User login
From famous to infamous: Psychiatric aspects of the fall from grace
It’s an all-too-common news item: The crash and burn of yet another politician, celebrity, or prominent individual. It’s painful to watch someone who spent years to achieve the status of a household name suddenly, and often ignominiously, lose it all. This is the equivalent of a human train wreck.
Some adversaries (who doesn’t have a few?) will rejoice or express schadenfreude, but many people will experience some empathy or sorrow as they witness the implosion of a celebrity. Fans, followers, or voters may grieve as the object of their respect and adulation falls off the high pedestal of fame. What starts as a drip-drip of rumors and innuendos soon eventuates in a denouement. And with time, as additional public figures fall from grace, the previous casualties will become mere footnotes in the annals of human self-destruction. Their loss of face, shame, and wrenching emotional and financial toll will be forgotten from the public’s collective memory, but the embers of bitterness and regret will continue to smolder in the hearts and souls of those who inadvertently contributed to their own social or professional demise due to a mistake, error of judgement, or plain old-fashioned stupidity. For the fallen, forgiveness and redemption are hard to come by.
Oh, how the mighty have fallen over centuries, and they include historical figures such as kings, military leaders, religious leaders, and politicians. The fall from grace in the past often led to executions, excommunication, or persecution. In the contemporary era, the oppressive “cancel culture” will mercilessly discard anyone, regardless of stature, after only 1 “wrong” tweet. In the digital age of mass communication, being “cancelled” is a frequent fall from grace and is the equivalent of being ostracized from millions of denizens on social media, which can spell doom for one’s career and social interactions.
The list of those whose careers ended calamitously include many familiar names, but I will only cite their prominent roles (you can easily guess their names!):
- emperors, kings, presidents, prime ministers, and political demagogues
- congressmen, senators, governors, and mayors
- Nobel Laureates (a Medicine and Physiology winner went to prison for pedophilia, and a Peace Prize winner fell from grace for supporting a military dictatorship)
- Cardinals and bishops in various countries (for sexual or financial crimes)
- billionaires, often for erratic personal lives
- sport legends, including decorated athletes and coaches of college and professional teams
- world chess masters
- Wall Street moguls
- Hollywood celebrities, including actors and directors, some with Oscars and related recognitions
- television news anchors and commentators
- comedians of various stripes
- CEOs of major media companies
- talk show hosts watched by millions
- celebrated musicians (classical, pop, rap, or blues)
- university presidents
- others in esteemed positions (including some psychiatrists).
Why is this so common?
From a psychiatric perspective, the most compelling question is why is the fall from grace so common? What are the transgressions, flaws, and shortcomings of successful individuals whose reputations end up smeared or who lose everything they worked for? Why do high achievers, talented and successful, at the apogee of fame and fortune, lose it all with nary a chance for recovery
The answer is all too obvious: human frailties. Successful persons are by no means immune from poor judgment. They can be as error-prone as the rest of us mortals. Having robust cognitive intelligence can be undermined by stunted emotional intelligence or poor interpersonal or social judgment. In Freudian terms, famous people who crash and burn may have a “Swiss cheese superego” that allows their id to viciously weaken their ego. From a neuroscience perspective, their limbic system conquers their cortical circuitry with relentless innate forces, including:
- fervent sexual appetite, compounded by the cockiness that comes with fame
- felonious paraphilias, such as pedophilia or public indecency
- intense greed that clouds one’s judgment (a trait exhibited by some ultra-rich persons)
- narcissism, either inborn or acquired with unexpected success and power
- impulsivity and recklessness, with injurious words or actions.
- substance use.
Consideration should be given to psychopathology. Some may have a personality disorder. Others may be both blessed and cursed with hypomania that leads to high achievement but also to foolish and impulsive behavior.1 Some may have maladaptive social skills seen in autism spectrum disorder (recently, a very prominent and innovative billionaire casually announced that he has autistic traits). And others my have limited coping skills to deal with fame and fortune and unwittingly end up shooting themselves in both feet.
Continue to: But perhaps the most common thread...
But perhaps the most common thread across all the tragic cases of self-destruction is hubris. As humans become rich, famous, or powerful, they gradually develop the fallacious belief that they can get away with anything because they have masses of fans and followers who “love them no matter what.” This dangerous “acquired narcissism” is an unfortunate byproduct of success. Humility is rare among celebrities and powerful leaders. Modest celebrities almost never fall from grace and are endowed with an innate antidote to self-aggrandizement. A few years ago, I wrote an editorial in
In contemporary society, with the era of social media and toxic political zeitgeist, there are many inadvertent “opportunities” to stumble and ruin one’s career by uttering an “unacceptable” word or dispatching an “offensive tweet” or posting a politically incorrect photo. And even if one is currently careful, there are now social media detectives and fact-finding “archeologists” who can excavate and disseminate the faux pas, peccadillos, or misdeeds from a prominent person’s immature youth, which will destroy a famous person overnight. That can be a nightmare for anyone who becomes a bona fide celebrity after years of working hard to get there.
High achievers: Beware!
1. Gartner JD. The hypomanic edge: the link between (a little) craziness and (a lot of) success in America. Simon & Schuster; 2005.
2. Nasrallah HA. Should psychiatry list hubris in DSM-V? Current Psychiatry. 2008;7(12):14-15.
It’s an all-too-common news item: The crash and burn of yet another politician, celebrity, or prominent individual. It’s painful to watch someone who spent years to achieve the status of a household name suddenly, and often ignominiously, lose it all. This is the equivalent of a human train wreck.
Some adversaries (who doesn’t have a few?) will rejoice or express schadenfreude, but many people will experience some empathy or sorrow as they witness the implosion of a celebrity. Fans, followers, or voters may grieve as the object of their respect and adulation falls off the high pedestal of fame. What starts as a drip-drip of rumors and innuendos soon eventuates in a denouement. And with time, as additional public figures fall from grace, the previous casualties will become mere footnotes in the annals of human self-destruction. Their loss of face, shame, and wrenching emotional and financial toll will be forgotten from the public’s collective memory, but the embers of bitterness and regret will continue to smolder in the hearts and souls of those who inadvertently contributed to their own social or professional demise due to a mistake, error of judgement, or plain old-fashioned stupidity. For the fallen, forgiveness and redemption are hard to come by.
Oh, how the mighty have fallen over centuries, and they include historical figures such as kings, military leaders, religious leaders, and politicians. The fall from grace in the past often led to executions, excommunication, or persecution. In the contemporary era, the oppressive “cancel culture” will mercilessly discard anyone, regardless of stature, after only 1 “wrong” tweet. In the digital age of mass communication, being “cancelled” is a frequent fall from grace and is the equivalent of being ostracized from millions of denizens on social media, which can spell doom for one’s career and social interactions.
The list of those whose careers ended calamitously include many familiar names, but I will only cite their prominent roles (you can easily guess their names!):
- emperors, kings, presidents, prime ministers, and political demagogues
- congressmen, senators, governors, and mayors
- Nobel Laureates (a Medicine and Physiology winner went to prison for pedophilia, and a Peace Prize winner fell from grace for supporting a military dictatorship)
- Cardinals and bishops in various countries (for sexual or financial crimes)
- billionaires, often for erratic personal lives
- sport legends, including decorated athletes and coaches of college and professional teams
- world chess masters
- Wall Street moguls
- Hollywood celebrities, including actors and directors, some with Oscars and related recognitions
- television news anchors and commentators
- comedians of various stripes
- CEOs of major media companies
- talk show hosts watched by millions
- celebrated musicians (classical, pop, rap, or blues)
- university presidents
- others in esteemed positions (including some psychiatrists).
Why is this so common?
From a psychiatric perspective, the most compelling question is why is the fall from grace so common? What are the transgressions, flaws, and shortcomings of successful individuals whose reputations end up smeared or who lose everything they worked for? Why do high achievers, talented and successful, at the apogee of fame and fortune, lose it all with nary a chance for recovery
The answer is all too obvious: human frailties. Successful persons are by no means immune from poor judgment. They can be as error-prone as the rest of us mortals. Having robust cognitive intelligence can be undermined by stunted emotional intelligence or poor interpersonal or social judgment. In Freudian terms, famous people who crash and burn may have a “Swiss cheese superego” that allows their id to viciously weaken their ego. From a neuroscience perspective, their limbic system conquers their cortical circuitry with relentless innate forces, including:
- fervent sexual appetite, compounded by the cockiness that comes with fame
- felonious paraphilias, such as pedophilia or public indecency
- intense greed that clouds one’s judgment (a trait exhibited by some ultra-rich persons)
- narcissism, either inborn or acquired with unexpected success and power
- impulsivity and recklessness, with injurious words or actions.
- substance use.
Consideration should be given to psychopathology. Some may have a personality disorder. Others may be both blessed and cursed with hypomania that leads to high achievement but also to foolish and impulsive behavior.1 Some may have maladaptive social skills seen in autism spectrum disorder (recently, a very prominent and innovative billionaire casually announced that he has autistic traits). And others my have limited coping skills to deal with fame and fortune and unwittingly end up shooting themselves in both feet.
Continue to: But perhaps the most common thread...
But perhaps the most common thread across all the tragic cases of self-destruction is hubris. As humans become rich, famous, or powerful, they gradually develop the fallacious belief that they can get away with anything because they have masses of fans and followers who “love them no matter what.” This dangerous “acquired narcissism” is an unfortunate byproduct of success. Humility is rare among celebrities and powerful leaders. Modest celebrities almost never fall from grace and are endowed with an innate antidote to self-aggrandizement. A few years ago, I wrote an editorial in
In contemporary society, with the era of social media and toxic political zeitgeist, there are many inadvertent “opportunities” to stumble and ruin one’s career by uttering an “unacceptable” word or dispatching an “offensive tweet” or posting a politically incorrect photo. And even if one is currently careful, there are now social media detectives and fact-finding “archeologists” who can excavate and disseminate the faux pas, peccadillos, or misdeeds from a prominent person’s immature youth, which will destroy a famous person overnight. That can be a nightmare for anyone who becomes a bona fide celebrity after years of working hard to get there.
High achievers: Beware!
It’s an all-too-common news item: The crash and burn of yet another politician, celebrity, or prominent individual. It’s painful to watch someone who spent years to achieve the status of a household name suddenly, and often ignominiously, lose it all. This is the equivalent of a human train wreck.
Some adversaries (who doesn’t have a few?) will rejoice or express schadenfreude, but many people will experience some empathy or sorrow as they witness the implosion of a celebrity. Fans, followers, or voters may grieve as the object of their respect and adulation falls off the high pedestal of fame. What starts as a drip-drip of rumors and innuendos soon eventuates in a denouement. And with time, as additional public figures fall from grace, the previous casualties will become mere footnotes in the annals of human self-destruction. Their loss of face, shame, and wrenching emotional and financial toll will be forgotten from the public’s collective memory, but the embers of bitterness and regret will continue to smolder in the hearts and souls of those who inadvertently contributed to their own social or professional demise due to a mistake, error of judgement, or plain old-fashioned stupidity. For the fallen, forgiveness and redemption are hard to come by.
Oh, how the mighty have fallen over centuries, and they include historical figures such as kings, military leaders, religious leaders, and politicians. The fall from grace in the past often led to executions, excommunication, or persecution. In the contemporary era, the oppressive “cancel culture” will mercilessly discard anyone, regardless of stature, after only 1 “wrong” tweet. In the digital age of mass communication, being “cancelled” is a frequent fall from grace and is the equivalent of being ostracized from millions of denizens on social media, which can spell doom for one’s career and social interactions.
The list of those whose careers ended calamitously include many familiar names, but I will only cite their prominent roles (you can easily guess their names!):
- emperors, kings, presidents, prime ministers, and political demagogues
- congressmen, senators, governors, and mayors
- Nobel Laureates (a Medicine and Physiology winner went to prison for pedophilia, and a Peace Prize winner fell from grace for supporting a military dictatorship)
- Cardinals and bishops in various countries (for sexual or financial crimes)
- billionaires, often for erratic personal lives
- sport legends, including decorated athletes and coaches of college and professional teams
- world chess masters
- Wall Street moguls
- Hollywood celebrities, including actors and directors, some with Oscars and related recognitions
- television news anchors and commentators
- comedians of various stripes
- CEOs of major media companies
- talk show hosts watched by millions
- celebrated musicians (classical, pop, rap, or blues)
- university presidents
- others in esteemed positions (including some psychiatrists).
Why is this so common?
From a psychiatric perspective, the most compelling question is why is the fall from grace so common? What are the transgressions, flaws, and shortcomings of successful individuals whose reputations end up smeared or who lose everything they worked for? Why do high achievers, talented and successful, at the apogee of fame and fortune, lose it all with nary a chance for recovery
The answer is all too obvious: human frailties. Successful persons are by no means immune from poor judgment. They can be as error-prone as the rest of us mortals. Having robust cognitive intelligence can be undermined by stunted emotional intelligence or poor interpersonal or social judgment. In Freudian terms, famous people who crash and burn may have a “Swiss cheese superego” that allows their id to viciously weaken their ego. From a neuroscience perspective, their limbic system conquers their cortical circuitry with relentless innate forces, including:
- fervent sexual appetite, compounded by the cockiness that comes with fame
- felonious paraphilias, such as pedophilia or public indecency
- intense greed that clouds one’s judgment (a trait exhibited by some ultra-rich persons)
- narcissism, either inborn or acquired with unexpected success and power
- impulsivity and recklessness, with injurious words or actions.
- substance use.
Consideration should be given to psychopathology. Some may have a personality disorder. Others may be both blessed and cursed with hypomania that leads to high achievement but also to foolish and impulsive behavior.1 Some may have maladaptive social skills seen in autism spectrum disorder (recently, a very prominent and innovative billionaire casually announced that he has autistic traits). And others my have limited coping skills to deal with fame and fortune and unwittingly end up shooting themselves in both feet.
Continue to: But perhaps the most common thread...
But perhaps the most common thread across all the tragic cases of self-destruction is hubris. As humans become rich, famous, or powerful, they gradually develop the fallacious belief that they can get away with anything because they have masses of fans and followers who “love them no matter what.” This dangerous “acquired narcissism” is an unfortunate byproduct of success. Humility is rare among celebrities and powerful leaders. Modest celebrities almost never fall from grace and are endowed with an innate antidote to self-aggrandizement. A few years ago, I wrote an editorial in
In contemporary society, with the era of social media and toxic political zeitgeist, there are many inadvertent “opportunities” to stumble and ruin one’s career by uttering an “unacceptable” word or dispatching an “offensive tweet” or posting a politically incorrect photo. And even if one is currently careful, there are now social media detectives and fact-finding “archeologists” who can excavate and disseminate the faux pas, peccadillos, or misdeeds from a prominent person’s immature youth, which will destroy a famous person overnight. That can be a nightmare for anyone who becomes a bona fide celebrity after years of working hard to get there.
High achievers: Beware!
1. Gartner JD. The hypomanic edge: the link between (a little) craziness and (a lot of) success in America. Simon & Schuster; 2005.
2. Nasrallah HA. Should psychiatry list hubris in DSM-V? Current Psychiatry. 2008;7(12):14-15.
1. Gartner JD. The hypomanic edge: the link between (a little) craziness and (a lot of) success in America. Simon & Schuster; 2005.
2. Nasrallah HA. Should psychiatry list hubris in DSM-V? Current Psychiatry. 2008;7(12):14-15.
Persistent altered mental status
CASE Sluggish, weak, and incoherent
Mr. O, age 24, who has a history of schizophrenia and obesity, presents to the emergency department (ED) for altered mental status (AMS). His mother reports that he has been sluggish, weak, incoherent, had no appetite, and that on the day before admission, he was drinking excessive amounts of water and urinating every 10 minutes.
HISTORY Multiple ineffective antipsychotics
Mr. O was diagnosed with schizophrenia at age 21 and struggled with medication adherence, which resulted in multiple hospitalizations for stabilization. Trials of haloperidol, risperidone, paliperidone palmitate, and valproic acid had been ineffective. At the time of admission, his psychotropic medication regimen is fluphenazine decanoate, 25 mg injection every 2 weeks; clozapine, 50 mg/d; lithium carbonate, 300 mg twice a day; benztropine, 2 mg every night; and trazodone, 50 mg every night.
EVALUATION Fever, tachycardia, and diabetic ketoacidosis
Upon arrival to the ED, Mr. O is obtunded, unable to follow commands, and does not respond to painful stimuli. On physical exam, he has a fever of 38.4°C (reference range 35.1°C to 37.9°C); tachycardia with a heart rate of 142 beats per minute (bpm) (reference range 60 to 100); tachypnea with a respiratory rate of 35 breaths per minute (reference range 12 to 20); a blood pressure of 116/76 mmHg (reference range 90/60 to 130/80); and hypoxemia with an oxygen saturation of 90% on room air (reference range 94% to 100%).
Mr. O is admitted to the hospital and his laboratory workup indicates diabetic ketoacidosis (DKA), with a glucose of 1,700 mg/dL; anion gap of 30 (reference range 4 to 12 mmol/L); pH 7.04 (reference range 7.32 to 7.42); serum bicarbonate 6 (reference range 20 to 24 mEq/L); beta-hydroxybutyrate 11.04 (reference range 0 to 0.27 mmol/L); urine ketones, serum osmolality 407 (reference range 280 to 300 mOsm/kg); and an elevated white blood cell count of 18.4 (reference range 4.5 to 11.0 × 109/L). A CT scan of the head is negative for acute pathology.
Initially, all psychotropic medications are held. On Day 3 of hospitalization, psychiatry is consulted and clozapine, 50 mg/d; lithium, 300 mg/d; and benztropine, 1 mg at night, are restarted; however, fluphenazine decanoate and trazodone are held. The team recommends IV haloperidol, 2 mg as needed for agitation; however, it is never administered.
Imaging rules out deep vein thrombosis, cardiac dysfunction, and stroke, but a CT chest scan is notable for bilateral lung infiltrates, which suggests aspiration pneumonia.
Mr. O is diagnosed with diabetes, complicated by DKA, and is treated in the intensive care unit (ICU). Despite resolution of the DKA, he remains altered with fever and tachycardia.
Continue to: On Day 6 of hospitalization...
On Day 6 of hospitalization, Mr. O continues to be tachycardic and obtunded with nuchal rigidity. The team decides to transfer Mr. O to another hospital for a higher level of care and continued workup of his persistent AMS.
Immediately upon arrival at the second hospital, infectious disease and neurology teams are consulted for further evaluation. Mr. O’s AMS continues despite no clear signs of infection or other neurologic insults.
[polldaddy:10930631]
The authors’ observations
Based on Mr. O’s psychiatric history and laboratory results, the first medical team concluded his initial AMS was likely secondary to DKA; however, the AMS continued after the DKA resolved. At the second hospital, Mr. O’s treatment team continued to dig for answers.
EVALUATION Exploring the differential diagnosis
At the second hospital, Mr. O is admitted to the ICU with fever (37.8°C), tachycardia (120 bpm), tachypnea, withdrawal from painful stimuli, decreased reflexes, and muscle rigidity, including clenched jaw. The differential diagnoses include meningitis, sepsis from aspiration pneumonia, severe metabolic encephalopathy with prolonged recovery, central pontine myelinolysis, anoxic brain injury, and subclinical seizures.
Empiric vancomycin, 1.75 g every 12 hours; ceftriaxone, 2 g/d; and acyclovir, 900 mg every 8 hours are started for meningoencephalitis, and all psychotropic medications are discontinued. Case reports have documented a relationship between hyperglycemic hyperosmolar syndrome (HHS) and malignant hyperthermia in rare cases1; however, HHS is ruled out based on Mr. O’s laboratory results.A lumbar puncture and imaging rules out CNS infection. Antibiotic treatment is narrowed to ampicillin-sulbactam due to Mr. O’s prior CT chest showing concern for aspiration pneumonia. An MRI of the brain rules out central pontine myelinolysis, acute stroke, and anoxic brain injury, and an EEG shows nonspecific encephalopathy. On Day 10 of hospitalization, a neurologic exam shows flaccid paralysis and bilateral clonus, and Mr. O is mute. On Day 14 of hospitalization, his fever resolves, and his blood cultures are negative. On Day 15 of hospitalization, Mr. O’s creatine kinase (CK) level is elevated at 1,308 U/L (reference range 26 to 192 U/L), suggesting rhabdomyolysis.
Continue to: Given the neurologic exam findings...
Given the neurologic exam findings, and the limited evidence of infection, the differential diagnosis for Mr. O’s AMS is broadened to include catatonia, neuroleptic malignant syndrome (NMS), serotonin syndrome, and autoimmune encephalitis. The psychiatry team evaluates Mr. O for catatonia. He scores 14 on the Bush-Francis Catatonia Rating Scale, with findings of immobility/stupor, mutism, staring, autonomic instability, and withdrawal indicating the presence of catatonia.2
The authors’ observations
When Mr. O was transferred to the second hospital, the primary concern was to rule out meningitis due to his unstable vitals, obtunded mental state, and nuchal rigidity. A comprehensive infectious workup, including lumbar puncture, was imperative because infection can not only lead to AMS, but also precipitate episodes of DKA. Mr. O’s persistently abnormal vital signs indicated an underlying process may have been missed by focusing on treating DKA.
TREATMENT Finally, the diagnosis is established
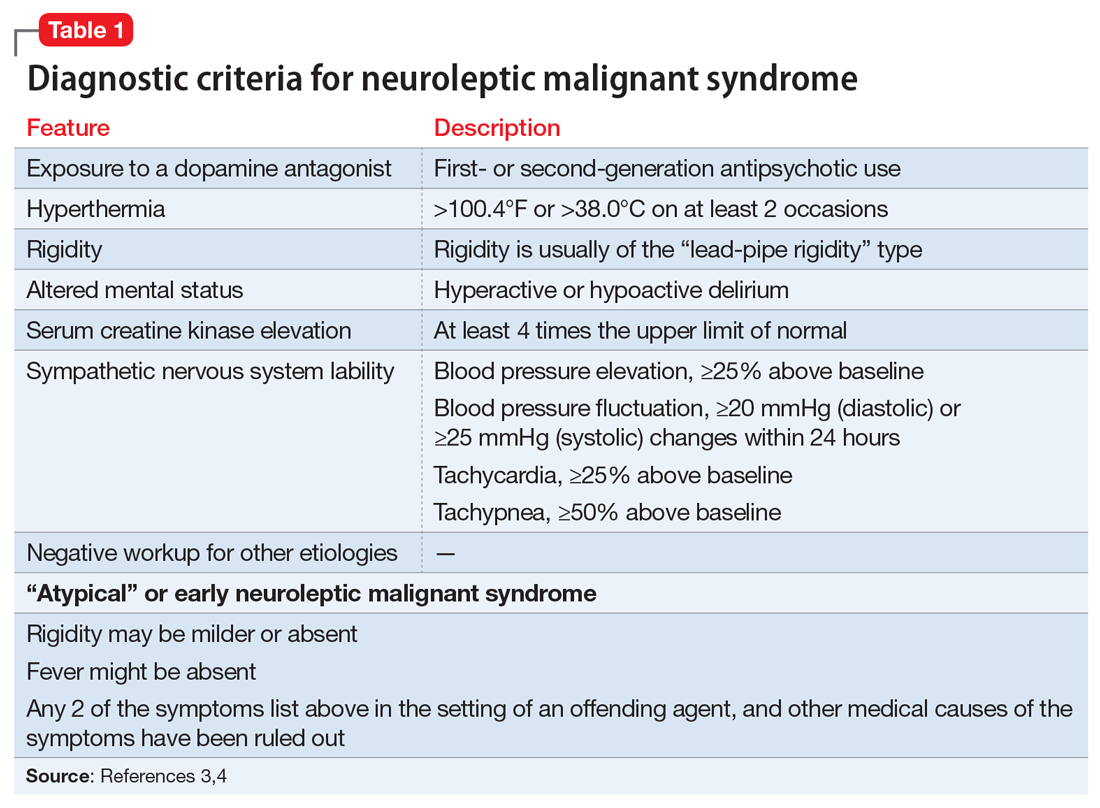
A lorazepam challenge is performed, and Mr. O receives 4 mg of lorazepam over 24 hours with little change in his catatonia symptoms. Given his persistent fever, tachycardia, and an elevated CK levels in the context of recent exposure to antipsychotic medications, Mr. O is diagnosed with NMS (Table 13,4 ) and is started on bromocriptine, 5 mg 3 times daily.
[polldaddy:10930632]
The authors’ observations
Mr. O’s complicated medical state—starting with DKA, halting the use of antipsychotic medications, and the suspicion of catatonia due to his history of schizophrenia—all distracted from the ultimate diagnosis of NMS as the cause of his enduring AMS and autonomic instability. Catatonia and NMS have overlapping symptomatology, including rigidity, autonomic instability, and stupor, which make the diagnosis of either condition complicated. A positive lorazepam test to diagnose catatonia is defined as a marked reduction in catatonia symptoms (typically a 50% reduction) as measured on a standardized rating scale.5 However, a negative lorazepam challenge does not definitely rule out catatonia because some cases are resistant to benzodiazepines.6
NMS risk factors relevant in this case include male sex, young age, acute medical illness, dehydration, and exposure to multiple psychotropic medications, including 2 antipsychotics, clozapine and fluphenazine.7 DKA is especially pertinent due to its acute onset and cause of significant dehydration. NMS can occur at any point of antipsychotic exposure, although the risk is highest during the initial weeks of treatment and during dosage changes. Unfortunately, Mr. O’s treatment team was unable to determine whether his medication had been recently changed, so it is not known what role this may have played in the development of NMS. Although first-generation antipsychotics are considered more likely to cause NMS, second-generation antipsychotics (SGAs) dominate the treatment of schizophrenia and bipolar disorder, and these medications also can cause NMS.8 As occurred in this case, long-acting injectable antipsychotics can be easily forgotten when not administered in the hospital, and their presence in the body persists for weeks. For example, the half-life of fluphenazine decanoate is approximately 10 days, and the half-life of haloperidol decanoate is 21 days.9
Continue to: OUTCOME Improvement with bromocriptine
OUTCOME Improvement with bromocriptine
After 4 days of bromocriptine, 5 mg 3 times daily, Mr. O is more alert, able to say “hello,” and can follow 1-step commands. By Day 26 of hospitalization, his CK levels decrease to 296 U/L, his CSF autoimmune panel is negative, and he is able to participate in physical therapy. After failing multiple swallow tests, Mr. O requires a percutaneous endoscopic gastrostomy (PEG) tube. He is discharged from the hospital to a long-term acute care facility with the plan to taper bromocriptine and restart a psychotropic regimen with his outpatient psychiatrist. At the time of discharge, he is able to sit at the edge of the bed independently, state his name, and respond to questions with multiple-word answers.
[polldaddy:10930633]
The authors’ observations
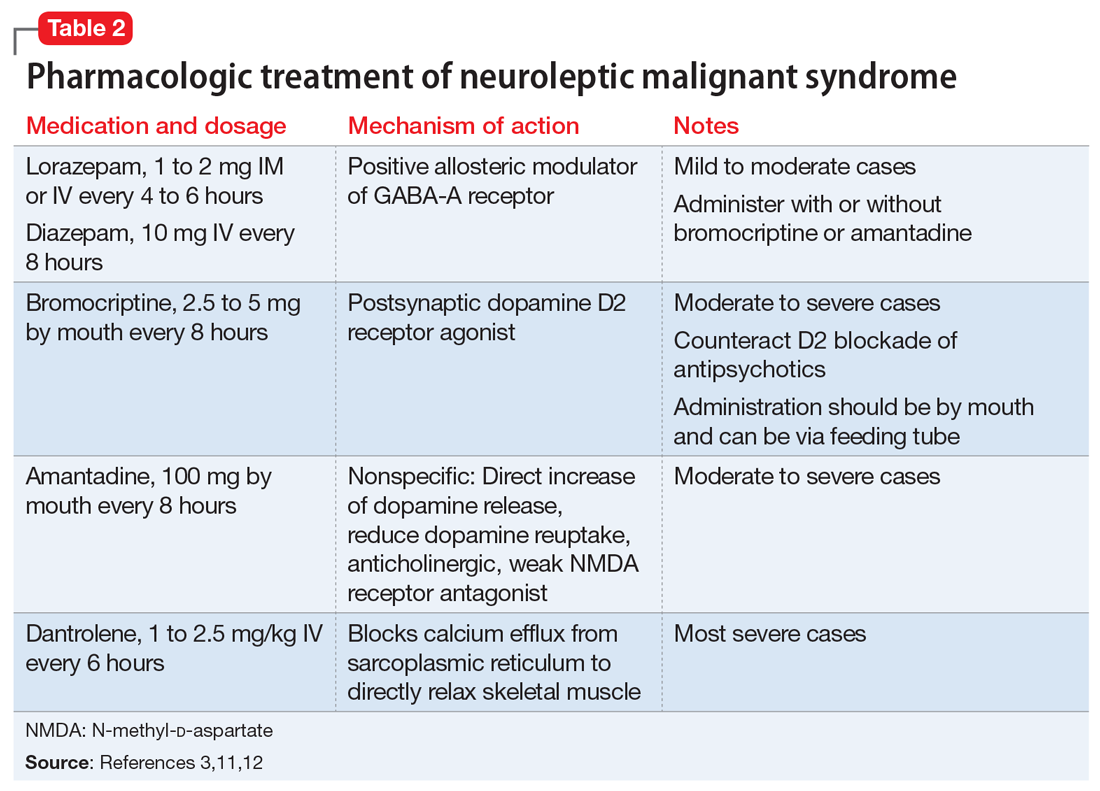
The most common pharmacologic treatments for NMS are dantrolene, bromocriptine, benzodiazepines (lorazepam or diazepam), and amantadine.3 Mild cases of NMS should be treated with discontinuation of all antipsychotics, supportive care, and benzodiazepines.3 Bromocriptine or amantadine are more appropriate for moderate cases and dantrolene for severe cases of NMS.3 All antipsychotics should be discontinued while a patient is experiencing an episode of NMS; however, once the NMS has resolved, clinicians must thoroughly evaluate the risks and benefits of restarting antipsychotic medication. After a patient has experienced an episode of NMS, clinicians generally should avoid prescribing the agent(s) that caused NMS and long-acting injections, and slowly titrate a low-potency SGA such as quetiapine.10Table 23,11,12 outlines the pharmacologic treatment of NMS.
Bottom Line
Neuroleptic malignant syndrome (NMS) should always be part of the differential diagnosis in patients with mental illness and altered mental status. The risk of NMS is especially high in patients with acute medical illness and exposure to antipsychotic medications.
Related Resource
- Turner AH, Kim JJ, McCarron RM. Differentiating serotonin syndrome and neuroleptic malignant syndrome. Current Psychiatry. 2019;18(2):30-36.
Drug Brand Names
Acyclovir • Zovirax
Amantadine • Gocovri
Ampicillin-sulbactam • Unasyn
Aripiprazole • Abilify Maintena
Benztropine • Cogentin
Bromocriptine • Cycloset, Parlodel
Ceftriaxone • Rocephin
Clozapine • Clozaril
Dantrolene • Dantrium
Diazepam • Valium
Haloperidol • Haldol
Lithium • Eskalith, Lithobid
Lorazepam • Ativan
Paliperidone palmitate • Invega Sustenna
Quetiapine • Seroquel
Risperidone • Risperdal
Valproate sodium • Depakote
Trazodone • Oleptro
Vancomycin • Vancocin
1. Zeitler P, Haqq A, Rosenbloom A, et al. Hyperglycemic hyperosmolar syndrome in children: pathophysiological considerations and suggested guidelines for treatment. J Pediatr. 2011;158(1):9-14.e1-2. doi: 10.1016/j.jpeds.2010.09.048
2. Francis A. Catatonia: diagnosis, classification, and treatment. Curr Psychiatry Rep. 2010;12(3):180-185. doi: 10.1007/s11920-010-0113-y
3. Pileggi DJ, Cook AM. Neuroleptic malignant syndrome. Ann Pharmacother. 2016;50(11):973-981. doi:10.1177/1060028016657553
4. Gurrera RJ, Caroff SN, Cohen A, et al. An international consensus study of neuroleptic malignant syndrome diagnostic criteria using the Delphi method. J Clin Psychiatry. 2011;72(9):1222-1228. doi:10.4088/JCP.10m06438
5. Sienaert P, Dhossche DM, Vancampfort D, et al. A clinical review of the treatment of catatonia. Front Psychiatry. 2014;5:181. doi:10.3389/fpsyt.2014.00181
6. Daniels J. Catatonia: clinical aspects and neurobiological correlates. J Neuropsychiatry Clin Neurosci. 2009;21(4):371-380. doi:10.1176/jnp.2009.21.4.371
7. Bhanushali MJ, Tuite PJ. The evaluation and management of patients with neuroleptic malignant syndrome. Neurol Clin. 2004;22(2):389-411. doi:10.1016/j.ncl.2003.12.006
8. Tse L, Barr AM, Scarapicchia V, et al. Neuroleptic malignant syndrome: a review from a clinically oriented perspective. Curr Neuropharmacol. 2015;13(3):395-406. doi:10.2174/1570159x13999150424113345
9. Correll CU, Kim E, Sliwa JK, et al. Pharmacokinetic characteristics of long-acting injectable antipsychotics for schizophrenia: an overview. CNS Drugs. 2021;35(1):39-59. doi:10.1007/s40263-020-00779-5
10. Strawn JR, Keck PE Jr, Caroff SN. Neuroleptic malignant syndrome. Am J Psychiatry. 2007;164(6):870-876. doi:10.1176/ajp.2007.164.6.870
11. Griffin CE 3rd, Kaye AM, Bueno FR, et al. Benzodiazepine pharmacology and central nervous system-mediated effects. Ochsner J. 2013;13(2):214-223.
12. Reulbach U, Dütsch C, Biermann T, et al. Managing an effective treatment for neuroleptic malignant syndrome. Crit Care. 2007;11(1):R4. doi:10.1186/cc5148
CASE Sluggish, weak, and incoherent
Mr. O, age 24, who has a history of schizophrenia and obesity, presents to the emergency department (ED) for altered mental status (AMS). His mother reports that he has been sluggish, weak, incoherent, had no appetite, and that on the day before admission, he was drinking excessive amounts of water and urinating every 10 minutes.
HISTORY Multiple ineffective antipsychotics
Mr. O was diagnosed with schizophrenia at age 21 and struggled with medication adherence, which resulted in multiple hospitalizations for stabilization. Trials of haloperidol, risperidone, paliperidone palmitate, and valproic acid had been ineffective. At the time of admission, his psychotropic medication regimen is fluphenazine decanoate, 25 mg injection every 2 weeks; clozapine, 50 mg/d; lithium carbonate, 300 mg twice a day; benztropine, 2 mg every night; and trazodone, 50 mg every night.
EVALUATION Fever, tachycardia, and diabetic ketoacidosis
Upon arrival to the ED, Mr. O is obtunded, unable to follow commands, and does not respond to painful stimuli. On physical exam, he has a fever of 38.4°C (reference range 35.1°C to 37.9°C); tachycardia with a heart rate of 142 beats per minute (bpm) (reference range 60 to 100); tachypnea with a respiratory rate of 35 breaths per minute (reference range 12 to 20); a blood pressure of 116/76 mmHg (reference range 90/60 to 130/80); and hypoxemia with an oxygen saturation of 90% on room air (reference range 94% to 100%).
Mr. O is admitted to the hospital and his laboratory workup indicates diabetic ketoacidosis (DKA), with a glucose of 1,700 mg/dL; anion gap of 30 (reference range 4 to 12 mmol/L); pH 7.04 (reference range 7.32 to 7.42); serum bicarbonate 6 (reference range 20 to 24 mEq/L); beta-hydroxybutyrate 11.04 (reference range 0 to 0.27 mmol/L); urine ketones, serum osmolality 407 (reference range 280 to 300 mOsm/kg); and an elevated white blood cell count of 18.4 (reference range 4.5 to 11.0 × 109/L). A CT scan of the head is negative for acute pathology.
Initially, all psychotropic medications are held. On Day 3 of hospitalization, psychiatry is consulted and clozapine, 50 mg/d; lithium, 300 mg/d; and benztropine, 1 mg at night, are restarted; however, fluphenazine decanoate and trazodone are held. The team recommends IV haloperidol, 2 mg as needed for agitation; however, it is never administered.
Imaging rules out deep vein thrombosis, cardiac dysfunction, and stroke, but a CT chest scan is notable for bilateral lung infiltrates, which suggests aspiration pneumonia.
Mr. O is diagnosed with diabetes, complicated by DKA, and is treated in the intensive care unit (ICU). Despite resolution of the DKA, he remains altered with fever and tachycardia.
Continue to: On Day 6 of hospitalization...
On Day 6 of hospitalization, Mr. O continues to be tachycardic and obtunded with nuchal rigidity. The team decides to transfer Mr. O to another hospital for a higher level of care and continued workup of his persistent AMS.
Immediately upon arrival at the second hospital, infectious disease and neurology teams are consulted for further evaluation. Mr. O’s AMS continues despite no clear signs of infection or other neurologic insults.
[polldaddy:10930631]
The authors’ observations
Based on Mr. O’s psychiatric history and laboratory results, the first medical team concluded his initial AMS was likely secondary to DKA; however, the AMS continued after the DKA resolved. At the second hospital, Mr. O’s treatment team continued to dig for answers.
EVALUATION Exploring the differential diagnosis
At the second hospital, Mr. O is admitted to the ICU with fever (37.8°C), tachycardia (120 bpm), tachypnea, withdrawal from painful stimuli, decreased reflexes, and muscle rigidity, including clenched jaw. The differential diagnoses include meningitis, sepsis from aspiration pneumonia, severe metabolic encephalopathy with prolonged recovery, central pontine myelinolysis, anoxic brain injury, and subclinical seizures.
Empiric vancomycin, 1.75 g every 12 hours; ceftriaxone, 2 g/d; and acyclovir, 900 mg every 8 hours are started for meningoencephalitis, and all psychotropic medications are discontinued. Case reports have documented a relationship between hyperglycemic hyperosmolar syndrome (HHS) and malignant hyperthermia in rare cases1; however, HHS is ruled out based on Mr. O’s laboratory results.A lumbar puncture and imaging rules out CNS infection. Antibiotic treatment is narrowed to ampicillin-sulbactam due to Mr. O’s prior CT chest showing concern for aspiration pneumonia. An MRI of the brain rules out central pontine myelinolysis, acute stroke, and anoxic brain injury, and an EEG shows nonspecific encephalopathy. On Day 10 of hospitalization, a neurologic exam shows flaccid paralysis and bilateral clonus, and Mr. O is mute. On Day 14 of hospitalization, his fever resolves, and his blood cultures are negative. On Day 15 of hospitalization, Mr. O’s creatine kinase (CK) level is elevated at 1,308 U/L (reference range 26 to 192 U/L), suggesting rhabdomyolysis.
Continue to: Given the neurologic exam findings...
Given the neurologic exam findings, and the limited evidence of infection, the differential diagnosis for Mr. O’s AMS is broadened to include catatonia, neuroleptic malignant syndrome (NMS), serotonin syndrome, and autoimmune encephalitis. The psychiatry team evaluates Mr. O for catatonia. He scores 14 on the Bush-Francis Catatonia Rating Scale, with findings of immobility/stupor, mutism, staring, autonomic instability, and withdrawal indicating the presence of catatonia.2
The authors’ observations
When Mr. O was transferred to the second hospital, the primary concern was to rule out meningitis due to his unstable vitals, obtunded mental state, and nuchal rigidity. A comprehensive infectious workup, including lumbar puncture, was imperative because infection can not only lead to AMS, but also precipitate episodes of DKA. Mr. O’s persistently abnormal vital signs indicated an underlying process may have been missed by focusing on treating DKA.
TREATMENT Finally, the diagnosis is established
A lorazepam challenge is performed, and Mr. O receives 4 mg of lorazepam over 24 hours with little change in his catatonia symptoms. Given his persistent fever, tachycardia, and an elevated CK levels in the context of recent exposure to antipsychotic medications, Mr. O is diagnosed with NMS (Table 13,4 ) and is started on bromocriptine, 5 mg 3 times daily.

[polldaddy:10930632]
The authors’ observations
Mr. O’s complicated medical state—starting with DKA, halting the use of antipsychotic medications, and the suspicion of catatonia due to his history of schizophrenia—all distracted from the ultimate diagnosis of NMS as the cause of his enduring AMS and autonomic instability. Catatonia and NMS have overlapping symptomatology, including rigidity, autonomic instability, and stupor, which make the diagnosis of either condition complicated. A positive lorazepam test to diagnose catatonia is defined as a marked reduction in catatonia symptoms (typically a 50% reduction) as measured on a standardized rating scale.5 However, a negative lorazepam challenge does not definitely rule out catatonia because some cases are resistant to benzodiazepines.6
NMS risk factors relevant in this case include male sex, young age, acute medical illness, dehydration, and exposure to multiple psychotropic medications, including 2 antipsychotics, clozapine and fluphenazine.7 DKA is especially pertinent due to its acute onset and cause of significant dehydration. NMS can occur at any point of antipsychotic exposure, although the risk is highest during the initial weeks of treatment and during dosage changes. Unfortunately, Mr. O’s treatment team was unable to determine whether his medication had been recently changed, so it is not known what role this may have played in the development of NMS. Although first-generation antipsychotics are considered more likely to cause NMS, second-generation antipsychotics (SGAs) dominate the treatment of schizophrenia and bipolar disorder, and these medications also can cause NMS.8 As occurred in this case, long-acting injectable antipsychotics can be easily forgotten when not administered in the hospital, and their presence in the body persists for weeks. For example, the half-life of fluphenazine decanoate is approximately 10 days, and the half-life of haloperidol decanoate is 21 days.9
Continue to: OUTCOME Improvement with bromocriptine
OUTCOME Improvement with bromocriptine
After 4 days of bromocriptine, 5 mg 3 times daily, Mr. O is more alert, able to say “hello,” and can follow 1-step commands. By Day 26 of hospitalization, his CK levels decrease to 296 U/L, his CSF autoimmune panel is negative, and he is able to participate in physical therapy. After failing multiple swallow tests, Mr. O requires a percutaneous endoscopic gastrostomy (PEG) tube. He is discharged from the hospital to a long-term acute care facility with the plan to taper bromocriptine and restart a psychotropic regimen with his outpatient psychiatrist. At the time of discharge, he is able to sit at the edge of the bed independently, state his name, and respond to questions with multiple-word answers.
[polldaddy:10930633]
The authors’ observations
The most common pharmacologic treatments for NMS are dantrolene, bromocriptine, benzodiazepines (lorazepam or diazepam), and amantadine.3 Mild cases of NMS should be treated with discontinuation of all antipsychotics, supportive care, and benzodiazepines.3 Bromocriptine or amantadine are more appropriate for moderate cases and dantrolene for severe cases of NMS.3 All antipsychotics should be discontinued while a patient is experiencing an episode of NMS; however, once the NMS has resolved, clinicians must thoroughly evaluate the risks and benefits of restarting antipsychotic medication. After a patient has experienced an episode of NMS, clinicians generally should avoid prescribing the agent(s) that caused NMS and long-acting injections, and slowly titrate a low-potency SGA such as quetiapine.10Table 23,11,12 outlines the pharmacologic treatment of NMS.

Bottom Line
Neuroleptic malignant syndrome (NMS) should always be part of the differential diagnosis in patients with mental illness and altered mental status. The risk of NMS is especially high in patients with acute medical illness and exposure to antipsychotic medications.
Related Resource
- Turner AH, Kim JJ, McCarron RM. Differentiating serotonin syndrome and neuroleptic malignant syndrome. Current Psychiatry. 2019;18(2):30-36.
Drug Brand Names
Acyclovir • Zovirax
Amantadine • Gocovri
Ampicillin-sulbactam • Unasyn
Aripiprazole • Abilify Maintena
Benztropine • Cogentin
Bromocriptine • Cycloset, Parlodel
Ceftriaxone • Rocephin
Clozapine • Clozaril
Dantrolene • Dantrium
Diazepam • Valium
Haloperidol • Haldol
Lithium • Eskalith, Lithobid
Lorazepam • Ativan
Paliperidone palmitate • Invega Sustenna
Quetiapine • Seroquel
Risperidone • Risperdal
Valproate sodium • Depakote
Trazodone • Oleptro
Vancomycin • Vancocin
CASE Sluggish, weak, and incoherent
Mr. O, age 24, who has a history of schizophrenia and obesity, presents to the emergency department (ED) for altered mental status (AMS). His mother reports that he has been sluggish, weak, incoherent, had no appetite, and that on the day before admission, he was drinking excessive amounts of water and urinating every 10 minutes.
HISTORY Multiple ineffective antipsychotics
Mr. O was diagnosed with schizophrenia at age 21 and struggled with medication adherence, which resulted in multiple hospitalizations for stabilization. Trials of haloperidol, risperidone, paliperidone palmitate, and valproic acid had been ineffective. At the time of admission, his psychotropic medication regimen is fluphenazine decanoate, 25 mg injection every 2 weeks; clozapine, 50 mg/d; lithium carbonate, 300 mg twice a day; benztropine, 2 mg every night; and trazodone, 50 mg every night.
EVALUATION Fever, tachycardia, and diabetic ketoacidosis
Upon arrival to the ED, Mr. O is obtunded, unable to follow commands, and does not respond to painful stimuli. On physical exam, he has a fever of 38.4°C (reference range 35.1°C to 37.9°C); tachycardia with a heart rate of 142 beats per minute (bpm) (reference range 60 to 100); tachypnea with a respiratory rate of 35 breaths per minute (reference range 12 to 20); a blood pressure of 116/76 mmHg (reference range 90/60 to 130/80); and hypoxemia with an oxygen saturation of 90% on room air (reference range 94% to 100%).
Mr. O is admitted to the hospital and his laboratory workup indicates diabetic ketoacidosis (DKA), with a glucose of 1,700 mg/dL; anion gap of 30 (reference range 4 to 12 mmol/L); pH 7.04 (reference range 7.32 to 7.42); serum bicarbonate 6 (reference range 20 to 24 mEq/L); beta-hydroxybutyrate 11.04 (reference range 0 to 0.27 mmol/L); urine ketones, serum osmolality 407 (reference range 280 to 300 mOsm/kg); and an elevated white blood cell count of 18.4 (reference range 4.5 to 11.0 × 109/L). A CT scan of the head is negative for acute pathology.
Initially, all psychotropic medications are held. On Day 3 of hospitalization, psychiatry is consulted and clozapine, 50 mg/d; lithium, 300 mg/d; and benztropine, 1 mg at night, are restarted; however, fluphenazine decanoate and trazodone are held. The team recommends IV haloperidol, 2 mg as needed for agitation; however, it is never administered.
Imaging rules out deep vein thrombosis, cardiac dysfunction, and stroke, but a CT chest scan is notable for bilateral lung infiltrates, which suggests aspiration pneumonia.
Mr. O is diagnosed with diabetes, complicated by DKA, and is treated in the intensive care unit (ICU). Despite resolution of the DKA, he remains altered with fever and tachycardia.
Continue to: On Day 6 of hospitalization...
On Day 6 of hospitalization, Mr. O continues to be tachycardic and obtunded with nuchal rigidity. The team decides to transfer Mr. O to another hospital for a higher level of care and continued workup of his persistent AMS.
Immediately upon arrival at the second hospital, infectious disease and neurology teams are consulted for further evaluation. Mr. O’s AMS continues despite no clear signs of infection or other neurologic insults.
[polldaddy:10930631]
The authors’ observations
Based on Mr. O’s psychiatric history and laboratory results, the first medical team concluded his initial AMS was likely secondary to DKA; however, the AMS continued after the DKA resolved. At the second hospital, Mr. O’s treatment team continued to dig for answers.
EVALUATION Exploring the differential diagnosis
At the second hospital, Mr. O is admitted to the ICU with fever (37.8°C), tachycardia (120 bpm), tachypnea, withdrawal from painful stimuli, decreased reflexes, and muscle rigidity, including clenched jaw. The differential diagnoses include meningitis, sepsis from aspiration pneumonia, severe metabolic encephalopathy with prolonged recovery, central pontine myelinolysis, anoxic brain injury, and subclinical seizures.
Empiric vancomycin, 1.75 g every 12 hours; ceftriaxone, 2 g/d; and acyclovir, 900 mg every 8 hours are started for meningoencephalitis, and all psychotropic medications are discontinued. Case reports have documented a relationship between hyperglycemic hyperosmolar syndrome (HHS) and malignant hyperthermia in rare cases1; however, HHS is ruled out based on Mr. O’s laboratory results.A lumbar puncture and imaging rules out CNS infection. Antibiotic treatment is narrowed to ampicillin-sulbactam due to Mr. O’s prior CT chest showing concern for aspiration pneumonia. An MRI of the brain rules out central pontine myelinolysis, acute stroke, and anoxic brain injury, and an EEG shows nonspecific encephalopathy. On Day 10 of hospitalization, a neurologic exam shows flaccid paralysis and bilateral clonus, and Mr. O is mute. On Day 14 of hospitalization, his fever resolves, and his blood cultures are negative. On Day 15 of hospitalization, Mr. O’s creatine kinase (CK) level is elevated at 1,308 U/L (reference range 26 to 192 U/L), suggesting rhabdomyolysis.
Continue to: Given the neurologic exam findings...
Given the neurologic exam findings, and the limited evidence of infection, the differential diagnosis for Mr. O’s AMS is broadened to include catatonia, neuroleptic malignant syndrome (NMS), serotonin syndrome, and autoimmune encephalitis. The psychiatry team evaluates Mr. O for catatonia. He scores 14 on the Bush-Francis Catatonia Rating Scale, with findings of immobility/stupor, mutism, staring, autonomic instability, and withdrawal indicating the presence of catatonia.2
The authors’ observations
When Mr. O was transferred to the second hospital, the primary concern was to rule out meningitis due to his unstable vitals, obtunded mental state, and nuchal rigidity. A comprehensive infectious workup, including lumbar puncture, was imperative because infection can not only lead to AMS, but also precipitate episodes of DKA. Mr. O’s persistently abnormal vital signs indicated an underlying process may have been missed by focusing on treating DKA.
TREATMENT Finally, the diagnosis is established
A lorazepam challenge is performed, and Mr. O receives 4 mg of lorazepam over 24 hours with little change in his catatonia symptoms. Given his persistent fever, tachycardia, and an elevated CK levels in the context of recent exposure to antipsychotic medications, Mr. O is diagnosed with NMS (Table 13,4 ) and is started on bromocriptine, 5 mg 3 times daily.

[polldaddy:10930632]
The authors’ observations
Mr. O’s complicated medical state—starting with DKA, halting the use of antipsychotic medications, and the suspicion of catatonia due to his history of schizophrenia—all distracted from the ultimate diagnosis of NMS as the cause of his enduring AMS and autonomic instability. Catatonia and NMS have overlapping symptomatology, including rigidity, autonomic instability, and stupor, which make the diagnosis of either condition complicated. A positive lorazepam test to diagnose catatonia is defined as a marked reduction in catatonia symptoms (typically a 50% reduction) as measured on a standardized rating scale.5 However, a negative lorazepam challenge does not definitely rule out catatonia because some cases are resistant to benzodiazepines.6
NMS risk factors relevant in this case include male sex, young age, acute medical illness, dehydration, and exposure to multiple psychotropic medications, including 2 antipsychotics, clozapine and fluphenazine.7 DKA is especially pertinent due to its acute onset and cause of significant dehydration. NMS can occur at any point of antipsychotic exposure, although the risk is highest during the initial weeks of treatment and during dosage changes. Unfortunately, Mr. O’s treatment team was unable to determine whether his medication had been recently changed, so it is not known what role this may have played in the development of NMS. Although first-generation antipsychotics are considered more likely to cause NMS, second-generation antipsychotics (SGAs) dominate the treatment of schizophrenia and bipolar disorder, and these medications also can cause NMS.8 As occurred in this case, long-acting injectable antipsychotics can be easily forgotten when not administered in the hospital, and their presence in the body persists for weeks. For example, the half-life of fluphenazine decanoate is approximately 10 days, and the half-life of haloperidol decanoate is 21 days.9
Continue to: OUTCOME Improvement with bromocriptine
OUTCOME Improvement with bromocriptine
After 4 days of bromocriptine, 5 mg 3 times daily, Mr. O is more alert, able to say “hello,” and can follow 1-step commands. By Day 26 of hospitalization, his CK levels decrease to 296 U/L, his CSF autoimmune panel is negative, and he is able to participate in physical therapy. After failing multiple swallow tests, Mr. O requires a percutaneous endoscopic gastrostomy (PEG) tube. He is discharged from the hospital to a long-term acute care facility with the plan to taper bromocriptine and restart a psychotropic regimen with his outpatient psychiatrist. At the time of discharge, he is able to sit at the edge of the bed independently, state his name, and respond to questions with multiple-word answers.
[polldaddy:10930633]
The authors’ observations
The most common pharmacologic treatments for NMS are dantrolene, bromocriptine, benzodiazepines (lorazepam or diazepam), and amantadine.3 Mild cases of NMS should be treated with discontinuation of all antipsychotics, supportive care, and benzodiazepines.3 Bromocriptine or amantadine are more appropriate for moderate cases and dantrolene for severe cases of NMS.3 All antipsychotics should be discontinued while a patient is experiencing an episode of NMS; however, once the NMS has resolved, clinicians must thoroughly evaluate the risks and benefits of restarting antipsychotic medication. After a patient has experienced an episode of NMS, clinicians generally should avoid prescribing the agent(s) that caused NMS and long-acting injections, and slowly titrate a low-potency SGA such as quetiapine.10Table 23,11,12 outlines the pharmacologic treatment of NMS.

Bottom Line
Neuroleptic malignant syndrome (NMS) should always be part of the differential diagnosis in patients with mental illness and altered mental status. The risk of NMS is especially high in patients with acute medical illness and exposure to antipsychotic medications.
Related Resource
- Turner AH, Kim JJ, McCarron RM. Differentiating serotonin syndrome and neuroleptic malignant syndrome. Current Psychiatry. 2019;18(2):30-36.
Drug Brand Names
Acyclovir • Zovirax
Amantadine • Gocovri
Ampicillin-sulbactam • Unasyn
Aripiprazole • Abilify Maintena
Benztropine • Cogentin
Bromocriptine • Cycloset, Parlodel
Ceftriaxone • Rocephin
Clozapine • Clozaril
Dantrolene • Dantrium
Diazepam • Valium
Haloperidol • Haldol
Lithium • Eskalith, Lithobid
Lorazepam • Ativan
Paliperidone palmitate • Invega Sustenna
Quetiapine • Seroquel
Risperidone • Risperdal
Valproate sodium • Depakote
Trazodone • Oleptro
Vancomycin • Vancocin
1. Zeitler P, Haqq A, Rosenbloom A, et al. Hyperglycemic hyperosmolar syndrome in children: pathophysiological considerations and suggested guidelines for treatment. J Pediatr. 2011;158(1):9-14.e1-2. doi: 10.1016/j.jpeds.2010.09.048
2. Francis A. Catatonia: diagnosis, classification, and treatment. Curr Psychiatry Rep. 2010;12(3):180-185. doi: 10.1007/s11920-010-0113-y
3. Pileggi DJ, Cook AM. Neuroleptic malignant syndrome. Ann Pharmacother. 2016;50(11):973-981. doi:10.1177/1060028016657553
4. Gurrera RJ, Caroff SN, Cohen A, et al. An international consensus study of neuroleptic malignant syndrome diagnostic criteria using the Delphi method. J Clin Psychiatry. 2011;72(9):1222-1228. doi:10.4088/JCP.10m06438
5. Sienaert P, Dhossche DM, Vancampfort D, et al. A clinical review of the treatment of catatonia. Front Psychiatry. 2014;5:181. doi:10.3389/fpsyt.2014.00181
6. Daniels J. Catatonia: clinical aspects and neurobiological correlates. J Neuropsychiatry Clin Neurosci. 2009;21(4):371-380. doi:10.1176/jnp.2009.21.4.371
7. Bhanushali MJ, Tuite PJ. The evaluation and management of patients with neuroleptic malignant syndrome. Neurol Clin. 2004;22(2):389-411. doi:10.1016/j.ncl.2003.12.006
8. Tse L, Barr AM, Scarapicchia V, et al. Neuroleptic malignant syndrome: a review from a clinically oriented perspective. Curr Neuropharmacol. 2015;13(3):395-406. doi:10.2174/1570159x13999150424113345
9. Correll CU, Kim E, Sliwa JK, et al. Pharmacokinetic characteristics of long-acting injectable antipsychotics for schizophrenia: an overview. CNS Drugs. 2021;35(1):39-59. doi:10.1007/s40263-020-00779-5
10. Strawn JR, Keck PE Jr, Caroff SN. Neuroleptic malignant syndrome. Am J Psychiatry. 2007;164(6):870-876. doi:10.1176/ajp.2007.164.6.870
11. Griffin CE 3rd, Kaye AM, Bueno FR, et al. Benzodiazepine pharmacology and central nervous system-mediated effects. Ochsner J. 2013;13(2):214-223.
12. Reulbach U, Dütsch C, Biermann T, et al. Managing an effective treatment for neuroleptic malignant syndrome. Crit Care. 2007;11(1):R4. doi:10.1186/cc5148
1. Zeitler P, Haqq A, Rosenbloom A, et al. Hyperglycemic hyperosmolar syndrome in children: pathophysiological considerations and suggested guidelines for treatment. J Pediatr. 2011;158(1):9-14.e1-2. doi: 10.1016/j.jpeds.2010.09.048
2. Francis A. Catatonia: diagnosis, classification, and treatment. Curr Psychiatry Rep. 2010;12(3):180-185. doi: 10.1007/s11920-010-0113-y
3. Pileggi DJ, Cook AM. Neuroleptic malignant syndrome. Ann Pharmacother. 2016;50(11):973-981. doi:10.1177/1060028016657553
4. Gurrera RJ, Caroff SN, Cohen A, et al. An international consensus study of neuroleptic malignant syndrome diagnostic criteria using the Delphi method. J Clin Psychiatry. 2011;72(9):1222-1228. doi:10.4088/JCP.10m06438
5. Sienaert P, Dhossche DM, Vancampfort D, et al. A clinical review of the treatment of catatonia. Front Psychiatry. 2014;5:181. doi:10.3389/fpsyt.2014.00181
6. Daniels J. Catatonia: clinical aspects and neurobiological correlates. J Neuropsychiatry Clin Neurosci. 2009;21(4):371-380. doi:10.1176/jnp.2009.21.4.371
7. Bhanushali MJ, Tuite PJ. The evaluation and management of patients with neuroleptic malignant syndrome. Neurol Clin. 2004;22(2):389-411. doi:10.1016/j.ncl.2003.12.006
8. Tse L, Barr AM, Scarapicchia V, et al. Neuroleptic malignant syndrome: a review from a clinically oriented perspective. Curr Neuropharmacol. 2015;13(3):395-406. doi:10.2174/1570159x13999150424113345
9. Correll CU, Kim E, Sliwa JK, et al. Pharmacokinetic characteristics of long-acting injectable antipsychotics for schizophrenia: an overview. CNS Drugs. 2021;35(1):39-59. doi:10.1007/s40263-020-00779-5
10. Strawn JR, Keck PE Jr, Caroff SN. Neuroleptic malignant syndrome. Am J Psychiatry. 2007;164(6):870-876. doi:10.1176/ajp.2007.164.6.870
11. Griffin CE 3rd, Kaye AM, Bueno FR, et al. Benzodiazepine pharmacology and central nervous system-mediated effects. Ochsner J. 2013;13(2):214-223.
12. Reulbach U, Dütsch C, Biermann T, et al. Managing an effective treatment for neuroleptic malignant syndrome. Crit Care. 2007;11(1):R4. doi:10.1186/cc5148
Intimate partner violence: Assessment in the era of telehealth
Intimate partner violence (IPV) includes “physical violence, sexual violence, stalking, and psychological aggression (including coercive tactics) by a current or former intimate partner.”1
Ensure a safe environment
At the onset of a telehealth appointment, ask the patient “Who is in the room with you?” If an adult or child age >2 years is present, do not assess for IPV because it may be unsafe for the patient to answer such questions. Encourage the patient to use privacy-enhancing strategies (eg, wearing headphones, going outside, calling from a vehicle). Be flexible; someone may not be able to discuss IPV during an appointment but might be able to at a different time, such as when their partner goes to work. For patients who disclose IPV, identify a word, phrase, or gesture to quickly communicate their partner’s presence or need for immediate help.2 While the “Signal for Help” (ie, thumb first tucked into the palm, then covered with fingers to form a fist) has been developed,3 it is not universally familiar; until then, establish specific communications and preferences with each patient. Include a plan for the patient to abruptly disconnect (eg, “You have the wrong number”) with a pre-determined method of follow-up.
Obtain informed consent
Before asking a patient about IPV, provide psychoeducation about the purpose, including its relationship to one’s health. Acknowledge reasons it may not be safe to provide and/or document answers, and describe limits of confidentiality and local mandated reporting requirements.
Standardize the assessment
Intimate partner violence assessment should be normalized (eg, “Because violence is common, I ask everyone about their relationships”), direct, and well-integrated. Know whether your site uses a specific IPV screening tool, such as the Relationship Health and Safety Screen (RHSS), which is used at the VA; if so, learn and practice asking the specific questions aloud until it feels routine and you can maintain eye contact throughout. Examples of other IPV assessment instruments include the Abuse Assessment Screen (AAS); Hurt, Insult, Threaten, and Scream (HITS), Partner Violence Screen (PVS), and Women Abuse Screening Tool (WAST).4 Pay attention to the populations in which a tool has been studied, any associated copyright fees, and gender-neutral and non-heteronormative language. Avoid asking leading questions (eg, “You’re not being hurt, are you?”) or using charged/interpretable terms (eg, “Is someone abusing you?”).
Document with intention
Use person-centered, recovery-oriented language (eg, someone who experiences or uses IPV) rather than stigmatizing language (eg, victim, batterer, abuser). Describe what happened using the individual’s own words and clearly identify the source of information, witnesses, and any weapons used. Choose nonpejorative language (ie, “states” instead of “claims”). Do not document details of the safety plan in the chart because doing so can compromise safety.
Provide resources and referrals
Regardless of whether a patient consents to screening/documentation or discloses IPV, you should offer universal education, resources, and referrals. Review national contacts (National Domestic Violence Hotline: 1-800-799-7233), community agencies (available through www.domesticshelters.org), and suggested safety apps such as myPlan (www.myplanapp.org), but do not send a patient electronic or physical materials without first confirming it is safe to do so. Assess the patient’s interest in legal steps (eg, obtaining a protection order, pressing charges) while recognizing and respecting valid concerns about law enforcement involvement, particularly among the Black community and Black transgender women. Provide options instead of instructions, which will empower patients to choose what is best for their situation, and support their decisions.
1. Breiding MJ, Chen J, Black MC. Intimate partner violence in the United States – 2010. National Center for Injury Prevention and Control, Centers for Disease Control and Prevention. Published February 2014. Accessed January 12, 2021. https://www.cdc.gov/violenceprevention/pdf/cdc_nisvs_ipv_report_2013_v17_single_a.pdf
2. Evans ML, Lindauer JD, Farrell ME. A pandemic within a pandemic – intimate partner violence during Covid-19. N Engl J Med. 2020;383(24):2302-2304. doi:10.1056/NEJMp2024046
3. Canadian Women’s Foundation. Signal for help. 2020. Accessed January 12, 2021. https://canadianwomen.org/signal-for-help/
4. Basile KC, Hertz MF, Back SE. Intimate partner violence and sexual violence victimization assessment instruments for use in healthcare settings: Version 1. Centers for Disease Control and Prevention, National Center for Injury Prevention and Control. 2007. Accessed January 12, 2021. https://www.cdc.gov/violenceprevention/pdf/ipv/ipvandsvscreening.pdf
Intimate partner violence (IPV) includes “physical violence, sexual violence, stalking, and psychological aggression (including coercive tactics) by a current or former intimate partner.”1
Ensure a safe environment
At the onset of a telehealth appointment, ask the patient “Who is in the room with you?” If an adult or child age >2 years is present, do not assess for IPV because it may be unsafe for the patient to answer such questions. Encourage the patient to use privacy-enhancing strategies (eg, wearing headphones, going outside, calling from a vehicle). Be flexible; someone may not be able to discuss IPV during an appointment but might be able to at a different time, such as when their partner goes to work. For patients who disclose IPV, identify a word, phrase, or gesture to quickly communicate their partner’s presence or need for immediate help.2 While the “Signal for Help” (ie, thumb first tucked into the palm, then covered with fingers to form a fist) has been developed,3 it is not universally familiar; until then, establish specific communications and preferences with each patient. Include a plan for the patient to abruptly disconnect (eg, “You have the wrong number”) with a pre-determined method of follow-up.
Obtain informed consent
Before asking a patient about IPV, provide psychoeducation about the purpose, including its relationship to one’s health. Acknowledge reasons it may not be safe to provide and/or document answers, and describe limits of confidentiality and local mandated reporting requirements.
Standardize the assessment
Intimate partner violence assessment should be normalized (eg, “Because violence is common, I ask everyone about their relationships”), direct, and well-integrated. Know whether your site uses a specific IPV screening tool, such as the Relationship Health and Safety Screen (RHSS), which is used at the VA; if so, learn and practice asking the specific questions aloud until it feels routine and you can maintain eye contact throughout. Examples of other IPV assessment instruments include the Abuse Assessment Screen (AAS); Hurt, Insult, Threaten, and Scream (HITS), Partner Violence Screen (PVS), and Women Abuse Screening Tool (WAST).4 Pay attention to the populations in which a tool has been studied, any associated copyright fees, and gender-neutral and non-heteronormative language. Avoid asking leading questions (eg, “You’re not being hurt, are you?”) or using charged/interpretable terms (eg, “Is someone abusing you?”).
Document with intention
Use person-centered, recovery-oriented language (eg, someone who experiences or uses IPV) rather than stigmatizing language (eg, victim, batterer, abuser). Describe what happened using the individual’s own words and clearly identify the source of information, witnesses, and any weapons used. Choose nonpejorative language (ie, “states” instead of “claims”). Do not document details of the safety plan in the chart because doing so can compromise safety.
Provide resources and referrals
Regardless of whether a patient consents to screening/documentation or discloses IPV, you should offer universal education, resources, and referrals. Review national contacts (National Domestic Violence Hotline: 1-800-799-7233), community agencies (available through www.domesticshelters.org), and suggested safety apps such as myPlan (www.myplanapp.org), but do not send a patient electronic or physical materials without first confirming it is safe to do so. Assess the patient’s interest in legal steps (eg, obtaining a protection order, pressing charges) while recognizing and respecting valid concerns about law enforcement involvement, particularly among the Black community and Black transgender women. Provide options instead of instructions, which will empower patients to choose what is best for their situation, and support their decisions.
Intimate partner violence (IPV) includes “physical violence, sexual violence, stalking, and psychological aggression (including coercive tactics) by a current or former intimate partner.”1
Ensure a safe environment
At the onset of a telehealth appointment, ask the patient “Who is in the room with you?” If an adult or child age >2 years is present, do not assess for IPV because it may be unsafe for the patient to answer such questions. Encourage the patient to use privacy-enhancing strategies (eg, wearing headphones, going outside, calling from a vehicle). Be flexible; someone may not be able to discuss IPV during an appointment but might be able to at a different time, such as when their partner goes to work. For patients who disclose IPV, identify a word, phrase, or gesture to quickly communicate their partner’s presence or need for immediate help.2 While the “Signal for Help” (ie, thumb first tucked into the palm, then covered with fingers to form a fist) has been developed,3 it is not universally familiar; until then, establish specific communications and preferences with each patient. Include a plan for the patient to abruptly disconnect (eg, “You have the wrong number”) with a pre-determined method of follow-up.
Obtain informed consent
Before asking a patient about IPV, provide psychoeducation about the purpose, including its relationship to one’s health. Acknowledge reasons it may not be safe to provide and/or document answers, and describe limits of confidentiality and local mandated reporting requirements.
Standardize the assessment
Intimate partner violence assessment should be normalized (eg, “Because violence is common, I ask everyone about their relationships”), direct, and well-integrated. Know whether your site uses a specific IPV screening tool, such as the Relationship Health and Safety Screen (RHSS), which is used at the VA; if so, learn and practice asking the specific questions aloud until it feels routine and you can maintain eye contact throughout. Examples of other IPV assessment instruments include the Abuse Assessment Screen (AAS); Hurt, Insult, Threaten, and Scream (HITS), Partner Violence Screen (PVS), and Women Abuse Screening Tool (WAST).4 Pay attention to the populations in which a tool has been studied, any associated copyright fees, and gender-neutral and non-heteronormative language. Avoid asking leading questions (eg, “You’re not being hurt, are you?”) or using charged/interpretable terms (eg, “Is someone abusing you?”).
Document with intention
Use person-centered, recovery-oriented language (eg, someone who experiences or uses IPV) rather than stigmatizing language (eg, victim, batterer, abuser). Describe what happened using the individual’s own words and clearly identify the source of information, witnesses, and any weapons used. Choose nonpejorative language (ie, “states” instead of “claims”). Do not document details of the safety plan in the chart because doing so can compromise safety.
Provide resources and referrals
Regardless of whether a patient consents to screening/documentation or discloses IPV, you should offer universal education, resources, and referrals. Review national contacts (National Domestic Violence Hotline: 1-800-799-7233), community agencies (available through www.domesticshelters.org), and suggested safety apps such as myPlan (www.myplanapp.org), but do not send a patient electronic or physical materials without first confirming it is safe to do so. Assess the patient’s interest in legal steps (eg, obtaining a protection order, pressing charges) while recognizing and respecting valid concerns about law enforcement involvement, particularly among the Black community and Black transgender women. Provide options instead of instructions, which will empower patients to choose what is best for their situation, and support their decisions.
1. Breiding MJ, Chen J, Black MC. Intimate partner violence in the United States – 2010. National Center for Injury Prevention and Control, Centers for Disease Control and Prevention. Published February 2014. Accessed January 12, 2021. https://www.cdc.gov/violenceprevention/pdf/cdc_nisvs_ipv_report_2013_v17_single_a.pdf
2. Evans ML, Lindauer JD, Farrell ME. A pandemic within a pandemic – intimate partner violence during Covid-19. N Engl J Med. 2020;383(24):2302-2304. doi:10.1056/NEJMp2024046
3. Canadian Women’s Foundation. Signal for help. 2020. Accessed January 12, 2021. https://canadianwomen.org/signal-for-help/
4. Basile KC, Hertz MF, Back SE. Intimate partner violence and sexual violence victimization assessment instruments for use in healthcare settings: Version 1. Centers for Disease Control and Prevention, National Center for Injury Prevention and Control. 2007. Accessed January 12, 2021. https://www.cdc.gov/violenceprevention/pdf/ipv/ipvandsvscreening.pdf
1. Breiding MJ, Chen J, Black MC. Intimate partner violence in the United States – 2010. National Center for Injury Prevention and Control, Centers for Disease Control and Prevention. Published February 2014. Accessed January 12, 2021. https://www.cdc.gov/violenceprevention/pdf/cdc_nisvs_ipv_report_2013_v17_single_a.pdf
2. Evans ML, Lindauer JD, Farrell ME. A pandemic within a pandemic – intimate partner violence during Covid-19. N Engl J Med. 2020;383(24):2302-2304. doi:10.1056/NEJMp2024046
3. Canadian Women’s Foundation. Signal for help. 2020. Accessed January 12, 2021. https://canadianwomen.org/signal-for-help/
4. Basile KC, Hertz MF, Back SE. Intimate partner violence and sexual violence victimization assessment instruments for use in healthcare settings: Version 1. Centers for Disease Control and Prevention, National Center for Injury Prevention and Control. 2007. Accessed January 12, 2021. https://www.cdc.gov/violenceprevention/pdf/ipv/ipvandsvscreening.pdf
Treating major depressive disorder after limited response to an initial agent
Selective serotonin reuptake inhibitors (SSRIs) and serotonin-norepinephrine reuptake inhibitors (SNRIs) are commonly used first-line agents for treating major depressive disorder. Less than one-half of patients with major depressive disorder experience remission after 1 acute trial of an antidepressant.1 After optimization of an initial agent’s dose and duration, potential next steps include switching agents or augmentation. Augmentation strategies may lead to clinical improvement but carry the risks of polypharmacy, including increased risk of adverse effects and drug interactions. Clinicians can consider the following evidence-based options for a patient with a limited response to an initial SSRI or SNRI.
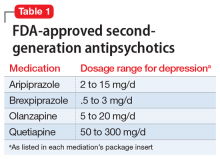
Second-generation antipsychotics, when used as augmentation agents to treat a patient with major depressive disorder, can lead to an approximately 10% improvement in remission rate compared with placebo.2 Aripiprazole, brexpiprazole, olanzapine (in combination with fluoxetine only), and quetiapine are FDA-approved as adjunctive therapies with an antidepressant (Table 1). Second-generation antipsychotics should be started at lower doses than those used for schizophrenia, and these agents have an increased risk of metabolic adverse effects as well as extrapyramidal symptoms.
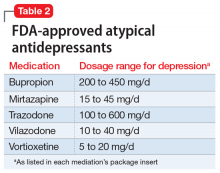
Atypical antidepressants are those that are not classified as an SSRI, SNRI, tricyclic antidepressant (TCA), or monoamine oxidase inhibitor (MAOI). These include bupropion, mirtazapine, trazodone, vilazodone, and vortioxetine (Table 2). Bupropion is a dopamine and norepinephrine reuptake inhibitor. When used for augmentation in clinical studies, it led to a 30% remission rate.3 Mirtazapine is an alpha-2 antagonist that can be used as monotherapy or in combination with another antidepressant.4 Trazodone is an antidepressant with activity at histamine and alpha-1-adrenergic receptors that is often used off-label for insomnia. Trazodone can be used safely and effectively in combination with other agents for treatment-resistant depression.5 Vilazodone is a 5-HT1A partial agonist, and vortioxetine is a 5-HT1A agonist and 5-HT3 antagonist; both are FDA-approved as alternative agents for monotherapy for major depressive disorder. Choosing among these agents for switching or augmenting can be guided by patient preference, adverse effect profile, and targeting specific symptoms, such as using mirtazapine to address poor sleep and appetite.
Lithium augmentation has been frequently investigated in placebo-controlled, double-blind studies. A meta-analysis showed that patients receiving lithium augmentation with a serum level of ≥0.5 mEq/L were >3 times more likely to respond than those receiving placebo.6 When lithium is used to treat bipolar disorder, the therapeutic serum range for lithium is 0.8 to 1.2 mEq/L, with an increased risk of adverse effects (including toxicity) at higher levels.7
Triiodothyronine (T3) augmentation of antidepressants led to remission in approximately 1 in 4 patients who had not achieved remission or who were intolerant to an initial treatment with citalopram and a second switch or augmentation trial.8 In this study, the mean dose of T3 was 45.2 µg/d, with an average length of treatment of 9 weeks.
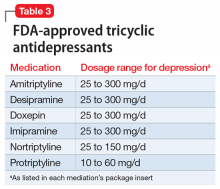
Tricyclic antidepressants are another option when considering switching agents (Table 3). TCAs are additionally effective for comorbid pain conditions.9 When TCAs are used in combination with SSRIs, drug interactions may occur that increase TCA plasma levels. There is also an increased risk of serotonin syndrome when used with serotonergic agents, though an SSRI/ TCA combination may be appropriate for a patient with treatment-resistant depression.10 Additionally, TCAs carry unique risks of cardiovascular effects, including cardiac arrhythmias. A meta-analysis comparing fluoxetine, paroxetine, and sertraline to TCAs (amitriptyline, clomipramine, desipramine, doxepin, imipramine, and nortriptyline) concluded that both classes had similar efficacy in treating depression, though the drop-out rate was significantly higher among patients receiving TCAs.11
Buspirone is approved for generalized anxiety disorder. In studies where buspirone was used as an augmentation agent for major depressive disorder at a mean daily dose of 40.9 mg divided into 2 doses, it led to a remission rate >30%.3
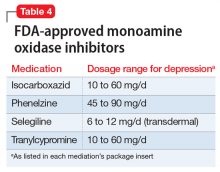
Continue to: Monoamine oxidase inhibitors
Monoamine oxidase inhibitors should typically be avoided in initial or early treatment of depression due to tolerability issues, drug interactions, and dietary restrictions to avoid hypertensive crisis. MAOIs are generally not recommended to be used with SSRIs, SNRIs, or TCAs, and typically require a “washout” period from other antidepressants (Table 4). One review found that MAOI treatment had advantage over TCA treatment for patients with early-stage treatment-resistant depression, though this advantage decreased as the number of failed antidepressant trials increased.12 One MAOI, selegiline, is available in a transdermal patch, and the 6-mg patch does not require dietary restriction.
Esketamine (intranasal) is FDA-approved for treatment-resistant depression (failure of response after at least 2 antidepressant trials with adequate dose and duration) in conjunction with an oral antidepressant. In clinical studies, a significant response was noted after 1 week of treatment.13 Esketamine requires an induction period of twice-weekly doses of 56 or 84 mg, with maintenance doses every 1 to 2 weeks. Each dosage administration requires monitoring for at least 2 hours by a health care professional at a certified treatment center. Esketamine’s indication was recently expanded to include treatment of patients with major depressive disorder with suicidal ideation or behavior.
Stimulants such as amphetamines, methylphenidate, or modafinil have been effective in open studies for augmentation in depression.14 However, no stimulant is FDA-approved for the treatment of depression. In addition to other adverse effects, these medications are controlled substances and carry risk of misuse, and their use may not be appropriate for all patients.
1. Trivedi MH, Rush AJ, Wisniewski SR, et al. Evaluation of outcomes with citalopram for depression using measurement-based care in STAR*D: implications for clinical practice. Am J Psychiatry. 2006;163(1):28-40.
2. Kato M, Chang CM. Augmentation treatments with second-generation antipsychotics to antidepressants in treatment-resistant depression. CNS Drugs. 2013;27 Suppl 1:S11-S19.
3. Trivedi MH, Fava M, Wisniewski SR, et al. Medication augmentation after the failure of SSRIs for depression. N Engl J Med. 2006;354(12):1243-1252.
4. Carpenter LL, Jocic Z, Hall JM, et al. Mirtazapine augmentation in the treatment of refractory depression. J Clin Psychiatry. 1999;60(1):45-49.
5. Maes M, Vandoolaeghe E, Desnyder R. Efficacy of treatment with trazodone in combination with pindolol or fluoxetine in major depression. J Affect Disord. 1996;41(3):201-210.
6. Bauer M, Dopfmer S. Lithium augmentation in treatment-resistant depression: meta-analysis of placebo-controlled studies. J Clin Psychopharmacol. 1999;19(5):427-434.
7. Yatham LN, Kennedy SH, Parikh SV, et al. Canadian Network for Mood and Anxiety Treatments (CANMAT) and International Society for Bipolar Disorders (ISBD) 2018 guidelines for the management of patients with bipolar disorder. Bipolar Disord. 2018;20(2):97-170.
8. Nierenberg AA, Fava M, Trivedi MH, et al. A comparison of lithium and T(3) augmentation following two failed medication treatments for depression: a STAR*D report. Am J Psychiatry. 2006;163(9):1519-1530; quiz 1665.
9. Saarto T, Wiffen PJ. Antidepressants for neuropathic pain. Cochrane Database Syst Rev. 2007;17(4):CD005454.
10. Taylor D. Selective serotonin reuptake inhibitors and tricyclic antidepressants in combination. Interactions and therapeutic uses. Br J Psychiatry. 1995;167(5):575-580.
11. Steffens DC, Krishnan KR, Helms MJ. Are SSRIs better than TCAs? Comparison of SSRIs and TCAs: a meta-analysis. Depress Anxiety. 1997;6(1):10-18.
12. Kim T, Xu C, Amsterdam JD. Relative effectiveness of tricyclic antidepressant versus monoamine oxidase inhibitor monotherapy for treatment-resistant depression. J Affect Disord. 2019;250:199-203.
13. Daly EJ, Singh JB, Fedgchin M, et al. Efficacy and safety of intranasal esketamine adjunctive to oral antidepressant therapy in treatment-resistant depression: a randomized clinical trial. JAMA Psychiatry. 2018;75(2):139-148.
14. DeBattista C. Augmentation and combination strategies for depression. J Psychopharmacol. 2006;20(3 Suppl):11-18.
Selective serotonin reuptake inhibitors (SSRIs) and serotonin-norepinephrine reuptake inhibitors (SNRIs) are commonly used first-line agents for treating major depressive disorder. Less than one-half of patients with major depressive disorder experience remission after 1 acute trial of an antidepressant.1 After optimization of an initial agent’s dose and duration, potential next steps include switching agents or augmentation. Augmentation strategies may lead to clinical improvement but carry the risks of polypharmacy, including increased risk of adverse effects and drug interactions. Clinicians can consider the following evidence-based options for a patient with a limited response to an initial SSRI or SNRI.
Second-generation antipsychotics, when used as augmentation agents to treat a patient with major depressive disorder, can lead to an approximately 10% improvement in remission rate compared with placebo.2 Aripiprazole, brexpiprazole, olanzapine (in combination with fluoxetine only), and quetiapine are FDA-approved as adjunctive therapies with an antidepressant (Table 1). Second-generation antipsychotics should be started at lower doses than those used for schizophrenia, and these agents have an increased risk of metabolic adverse effects as well as extrapyramidal symptoms.
Atypical antidepressants are those that are not classified as an SSRI, SNRI, tricyclic antidepressant (TCA), or monoamine oxidase inhibitor (MAOI). These include bupropion, mirtazapine, trazodone, vilazodone, and vortioxetine (Table 2). Bupropion is a dopamine and norepinephrine reuptake inhibitor. When used for augmentation in clinical studies, it led to a 30% remission rate.3 Mirtazapine is an alpha-2 antagonist that can be used as monotherapy or in combination with another antidepressant.4 Trazodone is an antidepressant with activity at histamine and alpha-1-adrenergic receptors that is often used off-label for insomnia. Trazodone can be used safely and effectively in combination with other agents for treatment-resistant depression.5 Vilazodone is a 5-HT1A partial agonist, and vortioxetine is a 5-HT1A agonist and 5-HT3 antagonist; both are FDA-approved as alternative agents for monotherapy for major depressive disorder. Choosing among these agents for switching or augmenting can be guided by patient preference, adverse effect profile, and targeting specific symptoms, such as using mirtazapine to address poor sleep and appetite.
Lithium augmentation has been frequently investigated in placebo-controlled, double-blind studies. A meta-analysis showed that patients receiving lithium augmentation with a serum level of ≥0.5 mEq/L were >3 times more likely to respond than those receiving placebo.6 When lithium is used to treat bipolar disorder, the therapeutic serum range for lithium is 0.8 to 1.2 mEq/L, with an increased risk of adverse effects (including toxicity) at higher levels.7
Triiodothyronine (T3) augmentation of antidepressants led to remission in approximately 1 in 4 patients who had not achieved remission or who were intolerant to an initial treatment with citalopram and a second switch or augmentation trial.8 In this study, the mean dose of T3 was 45.2 µg/d, with an average length of treatment of 9 weeks.
Tricyclic antidepressants are another option when considering switching agents (Table 3). TCAs are additionally effective for comorbid pain conditions.9 When TCAs are used in combination with SSRIs, drug interactions may occur that increase TCA plasma levels. There is also an increased risk of serotonin syndrome when used with serotonergic agents, though an SSRI/ TCA combination may be appropriate for a patient with treatment-resistant depression.10 Additionally, TCAs carry unique risks of cardiovascular effects, including cardiac arrhythmias. A meta-analysis comparing fluoxetine, paroxetine, and sertraline to TCAs (amitriptyline, clomipramine, desipramine, doxepin, imipramine, and nortriptyline) concluded that both classes had similar efficacy in treating depression, though the drop-out rate was significantly higher among patients receiving TCAs.11
Buspirone is approved for generalized anxiety disorder. In studies where buspirone was used as an augmentation agent for major depressive disorder at a mean daily dose of 40.9 mg divided into 2 doses, it led to a remission rate >30%.3
Continue to: Monoamine oxidase inhibitors
Monoamine oxidase inhibitors should typically be avoided in initial or early treatment of depression due to tolerability issues, drug interactions, and dietary restrictions to avoid hypertensive crisis. MAOIs are generally not recommended to be used with SSRIs, SNRIs, or TCAs, and typically require a “washout” period from other antidepressants (Table 4). One review found that MAOI treatment had advantage over TCA treatment for patients with early-stage treatment-resistant depression, though this advantage decreased as the number of failed antidepressant trials increased.12 One MAOI, selegiline, is available in a transdermal patch, and the 6-mg patch does not require dietary restriction.
Esketamine (intranasal) is FDA-approved for treatment-resistant depression (failure of response after at least 2 antidepressant trials with adequate dose and duration) in conjunction with an oral antidepressant. In clinical studies, a significant response was noted after 1 week of treatment.13 Esketamine requires an induction period of twice-weekly doses of 56 or 84 mg, with maintenance doses every 1 to 2 weeks. Each dosage administration requires monitoring for at least 2 hours by a health care professional at a certified treatment center. Esketamine’s indication was recently expanded to include treatment of patients with major depressive disorder with suicidal ideation or behavior.
Stimulants such as amphetamines, methylphenidate, or modafinil have been effective in open studies for augmentation in depression.14 However, no stimulant is FDA-approved for the treatment of depression. In addition to other adverse effects, these medications are controlled substances and carry risk of misuse, and their use may not be appropriate for all patients.
Selective serotonin reuptake inhibitors (SSRIs) and serotonin-norepinephrine reuptake inhibitors (SNRIs) are commonly used first-line agents for treating major depressive disorder. Less than one-half of patients with major depressive disorder experience remission after 1 acute trial of an antidepressant.1 After optimization of an initial agent’s dose and duration, potential next steps include switching agents or augmentation. Augmentation strategies may lead to clinical improvement but carry the risks of polypharmacy, including increased risk of adverse effects and drug interactions. Clinicians can consider the following evidence-based options for a patient with a limited response to an initial SSRI or SNRI.
Second-generation antipsychotics, when used as augmentation agents to treat a patient with major depressive disorder, can lead to an approximately 10% improvement in remission rate compared with placebo.2 Aripiprazole, brexpiprazole, olanzapine (in combination with fluoxetine only), and quetiapine are FDA-approved as adjunctive therapies with an antidepressant (Table 1). Second-generation antipsychotics should be started at lower doses than those used for schizophrenia, and these agents have an increased risk of metabolic adverse effects as well as extrapyramidal symptoms.
Atypical antidepressants are those that are not classified as an SSRI, SNRI, tricyclic antidepressant (TCA), or monoamine oxidase inhibitor (MAOI). These include bupropion, mirtazapine, trazodone, vilazodone, and vortioxetine (Table 2). Bupropion is a dopamine and norepinephrine reuptake inhibitor. When used for augmentation in clinical studies, it led to a 30% remission rate.3 Mirtazapine is an alpha-2 antagonist that can be used as monotherapy or in combination with another antidepressant.4 Trazodone is an antidepressant with activity at histamine and alpha-1-adrenergic receptors that is often used off-label for insomnia. Trazodone can be used safely and effectively in combination with other agents for treatment-resistant depression.5 Vilazodone is a 5-HT1A partial agonist, and vortioxetine is a 5-HT1A agonist and 5-HT3 antagonist; both are FDA-approved as alternative agents for monotherapy for major depressive disorder. Choosing among these agents for switching or augmenting can be guided by patient preference, adverse effect profile, and targeting specific symptoms, such as using mirtazapine to address poor sleep and appetite.
Lithium augmentation has been frequently investigated in placebo-controlled, double-blind studies. A meta-analysis showed that patients receiving lithium augmentation with a serum level of ≥0.5 mEq/L were >3 times more likely to respond than those receiving placebo.6 When lithium is used to treat bipolar disorder, the therapeutic serum range for lithium is 0.8 to 1.2 mEq/L, with an increased risk of adverse effects (including toxicity) at higher levels.7
Triiodothyronine (T3) augmentation of antidepressants led to remission in approximately 1 in 4 patients who had not achieved remission or who were intolerant to an initial treatment with citalopram and a second switch or augmentation trial.8 In this study, the mean dose of T3 was 45.2 µg/d, with an average length of treatment of 9 weeks.
Tricyclic antidepressants are another option when considering switching agents (Table 3). TCAs are additionally effective for comorbid pain conditions.9 When TCAs are used in combination with SSRIs, drug interactions may occur that increase TCA plasma levels. There is also an increased risk of serotonin syndrome when used with serotonergic agents, though an SSRI/ TCA combination may be appropriate for a patient with treatment-resistant depression.10 Additionally, TCAs carry unique risks of cardiovascular effects, including cardiac arrhythmias. A meta-analysis comparing fluoxetine, paroxetine, and sertraline to TCAs (amitriptyline, clomipramine, desipramine, doxepin, imipramine, and nortriptyline) concluded that both classes had similar efficacy in treating depression, though the drop-out rate was significantly higher among patients receiving TCAs.11
Buspirone is approved for generalized anxiety disorder. In studies where buspirone was used as an augmentation agent for major depressive disorder at a mean daily dose of 40.9 mg divided into 2 doses, it led to a remission rate >30%.3
Continue to: Monoamine oxidase inhibitors
Monoamine oxidase inhibitors should typically be avoided in initial or early treatment of depression due to tolerability issues, drug interactions, and dietary restrictions to avoid hypertensive crisis. MAOIs are generally not recommended to be used with SSRIs, SNRIs, or TCAs, and typically require a “washout” period from other antidepressants (Table 4). One review found that MAOI treatment had advantage over TCA treatment for patients with early-stage treatment-resistant depression, though this advantage decreased as the number of failed antidepressant trials increased.12 One MAOI, selegiline, is available in a transdermal patch, and the 6-mg patch does not require dietary restriction.
Esketamine (intranasal) is FDA-approved for treatment-resistant depression (failure of response after at least 2 antidepressant trials with adequate dose and duration) in conjunction with an oral antidepressant. In clinical studies, a significant response was noted after 1 week of treatment.13 Esketamine requires an induction period of twice-weekly doses of 56 or 84 mg, with maintenance doses every 1 to 2 weeks. Each dosage administration requires monitoring for at least 2 hours by a health care professional at a certified treatment center. Esketamine’s indication was recently expanded to include treatment of patients with major depressive disorder with suicidal ideation or behavior.
Stimulants such as amphetamines, methylphenidate, or modafinil have been effective in open studies for augmentation in depression.14 However, no stimulant is FDA-approved for the treatment of depression. In addition to other adverse effects, these medications are controlled substances and carry risk of misuse, and their use may not be appropriate for all patients.
1. Trivedi MH, Rush AJ, Wisniewski SR, et al. Evaluation of outcomes with citalopram for depression using measurement-based care in STAR*D: implications for clinical practice. Am J Psychiatry. 2006;163(1):28-40.
2. Kato M, Chang CM. Augmentation treatments with second-generation antipsychotics to antidepressants in treatment-resistant depression. CNS Drugs. 2013;27 Suppl 1:S11-S19.
3. Trivedi MH, Fava M, Wisniewski SR, et al. Medication augmentation after the failure of SSRIs for depression. N Engl J Med. 2006;354(12):1243-1252.
4. Carpenter LL, Jocic Z, Hall JM, et al. Mirtazapine augmentation in the treatment of refractory depression. J Clin Psychiatry. 1999;60(1):45-49.
5. Maes M, Vandoolaeghe E, Desnyder R. Efficacy of treatment with trazodone in combination with pindolol or fluoxetine in major depression. J Affect Disord. 1996;41(3):201-210.
6. Bauer M, Dopfmer S. Lithium augmentation in treatment-resistant depression: meta-analysis of placebo-controlled studies. J Clin Psychopharmacol. 1999;19(5):427-434.
7. Yatham LN, Kennedy SH, Parikh SV, et al. Canadian Network for Mood and Anxiety Treatments (CANMAT) and International Society for Bipolar Disorders (ISBD) 2018 guidelines for the management of patients with bipolar disorder. Bipolar Disord. 2018;20(2):97-170.
8. Nierenberg AA, Fava M, Trivedi MH, et al. A comparison of lithium and T(3) augmentation following two failed medication treatments for depression: a STAR*D report. Am J Psychiatry. 2006;163(9):1519-1530; quiz 1665.
9. Saarto T, Wiffen PJ. Antidepressants for neuropathic pain. Cochrane Database Syst Rev. 2007;17(4):CD005454.
10. Taylor D. Selective serotonin reuptake inhibitors and tricyclic antidepressants in combination. Interactions and therapeutic uses. Br J Psychiatry. 1995;167(5):575-580.
11. Steffens DC, Krishnan KR, Helms MJ. Are SSRIs better than TCAs? Comparison of SSRIs and TCAs: a meta-analysis. Depress Anxiety. 1997;6(1):10-18.
12. Kim T, Xu C, Amsterdam JD. Relative effectiveness of tricyclic antidepressant versus monoamine oxidase inhibitor monotherapy for treatment-resistant depression. J Affect Disord. 2019;250:199-203.
13. Daly EJ, Singh JB, Fedgchin M, et al. Efficacy and safety of intranasal esketamine adjunctive to oral antidepressant therapy in treatment-resistant depression: a randomized clinical trial. JAMA Psychiatry. 2018;75(2):139-148.
14. DeBattista C. Augmentation and combination strategies for depression. J Psychopharmacol. 2006;20(3 Suppl):11-18.
1. Trivedi MH, Rush AJ, Wisniewski SR, et al. Evaluation of outcomes with citalopram for depression using measurement-based care in STAR*D: implications for clinical practice. Am J Psychiatry. 2006;163(1):28-40.
2. Kato M, Chang CM. Augmentation treatments with second-generation antipsychotics to antidepressants in treatment-resistant depression. CNS Drugs. 2013;27 Suppl 1:S11-S19.
3. Trivedi MH, Fava M, Wisniewski SR, et al. Medication augmentation after the failure of SSRIs for depression. N Engl J Med. 2006;354(12):1243-1252.
4. Carpenter LL, Jocic Z, Hall JM, et al. Mirtazapine augmentation in the treatment of refractory depression. J Clin Psychiatry. 1999;60(1):45-49.
5. Maes M, Vandoolaeghe E, Desnyder R. Efficacy of treatment with trazodone in combination with pindolol or fluoxetine in major depression. J Affect Disord. 1996;41(3):201-210.
6. Bauer M, Dopfmer S. Lithium augmentation in treatment-resistant depression: meta-analysis of placebo-controlled studies. J Clin Psychopharmacol. 1999;19(5):427-434.
7. Yatham LN, Kennedy SH, Parikh SV, et al. Canadian Network for Mood and Anxiety Treatments (CANMAT) and International Society for Bipolar Disorders (ISBD) 2018 guidelines for the management of patients with bipolar disorder. Bipolar Disord. 2018;20(2):97-170.
8. Nierenberg AA, Fava M, Trivedi MH, et al. A comparison of lithium and T(3) augmentation following two failed medication treatments for depression: a STAR*D report. Am J Psychiatry. 2006;163(9):1519-1530; quiz 1665.
9. Saarto T, Wiffen PJ. Antidepressants for neuropathic pain. Cochrane Database Syst Rev. 2007;17(4):CD005454.
10. Taylor D. Selective serotonin reuptake inhibitors and tricyclic antidepressants in combination. Interactions and therapeutic uses. Br J Psychiatry. 1995;167(5):575-580.
11. Steffens DC, Krishnan KR, Helms MJ. Are SSRIs better than TCAs? Comparison of SSRIs and TCAs: a meta-analysis. Depress Anxiety. 1997;6(1):10-18.
12. Kim T, Xu C, Amsterdam JD. Relative effectiveness of tricyclic antidepressant versus monoamine oxidase inhibitor monotherapy for treatment-resistant depression. J Affect Disord. 2019;250:199-203.
13. Daly EJ, Singh JB, Fedgchin M, et al. Efficacy and safety of intranasal esketamine adjunctive to oral antidepressant therapy in treatment-resistant depression: a randomized clinical trial. JAMA Psychiatry. 2018;75(2):139-148.
14. DeBattista C. Augmentation and combination strategies for depression. J Psychopharmacol. 2006;20(3 Suppl):11-18.
Toy soldier syndrome: A consequence of parental cognitive dissonance
Editor’s note: Readers’ Forum is a department for correspondence from readers that is not in response to articles published in
Childhood and adolescence are periods with marked psychobehavioral development of the brain. The sense of self, identity, and role are established. This process is not without risk because brain regions governing reward, impulsivity, and sensation-seeking are relatively more developed and influential than higher-order cognitive regions regulating behavioral inhibition, decision-making, and planning, which continue to mature into one’s early to mid-20s. Consequently, while the developing brain is “under construction” by forging new pathways and taking advantage of its immense neuroplasticity, it is also prone to psychological insults under exposure to stressful events, attitudes, and behaviors, including those that can arise in the family.1
Most people would agree that there is no stronger emotion than parental love. The origins of this powerful biobehavioral bonding with a child have been attributed to maternal release of oxytocin, known colloquially as the “love hormone,” during the birthing process, and to both biological parents experiencing psychosocial attachment with their infant. Therefore, common sense dictates that parents would do anything to protect their offspring, and that no parent would deliberately behave in a manner that harms their child.
Common sense notwithstanding, reports of both child neglect and abuse are common. States have established agencies to protect children from their own parents. The answers to the question “Whose kids are they?” and under what circumstances the state has the authority to warn or reprimand parents, or even temporarily or permanently separate minors from their parents, are complex and vary by state.
In this commentary, we describe harmful actions committed by parents with the intention of protecting the impressionable minds of their children from malevolent forces or intrusive and unhealthy ideas. Second, we examine how to protect a minor from parental actions that are well-meaning but potentially harmful.
Parent-child communication
Delusional family interactions. Originally described in 1877 as “folie à deux,”2 shared madness is an extreme and uncommon parental psychiatric condition harmful to a child’s mental health. It is primarily characterized by parental-initiated delusions shared with the child that are typically persecutory and attributed to danger from vengeful folks or grandiose in nature. The question of whether the “folie” or “madness” is contagious arises due to the propensity of the child to adopt these delusions under an imposed insular or restrictive environment. Separating the child from the environment dominated by the delusional adult usually is sufficient to reverse the symptoms due to reality testing.
Normative familial communication. In contrary to a delusional familial interaction, normative family traditions and values are a unifying psychosocial force and a source of bonding and loyalty from an early age. A ubiquitous example is the support of a local sports team, and the emotional turmoil associated with the team’s wins and losses, accompanied by “hating” a rival. These family rituals are commonly devoid of emotional negative consequences for an impressionable young mind unless the child is exposed to unsportsmanlike emotional, verbal, or aggressive behavior by an adult at home in front of the television or in the stands at a game.
Continue to: Unfortunately...
Unfortunately, the “love-hate” dichotomy rooted in family-generated traditions of loyalty is becoming more evident in today’s turbulent sociopolitical environment. Children and young adolescents are not prepared to cope with the stressful effects of repeated exposure to intense conflictual events at home when parents adopt opposing sociopolitical ideologies. Furthermore, a parent might intentionally expose their child to emotionally conflictual circumstances in the name of a perceived value that might create and exacerbate stress, fear, and self-loathing. Ironically, by doing what a parent believes is right for their child, they might be transforming the child without their consent into a variant of a “toy soldier by proxy.” Such a child is a tool expected to follow the parental pathway and belief system without questioning, or even having the cognitive ability to do so, given their ongoing bio-behavioral and moral developmental phase.3
This new normative exposure to conflictual situations at the will of the parent is not only limited to watching them remotely but also may include participating in what is meant to be a peaceful protest or march. As we all witnessed in 2020, such events can easily deteriorate into unsafe environments rife with lawlessness and uncontrolled violence. This has included clashes between opposing groups who are matched in zeal and conviction, as well as opposition to or endangerment by law enforcement personnel trying to restore order by force. This is not where a responsible parent should take their child. Furthermore, there is the danger of loss of privacy of children exposed by media following their participation in public activity. This may lead to hate mail as that would further confuse and jeopardize a peaceful lifestyle, which is highly desirable for a developing child.
Cognitive dissonance. Have these parents temporarily allowed the limbic system to trump the restraints of the prefrontal cortex, as exhibited by an impulsive and risky behavior driven by poor insight? Have these parents thoughtfully weighed the balance between the merit of a child’s exposure to such conflictual circumstances and the peril of negative emotional consequences? This is illustrated by a mother who has been taking her preadolescent son to demonstrations regularly because “I want him to see how democracy works.”
Might this be a case of cognitive dissonance (CD) that amounts to unwitting mental child abuse if it happens repeatedly? According to the CD theory, there is a tendency to seek consistency between cognitions (eg, beliefs, opinions) and attitudes or behaviors. Inconsistency between these variables is termed “dissonance.”4,5 The importance attached to the dissonant belief affects the severity of the dissonance. The dissonance occurs when a parent must choose between 2 incompatible beliefs or actions. A classic demonstration of CD is when an adult requests that an adolescent follows his instructions (eg, “do not smoke or drink alcohol”), yet the adult does not act accordingly (eg, they smoke or drink). Role modeling demonstrated by such a discrepancy is a cause of confusion in the child. In terms of this article, the CD is between what the parent believes is an important learning experience by exercising the perceived right to pass to the child the parental value system vs compromising the protection of the child by exposing them to the potential negative consequences of a risky situation.
What can parents and therapists do?
Usually, parents mean well. It is important to communicate to parents the importance of refraining from forcing their children to join their battles. Calculating risks based on an intuitive approach is flawed because doing so is based on beliefs and emotions that originated in the limbic system (“I feel that”…) and are neither precise nor accurate.6 Teaching our youth in the school system how to think (eg, the science of logic and history of science) vs what to think (ie, indoctrination) is a key to healthy cognitive development. Furthermore, children need to have the time, space, and opportunities (learning moments) to develop this capacity. It is not until approximately age 16 that abstract thinking capabilities are developed. Cognitive dissonance can be eliminated by reducing the valence of the conflicting beliefs or by removing the conflicting attitude or behavior.
As parents and as mental health professionals, we should carry the necessary burden of responsibility to prevent the risk of “lost childhood” due to parental emotional zeal and righteousness that lead to early exposure to damaging adversity. We cannot afford to turn our children into exploitable tools (ie, toy soldiers) in conflicts they do not fully grasp.
1. Bagot KS, Kaminer Y. Harm reduction for youth in treatment with substance use disorders: one size does not fit all. Curr Addict Rep. 2018;5:379-385.
2. Arnon D, Patel A, Tan GM. The nosological significance of Folie à Deux: a review of the literature. Ann Gen Psychiatry. 2006;5:11.
3. Kohlberg L. The philosophy of moral development: the nature and validity of moral stages. Harper & Row; 1984.
4. Festinger L. A theory of cognitive dissonance. Stanford University Press; 1957.
5. Festinger L. Cognitive dissonance. Sci Am. 1962;207:93-102.
6. Henderson SW, Gerson R, Phillips B. What is “high risk” and what are we actually supposed to do about it? J Am Acad Child Adolesc Psychiatry. 2019;58(6):561-564.
Editor’s note: Readers’ Forum is a department for correspondence from readers that is not in response to articles published in
Childhood and adolescence are periods with marked psychobehavioral development of the brain. The sense of self, identity, and role are established. This process is not without risk because brain regions governing reward, impulsivity, and sensation-seeking are relatively more developed and influential than higher-order cognitive regions regulating behavioral inhibition, decision-making, and planning, which continue to mature into one’s early to mid-20s. Consequently, while the developing brain is “under construction” by forging new pathways and taking advantage of its immense neuroplasticity, it is also prone to psychological insults under exposure to stressful events, attitudes, and behaviors, including those that can arise in the family.1
Most people would agree that there is no stronger emotion than parental love. The origins of this powerful biobehavioral bonding with a child have been attributed to maternal release of oxytocin, known colloquially as the “love hormone,” during the birthing process, and to both biological parents experiencing psychosocial attachment with their infant. Therefore, common sense dictates that parents would do anything to protect their offspring, and that no parent would deliberately behave in a manner that harms their child.
Common sense notwithstanding, reports of both child neglect and abuse are common. States have established agencies to protect children from their own parents. The answers to the question “Whose kids are they?” and under what circumstances the state has the authority to warn or reprimand parents, or even temporarily or permanently separate minors from their parents, are complex and vary by state.
In this commentary, we describe harmful actions committed by parents with the intention of protecting the impressionable minds of their children from malevolent forces or intrusive and unhealthy ideas. Second, we examine how to protect a minor from parental actions that are well-meaning but potentially harmful.
Parent-child communication
Delusional family interactions. Originally described in 1877 as “folie à deux,”2 shared madness is an extreme and uncommon parental psychiatric condition harmful to a child’s mental health. It is primarily characterized by parental-initiated delusions shared with the child that are typically persecutory and attributed to danger from vengeful folks or grandiose in nature. The question of whether the “folie” or “madness” is contagious arises due to the propensity of the child to adopt these delusions under an imposed insular or restrictive environment. Separating the child from the environment dominated by the delusional adult usually is sufficient to reverse the symptoms due to reality testing.
Normative familial communication. In contrary to a delusional familial interaction, normative family traditions and values are a unifying psychosocial force and a source of bonding and loyalty from an early age. A ubiquitous example is the support of a local sports team, and the emotional turmoil associated with the team’s wins and losses, accompanied by “hating” a rival. These family rituals are commonly devoid of emotional negative consequences for an impressionable young mind unless the child is exposed to unsportsmanlike emotional, verbal, or aggressive behavior by an adult at home in front of the television or in the stands at a game.
Continue to: Unfortunately...
Unfortunately, the “love-hate” dichotomy rooted in family-generated traditions of loyalty is becoming more evident in today’s turbulent sociopolitical environment. Children and young adolescents are not prepared to cope with the stressful effects of repeated exposure to intense conflictual events at home when parents adopt opposing sociopolitical ideologies. Furthermore, a parent might intentionally expose their child to emotionally conflictual circumstances in the name of a perceived value that might create and exacerbate stress, fear, and self-loathing. Ironically, by doing what a parent believes is right for their child, they might be transforming the child without their consent into a variant of a “toy soldier by proxy.” Such a child is a tool expected to follow the parental pathway and belief system without questioning, or even having the cognitive ability to do so, given their ongoing bio-behavioral and moral developmental phase.3
This new normative exposure to conflictual situations at the will of the parent is not only limited to watching them remotely but also may include participating in what is meant to be a peaceful protest or march. As we all witnessed in 2020, such events can easily deteriorate into unsafe environments rife with lawlessness and uncontrolled violence. This has included clashes between opposing groups who are matched in zeal and conviction, as well as opposition to or endangerment by law enforcement personnel trying to restore order by force. This is not where a responsible parent should take their child. Furthermore, there is the danger of loss of privacy of children exposed by media following their participation in public activity. This may lead to hate mail as that would further confuse and jeopardize a peaceful lifestyle, which is highly desirable for a developing child.
Cognitive dissonance. Have these parents temporarily allowed the limbic system to trump the restraints of the prefrontal cortex, as exhibited by an impulsive and risky behavior driven by poor insight? Have these parents thoughtfully weighed the balance between the merit of a child’s exposure to such conflictual circumstances and the peril of negative emotional consequences? This is illustrated by a mother who has been taking her preadolescent son to demonstrations regularly because “I want him to see how democracy works.”
Might this be a case of cognitive dissonance (CD) that amounts to unwitting mental child abuse if it happens repeatedly? According to the CD theory, there is a tendency to seek consistency between cognitions (eg, beliefs, opinions) and attitudes or behaviors. Inconsistency between these variables is termed “dissonance.”4,5 The importance attached to the dissonant belief affects the severity of the dissonance. The dissonance occurs when a parent must choose between 2 incompatible beliefs or actions. A classic demonstration of CD is when an adult requests that an adolescent follows his instructions (eg, “do not smoke or drink alcohol”), yet the adult does not act accordingly (eg, they smoke or drink). Role modeling demonstrated by such a discrepancy is a cause of confusion in the child. In terms of this article, the CD is between what the parent believes is an important learning experience by exercising the perceived right to pass to the child the parental value system vs compromising the protection of the child by exposing them to the potential negative consequences of a risky situation.
What can parents and therapists do?
Usually, parents mean well. It is important to communicate to parents the importance of refraining from forcing their children to join their battles. Calculating risks based on an intuitive approach is flawed because doing so is based on beliefs and emotions that originated in the limbic system (“I feel that”…) and are neither precise nor accurate.6 Teaching our youth in the school system how to think (eg, the science of logic and history of science) vs what to think (ie, indoctrination) is a key to healthy cognitive development. Furthermore, children need to have the time, space, and opportunities (learning moments) to develop this capacity. It is not until approximately age 16 that abstract thinking capabilities are developed. Cognitive dissonance can be eliminated by reducing the valence of the conflicting beliefs or by removing the conflicting attitude or behavior.
As parents and as mental health professionals, we should carry the necessary burden of responsibility to prevent the risk of “lost childhood” due to parental emotional zeal and righteousness that lead to early exposure to damaging adversity. We cannot afford to turn our children into exploitable tools (ie, toy soldiers) in conflicts they do not fully grasp.
Editor’s note: Readers’ Forum is a department for correspondence from readers that is not in response to articles published in
Childhood and adolescence are periods with marked psychobehavioral development of the brain. The sense of self, identity, and role are established. This process is not without risk because brain regions governing reward, impulsivity, and sensation-seeking are relatively more developed and influential than higher-order cognitive regions regulating behavioral inhibition, decision-making, and planning, which continue to mature into one’s early to mid-20s. Consequently, while the developing brain is “under construction” by forging new pathways and taking advantage of its immense neuroplasticity, it is also prone to psychological insults under exposure to stressful events, attitudes, and behaviors, including those that can arise in the family.1
Most people would agree that there is no stronger emotion than parental love. The origins of this powerful biobehavioral bonding with a child have been attributed to maternal release of oxytocin, known colloquially as the “love hormone,” during the birthing process, and to both biological parents experiencing psychosocial attachment with their infant. Therefore, common sense dictates that parents would do anything to protect their offspring, and that no parent would deliberately behave in a manner that harms their child.
Common sense notwithstanding, reports of both child neglect and abuse are common. States have established agencies to protect children from their own parents. The answers to the question “Whose kids are they?” and under what circumstances the state has the authority to warn or reprimand parents, or even temporarily or permanently separate minors from their parents, are complex and vary by state.
In this commentary, we describe harmful actions committed by parents with the intention of protecting the impressionable minds of their children from malevolent forces or intrusive and unhealthy ideas. Second, we examine how to protect a minor from parental actions that are well-meaning but potentially harmful.
Parent-child communication
Delusional family interactions. Originally described in 1877 as “folie à deux,”2 shared madness is an extreme and uncommon parental psychiatric condition harmful to a child’s mental health. It is primarily characterized by parental-initiated delusions shared with the child that are typically persecutory and attributed to danger from vengeful folks or grandiose in nature. The question of whether the “folie” or “madness” is contagious arises due to the propensity of the child to adopt these delusions under an imposed insular or restrictive environment. Separating the child from the environment dominated by the delusional adult usually is sufficient to reverse the symptoms due to reality testing.
Normative familial communication. In contrary to a delusional familial interaction, normative family traditions and values are a unifying psychosocial force and a source of bonding and loyalty from an early age. A ubiquitous example is the support of a local sports team, and the emotional turmoil associated with the team’s wins and losses, accompanied by “hating” a rival. These family rituals are commonly devoid of emotional negative consequences for an impressionable young mind unless the child is exposed to unsportsmanlike emotional, verbal, or aggressive behavior by an adult at home in front of the television or in the stands at a game.
Continue to: Unfortunately...
Unfortunately, the “love-hate” dichotomy rooted in family-generated traditions of loyalty is becoming more evident in today’s turbulent sociopolitical environment. Children and young adolescents are not prepared to cope with the stressful effects of repeated exposure to intense conflictual events at home when parents adopt opposing sociopolitical ideologies. Furthermore, a parent might intentionally expose their child to emotionally conflictual circumstances in the name of a perceived value that might create and exacerbate stress, fear, and self-loathing. Ironically, by doing what a parent believes is right for their child, they might be transforming the child without their consent into a variant of a “toy soldier by proxy.” Such a child is a tool expected to follow the parental pathway and belief system without questioning, or even having the cognitive ability to do so, given their ongoing bio-behavioral and moral developmental phase.3
This new normative exposure to conflictual situations at the will of the parent is not only limited to watching them remotely but also may include participating in what is meant to be a peaceful protest or march. As we all witnessed in 2020, such events can easily deteriorate into unsafe environments rife with lawlessness and uncontrolled violence. This has included clashes between opposing groups who are matched in zeal and conviction, as well as opposition to or endangerment by law enforcement personnel trying to restore order by force. This is not where a responsible parent should take their child. Furthermore, there is the danger of loss of privacy of children exposed by media following their participation in public activity. This may lead to hate mail as that would further confuse and jeopardize a peaceful lifestyle, which is highly desirable for a developing child.
Cognitive dissonance. Have these parents temporarily allowed the limbic system to trump the restraints of the prefrontal cortex, as exhibited by an impulsive and risky behavior driven by poor insight? Have these parents thoughtfully weighed the balance between the merit of a child’s exposure to such conflictual circumstances and the peril of negative emotional consequences? This is illustrated by a mother who has been taking her preadolescent son to demonstrations regularly because “I want him to see how democracy works.”
Might this be a case of cognitive dissonance (CD) that amounts to unwitting mental child abuse if it happens repeatedly? According to the CD theory, there is a tendency to seek consistency between cognitions (eg, beliefs, opinions) and attitudes or behaviors. Inconsistency between these variables is termed “dissonance.”4,5 The importance attached to the dissonant belief affects the severity of the dissonance. The dissonance occurs when a parent must choose between 2 incompatible beliefs or actions. A classic demonstration of CD is when an adult requests that an adolescent follows his instructions (eg, “do not smoke or drink alcohol”), yet the adult does not act accordingly (eg, they smoke or drink). Role modeling demonstrated by such a discrepancy is a cause of confusion in the child. In terms of this article, the CD is between what the parent believes is an important learning experience by exercising the perceived right to pass to the child the parental value system vs compromising the protection of the child by exposing them to the potential negative consequences of a risky situation.
What can parents and therapists do?
Usually, parents mean well. It is important to communicate to parents the importance of refraining from forcing their children to join their battles. Calculating risks based on an intuitive approach is flawed because doing so is based on beliefs and emotions that originated in the limbic system (“I feel that”…) and are neither precise nor accurate.6 Teaching our youth in the school system how to think (eg, the science of logic and history of science) vs what to think (ie, indoctrination) is a key to healthy cognitive development. Furthermore, children need to have the time, space, and opportunities (learning moments) to develop this capacity. It is not until approximately age 16 that abstract thinking capabilities are developed. Cognitive dissonance can be eliminated by reducing the valence of the conflicting beliefs or by removing the conflicting attitude or behavior.
As parents and as mental health professionals, we should carry the necessary burden of responsibility to prevent the risk of “lost childhood” due to parental emotional zeal and righteousness that lead to early exposure to damaging adversity. We cannot afford to turn our children into exploitable tools (ie, toy soldiers) in conflicts they do not fully grasp.
1. Bagot KS, Kaminer Y. Harm reduction for youth in treatment with substance use disorders: one size does not fit all. Curr Addict Rep. 2018;5:379-385.
2. Arnon D, Patel A, Tan GM. The nosological significance of Folie à Deux: a review of the literature. Ann Gen Psychiatry. 2006;5:11.
3. Kohlberg L. The philosophy of moral development: the nature and validity of moral stages. Harper & Row; 1984.
4. Festinger L. A theory of cognitive dissonance. Stanford University Press; 1957.
5. Festinger L. Cognitive dissonance. Sci Am. 1962;207:93-102.
6. Henderson SW, Gerson R, Phillips B. What is “high risk” and what are we actually supposed to do about it? J Am Acad Child Adolesc Psychiatry. 2019;58(6):561-564.
1. Bagot KS, Kaminer Y. Harm reduction for youth in treatment with substance use disorders: one size does not fit all. Curr Addict Rep. 2018;5:379-385.
2. Arnon D, Patel A, Tan GM. The nosological significance of Folie à Deux: a review of the literature. Ann Gen Psychiatry. 2006;5:11.
3. Kohlberg L. The philosophy of moral development: the nature and validity of moral stages. Harper & Row; 1984.
4. Festinger L. A theory of cognitive dissonance. Stanford University Press; 1957.
5. Festinger L. Cognitive dissonance. Sci Am. 1962;207:93-102.
6. Henderson SW, Gerson R, Phillips B. What is “high risk” and what are we actually supposed to do about it? J Am Acad Child Adolesc Psychiatry. 2019;58(6):561-564.
The world authority
Editor’s note: Readers’ Forum is a department for correspondence from readers that is not in response to articles published in
It was the late 1970s. She arrived by limousine; an attractive young woman, “Jasmine,” daughter of one of the richest men in the country. She was wearing a low-cut silk blouse and was adorned with an abundance of jewelry: large earrings, a necklace, and rings on nearly every finger. She smiled broadly when I introduced myself, shook her hand, and ushered her into my office.
She moved quickly and spoke in a rapid and pressured manner. She complained of poor sleep, mood swings, and racing thoughts. I began to ask her questions. “Have you used cocaine recently?” “What about Ritalin or amphetamines?” “Do you take prednisone or any other steroids?” “Do you have thyroid disease?” “Do you or anyone in your family have a history of manic depressive illness? Who? Have they ever required hospitalization for its treatment?”
“Well, doctor, what do you think?” she asked.
“I think that you probably have manic depressive illness and that you are currently having a manic episode. You should go into the hospital and begin treatment with lithium.”
“Who is the authority on this illness?” she asked.
“…The US authority or the world authority?” I replied.
“The world authority” she answered.
“Probably Professor Mogens Schou in Copenhagen,” I said.
“I’ll go see what he thinks,” she responded.
“Okay, you do that,” I replied.
When she left, I thought, “What a grandiose young woman, I doubt I’ll hear from her again.”
Continue to: Three days...
Three days later I received an unusual phone call.
“Dr. Jaffe, this is the long-distance operator, will you hold for Dr. Schou?”
“Of course,” I replied.
“Dr. Jaffe, I’m here with your patient, a charming young woman. I told her that I am in complete agreement with your diagnosis and treatment plan. She will be flying home tomorrow.”
A few days after she arrived home, I had Jasmine hospitalized under my care at one of the local psychiatric units. She stabilized nicely on lithium and tolerated it well. She remained there for about 3 weeks and was then discharged. I began seeing her in my office for weekly visits. After a few months we started meeting every 2 weeks, and eventually monthly.
She was doing well. Her mood swings were now mild and infrequent. Her sleep had normalized. Most important, she felt a lot more in control of her life.
Jasmine offered me a small window into the world of the super-rich and powerful. Basically, what I learned was that they are just like the rest of us, only more so. All the money provides both the opportunity to do a lot more good as well as to get into a lot more trouble. When a middle-class person gets manic and goes on a spending spree, they may blow a few hundred dollars on lottery tickets and perhaps a thousand dollars on clothing or gifts they don’t need. When the very rich do this, they buy airplanes, Ferraris, and vacation homes.
Jasmine and her siblings were often pestered—usually by acquaintances, but sometimes friends—for favors, usually loans, jobs, or introductions to other famous or powerful people. Jasmine turned out to be a lovely young woman, kind, generous, loyal to her friends and with a fine sense of humor. Getting to know her well helped dispel some of my prejudices about the adult children of the super-rich. I had incorrectly assumed that she would be quite spoiled and entitled.
After working together for approximately 2 years, we said our goodbyes because I was moving to a different part of the country. She thanked me for helping her get well. I asked her if there was anything in particular that she found most helpful. She surprised me when she answered so quickly.
“Yes. When you come from a very wealthy family, most people want something from you. You never wanted anything from me except my honesty” she said.
I thanked Jasmine for her gift.
Editor’s note: Readers’ Forum is a department for correspondence from readers that is not in response to articles published in
It was the late 1970s. She arrived by limousine; an attractive young woman, “Jasmine,” daughter of one of the richest men in the country. She was wearing a low-cut silk blouse and was adorned with an abundance of jewelry: large earrings, a necklace, and rings on nearly every finger. She smiled broadly when I introduced myself, shook her hand, and ushered her into my office.
She moved quickly and spoke in a rapid and pressured manner. She complained of poor sleep, mood swings, and racing thoughts. I began to ask her questions. “Have you used cocaine recently?” “What about Ritalin or amphetamines?” “Do you take prednisone or any other steroids?” “Do you have thyroid disease?” “Do you or anyone in your family have a history of manic depressive illness? Who? Have they ever required hospitalization for its treatment?”
“Well, doctor, what do you think?” she asked.
“I think that you probably have manic depressive illness and that you are currently having a manic episode. You should go into the hospital and begin treatment with lithium.”
“Who is the authority on this illness?” she asked.
“…The US authority or the world authority?” I replied.
“The world authority” she answered.
“Probably Professor Mogens Schou in Copenhagen,” I said.
“I’ll go see what he thinks,” she responded.
“Okay, you do that,” I replied.
When she left, I thought, “What a grandiose young woman, I doubt I’ll hear from her again.”
Continue to: Three days...
Three days later I received an unusual phone call.
“Dr. Jaffe, this is the long-distance operator, will you hold for Dr. Schou?”
“Of course,” I replied.
“Dr. Jaffe, I’m here with your patient, a charming young woman. I told her that I am in complete agreement with your diagnosis and treatment plan. She will be flying home tomorrow.”
A few days after she arrived home, I had Jasmine hospitalized under my care at one of the local psychiatric units. She stabilized nicely on lithium and tolerated it well. She remained there for about 3 weeks and was then discharged. I began seeing her in my office for weekly visits. After a few months we started meeting every 2 weeks, and eventually monthly.
She was doing well. Her mood swings were now mild and infrequent. Her sleep had normalized. Most important, she felt a lot more in control of her life.
Jasmine offered me a small window into the world of the super-rich and powerful. Basically, what I learned was that they are just like the rest of us, only more so. All the money provides both the opportunity to do a lot more good as well as to get into a lot more trouble. When a middle-class person gets manic and goes on a spending spree, they may blow a few hundred dollars on lottery tickets and perhaps a thousand dollars on clothing or gifts they don’t need. When the very rich do this, they buy airplanes, Ferraris, and vacation homes.
Jasmine and her siblings were often pestered—usually by acquaintances, but sometimes friends—for favors, usually loans, jobs, or introductions to other famous or powerful people. Jasmine turned out to be a lovely young woman, kind, generous, loyal to her friends and with a fine sense of humor. Getting to know her well helped dispel some of my prejudices about the adult children of the super-rich. I had incorrectly assumed that she would be quite spoiled and entitled.
After working together for approximately 2 years, we said our goodbyes because I was moving to a different part of the country. She thanked me for helping her get well. I asked her if there was anything in particular that she found most helpful. She surprised me when she answered so quickly.
“Yes. When you come from a very wealthy family, most people want something from you. You never wanted anything from me except my honesty” she said.
I thanked Jasmine for her gift.
Editor’s note: Readers’ Forum is a department for correspondence from readers that is not in response to articles published in
It was the late 1970s. She arrived by limousine; an attractive young woman, “Jasmine,” daughter of one of the richest men in the country. She was wearing a low-cut silk blouse and was adorned with an abundance of jewelry: large earrings, a necklace, and rings on nearly every finger. She smiled broadly when I introduced myself, shook her hand, and ushered her into my office.
She moved quickly and spoke in a rapid and pressured manner. She complained of poor sleep, mood swings, and racing thoughts. I began to ask her questions. “Have you used cocaine recently?” “What about Ritalin or amphetamines?” “Do you take prednisone or any other steroids?” “Do you have thyroid disease?” “Do you or anyone in your family have a history of manic depressive illness? Who? Have they ever required hospitalization for its treatment?”
“Well, doctor, what do you think?” she asked.
“I think that you probably have manic depressive illness and that you are currently having a manic episode. You should go into the hospital and begin treatment with lithium.”
“Who is the authority on this illness?” she asked.
“…The US authority or the world authority?” I replied.
“The world authority” she answered.
“Probably Professor Mogens Schou in Copenhagen,” I said.
“I’ll go see what he thinks,” she responded.
“Okay, you do that,” I replied.
When she left, I thought, “What a grandiose young woman, I doubt I’ll hear from her again.”
Continue to: Three days...
Three days later I received an unusual phone call.
“Dr. Jaffe, this is the long-distance operator, will you hold for Dr. Schou?”
“Of course,” I replied.
“Dr. Jaffe, I’m here with your patient, a charming young woman. I told her that I am in complete agreement with your diagnosis and treatment plan. She will be flying home tomorrow.”
A few days after she arrived home, I had Jasmine hospitalized under my care at one of the local psychiatric units. She stabilized nicely on lithium and tolerated it well. She remained there for about 3 weeks and was then discharged. I began seeing her in my office for weekly visits. After a few months we started meeting every 2 weeks, and eventually monthly.
She was doing well. Her mood swings were now mild and infrequent. Her sleep had normalized. Most important, she felt a lot more in control of her life.
Jasmine offered me a small window into the world of the super-rich and powerful. Basically, what I learned was that they are just like the rest of us, only more so. All the money provides both the opportunity to do a lot more good as well as to get into a lot more trouble. When a middle-class person gets manic and goes on a spending spree, they may blow a few hundred dollars on lottery tickets and perhaps a thousand dollars on clothing or gifts they don’t need. When the very rich do this, they buy airplanes, Ferraris, and vacation homes.
Jasmine and her siblings were often pestered—usually by acquaintances, but sometimes friends—for favors, usually loans, jobs, or introductions to other famous or powerful people. Jasmine turned out to be a lovely young woman, kind, generous, loyal to her friends and with a fine sense of humor. Getting to know her well helped dispel some of my prejudices about the adult children of the super-rich. I had incorrectly assumed that she would be quite spoiled and entitled.
After working together for approximately 2 years, we said our goodbyes because I was moving to a different part of the country. She thanked me for helping her get well. I asked her if there was anything in particular that she found most helpful. She surprised me when she answered so quickly.
“Yes. When you come from a very wealthy family, most people want something from you. You never wanted anything from me except my honesty” she said.
I thanked Jasmine for her gift.
Seasons of change
Since its inaugural issue in January 2007, the newspaper has evolved into a trusted source of clinically relevant updates on emerging practice trends and technological advances. I am honored to serve as the fourth editor of GIHN, building on the strong foundation set by former editors Charles J. Lightdale, MD, AGAF; Colin W. Howden, MD, AGAF; and most recently John I. Allen, MD, MBA, AGAF. Each of them has played an instrumental role in the publication’s growth and success over the past 15 years.
GIHN is unique among AGA’s flagship publications in that it is designed to bring together content from a variety of sources, including innovative scientific research from leading academic journals, practice management updates, and information regarding emerging policy initiatives impacting frontline GI practice. It also provides a platform to highlight AGA’s important work on behalf of its members. My goal as EIC is to continue to curate high-yield content that has the potential to directly impact how we manage our patients and practices. Several new initiatives are planned, which I am excited to introduce over the next few months. My door is always open, and I welcome your feedback about how GIHN can best serve the needs of AGA’s diverse membership in both academics and community practice.
Highlights of this month’s issue include updates on a unique multidisciplinary collaboration designed to promote a coordinated response among health care providers in caring for patients with NAFLD/NASH and AGA’s Clinical Practice Update on dysplasia management in patients with IBD. If you haven’t already, please consider nominating yourself or a colleague for an AGA committee appointment – the deadline is Nov. 1, and this is a fantastic way to contribute to the national dialogue on important issues affecting frontline GI practice.
Megan A. Adams, MD, JD, MSc
Since its inaugural issue in January 2007, the newspaper has evolved into a trusted source of clinically relevant updates on emerging practice trends and technological advances. I am honored to serve as the fourth editor of GIHN, building on the strong foundation set by former editors Charles J. Lightdale, MD, AGAF; Colin W. Howden, MD, AGAF; and most recently John I. Allen, MD, MBA, AGAF. Each of them has played an instrumental role in the publication’s growth and success over the past 15 years.
GIHN is unique among AGA’s flagship publications in that it is designed to bring together content from a variety of sources, including innovative scientific research from leading academic journals, practice management updates, and information regarding emerging policy initiatives impacting frontline GI practice. It also provides a platform to highlight AGA’s important work on behalf of its members. My goal as EIC is to continue to curate high-yield content that has the potential to directly impact how we manage our patients and practices. Several new initiatives are planned, which I am excited to introduce over the next few months. My door is always open, and I welcome your feedback about how GIHN can best serve the needs of AGA’s diverse membership in both academics and community practice.
Highlights of this month’s issue include updates on a unique multidisciplinary collaboration designed to promote a coordinated response among health care providers in caring for patients with NAFLD/NASH and AGA’s Clinical Practice Update on dysplasia management in patients with IBD. If you haven’t already, please consider nominating yourself or a colleague for an AGA committee appointment – the deadline is Nov. 1, and this is a fantastic way to contribute to the national dialogue on important issues affecting frontline GI practice.
Megan A. Adams, MD, JD, MSc
Since its inaugural issue in January 2007, the newspaper has evolved into a trusted source of clinically relevant updates on emerging practice trends and technological advances. I am honored to serve as the fourth editor of GIHN, building on the strong foundation set by former editors Charles J. Lightdale, MD, AGAF; Colin W. Howden, MD, AGAF; and most recently John I. Allen, MD, MBA, AGAF. Each of them has played an instrumental role in the publication’s growth and success over the past 15 years.
GIHN is unique among AGA’s flagship publications in that it is designed to bring together content from a variety of sources, including innovative scientific research from leading academic journals, practice management updates, and information regarding emerging policy initiatives impacting frontline GI practice. It also provides a platform to highlight AGA’s important work on behalf of its members. My goal as EIC is to continue to curate high-yield content that has the potential to directly impact how we manage our patients and practices. Several new initiatives are planned, which I am excited to introduce over the next few months. My door is always open, and I welcome your feedback about how GIHN can best serve the needs of AGA’s diverse membership in both academics and community practice.
Highlights of this month’s issue include updates on a unique multidisciplinary collaboration designed to promote a coordinated response among health care providers in caring for patients with NAFLD/NASH and AGA’s Clinical Practice Update on dysplasia management in patients with IBD. If you haven’t already, please consider nominating yourself or a colleague for an AGA committee appointment – the deadline is Nov. 1, and this is a fantastic way to contribute to the national dialogue on important issues affecting frontline GI practice.
Megan A. Adams, MD, JD, MSc
Use of Biomarkers to Optimize Treatment of NSCLC
Benjamin Cooper, MD, Director of Proton Therapy at NYU Langone Health, discusses how physicians who treat patients with non-small cell lung cancer (NSCLC) can use genetic profiling results to select effective therapy. Although the current list of therapies is not applicable to all genetic mutations, there are approved treatments for several biomarkers and agents targeting other biomarkers are in clinical trials.
Dr. Cooper explains that biomarkers in NSCLC either boost the immune system’s capability to destroy oncogenes or they block driver and escape mutations that advance disease.
Immunotherapies that target either PD-1 or PD-L1 are now mainstays of NSCLC treatment. To gauge whether these therapies have potential effectiveness for a given patient, oncologists test for the presence of PD-L1 in the tumor. Higher expression of PD-L1 indicates stronger potential response to therapy.
Dr. Cooper then turns to a discussion of oncogenic driver mutations, focusing on EGFR, ALK, ROS1, BRAF, NTRK, RET, MET, KRAS, and HER2. Although there are hundreds of oncogenic driver mutations, not all are currently actionable. Effective therapy options have been available for EGFR, ALK, and BRAF for more than a decade, and treatments for other drivers such as NTRK, MET, KRAS, and HER2 have shown promising results in recent trials.
--
Benjamin Cooper, MD is an Assistant Professor, Department of Radiation Oncology, Director, Proton Therapy Services, NYU Grossman School of Medicine,
New York, New York
Benjamin Cooper, MD, has disclosed the following relevant financial relationships:
Serve(d) as a director, officer, partner, employee, advisor, consultant, or trustee for: AstraZeneca.
Benjamin Cooper, MD, Director of Proton Therapy at NYU Langone Health, discusses how physicians who treat patients with non-small cell lung cancer (NSCLC) can use genetic profiling results to select effective therapy. Although the current list of therapies is not applicable to all genetic mutations, there are approved treatments for several biomarkers and agents targeting other biomarkers are in clinical trials.
Dr. Cooper explains that biomarkers in NSCLC either boost the immune system’s capability to destroy oncogenes or they block driver and escape mutations that advance disease.
Immunotherapies that target either PD-1 or PD-L1 are now mainstays of NSCLC treatment. To gauge whether these therapies have potential effectiveness for a given patient, oncologists test for the presence of PD-L1 in the tumor. Higher expression of PD-L1 indicates stronger potential response to therapy.
Dr. Cooper then turns to a discussion of oncogenic driver mutations, focusing on EGFR, ALK, ROS1, BRAF, NTRK, RET, MET, KRAS, and HER2. Although there are hundreds of oncogenic driver mutations, not all are currently actionable. Effective therapy options have been available for EGFR, ALK, and BRAF for more than a decade, and treatments for other drivers such as NTRK, MET, KRAS, and HER2 have shown promising results in recent trials.
--
Benjamin Cooper, MD is an Assistant Professor, Department of Radiation Oncology, Director, Proton Therapy Services, NYU Grossman School of Medicine,
New York, New York
Benjamin Cooper, MD, has disclosed the following relevant financial relationships:
Serve(d) as a director, officer, partner, employee, advisor, consultant, or trustee for: AstraZeneca.
Benjamin Cooper, MD, Director of Proton Therapy at NYU Langone Health, discusses how physicians who treat patients with non-small cell lung cancer (NSCLC) can use genetic profiling results to select effective therapy. Although the current list of therapies is not applicable to all genetic mutations, there are approved treatments for several biomarkers and agents targeting other biomarkers are in clinical trials.
Dr. Cooper explains that biomarkers in NSCLC either boost the immune system’s capability to destroy oncogenes or they block driver and escape mutations that advance disease.
Immunotherapies that target either PD-1 or PD-L1 are now mainstays of NSCLC treatment. To gauge whether these therapies have potential effectiveness for a given patient, oncologists test for the presence of PD-L1 in the tumor. Higher expression of PD-L1 indicates stronger potential response to therapy.
Dr. Cooper then turns to a discussion of oncogenic driver mutations, focusing on EGFR, ALK, ROS1, BRAF, NTRK, RET, MET, KRAS, and HER2. Although there are hundreds of oncogenic driver mutations, not all are currently actionable. Effective therapy options have been available for EGFR, ALK, and BRAF for more than a decade, and treatments for other drivers such as NTRK, MET, KRAS, and HER2 have shown promising results in recent trials.
--
Benjamin Cooper, MD is an Assistant Professor, Department of Radiation Oncology, Director, Proton Therapy Services, NYU Grossman School of Medicine,
New York, New York
Benjamin Cooper, MD, has disclosed the following relevant financial relationships:
Serve(d) as a director, officer, partner, employee, advisor, consultant, or trustee for: AstraZeneca.

Improving access to liver disease screening in at-risk and underserved communities
Dr. Ponni V. Perumalswami is an Associate Professor of Internal Medicine in the Division of Gastroenterology and Hepatology at the University of Michigan; Ann Arbor VA Healthcare System. Dr. Perumalswami's areas of clinical interest include cirrhosis, acute/chronic liver diseases and liver transplantation. Her research program focuses on community outreach for hepatitis B and C screening and linking patients to care.
Q: For patients with liver disease who live in underserved and vulnerable communities, what barriers to that care are more prominent or at the primary systemic-level?
Dr. Perumalswami: I think a major barrier has been our approach to thinking about these barriers, so I'm glad you are asking this question in terms of what the systemic-level barriers are rather than, for example, patient-level barriers. I'll use viral hepatitis as an example in terms of liver-disease care. I think for a very long time we've placed an unfair, onus on patients, leaving them to find their own care and to navigate existing system-level barriers such as language proficiency, health literacy, lack of insurance, and long distances to access specialists by themselves. This “do-it-yourself” approach has created a systemic-level barrier to finding specialists, and it remains a major problem. We would do a better job of improving access to care by re-thinking all barriers to care as system-and provider-level barriers rather than patient-level barriers because it is often the case that solutions that address systems barriers can address these issues.
Many specialists, like myself, are often geographically clustered at tertiary care and urban academic centers but the reality is that patients who are at risk for living with liver disease live all over, including rural areas, where fewer specialists practice. But advancements in treatments of certain liver diseases, such as hepatitis C virus (HCV), have made it possible for frontline community providers to treat patients. Expanding these patients' treatment options, in large part, is dependent on payer policy changes to allow treatment by non-specialists, reducing the cost of treatment and giving frontline workers the support they need so that they are more confident to offer treatment while differentiating the occasional patient who may need referral to a specialist.
Cost is also a systemic-level barrier for our underserved patients with liver disease and comes in many forms. Barriers related to cost include lack or type of insurance, traveling distances which also entails a cost for time (i.e. loss of wages, caregiver support), and cost of treatments. There are certain restrictions on HCV treatments that designate who can administer them and at what point in disease progression the therapy be introduced. These restrictions have been arbitrarily set by payers for treatments like direct-acting antivirals (DAA) and unfortunately dictate "when they can be obtained" and "who can prescribe them." Instead, we should spend time and effort to determine how we can have more providers practicing in different spaces, who might be equipped and motivated to provide treatment, and do it safely and easily.
Another barrier I will mention is the lack of integrated care for patients with certain liver diseases within healthcare systems. Notable examples include integrated care for those with alcohol-associated liver disease and viral hepatitis, who often have co-occurring mental health issues and substance use, addiction, or opioid use disorders. We need to think through how we can get integrated treatment and care to these patients, instead of making them come to us as individual specialists. By integrating medical care into behavioral health practices or other treatment settings, and perhaps by considering nontraditional treatment modalities, we can overcome barriers to care that are all too often siloed.
The last thing that I will mention with regards to patient-level barriers is that liver diseases and their care by providers has been very stigmatized, particularly for patients with underlying mental health and/or addiction disorders. These patients do not always feel comfortable coming to see clinicians in their practices so we must recognize that our offices may be stigmatized places for some patients with liver disease. Because of this, it is vital to think about how we can integrate care into trusted spaces for patient populations who might be at risk or are living with liver disease.
Q: What aspects of these barriers have you focused on to improve screening and links to care in communities at risk?
Dr. Perumalswami: A lot of my work is focused on patients in populations who are at risk for viral hepatitis and on screening them, educating them, and linking them to care in their communities. The challenge in successfully treating patients with a liver disease is that most liver diseases remain silent until they've progressed to a very advanced stage. Certain populations are at a higher risk for contracting these diseases compared to others. For example, with hepatitis B virus, we know that foreign-born populations have a higher infection rate, and how and when they seek care might be very different in terms of being symptom-driven versus preventive care as a result of cultural factors around health seeking behaviors. Our team has attempted to take a more proactive approach; first, to understand who might be at risk, and second, to try to bring screening to trusted places where patients can easily access care. We have found this proactive approach to be very successful in terms of identifying people who are not yet diagnosed with liver disease and then linking them into care.
The first step is knowing which populations you want to target with respect to individual types of liver disease, then working with community partners to bring screening out into the community. Obviously, the challenging part is getting people linked into care. As stated previously, many liver diseases in their earlier stages stay silent and manifest without symptoms, thus why it is vital to offer at-risk patients testing or screening.
The next step is to raise patient awareness and provide education as to why it is important to seek care; to get a thorough evaluation in terms of the extent of the liver disease and how to best manage and treat it, long term. For example, we have found that care coordination works very well with patients living with HCV. For patients with hepatitis B, we have found that culturally informed patient navigation services are very helpful, so we work with peers in the community who speak the same language and who come from the same communities as the patients identified as at-risk. This combined strategy of testing and then linking to care has been very successful.
I will say an important part of the care-coordination piece is addressing the competing priorities that patients have in their lives. For example, if they need housing, we refer them to housing services; if they have food insecurities, we try to address the need. Once you address their basic determinants of health, you have established a basis for trust while helping patients contend with important competing priorities. This way, your team has enabled potential patients to prioritize and engage in health care.
Q: How have you integrated HCV treatment into harm reduction and opioid use disorder settings?
Dr. Perumalswami: I am fortunate to be involved with a program here in Michigan whose goal is to increase HCV treatment through an open access, HCV consultation program through the Michigan Opioid Collaborative. The premise is to find motivated, interested providers who want to learn how to offer HCV treatment to patients in their communities; the majority of these providers are in rural parts of Michigan. In this setting, we are working with frontline medical personnel in the community, many of whom are either addiction providers or are offering opioid use disorder treatment, and who are also seeing HCV patients. We have set up an open-case consulting program where providers can submit cases for review with guidance from hepatologists. Attendance is optional and we meet for an hour, every other week and we talk through cases in more detail as a group. The result is that the providers have reported that they feel less isolated doing this as a team, having a place to discuss cases and work through practical challenges that can arise with this patient base. While HCV treatment advances have made great strides, many providers want reassurance or guidance in terms how to implement these programs so as a group, we walk through a few cases, demonstrate how to check for drug-drug interactions and how to perform fibrosis assessments. After these providers go through this training, they become more comfortable giving treatment on their own.
The second project, which I have also been fortunate to be involved in, is led by my colleague Dr. Jeffrey Weiss at Mount Sinai Hospital and is located at a syringe exchange program in a Brooklyn, New York. Here, patients attend receive in-person and/or telemedicine-based HCV treatment, which is a new model of care for us. While it has produced a different set of challenges in terms of engaging and bringing treatment to patients in a new space, it has been a great way to meet our objectives of helping patients to be treated where they are comfortable accessing care and services.
Q: Has the pandemic created any new challenges in treating at risk or special populations?
Dr. Perumalswami: The pandemic presented many new challenges. The primary impact that COVID-19 has had on our patients has been with the disruption in care; particularly for those patients who already found it challenging to seek and receive care. For patients who benefitted from following a routine, other pandemic-related challenges were the restrictions placed on our practices, and the reduced hours patients had to contend with access services and treatments at places such as syringe exchange programs or methadone programs.
Many of our patients have expressed feeling isolated as they are not able to get the same type of support that they were previously receiving. The decreases in viral hepatitis outreach, in screening in the community, and in practices resulted in a decrease in diagnosis and treatment.
We have also heard numerous discussions with regards to better reimbursements for phone call and telehealth sessions, but we must recognize that those things are not accessible to all patients. Many of our most vulnerable populations, do not have working phones, stable housing, or smart devices to access telehealth, so while there have been technological advances that can provide access to care and better reimbursement procedures, there are still many limitations that our patients are facing.
(AGA applauds researchers who are working to raise our awareness of health disparities in digestive diseases. AGA is committed to addressing this important societal issue head on. Learn more about AGA’s commitment through the AGA Equity Project).
Dr. Ponni V. Perumalswami is an Associate Professor of Internal Medicine in the Division of Gastroenterology and Hepatology at the University of Michigan; Ann Arbor VA Healthcare System. Dr. Perumalswami's areas of clinical interest include cirrhosis, acute/chronic liver diseases and liver transplantation. Her research program focuses on community outreach for hepatitis B and C screening and linking patients to care.
Q: For patients with liver disease who live in underserved and vulnerable communities, what barriers to that care are more prominent or at the primary systemic-level?
Dr. Perumalswami: I think a major barrier has been our approach to thinking about these barriers, so I'm glad you are asking this question in terms of what the systemic-level barriers are rather than, for example, patient-level barriers. I'll use viral hepatitis as an example in terms of liver-disease care. I think for a very long time we've placed an unfair, onus on patients, leaving them to find their own care and to navigate existing system-level barriers such as language proficiency, health literacy, lack of insurance, and long distances to access specialists by themselves. This “do-it-yourself” approach has created a systemic-level barrier to finding specialists, and it remains a major problem. We would do a better job of improving access to care by re-thinking all barriers to care as system-and provider-level barriers rather than patient-level barriers because it is often the case that solutions that address systems barriers can address these issues.
Many specialists, like myself, are often geographically clustered at tertiary care and urban academic centers but the reality is that patients who are at risk for living with liver disease live all over, including rural areas, where fewer specialists practice. But advancements in treatments of certain liver diseases, such as hepatitis C virus (HCV), have made it possible for frontline community providers to treat patients. Expanding these patients' treatment options, in large part, is dependent on payer policy changes to allow treatment by non-specialists, reducing the cost of treatment and giving frontline workers the support they need so that they are more confident to offer treatment while differentiating the occasional patient who may need referral to a specialist.
Cost is also a systemic-level barrier for our underserved patients with liver disease and comes in many forms. Barriers related to cost include lack or type of insurance, traveling distances which also entails a cost for time (i.e. loss of wages, caregiver support), and cost of treatments. There are certain restrictions on HCV treatments that designate who can administer them and at what point in disease progression the therapy be introduced. These restrictions have been arbitrarily set by payers for treatments like direct-acting antivirals (DAA) and unfortunately dictate "when they can be obtained" and "who can prescribe them." Instead, we should spend time and effort to determine how we can have more providers practicing in different spaces, who might be equipped and motivated to provide treatment, and do it safely and easily.
Another barrier I will mention is the lack of integrated care for patients with certain liver diseases within healthcare systems. Notable examples include integrated care for those with alcohol-associated liver disease and viral hepatitis, who often have co-occurring mental health issues and substance use, addiction, or opioid use disorders. We need to think through how we can get integrated treatment and care to these patients, instead of making them come to us as individual specialists. By integrating medical care into behavioral health practices or other treatment settings, and perhaps by considering nontraditional treatment modalities, we can overcome barriers to care that are all too often siloed.
The last thing that I will mention with regards to patient-level barriers is that liver diseases and their care by providers has been very stigmatized, particularly for patients with underlying mental health and/or addiction disorders. These patients do not always feel comfortable coming to see clinicians in their practices so we must recognize that our offices may be stigmatized places for some patients with liver disease. Because of this, it is vital to think about how we can integrate care into trusted spaces for patient populations who might be at risk or are living with liver disease.
Q: What aspects of these barriers have you focused on to improve screening and links to care in communities at risk?
Dr. Perumalswami: A lot of my work is focused on patients in populations who are at risk for viral hepatitis and on screening them, educating them, and linking them to care in their communities. The challenge in successfully treating patients with a liver disease is that most liver diseases remain silent until they've progressed to a very advanced stage. Certain populations are at a higher risk for contracting these diseases compared to others. For example, with hepatitis B virus, we know that foreign-born populations have a higher infection rate, and how and when they seek care might be very different in terms of being symptom-driven versus preventive care as a result of cultural factors around health seeking behaviors. Our team has attempted to take a more proactive approach; first, to understand who might be at risk, and second, to try to bring screening to trusted places where patients can easily access care. We have found this proactive approach to be very successful in terms of identifying people who are not yet diagnosed with liver disease and then linking them into care.
The first step is knowing which populations you want to target with respect to individual types of liver disease, then working with community partners to bring screening out into the community. Obviously, the challenging part is getting people linked into care. As stated previously, many liver diseases in their earlier stages stay silent and manifest without symptoms, thus why it is vital to offer at-risk patients testing or screening.
The next step is to raise patient awareness and provide education as to why it is important to seek care; to get a thorough evaluation in terms of the extent of the liver disease and how to best manage and treat it, long term. For example, we have found that care coordination works very well with patients living with HCV. For patients with hepatitis B, we have found that culturally informed patient navigation services are very helpful, so we work with peers in the community who speak the same language and who come from the same communities as the patients identified as at-risk. This combined strategy of testing and then linking to care has been very successful.
I will say an important part of the care-coordination piece is addressing the competing priorities that patients have in their lives. For example, if they need housing, we refer them to housing services; if they have food insecurities, we try to address the need. Once you address their basic determinants of health, you have established a basis for trust while helping patients contend with important competing priorities. This way, your team has enabled potential patients to prioritize and engage in health care.
Q: How have you integrated HCV treatment into harm reduction and opioid use disorder settings?
Dr. Perumalswami: I am fortunate to be involved with a program here in Michigan whose goal is to increase HCV treatment through an open access, HCV consultation program through the Michigan Opioid Collaborative. The premise is to find motivated, interested providers who want to learn how to offer HCV treatment to patients in their communities; the majority of these providers are in rural parts of Michigan. In this setting, we are working with frontline medical personnel in the community, many of whom are either addiction providers or are offering opioid use disorder treatment, and who are also seeing HCV patients. We have set up an open-case consulting program where providers can submit cases for review with guidance from hepatologists. Attendance is optional and we meet for an hour, every other week and we talk through cases in more detail as a group. The result is that the providers have reported that they feel less isolated doing this as a team, having a place to discuss cases and work through practical challenges that can arise with this patient base. While HCV treatment advances have made great strides, many providers want reassurance or guidance in terms how to implement these programs so as a group, we walk through a few cases, demonstrate how to check for drug-drug interactions and how to perform fibrosis assessments. After these providers go through this training, they become more comfortable giving treatment on their own.
The second project, which I have also been fortunate to be involved in, is led by my colleague Dr. Jeffrey Weiss at Mount Sinai Hospital and is located at a syringe exchange program in a Brooklyn, New York. Here, patients attend receive in-person and/or telemedicine-based HCV treatment, which is a new model of care for us. While it has produced a different set of challenges in terms of engaging and bringing treatment to patients in a new space, it has been a great way to meet our objectives of helping patients to be treated where they are comfortable accessing care and services.
Q: Has the pandemic created any new challenges in treating at risk or special populations?
Dr. Perumalswami: The pandemic presented many new challenges. The primary impact that COVID-19 has had on our patients has been with the disruption in care; particularly for those patients who already found it challenging to seek and receive care. For patients who benefitted from following a routine, other pandemic-related challenges were the restrictions placed on our practices, and the reduced hours patients had to contend with access services and treatments at places such as syringe exchange programs or methadone programs.
Many of our patients have expressed feeling isolated as they are not able to get the same type of support that they were previously receiving. The decreases in viral hepatitis outreach, in screening in the community, and in practices resulted in a decrease in diagnosis and treatment.
We have also heard numerous discussions with regards to better reimbursements for phone call and telehealth sessions, but we must recognize that those things are not accessible to all patients. Many of our most vulnerable populations, do not have working phones, stable housing, or smart devices to access telehealth, so while there have been technological advances that can provide access to care and better reimbursement procedures, there are still many limitations that our patients are facing.
(AGA applauds researchers who are working to raise our awareness of health disparities in digestive diseases. AGA is committed to addressing this important societal issue head on. Learn more about AGA’s commitment through the AGA Equity Project).
Dr. Ponni V. Perumalswami is an Associate Professor of Internal Medicine in the Division of Gastroenterology and Hepatology at the University of Michigan; Ann Arbor VA Healthcare System. Dr. Perumalswami's areas of clinical interest include cirrhosis, acute/chronic liver diseases and liver transplantation. Her research program focuses on community outreach for hepatitis B and C screening and linking patients to care.
Q: For patients with liver disease who live in underserved and vulnerable communities, what barriers to that care are more prominent or at the primary systemic-level?
Dr. Perumalswami: I think a major barrier has been our approach to thinking about these barriers, so I'm glad you are asking this question in terms of what the systemic-level barriers are rather than, for example, patient-level barriers. I'll use viral hepatitis as an example in terms of liver-disease care. I think for a very long time we've placed an unfair, onus on patients, leaving them to find their own care and to navigate existing system-level barriers such as language proficiency, health literacy, lack of insurance, and long distances to access specialists by themselves. This “do-it-yourself” approach has created a systemic-level barrier to finding specialists, and it remains a major problem. We would do a better job of improving access to care by re-thinking all barriers to care as system-and provider-level barriers rather than patient-level barriers because it is often the case that solutions that address systems barriers can address these issues.
Many specialists, like myself, are often geographically clustered at tertiary care and urban academic centers but the reality is that patients who are at risk for living with liver disease live all over, including rural areas, where fewer specialists practice. But advancements in treatments of certain liver diseases, such as hepatitis C virus (HCV), have made it possible for frontline community providers to treat patients. Expanding these patients' treatment options, in large part, is dependent on payer policy changes to allow treatment by non-specialists, reducing the cost of treatment and giving frontline workers the support they need so that they are more confident to offer treatment while differentiating the occasional patient who may need referral to a specialist.
Cost is also a systemic-level barrier for our underserved patients with liver disease and comes in many forms. Barriers related to cost include lack or type of insurance, traveling distances which also entails a cost for time (i.e. loss of wages, caregiver support), and cost of treatments. There are certain restrictions on HCV treatments that designate who can administer them and at what point in disease progression the therapy be introduced. These restrictions have been arbitrarily set by payers for treatments like direct-acting antivirals (DAA) and unfortunately dictate "when they can be obtained" and "who can prescribe them." Instead, we should spend time and effort to determine how we can have more providers practicing in different spaces, who might be equipped and motivated to provide treatment, and do it safely and easily.
Another barrier I will mention is the lack of integrated care for patients with certain liver diseases within healthcare systems. Notable examples include integrated care for those with alcohol-associated liver disease and viral hepatitis, who often have co-occurring mental health issues and substance use, addiction, or opioid use disorders. We need to think through how we can get integrated treatment and care to these patients, instead of making them come to us as individual specialists. By integrating medical care into behavioral health practices or other treatment settings, and perhaps by considering nontraditional treatment modalities, we can overcome barriers to care that are all too often siloed.
The last thing that I will mention with regards to patient-level barriers is that liver diseases and their care by providers has been very stigmatized, particularly for patients with underlying mental health and/or addiction disorders. These patients do not always feel comfortable coming to see clinicians in their practices so we must recognize that our offices may be stigmatized places for some patients with liver disease. Because of this, it is vital to think about how we can integrate care into trusted spaces for patient populations who might be at risk or are living with liver disease.
Q: What aspects of these barriers have you focused on to improve screening and links to care in communities at risk?
Dr. Perumalswami: A lot of my work is focused on patients in populations who are at risk for viral hepatitis and on screening them, educating them, and linking them to care in their communities. The challenge in successfully treating patients with a liver disease is that most liver diseases remain silent until they've progressed to a very advanced stage. Certain populations are at a higher risk for contracting these diseases compared to others. For example, with hepatitis B virus, we know that foreign-born populations have a higher infection rate, and how and when they seek care might be very different in terms of being symptom-driven versus preventive care as a result of cultural factors around health seeking behaviors. Our team has attempted to take a more proactive approach; first, to understand who might be at risk, and second, to try to bring screening to trusted places where patients can easily access care. We have found this proactive approach to be very successful in terms of identifying people who are not yet diagnosed with liver disease and then linking them into care.
The first step is knowing which populations you want to target with respect to individual types of liver disease, then working with community partners to bring screening out into the community. Obviously, the challenging part is getting people linked into care. As stated previously, many liver diseases in their earlier stages stay silent and manifest without symptoms, thus why it is vital to offer at-risk patients testing or screening.
The next step is to raise patient awareness and provide education as to why it is important to seek care; to get a thorough evaluation in terms of the extent of the liver disease and how to best manage and treat it, long term. For example, we have found that care coordination works very well with patients living with HCV. For patients with hepatitis B, we have found that culturally informed patient navigation services are very helpful, so we work with peers in the community who speak the same language and who come from the same communities as the patients identified as at-risk. This combined strategy of testing and then linking to care has been very successful.
I will say an important part of the care-coordination piece is addressing the competing priorities that patients have in their lives. For example, if they need housing, we refer them to housing services; if they have food insecurities, we try to address the need. Once you address their basic determinants of health, you have established a basis for trust while helping patients contend with important competing priorities. This way, your team has enabled potential patients to prioritize and engage in health care.
Q: How have you integrated HCV treatment into harm reduction and opioid use disorder settings?
Dr. Perumalswami: I am fortunate to be involved with a program here in Michigan whose goal is to increase HCV treatment through an open access, HCV consultation program through the Michigan Opioid Collaborative. The premise is to find motivated, interested providers who want to learn how to offer HCV treatment to patients in their communities; the majority of these providers are in rural parts of Michigan. In this setting, we are working with frontline medical personnel in the community, many of whom are either addiction providers or are offering opioid use disorder treatment, and who are also seeing HCV patients. We have set up an open-case consulting program where providers can submit cases for review with guidance from hepatologists. Attendance is optional and we meet for an hour, every other week and we talk through cases in more detail as a group. The result is that the providers have reported that they feel less isolated doing this as a team, having a place to discuss cases and work through practical challenges that can arise with this patient base. While HCV treatment advances have made great strides, many providers want reassurance or guidance in terms how to implement these programs so as a group, we walk through a few cases, demonstrate how to check for drug-drug interactions and how to perform fibrosis assessments. After these providers go through this training, they become more comfortable giving treatment on their own.
The second project, which I have also been fortunate to be involved in, is led by my colleague Dr. Jeffrey Weiss at Mount Sinai Hospital and is located at a syringe exchange program in a Brooklyn, New York. Here, patients attend receive in-person and/or telemedicine-based HCV treatment, which is a new model of care for us. While it has produced a different set of challenges in terms of engaging and bringing treatment to patients in a new space, it has been a great way to meet our objectives of helping patients to be treated where they are comfortable accessing care and services.
Q: Has the pandemic created any new challenges in treating at risk or special populations?
Dr. Perumalswami: The pandemic presented many new challenges. The primary impact that COVID-19 has had on our patients has been with the disruption in care; particularly for those patients who already found it challenging to seek and receive care. For patients who benefitted from following a routine, other pandemic-related challenges were the restrictions placed on our practices, and the reduced hours patients had to contend with access services and treatments at places such as syringe exchange programs or methadone programs.
Many of our patients have expressed feeling isolated as they are not able to get the same type of support that they were previously receiving. The decreases in viral hepatitis outreach, in screening in the community, and in practices resulted in a decrease in diagnosis and treatment.
We have also heard numerous discussions with regards to better reimbursements for phone call and telehealth sessions, but we must recognize that those things are not accessible to all patients. Many of our most vulnerable populations, do not have working phones, stable housing, or smart devices to access telehealth, so while there have been technological advances that can provide access to care and better reimbursement procedures, there are still many limitations that our patients are facing.
(AGA applauds researchers who are working to raise our awareness of health disparities in digestive diseases. AGA is committed to addressing this important societal issue head on. Learn more about AGA’s commitment through the AGA Equity Project).
The influence of uterine fibroids on fertility in women planning to become pregnant
Q1: How do/can fibroids influence fertility?
When considering how uterine fibroids influence fertility, it's important to understand that uterine fibroids are very common. Uterine fibroids are the most common pelvic tumor in women, and they're non-cancerous tumors that are developed from the muscle cells of the uterus. The lifetime risk, before the age of 50, of a woman having fibroids varies by race and ethnicity, but in general, about 80% of Black women and 70% of Caucasian women will have at least one uterine fibroid diagnosed before the age of 50.
It's also important to understand when considering fertility that the prevalence of uterine fibroids increases as someone gets older. So uterine fibroids are much less common in younger women in their 20s as they are in women in their upper 30s and 40s. That's important to understand when looking at fertility because we also know that with age, fertility decreases. Thus, uterine fibroids also can impact fertility. There's also this age-related factor, which makes it difficult to really look at fibroids as far as being a causative agent for infertility.
We do know that approximately 10% of women with infertility will be diagnosed with uterine fibroids during their evaluation, and there's multiple ways that uterine fibroids impact fertility. In general, it's going to depend on the location of the uterine fibroids, the size and the bulk or the number of uterine fibroids that a woman has. But when we look at the ways that uterine fibroids can impact fertility, what they can do is they distort the uterine cavity. This is the most common for submucosal fibroids or fibroids that have a component that's present inside the uterine cavity. Fibroids that are submucosal or intramural fibroids are in the muscle of the uterus and have an intracavitary component. They're well-known to distort the uterine cavity and that can impact implantation of an embryo. There's also thought that it can impact an ongoing pregnancy.
There's speculation that uterine fibroids can impact the blood flow to a pregnancy as well and they may impact fertility. Depending on the size of the uterine fibroid, they may block the fallopian tubes. And so, if you have a uterine fibroid that's in the corner of the uterus, that could cause a tubal factor type of infertility where there's occlusion of the fallopian tube. But in general, the most concern we have for uterine fibroids is how those fibroids impact the uterine lining and implantation of an embryo, and it's thought that those are most likely due to submucosal fibroids, or some intramural fibroids that may be particularly large, or that have a component that's inside the cavity.
Q2. Several studies have attempted to clarify the influence of fibroids on fertility, however, there have been various, sometimes contradictory findings and a lack of well-designed trials. Why is this?
One of the challenges in counseling patients regarding uterine fibroids is that there's really a lack of high-quality studies assessing uterine fibroids and fertility. And we all know that the gold standard research study is a randomized control trial, as they provide the highest level of evidence, but those are very difficult to conduct especially for women with uterine fibroids, as many women will decline randomization.
It's difficult to design a study where there's one treatment that can be beneficial versus no treatment. That's one challenge. Because of that, the study designs that we've had to date have mostly been retrospective, and there's been some observational studies. But even those studies, unfortunately, are complicated by the fact that fibroids themselves are very heterogeneous. It's a very heterogeneous condition. There's a lot of difference between the size of the uterine fibroids, the location and the bulk of the fibroid, and then there's also going to be the issue with age. If you have a woman who's older with uterine fibroids, obviously her age is also going to impact her fertility. We know that women with uterine fibroids tend to be older and that also impacts fertility. So that's going to have an impact on any research as well.
What we do know from some of the research to date is that it's well-known that submucosal fibroids impair fertility, that's well established. We do know that subserosal fibroids or the fibroids on the surface of the uterus do not impact uterine fibroids. The question that really hasn't been answered because there hasn't been adequate research and there's just not enough data of high quality is, whether intramural fibroids or fibroids inside the uterine muscle, whether they impact fertility.
Many women who have intramural uterine fibroids are asymptomatic. They don't have symptoms at all. So, the question is whether a woman should undergo an invasive procedure to remove that fibroid and if it’s going to help or not? That's one of the questions that we just don't have enough adequate research on because there are some limitations in the literature.
Q3. What are the current treatments, both surgical and nonsurgical, for patients with fibroids who may want to become pregnant?
I think if there's a patient, a woman with uterine fibroids who's interested in fertility, she may be a patient who is diagnosed with symptomatic fibroids, who wants to preserve her fertility, or she may be a patient who's an infertility patient who during her fertility evaluation discovers she has fibroids. It is important to determine whether treatment is appropriate for that patient, and as we just discussed, there's not a lot of answers in the literature for some patients. I think the most important thing to do first before deciding on a treatment is to determine the best type of treatment. At Johns Hopkins, for many of our patients with uterine fibroids, they'll undergo a pelvic MRI because the pelvic MRI can provide the most detailed information regarding the size and exact location of the uterine fibroids.
We then have a multidisciplinary conference every two weeks where we review the MRIs with a group of minimally invasive surgeons, interventional radiologists, and fertility specialist where we can really decide the best treatment for the individual patient. In deciding on a patient, it's important to make the right decision and have the most information. So as far as treatments that are available, for women who are wanting to preserve their fertility or planning to get pregnant very soon, the most common options are going to be surgical.
The least invasive surgical treatment would be a hysteroscopic myomectomy where we would do a hysteroscopy and remove the uterine fibroids by either shaving the pieces of the submucosal fibroid or we can remove it with a hysteroscopic morcellator. There are various techniques. But for the submucosal fibroids that are inside the uterine cavity, hysteroscopic myomectomy is very minimally invasive. It's an outpatient procedure. It's very safe and it's something that we will typically offer to patients who have submucosal fibroids.
For patients who have symptomatic uterine fibroids and may have bulk symptoms, or have numerous uterine fibroids, we typically would recommend either a laparoscopic robotic-assisted myomectomy, sometimes just a laparoscopic myomectomy, or for women who have the most severe, a very large fibroid uterus, let's say greater than 20 centimeters, they may actually need to undergo an exploratory laparotomy or abdominal myomectomy. For patients who have symptomatic subserosal fibroids and large intramural fibroids that need to be removed, it really depends on the size, location, and bulk of the uterine fibroids. And that's where the pelvic MRI becomes very useful.
I would say that for the majority of my patients that have a large amount of fibroids, are still able to undergo a robotic-assisted laparoscopic hysterectomy which oftentimes can be an outpatient procedure just because we've had this improvement in technology with robotic and laparoscopic surgery. But surgery can be very beneficial as far as removing the bulk of the uterine fibroids. And so that is typically our treatments that we would recommend for those who want future fertility or who are imminently trying to get pregnant.
There are medical treatments as well or non-surgical treatments such as GnRH analogs that can shrink the size of the uterine fibroids. Unfortunately, the uterine fibroids are still there and typically will still impact fertility. So that's not something that we do often for those that are actively trying to get pregnant. The same for uterine artery embolization or uterine fibroids embolization. We will not recommend that for patients who want to have future fertility because the fibroids will still be in that location and they're typically in a location that's impairing fertility.
Q4. How long do patients have to wait after a fibroid treatment to try to get pregnant?
The length of time that a patient needs to wait after having fibroids removed for surgical treatment typically depends on the type of surgery the patient undergoes as well as the size of the fibroids and the extent of the surgery. For a patient who's undergoing a hysteroscopic myomectomy, they typically only must wait a month or two. Once they're assessed that there's no residual fibroid that's left, then they can try to conceive.
For patients who need to undergo abdominal myomectomy or laparoscopic myomectomy, those are much more extensive procedures. Typically, surgeons will recommend a patient wait three to six months to try to conceive. It's also important for the surgeon to discuss with the patient the extent of the myomectomy and whether that patient, when she does become pregnant, will require a c-section because typically if the uterine cavity is entered or if there are multiple incisions on the uterus during the myomectomy surgery, surgeons will recommend a c-section for that patient when she does become pregnant to decrease the risk of uterine rupture. And typically, that will be documented in the operative note, but the surgeon will also counsel the patient regarding this.
Q1: How do/can fibroids influence fertility?
When considering how uterine fibroids influence fertility, it's important to understand that uterine fibroids are very common. Uterine fibroids are the most common pelvic tumor in women, and they're non-cancerous tumors that are developed from the muscle cells of the uterus. The lifetime risk, before the age of 50, of a woman having fibroids varies by race and ethnicity, but in general, about 80% of Black women and 70% of Caucasian women will have at least one uterine fibroid diagnosed before the age of 50.
It's also important to understand when considering fertility that the prevalence of uterine fibroids increases as someone gets older. So uterine fibroids are much less common in younger women in their 20s as they are in women in their upper 30s and 40s. That's important to understand when looking at fertility because we also know that with age, fertility decreases. Thus, uterine fibroids also can impact fertility. There's also this age-related factor, which makes it difficult to really look at fibroids as far as being a causative agent for infertility.
We do know that approximately 10% of women with infertility will be diagnosed with uterine fibroids during their evaluation, and there's multiple ways that uterine fibroids impact fertility. In general, it's going to depend on the location of the uterine fibroids, the size and the bulk or the number of uterine fibroids that a woman has. But when we look at the ways that uterine fibroids can impact fertility, what they can do is they distort the uterine cavity. This is the most common for submucosal fibroids or fibroids that have a component that's present inside the uterine cavity. Fibroids that are submucosal or intramural fibroids are in the muscle of the uterus and have an intracavitary component. They're well-known to distort the uterine cavity and that can impact implantation of an embryo. There's also thought that it can impact an ongoing pregnancy.
There's speculation that uterine fibroids can impact the blood flow to a pregnancy as well and they may impact fertility. Depending on the size of the uterine fibroid, they may block the fallopian tubes. And so, if you have a uterine fibroid that's in the corner of the uterus, that could cause a tubal factor type of infertility where there's occlusion of the fallopian tube. But in general, the most concern we have for uterine fibroids is how those fibroids impact the uterine lining and implantation of an embryo, and it's thought that those are most likely due to submucosal fibroids, or some intramural fibroids that may be particularly large, or that have a component that's inside the cavity.
Q2. Several studies have attempted to clarify the influence of fibroids on fertility, however, there have been various, sometimes contradictory findings and a lack of well-designed trials. Why is this?
One of the challenges in counseling patients regarding uterine fibroids is that there's really a lack of high-quality studies assessing uterine fibroids and fertility. And we all know that the gold standard research study is a randomized control trial, as they provide the highest level of evidence, but those are very difficult to conduct especially for women with uterine fibroids, as many women will decline randomization.
It's difficult to design a study where there's one treatment that can be beneficial versus no treatment. That's one challenge. Because of that, the study designs that we've had to date have mostly been retrospective, and there's been some observational studies. But even those studies, unfortunately, are complicated by the fact that fibroids themselves are very heterogeneous. It's a very heterogeneous condition. There's a lot of difference between the size of the uterine fibroids, the location and the bulk of the fibroid, and then there's also going to be the issue with age. If you have a woman who's older with uterine fibroids, obviously her age is also going to impact her fertility. We know that women with uterine fibroids tend to be older and that also impacts fertility. So that's going to have an impact on any research as well.
What we do know from some of the research to date is that it's well-known that submucosal fibroids impair fertility, that's well established. We do know that subserosal fibroids or the fibroids on the surface of the uterus do not impact uterine fibroids. The question that really hasn't been answered because there hasn't been adequate research and there's just not enough data of high quality is, whether intramural fibroids or fibroids inside the uterine muscle, whether they impact fertility.
Many women who have intramural uterine fibroids are asymptomatic. They don't have symptoms at all. So, the question is whether a woman should undergo an invasive procedure to remove that fibroid and if it’s going to help or not? That's one of the questions that we just don't have enough adequate research on because there are some limitations in the literature.
Q3. What are the current treatments, both surgical and nonsurgical, for patients with fibroids who may want to become pregnant?
I think if there's a patient, a woman with uterine fibroids who's interested in fertility, she may be a patient who is diagnosed with symptomatic fibroids, who wants to preserve her fertility, or she may be a patient who's an infertility patient who during her fertility evaluation discovers she has fibroids. It is important to determine whether treatment is appropriate for that patient, and as we just discussed, there's not a lot of answers in the literature for some patients. I think the most important thing to do first before deciding on a treatment is to determine the best type of treatment. At Johns Hopkins, for many of our patients with uterine fibroids, they'll undergo a pelvic MRI because the pelvic MRI can provide the most detailed information regarding the size and exact location of the uterine fibroids.
We then have a multidisciplinary conference every two weeks where we review the MRIs with a group of minimally invasive surgeons, interventional radiologists, and fertility specialist where we can really decide the best treatment for the individual patient. In deciding on a patient, it's important to make the right decision and have the most information. So as far as treatments that are available, for women who are wanting to preserve their fertility or planning to get pregnant very soon, the most common options are going to be surgical.
The least invasive surgical treatment would be a hysteroscopic myomectomy where we would do a hysteroscopy and remove the uterine fibroids by either shaving the pieces of the submucosal fibroid or we can remove it with a hysteroscopic morcellator. There are various techniques. But for the submucosal fibroids that are inside the uterine cavity, hysteroscopic myomectomy is very minimally invasive. It's an outpatient procedure. It's very safe and it's something that we will typically offer to patients who have submucosal fibroids.
For patients who have symptomatic uterine fibroids and may have bulk symptoms, or have numerous uterine fibroids, we typically would recommend either a laparoscopic robotic-assisted myomectomy, sometimes just a laparoscopic myomectomy, or for women who have the most severe, a very large fibroid uterus, let's say greater than 20 centimeters, they may actually need to undergo an exploratory laparotomy or abdominal myomectomy. For patients who have symptomatic subserosal fibroids and large intramural fibroids that need to be removed, it really depends on the size, location, and bulk of the uterine fibroids. And that's where the pelvic MRI becomes very useful.
I would say that for the majority of my patients that have a large amount of fibroids, are still able to undergo a robotic-assisted laparoscopic hysterectomy which oftentimes can be an outpatient procedure just because we've had this improvement in technology with robotic and laparoscopic surgery. But surgery can be very beneficial as far as removing the bulk of the uterine fibroids. And so that is typically our treatments that we would recommend for those who want future fertility or who are imminently trying to get pregnant.
There are medical treatments as well or non-surgical treatments such as GnRH analogs that can shrink the size of the uterine fibroids. Unfortunately, the uterine fibroids are still there and typically will still impact fertility. So that's not something that we do often for those that are actively trying to get pregnant. The same for uterine artery embolization or uterine fibroids embolization. We will not recommend that for patients who want to have future fertility because the fibroids will still be in that location and they're typically in a location that's impairing fertility.
Q4. How long do patients have to wait after a fibroid treatment to try to get pregnant?
The length of time that a patient needs to wait after having fibroids removed for surgical treatment typically depends on the type of surgery the patient undergoes as well as the size of the fibroids and the extent of the surgery. For a patient who's undergoing a hysteroscopic myomectomy, they typically only must wait a month or two. Once they're assessed that there's no residual fibroid that's left, then they can try to conceive.
For patients who need to undergo abdominal myomectomy or laparoscopic myomectomy, those are much more extensive procedures. Typically, surgeons will recommend a patient wait three to six months to try to conceive. It's also important for the surgeon to discuss with the patient the extent of the myomectomy and whether that patient, when she does become pregnant, will require a c-section because typically if the uterine cavity is entered or if there are multiple incisions on the uterus during the myomectomy surgery, surgeons will recommend a c-section for that patient when she does become pregnant to decrease the risk of uterine rupture. And typically, that will be documented in the operative note, but the surgeon will also counsel the patient regarding this.
Q1: How do/can fibroids influence fertility?
When considering how uterine fibroids influence fertility, it's important to understand that uterine fibroids are very common. Uterine fibroids are the most common pelvic tumor in women, and they're non-cancerous tumors that are developed from the muscle cells of the uterus. The lifetime risk, before the age of 50, of a woman having fibroids varies by race and ethnicity, but in general, about 80% of Black women and 70% of Caucasian women will have at least one uterine fibroid diagnosed before the age of 50.
It's also important to understand when considering fertility that the prevalence of uterine fibroids increases as someone gets older. So uterine fibroids are much less common in younger women in their 20s as they are in women in their upper 30s and 40s. That's important to understand when looking at fertility because we also know that with age, fertility decreases. Thus, uterine fibroids also can impact fertility. There's also this age-related factor, which makes it difficult to really look at fibroids as far as being a causative agent for infertility.
We do know that approximately 10% of women with infertility will be diagnosed with uterine fibroids during their evaluation, and there's multiple ways that uterine fibroids impact fertility. In general, it's going to depend on the location of the uterine fibroids, the size and the bulk or the number of uterine fibroids that a woman has. But when we look at the ways that uterine fibroids can impact fertility, what they can do is they distort the uterine cavity. This is the most common for submucosal fibroids or fibroids that have a component that's present inside the uterine cavity. Fibroids that are submucosal or intramural fibroids are in the muscle of the uterus and have an intracavitary component. They're well-known to distort the uterine cavity and that can impact implantation of an embryo. There's also thought that it can impact an ongoing pregnancy.
There's speculation that uterine fibroids can impact the blood flow to a pregnancy as well and they may impact fertility. Depending on the size of the uterine fibroid, they may block the fallopian tubes. And so, if you have a uterine fibroid that's in the corner of the uterus, that could cause a tubal factor type of infertility where there's occlusion of the fallopian tube. But in general, the most concern we have for uterine fibroids is how those fibroids impact the uterine lining and implantation of an embryo, and it's thought that those are most likely due to submucosal fibroids, or some intramural fibroids that may be particularly large, or that have a component that's inside the cavity.
Q2. Several studies have attempted to clarify the influence of fibroids on fertility, however, there have been various, sometimes contradictory findings and a lack of well-designed trials. Why is this?
One of the challenges in counseling patients regarding uterine fibroids is that there's really a lack of high-quality studies assessing uterine fibroids and fertility. And we all know that the gold standard research study is a randomized control trial, as they provide the highest level of evidence, but those are very difficult to conduct especially for women with uterine fibroids, as many women will decline randomization.
It's difficult to design a study where there's one treatment that can be beneficial versus no treatment. That's one challenge. Because of that, the study designs that we've had to date have mostly been retrospective, and there's been some observational studies. But even those studies, unfortunately, are complicated by the fact that fibroids themselves are very heterogeneous. It's a very heterogeneous condition. There's a lot of difference between the size of the uterine fibroids, the location and the bulk of the fibroid, and then there's also going to be the issue with age. If you have a woman who's older with uterine fibroids, obviously her age is also going to impact her fertility. We know that women with uterine fibroids tend to be older and that also impacts fertility. So that's going to have an impact on any research as well.
What we do know from some of the research to date is that it's well-known that submucosal fibroids impair fertility, that's well established. We do know that subserosal fibroids or the fibroids on the surface of the uterus do not impact uterine fibroids. The question that really hasn't been answered because there hasn't been adequate research and there's just not enough data of high quality is, whether intramural fibroids or fibroids inside the uterine muscle, whether they impact fertility.
Many women who have intramural uterine fibroids are asymptomatic. They don't have symptoms at all. So, the question is whether a woman should undergo an invasive procedure to remove that fibroid and if it’s going to help or not? That's one of the questions that we just don't have enough adequate research on because there are some limitations in the literature.
Q3. What are the current treatments, both surgical and nonsurgical, for patients with fibroids who may want to become pregnant?
I think if there's a patient, a woman with uterine fibroids who's interested in fertility, she may be a patient who is diagnosed with symptomatic fibroids, who wants to preserve her fertility, or she may be a patient who's an infertility patient who during her fertility evaluation discovers she has fibroids. It is important to determine whether treatment is appropriate for that patient, and as we just discussed, there's not a lot of answers in the literature for some patients. I think the most important thing to do first before deciding on a treatment is to determine the best type of treatment. At Johns Hopkins, for many of our patients with uterine fibroids, they'll undergo a pelvic MRI because the pelvic MRI can provide the most detailed information regarding the size and exact location of the uterine fibroids.
We then have a multidisciplinary conference every two weeks where we review the MRIs with a group of minimally invasive surgeons, interventional radiologists, and fertility specialist where we can really decide the best treatment for the individual patient. In deciding on a patient, it's important to make the right decision and have the most information. So as far as treatments that are available, for women who are wanting to preserve their fertility or planning to get pregnant very soon, the most common options are going to be surgical.
The least invasive surgical treatment would be a hysteroscopic myomectomy where we would do a hysteroscopy and remove the uterine fibroids by either shaving the pieces of the submucosal fibroid or we can remove it with a hysteroscopic morcellator. There are various techniques. But for the submucosal fibroids that are inside the uterine cavity, hysteroscopic myomectomy is very minimally invasive. It's an outpatient procedure. It's very safe and it's something that we will typically offer to patients who have submucosal fibroids.
For patients who have symptomatic uterine fibroids and may have bulk symptoms, or have numerous uterine fibroids, we typically would recommend either a laparoscopic robotic-assisted myomectomy, sometimes just a laparoscopic myomectomy, or for women who have the most severe, a very large fibroid uterus, let's say greater than 20 centimeters, they may actually need to undergo an exploratory laparotomy or abdominal myomectomy. For patients who have symptomatic subserosal fibroids and large intramural fibroids that need to be removed, it really depends on the size, location, and bulk of the uterine fibroids. And that's where the pelvic MRI becomes very useful.
I would say that for the majority of my patients that have a large amount of fibroids, are still able to undergo a robotic-assisted laparoscopic hysterectomy which oftentimes can be an outpatient procedure just because we've had this improvement in technology with robotic and laparoscopic surgery. But surgery can be very beneficial as far as removing the bulk of the uterine fibroids. And so that is typically our treatments that we would recommend for those who want future fertility or who are imminently trying to get pregnant.
There are medical treatments as well or non-surgical treatments such as GnRH analogs that can shrink the size of the uterine fibroids. Unfortunately, the uterine fibroids are still there and typically will still impact fertility. So that's not something that we do often for those that are actively trying to get pregnant. The same for uterine artery embolization or uterine fibroids embolization. We will not recommend that for patients who want to have future fertility because the fibroids will still be in that location and they're typically in a location that's impairing fertility.
Q4. How long do patients have to wait after a fibroid treatment to try to get pregnant?
The length of time that a patient needs to wait after having fibroids removed for surgical treatment typically depends on the type of surgery the patient undergoes as well as the size of the fibroids and the extent of the surgery. For a patient who's undergoing a hysteroscopic myomectomy, they typically only must wait a month or two. Once they're assessed that there's no residual fibroid that's left, then they can try to conceive.
For patients who need to undergo abdominal myomectomy or laparoscopic myomectomy, those are much more extensive procedures. Typically, surgeons will recommend a patient wait three to six months to try to conceive. It's also important for the surgeon to discuss with the patient the extent of the myomectomy and whether that patient, when she does become pregnant, will require a c-section because typically if the uterine cavity is entered or if there are multiple incisions on the uterus during the myomectomy surgery, surgeons will recommend a c-section for that patient when she does become pregnant to decrease the risk of uterine rupture. And typically, that will be documented in the operative note, but the surgeon will also counsel the patient regarding this.