User login
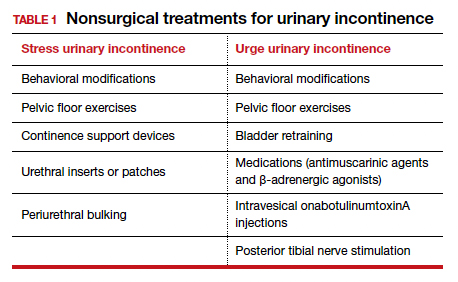
Nonsurgical treatments for patients with urinary incontinence
CASE Patient has urine leakage that worsens with exercise
At her annual preventative health visit, a 39-year-old woman reports that she has leakage of urine. She states that she drinks “a gallon of water daily” to help her lose the 20 lb she gained during the COVID-19 pandemic. She wants to resume Zumba fitness classes, but exercise makes her urine leakage worse. She started wearing protective pads because she finds herself often leaking urine on the way to the bathroom.
What nonsurgical treatment options are available for this patient?
Nearly half of all women experience urinary incontinence (UI), the involuntary loss of urine, and the condition increases with age.1 This common condition negatively impacts physical and psychological health and has been associated with social isolation, sexual dysfunction, and reduced independence.2,3 Symptoms of UI are underreported, and therefore universal screening is recommended for women of all ages.4 The diversity of available treatments (TABLE 1) provides patients and clinicians an opportunity to develop a plan that aligns with their symptom severity, goals, preferences, and resources.
Types of UI
The most common types of UI are stress urinary incontinence (SUI) and urgency urinary incontinence (UUI). Mixed urinary incontinence (MUI) occurs when symptoms of both SUI and UUI are present. Although the mechanisms that lead to urine leakage vary by the type of incontinence, many primary interventions improve both types of leakage, so a clinical diagnosis is sufficient to initiate treatment.
Stress urinary incontinence results from an impaired or weakened sphincter, which leads to involuntary, yet predictable, urine loss during increased abdominal pressure, such as coughing, laughing, sneezing, lifting, or physical activity.5 In UUI, involuntary loss of urine often accompanies the sudden urge to void. UUI is associated with overactive bladder (OAB), defined as urinary urgency, with or without urinary incontinence, usually accompanied by urinary frequency and/or nocturia (urination that interrupts sleep).6
In OAB, the detrusor muscle contracts randomly, leading to a sudden urge to void. When bladder pressure exceeds urethral sphincter closure pressure, urine leakage occurs. Women describe the urgency episodes as unpredictable, the urine leakage as prolonged with large volumes, and often occurring as they seek the toilet. Risk factors include age, obesity, parity, history of vaginal delivery, family history, ethnicity/race, medical comorbidities, menopausal status, and tobacco use.5
Making a diagnosis
A basic office evaluation is the most key step for diagnostic accuracy that leads to treatment success. This includes a detailed history, assessment of symptom severity, physical exam, pelvic exam, urinalysis, postvoid residual (to rule out urinary retention), and a cough stress test (to demonstrate SUI). The goal is to assess symptom severity, determine the type of UI, and identify contributing and potentially reversible factors, such as a urinary tract infection, medications, pelvic organ prolapse, incomplete bladder emptying, or impaired neurologic status. In the absence of the latter, advanced diagnostic tests, such as urodynamics, contribute little toward discerning the type of incontinence or changing first-line treatment plans.7
During the COVID-19 pandemic, abbreviated, virtual assessments for urinary symptoms were associated with high degrees of satisfaction (91% for fulfillment of personal needs, 94% overall satisfaction).8 This highlights the value of validated symptom questionnaires that help establish a working diagnosis and treatment plan in the absence of a physical exam. Questionnaire-based diagnoses have acceptable accuracy for classifying UUI and SUI among women with uncomplicated medical and surgical histories and for initiating low-risk therapies for defined intervals.
The 3 incontinence questions (3IQ) screen is an example of a useful, quick diagnostic tool designed for the primary care setting (FIGURE 1).9 It has been used in pharmaceutical treatment trials for UUI, with low frequency of misdiagnosis (1%–4%), resulting in no harm by the drug treatment prescribed or by the delay in appropriate care.10 Due to the limitations of an abbreviated remote evaluation, however, clinicians should assess patient response to primary interventions in a timely window. Patients who fail to experience satisfactory symptom reduction within 6 to 12 weeks should complete their evaluation in person or through a referral to a urogynecology program.

Continue to: Primary therapies for UI...
Primary therapies for UI
Primary therapies for UUI and SUI target strength training of the pelvic floor muscles, moderation of fluid intake, and adjustment in voiding behaviors and medications. Any functional barriers to continence also should be identified and addressed. Simple interventions, including a daily bowel regimen to address constipation, a bedside commode, and scheduled voiding, may reduce incontinence episodes without incurring significant cost or risk. For women suspected of having MUI, the treatment plan should prioritize their most bothersome symptoms.
Lifestyle and behavioral modifications
Everyday habits, medical comorbidities, and medications may exacerbate the severity of both SUI and UUI. Behavioral therapy alone or in combination with other interventions effectively reduces both SUI and UUI symptoms and has been shown to improve the efficacy of continence surgery.11 Information gained from a 3-day bladder diary (FIGURE 2)12 can guide clinicians on personalized patient recommendations, such as reducing excessive consumption of fluids and bladder irritants, limiting late evening drinking in the setting of bothersome nocturia, and scheduling voids (every 2–3 hours) to preempt incontinence episodes.

Weight loss
Obesity is a strong, independent, modifiable risk factor for both SUI and UUI. Each 5 kg/m2 increase in body mass index (BMI) has been associated with a 20% to 70% increased risk of UI, while weight loss of 5% or greater in overweight or obese women can lead to at least a 50% decrease in UI frequency.13
Reducing fluid intake and bladder irritants
Overactive bladder symptoms often respond to moderation of excessive fluid intake and reduction of bladder irritants (caffeine, carbonated beverages, diet beverages, and alcohol). While there is no established definition of excess caffeine intake, one study categorized high caffeine intake as greater than 400 mg/day (approximately four 8-oz cups of coffee).14
Information provided in a bladder diary can guide individualized recommendations for reducing fluid intake, particularly when 24-hour urine production exceeds the normative range (> 50–60 oz or 1.5-1.8 L/day).15 Hydration needs vary by activity, environment, and food; some general guidelines suggest 48 to 64 oz/day.5,16
Continue to: Pelvic floor muscle training...
Pelvic floor muscle training
An effective treatment for both UUI and SUI symptoms, pelvic floor muscle training (PFMT) leads to high degrees of patient satisfaction and improvement in quality of life.17 The presumed mechanisms of action of PFMT include improved urethral closure pressure and inhibition of detrusor muscle contractions.
Common exercise protocols recommend 3 sets of 10 contractions, held for 6 to 10 seconds per day, in varying positions of sitting, standing, and lying. While many women may be familiar with Kegel exercises, poor technique with straining and recruitment of gluteal and abdominal muscles can undermine the effect of PFMT. Clinicians can confirm successful pelvic muscle contractions by placing a finger in the vagina to appreciate contraction around and elevation of the finger toward the pubic symphysis in the absence of pushing.
Referral to supervised physical therapy and use of such teaching aid tools as booklets, mobile applications, and biofeedback can improve exercise adherence and outcomes.18,19 Systematic reviews report initial cure or improvement of incontinence symptoms as high as 74%, although little information is available about the long-term duration of effect.17
Vaginal pessaries
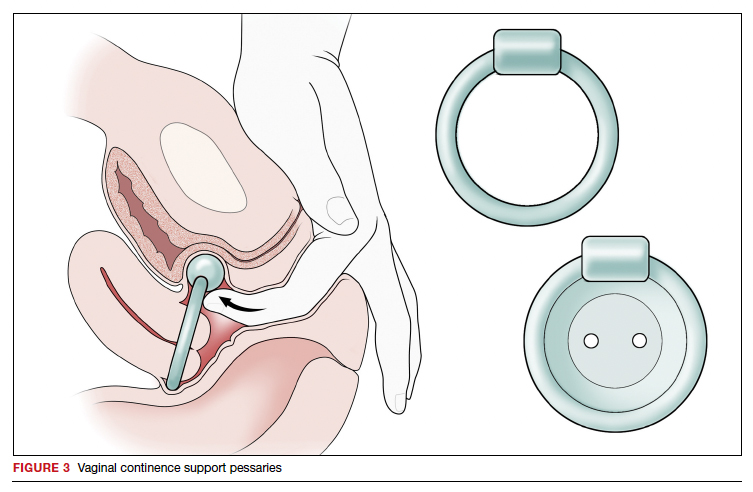
Vaginal continence support pessaries and devices work by stabilizing urethral mobility and compression of the bladder neck. Continence devices are particularly effective for situational SUI (such as during exercise).
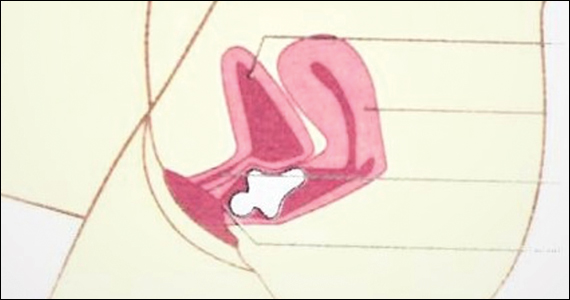
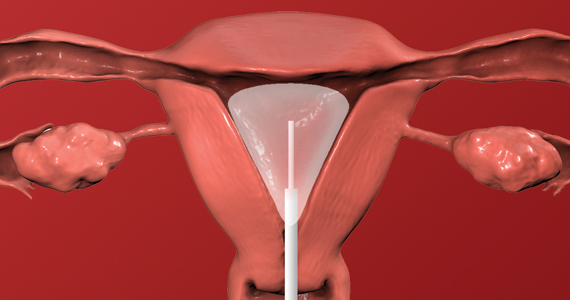
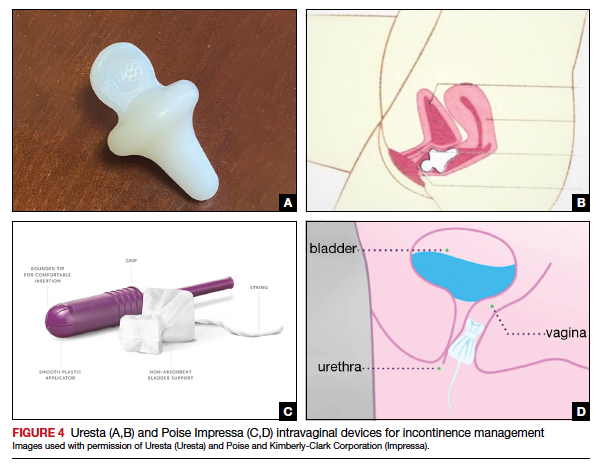
The reusable medical grade silicone pessaries are available in numerous shapes and sizes and are fitted by a health care clinician (FIGURE 3). Uresta is a self-fitted intravaginal device that women can purchase online with a prescription. The Poise Impressa bladder support is a disposable intravaginal device marketed for incontinence and available over-the-counter, without a prescription (FIGURE 4). Anecdotally, many women find that menstrual tampons provide a similar effect, but outcome data are lacking.
In a comparative effectiveness trial of a continence pessary and behavior therapy, behavioral therapy was more likely to result in no bothersome incontinence symptoms (49% vs 33%, P = .006) and greater treatment satisfaction at 3 months.20 However, these short-term group differences did not persist at 12 months, presumably due to waning adherence.
UUI-specific nonsurgical treatments
Drug therapy
All medications approved by the US Food and Drug Administration (FDA) for UI are for the indications of OAB or UUI. These second-line treatments are most effective as adjuncts to behavioral modifications and PFMT.
A multicenter randomized trial that evaluated the efficacy of drug therapy alone compared with drug therapy in combination with behavioral modification, PFMT, urge suppression strategies, timed voiding, and fluid management for UUI found that combined therapy was more successful in achieving greater than 70% reduction in incontinence episodes (58% for drug therapy vs 69% for combined therapy).21
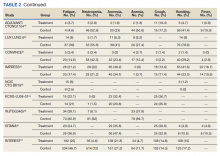
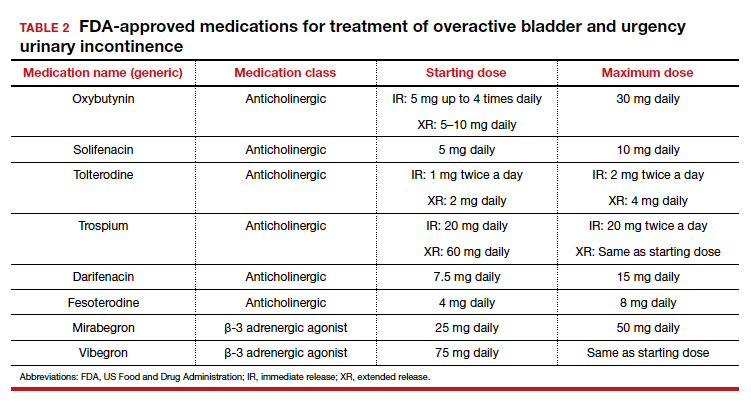
Of the 8 medications currently marketed in the United States for OAB or UUI, 6 are anticholinergic agents that block muscarinic receptors in the smooth muscle of the bladder, leading to inhibition of detrusor contractions, and 2 are β-adrenergic receptor agonists that promote bladder storage capacity by relaxing the detrusor muscle (TABLE 2). Similar efficacies lead most clinicians to initiate drug therapy based on formulary coverage and tolerance for adverse effects. Patients can expect a 53% to 80% reduction in UUI episodes and a 12% to 32% reduction in urinary frequency.22
Extended-release formulations are associated with reduced anticholinergic side effects (dry mouth, constipation, somnolence, dry eyes), leading to improved adherence. Notably, the anticholinergic medications are contraindicated in patients with untreated narrow-angle glaucoma, gastric retention, and supraventricular tachycardia. Mirabegron should be used with caution in patients with poorly controlled hypertension. 5 Due to concerns regarding the association between cumulative anticholinergic burden and the development of dementia, clinicians may consider avoiding the anticholinergic medications in older and at-risk patients.23
Continue to: UUI office-based procedure treatments...
UUI office-based procedure treatments
If behavioral therapies and medications are ineffective, contraindicated, or not the patient’s preference, additional FDA-approved therapies for UUI are available, typically through referral to a urogynecologist, urologist, or continence center.
Posterior tibial nerve stimulation (PTNS) is a nondrug treatment that delivers electrical stimulation using an acupuncture needle for 12 weekly 30-minute sessions followed by monthly maintenance for responders. The time commitment for this treatment plan can be a barrier for some patients. However, patients who adhere to the recommended protocol can expect a 60% improvement in symptoms, with minimal adverse events. Treatment efficacy is comparable to that of anticholinergic medication.24
OnabotulinumtoxinA injections into the bladder muscle are performed cystoscopically under local anesthetic. The toxin blocks the presynaptic release of acetylcholine at the neuromuscular junction, resulting in temporary muscle paralysis. This treatment is associated with high satisfaction. Efficacy varies by study population and outcome measure.
In one US comparative effectiveness trial, 67% of study participants with UUI symptoms refractory to oral medication reported a greater than 50% reduction in OAB symptoms at 6 months, 20% reported complete resolution of UUI, and 72% requested a second injection within 24 months.25 The interval between the first and second injection was nearly 1 year (350 days).Risks include urinary tract infection (12% within 1 month of the procedure and 35% through 6 months); urinary retention requiring catheterization has decreased to 6% with recognition that most moderate retention is tolerated by patients.
Some insurers limit onabotulinumtoxinA treatment coverage to patients who have failed to achieve symptom control with first- and second-line treatments.
SUI-specific nonsurgical treatments
Cystoscopic injection of urethral bulking agents into the urethral submucosa is designed to improve urethral coaptation. It is a minor procedure that can be performed in an ambulatory setting under local anesthetic with or without sedation.
Various bulking agents have been approved for use in the United States, some of which have been withdrawn due to complications of migration, erosion, and pseudoabscess formation. Cure or improvement after bulking agent injection was found to be superior to a home pelvic floor exercise program but inferior to a midurethral sling procedure for cure (9% vs 89%).26
The durability of currently available urethral bulking agents beyond 1 year is unknown. Complications are typically minor and transient and include pain at the injection site, urinary retention, de novo urgency, and implant leakage. The advantages include no postprocedure activity restrictions.
CASE Symptom presentation guides treatment plan
Our patient described symptoms of stress-predominant MUI. She was counseled to moderate her fluid intake to 2 L per day and to strategically time voids (before exercise, and at least every 4 hours). The patient was fitted with an incontinence pessary, and she elected to pursue a course of supervised physical therapy for pelvic floor muscle strengthening. Her follow-up visit is scheduled in 3 months to determine if other interventions are warranted. ●
1. Lee UJ, Feinstein L, Ward JB, et al. Prevalence of urinary incontinence among a nationally representative sample of women, 2005–2016: findings from the Urologic Diseases in America Project. J Urol. 2021;205:1718-1724. doi:10.1097 /JU.0000000000001634
2. Sims J, Browning C, Lundgren-Lindquist B, et al. Urinary incontinence in a community sample of older adults: prevalence and impact on quality of life. Disabil Rehabil. 2011;33:1389-1398. doi:10.3109/09638288.2010.532284
3. Sarikaya S, Yildiz FG, Senocak C, et al. Urinary incontinence as a cause of depression and sexual dysfunction: questionnaire-based study. Rev Int Androl. 2020:18:50-54. doi:10.1016 /j.androl.2018.08.003
4. O’Reilly N, Nelson HD, Conry JM, et al; Women’s Preventive Services Initiative. Screening for urinary incontinence in women: a recommendation from the Women’s Preventive Services Initiative. Ann Intern Med. 2018;169(5):320-328. doi:10.7326/M18-0595
5. Barber MD, Walters MD, Karram MM, et al. Walters & Karram Urogynecology and Reconstructive Pelvic Surgery. 5th ed. Elsevier Saunders; 2021.
6. Haylen BT, de Ridder D, Freeman RM, et al. An International Urogynecological Association (IUGA)/International Continence Society (ICS) joint report on the terminology for female pelvic floor dysfunction. Int Urogynecol J. 2010;21: 5-26. doi:10.1007/s00192-009-0976-9
7. ACOG practice bulletin no. 155. Urinary incontinence in women. Obstet Gynecol. 2015;126:e66-e81. doi:10.1097 /AOG.0000000000001148
8. Sansone S, Lu J, Drangsholt S, et al. No pelvic exam, no problem: patient satisfaction following the integration of comprehensive urogynecology telemedicine. Int Urogynecol J. 2022;1:3. doi:10.1007/s00192-022-05104-w
9. Brown JS, Bradley CS, Subak LL, et al; Diagnostic Aspects of Incontinence Study (DAISy) Research Group. The sensitivity and specificity of a simple test to distinguish between urge and stress urinary incontinence. Ann Intern Med. 2006;144:715723. doi:10.7326/0003-4819-144-10-200605160-00005
10. Hess R, Huang AJ, Richter HE, et al. Long-term efficacy and safety of questionnaire-based initiation of urgency urinary incontinence treatment. Am J Obstet Gynecol. 2013;209:244. e1-9. doi:10.1016/j.ajog.2013.05.008
11. Sung VW, Borello-France D, Newman DK, et al; NICHD Pelvic Floor Disorders Network. Effect of behavioral and pelvic floor muscle therapy combined with surgery vs surgery alone on incontinence symptoms among women with mixed urinary incontinence. JAMA. 2019;322:1066-1076. doi:10.1001 /jama.2019.12467
12. American Urogynecologic Society. Voices for PFD: intake and voiding diary. Accessed August 11, 2022. https://www .voicesforpfd.org/assets/2/6/Voiding_Diary.pdf
13. Subak LL, Richter HE, Hunskaar S. Obesity and urinary incontinence: epidemiology and clinical research update. J Urol. 2009;182(6 suppl):S2-7. doi:10.1016/j.juro.2009.08.071
14. Arya LA, Myers DL, Jackson ND. Dietary caffeine intake and the risk for detrusor instability: a case-control study. Obstet Gynecol. 2000;96:85-89. doi:10.1016/s0029-7844(00)00808-5
15. Wyman JF, Zhou J, LaCoursiere DY, et al. Normative noninvasive bladder function measurements in healthy women: a systematic review and meta-analysis. Neurourol Urodyn. 2020;39:507-522. doi:10.1002/nau.24265
16. Hashim H, Al Mousa R. Management of fluid intake in patients with overactive bladder. Curr Urol Rep. 2009;10: 428-433. doi:10.1007/s11934-009-0068-x
17. Dumoulin C, Cacciari LP, Hay-Smith EJC. Pelvic floor muscle training versus no treatment, or inactive control treatments, for urinary incontinence in women. Cochrane Database Syst Rev. 2018;10:CD005654. doi:10.1002/14651858.CD005654.pub4
18. Araujo CC, de A Marques A, Juliato CRT. The adherence of home pelvic floor muscles training using a mobile device application for women with urinary incontinence: a randomized controlled trial. Female Pelvic Med Reconstr Surg. 2020;26:697-703. doi:10.1097/SPV.0000000000000670
19. Sjöström M, Umefjord G, Stenlund H, et al. Internet-based treatment of stress urinary incontinence: a randomized controlled study with focus on pelvic floor muscle training. BJU Int. 2013;112:362-372. doi:10.1111/j.1464 -410X.2012.11713.x
20. Richter HE, Burgio KL, Brubaker L, et al; Pelvic Floor Disorders Network. Continence pessary compared with behavioral therapy or combined therapy for stress incontinence: a randomized controlled trial. Obstet Gynecol. 2010;115:609617. doi:10.1097/AOG.0b013e3181d055d4
21. Burgio KL, Kraus SR, Menefee S, et al. Behavioral therapy to enable women with urge incontinence to discontinue drug treatment: a randomized trial. Ann Intern Med. 2008;149(3): 161-169. doi:10.7326/0003-4819-149-3-200808050 -00005
22. Lukacz ES, Santiago-Lastra Y, Albo ME, et al. Urinary incontinence in women: a review. JAMA. 2017;318:1592-1604. doi:10.1001/jama.2017.12137
23. Welk B, Richardson K, Panicker JN. The cognitive effect of anticholinergics for patients with overactive bladder. Nat Rev Urol. 2021;18:686-700. doi:10.1038/s41585-021-00504-x
24. Burton C, Sajja A, Latthe PM. Effectiveness of percutaneous posterior tibial nerve stimulation for overactive bladder: a systematic review and meta-analysis. Neurourol Urodyn. 2012;31:1206-1216. doi:10.1002/nau.22251
25. Amundsen CL, Richter HE, Menefee SA, et al. OnabotulinumtoxinA vs sacral neuromodulation on refractory urgency urinary incontinence in women: A randomized clinical trial. JAMA. 2016;316:1366-1374. doi:10.1001/jama.2016.14617
26. Kirchin V, Page T, Keegan PE, et al. Urethral injection therapy for urinary incontinence in women. Cochrane Database Syst Rev. 2017;7:CD003881. doi:10.1002/14651858.CD003881.pub4
CASE Patient has urine leakage that worsens with exercise
At her annual preventative health visit, a 39-year-old woman reports that she has leakage of urine. She states that she drinks “a gallon of water daily” to help her lose the 20 lb she gained during the COVID-19 pandemic. She wants to resume Zumba fitness classes, but exercise makes her urine leakage worse. She started wearing protective pads because she finds herself often leaking urine on the way to the bathroom.
What nonsurgical treatment options are available for this patient?
Nearly half of all women experience urinary incontinence (UI), the involuntary loss of urine, and the condition increases with age.1 This common condition negatively impacts physical and psychological health and has been associated with social isolation, sexual dysfunction, and reduced independence.2,3 Symptoms of UI are underreported, and therefore universal screening is recommended for women of all ages.4 The diversity of available treatments (TABLE 1) provides patients and clinicians an opportunity to develop a plan that aligns with their symptom severity, goals, preferences, and resources.

Types of UI
The most common types of UI are stress urinary incontinence (SUI) and urgency urinary incontinence (UUI). Mixed urinary incontinence (MUI) occurs when symptoms of both SUI and UUI are present. Although the mechanisms that lead to urine leakage vary by the type of incontinence, many primary interventions improve both types of leakage, so a clinical diagnosis is sufficient to initiate treatment.
Stress urinary incontinence results from an impaired or weakened sphincter, which leads to involuntary, yet predictable, urine loss during increased abdominal pressure, such as coughing, laughing, sneezing, lifting, or physical activity.5 In UUI, involuntary loss of urine often accompanies the sudden urge to void. UUI is associated with overactive bladder (OAB), defined as urinary urgency, with or without urinary incontinence, usually accompanied by urinary frequency and/or nocturia (urination that interrupts sleep).6
In OAB, the detrusor muscle contracts randomly, leading to a sudden urge to void. When bladder pressure exceeds urethral sphincter closure pressure, urine leakage occurs. Women describe the urgency episodes as unpredictable, the urine leakage as prolonged with large volumes, and often occurring as they seek the toilet. Risk factors include age, obesity, parity, history of vaginal delivery, family history, ethnicity/race, medical comorbidities, menopausal status, and tobacco use.5
Making a diagnosis
A basic office evaluation is the most key step for diagnostic accuracy that leads to treatment success. This includes a detailed history, assessment of symptom severity, physical exam, pelvic exam, urinalysis, postvoid residual (to rule out urinary retention), and a cough stress test (to demonstrate SUI). The goal is to assess symptom severity, determine the type of UI, and identify contributing and potentially reversible factors, such as a urinary tract infection, medications, pelvic organ prolapse, incomplete bladder emptying, or impaired neurologic status. In the absence of the latter, advanced diagnostic tests, such as urodynamics, contribute little toward discerning the type of incontinence or changing first-line treatment plans.7
During the COVID-19 pandemic, abbreviated, virtual assessments for urinary symptoms were associated with high degrees of satisfaction (91% for fulfillment of personal needs, 94% overall satisfaction).8 This highlights the value of validated symptom questionnaires that help establish a working diagnosis and treatment plan in the absence of a physical exam. Questionnaire-based diagnoses have acceptable accuracy for classifying UUI and SUI among women with uncomplicated medical and surgical histories and for initiating low-risk therapies for defined intervals.
The 3 incontinence questions (3IQ) screen is an example of a useful, quick diagnostic tool designed for the primary care setting (FIGURE 1).9 It has been used in pharmaceutical treatment trials for UUI, with low frequency of misdiagnosis (1%–4%), resulting in no harm by the drug treatment prescribed or by the delay in appropriate care.10 Due to the limitations of an abbreviated remote evaluation, however, clinicians should assess patient response to primary interventions in a timely window. Patients who fail to experience satisfactory symptom reduction within 6 to 12 weeks should complete their evaluation in person or through a referral to a urogynecology program.

Continue to: Primary therapies for UI...
Primary therapies for UI
Primary therapies for UUI and SUI target strength training of the pelvic floor muscles, moderation of fluid intake, and adjustment in voiding behaviors and medications. Any functional barriers to continence also should be identified and addressed. Simple interventions, including a daily bowel regimen to address constipation, a bedside commode, and scheduled voiding, may reduce incontinence episodes without incurring significant cost or risk. For women suspected of having MUI, the treatment plan should prioritize their most bothersome symptoms.
Lifestyle and behavioral modifications
Everyday habits, medical comorbidities, and medications may exacerbate the severity of both SUI and UUI. Behavioral therapy alone or in combination with other interventions effectively reduces both SUI and UUI symptoms and has been shown to improve the efficacy of continence surgery.11 Information gained from a 3-day bladder diary (FIGURE 2)12 can guide clinicians on personalized patient recommendations, such as reducing excessive consumption of fluids and bladder irritants, limiting late evening drinking in the setting of bothersome nocturia, and scheduling voids (every 2–3 hours) to preempt incontinence episodes.

Weight loss
Obesity is a strong, independent, modifiable risk factor for both SUI and UUI. Each 5 kg/m2 increase in body mass index (BMI) has been associated with a 20% to 70% increased risk of UI, while weight loss of 5% or greater in overweight or obese women can lead to at least a 50% decrease in UI frequency.13
Reducing fluid intake and bladder irritants
Overactive bladder symptoms often respond to moderation of excessive fluid intake and reduction of bladder irritants (caffeine, carbonated beverages, diet beverages, and alcohol). While there is no established definition of excess caffeine intake, one study categorized high caffeine intake as greater than 400 mg/day (approximately four 8-oz cups of coffee).14
Information provided in a bladder diary can guide individualized recommendations for reducing fluid intake, particularly when 24-hour urine production exceeds the normative range (> 50–60 oz or 1.5-1.8 L/day).15 Hydration needs vary by activity, environment, and food; some general guidelines suggest 48 to 64 oz/day.5,16
Continue to: Pelvic floor muscle training...
Pelvic floor muscle training
An effective treatment for both UUI and SUI symptoms, pelvic floor muscle training (PFMT) leads to high degrees of patient satisfaction and improvement in quality of life.17 The presumed mechanisms of action of PFMT include improved urethral closure pressure and inhibition of detrusor muscle contractions.
Common exercise protocols recommend 3 sets of 10 contractions, held for 6 to 10 seconds per day, in varying positions of sitting, standing, and lying. While many women may be familiar with Kegel exercises, poor technique with straining and recruitment of gluteal and abdominal muscles can undermine the effect of PFMT. Clinicians can confirm successful pelvic muscle contractions by placing a finger in the vagina to appreciate contraction around and elevation of the finger toward the pubic symphysis in the absence of pushing.
Referral to supervised physical therapy and use of such teaching aid tools as booklets, mobile applications, and biofeedback can improve exercise adherence and outcomes.18,19 Systematic reviews report initial cure or improvement of incontinence symptoms as high as 74%, although little information is available about the long-term duration of effect.17
Vaginal pessaries
Vaginal continence support pessaries and devices work by stabilizing urethral mobility and compression of the bladder neck. Continence devices are particularly effective for situational SUI (such as during exercise).
The reusable medical grade silicone pessaries are available in numerous shapes and sizes and are fitted by a health care clinician (FIGURE 3). Uresta is a self-fitted intravaginal device that women can purchase online with a prescription. The Poise Impressa bladder support is a disposable intravaginal device marketed for incontinence and available over-the-counter, without a prescription (FIGURE 4). Anecdotally, many women find that menstrual tampons provide a similar effect, but outcome data are lacking.


In a comparative effectiveness trial of a continence pessary and behavior therapy, behavioral therapy was more likely to result in no bothersome incontinence symptoms (49% vs 33%, P = .006) and greater treatment satisfaction at 3 months.20 However, these short-term group differences did not persist at 12 months, presumably due to waning adherence.
UUI-specific nonsurgical treatments
Drug therapy
All medications approved by the US Food and Drug Administration (FDA) for UI are for the indications of OAB or UUI. These second-line treatments are most effective as adjuncts to behavioral modifications and PFMT.
A multicenter randomized trial that evaluated the efficacy of drug therapy alone compared with drug therapy in combination with behavioral modification, PFMT, urge suppression strategies, timed voiding, and fluid management for UUI found that combined therapy was more successful in achieving greater than 70% reduction in incontinence episodes (58% for drug therapy vs 69% for combined therapy).21
Of the 8 medications currently marketed in the United States for OAB or UUI, 6 are anticholinergic agents that block muscarinic receptors in the smooth muscle of the bladder, leading to inhibition of detrusor contractions, and 2 are β-adrenergic receptor agonists that promote bladder storage capacity by relaxing the detrusor muscle (TABLE 2). Similar efficacies lead most clinicians to initiate drug therapy based on formulary coverage and tolerance for adverse effects. Patients can expect a 53% to 80% reduction in UUI episodes and a 12% to 32% reduction in urinary frequency.22

Extended-release formulations are associated with reduced anticholinergic side effects (dry mouth, constipation, somnolence, dry eyes), leading to improved adherence. Notably, the anticholinergic medications are contraindicated in patients with untreated narrow-angle glaucoma, gastric retention, and supraventricular tachycardia. Mirabegron should be used with caution in patients with poorly controlled hypertension. 5 Due to concerns regarding the association between cumulative anticholinergic burden and the development of dementia, clinicians may consider avoiding the anticholinergic medications in older and at-risk patients.23
Continue to: UUI office-based procedure treatments...
UUI office-based procedure treatments
If behavioral therapies and medications are ineffective, contraindicated, or not the patient’s preference, additional FDA-approved therapies for UUI are available, typically through referral to a urogynecologist, urologist, or continence center.
Posterior tibial nerve stimulation (PTNS) is a nondrug treatment that delivers electrical stimulation using an acupuncture needle for 12 weekly 30-minute sessions followed by monthly maintenance for responders. The time commitment for this treatment plan can be a barrier for some patients. However, patients who adhere to the recommended protocol can expect a 60% improvement in symptoms, with minimal adverse events. Treatment efficacy is comparable to that of anticholinergic medication.24
OnabotulinumtoxinA injections into the bladder muscle are performed cystoscopically under local anesthetic. The toxin blocks the presynaptic release of acetylcholine at the neuromuscular junction, resulting in temporary muscle paralysis. This treatment is associated with high satisfaction. Efficacy varies by study population and outcome measure.
In one US comparative effectiveness trial, 67% of study participants with UUI symptoms refractory to oral medication reported a greater than 50% reduction in OAB symptoms at 6 months, 20% reported complete resolution of UUI, and 72% requested a second injection within 24 months.25 The interval between the first and second injection was nearly 1 year (350 days).Risks include urinary tract infection (12% within 1 month of the procedure and 35% through 6 months); urinary retention requiring catheterization has decreased to 6% with recognition that most moderate retention is tolerated by patients.
Some insurers limit onabotulinumtoxinA treatment coverage to patients who have failed to achieve symptom control with first- and second-line treatments.
SUI-specific nonsurgical treatments
Cystoscopic injection of urethral bulking agents into the urethral submucosa is designed to improve urethral coaptation. It is a minor procedure that can be performed in an ambulatory setting under local anesthetic with or without sedation.
Various bulking agents have been approved for use in the United States, some of which have been withdrawn due to complications of migration, erosion, and pseudoabscess formation. Cure or improvement after bulking agent injection was found to be superior to a home pelvic floor exercise program but inferior to a midurethral sling procedure for cure (9% vs 89%).26
The durability of currently available urethral bulking agents beyond 1 year is unknown. Complications are typically minor and transient and include pain at the injection site, urinary retention, de novo urgency, and implant leakage. The advantages include no postprocedure activity restrictions.
CASE Symptom presentation guides treatment plan
Our patient described symptoms of stress-predominant MUI. She was counseled to moderate her fluid intake to 2 L per day and to strategically time voids (before exercise, and at least every 4 hours). The patient was fitted with an incontinence pessary, and she elected to pursue a course of supervised physical therapy for pelvic floor muscle strengthening. Her follow-up visit is scheduled in 3 months to determine if other interventions are warranted. ●
CASE Patient has urine leakage that worsens with exercise
At her annual preventative health visit, a 39-year-old woman reports that she has leakage of urine. She states that she drinks “a gallon of water daily” to help her lose the 20 lb she gained during the COVID-19 pandemic. She wants to resume Zumba fitness classes, but exercise makes her urine leakage worse. She started wearing protective pads because she finds herself often leaking urine on the way to the bathroom.
What nonsurgical treatment options are available for this patient?
Nearly half of all women experience urinary incontinence (UI), the involuntary loss of urine, and the condition increases with age.1 This common condition negatively impacts physical and psychological health and has been associated with social isolation, sexual dysfunction, and reduced independence.2,3 Symptoms of UI are underreported, and therefore universal screening is recommended for women of all ages.4 The diversity of available treatments (TABLE 1) provides patients and clinicians an opportunity to develop a plan that aligns with their symptom severity, goals, preferences, and resources.

Types of UI
The most common types of UI are stress urinary incontinence (SUI) and urgency urinary incontinence (UUI). Mixed urinary incontinence (MUI) occurs when symptoms of both SUI and UUI are present. Although the mechanisms that lead to urine leakage vary by the type of incontinence, many primary interventions improve both types of leakage, so a clinical diagnosis is sufficient to initiate treatment.
Stress urinary incontinence results from an impaired or weakened sphincter, which leads to involuntary, yet predictable, urine loss during increased abdominal pressure, such as coughing, laughing, sneezing, lifting, or physical activity.5 In UUI, involuntary loss of urine often accompanies the sudden urge to void. UUI is associated with overactive bladder (OAB), defined as urinary urgency, with or without urinary incontinence, usually accompanied by urinary frequency and/or nocturia (urination that interrupts sleep).6
In OAB, the detrusor muscle contracts randomly, leading to a sudden urge to void. When bladder pressure exceeds urethral sphincter closure pressure, urine leakage occurs. Women describe the urgency episodes as unpredictable, the urine leakage as prolonged with large volumes, and often occurring as they seek the toilet. Risk factors include age, obesity, parity, history of vaginal delivery, family history, ethnicity/race, medical comorbidities, menopausal status, and tobacco use.5
Making a diagnosis
A basic office evaluation is the most key step for diagnostic accuracy that leads to treatment success. This includes a detailed history, assessment of symptom severity, physical exam, pelvic exam, urinalysis, postvoid residual (to rule out urinary retention), and a cough stress test (to demonstrate SUI). The goal is to assess symptom severity, determine the type of UI, and identify contributing and potentially reversible factors, such as a urinary tract infection, medications, pelvic organ prolapse, incomplete bladder emptying, or impaired neurologic status. In the absence of the latter, advanced diagnostic tests, such as urodynamics, contribute little toward discerning the type of incontinence or changing first-line treatment plans.7
During the COVID-19 pandemic, abbreviated, virtual assessments for urinary symptoms were associated with high degrees of satisfaction (91% for fulfillment of personal needs, 94% overall satisfaction).8 This highlights the value of validated symptom questionnaires that help establish a working diagnosis and treatment plan in the absence of a physical exam. Questionnaire-based diagnoses have acceptable accuracy for classifying UUI and SUI among women with uncomplicated medical and surgical histories and for initiating low-risk therapies for defined intervals.
The 3 incontinence questions (3IQ) screen is an example of a useful, quick diagnostic tool designed for the primary care setting (FIGURE 1).9 It has been used in pharmaceutical treatment trials for UUI, with low frequency of misdiagnosis (1%–4%), resulting in no harm by the drug treatment prescribed or by the delay in appropriate care.10 Due to the limitations of an abbreviated remote evaluation, however, clinicians should assess patient response to primary interventions in a timely window. Patients who fail to experience satisfactory symptom reduction within 6 to 12 weeks should complete their evaluation in person or through a referral to a urogynecology program.

Continue to: Primary therapies for UI...
Primary therapies for UI
Primary therapies for UUI and SUI target strength training of the pelvic floor muscles, moderation of fluid intake, and adjustment in voiding behaviors and medications. Any functional barriers to continence also should be identified and addressed. Simple interventions, including a daily bowel regimen to address constipation, a bedside commode, and scheduled voiding, may reduce incontinence episodes without incurring significant cost or risk. For women suspected of having MUI, the treatment plan should prioritize their most bothersome symptoms.
Lifestyle and behavioral modifications
Everyday habits, medical comorbidities, and medications may exacerbate the severity of both SUI and UUI. Behavioral therapy alone or in combination with other interventions effectively reduces both SUI and UUI symptoms and has been shown to improve the efficacy of continence surgery.11 Information gained from a 3-day bladder diary (FIGURE 2)12 can guide clinicians on personalized patient recommendations, such as reducing excessive consumption of fluids and bladder irritants, limiting late evening drinking in the setting of bothersome nocturia, and scheduling voids (every 2–3 hours) to preempt incontinence episodes.

Weight loss
Obesity is a strong, independent, modifiable risk factor for both SUI and UUI. Each 5 kg/m2 increase in body mass index (BMI) has been associated with a 20% to 70% increased risk of UI, while weight loss of 5% or greater in overweight or obese women can lead to at least a 50% decrease in UI frequency.13
Reducing fluid intake and bladder irritants
Overactive bladder symptoms often respond to moderation of excessive fluid intake and reduction of bladder irritants (caffeine, carbonated beverages, diet beverages, and alcohol). While there is no established definition of excess caffeine intake, one study categorized high caffeine intake as greater than 400 mg/day (approximately four 8-oz cups of coffee).14
Information provided in a bladder diary can guide individualized recommendations for reducing fluid intake, particularly when 24-hour urine production exceeds the normative range (> 50–60 oz or 1.5-1.8 L/day).15 Hydration needs vary by activity, environment, and food; some general guidelines suggest 48 to 64 oz/day.5,16
Continue to: Pelvic floor muscle training...
Pelvic floor muscle training
An effective treatment for both UUI and SUI symptoms, pelvic floor muscle training (PFMT) leads to high degrees of patient satisfaction and improvement in quality of life.17 The presumed mechanisms of action of PFMT include improved urethral closure pressure and inhibition of detrusor muscle contractions.
Common exercise protocols recommend 3 sets of 10 contractions, held for 6 to 10 seconds per day, in varying positions of sitting, standing, and lying. While many women may be familiar with Kegel exercises, poor technique with straining and recruitment of gluteal and abdominal muscles can undermine the effect of PFMT. Clinicians can confirm successful pelvic muscle contractions by placing a finger in the vagina to appreciate contraction around and elevation of the finger toward the pubic symphysis in the absence of pushing.
Referral to supervised physical therapy and use of such teaching aid tools as booklets, mobile applications, and biofeedback can improve exercise adherence and outcomes.18,19 Systematic reviews report initial cure or improvement of incontinence symptoms as high as 74%, although little information is available about the long-term duration of effect.17
Vaginal pessaries
Vaginal continence support pessaries and devices work by stabilizing urethral mobility and compression of the bladder neck. Continence devices are particularly effective for situational SUI (such as during exercise).
The reusable medical grade silicone pessaries are available in numerous shapes and sizes and are fitted by a health care clinician (FIGURE 3). Uresta is a self-fitted intravaginal device that women can purchase online with a prescription. The Poise Impressa bladder support is a disposable intravaginal device marketed for incontinence and available over-the-counter, without a prescription (FIGURE 4). Anecdotally, many women find that menstrual tampons provide a similar effect, but outcome data are lacking.


In a comparative effectiveness trial of a continence pessary and behavior therapy, behavioral therapy was more likely to result in no bothersome incontinence symptoms (49% vs 33%, P = .006) and greater treatment satisfaction at 3 months.20 However, these short-term group differences did not persist at 12 months, presumably due to waning adherence.
UUI-specific nonsurgical treatments
Drug therapy
All medications approved by the US Food and Drug Administration (FDA) for UI are for the indications of OAB or UUI. These second-line treatments are most effective as adjuncts to behavioral modifications and PFMT.
A multicenter randomized trial that evaluated the efficacy of drug therapy alone compared with drug therapy in combination with behavioral modification, PFMT, urge suppression strategies, timed voiding, and fluid management for UUI found that combined therapy was more successful in achieving greater than 70% reduction in incontinence episodes (58% for drug therapy vs 69% for combined therapy).21
Of the 8 medications currently marketed in the United States for OAB or UUI, 6 are anticholinergic agents that block muscarinic receptors in the smooth muscle of the bladder, leading to inhibition of detrusor contractions, and 2 are β-adrenergic receptor agonists that promote bladder storage capacity by relaxing the detrusor muscle (TABLE 2). Similar efficacies lead most clinicians to initiate drug therapy based on formulary coverage and tolerance for adverse effects. Patients can expect a 53% to 80% reduction in UUI episodes and a 12% to 32% reduction in urinary frequency.22

Extended-release formulations are associated with reduced anticholinergic side effects (dry mouth, constipation, somnolence, dry eyes), leading to improved adherence. Notably, the anticholinergic medications are contraindicated in patients with untreated narrow-angle glaucoma, gastric retention, and supraventricular tachycardia. Mirabegron should be used with caution in patients with poorly controlled hypertension. 5 Due to concerns regarding the association between cumulative anticholinergic burden and the development of dementia, clinicians may consider avoiding the anticholinergic medications in older and at-risk patients.23
Continue to: UUI office-based procedure treatments...
UUI office-based procedure treatments
If behavioral therapies and medications are ineffective, contraindicated, or not the patient’s preference, additional FDA-approved therapies for UUI are available, typically through referral to a urogynecologist, urologist, or continence center.
Posterior tibial nerve stimulation (PTNS) is a nondrug treatment that delivers electrical stimulation using an acupuncture needle for 12 weekly 30-minute sessions followed by monthly maintenance for responders. The time commitment for this treatment plan can be a barrier for some patients. However, patients who adhere to the recommended protocol can expect a 60% improvement in symptoms, with minimal adverse events. Treatment efficacy is comparable to that of anticholinergic medication.24
OnabotulinumtoxinA injections into the bladder muscle are performed cystoscopically under local anesthetic. The toxin blocks the presynaptic release of acetylcholine at the neuromuscular junction, resulting in temporary muscle paralysis. This treatment is associated with high satisfaction. Efficacy varies by study population and outcome measure.
In one US comparative effectiveness trial, 67% of study participants with UUI symptoms refractory to oral medication reported a greater than 50% reduction in OAB symptoms at 6 months, 20% reported complete resolution of UUI, and 72% requested a second injection within 24 months.25 The interval between the first and second injection was nearly 1 year (350 days).Risks include urinary tract infection (12% within 1 month of the procedure and 35% through 6 months); urinary retention requiring catheterization has decreased to 6% with recognition that most moderate retention is tolerated by patients.
Some insurers limit onabotulinumtoxinA treatment coverage to patients who have failed to achieve symptom control with first- and second-line treatments.
SUI-specific nonsurgical treatments
Cystoscopic injection of urethral bulking agents into the urethral submucosa is designed to improve urethral coaptation. It is a minor procedure that can be performed in an ambulatory setting under local anesthetic with or without sedation.
Various bulking agents have been approved for use in the United States, some of which have been withdrawn due to complications of migration, erosion, and pseudoabscess formation. Cure or improvement after bulking agent injection was found to be superior to a home pelvic floor exercise program but inferior to a midurethral sling procedure for cure (9% vs 89%).26
The durability of currently available urethral bulking agents beyond 1 year is unknown. Complications are typically minor and transient and include pain at the injection site, urinary retention, de novo urgency, and implant leakage. The advantages include no postprocedure activity restrictions.
CASE Symptom presentation guides treatment plan
Our patient described symptoms of stress-predominant MUI. She was counseled to moderate her fluid intake to 2 L per day and to strategically time voids (before exercise, and at least every 4 hours). The patient was fitted with an incontinence pessary, and she elected to pursue a course of supervised physical therapy for pelvic floor muscle strengthening. Her follow-up visit is scheduled in 3 months to determine if other interventions are warranted. ●
1. Lee UJ, Feinstein L, Ward JB, et al. Prevalence of urinary incontinence among a nationally representative sample of women, 2005–2016: findings from the Urologic Diseases in America Project. J Urol. 2021;205:1718-1724. doi:10.1097 /JU.0000000000001634
2. Sims J, Browning C, Lundgren-Lindquist B, et al. Urinary incontinence in a community sample of older adults: prevalence and impact on quality of life. Disabil Rehabil. 2011;33:1389-1398. doi:10.3109/09638288.2010.532284
3. Sarikaya S, Yildiz FG, Senocak C, et al. Urinary incontinence as a cause of depression and sexual dysfunction: questionnaire-based study. Rev Int Androl. 2020:18:50-54. doi:10.1016 /j.androl.2018.08.003
4. O’Reilly N, Nelson HD, Conry JM, et al; Women’s Preventive Services Initiative. Screening for urinary incontinence in women: a recommendation from the Women’s Preventive Services Initiative. Ann Intern Med. 2018;169(5):320-328. doi:10.7326/M18-0595
5. Barber MD, Walters MD, Karram MM, et al. Walters & Karram Urogynecology and Reconstructive Pelvic Surgery. 5th ed. Elsevier Saunders; 2021.
6. Haylen BT, de Ridder D, Freeman RM, et al. An International Urogynecological Association (IUGA)/International Continence Society (ICS) joint report on the terminology for female pelvic floor dysfunction. Int Urogynecol J. 2010;21: 5-26. doi:10.1007/s00192-009-0976-9
7. ACOG practice bulletin no. 155. Urinary incontinence in women. Obstet Gynecol. 2015;126:e66-e81. doi:10.1097 /AOG.0000000000001148
8. Sansone S, Lu J, Drangsholt S, et al. No pelvic exam, no problem: patient satisfaction following the integration of comprehensive urogynecology telemedicine. Int Urogynecol J. 2022;1:3. doi:10.1007/s00192-022-05104-w
9. Brown JS, Bradley CS, Subak LL, et al; Diagnostic Aspects of Incontinence Study (DAISy) Research Group. The sensitivity and specificity of a simple test to distinguish between urge and stress urinary incontinence. Ann Intern Med. 2006;144:715723. doi:10.7326/0003-4819-144-10-200605160-00005
10. Hess R, Huang AJ, Richter HE, et al. Long-term efficacy and safety of questionnaire-based initiation of urgency urinary incontinence treatment. Am J Obstet Gynecol. 2013;209:244. e1-9. doi:10.1016/j.ajog.2013.05.008
11. Sung VW, Borello-France D, Newman DK, et al; NICHD Pelvic Floor Disorders Network. Effect of behavioral and pelvic floor muscle therapy combined with surgery vs surgery alone on incontinence symptoms among women with mixed urinary incontinence. JAMA. 2019;322:1066-1076. doi:10.1001 /jama.2019.12467
12. American Urogynecologic Society. Voices for PFD: intake and voiding diary. Accessed August 11, 2022. https://www .voicesforpfd.org/assets/2/6/Voiding_Diary.pdf
13. Subak LL, Richter HE, Hunskaar S. Obesity and urinary incontinence: epidemiology and clinical research update. J Urol. 2009;182(6 suppl):S2-7. doi:10.1016/j.juro.2009.08.071
14. Arya LA, Myers DL, Jackson ND. Dietary caffeine intake and the risk for detrusor instability: a case-control study. Obstet Gynecol. 2000;96:85-89. doi:10.1016/s0029-7844(00)00808-5
15. Wyman JF, Zhou J, LaCoursiere DY, et al. Normative noninvasive bladder function measurements in healthy women: a systematic review and meta-analysis. Neurourol Urodyn. 2020;39:507-522. doi:10.1002/nau.24265
16. Hashim H, Al Mousa R. Management of fluid intake in patients with overactive bladder. Curr Urol Rep. 2009;10: 428-433. doi:10.1007/s11934-009-0068-x
17. Dumoulin C, Cacciari LP, Hay-Smith EJC. Pelvic floor muscle training versus no treatment, or inactive control treatments, for urinary incontinence in women. Cochrane Database Syst Rev. 2018;10:CD005654. doi:10.1002/14651858.CD005654.pub4
18. Araujo CC, de A Marques A, Juliato CRT. The adherence of home pelvic floor muscles training using a mobile device application for women with urinary incontinence: a randomized controlled trial. Female Pelvic Med Reconstr Surg. 2020;26:697-703. doi:10.1097/SPV.0000000000000670
19. Sjöström M, Umefjord G, Stenlund H, et al. Internet-based treatment of stress urinary incontinence: a randomized controlled study with focus on pelvic floor muscle training. BJU Int. 2013;112:362-372. doi:10.1111/j.1464 -410X.2012.11713.x
20. Richter HE, Burgio KL, Brubaker L, et al; Pelvic Floor Disorders Network. Continence pessary compared with behavioral therapy or combined therapy for stress incontinence: a randomized controlled trial. Obstet Gynecol. 2010;115:609617. doi:10.1097/AOG.0b013e3181d055d4
21. Burgio KL, Kraus SR, Menefee S, et al. Behavioral therapy to enable women with urge incontinence to discontinue drug treatment: a randomized trial. Ann Intern Med. 2008;149(3): 161-169. doi:10.7326/0003-4819-149-3-200808050 -00005
22. Lukacz ES, Santiago-Lastra Y, Albo ME, et al. Urinary incontinence in women: a review. JAMA. 2017;318:1592-1604. doi:10.1001/jama.2017.12137
23. Welk B, Richardson K, Panicker JN. The cognitive effect of anticholinergics for patients with overactive bladder. Nat Rev Urol. 2021;18:686-700. doi:10.1038/s41585-021-00504-x
24. Burton C, Sajja A, Latthe PM. Effectiveness of percutaneous posterior tibial nerve stimulation for overactive bladder: a systematic review and meta-analysis. Neurourol Urodyn. 2012;31:1206-1216. doi:10.1002/nau.22251
25. Amundsen CL, Richter HE, Menefee SA, et al. OnabotulinumtoxinA vs sacral neuromodulation on refractory urgency urinary incontinence in women: A randomized clinical trial. JAMA. 2016;316:1366-1374. doi:10.1001/jama.2016.14617
26. Kirchin V, Page T, Keegan PE, et al. Urethral injection therapy for urinary incontinence in women. Cochrane Database Syst Rev. 2017;7:CD003881. doi:10.1002/14651858.CD003881.pub4
1. Lee UJ, Feinstein L, Ward JB, et al. Prevalence of urinary incontinence among a nationally representative sample of women, 2005–2016: findings from the Urologic Diseases in America Project. J Urol. 2021;205:1718-1724. doi:10.1097 /JU.0000000000001634
2. Sims J, Browning C, Lundgren-Lindquist B, et al. Urinary incontinence in a community sample of older adults: prevalence and impact on quality of life. Disabil Rehabil. 2011;33:1389-1398. doi:10.3109/09638288.2010.532284
3. Sarikaya S, Yildiz FG, Senocak C, et al. Urinary incontinence as a cause of depression and sexual dysfunction: questionnaire-based study. Rev Int Androl. 2020:18:50-54. doi:10.1016 /j.androl.2018.08.003
4. O’Reilly N, Nelson HD, Conry JM, et al; Women’s Preventive Services Initiative. Screening for urinary incontinence in women: a recommendation from the Women’s Preventive Services Initiative. Ann Intern Med. 2018;169(5):320-328. doi:10.7326/M18-0595
5. Barber MD, Walters MD, Karram MM, et al. Walters & Karram Urogynecology and Reconstructive Pelvic Surgery. 5th ed. Elsevier Saunders; 2021.
6. Haylen BT, de Ridder D, Freeman RM, et al. An International Urogynecological Association (IUGA)/International Continence Society (ICS) joint report on the terminology for female pelvic floor dysfunction. Int Urogynecol J. 2010;21: 5-26. doi:10.1007/s00192-009-0976-9
7. ACOG practice bulletin no. 155. Urinary incontinence in women. Obstet Gynecol. 2015;126:e66-e81. doi:10.1097 /AOG.0000000000001148
8. Sansone S, Lu J, Drangsholt S, et al. No pelvic exam, no problem: patient satisfaction following the integration of comprehensive urogynecology telemedicine. Int Urogynecol J. 2022;1:3. doi:10.1007/s00192-022-05104-w
9. Brown JS, Bradley CS, Subak LL, et al; Diagnostic Aspects of Incontinence Study (DAISy) Research Group. The sensitivity and specificity of a simple test to distinguish between urge and stress urinary incontinence. Ann Intern Med. 2006;144:715723. doi:10.7326/0003-4819-144-10-200605160-00005
10. Hess R, Huang AJ, Richter HE, et al. Long-term efficacy and safety of questionnaire-based initiation of urgency urinary incontinence treatment. Am J Obstet Gynecol. 2013;209:244. e1-9. doi:10.1016/j.ajog.2013.05.008
11. Sung VW, Borello-France D, Newman DK, et al; NICHD Pelvic Floor Disorders Network. Effect of behavioral and pelvic floor muscle therapy combined with surgery vs surgery alone on incontinence symptoms among women with mixed urinary incontinence. JAMA. 2019;322:1066-1076. doi:10.1001 /jama.2019.12467
12. American Urogynecologic Society. Voices for PFD: intake and voiding diary. Accessed August 11, 2022. https://www .voicesforpfd.org/assets/2/6/Voiding_Diary.pdf
13. Subak LL, Richter HE, Hunskaar S. Obesity and urinary incontinence: epidemiology and clinical research update. J Urol. 2009;182(6 suppl):S2-7. doi:10.1016/j.juro.2009.08.071
14. Arya LA, Myers DL, Jackson ND. Dietary caffeine intake and the risk for detrusor instability: a case-control study. Obstet Gynecol. 2000;96:85-89. doi:10.1016/s0029-7844(00)00808-5
15. Wyman JF, Zhou J, LaCoursiere DY, et al. Normative noninvasive bladder function measurements in healthy women: a systematic review and meta-analysis. Neurourol Urodyn. 2020;39:507-522. doi:10.1002/nau.24265
16. Hashim H, Al Mousa R. Management of fluid intake in patients with overactive bladder. Curr Urol Rep. 2009;10: 428-433. doi:10.1007/s11934-009-0068-x
17. Dumoulin C, Cacciari LP, Hay-Smith EJC. Pelvic floor muscle training versus no treatment, or inactive control treatments, for urinary incontinence in women. Cochrane Database Syst Rev. 2018;10:CD005654. doi:10.1002/14651858.CD005654.pub4
18. Araujo CC, de A Marques A, Juliato CRT. The adherence of home pelvic floor muscles training using a mobile device application for women with urinary incontinence: a randomized controlled trial. Female Pelvic Med Reconstr Surg. 2020;26:697-703. doi:10.1097/SPV.0000000000000670
19. Sjöström M, Umefjord G, Stenlund H, et al. Internet-based treatment of stress urinary incontinence: a randomized controlled study with focus on pelvic floor muscle training. BJU Int. 2013;112:362-372. doi:10.1111/j.1464 -410X.2012.11713.x
20. Richter HE, Burgio KL, Brubaker L, et al; Pelvic Floor Disorders Network. Continence pessary compared with behavioral therapy or combined therapy for stress incontinence: a randomized controlled trial. Obstet Gynecol. 2010;115:609617. doi:10.1097/AOG.0b013e3181d055d4
21. Burgio KL, Kraus SR, Menefee S, et al. Behavioral therapy to enable women with urge incontinence to discontinue drug treatment: a randomized trial. Ann Intern Med. 2008;149(3): 161-169. doi:10.7326/0003-4819-149-3-200808050 -00005
22. Lukacz ES, Santiago-Lastra Y, Albo ME, et al. Urinary incontinence in women: a review. JAMA. 2017;318:1592-1604. doi:10.1001/jama.2017.12137
23. Welk B, Richardson K, Panicker JN. The cognitive effect of anticholinergics for patients with overactive bladder. Nat Rev Urol. 2021;18:686-700. doi:10.1038/s41585-021-00504-x
24. Burton C, Sajja A, Latthe PM. Effectiveness of percutaneous posterior tibial nerve stimulation for overactive bladder: a systematic review and meta-analysis. Neurourol Urodyn. 2012;31:1206-1216. doi:10.1002/nau.22251
25. Amundsen CL, Richter HE, Menefee SA, et al. OnabotulinumtoxinA vs sacral neuromodulation on refractory urgency urinary incontinence in women: A randomized clinical trial. JAMA. 2016;316:1366-1374. doi:10.1001/jama.2016.14617
26. Kirchin V, Page T, Keegan PE, et al. Urethral injection therapy for urinary incontinence in women. Cochrane Database Syst Rev. 2017;7:CD003881. doi:10.1002/14651858.CD003881.pub4
2022 Update on abnormal uterine bleeding
In this Update, we focus on therapies for abnormal uterine bleeding (AUB) that include a new formulation of a progesterone-only pill (POP), drospirenone 4 mg in a 24/4 regimen (24 days of drospirenone/4 days of inert tablets), which recently showed benefit over the use of desogestrel in a European randomized clinical trial (RCT). Two other commonly used treatments for AUB— the levonorgestrel-releasing intrauterine system (LNG IUS) and endometrial ablation—were studied in terms of cost-effectiveness as well as whether they should be used in combination for added efficacy. In addition, although at times either COVID-19 disease or the COVID-19 vaccine has been blamed for societal and medical problems, one study showed that it is unlikely that significant changes in the menstrual cycle are a result of the COVID-19 vaccine.
COVID-19 vaccination had minimal effects on menstrual cycle length
Edelman A, Boniface ER, Benhar W, et al. Association between menstrual cycle length and coronavirus disease 2019 (COVID-19) vaccination: a US cohort. Obstet Gynecol. 2022;139:481-489.
Does receiving the COVID-19 vaccination result in abnormal menstrual cycles? Patients often ask this question, and it has been a topic of social media discussion (including NPR) and concerns about the possibility of vaccine hesitancy,1,2 as the menstrual cycle is often considered a sign of health and fertility.
To better understand this possible association, Edelman and colleagues conducted a study that prospectively tracked menstrual cycle data using the digital app Natural Cycles in US residents aged 18 to 45 years for 3 consecutive cycles in both a vaccinated and an unvaccinated cohort.3 Almost 4,000 individuals were studied; 2,403 were vaccinated and 1,556 were unvaccinated. The study vaccine types included the BioNTech (Pfizer), Moderna, Johnson & Johnson/Janssen, and unspecified vaccines.
The primary outcome was the within-individual change in cycle length in days, comparing a 3-cycle postvaccine average to a 3-cycle prevaccination average in the 2 groups. (For the unvaccinated group, cycles 1, 2, and 3 were considered the equivalent of prevaccination cycles; cycle 4 was designated as the artificial first vaccine dose-cycle and cycle 5 as the artificial second-dose cycle.)
Increase in cycle length clinically negligible
The investigators found that the vaccinated cohort had less than a 1-day unadjusted increase in the length of their menstrual cycle, which was essentially a 0.71-day increase (98.75% confidence interval [CI], 0.47–0.94). Although this is considered statistically significant, it is likely clinically insignificant in that the overlaid histograms comparing the distribution of change showed a cycle length distribution in vaccinated individuals that is essentially equivalent to that in unvaccinated individuals. After adjusting for confounders, the difference in cycle length was reduced to a 0.64 day (98.75% CI, 0.27–1.01).
An interesting finding was that a subset of individuals who received both vaccine doses in a single cycle had, on average, an adjusted 2-day increase in their menstrual cycle compared with unvaccinated individuals. To explain this slightly longer cycle length, the authors postulated that mRNA vaccines create an immune response, or stressor, which could temporarily affect the hypothalamic-pituitary-ovarian axis if timed correctly. It is certainly possible for an individual to receive 2 doses in a single cycle, which could have both been administered in the early follicular phase. Such cycle length variability can be caused by events, including stressors, that affect the recruitment and maturation of the dominant follicle.
Counseling takeaway
This study provides reassurance to most individuals who receive a COVID-19 vaccine that it likely will not affect their menstrual cycle in a clinically significant manner.
This robust study by Edelman and colleagues on COVID-19 vaccination effects on menstrual cycle length had more than 99% power to detect an unadjusted 1-day difference in cycle length. However, given that most of the study participants were White and had access to the Natural Cycles app, the results may not be generalizable to all individuals who receive the vaccine.
Continue to: Drospirenone improved bleeding profiles, lowered discontinuation rates compared with desogestrel...
Drospirenone improved bleeding profiles, lowered discontinuation rates compared with desogestrel
Regidor PA, Colli E, Palacios S. Overall and bleeding-related discontinuation rates of a new oral contraceptive containing 4 mg drospirenone only in a 24/4 regimen and comparison to 0.075 mg desogestrel. Gynecol Endocrinol. 2021;37:1121-1127.
A new POP, marketed under the name Slynd, recently came to market. It contains the progestin drospirenone (DRSP) 4 mg in a 24/4 regimen. This formulation has the advantage of being an antiandrogenic progestin, with a long enough half-life to allow for managing a missed pill in the same fashion as combined oral contraceptives (COCs).
Investigators in Europe conducted a double-blind, randomized trial to assess discontinuation rates due to adverse events (mainly bleeding disorders) in participants taking DRSP 4 mg in a 24/4 regimen compared with those taking the POP desogestrel (DSG) 0.075 mg, which is commonly used in Europe.4 Regidor and colleagues compared 858 women with 6,691 DRSP treatment cycles with 332 women with 2,487 DSG treatment cycles.
Top reasons for stopping a POP
The discontinuation rate for abnormal bleeding was 3.7% in the DRSP group versus 7.3% in the DSG group (55.7% lower). The most common reasons for stopping either POP formulation were vaginal bleeding and acne. Both of these adverse events were less common in the DRSP group. Pill discontinuation due to vaginal bleeding was 2.6% in the DRSP group versus 5.4% in the DSG group, while discontinuation due to acne occurred in 1% in the DRSP group versus 2.7% in the DSG group.
New oral contraception option
This study shows improved acceptability and bleeding profiles in women using this new DRSP contraception pill regimen.
Adherence to a contraceptive method is influenced by patient satisfaction, and this is particularly important in patients who cannot take COCs. It also should be noted that the discontinuation rate for DRSP as a POP used in this 24/4 regimen was similar to discontinuation rates for COCs containing 20 µg and 30 µg of ethinyl estradiol. Cost, however, may be an issue with DRSP, depending on a patient’s insurance coverage.
Continue to: Placing an LNG IUS after endometrial ablation for heavy menstrual bleeding reduced risk of hysterectomy...
Placing an LNG IUS after endometrial ablation for heavy menstrual bleeding reduced risk of hysterectomy
Oderkerk TJ, van de Kar MMA, van der Zanden CHM, et al. The combined use of endometrial ablation or resection and levonorgestrel-releasing intrauterine system in women with heavy menstrual bleeding: a systematic review. Acta Obstet Gynecol Scand. 2021;100:1779-1787.
Over the years, a smattering of articles have suggested that a reduction in uterine bleeding was associated with placement of an LNG IUS at the conclusion of endometrial ablation. We now have a systematic review of this surgical modification.
Oderkerk and colleagues sifted through 747 articles to find 7 publications that could provide meaningful data on the impact of combined use of endometrial ablation and LNG IUS insertion for women with heavy menstrual bleeding.5 These included 4 retrospective cohort studies with control groups, 2 retrospective studies without control groups, and 1 case series. The primary outcome was the hysterectomy rate after therapy.
Promising results for combined therapy
Although no statistically significant intergroup differences were seen in the combined treatment group versus the endometrial ablation alone group for the first 6 months of treatment, significant differences existed at the 12- and 24-month mark. Hysterectomy rates after combined treatment varied from 0% to 11% versus 9.4% to 24% after endometrial ablation alone. Complication rates for combined treatment did not appear higher than those for endometrial ablation alone.
The authors postulated that the failure of endometrial ablation is generally caused by either remaining or regenerating endometrial tissue and that the addition of an LNG IUS allows for suppression of endometrial tissue. Also encouraging was that, in general, the removal of the LNG IUS was relatively simple. A single difficult removal was described due to uterine synechiae, but hysteroscopic resection was not necessary. The authors acknowledged that the data from these 7 retrospective studies are limited and that high-quality research from prospective studies is needed.
Bottom line
The data available from this systematic review suggest that placement of an LNG IUS at the completion of an endometrial ablation may result in lower hysterectomy rates, without apparent risk, and without significantly difficult LNG IUS removal when needed.
The data provided by Oderkerk and colleagues’ systematic review are promising and, although not studied in the reviewed publications, the potential may exist to reduce the risk of endometrial hyperplasia and endometrial cancer by adding an LNG IUS.
Continue to: LNG IUS is less expensive, and less effective, than endometrial ablation for heavy menstrual bleeding, cost analysis shows...
LNG IUS is less expensive, and less effective, than endometrial ablation for heavy menstrual bleeding, cost analysis shows
van den Brink MJ, Beelen P, Herman MC, et al. The levonorgestrel intrauterine system versus endometrial ablation for heavy menstrual bleeding: a cost-effectiveness analysis. BJOG. 2021;128:2003-2011.
To assess the cost-effectiveness of the LNG IUS versus endometrial ablation in the treatment of heavy menstrual bleeding, van den Brink and colleagues conducted a randomized, noninferiority trial.6
Part of the rationale for this study was to better understand the cost differences between the LNG IUS and second-generation endometrial ablation. Some data have suggested that the LNG IUS is cost-effective when compared with first-generation endometrial ablation; however, definitive evidence about its cost compared with second-generation endometrial ablation is lacking, as these procedures should be less expensive than first-generation endometrial ablation since they frequently are performed in the office rather than in an operating room.
Cost-effectiveness and noninferiority assessed
A total of 270 women were randomly assigned to 1 of 2 treatment strategies. Eventually, 132 women were treated first with the 52-mg LNG IUS, and 138 were treated first with endometrial ablation by radiofrequency ablation. Menstrual blood loss after 24 months was the primary outcome.
At 24 months, the mean pictorial blood loss assessment chart (PBAC) scores were 64.8 in the LNG IUS group compared with 14.2 in the endometrial ablation group. Given that the noninferiority margin was defined as 25 points, noninferiority could not be demonstrated. However, when looking at PBAC scores less than 75 points, the LNG IUS group met this secondary end point in 87% of women versus 94% in the endometrial ablation group. When satisfaction was assessed, 74% of women in the LNG IUS group were satisfied compared with 84% in the endometrial ablation group.
Overall, the total costs per patient were €2,285 in the LNG IUS strategy and €3,465 in the endometrial ablation strategy (costs convert to $2,285 and $3,465 as of this writing).
Key takeaway
Treatment of heavy menstrual bleeding starting with the LNG IUS is cheaper, but it is slightly less effective than endometrial ablation. ●
It is interesting that there are minimal differences between satisfaction rates and PBAC scores less than 75, yet the mean PBAC scores were significantly more favorable for endometrial ablation. This study’s results support the use of a sequential therapy of a less invasive therapy, such as the LNG IUS, prior to performing endometrial ablation.
- Blumfiel G. Why reports of menstrual changes after COVID vaccine are tough to study. NPR. August 9, 2021. Accessed August 30, 2022. https://www.npr.org/sections/health-shots/2021/08/09/1024190379/covid-vaccine-period-menstrual-cycle-research
- Lee KMN, Junkins EJ, Fatima UA, et al. Characterizing menstrual bleeding changes occurring after SARSCoV-2 vaccinations. MedRxiv. February 11, 2022. doi:10.1101/2021.10.11.21264863
- Edelman A, Boniface ER, Benhar W, et al. Association between menstrual cycle length and coronavirus disease 2019 (COVID-19) vaccination: a US cohort. Obstet Gynecol. 2022;139:481-489.
- Regidor PA, Colli E, Palacios S. Overall and bleeding-related discontinuation rates of a new oral contraceptive containing 4 mg drospirenone only in a 24/4 regimen and comparison to 0.075 mg desogestrel. Gynecol Endocrinol. 2021;37:1121-1127.
- Oderkerk TJ, van de Kar MMA, van der Zanden CHM, et al. T he combined use of endometrial ablation or resection and levonorgestrel-releasing intrauterine system in women with heavy menstrual bleeding: a systematic review. Acta Obstet Gynecol Scand. 2021;100:1779-1787.
- van den Brink MJ, Beelen P, Herman MC, et al. The levonorgestrel intrauterine system versus endometrial ablation for heavy menstrual bleeding: a cost-effectiveness analysis. BJOG. 2021;128:2003-2011.
In this Update, we focus on therapies for abnormal uterine bleeding (AUB) that include a new formulation of a progesterone-only pill (POP), drospirenone 4 mg in a 24/4 regimen (24 days of drospirenone/4 days of inert tablets), which recently showed benefit over the use of desogestrel in a European randomized clinical trial (RCT). Two other commonly used treatments for AUB— the levonorgestrel-releasing intrauterine system (LNG IUS) and endometrial ablation—were studied in terms of cost-effectiveness as well as whether they should be used in combination for added efficacy. In addition, although at times either COVID-19 disease or the COVID-19 vaccine has been blamed for societal and medical problems, one study showed that it is unlikely that significant changes in the menstrual cycle are a result of the COVID-19 vaccine.
COVID-19 vaccination had minimal effects on menstrual cycle length
Edelman A, Boniface ER, Benhar W, et al. Association between menstrual cycle length and coronavirus disease 2019 (COVID-19) vaccination: a US cohort. Obstet Gynecol. 2022;139:481-489.
Does receiving the COVID-19 vaccination result in abnormal menstrual cycles? Patients often ask this question, and it has been a topic of social media discussion (including NPR) and concerns about the possibility of vaccine hesitancy,1,2 as the menstrual cycle is often considered a sign of health and fertility.
To better understand this possible association, Edelman and colleagues conducted a study that prospectively tracked menstrual cycle data using the digital app Natural Cycles in US residents aged 18 to 45 years for 3 consecutive cycles in both a vaccinated and an unvaccinated cohort.3 Almost 4,000 individuals were studied; 2,403 were vaccinated and 1,556 were unvaccinated. The study vaccine types included the BioNTech (Pfizer), Moderna, Johnson & Johnson/Janssen, and unspecified vaccines.
The primary outcome was the within-individual change in cycle length in days, comparing a 3-cycle postvaccine average to a 3-cycle prevaccination average in the 2 groups. (For the unvaccinated group, cycles 1, 2, and 3 were considered the equivalent of prevaccination cycles; cycle 4 was designated as the artificial first vaccine dose-cycle and cycle 5 as the artificial second-dose cycle.)
Increase in cycle length clinically negligible
The investigators found that the vaccinated cohort had less than a 1-day unadjusted increase in the length of their menstrual cycle, which was essentially a 0.71-day increase (98.75% confidence interval [CI], 0.47–0.94). Although this is considered statistically significant, it is likely clinically insignificant in that the overlaid histograms comparing the distribution of change showed a cycle length distribution in vaccinated individuals that is essentially equivalent to that in unvaccinated individuals. After adjusting for confounders, the difference in cycle length was reduced to a 0.64 day (98.75% CI, 0.27–1.01).
An interesting finding was that a subset of individuals who received both vaccine doses in a single cycle had, on average, an adjusted 2-day increase in their menstrual cycle compared with unvaccinated individuals. To explain this slightly longer cycle length, the authors postulated that mRNA vaccines create an immune response, or stressor, which could temporarily affect the hypothalamic-pituitary-ovarian axis if timed correctly. It is certainly possible for an individual to receive 2 doses in a single cycle, which could have both been administered in the early follicular phase. Such cycle length variability can be caused by events, including stressors, that affect the recruitment and maturation of the dominant follicle.
Counseling takeaway
This study provides reassurance to most individuals who receive a COVID-19 vaccine that it likely will not affect their menstrual cycle in a clinically significant manner.
This robust study by Edelman and colleagues on COVID-19 vaccination effects on menstrual cycle length had more than 99% power to detect an unadjusted 1-day difference in cycle length. However, given that most of the study participants were White and had access to the Natural Cycles app, the results may not be generalizable to all individuals who receive the vaccine.
Continue to: Drospirenone improved bleeding profiles, lowered discontinuation rates compared with desogestrel...
Drospirenone improved bleeding profiles, lowered discontinuation rates compared with desogestrel
Regidor PA, Colli E, Palacios S. Overall and bleeding-related discontinuation rates of a new oral contraceptive containing 4 mg drospirenone only in a 24/4 regimen and comparison to 0.075 mg desogestrel. Gynecol Endocrinol. 2021;37:1121-1127.
A new POP, marketed under the name Slynd, recently came to market. It contains the progestin drospirenone (DRSP) 4 mg in a 24/4 regimen. This formulation has the advantage of being an antiandrogenic progestin, with a long enough half-life to allow for managing a missed pill in the same fashion as combined oral contraceptives (COCs).
Investigators in Europe conducted a double-blind, randomized trial to assess discontinuation rates due to adverse events (mainly bleeding disorders) in participants taking DRSP 4 mg in a 24/4 regimen compared with those taking the POP desogestrel (DSG) 0.075 mg, which is commonly used in Europe.4 Regidor and colleagues compared 858 women with 6,691 DRSP treatment cycles with 332 women with 2,487 DSG treatment cycles.
Top reasons for stopping a POP
The discontinuation rate for abnormal bleeding was 3.7% in the DRSP group versus 7.3% in the DSG group (55.7% lower). The most common reasons for stopping either POP formulation were vaginal bleeding and acne. Both of these adverse events were less common in the DRSP group. Pill discontinuation due to vaginal bleeding was 2.6% in the DRSP group versus 5.4% in the DSG group, while discontinuation due to acne occurred in 1% in the DRSP group versus 2.7% in the DSG group.
New oral contraception option
This study shows improved acceptability and bleeding profiles in women using this new DRSP contraception pill regimen.
Adherence to a contraceptive method is influenced by patient satisfaction, and this is particularly important in patients who cannot take COCs. It also should be noted that the discontinuation rate for DRSP as a POP used in this 24/4 regimen was similar to discontinuation rates for COCs containing 20 µg and 30 µg of ethinyl estradiol. Cost, however, may be an issue with DRSP, depending on a patient’s insurance coverage.
Continue to: Placing an LNG IUS after endometrial ablation for heavy menstrual bleeding reduced risk of hysterectomy...
Placing an LNG IUS after endometrial ablation for heavy menstrual bleeding reduced risk of hysterectomy
Oderkerk TJ, van de Kar MMA, van der Zanden CHM, et al. The combined use of endometrial ablation or resection and levonorgestrel-releasing intrauterine system in women with heavy menstrual bleeding: a systematic review. Acta Obstet Gynecol Scand. 2021;100:1779-1787.
Over the years, a smattering of articles have suggested that a reduction in uterine bleeding was associated with placement of an LNG IUS at the conclusion of endometrial ablation. We now have a systematic review of this surgical modification.
Oderkerk and colleagues sifted through 747 articles to find 7 publications that could provide meaningful data on the impact of combined use of endometrial ablation and LNG IUS insertion for women with heavy menstrual bleeding.5 These included 4 retrospective cohort studies with control groups, 2 retrospective studies without control groups, and 1 case series. The primary outcome was the hysterectomy rate after therapy.
Promising results for combined therapy
Although no statistically significant intergroup differences were seen in the combined treatment group versus the endometrial ablation alone group for the first 6 months of treatment, significant differences existed at the 12- and 24-month mark. Hysterectomy rates after combined treatment varied from 0% to 11% versus 9.4% to 24% after endometrial ablation alone. Complication rates for combined treatment did not appear higher than those for endometrial ablation alone.
The authors postulated that the failure of endometrial ablation is generally caused by either remaining or regenerating endometrial tissue and that the addition of an LNG IUS allows for suppression of endometrial tissue. Also encouraging was that, in general, the removal of the LNG IUS was relatively simple. A single difficult removal was described due to uterine synechiae, but hysteroscopic resection was not necessary. The authors acknowledged that the data from these 7 retrospective studies are limited and that high-quality research from prospective studies is needed.
Bottom line
The data available from this systematic review suggest that placement of an LNG IUS at the completion of an endometrial ablation may result in lower hysterectomy rates, without apparent risk, and without significantly difficult LNG IUS removal when needed.
The data provided by Oderkerk and colleagues’ systematic review are promising and, although not studied in the reviewed publications, the potential may exist to reduce the risk of endometrial hyperplasia and endometrial cancer by adding an LNG IUS.
Continue to: LNG IUS is less expensive, and less effective, than endometrial ablation for heavy menstrual bleeding, cost analysis shows...
LNG IUS is less expensive, and less effective, than endometrial ablation for heavy menstrual bleeding, cost analysis shows
van den Brink MJ, Beelen P, Herman MC, et al. The levonorgestrel intrauterine system versus endometrial ablation for heavy menstrual bleeding: a cost-effectiveness analysis. BJOG. 2021;128:2003-2011.
To assess the cost-effectiveness of the LNG IUS versus endometrial ablation in the treatment of heavy menstrual bleeding, van den Brink and colleagues conducted a randomized, noninferiority trial.6
Part of the rationale for this study was to better understand the cost differences between the LNG IUS and second-generation endometrial ablation. Some data have suggested that the LNG IUS is cost-effective when compared with first-generation endometrial ablation; however, definitive evidence about its cost compared with second-generation endometrial ablation is lacking, as these procedures should be less expensive than first-generation endometrial ablation since they frequently are performed in the office rather than in an operating room.
Cost-effectiveness and noninferiority assessed
A total of 270 women were randomly assigned to 1 of 2 treatment strategies. Eventually, 132 women were treated first with the 52-mg LNG IUS, and 138 were treated first with endometrial ablation by radiofrequency ablation. Menstrual blood loss after 24 months was the primary outcome.
At 24 months, the mean pictorial blood loss assessment chart (PBAC) scores were 64.8 in the LNG IUS group compared with 14.2 in the endometrial ablation group. Given that the noninferiority margin was defined as 25 points, noninferiority could not be demonstrated. However, when looking at PBAC scores less than 75 points, the LNG IUS group met this secondary end point in 87% of women versus 94% in the endometrial ablation group. When satisfaction was assessed, 74% of women in the LNG IUS group were satisfied compared with 84% in the endometrial ablation group.
Overall, the total costs per patient were €2,285 in the LNG IUS strategy and €3,465 in the endometrial ablation strategy (costs convert to $2,285 and $3,465 as of this writing).
Key takeaway
Treatment of heavy menstrual bleeding starting with the LNG IUS is cheaper, but it is slightly less effective than endometrial ablation. ●
It is interesting that there are minimal differences between satisfaction rates and PBAC scores less than 75, yet the mean PBAC scores were significantly more favorable for endometrial ablation. This study’s results support the use of a sequential therapy of a less invasive therapy, such as the LNG IUS, prior to performing endometrial ablation.
In this Update, we focus on therapies for abnormal uterine bleeding (AUB) that include a new formulation of a progesterone-only pill (POP), drospirenone 4 mg in a 24/4 regimen (24 days of drospirenone/4 days of inert tablets), which recently showed benefit over the use of desogestrel in a European randomized clinical trial (RCT). Two other commonly used treatments for AUB— the levonorgestrel-releasing intrauterine system (LNG IUS) and endometrial ablation—were studied in terms of cost-effectiveness as well as whether they should be used in combination for added efficacy. In addition, although at times either COVID-19 disease or the COVID-19 vaccine has been blamed for societal and medical problems, one study showed that it is unlikely that significant changes in the menstrual cycle are a result of the COVID-19 vaccine.
COVID-19 vaccination had minimal effects on menstrual cycle length
Edelman A, Boniface ER, Benhar W, et al. Association between menstrual cycle length and coronavirus disease 2019 (COVID-19) vaccination: a US cohort. Obstet Gynecol. 2022;139:481-489.
Does receiving the COVID-19 vaccination result in abnormal menstrual cycles? Patients often ask this question, and it has been a topic of social media discussion (including NPR) and concerns about the possibility of vaccine hesitancy,1,2 as the menstrual cycle is often considered a sign of health and fertility.
To better understand this possible association, Edelman and colleagues conducted a study that prospectively tracked menstrual cycle data using the digital app Natural Cycles in US residents aged 18 to 45 years for 3 consecutive cycles in both a vaccinated and an unvaccinated cohort.3 Almost 4,000 individuals were studied; 2,403 were vaccinated and 1,556 were unvaccinated. The study vaccine types included the BioNTech (Pfizer), Moderna, Johnson & Johnson/Janssen, and unspecified vaccines.
The primary outcome was the within-individual change in cycle length in days, comparing a 3-cycle postvaccine average to a 3-cycle prevaccination average in the 2 groups. (For the unvaccinated group, cycles 1, 2, and 3 were considered the equivalent of prevaccination cycles; cycle 4 was designated as the artificial first vaccine dose-cycle and cycle 5 as the artificial second-dose cycle.)
Increase in cycle length clinically negligible
The investigators found that the vaccinated cohort had less than a 1-day unadjusted increase in the length of their menstrual cycle, which was essentially a 0.71-day increase (98.75% confidence interval [CI], 0.47–0.94). Although this is considered statistically significant, it is likely clinically insignificant in that the overlaid histograms comparing the distribution of change showed a cycle length distribution in vaccinated individuals that is essentially equivalent to that in unvaccinated individuals. After adjusting for confounders, the difference in cycle length was reduced to a 0.64 day (98.75% CI, 0.27–1.01).
An interesting finding was that a subset of individuals who received both vaccine doses in a single cycle had, on average, an adjusted 2-day increase in their menstrual cycle compared with unvaccinated individuals. To explain this slightly longer cycle length, the authors postulated that mRNA vaccines create an immune response, or stressor, which could temporarily affect the hypothalamic-pituitary-ovarian axis if timed correctly. It is certainly possible for an individual to receive 2 doses in a single cycle, which could have both been administered in the early follicular phase. Such cycle length variability can be caused by events, including stressors, that affect the recruitment and maturation of the dominant follicle.
Counseling takeaway
This study provides reassurance to most individuals who receive a COVID-19 vaccine that it likely will not affect their menstrual cycle in a clinically significant manner.
This robust study by Edelman and colleagues on COVID-19 vaccination effects on menstrual cycle length had more than 99% power to detect an unadjusted 1-day difference in cycle length. However, given that most of the study participants were White and had access to the Natural Cycles app, the results may not be generalizable to all individuals who receive the vaccine.
Continue to: Drospirenone improved bleeding profiles, lowered discontinuation rates compared with desogestrel...
Drospirenone improved bleeding profiles, lowered discontinuation rates compared with desogestrel
Regidor PA, Colli E, Palacios S. Overall and bleeding-related discontinuation rates of a new oral contraceptive containing 4 mg drospirenone only in a 24/4 regimen and comparison to 0.075 mg desogestrel. Gynecol Endocrinol. 2021;37:1121-1127.
A new POP, marketed under the name Slynd, recently came to market. It contains the progestin drospirenone (DRSP) 4 mg in a 24/4 regimen. This formulation has the advantage of being an antiandrogenic progestin, with a long enough half-life to allow for managing a missed pill in the same fashion as combined oral contraceptives (COCs).
Investigators in Europe conducted a double-blind, randomized trial to assess discontinuation rates due to adverse events (mainly bleeding disorders) in participants taking DRSP 4 mg in a 24/4 regimen compared with those taking the POP desogestrel (DSG) 0.075 mg, which is commonly used in Europe.4 Regidor and colleagues compared 858 women with 6,691 DRSP treatment cycles with 332 women with 2,487 DSG treatment cycles.
Top reasons for stopping a POP
The discontinuation rate for abnormal bleeding was 3.7% in the DRSP group versus 7.3% in the DSG group (55.7% lower). The most common reasons for stopping either POP formulation were vaginal bleeding and acne. Both of these adverse events were less common in the DRSP group. Pill discontinuation due to vaginal bleeding was 2.6% in the DRSP group versus 5.4% in the DSG group, while discontinuation due to acne occurred in 1% in the DRSP group versus 2.7% in the DSG group.
New oral contraception option
This study shows improved acceptability and bleeding profiles in women using this new DRSP contraception pill regimen.
Adherence to a contraceptive method is influenced by patient satisfaction, and this is particularly important in patients who cannot take COCs. It also should be noted that the discontinuation rate for DRSP as a POP used in this 24/4 regimen was similar to discontinuation rates for COCs containing 20 µg and 30 µg of ethinyl estradiol. Cost, however, may be an issue with DRSP, depending on a patient’s insurance coverage.
Continue to: Placing an LNG IUS after endometrial ablation for heavy menstrual bleeding reduced risk of hysterectomy...
Placing an LNG IUS after endometrial ablation for heavy menstrual bleeding reduced risk of hysterectomy
Oderkerk TJ, van de Kar MMA, van der Zanden CHM, et al. The combined use of endometrial ablation or resection and levonorgestrel-releasing intrauterine system in women with heavy menstrual bleeding: a systematic review. Acta Obstet Gynecol Scand. 2021;100:1779-1787.
Over the years, a smattering of articles have suggested that a reduction in uterine bleeding was associated with placement of an LNG IUS at the conclusion of endometrial ablation. We now have a systematic review of this surgical modification.
Oderkerk and colleagues sifted through 747 articles to find 7 publications that could provide meaningful data on the impact of combined use of endometrial ablation and LNG IUS insertion for women with heavy menstrual bleeding.5 These included 4 retrospective cohort studies with control groups, 2 retrospective studies without control groups, and 1 case series. The primary outcome was the hysterectomy rate after therapy.
Promising results for combined therapy
Although no statistically significant intergroup differences were seen in the combined treatment group versus the endometrial ablation alone group for the first 6 months of treatment, significant differences existed at the 12- and 24-month mark. Hysterectomy rates after combined treatment varied from 0% to 11% versus 9.4% to 24% after endometrial ablation alone. Complication rates for combined treatment did not appear higher than those for endometrial ablation alone.
The authors postulated that the failure of endometrial ablation is generally caused by either remaining or regenerating endometrial tissue and that the addition of an LNG IUS allows for suppression of endometrial tissue. Also encouraging was that, in general, the removal of the LNG IUS was relatively simple. A single difficult removal was described due to uterine synechiae, but hysteroscopic resection was not necessary. The authors acknowledged that the data from these 7 retrospective studies are limited and that high-quality research from prospective studies is needed.
Bottom line
The data available from this systematic review suggest that placement of an LNG IUS at the completion of an endometrial ablation may result in lower hysterectomy rates, without apparent risk, and without significantly difficult LNG IUS removal when needed.
The data provided by Oderkerk and colleagues’ systematic review are promising and, although not studied in the reviewed publications, the potential may exist to reduce the risk of endometrial hyperplasia and endometrial cancer by adding an LNG IUS.
Continue to: LNG IUS is less expensive, and less effective, than endometrial ablation for heavy menstrual bleeding, cost analysis shows...
LNG IUS is less expensive, and less effective, than endometrial ablation for heavy menstrual bleeding, cost analysis shows
van den Brink MJ, Beelen P, Herman MC, et al. The levonorgestrel intrauterine system versus endometrial ablation for heavy menstrual bleeding: a cost-effectiveness analysis. BJOG. 2021;128:2003-2011.
To assess the cost-effectiveness of the LNG IUS versus endometrial ablation in the treatment of heavy menstrual bleeding, van den Brink and colleagues conducted a randomized, noninferiority trial.6
Part of the rationale for this study was to better understand the cost differences between the LNG IUS and second-generation endometrial ablation. Some data have suggested that the LNG IUS is cost-effective when compared with first-generation endometrial ablation; however, definitive evidence about its cost compared with second-generation endometrial ablation is lacking, as these procedures should be less expensive than first-generation endometrial ablation since they frequently are performed in the office rather than in an operating room.
Cost-effectiveness and noninferiority assessed
A total of 270 women were randomly assigned to 1 of 2 treatment strategies. Eventually, 132 women were treated first with the 52-mg LNG IUS, and 138 were treated first with endometrial ablation by radiofrequency ablation. Menstrual blood loss after 24 months was the primary outcome.
At 24 months, the mean pictorial blood loss assessment chart (PBAC) scores were 64.8 in the LNG IUS group compared with 14.2 in the endometrial ablation group. Given that the noninferiority margin was defined as 25 points, noninferiority could not be demonstrated. However, when looking at PBAC scores less than 75 points, the LNG IUS group met this secondary end point in 87% of women versus 94% in the endometrial ablation group. When satisfaction was assessed, 74% of women in the LNG IUS group were satisfied compared with 84% in the endometrial ablation group.
Overall, the total costs per patient were €2,285 in the LNG IUS strategy and €3,465 in the endometrial ablation strategy (costs convert to $2,285 and $3,465 as of this writing).
Key takeaway
Treatment of heavy menstrual bleeding starting with the LNG IUS is cheaper, but it is slightly less effective than endometrial ablation. ●
It is interesting that there are minimal differences between satisfaction rates and PBAC scores less than 75, yet the mean PBAC scores were significantly more favorable for endometrial ablation. This study’s results support the use of a sequential therapy of a less invasive therapy, such as the LNG IUS, prior to performing endometrial ablation.
- Blumfiel G. Why reports of menstrual changes after COVID vaccine are tough to study. NPR. August 9, 2021. Accessed August 30, 2022. https://www.npr.org/sections/health-shots/2021/08/09/1024190379/covid-vaccine-period-menstrual-cycle-research
- Lee KMN, Junkins EJ, Fatima UA, et al. Characterizing menstrual bleeding changes occurring after SARSCoV-2 vaccinations. MedRxiv. February 11, 2022. doi:10.1101/2021.10.11.21264863
- Edelman A, Boniface ER, Benhar W, et al. Association between menstrual cycle length and coronavirus disease 2019 (COVID-19) vaccination: a US cohort. Obstet Gynecol. 2022;139:481-489.
- Regidor PA, Colli E, Palacios S. Overall and bleeding-related discontinuation rates of a new oral contraceptive containing 4 mg drospirenone only in a 24/4 regimen and comparison to 0.075 mg desogestrel. Gynecol Endocrinol. 2021;37:1121-1127.
- Oderkerk TJ, van de Kar MMA, van der Zanden CHM, et al. T he combined use of endometrial ablation or resection and levonorgestrel-releasing intrauterine system in women with heavy menstrual bleeding: a systematic review. Acta Obstet Gynecol Scand. 2021;100:1779-1787.
- van den Brink MJ, Beelen P, Herman MC, et al. The levonorgestrel intrauterine system versus endometrial ablation for heavy menstrual bleeding: a cost-effectiveness analysis. BJOG. 2021;128:2003-2011.
- Blumfiel G. Why reports of menstrual changes after COVID vaccine are tough to study. NPR. August 9, 2021. Accessed August 30, 2022. https://www.npr.org/sections/health-shots/2021/08/09/1024190379/covid-vaccine-period-menstrual-cycle-research
- Lee KMN, Junkins EJ, Fatima UA, et al. Characterizing menstrual bleeding changes occurring after SARSCoV-2 vaccinations. MedRxiv. February 11, 2022. doi:10.1101/2021.10.11.21264863
- Edelman A, Boniface ER, Benhar W, et al. Association between menstrual cycle length and coronavirus disease 2019 (COVID-19) vaccination: a US cohort. Obstet Gynecol. 2022;139:481-489.
- Regidor PA, Colli E, Palacios S. Overall and bleeding-related discontinuation rates of a new oral contraceptive containing 4 mg drospirenone only in a 24/4 regimen and comparison to 0.075 mg desogestrel. Gynecol Endocrinol. 2021;37:1121-1127.
- Oderkerk TJ, van de Kar MMA, van der Zanden CHM, et al. T he combined use of endometrial ablation or resection and levonorgestrel-releasing intrauterine system in women with heavy menstrual bleeding: a systematic review. Acta Obstet Gynecol Scand. 2021;100:1779-1787.
- van den Brink MJ, Beelen P, Herman MC, et al. The levonorgestrel intrauterine system versus endometrial ablation for heavy menstrual bleeding: a cost-effectiveness analysis. BJOG. 2021;128:2003-2011.
ObGyn: A leader in academic medicine, with progress still to be made in diversity
The nation’s population is quickly diversifying, making racial/ethnic disparities in health care outcomes even more apparent. Minority and non-English-speaking populations have grown and may become a majority in the next generation.1 A proposed strategy to reduce disparities in health care is to recruit more practitioners who better reflect the patient populations.2 Improved access to care with racial concordance between physicians and patients has been reported.3
Being increasingly aware of access-to-care data, more patients are advocating and asking for physicians of color to be their providers.4 Despite progress (ie, more women entering the medical profession), the proportion of physicians who are underrepresented in medicine (URiM—eg, Black, Hispanic, and Native American) still lags US population demographics.3
Why diversity in medicine matters
In addition to improving access to care, diversity in medicine offers other benefits. Working within diverse learning environments has demonstrated educational advantages.5,6 Medical students and residents from diverse backgrounds are less likely to report depression symptoms, regardless of their race. Diversity may accelerate advancements in health care as well, since it is well-established that diverse teams outperform nondiverse teams when it comes to innovation and productivity.7 Finally, as a profession committed to equity, advocacy, and justice, physicians are positioned to lead the way toward racial equity.
Overall, racial and gender diversity in all clinical specialties is improving, but not at the same pace. While the diversity of US medical students and residents by sex and race/ethnicity is greater than among faculty, change in racial diversity has been slow for all 3 groups.8 During the past 40 years the number of full-time faculty has increased 6-fold for females and more than tripled for males.8 However, this rise has not favored URiM faculty, because their proportion is still underrepresented relative to their group in the general population. Clinical departments that are making the most progress in recruiting URiM residents and faculty are often primary or preventive care specialties rather than surgical or service or hospital-based specialties.8,9 ObGyn has consistently had a proportion of URiM residents (18%) that is highest in the surgical specialties and comparable to family medicine and pediatrics.10
When examining physician workforce diversity, it is important to “drill down” to individual specialties to obtain a clearer understanding of trends. The continued need for increased resident and faculty diversity prompted us to examine ObGyn departments. The most recent nationwide data were gathered about full-time faculty from the 2021 AAMC Faculty Roster, residents from the 2021 Accreditation Counsel for Graduate Medical Education (ACGME) Data Resource Book, medical student matriculants from 2021 AAMC, and US adult women (defined arbitrarily as 15 years or older) from the 2019 American Community Survey.11-13
Increase in female faculty and residents
The expanding numbers of faculty and residents over a 40-year period (from 1973 to 2012) led to more women and underrepresented minorities in ObGyn than in other major clinical departments.14,15 Women now constitute two-thirds of all ObGyn faculty and are more likely to be junior rather than senior faculty.9 When looking at junior faculty, a higher proportion of junior faculty who are URiM are female. While more junior faculty and residents are female, male faculty are also racially and ethnically diverse.9
- ObGyn is a leader in racial/ethnic diversity in academic medicine.
- The rapid rise of faculty numbers in the past has not favored underrepresented faculty.
- The rise in ObGyn faculty and residents, who were predominantly female, has contributed to greater racial/ethnic diversity.
- Improved patient outcomes with racial concordance between physicians and patients have been reported.
- More patients are advocating and asking for physicians of color to be their clinicians.
- Racial/ethnic diversity of junior faculty and residents is similar to medical students.
- The most underrepresented group is Hispanic, due in part to its rapid growth in the US population.
Continue to: Growth of URiM physicians in ObGyn...
Growth of URiM physicians in ObGyn
The distribution of racial/ethnic groups in 2021 were compared between senior and junior ObGyn faculty and residents with the US adult female population.9 As shown in the FIGURE, the proportion of ObGyn faculty who are White approximates the White US adult female population. The most rapidly growing racial/ethnic group in the US population is Hispanic. Although Hispanic is the best represented ethnicity among junior faculty, the proportions of Hispanics among faculty and residents lag well behind the US population. The proportion of ObGyn faculty who are Black has consistently been less than in the US adult female population. ObGyns who are Asian constitute higher proportions of faculty and residents than in the US adult female population. This finding about Asians is consistent across all clinical specialties.7
Recruiting URiM students into ObGyn is important. Racial and ethnic representation in surgical and nonsurgical residency programs has not substantially improved in the past decade and continues to lag the changing demographics of the US population.10 More students than residents and faculty are Hispanic, which represents a much-needed opportunity for recruitment. By contrast, junior ObGyn faculty are more likely to be Black than residents and students. Native Americans constitute less than 1% of all faculty, residents, students, and US adult females.9 Lastly, race/ethnicity being self-reported as “other” or “unknown” is most common among students and residents, which perhaps represents greater diversity.
Looking back
Increasing diversity in medicine and in ObGyn has not happened by accident. Transformational change requires rectifying any factors that detrimentally affect the racial/ethnic diversity of our medical students, residents, and faculty. For example, biases inherent in key residency application metrics are being recognized, and use of holistic review is increasing. Change is also accelerated by an explicit and public commitment from national organizations. In 2009, the Liaison Committee of Medical Education (LCME) mandated that medical schools engage in practices that focus on recruitment and retention of a diverse workforce. Increases in Black and Hispanic medical students were noted after implementation of this new mandate.16 The ACGME followed suit with similar guidelines in 2019.10
Diversity is one of the foundational strengths of the ObGyn specialty. Important aspects of the specialty are built upon the contributions of women of color, some voluntary and some not. One example is the knowledge of gynecology that was gained through the involuntary and nonanesthetized surgeries performed on
Moving forward
Advancing diversity in ObGyn offers advantages: better representation of patient populations, improving public health by better access to care, enhancing learning in medical education, building more comprehensive research agendas, and driving institutional excellence. While progress has been made, significant work is still to be done. We must continue to critically examine the role of biases and structural racism that are embedded in evaluating medical students, screening of residency applicants, and selecting and retaining faculty. In future work, we should explore the hypothesis that continued change in racial/ethnic diversity of faculty will only occur once more URiM students, especially the growing number of Hispanics, are admitted into medical schools and recruited for residency positions. We should also examine whether further diversity improves patient outcomes.
It is encouraging to realize that ObGyn departments are leaders in racial/ethnic diversity at US medical schools. It is also critical that the specialty commits to the progress that still needs to be made, including increasing diversity among faculty and institutional leadership. To maintain diversity that mirrors the US adult female population, the specialty of ObGyn will require active surveillance and continued recruitment of Black and, especially Hispanic, faculty and residents.19 The national strategies aimed at building medical student and residency diversity are beginning to yield results. For those gains to help faculty diversity, institutional and departmental leaders will need to implement best practices for recruiting, retaining, and advancing URiM faculty.19 Those practices would include making workforce diversity an explicit priority, building diverse applicant pools, and establishing infrastructure and mentorship to advance URiM faculty to senior leadership positions.20
In conclusion
Building a physician workforce that is more representative of the US population should aid in addressing inequalities in health and health care. Significant strides have been made in racial/ethnic diversity in ObGyn. This has resulted in a specialty that is among the most diverse in academic medicine. At the same time, there is more work to be done. For example, the specialty is far from reaching racial equity for Hispanic physicians. Also, continued efforts are necessary to advance URiM faculty to leadership positions. The legacy of racial/ethnic diversity in ObGyn did not happen by accident and will not be maintained without intention. ●
- Hummes KR, Jones NA, Ramierez RR. United States Census: overview of race and Hispanic origin: 2010. http//www. census.gov/prod/cen2010/briefs/c2010br-02.pdf. Accessed May 22, 2022.
- Xierali IM, Castillo-Page L, Zhang K, et al. AM last page: the urgency of physician workforce diversity. Acad Med. 2014;89:1192.
- Association of American Medical College. Diversity in the physician workforce. Facts & figures 2014. http://www .aamcdiversityfactsandfigures.org. Accessed April 9, 2022.
- Marrast LM, Zallman L, Woolhandler S, et al. Minority physicians’ role in the care of underserved patients: Diversifying the physician workforce may be key in addressing health disparities. JAMA Int Med. 2014;174:289-291.
- Amalba A, Abantanga FA, Scherpbier AJ, et al. Community-based education: The influence of role modeling on career choice and practice location. Med Teac. 2017;39:174-180.
- Umbach PD. The contribution of faculty of color to undergraduate education. Res High Educ. 2006;47:317-345.
- Gonzalo JD, Chuang CH, Glod SA, et al. General internists as change agents: opportunities and barriers to leadership in health systems and medical education transformation. J Gen Intern Med. 2020;35:1865-1869.
- Xierali IM, Fair MA, Nivet MA. Faculty diversity in U.S. medical schools: Progress and gaps coexist. AAMC Analysis in Brief. 2016;16. https://www.aamc.org/system/files/reports/1/decem ber2016facultydiversityinu.s.medicalschoolsprogressandga ps.pdf. Accessed May 4, 2022.
- Rayburn WF, Xierali IM, McDade WA. Racial-ethnic diversity of obstetrics and gynecology faculty at medical schools in the United States. Am J Obstet Gynecol. 2022;S00029378(22)00106-5. doi: 10.1016/j.ajog.2022.02.007.
- Hucko L, Al-khersan H, Lopez Dominguez J, et al. Racial and ethnic diversity of U.S. residency programs, 2011-2019. N Engl J Med. 2022;386:22-23.
- Accreditation Council for Graduate Medical Education. Data Resource Book Academic Year 2020-2021. https://www.acgme.org/globalassets/pfassets /publicationsbooks/2020-2021_acgme_databook _document.pdf. Accessed October 24, 2021
- United States Census Bureau. The 2019 American Community Survey 5-Year Public Use Microdata Sample (PUMS) Files.
- Accreditation Council for Graduate Medical Education. Data Resource Book Academic Year 2020-2021. https://www.acgme .org/globalassets/pfassets/publicationsbooks/2020-2021 _acgme_databook_document.pdf. Accessed October 24, 2021.
- Rayburn WF, Liu CQ, Elwell EC, et al. Diversity of physician faculty in obstetrics and gynecology. J Reprod Med. 2016;61:22-26.
- Xierali IM, Nivet MA, Rayburn WF. Full-time faculty in clinical and basic science departments by sex and underrepresented in medicine status: A 40-year review. Acad Med. 2021;96: 568-575.
- Boatright DH, Samuels EA, Cramer LJ, et al. Association between the Liaison Committee on Medical Education’s Diversity Standards and Changes in percentage of medical student sex, race, and ethnicity. JAMA. 2018;320:2267-2269.
- United States National Library of Medicine. Changing the face of medicine.
- https://cfmedicine.nlm.nih.gov/physicians/biography_82. html. Accessed May 5, 2022.
- Christmas M. #SayHerName: Should obstetrics and gynecology reckon with the legacy of JM Sims? Reprod Sci. 2021;28:3282-3284.
- Morgan HK, Winkel AF, Bands E, et al. Promoting diversity, equity, and inclusion in the selection of obstetrician-gynecologists. Obstet Gynecol. 2021;138:272-277.
- Peek ME, Kim KE, Johnson JK, et al. “URM candidates are encouraged to apply”: a national study to identify effective strategies to enhance racial and ethnic faculty diversity in academic departments of medicine. Acad Med. 2013;88:405-412.
The nation’s population is quickly diversifying, making racial/ethnic disparities in health care outcomes even more apparent. Minority and non-English-speaking populations have grown and may become a majority in the next generation.1 A proposed strategy to reduce disparities in health care is to recruit more practitioners who better reflect the patient populations.2 Improved access to care with racial concordance between physicians and patients has been reported.3
Being increasingly aware of access-to-care data, more patients are advocating and asking for physicians of color to be their providers.4 Despite progress (ie, more women entering the medical profession), the proportion of physicians who are underrepresented in medicine (URiM—eg, Black, Hispanic, and Native American) still lags US population demographics.3
Why diversity in medicine matters
In addition to improving access to care, diversity in medicine offers other benefits. Working within diverse learning environments has demonstrated educational advantages.5,6 Medical students and residents from diverse backgrounds are less likely to report depression symptoms, regardless of their race. Diversity may accelerate advancements in health care as well, since it is well-established that diverse teams outperform nondiverse teams when it comes to innovation and productivity.7 Finally, as a profession committed to equity, advocacy, and justice, physicians are positioned to lead the way toward racial equity.
Overall, racial and gender diversity in all clinical specialties is improving, but not at the same pace. While the diversity of US medical students and residents by sex and race/ethnicity is greater than among faculty, change in racial diversity has been slow for all 3 groups.8 During the past 40 years the number of full-time faculty has increased 6-fold for females and more than tripled for males.8 However, this rise has not favored URiM faculty, because their proportion is still underrepresented relative to their group in the general population. Clinical departments that are making the most progress in recruiting URiM residents and faculty are often primary or preventive care specialties rather than surgical or service or hospital-based specialties.8,9 ObGyn has consistently had a proportion of URiM residents (18%) that is highest in the surgical specialties and comparable to family medicine and pediatrics.10
When examining physician workforce diversity, it is important to “drill down” to individual specialties to obtain a clearer understanding of trends. The continued need for increased resident and faculty diversity prompted us to examine ObGyn departments. The most recent nationwide data were gathered about full-time faculty from the 2021 AAMC Faculty Roster, residents from the 2021 Accreditation Counsel for Graduate Medical Education (ACGME) Data Resource Book, medical student matriculants from 2021 AAMC, and US adult women (defined arbitrarily as 15 years or older) from the 2019 American Community Survey.11-13
Increase in female faculty and residents
The expanding numbers of faculty and residents over a 40-year period (from 1973 to 2012) led to more women and underrepresented minorities in ObGyn than in other major clinical departments.14,15 Women now constitute two-thirds of all ObGyn faculty and are more likely to be junior rather than senior faculty.9 When looking at junior faculty, a higher proportion of junior faculty who are URiM are female. While more junior faculty and residents are female, male faculty are also racially and ethnically diverse.9
- ObGyn is a leader in racial/ethnic diversity in academic medicine.
- The rapid rise of faculty numbers in the past has not favored underrepresented faculty.
- The rise in ObGyn faculty and residents, who were predominantly female, has contributed to greater racial/ethnic diversity.
- Improved patient outcomes with racial concordance between physicians and patients have been reported.
- More patients are advocating and asking for physicians of color to be their clinicians.
- Racial/ethnic diversity of junior faculty and residents is similar to medical students.
- The most underrepresented group is Hispanic, due in part to its rapid growth in the US population.
Continue to: Growth of URiM physicians in ObGyn...
Growth of URiM physicians in ObGyn
The distribution of racial/ethnic groups in 2021 were compared between senior and junior ObGyn faculty and residents with the US adult female population.9 As shown in the FIGURE, the proportion of ObGyn faculty who are White approximates the White US adult female population. The most rapidly growing racial/ethnic group in the US population is Hispanic. Although Hispanic is the best represented ethnicity among junior faculty, the proportions of Hispanics among faculty and residents lag well behind the US population. The proportion of ObGyn faculty who are Black has consistently been less than in the US adult female population. ObGyns who are Asian constitute higher proportions of faculty and residents than in the US adult female population. This finding about Asians is consistent across all clinical specialties.7

Recruiting URiM students into ObGyn is important. Racial and ethnic representation in surgical and nonsurgical residency programs has not substantially improved in the past decade and continues to lag the changing demographics of the US population.10 More students than residents and faculty are Hispanic, which represents a much-needed opportunity for recruitment. By contrast, junior ObGyn faculty are more likely to be Black than residents and students. Native Americans constitute less than 1% of all faculty, residents, students, and US adult females.9 Lastly, race/ethnicity being self-reported as “other” or “unknown” is most common among students and residents, which perhaps represents greater diversity.
Looking back
Increasing diversity in medicine and in ObGyn has not happened by accident. Transformational change requires rectifying any factors that detrimentally affect the racial/ethnic diversity of our medical students, residents, and faculty. For example, biases inherent in key residency application metrics are being recognized, and use of holistic review is increasing. Change is also accelerated by an explicit and public commitment from national organizations. In 2009, the Liaison Committee of Medical Education (LCME) mandated that medical schools engage in practices that focus on recruitment and retention of a diverse workforce. Increases in Black and Hispanic medical students were noted after implementation of this new mandate.16 The ACGME followed suit with similar guidelines in 2019.10
Diversity is one of the foundational strengths of the ObGyn specialty. Important aspects of the specialty are built upon the contributions of women of color, some voluntary and some not. One example is the knowledge of gynecology that was gained through the involuntary and nonanesthetized surgeries performed on
Moving forward
Advancing diversity in ObGyn offers advantages: better representation of patient populations, improving public health by better access to care, enhancing learning in medical education, building more comprehensive research agendas, and driving institutional excellence. While progress has been made, significant work is still to be done. We must continue to critically examine the role of biases and structural racism that are embedded in evaluating medical students, screening of residency applicants, and selecting and retaining faculty. In future work, we should explore the hypothesis that continued change in racial/ethnic diversity of faculty will only occur once more URiM students, especially the growing number of Hispanics, are admitted into medical schools and recruited for residency positions. We should also examine whether further diversity improves patient outcomes.
It is encouraging to realize that ObGyn departments are leaders in racial/ethnic diversity at US medical schools. It is also critical that the specialty commits to the progress that still needs to be made, including increasing diversity among faculty and institutional leadership. To maintain diversity that mirrors the US adult female population, the specialty of ObGyn will require active surveillance and continued recruitment of Black and, especially Hispanic, faculty and residents.19 The national strategies aimed at building medical student and residency diversity are beginning to yield results. For those gains to help faculty diversity, institutional and departmental leaders will need to implement best practices for recruiting, retaining, and advancing URiM faculty.19 Those practices would include making workforce diversity an explicit priority, building diverse applicant pools, and establishing infrastructure and mentorship to advance URiM faculty to senior leadership positions.20
In conclusion
Building a physician workforce that is more representative of the US population should aid in addressing inequalities in health and health care. Significant strides have been made in racial/ethnic diversity in ObGyn. This has resulted in a specialty that is among the most diverse in academic medicine. At the same time, there is more work to be done. For example, the specialty is far from reaching racial equity for Hispanic physicians. Also, continued efforts are necessary to advance URiM faculty to leadership positions. The legacy of racial/ethnic diversity in ObGyn did not happen by accident and will not be maintained without intention. ●
The nation’s population is quickly diversifying, making racial/ethnic disparities in health care outcomes even more apparent. Minority and non-English-speaking populations have grown and may become a majority in the next generation.1 A proposed strategy to reduce disparities in health care is to recruit more practitioners who better reflect the patient populations.2 Improved access to care with racial concordance between physicians and patients has been reported.3
Being increasingly aware of access-to-care data, more patients are advocating and asking for physicians of color to be their providers.4 Despite progress (ie, more women entering the medical profession), the proportion of physicians who are underrepresented in medicine (URiM—eg, Black, Hispanic, and Native American) still lags US population demographics.3
Why diversity in medicine matters
In addition to improving access to care, diversity in medicine offers other benefits. Working within diverse learning environments has demonstrated educational advantages.5,6 Medical students and residents from diverse backgrounds are less likely to report depression symptoms, regardless of their race. Diversity may accelerate advancements in health care as well, since it is well-established that diverse teams outperform nondiverse teams when it comes to innovation and productivity.7 Finally, as a profession committed to equity, advocacy, and justice, physicians are positioned to lead the way toward racial equity.
Overall, racial and gender diversity in all clinical specialties is improving, but not at the same pace. While the diversity of US medical students and residents by sex and race/ethnicity is greater than among faculty, change in racial diversity has been slow for all 3 groups.8 During the past 40 years the number of full-time faculty has increased 6-fold for females and more than tripled for males.8 However, this rise has not favored URiM faculty, because their proportion is still underrepresented relative to their group in the general population. Clinical departments that are making the most progress in recruiting URiM residents and faculty are often primary or preventive care specialties rather than surgical or service or hospital-based specialties.8,9 ObGyn has consistently had a proportion of URiM residents (18%) that is highest in the surgical specialties and comparable to family medicine and pediatrics.10
When examining physician workforce diversity, it is important to “drill down” to individual specialties to obtain a clearer understanding of trends. The continued need for increased resident and faculty diversity prompted us to examine ObGyn departments. The most recent nationwide data were gathered about full-time faculty from the 2021 AAMC Faculty Roster, residents from the 2021 Accreditation Counsel for Graduate Medical Education (ACGME) Data Resource Book, medical student matriculants from 2021 AAMC, and US adult women (defined arbitrarily as 15 years or older) from the 2019 American Community Survey.11-13
Increase in female faculty and residents
The expanding numbers of faculty and residents over a 40-year period (from 1973 to 2012) led to more women and underrepresented minorities in ObGyn than in other major clinical departments.14,15 Women now constitute two-thirds of all ObGyn faculty and are more likely to be junior rather than senior faculty.9 When looking at junior faculty, a higher proportion of junior faculty who are URiM are female. While more junior faculty and residents are female, male faculty are also racially and ethnically diverse.9
- ObGyn is a leader in racial/ethnic diversity in academic medicine.
- The rapid rise of faculty numbers in the past has not favored underrepresented faculty.
- The rise in ObGyn faculty and residents, who were predominantly female, has contributed to greater racial/ethnic diversity.
- Improved patient outcomes with racial concordance between physicians and patients have been reported.
- More patients are advocating and asking for physicians of color to be their clinicians.
- Racial/ethnic diversity of junior faculty and residents is similar to medical students.
- The most underrepresented group is Hispanic, due in part to its rapid growth in the US population.
Continue to: Growth of URiM physicians in ObGyn...
Growth of URiM physicians in ObGyn
The distribution of racial/ethnic groups in 2021 were compared between senior and junior ObGyn faculty and residents with the US adult female population.9 As shown in the FIGURE, the proportion of ObGyn faculty who are White approximates the White US adult female population. The most rapidly growing racial/ethnic group in the US population is Hispanic. Although Hispanic is the best represented ethnicity among junior faculty, the proportions of Hispanics among faculty and residents lag well behind the US population. The proportion of ObGyn faculty who are Black has consistently been less than in the US adult female population. ObGyns who are Asian constitute higher proportions of faculty and residents than in the US adult female population. This finding about Asians is consistent across all clinical specialties.7

Recruiting URiM students into ObGyn is important. Racial and ethnic representation in surgical and nonsurgical residency programs has not substantially improved in the past decade and continues to lag the changing demographics of the US population.10 More students than residents and faculty are Hispanic, which represents a much-needed opportunity for recruitment. By contrast, junior ObGyn faculty are more likely to be Black than residents and students. Native Americans constitute less than 1% of all faculty, residents, students, and US adult females.9 Lastly, race/ethnicity being self-reported as “other” or “unknown” is most common among students and residents, which perhaps represents greater diversity.
Looking back
Increasing diversity in medicine and in ObGyn has not happened by accident. Transformational change requires rectifying any factors that detrimentally affect the racial/ethnic diversity of our medical students, residents, and faculty. For example, biases inherent in key residency application metrics are being recognized, and use of holistic review is increasing. Change is also accelerated by an explicit and public commitment from national organizations. In 2009, the Liaison Committee of Medical Education (LCME) mandated that medical schools engage in practices that focus on recruitment and retention of a diverse workforce. Increases in Black and Hispanic medical students were noted after implementation of this new mandate.16 The ACGME followed suit with similar guidelines in 2019.10
Diversity is one of the foundational strengths of the ObGyn specialty. Important aspects of the specialty are built upon the contributions of women of color, some voluntary and some not. One example is the knowledge of gynecology that was gained through the involuntary and nonanesthetized surgeries performed on
Moving forward
Advancing diversity in ObGyn offers advantages: better representation of patient populations, improving public health by better access to care, enhancing learning in medical education, building more comprehensive research agendas, and driving institutional excellence. While progress has been made, significant work is still to be done. We must continue to critically examine the role of biases and structural racism that are embedded in evaluating medical students, screening of residency applicants, and selecting and retaining faculty. In future work, we should explore the hypothesis that continued change in racial/ethnic diversity of faculty will only occur once more URiM students, especially the growing number of Hispanics, are admitted into medical schools and recruited for residency positions. We should also examine whether further diversity improves patient outcomes.
It is encouraging to realize that ObGyn departments are leaders in racial/ethnic diversity at US medical schools. It is also critical that the specialty commits to the progress that still needs to be made, including increasing diversity among faculty and institutional leadership. To maintain diversity that mirrors the US adult female population, the specialty of ObGyn will require active surveillance and continued recruitment of Black and, especially Hispanic, faculty and residents.19 The national strategies aimed at building medical student and residency diversity are beginning to yield results. For those gains to help faculty diversity, institutional and departmental leaders will need to implement best practices for recruiting, retaining, and advancing URiM faculty.19 Those practices would include making workforce diversity an explicit priority, building diverse applicant pools, and establishing infrastructure and mentorship to advance URiM faculty to senior leadership positions.20
In conclusion
Building a physician workforce that is more representative of the US population should aid in addressing inequalities in health and health care. Significant strides have been made in racial/ethnic diversity in ObGyn. This has resulted in a specialty that is among the most diverse in academic medicine. At the same time, there is more work to be done. For example, the specialty is far from reaching racial equity for Hispanic physicians. Also, continued efforts are necessary to advance URiM faculty to leadership positions. The legacy of racial/ethnic diversity in ObGyn did not happen by accident and will not be maintained without intention. ●
- Hummes KR, Jones NA, Ramierez RR. United States Census: overview of race and Hispanic origin: 2010. http//www. census.gov/prod/cen2010/briefs/c2010br-02.pdf. Accessed May 22, 2022.
- Xierali IM, Castillo-Page L, Zhang K, et al. AM last page: the urgency of physician workforce diversity. Acad Med. 2014;89:1192.
- Association of American Medical College. Diversity in the physician workforce. Facts & figures 2014. http://www .aamcdiversityfactsandfigures.org. Accessed April 9, 2022.
- Marrast LM, Zallman L, Woolhandler S, et al. Minority physicians’ role in the care of underserved patients: Diversifying the physician workforce may be key in addressing health disparities. JAMA Int Med. 2014;174:289-291.
- Amalba A, Abantanga FA, Scherpbier AJ, et al. Community-based education: The influence of role modeling on career choice and practice location. Med Teac. 2017;39:174-180.
- Umbach PD. The contribution of faculty of color to undergraduate education. Res High Educ. 2006;47:317-345.
- Gonzalo JD, Chuang CH, Glod SA, et al. General internists as change agents: opportunities and barriers to leadership in health systems and medical education transformation. J Gen Intern Med. 2020;35:1865-1869.
- Xierali IM, Fair MA, Nivet MA. Faculty diversity in U.S. medical schools: Progress and gaps coexist. AAMC Analysis in Brief. 2016;16. https://www.aamc.org/system/files/reports/1/decem ber2016facultydiversityinu.s.medicalschoolsprogressandga ps.pdf. Accessed May 4, 2022.
- Rayburn WF, Xierali IM, McDade WA. Racial-ethnic diversity of obstetrics and gynecology faculty at medical schools in the United States. Am J Obstet Gynecol. 2022;S00029378(22)00106-5. doi: 10.1016/j.ajog.2022.02.007.
- Hucko L, Al-khersan H, Lopez Dominguez J, et al. Racial and ethnic diversity of U.S. residency programs, 2011-2019. N Engl J Med. 2022;386:22-23.
- Accreditation Council for Graduate Medical Education. Data Resource Book Academic Year 2020-2021. https://www.acgme.org/globalassets/pfassets /publicationsbooks/2020-2021_acgme_databook _document.pdf. Accessed October 24, 2021
- United States Census Bureau. The 2019 American Community Survey 5-Year Public Use Microdata Sample (PUMS) Files.
- Accreditation Council for Graduate Medical Education. Data Resource Book Academic Year 2020-2021. https://www.acgme .org/globalassets/pfassets/publicationsbooks/2020-2021 _acgme_databook_document.pdf. Accessed October 24, 2021.
- Rayburn WF, Liu CQ, Elwell EC, et al. Diversity of physician faculty in obstetrics and gynecology. J Reprod Med. 2016;61:22-26.
- Xierali IM, Nivet MA, Rayburn WF. Full-time faculty in clinical and basic science departments by sex and underrepresented in medicine status: A 40-year review. Acad Med. 2021;96: 568-575.
- Boatright DH, Samuels EA, Cramer LJ, et al. Association between the Liaison Committee on Medical Education’s Diversity Standards and Changes in percentage of medical student sex, race, and ethnicity. JAMA. 2018;320:2267-2269.
- United States National Library of Medicine. Changing the face of medicine.
- https://cfmedicine.nlm.nih.gov/physicians/biography_82. html. Accessed May 5, 2022.
- Christmas M. #SayHerName: Should obstetrics and gynecology reckon with the legacy of JM Sims? Reprod Sci. 2021;28:3282-3284.
- Morgan HK, Winkel AF, Bands E, et al. Promoting diversity, equity, and inclusion in the selection of obstetrician-gynecologists. Obstet Gynecol. 2021;138:272-277.
- Peek ME, Kim KE, Johnson JK, et al. “URM candidates are encouraged to apply”: a national study to identify effective strategies to enhance racial and ethnic faculty diversity in academic departments of medicine. Acad Med. 2013;88:405-412.
- Hummes KR, Jones NA, Ramierez RR. United States Census: overview of race and Hispanic origin: 2010. http//www. census.gov/prod/cen2010/briefs/c2010br-02.pdf. Accessed May 22, 2022.
- Xierali IM, Castillo-Page L, Zhang K, et al. AM last page: the urgency of physician workforce diversity. Acad Med. 2014;89:1192.
- Association of American Medical College. Diversity in the physician workforce. Facts & figures 2014. http://www .aamcdiversityfactsandfigures.org. Accessed April 9, 2022.
- Marrast LM, Zallman L, Woolhandler S, et al. Minority physicians’ role in the care of underserved patients: Diversifying the physician workforce may be key in addressing health disparities. JAMA Int Med. 2014;174:289-291.
- Amalba A, Abantanga FA, Scherpbier AJ, et al. Community-based education: The influence of role modeling on career choice and practice location. Med Teac. 2017;39:174-180.
- Umbach PD. The contribution of faculty of color to undergraduate education. Res High Educ. 2006;47:317-345.
- Gonzalo JD, Chuang CH, Glod SA, et al. General internists as change agents: opportunities and barriers to leadership in health systems and medical education transformation. J Gen Intern Med. 2020;35:1865-1869.
- Xierali IM, Fair MA, Nivet MA. Faculty diversity in U.S. medical schools: Progress and gaps coexist. AAMC Analysis in Brief. 2016;16. https://www.aamc.org/system/files/reports/1/decem ber2016facultydiversityinu.s.medicalschoolsprogressandga ps.pdf. Accessed May 4, 2022.
- Rayburn WF, Xierali IM, McDade WA. Racial-ethnic diversity of obstetrics and gynecology faculty at medical schools in the United States. Am J Obstet Gynecol. 2022;S00029378(22)00106-5. doi: 10.1016/j.ajog.2022.02.007.
- Hucko L, Al-khersan H, Lopez Dominguez J, et al. Racial and ethnic diversity of U.S. residency programs, 2011-2019. N Engl J Med. 2022;386:22-23.
- Accreditation Council for Graduate Medical Education. Data Resource Book Academic Year 2020-2021. https://www.acgme.org/globalassets/pfassets /publicationsbooks/2020-2021_acgme_databook _document.pdf. Accessed October 24, 2021
- United States Census Bureau. The 2019 American Community Survey 5-Year Public Use Microdata Sample (PUMS) Files.
- Accreditation Council for Graduate Medical Education. Data Resource Book Academic Year 2020-2021. https://www.acgme .org/globalassets/pfassets/publicationsbooks/2020-2021 _acgme_databook_document.pdf. Accessed October 24, 2021.
- Rayburn WF, Liu CQ, Elwell EC, et al. Diversity of physician faculty in obstetrics and gynecology. J Reprod Med. 2016;61:22-26.
- Xierali IM, Nivet MA, Rayburn WF. Full-time faculty in clinical and basic science departments by sex and underrepresented in medicine status: A 40-year review. Acad Med. 2021;96: 568-575.
- Boatright DH, Samuels EA, Cramer LJ, et al. Association between the Liaison Committee on Medical Education’s Diversity Standards and Changes in percentage of medical student sex, race, and ethnicity. JAMA. 2018;320:2267-2269.
- United States National Library of Medicine. Changing the face of medicine.
- https://cfmedicine.nlm.nih.gov/physicians/biography_82. html. Accessed May 5, 2022.
- Christmas M. #SayHerName: Should obstetrics and gynecology reckon with the legacy of JM Sims? Reprod Sci. 2021;28:3282-3284.
- Morgan HK, Winkel AF, Bands E, et al. Promoting diversity, equity, and inclusion in the selection of obstetrician-gynecologists. Obstet Gynecol. 2021;138:272-277.
- Peek ME, Kim KE, Johnson JK, et al. “URM candidates are encouraged to apply”: a national study to identify effective strategies to enhance racial and ethnic faculty diversity in academic departments of medicine. Acad Med. 2013;88:405-412.
Vismodegib for Basal Cell Carcinoma and Beyond: What Dermatologists Need to Know
Basal cell carcinomas (BCCs) are considered the most common cutaneous cancers. Approximately 80% of nonmelanoma skin cancers are BCCs.1,2 Surgical management is the gold standard for early-stage and localized BCCs; it may include simple excision vs Mohs micrographic surgery.3,4 However, if left untreated, these lesions can progress to an advanced stage (locally advanced BCC) or infrequently may spread to distant sites (metastatic BCC). In the advanced stage, the lesions are no longer manageable by surgery or radiation therapy.5,6 Recently, inhibitors targeting the hedgehog (Hh) pathway have shown great promise for these patients. The first drug approved by the US Food and Drug Administration (FDA) for locally advanced and metastatic BCC is vismodegib.7 In this article, we provide a clinical review of vismodegib for the management of BCC, including a discussion of the Hh pathway in BCC, adverse effects of vismodegib, use of vismodegib in adnexal skin tumors, recommended doses for vismodegib therapy in BCC, and management of the side effects of treatment.
Hh Pathway in BCC
In embryonic development, the Hh signaling pathway is crucial across a broad spectrum of species, including humans. Various members of the Hh family have been recognized, all working as secreted regulatory proteins.8 The name of the Hh signaling pathway is derived from a polypeptide ligand called hedgehog found in some fruit flies. Mutations in the gene led to fruit fly larvae that had a spiky hairy pattern of denticles similar to hedgehogs, leading to the name of this molecule.9 The transmembrane protein smoothened (SMO) is the main component of the Hh signaling pathway and initiates a signaling cascade that in turn leads to an increased expression of target genes, such as GLI1. Patched (PTCH), also a transmembrane protein and a cell-surface receptor for the secreted Hh ligand, suppresses the signaling capacity of SMO. Upon binding of the Hh ligand to the PTCH receptor, the suppression of SMO is relieved and a signal is propagated, evoking a cellular response.10 Molecular and genetic studies have reported that genetic alterations in the Hh signaling pathway are almost universally present in all BCCs, leading to an aberrant activation of the pathway and an uncontrolled proliferation of the basal cells. Frequently, these alterations have been shown to cause loss of function of PTCH homologue 1, which usually acts to inhibit the SMO homologue signaling activity.11,12
Because of the potential importance of Hh signaling in other solid malignancies and the failure of topical inhibition of SMO,13 subsequent studies on the development of Hh pathway inhibitors have mostly focused on the systemic approach. A multitude of Hh pathway inhibitors have been developed thus far, such as SANT1-SANT4, GDC-0449, IPI-926, BMS-833923 (XL139), HhAntag-691, and MK-4101.14 Many of these inhibitors have been clinically investigated.13,15,16
Systemic SMO Inhibitor: Vismodegib
Vismodegib was the earliest systemic SMO inhibitor to fulfill early clinical evaluation15,16 and the first drug to receive FDA approval for the management of advanced or metastatic BCC. Vismodegib is a small-molecule SMO inhibitor used for the management of selected locally advanced BCC and metastatic BCC in adults.3,17 Although there is a possibility of recurrence following drug withdrawal, vismodegib constitutes a new therapeutic strategy presenting positive benefits to patients. It may provide superior improvement over sunitinib, which has shown efficacy in a few patients; however, the efficacy and tolerance of sunitinib have been shown to be limited.18,19
Adverse Effects of Vismodegib Therapy
Adverse events with vismodegib use have been reported in 98% of patients (N=491); most of these were mild to moderate.20 However, the frequency of adverse events could prove to be a therapeutic challenge for patients requiring extended treatment. The most frequently reported reversible side effects were muscle spasms (64%), alopecia (62%), weight loss (33%), fatigue (28%), decreased appetite (25%), diarrhea (17%), nausea (16%), dysgeusia (54%), and ageusia (22%).20 In clinical trials, amenorrhea was noticed in 30% (3/10) of females with reproductive potential.2 Apart from alopecia and possibly amenorrhea, these side effects are reversible.17 Alkeraye et al17 reported 3 clinical cases of persistent alopecia following the use of vismodegib. Amenorrhea is a possible side effect of unknown reversibility.7
Vismodegib is a pregnancy category D medication.4 Severe birth defects, including craniofacial abnormalities, retardations in normal growth, open perineum, and absence of digital fusion at a corresponding 20% of the recommended daily dose, were found in rat studies. Embryo-fetal death was noted when rats were exposedto concentrations comparable to the recommended human dose.4
Hepatic events with the use of vismodegib have been reported. The use of vismodegib in randomized controlled trials resulted in elevation of both alanine aminotransferase and aspartate aminotransferase levels compared with placebo.21 Moreover, severe hepatotoxicity with vismodegib has been reported.22-24 A study conducted by Edwards et al25 concluded that the use of vismodegib in patients with severe liver disease must include thorough risk-benefit assessment, with caution in using other concomitant hepatotoxic medications.
Rare adverse events also have been reported in the literature, including vismodegib-induced pancreatitis in a 79-year-old patient treated for locally advanced, recurrent BCC that was cleared following cessation of therapy.26 Additionally, atypical fibroxanthoma was observed in an 83-year-old patient after 30 days of treatment with vismodegib for multiple BCCs.27 The development of other secondary malignancies, such as squamous cell carcinoma, melanoma, keratoacanthomas, and pilomatricomas, during or after the long-term use of vismodegib also have been described.28-35
Use of Vismodegib for Adnexal Skin Tumors
The role of the sonic Hh–PTCH pathway in the pathogenesis of adnexal tumors varies in the literature. Some studies propose the involvement of this pathway in the formation of adnexal tumors such as trichoblastoma, trichoepithelioma, and cylindroma, as in BCC. Various lines of evidence support this involvement. Firstly, in mice, the spontaneous generation of numerous BCCs, trichoblastomas, trichoepitheliomas, and cylindromas has been observed following constitutive activation of the sonic Hh–PTCH pathway.36 Secondly, in trichoepitheliomas, there have been positive results in molecular research into the tumor suppressor gene PTCH homologue 1, PTCH1, whose mutations cause constitutive activation of the sonic Hh–PTCH pathway.37 Thirdly, GLI138 and SOX939 transcription factors associated with the signaling pathway of sonic Hh–PTCH appear to have increased levels in adnexal carcinomas.19 Lepesant et al19 reported a notable clinical response to vismodegib in trichoblastic carcinoma. Baur et al40 reported successful treatment of multiple familial trichoepitheliomas with vismodegib. Nonetheless, more studies are required to assess the efficacy and reliability of vismodegib in the management of adnexal tumors.
Recommended Dose of Vismodegib Therapy
The vismodegib dosage that is approved by the FDA is 150 mg/d until disease progression or the development of intolerable side effects.4 Higher dosing regimens were evaluated with 270 mg/d and 540 mg/d. No added therapeutic benefit was noted with the increase in the dose, and no dose-limiting toxic effects were observed.41
Management of Vismodegib Side Effects
Managing patient expectations is a crucial step in improving dysgeusia. The experience of dysgeusia varies among patients; thus, patients should be instructed to adjust their diets according to their level of dysgeusia, which can be achieved by changing ingredients or dressings used with their diet. This step has been proven to be effective in overcoming vismodegib-related dysgeusia. Also, fluid taste distortion may lead to dehydration and an increase in creatine level. Thus, patients should be encouraged to monitor fluid intake. Moreover, a treatment hiatus of 2 to 8 months results in near-complete improvement of taste distortion.
For muscle spasms, quinine, treatment break for 1 month, gentle exercise of affected areas, or muscle relaxants such as baclofen and temazepam all are effective methods. For vismodegib-related alopecia, managing patient expectations is key; patients should be aware that hair may take 6 to 12 months or even longer to regrow. In addition, shaving less frequently helps improve alopecia.
For gastrointestinal disorders, loperamide with or without codeine phosphate is effective in resolving diarrhea, and metoclopramide is mostly adequate in treating nausea. Another adverse event is weight loss; weight loss of 5% or more of total body weight prompts dietetic referral. If weight loss persists, a treatment break might be needed to regain weight.
Overall, treatment breaks are sufficient to resolve adverse events caused by vismodegib and do not compromise efficacy of treatment. The duration of a treatment break should be considered before initiation. In one clinical trial, a longer treatment break was associated with fewer adverse effects without affecting the efficacy of treatment.42
Conclusion
Vismodegib provides an effective alternative to surgical intervention in the management of BCC. However, patients must be monitored vigilantly, as adverse events are common (>90%).
- Sekulic A, Migden MR, Oro AE, et al. Efficacy and safety of vismodegib in advanced basal-cell carcinoma. N Engl J Med. 2012;366:2171-2179.
- Rogers HW, Weinstock MA, Harris AR, et al. Incidence estimate of nonmelanoma skin cancer in the United States, 2006. Arch Dermatol. 2010;146:283-287.
- Von Hoff DD, LoRusso PM, Rudin CM, et al. Inhibition of the hedgehog pathway in advanced basal-cell carcinoma. N Engl J Med. 2009;361:1164-1172.
- Cirrone F, Harris CS. Vismodegib and the hedgehog pathway: a new treatment for basal cell carcinoma. Clin Ther. 2012;34:2039-2050.
- Ruiz-Salas V, Alegre M, López-Ferrer A, et al. Vismodegib: a review [article in English, Spanish]. Actas Dermosifiliogr. 2014;105:744-751.
- Rubin AI, Chen EH, Ratner D. Basal-cell carcinoma. N Engl J Med. 2005;353:2262-2269.
- Cusack CA, Nijhawan R, Miller B, et al. Vismodegib for locally advanced basal cell carcinoma in a heart transplant patient. JAMA Dermatol. 2015;151:70-72.
- Aszterbaum M, Rothman A, Johnson RL, et al. Identification of mutations in the human PATCHED gene in sporadic basal cell carcinomas and in patients with the basal cell nevus syndrome. J Invest Dermatol. 1998;110:885-888.
- Abidi A. Hedgehog signaling pathway: a novel target for cancer therapy: vismodegib, a promising therapeutic option in treatment of basal cell carcinomas. Indian J Pharmacol. 2014;46:3-12.
- St-Jacques B, Dassule HR, Karavanova I, et al. Sonic hedgehog signaling is essential for hair development. Curr Biol. 1998;8:1058-1068.
- Gailani MR, Ståhle-Bäckdahl M, Leffell DJ, et al. The role of the human homologue of Drosophila patched in sporadic basal cell carcinomas. Nat Genet. 1996;14:78-81.
- Hall JM, Bell ML, Finger TE. Disruption of sonic hedgehog signaling alters growth and patterning of lingual taste papillae. Dev Biol. 2003;255:263-277.
- Bai CB, Stephen D, Joyner AL. All mouse ventral spinal cord patterning by hedgehog is Gli dependent and involves an activator function of Gli3. Dev Cell. 2004;6:103-115.
- Wang B, Fallon JF, Beachy PA. Hedgehog-regulated processing of Gli3 produces an anterior/posterior repressor gradient in the developing vertebrate limb. Cell. 2000;100:423-434.
- Sekulic A, Mangold AR, Northfelt DW, et al. Advanced basal cell carcinoma of the skin: targeting the hedgehog pathway. Curr Opin Oncol. 2013;25:218-223.
- Ingham PW, Placzek M. Orchestrating ontogenesis: variations on a theme by sonic hedgehog. Nature Rev Genet. 2006;7:841-850.
- Alkeraye S, Maire C, Desmedt E, et al. Persistent alopecia induced by vismodegib. Br J Dermatol. 2015;172:1671-1672.
- Battistella M, Mateus C, Lassau N, et al. Sunitinib efficacy in the treatment of metastatic skin adnexal carcinomas: report of two patients with hidradenocarcinoma and trichoblastic carcinoma. J Eur Acad Dermatol Venereol. 2010;24:199-203.
- Lepesant P, Crinquette M, Alkeraye S, et al. Vismodegib induces significant clinical response in locally advanced trichoblastic carcinoma. Br J Dermatol. 2015;173:1059-1062.
- Basset-Seguin N, Hauschild A, Grob JJ, et al. Vismodegib in patients with advanced basal cell carcinoma (STEVIE): a pre-plannedinterim analysis of an international, open-label trial. Lancet Oncol. 2015;16:729-736.
- Catenacci DV, Junttila MR, Karrison T, et al. Randomized phase Ib/II study of gemcitabine plus placebo or vismodegib, a hedgehog pathway inhibitor, in patients with metastatic pancreatic cancer. J Clin Oncol. 2015;33:4284-4292.
- Sanchez BE, Hajjafar L. Severe hepatotoxicity in a patient treated with hedgehog inhibitor: first case report. Gastroenterology. 2011;140:S974-S975.
- Ly P, Wolf K, Wilson J. A case of hepatotoxicity associated with vismodegib. JAAD Case Rep. 2018;5:57-59.
- Eiger-Moscovich M, Reich E, Tauber G, et al. Efficacy of vismodegib for the treatment of orbital and advanced periocular basal cell carcinoma. Am J Ophthalmol. 2019;207:62-70.
- Edwards BJ, Raisch DW, Saraykar SS, et al. Hepatotoxicity with vismodegib: an MD Anderson Cancer Center and Research on Adverse Drug Events and Reports Project. Drugs R D. 2017;17:211-218.
- Velter C, Blanc J, Robert C. Acute pancreatitis after vismodegib for basal cell carcinoma: a causal relation? Eur J Cancer. 2019;118:67-69.
- Giorgini C, Barbaccia V, Croci GA, et al. Rapid development of atypical fibroxanthoma during vismodegib treatment. Clin Exp Dermatol. 2019;44:86-88.
- Saintes C, Saint-Jean M, Brocard A, et al. Development of squamous cell carcinoma into basal cell carcinoma under treatment with vismodegib. J Eur Acad Dermatol Venereol. 2015;29:1006-1009.
- Zhu GA, Sundram U, Chang ALS. Two different scenarios of squamous cell carcinoma within advanced basal cell carcinomas: cases illustrating the importance of serial biopsy during vismodegib usage. JAMA Dermatol. 2014;150:970-973.
- Poulalhon N, Dalle S, Balme B, et al. Fast-growing cutaneous squamous cell carcinoma in a patient treated with vismodegib. Dermatology. 2015;230:101-104.
- Orouji A, Goerdt S, Utikal J, et al. Multiple highly and moderately differentiated squamous cell carcinomas of the skin during vismodegib treatment of inoperable basal cell carcinoma. Br J Dermatol. 2014;171:431-433.
- Iarrobino A, Messina JL, Kudchadkar R, et al. Emergence of a squamous cell carcinoma phenotype following treatment of metastatic basal cell carcinoma with vismodegib. J Am Acad Dermatol. 2013;69:E33-E34.
- Giuffrida R, Kashofer K, Dika E, et al. Fast growing melanoma following treatment with vismodegib for locally advanced basal cell carcinomas: report of two cases. Eur J Cancer. 2018;91:177-179.
- Aasi S, Silkiss R, Tang JY, et al. New onset of keratoacanthomas after vismodegib treatment for locally advanced basal cell carcinomas: a report of 2 cases. JAMA Dermatol. 2013;149:242-243.
- Magdaleno-Tapial J, Valenzuela-Oñate C, Ortiz-Salvador JM, et al. Pilomatricomas secondary to treatment with vismodegib. JAAD Case Rep. 2018;5:12-14.
- Nilsson M, Undèn AB, Krause D, et al. Induction of basal cell carcinomas and trichoepitheliomas in mice overexpressing GLI-1. Proc Natl Acad Sci U S A. 2000;97:3438-3443.
- Vorechovský I, Undén AB, Sandstedt B, et al. Trichoepitheliomas contain somatic mutations in the overexpressed PTCH gene: support for a gatekeeper mechanism in skin tumorigenesis. Cancer Res. 1997;57:4677-4681.
- Hatta N, Hirano T, Kimura T, et al. Molecular diagnosis of basal cell carcinoma and other basaloid cell neoplasms of the skin by the quantification of Gli1 transcript levels. J Cutan Pathol. 2005;32:131-136.
- Vidal VP, Ortonne N, Schedl A. SOX9 expression is a general marker of basal cell carcinoma and adnexal-related neoplasms. J Cutan Pathol. 2008;35:373-379.
- Baur V, Papadopoulos T, Kazakov DV, et al. A case of multiple familial trichoepitheliomas responding to treatment with the hedgehog signaling pathway inhibitor vismodegib. Virchows Arch. 2018;473:241-246.
- LoRusso PM, Rudin CM, Reddy JC, et al. Phase I trial of hedgehog pathway inhibitor vismodegib (GDC-0449) in patients with refractory, locally advanced or metastatic solid tumors. Clin Cancer Res. 2011;17:2502-2511.
- Fife K, Herd R, Lalondrelle S, et al. Managing adverse events associated with vismodegib in the treatment of basal cell carcinoma. Future Oncol. 2017;13:175-184.
Basal cell carcinomas (BCCs) are considered the most common cutaneous cancers. Approximately 80% of nonmelanoma skin cancers are BCCs.1,2 Surgical management is the gold standard for early-stage and localized BCCs; it may include simple excision vs Mohs micrographic surgery.3,4 However, if left untreated, these lesions can progress to an advanced stage (locally advanced BCC) or infrequently may spread to distant sites (metastatic BCC). In the advanced stage, the lesions are no longer manageable by surgery or radiation therapy.5,6 Recently, inhibitors targeting the hedgehog (Hh) pathway have shown great promise for these patients. The first drug approved by the US Food and Drug Administration (FDA) for locally advanced and metastatic BCC is vismodegib.7 In this article, we provide a clinical review of vismodegib for the management of BCC, including a discussion of the Hh pathway in BCC, adverse effects of vismodegib, use of vismodegib in adnexal skin tumors, recommended doses for vismodegib therapy in BCC, and management of the side effects of treatment.
Hh Pathway in BCC
In embryonic development, the Hh signaling pathway is crucial across a broad spectrum of species, including humans. Various members of the Hh family have been recognized, all working as secreted regulatory proteins.8 The name of the Hh signaling pathway is derived from a polypeptide ligand called hedgehog found in some fruit flies. Mutations in the gene led to fruit fly larvae that had a spiky hairy pattern of denticles similar to hedgehogs, leading to the name of this molecule.9 The transmembrane protein smoothened (SMO) is the main component of the Hh signaling pathway and initiates a signaling cascade that in turn leads to an increased expression of target genes, such as GLI1. Patched (PTCH), also a transmembrane protein and a cell-surface receptor for the secreted Hh ligand, suppresses the signaling capacity of SMO. Upon binding of the Hh ligand to the PTCH receptor, the suppression of SMO is relieved and a signal is propagated, evoking a cellular response.10 Molecular and genetic studies have reported that genetic alterations in the Hh signaling pathway are almost universally present in all BCCs, leading to an aberrant activation of the pathway and an uncontrolled proliferation of the basal cells. Frequently, these alterations have been shown to cause loss of function of PTCH homologue 1, which usually acts to inhibit the SMO homologue signaling activity.11,12
Because of the potential importance of Hh signaling in other solid malignancies and the failure of topical inhibition of SMO,13 subsequent studies on the development of Hh pathway inhibitors have mostly focused on the systemic approach. A multitude of Hh pathway inhibitors have been developed thus far, such as SANT1-SANT4, GDC-0449, IPI-926, BMS-833923 (XL139), HhAntag-691, and MK-4101.14 Many of these inhibitors have been clinically investigated.13,15,16
Systemic SMO Inhibitor: Vismodegib
Vismodegib was the earliest systemic SMO inhibitor to fulfill early clinical evaluation15,16 and the first drug to receive FDA approval for the management of advanced or metastatic BCC. Vismodegib is a small-molecule SMO inhibitor used for the management of selected locally advanced BCC and metastatic BCC in adults.3,17 Although there is a possibility of recurrence following drug withdrawal, vismodegib constitutes a new therapeutic strategy presenting positive benefits to patients. It may provide superior improvement over sunitinib, which has shown efficacy in a few patients; however, the efficacy and tolerance of sunitinib have been shown to be limited.18,19
Adverse Effects of Vismodegib Therapy
Adverse events with vismodegib use have been reported in 98% of patients (N=491); most of these were mild to moderate.20 However, the frequency of adverse events could prove to be a therapeutic challenge for patients requiring extended treatment. The most frequently reported reversible side effects were muscle spasms (64%), alopecia (62%), weight loss (33%), fatigue (28%), decreased appetite (25%), diarrhea (17%), nausea (16%), dysgeusia (54%), and ageusia (22%).20 In clinical trials, amenorrhea was noticed in 30% (3/10) of females with reproductive potential.2 Apart from alopecia and possibly amenorrhea, these side effects are reversible.17 Alkeraye et al17 reported 3 clinical cases of persistent alopecia following the use of vismodegib. Amenorrhea is a possible side effect of unknown reversibility.7
Vismodegib is a pregnancy category D medication.4 Severe birth defects, including craniofacial abnormalities, retardations in normal growth, open perineum, and absence of digital fusion at a corresponding 20% of the recommended daily dose, were found in rat studies. Embryo-fetal death was noted when rats were exposedto concentrations comparable to the recommended human dose.4
Hepatic events with the use of vismodegib have been reported. The use of vismodegib in randomized controlled trials resulted in elevation of both alanine aminotransferase and aspartate aminotransferase levels compared with placebo.21 Moreover, severe hepatotoxicity with vismodegib has been reported.22-24 A study conducted by Edwards et al25 concluded that the use of vismodegib in patients with severe liver disease must include thorough risk-benefit assessment, with caution in using other concomitant hepatotoxic medications.
Rare adverse events also have been reported in the literature, including vismodegib-induced pancreatitis in a 79-year-old patient treated for locally advanced, recurrent BCC that was cleared following cessation of therapy.26 Additionally, atypical fibroxanthoma was observed in an 83-year-old patient after 30 days of treatment with vismodegib for multiple BCCs.27 The development of other secondary malignancies, such as squamous cell carcinoma, melanoma, keratoacanthomas, and pilomatricomas, during or after the long-term use of vismodegib also have been described.28-35
Use of Vismodegib for Adnexal Skin Tumors
The role of the sonic Hh–PTCH pathway in the pathogenesis of adnexal tumors varies in the literature. Some studies propose the involvement of this pathway in the formation of adnexal tumors such as trichoblastoma, trichoepithelioma, and cylindroma, as in BCC. Various lines of evidence support this involvement. Firstly, in mice, the spontaneous generation of numerous BCCs, trichoblastomas, trichoepitheliomas, and cylindromas has been observed following constitutive activation of the sonic Hh–PTCH pathway.36 Secondly, in trichoepitheliomas, there have been positive results in molecular research into the tumor suppressor gene PTCH homologue 1, PTCH1, whose mutations cause constitutive activation of the sonic Hh–PTCH pathway.37 Thirdly, GLI138 and SOX939 transcription factors associated with the signaling pathway of sonic Hh–PTCH appear to have increased levels in adnexal carcinomas.19 Lepesant et al19 reported a notable clinical response to vismodegib in trichoblastic carcinoma. Baur et al40 reported successful treatment of multiple familial trichoepitheliomas with vismodegib. Nonetheless, more studies are required to assess the efficacy and reliability of vismodegib in the management of adnexal tumors.
Recommended Dose of Vismodegib Therapy
The vismodegib dosage that is approved by the FDA is 150 mg/d until disease progression or the development of intolerable side effects.4 Higher dosing regimens were evaluated with 270 mg/d and 540 mg/d. No added therapeutic benefit was noted with the increase in the dose, and no dose-limiting toxic effects were observed.41
Management of Vismodegib Side Effects
Managing patient expectations is a crucial step in improving dysgeusia. The experience of dysgeusia varies among patients; thus, patients should be instructed to adjust their diets according to their level of dysgeusia, which can be achieved by changing ingredients or dressings used with their diet. This step has been proven to be effective in overcoming vismodegib-related dysgeusia. Also, fluid taste distortion may lead to dehydration and an increase in creatine level. Thus, patients should be encouraged to monitor fluid intake. Moreover, a treatment hiatus of 2 to 8 months results in near-complete improvement of taste distortion.
For muscle spasms, quinine, treatment break for 1 month, gentle exercise of affected areas, or muscle relaxants such as baclofen and temazepam all are effective methods. For vismodegib-related alopecia, managing patient expectations is key; patients should be aware that hair may take 6 to 12 months or even longer to regrow. In addition, shaving less frequently helps improve alopecia.
For gastrointestinal disorders, loperamide with or without codeine phosphate is effective in resolving diarrhea, and metoclopramide is mostly adequate in treating nausea. Another adverse event is weight loss; weight loss of 5% or more of total body weight prompts dietetic referral. If weight loss persists, a treatment break might be needed to regain weight.
Overall, treatment breaks are sufficient to resolve adverse events caused by vismodegib and do not compromise efficacy of treatment. The duration of a treatment break should be considered before initiation. In one clinical trial, a longer treatment break was associated with fewer adverse effects without affecting the efficacy of treatment.42
Conclusion
Vismodegib provides an effective alternative to surgical intervention in the management of BCC. However, patients must be monitored vigilantly, as adverse events are common (>90%).
Basal cell carcinomas (BCCs) are considered the most common cutaneous cancers. Approximately 80% of nonmelanoma skin cancers are BCCs.1,2 Surgical management is the gold standard for early-stage and localized BCCs; it may include simple excision vs Mohs micrographic surgery.3,4 However, if left untreated, these lesions can progress to an advanced stage (locally advanced BCC) or infrequently may spread to distant sites (metastatic BCC). In the advanced stage, the lesions are no longer manageable by surgery or radiation therapy.5,6 Recently, inhibitors targeting the hedgehog (Hh) pathway have shown great promise for these patients. The first drug approved by the US Food and Drug Administration (FDA) for locally advanced and metastatic BCC is vismodegib.7 In this article, we provide a clinical review of vismodegib for the management of BCC, including a discussion of the Hh pathway in BCC, adverse effects of vismodegib, use of vismodegib in adnexal skin tumors, recommended doses for vismodegib therapy in BCC, and management of the side effects of treatment.
Hh Pathway in BCC
In embryonic development, the Hh signaling pathway is crucial across a broad spectrum of species, including humans. Various members of the Hh family have been recognized, all working as secreted regulatory proteins.8 The name of the Hh signaling pathway is derived from a polypeptide ligand called hedgehog found in some fruit flies. Mutations in the gene led to fruit fly larvae that had a spiky hairy pattern of denticles similar to hedgehogs, leading to the name of this molecule.9 The transmembrane protein smoothened (SMO) is the main component of the Hh signaling pathway and initiates a signaling cascade that in turn leads to an increased expression of target genes, such as GLI1. Patched (PTCH), also a transmembrane protein and a cell-surface receptor for the secreted Hh ligand, suppresses the signaling capacity of SMO. Upon binding of the Hh ligand to the PTCH receptor, the suppression of SMO is relieved and a signal is propagated, evoking a cellular response.10 Molecular and genetic studies have reported that genetic alterations in the Hh signaling pathway are almost universally present in all BCCs, leading to an aberrant activation of the pathway and an uncontrolled proliferation of the basal cells. Frequently, these alterations have been shown to cause loss of function of PTCH homologue 1, which usually acts to inhibit the SMO homologue signaling activity.11,12
Because of the potential importance of Hh signaling in other solid malignancies and the failure of topical inhibition of SMO,13 subsequent studies on the development of Hh pathway inhibitors have mostly focused on the systemic approach. A multitude of Hh pathway inhibitors have been developed thus far, such as SANT1-SANT4, GDC-0449, IPI-926, BMS-833923 (XL139), HhAntag-691, and MK-4101.14 Many of these inhibitors have been clinically investigated.13,15,16
Systemic SMO Inhibitor: Vismodegib
Vismodegib was the earliest systemic SMO inhibitor to fulfill early clinical evaluation15,16 and the first drug to receive FDA approval for the management of advanced or metastatic BCC. Vismodegib is a small-molecule SMO inhibitor used for the management of selected locally advanced BCC and metastatic BCC in adults.3,17 Although there is a possibility of recurrence following drug withdrawal, vismodegib constitutes a new therapeutic strategy presenting positive benefits to patients. It may provide superior improvement over sunitinib, which has shown efficacy in a few patients; however, the efficacy and tolerance of sunitinib have been shown to be limited.18,19
Adverse Effects of Vismodegib Therapy
Adverse events with vismodegib use have been reported in 98% of patients (N=491); most of these were mild to moderate.20 However, the frequency of adverse events could prove to be a therapeutic challenge for patients requiring extended treatment. The most frequently reported reversible side effects were muscle spasms (64%), alopecia (62%), weight loss (33%), fatigue (28%), decreased appetite (25%), diarrhea (17%), nausea (16%), dysgeusia (54%), and ageusia (22%).20 In clinical trials, amenorrhea was noticed in 30% (3/10) of females with reproductive potential.2 Apart from alopecia and possibly amenorrhea, these side effects are reversible.17 Alkeraye et al17 reported 3 clinical cases of persistent alopecia following the use of vismodegib. Amenorrhea is a possible side effect of unknown reversibility.7
Vismodegib is a pregnancy category D medication.4 Severe birth defects, including craniofacial abnormalities, retardations in normal growth, open perineum, and absence of digital fusion at a corresponding 20% of the recommended daily dose, were found in rat studies. Embryo-fetal death was noted when rats were exposedto concentrations comparable to the recommended human dose.4
Hepatic events with the use of vismodegib have been reported. The use of vismodegib in randomized controlled trials resulted in elevation of both alanine aminotransferase and aspartate aminotransferase levels compared with placebo.21 Moreover, severe hepatotoxicity with vismodegib has been reported.22-24 A study conducted by Edwards et al25 concluded that the use of vismodegib in patients with severe liver disease must include thorough risk-benefit assessment, with caution in using other concomitant hepatotoxic medications.
Rare adverse events also have been reported in the literature, including vismodegib-induced pancreatitis in a 79-year-old patient treated for locally advanced, recurrent BCC that was cleared following cessation of therapy.26 Additionally, atypical fibroxanthoma was observed in an 83-year-old patient after 30 days of treatment with vismodegib for multiple BCCs.27 The development of other secondary malignancies, such as squamous cell carcinoma, melanoma, keratoacanthomas, and pilomatricomas, during or after the long-term use of vismodegib also have been described.28-35
Use of Vismodegib for Adnexal Skin Tumors
The role of the sonic Hh–PTCH pathway in the pathogenesis of adnexal tumors varies in the literature. Some studies propose the involvement of this pathway in the formation of adnexal tumors such as trichoblastoma, trichoepithelioma, and cylindroma, as in BCC. Various lines of evidence support this involvement. Firstly, in mice, the spontaneous generation of numerous BCCs, trichoblastomas, trichoepitheliomas, and cylindromas has been observed following constitutive activation of the sonic Hh–PTCH pathway.36 Secondly, in trichoepitheliomas, there have been positive results in molecular research into the tumor suppressor gene PTCH homologue 1, PTCH1, whose mutations cause constitutive activation of the sonic Hh–PTCH pathway.37 Thirdly, GLI138 and SOX939 transcription factors associated with the signaling pathway of sonic Hh–PTCH appear to have increased levels in adnexal carcinomas.19 Lepesant et al19 reported a notable clinical response to vismodegib in trichoblastic carcinoma. Baur et al40 reported successful treatment of multiple familial trichoepitheliomas with vismodegib. Nonetheless, more studies are required to assess the efficacy and reliability of vismodegib in the management of adnexal tumors.
Recommended Dose of Vismodegib Therapy
The vismodegib dosage that is approved by the FDA is 150 mg/d until disease progression or the development of intolerable side effects.4 Higher dosing regimens were evaluated with 270 mg/d and 540 mg/d. No added therapeutic benefit was noted with the increase in the dose, and no dose-limiting toxic effects were observed.41
Management of Vismodegib Side Effects
Managing patient expectations is a crucial step in improving dysgeusia. The experience of dysgeusia varies among patients; thus, patients should be instructed to adjust their diets according to their level of dysgeusia, which can be achieved by changing ingredients or dressings used with their diet. This step has been proven to be effective in overcoming vismodegib-related dysgeusia. Also, fluid taste distortion may lead to dehydration and an increase in creatine level. Thus, patients should be encouraged to monitor fluid intake. Moreover, a treatment hiatus of 2 to 8 months results in near-complete improvement of taste distortion.
For muscle spasms, quinine, treatment break for 1 month, gentle exercise of affected areas, or muscle relaxants such as baclofen and temazepam all are effective methods. For vismodegib-related alopecia, managing patient expectations is key; patients should be aware that hair may take 6 to 12 months or even longer to regrow. In addition, shaving less frequently helps improve alopecia.
For gastrointestinal disorders, loperamide with or without codeine phosphate is effective in resolving diarrhea, and metoclopramide is mostly adequate in treating nausea. Another adverse event is weight loss; weight loss of 5% or more of total body weight prompts dietetic referral. If weight loss persists, a treatment break might be needed to regain weight.
Overall, treatment breaks are sufficient to resolve adverse events caused by vismodegib and do not compromise efficacy of treatment. The duration of a treatment break should be considered before initiation. In one clinical trial, a longer treatment break was associated with fewer adverse effects without affecting the efficacy of treatment.42
Conclusion
Vismodegib provides an effective alternative to surgical intervention in the management of BCC. However, patients must be monitored vigilantly, as adverse events are common (>90%).
- Sekulic A, Migden MR, Oro AE, et al. Efficacy and safety of vismodegib in advanced basal-cell carcinoma. N Engl J Med. 2012;366:2171-2179.
- Rogers HW, Weinstock MA, Harris AR, et al. Incidence estimate of nonmelanoma skin cancer in the United States, 2006. Arch Dermatol. 2010;146:283-287.
- Von Hoff DD, LoRusso PM, Rudin CM, et al. Inhibition of the hedgehog pathway in advanced basal-cell carcinoma. N Engl J Med. 2009;361:1164-1172.
- Cirrone F, Harris CS. Vismodegib and the hedgehog pathway: a new treatment for basal cell carcinoma. Clin Ther. 2012;34:2039-2050.
- Ruiz-Salas V, Alegre M, López-Ferrer A, et al. Vismodegib: a review [article in English, Spanish]. Actas Dermosifiliogr. 2014;105:744-751.
- Rubin AI, Chen EH, Ratner D. Basal-cell carcinoma. N Engl J Med. 2005;353:2262-2269.
- Cusack CA, Nijhawan R, Miller B, et al. Vismodegib for locally advanced basal cell carcinoma in a heart transplant patient. JAMA Dermatol. 2015;151:70-72.
- Aszterbaum M, Rothman A, Johnson RL, et al. Identification of mutations in the human PATCHED gene in sporadic basal cell carcinomas and in patients with the basal cell nevus syndrome. J Invest Dermatol. 1998;110:885-888.
- Abidi A. Hedgehog signaling pathway: a novel target for cancer therapy: vismodegib, a promising therapeutic option in treatment of basal cell carcinomas. Indian J Pharmacol. 2014;46:3-12.
- St-Jacques B, Dassule HR, Karavanova I, et al. Sonic hedgehog signaling is essential for hair development. Curr Biol. 1998;8:1058-1068.
- Gailani MR, Ståhle-Bäckdahl M, Leffell DJ, et al. The role of the human homologue of Drosophila patched in sporadic basal cell carcinomas. Nat Genet. 1996;14:78-81.
- Hall JM, Bell ML, Finger TE. Disruption of sonic hedgehog signaling alters growth and patterning of lingual taste papillae. Dev Biol. 2003;255:263-277.
- Bai CB, Stephen D, Joyner AL. All mouse ventral spinal cord patterning by hedgehog is Gli dependent and involves an activator function of Gli3. Dev Cell. 2004;6:103-115.
- Wang B, Fallon JF, Beachy PA. Hedgehog-regulated processing of Gli3 produces an anterior/posterior repressor gradient in the developing vertebrate limb. Cell. 2000;100:423-434.
- Sekulic A, Mangold AR, Northfelt DW, et al. Advanced basal cell carcinoma of the skin: targeting the hedgehog pathway. Curr Opin Oncol. 2013;25:218-223.
- Ingham PW, Placzek M. Orchestrating ontogenesis: variations on a theme by sonic hedgehog. Nature Rev Genet. 2006;7:841-850.
- Alkeraye S, Maire C, Desmedt E, et al. Persistent alopecia induced by vismodegib. Br J Dermatol. 2015;172:1671-1672.
- Battistella M, Mateus C, Lassau N, et al. Sunitinib efficacy in the treatment of metastatic skin adnexal carcinomas: report of two patients with hidradenocarcinoma and trichoblastic carcinoma. J Eur Acad Dermatol Venereol. 2010;24:199-203.
- Lepesant P, Crinquette M, Alkeraye S, et al. Vismodegib induces significant clinical response in locally advanced trichoblastic carcinoma. Br J Dermatol. 2015;173:1059-1062.
- Basset-Seguin N, Hauschild A, Grob JJ, et al. Vismodegib in patients with advanced basal cell carcinoma (STEVIE): a pre-plannedinterim analysis of an international, open-label trial. Lancet Oncol. 2015;16:729-736.
- Catenacci DV, Junttila MR, Karrison T, et al. Randomized phase Ib/II study of gemcitabine plus placebo or vismodegib, a hedgehog pathway inhibitor, in patients with metastatic pancreatic cancer. J Clin Oncol. 2015;33:4284-4292.
- Sanchez BE, Hajjafar L. Severe hepatotoxicity in a patient treated with hedgehog inhibitor: first case report. Gastroenterology. 2011;140:S974-S975.
- Ly P, Wolf K, Wilson J. A case of hepatotoxicity associated with vismodegib. JAAD Case Rep. 2018;5:57-59.
- Eiger-Moscovich M, Reich E, Tauber G, et al. Efficacy of vismodegib for the treatment of orbital and advanced periocular basal cell carcinoma. Am J Ophthalmol. 2019;207:62-70.
- Edwards BJ, Raisch DW, Saraykar SS, et al. Hepatotoxicity with vismodegib: an MD Anderson Cancer Center and Research on Adverse Drug Events and Reports Project. Drugs R D. 2017;17:211-218.
- Velter C, Blanc J, Robert C. Acute pancreatitis after vismodegib for basal cell carcinoma: a causal relation? Eur J Cancer. 2019;118:67-69.
- Giorgini C, Barbaccia V, Croci GA, et al. Rapid development of atypical fibroxanthoma during vismodegib treatment. Clin Exp Dermatol. 2019;44:86-88.
- Saintes C, Saint-Jean M, Brocard A, et al. Development of squamous cell carcinoma into basal cell carcinoma under treatment with vismodegib. J Eur Acad Dermatol Venereol. 2015;29:1006-1009.
- Zhu GA, Sundram U, Chang ALS. Two different scenarios of squamous cell carcinoma within advanced basal cell carcinomas: cases illustrating the importance of serial biopsy during vismodegib usage. JAMA Dermatol. 2014;150:970-973.
- Poulalhon N, Dalle S, Balme B, et al. Fast-growing cutaneous squamous cell carcinoma in a patient treated with vismodegib. Dermatology. 2015;230:101-104.
- Orouji A, Goerdt S, Utikal J, et al. Multiple highly and moderately differentiated squamous cell carcinomas of the skin during vismodegib treatment of inoperable basal cell carcinoma. Br J Dermatol. 2014;171:431-433.
- Iarrobino A, Messina JL, Kudchadkar R, et al. Emergence of a squamous cell carcinoma phenotype following treatment of metastatic basal cell carcinoma with vismodegib. J Am Acad Dermatol. 2013;69:E33-E34.
- Giuffrida R, Kashofer K, Dika E, et al. Fast growing melanoma following treatment with vismodegib for locally advanced basal cell carcinomas: report of two cases. Eur J Cancer. 2018;91:177-179.
- Aasi S, Silkiss R, Tang JY, et al. New onset of keratoacanthomas after vismodegib treatment for locally advanced basal cell carcinomas: a report of 2 cases. JAMA Dermatol. 2013;149:242-243.
- Magdaleno-Tapial J, Valenzuela-Oñate C, Ortiz-Salvador JM, et al. Pilomatricomas secondary to treatment with vismodegib. JAAD Case Rep. 2018;5:12-14.
- Nilsson M, Undèn AB, Krause D, et al. Induction of basal cell carcinomas and trichoepitheliomas in mice overexpressing GLI-1. Proc Natl Acad Sci U S A. 2000;97:3438-3443.
- Vorechovský I, Undén AB, Sandstedt B, et al. Trichoepitheliomas contain somatic mutations in the overexpressed PTCH gene: support for a gatekeeper mechanism in skin tumorigenesis. Cancer Res. 1997;57:4677-4681.
- Hatta N, Hirano T, Kimura T, et al. Molecular diagnosis of basal cell carcinoma and other basaloid cell neoplasms of the skin by the quantification of Gli1 transcript levels. J Cutan Pathol. 2005;32:131-136.
- Vidal VP, Ortonne N, Schedl A. SOX9 expression is a general marker of basal cell carcinoma and adnexal-related neoplasms. J Cutan Pathol. 2008;35:373-379.
- Baur V, Papadopoulos T, Kazakov DV, et al. A case of multiple familial trichoepitheliomas responding to treatment with the hedgehog signaling pathway inhibitor vismodegib. Virchows Arch. 2018;473:241-246.
- LoRusso PM, Rudin CM, Reddy JC, et al. Phase I trial of hedgehog pathway inhibitor vismodegib (GDC-0449) in patients with refractory, locally advanced or metastatic solid tumors. Clin Cancer Res. 2011;17:2502-2511.
- Fife K, Herd R, Lalondrelle S, et al. Managing adverse events associated with vismodegib in the treatment of basal cell carcinoma. Future Oncol. 2017;13:175-184.
- Sekulic A, Migden MR, Oro AE, et al. Efficacy and safety of vismodegib in advanced basal-cell carcinoma. N Engl J Med. 2012;366:2171-2179.
- Rogers HW, Weinstock MA, Harris AR, et al. Incidence estimate of nonmelanoma skin cancer in the United States, 2006. Arch Dermatol. 2010;146:283-287.
- Von Hoff DD, LoRusso PM, Rudin CM, et al. Inhibition of the hedgehog pathway in advanced basal-cell carcinoma. N Engl J Med. 2009;361:1164-1172.
- Cirrone F, Harris CS. Vismodegib and the hedgehog pathway: a new treatment for basal cell carcinoma. Clin Ther. 2012;34:2039-2050.
- Ruiz-Salas V, Alegre M, López-Ferrer A, et al. Vismodegib: a review [article in English, Spanish]. Actas Dermosifiliogr. 2014;105:744-751.
- Rubin AI, Chen EH, Ratner D. Basal-cell carcinoma. N Engl J Med. 2005;353:2262-2269.
- Cusack CA, Nijhawan R, Miller B, et al. Vismodegib for locally advanced basal cell carcinoma in a heart transplant patient. JAMA Dermatol. 2015;151:70-72.
- Aszterbaum M, Rothman A, Johnson RL, et al. Identification of mutations in the human PATCHED gene in sporadic basal cell carcinomas and in patients with the basal cell nevus syndrome. J Invest Dermatol. 1998;110:885-888.
- Abidi A. Hedgehog signaling pathway: a novel target for cancer therapy: vismodegib, a promising therapeutic option in treatment of basal cell carcinomas. Indian J Pharmacol. 2014;46:3-12.
- St-Jacques B, Dassule HR, Karavanova I, et al. Sonic hedgehog signaling is essential for hair development. Curr Biol. 1998;8:1058-1068.
- Gailani MR, Ståhle-Bäckdahl M, Leffell DJ, et al. The role of the human homologue of Drosophila patched in sporadic basal cell carcinomas. Nat Genet. 1996;14:78-81.
- Hall JM, Bell ML, Finger TE. Disruption of sonic hedgehog signaling alters growth and patterning of lingual taste papillae. Dev Biol. 2003;255:263-277.
- Bai CB, Stephen D, Joyner AL. All mouse ventral spinal cord patterning by hedgehog is Gli dependent and involves an activator function of Gli3. Dev Cell. 2004;6:103-115.
- Wang B, Fallon JF, Beachy PA. Hedgehog-regulated processing of Gli3 produces an anterior/posterior repressor gradient in the developing vertebrate limb. Cell. 2000;100:423-434.
- Sekulic A, Mangold AR, Northfelt DW, et al. Advanced basal cell carcinoma of the skin: targeting the hedgehog pathway. Curr Opin Oncol. 2013;25:218-223.
- Ingham PW, Placzek M. Orchestrating ontogenesis: variations on a theme by sonic hedgehog. Nature Rev Genet. 2006;7:841-850.
- Alkeraye S, Maire C, Desmedt E, et al. Persistent alopecia induced by vismodegib. Br J Dermatol. 2015;172:1671-1672.
- Battistella M, Mateus C, Lassau N, et al. Sunitinib efficacy in the treatment of metastatic skin adnexal carcinomas: report of two patients with hidradenocarcinoma and trichoblastic carcinoma. J Eur Acad Dermatol Venereol. 2010;24:199-203.
- Lepesant P, Crinquette M, Alkeraye S, et al. Vismodegib induces significant clinical response in locally advanced trichoblastic carcinoma. Br J Dermatol. 2015;173:1059-1062.
- Basset-Seguin N, Hauschild A, Grob JJ, et al. Vismodegib in patients with advanced basal cell carcinoma (STEVIE): a pre-plannedinterim analysis of an international, open-label trial. Lancet Oncol. 2015;16:729-736.
- Catenacci DV, Junttila MR, Karrison T, et al. Randomized phase Ib/II study of gemcitabine plus placebo or vismodegib, a hedgehog pathway inhibitor, in patients with metastatic pancreatic cancer. J Clin Oncol. 2015;33:4284-4292.
- Sanchez BE, Hajjafar L. Severe hepatotoxicity in a patient treated with hedgehog inhibitor: first case report. Gastroenterology. 2011;140:S974-S975.
- Ly P, Wolf K, Wilson J. A case of hepatotoxicity associated with vismodegib. JAAD Case Rep. 2018;5:57-59.
- Eiger-Moscovich M, Reich E, Tauber G, et al. Efficacy of vismodegib for the treatment of orbital and advanced periocular basal cell carcinoma. Am J Ophthalmol. 2019;207:62-70.
- Edwards BJ, Raisch DW, Saraykar SS, et al. Hepatotoxicity with vismodegib: an MD Anderson Cancer Center and Research on Adverse Drug Events and Reports Project. Drugs R D. 2017;17:211-218.
- Velter C, Blanc J, Robert C. Acute pancreatitis after vismodegib for basal cell carcinoma: a causal relation? Eur J Cancer. 2019;118:67-69.
- Giorgini C, Barbaccia V, Croci GA, et al. Rapid development of atypical fibroxanthoma during vismodegib treatment. Clin Exp Dermatol. 2019;44:86-88.
- Saintes C, Saint-Jean M, Brocard A, et al. Development of squamous cell carcinoma into basal cell carcinoma under treatment with vismodegib. J Eur Acad Dermatol Venereol. 2015;29:1006-1009.
- Zhu GA, Sundram U, Chang ALS. Two different scenarios of squamous cell carcinoma within advanced basal cell carcinomas: cases illustrating the importance of serial biopsy during vismodegib usage. JAMA Dermatol. 2014;150:970-973.
- Poulalhon N, Dalle S, Balme B, et al. Fast-growing cutaneous squamous cell carcinoma in a patient treated with vismodegib. Dermatology. 2015;230:101-104.
- Orouji A, Goerdt S, Utikal J, et al. Multiple highly and moderately differentiated squamous cell carcinomas of the skin during vismodegib treatment of inoperable basal cell carcinoma. Br J Dermatol. 2014;171:431-433.
- Iarrobino A, Messina JL, Kudchadkar R, et al. Emergence of a squamous cell carcinoma phenotype following treatment of metastatic basal cell carcinoma with vismodegib. J Am Acad Dermatol. 2013;69:E33-E34.
- Giuffrida R, Kashofer K, Dika E, et al. Fast growing melanoma following treatment with vismodegib for locally advanced basal cell carcinomas: report of two cases. Eur J Cancer. 2018;91:177-179.
- Aasi S, Silkiss R, Tang JY, et al. New onset of keratoacanthomas after vismodegib treatment for locally advanced basal cell carcinomas: a report of 2 cases. JAMA Dermatol. 2013;149:242-243.
- Magdaleno-Tapial J, Valenzuela-Oñate C, Ortiz-Salvador JM, et al. Pilomatricomas secondary to treatment with vismodegib. JAAD Case Rep. 2018;5:12-14.
- Nilsson M, Undèn AB, Krause D, et al. Induction of basal cell carcinomas and trichoepitheliomas in mice overexpressing GLI-1. Proc Natl Acad Sci U S A. 2000;97:3438-3443.
- Vorechovský I, Undén AB, Sandstedt B, et al. Trichoepitheliomas contain somatic mutations in the overexpressed PTCH gene: support for a gatekeeper mechanism in skin tumorigenesis. Cancer Res. 1997;57:4677-4681.
- Hatta N, Hirano T, Kimura T, et al. Molecular diagnosis of basal cell carcinoma and other basaloid cell neoplasms of the skin by the quantification of Gli1 transcript levels. J Cutan Pathol. 2005;32:131-136.
- Vidal VP, Ortonne N, Schedl A. SOX9 expression is a general marker of basal cell carcinoma and adnexal-related neoplasms. J Cutan Pathol. 2008;35:373-379.
- Baur V, Papadopoulos T, Kazakov DV, et al. A case of multiple familial trichoepitheliomas responding to treatment with the hedgehog signaling pathway inhibitor vismodegib. Virchows Arch. 2018;473:241-246.
- LoRusso PM, Rudin CM, Reddy JC, et al. Phase I trial of hedgehog pathway inhibitor vismodegib (GDC-0449) in patients with refractory, locally advanced or metastatic solid tumors. Clin Cancer Res. 2011;17:2502-2511.
- Fife K, Herd R, Lalondrelle S, et al. Managing adverse events associated with vismodegib in the treatment of basal cell carcinoma. Future Oncol. 2017;13:175-184.
Practice Points
- The recommended dosage of vismodegib is 150 mg/d until unendurable side effects develop or disease progression occurs.
- The efficacy of vismodegib in the management of locally advanced basal cell carcinoma (BCC) and metastatic BCC is promising. Thus, it is now considered an effective substitute to surgical therapy.
- Patients using vismodegib must be closely monitored, as it is commonly associated with adverse events.
Utilization and Clinical Benefit of Immune Checkpoint Inhibitor in Veterans With Microsatellite Instability-High Prostate Cancer
Background
The utilization of immune checkpoint inhibitors (ICI) in prostate cancer (PC) can be very effective for patients with mismatch repair-deficiency (as identified by MSI-H by PCR/NGS or dMMR IHC). The use of ICI in this patient population has been associated with high rates of durable response. There is limited published data on factors that may influence patient response and outcomes. The aim of this study is to describe the utilization of and tumor response to ICI in this patient population.
Methods
This is a retrospective study of men with MSI-H PC reported by somatic genomic testing from April 1, 2015 to March 31, 2022 through the VA National Precision Oncology Program (NPOP), who received at least one dose of ICI. The primary objectives are to describe the incidence of MSI-H PC and the utilization of ICI. Descriptive statistics and Kaplan- Meier estimator were used for secondary objectives to determine the prostate-specific antigen decline of at least 50% (PSA50), clinical progression free survival (cPFS), time on ICI as a function of number of prior therapies, the extent of metastasis prior to initiation of ICI, and the correlation of MMR genetic alterations with treatment response.
Results
66 patients with MSI-H PC were identified (1.5% of a total of 4267 patients with PC tested through NPOP). 23 patients (35%) received at least one dose of ICI. 12 of 23 patients (52%) had PSA response. PSA50 responses occurred in 6 patients (50%) and 5 continued to have durable PSA50 at six months. Median cPFS was 280 days (95% CI: 105 days-not reached) and the estimated PFS at six months was 72.2% (95% CI: 35.7%-90.2%). 8 of 12 (67%) responders have received multiple lines of therapy for M1 PC. 8 of 12 patients (67%) had high-volume disease at ICI initiation. Of those patients with a MMR genetic alteration, patients with MLH1 (3/3) and MSH2 (6/8) alterations responded more frequently than those with MSH6 alterations (1/4).
Conclusions
MSI-H PC is rare but response rates to ICI are high and durable. Patients with MLH1 and MSH2 alterations appeared to respond more frequently than those with MSH6. Additional follow-up is ongoing.
Background
The utilization of immune checkpoint inhibitors (ICI) in prostate cancer (PC) can be very effective for patients with mismatch repair-deficiency (as identified by MSI-H by PCR/NGS or dMMR IHC). The use of ICI in this patient population has been associated with high rates of durable response. There is limited published data on factors that may influence patient response and outcomes. The aim of this study is to describe the utilization of and tumor response to ICI in this patient population.
Methods
This is a retrospective study of men with MSI-H PC reported by somatic genomic testing from April 1, 2015 to March 31, 2022 through the VA National Precision Oncology Program (NPOP), who received at least one dose of ICI. The primary objectives are to describe the incidence of MSI-H PC and the utilization of ICI. Descriptive statistics and Kaplan- Meier estimator were used for secondary objectives to determine the prostate-specific antigen decline of at least 50% (PSA50), clinical progression free survival (cPFS), time on ICI as a function of number of prior therapies, the extent of metastasis prior to initiation of ICI, and the correlation of MMR genetic alterations with treatment response.
Results
66 patients with MSI-H PC were identified (1.5% of a total of 4267 patients with PC tested through NPOP). 23 patients (35%) received at least one dose of ICI. 12 of 23 patients (52%) had PSA response. PSA50 responses occurred in 6 patients (50%) and 5 continued to have durable PSA50 at six months. Median cPFS was 280 days (95% CI: 105 days-not reached) and the estimated PFS at six months was 72.2% (95% CI: 35.7%-90.2%). 8 of 12 (67%) responders have received multiple lines of therapy for M1 PC. 8 of 12 patients (67%) had high-volume disease at ICI initiation. Of those patients with a MMR genetic alteration, patients with MLH1 (3/3) and MSH2 (6/8) alterations responded more frequently than those with MSH6 alterations (1/4).
Conclusions
MSI-H PC is rare but response rates to ICI are high and durable. Patients with MLH1 and MSH2 alterations appeared to respond more frequently than those with MSH6. Additional follow-up is ongoing.
Background
The utilization of immune checkpoint inhibitors (ICI) in prostate cancer (PC) can be very effective for patients with mismatch repair-deficiency (as identified by MSI-H by PCR/NGS or dMMR IHC). The use of ICI in this patient population has been associated with high rates of durable response. There is limited published data on factors that may influence patient response and outcomes. The aim of this study is to describe the utilization of and tumor response to ICI in this patient population.
Methods
This is a retrospective study of men with MSI-H PC reported by somatic genomic testing from April 1, 2015 to March 31, 2022 through the VA National Precision Oncology Program (NPOP), who received at least one dose of ICI. The primary objectives are to describe the incidence of MSI-H PC and the utilization of ICI. Descriptive statistics and Kaplan- Meier estimator were used for secondary objectives to determine the prostate-specific antigen decline of at least 50% (PSA50), clinical progression free survival (cPFS), time on ICI as a function of number of prior therapies, the extent of metastasis prior to initiation of ICI, and the correlation of MMR genetic alterations with treatment response.
Results
66 patients with MSI-H PC were identified (1.5% of a total of 4267 patients with PC tested through NPOP). 23 patients (35%) received at least one dose of ICI. 12 of 23 patients (52%) had PSA response. PSA50 responses occurred in 6 patients (50%) and 5 continued to have durable PSA50 at six months. Median cPFS was 280 days (95% CI: 105 days-not reached) and the estimated PFS at six months was 72.2% (95% CI: 35.7%-90.2%). 8 of 12 (67%) responders have received multiple lines of therapy for M1 PC. 8 of 12 patients (67%) had high-volume disease at ICI initiation. Of those patients with a MMR genetic alteration, patients with MLH1 (3/3) and MSH2 (6/8) alterations responded more frequently than those with MSH6 alterations (1/4).
Conclusions
MSI-H PC is rare but response rates to ICI are high and durable. Patients with MLH1 and MSH2 alterations appeared to respond more frequently than those with MSH6. Additional follow-up is ongoing.
Castration-Resistant Prostate Cancer—Not Only Challenging to Treat, but Difficult to Define
Purpose
Examine the impact of different definitions of castration resistance used to identify patients with castration-resistant prostate cancer (CRPC) using electronic health records (EHR).
Background
CRPC is a form of prostate cancer that is resistant to treatment with androgen deprivation therapy (ADT) and is associated with higher morbidity and mortality. Widely used guidelines like the Prostate Cancer Working Group 3 (PCWG 3), the American Urological Association (AUA), and many others differ in their definitions of castration-resistance. Until now, the feasibility of identifying CRPC using different definitions from EHR data has not been studied.
Methods/Data Analyisis
EHR data from the Veterans Health Administration (01/2006-12/2020) were used to identify veterans with CRPC according to the following criteria: 1) PCWG 3—a PSA increase ?25% from the nadir with a minimum rise of 2 ng/mL, while castrate (testosterone < 50 ng/mL); 2) AUA—2 consecutive PSA rises of ?0.2 ng/mL; 3) CRPC screening—a PSA rise of > 0.0 ng/mL within a window of 7–90 days.
Results
36,101 unique patients were identified using 1 of (or a combination of) the 3 CRPC criteria. Approximately 12,775 (35%) patients met all 3 criteria, while 8,589 (24%) were identified by AUA, 4,785 (13%) by CRPC screening, and 145 (0.4%) by PCWG3. A total of 8,377 (23%) patients met both the AUA and CRPC screening criteria, 1,219 (3%) patients met the AUA and PCWG3 criteria, and 211 (1%) met the PCWG3 and CRPC screening criteria.
Conculsions/Implications
Although several definitions can be used to identify CRPC patients, a combination of these definitions results in the greatest yield of CRPC patients identified using EHR data. Even though the PCWG3 criterion is frequently used in both clinical trials research and retrospective observational research, PCWG3 may miss many patients meeting other criteria and should not be used by itself when studying patients with CRPC identified from EHR data.
Purpose
Examine the impact of different definitions of castration resistance used to identify patients with castration-resistant prostate cancer (CRPC) using electronic health records (EHR).
Background
CRPC is a form of prostate cancer that is resistant to treatment with androgen deprivation therapy (ADT) and is associated with higher morbidity and mortality. Widely used guidelines like the Prostate Cancer Working Group 3 (PCWG 3), the American Urological Association (AUA), and many others differ in their definitions of castration-resistance. Until now, the feasibility of identifying CRPC using different definitions from EHR data has not been studied.
Methods/Data Analyisis
EHR data from the Veterans Health Administration (01/2006-12/2020) were used to identify veterans with CRPC according to the following criteria: 1) PCWG 3—a PSA increase ?25% from the nadir with a minimum rise of 2 ng/mL, while castrate (testosterone < 50 ng/mL); 2) AUA—2 consecutive PSA rises of ?0.2 ng/mL; 3) CRPC screening—a PSA rise of > 0.0 ng/mL within a window of 7–90 days.
Results
36,101 unique patients were identified using 1 of (or a combination of) the 3 CRPC criteria. Approximately 12,775 (35%) patients met all 3 criteria, while 8,589 (24%) were identified by AUA, 4,785 (13%) by CRPC screening, and 145 (0.4%) by PCWG3. A total of 8,377 (23%) patients met both the AUA and CRPC screening criteria, 1,219 (3%) patients met the AUA and PCWG3 criteria, and 211 (1%) met the PCWG3 and CRPC screening criteria.
Conculsions/Implications
Although several definitions can be used to identify CRPC patients, a combination of these definitions results in the greatest yield of CRPC patients identified using EHR data. Even though the PCWG3 criterion is frequently used in both clinical trials research and retrospective observational research, PCWG3 may miss many patients meeting other criteria and should not be used by itself when studying patients with CRPC identified from EHR data.
Purpose
Examine the impact of different definitions of castration resistance used to identify patients with castration-resistant prostate cancer (CRPC) using electronic health records (EHR).
Background
CRPC is a form of prostate cancer that is resistant to treatment with androgen deprivation therapy (ADT) and is associated with higher morbidity and mortality. Widely used guidelines like the Prostate Cancer Working Group 3 (PCWG 3), the American Urological Association (AUA), and many others differ in their definitions of castration-resistance. Until now, the feasibility of identifying CRPC using different definitions from EHR data has not been studied.
Methods/Data Analyisis
EHR data from the Veterans Health Administration (01/2006-12/2020) were used to identify veterans with CRPC according to the following criteria: 1) PCWG 3—a PSA increase ?25% from the nadir with a minimum rise of 2 ng/mL, while castrate (testosterone < 50 ng/mL); 2) AUA—2 consecutive PSA rises of ?0.2 ng/mL; 3) CRPC screening—a PSA rise of > 0.0 ng/mL within a window of 7–90 days.
Results
36,101 unique patients were identified using 1 of (or a combination of) the 3 CRPC criteria. Approximately 12,775 (35%) patients met all 3 criteria, while 8,589 (24%) were identified by AUA, 4,785 (13%) by CRPC screening, and 145 (0.4%) by PCWG3. A total of 8,377 (23%) patients met both the AUA and CRPC screening criteria, 1,219 (3%) patients met the AUA and PCWG3 criteria, and 211 (1%) met the PCWG3 and CRPC screening criteria.
Conculsions/Implications
Although several definitions can be used to identify CRPC patients, a combination of these definitions results in the greatest yield of CRPC patients identified using EHR data. Even though the PCWG3 criterion is frequently used in both clinical trials research and retrospective observational research, PCWG3 may miss many patients meeting other criteria and should not be used by itself when studying patients with CRPC identified from EHR data.
MYO1E DNA Methylation in U.S. Military Veterans With Adenocarcinoma of the Lung Is Associated With Increased Mortality Risk
Project Purpose
The aim is to assess the role of MYO1E in survival among veterans with lung adenocarcinoma (LUAD).
Background
Veterans have a higher smoking exposure than civilians; a higher incidence of lung cancer; and a younger age at diagnosis of lung cancer. We recently showed that MYO1E DNA methylation and RNA expression in LUAD are associated with survival among civilians.
Methods
This is a retrospective cohort study involving LUAD among civilians and veterans with biopsy or pathologically proven LUAD from surgical specimens. DNA extraction and isolation from FFPE cancer tissues was performed using methylation-onbeads as previously published, followed by qMSP with bisulfite treatment to quantify DNA methylation. RNA extraction and quantification from lung tissues was obtained as described in previous publications.
Data Analysis
Differences were assessed with Wilcoxon rank sum test for continuous variables and Fisher’s exact test for categorical. Two-tailed log-rank test was used to estimate overall survival differences and Cox hazard models, to quantify risk of mortality using hazard ratios (HRs) with 95% confidence intervals (CIs).
Results
There were 91 LUAD patients, 27 veterans and 64 civilians. Veterans were older than civilians, aged 70 years vs aged 66 years (P = .003); with higher proportions of males, 93% vs 69% (P = .03); higher proportion of African Americans, 67% vs 39% (P = .03); smoking more, 50 pack-year vs 40 (0.005), and having a higher proportion of grade I, 78% vs 55% (P = .036). Survival was statistically longer for MYO1E high DNA methylation group 48 months vs 33 for low methylation (P = .049). MYO1E RNA expression did not show statistically significant differences (P = .32). Multivariate Cox regression analysis adjusted by age, veteran/civil status, gender, race, packyear, and stage showed that DNA methylation was significantly associated with mortality risk (HR 5.14; 95% CI, 1.12-23.60) (P = .035).
Conclusions/Implications
This study suggests the utility of MYO1E DNA methylation as a prognostic biomarker for veterans with LUAD. Further studies are necessary to understand the role of MYO1E in chemotherapy resistance and microenvironment immune modulation. Given the low expression of MYO1E in blood cells, MYO1E DNA methylation has the potential to be used as circulating tumor marker in liquid biopsies.
Project Purpose
The aim is to assess the role of MYO1E in survival among veterans with lung adenocarcinoma (LUAD).
Background
Veterans have a higher smoking exposure than civilians; a higher incidence of lung cancer; and a younger age at diagnosis of lung cancer. We recently showed that MYO1E DNA methylation and RNA expression in LUAD are associated with survival among civilians.
Methods
This is a retrospective cohort study involving LUAD among civilians and veterans with biopsy or pathologically proven LUAD from surgical specimens. DNA extraction and isolation from FFPE cancer tissues was performed using methylation-onbeads as previously published, followed by qMSP with bisulfite treatment to quantify DNA methylation. RNA extraction and quantification from lung tissues was obtained as described in previous publications.
Data Analysis
Differences were assessed with Wilcoxon rank sum test for continuous variables and Fisher’s exact test for categorical. Two-tailed log-rank test was used to estimate overall survival differences and Cox hazard models, to quantify risk of mortality using hazard ratios (HRs) with 95% confidence intervals (CIs).
Results
There were 91 LUAD patients, 27 veterans and 64 civilians. Veterans were older than civilians, aged 70 years vs aged 66 years (P = .003); with higher proportions of males, 93% vs 69% (P = .03); higher proportion of African Americans, 67% vs 39% (P = .03); smoking more, 50 pack-year vs 40 (0.005), and having a higher proportion of grade I, 78% vs 55% (P = .036). Survival was statistically longer for MYO1E high DNA methylation group 48 months vs 33 for low methylation (P = .049). MYO1E RNA expression did not show statistically significant differences (P = .32). Multivariate Cox regression analysis adjusted by age, veteran/civil status, gender, race, packyear, and stage showed that DNA methylation was significantly associated with mortality risk (HR 5.14; 95% CI, 1.12-23.60) (P = .035).
Conclusions/Implications
This study suggests the utility of MYO1E DNA methylation as a prognostic biomarker for veterans with LUAD. Further studies are necessary to understand the role of MYO1E in chemotherapy resistance and microenvironment immune modulation. Given the low expression of MYO1E in blood cells, MYO1E DNA methylation has the potential to be used as circulating tumor marker in liquid biopsies.
Project Purpose
The aim is to assess the role of MYO1E in survival among veterans with lung adenocarcinoma (LUAD).
Background
Veterans have a higher smoking exposure than civilians; a higher incidence of lung cancer; and a younger age at diagnosis of lung cancer. We recently showed that MYO1E DNA methylation and RNA expression in LUAD are associated with survival among civilians.
Methods
This is a retrospective cohort study involving LUAD among civilians and veterans with biopsy or pathologically proven LUAD from surgical specimens. DNA extraction and isolation from FFPE cancer tissues was performed using methylation-onbeads as previously published, followed by qMSP with bisulfite treatment to quantify DNA methylation. RNA extraction and quantification from lung tissues was obtained as described in previous publications.
Data Analysis
Differences were assessed with Wilcoxon rank sum test for continuous variables and Fisher’s exact test for categorical. Two-tailed log-rank test was used to estimate overall survival differences and Cox hazard models, to quantify risk of mortality using hazard ratios (HRs) with 95% confidence intervals (CIs).
Results
There were 91 LUAD patients, 27 veterans and 64 civilians. Veterans were older than civilians, aged 70 years vs aged 66 years (P = .003); with higher proportions of males, 93% vs 69% (P = .03); higher proportion of African Americans, 67% vs 39% (P = .03); smoking more, 50 pack-year vs 40 (0.005), and having a higher proportion of grade I, 78% vs 55% (P = .036). Survival was statistically longer for MYO1E high DNA methylation group 48 months vs 33 for low methylation (P = .049). MYO1E RNA expression did not show statistically significant differences (P = .32). Multivariate Cox regression analysis adjusted by age, veteran/civil status, gender, race, packyear, and stage showed that DNA methylation was significantly associated with mortality risk (HR 5.14; 95% CI, 1.12-23.60) (P = .035).
Conclusions/Implications
This study suggests the utility of MYO1E DNA methylation as a prognostic biomarker for veterans with LUAD. Further studies are necessary to understand the role of MYO1E in chemotherapy resistance and microenvironment immune modulation. Given the low expression of MYO1E in blood cells, MYO1E DNA methylation has the potential to be used as circulating tumor marker in liquid biopsies.
Safety Profile of Mutant EGFR-TK Inhibitors in Advanced Non–Small Cell Lung Cancer: A Meta-analysis
Lung cancer has been the leading cause of cancer-related mortality for decades. It is also predicted to remain as the leading cause of cancer-related mortality through 2030.1 Platinum-based chemotherapy, including carboplatin and paclitaxel, was introduced 3 decades ago and revolutionized the management of advanced non–small cell lung cancer (NSCLC). A more recent advancement has been mutant epidermal growth factor receptor–tyrosine kinase (EGFR-TK) inhibitors.1 EGFR is a transmembrane protein that functions by transducing essential growth factor signaling from the extracellular milieu to the cell. As 60% of the advanced NSCLC expresses this receptor, blocking the mutant EGFR receptor was a groundbreaking development in the management of advanced NSCLC.2 Development of mutant EGFR-TK inhibitors has revolutionized the management of advanced NSCLC. This study was conducted to determine the safety profile of mutant EGFR-TK inhibitors in the management of advanced NSCLC.
Methods
This meta-analysis was conducted according to Cochrane Collaboration guidelines and reported as per Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines. The findings are summarized in the PRISMA flow diagram (Figure 1). Two authors (MZ and MM) performed a systematic literature search using databases such as MEDLINE (via PubMed), Embase, and Cochrane Library using the medical search terms and their respective entry words with the following search strategy: safety, “mutant EGFR-TK inhibitors,” advanced, “non–small cell,” “lung cancer,” “adverse effect,” and literature. Additionally, unpublished trials were identified from clinicaltrials.gov, and references of all pertinent articles were also scrutinized to ensure the inclusion of all relevant studies. The search was completed on June 1, 2021, and we only included studies available in English. Two authors (MM and MZ) independently screened the search results in a 2-step process based on predetermined inclusion/exclusion criteria. First, 890 articles were evaluated for relevance on title and abstract level, followed by full-text screening of the final list of 140 articles. Any disagreements were resolved by discussion or third-party review, and a total of 9 articles were included in the study.
The following eligibility criteria were used: original articles reporting adverse effects (AEs) of mutant EGFR-TK inhibitors in patients with advanced NSCLC compared with control groups receiving platinum-based chemotherapy. All the patients included in the study had an EGFR mutation but randomly assigned to either treatment or control group. All articles with subjective data on mutant EGFR-TK inhibitors AEs in patients with advanced NSCLC compared with control groups receiving platinum-based chemotherapy were included in the analysis. Only 9 articles qualified the aforementioned selection criteria for eligibility. All qualifying studies were nationwide inpatient or pooled clinical trials data. The reasons for exclusion of the other 71 articles were irrelevant (n = 31), duplicate (n = 13), reviews (n = 14), and poor data reporting (n = 12). Out of the 9 included studies, 9 studies showed correlation of AEs, including rash, diarrhea, nausea, and fatigue. Seven studies showed correlation of AEs including neutropenia, anorexia, and vomiting. Six studies showed correlation of anemia, cough, and stomatitis. Five studies showed correlation of elevated aspartate aminotransferase (AST), alanine aminotransferase (ALT), and leucopenia. Four studies showed correlation of fever between mutant EGFR-TK inhibitors and platinum-based chemotherapy.
The primary endpoints were reported AEs including rash, diarrhea, elevated ALT, elevated AST, stomatitis, nausea, leucopenia, fatigue, neutropenia, anorexia, anemia, cough, vomiting, and fever, respectively. Data on baseline characteristics and clinical outcomes were then extracted, and summary tables were created. Summary estimates of the clinical endpoints were then calculated with risk ratio (RR) and 95% confidence intervals (CIs) using the random-effects model. Heterogeneity between studies was examined with the Cochran Q I2 statistic which can be defined as low (25% to 50%), moderate (50% to 75%), or high (> 75%). Statistical analysis was performed using Comprehensive Meta-Analysis Software CMA Version 3.0.
Results
A total of 9 studies including 3415 patients (1775 in EGFR-TK inhibitor treatment group while 1640 patients in platinum-based chemotherapy control group) were included in the study. All 9 studies were phase III randomized control clinical trials conducted to compare the safety profile of mutant EGFR-TK inhibitors in patients with advanced NSCLC. Mean age was 61 years in both treatment and control groups. Further details on study and participant characteristics and safety profile including AEs are summarized in Tables 1 and 2. No evidence of publication bias was found.
Rash developed in 45.8% of patients in the treatment group receiving mutant EGFR-TK inhibitors vs only 5.6% of patients in the control group receiving platinum-based chemotherapy. Overall RR of 7.38 with the 95% CI noted, which was statistically significant, confirming higher rash event rates in patients receiving EGFR-TK inhibitors for their advanced NSCLC (Figure 2).
Diarrhea occurred in 33.6% of patients in the mutant EGFR-TK inhibitors treatment group vs 13.5% of patients in the control group receiving platinum-based chemotherapy. Overall RR of 2.63 and 95% CI was noted, which was statistically significant, confirming higher diarrheal rates in patients receiving EGFR-TK inhibitors for their advanced NSCLC (Figure 3).
Elevated ALT levels developed in 27.9% of patients in the treatment group receiving mutant EGFR-TK inhibitors compared with 15.1% of patients in the control group receiving platinum-based chemotherapy. Overall RR of 1.37 and 95% CI was noted, which was statistically significant, confirming higher ALT levels in patients receiving EGFR-TK inhibitors for their advanced NSCLC (Figure 4).
Elevated AST levels occurred in 40.7% of patients in the mutant EGFR-TK inhibitors treatment group vs 12.8% of patients in the control group receiving platinum-based chemotherapy. Overall RR of 1.77 and 95% CI was noted, which was statistically significant, confirming elevated AST levels in patients receiving EGFR-TK inhibitors for their advanced NSCLC (Figure 5).
Stomatitis developed in 17.2% of patients in the treatment group receiving mutant EGFR-TK inhibitors compared with 7.9% of patients in the control group receiving platinum-based chemotherapy. Overall RR of 1.53 and 95% CI was noted, which was statistically significant, confirming higher stomatitis event rates in patients receiving EGFR-TK inhibitors for their advanced NSCLC (Figure 6).
Nausea occurred in 16.5% of patients in the mutant EGFR-TK inhibitors group vs 42.5% of patients in the control group receiving platinum-based chemotherapy. Overall RR of 0.37 and 95% CI was noted, which was statistically significant, confirming higher nausea rates in patients receiving platinum-based chemotherapy compared with treatment group for their advanced NSCLC (Figure 7).
Leucopenia developed in 9.7% of patients in the mutant EGFR-TK inhibitors group compared with 51.3% of patients in the control group receiving platinum-based chemotherapy. Overall RR of 0.18 and 95% CI was noted, which was statistically significant, confirming higher leucopenia incidence in patients receiving platinum-based chemotherapy compared with treatment group for their advanced NSCLC (Figure 8).
Fatigue was reported in 17% of patients in the mutant EGFR-TK inhibitors group compared with 29.5% of patients in the control group receiving platinum-based chemotherapy. Overall RR of 0.59 and 95% CI was noted, which was statistically significant, confirming higher fatigue rates in patients receiving platinum-based chemotherapy compared with treatment group for their advanced NSCLC (Figure 9).
Neutropenia developed in 6.1% of patients in the mutant EGFR-TK inhibitors group vs 48.2% of patients in the control group receiving platinum-based chemotherapy. Overall RR of 0.11 and 95% CI was noted, which was statistically significant, confirming higher neutropenia rates in patients receiving platinum-based chemotherapy compared with the treatment group for their advanced NSCLC (Figure 10).
Anorexia developed in 21.3% of patients in the mutant EGFR-TK inhibitors group vs 31.4% of patients in the control group receiving platinum-based chemotherapy. Overall RR of 0.44 and 95% CI was noted, which was statistically significant, confirming higher anorexia rates in patients receiving platinum-based chemotherapy compared with the treatment group for their advanced NSCLC (Figure 11).
Anemia occurred in 8.7% of patients in the mutant EGFR-TK inhibitors group compared with 32.1% of patients in the control group receiving platinum-based chemotherapy. Overall RR of 0.24 and 95% CI was noted, which was statistically significant, confirming higher anorexia rates in patients receiving platinum-based chemotherapy compared with treatment for their advanced NSCLC (Figure 12).
Cough was reported in 17.8% of patients in the mutant EGFR-TK inhibitors group compared with 18.9% of patients in the control group receiving platinum-based chemotherapy. Overall RR of 0.99 and 95% CI was noted, which was statistically significant, confirming slightly higher cough rates in patients receiving platinum-based chemotherapy compared with treatment for their advanced NSCLC (Figure 13).
Vomiting developed in 11% of patients in the mutant EGFR-TK inhibitors group vs 30.1% of patients in the control group receiving platinum-based chemotherapy. Overall RR of 0.35 and 95% CI was noted, which was statistically significant, confirming higher vomiting rates in patients receiving platinum-based chemotherapy compared with the treatment group for their advanced NSCLC (Figure 14).
Fever occurred in 5.6% of patients in the mutant EGFR-TK inhibitors group compared with 30.1% of patients in the control group receiving platinum-based chemotherapy. Overall RR of 0.41 and 95% CI was noted, which was statistically significant, confirming higher fever rates in patients receiving platinum-based chemotherapy compared with the treatment group for their advanced NSCLC (Figure 15).
Discussion
Despite the advancement in the treatment of metastatic NSCLC, lung cancer stays as most common cause of cancer-related death in North America and European countries, as patients usually have an advanced disease at the time of diagnosis.3 In the past, platinum-based chemotherapy remained the standard of care for most of the patients affected with advanced NSCLC, but the higher recurrence rate and increase in frequency and intensity of AEs with platinum-based chemotherapy led to the development of targeted therapy for NSCLC, one of which includes
Smoking is the most common reversible risk factor associated with lung cancer. The EURTAC trial was the first perspective study in this regard, which compared safety and efficacy of mutant EGFR-TK inhibitors with platinum-based chemotherapy. Results analyzed in this study were in favor of mutant EGFR-TK inhibitors except in the group of former smokers.5 On the contrary, the OPTIMAL trial showed results in favor of mutant EGFR-TK inhibitors both in active and former smokers; this trial also confirmed the efficacy of mutant EGFR-TK inhibitors in European and Asian populations, confirming the rationale for routine testing of EGFR mutation in all the patients being diagnosed with advanced NSCLC.6 Similarly, osimertinib is one of the most recent mutant EGFR-TK inhibitors developed for the treatment of advanced NSCLC in patients with EGFR-positive receptors.
According to the FLAURA trial, patients receiving osimertinib showed significantly longer progression-free survival compared with platinum-based chemotherapy and early mutant EGFR-TK inhibitors. Median progression-free survival was noted to be 18.9 months, which showed 54% lower risk of disease progression in the treatment group receiving osimertinib.7 The ARCHER study emphasized a significant improvement in overall survival as well as progression-free survival among a patient population receiving dacomitinib compared with platinum-based chemotherapy.8,9
Being a potent targeted therapy, mutant EGFR-TK inhibitors do come with some AEs including diarrhea, which was seen in 33.6% of the patients receiving mutant EGFR-TK inhibitors in our study vs 53% in the chemotherapy group, as was observed in the study conducted by Pless and colleagues.10 Similarly, only 16.5% of patients receiving mutant EGFR-TK inhibitors developed nausea compared with 66% being observed in patients receiving chemotherapy. Correspondingly, only a small fraction of patients (9.7%) receiving mutant EGFR-TK inhibitors developed leucopenia, which was 10 times less reported in mutant EGFR-TK inhibitors compared with patients receiving chemotherapy having a percentage of 100%. A similar trend was reported for neutropenia and anemia in mutant EGFR-TK inhibitors with an incidence of 6.1% and 8.7%, compared with the platinum-based chemotherapy group in which the incidence was found to be 80% and 100%, respectively. It was concluded that platinum-based chemotherapy had played a vital role in the treatment of advanced NSCLC but at an expense of serious and severe AEs which led to discontinuation or withdrawal of treatment, leading to relapse and recurrence of lung cancer.10,11
Zhong and colleagues conducted a phase 2 randomized clinical trial comparing mutant EGFR-TK inhibitors with platinum-based chemotherapy. They concluded that in patients receiving platinum-based chemotherapy, incidence of rash, vomiting, anorexia, neutropenia, and nausea were 29.4%, 47%, 41.2%, 55.8%, and 32.4% compared with 45.8%, 11%, 21.3%, 6.1%, and 16.5%, respectively, reported in patients receiving mutant EGFR-TK inhibitors for their advanced NSCLC.12
Another study was conducted in 2019 by Noronha and colleagues to determine the impact of platinum-based chemotherapy combined with gefitinib on patients with advanced NSCLC.13 They concluded that 70% of the patients receiving combination treatment developed rash, which was significantly higher compared with 45.8% patients receiving the mutant EGFR-TK inhibitors alone in our study. Also, 56% of patients receiving combination therapy developed diarrhea vs 33.6% of patients receiving mutant EGFR-TK inhibitors only. Similarly, 96% of patients in the combination therapy group developed some degree of anemia compared with only 8.7% patients in the mutant EGFR-TK inhibitors group included in our study. In the same way, neutropenia was observed in 55% of patients receiving combination therapy vs 6.1% in patients receiving mutant EGFR-TK inhibitors solely. They concluded that mutant EGFR-TK inhibitors when combined with platinum-based chemotherapy increase the incidence of AEs of chemotherapy by many folds.13,14
Kato and colleagues conducted a study to determine the impact on AEs when erlotinib was combined with anti–vascular endothelial growth factor (VEGF) inhibitors like bevacizumab, they stated that 98.7% of patient in combination therapy developed rash, the incidence of which was only 45.8% in patients receiving mutant EGFR-TK inhibitors as was observed in our study. Similar trends were noticed with other AEs, including diarrhea, fatigue, nausea, and elevated liver enzymes.15
With the latest advancements in the management of advanced NSCLC, nivolumab, a programmed death ligand 1 (PD-L1) inhibitor, was developed and either used as monotherapy in patients with PD-L1 expression or was combined with platinum-based chemotherapy regardless of PD-L1 expression.16,17 Patients expressing lower PD-L1 levels were not omitted from receiving nivolumab as no significant difference was noted in progression-free span and overall survival in patients receiving nivolumab irrespective of PD-L1 levels.15 Rash developed in 17% of patients after receiving nivolumab vs 45.8% patients being observed in our study. A similar trend was observed with diarrhea as only 17% of the population receiving nivolumab developed diarrhea compared with 33.6% of the population receiving mutant EGFR-TK inhibitors in our study. Likewise, only 9.9% of the patients receiving nivolumab developed nausea as an AE compared with 16.5% being observed in mutant EGFR-TK inhibitors in our study. Also, fatigue was observed in 14.4% of the population receiving nivolumab vs 17% observed in patients receiving mutant EGFR-TK inhibitors as was noticed in our study.7,8
Rizvi and colleagues conducted a study on the role of nivolumab when combined with platinum-based chemotherapy in patients with advanced NSCLC and reported that 40% of patients included in the study developed rash compared with 45.8% reported in mutant EGFR-TK inhibitors in our study. Similarly, only 13% of patients in the nivolumab group developed diarrhea vs 33.6% cases reported in the mutant EGFR-TK inhibitors group included in our study. Also, 7% of patients in the nivolumab group developed elevated ALT levels vs 27.9% of patients receiving mutant EGFR-TK inhibitors included in our study, concluding that addition of immune checkpoint inhibitors like nivolumab to platinum-based chemotherapy does not increase the frequency of AEs.18
Conclusions
Our study focused on the safety profile of mutant EGFR-TK inhibitors vs platinum-based chemotherapy in the treatment of advanced NSCLC. Mutant EGFR-TK inhibitors are safer than platinum-based chemotherapy when compared for nausea, leucopenia, fatigue, neutropenia, anorexia, anemia, cough, vomiting, and fever. On the other end, mutant EGFR-TK inhibitors cause slightly higher AEs, including rash, diarrhea, elevated AST and ALT levels, and stomatitis. However, considering that the development of mutant EGFR-TK inhibitors laid a foundation of targeted therapy, we recommend continuing using mutant EGFR-TK inhibitors in patients with advanced NSCLC especially in patients having mutant EGFR receptors. AEs caused by mutant EGFR-TK inhibitors are significant but are usually tolerable and can be avoided by reducing the dosage of it with each cycle or by skipping or delaying the dose until the patient is symptomatic.
1. Rahib L, Smith BD, Aizenberg R, Rosenzweig AB, Fleshman JM, Matrisian LM. Projecting cancer incidence and deaths to 2030: the unexpected burden of thyroid, liver, and pancreas cancers in the United States. Cancer Res. 2014;74(11):2913-2921. doi:10.1158/0008-5472.CAN-14-0155
2. da Cunha Santos G, Shepherd FA, Tsao MS. EGFR mutations and lung cancer. Annu Rev Pathol. 2011;6:49-69. doi:10.1146/annurev-pathol-011110-130206
3. Sgambato A, Casaluce F, Maione P, et al. The role of EGFR tyrosine kinase inhibitors in the first-line treatment of advanced non small cell lung cancer patients harboring EGFR mutation. Curr Med Chem. 2012;19(20):3337-3352. doi:10.2174/092986712801215973
4. Rossi A, Di Maio M. Platinum-based chemotherapy in advanced non–small-cell lung cancer: optimal number of treatment cycles. Expert Rev Anticancer Ther. 2016;16(6):653-660. doi:10.1586/14737140.2016.1170596
5. Rosell R, Carcereny E, Gervais R, et al. Erlotinib versus standard chemotherapy as first-line treatment for European patients with advanced EGFR mutation-positive non–small-cell lung cancer (EURTAC): a multicentre, open-label, randomised phase 3 trial. Lancet Oncol. 2012;13(3):239-246. doi:10.1016/S1470-2045(11)70393-X
6. Zhou C, Wu YL, Chen G, et al. Erlotinib versus chemotherapy as first-line treatment for patients with advanced EGFR mutation-positive non–small-cell lung cancer (OPTIMAL, CTONG-0802): a multicentre, open-label, randomised, phase 3 study. Lancet Oncol. 2011;12(8):735-742. doi:10.1016/S1470-2045(11)70184-X
7. Soria JC, Ohe Y, Vansteenkiste J, et al. Osimertinib in untreated EGFR-mutated advanced non–small-cell lung cancer. N Engl J Med. 2018;378(2):113-125. doi:10.1056/NEJMoa1713137
8. Mok TS, Cheng Y, Zhou X, et al. Improvement in overall survival in a randomized study that compared dacomitinib with gefitinib in patients with advanced non–small-cell lung cancer and EGFR-activating mutations. J Clin Oncol. 2018;36(22):2244-2250. doi:10.1200/JCO.2018.78.7994
9. Mok TS, Wu YL, Thongprasert S, et al. Gefitinib or carboplatin-paclitaxel in pulmonary adenocarcinoma. N Engl J Med. 2009;361(10):947-957. doi:10.1056/NEJMoa0810699
10. Pless M, Stupp R, Ris HB, et al. Induction chemoradiation in stage IIIA/N2 non–small-cell lung cancer: a phase 3 randomised trial. Lancet. 2015;386(9998):1049-1056. doi:10.1016/S0140-6736(15)60294-X
11. Albain KS, Rusch VW, Crowley JJ, et al. Concurrent cisplatin/etoposide plus chest radiotherapy followed by surgery for stages IIIA (N2) and IIIB non–small-cell lung cancer: mature results of Southwest Oncology Group phase II study 8805. J Clin Oncol. 1995;13(8):1880-1892. doi:10.1200/JCO.1995.13.8.1880
12. Zhong WZ, Chen KN, Chen C, et al. Erlotinib versus gemcitabine plus cisplatin as neoadjuvant treatment of Stage IIIA-N2 EGFR-mutant non–small-cell lung cancer (EMERGING-CTONG 1103): a randomized phase II study. J Clin Oncol. 2019;37(25):2235-2245. doi:10.1200/JCO.19.00075
13. Noronha V, Patil VM, Joshi A, et al. Gefitinib versus gefitinib plus pemetrexed and carboplatin chemotherapy in EGFR-mutated lung cancer. J Clin Oncol. 2020;38(2):124-136. doi:10.1200/JCO.19.01154
14. Noronha V, Prabhash K, Thavamani A, et al. EGFR mutations in Indian lung cancer patients: clinical correlation and outcome to EGFR targeted therapy. PLoS One. 2013;8(4):e61561. Published 2013 Apr 19. doi:10.1371/journal.pone.0061561
15. Kato T, Seto T, Nishio M, et al. Erlotinib plus bevacizumab phase ll study in patients with advanced non–small-cell lung cancer (JO25567): updated safety results. Drug Saf. 2018;41(2):229-237. doi:10.1007/s40264-017-0596-0
16. Hellmann MD, Paz-Ares L, Bernabe Caro R, et al. Nivolumab plus ipilimumab in advanced non–small-cell lung cancer. N Engl J Med. 2019;381(21):2020-2031. doi:10.1056/NEJMoa1910231
17. Hellmann MD, Ciuleanu TE, Pluzanski A, et al. Nivolumab plus ipilimumab in lung cancer with a high tumor mutational burden. N Engl J Med. 2018;378(22):2093-2104. doi:10.1056/NEJMoa1801946
18. Rizvi NA, Hellmann MD, Brahmer JR, et al. Nivolumab in combination with platinum-based doublet chemotherapy for first-line treatment of advanced non–small-cell lung cancer. J Clin Oncol. 2016;34(25):2969-2979. doi:10.1200/JCO.2016.66.9861
19. Zhong WZ, Wang Q, Mao WM, et al. Gefitinib versus vinorelbine plus cisplatin as adjuvant treatment for stage II-IIIA (N1-N2) EGFR-mutant NSCLC: final overall survival analysis of CTONG1104 Phase III Trial. J Clin Oncol. 2021;39(7):713-722. doi:10.1200/JCO.20.01820
20. Yang JC, Sequist LV, Geater SL, et al. Clinical activity of afatinib in patients with advanced non-small-cell lung cancer harbouring uncommon EGFR mutations: a combined post-hoc analysis of LUX-Lung 2, LUX-Lung 3, and LUX-Lung 6. Lancet Oncol. 2015;16(7):830-838. doi:10.1016/S1470-2045(15)00026-1
21. Shi YK, Wang L, Han BH, et al. First-line icotinib versus cisplatin/pemetrexed plus pemetrexed maintenance therapy for patients with advanced EGFR mutation-positive lung adenocarcinoma (CONVINCE): a phase 3, open-label, randomized study. Ann Oncol. 2017;28(10):2443-2450. doi:10.1093/annonc/mdx359
22. Soria JC, Wu YL, Nakagawa K, et al. Gefitinib plus chemotherapy versus placebo plus chemotherapy in EGFR-mutation-positive non-small-cell lung cancer after progression on first-line gefitinib (IMPRESS): a phase 3 randomized trial. Lancet Oncol. 2015;16(8):990-998 doi:10.1016/S1470-2045(15)00121-7
23. Goss GD, O’Callaghan C, Lorimer I, et al. Gefitinib versus placebo in completely resected non-small-cell lung cancer: results of the NCIC CTG BR19 study. J Clin Oncol. 2013;31(27):3320-3326. doi:10.1200/JCO.2013.51.1816
24. Sun JM, Lee KH, Kim SW, et al. Gefitinib versus pemetrexed as second-line treatment in patients with non-small cell lung cancer previously treated with platinum-based chemotherapy (KCSG-LU08-01): an open-label, phase 3 trial. Cancer. 2012;118(24):6234-6242. doi:10.1200/JCO.2013.51.1816
25. Mitsudomi T, Morita S, Yatabe Y, et al. Gefitinib versus cisplatin plus docetaxel in patients with non-small-cell lung cancer harbouring mutations of the epidermal growth factor receptor (WJTOG3405): an open label, randomized phase 3 trial. Lancet Oncol. 2010;11(2):121-128. doi:10.1016/S1470-2045(09)70364-X
26. Lee DH, Park K, Kim JH, Lee JS, et al. Randomized phase III trial of gefitinib versus docetaxel in non-small cell lung cancer patients who have previously received platinum-based chemotherapy. Clin Cancer Res. 2010;16(4):1307-1314. doi:10.1158/1078-0432.CCR-09-1903
27. Kim ES, Hirsh V, Mok T, et al. Gefitinib versus docetaxel in previously treated non-small-cell lung cancer (INTEREST): a randomized phase III trial. Lancet. 2008;22;372(9652):1809-1818. doi:10.1016/S0140-6736(08)61758-4
Lung cancer has been the leading cause of cancer-related mortality for decades. It is also predicted to remain as the leading cause of cancer-related mortality through 2030.1 Platinum-based chemotherapy, including carboplatin and paclitaxel, was introduced 3 decades ago and revolutionized the management of advanced non–small cell lung cancer (NSCLC). A more recent advancement has been mutant epidermal growth factor receptor–tyrosine kinase (EGFR-TK) inhibitors.1 EGFR is a transmembrane protein that functions by transducing essential growth factor signaling from the extracellular milieu to the cell. As 60% of the advanced NSCLC expresses this receptor, blocking the mutant EGFR receptor was a groundbreaking development in the management of advanced NSCLC.2 Development of mutant EGFR-TK inhibitors has revolutionized the management of advanced NSCLC. This study was conducted to determine the safety profile of mutant EGFR-TK inhibitors in the management of advanced NSCLC.
Methods
This meta-analysis was conducted according to Cochrane Collaboration guidelines and reported as per Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines. The findings are summarized in the PRISMA flow diagram (Figure 1). Two authors (MZ and MM) performed a systematic literature search using databases such as MEDLINE (via PubMed), Embase, and Cochrane Library using the medical search terms and their respective entry words with the following search strategy: safety, “mutant EGFR-TK inhibitors,” advanced, “non–small cell,” “lung cancer,” “adverse effect,” and literature. Additionally, unpublished trials were identified from clinicaltrials.gov, and references of all pertinent articles were also scrutinized to ensure the inclusion of all relevant studies. The search was completed on June 1, 2021, and we only included studies available in English. Two authors (MM and MZ) independently screened the search results in a 2-step process based on predetermined inclusion/exclusion criteria. First, 890 articles were evaluated for relevance on title and abstract level, followed by full-text screening of the final list of 140 articles. Any disagreements were resolved by discussion or third-party review, and a total of 9 articles were included in the study.
The following eligibility criteria were used: original articles reporting adverse effects (AEs) of mutant EGFR-TK inhibitors in patients with advanced NSCLC compared with control groups receiving platinum-based chemotherapy. All the patients included in the study had an EGFR mutation but randomly assigned to either treatment or control group. All articles with subjective data on mutant EGFR-TK inhibitors AEs in patients with advanced NSCLC compared with control groups receiving platinum-based chemotherapy were included in the analysis. Only 9 articles qualified the aforementioned selection criteria for eligibility. All qualifying studies were nationwide inpatient or pooled clinical trials data. The reasons for exclusion of the other 71 articles were irrelevant (n = 31), duplicate (n = 13), reviews (n = 14), and poor data reporting (n = 12). Out of the 9 included studies, 9 studies showed correlation of AEs, including rash, diarrhea, nausea, and fatigue. Seven studies showed correlation of AEs including neutropenia, anorexia, and vomiting. Six studies showed correlation of anemia, cough, and stomatitis. Five studies showed correlation of elevated aspartate aminotransferase (AST), alanine aminotransferase (ALT), and leucopenia. Four studies showed correlation of fever between mutant EGFR-TK inhibitors and platinum-based chemotherapy.
The primary endpoints were reported AEs including rash, diarrhea, elevated ALT, elevated AST, stomatitis, nausea, leucopenia, fatigue, neutropenia, anorexia, anemia, cough, vomiting, and fever, respectively. Data on baseline characteristics and clinical outcomes were then extracted, and summary tables were created. Summary estimates of the clinical endpoints were then calculated with risk ratio (RR) and 95% confidence intervals (CIs) using the random-effects model. Heterogeneity between studies was examined with the Cochran Q I2 statistic which can be defined as low (25% to 50%), moderate (50% to 75%), or high (> 75%). Statistical analysis was performed using Comprehensive Meta-Analysis Software CMA Version 3.0.
Results
A total of 9 studies including 3415 patients (1775 in EGFR-TK inhibitor treatment group while 1640 patients in platinum-based chemotherapy control group) were included in the study. All 9 studies were phase III randomized control clinical trials conducted to compare the safety profile of mutant EGFR-TK inhibitors in patients with advanced NSCLC. Mean age was 61 years in both treatment and control groups. Further details on study and participant characteristics and safety profile including AEs are summarized in Tables 1 and 2. No evidence of publication bias was found.
Rash developed in 45.8% of patients in the treatment group receiving mutant EGFR-TK inhibitors vs only 5.6% of patients in the control group receiving platinum-based chemotherapy. Overall RR of 7.38 with the 95% CI noted, which was statistically significant, confirming higher rash event rates in patients receiving EGFR-TK inhibitors for their advanced NSCLC (Figure 2).
Diarrhea occurred in 33.6% of patients in the mutant EGFR-TK inhibitors treatment group vs 13.5% of patients in the control group receiving platinum-based chemotherapy. Overall RR of 2.63 and 95% CI was noted, which was statistically significant, confirming higher diarrheal rates in patients receiving EGFR-TK inhibitors for their advanced NSCLC (Figure 3).
Elevated ALT levels developed in 27.9% of patients in the treatment group receiving mutant EGFR-TK inhibitors compared with 15.1% of patients in the control group receiving platinum-based chemotherapy. Overall RR of 1.37 and 95% CI was noted, which was statistically significant, confirming higher ALT levels in patients receiving EGFR-TK inhibitors for their advanced NSCLC (Figure 4).
Elevated AST levels occurred in 40.7% of patients in the mutant EGFR-TK inhibitors treatment group vs 12.8% of patients in the control group receiving platinum-based chemotherapy. Overall RR of 1.77 and 95% CI was noted, which was statistically significant, confirming elevated AST levels in patients receiving EGFR-TK inhibitors for their advanced NSCLC (Figure 5).
Stomatitis developed in 17.2% of patients in the treatment group receiving mutant EGFR-TK inhibitors compared with 7.9% of patients in the control group receiving platinum-based chemotherapy. Overall RR of 1.53 and 95% CI was noted, which was statistically significant, confirming higher stomatitis event rates in patients receiving EGFR-TK inhibitors for their advanced NSCLC (Figure 6).
Nausea occurred in 16.5% of patients in the mutant EGFR-TK inhibitors group vs 42.5% of patients in the control group receiving platinum-based chemotherapy. Overall RR of 0.37 and 95% CI was noted, which was statistically significant, confirming higher nausea rates in patients receiving platinum-based chemotherapy compared with treatment group for their advanced NSCLC (Figure 7).
Leucopenia developed in 9.7% of patients in the mutant EGFR-TK inhibitors group compared with 51.3% of patients in the control group receiving platinum-based chemotherapy. Overall RR of 0.18 and 95% CI was noted, which was statistically significant, confirming higher leucopenia incidence in patients receiving platinum-based chemotherapy compared with treatment group for their advanced NSCLC (Figure 8).
Fatigue was reported in 17% of patients in the mutant EGFR-TK inhibitors group compared with 29.5% of patients in the control group receiving platinum-based chemotherapy. Overall RR of 0.59 and 95% CI was noted, which was statistically significant, confirming higher fatigue rates in patients receiving platinum-based chemotherapy compared with treatment group for their advanced NSCLC (Figure 9).
Neutropenia developed in 6.1% of patients in the mutant EGFR-TK inhibitors group vs 48.2% of patients in the control group receiving platinum-based chemotherapy. Overall RR of 0.11 and 95% CI was noted, which was statistically significant, confirming higher neutropenia rates in patients receiving platinum-based chemotherapy compared with the treatment group for their advanced NSCLC (Figure 10).
Anorexia developed in 21.3% of patients in the mutant EGFR-TK inhibitors group vs 31.4% of patients in the control group receiving platinum-based chemotherapy. Overall RR of 0.44 and 95% CI was noted, which was statistically significant, confirming higher anorexia rates in patients receiving platinum-based chemotherapy compared with the treatment group for their advanced NSCLC (Figure 11).
Anemia occurred in 8.7% of patients in the mutant EGFR-TK inhibitors group compared with 32.1% of patients in the control group receiving platinum-based chemotherapy. Overall RR of 0.24 and 95% CI was noted, which was statistically significant, confirming higher anorexia rates in patients receiving platinum-based chemotherapy compared with treatment for their advanced NSCLC (Figure 12).
Cough was reported in 17.8% of patients in the mutant EGFR-TK inhibitors group compared with 18.9% of patients in the control group receiving platinum-based chemotherapy. Overall RR of 0.99 and 95% CI was noted, which was statistically significant, confirming slightly higher cough rates in patients receiving platinum-based chemotherapy compared with treatment for their advanced NSCLC (Figure 13).
Vomiting developed in 11% of patients in the mutant EGFR-TK inhibitors group vs 30.1% of patients in the control group receiving platinum-based chemotherapy. Overall RR of 0.35 and 95% CI was noted, which was statistically significant, confirming higher vomiting rates in patients receiving platinum-based chemotherapy compared with the treatment group for their advanced NSCLC (Figure 14).
Fever occurred in 5.6% of patients in the mutant EGFR-TK inhibitors group compared with 30.1% of patients in the control group receiving platinum-based chemotherapy. Overall RR of 0.41 and 95% CI was noted, which was statistically significant, confirming higher fever rates in patients receiving platinum-based chemotherapy compared with the treatment group for their advanced NSCLC (Figure 15).
Discussion
Despite the advancement in the treatment of metastatic NSCLC, lung cancer stays as most common cause of cancer-related death in North America and European countries, as patients usually have an advanced disease at the time of diagnosis.3 In the past, platinum-based chemotherapy remained the standard of care for most of the patients affected with advanced NSCLC, but the higher recurrence rate and increase in frequency and intensity of AEs with platinum-based chemotherapy led to the development of targeted therapy for NSCLC, one of which includes
Smoking is the most common reversible risk factor associated with lung cancer. The EURTAC trial was the first perspective study in this regard, which compared safety and efficacy of mutant EGFR-TK inhibitors with platinum-based chemotherapy. Results analyzed in this study were in favor of mutant EGFR-TK inhibitors except in the group of former smokers.5 On the contrary, the OPTIMAL trial showed results in favor of mutant EGFR-TK inhibitors both in active and former smokers; this trial also confirmed the efficacy of mutant EGFR-TK inhibitors in European and Asian populations, confirming the rationale for routine testing of EGFR mutation in all the patients being diagnosed with advanced NSCLC.6 Similarly, osimertinib is one of the most recent mutant EGFR-TK inhibitors developed for the treatment of advanced NSCLC in patients with EGFR-positive receptors.
According to the FLAURA trial, patients receiving osimertinib showed significantly longer progression-free survival compared with platinum-based chemotherapy and early mutant EGFR-TK inhibitors. Median progression-free survival was noted to be 18.9 months, which showed 54% lower risk of disease progression in the treatment group receiving osimertinib.7 The ARCHER study emphasized a significant improvement in overall survival as well as progression-free survival among a patient population receiving dacomitinib compared with platinum-based chemotherapy.8,9
Being a potent targeted therapy, mutant EGFR-TK inhibitors do come with some AEs including diarrhea, which was seen in 33.6% of the patients receiving mutant EGFR-TK inhibitors in our study vs 53% in the chemotherapy group, as was observed in the study conducted by Pless and colleagues.10 Similarly, only 16.5% of patients receiving mutant EGFR-TK inhibitors developed nausea compared with 66% being observed in patients receiving chemotherapy. Correspondingly, only a small fraction of patients (9.7%) receiving mutant EGFR-TK inhibitors developed leucopenia, which was 10 times less reported in mutant EGFR-TK inhibitors compared with patients receiving chemotherapy having a percentage of 100%. A similar trend was reported for neutropenia and anemia in mutant EGFR-TK inhibitors with an incidence of 6.1% and 8.7%, compared with the platinum-based chemotherapy group in which the incidence was found to be 80% and 100%, respectively. It was concluded that platinum-based chemotherapy had played a vital role in the treatment of advanced NSCLC but at an expense of serious and severe AEs which led to discontinuation or withdrawal of treatment, leading to relapse and recurrence of lung cancer.10,11
Zhong and colleagues conducted a phase 2 randomized clinical trial comparing mutant EGFR-TK inhibitors with platinum-based chemotherapy. They concluded that in patients receiving platinum-based chemotherapy, incidence of rash, vomiting, anorexia, neutropenia, and nausea were 29.4%, 47%, 41.2%, 55.8%, and 32.4% compared with 45.8%, 11%, 21.3%, 6.1%, and 16.5%, respectively, reported in patients receiving mutant EGFR-TK inhibitors for their advanced NSCLC.12
Another study was conducted in 2019 by Noronha and colleagues to determine the impact of platinum-based chemotherapy combined with gefitinib on patients with advanced NSCLC.13 They concluded that 70% of the patients receiving combination treatment developed rash, which was significantly higher compared with 45.8% patients receiving the mutant EGFR-TK inhibitors alone in our study. Also, 56% of patients receiving combination therapy developed diarrhea vs 33.6% of patients receiving mutant EGFR-TK inhibitors only. Similarly, 96% of patients in the combination therapy group developed some degree of anemia compared with only 8.7% patients in the mutant EGFR-TK inhibitors group included in our study. In the same way, neutropenia was observed in 55% of patients receiving combination therapy vs 6.1% in patients receiving mutant EGFR-TK inhibitors solely. They concluded that mutant EGFR-TK inhibitors when combined with platinum-based chemotherapy increase the incidence of AEs of chemotherapy by many folds.13,14
Kato and colleagues conducted a study to determine the impact on AEs when erlotinib was combined with anti–vascular endothelial growth factor (VEGF) inhibitors like bevacizumab, they stated that 98.7% of patient in combination therapy developed rash, the incidence of which was only 45.8% in patients receiving mutant EGFR-TK inhibitors as was observed in our study. Similar trends were noticed with other AEs, including diarrhea, fatigue, nausea, and elevated liver enzymes.15
With the latest advancements in the management of advanced NSCLC, nivolumab, a programmed death ligand 1 (PD-L1) inhibitor, was developed and either used as monotherapy in patients with PD-L1 expression or was combined with platinum-based chemotherapy regardless of PD-L1 expression.16,17 Patients expressing lower PD-L1 levels were not omitted from receiving nivolumab as no significant difference was noted in progression-free span and overall survival in patients receiving nivolumab irrespective of PD-L1 levels.15 Rash developed in 17% of patients after receiving nivolumab vs 45.8% patients being observed in our study. A similar trend was observed with diarrhea as only 17% of the population receiving nivolumab developed diarrhea compared with 33.6% of the population receiving mutant EGFR-TK inhibitors in our study. Likewise, only 9.9% of the patients receiving nivolumab developed nausea as an AE compared with 16.5% being observed in mutant EGFR-TK inhibitors in our study. Also, fatigue was observed in 14.4% of the population receiving nivolumab vs 17% observed in patients receiving mutant EGFR-TK inhibitors as was noticed in our study.7,8
Rizvi and colleagues conducted a study on the role of nivolumab when combined with platinum-based chemotherapy in patients with advanced NSCLC and reported that 40% of patients included in the study developed rash compared with 45.8% reported in mutant EGFR-TK inhibitors in our study. Similarly, only 13% of patients in the nivolumab group developed diarrhea vs 33.6% cases reported in the mutant EGFR-TK inhibitors group included in our study. Also, 7% of patients in the nivolumab group developed elevated ALT levels vs 27.9% of patients receiving mutant EGFR-TK inhibitors included in our study, concluding that addition of immune checkpoint inhibitors like nivolumab to platinum-based chemotherapy does not increase the frequency of AEs.18
Conclusions
Our study focused on the safety profile of mutant EGFR-TK inhibitors vs platinum-based chemotherapy in the treatment of advanced NSCLC. Mutant EGFR-TK inhibitors are safer than platinum-based chemotherapy when compared for nausea, leucopenia, fatigue, neutropenia, anorexia, anemia, cough, vomiting, and fever. On the other end, mutant EGFR-TK inhibitors cause slightly higher AEs, including rash, diarrhea, elevated AST and ALT levels, and stomatitis. However, considering that the development of mutant EGFR-TK inhibitors laid a foundation of targeted therapy, we recommend continuing using mutant EGFR-TK inhibitors in patients with advanced NSCLC especially in patients having mutant EGFR receptors. AEs caused by mutant EGFR-TK inhibitors are significant but are usually tolerable and can be avoided by reducing the dosage of it with each cycle or by skipping or delaying the dose until the patient is symptomatic.
Lung cancer has been the leading cause of cancer-related mortality for decades. It is also predicted to remain as the leading cause of cancer-related mortality through 2030.1 Platinum-based chemotherapy, including carboplatin and paclitaxel, was introduced 3 decades ago and revolutionized the management of advanced non–small cell lung cancer (NSCLC). A more recent advancement has been mutant epidermal growth factor receptor–tyrosine kinase (EGFR-TK) inhibitors.1 EGFR is a transmembrane protein that functions by transducing essential growth factor signaling from the extracellular milieu to the cell. As 60% of the advanced NSCLC expresses this receptor, blocking the mutant EGFR receptor was a groundbreaking development in the management of advanced NSCLC.2 Development of mutant EGFR-TK inhibitors has revolutionized the management of advanced NSCLC. This study was conducted to determine the safety profile of mutant EGFR-TK inhibitors in the management of advanced NSCLC.
Methods
This meta-analysis was conducted according to Cochrane Collaboration guidelines and reported as per Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines. The findings are summarized in the PRISMA flow diagram (Figure 1). Two authors (MZ and MM) performed a systematic literature search using databases such as MEDLINE (via PubMed), Embase, and Cochrane Library using the medical search terms and their respective entry words with the following search strategy: safety, “mutant EGFR-TK inhibitors,” advanced, “non–small cell,” “lung cancer,” “adverse effect,” and literature. Additionally, unpublished trials were identified from clinicaltrials.gov, and references of all pertinent articles were also scrutinized to ensure the inclusion of all relevant studies. The search was completed on June 1, 2021, and we only included studies available in English. Two authors (MM and MZ) independently screened the search results in a 2-step process based on predetermined inclusion/exclusion criteria. First, 890 articles were evaluated for relevance on title and abstract level, followed by full-text screening of the final list of 140 articles. Any disagreements were resolved by discussion or third-party review, and a total of 9 articles were included in the study.
The following eligibility criteria were used: original articles reporting adverse effects (AEs) of mutant EGFR-TK inhibitors in patients with advanced NSCLC compared with control groups receiving platinum-based chemotherapy. All the patients included in the study had an EGFR mutation but randomly assigned to either treatment or control group. All articles with subjective data on mutant EGFR-TK inhibitors AEs in patients with advanced NSCLC compared with control groups receiving platinum-based chemotherapy were included in the analysis. Only 9 articles qualified the aforementioned selection criteria for eligibility. All qualifying studies were nationwide inpatient or pooled clinical trials data. The reasons for exclusion of the other 71 articles were irrelevant (n = 31), duplicate (n = 13), reviews (n = 14), and poor data reporting (n = 12). Out of the 9 included studies, 9 studies showed correlation of AEs, including rash, diarrhea, nausea, and fatigue. Seven studies showed correlation of AEs including neutropenia, anorexia, and vomiting. Six studies showed correlation of anemia, cough, and stomatitis. Five studies showed correlation of elevated aspartate aminotransferase (AST), alanine aminotransferase (ALT), and leucopenia. Four studies showed correlation of fever between mutant EGFR-TK inhibitors and platinum-based chemotherapy.
The primary endpoints were reported AEs including rash, diarrhea, elevated ALT, elevated AST, stomatitis, nausea, leucopenia, fatigue, neutropenia, anorexia, anemia, cough, vomiting, and fever, respectively. Data on baseline characteristics and clinical outcomes were then extracted, and summary tables were created. Summary estimates of the clinical endpoints were then calculated with risk ratio (RR) and 95% confidence intervals (CIs) using the random-effects model. Heterogeneity between studies was examined with the Cochran Q I2 statistic which can be defined as low (25% to 50%), moderate (50% to 75%), or high (> 75%). Statistical analysis was performed using Comprehensive Meta-Analysis Software CMA Version 3.0.
Results
A total of 9 studies including 3415 patients (1775 in EGFR-TK inhibitor treatment group while 1640 patients in platinum-based chemotherapy control group) were included in the study. All 9 studies were phase III randomized control clinical trials conducted to compare the safety profile of mutant EGFR-TK inhibitors in patients with advanced NSCLC. Mean age was 61 years in both treatment and control groups. Further details on study and participant characteristics and safety profile including AEs are summarized in Tables 1 and 2. No evidence of publication bias was found.
Rash developed in 45.8% of patients in the treatment group receiving mutant EGFR-TK inhibitors vs only 5.6% of patients in the control group receiving platinum-based chemotherapy. Overall RR of 7.38 with the 95% CI noted, which was statistically significant, confirming higher rash event rates in patients receiving EGFR-TK inhibitors for their advanced NSCLC (Figure 2).
Diarrhea occurred in 33.6% of patients in the mutant EGFR-TK inhibitors treatment group vs 13.5% of patients in the control group receiving platinum-based chemotherapy. Overall RR of 2.63 and 95% CI was noted, which was statistically significant, confirming higher diarrheal rates in patients receiving EGFR-TK inhibitors for their advanced NSCLC (Figure 3).
Elevated ALT levels developed in 27.9% of patients in the treatment group receiving mutant EGFR-TK inhibitors compared with 15.1% of patients in the control group receiving platinum-based chemotherapy. Overall RR of 1.37 and 95% CI was noted, which was statistically significant, confirming higher ALT levels in patients receiving EGFR-TK inhibitors for their advanced NSCLC (Figure 4).
Elevated AST levels occurred in 40.7% of patients in the mutant EGFR-TK inhibitors treatment group vs 12.8% of patients in the control group receiving platinum-based chemotherapy. Overall RR of 1.77 and 95% CI was noted, which was statistically significant, confirming elevated AST levels in patients receiving EGFR-TK inhibitors for their advanced NSCLC (Figure 5).
Stomatitis developed in 17.2% of patients in the treatment group receiving mutant EGFR-TK inhibitors compared with 7.9% of patients in the control group receiving platinum-based chemotherapy. Overall RR of 1.53 and 95% CI was noted, which was statistically significant, confirming higher stomatitis event rates in patients receiving EGFR-TK inhibitors for their advanced NSCLC (Figure 6).
Nausea occurred in 16.5% of patients in the mutant EGFR-TK inhibitors group vs 42.5% of patients in the control group receiving platinum-based chemotherapy. Overall RR of 0.37 and 95% CI was noted, which was statistically significant, confirming higher nausea rates in patients receiving platinum-based chemotherapy compared with treatment group for their advanced NSCLC (Figure 7).
Leucopenia developed in 9.7% of patients in the mutant EGFR-TK inhibitors group compared with 51.3% of patients in the control group receiving platinum-based chemotherapy. Overall RR of 0.18 and 95% CI was noted, which was statistically significant, confirming higher leucopenia incidence in patients receiving platinum-based chemotherapy compared with treatment group for their advanced NSCLC (Figure 8).
Fatigue was reported in 17% of patients in the mutant EGFR-TK inhibitors group compared with 29.5% of patients in the control group receiving platinum-based chemotherapy. Overall RR of 0.59 and 95% CI was noted, which was statistically significant, confirming higher fatigue rates in patients receiving platinum-based chemotherapy compared with treatment group for their advanced NSCLC (Figure 9).
Neutropenia developed in 6.1% of patients in the mutant EGFR-TK inhibitors group vs 48.2% of patients in the control group receiving platinum-based chemotherapy. Overall RR of 0.11 and 95% CI was noted, which was statistically significant, confirming higher neutropenia rates in patients receiving platinum-based chemotherapy compared with the treatment group for their advanced NSCLC (Figure 10).
Anorexia developed in 21.3% of patients in the mutant EGFR-TK inhibitors group vs 31.4% of patients in the control group receiving platinum-based chemotherapy. Overall RR of 0.44 and 95% CI was noted, which was statistically significant, confirming higher anorexia rates in patients receiving platinum-based chemotherapy compared with the treatment group for their advanced NSCLC (Figure 11).
Anemia occurred in 8.7% of patients in the mutant EGFR-TK inhibitors group compared with 32.1% of patients in the control group receiving platinum-based chemotherapy. Overall RR of 0.24 and 95% CI was noted, which was statistically significant, confirming higher anorexia rates in patients receiving platinum-based chemotherapy compared with treatment for their advanced NSCLC (Figure 12).
Cough was reported in 17.8% of patients in the mutant EGFR-TK inhibitors group compared with 18.9% of patients in the control group receiving platinum-based chemotherapy. Overall RR of 0.99 and 95% CI was noted, which was statistically significant, confirming slightly higher cough rates in patients receiving platinum-based chemotherapy compared with treatment for their advanced NSCLC (Figure 13).
Vomiting developed in 11% of patients in the mutant EGFR-TK inhibitors group vs 30.1% of patients in the control group receiving platinum-based chemotherapy. Overall RR of 0.35 and 95% CI was noted, which was statistically significant, confirming higher vomiting rates in patients receiving platinum-based chemotherapy compared with the treatment group for their advanced NSCLC (Figure 14).
Fever occurred in 5.6% of patients in the mutant EGFR-TK inhibitors group compared with 30.1% of patients in the control group receiving platinum-based chemotherapy. Overall RR of 0.41 and 95% CI was noted, which was statistically significant, confirming higher fever rates in patients receiving platinum-based chemotherapy compared with the treatment group for their advanced NSCLC (Figure 15).
Discussion
Despite the advancement in the treatment of metastatic NSCLC, lung cancer stays as most common cause of cancer-related death in North America and European countries, as patients usually have an advanced disease at the time of diagnosis.3 In the past, platinum-based chemotherapy remained the standard of care for most of the patients affected with advanced NSCLC, but the higher recurrence rate and increase in frequency and intensity of AEs with platinum-based chemotherapy led to the development of targeted therapy for NSCLC, one of which includes
Smoking is the most common reversible risk factor associated with lung cancer. The EURTAC trial was the first perspective study in this regard, which compared safety and efficacy of mutant EGFR-TK inhibitors with platinum-based chemotherapy. Results analyzed in this study were in favor of mutant EGFR-TK inhibitors except in the group of former smokers.5 On the contrary, the OPTIMAL trial showed results in favor of mutant EGFR-TK inhibitors both in active and former smokers; this trial also confirmed the efficacy of mutant EGFR-TK inhibitors in European and Asian populations, confirming the rationale for routine testing of EGFR mutation in all the patients being diagnosed with advanced NSCLC.6 Similarly, osimertinib is one of the most recent mutant EGFR-TK inhibitors developed for the treatment of advanced NSCLC in patients with EGFR-positive receptors.
According to the FLAURA trial, patients receiving osimertinib showed significantly longer progression-free survival compared with platinum-based chemotherapy and early mutant EGFR-TK inhibitors. Median progression-free survival was noted to be 18.9 months, which showed 54% lower risk of disease progression in the treatment group receiving osimertinib.7 The ARCHER study emphasized a significant improvement in overall survival as well as progression-free survival among a patient population receiving dacomitinib compared with platinum-based chemotherapy.8,9
Being a potent targeted therapy, mutant EGFR-TK inhibitors do come with some AEs including diarrhea, which was seen in 33.6% of the patients receiving mutant EGFR-TK inhibitors in our study vs 53% in the chemotherapy group, as was observed in the study conducted by Pless and colleagues.10 Similarly, only 16.5% of patients receiving mutant EGFR-TK inhibitors developed nausea compared with 66% being observed in patients receiving chemotherapy. Correspondingly, only a small fraction of patients (9.7%) receiving mutant EGFR-TK inhibitors developed leucopenia, which was 10 times less reported in mutant EGFR-TK inhibitors compared with patients receiving chemotherapy having a percentage of 100%. A similar trend was reported for neutropenia and anemia in mutant EGFR-TK inhibitors with an incidence of 6.1% and 8.7%, compared with the platinum-based chemotherapy group in which the incidence was found to be 80% and 100%, respectively. It was concluded that platinum-based chemotherapy had played a vital role in the treatment of advanced NSCLC but at an expense of serious and severe AEs which led to discontinuation or withdrawal of treatment, leading to relapse and recurrence of lung cancer.10,11
Zhong and colleagues conducted a phase 2 randomized clinical trial comparing mutant EGFR-TK inhibitors with platinum-based chemotherapy. They concluded that in patients receiving platinum-based chemotherapy, incidence of rash, vomiting, anorexia, neutropenia, and nausea were 29.4%, 47%, 41.2%, 55.8%, and 32.4% compared with 45.8%, 11%, 21.3%, 6.1%, and 16.5%, respectively, reported in patients receiving mutant EGFR-TK inhibitors for their advanced NSCLC.12
Another study was conducted in 2019 by Noronha and colleagues to determine the impact of platinum-based chemotherapy combined with gefitinib on patients with advanced NSCLC.13 They concluded that 70% of the patients receiving combination treatment developed rash, which was significantly higher compared with 45.8% patients receiving the mutant EGFR-TK inhibitors alone in our study. Also, 56% of patients receiving combination therapy developed diarrhea vs 33.6% of patients receiving mutant EGFR-TK inhibitors only. Similarly, 96% of patients in the combination therapy group developed some degree of anemia compared with only 8.7% patients in the mutant EGFR-TK inhibitors group included in our study. In the same way, neutropenia was observed in 55% of patients receiving combination therapy vs 6.1% in patients receiving mutant EGFR-TK inhibitors solely. They concluded that mutant EGFR-TK inhibitors when combined with platinum-based chemotherapy increase the incidence of AEs of chemotherapy by many folds.13,14
Kato and colleagues conducted a study to determine the impact on AEs when erlotinib was combined with anti–vascular endothelial growth factor (VEGF) inhibitors like bevacizumab, they stated that 98.7% of patient in combination therapy developed rash, the incidence of which was only 45.8% in patients receiving mutant EGFR-TK inhibitors as was observed in our study. Similar trends were noticed with other AEs, including diarrhea, fatigue, nausea, and elevated liver enzymes.15
With the latest advancements in the management of advanced NSCLC, nivolumab, a programmed death ligand 1 (PD-L1) inhibitor, was developed and either used as monotherapy in patients with PD-L1 expression or was combined with platinum-based chemotherapy regardless of PD-L1 expression.16,17 Patients expressing lower PD-L1 levels were not omitted from receiving nivolumab as no significant difference was noted in progression-free span and overall survival in patients receiving nivolumab irrespective of PD-L1 levels.15 Rash developed in 17% of patients after receiving nivolumab vs 45.8% patients being observed in our study. A similar trend was observed with diarrhea as only 17% of the population receiving nivolumab developed diarrhea compared with 33.6% of the population receiving mutant EGFR-TK inhibitors in our study. Likewise, only 9.9% of the patients receiving nivolumab developed nausea as an AE compared with 16.5% being observed in mutant EGFR-TK inhibitors in our study. Also, fatigue was observed in 14.4% of the population receiving nivolumab vs 17% observed in patients receiving mutant EGFR-TK inhibitors as was noticed in our study.7,8
Rizvi and colleagues conducted a study on the role of nivolumab when combined with platinum-based chemotherapy in patients with advanced NSCLC and reported that 40% of patients included in the study developed rash compared with 45.8% reported in mutant EGFR-TK inhibitors in our study. Similarly, only 13% of patients in the nivolumab group developed diarrhea vs 33.6% cases reported in the mutant EGFR-TK inhibitors group included in our study. Also, 7% of patients in the nivolumab group developed elevated ALT levels vs 27.9% of patients receiving mutant EGFR-TK inhibitors included in our study, concluding that addition of immune checkpoint inhibitors like nivolumab to platinum-based chemotherapy does not increase the frequency of AEs.18
Conclusions
Our study focused on the safety profile of mutant EGFR-TK inhibitors vs platinum-based chemotherapy in the treatment of advanced NSCLC. Mutant EGFR-TK inhibitors are safer than platinum-based chemotherapy when compared for nausea, leucopenia, fatigue, neutropenia, anorexia, anemia, cough, vomiting, and fever. On the other end, mutant EGFR-TK inhibitors cause slightly higher AEs, including rash, diarrhea, elevated AST and ALT levels, and stomatitis. However, considering that the development of mutant EGFR-TK inhibitors laid a foundation of targeted therapy, we recommend continuing using mutant EGFR-TK inhibitors in patients with advanced NSCLC especially in patients having mutant EGFR receptors. AEs caused by mutant EGFR-TK inhibitors are significant but are usually tolerable and can be avoided by reducing the dosage of it with each cycle or by skipping or delaying the dose until the patient is symptomatic.
1. Rahib L, Smith BD, Aizenberg R, Rosenzweig AB, Fleshman JM, Matrisian LM. Projecting cancer incidence and deaths to 2030: the unexpected burden of thyroid, liver, and pancreas cancers in the United States. Cancer Res. 2014;74(11):2913-2921. doi:10.1158/0008-5472.CAN-14-0155
2. da Cunha Santos G, Shepherd FA, Tsao MS. EGFR mutations and lung cancer. Annu Rev Pathol. 2011;6:49-69. doi:10.1146/annurev-pathol-011110-130206
3. Sgambato A, Casaluce F, Maione P, et al. The role of EGFR tyrosine kinase inhibitors in the first-line treatment of advanced non small cell lung cancer patients harboring EGFR mutation. Curr Med Chem. 2012;19(20):3337-3352. doi:10.2174/092986712801215973
4. Rossi A, Di Maio M. Platinum-based chemotherapy in advanced non–small-cell lung cancer: optimal number of treatment cycles. Expert Rev Anticancer Ther. 2016;16(6):653-660. doi:10.1586/14737140.2016.1170596
5. Rosell R, Carcereny E, Gervais R, et al. Erlotinib versus standard chemotherapy as first-line treatment for European patients with advanced EGFR mutation-positive non–small-cell lung cancer (EURTAC): a multicentre, open-label, randomised phase 3 trial. Lancet Oncol. 2012;13(3):239-246. doi:10.1016/S1470-2045(11)70393-X
6. Zhou C, Wu YL, Chen G, et al. Erlotinib versus chemotherapy as first-line treatment for patients with advanced EGFR mutation-positive non–small-cell lung cancer (OPTIMAL, CTONG-0802): a multicentre, open-label, randomised, phase 3 study. Lancet Oncol. 2011;12(8):735-742. doi:10.1016/S1470-2045(11)70184-X
7. Soria JC, Ohe Y, Vansteenkiste J, et al. Osimertinib in untreated EGFR-mutated advanced non–small-cell lung cancer. N Engl J Med. 2018;378(2):113-125. doi:10.1056/NEJMoa1713137
8. Mok TS, Cheng Y, Zhou X, et al. Improvement in overall survival in a randomized study that compared dacomitinib with gefitinib in patients with advanced non–small-cell lung cancer and EGFR-activating mutations. J Clin Oncol. 2018;36(22):2244-2250. doi:10.1200/JCO.2018.78.7994
9. Mok TS, Wu YL, Thongprasert S, et al. Gefitinib or carboplatin-paclitaxel in pulmonary adenocarcinoma. N Engl J Med. 2009;361(10):947-957. doi:10.1056/NEJMoa0810699
10. Pless M, Stupp R, Ris HB, et al. Induction chemoradiation in stage IIIA/N2 non–small-cell lung cancer: a phase 3 randomised trial. Lancet. 2015;386(9998):1049-1056. doi:10.1016/S0140-6736(15)60294-X
11. Albain KS, Rusch VW, Crowley JJ, et al. Concurrent cisplatin/etoposide plus chest radiotherapy followed by surgery for stages IIIA (N2) and IIIB non–small-cell lung cancer: mature results of Southwest Oncology Group phase II study 8805. J Clin Oncol. 1995;13(8):1880-1892. doi:10.1200/JCO.1995.13.8.1880
12. Zhong WZ, Chen KN, Chen C, et al. Erlotinib versus gemcitabine plus cisplatin as neoadjuvant treatment of Stage IIIA-N2 EGFR-mutant non–small-cell lung cancer (EMERGING-CTONG 1103): a randomized phase II study. J Clin Oncol. 2019;37(25):2235-2245. doi:10.1200/JCO.19.00075
13. Noronha V, Patil VM, Joshi A, et al. Gefitinib versus gefitinib plus pemetrexed and carboplatin chemotherapy in EGFR-mutated lung cancer. J Clin Oncol. 2020;38(2):124-136. doi:10.1200/JCO.19.01154
14. Noronha V, Prabhash K, Thavamani A, et al. EGFR mutations in Indian lung cancer patients: clinical correlation and outcome to EGFR targeted therapy. PLoS One. 2013;8(4):e61561. Published 2013 Apr 19. doi:10.1371/journal.pone.0061561
15. Kato T, Seto T, Nishio M, et al. Erlotinib plus bevacizumab phase ll study in patients with advanced non–small-cell lung cancer (JO25567): updated safety results. Drug Saf. 2018;41(2):229-237. doi:10.1007/s40264-017-0596-0
16. Hellmann MD, Paz-Ares L, Bernabe Caro R, et al. Nivolumab plus ipilimumab in advanced non–small-cell lung cancer. N Engl J Med. 2019;381(21):2020-2031. doi:10.1056/NEJMoa1910231
17. Hellmann MD, Ciuleanu TE, Pluzanski A, et al. Nivolumab plus ipilimumab in lung cancer with a high tumor mutational burden. N Engl J Med. 2018;378(22):2093-2104. doi:10.1056/NEJMoa1801946
18. Rizvi NA, Hellmann MD, Brahmer JR, et al. Nivolumab in combination with platinum-based doublet chemotherapy for first-line treatment of advanced non–small-cell lung cancer. J Clin Oncol. 2016;34(25):2969-2979. doi:10.1200/JCO.2016.66.9861
19. Zhong WZ, Wang Q, Mao WM, et al. Gefitinib versus vinorelbine plus cisplatin as adjuvant treatment for stage II-IIIA (N1-N2) EGFR-mutant NSCLC: final overall survival analysis of CTONG1104 Phase III Trial. J Clin Oncol. 2021;39(7):713-722. doi:10.1200/JCO.20.01820
20. Yang JC, Sequist LV, Geater SL, et al. Clinical activity of afatinib in patients with advanced non-small-cell lung cancer harbouring uncommon EGFR mutations: a combined post-hoc analysis of LUX-Lung 2, LUX-Lung 3, and LUX-Lung 6. Lancet Oncol. 2015;16(7):830-838. doi:10.1016/S1470-2045(15)00026-1
21. Shi YK, Wang L, Han BH, et al. First-line icotinib versus cisplatin/pemetrexed plus pemetrexed maintenance therapy for patients with advanced EGFR mutation-positive lung adenocarcinoma (CONVINCE): a phase 3, open-label, randomized study. Ann Oncol. 2017;28(10):2443-2450. doi:10.1093/annonc/mdx359
22. Soria JC, Wu YL, Nakagawa K, et al. Gefitinib plus chemotherapy versus placebo plus chemotherapy in EGFR-mutation-positive non-small-cell lung cancer after progression on first-line gefitinib (IMPRESS): a phase 3 randomized trial. Lancet Oncol. 2015;16(8):990-998 doi:10.1016/S1470-2045(15)00121-7
23. Goss GD, O’Callaghan C, Lorimer I, et al. Gefitinib versus placebo in completely resected non-small-cell lung cancer: results of the NCIC CTG BR19 study. J Clin Oncol. 2013;31(27):3320-3326. doi:10.1200/JCO.2013.51.1816
24. Sun JM, Lee KH, Kim SW, et al. Gefitinib versus pemetrexed as second-line treatment in patients with non-small cell lung cancer previously treated with platinum-based chemotherapy (KCSG-LU08-01): an open-label, phase 3 trial. Cancer. 2012;118(24):6234-6242. doi:10.1200/JCO.2013.51.1816
25. Mitsudomi T, Morita S, Yatabe Y, et al. Gefitinib versus cisplatin plus docetaxel in patients with non-small-cell lung cancer harbouring mutations of the epidermal growth factor receptor (WJTOG3405): an open label, randomized phase 3 trial. Lancet Oncol. 2010;11(2):121-128. doi:10.1016/S1470-2045(09)70364-X
26. Lee DH, Park K, Kim JH, Lee JS, et al. Randomized phase III trial of gefitinib versus docetaxel in non-small cell lung cancer patients who have previously received platinum-based chemotherapy. Clin Cancer Res. 2010;16(4):1307-1314. doi:10.1158/1078-0432.CCR-09-1903
27. Kim ES, Hirsh V, Mok T, et al. Gefitinib versus docetaxel in previously treated non-small-cell lung cancer (INTEREST): a randomized phase III trial. Lancet. 2008;22;372(9652):1809-1818. doi:10.1016/S0140-6736(08)61758-4
1. Rahib L, Smith BD, Aizenberg R, Rosenzweig AB, Fleshman JM, Matrisian LM. Projecting cancer incidence and deaths to 2030: the unexpected burden of thyroid, liver, and pancreas cancers in the United States. Cancer Res. 2014;74(11):2913-2921. doi:10.1158/0008-5472.CAN-14-0155
2. da Cunha Santos G, Shepherd FA, Tsao MS. EGFR mutations and lung cancer. Annu Rev Pathol. 2011;6:49-69. doi:10.1146/annurev-pathol-011110-130206
3. Sgambato A, Casaluce F, Maione P, et al. The role of EGFR tyrosine kinase inhibitors in the first-line treatment of advanced non small cell lung cancer patients harboring EGFR mutation. Curr Med Chem. 2012;19(20):3337-3352. doi:10.2174/092986712801215973
4. Rossi A, Di Maio M. Platinum-based chemotherapy in advanced non–small-cell lung cancer: optimal number of treatment cycles. Expert Rev Anticancer Ther. 2016;16(6):653-660. doi:10.1586/14737140.2016.1170596
5. Rosell R, Carcereny E, Gervais R, et al. Erlotinib versus standard chemotherapy as first-line treatment for European patients with advanced EGFR mutation-positive non–small-cell lung cancer (EURTAC): a multicentre, open-label, randomised phase 3 trial. Lancet Oncol. 2012;13(3):239-246. doi:10.1016/S1470-2045(11)70393-X
6. Zhou C, Wu YL, Chen G, et al. Erlotinib versus chemotherapy as first-line treatment for patients with advanced EGFR mutation-positive non–small-cell lung cancer (OPTIMAL, CTONG-0802): a multicentre, open-label, randomised, phase 3 study. Lancet Oncol. 2011;12(8):735-742. doi:10.1016/S1470-2045(11)70184-X
7. Soria JC, Ohe Y, Vansteenkiste J, et al. Osimertinib in untreated EGFR-mutated advanced non–small-cell lung cancer. N Engl J Med. 2018;378(2):113-125. doi:10.1056/NEJMoa1713137
8. Mok TS, Cheng Y, Zhou X, et al. Improvement in overall survival in a randomized study that compared dacomitinib with gefitinib in patients with advanced non–small-cell lung cancer and EGFR-activating mutations. J Clin Oncol. 2018;36(22):2244-2250. doi:10.1200/JCO.2018.78.7994
9. Mok TS, Wu YL, Thongprasert S, et al. Gefitinib or carboplatin-paclitaxel in pulmonary adenocarcinoma. N Engl J Med. 2009;361(10):947-957. doi:10.1056/NEJMoa0810699
10. Pless M, Stupp R, Ris HB, et al. Induction chemoradiation in stage IIIA/N2 non–small-cell lung cancer: a phase 3 randomised trial. Lancet. 2015;386(9998):1049-1056. doi:10.1016/S0140-6736(15)60294-X
11. Albain KS, Rusch VW, Crowley JJ, et al. Concurrent cisplatin/etoposide plus chest radiotherapy followed by surgery for stages IIIA (N2) and IIIB non–small-cell lung cancer: mature results of Southwest Oncology Group phase II study 8805. J Clin Oncol. 1995;13(8):1880-1892. doi:10.1200/JCO.1995.13.8.1880
12. Zhong WZ, Chen KN, Chen C, et al. Erlotinib versus gemcitabine plus cisplatin as neoadjuvant treatment of Stage IIIA-N2 EGFR-mutant non–small-cell lung cancer (EMERGING-CTONG 1103): a randomized phase II study. J Clin Oncol. 2019;37(25):2235-2245. doi:10.1200/JCO.19.00075
13. Noronha V, Patil VM, Joshi A, et al. Gefitinib versus gefitinib plus pemetrexed and carboplatin chemotherapy in EGFR-mutated lung cancer. J Clin Oncol. 2020;38(2):124-136. doi:10.1200/JCO.19.01154
14. Noronha V, Prabhash K, Thavamani A, et al. EGFR mutations in Indian lung cancer patients: clinical correlation and outcome to EGFR targeted therapy. PLoS One. 2013;8(4):e61561. Published 2013 Apr 19. doi:10.1371/journal.pone.0061561
15. Kato T, Seto T, Nishio M, et al. Erlotinib plus bevacizumab phase ll study in patients with advanced non–small-cell lung cancer (JO25567): updated safety results. Drug Saf. 2018;41(2):229-237. doi:10.1007/s40264-017-0596-0
16. Hellmann MD, Paz-Ares L, Bernabe Caro R, et al. Nivolumab plus ipilimumab in advanced non–small-cell lung cancer. N Engl J Med. 2019;381(21):2020-2031. doi:10.1056/NEJMoa1910231
17. Hellmann MD, Ciuleanu TE, Pluzanski A, et al. Nivolumab plus ipilimumab in lung cancer with a high tumor mutational burden. N Engl J Med. 2018;378(22):2093-2104. doi:10.1056/NEJMoa1801946
18. Rizvi NA, Hellmann MD, Brahmer JR, et al. Nivolumab in combination with platinum-based doublet chemotherapy for first-line treatment of advanced non–small-cell lung cancer. J Clin Oncol. 2016;34(25):2969-2979. doi:10.1200/JCO.2016.66.9861
19. Zhong WZ, Wang Q, Mao WM, et al. Gefitinib versus vinorelbine plus cisplatin as adjuvant treatment for stage II-IIIA (N1-N2) EGFR-mutant NSCLC: final overall survival analysis of CTONG1104 Phase III Trial. J Clin Oncol. 2021;39(7):713-722. doi:10.1200/JCO.20.01820
20. Yang JC, Sequist LV, Geater SL, et al. Clinical activity of afatinib in patients with advanced non-small-cell lung cancer harbouring uncommon EGFR mutations: a combined post-hoc analysis of LUX-Lung 2, LUX-Lung 3, and LUX-Lung 6. Lancet Oncol. 2015;16(7):830-838. doi:10.1016/S1470-2045(15)00026-1
21. Shi YK, Wang L, Han BH, et al. First-line icotinib versus cisplatin/pemetrexed plus pemetrexed maintenance therapy for patients with advanced EGFR mutation-positive lung adenocarcinoma (CONVINCE): a phase 3, open-label, randomized study. Ann Oncol. 2017;28(10):2443-2450. doi:10.1093/annonc/mdx359
22. Soria JC, Wu YL, Nakagawa K, et al. Gefitinib plus chemotherapy versus placebo plus chemotherapy in EGFR-mutation-positive non-small-cell lung cancer after progression on first-line gefitinib (IMPRESS): a phase 3 randomized trial. Lancet Oncol. 2015;16(8):990-998 doi:10.1016/S1470-2045(15)00121-7
23. Goss GD, O’Callaghan C, Lorimer I, et al. Gefitinib versus placebo in completely resected non-small-cell lung cancer: results of the NCIC CTG BR19 study. J Clin Oncol. 2013;31(27):3320-3326. doi:10.1200/JCO.2013.51.1816
24. Sun JM, Lee KH, Kim SW, et al. Gefitinib versus pemetrexed as second-line treatment in patients with non-small cell lung cancer previously treated with platinum-based chemotherapy (KCSG-LU08-01): an open-label, phase 3 trial. Cancer. 2012;118(24):6234-6242. doi:10.1200/JCO.2013.51.1816
25. Mitsudomi T, Morita S, Yatabe Y, et al. Gefitinib versus cisplatin plus docetaxel in patients with non-small-cell lung cancer harbouring mutations of the epidermal growth factor receptor (WJTOG3405): an open label, randomized phase 3 trial. Lancet Oncol. 2010;11(2):121-128. doi:10.1016/S1470-2045(09)70364-X
26. Lee DH, Park K, Kim JH, Lee JS, et al. Randomized phase III trial of gefitinib versus docetaxel in non-small cell lung cancer patients who have previously received platinum-based chemotherapy. Clin Cancer Res. 2010;16(4):1307-1314. doi:10.1158/1078-0432.CCR-09-1903
27. Kim ES, Hirsh V, Mok T, et al. Gefitinib versus docetaxel in previously treated non-small-cell lung cancer (INTEREST): a randomized phase III trial. Lancet. 2008;22;372(9652):1809-1818. doi:10.1016/S0140-6736(08)61758-4