User login
A job to love
I would like to think it was the culmination of a series of clever decisions, but finding myself in a job that I enjoyed was more than likely the result of blind luck. Even as I filled out medical school applications during my senior year in college, I had no intention of actually becoming a physician. I was more focused on not becoming cannon fodder in Vietnam. I am hesitant to use the word love to describe my affection for a job I did for 40 years. But I can’t imagine any work I could have enjoyed more than being a general pediatrician in a small town.
Were there moments when I would have rather been watching one of my children play in a postseason soccer game than see a patient in the office? Sure, but I can’t recall a morning when I dreaded going to work. Having listened to many other people, including my father, complain about their work, I consider myself fortunate to have discovered a job that wasn’t just tolerable and a way to support my family, but one that I actually enjoyed enough to not mind working nights and weekends.
What was it about being a pediatrician that fueled my affection for it? Social scientists have asked the same question, and one of the answers they discovered is that jobs that offer a degree of autonomy and contribute positively to society are more likely to have satisfied workers (“The Incalculable Value of Finding a Job You Love,” by Robert Frank, the New York Times, July 22, 2016). If one assumes that the mission of pediatrics is to help children become and stay healthy, then when I was practicing solo or in a small physician-owned practice, my job easily met these two criteria. But autonomy and a good cause don’t necessarily pay the rent. However, unless I had foolishly chosen to open a practice in an area already saturated with physicians, doing pediatrics meant I would have an adequate income.
Like any craft, practicing pediatrics became easier and more enjoyable as I gained experience. I made fewer time-gobbling errors and had more therapeutic successes. It’s not that more children got better or better quicker under my care. They were going to get better, regardless of what I did. But over time, an increasing number of parents and patients seemed to be appreciative of my role in educating and reassuring them.
So what happened? I retired from office practice 3 years ago. Had I fallen out of love with pediatrics? My physical stamina was and still is good. I just go to bed earlier. But as my practice was swallowed by larger and larger entities, I lost most of the autonomy that had been so appealing. Practicing medicine has always been a business. It has to be unless you are living off an inherited trust fund. But despite praiseworthy mission statements, corporate decisions were being made that were no longer consistent with the kind of individualized care I thought the patients deserved. It was frustrating to hear families who I had been seeing for decades complain that the care delivery system in our office had taken several steps back.
At the risk of whipping the same old tired horse, I must say that it was the impending introduction of a third new and increasingly less-patient and physician-friendly EHR that made it too difficult to accept the accumulation of negatives in exchange for the wonderful feeling at the end of the workday during which at least one person had thanked me or told me I had done a good job.
For those of you that remain on the job, I urge you to fight the good fight to preserve what it is about practicing pediatrics that allows you to get up in the morning and head off to work without grumbling. It won’t be easy, but if you can make it into a job you love, the patients are going to benefit along with you.
Dr. Wilkoff practiced primary care pediatrics in Brunswick, Maine, for nearly 40 years. He has authored several books on behavioral pediatrics including “How to Say No to Your Toddler.”
I would like to think it was the culmination of a series of clever decisions, but finding myself in a job that I enjoyed was more than likely the result of blind luck. Even as I filled out medical school applications during my senior year in college, I had no intention of actually becoming a physician. I was more focused on not becoming cannon fodder in Vietnam. I am hesitant to use the word love to describe my affection for a job I did for 40 years. But I can’t imagine any work I could have enjoyed more than being a general pediatrician in a small town.
Were there moments when I would have rather been watching one of my children play in a postseason soccer game than see a patient in the office? Sure, but I can’t recall a morning when I dreaded going to work. Having listened to many other people, including my father, complain about their work, I consider myself fortunate to have discovered a job that wasn’t just tolerable and a way to support my family, but one that I actually enjoyed enough to not mind working nights and weekends.
What was it about being a pediatrician that fueled my affection for it? Social scientists have asked the same question, and one of the answers they discovered is that jobs that offer a degree of autonomy and contribute positively to society are more likely to have satisfied workers (“The Incalculable Value of Finding a Job You Love,” by Robert Frank, the New York Times, July 22, 2016). If one assumes that the mission of pediatrics is to help children become and stay healthy, then when I was practicing solo or in a small physician-owned practice, my job easily met these two criteria. But autonomy and a good cause don’t necessarily pay the rent. However, unless I had foolishly chosen to open a practice in an area already saturated with physicians, doing pediatrics meant I would have an adequate income.
Like any craft, practicing pediatrics became easier and more enjoyable as I gained experience. I made fewer time-gobbling errors and had more therapeutic successes. It’s not that more children got better or better quicker under my care. They were going to get better, regardless of what I did. But over time, an increasing number of parents and patients seemed to be appreciative of my role in educating and reassuring them.
So what happened? I retired from office practice 3 years ago. Had I fallen out of love with pediatrics? My physical stamina was and still is good. I just go to bed earlier. But as my practice was swallowed by larger and larger entities, I lost most of the autonomy that had been so appealing. Practicing medicine has always been a business. It has to be unless you are living off an inherited trust fund. But despite praiseworthy mission statements, corporate decisions were being made that were no longer consistent with the kind of individualized care I thought the patients deserved. It was frustrating to hear families who I had been seeing for decades complain that the care delivery system in our office had taken several steps back.
At the risk of whipping the same old tired horse, I must say that it was the impending introduction of a third new and increasingly less-patient and physician-friendly EHR that made it too difficult to accept the accumulation of negatives in exchange for the wonderful feeling at the end of the workday during which at least one person had thanked me or told me I had done a good job.
For those of you that remain on the job, I urge you to fight the good fight to preserve what it is about practicing pediatrics that allows you to get up in the morning and head off to work without grumbling. It won’t be easy, but if you can make it into a job you love, the patients are going to benefit along with you.
Dr. Wilkoff practiced primary care pediatrics in Brunswick, Maine, for nearly 40 years. He has authored several books on behavioral pediatrics including “How to Say No to Your Toddler.”
I would like to think it was the culmination of a series of clever decisions, but finding myself in a job that I enjoyed was more than likely the result of blind luck. Even as I filled out medical school applications during my senior year in college, I had no intention of actually becoming a physician. I was more focused on not becoming cannon fodder in Vietnam. I am hesitant to use the word love to describe my affection for a job I did for 40 years. But I can’t imagine any work I could have enjoyed more than being a general pediatrician in a small town.
Were there moments when I would have rather been watching one of my children play in a postseason soccer game than see a patient in the office? Sure, but I can’t recall a morning when I dreaded going to work. Having listened to many other people, including my father, complain about their work, I consider myself fortunate to have discovered a job that wasn’t just tolerable and a way to support my family, but one that I actually enjoyed enough to not mind working nights and weekends.
What was it about being a pediatrician that fueled my affection for it? Social scientists have asked the same question, and one of the answers they discovered is that jobs that offer a degree of autonomy and contribute positively to society are more likely to have satisfied workers (“The Incalculable Value of Finding a Job You Love,” by Robert Frank, the New York Times, July 22, 2016). If one assumes that the mission of pediatrics is to help children become and stay healthy, then when I was practicing solo or in a small physician-owned practice, my job easily met these two criteria. But autonomy and a good cause don’t necessarily pay the rent. However, unless I had foolishly chosen to open a practice in an area already saturated with physicians, doing pediatrics meant I would have an adequate income.
Like any craft, practicing pediatrics became easier and more enjoyable as I gained experience. I made fewer time-gobbling errors and had more therapeutic successes. It’s not that more children got better or better quicker under my care. They were going to get better, regardless of what I did. But over time, an increasing number of parents and patients seemed to be appreciative of my role in educating and reassuring them.
So what happened? I retired from office practice 3 years ago. Had I fallen out of love with pediatrics? My physical stamina was and still is good. I just go to bed earlier. But as my practice was swallowed by larger and larger entities, I lost most of the autonomy that had been so appealing. Practicing medicine has always been a business. It has to be unless you are living off an inherited trust fund. But despite praiseworthy mission statements, corporate decisions were being made that were no longer consistent with the kind of individualized care I thought the patients deserved. It was frustrating to hear families who I had been seeing for decades complain that the care delivery system in our office had taken several steps back.
At the risk of whipping the same old tired horse, I must say that it was the impending introduction of a third new and increasingly less-patient and physician-friendly EHR that made it too difficult to accept the accumulation of negatives in exchange for the wonderful feeling at the end of the workday during which at least one person had thanked me or told me I had done a good job.
For those of you that remain on the job, I urge you to fight the good fight to preserve what it is about practicing pediatrics that allows you to get up in the morning and head off to work without grumbling. It won’t be easy, but if you can make it into a job you love, the patients are going to benefit along with you.
Dr. Wilkoff practiced primary care pediatrics in Brunswick, Maine, for nearly 40 years. He has authored several books on behavioral pediatrics including “How to Say No to Your Toddler.”
Are Antipsychotics Necessary in Patients With Dementia?
TORONTO—Approximately 80% of elderly people with dementia in nursing homes do not have a reemergence of behavioral symptoms after cessation of antipsychotic drugs, according to research presented at the Alzheimer’s Association International Conference. The results may indicate that the antipsychotics are not necessary in these patients.
Many residents of nursing homes receive antipsychotics, but the drugs’ effect size is modest, said Henry Brodaty, MD, DSc, Professor of Aging and Mental Health and Codirector of the Center for Healthy Brain Aging at the University of New South Wales in Sydney. Antipsychotics can cause extrapyramidal side effects, falls, anticholinergic side effects such as worse cognition, urinary outflow difficulties and constipation, and orthostatic hypotension. The medicines also are associated with increased rates of stroke and death. In a UK study, Ballard and colleagues found no difference in the reemergence of behavioral symptoms between patients on continuous antipsychotics who were randomized to withdrawal and patients who continued their antipsychotics. However, Devenand and colleagues reported reemergence of behavioral symptoms when patients ceased antipsychotics.
Nurses Used Behavioral Strategies
To investigate the matter further, Dr. Brodaty and colleagues initiated the Halting Antipsychotics in Long-Term Care (HALT) study. The single-arm, 12-month, longitudinal study took place in 23 nursing homes in the Australian state of New South Wales. A nurse at each nursing home was named a “nurse champion” and identified residents who were receiving continuous antipsychotics. The researchers obtained permission from the nursing homes, the patients’ families, and the patients’ physicians to include the patients in the study.
During three days of training, each nurse champion learned how to manage behavioral symptoms and how to use person-centered care and behavioral strategies. These nurse champions subsequently trained the other nurses at their respective facilities in these techniques. Next, investigators monitored the patients for a month to establish their baseline conditions. Antipsychotics were stopped for all patients according to a protocol, and the researchers reassessed participants three, six, and 12 months later.
Participants’ mean age was about 85, and approximately two-thirds of patients were female. More than half of the sample was separated, widowed, or divorced. Approximately one-third of participants were married. About half of the sample had a high level of education, and half had a low level. A chart review indicated that one-third of patients had Alzheimer’s disease, and less than one-third had unspecified dementia. Smaller portions of the population had vascular or other dementias.
Participants received an average of 2.3 psychotropic medications and an average of nine medications for other indications. The total of about 11 medications per person was “about average,” said Dr. Brodaty. The most common antipsychotic was risperidone, followed by quetiapine, olanzapine, and haloperidol. Participants had been taking their antipsychotics for about two years and their current doses for at least one year. More than half of participants started antipsychotic medication after admission to the nursing home. About 20% of participants started antipsychotics during hospitalization before transfer to the nursing home, and 8% started antipsychotics in the community. For others, this information was not available.
Neuropsychiatric Symptoms Did Not Change
In all, 150 participants completed the prebaseline assessment, 139 completed the baseline assessment, 133 participants completed the three-month follow-up, and 118 participants completed the six-month follow-up. The 12-month follow-up was incomplete at the time of the presentation.
Of the 125 patients who had stopped their antipsychotics, the drugs were represcribed to 15 patients during the first three months. At six months, 10 of 118 patients for whom data were available had antipsychotics represcribed. In the final six months of the study, one patient had an antipsychotic represcribed. “In other words, almost 80% remained deprescribed, usually completely ceased,” said Dr. Brodaty. Many physicians wrote prn prescriptions, mainly for oxazepam as rescue medication, but the investigators have not analyzed the data to determine whether they were filled.
Despite the cessation of antipsychotics for most of the residents, there was no significant change in participants’ total Neuropsychiatric Inventory (NPI) score over the five follow-up visits, nor in their agitation (as measured by the Cohen Mansfield Agitation Inventory), aggression (as measured by the NPI), delusions, or hallucinations.
Should Standard of Care Change?
It was difficult to recruit patients for the study, said Dr. Brodaty. The investigators approached 58 nursing homes, and more than half declined to participate. Nurse champions also may not have included all patients on antipsychotics in the study. The results thus may have been influenced by selection bias. Staff at the nursing homes, physicians, and families appeared to be ignorant about the potential benefits of stopping antipsychotics such as preventing anticholinergic and other side effects.
“Inappropriate use of antipsychotics is an old story, yet we are still talking about it,” said Dr. Brodaty. “We have the knowledge and now we have to build on this knowledge and change practice. As a side issue, informed consent was pretty well lacking, and there is a lack of accountability about getting informed consent.”
Incentives for the owners, managers, and staff of nursing homes, as well as education for patients’ families, could promote person-centered care and reduce the inappropriate use of antipsychotics.
“Our next steps are to look at the prn medication and represcribing patterns. We are curious as to why some people seemed to need the antipsychotics,” said Dr. Brodaty. Although antipsychotics may be falling from favor, “there is a place for the use of antipsychotics, and our skill as clinicians is trying to identify which patients benefit from them,” Dr. Brodaty concluded.
—Erik Greb
Suggested Reading
Ballard C, Hanney
Declercq T, Petrovic M, Azermai M, et al. Withdrawal versus continuation of chronic antipsychotic drugs for behavioural and psychological symptoms in older people with dementia. Cochrane Database Syst Rev. 2013;(3):CD007726.
Devanand DP, Mintzer J, Schultz SK, et al. Relapse risk after discontinuation of risperidone in Alzheimer’s disease. N Engl J Med. 2012; 367(16): 1497–1507.
Rabey JM, Prokhorov T, Miniovitz A, et al. Effect of quetiapine in psychotic Parkinson’s disease patients: a double-blind labeled study of 3 months’ duration. Mov Disord. 2007;22(3):313-318.
TORONTO—Approximately 80% of elderly people with dementia in nursing homes do not have a reemergence of behavioral symptoms after cessation of antipsychotic drugs, according to research presented at the Alzheimer’s Association International Conference. The results may indicate that the antipsychotics are not necessary in these patients.
Many residents of nursing homes receive antipsychotics, but the drugs’ effect size is modest, said Henry Brodaty, MD, DSc, Professor of Aging and Mental Health and Codirector of the Center for Healthy Brain Aging at the University of New South Wales in Sydney. Antipsychotics can cause extrapyramidal side effects, falls, anticholinergic side effects such as worse cognition, urinary outflow difficulties and constipation, and orthostatic hypotension. The medicines also are associated with increased rates of stroke and death. In a UK study, Ballard and colleagues found no difference in the reemergence of behavioral symptoms between patients on continuous antipsychotics who were randomized to withdrawal and patients who continued their antipsychotics. However, Devenand and colleagues reported reemergence of behavioral symptoms when patients ceased antipsychotics.
Nurses Used Behavioral Strategies
To investigate the matter further, Dr. Brodaty and colleagues initiated the Halting Antipsychotics in Long-Term Care (HALT) study. The single-arm, 12-month, longitudinal study took place in 23 nursing homes in the Australian state of New South Wales. A nurse at each nursing home was named a “nurse champion” and identified residents who were receiving continuous antipsychotics. The researchers obtained permission from the nursing homes, the patients’ families, and the patients’ physicians to include the patients in the study.
During three days of training, each nurse champion learned how to manage behavioral symptoms and how to use person-centered care and behavioral strategies. These nurse champions subsequently trained the other nurses at their respective facilities in these techniques. Next, investigators monitored the patients for a month to establish their baseline conditions. Antipsychotics were stopped for all patients according to a protocol, and the researchers reassessed participants three, six, and 12 months later.
Participants’ mean age was about 85, and approximately two-thirds of patients were female. More than half of the sample was separated, widowed, or divorced. Approximately one-third of participants were married. About half of the sample had a high level of education, and half had a low level. A chart review indicated that one-third of patients had Alzheimer’s disease, and less than one-third had unspecified dementia. Smaller portions of the population had vascular or other dementias.
Participants received an average of 2.3 psychotropic medications and an average of nine medications for other indications. The total of about 11 medications per person was “about average,” said Dr. Brodaty. The most common antipsychotic was risperidone, followed by quetiapine, olanzapine, and haloperidol. Participants had been taking their antipsychotics for about two years and their current doses for at least one year. More than half of participants started antipsychotic medication after admission to the nursing home. About 20% of participants started antipsychotics during hospitalization before transfer to the nursing home, and 8% started antipsychotics in the community. For others, this information was not available.
Neuropsychiatric Symptoms Did Not Change
In all, 150 participants completed the prebaseline assessment, 139 completed the baseline assessment, 133 participants completed the three-month follow-up, and 118 participants completed the six-month follow-up. The 12-month follow-up was incomplete at the time of the presentation.
Of the 125 patients who had stopped their antipsychotics, the drugs were represcribed to 15 patients during the first three months. At six months, 10 of 118 patients for whom data were available had antipsychotics represcribed. In the final six months of the study, one patient had an antipsychotic represcribed. “In other words, almost 80% remained deprescribed, usually completely ceased,” said Dr. Brodaty. Many physicians wrote prn prescriptions, mainly for oxazepam as rescue medication, but the investigators have not analyzed the data to determine whether they were filled.
Despite the cessation of antipsychotics for most of the residents, there was no significant change in participants’ total Neuropsychiatric Inventory (NPI) score over the five follow-up visits, nor in their agitation (as measured by the Cohen Mansfield Agitation Inventory), aggression (as measured by the NPI), delusions, or hallucinations.
Should Standard of Care Change?
It was difficult to recruit patients for the study, said Dr. Brodaty. The investigators approached 58 nursing homes, and more than half declined to participate. Nurse champions also may not have included all patients on antipsychotics in the study. The results thus may have been influenced by selection bias. Staff at the nursing homes, physicians, and families appeared to be ignorant about the potential benefits of stopping antipsychotics such as preventing anticholinergic and other side effects.
“Inappropriate use of antipsychotics is an old story, yet we are still talking about it,” said Dr. Brodaty. “We have the knowledge and now we have to build on this knowledge and change practice. As a side issue, informed consent was pretty well lacking, and there is a lack of accountability about getting informed consent.”
Incentives for the owners, managers, and staff of nursing homes, as well as education for patients’ families, could promote person-centered care and reduce the inappropriate use of antipsychotics.
“Our next steps are to look at the prn medication and represcribing patterns. We are curious as to why some people seemed to need the antipsychotics,” said Dr. Brodaty. Although antipsychotics may be falling from favor, “there is a place for the use of antipsychotics, and our skill as clinicians is trying to identify which patients benefit from them,” Dr. Brodaty concluded.
—Erik Greb
Suggested Reading
Ballard C, Hanney
Declercq T, Petrovic M, Azermai M, et al. Withdrawal versus continuation of chronic antipsychotic drugs for behavioural and psychological symptoms in older people with dementia. Cochrane Database Syst Rev. 2013;(3):CD007726.
Devanand DP, Mintzer J, Schultz SK, et al. Relapse risk after discontinuation of risperidone in Alzheimer’s disease. N Engl J Med. 2012; 367(16): 1497–1507.
Rabey JM, Prokhorov T, Miniovitz A, et al. Effect of quetiapine in psychotic Parkinson’s disease patients: a double-blind labeled study of 3 months’ duration. Mov Disord. 2007;22(3):313-318.
TORONTO—Approximately 80% of elderly people with dementia in nursing homes do not have a reemergence of behavioral symptoms after cessation of antipsychotic drugs, according to research presented at the Alzheimer’s Association International Conference. The results may indicate that the antipsychotics are not necessary in these patients.
Many residents of nursing homes receive antipsychotics, but the drugs’ effect size is modest, said Henry Brodaty, MD, DSc, Professor of Aging and Mental Health and Codirector of the Center for Healthy Brain Aging at the University of New South Wales in Sydney. Antipsychotics can cause extrapyramidal side effects, falls, anticholinergic side effects such as worse cognition, urinary outflow difficulties and constipation, and orthostatic hypotension. The medicines also are associated with increased rates of stroke and death. In a UK study, Ballard and colleagues found no difference in the reemergence of behavioral symptoms between patients on continuous antipsychotics who were randomized to withdrawal and patients who continued their antipsychotics. However, Devenand and colleagues reported reemergence of behavioral symptoms when patients ceased antipsychotics.
Nurses Used Behavioral Strategies
To investigate the matter further, Dr. Brodaty and colleagues initiated the Halting Antipsychotics in Long-Term Care (HALT) study. The single-arm, 12-month, longitudinal study took place in 23 nursing homes in the Australian state of New South Wales. A nurse at each nursing home was named a “nurse champion” and identified residents who were receiving continuous antipsychotics. The researchers obtained permission from the nursing homes, the patients’ families, and the patients’ physicians to include the patients in the study.
During three days of training, each nurse champion learned how to manage behavioral symptoms and how to use person-centered care and behavioral strategies. These nurse champions subsequently trained the other nurses at their respective facilities in these techniques. Next, investigators monitored the patients for a month to establish their baseline conditions. Antipsychotics were stopped for all patients according to a protocol, and the researchers reassessed participants three, six, and 12 months later.
Participants’ mean age was about 85, and approximately two-thirds of patients were female. More than half of the sample was separated, widowed, or divorced. Approximately one-third of participants were married. About half of the sample had a high level of education, and half had a low level. A chart review indicated that one-third of patients had Alzheimer’s disease, and less than one-third had unspecified dementia. Smaller portions of the population had vascular or other dementias.
Participants received an average of 2.3 psychotropic medications and an average of nine medications for other indications. The total of about 11 medications per person was “about average,” said Dr. Brodaty. The most common antipsychotic was risperidone, followed by quetiapine, olanzapine, and haloperidol. Participants had been taking their antipsychotics for about two years and their current doses for at least one year. More than half of participants started antipsychotic medication after admission to the nursing home. About 20% of participants started antipsychotics during hospitalization before transfer to the nursing home, and 8% started antipsychotics in the community. For others, this information was not available.
Neuropsychiatric Symptoms Did Not Change
In all, 150 participants completed the prebaseline assessment, 139 completed the baseline assessment, 133 participants completed the three-month follow-up, and 118 participants completed the six-month follow-up. The 12-month follow-up was incomplete at the time of the presentation.
Of the 125 patients who had stopped their antipsychotics, the drugs were represcribed to 15 patients during the first three months. At six months, 10 of 118 patients for whom data were available had antipsychotics represcribed. In the final six months of the study, one patient had an antipsychotic represcribed. “In other words, almost 80% remained deprescribed, usually completely ceased,” said Dr. Brodaty. Many physicians wrote prn prescriptions, mainly for oxazepam as rescue medication, but the investigators have not analyzed the data to determine whether they were filled.
Despite the cessation of antipsychotics for most of the residents, there was no significant change in participants’ total Neuropsychiatric Inventory (NPI) score over the five follow-up visits, nor in their agitation (as measured by the Cohen Mansfield Agitation Inventory), aggression (as measured by the NPI), delusions, or hallucinations.
Should Standard of Care Change?
It was difficult to recruit patients for the study, said Dr. Brodaty. The investigators approached 58 nursing homes, and more than half declined to participate. Nurse champions also may not have included all patients on antipsychotics in the study. The results thus may have been influenced by selection bias. Staff at the nursing homes, physicians, and families appeared to be ignorant about the potential benefits of stopping antipsychotics such as preventing anticholinergic and other side effects.
“Inappropriate use of antipsychotics is an old story, yet we are still talking about it,” said Dr. Brodaty. “We have the knowledge and now we have to build on this knowledge and change practice. As a side issue, informed consent was pretty well lacking, and there is a lack of accountability about getting informed consent.”
Incentives for the owners, managers, and staff of nursing homes, as well as education for patients’ families, could promote person-centered care and reduce the inappropriate use of antipsychotics.
“Our next steps are to look at the prn medication and represcribing patterns. We are curious as to why some people seemed to need the antipsychotics,” said Dr. Brodaty. Although antipsychotics may be falling from favor, “there is a place for the use of antipsychotics, and our skill as clinicians is trying to identify which patients benefit from them,” Dr. Brodaty concluded.
—Erik Greb
Suggested Reading
Ballard C, Hanney
Declercq T, Petrovic M, Azermai M, et al. Withdrawal versus continuation of chronic antipsychotic drugs for behavioural and psychological symptoms in older people with dementia. Cochrane Database Syst Rev. 2013;(3):CD007726.
Devanand DP, Mintzer J, Schultz SK, et al. Relapse risk after discontinuation of risperidone in Alzheimer’s disease. N Engl J Med. 2012; 367(16): 1497–1507.
Rabey JM, Prokhorov T, Miniovitz A, et al. Effect of quetiapine in psychotic Parkinson’s disease patients: a double-blind labeled study of 3 months’ duration. Mov Disord. 2007;22(3):313-318.
Most children with JIA get different diagnosis as adults
Two-thirds of children diagnosed with juvenile idiopathic arthritis are classified later as having a different form of arthritis as adults, with 72% of them requiring disease-modifying medication and 13% forced into retirement, according to a cross-sectional analysis of a registry database.
But among patients with inactive disease, more than one-third are off medication, and the majority have either no or very mild disabilities, reported Filipa Oliveira-Ramos, MD, of Hospital de Santa Maria, Centro Hospitalar Lisboa Norte, Lisbon, and her colleagues (RMD Open 2016;2:e000304. doi: 10.1136/rmdopen-2016-000304).
The team used data from the Rheumatic Diseases Portuguese Register database to discern how rheumatic disease classifications evolve as people with JIA grow into adulthood. The analysis comprised 426 patients and examined fulfillment of adult classification criteria, function as assessed by the Health Assessment Questionnaire (HAQ), clinical disease characteristics as assessed by the Juvenile Arthritis Damage Index–articular (JADI-A) and Juvenile Arthritis Damage Index–extra-articular (JADI-E), and disease activity.
The patients were a mean of 34 years old at the time of the last visit entered into the database. The patients’ mean disease duration was 22.5 years, including 80% with at least 10 years and 24% with more than 30 years.
All had been diagnosed with JIA, at a mean age of about 10 years. Disease categories were persistent oligoarthritis (19%), extended oligoarthritis (14%), rheumatoid factor–positive polyarthritis (17%), rheumatoid factor–negative polyarthritis (18%), systemic disease (10%), enthesitis-related arthritis (19%), psoriatic arthritis (3%), and undifferentiated arthritis (1%).
A total of 72% of patients were still employed, although 13% had retired because of disease-related disability. Most (67%) still had active disease, and 72% were taking a disease-modifying antirheumatic drug.
JIA had evolved into numerous new diagnoses, the team observed. Most patients with systemic-onset JIA (92%) were classified as having adult Still’s disease – more than half (58%) with persistent systemic features and about 42% with predominately polyarticular involvement.
The majority of patients with RF-positive polyarthritis (96%) and of those with RF-negative polyarthritis as children (57%) fulfilled the adult criteria for rheumatoid arthritis.
Patients who had persistent oligoarthritis as children were, as adults, most likely be classified with spondyloarthritis (35%), although 59% remained unclassifiable.
Most of the patients with extended oligoarthritis as children were later classified as having either rheumatoid arthritis (39%) or spondyloarthritis (26%). Most patients with juvenile enthesitis-related arthritis were also reclassified as having spondyloarthritis (95%).
All of those with childhood psoriatic arthritis retained that classification as adults.
A smaller portion of patients (21%) were unclassified as adults, the investigators said. Most of these patients had RF-negative polyarticular or oligoarticular classifications as children.
In a series of multivariate analyses, the team found a number of significant associations with adult outcomes. After adjustment for International League of Associations for Rheumatology (ILAR) category, inactive adult disease was associated with shorter disease duration, less delay in diagnosis, a lower HAQ score, and less exposure to corticosteroids. A higher HAQ score was associated with a longer disease duration and exposure to biologics, while a lower HAQ was associated with the persistence of systemic disease features.
Higher JADI-A scores were associated with disability-related retirement, longer disease duration, and past or current use of biologics.
Another series of multivariate models assessed outcomes associated with inactive disease. Patients who were older at disease onset were more likely to have inactive disease as adults. A positive test for anticitrullinated protein antibodies decreased the likelihood of disease inactivity by 93%.
Finally, the investigators evaluated associations with function and clinical characteristics. Younger age at disease onset was associated with higher HAQ and JADI scores in adulthood. Patients with RF-positive polyarthritis and systemic-onset JIA were more likely to have worse JADI-A and JADI-E scores, compared with patients who had persistent oligoarthritis. Corticosteroid exposure was also predictive of worse extra-articular scores on the JADI.
“Understanding the way these juvenile diseases progress could add useful information for the ongoing discussion of a new classification capable of better unifying the language between pediatric and adult care,” the authors concluded.
None of the authors had financial disclosures.
[email protected]
On Twitter @alz_gal
This is an important article in that it highlights one of the fundamental flaws of the juvenile idiopathic arthritis classification.
The manifestations of the rheumatic diseases often evolve over time. There may be psoriasis before arthritis, but there also may be arthritis before psoriasis. Similarly, children with an ultimate diagnosis of Crohn’s disease may first present to the rheumatologist with arthritis.
The definition of JIA requires only the onset of arthritis lasting more than 3 weeks before 16 years of age with the exclusion of other obvious cause. A careful reading of the subclassification criteria quickly reveals that many children are “unclassifiable” because of family history or other factors.
The present study makes it clear that by the time they reach adulthood, a significant number of individuals who were told they had JIA will, in fact, meet criteria for a different classification. This would be of only casual interest were it not for the fact that these children will have previously been entered into databases about the natural history of JIA with erroneous classifications.
More worrisome is that fact that some will have been included in therapeutic trials with erroneous classifications as well. The pediatric rheumatology community would do well to recognize the urgent need for a reassessment of the classification criteria and nomenclature to better reflect the diversity of causes of childhood arthritis.
Thomas Lehman, MD, is chief of pediatric rheumatology at the Hospital for Special Surgery, New York. He has no relevant disclosures.
This is an important article in that it highlights one of the fundamental flaws of the juvenile idiopathic arthritis classification.
The manifestations of the rheumatic diseases often evolve over time. There may be psoriasis before arthritis, but there also may be arthritis before psoriasis. Similarly, children with an ultimate diagnosis of Crohn’s disease may first present to the rheumatologist with arthritis.
The definition of JIA requires only the onset of arthritis lasting more than 3 weeks before 16 years of age with the exclusion of other obvious cause. A careful reading of the subclassification criteria quickly reveals that many children are “unclassifiable” because of family history or other factors.
The present study makes it clear that by the time they reach adulthood, a significant number of individuals who were told they had JIA will, in fact, meet criteria for a different classification. This would be of only casual interest were it not for the fact that these children will have previously been entered into databases about the natural history of JIA with erroneous classifications.
More worrisome is that fact that some will have been included in therapeutic trials with erroneous classifications as well. The pediatric rheumatology community would do well to recognize the urgent need for a reassessment of the classification criteria and nomenclature to better reflect the diversity of causes of childhood arthritis.
Thomas Lehman, MD, is chief of pediatric rheumatology at the Hospital for Special Surgery, New York. He has no relevant disclosures.
This is an important article in that it highlights one of the fundamental flaws of the juvenile idiopathic arthritis classification.
The manifestations of the rheumatic diseases often evolve over time. There may be psoriasis before arthritis, but there also may be arthritis before psoriasis. Similarly, children with an ultimate diagnosis of Crohn’s disease may first present to the rheumatologist with arthritis.
The definition of JIA requires only the onset of arthritis lasting more than 3 weeks before 16 years of age with the exclusion of other obvious cause. A careful reading of the subclassification criteria quickly reveals that many children are “unclassifiable” because of family history or other factors.
The present study makes it clear that by the time they reach adulthood, a significant number of individuals who were told they had JIA will, in fact, meet criteria for a different classification. This would be of only casual interest were it not for the fact that these children will have previously been entered into databases about the natural history of JIA with erroneous classifications.
More worrisome is that fact that some will have been included in therapeutic trials with erroneous classifications as well. The pediatric rheumatology community would do well to recognize the urgent need for a reassessment of the classification criteria and nomenclature to better reflect the diversity of causes of childhood arthritis.
Thomas Lehman, MD, is chief of pediatric rheumatology at the Hospital for Special Surgery, New York. He has no relevant disclosures.
Two-thirds of children diagnosed with juvenile idiopathic arthritis are classified later as having a different form of arthritis as adults, with 72% of them requiring disease-modifying medication and 13% forced into retirement, according to a cross-sectional analysis of a registry database.
But among patients with inactive disease, more than one-third are off medication, and the majority have either no or very mild disabilities, reported Filipa Oliveira-Ramos, MD, of Hospital de Santa Maria, Centro Hospitalar Lisboa Norte, Lisbon, and her colleagues (RMD Open 2016;2:e000304. doi: 10.1136/rmdopen-2016-000304).
The team used data from the Rheumatic Diseases Portuguese Register database to discern how rheumatic disease classifications evolve as people with JIA grow into adulthood. The analysis comprised 426 patients and examined fulfillment of adult classification criteria, function as assessed by the Health Assessment Questionnaire (HAQ), clinical disease characteristics as assessed by the Juvenile Arthritis Damage Index–articular (JADI-A) and Juvenile Arthritis Damage Index–extra-articular (JADI-E), and disease activity.
The patients were a mean of 34 years old at the time of the last visit entered into the database. The patients’ mean disease duration was 22.5 years, including 80% with at least 10 years and 24% with more than 30 years.
All had been diagnosed with JIA, at a mean age of about 10 years. Disease categories were persistent oligoarthritis (19%), extended oligoarthritis (14%), rheumatoid factor–positive polyarthritis (17%), rheumatoid factor–negative polyarthritis (18%), systemic disease (10%), enthesitis-related arthritis (19%), psoriatic arthritis (3%), and undifferentiated arthritis (1%).
A total of 72% of patients were still employed, although 13% had retired because of disease-related disability. Most (67%) still had active disease, and 72% were taking a disease-modifying antirheumatic drug.
JIA had evolved into numerous new diagnoses, the team observed. Most patients with systemic-onset JIA (92%) were classified as having adult Still’s disease – more than half (58%) with persistent systemic features and about 42% with predominately polyarticular involvement.
The majority of patients with RF-positive polyarthritis (96%) and of those with RF-negative polyarthritis as children (57%) fulfilled the adult criteria for rheumatoid arthritis.
Patients who had persistent oligoarthritis as children were, as adults, most likely be classified with spondyloarthritis (35%), although 59% remained unclassifiable.
Most of the patients with extended oligoarthritis as children were later classified as having either rheumatoid arthritis (39%) or spondyloarthritis (26%). Most patients with juvenile enthesitis-related arthritis were also reclassified as having spondyloarthritis (95%).
All of those with childhood psoriatic arthritis retained that classification as adults.
A smaller portion of patients (21%) were unclassified as adults, the investigators said. Most of these patients had RF-negative polyarticular or oligoarticular classifications as children.
In a series of multivariate analyses, the team found a number of significant associations with adult outcomes. After adjustment for International League of Associations for Rheumatology (ILAR) category, inactive adult disease was associated with shorter disease duration, less delay in diagnosis, a lower HAQ score, and less exposure to corticosteroids. A higher HAQ score was associated with a longer disease duration and exposure to biologics, while a lower HAQ was associated with the persistence of systemic disease features.
Higher JADI-A scores were associated with disability-related retirement, longer disease duration, and past or current use of biologics.
Another series of multivariate models assessed outcomes associated with inactive disease. Patients who were older at disease onset were more likely to have inactive disease as adults. A positive test for anticitrullinated protein antibodies decreased the likelihood of disease inactivity by 93%.
Finally, the investigators evaluated associations with function and clinical characteristics. Younger age at disease onset was associated with higher HAQ and JADI scores in adulthood. Patients with RF-positive polyarthritis and systemic-onset JIA were more likely to have worse JADI-A and JADI-E scores, compared with patients who had persistent oligoarthritis. Corticosteroid exposure was also predictive of worse extra-articular scores on the JADI.
“Understanding the way these juvenile diseases progress could add useful information for the ongoing discussion of a new classification capable of better unifying the language between pediatric and adult care,” the authors concluded.
None of the authors had financial disclosures.
[email protected]
On Twitter @alz_gal
Two-thirds of children diagnosed with juvenile idiopathic arthritis are classified later as having a different form of arthritis as adults, with 72% of them requiring disease-modifying medication and 13% forced into retirement, according to a cross-sectional analysis of a registry database.
But among patients with inactive disease, more than one-third are off medication, and the majority have either no or very mild disabilities, reported Filipa Oliveira-Ramos, MD, of Hospital de Santa Maria, Centro Hospitalar Lisboa Norte, Lisbon, and her colleagues (RMD Open 2016;2:e000304. doi: 10.1136/rmdopen-2016-000304).
The team used data from the Rheumatic Diseases Portuguese Register database to discern how rheumatic disease classifications evolve as people with JIA grow into adulthood. The analysis comprised 426 patients and examined fulfillment of adult classification criteria, function as assessed by the Health Assessment Questionnaire (HAQ), clinical disease characteristics as assessed by the Juvenile Arthritis Damage Index–articular (JADI-A) and Juvenile Arthritis Damage Index–extra-articular (JADI-E), and disease activity.
The patients were a mean of 34 years old at the time of the last visit entered into the database. The patients’ mean disease duration was 22.5 years, including 80% with at least 10 years and 24% with more than 30 years.
All had been diagnosed with JIA, at a mean age of about 10 years. Disease categories were persistent oligoarthritis (19%), extended oligoarthritis (14%), rheumatoid factor–positive polyarthritis (17%), rheumatoid factor–negative polyarthritis (18%), systemic disease (10%), enthesitis-related arthritis (19%), psoriatic arthritis (3%), and undifferentiated arthritis (1%).
A total of 72% of patients were still employed, although 13% had retired because of disease-related disability. Most (67%) still had active disease, and 72% were taking a disease-modifying antirheumatic drug.
JIA had evolved into numerous new diagnoses, the team observed. Most patients with systemic-onset JIA (92%) were classified as having adult Still’s disease – more than half (58%) with persistent systemic features and about 42% with predominately polyarticular involvement.
The majority of patients with RF-positive polyarthritis (96%) and of those with RF-negative polyarthritis as children (57%) fulfilled the adult criteria for rheumatoid arthritis.
Patients who had persistent oligoarthritis as children were, as adults, most likely be classified with spondyloarthritis (35%), although 59% remained unclassifiable.
Most of the patients with extended oligoarthritis as children were later classified as having either rheumatoid arthritis (39%) or spondyloarthritis (26%). Most patients with juvenile enthesitis-related arthritis were also reclassified as having spondyloarthritis (95%).
All of those with childhood psoriatic arthritis retained that classification as adults.
A smaller portion of patients (21%) were unclassified as adults, the investigators said. Most of these patients had RF-negative polyarticular or oligoarticular classifications as children.
In a series of multivariate analyses, the team found a number of significant associations with adult outcomes. After adjustment for International League of Associations for Rheumatology (ILAR) category, inactive adult disease was associated with shorter disease duration, less delay in diagnosis, a lower HAQ score, and less exposure to corticosteroids. A higher HAQ score was associated with a longer disease duration and exposure to biologics, while a lower HAQ was associated with the persistence of systemic disease features.
Higher JADI-A scores were associated with disability-related retirement, longer disease duration, and past or current use of biologics.
Another series of multivariate models assessed outcomes associated with inactive disease. Patients who were older at disease onset were more likely to have inactive disease as adults. A positive test for anticitrullinated protein antibodies decreased the likelihood of disease inactivity by 93%.
Finally, the investigators evaluated associations with function and clinical characteristics. Younger age at disease onset was associated with higher HAQ and JADI scores in adulthood. Patients with RF-positive polyarthritis and systemic-onset JIA were more likely to have worse JADI-A and JADI-E scores, compared with patients who had persistent oligoarthritis. Corticosteroid exposure was also predictive of worse extra-articular scores on the JADI.
“Understanding the way these juvenile diseases progress could add useful information for the ongoing discussion of a new classification capable of better unifying the language between pediatric and adult care,” the authors concluded.
None of the authors had financial disclosures.
[email protected]
On Twitter @alz_gal
Key clinical point:
Major finding: The childhood diagnosis of JIA is reclassified to another form of arthritis in two-thirds of patients in adulthood.
Data source: The Rheumatic Diseases Portuguese Register.
Disclosures: None of the authors had financial disclosures.
Low-Dose Radiotherapy for Primary Cutaneous Anaplastic Large-Cell Lymphoma While on Low-Dose Methotrexate
CD30+ primary cutaneous lymphoproliferative disorders (pcLPDs) are the second most common cause of cutaneous T-cell lymphoma, accounting for approximately 25% to 30% of cases.1 These disorders comprise a spectrum that includes primary cutaneous anaplastic large-cell lymphoma (pcALCL); lymphomatoid papulosis (LyP); and borderline lesions, which share clinicopathologic features of both pcALCL and LyP. Lymphomatoid papulosis is characterized as chronic, recurrent, papular or papulonodular skin lesions that typically are multifocal and regress spontaneously within weeks to months, only leaving small scars with atrophy and/or hyperpigmentation.2 Cutaneous anaplastic large-cell lymphoma typically presents as solitary or grouped nodules or tumors that may undergo spontaneous partial or complete regression in approximately 25% of cases3 but often persist if not treated. Patients may have an array of lesions comprising the spectrum of CD30 pcLPDs.4
There is no curative therapy for CD30+ pcLPDs. Although active treatment is not necessary for LyP, low-dose methotrexate (MTX)(10–50 mg weekly) or phototherapy are the preferred initial suppressive therapies for symptomatic patients with scarring, facial lesions, or multiple symptomatic lesions.5 Observation with expectant follow-up is an option in pcALCL, though spontaneous regression is less likely than in LyP. For single or grouped pcALCL lesions, local radiation is the first-line therapy.6 Multifocal pcALCL lesions also can be treated with low-dose MTX,2,5 as in LyP, or local radiation to selected areas. Although local radiotherapy is considered a first-line treatment in pcALCL, there is limited evidence on its clinical efficacy as well as the optimal dose and technique. We report the complete response of refractory pcALCL lesions to low-dose radiation while remaining on MTX weekly without any adverse effects.
Case Report
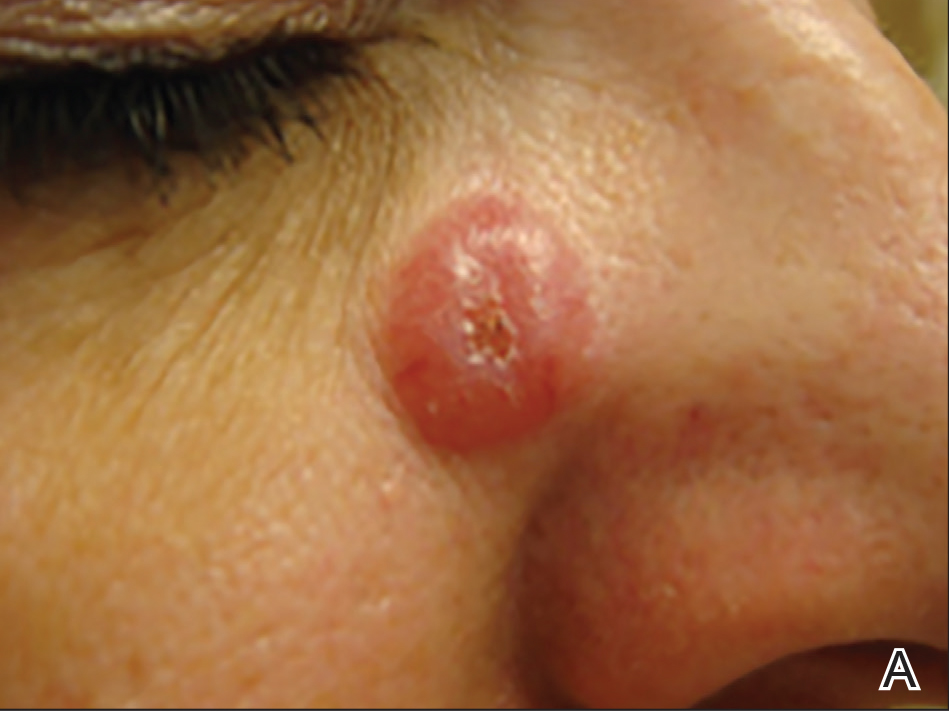
A 51-year-old woman presented with a 3-year history of CD30+ pcLPD manifesting primarily as pcALCL involving the head and neck, as well as LyP involving the head, arms, and trunk (T3N0M0). For 2 years her treatment regimen included clobetasol propionate cream 0.05% as needed for new lesions and 2 courses of standard-dose localized external beam radiation for larger pcALCL tumors on the right cheek and right side of the chin (Figure 1)(total dose for each course of treatment was 20 Gy and 36 Gy, respectively, each administered over 2–3 weeks). Because new unsightly papulonodules continued to develop on the patient’s face, she subsequently required low-dose oral MTX 30 mg once weekly for suppression of new lesions and was stable on this regimen for a year. However, she experienced an increase in LyP/pcALCL activity on the face during a 2-week break from MTX when she developed a herpes zoster infection on the right side of the forehead.
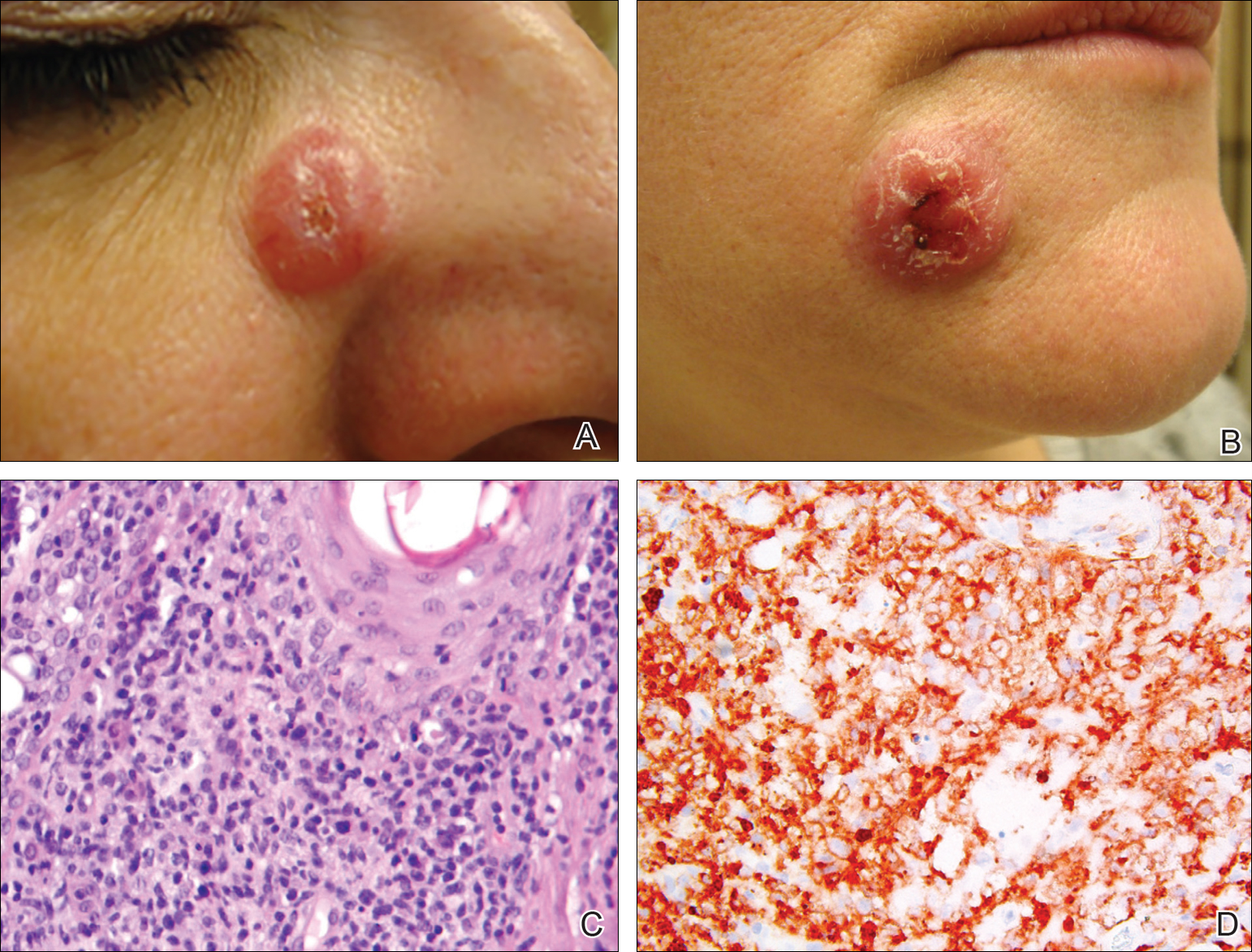
On physical examination 1 month later, 5 tiny pink papules scattered on the left eyebrow, left cheek, and left side of the chin were noted. She was advised to continue applying the clobetasol cream as needed and was restarted on MTX 10 mg once weekly. However, she developed 2 additional 1-cm nodules on the left side of the chin, neck, and shoulder. Methotrexate was increased to 30 mg once weekly over 2 weeks, which was the original dose prior to interruption, but the nodules grew to 1.5-cm in diameter. Due to their clinical appearance, the nodules were believed to be early pcALCL lesions (Figure 2A). Given the cosmetically sensitive location of the nodules, palliative radiotherapy was recommended rather than observe for possible regression. Based on a prior report by Neelis et al7 demonstrating efficacy of low-dose radiotherapy for cutaneous T-cell lymphoma and cutaneous B-cell lymphoma, we recommended starting with low radiation doses. Our patient was treated with 400 cGy twice to the left side of the chin and left side of the neck (800 cGy total at each site) while remaining on MTX 30 mg once weekly. This treatment was well tolerated without side effects and no evidence of radiation dermatitis. On follow-up examination 1 week later, the nodules had regressed and no new lesions were present (Figure 2B).
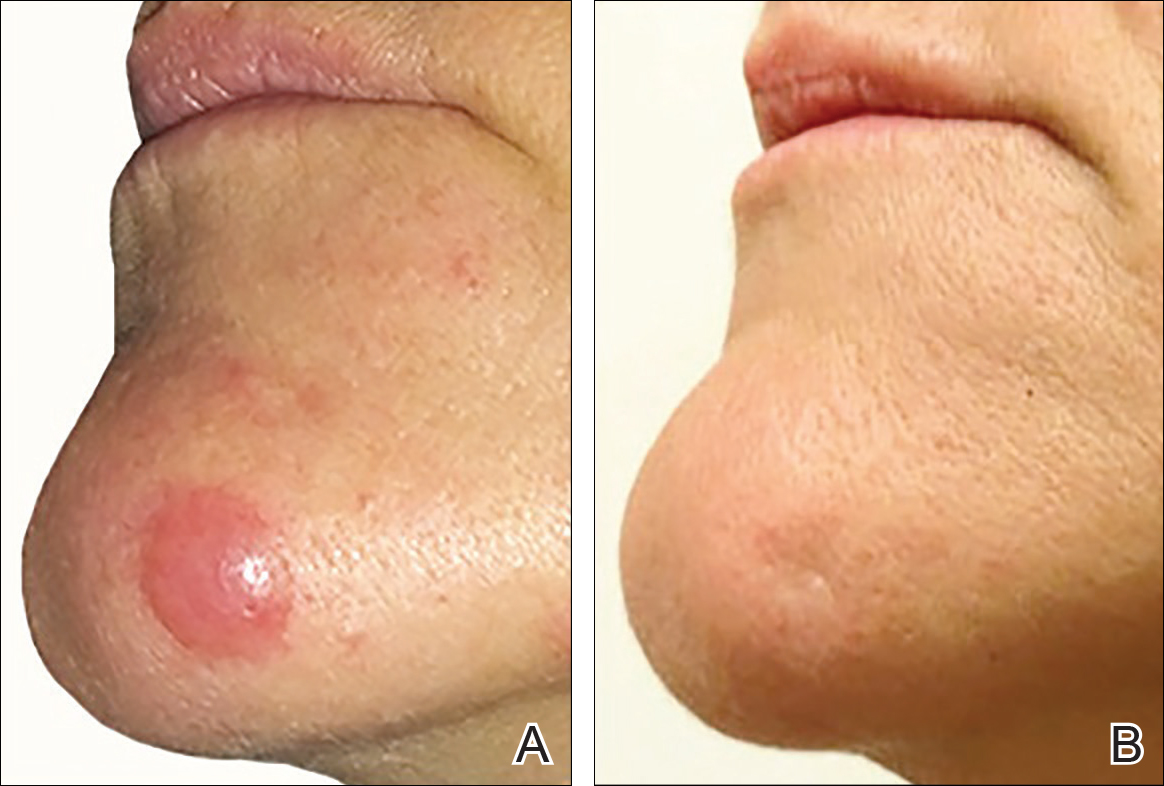
The patient has stayed on oral MTX and occasionally develops small lesions that quickly resolve with clobetasol cream. She has been followed for 3 years after radiotherapy and all 3 previously irradiated sites have remained recurrence free. Furthermore, she has not developed any new larger nodules or tumors and her MTX dose has been decreased to 15 mg once weekly.
Comment
Local radiotherapy is considered a first-line treatment of pcALCL; however, there is limited evidence on its clinical efficacy as well as the optimal dose and technique. Although no standard dose exists for pcALCL, the National Comprehensive Cancer Network guidelines8 recommend doses of 12 to 36 Gy in mycosis fungoides/Sézary syndrome subtypes of cutaneous T-cell lymphoma, which are consistent with guidelines published by the European Society for Medical Oncology.9 High complete response rates have been demonstrated in pcALCL at doses of 34 to 44 Gy6; however, lesions tend to recur elsewhere on the skin in 36% to 41% of patients despite treatment.2,10 Lower doses of radiation therapy would provide several advantages over higher-dose therapy if a complete response could be achieved without greatly increasing the local recurrence rate. In cases of local recurrence, low-dose radiation would more easily permit retreatment of lesions compared to higher doses of radiation. Similarly, in patients with multifocal pcALCL, lower doses of radiotherapy may allow for treatment of larger skin areas while limiting potential treatment risks. Furthermore, low-dose therapy would allow for treatments to be delivered more quickly and with less inconvenience to the patient who is likely to need multiple future treatments to other areas. Low-dose radiation has been described with a favorable efficacy profile for mycosis fungoides7,11 but has not been studied in patients with CD30+ pcLPDs.
Our case is notable because the patient remained on MTX during radiation therapy. B
Conclusion
We reported the use of low-dose radiation therapy for the treatment of localized pcALCL in a patient who remained on low-dose oral MTX. Additional studies will be necessary to more fully evaluate the efficacy of using low-dose radiation both as monotherapy and in combination with MTX for pcALCL.
- Willemze R, Jaffe ES, Burg G, et al. WHO-EORTC classification for cutaneous lymphomas. Blood. 2005;105:3768-3785.
- Bekkenk MW, Geelen FA, van Voorst Vader PC, et al. Primary and secondary cutaneous CD30+ lymphoproliferative disorders: a report from the Dutch Cutaneous Lymphoma Group on the long-term follow-up data of 219 patients and guidelines for diagnosis and treatment. Blood. 2000;95:3653-3661.
- Willemze R, Beljaards RC. Spectrum of primary cutaneous CD30 (Ki-1)-positive lymphoproliferative disorders: a proposal for classification and guidelines for management and treatment. J Am Acad Dermatol. 1993;28:973-980.
- Kadin ME. The spectrum of Ki-1+ cutaneous lymphomas. Curr Probl Dermatol. 1990;19:132-143.
- Vonderheid EC, Sajjadian A, Kadin ME. Methotrexate is effective therapy for lymphomatoid papulosis and other primary cutaneous CD30-positive lymphoproliferative disorders. J Am Acad Dermatol. 1996;34:470-481.
- Yu JB, McNiff JM, Lund MW, et al. Treatment of primary cutaneous CD30+ anaplastic large-cell lymphoma with radiation therapy. Int J Radiat Oncol Biol Phys. 2008;70:1542-1545.
- Neelis KJ, Schimmel EC, Vermeer MH, et al. Low-dose palliative radiotherapy B-cell and T-cell lymphomas. Int J Radiat Oncol Biol Phys. 2009;74:154-158.
- National Comprehensive Cancer Network. CD30 lymphoproliferative disorders section in non-Hodgkin’s lymphoma (Version 3.2016). http://www.nccn.org/professionals/physician_gls/pdf/nhl.pdf. Accessed September 26, 2016.
- Willemze R, Hodak E, Zinzani PL, et al; ESMO Guidelines Working Group. Primary cutaneous lymphomas: EMSO clinical practice guidelines for diagnosis, treatment, and follow-up [published online July 17, 2013]. Ann Onc. 2013;24(suppl 6):vi149-vi154.
- Liu HL, Hoppe RT, Kohler S, et al. CD30+ cutaneous lymphoproliferative disorders: the Stanford experience in lymphomatoid papulosis and primary cutaneous anaplastic large cell lymphoma. J Am Acad Dermatol. 2003;49:1049-1058.
- Harrison C, Young J, Navi D, et al. Revisiting low dose total skin electron beam radiotherapy in mycosis fungoides. Int J Radiat Oncol Biol Phys. 2011;81:651-657.
- Jaffe N, Farber S, Traggis D, et al. Favorable response of metastatic osteogenic sarcoma to pulse high-dose methotrexate with citrovorum rescue and radiation therapy. Cancer. 1973;31:1367-1373.
- Rosen G, Tefft M, Martinez A, et al. Combination chemotherapy and radiation therapy in the treatment of metastatic osteogenic sarcoma. Cancer. 1975;35:622-630.
- Kim YH, Aye MS, Fayos JV. Radiation necrosis of the scalp: a complication of cranial irradiation and methotrexate. Radiology. 1977;124:813-814.
CD30+ primary cutaneous lymphoproliferative disorders (pcLPDs) are the second most common cause of cutaneous T-cell lymphoma, accounting for approximately 25% to 30% of cases.1 These disorders comprise a spectrum that includes primary cutaneous anaplastic large-cell lymphoma (pcALCL); lymphomatoid papulosis (LyP); and borderline lesions, which share clinicopathologic features of both pcALCL and LyP. Lymphomatoid papulosis is characterized as chronic, recurrent, papular or papulonodular skin lesions that typically are multifocal and regress spontaneously within weeks to months, only leaving small scars with atrophy and/or hyperpigmentation.2 Cutaneous anaplastic large-cell lymphoma typically presents as solitary or grouped nodules or tumors that may undergo spontaneous partial or complete regression in approximately 25% of cases3 but often persist if not treated. Patients may have an array of lesions comprising the spectrum of CD30 pcLPDs.4
There is no curative therapy for CD30+ pcLPDs. Although active treatment is not necessary for LyP, low-dose methotrexate (MTX)(10–50 mg weekly) or phototherapy are the preferred initial suppressive therapies for symptomatic patients with scarring, facial lesions, or multiple symptomatic lesions.5 Observation with expectant follow-up is an option in pcALCL, though spontaneous regression is less likely than in LyP. For single or grouped pcALCL lesions, local radiation is the first-line therapy.6 Multifocal pcALCL lesions also can be treated with low-dose MTX,2,5 as in LyP, or local radiation to selected areas. Although local radiotherapy is considered a first-line treatment in pcALCL, there is limited evidence on its clinical efficacy as well as the optimal dose and technique. We report the complete response of refractory pcALCL lesions to low-dose radiation while remaining on MTX weekly without any adverse effects.
Case Report
A 51-year-old woman presented with a 3-year history of CD30+ pcLPD manifesting primarily as pcALCL involving the head and neck, as well as LyP involving the head, arms, and trunk (T3N0M0). For 2 years her treatment regimen included clobetasol propionate cream 0.05% as needed for new lesions and 2 courses of standard-dose localized external beam radiation for larger pcALCL tumors on the right cheek and right side of the chin (Figure 1)(total dose for each course of treatment was 20 Gy and 36 Gy, respectively, each administered over 2–3 weeks). Because new unsightly papulonodules continued to develop on the patient’s face, she subsequently required low-dose oral MTX 30 mg once weekly for suppression of new lesions and was stable on this regimen for a year. However, she experienced an increase in LyP/pcALCL activity on the face during a 2-week break from MTX when she developed a herpes zoster infection on the right side of the forehead.

On physical examination 1 month later, 5 tiny pink papules scattered on the left eyebrow, left cheek, and left side of the chin were noted. She was advised to continue applying the clobetasol cream as needed and was restarted on MTX 10 mg once weekly. However, she developed 2 additional 1-cm nodules on the left side of the chin, neck, and shoulder. Methotrexate was increased to 30 mg once weekly over 2 weeks, which was the original dose prior to interruption, but the nodules grew to 1.5-cm in diameter. Due to their clinical appearance, the nodules were believed to be early pcALCL lesions (Figure 2A). Given the cosmetically sensitive location of the nodules, palliative radiotherapy was recommended rather than observe for possible regression. Based on a prior report by Neelis et al7 demonstrating efficacy of low-dose radiotherapy for cutaneous T-cell lymphoma and cutaneous B-cell lymphoma, we recommended starting with low radiation doses. Our patient was treated with 400 cGy twice to the left side of the chin and left side of the neck (800 cGy total at each site) while remaining on MTX 30 mg once weekly. This treatment was well tolerated without side effects and no evidence of radiation dermatitis. On follow-up examination 1 week later, the nodules had regressed and no new lesions were present (Figure 2B).

The patient has stayed on oral MTX and occasionally develops small lesions that quickly resolve with clobetasol cream. She has been followed for 3 years after radiotherapy and all 3 previously irradiated sites have remained recurrence free. Furthermore, she has not developed any new larger nodules or tumors and her MTX dose has been decreased to 15 mg once weekly.
Comment
Local radiotherapy is considered a first-line treatment of pcALCL; however, there is limited evidence on its clinical efficacy as well as the optimal dose and technique. Although no standard dose exists for pcALCL, the National Comprehensive Cancer Network guidelines8 recommend doses of 12 to 36 Gy in mycosis fungoides/Sézary syndrome subtypes of cutaneous T-cell lymphoma, which are consistent with guidelines published by the European Society for Medical Oncology.9 High complete response rates have been demonstrated in pcALCL at doses of 34 to 44 Gy6; however, lesions tend to recur elsewhere on the skin in 36% to 41% of patients despite treatment.2,10 Lower doses of radiation therapy would provide several advantages over higher-dose therapy if a complete response could be achieved without greatly increasing the local recurrence rate. In cases of local recurrence, low-dose radiation would more easily permit retreatment of lesions compared to higher doses of radiation. Similarly, in patients with multifocal pcALCL, lower doses of radiotherapy may allow for treatment of larger skin areas while limiting potential treatment risks. Furthermore, low-dose therapy would allow for treatments to be delivered more quickly and with less inconvenience to the patient who is likely to need multiple future treatments to other areas. Low-dose radiation has been described with a favorable efficacy profile for mycosis fungoides7,11 but has not been studied in patients with CD30+ pcLPDs.
Our case is notable because the patient remained on MTX during radiation therapy. B
Conclusion
We reported the use of low-dose radiation therapy for the treatment of localized pcALCL in a patient who remained on low-dose oral MTX. Additional studies will be necessary to more fully evaluate the efficacy of using low-dose radiation both as monotherapy and in combination with MTX for pcALCL.
CD30+ primary cutaneous lymphoproliferative disorders (pcLPDs) are the second most common cause of cutaneous T-cell lymphoma, accounting for approximately 25% to 30% of cases.1 These disorders comprise a spectrum that includes primary cutaneous anaplastic large-cell lymphoma (pcALCL); lymphomatoid papulosis (LyP); and borderline lesions, which share clinicopathologic features of both pcALCL and LyP. Lymphomatoid papulosis is characterized as chronic, recurrent, papular or papulonodular skin lesions that typically are multifocal and regress spontaneously within weeks to months, only leaving small scars with atrophy and/or hyperpigmentation.2 Cutaneous anaplastic large-cell lymphoma typically presents as solitary or grouped nodules or tumors that may undergo spontaneous partial or complete regression in approximately 25% of cases3 but often persist if not treated. Patients may have an array of lesions comprising the spectrum of CD30 pcLPDs.4
There is no curative therapy for CD30+ pcLPDs. Although active treatment is not necessary for LyP, low-dose methotrexate (MTX)(10–50 mg weekly) or phototherapy are the preferred initial suppressive therapies for symptomatic patients with scarring, facial lesions, or multiple symptomatic lesions.5 Observation with expectant follow-up is an option in pcALCL, though spontaneous regression is less likely than in LyP. For single or grouped pcALCL lesions, local radiation is the first-line therapy.6 Multifocal pcALCL lesions also can be treated with low-dose MTX,2,5 as in LyP, or local radiation to selected areas. Although local radiotherapy is considered a first-line treatment in pcALCL, there is limited evidence on its clinical efficacy as well as the optimal dose and technique. We report the complete response of refractory pcALCL lesions to low-dose radiation while remaining on MTX weekly without any adverse effects.
Case Report
A 51-year-old woman presented with a 3-year history of CD30+ pcLPD manifesting primarily as pcALCL involving the head and neck, as well as LyP involving the head, arms, and trunk (T3N0M0). For 2 years her treatment regimen included clobetasol propionate cream 0.05% as needed for new lesions and 2 courses of standard-dose localized external beam radiation for larger pcALCL tumors on the right cheek and right side of the chin (Figure 1)(total dose for each course of treatment was 20 Gy and 36 Gy, respectively, each administered over 2–3 weeks). Because new unsightly papulonodules continued to develop on the patient’s face, she subsequently required low-dose oral MTX 30 mg once weekly for suppression of new lesions and was stable on this regimen for a year. However, she experienced an increase in LyP/pcALCL activity on the face during a 2-week break from MTX when she developed a herpes zoster infection on the right side of the forehead.

On physical examination 1 month later, 5 tiny pink papules scattered on the left eyebrow, left cheek, and left side of the chin were noted. She was advised to continue applying the clobetasol cream as needed and was restarted on MTX 10 mg once weekly. However, she developed 2 additional 1-cm nodules on the left side of the chin, neck, and shoulder. Methotrexate was increased to 30 mg once weekly over 2 weeks, which was the original dose prior to interruption, but the nodules grew to 1.5-cm in diameter. Due to their clinical appearance, the nodules were believed to be early pcALCL lesions (Figure 2A). Given the cosmetically sensitive location of the nodules, palliative radiotherapy was recommended rather than observe for possible regression. Based on a prior report by Neelis et al7 demonstrating efficacy of low-dose radiotherapy for cutaneous T-cell lymphoma and cutaneous B-cell lymphoma, we recommended starting with low radiation doses. Our patient was treated with 400 cGy twice to the left side of the chin and left side of the neck (800 cGy total at each site) while remaining on MTX 30 mg once weekly. This treatment was well tolerated without side effects and no evidence of radiation dermatitis. On follow-up examination 1 week later, the nodules had regressed and no new lesions were present (Figure 2B).

The patient has stayed on oral MTX and occasionally develops small lesions that quickly resolve with clobetasol cream. She has been followed for 3 years after radiotherapy and all 3 previously irradiated sites have remained recurrence free. Furthermore, she has not developed any new larger nodules or tumors and her MTX dose has been decreased to 15 mg once weekly.
Comment
Local radiotherapy is considered a first-line treatment of pcALCL; however, there is limited evidence on its clinical efficacy as well as the optimal dose and technique. Although no standard dose exists for pcALCL, the National Comprehensive Cancer Network guidelines8 recommend doses of 12 to 36 Gy in mycosis fungoides/Sézary syndrome subtypes of cutaneous T-cell lymphoma, which are consistent with guidelines published by the European Society for Medical Oncology.9 High complete response rates have been demonstrated in pcALCL at doses of 34 to 44 Gy6; however, lesions tend to recur elsewhere on the skin in 36% to 41% of patients despite treatment.2,10 Lower doses of radiation therapy would provide several advantages over higher-dose therapy if a complete response could be achieved without greatly increasing the local recurrence rate. In cases of local recurrence, low-dose radiation would more easily permit retreatment of lesions compared to higher doses of radiation. Similarly, in patients with multifocal pcALCL, lower doses of radiotherapy may allow for treatment of larger skin areas while limiting potential treatment risks. Furthermore, low-dose therapy would allow for treatments to be delivered more quickly and with less inconvenience to the patient who is likely to need multiple future treatments to other areas. Low-dose radiation has been described with a favorable efficacy profile for mycosis fungoides7,11 but has not been studied in patients with CD30+ pcLPDs.
Our case is notable because the patient remained on MTX during radiation therapy. B
Conclusion
We reported the use of low-dose radiation therapy for the treatment of localized pcALCL in a patient who remained on low-dose oral MTX. Additional studies will be necessary to more fully evaluate the efficacy of using low-dose radiation both as monotherapy and in combination with MTX for pcALCL.
- Willemze R, Jaffe ES, Burg G, et al. WHO-EORTC classification for cutaneous lymphomas. Blood. 2005;105:3768-3785.
- Bekkenk MW, Geelen FA, van Voorst Vader PC, et al. Primary and secondary cutaneous CD30+ lymphoproliferative disorders: a report from the Dutch Cutaneous Lymphoma Group on the long-term follow-up data of 219 patients and guidelines for diagnosis and treatment. Blood. 2000;95:3653-3661.
- Willemze R, Beljaards RC. Spectrum of primary cutaneous CD30 (Ki-1)-positive lymphoproliferative disorders: a proposal for classification and guidelines for management and treatment. J Am Acad Dermatol. 1993;28:973-980.
- Kadin ME. The spectrum of Ki-1+ cutaneous lymphomas. Curr Probl Dermatol. 1990;19:132-143.
- Vonderheid EC, Sajjadian A, Kadin ME. Methotrexate is effective therapy for lymphomatoid papulosis and other primary cutaneous CD30-positive lymphoproliferative disorders. J Am Acad Dermatol. 1996;34:470-481.
- Yu JB, McNiff JM, Lund MW, et al. Treatment of primary cutaneous CD30+ anaplastic large-cell lymphoma with radiation therapy. Int J Radiat Oncol Biol Phys. 2008;70:1542-1545.
- Neelis KJ, Schimmel EC, Vermeer MH, et al. Low-dose palliative radiotherapy B-cell and T-cell lymphomas. Int J Radiat Oncol Biol Phys. 2009;74:154-158.
- National Comprehensive Cancer Network. CD30 lymphoproliferative disorders section in non-Hodgkin’s lymphoma (Version 3.2016). http://www.nccn.org/professionals/physician_gls/pdf/nhl.pdf. Accessed September 26, 2016.
- Willemze R, Hodak E, Zinzani PL, et al; ESMO Guidelines Working Group. Primary cutaneous lymphomas: EMSO clinical practice guidelines for diagnosis, treatment, and follow-up [published online July 17, 2013]. Ann Onc. 2013;24(suppl 6):vi149-vi154.
- Liu HL, Hoppe RT, Kohler S, et al. CD30+ cutaneous lymphoproliferative disorders: the Stanford experience in lymphomatoid papulosis and primary cutaneous anaplastic large cell lymphoma. J Am Acad Dermatol. 2003;49:1049-1058.
- Harrison C, Young J, Navi D, et al. Revisiting low dose total skin electron beam radiotherapy in mycosis fungoides. Int J Radiat Oncol Biol Phys. 2011;81:651-657.
- Jaffe N, Farber S, Traggis D, et al. Favorable response of metastatic osteogenic sarcoma to pulse high-dose methotrexate with citrovorum rescue and radiation therapy. Cancer. 1973;31:1367-1373.
- Rosen G, Tefft M, Martinez A, et al. Combination chemotherapy and radiation therapy in the treatment of metastatic osteogenic sarcoma. Cancer. 1975;35:622-630.
- Kim YH, Aye MS, Fayos JV. Radiation necrosis of the scalp: a complication of cranial irradiation and methotrexate. Radiology. 1977;124:813-814.
- Willemze R, Jaffe ES, Burg G, et al. WHO-EORTC classification for cutaneous lymphomas. Blood. 2005;105:3768-3785.
- Bekkenk MW, Geelen FA, van Voorst Vader PC, et al. Primary and secondary cutaneous CD30+ lymphoproliferative disorders: a report from the Dutch Cutaneous Lymphoma Group on the long-term follow-up data of 219 patients and guidelines for diagnosis and treatment. Blood. 2000;95:3653-3661.
- Willemze R, Beljaards RC. Spectrum of primary cutaneous CD30 (Ki-1)-positive lymphoproliferative disorders: a proposal for classification and guidelines for management and treatment. J Am Acad Dermatol. 1993;28:973-980.
- Kadin ME. The spectrum of Ki-1+ cutaneous lymphomas. Curr Probl Dermatol. 1990;19:132-143.
- Vonderheid EC, Sajjadian A, Kadin ME. Methotrexate is effective therapy for lymphomatoid papulosis and other primary cutaneous CD30-positive lymphoproliferative disorders. J Am Acad Dermatol. 1996;34:470-481.
- Yu JB, McNiff JM, Lund MW, et al. Treatment of primary cutaneous CD30+ anaplastic large-cell lymphoma with radiation therapy. Int J Radiat Oncol Biol Phys. 2008;70:1542-1545.
- Neelis KJ, Schimmel EC, Vermeer MH, et al. Low-dose palliative radiotherapy B-cell and T-cell lymphomas. Int J Radiat Oncol Biol Phys. 2009;74:154-158.
- National Comprehensive Cancer Network. CD30 lymphoproliferative disorders section in non-Hodgkin’s lymphoma (Version 3.2016). http://www.nccn.org/professionals/physician_gls/pdf/nhl.pdf. Accessed September 26, 2016.
- Willemze R, Hodak E, Zinzani PL, et al; ESMO Guidelines Working Group. Primary cutaneous lymphomas: EMSO clinical practice guidelines for diagnosis, treatment, and follow-up [published online July 17, 2013]. Ann Onc. 2013;24(suppl 6):vi149-vi154.
- Liu HL, Hoppe RT, Kohler S, et al. CD30+ cutaneous lymphoproliferative disorders: the Stanford experience in lymphomatoid papulosis and primary cutaneous anaplastic large cell lymphoma. J Am Acad Dermatol. 2003;49:1049-1058.
- Harrison C, Young J, Navi D, et al. Revisiting low dose total skin electron beam radiotherapy in mycosis fungoides. Int J Radiat Oncol Biol Phys. 2011;81:651-657.
- Jaffe N, Farber S, Traggis D, et al. Favorable response of metastatic osteogenic sarcoma to pulse high-dose methotrexate with citrovorum rescue and radiation therapy. Cancer. 1973;31:1367-1373.
- Rosen G, Tefft M, Martinez A, et al. Combination chemotherapy and radiation therapy in the treatment of metastatic osteogenic sarcoma. Cancer. 1975;35:622-630.
- Kim YH, Aye MS, Fayos JV. Radiation necrosis of the scalp: a complication of cranial irradiation and methotrexate. Radiology. 1977;124:813-814.
Practice Points
- Cutaneous T-cell lymphoma tumors such as primary cutaneous anaplastic large-cell lymphoma can respond to low-dose radiation therapy, which enables future retreatment of sensitive sites.
- Low-dose radiation therapy requires a shorter course of therapy than traditional dosing, which is more convenient and less costly.
BEST PRACTICES: Multiplex Technology Delivers a Novel Tool to Assist in Ruling Out Systemic Lupus Erythematosus
Ellen Field, MD
Private Practice, Rheumatology
Lehigh Valley, Pennsylvania
Staff, Lehigh Valley Hospital
St. Luke’s Hospital
Sacred Heart Hospital
Lehigh Valley, Pennsylvania
Clinical Assistant Adjunct Professor of Medicine
Temple University School of Medicine
Philadelphia, Pennsylvania
Q&A with
| Peter Rumore, MD Rheumatology Associates of Long Island Smithtown, New York | Alan Kivitz, MD Altoona Arthritis and Osteoporosis Center Duncansville, Pennsylvania |
Click here to read this Best Practices supplement
Ellen Field, MD
Private Practice, Rheumatology
Lehigh Valley, Pennsylvania
Staff, Lehigh Valley Hospital
St. Luke’s Hospital
Sacred Heart Hospital
Lehigh Valley, Pennsylvania
Clinical Assistant Adjunct Professor of Medicine
Temple University School of Medicine
Philadelphia, Pennsylvania
Q&A with
| Peter Rumore, MD Rheumatology Associates of Long Island Smithtown, New York | Alan Kivitz, MD Altoona Arthritis and Osteoporosis Center Duncansville, Pennsylvania |
Click here to read this Best Practices supplement
Ellen Field, MD
Private Practice, Rheumatology
Lehigh Valley, Pennsylvania
Staff, Lehigh Valley Hospital
St. Luke’s Hospital
Sacred Heart Hospital
Lehigh Valley, Pennsylvania
Clinical Assistant Adjunct Professor of Medicine
Temple University School of Medicine
Philadelphia, Pennsylvania
Q&A with
| Peter Rumore, MD Rheumatology Associates of Long Island Smithtown, New York | Alan Kivitz, MD Altoona Arthritis and Osteoporosis Center Duncansville, Pennsylvania |
Click here to read this Best Practices supplement
Transcranial direct current stimulation enhances cognitive training in Parkinson’s
PORTLAND, ORE. – Combining transcranial direct current stimulation and cognitive training resulted in an improvement in a greater number of cognitive outcomes than either intervention alone in a small, randomized, controlled trial of patients with Parkinson’s disease and mild cognitive impairment.
Researchers at Curtin University in Perth, Western Australia, conducted the trial comparing the effects of standard (not individualized) cognitive training (SCT), tailored (individualized) cognitive training (TCT), transcranial direct current stimulation (tDCS), and a combination of tDCS with either form of cognitive training on cognitive outcomes, activities of daily living, and quality of life in such Parkinson’s patients with mild cognitive impairment. Previously, it was not known whether either form of cognitive training or tDCS or a combination of the two would be most efficacious in improving cognition in such patients.
Patients had cognitive deficits that did not interfere with functional independence and were responding to stable doses of antiparkinsonian medication. Forty-two eligible participants underwent neuropsychological testing at baseline and were randomly and equally assigned to one of six groups: SCT, TCT, tDCS, SCT+tDCS, TCT+tDCS, or control.
Cognitive training consisted of three 45-minute sessions per week for 4 weeks using Smartbrain Pro software in participants’ homes. tDCS involved constant 1.5 mA stimulation for 20 minutes in one session per week for 4 weeks at the university, with the anode placed over area F3 to stimulate the left dorsal lateral prefrontal cortex. Follow-up evaluations were at 12 weeks.
The following tests were used to evaluate each outcome: executive function – Stockings of Cambridge; attention/working memory – Stroop test; memory – paragraph recall; quality of life – PDQ-39; activities of daily living – Unified Parkinson’s Disease Rating Scale-II; and language – similarities test.
In general, combining tDCS with either form of cognitive training resulted in significantly greater improvements in more outcomes than any of the modalities alone. SCT showed positive results when compared against the control group in memory improvement at follow-up (effect size, 1.30), as well as quality of life and activities of daily living postintervention (effect sizes, 0.24 and 0.33, respectively). TCT showed benefits on quality of life at both time points (effect sizes 0.26 at postintervention and 0.12 at follow-up, respectively).
When combined with tDCS, SCT produced improvements in attention/working memory both postintervention and at 12-week follow-up (effect sizes, 0.60 and 0.24, respectively) as well as executive function at postintervention and follow-up (0.41 and 0.23). Improvement in activities of daily living and language were statistically significant only immediately postintervention.
Combining tDCS with TCT resulted in improvements postintervention and at follow-up on measures of memory (1.36 and 1.75) and executive function (0.19 and 0.92), as well as in language postintervention (1.06).
“The main takeaway was that the groups that completed both cognitive training and brain stimulation improved to a greater extent and in more outcomes than the groups that just completed the brain training or the stimulation individually,” Mr. Lawrence said. “The majority of the effects were shown immediately after the intervention, but some of the promising results ... actually maintained improvement at the 12-week follow-up, so that was after about 8 weeks, when they didn’t complete any intervention whatsoever.”
The improvements are probably clinically meaningful to patients since they themselves reported the outcomes on quality of life and activities of daily living scales, he said. He added that studies are coming out that look at the effect of brain stimulation and brain training at the same time, and they have shown improvement, but not many such studies have yet been done in Parkinson’s disease.
PORTLAND, ORE. – Combining transcranial direct current stimulation and cognitive training resulted in an improvement in a greater number of cognitive outcomes than either intervention alone in a small, randomized, controlled trial of patients with Parkinson’s disease and mild cognitive impairment.
Researchers at Curtin University in Perth, Western Australia, conducted the trial comparing the effects of standard (not individualized) cognitive training (SCT), tailored (individualized) cognitive training (TCT), transcranial direct current stimulation (tDCS), and a combination of tDCS with either form of cognitive training on cognitive outcomes, activities of daily living, and quality of life in such Parkinson’s patients with mild cognitive impairment. Previously, it was not known whether either form of cognitive training or tDCS or a combination of the two would be most efficacious in improving cognition in such patients.
Patients had cognitive deficits that did not interfere with functional independence and were responding to stable doses of antiparkinsonian medication. Forty-two eligible participants underwent neuropsychological testing at baseline and were randomly and equally assigned to one of six groups: SCT, TCT, tDCS, SCT+tDCS, TCT+tDCS, or control.
Cognitive training consisted of three 45-minute sessions per week for 4 weeks using Smartbrain Pro software in participants’ homes. tDCS involved constant 1.5 mA stimulation for 20 minutes in one session per week for 4 weeks at the university, with the anode placed over area F3 to stimulate the left dorsal lateral prefrontal cortex. Follow-up evaluations were at 12 weeks.
The following tests were used to evaluate each outcome: executive function – Stockings of Cambridge; attention/working memory – Stroop test; memory – paragraph recall; quality of life – PDQ-39; activities of daily living – Unified Parkinson’s Disease Rating Scale-II; and language – similarities test.
In general, combining tDCS with either form of cognitive training resulted in significantly greater improvements in more outcomes than any of the modalities alone. SCT showed positive results when compared against the control group in memory improvement at follow-up (effect size, 1.30), as well as quality of life and activities of daily living postintervention (effect sizes, 0.24 and 0.33, respectively). TCT showed benefits on quality of life at both time points (effect sizes 0.26 at postintervention and 0.12 at follow-up, respectively).
When combined with tDCS, SCT produced improvements in attention/working memory both postintervention and at 12-week follow-up (effect sizes, 0.60 and 0.24, respectively) as well as executive function at postintervention and follow-up (0.41 and 0.23). Improvement in activities of daily living and language were statistically significant only immediately postintervention.
Combining tDCS with TCT resulted in improvements postintervention and at follow-up on measures of memory (1.36 and 1.75) and executive function (0.19 and 0.92), as well as in language postintervention (1.06).
“The main takeaway was that the groups that completed both cognitive training and brain stimulation improved to a greater extent and in more outcomes than the groups that just completed the brain training or the stimulation individually,” Mr. Lawrence said. “The majority of the effects were shown immediately after the intervention, but some of the promising results ... actually maintained improvement at the 12-week follow-up, so that was after about 8 weeks, when they didn’t complete any intervention whatsoever.”
The improvements are probably clinically meaningful to patients since they themselves reported the outcomes on quality of life and activities of daily living scales, he said. He added that studies are coming out that look at the effect of brain stimulation and brain training at the same time, and they have shown improvement, but not many such studies have yet been done in Parkinson’s disease.
PORTLAND, ORE. – Combining transcranial direct current stimulation and cognitive training resulted in an improvement in a greater number of cognitive outcomes than either intervention alone in a small, randomized, controlled trial of patients with Parkinson’s disease and mild cognitive impairment.
Researchers at Curtin University in Perth, Western Australia, conducted the trial comparing the effects of standard (not individualized) cognitive training (SCT), tailored (individualized) cognitive training (TCT), transcranial direct current stimulation (tDCS), and a combination of tDCS with either form of cognitive training on cognitive outcomes, activities of daily living, and quality of life in such Parkinson’s patients with mild cognitive impairment. Previously, it was not known whether either form of cognitive training or tDCS or a combination of the two would be most efficacious in improving cognition in such patients.
Patients had cognitive deficits that did not interfere with functional independence and were responding to stable doses of antiparkinsonian medication. Forty-two eligible participants underwent neuropsychological testing at baseline and were randomly and equally assigned to one of six groups: SCT, TCT, tDCS, SCT+tDCS, TCT+tDCS, or control.
Cognitive training consisted of three 45-minute sessions per week for 4 weeks using Smartbrain Pro software in participants’ homes. tDCS involved constant 1.5 mA stimulation for 20 minutes in one session per week for 4 weeks at the university, with the anode placed over area F3 to stimulate the left dorsal lateral prefrontal cortex. Follow-up evaluations were at 12 weeks.
The following tests were used to evaluate each outcome: executive function – Stockings of Cambridge; attention/working memory – Stroop test; memory – paragraph recall; quality of life – PDQ-39; activities of daily living – Unified Parkinson’s Disease Rating Scale-II; and language – similarities test.
In general, combining tDCS with either form of cognitive training resulted in significantly greater improvements in more outcomes than any of the modalities alone. SCT showed positive results when compared against the control group in memory improvement at follow-up (effect size, 1.30), as well as quality of life and activities of daily living postintervention (effect sizes, 0.24 and 0.33, respectively). TCT showed benefits on quality of life at both time points (effect sizes 0.26 at postintervention and 0.12 at follow-up, respectively).
When combined with tDCS, SCT produced improvements in attention/working memory both postintervention and at 12-week follow-up (effect sizes, 0.60 and 0.24, respectively) as well as executive function at postintervention and follow-up (0.41 and 0.23). Improvement in activities of daily living and language were statistically significant only immediately postintervention.
Combining tDCS with TCT resulted in improvements postintervention and at follow-up on measures of memory (1.36 and 1.75) and executive function (0.19 and 0.92), as well as in language postintervention (1.06).
“The main takeaway was that the groups that completed both cognitive training and brain stimulation improved to a greater extent and in more outcomes than the groups that just completed the brain training or the stimulation individually,” Mr. Lawrence said. “The majority of the effects were shown immediately after the intervention, but some of the promising results ... actually maintained improvement at the 12-week follow-up, so that was after about 8 weeks, when they didn’t complete any intervention whatsoever.”
The improvements are probably clinically meaningful to patients since they themselves reported the outcomes on quality of life and activities of daily living scales, he said. He added that studies are coming out that look at the effect of brain stimulation and brain training at the same time, and they have shown improvement, but not many such studies have yet been done in Parkinson’s disease.
Key clinical point:
Major finding: tDCS adds cognitive benefit when combined with cognitive training alone.
Data source: Randomized, controlled trial of 42 patients with Parkinson’s disease and mild cognitive impairment.
Disclosures: There was no commercial funding of the study. Mr. Lawrence reported he had no financial disclosures.
Modern Indications, Results, and Global Trends in the Use of Unicompartmental Knee Arthroplasty and High Tibial Osteotomy in the Treatment of Isolated Medial Compartment Osteoarthritis
An increasingly number of patients with symptomatic isolated medial unicompartmental knee osteoarthritis (OA) are too young and too functionally active to be ideal candidates for total knee arthroplasty (TKA). Isolated medial compartment OA occurs in 10% to 29.5% of all cases, whereas the isolated lateral variant is less common, with a reported incidence of 1% to 7%.1,2 In 1961, Jackson and Waugh3 introduced the high tibial osteotomy (HTO) as a surgical treatment for single-compartment OA. This procedure is designed to increase the life span of articular cartilage by unloading and redistributing the mechanical forces over the nonaffected compartment. Unicompartmental knee arthroplasty (UKA) was introduced in the 1970s as an alternative to TKA or HTO for single-compartment OA.
Since the introduction of these methods, there has been debate about which patients are appropriate candidates for each procedure. Improved surgical techniques and implant designs have led surgeons to reexamine the selection criteria and contraindications for these procedures. Furthermore, given the increasing popularity and use of UKA, the question arises as to whether HTO still has a role in clinical practice in the surgical treatment of medial OA of the knee.
To clarify current ambiguities, we review the modern indications, subjective outcome scores, and survivorship results of UKA and HTO in the treatment of isolated medial compartment degeneration of the knee. In addition, in a thorough review of the literature, we evaluate global trends in the use of both methods.
High Tibial Osteotomy for Medial Compartment OA
Indications
Before the introduction of TKA and UKA for single-compartment OA, surgical management consisted of HTO. When the mechanical axis is slightly overcorrected, the medial compartment is decompressed, ensuring tissue viability and delaying progressive compartment degeneration.
Traditionally, HTO is indicated for young (age <60 years), normal-weight, active patients with radiographic single-compartment OA.6 The knee should be stable and have good range of motion (ROM; flexion >120°), and pain should be localized to the tibiofemoral joint line.
Over the past few decades, numerous authors have reported similar inclusion criteria, clarifying their definition. This definition should be further refined in order to optimize survivorship and clinical outcomes.
Confirming age as an inclusion criterion for HTO, Trieb and colleagues7 found that the risk of failure was significantly (P = .046) higher for HTO patients older than 65 years than for those younger than 65 years (relative risk, 1.5). This finding agrees with findings of other studies, which suggests that, in particular, young patients benefit from HTO.8-11
Moreover, there is a clear relation between HTO survival and obesity. In a study of 159 CWHTOs, Akizuki and colleagues12 reported that preoperative body mass index (BMI) higher than 27.5 kg/m2 was a significant risk factor for early failure. Using BMI higher than 30 kg/m2 as a threshold, Howells and colleagues9 found significantly inferior Knee Society Score (KSS) and Western Ontario and McMaster Universities Osteoarthritis Index (WOMAC) results for the obese group 5 years after HTO.
Radiographic evidence of severe preoperative compartment degeneration has been associated with early conversion to TKA. Flecher and colleagues11 and van Raaij and colleagues13 both concluded the best long-term survival grades are achieved in HTO patients with mild compartment OA (Ahlbäck14 grade I). The question then becomes whether these patients should be treated nonoperatively instead.15,16The literature supports strict adherence to inclusion criteria in the selection of a potential HTO candidate. Age, BMI, and the preoperative state of OA should be taken into account in order to optimize clinical outcome and survivorship results in patients about to undergo HTO.
Outcomes
Multiple authors have described or compared the midterm or long-term results of the various surgical HTO techniques. Howells and colleagues9 noted overall survival rates of 87% (5 years after CWHTO) and 79% (10 years after CWHTO). Over the 10-year postoperative period, there was significant deterioration in clinical outcome scores and survivorship. Others authors have had similar findings.17-19 van Raaij and colleagues13 found that the 10-year probability of survival after CWHTO was 75%. In 455 patients who underwent lateral CWHTO, Hui and colleagues8 found that 5-year probability of survival was 95%, 10-year probability was 79%, and 15-year probability was 56%. Niinimäki and colleagues10 used the Finnish Arthroplasty Register to report HTO survivorship at a national level. Using conversion to TKA as a cutoff, they noted 5-year survivorship of 89% and 10-year survivorship of 73%. To our knowledge, 2 groups, both in Japan, have reported substantially higher 15-year survival rates: 90%12 and 93%.20 The authors acknowledged that their results were significantly better than in other countries and that Japanese lifestyle, culture, and body habitus therefore require further investigation. At this time, it is not possible to compare their results with Western results.
In an attempt to compare the different survival rates of the various HTO techniques, Schallberger and colleagues21 conducted a retrospective study of OWHTOs and CWHTOs. At median follow-up of 16.5 years, comparative survival rates showed a trend of deterioration. Although data were limited, there were no significant differences in survival or functional outcome between the 2 techniques. In a recent randomized clinical trial, Duivenvoorden and colleagues5 compared these techniques’ midterm results (mean follow-up, 6 years). Clinical outcomes were not significantly different. There were more complications in the OWHTO group and more conversions to TKA in the CWHTO group. Considering these results, the authors suggested OWHTO without autologous bone graft is the best HTO treatment strategy for medial gonarthritis with varus malalignment of <12°.
The HTO results noted in these studies show a similar deteriorating trend; expected 10-year survivorship is 75%. Although modern implants and surgical techniques are being used, evidence supporting use of one surgical HTO method over another is lacking.
UKA for Medial Compartment OA
Indications
Since it was first introduced in the 1970s, use of UKA for single-compartment OA has been a subject of debate. The high failure rates reported at the time raised skepticism about the new treatment.22 Kozinn and Scott23 defined classic indications and contraindications. Indications included isolated medial or lateral compartment OA or osteonecrosis of the knee, age over 60 years, and weight under 82 kg. In addition, the angular deformity of the affected lower extremity had to be <15° and passively correctable to neutral at time of surgery. Last, the flexion contracture had to be <5°, and ideal ROM was 90°. Contraindications included high activity, age under 60 years, and inflammatory arthritis. Strict adherence led to improved implant survival and lower revision rates. Because of improved surgical techniques, modern implant designs, and accumulating experience with the procedure, the surgical indications for UKA have expanded. Exact thresholds for UKA inclusion, however, remain unclear.
The modern literature is overturning the traditional idea that UKA is not indicated for patients under age 60 years.23 Using KSS, Thompson and colleagues24 found that younger patients did better than older patients 2 years after UKA using various types of implants. Analyzing survivorship results, Heyse and colleagues25 concluded that UKA can be successful in patients under age 60 years and reported a 15-year survivorship rate of 85.6% and excellent outcome scores. Other authors have had similar findings.26-28
Evaluating the influence of weight, Thompson and colleagues24 found obese patients did not have a higher revision rate but did have slower progression of improvement 2 years after UKA. Cavaignac and colleagues29 concluded that, at minimum follow-up of 7 years (range, 7-22 years), weight did not influence UKA survivorship. Other authors30-33 have found no significant influence of BMI on survival.
Reports on preoperative radiographic parameters that can potentially influence UKA results are limited. In 113 medial UKAs studied by Niinimäki and colleagues,34 mild medial compartment degeneration, seen on preoperative radiographs, was associated with significantly higher failure rates. The authors concluded that other treatment options should be favored in the absence of severe isolated compartment OA.
Although the classic indications defined by Kozinn and Scott23 have yielded good to excellent UKA results, improvements in implants and surgical techniques35-38 have extended the criteria. The modern literature demonstrates that age and BMI should not be used as criteria for excluding UKA candidates. Radiographically, there should be significant isolated compartment degeneration in order to optimize patient-reported outcome and survivorship.
Outcomes
Improved implant designs and modern minimally invasive techniques have effected a change in outcome results and a renewed interest in implants. Over the past decade, multiple authors have described the various modern UKA implants and their survivorship. Reports published since UKA was introduced in the 1970s show a continual increase in implant survival. Koskinen and colleagues,39 using Finnish Arthroplasty Register data on 1819 UKAs performed between 1985 and 2003, found 10-year survival rates of 81% for Oxford implants (Zimmer Biomet), 79% for Miller-Galante II (Zimmer Biomet), 78% for Duracon (Howmedica), and 53% for PCA unicompartmental knee (Howmedica). Heyse and colleagues25 reported 10- and 15-year survivorship data (93.5% and 86.3%, respectively) for 223 patients under age 60 years at the time of their index surgery (Genesis Unicondylar implant, Smith & Nephew), performed between 1993 and 2005. KSS was good to excellent. Similar numbers in cohorts under age 60 years were reported by Schai and colleagues26 using the PFC system (Johnson & Johnson) and by Price and colleagues27 using the medial Oxford UKA. Both groups reported excellent survivorship rates: 93% at 2- to 6-year follow-up and 91% at 10-year follow-up. The outcome in older patients seems satisfactory as well. In another multicenter report, by Price and colleagues,40 medial Oxford UKAs had a 15-year survival rate of 93%. Berger and colleagues41 reported similar numbers for the Miller-Galante prosthesis. Survival rates were 98% (10 years) and 95.7% (13 years), and 92% of patients had good to excellent Hospital for Special Surgery knee scores.
Although various modern implants have had good to excellent results, the historical question of what type of UKA to use (mobile or fixed-bearing) remains unanswered. To try to address it, Peersman and colleagues42 performed a systematic review of 44 papers (9463 knees). The 2 implant types had comparable revision rates. Another recent retrospective study tried to determine what is crucial for implant survival: implant design or surgeon experience.43 The authors concluded that prosthetic component positioning is key. Other authors have reported high-volume centers are crucial for satisfactory UKA results and lower revision rates.44-46
Results of these studies indicate that, where UKAs are being performed in volume, 10-year survivorship rates higher than 90% and good to excellent outcomes can be expected.
UKA vs HTO
Cohort studies that have directly compared the 2 treatment modalities are scarce, and most have been retrospective. In a prospective study, Stukenborg-Colsman and colleagues47 randomized patients with medial compartment OA to undergo either CWHTO (32 patients) with a technique reported by Coventry48 or UKA (28 patients) with the unicondylar knee sliding prosthesis, Tübingen pattern (Aesculap), between 1988 and 1991. Patients were assessed 2.5, 4.5, and 7.5 years after surgery. More postoperative complications were noted in the HTO group. At 7- to 10-year follow-up, 71% of the HTO group and 65% of the UKA group had excellent KSS. Mean ROM was 103° after UKA (range, 35°-140°) and 117° after HTO (range, 85°-135°) during the same assessment. Although differences were not significant, Kaplan-Meier survival analysis was 60% for HTO and 77% for UKA at 10 years. Results were not promising for the implants used, compared with other implants, but the authors concluded that, because of improvements in implant designs and image-guided techniques, better long-term success can be expected with UKA than with HTO.
In another prospective study, Börjesson and colleagues49 evaluated pain during walking, ROM, British Orthopaedic Association (BOA) scores, and gait variables at 1- and 5-year follow-up. Patients with moderate medial OA (Ahlbäck14 grade I-III) were randomly selected to undergo CWHTO or UKA (Brigham, DePuy). There were no significant differences in BOA scores, ROM, or pain during walking between the 2 groups at 3 months, 1 year, and 5 years after surgery. Gait analysis showed a significant difference in favor of UKA only at 3 months after surgery. At 1- and 5-year follow-up, no significant differences were noted.
To clarify current ambiguities, Fu and colleagues50 performed a systematic review of all (11) comparative studies. These studies had a total of 5840 (5081 UKA, 759 HTO) patients. Although ROM was significantly better for the HTO group than the UKA group, the UKA group had significantly better functional results. Walking after surgery was significantly faster for the UKA group. The authors suggested the difference might be attributed to the different postoperative regimens—HTO patients wore a whole-leg plaster cast for 6 weeks, and UKA patients were allowed immediate postoperative weight-bearing. Regarding rates of survival and complications, pooled data showed no significant differences. Despite these results, the authors acknowledged the limitation of available randomized clinical trials and the multiple techniques and implants used. We share their assertion that larger prospective controlled trials are needed. These are crucial to getting a definitive answer regarding which of the 2 treatment strategies should be used for isolated compartment OA.
Current Trends in Use of UKA and HTO
Evaluation of national registries and recent reports showed a global shift in use of both HTO and UKA. Despite the lack of national HTO registries, a few reports have described use of TKA, UKA, and HTO in Western populations over the past 2 decades. Using 1998-2007 data from the Swedish Knee Arthroplasty Register, W-Dahl and colleagues51 found a 3-fold increase in UKA use, whereas HTO use was halved over the same period. Niinimäki and colleagues52 reported similar findings with the Finnish National Hospital Discharge Register. They noted a steady 6.8% annual decrease in osteotomies, whereas UKA use increased sharply after the Oxford UKA was introduced (Phase 3; Biomet). These findings are consistent with several reports from North America. In their epidemiologic analysis covering the period 1985-1990, Wright and colleagues53 found an 11% to 14% annual decrease in osteotomies among the elderly, compared with an annual decrease of only 3% to 4% among patients younger than 65 years. Nwachukwu and colleagues54 recently compared UKA and HTO practice patterns between 2007 and 2011, using data from a large US private payer insurance database. They noted an annual growth rate of 4.7% in UKA use, compared with an annual 3.9% decrease in HTO use. Furthermore, based on their subgroup analysis, they speculated there was a demographic shift toward UKA, as opposed to TKA, particularly in older women. Bolognesi and colleagues55 investigated further. Evaluating all Medicare beneficiaries who underwent knee arthroplasty in the United States between 2000 and 2009, they noted a 1.7-fold increase in TKA use and a 6.2-fold increase in UKA use. As there were no substantial changes in patient characteristics over that period, the authors hypothesized that a possible broadening of inclusion criteria may have led to the increased use of UKA.
There is a possible multifactorial explanation for the current global shift in favor of UKA. First, UKA was once a technically demanding procedure, but improved surgical techniques, image guidance, and robot assistance56 have made it relatively less difficult. Second, UKA surgery is associated with lower reported perioperative morbidities.57 We think these factors have contributed to the global trend of less HTO use and more UKA use in the treatment of unicompartmental OA.
Conclusion
The modern literature suggests the inclusion criteria for HTO have been well investigated and defined; the UKA criteria remain a matter of debate but seem to be expanding. Long-term survival results seem to favor UKA, though patient satisfaction with both procedures is good to excellent. The broadening range of inclusion criteria and consistent reports of durable outcomes, coupled with excellent patient satisfaction, likely explain the shift toward UKA in the treatment of isolated compartment degeneration.
Am J Orthop. 2016;45(6):E355-E361. Copyright Frontline Medical Communications Inc. 2016. All rights reserved.
1. Ledingham J, Regan M, Jones A, Doherty M. Radiographic patterns and associations of osteoarthritis of the knee in patients referred to hospital. Ann Rheum Dis. 1993;52(7): 520-526.
2. Wise BL, Niu J, Yang M, et al; Multicenter Osteoarthritis (MOST) Group. Patterns of compartment involvement in tibiofemoral osteoarthritis in men and women and in whites and African Americans. Arthritis Care Res. 2012;64(6): 847-852.
3. Jackson JP, Waugh W. Tibial osteotomy for osteoarthritis of the knee. J Bone Joint Surg Br. 1961;43:746-751.
4. Brouwer RW, Bierma-Zeinstra SM, van Raaij TM, Verhaar JA. Osteotomy for medial compartment arthritis of the knee using a closing wedge or an opening wedge controlled by a Puddu plate. A one-year randomised, controlled study. J Bone Joint Surg Br. 2006;88(11):1454-1459.
5. Duivenvoorden T, Brouwer RW, Baan A, et al. Comparison of closing-wedge and opening-wedge high tibial osteotomy for medial compartment osteoarthritis of the knee: a randomized controlled trial with a six-year follow-up. J Bone Joint Surg Am. 2014;96(17):1425-1432.
6. Hutchison CR, Cho B, Wong N, Agnidis Z, Gross AE. Proximal valgus tibial osteotomy for osteoarthritis of the knee. Instr Course Lect. 1999;48:131-134.
7. Trieb K, Grohs J, Hanslik-Schnabel B, Stulnig T, Panotopoulos J, Wanivenhaus A. Age predicts outcome of high-tibial osteotomy. Knee Surg Sports Traumatol Arthrosc. 2006;14(2):149-152.
8. Hui C, Salmon LJ, Kok A, et al. Long-term survival of high tibial osteotomy for medial compartment osteoarthritis of the knee. Am J Sports Med. 2011;39(1):64-70.
9. Howells NR, Salmon L, Waller A, Scanelli J, Pinczewski LA. The outcome at ten years of lateral closing-wedge high tibial osteotomy: determinants of survival and functional outcome. Bone Joint J Br. 2014;96(11):1491-1497.
10. Niinimäki TT, Eskelinen A, Mann BS, Junnila M, Ohtonen P, Leppilahti J. Survivorship of high tibial osteotomy in the treatment of osteoarthritis of the knee: Finnish registry-based study of 3195 knees. J Bone Joint Surg Br. 2012;94(11):1517-1521.
11. Flecher X, Parratte S, Aubaniac JM, Argenson JN. A 12-28-year followup study of closing wedge high tibial osteotomy. Clin Orthop Relat Res. 2006;(452):91-96.
12. Akizuki S, Shibakawa A, Takizawa T, Yamazaki I, Horiuchi H. The long-term outcome of high tibial osteotomy: a ten- to 20-year follow-up. J Bone Joint Surg Br. 2008;90(5):592-596.
13. van Raaij T, Reijman M, Brouwer RW, Jakma TS, Verhaar JN. Survival of closing-wedge high tibial osteotomy: good outcome in men with low-grade osteoarthritis after 10-16 years. Acta Orthop. 2008;79:230-234.
14. Ahlbäck S. Osteoarthrosis of the knee. A radiographic investigation. Acta Radiol Diagn. 1968;(suppl 277):7-72.
15. Bannuru RR, Natov NS, Obadan IE, Price LL, Schmid CH, McAlindon TE. Therapeutic trajectory of hyaluronic acid versus corticosteroids in the treatment of knee osteoarthritis: a systematic review and meta-analysis. Arthritis Rheum. 2009;61(12):1704-1711.
16. Evanich JD, Evanich CJ, Wright MB, Rydlewicz JA. Efficacy of intraarticular hyaluronic acid injections in knee osteoarthritis. Clin Orthop Relat Res. 2001;(390):173-181.
17. Naudie D, Bourne RB, Rorabeck CH, Bourne TJ. The Install Award. Survivorship of the high tibial valgus osteotomy. A 10- to -22-year followup study. Clin Orthop Relat Res. 1999;(367):18-27.
18. Sprenger TR, Doerzbacher JF. Tibial osteotomy for the treatment of varus gonarthrosis. Survival and failure analysis to twenty-two years. J Bone Joint Surg Br. 2003;85(3):469-474.
19. Billings A, Scott DF, Camargo MP, Hofmann AA. High tibial osteotomy with a calibrated osteotomy guide, rigid internal fixation, and early motion. Long-term follow-up. J Bone Joint Surg Am. 2000;82(1):70-79.
20. Koshino T, Yoshida T, Ara Y, Saito I, Saito T. Fifteen to twenty-eight years’ follow-up results of high tibial valgus osteotomy for osteoarthritic knee. Knee. 2004;11(6):439-444.
21. Schallberger A, Jacobi M, Wahl P, Maestretti G, Jakob RP. High tibial valgus osteotomy in unicompartmental medial osteoarthritis of the knee: a retrospective follow-up study over 13-21 years. Knee Surg Sports Traumatol Arthrosc. 2011;19(1):122-127.
22. Insall J, Aglietti P. A five to seven-year follow-up of unicondylar arthroplasty. J Bone Joint Surg Am. 1980;62(8):1329-1337.
23. Kozinn SC, Scott R. Unicondylar knee arthroplasty. J Bone Joint Surg Am. 1989;71(1):145-150.
24. Thompson SA, Liabaud B, Nellans KW, Geller JA. Factors associated with poor outcomes following unicompartmental knee arthroplasty: redefining the “classic” indications for surgery. J Arthroplasty. 2013;28(9):1561-1564.
25. Heyse TJ, Khefacha A, Peersman G, Cartier P. Survivorship of UKA in the middle-aged. Knee. 2012;19(5):585-591.
26. Schai PA, Suh JT, Thornhill TS, Scott RD. Unicompartmental knee arthroplasty in middle-aged patients: a 2- to 6-year follow-up evaluation. J Arthroplasty. 1998;13(4):365-372.
27. Price AJ, Dodd CA, Svard UG, Murray DW. Oxford medial unicompartmental knee arthroplasty in patients younger and older than 60 years of age. J Bone Joint Surg Br. 2005;87(11):1488-1492.
28. Pennington DW, Swienckowski JJ, Lutes WB, Drake GN. Unicompartmental knee arthroplasty in patients sixty years of age or younger. J Bone Joint Surg Am. 2003;85(10):1968-1973.
29. Cavaignac E, Lafontan V, Reina N, et al. Obesity has no adverse effect on the outcome of unicompartmental knee replacement at a minimum follow-up of seven years. Bone Joint J Br. 2013;95(8):1064-1068.
30. Tabor OB Jr, Tabor OB, Bernard M, Wan JY. Unicompartmental knee arthroplasty: long-term success in middle-age and obese patients. J Surg Orthop Adv. 2005;14(2):59-63.
31. Berend KR, Lombardi AV Jr, Adams JB. Obesity, young age, patellofemoral disease, and anterior knee pain: identifying the unicondylar arthroplasty patient in the United States. Orthopedics. 2007;30(5 suppl):19-23.
32. Xing Z, Katz J, Jiranek W. Unicompartmental knee arthroplasty: factors influencing the outcome. J Knee Surg. 2012;25(5):369-373.
33. Plate JF, Augart MA, Seyler TM, et al. Obesity has no effect on outcomes following unicompartmental knee arthroplasty [published online April 12, 2015]. Knee Surg Sports Traumatol Arthrosc. doi:10.1007/s00167-015-3597-5.
34. Niinimäki TT, Murray DW, Partanen J, Pajala A, Leppilahti JI. Unicompartmental knee arthroplasties implanted for osteoarthritis with partial loss of joint space have high re-operation rates. Knee. 2011;18(6):432-435.
35. Carlsson LV, Albrektsson BE, Regnér LR. Minimally invasive surgery vs conventional exposure using the Miller-Galante unicompartmental knee arthroplasty: a randomized radiostereometric study. J Arthroplasty. 2006;21(2):151-156.
36. Repicci JA. Mini-invasive knee unicompartmental arthroplasty: bone-sparing technique. Surg Technol Int. 2003;11:282-286.
37. Pandit H, Jenkins C, Barker K, Dodd CA, Murray DW. The Oxford medial unicompartmental knee replacement using a minimally-invasive approach. J Bone Joint Surg Br. 2006;88(1):54-60.
38. Romanowski MR, Repicci JA. Minimally invasive unicondylar arthroplasty: eight-year follow-up. J Knee Surg. 2002;15(1):17-22.
39. Koskinen E, Paavolainen P, Eskelinen A, Pulkkinen P, Remes V. Unicondylar knee replacement for primary osteoarthritis: a prospective follow-up study of 1,819 patients from the Finnish Arthroplasty Register. Acta Orthop. 2007;78(1):128-135.
40. Price AJ, Waite JC, Svard U. Long-term clinical results of the medial Oxford unicompartmental knee arthroplasty. Clin Orthop Relat Res. 2005;(435):171-180.
41. Berger RA, Meneghini RM, Jacobs JJ, et al. Results of unicompartmental knee arthroplasty at a minimum of ten years of follow-up. J Bone Joint Surg Am. 2005;87(5):999-1006.
42. Peersman G, Stuyts B, Vandenlangenbergh T, Cartier P, Fennema P. Fixed- versus mobile-bearing UKA: a systematic review and meta-analysis. Knee Surg Sports Traumatol Arthrosc. 2015;23(11):3296-3305.
43. Zambianchi F, Digennaro V, Giorgini A, et al. Surgeon’s experience influences UKA survivorship: a comparative study between all-poly and metal back designs. Knee Surg Sports Traumatol Arthrosc. 2015;23(7):2074-2080.
44. Robertsson O, Knutson K, Lewold S, Lidgren L. The routine of surgical management reduces failure after unicompartmental knee arthroplasty. J Bone Joint Surg Br. 2001;83(1):45-49.
45. Furnes O, Espehaug B, Lie SA, Vollset SE, Engesaeter LB, Havelin LI. Failure mechanisms after unicompartmental and tricompartmental primary knee replacement with cement. J Bone Joint Surg Am. 2007;89(3):519-525.
46. Robertsson O, Lidgren L. The short-term results of 3 common UKA implants during different periods in Sweden. J Arthroplasty. 2008;23(6):801-807.
47. Stukenborg-Colsman C, Wirth CJ, Lazovic D, Wefer A. High tibial osteotomy versus unicompartmental joint replacement in unicompartmental knee joint osteoarthritis: 7-10-year follow-up prospective randomised study. Knee. 2001;8(3):187-194.
48. Coventry MB. Osteotomy about the knee for degenerative and rheumatoid arthritis. J Bone Joint Surg Am. 1973;55(1):23-48.
49. Börjesson M, Weidenhielm L, Mattsson E, Olsson E. Gait and clinical measurements in patients with knee osteoarthritis after surgery: a prospective 5-year follow-up study. Knee. 2005;12(2):121-127.
50. Fu D, Li G, Chen K, Zhao Y, Hua Y, Cai Z. Comparison of high tibial osteotomy and unicompartmental knee arthroplasty in the treatment of unicompartmental osteoarthritis: a meta-analysis. J Arthroplasty. 2013;28(5):759-765.
51. W-Dahl A, Robertsson O, Lidgren L. Surgery for knee osteoarthritis in younger patients. Acta Orthop. 2010;81(2):161-164.
52. Niinimäki TT, Eskelinen A, Ohtonen P, Junnila M, Leppilahti J. Incidence of osteotomies around the knee for the treatment of knee osteoarthritis: a 22-year population-based study. Int Orthop. 2012;36(7):1399-1402.
53. Wright J, Heck D, Hawker G, et al. Rates of tibial osteotomies in Canada and the United States. Clin Orthop Relat Res. 1995;(319):266-275.
54. Nwachukwu BU, McCormick FM, Schairer WW, Frank RM, Provencher MT, Roche MW. Unicompartmental knee arthroplasty versus high tibial osteotomy: United States practice patterns for the surgical treatment of unicompartmental arthritis. J Arthroplasty. 2014;29(8):1586-1589.
55. Bolognesi MP, Greiner MA, Attarian DE, et al. Unicompartmental knee arthroplasty and total knee arthroplasty among Medicare beneficiaries, 2000 to 2009. J Bone Joint Surg Am. 2013;95(22):e174.
56. Pearle AD, O’Loughlin PF, Kendoff DO. Robot-assisted unicompartmental knee arthroplasty. J Arthroplasty. 2010;25(2):230-237.
57. Brown NM, Sheth NP, Davis K, et al. Total knee arthroplasty has higher postoperative morbidity than unicompartmental knee arthroplasty: a multicenter analysis. J Arthroplasty. 2012;27(8 suppl):86-90.
An increasingly number of patients with symptomatic isolated medial unicompartmental knee osteoarthritis (OA) are too young and too functionally active to be ideal candidates for total knee arthroplasty (TKA). Isolated medial compartment OA occurs in 10% to 29.5% of all cases, whereas the isolated lateral variant is less common, with a reported incidence of 1% to 7%.1,2 In 1961, Jackson and Waugh3 introduced the high tibial osteotomy (HTO) as a surgical treatment for single-compartment OA. This procedure is designed to increase the life span of articular cartilage by unloading and redistributing the mechanical forces over the nonaffected compartment. Unicompartmental knee arthroplasty (UKA) was introduced in the 1970s as an alternative to TKA or HTO for single-compartment OA.
Since the introduction of these methods, there has been debate about which patients are appropriate candidates for each procedure. Improved surgical techniques and implant designs have led surgeons to reexamine the selection criteria and contraindications for these procedures. Furthermore, given the increasing popularity and use of UKA, the question arises as to whether HTO still has a role in clinical practice in the surgical treatment of medial OA of the knee.
To clarify current ambiguities, we review the modern indications, subjective outcome scores, and survivorship results of UKA and HTO in the treatment of isolated medial compartment degeneration of the knee. In addition, in a thorough review of the literature, we evaluate global trends in the use of both methods.
High Tibial Osteotomy for Medial Compartment OA
Indications
Before the introduction of TKA and UKA for single-compartment OA, surgical management consisted of HTO. When the mechanical axis is slightly overcorrected, the medial compartment is decompressed, ensuring tissue viability and delaying progressive compartment degeneration.
Traditionally, HTO is indicated for young (age <60 years), normal-weight, active patients with radiographic single-compartment OA.6 The knee should be stable and have good range of motion (ROM; flexion >120°), and pain should be localized to the tibiofemoral joint line.
Over the past few decades, numerous authors have reported similar inclusion criteria, clarifying their definition. This definition should be further refined in order to optimize survivorship and clinical outcomes.
Confirming age as an inclusion criterion for HTO, Trieb and colleagues7 found that the risk of failure was significantly (P = .046) higher for HTO patients older than 65 years than for those younger than 65 years (relative risk, 1.5). This finding agrees with findings of other studies, which suggests that, in particular, young patients benefit from HTO.8-11
Moreover, there is a clear relation between HTO survival and obesity. In a study of 159 CWHTOs, Akizuki and colleagues12 reported that preoperative body mass index (BMI) higher than 27.5 kg/m2 was a significant risk factor for early failure. Using BMI higher than 30 kg/m2 as a threshold, Howells and colleagues9 found significantly inferior Knee Society Score (KSS) and Western Ontario and McMaster Universities Osteoarthritis Index (WOMAC) results for the obese group 5 years after HTO.
Radiographic evidence of severe preoperative compartment degeneration has been associated with early conversion to TKA. Flecher and colleagues11 and van Raaij and colleagues13 both concluded the best long-term survival grades are achieved in HTO patients with mild compartment OA (Ahlbäck14 grade I). The question then becomes whether these patients should be treated nonoperatively instead.15,16The literature supports strict adherence to inclusion criteria in the selection of a potential HTO candidate. Age, BMI, and the preoperative state of OA should be taken into account in order to optimize clinical outcome and survivorship results in patients about to undergo HTO.
Outcomes
Multiple authors have described or compared the midterm or long-term results of the various surgical HTO techniques. Howells and colleagues9 noted overall survival rates of 87% (5 years after CWHTO) and 79% (10 years after CWHTO). Over the 10-year postoperative period, there was significant deterioration in clinical outcome scores and survivorship. Others authors have had similar findings.17-19 van Raaij and colleagues13 found that the 10-year probability of survival after CWHTO was 75%. In 455 patients who underwent lateral CWHTO, Hui and colleagues8 found that 5-year probability of survival was 95%, 10-year probability was 79%, and 15-year probability was 56%. Niinimäki and colleagues10 used the Finnish Arthroplasty Register to report HTO survivorship at a national level. Using conversion to TKA as a cutoff, they noted 5-year survivorship of 89% and 10-year survivorship of 73%. To our knowledge, 2 groups, both in Japan, have reported substantially higher 15-year survival rates: 90%12 and 93%.20 The authors acknowledged that their results were significantly better than in other countries and that Japanese lifestyle, culture, and body habitus therefore require further investigation. At this time, it is not possible to compare their results with Western results.
In an attempt to compare the different survival rates of the various HTO techniques, Schallberger and colleagues21 conducted a retrospective study of OWHTOs and CWHTOs. At median follow-up of 16.5 years, comparative survival rates showed a trend of deterioration. Although data were limited, there were no significant differences in survival or functional outcome between the 2 techniques. In a recent randomized clinical trial, Duivenvoorden and colleagues5 compared these techniques’ midterm results (mean follow-up, 6 years). Clinical outcomes were not significantly different. There were more complications in the OWHTO group and more conversions to TKA in the CWHTO group. Considering these results, the authors suggested OWHTO without autologous bone graft is the best HTO treatment strategy for medial gonarthritis with varus malalignment of <12°.
The HTO results noted in these studies show a similar deteriorating trend; expected 10-year survivorship is 75%. Although modern implants and surgical techniques are being used, evidence supporting use of one surgical HTO method over another is lacking.
UKA for Medial Compartment OA
Indications
Since it was first introduced in the 1970s, use of UKA for single-compartment OA has been a subject of debate. The high failure rates reported at the time raised skepticism about the new treatment.22 Kozinn and Scott23 defined classic indications and contraindications. Indications included isolated medial or lateral compartment OA or osteonecrosis of the knee, age over 60 years, and weight under 82 kg. In addition, the angular deformity of the affected lower extremity had to be <15° and passively correctable to neutral at time of surgery. Last, the flexion contracture had to be <5°, and ideal ROM was 90°. Contraindications included high activity, age under 60 years, and inflammatory arthritis. Strict adherence led to improved implant survival and lower revision rates. Because of improved surgical techniques, modern implant designs, and accumulating experience with the procedure, the surgical indications for UKA have expanded. Exact thresholds for UKA inclusion, however, remain unclear.
The modern literature is overturning the traditional idea that UKA is not indicated for patients under age 60 years.23 Using KSS, Thompson and colleagues24 found that younger patients did better than older patients 2 years after UKA using various types of implants. Analyzing survivorship results, Heyse and colleagues25 concluded that UKA can be successful in patients under age 60 years and reported a 15-year survivorship rate of 85.6% and excellent outcome scores. Other authors have had similar findings.26-28
Evaluating the influence of weight, Thompson and colleagues24 found obese patients did not have a higher revision rate but did have slower progression of improvement 2 years after UKA. Cavaignac and colleagues29 concluded that, at minimum follow-up of 7 years (range, 7-22 years), weight did not influence UKA survivorship. Other authors30-33 have found no significant influence of BMI on survival.
Reports on preoperative radiographic parameters that can potentially influence UKA results are limited. In 113 medial UKAs studied by Niinimäki and colleagues,34 mild medial compartment degeneration, seen on preoperative radiographs, was associated with significantly higher failure rates. The authors concluded that other treatment options should be favored in the absence of severe isolated compartment OA.
Although the classic indications defined by Kozinn and Scott23 have yielded good to excellent UKA results, improvements in implants and surgical techniques35-38 have extended the criteria. The modern literature demonstrates that age and BMI should not be used as criteria for excluding UKA candidates. Radiographically, there should be significant isolated compartment degeneration in order to optimize patient-reported outcome and survivorship.
Outcomes
Improved implant designs and modern minimally invasive techniques have effected a change in outcome results and a renewed interest in implants. Over the past decade, multiple authors have described the various modern UKA implants and their survivorship. Reports published since UKA was introduced in the 1970s show a continual increase in implant survival. Koskinen and colleagues,39 using Finnish Arthroplasty Register data on 1819 UKAs performed between 1985 and 2003, found 10-year survival rates of 81% for Oxford implants (Zimmer Biomet), 79% for Miller-Galante II (Zimmer Biomet), 78% for Duracon (Howmedica), and 53% for PCA unicompartmental knee (Howmedica). Heyse and colleagues25 reported 10- and 15-year survivorship data (93.5% and 86.3%, respectively) for 223 patients under age 60 years at the time of their index surgery (Genesis Unicondylar implant, Smith & Nephew), performed between 1993 and 2005. KSS was good to excellent. Similar numbers in cohorts under age 60 years were reported by Schai and colleagues26 using the PFC system (Johnson & Johnson) and by Price and colleagues27 using the medial Oxford UKA. Both groups reported excellent survivorship rates: 93% at 2- to 6-year follow-up and 91% at 10-year follow-up. The outcome in older patients seems satisfactory as well. In another multicenter report, by Price and colleagues,40 medial Oxford UKAs had a 15-year survival rate of 93%. Berger and colleagues41 reported similar numbers for the Miller-Galante prosthesis. Survival rates were 98% (10 years) and 95.7% (13 years), and 92% of patients had good to excellent Hospital for Special Surgery knee scores.
Although various modern implants have had good to excellent results, the historical question of what type of UKA to use (mobile or fixed-bearing) remains unanswered. To try to address it, Peersman and colleagues42 performed a systematic review of 44 papers (9463 knees). The 2 implant types had comparable revision rates. Another recent retrospective study tried to determine what is crucial for implant survival: implant design or surgeon experience.43 The authors concluded that prosthetic component positioning is key. Other authors have reported high-volume centers are crucial for satisfactory UKA results and lower revision rates.44-46
Results of these studies indicate that, where UKAs are being performed in volume, 10-year survivorship rates higher than 90% and good to excellent outcomes can be expected.
UKA vs HTO
Cohort studies that have directly compared the 2 treatment modalities are scarce, and most have been retrospective. In a prospective study, Stukenborg-Colsman and colleagues47 randomized patients with medial compartment OA to undergo either CWHTO (32 patients) with a technique reported by Coventry48 or UKA (28 patients) with the unicondylar knee sliding prosthesis, Tübingen pattern (Aesculap), between 1988 and 1991. Patients were assessed 2.5, 4.5, and 7.5 years after surgery. More postoperative complications were noted in the HTO group. At 7- to 10-year follow-up, 71% of the HTO group and 65% of the UKA group had excellent KSS. Mean ROM was 103° after UKA (range, 35°-140°) and 117° after HTO (range, 85°-135°) during the same assessment. Although differences were not significant, Kaplan-Meier survival analysis was 60% for HTO and 77% for UKA at 10 years. Results were not promising for the implants used, compared with other implants, but the authors concluded that, because of improvements in implant designs and image-guided techniques, better long-term success can be expected with UKA than with HTO.
In another prospective study, Börjesson and colleagues49 evaluated pain during walking, ROM, British Orthopaedic Association (BOA) scores, and gait variables at 1- and 5-year follow-up. Patients with moderate medial OA (Ahlbäck14 grade I-III) were randomly selected to undergo CWHTO or UKA (Brigham, DePuy). There were no significant differences in BOA scores, ROM, or pain during walking between the 2 groups at 3 months, 1 year, and 5 years after surgery. Gait analysis showed a significant difference in favor of UKA only at 3 months after surgery. At 1- and 5-year follow-up, no significant differences were noted.
To clarify current ambiguities, Fu and colleagues50 performed a systematic review of all (11) comparative studies. These studies had a total of 5840 (5081 UKA, 759 HTO) patients. Although ROM was significantly better for the HTO group than the UKA group, the UKA group had significantly better functional results. Walking after surgery was significantly faster for the UKA group. The authors suggested the difference might be attributed to the different postoperative regimens—HTO patients wore a whole-leg plaster cast for 6 weeks, and UKA patients were allowed immediate postoperative weight-bearing. Regarding rates of survival and complications, pooled data showed no significant differences. Despite these results, the authors acknowledged the limitation of available randomized clinical trials and the multiple techniques and implants used. We share their assertion that larger prospective controlled trials are needed. These are crucial to getting a definitive answer regarding which of the 2 treatment strategies should be used for isolated compartment OA.
Current Trends in Use of UKA and HTO
Evaluation of national registries and recent reports showed a global shift in use of both HTO and UKA. Despite the lack of national HTO registries, a few reports have described use of TKA, UKA, and HTO in Western populations over the past 2 decades. Using 1998-2007 data from the Swedish Knee Arthroplasty Register, W-Dahl and colleagues51 found a 3-fold increase in UKA use, whereas HTO use was halved over the same period. Niinimäki and colleagues52 reported similar findings with the Finnish National Hospital Discharge Register. They noted a steady 6.8% annual decrease in osteotomies, whereas UKA use increased sharply after the Oxford UKA was introduced (Phase 3; Biomet). These findings are consistent with several reports from North America. In their epidemiologic analysis covering the period 1985-1990, Wright and colleagues53 found an 11% to 14% annual decrease in osteotomies among the elderly, compared with an annual decrease of only 3% to 4% among patients younger than 65 years. Nwachukwu and colleagues54 recently compared UKA and HTO practice patterns between 2007 and 2011, using data from a large US private payer insurance database. They noted an annual growth rate of 4.7% in UKA use, compared with an annual 3.9% decrease in HTO use. Furthermore, based on their subgroup analysis, they speculated there was a demographic shift toward UKA, as opposed to TKA, particularly in older women. Bolognesi and colleagues55 investigated further. Evaluating all Medicare beneficiaries who underwent knee arthroplasty in the United States between 2000 and 2009, they noted a 1.7-fold increase in TKA use and a 6.2-fold increase in UKA use. As there were no substantial changes in patient characteristics over that period, the authors hypothesized that a possible broadening of inclusion criteria may have led to the increased use of UKA.
There is a possible multifactorial explanation for the current global shift in favor of UKA. First, UKA was once a technically demanding procedure, but improved surgical techniques, image guidance, and robot assistance56 have made it relatively less difficult. Second, UKA surgery is associated with lower reported perioperative morbidities.57 We think these factors have contributed to the global trend of less HTO use and more UKA use in the treatment of unicompartmental OA.
Conclusion
The modern literature suggests the inclusion criteria for HTO have been well investigated and defined; the UKA criteria remain a matter of debate but seem to be expanding. Long-term survival results seem to favor UKA, though patient satisfaction with both procedures is good to excellent. The broadening range of inclusion criteria and consistent reports of durable outcomes, coupled with excellent patient satisfaction, likely explain the shift toward UKA in the treatment of isolated compartment degeneration.
Am J Orthop. 2016;45(6):E355-E361. Copyright Frontline Medical Communications Inc. 2016. All rights reserved.
An increasingly number of patients with symptomatic isolated medial unicompartmental knee osteoarthritis (OA) are too young and too functionally active to be ideal candidates for total knee arthroplasty (TKA). Isolated medial compartment OA occurs in 10% to 29.5% of all cases, whereas the isolated lateral variant is less common, with a reported incidence of 1% to 7%.1,2 In 1961, Jackson and Waugh3 introduced the high tibial osteotomy (HTO) as a surgical treatment for single-compartment OA. This procedure is designed to increase the life span of articular cartilage by unloading and redistributing the mechanical forces over the nonaffected compartment. Unicompartmental knee arthroplasty (UKA) was introduced in the 1970s as an alternative to TKA or HTO for single-compartment OA.
Since the introduction of these methods, there has been debate about which patients are appropriate candidates for each procedure. Improved surgical techniques and implant designs have led surgeons to reexamine the selection criteria and contraindications for these procedures. Furthermore, given the increasing popularity and use of UKA, the question arises as to whether HTO still has a role in clinical practice in the surgical treatment of medial OA of the knee.
To clarify current ambiguities, we review the modern indications, subjective outcome scores, and survivorship results of UKA and HTO in the treatment of isolated medial compartment degeneration of the knee. In addition, in a thorough review of the literature, we evaluate global trends in the use of both methods.
High Tibial Osteotomy for Medial Compartment OA
Indications
Before the introduction of TKA and UKA for single-compartment OA, surgical management consisted of HTO. When the mechanical axis is slightly overcorrected, the medial compartment is decompressed, ensuring tissue viability and delaying progressive compartment degeneration.
Traditionally, HTO is indicated for young (age <60 years), normal-weight, active patients with radiographic single-compartment OA.6 The knee should be stable and have good range of motion (ROM; flexion >120°), and pain should be localized to the tibiofemoral joint line.
Over the past few decades, numerous authors have reported similar inclusion criteria, clarifying their definition. This definition should be further refined in order to optimize survivorship and clinical outcomes.
Confirming age as an inclusion criterion for HTO, Trieb and colleagues7 found that the risk of failure was significantly (P = .046) higher for HTO patients older than 65 years than for those younger than 65 years (relative risk, 1.5). This finding agrees with findings of other studies, which suggests that, in particular, young patients benefit from HTO.8-11
Moreover, there is a clear relation between HTO survival and obesity. In a study of 159 CWHTOs, Akizuki and colleagues12 reported that preoperative body mass index (BMI) higher than 27.5 kg/m2 was a significant risk factor for early failure. Using BMI higher than 30 kg/m2 as a threshold, Howells and colleagues9 found significantly inferior Knee Society Score (KSS) and Western Ontario and McMaster Universities Osteoarthritis Index (WOMAC) results for the obese group 5 years after HTO.
Radiographic evidence of severe preoperative compartment degeneration has been associated with early conversion to TKA. Flecher and colleagues11 and van Raaij and colleagues13 both concluded the best long-term survival grades are achieved in HTO patients with mild compartment OA (Ahlbäck14 grade I). The question then becomes whether these patients should be treated nonoperatively instead.15,16The literature supports strict adherence to inclusion criteria in the selection of a potential HTO candidate. Age, BMI, and the preoperative state of OA should be taken into account in order to optimize clinical outcome and survivorship results in patients about to undergo HTO.
Outcomes
Multiple authors have described or compared the midterm or long-term results of the various surgical HTO techniques. Howells and colleagues9 noted overall survival rates of 87% (5 years after CWHTO) and 79% (10 years after CWHTO). Over the 10-year postoperative period, there was significant deterioration in clinical outcome scores and survivorship. Others authors have had similar findings.17-19 van Raaij and colleagues13 found that the 10-year probability of survival after CWHTO was 75%. In 455 patients who underwent lateral CWHTO, Hui and colleagues8 found that 5-year probability of survival was 95%, 10-year probability was 79%, and 15-year probability was 56%. Niinimäki and colleagues10 used the Finnish Arthroplasty Register to report HTO survivorship at a national level. Using conversion to TKA as a cutoff, they noted 5-year survivorship of 89% and 10-year survivorship of 73%. To our knowledge, 2 groups, both in Japan, have reported substantially higher 15-year survival rates: 90%12 and 93%.20 The authors acknowledged that their results were significantly better than in other countries and that Japanese lifestyle, culture, and body habitus therefore require further investigation. At this time, it is not possible to compare their results with Western results.
In an attempt to compare the different survival rates of the various HTO techniques, Schallberger and colleagues21 conducted a retrospective study of OWHTOs and CWHTOs. At median follow-up of 16.5 years, comparative survival rates showed a trend of deterioration. Although data were limited, there were no significant differences in survival or functional outcome between the 2 techniques. In a recent randomized clinical trial, Duivenvoorden and colleagues5 compared these techniques’ midterm results (mean follow-up, 6 years). Clinical outcomes were not significantly different. There were more complications in the OWHTO group and more conversions to TKA in the CWHTO group. Considering these results, the authors suggested OWHTO without autologous bone graft is the best HTO treatment strategy for medial gonarthritis with varus malalignment of <12°.
The HTO results noted in these studies show a similar deteriorating trend; expected 10-year survivorship is 75%. Although modern implants and surgical techniques are being used, evidence supporting use of one surgical HTO method over another is lacking.
UKA for Medial Compartment OA
Indications
Since it was first introduced in the 1970s, use of UKA for single-compartment OA has been a subject of debate. The high failure rates reported at the time raised skepticism about the new treatment.22 Kozinn and Scott23 defined classic indications and contraindications. Indications included isolated medial or lateral compartment OA or osteonecrosis of the knee, age over 60 years, and weight under 82 kg. In addition, the angular deformity of the affected lower extremity had to be <15° and passively correctable to neutral at time of surgery. Last, the flexion contracture had to be <5°, and ideal ROM was 90°. Contraindications included high activity, age under 60 years, and inflammatory arthritis. Strict adherence led to improved implant survival and lower revision rates. Because of improved surgical techniques, modern implant designs, and accumulating experience with the procedure, the surgical indications for UKA have expanded. Exact thresholds for UKA inclusion, however, remain unclear.
The modern literature is overturning the traditional idea that UKA is not indicated for patients under age 60 years.23 Using KSS, Thompson and colleagues24 found that younger patients did better than older patients 2 years after UKA using various types of implants. Analyzing survivorship results, Heyse and colleagues25 concluded that UKA can be successful in patients under age 60 years and reported a 15-year survivorship rate of 85.6% and excellent outcome scores. Other authors have had similar findings.26-28
Evaluating the influence of weight, Thompson and colleagues24 found obese patients did not have a higher revision rate but did have slower progression of improvement 2 years after UKA. Cavaignac and colleagues29 concluded that, at minimum follow-up of 7 years (range, 7-22 years), weight did not influence UKA survivorship. Other authors30-33 have found no significant influence of BMI on survival.
Reports on preoperative radiographic parameters that can potentially influence UKA results are limited. In 113 medial UKAs studied by Niinimäki and colleagues,34 mild medial compartment degeneration, seen on preoperative radiographs, was associated with significantly higher failure rates. The authors concluded that other treatment options should be favored in the absence of severe isolated compartment OA.
Although the classic indications defined by Kozinn and Scott23 have yielded good to excellent UKA results, improvements in implants and surgical techniques35-38 have extended the criteria. The modern literature demonstrates that age and BMI should not be used as criteria for excluding UKA candidates. Radiographically, there should be significant isolated compartment degeneration in order to optimize patient-reported outcome and survivorship.
Outcomes
Improved implant designs and modern minimally invasive techniques have effected a change in outcome results and a renewed interest in implants. Over the past decade, multiple authors have described the various modern UKA implants and their survivorship. Reports published since UKA was introduced in the 1970s show a continual increase in implant survival. Koskinen and colleagues,39 using Finnish Arthroplasty Register data on 1819 UKAs performed between 1985 and 2003, found 10-year survival rates of 81% for Oxford implants (Zimmer Biomet), 79% for Miller-Galante II (Zimmer Biomet), 78% for Duracon (Howmedica), and 53% for PCA unicompartmental knee (Howmedica). Heyse and colleagues25 reported 10- and 15-year survivorship data (93.5% and 86.3%, respectively) for 223 patients under age 60 years at the time of their index surgery (Genesis Unicondylar implant, Smith & Nephew), performed between 1993 and 2005. KSS was good to excellent. Similar numbers in cohorts under age 60 years were reported by Schai and colleagues26 using the PFC system (Johnson & Johnson) and by Price and colleagues27 using the medial Oxford UKA. Both groups reported excellent survivorship rates: 93% at 2- to 6-year follow-up and 91% at 10-year follow-up. The outcome in older patients seems satisfactory as well. In another multicenter report, by Price and colleagues,40 medial Oxford UKAs had a 15-year survival rate of 93%. Berger and colleagues41 reported similar numbers for the Miller-Galante prosthesis. Survival rates were 98% (10 years) and 95.7% (13 years), and 92% of patients had good to excellent Hospital for Special Surgery knee scores.
Although various modern implants have had good to excellent results, the historical question of what type of UKA to use (mobile or fixed-bearing) remains unanswered. To try to address it, Peersman and colleagues42 performed a systematic review of 44 papers (9463 knees). The 2 implant types had comparable revision rates. Another recent retrospective study tried to determine what is crucial for implant survival: implant design or surgeon experience.43 The authors concluded that prosthetic component positioning is key. Other authors have reported high-volume centers are crucial for satisfactory UKA results and lower revision rates.44-46
Results of these studies indicate that, where UKAs are being performed in volume, 10-year survivorship rates higher than 90% and good to excellent outcomes can be expected.
UKA vs HTO
Cohort studies that have directly compared the 2 treatment modalities are scarce, and most have been retrospective. In a prospective study, Stukenborg-Colsman and colleagues47 randomized patients with medial compartment OA to undergo either CWHTO (32 patients) with a technique reported by Coventry48 or UKA (28 patients) with the unicondylar knee sliding prosthesis, Tübingen pattern (Aesculap), between 1988 and 1991. Patients were assessed 2.5, 4.5, and 7.5 years after surgery. More postoperative complications were noted in the HTO group. At 7- to 10-year follow-up, 71% of the HTO group and 65% of the UKA group had excellent KSS. Mean ROM was 103° after UKA (range, 35°-140°) and 117° after HTO (range, 85°-135°) during the same assessment. Although differences were not significant, Kaplan-Meier survival analysis was 60% for HTO and 77% for UKA at 10 years. Results were not promising for the implants used, compared with other implants, but the authors concluded that, because of improvements in implant designs and image-guided techniques, better long-term success can be expected with UKA than with HTO.
In another prospective study, Börjesson and colleagues49 evaluated pain during walking, ROM, British Orthopaedic Association (BOA) scores, and gait variables at 1- and 5-year follow-up. Patients with moderate medial OA (Ahlbäck14 grade I-III) were randomly selected to undergo CWHTO or UKA (Brigham, DePuy). There were no significant differences in BOA scores, ROM, or pain during walking between the 2 groups at 3 months, 1 year, and 5 years after surgery. Gait analysis showed a significant difference in favor of UKA only at 3 months after surgery. At 1- and 5-year follow-up, no significant differences were noted.
To clarify current ambiguities, Fu and colleagues50 performed a systematic review of all (11) comparative studies. These studies had a total of 5840 (5081 UKA, 759 HTO) patients. Although ROM was significantly better for the HTO group than the UKA group, the UKA group had significantly better functional results. Walking after surgery was significantly faster for the UKA group. The authors suggested the difference might be attributed to the different postoperative regimens—HTO patients wore a whole-leg plaster cast for 6 weeks, and UKA patients were allowed immediate postoperative weight-bearing. Regarding rates of survival and complications, pooled data showed no significant differences. Despite these results, the authors acknowledged the limitation of available randomized clinical trials and the multiple techniques and implants used. We share their assertion that larger prospective controlled trials are needed. These are crucial to getting a definitive answer regarding which of the 2 treatment strategies should be used for isolated compartment OA.
Current Trends in Use of UKA and HTO
Evaluation of national registries and recent reports showed a global shift in use of both HTO and UKA. Despite the lack of national HTO registries, a few reports have described use of TKA, UKA, and HTO in Western populations over the past 2 decades. Using 1998-2007 data from the Swedish Knee Arthroplasty Register, W-Dahl and colleagues51 found a 3-fold increase in UKA use, whereas HTO use was halved over the same period. Niinimäki and colleagues52 reported similar findings with the Finnish National Hospital Discharge Register. They noted a steady 6.8% annual decrease in osteotomies, whereas UKA use increased sharply after the Oxford UKA was introduced (Phase 3; Biomet). These findings are consistent with several reports from North America. In their epidemiologic analysis covering the period 1985-1990, Wright and colleagues53 found an 11% to 14% annual decrease in osteotomies among the elderly, compared with an annual decrease of only 3% to 4% among patients younger than 65 years. Nwachukwu and colleagues54 recently compared UKA and HTO practice patterns between 2007 and 2011, using data from a large US private payer insurance database. They noted an annual growth rate of 4.7% in UKA use, compared with an annual 3.9% decrease in HTO use. Furthermore, based on their subgroup analysis, they speculated there was a demographic shift toward UKA, as opposed to TKA, particularly in older women. Bolognesi and colleagues55 investigated further. Evaluating all Medicare beneficiaries who underwent knee arthroplasty in the United States between 2000 and 2009, they noted a 1.7-fold increase in TKA use and a 6.2-fold increase in UKA use. As there were no substantial changes in patient characteristics over that period, the authors hypothesized that a possible broadening of inclusion criteria may have led to the increased use of UKA.
There is a possible multifactorial explanation for the current global shift in favor of UKA. First, UKA was once a technically demanding procedure, but improved surgical techniques, image guidance, and robot assistance56 have made it relatively less difficult. Second, UKA surgery is associated with lower reported perioperative morbidities.57 We think these factors have contributed to the global trend of less HTO use and more UKA use in the treatment of unicompartmental OA.
Conclusion
The modern literature suggests the inclusion criteria for HTO have been well investigated and defined; the UKA criteria remain a matter of debate but seem to be expanding. Long-term survival results seem to favor UKA, though patient satisfaction with both procedures is good to excellent. The broadening range of inclusion criteria and consistent reports of durable outcomes, coupled with excellent patient satisfaction, likely explain the shift toward UKA in the treatment of isolated compartment degeneration.
Am J Orthop. 2016;45(6):E355-E361. Copyright Frontline Medical Communications Inc. 2016. All rights reserved.
1. Ledingham J, Regan M, Jones A, Doherty M. Radiographic patterns and associations of osteoarthritis of the knee in patients referred to hospital. Ann Rheum Dis. 1993;52(7): 520-526.
2. Wise BL, Niu J, Yang M, et al; Multicenter Osteoarthritis (MOST) Group. Patterns of compartment involvement in tibiofemoral osteoarthritis in men and women and in whites and African Americans. Arthritis Care Res. 2012;64(6): 847-852.
3. Jackson JP, Waugh W. Tibial osteotomy for osteoarthritis of the knee. J Bone Joint Surg Br. 1961;43:746-751.
4. Brouwer RW, Bierma-Zeinstra SM, van Raaij TM, Verhaar JA. Osteotomy for medial compartment arthritis of the knee using a closing wedge or an opening wedge controlled by a Puddu plate. A one-year randomised, controlled study. J Bone Joint Surg Br. 2006;88(11):1454-1459.
5. Duivenvoorden T, Brouwer RW, Baan A, et al. Comparison of closing-wedge and opening-wedge high tibial osteotomy for medial compartment osteoarthritis of the knee: a randomized controlled trial with a six-year follow-up. J Bone Joint Surg Am. 2014;96(17):1425-1432.
6. Hutchison CR, Cho B, Wong N, Agnidis Z, Gross AE. Proximal valgus tibial osteotomy for osteoarthritis of the knee. Instr Course Lect. 1999;48:131-134.
7. Trieb K, Grohs J, Hanslik-Schnabel B, Stulnig T, Panotopoulos J, Wanivenhaus A. Age predicts outcome of high-tibial osteotomy. Knee Surg Sports Traumatol Arthrosc. 2006;14(2):149-152.
8. Hui C, Salmon LJ, Kok A, et al. Long-term survival of high tibial osteotomy for medial compartment osteoarthritis of the knee. Am J Sports Med. 2011;39(1):64-70.
9. Howells NR, Salmon L, Waller A, Scanelli J, Pinczewski LA. The outcome at ten years of lateral closing-wedge high tibial osteotomy: determinants of survival and functional outcome. Bone Joint J Br. 2014;96(11):1491-1497.
10. Niinimäki TT, Eskelinen A, Mann BS, Junnila M, Ohtonen P, Leppilahti J. Survivorship of high tibial osteotomy in the treatment of osteoarthritis of the knee: Finnish registry-based study of 3195 knees. J Bone Joint Surg Br. 2012;94(11):1517-1521.
11. Flecher X, Parratte S, Aubaniac JM, Argenson JN. A 12-28-year followup study of closing wedge high tibial osteotomy. Clin Orthop Relat Res. 2006;(452):91-96.
12. Akizuki S, Shibakawa A, Takizawa T, Yamazaki I, Horiuchi H. The long-term outcome of high tibial osteotomy: a ten- to 20-year follow-up. J Bone Joint Surg Br. 2008;90(5):592-596.
13. van Raaij T, Reijman M, Brouwer RW, Jakma TS, Verhaar JN. Survival of closing-wedge high tibial osteotomy: good outcome in men with low-grade osteoarthritis after 10-16 years. Acta Orthop. 2008;79:230-234.
14. Ahlbäck S. Osteoarthrosis of the knee. A radiographic investigation. Acta Radiol Diagn. 1968;(suppl 277):7-72.
15. Bannuru RR, Natov NS, Obadan IE, Price LL, Schmid CH, McAlindon TE. Therapeutic trajectory of hyaluronic acid versus corticosteroids in the treatment of knee osteoarthritis: a systematic review and meta-analysis. Arthritis Rheum. 2009;61(12):1704-1711.
16. Evanich JD, Evanich CJ, Wright MB, Rydlewicz JA. Efficacy of intraarticular hyaluronic acid injections in knee osteoarthritis. Clin Orthop Relat Res. 2001;(390):173-181.
17. Naudie D, Bourne RB, Rorabeck CH, Bourne TJ. The Install Award. Survivorship of the high tibial valgus osteotomy. A 10- to -22-year followup study. Clin Orthop Relat Res. 1999;(367):18-27.
18. Sprenger TR, Doerzbacher JF. Tibial osteotomy for the treatment of varus gonarthrosis. Survival and failure analysis to twenty-two years. J Bone Joint Surg Br. 2003;85(3):469-474.
19. Billings A, Scott DF, Camargo MP, Hofmann AA. High tibial osteotomy with a calibrated osteotomy guide, rigid internal fixation, and early motion. Long-term follow-up. J Bone Joint Surg Am. 2000;82(1):70-79.
20. Koshino T, Yoshida T, Ara Y, Saito I, Saito T. Fifteen to twenty-eight years’ follow-up results of high tibial valgus osteotomy for osteoarthritic knee. Knee. 2004;11(6):439-444.
21. Schallberger A, Jacobi M, Wahl P, Maestretti G, Jakob RP. High tibial valgus osteotomy in unicompartmental medial osteoarthritis of the knee: a retrospective follow-up study over 13-21 years. Knee Surg Sports Traumatol Arthrosc. 2011;19(1):122-127.
22. Insall J, Aglietti P. A five to seven-year follow-up of unicondylar arthroplasty. J Bone Joint Surg Am. 1980;62(8):1329-1337.
23. Kozinn SC, Scott R. Unicondylar knee arthroplasty. J Bone Joint Surg Am. 1989;71(1):145-150.
24. Thompson SA, Liabaud B, Nellans KW, Geller JA. Factors associated with poor outcomes following unicompartmental knee arthroplasty: redefining the “classic” indications for surgery. J Arthroplasty. 2013;28(9):1561-1564.
25. Heyse TJ, Khefacha A, Peersman G, Cartier P. Survivorship of UKA in the middle-aged. Knee. 2012;19(5):585-591.
26. Schai PA, Suh JT, Thornhill TS, Scott RD. Unicompartmental knee arthroplasty in middle-aged patients: a 2- to 6-year follow-up evaluation. J Arthroplasty. 1998;13(4):365-372.
27. Price AJ, Dodd CA, Svard UG, Murray DW. Oxford medial unicompartmental knee arthroplasty in patients younger and older than 60 years of age. J Bone Joint Surg Br. 2005;87(11):1488-1492.
28. Pennington DW, Swienckowski JJ, Lutes WB, Drake GN. Unicompartmental knee arthroplasty in patients sixty years of age or younger. J Bone Joint Surg Am. 2003;85(10):1968-1973.
29. Cavaignac E, Lafontan V, Reina N, et al. Obesity has no adverse effect on the outcome of unicompartmental knee replacement at a minimum follow-up of seven years. Bone Joint J Br. 2013;95(8):1064-1068.
30. Tabor OB Jr, Tabor OB, Bernard M, Wan JY. Unicompartmental knee arthroplasty: long-term success in middle-age and obese patients. J Surg Orthop Adv. 2005;14(2):59-63.
31. Berend KR, Lombardi AV Jr, Adams JB. Obesity, young age, patellofemoral disease, and anterior knee pain: identifying the unicondylar arthroplasty patient in the United States. Orthopedics. 2007;30(5 suppl):19-23.
32. Xing Z, Katz J, Jiranek W. Unicompartmental knee arthroplasty: factors influencing the outcome. J Knee Surg. 2012;25(5):369-373.
33. Plate JF, Augart MA, Seyler TM, et al. Obesity has no effect on outcomes following unicompartmental knee arthroplasty [published online April 12, 2015]. Knee Surg Sports Traumatol Arthrosc. doi:10.1007/s00167-015-3597-5.
34. Niinimäki TT, Murray DW, Partanen J, Pajala A, Leppilahti JI. Unicompartmental knee arthroplasties implanted for osteoarthritis with partial loss of joint space have high re-operation rates. Knee. 2011;18(6):432-435.
35. Carlsson LV, Albrektsson BE, Regnér LR. Minimally invasive surgery vs conventional exposure using the Miller-Galante unicompartmental knee arthroplasty: a randomized radiostereometric study. J Arthroplasty. 2006;21(2):151-156.
36. Repicci JA. Mini-invasive knee unicompartmental arthroplasty: bone-sparing technique. Surg Technol Int. 2003;11:282-286.
37. Pandit H, Jenkins C, Barker K, Dodd CA, Murray DW. The Oxford medial unicompartmental knee replacement using a minimally-invasive approach. J Bone Joint Surg Br. 2006;88(1):54-60.
38. Romanowski MR, Repicci JA. Minimally invasive unicondylar arthroplasty: eight-year follow-up. J Knee Surg. 2002;15(1):17-22.
39. Koskinen E, Paavolainen P, Eskelinen A, Pulkkinen P, Remes V. Unicondylar knee replacement for primary osteoarthritis: a prospective follow-up study of 1,819 patients from the Finnish Arthroplasty Register. Acta Orthop. 2007;78(1):128-135.
40. Price AJ, Waite JC, Svard U. Long-term clinical results of the medial Oxford unicompartmental knee arthroplasty. Clin Orthop Relat Res. 2005;(435):171-180.
41. Berger RA, Meneghini RM, Jacobs JJ, et al. Results of unicompartmental knee arthroplasty at a minimum of ten years of follow-up. J Bone Joint Surg Am. 2005;87(5):999-1006.
42. Peersman G, Stuyts B, Vandenlangenbergh T, Cartier P, Fennema P. Fixed- versus mobile-bearing UKA: a systematic review and meta-analysis. Knee Surg Sports Traumatol Arthrosc. 2015;23(11):3296-3305.
43. Zambianchi F, Digennaro V, Giorgini A, et al. Surgeon’s experience influences UKA survivorship: a comparative study between all-poly and metal back designs. Knee Surg Sports Traumatol Arthrosc. 2015;23(7):2074-2080.
44. Robertsson O, Knutson K, Lewold S, Lidgren L. The routine of surgical management reduces failure after unicompartmental knee arthroplasty. J Bone Joint Surg Br. 2001;83(1):45-49.
45. Furnes O, Espehaug B, Lie SA, Vollset SE, Engesaeter LB, Havelin LI. Failure mechanisms after unicompartmental and tricompartmental primary knee replacement with cement. J Bone Joint Surg Am. 2007;89(3):519-525.
46. Robertsson O, Lidgren L. The short-term results of 3 common UKA implants during different periods in Sweden. J Arthroplasty. 2008;23(6):801-807.
47. Stukenborg-Colsman C, Wirth CJ, Lazovic D, Wefer A. High tibial osteotomy versus unicompartmental joint replacement in unicompartmental knee joint osteoarthritis: 7-10-year follow-up prospective randomised study. Knee. 2001;8(3):187-194.
48. Coventry MB. Osteotomy about the knee for degenerative and rheumatoid arthritis. J Bone Joint Surg Am. 1973;55(1):23-48.
49. Börjesson M, Weidenhielm L, Mattsson E, Olsson E. Gait and clinical measurements in patients with knee osteoarthritis after surgery: a prospective 5-year follow-up study. Knee. 2005;12(2):121-127.
50. Fu D, Li G, Chen K, Zhao Y, Hua Y, Cai Z. Comparison of high tibial osteotomy and unicompartmental knee arthroplasty in the treatment of unicompartmental osteoarthritis: a meta-analysis. J Arthroplasty. 2013;28(5):759-765.
51. W-Dahl A, Robertsson O, Lidgren L. Surgery for knee osteoarthritis in younger patients. Acta Orthop. 2010;81(2):161-164.
52. Niinimäki TT, Eskelinen A, Ohtonen P, Junnila M, Leppilahti J. Incidence of osteotomies around the knee for the treatment of knee osteoarthritis: a 22-year population-based study. Int Orthop. 2012;36(7):1399-1402.
53. Wright J, Heck D, Hawker G, et al. Rates of tibial osteotomies in Canada and the United States. Clin Orthop Relat Res. 1995;(319):266-275.
54. Nwachukwu BU, McCormick FM, Schairer WW, Frank RM, Provencher MT, Roche MW. Unicompartmental knee arthroplasty versus high tibial osteotomy: United States practice patterns for the surgical treatment of unicompartmental arthritis. J Arthroplasty. 2014;29(8):1586-1589.
55. Bolognesi MP, Greiner MA, Attarian DE, et al. Unicompartmental knee arthroplasty and total knee arthroplasty among Medicare beneficiaries, 2000 to 2009. J Bone Joint Surg Am. 2013;95(22):e174.
56. Pearle AD, O’Loughlin PF, Kendoff DO. Robot-assisted unicompartmental knee arthroplasty. J Arthroplasty. 2010;25(2):230-237.
57. Brown NM, Sheth NP, Davis K, et al. Total knee arthroplasty has higher postoperative morbidity than unicompartmental knee arthroplasty: a multicenter analysis. J Arthroplasty. 2012;27(8 suppl):86-90.
1. Ledingham J, Regan M, Jones A, Doherty M. Radiographic patterns and associations of osteoarthritis of the knee in patients referred to hospital. Ann Rheum Dis. 1993;52(7): 520-526.
2. Wise BL, Niu J, Yang M, et al; Multicenter Osteoarthritis (MOST) Group. Patterns of compartment involvement in tibiofemoral osteoarthritis in men and women and in whites and African Americans. Arthritis Care Res. 2012;64(6): 847-852.
3. Jackson JP, Waugh W. Tibial osteotomy for osteoarthritis of the knee. J Bone Joint Surg Br. 1961;43:746-751.
4. Brouwer RW, Bierma-Zeinstra SM, van Raaij TM, Verhaar JA. Osteotomy for medial compartment arthritis of the knee using a closing wedge or an opening wedge controlled by a Puddu plate. A one-year randomised, controlled study. J Bone Joint Surg Br. 2006;88(11):1454-1459.
5. Duivenvoorden T, Brouwer RW, Baan A, et al. Comparison of closing-wedge and opening-wedge high tibial osteotomy for medial compartment osteoarthritis of the knee: a randomized controlled trial with a six-year follow-up. J Bone Joint Surg Am. 2014;96(17):1425-1432.
6. Hutchison CR, Cho B, Wong N, Agnidis Z, Gross AE. Proximal valgus tibial osteotomy for osteoarthritis of the knee. Instr Course Lect. 1999;48:131-134.
7. Trieb K, Grohs J, Hanslik-Schnabel B, Stulnig T, Panotopoulos J, Wanivenhaus A. Age predicts outcome of high-tibial osteotomy. Knee Surg Sports Traumatol Arthrosc. 2006;14(2):149-152.
8. Hui C, Salmon LJ, Kok A, et al. Long-term survival of high tibial osteotomy for medial compartment osteoarthritis of the knee. Am J Sports Med. 2011;39(1):64-70.
9. Howells NR, Salmon L, Waller A, Scanelli J, Pinczewski LA. The outcome at ten years of lateral closing-wedge high tibial osteotomy: determinants of survival and functional outcome. Bone Joint J Br. 2014;96(11):1491-1497.
10. Niinimäki TT, Eskelinen A, Mann BS, Junnila M, Ohtonen P, Leppilahti J. Survivorship of high tibial osteotomy in the treatment of osteoarthritis of the knee: Finnish registry-based study of 3195 knees. J Bone Joint Surg Br. 2012;94(11):1517-1521.
11. Flecher X, Parratte S, Aubaniac JM, Argenson JN. A 12-28-year followup study of closing wedge high tibial osteotomy. Clin Orthop Relat Res. 2006;(452):91-96.
12. Akizuki S, Shibakawa A, Takizawa T, Yamazaki I, Horiuchi H. The long-term outcome of high tibial osteotomy: a ten- to 20-year follow-up. J Bone Joint Surg Br. 2008;90(5):592-596.
13. van Raaij T, Reijman M, Brouwer RW, Jakma TS, Verhaar JN. Survival of closing-wedge high tibial osteotomy: good outcome in men with low-grade osteoarthritis after 10-16 years. Acta Orthop. 2008;79:230-234.
14. Ahlbäck S. Osteoarthrosis of the knee. A radiographic investigation. Acta Radiol Diagn. 1968;(suppl 277):7-72.
15. Bannuru RR, Natov NS, Obadan IE, Price LL, Schmid CH, McAlindon TE. Therapeutic trajectory of hyaluronic acid versus corticosteroids in the treatment of knee osteoarthritis: a systematic review and meta-analysis. Arthritis Rheum. 2009;61(12):1704-1711.
16. Evanich JD, Evanich CJ, Wright MB, Rydlewicz JA. Efficacy of intraarticular hyaluronic acid injections in knee osteoarthritis. Clin Orthop Relat Res. 2001;(390):173-181.
17. Naudie D, Bourne RB, Rorabeck CH, Bourne TJ. The Install Award. Survivorship of the high tibial valgus osteotomy. A 10- to -22-year followup study. Clin Orthop Relat Res. 1999;(367):18-27.
18. Sprenger TR, Doerzbacher JF. Tibial osteotomy for the treatment of varus gonarthrosis. Survival and failure analysis to twenty-two years. J Bone Joint Surg Br. 2003;85(3):469-474.
19. Billings A, Scott DF, Camargo MP, Hofmann AA. High tibial osteotomy with a calibrated osteotomy guide, rigid internal fixation, and early motion. Long-term follow-up. J Bone Joint Surg Am. 2000;82(1):70-79.
20. Koshino T, Yoshida T, Ara Y, Saito I, Saito T. Fifteen to twenty-eight years’ follow-up results of high tibial valgus osteotomy for osteoarthritic knee. Knee. 2004;11(6):439-444.
21. Schallberger A, Jacobi M, Wahl P, Maestretti G, Jakob RP. High tibial valgus osteotomy in unicompartmental medial osteoarthritis of the knee: a retrospective follow-up study over 13-21 years. Knee Surg Sports Traumatol Arthrosc. 2011;19(1):122-127.
22. Insall J, Aglietti P. A five to seven-year follow-up of unicondylar arthroplasty. J Bone Joint Surg Am. 1980;62(8):1329-1337.
23. Kozinn SC, Scott R. Unicondylar knee arthroplasty. J Bone Joint Surg Am. 1989;71(1):145-150.
24. Thompson SA, Liabaud B, Nellans KW, Geller JA. Factors associated with poor outcomes following unicompartmental knee arthroplasty: redefining the “classic” indications for surgery. J Arthroplasty. 2013;28(9):1561-1564.
25. Heyse TJ, Khefacha A, Peersman G, Cartier P. Survivorship of UKA in the middle-aged. Knee. 2012;19(5):585-591.
26. Schai PA, Suh JT, Thornhill TS, Scott RD. Unicompartmental knee arthroplasty in middle-aged patients: a 2- to 6-year follow-up evaluation. J Arthroplasty. 1998;13(4):365-372.
27. Price AJ, Dodd CA, Svard UG, Murray DW. Oxford medial unicompartmental knee arthroplasty in patients younger and older than 60 years of age. J Bone Joint Surg Br. 2005;87(11):1488-1492.
28. Pennington DW, Swienckowski JJ, Lutes WB, Drake GN. Unicompartmental knee arthroplasty in patients sixty years of age or younger. J Bone Joint Surg Am. 2003;85(10):1968-1973.
29. Cavaignac E, Lafontan V, Reina N, et al. Obesity has no adverse effect on the outcome of unicompartmental knee replacement at a minimum follow-up of seven years. Bone Joint J Br. 2013;95(8):1064-1068.
30. Tabor OB Jr, Tabor OB, Bernard M, Wan JY. Unicompartmental knee arthroplasty: long-term success in middle-age and obese patients. J Surg Orthop Adv. 2005;14(2):59-63.
31. Berend KR, Lombardi AV Jr, Adams JB. Obesity, young age, patellofemoral disease, and anterior knee pain: identifying the unicondylar arthroplasty patient in the United States. Orthopedics. 2007;30(5 suppl):19-23.
32. Xing Z, Katz J, Jiranek W. Unicompartmental knee arthroplasty: factors influencing the outcome. J Knee Surg. 2012;25(5):369-373.
33. Plate JF, Augart MA, Seyler TM, et al. Obesity has no effect on outcomes following unicompartmental knee arthroplasty [published online April 12, 2015]. Knee Surg Sports Traumatol Arthrosc. doi:10.1007/s00167-015-3597-5.
34. Niinimäki TT, Murray DW, Partanen J, Pajala A, Leppilahti JI. Unicompartmental knee arthroplasties implanted for osteoarthritis with partial loss of joint space have high re-operation rates. Knee. 2011;18(6):432-435.
35. Carlsson LV, Albrektsson BE, Regnér LR. Minimally invasive surgery vs conventional exposure using the Miller-Galante unicompartmental knee arthroplasty: a randomized radiostereometric study. J Arthroplasty. 2006;21(2):151-156.
36. Repicci JA. Mini-invasive knee unicompartmental arthroplasty: bone-sparing technique. Surg Technol Int. 2003;11:282-286.
37. Pandit H, Jenkins C, Barker K, Dodd CA, Murray DW. The Oxford medial unicompartmental knee replacement using a minimally-invasive approach. J Bone Joint Surg Br. 2006;88(1):54-60.
38. Romanowski MR, Repicci JA. Minimally invasive unicondylar arthroplasty: eight-year follow-up. J Knee Surg. 2002;15(1):17-22.
39. Koskinen E, Paavolainen P, Eskelinen A, Pulkkinen P, Remes V. Unicondylar knee replacement for primary osteoarthritis: a prospective follow-up study of 1,819 patients from the Finnish Arthroplasty Register. Acta Orthop. 2007;78(1):128-135.
40. Price AJ, Waite JC, Svard U. Long-term clinical results of the medial Oxford unicompartmental knee arthroplasty. Clin Orthop Relat Res. 2005;(435):171-180.
41. Berger RA, Meneghini RM, Jacobs JJ, et al. Results of unicompartmental knee arthroplasty at a minimum of ten years of follow-up. J Bone Joint Surg Am. 2005;87(5):999-1006.
42. Peersman G, Stuyts B, Vandenlangenbergh T, Cartier P, Fennema P. Fixed- versus mobile-bearing UKA: a systematic review and meta-analysis. Knee Surg Sports Traumatol Arthrosc. 2015;23(11):3296-3305.
43. Zambianchi F, Digennaro V, Giorgini A, et al. Surgeon’s experience influences UKA survivorship: a comparative study between all-poly and metal back designs. Knee Surg Sports Traumatol Arthrosc. 2015;23(7):2074-2080.
44. Robertsson O, Knutson K, Lewold S, Lidgren L. The routine of surgical management reduces failure after unicompartmental knee arthroplasty. J Bone Joint Surg Br. 2001;83(1):45-49.
45. Furnes O, Espehaug B, Lie SA, Vollset SE, Engesaeter LB, Havelin LI. Failure mechanisms after unicompartmental and tricompartmental primary knee replacement with cement. J Bone Joint Surg Am. 2007;89(3):519-525.
46. Robertsson O, Lidgren L. The short-term results of 3 common UKA implants during different periods in Sweden. J Arthroplasty. 2008;23(6):801-807.
47. Stukenborg-Colsman C, Wirth CJ, Lazovic D, Wefer A. High tibial osteotomy versus unicompartmental joint replacement in unicompartmental knee joint osteoarthritis: 7-10-year follow-up prospective randomised study. Knee. 2001;8(3):187-194.
48. Coventry MB. Osteotomy about the knee for degenerative and rheumatoid arthritis. J Bone Joint Surg Am. 1973;55(1):23-48.
49. Börjesson M, Weidenhielm L, Mattsson E, Olsson E. Gait and clinical measurements in patients with knee osteoarthritis after surgery: a prospective 5-year follow-up study. Knee. 2005;12(2):121-127.
50. Fu D, Li G, Chen K, Zhao Y, Hua Y, Cai Z. Comparison of high tibial osteotomy and unicompartmental knee arthroplasty in the treatment of unicompartmental osteoarthritis: a meta-analysis. J Arthroplasty. 2013;28(5):759-765.
51. W-Dahl A, Robertsson O, Lidgren L. Surgery for knee osteoarthritis in younger patients. Acta Orthop. 2010;81(2):161-164.
52. Niinimäki TT, Eskelinen A, Ohtonen P, Junnila M, Leppilahti J. Incidence of osteotomies around the knee for the treatment of knee osteoarthritis: a 22-year population-based study. Int Orthop. 2012;36(7):1399-1402.
53. Wright J, Heck D, Hawker G, et al. Rates of tibial osteotomies in Canada and the United States. Clin Orthop Relat Res. 1995;(319):266-275.
54. Nwachukwu BU, McCormick FM, Schairer WW, Frank RM, Provencher MT, Roche MW. Unicompartmental knee arthroplasty versus high tibial osteotomy: United States practice patterns for the surgical treatment of unicompartmental arthritis. J Arthroplasty. 2014;29(8):1586-1589.
55. Bolognesi MP, Greiner MA, Attarian DE, et al. Unicompartmental knee arthroplasty and total knee arthroplasty among Medicare beneficiaries, 2000 to 2009. J Bone Joint Surg Am. 2013;95(22):e174.
56. Pearle AD, O’Loughlin PF, Kendoff DO. Robot-assisted unicompartmental knee arthroplasty. J Arthroplasty. 2010;25(2):230-237.
57. Brown NM, Sheth NP, Davis K, et al. Total knee arthroplasty has higher postoperative morbidity than unicompartmental knee arthroplasty: a multicenter analysis. J Arthroplasty. 2012;27(8 suppl):86-90.
Medicare doesn’t lower TKI costs enough, study suggests

cut in half with a pill splitter
Photo by Patrick Pelletier
Significant out-of-pocket costs may delay treatment for Medicare beneficiaries with chronic myeloid leukemia (CML), according to a study published in the Journal of Clinical Oncology.
Researchers studied 393 patients with CML who had federally funded health insurance—specifically, a Medicare Part D plan.
Nearly a third of these patients did not start tyrosine kinase inhibitor (TKI) treatment within 6 months of their diagnosis.
However, patients who had access to subsidies that help cover treatment costs had a shorter median time to the start of therapy.
“There are 2 troubling findings here,” said study author Aaron Winn, a doctoral student at the University of North Carolina at Chapel Hill.
“First, we are seeing that more than 30% of people aren’t starting therapy within 6 months. Second, we are seeing long delays in starting drugs for people without subsidies. This is very concerning as these delays may be an indicator that the patient is trying to find funds to pay for their first treatment.”
Medicare Part D and TKIs
Previous studies have shown that patients insured through Medicare Part D have out of-pocket costs of nearly $3000 for the first month’s supply of a TKI.
According to researchers, the high upfront costs are due to the Medicare Part D benefit design, which requires patients to pay a larger share of medication costs until they have paid at least $4850 out-of-pocket in a year (cost in 2016). After that, patients pay 5% of the monthly drug costs.
In order to qualify for Medicare Part D’s low-income subsidy, an individual must have an annual income of less than $17,820 and assets of less than $13,640 (figures for 2016).
“Once you’re on Medicare Part D, there really aren’t ways to minimize these out-of-pocket costs, other than subsidies,” said Stacie Dusetzina, PhD, of the University of North Carolina at Chapel Hill.
“One of the challenges is that when the Medicare benefit was designed, I don’t think they were really considering these very expensive therapies. The benefit design makes a lot more sense when you’re looking at drugs that cost several hundred dollars versus several thousand dollars or more. We really need to think carefully about how much these high out-of-pocket costs are impacting patients’ access to life-saving drugs.”
Study results
For this study, Dr Dusetzina and her colleagues evaluated data on 393 patients who were diagnosed with CML between 2007 and 2011. The patients’ median age was 77, 47% were married, 48% were male, and 85% were white.
All of the patients were enrolled in Medicare Part D, and 40% qualified for subsidies to lower drug costs.
Of all the patients, there were 32% who had not started treatment with a first-line TKI (imatinib, nilotinib, or dasatinib) within 6 months of diagnosis.
However, having access to subsidies was associated with a shorter time to the start of treatment. The median time to the start of treatment was 58 days for patients with subsidies and 108 days for patients without them.
While the gap between the 2 groups widened after diagnosis, eventually, patients without subsidies did catch up, Dr Dusetzina said.
Ninety days from diagnosis, 48% of patients without subsidies had started treatment, compared to 63% of patients with subsidies. At 6 months from diagnosis, 64% of patients without subsidies had started treatment, compared to 65% of patients with subsidies.
Dr Dusetzina said patients without subsidies could be catching up as they find the financial resources to help cover those initial costs. But overall, patients with subsidies were 35% more likely to start TKI treatment faster.
“We recognize that people have a high cost to even start therapy, and this study really demonstrates the difference between people with and without a subsidy in initiating therapy,” Dr Dusetzina said. “The out-of-pocket costs may be delaying people starting these life-saving drugs.” ![]()

cut in half with a pill splitter
Photo by Patrick Pelletier
Significant out-of-pocket costs may delay treatment for Medicare beneficiaries with chronic myeloid leukemia (CML), according to a study published in the Journal of Clinical Oncology.
Researchers studied 393 patients with CML who had federally funded health insurance—specifically, a Medicare Part D plan.
Nearly a third of these patients did not start tyrosine kinase inhibitor (TKI) treatment within 6 months of their diagnosis.
However, patients who had access to subsidies that help cover treatment costs had a shorter median time to the start of therapy.
“There are 2 troubling findings here,” said study author Aaron Winn, a doctoral student at the University of North Carolina at Chapel Hill.
“First, we are seeing that more than 30% of people aren’t starting therapy within 6 months. Second, we are seeing long delays in starting drugs for people without subsidies. This is very concerning as these delays may be an indicator that the patient is trying to find funds to pay for their first treatment.”
Medicare Part D and TKIs
Previous studies have shown that patients insured through Medicare Part D have out of-pocket costs of nearly $3000 for the first month’s supply of a TKI.
According to researchers, the high upfront costs are due to the Medicare Part D benefit design, which requires patients to pay a larger share of medication costs until they have paid at least $4850 out-of-pocket in a year (cost in 2016). After that, patients pay 5% of the monthly drug costs.
In order to qualify for Medicare Part D’s low-income subsidy, an individual must have an annual income of less than $17,820 and assets of less than $13,640 (figures for 2016).
“Once you’re on Medicare Part D, there really aren’t ways to minimize these out-of-pocket costs, other than subsidies,” said Stacie Dusetzina, PhD, of the University of North Carolina at Chapel Hill.
“One of the challenges is that when the Medicare benefit was designed, I don’t think they were really considering these very expensive therapies. The benefit design makes a lot more sense when you’re looking at drugs that cost several hundred dollars versus several thousand dollars or more. We really need to think carefully about how much these high out-of-pocket costs are impacting patients’ access to life-saving drugs.”
Study results
For this study, Dr Dusetzina and her colleagues evaluated data on 393 patients who were diagnosed with CML between 2007 and 2011. The patients’ median age was 77, 47% were married, 48% were male, and 85% were white.
All of the patients were enrolled in Medicare Part D, and 40% qualified for subsidies to lower drug costs.
Of all the patients, there were 32% who had not started treatment with a first-line TKI (imatinib, nilotinib, or dasatinib) within 6 months of diagnosis.
However, having access to subsidies was associated with a shorter time to the start of treatment. The median time to the start of treatment was 58 days for patients with subsidies and 108 days for patients without them.
While the gap between the 2 groups widened after diagnosis, eventually, patients without subsidies did catch up, Dr Dusetzina said.
Ninety days from diagnosis, 48% of patients without subsidies had started treatment, compared to 63% of patients with subsidies. At 6 months from diagnosis, 64% of patients without subsidies had started treatment, compared to 65% of patients with subsidies.
Dr Dusetzina said patients without subsidies could be catching up as they find the financial resources to help cover those initial costs. But overall, patients with subsidies were 35% more likely to start TKI treatment faster.
“We recognize that people have a high cost to even start therapy, and this study really demonstrates the difference between people with and without a subsidy in initiating therapy,” Dr Dusetzina said. “The out-of-pocket costs may be delaying people starting these life-saving drugs.” ![]()

cut in half with a pill splitter
Photo by Patrick Pelletier
Significant out-of-pocket costs may delay treatment for Medicare beneficiaries with chronic myeloid leukemia (CML), according to a study published in the Journal of Clinical Oncology.
Researchers studied 393 patients with CML who had federally funded health insurance—specifically, a Medicare Part D plan.
Nearly a third of these patients did not start tyrosine kinase inhibitor (TKI) treatment within 6 months of their diagnosis.
However, patients who had access to subsidies that help cover treatment costs had a shorter median time to the start of therapy.
“There are 2 troubling findings here,” said study author Aaron Winn, a doctoral student at the University of North Carolina at Chapel Hill.
“First, we are seeing that more than 30% of people aren’t starting therapy within 6 months. Second, we are seeing long delays in starting drugs for people without subsidies. This is very concerning as these delays may be an indicator that the patient is trying to find funds to pay for their first treatment.”
Medicare Part D and TKIs
Previous studies have shown that patients insured through Medicare Part D have out of-pocket costs of nearly $3000 for the first month’s supply of a TKI.
According to researchers, the high upfront costs are due to the Medicare Part D benefit design, which requires patients to pay a larger share of medication costs until they have paid at least $4850 out-of-pocket in a year (cost in 2016). After that, patients pay 5% of the monthly drug costs.
In order to qualify for Medicare Part D’s low-income subsidy, an individual must have an annual income of less than $17,820 and assets of less than $13,640 (figures for 2016).
“Once you’re on Medicare Part D, there really aren’t ways to minimize these out-of-pocket costs, other than subsidies,” said Stacie Dusetzina, PhD, of the University of North Carolina at Chapel Hill.
“One of the challenges is that when the Medicare benefit was designed, I don’t think they were really considering these very expensive therapies. The benefit design makes a lot more sense when you’re looking at drugs that cost several hundred dollars versus several thousand dollars or more. We really need to think carefully about how much these high out-of-pocket costs are impacting patients’ access to life-saving drugs.”
Study results
For this study, Dr Dusetzina and her colleagues evaluated data on 393 patients who were diagnosed with CML between 2007 and 2011. The patients’ median age was 77, 47% were married, 48% were male, and 85% were white.
All of the patients were enrolled in Medicare Part D, and 40% qualified for subsidies to lower drug costs.
Of all the patients, there were 32% who had not started treatment with a first-line TKI (imatinib, nilotinib, or dasatinib) within 6 months of diagnosis.
However, having access to subsidies was associated with a shorter time to the start of treatment. The median time to the start of treatment was 58 days for patients with subsidies and 108 days for patients without them.
While the gap between the 2 groups widened after diagnosis, eventually, patients without subsidies did catch up, Dr Dusetzina said.
Ninety days from diagnosis, 48% of patients without subsidies had started treatment, compared to 63% of patients with subsidies. At 6 months from diagnosis, 64% of patients without subsidies had started treatment, compared to 65% of patients with subsidies.
Dr Dusetzina said patients without subsidies could be catching up as they find the financial resources to help cover those initial costs. But overall, patients with subsidies were 35% more likely to start TKI treatment faster.
“We recognize that people have a high cost to even start therapy, and this study really demonstrates the difference between people with and without a subsidy in initiating therapy,” Dr Dusetzina said. “The out-of-pocket costs may be delaying people starting these life-saving drugs.” ![]()
NIH: Some Complementary Treatments Safe and Effective for Chronic Pain
Do you have a patient with back pain? Suggest acupuncture and yoga. Osteoarthritis? Acupuncture and tai chi. Those are some of the approaches that are safe and effective in treating chronic pain, according to researchers from the National Center for Complementary and Integrative Health (NCCIH). They also gave a nod to massage for neck pain and relaxation techniques for severe headaches and migraine.
The researchers reviewed 105 U.S. randomized controlled trials from the past 50 years, focusing on 7 complementary treatments: acupuncture, spinal manipulation or osteopathic manipulation, massage therapy, tai chi, yoga, and relaxation techniques (such as mindfulness meditation). They also evaluated findings from studies involving natural product supplements: chondroitin, glucosamine, methylsulfonylmethane (MSM), S-adenosylmethionine (SAMe), and omega-3 fatty acids.
The researchers rated those treatments in 5 painful conditions often seen in primary care: back pain, osteoarthritis, neck pain, fibromyalgia, and severe headaches and migraine.
Although evidence was weaker, they also found that massage, spinal manipulation, and osteopathic manipulation may help in some back pain, and relaxation and tai chi may help patients with fibromyalgia.
No trial reported a serious adverse event linked to a complementary treatment. Most adverse events were gastrointestinal problems from glucosamine, chondroitin, MSM, or SAMe, or minor soreness from exercise.
Do you have a patient with back pain? Suggest acupuncture and yoga. Osteoarthritis? Acupuncture and tai chi. Those are some of the approaches that are safe and effective in treating chronic pain, according to researchers from the National Center for Complementary and Integrative Health (NCCIH). They also gave a nod to massage for neck pain and relaxation techniques for severe headaches and migraine.
The researchers reviewed 105 U.S. randomized controlled trials from the past 50 years, focusing on 7 complementary treatments: acupuncture, spinal manipulation or osteopathic manipulation, massage therapy, tai chi, yoga, and relaxation techniques (such as mindfulness meditation). They also evaluated findings from studies involving natural product supplements: chondroitin, glucosamine, methylsulfonylmethane (MSM), S-adenosylmethionine (SAMe), and omega-3 fatty acids.
The researchers rated those treatments in 5 painful conditions often seen in primary care: back pain, osteoarthritis, neck pain, fibromyalgia, and severe headaches and migraine.
Although evidence was weaker, they also found that massage, spinal manipulation, and osteopathic manipulation may help in some back pain, and relaxation and tai chi may help patients with fibromyalgia.
No trial reported a serious adverse event linked to a complementary treatment. Most adverse events were gastrointestinal problems from glucosamine, chondroitin, MSM, or SAMe, or minor soreness from exercise.
Do you have a patient with back pain? Suggest acupuncture and yoga. Osteoarthritis? Acupuncture and tai chi. Those are some of the approaches that are safe and effective in treating chronic pain, according to researchers from the National Center for Complementary and Integrative Health (NCCIH). They also gave a nod to massage for neck pain and relaxation techniques for severe headaches and migraine.
The researchers reviewed 105 U.S. randomized controlled trials from the past 50 years, focusing on 7 complementary treatments: acupuncture, spinal manipulation or osteopathic manipulation, massage therapy, tai chi, yoga, and relaxation techniques (such as mindfulness meditation). They also evaluated findings from studies involving natural product supplements: chondroitin, glucosamine, methylsulfonylmethane (MSM), S-adenosylmethionine (SAMe), and omega-3 fatty acids.
The researchers rated those treatments in 5 painful conditions often seen in primary care: back pain, osteoarthritis, neck pain, fibromyalgia, and severe headaches and migraine.
Although evidence was weaker, they also found that massage, spinal manipulation, and osteopathic manipulation may help in some back pain, and relaxation and tai chi may help patients with fibromyalgia.
No trial reported a serious adverse event linked to a complementary treatment. Most adverse events were gastrointestinal problems from glucosamine, chondroitin, MSM, or SAMe, or minor soreness from exercise.
How Should Hospitalists Manage Elderly Patients with Dysphagia?
The Case
A 74-year-old man with Alzheimer’s dementia presents with urinary tract infection (UTI), hypovolemia, and hypernatremia. He also has chronic dysphagia with a history of aspiration pneumonia and has been on thickened liquids at home for the past five months. As his infection is treated, he improves and requests water to drink.
Background
The diagnosis of dysphagia is clinical, and assessments from patients and family are often sufficient. The optimal test to assess the severity of dysphagia is a bedside swallow evaluation using small amounts of water.1 Video-assisted fluoroscopic examinations can identify problem areas within the oropharynx and esophagus and may help determine the etiology of dysphagia.
What evidence supports various treatment options for dysphagia?
Access to Water
Water is a thin liquid with low viscosity, which allows for rapid transit through the oropharynx. In debilitated and elderly patients, thin liquids easily reach the epiglottis and enter the trachea before pharyngeal muscles compensate. As such, access to water and other thin liquids is often restricted in patients suspected to have dysphagia.4
However, allowing access to water improves patient satisfaction, reduces the development of dehydration, and does not increase the incidence of AP. Bedside therapy interventions such as correct positioning and chin-tuck and sipping technique as well as attention to oral hygiene are recommended prior to more noxious options such as thickened liquids.1 The Frazier water protocol may help provide logistical guidance for facilities interested in improving access to water for patients with dysphagia.
Liquid Modification
Many clinicians manage dysphagia through restricting access to all thin liquids. In the hospital setting where video fluoroscopy and speech therapy are readily available, clinicians frequently employ the use of modified diets with thickened liquids in order to minimize the risk of aspiration despite the lack of high-quality evidence supporting liquid modification.2 Patients associate thickened liquids and restricted diets with a reduction in quality of life. Compliance studies have shown that only a minority of patients are compliant with thickened liquids at five days. In addition, thickening liquids has not been shown to decrease the risk of AP nor improve nutritional status, and it may actually cause harm by increasing the risk of dehydration and UTI.4
Tube Feeding
In patients with severe dysphagia in whom conservative management is not feasible or has failed, maintaining adequate nutrition can be a challenge. There are encouraging data with nutritionally enriching and modifying the texture of solid foods.1 Alternative methods of enteral nutrition delivery are often also considered. The most common vehicles of delivery are nasogastric tubes, post-pyloric feeding tubes, and percutaneous endoscopic gastrostomy (PEG) tubes. In theory, bypassing the pharynx and esophagus could result in fewer aspiration events and less AP.3 However, nasogastric, post-pyloric, or PEG feeding does not decrease the risk of AP. For patients with advanced dementia, there have been no randomized trials demonstrating an improvement in mortality with tube feeds.4 Tube feeding also carries with it a slight procedural risk and a high incidence of associated diarrhea, plus is associated with electrolyte derangements such as hypernatremia. The decision to pursue tube feeding should be weighed heavily in every patient and is highly influenced by the etiology and anticipated duration of dysphagia.
Selective Digestive Decontamination
Selective digestive decontamination (SDD) is a protocol-based treatment that aims to eradicate potentially pathogenic gut flora, particularly aerobic gram-negatives, in critically ill patients to reduce the impact of aspiration events. The utilization of SDD and the available literature center firmly on critically ill and ventilated patients. Subsequent studies have demonstrated recolonization after protocol cessation, and long-term effects are currently undefined.5 Until it can be studied in broader populations and proven to have clinical benefit, employing SSD in non-critically ill patients with dysphagia remains unsupported.
Multimodal Approach
Many rehabilitation centers incorporate a therapist-driven swallowing treatment program. Evidence suggests patient and family counseling alone may not be effective, so these programs variably incorporate diet/liquid modification, strengthening exercises, sensory processing techniques, and even neuromuscular electrical stimulation for muscle building.1 Accordingly, these programs are resource-intensive.
Management
Dysphagia remains a significant clinical problem for hospitalized patients. The existing literature and practice guidelines generally support a “less is more” approach. Though liquid/diet modification is common practice, it is not based in solid evidence and may contribute to unnecessary tube feeding. The best current evidence supports allowing access to water and ice chips. The ideal management plan for each patient will differ and should incorporate patient and family preferences in a multidisciplinary approach.
Back to the Case
Our patient requests water. He coughs after drinking during a bedside swallow evaluation. The risks of potential aspiration and AP are explained, and he expresses his understanding. He reiterates his choice to be allowed access to water as it is important to his quality of life. The speech therapy team is consulted and provides instruction on chin-tuck positioning, oral care, and timing water between meals rather than while eating food. He does well for the remainder of the hospital stay, and by time of discharge, his electrolytes are corrected, and he is much more comfortable being allowed to drink water. He is discharged home and encouraged to continue with these conservative measures.
Bottom Line
Evidence to support many common interventions for dysphagia is lacking; patients with dysphagia are best managed via a multidisciplinary, multimodal approach that provides access to water whenever possible. TH
Vijay G. Paryani, MD, is an internal medicine resident in the department of internal medicine at the University of Kentucky. Joseph R. Sweigart, MD, is a hospitalist and assistant professor of hospital medicine in the division of hospital medicine at the University of Kentucky. Laura C. Fanucchi, MD, is a hospitalist and assistant professor of hospital medicine in the division of hospital medicine at the University of Kentucky.
References
- Karagiannis MJ, Chivers L, Karagiannis TC. Effects of oral intake of water in patients with oropharyngeal dysphagia. BMC Geriatr. 2011;11(2):9.
- Foley N, Teasell R, Salter K, Kruger E, Martino R. Dysphagia treatment post stroke: a systematic review of randomized controlled trials. Age Ageing. 2008;37(3):258-264.
- Marik PE. Aspiration pneumonitis and aspiration pneumonia. N Engl J Med. 2001;344(9):665-671.
- Loeb MB, Becker M, Eady A, Walker-Dilks C. Interventions to prevent aspiration pneumonia in older adults: a systematic review. J Am Geriatr Soc. 2003;51(7):1018-1022.
- Gosney M, Martin MV, Wright AE. The role of selective decontamination of the digestive tract in acute stroke. Age Ageing 2006;35(1):42-47.
The Case
A 74-year-old man with Alzheimer’s dementia presents with urinary tract infection (UTI), hypovolemia, and hypernatremia. He also has chronic dysphagia with a history of aspiration pneumonia and has been on thickened liquids at home for the past five months. As his infection is treated, he improves and requests water to drink.
Background
The diagnosis of dysphagia is clinical, and assessments from patients and family are often sufficient. The optimal test to assess the severity of dysphagia is a bedside swallow evaluation using small amounts of water.1 Video-assisted fluoroscopic examinations can identify problem areas within the oropharynx and esophagus and may help determine the etiology of dysphagia.
What evidence supports various treatment options for dysphagia?
Access to Water
Water is a thin liquid with low viscosity, which allows for rapid transit through the oropharynx. In debilitated and elderly patients, thin liquids easily reach the epiglottis and enter the trachea before pharyngeal muscles compensate. As such, access to water and other thin liquids is often restricted in patients suspected to have dysphagia.4
However, allowing access to water improves patient satisfaction, reduces the development of dehydration, and does not increase the incidence of AP. Bedside therapy interventions such as correct positioning and chin-tuck and sipping technique as well as attention to oral hygiene are recommended prior to more noxious options such as thickened liquids.1 The Frazier water protocol may help provide logistical guidance for facilities interested in improving access to water for patients with dysphagia.
Liquid Modification
Many clinicians manage dysphagia through restricting access to all thin liquids. In the hospital setting where video fluoroscopy and speech therapy are readily available, clinicians frequently employ the use of modified diets with thickened liquids in order to minimize the risk of aspiration despite the lack of high-quality evidence supporting liquid modification.2 Patients associate thickened liquids and restricted diets with a reduction in quality of life. Compliance studies have shown that only a minority of patients are compliant with thickened liquids at five days. In addition, thickening liquids has not been shown to decrease the risk of AP nor improve nutritional status, and it may actually cause harm by increasing the risk of dehydration and UTI.4
Tube Feeding
In patients with severe dysphagia in whom conservative management is not feasible or has failed, maintaining adequate nutrition can be a challenge. There are encouraging data with nutritionally enriching and modifying the texture of solid foods.1 Alternative methods of enteral nutrition delivery are often also considered. The most common vehicles of delivery are nasogastric tubes, post-pyloric feeding tubes, and percutaneous endoscopic gastrostomy (PEG) tubes. In theory, bypassing the pharynx and esophagus could result in fewer aspiration events and less AP.3 However, nasogastric, post-pyloric, or PEG feeding does not decrease the risk of AP. For patients with advanced dementia, there have been no randomized trials demonstrating an improvement in mortality with tube feeds.4 Tube feeding also carries with it a slight procedural risk and a high incidence of associated diarrhea, plus is associated with electrolyte derangements such as hypernatremia. The decision to pursue tube feeding should be weighed heavily in every patient and is highly influenced by the etiology and anticipated duration of dysphagia.
Selective Digestive Decontamination
Selective digestive decontamination (SDD) is a protocol-based treatment that aims to eradicate potentially pathogenic gut flora, particularly aerobic gram-negatives, in critically ill patients to reduce the impact of aspiration events. The utilization of SDD and the available literature center firmly on critically ill and ventilated patients. Subsequent studies have demonstrated recolonization after protocol cessation, and long-term effects are currently undefined.5 Until it can be studied in broader populations and proven to have clinical benefit, employing SSD in non-critically ill patients with dysphagia remains unsupported.
Multimodal Approach
Many rehabilitation centers incorporate a therapist-driven swallowing treatment program. Evidence suggests patient and family counseling alone may not be effective, so these programs variably incorporate diet/liquid modification, strengthening exercises, sensory processing techniques, and even neuromuscular electrical stimulation for muscle building.1 Accordingly, these programs are resource-intensive.
Management
Dysphagia remains a significant clinical problem for hospitalized patients. The existing literature and practice guidelines generally support a “less is more” approach. Though liquid/diet modification is common practice, it is not based in solid evidence and may contribute to unnecessary tube feeding. The best current evidence supports allowing access to water and ice chips. The ideal management plan for each patient will differ and should incorporate patient and family preferences in a multidisciplinary approach.
Back to the Case
Our patient requests water. He coughs after drinking during a bedside swallow evaluation. The risks of potential aspiration and AP are explained, and he expresses his understanding. He reiterates his choice to be allowed access to water as it is important to his quality of life. The speech therapy team is consulted and provides instruction on chin-tuck positioning, oral care, and timing water between meals rather than while eating food. He does well for the remainder of the hospital stay, and by time of discharge, his electrolytes are corrected, and he is much more comfortable being allowed to drink water. He is discharged home and encouraged to continue with these conservative measures.
Bottom Line
Evidence to support many common interventions for dysphagia is lacking; patients with dysphagia are best managed via a multidisciplinary, multimodal approach that provides access to water whenever possible. TH
Vijay G. Paryani, MD, is an internal medicine resident in the department of internal medicine at the University of Kentucky. Joseph R. Sweigart, MD, is a hospitalist and assistant professor of hospital medicine in the division of hospital medicine at the University of Kentucky. Laura C. Fanucchi, MD, is a hospitalist and assistant professor of hospital medicine in the division of hospital medicine at the University of Kentucky.
References
- Karagiannis MJ, Chivers L, Karagiannis TC. Effects of oral intake of water in patients with oropharyngeal dysphagia. BMC Geriatr. 2011;11(2):9.
- Foley N, Teasell R, Salter K, Kruger E, Martino R. Dysphagia treatment post stroke: a systematic review of randomized controlled trials. Age Ageing. 2008;37(3):258-264.
- Marik PE. Aspiration pneumonitis and aspiration pneumonia. N Engl J Med. 2001;344(9):665-671.
- Loeb MB, Becker M, Eady A, Walker-Dilks C. Interventions to prevent aspiration pneumonia in older adults: a systematic review. J Am Geriatr Soc. 2003;51(7):1018-1022.
- Gosney M, Martin MV, Wright AE. The role of selective decontamination of the digestive tract in acute stroke. Age Ageing 2006;35(1):42-47.
The Case
A 74-year-old man with Alzheimer’s dementia presents with urinary tract infection (UTI), hypovolemia, and hypernatremia. He also has chronic dysphagia with a history of aspiration pneumonia and has been on thickened liquids at home for the past five months. As his infection is treated, he improves and requests water to drink.
Background
The diagnosis of dysphagia is clinical, and assessments from patients and family are often sufficient. The optimal test to assess the severity of dysphagia is a bedside swallow evaluation using small amounts of water.1 Video-assisted fluoroscopic examinations can identify problem areas within the oropharynx and esophagus and may help determine the etiology of dysphagia.
What evidence supports various treatment options for dysphagia?
Access to Water
Water is a thin liquid with low viscosity, which allows for rapid transit through the oropharynx. In debilitated and elderly patients, thin liquids easily reach the epiglottis and enter the trachea before pharyngeal muscles compensate. As such, access to water and other thin liquids is often restricted in patients suspected to have dysphagia.4
However, allowing access to water improves patient satisfaction, reduces the development of dehydration, and does not increase the incidence of AP. Bedside therapy interventions such as correct positioning and chin-tuck and sipping technique as well as attention to oral hygiene are recommended prior to more noxious options such as thickened liquids.1 The Frazier water protocol may help provide logistical guidance for facilities interested in improving access to water for patients with dysphagia.
Liquid Modification
Many clinicians manage dysphagia through restricting access to all thin liquids. In the hospital setting where video fluoroscopy and speech therapy are readily available, clinicians frequently employ the use of modified diets with thickened liquids in order to minimize the risk of aspiration despite the lack of high-quality evidence supporting liquid modification.2 Patients associate thickened liquids and restricted diets with a reduction in quality of life. Compliance studies have shown that only a minority of patients are compliant with thickened liquids at five days. In addition, thickening liquids has not been shown to decrease the risk of AP nor improve nutritional status, and it may actually cause harm by increasing the risk of dehydration and UTI.4
Tube Feeding
In patients with severe dysphagia in whom conservative management is not feasible or has failed, maintaining adequate nutrition can be a challenge. There are encouraging data with nutritionally enriching and modifying the texture of solid foods.1 Alternative methods of enteral nutrition delivery are often also considered. The most common vehicles of delivery are nasogastric tubes, post-pyloric feeding tubes, and percutaneous endoscopic gastrostomy (PEG) tubes. In theory, bypassing the pharynx and esophagus could result in fewer aspiration events and less AP.3 However, nasogastric, post-pyloric, or PEG feeding does not decrease the risk of AP. For patients with advanced dementia, there have been no randomized trials demonstrating an improvement in mortality with tube feeds.4 Tube feeding also carries with it a slight procedural risk and a high incidence of associated diarrhea, plus is associated with electrolyte derangements such as hypernatremia. The decision to pursue tube feeding should be weighed heavily in every patient and is highly influenced by the etiology and anticipated duration of dysphagia.
Selective Digestive Decontamination
Selective digestive decontamination (SDD) is a protocol-based treatment that aims to eradicate potentially pathogenic gut flora, particularly aerobic gram-negatives, in critically ill patients to reduce the impact of aspiration events. The utilization of SDD and the available literature center firmly on critically ill and ventilated patients. Subsequent studies have demonstrated recolonization after protocol cessation, and long-term effects are currently undefined.5 Until it can be studied in broader populations and proven to have clinical benefit, employing SSD in non-critically ill patients with dysphagia remains unsupported.
Multimodal Approach
Many rehabilitation centers incorporate a therapist-driven swallowing treatment program. Evidence suggests patient and family counseling alone may not be effective, so these programs variably incorporate diet/liquid modification, strengthening exercises, sensory processing techniques, and even neuromuscular electrical stimulation for muscle building.1 Accordingly, these programs are resource-intensive.
Management
Dysphagia remains a significant clinical problem for hospitalized patients. The existing literature and practice guidelines generally support a “less is more” approach. Though liquid/diet modification is common practice, it is not based in solid evidence and may contribute to unnecessary tube feeding. The best current evidence supports allowing access to water and ice chips. The ideal management plan for each patient will differ and should incorporate patient and family preferences in a multidisciplinary approach.
Back to the Case
Our patient requests water. He coughs after drinking during a bedside swallow evaluation. The risks of potential aspiration and AP are explained, and he expresses his understanding. He reiterates his choice to be allowed access to water as it is important to his quality of life. The speech therapy team is consulted and provides instruction on chin-tuck positioning, oral care, and timing water between meals rather than while eating food. He does well for the remainder of the hospital stay, and by time of discharge, his electrolytes are corrected, and he is much more comfortable being allowed to drink water. He is discharged home and encouraged to continue with these conservative measures.
Bottom Line
Evidence to support many common interventions for dysphagia is lacking; patients with dysphagia are best managed via a multidisciplinary, multimodal approach that provides access to water whenever possible. TH
Vijay G. Paryani, MD, is an internal medicine resident in the department of internal medicine at the University of Kentucky. Joseph R. Sweigart, MD, is a hospitalist and assistant professor of hospital medicine in the division of hospital medicine at the University of Kentucky. Laura C. Fanucchi, MD, is a hospitalist and assistant professor of hospital medicine in the division of hospital medicine at the University of Kentucky.
References
- Karagiannis MJ, Chivers L, Karagiannis TC. Effects of oral intake of water in patients with oropharyngeal dysphagia. BMC Geriatr. 2011;11(2):9.
- Foley N, Teasell R, Salter K, Kruger E, Martino R. Dysphagia treatment post stroke: a systematic review of randomized controlled trials. Age Ageing. 2008;37(3):258-264.
- Marik PE. Aspiration pneumonitis and aspiration pneumonia. N Engl J Med. 2001;344(9):665-671.
- Loeb MB, Becker M, Eady A, Walker-Dilks C. Interventions to prevent aspiration pneumonia in older adults: a systematic review. J Am Geriatr Soc. 2003;51(7):1018-1022.
- Gosney M, Martin MV, Wright AE. The role of selective decontamination of the digestive tract in acute stroke. Age Ageing 2006;35(1):42-47.