User login
Risk of stopping inhaled corticosteroids
A significant dissatisfier for both clinician and patient is that inhaled corticosteroids, commonly underutilized and potentially lifesaving medications, are almost never (if ever) covered at the lowest tier by insurance companies. We would select a first-tier medication if there were one that we could substitute for an ICS; but frequently there isn’t, so we can’t.
Because of this, patients may be financially motivated to simply stop the medication – especially if they perceive that they are on the lowest doses and believe the medication perhaps is not needed at all. Clinicians, meanwhile, are doing the balancing act of moving patients to the lowest doses in order to avoid side effects while maintaining optimal disease control.
So, what are the risks when patients stop using inhaled corticosteroids?
Dr. Matthew A. Rank of the Mayo Clinic, Rochester, Minn., and his colleagues recently published a systematic review of the literature to answer this question (J. Allergy Clin. Immunol. 2013;131:724-9). In this review, randomized, controlled clinical trials in which the study intervention was continuing or stopping low-dose ICSs were included. Studies had to have 4 or more weeks of a run-in with stable doses of ICSs to ensure a minimum period of asthma stability. Seven studies met inclusion criteria. Two studies were exclusively in children, and one was exclusively in adults.
Asthma exacerbations were more likely among patients who stopped ICSs, compared with those who did not (relative risk, 2.35; 95% CI: 1.88-2.92). The risk for an asthma exacerbation in the next 6 months on low-dose ICSs was 16% if patients continued taking the medications, and 38% if they stopped. For every five patients who stopped ICSs, one patient would be expected to have an asthma exacerbation in the next 6 months – which could have been prevented if steroids had been continued. The mean decrease in forced expiratory volume in 1 second associated with discontinued ICSs use was 130 mL.
Most patients can step down with ICSs if they are on long-acting beta-agonists. Expert panels have suggested that patients should be controlled for 3 months before stepping down therapy. Findings from this study further suggest that patients who discontinue low-dose ICSs are at an increased risk of asthma exacerbation.
We need to help our patients understand the risk of stopping low-dose ICSs and encourage them, as much as they are able, to stay on them.
Dr. Ebbert is professor of medicine and a primary care clinician at the Mayo Clinic in Rochester, Minn. He reported having no relevant financial conflicts. The opinions expressed are those of the author.
This column, "What Matters," appears regularly in Internal Medicine News.
A significant dissatisfier for both clinician and patient is that inhaled corticosteroids, commonly underutilized and potentially lifesaving medications, are almost never (if ever) covered at the lowest tier by insurance companies. We would select a first-tier medication if there were one that we could substitute for an ICS; but frequently there isn’t, so we can’t.
Because of this, patients may be financially motivated to simply stop the medication – especially if they perceive that they are on the lowest doses and believe the medication perhaps is not needed at all. Clinicians, meanwhile, are doing the balancing act of moving patients to the lowest doses in order to avoid side effects while maintaining optimal disease control.
So, what are the risks when patients stop using inhaled corticosteroids?
Dr. Matthew A. Rank of the Mayo Clinic, Rochester, Minn., and his colleagues recently published a systematic review of the literature to answer this question (J. Allergy Clin. Immunol. 2013;131:724-9). In this review, randomized, controlled clinical trials in which the study intervention was continuing or stopping low-dose ICSs were included. Studies had to have 4 or more weeks of a run-in with stable doses of ICSs to ensure a minimum period of asthma stability. Seven studies met inclusion criteria. Two studies were exclusively in children, and one was exclusively in adults.
Asthma exacerbations were more likely among patients who stopped ICSs, compared with those who did not (relative risk, 2.35; 95% CI: 1.88-2.92). The risk for an asthma exacerbation in the next 6 months on low-dose ICSs was 16% if patients continued taking the medications, and 38% if they stopped. For every five patients who stopped ICSs, one patient would be expected to have an asthma exacerbation in the next 6 months – which could have been prevented if steroids had been continued. The mean decrease in forced expiratory volume in 1 second associated with discontinued ICSs use was 130 mL.
Most patients can step down with ICSs if they are on long-acting beta-agonists. Expert panels have suggested that patients should be controlled for 3 months before stepping down therapy. Findings from this study further suggest that patients who discontinue low-dose ICSs are at an increased risk of asthma exacerbation.
We need to help our patients understand the risk of stopping low-dose ICSs and encourage them, as much as they are able, to stay on them.
Dr. Ebbert is professor of medicine and a primary care clinician at the Mayo Clinic in Rochester, Minn. He reported having no relevant financial conflicts. The opinions expressed are those of the author.
This column, "What Matters," appears regularly in Internal Medicine News.
A significant dissatisfier for both clinician and patient is that inhaled corticosteroids, commonly underutilized and potentially lifesaving medications, are almost never (if ever) covered at the lowest tier by insurance companies. We would select a first-tier medication if there were one that we could substitute for an ICS; but frequently there isn’t, so we can’t.
Because of this, patients may be financially motivated to simply stop the medication – especially if they perceive that they are on the lowest doses and believe the medication perhaps is not needed at all. Clinicians, meanwhile, are doing the balancing act of moving patients to the lowest doses in order to avoid side effects while maintaining optimal disease control.
So, what are the risks when patients stop using inhaled corticosteroids?
Dr. Matthew A. Rank of the Mayo Clinic, Rochester, Minn., and his colleagues recently published a systematic review of the literature to answer this question (J. Allergy Clin. Immunol. 2013;131:724-9). In this review, randomized, controlled clinical trials in which the study intervention was continuing or stopping low-dose ICSs were included. Studies had to have 4 or more weeks of a run-in with stable doses of ICSs to ensure a minimum period of asthma stability. Seven studies met inclusion criteria. Two studies were exclusively in children, and one was exclusively in adults.
Asthma exacerbations were more likely among patients who stopped ICSs, compared with those who did not (relative risk, 2.35; 95% CI: 1.88-2.92). The risk for an asthma exacerbation in the next 6 months on low-dose ICSs was 16% if patients continued taking the medications, and 38% if they stopped. For every five patients who stopped ICSs, one patient would be expected to have an asthma exacerbation in the next 6 months – which could have been prevented if steroids had been continued. The mean decrease in forced expiratory volume in 1 second associated with discontinued ICSs use was 130 mL.
Most patients can step down with ICSs if they are on long-acting beta-agonists. Expert panels have suggested that patients should be controlled for 3 months before stepping down therapy. Findings from this study further suggest that patients who discontinue low-dose ICSs are at an increased risk of asthma exacerbation.
We need to help our patients understand the risk of stopping low-dose ICSs and encourage them, as much as they are able, to stay on them.
Dr. Ebbert is professor of medicine and a primary care clinician at the Mayo Clinic in Rochester, Minn. He reported having no relevant financial conflicts. The opinions expressed are those of the author.
This column, "What Matters," appears regularly in Internal Medicine News.
Epi-Curious
The latest recommendations from the USPSTF
Since the last Practice Alert update on the US Preventive Services Task Force (USPSTF) recommendations,1 the Task Force released 16 final recommendations, through January of this year (TABLE).2 However, none of these were level A recommendations and only 4 were level B. This is significant in that USPSTF level A and B recommendations must now be covered by health insurance plans without patient cost sharing as a result of a clause in the Affordable Care Act. There were 5 D recommendations (recommend against), and some of the tests that fell into this category are in common use. I discuss the B and D recommendations below.
TABLE
Recent recommendations from the USPSTF2
| B recommendations |
The USPSTF recommends:
|
| C recommendations |
The USPSTF recommends against automatically:
|
| D recommendations |
The USPSTF recommends against:
|
| I statements |
The USPSTF concludes that the current evidence is insufficient to assess the balance of benefits and harms of:
|
| For more on the USPSTF’s grade definitions, see http://www.uspreventiveservicestaskforce.org/uspstf/grades.htm. |
B recommendations
Encourage vitamin D supplementation and regular exercise to prevent falls in elderly
Falls in the elderly are a significant cause of morbidity and mortality. The Task Force found that between 30% and 40% of community-dwelling adults ≥65 years fall each year, and 5% to 10% of those who fall will sustain a fracture, head injury, or laceration.3 Those at highest risk have a history of falls, report mobility problems, have chronic diseases, use psychotropic medications, or have difficulty on a “get up and go” test, which involves rising from a sitting position in an arm chair, walking 10 feet, turning, walking back, and sitting down. If this activity takes more than 10 seconds, the risk of a fall is increased.3
Two interventions were found to be effective in preventing falls: vitamin D supplementation and regular exercise or physical therapy. Vitamin D enhances muscular strength and balance, and supplementation of 800 IU daily for 12 months can decrease the risk of a fall by 17%, with a number needed to treat (NNT) of 10 to prevent one fall.3 Exercise or physical therapy that focuses on gait and balance, strength or resistance training, or general fitness can reduce the risk of falls with an NNT of 16. Individuals who benefit the most are those at higher risk.3
As for multifactorial risk assessment and comprehensive management of risks to prevent falls, a pooled analysis of studies showed that these interventions do little to reduce falls and do not warrant routine use. The Task Force evaluated other interventions—vision correction, medication discontinuation, protein supplementation, education or counseling, and home hazard modification—but could not find sufficient evidence to recommend for or against them.
Screen for obesity in adults
The Task Force reaffirmed its recommendation to screen all adults for obesity and to offer intensive behavioral interventions to those with a body mass index of ≥30 kg/m2. Helpful interventions include multiple behavioral management activities in group or individual sessions; setting weight-loss goals; improving diet or nutrition; physical activity sessions; addressing barriers to change; active use of self-monitoring; and strategizing ways to maintain lifestyle changes. High-intensity programs involve 12 to 26 sessions a year and result, on average, in a reduction of 6% of body weight.4
Counsel fair-skinned patients to minimize sun exposure
The Task Force now recommends counseling fair-skinned children, adolescents, and young adults (10-24 years of age) about reducing their exposure to ultraviolet (UV) radiation. UV radiation exposure occurs when outdoors in the sun, especially in the middle of the day; and when using artificial sources of UV light, such as an indoor tanning bed. Unprotected UV light exposure is a cause of skin cancer, especially when this exposure occurs in childhood or young adulthood.
Behaviors that protect from UV radiation exposure include using broad-spectrum sunscreen with a sun-protection factor of at least 15, wearing hats and protective clothing, avoiding the outdoors during midday hours (10 am-3 pm), and avoiding indoor tanning. Brief counseling offered in a primary care setting can increase protective behaviors in the targeted age group.
UV light exposure in adults is also linked to skin cancer, but the effectiveness of counseling in this population is less certain and the benefit from protective behaviors is less. In addition, almost all studies of skin cancer prevention have been conducted with fair-skinned subjects, so the Task Force limited this recommendation to those who have fair skin and are between the ages of 10 and 24.5
Screen for intimate partner violence
The USPSTF has changed its recommendation on screening women for intimate partner violence (IPV). Previously it said that the evidence was insufficient to make a recommendation. New evidence has since been published and the Task Force recommends that women of childbearing age (14-46 years, with most evidence for those over age 18) be screened using one of 6 screening tools found to have satisfactory performance characteristics.6 IPV means physical, sexual, or psychological abuse by a current or former partner or spouse, among heterosexual or same-sex couples. To learn more, see “Time to routinely screen for intimate partner violence?” (J Fam Pract. 2013;62:90-92).
Services found to be effective in preventing IPV include counseling, home visits, information cards, referrals to community services, and mentoring support provided by physicians or other health professionals.6
The evidence on screening for the prevention of elder abuse and abuse of vulnerable adults still remains insufficient for a recommendation.
D recommendations
No need for prostate cancer screening, or these other interventions
The list of new D recommendations (interventions that have no benefit or that cause more harm than benefit) includes:
- screening for ovarian and prostate cancer
- using estrogen or estrogen combined with progestin in postmenopausal women for the prevention of chronic conditions
- screening with resting or exercise electrocardiography for the prediction of coronary heart disease events in asymptomatic adults at low risk for such events.
The most controversial D recommendation is to avoid measuring prostate-specific antigen (PSA) to screen for prostate cancer. The Task Force has never endorsed use of the PSA test, previously stating that evidence was not of sufficient strength to recommend for or against it in men <75 years and recommending against it for older men. The evidence report conducted for the reconsideration of this topic provided sufficient evidence that the PSA test results in far more harm than benefit.
In February, the USPSTF finalized a recommendation on “Vitamin D and Calcium Supplementation to Prevent Fractures in Adults.” For more information, go to:
http://www.uspreventiveservicestaskforce.org/announcements.htm
The troublesome C recommendation
Proceed with caution with these 2 interventions
The wording of level C recommendations has undergone revision once again. In recognition that some preventive services may benefit select patients—although the overall benefit in the population is small—the USPSTF now states that a C recommendation means that the Task Force “recommends selectively offering or providing this service to individual patients based on professional judgment and patient preferences.” This past year, 2 interventions fell into this category: multifactorial risk assessment and management to prevent falls in community dwelling elders, and counseling adults about a healthy diet and exercise to prevent cardiovascular disease (TABLE).2
1. Campos-Outcalt D. The latest recommendations from the USPSTF. J Fam Pract. 2012;61:278-282.
2. USPSTF. Announcements. Available at: http://www.uspreventiveservicestaskforce.org/announcements.htm. Accessed March 6, 2013.
3. USPSTF. Prevention of falls in community dwelling older adults. Available at: http://www.uspreventiveservicestaskforce.org/uspstf11/fallsprevention/fallsprevrs.htm. Accessed March 6, 2013.
4. USPSTF. Screening for and management of obesity in adults. Available at: http://www.uspreventiveservicestaskforce.org/uspstf11/obeseadult/obesers.htm. Accessed March 6, 2013.
5. USPSTF. Behavioral counseling to prevent skin cancer. Available at: http://www.uspreventiveservicestaskforce.org/uspstf11/skincancouns/skincancounsrs.htm. Accessed March 6, 2013.
6. USPSTF. Screening for intimate partner violence and abuse of elderly and vulnerable adults. Available at: http://www.uspreventiveservicestaskforce.org/uspstf12/ipvelder/ipvelderfinalrs.htm. Accessed March 6, 2013.
Since the last Practice Alert update on the US Preventive Services Task Force (USPSTF) recommendations,1 the Task Force released 16 final recommendations, through January of this year (TABLE).2 However, none of these were level A recommendations and only 4 were level B. This is significant in that USPSTF level A and B recommendations must now be covered by health insurance plans without patient cost sharing as a result of a clause in the Affordable Care Act. There were 5 D recommendations (recommend against), and some of the tests that fell into this category are in common use. I discuss the B and D recommendations below.
TABLE
Recent recommendations from the USPSTF2
| B recommendations |
The USPSTF recommends:
|
| C recommendations |
The USPSTF recommends against automatically:
|
| D recommendations |
The USPSTF recommends against:
|
| I statements |
The USPSTF concludes that the current evidence is insufficient to assess the balance of benefits and harms of:
|
| For more on the USPSTF’s grade definitions, see http://www.uspreventiveservicestaskforce.org/uspstf/grades.htm. |
B recommendations
Encourage vitamin D supplementation and regular exercise to prevent falls in elderly
Falls in the elderly are a significant cause of morbidity and mortality. The Task Force found that between 30% and 40% of community-dwelling adults ≥65 years fall each year, and 5% to 10% of those who fall will sustain a fracture, head injury, or laceration.3 Those at highest risk have a history of falls, report mobility problems, have chronic diseases, use psychotropic medications, or have difficulty on a “get up and go” test, which involves rising from a sitting position in an arm chair, walking 10 feet, turning, walking back, and sitting down. If this activity takes more than 10 seconds, the risk of a fall is increased.3
Two interventions were found to be effective in preventing falls: vitamin D supplementation and regular exercise or physical therapy. Vitamin D enhances muscular strength and balance, and supplementation of 800 IU daily for 12 months can decrease the risk of a fall by 17%, with a number needed to treat (NNT) of 10 to prevent one fall.3 Exercise or physical therapy that focuses on gait and balance, strength or resistance training, or general fitness can reduce the risk of falls with an NNT of 16. Individuals who benefit the most are those at higher risk.3
As for multifactorial risk assessment and comprehensive management of risks to prevent falls, a pooled analysis of studies showed that these interventions do little to reduce falls and do not warrant routine use. The Task Force evaluated other interventions—vision correction, medication discontinuation, protein supplementation, education or counseling, and home hazard modification—but could not find sufficient evidence to recommend for or against them.
Screen for obesity in adults
The Task Force reaffirmed its recommendation to screen all adults for obesity and to offer intensive behavioral interventions to those with a body mass index of ≥30 kg/m2. Helpful interventions include multiple behavioral management activities in group or individual sessions; setting weight-loss goals; improving diet or nutrition; physical activity sessions; addressing barriers to change; active use of self-monitoring; and strategizing ways to maintain lifestyle changes. High-intensity programs involve 12 to 26 sessions a year and result, on average, in a reduction of 6% of body weight.4
Counsel fair-skinned patients to minimize sun exposure
The Task Force now recommends counseling fair-skinned children, adolescents, and young adults (10-24 years of age) about reducing their exposure to ultraviolet (UV) radiation. UV radiation exposure occurs when outdoors in the sun, especially in the middle of the day; and when using artificial sources of UV light, such as an indoor tanning bed. Unprotected UV light exposure is a cause of skin cancer, especially when this exposure occurs in childhood or young adulthood.
Behaviors that protect from UV radiation exposure include using broad-spectrum sunscreen with a sun-protection factor of at least 15, wearing hats and protective clothing, avoiding the outdoors during midday hours (10 am-3 pm), and avoiding indoor tanning. Brief counseling offered in a primary care setting can increase protective behaviors in the targeted age group.
UV light exposure in adults is also linked to skin cancer, but the effectiveness of counseling in this population is less certain and the benefit from protective behaviors is less. In addition, almost all studies of skin cancer prevention have been conducted with fair-skinned subjects, so the Task Force limited this recommendation to those who have fair skin and are between the ages of 10 and 24.5
Screen for intimate partner violence
The USPSTF has changed its recommendation on screening women for intimate partner violence (IPV). Previously it said that the evidence was insufficient to make a recommendation. New evidence has since been published and the Task Force recommends that women of childbearing age (14-46 years, with most evidence for those over age 18) be screened using one of 6 screening tools found to have satisfactory performance characteristics.6 IPV means physical, sexual, or psychological abuse by a current or former partner or spouse, among heterosexual or same-sex couples. To learn more, see “Time to routinely screen for intimate partner violence?” (J Fam Pract. 2013;62:90-92).
Services found to be effective in preventing IPV include counseling, home visits, information cards, referrals to community services, and mentoring support provided by physicians or other health professionals.6
The evidence on screening for the prevention of elder abuse and abuse of vulnerable adults still remains insufficient for a recommendation.
D recommendations
No need for prostate cancer screening, or these other interventions
The list of new D recommendations (interventions that have no benefit or that cause more harm than benefit) includes:
- screening for ovarian and prostate cancer
- using estrogen or estrogen combined with progestin in postmenopausal women for the prevention of chronic conditions
- screening with resting or exercise electrocardiography for the prediction of coronary heart disease events in asymptomatic adults at low risk for such events.
The most controversial D recommendation is to avoid measuring prostate-specific antigen (PSA) to screen for prostate cancer. The Task Force has never endorsed use of the PSA test, previously stating that evidence was not of sufficient strength to recommend for or against it in men <75 years and recommending against it for older men. The evidence report conducted for the reconsideration of this topic provided sufficient evidence that the PSA test results in far more harm than benefit.
In February, the USPSTF finalized a recommendation on “Vitamin D and Calcium Supplementation to Prevent Fractures in Adults.” For more information, go to:
http://www.uspreventiveservicestaskforce.org/announcements.htm
The troublesome C recommendation
Proceed with caution with these 2 interventions
The wording of level C recommendations has undergone revision once again. In recognition that some preventive services may benefit select patients—although the overall benefit in the population is small—the USPSTF now states that a C recommendation means that the Task Force “recommends selectively offering or providing this service to individual patients based on professional judgment and patient preferences.” This past year, 2 interventions fell into this category: multifactorial risk assessment and management to prevent falls in community dwelling elders, and counseling adults about a healthy diet and exercise to prevent cardiovascular disease (TABLE).2
Since the last Practice Alert update on the US Preventive Services Task Force (USPSTF) recommendations,1 the Task Force released 16 final recommendations, through January of this year (TABLE).2 However, none of these were level A recommendations and only 4 were level B. This is significant in that USPSTF level A and B recommendations must now be covered by health insurance plans without patient cost sharing as a result of a clause in the Affordable Care Act. There were 5 D recommendations (recommend against), and some of the tests that fell into this category are in common use. I discuss the B and D recommendations below.
TABLE
Recent recommendations from the USPSTF2
| B recommendations |
The USPSTF recommends:
|
| C recommendations |
The USPSTF recommends against automatically:
|
| D recommendations |
The USPSTF recommends against:
|
| I statements |
The USPSTF concludes that the current evidence is insufficient to assess the balance of benefits and harms of:
|
| For more on the USPSTF’s grade definitions, see http://www.uspreventiveservicestaskforce.org/uspstf/grades.htm. |
B recommendations
Encourage vitamin D supplementation and regular exercise to prevent falls in elderly
Falls in the elderly are a significant cause of morbidity and mortality. The Task Force found that between 30% and 40% of community-dwelling adults ≥65 years fall each year, and 5% to 10% of those who fall will sustain a fracture, head injury, or laceration.3 Those at highest risk have a history of falls, report mobility problems, have chronic diseases, use psychotropic medications, or have difficulty on a “get up and go” test, which involves rising from a sitting position in an arm chair, walking 10 feet, turning, walking back, and sitting down. If this activity takes more than 10 seconds, the risk of a fall is increased.3
Two interventions were found to be effective in preventing falls: vitamin D supplementation and regular exercise or physical therapy. Vitamin D enhances muscular strength and balance, and supplementation of 800 IU daily for 12 months can decrease the risk of a fall by 17%, with a number needed to treat (NNT) of 10 to prevent one fall.3 Exercise or physical therapy that focuses on gait and balance, strength or resistance training, or general fitness can reduce the risk of falls with an NNT of 16. Individuals who benefit the most are those at higher risk.3
As for multifactorial risk assessment and comprehensive management of risks to prevent falls, a pooled analysis of studies showed that these interventions do little to reduce falls and do not warrant routine use. The Task Force evaluated other interventions—vision correction, medication discontinuation, protein supplementation, education or counseling, and home hazard modification—but could not find sufficient evidence to recommend for or against them.
Screen for obesity in adults
The Task Force reaffirmed its recommendation to screen all adults for obesity and to offer intensive behavioral interventions to those with a body mass index of ≥30 kg/m2. Helpful interventions include multiple behavioral management activities in group or individual sessions; setting weight-loss goals; improving diet or nutrition; physical activity sessions; addressing barriers to change; active use of self-monitoring; and strategizing ways to maintain lifestyle changes. High-intensity programs involve 12 to 26 sessions a year and result, on average, in a reduction of 6% of body weight.4
Counsel fair-skinned patients to minimize sun exposure
The Task Force now recommends counseling fair-skinned children, adolescents, and young adults (10-24 years of age) about reducing their exposure to ultraviolet (UV) radiation. UV radiation exposure occurs when outdoors in the sun, especially in the middle of the day; and when using artificial sources of UV light, such as an indoor tanning bed. Unprotected UV light exposure is a cause of skin cancer, especially when this exposure occurs in childhood or young adulthood.
Behaviors that protect from UV radiation exposure include using broad-spectrum sunscreen with a sun-protection factor of at least 15, wearing hats and protective clothing, avoiding the outdoors during midday hours (10 am-3 pm), and avoiding indoor tanning. Brief counseling offered in a primary care setting can increase protective behaviors in the targeted age group.
UV light exposure in adults is also linked to skin cancer, but the effectiveness of counseling in this population is less certain and the benefit from protective behaviors is less. In addition, almost all studies of skin cancer prevention have been conducted with fair-skinned subjects, so the Task Force limited this recommendation to those who have fair skin and are between the ages of 10 and 24.5
Screen for intimate partner violence
The USPSTF has changed its recommendation on screening women for intimate partner violence (IPV). Previously it said that the evidence was insufficient to make a recommendation. New evidence has since been published and the Task Force recommends that women of childbearing age (14-46 years, with most evidence for those over age 18) be screened using one of 6 screening tools found to have satisfactory performance characteristics.6 IPV means physical, sexual, or psychological abuse by a current or former partner or spouse, among heterosexual or same-sex couples. To learn more, see “Time to routinely screen for intimate partner violence?” (J Fam Pract. 2013;62:90-92).
Services found to be effective in preventing IPV include counseling, home visits, information cards, referrals to community services, and mentoring support provided by physicians or other health professionals.6
The evidence on screening for the prevention of elder abuse and abuse of vulnerable adults still remains insufficient for a recommendation.
D recommendations
No need for prostate cancer screening, or these other interventions
The list of new D recommendations (interventions that have no benefit or that cause more harm than benefit) includes:
- screening for ovarian and prostate cancer
- using estrogen or estrogen combined with progestin in postmenopausal women for the prevention of chronic conditions
- screening with resting or exercise electrocardiography for the prediction of coronary heart disease events in asymptomatic adults at low risk for such events.
The most controversial D recommendation is to avoid measuring prostate-specific antigen (PSA) to screen for prostate cancer. The Task Force has never endorsed use of the PSA test, previously stating that evidence was not of sufficient strength to recommend for or against it in men <75 years and recommending against it for older men. The evidence report conducted for the reconsideration of this topic provided sufficient evidence that the PSA test results in far more harm than benefit.
In February, the USPSTF finalized a recommendation on “Vitamin D and Calcium Supplementation to Prevent Fractures in Adults.” For more information, go to:
http://www.uspreventiveservicestaskforce.org/announcements.htm
The troublesome C recommendation
Proceed with caution with these 2 interventions
The wording of level C recommendations has undergone revision once again. In recognition that some preventive services may benefit select patients—although the overall benefit in the population is small—the USPSTF now states that a C recommendation means that the Task Force “recommends selectively offering or providing this service to individual patients based on professional judgment and patient preferences.” This past year, 2 interventions fell into this category: multifactorial risk assessment and management to prevent falls in community dwelling elders, and counseling adults about a healthy diet and exercise to prevent cardiovascular disease (TABLE).2
1. Campos-Outcalt D. The latest recommendations from the USPSTF. J Fam Pract. 2012;61:278-282.
2. USPSTF. Announcements. Available at: http://www.uspreventiveservicestaskforce.org/announcements.htm. Accessed March 6, 2013.
3. USPSTF. Prevention of falls in community dwelling older adults. Available at: http://www.uspreventiveservicestaskforce.org/uspstf11/fallsprevention/fallsprevrs.htm. Accessed March 6, 2013.
4. USPSTF. Screening for and management of obesity in adults. Available at: http://www.uspreventiveservicestaskforce.org/uspstf11/obeseadult/obesers.htm. Accessed March 6, 2013.
5. USPSTF. Behavioral counseling to prevent skin cancer. Available at: http://www.uspreventiveservicestaskforce.org/uspstf11/skincancouns/skincancounsrs.htm. Accessed March 6, 2013.
6. USPSTF. Screening for intimate partner violence and abuse of elderly and vulnerable adults. Available at: http://www.uspreventiveservicestaskforce.org/uspstf12/ipvelder/ipvelderfinalrs.htm. Accessed March 6, 2013.
1. Campos-Outcalt D. The latest recommendations from the USPSTF. J Fam Pract. 2012;61:278-282.
2. USPSTF. Announcements. Available at: http://www.uspreventiveservicestaskforce.org/announcements.htm. Accessed March 6, 2013.
3. USPSTF. Prevention of falls in community dwelling older adults. Available at: http://www.uspreventiveservicestaskforce.org/uspstf11/fallsprevention/fallsprevrs.htm. Accessed March 6, 2013.
4. USPSTF. Screening for and management of obesity in adults. Available at: http://www.uspreventiveservicestaskforce.org/uspstf11/obeseadult/obesers.htm. Accessed March 6, 2013.
5. USPSTF. Behavioral counseling to prevent skin cancer. Available at: http://www.uspreventiveservicestaskforce.org/uspstf11/skincancouns/skincancounsrs.htm. Accessed March 6, 2013.
6. USPSTF. Screening for intimate partner violence and abuse of elderly and vulnerable adults. Available at: http://www.uspreventiveservicestaskforce.org/uspstf12/ipvelder/ipvelderfinalrs.htm. Accessed March 6, 2013.
Heparin-Induced Thrombocytopenia
Heparin-induced thrombocytopenia (HIT) is an immune-mediated drug reaction that requires prompt detection and treatment in order to minimize patient morbidity and mortality.1 HIT is caused by the development of antibodies to platelet factor 4 (PF4), although it is important to note that not all patients who develop PF4 antibodies will experience the clinical syndrome of HIT.2-4 In fact, about 50% of patients who undergo cardiovascular surgery develop PF4 antibodies, but only 1% to 2% of patients with antibodies actually experience HIT.5-7 There is currently no explanation for the phenomenon of HIT.8
In 2012, with an intent to limit HIT-associated morbidity and mortality, the American College of Chest Physicians (ACCP) unveiled the ninth edition of its evidence-based practice guidelines for the detection of HIT and appropriate treatment.5 Much of the information provided in this article emerged from these guidelines.
Epidemiology
Of the 12 million patients treated each year with either unfractionated heparin (UFH) or low-molecular-weight heparin (LMWH), 600,000 (0.5%) will develop HIT. Among these patients, 300,000 will develop thrombosis, and 90,000 will die. In 2009 alone, the HIT-associated cost to the US health care system was estimated at $100 million.1
As growing numbers of patients require anticoagulation therapy, it becomes increasingly important for clinicians to understand the importance of screening for deep vein thrombosis (DVT), one of the two most common thromboses; the other is pulmonary embolism.9,10 Continuing to administer heparin or warfarin to patients with undetected HIT predisposes them to severe complications, including venous and arterial thromboses and gangrenous skin lesions—which can result in loss of life and/or limb.1,11,12
Risk Factors for HIT
Several factors influence a patient's risk for HIT, including the type and dosing regimen of the heparin being administered. Generally, the risk for HIT is about 10-fold in patients treated with UFH (3% to 5%), compared with those receiving LMWH (0.5%).5,13 The risk for HIT is also greater in patients receiving UFH of bovine origin, compared with those taking porcine-derived UFH.8,14,15
In a recent meta-analysis of postsurgical patients who underwent heparin thromboprophylaxis, those given LMWH had a 76% relative risk reduction for HIT, compared with patients taking UFH.16 The incidence of HIT increases among patients receiving LMWH if they have been treated with UFH within the previous 100 days.9 HIT onset may be delayed for several days in patients given heparin for the first time (or for the first time in several months), whereas previously exposed patients who have already developed antiheparin PF4 antibodies can experience severe HIT within hours.9
Patient-Specific Risk Factors
Certain patient characteristics also have an impact on HIT risk. For example, the risk for HIT is approximately doubled in women, compared with men,1,5,15 and the incidence of HIT is greater in surgical patients than in medical patients.7,17 Among surgical patients, 5% of orthopedic patients have been reported to develop HIT, compared with 3% of cardiac patients and 1% of patients undergoing surgery for vascular illnesses.1 The reasons for these differences are poorly understood, but current theory focuses on the inflammatory response of individual patients and the degree of associated platelet activation.2,12
Patient Presentation and History
The typical patient with HIT presents with a new or progressing thrombosis between days 5 and 14 of heparin therapy (with day 0 representing the day the first dose is administered); thrombosis can be venous or arterial, although venous thrombosis occurs much more frequently.1,5,9,15 As patients rarely remain hospitalized for such a long period, it is imperative that providers in clinic and emergency settings obtain detailed histories for patients who present with thrombocytopenia and/or thrombosis. HIT should be suspected in any such patient whose history shows heparin use within the previous two weeks (even if the drug has been discontinued).15
Two forms of atypical HIT are rapid-onset HIT and delayed-onset HIT. Rapid-onset HIT is defined by a platelet count that falls within 24 hours of exposure to heparin. This form is usually associated with previous heparin exposure (ie, within the previous 100 days, but most commonly within the previous 30 days). Affected patients have already developed circulating antiheparin PF4 antibodies, causing an immediate reaction when the patient is re-exposed to the drug.1,15
The less common delayed-onset HIT occurs in patients in whom heparin has been discontinued for as long as 40 days. Delayed-onset HIT carries the greatest risk for severe thrombosis.1,15
Atypically, a patient may present with bleeding, skin necrosis, venous gangrene, or anaphylaxis,9 but skin necrosis at the site of heparin injection is strongly suggestive of HIT.12However, neither physical signs nor symptoms, nor a thrombotic event is required to make a diagnosis of HIT. In fact, the preference is for a diagnosis to be made before thrombosis formation.5
The major manifestation of HIT is thrombocytopenia itself2,18(see "Laboratory Findings"). Nevertheless, if physical signs and symptoms are evident, they will be related to the thrombosis, and the components of the physical exam will proceed accordingly.
Laboratory Findings
Platelet count monitoring and HIT antibody testing are the laboratory tests most commonly used when HIT is suspected. Although 25% of patients with HIT will experience a thrombotic event before the platelet count falls, monitoring the platelet count is considered the most effective means to identify patients with HIT.5 HIT antibody testing is not recommended unless the health care provider has a strong suspicion for HIT.19
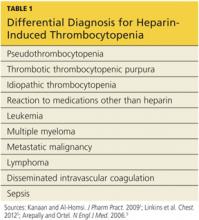
Thrombocytopenia is a common abnormality, especially in hospitalized patients, and its causes are numerous.11 Table 11,5,9 lists the differential diagnosis that the clinician who suspects HIT should consider.
Nevertheless, the ACCP guidelines5 recommend platelet count monitoring for all patients receiving heparin, beginning on day 4 of heparin therapy, then continuing every two to three days until treatment day 14 or heparin discontinuation, whichever occurs first.5,15 A platelet count decrease of 50% or more should raise a suspicion for HIT.15
The ACCP makes two principal exceptions to these recommendations.5 The first involves patients who have received UFH within the previous 100 days. These patients should undergo a baseline platelet count before heparin is administered, followed by a repeat platelet count within 24 hours. Any patient who experiences an anaphylactic reaction to UFH should undergo an immediate platelet count; a decrease in these patients is often transient.
The second exception pertains to medical and obstetric patients. Those receiving LMWH or UFH only to maintain line patency do not require platelet monitoring, as their risk for HIT is relatively low.1,5
Laboratory Interpretation
In HIT, thrombocytopenia is defined as a platelet count below 150,000/mm3 or a platelet count reduction of 50% or more from baseline, even if the platelet count remains above 150,000/mm3.9 (The patient's baseline platelet count is defined as the highest count recorded in the previous two weeks.1,5,15) The thrombocytopenia associated with HIT is rarely severe and can be easily overlooked.1
Once the platelet count suggests a diagnosis of HIT, heparin-dependent antibodies can be identified through immunologic or functional assays.1,9,15 Immunologic assays should be ordered immediately upon suspicion of HIT since they are simple tests with relatively rapid results. Immunologic assays detect immunoglobulin G (IgG), IgA, and IgM antibodies.9Though lacking in specificity, the immunologic assay is highly sensitive.2,12,20 The most frequently used immunologic assay is the enzyme-linked immunosorbent assay (ELISA).1,2,9,20The ELISA, which detects antiheparin PF4 antibodies, has a sensitivity greater than 97% but a specificity of only 74% to 86%.8,12
Functional assays,which are technically demanding, test the ability of PF4 antibodies to activate platelets in the presence of heparin. The functional assay is used to confirm the diagnosis of HIT when a positive ELISA result is obtained.1,9,15 Among the functional assays, the serotonin release assay (SRA) has been most completely studied. Though very expensive, the SRA is 89% to 100% specific in diagnosing HIT.2,12
Diagnosis
The diagnosis of HIT is determined by combining clinical and serologic assessment. HIT should be suspected in any patient who is in day 5 to 14 of heparin therapy and experiences a drop in platelet count of at least 50%, or in whom a new thrombotic event occurs (even if the patient is no longer receiving heparin therapy). The interpretation of all diagnostic information must be made in the context of the patient's clinical probability of HIT.15
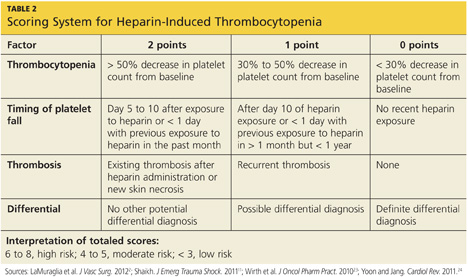
A scoring system referred to as the 4Ts (thrombocytopenia, timing of platelet fall, thrombosis or other sequelae, and test interpretation) is used to help determine the patient's probability of HIT5,21,22 (similar to the scoring strategy shown in Table 22,11,23,24).
The patient diagnosed with HIT must be positive for HIT antibodies and meet at least one additional criterion:
• A platelet count decrease of 30% to 50% below baseline, regardless of the actual value
• A venous or arterial thrombosis
• A skin lesion at the heparin injection site; and/or
• An anaphylactic reaction after IV bolus administration of heparin.15
Treatment/Management
The goal in management of HIT is to reduce the likelihood, then the severity, of thrombosis.9 Treatment should be started as soon as HIT is suspected, before laboratory confirmation.25 Treatment for HIT comprises two steps: stopping all exposure to heparin, and administering an alternative, non-heparin anticoagulant.
Discontinuation of Heparin Exposure
Stopping heparin exposure is the mainstay of treatment for HIT. This includes all potential sources of heparin exposure, including "flushes" that may be used to promote patency of central IV catheters, use of UFH-coated catheters, or addition of any heparin to total parenteral nutrition.9,25
Use of a Non-Heparin Anticoagulant
In addition to discontinuation of all heparin exposure, the patient must be started on a non-heparin anticoagulant, whether or not thrombosis is present.25 Forty percent to 50% of patients without thrombosis will develop a thrombosis within 30 days if alternative anticoagulation is not started.12,18,26
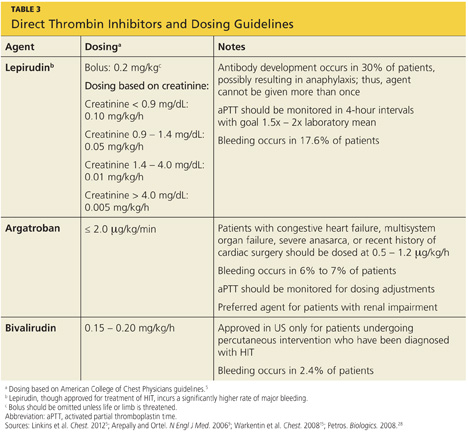
The principal choices for a non-heparin anticoagulant are the direct thrombin inhibitors (DTIs): lepirudin, argatroban, and bivalirudin.1,9,15,25,27 DTIs are the treatment of choice for patients with known or suspected HIT. The ACCP dosing guidelines for the DTIs are summarized in Table 3.9,5,15,28
The factor Xa inhibitor fondaparinux, though FDA approved for DVT prophylaxis,11 has not yet been systematically investigated for the treatment of HIT; thus, its use for this indication is considered off-label.29 However, small studies have shown no cross-reactivity between fondaparinux and PF4 antibodies.30 Due to the positive risk/benefit ratio, ease of use, and reduced need for monitoring in patients taking fondaparinux, it is considered an attractive alternative to DTIs that may receive approval in the near future.12,18,20,29
Currently, the ACCP limits its recommendation of fondaparinux use to patients with a previous history of HIT who require anticoagulation for an acute thrombotic event unrelated to HIT (grade 2C recommendation).5
The vitamin K antagonist warfarin is absolutely contraindicated in patients with HIT until the platelet count is at least 150,000/mm3, due to the risk for warfarin-induced skin necrosis and venous gangrene.9,15 If a patient is receiving warfarin at diagnosis, vitamin K (10 mg orally or 5 to 10 mg IV) should be administered.15
The patient should remain on the alternative non-heparin anticoagulant until the platelet count has stabilized at or above 150,000/mm3. Warfarin should then be started at a maximum of 5 mg/d.2,5 The non-heparin anticoagulant and warfarin should be continued until a therapeutic international normalized ratio (INR) is reached and maintained for 48 hours, with a minimum 5-day overlap of the two medications. Once the non-heparin anticoagulant is discontinued, the INR should be reevaluated for remaining within the therapeutic range, as DTIs can elevate the INR.2,5Warfarin should be continued for as long as four weeks, with frequent INR monitoring.15
Patient Education
The presence of PF4 antibodies is transient (50 to 80 days); however, concern persists regarding recurrent antibody development with subsequent heparin use. Thus, an alternative anticoagulant should be used whenever possible. Patients who have been diagnosed with HIT should be advised to inform future health care professionals regarding their need for alternative anticoagulation whenever possible.
Patients should also be made aware that when the risk for DTI-associated bleeding is too great (as in the case of cardiac surgery), heparin remains the anticoagulant of choice.9,15
Conclusion
Heparin-induced thrombocytopenia is a transient development of antibodies to heparin. While the condition carries a high risk for morbidity and mortality, early detection and prompt treatment can greatly reduce the associated risk to life and limb.
References
1. Kanaan AO, Al-Homsi AS. Heparin-induced thrombocytopenia: pathophysiology, diagnosis, and review of pharmacotherapy. J Pharm Pract. 2009;22:149-157.
2. LaMuraglia GM, Houbballah R, Laposata M. The identification and management of heparin-induced thrombocytopenia in the vascular patient. J Vasc Surg. 2012;55:562-570.
3. Rauova L, Zhai L, Kowalska MA, et al. Role of platelet surface PF4 antigenic complexes in heparin-induced thrombocytopenia pathogenesis: diagnostic and therapeutic implications. Blood. 2006;107:2346-2353.
4. Suvarna S, Espinasse B, Qi R, et al. Determinants of PF4/heparin immunogenicity. Blood. 2007;110:4253-4260.
5. Linkins LA, Dans AL, Moores LK, et al. Treatment and prevention of heparin-induced thrombocytopenia: antithrombotic therapy and prevention of thrombosis, 9th ed: American College of Chest Physicians Evidence-Based Clinical Practice Guidelines. Chest. 2012;141(2 suppl):e495S-e530S.
6. Demma LJ, Winkler AM, Levy JH. A diagnosis of heparin-induced thrombocytopenia with combined clinical and laboratory methods in cardiothoracic surgical intensive care unit patients. Anesth Analg. 2011;113:697-702.
7. Demma LJ, Levy JH. Diagnosing heparin-induced thrombocytopenia in cardiac surgical patients: not as easy as you think. Anesth Analg. 2011;112:747-749.
8. Alaraj A, Wallace A, Tesoro E, et al. Heparin-induced thrombocytopenia: diagnosis and management. J Neurointervent Surg. 2010;2:371-378.
9. Arepally GM, Ortel TL. Heparin-induced thrombocytopenia. N Engl J Med. 2006;355:809-817.
10. Sud S, Mittmann N, Cook DJ, et al. Screening and prevention of venous thromboembolism in critically ill patients: a decision analysis and economic evaluation. Am J Resp Crit Care Med. 2011;184:1289-1298.
11. Shaikh N. Heparin-induced thrombocytopenia. J Emerg Trauma Shock. 2011;14:97-102.
12. Cuker A. Heparin-induced thrombocytopenia: present and future. J Thromb Thrombolysis. 2011;31:353-366.
13. Locke CSF, Dooley J, Gerber J. Rates of clinically apparent heparin-induced thrombocytopenia for unfractionated heparin vs low molecular weight heparin in non-surgical patients are low and similar. Thromb J. 2005;3:4.
14. Cuker A. Recent advances in heparin-induced thrombocytopenia. Curr Opin Hematol. 2011;18:315-322.
15. Warkentin TE, Greinacher A, Koster A, Lincoff AM. Treatment and prevention of heparin-induced thrombocytopenia: American College of Chest Physicians Evidence-Based Clinical Practice Guidelines (8th ed). Chest. 2008;133(6 suppl):340S-380S.
16. Junqueira DR, Perini E, Penholati RR, Carvalho MG. Unfractionated heparin versus low molecular weight heparin for avoiding heparin-induced thrombocytopenia in postoperative patients. Cochrane Database Syst Rev. 2012;9:CD007557.
17. Berry C, Tcherniantchouk O, Ley EJ, et al. Overdiagnosis of heparin-induced thrombocytopenia in surgical ICU patients. J Am Coll Surg. 2011;213:10-17.
18. Cuker A. Current and emerging therapeutics for heparin-induced thrombocytopenia. Semin Thromb Hemost. 2012;38:31-37.
19. Warkentin TE. New approaches to the diagnosis of heparin-induced thrombocytopenia. Chest. 2005;127(2 suppl):35S-45S.
20. Fennessy-Cooney M. Heparin-induced thrombocytopenia. Nurse Pract. 2011;36:31-37.
21. Bryant A, Low J, Austin S, Joseph JE. Timely diagnosis and management of heparin-induced thrombocytopenia in a frequent request, low incidence single centre using clinical 4T's score and particle gel immunoassay. Br J Haematol. 2008;143:721-726.
22. Lo GK, Juhl D, Warkentin TE, et al. Evaluation of pretest clinical score (4 T's) for the diagnosis of heparin-induced thrombocytopenia in two clinical settings. J Thromb Haemost. 2006;4:759-765.
23. Wirth SM, Macaulay TE, Armistead JA, et al. Evaluation of a clinical scoring scale to direct early appropriate therapy in heparin-induced thrombocytopenia. J Oncol Pharm Pract. 2010;16:161-166.
24. Yoon JH, Jang IK. Heparin-induced thrombocytopenia in cardiovascular patients: pathophysiology, diagnosis, and treatment. Cardiol Rev. 2011;19:143-153.
25. Bartholomew JR. Heparin-induced thrombocytopenia: 2008 update. Curr Treat Options Cardiovasc Med. 2008;10:117-127.
26. Warkentin TE. Platelet count monitoring and laboratory testing for heparin-induced thrombocytopenia. Arch Pathol Lab Med. 2002;126:1415-1423.
27. Badger NO. Fondaparinux (Arixtra®), a safe alternative for the treatment of patients with heparin-induced thrombocytopenia? J Pharm Pract. 2010;23:235-238.
28. Petros S. Lepirudin in the management of patients with heparin-induced thrombocytopenia. Biologics. 2008;2:481-490.
29. Warkentin TE. How I diagnose and manage HIT. Hematology Am Soc Hematol Educ Program. 2011;2011:143-149.
30. Papadopoulos S, Flynn JD, Lewis DA. Fondaparinux as a treatment option for heparin-induced thrombocytopenia. Pharmacotherapy. 2007; 27:921-926.
Heparin-induced thrombocytopenia (HIT) is an immune-mediated drug reaction that requires prompt detection and treatment in order to minimize patient morbidity and mortality.1 HIT is caused by the development of antibodies to platelet factor 4 (PF4), although it is important to note that not all patients who develop PF4 antibodies will experience the clinical syndrome of HIT.2-4 In fact, about 50% of patients who undergo cardiovascular surgery develop PF4 antibodies, but only 1% to 2% of patients with antibodies actually experience HIT.5-7 There is currently no explanation for the phenomenon of HIT.8
In 2012, with an intent to limit HIT-associated morbidity and mortality, the American College of Chest Physicians (ACCP) unveiled the ninth edition of its evidence-based practice guidelines for the detection of HIT and appropriate treatment.5 Much of the information provided in this article emerged from these guidelines.
Epidemiology
Of the 12 million patients treated each year with either unfractionated heparin (UFH) or low-molecular-weight heparin (LMWH), 600,000 (0.5%) will develop HIT. Among these patients, 300,000 will develop thrombosis, and 90,000 will die. In 2009 alone, the HIT-associated cost to the US health care system was estimated at $100 million.1
As growing numbers of patients require anticoagulation therapy, it becomes increasingly important for clinicians to understand the importance of screening for deep vein thrombosis (DVT), one of the two most common thromboses; the other is pulmonary embolism.9,10 Continuing to administer heparin or warfarin to patients with undetected HIT predisposes them to severe complications, including venous and arterial thromboses and gangrenous skin lesions—which can result in loss of life and/or limb.1,11,12
Risk Factors for HIT
Several factors influence a patient's risk for HIT, including the type and dosing regimen of the heparin being administered. Generally, the risk for HIT is about 10-fold in patients treated with UFH (3% to 5%), compared with those receiving LMWH (0.5%).5,13 The risk for HIT is also greater in patients receiving UFH of bovine origin, compared with those taking porcine-derived UFH.8,14,15
In a recent meta-analysis of postsurgical patients who underwent heparin thromboprophylaxis, those given LMWH had a 76% relative risk reduction for HIT, compared with patients taking UFH.16 The incidence of HIT increases among patients receiving LMWH if they have been treated with UFH within the previous 100 days.9 HIT onset may be delayed for several days in patients given heparin for the first time (or for the first time in several months), whereas previously exposed patients who have already developed antiheparin PF4 antibodies can experience severe HIT within hours.9
Patient-Specific Risk Factors
Certain patient characteristics also have an impact on HIT risk. For example, the risk for HIT is approximately doubled in women, compared with men,1,5,15 and the incidence of HIT is greater in surgical patients than in medical patients.7,17 Among surgical patients, 5% of orthopedic patients have been reported to develop HIT, compared with 3% of cardiac patients and 1% of patients undergoing surgery for vascular illnesses.1 The reasons for these differences are poorly understood, but current theory focuses on the inflammatory response of individual patients and the degree of associated platelet activation.2,12
Patient Presentation and History
The typical patient with HIT presents with a new or progressing thrombosis between days 5 and 14 of heparin therapy (with day 0 representing the day the first dose is administered); thrombosis can be venous or arterial, although venous thrombosis occurs much more frequently.1,5,9,15 As patients rarely remain hospitalized for such a long period, it is imperative that providers in clinic and emergency settings obtain detailed histories for patients who present with thrombocytopenia and/or thrombosis. HIT should be suspected in any such patient whose history shows heparin use within the previous two weeks (even if the drug has been discontinued).15
Two forms of atypical HIT are rapid-onset HIT and delayed-onset HIT. Rapid-onset HIT is defined by a platelet count that falls within 24 hours of exposure to heparin. This form is usually associated with previous heparin exposure (ie, within the previous 100 days, but most commonly within the previous 30 days). Affected patients have already developed circulating antiheparin PF4 antibodies, causing an immediate reaction when the patient is re-exposed to the drug.1,15
The less common delayed-onset HIT occurs in patients in whom heparin has been discontinued for as long as 40 days. Delayed-onset HIT carries the greatest risk for severe thrombosis.1,15
Atypically, a patient may present with bleeding, skin necrosis, venous gangrene, or anaphylaxis,9 but skin necrosis at the site of heparin injection is strongly suggestive of HIT.12However, neither physical signs nor symptoms, nor a thrombotic event is required to make a diagnosis of HIT. In fact, the preference is for a diagnosis to be made before thrombosis formation.5
The major manifestation of HIT is thrombocytopenia itself2,18(see "Laboratory Findings"). Nevertheless, if physical signs and symptoms are evident, they will be related to the thrombosis, and the components of the physical exam will proceed accordingly.
Laboratory Findings
Platelet count monitoring and HIT antibody testing are the laboratory tests most commonly used when HIT is suspected. Although 25% of patients with HIT will experience a thrombotic event before the platelet count falls, monitoring the platelet count is considered the most effective means to identify patients with HIT.5 HIT antibody testing is not recommended unless the health care provider has a strong suspicion for HIT.19
Thrombocytopenia is a common abnormality, especially in hospitalized patients, and its causes are numerous.11 Table 11,5,9 lists the differential diagnosis that the clinician who suspects HIT should consider.
Nevertheless, the ACCP guidelines5 recommend platelet count monitoring for all patients receiving heparin, beginning on day 4 of heparin therapy, then continuing every two to three days until treatment day 14 or heparin discontinuation, whichever occurs first.5,15 A platelet count decrease of 50% or more should raise a suspicion for HIT.15
The ACCP makes two principal exceptions to these recommendations.5 The first involves patients who have received UFH within the previous 100 days. These patients should undergo a baseline platelet count before heparin is administered, followed by a repeat platelet count within 24 hours. Any patient who experiences an anaphylactic reaction to UFH should undergo an immediate platelet count; a decrease in these patients is often transient.
The second exception pertains to medical and obstetric patients. Those receiving LMWH or UFH only to maintain line patency do not require platelet monitoring, as their risk for HIT is relatively low.1,5
Laboratory Interpretation
In HIT, thrombocytopenia is defined as a platelet count below 150,000/mm3 or a platelet count reduction of 50% or more from baseline, even if the platelet count remains above 150,000/mm3.9 (The patient's baseline platelet count is defined as the highest count recorded in the previous two weeks.1,5,15) The thrombocytopenia associated with HIT is rarely severe and can be easily overlooked.1
Once the platelet count suggests a diagnosis of HIT, heparin-dependent antibodies can be identified through immunologic or functional assays.1,9,15 Immunologic assays should be ordered immediately upon suspicion of HIT since they are simple tests with relatively rapid results. Immunologic assays detect immunoglobulin G (IgG), IgA, and IgM antibodies.9Though lacking in specificity, the immunologic assay is highly sensitive.2,12,20 The most frequently used immunologic assay is the enzyme-linked immunosorbent assay (ELISA).1,2,9,20The ELISA, which detects antiheparin PF4 antibodies, has a sensitivity greater than 97% but a specificity of only 74% to 86%.8,12
Functional assays,which are technically demanding, test the ability of PF4 antibodies to activate platelets in the presence of heparin. The functional assay is used to confirm the diagnosis of HIT when a positive ELISA result is obtained.1,9,15 Among the functional assays, the serotonin release assay (SRA) has been most completely studied. Though very expensive, the SRA is 89% to 100% specific in diagnosing HIT.2,12
Diagnosis
The diagnosis of HIT is determined by combining clinical and serologic assessment. HIT should be suspected in any patient who is in day 5 to 14 of heparin therapy and experiences a drop in platelet count of at least 50%, or in whom a new thrombotic event occurs (even if the patient is no longer receiving heparin therapy). The interpretation of all diagnostic information must be made in the context of the patient's clinical probability of HIT.15
A scoring system referred to as the 4Ts (thrombocytopenia, timing of platelet fall, thrombosis or other sequelae, and test interpretation) is used to help determine the patient's probability of HIT5,21,22 (similar to the scoring strategy shown in Table 22,11,23,24).

The patient diagnosed with HIT must be positive for HIT antibodies and meet at least one additional criterion:
• A platelet count decrease of 30% to 50% below baseline, regardless of the actual value
• A venous or arterial thrombosis
• A skin lesion at the heparin injection site; and/or
• An anaphylactic reaction after IV bolus administration of heparin.15
Treatment/Management
The goal in management of HIT is to reduce the likelihood, then the severity, of thrombosis.9 Treatment should be started as soon as HIT is suspected, before laboratory confirmation.25 Treatment for HIT comprises two steps: stopping all exposure to heparin, and administering an alternative, non-heparin anticoagulant.
Discontinuation of Heparin Exposure
Stopping heparin exposure is the mainstay of treatment for HIT. This includes all potential sources of heparin exposure, including "flushes" that may be used to promote patency of central IV catheters, use of UFH-coated catheters, or addition of any heparin to total parenteral nutrition.9,25
Use of a Non-Heparin Anticoagulant
In addition to discontinuation of all heparin exposure, the patient must be started on a non-heparin anticoagulant, whether or not thrombosis is present.25 Forty percent to 50% of patients without thrombosis will develop a thrombosis within 30 days if alternative anticoagulation is not started.12,18,26
The principal choices for a non-heparin anticoagulant are the direct thrombin inhibitors (DTIs): lepirudin, argatroban, and bivalirudin.1,9,15,25,27 DTIs are the treatment of choice for patients with known or suspected HIT. The ACCP dosing guidelines for the DTIs are summarized in Table 3.9,5,15,28

The factor Xa inhibitor fondaparinux, though FDA approved for DVT prophylaxis,11 has not yet been systematically investigated for the treatment of HIT; thus, its use for this indication is considered off-label.29 However, small studies have shown no cross-reactivity between fondaparinux and PF4 antibodies.30 Due to the positive risk/benefit ratio, ease of use, and reduced need for monitoring in patients taking fondaparinux, it is considered an attractive alternative to DTIs that may receive approval in the near future.12,18,20,29
Currently, the ACCP limits its recommendation of fondaparinux use to patients with a previous history of HIT who require anticoagulation for an acute thrombotic event unrelated to HIT (grade 2C recommendation).5
The vitamin K antagonist warfarin is absolutely contraindicated in patients with HIT until the platelet count is at least 150,000/mm3, due to the risk for warfarin-induced skin necrosis and venous gangrene.9,15 If a patient is receiving warfarin at diagnosis, vitamin K (10 mg orally or 5 to 10 mg IV) should be administered.15
The patient should remain on the alternative non-heparin anticoagulant until the platelet count has stabilized at or above 150,000/mm3. Warfarin should then be started at a maximum of 5 mg/d.2,5 The non-heparin anticoagulant and warfarin should be continued until a therapeutic international normalized ratio (INR) is reached and maintained for 48 hours, with a minimum 5-day overlap of the two medications. Once the non-heparin anticoagulant is discontinued, the INR should be reevaluated for remaining within the therapeutic range, as DTIs can elevate the INR.2,5Warfarin should be continued for as long as four weeks, with frequent INR monitoring.15
Patient Education
The presence of PF4 antibodies is transient (50 to 80 days); however, concern persists regarding recurrent antibody development with subsequent heparin use. Thus, an alternative anticoagulant should be used whenever possible. Patients who have been diagnosed with HIT should be advised to inform future health care professionals regarding their need for alternative anticoagulation whenever possible.
Patients should also be made aware that when the risk for DTI-associated bleeding is too great (as in the case of cardiac surgery), heparin remains the anticoagulant of choice.9,15
Conclusion
Heparin-induced thrombocytopenia is a transient development of antibodies to heparin. While the condition carries a high risk for morbidity and mortality, early detection and prompt treatment can greatly reduce the associated risk to life and limb.
References
1. Kanaan AO, Al-Homsi AS. Heparin-induced thrombocytopenia: pathophysiology, diagnosis, and review of pharmacotherapy. J Pharm Pract. 2009;22:149-157.
2. LaMuraglia GM, Houbballah R, Laposata M. The identification and management of heparin-induced thrombocytopenia in the vascular patient. J Vasc Surg. 2012;55:562-570.
3. Rauova L, Zhai L, Kowalska MA, et al. Role of platelet surface PF4 antigenic complexes in heparin-induced thrombocytopenia pathogenesis: diagnostic and therapeutic implications. Blood. 2006;107:2346-2353.
4. Suvarna S, Espinasse B, Qi R, et al. Determinants of PF4/heparin immunogenicity. Blood. 2007;110:4253-4260.
5. Linkins LA, Dans AL, Moores LK, et al. Treatment and prevention of heparin-induced thrombocytopenia: antithrombotic therapy and prevention of thrombosis, 9th ed: American College of Chest Physicians Evidence-Based Clinical Practice Guidelines. Chest. 2012;141(2 suppl):e495S-e530S.
6. Demma LJ, Winkler AM, Levy JH. A diagnosis of heparin-induced thrombocytopenia with combined clinical and laboratory methods in cardiothoracic surgical intensive care unit patients. Anesth Analg. 2011;113:697-702.
7. Demma LJ, Levy JH. Diagnosing heparin-induced thrombocytopenia in cardiac surgical patients: not as easy as you think. Anesth Analg. 2011;112:747-749.
8. Alaraj A, Wallace A, Tesoro E, et al. Heparin-induced thrombocytopenia: diagnosis and management. J Neurointervent Surg. 2010;2:371-378.
9. Arepally GM, Ortel TL. Heparin-induced thrombocytopenia. N Engl J Med. 2006;355:809-817.
10. Sud S, Mittmann N, Cook DJ, et al. Screening and prevention of venous thromboembolism in critically ill patients: a decision analysis and economic evaluation. Am J Resp Crit Care Med. 2011;184:1289-1298.
11. Shaikh N. Heparin-induced thrombocytopenia. J Emerg Trauma Shock. 2011;14:97-102.
12. Cuker A. Heparin-induced thrombocytopenia: present and future. J Thromb Thrombolysis. 2011;31:353-366.
13. Locke CSF, Dooley J, Gerber J. Rates of clinically apparent heparin-induced thrombocytopenia for unfractionated heparin vs low molecular weight heparin in non-surgical patients are low and similar. Thromb J. 2005;3:4.
14. Cuker A. Recent advances in heparin-induced thrombocytopenia. Curr Opin Hematol. 2011;18:315-322.
15. Warkentin TE, Greinacher A, Koster A, Lincoff AM. Treatment and prevention of heparin-induced thrombocytopenia: American College of Chest Physicians Evidence-Based Clinical Practice Guidelines (8th ed). Chest. 2008;133(6 suppl):340S-380S.
16. Junqueira DR, Perini E, Penholati RR, Carvalho MG. Unfractionated heparin versus low molecular weight heparin for avoiding heparin-induced thrombocytopenia in postoperative patients. Cochrane Database Syst Rev. 2012;9:CD007557.
17. Berry C, Tcherniantchouk O, Ley EJ, et al. Overdiagnosis of heparin-induced thrombocytopenia in surgical ICU patients. J Am Coll Surg. 2011;213:10-17.
18. Cuker A. Current and emerging therapeutics for heparin-induced thrombocytopenia. Semin Thromb Hemost. 2012;38:31-37.
19. Warkentin TE. New approaches to the diagnosis of heparin-induced thrombocytopenia. Chest. 2005;127(2 suppl):35S-45S.
20. Fennessy-Cooney M. Heparin-induced thrombocytopenia. Nurse Pract. 2011;36:31-37.
21. Bryant A, Low J, Austin S, Joseph JE. Timely diagnosis and management of heparin-induced thrombocytopenia in a frequent request, low incidence single centre using clinical 4T's score and particle gel immunoassay. Br J Haematol. 2008;143:721-726.
22. Lo GK, Juhl D, Warkentin TE, et al. Evaluation of pretest clinical score (4 T's) for the diagnosis of heparin-induced thrombocytopenia in two clinical settings. J Thromb Haemost. 2006;4:759-765.
23. Wirth SM, Macaulay TE, Armistead JA, et al. Evaluation of a clinical scoring scale to direct early appropriate therapy in heparin-induced thrombocytopenia. J Oncol Pharm Pract. 2010;16:161-166.
24. Yoon JH, Jang IK. Heparin-induced thrombocytopenia in cardiovascular patients: pathophysiology, diagnosis, and treatment. Cardiol Rev. 2011;19:143-153.
25. Bartholomew JR. Heparin-induced thrombocytopenia: 2008 update. Curr Treat Options Cardiovasc Med. 2008;10:117-127.
26. Warkentin TE. Platelet count monitoring and laboratory testing for heparin-induced thrombocytopenia. Arch Pathol Lab Med. 2002;126:1415-1423.
27. Badger NO. Fondaparinux (Arixtra®), a safe alternative for the treatment of patients with heparin-induced thrombocytopenia? J Pharm Pract. 2010;23:235-238.
28. Petros S. Lepirudin in the management of patients with heparin-induced thrombocytopenia. Biologics. 2008;2:481-490.
29. Warkentin TE. How I diagnose and manage HIT. Hematology Am Soc Hematol Educ Program. 2011;2011:143-149.
30. Papadopoulos S, Flynn JD, Lewis DA. Fondaparinux as a treatment option for heparin-induced thrombocytopenia. Pharmacotherapy. 2007; 27:921-926.
Heparin-induced thrombocytopenia (HIT) is an immune-mediated drug reaction that requires prompt detection and treatment in order to minimize patient morbidity and mortality.1 HIT is caused by the development of antibodies to platelet factor 4 (PF4), although it is important to note that not all patients who develop PF4 antibodies will experience the clinical syndrome of HIT.2-4 In fact, about 50% of patients who undergo cardiovascular surgery develop PF4 antibodies, but only 1% to 2% of patients with antibodies actually experience HIT.5-7 There is currently no explanation for the phenomenon of HIT.8
In 2012, with an intent to limit HIT-associated morbidity and mortality, the American College of Chest Physicians (ACCP) unveiled the ninth edition of its evidence-based practice guidelines for the detection of HIT and appropriate treatment.5 Much of the information provided in this article emerged from these guidelines.
Epidemiology
Of the 12 million patients treated each year with either unfractionated heparin (UFH) or low-molecular-weight heparin (LMWH), 600,000 (0.5%) will develop HIT. Among these patients, 300,000 will develop thrombosis, and 90,000 will die. In 2009 alone, the HIT-associated cost to the US health care system was estimated at $100 million.1
As growing numbers of patients require anticoagulation therapy, it becomes increasingly important for clinicians to understand the importance of screening for deep vein thrombosis (DVT), one of the two most common thromboses; the other is pulmonary embolism.9,10 Continuing to administer heparin or warfarin to patients with undetected HIT predisposes them to severe complications, including venous and arterial thromboses and gangrenous skin lesions—which can result in loss of life and/or limb.1,11,12
Risk Factors for HIT
Several factors influence a patient's risk for HIT, including the type and dosing regimen of the heparin being administered. Generally, the risk for HIT is about 10-fold in patients treated with UFH (3% to 5%), compared with those receiving LMWH (0.5%).5,13 The risk for HIT is also greater in patients receiving UFH of bovine origin, compared with those taking porcine-derived UFH.8,14,15
In a recent meta-analysis of postsurgical patients who underwent heparin thromboprophylaxis, those given LMWH had a 76% relative risk reduction for HIT, compared with patients taking UFH.16 The incidence of HIT increases among patients receiving LMWH if they have been treated with UFH within the previous 100 days.9 HIT onset may be delayed for several days in patients given heparin for the first time (or for the first time in several months), whereas previously exposed patients who have already developed antiheparin PF4 antibodies can experience severe HIT within hours.9
Patient-Specific Risk Factors
Certain patient characteristics also have an impact on HIT risk. For example, the risk for HIT is approximately doubled in women, compared with men,1,5,15 and the incidence of HIT is greater in surgical patients than in medical patients.7,17 Among surgical patients, 5% of orthopedic patients have been reported to develop HIT, compared with 3% of cardiac patients and 1% of patients undergoing surgery for vascular illnesses.1 The reasons for these differences are poorly understood, but current theory focuses on the inflammatory response of individual patients and the degree of associated platelet activation.2,12
Patient Presentation and History
The typical patient with HIT presents with a new or progressing thrombosis between days 5 and 14 of heparin therapy (with day 0 representing the day the first dose is administered); thrombosis can be venous or arterial, although venous thrombosis occurs much more frequently.1,5,9,15 As patients rarely remain hospitalized for such a long period, it is imperative that providers in clinic and emergency settings obtain detailed histories for patients who present with thrombocytopenia and/or thrombosis. HIT should be suspected in any such patient whose history shows heparin use within the previous two weeks (even if the drug has been discontinued).15
Two forms of atypical HIT are rapid-onset HIT and delayed-onset HIT. Rapid-onset HIT is defined by a platelet count that falls within 24 hours of exposure to heparin. This form is usually associated with previous heparin exposure (ie, within the previous 100 days, but most commonly within the previous 30 days). Affected patients have already developed circulating antiheparin PF4 antibodies, causing an immediate reaction when the patient is re-exposed to the drug.1,15
The less common delayed-onset HIT occurs in patients in whom heparin has been discontinued for as long as 40 days. Delayed-onset HIT carries the greatest risk for severe thrombosis.1,15
Atypically, a patient may present with bleeding, skin necrosis, venous gangrene, or anaphylaxis,9 but skin necrosis at the site of heparin injection is strongly suggestive of HIT.12However, neither physical signs nor symptoms, nor a thrombotic event is required to make a diagnosis of HIT. In fact, the preference is for a diagnosis to be made before thrombosis formation.5
The major manifestation of HIT is thrombocytopenia itself2,18(see "Laboratory Findings"). Nevertheless, if physical signs and symptoms are evident, they will be related to the thrombosis, and the components of the physical exam will proceed accordingly.
Laboratory Findings
Platelet count monitoring and HIT antibody testing are the laboratory tests most commonly used when HIT is suspected. Although 25% of patients with HIT will experience a thrombotic event before the platelet count falls, monitoring the platelet count is considered the most effective means to identify patients with HIT.5 HIT antibody testing is not recommended unless the health care provider has a strong suspicion for HIT.19
Thrombocytopenia is a common abnormality, especially in hospitalized patients, and its causes are numerous.11 Table 11,5,9 lists the differential diagnosis that the clinician who suspects HIT should consider.
Nevertheless, the ACCP guidelines5 recommend platelet count monitoring for all patients receiving heparin, beginning on day 4 of heparin therapy, then continuing every two to three days until treatment day 14 or heparin discontinuation, whichever occurs first.5,15 A platelet count decrease of 50% or more should raise a suspicion for HIT.15
The ACCP makes two principal exceptions to these recommendations.5 The first involves patients who have received UFH within the previous 100 days. These patients should undergo a baseline platelet count before heparin is administered, followed by a repeat platelet count within 24 hours. Any patient who experiences an anaphylactic reaction to UFH should undergo an immediate platelet count; a decrease in these patients is often transient.
The second exception pertains to medical and obstetric patients. Those receiving LMWH or UFH only to maintain line patency do not require platelet monitoring, as their risk for HIT is relatively low.1,5
Laboratory Interpretation
In HIT, thrombocytopenia is defined as a platelet count below 150,000/mm3 or a platelet count reduction of 50% or more from baseline, even if the platelet count remains above 150,000/mm3.9 (The patient's baseline platelet count is defined as the highest count recorded in the previous two weeks.1,5,15) The thrombocytopenia associated with HIT is rarely severe and can be easily overlooked.1
Once the platelet count suggests a diagnosis of HIT, heparin-dependent antibodies can be identified through immunologic or functional assays.1,9,15 Immunologic assays should be ordered immediately upon suspicion of HIT since they are simple tests with relatively rapid results. Immunologic assays detect immunoglobulin G (IgG), IgA, and IgM antibodies.9Though lacking in specificity, the immunologic assay is highly sensitive.2,12,20 The most frequently used immunologic assay is the enzyme-linked immunosorbent assay (ELISA).1,2,9,20The ELISA, which detects antiheparin PF4 antibodies, has a sensitivity greater than 97% but a specificity of only 74% to 86%.8,12
Functional assays,which are technically demanding, test the ability of PF4 antibodies to activate platelets in the presence of heparin. The functional assay is used to confirm the diagnosis of HIT when a positive ELISA result is obtained.1,9,15 Among the functional assays, the serotonin release assay (SRA) has been most completely studied. Though very expensive, the SRA is 89% to 100% specific in diagnosing HIT.2,12
Diagnosis
The diagnosis of HIT is determined by combining clinical and serologic assessment. HIT should be suspected in any patient who is in day 5 to 14 of heparin therapy and experiences a drop in platelet count of at least 50%, or in whom a new thrombotic event occurs (even if the patient is no longer receiving heparin therapy). The interpretation of all diagnostic information must be made in the context of the patient's clinical probability of HIT.15
A scoring system referred to as the 4Ts (thrombocytopenia, timing of platelet fall, thrombosis or other sequelae, and test interpretation) is used to help determine the patient's probability of HIT5,21,22 (similar to the scoring strategy shown in Table 22,11,23,24).

The patient diagnosed with HIT must be positive for HIT antibodies and meet at least one additional criterion:
• A platelet count decrease of 30% to 50% below baseline, regardless of the actual value
• A venous or arterial thrombosis
• A skin lesion at the heparin injection site; and/or
• An anaphylactic reaction after IV bolus administration of heparin.15
Treatment/Management
The goal in management of HIT is to reduce the likelihood, then the severity, of thrombosis.9 Treatment should be started as soon as HIT is suspected, before laboratory confirmation.25 Treatment for HIT comprises two steps: stopping all exposure to heparin, and administering an alternative, non-heparin anticoagulant.
Discontinuation of Heparin Exposure
Stopping heparin exposure is the mainstay of treatment for HIT. This includes all potential sources of heparin exposure, including "flushes" that may be used to promote patency of central IV catheters, use of UFH-coated catheters, or addition of any heparin to total parenteral nutrition.9,25
Use of a Non-Heparin Anticoagulant
In addition to discontinuation of all heparin exposure, the patient must be started on a non-heparin anticoagulant, whether or not thrombosis is present.25 Forty percent to 50% of patients without thrombosis will develop a thrombosis within 30 days if alternative anticoagulation is not started.12,18,26
The principal choices for a non-heparin anticoagulant are the direct thrombin inhibitors (DTIs): lepirudin, argatroban, and bivalirudin.1,9,15,25,27 DTIs are the treatment of choice for patients with known or suspected HIT. The ACCP dosing guidelines for the DTIs are summarized in Table 3.9,5,15,28

The factor Xa inhibitor fondaparinux, though FDA approved for DVT prophylaxis,11 has not yet been systematically investigated for the treatment of HIT; thus, its use for this indication is considered off-label.29 However, small studies have shown no cross-reactivity between fondaparinux and PF4 antibodies.30 Due to the positive risk/benefit ratio, ease of use, and reduced need for monitoring in patients taking fondaparinux, it is considered an attractive alternative to DTIs that may receive approval in the near future.12,18,20,29
Currently, the ACCP limits its recommendation of fondaparinux use to patients with a previous history of HIT who require anticoagulation for an acute thrombotic event unrelated to HIT (grade 2C recommendation).5
The vitamin K antagonist warfarin is absolutely contraindicated in patients with HIT until the platelet count is at least 150,000/mm3, due to the risk for warfarin-induced skin necrosis and venous gangrene.9,15 If a patient is receiving warfarin at diagnosis, vitamin K (10 mg orally or 5 to 10 mg IV) should be administered.15
The patient should remain on the alternative non-heparin anticoagulant until the platelet count has stabilized at or above 150,000/mm3. Warfarin should then be started at a maximum of 5 mg/d.2,5 The non-heparin anticoagulant and warfarin should be continued until a therapeutic international normalized ratio (INR) is reached and maintained for 48 hours, with a minimum 5-day overlap of the two medications. Once the non-heparin anticoagulant is discontinued, the INR should be reevaluated for remaining within the therapeutic range, as DTIs can elevate the INR.2,5Warfarin should be continued for as long as four weeks, with frequent INR monitoring.15
Patient Education
The presence of PF4 antibodies is transient (50 to 80 days); however, concern persists regarding recurrent antibody development with subsequent heparin use. Thus, an alternative anticoagulant should be used whenever possible. Patients who have been diagnosed with HIT should be advised to inform future health care professionals regarding their need for alternative anticoagulation whenever possible.
Patients should also be made aware that when the risk for DTI-associated bleeding is too great (as in the case of cardiac surgery), heparin remains the anticoagulant of choice.9,15
Conclusion
Heparin-induced thrombocytopenia is a transient development of antibodies to heparin. While the condition carries a high risk for morbidity and mortality, early detection and prompt treatment can greatly reduce the associated risk to life and limb.
References
1. Kanaan AO, Al-Homsi AS. Heparin-induced thrombocytopenia: pathophysiology, diagnosis, and review of pharmacotherapy. J Pharm Pract. 2009;22:149-157.
2. LaMuraglia GM, Houbballah R, Laposata M. The identification and management of heparin-induced thrombocytopenia in the vascular patient. J Vasc Surg. 2012;55:562-570.
3. Rauova L, Zhai L, Kowalska MA, et al. Role of platelet surface PF4 antigenic complexes in heparin-induced thrombocytopenia pathogenesis: diagnostic and therapeutic implications. Blood. 2006;107:2346-2353.
4. Suvarna S, Espinasse B, Qi R, et al. Determinants of PF4/heparin immunogenicity. Blood. 2007;110:4253-4260.
5. Linkins LA, Dans AL, Moores LK, et al. Treatment and prevention of heparin-induced thrombocytopenia: antithrombotic therapy and prevention of thrombosis, 9th ed: American College of Chest Physicians Evidence-Based Clinical Practice Guidelines. Chest. 2012;141(2 suppl):e495S-e530S.
6. Demma LJ, Winkler AM, Levy JH. A diagnosis of heparin-induced thrombocytopenia with combined clinical and laboratory methods in cardiothoracic surgical intensive care unit patients. Anesth Analg. 2011;113:697-702.
7. Demma LJ, Levy JH. Diagnosing heparin-induced thrombocytopenia in cardiac surgical patients: not as easy as you think. Anesth Analg. 2011;112:747-749.
8. Alaraj A, Wallace A, Tesoro E, et al. Heparin-induced thrombocytopenia: diagnosis and management. J Neurointervent Surg. 2010;2:371-378.
9. Arepally GM, Ortel TL. Heparin-induced thrombocytopenia. N Engl J Med. 2006;355:809-817.
10. Sud S, Mittmann N, Cook DJ, et al. Screening and prevention of venous thromboembolism in critically ill patients: a decision analysis and economic evaluation. Am J Resp Crit Care Med. 2011;184:1289-1298.
11. Shaikh N. Heparin-induced thrombocytopenia. J Emerg Trauma Shock. 2011;14:97-102.
12. Cuker A. Heparin-induced thrombocytopenia: present and future. J Thromb Thrombolysis. 2011;31:353-366.
13. Locke CSF, Dooley J, Gerber J. Rates of clinically apparent heparin-induced thrombocytopenia for unfractionated heparin vs low molecular weight heparin in non-surgical patients are low and similar. Thromb J. 2005;3:4.
14. Cuker A. Recent advances in heparin-induced thrombocytopenia. Curr Opin Hematol. 2011;18:315-322.
15. Warkentin TE, Greinacher A, Koster A, Lincoff AM. Treatment and prevention of heparin-induced thrombocytopenia: American College of Chest Physicians Evidence-Based Clinical Practice Guidelines (8th ed). Chest. 2008;133(6 suppl):340S-380S.
16. Junqueira DR, Perini E, Penholati RR, Carvalho MG. Unfractionated heparin versus low molecular weight heparin for avoiding heparin-induced thrombocytopenia in postoperative patients. Cochrane Database Syst Rev. 2012;9:CD007557.
17. Berry C, Tcherniantchouk O, Ley EJ, et al. Overdiagnosis of heparin-induced thrombocytopenia in surgical ICU patients. J Am Coll Surg. 2011;213:10-17.
18. Cuker A. Current and emerging therapeutics for heparin-induced thrombocytopenia. Semin Thromb Hemost. 2012;38:31-37.
19. Warkentin TE. New approaches to the diagnosis of heparin-induced thrombocytopenia. Chest. 2005;127(2 suppl):35S-45S.
20. Fennessy-Cooney M. Heparin-induced thrombocytopenia. Nurse Pract. 2011;36:31-37.
21. Bryant A, Low J, Austin S, Joseph JE. Timely diagnosis and management of heparin-induced thrombocytopenia in a frequent request, low incidence single centre using clinical 4T's score and particle gel immunoassay. Br J Haematol. 2008;143:721-726.
22. Lo GK, Juhl D, Warkentin TE, et al. Evaluation of pretest clinical score (4 T's) for the diagnosis of heparin-induced thrombocytopenia in two clinical settings. J Thromb Haemost. 2006;4:759-765.
23. Wirth SM, Macaulay TE, Armistead JA, et al. Evaluation of a clinical scoring scale to direct early appropriate therapy in heparin-induced thrombocytopenia. J Oncol Pharm Pract. 2010;16:161-166.
24. Yoon JH, Jang IK. Heparin-induced thrombocytopenia in cardiovascular patients: pathophysiology, diagnosis, and treatment. Cardiol Rev. 2011;19:143-153.
25. Bartholomew JR. Heparin-induced thrombocytopenia: 2008 update. Curr Treat Options Cardiovasc Med. 2008;10:117-127.
26. Warkentin TE. Platelet count monitoring and laboratory testing for heparin-induced thrombocytopenia. Arch Pathol Lab Med. 2002;126:1415-1423.
27. Badger NO. Fondaparinux (Arixtra®), a safe alternative for the treatment of patients with heparin-induced thrombocytopenia? J Pharm Pract. 2010;23:235-238.
28. Petros S. Lepirudin in the management of patients with heparin-induced thrombocytopenia. Biologics. 2008;2:481-490.
29. Warkentin TE. How I diagnose and manage HIT. Hematology Am Soc Hematol Educ Program. 2011;2011:143-149.
30. Papadopoulos S, Flynn JD, Lewis DA. Fondaparinux as a treatment option for heparin-induced thrombocytopenia. Pharmacotherapy. 2007; 27:921-926.
Heparin-Induced Thrombocytopenia: Presentation and History
This is the second article in the series, "Heparin-Induced Thrombocytopenia." The remaining articles are Introduction; Diagnosis; Treatment/Management; and References.
Patient Presentation and History
The typical patient with HIT presents with a new or progressing thrombosis between days 5 and 14 of heparin therapy (with day 0 representing the day the first dose is administered); thrombosis can be venous or arterial, although venous thrombosis occurs much more frequently.1,5,9,15 As patients rarely remain hospitalized for such a long period, it is imperative that providers in clinic and emergency settings obtain detailed histories for patients who present with thrombocytopenia and/or thrombosis. HIT should be suspected in any such patient whose history shows heparin use within the previous two weeks (even if the drug has been discontinued).15
Two forms of atypical HIT are rapid-onset HIT and delayed-onset HIT. Rapid-onset HIT is defined by a platelet count that falls within 24 hours of exposure to heparin. This form is usually associated with previous heparin exposure (ie, within the previous 100 days, but most commonly within the previous 30 days). Affected patients have already developed circulating antiheparin PF4 antibodies, causing an immediate reaction when the patient is re-exposed to the drug.1,15
The less common delayed-onset HIT occurs in patients in whom heparin has been discontinued for as long as 40 days. Delayed-onset HIT carries the greatest risk for severe thrombosis.1,15
Physical Examination
Atypically, a patient may present with bleeding, skin necrosis, venous gangrene, or anaphylaxis,9 but skin necrosis at the site of heparin injection is strongly suggestive of HIT.12 However, neither physical signs nor symptoms, nor a thrombotic event is required to make a diagnosis of HIT. In fact, the preference is for a diagnosis to be made before thrombosis formation.5
The major manifestation of HIT is thrombocytopenia itself2,18 (see "Laboratory Findings"). Nevertheless, if physical signs and symptoms are evident, they will be related to the thrombosis, and the components of the physical exam will proceed accordingly.
Laboratory Findings
Platelet count monitoring and HIT antibody testing are the laboratory tests most commonly used when HIT is suspected. Although 25% of patients with HIT will experience a thrombotic event before the platelet count falls, monitoring the platelet count is considered the most effective means to identify patients with HIT.5 HIT antibody testing is not recommended unless the health care provider has a strong suspicion for HIT.19
Thrombocytopenia is a common abnormality, especially in hospitalized patients, and its causes are numerous.11 Table 11,5,9 lists the differential diagnosis that the clinician who suspects HIT should consider.
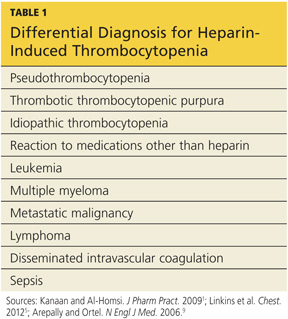
Nevertheless, the ACCP guidelines5 recommend platelet count monitoring for all patients receiving heparin, beginning on day 4 of heparin therapy, then continuing every two to three days until treatment day 14 or heparin discontinuation, whichever occurs first.5,15 A platelet count decrease of 50% or more should raise a suspicion for HIT.15
The ACCP makes two principal exceptions to these recommendations.5 The first involves patients who have received UFH within the previous 100 days. These patients should undergo a baseline platelet count before heparin is administered, followed by a repeat platelet count within 24 hours. Any patient who experiences an anaphylactic reaction to UFH should undergo an immediate platelet count; a decrease in these patients is often transient.
The second exception pertains to medical and obstetric patients. Those receiving LMWH or UFH only to maintain line patency do not require platelet monitoring, as their risk for HIT is relatively low.1,5
Laboratory Interpretation
In HIT, thrombocytopenia is defined as a platelet count below 150,000/mm3 or a platelet count reduction of 50% or more from baseline, even if the platelet count remains above 150,000/mm3.9 (The patient's baseline platelet count is defined as the highest count recorded in the previous two weeks.1,5,15) The thrombocytopenia associated with HIT is rarely severe and can be easily overlooked.1
Once the platelet count suggests a diagnosis of HIT, heparin-dependent antibodies can be identified through immunologic or functional assays.1,9,15 Immunologic assays should be ordered immediately upon suspicion of HIT since they are simple tests with relatively rapid results. Immunologic assays detect immunoglobulin G (IgG), IgA, and IgM antibodies.9 Though lacking in specificity, the immunologic assay is highly sensitive.2,12,20 The most frequently used immunologic assay is the enzyme-linked immunosorbent assay (ELISA).1,2,9,20 The ELISA, which detects antiheparin PF4 antibodies, has a sensitivity greater than 97% but a specificity of only 74% to 86%.8,12
Functional assays,which are technically demanding, test the ability of PF4 antibodies to activate platelets in the presence of heparin. The functional assay is used to confirm the diagnosis of HIT when a positive ELISA result is obtained.1,9,15 Among the functional assays, the serotonin release assay (SRA) has been most completely studied. Though very expensive, the SRA is 89% to 100% specific in diagnosing HIT.2,12
This is the second article in the series, "Heparin-Induced Thrombocytopenia." The remaining articles are Introduction; Diagnosis; Treatment/Management; and References.
This is the second article in the series, "Heparin-Induced Thrombocytopenia." The remaining articles are Introduction; Diagnosis; Treatment/Management; and References.
Patient Presentation and History
The typical patient with HIT presents with a new or progressing thrombosis between days 5 and 14 of heparin therapy (with day 0 representing the day the first dose is administered); thrombosis can be venous or arterial, although venous thrombosis occurs much more frequently.1,5,9,15 As patients rarely remain hospitalized for such a long period, it is imperative that providers in clinic and emergency settings obtain detailed histories for patients who present with thrombocytopenia and/or thrombosis. HIT should be suspected in any such patient whose history shows heparin use within the previous two weeks (even if the drug has been discontinued).15
Two forms of atypical HIT are rapid-onset HIT and delayed-onset HIT. Rapid-onset HIT is defined by a platelet count that falls within 24 hours of exposure to heparin. This form is usually associated with previous heparin exposure (ie, within the previous 100 days, but most commonly within the previous 30 days). Affected patients have already developed circulating antiheparin PF4 antibodies, causing an immediate reaction when the patient is re-exposed to the drug.1,15
The less common delayed-onset HIT occurs in patients in whom heparin has been discontinued for as long as 40 days. Delayed-onset HIT carries the greatest risk for severe thrombosis.1,15
Physical Examination
Atypically, a patient may present with bleeding, skin necrosis, venous gangrene, or anaphylaxis,9 but skin necrosis at the site of heparin injection is strongly suggestive of HIT.12 However, neither physical signs nor symptoms, nor a thrombotic event is required to make a diagnosis of HIT. In fact, the preference is for a diagnosis to be made before thrombosis formation.5
The major manifestation of HIT is thrombocytopenia itself2,18 (see "Laboratory Findings"). Nevertheless, if physical signs and symptoms are evident, they will be related to the thrombosis, and the components of the physical exam will proceed accordingly.
Laboratory Findings
Platelet count monitoring and HIT antibody testing are the laboratory tests most commonly used when HIT is suspected. Although 25% of patients with HIT will experience a thrombotic event before the platelet count falls, monitoring the platelet count is considered the most effective means to identify patients with HIT.5 HIT antibody testing is not recommended unless the health care provider has a strong suspicion for HIT.19
Thrombocytopenia is a common abnormality, especially in hospitalized patients, and its causes are numerous.11 Table 11,5,9 lists the differential diagnosis that the clinician who suspects HIT should consider.

Nevertheless, the ACCP guidelines5 recommend platelet count monitoring for all patients receiving heparin, beginning on day 4 of heparin therapy, then continuing every two to three days until treatment day 14 or heparin discontinuation, whichever occurs first.5,15 A platelet count decrease of 50% or more should raise a suspicion for HIT.15
The ACCP makes two principal exceptions to these recommendations.5 The first involves patients who have received UFH within the previous 100 days. These patients should undergo a baseline platelet count before heparin is administered, followed by a repeat platelet count within 24 hours. Any patient who experiences an anaphylactic reaction to UFH should undergo an immediate platelet count; a decrease in these patients is often transient.
The second exception pertains to medical and obstetric patients. Those receiving LMWH or UFH only to maintain line patency do not require platelet monitoring, as their risk for HIT is relatively low.1,5
Laboratory Interpretation
In HIT, thrombocytopenia is defined as a platelet count below 150,000/mm3 or a platelet count reduction of 50% or more from baseline, even if the platelet count remains above 150,000/mm3.9 (The patient's baseline platelet count is defined as the highest count recorded in the previous two weeks.1,5,15) The thrombocytopenia associated with HIT is rarely severe and can be easily overlooked.1
Once the platelet count suggests a diagnosis of HIT, heparin-dependent antibodies can be identified through immunologic or functional assays.1,9,15 Immunologic assays should be ordered immediately upon suspicion of HIT since they are simple tests with relatively rapid results. Immunologic assays detect immunoglobulin G (IgG), IgA, and IgM antibodies.9 Though lacking in specificity, the immunologic assay is highly sensitive.2,12,20 The most frequently used immunologic assay is the enzyme-linked immunosorbent assay (ELISA).1,2,9,20 The ELISA, which detects antiheparin PF4 antibodies, has a sensitivity greater than 97% but a specificity of only 74% to 86%.8,12
Functional assays,which are technically demanding, test the ability of PF4 antibodies to activate platelets in the presence of heparin. The functional assay is used to confirm the diagnosis of HIT when a positive ELISA result is obtained.1,9,15 Among the functional assays, the serotonin release assay (SRA) has been most completely studied. Though very expensive, the SRA is 89% to 100% specific in diagnosing HIT.2,12
This is the second article in the series, "Heparin-Induced Thrombocytopenia." The remaining articles are Introduction; Diagnosis; Treatment/Management; and References.
This is the second article in the series, "Heparin-Induced Thrombocytopenia." The remaining articles are Introduction; Diagnosis; Treatment/Management; and References.
Patient Presentation and History
The typical patient with HIT presents with a new or progressing thrombosis between days 5 and 14 of heparin therapy (with day 0 representing the day the first dose is administered); thrombosis can be venous or arterial, although venous thrombosis occurs much more frequently.1,5,9,15 As patients rarely remain hospitalized for such a long period, it is imperative that providers in clinic and emergency settings obtain detailed histories for patients who present with thrombocytopenia and/or thrombosis. HIT should be suspected in any such patient whose history shows heparin use within the previous two weeks (even if the drug has been discontinued).15
Two forms of atypical HIT are rapid-onset HIT and delayed-onset HIT. Rapid-onset HIT is defined by a platelet count that falls within 24 hours of exposure to heparin. This form is usually associated with previous heparin exposure (ie, within the previous 100 days, but most commonly within the previous 30 days). Affected patients have already developed circulating antiheparin PF4 antibodies, causing an immediate reaction when the patient is re-exposed to the drug.1,15
The less common delayed-onset HIT occurs in patients in whom heparin has been discontinued for as long as 40 days. Delayed-onset HIT carries the greatest risk for severe thrombosis.1,15
Physical Examination
Atypically, a patient may present with bleeding, skin necrosis, venous gangrene, or anaphylaxis,9 but skin necrosis at the site of heparin injection is strongly suggestive of HIT.12 However, neither physical signs nor symptoms, nor a thrombotic event is required to make a diagnosis of HIT. In fact, the preference is for a diagnosis to be made before thrombosis formation.5
The major manifestation of HIT is thrombocytopenia itself2,18 (see "Laboratory Findings"). Nevertheless, if physical signs and symptoms are evident, they will be related to the thrombosis, and the components of the physical exam will proceed accordingly.
Laboratory Findings
Platelet count monitoring and HIT antibody testing are the laboratory tests most commonly used when HIT is suspected. Although 25% of patients with HIT will experience a thrombotic event before the platelet count falls, monitoring the platelet count is considered the most effective means to identify patients with HIT.5 HIT antibody testing is not recommended unless the health care provider has a strong suspicion for HIT.19
Thrombocytopenia is a common abnormality, especially in hospitalized patients, and its causes are numerous.11 Table 11,5,9 lists the differential diagnosis that the clinician who suspects HIT should consider.

Nevertheless, the ACCP guidelines5 recommend platelet count monitoring for all patients receiving heparin, beginning on day 4 of heparin therapy, then continuing every two to three days until treatment day 14 or heparin discontinuation, whichever occurs first.5,15 A platelet count decrease of 50% or more should raise a suspicion for HIT.15
The ACCP makes two principal exceptions to these recommendations.5 The first involves patients who have received UFH within the previous 100 days. These patients should undergo a baseline platelet count before heparin is administered, followed by a repeat platelet count within 24 hours. Any patient who experiences an anaphylactic reaction to UFH should undergo an immediate platelet count; a decrease in these patients is often transient.
The second exception pertains to medical and obstetric patients. Those receiving LMWH or UFH only to maintain line patency do not require platelet monitoring, as their risk for HIT is relatively low.1,5
Laboratory Interpretation
In HIT, thrombocytopenia is defined as a platelet count below 150,000/mm3 or a platelet count reduction of 50% or more from baseline, even if the platelet count remains above 150,000/mm3.9 (The patient's baseline platelet count is defined as the highest count recorded in the previous two weeks.1,5,15) The thrombocytopenia associated with HIT is rarely severe and can be easily overlooked.1
Once the platelet count suggests a diagnosis of HIT, heparin-dependent antibodies can be identified through immunologic or functional assays.1,9,15 Immunologic assays should be ordered immediately upon suspicion of HIT since they are simple tests with relatively rapid results. Immunologic assays detect immunoglobulin G (IgG), IgA, and IgM antibodies.9 Though lacking in specificity, the immunologic assay is highly sensitive.2,12,20 The most frequently used immunologic assay is the enzyme-linked immunosorbent assay (ELISA).1,2,9,20 The ELISA, which detects antiheparin PF4 antibodies, has a sensitivity greater than 97% but a specificity of only 74% to 86%.8,12
Functional assays,which are technically demanding, test the ability of PF4 antibodies to activate platelets in the presence of heparin. The functional assay is used to confirm the diagnosis of HIT when a positive ELISA result is obtained.1,9,15 Among the functional assays, the serotonin release assay (SRA) has been most completely studied. Though very expensive, the SRA is 89% to 100% specific in diagnosing HIT.2,12
This is the second article in the series, "Heparin-Induced Thrombocytopenia." The remaining articles are Introduction; Diagnosis; Treatment/Management; and References.
Heparin-Induced Thrombocytopenia: Diagnosis
This is the third article in the series, "Heparin-Induced Thrombocytopenia." The remaining articles are Introduction; Presentation and History; Treatment/Management; and References.
Diagnosis
The diagnosis of HIT is determined by combining clinical and serologic assessment. HIT should be suspected in any patient who is in day 5 to 14 of heparin therapy and experiences a drop in platelet count of at least 50%, or in whom a new thrombotic event occurs (even if the patient is no longer receiving heparin therapy). The interpretation of all diagnostic information must be made in the context of the patient's clinical probability of HIT.15
A scoring system referred to as the 4Ts (thrombocytopenia, timing of platelet fall, thrombosis or other sequelae, and test interpretation) is used to help determine the patient's probability of HIT5,21,22 (similar to the scoring strategy shown in Table 22,11,23,24). The patient diagnosed with HIT must be positive for HIT antibodies and meet at least one additional criterion:
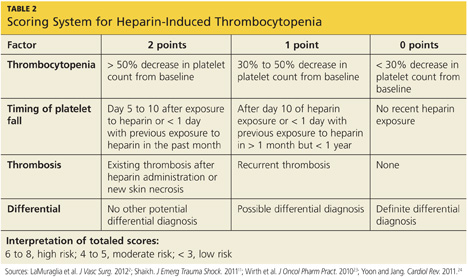
• A platelet count decrease of 30% to 50% below baseline, regardless of the actual value
• A venous or arterial thrombosis
• A skin lesion at the heparin injection site; and/or
• An anaphylactic reaction after IV bolus administration of heparin.15
This is the third article in the series, "Heparin-Induced Thrombocytopenia." The remaining articles are Introduction; Presentation and History; Treatment/Management; and References.
|
This is the third article in the series, "Heparin-Induced Thrombocytopenia." The remaining articles are Introduction; Presentation and History; Treatment/Management; and References.
Diagnosis
The diagnosis of HIT is determined by combining clinical and serologic assessment. HIT should be suspected in any patient who is in day 5 to 14 of heparin therapy and experiences a drop in platelet count of at least 50%, or in whom a new thrombotic event occurs (even if the patient is no longer receiving heparin therapy). The interpretation of all diagnostic information must be made in the context of the patient's clinical probability of HIT.15
A scoring system referred to as the 4Ts (thrombocytopenia, timing of platelet fall, thrombosis or other sequelae, and test interpretation) is used to help determine the patient's probability of HIT5,21,22 (similar to the scoring strategy shown in Table 22,11,23,24). The patient diagnosed with HIT must be positive for HIT antibodies and meet at least one additional criterion:

• A platelet count decrease of 30% to 50% below baseline, regardless of the actual value
• A venous or arterial thrombosis
• A skin lesion at the heparin injection site; and/or
• An anaphylactic reaction after IV bolus administration of heparin.15
This is the third article in the series, "Heparin-Induced Thrombocytopenia." The remaining articles are Introduction; Presentation and History; Treatment/Management; and References.
|
This is the third article in the series, "Heparin-Induced Thrombocytopenia." The remaining articles are Introduction; Presentation and History; Treatment/Management; and References.
Diagnosis
The diagnosis of HIT is determined by combining clinical and serologic assessment. HIT should be suspected in any patient who is in day 5 to 14 of heparin therapy and experiences a drop in platelet count of at least 50%, or in whom a new thrombotic event occurs (even if the patient is no longer receiving heparin therapy). The interpretation of all diagnostic information must be made in the context of the patient's clinical probability of HIT.15
A scoring system referred to as the 4Ts (thrombocytopenia, timing of platelet fall, thrombosis or other sequelae, and test interpretation) is used to help determine the patient's probability of HIT5,21,22 (similar to the scoring strategy shown in Table 22,11,23,24). The patient diagnosed with HIT must be positive for HIT antibodies and meet at least one additional criterion:

• A platelet count decrease of 30% to 50% below baseline, regardless of the actual value
• A venous or arterial thrombosis
• A skin lesion at the heparin injection site; and/or
• An anaphylactic reaction after IV bolus administration of heparin.15
This is the third article in the series, "Heparin-Induced Thrombocytopenia." The remaining articles are Introduction; Presentation and History; Treatment/Management; and References.
|
Heparin-Induced Thrombocytopenia: Treatment/Management
This is the fourth article in the series, "Heparin-Induced Thrombocytopenia." The remaining articles are Introduction; Presentation and History; Diagnosis; and References.
Treatment/Management
The goal in management of HIT is to reduce the likelihood, then the severity, of thrombosis.9 Treatment should be started as soon as HIT is suspected, before laboratory confirmation.25 Treatment for HIT comprises two steps: stopping all exposure to heparin, and administering an alternative, non-heparin anticoagulant.
Discontinuation of Heparin Exposure
Stopping heparin exposure is the mainstay of treatment for HIT. This includes all potential sources of heparin exposure, including "flushes" that may be used to promote patency of central IV catheters, use of UFH-coated catheters, or addition of any heparin to total parenteral nutrition.9,25
Once discontinuation is achieved, the patient's platelet count will begin to rise within two to three days, with a return to baseline within 14 days.1 Because bleeding is rare in patients with HIT, prophylactic administration of platelets is discouraged.2,26 Platelets may be given if there is clinical evidence of bleeding; however, platelet administration has been reported to precipitate thrombosis in patients with HIT.12
If the patient's platelet count remains below baseline after 14 days of heparin abstinence, it is important for the clinician to evaluate the patient for an alternative cause of thrombocytopenia.1
Use of a Non-Heparin Anticoagulant
In addition to discontinuation of all heparin exposure, the patient must be started on a non-heparin anticoagulant, whether or not thrombosis is present.25 Forty percent to 50% of patients without thrombosis will develop a thrombosis within 30 days if alternative anticoagulation is not started.12,18,26
The principal choices for a non-heparin anticoagulant are the direct thrombin inhibitors (DTIs): lepirudin, argatroban, and bivalirudin.1,9,15,25,27 DTIs are the treatment of choice for patients with known or suspected HIT. The ACCP dosing guidelines for the DTIs are summarized in Table 3.9,5,15,28
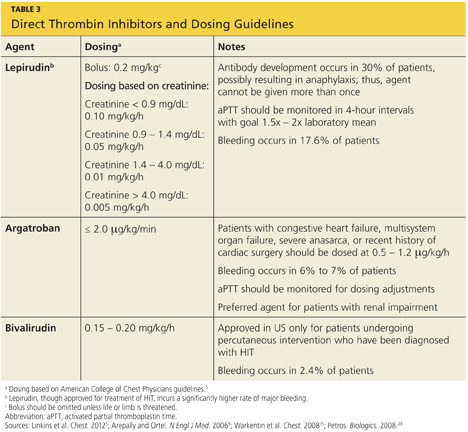
The factor Xa inhibitor fondaparinux, though FDA approved for DVT prophylaxis,11 has not yet been systematically investigated for the treatment of HIT; thus, its use for this indication is considered off-label.29 However, small studies have shown no cross-reactivity between fondaparinux and PF4 antibodies.30 Due to the positive risk/benefit ratio, ease of use, and reduced need for monitoring in patients taking fondaparinux, it is considered an attractive alternative to DTIs that may receive approval in the near future.12,18,20,29
Currently, the ACCP limits its recommendation of fondaparinux use to patients with a previous history of HIT who require anticoagulation for an acute thrombotic event unrelated to HIT (grade 2C recommendation).5
The vitamin K antagonist warfarin is absolutely contraindicated in patients with HIT until the platelet count is at least 150,000/mm3, due to the risk for warfarin-induced skin necrosis and venous gangrene.9,15 If a patient is receiving warfarin at diagnosis, vitamin K (10 mg orally or 5 to 10 mg IV) should be administered.15
The patient should remain on the alternative non-heparin anticoagulant until the platelet count has stabilized at or above 150,000/mm3. Warfarin should then be started at a maximum of 5 mg/d.2,5 The non-heparin anticoagulant and warfarin should be continued until a therapeutic international normalized ratio (INR) is reached and maintained for 48 hours, with a minimum 5-day overlap of the two medications. Once the non-heparin anticoagulant is discontinued, the INR should be reevaluated for remaining within the therapeutic range, as DTIs can elevate the INR.2,5 Warfarin should be continued for as long as four weeks, with frequent INR monitoring.15
Patient Education
The presence of PF4 antibodies is transient (50 to 80 days); however, concern persists regarding recurrent antibody development with subsequent heparin use. Thus, an alternative anticoagulant should be used whenever possible. Patients who have been diagnosed with HIT should be advised to inform future health care professionals regarding their need for alternative anticoagulation whenever possible.
Patients should also be made aware that when the risk for DTI-associated bleeding is too great (as in the case of cardiac surgery), heparin remains the anticoagulant of choice.9,15
Conclusion
Heparin-induced thrombocytopenia is a transient development of antibodies to heparin. While the condition carries a high risk for morbidity and mortality, early detection and prompt treatment can greatly reduce the associated risk to life and limb.
This is the fourth article in the series, "Heparin-Induced Thrombocytopenia." The remaining articles are Introduction; Presentation and History; Diagnosis; and References.
This is the fourth article in the series, "Heparin-Induced Thrombocytopenia." The remaining articles are Introduction; Presentation and History; Diagnosis; and References.
Treatment/Management
The goal in management of HIT is to reduce the likelihood, then the severity, of thrombosis.9 Treatment should be started as soon as HIT is suspected, before laboratory confirmation.25 Treatment for HIT comprises two steps: stopping all exposure to heparin, and administering an alternative, non-heparin anticoagulant.
Discontinuation of Heparin Exposure
Stopping heparin exposure is the mainstay of treatment for HIT. This includes all potential sources of heparin exposure, including "flushes" that may be used to promote patency of central IV catheters, use of UFH-coated catheters, or addition of any heparin to total parenteral nutrition.9,25
Once discontinuation is achieved, the patient's platelet count will begin to rise within two to three days, with a return to baseline within 14 days.1 Because bleeding is rare in patients with HIT, prophylactic administration of platelets is discouraged.2,26 Platelets may be given if there is clinical evidence of bleeding; however, platelet administration has been reported to precipitate thrombosis in patients with HIT.12
If the patient's platelet count remains below baseline after 14 days of heparin abstinence, it is important for the clinician to evaluate the patient for an alternative cause of thrombocytopenia.1
Use of a Non-Heparin Anticoagulant
In addition to discontinuation of all heparin exposure, the patient must be started on a non-heparin anticoagulant, whether or not thrombosis is present.25 Forty percent to 50% of patients without thrombosis will develop a thrombosis within 30 days if alternative anticoagulation is not started.12,18,26
The principal choices for a non-heparin anticoagulant are the direct thrombin inhibitors (DTIs): lepirudin, argatroban, and bivalirudin.1,9,15,25,27 DTIs are the treatment of choice for patients with known or suspected HIT. The ACCP dosing guidelines for the DTIs are summarized in Table 3.9,5,15,28

The factor Xa inhibitor fondaparinux, though FDA approved for DVT prophylaxis,11 has not yet been systematically investigated for the treatment of HIT; thus, its use for this indication is considered off-label.29 However, small studies have shown no cross-reactivity between fondaparinux and PF4 antibodies.30 Due to the positive risk/benefit ratio, ease of use, and reduced need for monitoring in patients taking fondaparinux, it is considered an attractive alternative to DTIs that may receive approval in the near future.12,18,20,29
Currently, the ACCP limits its recommendation of fondaparinux use to patients with a previous history of HIT who require anticoagulation for an acute thrombotic event unrelated to HIT (grade 2C recommendation).5
The vitamin K antagonist warfarin is absolutely contraindicated in patients with HIT until the platelet count is at least 150,000/mm3, due to the risk for warfarin-induced skin necrosis and venous gangrene.9,15 If a patient is receiving warfarin at diagnosis, vitamin K (10 mg orally or 5 to 10 mg IV) should be administered.15
The patient should remain on the alternative non-heparin anticoagulant until the platelet count has stabilized at or above 150,000/mm3. Warfarin should then be started at a maximum of 5 mg/d.2,5 The non-heparin anticoagulant and warfarin should be continued until a therapeutic international normalized ratio (INR) is reached and maintained for 48 hours, with a minimum 5-day overlap of the two medications. Once the non-heparin anticoagulant is discontinued, the INR should be reevaluated for remaining within the therapeutic range, as DTIs can elevate the INR.2,5 Warfarin should be continued for as long as four weeks, with frequent INR monitoring.15
Patient Education
The presence of PF4 antibodies is transient (50 to 80 days); however, concern persists regarding recurrent antibody development with subsequent heparin use. Thus, an alternative anticoagulant should be used whenever possible. Patients who have been diagnosed with HIT should be advised to inform future health care professionals regarding their need for alternative anticoagulation whenever possible.
Patients should also be made aware that when the risk for DTI-associated bleeding is too great (as in the case of cardiac surgery), heparin remains the anticoagulant of choice.9,15
Conclusion
Heparin-induced thrombocytopenia is a transient development of antibodies to heparin. While the condition carries a high risk for morbidity and mortality, early detection and prompt treatment can greatly reduce the associated risk to life and limb.
This is the fourth article in the series, "Heparin-Induced Thrombocytopenia." The remaining articles are Introduction; Presentation and History; Diagnosis; and References.
This is the fourth article in the series, "Heparin-Induced Thrombocytopenia." The remaining articles are Introduction; Presentation and History; Diagnosis; and References.
Treatment/Management
The goal in management of HIT is to reduce the likelihood, then the severity, of thrombosis.9 Treatment should be started as soon as HIT is suspected, before laboratory confirmation.25 Treatment for HIT comprises two steps: stopping all exposure to heparin, and administering an alternative, non-heparin anticoagulant.
Discontinuation of Heparin Exposure
Stopping heparin exposure is the mainstay of treatment for HIT. This includes all potential sources of heparin exposure, including "flushes" that may be used to promote patency of central IV catheters, use of UFH-coated catheters, or addition of any heparin to total parenteral nutrition.9,25
Once discontinuation is achieved, the patient's platelet count will begin to rise within two to three days, with a return to baseline within 14 days.1 Because bleeding is rare in patients with HIT, prophylactic administration of platelets is discouraged.2,26 Platelets may be given if there is clinical evidence of bleeding; however, platelet administration has been reported to precipitate thrombosis in patients with HIT.12
If the patient's platelet count remains below baseline after 14 days of heparin abstinence, it is important for the clinician to evaluate the patient for an alternative cause of thrombocytopenia.1
Use of a Non-Heparin Anticoagulant
In addition to discontinuation of all heparin exposure, the patient must be started on a non-heparin anticoagulant, whether or not thrombosis is present.25 Forty percent to 50% of patients without thrombosis will develop a thrombosis within 30 days if alternative anticoagulation is not started.12,18,26
The principal choices for a non-heparin anticoagulant are the direct thrombin inhibitors (DTIs): lepirudin, argatroban, and bivalirudin.1,9,15,25,27 DTIs are the treatment of choice for patients with known or suspected HIT. The ACCP dosing guidelines for the DTIs are summarized in Table 3.9,5,15,28

The factor Xa inhibitor fondaparinux, though FDA approved for DVT prophylaxis,11 has not yet been systematically investigated for the treatment of HIT; thus, its use for this indication is considered off-label.29 However, small studies have shown no cross-reactivity between fondaparinux and PF4 antibodies.30 Due to the positive risk/benefit ratio, ease of use, and reduced need for monitoring in patients taking fondaparinux, it is considered an attractive alternative to DTIs that may receive approval in the near future.12,18,20,29
Currently, the ACCP limits its recommendation of fondaparinux use to patients with a previous history of HIT who require anticoagulation for an acute thrombotic event unrelated to HIT (grade 2C recommendation).5
The vitamin K antagonist warfarin is absolutely contraindicated in patients with HIT until the platelet count is at least 150,000/mm3, due to the risk for warfarin-induced skin necrosis and venous gangrene.9,15 If a patient is receiving warfarin at diagnosis, vitamin K (10 mg orally or 5 to 10 mg IV) should be administered.15
The patient should remain on the alternative non-heparin anticoagulant until the platelet count has stabilized at or above 150,000/mm3. Warfarin should then be started at a maximum of 5 mg/d.2,5 The non-heparin anticoagulant and warfarin should be continued until a therapeutic international normalized ratio (INR) is reached and maintained for 48 hours, with a minimum 5-day overlap of the two medications. Once the non-heparin anticoagulant is discontinued, the INR should be reevaluated for remaining within the therapeutic range, as DTIs can elevate the INR.2,5 Warfarin should be continued for as long as four weeks, with frequent INR monitoring.15
Patient Education
The presence of PF4 antibodies is transient (50 to 80 days); however, concern persists regarding recurrent antibody development with subsequent heparin use. Thus, an alternative anticoagulant should be used whenever possible. Patients who have been diagnosed with HIT should be advised to inform future health care professionals regarding their need for alternative anticoagulation whenever possible.
Patients should also be made aware that when the risk for DTI-associated bleeding is too great (as in the case of cardiac surgery), heparin remains the anticoagulant of choice.9,15
Conclusion
Heparin-induced thrombocytopenia is a transient development of antibodies to heparin. While the condition carries a high risk for morbidity and mortality, early detection and prompt treatment can greatly reduce the associated risk to life and limb.
This is the fourth article in the series, "Heparin-Induced Thrombocytopenia." The remaining articles are Introduction; Presentation and History; Diagnosis; and References.
Heparin-Induced Thrombocytopenia: References
This is the reference list for the series, "Heparin-Induced Thrombocytopenia." The remaining articles are Introduction; Presentation and History; Diagnosis; and Treatment/Management. References 2. LaMuraglia GM, Houbballah R, Laposata M. The identification and management of heparin-induced thrombocytopenia in the vascular patient. J Vasc Surg. 2012;55:562-570. 3. Rauova L, Zhai L, Kowalska MA, et al. Role of platelet surface PF4 antigenic complexes in heparin-induced thrombocytopenia pathogenesis: diagnostic and therapeutic implications. Blood. 2006;107:2346-2353. 4. Suvarna S, Espinasse B, Qi R, et al. Determinants of PF4/heparin immunogenicity. Blood. 2007;110:4253-4260. 5. Linkins LA, Dans AL, Moores LK, et al. Treatment and prevention of heparin-induced thrombocytopenia: antithrombotic therapy and prevention of thrombosis, 9th ed: American College of Chest Physicians Evidence-Based Clinical Practice Guidelines. Chest. 2012;141(2 suppl):e495S-e530S. 6. Demma LJ, Winkler AM, Levy JH. A diagnosis of heparin-induced thrombocytopenia with combined clinical and laboratory methods in cardiothoracic surgical intensive care unit patients. Anesth Analg. 2011;113:697-702. 7. Demma LJ, Levy JH. Diagnosing heparin-induced thrombocytopenia in cardiac surgical patients: not as easy as you think. Anesth Analg. 2011;112:747-749. 8. Alaraj A, Wallace A, Tesoro E, et al. Heparin-induced thrombocytopenia: diagnosis and management. J Neurointervent Surg. 2010;2:371-378. 9. Arepally GM, Ortel TL. Heparin-induced thrombocytopenia. N Engl J Med. 2006;355:809-817. 10. Sud S, Mittmann N, Cook DJ, et al. Screening and prevention of venous thromboembolism in critically ill patients: a decision analysis and economic evaluation. Am J Resp Crit Care Med. 2011;184:1289-1298. 11. Shaikh N. Heparin-induced thrombocytopenia. J Emerg Trauma Shock. 2011;14:97-102. 12. Cuker A. Heparin-induced thrombocytopenia: present and future. J Thromb Thrombolysis. 2011;31:353-366. 13. Locke CSF, Dooley J, Gerber J. Rates of clinically apparent heparin-induced thrombocytopenia for unfractionated heparin vs low molecular weight heparin in non-surgical patients are low and similar. Thromb J. 2005;3:4. 14. Cuker A. Recent advances in heparin-induced thrombocytopenia. Curr Opin Hematol. 2011;18:315-322. 15. Warkentin TE, Greinacher A, Koster A, Lincoff AM. Treatment and prevention of heparin-induced thrombocytopenia: American College of Chest Physicians Evidence-Based Clinical Practice Guidelines (8th ed). Chest. 2008;133(6 suppl):340S-380S. 16. Junqueira DR, Perini E, Penholati RR, Carvalho MG. Unfractionated heparin versus low molecular weight heparin for avoiding heparin-induced thrombocytopenia in postoperative patients. Cochrane Database Syst Rev. 2012;9:CD007557. 17. Berry C, Tcherniantchouk O, Ley EJ, et al. Overdiagnosis of heparin-induced thrombocytopenia in surgical ICU patients. J Am Coll Surg. 2011;213:10-17. 18. Cuker A. Current and emerging therapeutics for heparin-induced thrombocytopenia. Semin Thromb Hemost. 2012;38:31-37. 19. Warkentin TE. New approaches to the diagnosis of heparin-induced thrombocytopenia. Chest. 2005;127(2 suppl):35S-45S. 20. Fennessy-Cooney M. Heparin-induced thrombocytopenia. Nurse Pract. 2011;36:31-37. 21. Bryant A, Low J, Austin S, Joseph JE. Timely diagnosis and management of heparin-induced thrombocytopenia in a frequent request, low incidence single centre using clinical 4T's score and particle gel immunoassay. Br J Haematol. 2008;143:721-726. 22. Lo GK, Juhl D, Warkentin TE, et al. Evaluation of pretest clinical score (4 T's) for the diagnosis of heparin-induced thrombocytopenia in two clinical settings. J Thromb Haemost. 2006;4:759-765. 23. Wirth SM, Macaulay TE, Armistead JA, et al. Evaluation of a clinical scoring scale to direct early appropriate therapy in heparin-induced thrombocytopenia. J Oncol Pharm Pract. 2010;16:161-166. 24. Yoon JH, Jang IK. Heparin-induced thrombocytopenia in cardiovascular patients: pathophysiology, diagnosis, and treatment. Cardiol Rev. 2011;19:143-153. 25. Bartholomew JR. Heparin-induced thrombocytopenia: 2008 update. Curr Treat Options Cardiovasc Med. 2008;10:117-127. 26. Warkentin TE. Platelet count monitoring and laboratory testing for heparin-induced thrombocytopenia. Arch Pathol Lab Med. 2002;126:1415-1423. 27. Badger NO. Fondaparinux (Arixtra®), a safe alternative for the treatment of patients with heparin-induced thrombocytopenia? J Pharm Pract. 2010;23:235-238. 28. Petros S. Lepirudin in the management of patients with heparin-induced thrombocytopenia. Biologics. 2008;2:481-490. 29. Warkentin TE. How I diagnose and manage HIT. Hematology Am Soc Hematol Educ Program. 2011;2011:143-149. 30. Papadopoulos S, Flynn JD, Lewis DA. Fondaparinux as a treatment option for heparin-induced thrombocytopenia. Pharmacotherapy. 2007; 27:921-926. This is the reference list for the series, "Heparin-Induced Thrombocytopenia." The remaining articles are Introduction; Presentation and History; Diagnosis; and Treatment/Management. |
This is the reference list for the series, "Heparin-Induced Thrombocytopenia." The remaining articles are Introduction; Presentation and History; Diagnosis; and Treatment/Management. References 2. LaMuraglia GM, Houbballah R, Laposata M. The identification and management of heparin-induced thrombocytopenia in the vascular patient. J Vasc Surg. 2012;55:562-570. 3. Rauova L, Zhai L, Kowalska MA, et al. Role of platelet surface PF4 antigenic complexes in heparin-induced thrombocytopenia pathogenesis: diagnostic and therapeutic implications. Blood. 2006;107:2346-2353. 4. Suvarna S, Espinasse B, Qi R, et al. Determinants of PF4/heparin immunogenicity. Blood. 2007;110:4253-4260. 5. Linkins LA, Dans AL, Moores LK, et al. Treatment and prevention of heparin-induced thrombocytopenia: antithrombotic therapy and prevention of thrombosis, 9th ed: American College of Chest Physicians Evidence-Based Clinical Practice Guidelines. Chest. 2012;141(2 suppl):e495S-e530S. 6. Demma LJ, Winkler AM, Levy JH. A diagnosis of heparin-induced thrombocytopenia with combined clinical and laboratory methods in cardiothoracic surgical intensive care unit patients. Anesth Analg. 2011;113:697-702. 7. Demma LJ, Levy JH. Diagnosing heparin-induced thrombocytopenia in cardiac surgical patients: not as easy as you think. Anesth Analg. 2011;112:747-749. 8. Alaraj A, Wallace A, Tesoro E, et al. Heparin-induced thrombocytopenia: diagnosis and management. J Neurointervent Surg. 2010;2:371-378. 9. Arepally GM, Ortel TL. Heparin-induced thrombocytopenia. N Engl J Med. 2006;355:809-817. 10. Sud S, Mittmann N, Cook DJ, et al. Screening and prevention of venous thromboembolism in critically ill patients: a decision analysis and economic evaluation. Am J Resp Crit Care Med. 2011;184:1289-1298. 11. Shaikh N. Heparin-induced thrombocytopenia. J Emerg Trauma Shock. 2011;14:97-102. 12. Cuker A. Heparin-induced thrombocytopenia: present and future. J Thromb Thrombolysis. 2011;31:353-366. 13. Locke CSF, Dooley J, Gerber J. Rates of clinically apparent heparin-induced thrombocytopenia for unfractionated heparin vs low molecular weight heparin in non-surgical patients are low and similar. Thromb J. 2005;3:4. 14. Cuker A. Recent advances in heparin-induced thrombocytopenia. Curr Opin Hematol. 2011;18:315-322. 15. Warkentin TE, Greinacher A, Koster A, Lincoff AM. Treatment and prevention of heparin-induced thrombocytopenia: American College of Chest Physicians Evidence-Based Clinical Practice Guidelines (8th ed). Chest. 2008;133(6 suppl):340S-380S. 16. Junqueira DR, Perini E, Penholati RR, Carvalho MG. Unfractionated heparin versus low molecular weight heparin for avoiding heparin-induced thrombocytopenia in postoperative patients. Cochrane Database Syst Rev. 2012;9:CD007557. 17. Berry C, Tcherniantchouk O, Ley EJ, et al. Overdiagnosis of heparin-induced thrombocytopenia in surgical ICU patients. J Am Coll Surg. 2011;213:10-17. 18. Cuker A. Current and emerging therapeutics for heparin-induced thrombocytopenia. Semin Thromb Hemost. 2012;38:31-37. 19. Warkentin TE. New approaches to the diagnosis of heparin-induced thrombocytopenia. Chest. 2005;127(2 suppl):35S-45S. 20. Fennessy-Cooney M. Heparin-induced thrombocytopenia. Nurse Pract. 2011;36:31-37. 21. Bryant A, Low J, Austin S, Joseph JE. Timely diagnosis and management of heparin-induced thrombocytopenia in a frequent request, low incidence single centre using clinical 4T's score and particle gel immunoassay. Br J Haematol. 2008;143:721-726. 22. Lo GK, Juhl D, Warkentin TE, et al. Evaluation of pretest clinical score (4 T's) for the diagnosis of heparin-induced thrombocytopenia in two clinical settings. J Thromb Haemost. 2006;4:759-765. 23. Wirth SM, Macaulay TE, Armistead JA, et al. Evaluation of a clinical scoring scale to direct early appropriate therapy in heparin-induced thrombocytopenia. J Oncol Pharm Pract. 2010;16:161-166. 24. Yoon JH, Jang IK. Heparin-induced thrombocytopenia in cardiovascular patients: pathophysiology, diagnosis, and treatment. Cardiol Rev. 2011;19:143-153. 25. Bartholomew JR. Heparin-induced thrombocytopenia: 2008 update. Curr Treat Options Cardiovasc Med. 2008;10:117-127. 26. Warkentin TE. Platelet count monitoring and laboratory testing for heparin-induced thrombocytopenia. Arch Pathol Lab Med. 2002;126:1415-1423. 27. Badger NO. Fondaparinux (Arixtra®), a safe alternative for the treatment of patients with heparin-induced thrombocytopenia? J Pharm Pract. 2010;23:235-238. 28. Petros S. Lepirudin in the management of patients with heparin-induced thrombocytopenia. Biologics. 2008;2:481-490. 29. Warkentin TE. How I diagnose and manage HIT. Hematology Am Soc Hematol Educ Program. 2011;2011:143-149. 30. Papadopoulos S, Flynn JD, Lewis DA. Fondaparinux as a treatment option for heparin-induced thrombocytopenia. Pharmacotherapy. 2007; 27:921-926. This is the reference list for the series, "Heparin-Induced Thrombocytopenia." The remaining articles are Introduction; Presentation and History; Diagnosis; and Treatment/Management. |
This is the reference list for the series, "Heparin-Induced Thrombocytopenia." The remaining articles are Introduction; Presentation and History; Diagnosis; and Treatment/Management. References 2. LaMuraglia GM, Houbballah R, Laposata M. The identification and management of heparin-induced thrombocytopenia in the vascular patient. J Vasc Surg. 2012;55:562-570. 3. Rauova L, Zhai L, Kowalska MA, et al. Role of platelet surface PF4 antigenic complexes in heparin-induced thrombocytopenia pathogenesis: diagnostic and therapeutic implications. Blood. 2006;107:2346-2353. 4. Suvarna S, Espinasse B, Qi R, et al. Determinants of PF4/heparin immunogenicity. Blood. 2007;110:4253-4260. 5. Linkins LA, Dans AL, Moores LK, et al. Treatment and prevention of heparin-induced thrombocytopenia: antithrombotic therapy and prevention of thrombosis, 9th ed: American College of Chest Physicians Evidence-Based Clinical Practice Guidelines. Chest. 2012;141(2 suppl):e495S-e530S. 6. Demma LJ, Winkler AM, Levy JH. A diagnosis of heparin-induced thrombocytopenia with combined clinical and laboratory methods in cardiothoracic surgical intensive care unit patients. Anesth Analg. 2011;113:697-702. 7. Demma LJ, Levy JH. Diagnosing heparin-induced thrombocytopenia in cardiac surgical patients: not as easy as you think. Anesth Analg. 2011;112:747-749. 8. Alaraj A, Wallace A, Tesoro E, et al. Heparin-induced thrombocytopenia: diagnosis and management. J Neurointervent Surg. 2010;2:371-378. 9. Arepally GM, Ortel TL. Heparin-induced thrombocytopenia. N Engl J Med. 2006;355:809-817. 10. Sud S, Mittmann N, Cook DJ, et al. Screening and prevention of venous thromboembolism in critically ill patients: a decision analysis and economic evaluation. Am J Resp Crit Care Med. 2011;184:1289-1298. 11. Shaikh N. Heparin-induced thrombocytopenia. J Emerg Trauma Shock. 2011;14:97-102. 12. Cuker A. Heparin-induced thrombocytopenia: present and future. J Thromb Thrombolysis. 2011;31:353-366. 13. Locke CSF, Dooley J, Gerber J. Rates of clinically apparent heparin-induced thrombocytopenia for unfractionated heparin vs low molecular weight heparin in non-surgical patients are low and similar. Thromb J. 2005;3:4. 14. Cuker A. Recent advances in heparin-induced thrombocytopenia. Curr Opin Hematol. 2011;18:315-322. 15. Warkentin TE, Greinacher A, Koster A, Lincoff AM. Treatment and prevention of heparin-induced thrombocytopenia: American College of Chest Physicians Evidence-Based Clinical Practice Guidelines (8th ed). Chest. 2008;133(6 suppl):340S-380S. 16. Junqueira DR, Perini E, Penholati RR, Carvalho MG. Unfractionated heparin versus low molecular weight heparin for avoiding heparin-induced thrombocytopenia in postoperative patients. Cochrane Database Syst Rev. 2012;9:CD007557. 17. Berry C, Tcherniantchouk O, Ley EJ, et al. Overdiagnosis of heparin-induced thrombocytopenia in surgical ICU patients. J Am Coll Surg. 2011;213:10-17. 18. Cuker A. Current and emerging therapeutics for heparin-induced thrombocytopenia. Semin Thromb Hemost. 2012;38:31-37. 19. Warkentin TE. New approaches to the diagnosis of heparin-induced thrombocytopenia. Chest. 2005;127(2 suppl):35S-45S. 20. Fennessy-Cooney M. Heparin-induced thrombocytopenia. Nurse Pract. 2011;36:31-37. 21. Bryant A, Low J, Austin S, Joseph JE. Timely diagnosis and management of heparin-induced thrombocytopenia in a frequent request, low incidence single centre using clinical 4T's score and particle gel immunoassay. Br J Haematol. 2008;143:721-726. 22. Lo GK, Juhl D, Warkentin TE, et al. Evaluation of pretest clinical score (4 T's) for the diagnosis of heparin-induced thrombocytopenia in two clinical settings. J Thromb Haemost. 2006;4:759-765. 23. Wirth SM, Macaulay TE, Armistead JA, et al. Evaluation of a clinical scoring scale to direct early appropriate therapy in heparin-induced thrombocytopenia. J Oncol Pharm Pract. 2010;16:161-166. 24. Yoon JH, Jang IK. Heparin-induced thrombocytopenia in cardiovascular patients: pathophysiology, diagnosis, and treatment. Cardiol Rev. 2011;19:143-153. 25. Bartholomew JR. Heparin-induced thrombocytopenia: 2008 update. Curr Treat Options Cardiovasc Med. 2008;10:117-127. 26. Warkentin TE. Platelet count monitoring and laboratory testing for heparin-induced thrombocytopenia. Arch Pathol Lab Med. 2002;126:1415-1423. 27. Badger NO. Fondaparinux (Arixtra®), a safe alternative for the treatment of patients with heparin-induced thrombocytopenia? J Pharm Pract. 2010;23:235-238. 28. Petros S. Lepirudin in the management of patients with heparin-induced thrombocytopenia. Biologics. 2008;2:481-490. 29. Warkentin TE. How I diagnose and manage HIT. Hematology Am Soc Hematol Educ Program. 2011;2011:143-149. 30. Papadopoulos S, Flynn JD, Lewis DA. Fondaparinux as a treatment option for heparin-induced thrombocytopenia. Pharmacotherapy. 2007; 27:921-926. This is the reference list for the series, "Heparin-Induced Thrombocytopenia." The remaining articles are Introduction; Presentation and History; Diagnosis; and Treatment/Management. |
More on Cognitive Screening Tools
Regarding our January 2013 CE/CME activity (Segal-Gidan FI. Cognitive screening tools. Clinician Reviews. 2013;23(1):12-18), Lieutenant Colonel Scott A. Arcand, PA-C, LTC, SP, of the Army National Guard* asked for more information: an assessment of the described tools in terms of six core neuropsychiatric domains; and more about scoring systems for the very useful clock drawing test.
Q: At the end of the section, "Which Test to Use?", the author states that a comprehensive screening instrument would assess six core neuropsychiatric domains, but then does not assess the six instruments discussed in the article against these criteria. I would have found this a very helpful discussion. I also think it would be helpful to have a table that showed which assessments met each criterion. If a meta-analysis were done to evaluate and compare these instruments, it would lead the drive to improved tools.
In the original article, Table 2 ("Brief Cognitive Assessment Instruments") includes a column listing the cognitive functions assessed by each of the six screening tools reviewed/discussed. Each of these components corresponds to one or more neuropsychological domain, and I refer the reader to the referenced paper for a more detailed discussion of this (Cullen et al,1 2007). Two screening tests, the Modified Mini–Mental State Exam (3MS) and the Mini–Mental State Exam (MMSE) include—at least in part—each of the suggested six core neuropsychological domains. These two tests would be considered the most complete according to the standard of the six core neuropsychological domains; however, they also take the most time to administer (10 to 15 minutes), as noted in the original article's Table 2.
The Clinician's Choice
For clinicians deciding which test is most appropriate to use as a screening tool in their practice or for an individual patient, they should weigh several factors, including the length and time to administer, as well as the domains assessed. Table 1, I hope, will more directly address the reader's issue of which neuropsychological domain is assessed in the test items for each of the cognitive screening tests reviewed in the article.

Q: Regarding the clock drawing test (CDT), the author makes two statements: that there are multiple scoring systems for the CDT, and that the CDT has been shown to be highly predictive of driver safety. I would be interested to see at least one of these scoring systems. Additionally, how low a score on the CDT would it take to indicate that a patient is (or could be) unfit for driving?
Scoring systems for the CDT, varying from four- to 20-point scales, evaluate accuracy of components and traits of the drawing, or they categorize errors. In a 2001 overview of various CDT scoring systems, Peter Braunberger2 included a detailed review of two scoring strategies:
A simple scoring system created by Shua et al,3 in which one point is awarded for each of the following:
• Approximate drawing of the clock
• Presence of numbers in the correct sequence
• Correct spatial arrangement of numbers
• Presence of clock hands
• Hands showing the approximate correct time
• Hands depicting the exact time
In the second scoring system, proposed by Shulman et al,4 six different scores are possible (see Table 24).
Driver Safety
Regarding use of the CDT as a predictor of driver safety, Freund and colleagues5 conducted a study of clock drawing and simulator driving assessment. In addition to finding four CDT scoring scales comparable, they discovered that as patients' CDT scores decreased, the number of "driving errors" they made increased. As shown in Table 3,5 the researchers' CDT scoring system focused on time, numbers, and spacing, with a maximum score of seven points.

References
1. Cullen B, O'Neill B, Evans JJ, et al. A review of screening tests for cognitive impairment. J Neurol Neurosurg Psychiatry. 2007;78(8):790-799.
2. Braunberger P. The clock drawing test (2001) www.neurosurvival.ca/ClinicalAssistant/scales/clock_drawing_test.htm. Accessed March 23, 2013.
3. Shua-Haim J, Koppuzha G, Gross J. A simple scoring system for clock drawing in patients with Alzheimer's disease. J Am Geriatr Soc. 1996;44:335.
4. Shulman KI, Gold DP, Cohen CA, Zucchero CA. Clock-drawing and dementia in the community: a longitudinal study. Int J Geriatr Psychiatry. 1993;8:487-496.
5. Freund B, Gravenstein S, Ferris R, et al. Drawing clocks and driving cars. J Gen Intern Med. 2005;20:240-244.
*Scott A. Arcandis currently serving on active duty as a member of the Army National Guard. The statements expressed here are those of the writer and do not constitute official policy of the Department of Defense, the Department of the Army, or the National Guard Bureau.
Regarding our January 2013 CE/CME activity (Segal-Gidan FI. Cognitive screening tools. Clinician Reviews. 2013;23(1):12-18), Lieutenant Colonel Scott A. Arcand, PA-C, LTC, SP, of the Army National Guard* asked for more information: an assessment of the described tools in terms of six core neuropsychiatric domains; and more about scoring systems for the very useful clock drawing test.
Q: At the end of the section, "Which Test to Use?", the author states that a comprehensive screening instrument would assess six core neuropsychiatric domains, but then does not assess the six instruments discussed in the article against these criteria. I would have found this a very helpful discussion. I also think it would be helpful to have a table that showed which assessments met each criterion. If a meta-analysis were done to evaluate and compare these instruments, it would lead the drive to improved tools.
In the original article, Table 2 ("Brief Cognitive Assessment Instruments") includes a column listing the cognitive functions assessed by each of the six screening tools reviewed/discussed. Each of these components corresponds to one or more neuropsychological domain, and I refer the reader to the referenced paper for a more detailed discussion of this (Cullen et al,1 2007). Two screening tests, the Modified Mini–Mental State Exam (3MS) and the Mini–Mental State Exam (MMSE) include—at least in part—each of the suggested six core neuropsychological domains. These two tests would be considered the most complete according to the standard of the six core neuropsychological domains; however, they also take the most time to administer (10 to 15 minutes), as noted in the original article's Table 2.
The Clinician's Choice
For clinicians deciding which test is most appropriate to use as a screening tool in their practice or for an individual patient, they should weigh several factors, including the length and time to administer, as well as the domains assessed. Table 1, I hope, will more directly address the reader's issue of which neuropsychological domain is assessed in the test items for each of the cognitive screening tests reviewed in the article.

Q: Regarding the clock drawing test (CDT), the author makes two statements: that there are multiple scoring systems for the CDT, and that the CDT has been shown to be highly predictive of driver safety. I would be interested to see at least one of these scoring systems. Additionally, how low a score on the CDT would it take to indicate that a patient is (or could be) unfit for driving?
Scoring systems for the CDT, varying from four- to 20-point scales, evaluate accuracy of components and traits of the drawing, or they categorize errors. In a 2001 overview of various CDT scoring systems, Peter Braunberger2 included a detailed review of two scoring strategies:
A simple scoring system created by Shua et al,3 in which one point is awarded for each of the following:
• Approximate drawing of the clock
• Presence of numbers in the correct sequence
• Correct spatial arrangement of numbers
• Presence of clock hands
• Hands showing the approximate correct time
• Hands depicting the exact time
In the second scoring system, proposed by Shulman et al,4 six different scores are possible (see Table 24).

Driver Safety
Regarding use of the CDT as a predictor of driver safety, Freund and colleagues5 conducted a study of clock drawing and simulator driving assessment. In addition to finding four CDT scoring scales comparable, they discovered that as patients' CDT scores decreased, the number of "driving errors" they made increased. As shown in Table 3,5 the researchers' CDT scoring system focused on time, numbers, and spacing, with a maximum score of seven points.

References
1. Cullen B, O'Neill B, Evans JJ, et al. A review of screening tests for cognitive impairment. J Neurol Neurosurg Psychiatry. 2007;78(8):790-799.
2. Braunberger P. The clock drawing test (2001) www.neurosurvival.ca/ClinicalAssistant/scales/clock_drawing_test.htm. Accessed March 23, 2013.
3. Shua-Haim J, Koppuzha G, Gross J. A simple scoring system for clock drawing in patients with Alzheimer's disease. J Am Geriatr Soc. 1996;44:335.
4. Shulman KI, Gold DP, Cohen CA, Zucchero CA. Clock-drawing and dementia in the community: a longitudinal study. Int J Geriatr Psychiatry. 1993;8:487-496.
5. Freund B, Gravenstein S, Ferris R, et al. Drawing clocks and driving cars. J Gen Intern Med. 2005;20:240-244.
*Scott A. Arcandis currently serving on active duty as a member of the Army National Guard. The statements expressed here are those of the writer and do not constitute official policy of the Department of Defense, the Department of the Army, or the National Guard Bureau.
Regarding our January 2013 CE/CME activity (Segal-Gidan FI. Cognitive screening tools. Clinician Reviews. 2013;23(1):12-18), Lieutenant Colonel Scott A. Arcand, PA-C, LTC, SP, of the Army National Guard* asked for more information: an assessment of the described tools in terms of six core neuropsychiatric domains; and more about scoring systems for the very useful clock drawing test.
Q: At the end of the section, "Which Test to Use?", the author states that a comprehensive screening instrument would assess six core neuropsychiatric domains, but then does not assess the six instruments discussed in the article against these criteria. I would have found this a very helpful discussion. I also think it would be helpful to have a table that showed which assessments met each criterion. If a meta-analysis were done to evaluate and compare these instruments, it would lead the drive to improved tools.
In the original article, Table 2 ("Brief Cognitive Assessment Instruments") includes a column listing the cognitive functions assessed by each of the six screening tools reviewed/discussed. Each of these components corresponds to one or more neuropsychological domain, and I refer the reader to the referenced paper for a more detailed discussion of this (Cullen et al,1 2007). Two screening tests, the Modified Mini–Mental State Exam (3MS) and the Mini–Mental State Exam (MMSE) include—at least in part—each of the suggested six core neuropsychological domains. These two tests would be considered the most complete according to the standard of the six core neuropsychological domains; however, they also take the most time to administer (10 to 15 minutes), as noted in the original article's Table 2.
The Clinician's Choice
For clinicians deciding which test is most appropriate to use as a screening tool in their practice or for an individual patient, they should weigh several factors, including the length and time to administer, as well as the domains assessed. Table 1, I hope, will more directly address the reader's issue of which neuropsychological domain is assessed in the test items for each of the cognitive screening tests reviewed in the article.

Q: Regarding the clock drawing test (CDT), the author makes two statements: that there are multiple scoring systems for the CDT, and that the CDT has been shown to be highly predictive of driver safety. I would be interested to see at least one of these scoring systems. Additionally, how low a score on the CDT would it take to indicate that a patient is (or could be) unfit for driving?
Scoring systems for the CDT, varying from four- to 20-point scales, evaluate accuracy of components and traits of the drawing, or they categorize errors. In a 2001 overview of various CDT scoring systems, Peter Braunberger2 included a detailed review of two scoring strategies:
A simple scoring system created by Shua et al,3 in which one point is awarded for each of the following:
• Approximate drawing of the clock
• Presence of numbers in the correct sequence
• Correct spatial arrangement of numbers
• Presence of clock hands
• Hands showing the approximate correct time
• Hands depicting the exact time
In the second scoring system, proposed by Shulman et al,4 six different scores are possible (see Table 24).

Driver Safety
Regarding use of the CDT as a predictor of driver safety, Freund and colleagues5 conducted a study of clock drawing and simulator driving assessment. In addition to finding four CDT scoring scales comparable, they discovered that as patients' CDT scores decreased, the number of "driving errors" they made increased. As shown in Table 3,5 the researchers' CDT scoring system focused on time, numbers, and spacing, with a maximum score of seven points.

References
1. Cullen B, O'Neill B, Evans JJ, et al. A review of screening tests for cognitive impairment. J Neurol Neurosurg Psychiatry. 2007;78(8):790-799.
2. Braunberger P. The clock drawing test (2001) www.neurosurvival.ca/ClinicalAssistant/scales/clock_drawing_test.htm. Accessed March 23, 2013.
3. Shua-Haim J, Koppuzha G, Gross J. A simple scoring system for clock drawing in patients with Alzheimer's disease. J Am Geriatr Soc. 1996;44:335.
4. Shulman KI, Gold DP, Cohen CA, Zucchero CA. Clock-drawing and dementia in the community: a longitudinal study. Int J Geriatr Psychiatry. 1993;8:487-496.
5. Freund B, Gravenstein S, Ferris R, et al. Drawing clocks and driving cars. J Gen Intern Med. 2005;20:240-244.
*Scott A. Arcandis currently serving on active duty as a member of the Army National Guard. The statements expressed here are those of the writer and do not constitute official policy of the Department of Defense, the Department of the Army, or the National Guard Bureau.
UPDATE ON CERVICAL DISEASE
To access 10 recent articles and audiocasts from OBG Management on cervical disease, click here.
Dr. Einstein reports that Montefiore Medical Center has received payment from Roche and Hologic for time he spent as an advisor or educational speaker. In some cases, his travel has been paid for when required for meetings. In addition, Dr. Einstein reports that Montefiore has received grant funding from Roche, Hologic, and Becton-Dickinson for research-related costs of clinical trials that he has been the overall or Montefiore principal investigator.
Dr. Cox reports that he is a consultant to OncoHealth; a member of the Scientific Advisory Boards for Roche and Hologic; a speaker for Roche; and on the Data and Safety Monitoring Board for HPV vaccines for Merck.
Cervical cancer screening is necessarily complex, and guidelines must change fairly frequently as our understanding of the natural history of HPV infection and cervical cancer continues to evolve. Up-to-date guidelines enhance our ability to detect cervical intraepithelial neoplasia and cancer early and manage them appropriately.
In April 2013, the American Society for Colposcopy and Cervical Pathology (ASCCP) updated guidelines for the management of abnormal cervical cytology and cervical cancer precursors for the first time since 2006.1 This update follows new cervical cancer screening guidelines published in 2012 by the ACS/ASCCP/ASCP,2 the USPSTF,3 and the American College of Obstetricians and Gynecologists4 (and reported in OBG Management in June 20125).
For many clinicians, all these modifications amount to a dizzying “sea change” in the way they have been screening and managing patients to prevent cervical cancer. Clinicians often express frustration with the guidelines, both for their complexity and for what seems like all-too-frequent changes. Do they really need to change … again? Do they really need to get even more complex? And what about them is really new?
This article addresses these questions by reviewing the guidelines and their updates in more depth. For a specific answer to the question of “What’s new?” see sidebar below.
The following features of the 2013 ASCCP update to cervical cancer screening guidelines are new:
- The return to “routine” screening is now better defined
- The management of women who have “unsatisfactory” cytology or a specimen lacking endocervical or transformation-zone components now includes the results of HPV testing
- Management guidelines previously used for adolescents (<21 years) now apply to young adult women (<25 years)
- There is now advice on the management of women aged 30 and older who have discordant cotest results, including HPV-positive/cytology-negative findings and HPV-negative/cytology-positive findings of ASC-US or more severe.
Did the guidelines really need to change … again?
Cervical cancer screening tests—be they the Pap test or a human papillomavirus (HPV) test—are not as clear-cut as other tests used to screen for sexually transmitted infections or their effects. We treat a patient whenever her gonorrhea or Chlamydia test is positive, for example. However, other than cytology classified as high-grade (ie, HSIL), which may prompt immediate treatment in women 25 years and older by “see-and-treat” loop electrosurgical excision procedure (LEEP), neither cervical cytology nor HPV testing is sufficiently specific for present disease (cervical intraepithelial neoplasia [CIN] 3 or cancer) to warrant treatment without a diagnostic work-up. That’s because the cause of cervical cancer (infection with HPV) usually does not produce CIN 3 or cancer, and the cell changes that it does produce most often (atypia and koilocytosis) are very common. And other cervical-vaginal changes associated with hormonal fluctuations, tampons, intercourse, and so on, may result in cervical cytologic changes unrelated to HPV and, therefore, do not represent a risk for cervical cancer.
How can we best sort out who needs to be evaluated without under- or overdoing it? When we find CIN, some of which is destined to progress and some not, how do we reduce the risk of overtreatment without increasing the likelihood that some will progress to cancer? If we have treated CIN or adenocarcinoma in situ (AIS), how do we make sure there is no recurrence without risking over-management and potential overtreatment?
The first thing we do is ensure that we use our best clinical judgment and also respect the informed wishes of the patient. Because the guidelines are based on the best available data, and on expert opinion when data are lacking, guidelines developed through a consensus process provide a framework for care that is optimal for most women at each phase of their lives. This knowledge can help the clinician—and often the patient—make the best-informed decisions.
Because only high-risk HPV types cause cervical cancer, testing should be restricted to high-risk (oncogenic) HPV types. Do not test for low-risk HPV types.
The guidelines are intended for use only with HPV tests that have been analytically and clinically validated, as documented by US Food and Drug Administration licensing and approval or by publication in peer-reviewed scientific literature. This distinction is important because management based on results of HPV tests that have not been similarly validated may not result in outcomes intended by these guidelines and may increase the potential for patient harm.
Do guidelines really need to get even more complex?
Consider the myriad management decisions that confront us in the field of cervical cancer screening, and the potential result of each choice. Even when cervical screening involves cytology alone, there are five major categories for abnormal results, each associated with a different level of risk requiring a unique level of management:
- atypical squamous cells – undetermined significance (ASC-US)
- atypical squamous cells – cannot rule out a high-grade lesion (ASC-H)
- atypical glandular cells (AGC)
- low-grade squamous intraepithelial lesion (LSIL)
- high-grade squamous intraepithelial lesion (HSIL).
Add in HPV testing with cervical cytology for women 30 years and older, and there is one more abnormal category—normal Pap/ HPV-positive. And these categories just cover initial management. Also needed are guidelines for appropriate follow-up of women who undergo colposcopy for each abnormal cytologic result when no CIN 2, CIN 3, or AIS is found that requires treatment, as well as guidelines for managing women following treatment when high-grade histology is found.
As our understanding of the natural history of HPV and cervical oncogenesis has increased, it has become clearer that we must further adjust management decisions on the basis of age, essentially creating many parallel sets of guidelines for women aged 21 to 24, 25 to 29, and 30 years and older.
Yes, cervical screening and management are complex. We are fortunate that the Internet and new “apps” for smartphones give us easy access to guidelines for most of the potential combinations of clinical findings and results. The guideline algorithms are available at www.asccp.org, and full explanatory articles are available at www.jlgtd.com and www.greenjournal.org (comprehensive apps are available for download for almost every smartphone device).
Remember, it is impossible to create guidelines for every possible clinical situation, so clinical judgment must always be paramount when applying guidelines to individual patients.1
What are the major changes of the latest set of guidelines and its update?
Massad LS, Einstein MH, Huh WK, et al. 2012 updated consensus guidelines for the management of abnormal cervical cancer screening tests and cancer precursors. Obstet Gynecol. 2013;121(4):829–846. [Also published in J Low Genit Tract Dis. 2013;17(5 Suppl 1):S1–S27.]
Let’s start by focusing on how the experts crafted the 2012 guidelines. New evidence to guide decisions about the management of abnormal screening tests, CIN, and AIS emerged in 2012 from a review of the world literature and from analyses of a large 7-year clinical database (1.4 million women) at the Kaiser Permanente Northern California Medical Care Plan, conducted in collaboration with scientists from the National Cancer Institute.1
Most of the 2006 guidelines remain valid, but new evidence has modified some of the guidelines and created others where gaps existed. Guideline developers recognized that cervical cancer prevention is a process that entails both benefits and potential harms, and that the potential risks cannot be reduced to zero with the strategies currently available. Attempts to achieve zero risk could result in unbalanced harms, including overtreatment.
- Anxiety from an abnormal test that the patient might fear to be a sign of cancer
- Stigma from diagnosis of a ubiquitous sexually transmitted infection (HPV)
- Time and patient expense related to screening and management
- Pain and injury from the procedures and treatment
- Increased risk of premature delivery and pregnancy loss.
Defining acceptable risk levels
Applying the concept of “similar management for similar risks,” guideline developers benchmarked risks to the risks associated with accepted screening and management strategies. Because the 5-year risk for CIN 3+ for a woman with an LSIL Pap finding is about 5.2%, and the recommendation for LSIL is colposcopy, 5.2% was set as the lower limit of the level of risk that provides enough benefit (detection of CIN 3+) to balance the potential harms of colposcopy.1 (See the box on harms above.)
When women return to prolonged screening as follow-up to abnormal cytology or a positive HPV test, acceptable risk was considered to be that approximating the risk for CIN3+ three years after negative cytology or 5 years after negative cotesting—as these risks were considered acceptable to guide recent primary cervical screening guidelines.2-4
To be as precise as possible, experts stratified the guidelines by risk, according to the woman’s age, cytologic diagnosis, and HPV status, including HPV genotyping for types 16 and 18, when tested. Of course, guidelines for management apply only to women who are found to have abnormalities during routine screening.1 Women who experience postcoital or unexplained abnormal vaginal bleeding, pelvic pain, abnormal discharge, or a visible lesion need individualized evaluations.1
Only changes or additions to the guidelines are listed here, so be sure to read the published guidelines and supplemental articles and/or visit the Web sites listed earlier for a review of all the guidelines.
What’s new in managing women with unsatisfactory Pap results?
In general, cytology should be repeated in 2 to 4 months.
If the unsatisfactory Pap test is part of a cotest, then the following strategies are appropriate:
- If the HPV test is positive, either repeating the Pap test or moving directly to colposcopy is acceptable
- If HPV genotyping was reported and is positive for type 16 or 18, colposcopy is indicated.
Colposcopy also is recommended when two consecutive Pap tests are unsatisfactory.
What’s new in managing women with normal cytology but no, or insufficient, endocervical cells/transformation-zone component?
The answer varies by age:
- For women 21 to 29 years – routine screening with cytology in 3 years is recommended
- For women 30 years and older:
- When cotesting is done, the HPV result guides management:
- HPV-negative: routine screening with cotesting in 5 years is preferred
- HPV-positive: either cotesting in 1 year or immediate genotyping is recommended
- If HPV testing was not done, then HPV testing is recommended, with management guided by results.
- When cotesting is done, the HPV result guides management:
What’s new in managing women aged 21 to 24 with abnormal cervical cytology or CIN?
Young women of this age are at high risk for HPV infection but very low risk for cancer. Aggressive management usually involves more harm than benefit, promoting observation. Adolescents are no longer screened; management previously reserved for adolescents is now appropriate for women aged 21 to 24 years.
If the Pap result is:
- ASC-US or LSIL:
- No colposcopy is needed. The Pap test should be repeated annually for 2 years, with colposcopy after 1 year only when the finding is HSIL and after 2 years if ASC-US or LSIL findings persist
- HPV triage for ASC-US is not recommended, but if it is done:
- HPV-negative women should continue routine screening with a Pap test in 3 years
- HPV-positive women should have annual cytology for 2 years, with colposcopy after 1 year only if the result is HSIL and after 2 years if ASC-US or LSIL findings persist.
- ASC-H or HSIL:
- Colposcopy is recommended, but immediate treatment (see-and-treat LEEP) is unacceptable
- Women with no CIN 2 or CIN 3 at colposcopy should be followed with colposcopy and cytology every 6 months for as long as 2 years, until two consecutive Pap tests are negative and no high-grade colposcopic abnormality is observed
- Repeat biopsies are indicated if cytology at 1 year is again ASC-H or HSIL
- Diagnostic excision is recommended if HSIL cytology persists for 2 years.
Changes in the management of histologic findings
If CIN 1 is detected, management depends on the antecedent cytology report:
- If the prior Pap finding was ASC-US or LSIL, observation with annual cytology is recommended
- If the prior Pap finding was ASC-H or HSIL, observation for as long as 24 months is recommended, using both colposcopy and cytology at 6-month intervals, provided the colposcopic examination is adequate and endocervical assessment is negative.
If CIN 2 is detected, observation is preferred but treatment is acceptable (see the guidelines for detailed recommendations).
If CIN 2/CIN 3 (not otherwise differentiated) is detected, either observation or treatment is acceptable (see the guidelines for detailed recommendations).
If CIN 3 is detected in a woman of any age, treatment is indicated.
What’s new in managing women 30 years and older who have discordant cotest results?
Use cotesting management recommendations only for women 30 years and older.
If the finding is:
- HPV-positive/Pap-negative (HPV+/ Pap-), the two options are:
- Repeat cotesting in 1 year, with colposcopy if the finding is again HPV+ or the Pap is ASC-US or more severe (including HPV-/ASC-US), and repeat cotesting in 3 years if results for both the HPV test and the Pap are negative (HPV-/Pap-)
- Genotyping, with colposcopy if HPV 16 or 18 is identified and repeat cotesting in 1 year if both HPV 16 and 18 are negative
- HPV-/ASC-US:
- Repeat the cotest in 3 years
- HPV-/LSIL, the options are:
- Cotesting in 1 year (preferred)
- Colposcopy (acceptable)
- HPV+/LSIL or LSIL/no HPV result:
- Colposcopy
- HPV-/HSIL or HPV-/ASC-H:
- Colposcopy
- HPV-/AGC
- Colposcopy, often with endometrial sampling.
New terminology unifies all lower genital tract HPV intraepithelial neoplasia
Darragh TM, Colgan TJ, Cox JT, et al; LAST Project Work Groups. The Lower Anogenital Squamous Terminology Standardization Project for HPV-associated lesions: background and consensus recommendations from the College of American Pathologists and the American Society for Colposcopy and Cervical Pathology. J Low Genit Tract Dis. 2012;16(3):205–242.
In 2012, the Lower Anogenital Squamous Terminology (LAST) standardization project created new histology terminology for HPV-related lesions of the lower genital tract. The LSIL finding was designated as the all-encompassing term for CIN 1, vaginal intraepithelial neoplasia 1 (VaIN 1), vulvar intraepithelial neoplasia 1 (VIN 1), penile intraepithelial neoplasia 1 (PeIN 1), perianal intraepithelial neoplasia 1 (PAIN 1) and anal intraepithelial neoplasia 1 (AIN 1). Intraepithelial neoplasia (IN) graded 2, 2/3, and 3 from each of these areas is designated HSIL.5
When CIN 2 and CIN 3 can be differentiated, these designations can be reported along with the HSIL diagnosis. However, after thoughtful deliberation, the delegates to the ASCCP consensus conference decided that there is not yet enough outcome data available to determine different management strategies when using the new LAST histopathology terminology. They recommended that, until evidence is available, results reported as histologic (not cytologic) LSIL should be managed as CIN 1, and histologic (not cytologic) HSIL should be managed as CIN 2/CIN 3.
Guidelines for the management of abnormal cervical cytology, CIN, and AIS are necessarily complicated, but they provide the best basis for evidence-based management of these medical challenges. The Web provides easy access to all of the ASCCP guidelines via www.asccp.org, www.jlgtd.com, and www.greenjournal.org.
We want to hear from you! Tell us what you think.
1. Massad LS, Einstein MH, Huh WK, et al. 2012 updated consensus guidelines for the management of abnormal cervical cancer screening tests and cancer precursors. Obstet Gynecol. 2013;121(4):829-846.(Also published in J Low Genit Tract Dis. 2013;17[5 Suppl 1]:S1–S27.)
2. Saslow D, Solomon D, Lawson HW, et al. American Cancer Society, American Society for Colposcopy and Cervical Pathology, American Society for Clinical Pathology screening guidelines for the prevention and early detection of cervical cancer. Am J Clin Pathol. 2012;137(4):516-542.
3. Moyer VA. US Preventive Services Task Force. Screening for cervical cancer: US Preventive Services Task Force recommendation statement. Ann Intern Med. 2012;156(12):880-891, W312.
4. Committee on Practice Bulletins—Gynecology. American College of Obstetricians and Gynecologists. ACOG Practice Bulletin No. 131: Screening for cervical cancer. Obstet Gynecol. 2012;120(5):1222-1238.
5. Yates J. New cervical Ca screening guidelines recommend less frequent assessment. OBG Manage. 2012;24(6):40-44.
To access 10 recent articles and audiocasts from OBG Management on cervical disease, click here.
Dr. Einstein reports that Montefiore Medical Center has received payment from Roche and Hologic for time he spent as an advisor or educational speaker. In some cases, his travel has been paid for when required for meetings. In addition, Dr. Einstein reports that Montefiore has received grant funding from Roche, Hologic, and Becton-Dickinson for research-related costs of clinical trials that he has been the overall or Montefiore principal investigator.
Dr. Cox reports that he is a consultant to OncoHealth; a member of the Scientific Advisory Boards for Roche and Hologic; a speaker for Roche; and on the Data and Safety Monitoring Board for HPV vaccines for Merck.

Cervical cancer screening is necessarily complex, and guidelines must change fairly frequently as our understanding of the natural history of HPV infection and cervical cancer continues to evolve. Up-to-date guidelines enhance our ability to detect cervical intraepithelial neoplasia and cancer early and manage them appropriately.
In April 2013, the American Society for Colposcopy and Cervical Pathology (ASCCP) updated guidelines for the management of abnormal cervical cytology and cervical cancer precursors for the first time since 2006.1 This update follows new cervical cancer screening guidelines published in 2012 by the ACS/ASCCP/ASCP,2 the USPSTF,3 and the American College of Obstetricians and Gynecologists4 (and reported in OBG Management in June 20125).
For many clinicians, all these modifications amount to a dizzying “sea change” in the way they have been screening and managing patients to prevent cervical cancer. Clinicians often express frustration with the guidelines, both for their complexity and for what seems like all-too-frequent changes. Do they really need to change … again? Do they really need to get even more complex? And what about them is really new?
This article addresses these questions by reviewing the guidelines and their updates in more depth. For a specific answer to the question of “What’s new?” see sidebar below.
The following features of the 2013 ASCCP update to cervical cancer screening guidelines are new:
- The return to “routine” screening is now better defined
- The management of women who have “unsatisfactory” cytology or a specimen lacking endocervical or transformation-zone components now includes the results of HPV testing
- Management guidelines previously used for adolescents (<21 years) now apply to young adult women (<25 years)
- There is now advice on the management of women aged 30 and older who have discordant cotest results, including HPV-positive/cytology-negative findings and HPV-negative/cytology-positive findings of ASC-US or more severe.
Did the guidelines really need to change … again?
Cervical cancer screening tests—be they the Pap test or a human papillomavirus (HPV) test—are not as clear-cut as other tests used to screen for sexually transmitted infections or their effects. We treat a patient whenever her gonorrhea or Chlamydia test is positive, for example. However, other than cytology classified as high-grade (ie, HSIL), which may prompt immediate treatment in women 25 years and older by “see-and-treat” loop electrosurgical excision procedure (LEEP), neither cervical cytology nor HPV testing is sufficiently specific for present disease (cervical intraepithelial neoplasia [CIN] 3 or cancer) to warrant treatment without a diagnostic work-up. That’s because the cause of cervical cancer (infection with HPV) usually does not produce CIN 3 or cancer, and the cell changes that it does produce most often (atypia and koilocytosis) are very common. And other cervical-vaginal changes associated with hormonal fluctuations, tampons, intercourse, and so on, may result in cervical cytologic changes unrelated to HPV and, therefore, do not represent a risk for cervical cancer.
How can we best sort out who needs to be evaluated without under- or overdoing it? When we find CIN, some of which is destined to progress and some not, how do we reduce the risk of overtreatment without increasing the likelihood that some will progress to cancer? If we have treated CIN or adenocarcinoma in situ (AIS), how do we make sure there is no recurrence without risking over-management and potential overtreatment?
The first thing we do is ensure that we use our best clinical judgment and also respect the informed wishes of the patient. Because the guidelines are based on the best available data, and on expert opinion when data are lacking, guidelines developed through a consensus process provide a framework for care that is optimal for most women at each phase of their lives. This knowledge can help the clinician—and often the patient—make the best-informed decisions.
Because only high-risk HPV types cause cervical cancer, testing should be restricted to high-risk (oncogenic) HPV types. Do not test for low-risk HPV types.
The guidelines are intended for use only with HPV tests that have been analytically and clinically validated, as documented by US Food and Drug Administration licensing and approval or by publication in peer-reviewed scientific literature. This distinction is important because management based on results of HPV tests that have not been similarly validated may not result in outcomes intended by these guidelines and may increase the potential for patient harm.
Do guidelines really need to get even more complex?
Consider the myriad management decisions that confront us in the field of cervical cancer screening, and the potential result of each choice. Even when cervical screening involves cytology alone, there are five major categories for abnormal results, each associated with a different level of risk requiring a unique level of management:
- atypical squamous cells – undetermined significance (ASC-US)
- atypical squamous cells – cannot rule out a high-grade lesion (ASC-H)
- atypical glandular cells (AGC)
- low-grade squamous intraepithelial lesion (LSIL)
- high-grade squamous intraepithelial lesion (HSIL).
Add in HPV testing with cervical cytology for women 30 years and older, and there is one more abnormal category—normal Pap/ HPV-positive. And these categories just cover initial management. Also needed are guidelines for appropriate follow-up of women who undergo colposcopy for each abnormal cytologic result when no CIN 2, CIN 3, or AIS is found that requires treatment, as well as guidelines for managing women following treatment when high-grade histology is found.
As our understanding of the natural history of HPV and cervical oncogenesis has increased, it has become clearer that we must further adjust management decisions on the basis of age, essentially creating many parallel sets of guidelines for women aged 21 to 24, 25 to 29, and 30 years and older.
Yes, cervical screening and management are complex. We are fortunate that the Internet and new “apps” for smartphones give us easy access to guidelines for most of the potential combinations of clinical findings and results. The guideline algorithms are available at www.asccp.org, and full explanatory articles are available at www.jlgtd.com and www.greenjournal.org (comprehensive apps are available for download for almost every smartphone device).
Remember, it is impossible to create guidelines for every possible clinical situation, so clinical judgment must always be paramount when applying guidelines to individual patients.1
What are the major changes of the latest set of guidelines and its update?
Massad LS, Einstein MH, Huh WK, et al. 2012 updated consensus guidelines for the management of abnormal cervical cancer screening tests and cancer precursors. Obstet Gynecol. 2013;121(4):829–846. [Also published in J Low Genit Tract Dis. 2013;17(5 Suppl 1):S1–S27.]
Let’s start by focusing on how the experts crafted the 2012 guidelines. New evidence to guide decisions about the management of abnormal screening tests, CIN, and AIS emerged in 2012 from a review of the world literature and from analyses of a large 7-year clinical database (1.4 million women) at the Kaiser Permanente Northern California Medical Care Plan, conducted in collaboration with scientists from the National Cancer Institute.1
Most of the 2006 guidelines remain valid, but new evidence has modified some of the guidelines and created others where gaps existed. Guideline developers recognized that cervical cancer prevention is a process that entails both benefits and potential harms, and that the potential risks cannot be reduced to zero with the strategies currently available. Attempts to achieve zero risk could result in unbalanced harms, including overtreatment.
- Anxiety from an abnormal test that the patient might fear to be a sign of cancer
- Stigma from diagnosis of a ubiquitous sexually transmitted infection (HPV)
- Time and patient expense related to screening and management
- Pain and injury from the procedures and treatment
- Increased risk of premature delivery and pregnancy loss.
Defining acceptable risk levels
Applying the concept of “similar management for similar risks,” guideline developers benchmarked risks to the risks associated with accepted screening and management strategies. Because the 5-year risk for CIN 3+ for a woman with an LSIL Pap finding is about 5.2%, and the recommendation for LSIL is colposcopy, 5.2% was set as the lower limit of the level of risk that provides enough benefit (detection of CIN 3+) to balance the potential harms of colposcopy.1 (See the box on harms above.)
When women return to prolonged screening as follow-up to abnormal cytology or a positive HPV test, acceptable risk was considered to be that approximating the risk for CIN3+ three years after negative cytology or 5 years after negative cotesting—as these risks were considered acceptable to guide recent primary cervical screening guidelines.2-4
To be as precise as possible, experts stratified the guidelines by risk, according to the woman’s age, cytologic diagnosis, and HPV status, including HPV genotyping for types 16 and 18, when tested. Of course, guidelines for management apply only to women who are found to have abnormalities during routine screening.1 Women who experience postcoital or unexplained abnormal vaginal bleeding, pelvic pain, abnormal discharge, or a visible lesion need individualized evaluations.1
Only changes or additions to the guidelines are listed here, so be sure to read the published guidelines and supplemental articles and/or visit the Web sites listed earlier for a review of all the guidelines.
What’s new in managing women with unsatisfactory Pap results?
In general, cytology should be repeated in 2 to 4 months.
If the unsatisfactory Pap test is part of a cotest, then the following strategies are appropriate:
- If the HPV test is positive, either repeating the Pap test or moving directly to colposcopy is acceptable
- If HPV genotyping was reported and is positive for type 16 or 18, colposcopy is indicated.
Colposcopy also is recommended when two consecutive Pap tests are unsatisfactory.
What’s new in managing women with normal cytology but no, or insufficient, endocervical cells/transformation-zone component?
The answer varies by age:
- For women 21 to 29 years – routine screening with cytology in 3 years is recommended
- For women 30 years and older:
- When cotesting is done, the HPV result guides management:
- HPV-negative: routine screening with cotesting in 5 years is preferred
- HPV-positive: either cotesting in 1 year or immediate genotyping is recommended
- If HPV testing was not done, then HPV testing is recommended, with management guided by results.
- When cotesting is done, the HPV result guides management:
What’s new in managing women aged 21 to 24 with abnormal cervical cytology or CIN?
Young women of this age are at high risk for HPV infection but very low risk for cancer. Aggressive management usually involves more harm than benefit, promoting observation. Adolescents are no longer screened; management previously reserved for adolescents is now appropriate for women aged 21 to 24 years.
If the Pap result is:
- ASC-US or LSIL:
- No colposcopy is needed. The Pap test should be repeated annually for 2 years, with colposcopy after 1 year only when the finding is HSIL and after 2 years if ASC-US or LSIL findings persist
- HPV triage for ASC-US is not recommended, but if it is done:
- HPV-negative women should continue routine screening with a Pap test in 3 years
- HPV-positive women should have annual cytology for 2 years, with colposcopy after 1 year only if the result is HSIL and after 2 years if ASC-US or LSIL findings persist.
- ASC-H or HSIL:
- Colposcopy is recommended, but immediate treatment (see-and-treat LEEP) is unacceptable
- Women with no CIN 2 or CIN 3 at colposcopy should be followed with colposcopy and cytology every 6 months for as long as 2 years, until two consecutive Pap tests are negative and no high-grade colposcopic abnormality is observed
- Repeat biopsies are indicated if cytology at 1 year is again ASC-H or HSIL
- Diagnostic excision is recommended if HSIL cytology persists for 2 years.
Changes in the management of histologic findings
If CIN 1 is detected, management depends on the antecedent cytology report:
- If the prior Pap finding was ASC-US or LSIL, observation with annual cytology is recommended
- If the prior Pap finding was ASC-H or HSIL, observation for as long as 24 months is recommended, using both colposcopy and cytology at 6-month intervals, provided the colposcopic examination is adequate and endocervical assessment is negative.
If CIN 2 is detected, observation is preferred but treatment is acceptable (see the guidelines for detailed recommendations).
If CIN 2/CIN 3 (not otherwise differentiated) is detected, either observation or treatment is acceptable (see the guidelines for detailed recommendations).
If CIN 3 is detected in a woman of any age, treatment is indicated.
What’s new in managing women 30 years and older who have discordant cotest results?
Use cotesting management recommendations only for women 30 years and older.
If the finding is:
- HPV-positive/Pap-negative (HPV+/ Pap-), the two options are:
- Repeat cotesting in 1 year, with colposcopy if the finding is again HPV+ or the Pap is ASC-US or more severe (including HPV-/ASC-US), and repeat cotesting in 3 years if results for both the HPV test and the Pap are negative (HPV-/Pap-)
- Genotyping, with colposcopy if HPV 16 or 18 is identified and repeat cotesting in 1 year if both HPV 16 and 18 are negative
- HPV-/ASC-US:
- Repeat the cotest in 3 years
- HPV-/LSIL, the options are:
- Cotesting in 1 year (preferred)
- Colposcopy (acceptable)
- HPV+/LSIL or LSIL/no HPV result:
- Colposcopy
- HPV-/HSIL or HPV-/ASC-H:
- Colposcopy
- HPV-/AGC
- Colposcopy, often with endometrial sampling.
New terminology unifies all lower genital tract HPV intraepithelial neoplasia
Darragh TM, Colgan TJ, Cox JT, et al; LAST Project Work Groups. The Lower Anogenital Squamous Terminology Standardization Project for HPV-associated lesions: background and consensus recommendations from the College of American Pathologists and the American Society for Colposcopy and Cervical Pathology. J Low Genit Tract Dis. 2012;16(3):205–242.
In 2012, the Lower Anogenital Squamous Terminology (LAST) standardization project created new histology terminology for HPV-related lesions of the lower genital tract. The LSIL finding was designated as the all-encompassing term for CIN 1, vaginal intraepithelial neoplasia 1 (VaIN 1), vulvar intraepithelial neoplasia 1 (VIN 1), penile intraepithelial neoplasia 1 (PeIN 1), perianal intraepithelial neoplasia 1 (PAIN 1) and anal intraepithelial neoplasia 1 (AIN 1). Intraepithelial neoplasia (IN) graded 2, 2/3, and 3 from each of these areas is designated HSIL.5
When CIN 2 and CIN 3 can be differentiated, these designations can be reported along with the HSIL diagnosis. However, after thoughtful deliberation, the delegates to the ASCCP consensus conference decided that there is not yet enough outcome data available to determine different management strategies when using the new LAST histopathology terminology. They recommended that, until evidence is available, results reported as histologic (not cytologic) LSIL should be managed as CIN 1, and histologic (not cytologic) HSIL should be managed as CIN 2/CIN 3.
Guidelines for the management of abnormal cervical cytology, CIN, and AIS are necessarily complicated, but they provide the best basis for evidence-based management of these medical challenges. The Web provides easy access to all of the ASCCP guidelines via www.asccp.org, www.jlgtd.com, and www.greenjournal.org.
We want to hear from you! Tell us what you think.
To access 10 recent articles and audiocasts from OBG Management on cervical disease, click here.
Dr. Einstein reports that Montefiore Medical Center has received payment from Roche and Hologic for time he spent as an advisor or educational speaker. In some cases, his travel has been paid for when required for meetings. In addition, Dr. Einstein reports that Montefiore has received grant funding from Roche, Hologic, and Becton-Dickinson for research-related costs of clinical trials that he has been the overall or Montefiore principal investigator.
Dr. Cox reports that he is a consultant to OncoHealth; a member of the Scientific Advisory Boards for Roche and Hologic; a speaker for Roche; and on the Data and Safety Monitoring Board for HPV vaccines for Merck.

Cervical cancer screening is necessarily complex, and guidelines must change fairly frequently as our understanding of the natural history of HPV infection and cervical cancer continues to evolve. Up-to-date guidelines enhance our ability to detect cervical intraepithelial neoplasia and cancer early and manage them appropriately.
In April 2013, the American Society for Colposcopy and Cervical Pathology (ASCCP) updated guidelines for the management of abnormal cervical cytology and cervical cancer precursors for the first time since 2006.1 This update follows new cervical cancer screening guidelines published in 2012 by the ACS/ASCCP/ASCP,2 the USPSTF,3 and the American College of Obstetricians and Gynecologists4 (and reported in OBG Management in June 20125).
For many clinicians, all these modifications amount to a dizzying “sea change” in the way they have been screening and managing patients to prevent cervical cancer. Clinicians often express frustration with the guidelines, both for their complexity and for what seems like all-too-frequent changes. Do they really need to change … again? Do they really need to get even more complex? And what about them is really new?
This article addresses these questions by reviewing the guidelines and their updates in more depth. For a specific answer to the question of “What’s new?” see sidebar below.
The following features of the 2013 ASCCP update to cervical cancer screening guidelines are new:
- The return to “routine” screening is now better defined
- The management of women who have “unsatisfactory” cytology or a specimen lacking endocervical or transformation-zone components now includes the results of HPV testing
- Management guidelines previously used for adolescents (<21 years) now apply to young adult women (<25 years)
- There is now advice on the management of women aged 30 and older who have discordant cotest results, including HPV-positive/cytology-negative findings and HPV-negative/cytology-positive findings of ASC-US or more severe.
Did the guidelines really need to change … again?
Cervical cancer screening tests—be they the Pap test or a human papillomavirus (HPV) test—are not as clear-cut as other tests used to screen for sexually transmitted infections or their effects. We treat a patient whenever her gonorrhea or Chlamydia test is positive, for example. However, other than cytology classified as high-grade (ie, HSIL), which may prompt immediate treatment in women 25 years and older by “see-and-treat” loop electrosurgical excision procedure (LEEP), neither cervical cytology nor HPV testing is sufficiently specific for present disease (cervical intraepithelial neoplasia [CIN] 3 or cancer) to warrant treatment without a diagnostic work-up. That’s because the cause of cervical cancer (infection with HPV) usually does not produce CIN 3 or cancer, and the cell changes that it does produce most often (atypia and koilocytosis) are very common. And other cervical-vaginal changes associated with hormonal fluctuations, tampons, intercourse, and so on, may result in cervical cytologic changes unrelated to HPV and, therefore, do not represent a risk for cervical cancer.
How can we best sort out who needs to be evaluated without under- or overdoing it? When we find CIN, some of which is destined to progress and some not, how do we reduce the risk of overtreatment without increasing the likelihood that some will progress to cancer? If we have treated CIN or adenocarcinoma in situ (AIS), how do we make sure there is no recurrence without risking over-management and potential overtreatment?
The first thing we do is ensure that we use our best clinical judgment and also respect the informed wishes of the patient. Because the guidelines are based on the best available data, and on expert opinion when data are lacking, guidelines developed through a consensus process provide a framework for care that is optimal for most women at each phase of their lives. This knowledge can help the clinician—and often the patient—make the best-informed decisions.
Because only high-risk HPV types cause cervical cancer, testing should be restricted to high-risk (oncogenic) HPV types. Do not test for low-risk HPV types.
The guidelines are intended for use only with HPV tests that have been analytically and clinically validated, as documented by US Food and Drug Administration licensing and approval or by publication in peer-reviewed scientific literature. This distinction is important because management based on results of HPV tests that have not been similarly validated may not result in outcomes intended by these guidelines and may increase the potential for patient harm.
Do guidelines really need to get even more complex?
Consider the myriad management decisions that confront us in the field of cervical cancer screening, and the potential result of each choice. Even when cervical screening involves cytology alone, there are five major categories for abnormal results, each associated with a different level of risk requiring a unique level of management:
- atypical squamous cells – undetermined significance (ASC-US)
- atypical squamous cells – cannot rule out a high-grade lesion (ASC-H)
- atypical glandular cells (AGC)
- low-grade squamous intraepithelial lesion (LSIL)
- high-grade squamous intraepithelial lesion (HSIL).
Add in HPV testing with cervical cytology for women 30 years and older, and there is one more abnormal category—normal Pap/ HPV-positive. And these categories just cover initial management. Also needed are guidelines for appropriate follow-up of women who undergo colposcopy for each abnormal cytologic result when no CIN 2, CIN 3, or AIS is found that requires treatment, as well as guidelines for managing women following treatment when high-grade histology is found.
As our understanding of the natural history of HPV and cervical oncogenesis has increased, it has become clearer that we must further adjust management decisions on the basis of age, essentially creating many parallel sets of guidelines for women aged 21 to 24, 25 to 29, and 30 years and older.
Yes, cervical screening and management are complex. We are fortunate that the Internet and new “apps” for smartphones give us easy access to guidelines for most of the potential combinations of clinical findings and results. The guideline algorithms are available at www.asccp.org, and full explanatory articles are available at www.jlgtd.com and www.greenjournal.org (comprehensive apps are available for download for almost every smartphone device).
Remember, it is impossible to create guidelines for every possible clinical situation, so clinical judgment must always be paramount when applying guidelines to individual patients.1
What are the major changes of the latest set of guidelines and its update?
Massad LS, Einstein MH, Huh WK, et al. 2012 updated consensus guidelines for the management of abnormal cervical cancer screening tests and cancer precursors. Obstet Gynecol. 2013;121(4):829–846. [Also published in J Low Genit Tract Dis. 2013;17(5 Suppl 1):S1–S27.]
Let’s start by focusing on how the experts crafted the 2012 guidelines. New evidence to guide decisions about the management of abnormal screening tests, CIN, and AIS emerged in 2012 from a review of the world literature and from analyses of a large 7-year clinical database (1.4 million women) at the Kaiser Permanente Northern California Medical Care Plan, conducted in collaboration with scientists from the National Cancer Institute.1
Most of the 2006 guidelines remain valid, but new evidence has modified some of the guidelines and created others where gaps existed. Guideline developers recognized that cervical cancer prevention is a process that entails both benefits and potential harms, and that the potential risks cannot be reduced to zero with the strategies currently available. Attempts to achieve zero risk could result in unbalanced harms, including overtreatment.
- Anxiety from an abnormal test that the patient might fear to be a sign of cancer
- Stigma from diagnosis of a ubiquitous sexually transmitted infection (HPV)
- Time and patient expense related to screening and management
- Pain and injury from the procedures and treatment
- Increased risk of premature delivery and pregnancy loss.
Defining acceptable risk levels
Applying the concept of “similar management for similar risks,” guideline developers benchmarked risks to the risks associated with accepted screening and management strategies. Because the 5-year risk for CIN 3+ for a woman with an LSIL Pap finding is about 5.2%, and the recommendation for LSIL is colposcopy, 5.2% was set as the lower limit of the level of risk that provides enough benefit (detection of CIN 3+) to balance the potential harms of colposcopy.1 (See the box on harms above.)
When women return to prolonged screening as follow-up to abnormal cytology or a positive HPV test, acceptable risk was considered to be that approximating the risk for CIN3+ three years after negative cytology or 5 years after negative cotesting—as these risks were considered acceptable to guide recent primary cervical screening guidelines.2-4
To be as precise as possible, experts stratified the guidelines by risk, according to the woman’s age, cytologic diagnosis, and HPV status, including HPV genotyping for types 16 and 18, when tested. Of course, guidelines for management apply only to women who are found to have abnormalities during routine screening.1 Women who experience postcoital or unexplained abnormal vaginal bleeding, pelvic pain, abnormal discharge, or a visible lesion need individualized evaluations.1
Only changes or additions to the guidelines are listed here, so be sure to read the published guidelines and supplemental articles and/or visit the Web sites listed earlier for a review of all the guidelines.
What’s new in managing women with unsatisfactory Pap results?
In general, cytology should be repeated in 2 to 4 months.
If the unsatisfactory Pap test is part of a cotest, then the following strategies are appropriate:
- If the HPV test is positive, either repeating the Pap test or moving directly to colposcopy is acceptable
- If HPV genotyping was reported and is positive for type 16 or 18, colposcopy is indicated.
Colposcopy also is recommended when two consecutive Pap tests are unsatisfactory.
What’s new in managing women with normal cytology but no, or insufficient, endocervical cells/transformation-zone component?
The answer varies by age:
- For women 21 to 29 years – routine screening with cytology in 3 years is recommended
- For women 30 years and older:
- When cotesting is done, the HPV result guides management:
- HPV-negative: routine screening with cotesting in 5 years is preferred
- HPV-positive: either cotesting in 1 year or immediate genotyping is recommended
- If HPV testing was not done, then HPV testing is recommended, with management guided by results.
- When cotesting is done, the HPV result guides management:
What’s new in managing women aged 21 to 24 with abnormal cervical cytology or CIN?
Young women of this age are at high risk for HPV infection but very low risk for cancer. Aggressive management usually involves more harm than benefit, promoting observation. Adolescents are no longer screened; management previously reserved for adolescents is now appropriate for women aged 21 to 24 years.
If the Pap result is:
- ASC-US or LSIL:
- No colposcopy is needed. The Pap test should be repeated annually for 2 years, with colposcopy after 1 year only when the finding is HSIL and after 2 years if ASC-US or LSIL findings persist
- HPV triage for ASC-US is not recommended, but if it is done:
- HPV-negative women should continue routine screening with a Pap test in 3 years
- HPV-positive women should have annual cytology for 2 years, with colposcopy after 1 year only if the result is HSIL and after 2 years if ASC-US or LSIL findings persist.
- ASC-H or HSIL:
- Colposcopy is recommended, but immediate treatment (see-and-treat LEEP) is unacceptable
- Women with no CIN 2 or CIN 3 at colposcopy should be followed with colposcopy and cytology every 6 months for as long as 2 years, until two consecutive Pap tests are negative and no high-grade colposcopic abnormality is observed
- Repeat biopsies are indicated if cytology at 1 year is again ASC-H or HSIL
- Diagnostic excision is recommended if HSIL cytology persists for 2 years.
Changes in the management of histologic findings
If CIN 1 is detected, management depends on the antecedent cytology report:
- If the prior Pap finding was ASC-US or LSIL, observation with annual cytology is recommended
- If the prior Pap finding was ASC-H or HSIL, observation for as long as 24 months is recommended, using both colposcopy and cytology at 6-month intervals, provided the colposcopic examination is adequate and endocervical assessment is negative.
If CIN 2 is detected, observation is preferred but treatment is acceptable (see the guidelines for detailed recommendations).
If CIN 2/CIN 3 (not otherwise differentiated) is detected, either observation or treatment is acceptable (see the guidelines for detailed recommendations).
If CIN 3 is detected in a woman of any age, treatment is indicated.
What’s new in managing women 30 years and older who have discordant cotest results?
Use cotesting management recommendations only for women 30 years and older.
If the finding is:
- HPV-positive/Pap-negative (HPV+/ Pap-), the two options are:
- Repeat cotesting in 1 year, with colposcopy if the finding is again HPV+ or the Pap is ASC-US or more severe (including HPV-/ASC-US), and repeat cotesting in 3 years if results for both the HPV test and the Pap are negative (HPV-/Pap-)
- Genotyping, with colposcopy if HPV 16 or 18 is identified and repeat cotesting in 1 year if both HPV 16 and 18 are negative
- HPV-/ASC-US:
- Repeat the cotest in 3 years
- HPV-/LSIL, the options are:
- Cotesting in 1 year (preferred)
- Colposcopy (acceptable)
- HPV+/LSIL or LSIL/no HPV result:
- Colposcopy
- HPV-/HSIL or HPV-/ASC-H:
- Colposcopy
- HPV-/AGC
- Colposcopy, often with endometrial sampling.
New terminology unifies all lower genital tract HPV intraepithelial neoplasia
Darragh TM, Colgan TJ, Cox JT, et al; LAST Project Work Groups. The Lower Anogenital Squamous Terminology Standardization Project for HPV-associated lesions: background and consensus recommendations from the College of American Pathologists and the American Society for Colposcopy and Cervical Pathology. J Low Genit Tract Dis. 2012;16(3):205–242.
In 2012, the Lower Anogenital Squamous Terminology (LAST) standardization project created new histology terminology for HPV-related lesions of the lower genital tract. The LSIL finding was designated as the all-encompassing term for CIN 1, vaginal intraepithelial neoplasia 1 (VaIN 1), vulvar intraepithelial neoplasia 1 (VIN 1), penile intraepithelial neoplasia 1 (PeIN 1), perianal intraepithelial neoplasia 1 (PAIN 1) and anal intraepithelial neoplasia 1 (AIN 1). Intraepithelial neoplasia (IN) graded 2, 2/3, and 3 from each of these areas is designated HSIL.5
When CIN 2 and CIN 3 can be differentiated, these designations can be reported along with the HSIL diagnosis. However, after thoughtful deliberation, the delegates to the ASCCP consensus conference decided that there is not yet enough outcome data available to determine different management strategies when using the new LAST histopathology terminology. They recommended that, until evidence is available, results reported as histologic (not cytologic) LSIL should be managed as CIN 1, and histologic (not cytologic) HSIL should be managed as CIN 2/CIN 3.
Guidelines for the management of abnormal cervical cytology, CIN, and AIS are necessarily complicated, but they provide the best basis for evidence-based management of these medical challenges. The Web provides easy access to all of the ASCCP guidelines via www.asccp.org, www.jlgtd.com, and www.greenjournal.org.
We want to hear from you! Tell us what you think.
1. Massad LS, Einstein MH, Huh WK, et al. 2012 updated consensus guidelines for the management of abnormal cervical cancer screening tests and cancer precursors. Obstet Gynecol. 2013;121(4):829-846.(Also published in J Low Genit Tract Dis. 2013;17[5 Suppl 1]:S1–S27.)
2. Saslow D, Solomon D, Lawson HW, et al. American Cancer Society, American Society for Colposcopy and Cervical Pathology, American Society for Clinical Pathology screening guidelines for the prevention and early detection of cervical cancer. Am J Clin Pathol. 2012;137(4):516-542.
3. Moyer VA. US Preventive Services Task Force. Screening for cervical cancer: US Preventive Services Task Force recommendation statement. Ann Intern Med. 2012;156(12):880-891, W312.
4. Committee on Practice Bulletins—Gynecology. American College of Obstetricians and Gynecologists. ACOG Practice Bulletin No. 131: Screening for cervical cancer. Obstet Gynecol. 2012;120(5):1222-1238.
5. Yates J. New cervical Ca screening guidelines recommend less frequent assessment. OBG Manage. 2012;24(6):40-44.
1. Massad LS, Einstein MH, Huh WK, et al. 2012 updated consensus guidelines for the management of abnormal cervical cancer screening tests and cancer precursors. Obstet Gynecol. 2013;121(4):829-846.(Also published in J Low Genit Tract Dis. 2013;17[5 Suppl 1]:S1–S27.)
2. Saslow D, Solomon D, Lawson HW, et al. American Cancer Society, American Society for Colposcopy and Cervical Pathology, American Society for Clinical Pathology screening guidelines for the prevention and early detection of cervical cancer. Am J Clin Pathol. 2012;137(4):516-542.
3. Moyer VA. US Preventive Services Task Force. Screening for cervical cancer: US Preventive Services Task Force recommendation statement. Ann Intern Med. 2012;156(12):880-891, W312.
4. Committee on Practice Bulletins—Gynecology. American College of Obstetricians and Gynecologists. ACOG Practice Bulletin No. 131: Screening for cervical cancer. Obstet Gynecol. 2012;120(5):1222-1238.
5. Yates J. New cervical Ca screening guidelines recommend less frequent assessment. OBG Manage. 2012;24(6):40-44.