User login
Using Contingency Management for the Treatment of Substance Use Disorders in Real-World Settings
From UConn Health, Farmington, CT.
Abstract
- Objective: To discuss the efficacy and generalizability of contingency management (CM) for the treatment of substance use disorders and design considerations for those considering implementing in clinical settings.
- Methods: Review of the literature.
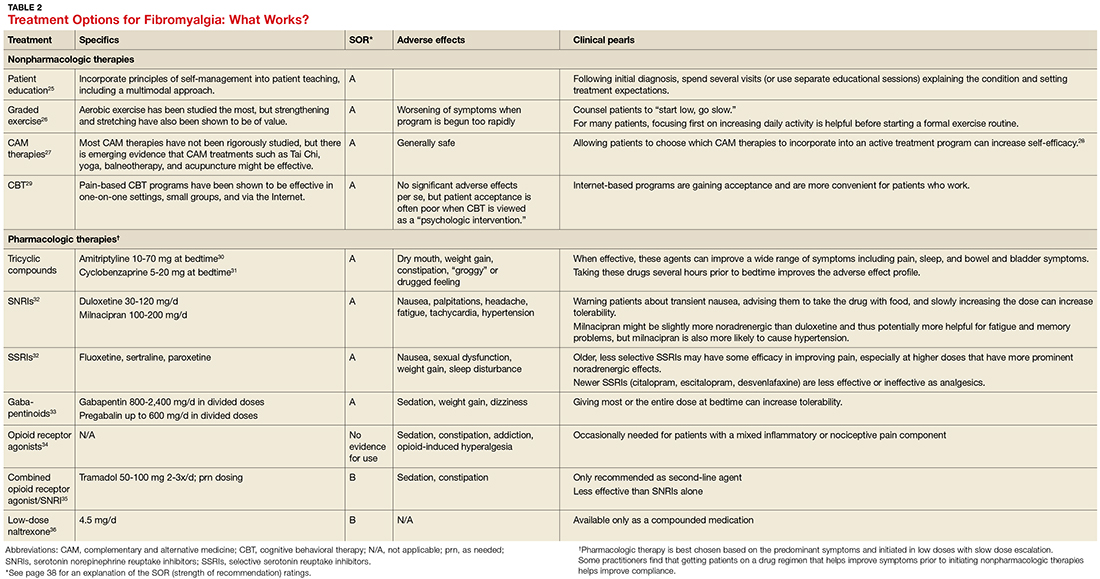
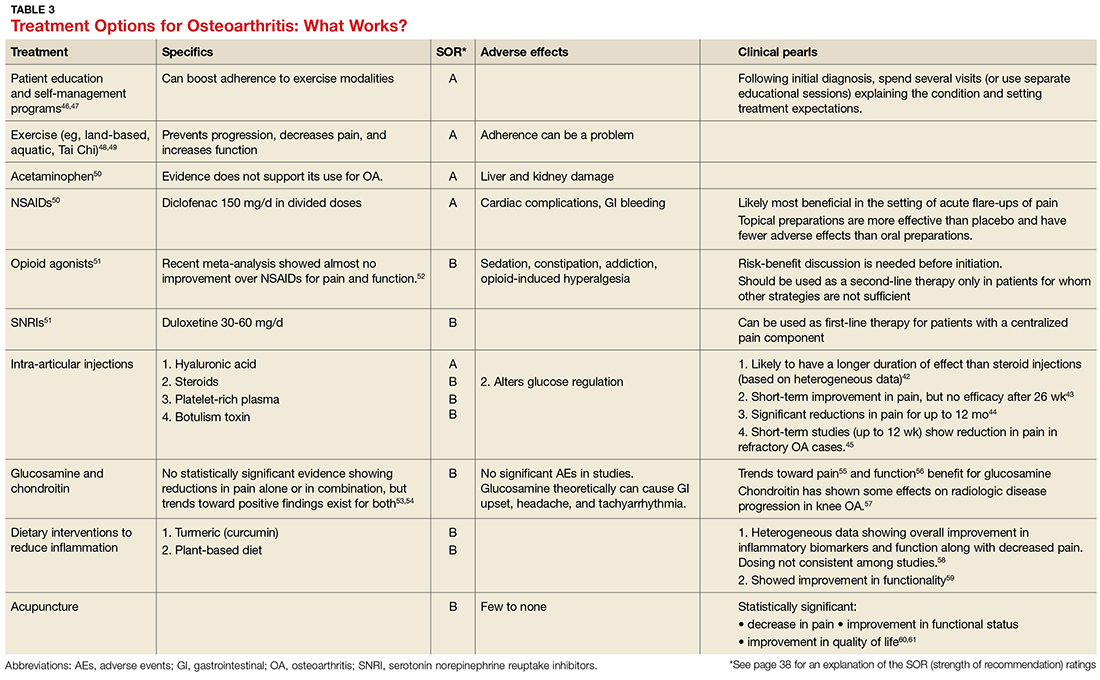
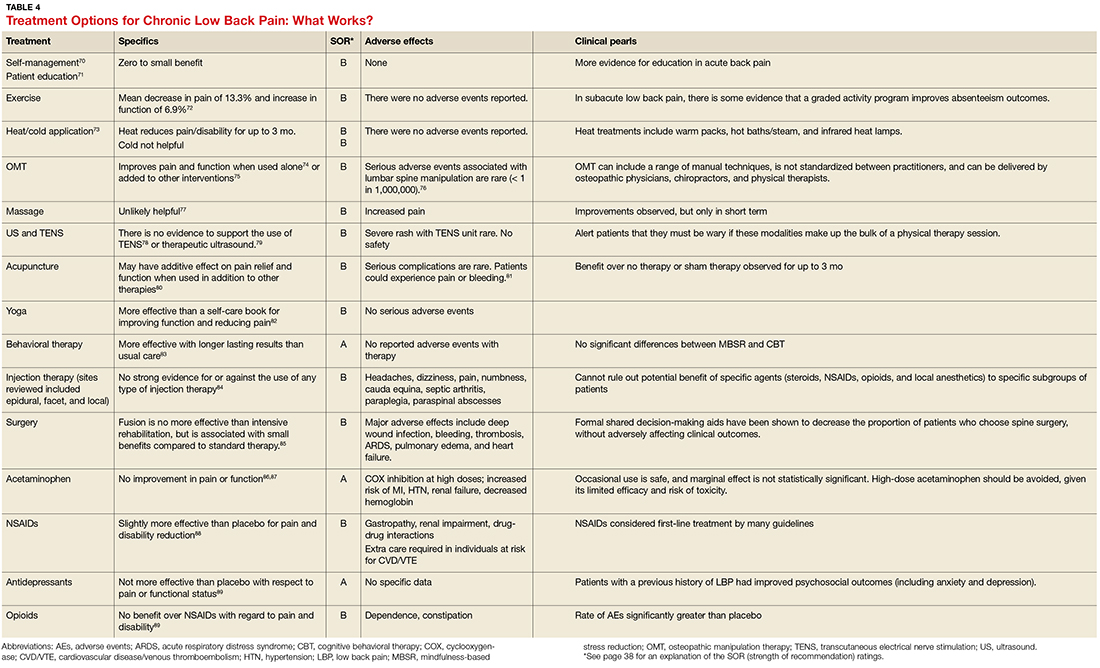
- Results: CM is an efficacious treatment for substance abuse disorders that is widely generalizable across substance use disorders and patient characteristics. CM can be implemented in a number of treatment programs, including residential and outpatient settings, and it can be administered in both individual and group formats. Abstinence and attendance are the most commonly targeted behaviors in substance abuse treatment settings. Design features, including the selection of target behaviors, delivery methods, and reinforcers, are discussed. Schedule parameters, such as frequency, magnitude, immediacy, and escalation of reinforcement, are associated with overall impact of the CM program and are important considerations for those interested in tailoring CM protocols to their needs.
- Conclusion: CM is an efficacious option that is applicable to most substance abuse treatment patients. A number of demonstrations of real-world implementation have been published and suggest CM can be adapted with success to clinic settings. In adopting CM protocols, clinics should aim for those protocols with established efficacy; however, if adaptations are necessary, careful consideration should be given to modifications to minimize risks of undermining CM’s effects.
Key words: incentives; reinforcement; substance abuse treatment; dissemination; implementation.
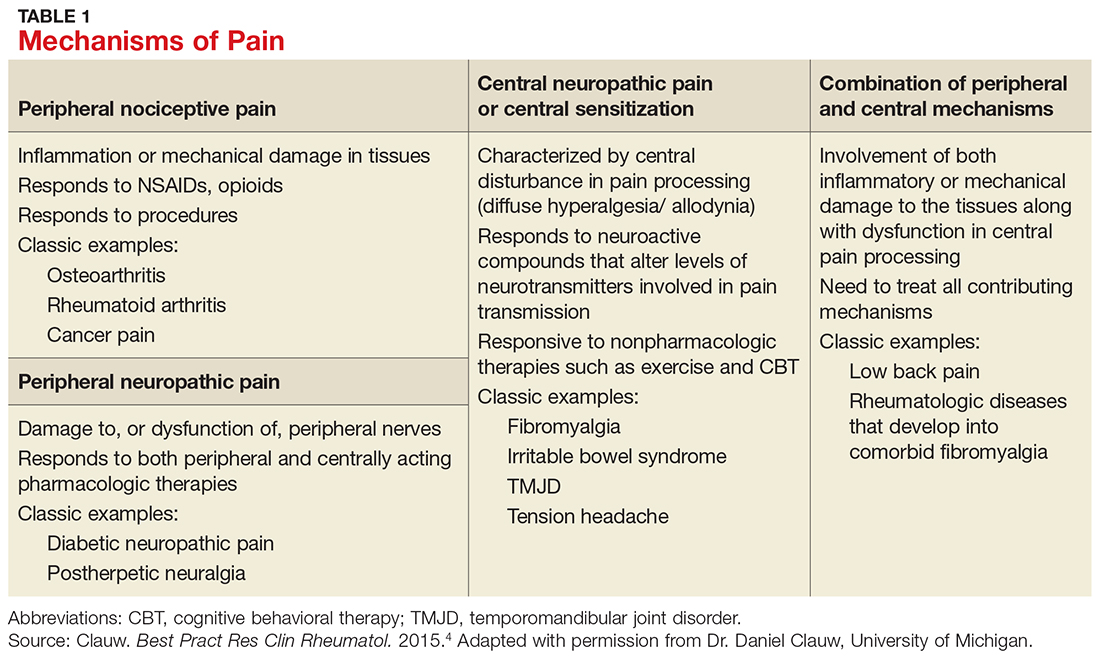
Contingency management (CM) is a behavioral intervention that is efficacious in the treatment of substance use disorders (SUDs). CM uses a behavior analytic framework and applies principles of learning theory, particularly operant conditioning theory, to change client behavior(s) [1–5]. In basic terms, operant conditioning principles suggests that whether a behavior continues or not is a function of consequences [6]. Reinforced behaviors are more likely to occur in the future. Substance abuse can be viewed as a behavior maintained by the reinforcing effects of the drug itself [5], including the feel-good aspects of intoxication or relaxation and the amelioration of withdrawal symptoms. CM extends these same principles of to a treatment context, such that reinforcers for abstinent behavior are introduced to compete with the reinforcing effects of continued drug use [5].
In CM’s application to substance abuse treatment, drug-negative samples or treatment attendance are reinforced using tangible incentives with the goal of motivating continued abstinence and/or treatment engagement. When clients demonstrate these target behaviors, they earn incentives in the form of goods or services of value to the client, such as small electronics, gift cards, and toiletries. Despite the promising effects observed in research trials, real-world implementation efforts have not kept pace [7–9]. This review briefly discusses CM’s efficacy and highlights key features for professionals considering adopting this intervention. Demonstration efforts that illustrate how CM can be effectively implemented within the constraints and limitations of non-research, clinical settings are also presented.
Efficacy of CM
CM’s efficacy spans a number of SUDs, including cocaine, opioids, alcohol, nicotine, and marijuana [10–13], making it amenable for treatment of most SUD clinic populations. It generates larger effect sizes than other SUD treatments, including cognitive behavioral therapy [14], and it has been evaluated in a wide range of settings. Large-scale evaluations have been conducted in both intensive outpatient [15] and methadone maintenance [16] settings as part of the National Institute on Drug Abuse Clinical Trials Network, demonstrating consistent benefits of CM when added to treatment as usual. In the first of these 2 studies, Petry et al [15] randomized 415 stimulant users from 1 of 8 intensive outpatient clinics to treatment as usual or treatment as usual plus CM for alcohol and stimulant abstinence. CM participants submitted more substance-negative urine and breath samples, achieved continuous abstinence at significantly higher rates, and had longer treatment retention compared to those receiving treatment as usual. The parallel study [16] focused on stimulant use in clients from methadone maintenance clinics and found similar benefits of CM on stimulant abstinence. Beyond these settings, CM has been applied in a number of other contexts, including drop-in centers [17], vocational rehabilitation [18,19], job-skills training [20], and residential programs [21–23]. In addition, several group-based adaptations have been explored [17,24–27].
CM benefits most clients and generalizes across several demographic variables, including gender [28,29], race [30], housing status [31], and income levels [32–34]. Among clinical characteristics, CM is efficacious for those with co-occurring SUDs [35], other substance use [36], psychiatric disorders [37–39], medical problems [40–42], and history of transactional sex [43].
Design Considerations
Design features, including what behavior will be reinforced and how to do so, are among the first decision points for clinicians interested in implementing CM. One of the advantages of CM is that it has a high degree of flexibility in design, which means that it can be readily tailored to client populations and clinic needs. However, this flexibility can lead clinicians astray from the foundational principles of CM and unknowingly weaken the impact of the program. Below, some key considerations for CM protocol design are reviewed. For additional coverage of these topics, readers are referred to additional articles [1,2] or Petry’s comprehensive book on implementing CM [44]. In this review, published examples of CM’s application in real-world settings are presented, highlighting how CM has been adapted in these clinical efforts.
Target Behaviors
The selection of the target behavior will drive many of the subsequent program design decisions. As such, it is important to identify this feature early. Target behaviors must be achievable, objectively verifiable, and well defined. The most common CM targets are drug abstinence or therapy session attendance. CM has also been used to target other behaviors, such as medication adherence [45,46], treatment-related activities [47,48], and exercise [49–51]. Client self-report of behaviors or vaguely defined behaviors (eg, “good participation”) should be avoided. While some of the decisions related to CM protocols are flexible, the use of objectively verifiable target behaviors is a core feature that should not be neglected. If the behavior of interest cannot be objectively verified, an alternate behavior should be chosen.
Selection of the target behavior is often considered in hand with defining which population is eligible to participate in the CM program. Client characteristics are often forefront in this decision, but clinic-driven logistical issues or unmet needs may also play a role. A real-world example of this decision process is evident in the nationwide rollout of CM among the intensive outpatient programs within the Veterans Administration (VA). The VA identified a treatment need for those with stimulant use disorders, as this group did not have efficacious pharmacotherapy options available that targeted stimulant use. As such, the VA applied CM to patients with a focus on stimulant abstinence as the behavioral target [52]. For others, the decision may revolve around addressing underutilization of specific treatment resources (eg, outpatient groups, vocational rehabilitation) [53–56] or treatment needs among certain subgroups of clients, such as adolescents [57–59].
For abstinence targets, clinics would need to select one or more specific substances as the focus of the CM program. In general, targeting a single substance rather than multiple substances is more effective [10,13], is more straightforward for clients to understand, and allows more clients to access the reinforcers. Exposure to the reinforcers is necessary for CM to work; thus, setting a goal that is achievable for most clients should be a priority. Requiring abstinence from multiple substances means that some clients may never experience the reinforcer and thus cannot benefit from its effects at all. Some clinicians or administrators may initially have reservations about reinforcing single drug abstinence in the event that other drug use continues. However, targeting a single substance for reinforcement often results in reduced use of other substances [60]. Clinicians may find that this makes intuitive sense; a client with cocaine use disorder who is trying to maintain cocaine abstinence over a long period is likely to avoid using alcohol or other substances that might lead to relapse. For abstinence, objective verification through urine or breath specimens using tests that include validity checks is relatively straightforward.
Attendance is a popular target for clinics in part because it does not require additional staff time to collect specimen samples and it was the most commonly reported target behavior in samples of SUD providers who use incentives [61,62]. Objective verification of attendance is usually via a staff member, but expectations must be clear to both parties. Clinics should consider potential problems that may arise (eg, arriving late, leaving group early, excused absences) and carefully define and communicate expectations for the CM program. Piloting [19] the CM program with a small group of clients may be valuable in trouble-shooting challenges before wider implementation.
In a recent study [55], clients earned reinforcers for attending clinician-led group counseling sessions and/or the in-clinic patient-led Methadone Anonymous (MA) groups. This non-research, clinical effort addressed historically poor therapy attendance at the clinic, and attendance rates were compared before, during, and after the CM program. CM increased attendance to both groups in the short-term after implementation, but effects were more robust for the MA groups in which increased attendance persisted 3 months following the withdrawal of the contingencies. Overall effects of this program were modest, but they are notable given the use of an ultra-low cost approach.
Delivery Methods
The majority of CM studies used voucher or prize-based methods. Head-to-head comparisons suggest that they are comparable in efficacy [63–65], and each has advantages and disadvantages that may make one option more appealing for a given clinic. Voucher programs are generally straightforward to administer. Clients earn vouchers for each instance of the target behavior. The value of the vouchers typically increases with consecutive performance. The schedule used in the influential Higgins et al studies [66,67] started at $2.50 for the first cocaine-negative sample and increased by $0.50 for each subsequent consecutive cocaine-negative sample. Earned vouchers are exchanged for goods or services selected by the client, increasing the likelihood that the selected items will be highly desirable and allowing for a wide range of client preferences. Clients appear to prefer this approach when given a choice between set schedules or those that introduce an element of chance (ie, prize-based CM, discussed below) [68]. However, voucher programs can be costly (~$1000 per client over 12 weeks) and may require more staff time to fulfill individual requests for specific items. However, staff burden related to shopping can be reduced by limiting these individual requests and using an on-site stocked cabinet of goods similar to prize-CM programs.
Prize-based CM is similar but introduces probabilistic earnings and variability in prize magnitude. Rather than earning vouchers, clients earn draws from a fishbowl for each instance of the target behavior, again typically in an escalating manner. For example, a client may earn one draw from the fishbowl for the first cocaine-negative sample, 2 draws for the second consecutive negative sample, 3 draws for the third, and so on. A typical fishbowl is composed of 500 slips, some noting prizes and some having no prize value. Typically, half the slips in the bowl are non-monetary “good jobs.” The remaining half are small prizes worth about $1 in value (eg, food coupons, bus tokens, small toiletries), large prizes worth about $20 in value (eg, small electronics, gift certificates), and one slip is the jumbo prize worth about $100. When a client draws a winning slip, they select a prize from that category (ie, small, large, jumbo) from an onsite, stocked cabinet. Due to the probabilistic feature of prize-based CM, overall costs of the program can be substantially lower than typical voucher programs, with average maximum expected earnings ranging $250 t $450 per client over a 12-week treatment period [15,16,65,69]. Advantages of this method include potentially lower costs and minimal shopping demands (a once-monthly shopping trip to restock the cabinet will usually suffice) while maintaining comparable efficacy. Relative to voucher programs, prize-based CM involves additional administration time to allow for drawing slips from the fishbowl, which can be compounded when multiple clients want to draw at the same time such as in a group setting. Many of the group-based CM adaptations address this issue by limiting the number of clients who can draw for prizes in a given group or by limiting the number of draws per client [25,27,54].
Reinforcers
Regardless of whether selecting voucher or prize CM, reinforcers are critically important to the success of the program. Reinforcers must be desirable. One of the quickest ways to undermine a CM program is lack of variety or undesirable reinforcers. If stocking a cabinet with prizes onsite, care should be taken to have numerous options within each of the small and large prize categories that are appealing to a wide range of clients. Since a client who is consistently earning draws will choose prizes often, it is imperative to include enough variety so that even these clients find desirable items each time they select a prize. Clients should be asked regularly if they have suggestions for prizes; one program [54] found suggestion boxes useful for encouraging clients to voice their preferences. Donations can be solicited from local businesses to reduce costs [53], and low-cost but high-value options, such as clinic privileges, can also be explored. Petry [1,44] provides some suggestions of the latter, and Amass and Kamien [70] describe their successful strategies to fund and sustain a clinic-based CM program through community donations. Some clinics may already have tangible goods, such as gas or metro cards, that are offered to clients based on need rather than behavior [53]. These existing resources might be redirected to a CM program, in which these goods are contingent on abstinence or attendance, if appropriate.
Schedule Parameters
Once the target behavior, client population, and CM delivery methods are selected, the next step is to design the reinforcement schedule. The following schedule parameters apply to both voucher and prize-based CM systems. The more closely a clinical program adheres to the parameters of effective protocols, the more likely the program is to generate comparable outcomes. If there is a parameter or design feature that is incompatible with clinic needs, modifications can be introduced. However, each deviation away from the ideal has a chance of undermining the success of the CM program. Any changes and their potential impacts should considered carefully, and consultation with a CM expert may aid in the development of successful and efficacious clinic-based protocols. Of note, a meta-analysis [13] of CM studies found that researcher involvement in the planning and design of CM programs is associated with larger treatment effects. CM researchers are especially attuned to the potential impacts and pitfalls associated with modifying CM protocols, and they can be valuable resources for clinics interested in tailoring a CM program to their specific needs. Several examples of clinical demonstration projects that used researcher input are available [19,53,71].
Magnitude
Incentive magnitude was directly related to the size of treatment effects in a meta-analysis [11] of CM studies. Although not all studies find significant differences in outcomes related to magnitude [65,72], the bulk of evidence suggests magnitude is an important parameter and is related to effect size for both voucher [73–75] and prize-based CM [69,76] systems. Thus, although clinics may have restrictive budgets, severely undercutting the magnitude of rewards is not usually the solution as it can undermine treatment effects [76]. Donations can reduce overall costs [53,57,70], and other protocol features discussed below, such as capping the amount of reinforcement available, can reduce the overall magnitude available per patient.
Another approach, used in group-based CM, limits the number of patients who earn prizes per week [25,27]. For example, in a 2011 study by Petry et al, clients added slips with their name to a bowl for attendance and negative samples. Once all names were collected in the bowl, the group leader would pull a specified number of slips (eg, 3 slips per group). These individuals were eligible to draw from the prize bowl for prizes. This approach was associated with longer durations of consecutive abstinence and better treatment attendance relative to treatment as usual. However, clinics can control the overall program costs by limiting the number of patients eligible for prizes.
Frequency
Frequent reinforcement opportunities are ideal, and more frequent assessment is associated with larger treatment effects [10]. However, a number of factors, including which target behavior is selected and logistical issues specific to the clinic such as when groups meet, will play a role in determining the frequency of CM sessions. For abstinence targets, the substance targeted and type of test will largely determine the frequency of CM sessions. The goal would be to test at a frequency that would detect most or nearly all instances of use. For cocaine or opioids, this equates to testing 2 to 3 times weekly. Breath samples for alcohol or cigarette smoking would necessitate testing daily or multiple times per day to detect most instances of use because these tests have short windows of detection. CM protocols based on these breath tests have often had daily or twice daily CM sessions [77,78]; technological adaptations [77,79,80] or residential settings [21,23] may reduce burden to the client for assessment of these substances. Tapering the number of breath tests over time or transitioning from daily breath tests to once or twice weekly urine testing after abstinence is established is another approach [81,82].
Marijuana, on the other hand, poses difficulties because it is detectable in urine samples for up to 2 weeks following use. If relying solely on urine results for reinforcement, clients may not test negative for several days or weeks after last use, resulting in a delay of reinforcement. To address this issue, some CM programs targeting marijuana abstinence initially reinforce attendance in the first 2 weeks and then transition to reinforcing marijuana-negative drug samples for the remainder of the treatment period [48].
In general, more frequent CM sessions can translate to higher costs; however, infrequent reinforcement (ie, less than weekly) is not as effective [45]. In real-world applications, clinics often need to balance feasibility and costs with the ideal CM schedule. In abstinence-based CM, this compromise may result in a testing schedule that may not capture all instances of use. For example, while thrice-weekly testing may be ideal for cocaine or opioids, a twice-weekly schedule may be selected because it lowers costs and is more consistent with clinic schedules.
Immediacy
In general, clinics should aim to deliver reinforcement as immediately as possible, as delays between the target behavior and reinforcement are associated with decreased treatment effects [10,11,83]. For drug abstinence, onsite urine testing systems that provide immediate results are preferred over sending samples for laboratory testing. Clinics that do not have access to or who cannot afford specimen testing that allows onsite collection and immediate results might consider other options for target behaviors, such as attendance.
Immediacy of reinforcement is also important when targeting attendance. One clinic [53] implemented a program that offered a $50 incentive if clients attended 1 month of group therapy sessions. This approach was not effective and no clients earned the incentive for several months. After consultation, the clinic revised the incentive program to a daily drawing for attendance using the fishbowl method, thereby decreasing the delay between the behavior and its consequence. This example illustrates not only problems with delayed reinforcement but also the common mistake of setting expectations for the target behavior too high. Attending a month of group therapy sessions is a high bar that few patients will achieve, resulting in a system that mostly rewards those already doing well [19]. In contrast, attending a single group session in order to earn reinforcers is a reachable goal and increases the likelihood that more clients are exposed to the reinforcers. These small steps (ie, attending a single group or submitting a single drug negative urine) encourage initiation of the behavior(s) targeted. Other features, such as escalation (discussed next), aim to sustain the behavior over time.
Escalation
Escalation involves increasing the amount of reinforcement for each consecutive target behavior. In the voucher programs, the amount earned per negative sample may increase for each consecutive negative sample (eg, $2.50 for the first negative sample, $3.00 for the second, $3.50 for the third, and so on). For prize-based programs, the number of draws escalates with consecutive performance (eg, 1 draw for the first group attended, 2 draws for the second, 3 for the third, and so on). Protocols that include escalation generate larger effects than those that have a set, flat incentive amount even when total costs are the same across comparison conditions [73].
Escalating schedules usually include a reset feature. Following a positive or refused sample or unexcused absence, the amount earned for the next negative sample is reduced to the initial amount and begins escalating anew with consecutive negative samples. Some schedules allow for a rapid reset in which after a specified period of time or consecutive performance, the value jumps to the value achieved when the relapse occurred [66].
Despite its consistent inclusion in CM protocols from randomized clinical trials, our data [61] suggest that more than half of providers using incentives in treatment as part of a clinical effort do not use escalating reinforcers. Escalating schedules require more careful tracking of client progress, possibly contributing to lower uptake of this design feature in clinical practice. Development of simple tracking forms can minimize this challenge.
Another drawback of escalation pertaining to prize-based CM is that escalating schedules can affect the duration of CM sessions when clients are drawing a large number of slips each session and escalation can increase costs of the overall program. Capping the number of draws will help mitigate both issues. For example, once a client reaches 10 draws for group attendance, they continue earning 10 draws for each consecutive session attended with no further escalation.
Putting It All Together
CM sessions can be conducted as stand-alone sessions or incorporated into group or individual therapy sessions. Many clinicians will find the latter approach sets a positive tone for the therapy session given CM's focus on what the client is doing well. Starting the treatment session with the CM component often naturally leads into a discussion of relevant therapeutic issues, such as effective coping, slips, or triggers. The CM session length can be variable, but it is typically under 10 minutes. Thus, the CM component need not dominate the clinical session or content. CM sessions for abstinence are scheduled according to a set schedule (eg, Mondays and Thursdays) and often coincide with other treatment aspects (eg, before or after group therapy on Mondays and Thursdays). CM sessions for attendance also generally follow a set schedule (eg, client expected to attend Monday and Wednesday group therapy sessions). The duration of the CM protocol can also vary, with most clinical trials ranging from 12 to 24 weeks. Very short durations are unlikely to produce lasting behavior change, particularly with complex behaviors such as abstinence. Petry [44] recommends no less than 8 weeks duration and a maximum duration of 24 weeks.
As discussed, CM offers many opportunities for tailoring to optimize its fit within the existing structure of clinics. However, this flexibility must be viewed together with an understanding of the principles that impact CM's efficacy. Specific recommendations for CM protocol development will depend on the behavior targeted, the delivery methods, and format (eg, individual versus group settings). For these reasons, consultation with a CM expert is ideal. However, some general guidelines for developing a CM program that incorporate the principles discussed above include an 8- to 12-week program that (1) provides sufficient magnitude to compete with the behavior you are attempting to change, (2) offers frequent opportunities for reinforcement (eg, 2-3 times/wk for opioids or stimulant abstinence, 1-2 times/week for attendance targets; not less than weekly for most behaviors), (3) delivers the reinforcement immediately or very close in time with the behavior (eg, reinforce attendance at the beginning of the group, use onsite urine testing and reinforce immediately after testing), and (4) incorporates escalating and reset features into the schedule.
Clinician Training and Supervision
Training in CM is an important part of the implementation process. Studies [62,84–87] have identified a number of perceived barriers to and negative beliefs about CM, including philosophical and logistical concerns. Tangible incentives, the core of most CM protocols, are generally viewed less favorably than social or nonspecified incentives [84,86,87]. Philosophical concerns relate to CM’s inability to address the underlying causes of addiction, that it does not address multiple behaviors, and that it may undermine internal motivation for sobriety [62,84]. An additional objection relates to paying someone to do what they should do on their own [86]. Logistical and practical concerns often represent implementation barriers such as costs and access to training and supervision, but they also reflect concern for what happens when contingencies are withdrawn, that clients may sell or trade prizes for drugs, and worries that CM’s evidence does not generalize to clinic populations [62].
Many of these beliefs reflect a limited understanding of CM, and addressing these misperceptions is a first step toward reducing resistance to implementation efforts. For example, a substantial body of literature points to CM’s wide generalizability across a range of characteristics, clients that sell or trade prizes for drugs are likely to disrupt their chain of negative samples or attendance, and most studies do not find negative impacts of CM on intrinsic motivation [88–90]. Fortunately, CM training appears to be an effective way to address negative beliefs. In the VA implementation effort [52], training workshops decreased perceived barriers and increased positive impressions of CM [91]. In other training efforts, brief educational materials were effective in changing perceptions of CM’s efficacy [92].
Beyond initial training, supervision of CM delivery is likely to be necessary [93,94]. Clinician skill in delivering CM is related to client outcomes [93,95] and relatively simple adherence measures are available for monitoring [96,97]. However, the best methods for training and supervision of CM have yet to be established. The VA initiative was developed in consultation with CM experts and employed ongoing phone consultation following initial training workshops [52,91]. This approach represented significant investment by the VA toward staff training and CM protocol development that may not be achievable for individual clinics. As attention to CM’s dissemination and implementation has grown, some free resources have been developed. Promoting Awareness of Motivational Incentives (PAMI; www.bettertxoutcomes.org/bettertxoutcomes/PAMI.html) is a collaborative initiative sponsored by the National Institute of Drug Abuse and the Substance Abuse and Mental Health Services Administration. It offers free resources and training materials.
Conclusion
Overall, CM is a highly efficacious treatment for SUDs that generalizes to most clients. Despite a robust evidence base, CM’s implementation in clinical settings lags behind other empirically supported treatments [92]. At least in part, CM’s costs, which include not only staff training and adherence monitoring (as with other treatments), but also costs of the incentives themselves, may contribute to slow uptake in clinical settings. Clinics often do not have the resources available for CM within their operating budgets. However, a growing number of projects [19,52,53,55–57,70,71] illustrate CM implementation within routine clinical care, and increased revenue from improved attendance to treatment groups may be one mechanism through which to fund a CM program [54,56,57]. These projects are valuable not only for demonstrating that CM can be efficacious outside the research setting, but also for highlighting how implementation barriers can be overcome. Continued efforts of this nature are likely to be particularly valuable for clinicians and administrators considering adopting CM within clinical settings.
1. Petry NM. A comprehensive guide to the application of contingency management procedures in clinical settings. Drug Alcohol Depend 2000;58:9–25.
2. Stitzer M, Petry N. Contingency management for treatment of substance abuse. Annu Rev Clin Psychol 2006;2:411–34.
3. Meredith SE, Jarvis BP, Raiff BR, et al. The ABCs of incentive-based treatment in health care: A behavior analytic framework to inform research and practice. Psychol Res Behav Manag 2014;7:103–14.
4. Bigelow GE, Silverman K. Theoretical and empirical foundations of contingency management treatments for drug abuse. In: Higgins ST, Silverman K, editors. Motivating behavior change among illicit drug users. Washington DC: American Psychological Association; 1999: 15–31.
5. Higgins ST, Budney AJ, Bickel WK. Applying behavioral concepts and principles to the treatment of cocaine dependence. Drug Alcohol Depend 1994;34:87–97.
6. Skinner BF. Science and human behavior. New York: Macmillan; 1953.
7. Herbeck DM, Hser YI, Teruya C. Empirically supported substance abuse treatment approaches: A survey of treatment providers’ perspectives and practices. Addict Behav 2008;33:699–712.
8. McGovern MP, Fox TS, Xie H, Drake RE. A survey of clinical practices and readiness to adopt evidence-based practices: Dissemination research in an addiction treatment system. J Subst Abuse Treat 2004;26:305–12.
9. Willenbring ML, Kivlahan D, Kenny M, et al. Beliefs about evidence-based practices in addiction treatment: A survey of Veterans Administration program leaders. J Subst Abuse Treat 2004;26:79–85.
10. Griffith JD, Rowan-Szal GA, Roark RR, Simpson DD. Contingency management in outpatient methadone treatment: A meta-analysis. Drug Alcohol Depend 2000;58:55–66.
11. Lussier JP, Heil SH, Mongeon JA, et al. A meta-analysis of voucher-based reinforcement therapy for substance use disorders. Addiction 2006;101:192–203.
12. Benishek LA, Dugosh KL, Kirby KC, et al. Prize-based contingency management for the treatment of substance abusers: A meta-analysis. Addiction 2014;109:1426–36.
13. Prendergast M, Podus D, Finney J, et al. Contingency management for treatment of substance use disorders: A meta-analysis. Addiction 2006;101:1546–60.
14. Dutra L, Stathopoulou G, Basden SL, et al. A meta-analytic review of psychosocial interventions for substance use disorders. Am J Psychiatry 2008;165:179–87.
15. Petry NM, Peirce JM, Stitzer ML, et al. Effect of prize-based incentives on outcomes in stimulant abusers in outpatient psychosocial treatment programs: A National Drug Abuse Treatment Clinical Trials Network study. Arch Gen Psychiatry 2005;62:1148–56.
16. Peirce JM, Petry NM, Stitzer ML, et al. Effects of lower-cost incentives on stimulant abstinence in methadone maintenance treatment - A National Drug Abuse Treatment Clinical Trials Network study. Arch Gen Psychiatry 2006;63:201–8.
17. Petry NM, Martin B, Finocche C. Contingency management in group treatment: a demonstration project in an HIV drop-in center. J Subst Abuse Treat 2001;21:89–96.
18. Drebing CE, Van Ormer EA, Mueller L, et al. Adding contingency management intervention to vocational rehabilitation: outcomes for dually diagnosed veterans. J Rehabil Res Dev 2007;44:851–65.
19. Kellogg SH, Burns M, Coleman P, et al. Something of value: The introduction of contingency management interventions into the New York City Health and Hospital Addiction Treatment Service. J Subst Abuse Treat 2005;28:57–65.
20. Koffarnus MN, Wong CJ, Fingerhood M, et al. Monetary incentives to reinforce engagement and achievement in a job-skills training program for homeless, unemployed adults. J Appl Behav Anal 2013;46:582–91.
21. Rohsenow D, Martin R, Tidey JW, et al. Treating smokers in substance treatment with contingent vouchers, nicotine replacement, and brief advice adapted for sobriety settings. J Subst Abuse Treat 2017.
22. Hunt YM, Rash CJ, Burke RS, Parker JD. Smoking cessation in recovery: Comparing two different cognitive behavioral treatments. Addict Disord Their Treat 2010;9:64–74.
23. Alessi SM, Petry NM, Urso J. Contingency management promotes smoking reductions in residential substance abuse patients. J Appl Behav Anal 2008;41:617–22.
24. Petry NM, Weinstock J, Alessi SM, et al. Group-based randomized trial of contingencies for health and abstinence in HIV patients. J Consult Clin Psychol 2010;78:89.
25. Petry NM, Weinstock J, Alessi SM. A randomized trial of contingency management delivered in the context of group counseling. J Consult Clin Psychol 2011;79:686–96.
26. Ledgerwood DM, Alessi SM, Hanson T, et al. Contingency management for attendance to group substance abuse treatment administered by clinicians in community clinics. J Appl Behav Anal 2008;41:517–26.
27. Alessi SM, Hanson T, Wieners M, Petry NM. Low-cost contingency management in community clinics: Delivering incentives partially in group therapy. Exp Clin Psychopharmacol 2007;15:293–300.
28. Burch AE, Rash CJ, Petry NM. Sex effects in cocaine-using methadone patients randomized to contingency management interventions. Exp Clin Psychopharmacol 2015;23:284–90.
29. Rash CJ, Petry NM. Contingency management treatments are equally efficacious for both sexes in intensive outpatient settings. Exp Clin Psychopharmacol 2015;23:369–76.
30. Barry D, Sullivan B, Petry NM. Comparable efficacy of contingency management for cocaine dependence among African American, Hispanic, and White methadone maintenance clients. Psychol Addict Behav 2009;23:168–74.
31. Rash CJ, Alessi SM, Petry NM. Substance abuse treatment patients in housing programs respond to contingency management interventions. J Subst Abuse Treat 2017;72:97–102.
32. Rash CJ, Olmstead TA, Petry NM. Income does not affect response to contingency management treatments among community substance abuse treatment-seekers. Drug Alcohol Depend 2009;104:249–53.
33. Rash CJ, Andrade LF, Petry NM. Income received during treatment does not affect response to contingency management treatments in cocaine-dependent outpatients. Drug Alcohol Depend 2013;132:528–34.
34. Secades-Villa R, García-Fernández G, Peña-Suárez E, et al. Contingency management is effective across cocaine-dependent outpatients with different socioeconomic status. J Subst Abuse Treat 2013;44:349–54.
35. Rash CJ, Alessi SM, Petry NM. Cocaine abusers with and without alcohol dependence respond equally well to contingency management treatments. Exp Clin Psychopharmacol 2008;16:275–81.
36. Alessi SM, Rash C, Petry NM. Contingency management is efficacious and improves outcomes in cocaine patients with pretreatment marijuana use. Drug Alcohol Depend 2011;118:62–7.
37. Ford JD, Hawke J, Alessi S, et al. Psychological trauma and PTSD symptoms as predictors of substance dependence treatment outcomes. Behav Res Ther 2007;45:2417–31.
38. Weinstock J, Alessi SM, Petry NM. Regardless of psychiatric severity the addition of contingency management to standard treatment improves retention and drug use outcomes. Drug Alcohol Depend 2007;87:288–96.
39. García-Fernández G, Secades-Villa R, García-Rodríguez O, et al. Contingency management improves outcomes in cocaine-dependent outpatients with depressive symptoms. Exp Clin Psychopharmacol 2013;21:482–9.
40. Walter KN, Petry NM. Patients with diabetes respond well to contingency management treatment targeting alcohol and substance use. Psychol Health Med 2015;20:916–26.
41. Burch AE, Morasco BJ, Petry NM. Patients undergoing substance abuse treatment and receiving financial sssistance for a physical disability respond well to contingency management treatment. J Subst Abuse Treat 2015;58:67–71.
42. Burch AE, Rash CJ, Petry NM. Cocaine-using substance abuse treatment patients with and without HIV respond well to contingency management treatment. J Subst Abuse Treat 2017;77:21–5.
43. Rash CJ, Burki M, Montezuma-Rusca JM, Petry NM. A retrospective and prospective analysis of trading sex for drugs or money in women substance abuse treatment patients. Drug Alcohol Depend 2016;162:182–9.
44. Petry NM. Contingency management for substance abuse treatment: A guide to implementing this evidence-based practice. New York: Routledge; 2012.
45. Petry NM, Rash CJ, Byrne S, et al. Financial reinforcers for improving medication adherence: Findings from a meta-analysis. Am J Med 2012;125:888–96.
46. Herrmann ES, Matusiewicz AK, Stitzer ML, et al. Contingency management interventions for HIV, tuberculosis, and hepatitis control among individuals with substance use disorders: a systematized review. J Subst Abuse Treat 2017;72:117–25.
47. Petry NM, Alessi SM, Carroll KM, et al. Contingency management treatments: Reinforcing abstinence versus adherence with goal-related activities. J Consult Clin Psychol 2006;74:592–601.
48. Litt MD, Kadden RM, Petry NM. Behavioral treatment for marijuana dependence: Randomized trial of contingency management and self-efficacy enhancement. Addict Behav 2013;38:1764–75.
49. Kurti AN, Dallery J. A laboratory-based evaluation of exercise plus contingency management for reducing cigarette smoking. Drug Alcohol Depend 2014;144:201–9.
50. Weinstock J, Capizzi J, Weber SM, et al. Exercise as an intervention for sedentary hazardous drinking college students: A pilot study. Ment Health Phys Act 2014;7:55–62.
51. Mitchell MS, Goodman JM, Alter DA, et al. Financial incentives for exercise adherence in adults: Systematic review and meta-analysis. Am J Prev Med 2013;45:658–67.
52. Petry NM, Dephilippis D, Rash CJ, et al. Nationwide dissemination of contingency management: The Veterans Administration initiative. Am J Addict 2014;23:205–10.
53. Walker R, Rosvall T, Field CA, et al. Disseminating contingency management to increase attendance in two community substance abuse treatment centers: Lessons learned. J Subst Abuse Treat 2010;39:202–9.
54. Sigmon SC, Stitzer ML. Use of a low-cost incentive intervention to improve counseling attendance among methadone-maintained patients. J Subst Abuse Treat 2005;29:253–8.
55. Kropp F, Lewis D, Winhusen T. The effectiveness of ultra-low magnitude reinforcers: Findings from a “real-world” application of contingency management. J Subst Abuse Treat 2017;72:111–6.
56. Fitzsimons H, Tuten M, Borsuk C, et al. Clinician-delivered contingency management increases engagement and attendance in drug and alcohol treatment. Drug Alcohol Depend 2015;152:62–7.
57. Lott DC, Jencius S. Effectiveness of very low-cost contingency management in a community adolescent treatment program. Drug Alcohol Depend 2009;102:162–5.
58. Henggeler SW, Chapman JE, Rowland MD, et al. Statewide adoption and initial implementation of contingency management for substance-abusing adolescents. 2008;76:556–67.
59. Henggeler SW, Chapman JE, Rowland MD, et al. If you build it , they will come: Statewide practitioner interest in contingency management for youths. 2007;32:121–31.
60. Petry NM, Martin B, Cooney JL, Kranzler HR. Give them prizes, and they will come: contingency management for treatment of alcohol dependence. J Consult Clin Psychol 2000;68:250–7.
61. Rash CJ, Petry NM, Alessi SM. Examining implementation of contingency management in real-world settings [Abstract]. Alcohol Clin Exp Res 2016;20:103A.
62. Rash CJ, Petry NM, Kirby KC, et al. Identifying provider beliefs related to contingency management adoption using the Contingency Management Beliefs Questionnaire. Drug Alcohol Depend 2012;121:205–12.
63. Petry NM, Alessi SM, Marx J, et al. Vouchers versus prizes: Contingency management treatment of substance abusers in community settings. J Consult Clin Psychol 2005;73:1005–14.
64. Petry NM, Alessi SM, Hanson T, Sierra S. Randomized trial of contingent prizes versus vouchers in cocaine-using methadone patients. J Consult Clin Psychol 2007;75:983–91.
65. Petry NM, Alessi SM, Barry D, Carroll KM. Standard magnitude prize reinforcers can be as efficacious as larger magnitude reinforcers in cocaine-dependent methadone patients. J Consult Clin Psychol 2015;83:464–72.
66. Higgins ST, Budney AJ, Bickel WK, et al. Incentives improve outcome in outpatient behavioral treatment of cocaine dependence. Arch Gen Psychiatry 1994;51:568.
67. Higgins ST, Wong CJ, Badger GJ, et al. Contingent reinforcement increases cocaine abstinence during outpatient treatment and 1 year of follow-up. J Consult Clin Psychol 2000;68:64–72.
68. Hartzler B, Garrett S. Interest and preferences for contingency management design among addiction treatment clientele. Am J Drug Alcohol Abuse 2016;42:287–95.
69. Petry NM, Barry D, Alessi SM, et al. A randomized trial adapting contingency management targets based on initial abstinence status of cocaine-dependent patients. J Consult Clin Psychol 2012;80:276–85.
70. Amass L, Kamien J. A tale of two cities: Financing two voucher programs for substance abusers through community donations. Exp Clin Psychopharmacol 2004;12:147–55.
71. Hartzler B. Building a bonfire that remains stoked: Sustainment of a contingency management intervention developed through collaborative design. Subst Abuse Treat Prev Policy 2015;10:30.
72. Carroll KM, Sinha R, Nich C, et al. Contingency management to enhance naltrexone treatment of opioid dependence: a randomized clinical trial of reinforcement magnitude. Exp Clin Psychopharmacol 2002;10:54–63.
73. Roll JM, Shoptaw S. Contingency management: Schedule effects. Psychiatry Res 2006;144:91–3.
74. Silverman K, Chutuape MA, Bigelow GE, Stitzer ML. Voucher-based reinforcement of cocaine abstinence in treatment-resistant methadone patients: Effects of reinforcement magnitude. Psychopharmacology (Berl) 1999;146:128–38.
75. Businelle MS, Rash CJ, Burke RS, Parker JD. Using vouchers to increase continuing care participation in veterans: does magnitude matter? Am J Addict 2009;18:122–9.
76. Petry NM, Tedford J, Austin M, et al. Prize reinforcement contingency management for treating cocaine users: How low can we go, and with whom? Addiction 2004;99:349–60.
77. Alessi SM, Petry NM. A randomized study of cellphone technology to reinforce alcohol abstinence in the natural environment. Addiction 2013;108:900–9.
78. Alessi SM, Petry NM. Smoking reductions and increased self-efficacy in a randomized controlled trial of smoking abstinence--contingent incentives in residential substance abuse treatment patients. Nicotine Tob Res 2014;16:1436–45.
79. Dougherty DM, Hill-Kapturczak N, Liang Y, et al. Use of continuous transdermal alcohol monitoring during a contingency management procedure to reduce excessive alcohol use. Drug Alcohol Depend 2014;142:301–6.
80. Dallery J, Meredith S, Jarvis B, Nuzzo PA. Internet-based group contingency management to promote smoking abstinence. Exp Clin Psychopharmacol 2015;23:176–83.
81. Higgins ST, Washio Y, Lopez AA, et al. Examining two different schedules of financial incentives for smoking cessation among pregnant women. Prev Med (Baltim) 2014;68:51–7.
82. Higgins ST, Heil SH, Solomon LJ, et al. A pilot study on voucher-based incentives to promote abstinence from cigarette smoking during pregnancy and postpartum. Nicotine Tob Res 2004;6:1015–20.
83. Packer RR, Howell DN, McPherson S, Roll JM. Investigating reinforcer magnitude and reinforcer delay: A contingency management analog study. Exp Clin Psychopharmacol 2012;20:287–92.
84. Kirby KC, Benishek LA, Dugosh KL, Kerwin ME. Substance abuse treatment providers’ beliefs and objections regarding contingency management: Implications for dissemination. Drug Alcohol Depend 2006;85:19–27.
85. Cameron J, Ritter A. Contingency management: Perspectives of Australian service providers. 2007;26:183–9.
86. Hartzler B, Rabun C. Community opioid treatment perspectives on contingency management: Perceived feasibility, effectiveness, and transportability of social and financial incentives. J Subst Abuse Treat 2013;45:242–8.
87. Aletraris L, Shelton JS, Roman PM. Counselor attitudes toward contingency management for substance use disorder: Effectiveness, acceptability, and endorsement of incentives for treatment attendance and abstinence. J Subst Abuse Treat 2015;57:41–8.
88. Budney AJ, Higgins ST, Radonovich KJ, Novy PL. Adding voucher-based incentives to coping skills and motivational enhancement improves outcomes during treatment for marijuana dependence. J Consult Clin Psychol 2000;68:1051–61.
89. Ledgerwood DM, Petry NM. Does contingency management affect motivation to change substance use? Drug Alcohol Depend 2006;83:65–72.
90. Litt MD, Kadden RM, Kabela-Cormier E, Petry NM. Coping skills training and contingency management treatments for marijuana dependence: Exploring mechanisms of behavior change. Addiction 2008;103:638–48.
91. Rash CJ, DePhilippis D, McKay JR, et al. Training workshops positively impact beliefs about contingency management in a nationwide dissemination effort. J Subst Abuse Treat 2013;45:306–12.
92. Benishek LA, Kirby KC, Dugosh KL, Padovano A. Beliefs about the empirical support of drug abuse treatment interventions: A survey of outpatient treatment providers. Drug Alcohol Depend 2010;107:202–8.
93. Petry NM, Alessi SM, Ledgerwood DM. Contingency management delivered by community therapists in outpatient settings. Drug Alcohol Depend 2012;122:86–92.
94. Petry NM, Alessi SM, Ledgerwood DM. A randomized trial of contingency management delivered by community therapists. J Consult Clin Psychol 2012;80:286–98.
95. Hartzler B, Beadnell B, Donovan D. Predictive validity of addiction treatment clinicians’ post-training contingency management skills for subsequent clinical outcomes. J Subst Abuse Treat 2017.
96. Hartzler B. Adapting the Helpful Responses Questionnaire to assess communication skills involved in delivering contingency management: Preliminary psychometrics. J Subst Abuse Treat 2014;55:52–7.
97. Petry NM, Alessi SM, Ledgerwood DM, Sierra S. Psychometric properties of the Contingency Management Competence Scale. Drug Alcohol Depend 2010;109:167–74.
From UConn Health, Farmington, CT.
Abstract
- Objective: To discuss the efficacy and generalizability of contingency management (CM) for the treatment of substance use disorders and design considerations for those considering implementing in clinical settings.
- Methods: Review of the literature.
- Results: CM is an efficacious treatment for substance abuse disorders that is widely generalizable across substance use disorders and patient characteristics. CM can be implemented in a number of treatment programs, including residential and outpatient settings, and it can be administered in both individual and group formats. Abstinence and attendance are the most commonly targeted behaviors in substance abuse treatment settings. Design features, including the selection of target behaviors, delivery methods, and reinforcers, are discussed. Schedule parameters, such as frequency, magnitude, immediacy, and escalation of reinforcement, are associated with overall impact of the CM program and are important considerations for those interested in tailoring CM protocols to their needs.
- Conclusion: CM is an efficacious option that is applicable to most substance abuse treatment patients. A number of demonstrations of real-world implementation have been published and suggest CM can be adapted with success to clinic settings. In adopting CM protocols, clinics should aim for those protocols with established efficacy; however, if adaptations are necessary, careful consideration should be given to modifications to minimize risks of undermining CM’s effects.
Key words: incentives; reinforcement; substance abuse treatment; dissemination; implementation.
Contingency management (CM) is a behavioral intervention that is efficacious in the treatment of substance use disorders (SUDs). CM uses a behavior analytic framework and applies principles of learning theory, particularly operant conditioning theory, to change client behavior(s) [1–5]. In basic terms, operant conditioning principles suggests that whether a behavior continues or not is a function of consequences [6]. Reinforced behaviors are more likely to occur in the future. Substance abuse can be viewed as a behavior maintained by the reinforcing effects of the drug itself [5], including the feel-good aspects of intoxication or relaxation and the amelioration of withdrawal symptoms. CM extends these same principles of to a treatment context, such that reinforcers for abstinent behavior are introduced to compete with the reinforcing effects of continued drug use [5].
In CM’s application to substance abuse treatment, drug-negative samples or treatment attendance are reinforced using tangible incentives with the goal of motivating continued abstinence and/or treatment engagement. When clients demonstrate these target behaviors, they earn incentives in the form of goods or services of value to the client, such as small electronics, gift cards, and toiletries. Despite the promising effects observed in research trials, real-world implementation efforts have not kept pace [7–9]. This review briefly discusses CM’s efficacy and highlights key features for professionals considering adopting this intervention. Demonstration efforts that illustrate how CM can be effectively implemented within the constraints and limitations of non-research, clinical settings are also presented.
Efficacy of CM
CM’s efficacy spans a number of SUDs, including cocaine, opioids, alcohol, nicotine, and marijuana [10–13], making it amenable for treatment of most SUD clinic populations. It generates larger effect sizes than other SUD treatments, including cognitive behavioral therapy [14], and it has been evaluated in a wide range of settings. Large-scale evaluations have been conducted in both intensive outpatient [15] and methadone maintenance [16] settings as part of the National Institute on Drug Abuse Clinical Trials Network, demonstrating consistent benefits of CM when added to treatment as usual. In the first of these 2 studies, Petry et al [15] randomized 415 stimulant users from 1 of 8 intensive outpatient clinics to treatment as usual or treatment as usual plus CM for alcohol and stimulant abstinence. CM participants submitted more substance-negative urine and breath samples, achieved continuous abstinence at significantly higher rates, and had longer treatment retention compared to those receiving treatment as usual. The parallel study [16] focused on stimulant use in clients from methadone maintenance clinics and found similar benefits of CM on stimulant abstinence. Beyond these settings, CM has been applied in a number of other contexts, including drop-in centers [17], vocational rehabilitation [18,19], job-skills training [20], and residential programs [21–23]. In addition, several group-based adaptations have been explored [17,24–27].
CM benefits most clients and generalizes across several demographic variables, including gender [28,29], race [30], housing status [31], and income levels [32–34]. Among clinical characteristics, CM is efficacious for those with co-occurring SUDs [35], other substance use [36], psychiatric disorders [37–39], medical problems [40–42], and history of transactional sex [43].
Design Considerations
Design features, including what behavior will be reinforced and how to do so, are among the first decision points for clinicians interested in implementing CM. One of the advantages of CM is that it has a high degree of flexibility in design, which means that it can be readily tailored to client populations and clinic needs. However, this flexibility can lead clinicians astray from the foundational principles of CM and unknowingly weaken the impact of the program. Below, some key considerations for CM protocol design are reviewed. For additional coverage of these topics, readers are referred to additional articles [1,2] or Petry’s comprehensive book on implementing CM [44]. In this review, published examples of CM’s application in real-world settings are presented, highlighting how CM has been adapted in these clinical efforts.
Target Behaviors
The selection of the target behavior will drive many of the subsequent program design decisions. As such, it is important to identify this feature early. Target behaviors must be achievable, objectively verifiable, and well defined. The most common CM targets are drug abstinence or therapy session attendance. CM has also been used to target other behaviors, such as medication adherence [45,46], treatment-related activities [47,48], and exercise [49–51]. Client self-report of behaviors or vaguely defined behaviors (eg, “good participation”) should be avoided. While some of the decisions related to CM protocols are flexible, the use of objectively verifiable target behaviors is a core feature that should not be neglected. If the behavior of interest cannot be objectively verified, an alternate behavior should be chosen.
Selection of the target behavior is often considered in hand with defining which population is eligible to participate in the CM program. Client characteristics are often forefront in this decision, but clinic-driven logistical issues or unmet needs may also play a role. A real-world example of this decision process is evident in the nationwide rollout of CM among the intensive outpatient programs within the Veterans Administration (VA). The VA identified a treatment need for those with stimulant use disorders, as this group did not have efficacious pharmacotherapy options available that targeted stimulant use. As such, the VA applied CM to patients with a focus on stimulant abstinence as the behavioral target [52]. For others, the decision may revolve around addressing underutilization of specific treatment resources (eg, outpatient groups, vocational rehabilitation) [53–56] or treatment needs among certain subgroups of clients, such as adolescents [57–59].
For abstinence targets, clinics would need to select one or more specific substances as the focus of the CM program. In general, targeting a single substance rather than multiple substances is more effective [10,13], is more straightforward for clients to understand, and allows more clients to access the reinforcers. Exposure to the reinforcers is necessary for CM to work; thus, setting a goal that is achievable for most clients should be a priority. Requiring abstinence from multiple substances means that some clients may never experience the reinforcer and thus cannot benefit from its effects at all. Some clinicians or administrators may initially have reservations about reinforcing single drug abstinence in the event that other drug use continues. However, targeting a single substance for reinforcement often results in reduced use of other substances [60]. Clinicians may find that this makes intuitive sense; a client with cocaine use disorder who is trying to maintain cocaine abstinence over a long period is likely to avoid using alcohol or other substances that might lead to relapse. For abstinence, objective verification through urine or breath specimens using tests that include validity checks is relatively straightforward.
Attendance is a popular target for clinics in part because it does not require additional staff time to collect specimen samples and it was the most commonly reported target behavior in samples of SUD providers who use incentives [61,62]. Objective verification of attendance is usually via a staff member, but expectations must be clear to both parties. Clinics should consider potential problems that may arise (eg, arriving late, leaving group early, excused absences) and carefully define and communicate expectations for the CM program. Piloting [19] the CM program with a small group of clients may be valuable in trouble-shooting challenges before wider implementation.
In a recent study [55], clients earned reinforcers for attending clinician-led group counseling sessions and/or the in-clinic patient-led Methadone Anonymous (MA) groups. This non-research, clinical effort addressed historically poor therapy attendance at the clinic, and attendance rates were compared before, during, and after the CM program. CM increased attendance to both groups in the short-term after implementation, but effects were more robust for the MA groups in which increased attendance persisted 3 months following the withdrawal of the contingencies. Overall effects of this program were modest, but they are notable given the use of an ultra-low cost approach.
Delivery Methods
The majority of CM studies used voucher or prize-based methods. Head-to-head comparisons suggest that they are comparable in efficacy [63–65], and each has advantages and disadvantages that may make one option more appealing for a given clinic. Voucher programs are generally straightforward to administer. Clients earn vouchers for each instance of the target behavior. The value of the vouchers typically increases with consecutive performance. The schedule used in the influential Higgins et al studies [66,67] started at $2.50 for the first cocaine-negative sample and increased by $0.50 for each subsequent consecutive cocaine-negative sample. Earned vouchers are exchanged for goods or services selected by the client, increasing the likelihood that the selected items will be highly desirable and allowing for a wide range of client preferences. Clients appear to prefer this approach when given a choice between set schedules or those that introduce an element of chance (ie, prize-based CM, discussed below) [68]. However, voucher programs can be costly (~$1000 per client over 12 weeks) and may require more staff time to fulfill individual requests for specific items. However, staff burden related to shopping can be reduced by limiting these individual requests and using an on-site stocked cabinet of goods similar to prize-CM programs.
Prize-based CM is similar but introduces probabilistic earnings and variability in prize magnitude. Rather than earning vouchers, clients earn draws from a fishbowl for each instance of the target behavior, again typically in an escalating manner. For example, a client may earn one draw from the fishbowl for the first cocaine-negative sample, 2 draws for the second consecutive negative sample, 3 draws for the third, and so on. A typical fishbowl is composed of 500 slips, some noting prizes and some having no prize value. Typically, half the slips in the bowl are non-monetary “good jobs.” The remaining half are small prizes worth about $1 in value (eg, food coupons, bus tokens, small toiletries), large prizes worth about $20 in value (eg, small electronics, gift certificates), and one slip is the jumbo prize worth about $100. When a client draws a winning slip, they select a prize from that category (ie, small, large, jumbo) from an onsite, stocked cabinet. Due to the probabilistic feature of prize-based CM, overall costs of the program can be substantially lower than typical voucher programs, with average maximum expected earnings ranging $250 t $450 per client over a 12-week treatment period [15,16,65,69]. Advantages of this method include potentially lower costs and minimal shopping demands (a once-monthly shopping trip to restock the cabinet will usually suffice) while maintaining comparable efficacy. Relative to voucher programs, prize-based CM involves additional administration time to allow for drawing slips from the fishbowl, which can be compounded when multiple clients want to draw at the same time such as in a group setting. Many of the group-based CM adaptations address this issue by limiting the number of clients who can draw for prizes in a given group or by limiting the number of draws per client [25,27,54].
Reinforcers
Regardless of whether selecting voucher or prize CM, reinforcers are critically important to the success of the program. Reinforcers must be desirable. One of the quickest ways to undermine a CM program is lack of variety or undesirable reinforcers. If stocking a cabinet with prizes onsite, care should be taken to have numerous options within each of the small and large prize categories that are appealing to a wide range of clients. Since a client who is consistently earning draws will choose prizes often, it is imperative to include enough variety so that even these clients find desirable items each time they select a prize. Clients should be asked regularly if they have suggestions for prizes; one program [54] found suggestion boxes useful for encouraging clients to voice their preferences. Donations can be solicited from local businesses to reduce costs [53], and low-cost but high-value options, such as clinic privileges, can also be explored. Petry [1,44] provides some suggestions of the latter, and Amass and Kamien [70] describe their successful strategies to fund and sustain a clinic-based CM program through community donations. Some clinics may already have tangible goods, such as gas or metro cards, that are offered to clients based on need rather than behavior [53]. These existing resources might be redirected to a CM program, in which these goods are contingent on abstinence or attendance, if appropriate.
Schedule Parameters
Once the target behavior, client population, and CM delivery methods are selected, the next step is to design the reinforcement schedule. The following schedule parameters apply to both voucher and prize-based CM systems. The more closely a clinical program adheres to the parameters of effective protocols, the more likely the program is to generate comparable outcomes. If there is a parameter or design feature that is incompatible with clinic needs, modifications can be introduced. However, each deviation away from the ideal has a chance of undermining the success of the CM program. Any changes and their potential impacts should considered carefully, and consultation with a CM expert may aid in the development of successful and efficacious clinic-based protocols. Of note, a meta-analysis [13] of CM studies found that researcher involvement in the planning and design of CM programs is associated with larger treatment effects. CM researchers are especially attuned to the potential impacts and pitfalls associated with modifying CM protocols, and they can be valuable resources for clinics interested in tailoring a CM program to their specific needs. Several examples of clinical demonstration projects that used researcher input are available [19,53,71].
Magnitude
Incentive magnitude was directly related to the size of treatment effects in a meta-analysis [11] of CM studies. Although not all studies find significant differences in outcomes related to magnitude [65,72], the bulk of evidence suggests magnitude is an important parameter and is related to effect size for both voucher [73–75] and prize-based CM [69,76] systems. Thus, although clinics may have restrictive budgets, severely undercutting the magnitude of rewards is not usually the solution as it can undermine treatment effects [76]. Donations can reduce overall costs [53,57,70], and other protocol features discussed below, such as capping the amount of reinforcement available, can reduce the overall magnitude available per patient.
Another approach, used in group-based CM, limits the number of patients who earn prizes per week [25,27]. For example, in a 2011 study by Petry et al, clients added slips with their name to a bowl for attendance and negative samples. Once all names were collected in the bowl, the group leader would pull a specified number of slips (eg, 3 slips per group). These individuals were eligible to draw from the prize bowl for prizes. This approach was associated with longer durations of consecutive abstinence and better treatment attendance relative to treatment as usual. However, clinics can control the overall program costs by limiting the number of patients eligible for prizes.
Frequency
Frequent reinforcement opportunities are ideal, and more frequent assessment is associated with larger treatment effects [10]. However, a number of factors, including which target behavior is selected and logistical issues specific to the clinic such as when groups meet, will play a role in determining the frequency of CM sessions. For abstinence targets, the substance targeted and type of test will largely determine the frequency of CM sessions. The goal would be to test at a frequency that would detect most or nearly all instances of use. For cocaine or opioids, this equates to testing 2 to 3 times weekly. Breath samples for alcohol or cigarette smoking would necessitate testing daily or multiple times per day to detect most instances of use because these tests have short windows of detection. CM protocols based on these breath tests have often had daily or twice daily CM sessions [77,78]; technological adaptations [77,79,80] or residential settings [21,23] may reduce burden to the client for assessment of these substances. Tapering the number of breath tests over time or transitioning from daily breath tests to once or twice weekly urine testing after abstinence is established is another approach [81,82].
Marijuana, on the other hand, poses difficulties because it is detectable in urine samples for up to 2 weeks following use. If relying solely on urine results for reinforcement, clients may not test negative for several days or weeks after last use, resulting in a delay of reinforcement. To address this issue, some CM programs targeting marijuana abstinence initially reinforce attendance in the first 2 weeks and then transition to reinforcing marijuana-negative drug samples for the remainder of the treatment period [48].
In general, more frequent CM sessions can translate to higher costs; however, infrequent reinforcement (ie, less than weekly) is not as effective [45]. In real-world applications, clinics often need to balance feasibility and costs with the ideal CM schedule. In abstinence-based CM, this compromise may result in a testing schedule that may not capture all instances of use. For example, while thrice-weekly testing may be ideal for cocaine or opioids, a twice-weekly schedule may be selected because it lowers costs and is more consistent with clinic schedules.
Immediacy
In general, clinics should aim to deliver reinforcement as immediately as possible, as delays between the target behavior and reinforcement are associated with decreased treatment effects [10,11,83]. For drug abstinence, onsite urine testing systems that provide immediate results are preferred over sending samples for laboratory testing. Clinics that do not have access to or who cannot afford specimen testing that allows onsite collection and immediate results might consider other options for target behaviors, such as attendance.
Immediacy of reinforcement is also important when targeting attendance. One clinic [53] implemented a program that offered a $50 incentive if clients attended 1 month of group therapy sessions. This approach was not effective and no clients earned the incentive for several months. After consultation, the clinic revised the incentive program to a daily drawing for attendance using the fishbowl method, thereby decreasing the delay between the behavior and its consequence. This example illustrates not only problems with delayed reinforcement but also the common mistake of setting expectations for the target behavior too high. Attending a month of group therapy sessions is a high bar that few patients will achieve, resulting in a system that mostly rewards those already doing well [19]. In contrast, attending a single group session in order to earn reinforcers is a reachable goal and increases the likelihood that more clients are exposed to the reinforcers. These small steps (ie, attending a single group or submitting a single drug negative urine) encourage initiation of the behavior(s) targeted. Other features, such as escalation (discussed next), aim to sustain the behavior over time.
Escalation
Escalation involves increasing the amount of reinforcement for each consecutive target behavior. In the voucher programs, the amount earned per negative sample may increase for each consecutive negative sample (eg, $2.50 for the first negative sample, $3.00 for the second, $3.50 for the third, and so on). For prize-based programs, the number of draws escalates with consecutive performance (eg, 1 draw for the first group attended, 2 draws for the second, 3 for the third, and so on). Protocols that include escalation generate larger effects than those that have a set, flat incentive amount even when total costs are the same across comparison conditions [73].
Escalating schedules usually include a reset feature. Following a positive or refused sample or unexcused absence, the amount earned for the next negative sample is reduced to the initial amount and begins escalating anew with consecutive negative samples. Some schedules allow for a rapid reset in which after a specified period of time or consecutive performance, the value jumps to the value achieved when the relapse occurred [66].
Despite its consistent inclusion in CM protocols from randomized clinical trials, our data [61] suggest that more than half of providers using incentives in treatment as part of a clinical effort do not use escalating reinforcers. Escalating schedules require more careful tracking of client progress, possibly contributing to lower uptake of this design feature in clinical practice. Development of simple tracking forms can minimize this challenge.
Another drawback of escalation pertaining to prize-based CM is that escalating schedules can affect the duration of CM sessions when clients are drawing a large number of slips each session and escalation can increase costs of the overall program. Capping the number of draws will help mitigate both issues. For example, once a client reaches 10 draws for group attendance, they continue earning 10 draws for each consecutive session attended with no further escalation.
Putting It All Together
CM sessions can be conducted as stand-alone sessions or incorporated into group or individual therapy sessions. Many clinicians will find the latter approach sets a positive tone for the therapy session given CM's focus on what the client is doing well. Starting the treatment session with the CM component often naturally leads into a discussion of relevant therapeutic issues, such as effective coping, slips, or triggers. The CM session length can be variable, but it is typically under 10 minutes. Thus, the CM component need not dominate the clinical session or content. CM sessions for abstinence are scheduled according to a set schedule (eg, Mondays and Thursdays) and often coincide with other treatment aspects (eg, before or after group therapy on Mondays and Thursdays). CM sessions for attendance also generally follow a set schedule (eg, client expected to attend Monday and Wednesday group therapy sessions). The duration of the CM protocol can also vary, with most clinical trials ranging from 12 to 24 weeks. Very short durations are unlikely to produce lasting behavior change, particularly with complex behaviors such as abstinence. Petry [44] recommends no less than 8 weeks duration and a maximum duration of 24 weeks.
As discussed, CM offers many opportunities for tailoring to optimize its fit within the existing structure of clinics. However, this flexibility must be viewed together with an understanding of the principles that impact CM's efficacy. Specific recommendations for CM protocol development will depend on the behavior targeted, the delivery methods, and format (eg, individual versus group settings). For these reasons, consultation with a CM expert is ideal. However, some general guidelines for developing a CM program that incorporate the principles discussed above include an 8- to 12-week program that (1) provides sufficient magnitude to compete with the behavior you are attempting to change, (2) offers frequent opportunities for reinforcement (eg, 2-3 times/wk for opioids or stimulant abstinence, 1-2 times/week for attendance targets; not less than weekly for most behaviors), (3) delivers the reinforcement immediately or very close in time with the behavior (eg, reinforce attendance at the beginning of the group, use onsite urine testing and reinforce immediately after testing), and (4) incorporates escalating and reset features into the schedule.
Clinician Training and Supervision
Training in CM is an important part of the implementation process. Studies [62,84–87] have identified a number of perceived barriers to and negative beliefs about CM, including philosophical and logistical concerns. Tangible incentives, the core of most CM protocols, are generally viewed less favorably than social or nonspecified incentives [84,86,87]. Philosophical concerns relate to CM’s inability to address the underlying causes of addiction, that it does not address multiple behaviors, and that it may undermine internal motivation for sobriety [62,84]. An additional objection relates to paying someone to do what they should do on their own [86]. Logistical and practical concerns often represent implementation barriers such as costs and access to training and supervision, but they also reflect concern for what happens when contingencies are withdrawn, that clients may sell or trade prizes for drugs, and worries that CM’s evidence does not generalize to clinic populations [62].
Many of these beliefs reflect a limited understanding of CM, and addressing these misperceptions is a first step toward reducing resistance to implementation efforts. For example, a substantial body of literature points to CM’s wide generalizability across a range of characteristics, clients that sell or trade prizes for drugs are likely to disrupt their chain of negative samples or attendance, and most studies do not find negative impacts of CM on intrinsic motivation [88–90]. Fortunately, CM training appears to be an effective way to address negative beliefs. In the VA implementation effort [52], training workshops decreased perceived barriers and increased positive impressions of CM [91]. In other training efforts, brief educational materials were effective in changing perceptions of CM’s efficacy [92].
Beyond initial training, supervision of CM delivery is likely to be necessary [93,94]. Clinician skill in delivering CM is related to client outcomes [93,95] and relatively simple adherence measures are available for monitoring [96,97]. However, the best methods for training and supervision of CM have yet to be established. The VA initiative was developed in consultation with CM experts and employed ongoing phone consultation following initial training workshops [52,91]. This approach represented significant investment by the VA toward staff training and CM protocol development that may not be achievable for individual clinics. As attention to CM’s dissemination and implementation has grown, some free resources have been developed. Promoting Awareness of Motivational Incentives (PAMI; www.bettertxoutcomes.org/bettertxoutcomes/PAMI.html) is a collaborative initiative sponsored by the National Institute of Drug Abuse and the Substance Abuse and Mental Health Services Administration. It offers free resources and training materials.
Conclusion
Overall, CM is a highly efficacious treatment for SUDs that generalizes to most clients. Despite a robust evidence base, CM’s implementation in clinical settings lags behind other empirically supported treatments [92]. At least in part, CM’s costs, which include not only staff training and adherence monitoring (as with other treatments), but also costs of the incentives themselves, may contribute to slow uptake in clinical settings. Clinics often do not have the resources available for CM within their operating budgets. However, a growing number of projects [19,52,53,55–57,70,71] illustrate CM implementation within routine clinical care, and increased revenue from improved attendance to treatment groups may be one mechanism through which to fund a CM program [54,56,57]. These projects are valuable not only for demonstrating that CM can be efficacious outside the research setting, but also for highlighting how implementation barriers can be overcome. Continued efforts of this nature are likely to be particularly valuable for clinicians and administrators considering adopting CM within clinical settings.
From UConn Health, Farmington, CT.
Abstract
- Objective: To discuss the efficacy and generalizability of contingency management (CM) for the treatment of substance use disorders and design considerations for those considering implementing in clinical settings.
- Methods: Review of the literature.
- Results: CM is an efficacious treatment for substance abuse disorders that is widely generalizable across substance use disorders and patient characteristics. CM can be implemented in a number of treatment programs, including residential and outpatient settings, and it can be administered in both individual and group formats. Abstinence and attendance are the most commonly targeted behaviors in substance abuse treatment settings. Design features, including the selection of target behaviors, delivery methods, and reinforcers, are discussed. Schedule parameters, such as frequency, magnitude, immediacy, and escalation of reinforcement, are associated with overall impact of the CM program and are important considerations for those interested in tailoring CM protocols to their needs.
- Conclusion: CM is an efficacious option that is applicable to most substance abuse treatment patients. A number of demonstrations of real-world implementation have been published and suggest CM can be adapted with success to clinic settings. In adopting CM protocols, clinics should aim for those protocols with established efficacy; however, if adaptations are necessary, careful consideration should be given to modifications to minimize risks of undermining CM’s effects.
Key words: incentives; reinforcement; substance abuse treatment; dissemination; implementation.
Contingency management (CM) is a behavioral intervention that is efficacious in the treatment of substance use disorders (SUDs). CM uses a behavior analytic framework and applies principles of learning theory, particularly operant conditioning theory, to change client behavior(s) [1–5]. In basic terms, operant conditioning principles suggests that whether a behavior continues or not is a function of consequences [6]. Reinforced behaviors are more likely to occur in the future. Substance abuse can be viewed as a behavior maintained by the reinforcing effects of the drug itself [5], including the feel-good aspects of intoxication or relaxation and the amelioration of withdrawal symptoms. CM extends these same principles of to a treatment context, such that reinforcers for abstinent behavior are introduced to compete with the reinforcing effects of continued drug use [5].
In CM’s application to substance abuse treatment, drug-negative samples or treatment attendance are reinforced using tangible incentives with the goal of motivating continued abstinence and/or treatment engagement. When clients demonstrate these target behaviors, they earn incentives in the form of goods or services of value to the client, such as small electronics, gift cards, and toiletries. Despite the promising effects observed in research trials, real-world implementation efforts have not kept pace [7–9]. This review briefly discusses CM’s efficacy and highlights key features for professionals considering adopting this intervention. Demonstration efforts that illustrate how CM can be effectively implemented within the constraints and limitations of non-research, clinical settings are also presented.
Efficacy of CM
CM’s efficacy spans a number of SUDs, including cocaine, opioids, alcohol, nicotine, and marijuana [10–13], making it amenable for treatment of most SUD clinic populations. It generates larger effect sizes than other SUD treatments, including cognitive behavioral therapy [14], and it has been evaluated in a wide range of settings. Large-scale evaluations have been conducted in both intensive outpatient [15] and methadone maintenance [16] settings as part of the National Institute on Drug Abuse Clinical Trials Network, demonstrating consistent benefits of CM when added to treatment as usual. In the first of these 2 studies, Petry et al [15] randomized 415 stimulant users from 1 of 8 intensive outpatient clinics to treatment as usual or treatment as usual plus CM for alcohol and stimulant abstinence. CM participants submitted more substance-negative urine and breath samples, achieved continuous abstinence at significantly higher rates, and had longer treatment retention compared to those receiving treatment as usual. The parallel study [16] focused on stimulant use in clients from methadone maintenance clinics and found similar benefits of CM on stimulant abstinence. Beyond these settings, CM has been applied in a number of other contexts, including drop-in centers [17], vocational rehabilitation [18,19], job-skills training [20], and residential programs [21–23]. In addition, several group-based adaptations have been explored [17,24–27].
CM benefits most clients and generalizes across several demographic variables, including gender [28,29], race [30], housing status [31], and income levels [32–34]. Among clinical characteristics, CM is efficacious for those with co-occurring SUDs [35], other substance use [36], psychiatric disorders [37–39], medical problems [40–42], and history of transactional sex [43].
Design Considerations
Design features, including what behavior will be reinforced and how to do so, are among the first decision points for clinicians interested in implementing CM. One of the advantages of CM is that it has a high degree of flexibility in design, which means that it can be readily tailored to client populations and clinic needs. However, this flexibility can lead clinicians astray from the foundational principles of CM and unknowingly weaken the impact of the program. Below, some key considerations for CM protocol design are reviewed. For additional coverage of these topics, readers are referred to additional articles [1,2] or Petry’s comprehensive book on implementing CM [44]. In this review, published examples of CM’s application in real-world settings are presented, highlighting how CM has been adapted in these clinical efforts.
Target Behaviors
The selection of the target behavior will drive many of the subsequent program design decisions. As such, it is important to identify this feature early. Target behaviors must be achievable, objectively verifiable, and well defined. The most common CM targets are drug abstinence or therapy session attendance. CM has also been used to target other behaviors, such as medication adherence [45,46], treatment-related activities [47,48], and exercise [49–51]. Client self-report of behaviors or vaguely defined behaviors (eg, “good participation”) should be avoided. While some of the decisions related to CM protocols are flexible, the use of objectively verifiable target behaviors is a core feature that should not be neglected. If the behavior of interest cannot be objectively verified, an alternate behavior should be chosen.
Selection of the target behavior is often considered in hand with defining which population is eligible to participate in the CM program. Client characteristics are often forefront in this decision, but clinic-driven logistical issues or unmet needs may also play a role. A real-world example of this decision process is evident in the nationwide rollout of CM among the intensive outpatient programs within the Veterans Administration (VA). The VA identified a treatment need for those with stimulant use disorders, as this group did not have efficacious pharmacotherapy options available that targeted stimulant use. As such, the VA applied CM to patients with a focus on stimulant abstinence as the behavioral target [52]. For others, the decision may revolve around addressing underutilization of specific treatment resources (eg, outpatient groups, vocational rehabilitation) [53–56] or treatment needs among certain subgroups of clients, such as adolescents [57–59].
For abstinence targets, clinics would need to select one or more specific substances as the focus of the CM program. In general, targeting a single substance rather than multiple substances is more effective [10,13], is more straightforward for clients to understand, and allows more clients to access the reinforcers. Exposure to the reinforcers is necessary for CM to work; thus, setting a goal that is achievable for most clients should be a priority. Requiring abstinence from multiple substances means that some clients may never experience the reinforcer and thus cannot benefit from its effects at all. Some clinicians or administrators may initially have reservations about reinforcing single drug abstinence in the event that other drug use continues. However, targeting a single substance for reinforcement often results in reduced use of other substances [60]. Clinicians may find that this makes intuitive sense; a client with cocaine use disorder who is trying to maintain cocaine abstinence over a long period is likely to avoid using alcohol or other substances that might lead to relapse. For abstinence, objective verification through urine or breath specimens using tests that include validity checks is relatively straightforward.
Attendance is a popular target for clinics in part because it does not require additional staff time to collect specimen samples and it was the most commonly reported target behavior in samples of SUD providers who use incentives [61,62]. Objective verification of attendance is usually via a staff member, but expectations must be clear to both parties. Clinics should consider potential problems that may arise (eg, arriving late, leaving group early, excused absences) and carefully define and communicate expectations for the CM program. Piloting [19] the CM program with a small group of clients may be valuable in trouble-shooting challenges before wider implementation.
In a recent study [55], clients earned reinforcers for attending clinician-led group counseling sessions and/or the in-clinic patient-led Methadone Anonymous (MA) groups. This non-research, clinical effort addressed historically poor therapy attendance at the clinic, and attendance rates were compared before, during, and after the CM program. CM increased attendance to both groups in the short-term after implementation, but effects were more robust for the MA groups in which increased attendance persisted 3 months following the withdrawal of the contingencies. Overall effects of this program were modest, but they are notable given the use of an ultra-low cost approach.
Delivery Methods
The majority of CM studies used voucher or prize-based methods. Head-to-head comparisons suggest that they are comparable in efficacy [63–65], and each has advantages and disadvantages that may make one option more appealing for a given clinic. Voucher programs are generally straightforward to administer. Clients earn vouchers for each instance of the target behavior. The value of the vouchers typically increases with consecutive performance. The schedule used in the influential Higgins et al studies [66,67] started at $2.50 for the first cocaine-negative sample and increased by $0.50 for each subsequent consecutive cocaine-negative sample. Earned vouchers are exchanged for goods or services selected by the client, increasing the likelihood that the selected items will be highly desirable and allowing for a wide range of client preferences. Clients appear to prefer this approach when given a choice between set schedules or those that introduce an element of chance (ie, prize-based CM, discussed below) [68]. However, voucher programs can be costly (~$1000 per client over 12 weeks) and may require more staff time to fulfill individual requests for specific items. However, staff burden related to shopping can be reduced by limiting these individual requests and using an on-site stocked cabinet of goods similar to prize-CM programs.
Prize-based CM is similar but introduces probabilistic earnings and variability in prize magnitude. Rather than earning vouchers, clients earn draws from a fishbowl for each instance of the target behavior, again typically in an escalating manner. For example, a client may earn one draw from the fishbowl for the first cocaine-negative sample, 2 draws for the second consecutive negative sample, 3 draws for the third, and so on. A typical fishbowl is composed of 500 slips, some noting prizes and some having no prize value. Typically, half the slips in the bowl are non-monetary “good jobs.” The remaining half are small prizes worth about $1 in value (eg, food coupons, bus tokens, small toiletries), large prizes worth about $20 in value (eg, small electronics, gift certificates), and one slip is the jumbo prize worth about $100. When a client draws a winning slip, they select a prize from that category (ie, small, large, jumbo) from an onsite, stocked cabinet. Due to the probabilistic feature of prize-based CM, overall costs of the program can be substantially lower than typical voucher programs, with average maximum expected earnings ranging $250 t $450 per client over a 12-week treatment period [15,16,65,69]. Advantages of this method include potentially lower costs and minimal shopping demands (a once-monthly shopping trip to restock the cabinet will usually suffice) while maintaining comparable efficacy. Relative to voucher programs, prize-based CM involves additional administration time to allow for drawing slips from the fishbowl, which can be compounded when multiple clients want to draw at the same time such as in a group setting. Many of the group-based CM adaptations address this issue by limiting the number of clients who can draw for prizes in a given group or by limiting the number of draws per client [25,27,54].
Reinforcers
Regardless of whether selecting voucher or prize CM, reinforcers are critically important to the success of the program. Reinforcers must be desirable. One of the quickest ways to undermine a CM program is lack of variety or undesirable reinforcers. If stocking a cabinet with prizes onsite, care should be taken to have numerous options within each of the small and large prize categories that are appealing to a wide range of clients. Since a client who is consistently earning draws will choose prizes often, it is imperative to include enough variety so that even these clients find desirable items each time they select a prize. Clients should be asked regularly if they have suggestions for prizes; one program [54] found suggestion boxes useful for encouraging clients to voice their preferences. Donations can be solicited from local businesses to reduce costs [53], and low-cost but high-value options, such as clinic privileges, can also be explored. Petry [1,44] provides some suggestions of the latter, and Amass and Kamien [70] describe their successful strategies to fund and sustain a clinic-based CM program through community donations. Some clinics may already have tangible goods, such as gas or metro cards, that are offered to clients based on need rather than behavior [53]. These existing resources might be redirected to a CM program, in which these goods are contingent on abstinence or attendance, if appropriate.
Schedule Parameters
Once the target behavior, client population, and CM delivery methods are selected, the next step is to design the reinforcement schedule. The following schedule parameters apply to both voucher and prize-based CM systems. The more closely a clinical program adheres to the parameters of effective protocols, the more likely the program is to generate comparable outcomes. If there is a parameter or design feature that is incompatible with clinic needs, modifications can be introduced. However, each deviation away from the ideal has a chance of undermining the success of the CM program. Any changes and their potential impacts should considered carefully, and consultation with a CM expert may aid in the development of successful and efficacious clinic-based protocols. Of note, a meta-analysis [13] of CM studies found that researcher involvement in the planning and design of CM programs is associated with larger treatment effects. CM researchers are especially attuned to the potential impacts and pitfalls associated with modifying CM protocols, and they can be valuable resources for clinics interested in tailoring a CM program to their specific needs. Several examples of clinical demonstration projects that used researcher input are available [19,53,71].
Magnitude
Incentive magnitude was directly related to the size of treatment effects in a meta-analysis [11] of CM studies. Although not all studies find significant differences in outcomes related to magnitude [65,72], the bulk of evidence suggests magnitude is an important parameter and is related to effect size for both voucher [73–75] and prize-based CM [69,76] systems. Thus, although clinics may have restrictive budgets, severely undercutting the magnitude of rewards is not usually the solution as it can undermine treatment effects [76]. Donations can reduce overall costs [53,57,70], and other protocol features discussed below, such as capping the amount of reinforcement available, can reduce the overall magnitude available per patient.
Another approach, used in group-based CM, limits the number of patients who earn prizes per week [25,27]. For example, in a 2011 study by Petry et al, clients added slips with their name to a bowl for attendance and negative samples. Once all names were collected in the bowl, the group leader would pull a specified number of slips (eg, 3 slips per group). These individuals were eligible to draw from the prize bowl for prizes. This approach was associated with longer durations of consecutive abstinence and better treatment attendance relative to treatment as usual. However, clinics can control the overall program costs by limiting the number of patients eligible for prizes.
Frequency
Frequent reinforcement opportunities are ideal, and more frequent assessment is associated with larger treatment effects [10]. However, a number of factors, including which target behavior is selected and logistical issues specific to the clinic such as when groups meet, will play a role in determining the frequency of CM sessions. For abstinence targets, the substance targeted and type of test will largely determine the frequency of CM sessions. The goal would be to test at a frequency that would detect most or nearly all instances of use. For cocaine or opioids, this equates to testing 2 to 3 times weekly. Breath samples for alcohol or cigarette smoking would necessitate testing daily or multiple times per day to detect most instances of use because these tests have short windows of detection. CM protocols based on these breath tests have often had daily or twice daily CM sessions [77,78]; technological adaptations [77,79,80] or residential settings [21,23] may reduce burden to the client for assessment of these substances. Tapering the number of breath tests over time or transitioning from daily breath tests to once or twice weekly urine testing after abstinence is established is another approach [81,82].
Marijuana, on the other hand, poses difficulties because it is detectable in urine samples for up to 2 weeks following use. If relying solely on urine results for reinforcement, clients may not test negative for several days or weeks after last use, resulting in a delay of reinforcement. To address this issue, some CM programs targeting marijuana abstinence initially reinforce attendance in the first 2 weeks and then transition to reinforcing marijuana-negative drug samples for the remainder of the treatment period [48].
In general, more frequent CM sessions can translate to higher costs; however, infrequent reinforcement (ie, less than weekly) is not as effective [45]. In real-world applications, clinics often need to balance feasibility and costs with the ideal CM schedule. In abstinence-based CM, this compromise may result in a testing schedule that may not capture all instances of use. For example, while thrice-weekly testing may be ideal for cocaine or opioids, a twice-weekly schedule may be selected because it lowers costs and is more consistent with clinic schedules.
Immediacy
In general, clinics should aim to deliver reinforcement as immediately as possible, as delays between the target behavior and reinforcement are associated with decreased treatment effects [10,11,83]. For drug abstinence, onsite urine testing systems that provide immediate results are preferred over sending samples for laboratory testing. Clinics that do not have access to or who cannot afford specimen testing that allows onsite collection and immediate results might consider other options for target behaviors, such as attendance.
Immediacy of reinforcement is also important when targeting attendance. One clinic [53] implemented a program that offered a $50 incentive if clients attended 1 month of group therapy sessions. This approach was not effective and no clients earned the incentive for several months. After consultation, the clinic revised the incentive program to a daily drawing for attendance using the fishbowl method, thereby decreasing the delay between the behavior and its consequence. This example illustrates not only problems with delayed reinforcement but also the common mistake of setting expectations for the target behavior too high. Attending a month of group therapy sessions is a high bar that few patients will achieve, resulting in a system that mostly rewards those already doing well [19]. In contrast, attending a single group session in order to earn reinforcers is a reachable goal and increases the likelihood that more clients are exposed to the reinforcers. These small steps (ie, attending a single group or submitting a single drug negative urine) encourage initiation of the behavior(s) targeted. Other features, such as escalation (discussed next), aim to sustain the behavior over time.
Escalation
Escalation involves increasing the amount of reinforcement for each consecutive target behavior. In the voucher programs, the amount earned per negative sample may increase for each consecutive negative sample (eg, $2.50 for the first negative sample, $3.00 for the second, $3.50 for the third, and so on). For prize-based programs, the number of draws escalates with consecutive performance (eg, 1 draw for the first group attended, 2 draws for the second, 3 for the third, and so on). Protocols that include escalation generate larger effects than those that have a set, flat incentive amount even when total costs are the same across comparison conditions [73].
Escalating schedules usually include a reset feature. Following a positive or refused sample or unexcused absence, the amount earned for the next negative sample is reduced to the initial amount and begins escalating anew with consecutive negative samples. Some schedules allow for a rapid reset in which after a specified period of time or consecutive performance, the value jumps to the value achieved when the relapse occurred [66].
Despite its consistent inclusion in CM protocols from randomized clinical trials, our data [61] suggest that more than half of providers using incentives in treatment as part of a clinical effort do not use escalating reinforcers. Escalating schedules require more careful tracking of client progress, possibly contributing to lower uptake of this design feature in clinical practice. Development of simple tracking forms can minimize this challenge.
Another drawback of escalation pertaining to prize-based CM is that escalating schedules can affect the duration of CM sessions when clients are drawing a large number of slips each session and escalation can increase costs of the overall program. Capping the number of draws will help mitigate both issues. For example, once a client reaches 10 draws for group attendance, they continue earning 10 draws for each consecutive session attended with no further escalation.
Putting It All Together
CM sessions can be conducted as stand-alone sessions or incorporated into group or individual therapy sessions. Many clinicians will find the latter approach sets a positive tone for the therapy session given CM's focus on what the client is doing well. Starting the treatment session with the CM component often naturally leads into a discussion of relevant therapeutic issues, such as effective coping, slips, or triggers. The CM session length can be variable, but it is typically under 10 minutes. Thus, the CM component need not dominate the clinical session or content. CM sessions for abstinence are scheduled according to a set schedule (eg, Mondays and Thursdays) and often coincide with other treatment aspects (eg, before or after group therapy on Mondays and Thursdays). CM sessions for attendance also generally follow a set schedule (eg, client expected to attend Monday and Wednesday group therapy sessions). The duration of the CM protocol can also vary, with most clinical trials ranging from 12 to 24 weeks. Very short durations are unlikely to produce lasting behavior change, particularly with complex behaviors such as abstinence. Petry [44] recommends no less than 8 weeks duration and a maximum duration of 24 weeks.
As discussed, CM offers many opportunities for tailoring to optimize its fit within the existing structure of clinics. However, this flexibility must be viewed together with an understanding of the principles that impact CM's efficacy. Specific recommendations for CM protocol development will depend on the behavior targeted, the delivery methods, and format (eg, individual versus group settings). For these reasons, consultation with a CM expert is ideal. However, some general guidelines for developing a CM program that incorporate the principles discussed above include an 8- to 12-week program that (1) provides sufficient magnitude to compete with the behavior you are attempting to change, (2) offers frequent opportunities for reinforcement (eg, 2-3 times/wk for opioids or stimulant abstinence, 1-2 times/week for attendance targets; not less than weekly for most behaviors), (3) delivers the reinforcement immediately or very close in time with the behavior (eg, reinforce attendance at the beginning of the group, use onsite urine testing and reinforce immediately after testing), and (4) incorporates escalating and reset features into the schedule.
Clinician Training and Supervision
Training in CM is an important part of the implementation process. Studies [62,84–87] have identified a number of perceived barriers to and negative beliefs about CM, including philosophical and logistical concerns. Tangible incentives, the core of most CM protocols, are generally viewed less favorably than social or nonspecified incentives [84,86,87]. Philosophical concerns relate to CM’s inability to address the underlying causes of addiction, that it does not address multiple behaviors, and that it may undermine internal motivation for sobriety [62,84]. An additional objection relates to paying someone to do what they should do on their own [86]. Logistical and practical concerns often represent implementation barriers such as costs and access to training and supervision, but they also reflect concern for what happens when contingencies are withdrawn, that clients may sell or trade prizes for drugs, and worries that CM’s evidence does not generalize to clinic populations [62].
Many of these beliefs reflect a limited understanding of CM, and addressing these misperceptions is a first step toward reducing resistance to implementation efforts. For example, a substantial body of literature points to CM’s wide generalizability across a range of characteristics, clients that sell or trade prizes for drugs are likely to disrupt their chain of negative samples or attendance, and most studies do not find negative impacts of CM on intrinsic motivation [88–90]. Fortunately, CM training appears to be an effective way to address negative beliefs. In the VA implementation effort [52], training workshops decreased perceived barriers and increased positive impressions of CM [91]. In other training efforts, brief educational materials were effective in changing perceptions of CM’s efficacy [92].
Beyond initial training, supervision of CM delivery is likely to be necessary [93,94]. Clinician skill in delivering CM is related to client outcomes [93,95] and relatively simple adherence measures are available for monitoring [96,97]. However, the best methods for training and supervision of CM have yet to be established. The VA initiative was developed in consultation with CM experts and employed ongoing phone consultation following initial training workshops [52,91]. This approach represented significant investment by the VA toward staff training and CM protocol development that may not be achievable for individual clinics. As attention to CM’s dissemination and implementation has grown, some free resources have been developed. Promoting Awareness of Motivational Incentives (PAMI; www.bettertxoutcomes.org/bettertxoutcomes/PAMI.html) is a collaborative initiative sponsored by the National Institute of Drug Abuse and the Substance Abuse and Mental Health Services Administration. It offers free resources and training materials.
Conclusion
Overall, CM is a highly efficacious treatment for SUDs that generalizes to most clients. Despite a robust evidence base, CM’s implementation in clinical settings lags behind other empirically supported treatments [92]. At least in part, CM’s costs, which include not only staff training and adherence monitoring (as with other treatments), but also costs of the incentives themselves, may contribute to slow uptake in clinical settings. Clinics often do not have the resources available for CM within their operating budgets. However, a growing number of projects [19,52,53,55–57,70,71] illustrate CM implementation within routine clinical care, and increased revenue from improved attendance to treatment groups may be one mechanism through which to fund a CM program [54,56,57]. These projects are valuable not only for demonstrating that CM can be efficacious outside the research setting, but also for highlighting how implementation barriers can be overcome. Continued efforts of this nature are likely to be particularly valuable for clinicians and administrators considering adopting CM within clinical settings.
1. Petry NM. A comprehensive guide to the application of contingency management procedures in clinical settings. Drug Alcohol Depend 2000;58:9–25.
2. Stitzer M, Petry N. Contingency management for treatment of substance abuse. Annu Rev Clin Psychol 2006;2:411–34.
3. Meredith SE, Jarvis BP, Raiff BR, et al. The ABCs of incentive-based treatment in health care: A behavior analytic framework to inform research and practice. Psychol Res Behav Manag 2014;7:103–14.
4. Bigelow GE, Silverman K. Theoretical and empirical foundations of contingency management treatments for drug abuse. In: Higgins ST, Silverman K, editors. Motivating behavior change among illicit drug users. Washington DC: American Psychological Association; 1999: 15–31.
5. Higgins ST, Budney AJ, Bickel WK. Applying behavioral concepts and principles to the treatment of cocaine dependence. Drug Alcohol Depend 1994;34:87–97.
6. Skinner BF. Science and human behavior. New York: Macmillan; 1953.
7. Herbeck DM, Hser YI, Teruya C. Empirically supported substance abuse treatment approaches: A survey of treatment providers’ perspectives and practices. Addict Behav 2008;33:699–712.
8. McGovern MP, Fox TS, Xie H, Drake RE. A survey of clinical practices and readiness to adopt evidence-based practices: Dissemination research in an addiction treatment system. J Subst Abuse Treat 2004;26:305–12.
9. Willenbring ML, Kivlahan D, Kenny M, et al. Beliefs about evidence-based practices in addiction treatment: A survey of Veterans Administration program leaders. J Subst Abuse Treat 2004;26:79–85.
10. Griffith JD, Rowan-Szal GA, Roark RR, Simpson DD. Contingency management in outpatient methadone treatment: A meta-analysis. Drug Alcohol Depend 2000;58:55–66.
11. Lussier JP, Heil SH, Mongeon JA, et al. A meta-analysis of voucher-based reinforcement therapy for substance use disorders. Addiction 2006;101:192–203.
12. Benishek LA, Dugosh KL, Kirby KC, et al. Prize-based contingency management for the treatment of substance abusers: A meta-analysis. Addiction 2014;109:1426–36.
13. Prendergast M, Podus D, Finney J, et al. Contingency management for treatment of substance use disorders: A meta-analysis. Addiction 2006;101:1546–60.
14. Dutra L, Stathopoulou G, Basden SL, et al. A meta-analytic review of psychosocial interventions for substance use disorders. Am J Psychiatry 2008;165:179–87.
15. Petry NM, Peirce JM, Stitzer ML, et al. Effect of prize-based incentives on outcomes in stimulant abusers in outpatient psychosocial treatment programs: A National Drug Abuse Treatment Clinical Trials Network study. Arch Gen Psychiatry 2005;62:1148–56.
16. Peirce JM, Petry NM, Stitzer ML, et al. Effects of lower-cost incentives on stimulant abstinence in methadone maintenance treatment - A National Drug Abuse Treatment Clinical Trials Network study. Arch Gen Psychiatry 2006;63:201–8.
17. Petry NM, Martin B, Finocche C. Contingency management in group treatment: a demonstration project in an HIV drop-in center. J Subst Abuse Treat 2001;21:89–96.
18. Drebing CE, Van Ormer EA, Mueller L, et al. Adding contingency management intervention to vocational rehabilitation: outcomes for dually diagnosed veterans. J Rehabil Res Dev 2007;44:851–65.
19. Kellogg SH, Burns M, Coleman P, et al. Something of value: The introduction of contingency management interventions into the New York City Health and Hospital Addiction Treatment Service. J Subst Abuse Treat 2005;28:57–65.
20. Koffarnus MN, Wong CJ, Fingerhood M, et al. Monetary incentives to reinforce engagement and achievement in a job-skills training program for homeless, unemployed adults. J Appl Behav Anal 2013;46:582–91.
21. Rohsenow D, Martin R, Tidey JW, et al. Treating smokers in substance treatment with contingent vouchers, nicotine replacement, and brief advice adapted for sobriety settings. J Subst Abuse Treat 2017.
22. Hunt YM, Rash CJ, Burke RS, Parker JD. Smoking cessation in recovery: Comparing two different cognitive behavioral treatments. Addict Disord Their Treat 2010;9:64–74.
23. Alessi SM, Petry NM, Urso J. Contingency management promotes smoking reductions in residential substance abuse patients. J Appl Behav Anal 2008;41:617–22.
24. Petry NM, Weinstock J, Alessi SM, et al. Group-based randomized trial of contingencies for health and abstinence in HIV patients. J Consult Clin Psychol 2010;78:89.
25. Petry NM, Weinstock J, Alessi SM. A randomized trial of contingency management delivered in the context of group counseling. J Consult Clin Psychol 2011;79:686–96.
26. Ledgerwood DM, Alessi SM, Hanson T, et al. Contingency management for attendance to group substance abuse treatment administered by clinicians in community clinics. J Appl Behav Anal 2008;41:517–26.
27. Alessi SM, Hanson T, Wieners M, Petry NM. Low-cost contingency management in community clinics: Delivering incentives partially in group therapy. Exp Clin Psychopharmacol 2007;15:293–300.
28. Burch AE, Rash CJ, Petry NM. Sex effects in cocaine-using methadone patients randomized to contingency management interventions. Exp Clin Psychopharmacol 2015;23:284–90.
29. Rash CJ, Petry NM. Contingency management treatments are equally efficacious for both sexes in intensive outpatient settings. Exp Clin Psychopharmacol 2015;23:369–76.
30. Barry D, Sullivan B, Petry NM. Comparable efficacy of contingency management for cocaine dependence among African American, Hispanic, and White methadone maintenance clients. Psychol Addict Behav 2009;23:168–74.
31. Rash CJ, Alessi SM, Petry NM. Substance abuse treatment patients in housing programs respond to contingency management interventions. J Subst Abuse Treat 2017;72:97–102.
32. Rash CJ, Olmstead TA, Petry NM. Income does not affect response to contingency management treatments among community substance abuse treatment-seekers. Drug Alcohol Depend 2009;104:249–53.
33. Rash CJ, Andrade LF, Petry NM. Income received during treatment does not affect response to contingency management treatments in cocaine-dependent outpatients. Drug Alcohol Depend 2013;132:528–34.
34. Secades-Villa R, García-Fernández G, Peña-Suárez E, et al. Contingency management is effective across cocaine-dependent outpatients with different socioeconomic status. J Subst Abuse Treat 2013;44:349–54.
35. Rash CJ, Alessi SM, Petry NM. Cocaine abusers with and without alcohol dependence respond equally well to contingency management treatments. Exp Clin Psychopharmacol 2008;16:275–81.
36. Alessi SM, Rash C, Petry NM. Contingency management is efficacious and improves outcomes in cocaine patients with pretreatment marijuana use. Drug Alcohol Depend 2011;118:62–7.
37. Ford JD, Hawke J, Alessi S, et al. Psychological trauma and PTSD symptoms as predictors of substance dependence treatment outcomes. Behav Res Ther 2007;45:2417–31.
38. Weinstock J, Alessi SM, Petry NM. Regardless of psychiatric severity the addition of contingency management to standard treatment improves retention and drug use outcomes. Drug Alcohol Depend 2007;87:288–96.
39. García-Fernández G, Secades-Villa R, García-Rodríguez O, et al. Contingency management improves outcomes in cocaine-dependent outpatients with depressive symptoms. Exp Clin Psychopharmacol 2013;21:482–9.
40. Walter KN, Petry NM. Patients with diabetes respond well to contingency management treatment targeting alcohol and substance use. Psychol Health Med 2015;20:916–26.
41. Burch AE, Morasco BJ, Petry NM. Patients undergoing substance abuse treatment and receiving financial sssistance for a physical disability respond well to contingency management treatment. J Subst Abuse Treat 2015;58:67–71.
42. Burch AE, Rash CJ, Petry NM. Cocaine-using substance abuse treatment patients with and without HIV respond well to contingency management treatment. J Subst Abuse Treat 2017;77:21–5.
43. Rash CJ, Burki M, Montezuma-Rusca JM, Petry NM. A retrospective and prospective analysis of trading sex for drugs or money in women substance abuse treatment patients. Drug Alcohol Depend 2016;162:182–9.
44. Petry NM. Contingency management for substance abuse treatment: A guide to implementing this evidence-based practice. New York: Routledge; 2012.
45. Petry NM, Rash CJ, Byrne S, et al. Financial reinforcers for improving medication adherence: Findings from a meta-analysis. Am J Med 2012;125:888–96.
46. Herrmann ES, Matusiewicz AK, Stitzer ML, et al. Contingency management interventions for HIV, tuberculosis, and hepatitis control among individuals with substance use disorders: a systematized review. J Subst Abuse Treat 2017;72:117–25.
47. Petry NM, Alessi SM, Carroll KM, et al. Contingency management treatments: Reinforcing abstinence versus adherence with goal-related activities. J Consult Clin Psychol 2006;74:592–601.
48. Litt MD, Kadden RM, Petry NM. Behavioral treatment for marijuana dependence: Randomized trial of contingency management and self-efficacy enhancement. Addict Behav 2013;38:1764–75.
49. Kurti AN, Dallery J. A laboratory-based evaluation of exercise plus contingency management for reducing cigarette smoking. Drug Alcohol Depend 2014;144:201–9.
50. Weinstock J, Capizzi J, Weber SM, et al. Exercise as an intervention for sedentary hazardous drinking college students: A pilot study. Ment Health Phys Act 2014;7:55–62.
51. Mitchell MS, Goodman JM, Alter DA, et al. Financial incentives for exercise adherence in adults: Systematic review and meta-analysis. Am J Prev Med 2013;45:658–67.
52. Petry NM, Dephilippis D, Rash CJ, et al. Nationwide dissemination of contingency management: The Veterans Administration initiative. Am J Addict 2014;23:205–10.
53. Walker R, Rosvall T, Field CA, et al. Disseminating contingency management to increase attendance in two community substance abuse treatment centers: Lessons learned. J Subst Abuse Treat 2010;39:202–9.
54. Sigmon SC, Stitzer ML. Use of a low-cost incentive intervention to improve counseling attendance among methadone-maintained patients. J Subst Abuse Treat 2005;29:253–8.
55. Kropp F, Lewis D, Winhusen T. The effectiveness of ultra-low magnitude reinforcers: Findings from a “real-world” application of contingency management. J Subst Abuse Treat 2017;72:111–6.
56. Fitzsimons H, Tuten M, Borsuk C, et al. Clinician-delivered contingency management increases engagement and attendance in drug and alcohol treatment. Drug Alcohol Depend 2015;152:62–7.
57. Lott DC, Jencius S. Effectiveness of very low-cost contingency management in a community adolescent treatment program. Drug Alcohol Depend 2009;102:162–5.
58. Henggeler SW, Chapman JE, Rowland MD, et al. Statewide adoption and initial implementation of contingency management for substance-abusing adolescents. 2008;76:556–67.
59. Henggeler SW, Chapman JE, Rowland MD, et al. If you build it , they will come: Statewide practitioner interest in contingency management for youths. 2007;32:121–31.
60. Petry NM, Martin B, Cooney JL, Kranzler HR. Give them prizes, and they will come: contingency management for treatment of alcohol dependence. J Consult Clin Psychol 2000;68:250–7.
61. Rash CJ, Petry NM, Alessi SM. Examining implementation of contingency management in real-world settings [Abstract]. Alcohol Clin Exp Res 2016;20:103A.
62. Rash CJ, Petry NM, Kirby KC, et al. Identifying provider beliefs related to contingency management adoption using the Contingency Management Beliefs Questionnaire. Drug Alcohol Depend 2012;121:205–12.
63. Petry NM, Alessi SM, Marx J, et al. Vouchers versus prizes: Contingency management treatment of substance abusers in community settings. J Consult Clin Psychol 2005;73:1005–14.
64. Petry NM, Alessi SM, Hanson T, Sierra S. Randomized trial of contingent prizes versus vouchers in cocaine-using methadone patients. J Consult Clin Psychol 2007;75:983–91.
65. Petry NM, Alessi SM, Barry D, Carroll KM. Standard magnitude prize reinforcers can be as efficacious as larger magnitude reinforcers in cocaine-dependent methadone patients. J Consult Clin Psychol 2015;83:464–72.
66. Higgins ST, Budney AJ, Bickel WK, et al. Incentives improve outcome in outpatient behavioral treatment of cocaine dependence. Arch Gen Psychiatry 1994;51:568.
67. Higgins ST, Wong CJ, Badger GJ, et al. Contingent reinforcement increases cocaine abstinence during outpatient treatment and 1 year of follow-up. J Consult Clin Psychol 2000;68:64–72.
68. Hartzler B, Garrett S. Interest and preferences for contingency management design among addiction treatment clientele. Am J Drug Alcohol Abuse 2016;42:287–95.
69. Petry NM, Barry D, Alessi SM, et al. A randomized trial adapting contingency management targets based on initial abstinence status of cocaine-dependent patients. J Consult Clin Psychol 2012;80:276–85.
70. Amass L, Kamien J. A tale of two cities: Financing two voucher programs for substance abusers through community donations. Exp Clin Psychopharmacol 2004;12:147–55.
71. Hartzler B. Building a bonfire that remains stoked: Sustainment of a contingency management intervention developed through collaborative design. Subst Abuse Treat Prev Policy 2015;10:30.
72. Carroll KM, Sinha R, Nich C, et al. Contingency management to enhance naltrexone treatment of opioid dependence: a randomized clinical trial of reinforcement magnitude. Exp Clin Psychopharmacol 2002;10:54–63.
73. Roll JM, Shoptaw S. Contingency management: Schedule effects. Psychiatry Res 2006;144:91–3.
74. Silverman K, Chutuape MA, Bigelow GE, Stitzer ML. Voucher-based reinforcement of cocaine abstinence in treatment-resistant methadone patients: Effects of reinforcement magnitude. Psychopharmacology (Berl) 1999;146:128–38.
75. Businelle MS, Rash CJ, Burke RS, Parker JD. Using vouchers to increase continuing care participation in veterans: does magnitude matter? Am J Addict 2009;18:122–9.
76. Petry NM, Tedford J, Austin M, et al. Prize reinforcement contingency management for treating cocaine users: How low can we go, and with whom? Addiction 2004;99:349–60.
77. Alessi SM, Petry NM. A randomized study of cellphone technology to reinforce alcohol abstinence in the natural environment. Addiction 2013;108:900–9.
78. Alessi SM, Petry NM. Smoking reductions and increased self-efficacy in a randomized controlled trial of smoking abstinence--contingent incentives in residential substance abuse treatment patients. Nicotine Tob Res 2014;16:1436–45.
79. Dougherty DM, Hill-Kapturczak N, Liang Y, et al. Use of continuous transdermal alcohol monitoring during a contingency management procedure to reduce excessive alcohol use. Drug Alcohol Depend 2014;142:301–6.
80. Dallery J, Meredith S, Jarvis B, Nuzzo PA. Internet-based group contingency management to promote smoking abstinence. Exp Clin Psychopharmacol 2015;23:176–83.
81. Higgins ST, Washio Y, Lopez AA, et al. Examining two different schedules of financial incentives for smoking cessation among pregnant women. Prev Med (Baltim) 2014;68:51–7.
82. Higgins ST, Heil SH, Solomon LJ, et al. A pilot study on voucher-based incentives to promote abstinence from cigarette smoking during pregnancy and postpartum. Nicotine Tob Res 2004;6:1015–20.
83. Packer RR, Howell DN, McPherson S, Roll JM. Investigating reinforcer magnitude and reinforcer delay: A contingency management analog study. Exp Clin Psychopharmacol 2012;20:287–92.
84. Kirby KC, Benishek LA, Dugosh KL, Kerwin ME. Substance abuse treatment providers’ beliefs and objections regarding contingency management: Implications for dissemination. Drug Alcohol Depend 2006;85:19–27.
85. Cameron J, Ritter A. Contingency management: Perspectives of Australian service providers. 2007;26:183–9.
86. Hartzler B, Rabun C. Community opioid treatment perspectives on contingency management: Perceived feasibility, effectiveness, and transportability of social and financial incentives. J Subst Abuse Treat 2013;45:242–8.
87. Aletraris L, Shelton JS, Roman PM. Counselor attitudes toward contingency management for substance use disorder: Effectiveness, acceptability, and endorsement of incentives for treatment attendance and abstinence. J Subst Abuse Treat 2015;57:41–8.
88. Budney AJ, Higgins ST, Radonovich KJ, Novy PL. Adding voucher-based incentives to coping skills and motivational enhancement improves outcomes during treatment for marijuana dependence. J Consult Clin Psychol 2000;68:1051–61.
89. Ledgerwood DM, Petry NM. Does contingency management affect motivation to change substance use? Drug Alcohol Depend 2006;83:65–72.
90. Litt MD, Kadden RM, Kabela-Cormier E, Petry NM. Coping skills training and contingency management treatments for marijuana dependence: Exploring mechanisms of behavior change. Addiction 2008;103:638–48.
91. Rash CJ, DePhilippis D, McKay JR, et al. Training workshops positively impact beliefs about contingency management in a nationwide dissemination effort. J Subst Abuse Treat 2013;45:306–12.
92. Benishek LA, Kirby KC, Dugosh KL, Padovano A. Beliefs about the empirical support of drug abuse treatment interventions: A survey of outpatient treatment providers. Drug Alcohol Depend 2010;107:202–8.
93. Petry NM, Alessi SM, Ledgerwood DM. Contingency management delivered by community therapists in outpatient settings. Drug Alcohol Depend 2012;122:86–92.
94. Petry NM, Alessi SM, Ledgerwood DM. A randomized trial of contingency management delivered by community therapists. J Consult Clin Psychol 2012;80:286–98.
95. Hartzler B, Beadnell B, Donovan D. Predictive validity of addiction treatment clinicians’ post-training contingency management skills for subsequent clinical outcomes. J Subst Abuse Treat 2017.
96. Hartzler B. Adapting the Helpful Responses Questionnaire to assess communication skills involved in delivering contingency management: Preliminary psychometrics. J Subst Abuse Treat 2014;55:52–7.
97. Petry NM, Alessi SM, Ledgerwood DM, Sierra S. Psychometric properties of the Contingency Management Competence Scale. Drug Alcohol Depend 2010;109:167–74.
1. Petry NM. A comprehensive guide to the application of contingency management procedures in clinical settings. Drug Alcohol Depend 2000;58:9–25.
2. Stitzer M, Petry N. Contingency management for treatment of substance abuse. Annu Rev Clin Psychol 2006;2:411–34.
3. Meredith SE, Jarvis BP, Raiff BR, et al. The ABCs of incentive-based treatment in health care: A behavior analytic framework to inform research and practice. Psychol Res Behav Manag 2014;7:103–14.
4. Bigelow GE, Silverman K. Theoretical and empirical foundations of contingency management treatments for drug abuse. In: Higgins ST, Silverman K, editors. Motivating behavior change among illicit drug users. Washington DC: American Psychological Association; 1999: 15–31.
5. Higgins ST, Budney AJ, Bickel WK. Applying behavioral concepts and principles to the treatment of cocaine dependence. Drug Alcohol Depend 1994;34:87–97.
6. Skinner BF. Science and human behavior. New York: Macmillan; 1953.
7. Herbeck DM, Hser YI, Teruya C. Empirically supported substance abuse treatment approaches: A survey of treatment providers’ perspectives and practices. Addict Behav 2008;33:699–712.
8. McGovern MP, Fox TS, Xie H, Drake RE. A survey of clinical practices and readiness to adopt evidence-based practices: Dissemination research in an addiction treatment system. J Subst Abuse Treat 2004;26:305–12.
9. Willenbring ML, Kivlahan D, Kenny M, et al. Beliefs about evidence-based practices in addiction treatment: A survey of Veterans Administration program leaders. J Subst Abuse Treat 2004;26:79–85.
10. Griffith JD, Rowan-Szal GA, Roark RR, Simpson DD. Contingency management in outpatient methadone treatment: A meta-analysis. Drug Alcohol Depend 2000;58:55–66.
11. Lussier JP, Heil SH, Mongeon JA, et al. A meta-analysis of voucher-based reinforcement therapy for substance use disorders. Addiction 2006;101:192–203.
12. Benishek LA, Dugosh KL, Kirby KC, et al. Prize-based contingency management for the treatment of substance abusers: A meta-analysis. Addiction 2014;109:1426–36.
13. Prendergast M, Podus D, Finney J, et al. Contingency management for treatment of substance use disorders: A meta-analysis. Addiction 2006;101:1546–60.
14. Dutra L, Stathopoulou G, Basden SL, et al. A meta-analytic review of psychosocial interventions for substance use disorders. Am J Psychiatry 2008;165:179–87.
15. Petry NM, Peirce JM, Stitzer ML, et al. Effect of prize-based incentives on outcomes in stimulant abusers in outpatient psychosocial treatment programs: A National Drug Abuse Treatment Clinical Trials Network study. Arch Gen Psychiatry 2005;62:1148–56.
16. Peirce JM, Petry NM, Stitzer ML, et al. Effects of lower-cost incentives on stimulant abstinence in methadone maintenance treatment - A National Drug Abuse Treatment Clinical Trials Network study. Arch Gen Psychiatry 2006;63:201–8.
17. Petry NM, Martin B, Finocche C. Contingency management in group treatment: a demonstration project in an HIV drop-in center. J Subst Abuse Treat 2001;21:89–96.
18. Drebing CE, Van Ormer EA, Mueller L, et al. Adding contingency management intervention to vocational rehabilitation: outcomes for dually diagnosed veterans. J Rehabil Res Dev 2007;44:851–65.
19. Kellogg SH, Burns M, Coleman P, et al. Something of value: The introduction of contingency management interventions into the New York City Health and Hospital Addiction Treatment Service. J Subst Abuse Treat 2005;28:57–65.
20. Koffarnus MN, Wong CJ, Fingerhood M, et al. Monetary incentives to reinforce engagement and achievement in a job-skills training program for homeless, unemployed adults. J Appl Behav Anal 2013;46:582–91.
21. Rohsenow D, Martin R, Tidey JW, et al. Treating smokers in substance treatment with contingent vouchers, nicotine replacement, and brief advice adapted for sobriety settings. J Subst Abuse Treat 2017.
22. Hunt YM, Rash CJ, Burke RS, Parker JD. Smoking cessation in recovery: Comparing two different cognitive behavioral treatments. Addict Disord Their Treat 2010;9:64–74.
23. Alessi SM, Petry NM, Urso J. Contingency management promotes smoking reductions in residential substance abuse patients. J Appl Behav Anal 2008;41:617–22.
24. Petry NM, Weinstock J, Alessi SM, et al. Group-based randomized trial of contingencies for health and abstinence in HIV patients. J Consult Clin Psychol 2010;78:89.
25. Petry NM, Weinstock J, Alessi SM. A randomized trial of contingency management delivered in the context of group counseling. J Consult Clin Psychol 2011;79:686–96.
26. Ledgerwood DM, Alessi SM, Hanson T, et al. Contingency management for attendance to group substance abuse treatment administered by clinicians in community clinics. J Appl Behav Anal 2008;41:517–26.
27. Alessi SM, Hanson T, Wieners M, Petry NM. Low-cost contingency management in community clinics: Delivering incentives partially in group therapy. Exp Clin Psychopharmacol 2007;15:293–300.
28. Burch AE, Rash CJ, Petry NM. Sex effects in cocaine-using methadone patients randomized to contingency management interventions. Exp Clin Psychopharmacol 2015;23:284–90.
29. Rash CJ, Petry NM. Contingency management treatments are equally efficacious for both sexes in intensive outpatient settings. Exp Clin Psychopharmacol 2015;23:369–76.
30. Barry D, Sullivan B, Petry NM. Comparable efficacy of contingency management for cocaine dependence among African American, Hispanic, and White methadone maintenance clients. Psychol Addict Behav 2009;23:168–74.
31. Rash CJ, Alessi SM, Petry NM. Substance abuse treatment patients in housing programs respond to contingency management interventions. J Subst Abuse Treat 2017;72:97–102.
32. Rash CJ, Olmstead TA, Petry NM. Income does not affect response to contingency management treatments among community substance abuse treatment-seekers. Drug Alcohol Depend 2009;104:249–53.
33. Rash CJ, Andrade LF, Petry NM. Income received during treatment does not affect response to contingency management treatments in cocaine-dependent outpatients. Drug Alcohol Depend 2013;132:528–34.
34. Secades-Villa R, García-Fernández G, Peña-Suárez E, et al. Contingency management is effective across cocaine-dependent outpatients with different socioeconomic status. J Subst Abuse Treat 2013;44:349–54.
35. Rash CJ, Alessi SM, Petry NM. Cocaine abusers with and without alcohol dependence respond equally well to contingency management treatments. Exp Clin Psychopharmacol 2008;16:275–81.
36. Alessi SM, Rash C, Petry NM. Contingency management is efficacious and improves outcomes in cocaine patients with pretreatment marijuana use. Drug Alcohol Depend 2011;118:62–7.
37. Ford JD, Hawke J, Alessi S, et al. Psychological trauma and PTSD symptoms as predictors of substance dependence treatment outcomes. Behav Res Ther 2007;45:2417–31.
38. Weinstock J, Alessi SM, Petry NM. Regardless of psychiatric severity the addition of contingency management to standard treatment improves retention and drug use outcomes. Drug Alcohol Depend 2007;87:288–96.
39. García-Fernández G, Secades-Villa R, García-Rodríguez O, et al. Contingency management improves outcomes in cocaine-dependent outpatients with depressive symptoms. Exp Clin Psychopharmacol 2013;21:482–9.
40. Walter KN, Petry NM. Patients with diabetes respond well to contingency management treatment targeting alcohol and substance use. Psychol Health Med 2015;20:916–26.
41. Burch AE, Morasco BJ, Petry NM. Patients undergoing substance abuse treatment and receiving financial sssistance for a physical disability respond well to contingency management treatment. J Subst Abuse Treat 2015;58:67–71.
42. Burch AE, Rash CJ, Petry NM. Cocaine-using substance abuse treatment patients with and without HIV respond well to contingency management treatment. J Subst Abuse Treat 2017;77:21–5.
43. Rash CJ, Burki M, Montezuma-Rusca JM, Petry NM. A retrospective and prospective analysis of trading sex for drugs or money in women substance abuse treatment patients. Drug Alcohol Depend 2016;162:182–9.
44. Petry NM. Contingency management for substance abuse treatment: A guide to implementing this evidence-based practice. New York: Routledge; 2012.
45. Petry NM, Rash CJ, Byrne S, et al. Financial reinforcers for improving medication adherence: Findings from a meta-analysis. Am J Med 2012;125:888–96.
46. Herrmann ES, Matusiewicz AK, Stitzer ML, et al. Contingency management interventions for HIV, tuberculosis, and hepatitis control among individuals with substance use disorders: a systematized review. J Subst Abuse Treat 2017;72:117–25.
47. Petry NM, Alessi SM, Carroll KM, et al. Contingency management treatments: Reinforcing abstinence versus adherence with goal-related activities. J Consult Clin Psychol 2006;74:592–601.
48. Litt MD, Kadden RM, Petry NM. Behavioral treatment for marijuana dependence: Randomized trial of contingency management and self-efficacy enhancement. Addict Behav 2013;38:1764–75.
49. Kurti AN, Dallery J. A laboratory-based evaluation of exercise plus contingency management for reducing cigarette smoking. Drug Alcohol Depend 2014;144:201–9.
50. Weinstock J, Capizzi J, Weber SM, et al. Exercise as an intervention for sedentary hazardous drinking college students: A pilot study. Ment Health Phys Act 2014;7:55–62.
51. Mitchell MS, Goodman JM, Alter DA, et al. Financial incentives for exercise adherence in adults: Systematic review and meta-analysis. Am J Prev Med 2013;45:658–67.
52. Petry NM, Dephilippis D, Rash CJ, et al. Nationwide dissemination of contingency management: The Veterans Administration initiative. Am J Addict 2014;23:205–10.
53. Walker R, Rosvall T, Field CA, et al. Disseminating contingency management to increase attendance in two community substance abuse treatment centers: Lessons learned. J Subst Abuse Treat 2010;39:202–9.
54. Sigmon SC, Stitzer ML. Use of a low-cost incentive intervention to improve counseling attendance among methadone-maintained patients. J Subst Abuse Treat 2005;29:253–8.
55. Kropp F, Lewis D, Winhusen T. The effectiveness of ultra-low magnitude reinforcers: Findings from a “real-world” application of contingency management. J Subst Abuse Treat 2017;72:111–6.
56. Fitzsimons H, Tuten M, Borsuk C, et al. Clinician-delivered contingency management increases engagement and attendance in drug and alcohol treatment. Drug Alcohol Depend 2015;152:62–7.
57. Lott DC, Jencius S. Effectiveness of very low-cost contingency management in a community adolescent treatment program. Drug Alcohol Depend 2009;102:162–5.
58. Henggeler SW, Chapman JE, Rowland MD, et al. Statewide adoption and initial implementation of contingency management for substance-abusing adolescents. 2008;76:556–67.
59. Henggeler SW, Chapman JE, Rowland MD, et al. If you build it , they will come: Statewide practitioner interest in contingency management for youths. 2007;32:121–31.
60. Petry NM, Martin B, Cooney JL, Kranzler HR. Give them prizes, and they will come: contingency management for treatment of alcohol dependence. J Consult Clin Psychol 2000;68:250–7.
61. Rash CJ, Petry NM, Alessi SM. Examining implementation of contingency management in real-world settings [Abstract]. Alcohol Clin Exp Res 2016;20:103A.
62. Rash CJ, Petry NM, Kirby KC, et al. Identifying provider beliefs related to contingency management adoption using the Contingency Management Beliefs Questionnaire. Drug Alcohol Depend 2012;121:205–12.
63. Petry NM, Alessi SM, Marx J, et al. Vouchers versus prizes: Contingency management treatment of substance abusers in community settings. J Consult Clin Psychol 2005;73:1005–14.
64. Petry NM, Alessi SM, Hanson T, Sierra S. Randomized trial of contingent prizes versus vouchers in cocaine-using methadone patients. J Consult Clin Psychol 2007;75:983–91.
65. Petry NM, Alessi SM, Barry D, Carroll KM. Standard magnitude prize reinforcers can be as efficacious as larger magnitude reinforcers in cocaine-dependent methadone patients. J Consult Clin Psychol 2015;83:464–72.
66. Higgins ST, Budney AJ, Bickel WK, et al. Incentives improve outcome in outpatient behavioral treatment of cocaine dependence. Arch Gen Psychiatry 1994;51:568.
67. Higgins ST, Wong CJ, Badger GJ, et al. Contingent reinforcement increases cocaine abstinence during outpatient treatment and 1 year of follow-up. J Consult Clin Psychol 2000;68:64–72.
68. Hartzler B, Garrett S. Interest and preferences for contingency management design among addiction treatment clientele. Am J Drug Alcohol Abuse 2016;42:287–95.
69. Petry NM, Barry D, Alessi SM, et al. A randomized trial adapting contingency management targets based on initial abstinence status of cocaine-dependent patients. J Consult Clin Psychol 2012;80:276–85.
70. Amass L, Kamien J. A tale of two cities: Financing two voucher programs for substance abusers through community donations. Exp Clin Psychopharmacol 2004;12:147–55.
71. Hartzler B. Building a bonfire that remains stoked: Sustainment of a contingency management intervention developed through collaborative design. Subst Abuse Treat Prev Policy 2015;10:30.
72. Carroll KM, Sinha R, Nich C, et al. Contingency management to enhance naltrexone treatment of opioid dependence: a randomized clinical trial of reinforcement magnitude. Exp Clin Psychopharmacol 2002;10:54–63.
73. Roll JM, Shoptaw S. Contingency management: Schedule effects. Psychiatry Res 2006;144:91–3.
74. Silverman K, Chutuape MA, Bigelow GE, Stitzer ML. Voucher-based reinforcement of cocaine abstinence in treatment-resistant methadone patients: Effects of reinforcement magnitude. Psychopharmacology (Berl) 1999;146:128–38.
75. Businelle MS, Rash CJ, Burke RS, Parker JD. Using vouchers to increase continuing care participation in veterans: does magnitude matter? Am J Addict 2009;18:122–9.
76. Petry NM, Tedford J, Austin M, et al. Prize reinforcement contingency management for treating cocaine users: How low can we go, and with whom? Addiction 2004;99:349–60.
77. Alessi SM, Petry NM. A randomized study of cellphone technology to reinforce alcohol abstinence in the natural environment. Addiction 2013;108:900–9.
78. Alessi SM, Petry NM. Smoking reductions and increased self-efficacy in a randomized controlled trial of smoking abstinence--contingent incentives in residential substance abuse treatment patients. Nicotine Tob Res 2014;16:1436–45.
79. Dougherty DM, Hill-Kapturczak N, Liang Y, et al. Use of continuous transdermal alcohol monitoring during a contingency management procedure to reduce excessive alcohol use. Drug Alcohol Depend 2014;142:301–6.
80. Dallery J, Meredith S, Jarvis B, Nuzzo PA. Internet-based group contingency management to promote smoking abstinence. Exp Clin Psychopharmacol 2015;23:176–83.
81. Higgins ST, Washio Y, Lopez AA, et al. Examining two different schedules of financial incentives for smoking cessation among pregnant women. Prev Med (Baltim) 2014;68:51–7.
82. Higgins ST, Heil SH, Solomon LJ, et al. A pilot study on voucher-based incentives to promote abstinence from cigarette smoking during pregnancy and postpartum. Nicotine Tob Res 2004;6:1015–20.
83. Packer RR, Howell DN, McPherson S, Roll JM. Investigating reinforcer magnitude and reinforcer delay: A contingency management analog study. Exp Clin Psychopharmacol 2012;20:287–92.
84. Kirby KC, Benishek LA, Dugosh KL, Kerwin ME. Substance abuse treatment providers’ beliefs and objections regarding contingency management: Implications for dissemination. Drug Alcohol Depend 2006;85:19–27.
85. Cameron J, Ritter A. Contingency management: Perspectives of Australian service providers. 2007;26:183–9.
86. Hartzler B, Rabun C. Community opioid treatment perspectives on contingency management: Perceived feasibility, effectiveness, and transportability of social and financial incentives. J Subst Abuse Treat 2013;45:242–8.
87. Aletraris L, Shelton JS, Roman PM. Counselor attitudes toward contingency management for substance use disorder: Effectiveness, acceptability, and endorsement of incentives for treatment attendance and abstinence. J Subst Abuse Treat 2015;57:41–8.
88. Budney AJ, Higgins ST, Radonovich KJ, Novy PL. Adding voucher-based incentives to coping skills and motivational enhancement improves outcomes during treatment for marijuana dependence. J Consult Clin Psychol 2000;68:1051–61.
89. Ledgerwood DM, Petry NM. Does contingency management affect motivation to change substance use? Drug Alcohol Depend 2006;83:65–72.
90. Litt MD, Kadden RM, Kabela-Cormier E, Petry NM. Coping skills training and contingency management treatments for marijuana dependence: Exploring mechanisms of behavior change. Addiction 2008;103:638–48.
91. Rash CJ, DePhilippis D, McKay JR, et al. Training workshops positively impact beliefs about contingency management in a nationwide dissemination effort. J Subst Abuse Treat 2013;45:306–12.
92. Benishek LA, Kirby KC, Dugosh KL, Padovano A. Beliefs about the empirical support of drug abuse treatment interventions: A survey of outpatient treatment providers. Drug Alcohol Depend 2010;107:202–8.
93. Petry NM, Alessi SM, Ledgerwood DM. Contingency management delivered by community therapists in outpatient settings. Drug Alcohol Depend 2012;122:86–92.
94. Petry NM, Alessi SM, Ledgerwood DM. A randomized trial of contingency management delivered by community therapists. J Consult Clin Psychol 2012;80:286–98.
95. Hartzler B, Beadnell B, Donovan D. Predictive validity of addiction treatment clinicians’ post-training contingency management skills for subsequent clinical outcomes. J Subst Abuse Treat 2017.
96. Hartzler B. Adapting the Helpful Responses Questionnaire to assess communication skills involved in delivering contingency management: Preliminary psychometrics. J Subst Abuse Treat 2014;55:52–7.
97. Petry NM, Alessi SM, Ledgerwood DM, Sierra S. Psychometric properties of the Contingency Management Competence Scale. Drug Alcohol Depend 2010;109:167–74.
Chronic Pain: How to Approach These 3 Common Conditions
Case 1 Lola, 28, has a history of muscular aches and joint pain throughout her body, fatigue, and mental fogginess. A rheumatologist diagnosed fibromyalgia, but Lola just moved to your town and is establishing care. She is feeling desperate because her pain has worsened and the medication previously prescribed (gabapentin 300 mg tid) is no longer working. She asks to try oxycodone.
Case 2 Matt is a 59-year-old truck driver with severe hip osteoarthritis (OA). His orthopedist recommended against hip replacement at this time because of his young age and a heart condition that makes him high risk. His pain makes sitting for long periods very difficult. He presents to you for help because he is worried he will be unable to continue working.
Case 3 Keith is a 56-year-old construction worker who has been experiencing back pain for many years. The pain has become more debilitating over time; it is now constant, and Keith can hardly make it through his work day. He has been getting hydrocodone/acetaminophen from urgent care centers and emergency departments, but he isn’t sure it is helping and is coming to you to assume his pain management.
Chronic pain (defined as > 3 mo in duration) is a complex, heterogeneous condition affecting an estimated 116 million US adults.1 Much of the management of chronic pain occurs in primary care settings, placing family practice providers (FPPs) on the frontlines of two epidemics: that of chronic pain and that of the abuse and misuse of opioid pain medications.
To improve communication about the risks and benefits of opioid therapy and the safety and effectiveness of pain treatments in general, many professional organizations, health care institutions, and recently the CDC, have published guidelines on the use of opioids for nonmalignant chronic pain.2 With these guidelines in mind—and in light of the latest evidence—we propose the following paradigm for the treatment of chronic pain. A critical aspect is determining the underlying pathophysiology of a patient’s pain in order to develop a well-rounded, multimodal, evidence-based treatment plan. Detailed here is the application of this approach to the treatment of three common diagnoses: fibromyalgia, osteoarthritis, and low back pain.
LOOK TO THE CENTRAL AND PERIPHERAL NERVOUS SYSTEM
Acute pain begins with activation of peripheral nociceptors at the site of injury. This causes depolarization up the spinal cord and through the brain stem to higher cortical centers where the pain is perceived and localized. Descending neural pathways transport both excitatory and inhibitory information from the brain to the periphery via the spinal cord, which either increases or decreases the perception of pain.3
When damage/injury doesn’t correlate with the perception of pain
Until recently, it was assumed that chronic pain worked much the same way as acute pain and was caused by ongoing nociceptive input in the periphery, but research has shown us that the central nervous system (CNS) can play a large role in the modulation of nociception. This new understanding comes from the lack of evidence pointing to any pain state in which the degree of nociceptive input correlates with the degree of pain experienced.
For most patients with chronic pain, regardless of their diagnosis, there is some degree of alteration in the processing of nociceptive signals by the CNS contributing to the experience of pain.4 This alteration is thought to result from peripheral nociceptive signaling persisting past the point of tissue healing, leading to a hypersensitivity of nerve fibers, which then continue to respond to low, or absent, sensory stimuli.
Central sensitization is when this hypersensitivity develops in the superficial, deep, and ventral cord nerves. When this happens, pain is often accompanied by systemic symptoms such as fatigue and slowed cognitive processing, often with little to no actual stimulation of the peripheral nociceptors.3
Table 1 lists the possible mechanisms of pain, which can be broken down into four categories: peripheral nociceptive (inflammatory or mechanical), peripheral neuropathic (underlying damage to a peripheral nerve), central (when the CNS is the primary entity involved in maintaining the pain), or any combination of the three.4
As pain becomes chronic, multiple mechanisms overlap
It is important to remember that for any single pain diagnosis, there is likely to be—at least initially—a principle underlying mechanism generating the pain. But as the pain becomes chronic, an overlap of multiple mechanisms develops, with central sensitization often playing a more dominant role than peripheral stimulation (regardless of the diagnosis).
For example, in a patient with rheumatoid arthritis (RA), peripheral nociceptive input (in the form of inflammation) is likely the initial mechanism at work, but as time goes on, central processing becomes more involved. The patient may then begin to experience pain that is disproportionate to what is generally expected with RA and may develop other somatic symptoms. The diagnosis then becomes pain primarily related to RA with central sensitization, and both need to be addressed in a treatment plan. In rheumatic conditions, comorbid fibromyalgia (indicative of central sensitization) is thought to occur in 15% to 30% of patients.5
FPPs can utilize the underlying mechanisms to cut across diagnostic labels and tailor treatments to those that are most likely to be effective. For a patient with more prominent peripheral involvement, a procedural intervention such as injections or surgery alone may suffice, whereas a broader approach including psychotherapy, medications, exercise, and other lifestyle interventions may be necessary for a patient with pain caused predominantly by central sensitization.
Addressing both peripheral and central components is essential. One prospective, observational cohort study of more than 600 patients scheduled for unilateral total knee or total hip arthroplasty found that patients with a higher degree of centralization of pain (measured by widespread pain index and modified fibromyalgia screening scales) were less likely to report improvement in the affected body part and in overall body pain following surgery.6,7
There is a high degree of overlap among many of the chronic pain syndromes (fibromyalgia, irritable bowel syndrome, interstitial cystitis, chronic headaches) that have been found to have a central sensitization component.8 Providers of primary care are aptly positioned to recognize central sensitization as the underlying pathology and target treatment effectively.
TAILOR TREATMENT TO THE UNDERLYING MECHANISMS OF PAIN
As with any chronic condition, a thorough workup (complete with history, physical exam, and diagnostic testing, as appropriate) is indicated. In the setting of chronic pain, it’s important to identify the primary mechanism, as well as secondary factors that may contribute to the patient’s pain, before developing your treatment plan. These secondary factors may include co-occurring affect disorders, a history of trauma, poor sleep, and tobacco use.9-12 A history of trauma, for example, co-exists with many pain syndromes. For these patients, central sensitization is responsible for much of their pain. As a result, traditional cognitive behavioral therapy (CBT) may not be the best option because of its focus on accepting pain as a chronic diagnosis; more trauma-focused treatments, such as those dealing in emotional awareness and understanding of the CNS’s role in chronic pain, need to be considered.13
Three common conditions. Below we present evidence-based treatment approaches for conditions typically associated with each of the major mechanisms of chronic pain: fibromyalgia (central sensitization), OA (peripheral nociceptive), and low back pain (mixed pain state).
Fibromyalgia: a case of central sensitization
Fibromyalgia is a hallmark diagnosis for patients in whom central sensitization is the dominant cause of pain. They usually present with widespread, diffuse pain and somatic symptoms such as fatigue, memory difficulties, and poor sleep quality.8 When explaining the pain mechanism to patients, it may be useful to use the analogy of a volume control dial that is stuck in the “high” position and can’t be turned down.
Genes, the environment, and neurotransmitters play a role. The origin of the pain amplification process is believed to be multifactorial.
Genetic factors are thought to contribute to a predisposition for amplification. To date, five sets of genes have been implicated in increased sensitivity to pain leading to increased risk of the development of chronic pain during a patient’s lifetime.14-19
Environmental factors (eg, early life trauma, physical trauma especially to the trunk, certain infections such as Lyme disease and Epstein-Barr virus, and emotional stress) may trigger or exacerbate symptoms.8 Of note: Only about 5% to 10% of people who experience these triggers actually develop a chronic pain state, while the rest regain their baseline health.4 This raises the question of whether there is a point during an acute pain episode in which one can intervene and prevent the acute pain from becoming chronic in those at higher risk.4
Imbalances of neurotransmitters (high glutamate; low norepinephrine, serotonin, and gamma-aminobutyric acid [GABA]) play a role in central amplification.20-22 These substances not only affect sensory transmission, but also control levels of alertness, sleep, mood, and memory.
The diagnostic criteria for fibromyalgia were modified in 2011 to remove the tender point examination and to add somatic symptoms.6 These criteria can be useful in the clinical setting in identifying not only fibromyalgia itself but also the degree of “fibromyalgianess” a patient has, which is an indicator of how large a role the centralization process plays in the maintenance of chronic pain.23,24
Treatment: multimodal and patient empowering. Evidence-based treatment options for fibromyalgia, as well as other conditions for which there is a high degree of centralized pain, can be found in Table 2.25-36 Multimodal treatment, with an emphasis on patient knowledge and empowerment, is generally thought to be the most beneficial.25,37 Treatment should almost always include CBT and exercise/activity therapies, which have high degrees of efficacy with few adverse effects.26,29
In terms of medication, centrally-acting agents (tricyclic antidepressants, serotonin norepinephrine reuptake inhibitors [SNRIs], and alpha 2 delta ligands) are the most effective. There is little to no data showing benefit from anti-inflammatories or opioids in the setting of fibromyalgia. There is some data to suggest that combination therapy, for example with an SNRI (milnacipran) and an alpha 2 delta ligand (pregabalin), may provide more benefit than treating with pregabalin alone.38
Complementary and alternative therapies (eg, yoga, chiropractic care, acupuncture, massage) are being studied more, and while evidence is only preliminary in terms of efficacy, there is increasing emphasis being placed on the need for patients with chronic pain to shift their treatment expectations to greater acceptance of pain and the need for ongoing self-care.28
OA: an example of peripheral nociceptive pain
OA is a condition long thought to be characterized by damage to the cartilage and bone; however, as with many other pain diagnoses, there is frequently little correlation between damage seen on radiographs and the amount of pain that patients experience.
One study analyzed data on almost 7,000 patients from the National Health and Nutrition Examination Survey (NHANES I) and found that between 30% and 50% of OA patients with moderate to severe radiographic changes were asymptomatic, and 10% of those with moderate to severe pain had normal radiographs or only mild changes.39 Research is showing that many factors may contribute to this discrepancy, including the typical “wear and tear” of the disease, subacute levels of inflammation that can lead to peripheral sensitization, and, in some patients, a centralized pain component.40 The patients with more centralized pain often have pain that is disproportionate to radiographic evidence, as well as more somatic symptoms, such as fatigue, sleep disturbance, and memory issues.41
Treatment should be multimodal and include interventions targeted at halting the progression of damage as well as palliation of pain. All treatment plans for OA should also include exercise, weight reduction, and self-management, in addition to pharmacologic interventions, to reduce both the micro-inflammation and the centralized pain component (when present). Intra-articular injections of various types have been studied with some having more efficacy in pain reduction and functional improvement than others.42-45 See Table 3 for a summary of evidence-based treatment options.42-61
Low back pain: a mixed pain state
Low back pain (LBP) has been recognized as a mixed pain state for quite some time. While some patients may experience purely nociceptive and/or neuropathic pain, most cases are nonspecific, with patients experiencing varying degrees of nociceptive (myofascial LBP), neuropathic (lumbar radiculopathy), and central sensitization pain.62,63 Evidence for centralized pain is demonstrated in studies showing hyperalgesia, augmented central pain processing, involvement of the emotional brain, and delayed recovery influenced by poor coping strategies.64-67
When developing a treatment plan for a patient with chronic LBP, remember that the pain derives from a complex combination of pathophysiologic contributors. Identifying where a patient lies on the pain centralization spectrum can help you tailor treatment.
In one study of 548 patients presenting to a tertiary pain clinic with primary spine pain diagnoses, 42% met diagnostic criteria for fibromyalgia.68 Compared to criteria-negative patients, these patients tended to be younger, unemployed, and receiving compensation; they had greater pain intensity, pain interference, and used stronger words to describe their neuropathic pain, as well as having higher levels of depression/anxiety and a lower level of physical function.
Because LBP is a condition with high prevalence and associated disability, many clinical boards have created guidelines for management. These guidelines tend to vary in the strength of evidence used, and the extent to which they are followed in clinical practice remains largely unknown. Recommendations frequently discourage the use of ultrasound/electrotherapy, but many encourage short-term use of medications, supervised exercise therapy, CBT, and multidisciplinary treatment.
Guidelines tend to differ most widely with regard to recommendations for spinal manipulation and specific drug therapies.69 The classes of drugs that may be most useful when centralized pain is present include the SNRIs and the alpha 2 delta calcium channel ligands.4 See Table 4 for a summary of evidence-based treatment options.70-89
Case 1 Lola is started on amitriptyline 25 mg at bedtime, which improves her fatigue and cognitive symptoms. During monthly office visits, her FPP educates her about the pathophysiology of fibromyalgia and uses motivational interviewing to get her slowly moving and increasing her activity level. She is weaned off the gabapentin previously prescribed, as her symptoms stabilize and improve.
Case 2 Matt is sent for a steroid injection, which decreases his pain temporarily. During this time, he begins physical therapy; slowly, with increased movement, his function improves. A trial of duloxetine provides pain relief; that combined with intermittent NSAIDs has allowed Matt to maintain his function and his job.
Case 3 Because Keith was only taking the narcotics intermittently and wasn’t certain they were helping, CBT was sufficient to wean him off the medication without any worsening of his pain in the process. By participating in physical therapy, he has learned how to perform certain tasks at his job without pain or injury. He uses NSAIDs as needed for pain.
The authors thank Drs. Daniel Clauw (University of Michigan, Ann Arbor) and Martha Rumschlag (Providence Family Medicine Residency Program, Southfield, Michigan), for their valuable contributions to this article.
1. Institute of Medicine (US) Committee on Advancing Pain Research, Care, and Education. Relieving pain in America: a blueprint for transforming prevention, care, education, and research. Washington (DC): National Academies Press (US); 2011.
2. Dowell D, Haegerich TM, Chou R. CDC Guideline for Prescribing Opioids for Chronic Pain—United States, 2016. MMWR Recomm Rep. 2016;65:1-49.
3. Aronoff GM. What do we know about the pathophysiology of chronic pain? Implications for treatment considerations. Med Clin North Am. 2016;100:31-42.
4. Clauw DJ. Diagnosing and treating chronic musculoskeletal pain based on the underlying mechanism(s). Best Pract Res Clin Rheumatol. 2015;29:6-19.
5. Clauw DJ, Katz P. The overlap between fibromyalgia and inflammatory rheumatic disease: when and why does it occur? J Clin Rheumatol. 1995;1:335-342.
6. Wolfe F, Clauw DJ, Fitzcharles MA, et al. Fibromyalgia criteria and severity scales for clinical and epidemiological studies: a modification of the ACR Preliminary Diagnostic Criteria for Fibromyalgia. J Rheumatol. 2011;38:1113-1122.
7. Brummett CM, Urquhart AG, Hassett AL, et al. Characteristics of fibromyalgia independently predict poorer long-term analgesic outcomes following total knee and hip arthroplasty. Arthritis Rheumatol. 2015;67:1386-1394.
8. Ablin K, Clauw DJ. From fibrositis to functional somatic syndromes to a bell-shaped curve of pain and sensory sensitivity: evolution of a clinical construct. Rheum Dis Clin North Am. 2009;35:233-251.
9. Giesecke T, Gracely RH, Williams DA, et al. The relationship between depression, clinical pain, and experimental pain in a chronic pain cohort. Arthritis Rheum. 2005;52:1577-1584.
10. Tesarz J, Eich W, Treede RD, et al. Altered pressure pain thresholds and increased wind-up in adult chronic back pain patients with a history of childhood maltreatment: a quantitative sensory testing study. Pain. 2016;157:1799-1809.
11. Finan PH, Goodin BR, Smith MT. The association of sleep and pain: an update and a path forward. J Pain. 2013;14:1539-1552.
12. Shi Y, Weingarten TN, Mantilla CB, et al. Smoking and pain: pathophysiology and clinical implications. Anesthesiology. 2010;113:977-992.
13. Burger AJ, Lumley MA, Carty JN, et al. The effects of a novel psychological attribution and emotional awareness and expression therapy for chronic musculoskeletal pain: a preliminary, uncontrolled trial. J Psychosom Res. 2016;81:1-8.
14. Zubieta JK, Heitzeg MM, Smith YR, et al. COMT val158met genotype affects mu-opioid neurotransmitter responses to a pain stressor. Science. 2003;299:1240-1243.
15. van Meurs JB, Uitterlinden AG, Stolk L, et al. A functional polymorphism in the catechol-O-methyltransferase gene is associated with osteoarthritis-related pain. Arthritis Rheum. 2009;60:628-629.
16. McLean SA, Diatchenko L, Lee YM, et al. Catechol O-methyltransferase haplotype predicts immediate musculoskeletal neck pain and psychological symptoms after motor vehicle collision. J Pain. 2011;12:101-107.
17. Costigan M, Belfer I, Griffin RS, et al. Multiple chronic pain states are associated with a common amino acid-changing allele in KCNS1. Brain. 2010;133:2519-2527.
18. Tegeder I, Costigan M, Griffin RS, et al. GTP cyclohydrolase and tetrahydrobiopterin regulate pain sensitivity and persistence. Nat Med. 2006;12:1269-1277.
19. Amaya F, Wang H, Costigan M, et al. The voltage-gated sodium channel Na(v)1.9 is an effector of peripheral inflammatory pain hypersensitivity. J Neurosci. 2006;26:12852-12860.
20. Harris RE, Napadow V, Huggins JP, et al. Pregabalin rectifies abberrant brain chemistry, connectivity, and functional responses in chronic pain patients. Anesthesiology. 2013;119:1453-1464.
21. Russell IJ, Vaeroy H, Javors M, et al. Cerebrospinal fluid biogenic amine metabolites in fibromyalgia/fibrositis syndrome and rheumatoid arthritis. Arthritis Rheum. 1992;35:550-556.
22. Foerster BR, Petrou M, Edden RAE, et al. Reduced insular gamma-aminobutyric acid in fibromyalgia. Arthritis Rheum. 2012;64:579-583.
23. Clauw DJ. Fibromyalgia: a clinical review. JAMA. 2014;311:1547-1555.
24. Wolfe F. Fibromyalgianess. Arthritis Rheum. 2009;61:715-716.
25. Hauser W, Bernardy K, Arnold B, et al. Efficacy of multicomponent treatment in fibromyalgia syndrome: a meta-analysis of randomized controlled clinical trials. Arthritis Rheum. 2009;61:216-224.
26. Hauser W, Klose P, Langhorst J, et al. Efficacy of different types of aerobic exercise in fibromyalgia syndrome: a systematic review and meta-analysis of randomised controlled trials. Arthritis Res Ther. 2010;12:R79.
27. Porter NS, Jason LA, Boulton A, et al. Alternative medical interventions used in the treatment and management of myalgic encephalomyelitis/chronic fatigue syndrome and fibromyalgia. J Altern Complement Med. 2010;16:235-249.
28. Eaves ER, Sherman KJ, Ritenbaugh C, et al. A qualitative study of changes in expectations over time among patients with chronic low back pain seeking four CAM therapies. BMC Complement Altern Med. 2015;15:12.
29. Bernardy K, Fuber N, Kollner V, et al. Efficacy of cognitive-behavioral therapies in fibromyalgia syndrome: a systematic review and metaanalysis of randomized controlled trials. J Rheumatol. 2010;37:1991-2005.
30. Arnold LM, Keck PE Jr, Welge JA. Antidepressant treatment of fibromyalgia. A meta-analysis and review. Psychosomatics. 2000;41:104-113.
31. Moldofsky H, Harris HW, Archambault WT, et al. Effects of bedtime very low dose cyclobenzaprine on symptoms and sleep physiology in patients with fibromyalgia syndrome: a double-blind randomized placebo-controlled study. J Rheumatol. 2011;38:2653-2663.
32. Arnold LM. Duloxetine and other antidepressants in the treatment of patients with fibromyalgia. Pain Med. 2007;(8 Suppl 2):S63-S74.
33. Häuser W, Bernardy K, Uceyler N, et al. Treatment of fibromyalgia syndrome with gabapentin and pregabalin—a meta-analysis of randomized controlled trials. Pain. 2009;145:69-81.
34. Gaskell H, Moore RA, Derry S, et al. Oxycodone for neuropathic pain and fibromyalgia in adults. Cochrane Database Syst Rev. 2014;Jun 23:CD010692.
35. MacLean AJ, Schwartz TL. Tramadol for the treatment of fibromyalgia. Expert Rev Neurother. 2015;15:469-475.
36. Younger J, Noor N, McCue R, et al. Low-dose naltrexone for the treatment of fibromyalgia: findings of a small, randomized, double-blind, placebo-controlled, counterbalanced, crossover trial assessing daily pain levels. Arthritis Rheum. 2013;65:529-538.
37. Camerini L, Schulz PJ, Nakamoto K. Differential effects of health knowledge and health empowerment over patients’ self-management and health outcomes: a cross-sectional evaluation. Patient Educ Couns. 2012;89:337-344.
38. Mease PJ, Farmer MV, Palmer RH, et al. Milnacipran combined with pregabalin in fibromyalgia: a randomized, open-label study evaluating the safety and efficacy of adding milnacipran in patients with incomplete response to pregabalin. Ther Adv Musculoskeletal Dis. 2013;5:113-126.
39. Hannan MT, Felson DT, Pincus T. Analysis of the discordance between radiographic changes and knee pain in osteoarthritis of the knee. J Rheumatol. 2000;27:1513-1517.
40. Daghestani HN, Kraus VB. Inflammatory biomarkers in osteoarthritis. Osteoarthritis Cartilage. 2015;23:1890-1896.
41. Fingleton C, Smart K, Moloney N, et al. Pain sensitization in people with knee osteoarthritis: a systematic review and meta-analysis. Osteoarthritis Cartilage. 2015;23:1043-1056.
42. Strand V, McIntyre LF, Beach WR, et al. Safety and efficacy of US-approved viscosupplements for knee osteoarthritis: a systematic review and meta-analysis of randomized, saline-controlled trials. J Pain Res. 2015;8:217-228.
43. Jüni P, Hari R, Rutjes AW, et al. Intra-articular corticosteroid for knee osteoarthritis. Cochrane Database Syst Rev. 2015:CD005328.
44. Meheux CJ, McCulloch PC, Lintner DM, et al. Efficacy of intra-articular platelet-rich plasma injections in knee osteoarthritis: a systematic review. Arthroscopy. 2016;32:495-505.
45. Wu T, Song HX, Dong Y, et al. Intra-articular injections of botulinum toxin a for refractory joint pain: a systematic review and meta-analysis. Clin Rehabil. 2017;31(4):435-443.
46. Jordan JL, Holden MA, Mason EE, et al. Interventions to improve adherence to exercise for chronic musculoskeletal pain in adults. Cochrane Database Syst Rev. 2010:CD005956.
47. Bodenheimer T, Lorig K, Holman H, et al. Patient self-management of chronic disease in primary care. JAMA. 2002;288:2469-2475.
48. Fransen M, McConnell S, Hernandez-Molina G, et al. Exercise for osteoarthritis of the hip. Cochrane Database Syst Rev. 2014:CD007912.
49. Bartels EM, Juhl CB, Christensen R, et al. Aquatic exercise for the treatment of knee and hip osteoarthritis. Cochrane Database Syst Rev. 2016;3:CD005523.
50. da Costa BR, Reichenbach S, Keller N, et al. Effectiveness of non-steroidal anti-inflammatory drugs for the treatment of pain in knee and hip osteoarthritis: a network meta-analysis. Lancet. 2016;387:2093-2105.
51. Myers J, Wielage RC, Han B, et al. The efficacy of duloxetine, non-steroidal anti-inflammatory drugs, and opioids in osteoarthritis: a systematic literature review and meta-analysis. BMC Musculoskelet Disord. 2014;15:76.
52. Berthelot JM, Darrieutort-Lafitte C, Le Goff B, et al. Strong opioids for noncancer pain due to musculoskeletal diseases: not more effective than acetaminophen or NSAIDs. Joint Bone Spine. 2015;82:397-401.
53. Clegg DO, Reda DJ, Harris CL, et al. Glucosamine, chondroitin sulfate, and the two in combination for painful knee osteoarthritis. N Engl J Med. 2006;354:795-808.
54. Wandel S, Jüni P, Tendal B, et al. Effects of glucosamine, chondroitin, or placebo in patients with osteoarthritis of hip or knee: network meta-analysis. BMJ. 2010;341:c4675.
55. Sawitzke AD, Shi H, Finco MF, et al. Clinical efficacy and safety of glucosamine, chondroitin sulphate, their combination, celecoxib or placebo taken to treat osteoarthritis of the knee: 2-year results from GAIT. Ann Rheum Dis. 2010;69:1459-1464.
56. Wu D, Huang Y, Gu Y, et al. Efficacies of different preparations of glucosamine for the treatment of osteoarthritis: a meta-analysis of randomised, double-blind, placebo-controlled trials. Int J Clin Pract. 2013;67:585-594.
57. Kahan A, Uebelhart D, De Vathaire F, et al. Long-term effects of chondroitins 4 and 6 sulfate on knee osteoarthritis: the study on osteoarthritis progression prevention, a two-year, randomized, double-blind, placebo-controlled trial. Arthritis Rheum. 2009;60:524-533.
58. Perkins K, Sahy W, Beckett RD. Efficacy of curcuma for treatment of osteoarthritis. J Evid Based Complementary Altern Med. 2017;22:156-165.
59. Clinton CM, O’Brien S, Law J, et al. Whole-foods, plant-based diet alleviates the symptoms of osteoarthritis. Arthritis. 2015;2015:708152.
60. Manyanga T, Froese M, Zarychanski R, et al. Pain management with acupuncture in osteoarthritis: a systematic review and meta-analysis. BMC Complement Altern Med. 2014;14:312.
61. Vickers AJ, Cronin AM, Maschino AC, et al. Acupuncture for chronic pain: individual patient data meta-analysis. Arch Intern Med. 2012;172:1444-1453.
62. Nijs J, Apeldoorn A, Hallegraeff H, et al. Low back pain: guidelines for the clinical classification of predominant neuropathic, nociceptive, or central sensitization pain. Pain Physician. 2015;18:E333-E346.
63. Fishbain DA, Cole B, Lewis JE, et al. What is the evidence that neuropathic pain is present in chronic low back pain and soft tissue syndromes? An evidence-based structured review. Pain Med. 2014;15:4-15.
64. Hübscher M, Moloney N, Rebbeck T, et al. Contributions of mood, pain catastrophizing, and cold hyperalgesia in acute and chronic low back pain: a comparison with pain-free controls. Clin J Pain. 2014;30:886-893.
65. Giesecke T, Gracely RH, Grant MA, et al. Evidence of augmented central pain processing in idiopathic chronic low back pain. Arthritis Rheum. 2004;50:613-623.
66. Baliki MN, Chialvo DR, Geha PY, et al. Chronic pain and the emotional brain: specific brain activity associated with spontaneous fluctuations of intensity of chronic back pain. J Neurosci. 2006;26:12165-12173.
67. Wertli MM, Eugster R, Held U, et al. Catastrophizing-a prognostic factor for outcome in patients with low back pain: a systematic review. Spine J. 2014;14:2639-2657.
68. Brummett CM, Goesling J, Tsodikov A, et al. Prevalence of the fibromyalgia phenotype in patients with spine pain presenting to a tertiary care pain clinic and the potential treatment implications. Arthritis Rheum. 2013;65:3285-3292.
69. Koes BW, van Tulder M, Lin CW, et al. An updated overview of clinical guidelines for the management of non-specific low back pain in primary care. Eur Spine J. 2010;19:2075-2094.
70. Oliveira VC, Ferreira PH, Maher CG, et al. Effectiveness of self-management of low back pain: systematic review with meta-analysis. Arthritis Care Res. 2012;64:1739-1748.
71. Engers A, Jellema P, Wensing M, et al. Individual patient education for low back pain. Cochrane Database Syst Rev. 2008:CD004057.
72. Hayden JA, van Tulder MW, Malmivaara A, et al. Exercise therapy for treatment of non-specific low back pain. Cochrane Database Syst Rev. 2005:CD000335.
73. French SD, Cameron M, Walker BF, et al. Superficial heat or cold for low back pain. Cochrane Database Syst Rev. 2006:CD004750.
74. Franke H, Franke JD, Fryer G. Osteopathic manipulative treatment for nonspecific low back pain: a systematic review and meta-analysis. BMC Musculoskeletal Disord. 2014;15:286.
75. Franke H, Fryer G, Ostelo RW, et al. Muscle energy technique for non-specific low back pain. Cochrane Database Syst Rev. 2015:CD009852.
76. Oliphant D. Safety of spinal manipulation in the treatment of lumbar disk herniations: a systematic review and risk assessment. J Manipulative Physiol Ther. 2004:197-210.
77. Furlan AD, Giraldo M, Baskwill A, et al. Massage for low-back pain. Cochrane Database Syst Rev. 2015:CD001929.
78. Khadilkar A, Odebiyi DO, Brosseau L, et al. Transcutaneous electrical nerve stimulation (TENS) versus placebo for chronic low back pain. Cochrane Database Syst Rev. 2008:CD003008.
79. Ebadi S, Henschke N, Nakhostin Ansari N, et al. Therapeutic ultrasound for chronic low back pain. Cochrane Database Syst Rev. 2014:CD009169.
80. Furlan AD, van Tulder MW, Cherkin DC, et al. Acupuncture and dry-needling for low back pain. Cochrane Database Syst Rev. 2005:CD001351.
81. Chou R, Huffman LH. Nonpharmacologic therapies for acute and chronic low back pain: a review of the evidence for an American Pain Society/American College of Physicians clinical practice guideline. Ann Intern Med. 2007;147:492-504.
82. Sherman KJ, Cherkin DC, Erro J, et al. Comparing yoga, exercise, and a self-care book for chronic low back pain: a randomized, controlled trial. Ann Intern Med. 2005;143:849-856.
83. Cherkin DC, Sherman KJ, Balderson BH, et al. Effect of mindfulness-based stress reduction vs cognitive behavioral therapy or usual care on back pain and functional limitations in adults with chronic low back pain: a randomized clinical trial. JAMA. 2016;315:1240-1249.
84. Staal JB, de Bie R, de Vet HC, et al. Injection therapy for subacute and chronic low back pain. Cochrane Database Syst Rev. 2008:CD001824.
85. Chou R, Baisden J, Carragee EJ, et al. Surgery for low back pain: a review of the evidence for an American Pain Society Clinical Practice Guideline. Spine. 2009;34:1094-1109.
86. Felson D. Paracetamol is ineffective for spinal pain and knee and hip osteoarthritis. Evid Based Med. 2015;20:205.
87. Machado GC, Maher CG, Ferreira PH, et al. Efficacy and safety of paracetamol for spinal pain and osteoarthritis: systematic review and meta-analysis of randomised placebo controlled trials. BMJ. 2015;350:h1225.
88. Enthoven WT, Roelofs PD, Deyo RA, et al. Non-steroidal anti-inflammatory drugs for chronic low back pain. Cochrane Database Syst Rev. 2016;2:CD012087.
89. White AP, Arnold PM, Norvell DC, et al. Pharmacologic management of chronic low back pain: synthesis of the evidence. Spine (Phila Pa 1976). 2011;36:S131-S143.
Case 1 Lola, 28, has a history of muscular aches and joint pain throughout her body, fatigue, and mental fogginess. A rheumatologist diagnosed fibromyalgia, but Lola just moved to your town and is establishing care. She is feeling desperate because her pain has worsened and the medication previously prescribed (gabapentin 300 mg tid) is no longer working. She asks to try oxycodone.
Case 2 Matt is a 59-year-old truck driver with severe hip osteoarthritis (OA). His orthopedist recommended against hip replacement at this time because of his young age and a heart condition that makes him high risk. His pain makes sitting for long periods very difficult. He presents to you for help because he is worried he will be unable to continue working.
Case 3 Keith is a 56-year-old construction worker who has been experiencing back pain for many years. The pain has become more debilitating over time; it is now constant, and Keith can hardly make it through his work day. He has been getting hydrocodone/acetaminophen from urgent care centers and emergency departments, but he isn’t sure it is helping and is coming to you to assume his pain management.
Chronic pain (defined as > 3 mo in duration) is a complex, heterogeneous condition affecting an estimated 116 million US adults.1 Much of the management of chronic pain occurs in primary care settings, placing family practice providers (FPPs) on the frontlines of two epidemics: that of chronic pain and that of the abuse and misuse of opioid pain medications.
To improve communication about the risks and benefits of opioid therapy and the safety and effectiveness of pain treatments in general, many professional organizations, health care institutions, and recently the CDC, have published guidelines on the use of opioids for nonmalignant chronic pain.2 With these guidelines in mind—and in light of the latest evidence—we propose the following paradigm for the treatment of chronic pain. A critical aspect is determining the underlying pathophysiology of a patient’s pain in order to develop a well-rounded, multimodal, evidence-based treatment plan. Detailed here is the application of this approach to the treatment of three common diagnoses: fibromyalgia, osteoarthritis, and low back pain.
LOOK TO THE CENTRAL AND PERIPHERAL NERVOUS SYSTEM
Acute pain begins with activation of peripheral nociceptors at the site of injury. This causes depolarization up the spinal cord and through the brain stem to higher cortical centers where the pain is perceived and localized. Descending neural pathways transport both excitatory and inhibitory information from the brain to the periphery via the spinal cord, which either increases or decreases the perception of pain.3
When damage/injury doesn’t correlate with the perception of pain
Until recently, it was assumed that chronic pain worked much the same way as acute pain and was caused by ongoing nociceptive input in the periphery, but research has shown us that the central nervous system (CNS) can play a large role in the modulation of nociception. This new understanding comes from the lack of evidence pointing to any pain state in which the degree of nociceptive input correlates with the degree of pain experienced.
For most patients with chronic pain, regardless of their diagnosis, there is some degree of alteration in the processing of nociceptive signals by the CNS contributing to the experience of pain.4 This alteration is thought to result from peripheral nociceptive signaling persisting past the point of tissue healing, leading to a hypersensitivity of nerve fibers, which then continue to respond to low, or absent, sensory stimuli.
Central sensitization is when this hypersensitivity develops in the superficial, deep, and ventral cord nerves. When this happens, pain is often accompanied by systemic symptoms such as fatigue and slowed cognitive processing, often with little to no actual stimulation of the peripheral nociceptors.3
Table 1 lists the possible mechanisms of pain, which can be broken down into four categories: peripheral nociceptive (inflammatory or mechanical), peripheral neuropathic (underlying damage to a peripheral nerve), central (when the CNS is the primary entity involved in maintaining the pain), or any combination of the three.4
As pain becomes chronic, multiple mechanisms overlap
It is important to remember that for any single pain diagnosis, there is likely to be—at least initially—a principle underlying mechanism generating the pain. But as the pain becomes chronic, an overlap of multiple mechanisms develops, with central sensitization often playing a more dominant role than peripheral stimulation (regardless of the diagnosis).
For example, in a patient with rheumatoid arthritis (RA), peripheral nociceptive input (in the form of inflammation) is likely the initial mechanism at work, but as time goes on, central processing becomes more involved. The patient may then begin to experience pain that is disproportionate to what is generally expected with RA and may develop other somatic symptoms. The diagnosis then becomes pain primarily related to RA with central sensitization, and both need to be addressed in a treatment plan. In rheumatic conditions, comorbid fibromyalgia (indicative of central sensitization) is thought to occur in 15% to 30% of patients.5
FPPs can utilize the underlying mechanisms to cut across diagnostic labels and tailor treatments to those that are most likely to be effective. For a patient with more prominent peripheral involvement, a procedural intervention such as injections or surgery alone may suffice, whereas a broader approach including psychotherapy, medications, exercise, and other lifestyle interventions may be necessary for a patient with pain caused predominantly by central sensitization.
Addressing both peripheral and central components is essential. One prospective, observational cohort study of more than 600 patients scheduled for unilateral total knee or total hip arthroplasty found that patients with a higher degree of centralization of pain (measured by widespread pain index and modified fibromyalgia screening scales) were less likely to report improvement in the affected body part and in overall body pain following surgery.6,7
There is a high degree of overlap among many of the chronic pain syndromes (fibromyalgia, irritable bowel syndrome, interstitial cystitis, chronic headaches) that have been found to have a central sensitization component.8 Providers of primary care are aptly positioned to recognize central sensitization as the underlying pathology and target treatment effectively.
TAILOR TREATMENT TO THE UNDERLYING MECHANISMS OF PAIN
As with any chronic condition, a thorough workup (complete with history, physical exam, and diagnostic testing, as appropriate) is indicated. In the setting of chronic pain, it’s important to identify the primary mechanism, as well as secondary factors that may contribute to the patient’s pain, before developing your treatment plan. These secondary factors may include co-occurring affect disorders, a history of trauma, poor sleep, and tobacco use.9-12 A history of trauma, for example, co-exists with many pain syndromes. For these patients, central sensitization is responsible for much of their pain. As a result, traditional cognitive behavioral therapy (CBT) may not be the best option because of its focus on accepting pain as a chronic diagnosis; more trauma-focused treatments, such as those dealing in emotional awareness and understanding of the CNS’s role in chronic pain, need to be considered.13
Three common conditions. Below we present evidence-based treatment approaches for conditions typically associated with each of the major mechanisms of chronic pain: fibromyalgia (central sensitization), OA (peripheral nociceptive), and low back pain (mixed pain state).
Fibromyalgia: a case of central sensitization
Fibromyalgia is a hallmark diagnosis for patients in whom central sensitization is the dominant cause of pain. They usually present with widespread, diffuse pain and somatic symptoms such as fatigue, memory difficulties, and poor sleep quality.8 When explaining the pain mechanism to patients, it may be useful to use the analogy of a volume control dial that is stuck in the “high” position and can’t be turned down.
Genes, the environment, and neurotransmitters play a role. The origin of the pain amplification process is believed to be multifactorial.
Genetic factors are thought to contribute to a predisposition for amplification. To date, five sets of genes have been implicated in increased sensitivity to pain leading to increased risk of the development of chronic pain during a patient’s lifetime.14-19
Environmental factors (eg, early life trauma, physical trauma especially to the trunk, certain infections such as Lyme disease and Epstein-Barr virus, and emotional stress) may trigger or exacerbate symptoms.8 Of note: Only about 5% to 10% of people who experience these triggers actually develop a chronic pain state, while the rest regain their baseline health.4 This raises the question of whether there is a point during an acute pain episode in which one can intervene and prevent the acute pain from becoming chronic in those at higher risk.4
Imbalances of neurotransmitters (high glutamate; low norepinephrine, serotonin, and gamma-aminobutyric acid [GABA]) play a role in central amplification.20-22 These substances not only affect sensory transmission, but also control levels of alertness, sleep, mood, and memory.
The diagnostic criteria for fibromyalgia were modified in 2011 to remove the tender point examination and to add somatic symptoms.6 These criteria can be useful in the clinical setting in identifying not only fibromyalgia itself but also the degree of “fibromyalgianess” a patient has, which is an indicator of how large a role the centralization process plays in the maintenance of chronic pain.23,24
Treatment: multimodal and patient empowering. Evidence-based treatment options for fibromyalgia, as well as other conditions for which there is a high degree of centralized pain, can be found in Table 2.25-36 Multimodal treatment, with an emphasis on patient knowledge and empowerment, is generally thought to be the most beneficial.25,37 Treatment should almost always include CBT and exercise/activity therapies, which have high degrees of efficacy with few adverse effects.26,29
In terms of medication, centrally-acting agents (tricyclic antidepressants, serotonin norepinephrine reuptake inhibitors [SNRIs], and alpha 2 delta ligands) are the most effective. There is little to no data showing benefit from anti-inflammatories or opioids in the setting of fibromyalgia. There is some data to suggest that combination therapy, for example with an SNRI (milnacipran) and an alpha 2 delta ligand (pregabalin), may provide more benefit than treating with pregabalin alone.38
Complementary and alternative therapies (eg, yoga, chiropractic care, acupuncture, massage) are being studied more, and while evidence is only preliminary in terms of efficacy, there is increasing emphasis being placed on the need for patients with chronic pain to shift their treatment expectations to greater acceptance of pain and the need for ongoing self-care.28
OA: an example of peripheral nociceptive pain
OA is a condition long thought to be characterized by damage to the cartilage and bone; however, as with many other pain diagnoses, there is frequently little correlation between damage seen on radiographs and the amount of pain that patients experience.
One study analyzed data on almost 7,000 patients from the National Health and Nutrition Examination Survey (NHANES I) and found that between 30% and 50% of OA patients with moderate to severe radiographic changes were asymptomatic, and 10% of those with moderate to severe pain had normal radiographs or only mild changes.39 Research is showing that many factors may contribute to this discrepancy, including the typical “wear and tear” of the disease, subacute levels of inflammation that can lead to peripheral sensitization, and, in some patients, a centralized pain component.40 The patients with more centralized pain often have pain that is disproportionate to radiographic evidence, as well as more somatic symptoms, such as fatigue, sleep disturbance, and memory issues.41
Treatment should be multimodal and include interventions targeted at halting the progression of damage as well as palliation of pain. All treatment plans for OA should also include exercise, weight reduction, and self-management, in addition to pharmacologic interventions, to reduce both the micro-inflammation and the centralized pain component (when present). Intra-articular injections of various types have been studied with some having more efficacy in pain reduction and functional improvement than others.42-45 See Table 3 for a summary of evidence-based treatment options.42-61
Low back pain: a mixed pain state
Low back pain (LBP) has been recognized as a mixed pain state for quite some time. While some patients may experience purely nociceptive and/or neuropathic pain, most cases are nonspecific, with patients experiencing varying degrees of nociceptive (myofascial LBP), neuropathic (lumbar radiculopathy), and central sensitization pain.62,63 Evidence for centralized pain is demonstrated in studies showing hyperalgesia, augmented central pain processing, involvement of the emotional brain, and delayed recovery influenced by poor coping strategies.64-67
When developing a treatment plan for a patient with chronic LBP, remember that the pain derives from a complex combination of pathophysiologic contributors. Identifying where a patient lies on the pain centralization spectrum can help you tailor treatment.
In one study of 548 patients presenting to a tertiary pain clinic with primary spine pain diagnoses, 42% met diagnostic criteria for fibromyalgia.68 Compared to criteria-negative patients, these patients tended to be younger, unemployed, and receiving compensation; they had greater pain intensity, pain interference, and used stronger words to describe their neuropathic pain, as well as having higher levels of depression/anxiety and a lower level of physical function.
Because LBP is a condition with high prevalence and associated disability, many clinical boards have created guidelines for management. These guidelines tend to vary in the strength of evidence used, and the extent to which they are followed in clinical practice remains largely unknown. Recommendations frequently discourage the use of ultrasound/electrotherapy, but many encourage short-term use of medications, supervised exercise therapy, CBT, and multidisciplinary treatment.
Guidelines tend to differ most widely with regard to recommendations for spinal manipulation and specific drug therapies.69 The classes of drugs that may be most useful when centralized pain is present include the SNRIs and the alpha 2 delta calcium channel ligands.4 See Table 4 for a summary of evidence-based treatment options.70-89
Case 1 Lola is started on amitriptyline 25 mg at bedtime, which improves her fatigue and cognitive symptoms. During monthly office visits, her FPP educates her about the pathophysiology of fibromyalgia and uses motivational interviewing to get her slowly moving and increasing her activity level. She is weaned off the gabapentin previously prescribed, as her symptoms stabilize and improve.
Case 2 Matt is sent for a steroid injection, which decreases his pain temporarily. During this time, he begins physical therapy; slowly, with increased movement, his function improves. A trial of duloxetine provides pain relief; that combined with intermittent NSAIDs has allowed Matt to maintain his function and his job.
Case 3 Because Keith was only taking the narcotics intermittently and wasn’t certain they were helping, CBT was sufficient to wean him off the medication without any worsening of his pain in the process. By participating in physical therapy, he has learned how to perform certain tasks at his job without pain or injury. He uses NSAIDs as needed for pain.
The authors thank Drs. Daniel Clauw (University of Michigan, Ann Arbor) and Martha Rumschlag (Providence Family Medicine Residency Program, Southfield, Michigan), for their valuable contributions to this article.
Case 1 Lola, 28, has a history of muscular aches and joint pain throughout her body, fatigue, and mental fogginess. A rheumatologist diagnosed fibromyalgia, but Lola just moved to your town and is establishing care. She is feeling desperate because her pain has worsened and the medication previously prescribed (gabapentin 300 mg tid) is no longer working. She asks to try oxycodone.
Case 2 Matt is a 59-year-old truck driver with severe hip osteoarthritis (OA). His orthopedist recommended against hip replacement at this time because of his young age and a heart condition that makes him high risk. His pain makes sitting for long periods very difficult. He presents to you for help because he is worried he will be unable to continue working.
Case 3 Keith is a 56-year-old construction worker who has been experiencing back pain for many years. The pain has become more debilitating over time; it is now constant, and Keith can hardly make it through his work day. He has been getting hydrocodone/acetaminophen from urgent care centers and emergency departments, but he isn’t sure it is helping and is coming to you to assume his pain management.
Chronic pain (defined as > 3 mo in duration) is a complex, heterogeneous condition affecting an estimated 116 million US adults.1 Much of the management of chronic pain occurs in primary care settings, placing family practice providers (FPPs) on the frontlines of two epidemics: that of chronic pain and that of the abuse and misuse of opioid pain medications.
To improve communication about the risks and benefits of opioid therapy and the safety and effectiveness of pain treatments in general, many professional organizations, health care institutions, and recently the CDC, have published guidelines on the use of opioids for nonmalignant chronic pain.2 With these guidelines in mind—and in light of the latest evidence—we propose the following paradigm for the treatment of chronic pain. A critical aspect is determining the underlying pathophysiology of a patient’s pain in order to develop a well-rounded, multimodal, evidence-based treatment plan. Detailed here is the application of this approach to the treatment of three common diagnoses: fibromyalgia, osteoarthritis, and low back pain.
LOOK TO THE CENTRAL AND PERIPHERAL NERVOUS SYSTEM
Acute pain begins with activation of peripheral nociceptors at the site of injury. This causes depolarization up the spinal cord and through the brain stem to higher cortical centers where the pain is perceived and localized. Descending neural pathways transport both excitatory and inhibitory information from the brain to the periphery via the spinal cord, which either increases or decreases the perception of pain.3
When damage/injury doesn’t correlate with the perception of pain
Until recently, it was assumed that chronic pain worked much the same way as acute pain and was caused by ongoing nociceptive input in the periphery, but research has shown us that the central nervous system (CNS) can play a large role in the modulation of nociception. This new understanding comes from the lack of evidence pointing to any pain state in which the degree of nociceptive input correlates with the degree of pain experienced.
For most patients with chronic pain, regardless of their diagnosis, there is some degree of alteration in the processing of nociceptive signals by the CNS contributing to the experience of pain.4 This alteration is thought to result from peripheral nociceptive signaling persisting past the point of tissue healing, leading to a hypersensitivity of nerve fibers, which then continue to respond to low, or absent, sensory stimuli.
Central sensitization is when this hypersensitivity develops in the superficial, deep, and ventral cord nerves. When this happens, pain is often accompanied by systemic symptoms such as fatigue and slowed cognitive processing, often with little to no actual stimulation of the peripheral nociceptors.3
Table 1 lists the possible mechanisms of pain, which can be broken down into four categories: peripheral nociceptive (inflammatory or mechanical), peripheral neuropathic (underlying damage to a peripheral nerve), central (when the CNS is the primary entity involved in maintaining the pain), or any combination of the three.4
As pain becomes chronic, multiple mechanisms overlap
It is important to remember that for any single pain diagnosis, there is likely to be—at least initially—a principle underlying mechanism generating the pain. But as the pain becomes chronic, an overlap of multiple mechanisms develops, with central sensitization often playing a more dominant role than peripheral stimulation (regardless of the diagnosis).
For example, in a patient with rheumatoid arthritis (RA), peripheral nociceptive input (in the form of inflammation) is likely the initial mechanism at work, but as time goes on, central processing becomes more involved. The patient may then begin to experience pain that is disproportionate to what is generally expected with RA and may develop other somatic symptoms. The diagnosis then becomes pain primarily related to RA with central sensitization, and both need to be addressed in a treatment plan. In rheumatic conditions, comorbid fibromyalgia (indicative of central sensitization) is thought to occur in 15% to 30% of patients.5
FPPs can utilize the underlying mechanisms to cut across diagnostic labels and tailor treatments to those that are most likely to be effective. For a patient with more prominent peripheral involvement, a procedural intervention such as injections or surgery alone may suffice, whereas a broader approach including psychotherapy, medications, exercise, and other lifestyle interventions may be necessary for a patient with pain caused predominantly by central sensitization.
Addressing both peripheral and central components is essential. One prospective, observational cohort study of more than 600 patients scheduled for unilateral total knee or total hip arthroplasty found that patients with a higher degree of centralization of pain (measured by widespread pain index and modified fibromyalgia screening scales) were less likely to report improvement in the affected body part and in overall body pain following surgery.6,7
There is a high degree of overlap among many of the chronic pain syndromes (fibromyalgia, irritable bowel syndrome, interstitial cystitis, chronic headaches) that have been found to have a central sensitization component.8 Providers of primary care are aptly positioned to recognize central sensitization as the underlying pathology and target treatment effectively.
TAILOR TREATMENT TO THE UNDERLYING MECHANISMS OF PAIN
As with any chronic condition, a thorough workup (complete with history, physical exam, and diagnostic testing, as appropriate) is indicated. In the setting of chronic pain, it’s important to identify the primary mechanism, as well as secondary factors that may contribute to the patient’s pain, before developing your treatment plan. These secondary factors may include co-occurring affect disorders, a history of trauma, poor sleep, and tobacco use.9-12 A history of trauma, for example, co-exists with many pain syndromes. For these patients, central sensitization is responsible for much of their pain. As a result, traditional cognitive behavioral therapy (CBT) may not be the best option because of its focus on accepting pain as a chronic diagnosis; more trauma-focused treatments, such as those dealing in emotional awareness and understanding of the CNS’s role in chronic pain, need to be considered.13
Three common conditions. Below we present evidence-based treatment approaches for conditions typically associated with each of the major mechanisms of chronic pain: fibromyalgia (central sensitization), OA (peripheral nociceptive), and low back pain (mixed pain state).
Fibromyalgia: a case of central sensitization
Fibromyalgia is a hallmark diagnosis for patients in whom central sensitization is the dominant cause of pain. They usually present with widespread, diffuse pain and somatic symptoms such as fatigue, memory difficulties, and poor sleep quality.8 When explaining the pain mechanism to patients, it may be useful to use the analogy of a volume control dial that is stuck in the “high” position and can’t be turned down.
Genes, the environment, and neurotransmitters play a role. The origin of the pain amplification process is believed to be multifactorial.
Genetic factors are thought to contribute to a predisposition for amplification. To date, five sets of genes have been implicated in increased sensitivity to pain leading to increased risk of the development of chronic pain during a patient’s lifetime.14-19
Environmental factors (eg, early life trauma, physical trauma especially to the trunk, certain infections such as Lyme disease and Epstein-Barr virus, and emotional stress) may trigger or exacerbate symptoms.8 Of note: Only about 5% to 10% of people who experience these triggers actually develop a chronic pain state, while the rest regain their baseline health.4 This raises the question of whether there is a point during an acute pain episode in which one can intervene and prevent the acute pain from becoming chronic in those at higher risk.4
Imbalances of neurotransmitters (high glutamate; low norepinephrine, serotonin, and gamma-aminobutyric acid [GABA]) play a role in central amplification.20-22 These substances not only affect sensory transmission, but also control levels of alertness, sleep, mood, and memory.
The diagnostic criteria for fibromyalgia were modified in 2011 to remove the tender point examination and to add somatic symptoms.6 These criteria can be useful in the clinical setting in identifying not only fibromyalgia itself but also the degree of “fibromyalgianess” a patient has, which is an indicator of how large a role the centralization process plays in the maintenance of chronic pain.23,24
Treatment: multimodal and patient empowering. Evidence-based treatment options for fibromyalgia, as well as other conditions for which there is a high degree of centralized pain, can be found in Table 2.25-36 Multimodal treatment, with an emphasis on patient knowledge and empowerment, is generally thought to be the most beneficial.25,37 Treatment should almost always include CBT and exercise/activity therapies, which have high degrees of efficacy with few adverse effects.26,29
In terms of medication, centrally-acting agents (tricyclic antidepressants, serotonin norepinephrine reuptake inhibitors [SNRIs], and alpha 2 delta ligands) are the most effective. There is little to no data showing benefit from anti-inflammatories or opioids in the setting of fibromyalgia. There is some data to suggest that combination therapy, for example with an SNRI (milnacipran) and an alpha 2 delta ligand (pregabalin), may provide more benefit than treating with pregabalin alone.38
Complementary and alternative therapies (eg, yoga, chiropractic care, acupuncture, massage) are being studied more, and while evidence is only preliminary in terms of efficacy, there is increasing emphasis being placed on the need for patients with chronic pain to shift their treatment expectations to greater acceptance of pain and the need for ongoing self-care.28
OA: an example of peripheral nociceptive pain
OA is a condition long thought to be characterized by damage to the cartilage and bone; however, as with many other pain diagnoses, there is frequently little correlation between damage seen on radiographs and the amount of pain that patients experience.
One study analyzed data on almost 7,000 patients from the National Health and Nutrition Examination Survey (NHANES I) and found that between 30% and 50% of OA patients with moderate to severe radiographic changes were asymptomatic, and 10% of those with moderate to severe pain had normal radiographs or only mild changes.39 Research is showing that many factors may contribute to this discrepancy, including the typical “wear and tear” of the disease, subacute levels of inflammation that can lead to peripheral sensitization, and, in some patients, a centralized pain component.40 The patients with more centralized pain often have pain that is disproportionate to radiographic evidence, as well as more somatic symptoms, such as fatigue, sleep disturbance, and memory issues.41
Treatment should be multimodal and include interventions targeted at halting the progression of damage as well as palliation of pain. All treatment plans for OA should also include exercise, weight reduction, and self-management, in addition to pharmacologic interventions, to reduce both the micro-inflammation and the centralized pain component (when present). Intra-articular injections of various types have been studied with some having more efficacy in pain reduction and functional improvement than others.42-45 See Table 3 for a summary of evidence-based treatment options.42-61
Low back pain: a mixed pain state
Low back pain (LBP) has been recognized as a mixed pain state for quite some time. While some patients may experience purely nociceptive and/or neuropathic pain, most cases are nonspecific, with patients experiencing varying degrees of nociceptive (myofascial LBP), neuropathic (lumbar radiculopathy), and central sensitization pain.62,63 Evidence for centralized pain is demonstrated in studies showing hyperalgesia, augmented central pain processing, involvement of the emotional brain, and delayed recovery influenced by poor coping strategies.64-67
When developing a treatment plan for a patient with chronic LBP, remember that the pain derives from a complex combination of pathophysiologic contributors. Identifying where a patient lies on the pain centralization spectrum can help you tailor treatment.
In one study of 548 patients presenting to a tertiary pain clinic with primary spine pain diagnoses, 42% met diagnostic criteria for fibromyalgia.68 Compared to criteria-negative patients, these patients tended to be younger, unemployed, and receiving compensation; they had greater pain intensity, pain interference, and used stronger words to describe their neuropathic pain, as well as having higher levels of depression/anxiety and a lower level of physical function.
Because LBP is a condition with high prevalence and associated disability, many clinical boards have created guidelines for management. These guidelines tend to vary in the strength of evidence used, and the extent to which they are followed in clinical practice remains largely unknown. Recommendations frequently discourage the use of ultrasound/electrotherapy, but many encourage short-term use of medications, supervised exercise therapy, CBT, and multidisciplinary treatment.
Guidelines tend to differ most widely with regard to recommendations for spinal manipulation and specific drug therapies.69 The classes of drugs that may be most useful when centralized pain is present include the SNRIs and the alpha 2 delta calcium channel ligands.4 See Table 4 for a summary of evidence-based treatment options.70-89
Case 1 Lola is started on amitriptyline 25 mg at bedtime, which improves her fatigue and cognitive symptoms. During monthly office visits, her FPP educates her about the pathophysiology of fibromyalgia and uses motivational interviewing to get her slowly moving and increasing her activity level. She is weaned off the gabapentin previously prescribed, as her symptoms stabilize and improve.
Case 2 Matt is sent for a steroid injection, which decreases his pain temporarily. During this time, he begins physical therapy; slowly, with increased movement, his function improves. A trial of duloxetine provides pain relief; that combined with intermittent NSAIDs has allowed Matt to maintain his function and his job.
Case 3 Because Keith was only taking the narcotics intermittently and wasn’t certain they were helping, CBT was sufficient to wean him off the medication without any worsening of his pain in the process. By participating in physical therapy, he has learned how to perform certain tasks at his job without pain or injury. He uses NSAIDs as needed for pain.
The authors thank Drs. Daniel Clauw (University of Michigan, Ann Arbor) and Martha Rumschlag (Providence Family Medicine Residency Program, Southfield, Michigan), for their valuable contributions to this article.
1. Institute of Medicine (US) Committee on Advancing Pain Research, Care, and Education. Relieving pain in America: a blueprint for transforming prevention, care, education, and research. Washington (DC): National Academies Press (US); 2011.
2. Dowell D, Haegerich TM, Chou R. CDC Guideline for Prescribing Opioids for Chronic Pain—United States, 2016. MMWR Recomm Rep. 2016;65:1-49.
3. Aronoff GM. What do we know about the pathophysiology of chronic pain? Implications for treatment considerations. Med Clin North Am. 2016;100:31-42.
4. Clauw DJ. Diagnosing and treating chronic musculoskeletal pain based on the underlying mechanism(s). Best Pract Res Clin Rheumatol. 2015;29:6-19.
5. Clauw DJ, Katz P. The overlap between fibromyalgia and inflammatory rheumatic disease: when and why does it occur? J Clin Rheumatol. 1995;1:335-342.
6. Wolfe F, Clauw DJ, Fitzcharles MA, et al. Fibromyalgia criteria and severity scales for clinical and epidemiological studies: a modification of the ACR Preliminary Diagnostic Criteria for Fibromyalgia. J Rheumatol. 2011;38:1113-1122.
7. Brummett CM, Urquhart AG, Hassett AL, et al. Characteristics of fibromyalgia independently predict poorer long-term analgesic outcomes following total knee and hip arthroplasty. Arthritis Rheumatol. 2015;67:1386-1394.
8. Ablin K, Clauw DJ. From fibrositis to functional somatic syndromes to a bell-shaped curve of pain and sensory sensitivity: evolution of a clinical construct. Rheum Dis Clin North Am. 2009;35:233-251.
9. Giesecke T, Gracely RH, Williams DA, et al. The relationship between depression, clinical pain, and experimental pain in a chronic pain cohort. Arthritis Rheum. 2005;52:1577-1584.
10. Tesarz J, Eich W, Treede RD, et al. Altered pressure pain thresholds and increased wind-up in adult chronic back pain patients with a history of childhood maltreatment: a quantitative sensory testing study. Pain. 2016;157:1799-1809.
11. Finan PH, Goodin BR, Smith MT. The association of sleep and pain: an update and a path forward. J Pain. 2013;14:1539-1552.
12. Shi Y, Weingarten TN, Mantilla CB, et al. Smoking and pain: pathophysiology and clinical implications. Anesthesiology. 2010;113:977-992.
13. Burger AJ, Lumley MA, Carty JN, et al. The effects of a novel psychological attribution and emotional awareness and expression therapy for chronic musculoskeletal pain: a preliminary, uncontrolled trial. J Psychosom Res. 2016;81:1-8.
14. Zubieta JK, Heitzeg MM, Smith YR, et al. COMT val158met genotype affects mu-opioid neurotransmitter responses to a pain stressor. Science. 2003;299:1240-1243.
15. van Meurs JB, Uitterlinden AG, Stolk L, et al. A functional polymorphism in the catechol-O-methyltransferase gene is associated with osteoarthritis-related pain. Arthritis Rheum. 2009;60:628-629.
16. McLean SA, Diatchenko L, Lee YM, et al. Catechol O-methyltransferase haplotype predicts immediate musculoskeletal neck pain and psychological symptoms after motor vehicle collision. J Pain. 2011;12:101-107.
17. Costigan M, Belfer I, Griffin RS, et al. Multiple chronic pain states are associated with a common amino acid-changing allele in KCNS1. Brain. 2010;133:2519-2527.
18. Tegeder I, Costigan M, Griffin RS, et al. GTP cyclohydrolase and tetrahydrobiopterin regulate pain sensitivity and persistence. Nat Med. 2006;12:1269-1277.
19. Amaya F, Wang H, Costigan M, et al. The voltage-gated sodium channel Na(v)1.9 is an effector of peripheral inflammatory pain hypersensitivity. J Neurosci. 2006;26:12852-12860.
20. Harris RE, Napadow V, Huggins JP, et al. Pregabalin rectifies abberrant brain chemistry, connectivity, and functional responses in chronic pain patients. Anesthesiology. 2013;119:1453-1464.
21. Russell IJ, Vaeroy H, Javors M, et al. Cerebrospinal fluid biogenic amine metabolites in fibromyalgia/fibrositis syndrome and rheumatoid arthritis. Arthritis Rheum. 1992;35:550-556.
22. Foerster BR, Petrou M, Edden RAE, et al. Reduced insular gamma-aminobutyric acid in fibromyalgia. Arthritis Rheum. 2012;64:579-583.
23. Clauw DJ. Fibromyalgia: a clinical review. JAMA. 2014;311:1547-1555.
24. Wolfe F. Fibromyalgianess. Arthritis Rheum. 2009;61:715-716.
25. Hauser W, Bernardy K, Arnold B, et al. Efficacy of multicomponent treatment in fibromyalgia syndrome: a meta-analysis of randomized controlled clinical trials. Arthritis Rheum. 2009;61:216-224.
26. Hauser W, Klose P, Langhorst J, et al. Efficacy of different types of aerobic exercise in fibromyalgia syndrome: a systematic review and meta-analysis of randomised controlled trials. Arthritis Res Ther. 2010;12:R79.
27. Porter NS, Jason LA, Boulton A, et al. Alternative medical interventions used in the treatment and management of myalgic encephalomyelitis/chronic fatigue syndrome and fibromyalgia. J Altern Complement Med. 2010;16:235-249.
28. Eaves ER, Sherman KJ, Ritenbaugh C, et al. A qualitative study of changes in expectations over time among patients with chronic low back pain seeking four CAM therapies. BMC Complement Altern Med. 2015;15:12.
29. Bernardy K, Fuber N, Kollner V, et al. Efficacy of cognitive-behavioral therapies in fibromyalgia syndrome: a systematic review and metaanalysis of randomized controlled trials. J Rheumatol. 2010;37:1991-2005.
30. Arnold LM, Keck PE Jr, Welge JA. Antidepressant treatment of fibromyalgia. A meta-analysis and review. Psychosomatics. 2000;41:104-113.
31. Moldofsky H, Harris HW, Archambault WT, et al. Effects of bedtime very low dose cyclobenzaprine on symptoms and sleep physiology in patients with fibromyalgia syndrome: a double-blind randomized placebo-controlled study. J Rheumatol. 2011;38:2653-2663.
32. Arnold LM. Duloxetine and other antidepressants in the treatment of patients with fibromyalgia. Pain Med. 2007;(8 Suppl 2):S63-S74.
33. Häuser W, Bernardy K, Uceyler N, et al. Treatment of fibromyalgia syndrome with gabapentin and pregabalin—a meta-analysis of randomized controlled trials. Pain. 2009;145:69-81.
34. Gaskell H, Moore RA, Derry S, et al. Oxycodone for neuropathic pain and fibromyalgia in adults. Cochrane Database Syst Rev. 2014;Jun 23:CD010692.
35. MacLean AJ, Schwartz TL. Tramadol for the treatment of fibromyalgia. Expert Rev Neurother. 2015;15:469-475.
36. Younger J, Noor N, McCue R, et al. Low-dose naltrexone for the treatment of fibromyalgia: findings of a small, randomized, double-blind, placebo-controlled, counterbalanced, crossover trial assessing daily pain levels. Arthritis Rheum. 2013;65:529-538.
37. Camerini L, Schulz PJ, Nakamoto K. Differential effects of health knowledge and health empowerment over patients’ self-management and health outcomes: a cross-sectional evaluation. Patient Educ Couns. 2012;89:337-344.
38. Mease PJ, Farmer MV, Palmer RH, et al. Milnacipran combined with pregabalin in fibromyalgia: a randomized, open-label study evaluating the safety and efficacy of adding milnacipran in patients with incomplete response to pregabalin. Ther Adv Musculoskeletal Dis. 2013;5:113-126.
39. Hannan MT, Felson DT, Pincus T. Analysis of the discordance between radiographic changes and knee pain in osteoarthritis of the knee. J Rheumatol. 2000;27:1513-1517.
40. Daghestani HN, Kraus VB. Inflammatory biomarkers in osteoarthritis. Osteoarthritis Cartilage. 2015;23:1890-1896.
41. Fingleton C, Smart K, Moloney N, et al. Pain sensitization in people with knee osteoarthritis: a systematic review and meta-analysis. Osteoarthritis Cartilage. 2015;23:1043-1056.
42. Strand V, McIntyre LF, Beach WR, et al. Safety and efficacy of US-approved viscosupplements for knee osteoarthritis: a systematic review and meta-analysis of randomized, saline-controlled trials. J Pain Res. 2015;8:217-228.
43. Jüni P, Hari R, Rutjes AW, et al. Intra-articular corticosteroid for knee osteoarthritis. Cochrane Database Syst Rev. 2015:CD005328.
44. Meheux CJ, McCulloch PC, Lintner DM, et al. Efficacy of intra-articular platelet-rich plasma injections in knee osteoarthritis: a systematic review. Arthroscopy. 2016;32:495-505.
45. Wu T, Song HX, Dong Y, et al. Intra-articular injections of botulinum toxin a for refractory joint pain: a systematic review and meta-analysis. Clin Rehabil. 2017;31(4):435-443.
46. Jordan JL, Holden MA, Mason EE, et al. Interventions to improve adherence to exercise for chronic musculoskeletal pain in adults. Cochrane Database Syst Rev. 2010:CD005956.
47. Bodenheimer T, Lorig K, Holman H, et al. Patient self-management of chronic disease in primary care. JAMA. 2002;288:2469-2475.
48. Fransen M, McConnell S, Hernandez-Molina G, et al. Exercise for osteoarthritis of the hip. Cochrane Database Syst Rev. 2014:CD007912.
49. Bartels EM, Juhl CB, Christensen R, et al. Aquatic exercise for the treatment of knee and hip osteoarthritis. Cochrane Database Syst Rev. 2016;3:CD005523.
50. da Costa BR, Reichenbach S, Keller N, et al. Effectiveness of non-steroidal anti-inflammatory drugs for the treatment of pain in knee and hip osteoarthritis: a network meta-analysis. Lancet. 2016;387:2093-2105.
51. Myers J, Wielage RC, Han B, et al. The efficacy of duloxetine, non-steroidal anti-inflammatory drugs, and opioids in osteoarthritis: a systematic literature review and meta-analysis. BMC Musculoskelet Disord. 2014;15:76.
52. Berthelot JM, Darrieutort-Lafitte C, Le Goff B, et al. Strong opioids for noncancer pain due to musculoskeletal diseases: not more effective than acetaminophen or NSAIDs. Joint Bone Spine. 2015;82:397-401.
53. Clegg DO, Reda DJ, Harris CL, et al. Glucosamine, chondroitin sulfate, and the two in combination for painful knee osteoarthritis. N Engl J Med. 2006;354:795-808.
54. Wandel S, Jüni P, Tendal B, et al. Effects of glucosamine, chondroitin, or placebo in patients with osteoarthritis of hip or knee: network meta-analysis. BMJ. 2010;341:c4675.
55. Sawitzke AD, Shi H, Finco MF, et al. Clinical efficacy and safety of glucosamine, chondroitin sulphate, their combination, celecoxib or placebo taken to treat osteoarthritis of the knee: 2-year results from GAIT. Ann Rheum Dis. 2010;69:1459-1464.
56. Wu D, Huang Y, Gu Y, et al. Efficacies of different preparations of glucosamine for the treatment of osteoarthritis: a meta-analysis of randomised, double-blind, placebo-controlled trials. Int J Clin Pract. 2013;67:585-594.
57. Kahan A, Uebelhart D, De Vathaire F, et al. Long-term effects of chondroitins 4 and 6 sulfate on knee osteoarthritis: the study on osteoarthritis progression prevention, a two-year, randomized, double-blind, placebo-controlled trial. Arthritis Rheum. 2009;60:524-533.
58. Perkins K, Sahy W, Beckett RD. Efficacy of curcuma for treatment of osteoarthritis. J Evid Based Complementary Altern Med. 2017;22:156-165.
59. Clinton CM, O’Brien S, Law J, et al. Whole-foods, plant-based diet alleviates the symptoms of osteoarthritis. Arthritis. 2015;2015:708152.
60. Manyanga T, Froese M, Zarychanski R, et al. Pain management with acupuncture in osteoarthritis: a systematic review and meta-analysis. BMC Complement Altern Med. 2014;14:312.
61. Vickers AJ, Cronin AM, Maschino AC, et al. Acupuncture for chronic pain: individual patient data meta-analysis. Arch Intern Med. 2012;172:1444-1453.
62. Nijs J, Apeldoorn A, Hallegraeff H, et al. Low back pain: guidelines for the clinical classification of predominant neuropathic, nociceptive, or central sensitization pain. Pain Physician. 2015;18:E333-E346.
63. Fishbain DA, Cole B, Lewis JE, et al. What is the evidence that neuropathic pain is present in chronic low back pain and soft tissue syndromes? An evidence-based structured review. Pain Med. 2014;15:4-15.
64. Hübscher M, Moloney N, Rebbeck T, et al. Contributions of mood, pain catastrophizing, and cold hyperalgesia in acute and chronic low back pain: a comparison with pain-free controls. Clin J Pain. 2014;30:886-893.
65. Giesecke T, Gracely RH, Grant MA, et al. Evidence of augmented central pain processing in idiopathic chronic low back pain. Arthritis Rheum. 2004;50:613-623.
66. Baliki MN, Chialvo DR, Geha PY, et al. Chronic pain and the emotional brain: specific brain activity associated with spontaneous fluctuations of intensity of chronic back pain. J Neurosci. 2006;26:12165-12173.
67. Wertli MM, Eugster R, Held U, et al. Catastrophizing-a prognostic factor for outcome in patients with low back pain: a systematic review. Spine J. 2014;14:2639-2657.
68. Brummett CM, Goesling J, Tsodikov A, et al. Prevalence of the fibromyalgia phenotype in patients with spine pain presenting to a tertiary care pain clinic and the potential treatment implications. Arthritis Rheum. 2013;65:3285-3292.
69. Koes BW, van Tulder M, Lin CW, et al. An updated overview of clinical guidelines for the management of non-specific low back pain in primary care. Eur Spine J. 2010;19:2075-2094.
70. Oliveira VC, Ferreira PH, Maher CG, et al. Effectiveness of self-management of low back pain: systematic review with meta-analysis. Arthritis Care Res. 2012;64:1739-1748.
71. Engers A, Jellema P, Wensing M, et al. Individual patient education for low back pain. Cochrane Database Syst Rev. 2008:CD004057.
72. Hayden JA, van Tulder MW, Malmivaara A, et al. Exercise therapy for treatment of non-specific low back pain. Cochrane Database Syst Rev. 2005:CD000335.
73. French SD, Cameron M, Walker BF, et al. Superficial heat or cold for low back pain. Cochrane Database Syst Rev. 2006:CD004750.
74. Franke H, Franke JD, Fryer G. Osteopathic manipulative treatment for nonspecific low back pain: a systematic review and meta-analysis. BMC Musculoskeletal Disord. 2014;15:286.
75. Franke H, Fryer G, Ostelo RW, et al. Muscle energy technique for non-specific low back pain. Cochrane Database Syst Rev. 2015:CD009852.
76. Oliphant D. Safety of spinal manipulation in the treatment of lumbar disk herniations: a systematic review and risk assessment. J Manipulative Physiol Ther. 2004:197-210.
77. Furlan AD, Giraldo M, Baskwill A, et al. Massage for low-back pain. Cochrane Database Syst Rev. 2015:CD001929.
78. Khadilkar A, Odebiyi DO, Brosseau L, et al. Transcutaneous electrical nerve stimulation (TENS) versus placebo for chronic low back pain. Cochrane Database Syst Rev. 2008:CD003008.
79. Ebadi S, Henschke N, Nakhostin Ansari N, et al. Therapeutic ultrasound for chronic low back pain. Cochrane Database Syst Rev. 2014:CD009169.
80. Furlan AD, van Tulder MW, Cherkin DC, et al. Acupuncture and dry-needling for low back pain. Cochrane Database Syst Rev. 2005:CD001351.
81. Chou R, Huffman LH. Nonpharmacologic therapies for acute and chronic low back pain: a review of the evidence for an American Pain Society/American College of Physicians clinical practice guideline. Ann Intern Med. 2007;147:492-504.
82. Sherman KJ, Cherkin DC, Erro J, et al. Comparing yoga, exercise, and a self-care book for chronic low back pain: a randomized, controlled trial. Ann Intern Med. 2005;143:849-856.
83. Cherkin DC, Sherman KJ, Balderson BH, et al. Effect of mindfulness-based stress reduction vs cognitive behavioral therapy or usual care on back pain and functional limitations in adults with chronic low back pain: a randomized clinical trial. JAMA. 2016;315:1240-1249.
84. Staal JB, de Bie R, de Vet HC, et al. Injection therapy for subacute and chronic low back pain. Cochrane Database Syst Rev. 2008:CD001824.
85. Chou R, Baisden J, Carragee EJ, et al. Surgery for low back pain: a review of the evidence for an American Pain Society Clinical Practice Guideline. Spine. 2009;34:1094-1109.
86. Felson D. Paracetamol is ineffective for spinal pain and knee and hip osteoarthritis. Evid Based Med. 2015;20:205.
87. Machado GC, Maher CG, Ferreira PH, et al. Efficacy and safety of paracetamol for spinal pain and osteoarthritis: systematic review and meta-analysis of randomised placebo controlled trials. BMJ. 2015;350:h1225.
88. Enthoven WT, Roelofs PD, Deyo RA, et al. Non-steroidal anti-inflammatory drugs for chronic low back pain. Cochrane Database Syst Rev. 2016;2:CD012087.
89. White AP, Arnold PM, Norvell DC, et al. Pharmacologic management of chronic low back pain: synthesis of the evidence. Spine (Phila Pa 1976). 2011;36:S131-S143.
1. Institute of Medicine (US) Committee on Advancing Pain Research, Care, and Education. Relieving pain in America: a blueprint for transforming prevention, care, education, and research. Washington (DC): National Academies Press (US); 2011.
2. Dowell D, Haegerich TM, Chou R. CDC Guideline for Prescribing Opioids for Chronic Pain—United States, 2016. MMWR Recomm Rep. 2016;65:1-49.
3. Aronoff GM. What do we know about the pathophysiology of chronic pain? Implications for treatment considerations. Med Clin North Am. 2016;100:31-42.
4. Clauw DJ. Diagnosing and treating chronic musculoskeletal pain based on the underlying mechanism(s). Best Pract Res Clin Rheumatol. 2015;29:6-19.
5. Clauw DJ, Katz P. The overlap between fibromyalgia and inflammatory rheumatic disease: when and why does it occur? J Clin Rheumatol. 1995;1:335-342.
6. Wolfe F, Clauw DJ, Fitzcharles MA, et al. Fibromyalgia criteria and severity scales for clinical and epidemiological studies: a modification of the ACR Preliminary Diagnostic Criteria for Fibromyalgia. J Rheumatol. 2011;38:1113-1122.
7. Brummett CM, Urquhart AG, Hassett AL, et al. Characteristics of fibromyalgia independently predict poorer long-term analgesic outcomes following total knee and hip arthroplasty. Arthritis Rheumatol. 2015;67:1386-1394.
8. Ablin K, Clauw DJ. From fibrositis to functional somatic syndromes to a bell-shaped curve of pain and sensory sensitivity: evolution of a clinical construct. Rheum Dis Clin North Am. 2009;35:233-251.
9. Giesecke T, Gracely RH, Williams DA, et al. The relationship between depression, clinical pain, and experimental pain in a chronic pain cohort. Arthritis Rheum. 2005;52:1577-1584.
10. Tesarz J, Eich W, Treede RD, et al. Altered pressure pain thresholds and increased wind-up in adult chronic back pain patients with a history of childhood maltreatment: a quantitative sensory testing study. Pain. 2016;157:1799-1809.
11. Finan PH, Goodin BR, Smith MT. The association of sleep and pain: an update and a path forward. J Pain. 2013;14:1539-1552.
12. Shi Y, Weingarten TN, Mantilla CB, et al. Smoking and pain: pathophysiology and clinical implications. Anesthesiology. 2010;113:977-992.
13. Burger AJ, Lumley MA, Carty JN, et al. The effects of a novel psychological attribution and emotional awareness and expression therapy for chronic musculoskeletal pain: a preliminary, uncontrolled trial. J Psychosom Res. 2016;81:1-8.
14. Zubieta JK, Heitzeg MM, Smith YR, et al. COMT val158met genotype affects mu-opioid neurotransmitter responses to a pain stressor. Science. 2003;299:1240-1243.
15. van Meurs JB, Uitterlinden AG, Stolk L, et al. A functional polymorphism in the catechol-O-methyltransferase gene is associated with osteoarthritis-related pain. Arthritis Rheum. 2009;60:628-629.
16. McLean SA, Diatchenko L, Lee YM, et al. Catechol O-methyltransferase haplotype predicts immediate musculoskeletal neck pain and psychological symptoms after motor vehicle collision. J Pain. 2011;12:101-107.
17. Costigan M, Belfer I, Griffin RS, et al. Multiple chronic pain states are associated with a common amino acid-changing allele in KCNS1. Brain. 2010;133:2519-2527.
18. Tegeder I, Costigan M, Griffin RS, et al. GTP cyclohydrolase and tetrahydrobiopterin regulate pain sensitivity and persistence. Nat Med. 2006;12:1269-1277.
19. Amaya F, Wang H, Costigan M, et al. The voltage-gated sodium channel Na(v)1.9 is an effector of peripheral inflammatory pain hypersensitivity. J Neurosci. 2006;26:12852-12860.
20. Harris RE, Napadow V, Huggins JP, et al. Pregabalin rectifies abberrant brain chemistry, connectivity, and functional responses in chronic pain patients. Anesthesiology. 2013;119:1453-1464.
21. Russell IJ, Vaeroy H, Javors M, et al. Cerebrospinal fluid biogenic amine metabolites in fibromyalgia/fibrositis syndrome and rheumatoid arthritis. Arthritis Rheum. 1992;35:550-556.
22. Foerster BR, Petrou M, Edden RAE, et al. Reduced insular gamma-aminobutyric acid in fibromyalgia. Arthritis Rheum. 2012;64:579-583.
23. Clauw DJ. Fibromyalgia: a clinical review. JAMA. 2014;311:1547-1555.
24. Wolfe F. Fibromyalgianess. Arthritis Rheum. 2009;61:715-716.
25. Hauser W, Bernardy K, Arnold B, et al. Efficacy of multicomponent treatment in fibromyalgia syndrome: a meta-analysis of randomized controlled clinical trials. Arthritis Rheum. 2009;61:216-224.
26. Hauser W, Klose P, Langhorst J, et al. Efficacy of different types of aerobic exercise in fibromyalgia syndrome: a systematic review and meta-analysis of randomised controlled trials. Arthritis Res Ther. 2010;12:R79.
27. Porter NS, Jason LA, Boulton A, et al. Alternative medical interventions used in the treatment and management of myalgic encephalomyelitis/chronic fatigue syndrome and fibromyalgia. J Altern Complement Med. 2010;16:235-249.
28. Eaves ER, Sherman KJ, Ritenbaugh C, et al. A qualitative study of changes in expectations over time among patients with chronic low back pain seeking four CAM therapies. BMC Complement Altern Med. 2015;15:12.
29. Bernardy K, Fuber N, Kollner V, et al. Efficacy of cognitive-behavioral therapies in fibromyalgia syndrome: a systematic review and metaanalysis of randomized controlled trials. J Rheumatol. 2010;37:1991-2005.
30. Arnold LM, Keck PE Jr, Welge JA. Antidepressant treatment of fibromyalgia. A meta-analysis and review. Psychosomatics. 2000;41:104-113.
31. Moldofsky H, Harris HW, Archambault WT, et al. Effects of bedtime very low dose cyclobenzaprine on symptoms and sleep physiology in patients with fibromyalgia syndrome: a double-blind randomized placebo-controlled study. J Rheumatol. 2011;38:2653-2663.
32. Arnold LM. Duloxetine and other antidepressants in the treatment of patients with fibromyalgia. Pain Med. 2007;(8 Suppl 2):S63-S74.
33. Häuser W, Bernardy K, Uceyler N, et al. Treatment of fibromyalgia syndrome with gabapentin and pregabalin—a meta-analysis of randomized controlled trials. Pain. 2009;145:69-81.
34. Gaskell H, Moore RA, Derry S, et al. Oxycodone for neuropathic pain and fibromyalgia in adults. Cochrane Database Syst Rev. 2014;Jun 23:CD010692.
35. MacLean AJ, Schwartz TL. Tramadol for the treatment of fibromyalgia. Expert Rev Neurother. 2015;15:469-475.
36. Younger J, Noor N, McCue R, et al. Low-dose naltrexone for the treatment of fibromyalgia: findings of a small, randomized, double-blind, placebo-controlled, counterbalanced, crossover trial assessing daily pain levels. Arthritis Rheum. 2013;65:529-538.
37. Camerini L, Schulz PJ, Nakamoto K. Differential effects of health knowledge and health empowerment over patients’ self-management and health outcomes: a cross-sectional evaluation. Patient Educ Couns. 2012;89:337-344.
38. Mease PJ, Farmer MV, Palmer RH, et al. Milnacipran combined with pregabalin in fibromyalgia: a randomized, open-label study evaluating the safety and efficacy of adding milnacipran in patients with incomplete response to pregabalin. Ther Adv Musculoskeletal Dis. 2013;5:113-126.
39. Hannan MT, Felson DT, Pincus T. Analysis of the discordance between radiographic changes and knee pain in osteoarthritis of the knee. J Rheumatol. 2000;27:1513-1517.
40. Daghestani HN, Kraus VB. Inflammatory biomarkers in osteoarthritis. Osteoarthritis Cartilage. 2015;23:1890-1896.
41. Fingleton C, Smart K, Moloney N, et al. Pain sensitization in people with knee osteoarthritis: a systematic review and meta-analysis. Osteoarthritis Cartilage. 2015;23:1043-1056.
42. Strand V, McIntyre LF, Beach WR, et al. Safety and efficacy of US-approved viscosupplements for knee osteoarthritis: a systematic review and meta-analysis of randomized, saline-controlled trials. J Pain Res. 2015;8:217-228.
43. Jüni P, Hari R, Rutjes AW, et al. Intra-articular corticosteroid for knee osteoarthritis. Cochrane Database Syst Rev. 2015:CD005328.
44. Meheux CJ, McCulloch PC, Lintner DM, et al. Efficacy of intra-articular platelet-rich plasma injections in knee osteoarthritis: a systematic review. Arthroscopy. 2016;32:495-505.
45. Wu T, Song HX, Dong Y, et al. Intra-articular injections of botulinum toxin a for refractory joint pain: a systematic review and meta-analysis. Clin Rehabil. 2017;31(4):435-443.
46. Jordan JL, Holden MA, Mason EE, et al. Interventions to improve adherence to exercise for chronic musculoskeletal pain in adults. Cochrane Database Syst Rev. 2010:CD005956.
47. Bodenheimer T, Lorig K, Holman H, et al. Patient self-management of chronic disease in primary care. JAMA. 2002;288:2469-2475.
48. Fransen M, McConnell S, Hernandez-Molina G, et al. Exercise for osteoarthritis of the hip. Cochrane Database Syst Rev. 2014:CD007912.
49. Bartels EM, Juhl CB, Christensen R, et al. Aquatic exercise for the treatment of knee and hip osteoarthritis. Cochrane Database Syst Rev. 2016;3:CD005523.
50. da Costa BR, Reichenbach S, Keller N, et al. Effectiveness of non-steroidal anti-inflammatory drugs for the treatment of pain in knee and hip osteoarthritis: a network meta-analysis. Lancet. 2016;387:2093-2105.
51. Myers J, Wielage RC, Han B, et al. The efficacy of duloxetine, non-steroidal anti-inflammatory drugs, and opioids in osteoarthritis: a systematic literature review and meta-analysis. BMC Musculoskelet Disord. 2014;15:76.
52. Berthelot JM, Darrieutort-Lafitte C, Le Goff B, et al. Strong opioids for noncancer pain due to musculoskeletal diseases: not more effective than acetaminophen or NSAIDs. Joint Bone Spine. 2015;82:397-401.
53. Clegg DO, Reda DJ, Harris CL, et al. Glucosamine, chondroitin sulfate, and the two in combination for painful knee osteoarthritis. N Engl J Med. 2006;354:795-808.
54. Wandel S, Jüni P, Tendal B, et al. Effects of glucosamine, chondroitin, or placebo in patients with osteoarthritis of hip or knee: network meta-analysis. BMJ. 2010;341:c4675.
55. Sawitzke AD, Shi H, Finco MF, et al. Clinical efficacy and safety of glucosamine, chondroitin sulphate, their combination, celecoxib or placebo taken to treat osteoarthritis of the knee: 2-year results from GAIT. Ann Rheum Dis. 2010;69:1459-1464.
56. Wu D, Huang Y, Gu Y, et al. Efficacies of different preparations of glucosamine for the treatment of osteoarthritis: a meta-analysis of randomised, double-blind, placebo-controlled trials. Int J Clin Pract. 2013;67:585-594.
57. Kahan A, Uebelhart D, De Vathaire F, et al. Long-term effects of chondroitins 4 and 6 sulfate on knee osteoarthritis: the study on osteoarthritis progression prevention, a two-year, randomized, double-blind, placebo-controlled trial. Arthritis Rheum. 2009;60:524-533.
58. Perkins K, Sahy W, Beckett RD. Efficacy of curcuma for treatment of osteoarthritis. J Evid Based Complementary Altern Med. 2017;22:156-165.
59. Clinton CM, O’Brien S, Law J, et al. Whole-foods, plant-based diet alleviates the symptoms of osteoarthritis. Arthritis. 2015;2015:708152.
60. Manyanga T, Froese M, Zarychanski R, et al. Pain management with acupuncture in osteoarthritis: a systematic review and meta-analysis. BMC Complement Altern Med. 2014;14:312.
61. Vickers AJ, Cronin AM, Maschino AC, et al. Acupuncture for chronic pain: individual patient data meta-analysis. Arch Intern Med. 2012;172:1444-1453.
62. Nijs J, Apeldoorn A, Hallegraeff H, et al. Low back pain: guidelines for the clinical classification of predominant neuropathic, nociceptive, or central sensitization pain. Pain Physician. 2015;18:E333-E346.
63. Fishbain DA, Cole B, Lewis JE, et al. What is the evidence that neuropathic pain is present in chronic low back pain and soft tissue syndromes? An evidence-based structured review. Pain Med. 2014;15:4-15.
64. Hübscher M, Moloney N, Rebbeck T, et al. Contributions of mood, pain catastrophizing, and cold hyperalgesia in acute and chronic low back pain: a comparison with pain-free controls. Clin J Pain. 2014;30:886-893.
65. Giesecke T, Gracely RH, Grant MA, et al. Evidence of augmented central pain processing in idiopathic chronic low back pain. Arthritis Rheum. 2004;50:613-623.
66. Baliki MN, Chialvo DR, Geha PY, et al. Chronic pain and the emotional brain: specific brain activity associated with spontaneous fluctuations of intensity of chronic back pain. J Neurosci. 2006;26:12165-12173.
67. Wertli MM, Eugster R, Held U, et al. Catastrophizing-a prognostic factor for outcome in patients with low back pain: a systematic review. Spine J. 2014;14:2639-2657.
68. Brummett CM, Goesling J, Tsodikov A, et al. Prevalence of the fibromyalgia phenotype in patients with spine pain presenting to a tertiary care pain clinic and the potential treatment implications. Arthritis Rheum. 2013;65:3285-3292.
69. Koes BW, van Tulder M, Lin CW, et al. An updated overview of clinical guidelines for the management of non-specific low back pain in primary care. Eur Spine J. 2010;19:2075-2094.
70. Oliveira VC, Ferreira PH, Maher CG, et al. Effectiveness of self-management of low back pain: systematic review with meta-analysis. Arthritis Care Res. 2012;64:1739-1748.
71. Engers A, Jellema P, Wensing M, et al. Individual patient education for low back pain. Cochrane Database Syst Rev. 2008:CD004057.
72. Hayden JA, van Tulder MW, Malmivaara A, et al. Exercise therapy for treatment of non-specific low back pain. Cochrane Database Syst Rev. 2005:CD000335.
73. French SD, Cameron M, Walker BF, et al. Superficial heat or cold for low back pain. Cochrane Database Syst Rev. 2006:CD004750.
74. Franke H, Franke JD, Fryer G. Osteopathic manipulative treatment for nonspecific low back pain: a systematic review and meta-analysis. BMC Musculoskeletal Disord. 2014;15:286.
75. Franke H, Fryer G, Ostelo RW, et al. Muscle energy technique for non-specific low back pain. Cochrane Database Syst Rev. 2015:CD009852.
76. Oliphant D. Safety of spinal manipulation in the treatment of lumbar disk herniations: a systematic review and risk assessment. J Manipulative Physiol Ther. 2004:197-210.
77. Furlan AD, Giraldo M, Baskwill A, et al. Massage for low-back pain. Cochrane Database Syst Rev. 2015:CD001929.
78. Khadilkar A, Odebiyi DO, Brosseau L, et al. Transcutaneous electrical nerve stimulation (TENS) versus placebo for chronic low back pain. Cochrane Database Syst Rev. 2008:CD003008.
79. Ebadi S, Henschke N, Nakhostin Ansari N, et al. Therapeutic ultrasound for chronic low back pain. Cochrane Database Syst Rev. 2014:CD009169.
80. Furlan AD, van Tulder MW, Cherkin DC, et al. Acupuncture and dry-needling for low back pain. Cochrane Database Syst Rev. 2005:CD001351.
81. Chou R, Huffman LH. Nonpharmacologic therapies for acute and chronic low back pain: a review of the evidence for an American Pain Society/American College of Physicians clinical practice guideline. Ann Intern Med. 2007;147:492-504.
82. Sherman KJ, Cherkin DC, Erro J, et al. Comparing yoga, exercise, and a self-care book for chronic low back pain: a randomized, controlled trial. Ann Intern Med. 2005;143:849-856.
83. Cherkin DC, Sherman KJ, Balderson BH, et al. Effect of mindfulness-based stress reduction vs cognitive behavioral therapy or usual care on back pain and functional limitations in adults with chronic low back pain: a randomized clinical trial. JAMA. 2016;315:1240-1249.
84. Staal JB, de Bie R, de Vet HC, et al. Injection therapy for subacute and chronic low back pain. Cochrane Database Syst Rev. 2008:CD001824.
85. Chou R, Baisden J, Carragee EJ, et al. Surgery for low back pain: a review of the evidence for an American Pain Society Clinical Practice Guideline. Spine. 2009;34:1094-1109.
86. Felson D. Paracetamol is ineffective for spinal pain and knee and hip osteoarthritis. Evid Based Med. 2015;20:205.
87. Machado GC, Maher CG, Ferreira PH, et al. Efficacy and safety of paracetamol for spinal pain and osteoarthritis: systematic review and meta-analysis of randomised placebo controlled trials. BMJ. 2015;350:h1225.
88. Enthoven WT, Roelofs PD, Deyo RA, et al. Non-steroidal anti-inflammatory drugs for chronic low back pain. Cochrane Database Syst Rev. 2016;2:CD012087.
89. White AP, Arnold PM, Norvell DC, et al. Pharmacologic management of chronic low back pain: synthesis of the evidence. Spine (Phila Pa 1976). 2011;36:S131-S143.
Pelvic Inflammatory Disease: How to Recognize and Treat Revised
IN THIS ARTICLE
- Diagnostic tests
- Complications of PID
- CDC treatment regimens
Pelvic inflammatory disease (PID) is an ascending polymicrobial infection of the female upper reproductive tract that primarily affects sexually active women ages 15 to 29. Around 5% of sexually active women in the United States were treated for PID from 2011-2013.1 The rates and severity of PID have declined in North America and Western Europe due to overall decrease in sexually transmitted infection (STI) rates, improved screening initiatives for Chlamydia trachomatis, better treatment compliance secondary to increased access to antibiotics, and diagnostic tests with higher sensitivity.2 Despite this rate reduction, PID remains a major public health concern given the significant long-term complications, which include infertility, ectopic pregnancy, and chronic pelvic pain.3
EPIDEMIOLOGY AND PATHOGENESIS
PID is caused by sexually transmitted bacteria or enteric organisms that have spread to internal reproductive organs. Historically, the two most common pathogens identified in cases of PID have been Chlamydia trachomatis and Neisseria gonorrhoeae; however, the decline in rates of gonorrhea has led to a diminished role for N gonorrhoeae (though it continues to be associated with more severe cases).4,5
More recent studies have suggested a shift in the causative organisms; less than half of women diagnosed with acute PID test positive for either N gonorrhoeae or C trachomatis.6 Emerging infectious agents associated with PID include Mycoplasma genitalium, Gardnerella vaginalis, and bacterial vaginosis–associated bacteria.5,7,8-10
RISK FACTORS
Women ages 15 to 25 are at an increased risk for PID. The high prevalence in this age group may be attributable to high-risk behaviors, including a high number of sexual partners, high frequency of new sexual partners, and engagement in sexual intercourse without condoms.11
Taking an accurate sexual history is imperative. Clinicians should maintain a high level of suspicion for PID in women with a history of the disease, as 25% will experience recurrence.12
Clinicians should not be deterred from screening for STIs and cervical cancer in women who report having sex with other women. In addition, transgender patients should be assessed for STIs and HIV-related risks based on current anatomy sexual practices.13
PHYSICAL EXAM
While some cases of PID are asymptomatic, the typical presentation includes bilateral abdominal pain and/or pelvic pain, with onset during or shortly after menses. The pain often worsens with movement and coitus. Associated signs and symptoms include abnormal uterine bleeding or vaginal discharge; dysuria; fever and chills; frequent urination; lower back pain; and nausea and/or vomiting.14,15
All females suspected of having PID should undergo both a bimanual exam and a speculum exam. On bimanual examination, adnexal tenderness has the highest sensitivity (93% to 95.5%) for ruling out acute PID, whereas on speculum exam, purulent endocervical discharge has the highest specificity (93%).16,17 Bimanual exam findings suggestive of PID include cervical motion tenderness, uterine tenderness, and/or adnexal tenderness. Suggestive speculum exam findings include abnormal discoloration or texture of the cervix and/or endocervical mucopurulent discharge.5,16,17
One cardinal rule that should not be overlooked is that all females of reproductive age who present with abdominal pain and/or pelvic pain should take a pregnancy test to rule out ectopic pregnancy and any other pregnancy-related complications.
DIAGNOSIS
The diagnosis of PID relies on clinical judgement and a high index of suspicion.5,18 The CDC’s diagnostic criteria for acute PID include
- Sexually active female AND
- Pelvic or lower abdominal pain AND
- Cervical motion tenderness OR uterine tenderness OR adnexal tenderness.5
Additional findings that support the diagnosis include
- Abnormal cervical mucopurulent discharge or cervical friability
- Abundant white blood cells (WBCs) on saline microscopy of vaginal fluid
- Elevated C-reactive protein
- Elevated erythrocyte sedimentation rate
- Laboratory documentation of infection with C trachomatis or N gonorrhea
- Oral temperature > 101°F.5,18
The CDC notes that the first two findings (mucopurulent discharge and evidence of WBCs on microscopy) occur in most women with PID; in their absence, the diagnosis is unlikely and other sources of pain should be considered.5 The differential for PID includes acute appendicitis; adhesions; carcinoid tumor; cholecystitis; ectopic pregnancy; endometriosis; inflammatory bowel disease; and ovarian cyst.19
Given the variability in presentation, clinicians may find it useful to perform further diagnostic testing. There are additional laboratory tests that may be ordered for patients with a suspected diagnosis of PID (see Table 1).
TREATMENT
According to the CDC’s 2015 treatment guidelines for PID, a negative endocervical exam and negative microbial screening do not rule out an upper reproductive tract infection. Therefore, all sexually active women who present with lower abdominal pain and/or pelvic pain and have evidence of cervical motion, uterine, or adnexal tenderness on bimanual exam should be treated immediately.5
Treatment guidelines are outlined in Table 2. The polymicrobial nature of PID requires gram-negative antibiotic coverage, such as doxycycline plus a second/third-generation cephalosporin.5 Clinicians should note that cefoxitin, a second-generation cephalosporin, is recommended as firstline therapy for inpatients, as it has better anaerobic coverage than ceftriaxone.19 A targeted change in antibiotic coverage—such as inclusion of a macrolide and/or metronidazole—might be necessary if a causative organism is identified by culture.7
Treatment is indicated for all patients with a presumptive diagnosis of PID regardless of symptoms or exam findings, as PID may be asymptomatic and long-term sequelae (eg, infertility, ectopic pregnancy) are often irreversible. At-risk patients include sexually active adolescents, women with multiple sexual partners, women with a history of STI, those whose sexual partner has an STI, and women living in communities with a high prevalence of disease.20,21
Women being treated for PID should be advised to abstain from sexual intercourse until symptoms have resolved, treatment is completed, and any sexual partners have been treated as well. It is essential to emphasize to patients (and their partners) the importance of compliance to treatment regimens and the risk for PID co-infection and reinfection, as recurrence leads to an increase in long-term complications.5
Treatment of sexual partners. The CDC instructs that a woman’s most recent partner should be treated if she had sexual intercourse within 60 days of onset of symptoms or diagnosis. Furthermore, men who have had sexual contact with a woman who has PID in the 60 days prior to onset of her symptoms should be evaluated, tested, and treated for chlamydia and gonorrhea, regardless of the etiology of PID or the pathogens isolated from the woman.5
Admission criteria. Hospitalization should be based on provider judgment despite patient age. The suggested admission criteria include surgical emergency (eg, appendicitis), tubo-ovarian abscess, pregnancy, severe illness, nausea and vomiting, high fever, inability to follow or tolerate an outpatient oral regimen, and lack of clinical response to oral antimicrobial therapy.5
Follow-up care. Clinical improvement (ie, reduction in abdominal, uterine, adnexal, and cervical motion tenderness) should occur within 72 hours of antimicrobial therapy initiation. If it does not, hospital admission or adjustment in antimicrobial regimen should be considered, as well as additional diagnostic testing (eg, laparoscopy). In addition, all women with chlamydial- or gonococcal-related PID should return in three months for surveillance testing.22
COMPLICATIONS
Long-term complications—including infertility, chronic pelvic pain, and ectopic pregnancy—may occur, even when there has been a clinical response to adequate treatment. Data from the PID Evaluation and Clinical Health (PEACH) study were analyzed to assess long-term sequelae at seven years postdiagnosis and treatment. The researchers found that about 21% of women experienced recurrent PID, 19% developed infertility, and 42% reported chronic pelvic pain.3 Other research has also shown that repeat episodes of PID and delayed treatment increase the risk for long-term complications.23,24
SCREENING AND PREVENTION
Ten percent of women with an untreated STI will go on to develop PID.4 It is imperative to educate patients on the dangers and consequences of STIs when they become sexually active. Adolescents benefit the most from preventive education; this group is twice as likely as any other age group to be diagnosed with PID due to their inclination toward risky sexual behavior. Additionally, younger women tend to have a more friable cervix, increasing their risk for infection.25,26 Providers should promote safe sexual practices, such as condom use and less frequent partner exchange, in order to reduce STI exposure.
In 2015, the rate of reported cases of C trachomatis was 645.5 per 100,000 females, and of N gonorrheae, 107.2 per 100,000 females.23 The United States Preventive Services Task Force and the CDC recommend annual screening for chlamydia and gonorrhea in all sexually active women younger than 25, as well as sexually active women ages 25 and older who are considered at increased risk.5
CONCLUSION
PID is often difficult to diagnose, since patients may be asymptomatic or present with vague symptoms. Clinicians should maintain a high level of suspicion for PID in adolescent females due to the high incidence of STI exposure in this population. The best way to prevent long-term complications of PID is to prevent the first episode of PID and/or first exposure to STIs. Therefore, clinicians should be proactive in offering STI screenings to all sexually active patients younger than 25 who request care, regardless of their chief complaint, and educating patients on the potential long-term effects of PID and STIs.
1. Leichliter JS, Chandra A, Aral SO. Correlates of self-reported pelvic inflammatory disease treatment in sexually experienced reproductive-aged women in the United States, 1995 and 2006-2010. Sex Transm Dis. 2013;40(5):413-418.
2. Owusu-Edusei K Jr, Bohm MK, Chesson HW, Kent CK. Chlamydia screening and pelvic inflammatory disease: insights from exploratory time-series analyses. Am J Prev Med. 2010;38(6):652-657.
3. Trent M, Bass D, Ness RB, Haggerty C. Recurrent PID, subsequent STI, and reproductive health outcomes: findings from the PID evaluation and clinical health (PEACH) study. Sex Transm Dis. 2011;38(9):879-881.
4. Mitchell C, Prabhu M. Pelvic inflammatory disease: current concepts in pathogenesis, diagnosis and treatment. Infect Dis Clin North Am. 2013;27(4):793-809.
5. CDC. Pelvic inflammatory disease (PID). www.cdc.gov/std/tg2015/pid.htm. Accessed July 13, 2017.
6. Burnett AM, Anderson CP, Zwank MD. Laboratory-confirmed gonorrhea and/or chlamydia rates in clinically diagnosed pelvic inflammatory disease and cervicitis. Am J Emerg Med. 2012;30:1114–1117.
7. Bjartling C, Osser S, Persson K. The association between Mycoplasma genitalium and pelvic inflammatory disease after termination of pregnancy. BJOG. 2010;117(3):361-364.
8. Ness RB, Kip KE, Hillier SL, et al. A cluster analysis of bacterial vaginosis-associated microflora and pelvic inflammatory disease. Am J Epidemiol. 2005;162(6):585-590.
9. Ness RB, Hillier SL, Kip KE, et al. Bacterial vaginosis and risk of pelvic inflammatory disease. Obstet Gynecol. 2004;104(4):761-769.
10. Cherpes TL, Wiesenfeld HC, Melan MA, et al. The associations between pelvic inflammatory disease, Trichomonas vaginalis infection, and positive herpes simplex virus type 2 serology. Sex Transm Dis. 2006;33(12):747-752.
11. Simms I, Stephenson JM, Mallinson H, et al. Risk factors associated with pelvic inflammatory disease. Sex Transm Infect. 2006;82(6):452-457.
12. Schindlbeck C, Dziura D, Mylonas I. Diagnosis of pelvic inflammatory disease (PID): intra-operative findings and comparison of vaginal and intra-abdominal cultures. Arch Gynecol Obstet. 2014;289(6):1263-1269.
13. CDC. 2015 sexually transmitted diseases treatment guidelines. www.cdc.gov/std/tg2015/default.htm. Accessed September 6, 2017.
14. Korn AP, Hessol NA, Padian NS, et al. Risk factors for plasma cell endometritis among women with cervical Neisseria gonorrhoeae, cervical Chlamydia trachomatis, or bacterial vaginosis. Am J Obstet Gynecol. 1998;178(5):987-990.
15. Wiesenfeld HC, Sweet RL, Ness RB, et al. Comparison of acute and subclinical pelvic inflammatory disease. Sex Transm Dis. 2005;32(7):400-405.
16. Peipert JF, Ness RB, Blume J, et al. Clinical predictors of endometritis in women with symptoms and signs of pelvic inflammatory disease. Am J Obstet Gynecol. 2001;184(5):856-864.
17. Gaitán H, Angel E, Diaz R, et al. Accuracy of five different diagnostic techniques in mild-to-moderate pelvic inflammatory disease. Infect Dis Obstet Gynecol. 2002;10(4):171-180.
18. Tukeva TA, Aronen HJ, Karjalainen PT, et al. MR imaging in pelvic inflammatory disease: comparison with laparoscopy and US. Radiology. 1999;210(1):209-216.
19. Morino M, Pellegrino L, Castagna E, et al. Acute nonspecific abdominal pain. Ann Surg. 2006;244(6):881-888.
20. Woods JL, Scurlock AM, Hensel DJ. Pelvic inflammatory disease in the adolescent: understanding diagnosis and treatment as a health care provider. Pediatric Emergency Care. 2013;29(6):720-725.
21. LeFevre ML; U.S. Preventive Services Task Force. Screening for chlamydia and gonorrhea: U.S. Preventive Services Task Force recommendation statement. Ann Intern Med. 2014;161(12):902-910.
22. Hosenfeld CB, Workowski KA, Berman S, et al. Repeat infection with Chlamydia and gonorrhea among females: a systematic review of the literature. Sex Transm Dis. 2009;36(8):478-489.
23. CDC. Sexually transmitted disease surveillance. www.cdc.gov/std/stats15/STD-Surveillance-2015-print.pdf. Accessed July 13, 2017.
24. Hillis SD, Joesoef R, Marchbanks PA, et al. Delayed care of pelvic inflammatory disease as a risk factor for impaired fertility. Am J Obstet Gynecol. 1993;168(5):1503-1509.
25. Goyal M, Hersh A, Luan X, et al. National trends in pelvic inflammatory disease among adolescents in the emergency department. J Adolesc Health. 2013;53(2):249-252.
26. Gray-Swain MR, Peipert JF. Pelvic inflammatory disease in adolescents. Curr Opin Obstet Gynecol. 2006;18(5):503-510.
27. Łój B, Brodowska A, Ciecwiez S, et al. The role of serological testing for Chlamydia trachomatis in differential diagnosis of pelvic pain. Ann Agric Environ Med. 2016;23(3):506-510.
28. Sam JW, Jacobs JE, Birnbaum BA. Spectrum of CT findings in acute pyogenic pelvic inflammatory disease. Radiographics. 2002;22(6):1327-1 334.
IN THIS ARTICLE
- Diagnostic tests
- Complications of PID
- CDC treatment regimens
Pelvic inflammatory disease (PID) is an ascending polymicrobial infection of the female upper reproductive tract that primarily affects sexually active women ages 15 to 29. Around 5% of sexually active women in the United States were treated for PID from 2011-2013.1 The rates and severity of PID have declined in North America and Western Europe due to overall decrease in sexually transmitted infection (STI) rates, improved screening initiatives for Chlamydia trachomatis, better treatment compliance secondary to increased access to antibiotics, and diagnostic tests with higher sensitivity.2 Despite this rate reduction, PID remains a major public health concern given the significant long-term complications, which include infertility, ectopic pregnancy, and chronic pelvic pain.3
EPIDEMIOLOGY AND PATHOGENESIS
PID is caused by sexually transmitted bacteria or enteric organisms that have spread to internal reproductive organs. Historically, the two most common pathogens identified in cases of PID have been Chlamydia trachomatis and Neisseria gonorrhoeae; however, the decline in rates of gonorrhea has led to a diminished role for N gonorrhoeae (though it continues to be associated with more severe cases).4,5
More recent studies have suggested a shift in the causative organisms; less than half of women diagnosed with acute PID test positive for either N gonorrhoeae or C trachomatis.6 Emerging infectious agents associated with PID include Mycoplasma genitalium, Gardnerella vaginalis, and bacterial vaginosis–associated bacteria.5,7,8-10
RISK FACTORS
Women ages 15 to 25 are at an increased risk for PID. The high prevalence in this age group may be attributable to high-risk behaviors, including a high number of sexual partners, high frequency of new sexual partners, and engagement in sexual intercourse without condoms.11
Taking an accurate sexual history is imperative. Clinicians should maintain a high level of suspicion for PID in women with a history of the disease, as 25% will experience recurrence.12
Clinicians should not be deterred from screening for STIs and cervical cancer in women who report having sex with other women. In addition, transgender patients should be assessed for STIs and HIV-related risks based on current anatomy sexual practices.13
PHYSICAL EXAM
While some cases of PID are asymptomatic, the typical presentation includes bilateral abdominal pain and/or pelvic pain, with onset during or shortly after menses. The pain often worsens with movement and coitus. Associated signs and symptoms include abnormal uterine bleeding or vaginal discharge; dysuria; fever and chills; frequent urination; lower back pain; and nausea and/or vomiting.14,15
All females suspected of having PID should undergo both a bimanual exam and a speculum exam. On bimanual examination, adnexal tenderness has the highest sensitivity (93% to 95.5%) for ruling out acute PID, whereas on speculum exam, purulent endocervical discharge has the highest specificity (93%).16,17 Bimanual exam findings suggestive of PID include cervical motion tenderness, uterine tenderness, and/or adnexal tenderness. Suggestive speculum exam findings include abnormal discoloration or texture of the cervix and/or endocervical mucopurulent discharge.5,16,17
One cardinal rule that should not be overlooked is that all females of reproductive age who present with abdominal pain and/or pelvic pain should take a pregnancy test to rule out ectopic pregnancy and any other pregnancy-related complications.
DIAGNOSIS
The diagnosis of PID relies on clinical judgement and a high index of suspicion.5,18 The CDC’s diagnostic criteria for acute PID include
- Sexually active female AND
- Pelvic or lower abdominal pain AND
- Cervical motion tenderness OR uterine tenderness OR adnexal tenderness.5
Additional findings that support the diagnosis include
- Abnormal cervical mucopurulent discharge or cervical friability
- Abundant white blood cells (WBCs) on saline microscopy of vaginal fluid
- Elevated C-reactive protein
- Elevated erythrocyte sedimentation rate
- Laboratory documentation of infection with C trachomatis or N gonorrhea
- Oral temperature > 101°F.5,18
The CDC notes that the first two findings (mucopurulent discharge and evidence of WBCs on microscopy) occur in most women with PID; in their absence, the diagnosis is unlikely and other sources of pain should be considered.5 The differential for PID includes acute appendicitis; adhesions; carcinoid tumor; cholecystitis; ectopic pregnancy; endometriosis; inflammatory bowel disease; and ovarian cyst.19
Given the variability in presentation, clinicians may find it useful to perform further diagnostic testing. There are additional laboratory tests that may be ordered for patients with a suspected diagnosis of PID (see Table 1).
TREATMENT
According to the CDC’s 2015 treatment guidelines for PID, a negative endocervical exam and negative microbial screening do not rule out an upper reproductive tract infection. Therefore, all sexually active women who present with lower abdominal pain and/or pelvic pain and have evidence of cervical motion, uterine, or adnexal tenderness on bimanual exam should be treated immediately.5
Treatment guidelines are outlined in Table 2. The polymicrobial nature of PID requires gram-negative antibiotic coverage, such as doxycycline plus a second/third-generation cephalosporin.5 Clinicians should note that cefoxitin, a second-generation cephalosporin, is recommended as firstline therapy for inpatients, as it has better anaerobic coverage than ceftriaxone.19 A targeted change in antibiotic coverage—such as inclusion of a macrolide and/or metronidazole—might be necessary if a causative organism is identified by culture.7
Treatment is indicated for all patients with a presumptive diagnosis of PID regardless of symptoms or exam findings, as PID may be asymptomatic and long-term sequelae (eg, infertility, ectopic pregnancy) are often irreversible. At-risk patients include sexually active adolescents, women with multiple sexual partners, women with a history of STI, those whose sexual partner has an STI, and women living in communities with a high prevalence of disease.20,21
Women being treated for PID should be advised to abstain from sexual intercourse until symptoms have resolved, treatment is completed, and any sexual partners have been treated as well. It is essential to emphasize to patients (and their partners) the importance of compliance to treatment regimens and the risk for PID co-infection and reinfection, as recurrence leads to an increase in long-term complications.5
Treatment of sexual partners. The CDC instructs that a woman’s most recent partner should be treated if she had sexual intercourse within 60 days of onset of symptoms or diagnosis. Furthermore, men who have had sexual contact with a woman who has PID in the 60 days prior to onset of her symptoms should be evaluated, tested, and treated for chlamydia and gonorrhea, regardless of the etiology of PID or the pathogens isolated from the woman.5
Admission criteria. Hospitalization should be based on provider judgment despite patient age. The suggested admission criteria include surgical emergency (eg, appendicitis), tubo-ovarian abscess, pregnancy, severe illness, nausea and vomiting, high fever, inability to follow or tolerate an outpatient oral regimen, and lack of clinical response to oral antimicrobial therapy.5
Follow-up care. Clinical improvement (ie, reduction in abdominal, uterine, adnexal, and cervical motion tenderness) should occur within 72 hours of antimicrobial therapy initiation. If it does not, hospital admission or adjustment in antimicrobial regimen should be considered, as well as additional diagnostic testing (eg, laparoscopy). In addition, all women with chlamydial- or gonococcal-related PID should return in three months for surveillance testing.22
COMPLICATIONS
Long-term complications—including infertility, chronic pelvic pain, and ectopic pregnancy—may occur, even when there has been a clinical response to adequate treatment. Data from the PID Evaluation and Clinical Health (PEACH) study were analyzed to assess long-term sequelae at seven years postdiagnosis and treatment. The researchers found that about 21% of women experienced recurrent PID, 19% developed infertility, and 42% reported chronic pelvic pain.3 Other research has also shown that repeat episodes of PID and delayed treatment increase the risk for long-term complications.23,24
SCREENING AND PREVENTION
Ten percent of women with an untreated STI will go on to develop PID.4 It is imperative to educate patients on the dangers and consequences of STIs when they become sexually active. Adolescents benefit the most from preventive education; this group is twice as likely as any other age group to be diagnosed with PID due to their inclination toward risky sexual behavior. Additionally, younger women tend to have a more friable cervix, increasing their risk for infection.25,26 Providers should promote safe sexual practices, such as condom use and less frequent partner exchange, in order to reduce STI exposure.
In 2015, the rate of reported cases of C trachomatis was 645.5 per 100,000 females, and of N gonorrheae, 107.2 per 100,000 females.23 The United States Preventive Services Task Force and the CDC recommend annual screening for chlamydia and gonorrhea in all sexually active women younger than 25, as well as sexually active women ages 25 and older who are considered at increased risk.5
CONCLUSION
PID is often difficult to diagnose, since patients may be asymptomatic or present with vague symptoms. Clinicians should maintain a high level of suspicion for PID in adolescent females due to the high incidence of STI exposure in this population. The best way to prevent long-term complications of PID is to prevent the first episode of PID and/or first exposure to STIs. Therefore, clinicians should be proactive in offering STI screenings to all sexually active patients younger than 25 who request care, regardless of their chief complaint, and educating patients on the potential long-term effects of PID and STIs.
IN THIS ARTICLE
- Diagnostic tests
- Complications of PID
- CDC treatment regimens
Pelvic inflammatory disease (PID) is an ascending polymicrobial infection of the female upper reproductive tract that primarily affects sexually active women ages 15 to 29. Around 5% of sexually active women in the United States were treated for PID from 2011-2013.1 The rates and severity of PID have declined in North America and Western Europe due to overall decrease in sexually transmitted infection (STI) rates, improved screening initiatives for Chlamydia trachomatis, better treatment compliance secondary to increased access to antibiotics, and diagnostic tests with higher sensitivity.2 Despite this rate reduction, PID remains a major public health concern given the significant long-term complications, which include infertility, ectopic pregnancy, and chronic pelvic pain.3
EPIDEMIOLOGY AND PATHOGENESIS
PID is caused by sexually transmitted bacteria or enteric organisms that have spread to internal reproductive organs. Historically, the two most common pathogens identified in cases of PID have been Chlamydia trachomatis and Neisseria gonorrhoeae; however, the decline in rates of gonorrhea has led to a diminished role for N gonorrhoeae (though it continues to be associated with more severe cases).4,5
More recent studies have suggested a shift in the causative organisms; less than half of women diagnosed with acute PID test positive for either N gonorrhoeae or C trachomatis.6 Emerging infectious agents associated with PID include Mycoplasma genitalium, Gardnerella vaginalis, and bacterial vaginosis–associated bacteria.5,7,8-10
RISK FACTORS
Women ages 15 to 25 are at an increased risk for PID. The high prevalence in this age group may be attributable to high-risk behaviors, including a high number of sexual partners, high frequency of new sexual partners, and engagement in sexual intercourse without condoms.11
Taking an accurate sexual history is imperative. Clinicians should maintain a high level of suspicion for PID in women with a history of the disease, as 25% will experience recurrence.12
Clinicians should not be deterred from screening for STIs and cervical cancer in women who report having sex with other women. In addition, transgender patients should be assessed for STIs and HIV-related risks based on current anatomy sexual practices.13
PHYSICAL EXAM
While some cases of PID are asymptomatic, the typical presentation includes bilateral abdominal pain and/or pelvic pain, with onset during or shortly after menses. The pain often worsens with movement and coitus. Associated signs and symptoms include abnormal uterine bleeding or vaginal discharge; dysuria; fever and chills; frequent urination; lower back pain; and nausea and/or vomiting.14,15
All females suspected of having PID should undergo both a bimanual exam and a speculum exam. On bimanual examination, adnexal tenderness has the highest sensitivity (93% to 95.5%) for ruling out acute PID, whereas on speculum exam, purulent endocervical discharge has the highest specificity (93%).16,17 Bimanual exam findings suggestive of PID include cervical motion tenderness, uterine tenderness, and/or adnexal tenderness. Suggestive speculum exam findings include abnormal discoloration or texture of the cervix and/or endocervical mucopurulent discharge.5,16,17
One cardinal rule that should not be overlooked is that all females of reproductive age who present with abdominal pain and/or pelvic pain should take a pregnancy test to rule out ectopic pregnancy and any other pregnancy-related complications.
DIAGNOSIS
The diagnosis of PID relies on clinical judgement and a high index of suspicion.5,18 The CDC’s diagnostic criteria for acute PID include
- Sexually active female AND
- Pelvic or lower abdominal pain AND
- Cervical motion tenderness OR uterine tenderness OR adnexal tenderness.5
Additional findings that support the diagnosis include
- Abnormal cervical mucopurulent discharge or cervical friability
- Abundant white blood cells (WBCs) on saline microscopy of vaginal fluid
- Elevated C-reactive protein
- Elevated erythrocyte sedimentation rate
- Laboratory documentation of infection with C trachomatis or N gonorrhea
- Oral temperature > 101°F.5,18
The CDC notes that the first two findings (mucopurulent discharge and evidence of WBCs on microscopy) occur in most women with PID; in their absence, the diagnosis is unlikely and other sources of pain should be considered.5 The differential for PID includes acute appendicitis; adhesions; carcinoid tumor; cholecystitis; ectopic pregnancy; endometriosis; inflammatory bowel disease; and ovarian cyst.19
Given the variability in presentation, clinicians may find it useful to perform further diagnostic testing. There are additional laboratory tests that may be ordered for patients with a suspected diagnosis of PID (see Table 1).
TREATMENT
According to the CDC’s 2015 treatment guidelines for PID, a negative endocervical exam and negative microbial screening do not rule out an upper reproductive tract infection. Therefore, all sexually active women who present with lower abdominal pain and/or pelvic pain and have evidence of cervical motion, uterine, or adnexal tenderness on bimanual exam should be treated immediately.5
Treatment guidelines are outlined in Table 2. The polymicrobial nature of PID requires gram-negative antibiotic coverage, such as doxycycline plus a second/third-generation cephalosporin.5 Clinicians should note that cefoxitin, a second-generation cephalosporin, is recommended as firstline therapy for inpatients, as it has better anaerobic coverage than ceftriaxone.19 A targeted change in antibiotic coverage—such as inclusion of a macrolide and/or metronidazole—might be necessary if a causative organism is identified by culture.7
Treatment is indicated for all patients with a presumptive diagnosis of PID regardless of symptoms or exam findings, as PID may be asymptomatic and long-term sequelae (eg, infertility, ectopic pregnancy) are often irreversible. At-risk patients include sexually active adolescents, women with multiple sexual partners, women with a history of STI, those whose sexual partner has an STI, and women living in communities with a high prevalence of disease.20,21
Women being treated for PID should be advised to abstain from sexual intercourse until symptoms have resolved, treatment is completed, and any sexual partners have been treated as well. It is essential to emphasize to patients (and their partners) the importance of compliance to treatment regimens and the risk for PID co-infection and reinfection, as recurrence leads to an increase in long-term complications.5
Treatment of sexual partners. The CDC instructs that a woman’s most recent partner should be treated if she had sexual intercourse within 60 days of onset of symptoms or diagnosis. Furthermore, men who have had sexual contact with a woman who has PID in the 60 days prior to onset of her symptoms should be evaluated, tested, and treated for chlamydia and gonorrhea, regardless of the etiology of PID or the pathogens isolated from the woman.5
Admission criteria. Hospitalization should be based on provider judgment despite patient age. The suggested admission criteria include surgical emergency (eg, appendicitis), tubo-ovarian abscess, pregnancy, severe illness, nausea and vomiting, high fever, inability to follow or tolerate an outpatient oral regimen, and lack of clinical response to oral antimicrobial therapy.5
Follow-up care. Clinical improvement (ie, reduction in abdominal, uterine, adnexal, and cervical motion tenderness) should occur within 72 hours of antimicrobial therapy initiation. If it does not, hospital admission or adjustment in antimicrobial regimen should be considered, as well as additional diagnostic testing (eg, laparoscopy). In addition, all women with chlamydial- or gonococcal-related PID should return in three months for surveillance testing.22
COMPLICATIONS
Long-term complications—including infertility, chronic pelvic pain, and ectopic pregnancy—may occur, even when there has been a clinical response to adequate treatment. Data from the PID Evaluation and Clinical Health (PEACH) study were analyzed to assess long-term sequelae at seven years postdiagnosis and treatment. The researchers found that about 21% of women experienced recurrent PID, 19% developed infertility, and 42% reported chronic pelvic pain.3 Other research has also shown that repeat episodes of PID and delayed treatment increase the risk for long-term complications.23,24
SCREENING AND PREVENTION
Ten percent of women with an untreated STI will go on to develop PID.4 It is imperative to educate patients on the dangers and consequences of STIs when they become sexually active. Adolescents benefit the most from preventive education; this group is twice as likely as any other age group to be diagnosed with PID due to their inclination toward risky sexual behavior. Additionally, younger women tend to have a more friable cervix, increasing their risk for infection.25,26 Providers should promote safe sexual practices, such as condom use and less frequent partner exchange, in order to reduce STI exposure.
In 2015, the rate of reported cases of C trachomatis was 645.5 per 100,000 females, and of N gonorrheae, 107.2 per 100,000 females.23 The United States Preventive Services Task Force and the CDC recommend annual screening for chlamydia and gonorrhea in all sexually active women younger than 25, as well as sexually active women ages 25 and older who are considered at increased risk.5
CONCLUSION
PID is often difficult to diagnose, since patients may be asymptomatic or present with vague symptoms. Clinicians should maintain a high level of suspicion for PID in adolescent females due to the high incidence of STI exposure in this population. The best way to prevent long-term complications of PID is to prevent the first episode of PID and/or first exposure to STIs. Therefore, clinicians should be proactive in offering STI screenings to all sexually active patients younger than 25 who request care, regardless of their chief complaint, and educating patients on the potential long-term effects of PID and STIs.
1. Leichliter JS, Chandra A, Aral SO. Correlates of self-reported pelvic inflammatory disease treatment in sexually experienced reproductive-aged women in the United States, 1995 and 2006-2010. Sex Transm Dis. 2013;40(5):413-418.
2. Owusu-Edusei K Jr, Bohm MK, Chesson HW, Kent CK. Chlamydia screening and pelvic inflammatory disease: insights from exploratory time-series analyses. Am J Prev Med. 2010;38(6):652-657.
3. Trent M, Bass D, Ness RB, Haggerty C. Recurrent PID, subsequent STI, and reproductive health outcomes: findings from the PID evaluation and clinical health (PEACH) study. Sex Transm Dis. 2011;38(9):879-881.
4. Mitchell C, Prabhu M. Pelvic inflammatory disease: current concepts in pathogenesis, diagnosis and treatment. Infect Dis Clin North Am. 2013;27(4):793-809.
5. CDC. Pelvic inflammatory disease (PID). www.cdc.gov/std/tg2015/pid.htm. Accessed July 13, 2017.
6. Burnett AM, Anderson CP, Zwank MD. Laboratory-confirmed gonorrhea and/or chlamydia rates in clinically diagnosed pelvic inflammatory disease and cervicitis. Am J Emerg Med. 2012;30:1114–1117.
7. Bjartling C, Osser S, Persson K. The association between Mycoplasma genitalium and pelvic inflammatory disease after termination of pregnancy. BJOG. 2010;117(3):361-364.
8. Ness RB, Kip KE, Hillier SL, et al. A cluster analysis of bacterial vaginosis-associated microflora and pelvic inflammatory disease. Am J Epidemiol. 2005;162(6):585-590.
9. Ness RB, Hillier SL, Kip KE, et al. Bacterial vaginosis and risk of pelvic inflammatory disease. Obstet Gynecol. 2004;104(4):761-769.
10. Cherpes TL, Wiesenfeld HC, Melan MA, et al. The associations between pelvic inflammatory disease, Trichomonas vaginalis infection, and positive herpes simplex virus type 2 serology. Sex Transm Dis. 2006;33(12):747-752.
11. Simms I, Stephenson JM, Mallinson H, et al. Risk factors associated with pelvic inflammatory disease. Sex Transm Infect. 2006;82(6):452-457.
12. Schindlbeck C, Dziura D, Mylonas I. Diagnosis of pelvic inflammatory disease (PID): intra-operative findings and comparison of vaginal and intra-abdominal cultures. Arch Gynecol Obstet. 2014;289(6):1263-1269.
13. CDC. 2015 sexually transmitted diseases treatment guidelines. www.cdc.gov/std/tg2015/default.htm. Accessed September 6, 2017.
14. Korn AP, Hessol NA, Padian NS, et al. Risk factors for plasma cell endometritis among women with cervical Neisseria gonorrhoeae, cervical Chlamydia trachomatis, or bacterial vaginosis. Am J Obstet Gynecol. 1998;178(5):987-990.
15. Wiesenfeld HC, Sweet RL, Ness RB, et al. Comparison of acute and subclinical pelvic inflammatory disease. Sex Transm Dis. 2005;32(7):400-405.
16. Peipert JF, Ness RB, Blume J, et al. Clinical predictors of endometritis in women with symptoms and signs of pelvic inflammatory disease. Am J Obstet Gynecol. 2001;184(5):856-864.
17. Gaitán H, Angel E, Diaz R, et al. Accuracy of five different diagnostic techniques in mild-to-moderate pelvic inflammatory disease. Infect Dis Obstet Gynecol. 2002;10(4):171-180.
18. Tukeva TA, Aronen HJ, Karjalainen PT, et al. MR imaging in pelvic inflammatory disease: comparison with laparoscopy and US. Radiology. 1999;210(1):209-216.
19. Morino M, Pellegrino L, Castagna E, et al. Acute nonspecific abdominal pain. Ann Surg. 2006;244(6):881-888.
20. Woods JL, Scurlock AM, Hensel DJ. Pelvic inflammatory disease in the adolescent: understanding diagnosis and treatment as a health care provider. Pediatric Emergency Care. 2013;29(6):720-725.
21. LeFevre ML; U.S. Preventive Services Task Force. Screening for chlamydia and gonorrhea: U.S. Preventive Services Task Force recommendation statement. Ann Intern Med. 2014;161(12):902-910.
22. Hosenfeld CB, Workowski KA, Berman S, et al. Repeat infection with Chlamydia and gonorrhea among females: a systematic review of the literature. Sex Transm Dis. 2009;36(8):478-489.
23. CDC. Sexually transmitted disease surveillance. www.cdc.gov/std/stats15/STD-Surveillance-2015-print.pdf. Accessed July 13, 2017.
24. Hillis SD, Joesoef R, Marchbanks PA, et al. Delayed care of pelvic inflammatory disease as a risk factor for impaired fertility. Am J Obstet Gynecol. 1993;168(5):1503-1509.
25. Goyal M, Hersh A, Luan X, et al. National trends in pelvic inflammatory disease among adolescents in the emergency department. J Adolesc Health. 2013;53(2):249-252.
26. Gray-Swain MR, Peipert JF. Pelvic inflammatory disease in adolescents. Curr Opin Obstet Gynecol. 2006;18(5):503-510.
27. Łój B, Brodowska A, Ciecwiez S, et al. The role of serological testing for Chlamydia trachomatis in differential diagnosis of pelvic pain. Ann Agric Environ Med. 2016;23(3):506-510.
28. Sam JW, Jacobs JE, Birnbaum BA. Spectrum of CT findings in acute pyogenic pelvic inflammatory disease. Radiographics. 2002;22(6):1327-1 334.
1. Leichliter JS, Chandra A, Aral SO. Correlates of self-reported pelvic inflammatory disease treatment in sexually experienced reproductive-aged women in the United States, 1995 and 2006-2010. Sex Transm Dis. 2013;40(5):413-418.
2. Owusu-Edusei K Jr, Bohm MK, Chesson HW, Kent CK. Chlamydia screening and pelvic inflammatory disease: insights from exploratory time-series analyses. Am J Prev Med. 2010;38(6):652-657.
3. Trent M, Bass D, Ness RB, Haggerty C. Recurrent PID, subsequent STI, and reproductive health outcomes: findings from the PID evaluation and clinical health (PEACH) study. Sex Transm Dis. 2011;38(9):879-881.
4. Mitchell C, Prabhu M. Pelvic inflammatory disease: current concepts in pathogenesis, diagnosis and treatment. Infect Dis Clin North Am. 2013;27(4):793-809.
5. CDC. Pelvic inflammatory disease (PID). www.cdc.gov/std/tg2015/pid.htm. Accessed July 13, 2017.
6. Burnett AM, Anderson CP, Zwank MD. Laboratory-confirmed gonorrhea and/or chlamydia rates in clinically diagnosed pelvic inflammatory disease and cervicitis. Am J Emerg Med. 2012;30:1114–1117.
7. Bjartling C, Osser S, Persson K. The association between Mycoplasma genitalium and pelvic inflammatory disease after termination of pregnancy. BJOG. 2010;117(3):361-364.
8. Ness RB, Kip KE, Hillier SL, et al. A cluster analysis of bacterial vaginosis-associated microflora and pelvic inflammatory disease. Am J Epidemiol. 2005;162(6):585-590.
9. Ness RB, Hillier SL, Kip KE, et al. Bacterial vaginosis and risk of pelvic inflammatory disease. Obstet Gynecol. 2004;104(4):761-769.
10. Cherpes TL, Wiesenfeld HC, Melan MA, et al. The associations between pelvic inflammatory disease, Trichomonas vaginalis infection, and positive herpes simplex virus type 2 serology. Sex Transm Dis. 2006;33(12):747-752.
11. Simms I, Stephenson JM, Mallinson H, et al. Risk factors associated with pelvic inflammatory disease. Sex Transm Infect. 2006;82(6):452-457.
12. Schindlbeck C, Dziura D, Mylonas I. Diagnosis of pelvic inflammatory disease (PID): intra-operative findings and comparison of vaginal and intra-abdominal cultures. Arch Gynecol Obstet. 2014;289(6):1263-1269.
13. CDC. 2015 sexually transmitted diseases treatment guidelines. www.cdc.gov/std/tg2015/default.htm. Accessed September 6, 2017.
14. Korn AP, Hessol NA, Padian NS, et al. Risk factors for plasma cell endometritis among women with cervical Neisseria gonorrhoeae, cervical Chlamydia trachomatis, or bacterial vaginosis. Am J Obstet Gynecol. 1998;178(5):987-990.
15. Wiesenfeld HC, Sweet RL, Ness RB, et al. Comparison of acute and subclinical pelvic inflammatory disease. Sex Transm Dis. 2005;32(7):400-405.
16. Peipert JF, Ness RB, Blume J, et al. Clinical predictors of endometritis in women with symptoms and signs of pelvic inflammatory disease. Am J Obstet Gynecol. 2001;184(5):856-864.
17. Gaitán H, Angel E, Diaz R, et al. Accuracy of five different diagnostic techniques in mild-to-moderate pelvic inflammatory disease. Infect Dis Obstet Gynecol. 2002;10(4):171-180.
18. Tukeva TA, Aronen HJ, Karjalainen PT, et al. MR imaging in pelvic inflammatory disease: comparison with laparoscopy and US. Radiology. 1999;210(1):209-216.
19. Morino M, Pellegrino L, Castagna E, et al. Acute nonspecific abdominal pain. Ann Surg. 2006;244(6):881-888.
20. Woods JL, Scurlock AM, Hensel DJ. Pelvic inflammatory disease in the adolescent: understanding diagnosis and treatment as a health care provider. Pediatric Emergency Care. 2013;29(6):720-725.
21. LeFevre ML; U.S. Preventive Services Task Force. Screening for chlamydia and gonorrhea: U.S. Preventive Services Task Force recommendation statement. Ann Intern Med. 2014;161(12):902-910.
22. Hosenfeld CB, Workowski KA, Berman S, et al. Repeat infection with Chlamydia and gonorrhea among females: a systematic review of the literature. Sex Transm Dis. 2009;36(8):478-489.
23. CDC. Sexually transmitted disease surveillance. www.cdc.gov/std/stats15/STD-Surveillance-2015-print.pdf. Accessed July 13, 2017.
24. Hillis SD, Joesoef R, Marchbanks PA, et al. Delayed care of pelvic inflammatory disease as a risk factor for impaired fertility. Am J Obstet Gynecol. 1993;168(5):1503-1509.
25. Goyal M, Hersh A, Luan X, et al. National trends in pelvic inflammatory disease among adolescents in the emergency department. J Adolesc Health. 2013;53(2):249-252.
26. Gray-Swain MR, Peipert JF. Pelvic inflammatory disease in adolescents. Curr Opin Obstet Gynecol. 2006;18(5):503-510.
27. Łój B, Brodowska A, Ciecwiez S, et al. The role of serological testing for Chlamydia trachomatis in differential diagnosis of pelvic pain. Ann Agric Environ Med. 2016;23(3):506-510.
28. Sam JW, Jacobs JE, Birnbaum BA. Spectrum of CT findings in acute pyogenic pelvic inflammatory disease. Radiographics. 2002;22(6):1327-1 334.
First EDition: New Assay Helps Distinguish Viral and Bacterial Infections in Children, more
New Assay Helps Distinguish Viral and Bacterial Infections in Children
BY IAN LACY
An assay testing the presence of three blood-borne host-proteins shows promise in accurately identifying viral and bacterial infections in febrile children, a validation study found.
The three proteins that the ImmunoXpert assay uses to differentiate between viral and bacterial infections are: viral-induced tumor necrosis factor-related apoptosis-inducing ligand (TRAIL), interferon gamma-induced protein-10 (IP-10), and bacterial-induced C-reactive protein (CRP). While TRAIL and IP-10 are novel identifiers, CRP has been used in traditional bacterial detecting assays, Isaac Srugo, MD, and colleagues reported.
The investigators identified 597 potential stored patient serum samples from patients admitted to multiple pediatric EDs and wards in Israel and Switzerland, and ultimately, 361 samples were selected for assay testing.1
Of the 361 patients whose samples were selected for testing, the assay identified 209 patients (58%) with a viral infection, 99 patients (27%) with a bacterial infection, and the remaining 53 patients (15%) with an equivocal outcome, according to Dr Srugo of the Ruth and Bruce Rappaport Faculty of Medicine, Technion-Israel Institute of Technology, Haifa, Israel, and his colleagues. The 307 patients with a bacterial or viral diagnosis had sensitivity of 93.8% (95% confidence interval [CI], 87.8%-99.8%) and specificity of 89.8% (CI, 85.6%-94.0%). There were four false-negative and 21 false-positive findings.
The study found that TRAIL and IP-10 were present in higher levels in children with viral infections compared to children with bacterial infections. The opposite was true of CRP results, with significantly lower levels of CRP in children with viral infections compared to those in children with bacterial infections.
“Notably, among the indeterminate diagnosis patients without a reference standard, the assay gave a bacterial or viral outcome for 69% of the cases (the rest were equivocal), with half of these yielding a score associated with a particularly high degree of assay diagnostic confidence,” investigators said. “This finding suggests that the assay may be applicable to ‘harder-to-diagnose’ cases in real-life clinical settings.”
Also, the assay “exhibits consistent performance across a wide range of ages [3 months to 18 years], time from symptom onset, and clinical syndromes,” Dr Srugo and his associates said.
1. Srugo I, Klein A, Stein M, et al. Validation of a novel assay to distinguish bacterial and viral infections. Pediatrics. 2017 Sep 13. doi:10.1542/peds.2016-3453 [Epub ahead of print]
Emergency Medicine Associate Editor-in-Chief, Francis L. Counselman, MD, to Receive ACEP Award for Outstanding Contribution in Education
Francis L. Counselman, MD, Associate Editor-in-Chief of Emergency Medicine will receive the 2017 American College of Emergency Physicians (ACEP) Award for Outstanding Contribution in Education, at the ACEP Scientific Assembly this month. This award is “presented to an ACEP member who has made a significant contribution to the educational aspects of the specialty,” and is the highest award ACEP gives annually regarding education and teaching.
Dr. Counselman is the founding Chairman of the Department of Emergency Medicine at Eastern Virginia Medical School (EVMS) in Norfolk, Virginia. Founded in 1992, he continues to serve as Chairman. He served as Program Director of the Emergency Medicine residency program at EVMS from 1990 to 2010. In 1999, he was made the EVMS Distinguished Professor of Emergency Medicine. He is the recipient of numerous teaching awards, including the Accreditation Council for Graduate Medical Education (ACGME) Parker Palmer “Courage to Teach” Award; the Emergency Medicine Residents’ Association (EMRA) Residency Director of the Year Award; and the EVMS Award for Outstanding Faculty Achievement. He has served as President of the Association of Academic Chairs of Emergency Medicine (AACEM) and the American Board of Emergency Medicine (ABEM). He is also a frequently invited speaker at state and national meetings. Dr Counselman has served on the Editorial Board of Emergency Medicine since 1999; and has been the Associate Editor-in-Chief since 2006.
Soccer-Playing Girls Five Times More Likely to Return to Same-Day Play After Concussion
BY KARI OAKES
Soccer-playing girls were five times more likely to return to play on the day of a concussion as were their male peers, according to a study presented at the annual meeting of the American Academy of Pediatrics.
Records from 87 soccer players aged 7 to 18 years (median age, 14 years) were examined in a retrospective review of patients seen over a 2-year period by a single physician at a pediatric sports medicine center. Of these, two thirds (n = 58) were girls.
Thirty of the 58 girls (51.7%) reported that they returned to play on the day of their concussion, compared to five of 29 boys (17.2%; P = .002). This difference in reporting yielded an odds ratio of 5.14 (95% CI, 1.72-15.3) for girls returning to same-day play, compared with boys who sustained a concussion.
The soccer players included children participating in recreational, club, and school-sponsored soccer, said senior author Shane M. Miller, MD, in an interview. All patients were assessed according to a standardized concussion protocol that involved a neurological examination and validated concussion evaluation tests, including the Immediate Post-Concussion Assessment and Cognitive Test and the Sports Concussion Assessment Tool.
As soccer has grown in popularity as a youth sport, so has the number of reported concussions. “The incidence of reported concussions has increased 1,600% from 1990 to 2014,” wrote Dr Miller and his coauthors in the abstract accompanying the presentation. Dr Miller said that girls are 1.5 times more likely than boys to sustain a concussion while playing soccer.
While seeing the patients who were the subject of the study, Dr Miller realized that most of the soccer players had not come out of play for evaluation after the head impact. Rather, they had continued to play, only later reporting concussion symptoms to coaches, trainers, or parents.
“The athletes may have chosen not to say anything because they didn’t want to come out of the game,” said Dr Miller, a sports medicine physician at Texas Scottish Rite Hospital for Children, Dallas.
“I was surprised by the significant degree of difference” between male and female soccer players, said Dr Miller. The study was not designed to get at the reason for the discrepancy, so Dr Miller could not say with certainty whether awareness of concussion symptoms is significantly lower for female athletes, or whether the athletic culture more strongly encourages minimization of symptoms for girls than boys. In any case, he said, there is room for education of players, coaches, and families to raise awareness of the importance of recognizing and reporting concussion, and then removing the affected athlete from play,
Dr Miller said that future research directions include collaboration with other facilities to conduct prospective research using a concussion registry. This will allow more robust statistical analysis and help ascertain the degree of regional variation in pediatric sports concussion management.
“Current education efforts may not be enough to help athletes, parents, and coaches identify concussion symptoms, know the guidelines for immediate removal from play, and understand the risks of returning to play after an injury. More research is needed on how to better spread this message intended to protect the health of young athletes….” Aaron Zynda, the study’s first author and clinical research coordinator at Texas Scottish Rite, said in a press release accompanying the abstract. “Concussion recognition and identification is a team effort,” he said.
Opioid Management Protocol Reduced Trauma Patient Pain Medication Use
BY ELI ZIMMERMAN
A pain management protocol implemented in a trauma service reduced opioid intake in trauma patients while improving patient satisfaction, according to a retrospective study.
The opioid epidemic continues to grow every day, partly as a result of irresponsible overprescribing of opioid medication, according to Jessica Gross, MB BAO BCh, a trauma surgeon from Wake Forest Baptist Health, North Carolina, at the American Association for the Surgery of Trauma annual meeting. Dr Gross and her colleagues developed a pain management protocol (PMP) to provide adequate pain control while using fewer opioids in the postdischarge setting. They tested their PMP through a retrospective chart review of 498 patients admitted to the trauma service between January 2015 and December 2016, half of whom were admitted before the PMP was initiated and half of whom were admitted afterward.
The PMP involved a stepped approach to treating pain, with acetaminophen or ibuprofen as needed for mild pain, one 5-mg tablet of oxycodone/acetaminophen every 6 hours for moderate pain, two tablets for severe pain, and 50 to 100 mg of tramadol every 6 hours for breakthrough pain.
Counseling services for patients who were found to be in danger of substance abuse were provided in the hospital, and at discharge, patients received a weaning plan for their medication, according to Dr Gross.
If the short-acting medications were found to be inadequate to control pain, patients were given slow-release pain medication as needed.
The average total medication, which included medication given at discharge and for refills, prescribed after PMP initiation was 1,242 morphine milligram equivalents (MME), compared with 2,421 MME prior to implementation of the protocol (P < .0001).
After the protocol was implemented, Dr Gross and her colleagues found the number of patients for whom a refill was prescribed dropped from 39.7% to 28.1%, and the size of those refills dropped from 1,032 MME to 213 MME on average.
“By having a comprehensive pain management protocol, we can reduce the amount of pain medications we prescribe for outpatient use, after discharge from the trauma service,” said Dr Gross, “…not only by decreasing the number of refills we were providing, but also the amount of pain medications that was prescribed within these refills.”
A Press Ganey survey analysis of patients during the month before and the month after the PMP implementation, found a significant increase in overall patient satisfaction and satisfaction with pain management, according to Dr Gross.
Certain limitations include not being able to confirm whether patients received prescription medication elsewhere, nor any concrete data on patient satisfaction after discharge other than an inference based on fewer refills and lower refill MME.
New Assay Helps Distinguish Viral and Bacterial Infections in Children
BY IAN LACY
An assay testing the presence of three blood-borne host-proteins shows promise in accurately identifying viral and bacterial infections in febrile children, a validation study found.
The three proteins that the ImmunoXpert assay uses to differentiate between viral and bacterial infections are: viral-induced tumor necrosis factor-related apoptosis-inducing ligand (TRAIL), interferon gamma-induced protein-10 (IP-10), and bacterial-induced C-reactive protein (CRP). While TRAIL and IP-10 are novel identifiers, CRP has been used in traditional bacterial detecting assays, Isaac Srugo, MD, and colleagues reported.
The investigators identified 597 potential stored patient serum samples from patients admitted to multiple pediatric EDs and wards in Israel and Switzerland, and ultimately, 361 samples were selected for assay testing.1
Of the 361 patients whose samples were selected for testing, the assay identified 209 patients (58%) with a viral infection, 99 patients (27%) with a bacterial infection, and the remaining 53 patients (15%) with an equivocal outcome, according to Dr Srugo of the Ruth and Bruce Rappaport Faculty of Medicine, Technion-Israel Institute of Technology, Haifa, Israel, and his colleagues. The 307 patients with a bacterial or viral diagnosis had sensitivity of 93.8% (95% confidence interval [CI], 87.8%-99.8%) and specificity of 89.8% (CI, 85.6%-94.0%). There were four false-negative and 21 false-positive findings.
The study found that TRAIL and IP-10 were present in higher levels in children with viral infections compared to children with bacterial infections. The opposite was true of CRP results, with significantly lower levels of CRP in children with viral infections compared to those in children with bacterial infections.
“Notably, among the indeterminate diagnosis patients without a reference standard, the assay gave a bacterial or viral outcome for 69% of the cases (the rest were equivocal), with half of these yielding a score associated with a particularly high degree of assay diagnostic confidence,” investigators said. “This finding suggests that the assay may be applicable to ‘harder-to-diagnose’ cases in real-life clinical settings.”
Also, the assay “exhibits consistent performance across a wide range of ages [3 months to 18 years], time from symptom onset, and clinical syndromes,” Dr Srugo and his associates said.
1. Srugo I, Klein A, Stein M, et al. Validation of a novel assay to distinguish bacterial and viral infections. Pediatrics. 2017 Sep 13. doi:10.1542/peds.2016-3453 [Epub ahead of print]
Emergency Medicine Associate Editor-in-Chief, Francis L. Counselman, MD, to Receive ACEP Award for Outstanding Contribution in Education
Francis L. Counselman, MD, Associate Editor-in-Chief of Emergency Medicine will receive the 2017 American College of Emergency Physicians (ACEP) Award for Outstanding Contribution in Education, at the ACEP Scientific Assembly this month. This award is “presented to an ACEP member who has made a significant contribution to the educational aspects of the specialty,” and is the highest award ACEP gives annually regarding education and teaching.
Dr. Counselman is the founding Chairman of the Department of Emergency Medicine at Eastern Virginia Medical School (EVMS) in Norfolk, Virginia. Founded in 1992, he continues to serve as Chairman. He served as Program Director of the Emergency Medicine residency program at EVMS from 1990 to 2010. In 1999, he was made the EVMS Distinguished Professor of Emergency Medicine. He is the recipient of numerous teaching awards, including the Accreditation Council for Graduate Medical Education (ACGME) Parker Palmer “Courage to Teach” Award; the Emergency Medicine Residents’ Association (EMRA) Residency Director of the Year Award; and the EVMS Award for Outstanding Faculty Achievement. He has served as President of the Association of Academic Chairs of Emergency Medicine (AACEM) and the American Board of Emergency Medicine (ABEM). He is also a frequently invited speaker at state and national meetings. Dr Counselman has served on the Editorial Board of Emergency Medicine since 1999; and has been the Associate Editor-in-Chief since 2006.
Soccer-Playing Girls Five Times More Likely to Return to Same-Day Play After Concussion
BY KARI OAKES
Soccer-playing girls were five times more likely to return to play on the day of a concussion as were their male peers, according to a study presented at the annual meeting of the American Academy of Pediatrics.
Records from 87 soccer players aged 7 to 18 years (median age, 14 years) were examined in a retrospective review of patients seen over a 2-year period by a single physician at a pediatric sports medicine center. Of these, two thirds (n = 58) were girls.
Thirty of the 58 girls (51.7%) reported that they returned to play on the day of their concussion, compared to five of 29 boys (17.2%; P = .002). This difference in reporting yielded an odds ratio of 5.14 (95% CI, 1.72-15.3) for girls returning to same-day play, compared with boys who sustained a concussion.
The soccer players included children participating in recreational, club, and school-sponsored soccer, said senior author Shane M. Miller, MD, in an interview. All patients were assessed according to a standardized concussion protocol that involved a neurological examination and validated concussion evaluation tests, including the Immediate Post-Concussion Assessment and Cognitive Test and the Sports Concussion Assessment Tool.
As soccer has grown in popularity as a youth sport, so has the number of reported concussions. “The incidence of reported concussions has increased 1,600% from 1990 to 2014,” wrote Dr Miller and his coauthors in the abstract accompanying the presentation. Dr Miller said that girls are 1.5 times more likely than boys to sustain a concussion while playing soccer.
While seeing the patients who were the subject of the study, Dr Miller realized that most of the soccer players had not come out of play for evaluation after the head impact. Rather, they had continued to play, only later reporting concussion symptoms to coaches, trainers, or parents.
“The athletes may have chosen not to say anything because they didn’t want to come out of the game,” said Dr Miller, a sports medicine physician at Texas Scottish Rite Hospital for Children, Dallas.
“I was surprised by the significant degree of difference” between male and female soccer players, said Dr Miller. The study was not designed to get at the reason for the discrepancy, so Dr Miller could not say with certainty whether awareness of concussion symptoms is significantly lower for female athletes, or whether the athletic culture more strongly encourages minimization of symptoms for girls than boys. In any case, he said, there is room for education of players, coaches, and families to raise awareness of the importance of recognizing and reporting concussion, and then removing the affected athlete from play,
Dr Miller said that future research directions include collaboration with other facilities to conduct prospective research using a concussion registry. This will allow more robust statistical analysis and help ascertain the degree of regional variation in pediatric sports concussion management.
“Current education efforts may not be enough to help athletes, parents, and coaches identify concussion symptoms, know the guidelines for immediate removal from play, and understand the risks of returning to play after an injury. More research is needed on how to better spread this message intended to protect the health of young athletes….” Aaron Zynda, the study’s first author and clinical research coordinator at Texas Scottish Rite, said in a press release accompanying the abstract. “Concussion recognition and identification is a team effort,” he said.
Opioid Management Protocol Reduced Trauma Patient Pain Medication Use
BY ELI ZIMMERMAN
A pain management protocol implemented in a trauma service reduced opioid intake in trauma patients while improving patient satisfaction, according to a retrospective study.
The opioid epidemic continues to grow every day, partly as a result of irresponsible overprescribing of opioid medication, according to Jessica Gross, MB BAO BCh, a trauma surgeon from Wake Forest Baptist Health, North Carolina, at the American Association for the Surgery of Trauma annual meeting. Dr Gross and her colleagues developed a pain management protocol (PMP) to provide adequate pain control while using fewer opioids in the postdischarge setting. They tested their PMP through a retrospective chart review of 498 patients admitted to the trauma service between January 2015 and December 2016, half of whom were admitted before the PMP was initiated and half of whom were admitted afterward.
The PMP involved a stepped approach to treating pain, with acetaminophen or ibuprofen as needed for mild pain, one 5-mg tablet of oxycodone/acetaminophen every 6 hours for moderate pain, two tablets for severe pain, and 50 to 100 mg of tramadol every 6 hours for breakthrough pain.
Counseling services for patients who were found to be in danger of substance abuse were provided in the hospital, and at discharge, patients received a weaning plan for their medication, according to Dr Gross.
If the short-acting medications were found to be inadequate to control pain, patients were given slow-release pain medication as needed.
The average total medication, which included medication given at discharge and for refills, prescribed after PMP initiation was 1,242 morphine milligram equivalents (MME), compared with 2,421 MME prior to implementation of the protocol (P < .0001).
After the protocol was implemented, Dr Gross and her colleagues found the number of patients for whom a refill was prescribed dropped from 39.7% to 28.1%, and the size of those refills dropped from 1,032 MME to 213 MME on average.
“By having a comprehensive pain management protocol, we can reduce the amount of pain medications we prescribe for outpatient use, after discharge from the trauma service,” said Dr Gross, “…not only by decreasing the number of refills we were providing, but also the amount of pain medications that was prescribed within these refills.”
A Press Ganey survey analysis of patients during the month before and the month after the PMP implementation, found a significant increase in overall patient satisfaction and satisfaction with pain management, according to Dr Gross.
Certain limitations include not being able to confirm whether patients received prescription medication elsewhere, nor any concrete data on patient satisfaction after discharge other than an inference based on fewer refills and lower refill MME.
New Assay Helps Distinguish Viral and Bacterial Infections in Children
BY IAN LACY
An assay testing the presence of three blood-borne host-proteins shows promise in accurately identifying viral and bacterial infections in febrile children, a validation study found.
The three proteins that the ImmunoXpert assay uses to differentiate between viral and bacterial infections are: viral-induced tumor necrosis factor-related apoptosis-inducing ligand (TRAIL), interferon gamma-induced protein-10 (IP-10), and bacterial-induced C-reactive protein (CRP). While TRAIL and IP-10 are novel identifiers, CRP has been used in traditional bacterial detecting assays, Isaac Srugo, MD, and colleagues reported.
The investigators identified 597 potential stored patient serum samples from patients admitted to multiple pediatric EDs and wards in Israel and Switzerland, and ultimately, 361 samples were selected for assay testing.1
Of the 361 patients whose samples were selected for testing, the assay identified 209 patients (58%) with a viral infection, 99 patients (27%) with a bacterial infection, and the remaining 53 patients (15%) with an equivocal outcome, according to Dr Srugo of the Ruth and Bruce Rappaport Faculty of Medicine, Technion-Israel Institute of Technology, Haifa, Israel, and his colleagues. The 307 patients with a bacterial or viral diagnosis had sensitivity of 93.8% (95% confidence interval [CI], 87.8%-99.8%) and specificity of 89.8% (CI, 85.6%-94.0%). There were four false-negative and 21 false-positive findings.
The study found that TRAIL and IP-10 were present in higher levels in children with viral infections compared to children with bacterial infections. The opposite was true of CRP results, with significantly lower levels of CRP in children with viral infections compared to those in children with bacterial infections.
“Notably, among the indeterminate diagnosis patients without a reference standard, the assay gave a bacterial or viral outcome for 69% of the cases (the rest were equivocal), with half of these yielding a score associated with a particularly high degree of assay diagnostic confidence,” investigators said. “This finding suggests that the assay may be applicable to ‘harder-to-diagnose’ cases in real-life clinical settings.”
Also, the assay “exhibits consistent performance across a wide range of ages [3 months to 18 years], time from symptom onset, and clinical syndromes,” Dr Srugo and his associates said.
1. Srugo I, Klein A, Stein M, et al. Validation of a novel assay to distinguish bacterial and viral infections. Pediatrics. 2017 Sep 13. doi:10.1542/peds.2016-3453 [Epub ahead of print]
Emergency Medicine Associate Editor-in-Chief, Francis L. Counselman, MD, to Receive ACEP Award for Outstanding Contribution in Education
Francis L. Counselman, MD, Associate Editor-in-Chief of Emergency Medicine will receive the 2017 American College of Emergency Physicians (ACEP) Award for Outstanding Contribution in Education, at the ACEP Scientific Assembly this month. This award is “presented to an ACEP member who has made a significant contribution to the educational aspects of the specialty,” and is the highest award ACEP gives annually regarding education and teaching.
Dr. Counselman is the founding Chairman of the Department of Emergency Medicine at Eastern Virginia Medical School (EVMS) in Norfolk, Virginia. Founded in 1992, he continues to serve as Chairman. He served as Program Director of the Emergency Medicine residency program at EVMS from 1990 to 2010. In 1999, he was made the EVMS Distinguished Professor of Emergency Medicine. He is the recipient of numerous teaching awards, including the Accreditation Council for Graduate Medical Education (ACGME) Parker Palmer “Courage to Teach” Award; the Emergency Medicine Residents’ Association (EMRA) Residency Director of the Year Award; and the EVMS Award for Outstanding Faculty Achievement. He has served as President of the Association of Academic Chairs of Emergency Medicine (AACEM) and the American Board of Emergency Medicine (ABEM). He is also a frequently invited speaker at state and national meetings. Dr Counselman has served on the Editorial Board of Emergency Medicine since 1999; and has been the Associate Editor-in-Chief since 2006.
Soccer-Playing Girls Five Times More Likely to Return to Same-Day Play After Concussion
BY KARI OAKES
Soccer-playing girls were five times more likely to return to play on the day of a concussion as were their male peers, according to a study presented at the annual meeting of the American Academy of Pediatrics.
Records from 87 soccer players aged 7 to 18 years (median age, 14 years) were examined in a retrospective review of patients seen over a 2-year period by a single physician at a pediatric sports medicine center. Of these, two thirds (n = 58) were girls.
Thirty of the 58 girls (51.7%) reported that they returned to play on the day of their concussion, compared to five of 29 boys (17.2%; P = .002). This difference in reporting yielded an odds ratio of 5.14 (95% CI, 1.72-15.3) for girls returning to same-day play, compared with boys who sustained a concussion.
The soccer players included children participating in recreational, club, and school-sponsored soccer, said senior author Shane M. Miller, MD, in an interview. All patients were assessed according to a standardized concussion protocol that involved a neurological examination and validated concussion evaluation tests, including the Immediate Post-Concussion Assessment and Cognitive Test and the Sports Concussion Assessment Tool.
As soccer has grown in popularity as a youth sport, so has the number of reported concussions. “The incidence of reported concussions has increased 1,600% from 1990 to 2014,” wrote Dr Miller and his coauthors in the abstract accompanying the presentation. Dr Miller said that girls are 1.5 times more likely than boys to sustain a concussion while playing soccer.
While seeing the patients who were the subject of the study, Dr Miller realized that most of the soccer players had not come out of play for evaluation after the head impact. Rather, they had continued to play, only later reporting concussion symptoms to coaches, trainers, or parents.
“The athletes may have chosen not to say anything because they didn’t want to come out of the game,” said Dr Miller, a sports medicine physician at Texas Scottish Rite Hospital for Children, Dallas.
“I was surprised by the significant degree of difference” between male and female soccer players, said Dr Miller. The study was not designed to get at the reason for the discrepancy, so Dr Miller could not say with certainty whether awareness of concussion symptoms is significantly lower for female athletes, or whether the athletic culture more strongly encourages minimization of symptoms for girls than boys. In any case, he said, there is room for education of players, coaches, and families to raise awareness of the importance of recognizing and reporting concussion, and then removing the affected athlete from play,
Dr Miller said that future research directions include collaboration with other facilities to conduct prospective research using a concussion registry. This will allow more robust statistical analysis and help ascertain the degree of regional variation in pediatric sports concussion management.
“Current education efforts may not be enough to help athletes, parents, and coaches identify concussion symptoms, know the guidelines for immediate removal from play, and understand the risks of returning to play after an injury. More research is needed on how to better spread this message intended to protect the health of young athletes….” Aaron Zynda, the study’s first author and clinical research coordinator at Texas Scottish Rite, said in a press release accompanying the abstract. “Concussion recognition and identification is a team effort,” he said.
Opioid Management Protocol Reduced Trauma Patient Pain Medication Use
BY ELI ZIMMERMAN
A pain management protocol implemented in a trauma service reduced opioid intake in trauma patients while improving patient satisfaction, according to a retrospective study.
The opioid epidemic continues to grow every day, partly as a result of irresponsible overprescribing of opioid medication, according to Jessica Gross, MB BAO BCh, a trauma surgeon from Wake Forest Baptist Health, North Carolina, at the American Association for the Surgery of Trauma annual meeting. Dr Gross and her colleagues developed a pain management protocol (PMP) to provide adequate pain control while using fewer opioids in the postdischarge setting. They tested their PMP through a retrospective chart review of 498 patients admitted to the trauma service between January 2015 and December 2016, half of whom were admitted before the PMP was initiated and half of whom were admitted afterward.
The PMP involved a stepped approach to treating pain, with acetaminophen or ibuprofen as needed for mild pain, one 5-mg tablet of oxycodone/acetaminophen every 6 hours for moderate pain, two tablets for severe pain, and 50 to 100 mg of tramadol every 6 hours for breakthrough pain.
Counseling services for patients who were found to be in danger of substance abuse were provided in the hospital, and at discharge, patients received a weaning plan for their medication, according to Dr Gross.
If the short-acting medications were found to be inadequate to control pain, patients were given slow-release pain medication as needed.
The average total medication, which included medication given at discharge and for refills, prescribed after PMP initiation was 1,242 morphine milligram equivalents (MME), compared with 2,421 MME prior to implementation of the protocol (P < .0001).
After the protocol was implemented, Dr Gross and her colleagues found the number of patients for whom a refill was prescribed dropped from 39.7% to 28.1%, and the size of those refills dropped from 1,032 MME to 213 MME on average.
“By having a comprehensive pain management protocol, we can reduce the amount of pain medications we prescribe for outpatient use, after discharge from the trauma service,” said Dr Gross, “…not only by decreasing the number of refills we were providing, but also the amount of pain medications that was prescribed within these refills.”
A Press Ganey survey analysis of patients during the month before and the month after the PMP implementation, found a significant increase in overall patient satisfaction and satisfaction with pain management, according to Dr Gross.
Certain limitations include not being able to confirm whether patients received prescription medication elsewhere, nor any concrete data on patient satisfaction after discharge other than an inference based on fewer refills and lower refill MME.
The Role of Synovial Cytokines in the Diagnosis of Periprosthetic Joint Infections: Current Concepts
Take-Home Points
- In cases of failed TJA, it is important to differentiate between septic and aseptic etiologies.
- Chronic and low-grade infections are challenging for orthopedic surgeons, as the symptoms often overlap with aseptic etiologies.
- Verification of infection eradication before beginning the second-stage reimplantation surgery is extremely important, but pre- and intraoperative findings can be unreliable.
- Synovial fluid cytokines have been shown to accurately diagnose PJIs.
- Synovial fluid cytokines may help surgeons differentiate between septic and aseptic cases of failed TJA.
Total joint arthroplasty (TJA) is an effective procedure that has been extensively used to relieve pain and improve quality of life in patients with various forms of joint disease. Although advances in technology and surgical technique have improved the success of TJA, periprosthetic joint infection (PJI) remains a serious complication. In the United States, it is estimated that PJI is the most common reason for total knee arthroplasty failure and the third most common reason for total hip arthroplasty revision.1 Although the incidence of PJI is 1% to 2%, the dramatic increase in TJA volume is expected to be accompanied by a similar rise in the number of infected TJAs; that number is expected to exceed 60,000 in the United States by 2020.2 Moreover, management of PJI is expensive and imposes a heavy burden on the healthcare system, with costs expected to hit $20 billion by 2020 in the US.2 Therefore, treating asepsis cases as infections imposes a heavy burden on the healthcare system and may result in excessive morbidity.3 At the same time, inadequate management of a PJI may result in recurrences that require infection treatment with morbid procedures, such as arthrodesis or amputation. Accurate diagnosis of PJI is of paramount importance in preventing potential implications of a misdiagnosed case. Unfortunately, the PJI diagnosis is extremely challenging, and the available diagnostic tests are often unreliable.4 Thus, research has recently focused on use of several synovial fluid cytokines in the detection of PJI.5-7 In this article, we provide an overview of the synovial biomarkers being used to diagnose PJI.
Diagnosis of Periprosthetic Joint Infection
Differentiating between septic and aseptic failed TJA is important, as the treatment options differ considerably. PJI can be broadly classified as acute or early postoperative (<6 weeks), late chronic (indolent onset), and acute-on-chronic (acute onset in well-functioning prosthesis, secondary to hematogenous spread).8 The acute and acute-on-chronic presentations are often associated with obvious signs of infection.9 However, chronic and low-grade infections pose a challenge to modern orthopedic practice, as the symptoms often overlap with that of aseptic causes of TJA failure.10 As a result, the International Consensus Group on Periprosthetic Joint Infection developed complex criteria using the Musculoskeletal Infection Society definition of PJI and involving a battery of tests for PJI diagnosis.11 According to these criteria, PJI is diagnosed when 1 of the 2 major criteria or 3 of the 5 minor criteria are met (Table 1).
Although these criteria constitute the most agreed on and widely used standard for PJI diagnosis, the definition is complex and often incomplete until surgical intervention. An ideal diagnostic test would aid in managing a PJI and provide results before a treatment decision is made. Many revision surgeries are being performed with insufficient information about the true diagnosis, and the diagnosis might change during or after surgery. About 10% of the revisions presumed to be aseptic may unexpectedly grow cultures during surgery and thereby satisfy the criteria for PJI after surgery.12 Moreover, with the use of novel methods such as polymerase chain reaction, microorganisms were identified in more than three-fourths of the presumed aseptic revisions.13 The optimal management of such cases is controversial, and it is unclear whether positive cultures should be treated as possible contaminants or true infection.12,14
Verification of Infection Eradication
A 2-stage revision procedure, widely accepted as the standard treatment for PJI, has success rates approaching 94%.15 In this procedure, it is important to verify infection eradication before beginning the second-stage reimplantation. Verification is crucial in avoiding reimplantation of an infected joint.16 After the first stage, patients are usually administered intravenous antibiotics for at least 6 weeks; these antibiotics are then withheld, and systemic inflammatory markers are evaluated for infection eradication. Although reliable criteria have been established for PJI diagnosis, guidelines for detecting eradication of infection are rudimentary. Most surgeons monitor the decrease in serologic markers, such as erythrocyte sedimentation rate and C-reactive protein (CRP) level, to assess the response to treatment. However, noninfectious etiologies may result in continued elevation of these markers.17 Even though aspirations are often performed to diagnose persistent infection before the second-stage procedure, their diagnostic utility may be limited.18 Use of cultures is also limited, as presence of antibiotic-loaded spacers can decrease the sensitivity of culture.19 Inadequate diagnosis often leads to unnecessary continuation of antimicrobial therapy or additional surgical débridement. Nuclear scans often remain positive because of aseptic inflammation related to surgery and are not useful in documenting sepsis arrest.20 Given the limitations of available tests, novel strategies for identifying the presence of infection at the second stage are being tested.
Synovial Fluid Cytokines
PJI pathogenesis begins with colonization of the implant surfaces with microorganisms and subsequent formation of biofilms.21 The human immune system is activated by the microbial products, cell wall components, and various biofilm proteins. Immune cells are recruited to the site, where they secrete a myriad of inflammatory biomarkers, such as cytokines, which promote further recruitment of inflammatory cells and aid in the eradication of pathogens.9 These inflammatory cytokines and cells are involved in aseptic inflammatory joint conditions, such as rheumatoid arthritis22,23; however, some are specifically involved in immune pathways combating pathogens.24 This action is the basis for increasing interest in using various synovial fluid cytokines and other biomarkers in the diagnosis of PJI. Here we describe some of the commonly studied cytokines.
Interleukin 1β
Interleukin 1β (IL-1β) is a major proinflammatory cytokine that is synthesized by multiple cells, including macrophages and monocytes.25 IL-1β is produced in response to microorganisms, other cytokines, antigen-presenting cells, and immune complexes; stimulates production of acute-phase proteins by the liver; and is an important pyrogen.25 Deirmengian and colleagues5 found that synovial IL-1β increased 258-fold in patients with a PJI. Studies have found that synovial IL-1β has sensitivity ranging from 66.7% to 100% and specificity ranging from 87% to 100%, with 1 study reporting an accuracy of 100%.5,6,26,27
Interleukin 6
Also produced by macrophages and monocytes, interleukin 6 (IL-6) is a potent stimulator of acute-phase proteins.28,29 IL-6 has a role as a chemoattractant and helps with cell differentiation when changing from innate to acquired immunity.30 It is also used as an aid in diagnosing PJI; it has sensitivity ranging from 62% to 100% and specificity ranging from 85% to 100%.5,6,26,31,32 Synovial IL-6 measurements were more accurate than serum IL-6 measurements.26 Furthermore, synovial IL-6 can be increased up to 27-fold in PJI cases.5 In one study, synovial IL-6 levels >2100 pg/mL had sensitivity of 62.5% and specificity of 85.7% in PJI diagnosis26; in another study, an IL-6 threshold of 4270 pg/mL had sensitivity of 87.1%, specificity of 100%, and accuracy of 94.6%.31
C-Reactive Protein
CRP is an acute-phase reactant. Blood levels increase in response to aseptic inflammatory processes and systemic infection.33 CRP plays an important role in host defense by activating complement and helping mediate phagocytosis.33,34 Although serum CRP levels have been used in diagnosing PJIs,6 they can yield false-negative results.35,36 Therefore, attention turned to synovial CRP levels, which were found to be increased 13-fold in PJI cases.5 It has been shown that synovial CRP levels are significantly higher in infected vs noninfected prosthetic joints34 and had diagnostic accuracy better than that of serum CRP levels in diagnosing PJI.37 One study found that CRP at a threshold of 3.7 mg/L had sensitivity of 84%, specificity of 97.1%, and accuracy of 91.5%,37 whereas another study found that CRP at a threshold of 3.61 mg/L had sensitivity of 87.1%, specificity of 97.7%, and accuracy of 93.3%.31
α-Defensin
α-Defensin, a natural peptide produced and secreted by neutrophils in response to pathogens, has antimicrobial and cytotoxic properties,38-40 signals for the secretion of various cytokines, and acts as a chemoattractant for various immune cells.41 Deirmengian and colleagues6 found that α-defensin was consistently elevated in patients with PJI. α-Defensin is extremely accurate in diagnosing PJI; it has sensitivity ranging from 97% to 100% and specificity ranging from 96% to 100%.6,27,42 Moreover, α-defensin was effective in diagnosing PJI caused by a wide spectrum of organisms, including various low-virulence bacteria and fungi.43
Leukocyte Esterase
Leukocyte esterase is an enzyme produced and secreted by neutrophils at sites of active infection.7,44 Testing for this enzyme is performed with a colorimetric strip and was originally performed for the diagnosis of urinary tract infections.44,45 In a study conducted by Parvizi and colleagues,7 this strip was used to test for leukocyte esterase in synovial fluid samples; a ++ reading was found to have sensitivity of 80.6% and specificity of 100% in diagnosing knee PJI. Similarly, De Vecchi and colleagues45 found sensitivity of 92.6% and specificity of 97%.
Other Synovial Markers
Research has identified numerous molecular biomarkers that may be associated with the pathogenesis of PJI. Although several (eg, cytokines) have demonstrated higher levels in synovial fluid in patients with PJI than in normal controls, only a few have had clinically relevant diagnostic utility.6 Deirmengian and colleagues6 screened 43 synovial fluid biomarkers that potentially could be used in the diagnosis of PJI. Besides the cytokine α-defensin, 4 other biomarkers—lactoferrin, neutrophil gelatinase-associated lipocalcin, neutrophil elastase 2, and bactericidal/permeability-increasing protein—had accuracy of 100%. In addition, 8 cytokines and biomarkers (IL-8, CRP, resistin, thrombospondin, IL-1β, IL-6, IL-10, IL-1α) had area under the curve values higher than 0.9. Studies have also evaluated the diagnostic utility of metabolic products such as lactate, lactate dehydrogenase, and glucose; their accuracy was comparable to that of serum CRP.32
Serum Markers
In addition to the synovial fluid cytokines, several serum inflammatory cytokines have been studied as potential targets in diagnosing infection. Serum IL-6 has had excellent diagnostic accuracy46 and, when combined with CRP, could increase sensitivity in diagnosing PJI; such a combination (vs either test alone) could be useful in screening patients.47,48 Biomarkers such as tumor necrosis factor α and procalcitonin are considered very specific for PJI and may be useful in confirmatory testing.48 Evidence also suggests that toll-like receptor 2 proteins are elevated in the serum of patients with PJI and therefore are a potential diagnostic tool.49
Limitations of Synovial Cytokines
The literature suggests that some synovial fluid cytokines have promise.6 However, the best biomarker or combination of biomarkers is yet to be determined. Results have been consistent with α-defensin and other cytokines but mixed with IL-6 and still others32,42,50 (Table 2).
Information on the utility of synovial biomarkers in detecting persistent infection is limited. Frangiamore and colleagues50 found that IL-1 and IL-6 levels decreased between the stages of 2-stage revision. Unfortunately, none of the synovial fluid cytokines investigated (IL-1, IL-2, IL-6, IL-8, Il-10, interferon γ, granulocyte macrophage-colony stimulating factor, tumor necrosis factor α, IL-12p70) satisfactorily detected resolution of infection in the setting of prior treatment for PJI. Although cytokines are expected to be elevated in the presence of infection, the internal milieu at the time of stage 2 of the revision makes diagnosis of infection difficult. In addition, presence of spacer particles and recent surgery may activate immune pathways and yield false-positive results. Furthermore, antibiotic cement spacers may suppress the microorganisms to very low levels and yield false-negative results even if these organisms remain virulent.19
Even though the synovial molecular markers can detect the presence of infection, they are unable to identify pathogens. As identifying the pathogen is important in the treatment of PJI, there has been interest in using polymerase chain reaction (PCR) techniques.51 These tests may also provide specific information about the pathogen, such as its antibiotic sensitivity. A recently developed technology, the Ibis T5000 Universal Biosensor (Ibis Biosciences), uses novel pan-domain primers in a series of PCRs. This biosensor is useful in diagnosing infections when cultures are negative and appears to be more accurate than conventional PCR.13 As reported by Jacovides and colleagues,13 this novel PCR technique identified an organism in about 88% of presumed cases of aseptic revision.
Conclusion
PJI poses an extreme challenge to the healthcare system. Given the morbidity associated with improper management of PJI, accurate diagnosis is of paramount importance. Given the limitations of current tests, synovial fluid cytokines hold promise in the diagnosis of PJIs. However, these cytokines are expensive, and their clinical utility in PJI management is not well established. More research is needed before guidelines for synovial fluid cytokines and biomarkers can replace or be incorporated into guidelines for the treatment of PJIs.
1 Parvizi J, Adeli B, Zmistowski B, Restrepo C, Greenwald AS. Management of periprosthetic joint infection: the current knowledge: AAOS exhibit selection. J Bone Joint Surg Am. 2012;94(14):e104.
2. Kurtz SM, Lau E, Watson H, Schmier JK, Parvizi J. Economic burden of periprosthetic joint infection in the United States. J Arthroplasty. 2012;27(8 suppl):61-65.e1.
3. Sierra RJ, Trousdale RT, Pagnano MW. Above-the-knee amputation after a total knee replacement: prevalence, etiology, and functional outcome. J Bone Joint Surg Am. 2003;85(6):1000-1004.
4. Bauer TW, Parvizi J, Kobayashi N, Krebs V. Diagnosis of periprosthetic infection. J Bone Joint Surg Am. 2006;88(4):869-882.
5. Deirmengian C, Hallab N, Tarabishy A, et al. Synovial fluid biomarkers for periprosthetic infection. Clin Orthop Relat Res. 2010;468(8):2017-2023.
6. Deirmengian C, Kardos K, Kilmartin P, Cameron A, Schiller K, Parvizi J. Diagnosing periprosthetic joint infection: has the era of the biomarker arrived? Clin Orthop Relat Res. 2014;472(11):3254-3262.
7. Parvizi J, Jacovides C, Antoci V, Ghanem E. Diagnosis of periprosthetic joint infection: the utility of a simple yet unappreciated enzyme. J Bone Joint Surg Am. 2011;93(24):2242-2248.
8. Kuiper JW, Willink RT, Moojen DJF, van den Bekerom MP, Colen S. Treatment of acute periprosthetic infections with prosthesis retention: review of current concepts. World J Orthop. 2014;5(5):667-676.
9. Zimmerli W, Trampuz A, Ochsner PE. Prosthetic-joint infections. N Engl J Med. 2004;351(16):1645-1654.
10. Osmon DR, Berbari EF, Berendt AR, et al. Diagnosis and management of prosthetic joint infection: clinical practice guidelines by the Infectious Diseases Society of America. Clin Infect Dis. 2013;56(1):e1-e25.
11. Parvizi J, Gehrke T; International Consensus Group on Periprosthetic Joint Infection. Definition of periprosthetic joint infection. J Arthroplasty. 2014;29(7):1331.
12. Saleh A, Guirguis A, Klika AK, Johnson L, Higuera CA, Barsoum WK. Unexpected positive intraoperative cultures in aseptic revision arthroplasty. J Arthroplasty. 2014;29(11):2181-2186.
13. Jacovides CL, Kreft R, Adeli B, Hozack B, Ehrlich GD, Parvizi J. Successful identification of pathogens by polymerase chain reaction (PCR)–based electron spray ionization time-of-flight mass spectrometry (ESI-TOF-MS) in culture-negative periprosthetic joint infection. J Bone Joint Surg Am. 2012;94(24):2247-2254.
14. Barrack RL, Aggarwal A, Burnett RS, et al. The fate of the unexpected positive intraoperative cultures after revision total knee arthroplasty. J Arthroplasty. 2007;22(6 suppl 2):94-99.
15. Macheras GA, Koutsostathis SD, Kateros K, Papadakis S, Anastasopoulos P. A two stage re-implantation protocol for the treatment of deep periprosthetic hip infection. Mid to long-term results. Hip Int. 2012;22(suppl 8):S54-S61.
16. George J, Kwiecien G, Klika AK, et al. Are frozen sections and MSIS criteria reliable at the time of reimplantation of two-stage revision arthroplasty? Clin Orthop Relat Res. 2016;474(7):1619-1626.
17. Kusuma SK, Ward J, Jacofsky M, Sporer SM, Della Valle CJ. What is the role of serological testing between stages of two-stage reconstruction of the infected prosthetic knee? Clin Orthop Relat Res. 2011;469(4):1002-1008.
18. Lonner JH, Siliski JM, Della Valle C, DiCesare P, Lotke PA. Role of knee aspiration after resection of the infected total knee arthroplasty. Am J Orthop. 2001;30(4):305-309.
19. Mont MA, Waldman BJ, Hungerford DS. Evaluation of preoperative cultures before second-stage reimplantation of a total knee prosthesis complicated by infection. A comparison-group study. J Bone Joint Surg Am. 2000;82(11):1552-1557.
20. Love C, Marwin SE, Palestro CJ. Nuclear medicine and the infected joint replacement. Semin Nucl Med. 2009;39(1):66-78.
21. Zimmerli W, Moser C. Pathogenesis and treatment concepts of orthopaedic biofilm infections. FEMS Immunol Med Microbiol. 2012;65(2):158-168.
22. Fontana A, Hengartner H, Weber E, Fehr K, Grob PJ, Cohen G. Interleukin 1 activity in the synovial fluid of patients with rheumatoid arthritis. Rheumatol Int. 1982;2(2):49-53.
23. Guerne PA, Zuraw BL, Vaughan JH, Carson DA, Lotz M. Synovium as a source of interleukin 6 in vitro. Contribution to local and systemic manifestations of arthritis. J Clin Invest. 1989;83(2):585-592.
24. Wang G. Human antimicrobial peptides and proteins. Pharmaceuticals (Basel). 2014;7(5):545-594.
25. Stylianou E, Saklatvala J. Interleukin-1. Int J Biochem Cell Biol. 1998;30(10):1075-1079.
26. Gollwitzer H, Dombrowski Y, Prodinger PM, et al. Antimicrobial peptides and proinflammatory cytokines in periprosthetic joint infection. J Bone Joint Surg Am. 2013;95(7):644-651.
27. Deirmengian C, Kardos K, Kilmartin P, Cameron A, Schiller K, Parvizi J. Combined measurement of synovial fluid α-defensin and C-reactive protein levels: highly accurate for diagnosing periprosthetic joint infection. J Bone Joint Surg Am. 2014;96(17):1439-1445.
28. Randau TM, Friedrich MJ, Wimmer MD, et al. Interleukin-6 in serum and in synovial fluid enhances the differentiation between periprosthetic joint infection and aseptic loosening. PLoS One. 2014;9(2):e89045.
29. Heinrich PC, Castell JV, Andus T. Interleukin-6 and the acute phase response. Biochem J. 1990;265(3):621-636.
30. Scheller J, Chalaris A, Schmidt-Arras D, Rose-John S. The pro- and anti-inflammatory properties of the cytokine interleukin-6. Biochim Biophys Acta. 2011;1813(5):878-888.
31. Jacovides CL, Parvizi J, Adeli B, Jung KA. Molecular markers for diagnosis of periprosthetic joint infection. J Arthroplasty. 2011;26(6 suppl):99-103.e1.
32. Lenski M, Scherer MA. Synovial IL-6 as inflammatory marker in periprosthetic joint infections. J Arthroplasty. 2014;29(6):1105-1109.
33. Mortensen RF. C-reactive protein, inflammation, and innate immunity. Immunol Res. 2001;24(2):163-176.
34. Parvizi J, McKenzie JC, Cashman JP. Diagnosis of periprosthetic joint infection using synovial C-reactive protein. J Arthroplasty. 2012;27(8 suppl):12-16.
35. Ghanem E, Antoci V, Pulido L, Joshi A, Hozack W, Parvizi J. The use of receiver operating characteristics analysis in determining erythrocyte sedimentation rate and C-reactive protein levels in diagnosing periprosthetic infection prior to revision total hip arthroplasty. Int J Infect Dis. 2009;13(6):e444-e449.
36. Johnson AJ, Zywiel MG, Stroh A, Marker DR, Mont MA. Serological markers can lead to false negative diagnoses of periprosthetic infections following total knee arthroplasty. Int Orthop. 2011;35(11):1621-1626.
37. Parvizi J, Jacovides C, Adeli B, Jung KA, Hozack WJ. Mark B. Coventry award: synovial C-reactive protein: a prospective evaluation of a molecular marker for periprosthetic knee joint infection. Clin Orthop Relat Res. 2012;470(1):54-60.
38. Lehrer RI, Lichtenstein AK, Ganz T. Defensins: antimicrobial and cytotoxic peptides of mammalian cells. Annu Rev Immunol. 1993;11:105-128.
39. Ganz T, Selsted ME, Szklarek D, et al. Defensins. Natural peptide antibiotics of human neutrophils. J Clin Invest. 1985;76(4):1427-1435.
40. Chalifour A, Jeannin P, Gauchat JF, et al. Direct bacterial protein PAMP recognition by human NK cells involves TLRs and triggers alpha-defensin production. Blood. 2004;104(6):1778-1783.
41. Ulm H, Wilmes M, Shai Y, Sahl HG. Antimicrobial host defensins—specific antibiotic activities and innate defense modulation. Front Immunol. 2012;3:249.
42. Bingham J, Clarke H, Spangehl M, Schwartz A, Beauchamp C, Goldberg B. The alpha defensin-1 biomarker assay can be used to evaluate the potentially infected total joint arthroplasty. Clin Orthop Relat Res. 2014;472(12):4006-4009.
43. Deirmengian C, Kardos K, Kilmartin P, Gulati S, Citrano P, Booth RE. The alpha-defensin test for periprosthetic joint infection responds to a wide spectrum of organisms. Clin Orthop Relat Res. 2015;473(7):2229-2235.
44. Tischler EH, Cavanaugh PK, Parvizi J. Leukocyte esterase strip test: matched for Musculoskeletal Infection Society criteria. J Bone Joint Surg Am. 2014;96(22):1917-1920.
45. De Vecchi E, Villa F, Bortolin M, et al. Leucocyte esterase, glucose and C-reactive protein in the diagnosis of prosthetic joint infections: a prospective study. Clin Microbiol Infect. 2016;22(6):555-560.
46. Di Cesare PE, Chang E, Preston CF, Liu C. Serum interleukin-6 as a marker of periprosthetic infection following total hip and knee arthroplasty. J Bone Joint Surg Am. 2005;87(9):1921-1927.
47. Ettinger M, Calliess T, Kielstein JT, et al. Circulating biomarkers for discrimination between aseptic joint failure, low-grade infection, and high-grade septic failure. Clin Infect Dis. 2015;61(3):332-341.
48. Bottner F, Wegner A, Winkelmann W, Becker K, Erren M, Götze C. Interleukin-6, procalcitonin and TNF-alpha: markers of peri-prosthetic infection following total joint replacement. J Bone Joint Surg Br. 2007;89(1):94-99.
49. Galliera E, Drago L, Vassena C, et al. Toll-like receptor 2 in serum: a potential diagnostic marker of prosthetic joint infection? J Clin Microbiol. 2014;52(2):620-623.
50. Frangiamore SJ, Siqueira MB, Saleh A, Daly T, Higuera CA, Barsoum WK. Synovial cytokines and the MSIS criteria are not useful for determining infection resolution after periprosthetic joint infection explantation. Clin Orthop Relat Res. 2016;474(7):1630-1639.
51. Rasouli MR, Harandi AA, Adeli B, Purtill JJ, Parvizi J. Revision total knee arthroplasty: infection should be ruled out in all cases. J Arthroplasty. 2012;27(6):1239-1243.e1-e2.
52. Omar M, Ettinger M, Reichling M, et al. Synovial C-reactive protein as a marker for chronic periprosthetic infection in total hip arthroplasty. Bone Joint J. 2015;97(2):173-176.
53. Tetreault MW, Wetters NG, Moric M, Gross CE, Della Valle CJ. Is synovial C-reactive protein a useful marker for periprosthetic joint infection? Clin Orthop Relat Res. 2014;472(12):3997-4003.
54. Omar M, Ettinger M, Reichling M, et al. Preliminary results of a new test for rapid diagnosis of septic arthritis with use of leukocyte esterase and glucose reagent strips. J Bone Joint Surg Am. 2014;96(24):2032-2037.
Take-Home Points
- In cases of failed TJA, it is important to differentiate between septic and aseptic etiologies.
- Chronic and low-grade infections are challenging for orthopedic surgeons, as the symptoms often overlap with aseptic etiologies.
- Verification of infection eradication before beginning the second-stage reimplantation surgery is extremely important, but pre- and intraoperative findings can be unreliable.
- Synovial fluid cytokines have been shown to accurately diagnose PJIs.
- Synovial fluid cytokines may help surgeons differentiate between septic and aseptic cases of failed TJA.
Total joint arthroplasty (TJA) is an effective procedure that has been extensively used to relieve pain and improve quality of life in patients with various forms of joint disease. Although advances in technology and surgical technique have improved the success of TJA, periprosthetic joint infection (PJI) remains a serious complication. In the United States, it is estimated that PJI is the most common reason for total knee arthroplasty failure and the third most common reason for total hip arthroplasty revision.1 Although the incidence of PJI is 1% to 2%, the dramatic increase in TJA volume is expected to be accompanied by a similar rise in the number of infected TJAs; that number is expected to exceed 60,000 in the United States by 2020.2 Moreover, management of PJI is expensive and imposes a heavy burden on the healthcare system, with costs expected to hit $20 billion by 2020 in the US.2 Therefore, treating asepsis cases as infections imposes a heavy burden on the healthcare system and may result in excessive morbidity.3 At the same time, inadequate management of a PJI may result in recurrences that require infection treatment with morbid procedures, such as arthrodesis or amputation. Accurate diagnosis of PJI is of paramount importance in preventing potential implications of a misdiagnosed case. Unfortunately, the PJI diagnosis is extremely challenging, and the available diagnostic tests are often unreliable.4 Thus, research has recently focused on use of several synovial fluid cytokines in the detection of PJI.5-7 In this article, we provide an overview of the synovial biomarkers being used to diagnose PJI.
Diagnosis of Periprosthetic Joint Infection
Differentiating between septic and aseptic failed TJA is important, as the treatment options differ considerably. PJI can be broadly classified as acute or early postoperative (<6 weeks), late chronic (indolent onset), and acute-on-chronic (acute onset in well-functioning prosthesis, secondary to hematogenous spread).8 The acute and acute-on-chronic presentations are often associated with obvious signs of infection.9 However, chronic and low-grade infections pose a challenge to modern orthopedic practice, as the symptoms often overlap with that of aseptic causes of TJA failure.10 As a result, the International Consensus Group on Periprosthetic Joint Infection developed complex criteria using the Musculoskeletal Infection Society definition of PJI and involving a battery of tests for PJI diagnosis.11 According to these criteria, PJI is diagnosed when 1 of the 2 major criteria or 3 of the 5 minor criteria are met (Table 1).
Although these criteria constitute the most agreed on and widely used standard for PJI diagnosis, the definition is complex and often incomplete until surgical intervention. An ideal diagnostic test would aid in managing a PJI and provide results before a treatment decision is made. Many revision surgeries are being performed with insufficient information about the true diagnosis, and the diagnosis might change during or after surgery. About 10% of the revisions presumed to be aseptic may unexpectedly grow cultures during surgery and thereby satisfy the criteria for PJI after surgery.12 Moreover, with the use of novel methods such as polymerase chain reaction, microorganisms were identified in more than three-fourths of the presumed aseptic revisions.13 The optimal management of such cases is controversial, and it is unclear whether positive cultures should be treated as possible contaminants or true infection.12,14
Verification of Infection Eradication
A 2-stage revision procedure, widely accepted as the standard treatment for PJI, has success rates approaching 94%.15 In this procedure, it is important to verify infection eradication before beginning the second-stage reimplantation. Verification is crucial in avoiding reimplantation of an infected joint.16 After the first stage, patients are usually administered intravenous antibiotics for at least 6 weeks; these antibiotics are then withheld, and systemic inflammatory markers are evaluated for infection eradication. Although reliable criteria have been established for PJI diagnosis, guidelines for detecting eradication of infection are rudimentary. Most surgeons monitor the decrease in serologic markers, such as erythrocyte sedimentation rate and C-reactive protein (CRP) level, to assess the response to treatment. However, noninfectious etiologies may result in continued elevation of these markers.17 Even though aspirations are often performed to diagnose persistent infection before the second-stage procedure, their diagnostic utility may be limited.18 Use of cultures is also limited, as presence of antibiotic-loaded spacers can decrease the sensitivity of culture.19 Inadequate diagnosis often leads to unnecessary continuation of antimicrobial therapy or additional surgical débridement. Nuclear scans often remain positive because of aseptic inflammation related to surgery and are not useful in documenting sepsis arrest.20 Given the limitations of available tests, novel strategies for identifying the presence of infection at the second stage are being tested.
Synovial Fluid Cytokines
PJI pathogenesis begins with colonization of the implant surfaces with microorganisms and subsequent formation of biofilms.21 The human immune system is activated by the microbial products, cell wall components, and various biofilm proteins. Immune cells are recruited to the site, where they secrete a myriad of inflammatory biomarkers, such as cytokines, which promote further recruitment of inflammatory cells and aid in the eradication of pathogens.9 These inflammatory cytokines and cells are involved in aseptic inflammatory joint conditions, such as rheumatoid arthritis22,23; however, some are specifically involved in immune pathways combating pathogens.24 This action is the basis for increasing interest in using various synovial fluid cytokines and other biomarkers in the diagnosis of PJI. Here we describe some of the commonly studied cytokines.
Interleukin 1β
Interleukin 1β (IL-1β) is a major proinflammatory cytokine that is synthesized by multiple cells, including macrophages and monocytes.25 IL-1β is produced in response to microorganisms, other cytokines, antigen-presenting cells, and immune complexes; stimulates production of acute-phase proteins by the liver; and is an important pyrogen.25 Deirmengian and colleagues5 found that synovial IL-1β increased 258-fold in patients with a PJI. Studies have found that synovial IL-1β has sensitivity ranging from 66.7% to 100% and specificity ranging from 87% to 100%, with 1 study reporting an accuracy of 100%.5,6,26,27
Interleukin 6
Also produced by macrophages and monocytes, interleukin 6 (IL-6) is a potent stimulator of acute-phase proteins.28,29 IL-6 has a role as a chemoattractant and helps with cell differentiation when changing from innate to acquired immunity.30 It is also used as an aid in diagnosing PJI; it has sensitivity ranging from 62% to 100% and specificity ranging from 85% to 100%.5,6,26,31,32 Synovial IL-6 measurements were more accurate than serum IL-6 measurements.26 Furthermore, synovial IL-6 can be increased up to 27-fold in PJI cases.5 In one study, synovial IL-6 levels >2100 pg/mL had sensitivity of 62.5% and specificity of 85.7% in PJI diagnosis26; in another study, an IL-6 threshold of 4270 pg/mL had sensitivity of 87.1%, specificity of 100%, and accuracy of 94.6%.31
C-Reactive Protein
CRP is an acute-phase reactant. Blood levels increase in response to aseptic inflammatory processes and systemic infection.33 CRP plays an important role in host defense by activating complement and helping mediate phagocytosis.33,34 Although serum CRP levels have been used in diagnosing PJIs,6 they can yield false-negative results.35,36 Therefore, attention turned to synovial CRP levels, which were found to be increased 13-fold in PJI cases.5 It has been shown that synovial CRP levels are significantly higher in infected vs noninfected prosthetic joints34 and had diagnostic accuracy better than that of serum CRP levels in diagnosing PJI.37 One study found that CRP at a threshold of 3.7 mg/L had sensitivity of 84%, specificity of 97.1%, and accuracy of 91.5%,37 whereas another study found that CRP at a threshold of 3.61 mg/L had sensitivity of 87.1%, specificity of 97.7%, and accuracy of 93.3%.31
α-Defensin
α-Defensin, a natural peptide produced and secreted by neutrophils in response to pathogens, has antimicrobial and cytotoxic properties,38-40 signals for the secretion of various cytokines, and acts as a chemoattractant for various immune cells.41 Deirmengian and colleagues6 found that α-defensin was consistently elevated in patients with PJI. α-Defensin is extremely accurate in diagnosing PJI; it has sensitivity ranging from 97% to 100% and specificity ranging from 96% to 100%.6,27,42 Moreover, α-defensin was effective in diagnosing PJI caused by a wide spectrum of organisms, including various low-virulence bacteria and fungi.43
Leukocyte Esterase
Leukocyte esterase is an enzyme produced and secreted by neutrophils at sites of active infection.7,44 Testing for this enzyme is performed with a colorimetric strip and was originally performed for the diagnosis of urinary tract infections.44,45 In a study conducted by Parvizi and colleagues,7 this strip was used to test for leukocyte esterase in synovial fluid samples; a ++ reading was found to have sensitivity of 80.6% and specificity of 100% in diagnosing knee PJI. Similarly, De Vecchi and colleagues45 found sensitivity of 92.6% and specificity of 97%.
Other Synovial Markers
Research has identified numerous molecular biomarkers that may be associated with the pathogenesis of PJI. Although several (eg, cytokines) have demonstrated higher levels in synovial fluid in patients with PJI than in normal controls, only a few have had clinically relevant diagnostic utility.6 Deirmengian and colleagues6 screened 43 synovial fluid biomarkers that potentially could be used in the diagnosis of PJI. Besides the cytokine α-defensin, 4 other biomarkers—lactoferrin, neutrophil gelatinase-associated lipocalcin, neutrophil elastase 2, and bactericidal/permeability-increasing protein—had accuracy of 100%. In addition, 8 cytokines and biomarkers (IL-8, CRP, resistin, thrombospondin, IL-1β, IL-6, IL-10, IL-1α) had area under the curve values higher than 0.9. Studies have also evaluated the diagnostic utility of metabolic products such as lactate, lactate dehydrogenase, and glucose; their accuracy was comparable to that of serum CRP.32
Serum Markers
In addition to the synovial fluid cytokines, several serum inflammatory cytokines have been studied as potential targets in diagnosing infection. Serum IL-6 has had excellent diagnostic accuracy46 and, when combined with CRP, could increase sensitivity in diagnosing PJI; such a combination (vs either test alone) could be useful in screening patients.47,48 Biomarkers such as tumor necrosis factor α and procalcitonin are considered very specific for PJI and may be useful in confirmatory testing.48 Evidence also suggests that toll-like receptor 2 proteins are elevated in the serum of patients with PJI and therefore are a potential diagnostic tool.49
Limitations of Synovial Cytokines
The literature suggests that some synovial fluid cytokines have promise.6 However, the best biomarker or combination of biomarkers is yet to be determined. Results have been consistent with α-defensin and other cytokines but mixed with IL-6 and still others32,42,50 (Table 2).
Information on the utility of synovial biomarkers in detecting persistent infection is limited. Frangiamore and colleagues50 found that IL-1 and IL-6 levels decreased between the stages of 2-stage revision. Unfortunately, none of the synovial fluid cytokines investigated (IL-1, IL-2, IL-6, IL-8, Il-10, interferon γ, granulocyte macrophage-colony stimulating factor, tumor necrosis factor α, IL-12p70) satisfactorily detected resolution of infection in the setting of prior treatment for PJI. Although cytokines are expected to be elevated in the presence of infection, the internal milieu at the time of stage 2 of the revision makes diagnosis of infection difficult. In addition, presence of spacer particles and recent surgery may activate immune pathways and yield false-positive results. Furthermore, antibiotic cement spacers may suppress the microorganisms to very low levels and yield false-negative results even if these organisms remain virulent.19
Even though the synovial molecular markers can detect the presence of infection, they are unable to identify pathogens. As identifying the pathogen is important in the treatment of PJI, there has been interest in using polymerase chain reaction (PCR) techniques.51 These tests may also provide specific information about the pathogen, such as its antibiotic sensitivity. A recently developed technology, the Ibis T5000 Universal Biosensor (Ibis Biosciences), uses novel pan-domain primers in a series of PCRs. This biosensor is useful in diagnosing infections when cultures are negative and appears to be more accurate than conventional PCR.13 As reported by Jacovides and colleagues,13 this novel PCR technique identified an organism in about 88% of presumed cases of aseptic revision.
Conclusion
PJI poses an extreme challenge to the healthcare system. Given the morbidity associated with improper management of PJI, accurate diagnosis is of paramount importance. Given the limitations of current tests, synovial fluid cytokines hold promise in the diagnosis of PJIs. However, these cytokines are expensive, and their clinical utility in PJI management is not well established. More research is needed before guidelines for synovial fluid cytokines and biomarkers can replace or be incorporated into guidelines for the treatment of PJIs.
Take-Home Points
- In cases of failed TJA, it is important to differentiate between septic and aseptic etiologies.
- Chronic and low-grade infections are challenging for orthopedic surgeons, as the symptoms often overlap with aseptic etiologies.
- Verification of infection eradication before beginning the second-stage reimplantation surgery is extremely important, but pre- and intraoperative findings can be unreliable.
- Synovial fluid cytokines have been shown to accurately diagnose PJIs.
- Synovial fluid cytokines may help surgeons differentiate between septic and aseptic cases of failed TJA.
Total joint arthroplasty (TJA) is an effective procedure that has been extensively used to relieve pain and improve quality of life in patients with various forms of joint disease. Although advances in technology and surgical technique have improved the success of TJA, periprosthetic joint infection (PJI) remains a serious complication. In the United States, it is estimated that PJI is the most common reason for total knee arthroplasty failure and the third most common reason for total hip arthroplasty revision.1 Although the incidence of PJI is 1% to 2%, the dramatic increase in TJA volume is expected to be accompanied by a similar rise in the number of infected TJAs; that number is expected to exceed 60,000 in the United States by 2020.2 Moreover, management of PJI is expensive and imposes a heavy burden on the healthcare system, with costs expected to hit $20 billion by 2020 in the US.2 Therefore, treating asepsis cases as infections imposes a heavy burden on the healthcare system and may result in excessive morbidity.3 At the same time, inadequate management of a PJI may result in recurrences that require infection treatment with morbid procedures, such as arthrodesis or amputation. Accurate diagnosis of PJI is of paramount importance in preventing potential implications of a misdiagnosed case. Unfortunately, the PJI diagnosis is extremely challenging, and the available diagnostic tests are often unreliable.4 Thus, research has recently focused on use of several synovial fluid cytokines in the detection of PJI.5-7 In this article, we provide an overview of the synovial biomarkers being used to diagnose PJI.
Diagnosis of Periprosthetic Joint Infection
Differentiating between septic and aseptic failed TJA is important, as the treatment options differ considerably. PJI can be broadly classified as acute or early postoperative (<6 weeks), late chronic (indolent onset), and acute-on-chronic (acute onset in well-functioning prosthesis, secondary to hematogenous spread).8 The acute and acute-on-chronic presentations are often associated with obvious signs of infection.9 However, chronic and low-grade infections pose a challenge to modern orthopedic practice, as the symptoms often overlap with that of aseptic causes of TJA failure.10 As a result, the International Consensus Group on Periprosthetic Joint Infection developed complex criteria using the Musculoskeletal Infection Society definition of PJI and involving a battery of tests for PJI diagnosis.11 According to these criteria, PJI is diagnosed when 1 of the 2 major criteria or 3 of the 5 minor criteria are met (Table 1).
Although these criteria constitute the most agreed on and widely used standard for PJI diagnosis, the definition is complex and often incomplete until surgical intervention. An ideal diagnostic test would aid in managing a PJI and provide results before a treatment decision is made. Many revision surgeries are being performed with insufficient information about the true diagnosis, and the diagnosis might change during or after surgery. About 10% of the revisions presumed to be aseptic may unexpectedly grow cultures during surgery and thereby satisfy the criteria for PJI after surgery.12 Moreover, with the use of novel methods such as polymerase chain reaction, microorganisms were identified in more than three-fourths of the presumed aseptic revisions.13 The optimal management of such cases is controversial, and it is unclear whether positive cultures should be treated as possible contaminants or true infection.12,14
Verification of Infection Eradication
A 2-stage revision procedure, widely accepted as the standard treatment for PJI, has success rates approaching 94%.15 In this procedure, it is important to verify infection eradication before beginning the second-stage reimplantation. Verification is crucial in avoiding reimplantation of an infected joint.16 After the first stage, patients are usually administered intravenous antibiotics for at least 6 weeks; these antibiotics are then withheld, and systemic inflammatory markers are evaluated for infection eradication. Although reliable criteria have been established for PJI diagnosis, guidelines for detecting eradication of infection are rudimentary. Most surgeons monitor the decrease in serologic markers, such as erythrocyte sedimentation rate and C-reactive protein (CRP) level, to assess the response to treatment. However, noninfectious etiologies may result in continued elevation of these markers.17 Even though aspirations are often performed to diagnose persistent infection before the second-stage procedure, their diagnostic utility may be limited.18 Use of cultures is also limited, as presence of antibiotic-loaded spacers can decrease the sensitivity of culture.19 Inadequate diagnosis often leads to unnecessary continuation of antimicrobial therapy or additional surgical débridement. Nuclear scans often remain positive because of aseptic inflammation related to surgery and are not useful in documenting sepsis arrest.20 Given the limitations of available tests, novel strategies for identifying the presence of infection at the second stage are being tested.
Synovial Fluid Cytokines
PJI pathogenesis begins with colonization of the implant surfaces with microorganisms and subsequent formation of biofilms.21 The human immune system is activated by the microbial products, cell wall components, and various biofilm proteins. Immune cells are recruited to the site, where they secrete a myriad of inflammatory biomarkers, such as cytokines, which promote further recruitment of inflammatory cells and aid in the eradication of pathogens.9 These inflammatory cytokines and cells are involved in aseptic inflammatory joint conditions, such as rheumatoid arthritis22,23; however, some are specifically involved in immune pathways combating pathogens.24 This action is the basis for increasing interest in using various synovial fluid cytokines and other biomarkers in the diagnosis of PJI. Here we describe some of the commonly studied cytokines.
Interleukin 1β
Interleukin 1β (IL-1β) is a major proinflammatory cytokine that is synthesized by multiple cells, including macrophages and monocytes.25 IL-1β is produced in response to microorganisms, other cytokines, antigen-presenting cells, and immune complexes; stimulates production of acute-phase proteins by the liver; and is an important pyrogen.25 Deirmengian and colleagues5 found that synovial IL-1β increased 258-fold in patients with a PJI. Studies have found that synovial IL-1β has sensitivity ranging from 66.7% to 100% and specificity ranging from 87% to 100%, with 1 study reporting an accuracy of 100%.5,6,26,27
Interleukin 6
Also produced by macrophages and monocytes, interleukin 6 (IL-6) is a potent stimulator of acute-phase proteins.28,29 IL-6 has a role as a chemoattractant and helps with cell differentiation when changing from innate to acquired immunity.30 It is also used as an aid in diagnosing PJI; it has sensitivity ranging from 62% to 100% and specificity ranging from 85% to 100%.5,6,26,31,32 Synovial IL-6 measurements were more accurate than serum IL-6 measurements.26 Furthermore, synovial IL-6 can be increased up to 27-fold in PJI cases.5 In one study, synovial IL-6 levels >2100 pg/mL had sensitivity of 62.5% and specificity of 85.7% in PJI diagnosis26; in another study, an IL-6 threshold of 4270 pg/mL had sensitivity of 87.1%, specificity of 100%, and accuracy of 94.6%.31
C-Reactive Protein
CRP is an acute-phase reactant. Blood levels increase in response to aseptic inflammatory processes and systemic infection.33 CRP plays an important role in host defense by activating complement and helping mediate phagocytosis.33,34 Although serum CRP levels have been used in diagnosing PJIs,6 they can yield false-negative results.35,36 Therefore, attention turned to synovial CRP levels, which were found to be increased 13-fold in PJI cases.5 It has been shown that synovial CRP levels are significantly higher in infected vs noninfected prosthetic joints34 and had diagnostic accuracy better than that of serum CRP levels in diagnosing PJI.37 One study found that CRP at a threshold of 3.7 mg/L had sensitivity of 84%, specificity of 97.1%, and accuracy of 91.5%,37 whereas another study found that CRP at a threshold of 3.61 mg/L had sensitivity of 87.1%, specificity of 97.7%, and accuracy of 93.3%.31
α-Defensin
α-Defensin, a natural peptide produced and secreted by neutrophils in response to pathogens, has antimicrobial and cytotoxic properties,38-40 signals for the secretion of various cytokines, and acts as a chemoattractant for various immune cells.41 Deirmengian and colleagues6 found that α-defensin was consistently elevated in patients with PJI. α-Defensin is extremely accurate in diagnosing PJI; it has sensitivity ranging from 97% to 100% and specificity ranging from 96% to 100%.6,27,42 Moreover, α-defensin was effective in diagnosing PJI caused by a wide spectrum of organisms, including various low-virulence bacteria and fungi.43
Leukocyte Esterase
Leukocyte esterase is an enzyme produced and secreted by neutrophils at sites of active infection.7,44 Testing for this enzyme is performed with a colorimetric strip and was originally performed for the diagnosis of urinary tract infections.44,45 In a study conducted by Parvizi and colleagues,7 this strip was used to test for leukocyte esterase in synovial fluid samples; a ++ reading was found to have sensitivity of 80.6% and specificity of 100% in diagnosing knee PJI. Similarly, De Vecchi and colleagues45 found sensitivity of 92.6% and specificity of 97%.
Other Synovial Markers
Research has identified numerous molecular biomarkers that may be associated with the pathogenesis of PJI. Although several (eg, cytokines) have demonstrated higher levels in synovial fluid in patients with PJI than in normal controls, only a few have had clinically relevant diagnostic utility.6 Deirmengian and colleagues6 screened 43 synovial fluid biomarkers that potentially could be used in the diagnosis of PJI. Besides the cytokine α-defensin, 4 other biomarkers—lactoferrin, neutrophil gelatinase-associated lipocalcin, neutrophil elastase 2, and bactericidal/permeability-increasing protein—had accuracy of 100%. In addition, 8 cytokines and biomarkers (IL-8, CRP, resistin, thrombospondin, IL-1β, IL-6, IL-10, IL-1α) had area under the curve values higher than 0.9. Studies have also evaluated the diagnostic utility of metabolic products such as lactate, lactate dehydrogenase, and glucose; their accuracy was comparable to that of serum CRP.32
Serum Markers
In addition to the synovial fluid cytokines, several serum inflammatory cytokines have been studied as potential targets in diagnosing infection. Serum IL-6 has had excellent diagnostic accuracy46 and, when combined with CRP, could increase sensitivity in diagnosing PJI; such a combination (vs either test alone) could be useful in screening patients.47,48 Biomarkers such as tumor necrosis factor α and procalcitonin are considered very specific for PJI and may be useful in confirmatory testing.48 Evidence also suggests that toll-like receptor 2 proteins are elevated in the serum of patients with PJI and therefore are a potential diagnostic tool.49
Limitations of Synovial Cytokines
The literature suggests that some synovial fluid cytokines have promise.6 However, the best biomarker or combination of biomarkers is yet to be determined. Results have been consistent with α-defensin and other cytokines but mixed with IL-6 and still others32,42,50 (Table 2).
Information on the utility of synovial biomarkers in detecting persistent infection is limited. Frangiamore and colleagues50 found that IL-1 and IL-6 levels decreased between the stages of 2-stage revision. Unfortunately, none of the synovial fluid cytokines investigated (IL-1, IL-2, IL-6, IL-8, Il-10, interferon γ, granulocyte macrophage-colony stimulating factor, tumor necrosis factor α, IL-12p70) satisfactorily detected resolution of infection in the setting of prior treatment for PJI. Although cytokines are expected to be elevated in the presence of infection, the internal milieu at the time of stage 2 of the revision makes diagnosis of infection difficult. In addition, presence of spacer particles and recent surgery may activate immune pathways and yield false-positive results. Furthermore, antibiotic cement spacers may suppress the microorganisms to very low levels and yield false-negative results even if these organisms remain virulent.19
Even though the synovial molecular markers can detect the presence of infection, they are unable to identify pathogens. As identifying the pathogen is important in the treatment of PJI, there has been interest in using polymerase chain reaction (PCR) techniques.51 These tests may also provide specific information about the pathogen, such as its antibiotic sensitivity. A recently developed technology, the Ibis T5000 Universal Biosensor (Ibis Biosciences), uses novel pan-domain primers in a series of PCRs. This biosensor is useful in diagnosing infections when cultures are negative and appears to be more accurate than conventional PCR.13 As reported by Jacovides and colleagues,13 this novel PCR technique identified an organism in about 88% of presumed cases of aseptic revision.
Conclusion
PJI poses an extreme challenge to the healthcare system. Given the morbidity associated with improper management of PJI, accurate diagnosis is of paramount importance. Given the limitations of current tests, synovial fluid cytokines hold promise in the diagnosis of PJIs. However, these cytokines are expensive, and their clinical utility in PJI management is not well established. More research is needed before guidelines for synovial fluid cytokines and biomarkers can replace or be incorporated into guidelines for the treatment of PJIs.
1 Parvizi J, Adeli B, Zmistowski B, Restrepo C, Greenwald AS. Management of periprosthetic joint infection: the current knowledge: AAOS exhibit selection. J Bone Joint Surg Am. 2012;94(14):e104.
2. Kurtz SM, Lau E, Watson H, Schmier JK, Parvizi J. Economic burden of periprosthetic joint infection in the United States. J Arthroplasty. 2012;27(8 suppl):61-65.e1.
3. Sierra RJ, Trousdale RT, Pagnano MW. Above-the-knee amputation after a total knee replacement: prevalence, etiology, and functional outcome. J Bone Joint Surg Am. 2003;85(6):1000-1004.
4. Bauer TW, Parvizi J, Kobayashi N, Krebs V. Diagnosis of periprosthetic infection. J Bone Joint Surg Am. 2006;88(4):869-882.
5. Deirmengian C, Hallab N, Tarabishy A, et al. Synovial fluid biomarkers for periprosthetic infection. Clin Orthop Relat Res. 2010;468(8):2017-2023.
6. Deirmengian C, Kardos K, Kilmartin P, Cameron A, Schiller K, Parvizi J. Diagnosing periprosthetic joint infection: has the era of the biomarker arrived? Clin Orthop Relat Res. 2014;472(11):3254-3262.
7. Parvizi J, Jacovides C, Antoci V, Ghanem E. Diagnosis of periprosthetic joint infection: the utility of a simple yet unappreciated enzyme. J Bone Joint Surg Am. 2011;93(24):2242-2248.
8. Kuiper JW, Willink RT, Moojen DJF, van den Bekerom MP, Colen S. Treatment of acute periprosthetic infections with prosthesis retention: review of current concepts. World J Orthop. 2014;5(5):667-676.
9. Zimmerli W, Trampuz A, Ochsner PE. Prosthetic-joint infections. N Engl J Med. 2004;351(16):1645-1654.
10. Osmon DR, Berbari EF, Berendt AR, et al. Diagnosis and management of prosthetic joint infection: clinical practice guidelines by the Infectious Diseases Society of America. Clin Infect Dis. 2013;56(1):e1-e25.
11. Parvizi J, Gehrke T; International Consensus Group on Periprosthetic Joint Infection. Definition of periprosthetic joint infection. J Arthroplasty. 2014;29(7):1331.
12. Saleh A, Guirguis A, Klika AK, Johnson L, Higuera CA, Barsoum WK. Unexpected positive intraoperative cultures in aseptic revision arthroplasty. J Arthroplasty. 2014;29(11):2181-2186.
13. Jacovides CL, Kreft R, Adeli B, Hozack B, Ehrlich GD, Parvizi J. Successful identification of pathogens by polymerase chain reaction (PCR)–based electron spray ionization time-of-flight mass spectrometry (ESI-TOF-MS) in culture-negative periprosthetic joint infection. J Bone Joint Surg Am. 2012;94(24):2247-2254.
14. Barrack RL, Aggarwal A, Burnett RS, et al. The fate of the unexpected positive intraoperative cultures after revision total knee arthroplasty. J Arthroplasty. 2007;22(6 suppl 2):94-99.
15. Macheras GA, Koutsostathis SD, Kateros K, Papadakis S, Anastasopoulos P. A two stage re-implantation protocol for the treatment of deep periprosthetic hip infection. Mid to long-term results. Hip Int. 2012;22(suppl 8):S54-S61.
16. George J, Kwiecien G, Klika AK, et al. Are frozen sections and MSIS criteria reliable at the time of reimplantation of two-stage revision arthroplasty? Clin Orthop Relat Res. 2016;474(7):1619-1626.
17. Kusuma SK, Ward J, Jacofsky M, Sporer SM, Della Valle CJ. What is the role of serological testing between stages of two-stage reconstruction of the infected prosthetic knee? Clin Orthop Relat Res. 2011;469(4):1002-1008.
18. Lonner JH, Siliski JM, Della Valle C, DiCesare P, Lotke PA. Role of knee aspiration after resection of the infected total knee arthroplasty. Am J Orthop. 2001;30(4):305-309.
19. Mont MA, Waldman BJ, Hungerford DS. Evaluation of preoperative cultures before second-stage reimplantation of a total knee prosthesis complicated by infection. A comparison-group study. J Bone Joint Surg Am. 2000;82(11):1552-1557.
20. Love C, Marwin SE, Palestro CJ. Nuclear medicine and the infected joint replacement. Semin Nucl Med. 2009;39(1):66-78.
21. Zimmerli W, Moser C. Pathogenesis and treatment concepts of orthopaedic biofilm infections. FEMS Immunol Med Microbiol. 2012;65(2):158-168.
22. Fontana A, Hengartner H, Weber E, Fehr K, Grob PJ, Cohen G. Interleukin 1 activity in the synovial fluid of patients with rheumatoid arthritis. Rheumatol Int. 1982;2(2):49-53.
23. Guerne PA, Zuraw BL, Vaughan JH, Carson DA, Lotz M. Synovium as a source of interleukin 6 in vitro. Contribution to local and systemic manifestations of arthritis. J Clin Invest. 1989;83(2):585-592.
24. Wang G. Human antimicrobial peptides and proteins. Pharmaceuticals (Basel). 2014;7(5):545-594.
25. Stylianou E, Saklatvala J. Interleukin-1. Int J Biochem Cell Biol. 1998;30(10):1075-1079.
26. Gollwitzer H, Dombrowski Y, Prodinger PM, et al. Antimicrobial peptides and proinflammatory cytokines in periprosthetic joint infection. J Bone Joint Surg Am. 2013;95(7):644-651.
27. Deirmengian C, Kardos K, Kilmartin P, Cameron A, Schiller K, Parvizi J. Combined measurement of synovial fluid α-defensin and C-reactive protein levels: highly accurate for diagnosing periprosthetic joint infection. J Bone Joint Surg Am. 2014;96(17):1439-1445.
28. Randau TM, Friedrich MJ, Wimmer MD, et al. Interleukin-6 in serum and in synovial fluid enhances the differentiation between periprosthetic joint infection and aseptic loosening. PLoS One. 2014;9(2):e89045.
29. Heinrich PC, Castell JV, Andus T. Interleukin-6 and the acute phase response. Biochem J. 1990;265(3):621-636.
30. Scheller J, Chalaris A, Schmidt-Arras D, Rose-John S. The pro- and anti-inflammatory properties of the cytokine interleukin-6. Biochim Biophys Acta. 2011;1813(5):878-888.
31. Jacovides CL, Parvizi J, Adeli B, Jung KA. Molecular markers for diagnosis of periprosthetic joint infection. J Arthroplasty. 2011;26(6 suppl):99-103.e1.
32. Lenski M, Scherer MA. Synovial IL-6 as inflammatory marker in periprosthetic joint infections. J Arthroplasty. 2014;29(6):1105-1109.
33. Mortensen RF. C-reactive protein, inflammation, and innate immunity. Immunol Res. 2001;24(2):163-176.
34. Parvizi J, McKenzie JC, Cashman JP. Diagnosis of periprosthetic joint infection using synovial C-reactive protein. J Arthroplasty. 2012;27(8 suppl):12-16.
35. Ghanem E, Antoci V, Pulido L, Joshi A, Hozack W, Parvizi J. The use of receiver operating characteristics analysis in determining erythrocyte sedimentation rate and C-reactive protein levels in diagnosing periprosthetic infection prior to revision total hip arthroplasty. Int J Infect Dis. 2009;13(6):e444-e449.
36. Johnson AJ, Zywiel MG, Stroh A, Marker DR, Mont MA. Serological markers can lead to false negative diagnoses of periprosthetic infections following total knee arthroplasty. Int Orthop. 2011;35(11):1621-1626.
37. Parvizi J, Jacovides C, Adeli B, Jung KA, Hozack WJ. Mark B. Coventry award: synovial C-reactive protein: a prospective evaluation of a molecular marker for periprosthetic knee joint infection. Clin Orthop Relat Res. 2012;470(1):54-60.
38. Lehrer RI, Lichtenstein AK, Ganz T. Defensins: antimicrobial and cytotoxic peptides of mammalian cells. Annu Rev Immunol. 1993;11:105-128.
39. Ganz T, Selsted ME, Szklarek D, et al. Defensins. Natural peptide antibiotics of human neutrophils. J Clin Invest. 1985;76(4):1427-1435.
40. Chalifour A, Jeannin P, Gauchat JF, et al. Direct bacterial protein PAMP recognition by human NK cells involves TLRs and triggers alpha-defensin production. Blood. 2004;104(6):1778-1783.
41. Ulm H, Wilmes M, Shai Y, Sahl HG. Antimicrobial host defensins—specific antibiotic activities and innate defense modulation. Front Immunol. 2012;3:249.
42. Bingham J, Clarke H, Spangehl M, Schwartz A, Beauchamp C, Goldberg B. The alpha defensin-1 biomarker assay can be used to evaluate the potentially infected total joint arthroplasty. Clin Orthop Relat Res. 2014;472(12):4006-4009.
43. Deirmengian C, Kardos K, Kilmartin P, Gulati S, Citrano P, Booth RE. The alpha-defensin test for periprosthetic joint infection responds to a wide spectrum of organisms. Clin Orthop Relat Res. 2015;473(7):2229-2235.
44. Tischler EH, Cavanaugh PK, Parvizi J. Leukocyte esterase strip test: matched for Musculoskeletal Infection Society criteria. J Bone Joint Surg Am. 2014;96(22):1917-1920.
45. De Vecchi E, Villa F, Bortolin M, et al. Leucocyte esterase, glucose and C-reactive protein in the diagnosis of prosthetic joint infections: a prospective study. Clin Microbiol Infect. 2016;22(6):555-560.
46. Di Cesare PE, Chang E, Preston CF, Liu C. Serum interleukin-6 as a marker of periprosthetic infection following total hip and knee arthroplasty. J Bone Joint Surg Am. 2005;87(9):1921-1927.
47. Ettinger M, Calliess T, Kielstein JT, et al. Circulating biomarkers for discrimination between aseptic joint failure, low-grade infection, and high-grade septic failure. Clin Infect Dis. 2015;61(3):332-341.
48. Bottner F, Wegner A, Winkelmann W, Becker K, Erren M, Götze C. Interleukin-6, procalcitonin and TNF-alpha: markers of peri-prosthetic infection following total joint replacement. J Bone Joint Surg Br. 2007;89(1):94-99.
49. Galliera E, Drago L, Vassena C, et al. Toll-like receptor 2 in serum: a potential diagnostic marker of prosthetic joint infection? J Clin Microbiol. 2014;52(2):620-623.
50. Frangiamore SJ, Siqueira MB, Saleh A, Daly T, Higuera CA, Barsoum WK. Synovial cytokines and the MSIS criteria are not useful for determining infection resolution after periprosthetic joint infection explantation. Clin Orthop Relat Res. 2016;474(7):1630-1639.
51. Rasouli MR, Harandi AA, Adeli B, Purtill JJ, Parvizi J. Revision total knee arthroplasty: infection should be ruled out in all cases. J Arthroplasty. 2012;27(6):1239-1243.e1-e2.
52. Omar M, Ettinger M, Reichling M, et al. Synovial C-reactive protein as a marker for chronic periprosthetic infection in total hip arthroplasty. Bone Joint J. 2015;97(2):173-176.
53. Tetreault MW, Wetters NG, Moric M, Gross CE, Della Valle CJ. Is synovial C-reactive protein a useful marker for periprosthetic joint infection? Clin Orthop Relat Res. 2014;472(12):3997-4003.
54. Omar M, Ettinger M, Reichling M, et al. Preliminary results of a new test for rapid diagnosis of septic arthritis with use of leukocyte esterase and glucose reagent strips. J Bone Joint Surg Am. 2014;96(24):2032-2037.
1 Parvizi J, Adeli B, Zmistowski B, Restrepo C, Greenwald AS. Management of periprosthetic joint infection: the current knowledge: AAOS exhibit selection. J Bone Joint Surg Am. 2012;94(14):e104.
2. Kurtz SM, Lau E, Watson H, Schmier JK, Parvizi J. Economic burden of periprosthetic joint infection in the United States. J Arthroplasty. 2012;27(8 suppl):61-65.e1.
3. Sierra RJ, Trousdale RT, Pagnano MW. Above-the-knee amputation after a total knee replacement: prevalence, etiology, and functional outcome. J Bone Joint Surg Am. 2003;85(6):1000-1004.
4. Bauer TW, Parvizi J, Kobayashi N, Krebs V. Diagnosis of periprosthetic infection. J Bone Joint Surg Am. 2006;88(4):869-882.
5. Deirmengian C, Hallab N, Tarabishy A, et al. Synovial fluid biomarkers for periprosthetic infection. Clin Orthop Relat Res. 2010;468(8):2017-2023.
6. Deirmengian C, Kardos K, Kilmartin P, Cameron A, Schiller K, Parvizi J. Diagnosing periprosthetic joint infection: has the era of the biomarker arrived? Clin Orthop Relat Res. 2014;472(11):3254-3262.
7. Parvizi J, Jacovides C, Antoci V, Ghanem E. Diagnosis of periprosthetic joint infection: the utility of a simple yet unappreciated enzyme. J Bone Joint Surg Am. 2011;93(24):2242-2248.
8. Kuiper JW, Willink RT, Moojen DJF, van den Bekerom MP, Colen S. Treatment of acute periprosthetic infections with prosthesis retention: review of current concepts. World J Orthop. 2014;5(5):667-676.
9. Zimmerli W, Trampuz A, Ochsner PE. Prosthetic-joint infections. N Engl J Med. 2004;351(16):1645-1654.
10. Osmon DR, Berbari EF, Berendt AR, et al. Diagnosis and management of prosthetic joint infection: clinical practice guidelines by the Infectious Diseases Society of America. Clin Infect Dis. 2013;56(1):e1-e25.
11. Parvizi J, Gehrke T; International Consensus Group on Periprosthetic Joint Infection. Definition of periprosthetic joint infection. J Arthroplasty. 2014;29(7):1331.
12. Saleh A, Guirguis A, Klika AK, Johnson L, Higuera CA, Barsoum WK. Unexpected positive intraoperative cultures in aseptic revision arthroplasty. J Arthroplasty. 2014;29(11):2181-2186.
13. Jacovides CL, Kreft R, Adeli B, Hozack B, Ehrlich GD, Parvizi J. Successful identification of pathogens by polymerase chain reaction (PCR)–based electron spray ionization time-of-flight mass spectrometry (ESI-TOF-MS) in culture-negative periprosthetic joint infection. J Bone Joint Surg Am. 2012;94(24):2247-2254.
14. Barrack RL, Aggarwal A, Burnett RS, et al. The fate of the unexpected positive intraoperative cultures after revision total knee arthroplasty. J Arthroplasty. 2007;22(6 suppl 2):94-99.
15. Macheras GA, Koutsostathis SD, Kateros K, Papadakis S, Anastasopoulos P. A two stage re-implantation protocol for the treatment of deep periprosthetic hip infection. Mid to long-term results. Hip Int. 2012;22(suppl 8):S54-S61.
16. George J, Kwiecien G, Klika AK, et al. Are frozen sections and MSIS criteria reliable at the time of reimplantation of two-stage revision arthroplasty? Clin Orthop Relat Res. 2016;474(7):1619-1626.
17. Kusuma SK, Ward J, Jacofsky M, Sporer SM, Della Valle CJ. What is the role of serological testing between stages of two-stage reconstruction of the infected prosthetic knee? Clin Orthop Relat Res. 2011;469(4):1002-1008.
18. Lonner JH, Siliski JM, Della Valle C, DiCesare P, Lotke PA. Role of knee aspiration after resection of the infected total knee arthroplasty. Am J Orthop. 2001;30(4):305-309.
19. Mont MA, Waldman BJ, Hungerford DS. Evaluation of preoperative cultures before second-stage reimplantation of a total knee prosthesis complicated by infection. A comparison-group study. J Bone Joint Surg Am. 2000;82(11):1552-1557.
20. Love C, Marwin SE, Palestro CJ. Nuclear medicine and the infected joint replacement. Semin Nucl Med. 2009;39(1):66-78.
21. Zimmerli W, Moser C. Pathogenesis and treatment concepts of orthopaedic biofilm infections. FEMS Immunol Med Microbiol. 2012;65(2):158-168.
22. Fontana A, Hengartner H, Weber E, Fehr K, Grob PJ, Cohen G. Interleukin 1 activity in the synovial fluid of patients with rheumatoid arthritis. Rheumatol Int. 1982;2(2):49-53.
23. Guerne PA, Zuraw BL, Vaughan JH, Carson DA, Lotz M. Synovium as a source of interleukin 6 in vitro. Contribution to local and systemic manifestations of arthritis. J Clin Invest. 1989;83(2):585-592.
24. Wang G. Human antimicrobial peptides and proteins. Pharmaceuticals (Basel). 2014;7(5):545-594.
25. Stylianou E, Saklatvala J. Interleukin-1. Int J Biochem Cell Biol. 1998;30(10):1075-1079.
26. Gollwitzer H, Dombrowski Y, Prodinger PM, et al. Antimicrobial peptides and proinflammatory cytokines in periprosthetic joint infection. J Bone Joint Surg Am. 2013;95(7):644-651.
27. Deirmengian C, Kardos K, Kilmartin P, Cameron A, Schiller K, Parvizi J. Combined measurement of synovial fluid α-defensin and C-reactive protein levels: highly accurate for diagnosing periprosthetic joint infection. J Bone Joint Surg Am. 2014;96(17):1439-1445.
28. Randau TM, Friedrich MJ, Wimmer MD, et al. Interleukin-6 in serum and in synovial fluid enhances the differentiation between periprosthetic joint infection and aseptic loosening. PLoS One. 2014;9(2):e89045.
29. Heinrich PC, Castell JV, Andus T. Interleukin-6 and the acute phase response. Biochem J. 1990;265(3):621-636.
30. Scheller J, Chalaris A, Schmidt-Arras D, Rose-John S. The pro- and anti-inflammatory properties of the cytokine interleukin-6. Biochim Biophys Acta. 2011;1813(5):878-888.
31. Jacovides CL, Parvizi J, Adeli B, Jung KA. Molecular markers for diagnosis of periprosthetic joint infection. J Arthroplasty. 2011;26(6 suppl):99-103.e1.
32. Lenski M, Scherer MA. Synovial IL-6 as inflammatory marker in periprosthetic joint infections. J Arthroplasty. 2014;29(6):1105-1109.
33. Mortensen RF. C-reactive protein, inflammation, and innate immunity. Immunol Res. 2001;24(2):163-176.
34. Parvizi J, McKenzie JC, Cashman JP. Diagnosis of periprosthetic joint infection using synovial C-reactive protein. J Arthroplasty. 2012;27(8 suppl):12-16.
35. Ghanem E, Antoci V, Pulido L, Joshi A, Hozack W, Parvizi J. The use of receiver operating characteristics analysis in determining erythrocyte sedimentation rate and C-reactive protein levels in diagnosing periprosthetic infection prior to revision total hip arthroplasty. Int J Infect Dis. 2009;13(6):e444-e449.
36. Johnson AJ, Zywiel MG, Stroh A, Marker DR, Mont MA. Serological markers can lead to false negative diagnoses of periprosthetic infections following total knee arthroplasty. Int Orthop. 2011;35(11):1621-1626.
37. Parvizi J, Jacovides C, Adeli B, Jung KA, Hozack WJ. Mark B. Coventry award: synovial C-reactive protein: a prospective evaluation of a molecular marker for periprosthetic knee joint infection. Clin Orthop Relat Res. 2012;470(1):54-60.
38. Lehrer RI, Lichtenstein AK, Ganz T. Defensins: antimicrobial and cytotoxic peptides of mammalian cells. Annu Rev Immunol. 1993;11:105-128.
39. Ganz T, Selsted ME, Szklarek D, et al. Defensins. Natural peptide antibiotics of human neutrophils. J Clin Invest. 1985;76(4):1427-1435.
40. Chalifour A, Jeannin P, Gauchat JF, et al. Direct bacterial protein PAMP recognition by human NK cells involves TLRs and triggers alpha-defensin production. Blood. 2004;104(6):1778-1783.
41. Ulm H, Wilmes M, Shai Y, Sahl HG. Antimicrobial host defensins—specific antibiotic activities and innate defense modulation. Front Immunol. 2012;3:249.
42. Bingham J, Clarke H, Spangehl M, Schwartz A, Beauchamp C, Goldberg B. The alpha defensin-1 biomarker assay can be used to evaluate the potentially infected total joint arthroplasty. Clin Orthop Relat Res. 2014;472(12):4006-4009.
43. Deirmengian C, Kardos K, Kilmartin P, Gulati S, Citrano P, Booth RE. The alpha-defensin test for periprosthetic joint infection responds to a wide spectrum of organisms. Clin Orthop Relat Res. 2015;473(7):2229-2235.
44. Tischler EH, Cavanaugh PK, Parvizi J. Leukocyte esterase strip test: matched for Musculoskeletal Infection Society criteria. J Bone Joint Surg Am. 2014;96(22):1917-1920.
45. De Vecchi E, Villa F, Bortolin M, et al. Leucocyte esterase, glucose and C-reactive protein in the diagnosis of prosthetic joint infections: a prospective study. Clin Microbiol Infect. 2016;22(6):555-560.
46. Di Cesare PE, Chang E, Preston CF, Liu C. Serum interleukin-6 as a marker of periprosthetic infection following total hip and knee arthroplasty. J Bone Joint Surg Am. 2005;87(9):1921-1927.
47. Ettinger M, Calliess T, Kielstein JT, et al. Circulating biomarkers for discrimination between aseptic joint failure, low-grade infection, and high-grade septic failure. Clin Infect Dis. 2015;61(3):332-341.
48. Bottner F, Wegner A, Winkelmann W, Becker K, Erren M, Götze C. Interleukin-6, procalcitonin and TNF-alpha: markers of peri-prosthetic infection following total joint replacement. J Bone Joint Surg Br. 2007;89(1):94-99.
49. Galliera E, Drago L, Vassena C, et al. Toll-like receptor 2 in serum: a potential diagnostic marker of prosthetic joint infection? J Clin Microbiol. 2014;52(2):620-623.
50. Frangiamore SJ, Siqueira MB, Saleh A, Daly T, Higuera CA, Barsoum WK. Synovial cytokines and the MSIS criteria are not useful for determining infection resolution after periprosthetic joint infection explantation. Clin Orthop Relat Res. 2016;474(7):1630-1639.
51. Rasouli MR, Harandi AA, Adeli B, Purtill JJ, Parvizi J. Revision total knee arthroplasty: infection should be ruled out in all cases. J Arthroplasty. 2012;27(6):1239-1243.e1-e2.
52. Omar M, Ettinger M, Reichling M, et al. Synovial C-reactive protein as a marker for chronic periprosthetic infection in total hip arthroplasty. Bone Joint J. 2015;97(2):173-176.
53. Tetreault MW, Wetters NG, Moric M, Gross CE, Della Valle CJ. Is synovial C-reactive protein a useful marker for periprosthetic joint infection? Clin Orthop Relat Res. 2014;472(12):3997-4003.
54. Omar M, Ettinger M, Reichling M, et al. Preliminary results of a new test for rapid diagnosis of septic arthritis with use of leukocyte esterase and glucose reagent strips. J Bone Joint Surg Am. 2014;96(24):2032-2037.
Paraskiing Crash and Knee Dislocation With Multiligament Reconstruction and Iliotibial Band Repair
Take-Home Points
- Reconstruction of a torn ITB is important in restoration of native anatomy and function given its properties in anterolateral stabilization and resistance to varus stress and internal tibial rotation.
- Restoration of posterolateral instability primarily involves reconstructing the FCL, PLT, and popliteofibular ligament.
- For combined PLC injuries, concurrent reconstruction of the cruciate ligaments in one stage is highly recommended.
- Post-surgery, a 6-week non-weight-bearing, limited flexion rehab protocol utilizing a dynamic PCL brace, such as the PCL Rebound brace, is recommended to prevent posterior tibial sag.
- Arthrofibrosis and decreased ROM can be seen following a violent knee injury which requires extensive multiligament reconstruction surgeries, occasionally requiring a secondary surgery for further restoration of knee motion.
Tibiofemoral knee dislocations are uncommon injuries that have devastating complications and potentially result in complex surgeries.1 Knee dislocations (KDs) can be classified with the Schenck system.2 KD-I is a multiligament injury involving the anterior cruciate ligament (ACL) or the posterior cruciate ligament (PCL), and the scale increases in severity/number of ligaments involved, with KD-V being a multiligament injury with periarticular fracture.2
In this article, we report the case of a complex multiligament knee reconstruction performed with a midsubstance iliotibial band (ITB) repair. The patient provided written informed consent for print and electronic publication of this case report.
Case Report
A 27-year-old man presented 12 days after a paraskiing crash in which he collided with a tree at 45 mph and fell 40 feet before hitting snow. Physical examination revealed a large hemarthrosis of the left lower extremity and ecchymosis about the posterolateral aspect of the knee and popliteal fossa. Range of motion (ROM) was limited from 5° of hyperextension to 90° of flexion. Additional motion was deferred secondary to pain. Varus stress testing at 0° and 30° of knee flexion demonstrated significant side-to-side differences. The Lachman test, posterior drawer test, and posterolateral drawer test were all 3+. The dial test was 3 to 4+ compared with the contralateral knee. Valgus stress testing at 0° and 30° of flexion did not reveal any side-to-side laxity. The calf was nontender, and all compartments were soft. The patient reported no neurovascular symptoms and had no neuromotor deficits other than mild common peroneal nerve dysesthesias.
Varus stress radiographs showed increased side-to-side gapping (8 mm) of the lateral compartment of the injured knee. Kneeling posterior stress radiographs, limited because of the patient’s inability to apply full stress on the injured knee secondary to pain, showed a difference of 6 mm in increased posterior translation on the uninjured leg (Figures 1A-1D).
First Surgery
1. PLC Approach. A lateral hockey-stick skin incision was made along the ITB and extended distally between the fibular head and the Gerdy tubercle. The subcutaneous tissue was then dissected, and a posteriorly based flap was developed for preservation of vascular support to the superficial tissues. The ITB and the lateral capsule had completely torn off of the femur, allowing exposure directly into the joint. The long and short heads of the biceps femoris were exposed, with about 50% of the biceps attachment torn. The FCL was torn midsubstance, and the PLT had no remnant attachment left on the femur.
2. ITB and Lateral Capsule Tag Stitched. The torn ends of the ITB were dissected and tag stitches placed in each end. Tag stitches were also placed in the lateral capsule in preparation for a direct repair.
3. Neurolysis. The common peroneal nerve was found encased in a significant amount of scar tissue, and extensive neurolysis was required. Slow, methodical dissection was performed under the partially torn long head of the biceps femoris and was continued through the scar tissue and adhesions. Distally, 5 mm to 7 mm of the peroneus longus fascia was incised as part of the neurolysis in order to prevent nerve irritation or foot drop caused by postoperative swelling.
4. PLC Tunnels. The margin between the lateral gastrocnemius tendon and the soleus muscle was identified by blunt dissection that allowed palpation of the posteromedial aspect of the fibular styloid and the popliteus musculotendinous junction. The underlying biceps bursa was incised in order to locate the midportion of the FCL remnant, which typically is tag-stitched with No. 2 FiberWire to help identify the femoral attachment (this was not done because of the complete tear at the midsubstance of the FCL).
Subperiosteal dissection of the lateral aspect of the fibular head was performed anterior to posterior and distally extended to the champagne-glass drop-off of the fibular head. Continuing the dissection distally beyond this point can endanger the common peroneal nerve. A small sulcus can be palpated where the distal FCL inserts on the fibular head. Posteriorly, a small elevator was used to dissect the soleus muscle off of the posteromedial aspect of the fibular head, where the fibular tunnel would later be created.
A Chandler retractor was placed posterior to the fibular head to protect the neurovascular bundle. With the aid of a collateral ligament aiming device, a guide pin was drilled from the lateral aspect of the fibular head (FCL attachment) to the posteromedial downslope of the fibular styloid (popliteofibular ligament attachment). The entry point of the guide pin was immediately above the champagne- glass drop-off, at the distal insertion site of the FCL, which was described as being 28.4 mm from the styloid tip and 8.2 mm posterior to the anterior margin of the fibular head.3 Care should be taken not to ream the tunnel too proximal, as doing so increases the risk of iatrogenic fracture. A 7-mm reamer was then used to drill the fibular tunnel. To facilitate later passage of the graft, a passing suture was placed through the tunnel, leaving the loop anterolateral.
Next, the starting point for the tibial tunnel was located on the flat spot of the anterolateral tibia distal and medial to the Gerdy tubercle, just lateral to the tibial tubercle. The tibial popliteal sulcus was identified by palpation of the posterolateral tibial plateau to localize the site of the popliteus musculotendinous junction, which is the ideal location of the posterior aperture of the tibial tunnel. This point is 1 cm proximal and 1 cm medial to the posteromedial exit of the fibular tunnel. A Chandler retractor was placed anterior to the lateral gastrocnemius to protect the neurovascular bundle. In the locations described earlier, a cruciate aiming device was used to place a guide pin anterior to posterior. A 9-mm tunnel was overreamed and a passing suture placed, leaving the loop posterior to facilitate graft passage.
The femoral insertions of the FCL and the PLT were then identified. ITB splitting was not necessary, given the complete midsubstance tear of this structure. The FCL attachment was identified 1.4 mm proximal and 3.1 mm posterior to the lateral epicondyle.3 Sharp dissection was performed in this location, proximal to distal, exposing the lateral epicondyle and the small sulcus at the FCL attachment site. A collateral ligament reconstruction aiming sleeve was used to drill a guide pin over the FCL femoral attachment site and out the medial aspect of the distal thigh, about 5 cm proximal and anterior to the adductor tubercle.
The femoral attachment of the PLT was reported located 18.5 mm anterior to the FCL insertion, in the anterior fifth of the popliteal sulcus.3 Although arthrotomy is usually required in order to access the PLT attachment, it was not necessary in this case, given the lateral capsule tear. A guide pin was inserted at the PLT attachment site, parallel to the FCL pin. After proper placement was verified, a 9-mm reamer was used to drill the FCL and PLT tunnels to a depth of 25 mm (socket), and a passing suture was placed into each tunnel to facilitate graft passage.
5. ACL Graft Harvest. The central third of the ipsilateral patellar tendon was harvested for use in the ACL reconstruction. Included were a 10-mm × 20-mm bone plug from the patella and a 10-mm × 25-mm bone plug from the tibial tubercle. The patella defect was then bone-grafted, and the patellar tendon closed side-to-side.
6. Graft Preparation. For the PLC, we used a split Achilles tendon allograft that had two 9-mm × 25-mm bone plugs proximally and were tubularized distally. For the PCL, we used an anterolateral bundle (ALB), which consisted of an Achilles tendon allograft that had an 11-mm × 25-mm bone plug proximally and was tubularized distally, and a posteromedial bundle (PMB), which consisted of a tibialis anterior allograft that was tubularized at both ends. For the ACL, we used a bone–patellar tendon–bone autograft 10 mm in diameter with a 20-mm femoral bone plug and a 25-mm tibial bone plug distally.
7. Arthroscopy. We created standard anterolateral and anteromedial parapatellar portals and performed arthroscopy, including lysis of adhesions. Cartilage and menisci were lesion-free.
8. PCL Femoral Tunnels. The ALB attachment was identified and outlined with a coagulator between the trochlear point and the medial arch point, adjacent to the edge of the articular cartilage. Similarly, the PMB attachment was marked about 8 mm or 9 mm posterior to the edge of the articular cartilage of the medial femoral condyle and slightly posterior to the ALB tunnel.4
In the anterolateral tunnel, an acorn reamer 11 mm in diameter was used to score the entry point of the ALB femoral tunnel. An eyelet pin was then drilled through the reamer anteromedially out the knee. Then a closed socket tunnel was reamed over the eyelet pin to a depth of 25 mm. A passing suture was pulled through the tunnel in preparation for graft passage.
With use of the same technique, a 7-mm reamer was placed against the outline of the PMB attachment site, and an eyelet pin was drilled through this reamer and out the anteromedial aspect of the knee. Again, a 25-mm deep closed socket was reamed. A bone bridge distance of 2 mm was maintained between the 2 femoral PCL bundle tunnels.
9. ACL Femoral Tunnel. The femoral ACL attachment was identified and outlined. An over-the-top guide was used to determine proper placement of the 10-mm low-profile reamer. A guide pin was drilled through the center of the reamer. The reamer was used to create a 25-mm deep closed socket tunnel, and a passing stitch was placed.
10. PCL Tibial Tunnel. With use of a 70° arthroscope for visualization, a posteromedial arthroscopic portal was created, and a shaver and a coagulator were used to identify the tibial PCL attachment, located distally along the PCL facet, until the proximal aspect of the popliteus muscle fibers were visualized. A guide pin was drilled starting at the anteromedial aspect of the tibia, about 6 cm distal to the joint line and centered between the anterior tibial crest and the medial tibial border. The pin exited posteriorly at the center of the PCL tibial attachment along the PCL bundle ridge, which was reported located between the ALB and the PMB on the tibia.5 Pin placement was verified with intraoperative lateral and anteroposterior radiographs. On the lateral radiograph, the pin should be about 6 mm or 7 mm proximal to the champagne-glass drop-off at the PCL facet on the posterior aspect of the tibia. On the anteroposterior radiograph, the pin should be 1 mm to 2 mm distal to the joint line and at the medial aspect of the lateral tibial eminence. A large curette was passed through the posteromedial arthroscopic portal both to retract the posterior tissues away from the reamer and to protect against guide-pin protrusion The guide pin was then overreamed with a 12-mm acorn reamer.
A large smoother was passed proximally up the tibial tunnel and then pulled out the anteromedial portal with a grasper. The smoother was gently cycled to smooth the intra-articular tibial tunnel aperture to remove any bony spicules that could interfere with graft passage. The smoother was then pulled back into the joint, passed out the anterolateral arthroscopic portal, and secured with a small clamp.4
11. ACL Tibial Tunnel. The ACL tibial attachment site was identified and cleaned of soft tissue. A guide pin was placed and then overreamed with a 10-mm acorn reamer.
12. PCL Femoral Fixation. The PMB graft was passed into its tunnel and secured with a 7-mm × 23-mm titanium screw. Next, the ALB was secured to the femur with a 7-mm × 20-mm titanium screw. The smoother was used to pull both grafts down through the tibial tunnel.
13. ACL Femoral Fixation. A 7-mm × 20-mm titanium screw was then used to fix the ACL autograft inside the femur. Traction was applied to the 3 cruciate grafts. There was no sign of impingement.
14. PLC Femoral Fixation. The FCL and the popliteus bone plugs were passed into their respective femoral sockets and secured with 7-mm × 20-mm titanium screws.
15. Lateral Capsule Femoral Anchors. Two suture anchors were placed into the femur, and the sutures were passed through the femoral portion of the lateral capsule for later repair.
16. PCL Tibial Fixation. Both grafts were fixed with a fully threaded bicortical 6.5-mm × 40-mm cannulated cancellous screw and an 18-mm spiked washer. The ALB was fixed first, with the knee flexed to 90°, traction on the graft, and the tibia in neutral rotation. Restoration of the normal tibiofemoral step-off was verified. The PMB was then fixed with the knee in full extension. A posterior drawer test was performed to verify restoration of stability.
17. PLC Fibula Fixation. The PLT graft was passed down the popliteal hiatus, and the FCL graft was passed under the remnant of the biceps bursa on the fibular head and then through the fibular head, anterolateral to posteromedial. The FCL graft was fixed in the fibular tunnel with the knee in 20° of flexion, a slight valgus reduction force, the tibia in neutral rotation, and traction on the graft. A 7-mm × 23-mm bioabsorbable screw was used.
18. Lateral Capsular Repair. The lateral capsule was directly repaired with the previously placed sutures. The sutures were tied with the knee in 20° of flexion.
19. PLC Tibial Fixation. The grafts were passed together, posterior to anterior, through the tibial tunnel. The knee was cycled several times through complete flexion/extension ROM. A 9-mm × 23-mm bioabsorbable screw was then used to fix the grafts to the tibia. During this fixation, the knee was kept in 60° of flexion and neutral rotation while traction was being applied to the distal end of both grafts.
20. ACL Tibial Fixation. A 9-mm × 20-mm titanium screw was used to fix the ACL graft with the knee in full extension. The graft was then viewed intra-articularly to confirm there was no impingement. The Lachman, posterior drawer, posterolateral drawer, dial, and varus stress tests were performed to ensure restoration of stability.
21. ITB Repair. A portion of the remaining Achilles tendon allograft was used to perform ITB reconstruction (reconstitution of the gaped portion of the ITB). Orthocord (DePuy Synthes) and Vicryl (Ethicon) sutures were used for this reconstruction. Knee stability was deemed restored, and the incisions were closed in standard layered fashion.
First Surgery: Postoperative Management
The patient remained non-weight-bearing the first 6 weeks after surgery, with prone knee flexion limited (0°-90°) the first 2 weeks. In addition, a PCL Jack brace (Albrecht) was placed 1 week after surgery and was to be worn at all times to decrease stress on the PCL grafts.
As ROM was not progressing as expected, the patient was instructed to use a continuous passive motion (CPM) machine 2 hours 3 times a day. About 4 weeks after surgery, with ROM still not progressing, the frequency of use of this machine was increased.
Despite continued physical therapy, use of the CPM machine, and pain management, ROM was limited (11°-90° of flexion) 5.5 months after left knee multiligament reconstruction. However, stress radiographs showed excellent stability. Varus stress radiographs showed a side-to-side difference of 0.3 mm less on the left (injured) knee, and kneeling PCL stress radiographs showed a side-to-side difference of 1.3 mm more on the left knee (Figures 3A-3D).
Second Surgery and Postoperative Management
As gentle manipulation under anesthesia was unsuccessful, the patient underwent knee arthroscopy, including 4-compartment lysis of adhesions, arthroscopically assisted posteromedial capsular release, and post-débridement manipulation under anesthesia. During manipulation, full extension and knee flexion up to 135° were achieved. ACL, PCL, and popliteus grafts were visualized and confirmed to be intact.
After this second surgery, the patient was to resume physical therapy and begin weight- bearing as tolerated. Active ROM was prioritized in an attempt to reach full ROM. In addition, a CPM machine was to be used from 0° to 135° of knee flexion 4 hours 3 times a day for 6 weeks.
Two weeks after surgery, the patient had continued pain, and extracapsular swelling in the left knee. However, ROM (0°-115° of flexion) was improved relative to before surgery (11°-90° of flexion), though it remained below the range on the contralateral side. Of note, the patient reported having a flexion contracture (~10°) in the immediate postoperative period. He had woken up with it after sleeping with the CPM machine the night before. The contracture delayed his physical therapy for several hours and resulted in a redesign of his therapy protocol to emphasize full, active knee extension and patellar mobilization, as well as discontinuation of use of the CPM machine. Corticosteroids were initiated to help with the extracapsular swelling, and the new therapy regimen brought adequate progress in ROM. Four months after the second surgery, the patient had full extension and 135° of flexion and was transitioned into wearing the PCL Rebound brace.
Discussion
This case was unique because of the midsubstance ITB tear and simultaneous multiligament injury caused by a KD-IIIL, a KD involving the ACL, the PCL, and the PLC with the medial side intact. There is limited research on ITB repair generally, with or without KD involvement. In a retrospective review of acute knee trauma cases, ITB pathologies were seen on 45% of reviewed MRI scans, and only 3% of the injuries were grade III; in addition, only 9 (5%) of the 200 cases involved both ITB and multiligament (ACL, PCL) knee injuries.6
After our patient’s ACL, PCL, and PLC were reconstructed, a fan piece of the Achilles tendon allograft from the PLC reconstruction was used to repair the ITB. The graft was used to reconstitute the torn gapped portion of the band in multiple locations, and this repair helped restore stability. The literature has reported numerous surgical uses for a portion of the ITB but few studies on repairing this anatomical structure. Preservation of the ITB is important to restoration of native anatomy and function. The ITB helps with anterolateral stabilization of the knee and with resistance of varus stress and internal tibial rotation.
The PLC reconstruction used in this case has been biomechanically validated as restoring the knee to near native stability through anatomical reconstruction of the PLC’s 3 main static stabilizers: the FCL, the PLT, and the popliteofibular ligament.7-9 First described in 2004,7 this anatomical PLC reconstruction technique has improved subjective and objective patient outcomes.10,11 For combined PLC injuries (eg, our patient’s injuries), Geeslin and LaPrade10 recommended concurrent reconstruction of the cruciate ligaments. In addition to the PLC reconstruction, the anatomical double-bundle PCL reconstruction used in this case has demonstrated significant improvements in subjective and objective outcome scores and objective knee stability.12
Although the stability and anatomy of this patient’s injured knee were reestablished, his development of arthrofibrosis is important. Many have discussed the commonality of arthrofibrosis or decreased ROM after extensive multiligament reconstruction surgeries.13,14 One study involving surgical management and outcomes of multiligament knee injuries found that, in more than half of its cases, restoration of full ROM required at least one operation after the initial one.13 Therefore, it is not unusual that our patient required a second operation for decreased ROM.
Conclusion
After surgery, excellent stabilization was achieved. Although the patient had setbacks related to pain and decreased ROM, his second surgery and continued physical therapy likely will help him return to his preoperative recreational activity levels.
1. Delos D, Warren RF, Marx RG. Multiligament knee injuries and their treatment. Oper Tech Sports Med. 2010;18(4):219-226.
2. Hobby B, Treme G, Wascher DC, Schenck RC. How I manage knee dislocations. Oper Tech Sports Med. 2010;18(4):227-234.
3. LaPrade RF, Ly TV, Wentorf FA, Engebretsen L. The posterolateral attachments of the knee: a qualitative and quantitative morphologic analysis of the fibular collateral ligament, popliteus tendon, popliteofibular ligament, and lateral gastrocnemius tendon. Am J Sports Med. 2003;31(6):854-860.
4. Chahla J, Nitri M, Civitarese D, Dean CS, Moulton SG, LaPrade RF. Anatomic double-bundle posterior cruciate ligament reconstruction. Arthrosc Tech. 2016;5(1):e149-e156.
5. Anderson CJ, Ziegler CG, Wijdicks CA, Engebretsen L, LaPrade RF. Arthroscopically pertinent anatomy of the anterolateral and posteromedial bundles of the posterior cruciate ligament. J Bone Joint Surg Am. 2012;94(21):1936-1945.
6. Mansour R, Yoong P, McKean D, Teh JL. The iliotibial band in acute knee trauma: patterns of injury on MR imaging. Skeletal Radiol. 2014;43(10):1369-1375.
7. LaPrade RF, Johansen S, Wentorf FA, Engebretsen L, Esterberg JL, Tso A. An analysis of an anatomical posterolateral knee reconstruction: an in vitro biomechanical study and development of a surgical technique. Am J Sports Med. 2004;32(6):1405-1414.
8. McCarthy M, Camarda L, Wijdicks CA, Johansen S, Engebretsen L, LaPrade RF. Anatomic posterolateral knee reconstructions require a popliteofibular ligament reconstruction through a tibial tunnel. Am J Sports Med. 2010;38(8):1674-1681.
9. LaPrade RF, Wozniczka JK, Stellmaker MP, Wijdicks CA. Analysis of the static function of the popliteus tendon and evaluation of an anatomic reconstruction: the “fifth ligament” of the knee. Am J Sports Med. 2010;38(3):543-549.
10. Geeslin AG, LaPrade RF. Outcomes of treatment of acute grade-III isolated and combined posterolateral knee injuries: a prospective case series and surgical technique. J Bone Joint Surg Am. 2011;93(18):1672-1683.
11. LaPrade RF, Johansen S, Agel J, Risberg MA, Moksnes H, Engebretsen L. Outcomes of an anatomic posterolateral knee reconstruction. J Bone Joint Surg Am. 2010;92(1):16-22.
12. Spiridonov SI, Slinkard NJ, LaPrade RF. Isolated and combined grade-III posterior cruciate ligament tears treated with double-bundle reconstruction with use of endoscopically placed femoral tunnels and grafts: operative technique and clinical outcomes. J Bone Joint Surg Am. 2011;93(19):1773-1780.
13. Noyes FR, Barber-Westin SD. Reconstruction of the anterior and posterior cruciate ligaments after knee dislocation. Use of early protected postoperative motion to decrease arthrofibrosis. Am J Sports Med. 1997;25(6):769-778.
14. Yenchak AJ, Wilk KE, Arrigo CA, Simpson CD, Andrews JR. Criteria-based management of an acute multistructure knee injury in a professional football player: a case report. J Orthop Sports Phys Ther. 2011;41(9):675-686.
Take-Home Points
- Reconstruction of a torn ITB is important in restoration of native anatomy and function given its properties in anterolateral stabilization and resistance to varus stress and internal tibial rotation.
- Restoration of posterolateral instability primarily involves reconstructing the FCL, PLT, and popliteofibular ligament.
- For combined PLC injuries, concurrent reconstruction of the cruciate ligaments in one stage is highly recommended.
- Post-surgery, a 6-week non-weight-bearing, limited flexion rehab protocol utilizing a dynamic PCL brace, such as the PCL Rebound brace, is recommended to prevent posterior tibial sag.
- Arthrofibrosis and decreased ROM can be seen following a violent knee injury which requires extensive multiligament reconstruction surgeries, occasionally requiring a secondary surgery for further restoration of knee motion.
Tibiofemoral knee dislocations are uncommon injuries that have devastating complications and potentially result in complex surgeries.1 Knee dislocations (KDs) can be classified with the Schenck system.2 KD-I is a multiligament injury involving the anterior cruciate ligament (ACL) or the posterior cruciate ligament (PCL), and the scale increases in severity/number of ligaments involved, with KD-V being a multiligament injury with periarticular fracture.2
In this article, we report the case of a complex multiligament knee reconstruction performed with a midsubstance iliotibial band (ITB) repair. The patient provided written informed consent for print and electronic publication of this case report.
Case Report
A 27-year-old man presented 12 days after a paraskiing crash in which he collided with a tree at 45 mph and fell 40 feet before hitting snow. Physical examination revealed a large hemarthrosis of the left lower extremity and ecchymosis about the posterolateral aspect of the knee and popliteal fossa. Range of motion (ROM) was limited from 5° of hyperextension to 90° of flexion. Additional motion was deferred secondary to pain. Varus stress testing at 0° and 30° of knee flexion demonstrated significant side-to-side differences. The Lachman test, posterior drawer test, and posterolateral drawer test were all 3+. The dial test was 3 to 4+ compared with the contralateral knee. Valgus stress testing at 0° and 30° of flexion did not reveal any side-to-side laxity. The calf was nontender, and all compartments were soft. The patient reported no neurovascular symptoms and had no neuromotor deficits other than mild common peroneal nerve dysesthesias.
Varus stress radiographs showed increased side-to-side gapping (8 mm) of the lateral compartment of the injured knee. Kneeling posterior stress radiographs, limited because of the patient’s inability to apply full stress on the injured knee secondary to pain, showed a difference of 6 mm in increased posterior translation on the uninjured leg (Figures 1A-1D).
First Surgery
1. PLC Approach. A lateral hockey-stick skin incision was made along the ITB and extended distally between the fibular head and the Gerdy tubercle. The subcutaneous tissue was then dissected, and a posteriorly based flap was developed for preservation of vascular support to the superficial tissues. The ITB and the lateral capsule had completely torn off of the femur, allowing exposure directly into the joint. The long and short heads of the biceps femoris were exposed, with about 50% of the biceps attachment torn. The FCL was torn midsubstance, and the PLT had no remnant attachment left on the femur.
2. ITB and Lateral Capsule Tag Stitched. The torn ends of the ITB were dissected and tag stitches placed in each end. Tag stitches were also placed in the lateral capsule in preparation for a direct repair.
3. Neurolysis. The common peroneal nerve was found encased in a significant amount of scar tissue, and extensive neurolysis was required. Slow, methodical dissection was performed under the partially torn long head of the biceps femoris and was continued through the scar tissue and adhesions. Distally, 5 mm to 7 mm of the peroneus longus fascia was incised as part of the neurolysis in order to prevent nerve irritation or foot drop caused by postoperative swelling.
4. PLC Tunnels. The margin between the lateral gastrocnemius tendon and the soleus muscle was identified by blunt dissection that allowed palpation of the posteromedial aspect of the fibular styloid and the popliteus musculotendinous junction. The underlying biceps bursa was incised in order to locate the midportion of the FCL remnant, which typically is tag-stitched with No. 2 FiberWire to help identify the femoral attachment (this was not done because of the complete tear at the midsubstance of the FCL).
Subperiosteal dissection of the lateral aspect of the fibular head was performed anterior to posterior and distally extended to the champagne-glass drop-off of the fibular head. Continuing the dissection distally beyond this point can endanger the common peroneal nerve. A small sulcus can be palpated where the distal FCL inserts on the fibular head. Posteriorly, a small elevator was used to dissect the soleus muscle off of the posteromedial aspect of the fibular head, where the fibular tunnel would later be created.
A Chandler retractor was placed posterior to the fibular head to protect the neurovascular bundle. With the aid of a collateral ligament aiming device, a guide pin was drilled from the lateral aspect of the fibular head (FCL attachment) to the posteromedial downslope of the fibular styloid (popliteofibular ligament attachment). The entry point of the guide pin was immediately above the champagne- glass drop-off, at the distal insertion site of the FCL, which was described as being 28.4 mm from the styloid tip and 8.2 mm posterior to the anterior margin of the fibular head.3 Care should be taken not to ream the tunnel too proximal, as doing so increases the risk of iatrogenic fracture. A 7-mm reamer was then used to drill the fibular tunnel. To facilitate later passage of the graft, a passing suture was placed through the tunnel, leaving the loop anterolateral.
Next, the starting point for the tibial tunnel was located on the flat spot of the anterolateral tibia distal and medial to the Gerdy tubercle, just lateral to the tibial tubercle. The tibial popliteal sulcus was identified by palpation of the posterolateral tibial plateau to localize the site of the popliteus musculotendinous junction, which is the ideal location of the posterior aperture of the tibial tunnel. This point is 1 cm proximal and 1 cm medial to the posteromedial exit of the fibular tunnel. A Chandler retractor was placed anterior to the lateral gastrocnemius to protect the neurovascular bundle. In the locations described earlier, a cruciate aiming device was used to place a guide pin anterior to posterior. A 9-mm tunnel was overreamed and a passing suture placed, leaving the loop posterior to facilitate graft passage.
The femoral insertions of the FCL and the PLT were then identified. ITB splitting was not necessary, given the complete midsubstance tear of this structure. The FCL attachment was identified 1.4 mm proximal and 3.1 mm posterior to the lateral epicondyle.3 Sharp dissection was performed in this location, proximal to distal, exposing the lateral epicondyle and the small sulcus at the FCL attachment site. A collateral ligament reconstruction aiming sleeve was used to drill a guide pin over the FCL femoral attachment site and out the medial aspect of the distal thigh, about 5 cm proximal and anterior to the adductor tubercle.
The femoral attachment of the PLT was reported located 18.5 mm anterior to the FCL insertion, in the anterior fifth of the popliteal sulcus.3 Although arthrotomy is usually required in order to access the PLT attachment, it was not necessary in this case, given the lateral capsule tear. A guide pin was inserted at the PLT attachment site, parallel to the FCL pin. After proper placement was verified, a 9-mm reamer was used to drill the FCL and PLT tunnels to a depth of 25 mm (socket), and a passing suture was placed into each tunnel to facilitate graft passage.
5. ACL Graft Harvest. The central third of the ipsilateral patellar tendon was harvested for use in the ACL reconstruction. Included were a 10-mm × 20-mm bone plug from the patella and a 10-mm × 25-mm bone plug from the tibial tubercle. The patella defect was then bone-grafted, and the patellar tendon closed side-to-side.
6. Graft Preparation. For the PLC, we used a split Achilles tendon allograft that had two 9-mm × 25-mm bone plugs proximally and were tubularized distally. For the PCL, we used an anterolateral bundle (ALB), which consisted of an Achilles tendon allograft that had an 11-mm × 25-mm bone plug proximally and was tubularized distally, and a posteromedial bundle (PMB), which consisted of a tibialis anterior allograft that was tubularized at both ends. For the ACL, we used a bone–patellar tendon–bone autograft 10 mm in diameter with a 20-mm femoral bone plug and a 25-mm tibial bone plug distally.
7. Arthroscopy. We created standard anterolateral and anteromedial parapatellar portals and performed arthroscopy, including lysis of adhesions. Cartilage and menisci were lesion-free.
8. PCL Femoral Tunnels. The ALB attachment was identified and outlined with a coagulator between the trochlear point and the medial arch point, adjacent to the edge of the articular cartilage. Similarly, the PMB attachment was marked about 8 mm or 9 mm posterior to the edge of the articular cartilage of the medial femoral condyle and slightly posterior to the ALB tunnel.4
In the anterolateral tunnel, an acorn reamer 11 mm in diameter was used to score the entry point of the ALB femoral tunnel. An eyelet pin was then drilled through the reamer anteromedially out the knee. Then a closed socket tunnel was reamed over the eyelet pin to a depth of 25 mm. A passing suture was pulled through the tunnel in preparation for graft passage.
With use of the same technique, a 7-mm reamer was placed against the outline of the PMB attachment site, and an eyelet pin was drilled through this reamer and out the anteromedial aspect of the knee. Again, a 25-mm deep closed socket was reamed. A bone bridge distance of 2 mm was maintained between the 2 femoral PCL bundle tunnels.
9. ACL Femoral Tunnel. The femoral ACL attachment was identified and outlined. An over-the-top guide was used to determine proper placement of the 10-mm low-profile reamer. A guide pin was drilled through the center of the reamer. The reamer was used to create a 25-mm deep closed socket tunnel, and a passing stitch was placed.
10. PCL Tibial Tunnel. With use of a 70° arthroscope for visualization, a posteromedial arthroscopic portal was created, and a shaver and a coagulator were used to identify the tibial PCL attachment, located distally along the PCL facet, until the proximal aspect of the popliteus muscle fibers were visualized. A guide pin was drilled starting at the anteromedial aspect of the tibia, about 6 cm distal to the joint line and centered between the anterior tibial crest and the medial tibial border. The pin exited posteriorly at the center of the PCL tibial attachment along the PCL bundle ridge, which was reported located between the ALB and the PMB on the tibia.5 Pin placement was verified with intraoperative lateral and anteroposterior radiographs. On the lateral radiograph, the pin should be about 6 mm or 7 mm proximal to the champagne-glass drop-off at the PCL facet on the posterior aspect of the tibia. On the anteroposterior radiograph, the pin should be 1 mm to 2 mm distal to the joint line and at the medial aspect of the lateral tibial eminence. A large curette was passed through the posteromedial arthroscopic portal both to retract the posterior tissues away from the reamer and to protect against guide-pin protrusion The guide pin was then overreamed with a 12-mm acorn reamer.
A large smoother was passed proximally up the tibial tunnel and then pulled out the anteromedial portal with a grasper. The smoother was gently cycled to smooth the intra-articular tibial tunnel aperture to remove any bony spicules that could interfere with graft passage. The smoother was then pulled back into the joint, passed out the anterolateral arthroscopic portal, and secured with a small clamp.4
11. ACL Tibial Tunnel. The ACL tibial attachment site was identified and cleaned of soft tissue. A guide pin was placed and then overreamed with a 10-mm acorn reamer.
12. PCL Femoral Fixation. The PMB graft was passed into its tunnel and secured with a 7-mm × 23-mm titanium screw. Next, the ALB was secured to the femur with a 7-mm × 20-mm titanium screw. The smoother was used to pull both grafts down through the tibial tunnel.
13. ACL Femoral Fixation. A 7-mm × 20-mm titanium screw was then used to fix the ACL autograft inside the femur. Traction was applied to the 3 cruciate grafts. There was no sign of impingement.
14. PLC Femoral Fixation. The FCL and the popliteus bone plugs were passed into their respective femoral sockets and secured with 7-mm × 20-mm titanium screws.
15. Lateral Capsule Femoral Anchors. Two suture anchors were placed into the femur, and the sutures were passed through the femoral portion of the lateral capsule for later repair.
16. PCL Tibial Fixation. Both grafts were fixed with a fully threaded bicortical 6.5-mm × 40-mm cannulated cancellous screw and an 18-mm spiked washer. The ALB was fixed first, with the knee flexed to 90°, traction on the graft, and the tibia in neutral rotation. Restoration of the normal tibiofemoral step-off was verified. The PMB was then fixed with the knee in full extension. A posterior drawer test was performed to verify restoration of stability.
17. PLC Fibula Fixation. The PLT graft was passed down the popliteal hiatus, and the FCL graft was passed under the remnant of the biceps bursa on the fibular head and then through the fibular head, anterolateral to posteromedial. The FCL graft was fixed in the fibular tunnel with the knee in 20° of flexion, a slight valgus reduction force, the tibia in neutral rotation, and traction on the graft. A 7-mm × 23-mm bioabsorbable screw was used.
18. Lateral Capsular Repair. The lateral capsule was directly repaired with the previously placed sutures. The sutures were tied with the knee in 20° of flexion.
19. PLC Tibial Fixation. The grafts were passed together, posterior to anterior, through the tibial tunnel. The knee was cycled several times through complete flexion/extension ROM. A 9-mm × 23-mm bioabsorbable screw was then used to fix the grafts to the tibia. During this fixation, the knee was kept in 60° of flexion and neutral rotation while traction was being applied to the distal end of both grafts.
20. ACL Tibial Fixation. A 9-mm × 20-mm titanium screw was used to fix the ACL graft with the knee in full extension. The graft was then viewed intra-articularly to confirm there was no impingement. The Lachman, posterior drawer, posterolateral drawer, dial, and varus stress tests were performed to ensure restoration of stability.
21. ITB Repair. A portion of the remaining Achilles tendon allograft was used to perform ITB reconstruction (reconstitution of the gaped portion of the ITB). Orthocord (DePuy Synthes) and Vicryl (Ethicon) sutures were used for this reconstruction. Knee stability was deemed restored, and the incisions were closed in standard layered fashion.
First Surgery: Postoperative Management
The patient remained non-weight-bearing the first 6 weeks after surgery, with prone knee flexion limited (0°-90°) the first 2 weeks. In addition, a PCL Jack brace (Albrecht) was placed 1 week after surgery and was to be worn at all times to decrease stress on the PCL grafts.
As ROM was not progressing as expected, the patient was instructed to use a continuous passive motion (CPM) machine 2 hours 3 times a day. About 4 weeks after surgery, with ROM still not progressing, the frequency of use of this machine was increased.
Despite continued physical therapy, use of the CPM machine, and pain management, ROM was limited (11°-90° of flexion) 5.5 months after left knee multiligament reconstruction. However, stress radiographs showed excellent stability. Varus stress radiographs showed a side-to-side difference of 0.3 mm less on the left (injured) knee, and kneeling PCL stress radiographs showed a side-to-side difference of 1.3 mm more on the left knee (Figures 3A-3D).
Second Surgery and Postoperative Management
As gentle manipulation under anesthesia was unsuccessful, the patient underwent knee arthroscopy, including 4-compartment lysis of adhesions, arthroscopically assisted posteromedial capsular release, and post-débridement manipulation under anesthesia. During manipulation, full extension and knee flexion up to 135° were achieved. ACL, PCL, and popliteus grafts were visualized and confirmed to be intact.
After this second surgery, the patient was to resume physical therapy and begin weight- bearing as tolerated. Active ROM was prioritized in an attempt to reach full ROM. In addition, a CPM machine was to be used from 0° to 135° of knee flexion 4 hours 3 times a day for 6 weeks.
Two weeks after surgery, the patient had continued pain, and extracapsular swelling in the left knee. However, ROM (0°-115° of flexion) was improved relative to before surgery (11°-90° of flexion), though it remained below the range on the contralateral side. Of note, the patient reported having a flexion contracture (~10°) in the immediate postoperative period. He had woken up with it after sleeping with the CPM machine the night before. The contracture delayed his physical therapy for several hours and resulted in a redesign of his therapy protocol to emphasize full, active knee extension and patellar mobilization, as well as discontinuation of use of the CPM machine. Corticosteroids were initiated to help with the extracapsular swelling, and the new therapy regimen brought adequate progress in ROM. Four months after the second surgery, the patient had full extension and 135° of flexion and was transitioned into wearing the PCL Rebound brace.
Discussion
This case was unique because of the midsubstance ITB tear and simultaneous multiligament injury caused by a KD-IIIL, a KD involving the ACL, the PCL, and the PLC with the medial side intact. There is limited research on ITB repair generally, with or without KD involvement. In a retrospective review of acute knee trauma cases, ITB pathologies were seen on 45% of reviewed MRI scans, and only 3% of the injuries were grade III; in addition, only 9 (5%) of the 200 cases involved both ITB and multiligament (ACL, PCL) knee injuries.6
After our patient’s ACL, PCL, and PLC were reconstructed, a fan piece of the Achilles tendon allograft from the PLC reconstruction was used to repair the ITB. The graft was used to reconstitute the torn gapped portion of the band in multiple locations, and this repair helped restore stability. The literature has reported numerous surgical uses for a portion of the ITB but few studies on repairing this anatomical structure. Preservation of the ITB is important to restoration of native anatomy and function. The ITB helps with anterolateral stabilization of the knee and with resistance of varus stress and internal tibial rotation.
The PLC reconstruction used in this case has been biomechanically validated as restoring the knee to near native stability through anatomical reconstruction of the PLC’s 3 main static stabilizers: the FCL, the PLT, and the popliteofibular ligament.7-9 First described in 2004,7 this anatomical PLC reconstruction technique has improved subjective and objective patient outcomes.10,11 For combined PLC injuries (eg, our patient’s injuries), Geeslin and LaPrade10 recommended concurrent reconstruction of the cruciate ligaments. In addition to the PLC reconstruction, the anatomical double-bundle PCL reconstruction used in this case has demonstrated significant improvements in subjective and objective outcome scores and objective knee stability.12
Although the stability and anatomy of this patient’s injured knee were reestablished, his development of arthrofibrosis is important. Many have discussed the commonality of arthrofibrosis or decreased ROM after extensive multiligament reconstruction surgeries.13,14 One study involving surgical management and outcomes of multiligament knee injuries found that, in more than half of its cases, restoration of full ROM required at least one operation after the initial one.13 Therefore, it is not unusual that our patient required a second operation for decreased ROM.
Conclusion
After surgery, excellent stabilization was achieved. Although the patient had setbacks related to pain and decreased ROM, his second surgery and continued physical therapy likely will help him return to his preoperative recreational activity levels.
Take-Home Points
- Reconstruction of a torn ITB is important in restoration of native anatomy and function given its properties in anterolateral stabilization and resistance to varus stress and internal tibial rotation.
- Restoration of posterolateral instability primarily involves reconstructing the FCL, PLT, and popliteofibular ligament.
- For combined PLC injuries, concurrent reconstruction of the cruciate ligaments in one stage is highly recommended.
- Post-surgery, a 6-week non-weight-bearing, limited flexion rehab protocol utilizing a dynamic PCL brace, such as the PCL Rebound brace, is recommended to prevent posterior tibial sag.
- Arthrofibrosis and decreased ROM can be seen following a violent knee injury which requires extensive multiligament reconstruction surgeries, occasionally requiring a secondary surgery for further restoration of knee motion.
Tibiofemoral knee dislocations are uncommon injuries that have devastating complications and potentially result in complex surgeries.1 Knee dislocations (KDs) can be classified with the Schenck system.2 KD-I is a multiligament injury involving the anterior cruciate ligament (ACL) or the posterior cruciate ligament (PCL), and the scale increases in severity/number of ligaments involved, with KD-V being a multiligament injury with periarticular fracture.2
In this article, we report the case of a complex multiligament knee reconstruction performed with a midsubstance iliotibial band (ITB) repair. The patient provided written informed consent for print and electronic publication of this case report.
Case Report
A 27-year-old man presented 12 days after a paraskiing crash in which he collided with a tree at 45 mph and fell 40 feet before hitting snow. Physical examination revealed a large hemarthrosis of the left lower extremity and ecchymosis about the posterolateral aspect of the knee and popliteal fossa. Range of motion (ROM) was limited from 5° of hyperextension to 90° of flexion. Additional motion was deferred secondary to pain. Varus stress testing at 0° and 30° of knee flexion demonstrated significant side-to-side differences. The Lachman test, posterior drawer test, and posterolateral drawer test were all 3+. The dial test was 3 to 4+ compared with the contralateral knee. Valgus stress testing at 0° and 30° of flexion did not reveal any side-to-side laxity. The calf was nontender, and all compartments were soft. The patient reported no neurovascular symptoms and had no neuromotor deficits other than mild common peroneal nerve dysesthesias.
Varus stress radiographs showed increased side-to-side gapping (8 mm) of the lateral compartment of the injured knee. Kneeling posterior stress radiographs, limited because of the patient’s inability to apply full stress on the injured knee secondary to pain, showed a difference of 6 mm in increased posterior translation on the uninjured leg (Figures 1A-1D).
First Surgery
1. PLC Approach. A lateral hockey-stick skin incision was made along the ITB and extended distally between the fibular head and the Gerdy tubercle. The subcutaneous tissue was then dissected, and a posteriorly based flap was developed for preservation of vascular support to the superficial tissues. The ITB and the lateral capsule had completely torn off of the femur, allowing exposure directly into the joint. The long and short heads of the biceps femoris were exposed, with about 50% of the biceps attachment torn. The FCL was torn midsubstance, and the PLT had no remnant attachment left on the femur.
2. ITB and Lateral Capsule Tag Stitched. The torn ends of the ITB were dissected and tag stitches placed in each end. Tag stitches were also placed in the lateral capsule in preparation for a direct repair.
3. Neurolysis. The common peroneal nerve was found encased in a significant amount of scar tissue, and extensive neurolysis was required. Slow, methodical dissection was performed under the partially torn long head of the biceps femoris and was continued through the scar tissue and adhesions. Distally, 5 mm to 7 mm of the peroneus longus fascia was incised as part of the neurolysis in order to prevent nerve irritation or foot drop caused by postoperative swelling.
4. PLC Tunnels. The margin between the lateral gastrocnemius tendon and the soleus muscle was identified by blunt dissection that allowed palpation of the posteromedial aspect of the fibular styloid and the popliteus musculotendinous junction. The underlying biceps bursa was incised in order to locate the midportion of the FCL remnant, which typically is tag-stitched with No. 2 FiberWire to help identify the femoral attachment (this was not done because of the complete tear at the midsubstance of the FCL).
Subperiosteal dissection of the lateral aspect of the fibular head was performed anterior to posterior and distally extended to the champagne-glass drop-off of the fibular head. Continuing the dissection distally beyond this point can endanger the common peroneal nerve. A small sulcus can be palpated where the distal FCL inserts on the fibular head. Posteriorly, a small elevator was used to dissect the soleus muscle off of the posteromedial aspect of the fibular head, where the fibular tunnel would later be created.
A Chandler retractor was placed posterior to the fibular head to protect the neurovascular bundle. With the aid of a collateral ligament aiming device, a guide pin was drilled from the lateral aspect of the fibular head (FCL attachment) to the posteromedial downslope of the fibular styloid (popliteofibular ligament attachment). The entry point of the guide pin was immediately above the champagne- glass drop-off, at the distal insertion site of the FCL, which was described as being 28.4 mm from the styloid tip and 8.2 mm posterior to the anterior margin of the fibular head.3 Care should be taken not to ream the tunnel too proximal, as doing so increases the risk of iatrogenic fracture. A 7-mm reamer was then used to drill the fibular tunnel. To facilitate later passage of the graft, a passing suture was placed through the tunnel, leaving the loop anterolateral.
Next, the starting point for the tibial tunnel was located on the flat spot of the anterolateral tibia distal and medial to the Gerdy tubercle, just lateral to the tibial tubercle. The tibial popliteal sulcus was identified by palpation of the posterolateral tibial plateau to localize the site of the popliteus musculotendinous junction, which is the ideal location of the posterior aperture of the tibial tunnel. This point is 1 cm proximal and 1 cm medial to the posteromedial exit of the fibular tunnel. A Chandler retractor was placed anterior to the lateral gastrocnemius to protect the neurovascular bundle. In the locations described earlier, a cruciate aiming device was used to place a guide pin anterior to posterior. A 9-mm tunnel was overreamed and a passing suture placed, leaving the loop posterior to facilitate graft passage.
The femoral insertions of the FCL and the PLT were then identified. ITB splitting was not necessary, given the complete midsubstance tear of this structure. The FCL attachment was identified 1.4 mm proximal and 3.1 mm posterior to the lateral epicondyle.3 Sharp dissection was performed in this location, proximal to distal, exposing the lateral epicondyle and the small sulcus at the FCL attachment site. A collateral ligament reconstruction aiming sleeve was used to drill a guide pin over the FCL femoral attachment site and out the medial aspect of the distal thigh, about 5 cm proximal and anterior to the adductor tubercle.
The femoral attachment of the PLT was reported located 18.5 mm anterior to the FCL insertion, in the anterior fifth of the popliteal sulcus.3 Although arthrotomy is usually required in order to access the PLT attachment, it was not necessary in this case, given the lateral capsule tear. A guide pin was inserted at the PLT attachment site, parallel to the FCL pin. After proper placement was verified, a 9-mm reamer was used to drill the FCL and PLT tunnels to a depth of 25 mm (socket), and a passing suture was placed into each tunnel to facilitate graft passage.
5. ACL Graft Harvest. The central third of the ipsilateral patellar tendon was harvested for use in the ACL reconstruction. Included were a 10-mm × 20-mm bone plug from the patella and a 10-mm × 25-mm bone plug from the tibial tubercle. The patella defect was then bone-grafted, and the patellar tendon closed side-to-side.
6. Graft Preparation. For the PLC, we used a split Achilles tendon allograft that had two 9-mm × 25-mm bone plugs proximally and were tubularized distally. For the PCL, we used an anterolateral bundle (ALB), which consisted of an Achilles tendon allograft that had an 11-mm × 25-mm bone plug proximally and was tubularized distally, and a posteromedial bundle (PMB), which consisted of a tibialis anterior allograft that was tubularized at both ends. For the ACL, we used a bone–patellar tendon–bone autograft 10 mm in diameter with a 20-mm femoral bone plug and a 25-mm tibial bone plug distally.
7. Arthroscopy. We created standard anterolateral and anteromedial parapatellar portals and performed arthroscopy, including lysis of adhesions. Cartilage and menisci were lesion-free.
8. PCL Femoral Tunnels. The ALB attachment was identified and outlined with a coagulator between the trochlear point and the medial arch point, adjacent to the edge of the articular cartilage. Similarly, the PMB attachment was marked about 8 mm or 9 mm posterior to the edge of the articular cartilage of the medial femoral condyle and slightly posterior to the ALB tunnel.4
In the anterolateral tunnel, an acorn reamer 11 mm in diameter was used to score the entry point of the ALB femoral tunnel. An eyelet pin was then drilled through the reamer anteromedially out the knee. Then a closed socket tunnel was reamed over the eyelet pin to a depth of 25 mm. A passing suture was pulled through the tunnel in preparation for graft passage.
With use of the same technique, a 7-mm reamer was placed against the outline of the PMB attachment site, and an eyelet pin was drilled through this reamer and out the anteromedial aspect of the knee. Again, a 25-mm deep closed socket was reamed. A bone bridge distance of 2 mm was maintained between the 2 femoral PCL bundle tunnels.
9. ACL Femoral Tunnel. The femoral ACL attachment was identified and outlined. An over-the-top guide was used to determine proper placement of the 10-mm low-profile reamer. A guide pin was drilled through the center of the reamer. The reamer was used to create a 25-mm deep closed socket tunnel, and a passing stitch was placed.
10. PCL Tibial Tunnel. With use of a 70° arthroscope for visualization, a posteromedial arthroscopic portal was created, and a shaver and a coagulator were used to identify the tibial PCL attachment, located distally along the PCL facet, until the proximal aspect of the popliteus muscle fibers were visualized. A guide pin was drilled starting at the anteromedial aspect of the tibia, about 6 cm distal to the joint line and centered between the anterior tibial crest and the medial tibial border. The pin exited posteriorly at the center of the PCL tibial attachment along the PCL bundle ridge, which was reported located between the ALB and the PMB on the tibia.5 Pin placement was verified with intraoperative lateral and anteroposterior radiographs. On the lateral radiograph, the pin should be about 6 mm or 7 mm proximal to the champagne-glass drop-off at the PCL facet on the posterior aspect of the tibia. On the anteroposterior radiograph, the pin should be 1 mm to 2 mm distal to the joint line and at the medial aspect of the lateral tibial eminence. A large curette was passed through the posteromedial arthroscopic portal both to retract the posterior tissues away from the reamer and to protect against guide-pin protrusion The guide pin was then overreamed with a 12-mm acorn reamer.
A large smoother was passed proximally up the tibial tunnel and then pulled out the anteromedial portal with a grasper. The smoother was gently cycled to smooth the intra-articular tibial tunnel aperture to remove any bony spicules that could interfere with graft passage. The smoother was then pulled back into the joint, passed out the anterolateral arthroscopic portal, and secured with a small clamp.4
11. ACL Tibial Tunnel. The ACL tibial attachment site was identified and cleaned of soft tissue. A guide pin was placed and then overreamed with a 10-mm acorn reamer.
12. PCL Femoral Fixation. The PMB graft was passed into its tunnel and secured with a 7-mm × 23-mm titanium screw. Next, the ALB was secured to the femur with a 7-mm × 20-mm titanium screw. The smoother was used to pull both grafts down through the tibial tunnel.
13. ACL Femoral Fixation. A 7-mm × 20-mm titanium screw was then used to fix the ACL autograft inside the femur. Traction was applied to the 3 cruciate grafts. There was no sign of impingement.
14. PLC Femoral Fixation. The FCL and the popliteus bone plugs were passed into their respective femoral sockets and secured with 7-mm × 20-mm titanium screws.
15. Lateral Capsule Femoral Anchors. Two suture anchors were placed into the femur, and the sutures were passed through the femoral portion of the lateral capsule for later repair.
16. PCL Tibial Fixation. Both grafts were fixed with a fully threaded bicortical 6.5-mm × 40-mm cannulated cancellous screw and an 18-mm spiked washer. The ALB was fixed first, with the knee flexed to 90°, traction on the graft, and the tibia in neutral rotation. Restoration of the normal tibiofemoral step-off was verified. The PMB was then fixed with the knee in full extension. A posterior drawer test was performed to verify restoration of stability.
17. PLC Fibula Fixation. The PLT graft was passed down the popliteal hiatus, and the FCL graft was passed under the remnant of the biceps bursa on the fibular head and then through the fibular head, anterolateral to posteromedial. The FCL graft was fixed in the fibular tunnel with the knee in 20° of flexion, a slight valgus reduction force, the tibia in neutral rotation, and traction on the graft. A 7-mm × 23-mm bioabsorbable screw was used.
18. Lateral Capsular Repair. The lateral capsule was directly repaired with the previously placed sutures. The sutures were tied with the knee in 20° of flexion.
19. PLC Tibial Fixation. The grafts were passed together, posterior to anterior, through the tibial tunnel. The knee was cycled several times through complete flexion/extension ROM. A 9-mm × 23-mm bioabsorbable screw was then used to fix the grafts to the tibia. During this fixation, the knee was kept in 60° of flexion and neutral rotation while traction was being applied to the distal end of both grafts.
20. ACL Tibial Fixation. A 9-mm × 20-mm titanium screw was used to fix the ACL graft with the knee in full extension. The graft was then viewed intra-articularly to confirm there was no impingement. The Lachman, posterior drawer, posterolateral drawer, dial, and varus stress tests were performed to ensure restoration of stability.
21. ITB Repair. A portion of the remaining Achilles tendon allograft was used to perform ITB reconstruction (reconstitution of the gaped portion of the ITB). Orthocord (DePuy Synthes) and Vicryl (Ethicon) sutures were used for this reconstruction. Knee stability was deemed restored, and the incisions were closed in standard layered fashion.
First Surgery: Postoperative Management
The patient remained non-weight-bearing the first 6 weeks after surgery, with prone knee flexion limited (0°-90°) the first 2 weeks. In addition, a PCL Jack brace (Albrecht) was placed 1 week after surgery and was to be worn at all times to decrease stress on the PCL grafts.
As ROM was not progressing as expected, the patient was instructed to use a continuous passive motion (CPM) machine 2 hours 3 times a day. About 4 weeks after surgery, with ROM still not progressing, the frequency of use of this machine was increased.
Despite continued physical therapy, use of the CPM machine, and pain management, ROM was limited (11°-90° of flexion) 5.5 months after left knee multiligament reconstruction. However, stress radiographs showed excellent stability. Varus stress radiographs showed a side-to-side difference of 0.3 mm less on the left (injured) knee, and kneeling PCL stress radiographs showed a side-to-side difference of 1.3 mm more on the left knee (Figures 3A-3D).
Second Surgery and Postoperative Management
As gentle manipulation under anesthesia was unsuccessful, the patient underwent knee arthroscopy, including 4-compartment lysis of adhesions, arthroscopically assisted posteromedial capsular release, and post-débridement manipulation under anesthesia. During manipulation, full extension and knee flexion up to 135° were achieved. ACL, PCL, and popliteus grafts were visualized and confirmed to be intact.
After this second surgery, the patient was to resume physical therapy and begin weight- bearing as tolerated. Active ROM was prioritized in an attempt to reach full ROM. In addition, a CPM machine was to be used from 0° to 135° of knee flexion 4 hours 3 times a day for 6 weeks.
Two weeks after surgery, the patient had continued pain, and extracapsular swelling in the left knee. However, ROM (0°-115° of flexion) was improved relative to before surgery (11°-90° of flexion), though it remained below the range on the contralateral side. Of note, the patient reported having a flexion contracture (~10°) in the immediate postoperative period. He had woken up with it after sleeping with the CPM machine the night before. The contracture delayed his physical therapy for several hours and resulted in a redesign of his therapy protocol to emphasize full, active knee extension and patellar mobilization, as well as discontinuation of use of the CPM machine. Corticosteroids were initiated to help with the extracapsular swelling, and the new therapy regimen brought adequate progress in ROM. Four months after the second surgery, the patient had full extension and 135° of flexion and was transitioned into wearing the PCL Rebound brace.
Discussion
This case was unique because of the midsubstance ITB tear and simultaneous multiligament injury caused by a KD-IIIL, a KD involving the ACL, the PCL, and the PLC with the medial side intact. There is limited research on ITB repair generally, with or without KD involvement. In a retrospective review of acute knee trauma cases, ITB pathologies were seen on 45% of reviewed MRI scans, and only 3% of the injuries were grade III; in addition, only 9 (5%) of the 200 cases involved both ITB and multiligament (ACL, PCL) knee injuries.6
After our patient’s ACL, PCL, and PLC were reconstructed, a fan piece of the Achilles tendon allograft from the PLC reconstruction was used to repair the ITB. The graft was used to reconstitute the torn gapped portion of the band in multiple locations, and this repair helped restore stability. The literature has reported numerous surgical uses for a portion of the ITB but few studies on repairing this anatomical structure. Preservation of the ITB is important to restoration of native anatomy and function. The ITB helps with anterolateral stabilization of the knee and with resistance of varus stress and internal tibial rotation.
The PLC reconstruction used in this case has been biomechanically validated as restoring the knee to near native stability through anatomical reconstruction of the PLC’s 3 main static stabilizers: the FCL, the PLT, and the popliteofibular ligament.7-9 First described in 2004,7 this anatomical PLC reconstruction technique has improved subjective and objective patient outcomes.10,11 For combined PLC injuries (eg, our patient’s injuries), Geeslin and LaPrade10 recommended concurrent reconstruction of the cruciate ligaments. In addition to the PLC reconstruction, the anatomical double-bundle PCL reconstruction used in this case has demonstrated significant improvements in subjective and objective outcome scores and objective knee stability.12
Although the stability and anatomy of this patient’s injured knee were reestablished, his development of arthrofibrosis is important. Many have discussed the commonality of arthrofibrosis or decreased ROM after extensive multiligament reconstruction surgeries.13,14 One study involving surgical management and outcomes of multiligament knee injuries found that, in more than half of its cases, restoration of full ROM required at least one operation after the initial one.13 Therefore, it is not unusual that our patient required a second operation for decreased ROM.
Conclusion
After surgery, excellent stabilization was achieved. Although the patient had setbacks related to pain and decreased ROM, his second surgery and continued physical therapy likely will help him return to his preoperative recreational activity levels.
1. Delos D, Warren RF, Marx RG. Multiligament knee injuries and their treatment. Oper Tech Sports Med. 2010;18(4):219-226.
2. Hobby B, Treme G, Wascher DC, Schenck RC. How I manage knee dislocations. Oper Tech Sports Med. 2010;18(4):227-234.
3. LaPrade RF, Ly TV, Wentorf FA, Engebretsen L. The posterolateral attachments of the knee: a qualitative and quantitative morphologic analysis of the fibular collateral ligament, popliteus tendon, popliteofibular ligament, and lateral gastrocnemius tendon. Am J Sports Med. 2003;31(6):854-860.
4. Chahla J, Nitri M, Civitarese D, Dean CS, Moulton SG, LaPrade RF. Anatomic double-bundle posterior cruciate ligament reconstruction. Arthrosc Tech. 2016;5(1):e149-e156.
5. Anderson CJ, Ziegler CG, Wijdicks CA, Engebretsen L, LaPrade RF. Arthroscopically pertinent anatomy of the anterolateral and posteromedial bundles of the posterior cruciate ligament. J Bone Joint Surg Am. 2012;94(21):1936-1945.
6. Mansour R, Yoong P, McKean D, Teh JL. The iliotibial band in acute knee trauma: patterns of injury on MR imaging. Skeletal Radiol. 2014;43(10):1369-1375.
7. LaPrade RF, Johansen S, Wentorf FA, Engebretsen L, Esterberg JL, Tso A. An analysis of an anatomical posterolateral knee reconstruction: an in vitro biomechanical study and development of a surgical technique. Am J Sports Med. 2004;32(6):1405-1414.
8. McCarthy M, Camarda L, Wijdicks CA, Johansen S, Engebretsen L, LaPrade RF. Anatomic posterolateral knee reconstructions require a popliteofibular ligament reconstruction through a tibial tunnel. Am J Sports Med. 2010;38(8):1674-1681.
9. LaPrade RF, Wozniczka JK, Stellmaker MP, Wijdicks CA. Analysis of the static function of the popliteus tendon and evaluation of an anatomic reconstruction: the “fifth ligament” of the knee. Am J Sports Med. 2010;38(3):543-549.
10. Geeslin AG, LaPrade RF. Outcomes of treatment of acute grade-III isolated and combined posterolateral knee injuries: a prospective case series and surgical technique. J Bone Joint Surg Am. 2011;93(18):1672-1683.
11. LaPrade RF, Johansen S, Agel J, Risberg MA, Moksnes H, Engebretsen L. Outcomes of an anatomic posterolateral knee reconstruction. J Bone Joint Surg Am. 2010;92(1):16-22.
12. Spiridonov SI, Slinkard NJ, LaPrade RF. Isolated and combined grade-III posterior cruciate ligament tears treated with double-bundle reconstruction with use of endoscopically placed femoral tunnels and grafts: operative technique and clinical outcomes. J Bone Joint Surg Am. 2011;93(19):1773-1780.
13. Noyes FR, Barber-Westin SD. Reconstruction of the anterior and posterior cruciate ligaments after knee dislocation. Use of early protected postoperative motion to decrease arthrofibrosis. Am J Sports Med. 1997;25(6):769-778.
14. Yenchak AJ, Wilk KE, Arrigo CA, Simpson CD, Andrews JR. Criteria-based management of an acute multistructure knee injury in a professional football player: a case report. J Orthop Sports Phys Ther. 2011;41(9):675-686.
1. Delos D, Warren RF, Marx RG. Multiligament knee injuries and their treatment. Oper Tech Sports Med. 2010;18(4):219-226.
2. Hobby B, Treme G, Wascher DC, Schenck RC. How I manage knee dislocations. Oper Tech Sports Med. 2010;18(4):227-234.
3. LaPrade RF, Ly TV, Wentorf FA, Engebretsen L. The posterolateral attachments of the knee: a qualitative and quantitative morphologic analysis of the fibular collateral ligament, popliteus tendon, popliteofibular ligament, and lateral gastrocnemius tendon. Am J Sports Med. 2003;31(6):854-860.
4. Chahla J, Nitri M, Civitarese D, Dean CS, Moulton SG, LaPrade RF. Anatomic double-bundle posterior cruciate ligament reconstruction. Arthrosc Tech. 2016;5(1):e149-e156.
5. Anderson CJ, Ziegler CG, Wijdicks CA, Engebretsen L, LaPrade RF. Arthroscopically pertinent anatomy of the anterolateral and posteromedial bundles of the posterior cruciate ligament. J Bone Joint Surg Am. 2012;94(21):1936-1945.
6. Mansour R, Yoong P, McKean D, Teh JL. The iliotibial band in acute knee trauma: patterns of injury on MR imaging. Skeletal Radiol. 2014;43(10):1369-1375.
7. LaPrade RF, Johansen S, Wentorf FA, Engebretsen L, Esterberg JL, Tso A. An analysis of an anatomical posterolateral knee reconstruction: an in vitro biomechanical study and development of a surgical technique. Am J Sports Med. 2004;32(6):1405-1414.
8. McCarthy M, Camarda L, Wijdicks CA, Johansen S, Engebretsen L, LaPrade RF. Anatomic posterolateral knee reconstructions require a popliteofibular ligament reconstruction through a tibial tunnel. Am J Sports Med. 2010;38(8):1674-1681.
9. LaPrade RF, Wozniczka JK, Stellmaker MP, Wijdicks CA. Analysis of the static function of the popliteus tendon and evaluation of an anatomic reconstruction: the “fifth ligament” of the knee. Am J Sports Med. 2010;38(3):543-549.
10. Geeslin AG, LaPrade RF. Outcomes of treatment of acute grade-III isolated and combined posterolateral knee injuries: a prospective case series and surgical technique. J Bone Joint Surg Am. 2011;93(18):1672-1683.
11. LaPrade RF, Johansen S, Agel J, Risberg MA, Moksnes H, Engebretsen L. Outcomes of an anatomic posterolateral knee reconstruction. J Bone Joint Surg Am. 2010;92(1):16-22.
12. Spiridonov SI, Slinkard NJ, LaPrade RF. Isolated and combined grade-III posterior cruciate ligament tears treated with double-bundle reconstruction with use of endoscopically placed femoral tunnels and grafts: operative technique and clinical outcomes. J Bone Joint Surg Am. 2011;93(19):1773-1780.
13. Noyes FR, Barber-Westin SD. Reconstruction of the anterior and posterior cruciate ligaments after knee dislocation. Use of early protected postoperative motion to decrease arthrofibrosis. Am J Sports Med. 1997;25(6):769-778.
14. Yenchak AJ, Wilk KE, Arrigo CA, Simpson CD, Andrews JR. Criteria-based management of an acute multistructure knee injury in a professional football player: a case report. J Orthop Sports Phys Ther. 2011;41(9):675-686.
Advanced Stage and Relapsed/Refractory Hodgkin Lymphoma
INTRODUCTION
Hodgkin lymphoma, previously known as Hodgkin’s disease, is a B-cell lymphoproliferative disease characterized by a unique set of pathologic and epidemiologic features. The disease is characterized by the presence of multinucleate giant cells called Hodgkin Reed-Sternberg (HRS) cells.1 Hodgkin lymphoma is unique compared to other B-cell lymphomas because of the relative rarity of the malignant cells within affected tissues. The HRS cells, which usually account for only 0.1% to 10% of the cells, induce accumulation of nonmalignant lymphocytes, macrophages, granulocytes, eosinophils, plasma cells, and histiocytes, which then constitute the majority of tumor cellularity.2 Although the disease was first described by Sir Thomas Hodgkin in 1832, in part because of this unique histopathology, it was not until the 1990s that it was conclusively demonstrated that HRS cells are in fact monoclonal germinal center–derived B cells.
Due to the development of highly effective therapies for Hodgkin lymphoma, cure is a reasonable goal for most patients. Because of the high cure rate, late complications of therapy must be considered when selecting treatment. This article reviews the clinical features and treatment options for advanced stage and relapsed/refractory Hodgkin lymphoma. A previously published article reviewed the epidemiology, etiology/pathogenesis, pathologic classification, initial workup, and staging evaluation of Hodgkin lymphoma, as well as the prognostic stratification and treatment of patients with early-stage Hodgkin lymphoma.3
PRESENTATION, INITIAL EVALUATION, AND PROGNOSIS
Overall, classical Hodgkin lymphoma (cHL) usually presents with asymptomatic mediastinal or cervical lymphadenopathy. At least 50% of patients will have stage I or II disease.4 A mediastinal mass is seen in most patients with nodular sclerosis cHL, at times showing the characteristics of bulky (> 10 cm) disease. Constitutional, or B, symptoms (fever, night sweats, and weight loss) are present in approximately 25% of all patients with cHL, but 50% of advanced stage patients. Between 10% and 15% of patients will have extranodal disease, most commonly involving lung, bone, and liver. Lymphocyte-predominant Hodgkin lymphoma (LPHL) is a rare histological subtype of Hodgkin lymphoma that is differentiated from cHL by distinct clinicopathological features. The clinical course and treatment approach for LPHL are dependent upon the stage of disease. The clinicopathological features of LPHL are discussed in the early-stage Hodgkin lymphoma article.3
For the purposes of prognosis and selection of treatment, Hodgkin lymphoma is commonly classified as early stage favorable, early stage unfavorable, and advanced stage. For advanced stage Hodgkin lymphoma patients, prognosis can be defined using a tool commonly referred to as the International Prognostic Score (IPS). This index consists of 7 factors: male gender, age 45 years or older, stage IV disease, hemoglobin < 10.5 g/dL, white blood cell (WBC) count > 15,000/μL, lymphopenia (absolute lymphocyte count < 600 cells/μL or lymphocytes < 8% of WBC count), and serum albumin < 4 g/dL.5 In the original study by Hasenclever et al,5 the 5-year freedom from progression (FFP) ranged from 42% to 84% and the 5-year overall survival (OS) ranged from 56% to 90%, depending on the number of factors present. This scoring system, however, was developed using a patient population treated prior to 1992. Using a more recently treated patient population, the British Columbia Cancer Agency (BCCA) found that the IPS is still valid for prognostication, but outcomes have improved across all IPS groups, with 5-year FFP now ranging from 62% to 88% and 5-year OS ranging from 67% to 98%.6 This improvement is likely a reflection of improved therapy and supportive care. Table 1 shows the PFS and OS within each IPS group, comparing the data from the German Hodgkin Study Group (GHSG) and BCCA group.5,6
High expression of CD68 is associated with adverse outcomes, whereas high FOXP3 and CD20 expression on tumor cells are predictors of superior outcomes.8 A recent study found that CD68 expression was associated with OS. Five-year OS was 88% in those with less than 25% CD68 expression, versus 63% in those with greater than 25% CD68 expression.9
Roemer and colleagues evaluated 108 newly diagnosed cHL biopsy specimens and found that almost all cHL patients had concordant alteration of PD-L1 (programmed death ligand-1) and PD-L2 loci, with a spectrum of 9p24.1 alterations ranging from low level polysomy to near uniform 9p24.1 amplification. PD-L1/PD-L2 copy number alterations are therefore a defining pathobiological feature of cHL.10 PFS was significantly shorter for patients with 9p24.1 amplification, and those patients were likely to have advanced disease suggesting that 9p24.1 amplification is associated with less favorable prognosis.10 This may change with the increasing use of PD-1 inhibitors in the treatment of cHL.
High baseline metabolic tumor volume and total lesion glycolysis have also been associated with adverse outcomes in cHL. While not routinely assessed in practice currently, these tools may ultimately be used to assess prognosis and guide therapy in clinical practice.11
ADVANCED STAGE HODGKIN LYMPHOMA
FRONTLINE THERAPY
First-line Chemotherapy
Chemotherapy plays an essential role in the treatment of advanced stage Hodgkin lymphoma. In the 1960s, the MOPP regimen (nitrogen mustard, vincristine, procarbazine, prednisone) was developed, with a 10-year OS of 50% and a progression-free survival (PFS) of 52% reported in advanced stage patients. The complete remission (CR) rate was 81%, and 36% of patients who achieved CR relapsed later.12 This chemotherapy regimen is associated with a significant rate of myelosuppression and infertility as well as long-term risk of secondary myelodysplasia and acute leukemias.13,14 This led to the development of newer regimens such as ABVD (doxorubicin, bleomycin, vinblastine, dacarbazine).15 In a randomized trial, ABVD showed improved failure-free survival (FFS) over MOPP (61% versus 50% at 5 years) but similar OS (66%–73%).16 In light of these findings, and considering the lower rate of infertility and myelotoxicity, ABVD became the standard of care for advanced stage cHL in the United States.
The Stanford V regimen was developed in an attempt to further minimize toxicity.17 Stanford V is a condensed, 12-week chemotherapy regimen that includes mechlorethamine, doxorubicin, vinblastine, etoposide, prednisone, vincristine, and bleomycin, followed by involved-field radiation therapy (IFRT). Subsequent trials compared the Stanford V and ABVD regimens and showed similar OS, freedom from treatment failure (FFTF), and response rates.18,19 The ABVD regimen was noted to have higher pulmonary toxicity, while other toxicities such as lymphopenia and neuropathy were higher with the Stanford V regimen. In addition, Stanford V requires patients to receive radiation therapy (RT) to original sites of disease larger than 5 cm in size and contiguous sites.
Another regimen which has been studied extensively for advanced stage Hodgkin lymphoma, and is considered a standard of care in some parts of the world, is escalated BEACOPP (bleomycin, etoposide, doxorubicin, cyclophosphamide, vincristine, procarbazine, prednisone). In the HD9 study (n = 1196), the GHSG evaluated BEACOPP, escalated BEACOPP, and COPP/ABVD in advanced stage Hodgkin lymphoma.20 All arms of the study included 30 Gy RT to sites of bulky disease or residual disease. This study showed improved OS and FFTF with escalated BEACOPP, but at the cost of higher rates of toxicity. At 10 years, FFTF was 64%, 70%, and 82% with OS rates of 75%, 80%, and 86% for COPP/ABVD, baseline BEACOPP, and escalated BEACOPP, respectively (P < 0.001). The rate of secondary acute leukemia 10 years after treatment was 0.4% for COPP/ABVD, 1.5% for BEACOPP, and 3.0% for escalated BEACOPP. However, 3 subsequent randomized trials did not confirm a survival benefit with escalated BEACOPP relative to ABVD. In the HD 2000 trial (n = 295)21 and in a trial by Viviani and colleagues (n = 331),22 an improvement in OS was not demonstrated in favor of escalated BEACOPP. These studies also confirmed a higher rate of toxicities as well as secondary malignancies associated with the escalated BEACOPP regimen. In the EORTC20012 Intergroup trial (n = 549), 8 cycles of ABVD was compared with 4 cycles of escalated BEACOPP followed by 4 cycles of baseline BEACOPP, without radiation, in patients with clinical stage III or IV Hodgkin lymphoma with IPS score ≥ 3. Both regimens resulted in statistically similar FFS (63.7% in ABVD × 8 versus 69.3% in BEACOPP 4+4) and OS (86.7% in ABVD × 8 vs 90.3% in BEACOPP 4+4).23
In the United States, ABVD (6–8 cycles) is commonly used, although escalated BEACOPP (particularly for patients with an IPS of 4 or higher) and Stanford V are considered appropriate as well.24 In the North American Intergroup study comparing ABVD to Stanford V, and in the trial by Viviani et al, ABVD was associated with a 5- to 7-year FFS of 73% to 79% and OS of 84% to 92%.19,22 Given these excellent results, as well as the potential to cure patients with second-line therapy consisting of autologous hematopoietic cell transplantation (auto-HCT), the general consensus among most U.S. hematologists and oncologists is that ABVD remains the treatment of choice, and that the improved FFS/PFS with escalated BEACOPP is not outweighed by the additional toxicity associated with the regimen. There may, however, be a role for escalated BEACOPP in select patients who have a suboptimal response to ABVD as defined by interim positron emission tomography (iPET) scan (see below).
Brentuximab vedotin is an anti-CD30 antibody-drug conjugate (ADC) consisting of an anti-CD30 antibody linked to monomethyl auristatin E (MMAE), a potent antitubulin agent. CD30 is highly expressed on HRS cells and also in anaplastic large cell lymphoma. Upon binding to CD30, the ADC/CD30 complex is then internalized and directed to the lysosome, where the ADC is proteolytically cleaved, releasing MMAE from the antibody. MMAE then disrupts microtubule networks within the cell, leading to G2/M cycle arrest and apoptosis. CD30 is consistently expressed on HRS cells. In addition to being studied in the relapsed/refractory setting (described below), brentuximab has been studied in the first-line setting. In a phase 1 trial, brentuximab combined with ABVD was associated with increased pulmonary toxicity, while brentuximab + AVD had no significant pulmonary toxicity, with an excellent CR rate (96%), suggesting that substituting brentuximab for bleomycin may be an effective strategy. In addition to possibly being more efficacious, this strategy would also have the benefit of eliminating the risk of bleomycin pulmonary toxicity.25 Based on this data, a large international phase 3 study (the ECHELON-1 trial) comparing ABVD versus brentuximab + AVD in advanced stage cHL patients was recently completed. This study enrolled 1334 patients, and preliminary results were recently announced. With a median follow-up of 24 months, the brentuximab + AVD arm had a 4.9% absolute improvement in PFS relative to the ABVD arm (82.1% versus 77.2%). The brentuximab + AVD arm had an increased incidence of febrile neutropenia, managed with growth factors and peripheral neuropathy requiring dose adjustments, whereas the ABVD arm had an increased rate and severity of pulmonary toxicity.26 Further follow-up will be required to determine whether this will translate into a survival benefit. See Table 2 for a summary of recent large randomized prospective phase 3 trials in advanced stage Hodgkin lymphoma.
Alternative Regimens in Older Patients
Patients older than 60 years of age often have poor tolerance for ABVD and especially escalated BEACOPP. This results in increased treatment-related mortality and reduced overall dose intensity, with higher relapse rates and poor OS. In an attempt to improve on the results of treatment of elderly patients with Hodgkin lymphoma, alternative regimens have been explored. One example is PVAG (prednisone, vinblastine, doxorubicin, gemcitabine). With this regimen, the 3-year OS was 66% and PFS was 58%. One patient out of 59 died from treatment-related toxicity, which is much improved over the historical figures for elderly patients with Hodgkin lymphoma.27 Another commonly used approach in practice is to simply omit bleomycin from ABVD. In the early-stage setting (GHSG HD-13 trial), this regimen (referred to as AVD) led to 89.6% PFS at 5 years, compared to 93.5% with ABVD.28 It therefore stands to reason that this should be a reasonable option in older or more frail advanced stage cHL patients as well.
Brentuximab has been evaluated as a single-agent therapy for first-line therapy of elderly patients with Hodgkin lymphoma. In a phase 2 study, 27 patients (63% with advanced stage disease) were treated, with a 92% overall response rate and 73% CR rate. However the median duration of remission was disappointing at only 9.1 months.29 Based on this data, single-agent brentuximab appears to be a reasonable and well tolerated option for frail or elderly patients, although with the caveat that long-term disease control is relatively uncommon.
RESPONSE-ADAPTED FRONTLINE THERAPY USING INTERIM PET SCAN
In recent years, response-adapted treatment approaches have been extensively researched in cHL using iPET. The goal is to reduce toxicity by minimizing therapy in those who achieve negative iPET and/or to intensify treatment for patients with suboptimal response on iPET. Gallamini et al evaluated the prognostic role of an early iPET scan in advanced Hodgkin lymphoma patients (n = 190) treated with ABVD. This study found that patients with positive iPET had a 2-year PFS of 12.8% versus 95.0% in patients with negative iPET. This result was highly statistically significant (P < 0.0001). This study also showed that PET-2 (iPET after 2 cycles of ABVD) superseded the prognostic value of the IPS at diagnosis.30 As a result, numerous subsequent studies have been pursued using iPET for risk-adapted treatment in cHL.
A critical element to the conduct of iPET risk-adapted treatment for cHL is the interpretation of the iPET. In hopes of standardizing iPET interpretation in clinical trials, a scoring system called the Deauville score was developed. The Deauville score ranges from 1 to 5 (Table 3).
The SWOG (Southwest Oncology Group) S0816 trial (n = 358) evaluated iPET-adapted treatment after 2 cycles of ABVD in stage III or IV Hodgkin lymphoma patients. Patients with positive iPET (Deauville score 4 to 5; n = 60) received escalated BEACOPP for 6 cycles, whereas iPET-negative (Deauville score 1 to 3; n = 271) patients continued to receive 4 more cycles of ABVD. The 2-year PFS was 64% for iPET-positive patients.33 This PFS was much higher than the expected 15% to 30% from prior studies such as Gallamini et al,30 suggesting that the treatment intensification may have been of benefit.
In the HD0801 study (n = 519), newly diagnosed advanced Hodgkin lymphoma patients with positive iPET after 2 cycles of ABVD (n = 103) received early ifosfamide-containing salvage therapy followed by high-dose therapy with autologous stem cell rescue. The 2-year PFS was 76% for PET-2–positive patients, comparable with PET-2–negative patients who had PFS of 81%.34 Again, this result for iPET-positive patients was much better than expected based on the historical control from Gallamini et al, suggesting that the treatment intensification may have been beneficial. It should be emphasized, however, that neither HD0801 nor S0816 were randomized prospective trials; rather, all iPET-positive patients were assigned to an intensified treatment approach.
In the HD18 trial (n = 1100), patients with advanced stage cHL started therapy with escalated BEACOPP and underwent an iPET after 2 cycles. For those with a positive iPET, rituximab was added to escalated BEACOPP in the experimental arm (n = 220) for cycles 3 through 8. The control group (n = 220) continued to receive 6 more cycles of escalated BEACOPP. In the 2 groups, the 3-year PFS was similar (91.4% in escalated BEACOPP, 93% in rituximab + escalated BEACOPP), suggesting no significant benefit with addition of rituximab.35 This study also calls into question whether iPET provides useful information for patients receiving intensive therapy such as escalated BEACOPP, and indicates that the historical control data for iPET-positive patients from Gallamini et al may not be consistently reproduced in other prospective trials. As a result, nonrandomized trials that implement an iPET risk-adapted approach should be interpreted with caution. See Table 4 for a summary of recent trials in advanced stage Hodgkin lymphoma using iPET scan to guide therapy.
RADIATION THERAPY IN FRONTLINE TREATMENT
In patients with advanced stage Hodgkin lymphoma, IFRT to initial bulky sites of disease may be incorporated into frontline therapy to improve local control. However, whether this provides a survival benefit and which patients benefit most from consolidative RT remain unclear.
The European Organization for Research and Treatment of Cancer (EORTC) completed a randomized study in advanced stage Hodgkin lymphoma patients who achieved complete or partial remission after MOPP-ABV.36 Patients in CR were randomly assigned to receive no further treatment versus IFRT (24 Gy to all initially involved nodal areas and 16 to 24 Gy to all initially involved extranodal sites). Patients in partial remission (PR) were treated with 30 Gy to nodal areas and 18 to 24 Gy to extranodal sites. Among the CR patients, the 5-year event-free survival (EFS) was 79% to 84% and did not differ for those who received radiation versus those who did not. Five-year OS was 85% to 91% and also did not differ between the 2 groups. However, among the patients in PR after chemotherapy, the 5-year EFS was 79% and the 5-year OS was 87%, which is better than expected for PR patients, indicating a possible benefit to RT in patients with a partial response after chemotherapy. In the GHSG HD12 trial, patients with advanced stage Hodgkin lymphoma who had a residual lesion by computed tomography (CT) (but not analyzed by PET) had a very subtle improvement in FFTF (90% versus 87%) in favor of consolidation with IFRT, but again no survival benefit was seen.37
The EORTC and HD12 studies described above utilized CT scan for assigning remission status following chemotherapy, and it is now well known that many patients with residual masses (by CT) after chemotherapy may in fact be cured, as such residual radiographic abnormalities may simply be composed of fibrosis. PET scan is more accurate than CT in identifying patients who truly have residual active disease following chemotherapy. As a result, the EORTC study discussed above and the GHSG HD12 trial are of limited relevance in the modern era, in which patients routinely undergo PET scan at the end of therapy. Restricting IFRT to sites that remain PET-positive after completing chemotherapy may be a reasonable strategy that would allow for the avoidance of RT in many patients, and may obviate the need for aggressive second-line therapy (eg, high-dose therapy and autologous hematopoietic cell transplant [auto-HCT]). This approach was taken in the GHSG HD15 trial (n = 2182) in which advanced stage patients were treated with 3 variations on the BEACOPP regimen (8 cycles of escalated BEACOPP, 6 cycles of escalated BEACOPP, or 8 cycles of baseline BEACOPP, randomized in a 1:1:1 ratio). Patients with a residual mass of 2.5 cm or greater on CT scan then underwent a PET scan; if the lesion was PET positive, it was treated with 30 Gy of IFRT. This overall strategy was very effective, with 5-year FFTF rates of 84.4%, 89.3%, and 85.4%, respectively. The OS rates were 91.9%, 95.3%, and 94.5%, respectively. For patients with lesions that remained PET positive after chemotherapy, the PFS rate was 86.2% at 48 months, whereas patients in PR with persistent mass ≥ 2.5 cm but with negative PET had a PFS of 92.6%, similar to that of patients in CR.38 With this approach of BEACOPP followed by PET-guided radiation, the proportion of patients receiving RT was reduced from 71% (in the HD9 study) to only 11% in the HD15 study,38 with no apparent loss in overall efficacy when comparing the results of the 2 studies.
UPFRONT STEM CELL TRANSPLANTATION
To further improve outcomes of patients with advanced Hodgkin lymphoma with high-risk disease, high-dose therapy with auto-HCT has been explored as part of frontline therapy. While this has been shown to be feasible in such patients,39 randomized trials have not shown a clear benefit in terms of FFS or OS with upfront auto-HCT. 40,41 Therefore, auto-HCT is not considered a standard component of frontline therapy for cHL patients who achieve CR by PET/CT scan.
RELAPSED AND REFRACTORY HODGKIN LYMPHOMA
Depending on the stage, risk factors, and frontline regimen utilized, between 5% and 40% of patients with Hodgkin lymphoma can be expected to experience either primary induction failure or a relapse after attaining remission with frontline therapy.3 Primary refractory Hodgkin lymphoma, which occurs in up to 5% to 10% of patients, is defined as progression or no response during induction treatment or within 90 days of completing treatment. In cases where remission status is in question, an updated tissue biopsy is recommended. Biopsy is also recommended in cases in which new sites of disease have appeared or if relapse has occurred after a durable period of remission. Restaging is recommended at the time of relapse.
For younger patients with relapsed/refractory Hodgkin lymphoma, the standard of care in most cases is second-line (or salvage) chemotherapy followed by high-dose therapy and auto-HCT. For patients not felt to be candidates for auto-HCT, options include conventional second-line chemotherapy alone, salvage radiotherapy, novel agents such as brentuximab or immune checkpoint inhibitors, and/or participation in clinical trials.
CONVENTIONAL MULTI-AGENT CHEMOTHERAPY REGIMENS
Numerous conventional regimens have been shown in phase 2 studies to be active in relapsed and refractory Hodgkin lymphoma. These include platinum-based regimens, gemcitabine-based regimens, and alkylator-based regimens. No randomized trials in Hodgkin lymphoma have been conducted comparing these regimens. In general, regimens are chosen based on the patient’s age, performance status, comorbidities, and whether auto-HCT is being considered.
In the United States, platinum-based regimens such as ICE (ifosfamide, carboplatin, etoposide),42 DHAP (dexamethasone, cisplatin, high-dose cytarabine),43 ESHAP (etoposide, methylprednisolone, high-dose cytarabine, cisplatin),44 GDP (gemcitabine, cisplatin, dexamethasone),45 and GCD (gemcitabine, carboplatin, dexamethasone)46 are all considered appropriate second-line therapy options for patients being considered for auto-HCT, due to their high response rates and because autologous hematopoietic stem cell collection remains feasible after these regimens. Response rates range from 60% to 88%, with CR rates between 17% and 41%, and toxic death rates generally well below 5%.
Other gemcitabine-based regimens such as IGEV (ifosfamide, gemcitabine, vinorelbine) and GVD (gemcitabine, vinorelbine, liposomal doxorubicin) are also effective.47,48 GVD is an excellent choice since it is a generally well-tolerated outpatient regimen with a 60% response rate even in heavily pretreated patients.48 Stem cell collection remains feasible after both IGEV and GVD as well. ABVD can produce CR in approximately 20% to 50% of patients initially treated with MOPP.49–51 In practice, however, most patients today with relapsed or refractory Hodgkin lymphoma have already received ABVD as part of their first-line therapy, and retreatment with ABVD is not a good option because it would be associated with prohibitively high cumulative doses of doxorubicin.
These multi-agent chemotherapy regimens may not be tolerated well in patients over age 65 to 70 years or those with significant underlying comorbidities. In recent years, bendamustine has emerged as one of the most active conventional agents for cHL, with overall response rates of 53% to 58% in heavily pre-treated patients.52,53 Bendamustine can generally be tolerated even in elderly patients as well.
Some centers, particularly in Europe, investigated aggressive salvage regimens such as mini-BEAM (carmustine, etoposide, cytarabine, melphalan)54 or dexa-BEAM (BEAM plus dexamethasone).55 These regimens, however, are associated with significant hematologic toxicity and high (2%–5%) treatment-related mortality. As a result, these are rarely used in the United States.
For patients who have progressed after (or are not candidates for) platinum- and/or gemcitabine-based therapy, older alkylator-based regimens such as MOPP, C-MOPP, or ChlVPP (chlorambucil, vinblastine, procarbazine, prednisone) can be considered.56–58 However, these regimens are associated with significant bone marrow suppression, and autologous hematopoietic stem cell collection may no longer be feasible after such regimens. Therefore, these regimens should only be given to patients not felt to be auto-HCT candidates, or patients for whom autologous hematopoietic stem cell collection has already been completed. Weekly vinblastine or single-agent gemcitabine are palliative chemotherapy options, with response rates in the 60% to 80% range. Patients can sometimes be maintained on such low-intensity palliative regimens for 6 to 12 months or longer.59,60
BRENTUXIMAB VEDOTIN
Several trials are evaluating incorporation of brentuximab into second-line therapy in transplant-eligible patients. These approaches have used brentuximab prior to, concurrent with, or following platinum-based chemotherapy.61 While there is currently no consensus on the optimal way to incorporate brentuximab into salvage therapy, it is possible that the use of brentuximab or other novel agents in salvage therapy may allow for avoidance of conventional chemotherapy in some patients. In addition, this may translate into more patients proceeding to auto-HCT in a PET negative state. PET negativity prior to auto-HCT is a powerful predictor of long-term remission after auto-HCT, so any intervention that increases the rate of PET negativity prior to auto-HCT would be expected to improve outcomes with auto-HCT.62–65
For patients not being considered for autoHCT, or those for whom platinum-based salvage therapy was ineffective, single-agent brentuximab is an excellent option. In 2 phase 2 studies, an overall response rate (ORR) of 60% to 75% (including a CR rate of 22%–34%) was seen in relapsed and refractory Hodgkin lymphoma patients.66 The US Food and Drug Administration (FDA) approved brentuximab vedotin in August 2011 for treatment of relapsed and refractory Hodgkin lymphoma, after a failed auto-HCT, or in patients who are not auto-HCT candidates and who have received at least 2 prior chemotherapy regimens. With more extended follow-up, it has become clear that a proportion of patients who achieve CR to brentuximab may maintain remission long-term—58% at 3 years and 38% at 5 years.67 These patients may in fact be cured, in many cases without having undergone allogeneic HCT (allo-HCT) after brentuximab.
PD-1 (IMMUNE CHECKPOINT) INHIBITORS
As discussed earlier, PD-L1/PD-L2 copy number alterations represent a disease-defining feature of cHL. Alterations in chromosome 9p24.1 increase the expression of PD-1 ligands PD-L1 and PD-L2. Nivolumab and pembrolizumab are PD-1-blocking antibodies, which have recently been FDA approved for relapsed and refractory cHL. In a study with 23 patients, with 78% of them relapsing after auto-HCT and 78% relapsing after brentuximab, nivolumab produced an objective response in 87% of the patients, with 17% achieving CR and 70% achieving PR. The rate of PFS was 86% at 24 weeks.68 Pembrolizumab, another PD-1 antagonist, was also tested in relapsed and refractory Hodgkin lymphoma. In the KEYNOTE-087 study (n = 210), pembrolizumab produced an ORR of 64% to 70% in 3 different cohorts of relapsed and refractory cHL patients. Overall CR rate was 22%.69 In general, these agents are well tolerated, although patients must be monitored closely for
inflammatory/autoimmune-type toxicities including skin rash, diarrhea/colitis, transaminitis, endocrine abnormalities, and pneumonitis. Prompt recognition and initiation of corticosteroids is essential in managing these toxicities. Of note, PD-1 inhibitors should be given very cautiously to patients with a prior history of allo-HCT, since 30% to 55% of such patients will experience acute graft-versus-host disease (GVHD) in this setting. In 2 retrospective studies, the response rate was very high at 77% to 95%; however, 10% to 26% of all patients treated with PD-1 inhibitors post-allo-HCT died from GVHD induced by PD-1 inhibition.70,71 These risks and benefits therefore need to be carefully weighed in the post-allo-HCT setting. In another recent study, the outcomes were reported for 39 patients who underwent allo-HCT after prior therapy with a PD-1 inhibitor. Three patients (7.7%) developed lethal acute GVHD, suggesting there may be an increased risk of GVHD in patients undergoing allo-HCT after prior PD-1 inhibitor therapy.72
AUTOLOGOUS STEM CELL TRANSPLANTATION
Several studies have shown an improved disease-free survival (DFS) or FFS in patients with relapsed cHL treated by auto-HCT as compared to those receiving conventional chemotherapy alone.55,73,74 Overall, for relapsed disease, one can expect an approximately 50% to 60% chance for DFS at 5 years post-transplant. In a retrospective, matched-pair analysis, FFP was 62% for auto-HCT patients, compared to 32% for conventional chemotherapy patients. OS, however, was similar for the 2 groups (47%–54%). Patients failing induction therapy or relapsing within 1 year were seen to benefit the most from auto-HCT, including an OS benefit.74
A European prospective randomized trial was conducted comparing conventional salvage therapy to auto-HCT. In this study, 161 patients with relapsed Hodgkin lymphoma were treated with 2 cycles of dexa-BEAM. Those with chemo-sensitive disease were then randomized to either 2 more cycles of dexa-BEAM or high-dose BEAM with auto-HCT. Auto-HCT was associated with an approximately 55% FFTF at 3 years, versus 34% with conventional chemotherapy alone.55 This benefit again was most apparent for patients relapsing within 1 year of completion of primary therapy, although an OS benefit was not seen with auto-HCT. For patients with late relapse (>1 year after completion of primary therapy), auto-HCT was associated with an approximately 75% FFTF at 3 years, versus 50% with chemotherapy alone. One other small randomized trial of auto-HCT in relapsed and refractory Hodgkin lymphoma also showed an improved 3-year EFS in favor of auto-HCT (53% versus 10%), again with no difference in OS.73
The lack of OS benefit seen in these studies suggests that auto-HCT at first or second relapse provides comparable outcomes. Auto-HCT offers the benefit of avoiding the long-term toxicities associated with multiple salvage regimens and the anxiety associated with multiple relapses. In addition, the treatment-related mortality with auto-HCT is now in the 1% to 2% range in younger patients, at centers that perform the procedure routinely. For all of these reasons, auto-HCT is commonly recommended by physicians for Hodgkin lymphoma patients in first or second relapse. In most cases, transplant is favored in first relapse, since waiting until second relapse may be associated with a lower chance of achieving CR and difficulty collecting sufficient hematopoietic stem cells. For patients with early relapse or primary refractory disease, an even stronger case can be made for auto-HCT as the best option to achieve sustained control of the disease. For patients with late relapse, conventional salvage therapy alone may be a reasonable option, particularly in older or frail patients, or those with significant comorbid conditions.
The optimal conditioning regimen for autoHCT for relapsed and refractory Hodgkin lymphoma remains undefined. No randomized trials have been performed comparing conditioning regimens for relapsed and refractory Hodgkin lymphoma. One retrospective study compared 92 patients with Hodgkin lymphoma who underwent auto-HCT using a total-body irradiation (TBI) regimen versus a chemotherapy-alone regimen. No difference in 5-year OS or EFS was seen.75 Given the lack of benefit seen with TBI, along with reports of increased rates of secondary malignancies and myelodysplasia with TBI,76 chemotherapy-alone conditioning regimens are most widely employed. For example, in the United States, either the BEAM or CBV (cyclophosphamide, carmustine, etoposide) regimens are used in over 80% of cases.77 This practice was justified in a Center for International Blood and Marrow Transplant Research (CIBMTR) retrospective study comparing outcomes by conditioning regimens, in which no regimen performed better than BEAM or CBV.78
IFRT is often given as an adjunctive therapy to sites of initial and/or relapsed disease following auto-HCT. Although a relatively common practice, whether this truly enhances outcomes beyond that obtained with auto-HCT alone is unclear. Two retrospective studies have shown some benefit in terms of improvement in OS at 3 to 5 years in the group that received IFRT (70%–73% versus 40%–56%).79,80 Given the retrospective nature and small size of these studies, a prospective study would be needed to properly define the potential role for IFRT following auto-HCT in relapsed/refractory Hodgkin lymphoma. Another retrospective study (n = 73) that evaluated peri-transplant IFRT in Hodgkin lymphoma patients receiving auto transplant found no improvement in survival for patients who received peri-transplant IFRT. This study, however, did show a survival benefit in the subgroup of patients with limited stage disease.81
Prognostic Factors Associated with Outcome with Auto-HCT
The factor most consistently associated with improved outcome for patients with relapsed and refractory Hodgkin lymphoma who undergo auto-HCT is the disease status at transplant.63,77 Those in a second CR, versus a chemo-sensitive relapse (but not CR), versus a chemo-refractory relapse have DFS rates of 60% to 70%, 30% to 40%, and 10% to 20%, respectively.63 The duration between remission and relapse also has important prognostic significance. Late relapse (> 1 year after completion of frontline therapy) is associated with better outcomes as compared to early relapse.55 Other factors with prognostic significance at relapse include anemia, time to relapse and clinical stage, B symptoms, extranodal disease, number of prior chemotherapy regimens, and performance status.42,82 The prognostic impact of pretransplant disease status has been confirmed by studies using functional imaging (eg, FDG-PET or gallium scans). In a report by Moskowitz et al, patients with negative functional imaging following second-line therapy had a 77% EFS post-auto-HCT versus 33% in those whose functional imaging remained positive.62 Very similar findings have been reported by other groups.63–65
Post-Auto-HCT Brentuximab Maintenance
In the multicenter, randomized, double-blinded phase 3 AETHERA trial (n = 329), brentuximab (n = 165) was compared with placebo (n = 164) in patients with unfavorable risk relapsed or primary refractory cHL who had undergone autologous transplant. Eligible patients had at least 1 of the following risk factors for progression after auto-HCT: primary refractory Hodgkin lymphoma (failure to achieve complete remission), relapsed Hodgkin lymphoma with an initial remission duration of less than 12 months, or extranodal involvement at the start of pre-transplantation salvage chemotherapy. Patients were required to have CR, PR, or stable disease after pretransplant salvage chemotherapy with adequate kidney, liver, and bone marrow function. Patients who previously received brentuximab were excluded. Patients received 16 cycles of brentuximab or placebo once every 3 weeks starting 30 to 45 days after transplant. The PFS was significantly improved in the brentuximab group when compared to the placebo group (hazard ratio 0.57; P = 0.0013) after a median observation time of 30 months. Median PFS was 42.9 months in the brentuximab group versus 24.1 months in the placebo group; estimated 2-year PFS rates were 63% in the brentuximab group and 51% in the placebo group. OS was not significantly different between the study groups (~85%), presumably due to the fact that patients in the control group who relapsed likely went on to receive brentuximab as a subsequent therapy.83
PRIMARY REFRACTORY HODGKIN LYMPHOMA
Patients with primary refractory Hodgkin lymphoma have a poor outcome. Salvage therapy using conventional chemotherapy and/or RT results in long-term DFS in 10% or fewer of such patients.13,84 Given these poor outcomes with conventional salvage therapy, auto-HCT is considered to be the standard of care for this subset of patients. The GHSG retrospectively analyzed the prognostic factors and outcomes of patients with primary refractory Hodgkin lymphoma. The 5-year freedom-from-second-failure and the 5-year OS were reported to be 31% and 43%, respectively, for those patients treated with auto-HCT. Patients with poor functional status at time of transplant, age greater than 50 years, and failure to attain a temporary remission had a 0% 5-year OS, as compared to 55% in patients without any of these risk factors.85 A large retrospective European study showed that patients with chemo-resistant disease who underwent transplant had a 19% survival at 5 years.63 Hence, even patients with primary refractory Hodgkin lymphoma have some chance of achieving long-term survival following auto-HCT.
SALVAGE RADIOTHERAPY
The GHSG performed a retrospective analysis of the efficacy of salvage RT in patients with refractory or first-relapsed Hodgkin lymphoma. Five-year FFTF and OS rates were 28% and 51%, respectively. Patients with a limited-stage relapse and without B symptoms were more likely to benefit from salvage RT.86 Campbell et al reported on 81 patients undergoing salvage RT for persistent or recurrent Hodgkin lymphoma after chemotherapy. The 10-year FFTF and OS rates were 33% and 46%, respectively.87 Similarly, Wirth et al reported a 5-year FFS of 26% and 5-year OS of 57%. These figures were 36% and 75%, respectively, in patients whose relapse was limited to supradiaphragmatic nodal sites without B symptoms.88 RT therefore may be a useful strategy for a subset of patients who relapse following chemotherapy, particularly those with a limited-stage relapse, without B symptoms, and those with relapsed disease after a CR, as opposed to those with a partial response or lack of response to the prior chemotherapy regimen.
INVESTIGATIONAL AGENTS AND NOVEL COMBINATIONS
Several biological therapies are emerging as options for the treatment of refractory or relapsed disease. These therapies consist of monoclonal antibodies and ADCs that target cell surface antigens, or small molecules that inhibit key intracellular pathways within neoplastic cells.
Rituximab
Rituximab is a chimeric anti-CD20 monoclonal antibody used widely in B-cell non-Hodgkin lymphomas. The CD20 molecule is typically highly expressed in nodular lymphocyte-predominant Hodgkin lymphoma (NLPHL). Two studies (one in relapsed patients, the other in a mixture of relapsed and previously untreated patients) showed significant activity of rituximab in relapsed NLPHL, with ORRs ranging from 94% to 100%, CR rates ranging from 41% to 53%, and median duration of remission in the 10- to 33-month range.89,90 In cHL, CD20 is expressed in HRS cells in 20% to 30% of cases. In such cases, single-agent rituximab has also shown activity. There is also evidence that rituximab may be effective even in cases in which the HRS cells are CD20-negative, presumably by virtue of depleting reactive B lymphocytes from the microenvironment, which may enhance anti-tumor immunity, or by eliminating a putative CD20-expressing Hodgkin lymphoma stem cell.91,92
Lenalidomide
Lenalidomide is an immunomodulatory drug that has multiple modes of action, including direct induction of apoptosis in tumor cells, antiangiogenic effects, and the activation of immune cells, such as natural killer cells and T cells. Lenalidomide has been shown to modify many features of the microenvironment of HRS cells and has demonstrated activity in other B-cell neoplasms. As a result, lenalidomide has been evaluated in relapsed and refractory Hodgkin lymphoma patients. A multicenter phase 2 study by Fehniger et al included 35 patients, 87% of whom had previously undergone HCT and 55% of whom were refractory to the last therapy.93 All patients were given lenalidomide 25 mg/day from days 1 to 21 of a 28-day cycle until disease progression. One patient was noted to achieve CR, 6 achieved PR, and 5 had stable disease lasting more than 6 months, for an ORR of 19% and a “cytostatic overall response rate” of 33%. The median duration of CR/partial remission was 6 months, with the median time-to-treatment failure in responders (including those with stable disease > 6 months) being 15 months. Similarly, in another study, Böll et al evaluated 12 patients across 4 German centers with relapsed or refractory disease who were treated with oral lenalidomide for 21 days in a 28-day cycle. No radiological evidence of disease progression after 2 cycles of lenalidomide was seen in any of the enrolled patients. ORR was noted to be 50%, with 6 patients with stable disease and 5 patients achieving PR after 2 cycles.94
Novel Brentuximab Combination Therapies
Brentuximab plus bendamustine. The combination of brentuximab and bendamustine was tested as an outpatient regimen in a phase 1/2 study (n = 55) in primary refractory Hodgkin lymphoma or after first relapse. The CR rate of the combination was 74%, with an overall objective response (CR + PR) of 93%. The CR rates were 64% and 84%, respectively, for refractory and relapsed patients. The PFS at 12 months was 80%, establishing this combination therapy as an effective salvage regimen with durable response.95
Brentuximab plus nivolumab. Preliminary results have recently been presented from 2 studies96,97 evaluating the combination of brentuximab and nivolumab. While this combination would still be considered investigational, these studies showed very encouraging ORRs of 90% to 100% and a CR rate of 62% to 66%. Longer follow-up is needed to determine whether these responses are durable and to document the toxicity profile of this combination.
Mammalian Target of Rapamycin Inhibitors
Two mammalian target of rapamycin (mTOR) inhibitors, everolimus and temsirolimus, are currently available in the United States. While neither drug currently has FDA approval for Hodgkin lymphoma, everolimus was evaluated in a phase 2 trial in a heavily pretreated group of relapsed/refractory patients. An ORR of 47% was seen, with a median time to progression of 7.2 months.98
ALLOGENEIC STEM CELL TRANSPLANTATION
Historically, patients who relapse after having an auto-HCT generally had a poor outcome, with a median survival of 2 to 3 years after failure of auto-HCT.99 These patients may be offered palliative chemotherapy (see above), treatment with novel agents (see above), or enrollment in a clinical trial. Select patients may benefit from a second hematopoietic stem cell transplant, most commonly an allo-HCT. However, rare patients with late relapse after auto-HCT may be considered for a second auto-HCT, with a minority of such patients achieving a durable remission after the second auto-HCT.100,101 Because relapse or progressive disease occurs most commonly in the first several months following auto-HCT, patients are more often considered for allo-HCT than a second auto-HCT. In addition, a second auto-HCT may not be feasible due to impaired bone marrow reserve and/or concerns for development of secondary myelodysplasia or acute myeloid leukemia.
Several studies have evaluated allo-HCT in relapsed/ refractory Hodgkin lymphoma. Early studies evaluating myeloablative allo-HCT for Hodgkin lymphoma showed excessive treatment-related mortality (up to 50%) and disappointingly low rates of long-term survival (< 25%).102,103 This was likely related to the fact that, in that era, most of the patients with Hodgkin lymphoma evaluated for allo-HCT were heavily pretreated and therefore at a higher risk for toxicity as well as lymphoma progression.
More recently, several studies have focused on the use of reduced-intensity conditioning (RIC) allo-HCT for relapsed and refractory Hodgkin lymphoma. This approach relies more on a “graft-versus-lymphoma” effect, the existence of which has been debated in Hodgkin lymphoma. Three single-center studies of RIC allo-HCT in patients with multiply recurrent Hodgkin lymphoma showed improved rates of treatment-related mortality (8%–16%) but still relatively low rates of long-term PFS (23%–39% at 2 to 4 years).104–106 Interestingly, in one of these studies the outcomes were more favorable for patients who underwent haploidentical (versus matched sibling or matched unrelated donor) transplants.105
Two large registry studies have also reported on the outcomes of RIC allo-HCT in patients with relapsed and refractory Hodgkin lymphoma.107,108 These studies also confirmed a modest improvement in outcomes compared with those seen historically with myeloablative transplants. Treatment-related mortality at 1 to 2 years was 23% to 33%, depending on whether a matched sibling donor versus an unrelated donor was used. However, long-term PFS (18%–20% at 2 to 5 years) and OS (28%–37% at 2 to 5 years) remained poor, primarily due to high rates of progressive lymphoma post-transplant. In both of these studies, patients were heavily pretreated (84%–96% had received 3 or more prior lines of chemotherapy, and 62%–89% received a prior auto-HCT), with 47% to 55% of patients chemo-resistant prior to transplant. Of note, both of these registry studies reflect patients who underwent transplant prior to the widespread use of brentuximab and PD-1 inhibitors.
Based on the single-center and registry data above, a prospective multicenter European phase 2 trial was conducted to evaluate the benefit of RIC allo-HCT in Hodgkin lymphoma.109 Ninety-two patients (86% with prior auto-HCT, 90% with 3 or more prior lines of therapy) were enrolled and given salvage therapy. Those who had stable disease or better following salvage therapy remained on protocol (n = 78) and underwent RIC with fludarabine and melphalan, followed by allo-HCT (70% with matched sibling donors). Treatment-related mortality was 15% at 1 year. Relapse or progression occurred in 49% at 2 years (35% if chemo-sensitive prior to transplant). Chronic GVHD was associated with a decreased rate of relapse, supporting the existence of a graft-versus-lymphoma effect in Hodgkin lymphoma. Unfortunately, PFS among all allografted patients was still relatively poor (24% at 4 years). However, among patients in CR prior to allo-HCT, a 50% PFS was seen at 4 years. Therefore, even in a prospective multicenter study, RIC allo-HCT offered significant benefit with manageable toxicity in relapsed and refractory Hodgkin lymphoma patients with chemo-sensitive disease.
These studies suggest that outcomes with allo-HCT would improve further if implemented earlier in the course of disease and/or with a lower burden of disease at transplant. It has therefore been suggested that allo-HCT should be considered soon after failure of auto-HCT is documented. In a retrospective study by Sarina et al, 185 Hodgkin lymphoma patients who relapsed following auto-HCT were then immediately considered for reduced-intensity allo-HCT.110 Of these, 122 had a donor identified, and 104 (85%) actually underwent allo-HCT. These 104 patients were then compared to the other 81 patients who either had no donor identified or had a donor but did not receive the planned allo-HCT. Two-year PFS and OS were superior in the patients undergoing allo-HCT (39% versus 14% and 66% versus 42%, respectively, P < 0.001), with a median follow-up of 4 years. The presence of chronic GVHD again was associated with improved PFS and OS. Disease status prior to transplant remained highly predictive of PFS and OS by multivariate analysis. Two other smaller retrospective studies similarly found a survival benefit associated with allo-HCT compared with patients who underwent conventional salvage therapies alone.111,112 These studies, although subject to the usual limitations of retrospective analyses, suggest that the results with reduced-intensity allo-HCT are in fact enhanced if applied earlier in the disease course, and are superior to those with conventional therapy alone.
Currently, the exact role of allo-HSCT, including the optimal timing and optimal donor source (matched sibling versus haploidentical sibling versus matched unrelated donor), remain undefined for relapsed and refractory Hodgkin lymphoma. As discussed earlier, brentuximab is highly active in relapsed Hodgkin lymphoma patients, with a subset of patients still in CR at 5 years.67 For such patients, avoiding the risks of allo-HCT is a desirable goal.
For those who relapse or progress after auto-HCT, a reasonable strategy therefore is to treat initially with brentuximab, unless the patient is already known to have responded poorly to brentuximab, or already has significant neuropathy. Those who achieve a CR to brentuximab are then observed. A subset of those patients will remain in remission at 5 years without further therapy. For those who relapse, or who achieve less than a CR to brentuximab, additional treatment (with brentuximab re-treatment being one option) followed by a reduced-intensity allo-HCT is a reasonable consideration. This approach has the theoretical advantages of (1) avoiding the risk of allo-HCT in the subset potentially cured by brentuximab, (2) getting patients to allo-HCT with fewer comorbidities (due to a lower total exposure to conventional chemotherapy pre-transplant), and (3) applying allo-HCT in the setting of sensitive disease/lower disease burden (due to the high efficacy of brentuximab). The results of a small study suggest that brentuximab may in fact be a very effective “bridge” to allotransplant. Chen et al113 reported on 18 patients with relapsed/refractory Hodgkin lymphoma (17 of whom had previously undergone auto-HCT) who were treated on brentuximab vedotin clinical trials. The data were retrospectively evaluated to determine the efficacy and safety of subsequent reduced-intensity allo-HCT. Remarkably, at 1 year the OS was 100%, PFS was 92%, and nonrelapse mortality was 0% with a median follow-up of 14 months. Hence, brentuximab is safe for use prior to reduced-intensity allo-HCT in heavily pre-treated patients and appears to be associated with very favorable post-transplant outcomes, particularly in comparison to older studies of allo-HCT in the era prior to brentuximab.
SUMMARY
Currently, cure is possible for the majority of patients diagnosed with advanced stage Hodgkin lymphoma. The challenge to the clinician is to provide curative treatment with the lowest risk of serious toxicities. Which regimen will best provide this balance of risk and benefit needs to be assessed based on the relapse risk, age, frailty, and comorbidity profile for an individual patient. For many patients with relapsed or refractory Hodgkin lymphoma, cure remains possible using approaches based on hematopoietic stem cell transplantation, RT, and/or brentuximab. In addition, there are now numerous conventional chemotherapy agents, RT strategies, and exciting newer agents such as PD-1 inhibitors, that can provide significant clinical benefit even when cure is not feasible.
1. Kuppers R, Rajewsky K, Zhao M, et al. Hodgkin disease: Hodgkin and Reed-Sternberg cells picked from histological sections show clonal immunoglobulin gene rearrangements and appear to be derived from B cells at various stages of development. Proc Natl Acad Sci U S A 1994;91:10962–6.
2. Kuppers R. The biology of Hodgkin’s lymphoma. Nature Rev Cancer 2009;9:15–27.
3. Narra RK, Pingali SR, Fenske TS. Early-stage Hodgkin’s lymphoma. Hospital Physician Hematology-Oncology Board Review Manual. 2017;12(Part 3).
4. Mauch PM, Kalish LA, Kadin M, et al. Patterns of presentation of Hodgkin disease. Implications for etiology and pathogenesis. Cancer 1993;71:2062–71.
5. Hasenclever D, Diehl V. A prognostic score for advanced Hodgkin’s disease. International Prognostic Factors Project on Advanced Hodgkin’s Disease. N Engl J Med 1998;339:1506–14.
6. Moccia AA, Donaldson J, Chhanabhai M, et al. International Prognostic Score in advanced-stage Hodgkin’s lymphoma: altered utility in the modern era. J Clin Oncol 2012;30:3383–8.
7. Diefenbach CS, Li H, Hong F, et al. Evaluation of the International Prognostic Score (IPS-7) and a Simpler Prognostic Score (IPS-3) for advanced Hodgkin lymphoma in the modern era. Br J Haematol 2015;171:530–8.
8. Greaves P, Clear A, Coutinho R, et al. Expression of FOXP3, CD68, and CD20 at diagnosis in the microenvironment of cassical hodgkin lymphoma is predictive of outcome. J Clin Oncol 2013;31:256–62.
9. Touati M, Delage-Corre M, Monteil J, et al. CD68-positive tumor-associated macrophages predict unfavorable treatment outcomes in classical Hodgkin lymphoma in correlation with interim fluorodeoxyglucose-positron emission tomography assessment. Leuk Lymphoma 2015;56:332–41.
10. Roemer MG, Advani RH, Ligon AH, et al. PD-L1 and PD-L2 genetic alterations define classical Hodgkin lymphoma and predict outcome. J Clin Oncol 2016;34:2690–7.
11. Pike LC, Kirkwood AA, Patrick P, et al. Can baseline PET-CT features predict outcomes in advanced hodgkin lymphoma? A prospective evaluation of UK patients in The RATHL Trial (CRUK/07/033). Hematological Oncology 2017;35(Supplement S2):37–8.
12. DeVita VT Jr, Simon RM, Hubbard SM, et al. Curability of advanced Hodgkin’s disease with chemotherapy. Long-term follow-up of MOPP-treated patients at the National Cancer Institute. Ann Intern Med 1980;92:587–95.
13. Longo DL, Duffey PL, Young RC, et al. Conventional-dose salvage combination chemotherapy in patients relapsing with Hodgkin’s disease after combination chemotherapy: the low probability for cure. J Clin Oncol 1992;10:210–8.
14. Kaldor JM, Day NE, Clarke EA, et al. Leukemia following Hodgkin’s disease. N Engl J Med 1990;322:7–13.
15. Bonadonna G, Zucali R, Monfardini S, et al. Combination chemotherapy of Hodgkin’s disease with adriamycin, bleomycin, vinblastine, and imidazole carboxamide versus MOPP. Cancer 1975;36:252–9.
16. Canellos GP, Anderson JR, Propert KJ, et al. Chemotherapy of advanced Hodgkin’s disease with MOPP, ABVD, or MOPP alternating with ABVD. N Engl J Med 1992;327:1478–84.
17. Horning SJ, Williams J, Bartlett NL, et al. Assessment of the stanford V regimen and consolidative radiotherapy for bulky and advanced Hodgkin’s disease: Eastern Cooperative Oncology Group pilot study E1492. J Clin Oncol 2000;18:972–80.
18. Hoskin PJ, Lowry L, Horwich A, et al. Randomized comparison of the stanford V regimen and ABVD in the treatment of advanced Hodgkin’s Lymphoma: United Kingdom National Cancer Research Institute Lymphoma Group Study ISRCTN 64141244. J Clin Oncol 2009;27:5390–6.
19. Gordon LI, Hong F, Fisher RI, et al. Randomized phase III trial of ABVD versus Stanford V with or without radiation therapy in locally extensive and advanced-stage Hodgkin lymphoma: an intergroup study coordinated by the Eastern Cooperative Oncology Group (E2496). J Clin Oncol 2013;31:684–91.
20. Engert A, Diehl V, Franklin J, et al. Escalated-dose BEACOPP in the treatment of patients with advanced-stage Hodgkin’s lymphoma: 10 years of follow-up of the GHSG HD9 study. J Clin Oncol 2009;27:4548–54.
21. Merli F, Luminari S, Gobbi PG, et al. Long-term results of the HD2000 trial comparing ABVD versus BEACOPP versus COPP-EBV-CAD in untreated patients with advanced Hodgkin lymphoma: a study by Fondazione Italiana Linfomi. J Clin Oncol 2016;34:1175–81.
22. Viviani S, Zinzani PL, Rambaldi A, et al. ABVD versus BEACOPP for Hodgkin’s lymphoma when high-dose salvage is planned. N Engl J Med 2011;365:203–12.
23. Carde P, Karrasch M, Fortpied C, et al. Eight cycles of ABVD versus four cycles of BEACOPPescalated plus four cycles of BEACOPPbaseline in stage III to IV, International Prognostic Score >/= 3, high-risk Hodgkin lymphoma: first results of the phase III EORTC 20012 intergroup trial. J Clin Oncol 2016;34:2028–36.
24. National Comprehensive Cancer Network I. NCCN Guidelines Version 1.2017 Hodgkin Lymphoma. Accessed July 20, 2017.
25. Younes A, Connors JM, Park SI, et al. Brentuximab vedotin combined with ABVD or AVD for patients with newly diagnosed Hodgkin’s lymphoma: a phase 1, open-label, dose-escalation study. Lancet Oncology 2013;14:1348–56.
26. Takeda and Seattle Genetics announce positive results from phase 3 ECHELON-1 clinical trial evaluating ADCETRIS® (brentuximab vedotin) in frontline advanced Hodgkin lymphoma [press release]. Cambridge, MA: Takeda Oncology; June 26, 2017.
27. Boll B, Bredenfeld H, Gorgen H, et al. Phase 2 study of PVAG (prednisone, vinblastine, doxorubicin, gemcitabine) in elderly patients with early unfavorable or advanced stage Hodgkin lymphoma. Blood 2011;118:6292–8.
28. Behringer K, Goergen H, Hitz F, et al. Omission of dacarbazine or bleomycin, or both, from the ABVD regimen in treatment of early-stage favourable Hodgkin’s lymphoma (GHSG HD13): an open-label, randomised, non-inferiority trial. Lancet 2015;385:1418–27.
29. Forero-Torres A, Holkova B, Goldschmidt J, et al. Phase 2 study of frontline brentuximab vedotin monotherapy in Hodgkin lymphoma patients aged 60 years and older. Blood 2015;126:2798–804.
30. Gallamini A, Hutchings M, Rigacci L, et al. Early interim 2-[18F]fluoro-2-deoxy-D-glucose positron emission tomography is prognostically superior to international prognostic score in advanced-stage Hodgkin’s lymphoma: a report from a joint Italian-Danish study. J Clin Oncol 2007;25:3746–52.
31. Meignan M, Gallamini A, Meignan M, et al. Report on the First International Workshop on Interim-PET-Scan in Lymphoma. Leuk Lymphoma 2009;50:1257–60.
32. Barrington SF, Mikhaeel NG, Kostakoglu L, et al. Role of Imaging in the Staging and Response Assessment of Lymphoma: Consensus of the International Conference on Malignant Lymphomas Imaging Working Group. J Clin Oncol 2014;32:3048–58.
33. Press OW, Li H, Schoder H, et al. US Intergroup trial of response-adapted therapy for stage III to IV Hodgkin lymphoma using early interim fluorodeoxyglucose-positron emission tomography imaging: Southwest Oncology Group S0816. J Clin Oncol 2016;34:2020–7.
34. Zinzani PL, Broccoli A, Gioia DM, et al. Interim positron emission tomography response-adapted therapy in advanced-stage Hodgkin lymphoma: final results of the pPhase II part of the HD0801 study. J Clin Oncol 2016;34:1376–85.
35. Borchmann P, Haverkamp H, Lohri A, et al. Progression-free survival of early interim PET-positive patients with advanced stage Hodgkin’s lymphoma treated with BEACOPPescalated alone or in combination with rituximab (HD18): an open-label, international, randomised phase 3 study by the German Hodgkin Study Group. Lancet Oncology 2017;18:454–63.
36. Aleman BM, Raemaekers JM, Tirelli U, et al. Involved-field radiotherapy for advanced Hodgkin’s lymphoma. N Engl J Med 2003;348:2396–406.
37. Borchmann P, Haverkamp H, Diehl V, et al. Eight cycles of escalated-dose BEACOPP compared with four cycles of escalated-dose BEACOPP followed by four cycles of baseline-dose BEACOPP with or without radiotherapy in patients with advanced-stage hodgkin’s lymphoma: final analysis of the HD12 trial of the German Hodgkin Study Group. J Clin Oncol 2011;29:4234–42.
38. Engert A, Haverkamp H, Kobe C, et al. Reduced-intensity chemotherapy and PET-guided radiotherapy in patients with advanced stage Hodgkin’s lymphoma (HD15 trial): a randomised, open-label, phase 3 non-inferiority trial. Lancet 2012;379:1791–9.
39. Nademanee A, Molina A, Fung H, et al. High-dose chemo/radiotherapy and autologous bone marrow or stem cell transplantation for poor-risk advanced-stage Hodgkin’s disease during first partial or complete remission. Biol Blood Marrow Transplant 1999;5:292–8.
40. Federico M, Bellei M, Brice P, et al. High-dose therapy and autologous stem-cell transplantation versus conventional therapy for patients with advanced Hodgkin’s lymphoma responding to front-line therapy. J Clin Oncol 2003;21:2320–5.
41. Arakelyan N, Berthou C, Desablens B, et al. Early versus late intensification for patients with high-risk Hodgkin lymphoma-3 cycles of intensive chemotherapy plus low-dose lymph node radiation therapy versus 4 cycles of combined doxorubicin, bleomycin, vinblastine, and dacarbazine plus myeloablative chemotherapy with autologous stem cell transplantation: five-year results of a randomized trial on behalf of the GOELAMS Group. Cancer 2008;113:3323–30.
42. Moskowitz CH, Nimer SD, Zelenetz AD, et al. A 2-step comprehensive high-dose chemoradiotherapy second-line program for relapsed and refractory Hodgkin disease: analysis by intent to treat and development of a prognostic model. Blood 2001;97:616–23.
43. Josting A, Rudolph C, Reiser M, et al. Time-intensified dexamethasone/cisplatin/cytarabine: an effective salvage therapy with low toxicity in patients with relapsed and refractory Hodgkin’s disease. Ann Oncol 2002;13:1628–35.
44. Aparicio J, Segura A, Garcera S, et al. ESHAP is an active regimen for relapsing Hodgkin’s disease. Ann Oncol 1999;10:593–5.
45. Baetz T, Belch A, Couban S, et al. Gemcitabine, dexamethasone and cisplatin is an active and non-toxic chemotherapy regimen in relapsed or refractory Hodgkin’s disease: a phase II study by the National Cancer Institute of Canada Clinical Trials Group. Ann Oncol 2003;14:1762–7.
46. Gopal AK, Press OW, Shustov AR, et al. Efficacy and safety of gemcitabine, carboplatin, dexamethasone, and rituximab in patients with relapsed/refractory lymphoma: a prospective multi-center phase II study by the Puget Sound Oncology Consortium. Leuk Lymphoma 2010;51:1523–9.
47. Santoro A, Magagnoli M, Spina M, et al. Ifosfamide, gemcitabine, and vinorelbine: a new induction regimen for refractory and relapsed Hodgkin’s lymphoma. Haematologica 2007;92:35–41.
48. Bartlett NL, Niedzwiecki D, Johnson JL, et al. Gemcitabine, vinorelbine, and pegylated liposomal doxorubicin (GVD), a salvage regimen in relapsed Hodgkin’s lymphoma: CALGB 59804. Ann Oncol 2007;18:1071–9.
49. Santoro A, Bonadonna G. Prolonged disease-free survival in MOPP-resistant Hodgkin’s disease after treatment with adriamycin, bleomycin, vinblastine and dacarbazine (ABVD). Cancer Chemother Pharmacol 1979;2:101–5.
50. Krikorian JG, Portlock CS, Rosenberg SA. Treatment of advanced Hodgkin’s disease with adriamycin, bleomycin, vinblastine, and imidazole carboxamide (ABVD) after failure of MOPP therapy. Cancer 1978;41:2107–11.
51. Piga A, Ambrosetti A, Todeschini G, et al. Doxorubicin, bleomycin, vinblastine, and dacarbazine (ABVD) salvage of mechlorethamine, vincristine, prednisone, and procarbazine (MOPP)-resistant advanced Hodgkin’s disease. Cancer Treat Rep 1984;68:947–51.
52. Moskowitz AJ, Hamlin PA Jr, Perales MA, et al. Phase II study of bendamustine in relapsed and refractory Hodgkin lymphoma. J Clin Oncol 2013;31:456–60.
53. Anastasia A, Carlo-Stella C, Corradini P, et al. Bendamustine for relapsed/refractory classical Hodgkin lymphoma after high dose chemotherapy and or allogeneic transplant: a study of Fondazione Italiana Linfomi (FIL). Blood 2012;120:3652.
54. Martin A, Fernandez-Jimenez MC, Caballero MD, et al. Long-term follow-up in patients treated with Mini-BEAM as salvage therapy for relapsed or refractory Hodgkin’s disease. Br J Haematol 2001;113:161–71.
55. Schmitz N, Pfistner B, Sextro M, et al. Aggressive conventional chemotherapy compared with high-dose chemotherapy with autologous haemopoietic stem-cell transplantation for relapsed chemosensitive Hodgkin’s disease: a randomised trial. Lancet 2002;359:2065–71.
56. Fisher RI, DeVita VT, Hubbard SP, et al. Prolonged disease-free survival in Hodgkin’s disease with MOPP reinduction after first relapse. Ann Intern Med 1979;90:761–3.
57. ChlVPP therapy for Hodgkin’s disease: experience of 960 patients. The International ChlVPP Treatment Group. Ann Oncol 1995;6:167–72.
58. Selby P, Patel P, Milan S, et al. ChlVPP combination chemotherapy for Hodgkin’s disease: long-term results. Br J Cancer 1990;62:279–85.
59. Santoro A, Bredenfeld H, Devizzi L, et al. Gemcitabine in the treatment of refractory Hodgkin’s disease: results of a multicenter phase II study. J Clin Oncol 2000;18:2615–9.
60. Little R, Wittes RE, Longo DL, Wilson WH. Vinblastine for recurrent Hodgkin’s disease following autologous bone marrow transplant. J Clin Oncol 1998;16:584–8.
61. Chen R, Palmer JM, Martin P, et al. Results of a multicenter phase II trial of brentuximab vedotin as second-line therapy before autologous transplantation in relapsed/refractory Hodgkin lymphoma. Biol Blood Marrow Transplant 2015;21:2136–40.
62. Moskowitz CH, Yahalom J, Zelenetz AD, et al. High-dose chemo-radiotherapy for relapsed or refractory Hodgkin lymphoma and the significance of pre-transplant functional imaging. Br J Haematol 2010;148:890–7.
63. Sureda A, Constans M, Iriondo A, et al. Prognostic factors affecting long-term outcome after stem cell transplantation in Hodgkin’s lymphoma autografted after a first relapse. Ann Oncol 2005;16:625–33.
64. Crocchiolo R, Canevari C, Assanelli A, et al. Pre-transplant 18FDG-PET predicts outcome in lymphoma patients treated with high-dose sequential chemotherapy followed by autologous stem cell transplantation. Leuk Lymphoma 2008;49:727–33.
65. Mocikova H, Pytlik R, Markova J, et al. Pre-transplant positron emission tomography in patients with relapsed Hodgkin lymphoma. Leuk Lymphoma 2011;52:1668–74.
66. Younes A, Gopal AK, Smith SE, et al. Results of a pivotal phase II study of brentuximab vedotin for patients with relapsed or refractory Hodgkin’s lymphoma. J Clin Oncol 2012;30:2183–9.
67. Gopal AK, Chen R, Smith SE, et al. Durable remissions in a pivotal phase 2 study of brentuximab vedotin in relapsed or refractory Hodgkin lymphoma. Blood 2015;125:1236–43.
68. Ansell SM, Lesokhin AM, Borrello I, et al. PD-1 blockade with nivolumab in relapsed or refractory Hodgkin’s lymphoma.N Engl J Med 2015;372:311–9.
69. Chen R, Zinzani PL, Fanale MA, et al. Phase II study of the efficacy and safety of pembrolizumab for relapsed/refractory classic Hodgkin lymphoma. J Clin Oncol 2017;35:2125–32.
70. Haverkos BM, Abbott D, Hamadani M, et al. PD-1 blockade for relapsed lymphoma post-allogeneic hematopoietic cell transplant: high response rate but frequent GVHD. Blood 2017;130:221–8.
71. Herbaux C, Gauthier J, Brice P, et al. Efficacy and tolerability of nivolumab after allogeneic transplantation for relapsed Hodgkin lymphoma. Blood 2017;129:2471–8.
72. Merryman RW, Kim HT, Zinzani PL et al. Safety and efficacy of allogeneic hematopoietic stem cell transplant after PD-1 blockage in relapsed/refractory lymphoma. Blood 2017;129:1380–8.
73. Linch DC, Winfield D, Goldstone AH, et al. Dose intensification with autologous bone-marrow transplantation in relapsed and resistant Hodgkin’s disease: results of a BNLI randomised trial. Lancet 1993;341:1051–4.
74. Yuen AR, Rosenberg SA, Hoppe RT, et al. Comparison between conventional salvage therapy and high-dose therapy with autografting for recurrent or refractory Hodgkin’s disease. Blood 1997;89:814–22.
75. Gutierrez-Delgado F, Holmberg L, Hooper H, et al. Autologous stem cell transplantation for Hodgkin’s disease: busulfan, melphalan and thiotepa compared to a radiation-based regimen. Bone Marrow Transplant 2003;32:279–85.
76. Hake CR, Graubert TA, Fenske TS. Does autologous transplantation directly increase the risk of secondary leukemia in lymphoma patients? Bone Marrow Transplant 2007;39:59–70.
77. Hahn T, McCarthy PL, Carreras J, et al. Comparison of prognostic models for autologous hematopoietic stem cell transplantation (AHCT) for relapsed Hodgkin lymphoma. Blood 2009;114:1215.
78. Chen Y-B, Lane AA, Logan BR, et al. Impact of conditioning regimen on outcomes for patients with lymphoma undergoing high-dose therapy with autologous hematopoietic cell transplantation. Biology Blood Marrow Transplant 2015;21:1046–53.
79. Wendland MM, Asch JD, Pulsipher MA, et al. The impact of involved field radiation therapy for patients receiving high-dose chemotherapy followed by hematopoietic progenitor cell transplant for the treatment of relapsed or refractory Hodgkin disease. Am J Clin Oncol 2006;29:189–95.
80. Biswas T, Culakova E, Friedberg JW, et al. Involved field radiation therapy following high dose chemotherapy and autologous stem cell transplant benefits local control and survival in refractory or recurrent Hodgkin lymphoma. Radiother Oncol 2012;103:367–72.
81. Levis M, Piva C, Filippi AR, et al. Potential benefit of involved-field radiotherapy for patients with Relapsed-refractory Hodgkin’s lymphoma with incomplete response before autologous stem cell transplantation. Clin Lymphoma Myeloma Leuk 2017;17:14–22.
82. Josting A, Franklin J, May M, et al. New prognostic score based on treatment outcome of patients with relapsed Hodgkin’s lymphoma registered in the database of the German Hodgkin’s lymphoma study group. J Clin Oncol 2002;20:221–30.
83. Moskowitz CH, Nademanee A, Masszi T, et al. Brentuximab vedotin as consolidation therapy after autologous stem-cell transplantation in patients with Hodgkin’s lymphoma at risk of relapse or progression (AETHERA): a randomised, double-blind, placebo-controlled, phase 3 trial. Lancet 2015;385:1853–62.
84. Bonfante V, Santoro A, Viviani S, et al. Outcome of patients with Hodgkin’s disease failing after primary MOPP-ABVD. J Clin Oncol 1997;15:528–34.
85. Josting A, Rueffer U, Franklin J, et al. Prognostic factors and treatment outcome in primary progressive Hodgkin lymphoma: a report from the German Hodgkin Lymphoma Study Group. Blood 2000;96:1280–6.
86. Josting A, Nogova L, Franklin J, et al. Salvage radiotherapy in patients with relapsed and refractory Hodgkin’s lymphoma: a retrospective analysis from the German Hodgkin Lymphoma Study Group. J Clin Oncol 2005;23:1522–9.
87. Campbell B WA, Milner A, Di Iulio J, et al. Long-term follow-up of salvage radiotherapy in Hodgkin’s lymphoma after chemotherapy failure. Int J Radiat Oncol Biol Phys 2005;63:1538–45.
88. Wirth A, Corry J, Laidlaw C, et al. Salvage radiotherapy for Hodgkin’s disease following chemotherapy failure. Int J Radiat Oncol Biol Phys 1997;39:599–607.
89. Schulz H, Rehwald U, Morschhauser F, et al. Rituximab in relapsed lymphocyte-predominant Hodgkin lymphoma: long-term results of a phase 2 trial by the German Hodgkin Lymphoma Study Group (GHSG). Blood 2008;111:109–11.
90. Ekstrand BC, Lucas JB, Horwitz SM, et al. Rituximab in lymphocyte-predominant Hodgkin disease: results of a phase 2 trial. Blood 2003;101:4285–9.
91. Younes A, Romaguera J, Hagemeister F, et al. A pilot study of rituximab in patients with recurrent, classic Hodgkin disease. Cancer 2003;98:310–4.
92. Rehwald U, Schulz H, Reiser M, et al. Treatment of relapsed CD20+ Hodgkin lymphoma with the monoclonal antibody rituximab is effective and well tolerated: results of a phase 2 trial of the German Hodgkin Lymphoma Study Group. Blood 2003;101:420–4.
93. Fehniger TA, Larson S, Trinkaus K, et al. A phase 2 multicenter study of lenalidomide in relapsed or refractory classical Hodgkin lymphoma. Blood 2011;118:5119–25.
94. Boll B, Borchmann P, Topp MS, et al. Lenalidomide in patients with refractory or multiple relapsed Hodgkin lymphoma. Br J Haematol 2010;148:480–2.
95. LaCasce AS, Bociek G, Sawas A, et al. Brentuximab vedotin plus bendamustine: a highly active salvage treatment regimen for patients with relapsed or refractory Hodgkin lymphoma. Blood 2015;126:3982.
96. Diefenbach CS, Hong F, David KA, et al. A phase I study with an expansion cohort of the combination of ipilimumab and nivolumab and brentuximab vedotin in patients with relapsed/refractory Hodgkin lymphoma: A trial of the ECOG-ACRIN Cancer Research Group (E4412 Arms D and E). Blood 2016;128:1106.
97. Herrera AF, Bartlett NL, Ramchandren R, et al. Preliminary results from a phase 1/2 study of brentuximab vedotin in combination with nivolumab in patients with relapsed or refractory Hodgkin lymphoma. Blood 2016;128:1105.
98. Johnston PB, Inwards DJ, Colgan JP, et al. A phase II trial of the oral mTOR inhibitor everolimus in relapsed Hodgkin lymphoma. Am J Hematol 2010;85:320–4.
99. Kewalramani T, Nimer SD, Zelenetz AD, et al. Progressive disease following autologous transplantation in patients with chemosensitive relapsed or primary refractory Hodgkin’s disease or aggressive non-Hodgkin’s lymphoma. Bone Marrow Transplant 2003;32:673–9.
100. Lin TS, Avalos BR, Penza SL, et al. Second autologous stem cell transplant for multiply relapsed Hodgkin’s disease. Bone Marrow Transplant 2002;29:763–7.
101. Smith SM, van Besien K, Carreras J, et al. Second autologous stem cell transplantation for relapsed lymphoma after a prior autologous transplant. Biol Blood Marrow Transplant 2008;14:904–12.
102. Gajewski JL, Phillips GL, Sobocinski KA, et al. Bone marrow transplants from HLA-identical siblings in advanced Hodgkin’s disease. J Clin Oncol 1996;14:572–8.
103. Peniket AJ, Ruiz de Elvira MC, Taghipour G, et al. An EBMT registry matched study of allogeneic stem cell transplants for lymphoma: allogeneic transplantation is associated with a lower relapse rate but a higher procedure-related mortality rate than autologous transplantation. Bone Marrow Transplant 2003;31:667–78.
104. Anderlini P, Saliba R, Acholonu S, et al. Fludarabine-melphalan as a preparative regimen for reduced-intensity conditioning allogeneic stem cell transplantation in relapsed and refractory Hodgkin’s lymphoma: the updated M.D. Anderson Cancer Center experience. Haematologica 2008;93:257–64.
105. Burroughs LM, O’Donnell PV, Sandmaier BM, et al. Comparison of outcomes of HLA-matched related, unrelated, or HLA-haploidentical related hematopoietic cell transplantation following nonmyeloablative conditioning for relapsed or refractory Hodgkin lymphoma. Biol Blood Marrow Transplant 2008;14:1279–87.
106. Peggs KS, Hunter A, Chopra R, et al. Clinical evidence of a graft-versus-Hodgkin’s-lymphoma effect after reduced-intensity allogeneic transplantation. Lancet 2005;365:1934–41.
107. Sureda A, Robinson S, Canals C, et al. Reduced-intensity conditioning compared with conventional allogeneic stem-cell transplantation in relapsed or refractory Hodgkin’s lymphoma: an analysis from the Lymphoma Working Party of the European Group for Blood and Marrow Transplantation. J Clin Oncol 2008;26:455–62.
108. Devetten MP, Hari PN, Carreras J, et al. Unrelated donor reduced-intensity allogeneic hematopoietic stem cell transplantation for relapsed and refractory Hodgkin lymphoma. Biol Blood Marrow Transplant 2009;15:109–17.
109. Sureda A, Canals C, Arranz R, et al. Allogeneic stem cell transplantation after reduced intensity conditioning in patients with relapsed or refractory Hodgkin’s lymphoma. Results of the HDR-ALLO study - a prospective clinical trial by the Grupo Espanol de Linfomas/Trasplante de Medula Osea (GEL/TAMO) and the Lymphoma Working Party of the European Group for Blood and Marrow Transplantation. Haematologica 2012;97:310–7.
110. Sarina B, Castagna L, Farina L, et al. Allogeneic transplantation improves the overall and progression-free survival of Hodgkin lymphoma patients relapsing after autologous transplantation: a retrospective study based on the time of HLA typing and donor availability. Blood 2010;115:3671–7.
111. Castagna L, Sarina B, Todisco E, et al. Allogeneic stem cell transplantation compared with chemotherapy for poor-risk Hodgkin lymphoma. Biol Blood Marrow Transplant 2009;15:432–8.
112. Thomson KJ, Peggs KS, Smith P, et al. Superiority of reduced-intensity allogeneic transplantation over conventional treatment for relapse of Hodgkin’s lymphoma following autologous stem cell transplantation. Bone Marrow Transplant 2008;41:765–70.
113. Chen R, Palmer JM, Thomas SH, et al. Brentuximab vedotin enables successful reduced-intensity allogeneic hematopoietic cell transplantation in patients with relapsed or refractory Hodgkin lymphoma. Blood 2012;119:6379–81.
INTRODUCTION
Hodgkin lymphoma, previously known as Hodgkin’s disease, is a B-cell lymphoproliferative disease characterized by a unique set of pathologic and epidemiologic features. The disease is characterized by the presence of multinucleate giant cells called Hodgkin Reed-Sternberg (HRS) cells.1 Hodgkin lymphoma is unique compared to other B-cell lymphomas because of the relative rarity of the malignant cells within affected tissues. The HRS cells, which usually account for only 0.1% to 10% of the cells, induce accumulation of nonmalignant lymphocytes, macrophages, granulocytes, eosinophils, plasma cells, and histiocytes, which then constitute the majority of tumor cellularity.2 Although the disease was first described by Sir Thomas Hodgkin in 1832, in part because of this unique histopathology, it was not until the 1990s that it was conclusively demonstrated that HRS cells are in fact monoclonal germinal center–derived B cells.
Due to the development of highly effective therapies for Hodgkin lymphoma, cure is a reasonable goal for most patients. Because of the high cure rate, late complications of therapy must be considered when selecting treatment. This article reviews the clinical features and treatment options for advanced stage and relapsed/refractory Hodgkin lymphoma. A previously published article reviewed the epidemiology, etiology/pathogenesis, pathologic classification, initial workup, and staging evaluation of Hodgkin lymphoma, as well as the prognostic stratification and treatment of patients with early-stage Hodgkin lymphoma.3
PRESENTATION, INITIAL EVALUATION, AND PROGNOSIS
Overall, classical Hodgkin lymphoma (cHL) usually presents with asymptomatic mediastinal or cervical lymphadenopathy. At least 50% of patients will have stage I or II disease.4 A mediastinal mass is seen in most patients with nodular sclerosis cHL, at times showing the characteristics of bulky (> 10 cm) disease. Constitutional, or B, symptoms (fever, night sweats, and weight loss) are present in approximately 25% of all patients with cHL, but 50% of advanced stage patients. Between 10% and 15% of patients will have extranodal disease, most commonly involving lung, bone, and liver. Lymphocyte-predominant Hodgkin lymphoma (LPHL) is a rare histological subtype of Hodgkin lymphoma that is differentiated from cHL by distinct clinicopathological features. The clinical course and treatment approach for LPHL are dependent upon the stage of disease. The clinicopathological features of LPHL are discussed in the early-stage Hodgkin lymphoma article.3
For the purposes of prognosis and selection of treatment, Hodgkin lymphoma is commonly classified as early stage favorable, early stage unfavorable, and advanced stage. For advanced stage Hodgkin lymphoma patients, prognosis can be defined using a tool commonly referred to as the International Prognostic Score (IPS). This index consists of 7 factors: male gender, age 45 years or older, stage IV disease, hemoglobin < 10.5 g/dL, white blood cell (WBC) count > 15,000/μL, lymphopenia (absolute lymphocyte count < 600 cells/μL or lymphocytes < 8% of WBC count), and serum albumin < 4 g/dL.5 In the original study by Hasenclever et al,5 the 5-year freedom from progression (FFP) ranged from 42% to 84% and the 5-year overall survival (OS) ranged from 56% to 90%, depending on the number of factors present. This scoring system, however, was developed using a patient population treated prior to 1992. Using a more recently treated patient population, the British Columbia Cancer Agency (BCCA) found that the IPS is still valid for prognostication, but outcomes have improved across all IPS groups, with 5-year FFP now ranging from 62% to 88% and 5-year OS ranging from 67% to 98%.6 This improvement is likely a reflection of improved therapy and supportive care. Table 1 shows the PFS and OS within each IPS group, comparing the data from the German Hodgkin Study Group (GHSG) and BCCA group.5,6
High expression of CD68 is associated with adverse outcomes, whereas high FOXP3 and CD20 expression on tumor cells are predictors of superior outcomes.8 A recent study found that CD68 expression was associated with OS. Five-year OS was 88% in those with less than 25% CD68 expression, versus 63% in those with greater than 25% CD68 expression.9
Roemer and colleagues evaluated 108 newly diagnosed cHL biopsy specimens and found that almost all cHL patients had concordant alteration of PD-L1 (programmed death ligand-1) and PD-L2 loci, with a spectrum of 9p24.1 alterations ranging from low level polysomy to near uniform 9p24.1 amplification. PD-L1/PD-L2 copy number alterations are therefore a defining pathobiological feature of cHL.10 PFS was significantly shorter for patients with 9p24.1 amplification, and those patients were likely to have advanced disease suggesting that 9p24.1 amplification is associated with less favorable prognosis.10 This may change with the increasing use of PD-1 inhibitors in the treatment of cHL.
High baseline metabolic tumor volume and total lesion glycolysis have also been associated with adverse outcomes in cHL. While not routinely assessed in practice currently, these tools may ultimately be used to assess prognosis and guide therapy in clinical practice.11
ADVANCED STAGE HODGKIN LYMPHOMA
FRONTLINE THERAPY
First-line Chemotherapy
Chemotherapy plays an essential role in the treatment of advanced stage Hodgkin lymphoma. In the 1960s, the MOPP regimen (nitrogen mustard, vincristine, procarbazine, prednisone) was developed, with a 10-year OS of 50% and a progression-free survival (PFS) of 52% reported in advanced stage patients. The complete remission (CR) rate was 81%, and 36% of patients who achieved CR relapsed later.12 This chemotherapy regimen is associated with a significant rate of myelosuppression and infertility as well as long-term risk of secondary myelodysplasia and acute leukemias.13,14 This led to the development of newer regimens such as ABVD (doxorubicin, bleomycin, vinblastine, dacarbazine).15 In a randomized trial, ABVD showed improved failure-free survival (FFS) over MOPP (61% versus 50% at 5 years) but similar OS (66%–73%).16 In light of these findings, and considering the lower rate of infertility and myelotoxicity, ABVD became the standard of care for advanced stage cHL in the United States.
The Stanford V regimen was developed in an attempt to further minimize toxicity.17 Stanford V is a condensed, 12-week chemotherapy regimen that includes mechlorethamine, doxorubicin, vinblastine, etoposide, prednisone, vincristine, and bleomycin, followed by involved-field radiation therapy (IFRT). Subsequent trials compared the Stanford V and ABVD regimens and showed similar OS, freedom from treatment failure (FFTF), and response rates.18,19 The ABVD regimen was noted to have higher pulmonary toxicity, while other toxicities such as lymphopenia and neuropathy were higher with the Stanford V regimen. In addition, Stanford V requires patients to receive radiation therapy (RT) to original sites of disease larger than 5 cm in size and contiguous sites.
Another regimen which has been studied extensively for advanced stage Hodgkin lymphoma, and is considered a standard of care in some parts of the world, is escalated BEACOPP (bleomycin, etoposide, doxorubicin, cyclophosphamide, vincristine, procarbazine, prednisone). In the HD9 study (n = 1196), the GHSG evaluated BEACOPP, escalated BEACOPP, and COPP/ABVD in advanced stage Hodgkin lymphoma.20 All arms of the study included 30 Gy RT to sites of bulky disease or residual disease. This study showed improved OS and FFTF with escalated BEACOPP, but at the cost of higher rates of toxicity. At 10 years, FFTF was 64%, 70%, and 82% with OS rates of 75%, 80%, and 86% for COPP/ABVD, baseline BEACOPP, and escalated BEACOPP, respectively (P < 0.001). The rate of secondary acute leukemia 10 years after treatment was 0.4% for COPP/ABVD, 1.5% for BEACOPP, and 3.0% for escalated BEACOPP. However, 3 subsequent randomized trials did not confirm a survival benefit with escalated BEACOPP relative to ABVD. In the HD 2000 trial (n = 295)21 and in a trial by Viviani and colleagues (n = 331),22 an improvement in OS was not demonstrated in favor of escalated BEACOPP. These studies also confirmed a higher rate of toxicities as well as secondary malignancies associated with the escalated BEACOPP regimen. In the EORTC20012 Intergroup trial (n = 549), 8 cycles of ABVD was compared with 4 cycles of escalated BEACOPP followed by 4 cycles of baseline BEACOPP, without radiation, in patients with clinical stage III or IV Hodgkin lymphoma with IPS score ≥ 3. Both regimens resulted in statistically similar FFS (63.7% in ABVD × 8 versus 69.3% in BEACOPP 4+4) and OS (86.7% in ABVD × 8 vs 90.3% in BEACOPP 4+4).23
In the United States, ABVD (6–8 cycles) is commonly used, although escalated BEACOPP (particularly for patients with an IPS of 4 or higher) and Stanford V are considered appropriate as well.24 In the North American Intergroup study comparing ABVD to Stanford V, and in the trial by Viviani et al, ABVD was associated with a 5- to 7-year FFS of 73% to 79% and OS of 84% to 92%.19,22 Given these excellent results, as well as the potential to cure patients with second-line therapy consisting of autologous hematopoietic cell transplantation (auto-HCT), the general consensus among most U.S. hematologists and oncologists is that ABVD remains the treatment of choice, and that the improved FFS/PFS with escalated BEACOPP is not outweighed by the additional toxicity associated with the regimen. There may, however, be a role for escalated BEACOPP in select patients who have a suboptimal response to ABVD as defined by interim positron emission tomography (iPET) scan (see below).
Brentuximab vedotin is an anti-CD30 antibody-drug conjugate (ADC) consisting of an anti-CD30 antibody linked to monomethyl auristatin E (MMAE), a potent antitubulin agent. CD30 is highly expressed on HRS cells and also in anaplastic large cell lymphoma. Upon binding to CD30, the ADC/CD30 complex is then internalized and directed to the lysosome, where the ADC is proteolytically cleaved, releasing MMAE from the antibody. MMAE then disrupts microtubule networks within the cell, leading to G2/M cycle arrest and apoptosis. CD30 is consistently expressed on HRS cells. In addition to being studied in the relapsed/refractory setting (described below), brentuximab has been studied in the first-line setting. In a phase 1 trial, brentuximab combined with ABVD was associated with increased pulmonary toxicity, while brentuximab + AVD had no significant pulmonary toxicity, with an excellent CR rate (96%), suggesting that substituting brentuximab for bleomycin may be an effective strategy. In addition to possibly being more efficacious, this strategy would also have the benefit of eliminating the risk of bleomycin pulmonary toxicity.25 Based on this data, a large international phase 3 study (the ECHELON-1 trial) comparing ABVD versus brentuximab + AVD in advanced stage cHL patients was recently completed. This study enrolled 1334 patients, and preliminary results were recently announced. With a median follow-up of 24 months, the brentuximab + AVD arm had a 4.9% absolute improvement in PFS relative to the ABVD arm (82.1% versus 77.2%). The brentuximab + AVD arm had an increased incidence of febrile neutropenia, managed with growth factors and peripheral neuropathy requiring dose adjustments, whereas the ABVD arm had an increased rate and severity of pulmonary toxicity.26 Further follow-up will be required to determine whether this will translate into a survival benefit. See Table 2 for a summary of recent large randomized prospective phase 3 trials in advanced stage Hodgkin lymphoma.
Alternative Regimens in Older Patients
Patients older than 60 years of age often have poor tolerance for ABVD and especially escalated BEACOPP. This results in increased treatment-related mortality and reduced overall dose intensity, with higher relapse rates and poor OS. In an attempt to improve on the results of treatment of elderly patients with Hodgkin lymphoma, alternative regimens have been explored. One example is PVAG (prednisone, vinblastine, doxorubicin, gemcitabine). With this regimen, the 3-year OS was 66% and PFS was 58%. One patient out of 59 died from treatment-related toxicity, which is much improved over the historical figures for elderly patients with Hodgkin lymphoma.27 Another commonly used approach in practice is to simply omit bleomycin from ABVD. In the early-stage setting (GHSG HD-13 trial), this regimen (referred to as AVD) led to 89.6% PFS at 5 years, compared to 93.5% with ABVD.28 It therefore stands to reason that this should be a reasonable option in older or more frail advanced stage cHL patients as well.
Brentuximab has been evaluated as a single-agent therapy for first-line therapy of elderly patients with Hodgkin lymphoma. In a phase 2 study, 27 patients (63% with advanced stage disease) were treated, with a 92% overall response rate and 73% CR rate. However the median duration of remission was disappointing at only 9.1 months.29 Based on this data, single-agent brentuximab appears to be a reasonable and well tolerated option for frail or elderly patients, although with the caveat that long-term disease control is relatively uncommon.
RESPONSE-ADAPTED FRONTLINE THERAPY USING INTERIM PET SCAN
In recent years, response-adapted treatment approaches have been extensively researched in cHL using iPET. The goal is to reduce toxicity by minimizing therapy in those who achieve negative iPET and/or to intensify treatment for patients with suboptimal response on iPET. Gallamini et al evaluated the prognostic role of an early iPET scan in advanced Hodgkin lymphoma patients (n = 190) treated with ABVD. This study found that patients with positive iPET had a 2-year PFS of 12.8% versus 95.0% in patients with negative iPET. This result was highly statistically significant (P < 0.0001). This study also showed that PET-2 (iPET after 2 cycles of ABVD) superseded the prognostic value of the IPS at diagnosis.30 As a result, numerous subsequent studies have been pursued using iPET for risk-adapted treatment in cHL.
A critical element to the conduct of iPET risk-adapted treatment for cHL is the interpretation of the iPET. In hopes of standardizing iPET interpretation in clinical trials, a scoring system called the Deauville score was developed. The Deauville score ranges from 1 to 5 (Table 3).
The SWOG (Southwest Oncology Group) S0816 trial (n = 358) evaluated iPET-adapted treatment after 2 cycles of ABVD in stage III or IV Hodgkin lymphoma patients. Patients with positive iPET (Deauville score 4 to 5; n = 60) received escalated BEACOPP for 6 cycles, whereas iPET-negative (Deauville score 1 to 3; n = 271) patients continued to receive 4 more cycles of ABVD. The 2-year PFS was 64% for iPET-positive patients.33 This PFS was much higher than the expected 15% to 30% from prior studies such as Gallamini et al,30 suggesting that the treatment intensification may have been of benefit.
In the HD0801 study (n = 519), newly diagnosed advanced Hodgkin lymphoma patients with positive iPET after 2 cycles of ABVD (n = 103) received early ifosfamide-containing salvage therapy followed by high-dose therapy with autologous stem cell rescue. The 2-year PFS was 76% for PET-2–positive patients, comparable with PET-2–negative patients who had PFS of 81%.34 Again, this result for iPET-positive patients was much better than expected based on the historical control from Gallamini et al, suggesting that the treatment intensification may have been beneficial. It should be emphasized, however, that neither HD0801 nor S0816 were randomized prospective trials; rather, all iPET-positive patients were assigned to an intensified treatment approach.
In the HD18 trial (n = 1100), patients with advanced stage cHL started therapy with escalated BEACOPP and underwent an iPET after 2 cycles. For those with a positive iPET, rituximab was added to escalated BEACOPP in the experimental arm (n = 220) for cycles 3 through 8. The control group (n = 220) continued to receive 6 more cycles of escalated BEACOPP. In the 2 groups, the 3-year PFS was similar (91.4% in escalated BEACOPP, 93% in rituximab + escalated BEACOPP), suggesting no significant benefit with addition of rituximab.35 This study also calls into question whether iPET provides useful information for patients receiving intensive therapy such as escalated BEACOPP, and indicates that the historical control data for iPET-positive patients from Gallamini et al may not be consistently reproduced in other prospective trials. As a result, nonrandomized trials that implement an iPET risk-adapted approach should be interpreted with caution. See Table 4 for a summary of recent trials in advanced stage Hodgkin lymphoma using iPET scan to guide therapy.
RADIATION THERAPY IN FRONTLINE TREATMENT
In patients with advanced stage Hodgkin lymphoma, IFRT to initial bulky sites of disease may be incorporated into frontline therapy to improve local control. However, whether this provides a survival benefit and which patients benefit most from consolidative RT remain unclear.
The European Organization for Research and Treatment of Cancer (EORTC) completed a randomized study in advanced stage Hodgkin lymphoma patients who achieved complete or partial remission after MOPP-ABV.36 Patients in CR were randomly assigned to receive no further treatment versus IFRT (24 Gy to all initially involved nodal areas and 16 to 24 Gy to all initially involved extranodal sites). Patients in partial remission (PR) were treated with 30 Gy to nodal areas and 18 to 24 Gy to extranodal sites. Among the CR patients, the 5-year event-free survival (EFS) was 79% to 84% and did not differ for those who received radiation versus those who did not. Five-year OS was 85% to 91% and also did not differ between the 2 groups. However, among the patients in PR after chemotherapy, the 5-year EFS was 79% and the 5-year OS was 87%, which is better than expected for PR patients, indicating a possible benefit to RT in patients with a partial response after chemotherapy. In the GHSG HD12 trial, patients with advanced stage Hodgkin lymphoma who had a residual lesion by computed tomography (CT) (but not analyzed by PET) had a very subtle improvement in FFTF (90% versus 87%) in favor of consolidation with IFRT, but again no survival benefit was seen.37
The EORTC and HD12 studies described above utilized CT scan for assigning remission status following chemotherapy, and it is now well known that many patients with residual masses (by CT) after chemotherapy may in fact be cured, as such residual radiographic abnormalities may simply be composed of fibrosis. PET scan is more accurate than CT in identifying patients who truly have residual active disease following chemotherapy. As a result, the EORTC study discussed above and the GHSG HD12 trial are of limited relevance in the modern era, in which patients routinely undergo PET scan at the end of therapy. Restricting IFRT to sites that remain PET-positive after completing chemotherapy may be a reasonable strategy that would allow for the avoidance of RT in many patients, and may obviate the need for aggressive second-line therapy (eg, high-dose therapy and autologous hematopoietic cell transplant [auto-HCT]). This approach was taken in the GHSG HD15 trial (n = 2182) in which advanced stage patients were treated with 3 variations on the BEACOPP regimen (8 cycles of escalated BEACOPP, 6 cycles of escalated BEACOPP, or 8 cycles of baseline BEACOPP, randomized in a 1:1:1 ratio). Patients with a residual mass of 2.5 cm or greater on CT scan then underwent a PET scan; if the lesion was PET positive, it was treated with 30 Gy of IFRT. This overall strategy was very effective, with 5-year FFTF rates of 84.4%, 89.3%, and 85.4%, respectively. The OS rates were 91.9%, 95.3%, and 94.5%, respectively. For patients with lesions that remained PET positive after chemotherapy, the PFS rate was 86.2% at 48 months, whereas patients in PR with persistent mass ≥ 2.5 cm but with negative PET had a PFS of 92.6%, similar to that of patients in CR.38 With this approach of BEACOPP followed by PET-guided radiation, the proportion of patients receiving RT was reduced from 71% (in the HD9 study) to only 11% in the HD15 study,38 with no apparent loss in overall efficacy when comparing the results of the 2 studies.
UPFRONT STEM CELL TRANSPLANTATION
To further improve outcomes of patients with advanced Hodgkin lymphoma with high-risk disease, high-dose therapy with auto-HCT has been explored as part of frontline therapy. While this has been shown to be feasible in such patients,39 randomized trials have not shown a clear benefit in terms of FFS or OS with upfront auto-HCT. 40,41 Therefore, auto-HCT is not considered a standard component of frontline therapy for cHL patients who achieve CR by PET/CT scan.
RELAPSED AND REFRACTORY HODGKIN LYMPHOMA
Depending on the stage, risk factors, and frontline regimen utilized, between 5% and 40% of patients with Hodgkin lymphoma can be expected to experience either primary induction failure or a relapse after attaining remission with frontline therapy.3 Primary refractory Hodgkin lymphoma, which occurs in up to 5% to 10% of patients, is defined as progression or no response during induction treatment or within 90 days of completing treatment. In cases where remission status is in question, an updated tissue biopsy is recommended. Biopsy is also recommended in cases in which new sites of disease have appeared or if relapse has occurred after a durable period of remission. Restaging is recommended at the time of relapse.
For younger patients with relapsed/refractory Hodgkin lymphoma, the standard of care in most cases is second-line (or salvage) chemotherapy followed by high-dose therapy and auto-HCT. For patients not felt to be candidates for auto-HCT, options include conventional second-line chemotherapy alone, salvage radiotherapy, novel agents such as brentuximab or immune checkpoint inhibitors, and/or participation in clinical trials.
CONVENTIONAL MULTI-AGENT CHEMOTHERAPY REGIMENS
Numerous conventional regimens have been shown in phase 2 studies to be active in relapsed and refractory Hodgkin lymphoma. These include platinum-based regimens, gemcitabine-based regimens, and alkylator-based regimens. No randomized trials in Hodgkin lymphoma have been conducted comparing these regimens. In general, regimens are chosen based on the patient’s age, performance status, comorbidities, and whether auto-HCT is being considered.
In the United States, platinum-based regimens such as ICE (ifosfamide, carboplatin, etoposide),42 DHAP (dexamethasone, cisplatin, high-dose cytarabine),43 ESHAP (etoposide, methylprednisolone, high-dose cytarabine, cisplatin),44 GDP (gemcitabine, cisplatin, dexamethasone),45 and GCD (gemcitabine, carboplatin, dexamethasone)46 are all considered appropriate second-line therapy options for patients being considered for auto-HCT, due to their high response rates and because autologous hematopoietic stem cell collection remains feasible after these regimens. Response rates range from 60% to 88%, with CR rates between 17% and 41%, and toxic death rates generally well below 5%.
Other gemcitabine-based regimens such as IGEV (ifosfamide, gemcitabine, vinorelbine) and GVD (gemcitabine, vinorelbine, liposomal doxorubicin) are also effective.47,48 GVD is an excellent choice since it is a generally well-tolerated outpatient regimen with a 60% response rate even in heavily pretreated patients.48 Stem cell collection remains feasible after both IGEV and GVD as well. ABVD can produce CR in approximately 20% to 50% of patients initially treated with MOPP.49–51 In practice, however, most patients today with relapsed or refractory Hodgkin lymphoma have already received ABVD as part of their first-line therapy, and retreatment with ABVD is not a good option because it would be associated with prohibitively high cumulative doses of doxorubicin.
These multi-agent chemotherapy regimens may not be tolerated well in patients over age 65 to 70 years or those with significant underlying comorbidities. In recent years, bendamustine has emerged as one of the most active conventional agents for cHL, with overall response rates of 53% to 58% in heavily pre-treated patients.52,53 Bendamustine can generally be tolerated even in elderly patients as well.
Some centers, particularly in Europe, investigated aggressive salvage regimens such as mini-BEAM (carmustine, etoposide, cytarabine, melphalan)54 or dexa-BEAM (BEAM plus dexamethasone).55 These regimens, however, are associated with significant hematologic toxicity and high (2%–5%) treatment-related mortality. As a result, these are rarely used in the United States.
For patients who have progressed after (or are not candidates for) platinum- and/or gemcitabine-based therapy, older alkylator-based regimens such as MOPP, C-MOPP, or ChlVPP (chlorambucil, vinblastine, procarbazine, prednisone) can be considered.56–58 However, these regimens are associated with significant bone marrow suppression, and autologous hematopoietic stem cell collection may no longer be feasible after such regimens. Therefore, these regimens should only be given to patients not felt to be auto-HCT candidates, or patients for whom autologous hematopoietic stem cell collection has already been completed. Weekly vinblastine or single-agent gemcitabine are palliative chemotherapy options, with response rates in the 60% to 80% range. Patients can sometimes be maintained on such low-intensity palliative regimens for 6 to 12 months or longer.59,60
BRENTUXIMAB VEDOTIN
Several trials are evaluating incorporation of brentuximab into second-line therapy in transplant-eligible patients. These approaches have used brentuximab prior to, concurrent with, or following platinum-based chemotherapy.61 While there is currently no consensus on the optimal way to incorporate brentuximab into salvage therapy, it is possible that the use of brentuximab or other novel agents in salvage therapy may allow for avoidance of conventional chemotherapy in some patients. In addition, this may translate into more patients proceeding to auto-HCT in a PET negative state. PET negativity prior to auto-HCT is a powerful predictor of long-term remission after auto-HCT, so any intervention that increases the rate of PET negativity prior to auto-HCT would be expected to improve outcomes with auto-HCT.62–65
For patients not being considered for autoHCT, or those for whom platinum-based salvage therapy was ineffective, single-agent brentuximab is an excellent option. In 2 phase 2 studies, an overall response rate (ORR) of 60% to 75% (including a CR rate of 22%–34%) was seen in relapsed and refractory Hodgkin lymphoma patients.66 The US Food and Drug Administration (FDA) approved brentuximab vedotin in August 2011 for treatment of relapsed and refractory Hodgkin lymphoma, after a failed auto-HCT, or in patients who are not auto-HCT candidates and who have received at least 2 prior chemotherapy regimens. With more extended follow-up, it has become clear that a proportion of patients who achieve CR to brentuximab may maintain remission long-term—58% at 3 years and 38% at 5 years.67 These patients may in fact be cured, in many cases without having undergone allogeneic HCT (allo-HCT) after brentuximab.
PD-1 (IMMUNE CHECKPOINT) INHIBITORS
As discussed earlier, PD-L1/PD-L2 copy number alterations represent a disease-defining feature of cHL. Alterations in chromosome 9p24.1 increase the expression of PD-1 ligands PD-L1 and PD-L2. Nivolumab and pembrolizumab are PD-1-blocking antibodies, which have recently been FDA approved for relapsed and refractory cHL. In a study with 23 patients, with 78% of them relapsing after auto-HCT and 78% relapsing after brentuximab, nivolumab produced an objective response in 87% of the patients, with 17% achieving CR and 70% achieving PR. The rate of PFS was 86% at 24 weeks.68 Pembrolizumab, another PD-1 antagonist, was also tested in relapsed and refractory Hodgkin lymphoma. In the KEYNOTE-087 study (n = 210), pembrolizumab produced an ORR of 64% to 70% in 3 different cohorts of relapsed and refractory cHL patients. Overall CR rate was 22%.69 In general, these agents are well tolerated, although patients must be monitored closely for
inflammatory/autoimmune-type toxicities including skin rash, diarrhea/colitis, transaminitis, endocrine abnormalities, and pneumonitis. Prompt recognition and initiation of corticosteroids is essential in managing these toxicities. Of note, PD-1 inhibitors should be given very cautiously to patients with a prior history of allo-HCT, since 30% to 55% of such patients will experience acute graft-versus-host disease (GVHD) in this setting. In 2 retrospective studies, the response rate was very high at 77% to 95%; however, 10% to 26% of all patients treated with PD-1 inhibitors post-allo-HCT died from GVHD induced by PD-1 inhibition.70,71 These risks and benefits therefore need to be carefully weighed in the post-allo-HCT setting. In another recent study, the outcomes were reported for 39 patients who underwent allo-HCT after prior therapy with a PD-1 inhibitor. Three patients (7.7%) developed lethal acute GVHD, suggesting there may be an increased risk of GVHD in patients undergoing allo-HCT after prior PD-1 inhibitor therapy.72
AUTOLOGOUS STEM CELL TRANSPLANTATION
Several studies have shown an improved disease-free survival (DFS) or FFS in patients with relapsed cHL treated by auto-HCT as compared to those receiving conventional chemotherapy alone.55,73,74 Overall, for relapsed disease, one can expect an approximately 50% to 60% chance for DFS at 5 years post-transplant. In a retrospective, matched-pair analysis, FFP was 62% for auto-HCT patients, compared to 32% for conventional chemotherapy patients. OS, however, was similar for the 2 groups (47%–54%). Patients failing induction therapy or relapsing within 1 year were seen to benefit the most from auto-HCT, including an OS benefit.74
A European prospective randomized trial was conducted comparing conventional salvage therapy to auto-HCT. In this study, 161 patients with relapsed Hodgkin lymphoma were treated with 2 cycles of dexa-BEAM. Those with chemo-sensitive disease were then randomized to either 2 more cycles of dexa-BEAM or high-dose BEAM with auto-HCT. Auto-HCT was associated with an approximately 55% FFTF at 3 years, versus 34% with conventional chemotherapy alone.55 This benefit again was most apparent for patients relapsing within 1 year of completion of primary therapy, although an OS benefit was not seen with auto-HCT. For patients with late relapse (>1 year after completion of primary therapy), auto-HCT was associated with an approximately 75% FFTF at 3 years, versus 50% with chemotherapy alone. One other small randomized trial of auto-HCT in relapsed and refractory Hodgkin lymphoma also showed an improved 3-year EFS in favor of auto-HCT (53% versus 10%), again with no difference in OS.73
The lack of OS benefit seen in these studies suggests that auto-HCT at first or second relapse provides comparable outcomes. Auto-HCT offers the benefit of avoiding the long-term toxicities associated with multiple salvage regimens and the anxiety associated with multiple relapses. In addition, the treatment-related mortality with auto-HCT is now in the 1% to 2% range in younger patients, at centers that perform the procedure routinely. For all of these reasons, auto-HCT is commonly recommended by physicians for Hodgkin lymphoma patients in first or second relapse. In most cases, transplant is favored in first relapse, since waiting until second relapse may be associated with a lower chance of achieving CR and difficulty collecting sufficient hematopoietic stem cells. For patients with early relapse or primary refractory disease, an even stronger case can be made for auto-HCT as the best option to achieve sustained control of the disease. For patients with late relapse, conventional salvage therapy alone may be a reasonable option, particularly in older or frail patients, or those with significant comorbid conditions.
The optimal conditioning regimen for autoHCT for relapsed and refractory Hodgkin lymphoma remains undefined. No randomized trials have been performed comparing conditioning regimens for relapsed and refractory Hodgkin lymphoma. One retrospective study compared 92 patients with Hodgkin lymphoma who underwent auto-HCT using a total-body irradiation (TBI) regimen versus a chemotherapy-alone regimen. No difference in 5-year OS or EFS was seen.75 Given the lack of benefit seen with TBI, along with reports of increased rates of secondary malignancies and myelodysplasia with TBI,76 chemotherapy-alone conditioning regimens are most widely employed. For example, in the United States, either the BEAM or CBV (cyclophosphamide, carmustine, etoposide) regimens are used in over 80% of cases.77 This practice was justified in a Center for International Blood and Marrow Transplant Research (CIBMTR) retrospective study comparing outcomes by conditioning regimens, in which no regimen performed better than BEAM or CBV.78
IFRT is often given as an adjunctive therapy to sites of initial and/or relapsed disease following auto-HCT. Although a relatively common practice, whether this truly enhances outcomes beyond that obtained with auto-HCT alone is unclear. Two retrospective studies have shown some benefit in terms of improvement in OS at 3 to 5 years in the group that received IFRT (70%–73% versus 40%–56%).79,80 Given the retrospective nature and small size of these studies, a prospective study would be needed to properly define the potential role for IFRT following auto-HCT in relapsed/refractory Hodgkin lymphoma. Another retrospective study (n = 73) that evaluated peri-transplant IFRT in Hodgkin lymphoma patients receiving auto transplant found no improvement in survival for patients who received peri-transplant IFRT. This study, however, did show a survival benefit in the subgroup of patients with limited stage disease.81
Prognostic Factors Associated with Outcome with Auto-HCT
The factor most consistently associated with improved outcome for patients with relapsed and refractory Hodgkin lymphoma who undergo auto-HCT is the disease status at transplant.63,77 Those in a second CR, versus a chemo-sensitive relapse (but not CR), versus a chemo-refractory relapse have DFS rates of 60% to 70%, 30% to 40%, and 10% to 20%, respectively.63 The duration between remission and relapse also has important prognostic significance. Late relapse (> 1 year after completion of frontline therapy) is associated with better outcomes as compared to early relapse.55 Other factors with prognostic significance at relapse include anemia, time to relapse and clinical stage, B symptoms, extranodal disease, number of prior chemotherapy regimens, and performance status.42,82 The prognostic impact of pretransplant disease status has been confirmed by studies using functional imaging (eg, FDG-PET or gallium scans). In a report by Moskowitz et al, patients with negative functional imaging following second-line therapy had a 77% EFS post-auto-HCT versus 33% in those whose functional imaging remained positive.62 Very similar findings have been reported by other groups.63–65
Post-Auto-HCT Brentuximab Maintenance
In the multicenter, randomized, double-blinded phase 3 AETHERA trial (n = 329), brentuximab (n = 165) was compared with placebo (n = 164) in patients with unfavorable risk relapsed or primary refractory cHL who had undergone autologous transplant. Eligible patients had at least 1 of the following risk factors for progression after auto-HCT: primary refractory Hodgkin lymphoma (failure to achieve complete remission), relapsed Hodgkin lymphoma with an initial remission duration of less than 12 months, or extranodal involvement at the start of pre-transplantation salvage chemotherapy. Patients were required to have CR, PR, or stable disease after pretransplant salvage chemotherapy with adequate kidney, liver, and bone marrow function. Patients who previously received brentuximab were excluded. Patients received 16 cycles of brentuximab or placebo once every 3 weeks starting 30 to 45 days after transplant. The PFS was significantly improved in the brentuximab group when compared to the placebo group (hazard ratio 0.57; P = 0.0013) after a median observation time of 30 months. Median PFS was 42.9 months in the brentuximab group versus 24.1 months in the placebo group; estimated 2-year PFS rates were 63% in the brentuximab group and 51% in the placebo group. OS was not significantly different between the study groups (~85%), presumably due to the fact that patients in the control group who relapsed likely went on to receive brentuximab as a subsequent therapy.83
PRIMARY REFRACTORY HODGKIN LYMPHOMA
Patients with primary refractory Hodgkin lymphoma have a poor outcome. Salvage therapy using conventional chemotherapy and/or RT results in long-term DFS in 10% or fewer of such patients.13,84 Given these poor outcomes with conventional salvage therapy, auto-HCT is considered to be the standard of care for this subset of patients. The GHSG retrospectively analyzed the prognostic factors and outcomes of patients with primary refractory Hodgkin lymphoma. The 5-year freedom-from-second-failure and the 5-year OS were reported to be 31% and 43%, respectively, for those patients treated with auto-HCT. Patients with poor functional status at time of transplant, age greater than 50 years, and failure to attain a temporary remission had a 0% 5-year OS, as compared to 55% in patients without any of these risk factors.85 A large retrospective European study showed that patients with chemo-resistant disease who underwent transplant had a 19% survival at 5 years.63 Hence, even patients with primary refractory Hodgkin lymphoma have some chance of achieving long-term survival following auto-HCT.
SALVAGE RADIOTHERAPY
The GHSG performed a retrospective analysis of the efficacy of salvage RT in patients with refractory or first-relapsed Hodgkin lymphoma. Five-year FFTF and OS rates were 28% and 51%, respectively. Patients with a limited-stage relapse and without B symptoms were more likely to benefit from salvage RT.86 Campbell et al reported on 81 patients undergoing salvage RT for persistent or recurrent Hodgkin lymphoma after chemotherapy. The 10-year FFTF and OS rates were 33% and 46%, respectively.87 Similarly, Wirth et al reported a 5-year FFS of 26% and 5-year OS of 57%. These figures were 36% and 75%, respectively, in patients whose relapse was limited to supradiaphragmatic nodal sites without B symptoms.88 RT therefore may be a useful strategy for a subset of patients who relapse following chemotherapy, particularly those with a limited-stage relapse, without B symptoms, and those with relapsed disease after a CR, as opposed to those with a partial response or lack of response to the prior chemotherapy regimen.
INVESTIGATIONAL AGENTS AND NOVEL COMBINATIONS
Several biological therapies are emerging as options for the treatment of refractory or relapsed disease. These therapies consist of monoclonal antibodies and ADCs that target cell surface antigens, or small molecules that inhibit key intracellular pathways within neoplastic cells.
Rituximab
Rituximab is a chimeric anti-CD20 monoclonal antibody used widely in B-cell non-Hodgkin lymphomas. The CD20 molecule is typically highly expressed in nodular lymphocyte-predominant Hodgkin lymphoma (NLPHL). Two studies (one in relapsed patients, the other in a mixture of relapsed and previously untreated patients) showed significant activity of rituximab in relapsed NLPHL, with ORRs ranging from 94% to 100%, CR rates ranging from 41% to 53%, and median duration of remission in the 10- to 33-month range.89,90 In cHL, CD20 is expressed in HRS cells in 20% to 30% of cases. In such cases, single-agent rituximab has also shown activity. There is also evidence that rituximab may be effective even in cases in which the HRS cells are CD20-negative, presumably by virtue of depleting reactive B lymphocytes from the microenvironment, which may enhance anti-tumor immunity, or by eliminating a putative CD20-expressing Hodgkin lymphoma stem cell.91,92
Lenalidomide
Lenalidomide is an immunomodulatory drug that has multiple modes of action, including direct induction of apoptosis in tumor cells, antiangiogenic effects, and the activation of immune cells, such as natural killer cells and T cells. Lenalidomide has been shown to modify many features of the microenvironment of HRS cells and has demonstrated activity in other B-cell neoplasms. As a result, lenalidomide has been evaluated in relapsed and refractory Hodgkin lymphoma patients. A multicenter phase 2 study by Fehniger et al included 35 patients, 87% of whom had previously undergone HCT and 55% of whom were refractory to the last therapy.93 All patients were given lenalidomide 25 mg/day from days 1 to 21 of a 28-day cycle until disease progression. One patient was noted to achieve CR, 6 achieved PR, and 5 had stable disease lasting more than 6 months, for an ORR of 19% and a “cytostatic overall response rate” of 33%. The median duration of CR/partial remission was 6 months, with the median time-to-treatment failure in responders (including those with stable disease > 6 months) being 15 months. Similarly, in another study, Böll et al evaluated 12 patients across 4 German centers with relapsed or refractory disease who were treated with oral lenalidomide for 21 days in a 28-day cycle. No radiological evidence of disease progression after 2 cycles of lenalidomide was seen in any of the enrolled patients. ORR was noted to be 50%, with 6 patients with stable disease and 5 patients achieving PR after 2 cycles.94
Novel Brentuximab Combination Therapies
Brentuximab plus bendamustine. The combination of brentuximab and bendamustine was tested as an outpatient regimen in a phase 1/2 study (n = 55) in primary refractory Hodgkin lymphoma or after first relapse. The CR rate of the combination was 74%, with an overall objective response (CR + PR) of 93%. The CR rates were 64% and 84%, respectively, for refractory and relapsed patients. The PFS at 12 months was 80%, establishing this combination therapy as an effective salvage regimen with durable response.95
Brentuximab plus nivolumab. Preliminary results have recently been presented from 2 studies96,97 evaluating the combination of brentuximab and nivolumab. While this combination would still be considered investigational, these studies showed very encouraging ORRs of 90% to 100% and a CR rate of 62% to 66%. Longer follow-up is needed to determine whether these responses are durable and to document the toxicity profile of this combination.
Mammalian Target of Rapamycin Inhibitors
Two mammalian target of rapamycin (mTOR) inhibitors, everolimus and temsirolimus, are currently available in the United States. While neither drug currently has FDA approval for Hodgkin lymphoma, everolimus was evaluated in a phase 2 trial in a heavily pretreated group of relapsed/refractory patients. An ORR of 47% was seen, with a median time to progression of 7.2 months.98
ALLOGENEIC STEM CELL TRANSPLANTATION
Historically, patients who relapse after having an auto-HCT generally had a poor outcome, with a median survival of 2 to 3 years after failure of auto-HCT.99 These patients may be offered palliative chemotherapy (see above), treatment with novel agents (see above), or enrollment in a clinical trial. Select patients may benefit from a second hematopoietic stem cell transplant, most commonly an allo-HCT. However, rare patients with late relapse after auto-HCT may be considered for a second auto-HCT, with a minority of such patients achieving a durable remission after the second auto-HCT.100,101 Because relapse or progressive disease occurs most commonly in the first several months following auto-HCT, patients are more often considered for allo-HCT than a second auto-HCT. In addition, a second auto-HCT may not be feasible due to impaired bone marrow reserve and/or concerns for development of secondary myelodysplasia or acute myeloid leukemia.
Several studies have evaluated allo-HCT in relapsed/ refractory Hodgkin lymphoma. Early studies evaluating myeloablative allo-HCT for Hodgkin lymphoma showed excessive treatment-related mortality (up to 50%) and disappointingly low rates of long-term survival (< 25%).102,103 This was likely related to the fact that, in that era, most of the patients with Hodgkin lymphoma evaluated for allo-HCT were heavily pretreated and therefore at a higher risk for toxicity as well as lymphoma progression.
More recently, several studies have focused on the use of reduced-intensity conditioning (RIC) allo-HCT for relapsed and refractory Hodgkin lymphoma. This approach relies more on a “graft-versus-lymphoma” effect, the existence of which has been debated in Hodgkin lymphoma. Three single-center studies of RIC allo-HCT in patients with multiply recurrent Hodgkin lymphoma showed improved rates of treatment-related mortality (8%–16%) but still relatively low rates of long-term PFS (23%–39% at 2 to 4 years).104–106 Interestingly, in one of these studies the outcomes were more favorable for patients who underwent haploidentical (versus matched sibling or matched unrelated donor) transplants.105
Two large registry studies have also reported on the outcomes of RIC allo-HCT in patients with relapsed and refractory Hodgkin lymphoma.107,108 These studies also confirmed a modest improvement in outcomes compared with those seen historically with myeloablative transplants. Treatment-related mortality at 1 to 2 years was 23% to 33%, depending on whether a matched sibling donor versus an unrelated donor was used. However, long-term PFS (18%–20% at 2 to 5 years) and OS (28%–37% at 2 to 5 years) remained poor, primarily due to high rates of progressive lymphoma post-transplant. In both of these studies, patients were heavily pretreated (84%–96% had received 3 or more prior lines of chemotherapy, and 62%–89% received a prior auto-HCT), with 47% to 55% of patients chemo-resistant prior to transplant. Of note, both of these registry studies reflect patients who underwent transplant prior to the widespread use of brentuximab and PD-1 inhibitors.
Based on the single-center and registry data above, a prospective multicenter European phase 2 trial was conducted to evaluate the benefit of RIC allo-HCT in Hodgkin lymphoma.109 Ninety-two patients (86% with prior auto-HCT, 90% with 3 or more prior lines of therapy) were enrolled and given salvage therapy. Those who had stable disease or better following salvage therapy remained on protocol (n = 78) and underwent RIC with fludarabine and melphalan, followed by allo-HCT (70% with matched sibling donors). Treatment-related mortality was 15% at 1 year. Relapse or progression occurred in 49% at 2 years (35% if chemo-sensitive prior to transplant). Chronic GVHD was associated with a decreased rate of relapse, supporting the existence of a graft-versus-lymphoma effect in Hodgkin lymphoma. Unfortunately, PFS among all allografted patients was still relatively poor (24% at 4 years). However, among patients in CR prior to allo-HCT, a 50% PFS was seen at 4 years. Therefore, even in a prospective multicenter study, RIC allo-HCT offered significant benefit with manageable toxicity in relapsed and refractory Hodgkin lymphoma patients with chemo-sensitive disease.
These studies suggest that outcomes with allo-HCT would improve further if implemented earlier in the course of disease and/or with a lower burden of disease at transplant. It has therefore been suggested that allo-HCT should be considered soon after failure of auto-HCT is documented. In a retrospective study by Sarina et al, 185 Hodgkin lymphoma patients who relapsed following auto-HCT were then immediately considered for reduced-intensity allo-HCT.110 Of these, 122 had a donor identified, and 104 (85%) actually underwent allo-HCT. These 104 patients were then compared to the other 81 patients who either had no donor identified or had a donor but did not receive the planned allo-HCT. Two-year PFS and OS were superior in the patients undergoing allo-HCT (39% versus 14% and 66% versus 42%, respectively, P < 0.001), with a median follow-up of 4 years. The presence of chronic GVHD again was associated with improved PFS and OS. Disease status prior to transplant remained highly predictive of PFS and OS by multivariate analysis. Two other smaller retrospective studies similarly found a survival benefit associated with allo-HCT compared with patients who underwent conventional salvage therapies alone.111,112 These studies, although subject to the usual limitations of retrospective analyses, suggest that the results with reduced-intensity allo-HCT are in fact enhanced if applied earlier in the disease course, and are superior to those with conventional therapy alone.
Currently, the exact role of allo-HSCT, including the optimal timing and optimal donor source (matched sibling versus haploidentical sibling versus matched unrelated donor), remain undefined for relapsed and refractory Hodgkin lymphoma. As discussed earlier, brentuximab is highly active in relapsed Hodgkin lymphoma patients, with a subset of patients still in CR at 5 years.67 For such patients, avoiding the risks of allo-HCT is a desirable goal.
For those who relapse or progress after auto-HCT, a reasonable strategy therefore is to treat initially with brentuximab, unless the patient is already known to have responded poorly to brentuximab, or already has significant neuropathy. Those who achieve a CR to brentuximab are then observed. A subset of those patients will remain in remission at 5 years without further therapy. For those who relapse, or who achieve less than a CR to brentuximab, additional treatment (with brentuximab re-treatment being one option) followed by a reduced-intensity allo-HCT is a reasonable consideration. This approach has the theoretical advantages of (1) avoiding the risk of allo-HCT in the subset potentially cured by brentuximab, (2) getting patients to allo-HCT with fewer comorbidities (due to a lower total exposure to conventional chemotherapy pre-transplant), and (3) applying allo-HCT in the setting of sensitive disease/lower disease burden (due to the high efficacy of brentuximab). The results of a small study suggest that brentuximab may in fact be a very effective “bridge” to allotransplant. Chen et al113 reported on 18 patients with relapsed/refractory Hodgkin lymphoma (17 of whom had previously undergone auto-HCT) who were treated on brentuximab vedotin clinical trials. The data were retrospectively evaluated to determine the efficacy and safety of subsequent reduced-intensity allo-HCT. Remarkably, at 1 year the OS was 100%, PFS was 92%, and nonrelapse mortality was 0% with a median follow-up of 14 months. Hence, brentuximab is safe for use prior to reduced-intensity allo-HCT in heavily pre-treated patients and appears to be associated with very favorable post-transplant outcomes, particularly in comparison to older studies of allo-HCT in the era prior to brentuximab.
SUMMARY
Currently, cure is possible for the majority of patients diagnosed with advanced stage Hodgkin lymphoma. The challenge to the clinician is to provide curative treatment with the lowest risk of serious toxicities. Which regimen will best provide this balance of risk and benefit needs to be assessed based on the relapse risk, age, frailty, and comorbidity profile for an individual patient. For many patients with relapsed or refractory Hodgkin lymphoma, cure remains possible using approaches based on hematopoietic stem cell transplantation, RT, and/or brentuximab. In addition, there are now numerous conventional chemotherapy agents, RT strategies, and exciting newer agents such as PD-1 inhibitors, that can provide significant clinical benefit even when cure is not feasible.
INTRODUCTION
Hodgkin lymphoma, previously known as Hodgkin’s disease, is a B-cell lymphoproliferative disease characterized by a unique set of pathologic and epidemiologic features. The disease is characterized by the presence of multinucleate giant cells called Hodgkin Reed-Sternberg (HRS) cells.1 Hodgkin lymphoma is unique compared to other B-cell lymphomas because of the relative rarity of the malignant cells within affected tissues. The HRS cells, which usually account for only 0.1% to 10% of the cells, induce accumulation of nonmalignant lymphocytes, macrophages, granulocytes, eosinophils, plasma cells, and histiocytes, which then constitute the majority of tumor cellularity.2 Although the disease was first described by Sir Thomas Hodgkin in 1832, in part because of this unique histopathology, it was not until the 1990s that it was conclusively demonstrated that HRS cells are in fact monoclonal germinal center–derived B cells.
Due to the development of highly effective therapies for Hodgkin lymphoma, cure is a reasonable goal for most patients. Because of the high cure rate, late complications of therapy must be considered when selecting treatment. This article reviews the clinical features and treatment options for advanced stage and relapsed/refractory Hodgkin lymphoma. A previously published article reviewed the epidemiology, etiology/pathogenesis, pathologic classification, initial workup, and staging evaluation of Hodgkin lymphoma, as well as the prognostic stratification and treatment of patients with early-stage Hodgkin lymphoma.3
PRESENTATION, INITIAL EVALUATION, AND PROGNOSIS
Overall, classical Hodgkin lymphoma (cHL) usually presents with asymptomatic mediastinal or cervical lymphadenopathy. At least 50% of patients will have stage I or II disease.4 A mediastinal mass is seen in most patients with nodular sclerosis cHL, at times showing the characteristics of bulky (> 10 cm) disease. Constitutional, or B, symptoms (fever, night sweats, and weight loss) are present in approximately 25% of all patients with cHL, but 50% of advanced stage patients. Between 10% and 15% of patients will have extranodal disease, most commonly involving lung, bone, and liver. Lymphocyte-predominant Hodgkin lymphoma (LPHL) is a rare histological subtype of Hodgkin lymphoma that is differentiated from cHL by distinct clinicopathological features. The clinical course and treatment approach for LPHL are dependent upon the stage of disease. The clinicopathological features of LPHL are discussed in the early-stage Hodgkin lymphoma article.3
For the purposes of prognosis and selection of treatment, Hodgkin lymphoma is commonly classified as early stage favorable, early stage unfavorable, and advanced stage. For advanced stage Hodgkin lymphoma patients, prognosis can be defined using a tool commonly referred to as the International Prognostic Score (IPS). This index consists of 7 factors: male gender, age 45 years or older, stage IV disease, hemoglobin < 10.5 g/dL, white blood cell (WBC) count > 15,000/μL, lymphopenia (absolute lymphocyte count < 600 cells/μL or lymphocytes < 8% of WBC count), and serum albumin < 4 g/dL.5 In the original study by Hasenclever et al,5 the 5-year freedom from progression (FFP) ranged from 42% to 84% and the 5-year overall survival (OS) ranged from 56% to 90%, depending on the number of factors present. This scoring system, however, was developed using a patient population treated prior to 1992. Using a more recently treated patient population, the British Columbia Cancer Agency (BCCA) found that the IPS is still valid for prognostication, but outcomes have improved across all IPS groups, with 5-year FFP now ranging from 62% to 88% and 5-year OS ranging from 67% to 98%.6 This improvement is likely a reflection of improved therapy and supportive care. Table 1 shows the PFS and OS within each IPS group, comparing the data from the German Hodgkin Study Group (GHSG) and BCCA group.5,6
High expression of CD68 is associated with adverse outcomes, whereas high FOXP3 and CD20 expression on tumor cells are predictors of superior outcomes.8 A recent study found that CD68 expression was associated with OS. Five-year OS was 88% in those with less than 25% CD68 expression, versus 63% in those with greater than 25% CD68 expression.9
Roemer and colleagues evaluated 108 newly diagnosed cHL biopsy specimens and found that almost all cHL patients had concordant alteration of PD-L1 (programmed death ligand-1) and PD-L2 loci, with a spectrum of 9p24.1 alterations ranging from low level polysomy to near uniform 9p24.1 amplification. PD-L1/PD-L2 copy number alterations are therefore a defining pathobiological feature of cHL.10 PFS was significantly shorter for patients with 9p24.1 amplification, and those patients were likely to have advanced disease suggesting that 9p24.1 amplification is associated with less favorable prognosis.10 This may change with the increasing use of PD-1 inhibitors in the treatment of cHL.
High baseline metabolic tumor volume and total lesion glycolysis have also been associated with adverse outcomes in cHL. While not routinely assessed in practice currently, these tools may ultimately be used to assess prognosis and guide therapy in clinical practice.11
ADVANCED STAGE HODGKIN LYMPHOMA
FRONTLINE THERAPY
First-line Chemotherapy
Chemotherapy plays an essential role in the treatment of advanced stage Hodgkin lymphoma. In the 1960s, the MOPP regimen (nitrogen mustard, vincristine, procarbazine, prednisone) was developed, with a 10-year OS of 50% and a progression-free survival (PFS) of 52% reported in advanced stage patients. The complete remission (CR) rate was 81%, and 36% of patients who achieved CR relapsed later.12 This chemotherapy regimen is associated with a significant rate of myelosuppression and infertility as well as long-term risk of secondary myelodysplasia and acute leukemias.13,14 This led to the development of newer regimens such as ABVD (doxorubicin, bleomycin, vinblastine, dacarbazine).15 In a randomized trial, ABVD showed improved failure-free survival (FFS) over MOPP (61% versus 50% at 5 years) but similar OS (66%–73%).16 In light of these findings, and considering the lower rate of infertility and myelotoxicity, ABVD became the standard of care for advanced stage cHL in the United States.
The Stanford V regimen was developed in an attempt to further minimize toxicity.17 Stanford V is a condensed, 12-week chemotherapy regimen that includes mechlorethamine, doxorubicin, vinblastine, etoposide, prednisone, vincristine, and bleomycin, followed by involved-field radiation therapy (IFRT). Subsequent trials compared the Stanford V and ABVD regimens and showed similar OS, freedom from treatment failure (FFTF), and response rates.18,19 The ABVD regimen was noted to have higher pulmonary toxicity, while other toxicities such as lymphopenia and neuropathy were higher with the Stanford V regimen. In addition, Stanford V requires patients to receive radiation therapy (RT) to original sites of disease larger than 5 cm in size and contiguous sites.
Another regimen which has been studied extensively for advanced stage Hodgkin lymphoma, and is considered a standard of care in some parts of the world, is escalated BEACOPP (bleomycin, etoposide, doxorubicin, cyclophosphamide, vincristine, procarbazine, prednisone). In the HD9 study (n = 1196), the GHSG evaluated BEACOPP, escalated BEACOPP, and COPP/ABVD in advanced stage Hodgkin lymphoma.20 All arms of the study included 30 Gy RT to sites of bulky disease or residual disease. This study showed improved OS and FFTF with escalated BEACOPP, but at the cost of higher rates of toxicity. At 10 years, FFTF was 64%, 70%, and 82% with OS rates of 75%, 80%, and 86% for COPP/ABVD, baseline BEACOPP, and escalated BEACOPP, respectively (P < 0.001). The rate of secondary acute leukemia 10 years after treatment was 0.4% for COPP/ABVD, 1.5% for BEACOPP, and 3.0% for escalated BEACOPP. However, 3 subsequent randomized trials did not confirm a survival benefit with escalated BEACOPP relative to ABVD. In the HD 2000 trial (n = 295)21 and in a trial by Viviani and colleagues (n = 331),22 an improvement in OS was not demonstrated in favor of escalated BEACOPP. These studies also confirmed a higher rate of toxicities as well as secondary malignancies associated with the escalated BEACOPP regimen. In the EORTC20012 Intergroup trial (n = 549), 8 cycles of ABVD was compared with 4 cycles of escalated BEACOPP followed by 4 cycles of baseline BEACOPP, without radiation, in patients with clinical stage III or IV Hodgkin lymphoma with IPS score ≥ 3. Both regimens resulted in statistically similar FFS (63.7% in ABVD × 8 versus 69.3% in BEACOPP 4+4) and OS (86.7% in ABVD × 8 vs 90.3% in BEACOPP 4+4).23
In the United States, ABVD (6–8 cycles) is commonly used, although escalated BEACOPP (particularly for patients with an IPS of 4 or higher) and Stanford V are considered appropriate as well.24 In the North American Intergroup study comparing ABVD to Stanford V, and in the trial by Viviani et al, ABVD was associated with a 5- to 7-year FFS of 73% to 79% and OS of 84% to 92%.19,22 Given these excellent results, as well as the potential to cure patients with second-line therapy consisting of autologous hematopoietic cell transplantation (auto-HCT), the general consensus among most U.S. hematologists and oncologists is that ABVD remains the treatment of choice, and that the improved FFS/PFS with escalated BEACOPP is not outweighed by the additional toxicity associated with the regimen. There may, however, be a role for escalated BEACOPP in select patients who have a suboptimal response to ABVD as defined by interim positron emission tomography (iPET) scan (see below).
Brentuximab vedotin is an anti-CD30 antibody-drug conjugate (ADC) consisting of an anti-CD30 antibody linked to monomethyl auristatin E (MMAE), a potent antitubulin agent. CD30 is highly expressed on HRS cells and also in anaplastic large cell lymphoma. Upon binding to CD30, the ADC/CD30 complex is then internalized and directed to the lysosome, where the ADC is proteolytically cleaved, releasing MMAE from the antibody. MMAE then disrupts microtubule networks within the cell, leading to G2/M cycle arrest and apoptosis. CD30 is consistently expressed on HRS cells. In addition to being studied in the relapsed/refractory setting (described below), brentuximab has been studied in the first-line setting. In a phase 1 trial, brentuximab combined with ABVD was associated with increased pulmonary toxicity, while brentuximab + AVD had no significant pulmonary toxicity, with an excellent CR rate (96%), suggesting that substituting brentuximab for bleomycin may be an effective strategy. In addition to possibly being more efficacious, this strategy would also have the benefit of eliminating the risk of bleomycin pulmonary toxicity.25 Based on this data, a large international phase 3 study (the ECHELON-1 trial) comparing ABVD versus brentuximab + AVD in advanced stage cHL patients was recently completed. This study enrolled 1334 patients, and preliminary results were recently announced. With a median follow-up of 24 months, the brentuximab + AVD arm had a 4.9% absolute improvement in PFS relative to the ABVD arm (82.1% versus 77.2%). The brentuximab + AVD arm had an increased incidence of febrile neutropenia, managed with growth factors and peripheral neuropathy requiring dose adjustments, whereas the ABVD arm had an increased rate and severity of pulmonary toxicity.26 Further follow-up will be required to determine whether this will translate into a survival benefit. See Table 2 for a summary of recent large randomized prospective phase 3 trials in advanced stage Hodgkin lymphoma.
Alternative Regimens in Older Patients
Patients older than 60 years of age often have poor tolerance for ABVD and especially escalated BEACOPP. This results in increased treatment-related mortality and reduced overall dose intensity, with higher relapse rates and poor OS. In an attempt to improve on the results of treatment of elderly patients with Hodgkin lymphoma, alternative regimens have been explored. One example is PVAG (prednisone, vinblastine, doxorubicin, gemcitabine). With this regimen, the 3-year OS was 66% and PFS was 58%. One patient out of 59 died from treatment-related toxicity, which is much improved over the historical figures for elderly patients with Hodgkin lymphoma.27 Another commonly used approach in practice is to simply omit bleomycin from ABVD. In the early-stage setting (GHSG HD-13 trial), this regimen (referred to as AVD) led to 89.6% PFS at 5 years, compared to 93.5% with ABVD.28 It therefore stands to reason that this should be a reasonable option in older or more frail advanced stage cHL patients as well.
Brentuximab has been evaluated as a single-agent therapy for first-line therapy of elderly patients with Hodgkin lymphoma. In a phase 2 study, 27 patients (63% with advanced stage disease) were treated, with a 92% overall response rate and 73% CR rate. However the median duration of remission was disappointing at only 9.1 months.29 Based on this data, single-agent brentuximab appears to be a reasonable and well tolerated option for frail or elderly patients, although with the caveat that long-term disease control is relatively uncommon.
RESPONSE-ADAPTED FRONTLINE THERAPY USING INTERIM PET SCAN
In recent years, response-adapted treatment approaches have been extensively researched in cHL using iPET. The goal is to reduce toxicity by minimizing therapy in those who achieve negative iPET and/or to intensify treatment for patients with suboptimal response on iPET. Gallamini et al evaluated the prognostic role of an early iPET scan in advanced Hodgkin lymphoma patients (n = 190) treated with ABVD. This study found that patients with positive iPET had a 2-year PFS of 12.8% versus 95.0% in patients with negative iPET. This result was highly statistically significant (P < 0.0001). This study also showed that PET-2 (iPET after 2 cycles of ABVD) superseded the prognostic value of the IPS at diagnosis.30 As a result, numerous subsequent studies have been pursued using iPET for risk-adapted treatment in cHL.
A critical element to the conduct of iPET risk-adapted treatment for cHL is the interpretation of the iPET. In hopes of standardizing iPET interpretation in clinical trials, a scoring system called the Deauville score was developed. The Deauville score ranges from 1 to 5 (Table 3).
The SWOG (Southwest Oncology Group) S0816 trial (n = 358) evaluated iPET-adapted treatment after 2 cycles of ABVD in stage III or IV Hodgkin lymphoma patients. Patients with positive iPET (Deauville score 4 to 5; n = 60) received escalated BEACOPP for 6 cycles, whereas iPET-negative (Deauville score 1 to 3; n = 271) patients continued to receive 4 more cycles of ABVD. The 2-year PFS was 64% for iPET-positive patients.33 This PFS was much higher than the expected 15% to 30% from prior studies such as Gallamini et al,30 suggesting that the treatment intensification may have been of benefit.
In the HD0801 study (n = 519), newly diagnosed advanced Hodgkin lymphoma patients with positive iPET after 2 cycles of ABVD (n = 103) received early ifosfamide-containing salvage therapy followed by high-dose therapy with autologous stem cell rescue. The 2-year PFS was 76% for PET-2–positive patients, comparable with PET-2–negative patients who had PFS of 81%.34 Again, this result for iPET-positive patients was much better than expected based on the historical control from Gallamini et al, suggesting that the treatment intensification may have been beneficial. It should be emphasized, however, that neither HD0801 nor S0816 were randomized prospective trials; rather, all iPET-positive patients were assigned to an intensified treatment approach.
In the HD18 trial (n = 1100), patients with advanced stage cHL started therapy with escalated BEACOPP and underwent an iPET after 2 cycles. For those with a positive iPET, rituximab was added to escalated BEACOPP in the experimental arm (n = 220) for cycles 3 through 8. The control group (n = 220) continued to receive 6 more cycles of escalated BEACOPP. In the 2 groups, the 3-year PFS was similar (91.4% in escalated BEACOPP, 93% in rituximab + escalated BEACOPP), suggesting no significant benefit with addition of rituximab.35 This study also calls into question whether iPET provides useful information for patients receiving intensive therapy such as escalated BEACOPP, and indicates that the historical control data for iPET-positive patients from Gallamini et al may not be consistently reproduced in other prospective trials. As a result, nonrandomized trials that implement an iPET risk-adapted approach should be interpreted with caution. See Table 4 for a summary of recent trials in advanced stage Hodgkin lymphoma using iPET scan to guide therapy.
RADIATION THERAPY IN FRONTLINE TREATMENT
In patients with advanced stage Hodgkin lymphoma, IFRT to initial bulky sites of disease may be incorporated into frontline therapy to improve local control. However, whether this provides a survival benefit and which patients benefit most from consolidative RT remain unclear.
The European Organization for Research and Treatment of Cancer (EORTC) completed a randomized study in advanced stage Hodgkin lymphoma patients who achieved complete or partial remission after MOPP-ABV.36 Patients in CR were randomly assigned to receive no further treatment versus IFRT (24 Gy to all initially involved nodal areas and 16 to 24 Gy to all initially involved extranodal sites). Patients in partial remission (PR) were treated with 30 Gy to nodal areas and 18 to 24 Gy to extranodal sites. Among the CR patients, the 5-year event-free survival (EFS) was 79% to 84% and did not differ for those who received radiation versus those who did not. Five-year OS was 85% to 91% and also did not differ between the 2 groups. However, among the patients in PR after chemotherapy, the 5-year EFS was 79% and the 5-year OS was 87%, which is better than expected for PR patients, indicating a possible benefit to RT in patients with a partial response after chemotherapy. In the GHSG HD12 trial, patients with advanced stage Hodgkin lymphoma who had a residual lesion by computed tomography (CT) (but not analyzed by PET) had a very subtle improvement in FFTF (90% versus 87%) in favor of consolidation with IFRT, but again no survival benefit was seen.37
The EORTC and HD12 studies described above utilized CT scan for assigning remission status following chemotherapy, and it is now well known that many patients with residual masses (by CT) after chemotherapy may in fact be cured, as such residual radiographic abnormalities may simply be composed of fibrosis. PET scan is more accurate than CT in identifying patients who truly have residual active disease following chemotherapy. As a result, the EORTC study discussed above and the GHSG HD12 trial are of limited relevance in the modern era, in which patients routinely undergo PET scan at the end of therapy. Restricting IFRT to sites that remain PET-positive after completing chemotherapy may be a reasonable strategy that would allow for the avoidance of RT in many patients, and may obviate the need for aggressive second-line therapy (eg, high-dose therapy and autologous hematopoietic cell transplant [auto-HCT]). This approach was taken in the GHSG HD15 trial (n = 2182) in which advanced stage patients were treated with 3 variations on the BEACOPP regimen (8 cycles of escalated BEACOPP, 6 cycles of escalated BEACOPP, or 8 cycles of baseline BEACOPP, randomized in a 1:1:1 ratio). Patients with a residual mass of 2.5 cm or greater on CT scan then underwent a PET scan; if the lesion was PET positive, it was treated with 30 Gy of IFRT. This overall strategy was very effective, with 5-year FFTF rates of 84.4%, 89.3%, and 85.4%, respectively. The OS rates were 91.9%, 95.3%, and 94.5%, respectively. For patients with lesions that remained PET positive after chemotherapy, the PFS rate was 86.2% at 48 months, whereas patients in PR with persistent mass ≥ 2.5 cm but with negative PET had a PFS of 92.6%, similar to that of patients in CR.38 With this approach of BEACOPP followed by PET-guided radiation, the proportion of patients receiving RT was reduced from 71% (in the HD9 study) to only 11% in the HD15 study,38 with no apparent loss in overall efficacy when comparing the results of the 2 studies.
UPFRONT STEM CELL TRANSPLANTATION
To further improve outcomes of patients with advanced Hodgkin lymphoma with high-risk disease, high-dose therapy with auto-HCT has been explored as part of frontline therapy. While this has been shown to be feasible in such patients,39 randomized trials have not shown a clear benefit in terms of FFS or OS with upfront auto-HCT. 40,41 Therefore, auto-HCT is not considered a standard component of frontline therapy for cHL patients who achieve CR by PET/CT scan.
RELAPSED AND REFRACTORY HODGKIN LYMPHOMA
Depending on the stage, risk factors, and frontline regimen utilized, between 5% and 40% of patients with Hodgkin lymphoma can be expected to experience either primary induction failure or a relapse after attaining remission with frontline therapy.3 Primary refractory Hodgkin lymphoma, which occurs in up to 5% to 10% of patients, is defined as progression or no response during induction treatment or within 90 days of completing treatment. In cases where remission status is in question, an updated tissue biopsy is recommended. Biopsy is also recommended in cases in which new sites of disease have appeared or if relapse has occurred after a durable period of remission. Restaging is recommended at the time of relapse.
For younger patients with relapsed/refractory Hodgkin lymphoma, the standard of care in most cases is second-line (or salvage) chemotherapy followed by high-dose therapy and auto-HCT. For patients not felt to be candidates for auto-HCT, options include conventional second-line chemotherapy alone, salvage radiotherapy, novel agents such as brentuximab or immune checkpoint inhibitors, and/or participation in clinical trials.
CONVENTIONAL MULTI-AGENT CHEMOTHERAPY REGIMENS
Numerous conventional regimens have been shown in phase 2 studies to be active in relapsed and refractory Hodgkin lymphoma. These include platinum-based regimens, gemcitabine-based regimens, and alkylator-based regimens. No randomized trials in Hodgkin lymphoma have been conducted comparing these regimens. In general, regimens are chosen based on the patient’s age, performance status, comorbidities, and whether auto-HCT is being considered.
In the United States, platinum-based regimens such as ICE (ifosfamide, carboplatin, etoposide),42 DHAP (dexamethasone, cisplatin, high-dose cytarabine),43 ESHAP (etoposide, methylprednisolone, high-dose cytarabine, cisplatin),44 GDP (gemcitabine, cisplatin, dexamethasone),45 and GCD (gemcitabine, carboplatin, dexamethasone)46 are all considered appropriate second-line therapy options for patients being considered for auto-HCT, due to their high response rates and because autologous hematopoietic stem cell collection remains feasible after these regimens. Response rates range from 60% to 88%, with CR rates between 17% and 41%, and toxic death rates generally well below 5%.
Other gemcitabine-based regimens such as IGEV (ifosfamide, gemcitabine, vinorelbine) and GVD (gemcitabine, vinorelbine, liposomal doxorubicin) are also effective.47,48 GVD is an excellent choice since it is a generally well-tolerated outpatient regimen with a 60% response rate even in heavily pretreated patients.48 Stem cell collection remains feasible after both IGEV and GVD as well. ABVD can produce CR in approximately 20% to 50% of patients initially treated with MOPP.49–51 In practice, however, most patients today with relapsed or refractory Hodgkin lymphoma have already received ABVD as part of their first-line therapy, and retreatment with ABVD is not a good option because it would be associated with prohibitively high cumulative doses of doxorubicin.
These multi-agent chemotherapy regimens may not be tolerated well in patients over age 65 to 70 years or those with significant underlying comorbidities. In recent years, bendamustine has emerged as one of the most active conventional agents for cHL, with overall response rates of 53% to 58% in heavily pre-treated patients.52,53 Bendamustine can generally be tolerated even in elderly patients as well.
Some centers, particularly in Europe, investigated aggressive salvage regimens such as mini-BEAM (carmustine, etoposide, cytarabine, melphalan)54 or dexa-BEAM (BEAM plus dexamethasone).55 These regimens, however, are associated with significant hematologic toxicity and high (2%–5%) treatment-related mortality. As a result, these are rarely used in the United States.
For patients who have progressed after (or are not candidates for) platinum- and/or gemcitabine-based therapy, older alkylator-based regimens such as MOPP, C-MOPP, or ChlVPP (chlorambucil, vinblastine, procarbazine, prednisone) can be considered.56–58 However, these regimens are associated with significant bone marrow suppression, and autologous hematopoietic stem cell collection may no longer be feasible after such regimens. Therefore, these regimens should only be given to patients not felt to be auto-HCT candidates, or patients for whom autologous hematopoietic stem cell collection has already been completed. Weekly vinblastine or single-agent gemcitabine are palliative chemotherapy options, with response rates in the 60% to 80% range. Patients can sometimes be maintained on such low-intensity palliative regimens for 6 to 12 months or longer.59,60
BRENTUXIMAB VEDOTIN
Several trials are evaluating incorporation of brentuximab into second-line therapy in transplant-eligible patients. These approaches have used brentuximab prior to, concurrent with, or following platinum-based chemotherapy.61 While there is currently no consensus on the optimal way to incorporate brentuximab into salvage therapy, it is possible that the use of brentuximab or other novel agents in salvage therapy may allow for avoidance of conventional chemotherapy in some patients. In addition, this may translate into more patients proceeding to auto-HCT in a PET negative state. PET negativity prior to auto-HCT is a powerful predictor of long-term remission after auto-HCT, so any intervention that increases the rate of PET negativity prior to auto-HCT would be expected to improve outcomes with auto-HCT.62–65
For patients not being considered for autoHCT, or those for whom platinum-based salvage therapy was ineffective, single-agent brentuximab is an excellent option. In 2 phase 2 studies, an overall response rate (ORR) of 60% to 75% (including a CR rate of 22%–34%) was seen in relapsed and refractory Hodgkin lymphoma patients.66 The US Food and Drug Administration (FDA) approved brentuximab vedotin in August 2011 for treatment of relapsed and refractory Hodgkin lymphoma, after a failed auto-HCT, or in patients who are not auto-HCT candidates and who have received at least 2 prior chemotherapy regimens. With more extended follow-up, it has become clear that a proportion of patients who achieve CR to brentuximab may maintain remission long-term—58% at 3 years and 38% at 5 years.67 These patients may in fact be cured, in many cases without having undergone allogeneic HCT (allo-HCT) after brentuximab.
PD-1 (IMMUNE CHECKPOINT) INHIBITORS
As discussed earlier, PD-L1/PD-L2 copy number alterations represent a disease-defining feature of cHL. Alterations in chromosome 9p24.1 increase the expression of PD-1 ligands PD-L1 and PD-L2. Nivolumab and pembrolizumab are PD-1-blocking antibodies, which have recently been FDA approved for relapsed and refractory cHL. In a study with 23 patients, with 78% of them relapsing after auto-HCT and 78% relapsing after brentuximab, nivolumab produced an objective response in 87% of the patients, with 17% achieving CR and 70% achieving PR. The rate of PFS was 86% at 24 weeks.68 Pembrolizumab, another PD-1 antagonist, was also tested in relapsed and refractory Hodgkin lymphoma. In the KEYNOTE-087 study (n = 210), pembrolizumab produced an ORR of 64% to 70% in 3 different cohorts of relapsed and refractory cHL patients. Overall CR rate was 22%.69 In general, these agents are well tolerated, although patients must be monitored closely for
inflammatory/autoimmune-type toxicities including skin rash, diarrhea/colitis, transaminitis, endocrine abnormalities, and pneumonitis. Prompt recognition and initiation of corticosteroids is essential in managing these toxicities. Of note, PD-1 inhibitors should be given very cautiously to patients with a prior history of allo-HCT, since 30% to 55% of such patients will experience acute graft-versus-host disease (GVHD) in this setting. In 2 retrospective studies, the response rate was very high at 77% to 95%; however, 10% to 26% of all patients treated with PD-1 inhibitors post-allo-HCT died from GVHD induced by PD-1 inhibition.70,71 These risks and benefits therefore need to be carefully weighed in the post-allo-HCT setting. In another recent study, the outcomes were reported for 39 patients who underwent allo-HCT after prior therapy with a PD-1 inhibitor. Three patients (7.7%) developed lethal acute GVHD, suggesting there may be an increased risk of GVHD in patients undergoing allo-HCT after prior PD-1 inhibitor therapy.72
AUTOLOGOUS STEM CELL TRANSPLANTATION
Several studies have shown an improved disease-free survival (DFS) or FFS in patients with relapsed cHL treated by auto-HCT as compared to those receiving conventional chemotherapy alone.55,73,74 Overall, for relapsed disease, one can expect an approximately 50% to 60% chance for DFS at 5 years post-transplant. In a retrospective, matched-pair analysis, FFP was 62% for auto-HCT patients, compared to 32% for conventional chemotherapy patients. OS, however, was similar for the 2 groups (47%–54%). Patients failing induction therapy or relapsing within 1 year were seen to benefit the most from auto-HCT, including an OS benefit.74
A European prospective randomized trial was conducted comparing conventional salvage therapy to auto-HCT. In this study, 161 patients with relapsed Hodgkin lymphoma were treated with 2 cycles of dexa-BEAM. Those with chemo-sensitive disease were then randomized to either 2 more cycles of dexa-BEAM or high-dose BEAM with auto-HCT. Auto-HCT was associated with an approximately 55% FFTF at 3 years, versus 34% with conventional chemotherapy alone.55 This benefit again was most apparent for patients relapsing within 1 year of completion of primary therapy, although an OS benefit was not seen with auto-HCT. For patients with late relapse (>1 year after completion of primary therapy), auto-HCT was associated with an approximately 75% FFTF at 3 years, versus 50% with chemotherapy alone. One other small randomized trial of auto-HCT in relapsed and refractory Hodgkin lymphoma also showed an improved 3-year EFS in favor of auto-HCT (53% versus 10%), again with no difference in OS.73
The lack of OS benefit seen in these studies suggests that auto-HCT at first or second relapse provides comparable outcomes. Auto-HCT offers the benefit of avoiding the long-term toxicities associated with multiple salvage regimens and the anxiety associated with multiple relapses. In addition, the treatment-related mortality with auto-HCT is now in the 1% to 2% range in younger patients, at centers that perform the procedure routinely. For all of these reasons, auto-HCT is commonly recommended by physicians for Hodgkin lymphoma patients in first or second relapse. In most cases, transplant is favored in first relapse, since waiting until second relapse may be associated with a lower chance of achieving CR and difficulty collecting sufficient hematopoietic stem cells. For patients with early relapse or primary refractory disease, an even stronger case can be made for auto-HCT as the best option to achieve sustained control of the disease. For patients with late relapse, conventional salvage therapy alone may be a reasonable option, particularly in older or frail patients, or those with significant comorbid conditions.
The optimal conditioning regimen for autoHCT for relapsed and refractory Hodgkin lymphoma remains undefined. No randomized trials have been performed comparing conditioning regimens for relapsed and refractory Hodgkin lymphoma. One retrospective study compared 92 patients with Hodgkin lymphoma who underwent auto-HCT using a total-body irradiation (TBI) regimen versus a chemotherapy-alone regimen. No difference in 5-year OS or EFS was seen.75 Given the lack of benefit seen with TBI, along with reports of increased rates of secondary malignancies and myelodysplasia with TBI,76 chemotherapy-alone conditioning regimens are most widely employed. For example, in the United States, either the BEAM or CBV (cyclophosphamide, carmustine, etoposide) regimens are used in over 80% of cases.77 This practice was justified in a Center for International Blood and Marrow Transplant Research (CIBMTR) retrospective study comparing outcomes by conditioning regimens, in which no regimen performed better than BEAM or CBV.78
IFRT is often given as an adjunctive therapy to sites of initial and/or relapsed disease following auto-HCT. Although a relatively common practice, whether this truly enhances outcomes beyond that obtained with auto-HCT alone is unclear. Two retrospective studies have shown some benefit in terms of improvement in OS at 3 to 5 years in the group that received IFRT (70%–73% versus 40%–56%).79,80 Given the retrospective nature and small size of these studies, a prospective study would be needed to properly define the potential role for IFRT following auto-HCT in relapsed/refractory Hodgkin lymphoma. Another retrospective study (n = 73) that evaluated peri-transplant IFRT in Hodgkin lymphoma patients receiving auto transplant found no improvement in survival for patients who received peri-transplant IFRT. This study, however, did show a survival benefit in the subgroup of patients with limited stage disease.81
Prognostic Factors Associated with Outcome with Auto-HCT
The factor most consistently associated with improved outcome for patients with relapsed and refractory Hodgkin lymphoma who undergo auto-HCT is the disease status at transplant.63,77 Those in a second CR, versus a chemo-sensitive relapse (but not CR), versus a chemo-refractory relapse have DFS rates of 60% to 70%, 30% to 40%, and 10% to 20%, respectively.63 The duration between remission and relapse also has important prognostic significance. Late relapse (> 1 year after completion of frontline therapy) is associated with better outcomes as compared to early relapse.55 Other factors with prognostic significance at relapse include anemia, time to relapse and clinical stage, B symptoms, extranodal disease, number of prior chemotherapy regimens, and performance status.42,82 The prognostic impact of pretransplant disease status has been confirmed by studies using functional imaging (eg, FDG-PET or gallium scans). In a report by Moskowitz et al, patients with negative functional imaging following second-line therapy had a 77% EFS post-auto-HCT versus 33% in those whose functional imaging remained positive.62 Very similar findings have been reported by other groups.63–65
Post-Auto-HCT Brentuximab Maintenance
In the multicenter, randomized, double-blinded phase 3 AETHERA trial (n = 329), brentuximab (n = 165) was compared with placebo (n = 164) in patients with unfavorable risk relapsed or primary refractory cHL who had undergone autologous transplant. Eligible patients had at least 1 of the following risk factors for progression after auto-HCT: primary refractory Hodgkin lymphoma (failure to achieve complete remission), relapsed Hodgkin lymphoma with an initial remission duration of less than 12 months, or extranodal involvement at the start of pre-transplantation salvage chemotherapy. Patients were required to have CR, PR, or stable disease after pretransplant salvage chemotherapy with adequate kidney, liver, and bone marrow function. Patients who previously received brentuximab were excluded. Patients received 16 cycles of brentuximab or placebo once every 3 weeks starting 30 to 45 days after transplant. The PFS was significantly improved in the brentuximab group when compared to the placebo group (hazard ratio 0.57; P = 0.0013) after a median observation time of 30 months. Median PFS was 42.9 months in the brentuximab group versus 24.1 months in the placebo group; estimated 2-year PFS rates were 63% in the brentuximab group and 51% in the placebo group. OS was not significantly different between the study groups (~85%), presumably due to the fact that patients in the control group who relapsed likely went on to receive brentuximab as a subsequent therapy.83
PRIMARY REFRACTORY HODGKIN LYMPHOMA
Patients with primary refractory Hodgkin lymphoma have a poor outcome. Salvage therapy using conventional chemotherapy and/or RT results in long-term DFS in 10% or fewer of such patients.13,84 Given these poor outcomes with conventional salvage therapy, auto-HCT is considered to be the standard of care for this subset of patients. The GHSG retrospectively analyzed the prognostic factors and outcomes of patients with primary refractory Hodgkin lymphoma. The 5-year freedom-from-second-failure and the 5-year OS were reported to be 31% and 43%, respectively, for those patients treated with auto-HCT. Patients with poor functional status at time of transplant, age greater than 50 years, and failure to attain a temporary remission had a 0% 5-year OS, as compared to 55% in patients without any of these risk factors.85 A large retrospective European study showed that patients with chemo-resistant disease who underwent transplant had a 19% survival at 5 years.63 Hence, even patients with primary refractory Hodgkin lymphoma have some chance of achieving long-term survival following auto-HCT.
SALVAGE RADIOTHERAPY
The GHSG performed a retrospective analysis of the efficacy of salvage RT in patients with refractory or first-relapsed Hodgkin lymphoma. Five-year FFTF and OS rates were 28% and 51%, respectively. Patients with a limited-stage relapse and without B symptoms were more likely to benefit from salvage RT.86 Campbell et al reported on 81 patients undergoing salvage RT for persistent or recurrent Hodgkin lymphoma after chemotherapy. The 10-year FFTF and OS rates were 33% and 46%, respectively.87 Similarly, Wirth et al reported a 5-year FFS of 26% and 5-year OS of 57%. These figures were 36% and 75%, respectively, in patients whose relapse was limited to supradiaphragmatic nodal sites without B symptoms.88 RT therefore may be a useful strategy for a subset of patients who relapse following chemotherapy, particularly those with a limited-stage relapse, without B symptoms, and those with relapsed disease after a CR, as opposed to those with a partial response or lack of response to the prior chemotherapy regimen.
INVESTIGATIONAL AGENTS AND NOVEL COMBINATIONS
Several biological therapies are emerging as options for the treatment of refractory or relapsed disease. These therapies consist of monoclonal antibodies and ADCs that target cell surface antigens, or small molecules that inhibit key intracellular pathways within neoplastic cells.
Rituximab
Rituximab is a chimeric anti-CD20 monoclonal antibody used widely in B-cell non-Hodgkin lymphomas. The CD20 molecule is typically highly expressed in nodular lymphocyte-predominant Hodgkin lymphoma (NLPHL). Two studies (one in relapsed patients, the other in a mixture of relapsed and previously untreated patients) showed significant activity of rituximab in relapsed NLPHL, with ORRs ranging from 94% to 100%, CR rates ranging from 41% to 53%, and median duration of remission in the 10- to 33-month range.89,90 In cHL, CD20 is expressed in HRS cells in 20% to 30% of cases. In such cases, single-agent rituximab has also shown activity. There is also evidence that rituximab may be effective even in cases in which the HRS cells are CD20-negative, presumably by virtue of depleting reactive B lymphocytes from the microenvironment, which may enhance anti-tumor immunity, or by eliminating a putative CD20-expressing Hodgkin lymphoma stem cell.91,92
Lenalidomide
Lenalidomide is an immunomodulatory drug that has multiple modes of action, including direct induction of apoptosis in tumor cells, antiangiogenic effects, and the activation of immune cells, such as natural killer cells and T cells. Lenalidomide has been shown to modify many features of the microenvironment of HRS cells and has demonstrated activity in other B-cell neoplasms. As a result, lenalidomide has been evaluated in relapsed and refractory Hodgkin lymphoma patients. A multicenter phase 2 study by Fehniger et al included 35 patients, 87% of whom had previously undergone HCT and 55% of whom were refractory to the last therapy.93 All patients were given lenalidomide 25 mg/day from days 1 to 21 of a 28-day cycle until disease progression. One patient was noted to achieve CR, 6 achieved PR, and 5 had stable disease lasting more than 6 months, for an ORR of 19% and a “cytostatic overall response rate” of 33%. The median duration of CR/partial remission was 6 months, with the median time-to-treatment failure in responders (including those with stable disease > 6 months) being 15 months. Similarly, in another study, Böll et al evaluated 12 patients across 4 German centers with relapsed or refractory disease who were treated with oral lenalidomide for 21 days in a 28-day cycle. No radiological evidence of disease progression after 2 cycles of lenalidomide was seen in any of the enrolled patients. ORR was noted to be 50%, with 6 patients with stable disease and 5 patients achieving PR after 2 cycles.94
Novel Brentuximab Combination Therapies
Brentuximab plus bendamustine. The combination of brentuximab and bendamustine was tested as an outpatient regimen in a phase 1/2 study (n = 55) in primary refractory Hodgkin lymphoma or after first relapse. The CR rate of the combination was 74%, with an overall objective response (CR + PR) of 93%. The CR rates were 64% and 84%, respectively, for refractory and relapsed patients. The PFS at 12 months was 80%, establishing this combination therapy as an effective salvage regimen with durable response.95
Brentuximab plus nivolumab. Preliminary results have recently been presented from 2 studies96,97 evaluating the combination of brentuximab and nivolumab. While this combination would still be considered investigational, these studies showed very encouraging ORRs of 90% to 100% and a CR rate of 62% to 66%. Longer follow-up is needed to determine whether these responses are durable and to document the toxicity profile of this combination.
Mammalian Target of Rapamycin Inhibitors
Two mammalian target of rapamycin (mTOR) inhibitors, everolimus and temsirolimus, are currently available in the United States. While neither drug currently has FDA approval for Hodgkin lymphoma, everolimus was evaluated in a phase 2 trial in a heavily pretreated group of relapsed/refractory patients. An ORR of 47% was seen, with a median time to progression of 7.2 months.98
ALLOGENEIC STEM CELL TRANSPLANTATION
Historically, patients who relapse after having an auto-HCT generally had a poor outcome, with a median survival of 2 to 3 years after failure of auto-HCT.99 These patients may be offered palliative chemotherapy (see above), treatment with novel agents (see above), or enrollment in a clinical trial. Select patients may benefit from a second hematopoietic stem cell transplant, most commonly an allo-HCT. However, rare patients with late relapse after auto-HCT may be considered for a second auto-HCT, with a minority of such patients achieving a durable remission after the second auto-HCT.100,101 Because relapse or progressive disease occurs most commonly in the first several months following auto-HCT, patients are more often considered for allo-HCT than a second auto-HCT. In addition, a second auto-HCT may not be feasible due to impaired bone marrow reserve and/or concerns for development of secondary myelodysplasia or acute myeloid leukemia.
Several studies have evaluated allo-HCT in relapsed/ refractory Hodgkin lymphoma. Early studies evaluating myeloablative allo-HCT for Hodgkin lymphoma showed excessive treatment-related mortality (up to 50%) and disappointingly low rates of long-term survival (< 25%).102,103 This was likely related to the fact that, in that era, most of the patients with Hodgkin lymphoma evaluated for allo-HCT were heavily pretreated and therefore at a higher risk for toxicity as well as lymphoma progression.
More recently, several studies have focused on the use of reduced-intensity conditioning (RIC) allo-HCT for relapsed and refractory Hodgkin lymphoma. This approach relies more on a “graft-versus-lymphoma” effect, the existence of which has been debated in Hodgkin lymphoma. Three single-center studies of RIC allo-HCT in patients with multiply recurrent Hodgkin lymphoma showed improved rates of treatment-related mortality (8%–16%) but still relatively low rates of long-term PFS (23%–39% at 2 to 4 years).104–106 Interestingly, in one of these studies the outcomes were more favorable for patients who underwent haploidentical (versus matched sibling or matched unrelated donor) transplants.105
Two large registry studies have also reported on the outcomes of RIC allo-HCT in patients with relapsed and refractory Hodgkin lymphoma.107,108 These studies also confirmed a modest improvement in outcomes compared with those seen historically with myeloablative transplants. Treatment-related mortality at 1 to 2 years was 23% to 33%, depending on whether a matched sibling donor versus an unrelated donor was used. However, long-term PFS (18%–20% at 2 to 5 years) and OS (28%–37% at 2 to 5 years) remained poor, primarily due to high rates of progressive lymphoma post-transplant. In both of these studies, patients were heavily pretreated (84%–96% had received 3 or more prior lines of chemotherapy, and 62%–89% received a prior auto-HCT), with 47% to 55% of patients chemo-resistant prior to transplant. Of note, both of these registry studies reflect patients who underwent transplant prior to the widespread use of brentuximab and PD-1 inhibitors.
Based on the single-center and registry data above, a prospective multicenter European phase 2 trial was conducted to evaluate the benefit of RIC allo-HCT in Hodgkin lymphoma.109 Ninety-two patients (86% with prior auto-HCT, 90% with 3 or more prior lines of therapy) were enrolled and given salvage therapy. Those who had stable disease or better following salvage therapy remained on protocol (n = 78) and underwent RIC with fludarabine and melphalan, followed by allo-HCT (70% with matched sibling donors). Treatment-related mortality was 15% at 1 year. Relapse or progression occurred in 49% at 2 years (35% if chemo-sensitive prior to transplant). Chronic GVHD was associated with a decreased rate of relapse, supporting the existence of a graft-versus-lymphoma effect in Hodgkin lymphoma. Unfortunately, PFS among all allografted patients was still relatively poor (24% at 4 years). However, among patients in CR prior to allo-HCT, a 50% PFS was seen at 4 years. Therefore, even in a prospective multicenter study, RIC allo-HCT offered significant benefit with manageable toxicity in relapsed and refractory Hodgkin lymphoma patients with chemo-sensitive disease.
These studies suggest that outcomes with allo-HCT would improve further if implemented earlier in the course of disease and/or with a lower burden of disease at transplant. It has therefore been suggested that allo-HCT should be considered soon after failure of auto-HCT is documented. In a retrospective study by Sarina et al, 185 Hodgkin lymphoma patients who relapsed following auto-HCT were then immediately considered for reduced-intensity allo-HCT.110 Of these, 122 had a donor identified, and 104 (85%) actually underwent allo-HCT. These 104 patients were then compared to the other 81 patients who either had no donor identified or had a donor but did not receive the planned allo-HCT. Two-year PFS and OS were superior in the patients undergoing allo-HCT (39% versus 14% and 66% versus 42%, respectively, P < 0.001), with a median follow-up of 4 years. The presence of chronic GVHD again was associated with improved PFS and OS. Disease status prior to transplant remained highly predictive of PFS and OS by multivariate analysis. Two other smaller retrospective studies similarly found a survival benefit associated with allo-HCT compared with patients who underwent conventional salvage therapies alone.111,112 These studies, although subject to the usual limitations of retrospective analyses, suggest that the results with reduced-intensity allo-HCT are in fact enhanced if applied earlier in the disease course, and are superior to those with conventional therapy alone.
Currently, the exact role of allo-HSCT, including the optimal timing and optimal donor source (matched sibling versus haploidentical sibling versus matched unrelated donor), remain undefined for relapsed and refractory Hodgkin lymphoma. As discussed earlier, brentuximab is highly active in relapsed Hodgkin lymphoma patients, with a subset of patients still in CR at 5 years.67 For such patients, avoiding the risks of allo-HCT is a desirable goal.
For those who relapse or progress after auto-HCT, a reasonable strategy therefore is to treat initially with brentuximab, unless the patient is already known to have responded poorly to brentuximab, or already has significant neuropathy. Those who achieve a CR to brentuximab are then observed. A subset of those patients will remain in remission at 5 years without further therapy. For those who relapse, or who achieve less than a CR to brentuximab, additional treatment (with brentuximab re-treatment being one option) followed by a reduced-intensity allo-HCT is a reasonable consideration. This approach has the theoretical advantages of (1) avoiding the risk of allo-HCT in the subset potentially cured by brentuximab, (2) getting patients to allo-HCT with fewer comorbidities (due to a lower total exposure to conventional chemotherapy pre-transplant), and (3) applying allo-HCT in the setting of sensitive disease/lower disease burden (due to the high efficacy of brentuximab). The results of a small study suggest that brentuximab may in fact be a very effective “bridge” to allotransplant. Chen et al113 reported on 18 patients with relapsed/refractory Hodgkin lymphoma (17 of whom had previously undergone auto-HCT) who were treated on brentuximab vedotin clinical trials. The data were retrospectively evaluated to determine the efficacy and safety of subsequent reduced-intensity allo-HCT. Remarkably, at 1 year the OS was 100%, PFS was 92%, and nonrelapse mortality was 0% with a median follow-up of 14 months. Hence, brentuximab is safe for use prior to reduced-intensity allo-HCT in heavily pre-treated patients and appears to be associated with very favorable post-transplant outcomes, particularly in comparison to older studies of allo-HCT in the era prior to brentuximab.
SUMMARY
Currently, cure is possible for the majority of patients diagnosed with advanced stage Hodgkin lymphoma. The challenge to the clinician is to provide curative treatment with the lowest risk of serious toxicities. Which regimen will best provide this balance of risk and benefit needs to be assessed based on the relapse risk, age, frailty, and comorbidity profile for an individual patient. For many patients with relapsed or refractory Hodgkin lymphoma, cure remains possible using approaches based on hematopoietic stem cell transplantation, RT, and/or brentuximab. In addition, there are now numerous conventional chemotherapy agents, RT strategies, and exciting newer agents such as PD-1 inhibitors, that can provide significant clinical benefit even when cure is not feasible.
1. Kuppers R, Rajewsky K, Zhao M, et al. Hodgkin disease: Hodgkin and Reed-Sternberg cells picked from histological sections show clonal immunoglobulin gene rearrangements and appear to be derived from B cells at various stages of development. Proc Natl Acad Sci U S A 1994;91:10962–6.
2. Kuppers R. The biology of Hodgkin’s lymphoma. Nature Rev Cancer 2009;9:15–27.
3. Narra RK, Pingali SR, Fenske TS. Early-stage Hodgkin’s lymphoma. Hospital Physician Hematology-Oncology Board Review Manual. 2017;12(Part 3).
4. Mauch PM, Kalish LA, Kadin M, et al. Patterns of presentation of Hodgkin disease. Implications for etiology and pathogenesis. Cancer 1993;71:2062–71.
5. Hasenclever D, Diehl V. A prognostic score for advanced Hodgkin’s disease. International Prognostic Factors Project on Advanced Hodgkin’s Disease. N Engl J Med 1998;339:1506–14.
6. Moccia AA, Donaldson J, Chhanabhai M, et al. International Prognostic Score in advanced-stage Hodgkin’s lymphoma: altered utility in the modern era. J Clin Oncol 2012;30:3383–8.
7. Diefenbach CS, Li H, Hong F, et al. Evaluation of the International Prognostic Score (IPS-7) and a Simpler Prognostic Score (IPS-3) for advanced Hodgkin lymphoma in the modern era. Br J Haematol 2015;171:530–8.
8. Greaves P, Clear A, Coutinho R, et al. Expression of FOXP3, CD68, and CD20 at diagnosis in the microenvironment of cassical hodgkin lymphoma is predictive of outcome. J Clin Oncol 2013;31:256–62.
9. Touati M, Delage-Corre M, Monteil J, et al. CD68-positive tumor-associated macrophages predict unfavorable treatment outcomes in classical Hodgkin lymphoma in correlation with interim fluorodeoxyglucose-positron emission tomography assessment. Leuk Lymphoma 2015;56:332–41.
10. Roemer MG, Advani RH, Ligon AH, et al. PD-L1 and PD-L2 genetic alterations define classical Hodgkin lymphoma and predict outcome. J Clin Oncol 2016;34:2690–7.
11. Pike LC, Kirkwood AA, Patrick P, et al. Can baseline PET-CT features predict outcomes in advanced hodgkin lymphoma? A prospective evaluation of UK patients in The RATHL Trial (CRUK/07/033). Hematological Oncology 2017;35(Supplement S2):37–8.
12. DeVita VT Jr, Simon RM, Hubbard SM, et al. Curability of advanced Hodgkin’s disease with chemotherapy. Long-term follow-up of MOPP-treated patients at the National Cancer Institute. Ann Intern Med 1980;92:587–95.
13. Longo DL, Duffey PL, Young RC, et al. Conventional-dose salvage combination chemotherapy in patients relapsing with Hodgkin’s disease after combination chemotherapy: the low probability for cure. J Clin Oncol 1992;10:210–8.
14. Kaldor JM, Day NE, Clarke EA, et al. Leukemia following Hodgkin’s disease. N Engl J Med 1990;322:7–13.
15. Bonadonna G, Zucali R, Monfardini S, et al. Combination chemotherapy of Hodgkin’s disease with adriamycin, bleomycin, vinblastine, and imidazole carboxamide versus MOPP. Cancer 1975;36:252–9.
16. Canellos GP, Anderson JR, Propert KJ, et al. Chemotherapy of advanced Hodgkin’s disease with MOPP, ABVD, or MOPP alternating with ABVD. N Engl J Med 1992;327:1478–84.
17. Horning SJ, Williams J, Bartlett NL, et al. Assessment of the stanford V regimen and consolidative radiotherapy for bulky and advanced Hodgkin’s disease: Eastern Cooperative Oncology Group pilot study E1492. J Clin Oncol 2000;18:972–80.
18. Hoskin PJ, Lowry L, Horwich A, et al. Randomized comparison of the stanford V regimen and ABVD in the treatment of advanced Hodgkin’s Lymphoma: United Kingdom National Cancer Research Institute Lymphoma Group Study ISRCTN 64141244. J Clin Oncol 2009;27:5390–6.
19. Gordon LI, Hong F, Fisher RI, et al. Randomized phase III trial of ABVD versus Stanford V with or without radiation therapy in locally extensive and advanced-stage Hodgkin lymphoma: an intergroup study coordinated by the Eastern Cooperative Oncology Group (E2496). J Clin Oncol 2013;31:684–91.
20. Engert A, Diehl V, Franklin J, et al. Escalated-dose BEACOPP in the treatment of patients with advanced-stage Hodgkin’s lymphoma: 10 years of follow-up of the GHSG HD9 study. J Clin Oncol 2009;27:4548–54.
21. Merli F, Luminari S, Gobbi PG, et al. Long-term results of the HD2000 trial comparing ABVD versus BEACOPP versus COPP-EBV-CAD in untreated patients with advanced Hodgkin lymphoma: a study by Fondazione Italiana Linfomi. J Clin Oncol 2016;34:1175–81.
22. Viviani S, Zinzani PL, Rambaldi A, et al. ABVD versus BEACOPP for Hodgkin’s lymphoma when high-dose salvage is planned. N Engl J Med 2011;365:203–12.
23. Carde P, Karrasch M, Fortpied C, et al. Eight cycles of ABVD versus four cycles of BEACOPPescalated plus four cycles of BEACOPPbaseline in stage III to IV, International Prognostic Score >/= 3, high-risk Hodgkin lymphoma: first results of the phase III EORTC 20012 intergroup trial. J Clin Oncol 2016;34:2028–36.
24. National Comprehensive Cancer Network I. NCCN Guidelines Version 1.2017 Hodgkin Lymphoma. Accessed July 20, 2017.
25. Younes A, Connors JM, Park SI, et al. Brentuximab vedotin combined with ABVD or AVD for patients with newly diagnosed Hodgkin’s lymphoma: a phase 1, open-label, dose-escalation study. Lancet Oncology 2013;14:1348–56.
26. Takeda and Seattle Genetics announce positive results from phase 3 ECHELON-1 clinical trial evaluating ADCETRIS® (brentuximab vedotin) in frontline advanced Hodgkin lymphoma [press release]. Cambridge, MA: Takeda Oncology; June 26, 2017.
27. Boll B, Bredenfeld H, Gorgen H, et al. Phase 2 study of PVAG (prednisone, vinblastine, doxorubicin, gemcitabine) in elderly patients with early unfavorable or advanced stage Hodgkin lymphoma. Blood 2011;118:6292–8.
28. Behringer K, Goergen H, Hitz F, et al. Omission of dacarbazine or bleomycin, or both, from the ABVD regimen in treatment of early-stage favourable Hodgkin’s lymphoma (GHSG HD13): an open-label, randomised, non-inferiority trial. Lancet 2015;385:1418–27.
29. Forero-Torres A, Holkova B, Goldschmidt J, et al. Phase 2 study of frontline brentuximab vedotin monotherapy in Hodgkin lymphoma patients aged 60 years and older. Blood 2015;126:2798–804.
30. Gallamini A, Hutchings M, Rigacci L, et al. Early interim 2-[18F]fluoro-2-deoxy-D-glucose positron emission tomography is prognostically superior to international prognostic score in advanced-stage Hodgkin’s lymphoma: a report from a joint Italian-Danish study. J Clin Oncol 2007;25:3746–52.
31. Meignan M, Gallamini A, Meignan M, et al. Report on the First International Workshop on Interim-PET-Scan in Lymphoma. Leuk Lymphoma 2009;50:1257–60.
32. Barrington SF, Mikhaeel NG, Kostakoglu L, et al. Role of Imaging in the Staging and Response Assessment of Lymphoma: Consensus of the International Conference on Malignant Lymphomas Imaging Working Group. J Clin Oncol 2014;32:3048–58.
33. Press OW, Li H, Schoder H, et al. US Intergroup trial of response-adapted therapy for stage III to IV Hodgkin lymphoma using early interim fluorodeoxyglucose-positron emission tomography imaging: Southwest Oncology Group S0816. J Clin Oncol 2016;34:2020–7.
34. Zinzani PL, Broccoli A, Gioia DM, et al. Interim positron emission tomography response-adapted therapy in advanced-stage Hodgkin lymphoma: final results of the pPhase II part of the HD0801 study. J Clin Oncol 2016;34:1376–85.
35. Borchmann P, Haverkamp H, Lohri A, et al. Progression-free survival of early interim PET-positive patients with advanced stage Hodgkin’s lymphoma treated with BEACOPPescalated alone or in combination with rituximab (HD18): an open-label, international, randomised phase 3 study by the German Hodgkin Study Group. Lancet Oncology 2017;18:454–63.
36. Aleman BM, Raemaekers JM, Tirelli U, et al. Involved-field radiotherapy for advanced Hodgkin’s lymphoma. N Engl J Med 2003;348:2396–406.
37. Borchmann P, Haverkamp H, Diehl V, et al. Eight cycles of escalated-dose BEACOPP compared with four cycles of escalated-dose BEACOPP followed by four cycles of baseline-dose BEACOPP with or without radiotherapy in patients with advanced-stage hodgkin’s lymphoma: final analysis of the HD12 trial of the German Hodgkin Study Group. J Clin Oncol 2011;29:4234–42.
38. Engert A, Haverkamp H, Kobe C, et al. Reduced-intensity chemotherapy and PET-guided radiotherapy in patients with advanced stage Hodgkin’s lymphoma (HD15 trial): a randomised, open-label, phase 3 non-inferiority trial. Lancet 2012;379:1791–9.
39. Nademanee A, Molina A, Fung H, et al. High-dose chemo/radiotherapy and autologous bone marrow or stem cell transplantation for poor-risk advanced-stage Hodgkin’s disease during first partial or complete remission. Biol Blood Marrow Transplant 1999;5:292–8.
40. Federico M, Bellei M, Brice P, et al. High-dose therapy and autologous stem-cell transplantation versus conventional therapy for patients with advanced Hodgkin’s lymphoma responding to front-line therapy. J Clin Oncol 2003;21:2320–5.
41. Arakelyan N, Berthou C, Desablens B, et al. Early versus late intensification for patients with high-risk Hodgkin lymphoma-3 cycles of intensive chemotherapy plus low-dose lymph node radiation therapy versus 4 cycles of combined doxorubicin, bleomycin, vinblastine, and dacarbazine plus myeloablative chemotherapy with autologous stem cell transplantation: five-year results of a randomized trial on behalf of the GOELAMS Group. Cancer 2008;113:3323–30.
42. Moskowitz CH, Nimer SD, Zelenetz AD, et al. A 2-step comprehensive high-dose chemoradiotherapy second-line program for relapsed and refractory Hodgkin disease: analysis by intent to treat and development of a prognostic model. Blood 2001;97:616–23.
43. Josting A, Rudolph C, Reiser M, et al. Time-intensified dexamethasone/cisplatin/cytarabine: an effective salvage therapy with low toxicity in patients with relapsed and refractory Hodgkin’s disease. Ann Oncol 2002;13:1628–35.
44. Aparicio J, Segura A, Garcera S, et al. ESHAP is an active regimen for relapsing Hodgkin’s disease. Ann Oncol 1999;10:593–5.
45. Baetz T, Belch A, Couban S, et al. Gemcitabine, dexamethasone and cisplatin is an active and non-toxic chemotherapy regimen in relapsed or refractory Hodgkin’s disease: a phase II study by the National Cancer Institute of Canada Clinical Trials Group. Ann Oncol 2003;14:1762–7.
46. Gopal AK, Press OW, Shustov AR, et al. Efficacy and safety of gemcitabine, carboplatin, dexamethasone, and rituximab in patients with relapsed/refractory lymphoma: a prospective multi-center phase II study by the Puget Sound Oncology Consortium. Leuk Lymphoma 2010;51:1523–9.
47. Santoro A, Magagnoli M, Spina M, et al. Ifosfamide, gemcitabine, and vinorelbine: a new induction regimen for refractory and relapsed Hodgkin’s lymphoma. Haematologica 2007;92:35–41.
48. Bartlett NL, Niedzwiecki D, Johnson JL, et al. Gemcitabine, vinorelbine, and pegylated liposomal doxorubicin (GVD), a salvage regimen in relapsed Hodgkin’s lymphoma: CALGB 59804. Ann Oncol 2007;18:1071–9.
49. Santoro A, Bonadonna G. Prolonged disease-free survival in MOPP-resistant Hodgkin’s disease after treatment with adriamycin, bleomycin, vinblastine and dacarbazine (ABVD). Cancer Chemother Pharmacol 1979;2:101–5.
50. Krikorian JG, Portlock CS, Rosenberg SA. Treatment of advanced Hodgkin’s disease with adriamycin, bleomycin, vinblastine, and imidazole carboxamide (ABVD) after failure of MOPP therapy. Cancer 1978;41:2107–11.
51. Piga A, Ambrosetti A, Todeschini G, et al. Doxorubicin, bleomycin, vinblastine, and dacarbazine (ABVD) salvage of mechlorethamine, vincristine, prednisone, and procarbazine (MOPP)-resistant advanced Hodgkin’s disease. Cancer Treat Rep 1984;68:947–51.
52. Moskowitz AJ, Hamlin PA Jr, Perales MA, et al. Phase II study of bendamustine in relapsed and refractory Hodgkin lymphoma. J Clin Oncol 2013;31:456–60.
53. Anastasia A, Carlo-Stella C, Corradini P, et al. Bendamustine for relapsed/refractory classical Hodgkin lymphoma after high dose chemotherapy and or allogeneic transplant: a study of Fondazione Italiana Linfomi (FIL). Blood 2012;120:3652.
54. Martin A, Fernandez-Jimenez MC, Caballero MD, et al. Long-term follow-up in patients treated with Mini-BEAM as salvage therapy for relapsed or refractory Hodgkin’s disease. Br J Haematol 2001;113:161–71.
55. Schmitz N, Pfistner B, Sextro M, et al. Aggressive conventional chemotherapy compared with high-dose chemotherapy with autologous haemopoietic stem-cell transplantation for relapsed chemosensitive Hodgkin’s disease: a randomised trial. Lancet 2002;359:2065–71.
56. Fisher RI, DeVita VT, Hubbard SP, et al. Prolonged disease-free survival in Hodgkin’s disease with MOPP reinduction after first relapse. Ann Intern Med 1979;90:761–3.
57. ChlVPP therapy for Hodgkin’s disease: experience of 960 patients. The International ChlVPP Treatment Group. Ann Oncol 1995;6:167–72.
58. Selby P, Patel P, Milan S, et al. ChlVPP combination chemotherapy for Hodgkin’s disease: long-term results. Br J Cancer 1990;62:279–85.
59. Santoro A, Bredenfeld H, Devizzi L, et al. Gemcitabine in the treatment of refractory Hodgkin’s disease: results of a multicenter phase II study. J Clin Oncol 2000;18:2615–9.
60. Little R, Wittes RE, Longo DL, Wilson WH. Vinblastine for recurrent Hodgkin’s disease following autologous bone marrow transplant. J Clin Oncol 1998;16:584–8.
61. Chen R, Palmer JM, Martin P, et al. Results of a multicenter phase II trial of brentuximab vedotin as second-line therapy before autologous transplantation in relapsed/refractory Hodgkin lymphoma. Biol Blood Marrow Transplant 2015;21:2136–40.
62. Moskowitz CH, Yahalom J, Zelenetz AD, et al. High-dose chemo-radiotherapy for relapsed or refractory Hodgkin lymphoma and the significance of pre-transplant functional imaging. Br J Haematol 2010;148:890–7.
63. Sureda A, Constans M, Iriondo A, et al. Prognostic factors affecting long-term outcome after stem cell transplantation in Hodgkin’s lymphoma autografted after a first relapse. Ann Oncol 2005;16:625–33.
64. Crocchiolo R, Canevari C, Assanelli A, et al. Pre-transplant 18FDG-PET predicts outcome in lymphoma patients treated with high-dose sequential chemotherapy followed by autologous stem cell transplantation. Leuk Lymphoma 2008;49:727–33.
65. Mocikova H, Pytlik R, Markova J, et al. Pre-transplant positron emission tomography in patients with relapsed Hodgkin lymphoma. Leuk Lymphoma 2011;52:1668–74.
66. Younes A, Gopal AK, Smith SE, et al. Results of a pivotal phase II study of brentuximab vedotin for patients with relapsed or refractory Hodgkin’s lymphoma. J Clin Oncol 2012;30:2183–9.
67. Gopal AK, Chen R, Smith SE, et al. Durable remissions in a pivotal phase 2 study of brentuximab vedotin in relapsed or refractory Hodgkin lymphoma. Blood 2015;125:1236–43.
68. Ansell SM, Lesokhin AM, Borrello I, et al. PD-1 blockade with nivolumab in relapsed or refractory Hodgkin’s lymphoma.N Engl J Med 2015;372:311–9.
69. Chen R, Zinzani PL, Fanale MA, et al. Phase II study of the efficacy and safety of pembrolizumab for relapsed/refractory classic Hodgkin lymphoma. J Clin Oncol 2017;35:2125–32.
70. Haverkos BM, Abbott D, Hamadani M, et al. PD-1 blockade for relapsed lymphoma post-allogeneic hematopoietic cell transplant: high response rate but frequent GVHD. Blood 2017;130:221–8.
71. Herbaux C, Gauthier J, Brice P, et al. Efficacy and tolerability of nivolumab after allogeneic transplantation for relapsed Hodgkin lymphoma. Blood 2017;129:2471–8.
72. Merryman RW, Kim HT, Zinzani PL et al. Safety and efficacy of allogeneic hematopoietic stem cell transplant after PD-1 blockage in relapsed/refractory lymphoma. Blood 2017;129:1380–8.
73. Linch DC, Winfield D, Goldstone AH, et al. Dose intensification with autologous bone-marrow transplantation in relapsed and resistant Hodgkin’s disease: results of a BNLI randomised trial. Lancet 1993;341:1051–4.
74. Yuen AR, Rosenberg SA, Hoppe RT, et al. Comparison between conventional salvage therapy and high-dose therapy with autografting for recurrent or refractory Hodgkin’s disease. Blood 1997;89:814–22.
75. Gutierrez-Delgado F, Holmberg L, Hooper H, et al. Autologous stem cell transplantation for Hodgkin’s disease: busulfan, melphalan and thiotepa compared to a radiation-based regimen. Bone Marrow Transplant 2003;32:279–85.
76. Hake CR, Graubert TA, Fenske TS. Does autologous transplantation directly increase the risk of secondary leukemia in lymphoma patients? Bone Marrow Transplant 2007;39:59–70.
77. Hahn T, McCarthy PL, Carreras J, et al. Comparison of prognostic models for autologous hematopoietic stem cell transplantation (AHCT) for relapsed Hodgkin lymphoma. Blood 2009;114:1215.
78. Chen Y-B, Lane AA, Logan BR, et al. Impact of conditioning regimen on outcomes for patients with lymphoma undergoing high-dose therapy with autologous hematopoietic cell transplantation. Biology Blood Marrow Transplant 2015;21:1046–53.
79. Wendland MM, Asch JD, Pulsipher MA, et al. The impact of involved field radiation therapy for patients receiving high-dose chemotherapy followed by hematopoietic progenitor cell transplant for the treatment of relapsed or refractory Hodgkin disease. Am J Clin Oncol 2006;29:189–95.
80. Biswas T, Culakova E, Friedberg JW, et al. Involved field radiation therapy following high dose chemotherapy and autologous stem cell transplant benefits local control and survival in refractory or recurrent Hodgkin lymphoma. Radiother Oncol 2012;103:367–72.
81. Levis M, Piva C, Filippi AR, et al. Potential benefit of involved-field radiotherapy for patients with Relapsed-refractory Hodgkin’s lymphoma with incomplete response before autologous stem cell transplantation. Clin Lymphoma Myeloma Leuk 2017;17:14–22.
82. Josting A, Franklin J, May M, et al. New prognostic score based on treatment outcome of patients with relapsed Hodgkin’s lymphoma registered in the database of the German Hodgkin’s lymphoma study group. J Clin Oncol 2002;20:221–30.
83. Moskowitz CH, Nademanee A, Masszi T, et al. Brentuximab vedotin as consolidation therapy after autologous stem-cell transplantation in patients with Hodgkin’s lymphoma at risk of relapse or progression (AETHERA): a randomised, double-blind, placebo-controlled, phase 3 trial. Lancet 2015;385:1853–62.
84. Bonfante V, Santoro A, Viviani S, et al. Outcome of patients with Hodgkin’s disease failing after primary MOPP-ABVD. J Clin Oncol 1997;15:528–34.
85. Josting A, Rueffer U, Franklin J, et al. Prognostic factors and treatment outcome in primary progressive Hodgkin lymphoma: a report from the German Hodgkin Lymphoma Study Group. Blood 2000;96:1280–6.
86. Josting A, Nogova L, Franklin J, et al. Salvage radiotherapy in patients with relapsed and refractory Hodgkin’s lymphoma: a retrospective analysis from the German Hodgkin Lymphoma Study Group. J Clin Oncol 2005;23:1522–9.
87. Campbell B WA, Milner A, Di Iulio J, et al. Long-term follow-up of salvage radiotherapy in Hodgkin’s lymphoma after chemotherapy failure. Int J Radiat Oncol Biol Phys 2005;63:1538–45.
88. Wirth A, Corry J, Laidlaw C, et al. Salvage radiotherapy for Hodgkin’s disease following chemotherapy failure. Int J Radiat Oncol Biol Phys 1997;39:599–607.
89. Schulz H, Rehwald U, Morschhauser F, et al. Rituximab in relapsed lymphocyte-predominant Hodgkin lymphoma: long-term results of a phase 2 trial by the German Hodgkin Lymphoma Study Group (GHSG). Blood 2008;111:109–11.
90. Ekstrand BC, Lucas JB, Horwitz SM, et al. Rituximab in lymphocyte-predominant Hodgkin disease: results of a phase 2 trial. Blood 2003;101:4285–9.
91. Younes A, Romaguera J, Hagemeister F, et al. A pilot study of rituximab in patients with recurrent, classic Hodgkin disease. Cancer 2003;98:310–4.
92. Rehwald U, Schulz H, Reiser M, et al. Treatment of relapsed CD20+ Hodgkin lymphoma with the monoclonal antibody rituximab is effective and well tolerated: results of a phase 2 trial of the German Hodgkin Lymphoma Study Group. Blood 2003;101:420–4.
93. Fehniger TA, Larson S, Trinkaus K, et al. A phase 2 multicenter study of lenalidomide in relapsed or refractory classical Hodgkin lymphoma. Blood 2011;118:5119–25.
94. Boll B, Borchmann P, Topp MS, et al. Lenalidomide in patients with refractory or multiple relapsed Hodgkin lymphoma. Br J Haematol 2010;148:480–2.
95. LaCasce AS, Bociek G, Sawas A, et al. Brentuximab vedotin plus bendamustine: a highly active salvage treatment regimen for patients with relapsed or refractory Hodgkin lymphoma. Blood 2015;126:3982.
96. Diefenbach CS, Hong F, David KA, et al. A phase I study with an expansion cohort of the combination of ipilimumab and nivolumab and brentuximab vedotin in patients with relapsed/refractory Hodgkin lymphoma: A trial of the ECOG-ACRIN Cancer Research Group (E4412 Arms D and E). Blood 2016;128:1106.
97. Herrera AF, Bartlett NL, Ramchandren R, et al. Preliminary results from a phase 1/2 study of brentuximab vedotin in combination with nivolumab in patients with relapsed or refractory Hodgkin lymphoma. Blood 2016;128:1105.
98. Johnston PB, Inwards DJ, Colgan JP, et al. A phase II trial of the oral mTOR inhibitor everolimus in relapsed Hodgkin lymphoma. Am J Hematol 2010;85:320–4.
99. Kewalramani T, Nimer SD, Zelenetz AD, et al. Progressive disease following autologous transplantation in patients with chemosensitive relapsed or primary refractory Hodgkin’s disease or aggressive non-Hodgkin’s lymphoma. Bone Marrow Transplant 2003;32:673–9.
100. Lin TS, Avalos BR, Penza SL, et al. Second autologous stem cell transplant for multiply relapsed Hodgkin’s disease. Bone Marrow Transplant 2002;29:763–7.
101. Smith SM, van Besien K, Carreras J, et al. Second autologous stem cell transplantation for relapsed lymphoma after a prior autologous transplant. Biol Blood Marrow Transplant 2008;14:904–12.
102. Gajewski JL, Phillips GL, Sobocinski KA, et al. Bone marrow transplants from HLA-identical siblings in advanced Hodgkin’s disease. J Clin Oncol 1996;14:572–8.
103. Peniket AJ, Ruiz de Elvira MC, Taghipour G, et al. An EBMT registry matched study of allogeneic stem cell transplants for lymphoma: allogeneic transplantation is associated with a lower relapse rate but a higher procedure-related mortality rate than autologous transplantation. Bone Marrow Transplant 2003;31:667–78.
104. Anderlini P, Saliba R, Acholonu S, et al. Fludarabine-melphalan as a preparative regimen for reduced-intensity conditioning allogeneic stem cell transplantation in relapsed and refractory Hodgkin’s lymphoma: the updated M.D. Anderson Cancer Center experience. Haematologica 2008;93:257–64.
105. Burroughs LM, O’Donnell PV, Sandmaier BM, et al. Comparison of outcomes of HLA-matched related, unrelated, or HLA-haploidentical related hematopoietic cell transplantation following nonmyeloablative conditioning for relapsed or refractory Hodgkin lymphoma. Biol Blood Marrow Transplant 2008;14:1279–87.
106. Peggs KS, Hunter A, Chopra R, et al. Clinical evidence of a graft-versus-Hodgkin’s-lymphoma effect after reduced-intensity allogeneic transplantation. Lancet 2005;365:1934–41.
107. Sureda A, Robinson S, Canals C, et al. Reduced-intensity conditioning compared with conventional allogeneic stem-cell transplantation in relapsed or refractory Hodgkin’s lymphoma: an analysis from the Lymphoma Working Party of the European Group for Blood and Marrow Transplantation. J Clin Oncol 2008;26:455–62.
108. Devetten MP, Hari PN, Carreras J, et al. Unrelated donor reduced-intensity allogeneic hematopoietic stem cell transplantation for relapsed and refractory Hodgkin lymphoma. Biol Blood Marrow Transplant 2009;15:109–17.
109. Sureda A, Canals C, Arranz R, et al. Allogeneic stem cell transplantation after reduced intensity conditioning in patients with relapsed or refractory Hodgkin’s lymphoma. Results of the HDR-ALLO study - a prospective clinical trial by the Grupo Espanol de Linfomas/Trasplante de Medula Osea (GEL/TAMO) and the Lymphoma Working Party of the European Group for Blood and Marrow Transplantation. Haematologica 2012;97:310–7.
110. Sarina B, Castagna L, Farina L, et al. Allogeneic transplantation improves the overall and progression-free survival of Hodgkin lymphoma patients relapsing after autologous transplantation: a retrospective study based on the time of HLA typing and donor availability. Blood 2010;115:3671–7.
111. Castagna L, Sarina B, Todisco E, et al. Allogeneic stem cell transplantation compared with chemotherapy for poor-risk Hodgkin lymphoma. Biol Blood Marrow Transplant 2009;15:432–8.
112. Thomson KJ, Peggs KS, Smith P, et al. Superiority of reduced-intensity allogeneic transplantation over conventional treatment for relapse of Hodgkin’s lymphoma following autologous stem cell transplantation. Bone Marrow Transplant 2008;41:765–70.
113. Chen R, Palmer JM, Thomas SH, et al. Brentuximab vedotin enables successful reduced-intensity allogeneic hematopoietic cell transplantation in patients with relapsed or refractory Hodgkin lymphoma. Blood 2012;119:6379–81.
1. Kuppers R, Rajewsky K, Zhao M, et al. Hodgkin disease: Hodgkin and Reed-Sternberg cells picked from histological sections show clonal immunoglobulin gene rearrangements and appear to be derived from B cells at various stages of development. Proc Natl Acad Sci U S A 1994;91:10962–6.
2. Kuppers R. The biology of Hodgkin’s lymphoma. Nature Rev Cancer 2009;9:15–27.
3. Narra RK, Pingali SR, Fenske TS. Early-stage Hodgkin’s lymphoma. Hospital Physician Hematology-Oncology Board Review Manual. 2017;12(Part 3).
4. Mauch PM, Kalish LA, Kadin M, et al. Patterns of presentation of Hodgkin disease. Implications for etiology and pathogenesis. Cancer 1993;71:2062–71.
5. Hasenclever D, Diehl V. A prognostic score for advanced Hodgkin’s disease. International Prognostic Factors Project on Advanced Hodgkin’s Disease. N Engl J Med 1998;339:1506–14.
6. Moccia AA, Donaldson J, Chhanabhai M, et al. International Prognostic Score in advanced-stage Hodgkin’s lymphoma: altered utility in the modern era. J Clin Oncol 2012;30:3383–8.
7. Diefenbach CS, Li H, Hong F, et al. Evaluation of the International Prognostic Score (IPS-7) and a Simpler Prognostic Score (IPS-3) for advanced Hodgkin lymphoma in the modern era. Br J Haematol 2015;171:530–8.
8. Greaves P, Clear A, Coutinho R, et al. Expression of FOXP3, CD68, and CD20 at diagnosis in the microenvironment of cassical hodgkin lymphoma is predictive of outcome. J Clin Oncol 2013;31:256–62.
9. Touati M, Delage-Corre M, Monteil J, et al. CD68-positive tumor-associated macrophages predict unfavorable treatment outcomes in classical Hodgkin lymphoma in correlation with interim fluorodeoxyglucose-positron emission tomography assessment. Leuk Lymphoma 2015;56:332–41.
10. Roemer MG, Advani RH, Ligon AH, et al. PD-L1 and PD-L2 genetic alterations define classical Hodgkin lymphoma and predict outcome. J Clin Oncol 2016;34:2690–7.
11. Pike LC, Kirkwood AA, Patrick P, et al. Can baseline PET-CT features predict outcomes in advanced hodgkin lymphoma? A prospective evaluation of UK patients in The RATHL Trial (CRUK/07/033). Hematological Oncology 2017;35(Supplement S2):37–8.
12. DeVita VT Jr, Simon RM, Hubbard SM, et al. Curability of advanced Hodgkin’s disease with chemotherapy. Long-term follow-up of MOPP-treated patients at the National Cancer Institute. Ann Intern Med 1980;92:587–95.
13. Longo DL, Duffey PL, Young RC, et al. Conventional-dose salvage combination chemotherapy in patients relapsing with Hodgkin’s disease after combination chemotherapy: the low probability for cure. J Clin Oncol 1992;10:210–8.
14. Kaldor JM, Day NE, Clarke EA, et al. Leukemia following Hodgkin’s disease. N Engl J Med 1990;322:7–13.
15. Bonadonna G, Zucali R, Monfardini S, et al. Combination chemotherapy of Hodgkin’s disease with adriamycin, bleomycin, vinblastine, and imidazole carboxamide versus MOPP. Cancer 1975;36:252–9.
16. Canellos GP, Anderson JR, Propert KJ, et al. Chemotherapy of advanced Hodgkin’s disease with MOPP, ABVD, or MOPP alternating with ABVD. N Engl J Med 1992;327:1478–84.
17. Horning SJ, Williams J, Bartlett NL, et al. Assessment of the stanford V regimen and consolidative radiotherapy for bulky and advanced Hodgkin’s disease: Eastern Cooperative Oncology Group pilot study E1492. J Clin Oncol 2000;18:972–80.
18. Hoskin PJ, Lowry L, Horwich A, et al. Randomized comparison of the stanford V regimen and ABVD in the treatment of advanced Hodgkin’s Lymphoma: United Kingdom National Cancer Research Institute Lymphoma Group Study ISRCTN 64141244. J Clin Oncol 2009;27:5390–6.
19. Gordon LI, Hong F, Fisher RI, et al. Randomized phase III trial of ABVD versus Stanford V with or without radiation therapy in locally extensive and advanced-stage Hodgkin lymphoma: an intergroup study coordinated by the Eastern Cooperative Oncology Group (E2496). J Clin Oncol 2013;31:684–91.
20. Engert A, Diehl V, Franklin J, et al. Escalated-dose BEACOPP in the treatment of patients with advanced-stage Hodgkin’s lymphoma: 10 years of follow-up of the GHSG HD9 study. J Clin Oncol 2009;27:4548–54.
21. Merli F, Luminari S, Gobbi PG, et al. Long-term results of the HD2000 trial comparing ABVD versus BEACOPP versus COPP-EBV-CAD in untreated patients with advanced Hodgkin lymphoma: a study by Fondazione Italiana Linfomi. J Clin Oncol 2016;34:1175–81.
22. Viviani S, Zinzani PL, Rambaldi A, et al. ABVD versus BEACOPP for Hodgkin’s lymphoma when high-dose salvage is planned. N Engl J Med 2011;365:203–12.
23. Carde P, Karrasch M, Fortpied C, et al. Eight cycles of ABVD versus four cycles of BEACOPPescalated plus four cycles of BEACOPPbaseline in stage III to IV, International Prognostic Score >/= 3, high-risk Hodgkin lymphoma: first results of the phase III EORTC 20012 intergroup trial. J Clin Oncol 2016;34:2028–36.
24. National Comprehensive Cancer Network I. NCCN Guidelines Version 1.2017 Hodgkin Lymphoma. Accessed July 20, 2017.
25. Younes A, Connors JM, Park SI, et al. Brentuximab vedotin combined with ABVD or AVD for patients with newly diagnosed Hodgkin’s lymphoma: a phase 1, open-label, dose-escalation study. Lancet Oncology 2013;14:1348–56.
26. Takeda and Seattle Genetics announce positive results from phase 3 ECHELON-1 clinical trial evaluating ADCETRIS® (brentuximab vedotin) in frontline advanced Hodgkin lymphoma [press release]. Cambridge, MA: Takeda Oncology; June 26, 2017.
27. Boll B, Bredenfeld H, Gorgen H, et al. Phase 2 study of PVAG (prednisone, vinblastine, doxorubicin, gemcitabine) in elderly patients with early unfavorable or advanced stage Hodgkin lymphoma. Blood 2011;118:6292–8.
28. Behringer K, Goergen H, Hitz F, et al. Omission of dacarbazine or bleomycin, or both, from the ABVD regimen in treatment of early-stage favourable Hodgkin’s lymphoma (GHSG HD13): an open-label, randomised, non-inferiority trial. Lancet 2015;385:1418–27.
29. Forero-Torres A, Holkova B, Goldschmidt J, et al. Phase 2 study of frontline brentuximab vedotin monotherapy in Hodgkin lymphoma patients aged 60 years and older. Blood 2015;126:2798–804.
30. Gallamini A, Hutchings M, Rigacci L, et al. Early interim 2-[18F]fluoro-2-deoxy-D-glucose positron emission tomography is prognostically superior to international prognostic score in advanced-stage Hodgkin’s lymphoma: a report from a joint Italian-Danish study. J Clin Oncol 2007;25:3746–52.
31. Meignan M, Gallamini A, Meignan M, et al. Report on the First International Workshop on Interim-PET-Scan in Lymphoma. Leuk Lymphoma 2009;50:1257–60.
32. Barrington SF, Mikhaeel NG, Kostakoglu L, et al. Role of Imaging in the Staging and Response Assessment of Lymphoma: Consensus of the International Conference on Malignant Lymphomas Imaging Working Group. J Clin Oncol 2014;32:3048–58.
33. Press OW, Li H, Schoder H, et al. US Intergroup trial of response-adapted therapy for stage III to IV Hodgkin lymphoma using early interim fluorodeoxyglucose-positron emission tomography imaging: Southwest Oncology Group S0816. J Clin Oncol 2016;34:2020–7.
34. Zinzani PL, Broccoli A, Gioia DM, et al. Interim positron emission tomography response-adapted therapy in advanced-stage Hodgkin lymphoma: final results of the pPhase II part of the HD0801 study. J Clin Oncol 2016;34:1376–85.
35. Borchmann P, Haverkamp H, Lohri A, et al. Progression-free survival of early interim PET-positive patients with advanced stage Hodgkin’s lymphoma treated with BEACOPPescalated alone or in combination with rituximab (HD18): an open-label, international, randomised phase 3 study by the German Hodgkin Study Group. Lancet Oncology 2017;18:454–63.
36. Aleman BM, Raemaekers JM, Tirelli U, et al. Involved-field radiotherapy for advanced Hodgkin’s lymphoma. N Engl J Med 2003;348:2396–406.
37. Borchmann P, Haverkamp H, Diehl V, et al. Eight cycles of escalated-dose BEACOPP compared with four cycles of escalated-dose BEACOPP followed by four cycles of baseline-dose BEACOPP with or without radiotherapy in patients with advanced-stage hodgkin’s lymphoma: final analysis of the HD12 trial of the German Hodgkin Study Group. J Clin Oncol 2011;29:4234–42.
38. Engert A, Haverkamp H, Kobe C, et al. Reduced-intensity chemotherapy and PET-guided radiotherapy in patients with advanced stage Hodgkin’s lymphoma (HD15 trial): a randomised, open-label, phase 3 non-inferiority trial. Lancet 2012;379:1791–9.
39. Nademanee A, Molina A, Fung H, et al. High-dose chemo/radiotherapy and autologous bone marrow or stem cell transplantation for poor-risk advanced-stage Hodgkin’s disease during first partial or complete remission. Biol Blood Marrow Transplant 1999;5:292–8.
40. Federico M, Bellei M, Brice P, et al. High-dose therapy and autologous stem-cell transplantation versus conventional therapy for patients with advanced Hodgkin’s lymphoma responding to front-line therapy. J Clin Oncol 2003;21:2320–5.
41. Arakelyan N, Berthou C, Desablens B, et al. Early versus late intensification for patients with high-risk Hodgkin lymphoma-3 cycles of intensive chemotherapy plus low-dose lymph node radiation therapy versus 4 cycles of combined doxorubicin, bleomycin, vinblastine, and dacarbazine plus myeloablative chemotherapy with autologous stem cell transplantation: five-year results of a randomized trial on behalf of the GOELAMS Group. Cancer 2008;113:3323–30.
42. Moskowitz CH, Nimer SD, Zelenetz AD, et al. A 2-step comprehensive high-dose chemoradiotherapy second-line program for relapsed and refractory Hodgkin disease: analysis by intent to treat and development of a prognostic model. Blood 2001;97:616–23.
43. Josting A, Rudolph C, Reiser M, et al. Time-intensified dexamethasone/cisplatin/cytarabine: an effective salvage therapy with low toxicity in patients with relapsed and refractory Hodgkin’s disease. Ann Oncol 2002;13:1628–35.
44. Aparicio J, Segura A, Garcera S, et al. ESHAP is an active regimen for relapsing Hodgkin’s disease. Ann Oncol 1999;10:593–5.
45. Baetz T, Belch A, Couban S, et al. Gemcitabine, dexamethasone and cisplatin is an active and non-toxic chemotherapy regimen in relapsed or refractory Hodgkin’s disease: a phase II study by the National Cancer Institute of Canada Clinical Trials Group. Ann Oncol 2003;14:1762–7.
46. Gopal AK, Press OW, Shustov AR, et al. Efficacy and safety of gemcitabine, carboplatin, dexamethasone, and rituximab in patients with relapsed/refractory lymphoma: a prospective multi-center phase II study by the Puget Sound Oncology Consortium. Leuk Lymphoma 2010;51:1523–9.
47. Santoro A, Magagnoli M, Spina M, et al. Ifosfamide, gemcitabine, and vinorelbine: a new induction regimen for refractory and relapsed Hodgkin’s lymphoma. Haematologica 2007;92:35–41.
48. Bartlett NL, Niedzwiecki D, Johnson JL, et al. Gemcitabine, vinorelbine, and pegylated liposomal doxorubicin (GVD), a salvage regimen in relapsed Hodgkin’s lymphoma: CALGB 59804. Ann Oncol 2007;18:1071–9.
49. Santoro A, Bonadonna G. Prolonged disease-free survival in MOPP-resistant Hodgkin’s disease after treatment with adriamycin, bleomycin, vinblastine and dacarbazine (ABVD). Cancer Chemother Pharmacol 1979;2:101–5.
50. Krikorian JG, Portlock CS, Rosenberg SA. Treatment of advanced Hodgkin’s disease with adriamycin, bleomycin, vinblastine, and imidazole carboxamide (ABVD) after failure of MOPP therapy. Cancer 1978;41:2107–11.
51. Piga A, Ambrosetti A, Todeschini G, et al. Doxorubicin, bleomycin, vinblastine, and dacarbazine (ABVD) salvage of mechlorethamine, vincristine, prednisone, and procarbazine (MOPP)-resistant advanced Hodgkin’s disease. Cancer Treat Rep 1984;68:947–51.
52. Moskowitz AJ, Hamlin PA Jr, Perales MA, et al. Phase II study of bendamustine in relapsed and refractory Hodgkin lymphoma. J Clin Oncol 2013;31:456–60.
53. Anastasia A, Carlo-Stella C, Corradini P, et al. Bendamustine for relapsed/refractory classical Hodgkin lymphoma after high dose chemotherapy and or allogeneic transplant: a study of Fondazione Italiana Linfomi (FIL). Blood 2012;120:3652.
54. Martin A, Fernandez-Jimenez MC, Caballero MD, et al. Long-term follow-up in patients treated with Mini-BEAM as salvage therapy for relapsed or refractory Hodgkin’s disease. Br J Haematol 2001;113:161–71.
55. Schmitz N, Pfistner B, Sextro M, et al. Aggressive conventional chemotherapy compared with high-dose chemotherapy with autologous haemopoietic stem-cell transplantation for relapsed chemosensitive Hodgkin’s disease: a randomised trial. Lancet 2002;359:2065–71.
56. Fisher RI, DeVita VT, Hubbard SP, et al. Prolonged disease-free survival in Hodgkin’s disease with MOPP reinduction after first relapse. Ann Intern Med 1979;90:761–3.
57. ChlVPP therapy for Hodgkin’s disease: experience of 960 patients. The International ChlVPP Treatment Group. Ann Oncol 1995;6:167–72.
58. Selby P, Patel P, Milan S, et al. ChlVPP combination chemotherapy for Hodgkin’s disease: long-term results. Br J Cancer 1990;62:279–85.
59. Santoro A, Bredenfeld H, Devizzi L, et al. Gemcitabine in the treatment of refractory Hodgkin’s disease: results of a multicenter phase II study. J Clin Oncol 2000;18:2615–9.
60. Little R, Wittes RE, Longo DL, Wilson WH. Vinblastine for recurrent Hodgkin’s disease following autologous bone marrow transplant. J Clin Oncol 1998;16:584–8.
61. Chen R, Palmer JM, Martin P, et al. Results of a multicenter phase II trial of brentuximab vedotin as second-line therapy before autologous transplantation in relapsed/refractory Hodgkin lymphoma. Biol Blood Marrow Transplant 2015;21:2136–40.
62. Moskowitz CH, Yahalom J, Zelenetz AD, et al. High-dose chemo-radiotherapy for relapsed or refractory Hodgkin lymphoma and the significance of pre-transplant functional imaging. Br J Haematol 2010;148:890–7.
63. Sureda A, Constans M, Iriondo A, et al. Prognostic factors affecting long-term outcome after stem cell transplantation in Hodgkin’s lymphoma autografted after a first relapse. Ann Oncol 2005;16:625–33.
64. Crocchiolo R, Canevari C, Assanelli A, et al. Pre-transplant 18FDG-PET predicts outcome in lymphoma patients treated with high-dose sequential chemotherapy followed by autologous stem cell transplantation. Leuk Lymphoma 2008;49:727–33.
65. Mocikova H, Pytlik R, Markova J, et al. Pre-transplant positron emission tomography in patients with relapsed Hodgkin lymphoma. Leuk Lymphoma 2011;52:1668–74.
66. Younes A, Gopal AK, Smith SE, et al. Results of a pivotal phase II study of brentuximab vedotin for patients with relapsed or refractory Hodgkin’s lymphoma. J Clin Oncol 2012;30:2183–9.
67. Gopal AK, Chen R, Smith SE, et al. Durable remissions in a pivotal phase 2 study of brentuximab vedotin in relapsed or refractory Hodgkin lymphoma. Blood 2015;125:1236–43.
68. Ansell SM, Lesokhin AM, Borrello I, et al. PD-1 blockade with nivolumab in relapsed or refractory Hodgkin’s lymphoma.N Engl J Med 2015;372:311–9.
69. Chen R, Zinzani PL, Fanale MA, et al. Phase II study of the efficacy and safety of pembrolizumab for relapsed/refractory classic Hodgkin lymphoma. J Clin Oncol 2017;35:2125–32.
70. Haverkos BM, Abbott D, Hamadani M, et al. PD-1 blockade for relapsed lymphoma post-allogeneic hematopoietic cell transplant: high response rate but frequent GVHD. Blood 2017;130:221–8.
71. Herbaux C, Gauthier J, Brice P, et al. Efficacy and tolerability of nivolumab after allogeneic transplantation for relapsed Hodgkin lymphoma. Blood 2017;129:2471–8.
72. Merryman RW, Kim HT, Zinzani PL et al. Safety and efficacy of allogeneic hematopoietic stem cell transplant after PD-1 blockage in relapsed/refractory lymphoma. Blood 2017;129:1380–8.
73. Linch DC, Winfield D, Goldstone AH, et al. Dose intensification with autologous bone-marrow transplantation in relapsed and resistant Hodgkin’s disease: results of a BNLI randomised trial. Lancet 1993;341:1051–4.
74. Yuen AR, Rosenberg SA, Hoppe RT, et al. Comparison between conventional salvage therapy and high-dose therapy with autografting for recurrent or refractory Hodgkin’s disease. Blood 1997;89:814–22.
75. Gutierrez-Delgado F, Holmberg L, Hooper H, et al. Autologous stem cell transplantation for Hodgkin’s disease: busulfan, melphalan and thiotepa compared to a radiation-based regimen. Bone Marrow Transplant 2003;32:279–85.
76. Hake CR, Graubert TA, Fenske TS. Does autologous transplantation directly increase the risk of secondary leukemia in lymphoma patients? Bone Marrow Transplant 2007;39:59–70.
77. Hahn T, McCarthy PL, Carreras J, et al. Comparison of prognostic models for autologous hematopoietic stem cell transplantation (AHCT) for relapsed Hodgkin lymphoma. Blood 2009;114:1215.
78. Chen Y-B, Lane AA, Logan BR, et al. Impact of conditioning regimen on outcomes for patients with lymphoma undergoing high-dose therapy with autologous hematopoietic cell transplantation. Biology Blood Marrow Transplant 2015;21:1046–53.
79. Wendland MM, Asch JD, Pulsipher MA, et al. The impact of involved field radiation therapy for patients receiving high-dose chemotherapy followed by hematopoietic progenitor cell transplant for the treatment of relapsed or refractory Hodgkin disease. Am J Clin Oncol 2006;29:189–95.
80. Biswas T, Culakova E, Friedberg JW, et al. Involved field radiation therapy following high dose chemotherapy and autologous stem cell transplant benefits local control and survival in refractory or recurrent Hodgkin lymphoma. Radiother Oncol 2012;103:367–72.
81. Levis M, Piva C, Filippi AR, et al. Potential benefit of involved-field radiotherapy for patients with Relapsed-refractory Hodgkin’s lymphoma with incomplete response before autologous stem cell transplantation. Clin Lymphoma Myeloma Leuk 2017;17:14–22.
82. Josting A, Franklin J, May M, et al. New prognostic score based on treatment outcome of patients with relapsed Hodgkin’s lymphoma registered in the database of the German Hodgkin’s lymphoma study group. J Clin Oncol 2002;20:221–30.
83. Moskowitz CH, Nademanee A, Masszi T, et al. Brentuximab vedotin as consolidation therapy after autologous stem-cell transplantation in patients with Hodgkin’s lymphoma at risk of relapse or progression (AETHERA): a randomised, double-blind, placebo-controlled, phase 3 trial. Lancet 2015;385:1853–62.
84. Bonfante V, Santoro A, Viviani S, et al. Outcome of patients with Hodgkin’s disease failing after primary MOPP-ABVD. J Clin Oncol 1997;15:528–34.
85. Josting A, Rueffer U, Franklin J, et al. Prognostic factors and treatment outcome in primary progressive Hodgkin lymphoma: a report from the German Hodgkin Lymphoma Study Group. Blood 2000;96:1280–6.
86. Josting A, Nogova L, Franklin J, et al. Salvage radiotherapy in patients with relapsed and refractory Hodgkin’s lymphoma: a retrospective analysis from the German Hodgkin Lymphoma Study Group. J Clin Oncol 2005;23:1522–9.
87. Campbell B WA, Milner A, Di Iulio J, et al. Long-term follow-up of salvage radiotherapy in Hodgkin’s lymphoma after chemotherapy failure. Int J Radiat Oncol Biol Phys 2005;63:1538–45.
88. Wirth A, Corry J, Laidlaw C, et al. Salvage radiotherapy for Hodgkin’s disease following chemotherapy failure. Int J Radiat Oncol Biol Phys 1997;39:599–607.
89. Schulz H, Rehwald U, Morschhauser F, et al. Rituximab in relapsed lymphocyte-predominant Hodgkin lymphoma: long-term results of a phase 2 trial by the German Hodgkin Lymphoma Study Group (GHSG). Blood 2008;111:109–11.
90. Ekstrand BC, Lucas JB, Horwitz SM, et al. Rituximab in lymphocyte-predominant Hodgkin disease: results of a phase 2 trial. Blood 2003;101:4285–9.
91. Younes A, Romaguera J, Hagemeister F, et al. A pilot study of rituximab in patients with recurrent, classic Hodgkin disease. Cancer 2003;98:310–4.
92. Rehwald U, Schulz H, Reiser M, et al. Treatment of relapsed CD20+ Hodgkin lymphoma with the monoclonal antibody rituximab is effective and well tolerated: results of a phase 2 trial of the German Hodgkin Lymphoma Study Group. Blood 2003;101:420–4.
93. Fehniger TA, Larson S, Trinkaus K, et al. A phase 2 multicenter study of lenalidomide in relapsed or refractory classical Hodgkin lymphoma. Blood 2011;118:5119–25.
94. Boll B, Borchmann P, Topp MS, et al. Lenalidomide in patients with refractory or multiple relapsed Hodgkin lymphoma. Br J Haematol 2010;148:480–2.
95. LaCasce AS, Bociek G, Sawas A, et al. Brentuximab vedotin plus bendamustine: a highly active salvage treatment regimen for patients with relapsed or refractory Hodgkin lymphoma. Blood 2015;126:3982.
96. Diefenbach CS, Hong F, David KA, et al. A phase I study with an expansion cohort of the combination of ipilimumab and nivolumab and brentuximab vedotin in patients with relapsed/refractory Hodgkin lymphoma: A trial of the ECOG-ACRIN Cancer Research Group (E4412 Arms D and E). Blood 2016;128:1106.
97. Herrera AF, Bartlett NL, Ramchandren R, et al. Preliminary results from a phase 1/2 study of brentuximab vedotin in combination with nivolumab in patients with relapsed or refractory Hodgkin lymphoma. Blood 2016;128:1105.
98. Johnston PB, Inwards DJ, Colgan JP, et al. A phase II trial of the oral mTOR inhibitor everolimus in relapsed Hodgkin lymphoma. Am J Hematol 2010;85:320–4.
99. Kewalramani T, Nimer SD, Zelenetz AD, et al. Progressive disease following autologous transplantation in patients with chemosensitive relapsed or primary refractory Hodgkin’s disease or aggressive non-Hodgkin’s lymphoma. Bone Marrow Transplant 2003;32:673–9.
100. Lin TS, Avalos BR, Penza SL, et al. Second autologous stem cell transplant for multiply relapsed Hodgkin’s disease. Bone Marrow Transplant 2002;29:763–7.
101. Smith SM, van Besien K, Carreras J, et al. Second autologous stem cell transplantation for relapsed lymphoma after a prior autologous transplant. Biol Blood Marrow Transplant 2008;14:904–12.
102. Gajewski JL, Phillips GL, Sobocinski KA, et al. Bone marrow transplants from HLA-identical siblings in advanced Hodgkin’s disease. J Clin Oncol 1996;14:572–8.
103. Peniket AJ, Ruiz de Elvira MC, Taghipour G, et al. An EBMT registry matched study of allogeneic stem cell transplants for lymphoma: allogeneic transplantation is associated with a lower relapse rate but a higher procedure-related mortality rate than autologous transplantation. Bone Marrow Transplant 2003;31:667–78.
104. Anderlini P, Saliba R, Acholonu S, et al. Fludarabine-melphalan as a preparative regimen for reduced-intensity conditioning allogeneic stem cell transplantation in relapsed and refractory Hodgkin’s lymphoma: the updated M.D. Anderson Cancer Center experience. Haematologica 2008;93:257–64.
105. Burroughs LM, O’Donnell PV, Sandmaier BM, et al. Comparison of outcomes of HLA-matched related, unrelated, or HLA-haploidentical related hematopoietic cell transplantation following nonmyeloablative conditioning for relapsed or refractory Hodgkin lymphoma. Biol Blood Marrow Transplant 2008;14:1279–87.
106. Peggs KS, Hunter A, Chopra R, et al. Clinical evidence of a graft-versus-Hodgkin’s-lymphoma effect after reduced-intensity allogeneic transplantation. Lancet 2005;365:1934–41.
107. Sureda A, Robinson S, Canals C, et al. Reduced-intensity conditioning compared with conventional allogeneic stem-cell transplantation in relapsed or refractory Hodgkin’s lymphoma: an analysis from the Lymphoma Working Party of the European Group for Blood and Marrow Transplantation. J Clin Oncol 2008;26:455–62.
108. Devetten MP, Hari PN, Carreras J, et al. Unrelated donor reduced-intensity allogeneic hematopoietic stem cell transplantation for relapsed and refractory Hodgkin lymphoma. Biol Blood Marrow Transplant 2009;15:109–17.
109. Sureda A, Canals C, Arranz R, et al. Allogeneic stem cell transplantation after reduced intensity conditioning in patients with relapsed or refractory Hodgkin’s lymphoma. Results of the HDR-ALLO study - a prospective clinical trial by the Grupo Espanol de Linfomas/Trasplante de Medula Osea (GEL/TAMO) and the Lymphoma Working Party of the European Group for Blood and Marrow Transplantation. Haematologica 2012;97:310–7.
110. Sarina B, Castagna L, Farina L, et al. Allogeneic transplantation improves the overall and progression-free survival of Hodgkin lymphoma patients relapsing after autologous transplantation: a retrospective study based on the time of HLA typing and donor availability. Blood 2010;115:3671–7.
111. Castagna L, Sarina B, Todisco E, et al. Allogeneic stem cell transplantation compared with chemotherapy for poor-risk Hodgkin lymphoma. Biol Blood Marrow Transplant 2009;15:432–8.
112. Thomson KJ, Peggs KS, Smith P, et al. Superiority of reduced-intensity allogeneic transplantation over conventional treatment for relapse of Hodgkin’s lymphoma following autologous stem cell transplantation. Bone Marrow Transplant 2008;41:765–70.
113. Chen R, Palmer JM, Thomas SH, et al. Brentuximab vedotin enables successful reduced-intensity allogeneic hematopoietic cell transplantation in patients with relapsed or refractory Hodgkin lymphoma. Blood 2012;119:6379–81.
2017 Update on pelvic floor dysfunction
The International Continence Society (ICS) defines overactive bladder (OAB) as a syndrome of "urinary urgency, usually accompanied by frequency and nocturia, with or without urgency urinary incontinence (UUI), in the absence of urinary tract infection [UTI] or obvious pathology."1 The Agency for Healthcare Research and Quality (AHRQ) reported OAB prevalence to be 15% in US women, with 11% reporting UUI.2 OAB represents a significant health care burden that impacts nearly every aspect of life, including physical, emotional, and psychological domains.3,4 The economic impact is notable; the projected cost is estimated to reach $82.6 billion annually by 2020.5
The American Urological Association (AUA) and the Society for Urodynamics, Female Pelvic Medicine and Urogenital Reconstruction (SUFU) have endorsed an algorithm for use in the evaluation of idiopathic OAB (FIGURE).6 If the patient's symptoms are certain, minimal evaluation is needed and it is reasonable to proceed with first-line therapy, which includes fluid management (decreasing caffeine intake and limiting evening fluid intake), bladder retraining drills such as timed voiding, and improving pelvic floor muscles with the use of biofeedback and functional electrical stimulation.6,7 Pelvic floor muscle training can be facilitated with a referral to a physical therapist trained in pelvic floor muscle education.
If treatment goals are not met with first-line strategies, second-line therapy may be initiated with anticholinergic or β3-adrenergic receptor agonist medications. If symptoms persist after 4 to 8 weeks of pharmacologic therapy, clinicians are encouraged to reassess or refer the patient to a specialist. Further evaluation may include a bladder diary in which the patient documents voided volumes, voiding frequency, and number of incontinent episodes; symptom-specific questionnaires; and/or urodynamic testing.
Related article:
The latest treatments for urinary and fecal incontinence: Which hold water?
Based on that evaluation, the patient may be a candidate for third-line therapy with either intradetrusor onabotulinumtoxinA, posterior tibial nerve stimulation (PTNS), or sacral neuromodulation.
There is a paucity of information comparing third-line therapies. In this Update, we focus on 4 randomized clinical trials that compare third-line treatment options for idiopathic OAB.
Read about how anticholinergic medication and onabotulinumtoxinA compare for treating UUI.
Anticholinergic therapy and onabotulinumtoxinA produce equivalent reductions in the frequency of daily UUI episodes
Visco AG, Brubaker L, Richter HE, et al; for the Pelvic Floor Disorders Network. Anticholinergic therapy vs onabotulinumtoxinA for urgency urinary incontinence. N Engl J Med. 2012;367(19):1803-1813.
In a double-blind, double-placebo-controlled randomized trial, Visco and colleagues compared anticholinergic medication with onabotulinumtoxinA 100 U for the treatment of women with UUI.
Details of the study
Two hundred forty-one women with moderate to severe UUI received either 6 months of oral anticholinergic therapy (solifenacin 5 mg daily with the option of dose escalation to 10 mg daily or change to trospium XR 60 mg daily based on the Patient Global Symptom Control score) plus a single intradetrusor injection of saline, or a single intradetrusor injection of onabotulinumtoxinA 100 U plus a 6-month oral placebo regimen.
Inclusion criteria were 5 or more UUI episodes on a 3-day diary, insufficient resolution of symptoms after 2 medications, or being drug naive. Exclusions included a postvoid residual (PVR) urine volume greater than 150 mL or previous therapy with onabotulinumtoxinA.
Participants were scheduled for follow up every 2 to 6 months post randomization, at which time all study medications were discontinued. The primary outcome was reduction from baseline in the mean number of UUI episodes per day over the 6-month period, as recorded in the monthly 3-day bladder diaries. Secondary outcomes included the proportion of participants with complete resolution of UUI, the proportion of participants with 75% or more reduction in UUI episodes, Overactive Bladder Questionnaire Short Form (OABq-SF) scores, other symptom-specific questionnaire scores, and adverse events.
Related article:
Which treatments for pelvic floor disorders are backed by evidence?
Both treatments significantly reduced UUI episodes
At baseline, participants reported a mean (SD) of 5.0 (2.7) UUI episodes per day, and 41% of participants were drug naive. Both treatment groups experienced significant reductions compared with baseline in mean UUI episodes, and the reductions were similar between the 2 groups (reduction of 3.4 episodes per day in the anticholinergic group, reduction of 3.3 episodes in the onabotulinumtoxinA group; P = .81). Complete resolution of UUI was more common in the onabotulinumtoxinA group (27%) as compared with the anticholinergic group (13%) (P = .003). There were no differences in improvement in OABq-SF scores (37.05 in the anticholinergic group vs 37.13 in the onabotulinumtoxinA group; P = .98) or other quality-of-life measures.
Adverse events. The anticholinergic group experienced a higher rate of dry mouth compared with the onabotulinumtoxinA group (46% vs 31%; P = .02) but had lower rates of intermittent catheterization use at 2 months (0% vs 5%, P = .01) and UTIs (13% vs 33%, P<.001).
Strengths and limitations. This was a well-designed, multicenter, randomized double-blind, double placebo-controlled trial. The study design allowed for dose escalation and change to another medication for inadequate symptom control and included drug-naive participants, which increases the generalizability of the results. However, current guidelines recommend reserving onabotulinumtoxinA therapy for third-line therapy, thus deterring this treatment's use in the drug-naive population. Additionally, the lack of a pure placebo arm makes it difficult to interpret the extent to which a placebo effect contributed to observed improvements in clinical symptoms.
Through 6 months, both a single intradetrusor injection of onabotulinumtoxinA 100 U and anticholinergic therapy reduce UUI episodes and improve quality-of-life measures in women who have failed medications or are drug naive. Use of onabotulinumtoxinA, however, more likely will lead to complete resolution of UUI, although with an increased risk of transient urinary retention and UTI. Even given the study findings supporting the use of onabotulinumtoxinA over anticholinergic therapy for complete resolution of UUI, it is most appropriate to align with current practice, which includes a trial of pharmacotherapy before proceeding with third-line onabotulinumtoxinA.
Read: onabotulinumtoxinA vs PTNS for OAB.
OnabotulinumtoxinA has greater 9-month durability for OAB symptoms compared with12 weeks of PTNS
Sherif H, Khalil M, Omar R. Management of refractory idiopathic overactive bladder: intradetrusor injection of botulinum toxin type A versus posterior tibial nerve stimulation. Can J Urol. 2017;24(3):8838-8846.
In this randomized clinical trial, Sherif and colleagues compared the safety and efficacy of a single intradetrusor injection of onabotulinumtoxinA 100 U with that of PTNS for OAB.
Details of the study
Sixty adult men and women with OAB who did not respond to medical therapy were randomly assigned to treatment with either onabotulinumtoxinA 100 U or PTNS. Criteria for exclusion were current UTI, PVR urine volume of more than 150 mL, previous radiation therapy or chemotherapy, previous incontinence surgery or bladder malignancy, or presence of mixed urinary incontinence.
At baseline, participants completed a 3-day bladder diary, an OAB symptom score (OABSS) questionnaire, and urodynamic testing. The OABSS questionnaire included 7 questions (scoring range, 0-28), with higher scores indicating worse symptoms, and included subscales for urgency and quality-of-life measures. Total OABSS, urgency score, quality-of-life score, bladder diary records, and urodynamic testing parameters were assessed at 6, 12, 24, and 36 weeks, along with adverse events.
OnabotulinumtoxinA injections were performed under spinal anesthesia. If PVR urine volume was greater than 200 mL at any follow-up visit, participants were instructed to begin clean intermittent self-catheterization. PTNS was administered as weekly 30-minute sessions for 12 consecutive weeks.
Participants' baseline demographics and symptoms were similar. Average age was 45 years. Averages (SD) for duration of anticholinergic use was 13 (0.8) weeks, UUI episode score was 4.5 (1) on 3-day bladder diary, and OABSS was 22 (2.7). Nine-month data were available for 29 participants in the onabotulinumtoxinA group and for 8 in the PTNS group.
Related article:
Update on pelvic floor dysfunction: Focus on urinary incontinence
OnabotulinumtoxinA treatment benefits sustained for 9 months
Through 6 months, compared with baseline assessments, both treatment groups had significant improvements in clinical symptoms and OABSS total score, as well as urgency and quality-of-life subscales. At 3 months, urodynamic study parameters were similarly improved from baseline in both groups.
At 9 months, however, only the onabotulinumtoxinA group, compared with the PTNS group, maintained the significant improvement from baseline in 3-day bladder diary voiding episodes (average [SD], 10.7 [1.01] vs 11.6 [1.09]; P = .009), 3-day bladder diary nocturia episodes (average [SD], 3.8 [1.09] vs 4.4 [0.8]; P = .02), and average [SD] UUI episodes over 3 days (3.5 [1.2] vs 4.2 [1.04]; P = .02). Similarly, onabotulinumtoxinA-treated participants, compared with those treated with PTNS, maintained improvements at 9 months in average (SD): OABSS total score (19.2 [2.4] vs 20.4 [1.7]; P = .03), urgency scores (10.9 [1.3] vs 11.8 [1.4]; P = .009), urine volume at first desire (177.8 [9.2] vs 171.8 [7.7]), maximum cystometric capacity (304 [17.6] vs 290 [13.1]), and Qmax (mL/sec) (20.7 [1.6] vs 22.2 [1.2]).
Adverse events. Average PVR urine volumes were higher in the onabotulinumtoxinA group compared with the PTNS group (36.8 [2.7] vs 32.4 [3.03]; P = .0001) at all time points, and self-catheterization was required in 6.6% of onabotulinumtoxinA-treated participants. Urinary tract infection occurred in 6.6% of participants in the onabotulinumtoxinA group and in none of the PTNS group. In the PTNS group, few experienced pain and minor bleeding at the needle site.
Strengths and limitations. This randomized, open-label trial comparing treatment with onabotulinumtoxinA 100 U and PTNS included both men and women with idiopathic OAB symptoms. The participants were assessed at regular intervals with various measures, and follow-up adherence was good. The sample size was small, so the study may not have been powered to see differences prior to 9 months.
Although at 9 months only the onabotulinumtoxinA group maintained significant improvement over baseline levels, the improvement was diminished, and therefore the clinical meaningfulness is uncertain. Further, participants in the PTNS group did not undergo monthly maintenance therapy after 3 months, which is recommended for those with a 12-week therapeutic response; this may have affected 9-month outcomes in this group. Since the one-time onabotulinumtoxinA 100 U injection was performed under spinal anesthesia, cost comparisons should be considered, since future onabotulinumtoxinA injections would be necessary.
A one-time onabotulinumtoxinA 100 U injection and 12 weeks of PTNS therapy are reasonable short-term options for symptomatic OAB relief after unsuccessful therapy with medications. OnabotulinumtoxinA injection may provide more durable OAB symptom control at 9 months but with a risk of UTI and need for self-catheterization.
Read about using different doses of onabotulinumtoxinA for OAB.
OnabotulinumtoxinA 200-U injection provides longer OAB symptom improvement than 100-U injection
Abdelwahab O, Sherif H, Soliman T, Elbarky I, Eshazly A. Efficacy of botulinum toxin type A 100 units versus 200 units for treatment of refractory idiopathic overactive bladder. Int Braz J Urol. 2015;41(6):1132-1140.
Abdelwahab and colleagues conducted a single-center, randomized clinical trial to investigate the safety and efficacy of a single injection of intradetrusor onabotulinumtoxinA in 2 different doses (100 U and 200 U) for treatment of OAB.
Details of the study
Eighty adults (63 women, 17 men) who did not benefit from anticholinergic medication during the previous 3 months were randomly assigned to receive either a 100-U (n = 40) or a 200-U (n = 40) injection of onabotulinumtoxinA. Exclusion criteria were PVR urine volume greater than 150 mL and previous radiation therapy or chemotherapy.
Initial assessments -- completed at baseline and at 1, 3, 6, and 9 months -- included the health-related quality-of-life (HR-QOL) questionnaire (maximum score, 100; higher score indicates better quality of life), an abbreviated OABSS questionnaire (4 questions; score range, 0-15; higher score indicates more severe symptoms), and urodynamic evaluation. Outcomes included OABSS, HR-QOL score, and urodynamic parameters at the various time points.
Related article:
Is there a link between impaired mobility and urinary incontinence in elderly, community-dwelling women?
Higher dose, greater symptom improvement and higher adverse event rate
At baseline, participants (average age, 31 years) had an average (SD) OABSS of 1.7 (1.6). OnabotulinumtoxinA treatment with both a 100-U and a 200-U dose resulted in significant improvements (compared with baseline levels) in frequency, nocturia, UUI episodes, OABSS, and urodynamic parameters throughout the 9 months. At 9 months, however, the group treated with the 200-U dose had greater improvements, compared with the group who received a 100-U dose, in urinary frequency symptom scores (mean [SD], 0.32 [0.47] vs 1.1 [0.51]; P<.05), nocturia symptom scores (mean [SD], 0.13 [0.34] vs 0.36 [0.49]; P<.05), UUI symptom scores (mean [SD], 0.68 [0.16] vs 1.26 [1.1]; P<.05), and mean (SD) total OABSS (2.6 [2.31] vs 5.3 [2.11]; P<.05). Similarly, at 9 months the 200-U dose resulted in greater improvements in volume at first desire (mean [SD], 291.8 [42.8] vs 246.8 [53.8] mL; P<.05), volume at strong desire (mean [SD], 392.1 [37.3] vs 313.1 [67.4] mL; P<.05), detrusor pressure (mean [SD], 10.4 [4.0] vs 19.2 [7.8] cm H2O; P<.05), and maximum cystometric capacity (mean [SD], 430.5 [34.2] vs 350 [69.1] mL; P<.05) compared with the 100-U dose.
Adverse events. No participant had a PVR urine volume greater than 100 mL at any follow-up visit. Postoperative hematuria occurred in 23% of the group treated with onabotulinumtoxinA 200 U versus in 15% of those treated with a 100-U dose. Similarly, UTIs occurred in 17.5% of the 200-U dose group and in 7.5% of the 100-U dose group. Dysuria was reported in 37.5% and 15% of the 200-U and 100-U dose groups, respectively.
Strengths and limitations. This randomized, open-label trial comparing a single injection of 100 U versus 200 U of onabotulinumtoxinA included mostly women. OAB symptoms and urodynamic parameters improved after treatment with both dose levels, but a longer duration of improvement was seen with the 200-U dose. The cohort had a low baseline OAB severity, based on the OABSS questionnaire, and a young average age of participants, which limits the generalizability of the study results to a population with refractory OAB. The 0% rate of clean intermittent self-catheterization postinjection might be based on the study's criteria for requiring clean intermittent catheterization. In addition, the initial postinjection visit occurred at 1 month, possibly missing participants who had symptoms of retention soon after injection.
Two dose levels (100 U and 200 U) of a single injection of onabotulinumtoxinA are associated with comparable OAB symptom and urodyanamic improvements. The benefits of a longer duration of effect with the 200-U dose must be weighed against the possible higher risks of transient hematuria, dysuria, and UTI.
Read: onabotulinumtoxinA vs sacral neuromodulation therapy for UUI.
Treatment with onabotulinumtoxinA may control UUI symptoms better than sacral neuromodulation therapy
Amundsen CL, Richter HE, Menefee SA, et al; Pelvic Floor Disorders Network. OnabotulinumtoxinA vs sacral neuromodulation on refractory urgency urinary incontinence in women: a randomized clinical trial. JAMA. 2016;316(13):1366-1374.
In this multicenter open-label randomized trial, Amundsen and colleagues compared the efficacy and safety of onabotulinumtoxinA 200 U with that of sacral neuromodulation.
Details of the study
Three hundred sixty-four women with UUI had data available for primary analysis at 6 months. Women were considered eligible for the study if they had 6 or more UUI episodes on a 3-day bladder diary, persistent symptoms despite anticholinergic therapy, a PVR urine volume of less than 150 mL, and had never previously received either study treatment.
There were no differences in baseline characteristics of the participants. The average (SD) age of the study population was 63 (11.6) years, with an average (SD) daily number of UUI episodes of 5.3 (2.8). The average (SD) body mass index was 32 (8) kg/m2.
Participants were randomly assigned to undergo either sacral neuromodulation (n = 174) or intradetrusor injection of onabotulinumtoxinA 200 U (n = 190). The primary outcome was change from baseline in mean number of daily UUI episodes averaged over 6 months as recorded on a monthly 3-day bladder diary. Secondary outcomes included complete resolution of urgency incontinence, 75% or more reduction in UUI episodes, the Overactive Bladder Questionnaire Short Form (SF) score (range, 0-100; higher score indicates higher symptom severity), the Overactive Bladder Satisfaction of Treatment questionnaire (range, 0-100; higher score indicates better satisfaction), other quality-of-life measures, and adverse events.
Related article:
2015 Update on pelvic floor dysfunction: Bladder pain syndrome
Greater symptom bother improvement, treatment satisfaction with onabotulinumtoxinA 200 U
Participants treated with onabotulinumtoxinA had a greater mean reduction of 3.9 UUI episodes per day than the sacral neuromodulation group's reduction of 3.3 UUI episodes per day (mean difference, 0.63; 95% confidence interval [CI], 0.13-1.14; P = .01). In addition, complete UUI resolution was higher in the onabotulinumtoxinA group as compared with the sacral neuromodulation group (20% vs 4%; P<.001). The onabotulinumtoxinA group also had higher rates of 75% or more reduction of UUI episodes compared with the sacral neuromodulation group (46% vs 26%; P<.001). Over 6 months, both groups had improvements in all quality-of-life measures, but the onabotulinumtoxinA group had greater improvement in symptom bother compared with the sacral neuromodulation group (-46.7 vs -38.6; mean difference, 8.1; 95% CI, 3.0-13.3; P = .002). Furthermore, the onabotulinumtoxinA group had greater treatment satisfaction compared with the sacral neuromodulation group (mean difference, 7.8; 95% CI, 1.6-14.1; P = .01).
Adverse events. Six women (3%) underwent sacral neuromodulation device revision or removal. Approximately 8% of onabotulinumtoxinA-treated participants required intermittent self-catheterization at 1 month, 4% at 3 months, and 2% at 6 months. The risk of UTI was higher in the onabotulinumtoxinA group compared with the sacral neuromodulation group (35% vs 11%; risk difference, 23%; 95% CI, -33% to -13%; P<.001).
Strengths and limitations. This is a well-designed randomized clinical trial comparing clinical outcomes and adverse events after treatment with onabotulinumtoxinA 200-U versus sacral neuromodulation. The interventions were standardized across investigators at multiple sites, and the study design required close follow-up to assess efficacy and adverse events. The study used a 200-U dose based on reported durability of effect at that time and findings of equivalency between onabotulinumtoxinA 100 U and anticholinergic therapy. The US Food and Drug Administration's recommendation to use a 100-U dose in all patients with idiopathic OAB might dissuade clinicians from considering the higher dose of onabotulinumtoxinA. The study was limited by the lack of a placebo group.
Both onabotulinumtoxinA 200 U and sacral neuromodulation provide significant improvement in UUI episodes and quality of life over 6 months. However, while treatment with onabotulinumtoxinA has a likelihood of complete UUI resolution, greater improvements in symptom bother and treatment satisfaction, these benefits must be weighed against the risks of transient catheterization and UTI.
Share your thoughts! Send your Letter to the Editor to [email protected]. Please include your name and the city and state in which you practice.
- Haylen BT, de Ridder D, Freeman RM, et al; International Urogynecological Association; International Continence Society. An International Urogynecological Association (IUGA)/International Continence Society (ICS) joint report on the terminology for female pelvic floor dysfunction. Neurourol Urodyn. 2010;29(1):4-20.
- Hartmann KE, McPheeters ML, Biller DH, et al. Treatment of overactive bladder in women. Evid Rep Technol Assess (Full Rep). 2009;187:1-120.
- Reynolds,WS, Fowke J, Dmochowski, R. The burden of overactive bladder on US public health. Curr Bladder Dysfunct Rep. 2016;11(1):8-13.
- Willis-Gray MG, Dieter AA, Geller EJ. Evaluation and management of overactive bladder: strategies for optimizing care. Res Rep Urol. 2016;8:113-122.
- Ganz ML, Smalarz AM, Krupski TL, et al. Economic costs of overactive bladder in the United States. Urology. 2010;75(3):526-532.
- Gormley EA, Lightner DJ, Faraday M, Vasavada SP; American Urological Association; Society of Urodyndamics, Female Pelvic Medicine. Diagnosis and treatment of overactive bladder (non-neurogenic) in adults: AUA/SUFU guideline amendment. J Urol. 2015; 193(5):1572-1580.
- Gormley EA, Lightner DJ, Burgio KL, et al; American Urological Association; Society of Urodynamics, Female Pelvic Medicine & Urogenital Reconstruction. Diagnosis and treatment of overactive bladder (non-neurogenic) in adults: AUA/SUFU guideline. J Urol. 2012;188(6 suppl):2455-2463.
The International Continence Society (ICS) defines overactive bladder (OAB) as a syndrome of "urinary urgency, usually accompanied by frequency and nocturia, with or without urgency urinary incontinence (UUI), in the absence of urinary tract infection [UTI] or obvious pathology."1 The Agency for Healthcare Research and Quality (AHRQ) reported OAB prevalence to be 15% in US women, with 11% reporting UUI.2 OAB represents a significant health care burden that impacts nearly every aspect of life, including physical, emotional, and psychological domains.3,4 The economic impact is notable; the projected cost is estimated to reach $82.6 billion annually by 2020.5
The American Urological Association (AUA) and the Society for Urodynamics, Female Pelvic Medicine and Urogenital Reconstruction (SUFU) have endorsed an algorithm for use in the evaluation of idiopathic OAB (FIGURE).6 If the patient's symptoms are certain, minimal evaluation is needed and it is reasonable to proceed with first-line therapy, which includes fluid management (decreasing caffeine intake and limiting evening fluid intake), bladder retraining drills such as timed voiding, and improving pelvic floor muscles with the use of biofeedback and functional electrical stimulation.6,7 Pelvic floor muscle training can be facilitated with a referral to a physical therapist trained in pelvic floor muscle education.
If treatment goals are not met with first-line strategies, second-line therapy may be initiated with anticholinergic or β3-adrenergic receptor agonist medications. If symptoms persist after 4 to 8 weeks of pharmacologic therapy, clinicians are encouraged to reassess or refer the patient to a specialist. Further evaluation may include a bladder diary in which the patient documents voided volumes, voiding frequency, and number of incontinent episodes; symptom-specific questionnaires; and/or urodynamic testing.
Related article:
The latest treatments for urinary and fecal incontinence: Which hold water?
Based on that evaluation, the patient may be a candidate for third-line therapy with either intradetrusor onabotulinumtoxinA, posterior tibial nerve stimulation (PTNS), or sacral neuromodulation.
There is a paucity of information comparing third-line therapies. In this Update, we focus on 4 randomized clinical trials that compare third-line treatment options for idiopathic OAB.
Read about how anticholinergic medication and onabotulinumtoxinA compare for treating UUI.
Anticholinergic therapy and onabotulinumtoxinA produce equivalent reductions in the frequency of daily UUI episodes
Visco AG, Brubaker L, Richter HE, et al; for the Pelvic Floor Disorders Network. Anticholinergic therapy vs onabotulinumtoxinA for urgency urinary incontinence. N Engl J Med. 2012;367(19):1803-1813.
In a double-blind, double-placebo-controlled randomized trial, Visco and colleagues compared anticholinergic medication with onabotulinumtoxinA 100 U for the treatment of women with UUI.
Details of the study
Two hundred forty-one women with moderate to severe UUI received either 6 months of oral anticholinergic therapy (solifenacin 5 mg daily with the option of dose escalation to 10 mg daily or change to trospium XR 60 mg daily based on the Patient Global Symptom Control score) plus a single intradetrusor injection of saline, or a single intradetrusor injection of onabotulinumtoxinA 100 U plus a 6-month oral placebo regimen.
Inclusion criteria were 5 or more UUI episodes on a 3-day diary, insufficient resolution of symptoms after 2 medications, or being drug naive. Exclusions included a postvoid residual (PVR) urine volume greater than 150 mL or previous therapy with onabotulinumtoxinA.
Participants were scheduled for follow up every 2 to 6 months post randomization, at which time all study medications were discontinued. The primary outcome was reduction from baseline in the mean number of UUI episodes per day over the 6-month period, as recorded in the monthly 3-day bladder diaries. Secondary outcomes included the proportion of participants with complete resolution of UUI, the proportion of participants with 75% or more reduction in UUI episodes, Overactive Bladder Questionnaire Short Form (OABq-SF) scores, other symptom-specific questionnaire scores, and adverse events.
Related article:
Which treatments for pelvic floor disorders are backed by evidence?
Both treatments significantly reduced UUI episodes
At baseline, participants reported a mean (SD) of 5.0 (2.7) UUI episodes per day, and 41% of participants were drug naive. Both treatment groups experienced significant reductions compared with baseline in mean UUI episodes, and the reductions were similar between the 2 groups (reduction of 3.4 episodes per day in the anticholinergic group, reduction of 3.3 episodes in the onabotulinumtoxinA group; P = .81). Complete resolution of UUI was more common in the onabotulinumtoxinA group (27%) as compared with the anticholinergic group (13%) (P = .003). There were no differences in improvement in OABq-SF scores (37.05 in the anticholinergic group vs 37.13 in the onabotulinumtoxinA group; P = .98) or other quality-of-life measures.
Adverse events. The anticholinergic group experienced a higher rate of dry mouth compared with the onabotulinumtoxinA group (46% vs 31%; P = .02) but had lower rates of intermittent catheterization use at 2 months (0% vs 5%, P = .01) and UTIs (13% vs 33%, P<.001).
Strengths and limitations. This was a well-designed, multicenter, randomized double-blind, double placebo-controlled trial. The study design allowed for dose escalation and change to another medication for inadequate symptom control and included drug-naive participants, which increases the generalizability of the results. However, current guidelines recommend reserving onabotulinumtoxinA therapy for third-line therapy, thus deterring this treatment's use in the drug-naive population. Additionally, the lack of a pure placebo arm makes it difficult to interpret the extent to which a placebo effect contributed to observed improvements in clinical symptoms.
Through 6 months, both a single intradetrusor injection of onabotulinumtoxinA 100 U and anticholinergic therapy reduce UUI episodes and improve quality-of-life measures in women who have failed medications or are drug naive. Use of onabotulinumtoxinA, however, more likely will lead to complete resolution of UUI, although with an increased risk of transient urinary retention and UTI. Even given the study findings supporting the use of onabotulinumtoxinA over anticholinergic therapy for complete resolution of UUI, it is most appropriate to align with current practice, which includes a trial of pharmacotherapy before proceeding with third-line onabotulinumtoxinA.
Read: onabotulinumtoxinA vs PTNS for OAB.
OnabotulinumtoxinA has greater 9-month durability for OAB symptoms compared with12 weeks of PTNS
Sherif H, Khalil M, Omar R. Management of refractory idiopathic overactive bladder: intradetrusor injection of botulinum toxin type A versus posterior tibial nerve stimulation. Can J Urol. 2017;24(3):8838-8846.
In this randomized clinical trial, Sherif and colleagues compared the safety and efficacy of a single intradetrusor injection of onabotulinumtoxinA 100 U with that of PTNS for OAB.
Details of the study
Sixty adult men and women with OAB who did not respond to medical therapy were randomly assigned to treatment with either onabotulinumtoxinA 100 U or PTNS. Criteria for exclusion were current UTI, PVR urine volume of more than 150 mL, previous radiation therapy or chemotherapy, previous incontinence surgery or bladder malignancy, or presence of mixed urinary incontinence.
At baseline, participants completed a 3-day bladder diary, an OAB symptom score (OABSS) questionnaire, and urodynamic testing. The OABSS questionnaire included 7 questions (scoring range, 0-28), with higher scores indicating worse symptoms, and included subscales for urgency and quality-of-life measures. Total OABSS, urgency score, quality-of-life score, bladder diary records, and urodynamic testing parameters were assessed at 6, 12, 24, and 36 weeks, along with adverse events.
OnabotulinumtoxinA injections were performed under spinal anesthesia. If PVR urine volume was greater than 200 mL at any follow-up visit, participants were instructed to begin clean intermittent self-catheterization. PTNS was administered as weekly 30-minute sessions for 12 consecutive weeks.
Participants' baseline demographics and symptoms were similar. Average age was 45 years. Averages (SD) for duration of anticholinergic use was 13 (0.8) weeks, UUI episode score was 4.5 (1) on 3-day bladder diary, and OABSS was 22 (2.7). Nine-month data were available for 29 participants in the onabotulinumtoxinA group and for 8 in the PTNS group.
Related article:
Update on pelvic floor dysfunction: Focus on urinary incontinence
OnabotulinumtoxinA treatment benefits sustained for 9 months
Through 6 months, compared with baseline assessments, both treatment groups had significant improvements in clinical symptoms and OABSS total score, as well as urgency and quality-of-life subscales. At 3 months, urodynamic study parameters were similarly improved from baseline in both groups.
At 9 months, however, only the onabotulinumtoxinA group, compared with the PTNS group, maintained the significant improvement from baseline in 3-day bladder diary voiding episodes (average [SD], 10.7 [1.01] vs 11.6 [1.09]; P = .009), 3-day bladder diary nocturia episodes (average [SD], 3.8 [1.09] vs 4.4 [0.8]; P = .02), and average [SD] UUI episodes over 3 days (3.5 [1.2] vs 4.2 [1.04]; P = .02). Similarly, onabotulinumtoxinA-treated participants, compared with those treated with PTNS, maintained improvements at 9 months in average (SD): OABSS total score (19.2 [2.4] vs 20.4 [1.7]; P = .03), urgency scores (10.9 [1.3] vs 11.8 [1.4]; P = .009), urine volume at first desire (177.8 [9.2] vs 171.8 [7.7]), maximum cystometric capacity (304 [17.6] vs 290 [13.1]), and Qmax (mL/sec) (20.7 [1.6] vs 22.2 [1.2]).
Adverse events. Average PVR urine volumes were higher in the onabotulinumtoxinA group compared with the PTNS group (36.8 [2.7] vs 32.4 [3.03]; P = .0001) at all time points, and self-catheterization was required in 6.6% of onabotulinumtoxinA-treated participants. Urinary tract infection occurred in 6.6% of participants in the onabotulinumtoxinA group and in none of the PTNS group. In the PTNS group, few experienced pain and minor bleeding at the needle site.
Strengths and limitations. This randomized, open-label trial comparing treatment with onabotulinumtoxinA 100 U and PTNS included both men and women with idiopathic OAB symptoms. The participants were assessed at regular intervals with various measures, and follow-up adherence was good. The sample size was small, so the study may not have been powered to see differences prior to 9 months.
Although at 9 months only the onabotulinumtoxinA group maintained significant improvement over baseline levels, the improvement was diminished, and therefore the clinical meaningfulness is uncertain. Further, participants in the PTNS group did not undergo monthly maintenance therapy after 3 months, which is recommended for those with a 12-week therapeutic response; this may have affected 9-month outcomes in this group. Since the one-time onabotulinumtoxinA 100 U injection was performed under spinal anesthesia, cost comparisons should be considered, since future onabotulinumtoxinA injections would be necessary.
A one-time onabotulinumtoxinA 100 U injection and 12 weeks of PTNS therapy are reasonable short-term options for symptomatic OAB relief after unsuccessful therapy with medications. OnabotulinumtoxinA injection may provide more durable OAB symptom control at 9 months but with a risk of UTI and need for self-catheterization.
Read about using different doses of onabotulinumtoxinA for OAB.
OnabotulinumtoxinA 200-U injection provides longer OAB symptom improvement than 100-U injection
Abdelwahab O, Sherif H, Soliman T, Elbarky I, Eshazly A. Efficacy of botulinum toxin type A 100 units versus 200 units for treatment of refractory idiopathic overactive bladder. Int Braz J Urol. 2015;41(6):1132-1140.
Abdelwahab and colleagues conducted a single-center, randomized clinical trial to investigate the safety and efficacy of a single injection of intradetrusor onabotulinumtoxinA in 2 different doses (100 U and 200 U) for treatment of OAB.
Details of the study
Eighty adults (63 women, 17 men) who did not benefit from anticholinergic medication during the previous 3 months were randomly assigned to receive either a 100-U (n = 40) or a 200-U (n = 40) injection of onabotulinumtoxinA. Exclusion criteria were PVR urine volume greater than 150 mL and previous radiation therapy or chemotherapy.
Initial assessments -- completed at baseline and at 1, 3, 6, and 9 months -- included the health-related quality-of-life (HR-QOL) questionnaire (maximum score, 100; higher score indicates better quality of life), an abbreviated OABSS questionnaire (4 questions; score range, 0-15; higher score indicates more severe symptoms), and urodynamic evaluation. Outcomes included OABSS, HR-QOL score, and urodynamic parameters at the various time points.
Related article:
Is there a link between impaired mobility and urinary incontinence in elderly, community-dwelling women?
Higher dose, greater symptom improvement and higher adverse event rate
At baseline, participants (average age, 31 years) had an average (SD) OABSS of 1.7 (1.6). OnabotulinumtoxinA treatment with both a 100-U and a 200-U dose resulted in significant improvements (compared with baseline levels) in frequency, nocturia, UUI episodes, OABSS, and urodynamic parameters throughout the 9 months. At 9 months, however, the group treated with the 200-U dose had greater improvements, compared with the group who received a 100-U dose, in urinary frequency symptom scores (mean [SD], 0.32 [0.47] vs 1.1 [0.51]; P<.05), nocturia symptom scores (mean [SD], 0.13 [0.34] vs 0.36 [0.49]; P<.05), UUI symptom scores (mean [SD], 0.68 [0.16] vs 1.26 [1.1]; P<.05), and mean (SD) total OABSS (2.6 [2.31] vs 5.3 [2.11]; P<.05). Similarly, at 9 months the 200-U dose resulted in greater improvements in volume at first desire (mean [SD], 291.8 [42.8] vs 246.8 [53.8] mL; P<.05), volume at strong desire (mean [SD], 392.1 [37.3] vs 313.1 [67.4] mL; P<.05), detrusor pressure (mean [SD], 10.4 [4.0] vs 19.2 [7.8] cm H2O; P<.05), and maximum cystometric capacity (mean [SD], 430.5 [34.2] vs 350 [69.1] mL; P<.05) compared with the 100-U dose.
Adverse events. No participant had a PVR urine volume greater than 100 mL at any follow-up visit. Postoperative hematuria occurred in 23% of the group treated with onabotulinumtoxinA 200 U versus in 15% of those treated with a 100-U dose. Similarly, UTIs occurred in 17.5% of the 200-U dose group and in 7.5% of the 100-U dose group. Dysuria was reported in 37.5% and 15% of the 200-U and 100-U dose groups, respectively.
Strengths and limitations. This randomized, open-label trial comparing a single injection of 100 U versus 200 U of onabotulinumtoxinA included mostly women. OAB symptoms and urodynamic parameters improved after treatment with both dose levels, but a longer duration of improvement was seen with the 200-U dose. The cohort had a low baseline OAB severity, based on the OABSS questionnaire, and a young average age of participants, which limits the generalizability of the study results to a population with refractory OAB. The 0% rate of clean intermittent self-catheterization postinjection might be based on the study's criteria for requiring clean intermittent catheterization. In addition, the initial postinjection visit occurred at 1 month, possibly missing participants who had symptoms of retention soon after injection.
Two dose levels (100 U and 200 U) of a single injection of onabotulinumtoxinA are associated with comparable OAB symptom and urodyanamic improvements. The benefits of a longer duration of effect with the 200-U dose must be weighed against the possible higher risks of transient hematuria, dysuria, and UTI.
Read: onabotulinumtoxinA vs sacral neuromodulation therapy for UUI.
Treatment with onabotulinumtoxinA may control UUI symptoms better than sacral neuromodulation therapy
Amundsen CL, Richter HE, Menefee SA, et al; Pelvic Floor Disorders Network. OnabotulinumtoxinA vs sacral neuromodulation on refractory urgency urinary incontinence in women: a randomized clinical trial. JAMA. 2016;316(13):1366-1374.
In this multicenter open-label randomized trial, Amundsen and colleagues compared the efficacy and safety of onabotulinumtoxinA 200 U with that of sacral neuromodulation.
Details of the study
Three hundred sixty-four women with UUI had data available for primary analysis at 6 months. Women were considered eligible for the study if they had 6 or more UUI episodes on a 3-day bladder diary, persistent symptoms despite anticholinergic therapy, a PVR urine volume of less than 150 mL, and had never previously received either study treatment.
There were no differences in baseline characteristics of the participants. The average (SD) age of the study population was 63 (11.6) years, with an average (SD) daily number of UUI episodes of 5.3 (2.8). The average (SD) body mass index was 32 (8) kg/m2.
Participants were randomly assigned to undergo either sacral neuromodulation (n = 174) or intradetrusor injection of onabotulinumtoxinA 200 U (n = 190). The primary outcome was change from baseline in mean number of daily UUI episodes averaged over 6 months as recorded on a monthly 3-day bladder diary. Secondary outcomes included complete resolution of urgency incontinence, 75% or more reduction in UUI episodes, the Overactive Bladder Questionnaire Short Form (SF) score (range, 0-100; higher score indicates higher symptom severity), the Overactive Bladder Satisfaction of Treatment questionnaire (range, 0-100; higher score indicates better satisfaction), other quality-of-life measures, and adverse events.
Related article:
2015 Update on pelvic floor dysfunction: Bladder pain syndrome
Greater symptom bother improvement, treatment satisfaction with onabotulinumtoxinA 200 U
Participants treated with onabotulinumtoxinA had a greater mean reduction of 3.9 UUI episodes per day than the sacral neuromodulation group's reduction of 3.3 UUI episodes per day (mean difference, 0.63; 95% confidence interval [CI], 0.13-1.14; P = .01). In addition, complete UUI resolution was higher in the onabotulinumtoxinA group as compared with the sacral neuromodulation group (20% vs 4%; P<.001). The onabotulinumtoxinA group also had higher rates of 75% or more reduction of UUI episodes compared with the sacral neuromodulation group (46% vs 26%; P<.001). Over 6 months, both groups had improvements in all quality-of-life measures, but the onabotulinumtoxinA group had greater improvement in symptom bother compared with the sacral neuromodulation group (-46.7 vs -38.6; mean difference, 8.1; 95% CI, 3.0-13.3; P = .002). Furthermore, the onabotulinumtoxinA group had greater treatment satisfaction compared with the sacral neuromodulation group (mean difference, 7.8; 95% CI, 1.6-14.1; P = .01).
Adverse events. Six women (3%) underwent sacral neuromodulation device revision or removal. Approximately 8% of onabotulinumtoxinA-treated participants required intermittent self-catheterization at 1 month, 4% at 3 months, and 2% at 6 months. The risk of UTI was higher in the onabotulinumtoxinA group compared with the sacral neuromodulation group (35% vs 11%; risk difference, 23%; 95% CI, -33% to -13%; P<.001).
Strengths and limitations. This is a well-designed randomized clinical trial comparing clinical outcomes and adverse events after treatment with onabotulinumtoxinA 200-U versus sacral neuromodulation. The interventions were standardized across investigators at multiple sites, and the study design required close follow-up to assess efficacy and adverse events. The study used a 200-U dose based on reported durability of effect at that time and findings of equivalency between onabotulinumtoxinA 100 U and anticholinergic therapy. The US Food and Drug Administration's recommendation to use a 100-U dose in all patients with idiopathic OAB might dissuade clinicians from considering the higher dose of onabotulinumtoxinA. The study was limited by the lack of a placebo group.
Both onabotulinumtoxinA 200 U and sacral neuromodulation provide significant improvement in UUI episodes and quality of life over 6 months. However, while treatment with onabotulinumtoxinA has a likelihood of complete UUI resolution, greater improvements in symptom bother and treatment satisfaction, these benefits must be weighed against the risks of transient catheterization and UTI.
Share your thoughts! Send your Letter to the Editor to [email protected]. Please include your name and the city and state in which you practice.
The International Continence Society (ICS) defines overactive bladder (OAB) as a syndrome of "urinary urgency, usually accompanied by frequency and nocturia, with or without urgency urinary incontinence (UUI), in the absence of urinary tract infection [UTI] or obvious pathology."1 The Agency for Healthcare Research and Quality (AHRQ) reported OAB prevalence to be 15% in US women, with 11% reporting UUI.2 OAB represents a significant health care burden that impacts nearly every aspect of life, including physical, emotional, and psychological domains.3,4 The economic impact is notable; the projected cost is estimated to reach $82.6 billion annually by 2020.5
The American Urological Association (AUA) and the Society for Urodynamics, Female Pelvic Medicine and Urogenital Reconstruction (SUFU) have endorsed an algorithm for use in the evaluation of idiopathic OAB (FIGURE).6 If the patient's symptoms are certain, minimal evaluation is needed and it is reasonable to proceed with first-line therapy, which includes fluid management (decreasing caffeine intake and limiting evening fluid intake), bladder retraining drills such as timed voiding, and improving pelvic floor muscles with the use of biofeedback and functional electrical stimulation.6,7 Pelvic floor muscle training can be facilitated with a referral to a physical therapist trained in pelvic floor muscle education.
If treatment goals are not met with first-line strategies, second-line therapy may be initiated with anticholinergic or β3-adrenergic receptor agonist medications. If symptoms persist after 4 to 8 weeks of pharmacologic therapy, clinicians are encouraged to reassess or refer the patient to a specialist. Further evaluation may include a bladder diary in which the patient documents voided volumes, voiding frequency, and number of incontinent episodes; symptom-specific questionnaires; and/or urodynamic testing.
Related article:
The latest treatments for urinary and fecal incontinence: Which hold water?
Based on that evaluation, the patient may be a candidate for third-line therapy with either intradetrusor onabotulinumtoxinA, posterior tibial nerve stimulation (PTNS), or sacral neuromodulation.
There is a paucity of information comparing third-line therapies. In this Update, we focus on 4 randomized clinical trials that compare third-line treatment options for idiopathic OAB.
Read about how anticholinergic medication and onabotulinumtoxinA compare for treating UUI.
Anticholinergic therapy and onabotulinumtoxinA produce equivalent reductions in the frequency of daily UUI episodes
Visco AG, Brubaker L, Richter HE, et al; for the Pelvic Floor Disorders Network. Anticholinergic therapy vs onabotulinumtoxinA for urgency urinary incontinence. N Engl J Med. 2012;367(19):1803-1813.
In a double-blind, double-placebo-controlled randomized trial, Visco and colleagues compared anticholinergic medication with onabotulinumtoxinA 100 U for the treatment of women with UUI.
Details of the study
Two hundred forty-one women with moderate to severe UUI received either 6 months of oral anticholinergic therapy (solifenacin 5 mg daily with the option of dose escalation to 10 mg daily or change to trospium XR 60 mg daily based on the Patient Global Symptom Control score) plus a single intradetrusor injection of saline, or a single intradetrusor injection of onabotulinumtoxinA 100 U plus a 6-month oral placebo regimen.
Inclusion criteria were 5 or more UUI episodes on a 3-day diary, insufficient resolution of symptoms after 2 medications, or being drug naive. Exclusions included a postvoid residual (PVR) urine volume greater than 150 mL or previous therapy with onabotulinumtoxinA.
Participants were scheduled for follow up every 2 to 6 months post randomization, at which time all study medications were discontinued. The primary outcome was reduction from baseline in the mean number of UUI episodes per day over the 6-month period, as recorded in the monthly 3-day bladder diaries. Secondary outcomes included the proportion of participants with complete resolution of UUI, the proportion of participants with 75% or more reduction in UUI episodes, Overactive Bladder Questionnaire Short Form (OABq-SF) scores, other symptom-specific questionnaire scores, and adverse events.
Related article:
Which treatments for pelvic floor disorders are backed by evidence?
Both treatments significantly reduced UUI episodes
At baseline, participants reported a mean (SD) of 5.0 (2.7) UUI episodes per day, and 41% of participants were drug naive. Both treatment groups experienced significant reductions compared with baseline in mean UUI episodes, and the reductions were similar between the 2 groups (reduction of 3.4 episodes per day in the anticholinergic group, reduction of 3.3 episodes in the onabotulinumtoxinA group; P = .81). Complete resolution of UUI was more common in the onabotulinumtoxinA group (27%) as compared with the anticholinergic group (13%) (P = .003). There were no differences in improvement in OABq-SF scores (37.05 in the anticholinergic group vs 37.13 in the onabotulinumtoxinA group; P = .98) or other quality-of-life measures.
Adverse events. The anticholinergic group experienced a higher rate of dry mouth compared with the onabotulinumtoxinA group (46% vs 31%; P = .02) but had lower rates of intermittent catheterization use at 2 months (0% vs 5%, P = .01) and UTIs (13% vs 33%, P<.001).
Strengths and limitations. This was a well-designed, multicenter, randomized double-blind, double placebo-controlled trial. The study design allowed for dose escalation and change to another medication for inadequate symptom control and included drug-naive participants, which increases the generalizability of the results. However, current guidelines recommend reserving onabotulinumtoxinA therapy for third-line therapy, thus deterring this treatment's use in the drug-naive population. Additionally, the lack of a pure placebo arm makes it difficult to interpret the extent to which a placebo effect contributed to observed improvements in clinical symptoms.
Through 6 months, both a single intradetrusor injection of onabotulinumtoxinA 100 U and anticholinergic therapy reduce UUI episodes and improve quality-of-life measures in women who have failed medications or are drug naive. Use of onabotulinumtoxinA, however, more likely will lead to complete resolution of UUI, although with an increased risk of transient urinary retention and UTI. Even given the study findings supporting the use of onabotulinumtoxinA over anticholinergic therapy for complete resolution of UUI, it is most appropriate to align with current practice, which includes a trial of pharmacotherapy before proceeding with third-line onabotulinumtoxinA.
Read: onabotulinumtoxinA vs PTNS for OAB.
OnabotulinumtoxinA has greater 9-month durability for OAB symptoms compared with12 weeks of PTNS
Sherif H, Khalil M, Omar R. Management of refractory idiopathic overactive bladder: intradetrusor injection of botulinum toxin type A versus posterior tibial nerve stimulation. Can J Urol. 2017;24(3):8838-8846.
In this randomized clinical trial, Sherif and colleagues compared the safety and efficacy of a single intradetrusor injection of onabotulinumtoxinA 100 U with that of PTNS for OAB.
Details of the study
Sixty adult men and women with OAB who did not respond to medical therapy were randomly assigned to treatment with either onabotulinumtoxinA 100 U or PTNS. Criteria for exclusion were current UTI, PVR urine volume of more than 150 mL, previous radiation therapy or chemotherapy, previous incontinence surgery or bladder malignancy, or presence of mixed urinary incontinence.
At baseline, participants completed a 3-day bladder diary, an OAB symptom score (OABSS) questionnaire, and urodynamic testing. The OABSS questionnaire included 7 questions (scoring range, 0-28), with higher scores indicating worse symptoms, and included subscales for urgency and quality-of-life measures. Total OABSS, urgency score, quality-of-life score, bladder diary records, and urodynamic testing parameters were assessed at 6, 12, 24, and 36 weeks, along with adverse events.
OnabotulinumtoxinA injections were performed under spinal anesthesia. If PVR urine volume was greater than 200 mL at any follow-up visit, participants were instructed to begin clean intermittent self-catheterization. PTNS was administered as weekly 30-minute sessions for 12 consecutive weeks.
Participants' baseline demographics and symptoms were similar. Average age was 45 years. Averages (SD) for duration of anticholinergic use was 13 (0.8) weeks, UUI episode score was 4.5 (1) on 3-day bladder diary, and OABSS was 22 (2.7). Nine-month data were available for 29 participants in the onabotulinumtoxinA group and for 8 in the PTNS group.
Related article:
Update on pelvic floor dysfunction: Focus on urinary incontinence
OnabotulinumtoxinA treatment benefits sustained for 9 months
Through 6 months, compared with baseline assessments, both treatment groups had significant improvements in clinical symptoms and OABSS total score, as well as urgency and quality-of-life subscales. At 3 months, urodynamic study parameters were similarly improved from baseline in both groups.
At 9 months, however, only the onabotulinumtoxinA group, compared with the PTNS group, maintained the significant improvement from baseline in 3-day bladder diary voiding episodes (average [SD], 10.7 [1.01] vs 11.6 [1.09]; P = .009), 3-day bladder diary nocturia episodes (average [SD], 3.8 [1.09] vs 4.4 [0.8]; P = .02), and average [SD] UUI episodes over 3 days (3.5 [1.2] vs 4.2 [1.04]; P = .02). Similarly, onabotulinumtoxinA-treated participants, compared with those treated with PTNS, maintained improvements at 9 months in average (SD): OABSS total score (19.2 [2.4] vs 20.4 [1.7]; P = .03), urgency scores (10.9 [1.3] vs 11.8 [1.4]; P = .009), urine volume at first desire (177.8 [9.2] vs 171.8 [7.7]), maximum cystometric capacity (304 [17.6] vs 290 [13.1]), and Qmax (mL/sec) (20.7 [1.6] vs 22.2 [1.2]).
Adverse events. Average PVR urine volumes were higher in the onabotulinumtoxinA group compared with the PTNS group (36.8 [2.7] vs 32.4 [3.03]; P = .0001) at all time points, and self-catheterization was required in 6.6% of onabotulinumtoxinA-treated participants. Urinary tract infection occurred in 6.6% of participants in the onabotulinumtoxinA group and in none of the PTNS group. In the PTNS group, few experienced pain and minor bleeding at the needle site.
Strengths and limitations. This randomized, open-label trial comparing treatment with onabotulinumtoxinA 100 U and PTNS included both men and women with idiopathic OAB symptoms. The participants were assessed at regular intervals with various measures, and follow-up adherence was good. The sample size was small, so the study may not have been powered to see differences prior to 9 months.
Although at 9 months only the onabotulinumtoxinA group maintained significant improvement over baseline levels, the improvement was diminished, and therefore the clinical meaningfulness is uncertain. Further, participants in the PTNS group did not undergo monthly maintenance therapy after 3 months, which is recommended for those with a 12-week therapeutic response; this may have affected 9-month outcomes in this group. Since the one-time onabotulinumtoxinA 100 U injection was performed under spinal anesthesia, cost comparisons should be considered, since future onabotulinumtoxinA injections would be necessary.
A one-time onabotulinumtoxinA 100 U injection and 12 weeks of PTNS therapy are reasonable short-term options for symptomatic OAB relief after unsuccessful therapy with medications. OnabotulinumtoxinA injection may provide more durable OAB symptom control at 9 months but with a risk of UTI and need for self-catheterization.
Read about using different doses of onabotulinumtoxinA for OAB.
OnabotulinumtoxinA 200-U injection provides longer OAB symptom improvement than 100-U injection
Abdelwahab O, Sherif H, Soliman T, Elbarky I, Eshazly A. Efficacy of botulinum toxin type A 100 units versus 200 units for treatment of refractory idiopathic overactive bladder. Int Braz J Urol. 2015;41(6):1132-1140.
Abdelwahab and colleagues conducted a single-center, randomized clinical trial to investigate the safety and efficacy of a single injection of intradetrusor onabotulinumtoxinA in 2 different doses (100 U and 200 U) for treatment of OAB.
Details of the study
Eighty adults (63 women, 17 men) who did not benefit from anticholinergic medication during the previous 3 months were randomly assigned to receive either a 100-U (n = 40) or a 200-U (n = 40) injection of onabotulinumtoxinA. Exclusion criteria were PVR urine volume greater than 150 mL and previous radiation therapy or chemotherapy.
Initial assessments -- completed at baseline and at 1, 3, 6, and 9 months -- included the health-related quality-of-life (HR-QOL) questionnaire (maximum score, 100; higher score indicates better quality of life), an abbreviated OABSS questionnaire (4 questions; score range, 0-15; higher score indicates more severe symptoms), and urodynamic evaluation. Outcomes included OABSS, HR-QOL score, and urodynamic parameters at the various time points.
Related article:
Is there a link between impaired mobility and urinary incontinence in elderly, community-dwelling women?
Higher dose, greater symptom improvement and higher adverse event rate
At baseline, participants (average age, 31 years) had an average (SD) OABSS of 1.7 (1.6). OnabotulinumtoxinA treatment with both a 100-U and a 200-U dose resulted in significant improvements (compared with baseline levels) in frequency, nocturia, UUI episodes, OABSS, and urodynamic parameters throughout the 9 months. At 9 months, however, the group treated with the 200-U dose had greater improvements, compared with the group who received a 100-U dose, in urinary frequency symptom scores (mean [SD], 0.32 [0.47] vs 1.1 [0.51]; P<.05), nocturia symptom scores (mean [SD], 0.13 [0.34] vs 0.36 [0.49]; P<.05), UUI symptom scores (mean [SD], 0.68 [0.16] vs 1.26 [1.1]; P<.05), and mean (SD) total OABSS (2.6 [2.31] vs 5.3 [2.11]; P<.05). Similarly, at 9 months the 200-U dose resulted in greater improvements in volume at first desire (mean [SD], 291.8 [42.8] vs 246.8 [53.8] mL; P<.05), volume at strong desire (mean [SD], 392.1 [37.3] vs 313.1 [67.4] mL; P<.05), detrusor pressure (mean [SD], 10.4 [4.0] vs 19.2 [7.8] cm H2O; P<.05), and maximum cystometric capacity (mean [SD], 430.5 [34.2] vs 350 [69.1] mL; P<.05) compared with the 100-U dose.
Adverse events. No participant had a PVR urine volume greater than 100 mL at any follow-up visit. Postoperative hematuria occurred in 23% of the group treated with onabotulinumtoxinA 200 U versus in 15% of those treated with a 100-U dose. Similarly, UTIs occurred in 17.5% of the 200-U dose group and in 7.5% of the 100-U dose group. Dysuria was reported in 37.5% and 15% of the 200-U and 100-U dose groups, respectively.
Strengths and limitations. This randomized, open-label trial comparing a single injection of 100 U versus 200 U of onabotulinumtoxinA included mostly women. OAB symptoms and urodynamic parameters improved after treatment with both dose levels, but a longer duration of improvement was seen with the 200-U dose. The cohort had a low baseline OAB severity, based on the OABSS questionnaire, and a young average age of participants, which limits the generalizability of the study results to a population with refractory OAB. The 0% rate of clean intermittent self-catheterization postinjection might be based on the study's criteria for requiring clean intermittent catheterization. In addition, the initial postinjection visit occurred at 1 month, possibly missing participants who had symptoms of retention soon after injection.
Two dose levels (100 U and 200 U) of a single injection of onabotulinumtoxinA are associated with comparable OAB symptom and urodyanamic improvements. The benefits of a longer duration of effect with the 200-U dose must be weighed against the possible higher risks of transient hematuria, dysuria, and UTI.
Read: onabotulinumtoxinA vs sacral neuromodulation therapy for UUI.
Treatment with onabotulinumtoxinA may control UUI symptoms better than sacral neuromodulation therapy
Amundsen CL, Richter HE, Menefee SA, et al; Pelvic Floor Disorders Network. OnabotulinumtoxinA vs sacral neuromodulation on refractory urgency urinary incontinence in women: a randomized clinical trial. JAMA. 2016;316(13):1366-1374.
In this multicenter open-label randomized trial, Amundsen and colleagues compared the efficacy and safety of onabotulinumtoxinA 200 U with that of sacral neuromodulation.
Details of the study
Three hundred sixty-four women with UUI had data available for primary analysis at 6 months. Women were considered eligible for the study if they had 6 or more UUI episodes on a 3-day bladder diary, persistent symptoms despite anticholinergic therapy, a PVR urine volume of less than 150 mL, and had never previously received either study treatment.
There were no differences in baseline characteristics of the participants. The average (SD) age of the study population was 63 (11.6) years, with an average (SD) daily number of UUI episodes of 5.3 (2.8). The average (SD) body mass index was 32 (8) kg/m2.
Participants were randomly assigned to undergo either sacral neuromodulation (n = 174) or intradetrusor injection of onabotulinumtoxinA 200 U (n = 190). The primary outcome was change from baseline in mean number of daily UUI episodes averaged over 6 months as recorded on a monthly 3-day bladder diary. Secondary outcomes included complete resolution of urgency incontinence, 75% or more reduction in UUI episodes, the Overactive Bladder Questionnaire Short Form (SF) score (range, 0-100; higher score indicates higher symptom severity), the Overactive Bladder Satisfaction of Treatment questionnaire (range, 0-100; higher score indicates better satisfaction), other quality-of-life measures, and adverse events.
Related article:
2015 Update on pelvic floor dysfunction: Bladder pain syndrome
Greater symptom bother improvement, treatment satisfaction with onabotulinumtoxinA 200 U
Participants treated with onabotulinumtoxinA had a greater mean reduction of 3.9 UUI episodes per day than the sacral neuromodulation group's reduction of 3.3 UUI episodes per day (mean difference, 0.63; 95% confidence interval [CI], 0.13-1.14; P = .01). In addition, complete UUI resolution was higher in the onabotulinumtoxinA group as compared with the sacral neuromodulation group (20% vs 4%; P<.001). The onabotulinumtoxinA group also had higher rates of 75% or more reduction of UUI episodes compared with the sacral neuromodulation group (46% vs 26%; P<.001). Over 6 months, both groups had improvements in all quality-of-life measures, but the onabotulinumtoxinA group had greater improvement in symptom bother compared with the sacral neuromodulation group (-46.7 vs -38.6; mean difference, 8.1; 95% CI, 3.0-13.3; P = .002). Furthermore, the onabotulinumtoxinA group had greater treatment satisfaction compared with the sacral neuromodulation group (mean difference, 7.8; 95% CI, 1.6-14.1; P = .01).
Adverse events. Six women (3%) underwent sacral neuromodulation device revision or removal. Approximately 8% of onabotulinumtoxinA-treated participants required intermittent self-catheterization at 1 month, 4% at 3 months, and 2% at 6 months. The risk of UTI was higher in the onabotulinumtoxinA group compared with the sacral neuromodulation group (35% vs 11%; risk difference, 23%; 95% CI, -33% to -13%; P<.001).
Strengths and limitations. This is a well-designed randomized clinical trial comparing clinical outcomes and adverse events after treatment with onabotulinumtoxinA 200-U versus sacral neuromodulation. The interventions were standardized across investigators at multiple sites, and the study design required close follow-up to assess efficacy and adverse events. The study used a 200-U dose based on reported durability of effect at that time and findings of equivalency between onabotulinumtoxinA 100 U and anticholinergic therapy. The US Food and Drug Administration's recommendation to use a 100-U dose in all patients with idiopathic OAB might dissuade clinicians from considering the higher dose of onabotulinumtoxinA. The study was limited by the lack of a placebo group.
Both onabotulinumtoxinA 200 U and sacral neuromodulation provide significant improvement in UUI episodes and quality of life over 6 months. However, while treatment with onabotulinumtoxinA has a likelihood of complete UUI resolution, greater improvements in symptom bother and treatment satisfaction, these benefits must be weighed against the risks of transient catheterization and UTI.
Share your thoughts! Send your Letter to the Editor to [email protected]. Please include your name and the city and state in which you practice.
- Haylen BT, de Ridder D, Freeman RM, et al; International Urogynecological Association; International Continence Society. An International Urogynecological Association (IUGA)/International Continence Society (ICS) joint report on the terminology for female pelvic floor dysfunction. Neurourol Urodyn. 2010;29(1):4-20.
- Hartmann KE, McPheeters ML, Biller DH, et al. Treatment of overactive bladder in women. Evid Rep Technol Assess (Full Rep). 2009;187:1-120.
- Reynolds,WS, Fowke J, Dmochowski, R. The burden of overactive bladder on US public health. Curr Bladder Dysfunct Rep. 2016;11(1):8-13.
- Willis-Gray MG, Dieter AA, Geller EJ. Evaluation and management of overactive bladder: strategies for optimizing care. Res Rep Urol. 2016;8:113-122.
- Ganz ML, Smalarz AM, Krupski TL, et al. Economic costs of overactive bladder in the United States. Urology. 2010;75(3):526-532.
- Gormley EA, Lightner DJ, Faraday M, Vasavada SP; American Urological Association; Society of Urodyndamics, Female Pelvic Medicine. Diagnosis and treatment of overactive bladder (non-neurogenic) in adults: AUA/SUFU guideline amendment. J Urol. 2015; 193(5):1572-1580.
- Gormley EA, Lightner DJ, Burgio KL, et al; American Urological Association; Society of Urodynamics, Female Pelvic Medicine & Urogenital Reconstruction. Diagnosis and treatment of overactive bladder (non-neurogenic) in adults: AUA/SUFU guideline. J Urol. 2012;188(6 suppl):2455-2463.
- Haylen BT, de Ridder D, Freeman RM, et al; International Urogynecological Association; International Continence Society. An International Urogynecological Association (IUGA)/International Continence Society (ICS) joint report on the terminology for female pelvic floor dysfunction. Neurourol Urodyn. 2010;29(1):4-20.
- Hartmann KE, McPheeters ML, Biller DH, et al. Treatment of overactive bladder in women. Evid Rep Technol Assess (Full Rep). 2009;187:1-120.
- Reynolds,WS, Fowke J, Dmochowski, R. The burden of overactive bladder on US public health. Curr Bladder Dysfunct Rep. 2016;11(1):8-13.
- Willis-Gray MG, Dieter AA, Geller EJ. Evaluation and management of overactive bladder: strategies for optimizing care. Res Rep Urol. 2016;8:113-122.
- Ganz ML, Smalarz AM, Krupski TL, et al. Economic costs of overactive bladder in the United States. Urology. 2010;75(3):526-532.
- Gormley EA, Lightner DJ, Faraday M, Vasavada SP; American Urological Association; Society of Urodyndamics, Female Pelvic Medicine. Diagnosis and treatment of overactive bladder (non-neurogenic) in adults: AUA/SUFU guideline amendment. J Urol. 2015; 193(5):1572-1580.
- Gormley EA, Lightner DJ, Burgio KL, et al; American Urological Association; Society of Urodynamics, Female Pelvic Medicine & Urogenital Reconstruction. Diagnosis and treatment of overactive bladder (non-neurogenic) in adults: AUA/SUFU guideline. J Urol. 2012;188(6 suppl):2455-2463.
Breast density and optimal screening for breast cancer
MY STORY: Prologue
My aunt received a breast cancer diagnosis at age 40, and she died at age 60, in 1970. Then, in 1975, my mother’s breast cancer was found at age 55, but only after she was examined for nipple retraction; on mammography, the cancer had been obscured by dense breast tissue. Mom had 2 metastatic nodes but participated in the earliest clinical trials of chemotherapy and lived free of breast cancer for another 41 years. Naturally I thought that, were I to develop this disease, I would want it found earlier. Ironically, it was, but only because I had spent my career trying to understand the optimal screening approaches for women with dense breasts—women like me.
Cancers are masked on mammography in dense breasts
For women, screening mammography is an important step in reducing the risk of dying from breast cancer. The greatest benefits are realized by those who start annual screening at age 40, or 45 at the latest.1 As it takes 9 to 10 years to see a benefit from breast cancer screening at the population level, it is not logical to continue this testing when life expectancy is less than 10 years, as is the case with women age 85 or older, even those in the healthiest quartile.2–4 However, despite recent advances, the development of 3D mammography (tomosynthesis) (FIGURE 1) in particular, cancers can still be masked by dense breast tissue. Both 2D and 3D mammograms are x-rays; both dense tissue and cancers absorb x-rays and appear white.
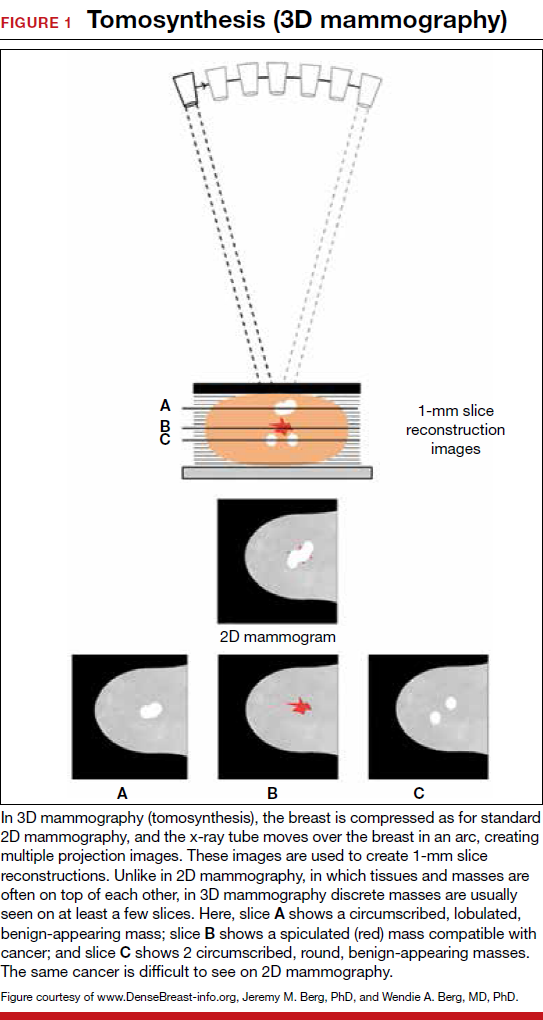
Breast density is determined on mammography and is categorized as fatty, scattered fibroglandular, heterogeneously dense, or extremely dense (FIGURE 2).5 Tissue in the heterogeneous and extreme categories is considered dense. More than half of women in their 40s have dense breasts; with some fatty involution occurring around menopause, the proportion drops to 25% for women in their 60s.6 About half of breast cancers have calcifications, which on mammography are usually easily visible even in dense breasts. The problem is with noncalcified invasive cancers that can be hidden by dense tissue (FIGURE 3).
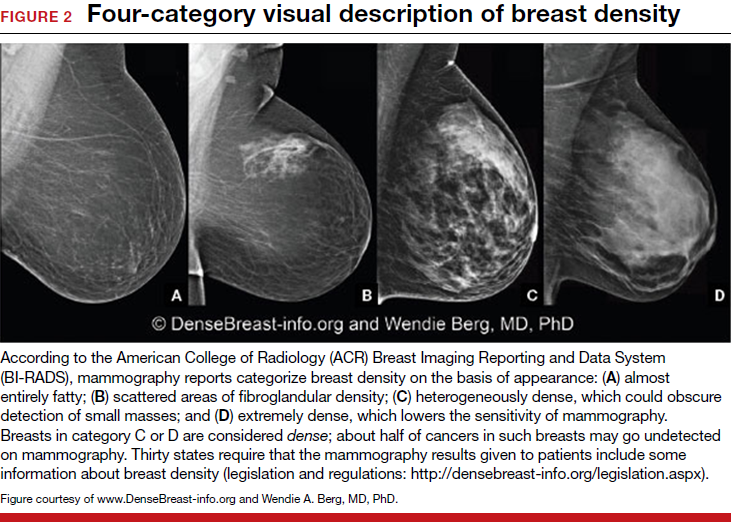
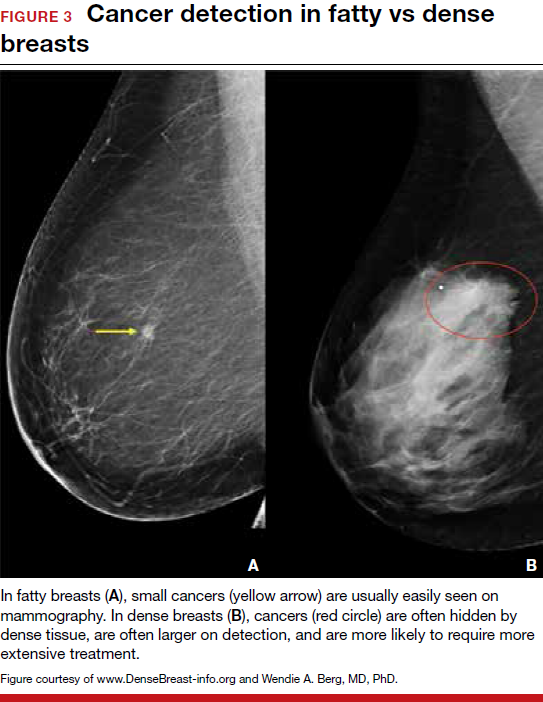
3D mammography improves cancer detection but is of minimal benefit in extremely dense breasts
Although 3D mammography improves cancer detection in most women, any benefit is minimal in women with extremely dense breasts, as there is no inherent soft-tissue contrast.7 Masked cancers are often only discovered because of a lump after a normal screening mammogram, as so-called “interval cancers.” Compared with screen-detected cancers, interval cancers tend to be more biologically aggressive, to have spread to lymph nodes, and to have worse prognoses. However, even some small screen-detected cancers are biologically aggressive and can spread to lymph nodes quickly, and no screening test or combination of screening tests can prevent this occurrence completely, regardless of breast density.
Related article:
Get smart about dense breasts
MRI provides early detection across all breast densities
In all tissue densities, contrast-enhanced magnetic resonance imaging (MRI) is far better than mammography in detecting breast cancer.8 Women at high risk for breast cancer caused by mutations in BRCA1, BRCA2, p53, and other genes have poor outcomes with screening mammography alone—up to 50% of cancers are interval cancers. Annual screening MRI reduces this percentage significantly, to 11% in women with pathogenic BRCA1 mutations and to 4% in women with BRCA2 mutations.9 Warner and colleagues found a decrease in late-stage cancers in high-risk women who underwent annual MRI screenings compared to high-risk women unable to have MRI.10
The use of MRI for screening is limited by availability, patient tolerance,11 and high cost. Research is being conducted to further validate approaches using shortened screening MRI times (so-called “abbreviated” or “fast” MRI) and, thereby, improve access, tolerance, and reduce associated costs; several investigators already have reported promising results, and a few centers offer this modality directly to patients willing to pay $300 to $350 out of pocket.12,13 Even in normal-risk women, MRI significantly increases detection of early breast cancer after a normal mammogram and ultrasound, and the cancer detection benefit of MRI is seen across all breast densities.14
Most health insurance plans cover screening MRI only for women who meet defined risk criteria, including women who have a known disease-causing mutation—or are suspected of having one, given a family history of breast cancer with higher than 20% to 25% lifetime risk by a model that predicts mutation carrier status—as well as women who had chest radiation therapy before age 30, typically for Hodgkin lymphoma, and at least 8 years earlier.15 In addition, MRI can be considered in women with atypical breast biopsy results or a personal history of lobular carcinoma in situ (LCIS).16
Screening MRI should start by age 25 in women with disease-causing mutations, or at the time of atypical or LCIS biopsy results, and should be performed annually unless the woman is pregnant or has a metallic implant, renal insufficiency, or another contraindication to MRI. MRI can be beneficial in women with a personal history of cancer, although annual mammography remains the standard of care.17–19
MRI and mammography can be performed at the same time or on an alternating 6-month basis, with mammography usually starting only after age 30 because of the small risk that radiation poses for younger women. There are a few other impediments to having breast MRI: The woman must lie on her stomach within a confined space (tunnel), the contrast that is injected may not be well tolerated, and insurance does not cover the test for women who do not meet the defined risk criteria.11
Read why mammography supplemented by US is best for women with dense breasts.
Ultrasonography supplements mammography
Mammography supplemented with ultrasonography (US) has been studied as a “Goldilocks” or best-fit solution for the screening of women with dense breasts, as detection of invasive cancers is improved with the 2 modalities over mammography alone, and US is less invasive, better tolerated, and lower in cost than the more sensitive MRI.
In women with dense breasts, US has been found to improve cancer detection over mammography alone, and early results suggest a larger cancer detection benefit from US than from 3D mammography, although research is ongoing.20 Adding US reduces the interval cancer rate in women with dense breasts to less than 10% of all cancers found—similar to results for women with fatty breasts.17,21,22
US can be performed by a trained technologist or a physician using a small transducer, which usually provides diagnostic images (so that most callbacks would be for a true finding), or a larger transducer and an automated system can be used to create more than a thousand images for radiologist review.23,24 Use of a hybrid system, a small transducer with an automated arm, has been validated as well.25 Screening US is not available universally, and with all these approaches optimal performance requires trained personnel. Supplemental screening US usually is covered by insurance but is nearly always subject to a deductible/copay.
Related article:
Educate patients about dense breasts and cancer risk
Reducing false-positives, callbacks, and additional testing
Mammography carries a risk of false-positives. On average, 11% to 12% of women are called back for additional testing after a screening mammogram, and in more than 95% of women brought back for extra testing, no cancer is found.26 Women with dense breasts are more likely than those with less dense breasts to be called back.27 US and MRI improve cancer detection and therefore yield additional positive, but also false-positive, findings. Notably, callbacks decrease after the first round of screening with any modality or combination of tests, as long as prior examinations are available for comparison.
One advantage of 3D over 2D mammography is a decrease in extra testing for areas of asymmetry, which are often recognizable on 3D mammography as representing normal superimposed tissue.28–30 Architectural distortion, which is better seen on 3D mammography and usually represents either cancer or a benign radial scar, can lead to false-positive biopsies, although the average biopsy rate is no higher for 3D than for 2D alone.31 Typically, the 3D and 2D examinations are performed together (slightly more than doubling the radiation dose), or synthetic 2D images can be created from the 3D slices (resulting in a total radiation dose almost the same as standard 2D alone).
Most additional cancers seen on 3D mammography or US are lower-grade invasive cancers with good prognoses. Some aggressive high-grade breast cancers go undetected even when mammography is supplemented with US, either because they are too small to be seen or because they resemble common benign masses and may not be recognized. MRI is particularly effective in depicting high-grade cancers, even small ones.
The TABLE summarizes the relative rates of cancer detection and additional testing by various breast screening tests or combinations of tests. Neither clinical breast examination by a physician or other health care professional nor routine breast self-examination reduces the number of deaths caused by breast cancer. Nevertheless, women should monitor any changes in their breasts and report these changes to their clinician. A new lump, skin or nipple retraction, or a spontaneous clear or bloody nipple discharge merits diagnostic breast imaging even if a recent screening mammogram was normal.
FIGURE 4 is an updated decision support tool that suggests strategies for optimizingcancer detection with widely available screening methods.
Read how to take advantage of today’s technology for breast density screening
MY STORY: Epilogue
My annual 3D mammograms were normal, even the year my cancer was present. In 2014, I entered my family history into the IBIS Breast Cancer Risk Evaluation Tool (Tyrer-Cuzick model of breast cancer risk) (http://www.ems-trials.org/riskevaluator/) and calculated my lifetime risk at 19.7%. That is when I decided to have a screening MRI. My invasive breast cancer was easily seen on MRI and then on US. The cancer was node-negative, easily confirmed with needle biopsy, and treated with lumpectomy and radiation. There was no need for chemotherapy.
My personal experience prompted me to join JoAnn Pushkin and Cindy Henke-Sarmento, RT(R)(M), BA, in developing a website, www.DenseBreast-info.org, to give women and their physicians easy access to information on making decisions about screening in dense breasts.
My colleagues and I are often asked what is the best way to order supplemental imaging for a patient who may have dense breasts. Even in cases in which a mammogram does not exist or is unavailable, the following prescription can be implemented easily at centers that offer US: “2D plus 3D mammogram if available; if dense, perform ultrasound as needed.”
Related article:
DenseBreast-info.org: What this resource can offer you, and your patients
Breast density screening: Take advantage of today’s technology
Breast screening and diagnostic imaging have improved significantly since the 1970s, when many of the randomized trials of mammography were conducted. Breast density is one of the most common and important risk factors for development of breast cancer and is now incorporated into the Breast Cancer Surveillance Consortium model (https://tools.bcsc-scc.org/BC5yearRisk/calculator.htm) and the Tyrer-Cuzick model (see also http://densebreast-info.org/explanation-of-dense-breast-risk-models.aspx).32 Although we continue to validate newer approaches, women should take advantage of the improved methods of early cancer detection, particularly if they have dense breasts or are at high risk for breast cancer.
Share your thoughts! Send your Letter to the Editor to [email protected]. Please include your name and the city and state in which you practice.
- Oeffinger KC, Fontham ET, Etzioni R, et al; American Cancer Society. Breast cancer screening for women at average risk: 2015 guideline update from the American Cancer Society. JAMA. 2015;314(15):1599–1614.
- Tabar L, Yen MF, Vitak B, Chen HH, Smith RA, Duffy SW. Mammography service screening and mortality in breast cancer patients: 20-year follow-up before and after introduction of screening. Lancet. 2003;361(9367):1405–1410.
- Lee SJ, Boscardin WJ, Stijacic-Cenzer I, Conell-Price J, O’Brien S, Walter LC. Time lag to benefit after screening for breast and colorectal cancer: meta-analysis of survival data from the United States, Sweden, United Kingdom, and Denmark. BMJ. 2013;346:e8441.
- Walter LC, Covinsky KE. Cancer screening in elderly patients: a framework for individualized decision making. JAMA. 2001;285(21):2750–2756.
- Sickles EA, D’Orsi CJ, Bassett LW, et al. ACR BI-RADS mammography. In: D’Orsi CJ, Sickles EA, Mendelson EB, et al, eds. ACR BI-RADS Atlas, Breast Imaging Reporting and Data System. 5th ed. Reston, VA: American College of Radiology; 2013.
- Sprague BL, Gangnon RE, Burt V, et al. Prevalence of mammographically dense breasts in the United States. J Natl Cancer Inst. 2014;106(10).
- Rafferty EA, Durand MA, Conant EF, et al. Breast cancer screening using tomosynthesis and digital mammography in dense and nondense breasts. JAMA. 2016;315(16):1784–1786.
- Berg WA. Tailored supplemental screening for breast cancer: what now and what next? AJR Am J Roentgenol. 2009;192(2):390–399.
- Heijnsdijk EA, Warner E, Gilbert FJ, et al. Differences in natural history between breast cancers in BRCA1 and BRCA2 mutation carriers and effects of MRI screening—MRISC, MARIBS, and Canadian studies combined. Cancer Epidemiol Biomarkers Prev. 2012;21(9):1458–1468.
- Warner E, Hill K, Causer P, et al. Prospective study of breast cancer incidence in women with a BRCA1 or BRCA2 mutation under surveillance with and without magnetic resonance imaging. J Clin Oncol. 2011;29(13):1664–1669.
- Berg WA, Blume JD, Adams AM, et al. Reasons women at elevated risk of breast cancer refuse breast MR imaging screening: ACRIN 6666. Radiology. 2010;254(1):79–87.
- Kuhl CK, Schrading S, Strobel K, Schild HH, Hilgers RD, Bieling HB. Abbreviated breast magnetic resonance imaging (MRI): first postcontrast subtracted images and maximum-intensity projection—a novel approach to breast cancer screening with MRI. J Clin Oncol. 2014;32(22):2304–2310.
- Strahle DA, Pathak DR, Sierra A, Saha S, Strahle C, Devisetty K. Systematic development of an abbreviated protocol for screening breast magnetic resonance imaging. Breast Cancer Res Treat. 2017;162(2):283–295.
- Kuhl CK, Strobel K, Bieling H, Leutner C, Schild HH, Schrading S. Supplemental breast MR imaging screening of women with average risk of breast cancer. Radiology. 2017;283(2):361–370.
- Saslow D, Boetes C, Burke W, et al; American Cancer Society Breast Cancer Advisory Group. American Cancer Society guidelines for breast screening with MRI as an adjunct to mammography. CA Cancer J Clin. 2007;57(2):75–89.
- National Comprehensive Cancer Network. NCCN guidelines for detection, prevention, and risk reduction: breast cancer screening and diagnosis. https://www.nccn.org/professionals/physician_gls/pdf/breast-screening.pdf.
- Berg WA, Zhang Z, Lehrer D, et al; ACRIN 6666 Investigators. Detection of breast cancer with addition of annual screening ultrasound or a single screening MRI to mammography in women with elevated breast cancer risk. JAMA. 2012;307(13):1394–1404.
- Brennan S, Liberman L, Dershaw DD, Morris E. Breast MRI screening of women with a personal history of breast cancer. AJR Am J Roentgenol. 2010;195(2):510–516.
- Lehman CD, Lee JM, DeMartini WB, et al. Screening MRI in women with a personal history of breast cancer. J Natl Cancer Inst. 2016;108(3).
- Tagliafico AS, Calabrese M, Mariscotti G, et al. Adjunct screening with tomosynthesis or ultrasound in women with mammography-negative dense breasts: interim report of a prospective comparative trial [published online ahead of print March 9, 2016]. J Clin Oncol. JCO634147.
- Corsetti V, Houssami N, Ghirardi M, et al. Evidence of the effect of adjunct ultrasound screening in women with mammography-negative dense breasts: interval breast cancers at 1 year follow-up. Eur J Cancer. 2011;47(7):1021–1026.
- Ohuchi N, Suzuki A, Sobue T, et al; J-START Investigator Groups. Sensitivity and specificity of mammography and adjunctive ultrasonography to screen for breast cancer in the Japan Strategic Anti-Cancer Randomized Trial (J-START): a randomised controlled trial. Lancet. 2016;387(10016):341–348.
- Berg WA, Mendelson EB. Technologist-performed handheld screening breast US imaging: how is it performed and what are the outcomes to date? Radiology. 2014;272(1):12–27.
- Brem RF, Tabár L, Duffy SW, et al. Assessing improvement in detection of breast cancer with three-dimensional automated breast US in women with dense breast tissue: the SomoInsight study. Radiology. 2015;274(3):663–673.
- Kelly KM, Dean J, Comulada WS, Lee SJ. Breast cancer detection using automated whole breast ultrasound and mammography in radiographically dense breasts. Eur Radiol. 2010;20(3):734–742.
- Lehman CD, Arao RF, Sprague BL, et al. National performance benchmarks for modern screening digital mammography: update from the Breast Cancer Surveillance Consortium. Radiology. 2017;283(1):49–58.
- Kerlikowske K, Zhu W, Hubbard RA, et al; Breast Cancer Surveillance Consortium. Outcomes of screening mammography by frequency, breast density, and postmenopausal hormone therapy. JAMA Intern Med. 2013;173(9):807–816.
- Friedewald SM, Rafferty EA, Rose SL, et al. Breast cancer screening using tomosynthesis in combination with digital mammography. JAMA. 2014;311(24):2499–2507.
- Skaane P, Bandos AI, Gullien R, et al. Comparison of digital mammography alone and digital mammography plus tomosynthesis in a population-based screening program. Radiology. 2013;267(1):47–56.
- Ciatto S, Houssami N, Bernardi D, et al. Integration of 3D digital mammography with tomosynthesis for population breast-cancer screening (STORM): a prospective comparison study. Lancet Oncol. 2013;14(7):583–589.
- Bahl M, Lamb LR, Lehman CD. Pathologic outcomes of architectural distortion on digital 2D versus tomosynthesis mammography [published online ahead of print August 23, 2017]. AJR Am J Roentgenol. doi:10.2214/AJR.17.17979.
- Engmann NJ, Golmakani MK, Miglioretti DL, Sprague BL, Kerlikowske K; Breast Cancer Surveillance Consortium. Population-attributable risk proportion of clinical risk factors for breast cancer [published online ahead of print February 2, 2017]. JAMA Oncol. doi:10.1001/jamaoncol.2016.6326.
MY STORY: Prologue
My aunt received a breast cancer diagnosis at age 40, and she died at age 60, in 1970. Then, in 1975, my mother’s breast cancer was found at age 55, but only after she was examined for nipple retraction; on mammography, the cancer had been obscured by dense breast tissue. Mom had 2 metastatic nodes but participated in the earliest clinical trials of chemotherapy and lived free of breast cancer for another 41 years. Naturally I thought that, were I to develop this disease, I would want it found earlier. Ironically, it was, but only because I had spent my career trying to understand the optimal screening approaches for women with dense breasts—women like me.
Cancers are masked on mammography in dense breasts
For women, screening mammography is an important step in reducing the risk of dying from breast cancer. The greatest benefits are realized by those who start annual screening at age 40, or 45 at the latest.1 As it takes 9 to 10 years to see a benefit from breast cancer screening at the population level, it is not logical to continue this testing when life expectancy is less than 10 years, as is the case with women age 85 or older, even those in the healthiest quartile.2–4 However, despite recent advances, the development of 3D mammography (tomosynthesis) (FIGURE 1) in particular, cancers can still be masked by dense breast tissue. Both 2D and 3D mammograms are x-rays; both dense tissue and cancers absorb x-rays and appear white.

Breast density is determined on mammography and is categorized as fatty, scattered fibroglandular, heterogeneously dense, or extremely dense (FIGURE 2).5 Tissue in the heterogeneous and extreme categories is considered dense. More than half of women in their 40s have dense breasts; with some fatty involution occurring around menopause, the proportion drops to 25% for women in their 60s.6 About half of breast cancers have calcifications, which on mammography are usually easily visible even in dense breasts. The problem is with noncalcified invasive cancers that can be hidden by dense tissue (FIGURE 3).


3D mammography improves cancer detection but is of minimal benefit in extremely dense breasts
Although 3D mammography improves cancer detection in most women, any benefit is minimal in women with extremely dense breasts, as there is no inherent soft-tissue contrast.7 Masked cancers are often only discovered because of a lump after a normal screening mammogram, as so-called “interval cancers.” Compared with screen-detected cancers, interval cancers tend to be more biologically aggressive, to have spread to lymph nodes, and to have worse prognoses. However, even some small screen-detected cancers are biologically aggressive and can spread to lymph nodes quickly, and no screening test or combination of screening tests can prevent this occurrence completely, regardless of breast density.
Related article:
Get smart about dense breasts

MRI provides early detection across all breast densities
In all tissue densities, contrast-enhanced magnetic resonance imaging (MRI) is far better than mammography in detecting breast cancer.8 Women at high risk for breast cancer caused by mutations in BRCA1, BRCA2, p53, and other genes have poor outcomes with screening mammography alone—up to 50% of cancers are interval cancers. Annual screening MRI reduces this percentage significantly, to 11% in women with pathogenic BRCA1 mutations and to 4% in women with BRCA2 mutations.9 Warner and colleagues found a decrease in late-stage cancers in high-risk women who underwent annual MRI screenings compared to high-risk women unable to have MRI.10
The use of MRI for screening is limited by availability, patient tolerance,11 and high cost. Research is being conducted to further validate approaches using shortened screening MRI times (so-called “abbreviated” or “fast” MRI) and, thereby, improve access, tolerance, and reduce associated costs; several investigators already have reported promising results, and a few centers offer this modality directly to patients willing to pay $300 to $350 out of pocket.12,13 Even in normal-risk women, MRI significantly increases detection of early breast cancer after a normal mammogram and ultrasound, and the cancer detection benefit of MRI is seen across all breast densities.14
Most health insurance plans cover screening MRI only for women who meet defined risk criteria, including women who have a known disease-causing mutation—or are suspected of having one, given a family history of breast cancer with higher than 20% to 25% lifetime risk by a model that predicts mutation carrier status—as well as women who had chest radiation therapy before age 30, typically for Hodgkin lymphoma, and at least 8 years earlier.15 In addition, MRI can be considered in women with atypical breast biopsy results or a personal history of lobular carcinoma in situ (LCIS).16
Screening MRI should start by age 25 in women with disease-causing mutations, or at the time of atypical or LCIS biopsy results, and should be performed annually unless the woman is pregnant or has a metallic implant, renal insufficiency, or another contraindication to MRI. MRI can be beneficial in women with a personal history of cancer, although annual mammography remains the standard of care.17–19
MRI and mammography can be performed at the same time or on an alternating 6-month basis, with mammography usually starting only after age 30 because of the small risk that radiation poses for younger women. There are a few other impediments to having breast MRI: The woman must lie on her stomach within a confined space (tunnel), the contrast that is injected may not be well tolerated, and insurance does not cover the test for women who do not meet the defined risk criteria.11
Read why mammography supplemented by US is best for women with dense breasts.
Ultrasonography supplements mammography
Mammography supplemented with ultrasonography (US) has been studied as a “Goldilocks” or best-fit solution for the screening of women with dense breasts, as detection of invasive cancers is improved with the 2 modalities over mammography alone, and US is less invasive, better tolerated, and lower in cost than the more sensitive MRI.
In women with dense breasts, US has been found to improve cancer detection over mammography alone, and early results suggest a larger cancer detection benefit from US than from 3D mammography, although research is ongoing.20 Adding US reduces the interval cancer rate in women with dense breasts to less than 10% of all cancers found—similar to results for women with fatty breasts.17,21,22
US can be performed by a trained technologist or a physician using a small transducer, which usually provides diagnostic images (so that most callbacks would be for a true finding), or a larger transducer and an automated system can be used to create more than a thousand images for radiologist review.23,24 Use of a hybrid system, a small transducer with an automated arm, has been validated as well.25 Screening US is not available universally, and with all these approaches optimal performance requires trained personnel. Supplemental screening US usually is covered by insurance but is nearly always subject to a deductible/copay.
Related article:
Educate patients about dense breasts and cancer risk
Reducing false-positives, callbacks, and additional testing
Mammography carries a risk of false-positives. On average, 11% to 12% of women are called back for additional testing after a screening mammogram, and in more than 95% of women brought back for extra testing, no cancer is found.26 Women with dense breasts are more likely than those with less dense breasts to be called back.27 US and MRI improve cancer detection and therefore yield additional positive, but also false-positive, findings. Notably, callbacks decrease after the first round of screening with any modality or combination of tests, as long as prior examinations are available for comparison.
One advantage of 3D over 2D mammography is a decrease in extra testing for areas of asymmetry, which are often recognizable on 3D mammography as representing normal superimposed tissue.28–30 Architectural distortion, which is better seen on 3D mammography and usually represents either cancer or a benign radial scar, can lead to false-positive biopsies, although the average biopsy rate is no higher for 3D than for 2D alone.31 Typically, the 3D and 2D examinations are performed together (slightly more than doubling the radiation dose), or synthetic 2D images can be created from the 3D slices (resulting in a total radiation dose almost the same as standard 2D alone).
Most additional cancers seen on 3D mammography or US are lower-grade invasive cancers with good prognoses. Some aggressive high-grade breast cancers go undetected even when mammography is supplemented with US, either because they are too small to be seen or because they resemble common benign masses and may not be recognized. MRI is particularly effective in depicting high-grade cancers, even small ones.
The TABLE summarizes the relative rates of cancer detection and additional testing by various breast screening tests or combinations of tests. Neither clinical breast examination by a physician or other health care professional nor routine breast self-examination reduces the number of deaths caused by breast cancer. Nevertheless, women should monitor any changes in their breasts and report these changes to their clinician. A new lump, skin or nipple retraction, or a spontaneous clear or bloody nipple discharge merits diagnostic breast imaging even if a recent screening mammogram was normal.
FIGURE 4 is an updated decision support tool that suggests strategies for optimizingcancer detection with widely available screening methods.
Read how to take advantage of today’s technology for breast density screening
MY STORY: Epilogue
My annual 3D mammograms were normal, even the year my cancer was present. In 2014, I entered my family history into the IBIS Breast Cancer Risk Evaluation Tool (Tyrer-Cuzick model of breast cancer risk) (http://www.ems-trials.org/riskevaluator/) and calculated my lifetime risk at 19.7%. That is when I decided to have a screening MRI. My invasive breast cancer was easily seen on MRI and then on US. The cancer was node-negative, easily confirmed with needle biopsy, and treated with lumpectomy and radiation. There was no need for chemotherapy.
My personal experience prompted me to join JoAnn Pushkin and Cindy Henke-Sarmento, RT(R)(M), BA, in developing a website, www.DenseBreast-info.org, to give women and their physicians easy access to information on making decisions about screening in dense breasts.
My colleagues and I are often asked what is the best way to order supplemental imaging for a patient who may have dense breasts. Even in cases in which a mammogram does not exist or is unavailable, the following prescription can be implemented easily at centers that offer US: “2D plus 3D mammogram if available; if dense, perform ultrasound as needed.”
Related article:
DenseBreast-info.org: What this resource can offer you, and your patients
Breast density screening: Take advantage of today’s technology
Breast screening and diagnostic imaging have improved significantly since the 1970s, when many of the randomized trials of mammography were conducted. Breast density is one of the most common and important risk factors for development of breast cancer and is now incorporated into the Breast Cancer Surveillance Consortium model (https://tools.bcsc-scc.org/BC5yearRisk/calculator.htm) and the Tyrer-Cuzick model (see also http://densebreast-info.org/explanation-of-dense-breast-risk-models.aspx).32 Although we continue to validate newer approaches, women should take advantage of the improved methods of early cancer detection, particularly if they have dense breasts or are at high risk for breast cancer.
Share your thoughts! Send your Letter to the Editor to [email protected]. Please include your name and the city and state in which you practice.
MY STORY: Prologue
My aunt received a breast cancer diagnosis at age 40, and she died at age 60, in 1970. Then, in 1975, my mother’s breast cancer was found at age 55, but only after she was examined for nipple retraction; on mammography, the cancer had been obscured by dense breast tissue. Mom had 2 metastatic nodes but participated in the earliest clinical trials of chemotherapy and lived free of breast cancer for another 41 years. Naturally I thought that, were I to develop this disease, I would want it found earlier. Ironically, it was, but only because I had spent my career trying to understand the optimal screening approaches for women with dense breasts—women like me.
Cancers are masked on mammography in dense breasts
For women, screening mammography is an important step in reducing the risk of dying from breast cancer. The greatest benefits are realized by those who start annual screening at age 40, or 45 at the latest.1 As it takes 9 to 10 years to see a benefit from breast cancer screening at the population level, it is not logical to continue this testing when life expectancy is less than 10 years, as is the case with women age 85 or older, even those in the healthiest quartile.2–4 However, despite recent advances, the development of 3D mammography (tomosynthesis) (FIGURE 1) in particular, cancers can still be masked by dense breast tissue. Both 2D and 3D mammograms are x-rays; both dense tissue and cancers absorb x-rays and appear white.

Breast density is determined on mammography and is categorized as fatty, scattered fibroglandular, heterogeneously dense, or extremely dense (FIGURE 2).5 Tissue in the heterogeneous and extreme categories is considered dense. More than half of women in their 40s have dense breasts; with some fatty involution occurring around menopause, the proportion drops to 25% for women in their 60s.6 About half of breast cancers have calcifications, which on mammography are usually easily visible even in dense breasts. The problem is with noncalcified invasive cancers that can be hidden by dense tissue (FIGURE 3).


3D mammography improves cancer detection but is of minimal benefit in extremely dense breasts
Although 3D mammography improves cancer detection in most women, any benefit is minimal in women with extremely dense breasts, as there is no inherent soft-tissue contrast.7 Masked cancers are often only discovered because of a lump after a normal screening mammogram, as so-called “interval cancers.” Compared with screen-detected cancers, interval cancers tend to be more biologically aggressive, to have spread to lymph nodes, and to have worse prognoses. However, even some small screen-detected cancers are biologically aggressive and can spread to lymph nodes quickly, and no screening test or combination of screening tests can prevent this occurrence completely, regardless of breast density.
Related article:
Get smart about dense breasts

MRI provides early detection across all breast densities
In all tissue densities, contrast-enhanced magnetic resonance imaging (MRI) is far better than mammography in detecting breast cancer.8 Women at high risk for breast cancer caused by mutations in BRCA1, BRCA2, p53, and other genes have poor outcomes with screening mammography alone—up to 50% of cancers are interval cancers. Annual screening MRI reduces this percentage significantly, to 11% in women with pathogenic BRCA1 mutations and to 4% in women with BRCA2 mutations.9 Warner and colleagues found a decrease in late-stage cancers in high-risk women who underwent annual MRI screenings compared to high-risk women unable to have MRI.10
The use of MRI for screening is limited by availability, patient tolerance,11 and high cost. Research is being conducted to further validate approaches using shortened screening MRI times (so-called “abbreviated” or “fast” MRI) and, thereby, improve access, tolerance, and reduce associated costs; several investigators already have reported promising results, and a few centers offer this modality directly to patients willing to pay $300 to $350 out of pocket.12,13 Even in normal-risk women, MRI significantly increases detection of early breast cancer after a normal mammogram and ultrasound, and the cancer detection benefit of MRI is seen across all breast densities.14
Most health insurance plans cover screening MRI only for women who meet defined risk criteria, including women who have a known disease-causing mutation—or are suspected of having one, given a family history of breast cancer with higher than 20% to 25% lifetime risk by a model that predicts mutation carrier status—as well as women who had chest radiation therapy before age 30, typically for Hodgkin lymphoma, and at least 8 years earlier.15 In addition, MRI can be considered in women with atypical breast biopsy results or a personal history of lobular carcinoma in situ (LCIS).16
Screening MRI should start by age 25 in women with disease-causing mutations, or at the time of atypical or LCIS biopsy results, and should be performed annually unless the woman is pregnant or has a metallic implant, renal insufficiency, or another contraindication to MRI. MRI can be beneficial in women with a personal history of cancer, although annual mammography remains the standard of care.17–19
MRI and mammography can be performed at the same time or on an alternating 6-month basis, with mammography usually starting only after age 30 because of the small risk that radiation poses for younger women. There are a few other impediments to having breast MRI: The woman must lie on her stomach within a confined space (tunnel), the contrast that is injected may not be well tolerated, and insurance does not cover the test for women who do not meet the defined risk criteria.11
Read why mammography supplemented by US is best for women with dense breasts.
Ultrasonography supplements mammography
Mammography supplemented with ultrasonography (US) has been studied as a “Goldilocks” or best-fit solution for the screening of women with dense breasts, as detection of invasive cancers is improved with the 2 modalities over mammography alone, and US is less invasive, better tolerated, and lower in cost than the more sensitive MRI.
In women with dense breasts, US has been found to improve cancer detection over mammography alone, and early results suggest a larger cancer detection benefit from US than from 3D mammography, although research is ongoing.20 Adding US reduces the interval cancer rate in women with dense breasts to less than 10% of all cancers found—similar to results for women with fatty breasts.17,21,22
US can be performed by a trained technologist or a physician using a small transducer, which usually provides diagnostic images (so that most callbacks would be for a true finding), or a larger transducer and an automated system can be used to create more than a thousand images for radiologist review.23,24 Use of a hybrid system, a small transducer with an automated arm, has been validated as well.25 Screening US is not available universally, and with all these approaches optimal performance requires trained personnel. Supplemental screening US usually is covered by insurance but is nearly always subject to a deductible/copay.
Related article:
Educate patients about dense breasts and cancer risk
Reducing false-positives, callbacks, and additional testing
Mammography carries a risk of false-positives. On average, 11% to 12% of women are called back for additional testing after a screening mammogram, and in more than 95% of women brought back for extra testing, no cancer is found.26 Women with dense breasts are more likely than those with less dense breasts to be called back.27 US and MRI improve cancer detection and therefore yield additional positive, but also false-positive, findings. Notably, callbacks decrease after the first round of screening with any modality or combination of tests, as long as prior examinations are available for comparison.
One advantage of 3D over 2D mammography is a decrease in extra testing for areas of asymmetry, which are often recognizable on 3D mammography as representing normal superimposed tissue.28–30 Architectural distortion, which is better seen on 3D mammography and usually represents either cancer or a benign radial scar, can lead to false-positive biopsies, although the average biopsy rate is no higher for 3D than for 2D alone.31 Typically, the 3D and 2D examinations are performed together (slightly more than doubling the radiation dose), or synthetic 2D images can be created from the 3D slices (resulting in a total radiation dose almost the same as standard 2D alone).
Most additional cancers seen on 3D mammography or US are lower-grade invasive cancers with good prognoses. Some aggressive high-grade breast cancers go undetected even when mammography is supplemented with US, either because they are too small to be seen or because they resemble common benign masses and may not be recognized. MRI is particularly effective in depicting high-grade cancers, even small ones.
The TABLE summarizes the relative rates of cancer detection and additional testing by various breast screening tests or combinations of tests. Neither clinical breast examination by a physician or other health care professional nor routine breast self-examination reduces the number of deaths caused by breast cancer. Nevertheless, women should monitor any changes in their breasts and report these changes to their clinician. A new lump, skin or nipple retraction, or a spontaneous clear or bloody nipple discharge merits diagnostic breast imaging even if a recent screening mammogram was normal.
FIGURE 4 is an updated decision support tool that suggests strategies for optimizingcancer detection with widely available screening methods.
Read how to take advantage of today’s technology for breast density screening
MY STORY: Epilogue
My annual 3D mammograms were normal, even the year my cancer was present. In 2014, I entered my family history into the IBIS Breast Cancer Risk Evaluation Tool (Tyrer-Cuzick model of breast cancer risk) (http://www.ems-trials.org/riskevaluator/) and calculated my lifetime risk at 19.7%. That is when I decided to have a screening MRI. My invasive breast cancer was easily seen on MRI and then on US. The cancer was node-negative, easily confirmed with needle biopsy, and treated with lumpectomy and radiation. There was no need for chemotherapy.
My personal experience prompted me to join JoAnn Pushkin and Cindy Henke-Sarmento, RT(R)(M), BA, in developing a website, www.DenseBreast-info.org, to give women and their physicians easy access to information on making decisions about screening in dense breasts.
My colleagues and I are often asked what is the best way to order supplemental imaging for a patient who may have dense breasts. Even in cases in which a mammogram does not exist or is unavailable, the following prescription can be implemented easily at centers that offer US: “2D plus 3D mammogram if available; if dense, perform ultrasound as needed.”
Related article:
DenseBreast-info.org: What this resource can offer you, and your patients
Breast density screening: Take advantage of today’s technology
Breast screening and diagnostic imaging have improved significantly since the 1970s, when many of the randomized trials of mammography were conducted. Breast density is one of the most common and important risk factors for development of breast cancer and is now incorporated into the Breast Cancer Surveillance Consortium model (https://tools.bcsc-scc.org/BC5yearRisk/calculator.htm) and the Tyrer-Cuzick model (see also http://densebreast-info.org/explanation-of-dense-breast-risk-models.aspx).32 Although we continue to validate newer approaches, women should take advantage of the improved methods of early cancer detection, particularly if they have dense breasts or are at high risk for breast cancer.
Share your thoughts! Send your Letter to the Editor to [email protected]. Please include your name and the city and state in which you practice.
- Oeffinger KC, Fontham ET, Etzioni R, et al; American Cancer Society. Breast cancer screening for women at average risk: 2015 guideline update from the American Cancer Society. JAMA. 2015;314(15):1599–1614.
- Tabar L, Yen MF, Vitak B, Chen HH, Smith RA, Duffy SW. Mammography service screening and mortality in breast cancer patients: 20-year follow-up before and after introduction of screening. Lancet. 2003;361(9367):1405–1410.
- Lee SJ, Boscardin WJ, Stijacic-Cenzer I, Conell-Price J, O’Brien S, Walter LC. Time lag to benefit after screening for breast and colorectal cancer: meta-analysis of survival data from the United States, Sweden, United Kingdom, and Denmark. BMJ. 2013;346:e8441.
- Walter LC, Covinsky KE. Cancer screening in elderly patients: a framework for individualized decision making. JAMA. 2001;285(21):2750–2756.
- Sickles EA, D’Orsi CJ, Bassett LW, et al. ACR BI-RADS mammography. In: D’Orsi CJ, Sickles EA, Mendelson EB, et al, eds. ACR BI-RADS Atlas, Breast Imaging Reporting and Data System. 5th ed. Reston, VA: American College of Radiology; 2013.
- Sprague BL, Gangnon RE, Burt V, et al. Prevalence of mammographically dense breasts in the United States. J Natl Cancer Inst. 2014;106(10).
- Rafferty EA, Durand MA, Conant EF, et al. Breast cancer screening using tomosynthesis and digital mammography in dense and nondense breasts. JAMA. 2016;315(16):1784–1786.
- Berg WA. Tailored supplemental screening for breast cancer: what now and what next? AJR Am J Roentgenol. 2009;192(2):390–399.
- Heijnsdijk EA, Warner E, Gilbert FJ, et al. Differences in natural history between breast cancers in BRCA1 and BRCA2 mutation carriers and effects of MRI screening—MRISC, MARIBS, and Canadian studies combined. Cancer Epidemiol Biomarkers Prev. 2012;21(9):1458–1468.
- Warner E, Hill K, Causer P, et al. Prospective study of breast cancer incidence in women with a BRCA1 or BRCA2 mutation under surveillance with and without magnetic resonance imaging. J Clin Oncol. 2011;29(13):1664–1669.
- Berg WA, Blume JD, Adams AM, et al. Reasons women at elevated risk of breast cancer refuse breast MR imaging screening: ACRIN 6666. Radiology. 2010;254(1):79–87.
- Kuhl CK, Schrading S, Strobel K, Schild HH, Hilgers RD, Bieling HB. Abbreviated breast magnetic resonance imaging (MRI): first postcontrast subtracted images and maximum-intensity projection—a novel approach to breast cancer screening with MRI. J Clin Oncol. 2014;32(22):2304–2310.
- Strahle DA, Pathak DR, Sierra A, Saha S, Strahle C, Devisetty K. Systematic development of an abbreviated protocol for screening breast magnetic resonance imaging. Breast Cancer Res Treat. 2017;162(2):283–295.
- Kuhl CK, Strobel K, Bieling H, Leutner C, Schild HH, Schrading S. Supplemental breast MR imaging screening of women with average risk of breast cancer. Radiology. 2017;283(2):361–370.
- Saslow D, Boetes C, Burke W, et al; American Cancer Society Breast Cancer Advisory Group. American Cancer Society guidelines for breast screening with MRI as an adjunct to mammography. CA Cancer J Clin. 2007;57(2):75–89.
- National Comprehensive Cancer Network. NCCN guidelines for detection, prevention, and risk reduction: breast cancer screening and diagnosis. https://www.nccn.org/professionals/physician_gls/pdf/breast-screening.pdf.
- Berg WA, Zhang Z, Lehrer D, et al; ACRIN 6666 Investigators. Detection of breast cancer with addition of annual screening ultrasound or a single screening MRI to mammography in women with elevated breast cancer risk. JAMA. 2012;307(13):1394–1404.
- Brennan S, Liberman L, Dershaw DD, Morris E. Breast MRI screening of women with a personal history of breast cancer. AJR Am J Roentgenol. 2010;195(2):510–516.
- Lehman CD, Lee JM, DeMartini WB, et al. Screening MRI in women with a personal history of breast cancer. J Natl Cancer Inst. 2016;108(3).
- Tagliafico AS, Calabrese M, Mariscotti G, et al. Adjunct screening with tomosynthesis or ultrasound in women with mammography-negative dense breasts: interim report of a prospective comparative trial [published online ahead of print March 9, 2016]. J Clin Oncol. JCO634147.
- Corsetti V, Houssami N, Ghirardi M, et al. Evidence of the effect of adjunct ultrasound screening in women with mammography-negative dense breasts: interval breast cancers at 1 year follow-up. Eur J Cancer. 2011;47(7):1021–1026.
- Ohuchi N, Suzuki A, Sobue T, et al; J-START Investigator Groups. Sensitivity and specificity of mammography and adjunctive ultrasonography to screen for breast cancer in the Japan Strategic Anti-Cancer Randomized Trial (J-START): a randomised controlled trial. Lancet. 2016;387(10016):341–348.
- Berg WA, Mendelson EB. Technologist-performed handheld screening breast US imaging: how is it performed and what are the outcomes to date? Radiology. 2014;272(1):12–27.
- Brem RF, Tabár L, Duffy SW, et al. Assessing improvement in detection of breast cancer with three-dimensional automated breast US in women with dense breast tissue: the SomoInsight study. Radiology. 2015;274(3):663–673.
- Kelly KM, Dean J, Comulada WS, Lee SJ. Breast cancer detection using automated whole breast ultrasound and mammography in radiographically dense breasts. Eur Radiol. 2010;20(3):734–742.
- Lehman CD, Arao RF, Sprague BL, et al. National performance benchmarks for modern screening digital mammography: update from the Breast Cancer Surveillance Consortium. Radiology. 2017;283(1):49–58.
- Kerlikowske K, Zhu W, Hubbard RA, et al; Breast Cancer Surveillance Consortium. Outcomes of screening mammography by frequency, breast density, and postmenopausal hormone therapy. JAMA Intern Med. 2013;173(9):807–816.
- Friedewald SM, Rafferty EA, Rose SL, et al. Breast cancer screening using tomosynthesis in combination with digital mammography. JAMA. 2014;311(24):2499–2507.
- Skaane P, Bandos AI, Gullien R, et al. Comparison of digital mammography alone and digital mammography plus tomosynthesis in a population-based screening program. Radiology. 2013;267(1):47–56.
- Ciatto S, Houssami N, Bernardi D, et al. Integration of 3D digital mammography with tomosynthesis for population breast-cancer screening (STORM): a prospective comparison study. Lancet Oncol. 2013;14(7):583–589.
- Bahl M, Lamb LR, Lehman CD. Pathologic outcomes of architectural distortion on digital 2D versus tomosynthesis mammography [published online ahead of print August 23, 2017]. AJR Am J Roentgenol. doi:10.2214/AJR.17.17979.
- Engmann NJ, Golmakani MK, Miglioretti DL, Sprague BL, Kerlikowske K; Breast Cancer Surveillance Consortium. Population-attributable risk proportion of clinical risk factors for breast cancer [published online ahead of print February 2, 2017]. JAMA Oncol. doi:10.1001/jamaoncol.2016.6326.
- Oeffinger KC, Fontham ET, Etzioni R, et al; American Cancer Society. Breast cancer screening for women at average risk: 2015 guideline update from the American Cancer Society. JAMA. 2015;314(15):1599–1614.
- Tabar L, Yen MF, Vitak B, Chen HH, Smith RA, Duffy SW. Mammography service screening and mortality in breast cancer patients: 20-year follow-up before and after introduction of screening. Lancet. 2003;361(9367):1405–1410.
- Lee SJ, Boscardin WJ, Stijacic-Cenzer I, Conell-Price J, O’Brien S, Walter LC. Time lag to benefit after screening for breast and colorectal cancer: meta-analysis of survival data from the United States, Sweden, United Kingdom, and Denmark. BMJ. 2013;346:e8441.
- Walter LC, Covinsky KE. Cancer screening in elderly patients: a framework for individualized decision making. JAMA. 2001;285(21):2750–2756.
- Sickles EA, D’Orsi CJ, Bassett LW, et al. ACR BI-RADS mammography. In: D’Orsi CJ, Sickles EA, Mendelson EB, et al, eds. ACR BI-RADS Atlas, Breast Imaging Reporting and Data System. 5th ed. Reston, VA: American College of Radiology; 2013.
- Sprague BL, Gangnon RE, Burt V, et al. Prevalence of mammographically dense breasts in the United States. J Natl Cancer Inst. 2014;106(10).
- Rafferty EA, Durand MA, Conant EF, et al. Breast cancer screening using tomosynthesis and digital mammography in dense and nondense breasts. JAMA. 2016;315(16):1784–1786.
- Berg WA. Tailored supplemental screening for breast cancer: what now and what next? AJR Am J Roentgenol. 2009;192(2):390–399.
- Heijnsdijk EA, Warner E, Gilbert FJ, et al. Differences in natural history between breast cancers in BRCA1 and BRCA2 mutation carriers and effects of MRI screening—MRISC, MARIBS, and Canadian studies combined. Cancer Epidemiol Biomarkers Prev. 2012;21(9):1458–1468.
- Warner E, Hill K, Causer P, et al. Prospective study of breast cancer incidence in women with a BRCA1 or BRCA2 mutation under surveillance with and without magnetic resonance imaging. J Clin Oncol. 2011;29(13):1664–1669.
- Berg WA, Blume JD, Adams AM, et al. Reasons women at elevated risk of breast cancer refuse breast MR imaging screening: ACRIN 6666. Radiology. 2010;254(1):79–87.
- Kuhl CK, Schrading S, Strobel K, Schild HH, Hilgers RD, Bieling HB. Abbreviated breast magnetic resonance imaging (MRI): first postcontrast subtracted images and maximum-intensity projection—a novel approach to breast cancer screening with MRI. J Clin Oncol. 2014;32(22):2304–2310.
- Strahle DA, Pathak DR, Sierra A, Saha S, Strahle C, Devisetty K. Systematic development of an abbreviated protocol for screening breast magnetic resonance imaging. Breast Cancer Res Treat. 2017;162(2):283–295.
- Kuhl CK, Strobel K, Bieling H, Leutner C, Schild HH, Schrading S. Supplemental breast MR imaging screening of women with average risk of breast cancer. Radiology. 2017;283(2):361–370.
- Saslow D, Boetes C, Burke W, et al; American Cancer Society Breast Cancer Advisory Group. American Cancer Society guidelines for breast screening with MRI as an adjunct to mammography. CA Cancer J Clin. 2007;57(2):75–89.
- National Comprehensive Cancer Network. NCCN guidelines for detection, prevention, and risk reduction: breast cancer screening and diagnosis. https://www.nccn.org/professionals/physician_gls/pdf/breast-screening.pdf.
- Berg WA, Zhang Z, Lehrer D, et al; ACRIN 6666 Investigators. Detection of breast cancer with addition of annual screening ultrasound or a single screening MRI to mammography in women with elevated breast cancer risk. JAMA. 2012;307(13):1394–1404.
- Brennan S, Liberman L, Dershaw DD, Morris E. Breast MRI screening of women with a personal history of breast cancer. AJR Am J Roentgenol. 2010;195(2):510–516.
- Lehman CD, Lee JM, DeMartini WB, et al. Screening MRI in women with a personal history of breast cancer. J Natl Cancer Inst. 2016;108(3).
- Tagliafico AS, Calabrese M, Mariscotti G, et al. Adjunct screening with tomosynthesis or ultrasound in women with mammography-negative dense breasts: interim report of a prospective comparative trial [published online ahead of print March 9, 2016]. J Clin Oncol. JCO634147.
- Corsetti V, Houssami N, Ghirardi M, et al. Evidence of the effect of adjunct ultrasound screening in women with mammography-negative dense breasts: interval breast cancers at 1 year follow-up. Eur J Cancer. 2011;47(7):1021–1026.
- Ohuchi N, Suzuki A, Sobue T, et al; J-START Investigator Groups. Sensitivity and specificity of mammography and adjunctive ultrasonography to screen for breast cancer in the Japan Strategic Anti-Cancer Randomized Trial (J-START): a randomised controlled trial. Lancet. 2016;387(10016):341–348.
- Berg WA, Mendelson EB. Technologist-performed handheld screening breast US imaging: how is it performed and what are the outcomes to date? Radiology. 2014;272(1):12–27.
- Brem RF, Tabár L, Duffy SW, et al. Assessing improvement in detection of breast cancer with three-dimensional automated breast US in women with dense breast tissue: the SomoInsight study. Radiology. 2015;274(3):663–673.
- Kelly KM, Dean J, Comulada WS, Lee SJ. Breast cancer detection using automated whole breast ultrasound and mammography in radiographically dense breasts. Eur Radiol. 2010;20(3):734–742.
- Lehman CD, Arao RF, Sprague BL, et al. National performance benchmarks for modern screening digital mammography: update from the Breast Cancer Surveillance Consortium. Radiology. 2017;283(1):49–58.
- Kerlikowske K, Zhu W, Hubbard RA, et al; Breast Cancer Surveillance Consortium. Outcomes of screening mammography by frequency, breast density, and postmenopausal hormone therapy. JAMA Intern Med. 2013;173(9):807–816.
- Friedewald SM, Rafferty EA, Rose SL, et al. Breast cancer screening using tomosynthesis in combination with digital mammography. JAMA. 2014;311(24):2499–2507.
- Skaane P, Bandos AI, Gullien R, et al. Comparison of digital mammography alone and digital mammography plus tomosynthesis in a population-based screening program. Radiology. 2013;267(1):47–56.
- Ciatto S, Houssami N, Bernardi D, et al. Integration of 3D digital mammography with tomosynthesis for population breast-cancer screening (STORM): a prospective comparison study. Lancet Oncol. 2013;14(7):583–589.
- Bahl M, Lamb LR, Lehman CD. Pathologic outcomes of architectural distortion on digital 2D versus tomosynthesis mammography [published online ahead of print August 23, 2017]. AJR Am J Roentgenol. doi:10.2214/AJR.17.17979.
- Engmann NJ, Golmakani MK, Miglioretti DL, Sprague BL, Kerlikowske K; Breast Cancer Surveillance Consortium. Population-attributable risk proportion of clinical risk factors for breast cancer [published online ahead of print February 2, 2017]. JAMA Oncol. doi:10.1001/jamaoncol.2016.6326.