User login
GLIMMER of hope for itch in primary biliary cholangitis
Patients with primary biliary cholangitis experienced rapid improvements in itch and quality of life after treatment with linerixibat in a randomized, placebo-controlled trial of the safety, efficacy, and tolerability of the small-molecule drug.
Moderate to severe pruritus “affects patients’ quality of life and is a huge burden for them,” said investigator Cynthia Levy, MD, from the University of Miami Health System.
“Finally having a medication that controls those symptoms is really important,” she said in an interview.
With a twice-daily mid-range dose of the drug for 12 weeks, patients with moderate to severe itch reported significantly less itch and better social and emotional quality of life, Dr. Levy reported at the Liver Meeting, where she presented findings from the phase 2 GLIMMER trial.
After a single-blind 4-week placebo run-in period for patients with itch scores of at least 4 on a 10-point rating scale, those with itch scores of at least 3 were then randomly assigned to one of five treatment regimens – once-daily linerixibat at doses of 20 mg, 90 mg, or 180 mg, or twice-daily doses of 40 mg or 90 mg – or to placebo.
After 12 weeks of treatment, all 147 participants once again received placebo for 4 weeks.
During the trial, participants recorded itch levels twice daily. The worst of these daily scores was averaged every 7 days to determine the mean worst daily itch.
The primary study endpoint was the change in worst daily itch from baseline after 12 weeks of treatment. Participants whose self-rated itch improved by 2 points on the 10-point scale were considered to have had a response to the drug.
Participants also completed the PBC-40, an instrument to measure quality of life in patients with primary biliary cholangitis, answering questions about itch and social and emotional status.
Reductions in worst daily itch from baseline to 12 weeks were steepest in the 40-mg twice-daily group, at 2.86 points, and in the 90-mg twice-daily group, at 2.25 points. In the placebo group, the mean decrease was 1.73 points.
During the subsequent 4 weeks of placebo, after treatment ended, the itch relief faded in all groups.
Scores on the PBC-40 itch domain improved significantly in every group, including placebo. However, only those in the twice-daily 40-mg group saw significant improvements on the social (P = .0016) and emotional (P = .0025) domains.
‘Between incremental and revolutionary’
The results are on a “kind of continuum between incremental and revolutionary,” said Jonathan A. Dranoff, MD, from the University of Arkansas for Medical Sciences, Little Rock, who was not involved in the study. “It doesn’t hit either extreme, but it’s the first new drug for this purpose in forever, which by itself is a good thing.”
The placebo effect suggests that “maybe the actual contribution of the noncognitive brain to pruritus is bigger than we thought, and that’s worth noting,” he added. Nevertheless, “the drug still appears to have effects that are statistically different from placebo.”
The placebo effect in itching studies is always high but tends to wane over time, said Dr. Levy. This trial had a 4-week placebo run-in period to allow that effect to fade somewhat, she explained.
About 10% of the study cohort experienced drug-related diarrhea, which was expected, and about 10% dropped out of the trial because of drug-related adverse events.
Linerixibat is an ileal sodium-dependent bile acid transporter inhibitor, so the gut has to deal with the excess bile acid fallout, but the diarrhea is likely manageable with antidiarrheals, said Dr. Levy.
It is unlikely that diarrhea will deter patients with severe itch from using an effective drug when other drugs have failed them. “These patients are consumed by itch most of the time,” said Dr. Dranoff. “I think for people who don’t regularly treat patients with primary biliary cholangitis, it’s one of the underappreciated aspects of the disease.”
The improvements in social and emotional quality of life seen with linerixibat are not only statistically significant, they are also clinically significant, said Dr. Levy. “We are really expecting this to impact the lives of our patients and are looking forward to phase 3.”
Dr. Levy disclosed support from GlaxoSmithKline. Dr. Dranoff disclosed no relevant financial relationships.
This article first appeared on Medscape.com.
Patients with primary biliary cholangitis experienced rapid improvements in itch and quality of life after treatment with linerixibat in a randomized, placebo-controlled trial of the safety, efficacy, and tolerability of the small-molecule drug.
Moderate to severe pruritus “affects patients’ quality of life and is a huge burden for them,” said investigator Cynthia Levy, MD, from the University of Miami Health System.
“Finally having a medication that controls those symptoms is really important,” she said in an interview.
With a twice-daily mid-range dose of the drug for 12 weeks, patients with moderate to severe itch reported significantly less itch and better social and emotional quality of life, Dr. Levy reported at the Liver Meeting, where she presented findings from the phase 2 GLIMMER trial.
After a single-blind 4-week placebo run-in period for patients with itch scores of at least 4 on a 10-point rating scale, those with itch scores of at least 3 were then randomly assigned to one of five treatment regimens – once-daily linerixibat at doses of 20 mg, 90 mg, or 180 mg, or twice-daily doses of 40 mg or 90 mg – or to placebo.
After 12 weeks of treatment, all 147 participants once again received placebo for 4 weeks.
During the trial, participants recorded itch levels twice daily. The worst of these daily scores was averaged every 7 days to determine the mean worst daily itch.
The primary study endpoint was the change in worst daily itch from baseline after 12 weeks of treatment. Participants whose self-rated itch improved by 2 points on the 10-point scale were considered to have had a response to the drug.
Participants also completed the PBC-40, an instrument to measure quality of life in patients with primary biliary cholangitis, answering questions about itch and social and emotional status.
Reductions in worst daily itch from baseline to 12 weeks were steepest in the 40-mg twice-daily group, at 2.86 points, and in the 90-mg twice-daily group, at 2.25 points. In the placebo group, the mean decrease was 1.73 points.
During the subsequent 4 weeks of placebo, after treatment ended, the itch relief faded in all groups.
Scores on the PBC-40 itch domain improved significantly in every group, including placebo. However, only those in the twice-daily 40-mg group saw significant improvements on the social (P = .0016) and emotional (P = .0025) domains.
‘Between incremental and revolutionary’
The results are on a “kind of continuum between incremental and revolutionary,” said Jonathan A. Dranoff, MD, from the University of Arkansas for Medical Sciences, Little Rock, who was not involved in the study. “It doesn’t hit either extreme, but it’s the first new drug for this purpose in forever, which by itself is a good thing.”
The placebo effect suggests that “maybe the actual contribution of the noncognitive brain to pruritus is bigger than we thought, and that’s worth noting,” he added. Nevertheless, “the drug still appears to have effects that are statistically different from placebo.”
The placebo effect in itching studies is always high but tends to wane over time, said Dr. Levy. This trial had a 4-week placebo run-in period to allow that effect to fade somewhat, she explained.
About 10% of the study cohort experienced drug-related diarrhea, which was expected, and about 10% dropped out of the trial because of drug-related adverse events.
Linerixibat is an ileal sodium-dependent bile acid transporter inhibitor, so the gut has to deal with the excess bile acid fallout, but the diarrhea is likely manageable with antidiarrheals, said Dr. Levy.
It is unlikely that diarrhea will deter patients with severe itch from using an effective drug when other drugs have failed them. “These patients are consumed by itch most of the time,” said Dr. Dranoff. “I think for people who don’t regularly treat patients with primary biliary cholangitis, it’s one of the underappreciated aspects of the disease.”
The improvements in social and emotional quality of life seen with linerixibat are not only statistically significant, they are also clinically significant, said Dr. Levy. “We are really expecting this to impact the lives of our patients and are looking forward to phase 3.”
Dr. Levy disclosed support from GlaxoSmithKline. Dr. Dranoff disclosed no relevant financial relationships.
This article first appeared on Medscape.com.
Patients with primary biliary cholangitis experienced rapid improvements in itch and quality of life after treatment with linerixibat in a randomized, placebo-controlled trial of the safety, efficacy, and tolerability of the small-molecule drug.
Moderate to severe pruritus “affects patients’ quality of life and is a huge burden for them,” said investigator Cynthia Levy, MD, from the University of Miami Health System.
“Finally having a medication that controls those symptoms is really important,” she said in an interview.
With a twice-daily mid-range dose of the drug for 12 weeks, patients with moderate to severe itch reported significantly less itch and better social and emotional quality of life, Dr. Levy reported at the Liver Meeting, where she presented findings from the phase 2 GLIMMER trial.
After a single-blind 4-week placebo run-in period for patients with itch scores of at least 4 on a 10-point rating scale, those with itch scores of at least 3 were then randomly assigned to one of five treatment regimens – once-daily linerixibat at doses of 20 mg, 90 mg, or 180 mg, or twice-daily doses of 40 mg or 90 mg – or to placebo.
After 12 weeks of treatment, all 147 participants once again received placebo for 4 weeks.
During the trial, participants recorded itch levels twice daily. The worst of these daily scores was averaged every 7 days to determine the mean worst daily itch.
The primary study endpoint was the change in worst daily itch from baseline after 12 weeks of treatment. Participants whose self-rated itch improved by 2 points on the 10-point scale were considered to have had a response to the drug.
Participants also completed the PBC-40, an instrument to measure quality of life in patients with primary biliary cholangitis, answering questions about itch and social and emotional status.
Reductions in worst daily itch from baseline to 12 weeks were steepest in the 40-mg twice-daily group, at 2.86 points, and in the 90-mg twice-daily group, at 2.25 points. In the placebo group, the mean decrease was 1.73 points.
During the subsequent 4 weeks of placebo, after treatment ended, the itch relief faded in all groups.
Scores on the PBC-40 itch domain improved significantly in every group, including placebo. However, only those in the twice-daily 40-mg group saw significant improvements on the social (P = .0016) and emotional (P = .0025) domains.
‘Between incremental and revolutionary’
The results are on a “kind of continuum between incremental and revolutionary,” said Jonathan A. Dranoff, MD, from the University of Arkansas for Medical Sciences, Little Rock, who was not involved in the study. “It doesn’t hit either extreme, but it’s the first new drug for this purpose in forever, which by itself is a good thing.”
The placebo effect suggests that “maybe the actual contribution of the noncognitive brain to pruritus is bigger than we thought, and that’s worth noting,” he added. Nevertheless, “the drug still appears to have effects that are statistically different from placebo.”
The placebo effect in itching studies is always high but tends to wane over time, said Dr. Levy. This trial had a 4-week placebo run-in period to allow that effect to fade somewhat, she explained.
About 10% of the study cohort experienced drug-related diarrhea, which was expected, and about 10% dropped out of the trial because of drug-related adverse events.
Linerixibat is an ileal sodium-dependent bile acid transporter inhibitor, so the gut has to deal with the excess bile acid fallout, but the diarrhea is likely manageable with antidiarrheals, said Dr. Levy.
It is unlikely that diarrhea will deter patients with severe itch from using an effective drug when other drugs have failed them. “These patients are consumed by itch most of the time,” said Dr. Dranoff. “I think for people who don’t regularly treat patients with primary biliary cholangitis, it’s one of the underappreciated aspects of the disease.”
The improvements in social and emotional quality of life seen with linerixibat are not only statistically significant, they are also clinically significant, said Dr. Levy. “We are really expecting this to impact the lives of our patients and are looking forward to phase 3.”
Dr. Levy disclosed support from GlaxoSmithKline. Dr. Dranoff disclosed no relevant financial relationships.
This article first appeared on Medscape.com.
Oral Hairy Leukoplakia Associated With the Use of Adalimumab
To the Editor:
Oral hairy leukoplakia (OHL) is an Epstein-Barr virus (EBV)–mediated mucocutaneous disease that often involves the lingual epithelium. The lateral portions of the tongue are the most commonly affected sites. The lesions often are described as asymptomatic, white, corrugated patches or plaques that are unable to be scraped off.1 Oral hairy leukoplakia was first identified in 1984 and was considered to be associated with AIDS.2 An association between the presence of OHL and the degree of immunosuppression as well as the severity of human immunodeficiency virus (HIV) has been reported.3 Although OHL initially was considered to be pathognomonic for HIV, it has since been described in multiple other immunosuppressive conditions.4 Numerous medical conditions and combinations of immunosuppressive medications have been associated with OHL in patients who were HIV negative.5
Adalimumab is an injectable human IgG1 recombinant antibody to tumor necrosis factor α (TNF-α).6 It currently is approved by the US Food and Drug Administration for the treatment of rheumatoid arthritis, juvenile idiopathic arthritis, psoriatic arthritis, ankylosing spondylitis, adult and pediatric Crohn disease, ulcerative colitis, noninfectious uveitis, hidradenitis suppurativa, and plaque psoriasis.7 We report a case of OHL associated with the use of adalimumab.
A 47-year-old woman initially presented with chronic plaque-type psoriasis. Her medical history was notable for bipolar disorder, migraines, hypertension, and tobacco use. The patient’s psoriasis initially was well controlled on a regimen of topical steroids and methotrexate; however, methotrexate was stopped after 2.5 years due to a mildly elevated alanine aminotransferase level, as well as an abnormal liver biopsy showing mildly active (grade 1 of 3) steatohepatitis with portal chronic inflammation, pericellular fibrosis, and portal and focal periportal fibrosis (stage 1-2 of 4). The patient and her dermatologist were uncomfortable continuing methotrexate with these findings. After baseline screening including a negative purified protein derivative skin test, adalimumab was initiated. A loading dose of 80 mg subcutaneously (SQ) was given, followed by adalimumab 40 mg SQ 1 week later and 40 mg every other week as maintenance.
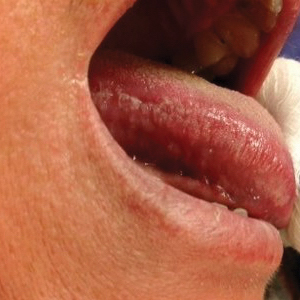
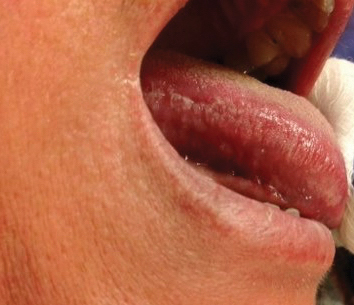
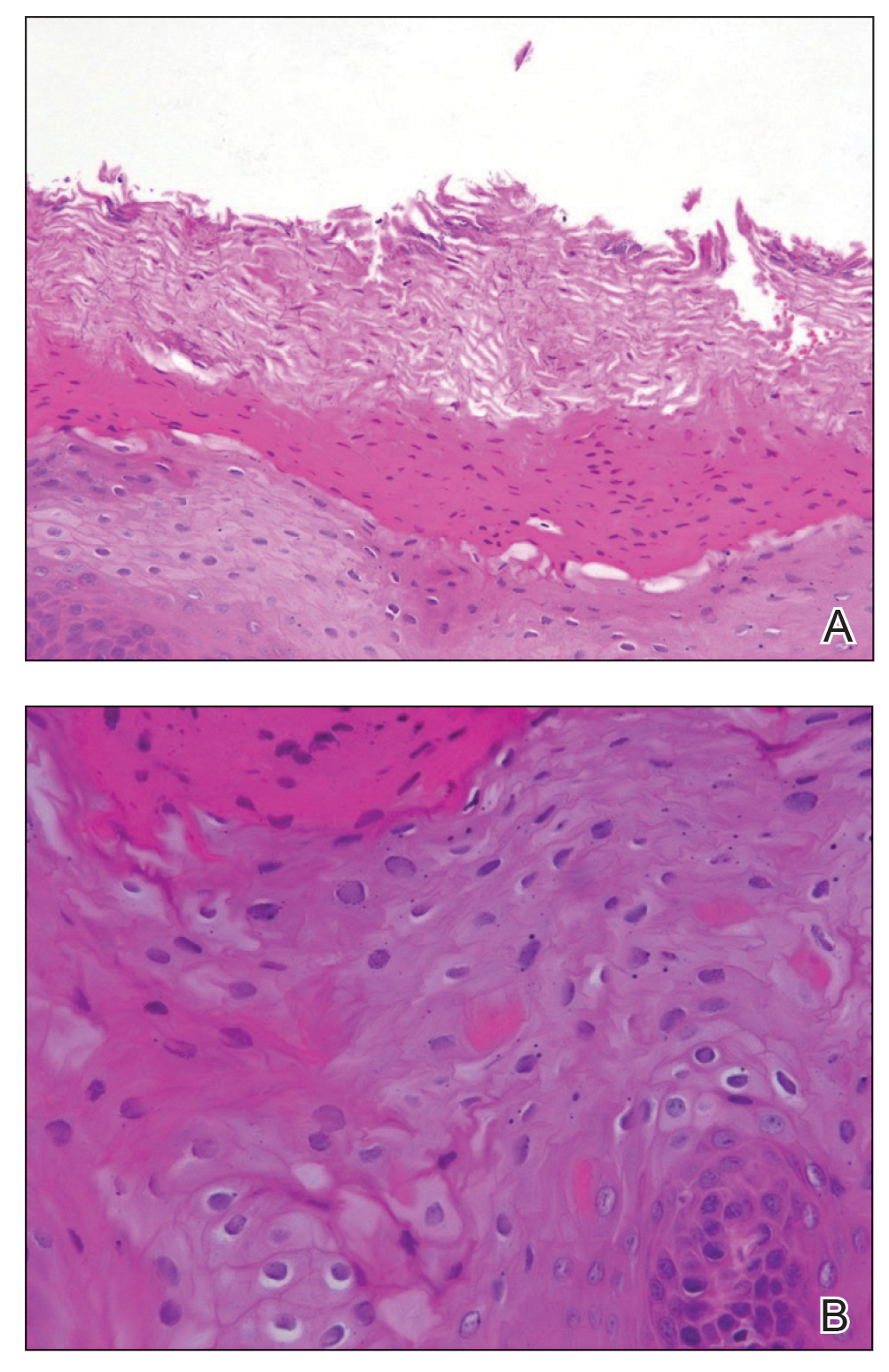
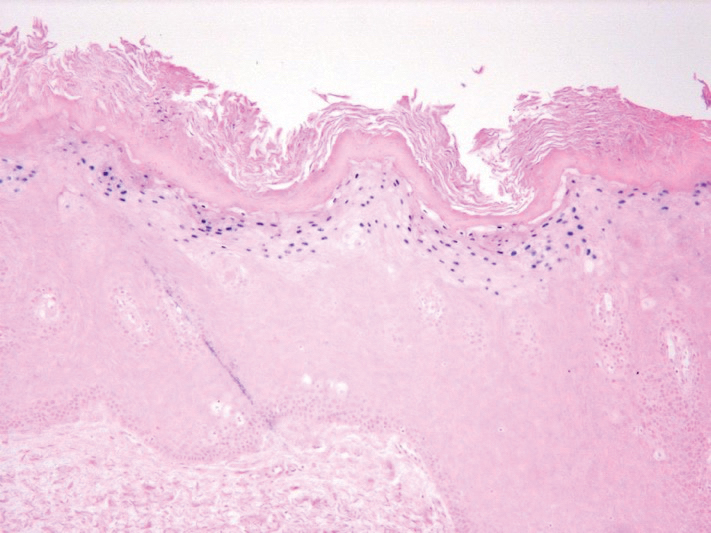
The patient’s psoriasis was well controlled with adalimumab for 22 months, but she then developed a thin white plaque on the right lateral tongue (Figure 1). An incisional biopsy of the tongue performed by an oral surgeon revealed hyperkeratosis with Candida colonization and viral cytopathic effect (Figure 2). An EBV DNA in situ hybridization stain revealed focal positivity within these cells (Figure 3), leading to a diagnosis of OHL. Laboratory evaluation demonstrated a normal complete blood cell count with differential and liver panel as well as a negative HIV test. The patient otherwise felt well and denied fevers, lymphadenopathy, and weight loss.
We consulted with an infectious disease and immunodeficiency specialist regarding the patient’s case. Before conducting further evaluation beyond HIV screening for immunodeficiency states, adalimumab was discontinued to see if the OHL would spontaneously resolve. Three months after discontinuation of adalimumab, the white plaque on the right lateral tongue was notably improved. The OHL continued to disappear and was completely resolved 1 year after discontinuation of adalimumab. The patient’s psoriasis had subsequently remained well controlled with diet and weight loss, smoking cessation, topical steroids, and apremilast without any recurrence of the OHL.
Oral hairy leukoplakia is associated with upregulated EBV replication and EBV-encoded proteins such as latent membrane protein 1.2 It often presents as white or gray patches on the lateral lingual margins with prominent folds and/or projections, giving a shaggy appearance. Oral hairy leukoplakia often is specific for HIV infection and rarely is associated with other immunodeficiencies.2 Prasad and Bilodeau5 performed a literature review of medical conditions and immunosuppressive medications associated with OHL in patients without HIV. Allogeneic transplant was associated with the highest incidence of OHL in HIV-negative patients (59.2% [45/76]).5 Various combinations of immunosuppressive medications (eg, prednisone, cyclosporine, azathioprine) also may be implicated in cases of HIV-negative patients with OHL. A case of OHL also has been reported with long-standing use of inhaled corticosteroids in an immunocompetent, HIV-negative patient.6 Another case was reported with long-term use of the aromatic antiepileptic lamotrigine, which resolved once stopping the medication.8 Although EBV is an oncovirus and has been associated with lymphoproliferative disorders and nasopharyngeal carcinoma, OHL is not considered to be a premalignant lesion.7 Despite the strong association between OHL and HIV, our patient was HIV negative. The only immunocompromising factor in our patient was the use of adalimumab to treat psoriasis. We did not conduct further testing for immunodeficiency states because the OHL spontaneously resolved when the adalimumab was discontinued.
PubMed and Ovid searches of articles indexed for MEDLINE using the terms adalimumab and oral hairy leukoplakia as well as TNF-alpha inhibitor and oral hairy leukoplakia with humans and English language as limitations revealed that no cases have been reported in the literature demonstrating an association between OHL and adalimumab or any other TNF-α inhibitor. However, Cetkovska et al9 reported a case of EBV hepatitis and subsequently chronic hepatitis as a complication of infliximab used for the treatment of chronic psoriasis. Because TNF-α and IFN-γ play an important role in controlling viral infections, there is an increased risk for reactivating a viral illness when depleting TNF through pharmacologic measures (ie, adalimumab, infliximab).8 Another case of EBV-associated plasmablastic lymphoma was reported after 1 year of adalimumab use in a patient with Crohn disease. The plasmablastic lymphoma resolved after 4 rounds of chemotherapy.10
The only contraindication for the use of adalimumab is a known hypersensitivity to the drug. Relative contraindications for use of adalimumab include active tuberculosis, demyelinating disease, hematologic diseases (ie, thrombocytopenia, pancytopenia), lymphoma, hepatitis C, and hepatitis B.11 The most common adverse effect of adalimumab is an injection-site reaction. Additional reported adverse effects of TNF-α inhibitors as a class are lymphoma, melanoma, nonmelanoma skin cancer, reactivation of latent tuberculosis, congestive heart failure, autoimmunity, and hematologic toxicity.11
This case demonstrates an association between adalimumab and OHL in an HIV-negative patient. Although the mechanism behind OHL and immunosuppression remains to be elucidated, this association is important to keep in mind when using adalimumab or other TNF-α inhibitors for the treatment of psoriasis or other medical conditions.
- Triantos D, Porter SR, Scully C, et al. Oral hairy leukoplakia: clinicopathologic features, pathogenesis, diagnosis, and clinical significance. Clin Infect Dis. 1997;25:1392-1396.
- Greenspan D, Greenspan JS, Conant M, et al. Oral “hairy” leucoplakia in male homosexuals: evidence of association with both papillomavirus and a herpes-group virus. Lancet. 1984;2:831-834.
- Glick M, Muzyka BC, Lurie D, et al. Oral manifestations associated with HIV-related disease as marks for immune suppression and AIDS. Oral Surg Oral Med Oral Pathol. 1994;77:344-349.
- Chambers AE, Conn B, Pemberton M, et al. Twenty-first-century oral hair leukoplakia—a non-HIV-associated entity. Oral Surg Oral Med Oral Patho Oral Radiol. 2015;119:326-332.
- Prasad JL, Bilodeau EA. Oral hairy leukoplakia in patients without HIV: presentation of 2 new cases. Oral Surg Oral Med Oral Pathol Oral Radiol. 2014;118:E151-E160.
- Moffat M, Jauhar S, Jones ME, et al. Oral hairy leukoplakia in an HIV-negative, immunocompetent patient. Oral Biosci Med. 2005;2:282-284.
- Greenspan JS, Greenspan D. Oral hairy leukoplakia: diagnosis and management. Oral Surg Oral Med Oral Pathol. 1989;67:396-403.
- Gordins P, Sloan P, Spickett GP, et al. Oral hairy leukoplakia in a patient on long-term anticonvulsant treatment with lamotrigine. Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 2011;111:E17-E23.
- Cetkovska P, Lomicova I, Mukensnabl P, et al. Anti-tumour necrosis factor treatment of severe psoriasis complicated by Epstein-Barr virus hepatitis and subsequently by chronic hepatitis. Dermatol Ther. 2015;28:369-372.
- Liu L, Charabaty A, Ozdemirli M. EBV-associated plasmablastic lymphoma in a patient with Crohn’s disease after adalimumab treatment. J Crohns Colitis. 2013;7:E118-E119.
- Humira [package insert]. North Chicago, IL: AbbVie Inc; 2018.
To the Editor:
Oral hairy leukoplakia (OHL) is an Epstein-Barr virus (EBV)–mediated mucocutaneous disease that often involves the lingual epithelium. The lateral portions of the tongue are the most commonly affected sites. The lesions often are described as asymptomatic, white, corrugated patches or plaques that are unable to be scraped off.1 Oral hairy leukoplakia was first identified in 1984 and was considered to be associated with AIDS.2 An association between the presence of OHL and the degree of immunosuppression as well as the severity of human immunodeficiency virus (HIV) has been reported.3 Although OHL initially was considered to be pathognomonic for HIV, it has since been described in multiple other immunosuppressive conditions.4 Numerous medical conditions and combinations of immunosuppressive medications have been associated with OHL in patients who were HIV negative.5
Adalimumab is an injectable human IgG1 recombinant antibody to tumor necrosis factor α (TNF-α).6 It currently is approved by the US Food and Drug Administration for the treatment of rheumatoid arthritis, juvenile idiopathic arthritis, psoriatic arthritis, ankylosing spondylitis, adult and pediatric Crohn disease, ulcerative colitis, noninfectious uveitis, hidradenitis suppurativa, and plaque psoriasis.7 We report a case of OHL associated with the use of adalimumab.
A 47-year-old woman initially presented with chronic plaque-type psoriasis. Her medical history was notable for bipolar disorder, migraines, hypertension, and tobacco use. The patient’s psoriasis initially was well controlled on a regimen of topical steroids and methotrexate; however, methotrexate was stopped after 2.5 years due to a mildly elevated alanine aminotransferase level, as well as an abnormal liver biopsy showing mildly active (grade 1 of 3) steatohepatitis with portal chronic inflammation, pericellular fibrosis, and portal and focal periportal fibrosis (stage 1-2 of 4). The patient and her dermatologist were uncomfortable continuing methotrexate with these findings. After baseline screening including a negative purified protein derivative skin test, adalimumab was initiated. A loading dose of 80 mg subcutaneously (SQ) was given, followed by adalimumab 40 mg SQ 1 week later and 40 mg every other week as maintenance.
The patient’s psoriasis was well controlled with adalimumab for 22 months, but she then developed a thin white plaque on the right lateral tongue (Figure 1). An incisional biopsy of the tongue performed by an oral surgeon revealed hyperkeratosis with Candida colonization and viral cytopathic effect (Figure 2). An EBV DNA in situ hybridization stain revealed focal positivity within these cells (Figure 3), leading to a diagnosis of OHL. Laboratory evaluation demonstrated a normal complete blood cell count with differential and liver panel as well as a negative HIV test. The patient otherwise felt well and denied fevers, lymphadenopathy, and weight loss.



We consulted with an infectious disease and immunodeficiency specialist regarding the patient’s case. Before conducting further evaluation beyond HIV screening for immunodeficiency states, adalimumab was discontinued to see if the OHL would spontaneously resolve. Three months after discontinuation of adalimumab, the white plaque on the right lateral tongue was notably improved. The OHL continued to disappear and was completely resolved 1 year after discontinuation of adalimumab. The patient’s psoriasis had subsequently remained well controlled with diet and weight loss, smoking cessation, topical steroids, and apremilast without any recurrence of the OHL.
Oral hairy leukoplakia is associated with upregulated EBV replication and EBV-encoded proteins such as latent membrane protein 1.2 It often presents as white or gray patches on the lateral lingual margins with prominent folds and/or projections, giving a shaggy appearance. Oral hairy leukoplakia often is specific for HIV infection and rarely is associated with other immunodeficiencies.2 Prasad and Bilodeau5 performed a literature review of medical conditions and immunosuppressive medications associated with OHL in patients without HIV. Allogeneic transplant was associated with the highest incidence of OHL in HIV-negative patients (59.2% [45/76]).5 Various combinations of immunosuppressive medications (eg, prednisone, cyclosporine, azathioprine) also may be implicated in cases of HIV-negative patients with OHL. A case of OHL also has been reported with long-standing use of inhaled corticosteroids in an immunocompetent, HIV-negative patient.6 Another case was reported with long-term use of the aromatic antiepileptic lamotrigine, which resolved once stopping the medication.8 Although EBV is an oncovirus and has been associated with lymphoproliferative disorders and nasopharyngeal carcinoma, OHL is not considered to be a premalignant lesion.7 Despite the strong association between OHL and HIV, our patient was HIV negative. The only immunocompromising factor in our patient was the use of adalimumab to treat psoriasis. We did not conduct further testing for immunodeficiency states because the OHL spontaneously resolved when the adalimumab was discontinued.
PubMed and Ovid searches of articles indexed for MEDLINE using the terms adalimumab and oral hairy leukoplakia as well as TNF-alpha inhibitor and oral hairy leukoplakia with humans and English language as limitations revealed that no cases have been reported in the literature demonstrating an association between OHL and adalimumab or any other TNF-α inhibitor. However, Cetkovska et al9 reported a case of EBV hepatitis and subsequently chronic hepatitis as a complication of infliximab used for the treatment of chronic psoriasis. Because TNF-α and IFN-γ play an important role in controlling viral infections, there is an increased risk for reactivating a viral illness when depleting TNF through pharmacologic measures (ie, adalimumab, infliximab).8 Another case of EBV-associated plasmablastic lymphoma was reported after 1 year of adalimumab use in a patient with Crohn disease. The plasmablastic lymphoma resolved after 4 rounds of chemotherapy.10
The only contraindication for the use of adalimumab is a known hypersensitivity to the drug. Relative contraindications for use of adalimumab include active tuberculosis, demyelinating disease, hematologic diseases (ie, thrombocytopenia, pancytopenia), lymphoma, hepatitis C, and hepatitis B.11 The most common adverse effect of adalimumab is an injection-site reaction. Additional reported adverse effects of TNF-α inhibitors as a class are lymphoma, melanoma, nonmelanoma skin cancer, reactivation of latent tuberculosis, congestive heart failure, autoimmunity, and hematologic toxicity.11
This case demonstrates an association between adalimumab and OHL in an HIV-negative patient. Although the mechanism behind OHL and immunosuppression remains to be elucidated, this association is important to keep in mind when using adalimumab or other TNF-α inhibitors for the treatment of psoriasis or other medical conditions.
To the Editor:
Oral hairy leukoplakia (OHL) is an Epstein-Barr virus (EBV)–mediated mucocutaneous disease that often involves the lingual epithelium. The lateral portions of the tongue are the most commonly affected sites. The lesions often are described as asymptomatic, white, corrugated patches or plaques that are unable to be scraped off.1 Oral hairy leukoplakia was first identified in 1984 and was considered to be associated with AIDS.2 An association between the presence of OHL and the degree of immunosuppression as well as the severity of human immunodeficiency virus (HIV) has been reported.3 Although OHL initially was considered to be pathognomonic for HIV, it has since been described in multiple other immunosuppressive conditions.4 Numerous medical conditions and combinations of immunosuppressive medications have been associated with OHL in patients who were HIV negative.5
Adalimumab is an injectable human IgG1 recombinant antibody to tumor necrosis factor α (TNF-α).6 It currently is approved by the US Food and Drug Administration for the treatment of rheumatoid arthritis, juvenile idiopathic arthritis, psoriatic arthritis, ankylosing spondylitis, adult and pediatric Crohn disease, ulcerative colitis, noninfectious uveitis, hidradenitis suppurativa, and plaque psoriasis.7 We report a case of OHL associated with the use of adalimumab.
A 47-year-old woman initially presented with chronic plaque-type psoriasis. Her medical history was notable for bipolar disorder, migraines, hypertension, and tobacco use. The patient’s psoriasis initially was well controlled on a regimen of topical steroids and methotrexate; however, methotrexate was stopped after 2.5 years due to a mildly elevated alanine aminotransferase level, as well as an abnormal liver biopsy showing mildly active (grade 1 of 3) steatohepatitis with portal chronic inflammation, pericellular fibrosis, and portal and focal periportal fibrosis (stage 1-2 of 4). The patient and her dermatologist were uncomfortable continuing methotrexate with these findings. After baseline screening including a negative purified protein derivative skin test, adalimumab was initiated. A loading dose of 80 mg subcutaneously (SQ) was given, followed by adalimumab 40 mg SQ 1 week later and 40 mg every other week as maintenance.
The patient’s psoriasis was well controlled with adalimumab for 22 months, but she then developed a thin white plaque on the right lateral tongue (Figure 1). An incisional biopsy of the tongue performed by an oral surgeon revealed hyperkeratosis with Candida colonization and viral cytopathic effect (Figure 2). An EBV DNA in situ hybridization stain revealed focal positivity within these cells (Figure 3), leading to a diagnosis of OHL. Laboratory evaluation demonstrated a normal complete blood cell count with differential and liver panel as well as a negative HIV test. The patient otherwise felt well and denied fevers, lymphadenopathy, and weight loss.



We consulted with an infectious disease and immunodeficiency specialist regarding the patient’s case. Before conducting further evaluation beyond HIV screening for immunodeficiency states, adalimumab was discontinued to see if the OHL would spontaneously resolve. Three months after discontinuation of adalimumab, the white plaque on the right lateral tongue was notably improved. The OHL continued to disappear and was completely resolved 1 year after discontinuation of adalimumab. The patient’s psoriasis had subsequently remained well controlled with diet and weight loss, smoking cessation, topical steroids, and apremilast without any recurrence of the OHL.
Oral hairy leukoplakia is associated with upregulated EBV replication and EBV-encoded proteins such as latent membrane protein 1.2 It often presents as white or gray patches on the lateral lingual margins with prominent folds and/or projections, giving a shaggy appearance. Oral hairy leukoplakia often is specific for HIV infection and rarely is associated with other immunodeficiencies.2 Prasad and Bilodeau5 performed a literature review of medical conditions and immunosuppressive medications associated with OHL in patients without HIV. Allogeneic transplant was associated with the highest incidence of OHL in HIV-negative patients (59.2% [45/76]).5 Various combinations of immunosuppressive medications (eg, prednisone, cyclosporine, azathioprine) also may be implicated in cases of HIV-negative patients with OHL. A case of OHL also has been reported with long-standing use of inhaled corticosteroids in an immunocompetent, HIV-negative patient.6 Another case was reported with long-term use of the aromatic antiepileptic lamotrigine, which resolved once stopping the medication.8 Although EBV is an oncovirus and has been associated with lymphoproliferative disorders and nasopharyngeal carcinoma, OHL is not considered to be a premalignant lesion.7 Despite the strong association between OHL and HIV, our patient was HIV negative. The only immunocompromising factor in our patient was the use of adalimumab to treat psoriasis. We did not conduct further testing for immunodeficiency states because the OHL spontaneously resolved when the adalimumab was discontinued.
PubMed and Ovid searches of articles indexed for MEDLINE using the terms adalimumab and oral hairy leukoplakia as well as TNF-alpha inhibitor and oral hairy leukoplakia with humans and English language as limitations revealed that no cases have been reported in the literature demonstrating an association between OHL and adalimumab or any other TNF-α inhibitor. However, Cetkovska et al9 reported a case of EBV hepatitis and subsequently chronic hepatitis as a complication of infliximab used for the treatment of chronic psoriasis. Because TNF-α and IFN-γ play an important role in controlling viral infections, there is an increased risk for reactivating a viral illness when depleting TNF through pharmacologic measures (ie, adalimumab, infliximab).8 Another case of EBV-associated plasmablastic lymphoma was reported after 1 year of adalimumab use in a patient with Crohn disease. The plasmablastic lymphoma resolved after 4 rounds of chemotherapy.10
The only contraindication for the use of adalimumab is a known hypersensitivity to the drug. Relative contraindications for use of adalimumab include active tuberculosis, demyelinating disease, hematologic diseases (ie, thrombocytopenia, pancytopenia), lymphoma, hepatitis C, and hepatitis B.11 The most common adverse effect of adalimumab is an injection-site reaction. Additional reported adverse effects of TNF-α inhibitors as a class are lymphoma, melanoma, nonmelanoma skin cancer, reactivation of latent tuberculosis, congestive heart failure, autoimmunity, and hematologic toxicity.11
This case demonstrates an association between adalimumab and OHL in an HIV-negative patient. Although the mechanism behind OHL and immunosuppression remains to be elucidated, this association is important to keep in mind when using adalimumab or other TNF-α inhibitors for the treatment of psoriasis or other medical conditions.
- Triantos D, Porter SR, Scully C, et al. Oral hairy leukoplakia: clinicopathologic features, pathogenesis, diagnosis, and clinical significance. Clin Infect Dis. 1997;25:1392-1396.
- Greenspan D, Greenspan JS, Conant M, et al. Oral “hairy” leucoplakia in male homosexuals: evidence of association with both papillomavirus and a herpes-group virus. Lancet. 1984;2:831-834.
- Glick M, Muzyka BC, Lurie D, et al. Oral manifestations associated with HIV-related disease as marks for immune suppression and AIDS. Oral Surg Oral Med Oral Pathol. 1994;77:344-349.
- Chambers AE, Conn B, Pemberton M, et al. Twenty-first-century oral hair leukoplakia—a non-HIV-associated entity. Oral Surg Oral Med Oral Patho Oral Radiol. 2015;119:326-332.
- Prasad JL, Bilodeau EA. Oral hairy leukoplakia in patients without HIV: presentation of 2 new cases. Oral Surg Oral Med Oral Pathol Oral Radiol. 2014;118:E151-E160.
- Moffat M, Jauhar S, Jones ME, et al. Oral hairy leukoplakia in an HIV-negative, immunocompetent patient. Oral Biosci Med. 2005;2:282-284.
- Greenspan JS, Greenspan D. Oral hairy leukoplakia: diagnosis and management. Oral Surg Oral Med Oral Pathol. 1989;67:396-403.
- Gordins P, Sloan P, Spickett GP, et al. Oral hairy leukoplakia in a patient on long-term anticonvulsant treatment with lamotrigine. Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 2011;111:E17-E23.
- Cetkovska P, Lomicova I, Mukensnabl P, et al. Anti-tumour necrosis factor treatment of severe psoriasis complicated by Epstein-Barr virus hepatitis and subsequently by chronic hepatitis. Dermatol Ther. 2015;28:369-372.
- Liu L, Charabaty A, Ozdemirli M. EBV-associated plasmablastic lymphoma in a patient with Crohn’s disease after adalimumab treatment. J Crohns Colitis. 2013;7:E118-E119.
- Humira [package insert]. North Chicago, IL: AbbVie Inc; 2018.
- Triantos D, Porter SR, Scully C, et al. Oral hairy leukoplakia: clinicopathologic features, pathogenesis, diagnosis, and clinical significance. Clin Infect Dis. 1997;25:1392-1396.
- Greenspan D, Greenspan JS, Conant M, et al. Oral “hairy” leucoplakia in male homosexuals: evidence of association with both papillomavirus and a herpes-group virus. Lancet. 1984;2:831-834.
- Glick M, Muzyka BC, Lurie D, et al. Oral manifestations associated with HIV-related disease as marks for immune suppression and AIDS. Oral Surg Oral Med Oral Pathol. 1994;77:344-349.
- Chambers AE, Conn B, Pemberton M, et al. Twenty-first-century oral hair leukoplakia—a non-HIV-associated entity. Oral Surg Oral Med Oral Patho Oral Radiol. 2015;119:326-332.
- Prasad JL, Bilodeau EA. Oral hairy leukoplakia in patients without HIV: presentation of 2 new cases. Oral Surg Oral Med Oral Pathol Oral Radiol. 2014;118:E151-E160.
- Moffat M, Jauhar S, Jones ME, et al. Oral hairy leukoplakia in an HIV-negative, immunocompetent patient. Oral Biosci Med. 2005;2:282-284.
- Greenspan JS, Greenspan D. Oral hairy leukoplakia: diagnosis and management. Oral Surg Oral Med Oral Pathol. 1989;67:396-403.
- Gordins P, Sloan P, Spickett GP, et al. Oral hairy leukoplakia in a patient on long-term anticonvulsant treatment with lamotrigine. Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 2011;111:E17-E23.
- Cetkovska P, Lomicova I, Mukensnabl P, et al. Anti-tumour necrosis factor treatment of severe psoriasis complicated by Epstein-Barr virus hepatitis and subsequently by chronic hepatitis. Dermatol Ther. 2015;28:369-372.
- Liu L, Charabaty A, Ozdemirli M. EBV-associated plasmablastic lymphoma in a patient with Crohn’s disease after adalimumab treatment. J Crohns Colitis. 2013;7:E118-E119.
- Humira [package insert]. North Chicago, IL: AbbVie Inc; 2018.
Practice Points
- Workup for new-onset oral hairy leukoplakia should include a comprehensive medication history.
- Oral hairy leukoplakia is an uncommon side effect of adalimumab.
Practicing medicine without judgment
“What do you think of all this election stuff?” I froze. Sitting on the exam table was a 50-something-year-old woman. Her hair was long, but not gray. She was wearing a mask without distinctive markings, such as Trump lips or #BLM to identify the political leanings of the owner. She had a subtle New York accent, perhaps dating back to the Giuliani years. It was hard to know her intention. “It’s a trap!” I could hear Admiral Ackbar’s voice in my head. “Don’t engage.” We all know nothing erodes trust faster than showing your blue or red colors before you know which your patient identifies.
Instead, I replied that indeed it has been a stressful year for us all. Then I paused. She shifted a bit and tugged at the gown sleeves and admitted this was the most stress she felt in years. She was seeing me for lichen sclerosus et atrophicus, a terribly itchy, sometimes-disfiguring eruption that can occur in the vulva. She was dealing with COVID-19, kids, divorce, a new partner, working from home, parents, and now the election drama.
At this point in the visit, I knew I could help her. First, the treatment for lichen sclerosus is straightforward and mostly effective. Second, I knew I’d have 7 minutes to spare to just listen. It was a lucky break, as often no such gift of time presents itself while seeing patients in a busy clinic. We take vitals, history (typing), do an exam, make a diagnosis (more typing), and maybe a procedure (yet more typing). All of this is necessary, but sometimes not what our patient needs. Some really need just to connect and share their burden with someone who isn’t a friend or family. As physicians, we have a unique opportunity to see and hear people without judgment.
This reminds me of a recent episode from Sam Harris’s podcast, “Making Sense.” Mr. Harris, a philosopher (and “blue” all the way through) revealed his insight into Presidents Trump’s appeal. Leaving policy aside, Mr. Harris notes that people are drawn to the President because he never judges you. He is incapable of being sanctimonious, Mr. Harris argues, and therefore creates a safe space for people to continue their lives, however flawed, without expectation that they improve.
I’m unsure just how much of this theory explains the devotion of his supporters, but it resonated with me. We doctors are sanctimonious by nature. The better part of my day is spent prodding people to be better: Wear more sunscreen, exercise more, stop believing in conspiracy theories, get your flu shot, and above all, stop scratching! In doing so, I’m in a way judging them. Finger wagging: You’re lazy or poor or dumb or stubborn. “You aren’t as good as me,” is what they might feel after 15 minutes of my pep talk.
But what if that’s wrong? What if they are just fine exactly the way they are? Perhaps what my lichen sclerosis patient needs more than anything is unconditional attention? She, like most of our patients, is well aware of how her shortcomings might contribute to her own anxiety or difficulties. And now she has this rash and that’s probably somehow her fault too, she thinks.
How can I best help her? Betamethasone dipropionate b.i.d. for 2 weeks and spend the last 7 minutes just sitting and listening without judgment or advice. I don’t know who she wanted to win the election. It didn’t matter, she was exactly right to believe what she believed, either way.
Dr. Benabio is director of Healthcare Transformation and chief of dermatology at Kaiser Permanente San Diego. The opinions expressed in this column are his own and do not represent those of Kaiser Permanente. Dr. Benabio is @Dermdoc on Twitter. Write to him at [email protected].
“What do you think of all this election stuff?” I froze. Sitting on the exam table was a 50-something-year-old woman. Her hair was long, but not gray. She was wearing a mask without distinctive markings, such as Trump lips or #BLM to identify the political leanings of the owner. She had a subtle New York accent, perhaps dating back to the Giuliani years. It was hard to know her intention. “It’s a trap!” I could hear Admiral Ackbar’s voice in my head. “Don’t engage.” We all know nothing erodes trust faster than showing your blue or red colors before you know which your patient identifies.
Instead, I replied that indeed it has been a stressful year for us all. Then I paused. She shifted a bit and tugged at the gown sleeves and admitted this was the most stress she felt in years. She was seeing me for lichen sclerosus et atrophicus, a terribly itchy, sometimes-disfiguring eruption that can occur in the vulva. She was dealing with COVID-19, kids, divorce, a new partner, working from home, parents, and now the election drama.
At this point in the visit, I knew I could help her. First, the treatment for lichen sclerosus is straightforward and mostly effective. Second, I knew I’d have 7 minutes to spare to just listen. It was a lucky break, as often no such gift of time presents itself while seeing patients in a busy clinic. We take vitals, history (typing), do an exam, make a diagnosis (more typing), and maybe a procedure (yet more typing). All of this is necessary, but sometimes not what our patient needs. Some really need just to connect and share their burden with someone who isn’t a friend or family. As physicians, we have a unique opportunity to see and hear people without judgment.
This reminds me of a recent episode from Sam Harris’s podcast, “Making Sense.” Mr. Harris, a philosopher (and “blue” all the way through) revealed his insight into Presidents Trump’s appeal. Leaving policy aside, Mr. Harris notes that people are drawn to the President because he never judges you. He is incapable of being sanctimonious, Mr. Harris argues, and therefore creates a safe space for people to continue their lives, however flawed, without expectation that they improve.
I’m unsure just how much of this theory explains the devotion of his supporters, but it resonated with me. We doctors are sanctimonious by nature. The better part of my day is spent prodding people to be better: Wear more sunscreen, exercise more, stop believing in conspiracy theories, get your flu shot, and above all, stop scratching! In doing so, I’m in a way judging them. Finger wagging: You’re lazy or poor or dumb or stubborn. “You aren’t as good as me,” is what they might feel after 15 minutes of my pep talk.
But what if that’s wrong? What if they are just fine exactly the way they are? Perhaps what my lichen sclerosis patient needs more than anything is unconditional attention? She, like most of our patients, is well aware of how her shortcomings might contribute to her own anxiety or difficulties. And now she has this rash and that’s probably somehow her fault too, she thinks.
How can I best help her? Betamethasone dipropionate b.i.d. for 2 weeks and spend the last 7 minutes just sitting and listening without judgment or advice. I don’t know who she wanted to win the election. It didn’t matter, she was exactly right to believe what she believed, either way.
Dr. Benabio is director of Healthcare Transformation and chief of dermatology at Kaiser Permanente San Diego. The opinions expressed in this column are his own and do not represent those of Kaiser Permanente. Dr. Benabio is @Dermdoc on Twitter. Write to him at [email protected].
“What do you think of all this election stuff?” I froze. Sitting on the exam table was a 50-something-year-old woman. Her hair was long, but not gray. She was wearing a mask without distinctive markings, such as Trump lips or #BLM to identify the political leanings of the owner. She had a subtle New York accent, perhaps dating back to the Giuliani years. It was hard to know her intention. “It’s a trap!” I could hear Admiral Ackbar’s voice in my head. “Don’t engage.” We all know nothing erodes trust faster than showing your blue or red colors before you know which your patient identifies.
Instead, I replied that indeed it has been a stressful year for us all. Then I paused. She shifted a bit and tugged at the gown sleeves and admitted this was the most stress she felt in years. She was seeing me for lichen sclerosus et atrophicus, a terribly itchy, sometimes-disfiguring eruption that can occur in the vulva. She was dealing with COVID-19, kids, divorce, a new partner, working from home, parents, and now the election drama.
At this point in the visit, I knew I could help her. First, the treatment for lichen sclerosus is straightforward and mostly effective. Second, I knew I’d have 7 minutes to spare to just listen. It was a lucky break, as often no such gift of time presents itself while seeing patients in a busy clinic. We take vitals, history (typing), do an exam, make a diagnosis (more typing), and maybe a procedure (yet more typing). All of this is necessary, but sometimes not what our patient needs. Some really need just to connect and share their burden with someone who isn’t a friend or family. As physicians, we have a unique opportunity to see and hear people without judgment.
This reminds me of a recent episode from Sam Harris’s podcast, “Making Sense.” Mr. Harris, a philosopher (and “blue” all the way through) revealed his insight into Presidents Trump’s appeal. Leaving policy aside, Mr. Harris notes that people are drawn to the President because he never judges you. He is incapable of being sanctimonious, Mr. Harris argues, and therefore creates a safe space for people to continue their lives, however flawed, without expectation that they improve.
I’m unsure just how much of this theory explains the devotion of his supporters, but it resonated with me. We doctors are sanctimonious by nature. The better part of my day is spent prodding people to be better: Wear more sunscreen, exercise more, stop believing in conspiracy theories, get your flu shot, and above all, stop scratching! In doing so, I’m in a way judging them. Finger wagging: You’re lazy or poor or dumb or stubborn. “You aren’t as good as me,” is what they might feel after 15 minutes of my pep talk.
But what if that’s wrong? What if they are just fine exactly the way they are? Perhaps what my lichen sclerosis patient needs more than anything is unconditional attention? She, like most of our patients, is well aware of how her shortcomings might contribute to her own anxiety or difficulties. And now she has this rash and that’s probably somehow her fault too, she thinks.
How can I best help her? Betamethasone dipropionate b.i.d. for 2 weeks and spend the last 7 minutes just sitting and listening without judgment or advice. I don’t know who she wanted to win the election. It didn’t matter, she was exactly right to believe what she believed, either way.
Dr. Benabio is director of Healthcare Transformation and chief of dermatology at Kaiser Permanente San Diego. The opinions expressed in this column are his own and do not represent those of Kaiser Permanente. Dr. Benabio is @Dermdoc on Twitter. Write to him at [email protected].
Moderna: Interim data show 94.5% efficacy for COVID-19 vaccine, will seek FDA EUA
The Moderna mRNA-1273 vaccine, in development to prevent COVID-19, yielded 94.5% efficacy in early results and is generally well tolerated, the company announced early Monday. The product can be stored at refrigeration temperatures common to many physician offices, pharmacies, and hospitals.
The first interim results of the phase 3 COVE trial included 95 participants with confirmed COVID-19. An independent data safety monitoring board, which was appointed by the National Institutes of Health, informed Moderna that 90 of the patients who were positive for COVID-19 were in a placebo group and that 5 patients were in the mRNA-1273 vaccine group, resulting in a vaccine efficacy of 94.5% (P < .0001).
Interim data included 11 patients with severe COVID-19, all of whom were in the placebo group.
“This positive interim analysis from our phase 3 study has given us the first clinical validation that our vaccine can prevent COVID-19 disease, including severe disease,” said Stéphane Bancel, CEO of Moderna, said in a statement.
The vaccine met its primary study endpoint, which was based on adjudicated data that were collected starting 2 weeks after the second dose of mRNA-1273. The interim study population included people who could be at higher risk for COVID-19, including 15 adults aged 65 years and older and 20 participants from diverse communities.
Safety data
The DSMB also reviewed safety data for the COVE study interim results. The vaccine was generally safe and well tolerated, as determined on the basis of solicited adverse events. Most adverse events were mild to moderate and were generally short-lived, according to a company news release.
Injection-site pain was reported in 2.7% of participants after the first dose. After the second dose, 9.7% of participants reported fatigue, 8.9% reported myalgia, 5.2% reported arthralgia, 4.5% reported headache, 4.1% reported pain, and 2.0% reported erythema or redness at the injection site.
Moderna plans to request emergency-use authorization (EUA) from the Food and Drug Administration in the coming weeks. The company expects that the EUA will be based on more data from the COVE study, including a final analysis of 151 patients with a median follow-up of more than 2 months. Moderna also plans to seek authorizations from global regulatory agencies.
The company expects to have approximately 20 million doses of mRNA-1273 ready to ship in the United States by the end of the year. In addition, the company says it remains on track to manufacture between 500 million and 1 billion doses globally in 2021.
Moderna is developing distribution plans in conjunction with the Centers for Disease Control and Prevention, the federal government’s Operation Warp Speed, and McKesson, a COVID-19 vaccine distributor contracted by the U.S. government.
Refrigeration requirements
The mRNA-1273 vaccine can be shipped and stored for up to 6 months at –20° C (about –4° F), a temperature maintained in most home or medical freezers, according to Moderna. The company expects that, after the product thaws, it will remain stable at standard refrigerator temperatures of 2°-8° C (36°-46° F) for up to 30 days within the 6-month shelf life.
Because the mRNA-1273 vaccine is stable at these refrigerator temperatures, it can be stored at most physicians’ offices, pharmacies, and hospitals, the company noted. In contrast, the similar Pfizer BTN162b2 vaccine – early results for which showed a 90% efficacy rate – requires shipment and storage at “deep-freeze” conditions of –70° C or –80° C, which is more challenging from a logistic point of view.
Moderna’s mRNA-1273 can be kept at room temperature for up to 12 hours after removal from a refrigerator for patient administration. The vaccine will not require dilution prior to use.
More than 30,000 people aged older than 18 years in the United States are enrolled in the COVE study. The research is being conducted in collaboration with the National Institute of Allergy and Infectious Diseases and the Biomedical Advanced Research and Development Authority, part of the Office of the Assistant Secretary for Preparedness and Response at the Department of Health & Human Services.
A version of this article originally appeared on Medscape.com.
The Moderna mRNA-1273 vaccine, in development to prevent COVID-19, yielded 94.5% efficacy in early results and is generally well tolerated, the company announced early Monday. The product can be stored at refrigeration temperatures common to many physician offices, pharmacies, and hospitals.
The first interim results of the phase 3 COVE trial included 95 participants with confirmed COVID-19. An independent data safety monitoring board, which was appointed by the National Institutes of Health, informed Moderna that 90 of the patients who were positive for COVID-19 were in a placebo group and that 5 patients were in the mRNA-1273 vaccine group, resulting in a vaccine efficacy of 94.5% (P < .0001).
Interim data included 11 patients with severe COVID-19, all of whom were in the placebo group.
“This positive interim analysis from our phase 3 study has given us the first clinical validation that our vaccine can prevent COVID-19 disease, including severe disease,” said Stéphane Bancel, CEO of Moderna, said in a statement.
The vaccine met its primary study endpoint, which was based on adjudicated data that were collected starting 2 weeks after the second dose of mRNA-1273. The interim study population included people who could be at higher risk for COVID-19, including 15 adults aged 65 years and older and 20 participants from diverse communities.
Safety data
The DSMB also reviewed safety data for the COVE study interim results. The vaccine was generally safe and well tolerated, as determined on the basis of solicited adverse events. Most adverse events were mild to moderate and were generally short-lived, according to a company news release.
Injection-site pain was reported in 2.7% of participants after the first dose. After the second dose, 9.7% of participants reported fatigue, 8.9% reported myalgia, 5.2% reported arthralgia, 4.5% reported headache, 4.1% reported pain, and 2.0% reported erythema or redness at the injection site.
Moderna plans to request emergency-use authorization (EUA) from the Food and Drug Administration in the coming weeks. The company expects that the EUA will be based on more data from the COVE study, including a final analysis of 151 patients with a median follow-up of more than 2 months. Moderna also plans to seek authorizations from global regulatory agencies.
The company expects to have approximately 20 million doses of mRNA-1273 ready to ship in the United States by the end of the year. In addition, the company says it remains on track to manufacture between 500 million and 1 billion doses globally in 2021.
Moderna is developing distribution plans in conjunction with the Centers for Disease Control and Prevention, the federal government’s Operation Warp Speed, and McKesson, a COVID-19 vaccine distributor contracted by the U.S. government.
Refrigeration requirements
The mRNA-1273 vaccine can be shipped and stored for up to 6 months at –20° C (about –4° F), a temperature maintained in most home or medical freezers, according to Moderna. The company expects that, after the product thaws, it will remain stable at standard refrigerator temperatures of 2°-8° C (36°-46° F) for up to 30 days within the 6-month shelf life.
Because the mRNA-1273 vaccine is stable at these refrigerator temperatures, it can be stored at most physicians’ offices, pharmacies, and hospitals, the company noted. In contrast, the similar Pfizer BTN162b2 vaccine – early results for which showed a 90% efficacy rate – requires shipment and storage at “deep-freeze” conditions of –70° C or –80° C, which is more challenging from a logistic point of view.
Moderna’s mRNA-1273 can be kept at room temperature for up to 12 hours after removal from a refrigerator for patient administration. The vaccine will not require dilution prior to use.
More than 30,000 people aged older than 18 years in the United States are enrolled in the COVE study. The research is being conducted in collaboration with the National Institute of Allergy and Infectious Diseases and the Biomedical Advanced Research and Development Authority, part of the Office of the Assistant Secretary for Preparedness and Response at the Department of Health & Human Services.
A version of this article originally appeared on Medscape.com.
The Moderna mRNA-1273 vaccine, in development to prevent COVID-19, yielded 94.5% efficacy in early results and is generally well tolerated, the company announced early Monday. The product can be stored at refrigeration temperatures common to many physician offices, pharmacies, and hospitals.
The first interim results of the phase 3 COVE trial included 95 participants with confirmed COVID-19. An independent data safety monitoring board, which was appointed by the National Institutes of Health, informed Moderna that 90 of the patients who were positive for COVID-19 were in a placebo group and that 5 patients were in the mRNA-1273 vaccine group, resulting in a vaccine efficacy of 94.5% (P < .0001).
Interim data included 11 patients with severe COVID-19, all of whom were in the placebo group.
“This positive interim analysis from our phase 3 study has given us the first clinical validation that our vaccine can prevent COVID-19 disease, including severe disease,” said Stéphane Bancel, CEO of Moderna, said in a statement.
The vaccine met its primary study endpoint, which was based on adjudicated data that were collected starting 2 weeks after the second dose of mRNA-1273. The interim study population included people who could be at higher risk for COVID-19, including 15 adults aged 65 years and older and 20 participants from diverse communities.
Safety data
The DSMB also reviewed safety data for the COVE study interim results. The vaccine was generally safe and well tolerated, as determined on the basis of solicited adverse events. Most adverse events were mild to moderate and were generally short-lived, according to a company news release.
Injection-site pain was reported in 2.7% of participants after the first dose. After the second dose, 9.7% of participants reported fatigue, 8.9% reported myalgia, 5.2% reported arthralgia, 4.5% reported headache, 4.1% reported pain, and 2.0% reported erythema or redness at the injection site.
Moderna plans to request emergency-use authorization (EUA) from the Food and Drug Administration in the coming weeks. The company expects that the EUA will be based on more data from the COVE study, including a final analysis of 151 patients with a median follow-up of more than 2 months. Moderna also plans to seek authorizations from global regulatory agencies.
The company expects to have approximately 20 million doses of mRNA-1273 ready to ship in the United States by the end of the year. In addition, the company says it remains on track to manufacture between 500 million and 1 billion doses globally in 2021.
Moderna is developing distribution plans in conjunction with the Centers for Disease Control and Prevention, the federal government’s Operation Warp Speed, and McKesson, a COVID-19 vaccine distributor contracted by the U.S. government.
Refrigeration requirements
The mRNA-1273 vaccine can be shipped and stored for up to 6 months at –20° C (about –4° F), a temperature maintained in most home or medical freezers, according to Moderna. The company expects that, after the product thaws, it will remain stable at standard refrigerator temperatures of 2°-8° C (36°-46° F) for up to 30 days within the 6-month shelf life.
Because the mRNA-1273 vaccine is stable at these refrigerator temperatures, it can be stored at most physicians’ offices, pharmacies, and hospitals, the company noted. In contrast, the similar Pfizer BTN162b2 vaccine – early results for which showed a 90% efficacy rate – requires shipment and storage at “deep-freeze” conditions of –70° C or –80° C, which is more challenging from a logistic point of view.
Moderna’s mRNA-1273 can be kept at room temperature for up to 12 hours after removal from a refrigerator for patient administration. The vaccine will not require dilution prior to use.
More than 30,000 people aged older than 18 years in the United States are enrolled in the COVE study. The research is being conducted in collaboration with the National Institute of Allergy and Infectious Diseases and the Biomedical Advanced Research and Development Authority, part of the Office of the Assistant Secretary for Preparedness and Response at the Department of Health & Human Services.
A version of this article originally appeared on Medscape.com.
Poor image quality may limit televulvology care
Seeing patients with vulvar problems via telemedicine can lead to efficient and successful care, but there are challenges and limitations with this approach, doctors are finding.
Image quality is one key factor that determines whether a clinician can assess and manage a condition remotely, said Aruna Venkatesan, MD, chief of dermatology and director of the genital dermatology clinic at Santa Clara Valley Medical Center in San Jose, Calif. Other issues may be especially relevant to televulvology, including privacy concerns.
“Who is helping with the positioning? Who is the photographer? Is the patient comfortable with having photos taken of this part of their body and submitted, even if they know it is submitted securely? Because they might not be,” Dr. Venkatesan said in a lecture at a virtual conference on diseases of the vulva and vagina, hosted by the International Society for the Study of Vulvovaginal Disease.
When quality photographs from referring providers are available, Dr. Venkatesan has conducted virtual new consultations. “But sometimes I will do a virtual telemedicine visit as the first visit and then figure out, okay, this isn’t really sufficient. I need to see them in person.”
Melissa Mauskar, MD, assistant professor of dermatology and obstetrics and gynecology at the University of Texas Southwestern Medical Center, Dallas, described a case early on during the COVID-19 pandemic that illustrates a limitation of virtual visits.
A patient sent in a photograph that appeared to show lichen sclerosus. “There looked like some classic lichen sclerosus changes,” Dr. Mauskar said during a discussion at the meeting. “But she was having a lot of pain, and after a week, her pain still was not better.”
Dr. Mauskar brought the patient into the office and ultimately diagnosed a squamous cell carcinoma. “What I thought was a normal erosion was actually an ulcerated plaque,” she said.
Like Dr. Venkatesan, Dr. Mauskar has found that image quality can be uneven. Photographs may be out of focus. Video visits have been a mixed bag. Some are successful. Other times, Dr. Mauskar has to tell the patient she needs to see her in the office.
Certain clinical scenarios require a vaginal exam, Dr. Venkatesan noted. Although some type of assessment may be possible if a patient is with a primary care provider during the telemedicine visit, the examination may not be equivalent. Doctors also should anticipate where a patient might go to have a biopsy if one is necessary.
Another telemedicine caveat pertains to patient counseling. When using store-and-forward telemedicine systems, advising patients in a written report can be challenging. “Is there an easy way ... to counsel patients how to apply their topical medications?” Dr. Venkatesan said.
Excellent care is possible
Vulvology is a small part of Dr. Venkatesan’s general dermatology practice, which has used telemedicine extensively since the pandemic.
In recent years, Dr. Venkatesan’s clinic began encouraging providers in their health system to submit photographs with referrals. “That has really paid off now because we have been able to help provide a lot of excellent quality care for patients without them having to come in,” she said. “We may be able to say: ‘These are excellent photos. We know what this patient has. We can manage it. They don’t need to come see us in person.’ ” That could be the case for certain types of acne, eczema, and psoriasis.
In other cases, they may be able to provide initial advice remotely but still want to see the patient. For a patient with severe acne, “I may be able to tell the referring doctor: ‘Please start the patient on these three medicines. It will take 2 months for those medicines to start working and then we will plan to have an in-person dermatology visit.’ ” In this case, telemedicine essentially replaces one in-person visit.
If photographs are poor, the differential diagnosis is broad, a procedure is required, the doctor needs to touch the lesion, or more involved history taking or counseling are required, the patient may need to go into the office.
Beyond its public health advantages during a pandemic, telemedicine can improve access for patients who live far away, lack transportation, or are unable to take time off from work. It also can decrease patient wait times. “Once we started doing some telemedicine work … we went from having a 5-month wait time for patients to see us in person to a 72-hour wait time for providing some care for patients if they had good photos as part of their referral,” Dr. Venkatesan said.
Telemedicine has been used in inpatient and outpatient dermatology settings. Primary care providers who consult with dermatologists using a store-and-forward telemedicine system may improve their dermatology knowledge and feel more confident in their ability to diagnose and manage dermatologic conditions, research indicates.
In obstetrics and gynecology, telemedicine may play a role in preconception, contraception, and medical abortion care, prenatal visits, well-woman exams, mental health, and pre- and postoperative counseling, a recent review suggests.
Image quality is key
“Quality of the image is so critical for being able to provide good care, especially in such a visual exam field as dermatology,” Dr. Venkatesan said.
To that end, doctors have offered recommendations on how to photograph skin conditions. A guide shared by the mobile telehealth system company ClickMedix suggests focusing on the area of importance, capturing the extent of involvement, and including involved and uninvolved areas.
Good lighting and checking the image resolution can help, Dr. Venkatesan offered. Nevertheless, patients may have difficulty photographing themselves. If a patient is with their primary care doctor, “we are much more likely to be able to get good quality photos,” she said.
Dr. Venkatesan is a paid consultant for DirectDerm, a store-and-forward teledermatology company. Dr. Mauskar had no relevant disclosures.
Seeing patients with vulvar problems via telemedicine can lead to efficient and successful care, but there are challenges and limitations with this approach, doctors are finding.
Image quality is one key factor that determines whether a clinician can assess and manage a condition remotely, said Aruna Venkatesan, MD, chief of dermatology and director of the genital dermatology clinic at Santa Clara Valley Medical Center in San Jose, Calif. Other issues may be especially relevant to televulvology, including privacy concerns.
“Who is helping with the positioning? Who is the photographer? Is the patient comfortable with having photos taken of this part of their body and submitted, even if they know it is submitted securely? Because they might not be,” Dr. Venkatesan said in a lecture at a virtual conference on diseases of the vulva and vagina, hosted by the International Society for the Study of Vulvovaginal Disease.
When quality photographs from referring providers are available, Dr. Venkatesan has conducted virtual new consultations. “But sometimes I will do a virtual telemedicine visit as the first visit and then figure out, okay, this isn’t really sufficient. I need to see them in person.”
Melissa Mauskar, MD, assistant professor of dermatology and obstetrics and gynecology at the University of Texas Southwestern Medical Center, Dallas, described a case early on during the COVID-19 pandemic that illustrates a limitation of virtual visits.
A patient sent in a photograph that appeared to show lichen sclerosus. “There looked like some classic lichen sclerosus changes,” Dr. Mauskar said during a discussion at the meeting. “But she was having a lot of pain, and after a week, her pain still was not better.”
Dr. Mauskar brought the patient into the office and ultimately diagnosed a squamous cell carcinoma. “What I thought was a normal erosion was actually an ulcerated plaque,” she said.
Like Dr. Venkatesan, Dr. Mauskar has found that image quality can be uneven. Photographs may be out of focus. Video visits have been a mixed bag. Some are successful. Other times, Dr. Mauskar has to tell the patient she needs to see her in the office.
Certain clinical scenarios require a vaginal exam, Dr. Venkatesan noted. Although some type of assessment may be possible if a patient is with a primary care provider during the telemedicine visit, the examination may not be equivalent. Doctors also should anticipate where a patient might go to have a biopsy if one is necessary.
Another telemedicine caveat pertains to patient counseling. When using store-and-forward telemedicine systems, advising patients in a written report can be challenging. “Is there an easy way ... to counsel patients how to apply their topical medications?” Dr. Venkatesan said.
Excellent care is possible
Vulvology is a small part of Dr. Venkatesan’s general dermatology practice, which has used telemedicine extensively since the pandemic.
In recent years, Dr. Venkatesan’s clinic began encouraging providers in their health system to submit photographs with referrals. “That has really paid off now because we have been able to help provide a lot of excellent quality care for patients without them having to come in,” she said. “We may be able to say: ‘These are excellent photos. We know what this patient has. We can manage it. They don’t need to come see us in person.’ ” That could be the case for certain types of acne, eczema, and psoriasis.
In other cases, they may be able to provide initial advice remotely but still want to see the patient. For a patient with severe acne, “I may be able to tell the referring doctor: ‘Please start the patient on these three medicines. It will take 2 months for those medicines to start working and then we will plan to have an in-person dermatology visit.’ ” In this case, telemedicine essentially replaces one in-person visit.
If photographs are poor, the differential diagnosis is broad, a procedure is required, the doctor needs to touch the lesion, or more involved history taking or counseling are required, the patient may need to go into the office.
Beyond its public health advantages during a pandemic, telemedicine can improve access for patients who live far away, lack transportation, or are unable to take time off from work. It also can decrease patient wait times. “Once we started doing some telemedicine work … we went from having a 5-month wait time for patients to see us in person to a 72-hour wait time for providing some care for patients if they had good photos as part of their referral,” Dr. Venkatesan said.
Telemedicine has been used in inpatient and outpatient dermatology settings. Primary care providers who consult with dermatologists using a store-and-forward telemedicine system may improve their dermatology knowledge and feel more confident in their ability to diagnose and manage dermatologic conditions, research indicates.
In obstetrics and gynecology, telemedicine may play a role in preconception, contraception, and medical abortion care, prenatal visits, well-woman exams, mental health, and pre- and postoperative counseling, a recent review suggests.
Image quality is key
“Quality of the image is so critical for being able to provide good care, especially in such a visual exam field as dermatology,” Dr. Venkatesan said.
To that end, doctors have offered recommendations on how to photograph skin conditions. A guide shared by the mobile telehealth system company ClickMedix suggests focusing on the area of importance, capturing the extent of involvement, and including involved and uninvolved areas.
Good lighting and checking the image resolution can help, Dr. Venkatesan offered. Nevertheless, patients may have difficulty photographing themselves. If a patient is with their primary care doctor, “we are much more likely to be able to get good quality photos,” she said.
Dr. Venkatesan is a paid consultant for DirectDerm, a store-and-forward teledermatology company. Dr. Mauskar had no relevant disclosures.
Seeing patients with vulvar problems via telemedicine can lead to efficient and successful care, but there are challenges and limitations with this approach, doctors are finding.
Image quality is one key factor that determines whether a clinician can assess and manage a condition remotely, said Aruna Venkatesan, MD, chief of dermatology and director of the genital dermatology clinic at Santa Clara Valley Medical Center in San Jose, Calif. Other issues may be especially relevant to televulvology, including privacy concerns.
“Who is helping with the positioning? Who is the photographer? Is the patient comfortable with having photos taken of this part of their body and submitted, even if they know it is submitted securely? Because they might not be,” Dr. Venkatesan said in a lecture at a virtual conference on diseases of the vulva and vagina, hosted by the International Society for the Study of Vulvovaginal Disease.
When quality photographs from referring providers are available, Dr. Venkatesan has conducted virtual new consultations. “But sometimes I will do a virtual telemedicine visit as the first visit and then figure out, okay, this isn’t really sufficient. I need to see them in person.”
Melissa Mauskar, MD, assistant professor of dermatology and obstetrics and gynecology at the University of Texas Southwestern Medical Center, Dallas, described a case early on during the COVID-19 pandemic that illustrates a limitation of virtual visits.
A patient sent in a photograph that appeared to show lichen sclerosus. “There looked like some classic lichen sclerosus changes,” Dr. Mauskar said during a discussion at the meeting. “But she was having a lot of pain, and after a week, her pain still was not better.”
Dr. Mauskar brought the patient into the office and ultimately diagnosed a squamous cell carcinoma. “What I thought was a normal erosion was actually an ulcerated plaque,” she said.
Like Dr. Venkatesan, Dr. Mauskar has found that image quality can be uneven. Photographs may be out of focus. Video visits have been a mixed bag. Some are successful. Other times, Dr. Mauskar has to tell the patient she needs to see her in the office.
Certain clinical scenarios require a vaginal exam, Dr. Venkatesan noted. Although some type of assessment may be possible if a patient is with a primary care provider during the telemedicine visit, the examination may not be equivalent. Doctors also should anticipate where a patient might go to have a biopsy if one is necessary.
Another telemedicine caveat pertains to patient counseling. When using store-and-forward telemedicine systems, advising patients in a written report can be challenging. “Is there an easy way ... to counsel patients how to apply their topical medications?” Dr. Venkatesan said.
Excellent care is possible
Vulvology is a small part of Dr. Venkatesan’s general dermatology practice, which has used telemedicine extensively since the pandemic.
In recent years, Dr. Venkatesan’s clinic began encouraging providers in their health system to submit photographs with referrals. “That has really paid off now because we have been able to help provide a lot of excellent quality care for patients without them having to come in,” she said. “We may be able to say: ‘These are excellent photos. We know what this patient has. We can manage it. They don’t need to come see us in person.’ ” That could be the case for certain types of acne, eczema, and psoriasis.
In other cases, they may be able to provide initial advice remotely but still want to see the patient. For a patient with severe acne, “I may be able to tell the referring doctor: ‘Please start the patient on these three medicines. It will take 2 months for those medicines to start working and then we will plan to have an in-person dermatology visit.’ ” In this case, telemedicine essentially replaces one in-person visit.
If photographs are poor, the differential diagnosis is broad, a procedure is required, the doctor needs to touch the lesion, or more involved history taking or counseling are required, the patient may need to go into the office.
Beyond its public health advantages during a pandemic, telemedicine can improve access for patients who live far away, lack transportation, or are unable to take time off from work. It also can decrease patient wait times. “Once we started doing some telemedicine work … we went from having a 5-month wait time for patients to see us in person to a 72-hour wait time for providing some care for patients if they had good photos as part of their referral,” Dr. Venkatesan said.
Telemedicine has been used in inpatient and outpatient dermatology settings. Primary care providers who consult with dermatologists using a store-and-forward telemedicine system may improve their dermatology knowledge and feel more confident in their ability to diagnose and manage dermatologic conditions, research indicates.
In obstetrics and gynecology, telemedicine may play a role in preconception, contraception, and medical abortion care, prenatal visits, well-woman exams, mental health, and pre- and postoperative counseling, a recent review suggests.
Image quality is key
“Quality of the image is so critical for being able to provide good care, especially in such a visual exam field as dermatology,” Dr. Venkatesan said.
To that end, doctors have offered recommendations on how to photograph skin conditions. A guide shared by the mobile telehealth system company ClickMedix suggests focusing on the area of importance, capturing the extent of involvement, and including involved and uninvolved areas.
Good lighting and checking the image resolution can help, Dr. Venkatesan offered. Nevertheless, patients may have difficulty photographing themselves. If a patient is with their primary care doctor, “we are much more likely to be able to get good quality photos,” she said.
Dr. Venkatesan is a paid consultant for DirectDerm, a store-and-forward teledermatology company. Dr. Mauskar had no relevant disclosures.
FROM THE ISSVD BIENNIAL CONFERENCE
Models for Implementing Buprenorphine Treatment in the VHA
STRENGTH trial questions CV benefit of high-dose omega-3s
Questions about the cardiovascular benefit of omega-3 fatty acids and the high-dose eicosapentaenoic acid (EPA) product, icosapent ethyl (Vascepa, Amarin), have resurfaced with the presentation and publication of the STRENGTH trial using a combined high-dose omega-3 fatty acid product.
The STRENGTH trial showed no benefit on cardiovascular event rates of a high-dose combination of EPA and docosahexaenoic acid (DHA) in a new branded product (Epanova, AstraZeneca).
It was announced in January that the trial was being stopped because of a low likelihood of showing any benefit.
Full results were presented Nov. 15 at the virtual American Heart Association scientific sessions and simultaneously published online in JAMA.
These results showed similar cardiovascular event rates with the high-dose EPA/DHA product and placebo, with a hazard ratio for the primary endpoint of 0.99. In addition to no benefit, more adverse effects occurred in the active treatment arm, with a higher rate of gastrointestinal adverse events and atrial fibrillation.
“We found no benefit of a high-dose combination of EPA and DHA. Despite a 270% to 300% increase in EPA, the hazard curves for the active and placebo groups were superimposable,” STRENGTH investigator A. Michael Lincoff, MD, of the Cleveland Clinic, said at the AHA meeting.
The big question is how the negative results of the STRENGTH trial can be reconciled with the very positive results of the REDUCE-IT trial, which showed an impressive 25% relative risk reduction in major adverse cardiovascular events with the high-dose purified EPA product, icosapent ethyl.
Dr. Lincoff proposed several possible explanations for the different results between these two trials, although he cautioned that all explanations were speculative.
The one explanation that Dr. Lincoff highlighted in particular was the different placebos used in the two trials. REDUCE-IT used a placebo of mineral oil, which Dr. Lincoff noted increases LDL, apolipoprotein B, and high-sensitivity C-reactive protein, whereas the corn oil placebo used in STRENGTH “is truly neutral on a broad range lipid and cardiovascular biomarkers,” he said.
“It must therefore be considered that at least part of the benefit in REDUCE-IT is due to an increase in adverse cardiovascular event rate in the control arm, and our results from STRENGTH cast uncertainly on the net benefit or harm of any omega-3 fatty acid preparation,” Dr. Lincoff said.
Asked whether he used omega-3 fatty acids in his practice, Dr. Lincoff replied, “Aside from patients with triglycerides greater than 500 – for which there is other evidence of benefit – I do not routinely prescribe omega-3 fatty acids. For the reasons discussed, I think there are questions about whether the risks and benefits have a favorable ratio.”
Asked at an AHA press conference what advice he would give to other physicians on the use of Vascepa, Dr. Lincoff said, “On the one hand, we could take the REDUCE-IT study results at face value, but there are potential concerns on the construct of that trial and whether the effects were exaggerated. That having been said, the [Food and Drug Administration] has approved that initial indication, so this is not a straightforward issue of whether or not that trial result is valid.
“What I would like to see is a trial in future with a clearly neutral comparator. It’s hard to recommend taking your patients off Vascepa now, but I have a high threshold at this point to start patients on it because of these concerns,” he added.
A “manufactured controversy”
The lead investigator of the REDUCE-IT trial, Deepak L. Bhatt, MD, professor of medicine, Harvard Medical School, Boston, described Dr. Lincoff’s comments as “absurd.”
In an interview, he said the Japanese JELIS trial, while having some limitations, also showed a benefit of icosapent ethyl, which “in the context of this manufactured controversy about the mineral oil placebo in REDUCE-IT, completely rebuts concerns about the placebo in REDUCE-IT being toxic.”
Dr. Bhatt also suggested that DHA may counter some of the benefits of EPA. “It appears that the STRENGTH trial leadership is trying to stir up controversy, rather than just reporting objectively that they have a negative trial,” he added.
Dr. Lincoff outlined other possible explanations for the difference between the two trials.
He noted that icosapent ethyl increased levels of EPA by 45% in REDUCE-IT more than did the combined product used in STRENGTH. “But this moderate difference seems insufficient to account for the markedly different results of the two trials,” Dr. Lincoff added, “and both trials showed a 19% reduction in triglycerides, suggesting they have similar biochemical effects.”
There is also the possibility of an adverse effect of DHA, he noted, “but this has never been seen in previous studies.”
Another explanation could be differences in trial populations, with REDUCE-IT including more patients with established cardiovascular disease. “But the results were no different in this group compared to the patients without established cardiovascular disease, so this explanation is unlikely,” Dr. Lincoff suggested.
STRENGTH trial
The STRENGTH trial included 13,078 statin-treated participants with high cardiovascular risk, hypertriglyceridemia, and low levels of HDL cholesterol from 22 countries.
They were randomly assigned to a 4 g per day of carboxylic acid formulation of EPA and DHA or to corn oil as an inert comparator.
The trial was halted when 1,384 patients had experienced a primary endpoint event (of a planned 1,600 events), based on an interim analysis that indicated a low probability of clinical benefit of the active treatment.
At this point, the primary efficacy endpoint – a composite of cardiovascular death, nonfatal myocardial infarction, nonfatal stroke, coronary revascularization, or unstable angina requiring hospitalization – occurred in 12.0% of patients treated with the omega-3 product vs. 12.2% of those who received corn oil (hazard ratio [HR], 0.99; P = .84).
A greater rate of gastrointestinal adverse events was observed in the omega-3 group (24.7%) than in corn oil–treated patients (14.7%). An increased rate of new-onset atrial fibrillation was also observed in the omega-3 group (2.2% vs 1.3%; HR, 1.69).
Uncertainty prevails
The moderator of an AHA press conference at which the STRENGTH trial was discussed, Amit Khera, MD, professor of medicine and director of preventive cardiology at UT Southwestern Medical Center, Dallas, said in an interview that questions about how Vascepa brought about the benefits shown in REDUCE-IT have been ongoing since that trial was published.
“I think for now, we have to accept the REDUCE-IT results as a positive finding. However, the STRENGTH trial did amplify these questions a bit since there was no signal at all for benefit, and this heightens the call for additional trials of high-dose EPA formulations, including icosapent ethyl, versus corn oil or another neutral comparator,” he said.
Discussant of the STRENGTH trial at the AHA late-breaker session, Alberico Catapano, MD, PhD, University of Milan, noted that DHA may have less biological activity than EPA.
“We don’t know for certain, but there are studies suggesting that EPA may have more effect on stabilizing plaque membranes,” Dr. Catapano said. “Certainly, the dose of EPA was different in the two studies, and in my view this could be part of the explanation. But we are still in place where we need more evidence.”
In an editorial accompanying the JAMA publication of STRENGTH, Garima Sharma, MD, Seth S. Martin, MD, and Roger S. Blumenthal, MD, Johns Hopkins University, Baltimore, said the trial’s findings “may invigorate further investigation regarding IPE [icosapent ethyl], generate additional constructive debate around the optimal placebo control, and should prompt reconsideration of over-the-counter mixed omega-3 fatty acid products for ASCVD [atherosclerotic cardiovascular disease] prevention.
“This latter point is especially important given the lack of evidence for benefit, and the potential for harm due to increased AF [atrial fibrillation],” they noted.
“The reasons the findings from the REDUCE-IT trial were positive and the STRENGTH trial were not, and that EPA levels correlated with outcomes in REDUCE-IT but did not in STRENGTH, remain uncertain,” they concluded. “The importance of the specific omega-3 formulation in achieving ASCVD risk reduction and the degree to which the placebo (i.e., mineral oil vs corn oil) may have affected outcomes remain unresolved.”
The STRENGTH trial was sponsored by AstraZeneca. Dr. Lincoff reported receiving grants from AstraZeneca during the conduct of the study. Dr. Catapano has received honoraria, lecture fees, or research grants from Sigma-Tau, Manarini, Kowa Pharmaceuticals, Recordati, Eli Lilly, AstraZeneca, Mediolanum, Pfizer, Merck, Sanofi, Aegerion, Amgen, Genzyme, Bayer, Sanofi, Regeneron Daiichi Sankyo, and Amarin. Dr. Martin reports receiving consulting fees from AstraZeneca, Amgen, Esperion, and REGENXBIO, and has a patent pending for a system of LDL-C estimation filed by Johns Hopkins University. Dr. Bhatt reports serving as principal investigator for REDUCE-IT and that Brigham and Women’s Hospital has received research funding from Amarin.
A version of this article originally appeared on Medscape.com.
Questions about the cardiovascular benefit of omega-3 fatty acids and the high-dose eicosapentaenoic acid (EPA) product, icosapent ethyl (Vascepa, Amarin), have resurfaced with the presentation and publication of the STRENGTH trial using a combined high-dose omega-3 fatty acid product.
The STRENGTH trial showed no benefit on cardiovascular event rates of a high-dose combination of EPA and docosahexaenoic acid (DHA) in a new branded product (Epanova, AstraZeneca).
It was announced in January that the trial was being stopped because of a low likelihood of showing any benefit.
Full results were presented Nov. 15 at the virtual American Heart Association scientific sessions and simultaneously published online in JAMA.
These results showed similar cardiovascular event rates with the high-dose EPA/DHA product and placebo, with a hazard ratio for the primary endpoint of 0.99. In addition to no benefit, more adverse effects occurred in the active treatment arm, with a higher rate of gastrointestinal adverse events and atrial fibrillation.
“We found no benefit of a high-dose combination of EPA and DHA. Despite a 270% to 300% increase in EPA, the hazard curves for the active and placebo groups were superimposable,” STRENGTH investigator A. Michael Lincoff, MD, of the Cleveland Clinic, said at the AHA meeting.
The big question is how the negative results of the STRENGTH trial can be reconciled with the very positive results of the REDUCE-IT trial, which showed an impressive 25% relative risk reduction in major adverse cardiovascular events with the high-dose purified EPA product, icosapent ethyl.
Dr. Lincoff proposed several possible explanations for the different results between these two trials, although he cautioned that all explanations were speculative.
The one explanation that Dr. Lincoff highlighted in particular was the different placebos used in the two trials. REDUCE-IT used a placebo of mineral oil, which Dr. Lincoff noted increases LDL, apolipoprotein B, and high-sensitivity C-reactive protein, whereas the corn oil placebo used in STRENGTH “is truly neutral on a broad range lipid and cardiovascular biomarkers,” he said.
“It must therefore be considered that at least part of the benefit in REDUCE-IT is due to an increase in adverse cardiovascular event rate in the control arm, and our results from STRENGTH cast uncertainly on the net benefit or harm of any omega-3 fatty acid preparation,” Dr. Lincoff said.
Asked whether he used omega-3 fatty acids in his practice, Dr. Lincoff replied, “Aside from patients with triglycerides greater than 500 – for which there is other evidence of benefit – I do not routinely prescribe omega-3 fatty acids. For the reasons discussed, I think there are questions about whether the risks and benefits have a favorable ratio.”
Asked at an AHA press conference what advice he would give to other physicians on the use of Vascepa, Dr. Lincoff said, “On the one hand, we could take the REDUCE-IT study results at face value, but there are potential concerns on the construct of that trial and whether the effects were exaggerated. That having been said, the [Food and Drug Administration] has approved that initial indication, so this is not a straightforward issue of whether or not that trial result is valid.
“What I would like to see is a trial in future with a clearly neutral comparator. It’s hard to recommend taking your patients off Vascepa now, but I have a high threshold at this point to start patients on it because of these concerns,” he added.
A “manufactured controversy”
The lead investigator of the REDUCE-IT trial, Deepak L. Bhatt, MD, professor of medicine, Harvard Medical School, Boston, described Dr. Lincoff’s comments as “absurd.”
In an interview, he said the Japanese JELIS trial, while having some limitations, also showed a benefit of icosapent ethyl, which “in the context of this manufactured controversy about the mineral oil placebo in REDUCE-IT, completely rebuts concerns about the placebo in REDUCE-IT being toxic.”
Dr. Bhatt also suggested that DHA may counter some of the benefits of EPA. “It appears that the STRENGTH trial leadership is trying to stir up controversy, rather than just reporting objectively that they have a negative trial,” he added.
Dr. Lincoff outlined other possible explanations for the difference between the two trials.
He noted that icosapent ethyl increased levels of EPA by 45% in REDUCE-IT more than did the combined product used in STRENGTH. “But this moderate difference seems insufficient to account for the markedly different results of the two trials,” Dr. Lincoff added, “and both trials showed a 19% reduction in triglycerides, suggesting they have similar biochemical effects.”
There is also the possibility of an adverse effect of DHA, he noted, “but this has never been seen in previous studies.”
Another explanation could be differences in trial populations, with REDUCE-IT including more patients with established cardiovascular disease. “But the results were no different in this group compared to the patients without established cardiovascular disease, so this explanation is unlikely,” Dr. Lincoff suggested.
STRENGTH trial
The STRENGTH trial included 13,078 statin-treated participants with high cardiovascular risk, hypertriglyceridemia, and low levels of HDL cholesterol from 22 countries.
They were randomly assigned to a 4 g per day of carboxylic acid formulation of EPA and DHA or to corn oil as an inert comparator.
The trial was halted when 1,384 patients had experienced a primary endpoint event (of a planned 1,600 events), based on an interim analysis that indicated a low probability of clinical benefit of the active treatment.
At this point, the primary efficacy endpoint – a composite of cardiovascular death, nonfatal myocardial infarction, nonfatal stroke, coronary revascularization, or unstable angina requiring hospitalization – occurred in 12.0% of patients treated with the omega-3 product vs. 12.2% of those who received corn oil (hazard ratio [HR], 0.99; P = .84).
A greater rate of gastrointestinal adverse events was observed in the omega-3 group (24.7%) than in corn oil–treated patients (14.7%). An increased rate of new-onset atrial fibrillation was also observed in the omega-3 group (2.2% vs 1.3%; HR, 1.69).
Uncertainty prevails
The moderator of an AHA press conference at which the STRENGTH trial was discussed, Amit Khera, MD, professor of medicine and director of preventive cardiology at UT Southwestern Medical Center, Dallas, said in an interview that questions about how Vascepa brought about the benefits shown in REDUCE-IT have been ongoing since that trial was published.
“I think for now, we have to accept the REDUCE-IT results as a positive finding. However, the STRENGTH trial did amplify these questions a bit since there was no signal at all for benefit, and this heightens the call for additional trials of high-dose EPA formulations, including icosapent ethyl, versus corn oil or another neutral comparator,” he said.
Discussant of the STRENGTH trial at the AHA late-breaker session, Alberico Catapano, MD, PhD, University of Milan, noted that DHA may have less biological activity than EPA.
“We don’t know for certain, but there are studies suggesting that EPA may have more effect on stabilizing plaque membranes,” Dr. Catapano said. “Certainly, the dose of EPA was different in the two studies, and in my view this could be part of the explanation. But we are still in place where we need more evidence.”
In an editorial accompanying the JAMA publication of STRENGTH, Garima Sharma, MD, Seth S. Martin, MD, and Roger S. Blumenthal, MD, Johns Hopkins University, Baltimore, said the trial’s findings “may invigorate further investigation regarding IPE [icosapent ethyl], generate additional constructive debate around the optimal placebo control, and should prompt reconsideration of over-the-counter mixed omega-3 fatty acid products for ASCVD [atherosclerotic cardiovascular disease] prevention.
“This latter point is especially important given the lack of evidence for benefit, and the potential for harm due to increased AF [atrial fibrillation],” they noted.
“The reasons the findings from the REDUCE-IT trial were positive and the STRENGTH trial were not, and that EPA levels correlated with outcomes in REDUCE-IT but did not in STRENGTH, remain uncertain,” they concluded. “The importance of the specific omega-3 formulation in achieving ASCVD risk reduction and the degree to which the placebo (i.e., mineral oil vs corn oil) may have affected outcomes remain unresolved.”
The STRENGTH trial was sponsored by AstraZeneca. Dr. Lincoff reported receiving grants from AstraZeneca during the conduct of the study. Dr. Catapano has received honoraria, lecture fees, or research grants from Sigma-Tau, Manarini, Kowa Pharmaceuticals, Recordati, Eli Lilly, AstraZeneca, Mediolanum, Pfizer, Merck, Sanofi, Aegerion, Amgen, Genzyme, Bayer, Sanofi, Regeneron Daiichi Sankyo, and Amarin. Dr. Martin reports receiving consulting fees from AstraZeneca, Amgen, Esperion, and REGENXBIO, and has a patent pending for a system of LDL-C estimation filed by Johns Hopkins University. Dr. Bhatt reports serving as principal investigator for REDUCE-IT and that Brigham and Women’s Hospital has received research funding from Amarin.
A version of this article originally appeared on Medscape.com.
Questions about the cardiovascular benefit of omega-3 fatty acids and the high-dose eicosapentaenoic acid (EPA) product, icosapent ethyl (Vascepa, Amarin), have resurfaced with the presentation and publication of the STRENGTH trial using a combined high-dose omega-3 fatty acid product.
The STRENGTH trial showed no benefit on cardiovascular event rates of a high-dose combination of EPA and docosahexaenoic acid (DHA) in a new branded product (Epanova, AstraZeneca).
It was announced in January that the trial was being stopped because of a low likelihood of showing any benefit.
Full results were presented Nov. 15 at the virtual American Heart Association scientific sessions and simultaneously published online in JAMA.
These results showed similar cardiovascular event rates with the high-dose EPA/DHA product and placebo, with a hazard ratio for the primary endpoint of 0.99. In addition to no benefit, more adverse effects occurred in the active treatment arm, with a higher rate of gastrointestinal adverse events and atrial fibrillation.
“We found no benefit of a high-dose combination of EPA and DHA. Despite a 270% to 300% increase in EPA, the hazard curves for the active and placebo groups were superimposable,” STRENGTH investigator A. Michael Lincoff, MD, of the Cleveland Clinic, said at the AHA meeting.
The big question is how the negative results of the STRENGTH trial can be reconciled with the very positive results of the REDUCE-IT trial, which showed an impressive 25% relative risk reduction in major adverse cardiovascular events with the high-dose purified EPA product, icosapent ethyl.
Dr. Lincoff proposed several possible explanations for the different results between these two trials, although he cautioned that all explanations were speculative.
The one explanation that Dr. Lincoff highlighted in particular was the different placebos used in the two trials. REDUCE-IT used a placebo of mineral oil, which Dr. Lincoff noted increases LDL, apolipoprotein B, and high-sensitivity C-reactive protein, whereas the corn oil placebo used in STRENGTH “is truly neutral on a broad range lipid and cardiovascular biomarkers,” he said.
“It must therefore be considered that at least part of the benefit in REDUCE-IT is due to an increase in adverse cardiovascular event rate in the control arm, and our results from STRENGTH cast uncertainly on the net benefit or harm of any omega-3 fatty acid preparation,” Dr. Lincoff said.
Asked whether he used omega-3 fatty acids in his practice, Dr. Lincoff replied, “Aside from patients with triglycerides greater than 500 – for which there is other evidence of benefit – I do not routinely prescribe omega-3 fatty acids. For the reasons discussed, I think there are questions about whether the risks and benefits have a favorable ratio.”
Asked at an AHA press conference what advice he would give to other physicians on the use of Vascepa, Dr. Lincoff said, “On the one hand, we could take the REDUCE-IT study results at face value, but there are potential concerns on the construct of that trial and whether the effects were exaggerated. That having been said, the [Food and Drug Administration] has approved that initial indication, so this is not a straightforward issue of whether or not that trial result is valid.
“What I would like to see is a trial in future with a clearly neutral comparator. It’s hard to recommend taking your patients off Vascepa now, but I have a high threshold at this point to start patients on it because of these concerns,” he added.
A “manufactured controversy”
The lead investigator of the REDUCE-IT trial, Deepak L. Bhatt, MD, professor of medicine, Harvard Medical School, Boston, described Dr. Lincoff’s comments as “absurd.”
In an interview, he said the Japanese JELIS trial, while having some limitations, also showed a benefit of icosapent ethyl, which “in the context of this manufactured controversy about the mineral oil placebo in REDUCE-IT, completely rebuts concerns about the placebo in REDUCE-IT being toxic.”
Dr. Bhatt also suggested that DHA may counter some of the benefits of EPA. “It appears that the STRENGTH trial leadership is trying to stir up controversy, rather than just reporting objectively that they have a negative trial,” he added.
Dr. Lincoff outlined other possible explanations for the difference between the two trials.
He noted that icosapent ethyl increased levels of EPA by 45% in REDUCE-IT more than did the combined product used in STRENGTH. “But this moderate difference seems insufficient to account for the markedly different results of the two trials,” Dr. Lincoff added, “and both trials showed a 19% reduction in triglycerides, suggesting they have similar biochemical effects.”
There is also the possibility of an adverse effect of DHA, he noted, “but this has never been seen in previous studies.”
Another explanation could be differences in trial populations, with REDUCE-IT including more patients with established cardiovascular disease. “But the results were no different in this group compared to the patients without established cardiovascular disease, so this explanation is unlikely,” Dr. Lincoff suggested.
STRENGTH trial
The STRENGTH trial included 13,078 statin-treated participants with high cardiovascular risk, hypertriglyceridemia, and low levels of HDL cholesterol from 22 countries.
They were randomly assigned to a 4 g per day of carboxylic acid formulation of EPA and DHA or to corn oil as an inert comparator.
The trial was halted when 1,384 patients had experienced a primary endpoint event (of a planned 1,600 events), based on an interim analysis that indicated a low probability of clinical benefit of the active treatment.
At this point, the primary efficacy endpoint – a composite of cardiovascular death, nonfatal myocardial infarction, nonfatal stroke, coronary revascularization, or unstable angina requiring hospitalization – occurred in 12.0% of patients treated with the omega-3 product vs. 12.2% of those who received corn oil (hazard ratio [HR], 0.99; P = .84).
A greater rate of gastrointestinal adverse events was observed in the omega-3 group (24.7%) than in corn oil–treated patients (14.7%). An increased rate of new-onset atrial fibrillation was also observed in the omega-3 group (2.2% vs 1.3%; HR, 1.69).
Uncertainty prevails
The moderator of an AHA press conference at which the STRENGTH trial was discussed, Amit Khera, MD, professor of medicine and director of preventive cardiology at UT Southwestern Medical Center, Dallas, said in an interview that questions about how Vascepa brought about the benefits shown in REDUCE-IT have been ongoing since that trial was published.
“I think for now, we have to accept the REDUCE-IT results as a positive finding. However, the STRENGTH trial did amplify these questions a bit since there was no signal at all for benefit, and this heightens the call for additional trials of high-dose EPA formulations, including icosapent ethyl, versus corn oil or another neutral comparator,” he said.
Discussant of the STRENGTH trial at the AHA late-breaker session, Alberico Catapano, MD, PhD, University of Milan, noted that DHA may have less biological activity than EPA.
“We don’t know for certain, but there are studies suggesting that EPA may have more effect on stabilizing plaque membranes,” Dr. Catapano said. “Certainly, the dose of EPA was different in the two studies, and in my view this could be part of the explanation. But we are still in place where we need more evidence.”
In an editorial accompanying the JAMA publication of STRENGTH, Garima Sharma, MD, Seth S. Martin, MD, and Roger S. Blumenthal, MD, Johns Hopkins University, Baltimore, said the trial’s findings “may invigorate further investigation regarding IPE [icosapent ethyl], generate additional constructive debate around the optimal placebo control, and should prompt reconsideration of over-the-counter mixed omega-3 fatty acid products for ASCVD [atherosclerotic cardiovascular disease] prevention.
“This latter point is especially important given the lack of evidence for benefit, and the potential for harm due to increased AF [atrial fibrillation],” they noted.
“The reasons the findings from the REDUCE-IT trial were positive and the STRENGTH trial were not, and that EPA levels correlated with outcomes in REDUCE-IT but did not in STRENGTH, remain uncertain,” they concluded. “The importance of the specific omega-3 formulation in achieving ASCVD risk reduction and the degree to which the placebo (i.e., mineral oil vs corn oil) may have affected outcomes remain unresolved.”
The STRENGTH trial was sponsored by AstraZeneca. Dr. Lincoff reported receiving grants from AstraZeneca during the conduct of the study. Dr. Catapano has received honoraria, lecture fees, or research grants from Sigma-Tau, Manarini, Kowa Pharmaceuticals, Recordati, Eli Lilly, AstraZeneca, Mediolanum, Pfizer, Merck, Sanofi, Aegerion, Amgen, Genzyme, Bayer, Sanofi, Regeneron Daiichi Sankyo, and Amarin. Dr. Martin reports receiving consulting fees from AstraZeneca, Amgen, Esperion, and REGENXBIO, and has a patent pending for a system of LDL-C estimation filed by Johns Hopkins University. Dr. Bhatt reports serving as principal investigator for REDUCE-IT and that Brigham and Women’s Hospital has received research funding from Amarin.
A version of this article originally appeared on Medscape.com.
Pembrolizumab approved for triple-negative breast cancer
The Food and Drug Administration has granted accelerated approval for pembrolizumab (Keytruda) in combination with chemotherapy to treat locally recurrent unresectable or metastatic triple-negative breast cancer (TNBC) that expresses PD-L1, as determined by a combined positive score of 10 or greater on an FDA-approved assay.
The FDA also approved a PD-L1 assay for selecting TNBC patients for pembrolizumab, the PD-L1 IHC 22C3 pharmDx.
Pembrolizumab is approved for numerous indications in the United States, but the new approval is its first breast cancer indication.
The accelerated approval for pembrolizumab in TNBC was based on progression-free survival (PFS) in the KEYNOTE-355 trial. The FDA noted that continued approval of pembrolizumab in TNBC “may be contingent upon verification and description of clinical benefit in the confirmatory trials.”
KEYNOTE-355 enrolled patients with locally recurrent unresectable or metastatic TNBC who had not received chemotherapy in the metastatic setting. Patients were randomized to chemotherapy (nab-paclitaxel, paclitaxel, or gemcitabine plus carboplatin) plus placebo (n = 281) or chemotherapy plus pembrolizumab at 200 mg on day 1 every 3 weeks (n = 562).
Among PD-L1-positive patients (n = 323), the median PFS was 5.6 months in the placebo arm and 9.7 months in the pembrolizumab arm (hazard ratio, 0.65; P = .0012).
The recommended pembrolizumab dose in TNBC is 200 mg every 3 weeks or 400 mg every 6 weeks administered prior to chemotherapy until disease progression, unacceptable toxicity, or up to 24 months.
Pembrolizumab can cause immune-mediated adverse reactions that may be severe or fatal, according to Merck, the manufacturer of pembrolizumab. These adverse reactions include pneumonitis, colitis, hepatitis, endocrinopathies, nephritis, severe skin reactions, solid organ transplant rejection, and complications of allogeneic hematopoietic stem cell transplant.
“Based on the severity of the adverse reaction, [pembrolizumab] should be withheld or discontinued and corticosteroids administered if appropriate,” the company noted.
For more details on pembrolizumab, see the full prescribing information.
The Food and Drug Administration has granted accelerated approval for pembrolizumab (Keytruda) in combination with chemotherapy to treat locally recurrent unresectable or metastatic triple-negative breast cancer (TNBC) that expresses PD-L1, as determined by a combined positive score of 10 or greater on an FDA-approved assay.
The FDA also approved a PD-L1 assay for selecting TNBC patients for pembrolizumab, the PD-L1 IHC 22C3 pharmDx.
Pembrolizumab is approved for numerous indications in the United States, but the new approval is its first breast cancer indication.
The accelerated approval for pembrolizumab in TNBC was based on progression-free survival (PFS) in the KEYNOTE-355 trial. The FDA noted that continued approval of pembrolizumab in TNBC “may be contingent upon verification and description of clinical benefit in the confirmatory trials.”
KEYNOTE-355 enrolled patients with locally recurrent unresectable or metastatic TNBC who had not received chemotherapy in the metastatic setting. Patients were randomized to chemotherapy (nab-paclitaxel, paclitaxel, or gemcitabine plus carboplatin) plus placebo (n = 281) or chemotherapy plus pembrolizumab at 200 mg on day 1 every 3 weeks (n = 562).
Among PD-L1-positive patients (n = 323), the median PFS was 5.6 months in the placebo arm and 9.7 months in the pembrolizumab arm (hazard ratio, 0.65; P = .0012).
The recommended pembrolizumab dose in TNBC is 200 mg every 3 weeks or 400 mg every 6 weeks administered prior to chemotherapy until disease progression, unacceptable toxicity, or up to 24 months.
Pembrolizumab can cause immune-mediated adverse reactions that may be severe or fatal, according to Merck, the manufacturer of pembrolizumab. These adverse reactions include pneumonitis, colitis, hepatitis, endocrinopathies, nephritis, severe skin reactions, solid organ transplant rejection, and complications of allogeneic hematopoietic stem cell transplant.
“Based on the severity of the adverse reaction, [pembrolizumab] should be withheld or discontinued and corticosteroids administered if appropriate,” the company noted.
For more details on pembrolizumab, see the full prescribing information.
The Food and Drug Administration has granted accelerated approval for pembrolizumab (Keytruda) in combination with chemotherapy to treat locally recurrent unresectable or metastatic triple-negative breast cancer (TNBC) that expresses PD-L1, as determined by a combined positive score of 10 or greater on an FDA-approved assay.
The FDA also approved a PD-L1 assay for selecting TNBC patients for pembrolizumab, the PD-L1 IHC 22C3 pharmDx.
Pembrolizumab is approved for numerous indications in the United States, but the new approval is its first breast cancer indication.
The accelerated approval for pembrolizumab in TNBC was based on progression-free survival (PFS) in the KEYNOTE-355 trial. The FDA noted that continued approval of pembrolizumab in TNBC “may be contingent upon verification and description of clinical benefit in the confirmatory trials.”
KEYNOTE-355 enrolled patients with locally recurrent unresectable or metastatic TNBC who had not received chemotherapy in the metastatic setting. Patients were randomized to chemotherapy (nab-paclitaxel, paclitaxel, or gemcitabine plus carboplatin) plus placebo (n = 281) or chemotherapy plus pembrolizumab at 200 mg on day 1 every 3 weeks (n = 562).
Among PD-L1-positive patients (n = 323), the median PFS was 5.6 months in the placebo arm and 9.7 months in the pembrolizumab arm (hazard ratio, 0.65; P = .0012).
The recommended pembrolizumab dose in TNBC is 200 mg every 3 weeks or 400 mg every 6 weeks administered prior to chemotherapy until disease progression, unacceptable toxicity, or up to 24 months.
Pembrolizumab can cause immune-mediated adverse reactions that may be severe or fatal, according to Merck, the manufacturer of pembrolizumab. These adverse reactions include pneumonitis, colitis, hepatitis, endocrinopathies, nephritis, severe skin reactions, solid organ transplant rejection, and complications of allogeneic hematopoietic stem cell transplant.
“Based on the severity of the adverse reaction, [pembrolizumab] should be withheld or discontinued and corticosteroids administered if appropriate,” the company noted.
For more details on pembrolizumab, see the full prescribing information.
FROM THE FOOD AND DRUG ADMINISTRATION
Evinacumab, novel lipid-lowerer, extends promise in phase 2 results
Treatment with evinacumab, an investigational lipid-lowering drug with a novel mechanism of action, safely led to roughly a halving of LDL cholesterol levels in patients with treatment-refractory hypercholesterolemia in a multicenter, phase 2 study of 272 patients treated for 16 weeks.
The study enrolled patients with either heterozygous familial hypercholesterolemia (FH) (72% of patients), or patients with hypercholesterolemia and clinical evidence of atherosclerotic cardiovascular disease who had failed to reached their recommended level of LDL cholesterol by treatment (when tolerated) with a statin, ezetimibe, and a PCSK9 (proprotein convertase subtilisin/kexin type 9) inhibitor.
Notably, only 8 of the 272 randomized patients entered the study not on treatment with a PCSK9 inhibitor. Despite these background treatments, all enrolled patients were above their goal LDL-cholesterol level, with an average level of 148 mg/dL.
The study’s primary endpoint was the percent change from baseline in LDL cholesterol after 16 weeks compared with placebo among patients treated with subcutaneous drug delivery either weekly or every other week, and among patients treated with intravenous delivery every 4 weeks. Results of the dose-ranging study showed that the highest subcutaneous dosage tested produced a 56% cut in LDL cholesterol, while the highest IV dosage led to a 51% drop, Robert S. Rosenson, MD, said at the virtual scientific sessions of the American Heart Association. Concurrently with his report, the results were published online in the New England Journal of Medicine.
The drug’s safety among 194 patients who received evinacumab was “reassuring,” said Dr. Rosenson, professor of medicine and director of the cardiometabolic disorders unit at Icahn School of Medicine at Mount Sinai in New York. “I see no concerning signals in the safety profile,” he said in an interview, an assessment that other experts shared.
“Safety looks pretty good. I don’t see any major concerns,” said lipidologist and endocrinologist Anne C. Goldberg, MD, professor of medicine at Washington University in St. Louis. The LDL-cholesterol effect shown was “very, very impressive in these hard to treat patients,” added Dr. Goldberg, who was a coinvestigator on the study.
“Nothing stands out” as a safety concern in the new data, agreed Robert H. Eckel, MD, an endocrinologist and lipid specialist at the University of Colorado in Aurora.
Drug’s unique mechanism extends potential benefits
The phase 2 study included dose-ranging assessments of both subcutaneous and intravenous treatment with evinacumab, a fully human monoclonal antibody against angiopoietin-like 3, an enzyme that inhibits two different lipases involved in metabolizing LDL cholesterol and other lipoproteins including triglycerides. When the drug inhibits angiopoietin-like 3, the lipases remain more active and further reduce levels of their target lipoproteins.
“The powerful contribution of this drug is that it works by a pathway independent of the LDL receptor,” said Dr. Rosenson.
By this mechanism evinacumab cut not only LDL cholesterol, but also lowered triglycerides by 53%-62% at the highest dosages, an effect seen as a potential plus. “Prospects are favorable for a drug that not only lowers atherogenic lipoproteins but also lowers triglycerides [TGs]. That’s a distinguishing feature of this treatment,” compared with other agents that lower LDL cholesterol, Dr. Rosenson said. It could make evinacumab especially attractive for treating patients with diabetes, who often have elevated TG levels, he noted. But Dr. Eckel cautioned that a clinical benefit directly linked to TG lowering has not yet been proven.
The drug also cut HDL cholesterol by an average of as much as 31% from baseline, though the consequence of this effect isn’t clear. “I’m not worried about the HDL levels,” said Dr. Goldberg, who noted that changes in HDL cholesterol produced by drug treatment have often not shown discernible effects.
Reaching goals by IV or subcutaneous delivery
Another measure of evinacumab’s efficacy was the percentage of patients who fell below the LDL-cholesterol threshold of 70 mg/dL set by recommendations of the American Heart Association and American College of Cardiology for the highest risk patients, and the less than 55 mg/dL goal set for similar patients by the European Society of Cardiology. Among the subcutaneously-treated patients, 64% achieved the goal of less than 70 mg/dL, and 49% hit the goal of less than 55 mg/dL. Among those who received IV treatment, 71% fell below the 70 mg/dL threshold, and 50% dropped below 55 mg/dL.
The good efficacy shown with subcutaneous dosing is critical, noted Dr. Eckel, as this represents a new dimension for evinacumab that had previously been tested only as an IV agent in patients with homozygous FH (N Engl J Med. 2020 Aug 20;383[8]:711-20).
“Subcutaneous delivery is needed for wide real world use,” Dr. Eckel noted in an interview.
Evinacumab’s role hangs on further studies
The path that evinacumab takes from here into U.S. practice is not yet clear, said Dr. Rosenson. He cited the approval earlier in 2020 of another LDL-lowering drug, bempedoic acid (Nexletol) that received U.S. regulatory approval for a similar patient population after studies that proved only lipid-lowering safety and efficacy, without any clinical-endpoint data. He wondered: “Will the [Food and Drug Administration] require a cardiovascular outcomes trial” for evinacumab?
The growing experience using the PCSK9 inhibitor antibodies to treat hyperlipidemia has made clinicians comfortable with this general approach to lipid management, but if evinacumab never accumulates similar efficacy evidence that may relegate it to the backseat compared with the PCSK9 inhibitors for quite some time, suggested Dr. Goldberg, though she said she’d be willing to prescribe evinacumab to selected patients based on lipid-lowering evidence alone.
By providing an alternative mechanism for lipid lowering, evinacumab can serve as a useful add-on for patients not reaching their LDL-cholesterol goal with more established agents, thereby providing an alternative to LDL apheresis, which now serves as the lipid-lowering therapy of last resort, said both Dr. Rosenson and Dr. Goldberg.
The study was sponsored by Regeneron, the company developing evinacumab. Dr. Rosenson has been a consultant to Regeneron, and has also been a consultant to or received research funding from Amgen, 89Bio, Corvidia, CVS Caremark, Kowa, Novartis, and The Medicines Company. Dr. Goldberg has received research grants, personal fees, and nonfinancial support from Regeneron and Sanofi, research grants from Amarin, Amgen, Ionis/AKCEA, Novartis, and Pfizer, and personal fees from AKCEA, Esperion, Merck and Novartis. Dr. Eckel has been a consultant to KOWA and Novo Nordisk.
Treatment with evinacumab, an investigational lipid-lowering drug with a novel mechanism of action, safely led to roughly a halving of LDL cholesterol levels in patients with treatment-refractory hypercholesterolemia in a multicenter, phase 2 study of 272 patients treated for 16 weeks.
The study enrolled patients with either heterozygous familial hypercholesterolemia (FH) (72% of patients), or patients with hypercholesterolemia and clinical evidence of atherosclerotic cardiovascular disease who had failed to reached their recommended level of LDL cholesterol by treatment (when tolerated) with a statin, ezetimibe, and a PCSK9 (proprotein convertase subtilisin/kexin type 9) inhibitor.
Notably, only 8 of the 272 randomized patients entered the study not on treatment with a PCSK9 inhibitor. Despite these background treatments, all enrolled patients were above their goal LDL-cholesterol level, with an average level of 148 mg/dL.
The study’s primary endpoint was the percent change from baseline in LDL cholesterol after 16 weeks compared with placebo among patients treated with subcutaneous drug delivery either weekly or every other week, and among patients treated with intravenous delivery every 4 weeks. Results of the dose-ranging study showed that the highest subcutaneous dosage tested produced a 56% cut in LDL cholesterol, while the highest IV dosage led to a 51% drop, Robert S. Rosenson, MD, said at the virtual scientific sessions of the American Heart Association. Concurrently with his report, the results were published online in the New England Journal of Medicine.
The drug’s safety among 194 patients who received evinacumab was “reassuring,” said Dr. Rosenson, professor of medicine and director of the cardiometabolic disorders unit at Icahn School of Medicine at Mount Sinai in New York. “I see no concerning signals in the safety profile,” he said in an interview, an assessment that other experts shared.
“Safety looks pretty good. I don’t see any major concerns,” said lipidologist and endocrinologist Anne C. Goldberg, MD, professor of medicine at Washington University in St. Louis. The LDL-cholesterol effect shown was “very, very impressive in these hard to treat patients,” added Dr. Goldberg, who was a coinvestigator on the study.
“Nothing stands out” as a safety concern in the new data, agreed Robert H. Eckel, MD, an endocrinologist and lipid specialist at the University of Colorado in Aurora.
Drug’s unique mechanism extends potential benefits
The phase 2 study included dose-ranging assessments of both subcutaneous and intravenous treatment with evinacumab, a fully human monoclonal antibody against angiopoietin-like 3, an enzyme that inhibits two different lipases involved in metabolizing LDL cholesterol and other lipoproteins including triglycerides. When the drug inhibits angiopoietin-like 3, the lipases remain more active and further reduce levels of their target lipoproteins.
“The powerful contribution of this drug is that it works by a pathway independent of the LDL receptor,” said Dr. Rosenson.
By this mechanism evinacumab cut not only LDL cholesterol, but also lowered triglycerides by 53%-62% at the highest dosages, an effect seen as a potential plus. “Prospects are favorable for a drug that not only lowers atherogenic lipoproteins but also lowers triglycerides [TGs]. That’s a distinguishing feature of this treatment,” compared with other agents that lower LDL cholesterol, Dr. Rosenson said. It could make evinacumab especially attractive for treating patients with diabetes, who often have elevated TG levels, he noted. But Dr. Eckel cautioned that a clinical benefit directly linked to TG lowering has not yet been proven.
The drug also cut HDL cholesterol by an average of as much as 31% from baseline, though the consequence of this effect isn’t clear. “I’m not worried about the HDL levels,” said Dr. Goldberg, who noted that changes in HDL cholesterol produced by drug treatment have often not shown discernible effects.
Reaching goals by IV or subcutaneous delivery
Another measure of evinacumab’s efficacy was the percentage of patients who fell below the LDL-cholesterol threshold of 70 mg/dL set by recommendations of the American Heart Association and American College of Cardiology for the highest risk patients, and the less than 55 mg/dL goal set for similar patients by the European Society of Cardiology. Among the subcutaneously-treated patients, 64% achieved the goal of less than 70 mg/dL, and 49% hit the goal of less than 55 mg/dL. Among those who received IV treatment, 71% fell below the 70 mg/dL threshold, and 50% dropped below 55 mg/dL.
The good efficacy shown with subcutaneous dosing is critical, noted Dr. Eckel, as this represents a new dimension for evinacumab that had previously been tested only as an IV agent in patients with homozygous FH (N Engl J Med. 2020 Aug 20;383[8]:711-20).
“Subcutaneous delivery is needed for wide real world use,” Dr. Eckel noted in an interview.
Evinacumab’s role hangs on further studies
The path that evinacumab takes from here into U.S. practice is not yet clear, said Dr. Rosenson. He cited the approval earlier in 2020 of another LDL-lowering drug, bempedoic acid (Nexletol) that received U.S. regulatory approval for a similar patient population after studies that proved only lipid-lowering safety and efficacy, without any clinical-endpoint data. He wondered: “Will the [Food and Drug Administration] require a cardiovascular outcomes trial” for evinacumab?
The growing experience using the PCSK9 inhibitor antibodies to treat hyperlipidemia has made clinicians comfortable with this general approach to lipid management, but if evinacumab never accumulates similar efficacy evidence that may relegate it to the backseat compared with the PCSK9 inhibitors for quite some time, suggested Dr. Goldberg, though she said she’d be willing to prescribe evinacumab to selected patients based on lipid-lowering evidence alone.
By providing an alternative mechanism for lipid lowering, evinacumab can serve as a useful add-on for patients not reaching their LDL-cholesterol goal with more established agents, thereby providing an alternative to LDL apheresis, which now serves as the lipid-lowering therapy of last resort, said both Dr. Rosenson and Dr. Goldberg.
The study was sponsored by Regeneron, the company developing evinacumab. Dr. Rosenson has been a consultant to Regeneron, and has also been a consultant to or received research funding from Amgen, 89Bio, Corvidia, CVS Caremark, Kowa, Novartis, and The Medicines Company. Dr. Goldberg has received research grants, personal fees, and nonfinancial support from Regeneron and Sanofi, research grants from Amarin, Amgen, Ionis/AKCEA, Novartis, and Pfizer, and personal fees from AKCEA, Esperion, Merck and Novartis. Dr. Eckel has been a consultant to KOWA and Novo Nordisk.
Treatment with evinacumab, an investigational lipid-lowering drug with a novel mechanism of action, safely led to roughly a halving of LDL cholesterol levels in patients with treatment-refractory hypercholesterolemia in a multicenter, phase 2 study of 272 patients treated for 16 weeks.
The study enrolled patients with either heterozygous familial hypercholesterolemia (FH) (72% of patients), or patients with hypercholesterolemia and clinical evidence of atherosclerotic cardiovascular disease who had failed to reached their recommended level of LDL cholesterol by treatment (when tolerated) with a statin, ezetimibe, and a PCSK9 (proprotein convertase subtilisin/kexin type 9) inhibitor.
Notably, only 8 of the 272 randomized patients entered the study not on treatment with a PCSK9 inhibitor. Despite these background treatments, all enrolled patients were above their goal LDL-cholesterol level, with an average level of 148 mg/dL.
The study’s primary endpoint was the percent change from baseline in LDL cholesterol after 16 weeks compared with placebo among patients treated with subcutaneous drug delivery either weekly or every other week, and among patients treated with intravenous delivery every 4 weeks. Results of the dose-ranging study showed that the highest subcutaneous dosage tested produced a 56% cut in LDL cholesterol, while the highest IV dosage led to a 51% drop, Robert S. Rosenson, MD, said at the virtual scientific sessions of the American Heart Association. Concurrently with his report, the results were published online in the New England Journal of Medicine.
The drug’s safety among 194 patients who received evinacumab was “reassuring,” said Dr. Rosenson, professor of medicine and director of the cardiometabolic disorders unit at Icahn School of Medicine at Mount Sinai in New York. “I see no concerning signals in the safety profile,” he said in an interview, an assessment that other experts shared.
“Safety looks pretty good. I don’t see any major concerns,” said lipidologist and endocrinologist Anne C. Goldberg, MD, professor of medicine at Washington University in St. Louis. The LDL-cholesterol effect shown was “very, very impressive in these hard to treat patients,” added Dr. Goldberg, who was a coinvestigator on the study.
“Nothing stands out” as a safety concern in the new data, agreed Robert H. Eckel, MD, an endocrinologist and lipid specialist at the University of Colorado in Aurora.
Drug’s unique mechanism extends potential benefits
The phase 2 study included dose-ranging assessments of both subcutaneous and intravenous treatment with evinacumab, a fully human monoclonal antibody against angiopoietin-like 3, an enzyme that inhibits two different lipases involved in metabolizing LDL cholesterol and other lipoproteins including triglycerides. When the drug inhibits angiopoietin-like 3, the lipases remain more active and further reduce levels of their target lipoproteins.
“The powerful contribution of this drug is that it works by a pathway independent of the LDL receptor,” said Dr. Rosenson.
By this mechanism evinacumab cut not only LDL cholesterol, but also lowered triglycerides by 53%-62% at the highest dosages, an effect seen as a potential plus. “Prospects are favorable for a drug that not only lowers atherogenic lipoproteins but also lowers triglycerides [TGs]. That’s a distinguishing feature of this treatment,” compared with other agents that lower LDL cholesterol, Dr. Rosenson said. It could make evinacumab especially attractive for treating patients with diabetes, who often have elevated TG levels, he noted. But Dr. Eckel cautioned that a clinical benefit directly linked to TG lowering has not yet been proven.
The drug also cut HDL cholesterol by an average of as much as 31% from baseline, though the consequence of this effect isn’t clear. “I’m not worried about the HDL levels,” said Dr. Goldberg, who noted that changes in HDL cholesterol produced by drug treatment have often not shown discernible effects.
Reaching goals by IV or subcutaneous delivery
Another measure of evinacumab’s efficacy was the percentage of patients who fell below the LDL-cholesterol threshold of 70 mg/dL set by recommendations of the American Heart Association and American College of Cardiology for the highest risk patients, and the less than 55 mg/dL goal set for similar patients by the European Society of Cardiology. Among the subcutaneously-treated patients, 64% achieved the goal of less than 70 mg/dL, and 49% hit the goal of less than 55 mg/dL. Among those who received IV treatment, 71% fell below the 70 mg/dL threshold, and 50% dropped below 55 mg/dL.
The good efficacy shown with subcutaneous dosing is critical, noted Dr. Eckel, as this represents a new dimension for evinacumab that had previously been tested only as an IV agent in patients with homozygous FH (N Engl J Med. 2020 Aug 20;383[8]:711-20).
“Subcutaneous delivery is needed for wide real world use,” Dr. Eckel noted in an interview.
Evinacumab’s role hangs on further studies
The path that evinacumab takes from here into U.S. practice is not yet clear, said Dr. Rosenson. He cited the approval earlier in 2020 of another LDL-lowering drug, bempedoic acid (Nexletol) that received U.S. regulatory approval for a similar patient population after studies that proved only lipid-lowering safety and efficacy, without any clinical-endpoint data. He wondered: “Will the [Food and Drug Administration] require a cardiovascular outcomes trial” for evinacumab?
The growing experience using the PCSK9 inhibitor antibodies to treat hyperlipidemia has made clinicians comfortable with this general approach to lipid management, but if evinacumab never accumulates similar efficacy evidence that may relegate it to the backseat compared with the PCSK9 inhibitors for quite some time, suggested Dr. Goldberg, though she said she’d be willing to prescribe evinacumab to selected patients based on lipid-lowering evidence alone.
By providing an alternative mechanism for lipid lowering, evinacumab can serve as a useful add-on for patients not reaching their LDL-cholesterol goal with more established agents, thereby providing an alternative to LDL apheresis, which now serves as the lipid-lowering therapy of last resort, said both Dr. Rosenson and Dr. Goldberg.
The study was sponsored by Regeneron, the company developing evinacumab. Dr. Rosenson has been a consultant to Regeneron, and has also been a consultant to or received research funding from Amgen, 89Bio, Corvidia, CVS Caremark, Kowa, Novartis, and The Medicines Company. Dr. Goldberg has received research grants, personal fees, and nonfinancial support from Regeneron and Sanofi, research grants from Amarin, Amgen, Ionis/AKCEA, Novartis, and Pfizer, and personal fees from AKCEA, Esperion, Merck and Novartis. Dr. Eckel has been a consultant to KOWA and Novo Nordisk.
AHA 2020
Chronic inflammatory diseases vary widely in CHD risk
Not all chronic systemic inflammatory diseases are equal enhancers of atherosclerotic cardiovascular disease risk, according to a large case-control study.
Current AHA/American College of Cardiology guidelines cite three chronic inflammatory diseases as atherosclerotic cardiovascular disease risk enhancers: rheumatoid arthritis, psoriasis, and HIV infection. But this study of those three diseases, along with three others marked by elevated high sensitivity C-reactive protein (systemic sclerosis, inflammatory bowel disease, and systemic lupus erythematosus [SLE]), showed that chronic inflammatory diseases are not monolithic in terms of their associated risk of incident coronary heart disease (CHD).
Indeed, two of the six inflammatory diseases – psoriasis and inflammatory bowel disease – turned out to be not at all associated with increased cardiovascular risk in the 37,117-patient study. The highest-risk disease was SLE, not specifically mentioned in the guidelines, Arjun Sinha, MD, a cardiology fellow at Northwestern University, Chicago, noted in his presentation at the virtual American Heart Association scientific sessions.
The study included 18,129 patients with one of the six chronic inflammatory diseases and 18,988 matched controls, none with CHD at baseline. All regularly received outpatient care at Northwestern during 2000-2019. There were 1,011 incident CHD events during a median of 3.5 years of follow-up.
In a Cox proportional hazards analysis adjusted for demographics, insurance status, hypertension, diabetes, current smoking, total cholesterol, and estimated glomerular filtration rate, here’s how the chronic inflammatory diseases stacked up in terms of incident CHD and MI risks:
- SLE: hazard ratio for CHD, 2.85; for MI, 4.76.
- Systemic sclerosis: HR for CHD, 2.14; for MI, 3.19.
- HIV: HR for CHD, 1.38; for MI, 1.69.
- Rheumatoid arthritis: HR for CHD, 1.22; for MI, 1.45.
- Psoriasis: no significant increase.
- Inflammatory bowel disease: no significant increase.
In an exploratory analysis, Dr. Sinha and coinvestigators evaluated the risk of incident CHD stratified by disease severity. For lack of standardized disease severity scales, the investigators relied upon tertiles of CD4 T cell count in the HIV group and CRP in the others. The HR for new-onset CHD in the more than 5,000 patients with psoriasis didn’t vary by CRP tertile. However, there was a nonsignificant trend for greater disease severity, as reflected by CRP tertile, to be associated with increased incident CHD risk in the HIV and inflammatory bowel disease groups.
In contrast, patients with rheumatoid arthritis or systemic sclerosis who were in the top CRP tertile had a significantly greater risk of developing CHD than that of controls, with HRs of 2.11 in the rheumatoid arthritis group and 4.59 with systemic sclerosis, although patients in the other two tertiles weren’t at significantly increased risk. But all three tertiles of CRP in patients with SLE were associated with significantly increased CHD risk: 3.17-fold in the lowest tertile of lupus severity, 5.38-fold in the middle tertile, and 4.04-fold in the top tertile for inflammation.
These findings could be used in clinical practice to fine-tune atherosclerotic cardiovascular disease risk assessment based upon chronic inflammatory disease type and severity. That’s information which in turn can help guide the timing and intensity of preventive therapy for patients with each disease type.
But studying the association between chronic systemic inflammatory diseases and CHD risk can be useful in additional ways, according to Dr. Sinha. These inflammatory diseases can serve as models of atherosclerosis that shed light on the non–lipid-related mechanisms involved in cardiovascular disease.
“The gradient in risk may be hypothesis-generating with respect to which specific inflammatory pathways may contribute to CHD,” he explained.
Each of these six chronic inflammatory diseases is characterized by a different form of major immune dysfunction, Dr. Sinha continued. A case in point is SLE, the inflammatory disease associated with the highest risk of CHD and MI. Lupus is characterized by a form of neutrophil dysfunction marked by increased formation and reduced degradation of neutrophil extracellular traps, or NETs, as well as by an increase in autoreactive B cells and dysfunctional CD4+ T helper cells. The increase in NETs of of particular interest because NETs have also been shown to contribute to the development of atherosclerosis, endothelial dysfunction, plaque erosion, and thrombosis.
In another exploratory analysis, Dr. Sinha and coworkers found that SLE patients with a neutrophil count above the median level were twice as likely to develop CHD than were those with a neutrophil count below the median.
A better understanding of the upstream pathways linking NET formation in SLE and atherosclerosis could lead to development of new or repurposed medications that target immune dysfunction in order to curb atherosclerosis, said Dr. Sinha, whose study won the AHA’s Samuel A. Levine Early Career Clinical Investigator Award.
He reported having no financial conflicts regarding his study.
Not all chronic systemic inflammatory diseases are equal enhancers of atherosclerotic cardiovascular disease risk, according to a large case-control study.
Current AHA/American College of Cardiology guidelines cite three chronic inflammatory diseases as atherosclerotic cardiovascular disease risk enhancers: rheumatoid arthritis, psoriasis, and HIV infection. But this study of those three diseases, along with three others marked by elevated high sensitivity C-reactive protein (systemic sclerosis, inflammatory bowel disease, and systemic lupus erythematosus [SLE]), showed that chronic inflammatory diseases are not monolithic in terms of their associated risk of incident coronary heart disease (CHD).
Indeed, two of the six inflammatory diseases – psoriasis and inflammatory bowel disease – turned out to be not at all associated with increased cardiovascular risk in the 37,117-patient study. The highest-risk disease was SLE, not specifically mentioned in the guidelines, Arjun Sinha, MD, a cardiology fellow at Northwestern University, Chicago, noted in his presentation at the virtual American Heart Association scientific sessions.
The study included 18,129 patients with one of the six chronic inflammatory diseases and 18,988 matched controls, none with CHD at baseline. All regularly received outpatient care at Northwestern during 2000-2019. There were 1,011 incident CHD events during a median of 3.5 years of follow-up.
In a Cox proportional hazards analysis adjusted for demographics, insurance status, hypertension, diabetes, current smoking, total cholesterol, and estimated glomerular filtration rate, here’s how the chronic inflammatory diseases stacked up in terms of incident CHD and MI risks:
- SLE: hazard ratio for CHD, 2.85; for MI, 4.76.
- Systemic sclerosis: HR for CHD, 2.14; for MI, 3.19.
- HIV: HR for CHD, 1.38; for MI, 1.69.
- Rheumatoid arthritis: HR for CHD, 1.22; for MI, 1.45.
- Psoriasis: no significant increase.
- Inflammatory bowel disease: no significant increase.
In an exploratory analysis, Dr. Sinha and coinvestigators evaluated the risk of incident CHD stratified by disease severity. For lack of standardized disease severity scales, the investigators relied upon tertiles of CD4 T cell count in the HIV group and CRP in the others. The HR for new-onset CHD in the more than 5,000 patients with psoriasis didn’t vary by CRP tertile. However, there was a nonsignificant trend for greater disease severity, as reflected by CRP tertile, to be associated with increased incident CHD risk in the HIV and inflammatory bowel disease groups.
In contrast, patients with rheumatoid arthritis or systemic sclerosis who were in the top CRP tertile had a significantly greater risk of developing CHD than that of controls, with HRs of 2.11 in the rheumatoid arthritis group and 4.59 with systemic sclerosis, although patients in the other two tertiles weren’t at significantly increased risk. But all three tertiles of CRP in patients with SLE were associated with significantly increased CHD risk: 3.17-fold in the lowest tertile of lupus severity, 5.38-fold in the middle tertile, and 4.04-fold in the top tertile for inflammation.
These findings could be used in clinical practice to fine-tune atherosclerotic cardiovascular disease risk assessment based upon chronic inflammatory disease type and severity. That’s information which in turn can help guide the timing and intensity of preventive therapy for patients with each disease type.
But studying the association between chronic systemic inflammatory diseases and CHD risk can be useful in additional ways, according to Dr. Sinha. These inflammatory diseases can serve as models of atherosclerosis that shed light on the non–lipid-related mechanisms involved in cardiovascular disease.
“The gradient in risk may be hypothesis-generating with respect to which specific inflammatory pathways may contribute to CHD,” he explained.
Each of these six chronic inflammatory diseases is characterized by a different form of major immune dysfunction, Dr. Sinha continued. A case in point is SLE, the inflammatory disease associated with the highest risk of CHD and MI. Lupus is characterized by a form of neutrophil dysfunction marked by increased formation and reduced degradation of neutrophil extracellular traps, or NETs, as well as by an increase in autoreactive B cells and dysfunctional CD4+ T helper cells. The increase in NETs of of particular interest because NETs have also been shown to contribute to the development of atherosclerosis, endothelial dysfunction, plaque erosion, and thrombosis.
In another exploratory analysis, Dr. Sinha and coworkers found that SLE patients with a neutrophil count above the median level were twice as likely to develop CHD than were those with a neutrophil count below the median.
A better understanding of the upstream pathways linking NET formation in SLE and atherosclerosis could lead to development of new or repurposed medications that target immune dysfunction in order to curb atherosclerosis, said Dr. Sinha, whose study won the AHA’s Samuel A. Levine Early Career Clinical Investigator Award.
He reported having no financial conflicts regarding his study.
Not all chronic systemic inflammatory diseases are equal enhancers of atherosclerotic cardiovascular disease risk, according to a large case-control study.
Current AHA/American College of Cardiology guidelines cite three chronic inflammatory diseases as atherosclerotic cardiovascular disease risk enhancers: rheumatoid arthritis, psoriasis, and HIV infection. But this study of those three diseases, along with three others marked by elevated high sensitivity C-reactive protein (systemic sclerosis, inflammatory bowel disease, and systemic lupus erythematosus [SLE]), showed that chronic inflammatory diseases are not monolithic in terms of their associated risk of incident coronary heart disease (CHD).
Indeed, two of the six inflammatory diseases – psoriasis and inflammatory bowel disease – turned out to be not at all associated with increased cardiovascular risk in the 37,117-patient study. The highest-risk disease was SLE, not specifically mentioned in the guidelines, Arjun Sinha, MD, a cardiology fellow at Northwestern University, Chicago, noted in his presentation at the virtual American Heart Association scientific sessions.
The study included 18,129 patients with one of the six chronic inflammatory diseases and 18,988 matched controls, none with CHD at baseline. All regularly received outpatient care at Northwestern during 2000-2019. There were 1,011 incident CHD events during a median of 3.5 years of follow-up.
In a Cox proportional hazards analysis adjusted for demographics, insurance status, hypertension, diabetes, current smoking, total cholesterol, and estimated glomerular filtration rate, here’s how the chronic inflammatory diseases stacked up in terms of incident CHD and MI risks:
- SLE: hazard ratio for CHD, 2.85; for MI, 4.76.
- Systemic sclerosis: HR for CHD, 2.14; for MI, 3.19.
- HIV: HR for CHD, 1.38; for MI, 1.69.
- Rheumatoid arthritis: HR for CHD, 1.22; for MI, 1.45.
- Psoriasis: no significant increase.
- Inflammatory bowel disease: no significant increase.
In an exploratory analysis, Dr. Sinha and coinvestigators evaluated the risk of incident CHD stratified by disease severity. For lack of standardized disease severity scales, the investigators relied upon tertiles of CD4 T cell count in the HIV group and CRP in the others. The HR for new-onset CHD in the more than 5,000 patients with psoriasis didn’t vary by CRP tertile. However, there was a nonsignificant trend for greater disease severity, as reflected by CRP tertile, to be associated with increased incident CHD risk in the HIV and inflammatory bowel disease groups.
In contrast, patients with rheumatoid arthritis or systemic sclerosis who were in the top CRP tertile had a significantly greater risk of developing CHD than that of controls, with HRs of 2.11 in the rheumatoid arthritis group and 4.59 with systemic sclerosis, although patients in the other two tertiles weren’t at significantly increased risk. But all three tertiles of CRP in patients with SLE were associated with significantly increased CHD risk: 3.17-fold in the lowest tertile of lupus severity, 5.38-fold in the middle tertile, and 4.04-fold in the top tertile for inflammation.
These findings could be used in clinical practice to fine-tune atherosclerotic cardiovascular disease risk assessment based upon chronic inflammatory disease type and severity. That’s information which in turn can help guide the timing and intensity of preventive therapy for patients with each disease type.
But studying the association between chronic systemic inflammatory diseases and CHD risk can be useful in additional ways, according to Dr. Sinha. These inflammatory diseases can serve as models of atherosclerosis that shed light on the non–lipid-related mechanisms involved in cardiovascular disease.
“The gradient in risk may be hypothesis-generating with respect to which specific inflammatory pathways may contribute to CHD,” he explained.
Each of these six chronic inflammatory diseases is characterized by a different form of major immune dysfunction, Dr. Sinha continued. A case in point is SLE, the inflammatory disease associated with the highest risk of CHD and MI. Lupus is characterized by a form of neutrophil dysfunction marked by increased formation and reduced degradation of neutrophil extracellular traps, or NETs, as well as by an increase in autoreactive B cells and dysfunctional CD4+ T helper cells. The increase in NETs of of particular interest because NETs have also been shown to contribute to the development of atherosclerosis, endothelial dysfunction, plaque erosion, and thrombosis.
In another exploratory analysis, Dr. Sinha and coworkers found that SLE patients with a neutrophil count above the median level were twice as likely to develop CHD than were those with a neutrophil count below the median.
A better understanding of the upstream pathways linking NET formation in SLE and atherosclerosis could lead to development of new or repurposed medications that target immune dysfunction in order to curb atherosclerosis, said Dr. Sinha, whose study won the AHA’s Samuel A. Levine Early Career Clinical Investigator Award.
He reported having no financial conflicts regarding his study.
FROM AHA 2020