User login
Study confirms it’s possible to catch COVID-19 twice
Researchers in Hong Kong say they’ve confirmed that a person can be infected with COVID-19 twice.
The new proof comes from a 33-year-old man in Hong Kong who first caught COVID-19 in March. He was tested for the coronavirus after he developed a cough, sore throat, fever, and a headache for 3 days. He stayed in the hospital until he twice tested negative for the virus in mid-April.
On Aug. 15, the man returned to Hong Kong from a recent trip to Spain and the United Kingdom, areas that have recently seen a resurgence of COVID-19 cases. At the airport, he was screened for COVID-19 with a test that checks saliva for the virus. He tested positive, but this time, had no symptoms. He was taken to the hospital for monitoring. His viral load – the amount of virus he had in his body – went down over time, suggesting that his immune system was taking care of the intrusion on its own.
The special thing about his case is that each time he was hospitalized, doctors sequenced the genome of the virus that infected him. It was slightly different from one infection to the next, suggesting that the virus had mutated – or changed – in the 4 months between his infections. It also proves that it’s possible for this coronavirus to infect the same person twice.
Experts with the World Health Organization responded to the case at a news briefing.
“What we are learning about infection is that people do develop an immune response. What is not completely clear yet is how strong that immune response is and for how long that immune response lasts,” said Maria Van Kerkhove, PhD, an infectious disease epidemiologist with the World Health Organization in Geneva, Switzerland.
A study on the man’s case is being prepared for publication in the journal Clinical Infectious Diseases. Experts say the finding shouldn’t cause alarm, but it does have important implications for the development of herd immunity and efforts to come up with vaccines and treatments.
“This appears to be pretty clear-cut evidence of reinfection because of sequencing and isolation of two different viruses,” said Gregory Poland, MD, an expert on vaccine development and immunology at the Mayo Clinic in Rochester, Minn. “The big unknown is how often is this happening,” he said. More studies are needed to learn whether this was a rare case or something that is happening often.
Past experience guides present
Until we know more, Dr. Poland said, the possibility of getting COVID-19 twice shouldn’t make anyone worry.
This also happens with other kinds of coronaviruses – the ones that cause common colds. Those coronaviruses change slightly each year as they circle the globe, which allows them to keep spreading and causing their more run-of-the-mill kind of misery.
It also happens with seasonal flu. It is the reason people have to get vaccinated against the flu year after year, and why the flu vaccine has to change slightly each year in an effort to keep up with the ever-evolving influenza virus.
“We’ve been making flu vaccines for 80 years, and there are clinical trials happening as we speak to find new and better influenza vaccines,” Dr. Poland said.
There has been other evidence the virus that causes COVID-19 can change this way, too. Researchers at Howard Hughes Medical Center, at Rockefeller University in New York, recently used a key piece of the SARS-CoV-2 virus – the genetic instructions for its spike protein – to repeatedly infect human cells. Scientists watched as each new generation of the virus went on to infect a new batch of cells. Over time, as it copied itself, some of the copies changed their genes to allow them to survive after scientists attacked them with neutralizing antibodies. Those antibodies are among the main weapons used by the immune system to recognize and disable a virus.
Though that study is still a preprint, which means it hasn’t yet been reviewed by outside experts, the authors wrote that their findings suggest the virus can change in ways that help it evade our immune system. If true, they wrote in mid-July, it means reinfection is possible, especially in people who have a weak immune response to the virus the first time they encounter it.
Good news
That seems to be true in the case of the man from Hong Kong. When doctors tested his blood to look for antibodies to the virus, they didn’t find any. That could mean that he either had a weak immune response to the virus the first time around, or that the antibodies he made during his first infection diminished over time. But during his second infection, he quickly developed more antibodies, suggesting that the second infection acted a little bit like a booster to fire up his immune system. That’s probably the reason he didn’t have any symptoms the second time, too.
That’s good news, Dr. Poland said. It means our bodies can get better at fighting off the COVID-19 virus and that catching it once means the second time might not be so bad.
But the fact that the virus can change quickly this way does have some impact on the effort to come up with a vaccine that works well.
“I think a potential implication of this is that we will have to give booster doses. The question is how frequently,” Dr. Poland said. That will depend on how fast the virus is changing, and how often reinfection is happening in the real world.
“I’m a little surprised at 4½ months,” Dr. Poland said, referencing the time between the Hong Kong man’s infections. “I’m not surprised by, you know, I got infected last winter and I got infected again this winter,” he said.
It also suggests that immune-based therapies such as convalescent plasma and monoclonal antibodies may be of limited help over time, since the virus might be changing in ways that help it outsmart those treatments.
Convalescent plasma is essentially a concentrated dose of antibodies from people who have recovered from a COVID-19 infection. As the virus changes, the antibodies in that plasma may not work as well for future infections.
Drug companies have learned to harness the power of monoclonal antibodies as powerful treatments against cancer and other diseases. Monoclonal antibodies, which are mass-produced in a lab, mimic the body’s natural defenses against a pathogen. Just like the virus can become resistant to natural immunity, it can change in ways that help it outsmart lab-created treatments. Some drug companies that are developing monoclonal antibodies to fight COVID-19 have already prepared for that possibility by making antibody cocktails that are designed to disable the virus by locking onto it in different places, which may help prevent it from developing resistance to those therapies.
“We have a lot to learn,” Dr. Poland said. “Now that the proof of principle has been established, and I would say it has with this man, and with our knowledge of seasonal coronaviruses, we need to look more aggressively to define how often this occurs.”
A version of this article originally appeared on WebMD.com.
Researchers in Hong Kong say they’ve confirmed that a person can be infected with COVID-19 twice.
The new proof comes from a 33-year-old man in Hong Kong who first caught COVID-19 in March. He was tested for the coronavirus after he developed a cough, sore throat, fever, and a headache for 3 days. He stayed in the hospital until he twice tested negative for the virus in mid-April.
On Aug. 15, the man returned to Hong Kong from a recent trip to Spain and the United Kingdom, areas that have recently seen a resurgence of COVID-19 cases. At the airport, he was screened for COVID-19 with a test that checks saliva for the virus. He tested positive, but this time, had no symptoms. He was taken to the hospital for monitoring. His viral load – the amount of virus he had in his body – went down over time, suggesting that his immune system was taking care of the intrusion on its own.
The special thing about his case is that each time he was hospitalized, doctors sequenced the genome of the virus that infected him. It was slightly different from one infection to the next, suggesting that the virus had mutated – or changed – in the 4 months between his infections. It also proves that it’s possible for this coronavirus to infect the same person twice.
Experts with the World Health Organization responded to the case at a news briefing.
“What we are learning about infection is that people do develop an immune response. What is not completely clear yet is how strong that immune response is and for how long that immune response lasts,” said Maria Van Kerkhove, PhD, an infectious disease epidemiologist with the World Health Organization in Geneva, Switzerland.
A study on the man’s case is being prepared for publication in the journal Clinical Infectious Diseases. Experts say the finding shouldn’t cause alarm, but it does have important implications for the development of herd immunity and efforts to come up with vaccines and treatments.
“This appears to be pretty clear-cut evidence of reinfection because of sequencing and isolation of two different viruses,” said Gregory Poland, MD, an expert on vaccine development and immunology at the Mayo Clinic in Rochester, Minn. “The big unknown is how often is this happening,” he said. More studies are needed to learn whether this was a rare case or something that is happening often.
Past experience guides present
Until we know more, Dr. Poland said, the possibility of getting COVID-19 twice shouldn’t make anyone worry.
This also happens with other kinds of coronaviruses – the ones that cause common colds. Those coronaviruses change slightly each year as they circle the globe, which allows them to keep spreading and causing their more run-of-the-mill kind of misery.
It also happens with seasonal flu. It is the reason people have to get vaccinated against the flu year after year, and why the flu vaccine has to change slightly each year in an effort to keep up with the ever-evolving influenza virus.
“We’ve been making flu vaccines for 80 years, and there are clinical trials happening as we speak to find new and better influenza vaccines,” Dr. Poland said.
There has been other evidence the virus that causes COVID-19 can change this way, too. Researchers at Howard Hughes Medical Center, at Rockefeller University in New York, recently used a key piece of the SARS-CoV-2 virus – the genetic instructions for its spike protein – to repeatedly infect human cells. Scientists watched as each new generation of the virus went on to infect a new batch of cells. Over time, as it copied itself, some of the copies changed their genes to allow them to survive after scientists attacked them with neutralizing antibodies. Those antibodies are among the main weapons used by the immune system to recognize and disable a virus.
Though that study is still a preprint, which means it hasn’t yet been reviewed by outside experts, the authors wrote that their findings suggest the virus can change in ways that help it evade our immune system. If true, they wrote in mid-July, it means reinfection is possible, especially in people who have a weak immune response to the virus the first time they encounter it.
Good news
That seems to be true in the case of the man from Hong Kong. When doctors tested his blood to look for antibodies to the virus, they didn’t find any. That could mean that he either had a weak immune response to the virus the first time around, or that the antibodies he made during his first infection diminished over time. But during his second infection, he quickly developed more antibodies, suggesting that the second infection acted a little bit like a booster to fire up his immune system. That’s probably the reason he didn’t have any symptoms the second time, too.
That’s good news, Dr. Poland said. It means our bodies can get better at fighting off the COVID-19 virus and that catching it once means the second time might not be so bad.
But the fact that the virus can change quickly this way does have some impact on the effort to come up with a vaccine that works well.
“I think a potential implication of this is that we will have to give booster doses. The question is how frequently,” Dr. Poland said. That will depend on how fast the virus is changing, and how often reinfection is happening in the real world.
“I’m a little surprised at 4½ months,” Dr. Poland said, referencing the time between the Hong Kong man’s infections. “I’m not surprised by, you know, I got infected last winter and I got infected again this winter,” he said.
It also suggests that immune-based therapies such as convalescent plasma and monoclonal antibodies may be of limited help over time, since the virus might be changing in ways that help it outsmart those treatments.
Convalescent plasma is essentially a concentrated dose of antibodies from people who have recovered from a COVID-19 infection. As the virus changes, the antibodies in that plasma may not work as well for future infections.
Drug companies have learned to harness the power of monoclonal antibodies as powerful treatments against cancer and other diseases. Monoclonal antibodies, which are mass-produced in a lab, mimic the body’s natural defenses against a pathogen. Just like the virus can become resistant to natural immunity, it can change in ways that help it outsmart lab-created treatments. Some drug companies that are developing monoclonal antibodies to fight COVID-19 have already prepared for that possibility by making antibody cocktails that are designed to disable the virus by locking onto it in different places, which may help prevent it from developing resistance to those therapies.
“We have a lot to learn,” Dr. Poland said. “Now that the proof of principle has been established, and I would say it has with this man, and with our knowledge of seasonal coronaviruses, we need to look more aggressively to define how often this occurs.”
A version of this article originally appeared on WebMD.com.
Researchers in Hong Kong say they’ve confirmed that a person can be infected with COVID-19 twice.
The new proof comes from a 33-year-old man in Hong Kong who first caught COVID-19 in March. He was tested for the coronavirus after he developed a cough, sore throat, fever, and a headache for 3 days. He stayed in the hospital until he twice tested negative for the virus in mid-April.
On Aug. 15, the man returned to Hong Kong from a recent trip to Spain and the United Kingdom, areas that have recently seen a resurgence of COVID-19 cases. At the airport, he was screened for COVID-19 with a test that checks saliva for the virus. He tested positive, but this time, had no symptoms. He was taken to the hospital for monitoring. His viral load – the amount of virus he had in his body – went down over time, suggesting that his immune system was taking care of the intrusion on its own.
The special thing about his case is that each time he was hospitalized, doctors sequenced the genome of the virus that infected him. It was slightly different from one infection to the next, suggesting that the virus had mutated – or changed – in the 4 months between his infections. It also proves that it’s possible for this coronavirus to infect the same person twice.
Experts with the World Health Organization responded to the case at a news briefing.
“What we are learning about infection is that people do develop an immune response. What is not completely clear yet is how strong that immune response is and for how long that immune response lasts,” said Maria Van Kerkhove, PhD, an infectious disease epidemiologist with the World Health Organization in Geneva, Switzerland.
A study on the man’s case is being prepared for publication in the journal Clinical Infectious Diseases. Experts say the finding shouldn’t cause alarm, but it does have important implications for the development of herd immunity and efforts to come up with vaccines and treatments.
“This appears to be pretty clear-cut evidence of reinfection because of sequencing and isolation of two different viruses,” said Gregory Poland, MD, an expert on vaccine development and immunology at the Mayo Clinic in Rochester, Minn. “The big unknown is how often is this happening,” he said. More studies are needed to learn whether this was a rare case or something that is happening often.
Past experience guides present
Until we know more, Dr. Poland said, the possibility of getting COVID-19 twice shouldn’t make anyone worry.
This also happens with other kinds of coronaviruses – the ones that cause common colds. Those coronaviruses change slightly each year as they circle the globe, which allows them to keep spreading and causing their more run-of-the-mill kind of misery.
It also happens with seasonal flu. It is the reason people have to get vaccinated against the flu year after year, and why the flu vaccine has to change slightly each year in an effort to keep up with the ever-evolving influenza virus.
“We’ve been making flu vaccines for 80 years, and there are clinical trials happening as we speak to find new and better influenza vaccines,” Dr. Poland said.
There has been other evidence the virus that causes COVID-19 can change this way, too. Researchers at Howard Hughes Medical Center, at Rockefeller University in New York, recently used a key piece of the SARS-CoV-2 virus – the genetic instructions for its spike protein – to repeatedly infect human cells. Scientists watched as each new generation of the virus went on to infect a new batch of cells. Over time, as it copied itself, some of the copies changed their genes to allow them to survive after scientists attacked them with neutralizing antibodies. Those antibodies are among the main weapons used by the immune system to recognize and disable a virus.
Though that study is still a preprint, which means it hasn’t yet been reviewed by outside experts, the authors wrote that their findings suggest the virus can change in ways that help it evade our immune system. If true, they wrote in mid-July, it means reinfection is possible, especially in people who have a weak immune response to the virus the first time they encounter it.
Good news
That seems to be true in the case of the man from Hong Kong. When doctors tested his blood to look for antibodies to the virus, they didn’t find any. That could mean that he either had a weak immune response to the virus the first time around, or that the antibodies he made during his first infection diminished over time. But during his second infection, he quickly developed more antibodies, suggesting that the second infection acted a little bit like a booster to fire up his immune system. That’s probably the reason he didn’t have any symptoms the second time, too.
That’s good news, Dr. Poland said. It means our bodies can get better at fighting off the COVID-19 virus and that catching it once means the second time might not be so bad.
But the fact that the virus can change quickly this way does have some impact on the effort to come up with a vaccine that works well.
“I think a potential implication of this is that we will have to give booster doses. The question is how frequently,” Dr. Poland said. That will depend on how fast the virus is changing, and how often reinfection is happening in the real world.
“I’m a little surprised at 4½ months,” Dr. Poland said, referencing the time between the Hong Kong man’s infections. “I’m not surprised by, you know, I got infected last winter and I got infected again this winter,” he said.
It also suggests that immune-based therapies such as convalescent plasma and monoclonal antibodies may be of limited help over time, since the virus might be changing in ways that help it outsmart those treatments.
Convalescent plasma is essentially a concentrated dose of antibodies from people who have recovered from a COVID-19 infection. As the virus changes, the antibodies in that plasma may not work as well for future infections.
Drug companies have learned to harness the power of monoclonal antibodies as powerful treatments against cancer and other diseases. Monoclonal antibodies, which are mass-produced in a lab, mimic the body’s natural defenses against a pathogen. Just like the virus can become resistant to natural immunity, it can change in ways that help it outsmart lab-created treatments. Some drug companies that are developing monoclonal antibodies to fight COVID-19 have already prepared for that possibility by making antibody cocktails that are designed to disable the virus by locking onto it in different places, which may help prevent it from developing resistance to those therapies.
“We have a lot to learn,” Dr. Poland said. “Now that the proof of principle has been established, and I would say it has with this man, and with our knowledge of seasonal coronaviruses, we need to look more aggressively to define how often this occurs.”
A version of this article originally appeared on WebMD.com.
Research examines links between ‘long COVID’ and ME/CFS
Some patients who had COVID-19 continue to have symptoms weeks to months later, even after they no longer test positive for the virus. In two recent reports – one published in JAMA in July and another published in Morbidity and Mortality Weekly Report in August – chronic fatigue was listed as the top symptom among individuals still feeling unwell beyond 2 weeks after COVID-19 onset.
Although some of the reported persistent symptoms appear specific to SARS-CoV-2 – such as cough, chest pain, and dyspnea – others overlap with the diagnostic criteria for ME/CFS, which is defined by substantial, profound fatigue for at least 6 months, postexertional malaise, unrefreshing sleep, and one or both of orthostatic intolerance and/or cognitive impairment. Although the etiology of ME/CFS is unclear, the condition commonly arises following a viral illness.
At the virtual meeting of the International Association for Chronic Fatigue Syndrome/Myalgic Encephalomyelitis August 21, the opening session was devoted to research documenting the extent to which COVID-19 survivors subsequently meet ME/CFS criteria, and to exploring underlying mechanisms.
“It offers a lot of opportunities for us to study potentially early ME/CFS and how it develops, but in addition, a lot of the research that has been done on ME/CFS may also provide answers for COVID-19,” IACFS/ME vice president Lily Chu, MD, said in an interview.
A hint from the SARS outbreak
This isn’t the first time researchers have seen a possible link between a coronavirus and ME/CFS, Harvey Moldofsky, MD, told attendees. To illustrate that point, Dr. Moldofsky, of the department of psychiatry (emeritus) at the University of Toronto, reviewed data from a previously published case-controlled study, which included 22 health care workers who had been infected in 2003 with SARS-CoV-1 and continued to report chronic fatigue, musculoskeletal pain, and disturbed and unrefreshing sleep with EEG-documented sleep disturbances 1-3 years following the illness. None had been able to return to work by 1 year.
“We’re looking at similar symptoms now” among survivors of COVID-19, Dr. Moldofsky said. “[T]he key issue is that we have no idea of its prevalence. … We need epidemiologic studies.”
Distinguishing ME/CFS from other post–COVID-19 symptoms
Not everyone who has persistent symptoms after COVID-19 will develop ME/CFS, and distinguishing between cases may be important.
Clinically, Dr. Chu said, one way to assess whether a patient with persistent COVID-19 symptoms might be progressing to ME/CFS is to ask him or her specifically about the level of fatigue following physical exertion and the timing of any fatigue. With ME/CFS, postexertional malaise often involves a dramatic exacerbation of symptoms such as fatigue, pain, and cognitive impairment a day or 2 after exertion rather than immediately following it. In contrast, shortness of breath during exertion isn’t typical of ME/CFS.
Objective measures of ME/CFS include low natural killer cell function (the test can be ordered from commercial labs but requires rapid transport of the blood sample), and autonomic dysfunction assessed by a tilt-table test.
While there is currently no cure for ME/CFS, diagnosing it allows for the patient to be taught “pacing” in which the person conserves his or her energy by balancing activity with rest. “That type of behavioral technique is valuable for everyone who suffers from a chronic disease with fatigue. It can help them be more functional,” Dr. Chu said.
If a patient appears to be exhibiting signs of ME/CFS, “don’t wait until they hit the 6-month mark to start helping them manage their symptoms,” she said. “Teaching pacing to COVID-19 patients who have a lot of fatigue isn’t going to harm them. As they get better they’re going to just naturally do more. But if they do have ME/CFS, [pacing] stresses their system less, since the data seem to be pointing to deficiencies in producing energy.”
Will COVID-19 unleash a new wave of ME/CFS patients?
Much of the session at the virtual meeting was devoted to ongoing studies. For example, Leonard Jason, PhD, of the Center for Community Research at DePaul University, Chicago, described a prospective study launched in 2014 that looked at risk factors for developing ME/CFS in college students who contracted infectious mononucleosis as a result of Epstein-Barr virus. Now, his team is also following students from the same cohort who develop COVID-19.
Because the study included collection of baseline biological samples, the results could help reveal predisposing factors associated with long-term illness from either virus.
Another project, funded by the Open Medicine Foundation, will follow patients who are discharged from the ICU following severe COVID-19 illness. Blood, urine, and cerebrospinal fluid will be collected from those with persistent symptoms at 6 months, along with questionnaire data. At 18-24 months, those who continue to report symptoms will undergo more intensive evaluation using genomics, metabolomics, and proteomics.
“We’re taking advantage of this horrible situation, hoping to understand how a serious viral infection might lead to ME/CFS,” said lead investigator Ronald Tompkins, MD, ScD, chief medical officer at the Open Medicine Foundation and a faculty member at Harvard Medical School, Boston. The results, he said, “might give us insight into potential drug targets or biomarkers useful for prevention and treatment strategies.”
Meanwhile, Sadie Whittaker, PhD, head of the Solve ME/CFS initiative, described her organization’s new plan to use their registry to prospectively track the impact of COVID-19 on people with ME/CFS.
She noted that they’ve also teamed up with “long-COVID” communities including Body Politic. “Our goal is to form a coalition to study together or at least harmonize data … and understand what’s going on through the power of bigger sample sizes,” Dr. Whittaker said.
None of the speakers disclosed relevant financial relationships.
A version of this article originally appeared on Medscape.com.
Some patients who had COVID-19 continue to have symptoms weeks to months later, even after they no longer test positive for the virus. In two recent reports – one published in JAMA in July and another published in Morbidity and Mortality Weekly Report in August – chronic fatigue was listed as the top symptom among individuals still feeling unwell beyond 2 weeks after COVID-19 onset.
Although some of the reported persistent symptoms appear specific to SARS-CoV-2 – such as cough, chest pain, and dyspnea – others overlap with the diagnostic criteria for ME/CFS, which is defined by substantial, profound fatigue for at least 6 months, postexertional malaise, unrefreshing sleep, and one or both of orthostatic intolerance and/or cognitive impairment. Although the etiology of ME/CFS is unclear, the condition commonly arises following a viral illness.
At the virtual meeting of the International Association for Chronic Fatigue Syndrome/Myalgic Encephalomyelitis August 21, the opening session was devoted to research documenting the extent to which COVID-19 survivors subsequently meet ME/CFS criteria, and to exploring underlying mechanisms.
“It offers a lot of opportunities for us to study potentially early ME/CFS and how it develops, but in addition, a lot of the research that has been done on ME/CFS may also provide answers for COVID-19,” IACFS/ME vice president Lily Chu, MD, said in an interview.
A hint from the SARS outbreak
This isn’t the first time researchers have seen a possible link between a coronavirus and ME/CFS, Harvey Moldofsky, MD, told attendees. To illustrate that point, Dr. Moldofsky, of the department of psychiatry (emeritus) at the University of Toronto, reviewed data from a previously published case-controlled study, which included 22 health care workers who had been infected in 2003 with SARS-CoV-1 and continued to report chronic fatigue, musculoskeletal pain, and disturbed and unrefreshing sleep with EEG-documented sleep disturbances 1-3 years following the illness. None had been able to return to work by 1 year.
“We’re looking at similar symptoms now” among survivors of COVID-19, Dr. Moldofsky said. “[T]he key issue is that we have no idea of its prevalence. … We need epidemiologic studies.”
Distinguishing ME/CFS from other post–COVID-19 symptoms
Not everyone who has persistent symptoms after COVID-19 will develop ME/CFS, and distinguishing between cases may be important.
Clinically, Dr. Chu said, one way to assess whether a patient with persistent COVID-19 symptoms might be progressing to ME/CFS is to ask him or her specifically about the level of fatigue following physical exertion and the timing of any fatigue. With ME/CFS, postexertional malaise often involves a dramatic exacerbation of symptoms such as fatigue, pain, and cognitive impairment a day or 2 after exertion rather than immediately following it. In contrast, shortness of breath during exertion isn’t typical of ME/CFS.
Objective measures of ME/CFS include low natural killer cell function (the test can be ordered from commercial labs but requires rapid transport of the blood sample), and autonomic dysfunction assessed by a tilt-table test.
While there is currently no cure for ME/CFS, diagnosing it allows for the patient to be taught “pacing” in which the person conserves his or her energy by balancing activity with rest. “That type of behavioral technique is valuable for everyone who suffers from a chronic disease with fatigue. It can help them be more functional,” Dr. Chu said.
If a patient appears to be exhibiting signs of ME/CFS, “don’t wait until they hit the 6-month mark to start helping them manage their symptoms,” she said. “Teaching pacing to COVID-19 patients who have a lot of fatigue isn’t going to harm them. As they get better they’re going to just naturally do more. But if they do have ME/CFS, [pacing] stresses their system less, since the data seem to be pointing to deficiencies in producing energy.”
Will COVID-19 unleash a new wave of ME/CFS patients?
Much of the session at the virtual meeting was devoted to ongoing studies. For example, Leonard Jason, PhD, of the Center for Community Research at DePaul University, Chicago, described a prospective study launched in 2014 that looked at risk factors for developing ME/CFS in college students who contracted infectious mononucleosis as a result of Epstein-Barr virus. Now, his team is also following students from the same cohort who develop COVID-19.
Because the study included collection of baseline biological samples, the results could help reveal predisposing factors associated with long-term illness from either virus.
Another project, funded by the Open Medicine Foundation, will follow patients who are discharged from the ICU following severe COVID-19 illness. Blood, urine, and cerebrospinal fluid will be collected from those with persistent symptoms at 6 months, along with questionnaire data. At 18-24 months, those who continue to report symptoms will undergo more intensive evaluation using genomics, metabolomics, and proteomics.
“We’re taking advantage of this horrible situation, hoping to understand how a serious viral infection might lead to ME/CFS,” said lead investigator Ronald Tompkins, MD, ScD, chief medical officer at the Open Medicine Foundation and a faculty member at Harvard Medical School, Boston. The results, he said, “might give us insight into potential drug targets or biomarkers useful for prevention and treatment strategies.”
Meanwhile, Sadie Whittaker, PhD, head of the Solve ME/CFS initiative, described her organization’s new plan to use their registry to prospectively track the impact of COVID-19 on people with ME/CFS.
She noted that they’ve also teamed up with “long-COVID” communities including Body Politic. “Our goal is to form a coalition to study together or at least harmonize data … and understand what’s going on through the power of bigger sample sizes,” Dr. Whittaker said.
None of the speakers disclosed relevant financial relationships.
A version of this article originally appeared on Medscape.com.
Some patients who had COVID-19 continue to have symptoms weeks to months later, even after they no longer test positive for the virus. In two recent reports – one published in JAMA in July and another published in Morbidity and Mortality Weekly Report in August – chronic fatigue was listed as the top symptom among individuals still feeling unwell beyond 2 weeks after COVID-19 onset.
Although some of the reported persistent symptoms appear specific to SARS-CoV-2 – such as cough, chest pain, and dyspnea – others overlap with the diagnostic criteria for ME/CFS, which is defined by substantial, profound fatigue for at least 6 months, postexertional malaise, unrefreshing sleep, and one or both of orthostatic intolerance and/or cognitive impairment. Although the etiology of ME/CFS is unclear, the condition commonly arises following a viral illness.
At the virtual meeting of the International Association for Chronic Fatigue Syndrome/Myalgic Encephalomyelitis August 21, the opening session was devoted to research documenting the extent to which COVID-19 survivors subsequently meet ME/CFS criteria, and to exploring underlying mechanisms.
“It offers a lot of opportunities for us to study potentially early ME/CFS and how it develops, but in addition, a lot of the research that has been done on ME/CFS may also provide answers for COVID-19,” IACFS/ME vice president Lily Chu, MD, said in an interview.
A hint from the SARS outbreak
This isn’t the first time researchers have seen a possible link between a coronavirus and ME/CFS, Harvey Moldofsky, MD, told attendees. To illustrate that point, Dr. Moldofsky, of the department of psychiatry (emeritus) at the University of Toronto, reviewed data from a previously published case-controlled study, which included 22 health care workers who had been infected in 2003 with SARS-CoV-1 and continued to report chronic fatigue, musculoskeletal pain, and disturbed and unrefreshing sleep with EEG-documented sleep disturbances 1-3 years following the illness. None had been able to return to work by 1 year.
“We’re looking at similar symptoms now” among survivors of COVID-19, Dr. Moldofsky said. “[T]he key issue is that we have no idea of its prevalence. … We need epidemiologic studies.”
Distinguishing ME/CFS from other post–COVID-19 symptoms
Not everyone who has persistent symptoms after COVID-19 will develop ME/CFS, and distinguishing between cases may be important.
Clinically, Dr. Chu said, one way to assess whether a patient with persistent COVID-19 symptoms might be progressing to ME/CFS is to ask him or her specifically about the level of fatigue following physical exertion and the timing of any fatigue. With ME/CFS, postexertional malaise often involves a dramatic exacerbation of symptoms such as fatigue, pain, and cognitive impairment a day or 2 after exertion rather than immediately following it. In contrast, shortness of breath during exertion isn’t typical of ME/CFS.
Objective measures of ME/CFS include low natural killer cell function (the test can be ordered from commercial labs but requires rapid transport of the blood sample), and autonomic dysfunction assessed by a tilt-table test.
While there is currently no cure for ME/CFS, diagnosing it allows for the patient to be taught “pacing” in which the person conserves his or her energy by balancing activity with rest. “That type of behavioral technique is valuable for everyone who suffers from a chronic disease with fatigue. It can help them be more functional,” Dr. Chu said.
If a patient appears to be exhibiting signs of ME/CFS, “don’t wait until they hit the 6-month mark to start helping them manage their symptoms,” she said. “Teaching pacing to COVID-19 patients who have a lot of fatigue isn’t going to harm them. As they get better they’re going to just naturally do more. But if they do have ME/CFS, [pacing] stresses their system less, since the data seem to be pointing to deficiencies in producing energy.”
Will COVID-19 unleash a new wave of ME/CFS patients?
Much of the session at the virtual meeting was devoted to ongoing studies. For example, Leonard Jason, PhD, of the Center for Community Research at DePaul University, Chicago, described a prospective study launched in 2014 that looked at risk factors for developing ME/CFS in college students who contracted infectious mononucleosis as a result of Epstein-Barr virus. Now, his team is also following students from the same cohort who develop COVID-19.
Because the study included collection of baseline biological samples, the results could help reveal predisposing factors associated with long-term illness from either virus.
Another project, funded by the Open Medicine Foundation, will follow patients who are discharged from the ICU following severe COVID-19 illness. Blood, urine, and cerebrospinal fluid will be collected from those with persistent symptoms at 6 months, along with questionnaire data. At 18-24 months, those who continue to report symptoms will undergo more intensive evaluation using genomics, metabolomics, and proteomics.
“We’re taking advantage of this horrible situation, hoping to understand how a serious viral infection might lead to ME/CFS,” said lead investigator Ronald Tompkins, MD, ScD, chief medical officer at the Open Medicine Foundation and a faculty member at Harvard Medical School, Boston. The results, he said, “might give us insight into potential drug targets or biomarkers useful for prevention and treatment strategies.”
Meanwhile, Sadie Whittaker, PhD, head of the Solve ME/CFS initiative, described her organization’s new plan to use their registry to prospectively track the impact of COVID-19 on people with ME/CFS.
She noted that they’ve also teamed up with “long-COVID” communities including Body Politic. “Our goal is to form a coalition to study together or at least harmonize data … and understand what’s going on through the power of bigger sample sizes,” Dr. Whittaker said.
None of the speakers disclosed relevant financial relationships.
A version of this article originally appeared on Medscape.com.
FDA authorizes convalescent plasma for COVID-19
Convalescent plasma contains antibodies from the blood of recovered COVID-19 patients, which can be used to treat people with severe infections. Convalescent plasma has been used to treat patients for other infectious diseases. The authorization allows the plasma to be distributed in the United States and administered by health care providers.
“COVID-19 convalescent plasma is safe and shows promising efficacy,” Stephen Hahn, MD, commissioner of the FDA, said during a press briefing with President Donald Trump.
In April, the FDA approved a program to test convalescent plasma in COVID-19 patients at the Mayo Clinic, followed by other institutions. More than 90,000 patients have enrolled in the program, and 70,000 have received the treatment, Dr. Hahn said.
The data indicate that the plasma can reduce mortality in patients by 35%, particularly if patients are treated within 3 days of being diagnosed. Those who have benefited the most were under age 80 and not on artificial respiration, Alex Azar, the secretary for the Department of Health & Human Services, said during the briefing.
“We dream, in drug development, of something like a 35% mortality reduction,” he said.
But top scientists pushed back against the announcement.
Eric Topol, MD, director of the Scripps Research Translational Institute, professor of molecular medicine, and executive vice president of Scripps Research, said the data the FDA are relying on did not come from the rigorous randomized, double-blind placebo trials that best determine if a treatment is successful.
Still, convalescent plasma is “one more tool added to the arsenal” of combating COVID-19, Mr. Azar said. The FDA will continue to study convalescent plasma as a COVID-19 treatment, Dr. Hahn added.
“We’re waiting for more data. We’re going to continue to gather data,” Dr. Hahn said during the briefing, but the current results meet FDA criteria for issuing an emergency use authorization.
Convalescent plasma “may be effective in lessening the severity or shortening the length of COVID-19 illness in some hospitalized patients,” according to the FDA announcement. Potential side effects include allergic reactions, transfusion-transmitted infections, and transfusion-associated lung injury.
“We’ve seen a great deal of demand for this from doctors around the country,” Dr. Hahn said during the briefing. “The EUA … allows us to continue that and meet that demand.”
Dr. Topol, however, said it appears Trump and the FDA are playing politics with science.
“There’s no evidence to support any survival benefit,” Dr. Topol said on Twitter. “Two days ago [the] FDA’s website stated there was no evidence for an EUA.”
The American Red Cross and other blood centers put out a national call for blood donors in July, especially for patients who have recovered from COVID-19. Mr. Azar and Dr. Hahn emphasized the need for blood donors during the press briefing.
“If you donate plasma, you could save a life,” Mr. Azar said.
The study has not been peer reviewed and did not include a placebo group for comparison, STAT reported.
Last week several health officials warned that the scientific data were too weak to warrant an emergency authorization, the New York Times reported.
A version of this originally appeared on WebMD.com.
Convalescent plasma contains antibodies from the blood of recovered COVID-19 patients, which can be used to treat people with severe infections. Convalescent plasma has been used to treat patients for other infectious diseases. The authorization allows the plasma to be distributed in the United States and administered by health care providers.
“COVID-19 convalescent plasma is safe and shows promising efficacy,” Stephen Hahn, MD, commissioner of the FDA, said during a press briefing with President Donald Trump.
In April, the FDA approved a program to test convalescent plasma in COVID-19 patients at the Mayo Clinic, followed by other institutions. More than 90,000 patients have enrolled in the program, and 70,000 have received the treatment, Dr. Hahn said.
The data indicate that the plasma can reduce mortality in patients by 35%, particularly if patients are treated within 3 days of being diagnosed. Those who have benefited the most were under age 80 and not on artificial respiration, Alex Azar, the secretary for the Department of Health & Human Services, said during the briefing.
“We dream, in drug development, of something like a 35% mortality reduction,” he said.
But top scientists pushed back against the announcement.
Eric Topol, MD, director of the Scripps Research Translational Institute, professor of molecular medicine, and executive vice president of Scripps Research, said the data the FDA are relying on did not come from the rigorous randomized, double-blind placebo trials that best determine if a treatment is successful.
Still, convalescent plasma is “one more tool added to the arsenal” of combating COVID-19, Mr. Azar said. The FDA will continue to study convalescent plasma as a COVID-19 treatment, Dr. Hahn added.
“We’re waiting for more data. We’re going to continue to gather data,” Dr. Hahn said during the briefing, but the current results meet FDA criteria for issuing an emergency use authorization.
Convalescent plasma “may be effective in lessening the severity or shortening the length of COVID-19 illness in some hospitalized patients,” according to the FDA announcement. Potential side effects include allergic reactions, transfusion-transmitted infections, and transfusion-associated lung injury.
“We’ve seen a great deal of demand for this from doctors around the country,” Dr. Hahn said during the briefing. “The EUA … allows us to continue that and meet that demand.”
Dr. Topol, however, said it appears Trump and the FDA are playing politics with science.
“There’s no evidence to support any survival benefit,” Dr. Topol said on Twitter. “Two days ago [the] FDA’s website stated there was no evidence for an EUA.”
The American Red Cross and other blood centers put out a national call for blood donors in July, especially for patients who have recovered from COVID-19. Mr. Azar and Dr. Hahn emphasized the need for blood donors during the press briefing.
“If you donate plasma, you could save a life,” Mr. Azar said.
The study has not been peer reviewed and did not include a placebo group for comparison, STAT reported.
Last week several health officials warned that the scientific data were too weak to warrant an emergency authorization, the New York Times reported.
A version of this originally appeared on WebMD.com.
Convalescent plasma contains antibodies from the blood of recovered COVID-19 patients, which can be used to treat people with severe infections. Convalescent plasma has been used to treat patients for other infectious diseases. The authorization allows the plasma to be distributed in the United States and administered by health care providers.
“COVID-19 convalescent plasma is safe and shows promising efficacy,” Stephen Hahn, MD, commissioner of the FDA, said during a press briefing with President Donald Trump.
In April, the FDA approved a program to test convalescent plasma in COVID-19 patients at the Mayo Clinic, followed by other institutions. More than 90,000 patients have enrolled in the program, and 70,000 have received the treatment, Dr. Hahn said.
The data indicate that the plasma can reduce mortality in patients by 35%, particularly if patients are treated within 3 days of being diagnosed. Those who have benefited the most were under age 80 and not on artificial respiration, Alex Azar, the secretary for the Department of Health & Human Services, said during the briefing.
“We dream, in drug development, of something like a 35% mortality reduction,” he said.
But top scientists pushed back against the announcement.
Eric Topol, MD, director of the Scripps Research Translational Institute, professor of molecular medicine, and executive vice president of Scripps Research, said the data the FDA are relying on did not come from the rigorous randomized, double-blind placebo trials that best determine if a treatment is successful.
Still, convalescent plasma is “one more tool added to the arsenal” of combating COVID-19, Mr. Azar said. The FDA will continue to study convalescent plasma as a COVID-19 treatment, Dr. Hahn added.
“We’re waiting for more data. We’re going to continue to gather data,” Dr. Hahn said during the briefing, but the current results meet FDA criteria for issuing an emergency use authorization.
Convalescent plasma “may be effective in lessening the severity or shortening the length of COVID-19 illness in some hospitalized patients,” according to the FDA announcement. Potential side effects include allergic reactions, transfusion-transmitted infections, and transfusion-associated lung injury.
“We’ve seen a great deal of demand for this from doctors around the country,” Dr. Hahn said during the briefing. “The EUA … allows us to continue that and meet that demand.”
Dr. Topol, however, said it appears Trump and the FDA are playing politics with science.
“There’s no evidence to support any survival benefit,” Dr. Topol said on Twitter. “Two days ago [the] FDA’s website stated there was no evidence for an EUA.”
The American Red Cross and other blood centers put out a national call for blood donors in July, especially for patients who have recovered from COVID-19. Mr. Azar and Dr. Hahn emphasized the need for blood donors during the press briefing.
“If you donate plasma, you could save a life,” Mr. Azar said.
The study has not been peer reviewed and did not include a placebo group for comparison, STAT reported.
Last week several health officials warned that the scientific data were too weak to warrant an emergency authorization, the New York Times reported.
A version of this originally appeared on WebMD.com.
‘The pandemic within the pandemic’
The coronavirus has infected millions of Americans and killed over 174,000. But could it be worse? Maybe.
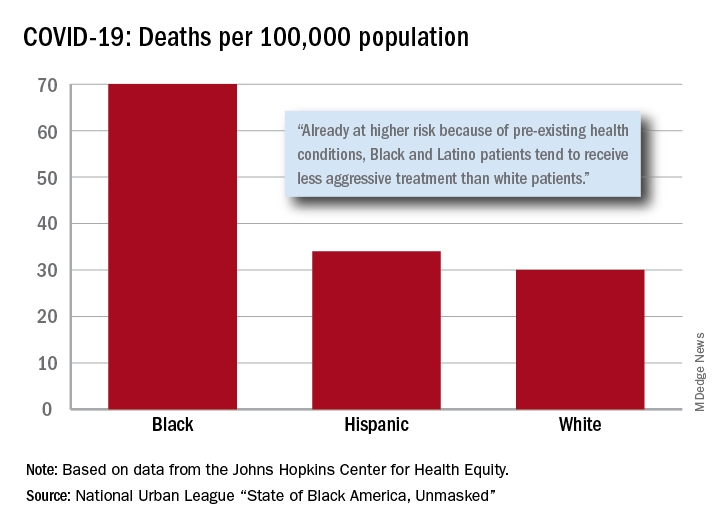
“Racism is the pandemic within the pandemic,” Marc H. Morial, president and CEO of the National Urban League, said in the 2020 “State of Black America, Unmasked” report.
“Black people with COVID-19 symptoms in February and March were less likely to get tested or treated than white patients,” he wrote.
After less testing and less treatment, the next step seems inevitable. The death rate from COVID-19 is 70 per 100,000 population among Black Americans, compared with 30 per 100,000 for Whites and 34 per 100,000 for Hispanics, the league said based on data from the Johns Hopkins Center for Health Equity.
Black and Hispanic patients with COVID-19 are more likely to have preexisting health conditions, but they “tend to receive less aggressive treatment than white patients,” the report noted. The lower death rate among Hispanics may be explained by the Black population’s greater age, although Hispanic Americans have a higher infection rate (73 per 10,000) than Blacks (62 per 10,000) or Whites (23 per 10,000).
Another possible explanation for the differences in infection rates: Blacks and Hispanics are less able to work at home because they “are overrepresented in low-wage jobs that offer the least flexibility and increase their risk of exposure to the coronavirus,” the league said.
Hispanics and Blacks also are more likely to be uninsured than Whites – 19.5% and 11.5%, respectively, vs. 7.5% – so “they tend to delay seeking treatment and are sicker than white patients when they finally do,” the league said. That may account for their much higher COVID-19 hospitalization rates: 213 per 100,000 for Blacks, 205 for Hispanics, and 46 for Whites.
“The silver lining during these dark times is that this pandemic has revealed our shared vulnerability and our interconnectedness. Many people are beginning to see that when others don’t have the opportunity to be healthy, it puts all of us at risk,” Lisa Cooper, MD, James F. Fries Professor of Medicine and Bloomberg Distinguished Professor in Health Equity at Johns Hopkins University, Baltimore, wrote in an essay accompanying the report.
The coronavirus has infected millions of Americans and killed over 174,000. But could it be worse? Maybe.

“Racism is the pandemic within the pandemic,” Marc H. Morial, president and CEO of the National Urban League, said in the 2020 “State of Black America, Unmasked” report.
“Black people with COVID-19 symptoms in February and March were less likely to get tested or treated than white patients,” he wrote.
After less testing and less treatment, the next step seems inevitable. The death rate from COVID-19 is 70 per 100,000 population among Black Americans, compared with 30 per 100,000 for Whites and 34 per 100,000 for Hispanics, the league said based on data from the Johns Hopkins Center for Health Equity.
Black and Hispanic patients with COVID-19 are more likely to have preexisting health conditions, but they “tend to receive less aggressive treatment than white patients,” the report noted. The lower death rate among Hispanics may be explained by the Black population’s greater age, although Hispanic Americans have a higher infection rate (73 per 10,000) than Blacks (62 per 10,000) or Whites (23 per 10,000).
Another possible explanation for the differences in infection rates: Blacks and Hispanics are less able to work at home because they “are overrepresented in low-wage jobs that offer the least flexibility and increase their risk of exposure to the coronavirus,” the league said.
Hispanics and Blacks also are more likely to be uninsured than Whites – 19.5% and 11.5%, respectively, vs. 7.5% – so “they tend to delay seeking treatment and are sicker than white patients when they finally do,” the league said. That may account for their much higher COVID-19 hospitalization rates: 213 per 100,000 for Blacks, 205 for Hispanics, and 46 for Whites.
“The silver lining during these dark times is that this pandemic has revealed our shared vulnerability and our interconnectedness. Many people are beginning to see that when others don’t have the opportunity to be healthy, it puts all of us at risk,” Lisa Cooper, MD, James F. Fries Professor of Medicine and Bloomberg Distinguished Professor in Health Equity at Johns Hopkins University, Baltimore, wrote in an essay accompanying the report.
The coronavirus has infected millions of Americans and killed over 174,000. But could it be worse? Maybe.

“Racism is the pandemic within the pandemic,” Marc H. Morial, president and CEO of the National Urban League, said in the 2020 “State of Black America, Unmasked” report.
“Black people with COVID-19 symptoms in February and March were less likely to get tested or treated than white patients,” he wrote.
After less testing and less treatment, the next step seems inevitable. The death rate from COVID-19 is 70 per 100,000 population among Black Americans, compared with 30 per 100,000 for Whites and 34 per 100,000 for Hispanics, the league said based on data from the Johns Hopkins Center for Health Equity.
Black and Hispanic patients with COVID-19 are more likely to have preexisting health conditions, but they “tend to receive less aggressive treatment than white patients,” the report noted. The lower death rate among Hispanics may be explained by the Black population’s greater age, although Hispanic Americans have a higher infection rate (73 per 10,000) than Blacks (62 per 10,000) or Whites (23 per 10,000).
Another possible explanation for the differences in infection rates: Blacks and Hispanics are less able to work at home because they “are overrepresented in low-wage jobs that offer the least flexibility and increase their risk of exposure to the coronavirus,” the league said.
Hispanics and Blacks also are more likely to be uninsured than Whites – 19.5% and 11.5%, respectively, vs. 7.5% – so “they tend to delay seeking treatment and are sicker than white patients when they finally do,” the league said. That may account for their much higher COVID-19 hospitalization rates: 213 per 100,000 for Blacks, 205 for Hispanics, and 46 for Whites.
“The silver lining during these dark times is that this pandemic has revealed our shared vulnerability and our interconnectedness. Many people are beginning to see that when others don’t have the opportunity to be healthy, it puts all of us at risk,” Lisa Cooper, MD, James F. Fries Professor of Medicine and Bloomberg Distinguished Professor in Health Equity at Johns Hopkins University, Baltimore, wrote in an essay accompanying the report.
Patient visits post COVID-19
Has telemedicine found its footing?
When Alexander Graham Bell invented the telephone, he accomplished something that many telegraph devotees never thought possible: the synchronous, bidirectional transmission of voice over electrical lines.
This was an incredible milestone in the advancement of mankind and enabled true revolutions in commerce, scientific collaboration, and human interaction. But Mr. Bell knew his invention didn’t represent the final advancement in telecommunication; he was quite prescient in imagining a day when individuals could see each other while speaking on the phone.
Many years later, what was once only a dream is now commonplace, and children growing up today can’t imagine a world where apps such as FaceTime and Skype don’t exist. Until recently, however, the medical community has been slow to adopt the idea of video interactions. This has dramatically changed because of the pandemic and the need for social distancing. It appears that telemedicine has found its footing, but whether it will remain popular once patients feel safe going to see their doctors in person again remains to be seen. This month, we’ll examine a few key issues that will determine the future of virtual medical visits.
Collect calling
The pandemic has wrought both human and economic casualties. With fear, job loss, and regulations leading to decreased spending, many large and small businesses have been and will continue to be unable to survive. Companies, including Brooks Brothers, Hertz, Lord and Taylor, GNC, and J.C. Penney, have declared bankruptcy.1 Medical practices and hospitals have taken cuts to their bottom line, and we’ve heard of many physician groups that have had to enact substantial salary cuts or even lay off providers – something previously unheard of. Recent months have demonstrated the health care community’s commitment to put patients first, but we simply cannot survive if we aren’t adequately reimbursed. Traditionally, this has been a significant roadblock toward the widespread adoption of telemedicine.
In most cases, these visits were not reimbursed at all. Thankfully, shortly after the coronavirus hit our shores, Medicare and Medicaid changed their policies, offering equal payment for video and in-person patient encounters. Most private insurers have followed suit, but the commitment to this payment parity appears – thus far – to be temporary. It is unclear that the financial support of telemedicine will continue post COVID-19, and this has many physicians feeling uncomfortable. In the meantime, many patients have come to prefer virtual visits, appreciating the convenience and efficiency.
Physicians don’t always have the same experience. Telemedicine can be technically challenging and take just as much – or sometimes more – time to navigate and document. Unless they are reimbursed equitably, providers will be forced to limit their use of virtual visits or not offer them at all. This leads to another issue: reliability.
‘Can you hear me now?’
Over the past several months, we have had the opportunity to use telemedicine firsthand and have spoken to many other physicians and patients about their experiences with it. The reports are all quite consistent: Most have had generally positive things to say. Still, some common concerns emerge when diving a bit deeper. Most notably are complaints about usability and reliability of the software.
While there are large telemedicine companies that have developed world-class cross-platform products, many in use today are proprietary and EHR dependent. As a result, the quality varies widely. Many EHR vendors were caught completely off guard by the sudden demand for telemedicine and are playing catch-up as they develop their own virtual visit platforms. While these vendor-developed platforms promise tight integration with patient records, some have significant shortcomings in stability when taxed under high utilization, including choppy video and garbled voice. This simply won’t do if telemedicine is to survive. It is incumbent on software developers and health care providers to invest in high-quality, reliable platforms on which to build their virtual visit offerings. This will ensure a more rapid adoption and the “staying power” of the new technology.
Dialing ‘0’ for the operator
Once seen as a “novelty” offered by only a small number of medical providers, virtual visits now represent a significant and ever-increasing percentage of patient encounters. The technology therefore must be easy to use. Given confidentiality and documentation requirements, along with the broad variety of available computing platforms and devices (e.g., PC, Mac, iOS, and Android), the process is often far from problem free. Patients may need help downloading apps, setting up webcams, or registering for the service. Providers may face issues with Internet connectivity or EHR-related delays.
It is critical that help be available to make the connection seamless and the experience a positive one. We are fortunate to work for a health care institution that has made this a priority, dedicating a team of individuals to provide real-time support to patients and clinicians. Small independent practices may not have this luxury, but we would encourage all providers to engage with their telemedicine or EHR vendors to determine what resources are available when problems arise, as they undoubtedly will.
Answering the call
Like the invention of the telephone, the advent of telemedicine is another milestone on the journey toward better communication with our patients, and it appears to be here to stay. Virtual visits won’t completely replace in-person care, nor minimize the benefit of human interaction, but they will continue to play an important role in the care continuum. By addressing the above concerns, we’ll lay a solid foundation for success and create a positive experience for physicians and patients alike.
Dr. Notte is a family physician and chief medical officer of Abington (Pa.) Hospital–Jefferson Health. Follow him on Twitter (@doctornotte). Dr. Skolnik is professor of family and community medicine at Sidney Kimmel Medical College, Philadelphia, and associate director of the family medicine residency program at Abington (Pa.) Hospital–Jefferson Health. They have no conflicts related to the content of this piece.
Reference
1. A running list of companies that have filed for bankruptcy during the coronavirus pandemic. Fortune.
Has telemedicine found its footing?
Has telemedicine found its footing?
When Alexander Graham Bell invented the telephone, he accomplished something that many telegraph devotees never thought possible: the synchronous, bidirectional transmission of voice over electrical lines.
This was an incredible milestone in the advancement of mankind and enabled true revolutions in commerce, scientific collaboration, and human interaction. But Mr. Bell knew his invention didn’t represent the final advancement in telecommunication; he was quite prescient in imagining a day when individuals could see each other while speaking on the phone.
Many years later, what was once only a dream is now commonplace, and children growing up today can’t imagine a world where apps such as FaceTime and Skype don’t exist. Until recently, however, the medical community has been slow to adopt the idea of video interactions. This has dramatically changed because of the pandemic and the need for social distancing. It appears that telemedicine has found its footing, but whether it will remain popular once patients feel safe going to see their doctors in person again remains to be seen. This month, we’ll examine a few key issues that will determine the future of virtual medical visits.
Collect calling
The pandemic has wrought both human and economic casualties. With fear, job loss, and regulations leading to decreased spending, many large and small businesses have been and will continue to be unable to survive. Companies, including Brooks Brothers, Hertz, Lord and Taylor, GNC, and J.C. Penney, have declared bankruptcy.1 Medical practices and hospitals have taken cuts to their bottom line, and we’ve heard of many physician groups that have had to enact substantial salary cuts or even lay off providers – something previously unheard of. Recent months have demonstrated the health care community’s commitment to put patients first, but we simply cannot survive if we aren’t adequately reimbursed. Traditionally, this has been a significant roadblock toward the widespread adoption of telemedicine.
In most cases, these visits were not reimbursed at all. Thankfully, shortly after the coronavirus hit our shores, Medicare and Medicaid changed their policies, offering equal payment for video and in-person patient encounters. Most private insurers have followed suit, but the commitment to this payment parity appears – thus far – to be temporary. It is unclear that the financial support of telemedicine will continue post COVID-19, and this has many physicians feeling uncomfortable. In the meantime, many patients have come to prefer virtual visits, appreciating the convenience and efficiency.
Physicians don’t always have the same experience. Telemedicine can be technically challenging and take just as much – or sometimes more – time to navigate and document. Unless they are reimbursed equitably, providers will be forced to limit their use of virtual visits or not offer them at all. This leads to another issue: reliability.
‘Can you hear me now?’
Over the past several months, we have had the opportunity to use telemedicine firsthand and have spoken to many other physicians and patients about their experiences with it. The reports are all quite consistent: Most have had generally positive things to say. Still, some common concerns emerge when diving a bit deeper. Most notably are complaints about usability and reliability of the software.
While there are large telemedicine companies that have developed world-class cross-platform products, many in use today are proprietary and EHR dependent. As a result, the quality varies widely. Many EHR vendors were caught completely off guard by the sudden demand for telemedicine and are playing catch-up as they develop their own virtual visit platforms. While these vendor-developed platforms promise tight integration with patient records, some have significant shortcomings in stability when taxed under high utilization, including choppy video and garbled voice. This simply won’t do if telemedicine is to survive. It is incumbent on software developers and health care providers to invest in high-quality, reliable platforms on which to build their virtual visit offerings. This will ensure a more rapid adoption and the “staying power” of the new technology.
Dialing ‘0’ for the operator
Once seen as a “novelty” offered by only a small number of medical providers, virtual visits now represent a significant and ever-increasing percentage of patient encounters. The technology therefore must be easy to use. Given confidentiality and documentation requirements, along with the broad variety of available computing platforms and devices (e.g., PC, Mac, iOS, and Android), the process is often far from problem free. Patients may need help downloading apps, setting up webcams, or registering for the service. Providers may face issues with Internet connectivity or EHR-related delays.
It is critical that help be available to make the connection seamless and the experience a positive one. We are fortunate to work for a health care institution that has made this a priority, dedicating a team of individuals to provide real-time support to patients and clinicians. Small independent practices may not have this luxury, but we would encourage all providers to engage with their telemedicine or EHR vendors to determine what resources are available when problems arise, as they undoubtedly will.
Answering the call
Like the invention of the telephone, the advent of telemedicine is another milestone on the journey toward better communication with our patients, and it appears to be here to stay. Virtual visits won’t completely replace in-person care, nor minimize the benefit of human interaction, but they will continue to play an important role in the care continuum. By addressing the above concerns, we’ll lay a solid foundation for success and create a positive experience for physicians and patients alike.
Dr. Notte is a family physician and chief medical officer of Abington (Pa.) Hospital–Jefferson Health. Follow him on Twitter (@doctornotte). Dr. Skolnik is professor of family and community medicine at Sidney Kimmel Medical College, Philadelphia, and associate director of the family medicine residency program at Abington (Pa.) Hospital–Jefferson Health. They have no conflicts related to the content of this piece.
Reference
1. A running list of companies that have filed for bankruptcy during the coronavirus pandemic. Fortune.
When Alexander Graham Bell invented the telephone, he accomplished something that many telegraph devotees never thought possible: the synchronous, bidirectional transmission of voice over electrical lines.
This was an incredible milestone in the advancement of mankind and enabled true revolutions in commerce, scientific collaboration, and human interaction. But Mr. Bell knew his invention didn’t represent the final advancement in telecommunication; he was quite prescient in imagining a day when individuals could see each other while speaking on the phone.
Many years later, what was once only a dream is now commonplace, and children growing up today can’t imagine a world where apps such as FaceTime and Skype don’t exist. Until recently, however, the medical community has been slow to adopt the idea of video interactions. This has dramatically changed because of the pandemic and the need for social distancing. It appears that telemedicine has found its footing, but whether it will remain popular once patients feel safe going to see their doctors in person again remains to be seen. This month, we’ll examine a few key issues that will determine the future of virtual medical visits.
Collect calling
The pandemic has wrought both human and economic casualties. With fear, job loss, and regulations leading to decreased spending, many large and small businesses have been and will continue to be unable to survive. Companies, including Brooks Brothers, Hertz, Lord and Taylor, GNC, and J.C. Penney, have declared bankruptcy.1 Medical practices and hospitals have taken cuts to their bottom line, and we’ve heard of many physician groups that have had to enact substantial salary cuts or even lay off providers – something previously unheard of. Recent months have demonstrated the health care community’s commitment to put patients first, but we simply cannot survive if we aren’t adequately reimbursed. Traditionally, this has been a significant roadblock toward the widespread adoption of telemedicine.
In most cases, these visits were not reimbursed at all. Thankfully, shortly after the coronavirus hit our shores, Medicare and Medicaid changed their policies, offering equal payment for video and in-person patient encounters. Most private insurers have followed suit, but the commitment to this payment parity appears – thus far – to be temporary. It is unclear that the financial support of telemedicine will continue post COVID-19, and this has many physicians feeling uncomfortable. In the meantime, many patients have come to prefer virtual visits, appreciating the convenience and efficiency.
Physicians don’t always have the same experience. Telemedicine can be technically challenging and take just as much – or sometimes more – time to navigate and document. Unless they are reimbursed equitably, providers will be forced to limit their use of virtual visits or not offer them at all. This leads to another issue: reliability.
‘Can you hear me now?’
Over the past several months, we have had the opportunity to use telemedicine firsthand and have spoken to many other physicians and patients about their experiences with it. The reports are all quite consistent: Most have had generally positive things to say. Still, some common concerns emerge when diving a bit deeper. Most notably are complaints about usability and reliability of the software.
While there are large telemedicine companies that have developed world-class cross-platform products, many in use today are proprietary and EHR dependent. As a result, the quality varies widely. Many EHR vendors were caught completely off guard by the sudden demand for telemedicine and are playing catch-up as they develop their own virtual visit platforms. While these vendor-developed platforms promise tight integration with patient records, some have significant shortcomings in stability when taxed under high utilization, including choppy video and garbled voice. This simply won’t do if telemedicine is to survive. It is incumbent on software developers and health care providers to invest in high-quality, reliable platforms on which to build their virtual visit offerings. This will ensure a more rapid adoption and the “staying power” of the new technology.
Dialing ‘0’ for the operator
Once seen as a “novelty” offered by only a small number of medical providers, virtual visits now represent a significant and ever-increasing percentage of patient encounters. The technology therefore must be easy to use. Given confidentiality and documentation requirements, along with the broad variety of available computing platforms and devices (e.g., PC, Mac, iOS, and Android), the process is often far from problem free. Patients may need help downloading apps, setting up webcams, or registering for the service. Providers may face issues with Internet connectivity or EHR-related delays.
It is critical that help be available to make the connection seamless and the experience a positive one. We are fortunate to work for a health care institution that has made this a priority, dedicating a team of individuals to provide real-time support to patients and clinicians. Small independent practices may not have this luxury, but we would encourage all providers to engage with their telemedicine or EHR vendors to determine what resources are available when problems arise, as they undoubtedly will.
Answering the call
Like the invention of the telephone, the advent of telemedicine is another milestone on the journey toward better communication with our patients, and it appears to be here to stay. Virtual visits won’t completely replace in-person care, nor minimize the benefit of human interaction, but they will continue to play an important role in the care continuum. By addressing the above concerns, we’ll lay a solid foundation for success and create a positive experience for physicians and patients alike.
Dr. Notte is a family physician and chief medical officer of Abington (Pa.) Hospital–Jefferson Health. Follow him on Twitter (@doctornotte). Dr. Skolnik is professor of family and community medicine at Sidney Kimmel Medical College, Philadelphia, and associate director of the family medicine residency program at Abington (Pa.) Hospital–Jefferson Health. They have no conflicts related to the content of this piece.
Reference
1. A running list of companies that have filed for bankruptcy during the coronavirus pandemic. Fortune.
Hospitalists confront administrative, financial challenges of COVID-19 crisis
Hospitalists nationwide have put in longer hours, played new clinical roles, and stretched beyond their medical specialty and comfort level to meet their hospital’s COVID-19 care demands. Can they expect some kind of financial recognition – perhaps in the form of “hazard pay” for going above and beyond – even though their institutions are experiencing negative financial fallout from the crisis?
Hospitals in regions experiencing a COVID-19 surge have limited elective procedures, discouraged non–COVID-19 admissions, and essentially entered crisis management mode. Other facilities in less hard-hit communities are also standing by, with reduced hospital census, smaller caseloads and less work to do, while trying to prepare their bottom lines for lower demand.
“This crisis has put most hospitals in financial jeopardy and that is likely to trickle down to all employees – including hospitalists,” said Ron Greeno, MD, FCCP, MHM, a past president of SHM and the society’s current senior advisor for government affairs. “But it’s not like hospitals could or would forgo an effective hospitalist program today. Hospitalists will be important players in defining the hospital’s future direction post crisis.”
That doesn’t mean tighter financials, caps on annual salary increases, or higher productivity expectations won’t be part of future conversations between hospital administrators and their hospitalists, Dr. Greeno said. Administrators are starting to look ahead to the post–COVID-19 era even as numbers of cases and rates of growth continue to rise in various regions, and Dr. Greeno sees a lot of uncertainty ahead.
Even prior to the crisis, he noted, hospital margins had been falling, while the cost of labor, including hospitalist labor, was going up. That was pointing toward an inevitable collision, which has only intensified with the new financial crisis facing hospitals – created by SARS-CoV-2 and by policies such as shutting down elective surgeries in anticipation of a COVID-19 patient surge that, for some institutions, may never come.
Brian Harte, MD, MHM, president of Cleveland Clinic Akron General and a past president of SHM, said that the Cleveland Clinic system has been planning since January its response to the coming crisis. “Governor Mike DeWine and the state Department of Health led the way in flattening the curve in Ohio. We engaged our hospitalists in brainstorming solutions. They have been excellent partners,” he said.
Approaching the crisis with a sense of urgency from the outset, the Cleveland Clinic built a COVID-19 surge team and incident command structure, with nursing, infectious diseases, critical care and hospital medicine represented. “We used that time to get ready for what was coming. We worked on streamlining consultant work flows.”
But utilization numbers are off in almost every service line, Dr. Harte said. “It has forced us to look at things we’ve always talked about, including greater use of telemedicine and exploring other ways of caring for patients, such as increased use of evening hours.”
Cleveland Clinic contracts with Sound Physicians of Tacoma, Wash., for its hospitalist coverage. “We have an excellent working relationship with Sound at the local, regional, and national levels, with common goals for quality and utilization. We tried to involve our hospitalists as early as possible in planning. We needed them to step in and role model and lead the way,” Dr. Harte said, for everybody’s anxiety levels.
“We’re still in the process of understanding the long-term financial impact of the epidemic,” Dr. Harte added. “But at this point I see no reason to think our relationship with our hospitalists needs to change. We’re the stewards of long-term finances. We’ll need to keep a close eye on this. But we’re committed to working through this together.”
Hazard pay for frontline health care workers was included in the COVID-19 relief package assembled in mid-May by Democrats in the House of Representatives. The $3 trillion HEROES Act includes $200 billion to award hazard pay to essential workers, including those in the health field, but Senate Majority Leader Mitch McConnell (R-Ky.) declared the legislation “dead on arrival” in the Senate.
Supplementary hazard payments made by hospitals to their hospitalists as a reward for sacrifices they made in the crisis is an interesting question, Dr. Greeno noted, and it’s definitely on the table at some hospitals. “But I think it is going to be a tough ask in these times.”
Dr. Harte said he has not offered nor been asked about hazard pay for hospitalists. Cleveland Clinic Akron General made a strategic decision that hazard pay was not going to be part of its response to the pandemic. Other hospital administrators interviewed for this article concur.
Hospitals respond to the fiscal crisis
Hospitals in other parts of the country also report significant fiscal fallout from the COVID-19 crisis, with predictions that 100 or more hospitals may be forced to close. Jeff Dye, president of the New Mexico Hospital Association, told the Albuquerque Journal on May 1 that hospitals in his state have been squeezed on all sides by increased costs, patients delaying routine care, and public health orders restricting elective surgeries. New Mexico hospitals, especially in rural areas, face incredible financial strain.
The University of Virginia Medical Center, Charlottesville, recently announced 20% reductions in total compensation for its providers through July 31, along with suspension of retirement contributions. Those changes won’t affect team members caring for COVID-19 patients. And the Spectrum Health Medical Group of 15 hospitals in western Michigan, according to Michigan Public Radio, told its doctors they either needed to sign “contract addendums” giving the system more control over their hours – or face a 25% pay cut, or worse.
Cheyenne (Wyo.) Regional Medical Center issued a statement April 24 that it expected losses of $10 million for the month of April. “CRMC, like every other hospital in Wyoming, is certainly feeling the financial impact that COVID-19 is having,” CEO Tim Thornell told the Cowboy State Daily on April 24. That includes a 30% reduction in inpatient care and 50% reduction in outpatient care, while the hospital has only had a handful of COVID-19 patients at any time. Capital projects are now on hold, overtime is limited, and a hiring freeze is in effect.
“We’re certainly prepared for a larger surge, which hasn’t come yet,” Mr. Thornell said in an interview. CRMC’s ICU was split to create a nine-bed dedicated COVID-19 unit. Intensivists see most of the critical care patients, while the hospital’s 15 directly-employed hospitalists are treating all of the non-ICU COVID-19 patients. “Among themselves, the hospitalists volunteered who would work on the unit. We’ve been fortunate enough to have enough volunteers and enough PPE [personal protective equipment],” he said.
Preparing for the COVID-19 pandemic has strengthened the medical center’s relationship with its hospitalists, Mr. Thornell explained. “Hospitalists are key to our operations, involved in so much that happens here. We’re trying to staff to volume with decreased utilization. We’ve scaled back, which only makes fiscal sense. Now, how do we reinfuse patients back into the mix? Our hospitalists are paid by the number of shifts, and as you distribute shift reductions over 15 providers, it shouldn’t be an intolerable burden.” But two open hospitalist positions have not been filled, he noted.
CRMC is trying to approach these changes with a Lean perspective, Mr. Thornell said. “We had already adopted a Lean program, but this has been a chance to go through a life-altering circumstance using the tools of Lean planning and applying them instantaneously.”
Providers step up
At Emory Healthcare in Atlanta, a major center for COVID-19 cases, communication has been essential in the crisis, said Bryce Gartland, MD, SFHM, Emory’s hospital group president and cochief of clinical operations. “Our group was prepared for a significant influx of patients. Like every other institution, we made the decision to postpone elective care, with a resulting plummet in volume,” he said.
As COVID-19 patients entered the Emory system, frontline hospitalists stepped up to care for those patients. “We’ve had ample providers in terms of clinical care. We guaranteed our physicians’ base compensation. They have flexed teams up and down as needed.” Advanced practice professionals also stepped up to bridge gaps.
With regard to the return of volumes of non–COVID-19 patients, the jury’s still out, Dr. Gartland said. “None of us has a crystal ball, and there are tremendous variables and decision points that will have significant impact. We have started to see numbers of time-sensitive and essential cases increase as of the first week of May.”
What lies ahead will likely include some rightsizing to future volumes. On top of that, the broader economic pressures on hospitals from high rates of unemployment, uninsured patients, bad debt, and charity care will push health care systems to significantly address costs and infrastructure, he said. “We’re still early in planning, and striving to maintain flexibility and nimbleness, given the uncertainties to this early understanding of our new normal. No hospital is immune from the financial impact. We’ll see and hear about more of these conversations in the months ahead.”
But the experience has also generated some positives, Dr. Gartland noted. “Things like telehealth, which we’ve been talking about for years but previously faced barriers to widespread adoption.” Now with COVID-19, the federal government issued waivers, and barriers – both internal and external – came down. “With telehealth, what will the role and deployment of hospitalists look like in this new model? How will traditional productivity expectations change, or the numbers and types of providers? This will make the relationship and partnership between hospitalist groups and hospital administrators ever more important as we consider the evolution toward new care models.”
Dr. Gartland said that “one of the great things about hospital medicine as a field is its flexibility and adaptability. Where there have been gaps, hospitalists were quick to step in. As long as hospital medicine continues to embrace those kinds of behaviors, it will be successful.” But if the conversation with hospitals is just about money, it will be harder, he acknowledged. “Where there is this kind of disruption in our usual way of doing things, there are also tremendous opportunities for care model innovation. I would encourage hospitalist groups to try to be true value partners.”
Command center mode
Like other physicians in hospital C-suites, Chad Whelan MD, FACP, SFHM, chief executive officer of Banner–University Medicine in Tucson, Ariz., led his two hospitals into command center mode when the crisis hit, planning for a surge of COVID-19 cases that could overwhelm hospital capacity.
“In terms of our hospitalists, we leaned in to them hard in the beginning, preparing them to supervise other physicians who came in to help if needed,” he said. “Our [non–COVID-19] census is down, revenues are down, and the implications are enormous – like nothing we’ve ever seen before.”
“We’re fortunate that we’re part of the Banner health system. We made a decision that we would essentially keep our physicians financially protected through this crisis,” Dr. Whelan said. “In return, we called on them to step up and be on the front lines and to put in enormous hours for planning. We asked them to consider: How could you contribute if the surge comes?”
He affirmed that hospital medicine has been a major part of his medical center’s planning and implementation. “I’ve been overwhelmed by the degree to which the entire delivery team has rallied around the pandemic, with everybody saying they want to keep people safe and be part of the solution. We have always had hospitalist leaders at the table as we’ve planned our response and as decisions were made,” said Dr. Whelan, a practicing hospitalist and teaching service attending since 2000 until he assumed his current executive position in Arizona 18 months ago.
“While we have kept people whole during the immediate crisis, we have acknowledged that we don’t know what our recovery will look like. What if [non–COVID-19] volume doesn’t return? That keeps me awake at night,” he said. “I have talked to our physician leadership in hospital medicine and more broadly. We need to ask ourselves many questions, including: do we have the right levels of staffing? Is this the time to consider alternate models of staffing, for example, advanced practice providers? And does the compensation plan need adjustments?”
Dr. Whelan thinks that the COVID-19 crisis is an opportunity for hospital medicine to more rapidly explore different models and to ask what additional value hospitalists can bring to the care model. “For example, what would it mean to redefine the hospitalist’s scope of practice as an acute medicine specialist, not defined by the hospital’s four walls?” he noted.
“One of the reasons our smaller hospital reached capacity with COVID-19 patients was the skilled nursing facility located a few hundred feet away that turned into a hot spot. If we had imported the hospital medicine model virtually into that SNF early on, could there have been a different scenario? Have we thought through what that would have even looked like?” Dr. Whelan asked.
He challenges the hospital medicine field, once it gets to the other side of this crisis, to not fall back on old way of doing things. “Instead, let’s use this time to create a better model today,” he said. “That’s what we’re trying to do at a system level at Banner, with our hospital medicine groups partnering with the hospital. I want to see our hospitalists create and thrive in that new model.”
Hospitalists nationwide have put in longer hours, played new clinical roles, and stretched beyond their medical specialty and comfort level to meet their hospital’s COVID-19 care demands. Can they expect some kind of financial recognition – perhaps in the form of “hazard pay” for going above and beyond – even though their institutions are experiencing negative financial fallout from the crisis?
Hospitals in regions experiencing a COVID-19 surge have limited elective procedures, discouraged non–COVID-19 admissions, and essentially entered crisis management mode. Other facilities in less hard-hit communities are also standing by, with reduced hospital census, smaller caseloads and less work to do, while trying to prepare their bottom lines for lower demand.
“This crisis has put most hospitals in financial jeopardy and that is likely to trickle down to all employees – including hospitalists,” said Ron Greeno, MD, FCCP, MHM, a past president of SHM and the society’s current senior advisor for government affairs. “But it’s not like hospitals could or would forgo an effective hospitalist program today. Hospitalists will be important players in defining the hospital’s future direction post crisis.”
That doesn’t mean tighter financials, caps on annual salary increases, or higher productivity expectations won’t be part of future conversations between hospital administrators and their hospitalists, Dr. Greeno said. Administrators are starting to look ahead to the post–COVID-19 era even as numbers of cases and rates of growth continue to rise in various regions, and Dr. Greeno sees a lot of uncertainty ahead.
Even prior to the crisis, he noted, hospital margins had been falling, while the cost of labor, including hospitalist labor, was going up. That was pointing toward an inevitable collision, which has only intensified with the new financial crisis facing hospitals – created by SARS-CoV-2 and by policies such as shutting down elective surgeries in anticipation of a COVID-19 patient surge that, for some institutions, may never come.
Brian Harte, MD, MHM, president of Cleveland Clinic Akron General and a past president of SHM, said that the Cleveland Clinic system has been planning since January its response to the coming crisis. “Governor Mike DeWine and the state Department of Health led the way in flattening the curve in Ohio. We engaged our hospitalists in brainstorming solutions. They have been excellent partners,” he said.
Approaching the crisis with a sense of urgency from the outset, the Cleveland Clinic built a COVID-19 surge team and incident command structure, with nursing, infectious diseases, critical care and hospital medicine represented. “We used that time to get ready for what was coming. We worked on streamlining consultant work flows.”
But utilization numbers are off in almost every service line, Dr. Harte said. “It has forced us to look at things we’ve always talked about, including greater use of telemedicine and exploring other ways of caring for patients, such as increased use of evening hours.”
Cleveland Clinic contracts with Sound Physicians of Tacoma, Wash., for its hospitalist coverage. “We have an excellent working relationship with Sound at the local, regional, and national levels, with common goals for quality and utilization. We tried to involve our hospitalists as early as possible in planning. We needed them to step in and role model and lead the way,” Dr. Harte said, for everybody’s anxiety levels.
“We’re still in the process of understanding the long-term financial impact of the epidemic,” Dr. Harte added. “But at this point I see no reason to think our relationship with our hospitalists needs to change. We’re the stewards of long-term finances. We’ll need to keep a close eye on this. But we’re committed to working through this together.”
Hazard pay for frontline health care workers was included in the COVID-19 relief package assembled in mid-May by Democrats in the House of Representatives. The $3 trillion HEROES Act includes $200 billion to award hazard pay to essential workers, including those in the health field, but Senate Majority Leader Mitch McConnell (R-Ky.) declared the legislation “dead on arrival” in the Senate.
Supplementary hazard payments made by hospitals to their hospitalists as a reward for sacrifices they made in the crisis is an interesting question, Dr. Greeno noted, and it’s definitely on the table at some hospitals. “But I think it is going to be a tough ask in these times.”
Dr. Harte said he has not offered nor been asked about hazard pay for hospitalists. Cleveland Clinic Akron General made a strategic decision that hazard pay was not going to be part of its response to the pandemic. Other hospital administrators interviewed for this article concur.
Hospitals respond to the fiscal crisis
Hospitals in other parts of the country also report significant fiscal fallout from the COVID-19 crisis, with predictions that 100 or more hospitals may be forced to close. Jeff Dye, president of the New Mexico Hospital Association, told the Albuquerque Journal on May 1 that hospitals in his state have been squeezed on all sides by increased costs, patients delaying routine care, and public health orders restricting elective surgeries. New Mexico hospitals, especially in rural areas, face incredible financial strain.
The University of Virginia Medical Center, Charlottesville, recently announced 20% reductions in total compensation for its providers through July 31, along with suspension of retirement contributions. Those changes won’t affect team members caring for COVID-19 patients. And the Spectrum Health Medical Group of 15 hospitals in western Michigan, according to Michigan Public Radio, told its doctors they either needed to sign “contract addendums” giving the system more control over their hours – or face a 25% pay cut, or worse.
Cheyenne (Wyo.) Regional Medical Center issued a statement April 24 that it expected losses of $10 million for the month of April. “CRMC, like every other hospital in Wyoming, is certainly feeling the financial impact that COVID-19 is having,” CEO Tim Thornell told the Cowboy State Daily on April 24. That includes a 30% reduction in inpatient care and 50% reduction in outpatient care, while the hospital has only had a handful of COVID-19 patients at any time. Capital projects are now on hold, overtime is limited, and a hiring freeze is in effect.
“We’re certainly prepared for a larger surge, which hasn’t come yet,” Mr. Thornell said in an interview. CRMC’s ICU was split to create a nine-bed dedicated COVID-19 unit. Intensivists see most of the critical care patients, while the hospital’s 15 directly-employed hospitalists are treating all of the non-ICU COVID-19 patients. “Among themselves, the hospitalists volunteered who would work on the unit. We’ve been fortunate enough to have enough volunteers and enough PPE [personal protective equipment],” he said.
Preparing for the COVID-19 pandemic has strengthened the medical center’s relationship with its hospitalists, Mr. Thornell explained. “Hospitalists are key to our operations, involved in so much that happens here. We’re trying to staff to volume with decreased utilization. We’ve scaled back, which only makes fiscal sense. Now, how do we reinfuse patients back into the mix? Our hospitalists are paid by the number of shifts, and as you distribute shift reductions over 15 providers, it shouldn’t be an intolerable burden.” But two open hospitalist positions have not been filled, he noted.
CRMC is trying to approach these changes with a Lean perspective, Mr. Thornell said. “We had already adopted a Lean program, but this has been a chance to go through a life-altering circumstance using the tools of Lean planning and applying them instantaneously.”
Providers step up
At Emory Healthcare in Atlanta, a major center for COVID-19 cases, communication has been essential in the crisis, said Bryce Gartland, MD, SFHM, Emory’s hospital group president and cochief of clinical operations. “Our group was prepared for a significant influx of patients. Like every other institution, we made the decision to postpone elective care, with a resulting plummet in volume,” he said.
As COVID-19 patients entered the Emory system, frontline hospitalists stepped up to care for those patients. “We’ve had ample providers in terms of clinical care. We guaranteed our physicians’ base compensation. They have flexed teams up and down as needed.” Advanced practice professionals also stepped up to bridge gaps.
With regard to the return of volumes of non–COVID-19 patients, the jury’s still out, Dr. Gartland said. “None of us has a crystal ball, and there are tremendous variables and decision points that will have significant impact. We have started to see numbers of time-sensitive and essential cases increase as of the first week of May.”
What lies ahead will likely include some rightsizing to future volumes. On top of that, the broader economic pressures on hospitals from high rates of unemployment, uninsured patients, bad debt, and charity care will push health care systems to significantly address costs and infrastructure, he said. “We’re still early in planning, and striving to maintain flexibility and nimbleness, given the uncertainties to this early understanding of our new normal. No hospital is immune from the financial impact. We’ll see and hear about more of these conversations in the months ahead.”
But the experience has also generated some positives, Dr. Gartland noted. “Things like telehealth, which we’ve been talking about for years but previously faced barriers to widespread adoption.” Now with COVID-19, the federal government issued waivers, and barriers – both internal and external – came down. “With telehealth, what will the role and deployment of hospitalists look like in this new model? How will traditional productivity expectations change, or the numbers and types of providers? This will make the relationship and partnership between hospitalist groups and hospital administrators ever more important as we consider the evolution toward new care models.”
Dr. Gartland said that “one of the great things about hospital medicine as a field is its flexibility and adaptability. Where there have been gaps, hospitalists were quick to step in. As long as hospital medicine continues to embrace those kinds of behaviors, it will be successful.” But if the conversation with hospitals is just about money, it will be harder, he acknowledged. “Where there is this kind of disruption in our usual way of doing things, there are also tremendous opportunities for care model innovation. I would encourage hospitalist groups to try to be true value partners.”
Command center mode
Like other physicians in hospital C-suites, Chad Whelan MD, FACP, SFHM, chief executive officer of Banner–University Medicine in Tucson, Ariz., led his two hospitals into command center mode when the crisis hit, planning for a surge of COVID-19 cases that could overwhelm hospital capacity.
“In terms of our hospitalists, we leaned in to them hard in the beginning, preparing them to supervise other physicians who came in to help if needed,” he said. “Our [non–COVID-19] census is down, revenues are down, and the implications are enormous – like nothing we’ve ever seen before.”
“We’re fortunate that we’re part of the Banner health system. We made a decision that we would essentially keep our physicians financially protected through this crisis,” Dr. Whelan said. “In return, we called on them to step up and be on the front lines and to put in enormous hours for planning. We asked them to consider: How could you contribute if the surge comes?”
He affirmed that hospital medicine has been a major part of his medical center’s planning and implementation. “I’ve been overwhelmed by the degree to which the entire delivery team has rallied around the pandemic, with everybody saying they want to keep people safe and be part of the solution. We have always had hospitalist leaders at the table as we’ve planned our response and as decisions were made,” said Dr. Whelan, a practicing hospitalist and teaching service attending since 2000 until he assumed his current executive position in Arizona 18 months ago.
“While we have kept people whole during the immediate crisis, we have acknowledged that we don’t know what our recovery will look like. What if [non–COVID-19] volume doesn’t return? That keeps me awake at night,” he said. “I have talked to our physician leadership in hospital medicine and more broadly. We need to ask ourselves many questions, including: do we have the right levels of staffing? Is this the time to consider alternate models of staffing, for example, advanced practice providers? And does the compensation plan need adjustments?”
Dr. Whelan thinks that the COVID-19 crisis is an opportunity for hospital medicine to more rapidly explore different models and to ask what additional value hospitalists can bring to the care model. “For example, what would it mean to redefine the hospitalist’s scope of practice as an acute medicine specialist, not defined by the hospital’s four walls?” he noted.
“One of the reasons our smaller hospital reached capacity with COVID-19 patients was the skilled nursing facility located a few hundred feet away that turned into a hot spot. If we had imported the hospital medicine model virtually into that SNF early on, could there have been a different scenario? Have we thought through what that would have even looked like?” Dr. Whelan asked.
He challenges the hospital medicine field, once it gets to the other side of this crisis, to not fall back on old way of doing things. “Instead, let’s use this time to create a better model today,” he said. “That’s what we’re trying to do at a system level at Banner, with our hospital medicine groups partnering with the hospital. I want to see our hospitalists create and thrive in that new model.”
Hospitalists nationwide have put in longer hours, played new clinical roles, and stretched beyond their medical specialty and comfort level to meet their hospital’s COVID-19 care demands. Can they expect some kind of financial recognition – perhaps in the form of “hazard pay” for going above and beyond – even though their institutions are experiencing negative financial fallout from the crisis?
Hospitals in regions experiencing a COVID-19 surge have limited elective procedures, discouraged non–COVID-19 admissions, and essentially entered crisis management mode. Other facilities in less hard-hit communities are also standing by, with reduced hospital census, smaller caseloads and less work to do, while trying to prepare their bottom lines for lower demand.
“This crisis has put most hospitals in financial jeopardy and that is likely to trickle down to all employees – including hospitalists,” said Ron Greeno, MD, FCCP, MHM, a past president of SHM and the society’s current senior advisor for government affairs. “But it’s not like hospitals could or would forgo an effective hospitalist program today. Hospitalists will be important players in defining the hospital’s future direction post crisis.”
That doesn’t mean tighter financials, caps on annual salary increases, or higher productivity expectations won’t be part of future conversations between hospital administrators and their hospitalists, Dr. Greeno said. Administrators are starting to look ahead to the post–COVID-19 era even as numbers of cases and rates of growth continue to rise in various regions, and Dr. Greeno sees a lot of uncertainty ahead.
Even prior to the crisis, he noted, hospital margins had been falling, while the cost of labor, including hospitalist labor, was going up. That was pointing toward an inevitable collision, which has only intensified with the new financial crisis facing hospitals – created by SARS-CoV-2 and by policies such as shutting down elective surgeries in anticipation of a COVID-19 patient surge that, for some institutions, may never come.
Brian Harte, MD, MHM, president of Cleveland Clinic Akron General and a past president of SHM, said that the Cleveland Clinic system has been planning since January its response to the coming crisis. “Governor Mike DeWine and the state Department of Health led the way in flattening the curve in Ohio. We engaged our hospitalists in brainstorming solutions. They have been excellent partners,” he said.
Approaching the crisis with a sense of urgency from the outset, the Cleveland Clinic built a COVID-19 surge team and incident command structure, with nursing, infectious diseases, critical care and hospital medicine represented. “We used that time to get ready for what was coming. We worked on streamlining consultant work flows.”
But utilization numbers are off in almost every service line, Dr. Harte said. “It has forced us to look at things we’ve always talked about, including greater use of telemedicine and exploring other ways of caring for patients, such as increased use of evening hours.”
Cleveland Clinic contracts with Sound Physicians of Tacoma, Wash., for its hospitalist coverage. “We have an excellent working relationship with Sound at the local, regional, and national levels, with common goals for quality and utilization. We tried to involve our hospitalists as early as possible in planning. We needed them to step in and role model and lead the way,” Dr. Harte said, for everybody’s anxiety levels.
“We’re still in the process of understanding the long-term financial impact of the epidemic,” Dr. Harte added. “But at this point I see no reason to think our relationship with our hospitalists needs to change. We’re the stewards of long-term finances. We’ll need to keep a close eye on this. But we’re committed to working through this together.”
Hazard pay for frontline health care workers was included in the COVID-19 relief package assembled in mid-May by Democrats in the House of Representatives. The $3 trillion HEROES Act includes $200 billion to award hazard pay to essential workers, including those in the health field, but Senate Majority Leader Mitch McConnell (R-Ky.) declared the legislation “dead on arrival” in the Senate.
Supplementary hazard payments made by hospitals to their hospitalists as a reward for sacrifices they made in the crisis is an interesting question, Dr. Greeno noted, and it’s definitely on the table at some hospitals. “But I think it is going to be a tough ask in these times.”
Dr. Harte said he has not offered nor been asked about hazard pay for hospitalists. Cleveland Clinic Akron General made a strategic decision that hazard pay was not going to be part of its response to the pandemic. Other hospital administrators interviewed for this article concur.
Hospitals respond to the fiscal crisis
Hospitals in other parts of the country also report significant fiscal fallout from the COVID-19 crisis, with predictions that 100 or more hospitals may be forced to close. Jeff Dye, president of the New Mexico Hospital Association, told the Albuquerque Journal on May 1 that hospitals in his state have been squeezed on all sides by increased costs, patients delaying routine care, and public health orders restricting elective surgeries. New Mexico hospitals, especially in rural areas, face incredible financial strain.
The University of Virginia Medical Center, Charlottesville, recently announced 20% reductions in total compensation for its providers through July 31, along with suspension of retirement contributions. Those changes won’t affect team members caring for COVID-19 patients. And the Spectrum Health Medical Group of 15 hospitals in western Michigan, according to Michigan Public Radio, told its doctors they either needed to sign “contract addendums” giving the system more control over their hours – or face a 25% pay cut, or worse.
Cheyenne (Wyo.) Regional Medical Center issued a statement April 24 that it expected losses of $10 million for the month of April. “CRMC, like every other hospital in Wyoming, is certainly feeling the financial impact that COVID-19 is having,” CEO Tim Thornell told the Cowboy State Daily on April 24. That includes a 30% reduction in inpatient care and 50% reduction in outpatient care, while the hospital has only had a handful of COVID-19 patients at any time. Capital projects are now on hold, overtime is limited, and a hiring freeze is in effect.
“We’re certainly prepared for a larger surge, which hasn’t come yet,” Mr. Thornell said in an interview. CRMC’s ICU was split to create a nine-bed dedicated COVID-19 unit. Intensivists see most of the critical care patients, while the hospital’s 15 directly-employed hospitalists are treating all of the non-ICU COVID-19 patients. “Among themselves, the hospitalists volunteered who would work on the unit. We’ve been fortunate enough to have enough volunteers and enough PPE [personal protective equipment],” he said.
Preparing for the COVID-19 pandemic has strengthened the medical center’s relationship with its hospitalists, Mr. Thornell explained. “Hospitalists are key to our operations, involved in so much that happens here. We’re trying to staff to volume with decreased utilization. We’ve scaled back, which only makes fiscal sense. Now, how do we reinfuse patients back into the mix? Our hospitalists are paid by the number of shifts, and as you distribute shift reductions over 15 providers, it shouldn’t be an intolerable burden.” But two open hospitalist positions have not been filled, he noted.
CRMC is trying to approach these changes with a Lean perspective, Mr. Thornell said. “We had already adopted a Lean program, but this has been a chance to go through a life-altering circumstance using the tools of Lean planning and applying them instantaneously.”
Providers step up
At Emory Healthcare in Atlanta, a major center for COVID-19 cases, communication has been essential in the crisis, said Bryce Gartland, MD, SFHM, Emory’s hospital group president and cochief of clinical operations. “Our group was prepared for a significant influx of patients. Like every other institution, we made the decision to postpone elective care, with a resulting plummet in volume,” he said.
As COVID-19 patients entered the Emory system, frontline hospitalists stepped up to care for those patients. “We’ve had ample providers in terms of clinical care. We guaranteed our physicians’ base compensation. They have flexed teams up and down as needed.” Advanced practice professionals also stepped up to bridge gaps.
With regard to the return of volumes of non–COVID-19 patients, the jury’s still out, Dr. Gartland said. “None of us has a crystal ball, and there are tremendous variables and decision points that will have significant impact. We have started to see numbers of time-sensitive and essential cases increase as of the first week of May.”
What lies ahead will likely include some rightsizing to future volumes. On top of that, the broader economic pressures on hospitals from high rates of unemployment, uninsured patients, bad debt, and charity care will push health care systems to significantly address costs and infrastructure, he said. “We’re still early in planning, and striving to maintain flexibility and nimbleness, given the uncertainties to this early understanding of our new normal. No hospital is immune from the financial impact. We’ll see and hear about more of these conversations in the months ahead.”
But the experience has also generated some positives, Dr. Gartland noted. “Things like telehealth, which we’ve been talking about for years but previously faced barriers to widespread adoption.” Now with COVID-19, the federal government issued waivers, and barriers – both internal and external – came down. “With telehealth, what will the role and deployment of hospitalists look like in this new model? How will traditional productivity expectations change, or the numbers and types of providers? This will make the relationship and partnership between hospitalist groups and hospital administrators ever more important as we consider the evolution toward new care models.”
Dr. Gartland said that “one of the great things about hospital medicine as a field is its flexibility and adaptability. Where there have been gaps, hospitalists were quick to step in. As long as hospital medicine continues to embrace those kinds of behaviors, it will be successful.” But if the conversation with hospitals is just about money, it will be harder, he acknowledged. “Where there is this kind of disruption in our usual way of doing things, there are also tremendous opportunities for care model innovation. I would encourage hospitalist groups to try to be true value partners.”
Command center mode
Like other physicians in hospital C-suites, Chad Whelan MD, FACP, SFHM, chief executive officer of Banner–University Medicine in Tucson, Ariz., led his two hospitals into command center mode when the crisis hit, planning for a surge of COVID-19 cases that could overwhelm hospital capacity.
“In terms of our hospitalists, we leaned in to them hard in the beginning, preparing them to supervise other physicians who came in to help if needed,” he said. “Our [non–COVID-19] census is down, revenues are down, and the implications are enormous – like nothing we’ve ever seen before.”
“We’re fortunate that we’re part of the Banner health system. We made a decision that we would essentially keep our physicians financially protected through this crisis,” Dr. Whelan said. “In return, we called on them to step up and be on the front lines and to put in enormous hours for planning. We asked them to consider: How could you contribute if the surge comes?”
He affirmed that hospital medicine has been a major part of his medical center’s planning and implementation. “I’ve been overwhelmed by the degree to which the entire delivery team has rallied around the pandemic, with everybody saying they want to keep people safe and be part of the solution. We have always had hospitalist leaders at the table as we’ve planned our response and as decisions were made,” said Dr. Whelan, a practicing hospitalist and teaching service attending since 2000 until he assumed his current executive position in Arizona 18 months ago.
“While we have kept people whole during the immediate crisis, we have acknowledged that we don’t know what our recovery will look like. What if [non–COVID-19] volume doesn’t return? That keeps me awake at night,” he said. “I have talked to our physician leadership in hospital medicine and more broadly. We need to ask ourselves many questions, including: do we have the right levels of staffing? Is this the time to consider alternate models of staffing, for example, advanced practice providers? And does the compensation plan need adjustments?”
Dr. Whelan thinks that the COVID-19 crisis is an opportunity for hospital medicine to more rapidly explore different models and to ask what additional value hospitalists can bring to the care model. “For example, what would it mean to redefine the hospitalist’s scope of practice as an acute medicine specialist, not defined by the hospital’s four walls?” he noted.
“One of the reasons our smaller hospital reached capacity with COVID-19 patients was the skilled nursing facility located a few hundred feet away that turned into a hot spot. If we had imported the hospital medicine model virtually into that SNF early on, could there have been a different scenario? Have we thought through what that would have even looked like?” Dr. Whelan asked.
He challenges the hospital medicine field, once it gets to the other side of this crisis, to not fall back on old way of doing things. “Instead, let’s use this time to create a better model today,” he said. “That’s what we’re trying to do at a system level at Banner, with our hospital medicine groups partnering with the hospital. I want to see our hospitalists create and thrive in that new model.”
Oleander extract for COVID-19? That’s a hard ‘no’ experts say
“Though renowned for its beauty and use in landscaping, this Mediterranean shrub is responsible for cases of accidental poisoning across the globe. All parts of the plant are poisonous,” Cassandra Quave, PhD, ethnobotanist and herbarium curator at Emory University, Atlanta, cautioned in an article in The Conversation, an independent, not-for-profit publication.
Oleandrin has properties similar to digoxin; the onset of toxicity occurs several hours after consumption.
The first symptoms of oleandrin poisoning may be gastrointestinal, such as nausea, vomiting, abdominal pain, diarrhea (which may contain blood), and loss of appetite.
After these first symptoms, the heart may be affected by tachyarrhythmia, bradyarrhythmia, premature ventricular contractions, or atrioventricular blockage. Xanthopsia (yellow vision), a burning sensation in the eyes, paralysis of the gastrointestinal tract, and respiratory symptoms may also occur.
Oleandrin poisoning may affect the central nervous system, as evidenced by drowsiness, tremors, seizures, collapse, and coma leading to death. When applied to the skin, oleander sap can cause skin irritations and allergic reactions characterized by dermatitis.
Diagnosis of oleandrin poisoning is mainly made on the basis of a description of the plant, how much of it was ingested, how much time has elapsed since ingestion, and symptoms. Confirmation of oleandrin in blood involves fluorescence polarization immunoassay, digoxin immunoassay, or liquid chromatography-electrospray tandem mass spectrometry.
Neither oleander nor oleandrin is approved by regulatory agencies as a prescription drug or dietary supplement.
In vitro study
Oleandrin for COVID-19 made headlines after President Trump met in the Oval Office with Andrew Whitney, vice chairman and director of Phoenix Biotechnology, along with Housing and Urban Development Secretary Ben Carson, MD, and MyPillow founder/CEO Mike Lindell, a strong supporter of Trump and an investor in the biotech company, to learn about oleandrin, which Whitney called a “cure” for COVID-19, Axios reported.
In an in vitro study, researchers from Phoenix Biotechnology and the University of Texas Medical Branch, Galveston, tested oleandrin against SARS-CoV-2 in cultured Vero cells.
“When administered both before and after virus infection, nanogram doses of oleandrin significantly inhibited replication by 45 to 3000-fold,” the researchers said in an article posted on bioRxiv, a free online archive and distribution service for unpublished preprints in the life sciences. The study has not been peer reviewed.
On the basis of these in vitro findings, the researchers said the plant extract has “potential to prevent disease and virus spread in persons recently exposed to SARS-CoV-2, as well as to prevent severe disease in persons at high risk.”
But it’s a far cry from test tube to human, one expert cautioned.
“This is an understatement: Care must be taken when inferring potential therapeutic benefits from in vitro antiviral effects,” Harlan Krumholz, MD, cardiologist and director, Yale New Haven Hospital Center for Outcomes Research and Evaluation, New Haven, Connecticut, told Medscape Medical News.
“There is a chasm between a single in vitro study and any use in humans outside of a protocol. People should be cautioned about that distance and the need [to] avoid such remedies unless part of a credible research project,” said Krumholz.
Yet Lindell told Axios that, in the Oval Office meeting, Trump expressed enthusiasm for the Food and Drug Administration to allow oleandrin to be marketed as a dietary supplement or approved for COVID-19.
“This is really just nonsense and a distraction,” Jonathan Reiner, MD, of George Washington University Medical Center, Washington, DC, said on CNN.
This article first appeared on Medscape.com.
“Though renowned for its beauty and use in landscaping, this Mediterranean shrub is responsible for cases of accidental poisoning across the globe. All parts of the plant are poisonous,” Cassandra Quave, PhD, ethnobotanist and herbarium curator at Emory University, Atlanta, cautioned in an article in The Conversation, an independent, not-for-profit publication.
Oleandrin has properties similar to digoxin; the onset of toxicity occurs several hours after consumption.
The first symptoms of oleandrin poisoning may be gastrointestinal, such as nausea, vomiting, abdominal pain, diarrhea (which may contain blood), and loss of appetite.
After these first symptoms, the heart may be affected by tachyarrhythmia, bradyarrhythmia, premature ventricular contractions, or atrioventricular blockage. Xanthopsia (yellow vision), a burning sensation in the eyes, paralysis of the gastrointestinal tract, and respiratory symptoms may also occur.
Oleandrin poisoning may affect the central nervous system, as evidenced by drowsiness, tremors, seizures, collapse, and coma leading to death. When applied to the skin, oleander sap can cause skin irritations and allergic reactions characterized by dermatitis.
Diagnosis of oleandrin poisoning is mainly made on the basis of a description of the plant, how much of it was ingested, how much time has elapsed since ingestion, and symptoms. Confirmation of oleandrin in blood involves fluorescence polarization immunoassay, digoxin immunoassay, or liquid chromatography-electrospray tandem mass spectrometry.
Neither oleander nor oleandrin is approved by regulatory agencies as a prescription drug or dietary supplement.
In vitro study
Oleandrin for COVID-19 made headlines after President Trump met in the Oval Office with Andrew Whitney, vice chairman and director of Phoenix Biotechnology, along with Housing and Urban Development Secretary Ben Carson, MD, and MyPillow founder/CEO Mike Lindell, a strong supporter of Trump and an investor in the biotech company, to learn about oleandrin, which Whitney called a “cure” for COVID-19, Axios reported.
In an in vitro study, researchers from Phoenix Biotechnology and the University of Texas Medical Branch, Galveston, tested oleandrin against SARS-CoV-2 in cultured Vero cells.
“When administered both before and after virus infection, nanogram doses of oleandrin significantly inhibited replication by 45 to 3000-fold,” the researchers said in an article posted on bioRxiv, a free online archive and distribution service for unpublished preprints in the life sciences. The study has not been peer reviewed.
On the basis of these in vitro findings, the researchers said the plant extract has “potential to prevent disease and virus spread in persons recently exposed to SARS-CoV-2, as well as to prevent severe disease in persons at high risk.”
But it’s a far cry from test tube to human, one expert cautioned.
“This is an understatement: Care must be taken when inferring potential therapeutic benefits from in vitro antiviral effects,” Harlan Krumholz, MD, cardiologist and director, Yale New Haven Hospital Center for Outcomes Research and Evaluation, New Haven, Connecticut, told Medscape Medical News.
“There is a chasm between a single in vitro study and any use in humans outside of a protocol. People should be cautioned about that distance and the need [to] avoid such remedies unless part of a credible research project,” said Krumholz.
Yet Lindell told Axios that, in the Oval Office meeting, Trump expressed enthusiasm for the Food and Drug Administration to allow oleandrin to be marketed as a dietary supplement or approved for COVID-19.
“This is really just nonsense and a distraction,” Jonathan Reiner, MD, of George Washington University Medical Center, Washington, DC, said on CNN.
This article first appeared on Medscape.com.
“Though renowned for its beauty and use in landscaping, this Mediterranean shrub is responsible for cases of accidental poisoning across the globe. All parts of the plant are poisonous,” Cassandra Quave, PhD, ethnobotanist and herbarium curator at Emory University, Atlanta, cautioned in an article in The Conversation, an independent, not-for-profit publication.
Oleandrin has properties similar to digoxin; the onset of toxicity occurs several hours after consumption.
The first symptoms of oleandrin poisoning may be gastrointestinal, such as nausea, vomiting, abdominal pain, diarrhea (which may contain blood), and loss of appetite.
After these first symptoms, the heart may be affected by tachyarrhythmia, bradyarrhythmia, premature ventricular contractions, or atrioventricular blockage. Xanthopsia (yellow vision), a burning sensation in the eyes, paralysis of the gastrointestinal tract, and respiratory symptoms may also occur.
Oleandrin poisoning may affect the central nervous system, as evidenced by drowsiness, tremors, seizures, collapse, and coma leading to death. When applied to the skin, oleander sap can cause skin irritations and allergic reactions characterized by dermatitis.
Diagnosis of oleandrin poisoning is mainly made on the basis of a description of the plant, how much of it was ingested, how much time has elapsed since ingestion, and symptoms. Confirmation of oleandrin in blood involves fluorescence polarization immunoassay, digoxin immunoassay, or liquid chromatography-electrospray tandem mass spectrometry.
Neither oleander nor oleandrin is approved by regulatory agencies as a prescription drug or dietary supplement.
In vitro study
Oleandrin for COVID-19 made headlines after President Trump met in the Oval Office with Andrew Whitney, vice chairman and director of Phoenix Biotechnology, along with Housing and Urban Development Secretary Ben Carson, MD, and MyPillow founder/CEO Mike Lindell, a strong supporter of Trump and an investor in the biotech company, to learn about oleandrin, which Whitney called a “cure” for COVID-19, Axios reported.
In an in vitro study, researchers from Phoenix Biotechnology and the University of Texas Medical Branch, Galveston, tested oleandrin against SARS-CoV-2 in cultured Vero cells.
“When administered both before and after virus infection, nanogram doses of oleandrin significantly inhibited replication by 45 to 3000-fold,” the researchers said in an article posted on bioRxiv, a free online archive and distribution service for unpublished preprints in the life sciences. The study has not been peer reviewed.
On the basis of these in vitro findings, the researchers said the plant extract has “potential to prevent disease and virus spread in persons recently exposed to SARS-CoV-2, as well as to prevent severe disease in persons at high risk.”
But it’s a far cry from test tube to human, one expert cautioned.
“This is an understatement: Care must be taken when inferring potential therapeutic benefits from in vitro antiviral effects,” Harlan Krumholz, MD, cardiologist and director, Yale New Haven Hospital Center for Outcomes Research and Evaluation, New Haven, Connecticut, told Medscape Medical News.
“There is a chasm between a single in vitro study and any use in humans outside of a protocol. People should be cautioned about that distance and the need [to] avoid such remedies unless part of a credible research project,” said Krumholz.
Yet Lindell told Axios that, in the Oval Office meeting, Trump expressed enthusiasm for the Food and Drug Administration to allow oleandrin to be marketed as a dietary supplement or approved for COVID-19.
“This is really just nonsense and a distraction,” Jonathan Reiner, MD, of George Washington University Medical Center, Washington, DC, said on CNN.
This article first appeared on Medscape.com.
Non-COVID-19 clinical trials grind to a halt during pandemic
The COVID-19 pandemic has created unique and unprecedented challenges for the clinical research world, with potentially long-lasting consequences.
A new analysis of the extent of disruption shows that the average rate of stopped trials nearly doubled during the first 5 months of 2020, compared with the 2 previous years.
“Typically, clinical research precedes clinical practice by several years, so this disruption we’re seeing now will be felt for many years to come,” said Mario Guadino, MD, of Weill Cornell Medicine, New York.
The analysis was published online July 31 in the Journal of the American College of Cardiology.
The researchers used Python software to query meta-data from all trials reported on ClinicalTrials.gov. Of 321,218 non-COVID-19 trials queried, 28,672 (8.9%) were reported as stopped, defined as a switch in trial status from “recruiting” to “active and not recruiting,” “completed,” “suspended,” “terminated,” or “withdrawn.”
The average rate of discontinuation was 638 trials/month from January 2017 to December 2019, rising to 1,147 trials/month between January 2020 and May 2020 (P < .001 for trend).
Once stopped (as opposed to paused), restarting a trial is a tricky prospect, said Dr. Guadino. “You can’t stop and restart a trial because it creates a lot of issues, so we should expect many of these stopped trials to never be completed.”
He said these figures likely represent an underestimate of the true impact of the pandemic because there is typically a delay in the updating of the status of a trial on ClinicalTrials.gov.
“We are likely looking only at the tip of the iceberg,” he added. “My impression is that the number of trials that will be affected and even canceled will be very high.”
As for cardiology trials, one of the report’s authors, Deepak Bhatt, MD, Brigham and Women’s Hospital, Boston, without naming specific trials, had this to say: “Several cardiovascular trials were paused, and some were permanently discontinued. It may be a while before we fully appreciate just how much information was lost and how much might be salvaged.”
He’s not worried, however, that upcoming cardiology meetings, which have moved online for the foreseeable future, might get a bit boring. “Fortunately, there is enough good work going on in the cardiovascular and cardiometabolic space that I believe there will still be ample randomized and observational data of high quality to present at the major meetings,” Dr. Bhatt said in an email.
The researchers found a weak correlation between the national population-adjusted numbers of COVID-19 cases and the proportion of non-COVID-19 trials stopped by country.
Even for trials that stopped recruiting for a period of time but are continuing, there are myriad issues involving compliance, data integrity, statistical interpretability, etc.
“Even if there is just a temporary disruption, that will most likely lead to reduced enrollment, missing follow-up visits, and protocol deviations, all things that would be red flags during normal times and impact the quality of the clinical trial,” said Dr. Guadino.
“And if your outcome of interest is mortality, well, how exactly do you measure that during a pandemic?” he added.
Stopped for lack of funding
Besides the logistical issues, another reason trials may be in jeopardy is funding. A warning early in the pandemic from the research community in Canada that funding was quickly drying up, leaving both jobs and data at risk, led to an aid package from the government to keep the lights on.
The National Institutes of Health (NIH), the Canadian Institutes of Health Research, and similar groups “have devoted large sums of money to research in COVID, which is of course very appropriate, but that clearly reduces the amount of funding that is available for other researchers,” said Dr. Guadino.
Some funding agencies around the world have canceled or put on hold all non-COVID-19 clinical trials still at the design state, Dr. Guadino said in an interview.
The NIH, he stressed, has not canceled funding and has been “extremely open and cooperative” in trying to help trialists navigate the many COVID-generated issues. They’ve even issued guidance on how to manage trials during COVID-19.
Of note, in the survey, the majority of the trials stopped (95.4%) had nongovernmental funding.
“The data are not very granular, so we’re only able to make some very simple, descriptive comments, but it does seem like the more fragile trials – those that are smaller and industry-funded – are the ones more likely to be disrupted,” said Dr. Guadino.
In some cases, he said, priorities have shifted to COVID-19. “If a small company is sponsoring a trial and they decide they want to sponsor something related to COVID, or they realize that because of the slow enrollment, the trial becomes too expensive to complete, they may opt to just abandon it,” said Dr. Guadino.
At what cost? It will take years to sort that out, he said.
This study received no funding. Dr. Guadino and Dr. Bhatt are both active trialists, participating in both industry- and government-sponsored clinical research.
A version of this article originally appeared on Medscape.com.
The COVID-19 pandemic has created unique and unprecedented challenges for the clinical research world, with potentially long-lasting consequences.
A new analysis of the extent of disruption shows that the average rate of stopped trials nearly doubled during the first 5 months of 2020, compared with the 2 previous years.
“Typically, clinical research precedes clinical practice by several years, so this disruption we’re seeing now will be felt for many years to come,” said Mario Guadino, MD, of Weill Cornell Medicine, New York.
The analysis was published online July 31 in the Journal of the American College of Cardiology.
The researchers used Python software to query meta-data from all trials reported on ClinicalTrials.gov. Of 321,218 non-COVID-19 trials queried, 28,672 (8.9%) were reported as stopped, defined as a switch in trial status from “recruiting” to “active and not recruiting,” “completed,” “suspended,” “terminated,” or “withdrawn.”
The average rate of discontinuation was 638 trials/month from January 2017 to December 2019, rising to 1,147 trials/month between January 2020 and May 2020 (P < .001 for trend).
Once stopped (as opposed to paused), restarting a trial is a tricky prospect, said Dr. Guadino. “You can’t stop and restart a trial because it creates a lot of issues, so we should expect many of these stopped trials to never be completed.”
He said these figures likely represent an underestimate of the true impact of the pandemic because there is typically a delay in the updating of the status of a trial on ClinicalTrials.gov.
“We are likely looking only at the tip of the iceberg,” he added. “My impression is that the number of trials that will be affected and even canceled will be very high.”
As for cardiology trials, one of the report’s authors, Deepak Bhatt, MD, Brigham and Women’s Hospital, Boston, without naming specific trials, had this to say: “Several cardiovascular trials were paused, and some were permanently discontinued. It may be a while before we fully appreciate just how much information was lost and how much might be salvaged.”
He’s not worried, however, that upcoming cardiology meetings, which have moved online for the foreseeable future, might get a bit boring. “Fortunately, there is enough good work going on in the cardiovascular and cardiometabolic space that I believe there will still be ample randomized and observational data of high quality to present at the major meetings,” Dr. Bhatt said in an email.
The researchers found a weak correlation between the national population-adjusted numbers of COVID-19 cases and the proportion of non-COVID-19 trials stopped by country.
Even for trials that stopped recruiting for a period of time but are continuing, there are myriad issues involving compliance, data integrity, statistical interpretability, etc.
“Even if there is just a temporary disruption, that will most likely lead to reduced enrollment, missing follow-up visits, and protocol deviations, all things that would be red flags during normal times and impact the quality of the clinical trial,” said Dr. Guadino.
“And if your outcome of interest is mortality, well, how exactly do you measure that during a pandemic?” he added.
Stopped for lack of funding
Besides the logistical issues, another reason trials may be in jeopardy is funding. A warning early in the pandemic from the research community in Canada that funding was quickly drying up, leaving both jobs and data at risk, led to an aid package from the government to keep the lights on.
The National Institutes of Health (NIH), the Canadian Institutes of Health Research, and similar groups “have devoted large sums of money to research in COVID, which is of course very appropriate, but that clearly reduces the amount of funding that is available for other researchers,” said Dr. Guadino.
Some funding agencies around the world have canceled or put on hold all non-COVID-19 clinical trials still at the design state, Dr. Guadino said in an interview.
The NIH, he stressed, has not canceled funding and has been “extremely open and cooperative” in trying to help trialists navigate the many COVID-generated issues. They’ve even issued guidance on how to manage trials during COVID-19.
Of note, in the survey, the majority of the trials stopped (95.4%) had nongovernmental funding.
“The data are not very granular, so we’re only able to make some very simple, descriptive comments, but it does seem like the more fragile trials – those that are smaller and industry-funded – are the ones more likely to be disrupted,” said Dr. Guadino.
In some cases, he said, priorities have shifted to COVID-19. “If a small company is sponsoring a trial and they decide they want to sponsor something related to COVID, or they realize that because of the slow enrollment, the trial becomes too expensive to complete, they may opt to just abandon it,” said Dr. Guadino.
At what cost? It will take years to sort that out, he said.
This study received no funding. Dr. Guadino and Dr. Bhatt are both active trialists, participating in both industry- and government-sponsored clinical research.
A version of this article originally appeared on Medscape.com.
The COVID-19 pandemic has created unique and unprecedented challenges for the clinical research world, with potentially long-lasting consequences.
A new analysis of the extent of disruption shows that the average rate of stopped trials nearly doubled during the first 5 months of 2020, compared with the 2 previous years.
“Typically, clinical research precedes clinical practice by several years, so this disruption we’re seeing now will be felt for many years to come,” said Mario Guadino, MD, of Weill Cornell Medicine, New York.
The analysis was published online July 31 in the Journal of the American College of Cardiology.
The researchers used Python software to query meta-data from all trials reported on ClinicalTrials.gov. Of 321,218 non-COVID-19 trials queried, 28,672 (8.9%) were reported as stopped, defined as a switch in trial status from “recruiting” to “active and not recruiting,” “completed,” “suspended,” “terminated,” or “withdrawn.”
The average rate of discontinuation was 638 trials/month from January 2017 to December 2019, rising to 1,147 trials/month between January 2020 and May 2020 (P < .001 for trend).
Once stopped (as opposed to paused), restarting a trial is a tricky prospect, said Dr. Guadino. “You can’t stop and restart a trial because it creates a lot of issues, so we should expect many of these stopped trials to never be completed.”
He said these figures likely represent an underestimate of the true impact of the pandemic because there is typically a delay in the updating of the status of a trial on ClinicalTrials.gov.
“We are likely looking only at the tip of the iceberg,” he added. “My impression is that the number of trials that will be affected and even canceled will be very high.”
As for cardiology trials, one of the report’s authors, Deepak Bhatt, MD, Brigham and Women’s Hospital, Boston, without naming specific trials, had this to say: “Several cardiovascular trials were paused, and some were permanently discontinued. It may be a while before we fully appreciate just how much information was lost and how much might be salvaged.”
He’s not worried, however, that upcoming cardiology meetings, which have moved online for the foreseeable future, might get a bit boring. “Fortunately, there is enough good work going on in the cardiovascular and cardiometabolic space that I believe there will still be ample randomized and observational data of high quality to present at the major meetings,” Dr. Bhatt said in an email.
The researchers found a weak correlation between the national population-adjusted numbers of COVID-19 cases and the proportion of non-COVID-19 trials stopped by country.
Even for trials that stopped recruiting for a period of time but are continuing, there are myriad issues involving compliance, data integrity, statistical interpretability, etc.
“Even if there is just a temporary disruption, that will most likely lead to reduced enrollment, missing follow-up visits, and protocol deviations, all things that would be red flags during normal times and impact the quality of the clinical trial,” said Dr. Guadino.
“And if your outcome of interest is mortality, well, how exactly do you measure that during a pandemic?” he added.
Stopped for lack of funding
Besides the logistical issues, another reason trials may be in jeopardy is funding. A warning early in the pandemic from the research community in Canada that funding was quickly drying up, leaving both jobs and data at risk, led to an aid package from the government to keep the lights on.
The National Institutes of Health (NIH), the Canadian Institutes of Health Research, and similar groups “have devoted large sums of money to research in COVID, which is of course very appropriate, but that clearly reduces the amount of funding that is available for other researchers,” said Dr. Guadino.
Some funding agencies around the world have canceled or put on hold all non-COVID-19 clinical trials still at the design state, Dr. Guadino said in an interview.
The NIH, he stressed, has not canceled funding and has been “extremely open and cooperative” in trying to help trialists navigate the many COVID-generated issues. They’ve even issued guidance on how to manage trials during COVID-19.
Of note, in the survey, the majority of the trials stopped (95.4%) had nongovernmental funding.
“The data are not very granular, so we’re only able to make some very simple, descriptive comments, but it does seem like the more fragile trials – those that are smaller and industry-funded – are the ones more likely to be disrupted,” said Dr. Guadino.
In some cases, he said, priorities have shifted to COVID-19. “If a small company is sponsoring a trial and they decide they want to sponsor something related to COVID, or they realize that because of the slow enrollment, the trial becomes too expensive to complete, they may opt to just abandon it,” said Dr. Guadino.
At what cost? It will take years to sort that out, he said.
This study received no funding. Dr. Guadino and Dr. Bhatt are both active trialists, participating in both industry- and government-sponsored clinical research.
A version of this article originally appeared on Medscape.com.
Send kids to school safely if possible, supplement virtually
The abrupt transition to online learning for American children in kindergarten through 12th grade has left educators and parents unprepared, but virtual learning can be a successful part of education going forward, according to a viewpoint published in JAMA Pediatrics. However, schools also can reopen safely if precautions are taken, and students would benefit in many ways, according to a second viewpoint.
“As policy makers, health care professionals, and parents prepare for the fall semester and as public and private schools grapple with how to make that possible, a better understanding of K-12 virtual learning options and outcomes may facilitate those difficult decisions,” wrote Erik Black, PhD, of the University of Florida, Gainesville; Richard Ferdig, PhD, of Kent State University, Ohio; and Lindsay A. Thompson, MD, of the University of Florida, Gainesville.
“Importantly, K-12 virtual schooling is not suited for all students or all families.”
In a viewpoint published in JAMA Pediatrics, the authors noted that virtual schooling has existed in the United States in various forms for some time. “Just like the myriad options that are available for face-to-face schooling in the U.S., virtual schooling exists in a complex landscape of for-profit, charter, and public options.”
Not all virtual schools are equal
Consequently, not all virtual schools are created equal, they emphasized. Virtual education can be successful for many students when presented by trained online instructors using a curriculum designed to be effective in an online venue.
“Parents need to seek reviews and ask for educational outcomes from each virtual school system to assess the quality of the provided education,” Dr. Black, Dr. Ferdig, and Dr. Thompson emphasized.
Key questions for parents to consider when faced with online learning include the type of technology needed to participate; whether their child can maintain a study schedule and complete assignments with limited supervision; whether their child could ask for help and communicate with teachers through technology including phone, text, email, or video; and whether their child has the basic reading, math, and computer literacy skills to engage in online learning, the authors said. Other questions include the school’s expectations for parents and caregivers, how student information may be shared, and how the virtual school lines up with state standards for K-12 educators (in the case of options outside the public school system).
“The COVID-19 pandemic offers a unique challenge for educators, policymakers, and health care professionals to partner with parents to make the best local and individual decisions for children,” Dr. Black, Dr. Ferdig, and Dr. Thompson concluded.
Schools may be able to open safely
Children continue to make up a low percentage of COVID-19 cases and appear less likely to experience illness, wrote C. Jason Wang, MD, PhD, and Henry Bair, BS, of Stanford (Calif.) University in a second viewpoint also published in JAMA Pediatrics. The impact of long-term school closures extends beyond education and can “exacerbate socioeconomic disparities, amplify existing educational inequalities, and aggravate food insecurity, domestic violence, and mental health disorders,” they wrote.
Dr. Wang and Mr. Bair proposed that school districts “engage key stakeholders to establish a COVID-19 task force, composed of the superintendent, members of the school board, teachers, parents, and health care professionals to develop policies and procedures,” that would allow schools to open safely.
The authors outlined strategies including adapting teaching spaces to accommodate physical distance, with the addition of temporary modular buildings if needed. They advised assigned seating on school buses, and acknowledged the need for the availability of protective equipment, including hand sanitizer and masks, as well as the possible use of transparent barriers on the sides of student desks.
“As the AAP [American Academy of Pediatrics] guidance suggests, teachers who must work closely with students with special needs or with students who are unable to wear masks should wear N95 masks if possible or wear face shields in addition to surgical masks,” Dr. Wang and Mr. Bair noted. Other elements of the AAP guidance include the creation of fixed cohorts of students and teachers to limit virus exposure.
“Even with all the precautions in place, COVID-19 outbreaks within schools are still likely,” they said. “Therefore, schools will need to remain flexible and consider temporary closures if there is an outbreak involving multiple students and/or staff and be ready to transition to online education.”
The AAP guidance does not address operational approaches to identifying signs and symptoms of COVID-19, the authors noted. “To address this, we recommend that schools implement multilevel screening for students and staff.”
“In summary, to maximize health and educational outcomes, school districts should adopt some or all of the measures of the AAP guidance and prioritize them after considering local COVID-19 incidence, key stakeholder input, and budgetary constraints,” Dr. Wang and Mr. Bair concluded.
Schools opening is a regional decision
“The mission of the AAP is to attain optimal physical, mental, and social health and well-being for all infants, children, adolescents, and young adults,” Howard Smart, MD, said in an interview. The question of school reopening “is of national importance, and the AAP has a national role in making recommendations regarding national policy affecting the health of the children.”
“The decision to open schools will be made regionally, but it is important for a nonpolitical national voice to make expert recommendations,” he emphasized.
“Many of the recommendations are ideal goals,” noted Dr. Smart, chairman of the department of pediatrics at the Sharp Rees-Stealy Medical Group in San Diego. “It will be difficult, for example, to implement symptom screening every day before school, no matter where it is performed. Some of the measures may be quite costly, and take time to implement, or require expansion of school staff, for which there may be no budget.”
In addition, “[n]ot all students are likely to comply with masking, distance, and hand-washing recommendations. One student who is noncompliant will be able to infect many other students and staff, as has been seen in other countries.” Also, parental attitudes toward control measures are likely to affect student attitudes, he noted.
“I have interviewed many families at recent checkups, and most have felt that the rush to remote learning that occurred at the end of the last school year resulted in fairly disorganized instruction,” Dr. Smart said. “They are hoping that, having had the summer to plan ahead, the remote teaching will be handled better. Remote learning will certainly work best for self-motivated, organized students with good family support, as noted in the Black, Ferdig, and Thompson article,” he said.
Pediatricians can support the schools by being a source of evidence-based information for parents, Dr. Smart said. “Pediatricians with time and energy might want to volunteer to hold informational video conferences for parents and/or school personnel if they feel they are up to date on current COVID-19 science and want to handle potentially contentious questions.”
The decision parents make to send their children back to school comes down to a risk-benefit calculation. “In some communities this may be left to parents, while in other communities this will a public health decision,” he said. “It is still not clear whether having students attend school in person will result in increased spread of COVID-19 among the students, or in their communities. Although some evidence from early in the pandemic suggests that children may not spread the virus as much as adults, more recent evidence suggests that children 10 years and older do transmit the virus at least as much as adults.”
“The risk to the students and the community, therefore, is unknown,” and difficult to compare with the benefit of in-person schooling, Dr. Smart noted.
“We will learn quite a bit from communities where students do go back to in-person class, as we follow the progression of COVID-19 over the weeks following the resumption of instruction.” Ultimately, advice to parents will need to be tailored to the current conditions of COVID-19 transmission in the community, he concluded.
It’s not just about education
“The AAP released its guidance to ensure that as school districts were contemplating reopening they were considering the full array of risks for children and adolescents. These risks included not only those related to COVID-19, but also those related to the impact of not reopening in-person,” Nathaniel Beers, MD, president of the HSC Health Care System in Washington, said in an interview.
“Students and families are dependent on schools for much more than just an education, and those [elements] need to be factored into the decisions to reopen,” the pediatrician said.
However, “[t]he major barrier for schools is resources to safely reopen,” said Dr. Beers. “The additional staffing and supplies will require additional funding. There are increased demands regardless of whether students are learning in-person or virtually or through hybrid models.”
“Another significant barrier is ensuring that parents and staff are actively engaged in planning for the type of model being used,” he said.
“All of the models require buy-in by staff and parents. This will require significant outreach and strong communication plans. Schools also need to ensure they are planning not just for how to return students to schools, but what will happen when staff or students test positive for COVID-19. Students, families, and staff all will need to know what these plans are up front to feel confident in returning to school,” he emphasized.
“There are students who can thrive in a virtual learning environment,” Dr. Beers said. “There are also students who benefit from the virtual learning environment because of their own risk, or because of a family member’s risk for COVID-19 or the complications from it.”
“However, many children with disabilities have struggled in a virtual environment,” he said. “These students struggle to access the educational services without the adequate supports at home. They often receive additional services in school, such as speech, occupational therapy or physical therapy, or nursing services, that may not have transitioned to home but are critical for their health and development. Many students with disabilities are dependent on family members to successfully access the educational services they need.”
“Pediatricians can play a role in providing feedback on recommendations related to physical distancing and face coverings in particular,” said Dr. Beers. “In addition, they can be helpful in developing plans for children with disabilities as well as what the response plan should be for students who become sick during the school day.”
The Centers for Disease Control and Prevention released a decision tool for parents who are considering whether to send their child to in-person school, and pediatricians can help parents walk through these questions, Dr. Beers noted. “In addition, pediatricians play an important role in helping patients and families think about the risks of COVID for the patient and other family members, and this can be helpful in addressing the anxiety that parents and patients may be experiencing.”
Further information can be found in Return to School During COVID-19, which can be located at HealthyChildren.org, by the American Academy of Pediatrics.
The authors of the viewpoints had no relevant financial disclosures. Dr. Smart, a member of the Pediatric News editorial advisory board, had no relevant financial disclosures. Dr. Beers has served on the editorial advisory board of Pediatric News in the past, but had no relevant financial disclosures.
SOURCES: Black E, Ferdig R, Thompson LA. JAMA Pediatr. 2020 Aug 11. doi: 10.1001/jamapediatrics.2020.3800. Wang CJ and Bair H. JAMA Pediatr. Aug 11. doi: 10.1001/jamapediatrics.2020.3871.
The abrupt transition to online learning for American children in kindergarten through 12th grade has left educators and parents unprepared, but virtual learning can be a successful part of education going forward, according to a viewpoint published in JAMA Pediatrics. However, schools also can reopen safely if precautions are taken, and students would benefit in many ways, according to a second viewpoint.
“As policy makers, health care professionals, and parents prepare for the fall semester and as public and private schools grapple with how to make that possible, a better understanding of K-12 virtual learning options and outcomes may facilitate those difficult decisions,” wrote Erik Black, PhD, of the University of Florida, Gainesville; Richard Ferdig, PhD, of Kent State University, Ohio; and Lindsay A. Thompson, MD, of the University of Florida, Gainesville.
“Importantly, K-12 virtual schooling is not suited for all students or all families.”
In a viewpoint published in JAMA Pediatrics, the authors noted that virtual schooling has existed in the United States in various forms for some time. “Just like the myriad options that are available for face-to-face schooling in the U.S., virtual schooling exists in a complex landscape of for-profit, charter, and public options.”
Not all virtual schools are equal
Consequently, not all virtual schools are created equal, they emphasized. Virtual education can be successful for many students when presented by trained online instructors using a curriculum designed to be effective in an online venue.
“Parents need to seek reviews and ask for educational outcomes from each virtual school system to assess the quality of the provided education,” Dr. Black, Dr. Ferdig, and Dr. Thompson emphasized.
Key questions for parents to consider when faced with online learning include the type of technology needed to participate; whether their child can maintain a study schedule and complete assignments with limited supervision; whether their child could ask for help and communicate with teachers through technology including phone, text, email, or video; and whether their child has the basic reading, math, and computer literacy skills to engage in online learning, the authors said. Other questions include the school’s expectations for parents and caregivers, how student information may be shared, and how the virtual school lines up with state standards for K-12 educators (in the case of options outside the public school system).
“The COVID-19 pandemic offers a unique challenge for educators, policymakers, and health care professionals to partner with parents to make the best local and individual decisions for children,” Dr. Black, Dr. Ferdig, and Dr. Thompson concluded.
Schools may be able to open safely
Children continue to make up a low percentage of COVID-19 cases and appear less likely to experience illness, wrote C. Jason Wang, MD, PhD, and Henry Bair, BS, of Stanford (Calif.) University in a second viewpoint also published in JAMA Pediatrics. The impact of long-term school closures extends beyond education and can “exacerbate socioeconomic disparities, amplify existing educational inequalities, and aggravate food insecurity, domestic violence, and mental health disorders,” they wrote.
Dr. Wang and Mr. Bair proposed that school districts “engage key stakeholders to establish a COVID-19 task force, composed of the superintendent, members of the school board, teachers, parents, and health care professionals to develop policies and procedures,” that would allow schools to open safely.
The authors outlined strategies including adapting teaching spaces to accommodate physical distance, with the addition of temporary modular buildings if needed. They advised assigned seating on school buses, and acknowledged the need for the availability of protective equipment, including hand sanitizer and masks, as well as the possible use of transparent barriers on the sides of student desks.
“As the AAP [American Academy of Pediatrics] guidance suggests, teachers who must work closely with students with special needs or with students who are unable to wear masks should wear N95 masks if possible or wear face shields in addition to surgical masks,” Dr. Wang and Mr. Bair noted. Other elements of the AAP guidance include the creation of fixed cohorts of students and teachers to limit virus exposure.
“Even with all the precautions in place, COVID-19 outbreaks within schools are still likely,” they said. “Therefore, schools will need to remain flexible and consider temporary closures if there is an outbreak involving multiple students and/or staff and be ready to transition to online education.”
The AAP guidance does not address operational approaches to identifying signs and symptoms of COVID-19, the authors noted. “To address this, we recommend that schools implement multilevel screening for students and staff.”
“In summary, to maximize health and educational outcomes, school districts should adopt some or all of the measures of the AAP guidance and prioritize them after considering local COVID-19 incidence, key stakeholder input, and budgetary constraints,” Dr. Wang and Mr. Bair concluded.
Schools opening is a regional decision
“The mission of the AAP is to attain optimal physical, mental, and social health and well-being for all infants, children, adolescents, and young adults,” Howard Smart, MD, said in an interview. The question of school reopening “is of national importance, and the AAP has a national role in making recommendations regarding national policy affecting the health of the children.”
“The decision to open schools will be made regionally, but it is important for a nonpolitical national voice to make expert recommendations,” he emphasized.
“Many of the recommendations are ideal goals,” noted Dr. Smart, chairman of the department of pediatrics at the Sharp Rees-Stealy Medical Group in San Diego. “It will be difficult, for example, to implement symptom screening every day before school, no matter where it is performed. Some of the measures may be quite costly, and take time to implement, or require expansion of school staff, for which there may be no budget.”
In addition, “[n]ot all students are likely to comply with masking, distance, and hand-washing recommendations. One student who is noncompliant will be able to infect many other students and staff, as has been seen in other countries.” Also, parental attitudes toward control measures are likely to affect student attitudes, he noted.
“I have interviewed many families at recent checkups, and most have felt that the rush to remote learning that occurred at the end of the last school year resulted in fairly disorganized instruction,” Dr. Smart said. “They are hoping that, having had the summer to plan ahead, the remote teaching will be handled better. Remote learning will certainly work best for self-motivated, organized students with good family support, as noted in the Black, Ferdig, and Thompson article,” he said.
Pediatricians can support the schools by being a source of evidence-based information for parents, Dr. Smart said. “Pediatricians with time and energy might want to volunteer to hold informational video conferences for parents and/or school personnel if they feel they are up to date on current COVID-19 science and want to handle potentially contentious questions.”
The decision parents make to send their children back to school comes down to a risk-benefit calculation. “In some communities this may be left to parents, while in other communities this will a public health decision,” he said. “It is still not clear whether having students attend school in person will result in increased spread of COVID-19 among the students, or in their communities. Although some evidence from early in the pandemic suggests that children may not spread the virus as much as adults, more recent evidence suggests that children 10 years and older do transmit the virus at least as much as adults.”
“The risk to the students and the community, therefore, is unknown,” and difficult to compare with the benefit of in-person schooling, Dr. Smart noted.
“We will learn quite a bit from communities where students do go back to in-person class, as we follow the progression of COVID-19 over the weeks following the resumption of instruction.” Ultimately, advice to parents will need to be tailored to the current conditions of COVID-19 transmission in the community, he concluded.
It’s not just about education
“The AAP released its guidance to ensure that as school districts were contemplating reopening they were considering the full array of risks for children and adolescents. These risks included not only those related to COVID-19, but also those related to the impact of not reopening in-person,” Nathaniel Beers, MD, president of the HSC Health Care System in Washington, said in an interview.
“Students and families are dependent on schools for much more than just an education, and those [elements] need to be factored into the decisions to reopen,” the pediatrician said.
However, “[t]he major barrier for schools is resources to safely reopen,” said Dr. Beers. “The additional staffing and supplies will require additional funding. There are increased demands regardless of whether students are learning in-person or virtually or through hybrid models.”
“Another significant barrier is ensuring that parents and staff are actively engaged in planning for the type of model being used,” he said.
“All of the models require buy-in by staff and parents. This will require significant outreach and strong communication plans. Schools also need to ensure they are planning not just for how to return students to schools, but what will happen when staff or students test positive for COVID-19. Students, families, and staff all will need to know what these plans are up front to feel confident in returning to school,” he emphasized.
“There are students who can thrive in a virtual learning environment,” Dr. Beers said. “There are also students who benefit from the virtual learning environment because of their own risk, or because of a family member’s risk for COVID-19 or the complications from it.”
“However, many children with disabilities have struggled in a virtual environment,” he said. “These students struggle to access the educational services without the adequate supports at home. They often receive additional services in school, such as speech, occupational therapy or physical therapy, or nursing services, that may not have transitioned to home but are critical for their health and development. Many students with disabilities are dependent on family members to successfully access the educational services they need.”
“Pediatricians can play a role in providing feedback on recommendations related to physical distancing and face coverings in particular,” said Dr. Beers. “In addition, they can be helpful in developing plans for children with disabilities as well as what the response plan should be for students who become sick during the school day.”
The Centers for Disease Control and Prevention released a decision tool for parents who are considering whether to send their child to in-person school, and pediatricians can help parents walk through these questions, Dr. Beers noted. “In addition, pediatricians play an important role in helping patients and families think about the risks of COVID for the patient and other family members, and this can be helpful in addressing the anxiety that parents and patients may be experiencing.”
Further information can be found in Return to School During COVID-19, which can be located at HealthyChildren.org, by the American Academy of Pediatrics.
The authors of the viewpoints had no relevant financial disclosures. Dr. Smart, a member of the Pediatric News editorial advisory board, had no relevant financial disclosures. Dr. Beers has served on the editorial advisory board of Pediatric News in the past, but had no relevant financial disclosures.
SOURCES: Black E, Ferdig R, Thompson LA. JAMA Pediatr. 2020 Aug 11. doi: 10.1001/jamapediatrics.2020.3800. Wang CJ and Bair H. JAMA Pediatr. Aug 11. doi: 10.1001/jamapediatrics.2020.3871.
The abrupt transition to online learning for American children in kindergarten through 12th grade has left educators and parents unprepared, but virtual learning can be a successful part of education going forward, according to a viewpoint published in JAMA Pediatrics. However, schools also can reopen safely if precautions are taken, and students would benefit in many ways, according to a second viewpoint.
“As policy makers, health care professionals, and parents prepare for the fall semester and as public and private schools grapple with how to make that possible, a better understanding of K-12 virtual learning options and outcomes may facilitate those difficult decisions,” wrote Erik Black, PhD, of the University of Florida, Gainesville; Richard Ferdig, PhD, of Kent State University, Ohio; and Lindsay A. Thompson, MD, of the University of Florida, Gainesville.
“Importantly, K-12 virtual schooling is not suited for all students or all families.”
In a viewpoint published in JAMA Pediatrics, the authors noted that virtual schooling has existed in the United States in various forms for some time. “Just like the myriad options that are available for face-to-face schooling in the U.S., virtual schooling exists in a complex landscape of for-profit, charter, and public options.”
Not all virtual schools are equal
Consequently, not all virtual schools are created equal, they emphasized. Virtual education can be successful for many students when presented by trained online instructors using a curriculum designed to be effective in an online venue.
“Parents need to seek reviews and ask for educational outcomes from each virtual school system to assess the quality of the provided education,” Dr. Black, Dr. Ferdig, and Dr. Thompson emphasized.
Key questions for parents to consider when faced with online learning include the type of technology needed to participate; whether their child can maintain a study schedule and complete assignments with limited supervision; whether their child could ask for help and communicate with teachers through technology including phone, text, email, or video; and whether their child has the basic reading, math, and computer literacy skills to engage in online learning, the authors said. Other questions include the school’s expectations for parents and caregivers, how student information may be shared, and how the virtual school lines up with state standards for K-12 educators (in the case of options outside the public school system).
“The COVID-19 pandemic offers a unique challenge for educators, policymakers, and health care professionals to partner with parents to make the best local and individual decisions for children,” Dr. Black, Dr. Ferdig, and Dr. Thompson concluded.
Schools may be able to open safely
Children continue to make up a low percentage of COVID-19 cases and appear less likely to experience illness, wrote C. Jason Wang, MD, PhD, and Henry Bair, BS, of Stanford (Calif.) University in a second viewpoint also published in JAMA Pediatrics. The impact of long-term school closures extends beyond education and can “exacerbate socioeconomic disparities, amplify existing educational inequalities, and aggravate food insecurity, domestic violence, and mental health disorders,” they wrote.
Dr. Wang and Mr. Bair proposed that school districts “engage key stakeholders to establish a COVID-19 task force, composed of the superintendent, members of the school board, teachers, parents, and health care professionals to develop policies and procedures,” that would allow schools to open safely.
The authors outlined strategies including adapting teaching spaces to accommodate physical distance, with the addition of temporary modular buildings if needed. They advised assigned seating on school buses, and acknowledged the need for the availability of protective equipment, including hand sanitizer and masks, as well as the possible use of transparent barriers on the sides of student desks.
“As the AAP [American Academy of Pediatrics] guidance suggests, teachers who must work closely with students with special needs or with students who are unable to wear masks should wear N95 masks if possible or wear face shields in addition to surgical masks,” Dr. Wang and Mr. Bair noted. Other elements of the AAP guidance include the creation of fixed cohorts of students and teachers to limit virus exposure.
“Even with all the precautions in place, COVID-19 outbreaks within schools are still likely,” they said. “Therefore, schools will need to remain flexible and consider temporary closures if there is an outbreak involving multiple students and/or staff and be ready to transition to online education.”
The AAP guidance does not address operational approaches to identifying signs and symptoms of COVID-19, the authors noted. “To address this, we recommend that schools implement multilevel screening for students and staff.”
“In summary, to maximize health and educational outcomes, school districts should adopt some or all of the measures of the AAP guidance and prioritize them after considering local COVID-19 incidence, key stakeholder input, and budgetary constraints,” Dr. Wang and Mr. Bair concluded.
Schools opening is a regional decision
“The mission of the AAP is to attain optimal physical, mental, and social health and well-being for all infants, children, adolescents, and young adults,” Howard Smart, MD, said in an interview. The question of school reopening “is of national importance, and the AAP has a national role in making recommendations regarding national policy affecting the health of the children.”
“The decision to open schools will be made regionally, but it is important for a nonpolitical national voice to make expert recommendations,” he emphasized.
“Many of the recommendations are ideal goals,” noted Dr. Smart, chairman of the department of pediatrics at the Sharp Rees-Stealy Medical Group in San Diego. “It will be difficult, for example, to implement symptom screening every day before school, no matter where it is performed. Some of the measures may be quite costly, and take time to implement, or require expansion of school staff, for which there may be no budget.”
In addition, “[n]ot all students are likely to comply with masking, distance, and hand-washing recommendations. One student who is noncompliant will be able to infect many other students and staff, as has been seen in other countries.” Also, parental attitudes toward control measures are likely to affect student attitudes, he noted.
“I have interviewed many families at recent checkups, and most have felt that the rush to remote learning that occurred at the end of the last school year resulted in fairly disorganized instruction,” Dr. Smart said. “They are hoping that, having had the summer to plan ahead, the remote teaching will be handled better. Remote learning will certainly work best for self-motivated, organized students with good family support, as noted in the Black, Ferdig, and Thompson article,” he said.
Pediatricians can support the schools by being a source of evidence-based information for parents, Dr. Smart said. “Pediatricians with time and energy might want to volunteer to hold informational video conferences for parents and/or school personnel if they feel they are up to date on current COVID-19 science and want to handle potentially contentious questions.”
The decision parents make to send their children back to school comes down to a risk-benefit calculation. “In some communities this may be left to parents, while in other communities this will a public health decision,” he said. “It is still not clear whether having students attend school in person will result in increased spread of COVID-19 among the students, or in their communities. Although some evidence from early in the pandemic suggests that children may not spread the virus as much as adults, more recent evidence suggests that children 10 years and older do transmit the virus at least as much as adults.”
“The risk to the students and the community, therefore, is unknown,” and difficult to compare with the benefit of in-person schooling, Dr. Smart noted.
“We will learn quite a bit from communities where students do go back to in-person class, as we follow the progression of COVID-19 over the weeks following the resumption of instruction.” Ultimately, advice to parents will need to be tailored to the current conditions of COVID-19 transmission in the community, he concluded.
It’s not just about education
“The AAP released its guidance to ensure that as school districts were contemplating reopening they were considering the full array of risks for children and adolescents. These risks included not only those related to COVID-19, but also those related to the impact of not reopening in-person,” Nathaniel Beers, MD, president of the HSC Health Care System in Washington, said in an interview.
“Students and families are dependent on schools for much more than just an education, and those [elements] need to be factored into the decisions to reopen,” the pediatrician said.
However, “[t]he major barrier for schools is resources to safely reopen,” said Dr. Beers. “The additional staffing and supplies will require additional funding. There are increased demands regardless of whether students are learning in-person or virtually or through hybrid models.”
“Another significant barrier is ensuring that parents and staff are actively engaged in planning for the type of model being used,” he said.
“All of the models require buy-in by staff and parents. This will require significant outreach and strong communication plans. Schools also need to ensure they are planning not just for how to return students to schools, but what will happen when staff or students test positive for COVID-19. Students, families, and staff all will need to know what these plans are up front to feel confident in returning to school,” he emphasized.
“There are students who can thrive in a virtual learning environment,” Dr. Beers said. “There are also students who benefit from the virtual learning environment because of their own risk, or because of a family member’s risk for COVID-19 or the complications from it.”
“However, many children with disabilities have struggled in a virtual environment,” he said. “These students struggle to access the educational services without the adequate supports at home. They often receive additional services in school, such as speech, occupational therapy or physical therapy, or nursing services, that may not have transitioned to home but are critical for their health and development. Many students with disabilities are dependent on family members to successfully access the educational services they need.”
“Pediatricians can play a role in providing feedback on recommendations related to physical distancing and face coverings in particular,” said Dr. Beers. “In addition, they can be helpful in developing plans for children with disabilities as well as what the response plan should be for students who become sick during the school day.”
The Centers for Disease Control and Prevention released a decision tool for parents who are considering whether to send their child to in-person school, and pediatricians can help parents walk through these questions, Dr. Beers noted. “In addition, pediatricians play an important role in helping patients and families think about the risks of COVID for the patient and other family members, and this can be helpful in addressing the anxiety that parents and patients may be experiencing.”
Further information can be found in Return to School During COVID-19, which can be located at HealthyChildren.org, by the American Academy of Pediatrics.
The authors of the viewpoints had no relevant financial disclosures. Dr. Smart, a member of the Pediatric News editorial advisory board, had no relevant financial disclosures. Dr. Beers has served on the editorial advisory board of Pediatric News in the past, but had no relevant financial disclosures.
SOURCES: Black E, Ferdig R, Thompson LA. JAMA Pediatr. 2020 Aug 11. doi: 10.1001/jamapediatrics.2020.3800. Wang CJ and Bair H. JAMA Pediatr. Aug 11. doi: 10.1001/jamapediatrics.2020.3871.
FROM JAMA PEDIATRICS
Only 40% of residents said training prepped them for COVID-19
Most residents who were asked whether their training prepared them for COVID-19 in a Medscape survey said it had not or they weren’t sure.
Whereas 40% said they felt prepared, 30% said they did not feel prepared and 31% answered they were unsure. (Numbers were rounded, so some answers pushed above 100%.)
One quarter have $300,000 or more in student debt
The Medscape Residents Salary & Debt Report 2020, with data collected April 3 to June 1, found that nearly one in four residents (24%) had medical school debt of more than $300,000. Half (49%) had more than $200,000.
The data include answers from 1,659 U.S. medical residents.
For the sixth straight year, female residents were more satisfied with their pay than were their male colleagues. This year the satisfaction gap was 45% female compared with 42% male. That imbalance came despite their making nearly the same pay overall ($63,700 for men and $63,000 for women).
Among practicing physicians, the pay gap is much wider: Men make 25% more in primary care and 31% more in specialties.
Ten percent thought they should earn 76%-100% more.
For those not satisfied with pay, the top reasons were feeling the pay was too low for the hours worked (81%) or too low compared with other medical staff, such as physician assistants (PAs) or nurses (77% chose that answer).
As for hours worked, 31% of residents reported they spend more than 60 hours/week seeing patients.
The top-paying specialties, averaging $69,500, were allergy and immunology, hematology, plastic surgery, aesthetic medicine, rheumatology, and specialized surgery. The lowest paid were family medicine residents at $58,500.
In primary care, overall, most residents said they planned to specialize. Only 47% planned to continue to work in primary care. Male residents were much more likely to say they will subspecialize than were their female colleagues (52% vs. 35%).
More than 90% of residents say future pay has influenced their choice of specialty, though more men than women felt that way (93% vs. 86%).
Good relationships with others
Overall, residents reported good relationships with attending physicians and nurses.
Most (88%) said they had good or very good relationships with attending physicians, 10% said the relationships were fair, and 2% said they were poor.
In addition, 89% of residents said the amount of supervision was appropriate, 4% said there was too much, and 7% said there was too little.
Relationships with nurses/PAs were slightly less positive overall: Eighty-two percent reported good or very good relationships with nurses/PAs, 15% said those relationships were fair, and 3% said they were poor.
One respondent said: “Our relationships could be better, but I think everyone is just overwhelmed with COVID-19, so emotions are heightened.”
Another said: “It takes time to earn the respect from nurses.”
Seventy-seven percent said they were satisfied with their learning experience overall, 12% were neutral on the question, and 11% said they were dissatisfied or very dissatisfied.
Work-life balance is the top concern
Work-life balance continues to be the top concern for residents. More than one-quarter (27%) in residency years 1 through 4 listed that as the top concern, and even more (32%) of those in years 5 through 8 agreed.
That was followed by demands on time and fear of failure or making a serious mistake.
The survey indicates that benefit packages for residents have stayed much the same over the past 2 years with health insurance and paid time off for sick leave, vacation, and personal time most commonly reported at 89% and 87%, respectively.
Much less common were benefits including commuter assistance (parking, public transportation) at 24%, housing allowance (8%), and child care (4%).
The vast majority of residents reported doing scut work (unskilled tasks): More than half (54%) reported doing 1-10 hours/week and 22% did 11-20 hours/week. Regardless of the number of hours, however, 62% said the time spent performing these tasks was appropriate.
A version of this article originally appeared on Medscape.com.
Most residents who were asked whether their training prepared them for COVID-19 in a Medscape survey said it had not or they weren’t sure.
Whereas 40% said they felt prepared, 30% said they did not feel prepared and 31% answered they were unsure. (Numbers were rounded, so some answers pushed above 100%.)
One quarter have $300,000 or more in student debt
The Medscape Residents Salary & Debt Report 2020, with data collected April 3 to June 1, found that nearly one in four residents (24%) had medical school debt of more than $300,000. Half (49%) had more than $200,000.
The data include answers from 1,659 U.S. medical residents.
For the sixth straight year, female residents were more satisfied with their pay than were their male colleagues. This year the satisfaction gap was 45% female compared with 42% male. That imbalance came despite their making nearly the same pay overall ($63,700 for men and $63,000 for women).
Among practicing physicians, the pay gap is much wider: Men make 25% more in primary care and 31% more in specialties.
Ten percent thought they should earn 76%-100% more.
For those not satisfied with pay, the top reasons were feeling the pay was too low for the hours worked (81%) or too low compared with other medical staff, such as physician assistants (PAs) or nurses (77% chose that answer).
As for hours worked, 31% of residents reported they spend more than 60 hours/week seeing patients.
The top-paying specialties, averaging $69,500, were allergy and immunology, hematology, plastic surgery, aesthetic medicine, rheumatology, and specialized surgery. The lowest paid were family medicine residents at $58,500.
In primary care, overall, most residents said they planned to specialize. Only 47% planned to continue to work in primary care. Male residents were much more likely to say they will subspecialize than were their female colleagues (52% vs. 35%).
More than 90% of residents say future pay has influenced their choice of specialty, though more men than women felt that way (93% vs. 86%).
Good relationships with others
Overall, residents reported good relationships with attending physicians and nurses.
Most (88%) said they had good or very good relationships with attending physicians, 10% said the relationships were fair, and 2% said they were poor.
In addition, 89% of residents said the amount of supervision was appropriate, 4% said there was too much, and 7% said there was too little.
Relationships with nurses/PAs were slightly less positive overall: Eighty-two percent reported good or very good relationships with nurses/PAs, 15% said those relationships were fair, and 3% said they were poor.
One respondent said: “Our relationships could be better, but I think everyone is just overwhelmed with COVID-19, so emotions are heightened.”
Another said: “It takes time to earn the respect from nurses.”
Seventy-seven percent said they were satisfied with their learning experience overall, 12% were neutral on the question, and 11% said they were dissatisfied or very dissatisfied.
Work-life balance is the top concern
Work-life balance continues to be the top concern for residents. More than one-quarter (27%) in residency years 1 through 4 listed that as the top concern, and even more (32%) of those in years 5 through 8 agreed.
That was followed by demands on time and fear of failure or making a serious mistake.
The survey indicates that benefit packages for residents have stayed much the same over the past 2 years with health insurance and paid time off for sick leave, vacation, and personal time most commonly reported at 89% and 87%, respectively.
Much less common were benefits including commuter assistance (parking, public transportation) at 24%, housing allowance (8%), and child care (4%).
The vast majority of residents reported doing scut work (unskilled tasks): More than half (54%) reported doing 1-10 hours/week and 22% did 11-20 hours/week. Regardless of the number of hours, however, 62% said the time spent performing these tasks was appropriate.
A version of this article originally appeared on Medscape.com.
Most residents who were asked whether their training prepared them for COVID-19 in a Medscape survey said it had not or they weren’t sure.
Whereas 40% said they felt prepared, 30% said they did not feel prepared and 31% answered they were unsure. (Numbers were rounded, so some answers pushed above 100%.)
One quarter have $300,000 or more in student debt
The Medscape Residents Salary & Debt Report 2020, with data collected April 3 to June 1, found that nearly one in four residents (24%) had medical school debt of more than $300,000. Half (49%) had more than $200,000.
The data include answers from 1,659 U.S. medical residents.
For the sixth straight year, female residents were more satisfied with their pay than were their male colleagues. This year the satisfaction gap was 45% female compared with 42% male. That imbalance came despite their making nearly the same pay overall ($63,700 for men and $63,000 for women).
Among practicing physicians, the pay gap is much wider: Men make 25% more in primary care and 31% more in specialties.
Ten percent thought they should earn 76%-100% more.
For those not satisfied with pay, the top reasons were feeling the pay was too low for the hours worked (81%) or too low compared with other medical staff, such as physician assistants (PAs) or nurses (77% chose that answer).
As for hours worked, 31% of residents reported they spend more than 60 hours/week seeing patients.
The top-paying specialties, averaging $69,500, were allergy and immunology, hematology, plastic surgery, aesthetic medicine, rheumatology, and specialized surgery. The lowest paid were family medicine residents at $58,500.
In primary care, overall, most residents said they planned to specialize. Only 47% planned to continue to work in primary care. Male residents were much more likely to say they will subspecialize than were their female colleagues (52% vs. 35%).
More than 90% of residents say future pay has influenced their choice of specialty, though more men than women felt that way (93% vs. 86%).
Good relationships with others
Overall, residents reported good relationships with attending physicians and nurses.
Most (88%) said they had good or very good relationships with attending physicians, 10% said the relationships were fair, and 2% said they were poor.
In addition, 89% of residents said the amount of supervision was appropriate, 4% said there was too much, and 7% said there was too little.
Relationships with nurses/PAs were slightly less positive overall: Eighty-two percent reported good or very good relationships with nurses/PAs, 15% said those relationships were fair, and 3% said they were poor.
One respondent said: “Our relationships could be better, but I think everyone is just overwhelmed with COVID-19, so emotions are heightened.”
Another said: “It takes time to earn the respect from nurses.”
Seventy-seven percent said they were satisfied with their learning experience overall, 12% were neutral on the question, and 11% said they were dissatisfied or very dissatisfied.
Work-life balance is the top concern
Work-life balance continues to be the top concern for residents. More than one-quarter (27%) in residency years 1 through 4 listed that as the top concern, and even more (32%) of those in years 5 through 8 agreed.
That was followed by demands on time and fear of failure or making a serious mistake.
The survey indicates that benefit packages for residents have stayed much the same over the past 2 years with health insurance and paid time off for sick leave, vacation, and personal time most commonly reported at 89% and 87%, respectively.
Much less common were benefits including commuter assistance (parking, public transportation) at 24%, housing allowance (8%), and child care (4%).
The vast majority of residents reported doing scut work (unskilled tasks): More than half (54%) reported doing 1-10 hours/week and 22% did 11-20 hours/week. Regardless of the number of hours, however, 62% said the time spent performing these tasks was appropriate.
A version of this article originally appeared on Medscape.com.













