According to Dr. Blalock, the future may include more routine use of noninvasive genetic molecular assays to assist with the diagnostics challenges linked to the visual image and pattern recognition approach of detecting cutaneous melanoma. For example, a two-gene classification method based on LINC00518 and preferentially expressed antigen in melanoma (PRAME) gene expression was evaluated and validated in 555 pigmented lesions obtained noninvasively via adhesive patch biopsy.
“Today, you can pick up a kit from your local pharmacy that can tell you a bit about broad genetic susceptibilities,” he said at the meeting, which was hosted by Scripps MD Anderson Cancer Center. He predicted that using adhesive patch biopsies to assess suspicious melanocytic lesions “is likely the wave of the future.” This may increase patient understanding “as to the types of risks they have, the different lesions they have, and minimize invasive disease, but it also will pose different challenges for us when it comes to deploying patient-centered health care. For example, in a patient with multiple different lesions, how are you going to keep track of them all?”
Dermoscopy
In Dr. Blalock’s clinical opinion, dermoscopy improves the sensitivity of human visual detection of melanoma and may allow detection before a lesion displays classical features described with the “ABCDE rule.” However, the learning curve for dermoscopy is steep, he added, and whether the technique should be considered a first-line tool or as a supplement to other methods of examining cutaneous lesions remains a matter of debate.
“Dermoscopy is our version of the stethoscope,” he said. “We need to figure out when we’re going to use it. Should we be using it all of the time or only some of the time? Based on the clinical setting, maybe it’s a personal choice, but this can be a helpful skill and art in your practice if you’re willing to take the time to learn.”
In 2007, the International Dermoscopy Society (IDS) established a proposal for the standardization and recommended criteria necessary to effectively convey dermoscopic findings to consulting physicians and colleagues. The document includes 10 points categorized as either recommended or optional for a standardized dermoscopy report.
“The first step is to assess the lesion to determine whether or not it’s melanocytic in the first place,” said Dr. Blalock. “There are many different features – the mile-high [global features] evaluation of the lesions – then more specific local features that may clue you in to specific diagnoses,” he noted. “Once we get past that first step of determining that a lesion is melanocytic, it’s not enough to stop there, because we don’t want to biopsy every single lesion that’s melanocytic,” so there is a need to determine which ones require intervention, which is where dermoscopy “gets trickier and a little more challenging.”
According to the IDS, a standard dermoscopy report should include the patient’s age, relevant history pertaining to the lesion, pertinent personal and family history (recommended); clinical description of the lesion (recommended); the two-step method of dermoscopy differentiating melanocytic from nonmelanocytic tumors (recommended); and the use of standardized terms to describe structures as defined by the Dermoscopy Consensus Report published in 2003.
For new terms, the document states, “it would be helpful” for the physician to provide a working definition (recommended); the dermoscopic algorithm used should be mentioned (optional); information on the imaging equipment and magnification (recommended); clinical and dermoscopic images of the tumor (recommended); a diagnosis or differential diagnosis (recommended); decision concerning management (recommended), and specific comments for the pathologist when excision and histopathologic examination are recommended (optional).
The 2007 IDS document also includes a proposed seven-point checklist to differentiate between benign and melanocytic lesions on dermoscopy. Three major criteria are worth two points each: The presence of an atypical pigment network, gray-blue areas (commonly known as the veil), and an atypical vascular pattern. Four minor criteria are worth one point each: Irregular streaks, irregular dots/globules, irregular pigmentation, and regression structures. A minimum total score of 3 is required to establish a diagnosis of melanoma.
Another diagnostic technique, digital mole mapping, involves the use of photography to detect new or changing lesions. Dr. Blalock described this approach as rife with limitations, including variations in quality, challenges of storing and maintaining records, cost, time required to evaluate them, and determining which patients are appropriate candidates.
Other techniques being evaluated include computer algorithms to help dermatologists determine the diagnosis of melanoma from dermoscopic images, electrical impedance spectroscopy for noninvasive evaluation of atypical pigmented lesions, and ultrasound for staging of cutaneous malignant tumors.
Ultimately, “I think we’ll have multiple tools in our belt,” Dr. Blalock said, adding, “How do we pull them out at the right time to improve the lives of our patients? Are we going to use ultrasound? Dermoscopy? Integrate them with some of the genetic findings?”
Dr. Blalock disclosed that he has served as a principal investigator for Castle Biosciences.
San Diego – .
“I don’t think that’s going to change in the short term,” Travis W. Blalock, MD, director of dermatologic surgery, Mohs micrographic surgery, and cutaneous oncology at Emory University, Atlanta, said at the annual Cutaneous Malignancy Update. “But I do think we can supplement that with other modalities that will improve the clinical examination and help dermatopathologists as they assess and evaluate these lesions,” he said, adding: “The reality is, histopathology, while it may be the gold standard, is not necessarily a consistently reproducible evaluation. That raises the question: What can we do better?”
According to Dr. Blalock, the future may include more routine use of noninvasive genetic molecular assays to assist with the diagnostics challenges linked to the visual image and pattern recognition approach of detecting cutaneous melanoma. For example, a two-gene classification method based on LINC00518 and preferentially expressed antigen in melanoma (PRAME) gene expression was evaluated and validated in 555 pigmented lesions obtained noninvasively via adhesive patch biopsy.
“Today, you can pick up a kit from your local pharmacy that can tell you a bit about broad genetic susceptibilities,” he said at the meeting, which was hosted by Scripps MD Anderson Cancer Center. He predicted that using adhesive patch biopsies to assess suspicious melanocytic lesions “is likely the wave of the future.” This may increase patient understanding “as to the types of risks they have, the different lesions they have, and minimize invasive disease, but it also will pose different challenges for us when it comes to deploying patient-centered health care. For example, in a patient with multiple different lesions, how are you going to keep track of them all?”
Dermoscopy
In Dr. Blalock’s clinical opinion, dermoscopy improves the sensitivity of human visual detection of melanoma and may allow detection before a lesion displays classical features described with the “ABCDE rule.” However, the learning curve for dermoscopy is steep, he added, and whether the technique should be considered a first-line tool or as a supplement to other methods of examining cutaneous lesions remains a matter of debate.
“Dermoscopy is our version of the stethoscope,” he said. “We need to figure out when we’re going to use it. Should we be using it all of the time or only some of the time? Based on the clinical setting, maybe it’s a personal choice, but this can be a helpful skill and art in your practice if you’re willing to take the time to learn.”
In 2007, the International Dermoscopy Society (IDS) established a proposal for the standardization and recommended criteria necessary to effectively convey dermoscopic findings to consulting physicians and colleagues. The document includes 10 points categorized as either recommended or optional for a standardized dermoscopy report.
“The first step is to assess the lesion to determine whether or not it’s melanocytic in the first place,” said Dr. Blalock. “There are many different features – the mile-high [global features] evaluation of the lesions – then more specific local features that may clue you in to specific diagnoses,” he noted. “Once we get past that first step of determining that a lesion is melanocytic, it’s not enough to stop there, because we don’t want to biopsy every single lesion that’s melanocytic,” so there is a need to determine which ones require intervention, which is where dermoscopy “gets trickier and a little more challenging.”
According to the IDS, a standard dermoscopy report should include the patient’s age, relevant history pertaining to the lesion, pertinent personal and family history (recommended); clinical description of the lesion (recommended); the two-step method of dermoscopy differentiating melanocytic from nonmelanocytic tumors (recommended); and the use of standardized terms to describe structures as defined by the Dermoscopy Consensus Report published in 2003.
For new terms, the document states, “it would be helpful” for the physician to provide a working definition (recommended); the dermoscopic algorithm used should be mentioned (optional); information on the imaging equipment and magnification (recommended); clinical and dermoscopic images of the tumor (recommended); a diagnosis or differential diagnosis (recommended); decision concerning management (recommended), and specific comments for the pathologist when excision and histopathologic examination are recommended (optional).
The 2007 IDS document also includes a proposed seven-point checklist to differentiate between benign and melanocytic lesions on dermoscopy. Three major criteria are worth two points each: The presence of an atypical pigment network, gray-blue areas (commonly known as the veil), and an atypical vascular pattern. Four minor criteria are worth one point each: Irregular streaks, irregular dots/globules, irregular pigmentation, and regression structures. A minimum total score of 3 is required to establish a diagnosis of melanoma.
Another diagnostic technique, digital mole mapping, involves the use of photography to detect new or changing lesions. Dr. Blalock described this approach as rife with limitations, including variations in quality, challenges of storing and maintaining records, cost, time required to evaluate them, and determining which patients are appropriate candidates.
Other techniques being evaluated include computer algorithms to help dermatologists determine the diagnosis of melanoma from dermoscopic images, electrical impedance spectroscopy for noninvasive evaluation of atypical pigmented lesions, and ultrasound for staging of cutaneous malignant tumors.
Ultimately, “I think we’ll have multiple tools in our belt,” Dr. Blalock said, adding, “How do we pull them out at the right time to improve the lives of our patients? Are we going to use ultrasound? Dermoscopy? Integrate them with some of the genetic findings?”
Dr. Blalock disclosed that he has served as a principal investigator for Castle Biosciences.
AT MELANOMA 2023
Gene test may offer insights into treatment response in advanced NSCLC
therapy despite their poor status, researchers reported.
Positive findings on the test, known as DetermaIO, were “associated with efficacy of response to ICI therapy in advanced NSCLC patients,” Matthew G. Varga, PhD, manager of scientific affairs at Oncocyte, said in an interview. “These data suggest that DetermaIO warrants further study in poor performance status patients as it has the potential to identify likely responders to ICI therapy.”
Oncocyte, which is developing the test, presented the findings in a poster at the annual meeting of the Society for Immunotherapy of Cancer.
According to Dr. Varga, “DetermaIO is an RT-qPCR test that can be applied to FFPE [formalin-fixed, paraffin-embedded] tissue specimens to quantify the relative gene expression of 27 genes and subsequently applies our proprietary algorithm to generate an IO score based on the gene expression profile. The DetermaIO score is a binary IO+ or IO– score, representing likely responder or nonresponder, respectively.”
The test was originally developed for triple negative breast cancer, Dr. Varga said, and it’s been validated in non–small cell lung cancer, metastatic urothelial carcinoma, and metastatic colorectal carcinoma.
For the study, the researchers retrospectively tracked associations between DetermaIO score and either progression-free survival (PFS) or overall survival (OS) in 147 patients in Canada with NSCLC who were treated with ICI monotherapy. All had programmed death-ligand 1 (PD-L1) ≥ 50%.
Overall, outcomes were poor: The median survival was 12.7 months, and median PFS was 7.0 months. These outcomes were even worse in those who underwent therapy as a second- line treatment: The median survival was 9.7 months, and median PFS was 4.4 months.
“DetermaIO was significantly associated with PFS at hazard ratio [HR] = 0.55, 95% [confidence interval] CI, 0.32-0.94, P = .028. In our analyses, a hazard ratio less than 1 suggests lower risk – i.e, that DetermaIO+ patients have lower risk of an event – death or progression – compared to a DetermaIO– patient,” Dr. Varga said. “The association for overall survival was not statistically significant, but it was suggestive of clinically meaningful benefit.”
He added that “we could identify likely responders from nonresponders, suggesting that the DetermaIO score adds both independent and incremental data to the existing gold standard biomarker. The objective response rate for all first-line patients – n = 78 – was 44.9%. Twenty-two DetermaIO– tumors had a 23% response rate (5 partial responses) whereas of the 56 DetermaIO+ patients, the response rate was 54% (2 complete response and 28 partial responses).”
A score on the test, he said, was not associated with OS or PFS in patients who received second-line or later treatment.
The study was not designed to evaluate the predictive power of the test. “For a biomarker to be defined as predictive requires a formal test of interaction between a treatment group (ICI monotherapy, for example) vs. a control group (chemo-only or other regimen),” Dr. Varga explained. “In our analysis, there was no group of patients who did not receive ICI monotherapy. Thus a test for interaction and a predictive claim cannot be made.”
The test is available for at no cost via an early access program, Dr. Varga said, and Oncocyte is getting ready to seek Medicare coverage. The ultimate cost of the test, he said, is unknown.
Oncocyte funded this study. Dr. Varga and several other study authors are Oncocyte employees, and another author is a paid consultant to the company.
therapy despite their poor status, researchers reported.
Positive findings on the test, known as DetermaIO, were “associated with efficacy of response to ICI therapy in advanced NSCLC patients,” Matthew G. Varga, PhD, manager of scientific affairs at Oncocyte, said in an interview. “These data suggest that DetermaIO warrants further study in poor performance status patients as it has the potential to identify likely responders to ICI therapy.”
Oncocyte, which is developing the test, presented the findings in a poster at the annual meeting of the Society for Immunotherapy of Cancer.
According to Dr. Varga, “DetermaIO is an RT-qPCR test that can be applied to FFPE [formalin-fixed, paraffin-embedded] tissue specimens to quantify the relative gene expression of 27 genes and subsequently applies our proprietary algorithm to generate an IO score based on the gene expression profile. The DetermaIO score is a binary IO+ or IO– score, representing likely responder or nonresponder, respectively.”
The test was originally developed for triple negative breast cancer, Dr. Varga said, and it’s been validated in non–small cell lung cancer, metastatic urothelial carcinoma, and metastatic colorectal carcinoma.
For the study, the researchers retrospectively tracked associations between DetermaIO score and either progression-free survival (PFS) or overall survival (OS) in 147 patients in Canada with NSCLC who were treated with ICI monotherapy. All had programmed death-ligand 1 (PD-L1) ≥ 50%.
Overall, outcomes were poor: The median survival was 12.7 months, and median PFS was 7.0 months. These outcomes were even worse in those who underwent therapy as a second- line treatment: The median survival was 9.7 months, and median PFS was 4.4 months.
“DetermaIO was significantly associated with PFS at hazard ratio [HR] = 0.55, 95% [confidence interval] CI, 0.32-0.94, P = .028. In our analyses, a hazard ratio less than 1 suggests lower risk – i.e, that DetermaIO+ patients have lower risk of an event – death or progression – compared to a DetermaIO– patient,” Dr. Varga said. “The association for overall survival was not statistically significant, but it was suggestive of clinically meaningful benefit.”
He added that “we could identify likely responders from nonresponders, suggesting that the DetermaIO score adds both independent and incremental data to the existing gold standard biomarker. The objective response rate for all first-line patients – n = 78 – was 44.9%. Twenty-two DetermaIO– tumors had a 23% response rate (5 partial responses) whereas of the 56 DetermaIO+ patients, the response rate was 54% (2 complete response and 28 partial responses).”
A score on the test, he said, was not associated with OS or PFS in patients who received second-line or later treatment.
The study was not designed to evaluate the predictive power of the test. “For a biomarker to be defined as predictive requires a formal test of interaction between a treatment group (ICI monotherapy, for example) vs. a control group (chemo-only or other regimen),” Dr. Varga explained. “In our analysis, there was no group of patients who did not receive ICI monotherapy. Thus a test for interaction and a predictive claim cannot be made.”
The test is available for at no cost via an early access program, Dr. Varga said, and Oncocyte is getting ready to seek Medicare coverage. The ultimate cost of the test, he said, is unknown.
Oncocyte funded this study. Dr. Varga and several other study authors are Oncocyte employees, and another author is a paid consultant to the company.
therapy despite their poor status, researchers reported.
Positive findings on the test, known as DetermaIO, were “associated with efficacy of response to ICI therapy in advanced NSCLC patients,” Matthew G. Varga, PhD, manager of scientific affairs at Oncocyte, said in an interview. “These data suggest that DetermaIO warrants further study in poor performance status patients as it has the potential to identify likely responders to ICI therapy.”
Oncocyte, which is developing the test, presented the findings in a poster at the annual meeting of the Society for Immunotherapy of Cancer.
According to Dr. Varga, “DetermaIO is an RT-qPCR test that can be applied to FFPE [formalin-fixed, paraffin-embedded] tissue specimens to quantify the relative gene expression of 27 genes and subsequently applies our proprietary algorithm to generate an IO score based on the gene expression profile. The DetermaIO score is a binary IO+ or IO– score, representing likely responder or nonresponder, respectively.”
The test was originally developed for triple negative breast cancer, Dr. Varga said, and it’s been validated in non–small cell lung cancer, metastatic urothelial carcinoma, and metastatic colorectal carcinoma.
For the study, the researchers retrospectively tracked associations between DetermaIO score and either progression-free survival (PFS) or overall survival (OS) in 147 patients in Canada with NSCLC who were treated with ICI monotherapy. All had programmed death-ligand 1 (PD-L1) ≥ 50%.
Overall, outcomes were poor: The median survival was 12.7 months, and median PFS was 7.0 months. These outcomes were even worse in those who underwent therapy as a second- line treatment: The median survival was 9.7 months, and median PFS was 4.4 months.
“DetermaIO was significantly associated with PFS at hazard ratio [HR] = 0.55, 95% [confidence interval] CI, 0.32-0.94, P = .028. In our analyses, a hazard ratio less than 1 suggests lower risk – i.e, that DetermaIO+ patients have lower risk of an event – death or progression – compared to a DetermaIO– patient,” Dr. Varga said. “The association for overall survival was not statistically significant, but it was suggestive of clinically meaningful benefit.”
He added that “we could identify likely responders from nonresponders, suggesting that the DetermaIO score adds both independent and incremental data to the existing gold standard biomarker. The objective response rate for all first-line patients – n = 78 – was 44.9%. Twenty-two DetermaIO– tumors had a 23% response rate (5 partial responses) whereas of the 56 DetermaIO+ patients, the response rate was 54% (2 complete response and 28 partial responses).”
A score on the test, he said, was not associated with OS or PFS in patients who received second-line or later treatment.
The study was not designed to evaluate the predictive power of the test. “For a biomarker to be defined as predictive requires a formal test of interaction between a treatment group (ICI monotherapy, for example) vs. a control group (chemo-only or other regimen),” Dr. Varga explained. “In our analysis, there was no group of patients who did not receive ICI monotherapy. Thus a test for interaction and a predictive claim cannot be made.”
The test is available for at no cost via an early access program, Dr. Varga said, and Oncocyte is getting ready to seek Medicare coverage. The ultimate cost of the test, he said, is unknown.
Oncocyte funded this study. Dr. Varga and several other study authors are Oncocyte employees, and another author is a paid consultant to the company.
FROM SITC 2022
Long-course radiation therapy better at organ-sparing in rectal cancer than short-term therapy
as part of a total neoadjuvant therapy (TNT) strategy, shows new research presented at the ASCO Gastrointestinal Cancers Symposium 2023.
“When we looked at the 2-year organ preservation rates, they were numerically higher in the long-course group versus the short-course group,” said study author J. Joshua Smith, MD, PhD,FACS, a colorectal surgeon with Memorial Sloan Kettering Cancer Center, New York. “Our study will be the first, to our knowledge, that examines a significant proportion of patients treated with the induction total neoadjuvant therapy approach – chemo first.”
An ideal outcome in rectal cancer is no need for surgery, Dr. Smith said. “If you can avoid surgery altogether and preserve the organ [the rectum], that’s a big win for the patient as they are usually able to avoid having a permanent or temporary ostomy.”
Long-course and short-course radiation have similar outcomes in terms of patients going on to need surgery, but it’s not clear which is superior in terms of organ sparing, toxicity, and side effects, said Paul Romesser, MD, a radiation oncologist with Memorial Sloan Kettering Cancer Center, New York, who served as first author of the study.
During the early months of the COVID-19 pandemic, the cancer center embraced short-course radiation in rectal cancer, Dr. Romesser said. “Once we emerged from the cloud of COVID, we said: ‘Well, what do we do now? Where do we go? Do we go back to what we did before? Or, do we go stick with the same? And what does that mean for organ preservation?’ ”
The researchers retrospectively identified 563 consecutive patients treated with TNT from 2018 to 2021. They focused on 332 who didn’t have metastatic disease, synchronous/metachronous malignancies, or nonadenocarcinoma histology (long course = 256, short course = 76). The groups had similar high-risk features, and about 82% were clinical stage III).
Patients most commonly received induction chemotherapy followed by consolidative radiation (78% long course, 70% short course).
The 2-year survival rates were similar, but organ preservation was higher in the long-course group versus the short-course group (40%; 95% confidence interval, 35%-47% vs. 29%; 95% CI, 20%-42%). And the 2-year local regrowth rate was also better in the long-course group versus the short-course group (20%; 95% CI, 12%-27% vs. 36%; 95% CI, 16%-52%).
Why might long-course therapy be better? “It’s probably just coming down to the biologically equivalent dose,” which is likely lower in short-course radiation, Dr. Romesser said.
Going forward, Dr. Romesser said he’ll tell patients about the findings of this study and a previous report published in 2022 that determined that “organ preservation is achievable in half of the patients with rectal cancer treated with total neoadjuvant therapy, without an apparent detriment in survival, compared with historical controls treated with chemoradiotherapy, TME [total mesorectal excision], and postoperative chemotherapy.” Dr. Smith is a coauthor of that study.
“Generally, I’ll steer patients toward long course, assuming all else is equal, and it’s not an undue burden on them financially and socially to come in for 5-6 weeks of chemoradiation,” Dr. Romesser said. He added that, “generally, the insurance companies recognize [short-course and long-course radiation] as both acceptable and standard treatment options for patients. We haven’t found that insurances will approve one, but not the other.”
The study was funded by the National Institutes of Health. Dr. Romesser disclosed consulting/advisory roles (EMD Serono, Faeth, Natera), research funding (XRad), and travel/accommodations/expenses (Elekta). Dr. Smith disclosed consulting/advisory roles (Foundation Medicine, Guardant Health). The other study authors reported no conflicts of interest.
The Gastrointestinal Cancers Symposium is sponsored by the American Gastroenterological Association, the American Society for Clinical Oncology, the American Society for Radiation Oncology, and the Society of Surgical Oncology.
as part of a total neoadjuvant therapy (TNT) strategy, shows new research presented at the ASCO Gastrointestinal Cancers Symposium 2023.
“When we looked at the 2-year organ preservation rates, they were numerically higher in the long-course group versus the short-course group,” said study author J. Joshua Smith, MD, PhD,FACS, a colorectal surgeon with Memorial Sloan Kettering Cancer Center, New York. “Our study will be the first, to our knowledge, that examines a significant proportion of patients treated with the induction total neoadjuvant therapy approach – chemo first.”
An ideal outcome in rectal cancer is no need for surgery, Dr. Smith said. “If you can avoid surgery altogether and preserve the organ [the rectum], that’s a big win for the patient as they are usually able to avoid having a permanent or temporary ostomy.”
Long-course and short-course radiation have similar outcomes in terms of patients going on to need surgery, but it’s not clear which is superior in terms of organ sparing, toxicity, and side effects, said Paul Romesser, MD, a radiation oncologist with Memorial Sloan Kettering Cancer Center, New York, who served as first author of the study.
During the early months of the COVID-19 pandemic, the cancer center embraced short-course radiation in rectal cancer, Dr. Romesser said. “Once we emerged from the cloud of COVID, we said: ‘Well, what do we do now? Where do we go? Do we go back to what we did before? Or, do we go stick with the same? And what does that mean for organ preservation?’ ”
The researchers retrospectively identified 563 consecutive patients treated with TNT from 2018 to 2021. They focused on 332 who didn’t have metastatic disease, synchronous/metachronous malignancies, or nonadenocarcinoma histology (long course = 256, short course = 76). The groups had similar high-risk features, and about 82% were clinical stage III).
Patients most commonly received induction chemotherapy followed by consolidative radiation (78% long course, 70% short course).
The 2-year survival rates were similar, but organ preservation was higher in the long-course group versus the short-course group (40%; 95% confidence interval, 35%-47% vs. 29%; 95% CI, 20%-42%). And the 2-year local regrowth rate was also better in the long-course group versus the short-course group (20%; 95% CI, 12%-27% vs. 36%; 95% CI, 16%-52%).
Why might long-course therapy be better? “It’s probably just coming down to the biologically equivalent dose,” which is likely lower in short-course radiation, Dr. Romesser said.
Going forward, Dr. Romesser said he’ll tell patients about the findings of this study and a previous report published in 2022 that determined that “organ preservation is achievable in half of the patients with rectal cancer treated with total neoadjuvant therapy, without an apparent detriment in survival, compared with historical controls treated with chemoradiotherapy, TME [total mesorectal excision], and postoperative chemotherapy.” Dr. Smith is a coauthor of that study.
“Generally, I’ll steer patients toward long course, assuming all else is equal, and it’s not an undue burden on them financially and socially to come in for 5-6 weeks of chemoradiation,” Dr. Romesser said. He added that, “generally, the insurance companies recognize [short-course and long-course radiation] as both acceptable and standard treatment options for patients. We haven’t found that insurances will approve one, but not the other.”
The study was funded by the National Institutes of Health. Dr. Romesser disclosed consulting/advisory roles (EMD Serono, Faeth, Natera), research funding (XRad), and travel/accommodations/expenses (Elekta). Dr. Smith disclosed consulting/advisory roles (Foundation Medicine, Guardant Health). The other study authors reported no conflicts of interest.
The Gastrointestinal Cancers Symposium is sponsored by the American Gastroenterological Association, the American Society for Clinical Oncology, the American Society for Radiation Oncology, and the Society of Surgical Oncology.
as part of a total neoadjuvant therapy (TNT) strategy, shows new research presented at the ASCO Gastrointestinal Cancers Symposium 2023.
“When we looked at the 2-year organ preservation rates, they were numerically higher in the long-course group versus the short-course group,” said study author J. Joshua Smith, MD, PhD,FACS, a colorectal surgeon with Memorial Sloan Kettering Cancer Center, New York. “Our study will be the first, to our knowledge, that examines a significant proportion of patients treated with the induction total neoadjuvant therapy approach – chemo first.”
An ideal outcome in rectal cancer is no need for surgery, Dr. Smith said. “If you can avoid surgery altogether and preserve the organ [the rectum], that’s a big win for the patient as they are usually able to avoid having a permanent or temporary ostomy.”
Long-course and short-course radiation have similar outcomes in terms of patients going on to need surgery, but it’s not clear which is superior in terms of organ sparing, toxicity, and side effects, said Paul Romesser, MD, a radiation oncologist with Memorial Sloan Kettering Cancer Center, New York, who served as first author of the study.
During the early months of the COVID-19 pandemic, the cancer center embraced short-course radiation in rectal cancer, Dr. Romesser said. “Once we emerged from the cloud of COVID, we said: ‘Well, what do we do now? Where do we go? Do we go back to what we did before? Or, do we go stick with the same? And what does that mean for organ preservation?’ ”
The researchers retrospectively identified 563 consecutive patients treated with TNT from 2018 to 2021. They focused on 332 who didn’t have metastatic disease, synchronous/metachronous malignancies, or nonadenocarcinoma histology (long course = 256, short course = 76). The groups had similar high-risk features, and about 82% were clinical stage III).
Patients most commonly received induction chemotherapy followed by consolidative radiation (78% long course, 70% short course).
The 2-year survival rates were similar, but organ preservation was higher in the long-course group versus the short-course group (40%; 95% confidence interval, 35%-47% vs. 29%; 95% CI, 20%-42%). And the 2-year local regrowth rate was also better in the long-course group versus the short-course group (20%; 95% CI, 12%-27% vs. 36%; 95% CI, 16%-52%).
Why might long-course therapy be better? “It’s probably just coming down to the biologically equivalent dose,” which is likely lower in short-course radiation, Dr. Romesser said.
Going forward, Dr. Romesser said he’ll tell patients about the findings of this study and a previous report published in 2022 that determined that “organ preservation is achievable in half of the patients with rectal cancer treated with total neoadjuvant therapy, without an apparent detriment in survival, compared with historical controls treated with chemoradiotherapy, TME [total mesorectal excision], and postoperative chemotherapy.” Dr. Smith is a coauthor of that study.
“Generally, I’ll steer patients toward long course, assuming all else is equal, and it’s not an undue burden on them financially and socially to come in for 5-6 weeks of chemoradiation,” Dr. Romesser said. He added that, “generally, the insurance companies recognize [short-course and long-course radiation] as both acceptable and standard treatment options for patients. We haven’t found that insurances will approve one, but not the other.”
The study was funded by the National Institutes of Health. Dr. Romesser disclosed consulting/advisory roles (EMD Serono, Faeth, Natera), research funding (XRad), and travel/accommodations/expenses (Elekta). Dr. Smith disclosed consulting/advisory roles (Foundation Medicine, Guardant Health). The other study authors reported no conflicts of interest.
The Gastrointestinal Cancers Symposium is sponsored by the American Gastroenterological Association, the American Society for Clinical Oncology, the American Society for Radiation Oncology, and the Society of Surgical Oncology.
FROM ASCO GI 2023
Colorectal cancer treatment outcomes in older adults
A phase 2, multi-institutional feasibility study found a completion rate of 67.3%, while a prospective study found that completion was associated with improved disease-free survival.
Both studies were presented in January at the ASCO Gastrointestinal Cancers Symposium 2023.
In HiSCO-04, Japanese researchers found that of 64 older patients with stage 3A colorectal cancer who underwent adjuvant chemotherapy, 53% completed the treatment with an improvement in disease-free survival. Patients who completed adjuvant chemotherapy had better disease-free survival (P = .03), while the survival was lower among those who did not receive adjuvant chemotherapy, and lowest among those who discontinued adjuvant chemotherapy.
“The results showed that adjuvant chemotherapy is not always recommended for elderly patients, and that patients who are able to complete treatment may have a better prognosis for survival. However, the results do not indicate which patients are unable to complete chemotherapy, and it will be necessary to identify patients who are intolerant of chemotherapy,” said the study’s lead author Manabu Shimomura, MD, PhD, an assistant professor of gastroenterological and transplant surgery at the Hiroshima University Graduate School of Biomedical and Health Sciences in Japan.
The study, which was conducted between 2013 and 2021, enrolled 214 patients (99 men, 115 women, 80-101 years old) who were in stage 3 cancer (27 cases 3A, 158 cases 3B, and 29 cases 3C). A total of 41 patients were ineligible for chemotherapy. Of the remaining patients, 65 received adjuvant chemotherapy and 108 did not receive adjuvant chemotherapy.
The 3-year disease-free survival was 63.6%, the 3-year overall survival was 76.9%, and the 3-year relapse-free survival was 63.1%. Thirty-six patients died because of colorectal cancer, and 30 patients died of other causes. There was recurrence in 58 cases and secondary cancers were observed in 17 cases during the 42.5 months–long follow-up period.
There were few reports of serious adverse events, but some cases of treatment discontinuation were because of adverse events.
In a second study presented by Dr. Shimomura’s group, called HiSCO-03, 65 patients (33 female) underwent curative resection and received five courses of uracil-tegafur and leucovorin (UFT/LV).
The completion rate of 67.3% had a 95% lower bound of 54.9%, which were lower than the predefined thresholds of 75% completion and a lower bound of 60%. “Based on the results of a previous (ACTS-CC phase III) study, we set the expected value of UFT/LV therapy in patients over 80 years of age at 75% and the threshold at 60%. Since the target age group of previous study was 75 years or younger, we concluded from the results of the current study that UFT/LV therapy is less well tolerated in patients 80 years of age and older than in patients 75 years of age and younger,” Dr. Shimomura said.
The treatment completion rate trended higher in males than females (77.6% versus 57.2%; P = .06) and performance status of 0 versus 1 or 2 (74.3% versus 58.9%; P = .10). The most common adverse events were anorexia (33.8%), diarrhea (30.8%), and anemia (24.6%). The median relative dose intensity was 84% for UFT and 100% for LV.
The challenges of treating older patients
If and how older patients with colorectal cancer should be treated is not clear cut. While 20% of patients in the United States who have colorectal cancer are over 80 years old, each case should be evaluated individually, experts say.
Writing in a 2015 review of colorectal cancer treatment in older adults, Monica Millan, MD, PhD, of Joan XXIII University Hospital, Tarragona, Spain, and colleagues, wrote that physiological heterogeneity and coexisting medical conditions make treating older patients with colorectal cancer challenging.
“Age in itself should not be an exclusion criterion for radical treatment, but there will be many elderly patients that will not tolerate or respond well to standard therapies. These patients need to be properly assessed before proposing treatment, and a tailored, individualized approach should be offered in a multidisciplinary setting,” wrote Dr. Millan, who is a colorectal surgeon.
The authors suggest that older patients who are fit could be treated similarly to younger patients, but there remain uncertainties about how to proceed in frail older adults with comorbidities.
“Most elderly patients with cancer will have priorities besides simply prolonging their lives. Surveys have found that their top concerns include avoiding suffering, strengthening relationships with family and friends, being mentally aware, not being a burden on others, and achieving a sense that their life is complete. The treatment plan should be comprehensive: cancer-specific treatment, symptom-specific treatment, supportive treatment modalities, and end-of-life care,” they wrote.
The U.S. Preventive Services Task Force recommends colorectal cancer screening for men and women who are between 45 and 75 years old; however, screening for patients between 76 and 85 years old should be done on a case-by-case basis based on a patient’s overall health, screening history, and the patient’s preferences.
Colorectal cancer incidence rates have been declining since the mid-1980s because of an increase in screening among adults 50 years and older, according to the American Cancer Society. Likewise, mortality rates have dropped from 29.2% in 1970 to 12.6% in 2020 – mostly because of screening.
Dr. Shimomura has no relevant financial disclosures.
The Gastrointestinal Cancers Symposium is sponsored by the American Gastroenterological Association, the American Society for Clinical Oncology, the American Society for Radiation Oncology, and the Society of Surgical Oncology.
A phase 2, multi-institutional feasibility study found a completion rate of 67.3%, while a prospective study found that completion was associated with improved disease-free survival.
Both studies were presented in January at the ASCO Gastrointestinal Cancers Symposium 2023.
In HiSCO-04, Japanese researchers found that of 64 older patients with stage 3A colorectal cancer who underwent adjuvant chemotherapy, 53% completed the treatment with an improvement in disease-free survival. Patients who completed adjuvant chemotherapy had better disease-free survival (P = .03), while the survival was lower among those who did not receive adjuvant chemotherapy, and lowest among those who discontinued adjuvant chemotherapy.
“The results showed that adjuvant chemotherapy is not always recommended for elderly patients, and that patients who are able to complete treatment may have a better prognosis for survival. However, the results do not indicate which patients are unable to complete chemotherapy, and it will be necessary to identify patients who are intolerant of chemotherapy,” said the study’s lead author Manabu Shimomura, MD, PhD, an assistant professor of gastroenterological and transplant surgery at the Hiroshima University Graduate School of Biomedical and Health Sciences in Japan.
The study, which was conducted between 2013 and 2021, enrolled 214 patients (99 men, 115 women, 80-101 years old) who were in stage 3 cancer (27 cases 3A, 158 cases 3B, and 29 cases 3C). A total of 41 patients were ineligible for chemotherapy. Of the remaining patients, 65 received adjuvant chemotherapy and 108 did not receive adjuvant chemotherapy.
The 3-year disease-free survival was 63.6%, the 3-year overall survival was 76.9%, and the 3-year relapse-free survival was 63.1%. Thirty-six patients died because of colorectal cancer, and 30 patients died of other causes. There was recurrence in 58 cases and secondary cancers were observed in 17 cases during the 42.5 months–long follow-up period.
There were few reports of serious adverse events, but some cases of treatment discontinuation were because of adverse events.
In a second study presented by Dr. Shimomura’s group, called HiSCO-03, 65 patients (33 female) underwent curative resection and received five courses of uracil-tegafur and leucovorin (UFT/LV).
The completion rate of 67.3% had a 95% lower bound of 54.9%, which were lower than the predefined thresholds of 75% completion and a lower bound of 60%. “Based on the results of a previous (ACTS-CC phase III) study, we set the expected value of UFT/LV therapy in patients over 80 years of age at 75% and the threshold at 60%. Since the target age group of previous study was 75 years or younger, we concluded from the results of the current study that UFT/LV therapy is less well tolerated in patients 80 years of age and older than in patients 75 years of age and younger,” Dr. Shimomura said.
The treatment completion rate trended higher in males than females (77.6% versus 57.2%; P = .06) and performance status of 0 versus 1 or 2 (74.3% versus 58.9%; P = .10). The most common adverse events were anorexia (33.8%), diarrhea (30.8%), and anemia (24.6%). The median relative dose intensity was 84% for UFT and 100% for LV.
The challenges of treating older patients
If and how older patients with colorectal cancer should be treated is not clear cut. While 20% of patients in the United States who have colorectal cancer are over 80 years old, each case should be evaluated individually, experts say.
Writing in a 2015 review of colorectal cancer treatment in older adults, Monica Millan, MD, PhD, of Joan XXIII University Hospital, Tarragona, Spain, and colleagues, wrote that physiological heterogeneity and coexisting medical conditions make treating older patients with colorectal cancer challenging.
“Age in itself should not be an exclusion criterion for radical treatment, but there will be many elderly patients that will not tolerate or respond well to standard therapies. These patients need to be properly assessed before proposing treatment, and a tailored, individualized approach should be offered in a multidisciplinary setting,” wrote Dr. Millan, who is a colorectal surgeon.
The authors suggest that older patients who are fit could be treated similarly to younger patients, but there remain uncertainties about how to proceed in frail older adults with comorbidities.
“Most elderly patients with cancer will have priorities besides simply prolonging their lives. Surveys have found that their top concerns include avoiding suffering, strengthening relationships with family and friends, being mentally aware, not being a burden on others, and achieving a sense that their life is complete. The treatment plan should be comprehensive: cancer-specific treatment, symptom-specific treatment, supportive treatment modalities, and end-of-life care,” they wrote.
The U.S. Preventive Services Task Force recommends colorectal cancer screening for men and women who are between 45 and 75 years old; however, screening for patients between 76 and 85 years old should be done on a case-by-case basis based on a patient’s overall health, screening history, and the patient’s preferences.
Colorectal cancer incidence rates have been declining since the mid-1980s because of an increase in screening among adults 50 years and older, according to the American Cancer Society. Likewise, mortality rates have dropped from 29.2% in 1970 to 12.6% in 2020 – mostly because of screening.
Dr. Shimomura has no relevant financial disclosures.
The Gastrointestinal Cancers Symposium is sponsored by the American Gastroenterological Association, the American Society for Clinical Oncology, the American Society for Radiation Oncology, and the Society of Surgical Oncology.
A phase 2, multi-institutional feasibility study found a completion rate of 67.3%, while a prospective study found that completion was associated with improved disease-free survival.
Both studies were presented in January at the ASCO Gastrointestinal Cancers Symposium 2023.
In HiSCO-04, Japanese researchers found that of 64 older patients with stage 3A colorectal cancer who underwent adjuvant chemotherapy, 53% completed the treatment with an improvement in disease-free survival. Patients who completed adjuvant chemotherapy had better disease-free survival (P = .03), while the survival was lower among those who did not receive adjuvant chemotherapy, and lowest among those who discontinued adjuvant chemotherapy.
“The results showed that adjuvant chemotherapy is not always recommended for elderly patients, and that patients who are able to complete treatment may have a better prognosis for survival. However, the results do not indicate which patients are unable to complete chemotherapy, and it will be necessary to identify patients who are intolerant of chemotherapy,” said the study’s lead author Manabu Shimomura, MD, PhD, an assistant professor of gastroenterological and transplant surgery at the Hiroshima University Graduate School of Biomedical and Health Sciences in Japan.
The study, which was conducted between 2013 and 2021, enrolled 214 patients (99 men, 115 women, 80-101 years old) who were in stage 3 cancer (27 cases 3A, 158 cases 3B, and 29 cases 3C). A total of 41 patients were ineligible for chemotherapy. Of the remaining patients, 65 received adjuvant chemotherapy and 108 did not receive adjuvant chemotherapy.
The 3-year disease-free survival was 63.6%, the 3-year overall survival was 76.9%, and the 3-year relapse-free survival was 63.1%. Thirty-six patients died because of colorectal cancer, and 30 patients died of other causes. There was recurrence in 58 cases and secondary cancers were observed in 17 cases during the 42.5 months–long follow-up period.
There were few reports of serious adverse events, but some cases of treatment discontinuation were because of adverse events.
In a second study presented by Dr. Shimomura’s group, called HiSCO-03, 65 patients (33 female) underwent curative resection and received five courses of uracil-tegafur and leucovorin (UFT/LV).
The completion rate of 67.3% had a 95% lower bound of 54.9%, which were lower than the predefined thresholds of 75% completion and a lower bound of 60%. “Based on the results of a previous (ACTS-CC phase III) study, we set the expected value of UFT/LV therapy in patients over 80 years of age at 75% and the threshold at 60%. Since the target age group of previous study was 75 years or younger, we concluded from the results of the current study that UFT/LV therapy is less well tolerated in patients 80 years of age and older than in patients 75 years of age and younger,” Dr. Shimomura said.
The treatment completion rate trended higher in males than females (77.6% versus 57.2%; P = .06) and performance status of 0 versus 1 or 2 (74.3% versus 58.9%; P = .10). The most common adverse events were anorexia (33.8%), diarrhea (30.8%), and anemia (24.6%). The median relative dose intensity was 84% for UFT and 100% for LV.
The challenges of treating older patients
If and how older patients with colorectal cancer should be treated is not clear cut. While 20% of patients in the United States who have colorectal cancer are over 80 years old, each case should be evaluated individually, experts say.
Writing in a 2015 review of colorectal cancer treatment in older adults, Monica Millan, MD, PhD, of Joan XXIII University Hospital, Tarragona, Spain, and colleagues, wrote that physiological heterogeneity and coexisting medical conditions make treating older patients with colorectal cancer challenging.
“Age in itself should not be an exclusion criterion for radical treatment, but there will be many elderly patients that will not tolerate or respond well to standard therapies. These patients need to be properly assessed before proposing treatment, and a tailored, individualized approach should be offered in a multidisciplinary setting,” wrote Dr. Millan, who is a colorectal surgeon.
The authors suggest that older patients who are fit could be treated similarly to younger patients, but there remain uncertainties about how to proceed in frail older adults with comorbidities.
“Most elderly patients with cancer will have priorities besides simply prolonging their lives. Surveys have found that their top concerns include avoiding suffering, strengthening relationships with family and friends, being mentally aware, not being a burden on others, and achieving a sense that their life is complete. The treatment plan should be comprehensive: cancer-specific treatment, symptom-specific treatment, supportive treatment modalities, and end-of-life care,” they wrote.
The U.S. Preventive Services Task Force recommends colorectal cancer screening for men and women who are between 45 and 75 years old; however, screening for patients between 76 and 85 years old should be done on a case-by-case basis based on a patient’s overall health, screening history, and the patient’s preferences.
Colorectal cancer incidence rates have been declining since the mid-1980s because of an increase in screening among adults 50 years and older, according to the American Cancer Society. Likewise, mortality rates have dropped from 29.2% in 1970 to 12.6% in 2020 – mostly because of screening.
Dr. Shimomura has no relevant financial disclosures.
The Gastrointestinal Cancers Symposium is sponsored by the American Gastroenterological Association, the American Society for Clinical Oncology, the American Society for Radiation Oncology, and the Society of Surgical Oncology.
FROM ASCO GI 2023