User login
Mutations could be therapeutic target for FL

Mutations in the RRAGC gene appear to be an “excellent candidate for therapeutic targeting” in follicular lymphoma (FL), according to investigators.
They analyzed mutations found in tumors with multiple relapses of FL without transformation to diffuse large B-cell lymphoma.
And they found that one commonly mutated gene encodes the protein RagC, which is essential for activating the amino-acid sensing mTORC1 pathway.
Although mutations in genes in the mTORC1 pathway have been associated with various cancers, this is the first time a genetic mutation in any of the 4 Rag proteins has been identified in malignancy.
“One of the mutations that we have identified allows follicular lymphoma tumors to turn on growth signals regardless of whether nutrients are available, thereby evading normal restrictions on its growth,” said study author Jessica Okosun, MB BChir, PhD, of Barts Cancer Institute at Queen Mary University of London in the UK.
“Remarkably, the mutations we have discovered have not been seen in other cancer types. However, drugs that directly target this nutrient-sensing mechanism are currently used to treat other types of cancer and may benefit patients with follicular lymphoma.”
Dr Okosun and her colleagues reported these findings in a letter to Nature Genetics.
In experiments with cell lines, the investigators found that expression of the mutated RagC proteins activate mTORC1 signaling in the absence of amino acids and increase binding to an important part of the mTORC1 complex, consistent with the established role of RagC in the mTORC1 pathway.
Because this research was performed exclusively in cell lines, the investigators have not yet deciphered the mutations’ mechanistic effect in patients. However, study author Rachel Wolfson, of the Whitehead Institute for Biomedical Research and Massachusetts Institute of Technology in Cambridge, Massachusetts, said there are clues to the mutations’ significance.
“mTORC1 is linked to cell growth, so it is not surprising that activation of the pathway could lead to some growth advantage for cancer cells,” Wolfson said. “But it leads to an interesting question: When is it a proliferative advantage versus a disadvantage to no longer be able to accurately sense amino acid levels? That is something we would need to investigate further, likely in vivo.”
The investigators would also like to know how the drug rapamycin affects FL with RagC mutations. Rapamycin binds to mTORC1 and inhibits its activity. If the drug interferes with mTORC1 dysregulation caused by RagC mutations, perhaps the drug could be used in FL treatment.
“If so, maybe these RagC mutations could be used as biomarkers to predict sensitivity to rapamycin treatment in follicular lymphoma patients,” Wolfson said. “That would be very exciting, and it’s something that should be investigated further.” ![]()

Mutations in the RRAGC gene appear to be an “excellent candidate for therapeutic targeting” in follicular lymphoma (FL), according to investigators.
They analyzed mutations found in tumors with multiple relapses of FL without transformation to diffuse large B-cell lymphoma.
And they found that one commonly mutated gene encodes the protein RagC, which is essential for activating the amino-acid sensing mTORC1 pathway.
Although mutations in genes in the mTORC1 pathway have been associated with various cancers, this is the first time a genetic mutation in any of the 4 Rag proteins has been identified in malignancy.
“One of the mutations that we have identified allows follicular lymphoma tumors to turn on growth signals regardless of whether nutrients are available, thereby evading normal restrictions on its growth,” said study author Jessica Okosun, MB BChir, PhD, of Barts Cancer Institute at Queen Mary University of London in the UK.
“Remarkably, the mutations we have discovered have not been seen in other cancer types. However, drugs that directly target this nutrient-sensing mechanism are currently used to treat other types of cancer and may benefit patients with follicular lymphoma.”
Dr Okosun and her colleagues reported these findings in a letter to Nature Genetics.
In experiments with cell lines, the investigators found that expression of the mutated RagC proteins activate mTORC1 signaling in the absence of amino acids and increase binding to an important part of the mTORC1 complex, consistent with the established role of RagC in the mTORC1 pathway.
Because this research was performed exclusively in cell lines, the investigators have not yet deciphered the mutations’ mechanistic effect in patients. However, study author Rachel Wolfson, of the Whitehead Institute for Biomedical Research and Massachusetts Institute of Technology in Cambridge, Massachusetts, said there are clues to the mutations’ significance.
“mTORC1 is linked to cell growth, so it is not surprising that activation of the pathway could lead to some growth advantage for cancer cells,” Wolfson said. “But it leads to an interesting question: When is it a proliferative advantage versus a disadvantage to no longer be able to accurately sense amino acid levels? That is something we would need to investigate further, likely in vivo.”
The investigators would also like to know how the drug rapamycin affects FL with RagC mutations. Rapamycin binds to mTORC1 and inhibits its activity. If the drug interferes with mTORC1 dysregulation caused by RagC mutations, perhaps the drug could be used in FL treatment.
“If so, maybe these RagC mutations could be used as biomarkers to predict sensitivity to rapamycin treatment in follicular lymphoma patients,” Wolfson said. “That would be very exciting, and it’s something that should be investigated further.” ![]()

Mutations in the RRAGC gene appear to be an “excellent candidate for therapeutic targeting” in follicular lymphoma (FL), according to investigators.
They analyzed mutations found in tumors with multiple relapses of FL without transformation to diffuse large B-cell lymphoma.
And they found that one commonly mutated gene encodes the protein RagC, which is essential for activating the amino-acid sensing mTORC1 pathway.
Although mutations in genes in the mTORC1 pathway have been associated with various cancers, this is the first time a genetic mutation in any of the 4 Rag proteins has been identified in malignancy.
“One of the mutations that we have identified allows follicular lymphoma tumors to turn on growth signals regardless of whether nutrients are available, thereby evading normal restrictions on its growth,” said study author Jessica Okosun, MB BChir, PhD, of Barts Cancer Institute at Queen Mary University of London in the UK.
“Remarkably, the mutations we have discovered have not been seen in other cancer types. However, drugs that directly target this nutrient-sensing mechanism are currently used to treat other types of cancer and may benefit patients with follicular lymphoma.”
Dr Okosun and her colleagues reported these findings in a letter to Nature Genetics.
In experiments with cell lines, the investigators found that expression of the mutated RagC proteins activate mTORC1 signaling in the absence of amino acids and increase binding to an important part of the mTORC1 complex, consistent with the established role of RagC in the mTORC1 pathway.
Because this research was performed exclusively in cell lines, the investigators have not yet deciphered the mutations’ mechanistic effect in patients. However, study author Rachel Wolfson, of the Whitehead Institute for Biomedical Research and Massachusetts Institute of Technology in Cambridge, Massachusetts, said there are clues to the mutations’ significance.
“mTORC1 is linked to cell growth, so it is not surprising that activation of the pathway could lead to some growth advantage for cancer cells,” Wolfson said. “But it leads to an interesting question: When is it a proliferative advantage versus a disadvantage to no longer be able to accurately sense amino acid levels? That is something we would need to investigate further, likely in vivo.”
The investigators would also like to know how the drug rapamycin affects FL with RagC mutations. Rapamycin binds to mTORC1 and inhibits its activity. If the drug interferes with mTORC1 dysregulation caused by RagC mutations, perhaps the drug could be used in FL treatment.
“If so, maybe these RagC mutations could be used as biomarkers to predict sensitivity to rapamycin treatment in follicular lymphoma patients,” Wolfson said. “That would be very exciting, and it’s something that should be investigated further.” ![]()
Studies reveal lack of transparency and poor reporting of research

Photo by Bill Branson
Two new studies suggest biomedical research may be hindered by poor reporting and a lack of transparency.
In one study, researchers analyzed more than 400 biomedical science articles and found the papers rarely provided full protocol information, complete data, and the necessary level of transparency to verify or replicate the work.
In the other study, researchers analyzed more than 500 preclinical experiments and found that most didn’t contain sufficient information on the animals used.
Both studies were published in PLOS Biology.
For the first study, Shareen Iqbal, PhD, of Emory University in Atlanta, Georgia, and her colleagues analyzed papers published between 2000 and 2014.
The team set out to determine the extent to which researchers report key information necessary for properly evaluating and replicating published research, including availability of protocols, data, and the frequency of published novel or replication studies.
Out of 441 articles drawn from across the biomedical literature, only 1 paper provided a full protocol, and none of the papers made all the data available. The majority of studies didn’t state funding or conflicts of interest, and replication studies were very rare.
Dr Iqbal and her colleagues said they hope their study will further sensitize scientists, funders, journals, and other science-related stakeholders about the need to improve these indicators.
For the second study, Ulrich Dirnagl, MD, of Charité Universitätsmedizin in Berlin, Germany, and his colleagues examined 100 papers describing preclinical research on stroke and cancer. These papers contained accounts of 316 experiments on infarct volume and 206 experiments on tumor shrinkage.
The vast majority of the reports didn’t contain sufficient information on how many animals were used in the experiments. What’s more, in many papers, animals “vanished” over the course of the study.
Using a computer model, the researchers simulated the effects of such animal loss on the validity of the experiments. They found that the more animals lost or removed, the shakier or more biased the experimental conclusions.
“The study began with an attempt to look at the robustness of findings in a handful of preclinical papers, but the sheer number of missing animals stopped us in our tracks,” said author Constance Holman, a graduate student at Charité Universitätsmedizin.
Researchers from both studies believe their findings add to the list of concerns about bias and reporting in research, but the results also establish ways in which research can become more transparent and potentially more reproducible. ![]()

Photo by Bill Branson
Two new studies suggest biomedical research may be hindered by poor reporting and a lack of transparency.
In one study, researchers analyzed more than 400 biomedical science articles and found the papers rarely provided full protocol information, complete data, and the necessary level of transparency to verify or replicate the work.
In the other study, researchers analyzed more than 500 preclinical experiments and found that most didn’t contain sufficient information on the animals used.
Both studies were published in PLOS Biology.
For the first study, Shareen Iqbal, PhD, of Emory University in Atlanta, Georgia, and her colleagues analyzed papers published between 2000 and 2014.
The team set out to determine the extent to which researchers report key information necessary for properly evaluating and replicating published research, including availability of protocols, data, and the frequency of published novel or replication studies.
Out of 441 articles drawn from across the biomedical literature, only 1 paper provided a full protocol, and none of the papers made all the data available. The majority of studies didn’t state funding or conflicts of interest, and replication studies were very rare.
Dr Iqbal and her colleagues said they hope their study will further sensitize scientists, funders, journals, and other science-related stakeholders about the need to improve these indicators.
For the second study, Ulrich Dirnagl, MD, of Charité Universitätsmedizin in Berlin, Germany, and his colleagues examined 100 papers describing preclinical research on stroke and cancer. These papers contained accounts of 316 experiments on infarct volume and 206 experiments on tumor shrinkage.
The vast majority of the reports didn’t contain sufficient information on how many animals were used in the experiments. What’s more, in many papers, animals “vanished” over the course of the study.
Using a computer model, the researchers simulated the effects of such animal loss on the validity of the experiments. They found that the more animals lost or removed, the shakier or more biased the experimental conclusions.
“The study began with an attempt to look at the robustness of findings in a handful of preclinical papers, but the sheer number of missing animals stopped us in our tracks,” said author Constance Holman, a graduate student at Charité Universitätsmedizin.
Researchers from both studies believe their findings add to the list of concerns about bias and reporting in research, but the results also establish ways in which research can become more transparent and potentially more reproducible. ![]()

Photo by Bill Branson
Two new studies suggest biomedical research may be hindered by poor reporting and a lack of transparency.
In one study, researchers analyzed more than 400 biomedical science articles and found the papers rarely provided full protocol information, complete data, and the necessary level of transparency to verify or replicate the work.
In the other study, researchers analyzed more than 500 preclinical experiments and found that most didn’t contain sufficient information on the animals used.
Both studies were published in PLOS Biology.
For the first study, Shareen Iqbal, PhD, of Emory University in Atlanta, Georgia, and her colleagues analyzed papers published between 2000 and 2014.
The team set out to determine the extent to which researchers report key information necessary for properly evaluating and replicating published research, including availability of protocols, data, and the frequency of published novel or replication studies.
Out of 441 articles drawn from across the biomedical literature, only 1 paper provided a full protocol, and none of the papers made all the data available. The majority of studies didn’t state funding or conflicts of interest, and replication studies were very rare.
Dr Iqbal and her colleagues said they hope their study will further sensitize scientists, funders, journals, and other science-related stakeholders about the need to improve these indicators.
For the second study, Ulrich Dirnagl, MD, of Charité Universitätsmedizin in Berlin, Germany, and his colleagues examined 100 papers describing preclinical research on stroke and cancer. These papers contained accounts of 316 experiments on infarct volume and 206 experiments on tumor shrinkage.
The vast majority of the reports didn’t contain sufficient information on how many animals were used in the experiments. What’s more, in many papers, animals “vanished” over the course of the study.
Using a computer model, the researchers simulated the effects of such animal loss on the validity of the experiments. They found that the more animals lost or removed, the shakier or more biased the experimental conclusions.
“The study began with an attempt to look at the robustness of findings in a handful of preclinical papers, but the sheer number of missing animals stopped us in our tracks,” said author Constance Holman, a graduate student at Charité Universitätsmedizin.
Researchers from both studies believe their findings add to the list of concerns about bias and reporting in research, but the results also establish ways in which research can become more transparent and potentially more reproducible. ![]()
Drug granted orphan designation for SCD
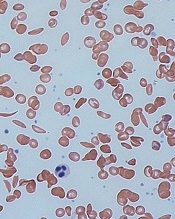
red blood cells
Image by Graham Beards
The US Food and Drug Administration (FDA) has granted orphan drug designation for the small molecule GBT440 to treat patients with sickle cell disease (SCD).
GBT440 is being developed as a potentially disease-modifying therapy for SCD. The drug works by increasing hemoglobin’s affinity for oxygen.
Since oxygenated sickle hemoglobin does not polymerize, it is believed that GBT440 blocks polymerization and the resultant sickling of red blood cells.
If GBT440 can restore normal hemoglobin function and improve oxygen delivery, the drug may be capable of modifying the progression of SCD.
“Receiving orphan drug designation, along with the previously announced fast track designation, are important milestones in our regulatory strategy for GBT440 and highlight the FDA’s agreement that the SCD community faces a critical need for new treatments,” said Ted W. Love, MD, chief executive officer of Global Blood Therapeutics, Inc., the company developing GBT440.
The FDA grants orphan designation to drugs that are intended to treat diseases or conditions affecting fewer than 200,000 patients in the US. The designation provides the drug’s sponsor with various development incentives, including opportunities to apply for research-related tax credits and grant funding, assistance in designing clinical trials, and 7 years of US market exclusivity if the drug is approved.
The FDA grants fast track designation to facilitate and expedite the development and review of new drugs intended to treat serious or life-threatening conditions and address unmet medical need. Through the fast track program, a drug may be eligible for priority review and rolling review, and the company developing the drug may receive additional help from the FDA to expedite development.
GBT440 trial
Early results from an ongoing phase 1/2 study of GBT440 were presented at the 2015 ASH Annual Meeting last month (abstract 542*).
The trial, which includes healthy subjects and patients with SCD, is being conducted in 2 parts: part A (single-dose administration) and part B (multiple-dose administration, daily for 15 days in healthy subjects and 28 days in SCD patients).
As of November 20, 2015, 8 SCD patients completed part A, and 30 SCD patients had either completed or were ongoing in part B.
Of the 30 SCD patients, 16 patients completed 700 mg daily dosing and follow-up (12 on GBT440 and 4 on placebo), and 14 patients completed or were ongoing at 500 mg daily dosing and follow-up (10 on GBT440 and 4 on placebo). A cohort of SCD patients on 1000 mg per day for 28 days is currently enrolling.
Thus far, GBT440 treatment has conferred several improvements from baseline to day 28.
Hemoglobin increases were evident by day 4 of treatment. And the researchers observed absolute hemoglobin increases of 0.5 and 0.7 g/dL with GBT440 at 500 and 700 mg, respectively, compared with a 0.1 g/dL decrease with placebo.
The median reticulocyte count decreased by 31% and 37% with GBT440 at 500 and 700 mg, respectively, compared with a 7% increase with placebo, indicating that the hemoglobin rise is due to decreased hemolysis.
Median erythropoietin levels decreased by 9 and 18 mU/mL with GBT440 at 500 and 700 mg, respectively, compared with an increase of 28 mU/mL with placebo.
Median unconjugated bilirubin levels decrease by 31% and 43% with GBT440 at 500 mg and 700 mg, respectively, compared with an increase of 2% with placebo.
Median lactate dehydrogenase levels decreased by 20% and 12% with GBT440 at 500 and 700 mg, respectively, compared with a decrease of 7% with placebo.
Median sickle cell counts decreased by 56% and 46% with GBT440 at 500 and 700 mg, respectively, compared with a 14% increase with placebo.
The researchers noted high inter- and intra-patient variability in circulating sickle cell counts.
They said inflammatory soluble adhesion molecules for the 700 mg dose cohort showed promising trends in improvement. The median P-selectin decreased 19%, compared with an increase of 20% with placebo. And the median ICAM-1 decreased 6%, compared with an increase of 33% in placebo. Data for the 500 mg dose cohort has not yet been analyzed.
The researchers said pharmacokinetic data demonstrated linear and dose-proportional properties, with a half-life amenable to once-daily dosing.
And GBT440 was well tolerated over the 28 days of dosing. None of the SCD patients discontinued GBT440. The most common adverse event was headache, and there have been no serious adverse events thought to be drug-related.
“We continue to believe that GBT440 has the potential to become the first mechanism-based and disease-modifying therapeutic for this grievous disease and look forward to sharing full results from our phase 1/2 trial and potentially initiating a pivotal trial in adult patients with SCD in 2016,” Dr Love said. ![]()
*Data in the abstract differ from the presentation.

red blood cells
Image by Graham Beards
The US Food and Drug Administration (FDA) has granted orphan drug designation for the small molecule GBT440 to treat patients with sickle cell disease (SCD).
GBT440 is being developed as a potentially disease-modifying therapy for SCD. The drug works by increasing hemoglobin’s affinity for oxygen.
Since oxygenated sickle hemoglobin does not polymerize, it is believed that GBT440 blocks polymerization and the resultant sickling of red blood cells.
If GBT440 can restore normal hemoglobin function and improve oxygen delivery, the drug may be capable of modifying the progression of SCD.
“Receiving orphan drug designation, along with the previously announced fast track designation, are important milestones in our regulatory strategy for GBT440 and highlight the FDA’s agreement that the SCD community faces a critical need for new treatments,” said Ted W. Love, MD, chief executive officer of Global Blood Therapeutics, Inc., the company developing GBT440.
The FDA grants orphan designation to drugs that are intended to treat diseases or conditions affecting fewer than 200,000 patients in the US. The designation provides the drug’s sponsor with various development incentives, including opportunities to apply for research-related tax credits and grant funding, assistance in designing clinical trials, and 7 years of US market exclusivity if the drug is approved.
The FDA grants fast track designation to facilitate and expedite the development and review of new drugs intended to treat serious or life-threatening conditions and address unmet medical need. Through the fast track program, a drug may be eligible for priority review and rolling review, and the company developing the drug may receive additional help from the FDA to expedite development.
GBT440 trial
Early results from an ongoing phase 1/2 study of GBT440 were presented at the 2015 ASH Annual Meeting last month (abstract 542*).
The trial, which includes healthy subjects and patients with SCD, is being conducted in 2 parts: part A (single-dose administration) and part B (multiple-dose administration, daily for 15 days in healthy subjects and 28 days in SCD patients).
As of November 20, 2015, 8 SCD patients completed part A, and 30 SCD patients had either completed or were ongoing in part B.
Of the 30 SCD patients, 16 patients completed 700 mg daily dosing and follow-up (12 on GBT440 and 4 on placebo), and 14 patients completed or were ongoing at 500 mg daily dosing and follow-up (10 on GBT440 and 4 on placebo). A cohort of SCD patients on 1000 mg per day for 28 days is currently enrolling.
Thus far, GBT440 treatment has conferred several improvements from baseline to day 28.
Hemoglobin increases were evident by day 4 of treatment. And the researchers observed absolute hemoglobin increases of 0.5 and 0.7 g/dL with GBT440 at 500 and 700 mg, respectively, compared with a 0.1 g/dL decrease with placebo.
The median reticulocyte count decreased by 31% and 37% with GBT440 at 500 and 700 mg, respectively, compared with a 7% increase with placebo, indicating that the hemoglobin rise is due to decreased hemolysis.
Median erythropoietin levels decreased by 9 and 18 mU/mL with GBT440 at 500 and 700 mg, respectively, compared with an increase of 28 mU/mL with placebo.
Median unconjugated bilirubin levels decrease by 31% and 43% with GBT440 at 500 mg and 700 mg, respectively, compared with an increase of 2% with placebo.
Median lactate dehydrogenase levels decreased by 20% and 12% with GBT440 at 500 and 700 mg, respectively, compared with a decrease of 7% with placebo.
Median sickle cell counts decreased by 56% and 46% with GBT440 at 500 and 700 mg, respectively, compared with a 14% increase with placebo.
The researchers noted high inter- and intra-patient variability in circulating sickle cell counts.
They said inflammatory soluble adhesion molecules for the 700 mg dose cohort showed promising trends in improvement. The median P-selectin decreased 19%, compared with an increase of 20% with placebo. And the median ICAM-1 decreased 6%, compared with an increase of 33% in placebo. Data for the 500 mg dose cohort has not yet been analyzed.
The researchers said pharmacokinetic data demonstrated linear and dose-proportional properties, with a half-life amenable to once-daily dosing.
And GBT440 was well tolerated over the 28 days of dosing. None of the SCD patients discontinued GBT440. The most common adverse event was headache, and there have been no serious adverse events thought to be drug-related.
“We continue to believe that GBT440 has the potential to become the first mechanism-based and disease-modifying therapeutic for this grievous disease and look forward to sharing full results from our phase 1/2 trial and potentially initiating a pivotal trial in adult patients with SCD in 2016,” Dr Love said. ![]()
*Data in the abstract differ from the presentation.

red blood cells
Image by Graham Beards
The US Food and Drug Administration (FDA) has granted orphan drug designation for the small molecule GBT440 to treat patients with sickle cell disease (SCD).
GBT440 is being developed as a potentially disease-modifying therapy for SCD. The drug works by increasing hemoglobin’s affinity for oxygen.
Since oxygenated sickle hemoglobin does not polymerize, it is believed that GBT440 blocks polymerization and the resultant sickling of red blood cells.
If GBT440 can restore normal hemoglobin function and improve oxygen delivery, the drug may be capable of modifying the progression of SCD.
“Receiving orphan drug designation, along with the previously announced fast track designation, are important milestones in our regulatory strategy for GBT440 and highlight the FDA’s agreement that the SCD community faces a critical need for new treatments,” said Ted W. Love, MD, chief executive officer of Global Blood Therapeutics, Inc., the company developing GBT440.
The FDA grants orphan designation to drugs that are intended to treat diseases or conditions affecting fewer than 200,000 patients in the US. The designation provides the drug’s sponsor with various development incentives, including opportunities to apply for research-related tax credits and grant funding, assistance in designing clinical trials, and 7 years of US market exclusivity if the drug is approved.
The FDA grants fast track designation to facilitate and expedite the development and review of new drugs intended to treat serious or life-threatening conditions and address unmet medical need. Through the fast track program, a drug may be eligible for priority review and rolling review, and the company developing the drug may receive additional help from the FDA to expedite development.
GBT440 trial
Early results from an ongoing phase 1/2 study of GBT440 were presented at the 2015 ASH Annual Meeting last month (abstract 542*).
The trial, which includes healthy subjects and patients with SCD, is being conducted in 2 parts: part A (single-dose administration) and part B (multiple-dose administration, daily for 15 days in healthy subjects and 28 days in SCD patients).
As of November 20, 2015, 8 SCD patients completed part A, and 30 SCD patients had either completed or were ongoing in part B.
Of the 30 SCD patients, 16 patients completed 700 mg daily dosing and follow-up (12 on GBT440 and 4 on placebo), and 14 patients completed or were ongoing at 500 mg daily dosing and follow-up (10 on GBT440 and 4 on placebo). A cohort of SCD patients on 1000 mg per day for 28 days is currently enrolling.
Thus far, GBT440 treatment has conferred several improvements from baseline to day 28.
Hemoglobin increases were evident by day 4 of treatment. And the researchers observed absolute hemoglobin increases of 0.5 and 0.7 g/dL with GBT440 at 500 and 700 mg, respectively, compared with a 0.1 g/dL decrease with placebo.
The median reticulocyte count decreased by 31% and 37% with GBT440 at 500 and 700 mg, respectively, compared with a 7% increase with placebo, indicating that the hemoglobin rise is due to decreased hemolysis.
Median erythropoietin levels decreased by 9 and 18 mU/mL with GBT440 at 500 and 700 mg, respectively, compared with an increase of 28 mU/mL with placebo.
Median unconjugated bilirubin levels decrease by 31% and 43% with GBT440 at 500 mg and 700 mg, respectively, compared with an increase of 2% with placebo.
Median lactate dehydrogenase levels decreased by 20% and 12% with GBT440 at 500 and 700 mg, respectively, compared with a decrease of 7% with placebo.
Median sickle cell counts decreased by 56% and 46% with GBT440 at 500 and 700 mg, respectively, compared with a 14% increase with placebo.
The researchers noted high inter- and intra-patient variability in circulating sickle cell counts.
They said inflammatory soluble adhesion molecules for the 700 mg dose cohort showed promising trends in improvement. The median P-selectin decreased 19%, compared with an increase of 20% with placebo. And the median ICAM-1 decreased 6%, compared with an increase of 33% in placebo. Data for the 500 mg dose cohort has not yet been analyzed.
The researchers said pharmacokinetic data demonstrated linear and dose-proportional properties, with a half-life amenable to once-daily dosing.
And GBT440 was well tolerated over the 28 days of dosing. None of the SCD patients discontinued GBT440. The most common adverse event was headache, and there have been no serious adverse events thought to be drug-related.
“We continue to believe that GBT440 has the potential to become the first mechanism-based and disease-modifying therapeutic for this grievous disease and look forward to sharing full results from our phase 1/2 trial and potentially initiating a pivotal trial in adult patients with SCD in 2016,” Dr Love said. ![]()
*Data in the abstract differ from the presentation.
Docs’ body language may convey racial bias

Doctors may convey racial bias with their body language, according to research published in The Journal of Pain and Symptom Management.
In this small study, a group of physicians, most of whom were white males, gave less compassionate nonverbal cues when interacting with black actors portraying seriously ill patients than when interacting with white actors portraying seriously ill patients.
“Although we found that physicians said the same things to their black and white patients, communication is not just the spoken word. It also involves nonverbal cues, such as eye contact, body positioning, and touch,” said study author Amber Barnato, MD, of the University of Pittsburg in Pennsylvania.
“Poor nonverbal communication—something the physician may not even be aware he or she is doing—could explain why many black patients perceive discrimination in the healthcare setting.”
For this study, Dr Barnato and her colleagues recruited 33 hospital-based attending emergency medicine physicians, hospitalists, and intensivists from Allegheny County, Pennsylvania, and put them in realistic simulations where actors portrayed dying black and white patients accompanied by a family member.
The actors portrayed comparable medical conditions—plummeting vital signs related to either metastatic gastric or pancreatic cancer—and read from matching scripts. The physicians were unaware of what the trial was testing.
The majority of the physicians were white men, so the researchers could not derive any statistically significant conclusions about whether a physician’s race impacted his or her actions.
Physicians were scored on a point system for both their verbal and nonverbal communication skills when interacting with the patient and family member. The physicians averaged 7% lower scores for their nonverbal interactions with the black patients than with the white patients.
“When explaining what was happening and what the next steps for care could be, with the white patients, the physicians were more likely to stand right at the patient’s bedside and touch them in a sympathetic manner,” Dr Barnato said.
She explained that something as simple as a physician staying near the door and holding a binder in front of his body could be perceived by the patient and family as defensive or disengaged. This could lead to a cascade of misunderstandings that result in patients and their families requesting extraordinary life-saving measures because they don’t trust the doctor has their best interests in mind when suggesting gentler, end-of-life care options.
“When you survey people in the community about their feelings on end-of-life care, blacks are only slightly more likely than whites to say they want aggressive, life-sustaining measures when terminally ill,” Dr Barnato said.
“However, blacks are much more likely than whites to request such care when they are faced with making the decision in the hospital. Body language is a significant tool in building trust—or mistrust—and physicians need to ensure that their body language isn’t contributing to that decision.”
“To help black patients and their families feel welcome and encouraged to be partners in medical decision-making, it is critical that doctors be aware of their verbal and nonverbal communication and any unintentional biases.” ![]()

Doctors may convey racial bias with their body language, according to research published in The Journal of Pain and Symptom Management.
In this small study, a group of physicians, most of whom were white males, gave less compassionate nonverbal cues when interacting with black actors portraying seriously ill patients than when interacting with white actors portraying seriously ill patients.
“Although we found that physicians said the same things to their black and white patients, communication is not just the spoken word. It also involves nonverbal cues, such as eye contact, body positioning, and touch,” said study author Amber Barnato, MD, of the University of Pittsburg in Pennsylvania.
“Poor nonverbal communication—something the physician may not even be aware he or she is doing—could explain why many black patients perceive discrimination in the healthcare setting.”
For this study, Dr Barnato and her colleagues recruited 33 hospital-based attending emergency medicine physicians, hospitalists, and intensivists from Allegheny County, Pennsylvania, and put them in realistic simulations where actors portrayed dying black and white patients accompanied by a family member.
The actors portrayed comparable medical conditions—plummeting vital signs related to either metastatic gastric or pancreatic cancer—and read from matching scripts. The physicians were unaware of what the trial was testing.
The majority of the physicians were white men, so the researchers could not derive any statistically significant conclusions about whether a physician’s race impacted his or her actions.
Physicians were scored on a point system for both their verbal and nonverbal communication skills when interacting with the patient and family member. The physicians averaged 7% lower scores for their nonverbal interactions with the black patients than with the white patients.
“When explaining what was happening and what the next steps for care could be, with the white patients, the physicians were more likely to stand right at the patient’s bedside and touch them in a sympathetic manner,” Dr Barnato said.
She explained that something as simple as a physician staying near the door and holding a binder in front of his body could be perceived by the patient and family as defensive or disengaged. This could lead to a cascade of misunderstandings that result in patients and their families requesting extraordinary life-saving measures because they don’t trust the doctor has their best interests in mind when suggesting gentler, end-of-life care options.
“When you survey people in the community about their feelings on end-of-life care, blacks are only slightly more likely than whites to say they want aggressive, life-sustaining measures when terminally ill,” Dr Barnato said.
“However, blacks are much more likely than whites to request such care when they are faced with making the decision in the hospital. Body language is a significant tool in building trust—or mistrust—and physicians need to ensure that their body language isn’t contributing to that decision.”
“To help black patients and their families feel welcome and encouraged to be partners in medical decision-making, it is critical that doctors be aware of their verbal and nonverbal communication and any unintentional biases.” ![]()

Doctors may convey racial bias with their body language, according to research published in The Journal of Pain and Symptom Management.
In this small study, a group of physicians, most of whom were white males, gave less compassionate nonverbal cues when interacting with black actors portraying seriously ill patients than when interacting with white actors portraying seriously ill patients.
“Although we found that physicians said the same things to their black and white patients, communication is not just the spoken word. It also involves nonverbal cues, such as eye contact, body positioning, and touch,” said study author Amber Barnato, MD, of the University of Pittsburg in Pennsylvania.
“Poor nonverbal communication—something the physician may not even be aware he or she is doing—could explain why many black patients perceive discrimination in the healthcare setting.”
For this study, Dr Barnato and her colleagues recruited 33 hospital-based attending emergency medicine physicians, hospitalists, and intensivists from Allegheny County, Pennsylvania, and put them in realistic simulations where actors portrayed dying black and white patients accompanied by a family member.
The actors portrayed comparable medical conditions—plummeting vital signs related to either metastatic gastric or pancreatic cancer—and read from matching scripts. The physicians were unaware of what the trial was testing.
The majority of the physicians were white men, so the researchers could not derive any statistically significant conclusions about whether a physician’s race impacted his or her actions.
Physicians were scored on a point system for both their verbal and nonverbal communication skills when interacting with the patient and family member. The physicians averaged 7% lower scores for their nonverbal interactions with the black patients than with the white patients.
“When explaining what was happening and what the next steps for care could be, with the white patients, the physicians were more likely to stand right at the patient’s bedside and touch them in a sympathetic manner,” Dr Barnato said.
She explained that something as simple as a physician staying near the door and holding a binder in front of his body could be perceived by the patient and family as defensive or disengaged. This could lead to a cascade of misunderstandings that result in patients and their families requesting extraordinary life-saving measures because they don’t trust the doctor has their best interests in mind when suggesting gentler, end-of-life care options.
“When you survey people in the community about their feelings on end-of-life care, blacks are only slightly more likely than whites to say they want aggressive, life-sustaining measures when terminally ill,” Dr Barnato said.
“However, blacks are much more likely than whites to request such care when they are faced with making the decision in the hospital. Body language is a significant tool in building trust—or mistrust—and physicians need to ensure that their body language isn’t contributing to that decision.”
“To help black patients and their families feel welcome and encouraged to be partners in medical decision-making, it is critical that doctors be aware of their verbal and nonverbal communication and any unintentional biases.” ![]()
Antiepileptic Drugs and Hyponatremia
Call for abstracts: Gut Microbiota for Health World Summit 2016
Share your research at the Gut Microbiota for Health World Summit 2016 with an international audience of researchers and clinicians.
Students, medical residents, trainees, postdoctoral fellows and junior faculty (within 3 years of their first appointment) are invited to submit an abstract for oral or poster presentation. Only a select number of abstracts will be accepted for oral presentation.
If selected, presenters will receive discounted registration and a travel grant to help cover the cost of airfare and accommodations. Abstracts must be submitted to Sandra Megally at [email protected] no later than Sunday, Jan. 17, 2016.
The summit is cosponsored by AGA Institute, European Society of Neurogastroenterology and Motility, European Society for Pediatric Gastroenterology, Hepatology and Nutrition, European Crohn’s and Colitis Organization, and the European Association for the Study of Liver.
Gut Microbiota for Health World Summit 2016 is supported by grants from Danone Dairy and Biocodex.
Share your research at the Gut Microbiota for Health World Summit 2016 with an international audience of researchers and clinicians.
Students, medical residents, trainees, postdoctoral fellows and junior faculty (within 3 years of their first appointment) are invited to submit an abstract for oral or poster presentation. Only a select number of abstracts will be accepted for oral presentation.
If selected, presenters will receive discounted registration and a travel grant to help cover the cost of airfare and accommodations. Abstracts must be submitted to Sandra Megally at [email protected] no later than Sunday, Jan. 17, 2016.
The summit is cosponsored by AGA Institute, European Society of Neurogastroenterology and Motility, European Society for Pediatric Gastroenterology, Hepatology and Nutrition, European Crohn’s and Colitis Organization, and the European Association for the Study of Liver.
Gut Microbiota for Health World Summit 2016 is supported by grants from Danone Dairy and Biocodex.
Share your research at the Gut Microbiota for Health World Summit 2016 with an international audience of researchers and clinicians.
Students, medical residents, trainees, postdoctoral fellows and junior faculty (within 3 years of their first appointment) are invited to submit an abstract for oral or poster presentation. Only a select number of abstracts will be accepted for oral presentation.
If selected, presenters will receive discounted registration and a travel grant to help cover the cost of airfare and accommodations. Abstracts must be submitted to Sandra Megally at [email protected] no later than Sunday, Jan. 17, 2016.
The summit is cosponsored by AGA Institute, European Society of Neurogastroenterology and Motility, European Society for Pediatric Gastroenterology, Hepatology and Nutrition, European Crohn’s and Colitis Organization, and the European Association for the Study of Liver.
Gut Microbiota for Health World Summit 2016 is supported by grants from Danone Dairy and Biocodex.
Help your patients manage chronic constipation
AGA has published the latest in a series of guideline-based resources for members to provide their patients. Based on the published AGA guideline on constipation, the new patient guide explains how a gastroenterologist can help when diet and lifestyle modifications fail.
The guide also explains defecatory disorders, slow-transit and normal-transit constipation, as well as information about tests and medication options in language that is accessible to patients.
AGA’s evidence-based patient guides can help improve the efficiency of office visits and the value you provide patients.
If your patients are suffering from chronic constipation, make sure to let them know about this helpful AGA patient guide.
AGA has published the latest in a series of guideline-based resources for members to provide their patients. Based on the published AGA guideline on constipation, the new patient guide explains how a gastroenterologist can help when diet and lifestyle modifications fail.
The guide also explains defecatory disorders, slow-transit and normal-transit constipation, as well as information about tests and medication options in language that is accessible to patients.
AGA’s evidence-based patient guides can help improve the efficiency of office visits and the value you provide patients.
If your patients are suffering from chronic constipation, make sure to let them know about this helpful AGA patient guide.
AGA has published the latest in a series of guideline-based resources for members to provide their patients. Based on the published AGA guideline on constipation, the new patient guide explains how a gastroenterologist can help when diet and lifestyle modifications fail.
The guide also explains defecatory disorders, slow-transit and normal-transit constipation, as well as information about tests and medication options in language that is accessible to patients.
AGA’s evidence-based patient guides can help improve the efficiency of office visits and the value you provide patients.
If your patients are suffering from chronic constipation, make sure to let them know about this helpful AGA patient guide.
Apply for 2016 research grants
Six grant deadlines are approaching in January 2016; make sure you’re prepared to submit your application by the deadline. Awards are available for both established and young investigators.
AGA is now accepting online applications through the new Grants Management System, which streamlines the application submission process and will be used to facilitate grant reviews.
Apply now for:
• The AGA-Elsevier Gut Microbiome Pilot Research Award. It will provide $25,000 to support pilot research projects pertaining to the gut microbiome. The application deadline is Jan. 15.
• The AGA-Elsevier Pilot Research Award. This is a 1-year grant providing young investigators, instructors, research associates, or equivalents $25,000 to support pilot research projects in gastroenterology- or hepatology-related areas. Two award recipients will be selected. The deadline is Jan. 15.
• The AGA–Rome Foundation Functional GI and Motility Disorders Pilot Research Award. This research initiative grant for $50,000 for 1 year is offered to early-stage investigators to support pilot research projects pertaining to functional GI and motility disorders. This award is jointly sponsored by the Rome Foundation and AGA. The deadline is Jan. 15.
• The AGA–Caroline Craig Augustyn & Damian Augustyn Award in Digestive Cancer. One award of $40,000 will support a young investigator, instructor, research associate, or equivalent who currently holds a federal or nonfederal career development award devoted to conducting research related to digestive cancer. The deadline is Jan. 29.
• The AGA–Covidien Research & Development Pilot Award in Technology. This $30,000 research initiative is offered to investigators to support the research and development of novel devices or technologies that will potentially impact the diagnosis or treatment of digestive disease. The award also includes a $1,000 travel stipend to attend the 2016 AGA Technology Summit. The deadline is Jan. 29.
• The 16th AGA–June & Donald O. Castell, M.D., Esophageal Clinical Research Award. This is a $25,000 grant, which provides research and/or salary support for junior faculty involved in clinical research in esophageal diseases. The deadline is Jan. 29.
We are also now accepting applications for the AGA Investing in the Future Student Research Fellowship. This provides 12 awards at $5,000 for underrepresented minority undergraduate and medical school students to perform 8-10 weeks of research related to digestive diseases or nutrition. The deadline to apply is Feb. 5.
Complete information about these and other research awards is available on www.gastro.org. All recipients will be acknowledged at the Research Recognition Celebration at Digestive Disease Week® 2016 in San Diego.
Six grant deadlines are approaching in January 2016; make sure you’re prepared to submit your application by the deadline. Awards are available for both established and young investigators.
AGA is now accepting online applications through the new Grants Management System, which streamlines the application submission process and will be used to facilitate grant reviews.
Apply now for:
• The AGA-Elsevier Gut Microbiome Pilot Research Award. It will provide $25,000 to support pilot research projects pertaining to the gut microbiome. The application deadline is Jan. 15.
• The AGA-Elsevier Pilot Research Award. This is a 1-year grant providing young investigators, instructors, research associates, or equivalents $25,000 to support pilot research projects in gastroenterology- or hepatology-related areas. Two award recipients will be selected. The deadline is Jan. 15.
• The AGA–Rome Foundation Functional GI and Motility Disorders Pilot Research Award. This research initiative grant for $50,000 for 1 year is offered to early-stage investigators to support pilot research projects pertaining to functional GI and motility disorders. This award is jointly sponsored by the Rome Foundation and AGA. The deadline is Jan. 15.
• The AGA–Caroline Craig Augustyn & Damian Augustyn Award in Digestive Cancer. One award of $40,000 will support a young investigator, instructor, research associate, or equivalent who currently holds a federal or nonfederal career development award devoted to conducting research related to digestive cancer. The deadline is Jan. 29.
• The AGA–Covidien Research & Development Pilot Award in Technology. This $30,000 research initiative is offered to investigators to support the research and development of novel devices or technologies that will potentially impact the diagnosis or treatment of digestive disease. The award also includes a $1,000 travel stipend to attend the 2016 AGA Technology Summit. The deadline is Jan. 29.
• The 16th AGA–June & Donald O. Castell, M.D., Esophageal Clinical Research Award. This is a $25,000 grant, which provides research and/or salary support for junior faculty involved in clinical research in esophageal diseases. The deadline is Jan. 29.
We are also now accepting applications for the AGA Investing in the Future Student Research Fellowship. This provides 12 awards at $5,000 for underrepresented minority undergraduate and medical school students to perform 8-10 weeks of research related to digestive diseases or nutrition. The deadline to apply is Feb. 5.
Complete information about these and other research awards is available on www.gastro.org. All recipients will be acknowledged at the Research Recognition Celebration at Digestive Disease Week® 2016 in San Diego.
Six grant deadlines are approaching in January 2016; make sure you’re prepared to submit your application by the deadline. Awards are available for both established and young investigators.
AGA is now accepting online applications through the new Grants Management System, which streamlines the application submission process and will be used to facilitate grant reviews.
Apply now for:
• The AGA-Elsevier Gut Microbiome Pilot Research Award. It will provide $25,000 to support pilot research projects pertaining to the gut microbiome. The application deadline is Jan. 15.
• The AGA-Elsevier Pilot Research Award. This is a 1-year grant providing young investigators, instructors, research associates, or equivalents $25,000 to support pilot research projects in gastroenterology- or hepatology-related areas. Two award recipients will be selected. The deadline is Jan. 15.
• The AGA–Rome Foundation Functional GI and Motility Disorders Pilot Research Award. This research initiative grant for $50,000 for 1 year is offered to early-stage investigators to support pilot research projects pertaining to functional GI and motility disorders. This award is jointly sponsored by the Rome Foundation and AGA. The deadline is Jan. 15.
• The AGA–Caroline Craig Augustyn & Damian Augustyn Award in Digestive Cancer. One award of $40,000 will support a young investigator, instructor, research associate, or equivalent who currently holds a federal or nonfederal career development award devoted to conducting research related to digestive cancer. The deadline is Jan. 29.
• The AGA–Covidien Research & Development Pilot Award in Technology. This $30,000 research initiative is offered to investigators to support the research and development of novel devices or technologies that will potentially impact the diagnosis or treatment of digestive disease. The award also includes a $1,000 travel stipend to attend the 2016 AGA Technology Summit. The deadline is Jan. 29.
• The 16th AGA–June & Donald O. Castell, M.D., Esophageal Clinical Research Award. This is a $25,000 grant, which provides research and/or salary support for junior faculty involved in clinical research in esophageal diseases. The deadline is Jan. 29.
We are also now accepting applications for the AGA Investing in the Future Student Research Fellowship. This provides 12 awards at $5,000 for underrepresented minority undergraduate and medical school students to perform 8-10 weeks of research related to digestive diseases or nutrition. The deadline to apply is Feb. 5.
Complete information about these and other research awards is available on www.gastro.org. All recipients will be acknowledged at the Research Recognition Celebration at Digestive Disease Week® 2016 in San Diego.
Learn how to avoid a reimbursement loss
The Physician Quality Reporting System (PQRS) will apply a negative 2% payment adjustment in 2017 if practices do not report data on quality measures for covered professional services furnished to Medicare beneficiaries in 2015. By participating in AGA’s Digestive Health Recognition Program™ (www.agarecognition.org), or DHRP, members can report on quality measures and work to avoid this negative payment adjustment.
To learn more about DHRP, register for a free, 30-minute webinar taking place during these two dates (attendee.gotowebinar.com/rt/2452555612218433025):
• Jan 28, 2016, 5 p.m. ET
• Feb. 25, 2016, noon ET
The deadline to enroll for the 2015 PQRS year through DHRP is Feb. 26, 2016. Members pay $300 to enroll.
DHRP is made possible by support from AbbVie; Gilead Science, Inc.; Janssen Biotech, Inc. and Janssen Therapeutics, Division of Janssen Products, LLP; and Shire Pharmaceuticals.
The Physician Quality Reporting System (PQRS) will apply a negative 2% payment adjustment in 2017 if practices do not report data on quality measures for covered professional services furnished to Medicare beneficiaries in 2015. By participating in AGA’s Digestive Health Recognition Program™ (www.agarecognition.org), or DHRP, members can report on quality measures and work to avoid this negative payment adjustment.
To learn more about DHRP, register for a free, 30-minute webinar taking place during these two dates (attendee.gotowebinar.com/rt/2452555612218433025):
• Jan 28, 2016, 5 p.m. ET
• Feb. 25, 2016, noon ET
The deadline to enroll for the 2015 PQRS year through DHRP is Feb. 26, 2016. Members pay $300 to enroll.
DHRP is made possible by support from AbbVie; Gilead Science, Inc.; Janssen Biotech, Inc. and Janssen Therapeutics, Division of Janssen Products, LLP; and Shire Pharmaceuticals.
The Physician Quality Reporting System (PQRS) will apply a negative 2% payment adjustment in 2017 if practices do not report data on quality measures for covered professional services furnished to Medicare beneficiaries in 2015. By participating in AGA’s Digestive Health Recognition Program™ (www.agarecognition.org), or DHRP, members can report on quality measures and work to avoid this negative payment adjustment.
To learn more about DHRP, register for a free, 30-minute webinar taking place during these two dates (attendee.gotowebinar.com/rt/2452555612218433025):
• Jan 28, 2016, 5 p.m. ET
• Feb. 25, 2016, noon ET
The deadline to enroll for the 2015 PQRS year through DHRP is Feb. 26, 2016. Members pay $300 to enroll.
DHRP is made possible by support from AbbVie; Gilead Science, Inc.; Janssen Biotech, Inc. and Janssen Therapeutics, Division of Janssen Products, LLP; and Shire Pharmaceuticals.
Medical Roundtable: Peripheral T-Cell Lymphomas: A Practical Approach to Newly Diagnosed and Relapsed Patients
Moderator: Steven M. Horwitz, MD1
Discussants: Alison Moskowitz, MD1; Michelle Fanale, MD2; Andrei Shustov, MD3
From Memorial Sloan Kettering Cancer Center, New York, NY1; MD Anderson Cancer Center, Houston, TX2; University of Washington Medical Center, Seattle, WA3
Address for correspondence: Steven M. Horwitz, MD, Memorial Sloan Kettering Cancer Center, 1275 York Avenue, New York, NY 10065
E-mail: [email protected]
DR. HORWITZ: My name is Dr. Steven Horwitz from Memorial Sloan-Kettering Cancer Center. I’m joined today by Drs. Alison Moskowitz, Memorial Sloan-Kettering, Michelle Fanale of MD Anderson, and Andrei Shustov, University of Washington in Seattle. Thank you all for joining us for this conversation on T-cell lymphoma. My colleagues are all well known as experts in T-cell lymphomas. Those of you who treat these diseases recognize the systemic T-cell lymphomas as one of our greater challenges in hematologic malignancies in terms of the treatment options for patients and the frequent lack of definitive data to guide our decisions.
I thought what we would do today is have a very practical discussion about the way we think about these diseases, the decisions we make, and the way we make those decisions. I'll start off by asking when you get a referral of a patient with a new diagnosis of peripheral T-cell lymphoma (PTCL), what are some of the basic things that you first think about in terms of approaching a new patient?
DR. SHUSTOV: I think one of the biggest challenges in T-cell lymphomas still remains making the proper diagnosis. In general, pathologists in the United States have a pretty good idea when they see T-cell lymphomas, however, subclassification remains a challenge even for expert hematopathologists due to frequent histologic overlap between the subtypes of PTCL, and even with non-malignant autoimmune disorders. I frequently see patients who are diagnosed with or misdiagnosed with a different subtype of T-cell lymphoma. The most challenging is differentiation between angioimmunoblastic T-cell lymphoma (AITL), anaplastic large cell T-cell lymphoma (ALCL), and PTCL not otherwise specified (PTCL-NOS), especially when the latter patients have high expression of CD30 and/or bear features resembling AITL.
Sometimes they are a slam-dunk diagnosis, but frequently our hematopathologists reverse the diagnosis after doing additional studies on the biopsy material. The most recent case I've seen in my clinic for consultation was a patient with a diagnosis of extranodal NK-cell lymphoma that was reclassified as a gamma-delta T-cell lymphoma after additional work-up. I truly believe that it is advisable that majority, if not all PTCL cases are reviewed by expert hematopathology teams at academic centers that see large volumes of these cases.
I think it's very important to educate community physicians and patients about a proper PTCL diagnosis, especially now that more targeted therapies are being developed and the gene expression profiling techniques will probably lead to identification of specific pathways that are amenable to therapy with specific biologic agents.
DR. FANALE: I'd like to expand upon what Andrei just said. For me the next step after confirming the pathology diagnosis is to think about two things. To think about whether or not this patient is a patient who might be eligible for an ongoing front-line trial, typically if the patient meets eligibility criteria for one of our ongoing front-line trials I would really recommend to the patient to consider being enrolled in that trial, and I also think about whether the patient, if he or she enters into remission with front-line therapy, can be considered for a consolidative autologous stem cell transplant.
I think it's very important to educate community physicians and patients about a proper PTCL diagnosis. Right now, our ongoing front-line trial is the ECHELON-2 trial which is evaluating brentuximab vedotin (BV) plus cyclophosphamide, doxorubicin, prednisone (CHP [BV-CHP]) chemotherapy compared to standard of care cyclophosphamide, doxorubicin, vincristine, prednisone (CHOP) chemotherapy, and that trial is based on the promising data that we've seen in the initial phase I trial that combined BV plus CHP chemotherapy followed by maintenance BV which demonstrated both high and durable complete remission (CR) rates.1 BV is an antibody drug conjugate that's targeted at CD30 and carries initial US Food and Drug Administration approval for patients who have relapsed or refractory ALCL which has a 100% level of CD30 positivity and then also has a National Comprehensive Cancer Network listing for treating other types of relapsed or refractory CD30 positive PTCL as well.
Also there is another upcoming front-line trial, which is to combine pralatrexate plus CHOP. If a patient isn't eligible for a clinical trial or it's just not feasible for that patient to enroll in a clinical trial, I will then look further at what would be potentially the best standard of care option for that patient.
I'll look at the patient's age and performance status and if they are generally less than age 65 or so and if otherwise well, I'll preferentially treat that patient with CHOEP which is CHOP plus etoposide. And then, for a patient who's either older than this or has multiple other comorbidities, I would treat that patient typically with CHOP alone.
DR. HORWITZ: Thank you, we'll go further into the selection of initial therapies but first circle back, I was curious as to how often your center comes to a different diagnosis than the referring center, and are there pitfalls you see that alert you to be suspicious of diagnosis.
DR. SHUSTOV: I'd say probably 10% of cases that we see at our center will be reclassified by our hematopathologists. In most cases, they do not necessarily reverse the diagnosis, but provide further clarification. It is occurring to me that in the community, pathologists are less likely to call the subtype of T-cell lymphoma and limit the report to the general description of T-cell lymphoprolipherative disorder, or state something like “consistent with T-cell lymphoma with features of AITL, or with features of anaplastic lymphoma.” I would admit though, that sometimes it's very difficult to identify the specific subtype of PTCL even in the expert hands; but I'd say these cases would constitute no more than 5% of PTCL patients.
DR. HORWITZ: And in your experience is it mostly fine tuning a T-cell lymphoma diagnosis, or do you see totally different diagnoses?
DR. MOSKOWITZ: Usually review by expert hematopathologists simply leads to fine-tuning the T-cell lymphoma diagnosis, however, I occasionally see significant changes in diagnosis. Often, alterations or clarification of a diagnosis are made possible only after we provide the pathologist with clinical history. For example, a lymph node biopsy may be interpreted as ALCL, however the knowledge that the particular patient has a history of mycosis fungoides would lead the pathologist to consider the diagnosis of large cell transformation of mycosis fungoides rather than ALCL. In such a case, molecular studies are helpful in confirming that the lymph node findings originate from the same clone as that in the original mycosis fungoides lesions, rather than representing a second primary.
DR. FANALE: Very occasionally, I've seen patients truly “with a misdiagnosis and a complete revision of diagnosis” and, usually, those pitfalls that I've seen them occasionally have been the young patients—the young patients who generally have disease in the thorax and neck, who are treated as though they have classical Hodgkin lymphoma, who have very significant progression of disease on standard of care treatment. So it's important that not all cases that have CD30 positivity are classical Hodgkin lymphoma even if the patient is young.
DR. HORWITZ: I often think the systemic T-cell lymphomas usually behave in an aggressive fashion so when the clinical picture and the diagnosis don't really fit, I think about getting a second biopsy before we finalize the diagnosis. Of course, when people are ill with obviously progressing disease, you may need to move more quickly.
I often think the systemic T-cell lymphomas usually behave in an aggressive fashion so when the clinical picture and the diagnosis don't really fit, I think about getting a second biopsy before we finalize the diagnosis. To move on, when you first see a patient, what is the decision tree in your mind in terms of picking therapy or planning therapy, or what kind of things do you consider?
DR. MOSKOWITZ: The first thing I think about when deciding upon treatment for a patient with T-cell lymphoma is whether or not I plan to use a curative approach to therapy. As was mentioned by Michelle, this decision is partly based upon the patients’ comorbidities and age. For patients who are eligible for curative therapy, our frontline approach for the most common types of T-cell lymphoma is to use CHOP with or without etoposide followed by consideration for autologous stem cell transplant in first remission. There are certainly individuals for whom such an aggressive strategy would not be appropriate due to age or co-morbidities. In these individuals, we may consider CHOP-based therapy alone or sometimes even milder approaches aimed at disease control.
DR. HORWITZ: Andrei, are you similar in the CHOP/CHOEP paradigm? And if so, what do you think of those data? How do you decide between the two and when do you do that regimen versus something different?
DR. SHUSTOV: I think I will double or triple what Michelle and Alison just said; for me, the two most important decisions that I have to make in the first encounter with patients with newly diagnosed PTCL are: 1) whether we're going to pursue curative approach strategy; and 2) whether the patient can tolerate the intensity of treatments that would provide him/her with the best chance of cure or long term remission. Patients who are elderly and have high risk disease would be very hard to cure, especially considering that the consolidative transplant might carry high rates of morbidity or mortality; more conservative strategies might be appropriate in these cases. On the other hand, younger and more fit patients might benefit more from intensified initial regimens—ie, CHOEP—followed by high-dose therapy and either autologous or allogeneic stem cell transplant (ALLO).
I usually have a long initial discussion with patients and families during which we decide on the intent of treatment and what to expect from certain regimens in terms of toxicities. I typically choose CHOEP regimen (or infusional version, etoposide, prednisone, vincristine, cyclophosphamide, doxorubicin [EPOCH]) for younger patients based on recent German data, even though this was a retrospective study and benefit of adding etoposide only approached statistical significance for the majority of PTCL subtypes.2 In older patients, I try to avoid anthracyclines, especially in the palliative intent setting, based on the retrospective analysis by the International T-Cell Lymphoma Project, and frequently use CEOP regimen (CHOEP minus anthracycline).3 I find that the majority of older patients tolerate this treatment somewhat better than anthracycline-containing combinations like CHOP.
In the very elderly and frail patients, I try to avoid combination chemotherapy all together. It is a somewhat easier decision for patients with AITL. Some of them are more indolent than other sub-types, and I would treat such patients with immunomodulatory approach, ie with a combination of prednisone and cyclosporine; then, I would consider single agent therapy with one of the recently approved agents for relapsed and refractory PTCL. I would also double what Michelle said in regard to making the best attempt to enroll patients into open clinical trials, because the current standards are not really satisfactory for many T-cell lymphoma patients.
DR. HORWITZ: It sounds like we all approach a new patient similarly. However, several of our up front trials are randomized against CHOP as opposed to CHOEP. When you're consenting a patient to those trials, how do you explain it to a patient or how important do you think the etoposide is?
DR. SHUSTOV: I share your frustration with quite a few of the current study designs for that exact reason. Some of these are confirmatory trials after conditional approvals of the new agents and are important. However, as often happens, in the confirmatory trials, we use controls that are a default from a lot of historical data, or the “common” way that the majority of patients are being treated. It is really a challenge when you consent patients to studies where a control arm is something that you think might not be quite adequate.
Having said that, the CHOEP study that was mentioned several times is a retrospective subgroup analysis and the addition of etoposide had marginal benefit that approached significance, but certainly was not a home run. I don't think we are ready to say that etoposide provides survival advantage in T-cell lymphoma patients and dismiss clinical trials in favor of just giving a patient the CHOEP, even though CHOP is admittedly not the best comparator. I discuss this controversy with patients and tell them frankly that the data that we base addition of etoposide on are not the best evidence one may have. Then, I ask patients to decide which approach sounds more reasonable to them and they make their choice.
DR. FANALE: To expand just slightly upon what Andrei said, I do emphasize to the patient and to the referring doctors that if you look at National Comprehensive Cancer Network guidelines, CHOP, CHOEP, EPOCH, all of these are potential options. So there's really not yet one trial that would say that one is clearly superior to the other at this time. I also emphasize to the patient and to the referring clinician that the only way, let's say, the patient could get the targeted agent plus the chemotherapy and have that 50% chance of, potentially, receiving that combination is really through the randomized clinical trial.
DR. HORWITZ: That was excellent, thank you. So you have alluded to this a bit. How much does subtype specific treatment come into play?
DR. MOSKOWITZ: At this point in time, outside of a clinical trial, for PTCL-NOS, AITL, and ALCL, the front-line approach would typically be CHOEP followed by autologous stem cell transplant. As Michelle mentioned, for a patient with ALCL, I would offer enrollment on the ECHELON-2 study in which patients are randomized to CHOP or BV-CHP, in order to give patients the option of potentially receiving BV.
For AITL, we are now obtaining molecular testing for certain mutations, particularly IDH2, because we currently have a study open with an IDH2 inhibitor that specifically enrolls patients with IDH2-mutant AITL. Because the testing takes some time to get back, we typically test patients’ biopsies at the time of diagnosis, so that we know if they're eligible for future clinical trials in case their disease does not respond to upfront therapy.
There are T-cell lymphoma subtypes that we treat quite differently from the entities discussed so far. For human T-lymphotropic virus-1 associated adult T-cell lymphoma, for example, the data with CHOP are really not good and we typically used EPOCH with the aim to consolidate with an allogeneic stem cell transplant.
Likewise, for hepatosplenic T-cell lymphoma, we have not observed good responses with the CHOP-based therapy and therefore we typically use platinum-based or ifosfamide-based therapy upfront with the aim to consolidate with allogeneic stem cell transplant. Extranodal NK/T-cell lymphoma is another entity for which CHOP is typically not effective and we have adopted asparaginase-based regimens, such as dexamethasone, methotrexate, ifosfamide, L-asparaginase, etoposide (SMILE) for this disease.
DR. SHUSTOV: I completely agree with Alison; for more common nodal lymphomas it is really hard at this point to base a treatment decision on histology, at least, in the front-line setting. However, some of the rare and unique subtypes, we generally treat with a completely different approach (ie, extranodal NK-cell lymphomas, enteropathy-associated T-cell lymphomas, adult T-cell leukemia/lymphomas).
DR. HORWITZ: Excellent. I’m curious, in a patient with ALCL who refused randomization of the ECHELON-2 trial, would you give them BV-CHP off study or would you discuss that as an alternative were they not to participate in the trial?
DR. FANALE: No. I don't urge treatment that's not approved for a particular line of therapy off clinical protocol with the exception that, let's say, if a patient is not a candidate for chemotherapy in second line setting for a particular lymphoma such as Hodgkin lymphoma—then, I would reference published data like Alison's data for a second line use of BV say for classical Hodgkin lymphoma and work with the insurance company to get the drug approved for that patient population, but if a patient just doesn't want to go on trial because they don't like the 50/50 chance of receiving BV-CHP compared to CHOP and they say, “I really want the 100% chance. Why don't you just contact my insurance?” I will explain to them the rationale of why the trial is being conducted including the hope that if the endpoints are met and the regimen is approved for front-line therapy then patients in the future can get BV-CHP at any oncology clinic, but I tell them for now it is a protocol based treatment option.
DR. HORWITZ: That's a very good answer. Can we now touch on the idea of transplant vs no transplant in first remission. Always, sometimes, never; how do you all think about that?
DR. SHUSTOV: I think that the idea of consolidative transplant is very heavily debated and discussed at the majority of specialized PTCL meetings. The reason for that is controversial nature of current clinical evidence. In the absence of randomized trials it is very hard to compare prospective phase II trial data to historical controls. You can interpret this in two ways; you might say, well, these are good data, outcome numbers look better than historical controls, and all patients with PTCL should have a consolidative transplant; or you can say, well, I don't have randomized data, and only a randomized study would really tell us whether post-induction consolidative transplant improves survival.
That is why it is such a controversial subject, and we agree that the phase II perspective studies of transplantation are hampered by significant bias; patients who are able and willing to travel to academic centers, were more robust and able to tolerate high dose therapy. They also have chemosensitive disease and that puts them in a completely different category than those you see in populational or retrospective studies, or other historical control trials.
Having said that, when I talk to patients, and I think that this is where I involve patients into treatment decision very heavily, I present them with the data and say that it is not wrong to do or not do the transplant in first CR. I make patients and family a big part of the decision. When they ask me what would give the patient the best chance being in remission five years from today, my answer is transplant, however, the data are not perfect, and the curative potential of autologous transplant in PTCL is not known.
DR. FANALE: So typically here, we would refer most patients on for front-line consolidative autologous stem cell transplant, I think, some exceptions are the ones that Andrei already touched on. If we're seeing a patient who has PTCL-NOS and this patient is the very rare patient who has early stage disease, with really no significant risk factors, that patient, unless there were pathologic features that showed a higher level of aggressiveness as Alison commented on, this patient might be one where we might defer doing the consolidative stem cell transplant for particularly if the patient’s disease entered into remission very quickly, but this still is controversial.
I'm not sure how you practice within each of your centers, but another patient, or where we would potentially defer, is a patient with fairly limited nasal NK/T-cell lymphoma, a patient that we’ve treated, let's say, with the dexamethasone, etoposide, ifosfamide, carboplatin (DeVIC) chemotherapy regimen plus a concomitant high-dose radiation, we typically historically here have deferred doing consolidative stem cell transplant for that patient population.
In terms of what Andrei commented on for when we might consider an ALLO and for a frontline consolidation, here we typically would not. The exception to that rule is that I've had a couple of patients here who have PTCL with a secondary hemophagocytic lymphohistiocytosis syndrome and then, for those patients because the concern is that the autologous stem cell transplant probably wouldn't be enough and for those patients given that they have such a dismal prognosis from the secondary hemophagocytic lymphohistiocytosis, we send those patients on for an ALLO.
DR. MOSKOWITZ: To expand upon early stage extranodal NK/T-cell lymphoma, I agree with Michelle that we wouldn't use an autologous stem cell transplant in first remission. Typically, for patients with localized disease, we recommend 2 cycles of SMILE followed by involved field radiation. Our patients treated with this approach have typically done quite well without needing a transplant.
DR. HORWITZ: That was great. If there are no further comments on upfront therapy, we can move over to relapse setting. When you see a patient at relapse, what are your strategies and how do you approach that patient?
DR. MOSKOWITZ: The first question in my mind when a patient is either not responding to or relapsing following front-line therapy is: what is my ultimate goal? For a patient who is fit and refractory to CHOP or has relapsed after CHOP or autologous stem cell transplant, my ultimate goal would be to try to get them to an ALLO. My choice of treatment following front-line therapy depends upon whether or not we are aiming for an ALLO as well as whether a donor has been identified for the patient.
Usually, if a patient just relapsed following front-line therapy, we are not necessarily ready to proceed quickly to ALLO. This may be because a donor has not yet been identified or because the patients’ eligibility for an ALLO is not clear. In this situation, my choice of treatment would be something that can be continued, potentially for several months, while we get things in place for the transplant. Treatment options in this situation would include the approved single agents for T-cell lymphoma, such as romidepsin, belinostat, pralatrexate, as well as BV (which would be specific for ALCL). In addition, I would consider enrollment on a promising clinical trial. Among these options, my choice of treatment typically depends upon the side effect profile and/or schedule, which is individualized for the patient. The one caveat would be that I typically will aim to use a histone deacetylase inhibitor (HDAC) inhibitor as my first choice for a patient with AITL.
Usually, if a patient just relapsed following front-line therapy, we are not necessarily ready to proceed quickly to ALLO. This may be because a donor has not yet been identified or because the patients’ eligibility for an ALLO is not clear. If my patient has a donor available and we are ready to move quickly to transplant, I would use one of the multi-agent regimens, such as ifosfamide, carboplatin, etoposide (ICE) or cisplatin, cytosine arabinoside, dexamethasone (DHAP), with the aim to try to get them into remission and relatively quickly proceed to transplant. The problem with using one of these types of regimens for everyone in the second-line setting is that the treatment cannot be continued indefinitely due to cumulative toxicity; therefore we need to know that we are ready to proceed to transplant if we are going to use one of the more intense regimens.
DR. HORWITZ: How do the others approach relapse?
DR. FANALE: Generally, a lot of similarities with what Alison discussed. Typically, it's if we're going to be sending the patient on for consideration for ALLO, I think the favor would be to try to use a regimen that has a high CR rate knowing that there is a trend toward improved progression free survival for the patient who enters into CR when compared to those who did not. I think the only slight difference might be just the timing and the selection, so, typically, here, even if the patient doesn't actually have a definite established donor quite yet we'll typically favor a combination type of a treatment approach, so whether or not that would be a platinum-based regimen such as ICE, as Alison mentioned, or gemcitabine-based chemotherapy regimen or similar.
I would generally favor one of those options, usually, over a single-agent therapeutic approach just knowing that the CR rates, generally, with exception of BV for ALCL are, generally, ranging about the 10%–15% range. And then, of course, first priority would be if there is a clinical trial available whether that's with a doublet of targeted agents or with the targeted agent plus chemotherapy in the relapse setting, we would typically favor that approach for a patient being considered for a stem cell transplant, ALLO approach.
DR. SHUSTOV: I'll consolidate what Michelle and Alison just said. I think we're on the same page. To me, the most important decision (even more important in the relapse setting) is to figure out whether we are still going to try to cure the lymphoma or we will pursue a palliative approach for which now we have several treatment options. So again, I’d have a long discussion with the patient, whether we're going to try and cure the disease, or we will pursue a palliative approach.
If this is a curative approach strategy and we're heading toward ALLO, we would start searching for the donor immediately. The choice of salvage therapy depends on duration of first remission. Primary refractory disease patients who failed CHOP, CHOEP, or EPOCH outright or within 3 months, I typically consider using the single agent approach, because even ICE and DHAP type regimens don’t work well for these patients. I put my “money” into HDAC inhibitors or antifolates, or other targeted therapy, if this is anaplastic lymphoma.
For patients who had at least 6 months remission from the initial therapy, younger patients who are still fit, I agree, I would consider standard salvage regimens like DHAP, etoposide, methylprednisolone, high-dosecytarabine, cisplatin (ESHAP) and ICE, and then, follow this with allograft.
DR. HORWITZ: You have all talked about allografting, I have two questions. What is your best guess in terms of percentage of patients you see with relapsed T-cell lymphoma who actually undergo ALLO at some point and do you ever consider autologous transplantation in those who didn't have it as part of their initial therapy?
DR. SHUSTOV: It certainly depends on the type of the academic center that patients are referred to, and we all practice at academic institutions with very strong ALLO programs. In patients who we select for a curative approach (one half to two thirds) we will at least make the best effort to take them to allogeneic grafting. Patients might fall off this track if they don't respond to salvage therapies or develop significant treatment/disease-related complications, ie fungal infections, organ toxicities, poor performance status, etc. Overall, I'd say that we successfully take about half of the patients with relapsed PTCL that are willing to pursue curative approach to allograft.
In addition, I consider autologous stem cell transplant in relapse setting for three situations. One is relapsed ALK-positive ALCL, some patients with ALK-negative ALCL who have had long initial remission and after relapse, and achieved second remission with BV or other salvage therapy. Finally, I’d consider auto-transplant in patients with AITL who achieved second CR with salvage therapy.
DR. HORWITZ: In terms of the relapse setting—some of you talked about angioimmunoblastic and HDAC inhibitors or BV for ALCL—when you have a relapse patient with PTCL-NOS, how do you pick a second line agent if you're not going directly to transplant, and are there new drugs that you're particularly excited about?
DR. FANALE: To answer your questions in terms of how I might choose a targeted agent for a patient who has PTCL-NOS and doesn't have AITL or ALCL, if the patient isn't eligible for trial or trial isn't feasible for that patient, I'll typically look at schedule, and then I'll look at side effect profile. For instance, for comparison, there are two HDAC inhibitor drugs right now approved, romidepsin and belinostat, so I think that potentially in a private practice setting, belinostat might be one when you look at the efficacy, being generally, around equivalent on where a patient might prefer that regimen because of the fact that they come in for five days in a row and then, their treatment is done for that cycle.
At a larger referral center to which patients are often traveling back and forth, it can be quite difficult for a patient to stay in town to do five days of treatment in a row. Often, these patients will prefer the romidepsin schedule because of the fact that they're coming back in once per week for three weeks in a row per cycle. The other way that I'll look at it is side effect profile, so, let's say, pralatrexate compared to the HDAC inhibitors, I will usually favor the HDAC inhibitors because of the potential side effect of mucositis with pralatrexate, although I have been able to manage through the mucositis by giving the patients more of a cutaneous T-cell lymphoma dosing format.
There had been a high level of interest in new emerging agents such as the aurora A-kinase inhibitors, alisertib, and that trial is now complete and the data are to be presented at The American Society of Hematology’s Annual Meeting this year.4 Other new therapies that are emerging are the PI3K inhibitors such as duvelisib and others. One advantage for many of the drugs in current trials is that they are oral agents.
My interest also lies in the combination approach, such as two or three targeted agent combinations, with the goal that if you see favorable CR rates and progression-free survival rates in the relapsed setting that you can then potentially move these treatments to the front-line setting to potentially move beyond the CHOP-based platform of therapies.
DR. SHUSTOV: For patients outside anaplastic lymphoma where, obviously, most of us would likely use BV, given the response rate to this agent, the decision is really based on convenience of the schedule, toxicity profile, and—sometimes—patient’s preference. So, toxicity and quality of life become kind of more decision-driving factors than disease biology. I’d have a discussion with patients about all three drugs, how they are administered, and what toxicities can be expected. Four-hour infusion of romidepsin might not be acceptable for some patients who have to travel long distances to treatment centers. They may prefer more rapid infusion (they would favor a trial of pralatrexate; or, as Alison mentioned, patient prefers a five-day/daily versus weekly administration.
It's kind of a coin toss decision outside clinical trials. Certainly, participation in studies in relapse setting is the high priority. We are all really looking forward to developing doublet combinations of novel agents, ie studies like the one Michelle is running at MD Anderson with a combination of alisertib and romidepsin.
DR. MOSKOWITZ: I'll echo what both Andrei and Michelle said that our choice of treatment is individualized and depends upon side-effect profile and the schedule. With regard to novel agents, I am also very excited about the studies that will be opening with the PI-3 kinase inhibitor, IPI-145. There have been promising results seen with single-agent IPI-1455 and we will be opening a study evaluating it in combination with bortezomib or romidepsin.
In addition, I mentioned earlier the IDH2 inhibitor, which we're studying right now in patients with AITL that is characterized by the presence of the IDH2 mutation. It would be exciting to see if targeting a specific mutation in this disease translates into responses.
DR. HORWITZ: Great, I think that was fantastic. I would like to thank Drs. Moskowitz, Shustov, and Fanale for a very thorough impractical discussion on how they approach and manage patients with T-cell lymphoma. I’m impressed that there is significant consistency among these experts in terms of how they manage these uncommon and often challenging lymphomas in terms of upfront combination chemotherapy approaches, combination considerations for the use of transplantation, as well as their enthusiasm for and dedication to incorporating clinical trials as part of everyday management.
References
1. Fanale MA, Horwitz SM, Forero-Torres A, et al. Brentuximab vedotin in the front-line treatment of patients with CD30+ peripheral T-cell lymphomas: results of a phase I study. J Clin Oncol. 2014;32(28):3137–3143.
2. Schmitz N, Trümper L, Ziepert M, et al. Treatment and prognosis of mature T-cell and NK-cell lymphoma: an analysis of patients with T-cell lymphoma treated in studies of the German High-Grade Non-Hodgkin Lymphoma Study Group. Blood. 2010;116(18):3418–3425. doi: 10.1182/blood-2010-02-270785. Epub 2010 Jul 21.
3. Vose J, Armitage J, Weisenburger D, for the International T-Cell Lymphoma Project. J Clin Oncol. 2008;26(25):4124–4130. doi: 10.1200/JCO.2008.16.4558. Epub 2008 Jul 14.
4. O’Connor OA, Ozcan M, Jacobsen ED, et al. First multicenter, randomized phase 3 study in patients with relapsed/refractory peripheral T-cell lymphoma: alisertib versus investigator’s choice. Paper to be presented at: American Society of Hematology 57th Annual Meeting & Exposition; December 5–8, 2015; Orlando, FL.
5. Horwitz SM, Porcu P, Flinn I, et al. Duvelisib (IPI-145), a phosphoinositide-3-kinsae-γδ inhibitor, shows activity in patients with relapsed/refractory T-cell lymphoma. Paper presented at: American Society of Hematology 56th Annual Meeting & Exposition; December 6–9, 2014; San Francisco, CA.
Moderator: Steven M. Horwitz, MD1
Discussants: Alison Moskowitz, MD1; Michelle Fanale, MD2; Andrei Shustov, MD3
From Memorial Sloan Kettering Cancer Center, New York, NY1; MD Anderson Cancer Center, Houston, TX2; University of Washington Medical Center, Seattle, WA3
Address for correspondence: Steven M. Horwitz, MD, Memorial Sloan Kettering Cancer Center, 1275 York Avenue, New York, NY 10065
E-mail: [email protected]
DR. HORWITZ: My name is Dr. Steven Horwitz from Memorial Sloan-Kettering Cancer Center. I’m joined today by Drs. Alison Moskowitz, Memorial Sloan-Kettering, Michelle Fanale of MD Anderson, and Andrei Shustov, University of Washington in Seattle. Thank you all for joining us for this conversation on T-cell lymphoma. My colleagues are all well known as experts in T-cell lymphomas. Those of you who treat these diseases recognize the systemic T-cell lymphomas as one of our greater challenges in hematologic malignancies in terms of the treatment options for patients and the frequent lack of definitive data to guide our decisions.
I thought what we would do today is have a very practical discussion about the way we think about these diseases, the decisions we make, and the way we make those decisions. I'll start off by asking when you get a referral of a patient with a new diagnosis of peripheral T-cell lymphoma (PTCL), what are some of the basic things that you first think about in terms of approaching a new patient?
DR. SHUSTOV: I think one of the biggest challenges in T-cell lymphomas still remains making the proper diagnosis. In general, pathologists in the United States have a pretty good idea when they see T-cell lymphomas, however, subclassification remains a challenge even for expert hematopathologists due to frequent histologic overlap between the subtypes of PTCL, and even with non-malignant autoimmune disorders. I frequently see patients who are diagnosed with or misdiagnosed with a different subtype of T-cell lymphoma. The most challenging is differentiation between angioimmunoblastic T-cell lymphoma (AITL), anaplastic large cell T-cell lymphoma (ALCL), and PTCL not otherwise specified (PTCL-NOS), especially when the latter patients have high expression of CD30 and/or bear features resembling AITL.
Sometimes they are a slam-dunk diagnosis, but frequently our hematopathologists reverse the diagnosis after doing additional studies on the biopsy material. The most recent case I've seen in my clinic for consultation was a patient with a diagnosis of extranodal NK-cell lymphoma that was reclassified as a gamma-delta T-cell lymphoma after additional work-up. I truly believe that it is advisable that majority, if not all PTCL cases are reviewed by expert hematopathology teams at academic centers that see large volumes of these cases.
I think it's very important to educate community physicians and patients about a proper PTCL diagnosis, especially now that more targeted therapies are being developed and the gene expression profiling techniques will probably lead to identification of specific pathways that are amenable to therapy with specific biologic agents.
DR. FANALE: I'd like to expand upon what Andrei just said. For me the next step after confirming the pathology diagnosis is to think about two things. To think about whether or not this patient is a patient who might be eligible for an ongoing front-line trial, typically if the patient meets eligibility criteria for one of our ongoing front-line trials I would really recommend to the patient to consider being enrolled in that trial, and I also think about whether the patient, if he or she enters into remission with front-line therapy, can be considered for a consolidative autologous stem cell transplant.
I think it's very important to educate community physicians and patients about a proper PTCL diagnosis. Right now, our ongoing front-line trial is the ECHELON-2 trial which is evaluating brentuximab vedotin (BV) plus cyclophosphamide, doxorubicin, prednisone (CHP [BV-CHP]) chemotherapy compared to standard of care cyclophosphamide, doxorubicin, vincristine, prednisone (CHOP) chemotherapy, and that trial is based on the promising data that we've seen in the initial phase I trial that combined BV plus CHP chemotherapy followed by maintenance BV which demonstrated both high and durable complete remission (CR) rates.1 BV is an antibody drug conjugate that's targeted at CD30 and carries initial US Food and Drug Administration approval for patients who have relapsed or refractory ALCL which has a 100% level of CD30 positivity and then also has a National Comprehensive Cancer Network listing for treating other types of relapsed or refractory CD30 positive PTCL as well.
Also there is another upcoming front-line trial, which is to combine pralatrexate plus CHOP. If a patient isn't eligible for a clinical trial or it's just not feasible for that patient to enroll in a clinical trial, I will then look further at what would be potentially the best standard of care option for that patient.
I'll look at the patient's age and performance status and if they are generally less than age 65 or so and if otherwise well, I'll preferentially treat that patient with CHOEP which is CHOP plus etoposide. And then, for a patient who's either older than this or has multiple other comorbidities, I would treat that patient typically with CHOP alone.
DR. HORWITZ: Thank you, we'll go further into the selection of initial therapies but first circle back, I was curious as to how often your center comes to a different diagnosis than the referring center, and are there pitfalls you see that alert you to be suspicious of diagnosis.
DR. SHUSTOV: I'd say probably 10% of cases that we see at our center will be reclassified by our hematopathologists. In most cases, they do not necessarily reverse the diagnosis, but provide further clarification. It is occurring to me that in the community, pathologists are less likely to call the subtype of T-cell lymphoma and limit the report to the general description of T-cell lymphoprolipherative disorder, or state something like “consistent with T-cell lymphoma with features of AITL, or with features of anaplastic lymphoma.” I would admit though, that sometimes it's very difficult to identify the specific subtype of PTCL even in the expert hands; but I'd say these cases would constitute no more than 5% of PTCL patients.
DR. HORWITZ: And in your experience is it mostly fine tuning a T-cell lymphoma diagnosis, or do you see totally different diagnoses?
DR. MOSKOWITZ: Usually review by expert hematopathologists simply leads to fine-tuning the T-cell lymphoma diagnosis, however, I occasionally see significant changes in diagnosis. Often, alterations or clarification of a diagnosis are made possible only after we provide the pathologist with clinical history. For example, a lymph node biopsy may be interpreted as ALCL, however the knowledge that the particular patient has a history of mycosis fungoides would lead the pathologist to consider the diagnosis of large cell transformation of mycosis fungoides rather than ALCL. In such a case, molecular studies are helpful in confirming that the lymph node findings originate from the same clone as that in the original mycosis fungoides lesions, rather than representing a second primary.
DR. FANALE: Very occasionally, I've seen patients truly “with a misdiagnosis and a complete revision of diagnosis” and, usually, those pitfalls that I've seen them occasionally have been the young patients—the young patients who generally have disease in the thorax and neck, who are treated as though they have classical Hodgkin lymphoma, who have very significant progression of disease on standard of care treatment. So it's important that not all cases that have CD30 positivity are classical Hodgkin lymphoma even if the patient is young.
DR. HORWITZ: I often think the systemic T-cell lymphomas usually behave in an aggressive fashion so when the clinical picture and the diagnosis don't really fit, I think about getting a second biopsy before we finalize the diagnosis. Of course, when people are ill with obviously progressing disease, you may need to move more quickly.
I often think the systemic T-cell lymphomas usually behave in an aggressive fashion so when the clinical picture and the diagnosis don't really fit, I think about getting a second biopsy before we finalize the diagnosis. To move on, when you first see a patient, what is the decision tree in your mind in terms of picking therapy or planning therapy, or what kind of things do you consider?
DR. MOSKOWITZ: The first thing I think about when deciding upon treatment for a patient with T-cell lymphoma is whether or not I plan to use a curative approach to therapy. As was mentioned by Michelle, this decision is partly based upon the patients’ comorbidities and age. For patients who are eligible for curative therapy, our frontline approach for the most common types of T-cell lymphoma is to use CHOP with or without etoposide followed by consideration for autologous stem cell transplant in first remission. There are certainly individuals for whom such an aggressive strategy would not be appropriate due to age or co-morbidities. In these individuals, we may consider CHOP-based therapy alone or sometimes even milder approaches aimed at disease control.
DR. HORWITZ: Andrei, are you similar in the CHOP/CHOEP paradigm? And if so, what do you think of those data? How do you decide between the two and when do you do that regimen versus something different?
DR. SHUSTOV: I think I will double or triple what Michelle and Alison just said; for me, the two most important decisions that I have to make in the first encounter with patients with newly diagnosed PTCL are: 1) whether we're going to pursue curative approach strategy; and 2) whether the patient can tolerate the intensity of treatments that would provide him/her with the best chance of cure or long term remission. Patients who are elderly and have high risk disease would be very hard to cure, especially considering that the consolidative transplant might carry high rates of morbidity or mortality; more conservative strategies might be appropriate in these cases. On the other hand, younger and more fit patients might benefit more from intensified initial regimens—ie, CHOEP—followed by high-dose therapy and either autologous or allogeneic stem cell transplant (ALLO).
I usually have a long initial discussion with patients and families during which we decide on the intent of treatment and what to expect from certain regimens in terms of toxicities. I typically choose CHOEP regimen (or infusional version, etoposide, prednisone, vincristine, cyclophosphamide, doxorubicin [EPOCH]) for younger patients based on recent German data, even though this was a retrospective study and benefit of adding etoposide only approached statistical significance for the majority of PTCL subtypes.2 In older patients, I try to avoid anthracyclines, especially in the palliative intent setting, based on the retrospective analysis by the International T-Cell Lymphoma Project, and frequently use CEOP regimen (CHOEP minus anthracycline).3 I find that the majority of older patients tolerate this treatment somewhat better than anthracycline-containing combinations like CHOP.
In the very elderly and frail patients, I try to avoid combination chemotherapy all together. It is a somewhat easier decision for patients with AITL. Some of them are more indolent than other sub-types, and I would treat such patients with immunomodulatory approach, ie with a combination of prednisone and cyclosporine; then, I would consider single agent therapy with one of the recently approved agents for relapsed and refractory PTCL. I would also double what Michelle said in regard to making the best attempt to enroll patients into open clinical trials, because the current standards are not really satisfactory for many T-cell lymphoma patients.
DR. HORWITZ: It sounds like we all approach a new patient similarly. However, several of our up front trials are randomized against CHOP as opposed to CHOEP. When you're consenting a patient to those trials, how do you explain it to a patient or how important do you think the etoposide is?
DR. SHUSTOV: I share your frustration with quite a few of the current study designs for that exact reason. Some of these are confirmatory trials after conditional approvals of the new agents and are important. However, as often happens, in the confirmatory trials, we use controls that are a default from a lot of historical data, or the “common” way that the majority of patients are being treated. It is really a challenge when you consent patients to studies where a control arm is something that you think might not be quite adequate.
Having said that, the CHOEP study that was mentioned several times is a retrospective subgroup analysis and the addition of etoposide had marginal benefit that approached significance, but certainly was not a home run. I don't think we are ready to say that etoposide provides survival advantage in T-cell lymphoma patients and dismiss clinical trials in favor of just giving a patient the CHOEP, even though CHOP is admittedly not the best comparator. I discuss this controversy with patients and tell them frankly that the data that we base addition of etoposide on are not the best evidence one may have. Then, I ask patients to decide which approach sounds more reasonable to them and they make their choice.
DR. FANALE: To expand just slightly upon what Andrei said, I do emphasize to the patient and to the referring doctors that if you look at National Comprehensive Cancer Network guidelines, CHOP, CHOEP, EPOCH, all of these are potential options. So there's really not yet one trial that would say that one is clearly superior to the other at this time. I also emphasize to the patient and to the referring clinician that the only way, let's say, the patient could get the targeted agent plus the chemotherapy and have that 50% chance of, potentially, receiving that combination is really through the randomized clinical trial.
DR. HORWITZ: That was excellent, thank you. So you have alluded to this a bit. How much does subtype specific treatment come into play?
DR. MOSKOWITZ: At this point in time, outside of a clinical trial, for PTCL-NOS, AITL, and ALCL, the front-line approach would typically be CHOEP followed by autologous stem cell transplant. As Michelle mentioned, for a patient with ALCL, I would offer enrollment on the ECHELON-2 study in which patients are randomized to CHOP or BV-CHP, in order to give patients the option of potentially receiving BV.
For AITL, we are now obtaining molecular testing for certain mutations, particularly IDH2, because we currently have a study open with an IDH2 inhibitor that specifically enrolls patients with IDH2-mutant AITL. Because the testing takes some time to get back, we typically test patients’ biopsies at the time of diagnosis, so that we know if they're eligible for future clinical trials in case their disease does not respond to upfront therapy.
There are T-cell lymphoma subtypes that we treat quite differently from the entities discussed so far. For human T-lymphotropic virus-1 associated adult T-cell lymphoma, for example, the data with CHOP are really not good and we typically used EPOCH with the aim to consolidate with an allogeneic stem cell transplant.
Likewise, for hepatosplenic T-cell lymphoma, we have not observed good responses with the CHOP-based therapy and therefore we typically use platinum-based or ifosfamide-based therapy upfront with the aim to consolidate with allogeneic stem cell transplant. Extranodal NK/T-cell lymphoma is another entity for which CHOP is typically not effective and we have adopted asparaginase-based regimens, such as dexamethasone, methotrexate, ifosfamide, L-asparaginase, etoposide (SMILE) for this disease.
DR. SHUSTOV: I completely agree with Alison; for more common nodal lymphomas it is really hard at this point to base a treatment decision on histology, at least, in the front-line setting. However, some of the rare and unique subtypes, we generally treat with a completely different approach (ie, extranodal NK-cell lymphomas, enteropathy-associated T-cell lymphomas, adult T-cell leukemia/lymphomas).
DR. HORWITZ: Excellent. I’m curious, in a patient with ALCL who refused randomization of the ECHELON-2 trial, would you give them BV-CHP off study or would you discuss that as an alternative were they not to participate in the trial?
DR. FANALE: No. I don't urge treatment that's not approved for a particular line of therapy off clinical protocol with the exception that, let's say, if a patient is not a candidate for chemotherapy in second line setting for a particular lymphoma such as Hodgkin lymphoma—then, I would reference published data like Alison's data for a second line use of BV say for classical Hodgkin lymphoma and work with the insurance company to get the drug approved for that patient population, but if a patient just doesn't want to go on trial because they don't like the 50/50 chance of receiving BV-CHP compared to CHOP and they say, “I really want the 100% chance. Why don't you just contact my insurance?” I will explain to them the rationale of why the trial is being conducted including the hope that if the endpoints are met and the regimen is approved for front-line therapy then patients in the future can get BV-CHP at any oncology clinic, but I tell them for now it is a protocol based treatment option.
DR. HORWITZ: That's a very good answer. Can we now touch on the idea of transplant vs no transplant in first remission. Always, sometimes, never; how do you all think about that?
DR. SHUSTOV: I think that the idea of consolidative transplant is very heavily debated and discussed at the majority of specialized PTCL meetings. The reason for that is controversial nature of current clinical evidence. In the absence of randomized trials it is very hard to compare prospective phase II trial data to historical controls. You can interpret this in two ways; you might say, well, these are good data, outcome numbers look better than historical controls, and all patients with PTCL should have a consolidative transplant; or you can say, well, I don't have randomized data, and only a randomized study would really tell us whether post-induction consolidative transplant improves survival.
That is why it is such a controversial subject, and we agree that the phase II perspective studies of transplantation are hampered by significant bias; patients who are able and willing to travel to academic centers, were more robust and able to tolerate high dose therapy. They also have chemosensitive disease and that puts them in a completely different category than those you see in populational or retrospective studies, or other historical control trials.
Having said that, when I talk to patients, and I think that this is where I involve patients into treatment decision very heavily, I present them with the data and say that it is not wrong to do or not do the transplant in first CR. I make patients and family a big part of the decision. When they ask me what would give the patient the best chance being in remission five years from today, my answer is transplant, however, the data are not perfect, and the curative potential of autologous transplant in PTCL is not known.
DR. FANALE: So typically here, we would refer most patients on for front-line consolidative autologous stem cell transplant, I think, some exceptions are the ones that Andrei already touched on. If we're seeing a patient who has PTCL-NOS and this patient is the very rare patient who has early stage disease, with really no significant risk factors, that patient, unless there were pathologic features that showed a higher level of aggressiveness as Alison commented on, this patient might be one where we might defer doing the consolidative stem cell transplant for particularly if the patient’s disease entered into remission very quickly, but this still is controversial.
I'm not sure how you practice within each of your centers, but another patient, or where we would potentially defer, is a patient with fairly limited nasal NK/T-cell lymphoma, a patient that we’ve treated, let's say, with the dexamethasone, etoposide, ifosfamide, carboplatin (DeVIC) chemotherapy regimen plus a concomitant high-dose radiation, we typically historically here have deferred doing consolidative stem cell transplant for that patient population.
In terms of what Andrei commented on for when we might consider an ALLO and for a frontline consolidation, here we typically would not. The exception to that rule is that I've had a couple of patients here who have PTCL with a secondary hemophagocytic lymphohistiocytosis syndrome and then, for those patients because the concern is that the autologous stem cell transplant probably wouldn't be enough and for those patients given that they have such a dismal prognosis from the secondary hemophagocytic lymphohistiocytosis, we send those patients on for an ALLO.
DR. MOSKOWITZ: To expand upon early stage extranodal NK/T-cell lymphoma, I agree with Michelle that we wouldn't use an autologous stem cell transplant in first remission. Typically, for patients with localized disease, we recommend 2 cycles of SMILE followed by involved field radiation. Our patients treated with this approach have typically done quite well without needing a transplant.
DR. HORWITZ: That was great. If there are no further comments on upfront therapy, we can move over to relapse setting. When you see a patient at relapse, what are your strategies and how do you approach that patient?
DR. MOSKOWITZ: The first question in my mind when a patient is either not responding to or relapsing following front-line therapy is: what is my ultimate goal? For a patient who is fit and refractory to CHOP or has relapsed after CHOP or autologous stem cell transplant, my ultimate goal would be to try to get them to an ALLO. My choice of treatment following front-line therapy depends upon whether or not we are aiming for an ALLO as well as whether a donor has been identified for the patient.
Usually, if a patient just relapsed following front-line therapy, we are not necessarily ready to proceed quickly to ALLO. This may be because a donor has not yet been identified or because the patients’ eligibility for an ALLO is not clear. In this situation, my choice of treatment would be something that can be continued, potentially for several months, while we get things in place for the transplant. Treatment options in this situation would include the approved single agents for T-cell lymphoma, such as romidepsin, belinostat, pralatrexate, as well as BV (which would be specific for ALCL). In addition, I would consider enrollment on a promising clinical trial. Among these options, my choice of treatment typically depends upon the side effect profile and/or schedule, which is individualized for the patient. The one caveat would be that I typically will aim to use a histone deacetylase inhibitor (HDAC) inhibitor as my first choice for a patient with AITL.
Usually, if a patient just relapsed following front-line therapy, we are not necessarily ready to proceed quickly to ALLO. This may be because a donor has not yet been identified or because the patients’ eligibility for an ALLO is not clear. If my patient has a donor available and we are ready to move quickly to transplant, I would use one of the multi-agent regimens, such as ifosfamide, carboplatin, etoposide (ICE) or cisplatin, cytosine arabinoside, dexamethasone (DHAP), with the aim to try to get them into remission and relatively quickly proceed to transplant. The problem with using one of these types of regimens for everyone in the second-line setting is that the treatment cannot be continued indefinitely due to cumulative toxicity; therefore we need to know that we are ready to proceed to transplant if we are going to use one of the more intense regimens.
DR. HORWITZ: How do the others approach relapse?
DR. FANALE: Generally, a lot of similarities with what Alison discussed. Typically, it's if we're going to be sending the patient on for consideration for ALLO, I think the favor would be to try to use a regimen that has a high CR rate knowing that there is a trend toward improved progression free survival for the patient who enters into CR when compared to those who did not. I think the only slight difference might be just the timing and the selection, so, typically, here, even if the patient doesn't actually have a definite established donor quite yet we'll typically favor a combination type of a treatment approach, so whether or not that would be a platinum-based regimen such as ICE, as Alison mentioned, or gemcitabine-based chemotherapy regimen or similar.
I would generally favor one of those options, usually, over a single-agent therapeutic approach just knowing that the CR rates, generally, with exception of BV for ALCL are, generally, ranging about the 10%–15% range. And then, of course, first priority would be if there is a clinical trial available whether that's with a doublet of targeted agents or with the targeted agent plus chemotherapy in the relapse setting, we would typically favor that approach for a patient being considered for a stem cell transplant, ALLO approach.
DR. SHUSTOV: I'll consolidate what Michelle and Alison just said. I think we're on the same page. To me, the most important decision (even more important in the relapse setting) is to figure out whether we are still going to try to cure the lymphoma or we will pursue a palliative approach for which now we have several treatment options. So again, I’d have a long discussion with the patient, whether we're going to try and cure the disease, or we will pursue a palliative approach.
If this is a curative approach strategy and we're heading toward ALLO, we would start searching for the donor immediately. The choice of salvage therapy depends on duration of first remission. Primary refractory disease patients who failed CHOP, CHOEP, or EPOCH outright or within 3 months, I typically consider using the single agent approach, because even ICE and DHAP type regimens don’t work well for these patients. I put my “money” into HDAC inhibitors or antifolates, or other targeted therapy, if this is anaplastic lymphoma.
For patients who had at least 6 months remission from the initial therapy, younger patients who are still fit, I agree, I would consider standard salvage regimens like DHAP, etoposide, methylprednisolone, high-dosecytarabine, cisplatin (ESHAP) and ICE, and then, follow this with allograft.
DR. HORWITZ: You have all talked about allografting, I have two questions. What is your best guess in terms of percentage of patients you see with relapsed T-cell lymphoma who actually undergo ALLO at some point and do you ever consider autologous transplantation in those who didn't have it as part of their initial therapy?
DR. SHUSTOV: It certainly depends on the type of the academic center that patients are referred to, and we all practice at academic institutions with very strong ALLO programs. In patients who we select for a curative approach (one half to two thirds) we will at least make the best effort to take them to allogeneic grafting. Patients might fall off this track if they don't respond to salvage therapies or develop significant treatment/disease-related complications, ie fungal infections, organ toxicities, poor performance status, etc. Overall, I'd say that we successfully take about half of the patients with relapsed PTCL that are willing to pursue curative approach to allograft.
In addition, I consider autologous stem cell transplant in relapse setting for three situations. One is relapsed ALK-positive ALCL, some patients with ALK-negative ALCL who have had long initial remission and after relapse, and achieved second remission with BV or other salvage therapy. Finally, I’d consider auto-transplant in patients with AITL who achieved second CR with salvage therapy.
DR. HORWITZ: In terms of the relapse setting—some of you talked about angioimmunoblastic and HDAC inhibitors or BV for ALCL—when you have a relapse patient with PTCL-NOS, how do you pick a second line agent if you're not going directly to transplant, and are there new drugs that you're particularly excited about?
DR. FANALE: To answer your questions in terms of how I might choose a targeted agent for a patient who has PTCL-NOS and doesn't have AITL or ALCL, if the patient isn't eligible for trial or trial isn't feasible for that patient, I'll typically look at schedule, and then I'll look at side effect profile. For instance, for comparison, there are two HDAC inhibitor drugs right now approved, romidepsin and belinostat, so I think that potentially in a private practice setting, belinostat might be one when you look at the efficacy, being generally, around equivalent on where a patient might prefer that regimen because of the fact that they come in for five days in a row and then, their treatment is done for that cycle.
At a larger referral center to which patients are often traveling back and forth, it can be quite difficult for a patient to stay in town to do five days of treatment in a row. Often, these patients will prefer the romidepsin schedule because of the fact that they're coming back in once per week for three weeks in a row per cycle. The other way that I'll look at it is side effect profile, so, let's say, pralatrexate compared to the HDAC inhibitors, I will usually favor the HDAC inhibitors because of the potential side effect of mucositis with pralatrexate, although I have been able to manage through the mucositis by giving the patients more of a cutaneous T-cell lymphoma dosing format.
There had been a high level of interest in new emerging agents such as the aurora A-kinase inhibitors, alisertib, and that trial is now complete and the data are to be presented at The American Society of Hematology’s Annual Meeting this year.4 Other new therapies that are emerging are the PI3K inhibitors such as duvelisib and others. One advantage for many of the drugs in current trials is that they are oral agents.
My interest also lies in the combination approach, such as two or three targeted agent combinations, with the goal that if you see favorable CR rates and progression-free survival rates in the relapsed setting that you can then potentially move these treatments to the front-line setting to potentially move beyond the CHOP-based platform of therapies.
DR. SHUSTOV: For patients outside anaplastic lymphoma where, obviously, most of us would likely use BV, given the response rate to this agent, the decision is really based on convenience of the schedule, toxicity profile, and—sometimes—patient’s preference. So, toxicity and quality of life become kind of more decision-driving factors than disease biology. I’d have a discussion with patients about all three drugs, how they are administered, and what toxicities can be expected. Four-hour infusion of romidepsin might not be acceptable for some patients who have to travel long distances to treatment centers. They may prefer more rapid infusion (they would favor a trial of pralatrexate; or, as Alison mentioned, patient prefers a five-day/daily versus weekly administration.
It's kind of a coin toss decision outside clinical trials. Certainly, participation in studies in relapse setting is the high priority. We are all really looking forward to developing doublet combinations of novel agents, ie studies like the one Michelle is running at MD Anderson with a combination of alisertib and romidepsin.
DR. MOSKOWITZ: I'll echo what both Andrei and Michelle said that our choice of treatment is individualized and depends upon side-effect profile and the schedule. With regard to novel agents, I am also very excited about the studies that will be opening with the PI-3 kinase inhibitor, IPI-145. There have been promising results seen with single-agent IPI-1455 and we will be opening a study evaluating it in combination with bortezomib or romidepsin.
In addition, I mentioned earlier the IDH2 inhibitor, which we're studying right now in patients with AITL that is characterized by the presence of the IDH2 mutation. It would be exciting to see if targeting a specific mutation in this disease translates into responses.
DR. HORWITZ: Great, I think that was fantastic. I would like to thank Drs. Moskowitz, Shustov, and Fanale for a very thorough impractical discussion on how they approach and manage patients with T-cell lymphoma. I’m impressed that there is significant consistency among these experts in terms of how they manage these uncommon and often challenging lymphomas in terms of upfront combination chemotherapy approaches, combination considerations for the use of transplantation, as well as their enthusiasm for and dedication to incorporating clinical trials as part of everyday management.
References
1. Fanale MA, Horwitz SM, Forero-Torres A, et al. Brentuximab vedotin in the front-line treatment of patients with CD30+ peripheral T-cell lymphomas: results of a phase I study. J Clin Oncol. 2014;32(28):3137–3143.
2. Schmitz N, Trümper L, Ziepert M, et al. Treatment and prognosis of mature T-cell and NK-cell lymphoma: an analysis of patients with T-cell lymphoma treated in studies of the German High-Grade Non-Hodgkin Lymphoma Study Group. Blood. 2010;116(18):3418–3425. doi: 10.1182/blood-2010-02-270785. Epub 2010 Jul 21.
3. Vose J, Armitage J, Weisenburger D, for the International T-Cell Lymphoma Project. J Clin Oncol. 2008;26(25):4124–4130. doi: 10.1200/JCO.2008.16.4558. Epub 2008 Jul 14.
4. O’Connor OA, Ozcan M, Jacobsen ED, et al. First multicenter, randomized phase 3 study in patients with relapsed/refractory peripheral T-cell lymphoma: alisertib versus investigator’s choice. Paper to be presented at: American Society of Hematology 57th Annual Meeting & Exposition; December 5–8, 2015; Orlando, FL.
5. Horwitz SM, Porcu P, Flinn I, et al. Duvelisib (IPI-145), a phosphoinositide-3-kinsae-γδ inhibitor, shows activity in patients with relapsed/refractory T-cell lymphoma. Paper presented at: American Society of Hematology 56th Annual Meeting & Exposition; December 6–9, 2014; San Francisco, CA.
Moderator: Steven M. Horwitz, MD1
Discussants: Alison Moskowitz, MD1; Michelle Fanale, MD2; Andrei Shustov, MD3
From Memorial Sloan Kettering Cancer Center, New York, NY1; MD Anderson Cancer Center, Houston, TX2; University of Washington Medical Center, Seattle, WA3
Address for correspondence: Steven M. Horwitz, MD, Memorial Sloan Kettering Cancer Center, 1275 York Avenue, New York, NY 10065
E-mail: [email protected]
DR. HORWITZ: My name is Dr. Steven Horwitz from Memorial Sloan-Kettering Cancer Center. I’m joined today by Drs. Alison Moskowitz, Memorial Sloan-Kettering, Michelle Fanale of MD Anderson, and Andrei Shustov, University of Washington in Seattle. Thank you all for joining us for this conversation on T-cell lymphoma. My colleagues are all well known as experts in T-cell lymphomas. Those of you who treat these diseases recognize the systemic T-cell lymphomas as one of our greater challenges in hematologic malignancies in terms of the treatment options for patients and the frequent lack of definitive data to guide our decisions.
I thought what we would do today is have a very practical discussion about the way we think about these diseases, the decisions we make, and the way we make those decisions. I'll start off by asking when you get a referral of a patient with a new diagnosis of peripheral T-cell lymphoma (PTCL), what are some of the basic things that you first think about in terms of approaching a new patient?
DR. SHUSTOV: I think one of the biggest challenges in T-cell lymphomas still remains making the proper diagnosis. In general, pathologists in the United States have a pretty good idea when they see T-cell lymphomas, however, subclassification remains a challenge even for expert hematopathologists due to frequent histologic overlap between the subtypes of PTCL, and even with non-malignant autoimmune disorders. I frequently see patients who are diagnosed with or misdiagnosed with a different subtype of T-cell lymphoma. The most challenging is differentiation between angioimmunoblastic T-cell lymphoma (AITL), anaplastic large cell T-cell lymphoma (ALCL), and PTCL not otherwise specified (PTCL-NOS), especially when the latter patients have high expression of CD30 and/or bear features resembling AITL.
Sometimes they are a slam-dunk diagnosis, but frequently our hematopathologists reverse the diagnosis after doing additional studies on the biopsy material. The most recent case I've seen in my clinic for consultation was a patient with a diagnosis of extranodal NK-cell lymphoma that was reclassified as a gamma-delta T-cell lymphoma after additional work-up. I truly believe that it is advisable that majority, if not all PTCL cases are reviewed by expert hematopathology teams at academic centers that see large volumes of these cases.
I think it's very important to educate community physicians and patients about a proper PTCL diagnosis, especially now that more targeted therapies are being developed and the gene expression profiling techniques will probably lead to identification of specific pathways that are amenable to therapy with specific biologic agents.
DR. FANALE: I'd like to expand upon what Andrei just said. For me the next step after confirming the pathology diagnosis is to think about two things. To think about whether or not this patient is a patient who might be eligible for an ongoing front-line trial, typically if the patient meets eligibility criteria for one of our ongoing front-line trials I would really recommend to the patient to consider being enrolled in that trial, and I also think about whether the patient, if he or she enters into remission with front-line therapy, can be considered for a consolidative autologous stem cell transplant.
I think it's very important to educate community physicians and patients about a proper PTCL diagnosis. Right now, our ongoing front-line trial is the ECHELON-2 trial which is evaluating brentuximab vedotin (BV) plus cyclophosphamide, doxorubicin, prednisone (CHP [BV-CHP]) chemotherapy compared to standard of care cyclophosphamide, doxorubicin, vincristine, prednisone (CHOP) chemotherapy, and that trial is based on the promising data that we've seen in the initial phase I trial that combined BV plus CHP chemotherapy followed by maintenance BV which demonstrated both high and durable complete remission (CR) rates.1 BV is an antibody drug conjugate that's targeted at CD30 and carries initial US Food and Drug Administration approval for patients who have relapsed or refractory ALCL which has a 100% level of CD30 positivity and then also has a National Comprehensive Cancer Network listing for treating other types of relapsed or refractory CD30 positive PTCL as well.
Also there is another upcoming front-line trial, which is to combine pralatrexate plus CHOP. If a patient isn't eligible for a clinical trial or it's just not feasible for that patient to enroll in a clinical trial, I will then look further at what would be potentially the best standard of care option for that patient.
I'll look at the patient's age and performance status and if they are generally less than age 65 or so and if otherwise well, I'll preferentially treat that patient with CHOEP which is CHOP plus etoposide. And then, for a patient who's either older than this or has multiple other comorbidities, I would treat that patient typically with CHOP alone.
DR. HORWITZ: Thank you, we'll go further into the selection of initial therapies but first circle back, I was curious as to how often your center comes to a different diagnosis than the referring center, and are there pitfalls you see that alert you to be suspicious of diagnosis.
DR. SHUSTOV: I'd say probably 10% of cases that we see at our center will be reclassified by our hematopathologists. In most cases, they do not necessarily reverse the diagnosis, but provide further clarification. It is occurring to me that in the community, pathologists are less likely to call the subtype of T-cell lymphoma and limit the report to the general description of T-cell lymphoprolipherative disorder, or state something like “consistent with T-cell lymphoma with features of AITL, or with features of anaplastic lymphoma.” I would admit though, that sometimes it's very difficult to identify the specific subtype of PTCL even in the expert hands; but I'd say these cases would constitute no more than 5% of PTCL patients.
DR. HORWITZ: And in your experience is it mostly fine tuning a T-cell lymphoma diagnosis, or do you see totally different diagnoses?
DR. MOSKOWITZ: Usually review by expert hematopathologists simply leads to fine-tuning the T-cell lymphoma diagnosis, however, I occasionally see significant changes in diagnosis. Often, alterations or clarification of a diagnosis are made possible only after we provide the pathologist with clinical history. For example, a lymph node biopsy may be interpreted as ALCL, however the knowledge that the particular patient has a history of mycosis fungoides would lead the pathologist to consider the diagnosis of large cell transformation of mycosis fungoides rather than ALCL. In such a case, molecular studies are helpful in confirming that the lymph node findings originate from the same clone as that in the original mycosis fungoides lesions, rather than representing a second primary.
DR. FANALE: Very occasionally, I've seen patients truly “with a misdiagnosis and a complete revision of diagnosis” and, usually, those pitfalls that I've seen them occasionally have been the young patients—the young patients who generally have disease in the thorax and neck, who are treated as though they have classical Hodgkin lymphoma, who have very significant progression of disease on standard of care treatment. So it's important that not all cases that have CD30 positivity are classical Hodgkin lymphoma even if the patient is young.
DR. HORWITZ: I often think the systemic T-cell lymphomas usually behave in an aggressive fashion so when the clinical picture and the diagnosis don't really fit, I think about getting a second biopsy before we finalize the diagnosis. Of course, when people are ill with obviously progressing disease, you may need to move more quickly.
I often think the systemic T-cell lymphomas usually behave in an aggressive fashion so when the clinical picture and the diagnosis don't really fit, I think about getting a second biopsy before we finalize the diagnosis. To move on, when you first see a patient, what is the decision tree in your mind in terms of picking therapy or planning therapy, or what kind of things do you consider?
DR. MOSKOWITZ: The first thing I think about when deciding upon treatment for a patient with T-cell lymphoma is whether or not I plan to use a curative approach to therapy. As was mentioned by Michelle, this decision is partly based upon the patients’ comorbidities and age. For patients who are eligible for curative therapy, our frontline approach for the most common types of T-cell lymphoma is to use CHOP with or without etoposide followed by consideration for autologous stem cell transplant in first remission. There are certainly individuals for whom such an aggressive strategy would not be appropriate due to age or co-morbidities. In these individuals, we may consider CHOP-based therapy alone or sometimes even milder approaches aimed at disease control.
DR. HORWITZ: Andrei, are you similar in the CHOP/CHOEP paradigm? And if so, what do you think of those data? How do you decide between the two and when do you do that regimen versus something different?
DR. SHUSTOV: I think I will double or triple what Michelle and Alison just said; for me, the two most important decisions that I have to make in the first encounter with patients with newly diagnosed PTCL are: 1) whether we're going to pursue curative approach strategy; and 2) whether the patient can tolerate the intensity of treatments that would provide him/her with the best chance of cure or long term remission. Patients who are elderly and have high risk disease would be very hard to cure, especially considering that the consolidative transplant might carry high rates of morbidity or mortality; more conservative strategies might be appropriate in these cases. On the other hand, younger and more fit patients might benefit more from intensified initial regimens—ie, CHOEP—followed by high-dose therapy and either autologous or allogeneic stem cell transplant (ALLO).
I usually have a long initial discussion with patients and families during which we decide on the intent of treatment and what to expect from certain regimens in terms of toxicities. I typically choose CHOEP regimen (or infusional version, etoposide, prednisone, vincristine, cyclophosphamide, doxorubicin [EPOCH]) for younger patients based on recent German data, even though this was a retrospective study and benefit of adding etoposide only approached statistical significance for the majority of PTCL subtypes.2 In older patients, I try to avoid anthracyclines, especially in the palliative intent setting, based on the retrospective analysis by the International T-Cell Lymphoma Project, and frequently use CEOP regimen (CHOEP minus anthracycline).3 I find that the majority of older patients tolerate this treatment somewhat better than anthracycline-containing combinations like CHOP.
In the very elderly and frail patients, I try to avoid combination chemotherapy all together. It is a somewhat easier decision for patients with AITL. Some of them are more indolent than other sub-types, and I would treat such patients with immunomodulatory approach, ie with a combination of prednisone and cyclosporine; then, I would consider single agent therapy with one of the recently approved agents for relapsed and refractory PTCL. I would also double what Michelle said in regard to making the best attempt to enroll patients into open clinical trials, because the current standards are not really satisfactory for many T-cell lymphoma patients.
DR. HORWITZ: It sounds like we all approach a new patient similarly. However, several of our up front trials are randomized against CHOP as opposed to CHOEP. When you're consenting a patient to those trials, how do you explain it to a patient or how important do you think the etoposide is?
DR. SHUSTOV: I share your frustration with quite a few of the current study designs for that exact reason. Some of these are confirmatory trials after conditional approvals of the new agents and are important. However, as often happens, in the confirmatory trials, we use controls that are a default from a lot of historical data, or the “common” way that the majority of patients are being treated. It is really a challenge when you consent patients to studies where a control arm is something that you think might not be quite adequate.
Having said that, the CHOEP study that was mentioned several times is a retrospective subgroup analysis and the addition of etoposide had marginal benefit that approached significance, but certainly was not a home run. I don't think we are ready to say that etoposide provides survival advantage in T-cell lymphoma patients and dismiss clinical trials in favor of just giving a patient the CHOEP, even though CHOP is admittedly not the best comparator. I discuss this controversy with patients and tell them frankly that the data that we base addition of etoposide on are not the best evidence one may have. Then, I ask patients to decide which approach sounds more reasonable to them and they make their choice.
DR. FANALE: To expand just slightly upon what Andrei said, I do emphasize to the patient and to the referring doctors that if you look at National Comprehensive Cancer Network guidelines, CHOP, CHOEP, EPOCH, all of these are potential options. So there's really not yet one trial that would say that one is clearly superior to the other at this time. I also emphasize to the patient and to the referring clinician that the only way, let's say, the patient could get the targeted agent plus the chemotherapy and have that 50% chance of, potentially, receiving that combination is really through the randomized clinical trial.
DR. HORWITZ: That was excellent, thank you. So you have alluded to this a bit. How much does subtype specific treatment come into play?
DR. MOSKOWITZ: At this point in time, outside of a clinical trial, for PTCL-NOS, AITL, and ALCL, the front-line approach would typically be CHOEP followed by autologous stem cell transplant. As Michelle mentioned, for a patient with ALCL, I would offer enrollment on the ECHELON-2 study in which patients are randomized to CHOP or BV-CHP, in order to give patients the option of potentially receiving BV.
For AITL, we are now obtaining molecular testing for certain mutations, particularly IDH2, because we currently have a study open with an IDH2 inhibitor that specifically enrolls patients with IDH2-mutant AITL. Because the testing takes some time to get back, we typically test patients’ biopsies at the time of diagnosis, so that we know if they're eligible for future clinical trials in case their disease does not respond to upfront therapy.
There are T-cell lymphoma subtypes that we treat quite differently from the entities discussed so far. For human T-lymphotropic virus-1 associated adult T-cell lymphoma, for example, the data with CHOP are really not good and we typically used EPOCH with the aim to consolidate with an allogeneic stem cell transplant.
Likewise, for hepatosplenic T-cell lymphoma, we have not observed good responses with the CHOP-based therapy and therefore we typically use platinum-based or ifosfamide-based therapy upfront with the aim to consolidate with allogeneic stem cell transplant. Extranodal NK/T-cell lymphoma is another entity for which CHOP is typically not effective and we have adopted asparaginase-based regimens, such as dexamethasone, methotrexate, ifosfamide, L-asparaginase, etoposide (SMILE) for this disease.
DR. SHUSTOV: I completely agree with Alison; for more common nodal lymphomas it is really hard at this point to base a treatment decision on histology, at least, in the front-line setting. However, some of the rare and unique subtypes, we generally treat with a completely different approach (ie, extranodal NK-cell lymphomas, enteropathy-associated T-cell lymphomas, adult T-cell leukemia/lymphomas).
DR. HORWITZ: Excellent. I’m curious, in a patient with ALCL who refused randomization of the ECHELON-2 trial, would you give them BV-CHP off study or would you discuss that as an alternative were they not to participate in the trial?
DR. FANALE: No. I don't urge treatment that's not approved for a particular line of therapy off clinical protocol with the exception that, let's say, if a patient is not a candidate for chemotherapy in second line setting for a particular lymphoma such as Hodgkin lymphoma—then, I would reference published data like Alison's data for a second line use of BV say for classical Hodgkin lymphoma and work with the insurance company to get the drug approved for that patient population, but if a patient just doesn't want to go on trial because they don't like the 50/50 chance of receiving BV-CHP compared to CHOP and they say, “I really want the 100% chance. Why don't you just contact my insurance?” I will explain to them the rationale of why the trial is being conducted including the hope that if the endpoints are met and the regimen is approved for front-line therapy then patients in the future can get BV-CHP at any oncology clinic, but I tell them for now it is a protocol based treatment option.
DR. HORWITZ: That's a very good answer. Can we now touch on the idea of transplant vs no transplant in first remission. Always, sometimes, never; how do you all think about that?
DR. SHUSTOV: I think that the idea of consolidative transplant is very heavily debated and discussed at the majority of specialized PTCL meetings. The reason for that is controversial nature of current clinical evidence. In the absence of randomized trials it is very hard to compare prospective phase II trial data to historical controls. You can interpret this in two ways; you might say, well, these are good data, outcome numbers look better than historical controls, and all patients with PTCL should have a consolidative transplant; or you can say, well, I don't have randomized data, and only a randomized study would really tell us whether post-induction consolidative transplant improves survival.
That is why it is such a controversial subject, and we agree that the phase II perspective studies of transplantation are hampered by significant bias; patients who are able and willing to travel to academic centers, were more robust and able to tolerate high dose therapy. They also have chemosensitive disease and that puts them in a completely different category than those you see in populational or retrospective studies, or other historical control trials.
Having said that, when I talk to patients, and I think that this is where I involve patients into treatment decision very heavily, I present them with the data and say that it is not wrong to do or not do the transplant in first CR. I make patients and family a big part of the decision. When they ask me what would give the patient the best chance being in remission five years from today, my answer is transplant, however, the data are not perfect, and the curative potential of autologous transplant in PTCL is not known.
DR. FANALE: So typically here, we would refer most patients on for front-line consolidative autologous stem cell transplant, I think, some exceptions are the ones that Andrei already touched on. If we're seeing a patient who has PTCL-NOS and this patient is the very rare patient who has early stage disease, with really no significant risk factors, that patient, unless there were pathologic features that showed a higher level of aggressiveness as Alison commented on, this patient might be one where we might defer doing the consolidative stem cell transplant for particularly if the patient’s disease entered into remission very quickly, but this still is controversial.
I'm not sure how you practice within each of your centers, but another patient, or where we would potentially defer, is a patient with fairly limited nasal NK/T-cell lymphoma, a patient that we’ve treated, let's say, with the dexamethasone, etoposide, ifosfamide, carboplatin (DeVIC) chemotherapy regimen plus a concomitant high-dose radiation, we typically historically here have deferred doing consolidative stem cell transplant for that patient population.
In terms of what Andrei commented on for when we might consider an ALLO and for a frontline consolidation, here we typically would not. The exception to that rule is that I've had a couple of patients here who have PTCL with a secondary hemophagocytic lymphohistiocytosis syndrome and then, for those patients because the concern is that the autologous stem cell transplant probably wouldn't be enough and for those patients given that they have such a dismal prognosis from the secondary hemophagocytic lymphohistiocytosis, we send those patients on for an ALLO.
DR. MOSKOWITZ: To expand upon early stage extranodal NK/T-cell lymphoma, I agree with Michelle that we wouldn't use an autologous stem cell transplant in first remission. Typically, for patients with localized disease, we recommend 2 cycles of SMILE followed by involved field radiation. Our patients treated with this approach have typically done quite well without needing a transplant.
DR. HORWITZ: That was great. If there are no further comments on upfront therapy, we can move over to relapse setting. When you see a patient at relapse, what are your strategies and how do you approach that patient?
DR. MOSKOWITZ: The first question in my mind when a patient is either not responding to or relapsing following front-line therapy is: what is my ultimate goal? For a patient who is fit and refractory to CHOP or has relapsed after CHOP or autologous stem cell transplant, my ultimate goal would be to try to get them to an ALLO. My choice of treatment following front-line therapy depends upon whether or not we are aiming for an ALLO as well as whether a donor has been identified for the patient.
Usually, if a patient just relapsed following front-line therapy, we are not necessarily ready to proceed quickly to ALLO. This may be because a donor has not yet been identified or because the patients’ eligibility for an ALLO is not clear. In this situation, my choice of treatment would be something that can be continued, potentially for several months, while we get things in place for the transplant. Treatment options in this situation would include the approved single agents for T-cell lymphoma, such as romidepsin, belinostat, pralatrexate, as well as BV (which would be specific for ALCL). In addition, I would consider enrollment on a promising clinical trial. Among these options, my choice of treatment typically depends upon the side effect profile and/or schedule, which is individualized for the patient. The one caveat would be that I typically will aim to use a histone deacetylase inhibitor (HDAC) inhibitor as my first choice for a patient with AITL.
Usually, if a patient just relapsed following front-line therapy, we are not necessarily ready to proceed quickly to ALLO. This may be because a donor has not yet been identified or because the patients’ eligibility for an ALLO is not clear. If my patient has a donor available and we are ready to move quickly to transplant, I would use one of the multi-agent regimens, such as ifosfamide, carboplatin, etoposide (ICE) or cisplatin, cytosine arabinoside, dexamethasone (DHAP), with the aim to try to get them into remission and relatively quickly proceed to transplant. The problem with using one of these types of regimens for everyone in the second-line setting is that the treatment cannot be continued indefinitely due to cumulative toxicity; therefore we need to know that we are ready to proceed to transplant if we are going to use one of the more intense regimens.
DR. HORWITZ: How do the others approach relapse?
DR. FANALE: Generally, a lot of similarities with what Alison discussed. Typically, it's if we're going to be sending the patient on for consideration for ALLO, I think the favor would be to try to use a regimen that has a high CR rate knowing that there is a trend toward improved progression free survival for the patient who enters into CR when compared to those who did not. I think the only slight difference might be just the timing and the selection, so, typically, here, even if the patient doesn't actually have a definite established donor quite yet we'll typically favor a combination type of a treatment approach, so whether or not that would be a platinum-based regimen such as ICE, as Alison mentioned, or gemcitabine-based chemotherapy regimen or similar.
I would generally favor one of those options, usually, over a single-agent therapeutic approach just knowing that the CR rates, generally, with exception of BV for ALCL are, generally, ranging about the 10%–15% range. And then, of course, first priority would be if there is a clinical trial available whether that's with a doublet of targeted agents or with the targeted agent plus chemotherapy in the relapse setting, we would typically favor that approach for a patient being considered for a stem cell transplant, ALLO approach.
DR. SHUSTOV: I'll consolidate what Michelle and Alison just said. I think we're on the same page. To me, the most important decision (even more important in the relapse setting) is to figure out whether we are still going to try to cure the lymphoma or we will pursue a palliative approach for which now we have several treatment options. So again, I’d have a long discussion with the patient, whether we're going to try and cure the disease, or we will pursue a palliative approach.
If this is a curative approach strategy and we're heading toward ALLO, we would start searching for the donor immediately. The choice of salvage therapy depends on duration of first remission. Primary refractory disease patients who failed CHOP, CHOEP, or EPOCH outright or within 3 months, I typically consider using the single agent approach, because even ICE and DHAP type regimens don’t work well for these patients. I put my “money” into HDAC inhibitors or antifolates, or other targeted therapy, if this is anaplastic lymphoma.
For patients who had at least 6 months remission from the initial therapy, younger patients who are still fit, I agree, I would consider standard salvage regimens like DHAP, etoposide, methylprednisolone, high-dosecytarabine, cisplatin (ESHAP) and ICE, and then, follow this with allograft.
DR. HORWITZ: You have all talked about allografting, I have two questions. What is your best guess in terms of percentage of patients you see with relapsed T-cell lymphoma who actually undergo ALLO at some point and do you ever consider autologous transplantation in those who didn't have it as part of their initial therapy?
DR. SHUSTOV: It certainly depends on the type of the academic center that patients are referred to, and we all practice at academic institutions with very strong ALLO programs. In patients who we select for a curative approach (one half to two thirds) we will at least make the best effort to take them to allogeneic grafting. Patients might fall off this track if they don't respond to salvage therapies or develop significant treatment/disease-related complications, ie fungal infections, organ toxicities, poor performance status, etc. Overall, I'd say that we successfully take about half of the patients with relapsed PTCL that are willing to pursue curative approach to allograft.
In addition, I consider autologous stem cell transplant in relapse setting for three situations. One is relapsed ALK-positive ALCL, some patients with ALK-negative ALCL who have had long initial remission and after relapse, and achieved second remission with BV or other salvage therapy. Finally, I’d consider auto-transplant in patients with AITL who achieved second CR with salvage therapy.
DR. HORWITZ: In terms of the relapse setting—some of you talked about angioimmunoblastic and HDAC inhibitors or BV for ALCL—when you have a relapse patient with PTCL-NOS, how do you pick a second line agent if you're not going directly to transplant, and are there new drugs that you're particularly excited about?
DR. FANALE: To answer your questions in terms of how I might choose a targeted agent for a patient who has PTCL-NOS and doesn't have AITL or ALCL, if the patient isn't eligible for trial or trial isn't feasible for that patient, I'll typically look at schedule, and then I'll look at side effect profile. For instance, for comparison, there are two HDAC inhibitor drugs right now approved, romidepsin and belinostat, so I think that potentially in a private practice setting, belinostat might be one when you look at the efficacy, being generally, around equivalent on where a patient might prefer that regimen because of the fact that they come in for five days in a row and then, their treatment is done for that cycle.
At a larger referral center to which patients are often traveling back and forth, it can be quite difficult for a patient to stay in town to do five days of treatment in a row. Often, these patients will prefer the romidepsin schedule because of the fact that they're coming back in once per week for three weeks in a row per cycle. The other way that I'll look at it is side effect profile, so, let's say, pralatrexate compared to the HDAC inhibitors, I will usually favor the HDAC inhibitors because of the potential side effect of mucositis with pralatrexate, although I have been able to manage through the mucositis by giving the patients more of a cutaneous T-cell lymphoma dosing format.
There had been a high level of interest in new emerging agents such as the aurora A-kinase inhibitors, alisertib, and that trial is now complete and the data are to be presented at The American Society of Hematology’s Annual Meeting this year.4 Other new therapies that are emerging are the PI3K inhibitors such as duvelisib and others. One advantage for many of the drugs in current trials is that they are oral agents.
My interest also lies in the combination approach, such as two or three targeted agent combinations, with the goal that if you see favorable CR rates and progression-free survival rates in the relapsed setting that you can then potentially move these treatments to the front-line setting to potentially move beyond the CHOP-based platform of therapies.
DR. SHUSTOV: For patients outside anaplastic lymphoma where, obviously, most of us would likely use BV, given the response rate to this agent, the decision is really based on convenience of the schedule, toxicity profile, and—sometimes—patient’s preference. So, toxicity and quality of life become kind of more decision-driving factors than disease biology. I’d have a discussion with patients about all three drugs, how they are administered, and what toxicities can be expected. Four-hour infusion of romidepsin might not be acceptable for some patients who have to travel long distances to treatment centers. They may prefer more rapid infusion (they would favor a trial of pralatrexate; or, as Alison mentioned, patient prefers a five-day/daily versus weekly administration.
It's kind of a coin toss decision outside clinical trials. Certainly, participation in studies in relapse setting is the high priority. We are all really looking forward to developing doublet combinations of novel agents, ie studies like the one Michelle is running at MD Anderson with a combination of alisertib and romidepsin.
DR. MOSKOWITZ: I'll echo what both Andrei and Michelle said that our choice of treatment is individualized and depends upon side-effect profile and the schedule. With regard to novel agents, I am also very excited about the studies that will be opening with the PI-3 kinase inhibitor, IPI-145. There have been promising results seen with single-agent IPI-1455 and we will be opening a study evaluating it in combination with bortezomib or romidepsin.
In addition, I mentioned earlier the IDH2 inhibitor, which we're studying right now in patients with AITL that is characterized by the presence of the IDH2 mutation. It would be exciting to see if targeting a specific mutation in this disease translates into responses.
DR. HORWITZ: Great, I think that was fantastic. I would like to thank Drs. Moskowitz, Shustov, and Fanale for a very thorough impractical discussion on how they approach and manage patients with T-cell lymphoma. I’m impressed that there is significant consistency among these experts in terms of how they manage these uncommon and often challenging lymphomas in terms of upfront combination chemotherapy approaches, combination considerations for the use of transplantation, as well as their enthusiasm for and dedication to incorporating clinical trials as part of everyday management.
References
1. Fanale MA, Horwitz SM, Forero-Torres A, et al. Brentuximab vedotin in the front-line treatment of patients with CD30+ peripheral T-cell lymphomas: results of a phase I study. J Clin Oncol. 2014;32(28):3137–3143.
2. Schmitz N, Trümper L, Ziepert M, et al. Treatment and prognosis of mature T-cell and NK-cell lymphoma: an analysis of patients with T-cell lymphoma treated in studies of the German High-Grade Non-Hodgkin Lymphoma Study Group. Blood. 2010;116(18):3418–3425. doi: 10.1182/blood-2010-02-270785. Epub 2010 Jul 21.
3. Vose J, Armitage J, Weisenburger D, for the International T-Cell Lymphoma Project. J Clin Oncol. 2008;26(25):4124–4130. doi: 10.1200/JCO.2008.16.4558. Epub 2008 Jul 14.
4. O’Connor OA, Ozcan M, Jacobsen ED, et al. First multicenter, randomized phase 3 study in patients with relapsed/refractory peripheral T-cell lymphoma: alisertib versus investigator’s choice. Paper to be presented at: American Society of Hematology 57th Annual Meeting & Exposition; December 5–8, 2015; Orlando, FL.
5. Horwitz SM, Porcu P, Flinn I, et al. Duvelisib (IPI-145), a phosphoinositide-3-kinsae-γδ inhibitor, shows activity in patients with relapsed/refractory T-cell lymphoma. Paper presented at: American Society of Hematology 56th Annual Meeting & Exposition; December 6–9, 2014; San Francisco, CA.

