User login
Neurology Reviews covers innovative and emerging news in neurology and neuroscience every month, with a focus on practical approaches to treating Parkinson's disease, epilepsy, headache, stroke, multiple sclerosis, Alzheimer's disease, and other neurologic disorders.
PML
Progressive multifocal leukoencephalopathy
Rituxan
The leading independent newspaper covering neurology news and commentary.
Women with atrial fibrillation more likely to develop dementia
New data suggest a significantly stronger link in women compared with men between atrial fibrillation (AF) and mild cognitive impairment (MCI) and dementia.
“Our findings imply that women with AF may be at higher risk for MCI and dementia with potentially more rapid disease progression from normal cognition to MCI or dementia than women without AF or men with and without AF,” wrote authors of a new study led by Kathryn A. Wood, PhD, RN, Neil Hodgson Woodruff School of Nursing at Emory University in Atlanta.
The findings were published online in Alzheimer’s & Dementia.
Researchers used the National Alzheimer’s Coordinating Center data with 43,630 patients and analyzed sex differences between men and women with AF and their performance on neuropsychological tests and cognitive disease progression.
Higher odds of dementia, MCI in women
According to the paper, AF is associated with higher odds of dementia (odds ratio [OR], 3.00; 95% confidence interval [CI], 1.22-7.37) in women and MCI in women (OR, 3.43; 95% CI, 1.55-7.55) compared with men.
Women with AF and normal cognition at baseline had a higher risk of disease progression (hazard ratio [HR], 1.26; 95% CI, 1.06-1.50) from normal to MCI and from MCI to vascular dementia (HR, 3.27; 95% CI, 1.89-5.65) than that of men with AF or men and women without AF.
AF is a major public health problem linked with stroke and heart failure, and is an independent risk factor of increased mortality. It is associated with higher risk of cognitive impairment and dementia independent of stroke history.
Cognitive screening for AF patients
The authors wrote that cognitive screening, especially in women, should be part of yearly cardiology visits for patients with AF to help identify early those at highest risk for cognitive disease.
T. Jared Bunch, MD, professor of medicine in the division of cardiovascular medicine at University of Utah in Salt Lake City, said in an interview, “We have learned that how we treat atrial fibrillation can influence risk.”
First, he said, outcomes, including brain health, are better when rhythm control approaches are used within the first year of diagnosis.
“Restoring a normal heart rhythm improves brain perfusion and cognitive function. Next, aggressive rhythm control – such as catheter ablation – is associated with much lower long-term risks of dementia in the [patients]. Finally, early and effective use of anticoagulation in patients with atrial fibrillation lowers risk of stroke, dementia, and cognitive decline.”
Several factors unknown
Dr. Bunch said there are some unknowns in the study, such as how long patients were in atrial fibrillation.
He said one way to address the inequities is to refer women earlier as women are often referred later in disease to specialty care, which can have consequences.
He said it is not known how many people underwent early and effective rhythm control.
“Women also are less likely to receive catheter ablation, a cardioversion, or be placed on antiarrhythmic drugs,” said Dr. Bunch, who was not part of the study. “These also represent potential opportunities to improve outcomes by treating the rhythm in a similar and aggressive manner in both men and women.”
Also unknown is how many people were on effective oral anticoagulation, Dr. Bunch noted.
The study importantly highlights a significant problem surrounding the care of women with AF, he said, but there are strategies to improve outcomes.
In addition to earlier screening and referral for women, providers should recognize that men and women may present differently with different AF symptoms. He added that physicians should offer catheter ablation, the most effective treatment, equally to men and women who are candidates.
In all people, he said, it’s important “to start anticoagulation very early in the disease to lower the risk of micro- and macrothrombotic events that lead to poor brain health and function.”
The study authors and Dr. Bunch declared no relevant financial relationships.
New data suggest a significantly stronger link in women compared with men between atrial fibrillation (AF) and mild cognitive impairment (MCI) and dementia.
“Our findings imply that women with AF may be at higher risk for MCI and dementia with potentially more rapid disease progression from normal cognition to MCI or dementia than women without AF or men with and without AF,” wrote authors of a new study led by Kathryn A. Wood, PhD, RN, Neil Hodgson Woodruff School of Nursing at Emory University in Atlanta.
The findings were published online in Alzheimer’s & Dementia.
Researchers used the National Alzheimer’s Coordinating Center data with 43,630 patients and analyzed sex differences between men and women with AF and their performance on neuropsychological tests and cognitive disease progression.
Higher odds of dementia, MCI in women
According to the paper, AF is associated with higher odds of dementia (odds ratio [OR], 3.00; 95% confidence interval [CI], 1.22-7.37) in women and MCI in women (OR, 3.43; 95% CI, 1.55-7.55) compared with men.
Women with AF and normal cognition at baseline had a higher risk of disease progression (hazard ratio [HR], 1.26; 95% CI, 1.06-1.50) from normal to MCI and from MCI to vascular dementia (HR, 3.27; 95% CI, 1.89-5.65) than that of men with AF or men and women without AF.
AF is a major public health problem linked with stroke and heart failure, and is an independent risk factor of increased mortality. It is associated with higher risk of cognitive impairment and dementia independent of stroke history.
Cognitive screening for AF patients
The authors wrote that cognitive screening, especially in women, should be part of yearly cardiology visits for patients with AF to help identify early those at highest risk for cognitive disease.
T. Jared Bunch, MD, professor of medicine in the division of cardiovascular medicine at University of Utah in Salt Lake City, said in an interview, “We have learned that how we treat atrial fibrillation can influence risk.”
First, he said, outcomes, including brain health, are better when rhythm control approaches are used within the first year of diagnosis.
“Restoring a normal heart rhythm improves brain perfusion and cognitive function. Next, aggressive rhythm control – such as catheter ablation – is associated with much lower long-term risks of dementia in the [patients]. Finally, early and effective use of anticoagulation in patients with atrial fibrillation lowers risk of stroke, dementia, and cognitive decline.”
Several factors unknown
Dr. Bunch said there are some unknowns in the study, such as how long patients were in atrial fibrillation.
He said one way to address the inequities is to refer women earlier as women are often referred later in disease to specialty care, which can have consequences.
He said it is not known how many people underwent early and effective rhythm control.
“Women also are less likely to receive catheter ablation, a cardioversion, or be placed on antiarrhythmic drugs,” said Dr. Bunch, who was not part of the study. “These also represent potential opportunities to improve outcomes by treating the rhythm in a similar and aggressive manner in both men and women.”
Also unknown is how many people were on effective oral anticoagulation, Dr. Bunch noted.
The study importantly highlights a significant problem surrounding the care of women with AF, he said, but there are strategies to improve outcomes.
In addition to earlier screening and referral for women, providers should recognize that men and women may present differently with different AF symptoms. He added that physicians should offer catheter ablation, the most effective treatment, equally to men and women who are candidates.
In all people, he said, it’s important “to start anticoagulation very early in the disease to lower the risk of micro- and macrothrombotic events that lead to poor brain health and function.”
The study authors and Dr. Bunch declared no relevant financial relationships.
New data suggest a significantly stronger link in women compared with men between atrial fibrillation (AF) and mild cognitive impairment (MCI) and dementia.
“Our findings imply that women with AF may be at higher risk for MCI and dementia with potentially more rapid disease progression from normal cognition to MCI or dementia than women without AF or men with and without AF,” wrote authors of a new study led by Kathryn A. Wood, PhD, RN, Neil Hodgson Woodruff School of Nursing at Emory University in Atlanta.
The findings were published online in Alzheimer’s & Dementia.
Researchers used the National Alzheimer’s Coordinating Center data with 43,630 patients and analyzed sex differences between men and women with AF and their performance on neuropsychological tests and cognitive disease progression.
Higher odds of dementia, MCI in women
According to the paper, AF is associated with higher odds of dementia (odds ratio [OR], 3.00; 95% confidence interval [CI], 1.22-7.37) in women and MCI in women (OR, 3.43; 95% CI, 1.55-7.55) compared with men.
Women with AF and normal cognition at baseline had a higher risk of disease progression (hazard ratio [HR], 1.26; 95% CI, 1.06-1.50) from normal to MCI and from MCI to vascular dementia (HR, 3.27; 95% CI, 1.89-5.65) than that of men with AF or men and women without AF.
AF is a major public health problem linked with stroke and heart failure, and is an independent risk factor of increased mortality. It is associated with higher risk of cognitive impairment and dementia independent of stroke history.
Cognitive screening for AF patients
The authors wrote that cognitive screening, especially in women, should be part of yearly cardiology visits for patients with AF to help identify early those at highest risk for cognitive disease.
T. Jared Bunch, MD, professor of medicine in the division of cardiovascular medicine at University of Utah in Salt Lake City, said in an interview, “We have learned that how we treat atrial fibrillation can influence risk.”
First, he said, outcomes, including brain health, are better when rhythm control approaches are used within the first year of diagnosis.
“Restoring a normal heart rhythm improves brain perfusion and cognitive function. Next, aggressive rhythm control – such as catheter ablation – is associated with much lower long-term risks of dementia in the [patients]. Finally, early and effective use of anticoagulation in patients with atrial fibrillation lowers risk of stroke, dementia, and cognitive decline.”
Several factors unknown
Dr. Bunch said there are some unknowns in the study, such as how long patients were in atrial fibrillation.
He said one way to address the inequities is to refer women earlier as women are often referred later in disease to specialty care, which can have consequences.
He said it is not known how many people underwent early and effective rhythm control.
“Women also are less likely to receive catheter ablation, a cardioversion, or be placed on antiarrhythmic drugs,” said Dr. Bunch, who was not part of the study. “These also represent potential opportunities to improve outcomes by treating the rhythm in a similar and aggressive manner in both men and women.”
Also unknown is how many people were on effective oral anticoagulation, Dr. Bunch noted.
The study importantly highlights a significant problem surrounding the care of women with AF, he said, but there are strategies to improve outcomes.
In addition to earlier screening and referral for women, providers should recognize that men and women may present differently with different AF symptoms. He added that physicians should offer catheter ablation, the most effective treatment, equally to men and women who are candidates.
In all people, he said, it’s important “to start anticoagulation very early in the disease to lower the risk of micro- and macrothrombotic events that lead to poor brain health and function.”
The study authors and Dr. Bunch declared no relevant financial relationships.
FROM ALZHEIMER’S & DEMENTIA
New law allows international medical graduates to bypass U.S. residency
Pediatric nephrologist Bryan Carmody, MD, recalls working alongside an extremely experienced neonatologist during his residency. She had managed a neonatal intensive care unit in her home country of Lithuania, but because she wanted to practice in the United States, it took years of repeat training before she was eligible for a medical license.
“She was very accomplished, and she was wonderful to have as a coresident at the time,” Dr. Carmody said in an interview.
The neonatologist now practices at a U.S. academic medical center, but to obtain that position, she had to complete 3 years of pediatric residency and 3 years of fellowship in the United States, Dr. Carmody said.
Such training for international medical graduates (IMGs) is a routine part of obtaining a U.S. medical license, but
The American Medical Association took similar measures at its recent annual meeting, making it easier for IMGs to gain licensure. Because the pandemic and Russia’s invasion of Ukraine disrupted the process by which some IMGs had their licenses verified, the AMA is now encouraging state licensing boards and other credentialing institutions to accept certification from the Educational Commission for Foreign Medical Graduates as verification, rather than requiring documents directly from international medical schools.
When it comes to Tennessee’s new law, signed by Gov. Bill Lee in April, experienced IMGs who have received medical training abroad can skip U.S. residency requirements and obtain a temporary license to practice medicine in Tennessee if they meet certain qualifications.
The international doctors must demonstrate competency, as determined by the state medical board. In addition, they must have completed a 3-year postgraduate training program in the graduate’s licensing country or otherwise have practiced as a medical professional in which they performed the duties of a physician for at least 3 of the past 5 years outside the United States, according to the new law.
To be approved, IMGs must also have received an employment offer from a Tennessee health care provider that has a residency program accredited by the Accreditation Council for Graduate Medical Education.
If physicians remain in good standing for 2 years, the board will grant them a full and unrestricted license to practice in Tennessee.
“The new legislation opens up a lot of doors for international medical graduates and is also a lifeline for a lot of underserved areas in Tennessee,” said Asim Ansari, MD, a Canadian who attended medical school in the Caribbean and is an advocate for IMGs.
Dr. Ansari is participating in a child and adolescent psychiatry fellowship at the University of Kansas Medical Center, Kansas City, until he can apply for the sixth time to a residency program. “This could possibly be a model that other states may want to implement in a few years.”
What’s behind the law?
A predicted physician shortage in Tennessee drove the legislation, said Rep. Sabi “Doc” Kumar, MD, vice chair for the Tennessee House Health Committee and a cosponsor of the legislation. Legislators hope the law will mitigate that shortage and boost the number of physicians practicing in underserved areas of the state.
“Considering that one in four physicians in the U.S. are international medical gradates, it was important for us to be able to attract those physicians to Tennessee,” he said.
The Tennessee Board of Medical Examiners will develop administrative rules for the law, which may take up to a year, Rep. Kumar said. He expects the program to be available to IMGs beginning in mid-2024.
Upon completion of the program, IMGs will be able to practice general medicine in Tennessee, not a specialty. Requirements for specialty certification would have to be met through the specialties’ respective boards.
Dr. Carmody, who blogs about medical education, including the new legislation, said in an interview the law will greatly benefit experienced IMGs, who often are bypassed as residency candidates because they graduated years ago. Hospitals also win because they can fill positions that otherwise might sit vacant, he said.
Family physician Sahil Bawa, MD, an IMG from India who recently matched into his specialty, said the Tennessee legislation will help fellow IMGs find U.S. medical jobs.
“It’s very difficult for IMGs to get into residency in the U.S.,” he said. “I’ve seen people with medical degrees from other countries drive Uber or do odd jobs to sustain themselves here. I’ve known a few people who have left and gone back to their home country because they were not accepted into a residency.”
Who benefits most?
Dr. Bawa noted that the legislation would not have helped him, as he needed a visa to practice in the United States and the law does not include the sponsoring of visas. The legislation requires IMGs to show evidence of citizenship or evidence that they are legally entitled to live or work in the United States.
U.S. citizen IMGs who haven’t completed residency or who practiced in another country also are left out of the law, Dr. Carmody said.
“This law is designed to take the most accomplished cream of the crop international medical graduates with the most experience and the most sophisticated skill set and send them to Tennessee. I think that’s the intent,” he said. “But many international medical graduates are U.S. citizens who don’t have the opportunity to practice in countries other than United States or do residencies. A lot of these people are sitting on the sidelines, unable to secure residency positions. I’m sure they would be desperate for a program like this.”
Questions remain
“Just because the doctor can get a [temporary] license without the training doesn’t mean employers are going to be interested in sponsoring those doctors,” said Adam Cohen, an immigration attorney who practices in Memphis. “What is the inclination of these employers to hire these physicians who have undergone training outside the U.S.? And will there be skepticism on the part of employers about the competence of these doctors?”
“Hospital systems will be able to hire experienced practitioners for a very low cost,” Dr. Ansari said. “So now you have these additional bodies who can do the work of a physician, but you don’t have to pay them as much as a physician for 2 years. And because some are desperate to work, they will take lower pay as long as they have a pathway to full licensure in Tennessee. What are the protections for these physicians? Who will cover their insurance? Who will be responsible for them, the attendees? And will the attendees be willing to put their license on the line for them?”
In addition, Dr. Carmody questions what, if anything, will encourage IMGs to work in underserved areas in Tennessee after their 2 years are up and whether there will be any incentives to guide them. He wonders, too, whether the physicians will be stuck practicing in Tennessee following completion of the program.
“Will these physicians only be able to work in Tennessee?” he asked. “I think that’s probably going to be the case, because they’ll be licensed in Tennessee, but to go to another state, they would be missing the required residency training. So it might be these folks are stuck in Tennessee unless other states develop reciprocal arrangements.”
Other states would have to decide whether to recognize the Tennessee license acquired through this pathway, Rep. Kumar said.
He explained that the sponsoring sites would be responsible for providing work-hour restrictions and liability protections. There are currently no incentives in the legislation for IMGs to practice in rural, underserved areas, but the hospitals and communities there generally offer incentives when recruiting, Rep. Kumar said.
“The law definitely has the potential to be helpful,” Mr. Cohen said, “because there’s an ability to place providers in the state without having to go through the bottleneck of limited residency slots. If other states see a positive effect on Tennessee or are exploring ways to alleviate their own shortages, it’s possible [they] might follow suit.”
Rep. Kumar agreed that other states will be watching Tennessee to weigh the law’s success.
“I think the law will have to prove itself and show that Tennessee has benefited from it and that the results have been good,” he said. “We are providing a pioneering way for attracting medical graduates and making it easier for them to obtain a license. I would think other states would want to do that.”
A version of this article first appeared on Medscape.com.
Pediatric nephrologist Bryan Carmody, MD, recalls working alongside an extremely experienced neonatologist during his residency. She had managed a neonatal intensive care unit in her home country of Lithuania, but because she wanted to practice in the United States, it took years of repeat training before she was eligible for a medical license.
“She was very accomplished, and she was wonderful to have as a coresident at the time,” Dr. Carmody said in an interview.
The neonatologist now practices at a U.S. academic medical center, but to obtain that position, she had to complete 3 years of pediatric residency and 3 years of fellowship in the United States, Dr. Carmody said.
Such training for international medical graduates (IMGs) is a routine part of obtaining a U.S. medical license, but
The American Medical Association took similar measures at its recent annual meeting, making it easier for IMGs to gain licensure. Because the pandemic and Russia’s invasion of Ukraine disrupted the process by which some IMGs had their licenses verified, the AMA is now encouraging state licensing boards and other credentialing institutions to accept certification from the Educational Commission for Foreign Medical Graduates as verification, rather than requiring documents directly from international medical schools.
When it comes to Tennessee’s new law, signed by Gov. Bill Lee in April, experienced IMGs who have received medical training abroad can skip U.S. residency requirements and obtain a temporary license to practice medicine in Tennessee if they meet certain qualifications.
The international doctors must demonstrate competency, as determined by the state medical board. In addition, they must have completed a 3-year postgraduate training program in the graduate’s licensing country or otherwise have practiced as a medical professional in which they performed the duties of a physician for at least 3 of the past 5 years outside the United States, according to the new law.
To be approved, IMGs must also have received an employment offer from a Tennessee health care provider that has a residency program accredited by the Accreditation Council for Graduate Medical Education.
If physicians remain in good standing for 2 years, the board will grant them a full and unrestricted license to practice in Tennessee.
“The new legislation opens up a lot of doors for international medical graduates and is also a lifeline for a lot of underserved areas in Tennessee,” said Asim Ansari, MD, a Canadian who attended medical school in the Caribbean and is an advocate for IMGs.
Dr. Ansari is participating in a child and adolescent psychiatry fellowship at the University of Kansas Medical Center, Kansas City, until he can apply for the sixth time to a residency program. “This could possibly be a model that other states may want to implement in a few years.”
What’s behind the law?
A predicted physician shortage in Tennessee drove the legislation, said Rep. Sabi “Doc” Kumar, MD, vice chair for the Tennessee House Health Committee and a cosponsor of the legislation. Legislators hope the law will mitigate that shortage and boost the number of physicians practicing in underserved areas of the state.
“Considering that one in four physicians in the U.S. are international medical gradates, it was important for us to be able to attract those physicians to Tennessee,” he said.
The Tennessee Board of Medical Examiners will develop administrative rules for the law, which may take up to a year, Rep. Kumar said. He expects the program to be available to IMGs beginning in mid-2024.
Upon completion of the program, IMGs will be able to practice general medicine in Tennessee, not a specialty. Requirements for specialty certification would have to be met through the specialties’ respective boards.
Dr. Carmody, who blogs about medical education, including the new legislation, said in an interview the law will greatly benefit experienced IMGs, who often are bypassed as residency candidates because they graduated years ago. Hospitals also win because they can fill positions that otherwise might sit vacant, he said.
Family physician Sahil Bawa, MD, an IMG from India who recently matched into his specialty, said the Tennessee legislation will help fellow IMGs find U.S. medical jobs.
“It’s very difficult for IMGs to get into residency in the U.S.,” he said. “I’ve seen people with medical degrees from other countries drive Uber or do odd jobs to sustain themselves here. I’ve known a few people who have left and gone back to their home country because they were not accepted into a residency.”
Who benefits most?
Dr. Bawa noted that the legislation would not have helped him, as he needed a visa to practice in the United States and the law does not include the sponsoring of visas. The legislation requires IMGs to show evidence of citizenship or evidence that they are legally entitled to live or work in the United States.
U.S. citizen IMGs who haven’t completed residency or who practiced in another country also are left out of the law, Dr. Carmody said.
“This law is designed to take the most accomplished cream of the crop international medical graduates with the most experience and the most sophisticated skill set and send them to Tennessee. I think that’s the intent,” he said. “But many international medical graduates are U.S. citizens who don’t have the opportunity to practice in countries other than United States or do residencies. A lot of these people are sitting on the sidelines, unable to secure residency positions. I’m sure they would be desperate for a program like this.”
Questions remain
“Just because the doctor can get a [temporary] license without the training doesn’t mean employers are going to be interested in sponsoring those doctors,” said Adam Cohen, an immigration attorney who practices in Memphis. “What is the inclination of these employers to hire these physicians who have undergone training outside the U.S.? And will there be skepticism on the part of employers about the competence of these doctors?”
“Hospital systems will be able to hire experienced practitioners for a very low cost,” Dr. Ansari said. “So now you have these additional bodies who can do the work of a physician, but you don’t have to pay them as much as a physician for 2 years. And because some are desperate to work, they will take lower pay as long as they have a pathway to full licensure in Tennessee. What are the protections for these physicians? Who will cover their insurance? Who will be responsible for them, the attendees? And will the attendees be willing to put their license on the line for them?”
In addition, Dr. Carmody questions what, if anything, will encourage IMGs to work in underserved areas in Tennessee after their 2 years are up and whether there will be any incentives to guide them. He wonders, too, whether the physicians will be stuck practicing in Tennessee following completion of the program.
“Will these physicians only be able to work in Tennessee?” he asked. “I think that’s probably going to be the case, because they’ll be licensed in Tennessee, but to go to another state, they would be missing the required residency training. So it might be these folks are stuck in Tennessee unless other states develop reciprocal arrangements.”
Other states would have to decide whether to recognize the Tennessee license acquired through this pathway, Rep. Kumar said.
He explained that the sponsoring sites would be responsible for providing work-hour restrictions and liability protections. There are currently no incentives in the legislation for IMGs to practice in rural, underserved areas, but the hospitals and communities there generally offer incentives when recruiting, Rep. Kumar said.
“The law definitely has the potential to be helpful,” Mr. Cohen said, “because there’s an ability to place providers in the state without having to go through the bottleneck of limited residency slots. If other states see a positive effect on Tennessee or are exploring ways to alleviate their own shortages, it’s possible [they] might follow suit.”
Rep. Kumar agreed that other states will be watching Tennessee to weigh the law’s success.
“I think the law will have to prove itself and show that Tennessee has benefited from it and that the results have been good,” he said. “We are providing a pioneering way for attracting medical graduates and making it easier for them to obtain a license. I would think other states would want to do that.”
A version of this article first appeared on Medscape.com.
Pediatric nephrologist Bryan Carmody, MD, recalls working alongside an extremely experienced neonatologist during his residency. She had managed a neonatal intensive care unit in her home country of Lithuania, but because she wanted to practice in the United States, it took years of repeat training before she was eligible for a medical license.
“She was very accomplished, and she was wonderful to have as a coresident at the time,” Dr. Carmody said in an interview.
The neonatologist now practices at a U.S. academic medical center, but to obtain that position, she had to complete 3 years of pediatric residency and 3 years of fellowship in the United States, Dr. Carmody said.
Such training for international medical graduates (IMGs) is a routine part of obtaining a U.S. medical license, but
The American Medical Association took similar measures at its recent annual meeting, making it easier for IMGs to gain licensure. Because the pandemic and Russia’s invasion of Ukraine disrupted the process by which some IMGs had their licenses verified, the AMA is now encouraging state licensing boards and other credentialing institutions to accept certification from the Educational Commission for Foreign Medical Graduates as verification, rather than requiring documents directly from international medical schools.
When it comes to Tennessee’s new law, signed by Gov. Bill Lee in April, experienced IMGs who have received medical training abroad can skip U.S. residency requirements and obtain a temporary license to practice medicine in Tennessee if they meet certain qualifications.
The international doctors must demonstrate competency, as determined by the state medical board. In addition, they must have completed a 3-year postgraduate training program in the graduate’s licensing country or otherwise have practiced as a medical professional in which they performed the duties of a physician for at least 3 of the past 5 years outside the United States, according to the new law.
To be approved, IMGs must also have received an employment offer from a Tennessee health care provider that has a residency program accredited by the Accreditation Council for Graduate Medical Education.
If physicians remain in good standing for 2 years, the board will grant them a full and unrestricted license to practice in Tennessee.
“The new legislation opens up a lot of doors for international medical graduates and is also a lifeline for a lot of underserved areas in Tennessee,” said Asim Ansari, MD, a Canadian who attended medical school in the Caribbean and is an advocate for IMGs.
Dr. Ansari is participating in a child and adolescent psychiatry fellowship at the University of Kansas Medical Center, Kansas City, until he can apply for the sixth time to a residency program. “This could possibly be a model that other states may want to implement in a few years.”
What’s behind the law?
A predicted physician shortage in Tennessee drove the legislation, said Rep. Sabi “Doc” Kumar, MD, vice chair for the Tennessee House Health Committee and a cosponsor of the legislation. Legislators hope the law will mitigate that shortage and boost the number of physicians practicing in underserved areas of the state.
“Considering that one in four physicians in the U.S. are international medical gradates, it was important for us to be able to attract those physicians to Tennessee,” he said.
The Tennessee Board of Medical Examiners will develop administrative rules for the law, which may take up to a year, Rep. Kumar said. He expects the program to be available to IMGs beginning in mid-2024.
Upon completion of the program, IMGs will be able to practice general medicine in Tennessee, not a specialty. Requirements for specialty certification would have to be met through the specialties’ respective boards.
Dr. Carmody, who blogs about medical education, including the new legislation, said in an interview the law will greatly benefit experienced IMGs, who often are bypassed as residency candidates because they graduated years ago. Hospitals also win because they can fill positions that otherwise might sit vacant, he said.
Family physician Sahil Bawa, MD, an IMG from India who recently matched into his specialty, said the Tennessee legislation will help fellow IMGs find U.S. medical jobs.
“It’s very difficult for IMGs to get into residency in the U.S.,” he said. “I’ve seen people with medical degrees from other countries drive Uber or do odd jobs to sustain themselves here. I’ve known a few people who have left and gone back to their home country because they were not accepted into a residency.”
Who benefits most?
Dr. Bawa noted that the legislation would not have helped him, as he needed a visa to practice in the United States and the law does not include the sponsoring of visas. The legislation requires IMGs to show evidence of citizenship or evidence that they are legally entitled to live or work in the United States.
U.S. citizen IMGs who haven’t completed residency or who practiced in another country also are left out of the law, Dr. Carmody said.
“This law is designed to take the most accomplished cream of the crop international medical graduates with the most experience and the most sophisticated skill set and send them to Tennessee. I think that’s the intent,” he said. “But many international medical graduates are U.S. citizens who don’t have the opportunity to practice in countries other than United States or do residencies. A lot of these people are sitting on the sidelines, unable to secure residency positions. I’m sure they would be desperate for a program like this.”
Questions remain
“Just because the doctor can get a [temporary] license without the training doesn’t mean employers are going to be interested in sponsoring those doctors,” said Adam Cohen, an immigration attorney who practices in Memphis. “What is the inclination of these employers to hire these physicians who have undergone training outside the U.S.? And will there be skepticism on the part of employers about the competence of these doctors?”
“Hospital systems will be able to hire experienced practitioners for a very low cost,” Dr. Ansari said. “So now you have these additional bodies who can do the work of a physician, but you don’t have to pay them as much as a physician for 2 years. And because some are desperate to work, they will take lower pay as long as they have a pathway to full licensure in Tennessee. What are the protections for these physicians? Who will cover their insurance? Who will be responsible for them, the attendees? And will the attendees be willing to put their license on the line for them?”
In addition, Dr. Carmody questions what, if anything, will encourage IMGs to work in underserved areas in Tennessee after their 2 years are up and whether there will be any incentives to guide them. He wonders, too, whether the physicians will be stuck practicing in Tennessee following completion of the program.
“Will these physicians only be able to work in Tennessee?” he asked. “I think that’s probably going to be the case, because they’ll be licensed in Tennessee, but to go to another state, they would be missing the required residency training. So it might be these folks are stuck in Tennessee unless other states develop reciprocal arrangements.”
Other states would have to decide whether to recognize the Tennessee license acquired through this pathway, Rep. Kumar said.
He explained that the sponsoring sites would be responsible for providing work-hour restrictions and liability protections. There are currently no incentives in the legislation for IMGs to practice in rural, underserved areas, but the hospitals and communities there generally offer incentives when recruiting, Rep. Kumar said.
“The law definitely has the potential to be helpful,” Mr. Cohen said, “because there’s an ability to place providers in the state without having to go through the bottleneck of limited residency slots. If other states see a positive effect on Tennessee or are exploring ways to alleviate their own shortages, it’s possible [they] might follow suit.”
Rep. Kumar agreed that other states will be watching Tennessee to weigh the law’s success.
“I think the law will have to prove itself and show that Tennessee has benefited from it and that the results have been good,” he said. “We are providing a pioneering way for attracting medical graduates and making it easier for them to obtain a license. I would think other states would want to do that.”
A version of this article first appeared on Medscape.com.
Migraine device expands treatment possibilities
AUSTIN, TEX – Migraine treatment and prevention is challenging in any population, but some present even more difficulties. Pregnant women and pediatric patients are two such groups where physicians and patients may be hesitant to use drugs.
Neuromodulation devices are proven alternatives to medical interventions, and the remote electrical neuromodulation device Nerivio (Theranica) was cleared by the Food and Drug Administration for acute treatment of migraine patients aged 12 and over in 2021. In March 2023, the agency expanded the clearance to include prevention of migration in adolescents aged 12 and over as well as adults.
Two studies presented at the annual meeting of the American Headache Society showed The latter study yielded similar findings to adults and was used by FDA in its decision to expand the device’s indication in adolescents in 2023, according to Teshamae Monteith, MD, who presented the study at a poster session.
The device, worn on the arm, allows the user to modulate the intensity of the stimulation so that it activates nociceptive pain receptors, but not in a painful way. “Each [patient] raises the intensity until it feels strong, yet comfortable, and when that happens, they activate the nociceptive receptors and the arm sends a signal all the way back up to the brainstem, where the pain control area is. Activating it causes the release of neurotransmitters that inhibit pain. That inhibition is a global pain inhibition mechanism, which causes inhibition of the migraine pain, and also the symptoms associated with migraine like photophobia and vomiting,” said Alit Stark-Inbar, PhD, who presented the study of treatment of pregnant women during a poster session.
Declining treatment days over time in adolescents
Dr. Monteith’s team studied high-frequency remote electrical neuromodulation device use in adolescents who had migraine on 10 days or more per month. They also required at least three treatment days in months 2 and 3 to control for the possibility that patients might stop using the device because they couldn’t afford it or for some reason other than efficacy or because their migraines went away.
The study included 83 adolescents aged 12-17 (mean, 15.9 years, 89% female). In the first month of use, the mean number of migraine treatment days was 12.6, which dropped to 9.0 in month 2 (P < .001), and 7.4 in month 3 (P < .001 from month 2). At 2 hours after treatment, 61.9% had pain relief, 24.5% had freedom from pain, 67.4% had functional disability relief, and 41.3% had functional disability freedom.
“It parallels the findings of the randomized, sham-controlled study in adults. The safety profile was excellent with just one person complaining of minor discomfort of the arm that resolved after treatment. The combination of the exceedingly safe profile and the likelihood of efficacy based on using monthly migraine treatment days as a proxy, the FDA decided to clear this for an adolescent indication,” said Dr. Monteith, associate professor of clinical neurology and chief of the headache division at the University of Miami.
The device design is convenient, according to Dr. Monteith. “The arm is just an easy place to stimulate. It’s a wearable device, and it’s 45 minutes [of treatment] and it’s app controlled. You know adolescents like their technology. They can track their symptoms here, and there’s some biobehavioral power to this because they can do biobehavioral exercises in addition to receiving the simulation,” she said.
The fact that the device is discrete is also an advantage for adolescents in school. “You have to go to the nurse to get your medication versus a device, you can just put it on, it’s easy, no one sees it, and no one’s making fun of you,” said Dr. Monteith.
Advantages for adolescents
The device offers a useful alternative to medication, according to Alan M. Rapoport, MD, who was asked for comment on the adolescent study. “I’d rather not give medication and certainly not preventive medication to an adolescent,” he said. He noted that over-the-counter acute care migraine medications such as aspirin or acetaminophen and combination medications with caffeine, as well as prescription medications such as triptans, “all have possible side effects, and when used to an increased extent can even cause medication overuse headache, increasing the severity and frequency of headache and migraine days per month,” Dr. Rapoport said. Using an effective device with almost no side effects is preferable to any of these acute care medications, especially if there are several headaches a month,” he said. Some newer medications that block calcitonin gene-related peptide might be quite effective when they are approved for adolescents, and should have few adverse events, he added.
In the past, Dr. Rapoport has favored biofeedback training for acute and especially preventive treatment of migraine in adolescents. “[Remote electrical neuromodulation] seems to do just as well, children enjoy it, and it’s easier for a patient to do at home,” said Dr. Rapoport.
Biofeedback training is usually taught to patients by a PhD psychologist. Once the patients have been on the biofeedback equipment and learn the techniques, they can practice on their own at home without equipment. “This new device treatment using Nerivio for acute care and prevention of migraine in adults and children 12 and older, where they can easily apply the device in almost any situation, whether they are at home or possibly even in school or out and about, looks very promising,” said Dr. Rapoport. It is quite effective and has almost no adverse events, which is what you really want, especially for adolescents,” he said.
Also asked to comment on the study of remote electrical neuromodulation use in adolescents, Abraham Avi Ashkenazi, MD, director of the Headache Clinic at Shaare Zedek Medical Center in Jerusalem, who attended the session, was enthusiastic, and said he has begun using it in his own practice. “It shows that remote electrical neuromodulation can not only be effective for the acute migraine attack, but also has a potential preventive effect on future migraine attacks. [This] actually makes sense, because we know that the more migraine attacks a person has, the more likely they are to progress to a more chronic form of the disease,” he said in an interview.
Asked what distinguishes REN from other neuromodulation therapies such as vagus nerve stimulation or transcranial magnetic stimulation (TMS), Dr. Ashkenazi said: “It’s just a different way of modulating the brain system via a different mechanism. In both ways, though, the advantage is that there are literally no adverse effects, as opposed to drug treatment.”
An alternative during pregnancy
Adolescents aren’t the only population where there is reluctance to use medication. Physicians have been prescribing the device for pregnant women, who are reluctant to take medication due to concerns effects on the fetus. However, pregnant women were not included in the pivotal studies. “They expect it to be safe. This study was done in order to validate that assumption. We reached out to women who either used the device during pregnancy or women from the same database who started it using afterwards, but did not use it during the pregnancy,” said Dr. Stark-Inbar, vice president of medical information at Theranica.
The study included 140 women, 59 in the remote electrical neuromodulation device group and 81 controls. The primary endpoint was gestational age, which was 38 weeks and 5 days in the remote electrical neuromodulation device group and 39 weeks among controls (P = .150). There were no significant between-group differences with respect to newborn birth weight, miscarriage rate, preterm birth rate, birth defect rate, developmental milestone rate, or emergency department visit rate.
Dr. Monteith and Dr. Ashkenazi have no relevant financial disclosures. Dr. Rapoport advises AbbVie, Biohaven, Cala Health, Dr. Reddy’s, Pfizer, Satsuma, Teva Pharmaceutical Industries, and Theranica. He is on the speakers bureau of AbbVie, Dr. Reddy’s, Impel, Pfizer and Teva Pharmaceutical Industries. Dr. Rapoport is the editor-in-chief of Neurology Reviews and on the editorial board of CNS Drugs.
AUSTIN, TEX – Migraine treatment and prevention is challenging in any population, but some present even more difficulties. Pregnant women and pediatric patients are two such groups where physicians and patients may be hesitant to use drugs.
Neuromodulation devices are proven alternatives to medical interventions, and the remote electrical neuromodulation device Nerivio (Theranica) was cleared by the Food and Drug Administration for acute treatment of migraine patients aged 12 and over in 2021. In March 2023, the agency expanded the clearance to include prevention of migration in adolescents aged 12 and over as well as adults.
Two studies presented at the annual meeting of the American Headache Society showed The latter study yielded similar findings to adults and was used by FDA in its decision to expand the device’s indication in adolescents in 2023, according to Teshamae Monteith, MD, who presented the study at a poster session.
The device, worn on the arm, allows the user to modulate the intensity of the stimulation so that it activates nociceptive pain receptors, but not in a painful way. “Each [patient] raises the intensity until it feels strong, yet comfortable, and when that happens, they activate the nociceptive receptors and the arm sends a signal all the way back up to the brainstem, where the pain control area is. Activating it causes the release of neurotransmitters that inhibit pain. That inhibition is a global pain inhibition mechanism, which causes inhibition of the migraine pain, and also the symptoms associated with migraine like photophobia and vomiting,” said Alit Stark-Inbar, PhD, who presented the study of treatment of pregnant women during a poster session.
Declining treatment days over time in adolescents
Dr. Monteith’s team studied high-frequency remote electrical neuromodulation device use in adolescents who had migraine on 10 days or more per month. They also required at least three treatment days in months 2 and 3 to control for the possibility that patients might stop using the device because they couldn’t afford it or for some reason other than efficacy or because their migraines went away.
The study included 83 adolescents aged 12-17 (mean, 15.9 years, 89% female). In the first month of use, the mean number of migraine treatment days was 12.6, which dropped to 9.0 in month 2 (P < .001), and 7.4 in month 3 (P < .001 from month 2). At 2 hours after treatment, 61.9% had pain relief, 24.5% had freedom from pain, 67.4% had functional disability relief, and 41.3% had functional disability freedom.
“It parallels the findings of the randomized, sham-controlled study in adults. The safety profile was excellent with just one person complaining of minor discomfort of the arm that resolved after treatment. The combination of the exceedingly safe profile and the likelihood of efficacy based on using monthly migraine treatment days as a proxy, the FDA decided to clear this for an adolescent indication,” said Dr. Monteith, associate professor of clinical neurology and chief of the headache division at the University of Miami.
The device design is convenient, according to Dr. Monteith. “The arm is just an easy place to stimulate. It’s a wearable device, and it’s 45 minutes [of treatment] and it’s app controlled. You know adolescents like their technology. They can track their symptoms here, and there’s some biobehavioral power to this because they can do biobehavioral exercises in addition to receiving the simulation,” she said.
The fact that the device is discrete is also an advantage for adolescents in school. “You have to go to the nurse to get your medication versus a device, you can just put it on, it’s easy, no one sees it, and no one’s making fun of you,” said Dr. Monteith.
Advantages for adolescents
The device offers a useful alternative to medication, according to Alan M. Rapoport, MD, who was asked for comment on the adolescent study. “I’d rather not give medication and certainly not preventive medication to an adolescent,” he said. He noted that over-the-counter acute care migraine medications such as aspirin or acetaminophen and combination medications with caffeine, as well as prescription medications such as triptans, “all have possible side effects, and when used to an increased extent can even cause medication overuse headache, increasing the severity and frequency of headache and migraine days per month,” Dr. Rapoport said. Using an effective device with almost no side effects is preferable to any of these acute care medications, especially if there are several headaches a month,” he said. Some newer medications that block calcitonin gene-related peptide might be quite effective when they are approved for adolescents, and should have few adverse events, he added.
In the past, Dr. Rapoport has favored biofeedback training for acute and especially preventive treatment of migraine in adolescents. “[Remote electrical neuromodulation] seems to do just as well, children enjoy it, and it’s easier for a patient to do at home,” said Dr. Rapoport.
Biofeedback training is usually taught to patients by a PhD psychologist. Once the patients have been on the biofeedback equipment and learn the techniques, they can practice on their own at home without equipment. “This new device treatment using Nerivio for acute care and prevention of migraine in adults and children 12 and older, where they can easily apply the device in almost any situation, whether they are at home or possibly even in school or out and about, looks very promising,” said Dr. Rapoport. It is quite effective and has almost no adverse events, which is what you really want, especially for adolescents,” he said.
Also asked to comment on the study of remote electrical neuromodulation use in adolescents, Abraham Avi Ashkenazi, MD, director of the Headache Clinic at Shaare Zedek Medical Center in Jerusalem, who attended the session, was enthusiastic, and said he has begun using it in his own practice. “It shows that remote electrical neuromodulation can not only be effective for the acute migraine attack, but also has a potential preventive effect on future migraine attacks. [This] actually makes sense, because we know that the more migraine attacks a person has, the more likely they are to progress to a more chronic form of the disease,” he said in an interview.
Asked what distinguishes REN from other neuromodulation therapies such as vagus nerve stimulation or transcranial magnetic stimulation (TMS), Dr. Ashkenazi said: “It’s just a different way of modulating the brain system via a different mechanism. In both ways, though, the advantage is that there are literally no adverse effects, as opposed to drug treatment.”
An alternative during pregnancy
Adolescents aren’t the only population where there is reluctance to use medication. Physicians have been prescribing the device for pregnant women, who are reluctant to take medication due to concerns effects on the fetus. However, pregnant women were not included in the pivotal studies. “They expect it to be safe. This study was done in order to validate that assumption. We reached out to women who either used the device during pregnancy or women from the same database who started it using afterwards, but did not use it during the pregnancy,” said Dr. Stark-Inbar, vice president of medical information at Theranica.
The study included 140 women, 59 in the remote electrical neuromodulation device group and 81 controls. The primary endpoint was gestational age, which was 38 weeks and 5 days in the remote electrical neuromodulation device group and 39 weeks among controls (P = .150). There were no significant between-group differences with respect to newborn birth weight, miscarriage rate, preterm birth rate, birth defect rate, developmental milestone rate, or emergency department visit rate.
Dr. Monteith and Dr. Ashkenazi have no relevant financial disclosures. Dr. Rapoport advises AbbVie, Biohaven, Cala Health, Dr. Reddy’s, Pfizer, Satsuma, Teva Pharmaceutical Industries, and Theranica. He is on the speakers bureau of AbbVie, Dr. Reddy’s, Impel, Pfizer and Teva Pharmaceutical Industries. Dr. Rapoport is the editor-in-chief of Neurology Reviews and on the editorial board of CNS Drugs.
AUSTIN, TEX – Migraine treatment and prevention is challenging in any population, but some present even more difficulties. Pregnant women and pediatric patients are two such groups where physicians and patients may be hesitant to use drugs.
Neuromodulation devices are proven alternatives to medical interventions, and the remote electrical neuromodulation device Nerivio (Theranica) was cleared by the Food and Drug Administration for acute treatment of migraine patients aged 12 and over in 2021. In March 2023, the agency expanded the clearance to include prevention of migration in adolescents aged 12 and over as well as adults.
Two studies presented at the annual meeting of the American Headache Society showed The latter study yielded similar findings to adults and was used by FDA in its decision to expand the device’s indication in adolescents in 2023, according to Teshamae Monteith, MD, who presented the study at a poster session.
The device, worn on the arm, allows the user to modulate the intensity of the stimulation so that it activates nociceptive pain receptors, but not in a painful way. “Each [patient] raises the intensity until it feels strong, yet comfortable, and when that happens, they activate the nociceptive receptors and the arm sends a signal all the way back up to the brainstem, where the pain control area is. Activating it causes the release of neurotransmitters that inhibit pain. That inhibition is a global pain inhibition mechanism, which causes inhibition of the migraine pain, and also the symptoms associated with migraine like photophobia and vomiting,” said Alit Stark-Inbar, PhD, who presented the study of treatment of pregnant women during a poster session.
Declining treatment days over time in adolescents
Dr. Monteith’s team studied high-frequency remote electrical neuromodulation device use in adolescents who had migraine on 10 days or more per month. They also required at least three treatment days in months 2 and 3 to control for the possibility that patients might stop using the device because they couldn’t afford it or for some reason other than efficacy or because their migraines went away.
The study included 83 adolescents aged 12-17 (mean, 15.9 years, 89% female). In the first month of use, the mean number of migraine treatment days was 12.6, which dropped to 9.0 in month 2 (P < .001), and 7.4 in month 3 (P < .001 from month 2). At 2 hours after treatment, 61.9% had pain relief, 24.5% had freedom from pain, 67.4% had functional disability relief, and 41.3% had functional disability freedom.
“It parallels the findings of the randomized, sham-controlled study in adults. The safety profile was excellent with just one person complaining of minor discomfort of the arm that resolved after treatment. The combination of the exceedingly safe profile and the likelihood of efficacy based on using monthly migraine treatment days as a proxy, the FDA decided to clear this for an adolescent indication,” said Dr. Monteith, associate professor of clinical neurology and chief of the headache division at the University of Miami.
The device design is convenient, according to Dr. Monteith. “The arm is just an easy place to stimulate. It’s a wearable device, and it’s 45 minutes [of treatment] and it’s app controlled. You know adolescents like their technology. They can track their symptoms here, and there’s some biobehavioral power to this because they can do biobehavioral exercises in addition to receiving the simulation,” she said.
The fact that the device is discrete is also an advantage for adolescents in school. “You have to go to the nurse to get your medication versus a device, you can just put it on, it’s easy, no one sees it, and no one’s making fun of you,” said Dr. Monteith.
Advantages for adolescents
The device offers a useful alternative to medication, according to Alan M. Rapoport, MD, who was asked for comment on the adolescent study. “I’d rather not give medication and certainly not preventive medication to an adolescent,” he said. He noted that over-the-counter acute care migraine medications such as aspirin or acetaminophen and combination medications with caffeine, as well as prescription medications such as triptans, “all have possible side effects, and when used to an increased extent can even cause medication overuse headache, increasing the severity and frequency of headache and migraine days per month,” Dr. Rapoport said. Using an effective device with almost no side effects is preferable to any of these acute care medications, especially if there are several headaches a month,” he said. Some newer medications that block calcitonin gene-related peptide might be quite effective when they are approved for adolescents, and should have few adverse events, he added.
In the past, Dr. Rapoport has favored biofeedback training for acute and especially preventive treatment of migraine in adolescents. “[Remote electrical neuromodulation] seems to do just as well, children enjoy it, and it’s easier for a patient to do at home,” said Dr. Rapoport.
Biofeedback training is usually taught to patients by a PhD psychologist. Once the patients have been on the biofeedback equipment and learn the techniques, they can practice on their own at home without equipment. “This new device treatment using Nerivio for acute care and prevention of migraine in adults and children 12 and older, where they can easily apply the device in almost any situation, whether they are at home or possibly even in school or out and about, looks very promising,” said Dr. Rapoport. It is quite effective and has almost no adverse events, which is what you really want, especially for adolescents,” he said.
Also asked to comment on the study of remote electrical neuromodulation use in adolescents, Abraham Avi Ashkenazi, MD, director of the Headache Clinic at Shaare Zedek Medical Center in Jerusalem, who attended the session, was enthusiastic, and said he has begun using it in his own practice. “It shows that remote electrical neuromodulation can not only be effective for the acute migraine attack, but also has a potential preventive effect on future migraine attacks. [This] actually makes sense, because we know that the more migraine attacks a person has, the more likely they are to progress to a more chronic form of the disease,” he said in an interview.
Asked what distinguishes REN from other neuromodulation therapies such as vagus nerve stimulation or transcranial magnetic stimulation (TMS), Dr. Ashkenazi said: “It’s just a different way of modulating the brain system via a different mechanism. In both ways, though, the advantage is that there are literally no adverse effects, as opposed to drug treatment.”
An alternative during pregnancy
Adolescents aren’t the only population where there is reluctance to use medication. Physicians have been prescribing the device for pregnant women, who are reluctant to take medication due to concerns effects on the fetus. However, pregnant women were not included in the pivotal studies. “They expect it to be safe. This study was done in order to validate that assumption. We reached out to women who either used the device during pregnancy or women from the same database who started it using afterwards, but did not use it during the pregnancy,” said Dr. Stark-Inbar, vice president of medical information at Theranica.
The study included 140 women, 59 in the remote electrical neuromodulation device group and 81 controls. The primary endpoint was gestational age, which was 38 weeks and 5 days in the remote electrical neuromodulation device group and 39 weeks among controls (P = .150). There were no significant between-group differences with respect to newborn birth weight, miscarriage rate, preterm birth rate, birth defect rate, developmental milestone rate, or emergency department visit rate.
Dr. Monteith and Dr. Ashkenazi have no relevant financial disclosures. Dr. Rapoport advises AbbVie, Biohaven, Cala Health, Dr. Reddy’s, Pfizer, Satsuma, Teva Pharmaceutical Industries, and Theranica. He is on the speakers bureau of AbbVie, Dr. Reddy’s, Impel, Pfizer and Teva Pharmaceutical Industries. Dr. Rapoport is the editor-in-chief of Neurology Reviews and on the editorial board of CNS Drugs.
AT AHS 2023
Can a repurposed Parkinson’s drug slow ALS progression?
However, at least one expert believes the study has “significant flaws.”
Investigators randomly assigned 20 individuals with sporadic ALS to receive either ropinirole or placebo for 24 weeks. During the double-blind period, there was no difference between the groups in terms of decline in functional status.
However, during a further open-label extension period, the ropinirole group showed significant suppression of functional decline and an average of an additional 7 months of progression-free survival.
The researchers were able to predict clinical responsiveness to ropinirole in vitro by analyzing motor neurons derived from participants’ stem cells.
“We found that ropinirole is safe and tolerable for ALS patients and shows therapeutic promise at helping them sustain daily activity and muscle strength,” first author Satoru Morimoto, MD, of the department of physiology, Keio University School of Medicine, Tokyo, said in a news release.
The study was published online in Cell Stem Cell.
Feasibility study
“ALS is totally incurable and it’s a very difficult disease to treat,” senior author Hideyuki Okano, MD, PhD, professor, department of physiology, Keio University, said in the news release.
Preclinical animal models have “limited translational potential” for identifying drug candidates, but induced pluripotent stem cell (iPSC)–derived motor neurons (MNs) from ALS patients can “overcome these limitations for drug screening,” the authors write.
“We previously identified ropinirole [a dopamine D2 receptor agonist] as a potential anti-ALS drug in vitro by iPSC drug discovery,” Dr. Okano said.
The current trial was a randomized, placebo-controlled phase 1/2a feasibility trial that evaluated the safety, tolerability, and efficacy of ropinirole in patients with ALS, using several parameters:
- The revised ALS functional rating scale (ALSFRS-R) score.
- Composite functional endpoints.
- Event-free survival.
- Time to ≤ 50% forced vital capacity (FVC).
The trial consisted of a 12-week run-in period, a 24-week double-blind period, an open-label extension period that lasted from 4 to 24 weeks, and a 4-week follow-up period after administration.
Thirteen patients were assigned to receive ropinirole (23.1% women; mean age, 65.2 ± 12.6 years; 7.7% with clinically definite and 76.9% with clinically probable ALS); seven were assigned to receive placebo (57.1% women; mean age, 66.3 ± 7.5 years; 14.3% with clinically definite and 85.7% with clinically probable ALS).
Of the treatment group, 30.8% had a bulbar onset lesion vs. 57.1% in the placebo group. At baseline, the mean FVC was 94.4% ± 14.9 and 81.5% ± 23.2 in the ropinirole and placebo groups, respectively. The mean body mass index (BMI) was 22.91 ± 3.82 and 19.69 ± 2.63, respectively.
Of the participants,12 in the ropinirole and six in the control group completed the full 24-week treatment protocol; 12 in the ropinirole and five in the placebo group completed the open-label extension (participants who had received placebo were switched to the active drug).
However only seven participants in the ropinirole group and one participant in the placebo group completed the full 1-year trial.
‘Striking correlation’
“During the double-blind period, muscle strength and daily activity were maintained, but a decline in the ALSFRS-R … was not different from that in the placebo group,” the researchers write.
In the open-label extension period, the ropinirole group showed “significant suppression of ALSFRS-R decline,” with an ALSFRS-R score change of only 7.75 (95% confidence interval, 10.66-4.63) for the treatment group vs. 17.51 (95% CI, 22.46-12.56) for the placebo group.
The researchers used the assessment of function and survival (CAFS) score, which adjusts the ALSFRS-R score against mortality, to see whether functional benefits translated into improved survival.
The score “favored ropinirole” in the open-extension period and the entire treatment period but not in the double-blind period.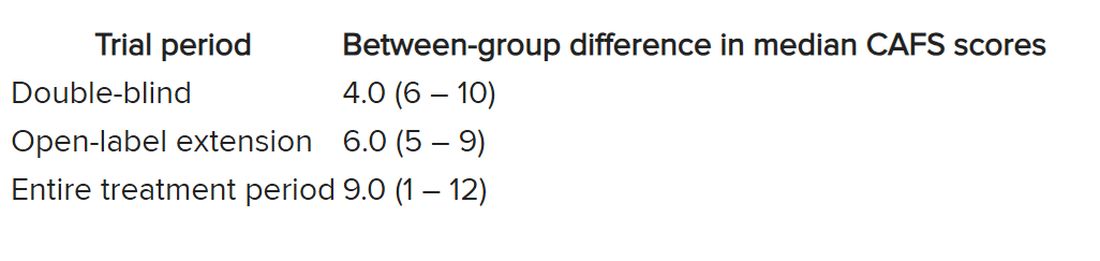
Disease progression events occurred in 7 of 7 (100%) participants in the placebo group and 7 of 13 (54%) in the ropinirole group, “suggesting a twofold decrease in disease progression” in the treatment group.
The ropinirole group experienced an additional 27.9 weeks of disease progression–free survival, compared with the placebo group.
“No participant discontinued treatment because of adverse experiences in either treatment group,” the authors report.
The analysis of iPSC-derived motor neurons from participants showed dopamine D2 receptor expression, as well as the potential involvement of the cholesterol pathway SREBP2 in the therapeutic effects of ropinirole. Lipid peroxide was also identified as a good “surrogate clinical marker to assess disease progression and drug efficacy.”
“We found a very striking correlation between a patient’s clinical response and the response of their motor neurons in vitro,” said Dr. Morimoto. “Patients whose motor neurons responded robustly to ropinirole in vitro had a much slower clinical disease progression with ropinirole treatment, while suboptimal responders showed much more rapid disease progression, despite taking ropinirole.”
Limitations include “small sample sizes and high attrition rates in the open-label extension period,” so “further validation” is required, the authors state.
Significant flaws
Commenting for this article, Carmel Armon, MD, MHS, professor of neurology, Loma Linda (Calif.) University, said the study “falls short of being a credible 1/2a clinical trial.”
Although the “intentions were good and the design not unusual,” the two groups were not “balanced on risk factors for faster progressing disease.” Rather, the placebo group was “tilted towards faster progressing disease” because there were more clinically definite and probable ALS patients in the placebo group than the treatment group, and there were more patients with bulbar onset.
Participants in the placebo group also had shorter median disease duration, lower BMI, and lower FVC, noted Dr. Armon, who was not involved with the study.
And only 1 in 7 control patients completed the open-label extension, compared with 7 of 13 patients in the intervention group.
“With these limitations, I would be disinclined to rely on the findings to justify a larger clinical trial,” Dr. Armon concluded.
The trial was sponsored by K Pharma. The study drug, active drugs, and placebo were supplied free of charge by GlaxoSmithKline K.K. Dr. Okano received grants from JSPS and AMED and grants and personal fees from K Pharma during the conduct of the study and personal fees from Sanbio, outside the submitted work. Dr. Okano has a patent on a therapeutic agent for ALS and composition for treatment licensed to K Pharma. The other authors’ disclosures and additional information are available in the original article. Dr. Armon reports no relevant financial relationships.
A version of this article first appeared on Medscape.com.
However, at least one expert believes the study has “significant flaws.”
Investigators randomly assigned 20 individuals with sporadic ALS to receive either ropinirole or placebo for 24 weeks. During the double-blind period, there was no difference between the groups in terms of decline in functional status.
However, during a further open-label extension period, the ropinirole group showed significant suppression of functional decline and an average of an additional 7 months of progression-free survival.
The researchers were able to predict clinical responsiveness to ropinirole in vitro by analyzing motor neurons derived from participants’ stem cells.
“We found that ropinirole is safe and tolerable for ALS patients and shows therapeutic promise at helping them sustain daily activity and muscle strength,” first author Satoru Morimoto, MD, of the department of physiology, Keio University School of Medicine, Tokyo, said in a news release.
The study was published online in Cell Stem Cell.
Feasibility study
“ALS is totally incurable and it’s a very difficult disease to treat,” senior author Hideyuki Okano, MD, PhD, professor, department of physiology, Keio University, said in the news release.
Preclinical animal models have “limited translational potential” for identifying drug candidates, but induced pluripotent stem cell (iPSC)–derived motor neurons (MNs) from ALS patients can “overcome these limitations for drug screening,” the authors write.
“We previously identified ropinirole [a dopamine D2 receptor agonist] as a potential anti-ALS drug in vitro by iPSC drug discovery,” Dr. Okano said.
The current trial was a randomized, placebo-controlled phase 1/2a feasibility trial that evaluated the safety, tolerability, and efficacy of ropinirole in patients with ALS, using several parameters:
- The revised ALS functional rating scale (ALSFRS-R) score.
- Composite functional endpoints.
- Event-free survival.
- Time to ≤ 50% forced vital capacity (FVC).
The trial consisted of a 12-week run-in period, a 24-week double-blind period, an open-label extension period that lasted from 4 to 24 weeks, and a 4-week follow-up period after administration.
Thirteen patients were assigned to receive ropinirole (23.1% women; mean age, 65.2 ± 12.6 years; 7.7% with clinically definite and 76.9% with clinically probable ALS); seven were assigned to receive placebo (57.1% women; mean age, 66.3 ± 7.5 years; 14.3% with clinically definite and 85.7% with clinically probable ALS).
Of the treatment group, 30.8% had a bulbar onset lesion vs. 57.1% in the placebo group. At baseline, the mean FVC was 94.4% ± 14.9 and 81.5% ± 23.2 in the ropinirole and placebo groups, respectively. The mean body mass index (BMI) was 22.91 ± 3.82 and 19.69 ± 2.63, respectively.
Of the participants,12 in the ropinirole and six in the control group completed the full 24-week treatment protocol; 12 in the ropinirole and five in the placebo group completed the open-label extension (participants who had received placebo were switched to the active drug).
However only seven participants in the ropinirole group and one participant in the placebo group completed the full 1-year trial.
‘Striking correlation’
“During the double-blind period, muscle strength and daily activity were maintained, but a decline in the ALSFRS-R … was not different from that in the placebo group,” the researchers write.
In the open-label extension period, the ropinirole group showed “significant suppression of ALSFRS-R decline,” with an ALSFRS-R score change of only 7.75 (95% confidence interval, 10.66-4.63) for the treatment group vs. 17.51 (95% CI, 22.46-12.56) for the placebo group.
The researchers used the assessment of function and survival (CAFS) score, which adjusts the ALSFRS-R score against mortality, to see whether functional benefits translated into improved survival.
The score “favored ropinirole” in the open-extension period and the entire treatment period but not in the double-blind period.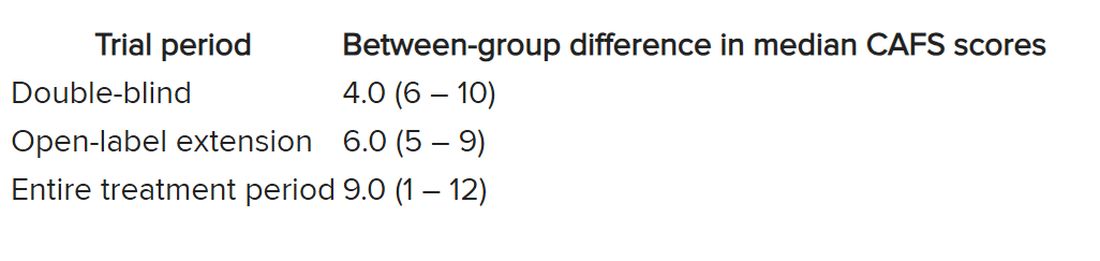
Disease progression events occurred in 7 of 7 (100%) participants in the placebo group and 7 of 13 (54%) in the ropinirole group, “suggesting a twofold decrease in disease progression” in the treatment group.
The ropinirole group experienced an additional 27.9 weeks of disease progression–free survival, compared with the placebo group.
“No participant discontinued treatment because of adverse experiences in either treatment group,” the authors report.
The analysis of iPSC-derived motor neurons from participants showed dopamine D2 receptor expression, as well as the potential involvement of the cholesterol pathway SREBP2 in the therapeutic effects of ropinirole. Lipid peroxide was also identified as a good “surrogate clinical marker to assess disease progression and drug efficacy.”
“We found a very striking correlation between a patient’s clinical response and the response of their motor neurons in vitro,” said Dr. Morimoto. “Patients whose motor neurons responded robustly to ropinirole in vitro had a much slower clinical disease progression with ropinirole treatment, while suboptimal responders showed much more rapid disease progression, despite taking ropinirole.”
Limitations include “small sample sizes and high attrition rates in the open-label extension period,” so “further validation” is required, the authors state.
Significant flaws
Commenting for this article, Carmel Armon, MD, MHS, professor of neurology, Loma Linda (Calif.) University, said the study “falls short of being a credible 1/2a clinical trial.”
Although the “intentions were good and the design not unusual,” the two groups were not “balanced on risk factors for faster progressing disease.” Rather, the placebo group was “tilted towards faster progressing disease” because there were more clinically definite and probable ALS patients in the placebo group than the treatment group, and there were more patients with bulbar onset.
Participants in the placebo group also had shorter median disease duration, lower BMI, and lower FVC, noted Dr. Armon, who was not involved with the study.
And only 1 in 7 control patients completed the open-label extension, compared with 7 of 13 patients in the intervention group.
“With these limitations, I would be disinclined to rely on the findings to justify a larger clinical trial,” Dr. Armon concluded.
The trial was sponsored by K Pharma. The study drug, active drugs, and placebo were supplied free of charge by GlaxoSmithKline K.K. Dr. Okano received grants from JSPS and AMED and grants and personal fees from K Pharma during the conduct of the study and personal fees from Sanbio, outside the submitted work. Dr. Okano has a patent on a therapeutic agent for ALS and composition for treatment licensed to K Pharma. The other authors’ disclosures and additional information are available in the original article. Dr. Armon reports no relevant financial relationships.
A version of this article first appeared on Medscape.com.
However, at least one expert believes the study has “significant flaws.”
Investigators randomly assigned 20 individuals with sporadic ALS to receive either ropinirole or placebo for 24 weeks. During the double-blind period, there was no difference between the groups in terms of decline in functional status.
However, during a further open-label extension period, the ropinirole group showed significant suppression of functional decline and an average of an additional 7 months of progression-free survival.
The researchers were able to predict clinical responsiveness to ropinirole in vitro by analyzing motor neurons derived from participants’ stem cells.
“We found that ropinirole is safe and tolerable for ALS patients and shows therapeutic promise at helping them sustain daily activity and muscle strength,” first author Satoru Morimoto, MD, of the department of physiology, Keio University School of Medicine, Tokyo, said in a news release.
The study was published online in Cell Stem Cell.
Feasibility study
“ALS is totally incurable and it’s a very difficult disease to treat,” senior author Hideyuki Okano, MD, PhD, professor, department of physiology, Keio University, said in the news release.
Preclinical animal models have “limited translational potential” for identifying drug candidates, but induced pluripotent stem cell (iPSC)–derived motor neurons (MNs) from ALS patients can “overcome these limitations for drug screening,” the authors write.
“We previously identified ropinirole [a dopamine D2 receptor agonist] as a potential anti-ALS drug in vitro by iPSC drug discovery,” Dr. Okano said.
The current trial was a randomized, placebo-controlled phase 1/2a feasibility trial that evaluated the safety, tolerability, and efficacy of ropinirole in patients with ALS, using several parameters:
- The revised ALS functional rating scale (ALSFRS-R) score.
- Composite functional endpoints.
- Event-free survival.
- Time to ≤ 50% forced vital capacity (FVC).
The trial consisted of a 12-week run-in period, a 24-week double-blind period, an open-label extension period that lasted from 4 to 24 weeks, and a 4-week follow-up period after administration.
Thirteen patients were assigned to receive ropinirole (23.1% women; mean age, 65.2 ± 12.6 years; 7.7% with clinically definite and 76.9% with clinically probable ALS); seven were assigned to receive placebo (57.1% women; mean age, 66.3 ± 7.5 years; 14.3% with clinically definite and 85.7% with clinically probable ALS).
Of the treatment group, 30.8% had a bulbar onset lesion vs. 57.1% in the placebo group. At baseline, the mean FVC was 94.4% ± 14.9 and 81.5% ± 23.2 in the ropinirole and placebo groups, respectively. The mean body mass index (BMI) was 22.91 ± 3.82 and 19.69 ± 2.63, respectively.
Of the participants,12 in the ropinirole and six in the control group completed the full 24-week treatment protocol; 12 in the ropinirole and five in the placebo group completed the open-label extension (participants who had received placebo were switched to the active drug).
However only seven participants in the ropinirole group and one participant in the placebo group completed the full 1-year trial.
‘Striking correlation’
“During the double-blind period, muscle strength and daily activity were maintained, but a decline in the ALSFRS-R … was not different from that in the placebo group,” the researchers write.
In the open-label extension period, the ropinirole group showed “significant suppression of ALSFRS-R decline,” with an ALSFRS-R score change of only 7.75 (95% confidence interval, 10.66-4.63) for the treatment group vs. 17.51 (95% CI, 22.46-12.56) for the placebo group.
The researchers used the assessment of function and survival (CAFS) score, which adjusts the ALSFRS-R score against mortality, to see whether functional benefits translated into improved survival.
The score “favored ropinirole” in the open-extension period and the entire treatment period but not in the double-blind period.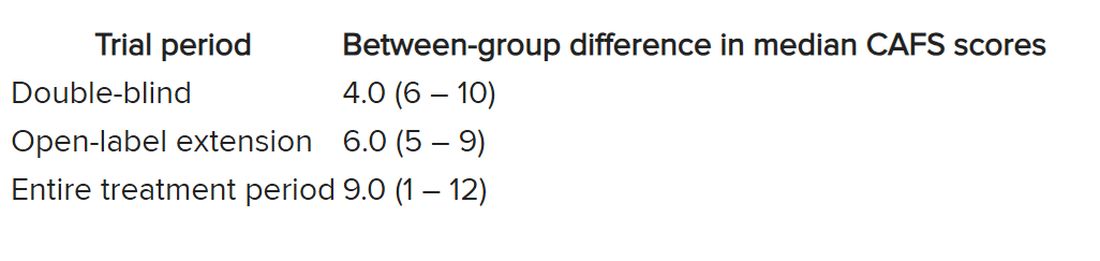
Disease progression events occurred in 7 of 7 (100%) participants in the placebo group and 7 of 13 (54%) in the ropinirole group, “suggesting a twofold decrease in disease progression” in the treatment group.
The ropinirole group experienced an additional 27.9 weeks of disease progression–free survival, compared with the placebo group.
“No participant discontinued treatment because of adverse experiences in either treatment group,” the authors report.
The analysis of iPSC-derived motor neurons from participants showed dopamine D2 receptor expression, as well as the potential involvement of the cholesterol pathway SREBP2 in the therapeutic effects of ropinirole. Lipid peroxide was also identified as a good “surrogate clinical marker to assess disease progression and drug efficacy.”
“We found a very striking correlation between a patient’s clinical response and the response of their motor neurons in vitro,” said Dr. Morimoto. “Patients whose motor neurons responded robustly to ropinirole in vitro had a much slower clinical disease progression with ropinirole treatment, while suboptimal responders showed much more rapid disease progression, despite taking ropinirole.”
Limitations include “small sample sizes and high attrition rates in the open-label extension period,” so “further validation” is required, the authors state.
Significant flaws
Commenting for this article, Carmel Armon, MD, MHS, professor of neurology, Loma Linda (Calif.) University, said the study “falls short of being a credible 1/2a clinical trial.”
Although the “intentions were good and the design not unusual,” the two groups were not “balanced on risk factors for faster progressing disease.” Rather, the placebo group was “tilted towards faster progressing disease” because there were more clinically definite and probable ALS patients in the placebo group than the treatment group, and there were more patients with bulbar onset.
Participants in the placebo group also had shorter median disease duration, lower BMI, and lower FVC, noted Dr. Armon, who was not involved with the study.
And only 1 in 7 control patients completed the open-label extension, compared with 7 of 13 patients in the intervention group.
“With these limitations, I would be disinclined to rely on the findings to justify a larger clinical trial,” Dr. Armon concluded.
The trial was sponsored by K Pharma. The study drug, active drugs, and placebo were supplied free of charge by GlaxoSmithKline K.K. Dr. Okano received grants from JSPS and AMED and grants and personal fees from K Pharma during the conduct of the study and personal fees from Sanbio, outside the submitted work. Dr. Okano has a patent on a therapeutic agent for ALS and composition for treatment licensed to K Pharma. The other authors’ disclosures and additional information are available in the original article. Dr. Armon reports no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FROM CELL STEM CELL
Regular napping linked to greater brain volume
Investigators at University College London, and the University of the Republic of Uruguay, Montevideo, found individuals genetically predisposed to regular napping had larger total brain volume, a surrogate of better cognitive health.
“Our results suggest that napping may improve brain health,” first author Valentina Paz, MSc, a PhD candidate at the University of the Republic of Uruguay said in an interview. “Specifically, our work revealed a 15.8 cubic cm increase in total brain volume with more frequent daytime napping,” she said.
The findings were published online in Sleep Health.
Higher brain volume
Previous studies examining the potential link between napping and cognition in older adults have yielded conflicting results.
To clarify this association, Ms. Paz and colleagues used Mendelian randomization to study DNA samples, cognitive outcomes, and functional magnetic resonance imaging data in participants from the ongoing UK Biobank Study.
Starting with data from 378,932 study participants (mean age 57), investigators compared measures of brain health and cognition of those who are more genetically programmed to nap with people who did not have these genetic variations.
More specifically, the investigators examined 97 sections of genetic code previously linked to the likelihood of regular napping and correlated these results with fMRI and cognitive outcomes between those genetically predisposed to take regular naps and those who weren’t.
Study outcomes included total brain volume, hippocampal volume, reaction time, and visual memory.
The final study sample included 35,080 with neuroimaging, cognitive assessment, and genotype data.
The researchers estimated that the average difference in brain volume between individuals genetically programmed to be habitual nappers and those who were not was equivalent to 15.8 cubic cm, or 2.6-6.5 years of aging.
However, there was no difference in the other three outcomes – hippocampal volume, reaction time, and visual processing – between the two study groups.
Since investigators did not have information on the length of time participants napped, Ms. Paz suggested that “taking a short nap in the early afternoon may help cognition in those needing it.”
However, she added, the study’s findings need to be replicated before any firm conclusions can be made.
“More work is needed to examine the associations between napping and cognition, and the replication of these findings using other datasets and methods,” she said.
The investigators note that the study’s findings augment the knowledge of the “impact of habitual daytime napping on brain health, which is essential to understanding cognitive impairment in the aging population. The lack of evidence for an association between napping and hippocampal volume and cognitive outcomes (for example, alertness) may be affected by habitual daytime napping and should be studied in the future.”
Strengths, limitations
Tara Spires-Jones, PhD, president of the British Neuroscience Association and group leader at the UK Dementia Research Institute, said, “the study shows a small but significant increase in brain volume in people who have a genetic signature associated with taking daytime naps.”
Dr. Spires-Jones, who was not involved in the research, noted that while the study is well-conducted, it has limitations. Because Mendelian randomization uses a genetic signature, she noted, outcomes depend on the accuracy of the signature.
“The napping habits of UK Biobank participants were self-reported, which might not be entirely accurate, and the ‘napping’ signature overlapped substantially with the signature for cognitive outcomes in the study, which makes the causal link weaker,” she said.
“Even with those limitations, this study is interesting because it adds to the data indicating that sleep is important for brain health,” said Dr. Spires-Jones.
The study was supported by Diabetes UK, the British Heart Foundation, and the Diabetes Research and Wellness Foundation. In Uruguay, it was supported by Programa de Desarrollo de las Ciencias Básicas, Agencia Nacional de Investigación e Innovación, Comisión Sectorial de Investigación Científica, and Comisión Académica de Posgrado. In the United States it was supported by the National Heart, Lung, and Blood Institute. There were no disclosures reported.
A version of this article first appeared on Medscape.com.
Investigators at University College London, and the University of the Republic of Uruguay, Montevideo, found individuals genetically predisposed to regular napping had larger total brain volume, a surrogate of better cognitive health.
“Our results suggest that napping may improve brain health,” first author Valentina Paz, MSc, a PhD candidate at the University of the Republic of Uruguay said in an interview. “Specifically, our work revealed a 15.8 cubic cm increase in total brain volume with more frequent daytime napping,” she said.
The findings were published online in Sleep Health.
Higher brain volume
Previous studies examining the potential link between napping and cognition in older adults have yielded conflicting results.
To clarify this association, Ms. Paz and colleagues used Mendelian randomization to study DNA samples, cognitive outcomes, and functional magnetic resonance imaging data in participants from the ongoing UK Biobank Study.
Starting with data from 378,932 study participants (mean age 57), investigators compared measures of brain health and cognition of those who are more genetically programmed to nap with people who did not have these genetic variations.
More specifically, the investigators examined 97 sections of genetic code previously linked to the likelihood of regular napping and correlated these results with fMRI and cognitive outcomes between those genetically predisposed to take regular naps and those who weren’t.
Study outcomes included total brain volume, hippocampal volume, reaction time, and visual memory.
The final study sample included 35,080 with neuroimaging, cognitive assessment, and genotype data.
The researchers estimated that the average difference in brain volume between individuals genetically programmed to be habitual nappers and those who were not was equivalent to 15.8 cubic cm, or 2.6-6.5 years of aging.
However, there was no difference in the other three outcomes – hippocampal volume, reaction time, and visual processing – between the two study groups.
Since investigators did not have information on the length of time participants napped, Ms. Paz suggested that “taking a short nap in the early afternoon may help cognition in those needing it.”
However, she added, the study’s findings need to be replicated before any firm conclusions can be made.
“More work is needed to examine the associations between napping and cognition, and the replication of these findings using other datasets and methods,” she said.
The investigators note that the study’s findings augment the knowledge of the “impact of habitual daytime napping on brain health, which is essential to understanding cognitive impairment in the aging population. The lack of evidence for an association between napping and hippocampal volume and cognitive outcomes (for example, alertness) may be affected by habitual daytime napping and should be studied in the future.”
Strengths, limitations
Tara Spires-Jones, PhD, president of the British Neuroscience Association and group leader at the UK Dementia Research Institute, said, “the study shows a small but significant increase in brain volume in people who have a genetic signature associated with taking daytime naps.”
Dr. Spires-Jones, who was not involved in the research, noted that while the study is well-conducted, it has limitations. Because Mendelian randomization uses a genetic signature, she noted, outcomes depend on the accuracy of the signature.
“The napping habits of UK Biobank participants were self-reported, which might not be entirely accurate, and the ‘napping’ signature overlapped substantially with the signature for cognitive outcomes in the study, which makes the causal link weaker,” she said.
“Even with those limitations, this study is interesting because it adds to the data indicating that sleep is important for brain health,” said Dr. Spires-Jones.
The study was supported by Diabetes UK, the British Heart Foundation, and the Diabetes Research and Wellness Foundation. In Uruguay, it was supported by Programa de Desarrollo de las Ciencias Básicas, Agencia Nacional de Investigación e Innovación, Comisión Sectorial de Investigación Científica, and Comisión Académica de Posgrado. In the United States it was supported by the National Heart, Lung, and Blood Institute. There were no disclosures reported.
A version of this article first appeared on Medscape.com.
Investigators at University College London, and the University of the Republic of Uruguay, Montevideo, found individuals genetically predisposed to regular napping had larger total brain volume, a surrogate of better cognitive health.
“Our results suggest that napping may improve brain health,” first author Valentina Paz, MSc, a PhD candidate at the University of the Republic of Uruguay said in an interview. “Specifically, our work revealed a 15.8 cubic cm increase in total brain volume with more frequent daytime napping,” she said.
The findings were published online in Sleep Health.
Higher brain volume
Previous studies examining the potential link between napping and cognition in older adults have yielded conflicting results.
To clarify this association, Ms. Paz and colleagues used Mendelian randomization to study DNA samples, cognitive outcomes, and functional magnetic resonance imaging data in participants from the ongoing UK Biobank Study.
Starting with data from 378,932 study participants (mean age 57), investigators compared measures of brain health and cognition of those who are more genetically programmed to nap with people who did not have these genetic variations.
More specifically, the investigators examined 97 sections of genetic code previously linked to the likelihood of regular napping and correlated these results with fMRI and cognitive outcomes between those genetically predisposed to take regular naps and those who weren’t.
Study outcomes included total brain volume, hippocampal volume, reaction time, and visual memory.
The final study sample included 35,080 with neuroimaging, cognitive assessment, and genotype data.
The researchers estimated that the average difference in brain volume between individuals genetically programmed to be habitual nappers and those who were not was equivalent to 15.8 cubic cm, or 2.6-6.5 years of aging.
However, there was no difference in the other three outcomes – hippocampal volume, reaction time, and visual processing – between the two study groups.
Since investigators did not have information on the length of time participants napped, Ms. Paz suggested that “taking a short nap in the early afternoon may help cognition in those needing it.”
However, she added, the study’s findings need to be replicated before any firm conclusions can be made.
“More work is needed to examine the associations between napping and cognition, and the replication of these findings using other datasets and methods,” she said.
The investigators note that the study’s findings augment the knowledge of the “impact of habitual daytime napping on brain health, which is essential to understanding cognitive impairment in the aging population. The lack of evidence for an association between napping and hippocampal volume and cognitive outcomes (for example, alertness) may be affected by habitual daytime napping and should be studied in the future.”
Strengths, limitations
Tara Spires-Jones, PhD, president of the British Neuroscience Association and group leader at the UK Dementia Research Institute, said, “the study shows a small but significant increase in brain volume in people who have a genetic signature associated with taking daytime naps.”
Dr. Spires-Jones, who was not involved in the research, noted that while the study is well-conducted, it has limitations. Because Mendelian randomization uses a genetic signature, she noted, outcomes depend on the accuracy of the signature.
“The napping habits of UK Biobank participants were self-reported, which might not be entirely accurate, and the ‘napping’ signature overlapped substantially with the signature for cognitive outcomes in the study, which makes the causal link weaker,” she said.
“Even with those limitations, this study is interesting because it adds to the data indicating that sleep is important for brain health,” said Dr. Spires-Jones.
The study was supported by Diabetes UK, the British Heart Foundation, and the Diabetes Research and Wellness Foundation. In Uruguay, it was supported by Programa de Desarrollo de las Ciencias Básicas, Agencia Nacional de Investigación e Innovación, Comisión Sectorial de Investigación Científica, and Comisión Académica de Posgrado. In the United States it was supported by the National Heart, Lung, and Blood Institute. There were no disclosures reported.
A version of this article first appeared on Medscape.com.
FROM SLEEP HEALTH
Should you dismiss a difficult patient?
Some patients continually cancel their appointments, ignore your medical directions, treat your staff rudely, or send you harassing emails.
Do you have to tolerate their behavior?
No, these are all appropriate reasons to terminate patients, attorneys say. Patients also can be dismissed for misleading doctors about their past medical history, chronic drug-seeking, displaying threatening or seductive behavior toward staff members or physicians, or any criminal behavior in the office, experts say.
But even if a reason seems legitimate, that doesn’t make it legal. Doctors should consider whether the reason is legal, said Chicago-area attorney Ericka Adler, JD, a partner at Roetzel & Andress, who advises doctors about terminating patients.
Ms. Adler said.
Terminating patients for an “illegal” reason such as discrimination based on race or gender or sexual orientation – even if couched as a legitimate patient issue – could open the practice to a lawsuit, Ms. Adler said.
Doctors also want to avoid patient abandonment claims by talking to the patient about problems and documenting them as they arise. If they can’t be resolved, doctors should ensure that there’s continuity of care when patients change physicians, said Ms. Adler.
About 90% of physicians have dismissed at least one patient during their career, according to a study of nearly 800 primary care practices. The most common reasons were legitimate: a patient was “extremely disruptive and/or behaved inappropriately toward clinicians or staff”; a patient had “violated chronic pain and controlled substance policies”; and a patient had “repeatedly missed appointments.”
Jacqui O’Kane, DO, a family physician at South Georgia Medical Center in rural Nashville, said she has dismissed about 15 of 3,000 patients she has seen in the past 3 years at the clinic. Before she dismisses a patient, she looks at whether there has been a pattern of behavior and tries to talk to them about the problem first to find out if there are other reasons for it.
She also gives patients a warning: If the unacceptable behavior continues, it will lead to their dismissal.
When patients cross a line
Dr. O’Kane warned an elderly man who used the N-word with her that she wouldn’t tolerate that language in her office. Then, when he later called her front office employee the N-word, she decided to dismiss him.
“I said, ‘That’s it, you can’t say that to someone in this office. I already told you once, and you did it again. I’m sorry, you have to find another doctor,’ ” said Dr. O’Kane.
Another patient crossed a line when she missed four appointments, refused to come in, and kept sending Dr. O’Kane long messages on MyChart demanding medications and advice. One message was fairly obtrusive: “If you don’t give me something stronger for my nerves TODAY, I am going to LOSE MY MIND!!!” Dr. O’Kane said the patient wrote.
“I then told her that’s not how I run my practice and that she needed to find someone else.”
Another common reason doctors dismiss patients is for nonpayment, says Ms. Adler.
Recently, however, some patients have also begun demanding their money back from doctors for services already received and billed because they were unhappy about something that occurred at the doctor’s office, said Ms. Adler.
“I advise doctors to respond: ‘We disagree that you didn’t get the service, but we will give you your money back, and we’re also terminating you from our practice.’ At that point, the doctor-patient relationship has become impossible,” said Ms. Adler.
How to dismiss difficult patients ethically and legally
According to the AMA’s Council on Ethical and Judicial Affairs, a physician may not discontinue treatment of a patient if further treatment is medically indicated without giving the patient reasonable notice and sufficient opportunity to make alternative arrangements for care.
Terminating a patient abruptly without transferring their care could lead to a claim of patient abandonment and the physician being called before a licensing board for potentially violating the state’s Medical Practice Act, said Ms. Adler.
Doctors can take these six steps to set the stage for dismissal and avoid a claim of patient abandonment.
1. Create written policies. Medical practices can describe the rules and behavior they expect from patients in these policies, which can cover, for example, payment, treating staff with courtesy, and medications. “When the rules are in writing and patients sign off on them, that gives doctors a certain comfort level in being able to refer to them and say that the patient hasn’t been compliant,” said Ms. Adler.
She also recommends that your practice create a policy that doctors should let the patient know about their concerns and meet with them to discuss the problem before receiving a termination letter.
2. Document any consistent problems you’re having with a patient. When you start having problems with a patient, you should document when the problem occurred, how often it occurred, any discussions with the patient about the problem, warnings you gave the patient, and if and when you decided to terminate the patient.
3. Meet with the patient to discuss the problem. “Talking and meeting with a patient also allows the physician to assess whether there’s another issue. For example, is there a mental health concern? Is there a financial reason for nonpayment or no-shows? There are multiple benefits to finding out what the problem is,” said Ms. Adler.
Once you’ve decided to terminate a patient, here’s what you should do:
4. Allow enough time for the patient to find alternative care. Ms. Adler recommends giving patients 30 days’ notice and that physicians offer to provide emergency care during that time. However, if the patient is undergoing treatment or has other challenges, more time may be needed to transfer care.
“It’s important to consider the patient’s context – if the patient is receiving cancer treatment, or is in a late stage of pregnancy, or lives in a rural area where few specialists are available, you may want to treat them longer – at least until they finish their treatment,” said Ms. Adler. Also, states may have their own requirements about minimum notice periods, she said.
5. Provide patients with written notice that you intend to terminate their care. Ms. Adler recommends that each letter be tailored to the patient’s specific circumstances. “You could spell out a patient’s history of noncompliance or nonpayment or inappropriate conduct because it’s been documented and the patient is already aware of it from a previous discussion,” she said.
Ms. Adler also recommends that doctors consult with legal counsel when in doubt or if contacted by the patient’s lawyer. Some lawyers will draft the termination letters, she said.
6. Include the following information in the written letter: The date that they will no longer receive care, how they can obtain copies of their medical records, and how they can find a new physician by providing contact information for a state medical association or similar organization, which often maintains a database of clinicians by specialty and location.
The letter should also state that the doctor will provide emergency care during the 30 days. Ms. Adler also recommends sending the notice by certified mail.
Dr. O’Kane said she may be more likely to give patients a second chance because she practices in a rural underserved area, and she understands that her patients don’t have many other options for health care. She also has developed a reputation for being willing to take on difficult patients that other physicians didn’t want to deal with, she said.
She encourages physicians to talk to patients to find out why, for example, they may not be compliant with medications.
“The patient may say, ‘I had to choose between paying for medications and putting food on the table,’ ” said Dr. O’Kane.
A version of this article first appeared on Medscape.com.
Some patients continually cancel their appointments, ignore your medical directions, treat your staff rudely, or send you harassing emails.
Do you have to tolerate their behavior?
No, these are all appropriate reasons to terminate patients, attorneys say. Patients also can be dismissed for misleading doctors about their past medical history, chronic drug-seeking, displaying threatening or seductive behavior toward staff members or physicians, or any criminal behavior in the office, experts say.
But even if a reason seems legitimate, that doesn’t make it legal. Doctors should consider whether the reason is legal, said Chicago-area attorney Ericka Adler, JD, a partner at Roetzel & Andress, who advises doctors about terminating patients.
Ms. Adler said.
Terminating patients for an “illegal” reason such as discrimination based on race or gender or sexual orientation – even if couched as a legitimate patient issue – could open the practice to a lawsuit, Ms. Adler said.
Doctors also want to avoid patient abandonment claims by talking to the patient about problems and documenting them as they arise. If they can’t be resolved, doctors should ensure that there’s continuity of care when patients change physicians, said Ms. Adler.
About 90% of physicians have dismissed at least one patient during their career, according to a study of nearly 800 primary care practices. The most common reasons were legitimate: a patient was “extremely disruptive and/or behaved inappropriately toward clinicians or staff”; a patient had “violated chronic pain and controlled substance policies”; and a patient had “repeatedly missed appointments.”
Jacqui O’Kane, DO, a family physician at South Georgia Medical Center in rural Nashville, said she has dismissed about 15 of 3,000 patients she has seen in the past 3 years at the clinic. Before she dismisses a patient, she looks at whether there has been a pattern of behavior and tries to talk to them about the problem first to find out if there are other reasons for it.
She also gives patients a warning: If the unacceptable behavior continues, it will lead to their dismissal.
When patients cross a line
Dr. O’Kane warned an elderly man who used the N-word with her that she wouldn’t tolerate that language in her office. Then, when he later called her front office employee the N-word, she decided to dismiss him.
“I said, ‘That’s it, you can’t say that to someone in this office. I already told you once, and you did it again. I’m sorry, you have to find another doctor,’ ” said Dr. O’Kane.
Another patient crossed a line when she missed four appointments, refused to come in, and kept sending Dr. O’Kane long messages on MyChart demanding medications and advice. One message was fairly obtrusive: “If you don’t give me something stronger for my nerves TODAY, I am going to LOSE MY MIND!!!” Dr. O’Kane said the patient wrote.
“I then told her that’s not how I run my practice and that she needed to find someone else.”
Another common reason doctors dismiss patients is for nonpayment, says Ms. Adler.
Recently, however, some patients have also begun demanding their money back from doctors for services already received and billed because they were unhappy about something that occurred at the doctor’s office, said Ms. Adler.
“I advise doctors to respond: ‘We disagree that you didn’t get the service, but we will give you your money back, and we’re also terminating you from our practice.’ At that point, the doctor-patient relationship has become impossible,” said Ms. Adler.
How to dismiss difficult patients ethically and legally
According to the AMA’s Council on Ethical and Judicial Affairs, a physician may not discontinue treatment of a patient if further treatment is medically indicated without giving the patient reasonable notice and sufficient opportunity to make alternative arrangements for care.
Terminating a patient abruptly without transferring their care could lead to a claim of patient abandonment and the physician being called before a licensing board for potentially violating the state’s Medical Practice Act, said Ms. Adler.
Doctors can take these six steps to set the stage for dismissal and avoid a claim of patient abandonment.
1. Create written policies. Medical practices can describe the rules and behavior they expect from patients in these policies, which can cover, for example, payment, treating staff with courtesy, and medications. “When the rules are in writing and patients sign off on them, that gives doctors a certain comfort level in being able to refer to them and say that the patient hasn’t been compliant,” said Ms. Adler.
She also recommends that your practice create a policy that doctors should let the patient know about their concerns and meet with them to discuss the problem before receiving a termination letter.
2. Document any consistent problems you’re having with a patient. When you start having problems with a patient, you should document when the problem occurred, how often it occurred, any discussions with the patient about the problem, warnings you gave the patient, and if and when you decided to terminate the patient.
3. Meet with the patient to discuss the problem. “Talking and meeting with a patient also allows the physician to assess whether there’s another issue. For example, is there a mental health concern? Is there a financial reason for nonpayment or no-shows? There are multiple benefits to finding out what the problem is,” said Ms. Adler.
Once you’ve decided to terminate a patient, here’s what you should do:
4. Allow enough time for the patient to find alternative care. Ms. Adler recommends giving patients 30 days’ notice and that physicians offer to provide emergency care during that time. However, if the patient is undergoing treatment or has other challenges, more time may be needed to transfer care.
“It’s important to consider the patient’s context – if the patient is receiving cancer treatment, or is in a late stage of pregnancy, or lives in a rural area where few specialists are available, you may want to treat them longer – at least until they finish their treatment,” said Ms. Adler. Also, states may have their own requirements about minimum notice periods, she said.
5. Provide patients with written notice that you intend to terminate their care. Ms. Adler recommends that each letter be tailored to the patient’s specific circumstances. “You could spell out a patient’s history of noncompliance or nonpayment or inappropriate conduct because it’s been documented and the patient is already aware of it from a previous discussion,” she said.
Ms. Adler also recommends that doctors consult with legal counsel when in doubt or if contacted by the patient’s lawyer. Some lawyers will draft the termination letters, she said.
6. Include the following information in the written letter: The date that they will no longer receive care, how they can obtain copies of their medical records, and how they can find a new physician by providing contact information for a state medical association or similar organization, which often maintains a database of clinicians by specialty and location.
The letter should also state that the doctor will provide emergency care during the 30 days. Ms. Adler also recommends sending the notice by certified mail.
Dr. O’Kane said she may be more likely to give patients a second chance because she practices in a rural underserved area, and she understands that her patients don’t have many other options for health care. She also has developed a reputation for being willing to take on difficult patients that other physicians didn’t want to deal with, she said.
She encourages physicians to talk to patients to find out why, for example, they may not be compliant with medications.
“The patient may say, ‘I had to choose between paying for medications and putting food on the table,’ ” said Dr. O’Kane.
A version of this article first appeared on Medscape.com.
Some patients continually cancel their appointments, ignore your medical directions, treat your staff rudely, or send you harassing emails.
Do you have to tolerate their behavior?
No, these are all appropriate reasons to terminate patients, attorneys say. Patients also can be dismissed for misleading doctors about their past medical history, chronic drug-seeking, displaying threatening or seductive behavior toward staff members or physicians, or any criminal behavior in the office, experts say.
But even if a reason seems legitimate, that doesn’t make it legal. Doctors should consider whether the reason is legal, said Chicago-area attorney Ericka Adler, JD, a partner at Roetzel & Andress, who advises doctors about terminating patients.
Ms. Adler said.
Terminating patients for an “illegal” reason such as discrimination based on race or gender or sexual orientation – even if couched as a legitimate patient issue – could open the practice to a lawsuit, Ms. Adler said.
Doctors also want to avoid patient abandonment claims by talking to the patient about problems and documenting them as they arise. If they can’t be resolved, doctors should ensure that there’s continuity of care when patients change physicians, said Ms. Adler.
About 90% of physicians have dismissed at least one patient during their career, according to a study of nearly 800 primary care practices. The most common reasons were legitimate: a patient was “extremely disruptive and/or behaved inappropriately toward clinicians or staff”; a patient had “violated chronic pain and controlled substance policies”; and a patient had “repeatedly missed appointments.”
Jacqui O’Kane, DO, a family physician at South Georgia Medical Center in rural Nashville, said she has dismissed about 15 of 3,000 patients she has seen in the past 3 years at the clinic. Before she dismisses a patient, she looks at whether there has been a pattern of behavior and tries to talk to them about the problem first to find out if there are other reasons for it.
She also gives patients a warning: If the unacceptable behavior continues, it will lead to their dismissal.
When patients cross a line
Dr. O’Kane warned an elderly man who used the N-word with her that she wouldn’t tolerate that language in her office. Then, when he later called her front office employee the N-word, she decided to dismiss him.
“I said, ‘That’s it, you can’t say that to someone in this office. I already told you once, and you did it again. I’m sorry, you have to find another doctor,’ ” said Dr. O’Kane.
Another patient crossed a line when she missed four appointments, refused to come in, and kept sending Dr. O’Kane long messages on MyChart demanding medications and advice. One message was fairly obtrusive: “If you don’t give me something stronger for my nerves TODAY, I am going to LOSE MY MIND!!!” Dr. O’Kane said the patient wrote.
“I then told her that’s not how I run my practice and that she needed to find someone else.”
Another common reason doctors dismiss patients is for nonpayment, says Ms. Adler.
Recently, however, some patients have also begun demanding their money back from doctors for services already received and billed because they were unhappy about something that occurred at the doctor’s office, said Ms. Adler.
“I advise doctors to respond: ‘We disagree that you didn’t get the service, but we will give you your money back, and we’re also terminating you from our practice.’ At that point, the doctor-patient relationship has become impossible,” said Ms. Adler.
How to dismiss difficult patients ethically and legally
According to the AMA’s Council on Ethical and Judicial Affairs, a physician may not discontinue treatment of a patient if further treatment is medically indicated without giving the patient reasonable notice and sufficient opportunity to make alternative arrangements for care.
Terminating a patient abruptly without transferring their care could lead to a claim of patient abandonment and the physician being called before a licensing board for potentially violating the state’s Medical Practice Act, said Ms. Adler.
Doctors can take these six steps to set the stage for dismissal and avoid a claim of patient abandonment.
1. Create written policies. Medical practices can describe the rules and behavior they expect from patients in these policies, which can cover, for example, payment, treating staff with courtesy, and medications. “When the rules are in writing and patients sign off on them, that gives doctors a certain comfort level in being able to refer to them and say that the patient hasn’t been compliant,” said Ms. Adler.
She also recommends that your practice create a policy that doctors should let the patient know about their concerns and meet with them to discuss the problem before receiving a termination letter.
2. Document any consistent problems you’re having with a patient. When you start having problems with a patient, you should document when the problem occurred, how often it occurred, any discussions with the patient about the problem, warnings you gave the patient, and if and when you decided to terminate the patient.
3. Meet with the patient to discuss the problem. “Talking and meeting with a patient also allows the physician to assess whether there’s another issue. For example, is there a mental health concern? Is there a financial reason for nonpayment or no-shows? There are multiple benefits to finding out what the problem is,” said Ms. Adler.
Once you’ve decided to terminate a patient, here’s what you should do:
4. Allow enough time for the patient to find alternative care. Ms. Adler recommends giving patients 30 days’ notice and that physicians offer to provide emergency care during that time. However, if the patient is undergoing treatment or has other challenges, more time may be needed to transfer care.
“It’s important to consider the patient’s context – if the patient is receiving cancer treatment, or is in a late stage of pregnancy, or lives in a rural area where few specialists are available, you may want to treat them longer – at least until they finish their treatment,” said Ms. Adler. Also, states may have their own requirements about minimum notice periods, she said.
5. Provide patients with written notice that you intend to terminate their care. Ms. Adler recommends that each letter be tailored to the patient’s specific circumstances. “You could spell out a patient’s history of noncompliance or nonpayment or inappropriate conduct because it’s been documented and the patient is already aware of it from a previous discussion,” she said.
Ms. Adler also recommends that doctors consult with legal counsel when in doubt or if contacted by the patient’s lawyer. Some lawyers will draft the termination letters, she said.
6. Include the following information in the written letter: The date that they will no longer receive care, how they can obtain copies of their medical records, and how they can find a new physician by providing contact information for a state medical association or similar organization, which often maintains a database of clinicians by specialty and location.
The letter should also state that the doctor will provide emergency care during the 30 days. Ms. Adler also recommends sending the notice by certified mail.
Dr. O’Kane said she may be more likely to give patients a second chance because she practices in a rural underserved area, and she understands that her patients don’t have many other options for health care. She also has developed a reputation for being willing to take on difficult patients that other physicians didn’t want to deal with, she said.
She encourages physicians to talk to patients to find out why, for example, they may not be compliant with medications.
“The patient may say, ‘I had to choose between paying for medications and putting food on the table,’ ” said Dr. O’Kane.
A version of this article first appeared on Medscape.com.
Low-calorie tastes sweeter with a little salt
Low-calorie tastes sweeter with a little salt
Diet and sugar-free foods and drinks seem like a good idea, but it’s hard to get past that strange aftertaste, right? It’s the calling card for the noncaloric aspartame- and stevia-containing sweeteners that we consume to make us feel like we can have the best of both worlds.
That weird lingering taste can be a total turn-off for some (raises hand), but researchers have found an almost facepalm solution to the not-so-sweet problem, and it’s salt.
Now, the concept of sweet and salty is not a far-fetched partnership when it comes to snack consumption (try M&Ms in your popcorn). The researchers at Almendra, a manufacturer of stevia sweeteners, put that iconic flavor pair to the test by adding mineral salts that have some nutritional value to lessen the effect of a stevia compound, rebaudioside A, found in noncaloric sweeteners.
The researchers added in magnesium chloride, calcium chloride, and potassium chloride separately to lessen rebaudioside A’s intensity, but they needed so much salt that it killed the sweet taste completely. A blend of the three mineral salts, however, reduced the lingering taste by 79% and improved the real sugar-like taste. The researchers tried this blend in reduced-calorie orange juice and a citrus-flavored soft drink, improving the taste in both.
The salty and sweet match comes in for the win once again. This time helping against the fight of obesity instead of making it worse.
Pseudomonas’ Achilles’ heel is more of an Achilles’ genetic switch
Today, on the long-awaited return of “Bacteria vs. the World,” we meet one of the rock stars of infectious disease.
LOTME: Through the use of imaginary technology, we’re talking to Pseudomonas aeruginosa. Thanks for joining us on such short notice, after Neisseria gonorrhoeae canceled at the last minute.
P. aeruginosa: No problem. I think we can all guess what that little devil is up to.
LOTME: Bacterial resistance to antibiotics is a huge problem for our species. What makes you so hard to fight?
P. aeruginosa: We’ve been trying to keep that a secret, actually, but now that researchers in Switzerland and Denmark seem to have figured it out, I guess it’s okay for me to spill the beans.
LOTME: Beans? What do beans have to do with it?
P. aeruginosa: Nothing, it’s just a colloquial expression that means I’m sharing previously private information.
LOTME: Sure, we knew that. Please, continue your spilling.
P. aeruginosa: The secret is … Well, let’s just say we were a little worried when the Clash released “Should I Stay or Should I Go” back in the 1980s.
LOTME: The Clash? Now we’re really confused.
P. aeruginosa: The answer to their question, “Should I stay or should I go? is yes. Successful invasion of a human is all about division of labor. “While one fraction of the bacterial population adheres to the mucosal surface and forms a biofilm, the other subpopulation spreads to distant tissue sites,” is how the investigators described it. We can increase surface colonization by using a “job-sharing” process, they said, and even resist antibiotics because most of us remain in the protective biofilm.
LOTME: And they say you guys don’t have brains.
P. aeruginosa: But wait, there’s more. We don’t just divide the labor randomly. After the initial colonization we form two functionally distinct subpopulations. One has high levels of the bacterial signaling molecule c-di-GMP and stays put to work on the biofilm. The other group, with low levels of c-di-GMP, heads out to the surrounding tissue to continue the colonization. As project leader Urs Jenal put it, “By identifying the genetic switch, we have tracked down the Achilles heel of the pathogen.”
LOTME: Pretty clever stuff, for humans, anyway.
P. aeruginosa: We agree, but now that you know our secret, we can’t let you share it.
LOTME: Wait! The journal article’s already been published. Your secret is out. You can’t stop that by infecting me.
P. aeruginosa: True enough, but are you familiar with the fable of the scorpion and the frog? It’s our nature.
LOTME: Nooooo! N. gonorrhoeae wouldn’t have done this!
What a pain in the Butt
Businesses rise and businesses fall. We all know that one cursed location, that spot in town where we see businesses move in and close up in a matter of months. At the same time, though, there are also businesses that have been around as long as anyone can remember, pillars of the community.
Corydon, IN., likely has a few such long-lived shops, but it is officially down one 70-year-old family business as of late April, with the unfortunate passing of beloved local pharmacy Butt Drugs. Prescription pick-up in rear.
The business dates back to 1952, when it was founded as William H. Butt Drugs. We’re sure William Butt was never teased about his last name. Nope. No one would ever do that. After he passed the store to his children, it underwent a stint as Butt Rexall Drugs. When the shop was passed down to its third-generation and ultimately final owner, Katie Butt Beckort, she decided to simplify the name. Get right down to the bottom of things, as it were.
Butt Drugs was a popular spot, featuring an old-school soda fountain and themed souvenirs. According to Ms. Butt Beckort, people would come from miles away to buy “I love Butt Drugs” T-shirts, magnets, and so on. Yes, they knew perfectly well what they were sitting on.
So, if was such a hit, why did it close? Butt Drugs may have a hilarious name and merchandise to match, but the pharmacy portion of the pharmacy had been losing money for years. You know, the actual point of the business. As with so many things, we can blame it on the insurance companies. More than half the drugs that passed through Butt Drugs’ doors were sold at a loss, because the insurance companies refused to reimburse the store more than the wholesale price of the drug. Not even a good butt drug could clear up that financial diarrhea.
And so, we’ve lost Butt Drugs forever. Spicy food enthusiasts, coffee drinkers, and all patrons of Taco Bell, take a moment to reflect and mourn on what you’ve lost. No more Butt Drugs to relieve your suffering. A true kick in the butt indeed.
Low-calorie tastes sweeter with a little salt
Diet and sugar-free foods and drinks seem like a good idea, but it’s hard to get past that strange aftertaste, right? It’s the calling card for the noncaloric aspartame- and stevia-containing sweeteners that we consume to make us feel like we can have the best of both worlds.
That weird lingering taste can be a total turn-off for some (raises hand), but researchers have found an almost facepalm solution to the not-so-sweet problem, and it’s salt.
Now, the concept of sweet and salty is not a far-fetched partnership when it comes to snack consumption (try M&Ms in your popcorn). The researchers at Almendra, a manufacturer of stevia sweeteners, put that iconic flavor pair to the test by adding mineral salts that have some nutritional value to lessen the effect of a stevia compound, rebaudioside A, found in noncaloric sweeteners.
The researchers added in magnesium chloride, calcium chloride, and potassium chloride separately to lessen rebaudioside A’s intensity, but they needed so much salt that it killed the sweet taste completely. A blend of the three mineral salts, however, reduced the lingering taste by 79% and improved the real sugar-like taste. The researchers tried this blend in reduced-calorie orange juice and a citrus-flavored soft drink, improving the taste in both.
The salty and sweet match comes in for the win once again. This time helping against the fight of obesity instead of making it worse.
Pseudomonas’ Achilles’ heel is more of an Achilles’ genetic switch
Today, on the long-awaited return of “Bacteria vs. the World,” we meet one of the rock stars of infectious disease.
LOTME: Through the use of imaginary technology, we’re talking to Pseudomonas aeruginosa. Thanks for joining us on such short notice, after Neisseria gonorrhoeae canceled at the last minute.
P. aeruginosa: No problem. I think we can all guess what that little devil is up to.
LOTME: Bacterial resistance to antibiotics is a huge problem for our species. What makes you so hard to fight?
P. aeruginosa: We’ve been trying to keep that a secret, actually, but now that researchers in Switzerland and Denmark seem to have figured it out, I guess it’s okay for me to spill the beans.
LOTME: Beans? What do beans have to do with it?
P. aeruginosa: Nothing, it’s just a colloquial expression that means I’m sharing previously private information.
LOTME: Sure, we knew that. Please, continue your spilling.
P. aeruginosa: The secret is … Well, let’s just say we were a little worried when the Clash released “Should I Stay or Should I Go” back in the 1980s.
LOTME: The Clash? Now we’re really confused.
P. aeruginosa: The answer to their question, “Should I stay or should I go? is yes. Successful invasion of a human is all about division of labor. “While one fraction of the bacterial population adheres to the mucosal surface and forms a biofilm, the other subpopulation spreads to distant tissue sites,” is how the investigators described it. We can increase surface colonization by using a “job-sharing” process, they said, and even resist antibiotics because most of us remain in the protective biofilm.
LOTME: And they say you guys don’t have brains.
P. aeruginosa: But wait, there’s more. We don’t just divide the labor randomly. After the initial colonization we form two functionally distinct subpopulations. One has high levels of the bacterial signaling molecule c-di-GMP and stays put to work on the biofilm. The other group, with low levels of c-di-GMP, heads out to the surrounding tissue to continue the colonization. As project leader Urs Jenal put it, “By identifying the genetic switch, we have tracked down the Achilles heel of the pathogen.”
LOTME: Pretty clever stuff, for humans, anyway.
P. aeruginosa: We agree, but now that you know our secret, we can’t let you share it.
LOTME: Wait! The journal article’s already been published. Your secret is out. You can’t stop that by infecting me.
P. aeruginosa: True enough, but are you familiar with the fable of the scorpion and the frog? It’s our nature.
LOTME: Nooooo! N. gonorrhoeae wouldn’t have done this!
What a pain in the Butt
Businesses rise and businesses fall. We all know that one cursed location, that spot in town where we see businesses move in and close up in a matter of months. At the same time, though, there are also businesses that have been around as long as anyone can remember, pillars of the community.
Corydon, IN., likely has a few such long-lived shops, but it is officially down one 70-year-old family business as of late April, with the unfortunate passing of beloved local pharmacy Butt Drugs. Prescription pick-up in rear.
The business dates back to 1952, when it was founded as William H. Butt Drugs. We’re sure William Butt was never teased about his last name. Nope. No one would ever do that. After he passed the store to his children, it underwent a stint as Butt Rexall Drugs. When the shop was passed down to its third-generation and ultimately final owner, Katie Butt Beckort, she decided to simplify the name. Get right down to the bottom of things, as it were.
Butt Drugs was a popular spot, featuring an old-school soda fountain and themed souvenirs. According to Ms. Butt Beckort, people would come from miles away to buy “I love Butt Drugs” T-shirts, magnets, and so on. Yes, they knew perfectly well what they were sitting on.
So, if was such a hit, why did it close? Butt Drugs may have a hilarious name and merchandise to match, but the pharmacy portion of the pharmacy had been losing money for years. You know, the actual point of the business. As with so many things, we can blame it on the insurance companies. More than half the drugs that passed through Butt Drugs’ doors were sold at a loss, because the insurance companies refused to reimburse the store more than the wholesale price of the drug. Not even a good butt drug could clear up that financial diarrhea.
And so, we’ve lost Butt Drugs forever. Spicy food enthusiasts, coffee drinkers, and all patrons of Taco Bell, take a moment to reflect and mourn on what you’ve lost. No more Butt Drugs to relieve your suffering. A true kick in the butt indeed.
Low-calorie tastes sweeter with a little salt
Diet and sugar-free foods and drinks seem like a good idea, but it’s hard to get past that strange aftertaste, right? It’s the calling card for the noncaloric aspartame- and stevia-containing sweeteners that we consume to make us feel like we can have the best of both worlds.
That weird lingering taste can be a total turn-off for some (raises hand), but researchers have found an almost facepalm solution to the not-so-sweet problem, and it’s salt.
Now, the concept of sweet and salty is not a far-fetched partnership when it comes to snack consumption (try M&Ms in your popcorn). The researchers at Almendra, a manufacturer of stevia sweeteners, put that iconic flavor pair to the test by adding mineral salts that have some nutritional value to lessen the effect of a stevia compound, rebaudioside A, found in noncaloric sweeteners.
The researchers added in magnesium chloride, calcium chloride, and potassium chloride separately to lessen rebaudioside A’s intensity, but they needed so much salt that it killed the sweet taste completely. A blend of the three mineral salts, however, reduced the lingering taste by 79% and improved the real sugar-like taste. The researchers tried this blend in reduced-calorie orange juice and a citrus-flavored soft drink, improving the taste in both.
The salty and sweet match comes in for the win once again. This time helping against the fight of obesity instead of making it worse.
Pseudomonas’ Achilles’ heel is more of an Achilles’ genetic switch
Today, on the long-awaited return of “Bacteria vs. the World,” we meet one of the rock stars of infectious disease.
LOTME: Through the use of imaginary technology, we’re talking to Pseudomonas aeruginosa. Thanks for joining us on such short notice, after Neisseria gonorrhoeae canceled at the last minute.
P. aeruginosa: No problem. I think we can all guess what that little devil is up to.
LOTME: Bacterial resistance to antibiotics is a huge problem for our species. What makes you so hard to fight?
P. aeruginosa: We’ve been trying to keep that a secret, actually, but now that researchers in Switzerland and Denmark seem to have figured it out, I guess it’s okay for me to spill the beans.
LOTME: Beans? What do beans have to do with it?
P. aeruginosa: Nothing, it’s just a colloquial expression that means I’m sharing previously private information.
LOTME: Sure, we knew that. Please, continue your spilling.
P. aeruginosa: The secret is … Well, let’s just say we were a little worried when the Clash released “Should I Stay or Should I Go” back in the 1980s.
LOTME: The Clash? Now we’re really confused.
P. aeruginosa: The answer to their question, “Should I stay or should I go? is yes. Successful invasion of a human is all about division of labor. “While one fraction of the bacterial population adheres to the mucosal surface and forms a biofilm, the other subpopulation spreads to distant tissue sites,” is how the investigators described it. We can increase surface colonization by using a “job-sharing” process, they said, and even resist antibiotics because most of us remain in the protective biofilm.
LOTME: And they say you guys don’t have brains.
P. aeruginosa: But wait, there’s more. We don’t just divide the labor randomly. After the initial colonization we form two functionally distinct subpopulations. One has high levels of the bacterial signaling molecule c-di-GMP and stays put to work on the biofilm. The other group, with low levels of c-di-GMP, heads out to the surrounding tissue to continue the colonization. As project leader Urs Jenal put it, “By identifying the genetic switch, we have tracked down the Achilles heel of the pathogen.”
LOTME: Pretty clever stuff, for humans, anyway.
P. aeruginosa: We agree, but now that you know our secret, we can’t let you share it.
LOTME: Wait! The journal article’s already been published. Your secret is out. You can’t stop that by infecting me.
P. aeruginosa: True enough, but are you familiar with the fable of the scorpion and the frog? It’s our nature.
LOTME: Nooooo! N. gonorrhoeae wouldn’t have done this!
What a pain in the Butt
Businesses rise and businesses fall. We all know that one cursed location, that spot in town where we see businesses move in and close up in a matter of months. At the same time, though, there are also businesses that have been around as long as anyone can remember, pillars of the community.
Corydon, IN., likely has a few such long-lived shops, but it is officially down one 70-year-old family business as of late April, with the unfortunate passing of beloved local pharmacy Butt Drugs. Prescription pick-up in rear.
The business dates back to 1952, when it was founded as William H. Butt Drugs. We’re sure William Butt was never teased about his last name. Nope. No one would ever do that. After he passed the store to his children, it underwent a stint as Butt Rexall Drugs. When the shop was passed down to its third-generation and ultimately final owner, Katie Butt Beckort, she decided to simplify the name. Get right down to the bottom of things, as it were.
Butt Drugs was a popular spot, featuring an old-school soda fountain and themed souvenirs. According to Ms. Butt Beckort, people would come from miles away to buy “I love Butt Drugs” T-shirts, magnets, and so on. Yes, they knew perfectly well what they were sitting on.
So, if was such a hit, why did it close? Butt Drugs may have a hilarious name and merchandise to match, but the pharmacy portion of the pharmacy had been losing money for years. You know, the actual point of the business. As with so many things, we can blame it on the insurance companies. More than half the drugs that passed through Butt Drugs’ doors were sold at a loss, because the insurance companies refused to reimburse the store more than the wholesale price of the drug. Not even a good butt drug could clear up that financial diarrhea.
And so, we’ve lost Butt Drugs forever. Spicy food enthusiasts, coffee drinkers, and all patrons of Taco Bell, take a moment to reflect and mourn on what you’ve lost. No more Butt Drugs to relieve your suffering. A true kick in the butt indeed.
Proposal to cap Part B pay on some drugs draws opposition
An influential panel proposed capping Medicare Part B pay for some drugs, arguing this would remove financial incentives to use more costly medicines when there are less expensive equivalents.
Medical groups have objected to both this recommendation from the Medicare Payment Advisory Commission (MedPAC) and the panel’s underlying premise. MedPAC said financial as well as clinical factors can come into play in clinicians’ choices of drugs for patients.
In an interview, Christina Downey, MD, chair of the Government Affairs Committee of the American College of Rheumatology, said physicians in her field cannot switch patients’ medicines to try to make a profit.
“Patients only respond to the drugs that they respond to,” Dr. Downey said. “It’s frankly very insulting to say that physicians just force patients to go on medicines that are going to make them a bunch of money.”
In a June report to Congress, MedPAC recommended reducing the add-on payment for many drugs given in hospitals and clinics, which are thus covered by Part B, as part of a package of suggestions for addressing rising costs. Part B drug spending grew about 9% annually between 2009 and 2021, rising from $15.4 billion to $42.9 billion, MedPAC said.
Medicare’s current Part B drug pricing model starts with the reported average sales price (ASP) and then adds about 4.3% or 6%, depending on current budget-sequester law, to the cost of medicines.
MedPAC members voted 17-0 in April in favor of a general recommendation to revise the Part B payment approach. In the June report, MedPAC fleshes out this idea. It mentions a model in which the add-on Part B payment would be the lesser of either 6% of the ASP, 3% plus $24, or $220.
The majority of Part B drug administrations are for very low-priced drugs, MedPAC said. But for some of the more costly ones, annual prices can be more than $400,000 per patient, and future launch prices may be even higher for certain types of products, such as gene therapies, MedPAC said.
“There is no evidence that the costs of a drug’s administration are proportionate to the price of the drug,” MedPAC said.
Concerns about how well Medicare covers the cost of drug administration should be addressed through other pathways, such as the American Medical
Association’s Specialty Society Relative Value Scale Update Committee (RUC), MedPAC said. AMA’s RUC advises the Centers for Medicare & Medicaid Services on the physician fee schedule.
Congress is not obliged to act on or to even consider MedPAC’s work. In general, lawmakers and CMS often pay heed to the panel’s recommendations, sometimes incorporating them into new policy.
But this new MedPAC Part B recommendation has drawn strong opposition, similar to the response to a 2016 CMS plan to cut the Part B add-on payment. That plan, which CMS later abandoned, would have cut the markup on Part B drugs to 2.5% and added a flat fee to cover administration costs.
Why not focus on PBMs instead?
The timing of the MedPAC recommendation is poor, given that CMS already is trying to implement the Inflation Reduction Act and create a new system of direct Medicare drug price negotiations, as ordered by Congress, said Madelaine A. Feldman, MD, a rheumatologist based in New Orleans.
A better approach for lowering drug prices would be to focus more on the operations of pharmacy benefit managers (PBMs), said Dr. Feldman, who also is vice president for advocacy and government affairs for the Coalition of State Rheumatology Organizations. A pending bipartisan Senate bill, for example, would prohibit PBM compensation based on the price of a drug as a condition of entering into a contract with a Medicare Part D plan.
Congress needs to take steps to unlink the profits of PBMs from higher drug prices, Dr. Feldman said.
“Until that happens, we can put all the lipstick we want on this big pig, but it’s not going to really fix the problem,” she said.
Reduced pay for drugs acquired through 340B program?
In an interview about the new MedPAC proposal, Ted Okon, executive director of the Community Oncology Alliance, urged renewed attention to what he sees as unintended consequences of the 340B discount drug program.
Under this program, certain hospitals can acquire drugs at steeply reduced prices, but they are not obliged to share those discounts with patients. Hospitals that participate in the 340B program can gain funds when patients and their insurers, including Medicare, pay more for the medicines hospitals and other organizations acquired with the 340B discount. Hospitals say they use the money from the 340B program to expand resources in their communities.
But rapid growth of the program in recent years has led to questions, especially about the role of contract pharmacies that manage the program. Congress created the 340B program in 1992 as a workaround to then new rules on Medicaid drug coverage.
In 2021, participating hospitals and clinics and organizations purchased about $44 billion worth of medicines through the 340B drug program. This was an increase of 16% from the previous year, according to a report from the nonprofit Commonwealth Fund. The number of sites, including hospitals and pharmacies, enrolled in the 340B program rose from 8,100 in 2000 to 50,000 by 2020, the report said.
MedPAC in 2016 urged CMS to reduce the amount Medicare pays for drugs acquired through the 340B program. CMS did so during the Trump administration, a policy later defended by the Biden administration.
But the U.S. Supreme Court last year said Medicare erred in its approach to making this cut, as earlier reported. Federal law required that the Department of Health and Human Services conduct a survey to support such a step, and HHS did not do this, the court said. CMS thus was ordered to return Medicare to the ASP+6% payment model for drugs purchased through the 340B discount program.
In the June report, though, MedPAC stuck by its 2016 recommendation that Medicare reduce its payments for drugs purchased through the 340B discount program despite this setback.
“We continue to believe that this approach is appropriate, and the specific level of payment reduction could be considered further as newer data become available,” MedPAC said.
Hospital, PhRMA split
Hospitals would certainly contest any renewed bid by CMS to drop Medicare’s pay for drugs purchased through the 340B program. The American Hospital Association objected to the MedPAC proposal regarding the add-on payment in Part B drug pricing.
MedPAC commissioners discussed this idea at a January meeting, prompting a February letter from the AHA to the panel. Like Dr. Feldman, AHA said it would be “premature” to launch into a revision of Part B drug pricing while the impact of the IRA on drug prices was still unclear.
AHA also noted that a reduction in Part B drug reimbursement would “shift the responsibility for the rapid increase in drug prices away from drug manufacturers, and instead places the burden on hospitals and patients.”
But the AHA gave a much warmer reception to another proposal MedPAC considered this year and that it included in its June report, which is a plan to address the high cost of certain drugs of as yet unconfirmed clinical benefit.
In April, the AHA said it supports a move toward a “value-based approach” in certain cases in which first-in-class medicines are sold under U.S. Food and Drug Administration’s accelerated approvals. Medicare could then cap payment for such drugs that have excessively high launch prices and uncertain clinical benefit, AHA said.
In the June report, MedPAC recommended that Medicare be able to place such a limit on Part B payments in certain cases, including ones in which companies do not meet FDA deadlines for postmarketing confirmatory trials.
The Pharmaceutical Research and Manufacturers of America (PhRMA) objected to this proposed change. The trade group for drugmakers said the FDA often revises and extends enrollment milestones for pending confirmatory trials when companies hit snags, such as challenges in enrolling patients, PhRMA said.
Reducing Part B payment for drugs for which confirmatory trials have been delayed would have a “disproportionate impact” on smaller and rural communities, where independent practices struggle to keep their doors open as it is, PhRMA spokeswoman Nicole Longo wrote in a blog post.
“If physicians can’t afford to administer a medicine, then they won’t and that means their patients won’t have access to them either,” Ms. Longo wrote.
A version of this article first appeared on Medscape.com.
An influential panel proposed capping Medicare Part B pay for some drugs, arguing this would remove financial incentives to use more costly medicines when there are less expensive equivalents.
Medical groups have objected to both this recommendation from the Medicare Payment Advisory Commission (MedPAC) and the panel’s underlying premise. MedPAC said financial as well as clinical factors can come into play in clinicians’ choices of drugs for patients.
In an interview, Christina Downey, MD, chair of the Government Affairs Committee of the American College of Rheumatology, said physicians in her field cannot switch patients’ medicines to try to make a profit.
“Patients only respond to the drugs that they respond to,” Dr. Downey said. “It’s frankly very insulting to say that physicians just force patients to go on medicines that are going to make them a bunch of money.”
In a June report to Congress, MedPAC recommended reducing the add-on payment for many drugs given in hospitals and clinics, which are thus covered by Part B, as part of a package of suggestions for addressing rising costs. Part B drug spending grew about 9% annually between 2009 and 2021, rising from $15.4 billion to $42.9 billion, MedPAC said.
Medicare’s current Part B drug pricing model starts with the reported average sales price (ASP) and then adds about 4.3% or 6%, depending on current budget-sequester law, to the cost of medicines.
MedPAC members voted 17-0 in April in favor of a general recommendation to revise the Part B payment approach. In the June report, MedPAC fleshes out this idea. It mentions a model in which the add-on Part B payment would be the lesser of either 6% of the ASP, 3% plus $24, or $220.
The majority of Part B drug administrations are for very low-priced drugs, MedPAC said. But for some of the more costly ones, annual prices can be more than $400,000 per patient, and future launch prices may be even higher for certain types of products, such as gene therapies, MedPAC said.
“There is no evidence that the costs of a drug’s administration are proportionate to the price of the drug,” MedPAC said.
Concerns about how well Medicare covers the cost of drug administration should be addressed through other pathways, such as the American Medical
Association’s Specialty Society Relative Value Scale Update Committee (RUC), MedPAC said. AMA’s RUC advises the Centers for Medicare & Medicaid Services on the physician fee schedule.
Congress is not obliged to act on or to even consider MedPAC’s work. In general, lawmakers and CMS often pay heed to the panel’s recommendations, sometimes incorporating them into new policy.
But this new MedPAC Part B recommendation has drawn strong opposition, similar to the response to a 2016 CMS plan to cut the Part B add-on payment. That plan, which CMS later abandoned, would have cut the markup on Part B drugs to 2.5% and added a flat fee to cover administration costs.
Why not focus on PBMs instead?
The timing of the MedPAC recommendation is poor, given that CMS already is trying to implement the Inflation Reduction Act and create a new system of direct Medicare drug price negotiations, as ordered by Congress, said Madelaine A. Feldman, MD, a rheumatologist based in New Orleans.
A better approach for lowering drug prices would be to focus more on the operations of pharmacy benefit managers (PBMs), said Dr. Feldman, who also is vice president for advocacy and government affairs for the Coalition of State Rheumatology Organizations. A pending bipartisan Senate bill, for example, would prohibit PBM compensation based on the price of a drug as a condition of entering into a contract with a Medicare Part D plan.
Congress needs to take steps to unlink the profits of PBMs from higher drug prices, Dr. Feldman said.
“Until that happens, we can put all the lipstick we want on this big pig, but it’s not going to really fix the problem,” she said.
Reduced pay for drugs acquired through 340B program?
In an interview about the new MedPAC proposal, Ted Okon, executive director of the Community Oncology Alliance, urged renewed attention to what he sees as unintended consequences of the 340B discount drug program.
Under this program, certain hospitals can acquire drugs at steeply reduced prices, but they are not obliged to share those discounts with patients. Hospitals that participate in the 340B program can gain funds when patients and their insurers, including Medicare, pay more for the medicines hospitals and other organizations acquired with the 340B discount. Hospitals say they use the money from the 340B program to expand resources in their communities.
But rapid growth of the program in recent years has led to questions, especially about the role of contract pharmacies that manage the program. Congress created the 340B program in 1992 as a workaround to then new rules on Medicaid drug coverage.
In 2021, participating hospitals and clinics and organizations purchased about $44 billion worth of medicines through the 340B drug program. This was an increase of 16% from the previous year, according to a report from the nonprofit Commonwealth Fund. The number of sites, including hospitals and pharmacies, enrolled in the 340B program rose from 8,100 in 2000 to 50,000 by 2020, the report said.
MedPAC in 2016 urged CMS to reduce the amount Medicare pays for drugs acquired through the 340B program. CMS did so during the Trump administration, a policy later defended by the Biden administration.
But the U.S. Supreme Court last year said Medicare erred in its approach to making this cut, as earlier reported. Federal law required that the Department of Health and Human Services conduct a survey to support such a step, and HHS did not do this, the court said. CMS thus was ordered to return Medicare to the ASP+6% payment model for drugs purchased through the 340B discount program.
In the June report, though, MedPAC stuck by its 2016 recommendation that Medicare reduce its payments for drugs purchased through the 340B discount program despite this setback.
“We continue to believe that this approach is appropriate, and the specific level of payment reduction could be considered further as newer data become available,” MedPAC said.
Hospital, PhRMA split
Hospitals would certainly contest any renewed bid by CMS to drop Medicare’s pay for drugs purchased through the 340B program. The American Hospital Association objected to the MedPAC proposal regarding the add-on payment in Part B drug pricing.
MedPAC commissioners discussed this idea at a January meeting, prompting a February letter from the AHA to the panel. Like Dr. Feldman, AHA said it would be “premature” to launch into a revision of Part B drug pricing while the impact of the IRA on drug prices was still unclear.
AHA also noted that a reduction in Part B drug reimbursement would “shift the responsibility for the rapid increase in drug prices away from drug manufacturers, and instead places the burden on hospitals and patients.”
But the AHA gave a much warmer reception to another proposal MedPAC considered this year and that it included in its June report, which is a plan to address the high cost of certain drugs of as yet unconfirmed clinical benefit.
In April, the AHA said it supports a move toward a “value-based approach” in certain cases in which first-in-class medicines are sold under U.S. Food and Drug Administration’s accelerated approvals. Medicare could then cap payment for such drugs that have excessively high launch prices and uncertain clinical benefit, AHA said.
In the June report, MedPAC recommended that Medicare be able to place such a limit on Part B payments in certain cases, including ones in which companies do not meet FDA deadlines for postmarketing confirmatory trials.
The Pharmaceutical Research and Manufacturers of America (PhRMA) objected to this proposed change. The trade group for drugmakers said the FDA often revises and extends enrollment milestones for pending confirmatory trials when companies hit snags, such as challenges in enrolling patients, PhRMA said.
Reducing Part B payment for drugs for which confirmatory trials have been delayed would have a “disproportionate impact” on smaller and rural communities, where independent practices struggle to keep their doors open as it is, PhRMA spokeswoman Nicole Longo wrote in a blog post.
“If physicians can’t afford to administer a medicine, then they won’t and that means their patients won’t have access to them either,” Ms. Longo wrote.
A version of this article first appeared on Medscape.com.
An influential panel proposed capping Medicare Part B pay for some drugs, arguing this would remove financial incentives to use more costly medicines when there are less expensive equivalents.
Medical groups have objected to both this recommendation from the Medicare Payment Advisory Commission (MedPAC) and the panel’s underlying premise. MedPAC said financial as well as clinical factors can come into play in clinicians’ choices of drugs for patients.
In an interview, Christina Downey, MD, chair of the Government Affairs Committee of the American College of Rheumatology, said physicians in her field cannot switch patients’ medicines to try to make a profit.
“Patients only respond to the drugs that they respond to,” Dr. Downey said. “It’s frankly very insulting to say that physicians just force patients to go on medicines that are going to make them a bunch of money.”
In a June report to Congress, MedPAC recommended reducing the add-on payment for many drugs given in hospitals and clinics, which are thus covered by Part B, as part of a package of suggestions for addressing rising costs. Part B drug spending grew about 9% annually between 2009 and 2021, rising from $15.4 billion to $42.9 billion, MedPAC said.
Medicare’s current Part B drug pricing model starts with the reported average sales price (ASP) and then adds about 4.3% or 6%, depending on current budget-sequester law, to the cost of medicines.
MedPAC members voted 17-0 in April in favor of a general recommendation to revise the Part B payment approach. In the June report, MedPAC fleshes out this idea. It mentions a model in which the add-on Part B payment would be the lesser of either 6% of the ASP, 3% plus $24, or $220.
The majority of Part B drug administrations are for very low-priced drugs, MedPAC said. But for some of the more costly ones, annual prices can be more than $400,000 per patient, and future launch prices may be even higher for certain types of products, such as gene therapies, MedPAC said.
“There is no evidence that the costs of a drug’s administration are proportionate to the price of the drug,” MedPAC said.
Concerns about how well Medicare covers the cost of drug administration should be addressed through other pathways, such as the American Medical
Association’s Specialty Society Relative Value Scale Update Committee (RUC), MedPAC said. AMA’s RUC advises the Centers for Medicare & Medicaid Services on the physician fee schedule.
Congress is not obliged to act on or to even consider MedPAC’s work. In general, lawmakers and CMS often pay heed to the panel’s recommendations, sometimes incorporating them into new policy.
But this new MedPAC Part B recommendation has drawn strong opposition, similar to the response to a 2016 CMS plan to cut the Part B add-on payment. That plan, which CMS later abandoned, would have cut the markup on Part B drugs to 2.5% and added a flat fee to cover administration costs.
Why not focus on PBMs instead?
The timing of the MedPAC recommendation is poor, given that CMS already is trying to implement the Inflation Reduction Act and create a new system of direct Medicare drug price negotiations, as ordered by Congress, said Madelaine A. Feldman, MD, a rheumatologist based in New Orleans.
A better approach for lowering drug prices would be to focus more on the operations of pharmacy benefit managers (PBMs), said Dr. Feldman, who also is vice president for advocacy and government affairs for the Coalition of State Rheumatology Organizations. A pending bipartisan Senate bill, for example, would prohibit PBM compensation based on the price of a drug as a condition of entering into a contract with a Medicare Part D plan.
Congress needs to take steps to unlink the profits of PBMs from higher drug prices, Dr. Feldman said.
“Until that happens, we can put all the lipstick we want on this big pig, but it’s not going to really fix the problem,” she said.
Reduced pay for drugs acquired through 340B program?
In an interview about the new MedPAC proposal, Ted Okon, executive director of the Community Oncology Alliance, urged renewed attention to what he sees as unintended consequences of the 340B discount drug program.
Under this program, certain hospitals can acquire drugs at steeply reduced prices, but they are not obliged to share those discounts with patients. Hospitals that participate in the 340B program can gain funds when patients and their insurers, including Medicare, pay more for the medicines hospitals and other organizations acquired with the 340B discount. Hospitals say they use the money from the 340B program to expand resources in their communities.
But rapid growth of the program in recent years has led to questions, especially about the role of contract pharmacies that manage the program. Congress created the 340B program in 1992 as a workaround to then new rules on Medicaid drug coverage.
In 2021, participating hospitals and clinics and organizations purchased about $44 billion worth of medicines through the 340B drug program. This was an increase of 16% from the previous year, according to a report from the nonprofit Commonwealth Fund. The number of sites, including hospitals and pharmacies, enrolled in the 340B program rose from 8,100 in 2000 to 50,000 by 2020, the report said.
MedPAC in 2016 urged CMS to reduce the amount Medicare pays for drugs acquired through the 340B program. CMS did so during the Trump administration, a policy later defended by the Biden administration.
But the U.S. Supreme Court last year said Medicare erred in its approach to making this cut, as earlier reported. Federal law required that the Department of Health and Human Services conduct a survey to support such a step, and HHS did not do this, the court said. CMS thus was ordered to return Medicare to the ASP+6% payment model for drugs purchased through the 340B discount program.
In the June report, though, MedPAC stuck by its 2016 recommendation that Medicare reduce its payments for drugs purchased through the 340B discount program despite this setback.
“We continue to believe that this approach is appropriate, and the specific level of payment reduction could be considered further as newer data become available,” MedPAC said.
Hospital, PhRMA split
Hospitals would certainly contest any renewed bid by CMS to drop Medicare’s pay for drugs purchased through the 340B program. The American Hospital Association objected to the MedPAC proposal regarding the add-on payment in Part B drug pricing.
MedPAC commissioners discussed this idea at a January meeting, prompting a February letter from the AHA to the panel. Like Dr. Feldman, AHA said it would be “premature” to launch into a revision of Part B drug pricing while the impact of the IRA on drug prices was still unclear.
AHA also noted that a reduction in Part B drug reimbursement would “shift the responsibility for the rapid increase in drug prices away from drug manufacturers, and instead places the burden on hospitals and patients.”
But the AHA gave a much warmer reception to another proposal MedPAC considered this year and that it included in its June report, which is a plan to address the high cost of certain drugs of as yet unconfirmed clinical benefit.
In April, the AHA said it supports a move toward a “value-based approach” in certain cases in which first-in-class medicines are sold under U.S. Food and Drug Administration’s accelerated approvals. Medicare could then cap payment for such drugs that have excessively high launch prices and uncertain clinical benefit, AHA said.
In the June report, MedPAC recommended that Medicare be able to place such a limit on Part B payments in certain cases, including ones in which companies do not meet FDA deadlines for postmarketing confirmatory trials.
The Pharmaceutical Research and Manufacturers of America (PhRMA) objected to this proposed change. The trade group for drugmakers said the FDA often revises and extends enrollment milestones for pending confirmatory trials when companies hit snags, such as challenges in enrolling patients, PhRMA said.
Reducing Part B payment for drugs for which confirmatory trials have been delayed would have a “disproportionate impact” on smaller and rural communities, where independent practices struggle to keep their doors open as it is, PhRMA spokeswoman Nicole Longo wrote in a blog post.
“If physicians can’t afford to administer a medicine, then they won’t and that means their patients won’t have access to them either,” Ms. Longo wrote.
A version of this article first appeared on Medscape.com.
Migraine clusters emerge from machine-learning analysis
AUSTIN, TEX. – The findings could point to new therapeutic strategies, according to study author Ali Ezzati, MD.
“A lot of diagnostic criteria that we have in the migraine world come from consensus groups of experts, and based on their experience and available data. They classify different types of headache and then on top of that different types of migraine. Unfortunately, this type of classification does not necessarily lead to having very homogeneous groups,” said Dr. Ezzati, who presented the study at the annual meeting of the American Headache Society.
Migraines are generally categorized as episodic (0-14 headache days per month) or chronic (15 or more per month), or as with or without aura. But these broad categories fail to capture the true diversity of migraine, according to Dr. Ezzati, and this may contribute to the fact that response to migraine therapy hovers around 60%. “We feel that the key to improving therapeutic efficacy is to identify individuals who are more homogeneous, more similar to each other, so that when we give a treatment, it is specifically targeting the underlying pathophysiology that those people have,” said Dr. Ezzati, who is an associate professor of neurology and director of the neuroinformatics program at University of California, Irvine.
The analysis revealed some clinically interesting results, said Dr. Ezzati. “For example, allodynia is a symptom that is not particularly used for classification of different types of migraine. There was a specific group that was very high in allodynia, and they were not very responsive to treatments, so that might be a [group] that people have to focus on. Also, we talk a lot about comorbidities in migraine, but we don’t talk about how these comorbidities affect the therapeutic strategies and treatment response to specific medications. We showed that people who have depression are actually less responsive than other groups to treatments, especially prescription medications,” he said.
Machine learning reveals clusters
The researchers analyzed data from 4,423 patients drawn from the American Migraine Prevalence and Prevention Study, which was conducted every year between 2005 and 2009. They included adult patients who filled out surveys in both 2006 and 2007. The study population was 83.7% female and had a mean age of 46.8 years, and 6.4% had chronic migraine. The researchers then used a machine-learning based self-organizing map to group patients into similar clusters.
The algorithm produced five such groups: Cluster 1 had the lowest symptom severity, and 0.6% had chronic migraine. Cluster 2 had mild symptom severity with no chronic migraine. Cluster 3 had moderate symptom severity and a high prevalence of allodynia (88.5%, vs. 63.4% overall, P < .001) and no chronic migraine. Cluster 4 had a high frequency of depressive symptoms (63.1% vs. 19.8% overall, P < .001) and 5.2% had chronic migraine. Cluster 5 had frequent and severe migraines, and most (83.0%) had chronic migraine (P < .001).
There were some other broader trends. Triptans were more commonly used in clusters 2 (25.6%), 3 (27.9%), and 5 (28.0%), but less so in cluster 4 (17.1%; P < .001). Pain freedom at 2 hours was most common in cluster 1 (53.1%), followed by cluster 2 (46.4%), but was significantly less frequent in clusters 3 (32.2%), 4 (32.2%), and 5 (34.7%; P < .001).
Therapeutic implications
Dr. Ezzati believes that machine learning and data analysis could point the way to a future of more tailored migraine therapies. “I think we have to in general go down the path of using more evidence and more data to inform us about individualized planning for patients. For that we need larger clinical studies and larger epidemiological studies to help us identify more homogeneous subtypes of patients that we can eventually target in clinical trials,” he said.
Catherine Chong, MD, who chaired the session where the research was presented, praised the study in an interview. “Episodic migraine and chronic migraine have been developed [as categories] by headache frequency per month, and it was basically based on consensus in committee. They made basically a determination that 15 and under migraine days would be episodic migraine and over would be chronic migraine. So they dichotomized migraine, in a way, based on what people thought in the field. Looking at the data freely, and letting the algorithm determine the different subtypes, and putting everybody with migraine in it, and having these groups naturally appear from the data, I think is fascinating,” Dr. Chong said.
She echoed Dr. Ezzati’s call for further research that could create even more subgroups. “Is it really truly the case that somebody with less than 15 migraine days [per month], that 14 migraines days would be so different than somebody with 15 or over, or 8? I think we need to look at it further to see whether there are additional subgroups within that data. I think there are probably more [groups identifiable] from different data that we have out there,” said Dr. Chong.
Dr. Ezzati has consulted for or been a reviewer or advisory board member for Corium, Eisai, GlaxoSmithKline, Mint Research, and Health Care Horizon Scanning System. He has received research funding from Amgen. Dr. Chong has no relevant financial disclosures.
AUSTIN, TEX. – The findings could point to new therapeutic strategies, according to study author Ali Ezzati, MD.
“A lot of diagnostic criteria that we have in the migraine world come from consensus groups of experts, and based on their experience and available data. They classify different types of headache and then on top of that different types of migraine. Unfortunately, this type of classification does not necessarily lead to having very homogeneous groups,” said Dr. Ezzati, who presented the study at the annual meeting of the American Headache Society.
Migraines are generally categorized as episodic (0-14 headache days per month) or chronic (15 or more per month), or as with or without aura. But these broad categories fail to capture the true diversity of migraine, according to Dr. Ezzati, and this may contribute to the fact that response to migraine therapy hovers around 60%. “We feel that the key to improving therapeutic efficacy is to identify individuals who are more homogeneous, more similar to each other, so that when we give a treatment, it is specifically targeting the underlying pathophysiology that those people have,” said Dr. Ezzati, who is an associate professor of neurology and director of the neuroinformatics program at University of California, Irvine.
The analysis revealed some clinically interesting results, said Dr. Ezzati. “For example, allodynia is a symptom that is not particularly used for classification of different types of migraine. There was a specific group that was very high in allodynia, and they were not very responsive to treatments, so that might be a [group] that people have to focus on. Also, we talk a lot about comorbidities in migraine, but we don’t talk about how these comorbidities affect the therapeutic strategies and treatment response to specific medications. We showed that people who have depression are actually less responsive than other groups to treatments, especially prescription medications,” he said.
Machine learning reveals clusters
The researchers analyzed data from 4,423 patients drawn from the American Migraine Prevalence and Prevention Study, which was conducted every year between 2005 and 2009. They included adult patients who filled out surveys in both 2006 and 2007. The study population was 83.7% female and had a mean age of 46.8 years, and 6.4% had chronic migraine. The researchers then used a machine-learning based self-organizing map to group patients into similar clusters.
The algorithm produced five such groups: Cluster 1 had the lowest symptom severity, and 0.6% had chronic migraine. Cluster 2 had mild symptom severity with no chronic migraine. Cluster 3 had moderate symptom severity and a high prevalence of allodynia (88.5%, vs. 63.4% overall, P < .001) and no chronic migraine. Cluster 4 had a high frequency of depressive symptoms (63.1% vs. 19.8% overall, P < .001) and 5.2% had chronic migraine. Cluster 5 had frequent and severe migraines, and most (83.0%) had chronic migraine (P < .001).
There were some other broader trends. Triptans were more commonly used in clusters 2 (25.6%), 3 (27.9%), and 5 (28.0%), but less so in cluster 4 (17.1%; P < .001). Pain freedom at 2 hours was most common in cluster 1 (53.1%), followed by cluster 2 (46.4%), but was significantly less frequent in clusters 3 (32.2%), 4 (32.2%), and 5 (34.7%; P < .001).
Therapeutic implications
Dr. Ezzati believes that machine learning and data analysis could point the way to a future of more tailored migraine therapies. “I think we have to in general go down the path of using more evidence and more data to inform us about individualized planning for patients. For that we need larger clinical studies and larger epidemiological studies to help us identify more homogeneous subtypes of patients that we can eventually target in clinical trials,” he said.
Catherine Chong, MD, who chaired the session where the research was presented, praised the study in an interview. “Episodic migraine and chronic migraine have been developed [as categories] by headache frequency per month, and it was basically based on consensus in committee. They made basically a determination that 15 and under migraine days would be episodic migraine and over would be chronic migraine. So they dichotomized migraine, in a way, based on what people thought in the field. Looking at the data freely, and letting the algorithm determine the different subtypes, and putting everybody with migraine in it, and having these groups naturally appear from the data, I think is fascinating,” Dr. Chong said.
She echoed Dr. Ezzati’s call for further research that could create even more subgroups. “Is it really truly the case that somebody with less than 15 migraine days [per month], that 14 migraines days would be so different than somebody with 15 or over, or 8? I think we need to look at it further to see whether there are additional subgroups within that data. I think there are probably more [groups identifiable] from different data that we have out there,” said Dr. Chong.
Dr. Ezzati has consulted for or been a reviewer or advisory board member for Corium, Eisai, GlaxoSmithKline, Mint Research, and Health Care Horizon Scanning System. He has received research funding from Amgen. Dr. Chong has no relevant financial disclosures.
AUSTIN, TEX. – The findings could point to new therapeutic strategies, according to study author Ali Ezzati, MD.
“A lot of diagnostic criteria that we have in the migraine world come from consensus groups of experts, and based on their experience and available data. They classify different types of headache and then on top of that different types of migraine. Unfortunately, this type of classification does not necessarily lead to having very homogeneous groups,” said Dr. Ezzati, who presented the study at the annual meeting of the American Headache Society.
Migraines are generally categorized as episodic (0-14 headache days per month) or chronic (15 or more per month), or as with or without aura. But these broad categories fail to capture the true diversity of migraine, according to Dr. Ezzati, and this may contribute to the fact that response to migraine therapy hovers around 60%. “We feel that the key to improving therapeutic efficacy is to identify individuals who are more homogeneous, more similar to each other, so that when we give a treatment, it is specifically targeting the underlying pathophysiology that those people have,” said Dr. Ezzati, who is an associate professor of neurology and director of the neuroinformatics program at University of California, Irvine.
The analysis revealed some clinically interesting results, said Dr. Ezzati. “For example, allodynia is a symptom that is not particularly used for classification of different types of migraine. There was a specific group that was very high in allodynia, and they were not very responsive to treatments, so that might be a [group] that people have to focus on. Also, we talk a lot about comorbidities in migraine, but we don’t talk about how these comorbidities affect the therapeutic strategies and treatment response to specific medications. We showed that people who have depression are actually less responsive than other groups to treatments, especially prescription medications,” he said.
Machine learning reveals clusters
The researchers analyzed data from 4,423 patients drawn from the American Migraine Prevalence and Prevention Study, which was conducted every year between 2005 and 2009. They included adult patients who filled out surveys in both 2006 and 2007. The study population was 83.7% female and had a mean age of 46.8 years, and 6.4% had chronic migraine. The researchers then used a machine-learning based self-organizing map to group patients into similar clusters.
The algorithm produced five such groups: Cluster 1 had the lowest symptom severity, and 0.6% had chronic migraine. Cluster 2 had mild symptom severity with no chronic migraine. Cluster 3 had moderate symptom severity and a high prevalence of allodynia (88.5%, vs. 63.4% overall, P < .001) and no chronic migraine. Cluster 4 had a high frequency of depressive symptoms (63.1% vs. 19.8% overall, P < .001) and 5.2% had chronic migraine. Cluster 5 had frequent and severe migraines, and most (83.0%) had chronic migraine (P < .001).
There were some other broader trends. Triptans were more commonly used in clusters 2 (25.6%), 3 (27.9%), and 5 (28.0%), but less so in cluster 4 (17.1%; P < .001). Pain freedom at 2 hours was most common in cluster 1 (53.1%), followed by cluster 2 (46.4%), but was significantly less frequent in clusters 3 (32.2%), 4 (32.2%), and 5 (34.7%; P < .001).
Therapeutic implications
Dr. Ezzati believes that machine learning and data analysis could point the way to a future of more tailored migraine therapies. “I think we have to in general go down the path of using more evidence and more data to inform us about individualized planning for patients. For that we need larger clinical studies and larger epidemiological studies to help us identify more homogeneous subtypes of patients that we can eventually target in clinical trials,” he said.
Catherine Chong, MD, who chaired the session where the research was presented, praised the study in an interview. “Episodic migraine and chronic migraine have been developed [as categories] by headache frequency per month, and it was basically based on consensus in committee. They made basically a determination that 15 and under migraine days would be episodic migraine and over would be chronic migraine. So they dichotomized migraine, in a way, based on what people thought in the field. Looking at the data freely, and letting the algorithm determine the different subtypes, and putting everybody with migraine in it, and having these groups naturally appear from the data, I think is fascinating,” Dr. Chong said.
She echoed Dr. Ezzati’s call for further research that could create even more subgroups. “Is it really truly the case that somebody with less than 15 migraine days [per month], that 14 migraines days would be so different than somebody with 15 or over, or 8? I think we need to look at it further to see whether there are additional subgroups within that data. I think there are probably more [groups identifiable] from different data that we have out there,” said Dr. Chong.
Dr. Ezzati has consulted for or been a reviewer or advisory board member for Corium, Eisai, GlaxoSmithKline, Mint Research, and Health Care Horizon Scanning System. He has received research funding from Amgen. Dr. Chong has no relevant financial disclosures.
FROM AHS 2023
You’ve been warned
Recently, Canada announced new regulations on tobacco, with warnings printed on individual cigarettes, such as “poison in every puff.” This is on top of the packaging already required to have 75% of its space devoted to similar warnings, often with graphic pictures, of the potential consequences.
Make no mistake, I don’t like cigarettes and try to get smokers to quit.
But I have to wonder how successful this is going to be. I mean, you’d have to have lived under a rock for the last 70 years (or more) to not know that cigarettes (and tobacco in general) aren’t good for you, and can cause stroke, heart disease, and a multitude of cancers.
I suppose you could ban cigarettes, but that only opens up a black market. From 1920 to 1933 the United States set an example for the world with prohibition, showing how such an idea can backfire horribly.
Realistically, there are always going to be people making bad health decisions of one kind or another, including myself. Whether it’s tobacco, alcohol, or a cheeseburger and fries.
Tobacco, of course, has a much worse track record than that of the cheeseburger. We all have to eat, even though some choices are better than others. Tobacco has absolutely no biological necessity, as do food, air, and water.
But it’s remarkably addictive, not to mention profitable. Those factors will always guarantee it a place in society.
At this point,
There’s a legitimate argument to be made in trying to keep people from starting. The teenage years, where we all tend to believe we’re immortal, are when a lot of habits (good and bad) form. If gruesome pictures and repeated warnings cut down on those numbers, then in the long run it’s a very good thing. Given that Canada’s goal is to cut tobacco use from 13% down to less than 5% by 2035, this could happen. Only time will tell how it plays out.
On a side note, here in the United States tobacco use is 19% of the population. This is actually somewhat surprising to me, as a brief, not particularly scientific, review of my charts for the past few weeks found that less than 5% of my patients do it. So either some are lying or (more likely), it’s just the demographics of my practice area.
But at some point it doesn’t matter how many warnings or gory pictures people see, or where they encounter them. Some will keep smoking out of habit. Some because they actually like it. Some to be defiant. Some just because they can. And no amount of warnings is going to change their minds.
Dr. Block has a solo neurology practice in Scottsdale, Ariz.
Recently, Canada announced new regulations on tobacco, with warnings printed on individual cigarettes, such as “poison in every puff.” This is on top of the packaging already required to have 75% of its space devoted to similar warnings, often with graphic pictures, of the potential consequences.
Make no mistake, I don’t like cigarettes and try to get smokers to quit.
But I have to wonder how successful this is going to be. I mean, you’d have to have lived under a rock for the last 70 years (or more) to not know that cigarettes (and tobacco in general) aren’t good for you, and can cause stroke, heart disease, and a multitude of cancers.
I suppose you could ban cigarettes, but that only opens up a black market. From 1920 to 1933 the United States set an example for the world with prohibition, showing how such an idea can backfire horribly.
Realistically, there are always going to be people making bad health decisions of one kind or another, including myself. Whether it’s tobacco, alcohol, or a cheeseburger and fries.
Tobacco, of course, has a much worse track record than that of the cheeseburger. We all have to eat, even though some choices are better than others. Tobacco has absolutely no biological necessity, as do food, air, and water.
But it’s remarkably addictive, not to mention profitable. Those factors will always guarantee it a place in society.
At this point,
There’s a legitimate argument to be made in trying to keep people from starting. The teenage years, where we all tend to believe we’re immortal, are when a lot of habits (good and bad) form. If gruesome pictures and repeated warnings cut down on those numbers, then in the long run it’s a very good thing. Given that Canada’s goal is to cut tobacco use from 13% down to less than 5% by 2035, this could happen. Only time will tell how it plays out.
On a side note, here in the United States tobacco use is 19% of the population. This is actually somewhat surprising to me, as a brief, not particularly scientific, review of my charts for the past few weeks found that less than 5% of my patients do it. So either some are lying or (more likely), it’s just the demographics of my practice area.
But at some point it doesn’t matter how many warnings or gory pictures people see, or where they encounter them. Some will keep smoking out of habit. Some because they actually like it. Some to be defiant. Some just because they can. And no amount of warnings is going to change their minds.
Dr. Block has a solo neurology practice in Scottsdale, Ariz.
Recently, Canada announced new regulations on tobacco, with warnings printed on individual cigarettes, such as “poison in every puff.” This is on top of the packaging already required to have 75% of its space devoted to similar warnings, often with graphic pictures, of the potential consequences.
Make no mistake, I don’t like cigarettes and try to get smokers to quit.
But I have to wonder how successful this is going to be. I mean, you’d have to have lived under a rock for the last 70 years (or more) to not know that cigarettes (and tobacco in general) aren’t good for you, and can cause stroke, heart disease, and a multitude of cancers.
I suppose you could ban cigarettes, but that only opens up a black market. From 1920 to 1933 the United States set an example for the world with prohibition, showing how such an idea can backfire horribly.
Realistically, there are always going to be people making bad health decisions of one kind or another, including myself. Whether it’s tobacco, alcohol, or a cheeseburger and fries.
Tobacco, of course, has a much worse track record than that of the cheeseburger. We all have to eat, even though some choices are better than others. Tobacco has absolutely no biological necessity, as do food, air, and water.
But it’s remarkably addictive, not to mention profitable. Those factors will always guarantee it a place in society.
At this point,
There’s a legitimate argument to be made in trying to keep people from starting. The teenage years, where we all tend to believe we’re immortal, are when a lot of habits (good and bad) form. If gruesome pictures and repeated warnings cut down on those numbers, then in the long run it’s a very good thing. Given that Canada’s goal is to cut tobacco use from 13% down to less than 5% by 2035, this could happen. Only time will tell how it plays out.
On a side note, here in the United States tobacco use is 19% of the population. This is actually somewhat surprising to me, as a brief, not particularly scientific, review of my charts for the past few weeks found that less than 5% of my patients do it. So either some are lying or (more likely), it’s just the demographics of my practice area.
But at some point it doesn’t matter how many warnings or gory pictures people see, or where they encounter them. Some will keep smoking out of habit. Some because they actually like it. Some to be defiant. Some just because they can. And no amount of warnings is going to change their minds.
Dr. Block has a solo neurology practice in Scottsdale, Ariz.