User login
Cosmetic Corner: Dermatologists Weigh in on OTC Adult Acne Products
To improve patient care and outcomes, leading dermatologists offered their recommendations on adult acne products. Consideration must be given to:
- Bioclear Face Lotion and Face Cream
Jan Marini Skin Research, Inc
“These products contain a powerful combination of glycolic, salicylic, and azelaic acids to smooth and brighten acne-prone skin.”—Larisa Ravitskiy, MD, Gahanna, Ohio
- Neutrogena Clear Pore Cleanser/Mask
Johnson & Johnson Consumer Inc
“This is a good daily product for acne-prone skin. It is formulated with benzoyl peroxide and can be used as a daily wash or mask.”—Anthony M. Rossi, MD, New York, New York
- Offects Sulfur Masque Acne Treatment
ZO Skin Health Inc
“This nonirritating mask reduces inflammation and oiliness and is safe to use in pregnancy.”—Larisa Ravitskiy, MD, Gahanna, Ohio
- PanOxyl Acne Foaming Wash and Acne Creamy Wash
Stiefel, a GSK company
Recommended by Gary Goldenberg, MD, New York, New York
Cutis invites readers to send us their recommendations. Athlete’s foot treatments, as well as products for dry cuticles, hyperhidrosis, and sensitive skin will be featured in upcoming editions of Cosmetic Corner. Please e-mail your recommendation(s) to the Editorial Office.
Disclaimer: Opinions expressed herein do not necessarily reflect those of Cutis or Frontline Medical Communications Inc. and shall not be used for product endorsement purposes. Any reference made to a specific commercial product does not indicate or imply that Cutis or Frontline Medical Communications Inc. endorses, recommends, or favors the product mentioned. No guarantee is given to the effects of recommended products.
[polldaddy:9711250]
To improve patient care and outcomes, leading dermatologists offered their recommendations on adult acne products. Consideration must be given to:
- Bioclear Face Lotion and Face Cream
Jan Marini Skin Research, Inc
“These products contain a powerful combination of glycolic, salicylic, and azelaic acids to smooth and brighten acne-prone skin.”—Larisa Ravitskiy, MD, Gahanna, Ohio
- Neutrogena Clear Pore Cleanser/Mask
Johnson & Johnson Consumer Inc
“This is a good daily product for acne-prone skin. It is formulated with benzoyl peroxide and can be used as a daily wash or mask.”—Anthony M. Rossi, MD, New York, New York
- Offects Sulfur Masque Acne Treatment
ZO Skin Health Inc
“This nonirritating mask reduces inflammation and oiliness and is safe to use in pregnancy.”—Larisa Ravitskiy, MD, Gahanna, Ohio
- PanOxyl Acne Foaming Wash and Acne Creamy Wash
Stiefel, a GSK company
Recommended by Gary Goldenberg, MD, New York, New York
Cutis invites readers to send us their recommendations. Athlete’s foot treatments, as well as products for dry cuticles, hyperhidrosis, and sensitive skin will be featured in upcoming editions of Cosmetic Corner. Please e-mail your recommendation(s) to the Editorial Office.
Disclaimer: Opinions expressed herein do not necessarily reflect those of Cutis or Frontline Medical Communications Inc. and shall not be used for product endorsement purposes. Any reference made to a specific commercial product does not indicate or imply that Cutis or Frontline Medical Communications Inc. endorses, recommends, or favors the product mentioned. No guarantee is given to the effects of recommended products.
[polldaddy:9711250]
To improve patient care and outcomes, leading dermatologists offered their recommendations on adult acne products. Consideration must be given to:
- Bioclear Face Lotion and Face Cream
Jan Marini Skin Research, Inc
“These products contain a powerful combination of glycolic, salicylic, and azelaic acids to smooth and brighten acne-prone skin.”—Larisa Ravitskiy, MD, Gahanna, Ohio
- Neutrogena Clear Pore Cleanser/Mask
Johnson & Johnson Consumer Inc
“This is a good daily product for acne-prone skin. It is formulated with benzoyl peroxide and can be used as a daily wash or mask.”—Anthony M. Rossi, MD, New York, New York
- Offects Sulfur Masque Acne Treatment
ZO Skin Health Inc
“This nonirritating mask reduces inflammation and oiliness and is safe to use in pregnancy.”—Larisa Ravitskiy, MD, Gahanna, Ohio
- PanOxyl Acne Foaming Wash and Acne Creamy Wash
Stiefel, a GSK company
Recommended by Gary Goldenberg, MD, New York, New York
Cutis invites readers to send us their recommendations. Athlete’s foot treatments, as well as products for dry cuticles, hyperhidrosis, and sensitive skin will be featured in upcoming editions of Cosmetic Corner. Please e-mail your recommendation(s) to the Editorial Office.
Disclaimer: Opinions expressed herein do not necessarily reflect those of Cutis or Frontline Medical Communications Inc. and shall not be used for product endorsement purposes. Any reference made to a specific commercial product does not indicate or imply that Cutis or Frontline Medical Communications Inc. endorses, recommends, or favors the product mentioned. No guarantee is given to the effects of recommended products.
[polldaddy:9711250]
Active surveillance an option for patients with mRCC
ORLANDO – Active surveillance prior to initiating targeted therapy could be an option for some patients with metastatic renal cell carcinoma (mRCC), according to new findings.
“Active surveillance does not affect the efficacy of subsequent therapies,” said lead study author Davide Bimbatti, MD, of the Azienda Ospedaliera Universitaria Integrata, University of Verona (Italy). “In selected patients, active surveillance allows us to delay the start of systemic treatment, and patients in active surveillance rarely have a worsening of prognostic class,” he said in a press briefing held at the 2017 Genitourinary Cancers Symposium sponsored by the American Society of Clinical Oncology, ASTRO, and the Society of Urologic Oncology.
Targeted therapies can improve survival in mRCC but treatment is not curative and associated toxicities can interfere with quality of life and force patients to discontinue treatment. Active surveillance delays the use of systemic therapy and associated toxicities, and is a feasible strategy for patients with indolent disease.
However, the effect of active surveillance on tumor burden and prognosis have not been investigated.
To address this issue, Dr. Bimbatti and his colleagues conducted a retrospective study and evaluated the effect of active surveillance on overall survival and postsurveillance overall survival, progression-free survival, and tumor burden (a measure of the number of sites and tumor size).
The study cohort included 48 patients with mRCC who underwent active surveillance during 2007-2016 and changes in the International mRCC Database Consortium (IMDC) prognostic class and tumor burden were analyzed for associations with these endpoints.
At baseline, 69% of patients had a favorable prognostic IMDC class, 25% were at an intermediate class, and 6% had a poor class designation. The main sites of metastases were lung (56%), lymph nodes (25%), pancreas (14%), adrenal gland (8%), CNS (8%), and bone (6%).
At a median follow-up of 37.3 months, 79.2% of patients were still alive and, at a median surveillance duration of 16.7 months, 71% of patients had begun a targeted therapy. There were a total of 17 deaths (33%).
Median progression-free survival was 16.6 months and median postsurveillance overall survival was 39.1 months.
During active surveillance, only four patients transitioned from good to intermediate IMDC prognostic class. IMDC classes overall maintained their prognostic value during active surveillance, Dr. Bimbatti said. IMDC class was also the only factor that was associated with time on surveillance.
At baseline, tumor burden was in one site for 65% of patients, two sites in 31%, and three or more sites for 4%, but during active surveillance, changes occurred in one site in 35% of patients, two in 48%, and more than two in 17%.
A change in tumor burden (greater than or equal to 2.2 times the original burden), however, was related to poorer postsurveillance survival (hazard ratio,1.23; P less than .01), but not with overall survival (HR, 1.0; P less than .05).
Conversely, any increase in metastatic sites was associated with both significantly worse postsurveillance and overall survival (HR = 2.6, P = 0.04; and HR = 3.3, P less than 0.01).
Commenting on the study, Primo N. Lara Jr., MD, of the University of California, Davis, Comprehensive Cancer Center explained that active surveillance “remains a reasonable option for highly selected patients.”
“[The investigators] provide reassuring data that active surveillance does not alter the IMDC risk grouping,” he said, and that the results of this study overall, were similar to those of prior research.
There were some limitations to the study, in that it was retrospective and conducted at a single institution. “It also failed to account for psychosocial issues such as anxiety during the active surveillance phase,” he said.
Critical questions also remain, Dr. Lara added, such as what are the validated selection criteria for patients entering active surveillance or what is the threshold of tumor burden to warrant active surveillance discontinuation.
The funding source was not disclosed. Dr. Bimbatti and his coauthors have no disclosures. Dr. Lara reports financial ties to multiple pharmaceutical companies.
ORLANDO – Active surveillance prior to initiating targeted therapy could be an option for some patients with metastatic renal cell carcinoma (mRCC), according to new findings.
“Active surveillance does not affect the efficacy of subsequent therapies,” said lead study author Davide Bimbatti, MD, of the Azienda Ospedaliera Universitaria Integrata, University of Verona (Italy). “In selected patients, active surveillance allows us to delay the start of systemic treatment, and patients in active surveillance rarely have a worsening of prognostic class,” he said in a press briefing held at the 2017 Genitourinary Cancers Symposium sponsored by the American Society of Clinical Oncology, ASTRO, and the Society of Urologic Oncology.
Targeted therapies can improve survival in mRCC but treatment is not curative and associated toxicities can interfere with quality of life and force patients to discontinue treatment. Active surveillance delays the use of systemic therapy and associated toxicities, and is a feasible strategy for patients with indolent disease.
However, the effect of active surveillance on tumor burden and prognosis have not been investigated.
To address this issue, Dr. Bimbatti and his colleagues conducted a retrospective study and evaluated the effect of active surveillance on overall survival and postsurveillance overall survival, progression-free survival, and tumor burden (a measure of the number of sites and tumor size).
The study cohort included 48 patients with mRCC who underwent active surveillance during 2007-2016 and changes in the International mRCC Database Consortium (IMDC) prognostic class and tumor burden were analyzed for associations with these endpoints.
At baseline, 69% of patients had a favorable prognostic IMDC class, 25% were at an intermediate class, and 6% had a poor class designation. The main sites of metastases were lung (56%), lymph nodes (25%), pancreas (14%), adrenal gland (8%), CNS (8%), and bone (6%).
At a median follow-up of 37.3 months, 79.2% of patients were still alive and, at a median surveillance duration of 16.7 months, 71% of patients had begun a targeted therapy. There were a total of 17 deaths (33%).
Median progression-free survival was 16.6 months and median postsurveillance overall survival was 39.1 months.
During active surveillance, only four patients transitioned from good to intermediate IMDC prognostic class. IMDC classes overall maintained their prognostic value during active surveillance, Dr. Bimbatti said. IMDC class was also the only factor that was associated with time on surveillance.
At baseline, tumor burden was in one site for 65% of patients, two sites in 31%, and three or more sites for 4%, but during active surveillance, changes occurred in one site in 35% of patients, two in 48%, and more than two in 17%.
A change in tumor burden (greater than or equal to 2.2 times the original burden), however, was related to poorer postsurveillance survival (hazard ratio,1.23; P less than .01), but not with overall survival (HR, 1.0; P less than .05).
Conversely, any increase in metastatic sites was associated with both significantly worse postsurveillance and overall survival (HR = 2.6, P = 0.04; and HR = 3.3, P less than 0.01).
Commenting on the study, Primo N. Lara Jr., MD, of the University of California, Davis, Comprehensive Cancer Center explained that active surveillance “remains a reasonable option for highly selected patients.”
“[The investigators] provide reassuring data that active surveillance does not alter the IMDC risk grouping,” he said, and that the results of this study overall, were similar to those of prior research.
There were some limitations to the study, in that it was retrospective and conducted at a single institution. “It also failed to account for psychosocial issues such as anxiety during the active surveillance phase,” he said.
Critical questions also remain, Dr. Lara added, such as what are the validated selection criteria for patients entering active surveillance or what is the threshold of tumor burden to warrant active surveillance discontinuation.
The funding source was not disclosed. Dr. Bimbatti and his coauthors have no disclosures. Dr. Lara reports financial ties to multiple pharmaceutical companies.
ORLANDO – Active surveillance prior to initiating targeted therapy could be an option for some patients with metastatic renal cell carcinoma (mRCC), according to new findings.
“Active surveillance does not affect the efficacy of subsequent therapies,” said lead study author Davide Bimbatti, MD, of the Azienda Ospedaliera Universitaria Integrata, University of Verona (Italy). “In selected patients, active surveillance allows us to delay the start of systemic treatment, and patients in active surveillance rarely have a worsening of prognostic class,” he said in a press briefing held at the 2017 Genitourinary Cancers Symposium sponsored by the American Society of Clinical Oncology, ASTRO, and the Society of Urologic Oncology.
Targeted therapies can improve survival in mRCC but treatment is not curative and associated toxicities can interfere with quality of life and force patients to discontinue treatment. Active surveillance delays the use of systemic therapy and associated toxicities, and is a feasible strategy for patients with indolent disease.
However, the effect of active surveillance on tumor burden and prognosis have not been investigated.
To address this issue, Dr. Bimbatti and his colleagues conducted a retrospective study and evaluated the effect of active surveillance on overall survival and postsurveillance overall survival, progression-free survival, and tumor burden (a measure of the number of sites and tumor size).
The study cohort included 48 patients with mRCC who underwent active surveillance during 2007-2016 and changes in the International mRCC Database Consortium (IMDC) prognostic class and tumor burden were analyzed for associations with these endpoints.
At baseline, 69% of patients had a favorable prognostic IMDC class, 25% were at an intermediate class, and 6% had a poor class designation. The main sites of metastases were lung (56%), lymph nodes (25%), pancreas (14%), adrenal gland (8%), CNS (8%), and bone (6%).
At a median follow-up of 37.3 months, 79.2% of patients were still alive and, at a median surveillance duration of 16.7 months, 71% of patients had begun a targeted therapy. There were a total of 17 deaths (33%).
Median progression-free survival was 16.6 months and median postsurveillance overall survival was 39.1 months.
During active surveillance, only four patients transitioned from good to intermediate IMDC prognostic class. IMDC classes overall maintained their prognostic value during active surveillance, Dr. Bimbatti said. IMDC class was also the only factor that was associated with time on surveillance.
At baseline, tumor burden was in one site for 65% of patients, two sites in 31%, and three or more sites for 4%, but during active surveillance, changes occurred in one site in 35% of patients, two in 48%, and more than two in 17%.
A change in tumor burden (greater than or equal to 2.2 times the original burden), however, was related to poorer postsurveillance survival (hazard ratio,1.23; P less than .01), but not with overall survival (HR, 1.0; P less than .05).
Conversely, any increase in metastatic sites was associated with both significantly worse postsurveillance and overall survival (HR = 2.6, P = 0.04; and HR = 3.3, P less than 0.01).
Commenting on the study, Primo N. Lara Jr., MD, of the University of California, Davis, Comprehensive Cancer Center explained that active surveillance “remains a reasonable option for highly selected patients.”
“[The investigators] provide reassuring data that active surveillance does not alter the IMDC risk grouping,” he said, and that the results of this study overall, were similar to those of prior research.
There were some limitations to the study, in that it was retrospective and conducted at a single institution. “It also failed to account for psychosocial issues such as anxiety during the active surveillance phase,” he said.
Critical questions also remain, Dr. Lara added, such as what are the validated selection criteria for patients entering active surveillance or what is the threshold of tumor burden to warrant active surveillance discontinuation.
The funding source was not disclosed. Dr. Bimbatti and his coauthors have no disclosures. Dr. Lara reports financial ties to multiple pharmaceutical companies.
AT THE GENITOURINARY CANCERS SYMPOSIUM
Key clinical point: Active surveillance may be an option for some patients with metastatic renal cell carcinoma.
Major finding: A change in tumor burden (greater than or equal to 2.2 times the original burden), however, was related to poorer postsurveillance survival (HR,1.23; P less than .01) but not with overall survival (HR, 1.0; P less than.05).
Data source: Retrospective single center study that evaluated active surveillance in 48 patients with metastatic renal cell carcinoma.
Disclosures: The funding source was not disclosed. Dr. Bimbatti and his coauthors have no disclosures. Dr. Lara reports financial ties to multiple pharmaceutical companies.
Vascular surgeons underutilize palliative care planning
Investment in advanced palliative care planning has the potential to improve the quality of care for vascular surgery patients, according to investigators from Oregon Health and Science University, Portland.
Dale G. Wilson, MD, and his colleagues performed a retrospective review of electronic medical records for 111 patients, who died while on the vascular surgery service at the OHSU Hospital during 2005-2014.
Almost three-quarters (73%) of patients were transitioned to palliative care; of those, 14% presented with an advanced directive, and 28% received a palliative care consultation (JAMA Surg. 2017;152[2]:183-90. doi: 10.1001/jamasurg.2016.3970).
While palliative care services are increasing in hospitals, accounting for 4% of annual hospital admissions in 2012 according to the study, they are not implemented consistently. “Many teams from various specialties care for patients at end of life; however, we still do not know what prompts end-of-life discussions,” Dr. Wilson said. “There is still no consensus on when to involve palliative services in the care of critically ill patients.”
While the decision to advise a consultation is “variable and physician dependent,” the type of treatment required may help identify when consultations are appropriate.
Of the 14 patients who did not choose comfort care, 11 (79%) required CPR. Additionally, all had to be taken to the operating room and required mechanical ventilation.
Of 81 patients who chose palliative care, 31 did so despite potential medical options. These patients were older – average age, 77 years, as compared with 68 years for patients who did not choose comfort care – with 8 of the 31 (26%) presenting an advanced directive, compared with only 7 of 83 patients (8%) for those who did not receive palliative care.
Dr. Wilson and his colleagues found that patients who chose palliative care were more likely to have received a palliative care consultation, as well: 10 of 31 patients who chose comfort care received a consultation, as opposed to 1 of 83 who chose comfort care but did not receive a consultation.
The nature of the vascular surgery service calls for early efforts to gather information regarding patients’ views on end-of-life care, Dr. Wilson said, noting that 73% of patients studied were admitted emergently and 87% underwent surgery, leaving little time for patients to express their wishes.
“Because the events associated with withdrawal of care are often not anticipated, we argue that all vascular surgical patients should have an advance directive, and perhaps, those at particular high risk should have a preoperative palliative care consultation,” Dr. Wilson wrote.
Limitations to the study included the data abstraction, which was performed by a single unblinded physician. Researchers also gathered patients’ reasons for transitioning to comfort care retrospectively.
The low rate of palliative care consultations found in this study mirrors my own experience, as does the feeling of urgency to shed more light on the issue. The biggest hurdle surgeons face when it comes to palliative care consultations is that, in their minds, seeking these meetings is associated with immediate death care. Many surgeons are shy about bringing palliative care specialists on board because approaching families can be daunting.
Family members who do not know enough about comfort care can be upset by the idea. Addressing this misunderstanding is crucial. Consultations are not just conversations about hospice care but can be emotional and spiritual experiences that prepare both the family and the patient for alternative options when surgical intervention cannot guarantee a good quality of life. I would encourage surgeons to be more proactive and less defensive about comfort care . Luckily, understanding the importance of this issue among professionals is growing.
When I approach these situations, it’s important for me to have a full understanding of what families and patients usually expect. Decisions should not be based on how bad things are now but on the future. What was the patient’s last year like? What is the best-case scenario for moving forward on a proposed intervention? What will the patient’s quality of life be? Answering these questions helps the patient understand his or her situation, without diminishing a surgeon’s ability. If you are honest, the family will usually come to the conclusion that they do not want to subject the patient to ultimately unnecessary treatment.
Palliative care services help patients and their families deal with pain beyond the physical symptoms. Dealing with pain, depression, or delirium is only a part of comfort care – coping with a sense of hopelessness, family disruption, or feelings of guilt also can be a part and, significantly, a part that surgeons are not trained to diagnose or treat.
With more than 70 surgeons certified in hospice care and a growing number of fellowships in palliative care, I am extremely optimistic in the progress we have made and will continue to make.
Geoffrey Dunn, MD, FACS, is the medical director of the Palliative Care Consultation Service at UPMC Hamot Medical Center, Erie, Penn. He currently is Community Editor for the Pain and Palliative Care Community for the ACS’s web portal.
The low rate of palliative care consultations found in this study mirrors my own experience, as does the feeling of urgency to shed more light on the issue. The biggest hurdle surgeons face when it comes to palliative care consultations is that, in their minds, seeking these meetings is associated with immediate death care. Many surgeons are shy about bringing palliative care specialists on board because approaching families can be daunting.
Family members who do not know enough about comfort care can be upset by the idea. Addressing this misunderstanding is crucial. Consultations are not just conversations about hospice care but can be emotional and spiritual experiences that prepare both the family and the patient for alternative options when surgical intervention cannot guarantee a good quality of life. I would encourage surgeons to be more proactive and less defensive about comfort care . Luckily, understanding the importance of this issue among professionals is growing.
When I approach these situations, it’s important for me to have a full understanding of what families and patients usually expect. Decisions should not be based on how bad things are now but on the future. What was the patient’s last year like? What is the best-case scenario for moving forward on a proposed intervention? What will the patient’s quality of life be? Answering these questions helps the patient understand his or her situation, without diminishing a surgeon’s ability. If you are honest, the family will usually come to the conclusion that they do not want to subject the patient to ultimately unnecessary treatment.
Palliative care services help patients and their families deal with pain beyond the physical symptoms. Dealing with pain, depression, or delirium is only a part of comfort care – coping with a sense of hopelessness, family disruption, or feelings of guilt also can be a part and, significantly, a part that surgeons are not trained to diagnose or treat.
With more than 70 surgeons certified in hospice care and a growing number of fellowships in palliative care, I am extremely optimistic in the progress we have made and will continue to make.
Geoffrey Dunn, MD, FACS, is the medical director of the Palliative Care Consultation Service at UPMC Hamot Medical Center, Erie, Penn. He currently is Community Editor for the Pain and Palliative Care Community for the ACS’s web portal.
The low rate of palliative care consultations found in this study mirrors my own experience, as does the feeling of urgency to shed more light on the issue. The biggest hurdle surgeons face when it comes to palliative care consultations is that, in their minds, seeking these meetings is associated with immediate death care. Many surgeons are shy about bringing palliative care specialists on board because approaching families can be daunting.
Family members who do not know enough about comfort care can be upset by the idea. Addressing this misunderstanding is crucial. Consultations are not just conversations about hospice care but can be emotional and spiritual experiences that prepare both the family and the patient for alternative options when surgical intervention cannot guarantee a good quality of life. I would encourage surgeons to be more proactive and less defensive about comfort care . Luckily, understanding the importance of this issue among professionals is growing.
When I approach these situations, it’s important for me to have a full understanding of what families and patients usually expect. Decisions should not be based on how bad things are now but on the future. What was the patient’s last year like? What is the best-case scenario for moving forward on a proposed intervention? What will the patient’s quality of life be? Answering these questions helps the patient understand his or her situation, without diminishing a surgeon’s ability. If you are honest, the family will usually come to the conclusion that they do not want to subject the patient to ultimately unnecessary treatment.
Palliative care services help patients and their families deal with pain beyond the physical symptoms. Dealing with pain, depression, or delirium is only a part of comfort care – coping with a sense of hopelessness, family disruption, or feelings of guilt also can be a part and, significantly, a part that surgeons are not trained to diagnose or treat.
With more than 70 surgeons certified in hospice care and a growing number of fellowships in palliative care, I am extremely optimistic in the progress we have made and will continue to make.
Geoffrey Dunn, MD, FACS, is the medical director of the Palliative Care Consultation Service at UPMC Hamot Medical Center, Erie, Penn. He currently is Community Editor for the Pain and Palliative Care Community for the ACS’s web portal.
Investment in advanced palliative care planning has the potential to improve the quality of care for vascular surgery patients, according to investigators from Oregon Health and Science University, Portland.
Dale G. Wilson, MD, and his colleagues performed a retrospective review of electronic medical records for 111 patients, who died while on the vascular surgery service at the OHSU Hospital during 2005-2014.
Almost three-quarters (73%) of patients were transitioned to palliative care; of those, 14% presented with an advanced directive, and 28% received a palliative care consultation (JAMA Surg. 2017;152[2]:183-90. doi: 10.1001/jamasurg.2016.3970).
While palliative care services are increasing in hospitals, accounting for 4% of annual hospital admissions in 2012 according to the study, they are not implemented consistently. “Many teams from various specialties care for patients at end of life; however, we still do not know what prompts end-of-life discussions,” Dr. Wilson said. “There is still no consensus on when to involve palliative services in the care of critically ill patients.”
While the decision to advise a consultation is “variable and physician dependent,” the type of treatment required may help identify when consultations are appropriate.
Of the 14 patients who did not choose comfort care, 11 (79%) required CPR. Additionally, all had to be taken to the operating room and required mechanical ventilation.
Of 81 patients who chose palliative care, 31 did so despite potential medical options. These patients were older – average age, 77 years, as compared with 68 years for patients who did not choose comfort care – with 8 of the 31 (26%) presenting an advanced directive, compared with only 7 of 83 patients (8%) for those who did not receive palliative care.
Dr. Wilson and his colleagues found that patients who chose palliative care were more likely to have received a palliative care consultation, as well: 10 of 31 patients who chose comfort care received a consultation, as opposed to 1 of 83 who chose comfort care but did not receive a consultation.
The nature of the vascular surgery service calls for early efforts to gather information regarding patients’ views on end-of-life care, Dr. Wilson said, noting that 73% of patients studied were admitted emergently and 87% underwent surgery, leaving little time for patients to express their wishes.
“Because the events associated with withdrawal of care are often not anticipated, we argue that all vascular surgical patients should have an advance directive, and perhaps, those at particular high risk should have a preoperative palliative care consultation,” Dr. Wilson wrote.
Limitations to the study included the data abstraction, which was performed by a single unblinded physician. Researchers also gathered patients’ reasons for transitioning to comfort care retrospectively.
Investment in advanced palliative care planning has the potential to improve the quality of care for vascular surgery patients, according to investigators from Oregon Health and Science University, Portland.
Dale G. Wilson, MD, and his colleagues performed a retrospective review of electronic medical records for 111 patients, who died while on the vascular surgery service at the OHSU Hospital during 2005-2014.
Almost three-quarters (73%) of patients were transitioned to palliative care; of those, 14% presented with an advanced directive, and 28% received a palliative care consultation (JAMA Surg. 2017;152[2]:183-90. doi: 10.1001/jamasurg.2016.3970).
While palliative care services are increasing in hospitals, accounting for 4% of annual hospital admissions in 2012 according to the study, they are not implemented consistently. “Many teams from various specialties care for patients at end of life; however, we still do not know what prompts end-of-life discussions,” Dr. Wilson said. “There is still no consensus on when to involve palliative services in the care of critically ill patients.”
While the decision to advise a consultation is “variable and physician dependent,” the type of treatment required may help identify when consultations are appropriate.
Of the 14 patients who did not choose comfort care, 11 (79%) required CPR. Additionally, all had to be taken to the operating room and required mechanical ventilation.
Of 81 patients who chose palliative care, 31 did so despite potential medical options. These patients were older – average age, 77 years, as compared with 68 years for patients who did not choose comfort care – with 8 of the 31 (26%) presenting an advanced directive, compared with only 7 of 83 patients (8%) for those who did not receive palliative care.
Dr. Wilson and his colleagues found that patients who chose palliative care were more likely to have received a palliative care consultation, as well: 10 of 31 patients who chose comfort care received a consultation, as opposed to 1 of 83 who chose comfort care but did not receive a consultation.
The nature of the vascular surgery service calls for early efforts to gather information regarding patients’ views on end-of-life care, Dr. Wilson said, noting that 73% of patients studied were admitted emergently and 87% underwent surgery, leaving little time for patients to express their wishes.
“Because the events associated with withdrawal of care are often not anticipated, we argue that all vascular surgical patients should have an advance directive, and perhaps, those at particular high risk should have a preoperative palliative care consultation,” Dr. Wilson wrote.
Limitations to the study included the data abstraction, which was performed by a single unblinded physician. Researchers also gathered patients’ reasons for transitioning to comfort care retrospectively.
FROM JAMA SURGERY
Key clinical point:
Major finding: Of the 111 patients studied, 81 died on palliative care, but only 15 presented an advanced directive.
Data source: A retrospective cohort study of the records of patients aged 18-99 years who died in the vascular surgery service at Oregon Health and Science University Hospital from 2005-2014.
Disclosures: The authors reported no financial disclosures.
How and when umbilical cord gas analysis can justify your obstetric management
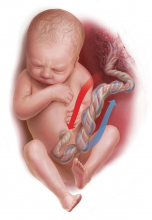
Umbilical cord blood (cord) gas values can aid both in understanding the cause of an infant’s acidosis and in providing reassurance that acute acidosis or asphyxia is not responsible for a compromised infant with a low Apgar score. Together with other clinical measurements (including fetal heart rate [FHR] tracings, Apgar scores, newborn nucleated red cell counts, and neonatal imaging), cord gas analysis can be remarkably helpful in determining the cause for a depressed newborn. It can help us determine, for example, if infant compromise was a result of an asphyxial event, and we often can differentiate whether the event was acute, prolonged, or occurred prior to presentation in labor. We further can use cord gas values to assess whether a decision for operative intervention for nonreassuring fetal well-being was appropriate (see “Brain injury at birth: Cord gas values presented as evidence at trial”). In addition, cord gas analysis can complement methods for determining fetal acidosis changes during labor, enabling improved assessment of FHR tracings.1−3
At 40 weeks' gestation, a woman presented to the hospital because of decreased fetal movement. On arrival, an external fetal heart-rate (FHR) monitor showed nonreassuring tracings, evidenced by absent to minimal variability and subtle decelerations occurring at 10- to 15-minute intervals. The on-call ObGyn requested induction of labor with oxytocin, and a low-dose infusion (1 mU/min) was initiated. An internal FHR monitor was then placed and late decelerations were observed with the first 2 induced contractions. The oxytocin infusion was discontinued and the ObGyn performed an emergency cesarean delivery. The infant's Apgar scores were 1, 2, and 2 at 1, 5, and 10 minutes, respectively. Cord samples were obtained and values from the umbilical artery were as follows: pH, 6.86; Pco2, 55 mm Hg; Po2, 6 mm Hg; and BDECF, 21.1 mmol/L. Values from the umbilical vein were: pH, 6.94; Pco2, 45 mm Hg; Po2, 17 mm Hg; and BDECF, 20.0 mmol/L. The infant was later diagnosed with a hypoxic brain injury resulting in cerebral palsy. At trial years later, the boy had cognitive and physical limitations and required 24-hour care.
The parents claimed that the ObGyn should have performed a cesarean delivery earlier when the external FHR monitor showed nonreassuring tracings.
The hospital and physician claimed that, while tracings were consistently nonreassuring, they were stable. They maintained that the child's brain damage was not due to a delivery delay, as the severe level of acidosis in both the umbilical artery and vein could not be a result of the few heart rate decelerations during the 2-hour period of monitoring prior to delivery. They argued that the clinical picture indicated a pre-hospital hypoxic event associated with decreased fetal movement.
A defense verdict was returned.
Case assessment
Cord gas results, together with other measures (eg, infant nucleated red blood cells, brain imaging) can aid the ObGyn in medicolegal cases. However, they are not always protective of adverse judgment.
I recommend checking umbilical cord blood gas values on all operative vaginal deliveries, cesarean deliveries for fetal concern, abnormal FHR patterns, clinical chorioamnionitis, multifetal gestations, premature deliveries, and all infants with low Apgar scores at 1 or 5 minutes. If you think you may need a cord gas analysis, go ahead and obtain it. Cord gas analysis often will aid in justifying your management or provide insight into the infant’s status.
Controversy remains as to the benefit of universal cord gas analysis. Assuming a variable cost of $15 for 2 (artery and vein) blood gas samples per neonate,4 the annual cost in the United States would be approximately $60 million. This would likely be cost effective as a result of medicolegal and educational benefits as well as potential improvements in perinatal outcome5 and reductions in special care nursery admissions.4
CASE 1: A newborn with unexpected acidosis
A 29-year-old woman (G2P1) at 38 weeks’ gestation was admitted to the hospital following an office visit during which oligohydramnios (amniotic fluid index, 3.5 cm) was found. The patient had a history of a prior cesarean delivery for failure to progress, and she desired a repeat cesarean delivery. Fetal monitoring revealed a heart rate of 140 beats per minute with moderate variability and uterine contractions every 3 to 5 minutes associated with moderate variable decelerations. A decision was made to proceed with the surgery. Blood samples were drawn for laboratory analysis, monitoring was discontinued, and the patient was taken to the operating room. An epidural anesthetic was placed and the cesarean delivery proceeded.
On uterine incision, there was no evidence of abruption or uterine rupture, but thick meconium-stained amniotic fluid was observed. A depressed infant was delivered, the umbilical cord clamped, and the infant handed to the pediatric team. Cord samples were obtained and values from the umbilical artery were as follows: pH, 6.80; P
What happened?
Read how to use cord gas values in practice
Using cord gas values in practice
Before analyzing the circumstances in Case 1,it is important to consider several key questions, including:
- What are the normal levels of cord pH, O2, CO2, and base deficit (BD)?
- How does cord gas indicate what happened during labor?
- What are the preventable errors in cord gas sampling or interpretation?
For a review of fetal cord gas physiology, see “Physiology of fetal cord gases: The basics”.
A review of basic fetal cord gas physiology will assist in understanding how values are interpreted.
Umbilical cord O2 and CO2
Fetal cord gas values result from the rapid transfer of gases and the slow clearance of acid across the placenta. Approximately 10% of maternal blood flow supplies the uteroplacental circulation, with the near-term placenta receiving approximately 70% of the uterine blood flow.1 Of the oxygen delivered, a surprising 50% provides for placental metabolism and 50% for the fetus. On the fetal side, 40% of fetal cardiac output supplies the umbilical circulation. Oxygen and carbon dioxide pass readily across the placental layers; exchange is limited by the amount of blood flow on both the maternal and the fetal side (flow limited). In the human placenta, maternal blood and fetal blood effectively travel in the same direction (concurrent exchange); thus, umbilical vein O2 and CO2 equilibrate with that in the maternal uterine vein.
Most of the O2 in fetal blood is carried by hemoglobin. Because of the markedly greater affinity of fetal hemoglobin for O2, the saturation curve is shifted to the left, resulting in increased hemoglobin saturation at the relatively low levels of fetal Po2. This greater affinity for oxygen results from the unique fetal hemoglobin gamma (γ) subunit, as compared with the adult beta (ß) subunit. Fetal hemoglobin has a reduced interaction with 2,3-bisphosphoglycerate, which itself decreases the affinity of adult hemoglobin for oxygen.
The majority of CO2 (85%) is carried as part of the bicarbonate buffer system. Fetal CO2 is converted into carbonic acid (H2CO3) in the red cell and dissociates into hydrogen (H+) and bicarbonate (HCO3−) ions, which diffuse out of the cell. When fetal blood reaches the placenta, this process is reversed and CO2 diffuses across the placenta to the maternal circulation. The production of H+ ions from CO2 explains the development of respiratory acidosis from high Pco2. In contrast, anaerobic metabolism, which produces lactic acid, results in metabolic acidosis.
Difference between pH and BD
The pH is calculated as the inverse log of the H+ ion concentration; thus, the pH falls as the H+ ion concentration exponentially increases, whether due to respiratory or metabolic acidosis. To quantify the more important metabolic acidosis, we use BD, which is a measure of how much of bicarbonate buffer base has been used by (lactic) acid. The BD and the base excess (BE) may be used interchangeably, with BE representing a negative number. Although BD represents the metabolic component of acidosis, a correction may be required to account for high levels of fetal Pco2 (see Case 1). In this situation, a more accurate measure is BD extracellular fluid (BDECF).
Why not just use pH? There are 2 major limitations to using pH as a measure of fetal or newborn acidosis. First, pH may be influenced by both respiratory and metabolic alterations, although only metabolic acidosis is associated with fetal neurologic injury.2 Furthermore, as pH is a log function, it does not change linearly with the amount of acid produced. In contrast to pH, BD is a measure of metabolic acidosis and changes in direct proportion to fetal acid production.
What about lactate? Measurements of lactate may also be included in blood gas analyses. Under hypoxic conditions, excess pyruvate is converted into lactate and released from the cell along with H+, resulting in acidosis. However, levels of umbilical cord lactate associated with neonatal hypoxic injury have not been established to the same degree as have pH or BD. Nevertheless, lactate has been measured in fetal scalp blood samples and offers the potential as a marker of fetal hypoxemia and acidosis.3
References
- Assali NS. Dynamics of the uteroplacental circulation in health and disease. Am J Perinatol. 1989;6(2):105-109.
- Low JA, Panagiotopoulos C, Derrick EJ. Newborn complications after intrapartum asphyxia with metabolic acidosis in the term fetus. Am J Obstet Gynecol. 1994;170(4):1081-1087.
- Mancho JP, Gamboa SM, Gimenez OR, Esteras RC, Solanilla BR, Mateo SC. Diagnostic accuracy of fetal scalp lactate for intrapartum acidosis compared with scalp pH [published online ahead of print October 8, 2016]. J Perinatal Med. doi: 10.1515/jpm-2016-004.
Normal values: The “20, 30, 40, 50 rule”
Among the values reported for umbilical blood gas, the pH, P
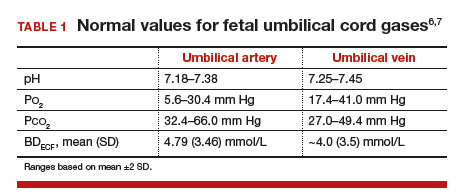
I recommend using the “20, 30, 40, 50 rule” as a simple tool for remembering normal umbilical artery and vein P
- P
o 2 values are lower than Pco 2 values; thus, the 20 and 30 represent Po 2 values - as fetal umbilical artery P
o 2 is lower than umbilical vein Po 2, 20 mm Hg represents the umbilical artery and 30 mm Hg represents the vein - P
co 2 values are higher in the umbilical artery than in the vein; thus, 50 mm Hg represents the umbilical artery and 40 mm Hg represents the umbilical vein.

Umbilical cord BD values change in relation to labor and FHR decelerations.8 Prior to labor, the normal fetus has a slight degree of acidosis (BD, 2 mmol/L). During the latent phase of labor, fetal BD typically does not change. With the increased frequency of contractions, BD may increase 1 mmol/L for every 3 to 6 hours during the active phase and up to 1 mmol/L per hour during the second stage, depending on FHR responses. Thus, following vaginal delivery the average umbilical artery BD is approximately 5 mmol/L and the umbilical vein BD is approximately 4 mmol/L. As lactate crosses the placenta slowly, BD values are typically only 1 mmol/L less in the umbilical vein than in the artery, unless there has been an obstruction to placental flow (see Case 1).
For pH, the umbilical artery value is always lower than that of the vein, a result of both the higher umbilical artery P
Possible causes of abnormal cord gas values
Because of the nearly fully saturated maternal hemoglobin under normal conditions, fetal arterial and venous P
In contrast, reduced fetal P
Effect of maternal oxygen administration on fetal oxygenation
Although maternal oxygen administration is commonly used during labor and delivery, controversy remains as to the benefit of oxygen supplementation.10 In a normal mother with oxygen saturation above 95%, the administration of oxygen will increase maternal arterial P
However, maternal oxygen supplementation may have marked benefit in cases in which maternal arterial P
How did the Case 1 circumstances lead to newborn acidosis?
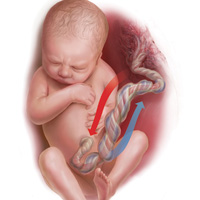
Most noticeable in this case is the large difference in BD between the umbilical artery and vein and the high P
Whereas BD normally is only about 1 mmol/L greater in the umbilical artery versus in the vein, occasionally the arterial value is markedly greater than the vein value. This can occur when there is a cessation of blood flow through the placenta, as a result of complete umbilical cord obstruction, or when there is a uterine abruption. In these situations, the umbilical vein (which has not had blood flow) represents the fetal status prior to the occlusion event. In contrast, despite bradycardia, fetal heart pulsations mix blood within the umbilical artery and therefore the artery generally represents the fetal status at the time of birth.
In response to complete cord occlusion, fetal BD increases by approximately 1 mmol/L every 2 minutes. Consequently, an 8 mmol/L difference in BD between the umbilical artery and vein is consistent with a 16-minute period of umbilical occlusion or placental abruption. Also in response to complete umbilical cord occlusion, P
The umbilical vein BD is also elevated for early labor. This value suggests that repetitive, intermittent cord occlusions (evident on the initial fetal monitor tracing) likely resulted in this moderate acidosis prior to the complete cord occlusion in the final 16 minutes.
Thus, BD and P
Read more cases plus procedures, equipment for cord sampling
CASE 2: An infant with unusual umbilical artery values
An infant born via vacuum delivery for a prolonged second stage of labor had 1- and 5-minute Apgar scores of 8 and 9, respectively. Cord gas values were obtained, and analysis revealed that for the umbilical artery, the pH was 7.29; P
The resident asked, “How is the P
The curious Case 2 values suggest an air bubble
Although it is possible that the aberrant values in Case 2 could have resulted from switching the artery and vein samples, the pH is lower in the artery, and both the artery P
Related article:
Is neonatal injury more likely outside of a 30-minute decision-to-incision time interval for cesarean delivery?
CASE 3: A vigorous baby with significant acidosis
A baby with 1- and 5-minute Apgar scores of 9 and 9 was delivered by cesarean and remained vigorous. Umbilical cord analysis revealed an umbilical artery pH level of 7.15, with normal P
Was there a collection error in Case 3?
On occasion, a falsely low pH level and, thus, a falsely elevated BD may result from excessive heparin in the collection syringe. Heparin is acidotic and should be used only to coat the syringe. Although syringes in current use are often pre-heparinized, if one is drawing up heparin into the syringe, it should be coated and then fully expelled.
Umbilical cord sampling: Procedures and equipment
Many issues remain regarding the optimal storage of cord samples. Ideally, a doubly clamped section of the cord promptly should be sampled into glass syringes that can be placed on ice and rapidly measured for cord values.
Stability of umbilical cord samples within the cord is within 20 to 30 minutes. Delayed sampling of clamped cord sections generally has minimal effect on pH and P
Plastic syringes can introduce interference. Several studies have demonstrated that collection of samples in plastic may result in an increase in P
Use glass, and “ice” the sample if necessary. Although it has been suggested that placing samples on ice minimizes metabolism, the cooled plastic may in fact be more susceptible to oxygen diffusion. Thus, unless samples will be analyzed promptly, it is best to use glass syringes on ice.13,14
Related article:
Protecting the newborn brain—the final frontier in obstetric and neonatal care
What if the umbilical cord is torn?
Sometimes the umbilical cord is torn and discarded or cannot be accessed for other reasons. A sample can still be obtained, however, by aspirating the placental surface artery and vein vessels. Although there is some potential variance in pH, P
How do you obtain cord analysis when delaying cord clamping?
The American College of Obstetricians and Gynecologists (ACOG) now advises delayed cord clamping in term and preterm deliveries, which raises the question of how you obtain a blood sample in this setting. Importantly, ACOG recommends delayed cord clamping only in vigorous infants,16 whereas potentially compromised infants should be transferred rapidly for newborn care. Although several studies have demonstrated some variation in cord gas values with delayed cord clamping,17–21 clamping after pulsation has ceased or after the recommended 30 to 60 seconds following birth results in minimal change in BD values. Thus, do not hesitate to perform delayed cord clamping in vigorous infants.
Share your thoughts! Send your Letter to the Editor to [email protected]. Please include your name and the city and state in which you practice.
- Ross MG, Gala R. Use of umbilical artery base excess: algorithm for the timing of hypoxic injury. Am J Obstet Gynecol. 2002;187(1):1–9.
- Uccella S, Cromi A, Colombo G, et al. Prediction of fetal base excess values at birth using an algorithm to interpret fetal heart rate tracings: a retrospective validation. BJOG. 2012;119(13):1657–1664.
- Uccella S, Cromi A, Colombo GF, et al. Interobserver reliability to interpret intrapartum electronic fetal heart rate monitoring: does a standardized algorithm improve agreement among clinicians? J Obstet Gynaecol. 2015;35(3):241–245.
- White CR, Doherty DA, Cannon JW, Kohan R, Newnham JP, Pennell CE. Cost effectiveness of universal umbilical cord blood gas and lactate analysis in a tertiary level maternity unit. J Perinat Med. 2016;44(5):573–584.
- White CR, Doherty DA, Henderson JJ, Kohan R, Newnham JP, Pennell CE. Benefits of introducing universal umbilical cord blood gas and lactate analysis into an obstetric unit. Aust N Z J Obstet Gynaecol. 2010;50(4):318–328.
- Yeomans ER, Hauth JC, Gilstrap LC III, Strickland DM. Umbilical cord pH, Pco2, and bicarbonate following uncomplicated term vaginal deliveries. Am J Obstet Gynecol. 1985;151(6):798–800.
- Wiberg N, Källén K, Olofsson P. Base deficit estimation in umbilical cord blood is influenced by gestational age, choice of fetal fluid compartment, and algorithm for calculation. Am J Obstet Gynecol. 2006;195(6):1651–1656.
- Ross MG, Gala R. Use of umbilical artery base excess: algorithm for the timing of hypoxic injury. Am J Obstet Gynecol. 2002;187(1):1–9.
- Executive summary: Neonatal encephalopathy and neurologic outcome, second edition. Report of the American College of Obstetricians and Gynecologists’ Task Force on Neonatal Encephalopathy. Obstet Gynecol. 2014;123(4):896-901.
- Hamel MS, Anderson BL, Rouse DJ. Oxygen for intrauterine resuscitation: of unproved benefit and potentially harmful. Am J Obstet Gynecol. 2014;211(2):124–127.
- Owen P, Farrell TA, Steyn W. Umbilical cord blood gas analysis; a comparison of two simple methods of sample storage. Early Hum Dev. 1995;42(1):67–71.
- Armstrong L, Stenson B. Effect of delayed sampling on umbilical cord arterial and venous lactate and blood gases in clamped and unclamped vessels. Arch Dis Child Fetal Neonatal Ed. 2006;91(5):F342–F345.
- White CR, Mok T, Doherty DA, Henderson JJ, Newnham JP, Pennell CE. The effect of time, temperature and storage device on umbilical cord blood gas and lactate measurement: a randomized controlled trial. J Matern Fetal Neonatal Med. 2012;25(6):587–594.
- Knowles TP, Mullin RA, Hunter JA, Douce FH. Effects of syringe material, sample storage time, and temperature on blood gases and oxygen saturation in arterialized human blood samples. Respir Care. 2006;51(7):732–736.
- Nodwell A, Carmichael L, Ross M, Richardson B. Placental compared with umbilical cord blood to assess fetal blood gas and acid-base status. Obstet Gynecol. 2005;105(1):129–138.
- American College of Obstetricians and Gynecologists. ACOG Committee Opinion No. 684. Delayed umbilical cord clamping after birth. Obstet Gynecol. 2017;129(1):e5–e10.
- De Paco C, Florido J, Garrido MC, Prados S, Navarrete L. Umbilical cord blood acid-base and gas analysis after early versus delayed cord clamping in neonates at term. Arch Gynecol Obstet. 2011;283(5):1011–1014.
- Valero J, Desantes D, Perales-Puchalt A, Rubio J, Diago Almela VJ, Perales A. Effect of delayed umbilical cord clamping on blood gas analysis. Eur J Obstet Gynecol Reprod Biol. 2012;162(1): 21–23.
- Andersson O, Hellström-Westas L, Andersson D, Clausen J, Domellöf M. Effects of delayed compared with early umbilical cord clamping on maternal postpartum hemorrhage and cord blood gas sampling: a randomized trial. Acta Obstet Gynecol Scand. 2013;92(5):567–574.
- Wiberg N, Källén K, Olofsson P. Delayed umbilical cord clamping at birth has effects on arterial and venous blood gases and lactate concentrations. BJOG. 2008;115(6):697–703.
- Mokarami P, Wiberg N, Olofsson P. Hidden acidosis: an explanation of acid-base and lactate changes occurring in umbilical cord blood after delayed sampling. BJOG. 2013;120(8):996–1002.
Umbilical cord blood (cord) gas values can aid both in understanding the cause of an infant’s acidosis and in providing reassurance that acute acidosis or asphyxia is not responsible for a compromised infant with a low Apgar score. Together with other clinical measurements (including fetal heart rate [FHR] tracings, Apgar scores, newborn nucleated red cell counts, and neonatal imaging), cord gas analysis can be remarkably helpful in determining the cause for a depressed newborn. It can help us determine, for example, if infant compromise was a result of an asphyxial event, and we often can differentiate whether the event was acute, prolonged, or occurred prior to presentation in labor. We further can use cord gas values to assess whether a decision for operative intervention for nonreassuring fetal well-being was appropriate (see “Brain injury at birth: Cord gas values presented as evidence at trial”). In addition, cord gas analysis can complement methods for determining fetal acidosis changes during labor, enabling improved assessment of FHR tracings.1−3
At 40 weeks' gestation, a woman presented to the hospital because of decreased fetal movement. On arrival, an external fetal heart-rate (FHR) monitor showed nonreassuring tracings, evidenced by absent to minimal variability and subtle decelerations occurring at 10- to 15-minute intervals. The on-call ObGyn requested induction of labor with oxytocin, and a low-dose infusion (1 mU/min) was initiated. An internal FHR monitor was then placed and late decelerations were observed with the first 2 induced contractions. The oxytocin infusion was discontinued and the ObGyn performed an emergency cesarean delivery. The infant's Apgar scores were 1, 2, and 2 at 1, 5, and 10 minutes, respectively. Cord samples were obtained and values from the umbilical artery were as follows: pH, 6.86; Pco2, 55 mm Hg; Po2, 6 mm Hg; and BDECF, 21.1 mmol/L. Values from the umbilical vein were: pH, 6.94; Pco2, 45 mm Hg; Po2, 17 mm Hg; and BDECF, 20.0 mmol/L. The infant was later diagnosed with a hypoxic brain injury resulting in cerebral palsy. At trial years later, the boy had cognitive and physical limitations and required 24-hour care.
The parents claimed that the ObGyn should have performed a cesarean delivery earlier when the external FHR monitor showed nonreassuring tracings.
The hospital and physician claimed that, while tracings were consistently nonreassuring, they were stable. They maintained that the child's brain damage was not due to a delivery delay, as the severe level of acidosis in both the umbilical artery and vein could not be a result of the few heart rate decelerations during the 2-hour period of monitoring prior to delivery. They argued that the clinical picture indicated a pre-hospital hypoxic event associated with decreased fetal movement.
A defense verdict was returned.
Case assessment
Cord gas results, together with other measures (eg, infant nucleated red blood cells, brain imaging) can aid the ObGyn in medicolegal cases. However, they are not always protective of adverse judgment.
I recommend checking umbilical cord blood gas values on all operative vaginal deliveries, cesarean deliveries for fetal concern, abnormal FHR patterns, clinical chorioamnionitis, multifetal gestations, premature deliveries, and all infants with low Apgar scores at 1 or 5 minutes. If you think you may need a cord gas analysis, go ahead and obtain it. Cord gas analysis often will aid in justifying your management or provide insight into the infant’s status.
Controversy remains as to the benefit of universal cord gas analysis. Assuming a variable cost of $15 for 2 (artery and vein) blood gas samples per neonate,4 the annual cost in the United States would be approximately $60 million. This would likely be cost effective as a result of medicolegal and educational benefits as well as potential improvements in perinatal outcome5 and reductions in special care nursery admissions.4
CASE 1: A newborn with unexpected acidosis
A 29-year-old woman (G2P1) at 38 weeks’ gestation was admitted to the hospital following an office visit during which oligohydramnios (amniotic fluid index, 3.5 cm) was found. The patient had a history of a prior cesarean delivery for failure to progress, and she desired a repeat cesarean delivery. Fetal monitoring revealed a heart rate of 140 beats per minute with moderate variability and uterine contractions every 3 to 5 minutes associated with moderate variable decelerations. A decision was made to proceed with the surgery. Blood samples were drawn for laboratory analysis, monitoring was discontinued, and the patient was taken to the operating room. An epidural anesthetic was placed and the cesarean delivery proceeded.
On uterine incision, there was no evidence of abruption or uterine rupture, but thick meconium-stained amniotic fluid was observed. A depressed infant was delivered, the umbilical cord clamped, and the infant handed to the pediatric team. Cord samples were obtained and values from the umbilical artery were as follows: pH, 6.80; P
What happened?
Read how to use cord gas values in practice
Using cord gas values in practice
Before analyzing the circumstances in Case 1,it is important to consider several key questions, including:
- What are the normal levels of cord pH, O2, CO2, and base deficit (BD)?
- How does cord gas indicate what happened during labor?
- What are the preventable errors in cord gas sampling or interpretation?
For a review of fetal cord gas physiology, see “Physiology of fetal cord gases: The basics”.
A review of basic fetal cord gas physiology will assist in understanding how values are interpreted.
Umbilical cord O2 and CO2
Fetal cord gas values result from the rapid transfer of gases and the slow clearance of acid across the placenta. Approximately 10% of maternal blood flow supplies the uteroplacental circulation, with the near-term placenta receiving approximately 70% of the uterine blood flow.1 Of the oxygen delivered, a surprising 50% provides for placental metabolism and 50% for the fetus. On the fetal side, 40% of fetal cardiac output supplies the umbilical circulation. Oxygen and carbon dioxide pass readily across the placental layers; exchange is limited by the amount of blood flow on both the maternal and the fetal side (flow limited). In the human placenta, maternal blood and fetal blood effectively travel in the same direction (concurrent exchange); thus, umbilical vein O2 and CO2 equilibrate with that in the maternal uterine vein.
Most of the O2 in fetal blood is carried by hemoglobin. Because of the markedly greater affinity of fetal hemoglobin for O2, the saturation curve is shifted to the left, resulting in increased hemoglobin saturation at the relatively low levels of fetal Po2. This greater affinity for oxygen results from the unique fetal hemoglobin gamma (γ) subunit, as compared with the adult beta (ß) subunit. Fetal hemoglobin has a reduced interaction with 2,3-bisphosphoglycerate, which itself decreases the affinity of adult hemoglobin for oxygen.
The majority of CO2 (85%) is carried as part of the bicarbonate buffer system. Fetal CO2 is converted into carbonic acid (H2CO3) in the red cell and dissociates into hydrogen (H+) and bicarbonate (HCO3−) ions, which diffuse out of the cell. When fetal blood reaches the placenta, this process is reversed and CO2 diffuses across the placenta to the maternal circulation. The production of H+ ions from CO2 explains the development of respiratory acidosis from high Pco2. In contrast, anaerobic metabolism, which produces lactic acid, results in metabolic acidosis.
Difference between pH and BD
The pH is calculated as the inverse log of the H+ ion concentration; thus, the pH falls as the H+ ion concentration exponentially increases, whether due to respiratory or metabolic acidosis. To quantify the more important metabolic acidosis, we use BD, which is a measure of how much of bicarbonate buffer base has been used by (lactic) acid. The BD and the base excess (BE) may be used interchangeably, with BE representing a negative number. Although BD represents the metabolic component of acidosis, a correction may be required to account for high levels of fetal Pco2 (see Case 1). In this situation, a more accurate measure is BD extracellular fluid (BDECF).
Why not just use pH? There are 2 major limitations to using pH as a measure of fetal or newborn acidosis. First, pH may be influenced by both respiratory and metabolic alterations, although only metabolic acidosis is associated with fetal neurologic injury.2 Furthermore, as pH is a log function, it does not change linearly with the amount of acid produced. In contrast to pH, BD is a measure of metabolic acidosis and changes in direct proportion to fetal acid production.
What about lactate? Measurements of lactate may also be included in blood gas analyses. Under hypoxic conditions, excess pyruvate is converted into lactate and released from the cell along with H+, resulting in acidosis. However, levels of umbilical cord lactate associated with neonatal hypoxic injury have not been established to the same degree as have pH or BD. Nevertheless, lactate has been measured in fetal scalp blood samples and offers the potential as a marker of fetal hypoxemia and acidosis.3
References
- Assali NS. Dynamics of the uteroplacental circulation in health and disease. Am J Perinatol. 1989;6(2):105-109.
- Low JA, Panagiotopoulos C, Derrick EJ. Newborn complications after intrapartum asphyxia with metabolic acidosis in the term fetus. Am J Obstet Gynecol. 1994;170(4):1081-1087.
- Mancho JP, Gamboa SM, Gimenez OR, Esteras RC, Solanilla BR, Mateo SC. Diagnostic accuracy of fetal scalp lactate for intrapartum acidosis compared with scalp pH [published online ahead of print October 8, 2016]. J Perinatal Med. doi: 10.1515/jpm-2016-004.
Normal values: The “20, 30, 40, 50 rule”
Among the values reported for umbilical blood gas, the pH, P

I recommend using the “20, 30, 40, 50 rule” as a simple tool for remembering normal umbilical artery and vein P
- P
o 2 values are lower than Pco 2 values; thus, the 20 and 30 represent Po 2 values - as fetal umbilical artery P
o 2 is lower than umbilical vein Po 2, 20 mm Hg represents the umbilical artery and 30 mm Hg represents the vein - P
co 2 values are higher in the umbilical artery than in the vein; thus, 50 mm Hg represents the umbilical artery and 40 mm Hg represents the umbilical vein.

Umbilical cord BD values change in relation to labor and FHR decelerations.8 Prior to labor, the normal fetus has a slight degree of acidosis (BD, 2 mmol/L). During the latent phase of labor, fetal BD typically does not change. With the increased frequency of contractions, BD may increase 1 mmol/L for every 3 to 6 hours during the active phase and up to 1 mmol/L per hour during the second stage, depending on FHR responses. Thus, following vaginal delivery the average umbilical artery BD is approximately 5 mmol/L and the umbilical vein BD is approximately 4 mmol/L. As lactate crosses the placenta slowly, BD values are typically only 1 mmol/L less in the umbilical vein than in the artery, unless there has been an obstruction to placental flow (see Case 1).
For pH, the umbilical artery value is always lower than that of the vein, a result of both the higher umbilical artery P
Possible causes of abnormal cord gas values
Because of the nearly fully saturated maternal hemoglobin under normal conditions, fetal arterial and venous P
In contrast, reduced fetal P
Effect of maternal oxygen administration on fetal oxygenation
Although maternal oxygen administration is commonly used during labor and delivery, controversy remains as to the benefit of oxygen supplementation.10 In a normal mother with oxygen saturation above 95%, the administration of oxygen will increase maternal arterial P
However, maternal oxygen supplementation may have marked benefit in cases in which maternal arterial P
How did the Case 1 circumstances lead to newborn acidosis?
Most noticeable in this case is the large difference in BD between the umbilical artery and vein and the high P
Whereas BD normally is only about 1 mmol/L greater in the umbilical artery versus in the vein, occasionally the arterial value is markedly greater than the vein value. This can occur when there is a cessation of blood flow through the placenta, as a result of complete umbilical cord obstruction, or when there is a uterine abruption. In these situations, the umbilical vein (which has not had blood flow) represents the fetal status prior to the occlusion event. In contrast, despite bradycardia, fetal heart pulsations mix blood within the umbilical artery and therefore the artery generally represents the fetal status at the time of birth.
In response to complete cord occlusion, fetal BD increases by approximately 1 mmol/L every 2 minutes. Consequently, an 8 mmol/L difference in BD between the umbilical artery and vein is consistent with a 16-minute period of umbilical occlusion or placental abruption. Also in response to complete umbilical cord occlusion, P
The umbilical vein BD is also elevated for early labor. This value suggests that repetitive, intermittent cord occlusions (evident on the initial fetal monitor tracing) likely resulted in this moderate acidosis prior to the complete cord occlusion in the final 16 minutes.
Thus, BD and P
Read more cases plus procedures, equipment for cord sampling
CASE 2: An infant with unusual umbilical artery values
An infant born via vacuum delivery for a prolonged second stage of labor had 1- and 5-minute Apgar scores of 8 and 9, respectively. Cord gas values were obtained, and analysis revealed that for the umbilical artery, the pH was 7.29; P
The resident asked, “How is the P
The curious Case 2 values suggest an air bubble
Although it is possible that the aberrant values in Case 2 could have resulted from switching the artery and vein samples, the pH is lower in the artery, and both the artery P
Related article:
Is neonatal injury more likely outside of a 30-minute decision-to-incision time interval for cesarean delivery?
CASE 3: A vigorous baby with significant acidosis
A baby with 1- and 5-minute Apgar scores of 9 and 9 was delivered by cesarean and remained vigorous. Umbilical cord analysis revealed an umbilical artery pH level of 7.15, with normal P
Was there a collection error in Case 3?
On occasion, a falsely low pH level and, thus, a falsely elevated BD may result from excessive heparin in the collection syringe. Heparin is acidotic and should be used only to coat the syringe. Although syringes in current use are often pre-heparinized, if one is drawing up heparin into the syringe, it should be coated and then fully expelled.
Umbilical cord sampling: Procedures and equipment
Many issues remain regarding the optimal storage of cord samples. Ideally, a doubly clamped section of the cord promptly should be sampled into glass syringes that can be placed on ice and rapidly measured for cord values.
Stability of umbilical cord samples within the cord is within 20 to 30 minutes. Delayed sampling of clamped cord sections generally has minimal effect on pH and P
Plastic syringes can introduce interference. Several studies have demonstrated that collection of samples in plastic may result in an increase in P
Use glass, and “ice” the sample if necessary. Although it has been suggested that placing samples on ice minimizes metabolism, the cooled plastic may in fact be more susceptible to oxygen diffusion. Thus, unless samples will be analyzed promptly, it is best to use glass syringes on ice.13,14
Related article:
Protecting the newborn brain—the final frontier in obstetric and neonatal care
What if the umbilical cord is torn?
Sometimes the umbilical cord is torn and discarded or cannot be accessed for other reasons. A sample can still be obtained, however, by aspirating the placental surface artery and vein vessels. Although there is some potential variance in pH, P
How do you obtain cord analysis when delaying cord clamping?
The American College of Obstetricians and Gynecologists (ACOG) now advises delayed cord clamping in term and preterm deliveries, which raises the question of how you obtain a blood sample in this setting. Importantly, ACOG recommends delayed cord clamping only in vigorous infants,16 whereas potentially compromised infants should be transferred rapidly for newborn care. Although several studies have demonstrated some variation in cord gas values with delayed cord clamping,17–21 clamping after pulsation has ceased or after the recommended 30 to 60 seconds following birth results in minimal change in BD values. Thus, do not hesitate to perform delayed cord clamping in vigorous infants.
Share your thoughts! Send your Letter to the Editor to [email protected]. Please include your name and the city and state in which you practice.
Umbilical cord blood (cord) gas values can aid both in understanding the cause of an infant’s acidosis and in providing reassurance that acute acidosis or asphyxia is not responsible for a compromised infant with a low Apgar score. Together with other clinical measurements (including fetal heart rate [FHR] tracings, Apgar scores, newborn nucleated red cell counts, and neonatal imaging), cord gas analysis can be remarkably helpful in determining the cause for a depressed newborn. It can help us determine, for example, if infant compromise was a result of an asphyxial event, and we often can differentiate whether the event was acute, prolonged, or occurred prior to presentation in labor. We further can use cord gas values to assess whether a decision for operative intervention for nonreassuring fetal well-being was appropriate (see “Brain injury at birth: Cord gas values presented as evidence at trial”). In addition, cord gas analysis can complement methods for determining fetal acidosis changes during labor, enabling improved assessment of FHR tracings.1−3
At 40 weeks' gestation, a woman presented to the hospital because of decreased fetal movement. On arrival, an external fetal heart-rate (FHR) monitor showed nonreassuring tracings, evidenced by absent to minimal variability and subtle decelerations occurring at 10- to 15-minute intervals. The on-call ObGyn requested induction of labor with oxytocin, and a low-dose infusion (1 mU/min) was initiated. An internal FHR monitor was then placed and late decelerations were observed with the first 2 induced contractions. The oxytocin infusion was discontinued and the ObGyn performed an emergency cesarean delivery. The infant's Apgar scores were 1, 2, and 2 at 1, 5, and 10 minutes, respectively. Cord samples were obtained and values from the umbilical artery were as follows: pH, 6.86; Pco2, 55 mm Hg; Po2, 6 mm Hg; and BDECF, 21.1 mmol/L. Values from the umbilical vein were: pH, 6.94; Pco2, 45 mm Hg; Po2, 17 mm Hg; and BDECF, 20.0 mmol/L. The infant was later diagnosed with a hypoxic brain injury resulting in cerebral palsy. At trial years later, the boy had cognitive and physical limitations and required 24-hour care.
The parents claimed that the ObGyn should have performed a cesarean delivery earlier when the external FHR monitor showed nonreassuring tracings.
The hospital and physician claimed that, while tracings were consistently nonreassuring, they were stable. They maintained that the child's brain damage was not due to a delivery delay, as the severe level of acidosis in both the umbilical artery and vein could not be a result of the few heart rate decelerations during the 2-hour period of monitoring prior to delivery. They argued that the clinical picture indicated a pre-hospital hypoxic event associated with decreased fetal movement.
A defense verdict was returned.
Case assessment
Cord gas results, together with other measures (eg, infant nucleated red blood cells, brain imaging) can aid the ObGyn in medicolegal cases. However, they are not always protective of adverse judgment.
I recommend checking umbilical cord blood gas values on all operative vaginal deliveries, cesarean deliveries for fetal concern, abnormal FHR patterns, clinical chorioamnionitis, multifetal gestations, premature deliveries, and all infants with low Apgar scores at 1 or 5 minutes. If you think you may need a cord gas analysis, go ahead and obtain it. Cord gas analysis often will aid in justifying your management or provide insight into the infant’s status.
Controversy remains as to the benefit of universal cord gas analysis. Assuming a variable cost of $15 for 2 (artery and vein) blood gas samples per neonate,4 the annual cost in the United States would be approximately $60 million. This would likely be cost effective as a result of medicolegal and educational benefits as well as potential improvements in perinatal outcome5 and reductions in special care nursery admissions.4
CASE 1: A newborn with unexpected acidosis
A 29-year-old woman (G2P1) at 38 weeks’ gestation was admitted to the hospital following an office visit during which oligohydramnios (amniotic fluid index, 3.5 cm) was found. The patient had a history of a prior cesarean delivery for failure to progress, and she desired a repeat cesarean delivery. Fetal monitoring revealed a heart rate of 140 beats per minute with moderate variability and uterine contractions every 3 to 5 minutes associated with moderate variable decelerations. A decision was made to proceed with the surgery. Blood samples were drawn for laboratory analysis, monitoring was discontinued, and the patient was taken to the operating room. An epidural anesthetic was placed and the cesarean delivery proceeded.
On uterine incision, there was no evidence of abruption or uterine rupture, but thick meconium-stained amniotic fluid was observed. A depressed infant was delivered, the umbilical cord clamped, and the infant handed to the pediatric team. Cord samples were obtained and values from the umbilical artery were as follows: pH, 6.80; P
What happened?
Read how to use cord gas values in practice
Using cord gas values in practice
Before analyzing the circumstances in Case 1,it is important to consider several key questions, including:
- What are the normal levels of cord pH, O2, CO2, and base deficit (BD)?
- How does cord gas indicate what happened during labor?
- What are the preventable errors in cord gas sampling or interpretation?
For a review of fetal cord gas physiology, see “Physiology of fetal cord gases: The basics”.
A review of basic fetal cord gas physiology will assist in understanding how values are interpreted.
Umbilical cord O2 and CO2
Fetal cord gas values result from the rapid transfer of gases and the slow clearance of acid across the placenta. Approximately 10% of maternal blood flow supplies the uteroplacental circulation, with the near-term placenta receiving approximately 70% of the uterine blood flow.1 Of the oxygen delivered, a surprising 50% provides for placental metabolism and 50% for the fetus. On the fetal side, 40% of fetal cardiac output supplies the umbilical circulation. Oxygen and carbon dioxide pass readily across the placental layers; exchange is limited by the amount of blood flow on both the maternal and the fetal side (flow limited). In the human placenta, maternal blood and fetal blood effectively travel in the same direction (concurrent exchange); thus, umbilical vein O2 and CO2 equilibrate with that in the maternal uterine vein.
Most of the O2 in fetal blood is carried by hemoglobin. Because of the markedly greater affinity of fetal hemoglobin for O2, the saturation curve is shifted to the left, resulting in increased hemoglobin saturation at the relatively low levels of fetal Po2. This greater affinity for oxygen results from the unique fetal hemoglobin gamma (γ) subunit, as compared with the adult beta (ß) subunit. Fetal hemoglobin has a reduced interaction with 2,3-bisphosphoglycerate, which itself decreases the affinity of adult hemoglobin for oxygen.
The majority of CO2 (85%) is carried as part of the bicarbonate buffer system. Fetal CO2 is converted into carbonic acid (H2CO3) in the red cell and dissociates into hydrogen (H+) and bicarbonate (HCO3−) ions, which diffuse out of the cell. When fetal blood reaches the placenta, this process is reversed and CO2 diffuses across the placenta to the maternal circulation. The production of H+ ions from CO2 explains the development of respiratory acidosis from high Pco2. In contrast, anaerobic metabolism, which produces lactic acid, results in metabolic acidosis.
Difference between pH and BD
The pH is calculated as the inverse log of the H+ ion concentration; thus, the pH falls as the H+ ion concentration exponentially increases, whether due to respiratory or metabolic acidosis. To quantify the more important metabolic acidosis, we use BD, which is a measure of how much of bicarbonate buffer base has been used by (lactic) acid. The BD and the base excess (BE) may be used interchangeably, with BE representing a negative number. Although BD represents the metabolic component of acidosis, a correction may be required to account for high levels of fetal Pco2 (see Case 1). In this situation, a more accurate measure is BD extracellular fluid (BDECF).
Why not just use pH? There are 2 major limitations to using pH as a measure of fetal or newborn acidosis. First, pH may be influenced by both respiratory and metabolic alterations, although only metabolic acidosis is associated with fetal neurologic injury.2 Furthermore, as pH is a log function, it does not change linearly with the amount of acid produced. In contrast to pH, BD is a measure of metabolic acidosis and changes in direct proportion to fetal acid production.
What about lactate? Measurements of lactate may also be included in blood gas analyses. Under hypoxic conditions, excess pyruvate is converted into lactate and released from the cell along with H+, resulting in acidosis. However, levels of umbilical cord lactate associated with neonatal hypoxic injury have not been established to the same degree as have pH or BD. Nevertheless, lactate has been measured in fetal scalp blood samples and offers the potential as a marker of fetal hypoxemia and acidosis.3
References
- Assali NS. Dynamics of the uteroplacental circulation in health and disease. Am J Perinatol. 1989;6(2):105-109.
- Low JA, Panagiotopoulos C, Derrick EJ. Newborn complications after intrapartum asphyxia with metabolic acidosis in the term fetus. Am J Obstet Gynecol. 1994;170(4):1081-1087.
- Mancho JP, Gamboa SM, Gimenez OR, Esteras RC, Solanilla BR, Mateo SC. Diagnostic accuracy of fetal scalp lactate for intrapartum acidosis compared with scalp pH [published online ahead of print October 8, 2016]. J Perinatal Med. doi: 10.1515/jpm-2016-004.
Normal values: The “20, 30, 40, 50 rule”
Among the values reported for umbilical blood gas, the pH, P

I recommend using the “20, 30, 40, 50 rule” as a simple tool for remembering normal umbilical artery and vein P
- P
o 2 values are lower than Pco 2 values; thus, the 20 and 30 represent Po 2 values - as fetal umbilical artery P
o 2 is lower than umbilical vein Po 2, 20 mm Hg represents the umbilical artery and 30 mm Hg represents the vein - P
co 2 values are higher in the umbilical artery than in the vein; thus, 50 mm Hg represents the umbilical artery and 40 mm Hg represents the umbilical vein.

Umbilical cord BD values change in relation to labor and FHR decelerations.8 Prior to labor, the normal fetus has a slight degree of acidosis (BD, 2 mmol/L). During the latent phase of labor, fetal BD typically does not change. With the increased frequency of contractions, BD may increase 1 mmol/L for every 3 to 6 hours during the active phase and up to 1 mmol/L per hour during the second stage, depending on FHR responses. Thus, following vaginal delivery the average umbilical artery BD is approximately 5 mmol/L and the umbilical vein BD is approximately 4 mmol/L. As lactate crosses the placenta slowly, BD values are typically only 1 mmol/L less in the umbilical vein than in the artery, unless there has been an obstruction to placental flow (see Case 1).
For pH, the umbilical artery value is always lower than that of the vein, a result of both the higher umbilical artery P
Possible causes of abnormal cord gas values
Because of the nearly fully saturated maternal hemoglobin under normal conditions, fetal arterial and venous P
In contrast, reduced fetal P
Effect of maternal oxygen administration on fetal oxygenation
Although maternal oxygen administration is commonly used during labor and delivery, controversy remains as to the benefit of oxygen supplementation.10 In a normal mother with oxygen saturation above 95%, the administration of oxygen will increase maternal arterial P
However, maternal oxygen supplementation may have marked benefit in cases in which maternal arterial P
How did the Case 1 circumstances lead to newborn acidosis?
Most noticeable in this case is the large difference in BD between the umbilical artery and vein and the high P
Whereas BD normally is only about 1 mmol/L greater in the umbilical artery versus in the vein, occasionally the arterial value is markedly greater than the vein value. This can occur when there is a cessation of blood flow through the placenta, as a result of complete umbilical cord obstruction, or when there is a uterine abruption. In these situations, the umbilical vein (which has not had blood flow) represents the fetal status prior to the occlusion event. In contrast, despite bradycardia, fetal heart pulsations mix blood within the umbilical artery and therefore the artery generally represents the fetal status at the time of birth.
In response to complete cord occlusion, fetal BD increases by approximately 1 mmol/L every 2 minutes. Consequently, an 8 mmol/L difference in BD between the umbilical artery and vein is consistent with a 16-minute period of umbilical occlusion or placental abruption. Also in response to complete umbilical cord occlusion, P
The umbilical vein BD is also elevated for early labor. This value suggests that repetitive, intermittent cord occlusions (evident on the initial fetal monitor tracing) likely resulted in this moderate acidosis prior to the complete cord occlusion in the final 16 minutes.
Thus, BD and P
Read more cases plus procedures, equipment for cord sampling
CASE 2: An infant with unusual umbilical artery values
An infant born via vacuum delivery for a prolonged second stage of labor had 1- and 5-minute Apgar scores of 8 and 9, respectively. Cord gas values were obtained, and analysis revealed that for the umbilical artery, the pH was 7.29; P
The resident asked, “How is the P
The curious Case 2 values suggest an air bubble
Although it is possible that the aberrant values in Case 2 could have resulted from switching the artery and vein samples, the pH is lower in the artery, and both the artery P
Related article:
Is neonatal injury more likely outside of a 30-minute decision-to-incision time interval for cesarean delivery?
CASE 3: A vigorous baby with significant acidosis
A baby with 1- and 5-minute Apgar scores of 9 and 9 was delivered by cesarean and remained vigorous. Umbilical cord analysis revealed an umbilical artery pH level of 7.15, with normal P
Was there a collection error in Case 3?
On occasion, a falsely low pH level and, thus, a falsely elevated BD may result from excessive heparin in the collection syringe. Heparin is acidotic and should be used only to coat the syringe. Although syringes in current use are often pre-heparinized, if one is drawing up heparin into the syringe, it should be coated and then fully expelled.
Umbilical cord sampling: Procedures and equipment
Many issues remain regarding the optimal storage of cord samples. Ideally, a doubly clamped section of the cord promptly should be sampled into glass syringes that can be placed on ice and rapidly measured for cord values.
Stability of umbilical cord samples within the cord is within 20 to 30 minutes. Delayed sampling of clamped cord sections generally has minimal effect on pH and P
Plastic syringes can introduce interference. Several studies have demonstrated that collection of samples in plastic may result in an increase in P
Use glass, and “ice” the sample if necessary. Although it has been suggested that placing samples on ice minimizes metabolism, the cooled plastic may in fact be more susceptible to oxygen diffusion. Thus, unless samples will be analyzed promptly, it is best to use glass syringes on ice.13,14
Related article:
Protecting the newborn brain—the final frontier in obstetric and neonatal care
What if the umbilical cord is torn?
Sometimes the umbilical cord is torn and discarded or cannot be accessed for other reasons. A sample can still be obtained, however, by aspirating the placental surface artery and vein vessels. Although there is some potential variance in pH, P
How do you obtain cord analysis when delaying cord clamping?
The American College of Obstetricians and Gynecologists (ACOG) now advises delayed cord clamping in term and preterm deliveries, which raises the question of how you obtain a blood sample in this setting. Importantly, ACOG recommends delayed cord clamping only in vigorous infants,16 whereas potentially compromised infants should be transferred rapidly for newborn care. Although several studies have demonstrated some variation in cord gas values with delayed cord clamping,17–21 clamping after pulsation has ceased or after the recommended 30 to 60 seconds following birth results in minimal change in BD values. Thus, do not hesitate to perform delayed cord clamping in vigorous infants.
Share your thoughts! Send your Letter to the Editor to [email protected]. Please include your name and the city and state in which you practice.
- Ross MG, Gala R. Use of umbilical artery base excess: algorithm for the timing of hypoxic injury. Am J Obstet Gynecol. 2002;187(1):1–9.
- Uccella S, Cromi A, Colombo G, et al. Prediction of fetal base excess values at birth using an algorithm to interpret fetal heart rate tracings: a retrospective validation. BJOG. 2012;119(13):1657–1664.
- Uccella S, Cromi A, Colombo GF, et al. Interobserver reliability to interpret intrapartum electronic fetal heart rate monitoring: does a standardized algorithm improve agreement among clinicians? J Obstet Gynaecol. 2015;35(3):241–245.
- White CR, Doherty DA, Cannon JW, Kohan R, Newnham JP, Pennell CE. Cost effectiveness of universal umbilical cord blood gas and lactate analysis in a tertiary level maternity unit. J Perinat Med. 2016;44(5):573–584.
- White CR, Doherty DA, Henderson JJ, Kohan R, Newnham JP, Pennell CE. Benefits of introducing universal umbilical cord blood gas and lactate analysis into an obstetric unit. Aust N Z J Obstet Gynaecol. 2010;50(4):318–328.
- Yeomans ER, Hauth JC, Gilstrap LC III, Strickland DM. Umbilical cord pH, Pco2, and bicarbonate following uncomplicated term vaginal deliveries. Am J Obstet Gynecol. 1985;151(6):798–800.
- Wiberg N, Källén K, Olofsson P. Base deficit estimation in umbilical cord blood is influenced by gestational age, choice of fetal fluid compartment, and algorithm for calculation. Am J Obstet Gynecol. 2006;195(6):1651–1656.
- Ross MG, Gala R. Use of umbilical artery base excess: algorithm for the timing of hypoxic injury. Am J Obstet Gynecol. 2002;187(1):1–9.
- Executive summary: Neonatal encephalopathy and neurologic outcome, second edition. Report of the American College of Obstetricians and Gynecologists’ Task Force on Neonatal Encephalopathy. Obstet Gynecol. 2014;123(4):896-901.
- Hamel MS, Anderson BL, Rouse DJ. Oxygen for intrauterine resuscitation: of unproved benefit and potentially harmful. Am J Obstet Gynecol. 2014;211(2):124–127.
- Owen P, Farrell TA, Steyn W. Umbilical cord blood gas analysis; a comparison of two simple methods of sample storage. Early Hum Dev. 1995;42(1):67–71.
- Armstrong L, Stenson B. Effect of delayed sampling on umbilical cord arterial and venous lactate and blood gases in clamped and unclamped vessels. Arch Dis Child Fetal Neonatal Ed. 2006;91(5):F342–F345.
- White CR, Mok T, Doherty DA, Henderson JJ, Newnham JP, Pennell CE. The effect of time, temperature and storage device on umbilical cord blood gas and lactate measurement: a randomized controlled trial. J Matern Fetal Neonatal Med. 2012;25(6):587–594.
- Knowles TP, Mullin RA, Hunter JA, Douce FH. Effects of syringe material, sample storage time, and temperature on blood gases and oxygen saturation in arterialized human blood samples. Respir Care. 2006;51(7):732–736.
- Nodwell A, Carmichael L, Ross M, Richardson B. Placental compared with umbilical cord blood to assess fetal blood gas and acid-base status. Obstet Gynecol. 2005;105(1):129–138.
- American College of Obstetricians and Gynecologists. ACOG Committee Opinion No. 684. Delayed umbilical cord clamping after birth. Obstet Gynecol. 2017;129(1):e5–e10.
- De Paco C, Florido J, Garrido MC, Prados S, Navarrete L. Umbilical cord blood acid-base and gas analysis after early versus delayed cord clamping in neonates at term. Arch Gynecol Obstet. 2011;283(5):1011–1014.
- Valero J, Desantes D, Perales-Puchalt A, Rubio J, Diago Almela VJ, Perales A. Effect of delayed umbilical cord clamping on blood gas analysis. Eur J Obstet Gynecol Reprod Biol. 2012;162(1): 21–23.
- Andersson O, Hellström-Westas L, Andersson D, Clausen J, Domellöf M. Effects of delayed compared with early umbilical cord clamping on maternal postpartum hemorrhage and cord blood gas sampling: a randomized trial. Acta Obstet Gynecol Scand. 2013;92(5):567–574.
- Wiberg N, Källén K, Olofsson P. Delayed umbilical cord clamping at birth has effects on arterial and venous blood gases and lactate concentrations. BJOG. 2008;115(6):697–703.
- Mokarami P, Wiberg N, Olofsson P. Hidden acidosis: an explanation of acid-base and lactate changes occurring in umbilical cord blood after delayed sampling. BJOG. 2013;120(8):996–1002.
- Ross MG, Gala R. Use of umbilical artery base excess: algorithm for the timing of hypoxic injury. Am J Obstet Gynecol. 2002;187(1):1–9.
- Uccella S, Cromi A, Colombo G, et al. Prediction of fetal base excess values at birth using an algorithm to interpret fetal heart rate tracings: a retrospective validation. BJOG. 2012;119(13):1657–1664.
- Uccella S, Cromi A, Colombo GF, et al. Interobserver reliability to interpret intrapartum electronic fetal heart rate monitoring: does a standardized algorithm improve agreement among clinicians? J Obstet Gynaecol. 2015;35(3):241–245.
- White CR, Doherty DA, Cannon JW, Kohan R, Newnham JP, Pennell CE. Cost effectiveness of universal umbilical cord blood gas and lactate analysis in a tertiary level maternity unit. J Perinat Med. 2016;44(5):573–584.
- White CR, Doherty DA, Henderson JJ, Kohan R, Newnham JP, Pennell CE. Benefits of introducing universal umbilical cord blood gas and lactate analysis into an obstetric unit. Aust N Z J Obstet Gynaecol. 2010;50(4):318–328.
- Yeomans ER, Hauth JC, Gilstrap LC III, Strickland DM. Umbilical cord pH, Pco2, and bicarbonate following uncomplicated term vaginal deliveries. Am J Obstet Gynecol. 1985;151(6):798–800.
- Wiberg N, Källén K, Olofsson P. Base deficit estimation in umbilical cord blood is influenced by gestational age, choice of fetal fluid compartment, and algorithm for calculation. Am J Obstet Gynecol. 2006;195(6):1651–1656.
- Ross MG, Gala R. Use of umbilical artery base excess: algorithm for the timing of hypoxic injury. Am J Obstet Gynecol. 2002;187(1):1–9.
- Executive summary: Neonatal encephalopathy and neurologic outcome, second edition. Report of the American College of Obstetricians and Gynecologists’ Task Force on Neonatal Encephalopathy. Obstet Gynecol. 2014;123(4):896-901.
- Hamel MS, Anderson BL, Rouse DJ. Oxygen for intrauterine resuscitation: of unproved benefit and potentially harmful. Am J Obstet Gynecol. 2014;211(2):124–127.
- Owen P, Farrell TA, Steyn W. Umbilical cord blood gas analysis; a comparison of two simple methods of sample storage. Early Hum Dev. 1995;42(1):67–71.
- Armstrong L, Stenson B. Effect of delayed sampling on umbilical cord arterial and venous lactate and blood gases in clamped and unclamped vessels. Arch Dis Child Fetal Neonatal Ed. 2006;91(5):F342–F345.
- White CR, Mok T, Doherty DA, Henderson JJ, Newnham JP, Pennell CE. The effect of time, temperature and storage device on umbilical cord blood gas and lactate measurement: a randomized controlled trial. J Matern Fetal Neonatal Med. 2012;25(6):587–594.
- Knowles TP, Mullin RA, Hunter JA, Douce FH. Effects of syringe material, sample storage time, and temperature on blood gases and oxygen saturation in arterialized human blood samples. Respir Care. 2006;51(7):732–736.
- Nodwell A, Carmichael L, Ross M, Richardson B. Placental compared with umbilical cord blood to assess fetal blood gas and acid-base status. Obstet Gynecol. 2005;105(1):129–138.
- American College of Obstetricians and Gynecologists. ACOG Committee Opinion No. 684. Delayed umbilical cord clamping after birth. Obstet Gynecol. 2017;129(1):e5–e10.
- De Paco C, Florido J, Garrido MC, Prados S, Navarrete L. Umbilical cord blood acid-base and gas analysis after early versus delayed cord clamping in neonates at term. Arch Gynecol Obstet. 2011;283(5):1011–1014.
- Valero J, Desantes D, Perales-Puchalt A, Rubio J, Diago Almela VJ, Perales A. Effect of delayed umbilical cord clamping on blood gas analysis. Eur J Obstet Gynecol Reprod Biol. 2012;162(1): 21–23.
- Andersson O, Hellström-Westas L, Andersson D, Clausen J, Domellöf M. Effects of delayed compared with early umbilical cord clamping on maternal postpartum hemorrhage and cord blood gas sampling: a randomized trial. Acta Obstet Gynecol Scand. 2013;92(5):567–574.
- Wiberg N, Källén K, Olofsson P. Delayed umbilical cord clamping at birth has effects on arterial and venous blood gases and lactate concentrations. BJOG. 2008;115(6):697–703.
- Mokarami P, Wiberg N, Olofsson P. Hidden acidosis: an explanation of acid-base and lactate changes occurring in umbilical cord blood after delayed sampling. BJOG. 2013;120(8):996–1002.
Humira Pen topped per-person drug spending in 2016
Humira Pen (adalimumab) was the most expensive drug in 2016 when ranked by spending per person, according to pharmacy benefits manager Express Scripts.
Total spending per person with employer-sponsored insurance was $45.11 last year for Humira Pen, which is indicated for rheumatoid arthritis, psoriatic arthritis, ankylosing spondylitis, Crohn’s disease, ulcerative colitis, and plaque psoriasis. Next in spending per person was Enbrel (etanercept) – another drug for arthritis, psoriatic arthritis, ankylosing spondylitis, and psoriasis – at $26.82, followed by the diabetes drug Lantus (insulin glargine) and two multiple sclerosis drugs: Tecfidera (dimethyl fumarate) and Copaxone (glatiramer), Express Scripts said in its “2016 Drug Trend Report.”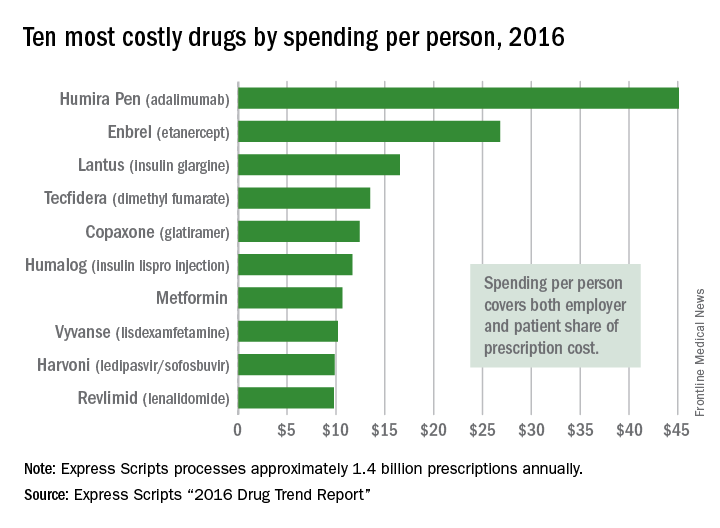
Humira Pen had the next-largest increase from 2015 – a mere 28% – while the hepatitis C drug Harvoni (ledipasvir/sofisbuvir) had the largest decrease in per-person spending among the top 10, dropping 54%, the report noted.
Express Scripts processes approximately 1.4 billion prescriptions annually for 85 million insured members from 3,000 client companies.
Humira Pen (adalimumab) was the most expensive drug in 2016 when ranked by spending per person, according to pharmacy benefits manager Express Scripts.
Total spending per person with employer-sponsored insurance was $45.11 last year for Humira Pen, which is indicated for rheumatoid arthritis, psoriatic arthritis, ankylosing spondylitis, Crohn’s disease, ulcerative colitis, and plaque psoriasis. Next in spending per person was Enbrel (etanercept) – another drug for arthritis, psoriatic arthritis, ankylosing spondylitis, and psoriasis – at $26.82, followed by the diabetes drug Lantus (insulin glargine) and two multiple sclerosis drugs: Tecfidera (dimethyl fumarate) and Copaxone (glatiramer), Express Scripts said in its “2016 Drug Trend Report.”
Humira Pen had the next-largest increase from 2015 – a mere 28% – while the hepatitis C drug Harvoni (ledipasvir/sofisbuvir) had the largest decrease in per-person spending among the top 10, dropping 54%, the report noted.
Express Scripts processes approximately 1.4 billion prescriptions annually for 85 million insured members from 3,000 client companies.
Humira Pen (adalimumab) was the most expensive drug in 2016 when ranked by spending per person, according to pharmacy benefits manager Express Scripts.
Total spending per person with employer-sponsored insurance was $45.11 last year for Humira Pen, which is indicated for rheumatoid arthritis, psoriatic arthritis, ankylosing spondylitis, Crohn’s disease, ulcerative colitis, and plaque psoriasis. Next in spending per person was Enbrel (etanercept) – another drug for arthritis, psoriatic arthritis, ankylosing spondylitis, and psoriasis – at $26.82, followed by the diabetes drug Lantus (insulin glargine) and two multiple sclerosis drugs: Tecfidera (dimethyl fumarate) and Copaxone (glatiramer), Express Scripts said in its “2016 Drug Trend Report.”
Humira Pen had the next-largest increase from 2015 – a mere 28% – while the hepatitis C drug Harvoni (ledipasvir/sofisbuvir) had the largest decrease in per-person spending among the top 10, dropping 54%, the report noted.
Express Scripts processes approximately 1.4 billion prescriptions annually for 85 million insured members from 3,000 client companies.
2017 Update on ovarian cancer
In 2017, an estimated 22,240 women will be diagnosed with ovarian cancer, and 14,080 women will die of the disease.1 The high mortality associated with ovarian cancer is due largely to the inability to detect the disease early and the lack of effective therapeutics for women with recurrent disease. In this Update, we review important advances in the diagnosis and treatment of ovarian cancer.
Development of an effective screening tool for women at average risk has been an elusive challenge. The United Kingdom Collaborative Trial of Ovarian Cancer Screening (UKCTOCS) examined the efficacy of transvaginal ultrasound and cancer antigen 125 (CA 125) monitoring for ovarian cancer in a large cohort of women.
For women diagnosed with ovarian cancer, treatment paradigms for the initial management of the disease have shifted dramatically. Based on data from multiple randomized controlled trials, neoadjuvant chemotherapy (NACT) is being used more frequently. The American Society of Clinical Oncology and the Society of Gynecologic Oncology developed consensus recommendations for the appropriate use of NACT and primary cytoreductive surgery for women with ovarian cancer.
Finally, all of oncology has moved toward incorporating molecularly targeted therapeutics directed toward individual genetic abnormalities in tumors, so-called precision medicine. In ovarian cancer, poly(adenosine diphosphate [ADP]–ribose) polymerase (PARP) has emerged as an important target, particularly for women with BRCA gene pathway mutations. We describe a recently published randomized controlled trial of the PARP inhibitor niraparib.
Read about ovarian cancer screening tests
Is CA 125 or ultrasound screening appropriate for the general population?
Jacobs IJ, Menon U, Ryan A, et al. Ovarian cancer screening and mortality in the UK Collaborative Trial of Ovarian Cancer Screening (UKCTOCS): a randomised controlled trial. Lancet. 2016;387(10022):945-956.
In the United States, the overall ovarian cancer 5-year survival rate is 46.2%, resulting in more than 14,000 deaths annually.2 The poor prognosis associated with this malignancy is largely attributable to the fact that almost 75% of women have stage III or stage IV disease at the time of diagnosis.2 Ovarian cancer is usually associated with vague, nonspecific symptoms as it progresses, which contributes to delayed diagnosis and increased mortality.
Multiple studies have examined pelvic ultrasonography and tumor markers, such as CA 125, as possible screening tools to increase early detection in asymptomatic women. However, neither modality alone or in combination has sufficient sensitivity or specificity to recommend it for use in the general population.3,4 Nevertheless, the search for an appropriate screening tool continues, and the UKCTOCS trial results have reinvigorated this discussion.5
The UKCTOCS findings
The UKCTOCS was a multicenter, randomized controlled trial in the United Kingdom in which researchers allocated 202,638 women aged 50 to 74 years to 1 of 3 groups: annual multimodal screening (MMS) with serum CA 125 interpreted with the use of the risk of ovarian cancer algorithm, annual transvaginal ultrasound screening (USS), or no screening. The median follow-up was more than 11 years.
The investigators found that equivalent rates of ovarian cancer were diagnosed in each group: 0.7% in the MMS group, 0.6% in the USS group, and 0.6% in the no-screening group. Overall, there was no significant reduction in the mortality rate from ovarian cancer in either of the 2 screening groups compared with the no-screening group.5
An important subset discovery
However, in a prespecified subset analysis excluding "prevalent cases" (women with ovarian cancer thought to be present prior to randomization and subsequent screening), ovarian cancer mortality was significantly lower in the MMS group compared with the no-screening group (P = .021). Compared with no screening, MMS was associated with a 20% reduction in mortality rate from ovarian cancer over time, with the most pronounced effects occurring at years 7 to 14 of follow-up, suggesting the possible increased effectiveness of screening over time.5
Related article:
Can CA 125 screening reduce mortality from ovarian cancer?
Concordance with other screening trials
While impressive in study magnitude and scope, the UKCTOCS results did not demonstrate a significant mortality benefit associated with MMS or USS when compared with no screening. Although the screening complications were low (<1% in both screening groups), the authors did note a false-positive surgery rate of 14 per 10,000 screens for the MMS group and 50 per 10,000 screens for the USS group. Based on the performance of screening in this trial, 641 women would need to be screened annually using MMS for 14 years to prevent 1 ovarian cancer death.
Like the UKCTOCS, the ovarian cancer-screening arm of the Prostate, Lung, Colorectal and Ovarian (PLCO) Cancer Screening Trial in the United States was also unable to demonstrate a reduction in mortality rate with screening with CA 125 and transvaginal ultrasound. Importantly, more than one-third of women with a false-positive screen underwent surgery and 15% of them experienced a major complication.6 Based on these findings, the US Preventive Services Task Force grades screening for ovarian cancer as D, suggesting that the harms of screening may outweigh the benefits.7
While screening for ovarian cancer remains an important need, there is currently no evidence to suggest that serum tumor marker or ultrasound screening is appropriate in the general population. Studies using more specific screening tests or strategies targeted to higher-risk women are ongoing.
Read about patient selection for neoadjuvant chemotherapy
New clinical practice guideline advises neoadjuvant chemotherapy for certain women with ovarian cancer
Wright AA, Bohlke K, Armstrong DK, et al. Neoadjuvant chemotherapy for newly diagnosed, advanced ovarian cancer: Society of Gynecologic Oncology and American Society of Clinical Oncology clinical practice guideline. J Clin Oncol. 2016;34(28):3460-3473.
It has long been held as a central dogma that primary cytoreductive surgery (PCS) is the preferred initial treatment for women with newly diagnosed ovarian cancer.8 However, PCS is associated with substantial morbidity, and the ability to achieve optimal cytoreduction (<1 cm of residual disease), an important prognostic factor, is often compromised in women with significant tumor burden.9,10
Neoadjuvant chemotherapy, in which chemotherapy is administered prior to surgical cytoreduction, challenges the traditional treatment paradigm for advanced-stage ovarian cancer. Several randomized controlled trials have reported equivalent survival for primary surgical cytoreduction and NACT. Importantly, women who received NACT had fewer complications and were more likely to have optimal cytoreduction at the time of surgery.11,12 These studies have limitations, however, and the role of NACT remains uncertain.
To help guide clinicians, the Society of Gynecologic Oncology and the American Society of Clinical Oncology convened an expert panel to provide recommendations and guidance on the evaluation of women for and the use of NACT in the setting of advanced ovarian cancer.13
Related article:
2015 Update on cancer
Recommendation: Clinical evaluation and patient selection
Strong clinical evidence supports that all women with suspected stage IIIC or stage IV ovarian cancer should be evaluated by a gynecologic oncologist prior to the initiation of therapy. The evaluation should include at least a computed tomography scan of the chest, abdomen, and pelvis to assess the extent of disease and resectability. A preoperative risk assessment should be performed to assess risk factors for increased morbidity and mortality.
Women who have a high perioperative risk profile or a low likelihood of achieving cytoreduction to 1 cm or less of residual tumor should receive NACT. Prior to the initiation of NACT, histologic confirmation of ovarian cancer should be obtained.13
Outcomes for neoadjuvant chemotherapy versus primary cytoreduction
Four phase 3 randomized controlled trials (EORTC 55971, CHORUS, JCOG0602, and SCORPION) suggest that NACT is noninferior to PCS with regard to progression-free survival and overall survival. NACT is associated with less perioperative and postoperative morbidity and mortality and is associated with shorter hospital stays.
To date, complete data are available only from the EORTC and CHORUS trials, which both demonstrated similar progression-free survival and overall survival for NACT and PCS. Critics have noted, however, that both trials have shorter median overall survival for the PCS groups than were previously reported in other phase 3 studies in the United States, suggesting the possibility of different patient populations or less aggressive "surgical effort." Thus, PCS remains the preferred management strategy for women with advanced-stage ovarian cancer in whom there is a high likelihood of optimal cytoreduction.13
Recommendation: Use of neoadjuvant chemotherapy
Patients who are appropriate candidates for NACT should be treated with a platinum and taxane doublet and should receive interval cytoreduction following 3 to 4 cycles of therapy if a favorable response is noted. Patients whose disease progresses despite NACT have a poor prognosis, and there is little role for surgical treatment with the exception of palliative purposes.13
Neoadjuvant chemotherapy is a noninferior and appropriate treatment option for women who are poor surgical candidates or who have a low likelihood of optimal cytoreduction. When optimal cytoreduction is possible, however, PCS is preferred (see FIGURE). The data on the efficacy of NACT for ovarian cancer have led to increased use of this treatment in the United States.
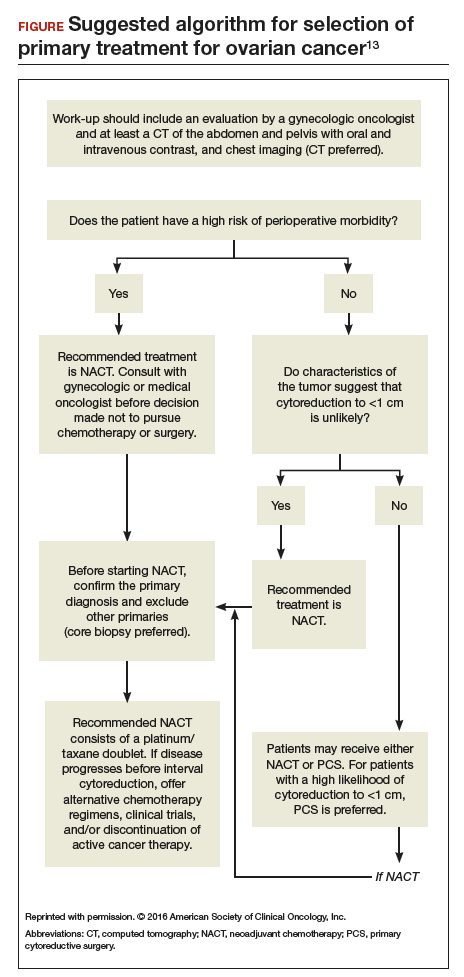
Read about a new PARP inhibitor for maintenance therapy
Niraparib is promising as maintenance therapy in ovarian cancer
Mirza MR, Monk BJ, Herrstedt J, et al; for the ENGOT-OV16/NOVA Investigators. Niraparib maintenance therapy in platinum-sensitive, recurrent ovarian cancer. N Engl J Med. 2016;375(22):2154-2164.
Approximately 85% of women with ovarian cancer will develop recurrent disease. Women with ovarian cancer are commonly treated with a range of antineoplastic agents over the course of their lifetime. As such, there is a great need for additional active therapeutic agents in this setting. Recently, substantial effort has been directed toward "precision" or "personalized medicine" in oncology.
Precision medicine, targeted therapies in oncology
Precision medicine refers to the customization of medical therapy based on the genetic characterization of the individual patient or the molecular profile of the patient's tumor. As a result of large-scale molecular profiling from projects such as the International Cancer Genome Consortium and The Cancer Genome Atlas, an abundance of molecular data has been generated through the characterization of multiple tumor types. This has led to the discovery of key cancer drivers, alterations, and specific molecular profiles that have distinct prognostic and treatment implications. These data, in combination with the commercial availability of molecular profiling tests, has made precision medicine a reality for women with ovarian cancer.
This wealth of new information has led to development of targeted therapeutics that block the growth and spread of cancer by acting on specific molecules or molecular pathways. Targeted therapies approved for cancer treatment include hormonal therapies, signal transduction inhibitors, gene expression modulators, apoptosis inducers, angiogenesis inhibitors, and immunotherapies.14
How PARP inhibitors work
PARP inhibitors are a class of agents that are emerging as important therapies for ovarian cancer. These agents block the nuclear protein PARP, which functions to detect and repair single-strand DNA breaks with the resulting accumulation of double-stranded DNA breaks.15 In the setting of DNA damage, the homologous recombination repair pathway is activated for repair. However, homologous recombination deficiencies (HRD) can arise as a result of BRCA1 or BRCA2 mutations or BRCA-independent pathways, which effectively disable this DNA repair pathway. As a result, when PARP inhibitors are used in patients with HRD, the cell cannot repair double-stranded DNA breaks and this leads to "synthetic lethality."16
Understanding this molecular mechanism of PARP inhibitors as well as the frequent abnormalities in the BRCA genes and HRD pathways in ovarian cancer has provided an important potential therapeutic target in ovarian cancer. A number of PARP inhibitors are now commercially available and are undergoing testing in ovarian cancer.
Related article:
Is a minimally invasive approach to hysterectomy for Gyn cancer utilized equally in all racial and income groups?
Niraparib for ovarian cancer
In a randomized, double-blind, phase 3 trial by Mizra and colleagues, 553 women with platinum-sensitive recurrent ovarian cancer who responded to therapy were divided according to the presence or absence of a germline BRCA (gBRCA) mutation and randomly assigned to niraparib 300 mg or placebo once daily. Women in the niraparib group had a significantly longer median duration of progression-free survival than did those in the placebo group. This was most pronounced in women in the gBRCA cohort (21.0 vs 5.5 months). Importantly, niraparib was associated with improved progression-free survival in HRD-positive patients without gBRCA mutations (12.9 vs 3.8 months) as well as in the HRD-negative subgroup (6.9 vs 3.8 months).17
Overall, niraparib was well tolerated. About 15% of women discontinued the drug due to toxicity. Significant (grade 3 or 4) adverse events were seen in three-quarters of women treated with niraparib, and they most commonly consisted of hematologic toxicities. Patient-reported outcomes were similar for both groups, indicating no significant effect from niraparib on quality of life.17
This study's results suggest that niraparib has clinical activity against ovarian cancer. Importantly, niraparib was active in women with gBRCA mutations, in those with HRD without a gBRCA mutation, and potentially in women without HRD. If approved by the US Food and Drug Administration, niraparib will join olaparib and rucaparib as a newly approved therapeutic agent for ovarian cancer. This study provides important evidence that suggests niraparib maintenance therapy may be an efficacious and important addition to the treatment armamentarium for platinum-sensitive ovarian cancer.
Share your thoughts! Send your Letter to the Editor to [email protected]. Please include your name and the city and state in which you practice.
- Siegel RL, Miller KD, Jemal A. Cancer statistics, 2017. CA Cancer J Clin. 2017;67(1):7–30.
- National Cancer Institute Surveillance, Epidemiology, and End Results Program. Cancer stat facts: ovarian cancer. https://seer.cancer.gov/statfacts/html/ovary.html. Accessed January 20, 2017.
- Jacobs I, Davies AP, Bridges J, et al. Prevalence screening for ovarian cancer in postmenopausal women by CA 125 measurement and ultrasonography. BMJ. 1993;306(6884):1030–1034.
- van Nagell JR Jr, Pavlik EJ. Ovarian cancer screening. Clin Obstet Gynecol. 2012;55:43–51.
- Jacobs IJ, Menon U, Ryan A, et al. Ovarian cancer screening and mortality in the UK Collaborative Trial of Ovarian Cancer Screening (UKCTOCS): a randomised controlled trial. Lancet. 2016;387(10022):945–956.
- Buys SS, Partridge E, Black A, et al; PLCO Project Team. Effect of screening on ovarian cancer mortality: the Prostate, Lung, Colorectal and Ovarian (PLCO) Cancer Screening randomized controlled trial. JAMA. 2011;305(22):2295–2303.
- Moyer VA, US Preventive Services Task Force. Screening for ovarian cancer: US Preventive Services Task Force reaffirmation recommendation statement. Ann Intern Med. 2012;157(12):900–904.
- Schorge JO, McCann C, Del Carmen MG. Surgical debulking of ovarian cancer: what difference does it make? Rev Obstet Gynecol. 2010;3(3):111–117.
- Hoskins WJ, McGuire WP, Brady MF, et al. The effect of diameter of largest residual disease on survival after primary cytoreductive surgery in patients with suboptimal residual epithelial ovarian carcinoma. Am J Obstet Gynecol. 1994;170(4):974–979; discussion 979–980.
- Bristow RE, Tomacruz RS, Armstrong DK, Trimble EL, Montz FJ. Survival effect of maximal cytoreductive surgery for advanced ovarian carcinoma during the platinum era: a meta-analysis. J Clin Oncol. 2002;20(5):1248–1259.
- Vergote I, Trope CG, Amant F, et al; European Organization for Research and Treatment of Cancer–Gynaecological Cancer Goup; NCIC Clinical Trials Group. Neoadjuvant chemotherapy or primary surgery in stage IIIC or IV ovarian cancer. N Engl J Med. 2010;363(10):943–953.
- Kehoe S, Hook J, Nankivell M, et al. Primary chemotherapy versus primary surgery for newly diagnosed advanced ovarian cancer (CHORUS): an open-label, randomised, controlled, non-inferiority trial. Lancet. 2015;386(9990):249–257.
- Wright AA, Bohlke K, Armstrong DK, et al. Neoadjuvant chemotherapy for newly diagnosed, advanced ovarian cancer: Society of Gynecologic Oncology and American Society of Clinical Oncology clinical practice guideline. J Clin Oncol. 2016;34(28):3460–3473.
- National Cancer Institute. Targeted cancer therapies. https://www.cancer.gov/about-cancer/treatment/types/targeted-therapies/targeted-therapies-fact-sheet. Updated April 25, 2014. Accessed January 21, 2017.
- Drean A, Lord CJ, Ashworth A. PARP inhibitor combination therapy. Crit Rev Oncol Hematol. 2016;108:73–85.
- Ledermann JA, El-Khouly F. PARP inhibitors in ovarian cancer: clinical evidence for informed treatment decisions. Br J Cancer. 2015;113(suppl 1):S10–S16.
- Mirza MR, Monk BJ, Herrstedt J, et al; ENGOT-OV16/NOVA Investigators. Niraparib maintenance therapy in platinum-sensitive, recurrent ovarian cancer. N Engl J Med. 2016;375(22):2154–2164.
In 2017, an estimated 22,240 women will be diagnosed with ovarian cancer, and 14,080 women will die of the disease.1 The high mortality associated with ovarian cancer is due largely to the inability to detect the disease early and the lack of effective therapeutics for women with recurrent disease. In this Update, we review important advances in the diagnosis and treatment of ovarian cancer.
Development of an effective screening tool for women at average risk has been an elusive challenge. The United Kingdom Collaborative Trial of Ovarian Cancer Screening (UKCTOCS) examined the efficacy of transvaginal ultrasound and cancer antigen 125 (CA 125) monitoring for ovarian cancer in a large cohort of women.
For women diagnosed with ovarian cancer, treatment paradigms for the initial management of the disease have shifted dramatically. Based on data from multiple randomized controlled trials, neoadjuvant chemotherapy (NACT) is being used more frequently. The American Society of Clinical Oncology and the Society of Gynecologic Oncology developed consensus recommendations for the appropriate use of NACT and primary cytoreductive surgery for women with ovarian cancer.
Finally, all of oncology has moved toward incorporating molecularly targeted therapeutics directed toward individual genetic abnormalities in tumors, so-called precision medicine. In ovarian cancer, poly(adenosine diphosphate [ADP]–ribose) polymerase (PARP) has emerged as an important target, particularly for women with BRCA gene pathway mutations. We describe a recently published randomized controlled trial of the PARP inhibitor niraparib.
Read about ovarian cancer screening tests
Is CA 125 or ultrasound screening appropriate for the general population?
Jacobs IJ, Menon U, Ryan A, et al. Ovarian cancer screening and mortality in the UK Collaborative Trial of Ovarian Cancer Screening (UKCTOCS): a randomised controlled trial. Lancet. 2016;387(10022):945-956.
In the United States, the overall ovarian cancer 5-year survival rate is 46.2%, resulting in more than 14,000 deaths annually.2 The poor prognosis associated with this malignancy is largely attributable to the fact that almost 75% of women have stage III or stage IV disease at the time of diagnosis.2 Ovarian cancer is usually associated with vague, nonspecific symptoms as it progresses, which contributes to delayed diagnosis and increased mortality.
Multiple studies have examined pelvic ultrasonography and tumor markers, such as CA 125, as possible screening tools to increase early detection in asymptomatic women. However, neither modality alone or in combination has sufficient sensitivity or specificity to recommend it for use in the general population.3,4 Nevertheless, the search for an appropriate screening tool continues, and the UKCTOCS trial results have reinvigorated this discussion.5

The UKCTOCS findings
The UKCTOCS was a multicenter, randomized controlled trial in the United Kingdom in which researchers allocated 202,638 women aged 50 to 74 years to 1 of 3 groups: annual multimodal screening (MMS) with serum CA 125 interpreted with the use of the risk of ovarian cancer algorithm, annual transvaginal ultrasound screening (USS), or no screening. The median follow-up was more than 11 years.
The investigators found that equivalent rates of ovarian cancer were diagnosed in each group: 0.7% in the MMS group, 0.6% in the USS group, and 0.6% in the no-screening group. Overall, there was no significant reduction in the mortality rate from ovarian cancer in either of the 2 screening groups compared with the no-screening group.5
An important subset discovery
However, in a prespecified subset analysis excluding "prevalent cases" (women with ovarian cancer thought to be present prior to randomization and subsequent screening), ovarian cancer mortality was significantly lower in the MMS group compared with the no-screening group (P = .021). Compared with no screening, MMS was associated with a 20% reduction in mortality rate from ovarian cancer over time, with the most pronounced effects occurring at years 7 to 14 of follow-up, suggesting the possible increased effectiveness of screening over time.5
Related article:
Can CA 125 screening reduce mortality from ovarian cancer?
Concordance with other screening trials
While impressive in study magnitude and scope, the UKCTOCS results did not demonstrate a significant mortality benefit associated with MMS or USS when compared with no screening. Although the screening complications were low (<1% in both screening groups), the authors did note a false-positive surgery rate of 14 per 10,000 screens for the MMS group and 50 per 10,000 screens for the USS group. Based on the performance of screening in this trial, 641 women would need to be screened annually using MMS for 14 years to prevent 1 ovarian cancer death.
Like the UKCTOCS, the ovarian cancer-screening arm of the Prostate, Lung, Colorectal and Ovarian (PLCO) Cancer Screening Trial in the United States was also unable to demonstrate a reduction in mortality rate with screening with CA 125 and transvaginal ultrasound. Importantly, more than one-third of women with a false-positive screen underwent surgery and 15% of them experienced a major complication.6 Based on these findings, the US Preventive Services Task Force grades screening for ovarian cancer as D, suggesting that the harms of screening may outweigh the benefits.7
While screening for ovarian cancer remains an important need, there is currently no evidence to suggest that serum tumor marker or ultrasound screening is appropriate in the general population. Studies using more specific screening tests or strategies targeted to higher-risk women are ongoing.
Read about patient selection for neoadjuvant chemotherapy
New clinical practice guideline advises neoadjuvant chemotherapy for certain women with ovarian cancer
Wright AA, Bohlke K, Armstrong DK, et al. Neoadjuvant chemotherapy for newly diagnosed, advanced ovarian cancer: Society of Gynecologic Oncology and American Society of Clinical Oncology clinical practice guideline. J Clin Oncol. 2016;34(28):3460-3473.
It has long been held as a central dogma that primary cytoreductive surgery (PCS) is the preferred initial treatment for women with newly diagnosed ovarian cancer.8 However, PCS is associated with substantial morbidity, and the ability to achieve optimal cytoreduction (<1 cm of residual disease), an important prognostic factor, is often compromised in women with significant tumor burden.9,10
Neoadjuvant chemotherapy, in which chemotherapy is administered prior to surgical cytoreduction, challenges the traditional treatment paradigm for advanced-stage ovarian cancer. Several randomized controlled trials have reported equivalent survival for primary surgical cytoreduction and NACT. Importantly, women who received NACT had fewer complications and were more likely to have optimal cytoreduction at the time of surgery.11,12 These studies have limitations, however, and the role of NACT remains uncertain.
To help guide clinicians, the Society of Gynecologic Oncology and the American Society of Clinical Oncology convened an expert panel to provide recommendations and guidance on the evaluation of women for and the use of NACT in the setting of advanced ovarian cancer.13
Related article:
2015 Update on cancer
Recommendation: Clinical evaluation and patient selection
Strong clinical evidence supports that all women with suspected stage IIIC or stage IV ovarian cancer should be evaluated by a gynecologic oncologist prior to the initiation of therapy. The evaluation should include at least a computed tomography scan of the chest, abdomen, and pelvis to assess the extent of disease and resectability. A preoperative risk assessment should be performed to assess risk factors for increased morbidity and mortality.
Women who have a high perioperative risk profile or a low likelihood of achieving cytoreduction to 1 cm or less of residual tumor should receive NACT. Prior to the initiation of NACT, histologic confirmation of ovarian cancer should be obtained.13
Outcomes for neoadjuvant chemotherapy versus primary cytoreduction
Four phase 3 randomized controlled trials (EORTC 55971, CHORUS, JCOG0602, and SCORPION) suggest that NACT is noninferior to PCS with regard to progression-free survival and overall survival. NACT is associated with less perioperative and postoperative morbidity and mortality and is associated with shorter hospital stays.
To date, complete data are available only from the EORTC and CHORUS trials, which both demonstrated similar progression-free survival and overall survival for NACT and PCS. Critics have noted, however, that both trials have shorter median overall survival for the PCS groups than were previously reported in other phase 3 studies in the United States, suggesting the possibility of different patient populations or less aggressive "surgical effort." Thus, PCS remains the preferred management strategy for women with advanced-stage ovarian cancer in whom there is a high likelihood of optimal cytoreduction.13
Recommendation: Use of neoadjuvant chemotherapy
Patients who are appropriate candidates for NACT should be treated with a platinum and taxane doublet and should receive interval cytoreduction following 3 to 4 cycles of therapy if a favorable response is noted. Patients whose disease progresses despite NACT have a poor prognosis, and there is little role for surgical treatment with the exception of palliative purposes.13
Neoadjuvant chemotherapy is a noninferior and appropriate treatment option for women who are poor surgical candidates or who have a low likelihood of optimal cytoreduction. When optimal cytoreduction is possible, however, PCS is preferred (see FIGURE). The data on the efficacy of NACT for ovarian cancer have led to increased use of this treatment in the United States.

Read about a new PARP inhibitor for maintenance therapy
Niraparib is promising as maintenance therapy in ovarian cancer
Mirza MR, Monk BJ, Herrstedt J, et al; for the ENGOT-OV16/NOVA Investigators. Niraparib maintenance therapy in platinum-sensitive, recurrent ovarian cancer. N Engl J Med. 2016;375(22):2154-2164.
Approximately 85% of women with ovarian cancer will develop recurrent disease. Women with ovarian cancer are commonly treated with a range of antineoplastic agents over the course of their lifetime. As such, there is a great need for additional active therapeutic agents in this setting. Recently, substantial effort has been directed toward "precision" or "personalized medicine" in oncology.
Precision medicine, targeted therapies in oncology
Precision medicine refers to the customization of medical therapy based on the genetic characterization of the individual patient or the molecular profile of the patient's tumor. As a result of large-scale molecular profiling from projects such as the International Cancer Genome Consortium and The Cancer Genome Atlas, an abundance of molecular data has been generated through the characterization of multiple tumor types. This has led to the discovery of key cancer drivers, alterations, and specific molecular profiles that have distinct prognostic and treatment implications. These data, in combination with the commercial availability of molecular profiling tests, has made precision medicine a reality for women with ovarian cancer.
This wealth of new information has led to development of targeted therapeutics that block the growth and spread of cancer by acting on specific molecules or molecular pathways. Targeted therapies approved for cancer treatment include hormonal therapies, signal transduction inhibitors, gene expression modulators, apoptosis inducers, angiogenesis inhibitors, and immunotherapies.14
How PARP inhibitors work
PARP inhibitors are a class of agents that are emerging as important therapies for ovarian cancer. These agents block the nuclear protein PARP, which functions to detect and repair single-strand DNA breaks with the resulting accumulation of double-stranded DNA breaks.15 In the setting of DNA damage, the homologous recombination repair pathway is activated for repair. However, homologous recombination deficiencies (HRD) can arise as a result of BRCA1 or BRCA2 mutations or BRCA-independent pathways, which effectively disable this DNA repair pathway. As a result, when PARP inhibitors are used in patients with HRD, the cell cannot repair double-stranded DNA breaks and this leads to "synthetic lethality."16
Understanding this molecular mechanism of PARP inhibitors as well as the frequent abnormalities in the BRCA genes and HRD pathways in ovarian cancer has provided an important potential therapeutic target in ovarian cancer. A number of PARP inhibitors are now commercially available and are undergoing testing in ovarian cancer.
Related article:
Is a minimally invasive approach to hysterectomy for Gyn cancer utilized equally in all racial and income groups?
Niraparib for ovarian cancer
In a randomized, double-blind, phase 3 trial by Mizra and colleagues, 553 women with platinum-sensitive recurrent ovarian cancer who responded to therapy were divided according to the presence or absence of a germline BRCA (gBRCA) mutation and randomly assigned to niraparib 300 mg or placebo once daily. Women in the niraparib group had a significantly longer median duration of progression-free survival than did those in the placebo group. This was most pronounced in women in the gBRCA cohort (21.0 vs 5.5 months). Importantly, niraparib was associated with improved progression-free survival in HRD-positive patients without gBRCA mutations (12.9 vs 3.8 months) as well as in the HRD-negative subgroup (6.9 vs 3.8 months).17
Overall, niraparib was well tolerated. About 15% of women discontinued the drug due to toxicity. Significant (grade 3 or 4) adverse events were seen in three-quarters of women treated with niraparib, and they most commonly consisted of hematologic toxicities. Patient-reported outcomes were similar for both groups, indicating no significant effect from niraparib on quality of life.17
This study's results suggest that niraparib has clinical activity against ovarian cancer. Importantly, niraparib was active in women with gBRCA mutations, in those with HRD without a gBRCA mutation, and potentially in women without HRD. If approved by the US Food and Drug Administration, niraparib will join olaparib and rucaparib as a newly approved therapeutic agent for ovarian cancer. This study provides important evidence that suggests niraparib maintenance therapy may be an efficacious and important addition to the treatment armamentarium for platinum-sensitive ovarian cancer.
Share your thoughts! Send your Letter to the Editor to [email protected]. Please include your name and the city and state in which you practice.
In 2017, an estimated 22,240 women will be diagnosed with ovarian cancer, and 14,080 women will die of the disease.1 The high mortality associated with ovarian cancer is due largely to the inability to detect the disease early and the lack of effective therapeutics for women with recurrent disease. In this Update, we review important advances in the diagnosis and treatment of ovarian cancer.
Development of an effective screening tool for women at average risk has been an elusive challenge. The United Kingdom Collaborative Trial of Ovarian Cancer Screening (UKCTOCS) examined the efficacy of transvaginal ultrasound and cancer antigen 125 (CA 125) monitoring for ovarian cancer in a large cohort of women.
For women diagnosed with ovarian cancer, treatment paradigms for the initial management of the disease have shifted dramatically. Based on data from multiple randomized controlled trials, neoadjuvant chemotherapy (NACT) is being used more frequently. The American Society of Clinical Oncology and the Society of Gynecologic Oncology developed consensus recommendations for the appropriate use of NACT and primary cytoreductive surgery for women with ovarian cancer.
Finally, all of oncology has moved toward incorporating molecularly targeted therapeutics directed toward individual genetic abnormalities in tumors, so-called precision medicine. In ovarian cancer, poly(adenosine diphosphate [ADP]–ribose) polymerase (PARP) has emerged as an important target, particularly for women with BRCA gene pathway mutations. We describe a recently published randomized controlled trial of the PARP inhibitor niraparib.
Read about ovarian cancer screening tests
Is CA 125 or ultrasound screening appropriate for the general population?
Jacobs IJ, Menon U, Ryan A, et al. Ovarian cancer screening and mortality in the UK Collaborative Trial of Ovarian Cancer Screening (UKCTOCS): a randomised controlled trial. Lancet. 2016;387(10022):945-956.
In the United States, the overall ovarian cancer 5-year survival rate is 46.2%, resulting in more than 14,000 deaths annually.2 The poor prognosis associated with this malignancy is largely attributable to the fact that almost 75% of women have stage III or stage IV disease at the time of diagnosis.2 Ovarian cancer is usually associated with vague, nonspecific symptoms as it progresses, which contributes to delayed diagnosis and increased mortality.
Multiple studies have examined pelvic ultrasonography and tumor markers, such as CA 125, as possible screening tools to increase early detection in asymptomatic women. However, neither modality alone or in combination has sufficient sensitivity or specificity to recommend it for use in the general population.3,4 Nevertheless, the search for an appropriate screening tool continues, and the UKCTOCS trial results have reinvigorated this discussion.5

The UKCTOCS findings
The UKCTOCS was a multicenter, randomized controlled trial in the United Kingdom in which researchers allocated 202,638 women aged 50 to 74 years to 1 of 3 groups: annual multimodal screening (MMS) with serum CA 125 interpreted with the use of the risk of ovarian cancer algorithm, annual transvaginal ultrasound screening (USS), or no screening. The median follow-up was more than 11 years.
The investigators found that equivalent rates of ovarian cancer were diagnosed in each group: 0.7% in the MMS group, 0.6% in the USS group, and 0.6% in the no-screening group. Overall, there was no significant reduction in the mortality rate from ovarian cancer in either of the 2 screening groups compared with the no-screening group.5
An important subset discovery
However, in a prespecified subset analysis excluding "prevalent cases" (women with ovarian cancer thought to be present prior to randomization and subsequent screening), ovarian cancer mortality was significantly lower in the MMS group compared with the no-screening group (P = .021). Compared with no screening, MMS was associated with a 20% reduction in mortality rate from ovarian cancer over time, with the most pronounced effects occurring at years 7 to 14 of follow-up, suggesting the possible increased effectiveness of screening over time.5
Related article:
Can CA 125 screening reduce mortality from ovarian cancer?
Concordance with other screening trials
While impressive in study magnitude and scope, the UKCTOCS results did not demonstrate a significant mortality benefit associated with MMS or USS when compared with no screening. Although the screening complications were low (<1% in both screening groups), the authors did note a false-positive surgery rate of 14 per 10,000 screens for the MMS group and 50 per 10,000 screens for the USS group. Based on the performance of screening in this trial, 641 women would need to be screened annually using MMS for 14 years to prevent 1 ovarian cancer death.
Like the UKCTOCS, the ovarian cancer-screening arm of the Prostate, Lung, Colorectal and Ovarian (PLCO) Cancer Screening Trial in the United States was also unable to demonstrate a reduction in mortality rate with screening with CA 125 and transvaginal ultrasound. Importantly, more than one-third of women with a false-positive screen underwent surgery and 15% of them experienced a major complication.6 Based on these findings, the US Preventive Services Task Force grades screening for ovarian cancer as D, suggesting that the harms of screening may outweigh the benefits.7
While screening for ovarian cancer remains an important need, there is currently no evidence to suggest that serum tumor marker or ultrasound screening is appropriate in the general population. Studies using more specific screening tests or strategies targeted to higher-risk women are ongoing.
Read about patient selection for neoadjuvant chemotherapy
New clinical practice guideline advises neoadjuvant chemotherapy for certain women with ovarian cancer
Wright AA, Bohlke K, Armstrong DK, et al. Neoadjuvant chemotherapy for newly diagnosed, advanced ovarian cancer: Society of Gynecologic Oncology and American Society of Clinical Oncology clinical practice guideline. J Clin Oncol. 2016;34(28):3460-3473.
It has long been held as a central dogma that primary cytoreductive surgery (PCS) is the preferred initial treatment for women with newly diagnosed ovarian cancer.8 However, PCS is associated with substantial morbidity, and the ability to achieve optimal cytoreduction (<1 cm of residual disease), an important prognostic factor, is often compromised in women with significant tumor burden.9,10
Neoadjuvant chemotherapy, in which chemotherapy is administered prior to surgical cytoreduction, challenges the traditional treatment paradigm for advanced-stage ovarian cancer. Several randomized controlled trials have reported equivalent survival for primary surgical cytoreduction and NACT. Importantly, women who received NACT had fewer complications and were more likely to have optimal cytoreduction at the time of surgery.11,12 These studies have limitations, however, and the role of NACT remains uncertain.
To help guide clinicians, the Society of Gynecologic Oncology and the American Society of Clinical Oncology convened an expert panel to provide recommendations and guidance on the evaluation of women for and the use of NACT in the setting of advanced ovarian cancer.13
Related article:
2015 Update on cancer
Recommendation: Clinical evaluation and patient selection
Strong clinical evidence supports that all women with suspected stage IIIC or stage IV ovarian cancer should be evaluated by a gynecologic oncologist prior to the initiation of therapy. The evaluation should include at least a computed tomography scan of the chest, abdomen, and pelvis to assess the extent of disease and resectability. A preoperative risk assessment should be performed to assess risk factors for increased morbidity and mortality.
Women who have a high perioperative risk profile or a low likelihood of achieving cytoreduction to 1 cm or less of residual tumor should receive NACT. Prior to the initiation of NACT, histologic confirmation of ovarian cancer should be obtained.13
Outcomes for neoadjuvant chemotherapy versus primary cytoreduction
Four phase 3 randomized controlled trials (EORTC 55971, CHORUS, JCOG0602, and SCORPION) suggest that NACT is noninferior to PCS with regard to progression-free survival and overall survival. NACT is associated with less perioperative and postoperative morbidity and mortality and is associated with shorter hospital stays.
To date, complete data are available only from the EORTC and CHORUS trials, which both demonstrated similar progression-free survival and overall survival for NACT and PCS. Critics have noted, however, that both trials have shorter median overall survival for the PCS groups than were previously reported in other phase 3 studies in the United States, suggesting the possibility of different patient populations or less aggressive "surgical effort." Thus, PCS remains the preferred management strategy for women with advanced-stage ovarian cancer in whom there is a high likelihood of optimal cytoreduction.13
Recommendation: Use of neoadjuvant chemotherapy
Patients who are appropriate candidates for NACT should be treated with a platinum and taxane doublet and should receive interval cytoreduction following 3 to 4 cycles of therapy if a favorable response is noted. Patients whose disease progresses despite NACT have a poor prognosis, and there is little role for surgical treatment with the exception of palliative purposes.13
Neoadjuvant chemotherapy is a noninferior and appropriate treatment option for women who are poor surgical candidates or who have a low likelihood of optimal cytoreduction. When optimal cytoreduction is possible, however, PCS is preferred (see FIGURE). The data on the efficacy of NACT for ovarian cancer have led to increased use of this treatment in the United States.

Read about a new PARP inhibitor for maintenance therapy
Niraparib is promising as maintenance therapy in ovarian cancer
Mirza MR, Monk BJ, Herrstedt J, et al; for the ENGOT-OV16/NOVA Investigators. Niraparib maintenance therapy in platinum-sensitive, recurrent ovarian cancer. N Engl J Med. 2016;375(22):2154-2164.
Approximately 85% of women with ovarian cancer will develop recurrent disease. Women with ovarian cancer are commonly treated with a range of antineoplastic agents over the course of their lifetime. As such, there is a great need for additional active therapeutic agents in this setting. Recently, substantial effort has been directed toward "precision" or "personalized medicine" in oncology.
Precision medicine, targeted therapies in oncology
Precision medicine refers to the customization of medical therapy based on the genetic characterization of the individual patient or the molecular profile of the patient's tumor. As a result of large-scale molecular profiling from projects such as the International Cancer Genome Consortium and The Cancer Genome Atlas, an abundance of molecular data has been generated through the characterization of multiple tumor types. This has led to the discovery of key cancer drivers, alterations, and specific molecular profiles that have distinct prognostic and treatment implications. These data, in combination with the commercial availability of molecular profiling tests, has made precision medicine a reality for women with ovarian cancer.
This wealth of new information has led to development of targeted therapeutics that block the growth and spread of cancer by acting on specific molecules or molecular pathways. Targeted therapies approved for cancer treatment include hormonal therapies, signal transduction inhibitors, gene expression modulators, apoptosis inducers, angiogenesis inhibitors, and immunotherapies.14
How PARP inhibitors work
PARP inhibitors are a class of agents that are emerging as important therapies for ovarian cancer. These agents block the nuclear protein PARP, which functions to detect and repair single-strand DNA breaks with the resulting accumulation of double-stranded DNA breaks.15 In the setting of DNA damage, the homologous recombination repair pathway is activated for repair. However, homologous recombination deficiencies (HRD) can arise as a result of BRCA1 or BRCA2 mutations or BRCA-independent pathways, which effectively disable this DNA repair pathway. As a result, when PARP inhibitors are used in patients with HRD, the cell cannot repair double-stranded DNA breaks and this leads to "synthetic lethality."16
Understanding this molecular mechanism of PARP inhibitors as well as the frequent abnormalities in the BRCA genes and HRD pathways in ovarian cancer has provided an important potential therapeutic target in ovarian cancer. A number of PARP inhibitors are now commercially available and are undergoing testing in ovarian cancer.
Related article:
Is a minimally invasive approach to hysterectomy for Gyn cancer utilized equally in all racial and income groups?
Niraparib for ovarian cancer
In a randomized, double-blind, phase 3 trial by Mizra and colleagues, 553 women with platinum-sensitive recurrent ovarian cancer who responded to therapy were divided according to the presence or absence of a germline BRCA (gBRCA) mutation and randomly assigned to niraparib 300 mg or placebo once daily. Women in the niraparib group had a significantly longer median duration of progression-free survival than did those in the placebo group. This was most pronounced in women in the gBRCA cohort (21.0 vs 5.5 months). Importantly, niraparib was associated with improved progression-free survival in HRD-positive patients without gBRCA mutations (12.9 vs 3.8 months) as well as in the HRD-negative subgroup (6.9 vs 3.8 months).17
Overall, niraparib was well tolerated. About 15% of women discontinued the drug due to toxicity. Significant (grade 3 or 4) adverse events were seen in three-quarters of women treated with niraparib, and they most commonly consisted of hematologic toxicities. Patient-reported outcomes were similar for both groups, indicating no significant effect from niraparib on quality of life.17
This study's results suggest that niraparib has clinical activity against ovarian cancer. Importantly, niraparib was active in women with gBRCA mutations, in those with HRD without a gBRCA mutation, and potentially in women without HRD. If approved by the US Food and Drug Administration, niraparib will join olaparib and rucaparib as a newly approved therapeutic agent for ovarian cancer. This study provides important evidence that suggests niraparib maintenance therapy may be an efficacious and important addition to the treatment armamentarium for platinum-sensitive ovarian cancer.
Share your thoughts! Send your Letter to the Editor to [email protected]. Please include your name and the city and state in which you practice.
- Siegel RL, Miller KD, Jemal A. Cancer statistics, 2017. CA Cancer J Clin. 2017;67(1):7–30.
- National Cancer Institute Surveillance, Epidemiology, and End Results Program. Cancer stat facts: ovarian cancer. https://seer.cancer.gov/statfacts/html/ovary.html. Accessed January 20, 2017.
- Jacobs I, Davies AP, Bridges J, et al. Prevalence screening for ovarian cancer in postmenopausal women by CA 125 measurement and ultrasonography. BMJ. 1993;306(6884):1030–1034.
- van Nagell JR Jr, Pavlik EJ. Ovarian cancer screening. Clin Obstet Gynecol. 2012;55:43–51.
- Jacobs IJ, Menon U, Ryan A, et al. Ovarian cancer screening and mortality in the UK Collaborative Trial of Ovarian Cancer Screening (UKCTOCS): a randomised controlled trial. Lancet. 2016;387(10022):945–956.
- Buys SS, Partridge E, Black A, et al; PLCO Project Team. Effect of screening on ovarian cancer mortality: the Prostate, Lung, Colorectal and Ovarian (PLCO) Cancer Screening randomized controlled trial. JAMA. 2011;305(22):2295–2303.
- Moyer VA, US Preventive Services Task Force. Screening for ovarian cancer: US Preventive Services Task Force reaffirmation recommendation statement. Ann Intern Med. 2012;157(12):900–904.
- Schorge JO, McCann C, Del Carmen MG. Surgical debulking of ovarian cancer: what difference does it make? Rev Obstet Gynecol. 2010;3(3):111–117.
- Hoskins WJ, McGuire WP, Brady MF, et al. The effect of diameter of largest residual disease on survival after primary cytoreductive surgery in patients with suboptimal residual epithelial ovarian carcinoma. Am J Obstet Gynecol. 1994;170(4):974–979; discussion 979–980.
- Bristow RE, Tomacruz RS, Armstrong DK, Trimble EL, Montz FJ. Survival effect of maximal cytoreductive surgery for advanced ovarian carcinoma during the platinum era: a meta-analysis. J Clin Oncol. 2002;20(5):1248–1259.
- Vergote I, Trope CG, Amant F, et al; European Organization for Research and Treatment of Cancer–Gynaecological Cancer Goup; NCIC Clinical Trials Group. Neoadjuvant chemotherapy or primary surgery in stage IIIC or IV ovarian cancer. N Engl J Med. 2010;363(10):943–953.
- Kehoe S, Hook J, Nankivell M, et al. Primary chemotherapy versus primary surgery for newly diagnosed advanced ovarian cancer (CHORUS): an open-label, randomised, controlled, non-inferiority trial. Lancet. 2015;386(9990):249–257.
- Wright AA, Bohlke K, Armstrong DK, et al. Neoadjuvant chemotherapy for newly diagnosed, advanced ovarian cancer: Society of Gynecologic Oncology and American Society of Clinical Oncology clinical practice guideline. J Clin Oncol. 2016;34(28):3460–3473.
- National Cancer Institute. Targeted cancer therapies. https://www.cancer.gov/about-cancer/treatment/types/targeted-therapies/targeted-therapies-fact-sheet. Updated April 25, 2014. Accessed January 21, 2017.
- Drean A, Lord CJ, Ashworth A. PARP inhibitor combination therapy. Crit Rev Oncol Hematol. 2016;108:73–85.
- Ledermann JA, El-Khouly F. PARP inhibitors in ovarian cancer: clinical evidence for informed treatment decisions. Br J Cancer. 2015;113(suppl 1):S10–S16.
- Mirza MR, Monk BJ, Herrstedt J, et al; ENGOT-OV16/NOVA Investigators. Niraparib maintenance therapy in platinum-sensitive, recurrent ovarian cancer. N Engl J Med. 2016;375(22):2154–2164.
- Siegel RL, Miller KD, Jemal A. Cancer statistics, 2017. CA Cancer J Clin. 2017;67(1):7–30.
- National Cancer Institute Surveillance, Epidemiology, and End Results Program. Cancer stat facts: ovarian cancer. https://seer.cancer.gov/statfacts/html/ovary.html. Accessed January 20, 2017.
- Jacobs I, Davies AP, Bridges J, et al. Prevalence screening for ovarian cancer in postmenopausal women by CA 125 measurement and ultrasonography. BMJ. 1993;306(6884):1030–1034.
- van Nagell JR Jr, Pavlik EJ. Ovarian cancer screening. Clin Obstet Gynecol. 2012;55:43–51.
- Jacobs IJ, Menon U, Ryan A, et al. Ovarian cancer screening and mortality in the UK Collaborative Trial of Ovarian Cancer Screening (UKCTOCS): a randomised controlled trial. Lancet. 2016;387(10022):945–956.
- Buys SS, Partridge E, Black A, et al; PLCO Project Team. Effect of screening on ovarian cancer mortality: the Prostate, Lung, Colorectal and Ovarian (PLCO) Cancer Screening randomized controlled trial. JAMA. 2011;305(22):2295–2303.
- Moyer VA, US Preventive Services Task Force. Screening for ovarian cancer: US Preventive Services Task Force reaffirmation recommendation statement. Ann Intern Med. 2012;157(12):900–904.
- Schorge JO, McCann C, Del Carmen MG. Surgical debulking of ovarian cancer: what difference does it make? Rev Obstet Gynecol. 2010;3(3):111–117.
- Hoskins WJ, McGuire WP, Brady MF, et al. The effect of diameter of largest residual disease on survival after primary cytoreductive surgery in patients with suboptimal residual epithelial ovarian carcinoma. Am J Obstet Gynecol. 1994;170(4):974–979; discussion 979–980.
- Bristow RE, Tomacruz RS, Armstrong DK, Trimble EL, Montz FJ. Survival effect of maximal cytoreductive surgery for advanced ovarian carcinoma during the platinum era: a meta-analysis. J Clin Oncol. 2002;20(5):1248–1259.
- Vergote I, Trope CG, Amant F, et al; European Organization for Research and Treatment of Cancer–Gynaecological Cancer Goup; NCIC Clinical Trials Group. Neoadjuvant chemotherapy or primary surgery in stage IIIC or IV ovarian cancer. N Engl J Med. 2010;363(10):943–953.
- Kehoe S, Hook J, Nankivell M, et al. Primary chemotherapy versus primary surgery for newly diagnosed advanced ovarian cancer (CHORUS): an open-label, randomised, controlled, non-inferiority trial. Lancet. 2015;386(9990):249–257.
- Wright AA, Bohlke K, Armstrong DK, et al. Neoadjuvant chemotherapy for newly diagnosed, advanced ovarian cancer: Society of Gynecologic Oncology and American Society of Clinical Oncology clinical practice guideline. J Clin Oncol. 2016;34(28):3460–3473.
- National Cancer Institute. Targeted cancer therapies. https://www.cancer.gov/about-cancer/treatment/types/targeted-therapies/targeted-therapies-fact-sheet. Updated April 25, 2014. Accessed January 21, 2017.
- Drean A, Lord CJ, Ashworth A. PARP inhibitor combination therapy. Crit Rev Oncol Hematol. 2016;108:73–85.
- Ledermann JA, El-Khouly F. PARP inhibitors in ovarian cancer: clinical evidence for informed treatment decisions. Br J Cancer. 2015;113(suppl 1):S10–S16.
- Mirza MR, Monk BJ, Herrstedt J, et al; ENGOT-OV16/NOVA Investigators. Niraparib maintenance therapy in platinum-sensitive, recurrent ovarian cancer. N Engl J Med. 2016;375(22):2154–2164.
Sirukumab found effective, safe for highly refractory RA
The investigational interleukin-6 inhibitor sirukumab proved effective and safe for rheumatoid arthritis patients who failed to respond to or were intolerant of multiple previous therapies in a phase III trial reported online in The Lancet.
These patients are “exceptionally treatment-resistant” and have “substantial clinical needs with the lowest chance of treatment success,” said first author Daniel Aletaha, MD, of the division of rheumatology at the Medical University of Vienna, and his associates.
Two different doses of sirukumab provided rapid and sustained improvements in rheumatoid arthritis (RA) signs and symptoms, physical function, and health status. These were accompanied by improvements in physical and mental well-being, a result that is particularly relevant for RA patients, who have a high prevalence of mood disorders. It has been estimated that 39% of RA patients have moderate to severe depressive symptoms, the investigators noted.
They assessed sirukumab in the manufacturer-funded trial involving 878 adults treated and followed at 183 centers in 20 countries across North America, Europe, Latin America, and Asia. These patients included 59% who had failed to respond to or had been intolerant of two or more biologic disease-modifying antirheumatic drugs (39% to two or more tumor necrosis factor inhibitors) and numerous other RA treatments.
The patients in the study, called SIRROUND-T, were randomly assigned to receive 50 mg sirukumab every 4 weeks (292 patients in the low-dose group), 100 mg sirukumab every 2 weeks (292 in the high-dose group), or matching placebo (294 patients in the control group) via subcutaneous injections for 52 weeks. Patients receiving placebo whose condition worsened were allowed an “early escape,” a blinded switch to either dose of the active drug for the remainder of the trial. At week 24, patients remaining in the placebo groups also were randomly assigned in a blinded manner to switch to active treatment (Lancet. 2017 Feb 15. doi: 10.1016/S0140-6736[17]30401-4). All patients were allowed to continue using any concomitant disease-modifying antirheumatic drugs (DMARDs). At baseline, only 80% were using a conventional synthetic DMARD, and about 25% of patients in each group were not taking methotrexate.
The primary efficacy endpoint was the proportion of patients achieving an ACR20 response (a 20% or greater improvement in swollen joint count and tender joint count plus a 20% or greater improvement in at least three factors: pain, patient-assessed disease activity, physician-assessed disease activity, patient-assessed physical function, and C-reactive protein levels) at week 16. Sirukumab achieved higher levels of response in both the low-dose (40% of patients) and high-dose (45%) groups than did placebo (24%).
In addition, significantly higher proportions of patients receiving active treatment achieved the secondary endpoints of ACR20, ACR50, and ACR70 at week 24. Greater improvements, including remission, also were noted with sirukumab than with placebo according to the Simplified Disease Activity Index, ACR/EULAR criteria, and the Health Assessment Questionnaire–Disability Index. And significantly greater improvements also were seen in the Functional Assessment of Chronic Illness Therapy–Fatigue score, the 36-Item Short Form Health Survey, the Work Limitations Questionnaire, the EuroQol-5 Dimension questionnaire, and the EuroQol Health State Visual Analogue Scale.
In both active-treatment groups, all of these improvements were noted as early as within 2 weeks of starting treatment and persisted through follow-ups at week 24 and week 52.
Rates of adverse events, serious adverse events, and adverse events leading to treatment discontinuation were similar across the three study groups. Infections (primarily pneumonia) and infestations were the most frequent adverse events leading to discontinuation. No cases of tuberculosis or serious opportunistic infections were reported. Both doses of sirukumab induced declines in neutrophil, platelet, and white blood cell counts, as well as increases in alanine aminotransferase, aspartate aminotransferase, bilirubin, cholesterol, and triglycerides. Nine patients had hypersensitivity reactions, including one classified as serious dermatitis; all of these patients were taking the high dose of sirukumab. One of the five patient deaths that occurred, a fatal case of pneumonia, was considered possibly related to sirukumab.
This trial was funded by Janssen and GlaxoSmithKline, which also participated in the study design, data collection and analysis, and writing the results. Dr. Aletaha reported serving as a consultant for or receiving research support from AbbVie, Pfizer, Grünenthal, Merck, Medac, UCB, Mitsubishi/Tanabe, Janssen, and Roche. His associates reported ties to numerous industry sources.
It would be useful to compare sirukumab’s efficacy against that of two other inhibitors of the interleukin-6 pathway, tocilizumab (Actemra) and sarilumab.
Roy Fleischmann, MD, is with the University of Texas Southwestern Medical Center and Metroplex Clinical Research Center, both in Dallas. He reported receiving research grants and consulting fees from Genentech-Roche, Sanofi-Aventis, and GlaxoSmithKline. Dr. Fleischmann made these remarks in editorial accompanying Dr. Aletaha and colleagues’ report ( Lancet. 2017 Feb 15. doi: 10.1016/S0140-6736[17]30405-1 ).
It would be useful to compare sirukumab’s efficacy against that of two other inhibitors of the interleukin-6 pathway, tocilizumab (Actemra) and sarilumab.
Roy Fleischmann, MD, is with the University of Texas Southwestern Medical Center and Metroplex Clinical Research Center, both in Dallas. He reported receiving research grants and consulting fees from Genentech-Roche, Sanofi-Aventis, and GlaxoSmithKline. Dr. Fleischmann made these remarks in editorial accompanying Dr. Aletaha and colleagues’ report ( Lancet. 2017 Feb 15. doi: 10.1016/S0140-6736[17]30405-1 ).
It would be useful to compare sirukumab’s efficacy against that of two other inhibitors of the interleukin-6 pathway, tocilizumab (Actemra) and sarilumab.
Roy Fleischmann, MD, is with the University of Texas Southwestern Medical Center and Metroplex Clinical Research Center, both in Dallas. He reported receiving research grants and consulting fees from Genentech-Roche, Sanofi-Aventis, and GlaxoSmithKline. Dr. Fleischmann made these remarks in editorial accompanying Dr. Aletaha and colleagues’ report ( Lancet. 2017 Feb 15. doi: 10.1016/S0140-6736[17]30405-1 ).
The investigational interleukin-6 inhibitor sirukumab proved effective and safe for rheumatoid arthritis patients who failed to respond to or were intolerant of multiple previous therapies in a phase III trial reported online in The Lancet.
These patients are “exceptionally treatment-resistant” and have “substantial clinical needs with the lowest chance of treatment success,” said first author Daniel Aletaha, MD, of the division of rheumatology at the Medical University of Vienna, and his associates.
Two different doses of sirukumab provided rapid and sustained improvements in rheumatoid arthritis (RA) signs and symptoms, physical function, and health status. These were accompanied by improvements in physical and mental well-being, a result that is particularly relevant for RA patients, who have a high prevalence of mood disorders. It has been estimated that 39% of RA patients have moderate to severe depressive symptoms, the investigators noted.
They assessed sirukumab in the manufacturer-funded trial involving 878 adults treated and followed at 183 centers in 20 countries across North America, Europe, Latin America, and Asia. These patients included 59% who had failed to respond to or had been intolerant of two or more biologic disease-modifying antirheumatic drugs (39% to two or more tumor necrosis factor inhibitors) and numerous other RA treatments.
The patients in the study, called SIRROUND-T, were randomly assigned to receive 50 mg sirukumab every 4 weeks (292 patients in the low-dose group), 100 mg sirukumab every 2 weeks (292 in the high-dose group), or matching placebo (294 patients in the control group) via subcutaneous injections for 52 weeks. Patients receiving placebo whose condition worsened were allowed an “early escape,” a blinded switch to either dose of the active drug for the remainder of the trial. At week 24, patients remaining in the placebo groups also were randomly assigned in a blinded manner to switch to active treatment (Lancet. 2017 Feb 15. doi: 10.1016/S0140-6736[17]30401-4). All patients were allowed to continue using any concomitant disease-modifying antirheumatic drugs (DMARDs). At baseline, only 80% were using a conventional synthetic DMARD, and about 25% of patients in each group were not taking methotrexate.
The primary efficacy endpoint was the proportion of patients achieving an ACR20 response (a 20% or greater improvement in swollen joint count and tender joint count plus a 20% or greater improvement in at least three factors: pain, patient-assessed disease activity, physician-assessed disease activity, patient-assessed physical function, and C-reactive protein levels) at week 16. Sirukumab achieved higher levels of response in both the low-dose (40% of patients) and high-dose (45%) groups than did placebo (24%).
In addition, significantly higher proportions of patients receiving active treatment achieved the secondary endpoints of ACR20, ACR50, and ACR70 at week 24. Greater improvements, including remission, also were noted with sirukumab than with placebo according to the Simplified Disease Activity Index, ACR/EULAR criteria, and the Health Assessment Questionnaire–Disability Index. And significantly greater improvements also were seen in the Functional Assessment of Chronic Illness Therapy–Fatigue score, the 36-Item Short Form Health Survey, the Work Limitations Questionnaire, the EuroQol-5 Dimension questionnaire, and the EuroQol Health State Visual Analogue Scale.
In both active-treatment groups, all of these improvements were noted as early as within 2 weeks of starting treatment and persisted through follow-ups at week 24 and week 52.
Rates of adverse events, serious adverse events, and adverse events leading to treatment discontinuation were similar across the three study groups. Infections (primarily pneumonia) and infestations were the most frequent adverse events leading to discontinuation. No cases of tuberculosis or serious opportunistic infections were reported. Both doses of sirukumab induced declines in neutrophil, platelet, and white blood cell counts, as well as increases in alanine aminotransferase, aspartate aminotransferase, bilirubin, cholesterol, and triglycerides. Nine patients had hypersensitivity reactions, including one classified as serious dermatitis; all of these patients were taking the high dose of sirukumab. One of the five patient deaths that occurred, a fatal case of pneumonia, was considered possibly related to sirukumab.
This trial was funded by Janssen and GlaxoSmithKline, which also participated in the study design, data collection and analysis, and writing the results. Dr. Aletaha reported serving as a consultant for or receiving research support from AbbVie, Pfizer, Grünenthal, Merck, Medac, UCB, Mitsubishi/Tanabe, Janssen, and Roche. His associates reported ties to numerous industry sources.
The investigational interleukin-6 inhibitor sirukumab proved effective and safe for rheumatoid arthritis patients who failed to respond to or were intolerant of multiple previous therapies in a phase III trial reported online in The Lancet.
These patients are “exceptionally treatment-resistant” and have “substantial clinical needs with the lowest chance of treatment success,” said first author Daniel Aletaha, MD, of the division of rheumatology at the Medical University of Vienna, and his associates.
Two different doses of sirukumab provided rapid and sustained improvements in rheumatoid arthritis (RA) signs and symptoms, physical function, and health status. These were accompanied by improvements in physical and mental well-being, a result that is particularly relevant for RA patients, who have a high prevalence of mood disorders. It has been estimated that 39% of RA patients have moderate to severe depressive symptoms, the investigators noted.
They assessed sirukumab in the manufacturer-funded trial involving 878 adults treated and followed at 183 centers in 20 countries across North America, Europe, Latin America, and Asia. These patients included 59% who had failed to respond to or had been intolerant of two or more biologic disease-modifying antirheumatic drugs (39% to two or more tumor necrosis factor inhibitors) and numerous other RA treatments.
The patients in the study, called SIRROUND-T, were randomly assigned to receive 50 mg sirukumab every 4 weeks (292 patients in the low-dose group), 100 mg sirukumab every 2 weeks (292 in the high-dose group), or matching placebo (294 patients in the control group) via subcutaneous injections for 52 weeks. Patients receiving placebo whose condition worsened were allowed an “early escape,” a blinded switch to either dose of the active drug for the remainder of the trial. At week 24, patients remaining in the placebo groups also were randomly assigned in a blinded manner to switch to active treatment (Lancet. 2017 Feb 15. doi: 10.1016/S0140-6736[17]30401-4). All patients were allowed to continue using any concomitant disease-modifying antirheumatic drugs (DMARDs). At baseline, only 80% were using a conventional synthetic DMARD, and about 25% of patients in each group were not taking methotrexate.
The primary efficacy endpoint was the proportion of patients achieving an ACR20 response (a 20% or greater improvement in swollen joint count and tender joint count plus a 20% or greater improvement in at least three factors: pain, patient-assessed disease activity, physician-assessed disease activity, patient-assessed physical function, and C-reactive protein levels) at week 16. Sirukumab achieved higher levels of response in both the low-dose (40% of patients) and high-dose (45%) groups than did placebo (24%).
In addition, significantly higher proportions of patients receiving active treatment achieved the secondary endpoints of ACR20, ACR50, and ACR70 at week 24. Greater improvements, including remission, also were noted with sirukumab than with placebo according to the Simplified Disease Activity Index, ACR/EULAR criteria, and the Health Assessment Questionnaire–Disability Index. And significantly greater improvements also were seen in the Functional Assessment of Chronic Illness Therapy–Fatigue score, the 36-Item Short Form Health Survey, the Work Limitations Questionnaire, the EuroQol-5 Dimension questionnaire, and the EuroQol Health State Visual Analogue Scale.
In both active-treatment groups, all of these improvements were noted as early as within 2 weeks of starting treatment and persisted through follow-ups at week 24 and week 52.
Rates of adverse events, serious adverse events, and adverse events leading to treatment discontinuation were similar across the three study groups. Infections (primarily pneumonia) and infestations were the most frequent adverse events leading to discontinuation. No cases of tuberculosis or serious opportunistic infections were reported. Both doses of sirukumab induced declines in neutrophil, platelet, and white blood cell counts, as well as increases in alanine aminotransferase, aspartate aminotransferase, bilirubin, cholesterol, and triglycerides. Nine patients had hypersensitivity reactions, including one classified as serious dermatitis; all of these patients were taking the high dose of sirukumab. One of the five patient deaths that occurred, a fatal case of pneumonia, was considered possibly related to sirukumab.
This trial was funded by Janssen and GlaxoSmithKline, which also participated in the study design, data collection and analysis, and writing the results. Dr. Aletaha reported serving as a consultant for or receiving research support from AbbVie, Pfizer, Grünenthal, Merck, Medac, UCB, Mitsubishi/Tanabe, Janssen, and Roche. His associates reported ties to numerous industry sources.
FROM THE LANCET
Key clinical point: Sirukumab proved effective and safe for RA patients who failed to respond to or were intolerant of multiple previous therapies.
Key numerical finding: The primary efficacy endpoint – the proportion of patients achieving an ACR20 response at week 16 – was 40% for low-dose and 45% for high-dose sirukumab, compared with 24% for placebo.
Data source: A manufacturer-sponsored, international, randomized, double-blind, placebo-controlled, phase III trial involving 878 adults with refractory RA.
Disclosures: This trial was funded by Janssen and GlaxoSmithKline, which also participated in the study design, data collection and analysis, and writing of the results. Dr. Aletaha reported serving as a consultant for or receiving research support from AbbVie, Pfizer, Grünenthal, Merck, Medac, UCB, Mitsubishi/Tanabe, Janssen, and Roche. His associates reported ties to numerous industry sources.
It is time for HPV vaccination to be considered part of routine preventive health care
The recognition that human papillomavirus (HPV) oncogenic viruses cause cervical carcinoma remains one of the most game-changing medical discoveries of the last century. Improvements in screening options for detecting cervical cancer precursors followed. We now have the ability to detect high-risk HPV subtypes in routine specimens. Finally, a highly effective vaccine was developed that targets HPV types 16 and 18, which are responsible for causing approximately 70% of all cases of cervical carcinoma.
In one of the original vaccines HPV types 6 and 11, responsible for 90% of all genital warts, were also targeted. In 2014, a 9-valent vaccine incorporating an additional 5 HPV strains (31, 33, 45, 52, and 58) was approved and is set to replace all previous vaccine versions. Together, these 7 oncogenic HPV types are responsible for approximately 90% of HPV-related cancers, including cervical, anal, oropharyngeal, vaginal, and vulvar cancer.
By vaccinating boys and girls between ages 9 and 21 (for males) and 9 and 26 (for females), we could effectively eliminate 90% of genital warts and 90% of all HPV-related cancers. So why have we not capitalized on this extraordinary discovery? In 2016, why were only 40% of teenage girls and less than 25% of teenage boys vaccinated against HPV when we are immunizing 80% to 90% of these populations with tetanus, diphtheria, and acellular pertusis (Tdap) and meningococcal vaccines?
Related article:
2016 Update on cervical disease
Barriers to HPV vaccination
When the first HPV vaccine was approved in 2006, cost was a significant factor. Many health insurance plans did not cover this “discretionary” vaccine, which was viewed as a prevention for sexually transmitted infections rather than as a valuable intervention for the prevention of cervical and other cancers. At well over $125 per dose with 3 doses required for a full series, ObGyns were reluctant to stock and provide these expensive vaccines without assurance of reimbursement. The logistics of recalling patients for their subsequent vaccine doses were challenging for offices that were not accustomed to seeing patients for preventive care activities more than once a year. In addition, the office infrastructure required to maintain the vaccine stock and manage the necessary paperwork could be daunting. Finally, the requirement that patients be observed for 15 to 30 minutes in the office after vaccine administration created efficiency and rooming problems in busy, active practices.
Over time, almost all payers covered the HPV vaccines, but the logistical issues in ObGyn practices remain. Pediatric practices, on the other hand, are ideally suited for vaccine administration. Unfortunately, our colleagues delivering preventive care to young teens have persisted in considering the HPV vaccine as an optional adjunct to routine vaccination despite the advice of the Advisory Committee on Immunization Practices (ACIP) of the Centers for Disease Control and Prevention (CDC), which for many years has recommended the HPV vaccine for girls. In 2011, the ACIP extended the HPV vaccine recommendation to include boys beginning at ages 11 to 12.
New 2-dose HPV vaccine schedule for children <15 years
In October 2016, 10 years after the first HPV vaccine approval, the ACIP and the CDC approved a reduced, 2-dose schedule for those younger than 15.1 The first dose can be administered simultaneously with other recommended vaccines for 11- to 12-year-olds (the meningococcal and Tdap vaccines) and the second dose, 6 or 12 months later.2 The 12-month interval would allow administration, once again, of all required vaccines at the annual visit.
Pivotal immunogenicity study
The new recommendation is based on robust multinational data (52 sites in 15 countries, N = 1,518) from an open-label trial.3 Immunogenicity of 2 doses of the 9-valent HPV vaccine in girls and boys ages 9 to 14 was compared with that of a standard 3-dose regimen in adolescents and young women ages 16 to 26. Five cohorts were studied: boys 9 to 14 given 2 doses at 6-month intervals; girls 9 to 14 given 2 doses at 6-month intervals; boys and girls 9 to 14 given 2 doses at a 12-month interval; girls 9 to 14 given the standard 3-dose regimen; and girls and young women 16 to 26 receiving 3 doses over 6 months.
The authors assessed the antibody responses against each HPV subtype 1 month after the final vaccine dose. Data from 1,377 participants (90.7% of the original cohort) were analyzed. Prespecified antibody titers were set conservatively to ensure adequate immunogenicity. Noninferiority criteria had to be met for all 9 HPV types.
Trial results. The immune responses for the 9- to 14-year-olds were consistently higher than those for the 16- to 26-year-old age group regardless of the regimen—not a surprising finding since the initial trials for HPV vaccine demonstrated a greater response among younger vaccine recipients. In this trial, higher antibody responses were found for the 12-month dosing interval than for the 6-month interval, although both regimens produced an adequate response.
Immunogenicity remained at 6 months. Antibody levels were retested 6 months after the last dose of HPV vaccine in a post hoc analysis. In all groups the antib
Related article:
2015 Update on cervical disease: New ammo for HPV prevention and screening
Simplified dosing may help increase vaccination rates
What does this new dosing regimen mean for practice? It will be simpler to incorporate HPV vaccination routinely into the standard vaccine regimen for preadolescent boys and girls. In addition, counseling for HPV vaccine administration can be combined with counseling for the meningococcal vaccine and routine Tdap booster.
Notably, primary care physicians have reported perceiving HPV vaccine discussions with parents as burdensome, and they tend to discuss it last after conversations about Tdap and meningococcal vaccines.4 Brewer and colleagues5 documented a 5% increase in first HPV vaccine doses among patients in practices in which the providers were taught to “announce” the need for HPV vaccine along with other routine vaccines. There was no increase in HPV vaccine uptake among practices in which providers were taught to “discuss” HPV with parents and to address their concerns, or in control practices. Therefore, less conversation about HPV and the HPV vaccine, as distinct from any other recommended vaccines, is better.
With the new 2-dose regimen, it should be easier to convey that the HPV vaccine is another necessary, routine intervention for children’s health. We should be able to achieve 90% vaccination rates for HPV—similar to rates for Tdap.
Share your thoughts! Send your Letter to the Editor to [email protected]. Please include your name and the city and state in which you practice.
- Centers for Disease Control and Prevention. CDC recommends only two HPV shots for younger adolescents. https://www.cdc.gov/media/releases/2016/p1020-hpv-shots.html. Published October 19, 2016. Accessed February 22, 2017.
- Meites E, Kempe A, Markowitz LE. Use of a 2-dose schedule for human papillomavirus vaccination—updated recommendations of the Advisory Committee on Immunization Practices. Morbid Mortal Weekly Rep MMWR. 2016;65(49)1405–1408.
- Iverson OE, Miranda MJ, Ulied A, et al. Immunogenicity of the 9-valent HPV vaccine using 2-dose regimens in girls and boys vs a 3-dose regimen in women. JAMA. 2016;316(22):2411–2421.
- Gilkey MB, Moss JL, Coyne-Beasley T, Hall ME, Shah PH, Brewer NT. Physician communication about adolescent vaccination: how is human papillomavirus vaccine different? Prev Med. 2015;77:181–185.
The recognition that human papillomavirus (HPV) oncogenic viruses cause cervical carcinoma remains one of the most game-changing medical discoveries of the last century. Improvements in screening options for detecting cervical cancer precursors followed. We now have the ability to detect high-risk HPV subtypes in routine specimens. Finally, a highly effective vaccine was developed that targets HPV types 16 and 18, which are responsible for causing approximately 70% of all cases of cervical carcinoma.
In one of the original vaccines HPV types 6 and 11, responsible for 90% of all genital warts, were also targeted. In 2014, a 9-valent vaccine incorporating an additional 5 HPV strains (31, 33, 45, 52, and 58) was approved and is set to replace all previous vaccine versions. Together, these 7 oncogenic HPV types are responsible for approximately 90% of HPV-related cancers, including cervical, anal, oropharyngeal, vaginal, and vulvar cancer.
By vaccinating boys and girls between ages 9 and 21 (for males) and 9 and 26 (for females), we could effectively eliminate 90% of genital warts and 90% of all HPV-related cancers. So why have we not capitalized on this extraordinary discovery? In 2016, why were only 40% of teenage girls and less than 25% of teenage boys vaccinated against HPV when we are immunizing 80% to 90% of these populations with tetanus, diphtheria, and acellular pertusis (Tdap) and meningococcal vaccines?
Related article:
2016 Update on cervical disease
Barriers to HPV vaccination
When the first HPV vaccine was approved in 2006, cost was a significant factor. Many health insurance plans did not cover this “discretionary” vaccine, which was viewed as a prevention for sexually transmitted infections rather than as a valuable intervention for the prevention of cervical and other cancers. At well over $125 per dose with 3 doses required for a full series, ObGyns were reluctant to stock and provide these expensive vaccines without assurance of reimbursement. The logistics of recalling patients for their subsequent vaccine doses were challenging for offices that were not accustomed to seeing patients for preventive care activities more than once a year. In addition, the office infrastructure required to maintain the vaccine stock and manage the necessary paperwork could be daunting. Finally, the requirement that patients be observed for 15 to 30 minutes in the office after vaccine administration created efficiency and rooming problems in busy, active practices.
Over time, almost all payers covered the HPV vaccines, but the logistical issues in ObGyn practices remain. Pediatric practices, on the other hand, are ideally suited for vaccine administration. Unfortunately, our colleagues delivering preventive care to young teens have persisted in considering the HPV vaccine as an optional adjunct to routine vaccination despite the advice of the Advisory Committee on Immunization Practices (ACIP) of the Centers for Disease Control and Prevention (CDC), which for many years has recommended the HPV vaccine for girls. In 2011, the ACIP extended the HPV vaccine recommendation to include boys beginning at ages 11 to 12.
New 2-dose HPV vaccine schedule for children <15 years
In October 2016, 10 years after the first HPV vaccine approval, the ACIP and the CDC approved a reduced, 2-dose schedule for those younger than 15.1 The first dose can be administered simultaneously with other recommended vaccines for 11- to 12-year-olds (the meningococcal and Tdap vaccines) and the second dose, 6 or 12 months later.2 The 12-month interval would allow administration, once again, of all required vaccines at the annual visit.
Pivotal immunogenicity study
The new recommendation is based on robust multinational data (52 sites in 15 countries, N = 1,518) from an open-label trial.3 Immunogenicity of 2 doses of the 9-valent HPV vaccine in girls and boys ages 9 to 14 was compared with that of a standard 3-dose regimen in adolescents and young women ages 16 to 26. Five cohorts were studied: boys 9 to 14 given 2 doses at 6-month intervals; girls 9 to 14 given 2 doses at 6-month intervals; boys and girls 9 to 14 given 2 doses at a 12-month interval; girls 9 to 14 given the standard 3-dose regimen; and girls and young women 16 to 26 receiving 3 doses over 6 months.
The authors assessed the antibody responses against each HPV subtype 1 month after the final vaccine dose. Data from 1,377 participants (90.7% of the original cohort) were analyzed. Prespecified antibody titers were set conservatively to ensure adequate immunogenicity. Noninferiority criteria had to be met for all 9 HPV types.
Trial results. The immune responses for the 9- to 14-year-olds were consistently higher than those for the 16- to 26-year-old age group regardless of the regimen—not a surprising finding since the initial trials for HPV vaccine demonstrated a greater response among younger vaccine recipients. In this trial, higher antibody responses were found for the 12-month dosing interval than for the 6-month interval, although both regimens produced an adequate response.
Immunogenicity remained at 6 months. Antibody levels were retested 6 months after the last dose of HPV vaccine in a post hoc analysis. In all groups the antib
Related article:
2015 Update on cervical disease: New ammo for HPV prevention and screening
Simplified dosing may help increase vaccination rates
What does this new dosing regimen mean for practice? It will be simpler to incorporate HPV vaccination routinely into the standard vaccine regimen for preadolescent boys and girls. In addition, counseling for HPV vaccine administration can be combined with counseling for the meningococcal vaccine and routine Tdap booster.
Notably, primary care physicians have reported perceiving HPV vaccine discussions with parents as burdensome, and they tend to discuss it last after conversations about Tdap and meningococcal vaccines.4 Brewer and colleagues5 documented a 5% increase in first HPV vaccine doses among patients in practices in which the providers were taught to “announce” the need for HPV vaccine along with other routine vaccines. There was no increase in HPV vaccine uptake among practices in which providers were taught to “discuss” HPV with parents and to address their concerns, or in control practices. Therefore, less conversation about HPV and the HPV vaccine, as distinct from any other recommended vaccines, is better.
With the new 2-dose regimen, it should be easier to convey that the HPV vaccine is another necessary, routine intervention for children’s health. We should be able to achieve 90% vaccination rates for HPV—similar to rates for Tdap.
Share your thoughts! Send your Letter to the Editor to [email protected]. Please include your name and the city and state in which you practice.
The recognition that human papillomavirus (HPV) oncogenic viruses cause cervical carcinoma remains one of the most game-changing medical discoveries of the last century. Improvements in screening options for detecting cervical cancer precursors followed. We now have the ability to detect high-risk HPV subtypes in routine specimens. Finally, a highly effective vaccine was developed that targets HPV types 16 and 18, which are responsible for causing approximately 70% of all cases of cervical carcinoma.
In one of the original vaccines HPV types 6 and 11, responsible for 90% of all genital warts, were also targeted. In 2014, a 9-valent vaccine incorporating an additional 5 HPV strains (31, 33, 45, 52, and 58) was approved and is set to replace all previous vaccine versions. Together, these 7 oncogenic HPV types are responsible for approximately 90% of HPV-related cancers, including cervical, anal, oropharyngeal, vaginal, and vulvar cancer.
By vaccinating boys and girls between ages 9 and 21 (for males) and 9 and 26 (for females), we could effectively eliminate 90% of genital warts and 90% of all HPV-related cancers. So why have we not capitalized on this extraordinary discovery? In 2016, why were only 40% of teenage girls and less than 25% of teenage boys vaccinated against HPV when we are immunizing 80% to 90% of these populations with tetanus, diphtheria, and acellular pertusis (Tdap) and meningococcal vaccines?
Related article:
2016 Update on cervical disease
Barriers to HPV vaccination
When the first HPV vaccine was approved in 2006, cost was a significant factor. Many health insurance plans did not cover this “discretionary” vaccine, which was viewed as a prevention for sexually transmitted infections rather than as a valuable intervention for the prevention of cervical and other cancers. At well over $125 per dose with 3 doses required for a full series, ObGyns were reluctant to stock and provide these expensive vaccines without assurance of reimbursement. The logistics of recalling patients for their subsequent vaccine doses were challenging for offices that were not accustomed to seeing patients for preventive care activities more than once a year. In addition, the office infrastructure required to maintain the vaccine stock and manage the necessary paperwork could be daunting. Finally, the requirement that patients be observed for 15 to 30 minutes in the office after vaccine administration created efficiency and rooming problems in busy, active practices.
Over time, almost all payers covered the HPV vaccines, but the logistical issues in ObGyn practices remain. Pediatric practices, on the other hand, are ideally suited for vaccine administration. Unfortunately, our colleagues delivering preventive care to young teens have persisted in considering the HPV vaccine as an optional adjunct to routine vaccination despite the advice of the Advisory Committee on Immunization Practices (ACIP) of the Centers for Disease Control and Prevention (CDC), which for many years has recommended the HPV vaccine for girls. In 2011, the ACIP extended the HPV vaccine recommendation to include boys beginning at ages 11 to 12.
New 2-dose HPV vaccine schedule for children <15 years
In October 2016, 10 years after the first HPV vaccine approval, the ACIP and the CDC approved a reduced, 2-dose schedule for those younger than 15.1 The first dose can be administered simultaneously with other recommended vaccines for 11- to 12-year-olds (the meningococcal and Tdap vaccines) and the second dose, 6 or 12 months later.2 The 12-month interval would allow administration, once again, of all required vaccines at the annual visit.
Pivotal immunogenicity study
The new recommendation is based on robust multinational data (52 sites in 15 countries, N = 1,518) from an open-label trial.3 Immunogenicity of 2 doses of the 9-valent HPV vaccine in girls and boys ages 9 to 14 was compared with that of a standard 3-dose regimen in adolescents and young women ages 16 to 26. Five cohorts were studied: boys 9 to 14 given 2 doses at 6-month intervals; girls 9 to 14 given 2 doses at 6-month intervals; boys and girls 9 to 14 given 2 doses at a 12-month interval; girls 9 to 14 given the standard 3-dose regimen; and girls and young women 16 to 26 receiving 3 doses over 6 months.
The authors assessed the antibody responses against each HPV subtype 1 month after the final vaccine dose. Data from 1,377 participants (90.7% of the original cohort) were analyzed. Prespecified antibody titers were set conservatively to ensure adequate immunogenicity. Noninferiority criteria had to be met for all 9 HPV types.
Trial results. The immune responses for the 9- to 14-year-olds were consistently higher than those for the 16- to 26-year-old age group regardless of the regimen—not a surprising finding since the initial trials for HPV vaccine demonstrated a greater response among younger vaccine recipients. In this trial, higher antibody responses were found for the 12-month dosing interval than for the 6-month interval, although both regimens produced an adequate response.
Immunogenicity remained at 6 months. Antibody levels were retested 6 months after the last dose of HPV vaccine in a post hoc analysis. In all groups the antib
Related article:
2015 Update on cervical disease: New ammo for HPV prevention and screening
Simplified dosing may help increase vaccination rates
What does this new dosing regimen mean for practice? It will be simpler to incorporate HPV vaccination routinely into the standard vaccine regimen for preadolescent boys and girls. In addition, counseling for HPV vaccine administration can be combined with counseling for the meningococcal vaccine and routine Tdap booster.
Notably, primary care physicians have reported perceiving HPV vaccine discussions with parents as burdensome, and they tend to discuss it last after conversations about Tdap and meningococcal vaccines.4 Brewer and colleagues5 documented a 5% increase in first HPV vaccine doses among patients in practices in which the providers were taught to “announce” the need for HPV vaccine along with other routine vaccines. There was no increase in HPV vaccine uptake among practices in which providers were taught to “discuss” HPV with parents and to address their concerns, or in control practices. Therefore, less conversation about HPV and the HPV vaccine, as distinct from any other recommended vaccines, is better.
With the new 2-dose regimen, it should be easier to convey that the HPV vaccine is another necessary, routine intervention for children’s health. We should be able to achieve 90% vaccination rates for HPV—similar to rates for Tdap.
Share your thoughts! Send your Letter to the Editor to [email protected]. Please include your name and the city and state in which you practice.
- Centers for Disease Control and Prevention. CDC recommends only two HPV shots for younger adolescents. https://www.cdc.gov/media/releases/2016/p1020-hpv-shots.html. Published October 19, 2016. Accessed February 22, 2017.
- Meites E, Kempe A, Markowitz LE. Use of a 2-dose schedule for human papillomavirus vaccination—updated recommendations of the Advisory Committee on Immunization Practices. Morbid Mortal Weekly Rep MMWR. 2016;65(49)1405–1408.
- Iverson OE, Miranda MJ, Ulied A, et al. Immunogenicity of the 9-valent HPV vaccine using 2-dose regimens in girls and boys vs a 3-dose regimen in women. JAMA. 2016;316(22):2411–2421.
- Gilkey MB, Moss JL, Coyne-Beasley T, Hall ME, Shah PH, Brewer NT. Physician communication about adolescent vaccination: how is human papillomavirus vaccine different? Prev Med. 2015;77:181–185.
- Centers for Disease Control and Prevention. CDC recommends only two HPV shots for younger adolescents. https://www.cdc.gov/media/releases/2016/p1020-hpv-shots.html. Published October 19, 2016. Accessed February 22, 2017.
- Meites E, Kempe A, Markowitz LE. Use of a 2-dose schedule for human papillomavirus vaccination—updated recommendations of the Advisory Committee on Immunization Practices. Morbid Mortal Weekly Rep MMWR. 2016;65(49)1405–1408.
- Iverson OE, Miranda MJ, Ulied A, et al. Immunogenicity of the 9-valent HPV vaccine using 2-dose regimens in girls and boys vs a 3-dose regimen in women. JAMA. 2016;316(22):2411–2421.
- Gilkey MB, Moss JL, Coyne-Beasley T, Hall ME, Shah PH, Brewer NT. Physician communication about adolescent vaccination: how is human papillomavirus vaccine different? Prev Med. 2015;77:181–185.
Alessandro Biffi, MD
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
Ticagrelor beats aspirin for recurrent stroke in patients with atherosclerosis
HOUSTON – Ticagrelor outperformed aspirin in preventing a combination of recurrent stroke, heart attack, and death – but only in patients whose index stroke was probably related to atherosclerosis.
The antiplatelet drug reduced the risk of the composite endpoint by 32%, compared with aspirin, in stroke patients with proven ipsilateral atherosclerotic stenosis (hazard ratio, 0.68). But ticagrelor (Brilinta) had no effect at all in those without stenosis (HR, 0.97), Pierre Amarenco, MD, said at the International Stroke Conference, sponsored by the American Heart Association.
The study was simultaneously published in Lancet Neurology (Lancet Neurol. 2017 Feb 23. doi: 10.1016/S1474-4422[17]30038-8). “The interaction that we found suggests what we already know in clinical practice: An understanding of stroke mechanisms and their causes is important to being able to deliver safe and effective treatment of early stroke prevention,” said Dr. Amarenco of Paris-Diderot Sorbonne University, Paris.
The findings come from a preplanned subgroup analysis of the large SOCRATES trial, published in 2016. The study determined that ticagrelor was no better than aspirin in preventing recurrent stroke, heart attack or death in patients who had a transient ischemic attack.
SOCRATES randomized 13,199 patients with a nonsevere ischemic stroke or high-risk transient ischemic attack to ticagrelor (180 mg loading dose on day 1 followed by 90 mg twice daily for days 2-90) or aspirin (300 mg on day 1 followed by 100 mg daily for days 2-90). The primary endpoint was the time to the occurrence of stroke, myocardial infarction, or death within 90 days.
The search for a potentially responsive group made sense, Dr. Amarenco said, because ticagrelor “is an effective antiplatelet therapy in patients with coronary atherosclerotic disease.” Therefore, investigators reasoned, it might be most effective in patients whose strokes were of atherosclerotic origin.
The substudy focused on 3,081 of the patients with proven ipsilateral atherosclerotic stenosis and/or a mobile thrombus or plaque in the aortic arch that was judged to potentially have caused their index stroke. Generally, the atherosclerotic patients were older and more likely to have dyslipidemia, hypertension, diabetes, coronary artery disease, and heart failure than were the patients with strokes of nonatherosclerotic origin. Atherosclerotic patients also were significantly more likely to have had a prior stroke or heart attack.
In the group with atherosclerosis, ticagrelor was significantly more effective at preventing the composite primary endpoint than was aspirin. There were 103 events in 1,542 patients in the ticagrelor group and 147 in 1,539 patients in the aspirin group (6.7% vs. 9.6%) – a “very impressive” risk reduction of 32% (HR, 0.68), Dr. Amarenco said.
In the group of patients without ipsilateral atherosclerotic stenosis, ticagrelor exerted no benefit over aspirin, with an event rate of 6.7% vs. 6.9% (HR, 0.97).
The rate of recurrent ischemic stroke was the driving force behind the significant between-group difference. Ischemic stroke occurred in 6.4% of those taking ticagrelor and 8.5% of those taking aspirin – a significant risk reduction of 27% (HR, 0.73). The drug exerted no benefit for recurrent ischemic stroke over aspirin in the group without atherosclerosis (5.8% vs. 6.1%; HR, 0.93).
There were no differences in the rate of heart attack or death, or in the secondary endpoints of all stroke, disabling stroke, or fatal stroke.
Ticagrelor was not associated with any major bleeding, compared with aspirin in either group, Dr. Amarenco noted.
The higher event rate in the patients with atherosclerosis is not surprising, he said.
“We had the exact same finding in our recent study with TIAregistry.org, which we found that patients with large artery atherosclerosis were at much higher risk than patients with other stroke subtypes.”
Dr. Amarenco disclosed financial relationships with numerous pharmaceutical companies, including AstraZeneca, which sponsored the study.
[email protected]
On Twitter @alz_gal
HOUSTON – Ticagrelor outperformed aspirin in preventing a combination of recurrent stroke, heart attack, and death – but only in patients whose index stroke was probably related to atherosclerosis.
The antiplatelet drug reduced the risk of the composite endpoint by 32%, compared with aspirin, in stroke patients with proven ipsilateral atherosclerotic stenosis (hazard ratio, 0.68). But ticagrelor (Brilinta) had no effect at all in those without stenosis (HR, 0.97), Pierre Amarenco, MD, said at the International Stroke Conference, sponsored by the American Heart Association.
The study was simultaneously published in Lancet Neurology (Lancet Neurol. 2017 Feb 23. doi: 10.1016/S1474-4422[17]30038-8). “The interaction that we found suggests what we already know in clinical practice: An understanding of stroke mechanisms and their causes is important to being able to deliver safe and effective treatment of early stroke prevention,” said Dr. Amarenco of Paris-Diderot Sorbonne University, Paris.
The findings come from a preplanned subgroup analysis of the large SOCRATES trial, published in 2016. The study determined that ticagrelor was no better than aspirin in preventing recurrent stroke, heart attack or death in patients who had a transient ischemic attack.
SOCRATES randomized 13,199 patients with a nonsevere ischemic stroke or high-risk transient ischemic attack to ticagrelor (180 mg loading dose on day 1 followed by 90 mg twice daily for days 2-90) or aspirin (300 mg on day 1 followed by 100 mg daily for days 2-90). The primary endpoint was the time to the occurrence of stroke, myocardial infarction, or death within 90 days.
The search for a potentially responsive group made sense, Dr. Amarenco said, because ticagrelor “is an effective antiplatelet therapy in patients with coronary atherosclerotic disease.” Therefore, investigators reasoned, it might be most effective in patients whose strokes were of atherosclerotic origin.
The substudy focused on 3,081 of the patients with proven ipsilateral atherosclerotic stenosis and/or a mobile thrombus or plaque in the aortic arch that was judged to potentially have caused their index stroke. Generally, the atherosclerotic patients were older and more likely to have dyslipidemia, hypertension, diabetes, coronary artery disease, and heart failure than were the patients with strokes of nonatherosclerotic origin. Atherosclerotic patients also were significantly more likely to have had a prior stroke or heart attack.
In the group with atherosclerosis, ticagrelor was significantly more effective at preventing the composite primary endpoint than was aspirin. There were 103 events in 1,542 patients in the ticagrelor group and 147 in 1,539 patients in the aspirin group (6.7% vs. 9.6%) – a “very impressive” risk reduction of 32% (HR, 0.68), Dr. Amarenco said.
In the group of patients without ipsilateral atherosclerotic stenosis, ticagrelor exerted no benefit over aspirin, with an event rate of 6.7% vs. 6.9% (HR, 0.97).
The rate of recurrent ischemic stroke was the driving force behind the significant between-group difference. Ischemic stroke occurred in 6.4% of those taking ticagrelor and 8.5% of those taking aspirin – a significant risk reduction of 27% (HR, 0.73). The drug exerted no benefit for recurrent ischemic stroke over aspirin in the group without atherosclerosis (5.8% vs. 6.1%; HR, 0.93).
There were no differences in the rate of heart attack or death, or in the secondary endpoints of all stroke, disabling stroke, or fatal stroke.
Ticagrelor was not associated with any major bleeding, compared with aspirin in either group, Dr. Amarenco noted.
The higher event rate in the patients with atherosclerosis is not surprising, he said.
“We had the exact same finding in our recent study with TIAregistry.org, which we found that patients with large artery atherosclerosis were at much higher risk than patients with other stroke subtypes.”
Dr. Amarenco disclosed financial relationships with numerous pharmaceutical companies, including AstraZeneca, which sponsored the study.
[email protected]
On Twitter @alz_gal
HOUSTON – Ticagrelor outperformed aspirin in preventing a combination of recurrent stroke, heart attack, and death – but only in patients whose index stroke was probably related to atherosclerosis.
The antiplatelet drug reduced the risk of the composite endpoint by 32%, compared with aspirin, in stroke patients with proven ipsilateral atherosclerotic stenosis (hazard ratio, 0.68). But ticagrelor (Brilinta) had no effect at all in those without stenosis (HR, 0.97), Pierre Amarenco, MD, said at the International Stroke Conference, sponsored by the American Heart Association.
The study was simultaneously published in Lancet Neurology (Lancet Neurol. 2017 Feb 23. doi: 10.1016/S1474-4422[17]30038-8). “The interaction that we found suggests what we already know in clinical practice: An understanding of stroke mechanisms and their causes is important to being able to deliver safe and effective treatment of early stroke prevention,” said Dr. Amarenco of Paris-Diderot Sorbonne University, Paris.
The findings come from a preplanned subgroup analysis of the large SOCRATES trial, published in 2016. The study determined that ticagrelor was no better than aspirin in preventing recurrent stroke, heart attack or death in patients who had a transient ischemic attack.
SOCRATES randomized 13,199 patients with a nonsevere ischemic stroke or high-risk transient ischemic attack to ticagrelor (180 mg loading dose on day 1 followed by 90 mg twice daily for days 2-90) or aspirin (300 mg on day 1 followed by 100 mg daily for days 2-90). The primary endpoint was the time to the occurrence of stroke, myocardial infarction, or death within 90 days.
The search for a potentially responsive group made sense, Dr. Amarenco said, because ticagrelor “is an effective antiplatelet therapy in patients with coronary atherosclerotic disease.” Therefore, investigators reasoned, it might be most effective in patients whose strokes were of atherosclerotic origin.
The substudy focused on 3,081 of the patients with proven ipsilateral atherosclerotic stenosis and/or a mobile thrombus or plaque in the aortic arch that was judged to potentially have caused their index stroke. Generally, the atherosclerotic patients were older and more likely to have dyslipidemia, hypertension, diabetes, coronary artery disease, and heart failure than were the patients with strokes of nonatherosclerotic origin. Atherosclerotic patients also were significantly more likely to have had a prior stroke or heart attack.
In the group with atherosclerosis, ticagrelor was significantly more effective at preventing the composite primary endpoint than was aspirin. There were 103 events in 1,542 patients in the ticagrelor group and 147 in 1,539 patients in the aspirin group (6.7% vs. 9.6%) – a “very impressive” risk reduction of 32% (HR, 0.68), Dr. Amarenco said.
In the group of patients without ipsilateral atherosclerotic stenosis, ticagrelor exerted no benefit over aspirin, with an event rate of 6.7% vs. 6.9% (HR, 0.97).
The rate of recurrent ischemic stroke was the driving force behind the significant between-group difference. Ischemic stroke occurred in 6.4% of those taking ticagrelor and 8.5% of those taking aspirin – a significant risk reduction of 27% (HR, 0.73). The drug exerted no benefit for recurrent ischemic stroke over aspirin in the group without atherosclerosis (5.8% vs. 6.1%; HR, 0.93).
There were no differences in the rate of heart attack or death, or in the secondary endpoints of all stroke, disabling stroke, or fatal stroke.
Ticagrelor was not associated with any major bleeding, compared with aspirin in either group, Dr. Amarenco noted.
The higher event rate in the patients with atherosclerosis is not surprising, he said.
“We had the exact same finding in our recent study with TIAregistry.org, which we found that patients with large artery atherosclerosis were at much higher risk than patients with other stroke subtypes.”
Dr. Amarenco disclosed financial relationships with numerous pharmaceutical companies, including AstraZeneca, which sponsored the study.
[email protected]
On Twitter @alz_gal
AT THE INTERNATIONAL STROKE CONFERENCE
Key clinical point:
Major finding: The drug cut risk of a combination of recurrent stroke, heart attack, and death by 32% among these patients, but was not better than aspirin for patients without atherosclerosis.
Data source: The subanalysis of the SOCRATES trial, comprising 3,081 patients with atherosclerosis and 10,118 without.
Disclosures: Dr. Amarenco disclosed financial relationships with numerous pharmaceutical companies, including AstraZeneca, which sponsored the study.