User login
Recent developments in the treatment of high-grade gliomas
Patients with glioblastoma and other high-grade gliomas have poor outcomes and are challenging to treat. The relative rarity of these tumors has made large-scale, practice-changing trials difficult to accomplish and has led to the formation of large multinational organizations that focus on neuro-oncology. This has resulted in the rapid completion of several large trials that in some cases have set new standards of care that can offer increased progression-free and overall survivals for some patients. The incorporation of correlative tissue studies in these trials has led to the identification of prognostic and predictive genetic markers that demonstrate the heterogeneity of these tumors and will assist in developing individualized treatment strategies as research continues to uncover new therapeutic targets. This review of recently completed and in-progress phase 3 trials in high-grade gliomas highlights the developments and future directions in the treatment of these tumors...
*For PDFs of the full article and related Commentary, click on the links to the left of this introduction.
Patients with glioblastoma and other high-grade gliomas have poor outcomes and are challenging to treat. The relative rarity of these tumors has made large-scale, practice-changing trials difficult to accomplish and has led to the formation of large multinational organizations that focus on neuro-oncology. This has resulted in the rapid completion of several large trials that in some cases have set new standards of care that can offer increased progression-free and overall survivals for some patients. The incorporation of correlative tissue studies in these trials has led to the identification of prognostic and predictive genetic markers that demonstrate the heterogeneity of these tumors and will assist in developing individualized treatment strategies as research continues to uncover new therapeutic targets. This review of recently completed and in-progress phase 3 trials in high-grade gliomas highlights the developments and future directions in the treatment of these tumors...
*For PDFs of the full article and related Commentary, click on the links to the left of this introduction.
Patients with glioblastoma and other high-grade gliomas have poor outcomes and are challenging to treat. The relative rarity of these tumors has made large-scale, practice-changing trials difficult to accomplish and has led to the formation of large multinational organizations that focus on neuro-oncology. This has resulted in the rapid completion of several large trials that in some cases have set new standards of care that can offer increased progression-free and overall survivals for some patients. The incorporation of correlative tissue studies in these trials has led to the identification of prognostic and predictive genetic markers that demonstrate the heterogeneity of these tumors and will assist in developing individualized treatment strategies as research continues to uncover new therapeutic targets. This review of recently completed and in-progress phase 3 trials in high-grade gliomas highlights the developments and future directions in the treatment of these tumors...
*For PDFs of the full article and related Commentary, click on the links to the left of this introduction.
Second TNF-Blocker Approved for Refractory Ulcerative Colitis
Adalimumab, a subcutaneously administered tumor necrosis factor blocker, has been approved for treating adults with moderately to severely active ulcerative colitis who have not had an adequate response with conventional treatments, the Food and Drug Administration announced on Sept. 28.
The safety and effectiveness of adalimumab for this patient population was established in two clinical studies of 908 patients with moderately to severely active ulcerative colitis (UC).
Adalimumab, marketed as Humira by Abbott Laboratories, was first approved for treating rheumatoid arthritis in 2002, followed by psoriatic arthritis in 2005, ankylosing spondylitis in 2006, Crohn’s disease in 2007, and plaque psoriasis and juvenile idiopathic arthritis in 2008.
Adalimumab is the second TNF blocker to be approved for ulcerative colitis; infliximab (Remicade), an intravenous TNF blocker, was previously approved for treating UC.
Clinical remission rates in the two studies were significantly greater among patients treated with infliximab than among those who received placebo: In an 8-week study, which did not include patients who had previously been treated with a TNF blocker, the clinical remission rate at 8 weeks was 18.5% among those on adalimumab vs. 9.2% in those on placebo, a 9.3% difference. In the second study, which followed patients for 1 year and included some who had been treated with infliximab, the clinical remission rate at 8 weeks was 16.5% among those on adalimumab, vs. 9.3% among those on placebo, a 7.2% difference.
At a meeting on Aug. 28 held to review these data, the majority of the FDA’s Gastrointestinal Drugs Advisory Committee agreed that these differences represented clinically meaningful benefits and supported approval of adalimumab for this indication. Panelists cited the need for more treatments for UC and for a subcutaneous TNF blocker for these patients, as well as its potential steroid-sparing effects.
In the studies, no new side effects were identified, the agency said. The FDA statement points out that the effectiveness of adalimumab "has not been established in patients with ulcerative colitis who have lost response to or were intolerant to TNF blockers."
The approved dosing regimen for adalimumab is a starting dose of 160 mg, followed by a second dose of 80 mg 2 weeks later and then a maintenance dose of 40 mg every other week. "The drug should only continue to be used in patients who have shown evidence of clinical remission by 8 weeks of therapy," according to the FDA statement.
Adalimumab is the first self-administered biologic treatment for ulcerative colitis to be approved.
Adalimumab, a subcutaneously administered tumor necrosis factor blocker, has been approved for treating adults with moderately to severely active ulcerative colitis who have not had an adequate response with conventional treatments, the Food and Drug Administration announced on Sept. 28.
The safety and effectiveness of adalimumab for this patient population was established in two clinical studies of 908 patients with moderately to severely active ulcerative colitis (UC).
Adalimumab, marketed as Humira by Abbott Laboratories, was first approved for treating rheumatoid arthritis in 2002, followed by psoriatic arthritis in 2005, ankylosing spondylitis in 2006, Crohn’s disease in 2007, and plaque psoriasis and juvenile idiopathic arthritis in 2008.
Adalimumab is the second TNF blocker to be approved for ulcerative colitis; infliximab (Remicade), an intravenous TNF blocker, was previously approved for treating UC.
Clinical remission rates in the two studies were significantly greater among patients treated with infliximab than among those who received placebo: In an 8-week study, which did not include patients who had previously been treated with a TNF blocker, the clinical remission rate at 8 weeks was 18.5% among those on adalimumab vs. 9.2% in those on placebo, a 9.3% difference. In the second study, which followed patients for 1 year and included some who had been treated with infliximab, the clinical remission rate at 8 weeks was 16.5% among those on adalimumab, vs. 9.3% among those on placebo, a 7.2% difference.
At a meeting on Aug. 28 held to review these data, the majority of the FDA’s Gastrointestinal Drugs Advisory Committee agreed that these differences represented clinically meaningful benefits and supported approval of adalimumab for this indication. Panelists cited the need for more treatments for UC and for a subcutaneous TNF blocker for these patients, as well as its potential steroid-sparing effects.
In the studies, no new side effects were identified, the agency said. The FDA statement points out that the effectiveness of adalimumab "has not been established in patients with ulcerative colitis who have lost response to or were intolerant to TNF blockers."
The approved dosing regimen for adalimumab is a starting dose of 160 mg, followed by a second dose of 80 mg 2 weeks later and then a maintenance dose of 40 mg every other week. "The drug should only continue to be used in patients who have shown evidence of clinical remission by 8 weeks of therapy," according to the FDA statement.
Adalimumab is the first self-administered biologic treatment for ulcerative colitis to be approved.
Adalimumab, a subcutaneously administered tumor necrosis factor blocker, has been approved for treating adults with moderately to severely active ulcerative colitis who have not had an adequate response with conventional treatments, the Food and Drug Administration announced on Sept. 28.
The safety and effectiveness of adalimumab for this patient population was established in two clinical studies of 908 patients with moderately to severely active ulcerative colitis (UC).
Adalimumab, marketed as Humira by Abbott Laboratories, was first approved for treating rheumatoid arthritis in 2002, followed by psoriatic arthritis in 2005, ankylosing spondylitis in 2006, Crohn’s disease in 2007, and plaque psoriasis and juvenile idiopathic arthritis in 2008.
Adalimumab is the second TNF blocker to be approved for ulcerative colitis; infliximab (Remicade), an intravenous TNF blocker, was previously approved for treating UC.
Clinical remission rates in the two studies were significantly greater among patients treated with infliximab than among those who received placebo: In an 8-week study, which did not include patients who had previously been treated with a TNF blocker, the clinical remission rate at 8 weeks was 18.5% among those on adalimumab vs. 9.2% in those on placebo, a 9.3% difference. In the second study, which followed patients for 1 year and included some who had been treated with infliximab, the clinical remission rate at 8 weeks was 16.5% among those on adalimumab, vs. 9.3% among those on placebo, a 7.2% difference.
At a meeting on Aug. 28 held to review these data, the majority of the FDA’s Gastrointestinal Drugs Advisory Committee agreed that these differences represented clinically meaningful benefits and supported approval of adalimumab for this indication. Panelists cited the need for more treatments for UC and for a subcutaneous TNF blocker for these patients, as well as its potential steroid-sparing effects.
In the studies, no new side effects were identified, the agency said. The FDA statement points out that the effectiveness of adalimumab "has not been established in patients with ulcerative colitis who have lost response to or were intolerant to TNF blockers."
The approved dosing regimen for adalimumab is a starting dose of 160 mg, followed by a second dose of 80 mg 2 weeks later and then a maintenance dose of 40 mg every other week. "The drug should only continue to be used in patients who have shown evidence of clinical remission by 8 weeks of therapy," according to the FDA statement.
Adalimumab is the first self-administered biologic treatment for ulcerative colitis to be approved.
New Gout Guidelines Inspired by Recent Data
The first guidelines on the management of gout from the American College of Rheumatology recommend new ways of using old drugs and changes in prophylaxis strategies, among other things.
The two-part guidelines, published online Sept. 28, should help speed up effective treatment of gout and get physicians to treat patients to a target urate level of less than 6 mg/dL in order to improve symptoms, Dr. John D. FitzGerald said in an interview.
"There has been a fair amount of recent movement on gout medications" including new alternatives to allopurinol and colchicine and new data on how to use those traditional drugs in safer ways, said Dr. FitzGerald, acting chief of the rheumatology division at the University of California, Los Angeles. "It’s a fair number of changes for medications that people had been using for decades."
The documents update previous guidelines from medical organizations in Europe, the Netherlands, and Japan. The new guidelines will be published in October 2012 by the journal Arthritis Care & Research.
Part 1 of the American College of Rheumatology (ACR) guidelines covers nonpharmacologic and pharmacologic approaches to managing hyperuricemia (Arthritis Care Res. 2012;64:1431-46 [doi:10.1002/acr.21772]).
Part 2 addresses prophylaxis and treatment for acute gouty arthritis (Arthritis Care Res. 2012;64:1447-61 [doi.wiley.com/10.1002/acr.21773]).
Dr. FitzGerald and two other co-leaders of the project, Dr. Dinesh Khanna of the University of Michigan, Ann Arbor and Dr. Robert Terkeltaub of the University of California, San Diego, reviewed the medical literature on gout from the 1950s to the present and drew up nine clinical case scenarios commonly seen in practice. A task force panel comprising seven rheumatologists, two primary care physicians, a nephrologist, and a patient representative used the scenarios to create consensus recommendations.
Among the recommendations, for example, on the use of allopurinol is to start at a low dose of 100 mg/day (instead of the common practice of starting with 300 mg/day), or even lower for patients with chronic kidney disease, and then gradually titrate upward every 2-5 weeks. That recommendation supports previous statements from the Food and Drug Administration and the European League Against Rheumatism.
Also, allopurinol therapy should be actively managed and patients followed to make sure the uric acid target is achieved. "You can’t just give a prescription and say your job is done," though some recent studies suggest that many physicians do just that, Dr. FitzGerald said. "The corollary would be if someone gave blood pressure medication and then didn’t follow the patient’s blood pressure. That wouldn’t be seen as good medicine."
Maintenance doses of allopurinol to prevent acute gout attacks can exceed 300 mg even in patients with chronic kidney disease provided there is adequate patient education and monitoring.
A new recommendation drops the starting dose of oral colchicine for acute gout attacks to a loading dose of 1.2 mg, followed by 0.6 mg an hour later, and then starting prophylaxis 12 hours later at dosing of 0.6 mg once or twice daily.
"We used to give up to eight tablets a day," Dr. FitzGerald said. "That is dropped down to three to four tablets at the start of an attacks, because of findings that more colchicine didn’t really help outcomes" and that smaller doses are safer. The authors called this recommendation from ACR "a paradigm shift" that’s in accordance with Food and Drug Administration-approved label language.
Other highlights of the new ACR recommendations include sections on screening for HLA-B*5801 in patients at high risk of severe adverse reaction to allopurinol, combination therapy when target urate levels are not achieved, medication options including new drugs, and more.
Although the reports are titled "Guidelines," the text makes clear that they are expert recommendations and that clinicians are expected to take active roles in choosing the best management strategies for their particular patients. The authors were "very concerned" that the guidelines not be used by third-party payers to restrict access to medications or to promote one drug over another if there isn’t clear evidence to support it, Dr. FitzGerald said.
The methodology of the project precluded evaluations of costs and cost effectiveness, instead focusing on efficacy. So, for example, the guidelines say that allopurinol and febuxostat can be used equivalently in some circumstances, but clinicians need to consider all other aspects of these options including cost, patient preference, and more.
The ACR plans to update the guidelines as new data become available. The task force panel did create specific indications for use of imaging studies because results should be available in the next few years from studies on the use of high-resolution ultrasound and dual-energy CT for patients with gout.
In the United States, gout affects an estimated 4% of adults – more than 8 million people.
"I’m most excited and hopeful about trying to get this out to internal medicine and family practice doctors," Dr. FitzGerald said. "They see more gout than rheumatologists."
Dr. FitzGerald reported having no financial disclosures. Some members of the task force reported financial associations with multiple pharmaceutical companies but, by design, a majority of task force members had no perceived potential conflicts of interest.
Writing guidelines on gout is a difficult task. I think they made a very good effort to cover as many treatment issues as they could.
Most patients with gout in the United States are cared for by primary care physicians. The guidelines will be helpful to both primary practitioners and rheumatologists, but the subtleties may be lost on the general practitioner, whereas the rheumatologist would pick these up right away. The devil is often in the details when it comes to treating gout. If physicians use the guidelines employing a cookbook approach, they might run into some problems.
For instance, the guidelines cover the use of colchicine as a first-line agent for an acute attack: It’s a good choice, but even the randomized controlled trials that have been published on this, especially using the low-dose approach, show that a significant proportion of patients will not respond to this regimen. The guidelines recommend a dosage higher than what has been advised previously for the low-dose colchicine approach. This may actually be a better method, so I hope this will allow primary practitioners to be able identify more people using this approach. But there are definitely going to be people who do not respond to the colchicine.
Another example of where the guidelines may mislead primary care physicians is the recommendation on when to start urate-lowering therapy (ULT). Their indications for starting pharmacologic ULT include an established diagnosis of gouty arthritis and at least two attacks per year. My colleagues and I think that may exclude too many people. Theoretically, you could have a patient with one attack per year who is having gout-related joint damage and, with this criteria, wouldn’t qualify for ULT. A rheumatologist would pick that up right away, but general practitioners who adhere to these guidelines might end up undertreating some patients.
Also, they recommend using adrenocorticotropic hormone (ACTH) for people who cannot take oral medications. Not only is ACTH is extremely expensive, but the Food and Drug Administration has taken gout off the list of indications for ACTH, so I doubt it would be readily available in a real clinical situation.
When the recommendations discuss using prednisone as a prophylactic against gout attacks, they suggest using 10 mg or less. I think that the authors are trying for the best of both worlds and ending up not having either. We generally try to avoid using steroids long term, so the authors suggest using low-dose prednisone; the problem is that 10 mg would probably be ineffective. There are data suggesting that gout prophylaxis requires higher doses, maybe as much as 20 mg/day. You could try 10 mg but I anticipate that it is not going to work very well.
In their defense, were the authors to go into the subtleties and side effects, what to do with a patient with liver or coronary disease, or issues of cost effectiveness, the guidelines would have become an unmanageable length. But the devil is in the details.
That said, it’s a major effort here. It’s good work. They tried to answer a lot of questions.
Dr. Christopher M. Burns is a rheumatologist at the Geisel School of Medicine at Dartmouth, Hanover, N.H. He reported having no financial disclosures.
Writing guidelines on gout is a difficult task. I think they made a very good effort to cover as many treatment issues as they could.
Most patients with gout in the United States are cared for by primary care physicians. The guidelines will be helpful to both primary practitioners and rheumatologists, but the subtleties may be lost on the general practitioner, whereas the rheumatologist would pick these up right away. The devil is often in the details when it comes to treating gout. If physicians use the guidelines employing a cookbook approach, they might run into some problems.
For instance, the guidelines cover the use of colchicine as a first-line agent for an acute attack: It’s a good choice, but even the randomized controlled trials that have been published on this, especially using the low-dose approach, show that a significant proportion of patients will not respond to this regimen. The guidelines recommend a dosage higher than what has been advised previously for the low-dose colchicine approach. This may actually be a better method, so I hope this will allow primary practitioners to be able identify more people using this approach. But there are definitely going to be people who do not respond to the colchicine.
Another example of where the guidelines may mislead primary care physicians is the recommendation on when to start urate-lowering therapy (ULT). Their indications for starting pharmacologic ULT include an established diagnosis of gouty arthritis and at least two attacks per year. My colleagues and I think that may exclude too many people. Theoretically, you could have a patient with one attack per year who is having gout-related joint damage and, with this criteria, wouldn’t qualify for ULT. A rheumatologist would pick that up right away, but general practitioners who adhere to these guidelines might end up undertreating some patients.
Also, they recommend using adrenocorticotropic hormone (ACTH) for people who cannot take oral medications. Not only is ACTH is extremely expensive, but the Food and Drug Administration has taken gout off the list of indications for ACTH, so I doubt it would be readily available in a real clinical situation.
When the recommendations discuss using prednisone as a prophylactic against gout attacks, they suggest using 10 mg or less. I think that the authors are trying for the best of both worlds and ending up not having either. We generally try to avoid using steroids long term, so the authors suggest using low-dose prednisone; the problem is that 10 mg would probably be ineffective. There are data suggesting that gout prophylaxis requires higher doses, maybe as much as 20 mg/day. You could try 10 mg but I anticipate that it is not going to work very well.
In their defense, were the authors to go into the subtleties and side effects, what to do with a patient with liver or coronary disease, or issues of cost effectiveness, the guidelines would have become an unmanageable length. But the devil is in the details.
That said, it’s a major effort here. It’s good work. They tried to answer a lot of questions.
Dr. Christopher M. Burns is a rheumatologist at the Geisel School of Medicine at Dartmouth, Hanover, N.H. He reported having no financial disclosures.
Writing guidelines on gout is a difficult task. I think they made a very good effort to cover as many treatment issues as they could.
Most patients with gout in the United States are cared for by primary care physicians. The guidelines will be helpful to both primary practitioners and rheumatologists, but the subtleties may be lost on the general practitioner, whereas the rheumatologist would pick these up right away. The devil is often in the details when it comes to treating gout. If physicians use the guidelines employing a cookbook approach, they might run into some problems.
For instance, the guidelines cover the use of colchicine as a first-line agent for an acute attack: It’s a good choice, but even the randomized controlled trials that have been published on this, especially using the low-dose approach, show that a significant proportion of patients will not respond to this regimen. The guidelines recommend a dosage higher than what has been advised previously for the low-dose colchicine approach. This may actually be a better method, so I hope this will allow primary practitioners to be able identify more people using this approach. But there are definitely going to be people who do not respond to the colchicine.
Another example of where the guidelines may mislead primary care physicians is the recommendation on when to start urate-lowering therapy (ULT). Their indications for starting pharmacologic ULT include an established diagnosis of gouty arthritis and at least two attacks per year. My colleagues and I think that may exclude too many people. Theoretically, you could have a patient with one attack per year who is having gout-related joint damage and, with this criteria, wouldn’t qualify for ULT. A rheumatologist would pick that up right away, but general practitioners who adhere to these guidelines might end up undertreating some patients.
Also, they recommend using adrenocorticotropic hormone (ACTH) for people who cannot take oral medications. Not only is ACTH is extremely expensive, but the Food and Drug Administration has taken gout off the list of indications for ACTH, so I doubt it would be readily available in a real clinical situation.
When the recommendations discuss using prednisone as a prophylactic against gout attacks, they suggest using 10 mg or less. I think that the authors are trying for the best of both worlds and ending up not having either. We generally try to avoid using steroids long term, so the authors suggest using low-dose prednisone; the problem is that 10 mg would probably be ineffective. There are data suggesting that gout prophylaxis requires higher doses, maybe as much as 20 mg/day. You could try 10 mg but I anticipate that it is not going to work very well.
In their defense, were the authors to go into the subtleties and side effects, what to do with a patient with liver or coronary disease, or issues of cost effectiveness, the guidelines would have become an unmanageable length. But the devil is in the details.
That said, it’s a major effort here. It’s good work. They tried to answer a lot of questions.
Dr. Christopher M. Burns is a rheumatologist at the Geisel School of Medicine at Dartmouth, Hanover, N.H. He reported having no financial disclosures.
The first guidelines on the management of gout from the American College of Rheumatology recommend new ways of using old drugs and changes in prophylaxis strategies, among other things.
The two-part guidelines, published online Sept. 28, should help speed up effective treatment of gout and get physicians to treat patients to a target urate level of less than 6 mg/dL in order to improve symptoms, Dr. John D. FitzGerald said in an interview.
"There has been a fair amount of recent movement on gout medications" including new alternatives to allopurinol and colchicine and new data on how to use those traditional drugs in safer ways, said Dr. FitzGerald, acting chief of the rheumatology division at the University of California, Los Angeles. "It’s a fair number of changes for medications that people had been using for decades."
The documents update previous guidelines from medical organizations in Europe, the Netherlands, and Japan. The new guidelines will be published in October 2012 by the journal Arthritis Care & Research.
Part 1 of the American College of Rheumatology (ACR) guidelines covers nonpharmacologic and pharmacologic approaches to managing hyperuricemia (Arthritis Care Res. 2012;64:1431-46 [doi:10.1002/acr.21772]).
Part 2 addresses prophylaxis and treatment for acute gouty arthritis (Arthritis Care Res. 2012;64:1447-61 [doi.wiley.com/10.1002/acr.21773]).
Dr. FitzGerald and two other co-leaders of the project, Dr. Dinesh Khanna of the University of Michigan, Ann Arbor and Dr. Robert Terkeltaub of the University of California, San Diego, reviewed the medical literature on gout from the 1950s to the present and drew up nine clinical case scenarios commonly seen in practice. A task force panel comprising seven rheumatologists, two primary care physicians, a nephrologist, and a patient representative used the scenarios to create consensus recommendations.
Among the recommendations, for example, on the use of allopurinol is to start at a low dose of 100 mg/day (instead of the common practice of starting with 300 mg/day), or even lower for patients with chronic kidney disease, and then gradually titrate upward every 2-5 weeks. That recommendation supports previous statements from the Food and Drug Administration and the European League Against Rheumatism.
Also, allopurinol therapy should be actively managed and patients followed to make sure the uric acid target is achieved. "You can’t just give a prescription and say your job is done," though some recent studies suggest that many physicians do just that, Dr. FitzGerald said. "The corollary would be if someone gave blood pressure medication and then didn’t follow the patient’s blood pressure. That wouldn’t be seen as good medicine."
Maintenance doses of allopurinol to prevent acute gout attacks can exceed 300 mg even in patients with chronic kidney disease provided there is adequate patient education and monitoring.
A new recommendation drops the starting dose of oral colchicine for acute gout attacks to a loading dose of 1.2 mg, followed by 0.6 mg an hour later, and then starting prophylaxis 12 hours later at dosing of 0.6 mg once or twice daily.
"We used to give up to eight tablets a day," Dr. FitzGerald said. "That is dropped down to three to four tablets at the start of an attacks, because of findings that more colchicine didn’t really help outcomes" and that smaller doses are safer. The authors called this recommendation from ACR "a paradigm shift" that’s in accordance with Food and Drug Administration-approved label language.
Other highlights of the new ACR recommendations include sections on screening for HLA-B*5801 in patients at high risk of severe adverse reaction to allopurinol, combination therapy when target urate levels are not achieved, medication options including new drugs, and more.
Although the reports are titled "Guidelines," the text makes clear that they are expert recommendations and that clinicians are expected to take active roles in choosing the best management strategies for their particular patients. The authors were "very concerned" that the guidelines not be used by third-party payers to restrict access to medications or to promote one drug over another if there isn’t clear evidence to support it, Dr. FitzGerald said.
The methodology of the project precluded evaluations of costs and cost effectiveness, instead focusing on efficacy. So, for example, the guidelines say that allopurinol and febuxostat can be used equivalently in some circumstances, but clinicians need to consider all other aspects of these options including cost, patient preference, and more.
The ACR plans to update the guidelines as new data become available. The task force panel did create specific indications for use of imaging studies because results should be available in the next few years from studies on the use of high-resolution ultrasound and dual-energy CT for patients with gout.
In the United States, gout affects an estimated 4% of adults – more than 8 million people.
"I’m most excited and hopeful about trying to get this out to internal medicine and family practice doctors," Dr. FitzGerald said. "They see more gout than rheumatologists."
Dr. FitzGerald reported having no financial disclosures. Some members of the task force reported financial associations with multiple pharmaceutical companies but, by design, a majority of task force members had no perceived potential conflicts of interest.
The first guidelines on the management of gout from the American College of Rheumatology recommend new ways of using old drugs and changes in prophylaxis strategies, among other things.
The two-part guidelines, published online Sept. 28, should help speed up effective treatment of gout and get physicians to treat patients to a target urate level of less than 6 mg/dL in order to improve symptoms, Dr. John D. FitzGerald said in an interview.
"There has been a fair amount of recent movement on gout medications" including new alternatives to allopurinol and colchicine and new data on how to use those traditional drugs in safer ways, said Dr. FitzGerald, acting chief of the rheumatology division at the University of California, Los Angeles. "It’s a fair number of changes for medications that people had been using for decades."
The documents update previous guidelines from medical organizations in Europe, the Netherlands, and Japan. The new guidelines will be published in October 2012 by the journal Arthritis Care & Research.
Part 1 of the American College of Rheumatology (ACR) guidelines covers nonpharmacologic and pharmacologic approaches to managing hyperuricemia (Arthritis Care Res. 2012;64:1431-46 [doi:10.1002/acr.21772]).
Part 2 addresses prophylaxis and treatment for acute gouty arthritis (Arthritis Care Res. 2012;64:1447-61 [doi.wiley.com/10.1002/acr.21773]).
Dr. FitzGerald and two other co-leaders of the project, Dr. Dinesh Khanna of the University of Michigan, Ann Arbor and Dr. Robert Terkeltaub of the University of California, San Diego, reviewed the medical literature on gout from the 1950s to the present and drew up nine clinical case scenarios commonly seen in practice. A task force panel comprising seven rheumatologists, two primary care physicians, a nephrologist, and a patient representative used the scenarios to create consensus recommendations.
Among the recommendations, for example, on the use of allopurinol is to start at a low dose of 100 mg/day (instead of the common practice of starting with 300 mg/day), or even lower for patients with chronic kidney disease, and then gradually titrate upward every 2-5 weeks. That recommendation supports previous statements from the Food and Drug Administration and the European League Against Rheumatism.
Also, allopurinol therapy should be actively managed and patients followed to make sure the uric acid target is achieved. "You can’t just give a prescription and say your job is done," though some recent studies suggest that many physicians do just that, Dr. FitzGerald said. "The corollary would be if someone gave blood pressure medication and then didn’t follow the patient’s blood pressure. That wouldn’t be seen as good medicine."
Maintenance doses of allopurinol to prevent acute gout attacks can exceed 300 mg even in patients with chronic kidney disease provided there is adequate patient education and monitoring.
A new recommendation drops the starting dose of oral colchicine for acute gout attacks to a loading dose of 1.2 mg, followed by 0.6 mg an hour later, and then starting prophylaxis 12 hours later at dosing of 0.6 mg once or twice daily.
"We used to give up to eight tablets a day," Dr. FitzGerald said. "That is dropped down to three to four tablets at the start of an attacks, because of findings that more colchicine didn’t really help outcomes" and that smaller doses are safer. The authors called this recommendation from ACR "a paradigm shift" that’s in accordance with Food and Drug Administration-approved label language.
Other highlights of the new ACR recommendations include sections on screening for HLA-B*5801 in patients at high risk of severe adverse reaction to allopurinol, combination therapy when target urate levels are not achieved, medication options including new drugs, and more.
Although the reports are titled "Guidelines," the text makes clear that they are expert recommendations and that clinicians are expected to take active roles in choosing the best management strategies for their particular patients. The authors were "very concerned" that the guidelines not be used by third-party payers to restrict access to medications or to promote one drug over another if there isn’t clear evidence to support it, Dr. FitzGerald said.
The methodology of the project precluded evaluations of costs and cost effectiveness, instead focusing on efficacy. So, for example, the guidelines say that allopurinol and febuxostat can be used equivalently in some circumstances, but clinicians need to consider all other aspects of these options including cost, patient preference, and more.
The ACR plans to update the guidelines as new data become available. The task force panel did create specific indications for use of imaging studies because results should be available in the next few years from studies on the use of high-resolution ultrasound and dual-energy CT for patients with gout.
In the United States, gout affects an estimated 4% of adults – more than 8 million people.
"I’m most excited and hopeful about trying to get this out to internal medicine and family practice doctors," Dr. FitzGerald said. "They see more gout than rheumatologists."
Dr. FitzGerald reported having no financial disclosures. Some members of the task force reported financial associations with multiple pharmaceutical companies but, by design, a majority of task force members had no perceived potential conflicts of interest.
Childhood Problem Flares at Age 50
ANSWER
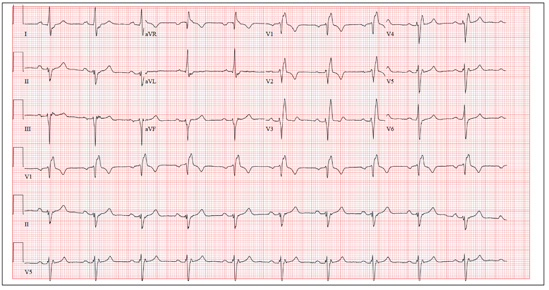
The correct interpretation includes normal sinus rhythm, right bundle branch block, and left anterior fascicular block. Normal sinus rhythm is evidenced by a rate between 60 and 100 beats/min, with a corresponding P for every QRS and a QRS for every P.
Right bundle branch block is evidenced by a QRS duration > 120 ms, a terminal broad S wave in lead I, and an RSR’ complex in lead V1. Left anterior fascicular block is evident from the finding that the S waves are greater than the R waves in leads II, III, and aVF.
The presence of a right ventricular block and left anterior fascicular block (bifascicular block) is consistent with a history of a VSD and/or surgical repair. The right and left bundles proceed from the atrioventricular node and bundle of His down the ventricular septum to the Purkinje fibers in the distal ventricular myocardium. Therefore, congenital anomalies of the ventricular septum, and/or surgical intervention within it, often affect conduction of the right and/or left bundle.
This patient’s symptoms were a result of his dilated aorta, and he underwent successful repair, with resolution of his symptoms.
ANSWER
The correct interpretation includes normal sinus rhythm, right bundle branch block, and left anterior fascicular block. Normal sinus rhythm is evidenced by a rate between 60 and 100 beats/min, with a corresponding P for every QRS and a QRS for every P.
Right bundle branch block is evidenced by a QRS duration > 120 ms, a terminal broad S wave in lead I, and an RSR’ complex in lead V1. Left anterior fascicular block is evident from the finding that the S waves are greater than the R waves in leads II, III, and aVF.
The presence of a right ventricular block and left anterior fascicular block (bifascicular block) is consistent with a history of a VSD and/or surgical repair. The right and left bundles proceed from the atrioventricular node and bundle of His down the ventricular septum to the Purkinje fibers in the distal ventricular myocardium. Therefore, congenital anomalies of the ventricular septum, and/or surgical intervention within it, often affect conduction of the right and/or left bundle.
This patient’s symptoms were a result of his dilated aorta, and he underwent successful repair, with resolution of his symptoms.
ANSWER
The correct interpretation includes normal sinus rhythm, right bundle branch block, and left anterior fascicular block. Normal sinus rhythm is evidenced by a rate between 60 and 100 beats/min, with a corresponding P for every QRS and a QRS for every P.
Right bundle branch block is evidenced by a QRS duration > 120 ms, a terminal broad S wave in lead I, and an RSR’ complex in lead V1. Left anterior fascicular block is evident from the finding that the S waves are greater than the R waves in leads II, III, and aVF.
The presence of a right ventricular block and left anterior fascicular block (bifascicular block) is consistent with a history of a VSD and/or surgical repair. The right and left bundles proceed from the atrioventricular node and bundle of His down the ventricular septum to the Purkinje fibers in the distal ventricular myocardium. Therefore, congenital anomalies of the ventricular septum, and/or surgical intervention within it, often affect conduction of the right and/or left bundle.
This patient’s symptoms were a result of his dilated aorta, and he underwent successful repair, with resolution of his symptoms.
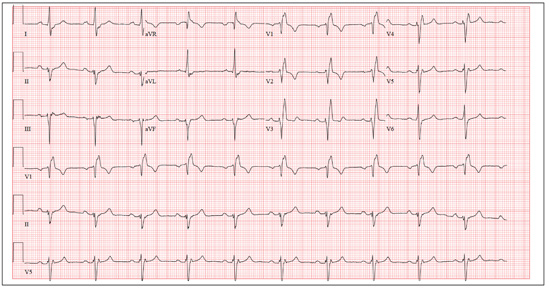
A man, 50, has a history of tetralogy of Fallot (ventricular septal defect [VSD], pulmonary stenosis, right ventricular hypertrophy, and overriding aorta). He underwent surgical correction at age 4, with placement of a Blalock-Taussig shunt and closure of his VSD, and was asymptomatic until one year ago. In the past year, he has developed progressive shortness of breath and dyspnea on exertion. In the past three months, he has developed chest pain that he describes as sharp, nonradiating, and occurring most often with dyspnea on exertion. He denies syncope, near-syncope, palpitations, or tachycardia. He cannot walk more than one-and-a-half blocks before stopping to rest, and he avoids hills and stairs if at all possible. A review of his most recent cardiac work-up (performed six months ago) reveals no significant coronary artery disease or evidence of aortic stenosis; it shows moderate aortic regurgitation, normal systolic aortic pressures, and normal left ventricular end diastolic pressures. The right ventricular pressures were elevated due to pulmonic stenosis; however, the estimated pulmonary artery pressures were normal. A cardiac MRI performed one month ago shows a significantly dilated aortic root with aneurysmal dilatation extending to the aortic arch, with effacement at the sinotubular junction and moderate aortic regurgitation. Additional findings include a markedly dilated right ventricular outflow tract with no pulmonic stenosis, evi-dence of a previous right Blalock-Taussig shunt, and moderate right atrial enlargement. Medical history is remarkable for hypertension. Family history is remarkable for hypertension, diabetes, and coronary artery disease, but not congenital heart disease. The patient does not smoke and drinks socially on the weekends. His medications include amlodipine, aspirin, and lisinopril. He is allergic to penicillin and amox-icillin. A review of systems reveals that he has had flulike symptoms for the past four days, with a dry, nonproductive cough. Physical exam reveals a well-developed, obese male in no distress. His height is 67”and his weight, 208 lb. Blood pressure is 102/70 mm Hg; pulse, 70 beats/min; respiratory rate, 16 breaths/min-1; and temperature, 98.4°F. His oxygen saturation is 98% on room air. Pertinent physical findings include a grade II/VI holosystolic murmur and a grade III/VI diastolic murmur, with a prominent S2 best heard at the left lower sternal border. There is no jugular venous distention, no peripheral edema, and no abnormal pulmonary finding. An ECG previously ordered for today’s visit reveals the following: a ventricular rate of 63 beats/min; PR interval, 196 ms; QRS duration, 174 ms; QT/QTc inter-val, 460/470 ms; P axis, 34°; R axis, –67°; and T axis, 56°. What is your interpretation of this ECG? How does the patient’s history predict the findings?
Wife is Worried That Her Husband's Condition is Contagious
ANSWER
The correct answer is petaloid seborrheic dermatitis (choice “d”), named for the flowerlike appearance of its polycyclic borders. Psoriasis (choice “a”) can present in this area, but tends to be scalier and usually involves multiple areas (eg, elbows, knees, and nails).
Rashes like this patient’s are often termed yeast infection (choice “b”). However, while a commensal yeast (Pityrosporum) can play a role in its formation, it appears that seborrhea represents an idiosyncratic reaction to increased numbers of this organism, rather than an actual infection.
Bowen’s disease (choice “c”) is a superficial squamous cell carcinoma, usually caused by overexposure to sunlight. Its lesions will be fixed, slowly growing larger with time, while seborrheic dermatitis will typically come and go. Biopsy is sometimes necessary to distinguish one from the other.
DISCUSSION
Seborrheic dermatitis (SD, aka seborrhea) is common, affecting up to 5% of the population. Dandruff is its usual manifestation, but it affects numerous other areas (as in this case), including the axillae, groin, beard, and genitals.
Presenting with scaling on an erythematous base, SD often flares and remits with the season (especially winter), with stress, and with increases in alcohol intake. Although it is usually mild, some cases can be severe. SD is associated with or accentuated by several other conditions, including Parkinson’s, stroke, and HIV. Severe SD in infants raises the possibility of Langerhans cell histiocytosis, especially when the presentation is atypical.
The diagnosis of SD can be difficult when it appears elsewhere than the scalp and face (eg, as an axillary or genital rash). Likewise, sternal petaloid SD is mystifying, unless other corroboratory manifestations are sought and found.
A few patients show signs of SD and psoriasis such that a definitive diagnosis cannot be made. Such overlap cases are sometimes termed sebopsoriasis. But psoriasis will usually exhibit signs not seen with SD, such as pitting of the nails, involvement of extensor surfaces of elbows and knees, and characteristic signs of psoriatic arthropathy in about 20% of cases. Pinpoint bleeding caused by peeling away scale, called the Auspitz sign, is seen with psoriasis and not with SD.
TREATMENT
This patient’s chest involvement responded rapidly to topical betamethasone foam, quickly tapered to avoid thinning the skin. Less powerful steroid creams, lotions, or gels (eg, triamcinolone 0.025%) can be used on other areas, such as ears and face. The daily use of an OTC dandruff shampoo (containing selenium sulfide, zinc pyrithione, tar, or ketoconazole) is an effective approach to controlling scalp involvement, but the product should be changed weekly.
Once the initial inflammation is controlled, topical antiyeast/antifungal preparations (eg, ketoconazole cream or any of the imidazoles, such as clotrimazole or oxiconazole) can be useful.
Finally, emphasis must be placed on educating the patient to expect control of the condition but not a cure.
ANSWER
The correct answer is petaloid seborrheic dermatitis (choice “d”), named for the flowerlike appearance of its polycyclic borders. Psoriasis (choice “a”) can present in this area, but tends to be scalier and usually involves multiple areas (eg, elbows, knees, and nails).
Rashes like this patient’s are often termed yeast infection (choice “b”). However, while a commensal yeast (Pityrosporum) can play a role in its formation, it appears that seborrhea represents an idiosyncratic reaction to increased numbers of this organism, rather than an actual infection.
Bowen’s disease (choice “c”) is a superficial squamous cell carcinoma, usually caused by overexposure to sunlight. Its lesions will be fixed, slowly growing larger with time, while seborrheic dermatitis will typically come and go. Biopsy is sometimes necessary to distinguish one from the other.
DISCUSSION
Seborrheic dermatitis (SD, aka seborrhea) is common, affecting up to 5% of the population. Dandruff is its usual manifestation, but it affects numerous other areas (as in this case), including the axillae, groin, beard, and genitals.
Presenting with scaling on an erythematous base, SD often flares and remits with the season (especially winter), with stress, and with increases in alcohol intake. Although it is usually mild, some cases can be severe. SD is associated with or accentuated by several other conditions, including Parkinson’s, stroke, and HIV. Severe SD in infants raises the possibility of Langerhans cell histiocytosis, especially when the presentation is atypical.
The diagnosis of SD can be difficult when it appears elsewhere than the scalp and face (eg, as an axillary or genital rash). Likewise, sternal petaloid SD is mystifying, unless other corroboratory manifestations are sought and found.
A few patients show signs of SD and psoriasis such that a definitive diagnosis cannot be made. Such overlap cases are sometimes termed sebopsoriasis. But psoriasis will usually exhibit signs not seen with SD, such as pitting of the nails, involvement of extensor surfaces of elbows and knees, and characteristic signs of psoriatic arthropathy in about 20% of cases. Pinpoint bleeding caused by peeling away scale, called the Auspitz sign, is seen with psoriasis and not with SD.
TREATMENT
This patient’s chest involvement responded rapidly to topical betamethasone foam, quickly tapered to avoid thinning the skin. Less powerful steroid creams, lotions, or gels (eg, triamcinolone 0.025%) can be used on other areas, such as ears and face. The daily use of an OTC dandruff shampoo (containing selenium sulfide, zinc pyrithione, tar, or ketoconazole) is an effective approach to controlling scalp involvement, but the product should be changed weekly.
Once the initial inflammation is controlled, topical antiyeast/antifungal preparations (eg, ketoconazole cream or any of the imidazoles, such as clotrimazole or oxiconazole) can be useful.
Finally, emphasis must be placed on educating the patient to expect control of the condition but not a cure.
ANSWER
The correct answer is petaloid seborrheic dermatitis (choice “d”), named for the flowerlike appearance of its polycyclic borders. Psoriasis (choice “a”) can present in this area, but tends to be scalier and usually involves multiple areas (eg, elbows, knees, and nails).
Rashes like this patient’s are often termed yeast infection (choice “b”). However, while a commensal yeast (Pityrosporum) can play a role in its formation, it appears that seborrhea represents an idiosyncratic reaction to increased numbers of this organism, rather than an actual infection.
Bowen’s disease (choice “c”) is a superficial squamous cell carcinoma, usually caused by overexposure to sunlight. Its lesions will be fixed, slowly growing larger with time, while seborrheic dermatitis will typically come and go. Biopsy is sometimes necessary to distinguish one from the other.
DISCUSSION
Seborrheic dermatitis (SD, aka seborrhea) is common, affecting up to 5% of the population. Dandruff is its usual manifestation, but it affects numerous other areas (as in this case), including the axillae, groin, beard, and genitals.
Presenting with scaling on an erythematous base, SD often flares and remits with the season (especially winter), with stress, and with increases in alcohol intake. Although it is usually mild, some cases can be severe. SD is associated with or accentuated by several other conditions, including Parkinson’s, stroke, and HIV. Severe SD in infants raises the possibility of Langerhans cell histiocytosis, especially when the presentation is atypical.
The diagnosis of SD can be difficult when it appears elsewhere than the scalp and face (eg, as an axillary or genital rash). Likewise, sternal petaloid SD is mystifying, unless other corroboratory manifestations are sought and found.
A few patients show signs of SD and psoriasis such that a definitive diagnosis cannot be made. Such overlap cases are sometimes termed sebopsoriasis. But psoriasis will usually exhibit signs not seen with SD, such as pitting of the nails, involvement of extensor surfaces of elbows and knees, and characteristic signs of psoriatic arthropathy in about 20% of cases. Pinpoint bleeding caused by peeling away scale, called the Auspitz sign, is seen with psoriasis and not with SD.
TREATMENT
This patient’s chest involvement responded rapidly to topical betamethasone foam, quickly tapered to avoid thinning the skin. Less powerful steroid creams, lotions, or gels (eg, triamcinolone 0.025%) can be used on other areas, such as ears and face. The daily use of an OTC dandruff shampoo (containing selenium sulfide, zinc pyrithione, tar, or ketoconazole) is an effective approach to controlling scalp involvement, but the product should be changed weekly.
Once the initial inflammation is controlled, topical antiyeast/antifungal preparations (eg, ketoconazole cream or any of the imidazoles, such as clotrimazole or oxiconazole) can be useful.
Finally, emphasis must be placed on educating the patient to expect control of the condition but not a cure.
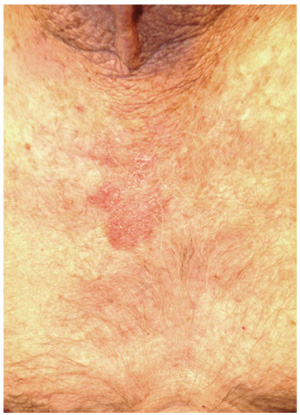
A 70-year-old man presents with a slightly itchy rash on his sternum that has appeared intermittently for years. Told it is “ringworm” by his primary care provider, the patient tried tolnaftate cream, to no avail. He is seeking additional consultation primarily because his wife is concerned she will catch the “infection.” The patient denies other skin problems, but then remembers that he has dandruff that flares from time to time, as well as a curious scaly red rash that “comes and goes” between his eyes, in his nasolabial folds, and behind his ears, especially in the winter. His father had similar problems. The patient is otherwise healthy, except for mild hypertension. The rash, located on the lower right sternum, measures about 6 cm at its largest dimension. Faintly pink, it has a papulosquamous surface, especially on its pol-ycyclic borders. Results of a KOH prep are negative for fungal elements. Elsewhere, a faintly scaly, orange-red rash is seen in the glabellar area and behind both ears. The man’s knees, elbows, and nails are free of any changes.
Proactive Rounding by RRT
Rapid response teams (RRT) have been promoted by numerous patient safety organizations to reduce preventable in‐hospital deaths.14 Initial studies of RRTs were promising,57 but recent literature,811 including systematic reviews and meta‐analyses, has called these findings into question. Nevertheless, RRTs remain popular in academic and community hospitals worldwide, and many have expanded their roles beyond solely responding to the deteriorating patient.12
Some RRTs, for example, proactively round on seriously ill ward patients and patients recently discharged from the intensive care unit (ICU) in an effort to prevent transitions to higher levels of care. Priestley and colleagues demonstrated that institution of such a team, referred to as a critical care outreach team (CCOT), decreased in‐hospital mortality while possibly increasing hospital length of stay (LOS).13 Three additional single‐center studies from the United Kingdom, where CCOTs are common, specifically examined proactive rounding by CCOTs on the ICU readmission rate: 2 observed no improvement,14, 15 while the third, limited by a small sample size, demonstrated a modest reduction in ICU readmissions.16
We sought to determine the impact of proactive rounding by an RRT on patients discharged from intensive care on the ICU readmission rate, ICU LOS, and in‐hospital mortality of patients discharged from the ICU. We hypothesized that proactive rounding by an RRT would decrease the ICU readmission rate, ICU LOS, and the in‐hospital mortality of patients discharged from the ICU.
MATERIALS AND METHODS
Site and Subjects
We carried out a retrospective, observational study of adult patients discharged from the ICU at University of California San Francisco (UCSF) Medical Center between January 2006 and June 2009. UCSF is a 790‐bed quaternary care academic hospital that admits approximately 17,000 patients annually and has 5 adult ICUs, with 62 beds and 3500 to 4000 ICU admissions annually. Our study was approved by the UCSF Medical Center Committee on Human Research; need for informed consent was waived.
Description of the RRT Before June 1, 2007
Throughout the study, the goal of the RRT was unchanged: to assess, triage, and institute early treatment in patients who experienced an acute decline in their clinical status. From November 2005 to October 2006, the RRT was staffed by an attending hospitalist and medicine resident during daytime, and by a critical care fellow at nighttime and on weekends. The RRT could be activated by any concerned staff member in response to a set of predetermined vital sign abnormalities, decreased urine output, or altered mental status, or simply if the staff member was concerned about the patient's clinical status. Despite extensive educational efforts, utilization of the team was low (2.7 calls per 1000 admissions) and, accordingly, it was discontinued in October 2006. After this time, staff would contact the primary team caring for the patient, should concerns regarding the patient's condition arise.
Description of the RRT After June 1, 2007
In an effort to expand its scope and utility, the RRT was reinstated on June 1, 2007 with a new composition and increased responsibilities. After this date, physician roles were eliminated, and the team composition changed to a dedicated critical care nurse and respiratory therapist, available 24 hours a day. Criteria for calling the team remained unchanged. In addition to responding to acute deteriorations in patients' clinical courses, the RRT began to proactively assess all patients within 12 hours of discharge from the ICU and would continue to round on these patients daily until it was felt that they were clinically stable. During these rounds, the RRT would provide consultation expertise to the bedside nurse and contact the patient's clinicians if concern existed about a patient's clinical trajectory; decisions to transfer a patient back to the ICU ultimately rested with the patient's primary team. During this time period, the RRT received an average of 110.6 calls per 1000 admissions.
Data Sources
Data collected included: demographics, clinical information (all patient refined [APR] severity of illness, APR risk of mortality, and the presence of 29 comorbidities), whether there was a readmission to the ICU, the total ICU LOS, and the vital status at the time of hospital discharge.
Outcomes
Outcomes included: readmission to the ICU, defined as 2 noncontiguous ICU stays during a single hospitalization; ICU LOS, defined as the total number of ICU days accrued during hospitalization; and in‐hospital mortality of patients discharged from the ICU.
Adjustment Variables
Patient age, gender, race, and ethnicity were available from administrative data. We used admission diagnosis code data to classify comorbidities using the method of Elixhauser et al.17
Statistical Analysis
For each of the 3 study outcomes, we assessed the effects of the intervention using multivariable models adjusting for patient‐ and service‐level factors, including a gamma model for ICU LOS and logistic models for ICU readmission and in‐hospital mortality of patients discharged from the ICU. We first compared unadjusted outcome levels before and after implementation. We then used an interrupted time series (ITS) framework to assess the effects of the intervention in terms of 5 measures: 1) the secular trend in the mean of the outcome before the intervention; 2) the change in the mean at the start of the implementation, or immediate effects; 3) the secular trend in the mean after implementation; 4) the change in secular trend, reflecting cumulative intervention effects; and 5) the net effect of the intervention, estimated as the adjusted difference between the fitted mean at the end of the postintervention period and the expected mean if the preintervention trend had continued without interruption or change.
Secondary Analyses
Given the heterogeneity of the RRT in the preintervention period, we assessed potential changes in trend at October 2006, the month in which the RRT was discontinued. We also examined changes in trend midway through the postimplementation period to evaluate for increased efficacy of the RRT with time.
Selection of Covariates
Age, race, and admitting service were included in both the prepost and ITS models by default for face validity. Additional covariates were selected for each outcome using backwards deletion with a retention criterion of P < 0.05, based on models that allowed the outcome rate to vary freely month to month. Because these data were obtained from administrative billing datasets, and the presence of comorbidities could not be definitively linked with time points during hospitalization, only those comorbidities that were likely present prior at ICU discharge were included. For similar reasons, APR severity of illness and risk of mortality scores, which were calculated from billing diagnoses at the end of hospitalization, were excluded from the models.
RESULTS
Patient Characteristics
During the study period, 11,687 patients were admitted to the ICU; 10,288 were discharged from the ICU alive and included in the analysis. In the 17 months prior to the introduction of proactive rounding by the RRT, 4902 (41.9%) patients were admitted, and during the 25 months afterwards, 6785 (58.1%) patients. Patients admitted in the 2 time periods were similar, although there were clinically small but statistically significant differences in race, APR severity of illness, APR risk of mortality, and certain comorbidities between the 2 groups (Table 1).
| Pre‐RRT (n = 4305) N (%) | Post‐RRT (n = 5983) N (%) | P Value | |
|---|---|---|---|
| |||
| Age, mean (y [SD]) | 57.7 [16.6] | 57.9 [16.5] | 0.50 |
| Female gender | 2,005 (46.6) | 2,824 (47.2) | 0.53 |
| Race | 0.0013 | ||
| White | 2,538 (59.0) | 3,520 (58.8) | |
| Black | 327 (7.6) | 436 (7.3) | |
| Asian | 642 (14.9) | 842 (14.1) | |
| Other | 719 (16.7) | 1,121 (18.7) | |
| Unknown | 79 (1.8) | 64 (1.1) | |
| Ethnicity | 0.87 | ||
| Hispanic | 480 (11.2) | 677 (11.3%) | |
| Non‐Hispanic | 3,547 (82.4) | 4,907 (82.0%) | |
| Unknown | 278 (6.5) | 399 (6.7) | |
| Insurance | 0.50 | ||
| Medicare | 1,788 (41.5) | 2,415 (40.4) | |
| Medicaid/Medi‐Cal | 699 (16.2) | 968 (16.2) | |
| Private | 1,642 (38.1) | 2,329 (38.9) | |
| Other | 176 (4.1) | 271 (4.5) | |
| Admission source | 0.41 | ||
| ED | 1,621 (37.7) | 2,244 (37.5) | |
| Outside hospital | 652 (15.2) | 855 (14.3) | |
| Direct admit | 2,032 (47.2) | 2,884 (48.2) | |
| Major surgery | 0.99 | ||
| Yes | 3,107 (72.2) | 4,319 (72.2) | |
| APR severity of illness | 0.0001 | ||
| Mild | 622 (14.5) | 828 (13.8) | |
| Moderate | 1,328 (30.9) | 1,626 (27.2) | |
| Major | 1,292 (30.0) | 1,908 (31.9) | |
| Extreme | 1,063 (24.7) | 1,621 (27.1) | |
| APR risk of mortality | 0.0109 | ||
| Mild | 1,422 (33.0) | 1,821 (30.4) | |
| Moderate | 1,074 (25.0) | 1,467 (24.5) | |
| Major | 947 (22.0) | 1,437 (24.0) | |
| Extreme | 862 (20.0) | 1,258 (21.0) | |
| Admitting service | 0.11 | ||
| Adult general surgery | 190 (4.4) | 260 (4.4) | |
| Cardiology | 347 (8.1) | 424 (7.1) | |
| Cardiothoracic surgery | 671 (15.6) | 930 (15.5) | |
| Kidney transplant surgery | 105 (2.4) | 112 (1.9) | |
| Liver transplant surgery | 298 (6.9) | 379 (6.3) | |
| Medicine | 683 (15.9) | 958 (16.0) | |
| Neurology | 420 (9.8) | 609 (10.2) | |
| Neurosurgery | 1,345 (31.2) | 1,995 (33.3) | |
| Vascular surgery | 246 (5.7) | 316 (5.3) | |
| Comorbidities | |||
| Hypertension | 2,054 (47.7) | 2,886 (48.2) | 0.60 |
| Fluid and electrolyte disorders | 998 (23.2) | 1,723 (28.8) | <0.0001 |
| Diabetes | 708 (16.5) | 880 (14.7) | 0.02 |
| Chronic obstructive pulmonary disease | 632 (14.7) | 849 (14.2) | 0.48 |
| Iron deficiency anemia | 582 (13.5) | 929 (15.5) | 0.005 |
| Renal failure | 541 (12.6) | 744 (12.4) | 0.84 |
| Coagulopathy | 418 (9.7) | 712 (11.9) | 0.0005 |
| Liver disease | 400 (9.3) | 553 (9.2) | 0.93 |
| Hypothyroidism | 330 (7.7) | 500 (8.4) | 0.20 |
| Depression | 306 (7.1) | 508 (8.5) | 0.01 |
| Peripheral vascular disease | 304 (7.1) | 422 (7.1) | 0.99 |
| Congestive heart failure | 263 (6.1) | 360 (6.0) | 0.85 |
| Weight loss | 236 (5.5) | 425 (7.1) | 0.0009 |
| Paralysis | 225 (5.2) | 328 (5.5) | 0.57 |
| Neurological disorders | 229 (5.3) | 276 (4.6) | 0.10 |
| Valvular disease | 210 (4.9) | 329 (5.5) | 0.16 |
| Drug abuse | 198 (4.6) | 268 (4.5) | 0.77 |
| Metastatic cancer | 198 (4.6) | 296 (5.0) | 0.42 |
| Obesity | 201 (4.7) | 306 (5.1) | 0.30 |
| Alcohol abuse | 178 (4.1) | 216 (3.6) | 0.17 |
| Diabetes with complications | 175 (4.1) | 218 (3.6) | 0.27 |
| Solid tumor without metastasis | 146 (3.4) | 245 (4.1) | 0.07 |
| Psychoses | 115 (2.7) | 183 (3.1) | 0.25 |
| Rheumatoid arthritis/collagen vascular disease | 96 (2.2) | 166 (2.8) | 0.08 |
| Pulmonary circulation disease | 83 (1.9) | 181 (3.0) | 0.0005 |
| Outcomes | |||
| Readmission to ICU | 288 (6.7) | 433 (7.3) | 0.24 |
| ICU length of stay, mean [SD] | 5.1 [9.7] | 4.9 [8.3] | 0.24 |
| In‐hospital mortality of patients discharged from the ICU | 260 (6.0) | 326 (5.5) | 0.24 |
ICU Readmission Rate
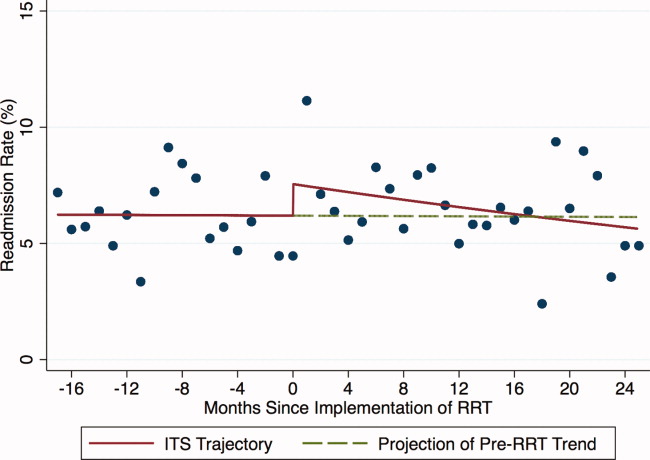
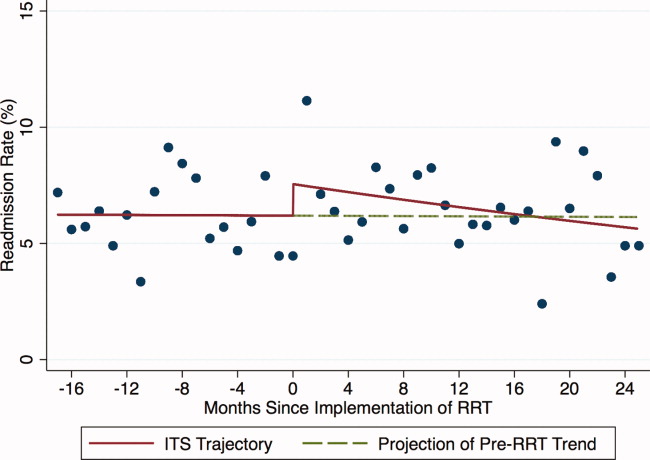
Introduction of proactive rounding by the RRT was not associated with unadjusted differences in the ICU readmission rate (6.7% preintervention vs 7.3% postintervention, P = 0.24; Table 1). In the adjusted ITS model, the intervention had no net effect on the odds of ICU readmission (adjusted odds ratio [AOR] for net intervention effect 0.98, 95% confidence interval [CI] 0.42, 2.28), with similar secular trends both preintervention (AOR 1.00 per year, 95% CI 0.97, 1.03), and afterwards (AOR 0.99 per year, 95% CI 0.98, 01.00), and a nonsignificant increase at implementation (Table 2). Figure 1 uses solid lines to show the fitted readmission rates, a hatched line to show the projection of the preintervention secular trend into the postintervention period, and circles to represent adjusted monthly means. The lack of a net intervention effect is indicated by the convergence of the solid and hatched lines 24 months postintervention.
| Outcome: Summary Effect Measure | Value (95% CI) | P Value |
|---|---|---|
| ||
| ICU readmission rateadjusted odds ratio | ||
| Pre‐RRT trend | 1.00 (0.97, 1.03) | 0.98 |
| Change at RRT implementation | 1.24 (0.94, 1.63) | 0.13 |
| Post‐RRT trend | 0.98 (0.97, 1.00) | 0.06 |
| Change in trend | 0.98 (0.96, 1.02) | 0.39 |
| Net intervention effect | 0.92 (0.40, 2.12) | 0.85 |
| ICU average length of stayadjusted ratio of means | ||
| Trend at 9 mo pre‐RRT | 0.98 (0.96, 1.00) | 0.05 |
| Trend at 3 mo pre‐RRT | 1.02 (0.99, 1.04) | 0.19 |
| Change in trend at 3 mo pre‐RRT | 1.03 (1.00, 1.07) | 0.07 |
| Change at RRT implementation | 0.92 (0.80, 1.06) | 0.27 |
| Post‐RRT trend | 1.00 (0.99, 1.00) | 0.35 |
| Change in trend at RRT implementation | 0.98 (0.96, 1.01) | 0.14 |
| Net intervention effect | 0.60 (0.31, 1.18) | 0.14 |
| In‐hospital mortality of patients discharged from the ICUadjusted odds ratio | ||
| Pre‐RRT trend | 1.02 (0.99, 1.06) | 0.15 |
| Change at RRT implementation | 0.74 (0.51, 1.08) | 0.12 |
| Post‐RRT trend | 1.00 (0.98, 1.01) | 0.68 |
| Change in trend | 0.97 (0.94, 1.01) | 0.14 |
| Net intervention effect | 0.39 (0.14, 1.10) | 0.08 |
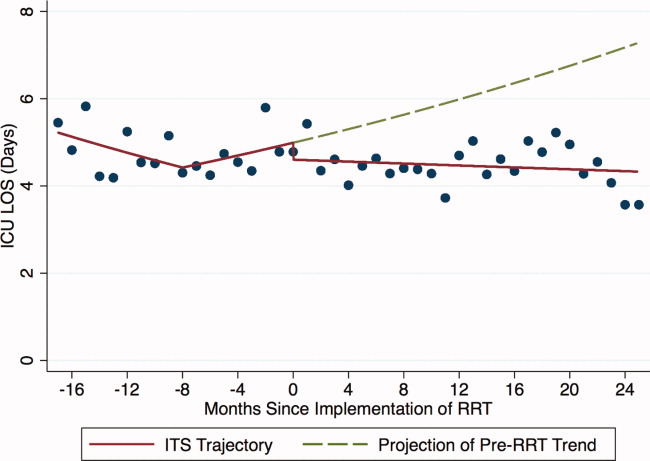
ICU Average LOS
Introduction of proactive rounding by the RRT was not associated with unadjusted differences in ICU average LOS (5.1 days preintervention vs 4.9 days postintervention, P = 0.24; Table 1). Trends in ICU LOS may have changed in October 2006 (P = 0.07), decreasing in the first half of the study period (adjusted rate ratio [ARR] 0.98 per year, 95% CI 0.961.00), but did not change significantly thereafter. As with the ICU readmission rate, neither the change in estimated secular trend after implementation (ARR 0.98, 95% CI 0.961.01), nor the net effect of the intervention (ARR 0.62, 95% CI 0.321.22) was statistically significant (Table 2); these results are depicted graphically in Figure 2.
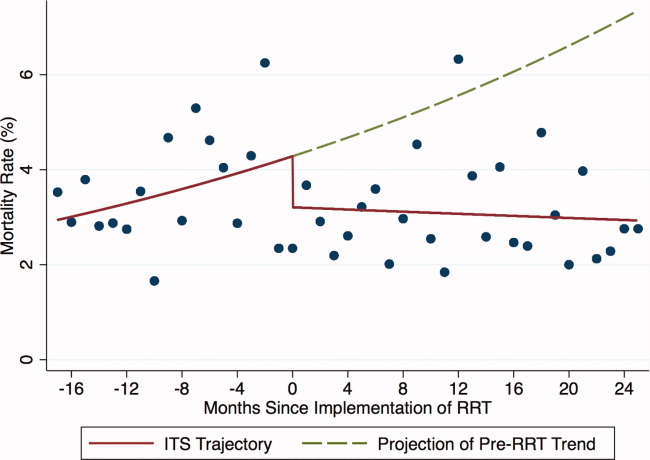
In‐Hospital Mortality of Patients Discharged From the ICU
Introduction of proactive rounding by the RRT was not associated with unadjusted differences in the mortality of patients discharged from the ICU (6.0% preintervention vs 5.5% postintervention, P = 0.24; Table 1). Similarly, in the adjusted ITS model, the intervention had no statistically significant net effect on the mortality outcome (Table 2 and Figure 3).
Secondary Analyses
Apart from weak evidence for a change in trend in ICU LOS in October 2006, no other changes in trend were found within the preintervention or postintervention periods (data not shown). This suggests that the heterogeneity of the preintervention RRT had no significant impact on the 3 outcomes examined, and that the RRT intervention failed to gain efficacy with time in the postintervention period. Additionally, we saw no outcome benefit in sensitivity analyses among all ICU patients or in service‐defined analyses (eg, surgical services), where ability to control for illness severity was improved.
DISCUSSION
In this single center study, introduction of an RRT that proactively rounded on patients discharged from the ICU did not reduce the ICU readmission rate, ICU LOS, or mortality of patients discharged from the ICU, after accounting for secular trends using robust ITS methods and adjusting for patient level factors.
Our study is consistent with 2 smaller studies that assessed the impact of proactive rounding by a CCOT on ICU readmission rate. Leary and Ridley14 found that proactively rounding by a CCOT did not reduce ICU readmissions or shorten the ICU LOS, although this study was limited by a surprisingly low ICU readmission rate and short ICU LOS prior to the intervention. Another study15 also observed no change in the ICU readmission rate following introduction of a proactively rounding CCOT but noted small reductions in both ICU and hospital mortality. The sole study showing an effect16 observed a lower ICU readmission rate and increased survival to hospital discharge (after excluding do not resuscitate [DNR] patients) with implementation of a CCOT, although some of their findings may be explained by their CCOT's use of palliative care services, a function not featured in our model.
Our study adds to the meta‐analyses and systematic reviews810 that have questioned the hypothesis that a trained and proactive team of caregivers should be able to prevent patients from returning to the ICU. Perhaps one reason why this is not true is that proactive rounding by RRTs may have minimal effect in systems where step‐down beds are readily available. At UCSF, nearly every patient transferred out of the ICU is triaged to a step‐down unit, where telemetry and pulse oximetry are continuously monitored. Despite this, however, our institution's 2 step‐down units generate more calls to our RRT than any other units in the hospital.
We were surprised to see that proactive rounding failed to shorten ICU LOS, hypothesizing that clinicians would be more comfortable discharging patients from the ICU knowing that the RRT would be closely monitoring them afterwards. Although we have no data to support this hypothesis, increased use of the RRT may have also increased step‐down bed use, as patients on the general medicalsurgical floors were transferred to a higher level of care upon recommendation of the RRT, thereby delaying transfers out of the ICU. Moreover, the opening of an additional 16‐bed ICU in October 2008 might have encouraged clinicians to transfer patients back to the ICU simply because beds were more easily accessible than before.
Introduction of proactive rounding by the RRT was also not associated with differences in the mortality rate of patients discharged from the ICU. This finding conflicts with the results of Garcea et al,15 Ball et al,16 and Priestley et al13, all of which found that implementation of a CCOT led to small but statistically significant reductions in in‐hospital mortality. All 3 of these studies, however, examined smaller patient populations (1380, 470, and 2903 patients, respectively), and both the Priestley and Ball studies13, 16 had significantly shorter periods of data collection (24 months and 32 weeks, respectively). Our results are based on models with confidence intervals and P values that account for variability in all 3 underlying effect estimates but assume a linear extrapolation of the preintervention trend. This approach allowed us to flexibly deal with changes related to the intervention, while relying on our large sample size to define time trends not dealt with adequately (or at all) in previous research.
The lack of improvement in outcomes cannot be attributed to immaturity of the RRT or failure of the clinical staff to use the RRT adequately. A prespecified secondary data analysis midway through the postintervention time period demonstrated that the RRT failed to gain efficacy with time with respect to all 3 outcomes. The postintervention RRT was also utilized far more frequently than its predecessor (110.6 vs 2.7 calls per 1000 admissions, respectively), and this degree of RRT utilization far surpasses the dose considered to be indicative of a mature RRT system.12
Our study has several limitations. First, we relied on administrative rather than chart‐collected data to determine the reason for ICU admission, and the APR severity of illness and risk of mortality scores. It seems unlikely, however, that coding deficiencies or biases affected the preintervention and postintervention patient populations differently. Even though we adjusted for all available measures, it is possible that we were not able to account for time trends in all potential confounders. Second, we did not have detailed clinical information on reasons for ICU readmission and whether readmissions occurred before or after the RRT proactively rounded on the patient. Therefore, potential readmissions to the ICU that might have been planned or which would have happened regardless of the presence of the RRT, such as for antibiotic desensitization, could not be accounted for. Third, introduction of proactive rounding by the RRT in June 2007 was accompanied by a change in the RRT's composition, from a physician‐led model to a nurse‐led model. Therefore, inherent differences in the way that physicians and nurses might assess and triage patients could not have been adjusted for. Lastly, this was a retrospective study conducted at a single academic medical center with a specific RRT model, and our results may not be directly applicable to nonteaching settings or to different RRT models.
Our findings raise further questions about the benefits of RRTs as they assume additional roles, such as proactive rounding on patients recently discharged from the ICU. The failure of our RRT to reduce the ICU readmission rate, the ICU average LOS, and the mortality of patients discharged from the ICU raises concerns that the benefits of our RRT are not commensurate with its cost. While defining the degree of impact and underlying mechanisms are worthy of prospective study, hospitals seeking to improve their RRT models should consider how to develop systems that achieve the RRT's promise in measurable ways.
Acknowledgements
The authors acknowledge Heather Leicester, MSPH, Senior Performance Improvement Analyst for Patient Safety and Quality Services at the University of California San Francisco for her work in data acquisition.
Disclosures: Dr Vittinghoff received salary support from an NIH grant during the time of this work for statistical consulting. He receives textbook royalties from Springer Verlag. Dr Auerbach was supported by 5K24HL098372‐02 from the National Heart Lung and Blood Institute during the period of this study although not specifically for this study; they had no role in the design or conduct of the study; the collection, analysis, or interpretation of the data; or the preparation, review, or approval of the manuscript. The other authors have no financial conflicts of interest.
- , , , . The 100,000 lives campaign: setting a goal and a deadline for improving health care quality. JAMA. 2006;295(3):324–327.
- Clinical Governance Unit, Quality and Safety Branch, Rural and Regional Health and Aged Care Services Division Safer Systems, Department of Human Services, State Government of Victoria. Safer Systems—Saving Lives Campaign. Available at: http://www.health.vic.gov.au/sssl. Accessed April 5, 2012.
- Canadian Patient Safety Institute. Safer Healthcare Now! Campaign. Available at: http://www.saferhealthcarenow.ca. Accessed April 5, 2012.
- , . The growth of rapid response systems. Jt Comm J Qual Patient Saf. 2008;34:489–495.
- , , , . The medical emergency team. Anaesth Intensive Care. 1995;23(2):183–186.
- , , , et al. Rates of in‐hospital arrests, deaths, and intensive care admission: the effect of a medical emergency team. Med J Aust. 2000;173:236–240.
- , , , , . The patient‐at‐risk team: identifying and managing seriously ill ward patients. Anaesthesia. 1999;54:853–860.
- , , , , . Effects of rapid response systems on clinical outcomes: systemic review and meta‐analysis. J Hosp Med. 2007;2:422–432.
- , , , , . Rapid response teams: a systemic review and meta‐analysis. Arch Intern Med. 2010;170(1):18–26.
- , , , , , . Rapid response systems: a systematic review. Crit Care Med. 2007;35(5):1238–1243.
- , , , et al. Introduction of the medical emergency team (MET) system: a cluster‐randomised controlled trial. Lancet. 2005;365:2091–2097.
- , , . Rapid response teams. N Engl J Med. 2011;365:139–146.
- , , , et al. Introducing critical care outreach: a ward‐randomised trial of phased introduction in a general hospital. Intensive Care Med. 2004;30(7):1398–1404.
- , . Impact of an outreach team on re‐admissions to a critical care unit. Anaesthesia. 2003;58:328–332.
- , , , , . Impact of a critical care outreach team on critical care readmissions and mortality. Acta Anaesthesiol Scand. 2004;48:1096–1100.
- , , . Effect of the critical care outreach team on patient survival to discharge from hospital and readmission to critical care: non‐randomised population based study. BMJ. 2003;327:1014–1017.
- , , , . Comorbidity measures for use with administrative data. Med Care. 1998;36(1):8–27.
Rapid response teams (RRT) have been promoted by numerous patient safety organizations to reduce preventable in‐hospital deaths.14 Initial studies of RRTs were promising,57 but recent literature,811 including systematic reviews and meta‐analyses, has called these findings into question. Nevertheless, RRTs remain popular in academic and community hospitals worldwide, and many have expanded their roles beyond solely responding to the deteriorating patient.12
Some RRTs, for example, proactively round on seriously ill ward patients and patients recently discharged from the intensive care unit (ICU) in an effort to prevent transitions to higher levels of care. Priestley and colleagues demonstrated that institution of such a team, referred to as a critical care outreach team (CCOT), decreased in‐hospital mortality while possibly increasing hospital length of stay (LOS).13 Three additional single‐center studies from the United Kingdom, where CCOTs are common, specifically examined proactive rounding by CCOTs on the ICU readmission rate: 2 observed no improvement,14, 15 while the third, limited by a small sample size, demonstrated a modest reduction in ICU readmissions.16
We sought to determine the impact of proactive rounding by an RRT on patients discharged from intensive care on the ICU readmission rate, ICU LOS, and in‐hospital mortality of patients discharged from the ICU. We hypothesized that proactive rounding by an RRT would decrease the ICU readmission rate, ICU LOS, and the in‐hospital mortality of patients discharged from the ICU.
MATERIALS AND METHODS
Site and Subjects
We carried out a retrospective, observational study of adult patients discharged from the ICU at University of California San Francisco (UCSF) Medical Center between January 2006 and June 2009. UCSF is a 790‐bed quaternary care academic hospital that admits approximately 17,000 patients annually and has 5 adult ICUs, with 62 beds and 3500 to 4000 ICU admissions annually. Our study was approved by the UCSF Medical Center Committee on Human Research; need for informed consent was waived.
Description of the RRT Before June 1, 2007
Throughout the study, the goal of the RRT was unchanged: to assess, triage, and institute early treatment in patients who experienced an acute decline in their clinical status. From November 2005 to October 2006, the RRT was staffed by an attending hospitalist and medicine resident during daytime, and by a critical care fellow at nighttime and on weekends. The RRT could be activated by any concerned staff member in response to a set of predetermined vital sign abnormalities, decreased urine output, or altered mental status, or simply if the staff member was concerned about the patient's clinical status. Despite extensive educational efforts, utilization of the team was low (2.7 calls per 1000 admissions) and, accordingly, it was discontinued in October 2006. After this time, staff would contact the primary team caring for the patient, should concerns regarding the patient's condition arise.
Description of the RRT After June 1, 2007
In an effort to expand its scope and utility, the RRT was reinstated on June 1, 2007 with a new composition and increased responsibilities. After this date, physician roles were eliminated, and the team composition changed to a dedicated critical care nurse and respiratory therapist, available 24 hours a day. Criteria for calling the team remained unchanged. In addition to responding to acute deteriorations in patients' clinical courses, the RRT began to proactively assess all patients within 12 hours of discharge from the ICU and would continue to round on these patients daily until it was felt that they were clinically stable. During these rounds, the RRT would provide consultation expertise to the bedside nurse and contact the patient's clinicians if concern existed about a patient's clinical trajectory; decisions to transfer a patient back to the ICU ultimately rested with the patient's primary team. During this time period, the RRT received an average of 110.6 calls per 1000 admissions.
Data Sources
Data collected included: demographics, clinical information (all patient refined [APR] severity of illness, APR risk of mortality, and the presence of 29 comorbidities), whether there was a readmission to the ICU, the total ICU LOS, and the vital status at the time of hospital discharge.
Outcomes
Outcomes included: readmission to the ICU, defined as 2 noncontiguous ICU stays during a single hospitalization; ICU LOS, defined as the total number of ICU days accrued during hospitalization; and in‐hospital mortality of patients discharged from the ICU.
Adjustment Variables
Patient age, gender, race, and ethnicity were available from administrative data. We used admission diagnosis code data to classify comorbidities using the method of Elixhauser et al.17
Statistical Analysis
For each of the 3 study outcomes, we assessed the effects of the intervention using multivariable models adjusting for patient‐ and service‐level factors, including a gamma model for ICU LOS and logistic models for ICU readmission and in‐hospital mortality of patients discharged from the ICU. We first compared unadjusted outcome levels before and after implementation. We then used an interrupted time series (ITS) framework to assess the effects of the intervention in terms of 5 measures: 1) the secular trend in the mean of the outcome before the intervention; 2) the change in the mean at the start of the implementation, or immediate effects; 3) the secular trend in the mean after implementation; 4) the change in secular trend, reflecting cumulative intervention effects; and 5) the net effect of the intervention, estimated as the adjusted difference between the fitted mean at the end of the postintervention period and the expected mean if the preintervention trend had continued without interruption or change.
Secondary Analyses
Given the heterogeneity of the RRT in the preintervention period, we assessed potential changes in trend at October 2006, the month in which the RRT was discontinued. We also examined changes in trend midway through the postimplementation period to evaluate for increased efficacy of the RRT with time.
Selection of Covariates
Age, race, and admitting service were included in both the prepost and ITS models by default for face validity. Additional covariates were selected for each outcome using backwards deletion with a retention criterion of P < 0.05, based on models that allowed the outcome rate to vary freely month to month. Because these data were obtained from administrative billing datasets, and the presence of comorbidities could not be definitively linked with time points during hospitalization, only those comorbidities that were likely present prior at ICU discharge were included. For similar reasons, APR severity of illness and risk of mortality scores, which were calculated from billing diagnoses at the end of hospitalization, were excluded from the models.
RESULTS
Patient Characteristics
During the study period, 11,687 patients were admitted to the ICU; 10,288 were discharged from the ICU alive and included in the analysis. In the 17 months prior to the introduction of proactive rounding by the RRT, 4902 (41.9%) patients were admitted, and during the 25 months afterwards, 6785 (58.1%) patients. Patients admitted in the 2 time periods were similar, although there were clinically small but statistically significant differences in race, APR severity of illness, APR risk of mortality, and certain comorbidities between the 2 groups (Table 1).
| Pre‐RRT (n = 4305) N (%) | Post‐RRT (n = 5983) N (%) | P Value | |
|---|---|---|---|
| |||
| Age, mean (y [SD]) | 57.7 [16.6] | 57.9 [16.5] | 0.50 |
| Female gender | 2,005 (46.6) | 2,824 (47.2) | 0.53 |
| Race | 0.0013 | ||
| White | 2,538 (59.0) | 3,520 (58.8) | |
| Black | 327 (7.6) | 436 (7.3) | |
| Asian | 642 (14.9) | 842 (14.1) | |
| Other | 719 (16.7) | 1,121 (18.7) | |
| Unknown | 79 (1.8) | 64 (1.1) | |
| Ethnicity | 0.87 | ||
| Hispanic | 480 (11.2) | 677 (11.3%) | |
| Non‐Hispanic | 3,547 (82.4) | 4,907 (82.0%) | |
| Unknown | 278 (6.5) | 399 (6.7) | |
| Insurance | 0.50 | ||
| Medicare | 1,788 (41.5) | 2,415 (40.4) | |
| Medicaid/Medi‐Cal | 699 (16.2) | 968 (16.2) | |
| Private | 1,642 (38.1) | 2,329 (38.9) | |
| Other | 176 (4.1) | 271 (4.5) | |
| Admission source | 0.41 | ||
| ED | 1,621 (37.7) | 2,244 (37.5) | |
| Outside hospital | 652 (15.2) | 855 (14.3) | |
| Direct admit | 2,032 (47.2) | 2,884 (48.2) | |
| Major surgery | 0.99 | ||
| Yes | 3,107 (72.2) | 4,319 (72.2) | |
| APR severity of illness | 0.0001 | ||
| Mild | 622 (14.5) | 828 (13.8) | |
| Moderate | 1,328 (30.9) | 1,626 (27.2) | |
| Major | 1,292 (30.0) | 1,908 (31.9) | |
| Extreme | 1,063 (24.7) | 1,621 (27.1) | |
| APR risk of mortality | 0.0109 | ||
| Mild | 1,422 (33.0) | 1,821 (30.4) | |
| Moderate | 1,074 (25.0) | 1,467 (24.5) | |
| Major | 947 (22.0) | 1,437 (24.0) | |
| Extreme | 862 (20.0) | 1,258 (21.0) | |
| Admitting service | 0.11 | ||
| Adult general surgery | 190 (4.4) | 260 (4.4) | |
| Cardiology | 347 (8.1) | 424 (7.1) | |
| Cardiothoracic surgery | 671 (15.6) | 930 (15.5) | |
| Kidney transplant surgery | 105 (2.4) | 112 (1.9) | |
| Liver transplant surgery | 298 (6.9) | 379 (6.3) | |
| Medicine | 683 (15.9) | 958 (16.0) | |
| Neurology | 420 (9.8) | 609 (10.2) | |
| Neurosurgery | 1,345 (31.2) | 1,995 (33.3) | |
| Vascular surgery | 246 (5.7) | 316 (5.3) | |
| Comorbidities | |||
| Hypertension | 2,054 (47.7) | 2,886 (48.2) | 0.60 |
| Fluid and electrolyte disorders | 998 (23.2) | 1,723 (28.8) | <0.0001 |
| Diabetes | 708 (16.5) | 880 (14.7) | 0.02 |
| Chronic obstructive pulmonary disease | 632 (14.7) | 849 (14.2) | 0.48 |
| Iron deficiency anemia | 582 (13.5) | 929 (15.5) | 0.005 |
| Renal failure | 541 (12.6) | 744 (12.4) | 0.84 |
| Coagulopathy | 418 (9.7) | 712 (11.9) | 0.0005 |
| Liver disease | 400 (9.3) | 553 (9.2) | 0.93 |
| Hypothyroidism | 330 (7.7) | 500 (8.4) | 0.20 |
| Depression | 306 (7.1) | 508 (8.5) | 0.01 |
| Peripheral vascular disease | 304 (7.1) | 422 (7.1) | 0.99 |
| Congestive heart failure | 263 (6.1) | 360 (6.0) | 0.85 |
| Weight loss | 236 (5.5) | 425 (7.1) | 0.0009 |
| Paralysis | 225 (5.2) | 328 (5.5) | 0.57 |
| Neurological disorders | 229 (5.3) | 276 (4.6) | 0.10 |
| Valvular disease | 210 (4.9) | 329 (5.5) | 0.16 |
| Drug abuse | 198 (4.6) | 268 (4.5) | 0.77 |
| Metastatic cancer | 198 (4.6) | 296 (5.0) | 0.42 |
| Obesity | 201 (4.7) | 306 (5.1) | 0.30 |
| Alcohol abuse | 178 (4.1) | 216 (3.6) | 0.17 |
| Diabetes with complications | 175 (4.1) | 218 (3.6) | 0.27 |
| Solid tumor without metastasis | 146 (3.4) | 245 (4.1) | 0.07 |
| Psychoses | 115 (2.7) | 183 (3.1) | 0.25 |
| Rheumatoid arthritis/collagen vascular disease | 96 (2.2) | 166 (2.8) | 0.08 |
| Pulmonary circulation disease | 83 (1.9) | 181 (3.0) | 0.0005 |
| Outcomes | |||
| Readmission to ICU | 288 (6.7) | 433 (7.3) | 0.24 |
| ICU length of stay, mean [SD] | 5.1 [9.7] | 4.9 [8.3] | 0.24 |
| In‐hospital mortality of patients discharged from the ICU | 260 (6.0) | 326 (5.5) | 0.24 |
ICU Readmission Rate
Introduction of proactive rounding by the RRT was not associated with unadjusted differences in the ICU readmission rate (6.7% preintervention vs 7.3% postintervention, P = 0.24; Table 1). In the adjusted ITS model, the intervention had no net effect on the odds of ICU readmission (adjusted odds ratio [AOR] for net intervention effect 0.98, 95% confidence interval [CI] 0.42, 2.28), with similar secular trends both preintervention (AOR 1.00 per year, 95% CI 0.97, 1.03), and afterwards (AOR 0.99 per year, 95% CI 0.98, 01.00), and a nonsignificant increase at implementation (Table 2). Figure 1 uses solid lines to show the fitted readmission rates, a hatched line to show the projection of the preintervention secular trend into the postintervention period, and circles to represent adjusted monthly means. The lack of a net intervention effect is indicated by the convergence of the solid and hatched lines 24 months postintervention.

| Outcome: Summary Effect Measure | Value (95% CI) | P Value |
|---|---|---|
| ||
| ICU readmission rateadjusted odds ratio | ||
| Pre‐RRT trend | 1.00 (0.97, 1.03) | 0.98 |
| Change at RRT implementation | 1.24 (0.94, 1.63) | 0.13 |
| Post‐RRT trend | 0.98 (0.97, 1.00) | 0.06 |
| Change in trend | 0.98 (0.96, 1.02) | 0.39 |
| Net intervention effect | 0.92 (0.40, 2.12) | 0.85 |
| ICU average length of stayadjusted ratio of means | ||
| Trend at 9 mo pre‐RRT | 0.98 (0.96, 1.00) | 0.05 |
| Trend at 3 mo pre‐RRT | 1.02 (0.99, 1.04) | 0.19 |
| Change in trend at 3 mo pre‐RRT | 1.03 (1.00, 1.07) | 0.07 |
| Change at RRT implementation | 0.92 (0.80, 1.06) | 0.27 |
| Post‐RRT trend | 1.00 (0.99, 1.00) | 0.35 |
| Change in trend at RRT implementation | 0.98 (0.96, 1.01) | 0.14 |
| Net intervention effect | 0.60 (0.31, 1.18) | 0.14 |
| In‐hospital mortality of patients discharged from the ICUadjusted odds ratio | ||
| Pre‐RRT trend | 1.02 (0.99, 1.06) | 0.15 |
| Change at RRT implementation | 0.74 (0.51, 1.08) | 0.12 |
| Post‐RRT trend | 1.00 (0.98, 1.01) | 0.68 |
| Change in trend | 0.97 (0.94, 1.01) | 0.14 |
| Net intervention effect | 0.39 (0.14, 1.10) | 0.08 |
ICU Average LOS
Introduction of proactive rounding by the RRT was not associated with unadjusted differences in ICU average LOS (5.1 days preintervention vs 4.9 days postintervention, P = 0.24; Table 1). Trends in ICU LOS may have changed in October 2006 (P = 0.07), decreasing in the first half of the study period (adjusted rate ratio [ARR] 0.98 per year, 95% CI 0.961.00), but did not change significantly thereafter. As with the ICU readmission rate, neither the change in estimated secular trend after implementation (ARR 0.98, 95% CI 0.961.01), nor the net effect of the intervention (ARR 0.62, 95% CI 0.321.22) was statistically significant (Table 2); these results are depicted graphically in Figure 2.

In‐Hospital Mortality of Patients Discharged From the ICU
Introduction of proactive rounding by the RRT was not associated with unadjusted differences in the mortality of patients discharged from the ICU (6.0% preintervention vs 5.5% postintervention, P = 0.24; Table 1). Similarly, in the adjusted ITS model, the intervention had no statistically significant net effect on the mortality outcome (Table 2 and Figure 3).

Secondary Analyses
Apart from weak evidence for a change in trend in ICU LOS in October 2006, no other changes in trend were found within the preintervention or postintervention periods (data not shown). This suggests that the heterogeneity of the preintervention RRT had no significant impact on the 3 outcomes examined, and that the RRT intervention failed to gain efficacy with time in the postintervention period. Additionally, we saw no outcome benefit in sensitivity analyses among all ICU patients or in service‐defined analyses (eg, surgical services), where ability to control for illness severity was improved.
DISCUSSION
In this single center study, introduction of an RRT that proactively rounded on patients discharged from the ICU did not reduce the ICU readmission rate, ICU LOS, or mortality of patients discharged from the ICU, after accounting for secular trends using robust ITS methods and adjusting for patient level factors.
Our study is consistent with 2 smaller studies that assessed the impact of proactive rounding by a CCOT on ICU readmission rate. Leary and Ridley14 found that proactively rounding by a CCOT did not reduce ICU readmissions or shorten the ICU LOS, although this study was limited by a surprisingly low ICU readmission rate and short ICU LOS prior to the intervention. Another study15 also observed no change in the ICU readmission rate following introduction of a proactively rounding CCOT but noted small reductions in both ICU and hospital mortality. The sole study showing an effect16 observed a lower ICU readmission rate and increased survival to hospital discharge (after excluding do not resuscitate [DNR] patients) with implementation of a CCOT, although some of their findings may be explained by their CCOT's use of palliative care services, a function not featured in our model.
Our study adds to the meta‐analyses and systematic reviews810 that have questioned the hypothesis that a trained and proactive team of caregivers should be able to prevent patients from returning to the ICU. Perhaps one reason why this is not true is that proactive rounding by RRTs may have minimal effect in systems where step‐down beds are readily available. At UCSF, nearly every patient transferred out of the ICU is triaged to a step‐down unit, where telemetry and pulse oximetry are continuously monitored. Despite this, however, our institution's 2 step‐down units generate more calls to our RRT than any other units in the hospital.
We were surprised to see that proactive rounding failed to shorten ICU LOS, hypothesizing that clinicians would be more comfortable discharging patients from the ICU knowing that the RRT would be closely monitoring them afterwards. Although we have no data to support this hypothesis, increased use of the RRT may have also increased step‐down bed use, as patients on the general medicalsurgical floors were transferred to a higher level of care upon recommendation of the RRT, thereby delaying transfers out of the ICU. Moreover, the opening of an additional 16‐bed ICU in October 2008 might have encouraged clinicians to transfer patients back to the ICU simply because beds were more easily accessible than before.
Introduction of proactive rounding by the RRT was also not associated with differences in the mortality rate of patients discharged from the ICU. This finding conflicts with the results of Garcea et al,15 Ball et al,16 and Priestley et al13, all of which found that implementation of a CCOT led to small but statistically significant reductions in in‐hospital mortality. All 3 of these studies, however, examined smaller patient populations (1380, 470, and 2903 patients, respectively), and both the Priestley and Ball studies13, 16 had significantly shorter periods of data collection (24 months and 32 weeks, respectively). Our results are based on models with confidence intervals and P values that account for variability in all 3 underlying effect estimates but assume a linear extrapolation of the preintervention trend. This approach allowed us to flexibly deal with changes related to the intervention, while relying on our large sample size to define time trends not dealt with adequately (or at all) in previous research.
The lack of improvement in outcomes cannot be attributed to immaturity of the RRT or failure of the clinical staff to use the RRT adequately. A prespecified secondary data analysis midway through the postintervention time period demonstrated that the RRT failed to gain efficacy with time with respect to all 3 outcomes. The postintervention RRT was also utilized far more frequently than its predecessor (110.6 vs 2.7 calls per 1000 admissions, respectively), and this degree of RRT utilization far surpasses the dose considered to be indicative of a mature RRT system.12
Our study has several limitations. First, we relied on administrative rather than chart‐collected data to determine the reason for ICU admission, and the APR severity of illness and risk of mortality scores. It seems unlikely, however, that coding deficiencies or biases affected the preintervention and postintervention patient populations differently. Even though we adjusted for all available measures, it is possible that we were not able to account for time trends in all potential confounders. Second, we did not have detailed clinical information on reasons for ICU readmission and whether readmissions occurred before or after the RRT proactively rounded on the patient. Therefore, potential readmissions to the ICU that might have been planned or which would have happened regardless of the presence of the RRT, such as for antibiotic desensitization, could not be accounted for. Third, introduction of proactive rounding by the RRT in June 2007 was accompanied by a change in the RRT's composition, from a physician‐led model to a nurse‐led model. Therefore, inherent differences in the way that physicians and nurses might assess and triage patients could not have been adjusted for. Lastly, this was a retrospective study conducted at a single academic medical center with a specific RRT model, and our results may not be directly applicable to nonteaching settings or to different RRT models.
Our findings raise further questions about the benefits of RRTs as they assume additional roles, such as proactive rounding on patients recently discharged from the ICU. The failure of our RRT to reduce the ICU readmission rate, the ICU average LOS, and the mortality of patients discharged from the ICU raises concerns that the benefits of our RRT are not commensurate with its cost. While defining the degree of impact and underlying mechanisms are worthy of prospective study, hospitals seeking to improve their RRT models should consider how to develop systems that achieve the RRT's promise in measurable ways.
Acknowledgements
The authors acknowledge Heather Leicester, MSPH, Senior Performance Improvement Analyst for Patient Safety and Quality Services at the University of California San Francisco for her work in data acquisition.
Disclosures: Dr Vittinghoff received salary support from an NIH grant during the time of this work for statistical consulting. He receives textbook royalties from Springer Verlag. Dr Auerbach was supported by 5K24HL098372‐02 from the National Heart Lung and Blood Institute during the period of this study although not specifically for this study; they had no role in the design or conduct of the study; the collection, analysis, or interpretation of the data; or the preparation, review, or approval of the manuscript. The other authors have no financial conflicts of interest.
Rapid response teams (RRT) have been promoted by numerous patient safety organizations to reduce preventable in‐hospital deaths.14 Initial studies of RRTs were promising,57 but recent literature,811 including systematic reviews and meta‐analyses, has called these findings into question. Nevertheless, RRTs remain popular in academic and community hospitals worldwide, and many have expanded their roles beyond solely responding to the deteriorating patient.12
Some RRTs, for example, proactively round on seriously ill ward patients and patients recently discharged from the intensive care unit (ICU) in an effort to prevent transitions to higher levels of care. Priestley and colleagues demonstrated that institution of such a team, referred to as a critical care outreach team (CCOT), decreased in‐hospital mortality while possibly increasing hospital length of stay (LOS).13 Three additional single‐center studies from the United Kingdom, where CCOTs are common, specifically examined proactive rounding by CCOTs on the ICU readmission rate: 2 observed no improvement,14, 15 while the third, limited by a small sample size, demonstrated a modest reduction in ICU readmissions.16
We sought to determine the impact of proactive rounding by an RRT on patients discharged from intensive care on the ICU readmission rate, ICU LOS, and in‐hospital mortality of patients discharged from the ICU. We hypothesized that proactive rounding by an RRT would decrease the ICU readmission rate, ICU LOS, and the in‐hospital mortality of patients discharged from the ICU.
MATERIALS AND METHODS
Site and Subjects
We carried out a retrospective, observational study of adult patients discharged from the ICU at University of California San Francisco (UCSF) Medical Center between January 2006 and June 2009. UCSF is a 790‐bed quaternary care academic hospital that admits approximately 17,000 patients annually and has 5 adult ICUs, with 62 beds and 3500 to 4000 ICU admissions annually. Our study was approved by the UCSF Medical Center Committee on Human Research; need for informed consent was waived.
Description of the RRT Before June 1, 2007
Throughout the study, the goal of the RRT was unchanged: to assess, triage, and institute early treatment in patients who experienced an acute decline in their clinical status. From November 2005 to October 2006, the RRT was staffed by an attending hospitalist and medicine resident during daytime, and by a critical care fellow at nighttime and on weekends. The RRT could be activated by any concerned staff member in response to a set of predetermined vital sign abnormalities, decreased urine output, or altered mental status, or simply if the staff member was concerned about the patient's clinical status. Despite extensive educational efforts, utilization of the team was low (2.7 calls per 1000 admissions) and, accordingly, it was discontinued in October 2006. After this time, staff would contact the primary team caring for the patient, should concerns regarding the patient's condition arise.
Description of the RRT After June 1, 2007
In an effort to expand its scope and utility, the RRT was reinstated on June 1, 2007 with a new composition and increased responsibilities. After this date, physician roles were eliminated, and the team composition changed to a dedicated critical care nurse and respiratory therapist, available 24 hours a day. Criteria for calling the team remained unchanged. In addition to responding to acute deteriorations in patients' clinical courses, the RRT began to proactively assess all patients within 12 hours of discharge from the ICU and would continue to round on these patients daily until it was felt that they were clinically stable. During these rounds, the RRT would provide consultation expertise to the bedside nurse and contact the patient's clinicians if concern existed about a patient's clinical trajectory; decisions to transfer a patient back to the ICU ultimately rested with the patient's primary team. During this time period, the RRT received an average of 110.6 calls per 1000 admissions.
Data Sources
Data collected included: demographics, clinical information (all patient refined [APR] severity of illness, APR risk of mortality, and the presence of 29 comorbidities), whether there was a readmission to the ICU, the total ICU LOS, and the vital status at the time of hospital discharge.
Outcomes
Outcomes included: readmission to the ICU, defined as 2 noncontiguous ICU stays during a single hospitalization; ICU LOS, defined as the total number of ICU days accrued during hospitalization; and in‐hospital mortality of patients discharged from the ICU.
Adjustment Variables
Patient age, gender, race, and ethnicity were available from administrative data. We used admission diagnosis code data to classify comorbidities using the method of Elixhauser et al.17
Statistical Analysis
For each of the 3 study outcomes, we assessed the effects of the intervention using multivariable models adjusting for patient‐ and service‐level factors, including a gamma model for ICU LOS and logistic models for ICU readmission and in‐hospital mortality of patients discharged from the ICU. We first compared unadjusted outcome levels before and after implementation. We then used an interrupted time series (ITS) framework to assess the effects of the intervention in terms of 5 measures: 1) the secular trend in the mean of the outcome before the intervention; 2) the change in the mean at the start of the implementation, or immediate effects; 3) the secular trend in the mean after implementation; 4) the change in secular trend, reflecting cumulative intervention effects; and 5) the net effect of the intervention, estimated as the adjusted difference between the fitted mean at the end of the postintervention period and the expected mean if the preintervention trend had continued without interruption or change.
Secondary Analyses
Given the heterogeneity of the RRT in the preintervention period, we assessed potential changes in trend at October 2006, the month in which the RRT was discontinued. We also examined changes in trend midway through the postimplementation period to evaluate for increased efficacy of the RRT with time.
Selection of Covariates
Age, race, and admitting service were included in both the prepost and ITS models by default for face validity. Additional covariates were selected for each outcome using backwards deletion with a retention criterion of P < 0.05, based on models that allowed the outcome rate to vary freely month to month. Because these data were obtained from administrative billing datasets, and the presence of comorbidities could not be definitively linked with time points during hospitalization, only those comorbidities that were likely present prior at ICU discharge were included. For similar reasons, APR severity of illness and risk of mortality scores, which were calculated from billing diagnoses at the end of hospitalization, were excluded from the models.
RESULTS
Patient Characteristics
During the study period, 11,687 patients were admitted to the ICU; 10,288 were discharged from the ICU alive and included in the analysis. In the 17 months prior to the introduction of proactive rounding by the RRT, 4902 (41.9%) patients were admitted, and during the 25 months afterwards, 6785 (58.1%) patients. Patients admitted in the 2 time periods were similar, although there were clinically small but statistically significant differences in race, APR severity of illness, APR risk of mortality, and certain comorbidities between the 2 groups (Table 1).
| Pre‐RRT (n = 4305) N (%) | Post‐RRT (n = 5983) N (%) | P Value | |
|---|---|---|---|
| |||
| Age, mean (y [SD]) | 57.7 [16.6] | 57.9 [16.5] | 0.50 |
| Female gender | 2,005 (46.6) | 2,824 (47.2) | 0.53 |
| Race | 0.0013 | ||
| White | 2,538 (59.0) | 3,520 (58.8) | |
| Black | 327 (7.6) | 436 (7.3) | |
| Asian | 642 (14.9) | 842 (14.1) | |
| Other | 719 (16.7) | 1,121 (18.7) | |
| Unknown | 79 (1.8) | 64 (1.1) | |
| Ethnicity | 0.87 | ||
| Hispanic | 480 (11.2) | 677 (11.3%) | |
| Non‐Hispanic | 3,547 (82.4) | 4,907 (82.0%) | |
| Unknown | 278 (6.5) | 399 (6.7) | |
| Insurance | 0.50 | ||
| Medicare | 1,788 (41.5) | 2,415 (40.4) | |
| Medicaid/Medi‐Cal | 699 (16.2) | 968 (16.2) | |
| Private | 1,642 (38.1) | 2,329 (38.9) | |
| Other | 176 (4.1) | 271 (4.5) | |
| Admission source | 0.41 | ||
| ED | 1,621 (37.7) | 2,244 (37.5) | |
| Outside hospital | 652 (15.2) | 855 (14.3) | |
| Direct admit | 2,032 (47.2) | 2,884 (48.2) | |
| Major surgery | 0.99 | ||
| Yes | 3,107 (72.2) | 4,319 (72.2) | |
| APR severity of illness | 0.0001 | ||
| Mild | 622 (14.5) | 828 (13.8) | |
| Moderate | 1,328 (30.9) | 1,626 (27.2) | |
| Major | 1,292 (30.0) | 1,908 (31.9) | |
| Extreme | 1,063 (24.7) | 1,621 (27.1) | |
| APR risk of mortality | 0.0109 | ||
| Mild | 1,422 (33.0) | 1,821 (30.4) | |
| Moderate | 1,074 (25.0) | 1,467 (24.5) | |
| Major | 947 (22.0) | 1,437 (24.0) | |
| Extreme | 862 (20.0) | 1,258 (21.0) | |
| Admitting service | 0.11 | ||
| Adult general surgery | 190 (4.4) | 260 (4.4) | |
| Cardiology | 347 (8.1) | 424 (7.1) | |
| Cardiothoracic surgery | 671 (15.6) | 930 (15.5) | |
| Kidney transplant surgery | 105 (2.4) | 112 (1.9) | |
| Liver transplant surgery | 298 (6.9) | 379 (6.3) | |
| Medicine | 683 (15.9) | 958 (16.0) | |
| Neurology | 420 (9.8) | 609 (10.2) | |
| Neurosurgery | 1,345 (31.2) | 1,995 (33.3) | |
| Vascular surgery | 246 (5.7) | 316 (5.3) | |
| Comorbidities | |||
| Hypertension | 2,054 (47.7) | 2,886 (48.2) | 0.60 |
| Fluid and electrolyte disorders | 998 (23.2) | 1,723 (28.8) | <0.0001 |
| Diabetes | 708 (16.5) | 880 (14.7) | 0.02 |
| Chronic obstructive pulmonary disease | 632 (14.7) | 849 (14.2) | 0.48 |
| Iron deficiency anemia | 582 (13.5) | 929 (15.5) | 0.005 |
| Renal failure | 541 (12.6) | 744 (12.4) | 0.84 |
| Coagulopathy | 418 (9.7) | 712 (11.9) | 0.0005 |
| Liver disease | 400 (9.3) | 553 (9.2) | 0.93 |
| Hypothyroidism | 330 (7.7) | 500 (8.4) | 0.20 |
| Depression | 306 (7.1) | 508 (8.5) | 0.01 |
| Peripheral vascular disease | 304 (7.1) | 422 (7.1) | 0.99 |
| Congestive heart failure | 263 (6.1) | 360 (6.0) | 0.85 |
| Weight loss | 236 (5.5) | 425 (7.1) | 0.0009 |
| Paralysis | 225 (5.2) | 328 (5.5) | 0.57 |
| Neurological disorders | 229 (5.3) | 276 (4.6) | 0.10 |
| Valvular disease | 210 (4.9) | 329 (5.5) | 0.16 |
| Drug abuse | 198 (4.6) | 268 (4.5) | 0.77 |
| Metastatic cancer | 198 (4.6) | 296 (5.0) | 0.42 |
| Obesity | 201 (4.7) | 306 (5.1) | 0.30 |
| Alcohol abuse | 178 (4.1) | 216 (3.6) | 0.17 |
| Diabetes with complications | 175 (4.1) | 218 (3.6) | 0.27 |
| Solid tumor without metastasis | 146 (3.4) | 245 (4.1) | 0.07 |
| Psychoses | 115 (2.7) | 183 (3.1) | 0.25 |
| Rheumatoid arthritis/collagen vascular disease | 96 (2.2) | 166 (2.8) | 0.08 |
| Pulmonary circulation disease | 83 (1.9) | 181 (3.0) | 0.0005 |
| Outcomes | |||
| Readmission to ICU | 288 (6.7) | 433 (7.3) | 0.24 |
| ICU length of stay, mean [SD] | 5.1 [9.7] | 4.9 [8.3] | 0.24 |
| In‐hospital mortality of patients discharged from the ICU | 260 (6.0) | 326 (5.5) | 0.24 |
ICU Readmission Rate
Introduction of proactive rounding by the RRT was not associated with unadjusted differences in the ICU readmission rate (6.7% preintervention vs 7.3% postintervention, P = 0.24; Table 1). In the adjusted ITS model, the intervention had no net effect on the odds of ICU readmission (adjusted odds ratio [AOR] for net intervention effect 0.98, 95% confidence interval [CI] 0.42, 2.28), with similar secular trends both preintervention (AOR 1.00 per year, 95% CI 0.97, 1.03), and afterwards (AOR 0.99 per year, 95% CI 0.98, 01.00), and a nonsignificant increase at implementation (Table 2). Figure 1 uses solid lines to show the fitted readmission rates, a hatched line to show the projection of the preintervention secular trend into the postintervention period, and circles to represent adjusted monthly means. The lack of a net intervention effect is indicated by the convergence of the solid and hatched lines 24 months postintervention.

| Outcome: Summary Effect Measure | Value (95% CI) | P Value |
|---|---|---|
| ||
| ICU readmission rateadjusted odds ratio | ||
| Pre‐RRT trend | 1.00 (0.97, 1.03) | 0.98 |
| Change at RRT implementation | 1.24 (0.94, 1.63) | 0.13 |
| Post‐RRT trend | 0.98 (0.97, 1.00) | 0.06 |
| Change in trend | 0.98 (0.96, 1.02) | 0.39 |
| Net intervention effect | 0.92 (0.40, 2.12) | 0.85 |
| ICU average length of stayadjusted ratio of means | ||
| Trend at 9 mo pre‐RRT | 0.98 (0.96, 1.00) | 0.05 |
| Trend at 3 mo pre‐RRT | 1.02 (0.99, 1.04) | 0.19 |
| Change in trend at 3 mo pre‐RRT | 1.03 (1.00, 1.07) | 0.07 |
| Change at RRT implementation | 0.92 (0.80, 1.06) | 0.27 |
| Post‐RRT trend | 1.00 (0.99, 1.00) | 0.35 |
| Change in trend at RRT implementation | 0.98 (0.96, 1.01) | 0.14 |
| Net intervention effect | 0.60 (0.31, 1.18) | 0.14 |
| In‐hospital mortality of patients discharged from the ICUadjusted odds ratio | ||
| Pre‐RRT trend | 1.02 (0.99, 1.06) | 0.15 |
| Change at RRT implementation | 0.74 (0.51, 1.08) | 0.12 |
| Post‐RRT trend | 1.00 (0.98, 1.01) | 0.68 |
| Change in trend | 0.97 (0.94, 1.01) | 0.14 |
| Net intervention effect | 0.39 (0.14, 1.10) | 0.08 |
ICU Average LOS
Introduction of proactive rounding by the RRT was not associated with unadjusted differences in ICU average LOS (5.1 days preintervention vs 4.9 days postintervention, P = 0.24; Table 1). Trends in ICU LOS may have changed in October 2006 (P = 0.07), decreasing in the first half of the study period (adjusted rate ratio [ARR] 0.98 per year, 95% CI 0.961.00), but did not change significantly thereafter. As with the ICU readmission rate, neither the change in estimated secular trend after implementation (ARR 0.98, 95% CI 0.961.01), nor the net effect of the intervention (ARR 0.62, 95% CI 0.321.22) was statistically significant (Table 2); these results are depicted graphically in Figure 2.

In‐Hospital Mortality of Patients Discharged From the ICU
Introduction of proactive rounding by the RRT was not associated with unadjusted differences in the mortality of patients discharged from the ICU (6.0% preintervention vs 5.5% postintervention, P = 0.24; Table 1). Similarly, in the adjusted ITS model, the intervention had no statistically significant net effect on the mortality outcome (Table 2 and Figure 3).

Secondary Analyses
Apart from weak evidence for a change in trend in ICU LOS in October 2006, no other changes in trend were found within the preintervention or postintervention periods (data not shown). This suggests that the heterogeneity of the preintervention RRT had no significant impact on the 3 outcomes examined, and that the RRT intervention failed to gain efficacy with time in the postintervention period. Additionally, we saw no outcome benefit in sensitivity analyses among all ICU patients or in service‐defined analyses (eg, surgical services), where ability to control for illness severity was improved.
DISCUSSION
In this single center study, introduction of an RRT that proactively rounded on patients discharged from the ICU did not reduce the ICU readmission rate, ICU LOS, or mortality of patients discharged from the ICU, after accounting for secular trends using robust ITS methods and adjusting for patient level factors.
Our study is consistent with 2 smaller studies that assessed the impact of proactive rounding by a CCOT on ICU readmission rate. Leary and Ridley14 found that proactively rounding by a CCOT did not reduce ICU readmissions or shorten the ICU LOS, although this study was limited by a surprisingly low ICU readmission rate and short ICU LOS prior to the intervention. Another study15 also observed no change in the ICU readmission rate following introduction of a proactively rounding CCOT but noted small reductions in both ICU and hospital mortality. The sole study showing an effect16 observed a lower ICU readmission rate and increased survival to hospital discharge (after excluding do not resuscitate [DNR] patients) with implementation of a CCOT, although some of their findings may be explained by their CCOT's use of palliative care services, a function not featured in our model.
Our study adds to the meta‐analyses and systematic reviews810 that have questioned the hypothesis that a trained and proactive team of caregivers should be able to prevent patients from returning to the ICU. Perhaps one reason why this is not true is that proactive rounding by RRTs may have minimal effect in systems where step‐down beds are readily available. At UCSF, nearly every patient transferred out of the ICU is triaged to a step‐down unit, where telemetry and pulse oximetry are continuously monitored. Despite this, however, our institution's 2 step‐down units generate more calls to our RRT than any other units in the hospital.
We were surprised to see that proactive rounding failed to shorten ICU LOS, hypothesizing that clinicians would be more comfortable discharging patients from the ICU knowing that the RRT would be closely monitoring them afterwards. Although we have no data to support this hypothesis, increased use of the RRT may have also increased step‐down bed use, as patients on the general medicalsurgical floors were transferred to a higher level of care upon recommendation of the RRT, thereby delaying transfers out of the ICU. Moreover, the opening of an additional 16‐bed ICU in October 2008 might have encouraged clinicians to transfer patients back to the ICU simply because beds were more easily accessible than before.
Introduction of proactive rounding by the RRT was also not associated with differences in the mortality rate of patients discharged from the ICU. This finding conflicts with the results of Garcea et al,15 Ball et al,16 and Priestley et al13, all of which found that implementation of a CCOT led to small but statistically significant reductions in in‐hospital mortality. All 3 of these studies, however, examined smaller patient populations (1380, 470, and 2903 patients, respectively), and both the Priestley and Ball studies13, 16 had significantly shorter periods of data collection (24 months and 32 weeks, respectively). Our results are based on models with confidence intervals and P values that account for variability in all 3 underlying effect estimates but assume a linear extrapolation of the preintervention trend. This approach allowed us to flexibly deal with changes related to the intervention, while relying on our large sample size to define time trends not dealt with adequately (or at all) in previous research.
The lack of improvement in outcomes cannot be attributed to immaturity of the RRT or failure of the clinical staff to use the RRT adequately. A prespecified secondary data analysis midway through the postintervention time period demonstrated that the RRT failed to gain efficacy with time with respect to all 3 outcomes. The postintervention RRT was also utilized far more frequently than its predecessor (110.6 vs 2.7 calls per 1000 admissions, respectively), and this degree of RRT utilization far surpasses the dose considered to be indicative of a mature RRT system.12
Our study has several limitations. First, we relied on administrative rather than chart‐collected data to determine the reason for ICU admission, and the APR severity of illness and risk of mortality scores. It seems unlikely, however, that coding deficiencies or biases affected the preintervention and postintervention patient populations differently. Even though we adjusted for all available measures, it is possible that we were not able to account for time trends in all potential confounders. Second, we did not have detailed clinical information on reasons for ICU readmission and whether readmissions occurred before or after the RRT proactively rounded on the patient. Therefore, potential readmissions to the ICU that might have been planned or which would have happened regardless of the presence of the RRT, such as for antibiotic desensitization, could not be accounted for. Third, introduction of proactive rounding by the RRT in June 2007 was accompanied by a change in the RRT's composition, from a physician‐led model to a nurse‐led model. Therefore, inherent differences in the way that physicians and nurses might assess and triage patients could not have been adjusted for. Lastly, this was a retrospective study conducted at a single academic medical center with a specific RRT model, and our results may not be directly applicable to nonteaching settings or to different RRT models.
Our findings raise further questions about the benefits of RRTs as they assume additional roles, such as proactive rounding on patients recently discharged from the ICU. The failure of our RRT to reduce the ICU readmission rate, the ICU average LOS, and the mortality of patients discharged from the ICU raises concerns that the benefits of our RRT are not commensurate with its cost. While defining the degree of impact and underlying mechanisms are worthy of prospective study, hospitals seeking to improve their RRT models should consider how to develop systems that achieve the RRT's promise in measurable ways.
Acknowledgements
The authors acknowledge Heather Leicester, MSPH, Senior Performance Improvement Analyst for Patient Safety and Quality Services at the University of California San Francisco for her work in data acquisition.
Disclosures: Dr Vittinghoff received salary support from an NIH grant during the time of this work for statistical consulting. He receives textbook royalties from Springer Verlag. Dr Auerbach was supported by 5K24HL098372‐02 from the National Heart Lung and Blood Institute during the period of this study although not specifically for this study; they had no role in the design or conduct of the study; the collection, analysis, or interpretation of the data; or the preparation, review, or approval of the manuscript. The other authors have no financial conflicts of interest.
- , , , . The 100,000 lives campaign: setting a goal and a deadline for improving health care quality. JAMA. 2006;295(3):324–327.
- Clinical Governance Unit, Quality and Safety Branch, Rural and Regional Health and Aged Care Services Division Safer Systems, Department of Human Services, State Government of Victoria. Safer Systems—Saving Lives Campaign. Available at: http://www.health.vic.gov.au/sssl. Accessed April 5, 2012.
- Canadian Patient Safety Institute. Safer Healthcare Now! Campaign. Available at: http://www.saferhealthcarenow.ca. Accessed April 5, 2012.
- , . The growth of rapid response systems. Jt Comm J Qual Patient Saf. 2008;34:489–495.
- , , , . The medical emergency team. Anaesth Intensive Care. 1995;23(2):183–186.
- , , , et al. Rates of in‐hospital arrests, deaths, and intensive care admission: the effect of a medical emergency team. Med J Aust. 2000;173:236–240.
- , , , , . The patient‐at‐risk team: identifying and managing seriously ill ward patients. Anaesthesia. 1999;54:853–860.
- , , , , . Effects of rapid response systems on clinical outcomes: systemic review and meta‐analysis. J Hosp Med. 2007;2:422–432.
- , , , , . Rapid response teams: a systemic review and meta‐analysis. Arch Intern Med. 2010;170(1):18–26.
- , , , , , . Rapid response systems: a systematic review. Crit Care Med. 2007;35(5):1238–1243.
- , , , et al. Introduction of the medical emergency team (MET) system: a cluster‐randomised controlled trial. Lancet. 2005;365:2091–2097.
- , , . Rapid response teams. N Engl J Med. 2011;365:139–146.
- , , , et al. Introducing critical care outreach: a ward‐randomised trial of phased introduction in a general hospital. Intensive Care Med. 2004;30(7):1398–1404.
- , . Impact of an outreach team on re‐admissions to a critical care unit. Anaesthesia. 2003;58:328–332.
- , , , , . Impact of a critical care outreach team on critical care readmissions and mortality. Acta Anaesthesiol Scand. 2004;48:1096–1100.
- , , . Effect of the critical care outreach team on patient survival to discharge from hospital and readmission to critical care: non‐randomised population based study. BMJ. 2003;327:1014–1017.
- , , , . Comorbidity measures for use with administrative data. Med Care. 1998;36(1):8–27.
- , , , . The 100,000 lives campaign: setting a goal and a deadline for improving health care quality. JAMA. 2006;295(3):324–327.
- Clinical Governance Unit, Quality and Safety Branch, Rural and Regional Health and Aged Care Services Division Safer Systems, Department of Human Services, State Government of Victoria. Safer Systems—Saving Lives Campaign. Available at: http://www.health.vic.gov.au/sssl. Accessed April 5, 2012.
- Canadian Patient Safety Institute. Safer Healthcare Now! Campaign. Available at: http://www.saferhealthcarenow.ca. Accessed April 5, 2012.
- , . The growth of rapid response systems. Jt Comm J Qual Patient Saf. 2008;34:489–495.
- , , , . The medical emergency team. Anaesth Intensive Care. 1995;23(2):183–186.
- , , , et al. Rates of in‐hospital arrests, deaths, and intensive care admission: the effect of a medical emergency team. Med J Aust. 2000;173:236–240.
- , , , , . The patient‐at‐risk team: identifying and managing seriously ill ward patients. Anaesthesia. 1999;54:853–860.
- , , , , . Effects of rapid response systems on clinical outcomes: systemic review and meta‐analysis. J Hosp Med. 2007;2:422–432.
- , , , , . Rapid response teams: a systemic review and meta‐analysis. Arch Intern Med. 2010;170(1):18–26.
- , , , , , . Rapid response systems: a systematic review. Crit Care Med. 2007;35(5):1238–1243.
- , , , et al. Introduction of the medical emergency team (MET) system: a cluster‐randomised controlled trial. Lancet. 2005;365:2091–2097.
- , , . Rapid response teams. N Engl J Med. 2011;365:139–146.
- , , , et al. Introducing critical care outreach: a ward‐randomised trial of phased introduction in a general hospital. Intensive Care Med. 2004;30(7):1398–1404.
- , . Impact of an outreach team on re‐admissions to a critical care unit. Anaesthesia. 2003;58:328–332.
- , , , , . Impact of a critical care outreach team on critical care readmissions and mortality. Acta Anaesthesiol Scand. 2004;48:1096–1100.
- , , . Effect of the critical care outreach team on patient survival to discharge from hospital and readmission to critical care: non‐randomised population based study. BMJ. 2003;327:1014–1017.
- , , , . Comorbidity measures for use with administrative data. Med Care. 1998;36(1):8–27.
Copyright © 2012 Society of Hospital Medicine
Risk Factors For Unplanned ICU Transfer
Emergency Department (ED) patients who are hospitalized and require unplanned transfer to the intensive care unit (ICU) within 24 hours of arrival on the ward have higher mortality than direct ICU admissions.1, 2 Previous research found that 5% of ED admissions experienced unplanned ICU transfer during their hospitalization, yet these patients account for 25% of in‐hospital deaths and have a longer length of stay than direct ICU admissions.1, 3 For these reasons, inpatient rapid‐response teams and early warning systems have been studied to reduce the mortality of patients who rapidly deteriorate on the hospital ward.410 However, there is little conclusive evidence that these interventions decrease mortality.710 It is possible that with better recognition and intervention in the ED, a portion of these unplanned ICU transfers and their subsequent adverse outcomes could be prevented.11
Previous research on risk factors for unplanned ICU transfers among ED admissions is limited. While 2 previous studies from non‐US hospitals used administrative data to identify some general populations at risk for unplanned ICU transfer,12, 13 these studies did not differentiate between transfers shortly after admission and those that occurred during a prolonged hospital staya critical distinction since the outcomes between these groups differs substantially.1 Another limitation of these studies is the absence of physiologic measures at ED presentation, which have been shown to be highly predictive of mortality.14
In this study, we describe risk factors for unplanned transfer to the ICU within 24 hours of arrival on the ward, among a large cohort of ED hospitalizations across 13 community hospitals. Focusing on admitting diagnoses most at risk, our goal was to inform efforts to improve the triage of ED admissions and determine which patients may benefit from additional interventions, such as improved resuscitation, closer monitoring, or risk stratification tools. We also hypothesized that higher volume hospitals would have lower rates of unplanned ICU transfers, as these hospitals are more likely have more patient care resources on the hospital ward and a higher threshold to transfer to the ICU.
METHODS
Setting and Patients
The setting for this study was Kaiser Permanente Northern California (KPNC), a large integrated healthcare delivery system serving approximately 3.3 million members.1, 3, 15, 16 We extracted data on all adult ED admissions (18 years old) to the hospital between 2007 and 2009. We excluded patients who went directly to the operating room or the ICU, as well as gynecological/pregnancy‐related admissions, as these patients have substantially different mortality risks.14 ED admissions to hospital wards could either go to medicalsurgical units or transitional care units (TCU), an intermediate level of care between the medicalsurgical units and the ICU. We chose to focus on hospitals with similar inpatient structures. Thus, 8 hospitals without TCUs were excluded, leaving 13 hospitals for analysis. The KPNC Institutional Review Board approved this study.
Main Outcome Measure
The main outcome measure was unplanned transfer to the ICU within 24 hours of arrival to the hospital ward, based upon bed history data. As in previous research, we make the assumptionwhich is supported by the high observed‐to‐expected mortality ratios found in these patientsthat these transfers to the ICU were due to clinical deterioration, and thus were unplanned, rather than a planned transfer to the ICU as is more common after an elective surgical procedure.13 The comparison population was patients admitted from the ED to the ward who never experienced a transfer to the ICU.
Patient and Hospital Characteristics
We extracted patient data on age, sex, admitting diagnosis, chronic illness burden, acute physiologic derangement in the ED, and hospital unit length of stay. Chronic illness was measured using the Comorbidity Point Score (COPS), and physiologic derangement was measured using the Laboratory Acute Physiology Score (LAPS) calculated from labs collected in the ED.1, 14, 17 The derivation of these variables from the electronic medical record has been previously described.14 The COPS was derived from International Classification of Diseases, Ninth Revision (ICD‐9) codes for all Kaiser Permanente Medical Care Program (KPMCP) inpatient and outpatient encounters prior to hospitalization. The LAPS is based on 14 possible lab tests that could be drawn in the ED or in the 72 hours prior to hospitalization. The admitting diagnosis is the ICD‐9 code assigned for the primary diagnosis determined by the admitting physician at the time when hospital admission orders are entered. We further collapsed a previously used categorization of 44 primary condition diagnoses, based on admission ICD‐9 codes,14 into 25 broad diagnostic categories based on pathophysiologic plausibility and mortality rates. We tabulated inpatient admissions originating in the ED to derive a hospital volume measure.
Statistical Analyses
We compared patient characteristics, hospital volume, and outcomes by whether or not an unplanned ICU transfer occurred. Unadjusted analyses were performed with analysis of variance (ANOVA) and chi‐square tests. We calculated crude rates of unplanned ICU transfer per 1,000 ED inpatient admissions by patient characteristics and by hospital, stratified by hospital volume.
We used a hierarchical multivariate logistic regression model to estimate adjusted odds ratios for unplanned ICU transfer as a function of both patient‐level variables (age, sex, COPS, LAPS, time of admission, admission to TCU vs ward, admitting diagnosis) and hospital‐level variables (volume) in the model. We planned to choose the reference group for admitting diagnosis as the one with an unadjusted odds ratio closest to the null (1.00). This model addresses correlations between patients with multiple hospitalizations and clustering by hospital, by fitting random intercepts for these clusters. All analyses were performed in Stata 12 (StataCorp, College Station, TX), and statistics are presented with 95% confidence intervals (CI). The Stata program gllamm (Generalized Linear Latent and Mixed Models) was used for hierarchical modeling.18
RESULTS
Of 178,315 ED non‐ICU hospitalizations meeting inclusion criteria, 4,252 (2.4%) were admitted to the ward and were transferred to the ICU within 24 hours of leaving the ED. There were 122,251 unique patients in our study population. Table 1 compares the characteristics of ED hospitalizations in which an unplanned transfer occurred to those that did not experience an unplanned transfer. Unplanned transfers were more likely to have a higher comorbidity burden, more deranged physiology, and more likely to arrive on the floor during the overnight shift.
| Characteristics | Unplanned Transfer to ICU Within 24 h of Leaving ED? | P Value* | |
|---|---|---|---|
| Yes | No | ||
| N = 4,252 (2.4%) | N = 174,063 (97.6%) | ||
| |||
| Age, median (IQR) | 69 (5680) | 70 (5681) | <0.01 |
| Male, % | 51.3 | 45.9 | <0.01 |
| Comorbidity Points Score (COPS), median (IQR) | 100 (46158) | 89 (42144) | <0.01 |
| Laboratory Acute Physiology Score (LAPS), median (IQR) | 26 (1342) | 18 (633) | <0.01 |
| Nursing shift on arrival to floor, % | |||
| Day: 7 am3 pm (Reference) | 20.1 | 20.1 | NS |
| Evening: 3 pm11 pm | 47.6 | 50.2 | NS |
| Overnight: 11 pm7 am | 32.3 | 29.7 | <0.01 |
| Weekend admission, % | 33.7 | 32.7 | NS |
| Admitted to monitored bed, % | 24.1 | 24.9 | NS |
| Emergency department annual volume, mean (SD) | 48,755 (15,379) | 50,570 (15,276) | <0.01 |
| Non‐ICU annual admission volume, mean (SD) | 5,562 (1,626) | 5,774 (1,568) | <0.01 |
| Admitting diagnosis, listed by descending frequency, % | NS | ||
| Pneumonia and respiratory infections | 16.3 | 11.8 | <0.01 |
| Gastrointestinal bleeding | 12.8 | 13.6 | NS |
| Chest pain | 7.3 | 10.0 | <0.01 |
| Miscellaneous conditions | 5.6 | 6.2 | NS |
| All other acute infections | 4.7 | 6.0 | <0.01 |
| Seizures | 4.1 | 5.9 | <0.01 |
| AMI | 3.9 | 3.3 | <0.05 |
| COPD | 3.8 | 3.0 | <0.01 |
| CHF | 3.5 | 3.7 | NS |
| Arrhythmias and pulmonary embolism | 3.5 | 3.3 | NS |
| Stroke | 3.4 | 3.5 | NS |
| Diabetic emergencies | 3.3 | 2.6 | <0.01 |
| Metabolic, endocrine, electrolytes | 3.0 | 2.9 | NS |
| Sepsis | 3.0 | 1.2 | <0.01 |
| Other neurology and toxicology | 3.0 | 2.9 | NS |
| Urinary tract infections | 2.9 | 3.2 | NS |
| Catastrophic conditions | 2.6 | 1.2 | <0.01 |
| Rheumatology | 2.5 | 3.5 | <0.01 |
| Hematology and oncology | 2.4 | 2.4 | NS |
| Acute renal failure | 1.9 | 1.1 | <0.01 |
| Pancreatic and liver | 1.7 | 2.0 | NS |
| Trauma, fractures, and dislocations | 1.6 | 1.8 | NS |
| Bowel obstructions and diseases | 1.6 | 2.9 | <0.01 |
| Other cardiac conditions | 1.5 | 1.3 | NS |
| Other renal conditions | 0.6 | 1.0 | <0.01 |
| Inpatient length of stay, median days (IQR) | 4.7 (2.78.6) | 2.6 (1.54.4) | <0.01 |
| Died during hospitalization, % | 12.7 | 2.4 | <0.01 |
Unplanned ICU transfers were more frequent in lower volume hospitals (Table 1). Figure 1 displays the inverse relationship between hospital annual ED inpatient admission volume and unplanned ICU transfers rates. The lowest volume hospital had a crude rate twice as high as the 2 highest volume hospitals (39 vs 20, per 1,000 admissions).
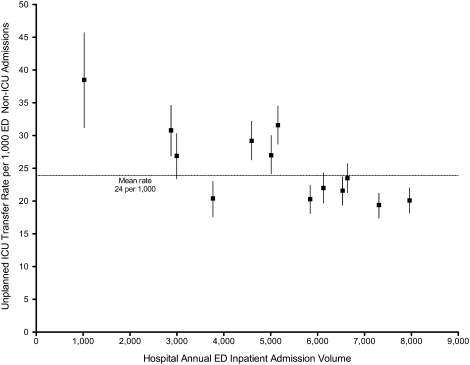
Pneumonia/respiratory infection was the most frequent admitting condition associated with unplanned transfer (16.3%) (Table 1). There was also wide variation in crude rates for unplanned ICU transfer by admitting condition (Figure 2). Patients admitted with sepsis had the highest rate (59 per 1,000 admissions), while patients admitted with renal conditions other than acute renal failure had the lowest rates (14.3 per 1,000 admissions).
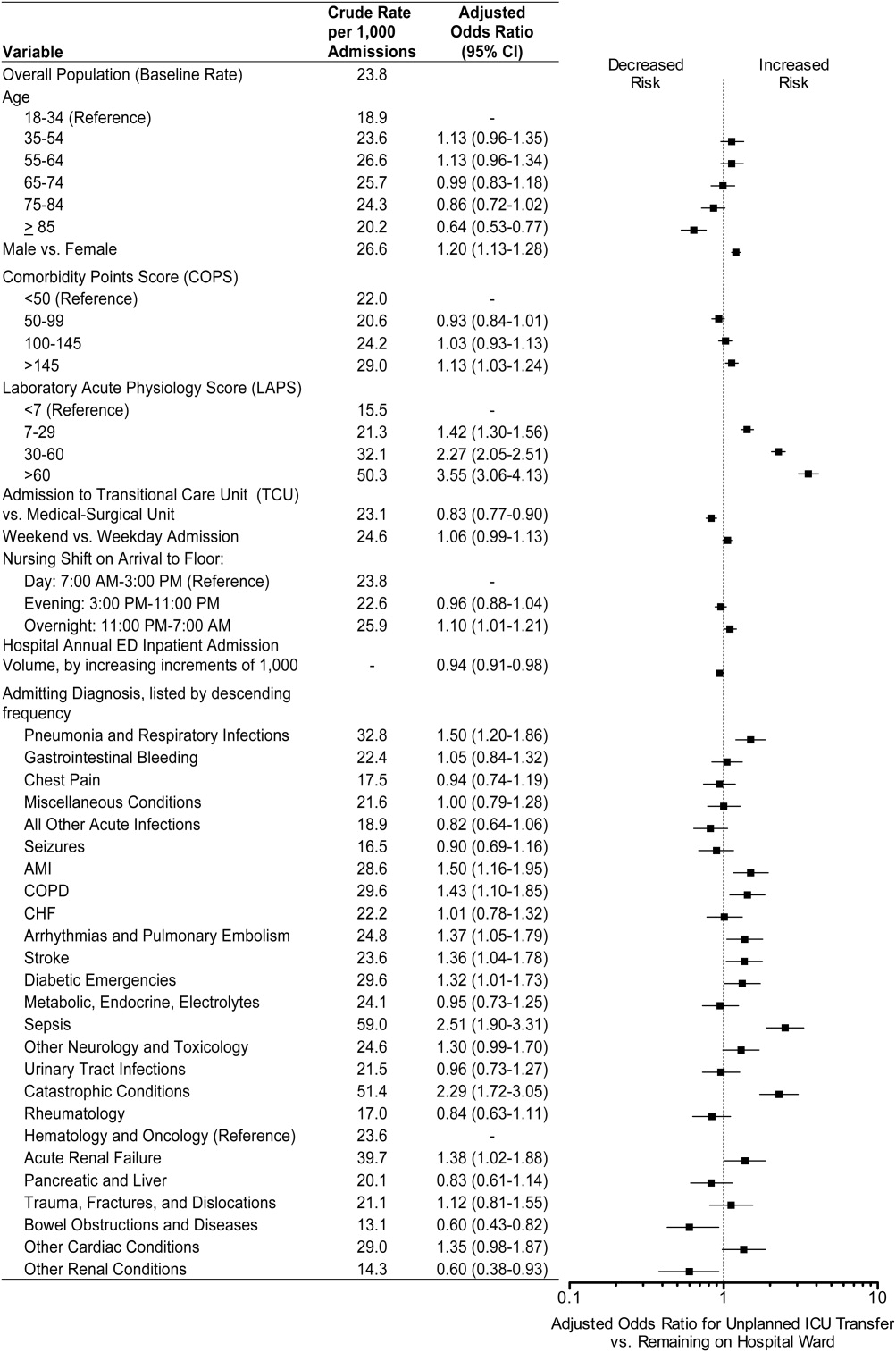
We confirmed that almost all diagnoses found to account for a disproportionately high share of unplanned ICU transfers in Table 1 were indeed independently associated with this phenomenon after adjustment for patient and hospital differences (Figure 2). Pneumonia remained the most frequent condition associated with unplanned ICU transfer (odds ratio [OR] 1.50; 95% CI 1.201.86). Although less frequent, sepsis had the strongest association of any condition with unplanned transfer (OR 2.51; 95% CI 1.903.31). However, metabolic, endocrine, and electrolyte conditions were no longer associated with unplanned transfer after adjustment, while arrhythmias and pulmonary embolism were. Other conditions confirmed to be associated with increased risk of unplanned transfer included: myocardial infarction (MI), chronic obstructive pulmonary disease (COPD), stroke, diabetic emergencies, catastrophic conditions (includes aortic catastrophes, all forms of shock except septic shock, and intracranial hemorrhage), and acute renal failure. After taking into account the frequency of admitting diagnoses, respiratory conditions (COPD, pneumonia/acute respiratory infection) comprised nearly half (47%) of all conditions associated with increased risk of unplanned ICU transfer.
Other factors confirmed to be independently associated with unplanned ICU transfer included: male sex (OR 1.20; 95% CI 1.131.28), high comorbidity burden as measured by COPS >145 (OR 1.13; 95% CI 1.031.24), increasingly abnormal physiology compared to a LAPS <7, and arrival on ward during the overnight shift (OR 1.10; 95% CI 1.011.21). After adjustment, we did find that admission to the TCU rather than a medicalsurgical unit was associated with decreased risk of unplanned ICU transfer (OR 0.83; 95% CI 0.770.90). Age 85 was associated with decreased risk of unplanned ICU transfer relative to the youngest age group of 1834‐year‐old patients (OR 0.64; 95% CI 0.530.77).
ED admissions to higher volume hospitals were 6% less likely to experience an unplanned transfer for each additional 1,000 annual ED hospitalizations over a lower volume hospital (OR 0.94; 95% CI 0.910.98). In other words, a patient admitted to a hospital with 8,000 annual ED hospitalizations had 30% decreased odds of unplanned ICU transfer compared to a hospital with only 3,000 annual ED hospitalizations.
DISCUSSION
Patients admitted with respiratory conditions accounted for half of all admitting diagnoses associated with increased risk of unplanned transfer to the ICU within 24 hours of arrival to the ward. We found that 1 in 30 ED ward admissions for pneumonia, and 1 in 33 for COPD, were transferred to the ICU within 24 hours. These findings indicate that there is some room for improvement in early care of respiratory conditions, given the average unplanned transfer rate of 1 in 42, and previous research showing that patients with pneumonia and patients with COPD, who experience unplanned ICU transfer, have substantially worse mortality than those directly admitted to the ICU.1
Although less frequent than hospitalizations for respiratory conditions, patients admitted with sepsis were at the highest risk of unplanned ICU transfer (1 in 17 ED non‐ICU hospitalizations). We also found that MI and stroke ward admissions had a higher risk of unplanned ICU transfer. However, we previously found that unplanned ICU transfers for sepsis, MI, and stroke did not have worse mortality than direct ICU admits for these conditions.1 Therefore, quality improvement efforts to reduce excess mortality related to early decompensation in the hospital and unplanned ICU transfer would be most effective if targeted towards respiratory conditions such as pneumonia and COPD.
This is the only in‐depth study, to our knowledge, to explore the association between a set of mutually exclusive diagnostic categories and risk of unplanned ICU transfer within 24 hours, and it is the first study to identify risk factors for unplanned ICU transfer in a multi‐hospital cohort adjusted for patient‐ and hospital‐level characteristics. We also identified a novel hospital volumeoutcome relationship: Unplanned ICU transfers are up to twice as likely to occur in the smallest volume hospitals compared with highest volume hospitals. Hospital volume has long been proposed as a proxy for hospital resources; there are several studies showing a relationship between low‐volume hospitals and worse outcomes for a number of conditions.19, 20 Possible mechanisms may include decreased ICU capacity, decreased on‐call intensivists in the hospital after hours, and less experience with certain critical care conditions seen more frequently in high‐volume hospitals.21
Patients at risk of unplanned ICU transfer were also more likely to have physiologic derangement identified on laboratory testing, high comorbidity burden, and arrive on the ward between 11 PM and 7 AM. Given the strong correlation between comorbidity burden and physiologic derangement and mortality,14 it is not surprising that the COPS and LAPS were independent predictors of unplanned transfer. It is unclear, however, why arriving on the ward on the overnight shift is associated with higher risk. One possibility is that patients who arrive on the wards during 11 PM to 7 AM are also likely to have been in the ED during evening peak hours most associated with ED crowding.22 High levels of ED crowding have been associated with delays in care, worse quality care, lapses in patient safety, and even increased in‐hospital mortality.22, 23 Other possible reasons include decreased in‐hospital staffing and longer delays in critical diagnostic tests and interventions.2428
Admission to TCUs was associated with decreased risk of unplanned ICU transfer in the first 24 hours of hospitalization. This may be due to the continuous monitoring, decreased nursing‐to‐patient ratios, or the availability to provide some critical care interventions. In our study, age 85 was associated with lower likelihood of unplanned transfer. Unfortunately, we did not have access to data on advanced directives or patient preferences. Data on advanced directives would help to distinguish whether this phenomenon was related to end‐of‐life care goals versus other explanations.
Our study confirms some risk factors identified in previous studies. These include specific diagnoses such as pneumonia and COPD,12, 13, 29 heavy comorbidity burden,12, 13, 29 abnormal labs,29 and male sex.13 Pneumonia has consistently been shown to be a risk factor for unplanned ICU transfer. This may stem from the dynamic nature of this condition and its ability to rapidly progress, and the fact that some ICUs may not accept pneumonia patients unless they demonstrate a need for mechanical ventilation.30 Recently, a prediction rule has been developed to determine which patients with pneumonia are likely to have an unplanned ICU transfer.30 It is possible that with validation and application of this rule, unplanned transfer rates for pneumonia could be reduced. It is unclear whether males have unmeasured factors associated with increased risk of unplanned transfer or whether a true gender disparity exists.
Our findings should be interpreted within the context of this study's limitations. First, this study was not designed to distinguish the underlying cause of the unplanned transfer such as under‐recognition of illness severity in the ED, evolving clinical disease after leaving the ED, or delays in critical interventions on the ward. These are a focus of our ongoing research efforts. Second, while previous studies have demonstrated that our automated risk adjustment variables can accurately predict in‐hospital mortality (0.88 area under curve in external populations),17 additional data on vital signs and mental status could further improve risk adjustment. However, using automated data allowed us to study risk factors for unplanned transfer in a multi‐hospital cohort with a much larger population than has been previously studied. Serial data on vital signs and mental status both in the ED and during hospitalization could also be helpful in determining which unplanned transfers could be prevented with earlier recognition and intervention. Finally, all patient care occurred within an integrated healthcare delivery system. Thus, differences in case‐mix, hospital resources, ICU structure, and geographic location should be considered when applying our results to other healthcare systems.
This study raises several new areas for future research. With access to richer data becoming available in electronic medical records, prediction rules should be developed to enable better triage to appropriate levels of care for ED admissions. Future research should also analyze the comparative effectiveness of intermediate monitored units versus non‐monitored wards for preventing clinical deterioration by admitting diagnosis. Diagnoses that have been shown to have an increased risk of death after unplanned ICU transfer, such as pneumonia/respiratory infection and COPD,1 should be prioritized in this research. Better understanding is needed on the diagnosis‐specific differences and the differences in ED triage process and ICU structure that may explain why high‐volume hospitals have significantly lower rates of early unplanned ICU transfers compared with low‐volume hospitals. In particular, determining the effect of TCU and ICU capacities and census at the time of admission, and comparing patient risk characteristics across hospital‐volume strata would be very useful. Finally, more work is needed to determine whether the higher rate of unplanned transfers during overnight nursing shifts is related to decreased resource availability, preceding ED crowding, or other organizational causes.
In conclusion, patients admitted with respiratory conditions, sepsis, MI, high comorbidity, and abnormal labs are at modestly increased risk of unplanned ICU transfer within 24 hours of admission from the ED. Patients admitted with respiratory conditions (pneumonia/respiratory infections and COPD) accounted for half of the admitting diagnoses that are at increased risk for unplanned ICU transfer. These patients may benefit from better inpatient triage from the ED, earlier intervention, or closer monitoring. More research is needed to determine the specific aspects of care associated with admission to intermediate care units and high‐volume hospitals that reduce the risk of unplanned ICU transfer.
Acknowledgements
The authors thank John D. Greene, Juan Carlos La Guardia, and Benjamin Turk for their assistance with formatting of the dataset; Dr Alan S. Go, Acting Director of the Division of Research, for reviewing the manuscript; and Alina Schnake‐Mahl for formatting the manuscript.
- , , , et al. Adverse outcomes associated with delayed intensive care unit transfers in an integrated healthcare system. J Hosp Med. 2011;7(3):224–230.
- , , , et al. Inpatient transfers to the intensive care unit. J Gen Intern Med. 2003;18(2):77–83.
- , , , et al. Intra‐hospital transfers to a higher level of care: contribution to total hospital and intensive care unit (ICU) mortality and length of stay (LOS). J Hosp Med. 2011;6:74–80.
- , , , et al. Hospital‐wide code rates and mortality before and after implementation of a rapid response team. JAMA. 2008;300(21):2506–2513.
- , , , et al. Effect of a rapid response team on hospital‐wide mortality and code rates outside the ICU in a children's hospital. JAMA. 2007;298(19):2267–2274.
- , , , et al. Introduction of the medical emergency team (MET) system: a cluster‐randomised controlled trial. Lancet. 2005;365(9477):2091–2097.
- , , , et al. Rapid response systems: A systematic review. Crit Care Med. 2007;35(5):1238–1243.
- , , , et al. Effects of rapid response systems on clinical outcomes: systematic review and meta‐analysis. J Hosp Med. 2007;2(6):422–432.
- , , , et al. Rapid response teams: a systematic review and meta‐analysis. Arch Intern Med. 2010;170(1):18–26.
- , , , et al. Outreach and early warning systems (EWS) for the prevention of intensive care admission and death of critically ill adult patients on general hospital wards. Cochrane Database Syst Rev. 2007;3:CD005529.
- , , , et al. Unplanned transfers to a medical intensive care unit: Causes and relationship to preventable errors in care. J Hosp Med. 2011;6:68–72.
- , , , et al. Using administrative data to develop a nomogram for individualising risk of unplanned admission to intensive care. Resuscitation. 2008;79(2):241–248.
- , , , et al. Unplanned admission to intensive care after emergency hospitalisation: risk factors and development of a nomogram for individualising risk. Resuscitation. 2009;80(2):224–230.
- , , , et al. Risk‐adjusting hospital inpatient mortality using automated inpatient, outpatient, and laboratory databases. Med Care. 2008;46(3):232–239.
- . Linking automated databases for research in managed care settings. Ann Intern Med. 1997;127(8 pt 2):719–724.
- , , , et al. Risk adjusting community‐acquired pneumonia hospital outcomes using automated databases. Am J Manag Care. 2008;14(3):158–166.
- , , , et al. The Kaiser Permanente inpatient risk adjustment methodology was valid in an external patient population. J Clin Epidemiol. 2011;63(7):798–803.
- , , . Maximum likelihood estimation of limited and discrete dependent variable models with nested random effects. J Econometrics. 2005;128(2):301–323.
- . The relation between volume and outcome in health care. N Engl J Med. 1999;340(21):1677–1679.
- , , . Is volume related to outcome in health care? A systematic review and methodologic critique of the literature. Ann Intern Med. 2002;137(6):511–520.
- , , . Working with capacity limitations: operations management in critical care. Crit Care. 2011;15(4):308.
- , . Systematic review of emergency department crowding: causes, effects, and solutions. Ann Intern Med. 2008;52(2):126–136.
- , , , et al. The effect of emergency department crowding on clinically oriented outcomes. Acad Emerg Med. 2009;16(1):1–10.
- , , , et al. Association between time of admission to the ICU and mortality. Chest. 2010;138(1):68–75.
- , , , et al. Off‐hour admission and in‐hospital stroke case fatality in the get with the guidelines‐stroke program. Stroke. 2009;40(2):569–576.
- , , , et al. Relationship between time of day, day of week, timeliness of reperfusion, and in‐hospital mortality for patients with acute ST‐segment elevation myocardial infarction. JAMA. 2005;294(7):803–812.
- , , , et al. Hospital mortality among adults admitted to and discharged from intensive care on weekends and evenings. J Crit Care. 2008;23(3):317–324.
- , , , et al. Association between ICU admission during morning rounds and mortality. Chest. 2009;136(6):1489–1495.
- , , , et al. Identifying infected emergency department patients admitted to the hospital ward at risk of clinical deterioration and intensive care unit transfer. Acad Emerg Med. 2010;17(10):1080–1085.
- , , , et al. Risk stratification of early admission to the intensive care unit of patients with no major criteria of severe community‐acquired pneumonia: development of an international prediction rule. Crit Care. 2009;13(2):R54.
Emergency Department (ED) patients who are hospitalized and require unplanned transfer to the intensive care unit (ICU) within 24 hours of arrival on the ward have higher mortality than direct ICU admissions.1, 2 Previous research found that 5% of ED admissions experienced unplanned ICU transfer during their hospitalization, yet these patients account for 25% of in‐hospital deaths and have a longer length of stay than direct ICU admissions.1, 3 For these reasons, inpatient rapid‐response teams and early warning systems have been studied to reduce the mortality of patients who rapidly deteriorate on the hospital ward.410 However, there is little conclusive evidence that these interventions decrease mortality.710 It is possible that with better recognition and intervention in the ED, a portion of these unplanned ICU transfers and their subsequent adverse outcomes could be prevented.11
Previous research on risk factors for unplanned ICU transfers among ED admissions is limited. While 2 previous studies from non‐US hospitals used administrative data to identify some general populations at risk for unplanned ICU transfer,12, 13 these studies did not differentiate between transfers shortly after admission and those that occurred during a prolonged hospital staya critical distinction since the outcomes between these groups differs substantially.1 Another limitation of these studies is the absence of physiologic measures at ED presentation, which have been shown to be highly predictive of mortality.14
In this study, we describe risk factors for unplanned transfer to the ICU within 24 hours of arrival on the ward, among a large cohort of ED hospitalizations across 13 community hospitals. Focusing on admitting diagnoses most at risk, our goal was to inform efforts to improve the triage of ED admissions and determine which patients may benefit from additional interventions, such as improved resuscitation, closer monitoring, or risk stratification tools. We also hypothesized that higher volume hospitals would have lower rates of unplanned ICU transfers, as these hospitals are more likely have more patient care resources on the hospital ward and a higher threshold to transfer to the ICU.
METHODS
Setting and Patients
The setting for this study was Kaiser Permanente Northern California (KPNC), a large integrated healthcare delivery system serving approximately 3.3 million members.1, 3, 15, 16 We extracted data on all adult ED admissions (18 years old) to the hospital between 2007 and 2009. We excluded patients who went directly to the operating room or the ICU, as well as gynecological/pregnancy‐related admissions, as these patients have substantially different mortality risks.14 ED admissions to hospital wards could either go to medicalsurgical units or transitional care units (TCU), an intermediate level of care between the medicalsurgical units and the ICU. We chose to focus on hospitals with similar inpatient structures. Thus, 8 hospitals without TCUs were excluded, leaving 13 hospitals for analysis. The KPNC Institutional Review Board approved this study.
Main Outcome Measure
The main outcome measure was unplanned transfer to the ICU within 24 hours of arrival to the hospital ward, based upon bed history data. As in previous research, we make the assumptionwhich is supported by the high observed‐to‐expected mortality ratios found in these patientsthat these transfers to the ICU were due to clinical deterioration, and thus were unplanned, rather than a planned transfer to the ICU as is more common after an elective surgical procedure.13 The comparison population was patients admitted from the ED to the ward who never experienced a transfer to the ICU.
Patient and Hospital Characteristics
We extracted patient data on age, sex, admitting diagnosis, chronic illness burden, acute physiologic derangement in the ED, and hospital unit length of stay. Chronic illness was measured using the Comorbidity Point Score (COPS), and physiologic derangement was measured using the Laboratory Acute Physiology Score (LAPS) calculated from labs collected in the ED.1, 14, 17 The derivation of these variables from the electronic medical record has been previously described.14 The COPS was derived from International Classification of Diseases, Ninth Revision (ICD‐9) codes for all Kaiser Permanente Medical Care Program (KPMCP) inpatient and outpatient encounters prior to hospitalization. The LAPS is based on 14 possible lab tests that could be drawn in the ED or in the 72 hours prior to hospitalization. The admitting diagnosis is the ICD‐9 code assigned for the primary diagnosis determined by the admitting physician at the time when hospital admission orders are entered. We further collapsed a previously used categorization of 44 primary condition diagnoses, based on admission ICD‐9 codes,14 into 25 broad diagnostic categories based on pathophysiologic plausibility and mortality rates. We tabulated inpatient admissions originating in the ED to derive a hospital volume measure.
Statistical Analyses
We compared patient characteristics, hospital volume, and outcomes by whether or not an unplanned ICU transfer occurred. Unadjusted analyses were performed with analysis of variance (ANOVA) and chi‐square tests. We calculated crude rates of unplanned ICU transfer per 1,000 ED inpatient admissions by patient characteristics and by hospital, stratified by hospital volume.
We used a hierarchical multivariate logistic regression model to estimate adjusted odds ratios for unplanned ICU transfer as a function of both patient‐level variables (age, sex, COPS, LAPS, time of admission, admission to TCU vs ward, admitting diagnosis) and hospital‐level variables (volume) in the model. We planned to choose the reference group for admitting diagnosis as the one with an unadjusted odds ratio closest to the null (1.00). This model addresses correlations between patients with multiple hospitalizations and clustering by hospital, by fitting random intercepts for these clusters. All analyses were performed in Stata 12 (StataCorp, College Station, TX), and statistics are presented with 95% confidence intervals (CI). The Stata program gllamm (Generalized Linear Latent and Mixed Models) was used for hierarchical modeling.18
RESULTS
Of 178,315 ED non‐ICU hospitalizations meeting inclusion criteria, 4,252 (2.4%) were admitted to the ward and were transferred to the ICU within 24 hours of leaving the ED. There were 122,251 unique patients in our study population. Table 1 compares the characteristics of ED hospitalizations in which an unplanned transfer occurred to those that did not experience an unplanned transfer. Unplanned transfers were more likely to have a higher comorbidity burden, more deranged physiology, and more likely to arrive on the floor during the overnight shift.
| Characteristics | Unplanned Transfer to ICU Within 24 h of Leaving ED? | P Value* | |
|---|---|---|---|
| Yes | No | ||
| N = 4,252 (2.4%) | N = 174,063 (97.6%) | ||
| |||
| Age, median (IQR) | 69 (5680) | 70 (5681) | <0.01 |
| Male, % | 51.3 | 45.9 | <0.01 |
| Comorbidity Points Score (COPS), median (IQR) | 100 (46158) | 89 (42144) | <0.01 |
| Laboratory Acute Physiology Score (LAPS), median (IQR) | 26 (1342) | 18 (633) | <0.01 |
| Nursing shift on arrival to floor, % | |||
| Day: 7 am3 pm (Reference) | 20.1 | 20.1 | NS |
| Evening: 3 pm11 pm | 47.6 | 50.2 | NS |
| Overnight: 11 pm7 am | 32.3 | 29.7 | <0.01 |
| Weekend admission, % | 33.7 | 32.7 | NS |
| Admitted to monitored bed, % | 24.1 | 24.9 | NS |
| Emergency department annual volume, mean (SD) | 48,755 (15,379) | 50,570 (15,276) | <0.01 |
| Non‐ICU annual admission volume, mean (SD) | 5,562 (1,626) | 5,774 (1,568) | <0.01 |
| Admitting diagnosis, listed by descending frequency, % | NS | ||
| Pneumonia and respiratory infections | 16.3 | 11.8 | <0.01 |
| Gastrointestinal bleeding | 12.8 | 13.6 | NS |
| Chest pain | 7.3 | 10.0 | <0.01 |
| Miscellaneous conditions | 5.6 | 6.2 | NS |
| All other acute infections | 4.7 | 6.0 | <0.01 |
| Seizures | 4.1 | 5.9 | <0.01 |
| AMI | 3.9 | 3.3 | <0.05 |
| COPD | 3.8 | 3.0 | <0.01 |
| CHF | 3.5 | 3.7 | NS |
| Arrhythmias and pulmonary embolism | 3.5 | 3.3 | NS |
| Stroke | 3.4 | 3.5 | NS |
| Diabetic emergencies | 3.3 | 2.6 | <0.01 |
| Metabolic, endocrine, electrolytes | 3.0 | 2.9 | NS |
| Sepsis | 3.0 | 1.2 | <0.01 |
| Other neurology and toxicology | 3.0 | 2.9 | NS |
| Urinary tract infections | 2.9 | 3.2 | NS |
| Catastrophic conditions | 2.6 | 1.2 | <0.01 |
| Rheumatology | 2.5 | 3.5 | <0.01 |
| Hematology and oncology | 2.4 | 2.4 | NS |
| Acute renal failure | 1.9 | 1.1 | <0.01 |
| Pancreatic and liver | 1.7 | 2.0 | NS |
| Trauma, fractures, and dislocations | 1.6 | 1.8 | NS |
| Bowel obstructions and diseases | 1.6 | 2.9 | <0.01 |
| Other cardiac conditions | 1.5 | 1.3 | NS |
| Other renal conditions | 0.6 | 1.0 | <0.01 |
| Inpatient length of stay, median days (IQR) | 4.7 (2.78.6) | 2.6 (1.54.4) | <0.01 |
| Died during hospitalization, % | 12.7 | 2.4 | <0.01 |
Unplanned ICU transfers were more frequent in lower volume hospitals (Table 1). Figure 1 displays the inverse relationship between hospital annual ED inpatient admission volume and unplanned ICU transfers rates. The lowest volume hospital had a crude rate twice as high as the 2 highest volume hospitals (39 vs 20, per 1,000 admissions).

Pneumonia/respiratory infection was the most frequent admitting condition associated with unplanned transfer (16.3%) (Table 1). There was also wide variation in crude rates for unplanned ICU transfer by admitting condition (Figure 2). Patients admitted with sepsis had the highest rate (59 per 1,000 admissions), while patients admitted with renal conditions other than acute renal failure had the lowest rates (14.3 per 1,000 admissions).

We confirmed that almost all diagnoses found to account for a disproportionately high share of unplanned ICU transfers in Table 1 were indeed independently associated with this phenomenon after adjustment for patient and hospital differences (Figure 2). Pneumonia remained the most frequent condition associated with unplanned ICU transfer (odds ratio [OR] 1.50; 95% CI 1.201.86). Although less frequent, sepsis had the strongest association of any condition with unplanned transfer (OR 2.51; 95% CI 1.903.31). However, metabolic, endocrine, and electrolyte conditions were no longer associated with unplanned transfer after adjustment, while arrhythmias and pulmonary embolism were. Other conditions confirmed to be associated with increased risk of unplanned transfer included: myocardial infarction (MI), chronic obstructive pulmonary disease (COPD), stroke, diabetic emergencies, catastrophic conditions (includes aortic catastrophes, all forms of shock except septic shock, and intracranial hemorrhage), and acute renal failure. After taking into account the frequency of admitting diagnoses, respiratory conditions (COPD, pneumonia/acute respiratory infection) comprised nearly half (47%) of all conditions associated with increased risk of unplanned ICU transfer.
Other factors confirmed to be independently associated with unplanned ICU transfer included: male sex (OR 1.20; 95% CI 1.131.28), high comorbidity burden as measured by COPS >145 (OR 1.13; 95% CI 1.031.24), increasingly abnormal physiology compared to a LAPS <7, and arrival on ward during the overnight shift (OR 1.10; 95% CI 1.011.21). After adjustment, we did find that admission to the TCU rather than a medicalsurgical unit was associated with decreased risk of unplanned ICU transfer (OR 0.83; 95% CI 0.770.90). Age 85 was associated with decreased risk of unplanned ICU transfer relative to the youngest age group of 1834‐year‐old patients (OR 0.64; 95% CI 0.530.77).
ED admissions to higher volume hospitals were 6% less likely to experience an unplanned transfer for each additional 1,000 annual ED hospitalizations over a lower volume hospital (OR 0.94; 95% CI 0.910.98). In other words, a patient admitted to a hospital with 8,000 annual ED hospitalizations had 30% decreased odds of unplanned ICU transfer compared to a hospital with only 3,000 annual ED hospitalizations.
DISCUSSION
Patients admitted with respiratory conditions accounted for half of all admitting diagnoses associated with increased risk of unplanned transfer to the ICU within 24 hours of arrival to the ward. We found that 1 in 30 ED ward admissions for pneumonia, and 1 in 33 for COPD, were transferred to the ICU within 24 hours. These findings indicate that there is some room for improvement in early care of respiratory conditions, given the average unplanned transfer rate of 1 in 42, and previous research showing that patients with pneumonia and patients with COPD, who experience unplanned ICU transfer, have substantially worse mortality than those directly admitted to the ICU.1
Although less frequent than hospitalizations for respiratory conditions, patients admitted with sepsis were at the highest risk of unplanned ICU transfer (1 in 17 ED non‐ICU hospitalizations). We also found that MI and stroke ward admissions had a higher risk of unplanned ICU transfer. However, we previously found that unplanned ICU transfers for sepsis, MI, and stroke did not have worse mortality than direct ICU admits for these conditions.1 Therefore, quality improvement efforts to reduce excess mortality related to early decompensation in the hospital and unplanned ICU transfer would be most effective if targeted towards respiratory conditions such as pneumonia and COPD.
This is the only in‐depth study, to our knowledge, to explore the association between a set of mutually exclusive diagnostic categories and risk of unplanned ICU transfer within 24 hours, and it is the first study to identify risk factors for unplanned ICU transfer in a multi‐hospital cohort adjusted for patient‐ and hospital‐level characteristics. We also identified a novel hospital volumeoutcome relationship: Unplanned ICU transfers are up to twice as likely to occur in the smallest volume hospitals compared with highest volume hospitals. Hospital volume has long been proposed as a proxy for hospital resources; there are several studies showing a relationship between low‐volume hospitals and worse outcomes for a number of conditions.19, 20 Possible mechanisms may include decreased ICU capacity, decreased on‐call intensivists in the hospital after hours, and less experience with certain critical care conditions seen more frequently in high‐volume hospitals.21
Patients at risk of unplanned ICU transfer were also more likely to have physiologic derangement identified on laboratory testing, high comorbidity burden, and arrive on the ward between 11 PM and 7 AM. Given the strong correlation between comorbidity burden and physiologic derangement and mortality,14 it is not surprising that the COPS and LAPS were independent predictors of unplanned transfer. It is unclear, however, why arriving on the ward on the overnight shift is associated with higher risk. One possibility is that patients who arrive on the wards during 11 PM to 7 AM are also likely to have been in the ED during evening peak hours most associated with ED crowding.22 High levels of ED crowding have been associated with delays in care, worse quality care, lapses in patient safety, and even increased in‐hospital mortality.22, 23 Other possible reasons include decreased in‐hospital staffing and longer delays in critical diagnostic tests and interventions.2428
Admission to TCUs was associated with decreased risk of unplanned ICU transfer in the first 24 hours of hospitalization. This may be due to the continuous monitoring, decreased nursing‐to‐patient ratios, or the availability to provide some critical care interventions. In our study, age 85 was associated with lower likelihood of unplanned transfer. Unfortunately, we did not have access to data on advanced directives or patient preferences. Data on advanced directives would help to distinguish whether this phenomenon was related to end‐of‐life care goals versus other explanations.
Our study confirms some risk factors identified in previous studies. These include specific diagnoses such as pneumonia and COPD,12, 13, 29 heavy comorbidity burden,12, 13, 29 abnormal labs,29 and male sex.13 Pneumonia has consistently been shown to be a risk factor for unplanned ICU transfer. This may stem from the dynamic nature of this condition and its ability to rapidly progress, and the fact that some ICUs may not accept pneumonia patients unless they demonstrate a need for mechanical ventilation.30 Recently, a prediction rule has been developed to determine which patients with pneumonia are likely to have an unplanned ICU transfer.30 It is possible that with validation and application of this rule, unplanned transfer rates for pneumonia could be reduced. It is unclear whether males have unmeasured factors associated with increased risk of unplanned transfer or whether a true gender disparity exists.
Our findings should be interpreted within the context of this study's limitations. First, this study was not designed to distinguish the underlying cause of the unplanned transfer such as under‐recognition of illness severity in the ED, evolving clinical disease after leaving the ED, or delays in critical interventions on the ward. These are a focus of our ongoing research efforts. Second, while previous studies have demonstrated that our automated risk adjustment variables can accurately predict in‐hospital mortality (0.88 area under curve in external populations),17 additional data on vital signs and mental status could further improve risk adjustment. However, using automated data allowed us to study risk factors for unplanned transfer in a multi‐hospital cohort with a much larger population than has been previously studied. Serial data on vital signs and mental status both in the ED and during hospitalization could also be helpful in determining which unplanned transfers could be prevented with earlier recognition and intervention. Finally, all patient care occurred within an integrated healthcare delivery system. Thus, differences in case‐mix, hospital resources, ICU structure, and geographic location should be considered when applying our results to other healthcare systems.
This study raises several new areas for future research. With access to richer data becoming available in electronic medical records, prediction rules should be developed to enable better triage to appropriate levels of care for ED admissions. Future research should also analyze the comparative effectiveness of intermediate monitored units versus non‐monitored wards for preventing clinical deterioration by admitting diagnosis. Diagnoses that have been shown to have an increased risk of death after unplanned ICU transfer, such as pneumonia/respiratory infection and COPD,1 should be prioritized in this research. Better understanding is needed on the diagnosis‐specific differences and the differences in ED triage process and ICU structure that may explain why high‐volume hospitals have significantly lower rates of early unplanned ICU transfers compared with low‐volume hospitals. In particular, determining the effect of TCU and ICU capacities and census at the time of admission, and comparing patient risk characteristics across hospital‐volume strata would be very useful. Finally, more work is needed to determine whether the higher rate of unplanned transfers during overnight nursing shifts is related to decreased resource availability, preceding ED crowding, or other organizational causes.
In conclusion, patients admitted with respiratory conditions, sepsis, MI, high comorbidity, and abnormal labs are at modestly increased risk of unplanned ICU transfer within 24 hours of admission from the ED. Patients admitted with respiratory conditions (pneumonia/respiratory infections and COPD) accounted for half of the admitting diagnoses that are at increased risk for unplanned ICU transfer. These patients may benefit from better inpatient triage from the ED, earlier intervention, or closer monitoring. More research is needed to determine the specific aspects of care associated with admission to intermediate care units and high‐volume hospitals that reduce the risk of unplanned ICU transfer.
Acknowledgements
The authors thank John D. Greene, Juan Carlos La Guardia, and Benjamin Turk for their assistance with formatting of the dataset; Dr Alan S. Go, Acting Director of the Division of Research, for reviewing the manuscript; and Alina Schnake‐Mahl for formatting the manuscript.
Emergency Department (ED) patients who are hospitalized and require unplanned transfer to the intensive care unit (ICU) within 24 hours of arrival on the ward have higher mortality than direct ICU admissions.1, 2 Previous research found that 5% of ED admissions experienced unplanned ICU transfer during their hospitalization, yet these patients account for 25% of in‐hospital deaths and have a longer length of stay than direct ICU admissions.1, 3 For these reasons, inpatient rapid‐response teams and early warning systems have been studied to reduce the mortality of patients who rapidly deteriorate on the hospital ward.410 However, there is little conclusive evidence that these interventions decrease mortality.710 It is possible that with better recognition and intervention in the ED, a portion of these unplanned ICU transfers and their subsequent adverse outcomes could be prevented.11
Previous research on risk factors for unplanned ICU transfers among ED admissions is limited. While 2 previous studies from non‐US hospitals used administrative data to identify some general populations at risk for unplanned ICU transfer,12, 13 these studies did not differentiate between transfers shortly after admission and those that occurred during a prolonged hospital staya critical distinction since the outcomes between these groups differs substantially.1 Another limitation of these studies is the absence of physiologic measures at ED presentation, which have been shown to be highly predictive of mortality.14
In this study, we describe risk factors for unplanned transfer to the ICU within 24 hours of arrival on the ward, among a large cohort of ED hospitalizations across 13 community hospitals. Focusing on admitting diagnoses most at risk, our goal was to inform efforts to improve the triage of ED admissions and determine which patients may benefit from additional interventions, such as improved resuscitation, closer monitoring, or risk stratification tools. We also hypothesized that higher volume hospitals would have lower rates of unplanned ICU transfers, as these hospitals are more likely have more patient care resources on the hospital ward and a higher threshold to transfer to the ICU.
METHODS
Setting and Patients
The setting for this study was Kaiser Permanente Northern California (KPNC), a large integrated healthcare delivery system serving approximately 3.3 million members.1, 3, 15, 16 We extracted data on all adult ED admissions (18 years old) to the hospital between 2007 and 2009. We excluded patients who went directly to the operating room or the ICU, as well as gynecological/pregnancy‐related admissions, as these patients have substantially different mortality risks.14 ED admissions to hospital wards could either go to medicalsurgical units or transitional care units (TCU), an intermediate level of care between the medicalsurgical units and the ICU. We chose to focus on hospitals with similar inpatient structures. Thus, 8 hospitals without TCUs were excluded, leaving 13 hospitals for analysis. The KPNC Institutional Review Board approved this study.
Main Outcome Measure
The main outcome measure was unplanned transfer to the ICU within 24 hours of arrival to the hospital ward, based upon bed history data. As in previous research, we make the assumptionwhich is supported by the high observed‐to‐expected mortality ratios found in these patientsthat these transfers to the ICU were due to clinical deterioration, and thus were unplanned, rather than a planned transfer to the ICU as is more common after an elective surgical procedure.13 The comparison population was patients admitted from the ED to the ward who never experienced a transfer to the ICU.
Patient and Hospital Characteristics
We extracted patient data on age, sex, admitting diagnosis, chronic illness burden, acute physiologic derangement in the ED, and hospital unit length of stay. Chronic illness was measured using the Comorbidity Point Score (COPS), and physiologic derangement was measured using the Laboratory Acute Physiology Score (LAPS) calculated from labs collected in the ED.1, 14, 17 The derivation of these variables from the electronic medical record has been previously described.14 The COPS was derived from International Classification of Diseases, Ninth Revision (ICD‐9) codes for all Kaiser Permanente Medical Care Program (KPMCP) inpatient and outpatient encounters prior to hospitalization. The LAPS is based on 14 possible lab tests that could be drawn in the ED or in the 72 hours prior to hospitalization. The admitting diagnosis is the ICD‐9 code assigned for the primary diagnosis determined by the admitting physician at the time when hospital admission orders are entered. We further collapsed a previously used categorization of 44 primary condition diagnoses, based on admission ICD‐9 codes,14 into 25 broad diagnostic categories based on pathophysiologic plausibility and mortality rates. We tabulated inpatient admissions originating in the ED to derive a hospital volume measure.
Statistical Analyses
We compared patient characteristics, hospital volume, and outcomes by whether or not an unplanned ICU transfer occurred. Unadjusted analyses were performed with analysis of variance (ANOVA) and chi‐square tests. We calculated crude rates of unplanned ICU transfer per 1,000 ED inpatient admissions by patient characteristics and by hospital, stratified by hospital volume.
We used a hierarchical multivariate logistic regression model to estimate adjusted odds ratios for unplanned ICU transfer as a function of both patient‐level variables (age, sex, COPS, LAPS, time of admission, admission to TCU vs ward, admitting diagnosis) and hospital‐level variables (volume) in the model. We planned to choose the reference group for admitting diagnosis as the one with an unadjusted odds ratio closest to the null (1.00). This model addresses correlations between patients with multiple hospitalizations and clustering by hospital, by fitting random intercepts for these clusters. All analyses were performed in Stata 12 (StataCorp, College Station, TX), and statistics are presented with 95% confidence intervals (CI). The Stata program gllamm (Generalized Linear Latent and Mixed Models) was used for hierarchical modeling.18
RESULTS
Of 178,315 ED non‐ICU hospitalizations meeting inclusion criteria, 4,252 (2.4%) were admitted to the ward and were transferred to the ICU within 24 hours of leaving the ED. There were 122,251 unique patients in our study population. Table 1 compares the characteristics of ED hospitalizations in which an unplanned transfer occurred to those that did not experience an unplanned transfer. Unplanned transfers were more likely to have a higher comorbidity burden, more deranged physiology, and more likely to arrive on the floor during the overnight shift.
| Characteristics | Unplanned Transfer to ICU Within 24 h of Leaving ED? | P Value* | |
|---|---|---|---|
| Yes | No | ||
| N = 4,252 (2.4%) | N = 174,063 (97.6%) | ||
| |||
| Age, median (IQR) | 69 (5680) | 70 (5681) | <0.01 |
| Male, % | 51.3 | 45.9 | <0.01 |
| Comorbidity Points Score (COPS), median (IQR) | 100 (46158) | 89 (42144) | <0.01 |
| Laboratory Acute Physiology Score (LAPS), median (IQR) | 26 (1342) | 18 (633) | <0.01 |
| Nursing shift on arrival to floor, % | |||
| Day: 7 am3 pm (Reference) | 20.1 | 20.1 | NS |
| Evening: 3 pm11 pm | 47.6 | 50.2 | NS |
| Overnight: 11 pm7 am | 32.3 | 29.7 | <0.01 |
| Weekend admission, % | 33.7 | 32.7 | NS |
| Admitted to monitored bed, % | 24.1 | 24.9 | NS |
| Emergency department annual volume, mean (SD) | 48,755 (15,379) | 50,570 (15,276) | <0.01 |
| Non‐ICU annual admission volume, mean (SD) | 5,562 (1,626) | 5,774 (1,568) | <0.01 |
| Admitting diagnosis, listed by descending frequency, % | NS | ||
| Pneumonia and respiratory infections | 16.3 | 11.8 | <0.01 |
| Gastrointestinal bleeding | 12.8 | 13.6 | NS |
| Chest pain | 7.3 | 10.0 | <0.01 |
| Miscellaneous conditions | 5.6 | 6.2 | NS |
| All other acute infections | 4.7 | 6.0 | <0.01 |
| Seizures | 4.1 | 5.9 | <0.01 |
| AMI | 3.9 | 3.3 | <0.05 |
| COPD | 3.8 | 3.0 | <0.01 |
| CHF | 3.5 | 3.7 | NS |
| Arrhythmias and pulmonary embolism | 3.5 | 3.3 | NS |
| Stroke | 3.4 | 3.5 | NS |
| Diabetic emergencies | 3.3 | 2.6 | <0.01 |
| Metabolic, endocrine, electrolytes | 3.0 | 2.9 | NS |
| Sepsis | 3.0 | 1.2 | <0.01 |
| Other neurology and toxicology | 3.0 | 2.9 | NS |
| Urinary tract infections | 2.9 | 3.2 | NS |
| Catastrophic conditions | 2.6 | 1.2 | <0.01 |
| Rheumatology | 2.5 | 3.5 | <0.01 |
| Hematology and oncology | 2.4 | 2.4 | NS |
| Acute renal failure | 1.9 | 1.1 | <0.01 |
| Pancreatic and liver | 1.7 | 2.0 | NS |
| Trauma, fractures, and dislocations | 1.6 | 1.8 | NS |
| Bowel obstructions and diseases | 1.6 | 2.9 | <0.01 |
| Other cardiac conditions | 1.5 | 1.3 | NS |
| Other renal conditions | 0.6 | 1.0 | <0.01 |
| Inpatient length of stay, median days (IQR) | 4.7 (2.78.6) | 2.6 (1.54.4) | <0.01 |
| Died during hospitalization, % | 12.7 | 2.4 | <0.01 |
Unplanned ICU transfers were more frequent in lower volume hospitals (Table 1). Figure 1 displays the inverse relationship between hospital annual ED inpatient admission volume and unplanned ICU transfers rates. The lowest volume hospital had a crude rate twice as high as the 2 highest volume hospitals (39 vs 20, per 1,000 admissions).

Pneumonia/respiratory infection was the most frequent admitting condition associated with unplanned transfer (16.3%) (Table 1). There was also wide variation in crude rates for unplanned ICU transfer by admitting condition (Figure 2). Patients admitted with sepsis had the highest rate (59 per 1,000 admissions), while patients admitted with renal conditions other than acute renal failure had the lowest rates (14.3 per 1,000 admissions).

We confirmed that almost all diagnoses found to account for a disproportionately high share of unplanned ICU transfers in Table 1 were indeed independently associated with this phenomenon after adjustment for patient and hospital differences (Figure 2). Pneumonia remained the most frequent condition associated with unplanned ICU transfer (odds ratio [OR] 1.50; 95% CI 1.201.86). Although less frequent, sepsis had the strongest association of any condition with unplanned transfer (OR 2.51; 95% CI 1.903.31). However, metabolic, endocrine, and electrolyte conditions were no longer associated with unplanned transfer after adjustment, while arrhythmias and pulmonary embolism were. Other conditions confirmed to be associated with increased risk of unplanned transfer included: myocardial infarction (MI), chronic obstructive pulmonary disease (COPD), stroke, diabetic emergencies, catastrophic conditions (includes aortic catastrophes, all forms of shock except septic shock, and intracranial hemorrhage), and acute renal failure. After taking into account the frequency of admitting diagnoses, respiratory conditions (COPD, pneumonia/acute respiratory infection) comprised nearly half (47%) of all conditions associated with increased risk of unplanned ICU transfer.
Other factors confirmed to be independently associated with unplanned ICU transfer included: male sex (OR 1.20; 95% CI 1.131.28), high comorbidity burden as measured by COPS >145 (OR 1.13; 95% CI 1.031.24), increasingly abnormal physiology compared to a LAPS <7, and arrival on ward during the overnight shift (OR 1.10; 95% CI 1.011.21). After adjustment, we did find that admission to the TCU rather than a medicalsurgical unit was associated with decreased risk of unplanned ICU transfer (OR 0.83; 95% CI 0.770.90). Age 85 was associated with decreased risk of unplanned ICU transfer relative to the youngest age group of 1834‐year‐old patients (OR 0.64; 95% CI 0.530.77).
ED admissions to higher volume hospitals were 6% less likely to experience an unplanned transfer for each additional 1,000 annual ED hospitalizations over a lower volume hospital (OR 0.94; 95% CI 0.910.98). In other words, a patient admitted to a hospital with 8,000 annual ED hospitalizations had 30% decreased odds of unplanned ICU transfer compared to a hospital with only 3,000 annual ED hospitalizations.
DISCUSSION
Patients admitted with respiratory conditions accounted for half of all admitting diagnoses associated with increased risk of unplanned transfer to the ICU within 24 hours of arrival to the ward. We found that 1 in 30 ED ward admissions for pneumonia, and 1 in 33 for COPD, were transferred to the ICU within 24 hours. These findings indicate that there is some room for improvement in early care of respiratory conditions, given the average unplanned transfer rate of 1 in 42, and previous research showing that patients with pneumonia and patients with COPD, who experience unplanned ICU transfer, have substantially worse mortality than those directly admitted to the ICU.1
Although less frequent than hospitalizations for respiratory conditions, patients admitted with sepsis were at the highest risk of unplanned ICU transfer (1 in 17 ED non‐ICU hospitalizations). We also found that MI and stroke ward admissions had a higher risk of unplanned ICU transfer. However, we previously found that unplanned ICU transfers for sepsis, MI, and stroke did not have worse mortality than direct ICU admits for these conditions.1 Therefore, quality improvement efforts to reduce excess mortality related to early decompensation in the hospital and unplanned ICU transfer would be most effective if targeted towards respiratory conditions such as pneumonia and COPD.
This is the only in‐depth study, to our knowledge, to explore the association between a set of mutually exclusive diagnostic categories and risk of unplanned ICU transfer within 24 hours, and it is the first study to identify risk factors for unplanned ICU transfer in a multi‐hospital cohort adjusted for patient‐ and hospital‐level characteristics. We also identified a novel hospital volumeoutcome relationship: Unplanned ICU transfers are up to twice as likely to occur in the smallest volume hospitals compared with highest volume hospitals. Hospital volume has long been proposed as a proxy for hospital resources; there are several studies showing a relationship between low‐volume hospitals and worse outcomes for a number of conditions.19, 20 Possible mechanisms may include decreased ICU capacity, decreased on‐call intensivists in the hospital after hours, and less experience with certain critical care conditions seen more frequently in high‐volume hospitals.21
Patients at risk of unplanned ICU transfer were also more likely to have physiologic derangement identified on laboratory testing, high comorbidity burden, and arrive on the ward between 11 PM and 7 AM. Given the strong correlation between comorbidity burden and physiologic derangement and mortality,14 it is not surprising that the COPS and LAPS were independent predictors of unplanned transfer. It is unclear, however, why arriving on the ward on the overnight shift is associated with higher risk. One possibility is that patients who arrive on the wards during 11 PM to 7 AM are also likely to have been in the ED during evening peak hours most associated with ED crowding.22 High levels of ED crowding have been associated with delays in care, worse quality care, lapses in patient safety, and even increased in‐hospital mortality.22, 23 Other possible reasons include decreased in‐hospital staffing and longer delays in critical diagnostic tests and interventions.2428
Admission to TCUs was associated with decreased risk of unplanned ICU transfer in the first 24 hours of hospitalization. This may be due to the continuous monitoring, decreased nursing‐to‐patient ratios, or the availability to provide some critical care interventions. In our study, age 85 was associated with lower likelihood of unplanned transfer. Unfortunately, we did not have access to data on advanced directives or patient preferences. Data on advanced directives would help to distinguish whether this phenomenon was related to end‐of‐life care goals versus other explanations.
Our study confirms some risk factors identified in previous studies. These include specific diagnoses such as pneumonia and COPD,12, 13, 29 heavy comorbidity burden,12, 13, 29 abnormal labs,29 and male sex.13 Pneumonia has consistently been shown to be a risk factor for unplanned ICU transfer. This may stem from the dynamic nature of this condition and its ability to rapidly progress, and the fact that some ICUs may not accept pneumonia patients unless they demonstrate a need for mechanical ventilation.30 Recently, a prediction rule has been developed to determine which patients with pneumonia are likely to have an unplanned ICU transfer.30 It is possible that with validation and application of this rule, unplanned transfer rates for pneumonia could be reduced. It is unclear whether males have unmeasured factors associated with increased risk of unplanned transfer or whether a true gender disparity exists.
Our findings should be interpreted within the context of this study's limitations. First, this study was not designed to distinguish the underlying cause of the unplanned transfer such as under‐recognition of illness severity in the ED, evolving clinical disease after leaving the ED, or delays in critical interventions on the ward. These are a focus of our ongoing research efforts. Second, while previous studies have demonstrated that our automated risk adjustment variables can accurately predict in‐hospital mortality (0.88 area under curve in external populations),17 additional data on vital signs and mental status could further improve risk adjustment. However, using automated data allowed us to study risk factors for unplanned transfer in a multi‐hospital cohort with a much larger population than has been previously studied. Serial data on vital signs and mental status both in the ED and during hospitalization could also be helpful in determining which unplanned transfers could be prevented with earlier recognition and intervention. Finally, all patient care occurred within an integrated healthcare delivery system. Thus, differences in case‐mix, hospital resources, ICU structure, and geographic location should be considered when applying our results to other healthcare systems.
This study raises several new areas for future research. With access to richer data becoming available in electronic medical records, prediction rules should be developed to enable better triage to appropriate levels of care for ED admissions. Future research should also analyze the comparative effectiveness of intermediate monitored units versus non‐monitored wards for preventing clinical deterioration by admitting diagnosis. Diagnoses that have been shown to have an increased risk of death after unplanned ICU transfer, such as pneumonia/respiratory infection and COPD,1 should be prioritized in this research. Better understanding is needed on the diagnosis‐specific differences and the differences in ED triage process and ICU structure that may explain why high‐volume hospitals have significantly lower rates of early unplanned ICU transfers compared with low‐volume hospitals. In particular, determining the effect of TCU and ICU capacities and census at the time of admission, and comparing patient risk characteristics across hospital‐volume strata would be very useful. Finally, more work is needed to determine whether the higher rate of unplanned transfers during overnight nursing shifts is related to decreased resource availability, preceding ED crowding, or other organizational causes.
In conclusion, patients admitted with respiratory conditions, sepsis, MI, high comorbidity, and abnormal labs are at modestly increased risk of unplanned ICU transfer within 24 hours of admission from the ED. Patients admitted with respiratory conditions (pneumonia/respiratory infections and COPD) accounted for half of the admitting diagnoses that are at increased risk for unplanned ICU transfer. These patients may benefit from better inpatient triage from the ED, earlier intervention, or closer monitoring. More research is needed to determine the specific aspects of care associated with admission to intermediate care units and high‐volume hospitals that reduce the risk of unplanned ICU transfer.
Acknowledgements
The authors thank John D. Greene, Juan Carlos La Guardia, and Benjamin Turk for their assistance with formatting of the dataset; Dr Alan S. Go, Acting Director of the Division of Research, for reviewing the manuscript; and Alina Schnake‐Mahl for formatting the manuscript.
- , , , et al. Adverse outcomes associated with delayed intensive care unit transfers in an integrated healthcare system. J Hosp Med. 2011;7(3):224–230.
- , , , et al. Inpatient transfers to the intensive care unit. J Gen Intern Med. 2003;18(2):77–83.
- , , , et al. Intra‐hospital transfers to a higher level of care: contribution to total hospital and intensive care unit (ICU) mortality and length of stay (LOS). J Hosp Med. 2011;6:74–80.
- , , , et al. Hospital‐wide code rates and mortality before and after implementation of a rapid response team. JAMA. 2008;300(21):2506–2513.
- , , , et al. Effect of a rapid response team on hospital‐wide mortality and code rates outside the ICU in a children's hospital. JAMA. 2007;298(19):2267–2274.
- , , , et al. Introduction of the medical emergency team (MET) system: a cluster‐randomised controlled trial. Lancet. 2005;365(9477):2091–2097.
- , , , et al. Rapid response systems: A systematic review. Crit Care Med. 2007;35(5):1238–1243.
- , , , et al. Effects of rapid response systems on clinical outcomes: systematic review and meta‐analysis. J Hosp Med. 2007;2(6):422–432.
- , , , et al. Rapid response teams: a systematic review and meta‐analysis. Arch Intern Med. 2010;170(1):18–26.
- , , , et al. Outreach and early warning systems (EWS) for the prevention of intensive care admission and death of critically ill adult patients on general hospital wards. Cochrane Database Syst Rev. 2007;3:CD005529.
- , , , et al. Unplanned transfers to a medical intensive care unit: Causes and relationship to preventable errors in care. J Hosp Med. 2011;6:68–72.
- , , , et al. Using administrative data to develop a nomogram for individualising risk of unplanned admission to intensive care. Resuscitation. 2008;79(2):241–248.
- , , , et al. Unplanned admission to intensive care after emergency hospitalisation: risk factors and development of a nomogram for individualising risk. Resuscitation. 2009;80(2):224–230.
- , , , et al. Risk‐adjusting hospital inpatient mortality using automated inpatient, outpatient, and laboratory databases. Med Care. 2008;46(3):232–239.
- . Linking automated databases for research in managed care settings. Ann Intern Med. 1997;127(8 pt 2):719–724.
- , , , et al. Risk adjusting community‐acquired pneumonia hospital outcomes using automated databases. Am J Manag Care. 2008;14(3):158–166.
- , , , et al. The Kaiser Permanente inpatient risk adjustment methodology was valid in an external patient population. J Clin Epidemiol. 2011;63(7):798–803.
- , , . Maximum likelihood estimation of limited and discrete dependent variable models with nested random effects. J Econometrics. 2005;128(2):301–323.
- . The relation between volume and outcome in health care. N Engl J Med. 1999;340(21):1677–1679.
- , , . Is volume related to outcome in health care? A systematic review and methodologic critique of the literature. Ann Intern Med. 2002;137(6):511–520.
- , , . Working with capacity limitations: operations management in critical care. Crit Care. 2011;15(4):308.
- , . Systematic review of emergency department crowding: causes, effects, and solutions. Ann Intern Med. 2008;52(2):126–136.
- , , , et al. The effect of emergency department crowding on clinically oriented outcomes. Acad Emerg Med. 2009;16(1):1–10.
- , , , et al. Association between time of admission to the ICU and mortality. Chest. 2010;138(1):68–75.
- , , , et al. Off‐hour admission and in‐hospital stroke case fatality in the get with the guidelines‐stroke program. Stroke. 2009;40(2):569–576.
- , , , et al. Relationship between time of day, day of week, timeliness of reperfusion, and in‐hospital mortality for patients with acute ST‐segment elevation myocardial infarction. JAMA. 2005;294(7):803–812.
- , , , et al. Hospital mortality among adults admitted to and discharged from intensive care on weekends and evenings. J Crit Care. 2008;23(3):317–324.
- , , , et al. Association between ICU admission during morning rounds and mortality. Chest. 2009;136(6):1489–1495.
- , , , et al. Identifying infected emergency department patients admitted to the hospital ward at risk of clinical deterioration and intensive care unit transfer. Acad Emerg Med. 2010;17(10):1080–1085.
- , , , et al. Risk stratification of early admission to the intensive care unit of patients with no major criteria of severe community‐acquired pneumonia: development of an international prediction rule. Crit Care. 2009;13(2):R54.
- , , , et al. Adverse outcomes associated with delayed intensive care unit transfers in an integrated healthcare system. J Hosp Med. 2011;7(3):224–230.
- , , , et al. Inpatient transfers to the intensive care unit. J Gen Intern Med. 2003;18(2):77–83.
- , , , et al. Intra‐hospital transfers to a higher level of care: contribution to total hospital and intensive care unit (ICU) mortality and length of stay (LOS). J Hosp Med. 2011;6:74–80.
- , , , et al. Hospital‐wide code rates and mortality before and after implementation of a rapid response team. JAMA. 2008;300(21):2506–2513.
- , , , et al. Effect of a rapid response team on hospital‐wide mortality and code rates outside the ICU in a children's hospital. JAMA. 2007;298(19):2267–2274.
- , , , et al. Introduction of the medical emergency team (MET) system: a cluster‐randomised controlled trial. Lancet. 2005;365(9477):2091–2097.
- , , , et al. Rapid response systems: A systematic review. Crit Care Med. 2007;35(5):1238–1243.
- , , , et al. Effects of rapid response systems on clinical outcomes: systematic review and meta‐analysis. J Hosp Med. 2007;2(6):422–432.
- , , , et al. Rapid response teams: a systematic review and meta‐analysis. Arch Intern Med. 2010;170(1):18–26.
- , , , et al. Outreach and early warning systems (EWS) for the prevention of intensive care admission and death of critically ill adult patients on general hospital wards. Cochrane Database Syst Rev. 2007;3:CD005529.
- , , , et al. Unplanned transfers to a medical intensive care unit: Causes and relationship to preventable errors in care. J Hosp Med. 2011;6:68–72.
- , , , et al. Using administrative data to develop a nomogram for individualising risk of unplanned admission to intensive care. Resuscitation. 2008;79(2):241–248.
- , , , et al. Unplanned admission to intensive care after emergency hospitalisation: risk factors and development of a nomogram for individualising risk. Resuscitation. 2009;80(2):224–230.
- , , , et al. Risk‐adjusting hospital inpatient mortality using automated inpatient, outpatient, and laboratory databases. Med Care. 2008;46(3):232–239.
- . Linking automated databases for research in managed care settings. Ann Intern Med. 1997;127(8 pt 2):719–724.
- , , , et al. Risk adjusting community‐acquired pneumonia hospital outcomes using automated databases. Am J Manag Care. 2008;14(3):158–166.
- , , , et al. The Kaiser Permanente inpatient risk adjustment methodology was valid in an external patient population. J Clin Epidemiol. 2011;63(7):798–803.
- , , . Maximum likelihood estimation of limited and discrete dependent variable models with nested random effects. J Econometrics. 2005;128(2):301–323.
- . The relation between volume and outcome in health care. N Engl J Med. 1999;340(21):1677–1679.
- , , . Is volume related to outcome in health care? A systematic review and methodologic critique of the literature. Ann Intern Med. 2002;137(6):511–520.
- , , . Working with capacity limitations: operations management in critical care. Crit Care. 2011;15(4):308.
- , . Systematic review of emergency department crowding: causes, effects, and solutions. Ann Intern Med. 2008;52(2):126–136.
- , , , et al. The effect of emergency department crowding on clinically oriented outcomes. Acad Emerg Med. 2009;16(1):1–10.
- , , , et al. Association between time of admission to the ICU and mortality. Chest. 2010;138(1):68–75.
- , , , et al. Off‐hour admission and in‐hospital stroke case fatality in the get with the guidelines‐stroke program. Stroke. 2009;40(2):569–576.
- , , , et al. Relationship between time of day, day of week, timeliness of reperfusion, and in‐hospital mortality for patients with acute ST‐segment elevation myocardial infarction. JAMA. 2005;294(7):803–812.
- , , , et al. Hospital mortality among adults admitted to and discharged from intensive care on weekends and evenings. J Crit Care. 2008;23(3):317–324.
- , , , et al. Association between ICU admission during morning rounds and mortality. Chest. 2009;136(6):1489–1495.
- , , , et al. Identifying infected emergency department patients admitted to the hospital ward at risk of clinical deterioration and intensive care unit transfer. Acad Emerg Med. 2010;17(10):1080–1085.
- , , , et al. Risk stratification of early admission to the intensive care unit of patients with no major criteria of severe community‐acquired pneumonia: development of an international prediction rule. Crit Care. 2009;13(2):R54.
Copyright © 2012 Society of Hospital Medicine
H1N1/09—Fever and Hospital Presentation
The Centers for Disease Control and Prevention (CDC) established a case definition during the 2009 influenza season for influenza‐like illness (ILI): a temperature of 100.0F (37.8C), oral or equivalent, and cough or sore throat, in the absence of a known cause other than influenza.1 Even though this definition is currently being revised, fever is still listed as a predominant symptom for influenza on the CDC Web site.2, 3
During the spring of 2009 in New York City, 95% of patients with pandemic hemagglutinin type 1 and neuraminidase type 1 (H1N1) influenza A met the case definition for ILI,4 whereas a report from Mexico5 and another from China6 reported that about one‐third of patients presented without fever. A recent study found that the clinical features of H1N1/09 influenza and seasonal strains were similar in hospitalized patients.7 Until recently, the CDC case definition for ILI included fever as a requisite symptom. Most clinicians still consider influenza a fever‐based illness.8, 9 Some studies have reported that seeking early medical care and early treatment for influenza resulted in reduced severity and duration of illness.1013 However, individuals without fever may not have sought early care because their illnesses did not meet all the criteria for ILI.
We hypothesized that significant number of our patients hospitalized during the H1N1/09 influenza pandemic did not have fever as part of the influenza‐like illness prior to hospital presentation. We further hypothesized that this absence of fever was associated with delayed presentation to the hospital and delayed treatment. No prior studies have assessed the effect of fever on hospital presentation, diagnosis, and treatment of H1N1/09 patients. We performed a retrospective cohort analysis comparing H1N1/09 patients with fever as part of their illness prior to hospital presentation to those without fever, to assess the influence of fever on hospital presentation and on diagnosis and treatment in hospitalized patients with laboratory‐confirmed H1N1/09 infection.
METHODS
Study Design and Setting
This retrospective cohort study was conducted at a tertiary care center with 1159 beds divided between 2 general hospitals (634 and 167 beds), a large cancer center (208 beds), and an inpatient cardiac hospital (150 beds).
Selection of Participants
During the pandemic influenza season, clinicians throughout the tertiary care center were encouraged to obtain nasopharyngeal swabs to screen for influenza from all patients presenting to the hospital with respiratory illness, unexplained gastrointestinal symptoms, ILI, or any suspicion for influenza infection based on physician discretion. Patients were admitted through routine hospital admission procedures and triage. Physicians were encouraged to treat all patients in whom pandemic H1N1/09 was suspected empirically with an antiviral drug (oseltamivir). Patients with a positive H1N1/09 test were then continued on treatment. Obese patients, critically ill patients requiring intensive care unit (ICU) care, pregnant women, and patients with chronic medical conditions such as chronic respiratory disease, chronic kidney disease, diabetes mellitus, and immune‐compromising conditions were treated with a higher dose of oseltamivir (150 mg twice a day). Other patients were treated with the standard dose of oseltamivir (75 mg twice a day). The present analyses include all patients at least 18 years of age, hospitalized with laboratory‐confirmed pandemic H1N1/09 virus infection from 1 June 2009 through 31 December 2009. Subjects younger than 18 years and prisoners were excluded.
Data Collection
Laboratory‐confirmed pandemic H1N1/09 patients were identified from the hospital's infection control database. After identifying eligible patients, physician investigators accessed each electronic medical record to obtain health history, diagnosis, test results, disposition, and clinical outcome information. We captured data on gender, race, age, comorbidities, symptoms and onset of illness, hospital presentation, diagnosis, treatment, ICU admission, and death. Data abstraction was performed by an infectious disease physician using a standardized form, and all data abstraction was validated by another infectious disease physician. The research was deemed exempt from further review by the Ohio State University Institutional Review Board (2009E0979).
Subjects with laboratory‐confirmed H1N1/09 virus infection were identified if nasal swab or respiratory secretions were positive for novel influenza A (H1N1) by specific rapid antigen or culture testing. Nasal swabs that were processed for confirmatory influenza testing by polymerase chain reaction (PCR) were confirmed by panel (xTAG) or specific influenza A and B PCR (Luminex Molecular Diagnostics, Toronto, CA).14 Positive tests were confirmed using the Prodesse ProFlu‐ST, Influenza A [2009] real‐time PCR (H1N1 subtyping) assay (Focus Diagnostics, Cypress, CA).
Day of onset of illness was defined as the day when the patient started developing one or more symptoms of influenza‐like illness. Day of hospital presentation was defined as the day when the patient either presented to the emergency department or a physician's office and was subsequently admitted or was directly admitted to the hospital. Fever was defined as a temperature of 100F (37.8C) or greater at presentation, or reported subjective fever upon presentation, and reported antipyretic use (nonsteroidal anti‐inflammatory drugs or acetaminophen) prior to admission. When multiple temperature readings were present in the emergency department record, any recorded temperature of 100F or greater was considered a fever. The Charlson comorbidity index predicts the 10‐year mortality for a patient with a range of comorbid conditions such as heart disease, acquired immune deficiency syndrome (AIDS), or cancer. The Charlson comorbidity index was calculated according to previously described methods.1517 Diabetes, chronic respiratory disease (asthma, chronic obstructive pulmonary disease), and chronic kidney disease with hemodialysis were recorded as comorbidities if these diagnoses were documented in the patient notes. Immunosuppression was defined as oral steroid use or other immunosuppressive medication, organ transplantation, human immunodeficiency virus (HIV) infection, or cancer chemotherapy. Empiric antibiotic treatment was defined as treatment with antibiotics regardless of the evidence of bacterial infection. Proven bacterial coinfection was defined as infection with bacterial pathogens documented by a positive bacterial culture from any site that was obtained in response to clinical suspicion of bacterial infection.
Data Analysis
Demographic and clinical characteristics were summarized for all patients, and for those patients with or without fever, as part of their illness prior to hospital presentation. Continuous variables were summarized as mean ( standard deviation) or median values. For categorical variables, the percentage of patients in each category was calculated. For comparison between patients with fever and those with no fever, for continuous variables, the 2‐sample t test for normally distributed data or the Wilcoxon rank‐sum test for non‐normal data was used. The Pearson chi‐square test was used for categorical data. We performed a logistic regression analysis to examine the association between fever and time to presentation and ICU admission, adjusting for patient age and Charlson index. All analyses were carried out using SAS 9.2 software (SAS, Cary, NC).
RESULTS
We identified 135 hospitalized patients with laboratory‐confirmed pandemic H1N1 virus infection during the study period. A substantial number of patients (n = 56, 42%) had no fever as part of their illness prior to hospital presentation. Thirty‐one patients (23%) required treatment in an ICU during hospitalization, and 9 (7%) died. While we observed no differences between patients with fever and those without fever prior to hospital presentation in gender, race, or age (Table 1), patients without fever had higher Charlson index compared to those with fever (P = 0.01, Wilcoxon rank‐sum test). Patients without fever also had significantly longer time to presentation to the hospital since the onset of illness, compared to those with fever (median of 4 vs 2 days, P < 0.001). Patients without fever also had significantly longer time to initiation of proper treatment since the onset of illness, compared to those with fever (median of 5 vs 2 days, P = 0.001).
| Fever | No Fever | Unadjusted Odds Ratio | P Value* | ||
|---|---|---|---|---|---|
| N = 79 (59%) | N = 56 (42%) | ||||
| |||||
| Gender | Female, n (%) | 41 (52) | 34 (61) | 0.7 | 0.31 |
| Race | White, n (%) | 45 (57) | 29 (52) | 1.03 | 0.30 |
| Black, n (%) | 30 (38) | 20 (36) | |||
| Other, n (%) | 4 (5) | 7 (13) | |||
| Age | Mean SD (range) | 4315 (1889) | 48 17 (1989) | 0.060 | |
| Charlson index | Median (range) | 0 (0, 13) | 1 (0, 13) | 0.010 | |
| Comorbidities | |||||
| Obese (BMI 30) | n (%) | 37 (50) | 29 (54) | 0.82 | 0.571 |
| Chronic respiratory illness (asthma, COPD) | n (%) | 19 (24) | 13 (23) | 1.05 | 0.910 |
| Diabetes | n (%) | 14 (18) | 19 (34) | 0.419 | 0.031 |
| CKD with hemodialysis | n (%) | 12 (15) | 6 (11) | 1.49 | 0.451 |
| Immunocompromised | n (%) | 29 (38) | 13 (23) | 1.92 | 0.095 |
| Days from onset to hospital presentation | Median (range) | 2 (010) | 4 (121) | 0.0001 | |
| No. with days >2 | n (%) | 31 (39) | 40 (71) | 0.26 | <0.001 |
| Days to diagnosis from hospital presentation | Median (range) | 0 (03) | 0 (05) | 0.166 | |
| No. with days >2 | n (%) | 1 (1) | 3 (5) | 0.24 | 0.170 |
| Days to oseltamivir since admission | Median (range) | 0 (013) | 1 (014) | 0.07 | |
| Days to oseltamivir since onset of illness | Median (range) | 2 (013) | 5 (119) | 0.0003 | |
| No. with days >2 | n (%) | 35 (44) | 39 (70) | 0.24 | 0.001 |
| Need for ICU care | n (%) | 12 (15) | 19 (34) | 0.35 | 0.010 |
| Death | n (%) | 6 (8) | 3 (5) | 1.45 | 0.061 |
| Bacterial coinfection | n (%) | 5 (n = 79; 6%) | 9 (15) | 0.35 | 0.067 |
| Empiric antibiotics | 65 (n = 77; 84%) | 33 (59) | 3.24 | 0.003 | |
| Lower respiratory symptoms | n (%) | 72 (91) | 50 (91) | 1.03 | 0.963 |
| Cough | n (%) | 58 (73) | 38 (69) | 1.24 | 0.585 |
| Shortness of breath | n (%) | 34 (43) | 25 (46) | 0.91 | 0.782 |
| Respiratory failure | n (%) | 7 (9) | 7 (13) | 0.67 | 0.472 |
| Sore throat/congestion/rhinorrhea | n (%) | 21 (27) | 10 (18) | 1.63 | 0.257 |
| Gastrointestinal symptoms (nausea, vomiting, diarrhea) | n (%) | 30 (38) | 13 (24) | 1.98 | 0.080 |
Patients were rapidly diagnosed and treated after presentation to the hospital with no significant difference by fever status at presentation (P = 0.17 and 0.07 for differences in time from hospital presentation to diagnosis and treatment, respectively). While 82% (65/79) of patients with fever received empiric antibiotic treatment, only 59% (33/56) of those with no fever received empiric antibiotic treatment (P = 0.003). Prevalence of bacterial coinfection was similar between the 2 groups (5% in the fever group vs 9% in the group without fever; P = 0.067). Patients without fever were more frequently in an ICU than those with fever (P = 0.01), but we observed no significant differences in mortality by fever status at presentation in the small number of deaths during the study period (P = 0.61). Compared to patients with no fever, after adjustment for age (<40 vs 40) and Charlson index (0, 12, 3), patients with fever had significantly decreased likelihood of late hospital presentation (>2 days from the onset of illness) (adjusted odds ratio [OR]: 0.27, P = 0.001). Patients with fever also had decreased likelihood of ICU stay compared to patients with no fever, after adjustment for age and Charlson index (adjusted OR: 0.42, P = 0.05) (Table 2).
| Adjusted Odds Ratio | P Value | |
|---|---|---|
| ||
| Late hospital presentation (>2 d from onset of illness) | ||
| Fever (vs no fever) | 0.27 | 0.001 |
| Age (40 y) | 2.06 | 0.071 |
| Charlson index | ||
| 12 | 0.69 | 0.38 |
| 3 | 1.01 | 0.99 |
| ICU admission | ||
| Fever (vs no fever) | 0.42 | 0.05 |
| Age (40 y) | 1.21 | 0.701 |
| Charlson index | ||
| 12 | 0.66 | 0.448 |
| 3 | 4.02 | 0.016 |
Patients who presented to the hospital more than 2 days after the onset of illness were significantly more likely to be treated in an ICU during their hospitalization (P < 0.001). Among 71 patients who presented to the hospital more than 2 days after the onset of illness, 27 patients were treated in an ICU and 44 patients did not require ICU care. Among 64 patients who presented to the hospital within 2 days of the onset of illness, 4 patients were treated in an ICU and 60 patients did not require ICU care. Those who presented to the hospital more than 2 days after the onset of illness also had a nonsignificantly higher death rate: 7 of 71 (10%) versus 2 of 64 (3%). Among the comorbidities tested, patients with no fever were significantly more likely to have diabetes mellitus (P = 0.03).
ICU patients had a slightly higher median Charlson index than those who did not receive ICU care (median score 1 vs score 0 for ICU patients vs non‐ICU patients, respectively; P = 0.052). We observed no difference in median Charlson index between patients with late hospital presentation (>2 days from onset of illness) and those with early hospital presentation (2 days from onset of illness) (P = 0.51).
DISCUSSION
We present the first study, to our knowledge, to assess the influence of presence of fever, as part of the illness prior to hospital presentation, on time to presentation and treatment for H1N1/09 patients. By comparing patients who had fever with those who did not, we showed that patients with fever tended to seek medical care sooner and thus received treatment sooner. Those without fever may not have realized they needed to seek medical care until much later in the course of disease compared to those with fever. Given that ICU stay was significantly more likely in patients with late presentation and no fever, these findings have important medical and economic consequences for individual patients and healthcare systems. Our findings, in conjunction with previous studies indicating reduced disease severity with early treatment of influenza,1013, can be used to educate the public to seek medical care for influenza early, even in the absence of fever.
Our study confirms previous observations that a significant number of patients with H1N1/09 did not have fever.5, 6 Nevertheless, fever appeared to trigger initiation of empiric antibiotic treatment. Empiric antibiotic use was significantly higher in the fever group. There was no difference in the median Charlson index among those who received antibiotic treatment and those who did not, suggesting that comorbidities are unlikely to explain the observed association. Empiric antibiotic treatment appeared not to affect whether or not a patient required ICU care during the hospitalization, though this association is difficult to measure because the majority of ICU patients (84%) were admitted to an ICU on the same day as hospital admission. We saw no difference in laboratory‐confirmed bacterial coinfection between patients presenting with and without fever. We did not observe a significant effect of late presentation or fever on mortality, though the small number of deaths in the study likely limited our ability to detect any association.
Recent studies have shown that clinical signs cannot reliably differentiate H1N1/09‐positive and H1N1/09‐negative patients.18, 19 In our study, patients in the fever and non‐fever groups received prompt diagnosis and treatment once they presented to the hospital. This may be because clinicians were encouraged to test patients based on any suspicion for influenza infection and to treat for influenza while waiting for the result of testing. This may explain the similar timeframes for diagnosis and treatment between the fever and non‐fever groups.
Limitations
Our study took place in a single medical center, and our findings may not be generalizable to other clinical settings. However, this large tertiary care center admits 25% of patients in central Ohio. Our study focused on hospitalized patients only and excluded patients who may have presented to the emergency department with H1N1/09 but were not hospitalized. Thus, our findings can be applied to patients with severe enough disease to warrant hospitalization. It is possible that patients without fever presented early (within 2 days of onset of illness) and were treated as outpatients; these individuals would not have been captured in the current analysis. However, our primary goal was to assess the effect of fever on the overall course of H1N1/09 presentation, diagnosis, and management in hospitalized patients. Provider bias may have played a role in the decision to admit patients to the hospital, but clinicians were encouraged to use a standardized protocol to drive decisions to test and admit. We studied H1N1/09 patients only, and our findings may not be generalizable to patients infected with other influenza types. However, H1N1/09 influenza symptoms are similar to other types of influenza. We also did not collect data on insurance status which may play a role in patients' choice to seek hospital care.
Strengths
Our study also has substantial strengths. Because clinicians tested all patients with symptoms of respiratory illness and unexplained gastrointestinal symptoms irrespective of the presence of fever, we were able to assess the proportion of H1N1/09 patients without fever as part of their illness and to determine that the most common presenting symptoms were respiratory. Our analysis includes only patients with laboratory‐confirmed H1N1/09 infection. Our surveillance was strengthened by the availability of real‐time PCR confirmatory testing performed in our own molecular microbiology laboratory.
Conclusions
In this study, nearly half of patients with H1N1/09 requiring hospitalization did not have fever as part of their illness. Patients with fever tended to seek medical care sooner and thus received treatment sooner. Patients with late presentation and no fever were more likely to need ICU admission. Our findings reinforce previous studies that indicate better outcomes with early treatment of influenza.1013 These findings can be used to prompt clinicians to consider treating hospitalized patients with influenza and to educate the public to seek medical care soon after the onset of illness, despite absence of fever, if symptoms of respiratory illness and unexplained gastrointestinal symptoms are present during the influenza season.
- CDC. Update: infections with a swine‐origin influenza A (H1N1) virus—United States and other countries, April 28, 2009. MMWR.2009;58:433–434.
- United States Centers for Disease Control and Prevention. Clinical Signs and Symptoms of Influenza. Available at: http://www.cdc.gov/flu/professionals/acip/clinical.htm#signs. Accessed June 11, 2012.
- United States Centers for Disease Control and Prevention. Cold Versus Flu. Available at: http://www.cdc.gov/flu/about/qa/coldflu.htm. Accessed June 11, 2012.
- CDC. Swine‐origin influenza A (H1N1) virus infections in a school—New York City, April 30, 2009. MMWR. 2009;58(Dispatch);1–3.
- The New York Times. Many swine flu cases have no fever. Available at: http://www.nytimes.com/2009/05/13/health/13fever.html?_r=1361(26):2507–2517.
- , , , et al. Clinical and epidemiological profile of patients with severe H1N1/09 pandemic influenza in Australia and New Zealand: an observational cohort study. BMJ Open.2011;1(1). Available at: http://dx.doi.org/10.1136/bmjopen‐2011‐000100.
- Colds and the Flu. Available at: http://familydoctor.org/familydoctor/en/diseases‐conditions/colds‐and‐the‐flu/symptoms.html. Accessed June 11, 2012.
- Flu or Cold Symptoms? Available at: http://www.webmd.com/cold‐and‐flu/cold‐guide/flu‐cold‐symptoms. Accessed June 11, 2012.
- . Neuraminidase inhibitors for influenza. N Engl J Med. 2005;353:1363–1373.
- , , , et al. Early administration of oral oseltamivir increases the benefits of influenza treatment. J Antimicrob Chemother. 2003;51:123–129.
- , , , et al. Factors influencing the effectiveness of oseltamivir and amantadine for the treatment of influenza: a multicenter study from Japan of the 2002‐2003 influenza season. Clin Infect Dis. 2005;40:1309–1316.
- , , , et al. Use of oseltamivir during influenza outbreaks in Ontario nursing homes, 1999‐2000. J Am Geriatr Soc. 2002;50:608–616.
- , . Likelihood that an unsubtypeable influenza A virus result obtained with the Luminex xTAG respiratory virus panel is indicative of infection with novel A/H1N1 (swine‐like) influenza virus. J Clin Microbiol. 2009;47:2347–2348.
- , , , . A new method of classifying prognostic comorbidity in longitudinal studies: development and validation. J Chronic Dis. 1987;40(5):373–383.
- , , , . A systematic review of the Charlson comorbidity index using Canadian administrative databases: a perspective on risk adjustment in critical care research. J Crit Care. 2005;20(1):12–19.
- , , . Adapting a clinical comorbidity index for use with ICD‐9‐CM administrative databases. J Clin Epidemiol. 1992;45(6):613–619.
- , , , , , . Characteristics of outpatients with pandemic H1N1/09 influenza in a tertiary care university hospital in Korea. Yonsei Med J. 2012;53(1):213–220.
- , , , et al. Comparison of clinical presentation of respiratory tract infections in H1N1/09‐positive and H1N1/09‐negative patients. Eur J Pediatr. 2012;171(1):159–166.
The Centers for Disease Control and Prevention (CDC) established a case definition during the 2009 influenza season for influenza‐like illness (ILI): a temperature of 100.0F (37.8C), oral or equivalent, and cough or sore throat, in the absence of a known cause other than influenza.1 Even though this definition is currently being revised, fever is still listed as a predominant symptom for influenza on the CDC Web site.2, 3
During the spring of 2009 in New York City, 95% of patients with pandemic hemagglutinin type 1 and neuraminidase type 1 (H1N1) influenza A met the case definition for ILI,4 whereas a report from Mexico5 and another from China6 reported that about one‐third of patients presented without fever. A recent study found that the clinical features of H1N1/09 influenza and seasonal strains were similar in hospitalized patients.7 Until recently, the CDC case definition for ILI included fever as a requisite symptom. Most clinicians still consider influenza a fever‐based illness.8, 9 Some studies have reported that seeking early medical care and early treatment for influenza resulted in reduced severity and duration of illness.1013 However, individuals without fever may not have sought early care because their illnesses did not meet all the criteria for ILI.
We hypothesized that significant number of our patients hospitalized during the H1N1/09 influenza pandemic did not have fever as part of the influenza‐like illness prior to hospital presentation. We further hypothesized that this absence of fever was associated with delayed presentation to the hospital and delayed treatment. No prior studies have assessed the effect of fever on hospital presentation, diagnosis, and treatment of H1N1/09 patients. We performed a retrospective cohort analysis comparing H1N1/09 patients with fever as part of their illness prior to hospital presentation to those without fever, to assess the influence of fever on hospital presentation and on diagnosis and treatment in hospitalized patients with laboratory‐confirmed H1N1/09 infection.
METHODS
Study Design and Setting
This retrospective cohort study was conducted at a tertiary care center with 1159 beds divided between 2 general hospitals (634 and 167 beds), a large cancer center (208 beds), and an inpatient cardiac hospital (150 beds).
Selection of Participants
During the pandemic influenza season, clinicians throughout the tertiary care center were encouraged to obtain nasopharyngeal swabs to screen for influenza from all patients presenting to the hospital with respiratory illness, unexplained gastrointestinal symptoms, ILI, or any suspicion for influenza infection based on physician discretion. Patients were admitted through routine hospital admission procedures and triage. Physicians were encouraged to treat all patients in whom pandemic H1N1/09 was suspected empirically with an antiviral drug (oseltamivir). Patients with a positive H1N1/09 test were then continued on treatment. Obese patients, critically ill patients requiring intensive care unit (ICU) care, pregnant women, and patients with chronic medical conditions such as chronic respiratory disease, chronic kidney disease, diabetes mellitus, and immune‐compromising conditions were treated with a higher dose of oseltamivir (150 mg twice a day). Other patients were treated with the standard dose of oseltamivir (75 mg twice a day). The present analyses include all patients at least 18 years of age, hospitalized with laboratory‐confirmed pandemic H1N1/09 virus infection from 1 June 2009 through 31 December 2009. Subjects younger than 18 years and prisoners were excluded.
Data Collection
Laboratory‐confirmed pandemic H1N1/09 patients were identified from the hospital's infection control database. After identifying eligible patients, physician investigators accessed each electronic medical record to obtain health history, diagnosis, test results, disposition, and clinical outcome information. We captured data on gender, race, age, comorbidities, symptoms and onset of illness, hospital presentation, diagnosis, treatment, ICU admission, and death. Data abstraction was performed by an infectious disease physician using a standardized form, and all data abstraction was validated by another infectious disease physician. The research was deemed exempt from further review by the Ohio State University Institutional Review Board (2009E0979).
Subjects with laboratory‐confirmed H1N1/09 virus infection were identified if nasal swab or respiratory secretions were positive for novel influenza A (H1N1) by specific rapid antigen or culture testing. Nasal swabs that were processed for confirmatory influenza testing by polymerase chain reaction (PCR) were confirmed by panel (xTAG) or specific influenza A and B PCR (Luminex Molecular Diagnostics, Toronto, CA).14 Positive tests were confirmed using the Prodesse ProFlu‐ST, Influenza A [2009] real‐time PCR (H1N1 subtyping) assay (Focus Diagnostics, Cypress, CA).
Day of onset of illness was defined as the day when the patient started developing one or more symptoms of influenza‐like illness. Day of hospital presentation was defined as the day when the patient either presented to the emergency department or a physician's office and was subsequently admitted or was directly admitted to the hospital. Fever was defined as a temperature of 100F (37.8C) or greater at presentation, or reported subjective fever upon presentation, and reported antipyretic use (nonsteroidal anti‐inflammatory drugs or acetaminophen) prior to admission. When multiple temperature readings were present in the emergency department record, any recorded temperature of 100F or greater was considered a fever. The Charlson comorbidity index predicts the 10‐year mortality for a patient with a range of comorbid conditions such as heart disease, acquired immune deficiency syndrome (AIDS), or cancer. The Charlson comorbidity index was calculated according to previously described methods.1517 Diabetes, chronic respiratory disease (asthma, chronic obstructive pulmonary disease), and chronic kidney disease with hemodialysis were recorded as comorbidities if these diagnoses were documented in the patient notes. Immunosuppression was defined as oral steroid use or other immunosuppressive medication, organ transplantation, human immunodeficiency virus (HIV) infection, or cancer chemotherapy. Empiric antibiotic treatment was defined as treatment with antibiotics regardless of the evidence of bacterial infection. Proven bacterial coinfection was defined as infection with bacterial pathogens documented by a positive bacterial culture from any site that was obtained in response to clinical suspicion of bacterial infection.
Data Analysis
Demographic and clinical characteristics were summarized for all patients, and for those patients with or without fever, as part of their illness prior to hospital presentation. Continuous variables were summarized as mean ( standard deviation) or median values. For categorical variables, the percentage of patients in each category was calculated. For comparison between patients with fever and those with no fever, for continuous variables, the 2‐sample t test for normally distributed data or the Wilcoxon rank‐sum test for non‐normal data was used. The Pearson chi‐square test was used for categorical data. We performed a logistic regression analysis to examine the association between fever and time to presentation and ICU admission, adjusting for patient age and Charlson index. All analyses were carried out using SAS 9.2 software (SAS, Cary, NC).
RESULTS
We identified 135 hospitalized patients with laboratory‐confirmed pandemic H1N1 virus infection during the study period. A substantial number of patients (n = 56, 42%) had no fever as part of their illness prior to hospital presentation. Thirty‐one patients (23%) required treatment in an ICU during hospitalization, and 9 (7%) died. While we observed no differences between patients with fever and those without fever prior to hospital presentation in gender, race, or age (Table 1), patients without fever had higher Charlson index compared to those with fever (P = 0.01, Wilcoxon rank‐sum test). Patients without fever also had significantly longer time to presentation to the hospital since the onset of illness, compared to those with fever (median of 4 vs 2 days, P < 0.001). Patients without fever also had significantly longer time to initiation of proper treatment since the onset of illness, compared to those with fever (median of 5 vs 2 days, P = 0.001).
| Fever | No Fever | Unadjusted Odds Ratio | P Value* | ||
|---|---|---|---|---|---|
| N = 79 (59%) | N = 56 (42%) | ||||
| |||||
| Gender | Female, n (%) | 41 (52) | 34 (61) | 0.7 | 0.31 |
| Race | White, n (%) | 45 (57) | 29 (52) | 1.03 | 0.30 |
| Black, n (%) | 30 (38) | 20 (36) | |||
| Other, n (%) | 4 (5) | 7 (13) | |||
| Age | Mean SD (range) | 4315 (1889) | 48 17 (1989) | 0.060 | |
| Charlson index | Median (range) | 0 (0, 13) | 1 (0, 13) | 0.010 | |
| Comorbidities | |||||
| Obese (BMI 30) | n (%) | 37 (50) | 29 (54) | 0.82 | 0.571 |
| Chronic respiratory illness (asthma, COPD) | n (%) | 19 (24) | 13 (23) | 1.05 | 0.910 |
| Diabetes | n (%) | 14 (18) | 19 (34) | 0.419 | 0.031 |
| CKD with hemodialysis | n (%) | 12 (15) | 6 (11) | 1.49 | 0.451 |
| Immunocompromised | n (%) | 29 (38) | 13 (23) | 1.92 | 0.095 |
| Days from onset to hospital presentation | Median (range) | 2 (010) | 4 (121) | 0.0001 | |
| No. with days >2 | n (%) | 31 (39) | 40 (71) | 0.26 | <0.001 |
| Days to diagnosis from hospital presentation | Median (range) | 0 (03) | 0 (05) | 0.166 | |
| No. with days >2 | n (%) | 1 (1) | 3 (5) | 0.24 | 0.170 |
| Days to oseltamivir since admission | Median (range) | 0 (013) | 1 (014) | 0.07 | |
| Days to oseltamivir since onset of illness | Median (range) | 2 (013) | 5 (119) | 0.0003 | |
| No. with days >2 | n (%) | 35 (44) | 39 (70) | 0.24 | 0.001 |
| Need for ICU care | n (%) | 12 (15) | 19 (34) | 0.35 | 0.010 |
| Death | n (%) | 6 (8) | 3 (5) | 1.45 | 0.061 |
| Bacterial coinfection | n (%) | 5 (n = 79; 6%) | 9 (15) | 0.35 | 0.067 |
| Empiric antibiotics | 65 (n = 77; 84%) | 33 (59) | 3.24 | 0.003 | |
| Lower respiratory symptoms | n (%) | 72 (91) | 50 (91) | 1.03 | 0.963 |
| Cough | n (%) | 58 (73) | 38 (69) | 1.24 | 0.585 |
| Shortness of breath | n (%) | 34 (43) | 25 (46) | 0.91 | 0.782 |
| Respiratory failure | n (%) | 7 (9) | 7 (13) | 0.67 | 0.472 |
| Sore throat/congestion/rhinorrhea | n (%) | 21 (27) | 10 (18) | 1.63 | 0.257 |
| Gastrointestinal symptoms (nausea, vomiting, diarrhea) | n (%) | 30 (38) | 13 (24) | 1.98 | 0.080 |
Patients were rapidly diagnosed and treated after presentation to the hospital with no significant difference by fever status at presentation (P = 0.17 and 0.07 for differences in time from hospital presentation to diagnosis and treatment, respectively). While 82% (65/79) of patients with fever received empiric antibiotic treatment, only 59% (33/56) of those with no fever received empiric antibiotic treatment (P = 0.003). Prevalence of bacterial coinfection was similar between the 2 groups (5% in the fever group vs 9% in the group without fever; P = 0.067). Patients without fever were more frequently in an ICU than those with fever (P = 0.01), but we observed no significant differences in mortality by fever status at presentation in the small number of deaths during the study period (P = 0.61). Compared to patients with no fever, after adjustment for age (<40 vs 40) and Charlson index (0, 12, 3), patients with fever had significantly decreased likelihood of late hospital presentation (>2 days from the onset of illness) (adjusted odds ratio [OR]: 0.27, P = 0.001). Patients with fever also had decreased likelihood of ICU stay compared to patients with no fever, after adjustment for age and Charlson index (adjusted OR: 0.42, P = 0.05) (Table 2).
| Adjusted Odds Ratio | P Value | |
|---|---|---|
| ||
| Late hospital presentation (>2 d from onset of illness) | ||
| Fever (vs no fever) | 0.27 | 0.001 |
| Age (40 y) | 2.06 | 0.071 |
| Charlson index | ||
| 12 | 0.69 | 0.38 |
| 3 | 1.01 | 0.99 |
| ICU admission | ||
| Fever (vs no fever) | 0.42 | 0.05 |
| Age (40 y) | 1.21 | 0.701 |
| Charlson index | ||
| 12 | 0.66 | 0.448 |
| 3 | 4.02 | 0.016 |
Patients who presented to the hospital more than 2 days after the onset of illness were significantly more likely to be treated in an ICU during their hospitalization (P < 0.001). Among 71 patients who presented to the hospital more than 2 days after the onset of illness, 27 patients were treated in an ICU and 44 patients did not require ICU care. Among 64 patients who presented to the hospital within 2 days of the onset of illness, 4 patients were treated in an ICU and 60 patients did not require ICU care. Those who presented to the hospital more than 2 days after the onset of illness also had a nonsignificantly higher death rate: 7 of 71 (10%) versus 2 of 64 (3%). Among the comorbidities tested, patients with no fever were significantly more likely to have diabetes mellitus (P = 0.03).
ICU patients had a slightly higher median Charlson index than those who did not receive ICU care (median score 1 vs score 0 for ICU patients vs non‐ICU patients, respectively; P = 0.052). We observed no difference in median Charlson index between patients with late hospital presentation (>2 days from onset of illness) and those with early hospital presentation (2 days from onset of illness) (P = 0.51).
DISCUSSION
We present the first study, to our knowledge, to assess the influence of presence of fever, as part of the illness prior to hospital presentation, on time to presentation and treatment for H1N1/09 patients. By comparing patients who had fever with those who did not, we showed that patients with fever tended to seek medical care sooner and thus received treatment sooner. Those without fever may not have realized they needed to seek medical care until much later in the course of disease compared to those with fever. Given that ICU stay was significantly more likely in patients with late presentation and no fever, these findings have important medical and economic consequences for individual patients and healthcare systems. Our findings, in conjunction with previous studies indicating reduced disease severity with early treatment of influenza,1013, can be used to educate the public to seek medical care for influenza early, even in the absence of fever.
Our study confirms previous observations that a significant number of patients with H1N1/09 did not have fever.5, 6 Nevertheless, fever appeared to trigger initiation of empiric antibiotic treatment. Empiric antibiotic use was significantly higher in the fever group. There was no difference in the median Charlson index among those who received antibiotic treatment and those who did not, suggesting that comorbidities are unlikely to explain the observed association. Empiric antibiotic treatment appeared not to affect whether or not a patient required ICU care during the hospitalization, though this association is difficult to measure because the majority of ICU patients (84%) were admitted to an ICU on the same day as hospital admission. We saw no difference in laboratory‐confirmed bacterial coinfection between patients presenting with and without fever. We did not observe a significant effect of late presentation or fever on mortality, though the small number of deaths in the study likely limited our ability to detect any association.
Recent studies have shown that clinical signs cannot reliably differentiate H1N1/09‐positive and H1N1/09‐negative patients.18, 19 In our study, patients in the fever and non‐fever groups received prompt diagnosis and treatment once they presented to the hospital. This may be because clinicians were encouraged to test patients based on any suspicion for influenza infection and to treat for influenza while waiting for the result of testing. This may explain the similar timeframes for diagnosis and treatment between the fever and non‐fever groups.
Limitations
Our study took place in a single medical center, and our findings may not be generalizable to other clinical settings. However, this large tertiary care center admits 25% of patients in central Ohio. Our study focused on hospitalized patients only and excluded patients who may have presented to the emergency department with H1N1/09 but were not hospitalized. Thus, our findings can be applied to patients with severe enough disease to warrant hospitalization. It is possible that patients without fever presented early (within 2 days of onset of illness) and were treated as outpatients; these individuals would not have been captured in the current analysis. However, our primary goal was to assess the effect of fever on the overall course of H1N1/09 presentation, diagnosis, and management in hospitalized patients. Provider bias may have played a role in the decision to admit patients to the hospital, but clinicians were encouraged to use a standardized protocol to drive decisions to test and admit. We studied H1N1/09 patients only, and our findings may not be generalizable to patients infected with other influenza types. However, H1N1/09 influenza symptoms are similar to other types of influenza. We also did not collect data on insurance status which may play a role in patients' choice to seek hospital care.
Strengths
Our study also has substantial strengths. Because clinicians tested all patients with symptoms of respiratory illness and unexplained gastrointestinal symptoms irrespective of the presence of fever, we were able to assess the proportion of H1N1/09 patients without fever as part of their illness and to determine that the most common presenting symptoms were respiratory. Our analysis includes only patients with laboratory‐confirmed H1N1/09 infection. Our surveillance was strengthened by the availability of real‐time PCR confirmatory testing performed in our own molecular microbiology laboratory.
Conclusions
In this study, nearly half of patients with H1N1/09 requiring hospitalization did not have fever as part of their illness. Patients with fever tended to seek medical care sooner and thus received treatment sooner. Patients with late presentation and no fever were more likely to need ICU admission. Our findings reinforce previous studies that indicate better outcomes with early treatment of influenza.1013 These findings can be used to prompt clinicians to consider treating hospitalized patients with influenza and to educate the public to seek medical care soon after the onset of illness, despite absence of fever, if symptoms of respiratory illness and unexplained gastrointestinal symptoms are present during the influenza season.
The Centers for Disease Control and Prevention (CDC) established a case definition during the 2009 influenza season for influenza‐like illness (ILI): a temperature of 100.0F (37.8C), oral or equivalent, and cough or sore throat, in the absence of a known cause other than influenza.1 Even though this definition is currently being revised, fever is still listed as a predominant symptom for influenza on the CDC Web site.2, 3
During the spring of 2009 in New York City, 95% of patients with pandemic hemagglutinin type 1 and neuraminidase type 1 (H1N1) influenza A met the case definition for ILI,4 whereas a report from Mexico5 and another from China6 reported that about one‐third of patients presented without fever. A recent study found that the clinical features of H1N1/09 influenza and seasonal strains were similar in hospitalized patients.7 Until recently, the CDC case definition for ILI included fever as a requisite symptom. Most clinicians still consider influenza a fever‐based illness.8, 9 Some studies have reported that seeking early medical care and early treatment for influenza resulted in reduced severity and duration of illness.1013 However, individuals without fever may not have sought early care because their illnesses did not meet all the criteria for ILI.
We hypothesized that significant number of our patients hospitalized during the H1N1/09 influenza pandemic did not have fever as part of the influenza‐like illness prior to hospital presentation. We further hypothesized that this absence of fever was associated with delayed presentation to the hospital and delayed treatment. No prior studies have assessed the effect of fever on hospital presentation, diagnosis, and treatment of H1N1/09 patients. We performed a retrospective cohort analysis comparing H1N1/09 patients with fever as part of their illness prior to hospital presentation to those without fever, to assess the influence of fever on hospital presentation and on diagnosis and treatment in hospitalized patients with laboratory‐confirmed H1N1/09 infection.
METHODS
Study Design and Setting
This retrospective cohort study was conducted at a tertiary care center with 1159 beds divided between 2 general hospitals (634 and 167 beds), a large cancer center (208 beds), and an inpatient cardiac hospital (150 beds).
Selection of Participants
During the pandemic influenza season, clinicians throughout the tertiary care center were encouraged to obtain nasopharyngeal swabs to screen for influenza from all patients presenting to the hospital with respiratory illness, unexplained gastrointestinal symptoms, ILI, or any suspicion for influenza infection based on physician discretion. Patients were admitted through routine hospital admission procedures and triage. Physicians were encouraged to treat all patients in whom pandemic H1N1/09 was suspected empirically with an antiviral drug (oseltamivir). Patients with a positive H1N1/09 test were then continued on treatment. Obese patients, critically ill patients requiring intensive care unit (ICU) care, pregnant women, and patients with chronic medical conditions such as chronic respiratory disease, chronic kidney disease, diabetes mellitus, and immune‐compromising conditions were treated with a higher dose of oseltamivir (150 mg twice a day). Other patients were treated with the standard dose of oseltamivir (75 mg twice a day). The present analyses include all patients at least 18 years of age, hospitalized with laboratory‐confirmed pandemic H1N1/09 virus infection from 1 June 2009 through 31 December 2009. Subjects younger than 18 years and prisoners were excluded.
Data Collection
Laboratory‐confirmed pandemic H1N1/09 patients were identified from the hospital's infection control database. After identifying eligible patients, physician investigators accessed each electronic medical record to obtain health history, diagnosis, test results, disposition, and clinical outcome information. We captured data on gender, race, age, comorbidities, symptoms and onset of illness, hospital presentation, diagnosis, treatment, ICU admission, and death. Data abstraction was performed by an infectious disease physician using a standardized form, and all data abstraction was validated by another infectious disease physician. The research was deemed exempt from further review by the Ohio State University Institutional Review Board (2009E0979).
Subjects with laboratory‐confirmed H1N1/09 virus infection were identified if nasal swab or respiratory secretions were positive for novel influenza A (H1N1) by specific rapid antigen or culture testing. Nasal swabs that were processed for confirmatory influenza testing by polymerase chain reaction (PCR) were confirmed by panel (xTAG) or specific influenza A and B PCR (Luminex Molecular Diagnostics, Toronto, CA).14 Positive tests were confirmed using the Prodesse ProFlu‐ST, Influenza A [2009] real‐time PCR (H1N1 subtyping) assay (Focus Diagnostics, Cypress, CA).
Day of onset of illness was defined as the day when the patient started developing one or more symptoms of influenza‐like illness. Day of hospital presentation was defined as the day when the patient either presented to the emergency department or a physician's office and was subsequently admitted or was directly admitted to the hospital. Fever was defined as a temperature of 100F (37.8C) or greater at presentation, or reported subjective fever upon presentation, and reported antipyretic use (nonsteroidal anti‐inflammatory drugs or acetaminophen) prior to admission. When multiple temperature readings were present in the emergency department record, any recorded temperature of 100F or greater was considered a fever. The Charlson comorbidity index predicts the 10‐year mortality for a patient with a range of comorbid conditions such as heart disease, acquired immune deficiency syndrome (AIDS), or cancer. The Charlson comorbidity index was calculated according to previously described methods.1517 Diabetes, chronic respiratory disease (asthma, chronic obstructive pulmonary disease), and chronic kidney disease with hemodialysis were recorded as comorbidities if these diagnoses were documented in the patient notes. Immunosuppression was defined as oral steroid use or other immunosuppressive medication, organ transplantation, human immunodeficiency virus (HIV) infection, or cancer chemotherapy. Empiric antibiotic treatment was defined as treatment with antibiotics regardless of the evidence of bacterial infection. Proven bacterial coinfection was defined as infection with bacterial pathogens documented by a positive bacterial culture from any site that was obtained in response to clinical suspicion of bacterial infection.
Data Analysis
Demographic and clinical characteristics were summarized for all patients, and for those patients with or without fever, as part of their illness prior to hospital presentation. Continuous variables were summarized as mean ( standard deviation) or median values. For categorical variables, the percentage of patients in each category was calculated. For comparison between patients with fever and those with no fever, for continuous variables, the 2‐sample t test for normally distributed data or the Wilcoxon rank‐sum test for non‐normal data was used. The Pearson chi‐square test was used for categorical data. We performed a logistic regression analysis to examine the association between fever and time to presentation and ICU admission, adjusting for patient age and Charlson index. All analyses were carried out using SAS 9.2 software (SAS, Cary, NC).
RESULTS
We identified 135 hospitalized patients with laboratory‐confirmed pandemic H1N1 virus infection during the study period. A substantial number of patients (n = 56, 42%) had no fever as part of their illness prior to hospital presentation. Thirty‐one patients (23%) required treatment in an ICU during hospitalization, and 9 (7%) died. While we observed no differences between patients with fever and those without fever prior to hospital presentation in gender, race, or age (Table 1), patients without fever had higher Charlson index compared to those with fever (P = 0.01, Wilcoxon rank‐sum test). Patients without fever also had significantly longer time to presentation to the hospital since the onset of illness, compared to those with fever (median of 4 vs 2 days, P < 0.001). Patients without fever also had significantly longer time to initiation of proper treatment since the onset of illness, compared to those with fever (median of 5 vs 2 days, P = 0.001).
| Fever | No Fever | Unadjusted Odds Ratio | P Value* | ||
|---|---|---|---|---|---|
| N = 79 (59%) | N = 56 (42%) | ||||
| |||||
| Gender | Female, n (%) | 41 (52) | 34 (61) | 0.7 | 0.31 |
| Race | White, n (%) | 45 (57) | 29 (52) | 1.03 | 0.30 |
| Black, n (%) | 30 (38) | 20 (36) | |||
| Other, n (%) | 4 (5) | 7 (13) | |||
| Age | Mean SD (range) | 4315 (1889) | 48 17 (1989) | 0.060 | |
| Charlson index | Median (range) | 0 (0, 13) | 1 (0, 13) | 0.010 | |
| Comorbidities | |||||
| Obese (BMI 30) | n (%) | 37 (50) | 29 (54) | 0.82 | 0.571 |
| Chronic respiratory illness (asthma, COPD) | n (%) | 19 (24) | 13 (23) | 1.05 | 0.910 |
| Diabetes | n (%) | 14 (18) | 19 (34) | 0.419 | 0.031 |
| CKD with hemodialysis | n (%) | 12 (15) | 6 (11) | 1.49 | 0.451 |
| Immunocompromised | n (%) | 29 (38) | 13 (23) | 1.92 | 0.095 |
| Days from onset to hospital presentation | Median (range) | 2 (010) | 4 (121) | 0.0001 | |
| No. with days >2 | n (%) | 31 (39) | 40 (71) | 0.26 | <0.001 |
| Days to diagnosis from hospital presentation | Median (range) | 0 (03) | 0 (05) | 0.166 | |
| No. with days >2 | n (%) | 1 (1) | 3 (5) | 0.24 | 0.170 |
| Days to oseltamivir since admission | Median (range) | 0 (013) | 1 (014) | 0.07 | |
| Days to oseltamivir since onset of illness | Median (range) | 2 (013) | 5 (119) | 0.0003 | |
| No. with days >2 | n (%) | 35 (44) | 39 (70) | 0.24 | 0.001 |
| Need for ICU care | n (%) | 12 (15) | 19 (34) | 0.35 | 0.010 |
| Death | n (%) | 6 (8) | 3 (5) | 1.45 | 0.061 |
| Bacterial coinfection | n (%) | 5 (n = 79; 6%) | 9 (15) | 0.35 | 0.067 |
| Empiric antibiotics | 65 (n = 77; 84%) | 33 (59) | 3.24 | 0.003 | |
| Lower respiratory symptoms | n (%) | 72 (91) | 50 (91) | 1.03 | 0.963 |
| Cough | n (%) | 58 (73) | 38 (69) | 1.24 | 0.585 |
| Shortness of breath | n (%) | 34 (43) | 25 (46) | 0.91 | 0.782 |
| Respiratory failure | n (%) | 7 (9) | 7 (13) | 0.67 | 0.472 |
| Sore throat/congestion/rhinorrhea | n (%) | 21 (27) | 10 (18) | 1.63 | 0.257 |
| Gastrointestinal symptoms (nausea, vomiting, diarrhea) | n (%) | 30 (38) | 13 (24) | 1.98 | 0.080 |
Patients were rapidly diagnosed and treated after presentation to the hospital with no significant difference by fever status at presentation (P = 0.17 and 0.07 for differences in time from hospital presentation to diagnosis and treatment, respectively). While 82% (65/79) of patients with fever received empiric antibiotic treatment, only 59% (33/56) of those with no fever received empiric antibiotic treatment (P = 0.003). Prevalence of bacterial coinfection was similar between the 2 groups (5% in the fever group vs 9% in the group without fever; P = 0.067). Patients without fever were more frequently in an ICU than those with fever (P = 0.01), but we observed no significant differences in mortality by fever status at presentation in the small number of deaths during the study period (P = 0.61). Compared to patients with no fever, after adjustment for age (<40 vs 40) and Charlson index (0, 12, 3), patients with fever had significantly decreased likelihood of late hospital presentation (>2 days from the onset of illness) (adjusted odds ratio [OR]: 0.27, P = 0.001). Patients with fever also had decreased likelihood of ICU stay compared to patients with no fever, after adjustment for age and Charlson index (adjusted OR: 0.42, P = 0.05) (Table 2).
| Adjusted Odds Ratio | P Value | |
|---|---|---|
| ||
| Late hospital presentation (>2 d from onset of illness) | ||
| Fever (vs no fever) | 0.27 | 0.001 |
| Age (40 y) | 2.06 | 0.071 |
| Charlson index | ||
| 12 | 0.69 | 0.38 |
| 3 | 1.01 | 0.99 |
| ICU admission | ||
| Fever (vs no fever) | 0.42 | 0.05 |
| Age (40 y) | 1.21 | 0.701 |
| Charlson index | ||
| 12 | 0.66 | 0.448 |
| 3 | 4.02 | 0.016 |
Patients who presented to the hospital more than 2 days after the onset of illness were significantly more likely to be treated in an ICU during their hospitalization (P < 0.001). Among 71 patients who presented to the hospital more than 2 days after the onset of illness, 27 patients were treated in an ICU and 44 patients did not require ICU care. Among 64 patients who presented to the hospital within 2 days of the onset of illness, 4 patients were treated in an ICU and 60 patients did not require ICU care. Those who presented to the hospital more than 2 days after the onset of illness also had a nonsignificantly higher death rate: 7 of 71 (10%) versus 2 of 64 (3%). Among the comorbidities tested, patients with no fever were significantly more likely to have diabetes mellitus (P = 0.03).
ICU patients had a slightly higher median Charlson index than those who did not receive ICU care (median score 1 vs score 0 for ICU patients vs non‐ICU patients, respectively; P = 0.052). We observed no difference in median Charlson index between patients with late hospital presentation (>2 days from onset of illness) and those with early hospital presentation (2 days from onset of illness) (P = 0.51).
DISCUSSION
We present the first study, to our knowledge, to assess the influence of presence of fever, as part of the illness prior to hospital presentation, on time to presentation and treatment for H1N1/09 patients. By comparing patients who had fever with those who did not, we showed that patients with fever tended to seek medical care sooner and thus received treatment sooner. Those without fever may not have realized they needed to seek medical care until much later in the course of disease compared to those with fever. Given that ICU stay was significantly more likely in patients with late presentation and no fever, these findings have important medical and economic consequences for individual patients and healthcare systems. Our findings, in conjunction with previous studies indicating reduced disease severity with early treatment of influenza,1013, can be used to educate the public to seek medical care for influenza early, even in the absence of fever.
Our study confirms previous observations that a significant number of patients with H1N1/09 did not have fever.5, 6 Nevertheless, fever appeared to trigger initiation of empiric antibiotic treatment. Empiric antibiotic use was significantly higher in the fever group. There was no difference in the median Charlson index among those who received antibiotic treatment and those who did not, suggesting that comorbidities are unlikely to explain the observed association. Empiric antibiotic treatment appeared not to affect whether or not a patient required ICU care during the hospitalization, though this association is difficult to measure because the majority of ICU patients (84%) were admitted to an ICU on the same day as hospital admission. We saw no difference in laboratory‐confirmed bacterial coinfection between patients presenting with and without fever. We did not observe a significant effect of late presentation or fever on mortality, though the small number of deaths in the study likely limited our ability to detect any association.
Recent studies have shown that clinical signs cannot reliably differentiate H1N1/09‐positive and H1N1/09‐negative patients.18, 19 In our study, patients in the fever and non‐fever groups received prompt diagnosis and treatment once they presented to the hospital. This may be because clinicians were encouraged to test patients based on any suspicion for influenza infection and to treat for influenza while waiting for the result of testing. This may explain the similar timeframes for diagnosis and treatment between the fever and non‐fever groups.
Limitations
Our study took place in a single medical center, and our findings may not be generalizable to other clinical settings. However, this large tertiary care center admits 25% of patients in central Ohio. Our study focused on hospitalized patients only and excluded patients who may have presented to the emergency department with H1N1/09 but were not hospitalized. Thus, our findings can be applied to patients with severe enough disease to warrant hospitalization. It is possible that patients without fever presented early (within 2 days of onset of illness) and were treated as outpatients; these individuals would not have been captured in the current analysis. However, our primary goal was to assess the effect of fever on the overall course of H1N1/09 presentation, diagnosis, and management in hospitalized patients. Provider bias may have played a role in the decision to admit patients to the hospital, but clinicians were encouraged to use a standardized protocol to drive decisions to test and admit. We studied H1N1/09 patients only, and our findings may not be generalizable to patients infected with other influenza types. However, H1N1/09 influenza symptoms are similar to other types of influenza. We also did not collect data on insurance status which may play a role in patients' choice to seek hospital care.
Strengths
Our study also has substantial strengths. Because clinicians tested all patients with symptoms of respiratory illness and unexplained gastrointestinal symptoms irrespective of the presence of fever, we were able to assess the proportion of H1N1/09 patients without fever as part of their illness and to determine that the most common presenting symptoms were respiratory. Our analysis includes only patients with laboratory‐confirmed H1N1/09 infection. Our surveillance was strengthened by the availability of real‐time PCR confirmatory testing performed in our own molecular microbiology laboratory.
Conclusions
In this study, nearly half of patients with H1N1/09 requiring hospitalization did not have fever as part of their illness. Patients with fever tended to seek medical care sooner and thus received treatment sooner. Patients with late presentation and no fever were more likely to need ICU admission. Our findings reinforce previous studies that indicate better outcomes with early treatment of influenza.1013 These findings can be used to prompt clinicians to consider treating hospitalized patients with influenza and to educate the public to seek medical care soon after the onset of illness, despite absence of fever, if symptoms of respiratory illness and unexplained gastrointestinal symptoms are present during the influenza season.
- CDC. Update: infections with a swine‐origin influenza A (H1N1) virus—United States and other countries, April 28, 2009. MMWR.2009;58:433–434.
- United States Centers for Disease Control and Prevention. Clinical Signs and Symptoms of Influenza. Available at: http://www.cdc.gov/flu/professionals/acip/clinical.htm#signs. Accessed June 11, 2012.
- United States Centers for Disease Control and Prevention. Cold Versus Flu. Available at: http://www.cdc.gov/flu/about/qa/coldflu.htm. Accessed June 11, 2012.
- CDC. Swine‐origin influenza A (H1N1) virus infections in a school—New York City, April 30, 2009. MMWR. 2009;58(Dispatch);1–3.
- The New York Times. Many swine flu cases have no fever. Available at: http://www.nytimes.com/2009/05/13/health/13fever.html?_r=1361(26):2507–2517.
- , , , et al. Clinical and epidemiological profile of patients with severe H1N1/09 pandemic influenza in Australia and New Zealand: an observational cohort study. BMJ Open.2011;1(1). Available at: http://dx.doi.org/10.1136/bmjopen‐2011‐000100.
- Colds and the Flu. Available at: http://familydoctor.org/familydoctor/en/diseases‐conditions/colds‐and‐the‐flu/symptoms.html. Accessed June 11, 2012.
- Flu or Cold Symptoms? Available at: http://www.webmd.com/cold‐and‐flu/cold‐guide/flu‐cold‐symptoms. Accessed June 11, 2012.
- . Neuraminidase inhibitors for influenza. N Engl J Med. 2005;353:1363–1373.
- , , , et al. Early administration of oral oseltamivir increases the benefits of influenza treatment. J Antimicrob Chemother. 2003;51:123–129.
- , , , et al. Factors influencing the effectiveness of oseltamivir and amantadine for the treatment of influenza: a multicenter study from Japan of the 2002‐2003 influenza season. Clin Infect Dis. 2005;40:1309–1316.
- , , , et al. Use of oseltamivir during influenza outbreaks in Ontario nursing homes, 1999‐2000. J Am Geriatr Soc. 2002;50:608–616.
- , . Likelihood that an unsubtypeable influenza A virus result obtained with the Luminex xTAG respiratory virus panel is indicative of infection with novel A/H1N1 (swine‐like) influenza virus. J Clin Microbiol. 2009;47:2347–2348.
- , , , . A new method of classifying prognostic comorbidity in longitudinal studies: development and validation. J Chronic Dis. 1987;40(5):373–383.
- , , , . A systematic review of the Charlson comorbidity index using Canadian administrative databases: a perspective on risk adjustment in critical care research. J Crit Care. 2005;20(1):12–19.
- , , . Adapting a clinical comorbidity index for use with ICD‐9‐CM administrative databases. J Clin Epidemiol. 1992;45(6):613–619.
- , , , , , . Characteristics of outpatients with pandemic H1N1/09 influenza in a tertiary care university hospital in Korea. Yonsei Med J. 2012;53(1):213–220.
- , , , et al. Comparison of clinical presentation of respiratory tract infections in H1N1/09‐positive and H1N1/09‐negative patients. Eur J Pediatr. 2012;171(1):159–166.
- CDC. Update: infections with a swine‐origin influenza A (H1N1) virus—United States and other countries, April 28, 2009. MMWR.2009;58:433–434.
- United States Centers for Disease Control and Prevention. Clinical Signs and Symptoms of Influenza. Available at: http://www.cdc.gov/flu/professionals/acip/clinical.htm#signs. Accessed June 11, 2012.
- United States Centers for Disease Control and Prevention. Cold Versus Flu. Available at: http://www.cdc.gov/flu/about/qa/coldflu.htm. Accessed June 11, 2012.
- CDC. Swine‐origin influenza A (H1N1) virus infections in a school—New York City, April 30, 2009. MMWR. 2009;58(Dispatch);1–3.
- The New York Times. Many swine flu cases have no fever. Available at: http://www.nytimes.com/2009/05/13/health/13fever.html?_r=1361(26):2507–2517.
- , , , et al. Clinical and epidemiological profile of patients with severe H1N1/09 pandemic influenza in Australia and New Zealand: an observational cohort study. BMJ Open.2011;1(1). Available at: http://dx.doi.org/10.1136/bmjopen‐2011‐000100.
- Colds and the Flu. Available at: http://familydoctor.org/familydoctor/en/diseases‐conditions/colds‐and‐the‐flu/symptoms.html. Accessed June 11, 2012.
- Flu or Cold Symptoms? Available at: http://www.webmd.com/cold‐and‐flu/cold‐guide/flu‐cold‐symptoms. Accessed June 11, 2012.
- . Neuraminidase inhibitors for influenza. N Engl J Med. 2005;353:1363–1373.
- , , , et al. Early administration of oral oseltamivir increases the benefits of influenza treatment. J Antimicrob Chemother. 2003;51:123–129.
- , , , et al. Factors influencing the effectiveness of oseltamivir and amantadine for the treatment of influenza: a multicenter study from Japan of the 2002‐2003 influenza season. Clin Infect Dis. 2005;40:1309–1316.
- , , , et al. Use of oseltamivir during influenza outbreaks in Ontario nursing homes, 1999‐2000. J Am Geriatr Soc. 2002;50:608–616.
- , . Likelihood that an unsubtypeable influenza A virus result obtained with the Luminex xTAG respiratory virus panel is indicative of infection with novel A/H1N1 (swine‐like) influenza virus. J Clin Microbiol. 2009;47:2347–2348.
- , , , . A new method of classifying prognostic comorbidity in longitudinal studies: development and validation. J Chronic Dis. 1987;40(5):373–383.
- , , , . A systematic review of the Charlson comorbidity index using Canadian administrative databases: a perspective on risk adjustment in critical care research. J Crit Care. 2005;20(1):12–19.
- , , . Adapting a clinical comorbidity index for use with ICD‐9‐CM administrative databases. J Clin Epidemiol. 1992;45(6):613–619.
- , , , , , . Characteristics of outpatients with pandemic H1N1/09 influenza in a tertiary care university hospital in Korea. Yonsei Med J. 2012;53(1):213–220.
- , , , et al. Comparison of clinical presentation of respiratory tract infections in H1N1/09‐positive and H1N1/09‐negative patients. Eur J Pediatr. 2012;171(1):159–166.
Copyright © 2012 Society of Hospital Medicine
Mortality After Therapeutic Hypothermia
There are over 350,000 cases of out‐of‐hospital cardiac arrest (OHCA) each year in the United States1, 2 and, with supportive therapy alone, only a fraction of victims survive to hospital discharge. Rapid intervention including cardiopulmonary resuscitation in the moments following arrest is critical to minimizing neurologic injury, morbidity, and mortality. In 2002, two small randomized controlled trials showed a survival benefit of therapeutic hypothermia (TH) when provided within 12 hours after return of circulation following an OHCA and, to date, TH remains one of the few interventions with proven mortality benefit after initial cardiopulmonary resuscitation.3, 4 Since 2003, TH has been incorporated into the American Heart Association practice guidelines57 and use of TH has steadily increased, but widespread clinical uptake remains low.8, 9
The initial studies that evaluated TH were small, with only 189 patients included in the TH arms of the 2 trials combined. To date, only a few studies have replicated this initial observation in real‐world settings, with little analysis of outcomes in US centers in particular.1013 Accordingly, we aimed to examine the real‐world experience with TH in the United States using a large administrative claims database of all California hospital admissions to describe utilization trends, hospital mortality, and volumeoutcome relationships associated with the intervention.
MATERIALS AND METHODS
Data
We identified all admissions to California hospitals during 19992008 based on discharge records from the California Office of Statewide Health Planning and Development. Our study period included cases of TH performed prior to the 2002 major clinical trials, since TH was in occasional use prior to the publication of these trials. The data was de‐identified and publicly available, and therefore exempt from review by the Institutional Review Board. In addition to hospital name, each discharge record included patient age, gender, admission year, International Classification of Disease, Ninth Revision (ICD‐9) code for presenting primary and secondary diagnoses, procedure codes, and disposition (discharge to home or rehabilitation, in‐hospital death). All California hospitals were included in the registry (n = 419). We defined teaching status for each hospital based on membership in the Council of Teaching Hospitals, as reported in the American Hospital Association's Annual Survey (n = 19 teaching hospitals).14
Setting and Participants
We used discharge diagnoses to identify patients who could be considered eligible for therapeutic hypothermia after cardiac arrest. We classified patients as eligible for therapeutic hypothermia after cardiac arrest based on ICD‐9 diagnosis codes that indicated the presence of both cardiac arrest and anoxic brain injury in the administrative diagnoses. Because of known imprecision in using billing codes to identify patients with cardiac arrest,15, 16 we broadly defined cardiac arrest to include those patients with ICD‐9 codes for cardiac arrest, ventricular fibrillation (VF), or ventricular tachycardia (VT) (see Supporting Table 1 in the online version of this article). We could not distinguish between out‐of‐hospital and in‐hospital cardiac arrest based on administrative diagnoses. To ensure that we included only patients with cardiac arrest complicated by neurologic insult, we required an ICD‐9 diagnosis of either anoxic brain injury, coma, or persistent vegetative state. Claims did not allow us to distinguish among initial cardiac arrest rhythms (VF vs pulseless VT vs asystole). Patients younger than 18 years of age, and those who were pregnant, suffered traumatic brain injury, intracranial hemorrhage, metastatic cancer, or dementia were excluded.3, 17 We did not exclude patients based on coagulopathy (which is considered a contraindication to TH), since ICD‐9 coding did not allow us to determine the severity of the coagulopathy or whether it was a result of therapeutic hypothermia itself.
We used the ICD‐9 procedure code (99.81) for TH to first identify patients who underwent TH from 1999 to 2008. Since this code also applies to TH used during cardiac and neurosurgery, we examined each of these cases and excluded individuals who underwent cardiac surgery or neurosurgery during the hospitalization. As in our eligible for TH definition, we excluded patients younger than 18 years of age, and those who were pregnant, suffered traumatic brain injury, intracranial hemorrhage, metastatic cancer, or dementia. Patients who underwent therapeutic hypothermia but for whom a specific procedure code was not recorded in the discharge abstractperhaps because the medical institution did not directly bill for the procedurecould not be identified.
Statistical Analysis
We used a multivariable logistic model to estimate differences in hospital mortality after cardiac arrest associated with use of therapeutic hypothermia. We conducted 2 specifications. In our baseline specification, we accounted for case‐mix differences between those who underwent TH and those who did not by adjusting for age, gender, year of admission, the number of Charlson‐Deyo comorbidities,1820 and hospital teaching status. Because mortality after cardiac arrest in centers that perform therapeutic hypothermia may be different from centers that do not, even for patients who do not undergo the procedure, we included indicators for volume tercile of therapeutic hypothermia cases performed. Volume of therapeutic hypothermia was defined at the hospital level as the total number of cases performed by that hospital from 1999 to 2008.
In order to explore how hospital teaching status, volume of therapeutic hypothermia procedures (broken into terciles), and year of admission affected the association between hospital mortality after cardiac arrest and therapeutic hypothermia, our baseline logistic model was expanded to include interactions between therapeutic hypothermia and each of these variables. The interaction between therapeutic hypothermia and year explored whether the effectiveness of the procedure changed over time, as case‐selection, method of therapeutic hypothermia (cold saline vs commercially available devices), and experience changed in California hospitals. For both specifications, we reported the odds ratio of hospital mortality among patients undergoing therapeutic hypothermia, as well as risk‐adjusted mortality for both TH and non‐TH groups.
STATA version 11 (STATA Corp, College Station, TX) was used for statistical analyses, and a 2‐sided P 0.05 was used.
RESULTS
Descriptive Data
Table 1 reports summary statistics for patients with cardiac arrest complicated by neurologic insult (anoxic brain injury, coma, or persistent vegetative state) between 1999 and 2008. Across all years, 204 patients were identified as undergoing TH. In comparison, 105 patients were identified as undergoing TH in 2008 alone. Patients who underwent TH were less likely to be male (30.7% vs 44.6% male, P < 0.01), were younger (63.9 15.0 years vs 67.3 15.7 years, P = 0.03), and had equivalent numbers of Charlson‐Deyo comorbidities (2.5 2.0 diagnoses vs 2.5 2.0 diagnoses, P = 0.89). Therapeutic hypothermia was more commonly employed at teaching hospitals (51/3709 [1.4%] vs 153/42,942 [0.4%], P < 0.01). There was a trend toward decreased unadjusted mortality among patients who underwent therapeutic hypothermia compared with those who did not (56.9% vs 62.8%, P = 0.08).
| Therapeutic Hypothermia | No Therapeutic Hypothermia | P Value | |
|---|---|---|---|
| |||
| No. observations | 204 | 46,629 | |
| No. cases in teaching hospitals | 51 | 3,658 | |
| No. cases in non‐teaching hospitals | 153 | 42,789 | |
| Age, y | 63.9 15.0 | 67.3 15.7 | 0.06 |
| Male | 30.7 | 44.6 | <0.01 |
| Hospital mortality, % | 56.9 | 62.8 | 0.08 |
| Comorbidities | |||
| No. Charlson‐Deyo comorbidities | 2.5 2.0 | 2.5 2.0 | 0.89 |
| Coronary artery disease, % | 48.0 | 38.0 | <0.01 |
| Acute myocardial infarction, % | 42.6 | 28.9 | <0.01 |
| Congestive heart failure, % | 27.9 | 35.3 | 0.03 |
| Hypertension, % | 36.3 | 33.2 | 0.83 |
| Acute renal failure, % | 33.3 | 26.6 | 0.03 |
| Diabetes mellitus, % | 30.9 | 23.0 | <0.01 |
| Chronic obstructive pulmonary disease, % | 10.3 | 19.3 | <0.01 |
Figures 1 and 2 provide additional aggregate statistics on therapeutic hypothermia in California hospitals. Figure 1 plots the number of therapeutic hypothermia cases recorded in the administrative discharge registry between 1999 and 2008. Of the 204 total cases identified during this period, 178 (87.3%) were performed between 2006 and 2008. Figure 2 shows the distribution of TH cases across centers that performed therapeutic hypothermia (n = 47 hospitals, 11.3 % of all hospitals). Ten centers accounted for 124/204 (60.7%) of the total patients treated with TH after cardiac arrest; the top 3 centers accounted for 64 (31.4%) of the treated patients. Twenty‐seven hospitals were identified as performing therapeutic hypothermia on only 1 or 2 patients between 1999 and 2008.
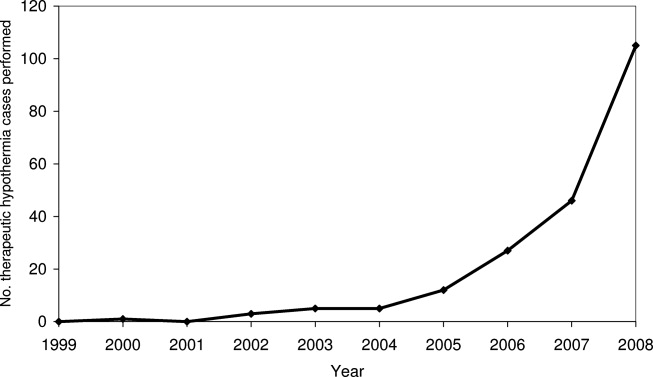
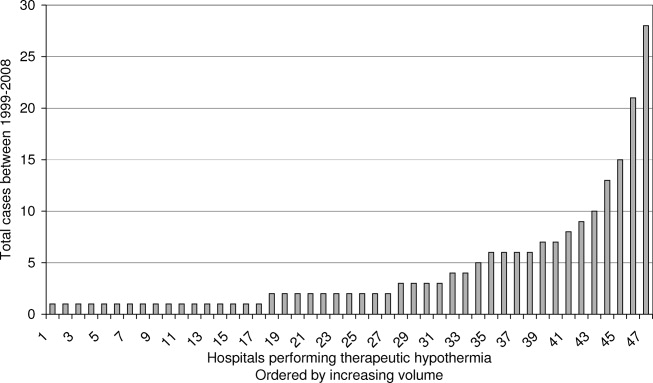
Risk‐Adjusted Mortality
Table 2 presents the odds ratio of factors predicting in‐hospital mortality after cardiac arrest complicated by neurologic insult. Factors include use of TH after cardiac arrest, age, gender, year of admission, number of Charlson‐Deyo comorbidities, hospital teaching status, and volume tercile of hospitals that performed therapeutic hypothermia. Overall, patients who were older, male, and had greater comorbidities were statistically more likely to die after cardiac arrest complicated by neurologic insult. Regardless of whether they underwent TH, patients admitted to hospitals in the highest volume tercile of TH use were more likely to die after cardiac arrest. Adjusting for volume tercile, teaching hospital status was not independently associated with mortality after cardiac arrest. The adjusted odds ratio of mortality among patients undergoing therapeutic hypothermia was 0.80 (95% confidence interval [CI] 0.601.06, P = 0.11). The adjusted probability of inpatient mortality among patients undergoing therapeutic hypothermia was 57.5% (95% CI 50.764.3%) compared to those who did not 62.8% (95% CI 61.763.9%, P = 0.11).
| Variable | Odds Ratio of Hospital Mortality (95% CI) | P Value |
|---|---|---|
| ||
| No. observations | 46,651 | |
| Age* | ||
| 6569 | 1.19 (1.121.28) | <0.001 |
| 7074 | 1.29 (1.201.39) | <0.001 |
| 7579 | 1.55 (1.441.67) | <0.001 |
| 8084 | 1.79 (1.651.93) | <0.001 |
| 85 and over | 2.06 (1.892.25) | <0.001 |
| Male | 1.15 (1.101.21) | <0.001 |
| Teaching hospital | 1.13 (0.951.34) | 0.17 |
| No. Charlson‐Deyo comorbidities | 1.09 (1.081.10) | <0.001 |
| Year trend | 0.98 (0.970.99) | <0.001 |
| Volume tercile among hospitals performing TH | ||
| First tercile | 0.94 (0.791.12) | 0.48 |
| Second tercile | 1.03 (0.801.33) | 0.82 |
| Third tercile | 1.20 (1.051.36) | 0.006 |
| Therapeutic hypothermia | 0.80 (0.601.06) | 0.11 |
Figure 3 presents adjusted mortality after cardiac arrest in hospitals that did not perform TH, as well as adjusted mortality associated with TH for each volume tercile of hospitals that performed the procedure. Hospital mortality rates among patients not receiving TH after cardiac arrest were slightly higher in hospitals in the high volume tercile of TH (66.3%, 95% CI 63.868.8%) compared to hospitals in low and moderate volume terciles and to hospitals not performing TH (P < 0.001). Hospital mortality rates among low and moderate TH volume centers and in centers not performing TH were similar (62.3%, 61.3%, and 63.4%, respectively). Among both the low volume and moderate volume terciles, however, patients who underwent TH after cardiac arrest were significantly less likely to die in‐hospital compared to those who did not. For patients admitted to hospitals in the low volume tercile, those undergoing therapeutic hypothermia had an adjusted hospital mortality rate of 25.5% (95% CI 3.047.9%) compared to those who did not undergo TH (adjusted mortality 61.3%, 95% CI 57.465.1%), P < 0.001. In the moderate volume tercile, patients receiving therapeutic hypothermia had an adjusted hospital mortality rate of 31.0% (95% CI 9.2%52.8%) compared to 63.4% (95% CI 57.769.1%), P < 0.001, among those not undergoing the procedure. There was no statistically significant difference in adjusted mortality between those who underwent TH and those who did not, in hospitals in the highest volume tercile (P = 0.211). In addition to examining how volume of therapeutic hypothermia performed by hospitals affected the association between TH and hospital mortality, we also examined whether year of admission and teaching hospital independently modified the association. Neither year of admission nor teaching hospital statistically significantly affected the association between therapeutic hypothermia and hospital mortality after cardiac arrest at the P < 0.10 level.
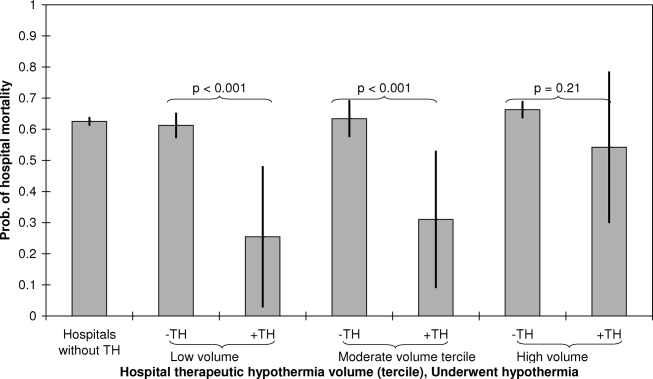
DISCUSSION
In an administrative database of all admissions to California hospitals, we demonstrated that use of TH increased steadily since the publication of the initial clinical trials in 2002. The absolute level of TH utilization in our study undoubtedly represents a significant underestimation of actual TH utilization, however, our study does provide an assessment of the utilization trends over time. The bulk of TH use appears to be performed in a small group of high volume centers, and 89% of California hospitals did not perform TH during the study period (as assessed by procedure billing codes). Additionally, within the limitations of a retrospective, administrative claims‐based study design, TH appears to be associated with a similar in‐hospital mortality rate to that seen in clinical trials.3, 4 In exploratory analyses, there appears to be a particular benefit of TH in low and moderate volume centers, though these findings should be considered hypothesis‐generating.
Despite the body of evidence in favor of TH, utilization in our study and others appears quite low. In a 2005 survey of physicians, 87% of respondents had never used therapeutic hypothermia, citing inadequate data, technical limitations, and lack of incorporation in the Advanced Cardiac Life Support (ACLS) protocol as principal justifications.8 Other surveys have shown similar results and noted that critical care physicians and those working in large medical centers were more likely to adopt the therapy.9 Advocates of the therapy have suggested that an explicit hospital‐based plan developed by key stakeholders can help facilitate implementation.21 Accordingly, there is growing interest in developing centers of expertise in highly intensive therapies such as TH. For instance, the New York City Emergency Medical Service has begun to explore a protocol to divert TH candidates to specialized centers.22, 23 Some favorable results have been reported in individual hospitals and local hospital systems.2428
Our data suggest that TH is associated with an in‐hospital mortality rate that is comparable to that reported in the clinical trials. For example, in a 2009 meta‐analysis of 4 clinical trials and 1 abstract (481 patients in total), TH was associated with a 35% relative mortality benefit as compared to standard post‐resuscitation care.29 It has been estimated that broad TH implementation could save thousands of lives30 and many authors have advocated for its use and outlined explicit protocols for implementation.17 Furthermore, TH appears to be cost‐effective in line with other accepted therapies. Assuming the Hypothermia After Cardiac Arrest (HACA) trial inclusion criteria, even at extreme estimates for costs, the cost‐effectiveness of hypothermia remains less than $100,000 per quality‐adjusted life year.31
There are important limitations of this study. Our use of administrative claims data certainly underestimates the level of TH utilization, since we could only identify cases in which TH was included in the billing codes for the hospitalization. Hospitals may vary in utilization of this particular billing code for TH in ways that bias our estimated associations. The ICD‐9 code 99.81 for therapeutic hypothermia was also not developed for post‐cardiac arrest TH specifically, so use of the code may actually lag clinical utilization. Although the observed trend in TH utilization is likely mainly due to a true increase in utilization, it is possible that some of the observed increase is due to an increase in utilization of TH procedure billing codes. Our TH utilization estimates should be construed as a lower bound of the actual rates. Additionally, although the estimated real‐world mortality benefit of TH may be comparable to that of clinical trials, the equivalence of patients in our sample to those in published randomized trials is uncertain. Similarly, even after adjusting for age, gender, year of admission, comorbidities, hospital teaching status, and TH volume, there are likely many unmeasured variables that influence mortality in both the TH and comparison groups. There are also likely patients included in our comparison group who had both cardiac arrest or ventricular tachycardia and anoxic brain injury, but who were not candidates for TH as the episode of cardiac arrest followed rather than preceded the anoxic brain injury. Since we lack detailed clinical data about the TH cases (initial rhythm, time before return of circulation, preexisting disease states, etc.), we are unable to match controls directly to cases. Additionally, we lack data to assess neurologic recovery or quality of life after arrest.
The observation that a mortality benefit in our study could be detected only in low and moderate volume centers requires further exploration. Indeed, one might expect that high volume centers may have better outcomes with TH as a result of more robust infrastructure, technical experience, and available resources. Our finding that mortality benefits of TH appear concentrated in centers with low to moderate volume of TH utilization suggest at least 1 of 2 possibilities. First, low and moderate volume centers may perform TH in a subset of patients who benefit most from the intervention or, alternatively, in the most viable cardiac arrest cases (those who may fare well with or without the therapy). Consequently, we may observe relatively favorable outcomes in this group due to this selection bias. Second, high volume centersdespite having more expertisemay also attract patients at higher mortality risk due to referral bias. This would lead us to estimate lower mortality benefits associated with TH in these high volume centers. Indeed, greater observed mortality at high volume centers regardless of TH status suggests that overall acuity is higher at high volume centers. While our inferences are greatly affected by issues of case selection and referral bias, it also important to consider the possibility that the estimated mortality benefit of TH in higher volume centers is lower because of the selection of patients who do not meet current guidelines for treatment with TH. Distinguishing whether the selection of patients undergoing TH at high volume centers is appropriate or inappropriate based on current guidelines is an important issue that merits further research with datasets with more refined patient clinical information.
In summary, therapeutic hypothermia utilization is low, but the rate of implementation has increased since the publication of the initial clinical trials in 2002. The bulk of TH utilization appears limited to a subset of high volume centers, and most centers in California appear to have not used the therapy. Real‐world in‐hospital mortality associated with TH is comparable to that reported in randomized clinical trials.
Acknowledgements
Disclosures: Dr Romley received support from NIH grant R03AG031990‐A1. Dr Noseworthy received support from the Max Schaldach Fellowship in Cardiac Pacing and Electrophysiology granted by the Heart Rhythm Society. The design, conduct, analysis, interpretation, and presentation of the data are the responsibility of the investigators, with no involvement from the funding sources. The contents of this article have not been published in any other peer‐reviewed media, and the manuscript is not under review elsewhere. All authors listed have contributed sufficiently to this project to be included as authors. The authors have no conflict of interest, financial or otherwise.
- , , , et al. Sudden cardiac death in the United States, 1989 to 1998. Circulation. 2001;104(18):2158–2163.
- , . Projections of global mortality and burden of disease from 2002 to 2030. PLoS Med. 2006;3(11):e442.
- , , , et al. Treatment of comatose survivors of out‐of‐hospital cardiac arrest with induced hypothermia. N Engl J Med. 2002;346(8):557–563.
- Hypothermia After Cardiac Arrest Study Group. Mild therapeutic hypothermia to improve the neurologic outcome after cardiac arrest. N Engl J Med. 2002;346(8):549–556.
- , , , et al. Therapeutic hypothermia after cardiac arrest: an advisory statement by the Advanced Life Support Task Force of the International Liaison Committee on Resuscitation. Circulation. 2003;108(1):118–121.
- 2005 American Heart Association guidelines for cardiopulmonary resuscitation and emergency cardiovascular care: part 7.5: postresuscitation support. Circulation. 2005;112(suppl I):IV‐84–IV‐88.
- , , , et al. Part 8: advanced life support: 2010 international consensus on cardiopulmonary resuscitation and emergency cardiovascular care science with treatment recommendations. Resuscitation. 2010;81(suppl 1):e93–e174.
- , , , et al. Induced hypothermia is underused after resuscitation from cardiac arrest: a current practice survey. Resuscitation. 2005;64(2):181–186.
- , , , et al. Therapeutic hypothermia utilization among physicians after resuscitation from cardiac arrest. Crit Care Med. 2006;34(7):1935–1940.
- , , , et al. Adverse events and their relation to mortality in out‐of‐hospital cardiac arrest patients treated with therapeutic hypothermia. Crit Care Med.39(1):57–64.
- , , , et al. Outcome, timing and adverse events in therapeutic hypothermia after out‐of‐hospital cardiac arrest. Acta Anaesthesiol Scand. 2009;53(7):926–934.
- . Clinical application of mild therapeutic hypothermia after cardiac arrest. Crit Care Med. 2007;35(4):1041–1047.
- , , , et al. Therapeutic hypothermia after out‐of‐hospital cardiac arrest: evaluation of a regional system to increase access to cooling. Circulation. 2011;124(2):206–214.
- American Hospital Association (AHA). American Hospital Association 2001 Annual Survey. Chicago, IL: Health Forum, LLC.
- , , . A multicenter study of the coding accuracy of hospital discharge administrative data for patients admitted to cardiac care units in Ontario. Am Heart J. 2002;144(2):290–296.
- . Accuracy of ICD‐9‐CM coding for the identification of patients with acute ischemic stroke: effect of modifier codes. Stroke. 1998;29(8):1602–1604.
- . Targeted temperature management for comatose survivors of cardiac arrest. N Engl J Med. 2010;363(13):1256–1264.
- , , , et al. A new method of classifying prognostic comorbidity in longitudinal studies: development and validation. J Chronic Dis. 1987;40(5):373–383.
- , , . Adapting a clinical comorbidity index for use with ICD‐9‐CM administrative databases. J Clin Epidemiol. 1992;45(6):613–619.
- , , . Adapting a clinical comorbidity index for use with ICD‐9‐CM administrative data: differing perspectives. J Clin Epidemiol. 1993;46(10):1075–1079; discussion 1081–1090.
- , , , et al. Practical implementation of therapeutic hypothermia after cardiac arrest. Hosp Pract (Minneap). 2009;37(1):71–83.
- . Paramedics will employ new therapy in cardiac arrest cases. New York Times. August 3, 2010:A18.
- , , , et al. Resuscitation center designation: recommendations for emergency medical services practices. Prehosp Emerg Care. 2010;14(1):51–61.
- , , , et al. Implementation of a hospital‐wide protocol for induced hypothermia following successfully resuscitated cardiac arrest. Crit Pathw Cardiol. 2010;9(4):216–220.
- , , , et al. Therapeutic hypothermia protocol in a community emergency department. West J Emerg Med. 2010;11(4):367–372.
- , , , et al. Implementation of a post‐cardiac arrest care bundle including therapeutic hypothermia and hemodynamic optimization in comatose patients with return of spontaneous circulation after out‐of‐hospital cardiac arrest: a feasibility study. Shock. 2011;35(4):360–366.
- , , , et al. Improved out‐of‐hospital cardiac arrest survival after the sequential implementation of 2005 AHA guidelines for compressions, ventilations, and induced hypothermia: the Wake County experience. Ann Emerg Med. 2010;56(4):348–357.
- , , , et al. Community‐based application of mild therapeutic hypothermia for survivors of cardiac arrest. South Med J. 2010;103(4):295–300.
- , , , et al. Hypothermia for neuroprotection in adults after cardiopulmonary resuscitation. Cochrane Database Syst Rev 2009(4):CD004128.
- , . Therapeutic hypothermia after cardiac arrest and myocardial infarction. Best Pract Res Clin Anaesthesiol. 2008;22(4):711–728.
- , , , et al. Cost‐effectiveness of therapeutic hypothermia after cardiac arrest. Circ Cardiovasc Qual Outcomes. 2009;2(5):421–428.
There are over 350,000 cases of out‐of‐hospital cardiac arrest (OHCA) each year in the United States1, 2 and, with supportive therapy alone, only a fraction of victims survive to hospital discharge. Rapid intervention including cardiopulmonary resuscitation in the moments following arrest is critical to minimizing neurologic injury, morbidity, and mortality. In 2002, two small randomized controlled trials showed a survival benefit of therapeutic hypothermia (TH) when provided within 12 hours after return of circulation following an OHCA and, to date, TH remains one of the few interventions with proven mortality benefit after initial cardiopulmonary resuscitation.3, 4 Since 2003, TH has been incorporated into the American Heart Association practice guidelines57 and use of TH has steadily increased, but widespread clinical uptake remains low.8, 9
The initial studies that evaluated TH were small, with only 189 patients included in the TH arms of the 2 trials combined. To date, only a few studies have replicated this initial observation in real‐world settings, with little analysis of outcomes in US centers in particular.1013 Accordingly, we aimed to examine the real‐world experience with TH in the United States using a large administrative claims database of all California hospital admissions to describe utilization trends, hospital mortality, and volumeoutcome relationships associated with the intervention.
MATERIALS AND METHODS
Data
We identified all admissions to California hospitals during 19992008 based on discharge records from the California Office of Statewide Health Planning and Development. Our study period included cases of TH performed prior to the 2002 major clinical trials, since TH was in occasional use prior to the publication of these trials. The data was de‐identified and publicly available, and therefore exempt from review by the Institutional Review Board. In addition to hospital name, each discharge record included patient age, gender, admission year, International Classification of Disease, Ninth Revision (ICD‐9) code for presenting primary and secondary diagnoses, procedure codes, and disposition (discharge to home or rehabilitation, in‐hospital death). All California hospitals were included in the registry (n = 419). We defined teaching status for each hospital based on membership in the Council of Teaching Hospitals, as reported in the American Hospital Association's Annual Survey (n = 19 teaching hospitals).14
Setting and Participants
We used discharge diagnoses to identify patients who could be considered eligible for therapeutic hypothermia after cardiac arrest. We classified patients as eligible for therapeutic hypothermia after cardiac arrest based on ICD‐9 diagnosis codes that indicated the presence of both cardiac arrest and anoxic brain injury in the administrative diagnoses. Because of known imprecision in using billing codes to identify patients with cardiac arrest,15, 16 we broadly defined cardiac arrest to include those patients with ICD‐9 codes for cardiac arrest, ventricular fibrillation (VF), or ventricular tachycardia (VT) (see Supporting Table 1 in the online version of this article). We could not distinguish between out‐of‐hospital and in‐hospital cardiac arrest based on administrative diagnoses. To ensure that we included only patients with cardiac arrest complicated by neurologic insult, we required an ICD‐9 diagnosis of either anoxic brain injury, coma, or persistent vegetative state. Claims did not allow us to distinguish among initial cardiac arrest rhythms (VF vs pulseless VT vs asystole). Patients younger than 18 years of age, and those who were pregnant, suffered traumatic brain injury, intracranial hemorrhage, metastatic cancer, or dementia were excluded.3, 17 We did not exclude patients based on coagulopathy (which is considered a contraindication to TH), since ICD‐9 coding did not allow us to determine the severity of the coagulopathy or whether it was a result of therapeutic hypothermia itself.
We used the ICD‐9 procedure code (99.81) for TH to first identify patients who underwent TH from 1999 to 2008. Since this code also applies to TH used during cardiac and neurosurgery, we examined each of these cases and excluded individuals who underwent cardiac surgery or neurosurgery during the hospitalization. As in our eligible for TH definition, we excluded patients younger than 18 years of age, and those who were pregnant, suffered traumatic brain injury, intracranial hemorrhage, metastatic cancer, or dementia. Patients who underwent therapeutic hypothermia but for whom a specific procedure code was not recorded in the discharge abstractperhaps because the medical institution did not directly bill for the procedurecould not be identified.
Statistical Analysis
We used a multivariable logistic model to estimate differences in hospital mortality after cardiac arrest associated with use of therapeutic hypothermia. We conducted 2 specifications. In our baseline specification, we accounted for case‐mix differences between those who underwent TH and those who did not by adjusting for age, gender, year of admission, the number of Charlson‐Deyo comorbidities,1820 and hospital teaching status. Because mortality after cardiac arrest in centers that perform therapeutic hypothermia may be different from centers that do not, even for patients who do not undergo the procedure, we included indicators for volume tercile of therapeutic hypothermia cases performed. Volume of therapeutic hypothermia was defined at the hospital level as the total number of cases performed by that hospital from 1999 to 2008.
In order to explore how hospital teaching status, volume of therapeutic hypothermia procedures (broken into terciles), and year of admission affected the association between hospital mortality after cardiac arrest and therapeutic hypothermia, our baseline logistic model was expanded to include interactions between therapeutic hypothermia and each of these variables. The interaction between therapeutic hypothermia and year explored whether the effectiveness of the procedure changed over time, as case‐selection, method of therapeutic hypothermia (cold saline vs commercially available devices), and experience changed in California hospitals. For both specifications, we reported the odds ratio of hospital mortality among patients undergoing therapeutic hypothermia, as well as risk‐adjusted mortality for both TH and non‐TH groups.
STATA version 11 (STATA Corp, College Station, TX) was used for statistical analyses, and a 2‐sided P 0.05 was used.
RESULTS
Descriptive Data
Table 1 reports summary statistics for patients with cardiac arrest complicated by neurologic insult (anoxic brain injury, coma, or persistent vegetative state) between 1999 and 2008. Across all years, 204 patients were identified as undergoing TH. In comparison, 105 patients were identified as undergoing TH in 2008 alone. Patients who underwent TH were less likely to be male (30.7% vs 44.6% male, P < 0.01), were younger (63.9 15.0 years vs 67.3 15.7 years, P = 0.03), and had equivalent numbers of Charlson‐Deyo comorbidities (2.5 2.0 diagnoses vs 2.5 2.0 diagnoses, P = 0.89). Therapeutic hypothermia was more commonly employed at teaching hospitals (51/3709 [1.4%] vs 153/42,942 [0.4%], P < 0.01). There was a trend toward decreased unadjusted mortality among patients who underwent therapeutic hypothermia compared with those who did not (56.9% vs 62.8%, P = 0.08).
| Therapeutic Hypothermia | No Therapeutic Hypothermia | P Value | |
|---|---|---|---|
| |||
| No. observations | 204 | 46,629 | |
| No. cases in teaching hospitals | 51 | 3,658 | |
| No. cases in non‐teaching hospitals | 153 | 42,789 | |
| Age, y | 63.9 15.0 | 67.3 15.7 | 0.06 |
| Male | 30.7 | 44.6 | <0.01 |
| Hospital mortality, % | 56.9 | 62.8 | 0.08 |
| Comorbidities | |||
| No. Charlson‐Deyo comorbidities | 2.5 2.0 | 2.5 2.0 | 0.89 |
| Coronary artery disease, % | 48.0 | 38.0 | <0.01 |
| Acute myocardial infarction, % | 42.6 | 28.9 | <0.01 |
| Congestive heart failure, % | 27.9 | 35.3 | 0.03 |
| Hypertension, % | 36.3 | 33.2 | 0.83 |
| Acute renal failure, % | 33.3 | 26.6 | 0.03 |
| Diabetes mellitus, % | 30.9 | 23.0 | <0.01 |
| Chronic obstructive pulmonary disease, % | 10.3 | 19.3 | <0.01 |
Figures 1 and 2 provide additional aggregate statistics on therapeutic hypothermia in California hospitals. Figure 1 plots the number of therapeutic hypothermia cases recorded in the administrative discharge registry between 1999 and 2008. Of the 204 total cases identified during this period, 178 (87.3%) were performed between 2006 and 2008. Figure 2 shows the distribution of TH cases across centers that performed therapeutic hypothermia (n = 47 hospitals, 11.3 % of all hospitals). Ten centers accounted for 124/204 (60.7%) of the total patients treated with TH after cardiac arrest; the top 3 centers accounted for 64 (31.4%) of the treated patients. Twenty‐seven hospitals were identified as performing therapeutic hypothermia on only 1 or 2 patients between 1999 and 2008.


Risk‐Adjusted Mortality
Table 2 presents the odds ratio of factors predicting in‐hospital mortality after cardiac arrest complicated by neurologic insult. Factors include use of TH after cardiac arrest, age, gender, year of admission, number of Charlson‐Deyo comorbidities, hospital teaching status, and volume tercile of hospitals that performed therapeutic hypothermia. Overall, patients who were older, male, and had greater comorbidities were statistically more likely to die after cardiac arrest complicated by neurologic insult. Regardless of whether they underwent TH, patients admitted to hospitals in the highest volume tercile of TH use were more likely to die after cardiac arrest. Adjusting for volume tercile, teaching hospital status was not independently associated with mortality after cardiac arrest. The adjusted odds ratio of mortality among patients undergoing therapeutic hypothermia was 0.80 (95% confidence interval [CI] 0.601.06, P = 0.11). The adjusted probability of inpatient mortality among patients undergoing therapeutic hypothermia was 57.5% (95% CI 50.764.3%) compared to those who did not 62.8% (95% CI 61.763.9%, P = 0.11).
| Variable | Odds Ratio of Hospital Mortality (95% CI) | P Value |
|---|---|---|
| ||
| No. observations | 46,651 | |
| Age* | ||
| 6569 | 1.19 (1.121.28) | <0.001 |
| 7074 | 1.29 (1.201.39) | <0.001 |
| 7579 | 1.55 (1.441.67) | <0.001 |
| 8084 | 1.79 (1.651.93) | <0.001 |
| 85 and over | 2.06 (1.892.25) | <0.001 |
| Male | 1.15 (1.101.21) | <0.001 |
| Teaching hospital | 1.13 (0.951.34) | 0.17 |
| No. Charlson‐Deyo comorbidities | 1.09 (1.081.10) | <0.001 |
| Year trend | 0.98 (0.970.99) | <0.001 |
| Volume tercile among hospitals performing TH | ||
| First tercile | 0.94 (0.791.12) | 0.48 |
| Second tercile | 1.03 (0.801.33) | 0.82 |
| Third tercile | 1.20 (1.051.36) | 0.006 |
| Therapeutic hypothermia | 0.80 (0.601.06) | 0.11 |
Figure 3 presents adjusted mortality after cardiac arrest in hospitals that did not perform TH, as well as adjusted mortality associated with TH for each volume tercile of hospitals that performed the procedure. Hospital mortality rates among patients not receiving TH after cardiac arrest were slightly higher in hospitals in the high volume tercile of TH (66.3%, 95% CI 63.868.8%) compared to hospitals in low and moderate volume terciles and to hospitals not performing TH (P < 0.001). Hospital mortality rates among low and moderate TH volume centers and in centers not performing TH were similar (62.3%, 61.3%, and 63.4%, respectively). Among both the low volume and moderate volume terciles, however, patients who underwent TH after cardiac arrest were significantly less likely to die in‐hospital compared to those who did not. For patients admitted to hospitals in the low volume tercile, those undergoing therapeutic hypothermia had an adjusted hospital mortality rate of 25.5% (95% CI 3.047.9%) compared to those who did not undergo TH (adjusted mortality 61.3%, 95% CI 57.465.1%), P < 0.001. In the moderate volume tercile, patients receiving therapeutic hypothermia had an adjusted hospital mortality rate of 31.0% (95% CI 9.2%52.8%) compared to 63.4% (95% CI 57.769.1%), P < 0.001, among those not undergoing the procedure. There was no statistically significant difference in adjusted mortality between those who underwent TH and those who did not, in hospitals in the highest volume tercile (P = 0.211). In addition to examining how volume of therapeutic hypothermia performed by hospitals affected the association between TH and hospital mortality, we also examined whether year of admission and teaching hospital independently modified the association. Neither year of admission nor teaching hospital statistically significantly affected the association between therapeutic hypothermia and hospital mortality after cardiac arrest at the P < 0.10 level.

DISCUSSION
In an administrative database of all admissions to California hospitals, we demonstrated that use of TH increased steadily since the publication of the initial clinical trials in 2002. The absolute level of TH utilization in our study undoubtedly represents a significant underestimation of actual TH utilization, however, our study does provide an assessment of the utilization trends over time. The bulk of TH use appears to be performed in a small group of high volume centers, and 89% of California hospitals did not perform TH during the study period (as assessed by procedure billing codes). Additionally, within the limitations of a retrospective, administrative claims‐based study design, TH appears to be associated with a similar in‐hospital mortality rate to that seen in clinical trials.3, 4 In exploratory analyses, there appears to be a particular benefit of TH in low and moderate volume centers, though these findings should be considered hypothesis‐generating.
Despite the body of evidence in favor of TH, utilization in our study and others appears quite low. In a 2005 survey of physicians, 87% of respondents had never used therapeutic hypothermia, citing inadequate data, technical limitations, and lack of incorporation in the Advanced Cardiac Life Support (ACLS) protocol as principal justifications.8 Other surveys have shown similar results and noted that critical care physicians and those working in large medical centers were more likely to adopt the therapy.9 Advocates of the therapy have suggested that an explicit hospital‐based plan developed by key stakeholders can help facilitate implementation.21 Accordingly, there is growing interest in developing centers of expertise in highly intensive therapies such as TH. For instance, the New York City Emergency Medical Service has begun to explore a protocol to divert TH candidates to specialized centers.22, 23 Some favorable results have been reported in individual hospitals and local hospital systems.2428
Our data suggest that TH is associated with an in‐hospital mortality rate that is comparable to that reported in the clinical trials. For example, in a 2009 meta‐analysis of 4 clinical trials and 1 abstract (481 patients in total), TH was associated with a 35% relative mortality benefit as compared to standard post‐resuscitation care.29 It has been estimated that broad TH implementation could save thousands of lives30 and many authors have advocated for its use and outlined explicit protocols for implementation.17 Furthermore, TH appears to be cost‐effective in line with other accepted therapies. Assuming the Hypothermia After Cardiac Arrest (HACA) trial inclusion criteria, even at extreme estimates for costs, the cost‐effectiveness of hypothermia remains less than $100,000 per quality‐adjusted life year.31
There are important limitations of this study. Our use of administrative claims data certainly underestimates the level of TH utilization, since we could only identify cases in which TH was included in the billing codes for the hospitalization. Hospitals may vary in utilization of this particular billing code for TH in ways that bias our estimated associations. The ICD‐9 code 99.81 for therapeutic hypothermia was also not developed for post‐cardiac arrest TH specifically, so use of the code may actually lag clinical utilization. Although the observed trend in TH utilization is likely mainly due to a true increase in utilization, it is possible that some of the observed increase is due to an increase in utilization of TH procedure billing codes. Our TH utilization estimates should be construed as a lower bound of the actual rates. Additionally, although the estimated real‐world mortality benefit of TH may be comparable to that of clinical trials, the equivalence of patients in our sample to those in published randomized trials is uncertain. Similarly, even after adjusting for age, gender, year of admission, comorbidities, hospital teaching status, and TH volume, there are likely many unmeasured variables that influence mortality in both the TH and comparison groups. There are also likely patients included in our comparison group who had both cardiac arrest or ventricular tachycardia and anoxic brain injury, but who were not candidates for TH as the episode of cardiac arrest followed rather than preceded the anoxic brain injury. Since we lack detailed clinical data about the TH cases (initial rhythm, time before return of circulation, preexisting disease states, etc.), we are unable to match controls directly to cases. Additionally, we lack data to assess neurologic recovery or quality of life after arrest.
The observation that a mortality benefit in our study could be detected only in low and moderate volume centers requires further exploration. Indeed, one might expect that high volume centers may have better outcomes with TH as a result of more robust infrastructure, technical experience, and available resources. Our finding that mortality benefits of TH appear concentrated in centers with low to moderate volume of TH utilization suggest at least 1 of 2 possibilities. First, low and moderate volume centers may perform TH in a subset of patients who benefit most from the intervention or, alternatively, in the most viable cardiac arrest cases (those who may fare well with or without the therapy). Consequently, we may observe relatively favorable outcomes in this group due to this selection bias. Second, high volume centersdespite having more expertisemay also attract patients at higher mortality risk due to referral bias. This would lead us to estimate lower mortality benefits associated with TH in these high volume centers. Indeed, greater observed mortality at high volume centers regardless of TH status suggests that overall acuity is higher at high volume centers. While our inferences are greatly affected by issues of case selection and referral bias, it also important to consider the possibility that the estimated mortality benefit of TH in higher volume centers is lower because of the selection of patients who do not meet current guidelines for treatment with TH. Distinguishing whether the selection of patients undergoing TH at high volume centers is appropriate or inappropriate based on current guidelines is an important issue that merits further research with datasets with more refined patient clinical information.
In summary, therapeutic hypothermia utilization is low, but the rate of implementation has increased since the publication of the initial clinical trials in 2002. The bulk of TH utilization appears limited to a subset of high volume centers, and most centers in California appear to have not used the therapy. Real‐world in‐hospital mortality associated with TH is comparable to that reported in randomized clinical trials.
Acknowledgements
Disclosures: Dr Romley received support from NIH grant R03AG031990‐A1. Dr Noseworthy received support from the Max Schaldach Fellowship in Cardiac Pacing and Electrophysiology granted by the Heart Rhythm Society. The design, conduct, analysis, interpretation, and presentation of the data are the responsibility of the investigators, with no involvement from the funding sources. The contents of this article have not been published in any other peer‐reviewed media, and the manuscript is not under review elsewhere. All authors listed have contributed sufficiently to this project to be included as authors. The authors have no conflict of interest, financial or otherwise.
There are over 350,000 cases of out‐of‐hospital cardiac arrest (OHCA) each year in the United States1, 2 and, with supportive therapy alone, only a fraction of victims survive to hospital discharge. Rapid intervention including cardiopulmonary resuscitation in the moments following arrest is critical to minimizing neurologic injury, morbidity, and mortality. In 2002, two small randomized controlled trials showed a survival benefit of therapeutic hypothermia (TH) when provided within 12 hours after return of circulation following an OHCA and, to date, TH remains one of the few interventions with proven mortality benefit after initial cardiopulmonary resuscitation.3, 4 Since 2003, TH has been incorporated into the American Heart Association practice guidelines57 and use of TH has steadily increased, but widespread clinical uptake remains low.8, 9
The initial studies that evaluated TH were small, with only 189 patients included in the TH arms of the 2 trials combined. To date, only a few studies have replicated this initial observation in real‐world settings, with little analysis of outcomes in US centers in particular.1013 Accordingly, we aimed to examine the real‐world experience with TH in the United States using a large administrative claims database of all California hospital admissions to describe utilization trends, hospital mortality, and volumeoutcome relationships associated with the intervention.
MATERIALS AND METHODS
Data
We identified all admissions to California hospitals during 19992008 based on discharge records from the California Office of Statewide Health Planning and Development. Our study period included cases of TH performed prior to the 2002 major clinical trials, since TH was in occasional use prior to the publication of these trials. The data was de‐identified and publicly available, and therefore exempt from review by the Institutional Review Board. In addition to hospital name, each discharge record included patient age, gender, admission year, International Classification of Disease, Ninth Revision (ICD‐9) code for presenting primary and secondary diagnoses, procedure codes, and disposition (discharge to home or rehabilitation, in‐hospital death). All California hospitals were included in the registry (n = 419). We defined teaching status for each hospital based on membership in the Council of Teaching Hospitals, as reported in the American Hospital Association's Annual Survey (n = 19 teaching hospitals).14
Setting and Participants
We used discharge diagnoses to identify patients who could be considered eligible for therapeutic hypothermia after cardiac arrest. We classified patients as eligible for therapeutic hypothermia after cardiac arrest based on ICD‐9 diagnosis codes that indicated the presence of both cardiac arrest and anoxic brain injury in the administrative diagnoses. Because of known imprecision in using billing codes to identify patients with cardiac arrest,15, 16 we broadly defined cardiac arrest to include those patients with ICD‐9 codes for cardiac arrest, ventricular fibrillation (VF), or ventricular tachycardia (VT) (see Supporting Table 1 in the online version of this article). We could not distinguish between out‐of‐hospital and in‐hospital cardiac arrest based on administrative diagnoses. To ensure that we included only patients with cardiac arrest complicated by neurologic insult, we required an ICD‐9 diagnosis of either anoxic brain injury, coma, or persistent vegetative state. Claims did not allow us to distinguish among initial cardiac arrest rhythms (VF vs pulseless VT vs asystole). Patients younger than 18 years of age, and those who were pregnant, suffered traumatic brain injury, intracranial hemorrhage, metastatic cancer, or dementia were excluded.3, 17 We did not exclude patients based on coagulopathy (which is considered a contraindication to TH), since ICD‐9 coding did not allow us to determine the severity of the coagulopathy or whether it was a result of therapeutic hypothermia itself.
We used the ICD‐9 procedure code (99.81) for TH to first identify patients who underwent TH from 1999 to 2008. Since this code also applies to TH used during cardiac and neurosurgery, we examined each of these cases and excluded individuals who underwent cardiac surgery or neurosurgery during the hospitalization. As in our eligible for TH definition, we excluded patients younger than 18 years of age, and those who were pregnant, suffered traumatic brain injury, intracranial hemorrhage, metastatic cancer, or dementia. Patients who underwent therapeutic hypothermia but for whom a specific procedure code was not recorded in the discharge abstractperhaps because the medical institution did not directly bill for the procedurecould not be identified.
Statistical Analysis
We used a multivariable logistic model to estimate differences in hospital mortality after cardiac arrest associated with use of therapeutic hypothermia. We conducted 2 specifications. In our baseline specification, we accounted for case‐mix differences between those who underwent TH and those who did not by adjusting for age, gender, year of admission, the number of Charlson‐Deyo comorbidities,1820 and hospital teaching status. Because mortality after cardiac arrest in centers that perform therapeutic hypothermia may be different from centers that do not, even for patients who do not undergo the procedure, we included indicators for volume tercile of therapeutic hypothermia cases performed. Volume of therapeutic hypothermia was defined at the hospital level as the total number of cases performed by that hospital from 1999 to 2008.
In order to explore how hospital teaching status, volume of therapeutic hypothermia procedures (broken into terciles), and year of admission affected the association between hospital mortality after cardiac arrest and therapeutic hypothermia, our baseline logistic model was expanded to include interactions between therapeutic hypothermia and each of these variables. The interaction between therapeutic hypothermia and year explored whether the effectiveness of the procedure changed over time, as case‐selection, method of therapeutic hypothermia (cold saline vs commercially available devices), and experience changed in California hospitals. For both specifications, we reported the odds ratio of hospital mortality among patients undergoing therapeutic hypothermia, as well as risk‐adjusted mortality for both TH and non‐TH groups.
STATA version 11 (STATA Corp, College Station, TX) was used for statistical analyses, and a 2‐sided P 0.05 was used.
RESULTS
Descriptive Data
Table 1 reports summary statistics for patients with cardiac arrest complicated by neurologic insult (anoxic brain injury, coma, or persistent vegetative state) between 1999 and 2008. Across all years, 204 patients were identified as undergoing TH. In comparison, 105 patients were identified as undergoing TH in 2008 alone. Patients who underwent TH were less likely to be male (30.7% vs 44.6% male, P < 0.01), were younger (63.9 15.0 years vs 67.3 15.7 years, P = 0.03), and had equivalent numbers of Charlson‐Deyo comorbidities (2.5 2.0 diagnoses vs 2.5 2.0 diagnoses, P = 0.89). Therapeutic hypothermia was more commonly employed at teaching hospitals (51/3709 [1.4%] vs 153/42,942 [0.4%], P < 0.01). There was a trend toward decreased unadjusted mortality among patients who underwent therapeutic hypothermia compared with those who did not (56.9% vs 62.8%, P = 0.08).
| Therapeutic Hypothermia | No Therapeutic Hypothermia | P Value | |
|---|---|---|---|
| |||
| No. observations | 204 | 46,629 | |
| No. cases in teaching hospitals | 51 | 3,658 | |
| No. cases in non‐teaching hospitals | 153 | 42,789 | |
| Age, y | 63.9 15.0 | 67.3 15.7 | 0.06 |
| Male | 30.7 | 44.6 | <0.01 |
| Hospital mortality, % | 56.9 | 62.8 | 0.08 |
| Comorbidities | |||
| No. Charlson‐Deyo comorbidities | 2.5 2.0 | 2.5 2.0 | 0.89 |
| Coronary artery disease, % | 48.0 | 38.0 | <0.01 |
| Acute myocardial infarction, % | 42.6 | 28.9 | <0.01 |
| Congestive heart failure, % | 27.9 | 35.3 | 0.03 |
| Hypertension, % | 36.3 | 33.2 | 0.83 |
| Acute renal failure, % | 33.3 | 26.6 | 0.03 |
| Diabetes mellitus, % | 30.9 | 23.0 | <0.01 |
| Chronic obstructive pulmonary disease, % | 10.3 | 19.3 | <0.01 |
Figures 1 and 2 provide additional aggregate statistics on therapeutic hypothermia in California hospitals. Figure 1 plots the number of therapeutic hypothermia cases recorded in the administrative discharge registry between 1999 and 2008. Of the 204 total cases identified during this period, 178 (87.3%) were performed between 2006 and 2008. Figure 2 shows the distribution of TH cases across centers that performed therapeutic hypothermia (n = 47 hospitals, 11.3 % of all hospitals). Ten centers accounted for 124/204 (60.7%) of the total patients treated with TH after cardiac arrest; the top 3 centers accounted for 64 (31.4%) of the treated patients. Twenty‐seven hospitals were identified as performing therapeutic hypothermia on only 1 or 2 patients between 1999 and 2008.


Risk‐Adjusted Mortality
Table 2 presents the odds ratio of factors predicting in‐hospital mortality after cardiac arrest complicated by neurologic insult. Factors include use of TH after cardiac arrest, age, gender, year of admission, number of Charlson‐Deyo comorbidities, hospital teaching status, and volume tercile of hospitals that performed therapeutic hypothermia. Overall, patients who were older, male, and had greater comorbidities were statistically more likely to die after cardiac arrest complicated by neurologic insult. Regardless of whether they underwent TH, patients admitted to hospitals in the highest volume tercile of TH use were more likely to die after cardiac arrest. Adjusting for volume tercile, teaching hospital status was not independently associated with mortality after cardiac arrest. The adjusted odds ratio of mortality among patients undergoing therapeutic hypothermia was 0.80 (95% confidence interval [CI] 0.601.06, P = 0.11). The adjusted probability of inpatient mortality among patients undergoing therapeutic hypothermia was 57.5% (95% CI 50.764.3%) compared to those who did not 62.8% (95% CI 61.763.9%, P = 0.11).
| Variable | Odds Ratio of Hospital Mortality (95% CI) | P Value |
|---|---|---|
| ||
| No. observations | 46,651 | |
| Age* | ||
| 6569 | 1.19 (1.121.28) | <0.001 |
| 7074 | 1.29 (1.201.39) | <0.001 |
| 7579 | 1.55 (1.441.67) | <0.001 |
| 8084 | 1.79 (1.651.93) | <0.001 |
| 85 and over | 2.06 (1.892.25) | <0.001 |
| Male | 1.15 (1.101.21) | <0.001 |
| Teaching hospital | 1.13 (0.951.34) | 0.17 |
| No. Charlson‐Deyo comorbidities | 1.09 (1.081.10) | <0.001 |
| Year trend | 0.98 (0.970.99) | <0.001 |
| Volume tercile among hospitals performing TH | ||
| First tercile | 0.94 (0.791.12) | 0.48 |
| Second tercile | 1.03 (0.801.33) | 0.82 |
| Third tercile | 1.20 (1.051.36) | 0.006 |
| Therapeutic hypothermia | 0.80 (0.601.06) | 0.11 |
Figure 3 presents adjusted mortality after cardiac arrest in hospitals that did not perform TH, as well as adjusted mortality associated with TH for each volume tercile of hospitals that performed the procedure. Hospital mortality rates among patients not receiving TH after cardiac arrest were slightly higher in hospitals in the high volume tercile of TH (66.3%, 95% CI 63.868.8%) compared to hospitals in low and moderate volume terciles and to hospitals not performing TH (P < 0.001). Hospital mortality rates among low and moderate TH volume centers and in centers not performing TH were similar (62.3%, 61.3%, and 63.4%, respectively). Among both the low volume and moderate volume terciles, however, patients who underwent TH after cardiac arrest were significantly less likely to die in‐hospital compared to those who did not. For patients admitted to hospitals in the low volume tercile, those undergoing therapeutic hypothermia had an adjusted hospital mortality rate of 25.5% (95% CI 3.047.9%) compared to those who did not undergo TH (adjusted mortality 61.3%, 95% CI 57.465.1%), P < 0.001. In the moderate volume tercile, patients receiving therapeutic hypothermia had an adjusted hospital mortality rate of 31.0% (95% CI 9.2%52.8%) compared to 63.4% (95% CI 57.769.1%), P < 0.001, among those not undergoing the procedure. There was no statistically significant difference in adjusted mortality between those who underwent TH and those who did not, in hospitals in the highest volume tercile (P = 0.211). In addition to examining how volume of therapeutic hypothermia performed by hospitals affected the association between TH and hospital mortality, we also examined whether year of admission and teaching hospital independently modified the association. Neither year of admission nor teaching hospital statistically significantly affected the association between therapeutic hypothermia and hospital mortality after cardiac arrest at the P < 0.10 level.

DISCUSSION
In an administrative database of all admissions to California hospitals, we demonstrated that use of TH increased steadily since the publication of the initial clinical trials in 2002. The absolute level of TH utilization in our study undoubtedly represents a significant underestimation of actual TH utilization, however, our study does provide an assessment of the utilization trends over time. The bulk of TH use appears to be performed in a small group of high volume centers, and 89% of California hospitals did not perform TH during the study period (as assessed by procedure billing codes). Additionally, within the limitations of a retrospective, administrative claims‐based study design, TH appears to be associated with a similar in‐hospital mortality rate to that seen in clinical trials.3, 4 In exploratory analyses, there appears to be a particular benefit of TH in low and moderate volume centers, though these findings should be considered hypothesis‐generating.
Despite the body of evidence in favor of TH, utilization in our study and others appears quite low. In a 2005 survey of physicians, 87% of respondents had never used therapeutic hypothermia, citing inadequate data, technical limitations, and lack of incorporation in the Advanced Cardiac Life Support (ACLS) protocol as principal justifications.8 Other surveys have shown similar results and noted that critical care physicians and those working in large medical centers were more likely to adopt the therapy.9 Advocates of the therapy have suggested that an explicit hospital‐based plan developed by key stakeholders can help facilitate implementation.21 Accordingly, there is growing interest in developing centers of expertise in highly intensive therapies such as TH. For instance, the New York City Emergency Medical Service has begun to explore a protocol to divert TH candidates to specialized centers.22, 23 Some favorable results have been reported in individual hospitals and local hospital systems.2428
Our data suggest that TH is associated with an in‐hospital mortality rate that is comparable to that reported in the clinical trials. For example, in a 2009 meta‐analysis of 4 clinical trials and 1 abstract (481 patients in total), TH was associated with a 35% relative mortality benefit as compared to standard post‐resuscitation care.29 It has been estimated that broad TH implementation could save thousands of lives30 and many authors have advocated for its use and outlined explicit protocols for implementation.17 Furthermore, TH appears to be cost‐effective in line with other accepted therapies. Assuming the Hypothermia After Cardiac Arrest (HACA) trial inclusion criteria, even at extreme estimates for costs, the cost‐effectiveness of hypothermia remains less than $100,000 per quality‐adjusted life year.31
There are important limitations of this study. Our use of administrative claims data certainly underestimates the level of TH utilization, since we could only identify cases in which TH was included in the billing codes for the hospitalization. Hospitals may vary in utilization of this particular billing code for TH in ways that bias our estimated associations. The ICD‐9 code 99.81 for therapeutic hypothermia was also not developed for post‐cardiac arrest TH specifically, so use of the code may actually lag clinical utilization. Although the observed trend in TH utilization is likely mainly due to a true increase in utilization, it is possible that some of the observed increase is due to an increase in utilization of TH procedure billing codes. Our TH utilization estimates should be construed as a lower bound of the actual rates. Additionally, although the estimated real‐world mortality benefit of TH may be comparable to that of clinical trials, the equivalence of patients in our sample to those in published randomized trials is uncertain. Similarly, even after adjusting for age, gender, year of admission, comorbidities, hospital teaching status, and TH volume, there are likely many unmeasured variables that influence mortality in both the TH and comparison groups. There are also likely patients included in our comparison group who had both cardiac arrest or ventricular tachycardia and anoxic brain injury, but who were not candidates for TH as the episode of cardiac arrest followed rather than preceded the anoxic brain injury. Since we lack detailed clinical data about the TH cases (initial rhythm, time before return of circulation, preexisting disease states, etc.), we are unable to match controls directly to cases. Additionally, we lack data to assess neurologic recovery or quality of life after arrest.
The observation that a mortality benefit in our study could be detected only in low and moderate volume centers requires further exploration. Indeed, one might expect that high volume centers may have better outcomes with TH as a result of more robust infrastructure, technical experience, and available resources. Our finding that mortality benefits of TH appear concentrated in centers with low to moderate volume of TH utilization suggest at least 1 of 2 possibilities. First, low and moderate volume centers may perform TH in a subset of patients who benefit most from the intervention or, alternatively, in the most viable cardiac arrest cases (those who may fare well with or without the therapy). Consequently, we may observe relatively favorable outcomes in this group due to this selection bias. Second, high volume centersdespite having more expertisemay also attract patients at higher mortality risk due to referral bias. This would lead us to estimate lower mortality benefits associated with TH in these high volume centers. Indeed, greater observed mortality at high volume centers regardless of TH status suggests that overall acuity is higher at high volume centers. While our inferences are greatly affected by issues of case selection and referral bias, it also important to consider the possibility that the estimated mortality benefit of TH in higher volume centers is lower because of the selection of patients who do not meet current guidelines for treatment with TH. Distinguishing whether the selection of patients undergoing TH at high volume centers is appropriate or inappropriate based on current guidelines is an important issue that merits further research with datasets with more refined patient clinical information.
In summary, therapeutic hypothermia utilization is low, but the rate of implementation has increased since the publication of the initial clinical trials in 2002. The bulk of TH utilization appears limited to a subset of high volume centers, and most centers in California appear to have not used the therapy. Real‐world in‐hospital mortality associated with TH is comparable to that reported in randomized clinical trials.
Acknowledgements
Disclosures: Dr Romley received support from NIH grant R03AG031990‐A1. Dr Noseworthy received support from the Max Schaldach Fellowship in Cardiac Pacing and Electrophysiology granted by the Heart Rhythm Society. The design, conduct, analysis, interpretation, and presentation of the data are the responsibility of the investigators, with no involvement from the funding sources. The contents of this article have not been published in any other peer‐reviewed media, and the manuscript is not under review elsewhere. All authors listed have contributed sufficiently to this project to be included as authors. The authors have no conflict of interest, financial or otherwise.
- , , , et al. Sudden cardiac death in the United States, 1989 to 1998. Circulation. 2001;104(18):2158–2163.
- , . Projections of global mortality and burden of disease from 2002 to 2030. PLoS Med. 2006;3(11):e442.
- , , , et al. Treatment of comatose survivors of out‐of‐hospital cardiac arrest with induced hypothermia. N Engl J Med. 2002;346(8):557–563.
- Hypothermia After Cardiac Arrest Study Group. Mild therapeutic hypothermia to improve the neurologic outcome after cardiac arrest. N Engl J Med. 2002;346(8):549–556.
- , , , et al. Therapeutic hypothermia after cardiac arrest: an advisory statement by the Advanced Life Support Task Force of the International Liaison Committee on Resuscitation. Circulation. 2003;108(1):118–121.
- 2005 American Heart Association guidelines for cardiopulmonary resuscitation and emergency cardiovascular care: part 7.5: postresuscitation support. Circulation. 2005;112(suppl I):IV‐84–IV‐88.
- , , , et al. Part 8: advanced life support: 2010 international consensus on cardiopulmonary resuscitation and emergency cardiovascular care science with treatment recommendations. Resuscitation. 2010;81(suppl 1):e93–e174.
- , , , et al. Induced hypothermia is underused after resuscitation from cardiac arrest: a current practice survey. Resuscitation. 2005;64(2):181–186.
- , , , et al. Therapeutic hypothermia utilization among physicians after resuscitation from cardiac arrest. Crit Care Med. 2006;34(7):1935–1940.
- , , , et al. Adverse events and their relation to mortality in out‐of‐hospital cardiac arrest patients treated with therapeutic hypothermia. Crit Care Med.39(1):57–64.
- , , , et al. Outcome, timing and adverse events in therapeutic hypothermia after out‐of‐hospital cardiac arrest. Acta Anaesthesiol Scand. 2009;53(7):926–934.
- . Clinical application of mild therapeutic hypothermia after cardiac arrest. Crit Care Med. 2007;35(4):1041–1047.
- , , , et al. Therapeutic hypothermia after out‐of‐hospital cardiac arrest: evaluation of a regional system to increase access to cooling. Circulation. 2011;124(2):206–214.
- American Hospital Association (AHA). American Hospital Association 2001 Annual Survey. Chicago, IL: Health Forum, LLC.
- , , . A multicenter study of the coding accuracy of hospital discharge administrative data for patients admitted to cardiac care units in Ontario. Am Heart J. 2002;144(2):290–296.
- . Accuracy of ICD‐9‐CM coding for the identification of patients with acute ischemic stroke: effect of modifier codes. Stroke. 1998;29(8):1602–1604.
- . Targeted temperature management for comatose survivors of cardiac arrest. N Engl J Med. 2010;363(13):1256–1264.
- , , , et al. A new method of classifying prognostic comorbidity in longitudinal studies: development and validation. J Chronic Dis. 1987;40(5):373–383.
- , , . Adapting a clinical comorbidity index for use with ICD‐9‐CM administrative databases. J Clin Epidemiol. 1992;45(6):613–619.
- , , . Adapting a clinical comorbidity index for use with ICD‐9‐CM administrative data: differing perspectives. J Clin Epidemiol. 1993;46(10):1075–1079; discussion 1081–1090.
- , , , et al. Practical implementation of therapeutic hypothermia after cardiac arrest. Hosp Pract (Minneap). 2009;37(1):71–83.
- . Paramedics will employ new therapy in cardiac arrest cases. New York Times. August 3, 2010:A18.
- , , , et al. Resuscitation center designation: recommendations for emergency medical services practices. Prehosp Emerg Care. 2010;14(1):51–61.
- , , , et al. Implementation of a hospital‐wide protocol for induced hypothermia following successfully resuscitated cardiac arrest. Crit Pathw Cardiol. 2010;9(4):216–220.
- , , , et al. Therapeutic hypothermia protocol in a community emergency department. West J Emerg Med. 2010;11(4):367–372.
- , , , et al. Implementation of a post‐cardiac arrest care bundle including therapeutic hypothermia and hemodynamic optimization in comatose patients with return of spontaneous circulation after out‐of‐hospital cardiac arrest: a feasibility study. Shock. 2011;35(4):360–366.
- , , , et al. Improved out‐of‐hospital cardiac arrest survival after the sequential implementation of 2005 AHA guidelines for compressions, ventilations, and induced hypothermia: the Wake County experience. Ann Emerg Med. 2010;56(4):348–357.
- , , , et al. Community‐based application of mild therapeutic hypothermia for survivors of cardiac arrest. South Med J. 2010;103(4):295–300.
- , , , et al. Hypothermia for neuroprotection in adults after cardiopulmonary resuscitation. Cochrane Database Syst Rev 2009(4):CD004128.
- , . Therapeutic hypothermia after cardiac arrest and myocardial infarction. Best Pract Res Clin Anaesthesiol. 2008;22(4):711–728.
- , , , et al. Cost‐effectiveness of therapeutic hypothermia after cardiac arrest. Circ Cardiovasc Qual Outcomes. 2009;2(5):421–428.
- , , , et al. Sudden cardiac death in the United States, 1989 to 1998. Circulation. 2001;104(18):2158–2163.
- , . Projections of global mortality and burden of disease from 2002 to 2030. PLoS Med. 2006;3(11):e442.
- , , , et al. Treatment of comatose survivors of out‐of‐hospital cardiac arrest with induced hypothermia. N Engl J Med. 2002;346(8):557–563.
- Hypothermia After Cardiac Arrest Study Group. Mild therapeutic hypothermia to improve the neurologic outcome after cardiac arrest. N Engl J Med. 2002;346(8):549–556.
- , , , et al. Therapeutic hypothermia after cardiac arrest: an advisory statement by the Advanced Life Support Task Force of the International Liaison Committee on Resuscitation. Circulation. 2003;108(1):118–121.
- 2005 American Heart Association guidelines for cardiopulmonary resuscitation and emergency cardiovascular care: part 7.5: postresuscitation support. Circulation. 2005;112(suppl I):IV‐84–IV‐88.
- , , , et al. Part 8: advanced life support: 2010 international consensus on cardiopulmonary resuscitation and emergency cardiovascular care science with treatment recommendations. Resuscitation. 2010;81(suppl 1):e93–e174.
- , , , et al. Induced hypothermia is underused after resuscitation from cardiac arrest: a current practice survey. Resuscitation. 2005;64(2):181–186.
- , , , et al. Therapeutic hypothermia utilization among physicians after resuscitation from cardiac arrest. Crit Care Med. 2006;34(7):1935–1940.
- , , , et al. Adverse events and their relation to mortality in out‐of‐hospital cardiac arrest patients treated with therapeutic hypothermia. Crit Care Med.39(1):57–64.
- , , , et al. Outcome, timing and adverse events in therapeutic hypothermia after out‐of‐hospital cardiac arrest. Acta Anaesthesiol Scand. 2009;53(7):926–934.
- . Clinical application of mild therapeutic hypothermia after cardiac arrest. Crit Care Med. 2007;35(4):1041–1047.
- , , , et al. Therapeutic hypothermia after out‐of‐hospital cardiac arrest: evaluation of a regional system to increase access to cooling. Circulation. 2011;124(2):206–214.
- American Hospital Association (AHA). American Hospital Association 2001 Annual Survey. Chicago, IL: Health Forum, LLC.
- , , . A multicenter study of the coding accuracy of hospital discharge administrative data for patients admitted to cardiac care units in Ontario. Am Heart J. 2002;144(2):290–296.
- . Accuracy of ICD‐9‐CM coding for the identification of patients with acute ischemic stroke: effect of modifier codes. Stroke. 1998;29(8):1602–1604.
- . Targeted temperature management for comatose survivors of cardiac arrest. N Engl J Med. 2010;363(13):1256–1264.
- , , , et al. A new method of classifying prognostic comorbidity in longitudinal studies: development and validation. J Chronic Dis. 1987;40(5):373–383.
- , , . Adapting a clinical comorbidity index for use with ICD‐9‐CM administrative databases. J Clin Epidemiol. 1992;45(6):613–619.
- , , . Adapting a clinical comorbidity index for use with ICD‐9‐CM administrative data: differing perspectives. J Clin Epidemiol. 1993;46(10):1075–1079; discussion 1081–1090.
- , , , et al. Practical implementation of therapeutic hypothermia after cardiac arrest. Hosp Pract (Minneap). 2009;37(1):71–83.
- . Paramedics will employ new therapy in cardiac arrest cases. New York Times. August 3, 2010:A18.
- , , , et al. Resuscitation center designation: recommendations for emergency medical services practices. Prehosp Emerg Care. 2010;14(1):51–61.
- , , , et al. Implementation of a hospital‐wide protocol for induced hypothermia following successfully resuscitated cardiac arrest. Crit Pathw Cardiol. 2010;9(4):216–220.
- , , , et al. Therapeutic hypothermia protocol in a community emergency department. West J Emerg Med. 2010;11(4):367–372.
- , , , et al. Implementation of a post‐cardiac arrest care bundle including therapeutic hypothermia and hemodynamic optimization in comatose patients with return of spontaneous circulation after out‐of‐hospital cardiac arrest: a feasibility study. Shock. 2011;35(4):360–366.
- , , , et al. Improved out‐of‐hospital cardiac arrest survival after the sequential implementation of 2005 AHA guidelines for compressions, ventilations, and induced hypothermia: the Wake County experience. Ann Emerg Med. 2010;56(4):348–357.
- , , , et al. Community‐based application of mild therapeutic hypothermia for survivors of cardiac arrest. South Med J. 2010;103(4):295–300.
- , , , et al. Hypothermia for neuroprotection in adults after cardiopulmonary resuscitation. Cochrane Database Syst Rev 2009(4):CD004128.
- , . Therapeutic hypothermia after cardiac arrest and myocardial infarction. Best Pract Res Clin Anaesthesiol. 2008;22(4):711–728.
- , , , et al. Cost‐effectiveness of therapeutic hypothermia after cardiac arrest. Circ Cardiovasc Qual Outcomes. 2009;2(5):421–428.
Copyright © 2012 Society of Hospital Medicine
Rethinking Cardiac Risk Reduction
Noncardiac surgery is frequently associated with major adverse cardiac events. Prevention of these events has traditionally focused on risk estimation and targeted interventions prior to surgical intervention.1 Cardiac risk is assessed through clinical prediction rules, such as the revised cardiac risk index (RCRI) or the American Society of Anesthesiology (ASA) classification. In patients deemed to be at high risk of adverse cardiac events, discretionary preoperative testing, medical treatments, and interventions are implemented.2 However, even when executed optimally, this approach fails to protect all patients. Thus, many patients undergoing noncardiac surgery continue to experience perioperative myocardial infarction (MI) and death.3
Recently, a prospective international study involving 15,133 patients reported that cardiac troponin levels measured within 3 days of noncardiac surgery were strongly associated with 30‐day mortality.4 This study is but the latest in a series of investigations that have concluded that postoperative measurement of cardiac troponin is a better predictor of cardiac outcomes than preoperative‐risk algorithms.48 Intuitively, this link is not surprising. Surgery represents the ultimate physiologic stress test. In the setting of hemodynamic changes and sustained oxygen supply:demand mismatch, it is hardly shocking that those with significant epicardial coronary stenosis or major subendocardial ischemia suffer poorer outcomes. What is perhaps most important about the association between troponin release and clinical outcomes, however, is the foundation it provides for a novel framework to improve perioperative care: using troponin measurement to guide treatment and interventions in the postoperative setting.
MODEL TO IMPROVE POSTOPERATIVE CARDIAC OUTCOMES
We hypothesize that the postoperative period can be seized as a golden moment to ameliorate cardiac risk and improve clinical outcomes for 3 reasons. First, those who release cardiac troponin during surgery declare themselves as harboring myocardial territories in jeopardy. In effect, the peripheral presence of troponin amounts to a cry for help in a population where many cardiac events are silent and thus remain clinically unnoticed.9, 10 Routine postoperative monitoring for this biomarker can thus help identify a population that would ordinarily escape detection until the precipitation of a major clinical event. Second, as the pathophysiology of cardiac events centers around rupture of vulnerable plaque (type‐1 MI), supply:demand mismatch (type‐2 MI), or both of these phenomena, escalation or institution of important medical risk‐reducing treatments (such as aspirin, beta‐blockers, or statins), can be established to improve outcomes. The ability to initiate these treatments in a monitored setting may offset and address many of the risks feared with these therapies, including hypotension, stroke, or rhabdomyolysis. Third, as patients convalesce from surgery, timely and discretionary cardiac testing or interventions can be undertaken in those identified as being at elevated cardiovascular risk prior to discharge. In sum, the trifecta of clearly recognizing those at risk, instituting interventions in controlled settings, and performing well‐timed interventions could translate into greater odds of survival in this cohort.
Owing to their intimate involvement with patients in the perioperative setting, hospitalists are uniquely suited for implementing a troponin surveillance paradigm. Yet, how may such a schema be realized? In Figure 1, we outline a pathway with which to implement this approach. This model replicates perioperative management workflow, calling for 3 discrete interventionspreoperative assessment, perioperative surveillance, and postoperative risk stratification:
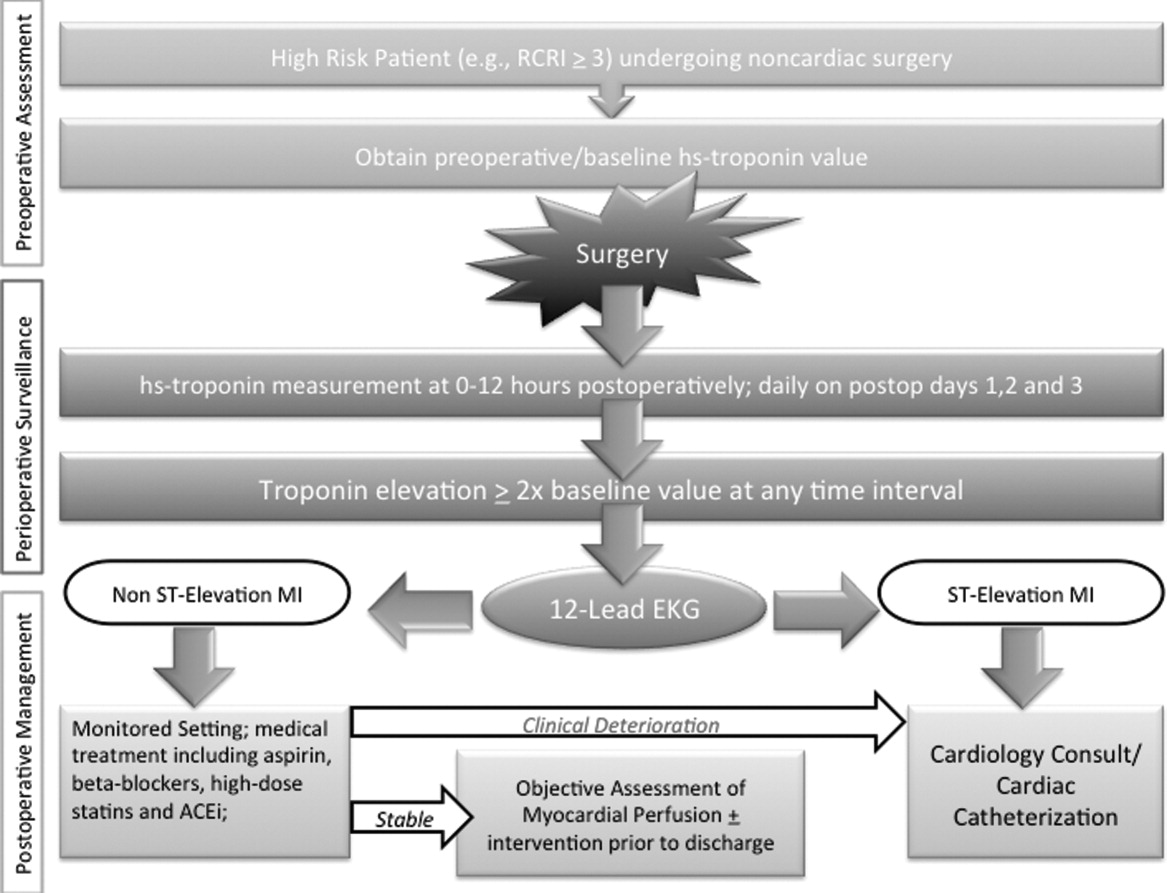
-
Preoperative assessment: Patients estimated to be at high risk of perioperative cardiac events (eg, RCRI 3) should undergo baseline, preoperative high‐sensitivity troponin measurement, as the frequency of major adverse cardiac outcomes is significant this group (11%).11 The importance of a baseline measurement of troponin cannot be overstated, as biologic variation of this marker can impact subsequent management.12, 13 Hospitalists can obtain these assays in preoperative clinics at the time of initial review and risk‐estimation.
-
Perioperative surveillance: Following the model implemented by a recent study, serial troponin surveillance should commence 612 hours after surgery, then daily on the first, second, and third day.4 As the optimal cardiac troponin threshold for the diagnosis of perioperative ischemia remains uncertain, doubling of baseline troponin levels may serve as a logical signal for further evaluation.14 The electrocardiographic pattern may be used as a branch point for immediate management at this stage: ST‐segment elevation MI may be treated aggressively with an early invasive strategy and cardiac catheterization when apropos (much as in the nonoperative context). However, as most perioperative cardiac events are non‐ST elevation MIs, treatment of this subset should center on initiation or escalation of medical therapy. Thus, achieving heart‐rate control in order to preserve coronary perfusion by careful titration of beta‐blockers, and initiation/up‐titration of statins to temper unstable atherosclerotic plaques represent cornerstones of this approach. Other potential therapeutic entities such as initiation of angiotensin‐converting‐enzyme (ACE)‐inhibitors in the context of left‐ventricular dysfunction, and/or antiplatelet agents such as aspirin, may also be relevant risk‐reducing tools.
-
Postoperative risk stratification: Prior to hospital discharge, objective assessment of myocardial perfusion and viability should occur in selected individuals with troponin elevations. Modalities that may be utilized in this context include either noninvasive imaging or occasional coronary angiography in patients with unstable coronary syndromes. Defining an optimal approach must be contextual, as interventions that may subsequently mandate uninterrupted antiplatelet treatment may be logistically challenging in this setting.
LIMITATIONS
Although this implementation model is plausible, it generates many questions that must be tested through the lens of a randomized controlled study. For instance, what is the optimal strategy for intervention in the postoperative setting? Though medical treatment with statins and beta‐blockade may represent the mainstay of treatment, are these therapies safe during potential clinical instability? Can net benefit be realized through judicious use of coronary angiography and revascularization? Can the artifact of heightened awareness and reporting of postoperative cardiac events mar the reported quality of a hospital? Finally, though cardiac troponin has been shown to be a strong and independent predictor of mortality, which troponin assay, reference range, and troponometric standard to use remain unclear.15 Whether or not these interventions translate into lesser mortality and improved clinical outcomes represents the raison d'etre for this experimental approach. As no alternative approach to define and target patients at high risk of adverse outcome after seemingly uneventful surgery currently exists, we believe that this hypothetical paradigm is worthy of further investigation.
CONCLUSION
For decades, perioperative practitioners have searched for a divining rod to reduce risk during surgery. While our efforts have been focused on the preoperative setting, the postoperative setting represents an as yet untapped and potentially profound period in the quest to improving surgical outcomes. Through our proximity to patients in the perioperative setting, hospitalists are ideal agents to test, deliver, and bring about this change. It is time for a postoperative carpe diem.
- , , , et al. Predictors of cardiac events after major vascular surgery: role of clinical characteristics, dobutamine echocardiography, and beta‐blocker therapy. JAMA. 2001;285(14):1865–1873.
- , , , et al. Combining clinical and thallium data optimizes preoperative assessment of cardiac risk before major vascular surgery. Ann Intern Med. 1989;110(11):859–866.
- , , , et al. An estimation of the global volume of surgery: a modelling strategy based on available data. Lancet. 2008;372(9633):139–144.
- VISION Study Investigators. Association between postoperative troponin levels and 30‐day mortlality among patients undergoing noncardiac surgery. JAMA. 2012;307(21):2295–2304.
- , , , et al. Postoperative levels of cardiac troponin versus CK‐MB and high‐sensitivity C‐reactive protein for the prediction of 1‐year cardiovascular outcome in patients undergoing vascular surgery. Coron Artery Dis. 2011;22(6):428–434.
- , , , et al. Peak postoperative troponin levels outperform preoperative cardiac risk indices as predictors of long‐term mortality after vascular surgery Troponins and postoperative outcomes. J Crit Care. 2012;27(1):66–72.
- , , , et al. Prognostic value of troponin and creatine kinase muscle and brain isoenzyme measurement after noncardiac surgery: a systematic review and meta‐analysis. Anesthesiology. 2011;114(4):796–806.
- , , . Outcomes in vascular surgical patients with isolated postoperative troponin leak: a meta‐analysis. Anaesthesia. 2011;66(7):604–610.
- , , , et al. Characteristics and short‐term prognosis of perioperative myocardial infarction in patients undergoing noncardiac surgery: a cohort study. Ann Intern Med. 2011;154(8):523–528.
- , , , , . Perioperative myocardial necrosis in patients at high cardiovascular risk undergoing elective non‐cardiac surgery. Heart. 2012;98(10):792–798.
- , , , et al. Derivation and prospective validation of a simple index for prediction of cardiac risk of major noncardiac surgery. Circulation. 1999;100(10):1043–1049.
- , , , , . Biologic variation of a novel cardiac troponin I assay. Clin Chem. 2011;57(7):1080–1081.
- , , , , . Short‐ and long‐term biological variation in cardiac troponin I measured with a high‐sensitivity assay: implications for clinical practice. Clin Chem. 2009;55(1):52–58.
- , , , et al. Relationship between perioperative troponin elevation and other indicators of myocardial injury in vascular surgery patients. Br J Anaesth. 2006;96(3):303–309.
- , , , et al. Which troponometric best predicts midterm outcome after coronary artery bypass graft surgery? Ann Thorac Surg. 2011;91(6):1860–1867.
Noncardiac surgery is frequently associated with major adverse cardiac events. Prevention of these events has traditionally focused on risk estimation and targeted interventions prior to surgical intervention.1 Cardiac risk is assessed through clinical prediction rules, such as the revised cardiac risk index (RCRI) or the American Society of Anesthesiology (ASA) classification. In patients deemed to be at high risk of adverse cardiac events, discretionary preoperative testing, medical treatments, and interventions are implemented.2 However, even when executed optimally, this approach fails to protect all patients. Thus, many patients undergoing noncardiac surgery continue to experience perioperative myocardial infarction (MI) and death.3
Recently, a prospective international study involving 15,133 patients reported that cardiac troponin levels measured within 3 days of noncardiac surgery were strongly associated with 30‐day mortality.4 This study is but the latest in a series of investigations that have concluded that postoperative measurement of cardiac troponin is a better predictor of cardiac outcomes than preoperative‐risk algorithms.48 Intuitively, this link is not surprising. Surgery represents the ultimate physiologic stress test. In the setting of hemodynamic changes and sustained oxygen supply:demand mismatch, it is hardly shocking that those with significant epicardial coronary stenosis or major subendocardial ischemia suffer poorer outcomes. What is perhaps most important about the association between troponin release and clinical outcomes, however, is the foundation it provides for a novel framework to improve perioperative care: using troponin measurement to guide treatment and interventions in the postoperative setting.
MODEL TO IMPROVE POSTOPERATIVE CARDIAC OUTCOMES
We hypothesize that the postoperative period can be seized as a golden moment to ameliorate cardiac risk and improve clinical outcomes for 3 reasons. First, those who release cardiac troponin during surgery declare themselves as harboring myocardial territories in jeopardy. In effect, the peripheral presence of troponin amounts to a cry for help in a population where many cardiac events are silent and thus remain clinically unnoticed.9, 10 Routine postoperative monitoring for this biomarker can thus help identify a population that would ordinarily escape detection until the precipitation of a major clinical event. Second, as the pathophysiology of cardiac events centers around rupture of vulnerable plaque (type‐1 MI), supply:demand mismatch (type‐2 MI), or both of these phenomena, escalation or institution of important medical risk‐reducing treatments (such as aspirin, beta‐blockers, or statins), can be established to improve outcomes. The ability to initiate these treatments in a monitored setting may offset and address many of the risks feared with these therapies, including hypotension, stroke, or rhabdomyolysis. Third, as patients convalesce from surgery, timely and discretionary cardiac testing or interventions can be undertaken in those identified as being at elevated cardiovascular risk prior to discharge. In sum, the trifecta of clearly recognizing those at risk, instituting interventions in controlled settings, and performing well‐timed interventions could translate into greater odds of survival in this cohort.
Owing to their intimate involvement with patients in the perioperative setting, hospitalists are uniquely suited for implementing a troponin surveillance paradigm. Yet, how may such a schema be realized? In Figure 1, we outline a pathway with which to implement this approach. This model replicates perioperative management workflow, calling for 3 discrete interventionspreoperative assessment, perioperative surveillance, and postoperative risk stratification:

-
Preoperative assessment: Patients estimated to be at high risk of perioperative cardiac events (eg, RCRI 3) should undergo baseline, preoperative high‐sensitivity troponin measurement, as the frequency of major adverse cardiac outcomes is significant this group (11%).11 The importance of a baseline measurement of troponin cannot be overstated, as biologic variation of this marker can impact subsequent management.12, 13 Hospitalists can obtain these assays in preoperative clinics at the time of initial review and risk‐estimation.
-
Perioperative surveillance: Following the model implemented by a recent study, serial troponin surveillance should commence 612 hours after surgery, then daily on the first, second, and third day.4 As the optimal cardiac troponin threshold for the diagnosis of perioperative ischemia remains uncertain, doubling of baseline troponin levels may serve as a logical signal for further evaluation.14 The electrocardiographic pattern may be used as a branch point for immediate management at this stage: ST‐segment elevation MI may be treated aggressively with an early invasive strategy and cardiac catheterization when apropos (much as in the nonoperative context). However, as most perioperative cardiac events are non‐ST elevation MIs, treatment of this subset should center on initiation or escalation of medical therapy. Thus, achieving heart‐rate control in order to preserve coronary perfusion by careful titration of beta‐blockers, and initiation/up‐titration of statins to temper unstable atherosclerotic plaques represent cornerstones of this approach. Other potential therapeutic entities such as initiation of angiotensin‐converting‐enzyme (ACE)‐inhibitors in the context of left‐ventricular dysfunction, and/or antiplatelet agents such as aspirin, may also be relevant risk‐reducing tools.
-
Postoperative risk stratification: Prior to hospital discharge, objective assessment of myocardial perfusion and viability should occur in selected individuals with troponin elevations. Modalities that may be utilized in this context include either noninvasive imaging or occasional coronary angiography in patients with unstable coronary syndromes. Defining an optimal approach must be contextual, as interventions that may subsequently mandate uninterrupted antiplatelet treatment may be logistically challenging in this setting.
LIMITATIONS
Although this implementation model is plausible, it generates many questions that must be tested through the lens of a randomized controlled study. For instance, what is the optimal strategy for intervention in the postoperative setting? Though medical treatment with statins and beta‐blockade may represent the mainstay of treatment, are these therapies safe during potential clinical instability? Can net benefit be realized through judicious use of coronary angiography and revascularization? Can the artifact of heightened awareness and reporting of postoperative cardiac events mar the reported quality of a hospital? Finally, though cardiac troponin has been shown to be a strong and independent predictor of mortality, which troponin assay, reference range, and troponometric standard to use remain unclear.15 Whether or not these interventions translate into lesser mortality and improved clinical outcomes represents the raison d'etre for this experimental approach. As no alternative approach to define and target patients at high risk of adverse outcome after seemingly uneventful surgery currently exists, we believe that this hypothetical paradigm is worthy of further investigation.
CONCLUSION
For decades, perioperative practitioners have searched for a divining rod to reduce risk during surgery. While our efforts have been focused on the preoperative setting, the postoperative setting represents an as yet untapped and potentially profound period in the quest to improving surgical outcomes. Through our proximity to patients in the perioperative setting, hospitalists are ideal agents to test, deliver, and bring about this change. It is time for a postoperative carpe diem.
Noncardiac surgery is frequently associated with major adverse cardiac events. Prevention of these events has traditionally focused on risk estimation and targeted interventions prior to surgical intervention.1 Cardiac risk is assessed through clinical prediction rules, such as the revised cardiac risk index (RCRI) or the American Society of Anesthesiology (ASA) classification. In patients deemed to be at high risk of adverse cardiac events, discretionary preoperative testing, medical treatments, and interventions are implemented.2 However, even when executed optimally, this approach fails to protect all patients. Thus, many patients undergoing noncardiac surgery continue to experience perioperative myocardial infarction (MI) and death.3
Recently, a prospective international study involving 15,133 patients reported that cardiac troponin levels measured within 3 days of noncardiac surgery were strongly associated with 30‐day mortality.4 This study is but the latest in a series of investigations that have concluded that postoperative measurement of cardiac troponin is a better predictor of cardiac outcomes than preoperative‐risk algorithms.48 Intuitively, this link is not surprising. Surgery represents the ultimate physiologic stress test. In the setting of hemodynamic changes and sustained oxygen supply:demand mismatch, it is hardly shocking that those with significant epicardial coronary stenosis or major subendocardial ischemia suffer poorer outcomes. What is perhaps most important about the association between troponin release and clinical outcomes, however, is the foundation it provides for a novel framework to improve perioperative care: using troponin measurement to guide treatment and interventions in the postoperative setting.
MODEL TO IMPROVE POSTOPERATIVE CARDIAC OUTCOMES
We hypothesize that the postoperative period can be seized as a golden moment to ameliorate cardiac risk and improve clinical outcomes for 3 reasons. First, those who release cardiac troponin during surgery declare themselves as harboring myocardial territories in jeopardy. In effect, the peripheral presence of troponin amounts to a cry for help in a population where many cardiac events are silent and thus remain clinically unnoticed.9, 10 Routine postoperative monitoring for this biomarker can thus help identify a population that would ordinarily escape detection until the precipitation of a major clinical event. Second, as the pathophysiology of cardiac events centers around rupture of vulnerable plaque (type‐1 MI), supply:demand mismatch (type‐2 MI), or both of these phenomena, escalation or institution of important medical risk‐reducing treatments (such as aspirin, beta‐blockers, or statins), can be established to improve outcomes. The ability to initiate these treatments in a monitored setting may offset and address many of the risks feared with these therapies, including hypotension, stroke, or rhabdomyolysis. Third, as patients convalesce from surgery, timely and discretionary cardiac testing or interventions can be undertaken in those identified as being at elevated cardiovascular risk prior to discharge. In sum, the trifecta of clearly recognizing those at risk, instituting interventions in controlled settings, and performing well‐timed interventions could translate into greater odds of survival in this cohort.
Owing to their intimate involvement with patients in the perioperative setting, hospitalists are uniquely suited for implementing a troponin surveillance paradigm. Yet, how may such a schema be realized? In Figure 1, we outline a pathway with which to implement this approach. This model replicates perioperative management workflow, calling for 3 discrete interventionspreoperative assessment, perioperative surveillance, and postoperative risk stratification:

-
Preoperative assessment: Patients estimated to be at high risk of perioperative cardiac events (eg, RCRI 3) should undergo baseline, preoperative high‐sensitivity troponin measurement, as the frequency of major adverse cardiac outcomes is significant this group (11%).11 The importance of a baseline measurement of troponin cannot be overstated, as biologic variation of this marker can impact subsequent management.12, 13 Hospitalists can obtain these assays in preoperative clinics at the time of initial review and risk‐estimation.
-
Perioperative surveillance: Following the model implemented by a recent study, serial troponin surveillance should commence 612 hours after surgery, then daily on the first, second, and third day.4 As the optimal cardiac troponin threshold for the diagnosis of perioperative ischemia remains uncertain, doubling of baseline troponin levels may serve as a logical signal for further evaluation.14 The electrocardiographic pattern may be used as a branch point for immediate management at this stage: ST‐segment elevation MI may be treated aggressively with an early invasive strategy and cardiac catheterization when apropos (much as in the nonoperative context). However, as most perioperative cardiac events are non‐ST elevation MIs, treatment of this subset should center on initiation or escalation of medical therapy. Thus, achieving heart‐rate control in order to preserve coronary perfusion by careful titration of beta‐blockers, and initiation/up‐titration of statins to temper unstable atherosclerotic plaques represent cornerstones of this approach. Other potential therapeutic entities such as initiation of angiotensin‐converting‐enzyme (ACE)‐inhibitors in the context of left‐ventricular dysfunction, and/or antiplatelet agents such as aspirin, may also be relevant risk‐reducing tools.
-
Postoperative risk stratification: Prior to hospital discharge, objective assessment of myocardial perfusion and viability should occur in selected individuals with troponin elevations. Modalities that may be utilized in this context include either noninvasive imaging or occasional coronary angiography in patients with unstable coronary syndromes. Defining an optimal approach must be contextual, as interventions that may subsequently mandate uninterrupted antiplatelet treatment may be logistically challenging in this setting.
LIMITATIONS
Although this implementation model is plausible, it generates many questions that must be tested through the lens of a randomized controlled study. For instance, what is the optimal strategy for intervention in the postoperative setting? Though medical treatment with statins and beta‐blockade may represent the mainstay of treatment, are these therapies safe during potential clinical instability? Can net benefit be realized through judicious use of coronary angiography and revascularization? Can the artifact of heightened awareness and reporting of postoperative cardiac events mar the reported quality of a hospital? Finally, though cardiac troponin has been shown to be a strong and independent predictor of mortality, which troponin assay, reference range, and troponometric standard to use remain unclear.15 Whether or not these interventions translate into lesser mortality and improved clinical outcomes represents the raison d'etre for this experimental approach. As no alternative approach to define and target patients at high risk of adverse outcome after seemingly uneventful surgery currently exists, we believe that this hypothetical paradigm is worthy of further investigation.
CONCLUSION
For decades, perioperative practitioners have searched for a divining rod to reduce risk during surgery. While our efforts have been focused on the preoperative setting, the postoperative setting represents an as yet untapped and potentially profound period in the quest to improving surgical outcomes. Through our proximity to patients in the perioperative setting, hospitalists are ideal agents to test, deliver, and bring about this change. It is time for a postoperative carpe diem.
- , , , et al. Predictors of cardiac events after major vascular surgery: role of clinical characteristics, dobutamine echocardiography, and beta‐blocker therapy. JAMA. 2001;285(14):1865–1873.
- , , , et al. Combining clinical and thallium data optimizes preoperative assessment of cardiac risk before major vascular surgery. Ann Intern Med. 1989;110(11):859–866.
- , , , et al. An estimation of the global volume of surgery: a modelling strategy based on available data. Lancet. 2008;372(9633):139–144.
- VISION Study Investigators. Association between postoperative troponin levels and 30‐day mortlality among patients undergoing noncardiac surgery. JAMA. 2012;307(21):2295–2304.
- , , , et al. Postoperative levels of cardiac troponin versus CK‐MB and high‐sensitivity C‐reactive protein for the prediction of 1‐year cardiovascular outcome in patients undergoing vascular surgery. Coron Artery Dis. 2011;22(6):428–434.
- , , , et al. Peak postoperative troponin levels outperform preoperative cardiac risk indices as predictors of long‐term mortality after vascular surgery Troponins and postoperative outcomes. J Crit Care. 2012;27(1):66–72.
- , , , et al. Prognostic value of troponin and creatine kinase muscle and brain isoenzyme measurement after noncardiac surgery: a systematic review and meta‐analysis. Anesthesiology. 2011;114(4):796–806.
- , , . Outcomes in vascular surgical patients with isolated postoperative troponin leak: a meta‐analysis. Anaesthesia. 2011;66(7):604–610.
- , , , et al. Characteristics and short‐term prognosis of perioperative myocardial infarction in patients undergoing noncardiac surgery: a cohort study. Ann Intern Med. 2011;154(8):523–528.
- , , , , . Perioperative myocardial necrosis in patients at high cardiovascular risk undergoing elective non‐cardiac surgery. Heart. 2012;98(10):792–798.
- , , , et al. Derivation and prospective validation of a simple index for prediction of cardiac risk of major noncardiac surgery. Circulation. 1999;100(10):1043–1049.
- , , , , . Biologic variation of a novel cardiac troponin I assay. Clin Chem. 2011;57(7):1080–1081.
- , , , , . Short‐ and long‐term biological variation in cardiac troponin I measured with a high‐sensitivity assay: implications for clinical practice. Clin Chem. 2009;55(1):52–58.
- , , , et al. Relationship between perioperative troponin elevation and other indicators of myocardial injury in vascular surgery patients. Br J Anaesth. 2006;96(3):303–309.
- , , , et al. Which troponometric best predicts midterm outcome after coronary artery bypass graft surgery? Ann Thorac Surg. 2011;91(6):1860–1867.
- , , , et al. Predictors of cardiac events after major vascular surgery: role of clinical characteristics, dobutamine echocardiography, and beta‐blocker therapy. JAMA. 2001;285(14):1865–1873.
- , , , et al. Combining clinical and thallium data optimizes preoperative assessment of cardiac risk before major vascular surgery. Ann Intern Med. 1989;110(11):859–866.
- , , , et al. An estimation of the global volume of surgery: a modelling strategy based on available data. Lancet. 2008;372(9633):139–144.
- VISION Study Investigators. Association between postoperative troponin levels and 30‐day mortlality among patients undergoing noncardiac surgery. JAMA. 2012;307(21):2295–2304.
- , , , et al. Postoperative levels of cardiac troponin versus CK‐MB and high‐sensitivity C‐reactive protein for the prediction of 1‐year cardiovascular outcome in patients undergoing vascular surgery. Coron Artery Dis. 2011;22(6):428–434.
- , , , et al. Peak postoperative troponin levels outperform preoperative cardiac risk indices as predictors of long‐term mortality after vascular surgery Troponins and postoperative outcomes. J Crit Care. 2012;27(1):66–72.
- , , , et al. Prognostic value of troponin and creatine kinase muscle and brain isoenzyme measurement after noncardiac surgery: a systematic review and meta‐analysis. Anesthesiology. 2011;114(4):796–806.
- , , . Outcomes in vascular surgical patients with isolated postoperative troponin leak: a meta‐analysis. Anaesthesia. 2011;66(7):604–610.
- , , , et al. Characteristics and short‐term prognosis of perioperative myocardial infarction in patients undergoing noncardiac surgery: a cohort study. Ann Intern Med. 2011;154(8):523–528.
- , , , , . Perioperative myocardial necrosis in patients at high cardiovascular risk undergoing elective non‐cardiac surgery. Heart. 2012;98(10):792–798.
- , , , et al. Derivation and prospective validation of a simple index for prediction of cardiac risk of major noncardiac surgery. Circulation. 1999;100(10):1043–1049.
- , , , , . Biologic variation of a novel cardiac troponin I assay. Clin Chem. 2011;57(7):1080–1081.
- , , , , . Short‐ and long‐term biological variation in cardiac troponin I measured with a high‐sensitivity assay: implications for clinical practice. Clin Chem. 2009;55(1):52–58.
- , , , et al. Relationship between perioperative troponin elevation and other indicators of myocardial injury in vascular surgery patients. Br J Anaesth. 2006;96(3):303–309.
- , , , et al. Which troponometric best predicts midterm outcome after coronary artery bypass graft surgery? Ann Thorac Surg. 2011;91(6):1860–1867.