User login
MDedge Daily News: Specialists warn Congress on Medicare Part B drug payments
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
Atrial fibrosis could be a key trigger for arrhythmia and stroke, how walnuts and the microbiome may cut cardiovascular risk, why some inflammatory bowel disease patients benefit by lighting up, and specialists warn Congress to protect Medicare Part B drug payments.
Listen to the MDedge Daily News podcast for all the details on today’s top news.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
Atrial fibrosis could be a key trigger for arrhythmia and stroke, how walnuts and the microbiome may cut cardiovascular risk, why some inflammatory bowel disease patients benefit by lighting up, and specialists warn Congress to protect Medicare Part B drug payments.
Listen to the MDedge Daily News podcast for all the details on today’s top news.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
Atrial fibrosis could be a key trigger for arrhythmia and stroke, how walnuts and the microbiome may cut cardiovascular risk, why some inflammatory bowel disease patients benefit by lighting up, and specialists warn Congress to protect Medicare Part B drug payments.
Listen to the MDedge Daily News podcast for all the details on today’s top news.
Closer to a Vaccine For a ‘Pandemic in Progress’?
A series of National Institute of Health (NIH) clinical trials are bringing Zika virus vaccines closer to the public.
According to preliminary findings from 3 phase 1 clinical trials, an investigational Zika purified inactivated virus (ZPIV) vaccine was well tolerated and induced an immune response. Scientists from Walter Reed Army Institute of Research are developing the vaccine and leading 1 of the trials.
Of 67 adult participants, 55 received the investigational vaccine; 12 received placebo. All participants received 2 intramuscular injections 4 weeks apart. The researchers detected antibodies in > 90% of those who received the vaccine, 4 weeks after the last dose.
In phase 2 clinical trials, 2 versions of an experimental gene-based Zika vaccine, developed by scientists at the National Institute of Allergy and Infectious Diseases, were both found to be safe and to induce an immune response. One candidate showed “the most promise,” paving the way for an international phase 2/2b safety and efficacy trial, which began in 2017 and will last for 2 years.
“This trial marks a significant milestone in our efforts to develop countermeasures for a pandemic in progress,” said Anthony Fauci, MD, NIAID director
A series of National Institute of Health (NIH) clinical trials are bringing Zika virus vaccines closer to the public.
According to preliminary findings from 3 phase 1 clinical trials, an investigational Zika purified inactivated virus (ZPIV) vaccine was well tolerated and induced an immune response. Scientists from Walter Reed Army Institute of Research are developing the vaccine and leading 1 of the trials.
Of 67 adult participants, 55 received the investigational vaccine; 12 received placebo. All participants received 2 intramuscular injections 4 weeks apart. The researchers detected antibodies in > 90% of those who received the vaccine, 4 weeks after the last dose.
In phase 2 clinical trials, 2 versions of an experimental gene-based Zika vaccine, developed by scientists at the National Institute of Allergy and Infectious Diseases, were both found to be safe and to induce an immune response. One candidate showed “the most promise,” paving the way for an international phase 2/2b safety and efficacy trial, which began in 2017 and will last for 2 years.
“This trial marks a significant milestone in our efforts to develop countermeasures for a pandemic in progress,” said Anthony Fauci, MD, NIAID director
A series of National Institute of Health (NIH) clinical trials are bringing Zika virus vaccines closer to the public.
According to preliminary findings from 3 phase 1 clinical trials, an investigational Zika purified inactivated virus (ZPIV) vaccine was well tolerated and induced an immune response. Scientists from Walter Reed Army Institute of Research are developing the vaccine and leading 1 of the trials.
Of 67 adult participants, 55 received the investigational vaccine; 12 received placebo. All participants received 2 intramuscular injections 4 weeks apart. The researchers detected antibodies in > 90% of those who received the vaccine, 4 weeks after the last dose.
In phase 2 clinical trials, 2 versions of an experimental gene-based Zika vaccine, developed by scientists at the National Institute of Allergy and Infectious Diseases, were both found to be safe and to induce an immune response. One candidate showed “the most promise,” paving the way for an international phase 2/2b safety and efficacy trial, which began in 2017 and will last for 2 years.
“This trial marks a significant milestone in our efforts to develop countermeasures for a pandemic in progress,” said Anthony Fauci, MD, NIAID director
IV bevacizumab improves severe bleeding in HHT
Intravenous (IV) bevacizumab “dramatically” improves severe bleeding associated with hereditary hemorrhagic telangiectasia (HHT), according to researchers.
In a retrospective study, HHT patients with severe bleeding had a substantial reduction in nose bleeds and gastrointestinal (GI) bleeding after treatment with IV bevacizumab.
In addition, patients were able to stop or considerably reduce red blood cell (RBC) transfusions.
The researchers therefore believe that IV bevacizumab should be considered as a first-line therapy for the treatment of refractory bleeding in patients with severe HHT.
The team detailed this research and in Mayo Clinic Proceedings alongside a related editorial.
“Some HHT patients suffer from severe epistaxis and gastrointestinal bleeding, which can result in severe anemia and years of blood transfusions,” said study author Vivek N. Iyer, MD, of the Mayo Clinic in Rochester, Minnesota.
“Both problems also appear to sometimes worsen with age. In some patients, both epistaxis and GI bleeding can become refractory/resistant to existing treatment options, leaving patients severely anemic and dependent on iron infusions or blood transfusions. Quality of life is very poor in these cases.”
With this in mind, Dr Iyer and his colleagues analyzed the records of 34 patients who were treated with IV bevacizumab for severe HHT-related bleeding from June 2013 through January 2017.
Patient characteristics
Patients had a median age of 63 (range, 57 to 72). Sixty-two percent of patients were female.
The primary source of bleeding was epistaxis in 15 patients, GI bleeding in 4 patients, and combined epistaxis and GI bleeding in 15 patients.
Prior epistaxis treatments included potassium-titanyl-phosphate/other laser procedures (62%), sclerotherapy (26%), endovascular angiographic embolization (21%), septodermoplasty (24%), subcutaneous bevacizumab injections (21%), and bevacizumab nasal spray (29%).
Seventy-one percent of patients underwent upper endoscopy (100% of these with telangiectasias), and 53% underwent colonoscopy (56% of these with telangiectasias).
Forty-one percent of patients had IV iron supplementation in the past 6 months.
Twenty-eight patients had received RBC transfusions. Sixteen patients were transfusion-dependent and had received a median of 75 transfusions before starting treatment with bevacizumab. The median duration of transfusion dependence was 6 years.
Treatment
The typical initial dosing cycle of bevacizumab consisted of 8 doses (4 doses each administered 2 weeks apart, followed by 4 doses each administered 1 month apart) over a period of around 22 weeks.
Further maintenance doses after the initial dosing cycle were individualized in each patient.
At last follow-up, 3 patients were still receiving bevacizumab from the initial dosing protocol. For the 31 patients who had completed the first dosing cycle, the median duration of follow-up was 13.6 months.
Eighteen patients required at least 1 top-up dose of IV bevacizumab because of worsening bleeding and/or anemia.
Efficacy
The median follow-up was 17.6 months (range, 3 to 42.5 months).
An Epistaxis Severity Score (ESS) questionnaire was used to assess the severity of nose bleeds both at the beginning of the study and after starting bevacizumab.
One month after starting treatment, there was a significant reduction in ESS scores (P<0.001). This improvement was maintained after patients completed the initial treatment cycle.
The median ESS score was 6.5 at baseline, 3.3 at 1 month, 4.0 at 3 months, 2.3 at the end of the first cycle, 2.0 at 1 to 3 months after the first cycle, 3.2 at 4 to 6 months, and 2.8 at 7 to 12 months.
GI bleeding also improved, with resolution or improvement of anemia in all 19 patients with this condition.
There was a reduction in RBC transfusions as well. The proportion of patients receiving transfusions was 53% in the 6 months before IV bevacizumab, 15% in the 1 to 3 months after starting IV bevacizumab, 14% at 4 to 6 months, 8% at 7 to 9 months, and 9% at 9 to 12 months.
Eighty-eight percent (n=14) of the transfusion-dependent patients had received a transfusion in the 6 months prior to starting IV bevacizumab. This compares to 31% (n=5) of patients in the 1 to 3 months after the start of treatment, 29% (n=4) at 4 to 6 months, 14% (n=2) at 7 to 9 months, and 8% (n=1) at 9 to 12 months.
Safety
Four patients had hypertension (HTN). One had pre-existing HTN and had to double the daily dose of lisinopril from 10 mg to 20 mg.
Two HTN patients had to start antihypertensive medications. The fourth patient experienced hypertensive urgency with a temporary decline in renal function. However, the patient was able to resume bevacizumab.
Two patients had infusion-related chills and fever, but premedication with acetaminophen and diphenhydramine prevented these events from recurring.
Three patients died during follow-up. Causes of death were stroke, infective endocarditis (methicillin-sensitive Staphylococcus aureus) with multiple cerebral infarcts, and postoperative respiratory failure (after left atrial appendectomy for paroxysmal atrial fibrillation).
None of these deaths were directly linked to bevacizumab.
“This study provides good-quality evidence for the excellent efficacy and safety of intravenous bevacizumab in the treatment of these patients,” Dr Iyer said. “Intravenous bevacizumab should be considered as a standard, first-line treatment option for HHT patients with severe bleeding and transfusion-dependent anemia.” ![]()
Intravenous (IV) bevacizumab “dramatically” improves severe bleeding associated with hereditary hemorrhagic telangiectasia (HHT), according to researchers.
In a retrospective study, HHT patients with severe bleeding had a substantial reduction in nose bleeds and gastrointestinal (GI) bleeding after treatment with IV bevacizumab.
In addition, patients were able to stop or considerably reduce red blood cell (RBC) transfusions.
The researchers therefore believe that IV bevacizumab should be considered as a first-line therapy for the treatment of refractory bleeding in patients with severe HHT.
The team detailed this research and in Mayo Clinic Proceedings alongside a related editorial.
“Some HHT patients suffer from severe epistaxis and gastrointestinal bleeding, which can result in severe anemia and years of blood transfusions,” said study author Vivek N. Iyer, MD, of the Mayo Clinic in Rochester, Minnesota.
“Both problems also appear to sometimes worsen with age. In some patients, both epistaxis and GI bleeding can become refractory/resistant to existing treatment options, leaving patients severely anemic and dependent on iron infusions or blood transfusions. Quality of life is very poor in these cases.”
With this in mind, Dr Iyer and his colleagues analyzed the records of 34 patients who were treated with IV bevacizumab for severe HHT-related bleeding from June 2013 through January 2017.
Patient characteristics
Patients had a median age of 63 (range, 57 to 72). Sixty-two percent of patients were female.
The primary source of bleeding was epistaxis in 15 patients, GI bleeding in 4 patients, and combined epistaxis and GI bleeding in 15 patients.
Prior epistaxis treatments included potassium-titanyl-phosphate/other laser procedures (62%), sclerotherapy (26%), endovascular angiographic embolization (21%), septodermoplasty (24%), subcutaneous bevacizumab injections (21%), and bevacizumab nasal spray (29%).
Seventy-one percent of patients underwent upper endoscopy (100% of these with telangiectasias), and 53% underwent colonoscopy (56% of these with telangiectasias).
Forty-one percent of patients had IV iron supplementation in the past 6 months.
Twenty-eight patients had received RBC transfusions. Sixteen patients were transfusion-dependent and had received a median of 75 transfusions before starting treatment with bevacizumab. The median duration of transfusion dependence was 6 years.
Treatment
The typical initial dosing cycle of bevacizumab consisted of 8 doses (4 doses each administered 2 weeks apart, followed by 4 doses each administered 1 month apart) over a period of around 22 weeks.
Further maintenance doses after the initial dosing cycle were individualized in each patient.
At last follow-up, 3 patients were still receiving bevacizumab from the initial dosing protocol. For the 31 patients who had completed the first dosing cycle, the median duration of follow-up was 13.6 months.
Eighteen patients required at least 1 top-up dose of IV bevacizumab because of worsening bleeding and/or anemia.
Efficacy
The median follow-up was 17.6 months (range, 3 to 42.5 months).
An Epistaxis Severity Score (ESS) questionnaire was used to assess the severity of nose bleeds both at the beginning of the study and after starting bevacizumab.
One month after starting treatment, there was a significant reduction in ESS scores (P<0.001). This improvement was maintained after patients completed the initial treatment cycle.
The median ESS score was 6.5 at baseline, 3.3 at 1 month, 4.0 at 3 months, 2.3 at the end of the first cycle, 2.0 at 1 to 3 months after the first cycle, 3.2 at 4 to 6 months, and 2.8 at 7 to 12 months.
GI bleeding also improved, with resolution or improvement of anemia in all 19 patients with this condition.
There was a reduction in RBC transfusions as well. The proportion of patients receiving transfusions was 53% in the 6 months before IV bevacizumab, 15% in the 1 to 3 months after starting IV bevacizumab, 14% at 4 to 6 months, 8% at 7 to 9 months, and 9% at 9 to 12 months.
Eighty-eight percent (n=14) of the transfusion-dependent patients had received a transfusion in the 6 months prior to starting IV bevacizumab. This compares to 31% (n=5) of patients in the 1 to 3 months after the start of treatment, 29% (n=4) at 4 to 6 months, 14% (n=2) at 7 to 9 months, and 8% (n=1) at 9 to 12 months.
Safety
Four patients had hypertension (HTN). One had pre-existing HTN and had to double the daily dose of lisinopril from 10 mg to 20 mg.
Two HTN patients had to start antihypertensive medications. The fourth patient experienced hypertensive urgency with a temporary decline in renal function. However, the patient was able to resume bevacizumab.
Two patients had infusion-related chills and fever, but premedication with acetaminophen and diphenhydramine prevented these events from recurring.
Three patients died during follow-up. Causes of death were stroke, infective endocarditis (methicillin-sensitive Staphylococcus aureus) with multiple cerebral infarcts, and postoperative respiratory failure (after left atrial appendectomy for paroxysmal atrial fibrillation).
None of these deaths were directly linked to bevacizumab.
“This study provides good-quality evidence for the excellent efficacy and safety of intravenous bevacizumab in the treatment of these patients,” Dr Iyer said. “Intravenous bevacizumab should be considered as a standard, first-line treatment option for HHT patients with severe bleeding and transfusion-dependent anemia.” ![]()
Intravenous (IV) bevacizumab “dramatically” improves severe bleeding associated with hereditary hemorrhagic telangiectasia (HHT), according to researchers.
In a retrospective study, HHT patients with severe bleeding had a substantial reduction in nose bleeds and gastrointestinal (GI) bleeding after treatment with IV bevacizumab.
In addition, patients were able to stop or considerably reduce red blood cell (RBC) transfusions.
The researchers therefore believe that IV bevacizumab should be considered as a first-line therapy for the treatment of refractory bleeding in patients with severe HHT.
The team detailed this research and in Mayo Clinic Proceedings alongside a related editorial.
“Some HHT patients suffer from severe epistaxis and gastrointestinal bleeding, which can result in severe anemia and years of blood transfusions,” said study author Vivek N. Iyer, MD, of the Mayo Clinic in Rochester, Minnesota.
“Both problems also appear to sometimes worsen with age. In some patients, both epistaxis and GI bleeding can become refractory/resistant to existing treatment options, leaving patients severely anemic and dependent on iron infusions or blood transfusions. Quality of life is very poor in these cases.”
With this in mind, Dr Iyer and his colleagues analyzed the records of 34 patients who were treated with IV bevacizumab for severe HHT-related bleeding from June 2013 through January 2017.
Patient characteristics
Patients had a median age of 63 (range, 57 to 72). Sixty-two percent of patients were female.
The primary source of bleeding was epistaxis in 15 patients, GI bleeding in 4 patients, and combined epistaxis and GI bleeding in 15 patients.
Prior epistaxis treatments included potassium-titanyl-phosphate/other laser procedures (62%), sclerotherapy (26%), endovascular angiographic embolization (21%), septodermoplasty (24%), subcutaneous bevacizumab injections (21%), and bevacizumab nasal spray (29%).
Seventy-one percent of patients underwent upper endoscopy (100% of these with telangiectasias), and 53% underwent colonoscopy (56% of these with telangiectasias).
Forty-one percent of patients had IV iron supplementation in the past 6 months.
Twenty-eight patients had received RBC transfusions. Sixteen patients were transfusion-dependent and had received a median of 75 transfusions before starting treatment with bevacizumab. The median duration of transfusion dependence was 6 years.
Treatment
The typical initial dosing cycle of bevacizumab consisted of 8 doses (4 doses each administered 2 weeks apart, followed by 4 doses each administered 1 month apart) over a period of around 22 weeks.
Further maintenance doses after the initial dosing cycle were individualized in each patient.
At last follow-up, 3 patients were still receiving bevacizumab from the initial dosing protocol. For the 31 patients who had completed the first dosing cycle, the median duration of follow-up was 13.6 months.
Eighteen patients required at least 1 top-up dose of IV bevacizumab because of worsening bleeding and/or anemia.
Efficacy
The median follow-up was 17.6 months (range, 3 to 42.5 months).
An Epistaxis Severity Score (ESS) questionnaire was used to assess the severity of nose bleeds both at the beginning of the study and after starting bevacizumab.
One month after starting treatment, there was a significant reduction in ESS scores (P<0.001). This improvement was maintained after patients completed the initial treatment cycle.
The median ESS score was 6.5 at baseline, 3.3 at 1 month, 4.0 at 3 months, 2.3 at the end of the first cycle, 2.0 at 1 to 3 months after the first cycle, 3.2 at 4 to 6 months, and 2.8 at 7 to 12 months.
GI bleeding also improved, with resolution or improvement of anemia in all 19 patients with this condition.
There was a reduction in RBC transfusions as well. The proportion of patients receiving transfusions was 53% in the 6 months before IV bevacizumab, 15% in the 1 to 3 months after starting IV bevacizumab, 14% at 4 to 6 months, 8% at 7 to 9 months, and 9% at 9 to 12 months.
Eighty-eight percent (n=14) of the transfusion-dependent patients had received a transfusion in the 6 months prior to starting IV bevacizumab. This compares to 31% (n=5) of patients in the 1 to 3 months after the start of treatment, 29% (n=4) at 4 to 6 months, 14% (n=2) at 7 to 9 months, and 8% (n=1) at 9 to 12 months.
Safety
Four patients had hypertension (HTN). One had pre-existing HTN and had to double the daily dose of lisinopril from 10 mg to 20 mg.
Two HTN patients had to start antihypertensive medications. The fourth patient experienced hypertensive urgency with a temporary decline in renal function. However, the patient was able to resume bevacizumab.
Two patients had infusion-related chills and fever, but premedication with acetaminophen and diphenhydramine prevented these events from recurring.
Three patients died during follow-up. Causes of death were stroke, infective endocarditis (methicillin-sensitive Staphylococcus aureus) with multiple cerebral infarcts, and postoperative respiratory failure (after left atrial appendectomy for paroxysmal atrial fibrillation).
None of these deaths were directly linked to bevacizumab.
“This study provides good-quality evidence for the excellent efficacy and safety of intravenous bevacizumab in the treatment of these patients,” Dr Iyer said. “Intravenous bevacizumab should be considered as a standard, first-line treatment option for HHT patients with severe bleeding and transfusion-dependent anemia.” ![]()
Trial protocols redacted by industry sponsors
New research has revealed a lack of public information regarding protocols for industry-sponsored, randomized drug trials in Denmark.
First, researchers found it difficult to obtain protocols for commercially sponsored trials, with some sponsors taking legal action in an attempt to keep protocols private.
When the researchers did receive protocols, many had widespread redactions.
The researchers reported these findings in the Journal of the Royal Society of Medicine.
“We wished to compare the information in the protocols with the information provided to the patients in order to evaluate whether the trials were ethical and necessary and whether essential information about the benefits and the harms of the drugs had been hidden from the patients,” said study author Peter Gøtzsche, MD, director of the Nordic Cochrane Centre in Copenhagen, Denmark.
To that end, Dr Gøtzsche and his colleagues used the Danish Freedom of Information Act to request access to 78 protocols for randomized trials that were approved by a research ethics committee from October 2012 to March 2013.
The researchers said several companies refused to provide protocols and involved their lawyers. In fact, Sanofi-Aventis sued the National Committee on Health Research Ethics but lost.
Three years after this project was started, the researchers were able to obtain all the protocols they had requested. Eight protocols were excluded from analysis because they did not meet the research inclusion criteria.
Seventeen of 34 protocols for commercially sponsored trials were unredacted, compared to 34 of 36 non-commercially sponsored trials.
The researchers said the redactions “were most widespread in those sections of the protocol where there is empirical evidence of substantial problems with the trustworthiness of published drug trials.”
This includes data analysis, the handling of missing data, the detection/analysis of adverse events, the definition of patient outcomes, interim analyses and premature study termination, the sponsor’s access to incoming data while the study is ongoing, ownership of the data, and investigators’ publication rights.
“The amount of redactions in the protocols we received was so vast that it made them rather useless for assessing the ethical justification for the studies and to identify discrepancies with subsequent publications,” Dr Gøtzsche said.
“We could not identify any legitimate rationale for the redactions. The current mistrust in industry-sponsored drug trials can only change if the industry offers unconditional access to its trial protocols and other relevant documents and data.” ![]()
New research has revealed a lack of public information regarding protocols for industry-sponsored, randomized drug trials in Denmark.
First, researchers found it difficult to obtain protocols for commercially sponsored trials, with some sponsors taking legal action in an attempt to keep protocols private.
When the researchers did receive protocols, many had widespread redactions.
The researchers reported these findings in the Journal of the Royal Society of Medicine.
“We wished to compare the information in the protocols with the information provided to the patients in order to evaluate whether the trials were ethical and necessary and whether essential information about the benefits and the harms of the drugs had been hidden from the patients,” said study author Peter Gøtzsche, MD, director of the Nordic Cochrane Centre in Copenhagen, Denmark.
To that end, Dr Gøtzsche and his colleagues used the Danish Freedom of Information Act to request access to 78 protocols for randomized trials that were approved by a research ethics committee from October 2012 to March 2013.
The researchers said several companies refused to provide protocols and involved their lawyers. In fact, Sanofi-Aventis sued the National Committee on Health Research Ethics but lost.
Three years after this project was started, the researchers were able to obtain all the protocols they had requested. Eight protocols were excluded from analysis because they did not meet the research inclusion criteria.
Seventeen of 34 protocols for commercially sponsored trials were unredacted, compared to 34 of 36 non-commercially sponsored trials.
The researchers said the redactions “were most widespread in those sections of the protocol where there is empirical evidence of substantial problems with the trustworthiness of published drug trials.”
This includes data analysis, the handling of missing data, the detection/analysis of adverse events, the definition of patient outcomes, interim analyses and premature study termination, the sponsor’s access to incoming data while the study is ongoing, ownership of the data, and investigators’ publication rights.
“The amount of redactions in the protocols we received was so vast that it made them rather useless for assessing the ethical justification for the studies and to identify discrepancies with subsequent publications,” Dr Gøtzsche said.
“We could not identify any legitimate rationale for the redactions. The current mistrust in industry-sponsored drug trials can only change if the industry offers unconditional access to its trial protocols and other relevant documents and data.” ![]()
New research has revealed a lack of public information regarding protocols for industry-sponsored, randomized drug trials in Denmark.
First, researchers found it difficult to obtain protocols for commercially sponsored trials, with some sponsors taking legal action in an attempt to keep protocols private.
When the researchers did receive protocols, many had widespread redactions.
The researchers reported these findings in the Journal of the Royal Society of Medicine.
“We wished to compare the information in the protocols with the information provided to the patients in order to evaluate whether the trials were ethical and necessary and whether essential information about the benefits and the harms of the drugs had been hidden from the patients,” said study author Peter Gøtzsche, MD, director of the Nordic Cochrane Centre in Copenhagen, Denmark.
To that end, Dr Gøtzsche and his colleagues used the Danish Freedom of Information Act to request access to 78 protocols for randomized trials that were approved by a research ethics committee from October 2012 to March 2013.
The researchers said several companies refused to provide protocols and involved their lawyers. In fact, Sanofi-Aventis sued the National Committee on Health Research Ethics but lost.
Three years after this project was started, the researchers were able to obtain all the protocols they had requested. Eight protocols were excluded from analysis because they did not meet the research inclusion criteria.
Seventeen of 34 protocols for commercially sponsored trials were unredacted, compared to 34 of 36 non-commercially sponsored trials.
The researchers said the redactions “were most widespread in those sections of the protocol where there is empirical evidence of substantial problems with the trustworthiness of published drug trials.”
This includes data analysis, the handling of missing data, the detection/analysis of adverse events, the definition of patient outcomes, interim analyses and premature study termination, the sponsor’s access to incoming data while the study is ongoing, ownership of the data, and investigators’ publication rights.
“The amount of redactions in the protocols we received was so vast that it made them rather useless for assessing the ethical justification for the studies and to identify discrepancies with subsequent publications,” Dr Gøtzsche said.
“We could not identify any legitimate rationale for the redactions. The current mistrust in industry-sponsored drug trials can only change if the industry offers unconditional access to its trial protocols and other relevant documents and data.” ![]()
Abstract: Reconciling the Effects of Screening on Prostate Cancer Mortality in the ERSPC and PLCO Trials
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
Tsodikov, A., et al, Ann Intern Med 167(7):449, October 3, 2017
BACKGROUND: In 2012 the U.S. Preventive Services Task Force recommended against routine prostate cancer screening because its lack of effect on long-term mortality. Their recommendations (which are being updated) were based mainly on the ERSPC (European Randomized Study of Screening for Prostate Cancer) and PLCO (Prostate, Lung, Colorectal, and Ovarian Cancer Screening Trial) trials. However, these two trials are substantially different in terms of design, settings, screening intensity, baseline risk and screening effect on mortality (a relative reduction of 21% vs. no reduction, respectively).
METHODS: To evaluate whether the mortality difference persisted after accounting for implementation and practice setting differences, these multinational authors combined data from both trials to conduct a traditional statistical analysis that adjusted for participant age and trial setting, and extended analyses that overcame variability in screening intensity by considering mean lead times, which indicated increased prostate cancer incidence and earlier diagnosis with vs. without screening. Follow-up was restricted to eleven years for both trials.
RESULTS: The traditional analysis demonstrated a marginally different screening effect on mortality between trials (p=0.087), and an overall relative risk reduction of 16% (p=0.010). Extended analyses indicated no difference in screening effect on mortality between trials (p=0.37 to 0.47), and an overall association of longer mean lead times with a lower risk of prostate cancer specific death (p=0.0027 to 0.0032). Screening was estimated to lead to a 25-31% and a 27-32% relative reduction in risk of prostate cancer death in the two trials, respectively, vs. no screening (NNT = ~ 1,111 for 11 years of regular screening).
CONCLUSIONS: This analysis of data from two large prostate cancer-screening trials found that screening significantly reduced prostate cancer mortality risk compared with no screening. 18 references ([email protected] for reprints)
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
Tsodikov, A., et al, Ann Intern Med 167(7):449, October 3, 2017
BACKGROUND: In 2012 the U.S. Preventive Services Task Force recommended against routine prostate cancer screening because its lack of effect on long-term mortality. Their recommendations (which are being updated) were based mainly on the ERSPC (European Randomized Study of Screening for Prostate Cancer) and PLCO (Prostate, Lung, Colorectal, and Ovarian Cancer Screening Trial) trials. However, these two trials are substantially different in terms of design, settings, screening intensity, baseline risk and screening effect on mortality (a relative reduction of 21% vs. no reduction, respectively).
METHODS: To evaluate whether the mortality difference persisted after accounting for implementation and practice setting differences, these multinational authors combined data from both trials to conduct a traditional statistical analysis that adjusted for participant age and trial setting, and extended analyses that overcame variability in screening intensity by considering mean lead times, which indicated increased prostate cancer incidence and earlier diagnosis with vs. without screening. Follow-up was restricted to eleven years for both trials.
RESULTS: The traditional analysis demonstrated a marginally different screening effect on mortality between trials (p=0.087), and an overall relative risk reduction of 16% (p=0.010). Extended analyses indicated no difference in screening effect on mortality between trials (p=0.37 to 0.47), and an overall association of longer mean lead times with a lower risk of prostate cancer specific death (p=0.0027 to 0.0032). Screening was estimated to lead to a 25-31% and a 27-32% relative reduction in risk of prostate cancer death in the two trials, respectively, vs. no screening (NNT = ~ 1,111 for 11 years of regular screening).
CONCLUSIONS: This analysis of data from two large prostate cancer-screening trials found that screening significantly reduced prostate cancer mortality risk compared with no screening. 18 references ([email protected] for reprints)
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
Tsodikov, A., et al, Ann Intern Med 167(7):449, October 3, 2017
BACKGROUND: In 2012 the U.S. Preventive Services Task Force recommended against routine prostate cancer screening because its lack of effect on long-term mortality. Their recommendations (which are being updated) were based mainly on the ERSPC (European Randomized Study of Screening for Prostate Cancer) and PLCO (Prostate, Lung, Colorectal, and Ovarian Cancer Screening Trial) trials. However, these two trials are substantially different in terms of design, settings, screening intensity, baseline risk and screening effect on mortality (a relative reduction of 21% vs. no reduction, respectively).
METHODS: To evaluate whether the mortality difference persisted after accounting for implementation and practice setting differences, these multinational authors combined data from both trials to conduct a traditional statistical analysis that adjusted for participant age and trial setting, and extended analyses that overcame variability in screening intensity by considering mean lead times, which indicated increased prostate cancer incidence and earlier diagnosis with vs. without screening. Follow-up was restricted to eleven years for both trials.
RESULTS: The traditional analysis demonstrated a marginally different screening effect on mortality between trials (p=0.087), and an overall relative risk reduction of 16% (p=0.010). Extended analyses indicated no difference in screening effect on mortality between trials (p=0.37 to 0.47), and an overall association of longer mean lead times with a lower risk of prostate cancer specific death (p=0.0027 to 0.0032). Screening was estimated to lead to a 25-31% and a 27-32% relative reduction in risk of prostate cancer death in the two trials, respectively, vs. no screening (NNT = ~ 1,111 for 11 years of regular screening).
CONCLUSIONS: This analysis of data from two large prostate cancer-screening trials found that screening significantly reduced prostate cancer mortality risk compared with no screening. 18 references ([email protected] for reprints)
Learn more about the Primary Care Medical Abstracts and podcasts, for which you can earn up to 9 CME credits per month.
Copyright © The Center for Medical Education
Reactive aggressive disorder in children with ADHD is looking for a name
NEW YORK – according to Robert L. Findling, MD.
Emphasizing the reactive component to this behavioral problem, he said: “They look okay until someone bumps into them at school. They do not have a mood disorder. They have a disorder of reactivity.”
The hurdle is that there is no accepted terminology to encourage clinicians to identify and initiate treatment in children with this behavior. The term conduct disorder has been used in the past, but Dr. Findling said that care delivered for conduct disorder is not reimbursable. This may be among the reasons that aggressive reactive behavior of ADHD is overlooked – even though treatment is likely to improve long-term outcome.
“I wish I had a magic label for this, but I don’t,” Dr. Findling said. However, he maintained that most clinicians who work with ADHD children are familiar with this type of behavior. Indeed, clinicians “grapple with this day to day. We all see these kids, and they are oftentimes the most impaired kids in our practices,” he said at a pediatric psychopharmacology update held by the American Academy of Child and Adolescent Psychiatry.
This behavior should not be confused with the aggression associated with mood disorders, such as disruptive mood dysregulation disorder (DMDD) or bipolar disease, according to Dr. Findling. Children with DMDD, for example, are chronically irritable or angry. Although bipolar disorder patients with aggressive behavior are not necessarily angry between episodes, they also have persistent mood disturbances.
In contrast, preadolescent children with ADHD who have episodes of aggression, a symptom far more common among males than females, do not otherwise exhibit disturbances in mood. In addition, the episodes of impulsive, reactive aggression are provoked. They require a perceived insult, threat, or similar trigger.
While many of these children continue to have episodes of impulsive aggressive behavior even on treatment effective for other ADHD symptoms, Dr. Findling said, “The good news is that there are treatments for aggression.” In addition to psychosocial support aimed at reducing aggressive behavior, once the diagnosis has been made, these include adjusting ADHD treatments to better target symptoms of episodic aggression. If needed, therapies known to treat aggression, such as atypical antipsychotics, anticonvulsants, or lithium also are options.
Dr. Findling did review one older double-blind study that associated methylphenidate with a reduction in aggression in children with conduct disorder, but said he believes that there is no guarantee for a response from any treatment. Rather, he recommended empirical strategies for symptom management and keeping in mind the benefit-to-risk relationship when considering treatments that impose a high burden of adverse events.
However, the first step to treatment is recognizing the problem.
“In my opinion, what is missing is the nosology for these kids,” Dr. Findling said. An evidence-based label will help increase awareness of the problem and encourage more extensive clinical study, he said.
“These children are not rare and they are really impaired. It is heartbreaking, because when you talk to them when they are still little, they know what people think of them. They know their teachers don’t like them. They know their parents think they’re bad. They know their peers are scared of them, and they cannot make friends,” he said. However, there is a potential for reversing these problems if treatment is initiated early.
“As you watch them get older, you watch them scarring over,” he added.
Dr. Findling reported financial ties with numerous pharmaceutical companies.
SOURCE: Findling RL. Psychopharmacology Update Institute
NEW YORK – according to Robert L. Findling, MD.
Emphasizing the reactive component to this behavioral problem, he said: “They look okay until someone bumps into them at school. They do not have a mood disorder. They have a disorder of reactivity.”
The hurdle is that there is no accepted terminology to encourage clinicians to identify and initiate treatment in children with this behavior. The term conduct disorder has been used in the past, but Dr. Findling said that care delivered for conduct disorder is not reimbursable. This may be among the reasons that aggressive reactive behavior of ADHD is overlooked – even though treatment is likely to improve long-term outcome.
“I wish I had a magic label for this, but I don’t,” Dr. Findling said. However, he maintained that most clinicians who work with ADHD children are familiar with this type of behavior. Indeed, clinicians “grapple with this day to day. We all see these kids, and they are oftentimes the most impaired kids in our practices,” he said at a pediatric psychopharmacology update held by the American Academy of Child and Adolescent Psychiatry.
This behavior should not be confused with the aggression associated with mood disorders, such as disruptive mood dysregulation disorder (DMDD) or bipolar disease, according to Dr. Findling. Children with DMDD, for example, are chronically irritable or angry. Although bipolar disorder patients with aggressive behavior are not necessarily angry between episodes, they also have persistent mood disturbances.
In contrast, preadolescent children with ADHD who have episodes of aggression, a symptom far more common among males than females, do not otherwise exhibit disturbances in mood. In addition, the episodes of impulsive, reactive aggression are provoked. They require a perceived insult, threat, or similar trigger.
While many of these children continue to have episodes of impulsive aggressive behavior even on treatment effective for other ADHD symptoms, Dr. Findling said, “The good news is that there are treatments for aggression.” In addition to psychosocial support aimed at reducing aggressive behavior, once the diagnosis has been made, these include adjusting ADHD treatments to better target symptoms of episodic aggression. If needed, therapies known to treat aggression, such as atypical antipsychotics, anticonvulsants, or lithium also are options.
Dr. Findling did review one older double-blind study that associated methylphenidate with a reduction in aggression in children with conduct disorder, but said he believes that there is no guarantee for a response from any treatment. Rather, he recommended empirical strategies for symptom management and keeping in mind the benefit-to-risk relationship when considering treatments that impose a high burden of adverse events.
However, the first step to treatment is recognizing the problem.
“In my opinion, what is missing is the nosology for these kids,” Dr. Findling said. An evidence-based label will help increase awareness of the problem and encourage more extensive clinical study, he said.
“These children are not rare and they are really impaired. It is heartbreaking, because when you talk to them when they are still little, they know what people think of them. They know their teachers don’t like them. They know their parents think they’re bad. They know their peers are scared of them, and they cannot make friends,” he said. However, there is a potential for reversing these problems if treatment is initiated early.
“As you watch them get older, you watch them scarring over,” he added.
Dr. Findling reported financial ties with numerous pharmaceutical companies.
SOURCE: Findling RL. Psychopharmacology Update Institute
NEW YORK – according to Robert L. Findling, MD.
Emphasizing the reactive component to this behavioral problem, he said: “They look okay until someone bumps into them at school. They do not have a mood disorder. They have a disorder of reactivity.”
The hurdle is that there is no accepted terminology to encourage clinicians to identify and initiate treatment in children with this behavior. The term conduct disorder has been used in the past, but Dr. Findling said that care delivered for conduct disorder is not reimbursable. This may be among the reasons that aggressive reactive behavior of ADHD is overlooked – even though treatment is likely to improve long-term outcome.
“I wish I had a magic label for this, but I don’t,” Dr. Findling said. However, he maintained that most clinicians who work with ADHD children are familiar with this type of behavior. Indeed, clinicians “grapple with this day to day. We all see these kids, and they are oftentimes the most impaired kids in our practices,” he said at a pediatric psychopharmacology update held by the American Academy of Child and Adolescent Psychiatry.
This behavior should not be confused with the aggression associated with mood disorders, such as disruptive mood dysregulation disorder (DMDD) or bipolar disease, according to Dr. Findling. Children with DMDD, for example, are chronically irritable or angry. Although bipolar disorder patients with aggressive behavior are not necessarily angry between episodes, they also have persistent mood disturbances.
In contrast, preadolescent children with ADHD who have episodes of aggression, a symptom far more common among males than females, do not otherwise exhibit disturbances in mood. In addition, the episodes of impulsive, reactive aggression are provoked. They require a perceived insult, threat, or similar trigger.
While many of these children continue to have episodes of impulsive aggressive behavior even on treatment effective for other ADHD symptoms, Dr. Findling said, “The good news is that there are treatments for aggression.” In addition to psychosocial support aimed at reducing aggressive behavior, once the diagnosis has been made, these include adjusting ADHD treatments to better target symptoms of episodic aggression. If needed, therapies known to treat aggression, such as atypical antipsychotics, anticonvulsants, or lithium also are options.
Dr. Findling did review one older double-blind study that associated methylphenidate with a reduction in aggression in children with conduct disorder, but said he believes that there is no guarantee for a response from any treatment. Rather, he recommended empirical strategies for symptom management and keeping in mind the benefit-to-risk relationship when considering treatments that impose a high burden of adverse events.
However, the first step to treatment is recognizing the problem.
“In my opinion, what is missing is the nosology for these kids,” Dr. Findling said. An evidence-based label will help increase awareness of the problem and encourage more extensive clinical study, he said.
“These children are not rare and they are really impaired. It is heartbreaking, because when you talk to them when they are still little, they know what people think of them. They know their teachers don’t like them. They know their parents think they’re bad. They know their peers are scared of them, and they cannot make friends,” he said. However, there is a potential for reversing these problems if treatment is initiated early.
“As you watch them get older, you watch them scarring over,” he added.
Dr. Findling reported financial ties with numerous pharmaceutical companies.
SOURCE: Findling RL. Psychopharmacology Update Institute
REPORTING FROM THE PSYCHOPHARMACOLOGY UPDATE INSTITUTE
Notable acute care surgery papers from 2017
LAKE BUENA VISTA, FLA. – Every year brings new studies, updates, and trials, and it can be a challenge to keep up.
Christian Jones, MD, FACS, a general surgeon in the division of acute care surgery at Johns Hopkins University, Baltimore, ranked some of the more notable trauma studies published in the past year and presented his perspective on them at the annual scientific assembly of the Eastern Association for the Surgery of Trauma.
Day 2 is the “sweet spot” for cholecystectomy
When it comes to cholecystectomy, acute cholecystitis (AC) patients appear to fare the best when operations are conducted on day 2 after admission, according to a study of patients registered in the Swedish Registry of Gallstone Surgery and Endoscopic Retrograde Cholangiopancreatography (GallRiks).
The retrospective study of 15,760 AC patients found that the rate of 30-day mortality of AC patients was significantly higher for patients who underwent a cholecystectomy on day of (odds ratio = .42) 3 days after (OR = .34), and 4 days after admission (OR = 1.0), compared with those who were operated on between 1 day after (OR = .23), and 2 days after (OR = .29) admission.
Lead author My Blohm, MD, of the department of clinical sciences, intervention, and technology at the Karolinska Institutet, Stockholm, and fellow investigators hypothesized that waiting allows patients to be medically optimized for surgery (J Gastrointest Surg. 2017;21[1]: 33-40).
With 90-day mortality rates showing nearly identical results for day 1 and day 2, holding off on surgery may be the best move for the patient, even if it is not the ideal situation for a provider.
“Sure, as we all know by now, delayed cholecystectomy is seldom necessary, later surgery is more difficult, and more likely to be associated with complications at least with an equal conversion to an open procedure, but even more surprising is the higher mortality on the admissions day,” said Dr. Jones.
Antibiotics for abscess drainage patients
For patients requiring abscess drainage, antibiotics may be the best bet to keep infection at bay, according to a study published in the New England Journal of Medicine in June 2017.
The prospective, randomized, placebo-controlled, double-blind, study of 786 simple skin abscess drainage patients found clindamycin and Bactrim (sulfamethoxazole and trimethoprim) outperformed a placebo in an evaluation of symptoms of true ongoing infection in patients even 30 days after the procedure (N Engl J Med. 2017 Jun 29;376[26]:2545-55).
Patients studied had Staphylococcus aureus (527) or methicillin-resistant S. aureus (388).
After 10 days of therapy, cure rate of infection for the clindamycin and Bactrim groups were 83% and 82% respectively, compared with 70% in the placebo group, according to Robert S. Daum, MD, principal investigator at the MRSA Research Center, University of Chicago. After 30 days, cure rate for both antibiotic groups remained superior to that of the placebo group.
While these treatments were successful, concern of drug resistance is notable and should be taken into consideration when deciding on treatment options.
“This does get to our typical concern with increased antibiotic usage, and that’s the concern of the health of the community versus the health of the individual patient,” said Dr. Jones. “Is the increased rate of [antibiotic] resistance important enough to have a lower cure rate of simple abscess drainage? We don’t know the answer to that.”
Loop ileostomies look good for C. diff patients
This minimally invasive procedure has been the subject of some well-received studies with findings that indicate it is a promising choice for patients with a Clostridium difficile–associated disease (CDAD) over total colectomy, Dr. Jones said.
In a study published in the Journal of Trauma and Acute Care Surgery, a study group of patients with CDAD who had loop ileostomy had no statistical difference in almost any recorded characteristic compared with those who underwent a total colectomy, except mortality rate. The retrospective, multicenter study of 98 CDAD patients found the mortality rate of the loop ileostomy group to be 17.2%, compared with 39.7% in the total colectomy group (J Trauma Acute Care Surg. 2017 Jul;83[1]:36-40).
“The outcomes all favored loop ileostomy in a statistically significant fashion,” said Dr. Jones. “Unsurprisingly, estimated blood loss and need for transfusions were all significantly less in the loop ileostomy patients, and the adjusted overall mortality, even if requiring a reoperation, still favored doing the loop ileostomy first.”
The one difference between LI and colectomy patients was a longer time from initial diagnosis to operation among LI patients, with about 12 hours from diagnosis for the colectomy versus 24 hours for LI patients, according to lead author Paula Ferrada, MD, FACS, director of the surgical and trauma intensive care unit at Virginia Commonwealth University, Richmond, and her fellow investigators,
Contrary to previous findings, the study found that LI can be performed on sick patients as well, according to the researchers, and failure of the procedure is not associated with increased mortality.
While these findings are encouraging, “there are things that the individual patient may reveal to you on your examination that tell you they are not a candidate and that you should go to total colectomy,” said Dr. Jones. “Keep in mind that perhaps we can be a bit more aggressive in this less invasive procedure.”
The skin vac actually works
A study published in Annals of Surgery found prophylactic negative-pressure dressings are associated with a decreased rate of surgical site infections in laparotomy wounds.
“The biggest surprise to me out of all of these studies is that a new piece of technology actually seems to work,” said Dr. Jones.
The randomized study included 50 laparotomy patients with a stapled wound, half of whom received a skin vac over their incision while the other half had a standard OpSite occlusive dressing (Ann Surg. 2017 Jun;265[6]:1082-6).
Patients in both arms had the same type of wound and had their dressings on for 4 days before being switched.
Rate of surgical site infections for the skin vac group was 8.3% over 30 days from operation, compared with 32% in the OpSite group. Average length of stay for patients with the pressure dressing was 6.1 days, while patients with an OpSite dressing had a length of 14.7 days, more than double, according to lead author Donal Peter O’Leary, MD, surgeon at Cork University Hospital, Ireland.
The difference in length of stay does become insignificant if six OpSite patients who stayed longer than 20 days are discounted, only two of whom were delayed because of wound complications as opposed to placement issues or unassociated infections.
“But a surgical site infection difference of 50% or more using a skin vac instead of a standard dressing, whether you’re talking about clean, clean-contaminated, or contaminated cases with a skin closure, seems to be worthy of notice,” explained Dr. Jones.
LAKE BUENA VISTA, FLA. – Every year brings new studies, updates, and trials, and it can be a challenge to keep up.
Christian Jones, MD, FACS, a general surgeon in the division of acute care surgery at Johns Hopkins University, Baltimore, ranked some of the more notable trauma studies published in the past year and presented his perspective on them at the annual scientific assembly of the Eastern Association for the Surgery of Trauma.
Day 2 is the “sweet spot” for cholecystectomy
When it comes to cholecystectomy, acute cholecystitis (AC) patients appear to fare the best when operations are conducted on day 2 after admission, according to a study of patients registered in the Swedish Registry of Gallstone Surgery and Endoscopic Retrograde Cholangiopancreatography (GallRiks).
The retrospective study of 15,760 AC patients found that the rate of 30-day mortality of AC patients was significantly higher for patients who underwent a cholecystectomy on day of (odds ratio = .42) 3 days after (OR = .34), and 4 days after admission (OR = 1.0), compared with those who were operated on between 1 day after (OR = .23), and 2 days after (OR = .29) admission.
Lead author My Blohm, MD, of the department of clinical sciences, intervention, and technology at the Karolinska Institutet, Stockholm, and fellow investigators hypothesized that waiting allows patients to be medically optimized for surgery (J Gastrointest Surg. 2017;21[1]: 33-40).
With 90-day mortality rates showing nearly identical results for day 1 and day 2, holding off on surgery may be the best move for the patient, even if it is not the ideal situation for a provider.
“Sure, as we all know by now, delayed cholecystectomy is seldom necessary, later surgery is more difficult, and more likely to be associated with complications at least with an equal conversion to an open procedure, but even more surprising is the higher mortality on the admissions day,” said Dr. Jones.
Antibiotics for abscess drainage patients
For patients requiring abscess drainage, antibiotics may be the best bet to keep infection at bay, according to a study published in the New England Journal of Medicine in June 2017.
The prospective, randomized, placebo-controlled, double-blind, study of 786 simple skin abscess drainage patients found clindamycin and Bactrim (sulfamethoxazole and trimethoprim) outperformed a placebo in an evaluation of symptoms of true ongoing infection in patients even 30 days after the procedure (N Engl J Med. 2017 Jun 29;376[26]:2545-55).
Patients studied had Staphylococcus aureus (527) or methicillin-resistant S. aureus (388).
After 10 days of therapy, cure rate of infection for the clindamycin and Bactrim groups were 83% and 82% respectively, compared with 70% in the placebo group, according to Robert S. Daum, MD, principal investigator at the MRSA Research Center, University of Chicago. After 30 days, cure rate for both antibiotic groups remained superior to that of the placebo group.
While these treatments were successful, concern of drug resistance is notable and should be taken into consideration when deciding on treatment options.
“This does get to our typical concern with increased antibiotic usage, and that’s the concern of the health of the community versus the health of the individual patient,” said Dr. Jones. “Is the increased rate of [antibiotic] resistance important enough to have a lower cure rate of simple abscess drainage? We don’t know the answer to that.”
Loop ileostomies look good for C. diff patients
This minimally invasive procedure has been the subject of some well-received studies with findings that indicate it is a promising choice for patients with a Clostridium difficile–associated disease (CDAD) over total colectomy, Dr. Jones said.
In a study published in the Journal of Trauma and Acute Care Surgery, a study group of patients with CDAD who had loop ileostomy had no statistical difference in almost any recorded characteristic compared with those who underwent a total colectomy, except mortality rate. The retrospective, multicenter study of 98 CDAD patients found the mortality rate of the loop ileostomy group to be 17.2%, compared with 39.7% in the total colectomy group (J Trauma Acute Care Surg. 2017 Jul;83[1]:36-40).
“The outcomes all favored loop ileostomy in a statistically significant fashion,” said Dr. Jones. “Unsurprisingly, estimated blood loss and need for transfusions were all significantly less in the loop ileostomy patients, and the adjusted overall mortality, even if requiring a reoperation, still favored doing the loop ileostomy first.”
The one difference between LI and colectomy patients was a longer time from initial diagnosis to operation among LI patients, with about 12 hours from diagnosis for the colectomy versus 24 hours for LI patients, according to lead author Paula Ferrada, MD, FACS, director of the surgical and trauma intensive care unit at Virginia Commonwealth University, Richmond, and her fellow investigators,
Contrary to previous findings, the study found that LI can be performed on sick patients as well, according to the researchers, and failure of the procedure is not associated with increased mortality.
While these findings are encouraging, “there are things that the individual patient may reveal to you on your examination that tell you they are not a candidate and that you should go to total colectomy,” said Dr. Jones. “Keep in mind that perhaps we can be a bit more aggressive in this less invasive procedure.”
The skin vac actually works
A study published in Annals of Surgery found prophylactic negative-pressure dressings are associated with a decreased rate of surgical site infections in laparotomy wounds.
“The biggest surprise to me out of all of these studies is that a new piece of technology actually seems to work,” said Dr. Jones.
The randomized study included 50 laparotomy patients with a stapled wound, half of whom received a skin vac over their incision while the other half had a standard OpSite occlusive dressing (Ann Surg. 2017 Jun;265[6]:1082-6).
Patients in both arms had the same type of wound and had their dressings on for 4 days before being switched.
Rate of surgical site infections for the skin vac group was 8.3% over 30 days from operation, compared with 32% in the OpSite group. Average length of stay for patients with the pressure dressing was 6.1 days, while patients with an OpSite dressing had a length of 14.7 days, more than double, according to lead author Donal Peter O’Leary, MD, surgeon at Cork University Hospital, Ireland.
The difference in length of stay does become insignificant if six OpSite patients who stayed longer than 20 days are discounted, only two of whom were delayed because of wound complications as opposed to placement issues or unassociated infections.
“But a surgical site infection difference of 50% or more using a skin vac instead of a standard dressing, whether you’re talking about clean, clean-contaminated, or contaminated cases with a skin closure, seems to be worthy of notice,” explained Dr. Jones.
LAKE BUENA VISTA, FLA. – Every year brings new studies, updates, and trials, and it can be a challenge to keep up.
Christian Jones, MD, FACS, a general surgeon in the division of acute care surgery at Johns Hopkins University, Baltimore, ranked some of the more notable trauma studies published in the past year and presented his perspective on them at the annual scientific assembly of the Eastern Association for the Surgery of Trauma.
Day 2 is the “sweet spot” for cholecystectomy
When it comes to cholecystectomy, acute cholecystitis (AC) patients appear to fare the best when operations are conducted on day 2 after admission, according to a study of patients registered in the Swedish Registry of Gallstone Surgery and Endoscopic Retrograde Cholangiopancreatography (GallRiks).
The retrospective study of 15,760 AC patients found that the rate of 30-day mortality of AC patients was significantly higher for patients who underwent a cholecystectomy on day of (odds ratio = .42) 3 days after (OR = .34), and 4 days after admission (OR = 1.0), compared with those who were operated on between 1 day after (OR = .23), and 2 days after (OR = .29) admission.
Lead author My Blohm, MD, of the department of clinical sciences, intervention, and technology at the Karolinska Institutet, Stockholm, and fellow investigators hypothesized that waiting allows patients to be medically optimized for surgery (J Gastrointest Surg. 2017;21[1]: 33-40).
With 90-day mortality rates showing nearly identical results for day 1 and day 2, holding off on surgery may be the best move for the patient, even if it is not the ideal situation for a provider.
“Sure, as we all know by now, delayed cholecystectomy is seldom necessary, later surgery is more difficult, and more likely to be associated with complications at least with an equal conversion to an open procedure, but even more surprising is the higher mortality on the admissions day,” said Dr. Jones.
Antibiotics for abscess drainage patients
For patients requiring abscess drainage, antibiotics may be the best bet to keep infection at bay, according to a study published in the New England Journal of Medicine in June 2017.
The prospective, randomized, placebo-controlled, double-blind, study of 786 simple skin abscess drainage patients found clindamycin and Bactrim (sulfamethoxazole and trimethoprim) outperformed a placebo in an evaluation of symptoms of true ongoing infection in patients even 30 days after the procedure (N Engl J Med. 2017 Jun 29;376[26]:2545-55).
Patients studied had Staphylococcus aureus (527) or methicillin-resistant S. aureus (388).
After 10 days of therapy, cure rate of infection for the clindamycin and Bactrim groups were 83% and 82% respectively, compared with 70% in the placebo group, according to Robert S. Daum, MD, principal investigator at the MRSA Research Center, University of Chicago. After 30 days, cure rate for both antibiotic groups remained superior to that of the placebo group.
While these treatments were successful, concern of drug resistance is notable and should be taken into consideration when deciding on treatment options.
“This does get to our typical concern with increased antibiotic usage, and that’s the concern of the health of the community versus the health of the individual patient,” said Dr. Jones. “Is the increased rate of [antibiotic] resistance important enough to have a lower cure rate of simple abscess drainage? We don’t know the answer to that.”
Loop ileostomies look good for C. diff patients
This minimally invasive procedure has been the subject of some well-received studies with findings that indicate it is a promising choice for patients with a Clostridium difficile–associated disease (CDAD) over total colectomy, Dr. Jones said.
In a study published in the Journal of Trauma and Acute Care Surgery, a study group of patients with CDAD who had loop ileostomy had no statistical difference in almost any recorded characteristic compared with those who underwent a total colectomy, except mortality rate. The retrospective, multicenter study of 98 CDAD patients found the mortality rate of the loop ileostomy group to be 17.2%, compared with 39.7% in the total colectomy group (J Trauma Acute Care Surg. 2017 Jul;83[1]:36-40).
“The outcomes all favored loop ileostomy in a statistically significant fashion,” said Dr. Jones. “Unsurprisingly, estimated blood loss and need for transfusions were all significantly less in the loop ileostomy patients, and the adjusted overall mortality, even if requiring a reoperation, still favored doing the loop ileostomy first.”
The one difference between LI and colectomy patients was a longer time from initial diagnosis to operation among LI patients, with about 12 hours from diagnosis for the colectomy versus 24 hours for LI patients, according to lead author Paula Ferrada, MD, FACS, director of the surgical and trauma intensive care unit at Virginia Commonwealth University, Richmond, and her fellow investigators,
Contrary to previous findings, the study found that LI can be performed on sick patients as well, according to the researchers, and failure of the procedure is not associated with increased mortality.
While these findings are encouraging, “there are things that the individual patient may reveal to you on your examination that tell you they are not a candidate and that you should go to total colectomy,” said Dr. Jones. “Keep in mind that perhaps we can be a bit more aggressive in this less invasive procedure.”
The skin vac actually works
A study published in Annals of Surgery found prophylactic negative-pressure dressings are associated with a decreased rate of surgical site infections in laparotomy wounds.
“The biggest surprise to me out of all of these studies is that a new piece of technology actually seems to work,” said Dr. Jones.
The randomized study included 50 laparotomy patients with a stapled wound, half of whom received a skin vac over their incision while the other half had a standard OpSite occlusive dressing (Ann Surg. 2017 Jun;265[6]:1082-6).
Patients in both arms had the same type of wound and had their dressings on for 4 days before being switched.
Rate of surgical site infections for the skin vac group was 8.3% over 30 days from operation, compared with 32% in the OpSite group. Average length of stay for patients with the pressure dressing was 6.1 days, while patients with an OpSite dressing had a length of 14.7 days, more than double, according to lead author Donal Peter O’Leary, MD, surgeon at Cork University Hospital, Ireland.
The difference in length of stay does become insignificant if six OpSite patients who stayed longer than 20 days are discounted, only two of whom were delayed because of wound complications as opposed to placement issues or unassociated infections.
“But a surgical site infection difference of 50% or more using a skin vac instead of a standard dressing, whether you’re talking about clean, clean-contaminated, or contaminated cases with a skin closure, seems to be worthy of notice,” explained Dr. Jones.
REPORTING FROM EAST 2018
Background color a dermoscopic clue to cutaneous B-cell lymphoma
A salmon-colored background and prominent serpentine blood vessels are two characteristic features of primary cutaneous B-cell lymphoma (PCBCL) that can be identified dermoscopically and may aid diagnosis, researchers say.
In the January issue of the Journal of the European Academy of Dermatology and Venereology, researchers reported the results of a retrospective observational study using the dermoscopic images of 58 biopsy-confirmed primary cutaneous B-cell lymphoma lesions in 51 patients.
While all the lesions were nonpigmented, 46 (79.3%) of them showed salmon- or yellow- to orange- colored background areas. More than three-quarters of the lesions also featured prominent blood vessels (77.6%), the majority of which were serpentine in nature.
, while only 8.6% of the lesions showed neither feature.
Of the 58 lesions, the authors selected 17 to be evaluated by two dermoscopy experts who were blinded to the diagnosis. In 70.6% of these cases they included cutaneous B-cell lymphoma in the differential diagnosis, while other diagnoses included spider bite (58.8%), basal cell carcinoma (52.9%), amelanotic melanoma (47.1%), and scar/keloid (47.1%). Overall, the two experts did not agree on almost 30% of the suggested differential diagnoses.
“The presentation of cutaneous lymphomas in general and of PCBCLs in particular can be nonspecific, and a biopsy is essential for a definitive diagnosis,” wrote Shamir Geller, MD, of the dermatology service at Memorial Sloan Kettering Cancer Center, New York, and his coauthors.
The 58 PCBCLs analyzed were among 172 biopsy-proven PCBCL lesions in the study, which were newly diagnosed and whose pathology reports included the clinical differential diagnosis in the pathology requisition slip, in patients referred to the cancer center between 1992 and 2016. In only 16.3% of these cases, the clinician suspected cutaneous lymphoma. Skin malignancies were suspected in 54.7% of cases, with the leading diagnosis being basal cell carcinoma in 17.4% of cases. Basal cell carcinoma was considered in nearly one-third of lesions, particularly those on the head and neck.
Nonneoplastic conditions suspected by clinicians included cyst in 21.5% of cases, granulomatous processes in 15.7%, and infectious disease in 4.7%.
The authors commented that a low index of suspicion for skin lymphoma was seen regardless of the subtype or site.
“While dermoscopy offers a bridge between the naked eye examination and the histopathological appearance, cutaneous lymphoma is diagnosed on a cellular level using histopathology, immunohistochemistry and molecular studies,” they wrote. “Therefore, dermoscopy may serve as an ancillary tool in PCBCL; however, it cannot be diagnostic.”
The study was supported in part by the National Institutes of Health/National Cancer Institute Cancer Center. Dr. Geller is a recipient of a grant from the American Physicians and Friends For Medicine in Israel. No conflicts of interest were declared.
SOURCE: Geller S et al. J Eur Acad Dermatol Venereol. 2018 Jan;32(1):53-6.
A salmon-colored background and prominent serpentine blood vessels are two characteristic features of primary cutaneous B-cell lymphoma (PCBCL) that can be identified dermoscopically and may aid diagnosis, researchers say.
In the January issue of the Journal of the European Academy of Dermatology and Venereology, researchers reported the results of a retrospective observational study using the dermoscopic images of 58 biopsy-confirmed primary cutaneous B-cell lymphoma lesions in 51 patients.
While all the lesions were nonpigmented, 46 (79.3%) of them showed salmon- or yellow- to orange- colored background areas. More than three-quarters of the lesions also featured prominent blood vessels (77.6%), the majority of which were serpentine in nature.
, while only 8.6% of the lesions showed neither feature.
Of the 58 lesions, the authors selected 17 to be evaluated by two dermoscopy experts who were blinded to the diagnosis. In 70.6% of these cases they included cutaneous B-cell lymphoma in the differential diagnosis, while other diagnoses included spider bite (58.8%), basal cell carcinoma (52.9%), amelanotic melanoma (47.1%), and scar/keloid (47.1%). Overall, the two experts did not agree on almost 30% of the suggested differential diagnoses.
“The presentation of cutaneous lymphomas in general and of PCBCLs in particular can be nonspecific, and a biopsy is essential for a definitive diagnosis,” wrote Shamir Geller, MD, of the dermatology service at Memorial Sloan Kettering Cancer Center, New York, and his coauthors.
The 58 PCBCLs analyzed were among 172 biopsy-proven PCBCL lesions in the study, which were newly diagnosed and whose pathology reports included the clinical differential diagnosis in the pathology requisition slip, in patients referred to the cancer center between 1992 and 2016. In only 16.3% of these cases, the clinician suspected cutaneous lymphoma. Skin malignancies were suspected in 54.7% of cases, with the leading diagnosis being basal cell carcinoma in 17.4% of cases. Basal cell carcinoma was considered in nearly one-third of lesions, particularly those on the head and neck.
Nonneoplastic conditions suspected by clinicians included cyst in 21.5% of cases, granulomatous processes in 15.7%, and infectious disease in 4.7%.
The authors commented that a low index of suspicion for skin lymphoma was seen regardless of the subtype or site.
“While dermoscopy offers a bridge between the naked eye examination and the histopathological appearance, cutaneous lymphoma is diagnosed on a cellular level using histopathology, immunohistochemistry and molecular studies,” they wrote. “Therefore, dermoscopy may serve as an ancillary tool in PCBCL; however, it cannot be diagnostic.”
The study was supported in part by the National Institutes of Health/National Cancer Institute Cancer Center. Dr. Geller is a recipient of a grant from the American Physicians and Friends For Medicine in Israel. No conflicts of interest were declared.
SOURCE: Geller S et al. J Eur Acad Dermatol Venereol. 2018 Jan;32(1):53-6.
A salmon-colored background and prominent serpentine blood vessels are two characteristic features of primary cutaneous B-cell lymphoma (PCBCL) that can be identified dermoscopically and may aid diagnosis, researchers say.
In the January issue of the Journal of the European Academy of Dermatology and Venereology, researchers reported the results of a retrospective observational study using the dermoscopic images of 58 biopsy-confirmed primary cutaneous B-cell lymphoma lesions in 51 patients.
While all the lesions were nonpigmented, 46 (79.3%) of them showed salmon- or yellow- to orange- colored background areas. More than three-quarters of the lesions also featured prominent blood vessels (77.6%), the majority of which were serpentine in nature.
, while only 8.6% of the lesions showed neither feature.
Of the 58 lesions, the authors selected 17 to be evaluated by two dermoscopy experts who were blinded to the diagnosis. In 70.6% of these cases they included cutaneous B-cell lymphoma in the differential diagnosis, while other diagnoses included spider bite (58.8%), basal cell carcinoma (52.9%), amelanotic melanoma (47.1%), and scar/keloid (47.1%). Overall, the two experts did not agree on almost 30% of the suggested differential diagnoses.
“The presentation of cutaneous lymphomas in general and of PCBCLs in particular can be nonspecific, and a biopsy is essential for a definitive diagnosis,” wrote Shamir Geller, MD, of the dermatology service at Memorial Sloan Kettering Cancer Center, New York, and his coauthors.
The 58 PCBCLs analyzed were among 172 biopsy-proven PCBCL lesions in the study, which were newly diagnosed and whose pathology reports included the clinical differential diagnosis in the pathology requisition slip, in patients referred to the cancer center between 1992 and 2016. In only 16.3% of these cases, the clinician suspected cutaneous lymphoma. Skin malignancies were suspected in 54.7% of cases, with the leading diagnosis being basal cell carcinoma in 17.4% of cases. Basal cell carcinoma was considered in nearly one-third of lesions, particularly those on the head and neck.
Nonneoplastic conditions suspected by clinicians included cyst in 21.5% of cases, granulomatous processes in 15.7%, and infectious disease in 4.7%.
The authors commented that a low index of suspicion for skin lymphoma was seen regardless of the subtype or site.
“While dermoscopy offers a bridge between the naked eye examination and the histopathological appearance, cutaneous lymphoma is diagnosed on a cellular level using histopathology, immunohistochemistry and molecular studies,” they wrote. “Therefore, dermoscopy may serve as an ancillary tool in PCBCL; however, it cannot be diagnostic.”
The study was supported in part by the National Institutes of Health/National Cancer Institute Cancer Center. Dr. Geller is a recipient of a grant from the American Physicians and Friends For Medicine in Israel. No conflicts of interest were declared.
SOURCE: Geller S et al. J Eur Acad Dermatol Venereol. 2018 Jan;32(1):53-6.
FROM THE JOURNAL OF THE EUROPEAN ACADEMY OF DERMATOLOGY AND VENEREOLOGY
Key clinical point: A salmon-colored background and prominent serpentine blood vessels are two characteristic dermoscopic features of primary cutaneous B-cell lymphoma (PCBCL).
Major finding: Nearly 80% of PCBCLs had a salmon-colored background on dermoscopy.
Data source: A retrospective observational study that analyzed 172 biopsy-proven PCBCLs, including 58 PCBCL dermoscopic images.
Disclosures: The study was supported by the NIH/NCI Cancer Center. The lead author received a grant from the American Physicians and Friends for Medicine in Israel. No conflicts of interest were declared.
Source: Geller S et al. J Eur Acad Dermatol Venereol. 2018 Jan;32(1):53-6.
Controversy surrounds calf vein thrombosis treatment
CHICAGO – The use of therapeutic-dose anticoagulation in hospitalized patients with calf vein thrombosis significantly reduces the risk of venous thromboembolic complications, compared with lower-dose prophylactic anticoagulation or surveillance alone, Heron E. Rodriguez, MD, said at a symposium on vascular surgery sponsored by Northwestern University.
Moreover, placement of an inferior vena cava filter in patients with calf vein thrombosis when anticoagulation is contraindicated accomplishes nothing beneficial and had a 10% complication rate in a large retrospective single-center study, added Dr. Rodriguez of Northwestern University, Chicago.
Deep vein thrombosis (DVT) remains a significant source of morbidity and mortality despite worldwide awareness of the problem.
“Specifically, calf vein thrombosis [CVT] is very common, and we know that in some series up to 30% of patients end up propagating proximally if they’re not treated, and a good number of them develop chronic venous insufficiency, with long-term consequences,” he noted.
“Unfortunately there is no consensus regarding treatment. The guidelines are very vague. For example, there is no mention [in current American College of Chest Physicians guidelines] of how to manage muscular vein thrombosis and much ambiguity on how to treat calf vein thrombosis,” he continued.
Dr. Rodriguez cited as an indication of the lack of consensus on management of CVT a single-institution survey by other investigators of the practices of physicians in various specialties. Forty-nine percent of respondents indicated they anticoagulate patients with CVT; 51% don’t. Of those who did, 62% prescribed low-molecular-weight heparin and 11% intravenous heparin. Fifty-eight percent of physicians who anticoagulated did so for 3 months, 30% for 6. And 46% of physicians used an inferior vena cava (IVC) filter when anticoagulation was contraindicated (Vascular. 2014 Apr;22[2]:93-7).
That rate of IVC placement “seemed really high” given the paucity of supporting evidence for safety and efficacy of filter placement in the setting of CVT, so Dr. Rodriguez and coinvestigators decided to conduct a retrospective review of practices at Northwestern Memorial Hospital. He explained the study hypothesis: “Our thinking was that these kinds of thrombi are associated with low risk of propagation and pulmonary embolism, and they can and should be managed conservatively.”
Of 647 patients with isolated thrombosis of the anterior and posterior tibial, soleal, peroneal, or gastrocnemius veins, 44% received an IVC filter, and the rest got medical treatment alone. Of the 362 patients managed medically, 49% received therapeutic anticoagulation, 12% got low-dose prophylactic anticoagulation, and 39% underwent surveillance without anticoagulation.
The primary outcome was the incidence of venous thromboembolic complications – that is, propagation of DVT or pulmonary embolism. The incidence was 35% in the surveillance-only group, 30% with prophylactic anticoagulation, and 10% in patients who got therapeutic anticoagulation.
Of note, the rate of the most feared complication, pulmonary embolism, was low and similar in the filter recipients and medically managed group: 2.5% in the IVC group, 3.3% with medical management.
“Distal vein thromboses have low rates of pulmonary embolism, regardless of whether or not they are so-called protected with a filter. On the other hand, a filter was associated with a 10% rate of complications. I have to make clear that these were radiographic abnormalities – tilting, migration, caval perforation – that didn’t have clinical consequences, but we were very aggressive in removing the IVC filters, and I’m guessing if they’d been left inside they would create problems in the long term,” Dr. Rodriguez said.
An important point about this study is that these were all sick patients, and most were hospitalized. The filter recipients and medical groups differed in key ways. For example, 49% of the filter patients had a malignancy, and 56% had a baseline history of venous thromboembolic events, compared with 26% and 16%, respectively, of the medical group. For that reason, the investigators performed propensity score matching and came up with 157 closely matched patient pairs. The outcomes remained unchanged.
Of course, this was a retrospective study, with its inherent limitations, but Dr. Rodriguez characterized the published randomized trials on management of CVT as “small and limited.” The most frequently quoted study is the double-blind multicenter CACTUS trial, which randomized 252 outpatients with symptomatic CVT to 6 weeks of low-molecular-weight heparin or placebo and found no difference in the rates of proximal extension of venous thromboembolic events (Lancet Haematol. 2016 Dec;3[12]:e556-62). But these were all low-risk patients. Prior DVT or malignancy were exclusion criteria, so this was a very different population than treated at Northwestern.
Based upon the results of the retrospective study at Northwestern, which have been published (J Vasc Surg Venous Lymphat Disord. 2017 Jan;5[1]:25-32), the vascular surgery service has developed a management algorithm for DVT management based upon the lesion site. If a patient is unable to undergo anticoagulation, venous duplex ultrasound is repeated at 1 week. If the imaging shows propagation into the popliteal vein and anticoagulation remains contraindicated, only then is placement of an IVC filter warranted.
Dr. Rodriguez reported serving as a paid speaker for Abbott, Endologix, and W.L. Gore.
CHICAGO – The use of therapeutic-dose anticoagulation in hospitalized patients with calf vein thrombosis significantly reduces the risk of venous thromboembolic complications, compared with lower-dose prophylactic anticoagulation or surveillance alone, Heron E. Rodriguez, MD, said at a symposium on vascular surgery sponsored by Northwestern University.
Moreover, placement of an inferior vena cava filter in patients with calf vein thrombosis when anticoagulation is contraindicated accomplishes nothing beneficial and had a 10% complication rate in a large retrospective single-center study, added Dr. Rodriguez of Northwestern University, Chicago.
Deep vein thrombosis (DVT) remains a significant source of morbidity and mortality despite worldwide awareness of the problem.
“Specifically, calf vein thrombosis [CVT] is very common, and we know that in some series up to 30% of patients end up propagating proximally if they’re not treated, and a good number of them develop chronic venous insufficiency, with long-term consequences,” he noted.
“Unfortunately there is no consensus regarding treatment. The guidelines are very vague. For example, there is no mention [in current American College of Chest Physicians guidelines] of how to manage muscular vein thrombosis and much ambiguity on how to treat calf vein thrombosis,” he continued.
Dr. Rodriguez cited as an indication of the lack of consensus on management of CVT a single-institution survey by other investigators of the practices of physicians in various specialties. Forty-nine percent of respondents indicated they anticoagulate patients with CVT; 51% don’t. Of those who did, 62% prescribed low-molecular-weight heparin and 11% intravenous heparin. Fifty-eight percent of physicians who anticoagulated did so for 3 months, 30% for 6. And 46% of physicians used an inferior vena cava (IVC) filter when anticoagulation was contraindicated (Vascular. 2014 Apr;22[2]:93-7).
That rate of IVC placement “seemed really high” given the paucity of supporting evidence for safety and efficacy of filter placement in the setting of CVT, so Dr. Rodriguez and coinvestigators decided to conduct a retrospective review of practices at Northwestern Memorial Hospital. He explained the study hypothesis: “Our thinking was that these kinds of thrombi are associated with low risk of propagation and pulmonary embolism, and they can and should be managed conservatively.”
Of 647 patients with isolated thrombosis of the anterior and posterior tibial, soleal, peroneal, or gastrocnemius veins, 44% received an IVC filter, and the rest got medical treatment alone. Of the 362 patients managed medically, 49% received therapeutic anticoagulation, 12% got low-dose prophylactic anticoagulation, and 39% underwent surveillance without anticoagulation.
The primary outcome was the incidence of venous thromboembolic complications – that is, propagation of DVT or pulmonary embolism. The incidence was 35% in the surveillance-only group, 30% with prophylactic anticoagulation, and 10% in patients who got therapeutic anticoagulation.
Of note, the rate of the most feared complication, pulmonary embolism, was low and similar in the filter recipients and medically managed group: 2.5% in the IVC group, 3.3% with medical management.
“Distal vein thromboses have low rates of pulmonary embolism, regardless of whether or not they are so-called protected with a filter. On the other hand, a filter was associated with a 10% rate of complications. I have to make clear that these were radiographic abnormalities – tilting, migration, caval perforation – that didn’t have clinical consequences, but we were very aggressive in removing the IVC filters, and I’m guessing if they’d been left inside they would create problems in the long term,” Dr. Rodriguez said.
An important point about this study is that these were all sick patients, and most were hospitalized. The filter recipients and medical groups differed in key ways. For example, 49% of the filter patients had a malignancy, and 56% had a baseline history of venous thromboembolic events, compared with 26% and 16%, respectively, of the medical group. For that reason, the investigators performed propensity score matching and came up with 157 closely matched patient pairs. The outcomes remained unchanged.
Of course, this was a retrospective study, with its inherent limitations, but Dr. Rodriguez characterized the published randomized trials on management of CVT as “small and limited.” The most frequently quoted study is the double-blind multicenter CACTUS trial, which randomized 252 outpatients with symptomatic CVT to 6 weeks of low-molecular-weight heparin or placebo and found no difference in the rates of proximal extension of venous thromboembolic events (Lancet Haematol. 2016 Dec;3[12]:e556-62). But these were all low-risk patients. Prior DVT or malignancy were exclusion criteria, so this was a very different population than treated at Northwestern.
Based upon the results of the retrospective study at Northwestern, which have been published (J Vasc Surg Venous Lymphat Disord. 2017 Jan;5[1]:25-32), the vascular surgery service has developed a management algorithm for DVT management based upon the lesion site. If a patient is unable to undergo anticoagulation, venous duplex ultrasound is repeated at 1 week. If the imaging shows propagation into the popliteal vein and anticoagulation remains contraindicated, only then is placement of an IVC filter warranted.
Dr. Rodriguez reported serving as a paid speaker for Abbott, Endologix, and W.L. Gore.
CHICAGO – The use of therapeutic-dose anticoagulation in hospitalized patients with calf vein thrombosis significantly reduces the risk of venous thromboembolic complications, compared with lower-dose prophylactic anticoagulation or surveillance alone, Heron E. Rodriguez, MD, said at a symposium on vascular surgery sponsored by Northwestern University.
Moreover, placement of an inferior vena cava filter in patients with calf vein thrombosis when anticoagulation is contraindicated accomplishes nothing beneficial and had a 10% complication rate in a large retrospective single-center study, added Dr. Rodriguez of Northwestern University, Chicago.
Deep vein thrombosis (DVT) remains a significant source of morbidity and mortality despite worldwide awareness of the problem.
“Specifically, calf vein thrombosis [CVT] is very common, and we know that in some series up to 30% of patients end up propagating proximally if they’re not treated, and a good number of them develop chronic venous insufficiency, with long-term consequences,” he noted.
“Unfortunately there is no consensus regarding treatment. The guidelines are very vague. For example, there is no mention [in current American College of Chest Physicians guidelines] of how to manage muscular vein thrombosis and much ambiguity on how to treat calf vein thrombosis,” he continued.
Dr. Rodriguez cited as an indication of the lack of consensus on management of CVT a single-institution survey by other investigators of the practices of physicians in various specialties. Forty-nine percent of respondents indicated they anticoagulate patients with CVT; 51% don’t. Of those who did, 62% prescribed low-molecular-weight heparin and 11% intravenous heparin. Fifty-eight percent of physicians who anticoagulated did so for 3 months, 30% for 6. And 46% of physicians used an inferior vena cava (IVC) filter when anticoagulation was contraindicated (Vascular. 2014 Apr;22[2]:93-7).
That rate of IVC placement “seemed really high” given the paucity of supporting evidence for safety and efficacy of filter placement in the setting of CVT, so Dr. Rodriguez and coinvestigators decided to conduct a retrospective review of practices at Northwestern Memorial Hospital. He explained the study hypothesis: “Our thinking was that these kinds of thrombi are associated with low risk of propagation and pulmonary embolism, and they can and should be managed conservatively.”
Of 647 patients with isolated thrombosis of the anterior and posterior tibial, soleal, peroneal, or gastrocnemius veins, 44% received an IVC filter, and the rest got medical treatment alone. Of the 362 patients managed medically, 49% received therapeutic anticoagulation, 12% got low-dose prophylactic anticoagulation, and 39% underwent surveillance without anticoagulation.
The primary outcome was the incidence of venous thromboembolic complications – that is, propagation of DVT or pulmonary embolism. The incidence was 35% in the surveillance-only group, 30% with prophylactic anticoagulation, and 10% in patients who got therapeutic anticoagulation.
Of note, the rate of the most feared complication, pulmonary embolism, was low and similar in the filter recipients and medically managed group: 2.5% in the IVC group, 3.3% with medical management.
“Distal vein thromboses have low rates of pulmonary embolism, regardless of whether or not they are so-called protected with a filter. On the other hand, a filter was associated with a 10% rate of complications. I have to make clear that these were radiographic abnormalities – tilting, migration, caval perforation – that didn’t have clinical consequences, but we were very aggressive in removing the IVC filters, and I’m guessing if they’d been left inside they would create problems in the long term,” Dr. Rodriguez said.
An important point about this study is that these were all sick patients, and most were hospitalized. The filter recipients and medical groups differed in key ways. For example, 49% of the filter patients had a malignancy, and 56% had a baseline history of venous thromboembolic events, compared with 26% and 16%, respectively, of the medical group. For that reason, the investigators performed propensity score matching and came up with 157 closely matched patient pairs. The outcomes remained unchanged.
Of course, this was a retrospective study, with its inherent limitations, but Dr. Rodriguez characterized the published randomized trials on management of CVT as “small and limited.” The most frequently quoted study is the double-blind multicenter CACTUS trial, which randomized 252 outpatients with symptomatic CVT to 6 weeks of low-molecular-weight heparin or placebo and found no difference in the rates of proximal extension of venous thromboembolic events (Lancet Haematol. 2016 Dec;3[12]:e556-62). But these were all low-risk patients. Prior DVT or malignancy were exclusion criteria, so this was a very different population than treated at Northwestern.
Based upon the results of the retrospective study at Northwestern, which have been published (J Vasc Surg Venous Lymphat Disord. 2017 Jan;5[1]:25-32), the vascular surgery service has developed a management algorithm for DVT management based upon the lesion site. If a patient is unable to undergo anticoagulation, venous duplex ultrasound is repeated at 1 week. If the imaging shows propagation into the popliteal vein and anticoagulation remains contraindicated, only then is placement of an IVC filter warranted.
Dr. Rodriguez reported serving as a paid speaker for Abbott, Endologix, and W.L. Gore.
EXPERT ANALYSIS FROM NORTHWESTERN VASCULAR SYMPOSIUM
Tumor Necrosis Factor α Inhibitors in the Treatment of Toxic Epidermal Necrolysis
Toxic epidermal necrolysis (TEN) is a rare, life-threatening adverse drug reaction with an estimated incidence of 0.4 to 1.9 cases per million persons per year worldwide and an estimated mortality rate of 25% to 35%.1,2 This dermatologic emergency is characterized by extensive detachment of the epidermis and erosions of the mucous membranes secondary to massive keratinocyte cell death via apoptosis, evolving quickly into full-thickness epidermal necrosis.
Primary treatment of TEN includes (1) prompt discontinuation of the suspected medication; (2) rapid transfer to an intensive care unit, burn center, or other specialty unit; and (3) supportive care, including wound care, fluid and electrolyte maintenance, and treatment of infections. Aside from the primary treatment, controversy remains over the most effective adjunctive therapy for TEN, as none has proven consistent superiority over well-conducted primary treatment alone. Therefore, established therapeutic guidelines do not exist.1-3
The use of adjunctive systemic therapy in TEN (eg, corticosteroids, intravenous immunoglobulin [IVIG], cyclosporine, plasmapheresis, granulocyte-colony stimulating factor) is based primarily on theories of pathogenesis, which unfortunately remain unclear. Activated CD8+ T cells are thought to increase the expression and production of granulysin, granzyme B, and perforins, leading to keratinocyte apoptosis. Fas ligand and tumor necrosis factor α (TNF-α) also are implicated as secondary mediators of cell death via the inducible nitric oxide synthase pathway.1,4-6
Since TNF-α was found to be elevated in serum and blister fluid in patients with TEN,7,8 medications aimed at decreasing the TNF-α concentration, such as pentoxifylline (PTX) and thalidomide, have been attempted for treatment.9,10 Biologic inhibitors of TNF-α, such as infliximab and etanercept, are novel therapeutic options in the treatment of TEN, as numerous reports document their successful use in the treatment of this disease.11-24 The purpose of this study is to systematically review the current literature on the use of TNF-α antagonists in the treatment of TEN.
METHODS
A PubMed search of all available articles indexed for MEDLINE using the terms toxic epidermal necrolysis and TNF-alpha and pentoxifylline or thalidomide or infliximab or etanercept or adalimumab was conducted.
RESULTS
Sixteen articles published between 1994 and 2014 were retrieved from PubMed and reviewed.9-24 Fourteen articles were case reports and case series involving the use of TNF-α inhibitors as either monotherapy, second-line agents, or in combination with other medications in the treatment of TEN, providing a total of 28 patients.9,11-23 Two articles were prospective trials, one evaluating the efficacy of thalidomide10 and the other infliximab24 in treating TEN. All studies implemented primary treatment (ie, prompt discontinuation of the suspected medication and aggressive supportive care) in addition to TNF-α inhibition.
Pentoxifylline
The first case report describing the use of an anti–TNF-α inhibitor for TEN was with PTX in 1994.9 Pentoxifylline, a vasoactive drug with immunomodulatory properties including the downregulation of TNF-α synthesis, was used to treat a 26-year-old woman with TEN on phenylhydantoin 15 days following resection of a grade II astrocytoma. The patient initially received intravenous N-acetylcysteine (NAC) (9 g once daily) and S-adenosyl-L-methionine (100 mg once daily) for antioxidant effects. On the second day of treatment, intravenous PTX (900 mg once daily) was added for TNF-α inhibition. Following PTX administration, the investigators reported quick stabilization of the eruption and achievement of reepithelialization after 7 days of therapy. Upon cessation of PTX therapy, a recurrence of generalized erythema occurred, suggesting a relapse of TEN; therefore, PTX was reinitiated for an additional 3 days, and the patient’s skin remained clear.9
Thalidomide
The earliest prospective trial we reviewed using anti–TNF-α therapy in TEN occurred in 1998 with thalidomide, a moderate inhibitor of TNF-α.10 In this randomized controlled trial, 22 TEN patients received either a 5-day course of thalidomide (400 mg once daily) or placebo. There was increased mortality in the thalidomide group (10/12 [83.3%]) versus the placebo group (3/10 [30.0%]). Additionally, the plasma TNF-α concentrations in the thalidomide group were higher than the control group. This study was stopped prematurely due to the excess mortality in the thalidomide group.10
Biologic TNF-α Antagonists
Following the PTX case report and the thalidomide trial, there was increased interest in using newer-generation TNF-α inhibitors, such as the monoclonal antibody infliximab or the fusion protein etanercept, in the treatment of TEN. To date, there are 10 known published case reports,11,12,15-21,23 3 case series,13,14,22 and 1 trial24 describing the use of these agents; however, treatment protocols vary. Categories of treatment protocols include the use of TNF-α inhibitors as monotherapy, following failure of other systemic agents, and in combination with other systemic therapies.
TNF-α Inhibitors as Monotherapy
Review of the literature yielded 2 case reports using infliximab monotherapy11,12 and 2 case series using infliximab or etanercept monotherapy13,14 with a total of 14 patients (Table 1). Fischer et al11 was the first of these reports to describe a patient successfully treated with supportive care and a single dose of infliximab 5 mg/kg. The dose was given 4 days after the onset of symptoms, and the rapid progression of the disease was stopped, with complete recovery in less than 4 weeks.11 Hunger et al12 also described the successful treatment of a patient using a similar protocol: a single dose of infliximab 5 mg/kg given 3 days after symptom onset. Epidermal detachment was abated within 24 hours and the patient had almost complete reepithelialization within 5 days.12 In a case series published by Zárate-Correa et al,13 2 patients with near 100% body surface area involvement were successfully treated with a single dose of infliximab 300 mg. Although both of these patients experienced fairly rapid recoveries, one patient’s course was complicated by methicillin-resistant Staphylococcus aureus bacteremia.13 Paradisi et al14 described 10 consecutive patients treated with a single dose of etanercept 50 mg given within 6 hours of hospital admission and within 72 hours of symptom onset. The SCORTEN (SCORe of Toxic Epidermal Necrolysis) scale—a severity-of-illness assessment for TEN based on body surface area involvement, comorbidities, and metabolic abnormalities—was used to predict mortality in these patients. The investigators reported an expected mortality of 46.9%; however, the observed mortality was 0%, and there were no reported infections.14
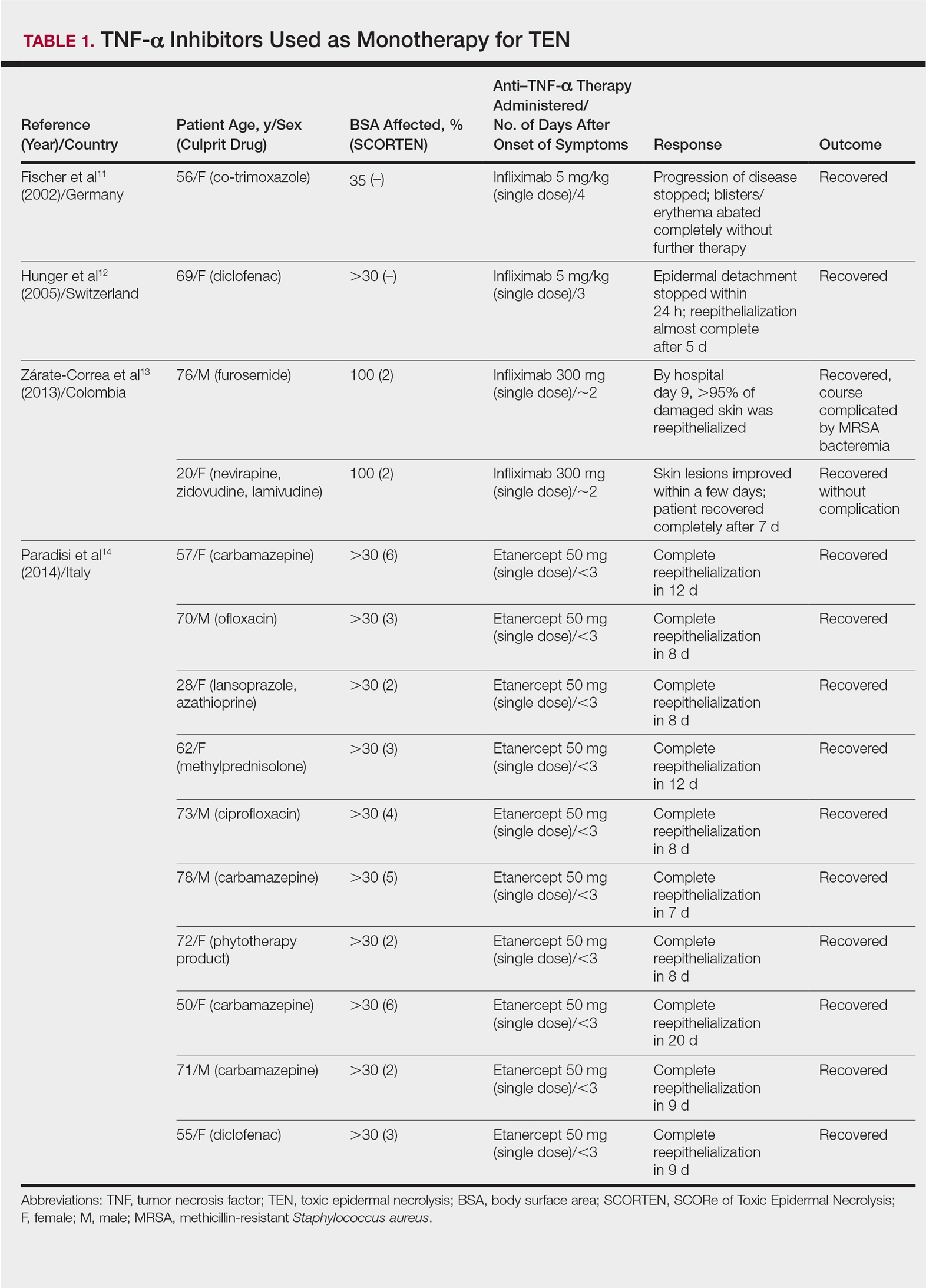
TNF-α Inhibitors Following Failure of Other Systemic Agents in TEN
Seven case reports and 1 case series using anti–TNF-α therapy following failure of other systemic agents were reviewed for a total of 9 patients (3 pediatric/adolescent patients, 6 adult patients)(Table 2).13,15-21 Seven patients were treated with infliximab,13,15,17,19-21 and the remaining 2 patients were treated with etanercept.16,18 All patients were treated initially with corticosteroids and/or IVIG. In each case, anti–TNF-α therapy was introduced when prior treatment failed to halt the progression of TEN. Most reports claimed a rapid and beneficial response to anti–TNF-α therapy. Eight of 9 (88.9%) patients recovered.13,15,17-21 Famularo et al16 described 1 patient who was treated with 2 doses of etanercept following prednisolone but died on the tenth day of hospitalization secondary to disseminated intravascular coagulation and multiorgan failure; however, the patient reportedly had near-complete reepithelialization of the skin on the sixth day of the hospital course.16 Of the 8 surviving patients, 3 (37.5%) experienced hospital courses complicated by nosocomial gram-negative bacteremia, including Pseudomonas aeruginosa, Escherichia coli, and Klebsiella pneumoniae.13,15 Interestingly, a patient described by Worsnop et al20 developed erosive lichen planus of the mouth and vulva 31 days after infliximab infusion.
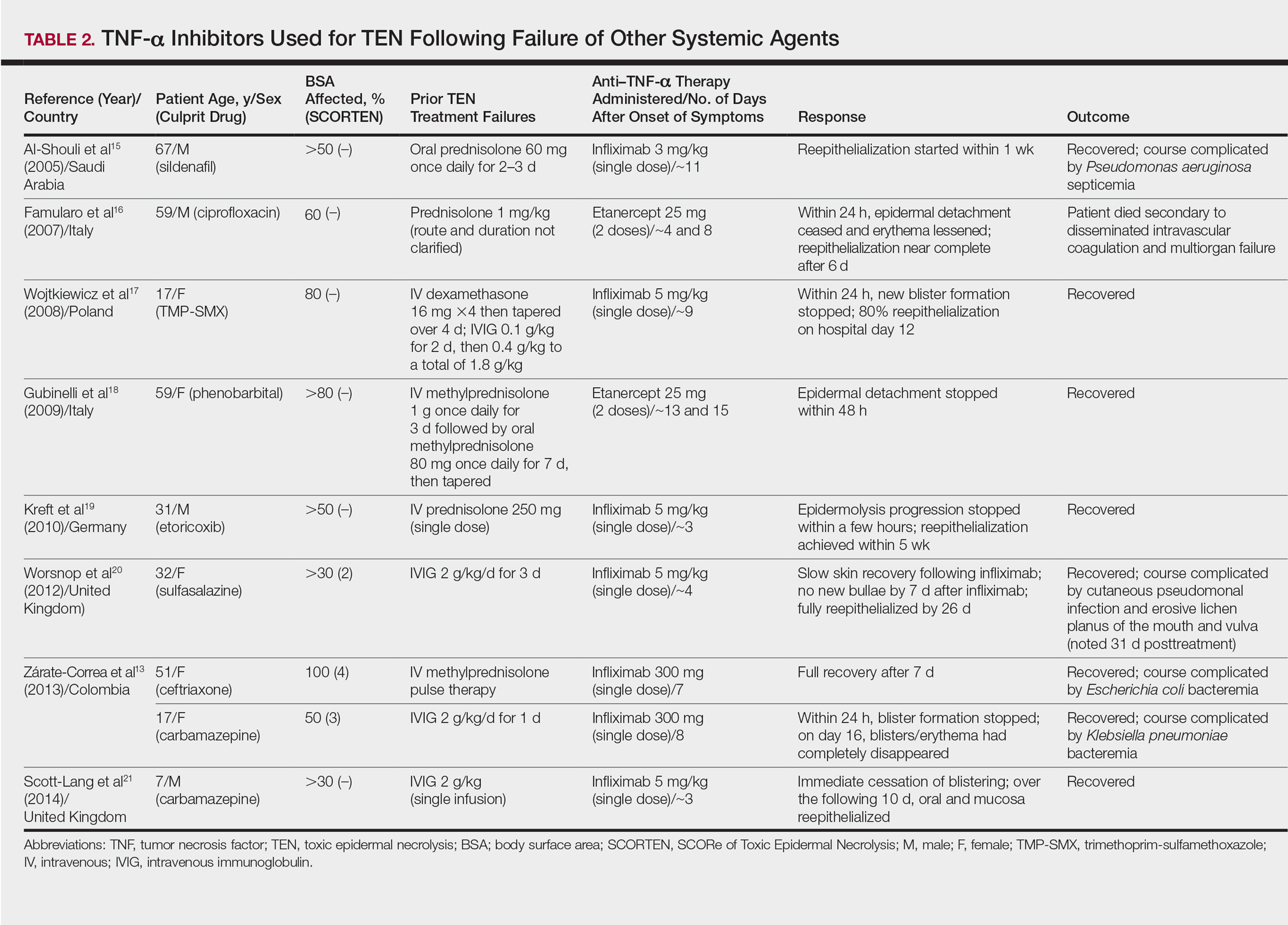
Combination of TNF-α Inhibitor With Other Systemic Agents in TEN
One case series22 and 1 case report23 using infliximab in combination with other systemic therapies were reviewed with a total of 4 patients (Table 3). Both reports utilized the same treatment protocol, which consisted of a single bolus of intravenous methylprednisolone 500 mg followed by a single dose of infliximab 5 mg/kg and then IVIG 2 g/kg over 5 days. Three of 4 (75%) patients recovered.22,23 Gaitanis et al22 reported a patient who died on the ninth day of hospitalization secondary to multiorgan dysfunction caused by a catheter-related bacteremia. Similar to the patient described by Famularo et al,16 this patient also was noted to have remarkably improved skin prior to death. Two of the other 3 patients that survived had their hospital course complicated by infection, requiring antibiotics.22 In the Gaitanis et al22 series, the average predicted mortality according to a SCORTEN assessment was 50.8%; however, mortality was observed in 33.3% (1/3) of patients in the case series.
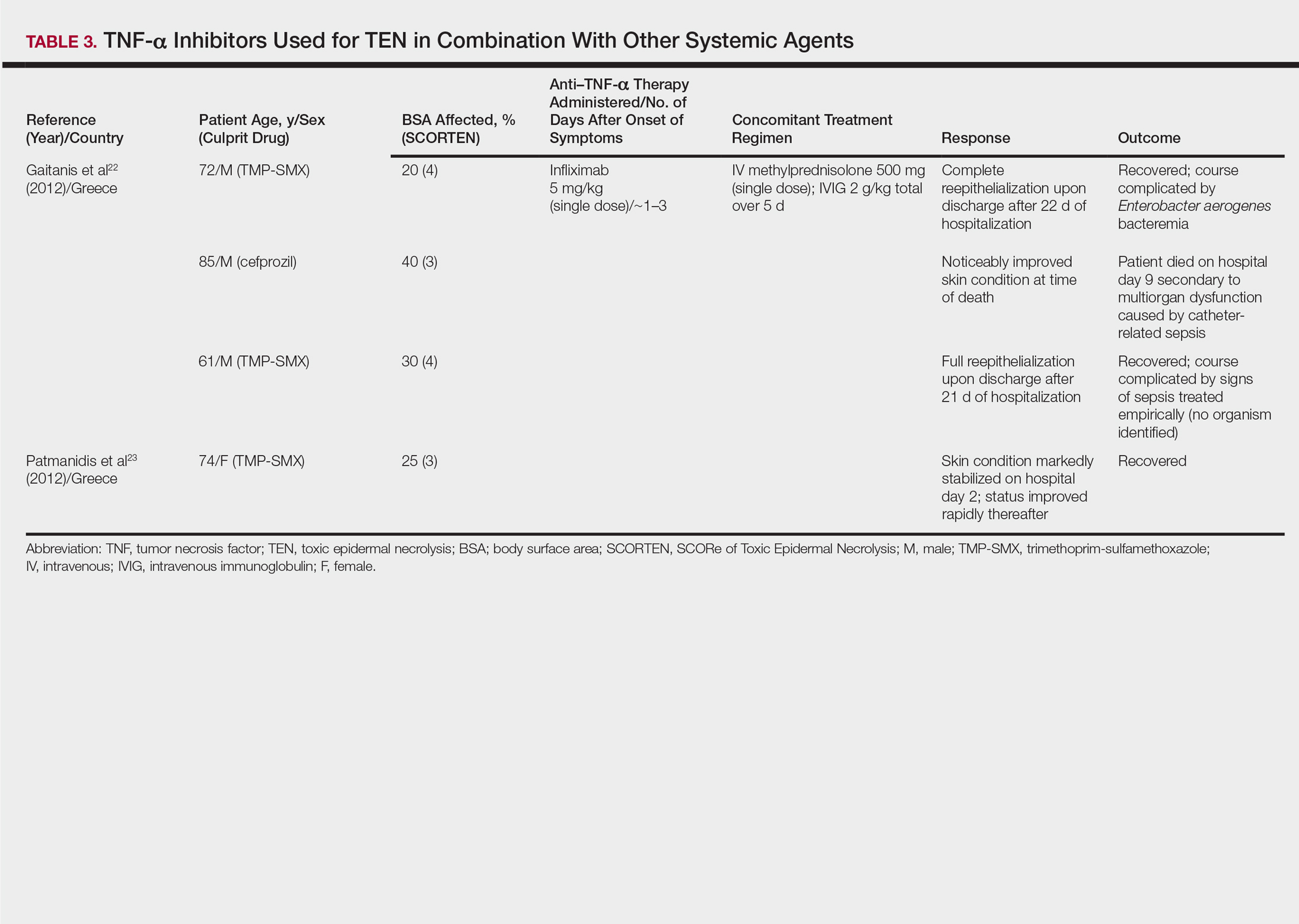
N-Acetylcysteine and Infliximab
The combination of NAC and infliximab was studied in a randomized controlled trial using TNF-α inhibition in TEN.24 In this study, 10 patients were admitted to a burn unit and treated with either 3 doses of intravenous NAC (150 mg/kg per dose) plus 1 dose of infliximab 5 mg/kg or NAC alone. Unlike some of the previously described articles, Paquet et al24 utilized an illness auxiliary score (IAS), which predicts both disease duration and mortality. An IAS was taken at admission and again 48 hours after completion of NAC and/or infliximab administration. The mean clinical IAS score was reported to have remained unchanged at treatment completion in the NAC group and slightly worsened in the NAC-infliximab group. One patient died in the NAC group and 2 patients died in the NAC-infliximab group, each due to infection. These fatalities corresponded to a mean mortality of 20% in the NAC-treated group and 40% for the NAC-infliximab group. To compare, the predicted mortalities based on the IAS were 20.4% and 21.4%, respectively.24
COMMENT
Tumor necrosis factor α inhibition in the treatment of TEN was first utilized in the 1990s with PTX and thalidomide.9,10 In 1994, PTX in addition to antioxidant therapy was found to successfully treat a 26-year-old woman with TEN attributed to anticonvulsant therapy.9 Other reports of PTX in the treatment of TEN were not found; however, there is a case series describing the successful treatment of 2 pediatric patients with Stevens-Johnson syndrome (SJS) and SJS-TEN overlap with PTX.25 Thalidomide, however, proved detrimental to patients with TEN as evidenced by an increased mortality in the 1998 trial.10 Paradoxically, the treatment group was found to have increased rather than decreased TNF-α concentrations, which was hypothesized to be the cause of increased mortality. This finding furthered the theory that TNF-α is an important mediator in TEN pathogenesis and a potential novel target in disease management.10
Since the PTX case report and the thalidomide trial, many physicians have reported the beneficial effects of biologic TNF-α inhibitors in the course of TEN; however, most of the literature is composed of case reports and case series describing a small number of patients. Therefore, the beneficial effects of anti–TNF-α therapy in TEN cannot be conclusively derived. Furthermore, cases using TNF-α inhibitors in combination with or after other systemic agents complicate the effects of TNF-α inhibitors themselves. Most of these case reports and case series describe the beneficial effects of TNF-α inhibitors in TEN; however, it is important to remember that cases in which these agents were ineffective are less likely to be published. The strongest evidence for TNF-α inhibitor use in the treatment TEN comes from the Paradisi et al14 case series, which showed a decrease in expected mortality with etanercept monotherapy in a relatively large cohort of patients. However, when evaluated prospectively by Paquet et al,24 there was no benefit seen by adding infliximab to NAC therapy and possibly an increased mortality in the group treated with both agents.
In the cases reviewed, a total of 32 patients were treated with infliximab or etanercept, and of these patients there were 4 deaths (12.5%).16,22,24 Three deaths were attributed to infection and 1 was attributed to disseminated intravascular coagulation. Furthermore, infection complicated the hospital course of 9 (28.1%) patients.13,15,22,24 The bacteria cultured from these patients included methicillin-resistant S aureus, P aeruginosa, E coli, Enterobacter aerogenes, and K pneumoniae. Patients who received TNF-α antagonists in combination with or after other systemic immunosuppressants appeared to have a higher incidence of infections. All patients treated with TNF-α antagonists in TEN should undergo careful evaluation and monitoring for infections due to the immunosuppressant effect of these drugs.
In our review, a total of 3 pediatric/adolescent patients received a TNF-α inhibitor for the treatment of TEN.13,17,21 Two patients received infliximab as a second-line medication after failure of IVIG to arrest progression of disease13,17 and one patient received infliximab as a second-line medication after dexamethasone.21 Each of these patients recovered without any reported infections or long-term complications.
Although excluded from this review, both infliximab and etanercept have been reported to show benefit in acute generalized exanthematous pustulosis/TEN overlap.26,27 Interestingly, in postmarketing surveillance, rare reports have implicated both infliximab and etanercept in causing both SJS and TEN.28 Also, there have been case reports of adalimumab causing SJS, but no cases of it causing TEN were identified.29,30
CONCLUSION
Rapid discontinuation of the culprit drug and aggressive supportive care remain the primary treatment of TEN. Tumor necrosis factor α inhibitors as monotherapy or as second-line agents show promise in the treatment of this complex disease state in both the adult and pediatric populations. The risks of these potent immunosuppressants must be weighed, and if administered, patients must be closely monitored for infections. Additional studies are needed to further characterize the role of TNF-α inhibition in the treatment of TEN.
- Schwartz R, McDonough P, Lee B. Toxic epidermal necrolysis: part I. introduction, history, classification, clinical features, systemic manifestations, etiology, and immunopathogenesis. J Am Acad Dermatol. 2013;69:173-186.
- Schwartz R, McDonough P, Lee B. Toxic epidermal necrolysis: part II. prognosis, sequelae, diagnosis, differential diagnosis, prevention, and treatment. J Am Acad Dermatol. 2013;69:187-203.
- Fernando S. The management of toxic epidermal necrolysis. Australas J Dermatol. 2012;55:165-171.
- Paquet P, Paquet F, Saleh W, et al. Immunoregulatory effector cells in drug-induced toxic epidermal necrolysis. Am J Dermatopathol. 2000;22:413-417.
- Nassif A, Moslehi H, Le Gouvello S, et al. Evaluation of the potential role of cytokines in toxic epidermal necrolysis. J Invest Dermatol. 2004;123:850-855.
- Viard-Leveugle I, Gaide O, Jankovic D, et al. TNF-α and INF-γ are potential inducers of Fas-mediated keratinocyte apoptosis thought activation of inducible nitric oxide synthase in toxic epidermal necrolysis. J Invest Dermatol. 2013;133:489-498.
- Paquet P, Pierard G. Soluble fractions of tumor necrosis factor-alpha, interleukin-6 and of their receptors in toxic epidermal necrolysis: a comparison with second-degree burns. Int J Mol Med. 1998;1:459-462.
- Correia O, Delgado L, Barbosa I, et al. Increased interleukin 10, tumor necrosis factor alpha, and interleukin 6 levels in blister fluid of toxic epidermal necrolysis. J Am Acad Dermatol. 2002;47:58-62.
- Redondo P, Rutz de Erenchun F, Iglesias M, et al. Toxic epidermal necrolysis. treatment with pentoxifylline. Br J Dermatol. 1994;130:688-689.
- Wolkenstein P, Latarjet J, Roujeau J, et al. Randomised comparison of thalidomide versus placebo in toxic epidermal necrolysis. Lancet. 1998;352:1586-1589.
- Fischer M, Fiedler E, Marsch W, et al. Antitumour necrosis factor-alpha antibodies (infliximab) in the treatment of a patient with toxic epidermal necrolysis. Br J Dermatol. 2002;146:707-708.
- Hunger R, Hunziker T, Buettiker U, et al. Rapid resolution of toxic epidermal necrolysis with anti-TNF-alpha treatment. J Allergy Clin Immunol. 2005;116:923-924.
- Zárate-Correa LC, Carrillo-Gómez DC, Ramírez-Escobar AF, et al. Toxic epidermal necrolysis successfully treated with infliximab. J Investig Allergol Clin Immunol. 2013;23:61-63.
- Paradisi A, Abeni D, Bergamo F, et al. Etanercept therapy for toxic epidermal necrolysis. J Am Acad Dermatol. 2014;71:278-283.
- Al-Shouli S, Bogusz M, Al Tufail M, et al. Toxic epidermal necrosis associated with high intake of sildenafil and its response to infliximab. Acta Derm Venereol. 2005;85:534-553.
- Famularo G, Di Dona B, Canzona F, et al. Etanercept for toxic epidermal necrolysis. Ann Pharmacother. 2007;41:1083-1084.
- Wojtkiewicz A, Wysocki M, Fortuna J, et al. Beneficial and rapid effect of infliximab on the course of toxic epidermal necrolysis. Acta Derm Venereol. 2008;88:420-421.
- Gubinelli E, Canzona F, Tonanzi T, et al. Toxic epidermal necrolysis successfully treated with etanercept. J Dermatol. 2009;36:150-153.
- Kreft B, Wohlrab J, Bramsiepe I, et al. Etoricoxib-induced toxic epidermal necrolysis: successful treatment with infliximab. J Dermatol. 2010;37:904-906.
- Worsnop F, Wee J, Moosa Y, et al. Reaction to biological drugs: infliximab for the treatment of toxic epidermal necrolysis subsequently triggering erosive lichen planus. Clin Exp Dermatol. 2012;37:879-881.
- Scott-Lang V, Tidman M, McKay D. Toxic epidermal necrolysis in a child successfully treated with infliximab. Pediatr Dermatol. 2014;31:532-534.
- Gaitanis G, Spyridonos P, Patmanidis K, et al. Treatment of toxic epidermal necrolysis with the combination of infliximab and high-dose intravenous immunoglobulins. Dermatology. 2012;224:134-139.
- Patmanidis K, Sidiras A, Dolianitis K, et al. Combination of infliximab and high-dose intravenous immunoglobulin for toxic epidermal necrolysis: successful treatment of an elderly patient. Case Rep Dermatol Med. 2012;2012:915314.
- Paquet P, Jennes S, Rousseua A, et al. Effect of N-acetylcysteine combined with infliximab on toxic epidermal necrolysis: a proof-of-concept study. Burns. 2014;1:1-6.
- Sanclemente G, De le Rouche C, Escobar C, et al. Pentoxifylline in toxic epidermal necrolysis and Stevens-Johnson syndrome. Int J Dermatol. 1998;38:878-879.
- Meiss F, Helmbold P, Meykadeh N, et al. Overlap of acute generalized exanthematous pustulosis and toxic epidermal necrolysis: response to antitumor necrosis factor-alpha antibody infliximab: report of three cases. J Eur Acad Dermatol Venereol. 2007;21:717-719.
- Sadighha A. Etanercept in the treatment of a patient with acute generalized exanthematous pustulosis/toxic epidermal necrolysis: definition of a new model based on translational research. Int J Dermatol. 2009;48:913-914.
- Borras-Blasco J, Navarro-Ruiz A, Borras C, et al. Adverse cutaneous reactions induced by TNF-α antagonist therapy. South Med J. 2009;102:1133-1140.
- Muna S, Lawrance I. Stevens-Johnson syndrome complicating adalimumab therapy in Crohn’s disease. World J Gastroenterol. 2009;15:4449-4452.
- Mounach A, Rezgi A, Nouijai A, et al. Stevens-Johnson syndrome complicating adalimumab therapy in rheumatoid arthritis disease. Rheumatol Int. 2013;33:1351-1353.
Toxic epidermal necrolysis (TEN) is a rare, life-threatening adverse drug reaction with an estimated incidence of 0.4 to 1.9 cases per million persons per year worldwide and an estimated mortality rate of 25% to 35%.1,2 This dermatologic emergency is characterized by extensive detachment of the epidermis and erosions of the mucous membranes secondary to massive keratinocyte cell death via apoptosis, evolving quickly into full-thickness epidermal necrosis.
Primary treatment of TEN includes (1) prompt discontinuation of the suspected medication; (2) rapid transfer to an intensive care unit, burn center, or other specialty unit; and (3) supportive care, including wound care, fluid and electrolyte maintenance, and treatment of infections. Aside from the primary treatment, controversy remains over the most effective adjunctive therapy for TEN, as none has proven consistent superiority over well-conducted primary treatment alone. Therefore, established therapeutic guidelines do not exist.1-3
The use of adjunctive systemic therapy in TEN (eg, corticosteroids, intravenous immunoglobulin [IVIG], cyclosporine, plasmapheresis, granulocyte-colony stimulating factor) is based primarily on theories of pathogenesis, which unfortunately remain unclear. Activated CD8+ T cells are thought to increase the expression and production of granulysin, granzyme B, and perforins, leading to keratinocyte apoptosis. Fas ligand and tumor necrosis factor α (TNF-α) also are implicated as secondary mediators of cell death via the inducible nitric oxide synthase pathway.1,4-6
Since TNF-α was found to be elevated in serum and blister fluid in patients with TEN,7,8 medications aimed at decreasing the TNF-α concentration, such as pentoxifylline (PTX) and thalidomide, have been attempted for treatment.9,10 Biologic inhibitors of TNF-α, such as infliximab and etanercept, are novel therapeutic options in the treatment of TEN, as numerous reports document their successful use in the treatment of this disease.11-24 The purpose of this study is to systematically review the current literature on the use of TNF-α antagonists in the treatment of TEN.
METHODS
A PubMed search of all available articles indexed for MEDLINE using the terms toxic epidermal necrolysis and TNF-alpha and pentoxifylline or thalidomide or infliximab or etanercept or adalimumab was conducted.
RESULTS
Sixteen articles published between 1994 and 2014 were retrieved from PubMed and reviewed.9-24 Fourteen articles were case reports and case series involving the use of TNF-α inhibitors as either monotherapy, second-line agents, or in combination with other medications in the treatment of TEN, providing a total of 28 patients.9,11-23 Two articles were prospective trials, one evaluating the efficacy of thalidomide10 and the other infliximab24 in treating TEN. All studies implemented primary treatment (ie, prompt discontinuation of the suspected medication and aggressive supportive care) in addition to TNF-α inhibition.
Pentoxifylline
The first case report describing the use of an anti–TNF-α inhibitor for TEN was with PTX in 1994.9 Pentoxifylline, a vasoactive drug with immunomodulatory properties including the downregulation of TNF-α synthesis, was used to treat a 26-year-old woman with TEN on phenylhydantoin 15 days following resection of a grade II astrocytoma. The patient initially received intravenous N-acetylcysteine (NAC) (9 g once daily) and S-adenosyl-L-methionine (100 mg once daily) for antioxidant effects. On the second day of treatment, intravenous PTX (900 mg once daily) was added for TNF-α inhibition. Following PTX administration, the investigators reported quick stabilization of the eruption and achievement of reepithelialization after 7 days of therapy. Upon cessation of PTX therapy, a recurrence of generalized erythema occurred, suggesting a relapse of TEN; therefore, PTX was reinitiated for an additional 3 days, and the patient’s skin remained clear.9
Thalidomide
The earliest prospective trial we reviewed using anti–TNF-α therapy in TEN occurred in 1998 with thalidomide, a moderate inhibitor of TNF-α.10 In this randomized controlled trial, 22 TEN patients received either a 5-day course of thalidomide (400 mg once daily) or placebo. There was increased mortality in the thalidomide group (10/12 [83.3%]) versus the placebo group (3/10 [30.0%]). Additionally, the plasma TNF-α concentrations in the thalidomide group were higher than the control group. This study was stopped prematurely due to the excess mortality in the thalidomide group.10
Biologic TNF-α Antagonists
Following the PTX case report and the thalidomide trial, there was increased interest in using newer-generation TNF-α inhibitors, such as the monoclonal antibody infliximab or the fusion protein etanercept, in the treatment of TEN. To date, there are 10 known published case reports,11,12,15-21,23 3 case series,13,14,22 and 1 trial24 describing the use of these agents; however, treatment protocols vary. Categories of treatment protocols include the use of TNF-α inhibitors as monotherapy, following failure of other systemic agents, and in combination with other systemic therapies.
TNF-α Inhibitors as Monotherapy
Review of the literature yielded 2 case reports using infliximab monotherapy11,12 and 2 case series using infliximab or etanercept monotherapy13,14 with a total of 14 patients (Table 1). Fischer et al11 was the first of these reports to describe a patient successfully treated with supportive care and a single dose of infliximab 5 mg/kg. The dose was given 4 days after the onset of symptoms, and the rapid progression of the disease was stopped, with complete recovery in less than 4 weeks.11 Hunger et al12 also described the successful treatment of a patient using a similar protocol: a single dose of infliximab 5 mg/kg given 3 days after symptom onset. Epidermal detachment was abated within 24 hours and the patient had almost complete reepithelialization within 5 days.12 In a case series published by Zárate-Correa et al,13 2 patients with near 100% body surface area involvement were successfully treated with a single dose of infliximab 300 mg. Although both of these patients experienced fairly rapid recoveries, one patient’s course was complicated by methicillin-resistant Staphylococcus aureus bacteremia.13 Paradisi et al14 described 10 consecutive patients treated with a single dose of etanercept 50 mg given within 6 hours of hospital admission and within 72 hours of symptom onset. The SCORTEN (SCORe of Toxic Epidermal Necrolysis) scale—a severity-of-illness assessment for TEN based on body surface area involvement, comorbidities, and metabolic abnormalities—was used to predict mortality in these patients. The investigators reported an expected mortality of 46.9%; however, the observed mortality was 0%, and there were no reported infections.14

TNF-α Inhibitors Following Failure of Other Systemic Agents in TEN
Seven case reports and 1 case series using anti–TNF-α therapy following failure of other systemic agents were reviewed for a total of 9 patients (3 pediatric/adolescent patients, 6 adult patients)(Table 2).13,15-21 Seven patients were treated with infliximab,13,15,17,19-21 and the remaining 2 patients were treated with etanercept.16,18 All patients were treated initially with corticosteroids and/or IVIG. In each case, anti–TNF-α therapy was introduced when prior treatment failed to halt the progression of TEN. Most reports claimed a rapid and beneficial response to anti–TNF-α therapy. Eight of 9 (88.9%) patients recovered.13,15,17-21 Famularo et al16 described 1 patient who was treated with 2 doses of etanercept following prednisolone but died on the tenth day of hospitalization secondary to disseminated intravascular coagulation and multiorgan failure; however, the patient reportedly had near-complete reepithelialization of the skin on the sixth day of the hospital course.16 Of the 8 surviving patients, 3 (37.5%) experienced hospital courses complicated by nosocomial gram-negative bacteremia, including Pseudomonas aeruginosa, Escherichia coli, and Klebsiella pneumoniae.13,15 Interestingly, a patient described by Worsnop et al20 developed erosive lichen planus of the mouth and vulva 31 days after infliximab infusion.

Combination of TNF-α Inhibitor With Other Systemic Agents in TEN
One case series22 and 1 case report23 using infliximab in combination with other systemic therapies were reviewed with a total of 4 patients (Table 3). Both reports utilized the same treatment protocol, which consisted of a single bolus of intravenous methylprednisolone 500 mg followed by a single dose of infliximab 5 mg/kg and then IVIG 2 g/kg over 5 days. Three of 4 (75%) patients recovered.22,23 Gaitanis et al22 reported a patient who died on the ninth day of hospitalization secondary to multiorgan dysfunction caused by a catheter-related bacteremia. Similar to the patient described by Famularo et al,16 this patient also was noted to have remarkably improved skin prior to death. Two of the other 3 patients that survived had their hospital course complicated by infection, requiring antibiotics.22 In the Gaitanis et al22 series, the average predicted mortality according to a SCORTEN assessment was 50.8%; however, mortality was observed in 33.3% (1/3) of patients in the case series.

N-Acetylcysteine and Infliximab
The combination of NAC and infliximab was studied in a randomized controlled trial using TNF-α inhibition in TEN.24 In this study, 10 patients were admitted to a burn unit and treated with either 3 doses of intravenous NAC (150 mg/kg per dose) plus 1 dose of infliximab 5 mg/kg or NAC alone. Unlike some of the previously described articles, Paquet et al24 utilized an illness auxiliary score (IAS), which predicts both disease duration and mortality. An IAS was taken at admission and again 48 hours after completion of NAC and/or infliximab administration. The mean clinical IAS score was reported to have remained unchanged at treatment completion in the NAC group and slightly worsened in the NAC-infliximab group. One patient died in the NAC group and 2 patients died in the NAC-infliximab group, each due to infection. These fatalities corresponded to a mean mortality of 20% in the NAC-treated group and 40% for the NAC-infliximab group. To compare, the predicted mortalities based on the IAS were 20.4% and 21.4%, respectively.24
COMMENT
Tumor necrosis factor α inhibition in the treatment of TEN was first utilized in the 1990s with PTX and thalidomide.9,10 In 1994, PTX in addition to antioxidant therapy was found to successfully treat a 26-year-old woman with TEN attributed to anticonvulsant therapy.9 Other reports of PTX in the treatment of TEN were not found; however, there is a case series describing the successful treatment of 2 pediatric patients with Stevens-Johnson syndrome (SJS) and SJS-TEN overlap with PTX.25 Thalidomide, however, proved detrimental to patients with TEN as evidenced by an increased mortality in the 1998 trial.10 Paradoxically, the treatment group was found to have increased rather than decreased TNF-α concentrations, which was hypothesized to be the cause of increased mortality. This finding furthered the theory that TNF-α is an important mediator in TEN pathogenesis and a potential novel target in disease management.10
Since the PTX case report and the thalidomide trial, many physicians have reported the beneficial effects of biologic TNF-α inhibitors in the course of TEN; however, most of the literature is composed of case reports and case series describing a small number of patients. Therefore, the beneficial effects of anti–TNF-α therapy in TEN cannot be conclusively derived. Furthermore, cases using TNF-α inhibitors in combination with or after other systemic agents complicate the effects of TNF-α inhibitors themselves. Most of these case reports and case series describe the beneficial effects of TNF-α inhibitors in TEN; however, it is important to remember that cases in which these agents were ineffective are less likely to be published. The strongest evidence for TNF-α inhibitor use in the treatment TEN comes from the Paradisi et al14 case series, which showed a decrease in expected mortality with etanercept monotherapy in a relatively large cohort of patients. However, when evaluated prospectively by Paquet et al,24 there was no benefit seen by adding infliximab to NAC therapy and possibly an increased mortality in the group treated with both agents.
In the cases reviewed, a total of 32 patients were treated with infliximab or etanercept, and of these patients there were 4 deaths (12.5%).16,22,24 Three deaths were attributed to infection and 1 was attributed to disseminated intravascular coagulation. Furthermore, infection complicated the hospital course of 9 (28.1%) patients.13,15,22,24 The bacteria cultured from these patients included methicillin-resistant S aureus, P aeruginosa, E coli, Enterobacter aerogenes, and K pneumoniae. Patients who received TNF-α antagonists in combination with or after other systemic immunosuppressants appeared to have a higher incidence of infections. All patients treated with TNF-α antagonists in TEN should undergo careful evaluation and monitoring for infections due to the immunosuppressant effect of these drugs.
In our review, a total of 3 pediatric/adolescent patients received a TNF-α inhibitor for the treatment of TEN.13,17,21 Two patients received infliximab as a second-line medication after failure of IVIG to arrest progression of disease13,17 and one patient received infliximab as a second-line medication after dexamethasone.21 Each of these patients recovered without any reported infections or long-term complications.
Although excluded from this review, both infliximab and etanercept have been reported to show benefit in acute generalized exanthematous pustulosis/TEN overlap.26,27 Interestingly, in postmarketing surveillance, rare reports have implicated both infliximab and etanercept in causing both SJS and TEN.28 Also, there have been case reports of adalimumab causing SJS, but no cases of it causing TEN were identified.29,30
CONCLUSION
Rapid discontinuation of the culprit drug and aggressive supportive care remain the primary treatment of TEN. Tumor necrosis factor α inhibitors as monotherapy or as second-line agents show promise in the treatment of this complex disease state in both the adult and pediatric populations. The risks of these potent immunosuppressants must be weighed, and if administered, patients must be closely monitored for infections. Additional studies are needed to further characterize the role of TNF-α inhibition in the treatment of TEN.
Toxic epidermal necrolysis (TEN) is a rare, life-threatening adverse drug reaction with an estimated incidence of 0.4 to 1.9 cases per million persons per year worldwide and an estimated mortality rate of 25% to 35%.1,2 This dermatologic emergency is characterized by extensive detachment of the epidermis and erosions of the mucous membranes secondary to massive keratinocyte cell death via apoptosis, evolving quickly into full-thickness epidermal necrosis.
Primary treatment of TEN includes (1) prompt discontinuation of the suspected medication; (2) rapid transfer to an intensive care unit, burn center, or other specialty unit; and (3) supportive care, including wound care, fluid and electrolyte maintenance, and treatment of infections. Aside from the primary treatment, controversy remains over the most effective adjunctive therapy for TEN, as none has proven consistent superiority over well-conducted primary treatment alone. Therefore, established therapeutic guidelines do not exist.1-3
The use of adjunctive systemic therapy in TEN (eg, corticosteroids, intravenous immunoglobulin [IVIG], cyclosporine, plasmapheresis, granulocyte-colony stimulating factor) is based primarily on theories of pathogenesis, which unfortunately remain unclear. Activated CD8+ T cells are thought to increase the expression and production of granulysin, granzyme B, and perforins, leading to keratinocyte apoptosis. Fas ligand and tumor necrosis factor α (TNF-α) also are implicated as secondary mediators of cell death via the inducible nitric oxide synthase pathway.1,4-6
Since TNF-α was found to be elevated in serum and blister fluid in patients with TEN,7,8 medications aimed at decreasing the TNF-α concentration, such as pentoxifylline (PTX) and thalidomide, have been attempted for treatment.9,10 Biologic inhibitors of TNF-α, such as infliximab and etanercept, are novel therapeutic options in the treatment of TEN, as numerous reports document their successful use in the treatment of this disease.11-24 The purpose of this study is to systematically review the current literature on the use of TNF-α antagonists in the treatment of TEN.
METHODS
A PubMed search of all available articles indexed for MEDLINE using the terms toxic epidermal necrolysis and TNF-alpha and pentoxifylline or thalidomide or infliximab or etanercept or adalimumab was conducted.
RESULTS
Sixteen articles published between 1994 and 2014 were retrieved from PubMed and reviewed.9-24 Fourteen articles were case reports and case series involving the use of TNF-α inhibitors as either monotherapy, second-line agents, or in combination with other medications in the treatment of TEN, providing a total of 28 patients.9,11-23 Two articles were prospective trials, one evaluating the efficacy of thalidomide10 and the other infliximab24 in treating TEN. All studies implemented primary treatment (ie, prompt discontinuation of the suspected medication and aggressive supportive care) in addition to TNF-α inhibition.
Pentoxifylline
The first case report describing the use of an anti–TNF-α inhibitor for TEN was with PTX in 1994.9 Pentoxifylline, a vasoactive drug with immunomodulatory properties including the downregulation of TNF-α synthesis, was used to treat a 26-year-old woman with TEN on phenylhydantoin 15 days following resection of a grade II astrocytoma. The patient initially received intravenous N-acetylcysteine (NAC) (9 g once daily) and S-adenosyl-L-methionine (100 mg once daily) for antioxidant effects. On the second day of treatment, intravenous PTX (900 mg once daily) was added for TNF-α inhibition. Following PTX administration, the investigators reported quick stabilization of the eruption and achievement of reepithelialization after 7 days of therapy. Upon cessation of PTX therapy, a recurrence of generalized erythema occurred, suggesting a relapse of TEN; therefore, PTX was reinitiated for an additional 3 days, and the patient’s skin remained clear.9
Thalidomide
The earliest prospective trial we reviewed using anti–TNF-α therapy in TEN occurred in 1998 with thalidomide, a moderate inhibitor of TNF-α.10 In this randomized controlled trial, 22 TEN patients received either a 5-day course of thalidomide (400 mg once daily) or placebo. There was increased mortality in the thalidomide group (10/12 [83.3%]) versus the placebo group (3/10 [30.0%]). Additionally, the plasma TNF-α concentrations in the thalidomide group were higher than the control group. This study was stopped prematurely due to the excess mortality in the thalidomide group.10
Biologic TNF-α Antagonists
Following the PTX case report and the thalidomide trial, there was increased interest in using newer-generation TNF-α inhibitors, such as the monoclonal antibody infliximab or the fusion protein etanercept, in the treatment of TEN. To date, there are 10 known published case reports,11,12,15-21,23 3 case series,13,14,22 and 1 trial24 describing the use of these agents; however, treatment protocols vary. Categories of treatment protocols include the use of TNF-α inhibitors as monotherapy, following failure of other systemic agents, and in combination with other systemic therapies.
TNF-α Inhibitors as Monotherapy
Review of the literature yielded 2 case reports using infliximab monotherapy11,12 and 2 case series using infliximab or etanercept monotherapy13,14 with a total of 14 patients (Table 1). Fischer et al11 was the first of these reports to describe a patient successfully treated with supportive care and a single dose of infliximab 5 mg/kg. The dose was given 4 days after the onset of symptoms, and the rapid progression of the disease was stopped, with complete recovery in less than 4 weeks.11 Hunger et al12 also described the successful treatment of a patient using a similar protocol: a single dose of infliximab 5 mg/kg given 3 days after symptom onset. Epidermal detachment was abated within 24 hours and the patient had almost complete reepithelialization within 5 days.12 In a case series published by Zárate-Correa et al,13 2 patients with near 100% body surface area involvement were successfully treated with a single dose of infliximab 300 mg. Although both of these patients experienced fairly rapid recoveries, one patient’s course was complicated by methicillin-resistant Staphylococcus aureus bacteremia.13 Paradisi et al14 described 10 consecutive patients treated with a single dose of etanercept 50 mg given within 6 hours of hospital admission and within 72 hours of symptom onset. The SCORTEN (SCORe of Toxic Epidermal Necrolysis) scale—a severity-of-illness assessment for TEN based on body surface area involvement, comorbidities, and metabolic abnormalities—was used to predict mortality in these patients. The investigators reported an expected mortality of 46.9%; however, the observed mortality was 0%, and there were no reported infections.14

TNF-α Inhibitors Following Failure of Other Systemic Agents in TEN
Seven case reports and 1 case series using anti–TNF-α therapy following failure of other systemic agents were reviewed for a total of 9 patients (3 pediatric/adolescent patients, 6 adult patients)(Table 2).13,15-21 Seven patients were treated with infliximab,13,15,17,19-21 and the remaining 2 patients were treated with etanercept.16,18 All patients were treated initially with corticosteroids and/or IVIG. In each case, anti–TNF-α therapy was introduced when prior treatment failed to halt the progression of TEN. Most reports claimed a rapid and beneficial response to anti–TNF-α therapy. Eight of 9 (88.9%) patients recovered.13,15,17-21 Famularo et al16 described 1 patient who was treated with 2 doses of etanercept following prednisolone but died on the tenth day of hospitalization secondary to disseminated intravascular coagulation and multiorgan failure; however, the patient reportedly had near-complete reepithelialization of the skin on the sixth day of the hospital course.16 Of the 8 surviving patients, 3 (37.5%) experienced hospital courses complicated by nosocomial gram-negative bacteremia, including Pseudomonas aeruginosa, Escherichia coli, and Klebsiella pneumoniae.13,15 Interestingly, a patient described by Worsnop et al20 developed erosive lichen planus of the mouth and vulva 31 days after infliximab infusion.

Combination of TNF-α Inhibitor With Other Systemic Agents in TEN
One case series22 and 1 case report23 using infliximab in combination with other systemic therapies were reviewed with a total of 4 patients (Table 3). Both reports utilized the same treatment protocol, which consisted of a single bolus of intravenous methylprednisolone 500 mg followed by a single dose of infliximab 5 mg/kg and then IVIG 2 g/kg over 5 days. Three of 4 (75%) patients recovered.22,23 Gaitanis et al22 reported a patient who died on the ninth day of hospitalization secondary to multiorgan dysfunction caused by a catheter-related bacteremia. Similar to the patient described by Famularo et al,16 this patient also was noted to have remarkably improved skin prior to death. Two of the other 3 patients that survived had their hospital course complicated by infection, requiring antibiotics.22 In the Gaitanis et al22 series, the average predicted mortality according to a SCORTEN assessment was 50.8%; however, mortality was observed in 33.3% (1/3) of patients in the case series.

N-Acetylcysteine and Infliximab
The combination of NAC and infliximab was studied in a randomized controlled trial using TNF-α inhibition in TEN.24 In this study, 10 patients were admitted to a burn unit and treated with either 3 doses of intravenous NAC (150 mg/kg per dose) plus 1 dose of infliximab 5 mg/kg or NAC alone. Unlike some of the previously described articles, Paquet et al24 utilized an illness auxiliary score (IAS), which predicts both disease duration and mortality. An IAS was taken at admission and again 48 hours after completion of NAC and/or infliximab administration. The mean clinical IAS score was reported to have remained unchanged at treatment completion in the NAC group and slightly worsened in the NAC-infliximab group. One patient died in the NAC group and 2 patients died in the NAC-infliximab group, each due to infection. These fatalities corresponded to a mean mortality of 20% in the NAC-treated group and 40% for the NAC-infliximab group. To compare, the predicted mortalities based on the IAS were 20.4% and 21.4%, respectively.24
COMMENT
Tumor necrosis factor α inhibition in the treatment of TEN was first utilized in the 1990s with PTX and thalidomide.9,10 In 1994, PTX in addition to antioxidant therapy was found to successfully treat a 26-year-old woman with TEN attributed to anticonvulsant therapy.9 Other reports of PTX in the treatment of TEN were not found; however, there is a case series describing the successful treatment of 2 pediatric patients with Stevens-Johnson syndrome (SJS) and SJS-TEN overlap with PTX.25 Thalidomide, however, proved detrimental to patients with TEN as evidenced by an increased mortality in the 1998 trial.10 Paradoxically, the treatment group was found to have increased rather than decreased TNF-α concentrations, which was hypothesized to be the cause of increased mortality. This finding furthered the theory that TNF-α is an important mediator in TEN pathogenesis and a potential novel target in disease management.10
Since the PTX case report and the thalidomide trial, many physicians have reported the beneficial effects of biologic TNF-α inhibitors in the course of TEN; however, most of the literature is composed of case reports and case series describing a small number of patients. Therefore, the beneficial effects of anti–TNF-α therapy in TEN cannot be conclusively derived. Furthermore, cases using TNF-α inhibitors in combination with or after other systemic agents complicate the effects of TNF-α inhibitors themselves. Most of these case reports and case series describe the beneficial effects of TNF-α inhibitors in TEN; however, it is important to remember that cases in which these agents were ineffective are less likely to be published. The strongest evidence for TNF-α inhibitor use in the treatment TEN comes from the Paradisi et al14 case series, which showed a decrease in expected mortality with etanercept monotherapy in a relatively large cohort of patients. However, when evaluated prospectively by Paquet et al,24 there was no benefit seen by adding infliximab to NAC therapy and possibly an increased mortality in the group treated with both agents.
In the cases reviewed, a total of 32 patients were treated with infliximab or etanercept, and of these patients there were 4 deaths (12.5%).16,22,24 Three deaths were attributed to infection and 1 was attributed to disseminated intravascular coagulation. Furthermore, infection complicated the hospital course of 9 (28.1%) patients.13,15,22,24 The bacteria cultured from these patients included methicillin-resistant S aureus, P aeruginosa, E coli, Enterobacter aerogenes, and K pneumoniae. Patients who received TNF-α antagonists in combination with or after other systemic immunosuppressants appeared to have a higher incidence of infections. All patients treated with TNF-α antagonists in TEN should undergo careful evaluation and monitoring for infections due to the immunosuppressant effect of these drugs.
In our review, a total of 3 pediatric/adolescent patients received a TNF-α inhibitor for the treatment of TEN.13,17,21 Two patients received infliximab as a second-line medication after failure of IVIG to arrest progression of disease13,17 and one patient received infliximab as a second-line medication after dexamethasone.21 Each of these patients recovered without any reported infections or long-term complications.
Although excluded from this review, both infliximab and etanercept have been reported to show benefit in acute generalized exanthematous pustulosis/TEN overlap.26,27 Interestingly, in postmarketing surveillance, rare reports have implicated both infliximab and etanercept in causing both SJS and TEN.28 Also, there have been case reports of adalimumab causing SJS, but no cases of it causing TEN were identified.29,30
CONCLUSION
Rapid discontinuation of the culprit drug and aggressive supportive care remain the primary treatment of TEN. Tumor necrosis factor α inhibitors as monotherapy or as second-line agents show promise in the treatment of this complex disease state in both the adult and pediatric populations. The risks of these potent immunosuppressants must be weighed, and if administered, patients must be closely monitored for infections. Additional studies are needed to further characterize the role of TNF-α inhibition in the treatment of TEN.
- Schwartz R, McDonough P, Lee B. Toxic epidermal necrolysis: part I. introduction, history, classification, clinical features, systemic manifestations, etiology, and immunopathogenesis. J Am Acad Dermatol. 2013;69:173-186.
- Schwartz R, McDonough P, Lee B. Toxic epidermal necrolysis: part II. prognosis, sequelae, diagnosis, differential diagnosis, prevention, and treatment. J Am Acad Dermatol. 2013;69:187-203.
- Fernando S. The management of toxic epidermal necrolysis. Australas J Dermatol. 2012;55:165-171.
- Paquet P, Paquet F, Saleh W, et al. Immunoregulatory effector cells in drug-induced toxic epidermal necrolysis. Am J Dermatopathol. 2000;22:413-417.
- Nassif A, Moslehi H, Le Gouvello S, et al. Evaluation of the potential role of cytokines in toxic epidermal necrolysis. J Invest Dermatol. 2004;123:850-855.
- Viard-Leveugle I, Gaide O, Jankovic D, et al. TNF-α and INF-γ are potential inducers of Fas-mediated keratinocyte apoptosis thought activation of inducible nitric oxide synthase in toxic epidermal necrolysis. J Invest Dermatol. 2013;133:489-498.
- Paquet P, Pierard G. Soluble fractions of tumor necrosis factor-alpha, interleukin-6 and of their receptors in toxic epidermal necrolysis: a comparison with second-degree burns. Int J Mol Med. 1998;1:459-462.
- Correia O, Delgado L, Barbosa I, et al. Increased interleukin 10, tumor necrosis factor alpha, and interleukin 6 levels in blister fluid of toxic epidermal necrolysis. J Am Acad Dermatol. 2002;47:58-62.
- Redondo P, Rutz de Erenchun F, Iglesias M, et al. Toxic epidermal necrolysis. treatment with pentoxifylline. Br J Dermatol. 1994;130:688-689.
- Wolkenstein P, Latarjet J, Roujeau J, et al. Randomised comparison of thalidomide versus placebo in toxic epidermal necrolysis. Lancet. 1998;352:1586-1589.
- Fischer M, Fiedler E, Marsch W, et al. Antitumour necrosis factor-alpha antibodies (infliximab) in the treatment of a patient with toxic epidermal necrolysis. Br J Dermatol. 2002;146:707-708.
- Hunger R, Hunziker T, Buettiker U, et al. Rapid resolution of toxic epidermal necrolysis with anti-TNF-alpha treatment. J Allergy Clin Immunol. 2005;116:923-924.
- Zárate-Correa LC, Carrillo-Gómez DC, Ramírez-Escobar AF, et al. Toxic epidermal necrolysis successfully treated with infliximab. J Investig Allergol Clin Immunol. 2013;23:61-63.
- Paradisi A, Abeni D, Bergamo F, et al. Etanercept therapy for toxic epidermal necrolysis. J Am Acad Dermatol. 2014;71:278-283.
- Al-Shouli S, Bogusz M, Al Tufail M, et al. Toxic epidermal necrosis associated with high intake of sildenafil and its response to infliximab. Acta Derm Venereol. 2005;85:534-553.
- Famularo G, Di Dona B, Canzona F, et al. Etanercept for toxic epidermal necrolysis. Ann Pharmacother. 2007;41:1083-1084.
- Wojtkiewicz A, Wysocki M, Fortuna J, et al. Beneficial and rapid effect of infliximab on the course of toxic epidermal necrolysis. Acta Derm Venereol. 2008;88:420-421.
- Gubinelli E, Canzona F, Tonanzi T, et al. Toxic epidermal necrolysis successfully treated with etanercept. J Dermatol. 2009;36:150-153.
- Kreft B, Wohlrab J, Bramsiepe I, et al. Etoricoxib-induced toxic epidermal necrolysis: successful treatment with infliximab. J Dermatol. 2010;37:904-906.
- Worsnop F, Wee J, Moosa Y, et al. Reaction to biological drugs: infliximab for the treatment of toxic epidermal necrolysis subsequently triggering erosive lichen planus. Clin Exp Dermatol. 2012;37:879-881.
- Scott-Lang V, Tidman M, McKay D. Toxic epidermal necrolysis in a child successfully treated with infliximab. Pediatr Dermatol. 2014;31:532-534.
- Gaitanis G, Spyridonos P, Patmanidis K, et al. Treatment of toxic epidermal necrolysis with the combination of infliximab and high-dose intravenous immunoglobulins. Dermatology. 2012;224:134-139.
- Patmanidis K, Sidiras A, Dolianitis K, et al. Combination of infliximab and high-dose intravenous immunoglobulin for toxic epidermal necrolysis: successful treatment of an elderly patient. Case Rep Dermatol Med. 2012;2012:915314.
- Paquet P, Jennes S, Rousseua A, et al. Effect of N-acetylcysteine combined with infliximab on toxic epidermal necrolysis: a proof-of-concept study. Burns. 2014;1:1-6.
- Sanclemente G, De le Rouche C, Escobar C, et al. Pentoxifylline in toxic epidermal necrolysis and Stevens-Johnson syndrome. Int J Dermatol. 1998;38:878-879.
- Meiss F, Helmbold P, Meykadeh N, et al. Overlap of acute generalized exanthematous pustulosis and toxic epidermal necrolysis: response to antitumor necrosis factor-alpha antibody infliximab: report of three cases. J Eur Acad Dermatol Venereol. 2007;21:717-719.
- Sadighha A. Etanercept in the treatment of a patient with acute generalized exanthematous pustulosis/toxic epidermal necrolysis: definition of a new model based on translational research. Int J Dermatol. 2009;48:913-914.
- Borras-Blasco J, Navarro-Ruiz A, Borras C, et al. Adverse cutaneous reactions induced by TNF-α antagonist therapy. South Med J. 2009;102:1133-1140.
- Muna S, Lawrance I. Stevens-Johnson syndrome complicating adalimumab therapy in Crohn’s disease. World J Gastroenterol. 2009;15:4449-4452.
- Mounach A, Rezgi A, Nouijai A, et al. Stevens-Johnson syndrome complicating adalimumab therapy in rheumatoid arthritis disease. Rheumatol Int. 2013;33:1351-1353.
- Schwartz R, McDonough P, Lee B. Toxic epidermal necrolysis: part I. introduction, history, classification, clinical features, systemic manifestations, etiology, and immunopathogenesis. J Am Acad Dermatol. 2013;69:173-186.
- Schwartz R, McDonough P, Lee B. Toxic epidermal necrolysis: part II. prognosis, sequelae, diagnosis, differential diagnosis, prevention, and treatment. J Am Acad Dermatol. 2013;69:187-203.
- Fernando S. The management of toxic epidermal necrolysis. Australas J Dermatol. 2012;55:165-171.
- Paquet P, Paquet F, Saleh W, et al. Immunoregulatory effector cells in drug-induced toxic epidermal necrolysis. Am J Dermatopathol. 2000;22:413-417.
- Nassif A, Moslehi H, Le Gouvello S, et al. Evaluation of the potential role of cytokines in toxic epidermal necrolysis. J Invest Dermatol. 2004;123:850-855.
- Viard-Leveugle I, Gaide O, Jankovic D, et al. TNF-α and INF-γ are potential inducers of Fas-mediated keratinocyte apoptosis thought activation of inducible nitric oxide synthase in toxic epidermal necrolysis. J Invest Dermatol. 2013;133:489-498.
- Paquet P, Pierard G. Soluble fractions of tumor necrosis factor-alpha, interleukin-6 and of their receptors in toxic epidermal necrolysis: a comparison with second-degree burns. Int J Mol Med. 1998;1:459-462.
- Correia O, Delgado L, Barbosa I, et al. Increased interleukin 10, tumor necrosis factor alpha, and interleukin 6 levels in blister fluid of toxic epidermal necrolysis. J Am Acad Dermatol. 2002;47:58-62.
- Redondo P, Rutz de Erenchun F, Iglesias M, et al. Toxic epidermal necrolysis. treatment with pentoxifylline. Br J Dermatol. 1994;130:688-689.
- Wolkenstein P, Latarjet J, Roujeau J, et al. Randomised comparison of thalidomide versus placebo in toxic epidermal necrolysis. Lancet. 1998;352:1586-1589.
- Fischer M, Fiedler E, Marsch W, et al. Antitumour necrosis factor-alpha antibodies (infliximab) in the treatment of a patient with toxic epidermal necrolysis. Br J Dermatol. 2002;146:707-708.
- Hunger R, Hunziker T, Buettiker U, et al. Rapid resolution of toxic epidermal necrolysis with anti-TNF-alpha treatment. J Allergy Clin Immunol. 2005;116:923-924.
- Zárate-Correa LC, Carrillo-Gómez DC, Ramírez-Escobar AF, et al. Toxic epidermal necrolysis successfully treated with infliximab. J Investig Allergol Clin Immunol. 2013;23:61-63.
- Paradisi A, Abeni D, Bergamo F, et al. Etanercept therapy for toxic epidermal necrolysis. J Am Acad Dermatol. 2014;71:278-283.
- Al-Shouli S, Bogusz M, Al Tufail M, et al. Toxic epidermal necrosis associated with high intake of sildenafil and its response to infliximab. Acta Derm Venereol. 2005;85:534-553.
- Famularo G, Di Dona B, Canzona F, et al. Etanercept for toxic epidermal necrolysis. Ann Pharmacother. 2007;41:1083-1084.
- Wojtkiewicz A, Wysocki M, Fortuna J, et al. Beneficial and rapid effect of infliximab on the course of toxic epidermal necrolysis. Acta Derm Venereol. 2008;88:420-421.
- Gubinelli E, Canzona F, Tonanzi T, et al. Toxic epidermal necrolysis successfully treated with etanercept. J Dermatol. 2009;36:150-153.
- Kreft B, Wohlrab J, Bramsiepe I, et al. Etoricoxib-induced toxic epidermal necrolysis: successful treatment with infliximab. J Dermatol. 2010;37:904-906.
- Worsnop F, Wee J, Moosa Y, et al. Reaction to biological drugs: infliximab for the treatment of toxic epidermal necrolysis subsequently triggering erosive lichen planus. Clin Exp Dermatol. 2012;37:879-881.
- Scott-Lang V, Tidman M, McKay D. Toxic epidermal necrolysis in a child successfully treated with infliximab. Pediatr Dermatol. 2014;31:532-534.
- Gaitanis G, Spyridonos P, Patmanidis K, et al. Treatment of toxic epidermal necrolysis with the combination of infliximab and high-dose intravenous immunoglobulins. Dermatology. 2012;224:134-139.
- Patmanidis K, Sidiras A, Dolianitis K, et al. Combination of infliximab and high-dose intravenous immunoglobulin for toxic epidermal necrolysis: successful treatment of an elderly patient. Case Rep Dermatol Med. 2012;2012:915314.
- Paquet P, Jennes S, Rousseua A, et al. Effect of N-acetylcysteine combined with infliximab on toxic epidermal necrolysis: a proof-of-concept study. Burns. 2014;1:1-6.
- Sanclemente G, De le Rouche C, Escobar C, et al. Pentoxifylline in toxic epidermal necrolysis and Stevens-Johnson syndrome. Int J Dermatol. 1998;38:878-879.
- Meiss F, Helmbold P, Meykadeh N, et al. Overlap of acute generalized exanthematous pustulosis and toxic epidermal necrolysis: response to antitumor necrosis factor-alpha antibody infliximab: report of three cases. J Eur Acad Dermatol Venereol. 2007;21:717-719.
- Sadighha A. Etanercept in the treatment of a patient with acute generalized exanthematous pustulosis/toxic epidermal necrolysis: definition of a new model based on translational research. Int J Dermatol. 2009;48:913-914.
- Borras-Blasco J, Navarro-Ruiz A, Borras C, et al. Adverse cutaneous reactions induced by TNF-α antagonist therapy. South Med J. 2009;102:1133-1140.
- Muna S, Lawrance I. Stevens-Johnson syndrome complicating adalimumab therapy in Crohn’s disease. World J Gastroenterol. 2009;15:4449-4452.
- Mounach A, Rezgi A, Nouijai A, et al. Stevens-Johnson syndrome complicating adalimumab therapy in rheumatoid arthritis disease. Rheumatol Int. 2013;33:1351-1353.
Practice Points
- Controversy remains over the most effective adjunctive therapy for toxic epidermal necrolysis (TEN), as none have consistently displayed superiority over rapid discontinuation of the culprit drug and aggressive supportive care alone.
- Since tumor necrosis factor α (TNF-α) was implicated in the pathogenesis of TEN, TNF-α inhibition has been attempted in treatment of the disease. These medications have shown positive outcomes.
- The risks of these potent immunosuppressants must be weighed, and if administered, patients must be closely monitored for infections.