User login
Table Salt Method Following Cryotherapy for Recurrent Pyogenic Granuloma on the Fingertip
Table Salt Method Following Cryotherapy for Recurrent Pyogenic Granuloma on the Fingertip
Practice Gap
Pyogenic granulomas (PGs) are benign endothelial tumors of the skin or mucosae that frequently become ulcerated and may cause patients substantial discomfort or distress due to rapid enlargement and bleeding.1 These lesions often manifest as solitary red papules or polyps following localized trauma or irritation. They can grow up to 1 cm over a few weeks to several months. Pyogenic granulomas can develop at any age, but they most commonly are seen in children and young adults, with a slight male predominance.1,2 The differential diagnosis for PG includes amelanotic melanoma, bacillary angiomatosis, Kaposi sarcoma, glomus tumor, infantile hemangioma, and irritated melanocytic nevus.1 Histologically, PGs are well-circumscribed exophytic or pedunculated proliferations of small capillaries that often are arranged in a lobular pattern. Early lesions show packed endothelial cells, while advanced lesions display more ectatic vessels, erosion, and crusting.3 The term pyogenic granuloma is a misnomer, as these lesions display neither an infectious etiology nor granulomatous tissue on dermatopathologic examination.4 A more accurate clinical description for this lesion is a lobular capillary hemangioma.
Numerous surgical and laser techniques have been used to treat PGs, with varying degrees of success. Treatment often consists of either shave excision followed by electrosurgery at the base or full excision with suturing under local anesthesia for patients who can tolerate anesthetic injections.1 Pulsed dye laser has been proven to be a safe alternative treatment option, particularly in children who otherwise would not tolerate surgical procedures.5 Topical beta-blockers, silver nitrate cauterization, sclerotherapy, and liquid nitrogen all have been used as alternative treatment methods.1
Pyogenic granulomas often recur after first-line treatments, and patients may hesitate to try more invasive techniques when the first choice has failed. Children may not be amenable to any of these curative techniques, as they may not tolerate the pain associated with lidocaine injection and/or have a fear of needles or surgical intervention; even adults may be reluctant to have a procedure they perceive as painful. We present a less invasive technique for treatment of recurrent PGs using table salt and cryotherapy.
The Technique
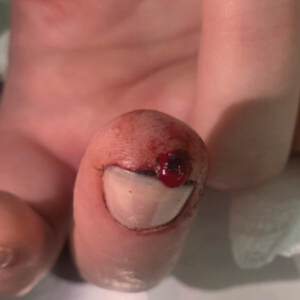
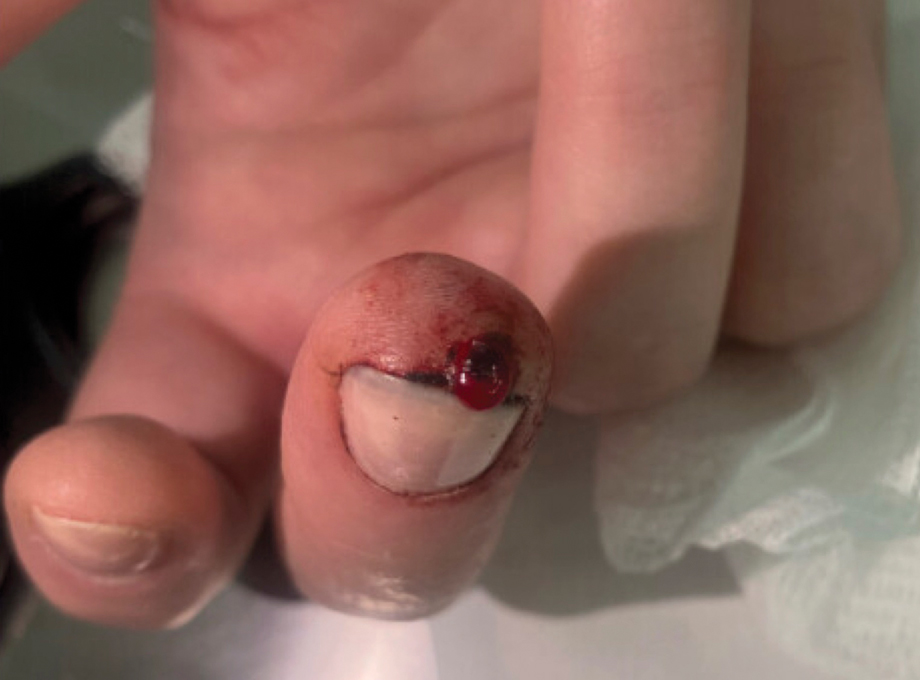
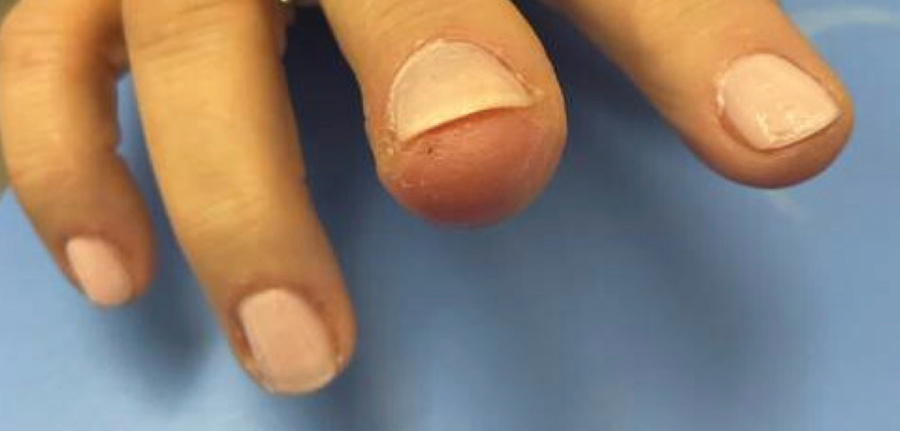
A 51-year-old woman with no notable medical history presented to the emergency department for evaluation of a black dot on the pulp of the right third fingertip of 1 week’s duration. The patient reported rapid progression to an ulcerated red nodule with associated bleeding for the past 3 days (Figure 1). Direct pressure temporarily alleviated the bleeding, but it started again upon cessation of pressure. She denied any preceding trauma to the area or any associated systemic symptoms such as fever, chills, nausea, or vomiting.
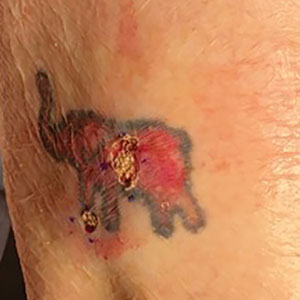
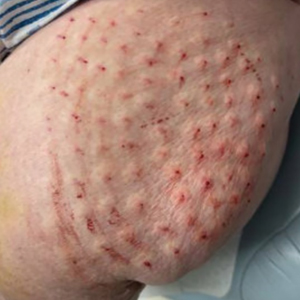
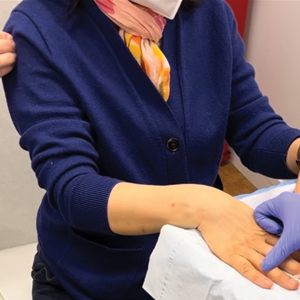
The inpatient dermatology team recommended that the patient be discharged following silver nitrate cautery, with a referral sent to outpatient dermatology; however, the patient returned to the dermatology clinic 4 days later, at which time physical examination revealed a well-circumscribed, 5-mm, bright-red, erosive papule with overlying hemorrhagic crust that was not actively bleeding, as well as fissuring of the surrounding skin. The entire lesion was removed using tangential excision followed by electrodesiccation at the base. Pathology revealed small capillaries arranged in a lobular pattern, confirming the diagnosis of PG. At a 2-week follow-up visit, the patient noted that the lesion had recurred within 24 hours after the procedure and was larger (Figure 2). At this visit, management was switched to a single treatment of cryotherapy (3 cycles for 5 seconds per cycle), and the table salt method was recommended based on a literature review for alternative nonpainful approaches for PG.6-11 We used this technique in our patient as an adjuvant to cryotherapy with the goal of reducing the need for additional painful procedures, but table salt also can be used as a standalone treatment without prior cryotherapy.
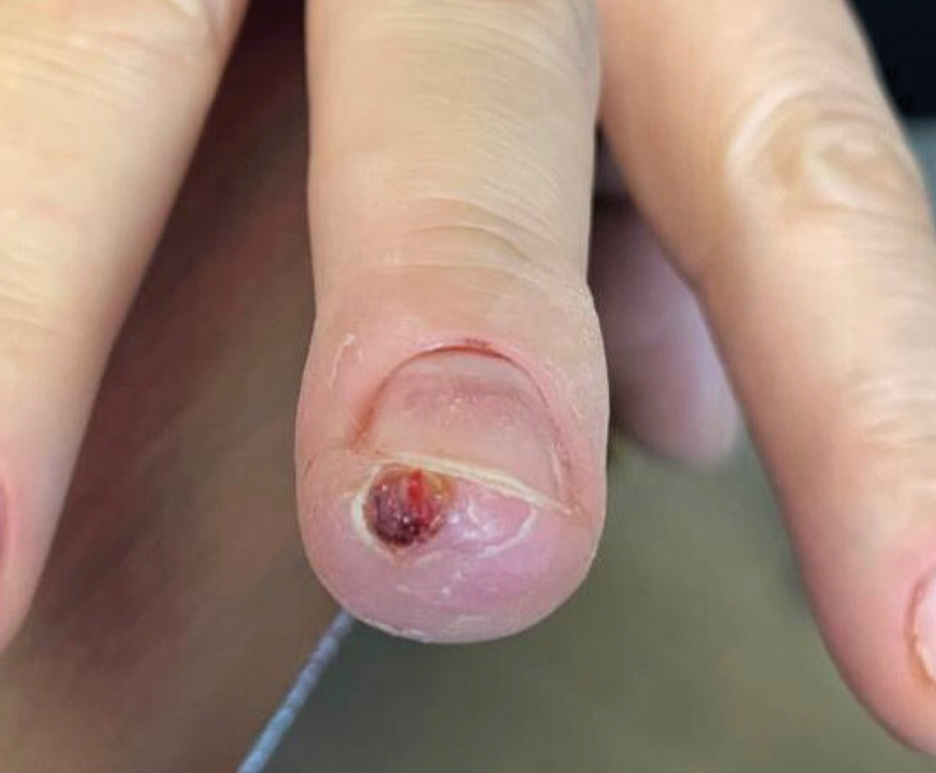
The patient was instructed to apply table salt to the lesion once daily for 2 weeks by pressing the lesion into a small amount of salt placed on a clean plate and then applying an occlusive dressing such as surgical or paper tape. She also was advised to apply petroleum jelly around the periphery of the lesion prior to salt application to protect the unaffected skin from irritation. Complete resolution of the lesion was seen when the patient followed up 2 weeks later (Figure 3). At the most recent follow-up 2 months after treatment, no further recurrence of the PG was reported.
Practice Implication
Pyogenic granulomas can be distressing for both patients and providers because they are cosmetically bothersome and prone to spontaneous bleeding. Various medical and surgical options exist to treat PGs, but there is no clear consensus on a superior modality. A 2019 study by Daruwalla and Dhurat6 highlighted a less invasive treatment option for PGs using table salt applied once daily for 2 weeks under an occlusive dressing with good outcomes and without involving other treatments such as cryotherapy. Several other case reports have endorsed this approach, adding anecdotal evidence for its utility.7-11 Topical sodium chloride may treat PGs primarily through osmotic desiccation, drawing water out of the lesion and leading to endothelial cell shrinkage and collapse of its capillary network. This hyperosmolar environment also may induce microvascular thrombosis and ischemia, promoting lesion necrosis. Additionally, repeated application creates a dry, mildly irritative surface that may suppress angiogenesis and encourage regression of the vascular proliferation.
Consider topical application of table salt for treatment of PGs in certain subsets of patients; for example, patients who are not amenable to surgery or are too young for advanced surgical techniques may be good candidates for this method, as it does not require anesthetic injections and generally is pain free. Patients with resistant or recurrent PGs that did not respond to first-line treatments such as cryotherapy, tangential excision, or electrodesiccation may be more amenable to a less invasive secondary approach.
Importantly, we used a dual-therapy approach in our patient, initially using a single application of cryotherapy followed by the table salt method once daily for 2 weeks. This imposes limitations on the generalizability of table salt as a standalone approach for treating PGs. In this case, we did not have prior practical experience using table salt for this condition and only had small reports to justify its use. As a result, we attempted a more traditional treatment initially (cryotherapy) to avoid potential delays in resolution. The clinicians recommended table salt as an adjuvant prior to seeing the cryotherapy results because this treatment was benign and offered potential additive results, and therefore waiting was not necessary. However, various other cases have reported similar success using table salt as monotherapy.6-9,11 Patients should be advised of potential mild adverse events, such as irritation to the surrounding skin. Higher-level evidence studies are required to further vet the utility of the table salt method for treatment of PGs.
- Bolognia JL, Schaffer JV, Cerroni L. Vascular neoplasms and neoplastic‑like proliferations. In: Dermatology. Elsevier; 2018.
- Harris MN, Desai R, Chuang TY, et al. Lobular capillary hemangiomas: an epidemiologic report, with emphasis on cutaneous lesions. J Am Acad Dermatol. 2000;42:1012-1016.
- Ferringer TK, DiCaudo DJ, Elston D, et al. Dermatopathology. W.B. Saunders; 2008.
- Gomes SR, Shakir QJ, Thaker PV, et al. Pyogenic granuloma of the gingiva: a misnomer? - a case report and review of literature. J Indian Soc Periodontol. 2013;17:514-519. doi:10.4103/0972-124X.118327
- Sud AR, Tan ST. Pyogenic granuloma-treatment by shave-excision and/or pulsed-dye laser. J Plast Reconstr Aesthet Surg. 2010;63:1364-1368. doi:10.1016/j.bjps.2009.06.031
- Daruwalla SB, Dhurat RS. A pinch of salt is all it takes! the novel use of table salt for the effective treatment of pyogenic granuloma. J Am Acad Dermatol. 2020;83:E107-E108. doi:10.1016/j.jaad.2019.12.013
- Alhammad G, Albaraka M, Alotaibi H, et al. The use of common salt for the treatment of pyogenic granuloma. JAAD Case Rep. 2024;53:40-42. doi:10.1016/j.jdcr.2024.08.016
- Weiss ES, Wood D. Simple, safe, and effective treatment for pyogenic granuloma. Can Fam Physician. 2023;69:479-480. doi:10.46747/cfp.6907479
- Bernales Salinas A, Toro Sepúlveda A, Meier Pincheira H, et al. Case report: pyogenic granuloma-just salt, a simple and pain-free treatment. Dermatol Ther. 2022;35:E15194. doi:10.1111/dth.15194
- Martín-Nieto González J, Rodríguez-Sánchez B, Berna-Rico E, et al. Pyogenic granuloma resolved with timolol and table salt. An Pediatr (Engl Ed). 2025;102:503706. doi:10.1016/j.anpede.2025.503706
- Bin Rubaian NF. Complete resolution of a refractory pyogenic granuloma following topical salt treatment. Open Access Emerg Med. 2021;13:445-448. doi:10.2147/OAEM.S323793
Practice Gap
Pyogenic granulomas (PGs) are benign endothelial tumors of the skin or mucosae that frequently become ulcerated and may cause patients substantial discomfort or distress due to rapid enlargement and bleeding.1 These lesions often manifest as solitary red papules or polyps following localized trauma or irritation. They can grow up to 1 cm over a few weeks to several months. Pyogenic granulomas can develop at any age, but they most commonly are seen in children and young adults, with a slight male predominance.1,2 The differential diagnosis for PG includes amelanotic melanoma, bacillary angiomatosis, Kaposi sarcoma, glomus tumor, infantile hemangioma, and irritated melanocytic nevus.1 Histologically, PGs are well-circumscribed exophytic or pedunculated proliferations of small capillaries that often are arranged in a lobular pattern. Early lesions show packed endothelial cells, while advanced lesions display more ectatic vessels, erosion, and crusting.3 The term pyogenic granuloma is a misnomer, as these lesions display neither an infectious etiology nor granulomatous tissue on dermatopathologic examination.4 A more accurate clinical description for this lesion is a lobular capillary hemangioma.
Numerous surgical and laser techniques have been used to treat PGs, with varying degrees of success. Treatment often consists of either shave excision followed by electrosurgery at the base or full excision with suturing under local anesthesia for patients who can tolerate anesthetic injections.1 Pulsed dye laser has been proven to be a safe alternative treatment option, particularly in children who otherwise would not tolerate surgical procedures.5 Topical beta-blockers, silver nitrate cauterization, sclerotherapy, and liquid nitrogen all have been used as alternative treatment methods.1
Pyogenic granulomas often recur after first-line treatments, and patients may hesitate to try more invasive techniques when the first choice has failed. Children may not be amenable to any of these curative techniques, as they may not tolerate the pain associated with lidocaine injection and/or have a fear of needles or surgical intervention; even adults may be reluctant to have a procedure they perceive as painful. We present a less invasive technique for treatment of recurrent PGs using table salt and cryotherapy.
The Technique
A 51-year-old woman with no notable medical history presented to the emergency department for evaluation of a black dot on the pulp of the right third fingertip of 1 week’s duration. The patient reported rapid progression to an ulcerated red nodule with associated bleeding for the past 3 days (Figure 1). Direct pressure temporarily alleviated the bleeding, but it started again upon cessation of pressure. She denied any preceding trauma to the area or any associated systemic symptoms such as fever, chills, nausea, or vomiting.

The inpatient dermatology team recommended that the patient be discharged following silver nitrate cautery, with a referral sent to outpatient dermatology; however, the patient returned to the dermatology clinic 4 days later, at which time physical examination revealed a well-circumscribed, 5-mm, bright-red, erosive papule with overlying hemorrhagic crust that was not actively bleeding, as well as fissuring of the surrounding skin. The entire lesion was removed using tangential excision followed by electrodesiccation at the base. Pathology revealed small capillaries arranged in a lobular pattern, confirming the diagnosis of PG. At a 2-week follow-up visit, the patient noted that the lesion had recurred within 24 hours after the procedure and was larger (Figure 2). At this visit, management was switched to a single treatment of cryotherapy (3 cycles for 5 seconds per cycle), and the table salt method was recommended based on a literature review for alternative nonpainful approaches for PG.6-11 We used this technique in our patient as an adjuvant to cryotherapy with the goal of reducing the need for additional painful procedures, but table salt also can be used as a standalone treatment without prior cryotherapy.

The patient was instructed to apply table salt to the lesion once daily for 2 weeks by pressing the lesion into a small amount of salt placed on a clean plate and then applying an occlusive dressing such as surgical or paper tape. She also was advised to apply petroleum jelly around the periphery of the lesion prior to salt application to protect the unaffected skin from irritation. Complete resolution of the lesion was seen when the patient followed up 2 weeks later (Figure 3). At the most recent follow-up 2 months after treatment, no further recurrence of the PG was reported.

Practice Implication
Pyogenic granulomas can be distressing for both patients and providers because they are cosmetically bothersome and prone to spontaneous bleeding. Various medical and surgical options exist to treat PGs, but there is no clear consensus on a superior modality. A 2019 study by Daruwalla and Dhurat6 highlighted a less invasive treatment option for PGs using table salt applied once daily for 2 weeks under an occlusive dressing with good outcomes and without involving other treatments such as cryotherapy. Several other case reports have endorsed this approach, adding anecdotal evidence for its utility.7-11 Topical sodium chloride may treat PGs primarily through osmotic desiccation, drawing water out of the lesion and leading to endothelial cell shrinkage and collapse of its capillary network. This hyperosmolar environment also may induce microvascular thrombosis and ischemia, promoting lesion necrosis. Additionally, repeated application creates a dry, mildly irritative surface that may suppress angiogenesis and encourage regression of the vascular proliferation.
Consider topical application of table salt for treatment of PGs in certain subsets of patients; for example, patients who are not amenable to surgery or are too young for advanced surgical techniques may be good candidates for this method, as it does not require anesthetic injections and generally is pain free. Patients with resistant or recurrent PGs that did not respond to first-line treatments such as cryotherapy, tangential excision, or electrodesiccation may be more amenable to a less invasive secondary approach.
Importantly, we used a dual-therapy approach in our patient, initially using a single application of cryotherapy followed by the table salt method once daily for 2 weeks. This imposes limitations on the generalizability of table salt as a standalone approach for treating PGs. In this case, we did not have prior practical experience using table salt for this condition and only had small reports to justify its use. As a result, we attempted a more traditional treatment initially (cryotherapy) to avoid potential delays in resolution. The clinicians recommended table salt as an adjuvant prior to seeing the cryotherapy results because this treatment was benign and offered potential additive results, and therefore waiting was not necessary. However, various other cases have reported similar success using table salt as monotherapy.6-9,11 Patients should be advised of potential mild adverse events, such as irritation to the surrounding skin. Higher-level evidence studies are required to further vet the utility of the table salt method for treatment of PGs.
Practice Gap
Pyogenic granulomas (PGs) are benign endothelial tumors of the skin or mucosae that frequently become ulcerated and may cause patients substantial discomfort or distress due to rapid enlargement and bleeding.1 These lesions often manifest as solitary red papules or polyps following localized trauma or irritation. They can grow up to 1 cm over a few weeks to several months. Pyogenic granulomas can develop at any age, but they most commonly are seen in children and young adults, with a slight male predominance.1,2 The differential diagnosis for PG includes amelanotic melanoma, bacillary angiomatosis, Kaposi sarcoma, glomus tumor, infantile hemangioma, and irritated melanocytic nevus.1 Histologically, PGs are well-circumscribed exophytic or pedunculated proliferations of small capillaries that often are arranged in a lobular pattern. Early lesions show packed endothelial cells, while advanced lesions display more ectatic vessels, erosion, and crusting.3 The term pyogenic granuloma is a misnomer, as these lesions display neither an infectious etiology nor granulomatous tissue on dermatopathologic examination.4 A more accurate clinical description for this lesion is a lobular capillary hemangioma.
Numerous surgical and laser techniques have been used to treat PGs, with varying degrees of success. Treatment often consists of either shave excision followed by electrosurgery at the base or full excision with suturing under local anesthesia for patients who can tolerate anesthetic injections.1 Pulsed dye laser has been proven to be a safe alternative treatment option, particularly in children who otherwise would not tolerate surgical procedures.5 Topical beta-blockers, silver nitrate cauterization, sclerotherapy, and liquid nitrogen all have been used as alternative treatment methods.1
Pyogenic granulomas often recur after first-line treatments, and patients may hesitate to try more invasive techniques when the first choice has failed. Children may not be amenable to any of these curative techniques, as they may not tolerate the pain associated with lidocaine injection and/or have a fear of needles or surgical intervention; even adults may be reluctant to have a procedure they perceive as painful. We present a less invasive technique for treatment of recurrent PGs using table salt and cryotherapy.
The Technique
A 51-year-old woman with no notable medical history presented to the emergency department for evaluation of a black dot on the pulp of the right third fingertip of 1 week’s duration. The patient reported rapid progression to an ulcerated red nodule with associated bleeding for the past 3 days (Figure 1). Direct pressure temporarily alleviated the bleeding, but it started again upon cessation of pressure. She denied any preceding trauma to the area or any associated systemic symptoms such as fever, chills, nausea, or vomiting.

The inpatient dermatology team recommended that the patient be discharged following silver nitrate cautery, with a referral sent to outpatient dermatology; however, the patient returned to the dermatology clinic 4 days later, at which time physical examination revealed a well-circumscribed, 5-mm, bright-red, erosive papule with overlying hemorrhagic crust that was not actively bleeding, as well as fissuring of the surrounding skin. The entire lesion was removed using tangential excision followed by electrodesiccation at the base. Pathology revealed small capillaries arranged in a lobular pattern, confirming the diagnosis of PG. At a 2-week follow-up visit, the patient noted that the lesion had recurred within 24 hours after the procedure and was larger (Figure 2). At this visit, management was switched to a single treatment of cryotherapy (3 cycles for 5 seconds per cycle), and the table salt method was recommended based on a literature review for alternative nonpainful approaches for PG.6-11 We used this technique in our patient as an adjuvant to cryotherapy with the goal of reducing the need for additional painful procedures, but table salt also can be used as a standalone treatment without prior cryotherapy.

The patient was instructed to apply table salt to the lesion once daily for 2 weeks by pressing the lesion into a small amount of salt placed on a clean plate and then applying an occlusive dressing such as surgical or paper tape. She also was advised to apply petroleum jelly around the periphery of the lesion prior to salt application to protect the unaffected skin from irritation. Complete resolution of the lesion was seen when the patient followed up 2 weeks later (Figure 3). At the most recent follow-up 2 months after treatment, no further recurrence of the PG was reported.

Practice Implication
Pyogenic granulomas can be distressing for both patients and providers because they are cosmetically bothersome and prone to spontaneous bleeding. Various medical and surgical options exist to treat PGs, but there is no clear consensus on a superior modality. A 2019 study by Daruwalla and Dhurat6 highlighted a less invasive treatment option for PGs using table salt applied once daily for 2 weeks under an occlusive dressing with good outcomes and without involving other treatments such as cryotherapy. Several other case reports have endorsed this approach, adding anecdotal evidence for its utility.7-11 Topical sodium chloride may treat PGs primarily through osmotic desiccation, drawing water out of the lesion and leading to endothelial cell shrinkage and collapse of its capillary network. This hyperosmolar environment also may induce microvascular thrombosis and ischemia, promoting lesion necrosis. Additionally, repeated application creates a dry, mildly irritative surface that may suppress angiogenesis and encourage regression of the vascular proliferation.
Consider topical application of table salt for treatment of PGs in certain subsets of patients; for example, patients who are not amenable to surgery or are too young for advanced surgical techniques may be good candidates for this method, as it does not require anesthetic injections and generally is pain free. Patients with resistant or recurrent PGs that did not respond to first-line treatments such as cryotherapy, tangential excision, or electrodesiccation may be more amenable to a less invasive secondary approach.
Importantly, we used a dual-therapy approach in our patient, initially using a single application of cryotherapy followed by the table salt method once daily for 2 weeks. This imposes limitations on the generalizability of table salt as a standalone approach for treating PGs. In this case, we did not have prior practical experience using table salt for this condition and only had small reports to justify its use. As a result, we attempted a more traditional treatment initially (cryotherapy) to avoid potential delays in resolution. The clinicians recommended table salt as an adjuvant prior to seeing the cryotherapy results because this treatment was benign and offered potential additive results, and therefore waiting was not necessary. However, various other cases have reported similar success using table salt as monotherapy.6-9,11 Patients should be advised of potential mild adverse events, such as irritation to the surrounding skin. Higher-level evidence studies are required to further vet the utility of the table salt method for treatment of PGs.
- Bolognia JL, Schaffer JV, Cerroni L. Vascular neoplasms and neoplastic‑like proliferations. In: Dermatology. Elsevier; 2018.
- Harris MN, Desai R, Chuang TY, et al. Lobular capillary hemangiomas: an epidemiologic report, with emphasis on cutaneous lesions. J Am Acad Dermatol. 2000;42:1012-1016.
- Ferringer TK, DiCaudo DJ, Elston D, et al. Dermatopathology. W.B. Saunders; 2008.
- Gomes SR, Shakir QJ, Thaker PV, et al. Pyogenic granuloma of the gingiva: a misnomer? - a case report and review of literature. J Indian Soc Periodontol. 2013;17:514-519. doi:10.4103/0972-124X.118327
- Sud AR, Tan ST. Pyogenic granuloma-treatment by shave-excision and/or pulsed-dye laser. J Plast Reconstr Aesthet Surg. 2010;63:1364-1368. doi:10.1016/j.bjps.2009.06.031
- Daruwalla SB, Dhurat RS. A pinch of salt is all it takes! the novel use of table salt for the effective treatment of pyogenic granuloma. J Am Acad Dermatol. 2020;83:E107-E108. doi:10.1016/j.jaad.2019.12.013
- Alhammad G, Albaraka M, Alotaibi H, et al. The use of common salt for the treatment of pyogenic granuloma. JAAD Case Rep. 2024;53:40-42. doi:10.1016/j.jdcr.2024.08.016
- Weiss ES, Wood D. Simple, safe, and effective treatment for pyogenic granuloma. Can Fam Physician. 2023;69:479-480. doi:10.46747/cfp.6907479
- Bernales Salinas A, Toro Sepúlveda A, Meier Pincheira H, et al. Case report: pyogenic granuloma-just salt, a simple and pain-free treatment. Dermatol Ther. 2022;35:E15194. doi:10.1111/dth.15194
- Martín-Nieto González J, Rodríguez-Sánchez B, Berna-Rico E, et al. Pyogenic granuloma resolved with timolol and table salt. An Pediatr (Engl Ed). 2025;102:503706. doi:10.1016/j.anpede.2025.503706
- Bin Rubaian NF. Complete resolution of a refractory pyogenic granuloma following topical salt treatment. Open Access Emerg Med. 2021;13:445-448. doi:10.2147/OAEM.S323793
- Bolognia JL, Schaffer JV, Cerroni L. Vascular neoplasms and neoplastic‑like proliferations. In: Dermatology. Elsevier; 2018.
- Harris MN, Desai R, Chuang TY, et al. Lobular capillary hemangiomas: an epidemiologic report, with emphasis on cutaneous lesions. J Am Acad Dermatol. 2000;42:1012-1016.
- Ferringer TK, DiCaudo DJ, Elston D, et al. Dermatopathology. W.B. Saunders; 2008.
- Gomes SR, Shakir QJ, Thaker PV, et al. Pyogenic granuloma of the gingiva: a misnomer? - a case report and review of literature. J Indian Soc Periodontol. 2013;17:514-519. doi:10.4103/0972-124X.118327
- Sud AR, Tan ST. Pyogenic granuloma-treatment by shave-excision and/or pulsed-dye laser. J Plast Reconstr Aesthet Surg. 2010;63:1364-1368. doi:10.1016/j.bjps.2009.06.031
- Daruwalla SB, Dhurat RS. A pinch of salt is all it takes! the novel use of table salt for the effective treatment of pyogenic granuloma. J Am Acad Dermatol. 2020;83:E107-E108. doi:10.1016/j.jaad.2019.12.013
- Alhammad G, Albaraka M, Alotaibi H, et al. The use of common salt for the treatment of pyogenic granuloma. JAAD Case Rep. 2024;53:40-42. doi:10.1016/j.jdcr.2024.08.016
- Weiss ES, Wood D. Simple, safe, and effective treatment for pyogenic granuloma. Can Fam Physician. 2023;69:479-480. doi:10.46747/cfp.6907479
- Bernales Salinas A, Toro Sepúlveda A, Meier Pincheira H, et al. Case report: pyogenic granuloma-just salt, a simple and pain-free treatment. Dermatol Ther. 2022;35:E15194. doi:10.1111/dth.15194
- Martín-Nieto González J, Rodríguez-Sánchez B, Berna-Rico E, et al. Pyogenic granuloma resolved with timolol and table salt. An Pediatr (Engl Ed). 2025;102:503706. doi:10.1016/j.anpede.2025.503706
- Bin Rubaian NF. Complete resolution of a refractory pyogenic granuloma following topical salt treatment. Open Access Emerg Med. 2021;13:445-448. doi:10.2147/OAEM.S323793
Table Salt Method Following Cryotherapy for Recurrent Pyogenic Granuloma on the Fingertip
Table Salt Method Following Cryotherapy for Recurrent Pyogenic Granuloma on the Fingertip
A Hybrid Suture Technique: Suture Modification With Dental Roll Insertion
A Hybrid Suture Technique: Suture Modification With Dental Roll Insertion
Practice Gap
If not cared for properly, epidermal suture knots can cause discomfort, skin irritation, and an increased risk for infection. There is limited guidance on a simple adaptable method to reduce tram-track marks from epidermal sutures exerting pressure on the epidermis while still facilitating healing in dermatologic procedures such as excision of cysts or lipomas. We present a hybrid suture method that combines elements of traditional simple interrupted and retention sutures with a layer of sterile, absorbent rolled gauze or a dental roll placed beneath the suture knots.
The Technique
Traditional epidermal sutures concentrate pressure at the knot, increasing the risk for tram-track marks and patient discomfort. To address this, we developed a hybrid technique combining simple interrupted sutures with a sterile dental roll beneath the knots to reduce pressure, protect the wound, and promote comfortable wound healing.
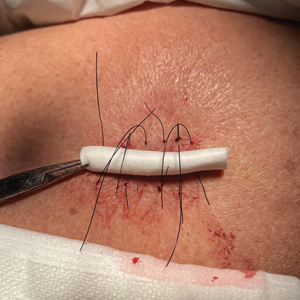
After excision of a cyst, we approximated the wound edges with buried vertical mattress sutures for eversion (a set-back buried dermal suture also may be used). The sutures initially were placed loosely but were left untied (eFigure 1A). A sterile dental roll with sterile petrolatum on the underside was positioned over the wound before the knots were secured, similar to a bolster dressing (eFigures 1B and 1C). The dressing then was covered and left in place for 24 to 48 hours. After removal of the dressing, no bandage was needed because the wound was clean and hemostatic and the dental roll had absorbed minimal drainage and protected the incision edges during the initial healing period. The patient applied petrolatum daily to prevent the dental roll from drying out. Sutures and the bolster were removed at 14 days without complications or complaints.
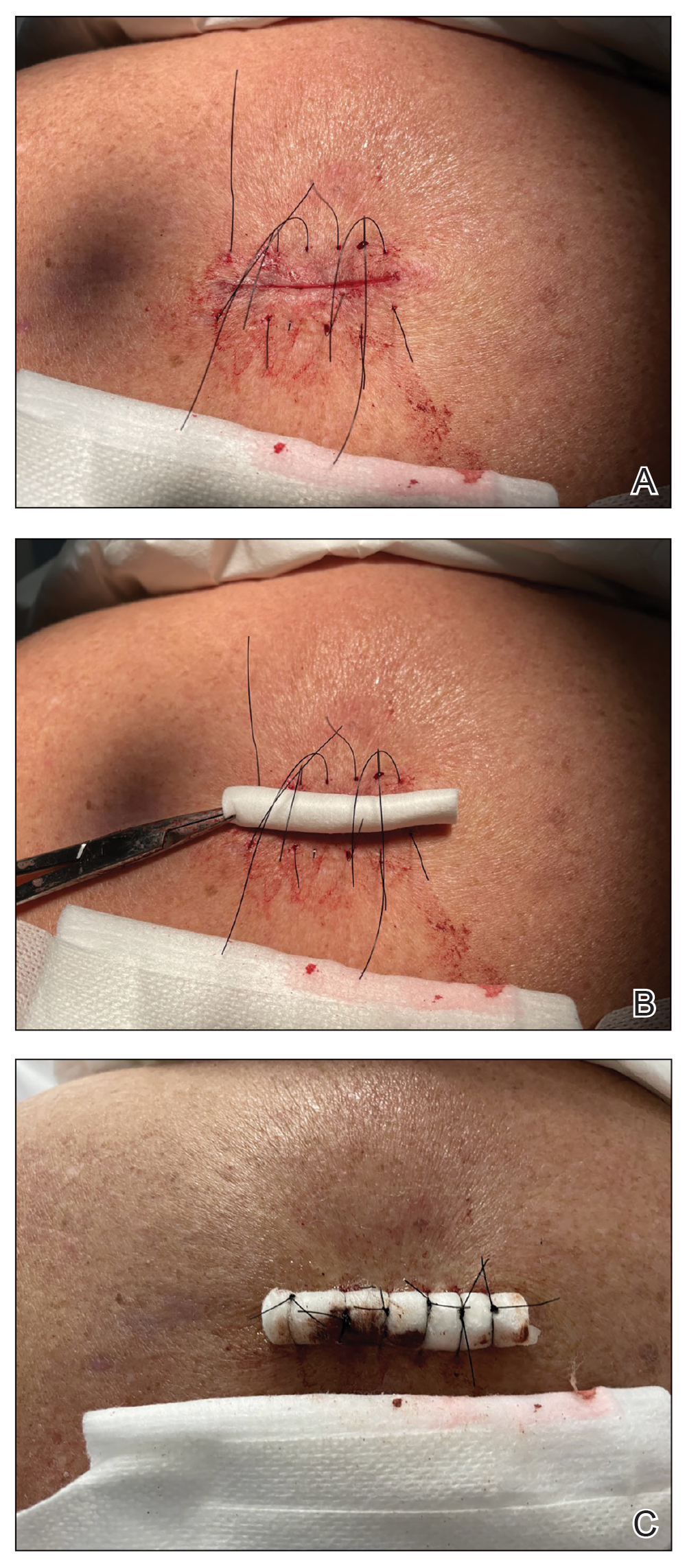
Rolled gauze may be used as an alternative to the dental roll. To maintain a clean surgical field, nonsterile gauze may be soaked in a disinfectant (eg, alcohol) and wrung out to remove excess moisture before placement on the skin. The side of the gauze in contact with the skin also should be lubricated with petroleum jelly to prevent sticking. If the sutures slip during knot tying, one end can be secured with a needle driver or hemostat. Patients should be advised to keep the dental roll dry to prevent maceration and promote optimal wound healing, but minor dampness is permissible if followed by air-drying.
This suturing method is most suitable for low- to moderate-tension closures such as cyst or lipoma excisions. The serosanguinous drainage can be absorbed by the gauze or dental roll while pressure is simultaneously applied to the wound. We do not recommend this technique for high-tension wounds in which large surface areas are removed (eg, skin cancer excisions on the posterior shoulder that require wide margins). Close monitoring of the wound for dehiscence is needed. As the sutures stretch and swelling decreases, the pressure is distributed accordingly without excessive compression to the wound line. Depending on the location, the sutures and dental roll can be removed in 7 to 14 days.
Practice Implications
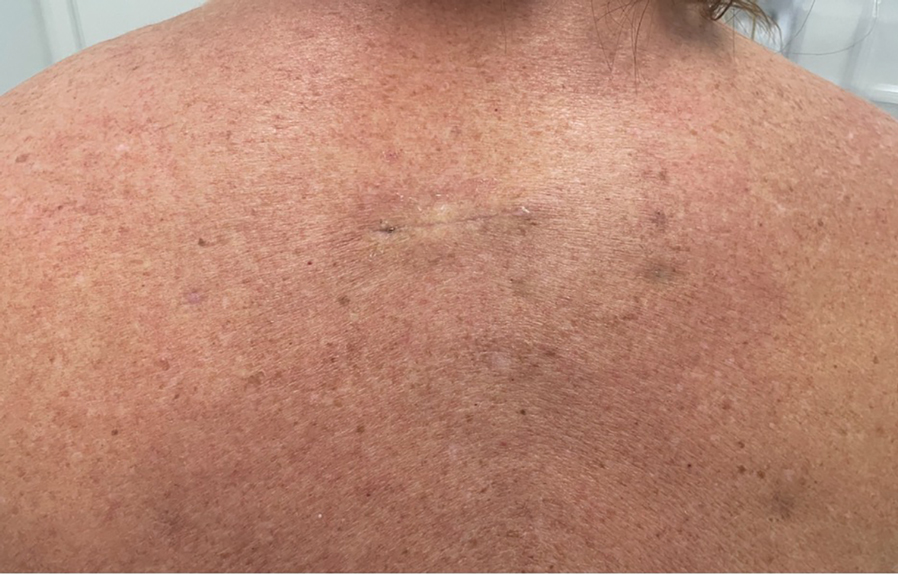
Placing a dental roll or rolled gauze beneath suture knots can prevent tram-track scarring by eliminating direct knot-to-skin contact (eFigure 2).1,2 This technique distributes tension evenly, reduces the risk for wound edge necrosis, and absorbs serosanguinous drainage while providing hemostasis. The modification is quick, inexpensive, and especially beneficial for patients who may struggle with complex wound care, maintaining a clean environment until sutures are removed.
- Li E, Zhang T, Ma Q, et al. Effect of modified Allgöwer-Donati suture technique on wound cosmetics in spinal surgery. Orthop Surg. 2022;14:678-685. doi:10.1111/os.13188
- Park L, Lum ZC, Joo JS. Allgöwer-Donati suture: a technique to preserve skin microcirculation in dermatologic surgery. J Am Acad Dermatol. 2024;90:E129-E130. doi:10.1016/j.jaad.2023.05.052
Practice Gap
If not cared for properly, epidermal suture knots can cause discomfort, skin irritation, and an increased risk for infection. There is limited guidance on a simple adaptable method to reduce tram-track marks from epidermal sutures exerting pressure on the epidermis while still facilitating healing in dermatologic procedures such as excision of cysts or lipomas. We present a hybrid suture method that combines elements of traditional simple interrupted and retention sutures with a layer of sterile, absorbent rolled gauze or a dental roll placed beneath the suture knots.
The Technique
Traditional epidermal sutures concentrate pressure at the knot, increasing the risk for tram-track marks and patient discomfort. To address this, we developed a hybrid technique combining simple interrupted sutures with a sterile dental roll beneath the knots to reduce pressure, protect the wound, and promote comfortable wound healing.
After excision of a cyst, we approximated the wound edges with buried vertical mattress sutures for eversion (a set-back buried dermal suture also may be used). The sutures initially were placed loosely but were left untied (eFigure 1A). A sterile dental roll with sterile petrolatum on the underside was positioned over the wound before the knots were secured, similar to a bolster dressing (eFigures 1B and 1C). The dressing then was covered and left in place for 24 to 48 hours. After removal of the dressing, no bandage was needed because the wound was clean and hemostatic and the dental roll had absorbed minimal drainage and protected the incision edges during the initial healing period. The patient applied petrolatum daily to prevent the dental roll from drying out. Sutures and the bolster were removed at 14 days without complications or complaints.

Rolled gauze may be used as an alternative to the dental roll. To maintain a clean surgical field, nonsterile gauze may be soaked in a disinfectant (eg, alcohol) and wrung out to remove excess moisture before placement on the skin. The side of the gauze in contact with the skin also should be lubricated with petroleum jelly to prevent sticking. If the sutures slip during knot tying, one end can be secured with a needle driver or hemostat. Patients should be advised to keep the dental roll dry to prevent maceration and promote optimal wound healing, but minor dampness is permissible if followed by air-drying.
This suturing method is most suitable for low- to moderate-tension closures such as cyst or lipoma excisions. The serosanguinous drainage can be absorbed by the gauze or dental roll while pressure is simultaneously applied to the wound. We do not recommend this technique for high-tension wounds in which large surface areas are removed (eg, skin cancer excisions on the posterior shoulder that require wide margins). Close monitoring of the wound for dehiscence is needed. As the sutures stretch and swelling decreases, the pressure is distributed accordingly without excessive compression to the wound line. Depending on the location, the sutures and dental roll can be removed in 7 to 14 days.
Practice Implications
Placing a dental roll or rolled gauze beneath suture knots can prevent tram-track scarring by eliminating direct knot-to-skin contact (eFigure 2).1,2 This technique distributes tension evenly, reduces the risk for wound edge necrosis, and absorbs serosanguinous drainage while providing hemostasis. The modification is quick, inexpensive, and especially beneficial for patients who may struggle with complex wound care, maintaining a clean environment until sutures are removed.

Practice Gap
If not cared for properly, epidermal suture knots can cause discomfort, skin irritation, and an increased risk for infection. There is limited guidance on a simple adaptable method to reduce tram-track marks from epidermal sutures exerting pressure on the epidermis while still facilitating healing in dermatologic procedures such as excision of cysts or lipomas. We present a hybrid suture method that combines elements of traditional simple interrupted and retention sutures with a layer of sterile, absorbent rolled gauze or a dental roll placed beneath the suture knots.
The Technique
Traditional epidermal sutures concentrate pressure at the knot, increasing the risk for tram-track marks and patient discomfort. To address this, we developed a hybrid technique combining simple interrupted sutures with a sterile dental roll beneath the knots to reduce pressure, protect the wound, and promote comfortable wound healing.
After excision of a cyst, we approximated the wound edges with buried vertical mattress sutures for eversion (a set-back buried dermal suture also may be used). The sutures initially were placed loosely but were left untied (eFigure 1A). A sterile dental roll with sterile petrolatum on the underside was positioned over the wound before the knots were secured, similar to a bolster dressing (eFigures 1B and 1C). The dressing then was covered and left in place for 24 to 48 hours. After removal of the dressing, no bandage was needed because the wound was clean and hemostatic and the dental roll had absorbed minimal drainage and protected the incision edges during the initial healing period. The patient applied petrolatum daily to prevent the dental roll from drying out. Sutures and the bolster were removed at 14 days without complications or complaints.

Rolled gauze may be used as an alternative to the dental roll. To maintain a clean surgical field, nonsterile gauze may be soaked in a disinfectant (eg, alcohol) and wrung out to remove excess moisture before placement on the skin. The side of the gauze in contact with the skin also should be lubricated with petroleum jelly to prevent sticking. If the sutures slip during knot tying, one end can be secured with a needle driver or hemostat. Patients should be advised to keep the dental roll dry to prevent maceration and promote optimal wound healing, but minor dampness is permissible if followed by air-drying.
This suturing method is most suitable for low- to moderate-tension closures such as cyst or lipoma excisions. The serosanguinous drainage can be absorbed by the gauze or dental roll while pressure is simultaneously applied to the wound. We do not recommend this technique for high-tension wounds in which large surface areas are removed (eg, skin cancer excisions on the posterior shoulder that require wide margins). Close monitoring of the wound for dehiscence is needed. As the sutures stretch and swelling decreases, the pressure is distributed accordingly without excessive compression to the wound line. Depending on the location, the sutures and dental roll can be removed in 7 to 14 days.
Practice Implications
Placing a dental roll or rolled gauze beneath suture knots can prevent tram-track scarring by eliminating direct knot-to-skin contact (eFigure 2).1,2 This technique distributes tension evenly, reduces the risk for wound edge necrosis, and absorbs serosanguinous drainage while providing hemostasis. The modification is quick, inexpensive, and especially beneficial for patients who may struggle with complex wound care, maintaining a clean environment until sutures are removed.

- Li E, Zhang T, Ma Q, et al. Effect of modified Allgöwer-Donati suture technique on wound cosmetics in spinal surgery. Orthop Surg. 2022;14:678-685. doi:10.1111/os.13188
- Park L, Lum ZC, Joo JS. Allgöwer-Donati suture: a technique to preserve skin microcirculation in dermatologic surgery. J Am Acad Dermatol. 2024;90:E129-E130. doi:10.1016/j.jaad.2023.05.052
- Li E, Zhang T, Ma Q, et al. Effect of modified Allgöwer-Donati suture technique on wound cosmetics in spinal surgery. Orthop Surg. 2022;14:678-685. doi:10.1111/os.13188
- Park L, Lum ZC, Joo JS. Allgöwer-Donati suture: a technique to preserve skin microcirculation in dermatologic surgery. J Am Acad Dermatol. 2024;90:E129-E130. doi:10.1016/j.jaad.2023.05.052
A Hybrid Suture Technique: Suture Modification With Dental Roll Insertion
A Hybrid Suture Technique: Suture Modification With Dental Roll Insertion
A Simple Alternative for Intralesional Cryosurgery of Keloids and Hypertrophic Scars Using a Disposable Infusion Set
A Simple Alternative for Intralesional Cryosurgery of Keloids and Hypertrophic Scars Using a Disposable Infusion Set
Practice Gap
Intralesional cryosurgery is a highly effective treatment for dermatologic conditions, notably keloids and hypertrophic scars.1 Conventional methods typically use specialized double-lumen intralesional probes or Luer lock adapters connected to hypodermic or lumbar puncture needles, allowing cryogen to flow internally to cool the probe or needle and treat the lesion via conduction.2 However, specialized intralesional probes are expensive and often are difficult to obtain. Furthermore, Luer lock adapters with needles directly attached to the handle unit can be ergonomically challenging, as the procedure requires simultaneous maintenance of a perpendicular handheld position, precise needle passage through the exact center of the lesion, and protection of the surrounding perilesional healthy skin from cold injury. Consequently, these limitations restrict widespread adoption, necessitating simpler, more accessible, and cost-effective alternatives. Herein, we present a novel, practical, and economical cryogen delivery method that adapts a disposable infusion set to a standard cryospray nozzle.
The Technique
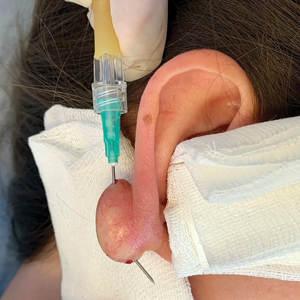
This technique involves detaching the infusion set tubing and securely connecting it to the cryospray nozzle (Figure 1). Brief activation of the cryospray to constrict the nozzle or a small incision in the tubing may be required to ensure a tight fit, which can be secured with medical tape to maintain consistent cryogen flow. Local anesthesia is administered directly into and around the lesion, particularly translesionally for keloids, to avoid unnecessary trauma to the surrounding healthy skin, which could trigger further keloid formation. A needle is inserted through the lesion with the tip extending beyond its distal boundary, ensuring the tip remains outside the lesion during cryogen application to prevent cryoinsufflation. If necessary, gentle bending of the needle helps ensure optimal cryogen distribution within the lesion (Figures 2A and 2B). However, this may slightly reduce flow and extend freezing duration; therefore, bending the needle should be performed cautiously and is specifically recommended for effectively treating lesions on curved anatomic sites (eg, the auricle of the ear) to optimize freezing and protect surrounding tissues.

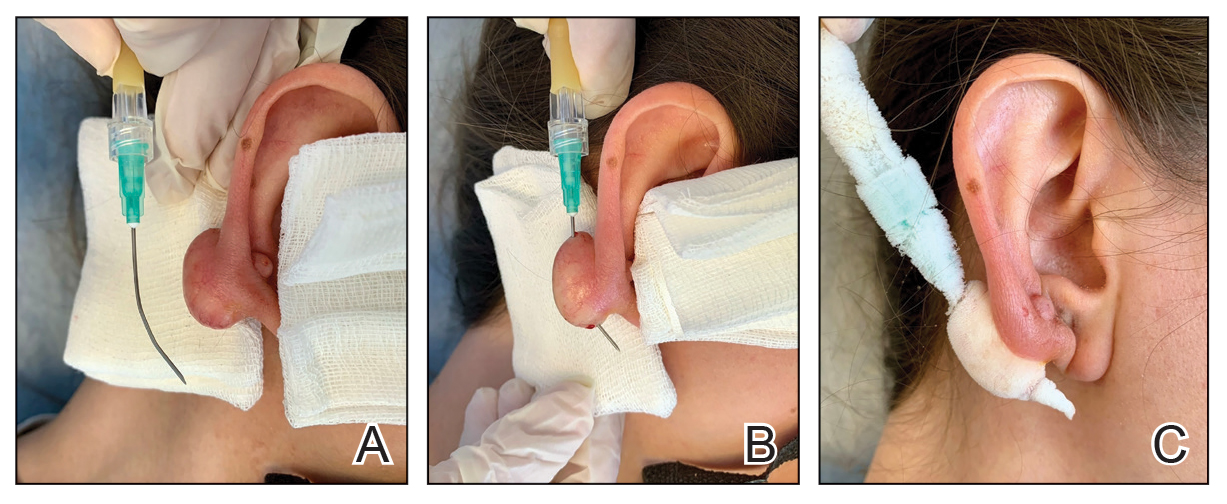
During initial cryogen release, covering the needle tip with gauze prevents aerosolization of biological debris, while placing a wooden tongue depressor between the needle tip and the patient’s skin prevents inadvertent cold injury. After cryogen flow is initiated, the lesion begins to freeze at both the needle entry and exit points, forming what is referred to as ice balls. Over time, typically within several seconds to a few minutes depending on lesion size and tissue characteristics, these ice balls merge centrally, forming a single ice ball encompassing the entire lesion (Figure 2C). Cryogen flow should be maintained during a single application until the unified ice ball appearance is achieved, confirming effective cooling.
Practice Implications
Studies have consistently shown that intralesional cryosurgery is associated with a reduction in the size and symptoms of hypertrophic scars and keloids.1,2 Multimodal treatment approaches, including intralesional methods, are especially valued for their targeted efficacy and minimal adverse effects. Our simplified method offers practical economic advantages, making it highly suitable for broad adoption across diverse clinical settings, particularly those that are resource limited. Clinicians can safely and effectively utilize this technique without specialized or costly equipment, considerably enhancing clinical efficiency and accessibility. The straightforwardness of this method also facilitates the training of medical personnel, enabling rapid integration into clinical practice and the flexibility to treat various lesion types and sizes effectively.
- McGoldrick RB, Theodorakopoulou E, Azzopardi E, et al. Lasers and ancillary treatments for scar management part 2: keloid, hypertrophic, pigmented and acne scars. Scars Burn Heal. 2017;3:1-16. doi:10.1177/2059513116689805
- Gupta S, Kumar B. Intralesional cryosurgery using lumbar puncture and/or hypodermic needles for large, bulky, recalcitrant keloids. Int J Dermatol. 2001;40:349-353. doi:10.1046/j.1365-4362.2001.01117.x
Practice Gap
Intralesional cryosurgery is a highly effective treatment for dermatologic conditions, notably keloids and hypertrophic scars.1 Conventional methods typically use specialized double-lumen intralesional probes or Luer lock adapters connected to hypodermic or lumbar puncture needles, allowing cryogen to flow internally to cool the probe or needle and treat the lesion via conduction.2 However, specialized intralesional probes are expensive and often are difficult to obtain. Furthermore, Luer lock adapters with needles directly attached to the handle unit can be ergonomically challenging, as the procedure requires simultaneous maintenance of a perpendicular handheld position, precise needle passage through the exact center of the lesion, and protection of the surrounding perilesional healthy skin from cold injury. Consequently, these limitations restrict widespread adoption, necessitating simpler, more accessible, and cost-effective alternatives. Herein, we present a novel, practical, and economical cryogen delivery method that adapts a disposable infusion set to a standard cryospray nozzle.
The Technique
This technique involves detaching the infusion set tubing and securely connecting it to the cryospray nozzle (Figure 1). Brief activation of the cryospray to constrict the nozzle or a small incision in the tubing may be required to ensure a tight fit, which can be secured with medical tape to maintain consistent cryogen flow. Local anesthesia is administered directly into and around the lesion, particularly translesionally for keloids, to avoid unnecessary trauma to the surrounding healthy skin, which could trigger further keloid formation. A needle is inserted through the lesion with the tip extending beyond its distal boundary, ensuring the tip remains outside the lesion during cryogen application to prevent cryoinsufflation. If necessary, gentle bending of the needle helps ensure optimal cryogen distribution within the lesion (Figures 2A and 2B). However, this may slightly reduce flow and extend freezing duration; therefore, bending the needle should be performed cautiously and is specifically recommended for effectively treating lesions on curved anatomic sites (eg, the auricle of the ear) to optimize freezing and protect surrounding tissues.


During initial cryogen release, covering the needle tip with gauze prevents aerosolization of biological debris, while placing a wooden tongue depressor between the needle tip and the patient’s skin prevents inadvertent cold injury. After cryogen flow is initiated, the lesion begins to freeze at both the needle entry and exit points, forming what is referred to as ice balls. Over time, typically within several seconds to a few minutes depending on lesion size and tissue characteristics, these ice balls merge centrally, forming a single ice ball encompassing the entire lesion (Figure 2C). Cryogen flow should be maintained during a single application until the unified ice ball appearance is achieved, confirming effective cooling.
Practice Implications
Studies have consistently shown that intralesional cryosurgery is associated with a reduction in the size and symptoms of hypertrophic scars and keloids.1,2 Multimodal treatment approaches, including intralesional methods, are especially valued for their targeted efficacy and minimal adverse effects. Our simplified method offers practical economic advantages, making it highly suitable for broad adoption across diverse clinical settings, particularly those that are resource limited. Clinicians can safely and effectively utilize this technique without specialized or costly equipment, considerably enhancing clinical efficiency and accessibility. The straightforwardness of this method also facilitates the training of medical personnel, enabling rapid integration into clinical practice and the flexibility to treat various lesion types and sizes effectively.
Practice Gap
Intralesional cryosurgery is a highly effective treatment for dermatologic conditions, notably keloids and hypertrophic scars.1 Conventional methods typically use specialized double-lumen intralesional probes or Luer lock adapters connected to hypodermic or lumbar puncture needles, allowing cryogen to flow internally to cool the probe or needle and treat the lesion via conduction.2 However, specialized intralesional probes are expensive and often are difficult to obtain. Furthermore, Luer lock adapters with needles directly attached to the handle unit can be ergonomically challenging, as the procedure requires simultaneous maintenance of a perpendicular handheld position, precise needle passage through the exact center of the lesion, and protection of the surrounding perilesional healthy skin from cold injury. Consequently, these limitations restrict widespread adoption, necessitating simpler, more accessible, and cost-effective alternatives. Herein, we present a novel, practical, and economical cryogen delivery method that adapts a disposable infusion set to a standard cryospray nozzle.
The Technique
This technique involves detaching the infusion set tubing and securely connecting it to the cryospray nozzle (Figure 1). Brief activation of the cryospray to constrict the nozzle or a small incision in the tubing may be required to ensure a tight fit, which can be secured with medical tape to maintain consistent cryogen flow. Local anesthesia is administered directly into and around the lesion, particularly translesionally for keloids, to avoid unnecessary trauma to the surrounding healthy skin, which could trigger further keloid formation. A needle is inserted through the lesion with the tip extending beyond its distal boundary, ensuring the tip remains outside the lesion during cryogen application to prevent cryoinsufflation. If necessary, gentle bending of the needle helps ensure optimal cryogen distribution within the lesion (Figures 2A and 2B). However, this may slightly reduce flow and extend freezing duration; therefore, bending the needle should be performed cautiously and is specifically recommended for effectively treating lesions on curved anatomic sites (eg, the auricle of the ear) to optimize freezing and protect surrounding tissues.


During initial cryogen release, covering the needle tip with gauze prevents aerosolization of biological debris, while placing a wooden tongue depressor between the needle tip and the patient’s skin prevents inadvertent cold injury. After cryogen flow is initiated, the lesion begins to freeze at both the needle entry and exit points, forming what is referred to as ice balls. Over time, typically within several seconds to a few minutes depending on lesion size and tissue characteristics, these ice balls merge centrally, forming a single ice ball encompassing the entire lesion (Figure 2C). Cryogen flow should be maintained during a single application until the unified ice ball appearance is achieved, confirming effective cooling.
Practice Implications
Studies have consistently shown that intralesional cryosurgery is associated with a reduction in the size and symptoms of hypertrophic scars and keloids.1,2 Multimodal treatment approaches, including intralesional methods, are especially valued for their targeted efficacy and minimal adverse effects. Our simplified method offers practical economic advantages, making it highly suitable for broad adoption across diverse clinical settings, particularly those that are resource limited. Clinicians can safely and effectively utilize this technique without specialized or costly equipment, considerably enhancing clinical efficiency and accessibility. The straightforwardness of this method also facilitates the training of medical personnel, enabling rapid integration into clinical practice and the flexibility to treat various lesion types and sizes effectively.
- McGoldrick RB, Theodorakopoulou E, Azzopardi E, et al. Lasers and ancillary treatments for scar management part 2: keloid, hypertrophic, pigmented and acne scars. Scars Burn Heal. 2017;3:1-16. doi:10.1177/2059513116689805
- Gupta S, Kumar B. Intralesional cryosurgery using lumbar puncture and/or hypodermic needles for large, bulky, recalcitrant keloids. Int J Dermatol. 2001;40:349-353. doi:10.1046/j.1365-4362.2001.01117.x
- McGoldrick RB, Theodorakopoulou E, Azzopardi E, et al. Lasers and ancillary treatments for scar management part 2: keloid, hypertrophic, pigmented and acne scars. Scars Burn Heal. 2017;3:1-16. doi:10.1177/2059513116689805
- Gupta S, Kumar B. Intralesional cryosurgery using lumbar puncture and/or hypodermic needles for large, bulky, recalcitrant keloids. Int J Dermatol. 2001;40:349-353. doi:10.1046/j.1365-4362.2001.01117.x
A Simple Alternative for Intralesional Cryosurgery of Keloids and Hypertrophic Scars Using a Disposable Infusion Set
A Simple Alternative for Intralesional Cryosurgery of Keloids and Hypertrophic Scars Using a Disposable Infusion Set
Waterproof Cast Protector Keeps Wound Dressing Intact Following Nail Surgery
Waterproof Cast Protector Keeps Wound Dressing Intact Following Nail Surgery
Practice Gap
Postoperative care after nail biopsies can be challenging for patients due to the bulky dressing that must remain in place for 48 hours.1 The dressing can restrict daily activities such as bathing, washing dishes, and other household tasks. A common solution is to cover the hand with a plastic bag secured with tape during water-related activities, but efficacy is variable. In one study, 23 participants tested this method by holding a paper towel with their hand covered by a plastic bag and measuring the weight of the paper towel before and after submersion of the hand in water.2 Any saturation of the paper towel was defined as failure; the failure rate was 52.2% (12/23) with motion (rotating the arm at the elbow for 30 seconds clockwise, counterclockwise, and left to right) and 60.9% (14/23) without motion. There was an average of 5.50 g of moisture accumulation without motion and 4.51 g with motion, with failure occurring most often immediately following submersion of the hand. Furthermore, the plastic bag with tape method was rated poorly by all 23 participants based on efficacy and comfort.2
In the same study, participants also reported that removal of the adhesive tape was unpleasant and irritating,2 which suggests these same complaints may apply to use of a waterproof bandage, another potential option for coverage of the wound dressing. As an alternative, we propose the use of a removable waterproof arm cast protector following nail surgery that allows patients to continue their regular activities while keeping the dressing dry and intact to allow for optimal wound healing.
The Technique
Our technique involves the use of a removable waterproof arm cast protector that is sealed with a thick rubber cuff, allowing patients to perform regular daily activities such as bathing, washing dishes, cleaning, and doing laundry without the wound dressing underneath becoming wet (Figure). Cast protectors made of flexible latex-free plastic are readily available and can slide on and off the arm as needed. We recommend that patients purchase the cast protector prior to undergoing surgery. There are options to fit most adults, with the opening generally accommodating arm diameters of 2 to 7 inches. These reusable cast protectors are available via popular online retailers and typically cost patients $10 to $15.

Practice Implications
In our experience, using a reusable waterproof cast protector following nail surgery is effective at keeping wound dressings dry and provides a practical solution for bathing and other activities involving water exposure. It is durable and easy to use, especially when compared to a plastic bag and waterproof tape. However, some patients find the waterproof seal uncomfortable, especially when worn for extended periods of time. According to online product feedback, limitations of the cast protector include potential leakage with prolonged immersion in water, swimming, or high-pressure water exposure. The cast protector should not be worn for more than 30 minutes, as it can restrict blood flow, and condensation from prolonged use may dampen the dressing. While we have not encountered allergic contact dermatitis associated with the use of cast protectors for this purpose in our practice, patients should be cautioned of this potential risk. While these cast protectors generally can accommodate a range of arm diameters, they may not fit all hand sizes or shapes and may reduce dexterity for motor tasks. Additionally, the patient must purchase the protector ahead of surgery.
Our technique involving the use of a waterproof arm cast protector is an affordable solution that allows patients to keep their wound dressing dry while continuing to perform regular daily activities. The cast protector also can be used following other dermatologic procedures (eg, biopsy, Mohs micrographic surgery) that involve the hand and lower arm when waterproof protection may be necessary.
- Ricardo JW, Lipner SR. How we do it: pressure-padded dressing with self-adherent elastic wrap for wound care after nail surgery. Dermatol Surg. 2021;47:442–444. doi:10.1097/DSS.0000000000002371
- Kwan S, Santoro A, Cheesman Q, et al. Efficacy of waterproof cast protectors and their ability to keep casts dry. J Hand Surg Am. 2023;48:803–809. doi:10.1016/j.jhsa.2022.05.006
Practice Gap
Postoperative care after nail biopsies can be challenging for patients due to the bulky dressing that must remain in place for 48 hours.1 The dressing can restrict daily activities such as bathing, washing dishes, and other household tasks. A common solution is to cover the hand with a plastic bag secured with tape during water-related activities, but efficacy is variable. In one study, 23 participants tested this method by holding a paper towel with their hand covered by a plastic bag and measuring the weight of the paper towel before and after submersion of the hand in water.2 Any saturation of the paper towel was defined as failure; the failure rate was 52.2% (12/23) with motion (rotating the arm at the elbow for 30 seconds clockwise, counterclockwise, and left to right) and 60.9% (14/23) without motion. There was an average of 5.50 g of moisture accumulation without motion and 4.51 g with motion, with failure occurring most often immediately following submersion of the hand. Furthermore, the plastic bag with tape method was rated poorly by all 23 participants based on efficacy and comfort.2
In the same study, participants also reported that removal of the adhesive tape was unpleasant and irritating,2 which suggests these same complaints may apply to use of a waterproof bandage, another potential option for coverage of the wound dressing. As an alternative, we propose the use of a removable waterproof arm cast protector following nail surgery that allows patients to continue their regular activities while keeping the dressing dry and intact to allow for optimal wound healing.
The Technique
Our technique involves the use of a removable waterproof arm cast protector that is sealed with a thick rubber cuff, allowing patients to perform regular daily activities such as bathing, washing dishes, cleaning, and doing laundry without the wound dressing underneath becoming wet (Figure). Cast protectors made of flexible latex-free plastic are readily available and can slide on and off the arm as needed. We recommend that patients purchase the cast protector prior to undergoing surgery. There are options to fit most adults, with the opening generally accommodating arm diameters of 2 to 7 inches. These reusable cast protectors are available via popular online retailers and typically cost patients $10 to $15.

Practice Implications
In our experience, using a reusable waterproof cast protector following nail surgery is effective at keeping wound dressings dry and provides a practical solution for bathing and other activities involving water exposure. It is durable and easy to use, especially when compared to a plastic bag and waterproof tape. However, some patients find the waterproof seal uncomfortable, especially when worn for extended periods of time. According to online product feedback, limitations of the cast protector include potential leakage with prolonged immersion in water, swimming, or high-pressure water exposure. The cast protector should not be worn for more than 30 minutes, as it can restrict blood flow, and condensation from prolonged use may dampen the dressing. While we have not encountered allergic contact dermatitis associated with the use of cast protectors for this purpose in our practice, patients should be cautioned of this potential risk. While these cast protectors generally can accommodate a range of arm diameters, they may not fit all hand sizes or shapes and may reduce dexterity for motor tasks. Additionally, the patient must purchase the protector ahead of surgery.
Our technique involving the use of a waterproof arm cast protector is an affordable solution that allows patients to keep their wound dressing dry while continuing to perform regular daily activities. The cast protector also can be used following other dermatologic procedures (eg, biopsy, Mohs micrographic surgery) that involve the hand and lower arm when waterproof protection may be necessary.
Practice Gap
Postoperative care after nail biopsies can be challenging for patients due to the bulky dressing that must remain in place for 48 hours.1 The dressing can restrict daily activities such as bathing, washing dishes, and other household tasks. A common solution is to cover the hand with a plastic bag secured with tape during water-related activities, but efficacy is variable. In one study, 23 participants tested this method by holding a paper towel with their hand covered by a plastic bag and measuring the weight of the paper towel before and after submersion of the hand in water.2 Any saturation of the paper towel was defined as failure; the failure rate was 52.2% (12/23) with motion (rotating the arm at the elbow for 30 seconds clockwise, counterclockwise, and left to right) and 60.9% (14/23) without motion. There was an average of 5.50 g of moisture accumulation without motion and 4.51 g with motion, with failure occurring most often immediately following submersion of the hand. Furthermore, the plastic bag with tape method was rated poorly by all 23 participants based on efficacy and comfort.2
In the same study, participants also reported that removal of the adhesive tape was unpleasant and irritating,2 which suggests these same complaints may apply to use of a waterproof bandage, another potential option for coverage of the wound dressing. As an alternative, we propose the use of a removable waterproof arm cast protector following nail surgery that allows patients to continue their regular activities while keeping the dressing dry and intact to allow for optimal wound healing.
The Technique
Our technique involves the use of a removable waterproof arm cast protector that is sealed with a thick rubber cuff, allowing patients to perform regular daily activities such as bathing, washing dishes, cleaning, and doing laundry without the wound dressing underneath becoming wet (Figure). Cast protectors made of flexible latex-free plastic are readily available and can slide on and off the arm as needed. We recommend that patients purchase the cast protector prior to undergoing surgery. There are options to fit most adults, with the opening generally accommodating arm diameters of 2 to 7 inches. These reusable cast protectors are available via popular online retailers and typically cost patients $10 to $15.

Practice Implications
In our experience, using a reusable waterproof cast protector following nail surgery is effective at keeping wound dressings dry and provides a practical solution for bathing and other activities involving water exposure. It is durable and easy to use, especially when compared to a plastic bag and waterproof tape. However, some patients find the waterproof seal uncomfortable, especially when worn for extended periods of time. According to online product feedback, limitations of the cast protector include potential leakage with prolonged immersion in water, swimming, or high-pressure water exposure. The cast protector should not be worn for more than 30 minutes, as it can restrict blood flow, and condensation from prolonged use may dampen the dressing. While we have not encountered allergic contact dermatitis associated with the use of cast protectors for this purpose in our practice, patients should be cautioned of this potential risk. While these cast protectors generally can accommodate a range of arm diameters, they may not fit all hand sizes or shapes and may reduce dexterity for motor tasks. Additionally, the patient must purchase the protector ahead of surgery.
Our technique involving the use of a waterproof arm cast protector is an affordable solution that allows patients to keep their wound dressing dry while continuing to perform regular daily activities. The cast protector also can be used following other dermatologic procedures (eg, biopsy, Mohs micrographic surgery) that involve the hand and lower arm when waterproof protection may be necessary.
- Ricardo JW, Lipner SR. How we do it: pressure-padded dressing with self-adherent elastic wrap for wound care after nail surgery. Dermatol Surg. 2021;47:442–444. doi:10.1097/DSS.0000000000002371
- Kwan S, Santoro A, Cheesman Q, et al. Efficacy of waterproof cast protectors and their ability to keep casts dry. J Hand Surg Am. 2023;48:803–809. doi:10.1016/j.jhsa.2022.05.006
- Ricardo JW, Lipner SR. How we do it: pressure-padded dressing with self-adherent elastic wrap for wound care after nail surgery. Dermatol Surg. 2021;47:442–444. doi:10.1097/DSS.0000000000002371
- Kwan S, Santoro A, Cheesman Q, et al. Efficacy of waterproof cast protectors and their ability to keep casts dry. J Hand Surg Am. 2023;48:803–809. doi:10.1016/j.jhsa.2022.05.006
Waterproof Cast Protector Keeps Wound Dressing Intact Following Nail Surgery
Waterproof Cast Protector Keeps Wound Dressing Intact Following Nail Surgery
Interactive Approach to Teaching Mohs Micrographic Surgery to Dermatology Residents
Interactive Approach to Teaching Mohs Micrographic Surgery to Dermatology Residents
Practice Gap
Tissue processing and complete margin assessment in Mohs micrographic surgery (MMS) are challenging concepts for residents, yet they are essential components of the dermatology residency curriculum. We propose a hands-on active teaching method using craft foam blocks to help residents master these techniques. Prior educational tools have included instructional videos1 as well as the peanut butter–cup and cantaloupe analogies.2,3 Specifically, our method utilizes inexpensive, readily available supplies that allow for repeated practice in a low-stakes environment without limitation of resources. This method provides an immersive, hands-on experience that allows residents to perform multiple practice excisions and simulate positive peripheral or deep margins, unlike tools that offer only fixed-depth or purely visual representations. Additionally, our learning model uniquely enables residents to flatten the simulated tissue, providing a clearer understanding of how a 3-dimensional specimen is transformed on a slide during histologic preparation. This step is particularly important, as tissue architecture can shift during processing, making it one of the most difficult concepts to grasp without hands-on experience. Having a multitude of teaching methods is crucial to accommodate various learning styles, and active learning has been shown to enhance retention for dermatology residents.4
The Technique
Residents use simple art supplies (including craft foam blocks and ink) and inexpensive, readily available surgical tools to simulate MMS (Table)(Figure 1). If desired, the resident can follow along with the comprehensive, stepwise textbook description of MMS, outlined by Benedetto et al5 to contextualize this hands-on exercise within a standardized didactic framework.


The foam block, which represents patient tissue, serves as the specimen. The resident begins by freehand drawing a simulated cutaneous tumor directly onto the foam using a surgical marking pen. At this point, the instructor discusses the advantages and limitations of tumor debulking with a sharp blade or curette. Residents then mark appropriate margins (1-3 mm) of normal-appearing “epidermis” on the foam block and add hash marks for orientation. This is another opportunity for the instructor to discuss common methods for marking tissue in vivo and to review situations when larger or smaller margins might be appropriate.
Next, the resident removes the first layer of simulated tissue using a disposable #15 blade scalpel at a 45° angle circumferentially and deep around the representative tumor. The resident also may use scissors and tissue forceps to remove the representative tumor. Next, the excised foam layer (the simulated “specimen”) is transferred to gauze. To demonstrate a positive margin, the resident or instructor marks the deep or peripheral foam block with a surgical marking pen, indicating residual tumor (Figure 2). This allows for multiple sequential layers of foam to be removed, demonstrating successive stages of MMS.

An inkwell holds different colors of washable paint to simulate tissue inking. After excision, the resident uses cotton-tipped applicators to apply different paint colors to the edges of the excised foam specimen at designated orientation points (eg, 3 o'clock and 12 o'clock). The resident then records the location of the excised sample by hand-drawing it on a printable Mohs map, labeling the corresponding paint colors to indicate orientation (Figure 3).
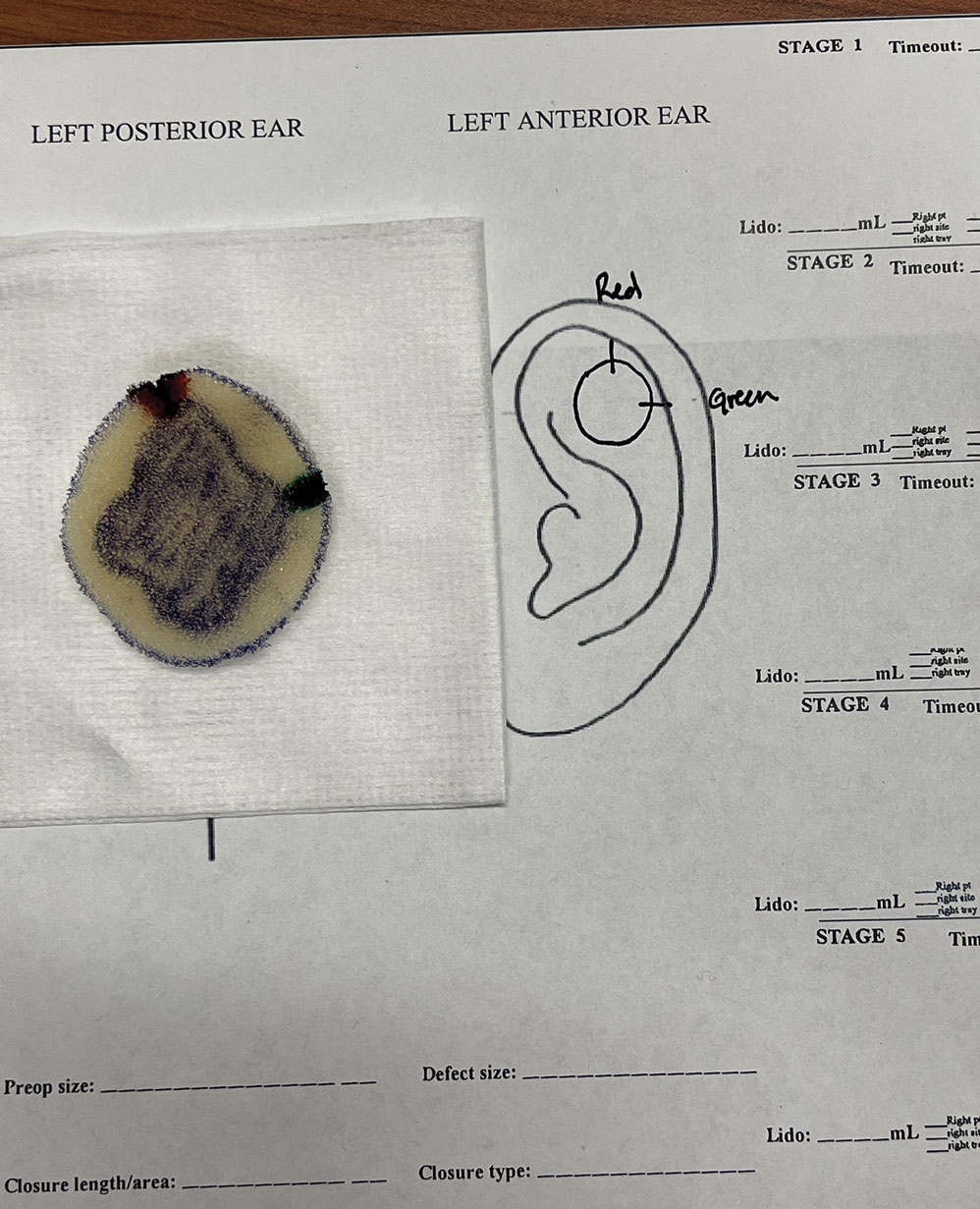
The resident then places the specimen between 2 plastic page protectors mimicking a glass slide and cover slip. Clear tape can be used to help flatten the specimen (Figure 4). The tissue is compressed between the page protector so that the simulated epidermis, dermis, and subcutaneous fat are all in the same plane. At this stage, the instructor may discuss the use of relaxing incisions, especially for deeper tissue specimens or when excision at a 45° bevel is not achieved.5 The view from the underside of the page protector reveals 100% of the specimen’s margin and mimics the first cut off the tissue block. The resident can visualize the complete circumferential, peripheral, and deep margins and can easily identify any positive margins. At this point, the exercise can conclude, or the resident can explore further stages for positive margins, bisected specimens, or other tissue preparation variations.

Practice Implications
By individually designing and removing a representative tumor with margins, creating hash marks, and preparing a tissue specimen for histologic analysis, our interactive teaching method provides dermatology residents with a relatively simple, effective, and active learning experience for MMS outside the surgical setting. Using a piece of craft foam allows the representative tissue to be manipulated and flattened, similar to cutaneous tissue. This method was implemented and refined across 3 separate teaching sessions held by teaching faculty (E.I.P and E.B.W.) at the San Antonio Uniformed Services Health Education Consortium Dermatology Residency Program (San Antonio, Texas). This method has consistently generated strong resident engagement and prompted insightful questions and discussions. Program directors at other residency programs can readily incorporate this method in their surgical curriculum by allocating a brief didactic period to the exercise and facilitating the discussion with a dermatologic surgeon. Its simplicity, low cost, and effectiveness make the foam block model an easily adoptable teaching tool for dermatology residency programs seeking to provide a comprehensive, hands-on understanding of MMS.
- McNeil E, Reich H, Hurliman E. Educational video improves dermatology residents’ understanding of Mohs micrographic surgery: a surveybased matched cohort study. J Am Acad Dermatol. 2020;83:926-927. doi:10.1016/j.jaad.2020.01.013
- Lee E, Wolverton JE, Somani AK. A simple, effective analogy to elucidate the Mohs micrographic surgery procedure—the peanut butter cup. JAMA Dermatol. 2017;153:743-744. doi:10.1001 /jamadermatol.2017.0614
- Vassantachart JM, Guccione J, Seeburger J. Clinical pearl: Mohs cantaloupe analogy for the dermatology resident. Cutis. 2018; 102:65-66.
- Stratman EJ, Vogel CA, Reck SJ, et al. Analysis of dermatology resident self-reported successful learning styles and implications for core competency curriculum development. Med Teach. 2008;30:420-425. doi:10.1080/01421590801946988
- Benedetto PX, Poblete-Lopez C. Mohs micrographic surgery technique. Dermatol Clinics. 2011;29:141-151. doi:10.1016/j.det.2011.02.002
Practice Gap
Tissue processing and complete margin assessment in Mohs micrographic surgery (MMS) are challenging concepts for residents, yet they are essential components of the dermatology residency curriculum. We propose a hands-on active teaching method using craft foam blocks to help residents master these techniques. Prior educational tools have included instructional videos1 as well as the peanut butter–cup and cantaloupe analogies.2,3 Specifically, our method utilizes inexpensive, readily available supplies that allow for repeated practice in a low-stakes environment without limitation of resources. This method provides an immersive, hands-on experience that allows residents to perform multiple practice excisions and simulate positive peripheral or deep margins, unlike tools that offer only fixed-depth or purely visual representations. Additionally, our learning model uniquely enables residents to flatten the simulated tissue, providing a clearer understanding of how a 3-dimensional specimen is transformed on a slide during histologic preparation. This step is particularly important, as tissue architecture can shift during processing, making it one of the most difficult concepts to grasp without hands-on experience. Having a multitude of teaching methods is crucial to accommodate various learning styles, and active learning has been shown to enhance retention for dermatology residents.4
The Technique
Residents use simple art supplies (including craft foam blocks and ink) and inexpensive, readily available surgical tools to simulate MMS (Table)(Figure 1). If desired, the resident can follow along with the comprehensive, stepwise textbook description of MMS, outlined by Benedetto et al5 to contextualize this hands-on exercise within a standardized didactic framework.


The foam block, which represents patient tissue, serves as the specimen. The resident begins by freehand drawing a simulated cutaneous tumor directly onto the foam using a surgical marking pen. At this point, the instructor discusses the advantages and limitations of tumor debulking with a sharp blade or curette. Residents then mark appropriate margins (1-3 mm) of normal-appearing “epidermis” on the foam block and add hash marks for orientation. This is another opportunity for the instructor to discuss common methods for marking tissue in vivo and to review situations when larger or smaller margins might be appropriate.
Next, the resident removes the first layer of simulated tissue using a disposable #15 blade scalpel at a 45° angle circumferentially and deep around the representative tumor. The resident also may use scissors and tissue forceps to remove the representative tumor. Next, the excised foam layer (the simulated “specimen”) is transferred to gauze. To demonstrate a positive margin, the resident or instructor marks the deep or peripheral foam block with a surgical marking pen, indicating residual tumor (Figure 2). This allows for multiple sequential layers of foam to be removed, demonstrating successive stages of MMS.

An inkwell holds different colors of washable paint to simulate tissue inking. After excision, the resident uses cotton-tipped applicators to apply different paint colors to the edges of the excised foam specimen at designated orientation points (eg, 3 o'clock and 12 o'clock). The resident then records the location of the excised sample by hand-drawing it on a printable Mohs map, labeling the corresponding paint colors to indicate orientation (Figure 3).

The resident then places the specimen between 2 plastic page protectors mimicking a glass slide and cover slip. Clear tape can be used to help flatten the specimen (Figure 4). The tissue is compressed between the page protector so that the simulated epidermis, dermis, and subcutaneous fat are all in the same plane. At this stage, the instructor may discuss the use of relaxing incisions, especially for deeper tissue specimens or when excision at a 45° bevel is not achieved.5 The view from the underside of the page protector reveals 100% of the specimen’s margin and mimics the first cut off the tissue block. The resident can visualize the complete circumferential, peripheral, and deep margins and can easily identify any positive margins. At this point, the exercise can conclude, or the resident can explore further stages for positive margins, bisected specimens, or other tissue preparation variations.

Practice Implications
By individually designing and removing a representative tumor with margins, creating hash marks, and preparing a tissue specimen for histologic analysis, our interactive teaching method provides dermatology residents with a relatively simple, effective, and active learning experience for MMS outside the surgical setting. Using a piece of craft foam allows the representative tissue to be manipulated and flattened, similar to cutaneous tissue. This method was implemented and refined across 3 separate teaching sessions held by teaching faculty (E.I.P and E.B.W.) at the San Antonio Uniformed Services Health Education Consortium Dermatology Residency Program (San Antonio, Texas). This method has consistently generated strong resident engagement and prompted insightful questions and discussions. Program directors at other residency programs can readily incorporate this method in their surgical curriculum by allocating a brief didactic period to the exercise and facilitating the discussion with a dermatologic surgeon. Its simplicity, low cost, and effectiveness make the foam block model an easily adoptable teaching tool for dermatology residency programs seeking to provide a comprehensive, hands-on understanding of MMS.
Practice Gap
Tissue processing and complete margin assessment in Mohs micrographic surgery (MMS) are challenging concepts for residents, yet they are essential components of the dermatology residency curriculum. We propose a hands-on active teaching method using craft foam blocks to help residents master these techniques. Prior educational tools have included instructional videos1 as well as the peanut butter–cup and cantaloupe analogies.2,3 Specifically, our method utilizes inexpensive, readily available supplies that allow for repeated practice in a low-stakes environment without limitation of resources. This method provides an immersive, hands-on experience that allows residents to perform multiple practice excisions and simulate positive peripheral or deep margins, unlike tools that offer only fixed-depth or purely visual representations. Additionally, our learning model uniquely enables residents to flatten the simulated tissue, providing a clearer understanding of how a 3-dimensional specimen is transformed on a slide during histologic preparation. This step is particularly important, as tissue architecture can shift during processing, making it one of the most difficult concepts to grasp without hands-on experience. Having a multitude of teaching methods is crucial to accommodate various learning styles, and active learning has been shown to enhance retention for dermatology residents.4
The Technique
Residents use simple art supplies (including craft foam blocks and ink) and inexpensive, readily available surgical tools to simulate MMS (Table)(Figure 1). If desired, the resident can follow along with the comprehensive, stepwise textbook description of MMS, outlined by Benedetto et al5 to contextualize this hands-on exercise within a standardized didactic framework.


The foam block, which represents patient tissue, serves as the specimen. The resident begins by freehand drawing a simulated cutaneous tumor directly onto the foam using a surgical marking pen. At this point, the instructor discusses the advantages and limitations of tumor debulking with a sharp blade or curette. Residents then mark appropriate margins (1-3 mm) of normal-appearing “epidermis” on the foam block and add hash marks for orientation. This is another opportunity for the instructor to discuss common methods for marking tissue in vivo and to review situations when larger or smaller margins might be appropriate.
Next, the resident removes the first layer of simulated tissue using a disposable #15 blade scalpel at a 45° angle circumferentially and deep around the representative tumor. The resident also may use scissors and tissue forceps to remove the representative tumor. Next, the excised foam layer (the simulated “specimen”) is transferred to gauze. To demonstrate a positive margin, the resident or instructor marks the deep or peripheral foam block with a surgical marking pen, indicating residual tumor (Figure 2). This allows for multiple sequential layers of foam to be removed, demonstrating successive stages of MMS.

An inkwell holds different colors of washable paint to simulate tissue inking. After excision, the resident uses cotton-tipped applicators to apply different paint colors to the edges of the excised foam specimen at designated orientation points (eg, 3 o'clock and 12 o'clock). The resident then records the location of the excised sample by hand-drawing it on a printable Mohs map, labeling the corresponding paint colors to indicate orientation (Figure 3).

The resident then places the specimen between 2 plastic page protectors mimicking a glass slide and cover slip. Clear tape can be used to help flatten the specimen (Figure 4). The tissue is compressed between the page protector so that the simulated epidermis, dermis, and subcutaneous fat are all in the same plane. At this stage, the instructor may discuss the use of relaxing incisions, especially for deeper tissue specimens or when excision at a 45° bevel is not achieved.5 The view from the underside of the page protector reveals 100% of the specimen’s margin and mimics the first cut off the tissue block. The resident can visualize the complete circumferential, peripheral, and deep margins and can easily identify any positive margins. At this point, the exercise can conclude, or the resident can explore further stages for positive margins, bisected specimens, or other tissue preparation variations.

Practice Implications
By individually designing and removing a representative tumor with margins, creating hash marks, and preparing a tissue specimen for histologic analysis, our interactive teaching method provides dermatology residents with a relatively simple, effective, and active learning experience for MMS outside the surgical setting. Using a piece of craft foam allows the representative tissue to be manipulated and flattened, similar to cutaneous tissue. This method was implemented and refined across 3 separate teaching sessions held by teaching faculty (E.I.P and E.B.W.) at the San Antonio Uniformed Services Health Education Consortium Dermatology Residency Program (San Antonio, Texas). This method has consistently generated strong resident engagement and prompted insightful questions and discussions. Program directors at other residency programs can readily incorporate this method in their surgical curriculum by allocating a brief didactic period to the exercise and facilitating the discussion with a dermatologic surgeon. Its simplicity, low cost, and effectiveness make the foam block model an easily adoptable teaching tool for dermatology residency programs seeking to provide a comprehensive, hands-on understanding of MMS.
- McNeil E, Reich H, Hurliman E. Educational video improves dermatology residents’ understanding of Mohs micrographic surgery: a surveybased matched cohort study. J Am Acad Dermatol. 2020;83:926-927. doi:10.1016/j.jaad.2020.01.013
- Lee E, Wolverton JE, Somani AK. A simple, effective analogy to elucidate the Mohs micrographic surgery procedure—the peanut butter cup. JAMA Dermatol. 2017;153:743-744. doi:10.1001 /jamadermatol.2017.0614
- Vassantachart JM, Guccione J, Seeburger J. Clinical pearl: Mohs cantaloupe analogy for the dermatology resident. Cutis. 2018; 102:65-66.
- Stratman EJ, Vogel CA, Reck SJ, et al. Analysis of dermatology resident self-reported successful learning styles and implications for core competency curriculum development. Med Teach. 2008;30:420-425. doi:10.1080/01421590801946988
- Benedetto PX, Poblete-Lopez C. Mohs micrographic surgery technique. Dermatol Clinics. 2011;29:141-151. doi:10.1016/j.det.2011.02.002
- McNeil E, Reich H, Hurliman E. Educational video improves dermatology residents’ understanding of Mohs micrographic surgery: a surveybased matched cohort study. J Am Acad Dermatol. 2020;83:926-927. doi:10.1016/j.jaad.2020.01.013
- Lee E, Wolverton JE, Somani AK. A simple, effective analogy to elucidate the Mohs micrographic surgery procedure—the peanut butter cup. JAMA Dermatol. 2017;153:743-744. doi:10.1001 /jamadermatol.2017.0614
- Vassantachart JM, Guccione J, Seeburger J. Clinical pearl: Mohs cantaloupe analogy for the dermatology resident. Cutis. 2018; 102:65-66.
- Stratman EJ, Vogel CA, Reck SJ, et al. Analysis of dermatology resident self-reported successful learning styles and implications for core competency curriculum development. Med Teach. 2008;30:420-425. doi:10.1080/01421590801946988
- Benedetto PX, Poblete-Lopez C. Mohs micrographic surgery technique. Dermatol Clinics. 2011;29:141-151. doi:10.1016/j.det.2011.02.002
Interactive Approach to Teaching Mohs Micrographic Surgery to Dermatology Residents
Interactive Approach to Teaching Mohs Micrographic Surgery to Dermatology Residents
Poly-L-Lactic Acid Reconstitution Technique to Reduce Needle Obstruction
Poly-L-Lactic Acid Reconstitution Technique to Reduce Needle Obstruction
Practice Gap
Poly-L-lactic acid is approved by the US Food and Drug Administration for addressing fat loss due to HAART in patients with HIV.2,3 When used as a dermal filler for correction of facial lipoatrophy, PLLA is well tolerated and has been shown to improve quality of life.2,3 Poly-L-lactic acid is available for clinical use as microparticles of lyophilized alpha hydroxy acid polymers. Once injected (after the carrier substance is absorbed), PLLA induces an inflammatory response that ultimately leads to the production of new collagen.3 Unfortunately, PLLA microparticles often obstruct needles and make the product difficult to use, potentially hindering effective injection; thus, it is in the best interest of the patient to mitigate needle obstruction during this procedure. In this article, we describe a simple and effective way to mitigate this problem by utilizing a water bath to warm the filler prior to injection.
Technique
The required supplies include a thermostatic water bath, reconstituted PLLA, a syringe, and a 26-gauge injection needle. Because laboratory-grade heated water baths typically cost between $300 and $3000,4 we recommend using a more affordable, commercially available thermostatic water bath (eg, baby bottle warmer)(Figure 1) to warm the filler prior to injection, as the optimal temperature for this technique can still be achieved while remaining cost effective. Vials of PLLA reconstituted with 7 mL of sterile water and 2 mL lidocaine hydrochloride 1% should be labeled with the date of reconstitution and manually agitated for 30 seconds. The reconstituted product should be stored for 24 hours to ensure even suspension and powder saturation.5 On the day of the procedure, the vial should be placed into the water bath (heated to 100 °C) for 10 minutes prior to injection (Figure 2) and agitated again immediately before withdrawal into the syringe. The clinician then should sterilize the rubber top and draw the product from the warmed vial using the same size needle that will be used for injection. Although a larger gauge needle may make drawing up the product easier in typical practice, drawing and injecting with the same gauge needle helps prevent larger particles from clogging a smaller injection needle. Using a 26-gauge injection needle for withdrawal further reduces clogging by serving as a filter to prevent larger product particles from entering the injection syringe. The vials of PLLA can be kept in the water bath throughout the procedure between uses to keep the filler at a consistent temperature.

Practice Implications
Although many clinicians reduce needle obstructions by warming PLLA before injection, a published protocol currently is not available. One consideration when utilizing this technique is the limited data on the clinical stability and efficacy of PLLA at varying temperatures. Two studies recommend bringing the reconstituted vial to room temperature prior to injection, while others have documented an endothermic melting point in the range of 120 °C to 180
- James J, Carruthers A, Carruthers J. HIV-associated facial lipoatrophy. Dermatol Surg. 2002;28:979-986. doi:10.1046/j.1524-4725.2002.02099.x
- Duracinsky M, Leclercq P, Herrmann S, et al. Safety of poly-L-lactic acid (New-Fill®) in the treatment of facial lipoatrophy: a large observational study among HIV-positive patients. BMC Infect Dis. 2014;14:474. doi:10.1186/1471-2334-14-474
- Sickles CK, Nassereddin A, Patel P, et al. Poly-L-lactic acid. StatPearls [Internet]. Updated February 28, 2024. Accessed October 31, 2025. https://www.ncbi.nlm.nih.gov/books/NBK507871/
- Laboratory equipment: Water bath. Global Lab Supply. (n.d.). http://www.globallabsupply.com/Water-Bath-s/2122.htm
- Lin MJ, Dubin DP, Goldberg DJ, et al. Practices in the usage and reconstitution of poly-L-lactic acid. J Drugs Dermatol. 2019;18:880-886.
- Vleggaar D, Fitzgerald R, Lorenc ZP, et al. Consensus recommendations on the use of injectable poly-L-lactic acid for facial and nonfacial volumization. J Drugs Dermatol. 2014;13:s44-51.
- Sedush NG, Kalinin KT, Azarkevich PN, et al. Physicochemical characteristics and hydrolytic degradation of polylactic acid dermal fillers: a comparative study. Cosmetics. 2023;10:110. doi:10.3390/cosmetics10040110
Practice Gap
Poly-L-lactic acid is approved by the US Food and Drug Administration for addressing fat loss due to HAART in patients with HIV.2,3 When used as a dermal filler for correction of facial lipoatrophy, PLLA is well tolerated and has been shown to improve quality of life.2,3 Poly-L-lactic acid is available for clinical use as microparticles of lyophilized alpha hydroxy acid polymers. Once injected (after the carrier substance is absorbed), PLLA induces an inflammatory response that ultimately leads to the production of new collagen.3 Unfortunately, PLLA microparticles often obstruct needles and make the product difficult to use, potentially hindering effective injection; thus, it is in the best interest of the patient to mitigate needle obstruction during this procedure. In this article, we describe a simple and effective way to mitigate this problem by utilizing a water bath to warm the filler prior to injection.
Technique
The required supplies include a thermostatic water bath, reconstituted PLLA, a syringe, and a 26-gauge injection needle. Because laboratory-grade heated water baths typically cost between $300 and $3000,4 we recommend using a more affordable, commercially available thermostatic water bath (eg, baby bottle warmer)(Figure 1) to warm the filler prior to injection, as the optimal temperature for this technique can still be achieved while remaining cost effective. Vials of PLLA reconstituted with 7 mL of sterile water and 2 mL lidocaine hydrochloride 1% should be labeled with the date of reconstitution and manually agitated for 30 seconds. The reconstituted product should be stored for 24 hours to ensure even suspension and powder saturation.5 On the day of the procedure, the vial should be placed into the water bath (heated to 100 °C) for 10 minutes prior to injection (Figure 2) and agitated again immediately before withdrawal into the syringe. The clinician then should sterilize the rubber top and draw the product from the warmed vial using the same size needle that will be used for injection. Although a larger gauge needle may make drawing up the product easier in typical practice, drawing and injecting with the same gauge needle helps prevent larger particles from clogging a smaller injection needle. Using a 26-gauge injection needle for withdrawal further reduces clogging by serving as a filter to prevent larger product particles from entering the injection syringe. The vials of PLLA can be kept in the water bath throughout the procedure between uses to keep the filler at a consistent temperature.


Practice Implications
Although many clinicians reduce needle obstructions by warming PLLA before injection, a published protocol currently is not available. One consideration when utilizing this technique is the limited data on the clinical stability and efficacy of PLLA at varying temperatures. Two studies recommend bringing the reconstituted vial to room temperature prior to injection, while others have documented an endothermic melting point in the range of 120 °C to 180
Practice Gap
Poly-L-lactic acid is approved by the US Food and Drug Administration for addressing fat loss due to HAART in patients with HIV.2,3 When used as a dermal filler for correction of facial lipoatrophy, PLLA is well tolerated and has been shown to improve quality of life.2,3 Poly-L-lactic acid is available for clinical use as microparticles of lyophilized alpha hydroxy acid polymers. Once injected (after the carrier substance is absorbed), PLLA induces an inflammatory response that ultimately leads to the production of new collagen.3 Unfortunately, PLLA microparticles often obstruct needles and make the product difficult to use, potentially hindering effective injection; thus, it is in the best interest of the patient to mitigate needle obstruction during this procedure. In this article, we describe a simple and effective way to mitigate this problem by utilizing a water bath to warm the filler prior to injection.
Technique
The required supplies include a thermostatic water bath, reconstituted PLLA, a syringe, and a 26-gauge injection needle. Because laboratory-grade heated water baths typically cost between $300 and $3000,4 we recommend using a more affordable, commercially available thermostatic water bath (eg, baby bottle warmer)(Figure 1) to warm the filler prior to injection, as the optimal temperature for this technique can still be achieved while remaining cost effective. Vials of PLLA reconstituted with 7 mL of sterile water and 2 mL lidocaine hydrochloride 1% should be labeled with the date of reconstitution and manually agitated for 30 seconds. The reconstituted product should be stored for 24 hours to ensure even suspension and powder saturation.5 On the day of the procedure, the vial should be placed into the water bath (heated to 100 °C) for 10 minutes prior to injection (Figure 2) and agitated again immediately before withdrawal into the syringe. The clinician then should sterilize the rubber top and draw the product from the warmed vial using the same size needle that will be used for injection. Although a larger gauge needle may make drawing up the product easier in typical practice, drawing and injecting with the same gauge needle helps prevent larger particles from clogging a smaller injection needle. Using a 26-gauge injection needle for withdrawal further reduces clogging by serving as a filter to prevent larger product particles from entering the injection syringe. The vials of PLLA can be kept in the water bath throughout the procedure between uses to keep the filler at a consistent temperature.


Practice Implications
Although many clinicians reduce needle obstructions by warming PLLA before injection, a published protocol currently is not available. One consideration when utilizing this technique is the limited data on the clinical stability and efficacy of PLLA at varying temperatures. Two studies recommend bringing the reconstituted vial to room temperature prior to injection, while others have documented an endothermic melting point in the range of 120 °C to 180
- James J, Carruthers A, Carruthers J. HIV-associated facial lipoatrophy. Dermatol Surg. 2002;28:979-986. doi:10.1046/j.1524-4725.2002.02099.x
- Duracinsky M, Leclercq P, Herrmann S, et al. Safety of poly-L-lactic acid (New-Fill®) in the treatment of facial lipoatrophy: a large observational study among HIV-positive patients. BMC Infect Dis. 2014;14:474. doi:10.1186/1471-2334-14-474
- Sickles CK, Nassereddin A, Patel P, et al. Poly-L-lactic acid. StatPearls [Internet]. Updated February 28, 2024. Accessed October 31, 2025. https://www.ncbi.nlm.nih.gov/books/NBK507871/
- Laboratory equipment: Water bath. Global Lab Supply. (n.d.). http://www.globallabsupply.com/Water-Bath-s/2122.htm
- Lin MJ, Dubin DP, Goldberg DJ, et al. Practices in the usage and reconstitution of poly-L-lactic acid. J Drugs Dermatol. 2019;18:880-886.
- Vleggaar D, Fitzgerald R, Lorenc ZP, et al. Consensus recommendations on the use of injectable poly-L-lactic acid for facial and nonfacial volumization. J Drugs Dermatol. 2014;13:s44-51.
- Sedush NG, Kalinin KT, Azarkevich PN, et al. Physicochemical characteristics and hydrolytic degradation of polylactic acid dermal fillers: a comparative study. Cosmetics. 2023;10:110. doi:10.3390/cosmetics10040110
- James J, Carruthers A, Carruthers J. HIV-associated facial lipoatrophy. Dermatol Surg. 2002;28:979-986. doi:10.1046/j.1524-4725.2002.02099.x
- Duracinsky M, Leclercq P, Herrmann S, et al. Safety of poly-L-lactic acid (New-Fill®) in the treatment of facial lipoatrophy: a large observational study among HIV-positive patients. BMC Infect Dis. 2014;14:474. doi:10.1186/1471-2334-14-474
- Sickles CK, Nassereddin A, Patel P, et al. Poly-L-lactic acid. StatPearls [Internet]. Updated February 28, 2024. Accessed October 31, 2025. https://www.ncbi.nlm.nih.gov/books/NBK507871/
- Laboratory equipment: Water bath. Global Lab Supply. (n.d.). http://www.globallabsupply.com/Water-Bath-s/2122.htm
- Lin MJ, Dubin DP, Goldberg DJ, et al. Practices in the usage and reconstitution of poly-L-lactic acid. J Drugs Dermatol. 2019;18:880-886.
- Vleggaar D, Fitzgerald R, Lorenc ZP, et al. Consensus recommendations on the use of injectable poly-L-lactic acid for facial and nonfacial volumization. J Drugs Dermatol. 2014;13:s44-51.
- Sedush NG, Kalinin KT, Azarkevich PN, et al. Physicochemical characteristics and hydrolytic degradation of polylactic acid dermal fillers: a comparative study. Cosmetics. 2023;10:110. doi:10.3390/cosmetics10040110
Poly-L-Lactic Acid Reconstitution Technique to Reduce Needle Obstruction
Poly-L-Lactic Acid Reconstitution Technique to Reduce Needle Obstruction
Conservative Thickness Layers to Preserve Tattoo Appearance During Excisional Procedures
Conservative Thickness Layers to Preserve Tattoo Appearance During Excisional Procedures
Practice Gap
Tattoos have become increasingly prevalent in Western culture, with approximately 1 in 4 Americans having at least 1 tattoo. Individuals invest money, time, and even pain in getting tattoos, many of which hold special personal, family, or religious significance.1 Various cutaneous pathologies may arise in areas of the skin with tattoos, including malignancies and inflammatory reactions to tattoo pigment, and in these cases, surgical management may be indicated.2,3
Nonmelanoma skin cancers (NMSCs) such as superficial basal cell carcinomas on broadly sun-damaged areas (eg, trunk, torso), squamous cell carcinomas, reactive keratoacanthomas, and reactive pseudoepitheliomatous squamous hyperplasia diagnosed as squamous cell carcinoma have been reported to occur in or near areas of the skin with tattoos.2 Mohs micrographic surgery (MMS) is the standard of care for removing NMSCs, particularly when they manifest in cosmetically sensitive areas.4 This treatment option allows for careful guided resection of tumors to minimize the risk for recurrence; it also preserves healthy tissue, which typically results in a smaller radial defect after the procedure is complete.
Chronic reactions to tattoo pigment may include granulomatous tattoo reactions and pseudolymphomas.3 Treatment options may include immunosuppressives such as intralesional triamcinolone as well as pigment destruction via lasers5; however, not all tattoos are responsive to these treatments. Surgical excision is an effective and definitive treatment in this context, as tattoo pigment resides in or above the mid dermis to a depth of approximately 400 μm. Intradermal excision effectively removes the antigenic pigment.5
In these clinical scenarios, patients may be hesitant to pursue surgical treatment due to concerns that it may alter tattoo appearance. Many clinicians and surgeons may consider definitive treatment and tattoo preservation to be mutually exclusive, but this is not always the case. We propose a technique that utilizes conservative thickness layers (CTL) to minimize disruption to the appearance of tattoos in MMS for treatment of cutaneous malignancies as well as intradermal excision of tattoo pigment in the setting of chronic inflammatory tattoo reactions.
The Technique
In the appropriate clinical context, CTL can effectively result in defects that heal well by secondary intention and minimize collateral tissue distortion.4 Lesions manifesting in or near tattooed skin often are responsive to treatment with CTL; furthermore, CTL may preserve some deeper tattoo pigment, resulting in only partial loss of the tattooed skin.
Conservative thickness layers are performed intradermally, similar to removing traditional layers in MMS. For treatment of NMSCs, a margin is scored around the lesion, and then the blade is passed carefully under the lesion nearly parallel to the skin through an intradermal plane. It is important to avoid entering the subcuticular fat (Figure 1). The tissue then is processed normally in the Mohs laboratory for complete circumferential margin evaluation. If necessary and possible, subsequent layers also can be performed in the intradermal plane. Once total circumferential margin control is obtained, the wound is allowed to granulate and heal by secondary intention. As these processes occur, we have found that wound contraction is less likely with the dermis intact, resulting in less impact on the overall appearance of the tattoo (Figures 1 and 2). For very thin lesions, resultant defects may retain some residual tattoo pigment. The residual scars also may be responsive to tattoo revision, although a period of monitoring for recurrence should be considered if there is concern that revising the tattoo could obscure early recurrent tumors. From our experience, utilizing CTL for NMSCs that arise within or near tattoos results in favorable preservation of the tattoo appearance and high patient satisfaction.
The procedure is performed similarly for removal of allergenic tattoo pigment, with careful excision to the mid dermis. Since the areas affected by the cutaneous reaction may be relatively large, surgical precision is required to maintain a uniform depth to remove the tattoo pigment and preserve the deep dermis (Figure 2). Once removed, the defect can be left to granulate and heal by secondary intention. If the patient wants to have the tattoo revised in the future, it would be prudent to utilize pigment that the patient has responded favorably to. In our experience, this approach is effective and yields high patient satisfaction and minimizes morbidity.
Practice Implications
Tattoos often hold special meaning for patients; therefore, treatment of pathologies arising in or near tattooed skin should emphasize maintaining the appearance of the tattoo while still being effective. Conservative thickness layers in MMS and intradermal excisions for allergic reactions to tattoo pigment are an effective treatment strategy that clinicians may consider.
One shortcoming of using CTL for MMS is the need for subsequent layers to clear the tumor; however, data suggest that first-stage cure rates are extremely high even with CTL for appropriately selected patients, with clearance of nearly 80% of tumors on the first stage. Tumors that may be most responsive to CTL include exophytic NMSCs and those arising in areas with a thicker dermis, including the back, legs, and scalp, although other locations including the face, hands, shins, ankles, and feet also may be well suited for CTL.4 Another shortcoming of CTL is that skin cancers arising in tattoos may not be considered appropriate for MMS based on the 2012
Conservative thickness layers in MMS and intradermal excisions of tattoo pigment are both effective techniques of minimizing disruption of tattoos while effectively treating patients.
- Roggenkamp H, Nicholls A, Pierre JM. Tattoos as a window to the psyche: how talking about skin art can inform psychiatric practice. World J Psychiatry. 2017;7:148-158. doi:10.5498/wjp.v7.i3.148
- Rubatto M, Gelato F, Mastorino L, et al. Nonmelanoma skin cancer arising on tattoos. Int J Dermatol. 2023;62:E155-E156. doi:10.1111/ijd.16381
- Atwater AR, Bembry R, Reeder M. Tattoo hypersensitivity reactions: inky business. Cutis. 2020;106:64-67. doi:10.12788/cutis.0028
- Tolkachjov SN, Cappel JA, Bryant EA, et al. Conservative thickness layers in Mohs micrographic surgery. Int J Dermatol. 2018;57:1128-1134. doi:10.1111/ijd.14043
- Sardana K, Ranjan R, Ghunawat S. Optimising laser tattoo removal. J Cutan Aesthet Surg. 2015;8:16-24. doi:10.4103/0974-2077.155068
- Connolly SM, Baker DR, Coldiron BM, et al. AAD/ACMS/ASDSA/ASMS 2012 appropriate use criteria for Mohs micrographic surgery: a report of the American Academy of Dermatology, American College of Mohs Surgery, American Society for Dermatologic Surgery Association, and the American Society for Mohs Surgery. J Am Acad Dermatol. 2012;67:531-550. doi:10.1016/j.jaad.2012.06.009.
- Amthor Croley JA. Current controversies in mohs micrographic surgery. Cutis. 2019;104:E29-E31.
Practice Gap
Tattoos have become increasingly prevalent in Western culture, with approximately 1 in 4 Americans having at least 1 tattoo. Individuals invest money, time, and even pain in getting tattoos, many of which hold special personal, family, or religious significance.1 Various cutaneous pathologies may arise in areas of the skin with tattoos, including malignancies and inflammatory reactions to tattoo pigment, and in these cases, surgical management may be indicated.2,3
Nonmelanoma skin cancers (NMSCs) such as superficial basal cell carcinomas on broadly sun-damaged areas (eg, trunk, torso), squamous cell carcinomas, reactive keratoacanthomas, and reactive pseudoepitheliomatous squamous hyperplasia diagnosed as squamous cell carcinoma have been reported to occur in or near areas of the skin with tattoos.2 Mohs micrographic surgery (MMS) is the standard of care for removing NMSCs, particularly when they manifest in cosmetically sensitive areas.4 This treatment option allows for careful guided resection of tumors to minimize the risk for recurrence; it also preserves healthy tissue, which typically results in a smaller radial defect after the procedure is complete.
Chronic reactions to tattoo pigment may include granulomatous tattoo reactions and pseudolymphomas.3 Treatment options may include immunosuppressives such as intralesional triamcinolone as well as pigment destruction via lasers5; however, not all tattoos are responsive to these treatments. Surgical excision is an effective and definitive treatment in this context, as tattoo pigment resides in or above the mid dermis to a depth of approximately 400 μm. Intradermal excision effectively removes the antigenic pigment.5
In these clinical scenarios, patients may be hesitant to pursue surgical treatment due to concerns that it may alter tattoo appearance. Many clinicians and surgeons may consider definitive treatment and tattoo preservation to be mutually exclusive, but this is not always the case. We propose a technique that utilizes conservative thickness layers (CTL) to minimize disruption to the appearance of tattoos in MMS for treatment of cutaneous malignancies as well as intradermal excision of tattoo pigment in the setting of chronic inflammatory tattoo reactions.
The Technique
In the appropriate clinical context, CTL can effectively result in defects that heal well by secondary intention and minimize collateral tissue distortion.4 Lesions manifesting in or near tattooed skin often are responsive to treatment with CTL; furthermore, CTL may preserve some deeper tattoo pigment, resulting in only partial loss of the tattooed skin.
Conservative thickness layers are performed intradermally, similar to removing traditional layers in MMS. For treatment of NMSCs, a margin is scored around the lesion, and then the blade is passed carefully under the lesion nearly parallel to the skin through an intradermal plane. It is important to avoid entering the subcuticular fat (Figure 1). The tissue then is processed normally in the Mohs laboratory for complete circumferential margin evaluation. If necessary and possible, subsequent layers also can be performed in the intradermal plane. Once total circumferential margin control is obtained, the wound is allowed to granulate and heal by secondary intention. As these processes occur, we have found that wound contraction is less likely with the dermis intact, resulting in less impact on the overall appearance of the tattoo (Figures 1 and 2). For very thin lesions, resultant defects may retain some residual tattoo pigment. The residual scars also may be responsive to tattoo revision, although a period of monitoring for recurrence should be considered if there is concern that revising the tattoo could obscure early recurrent tumors. From our experience, utilizing CTL for NMSCs that arise within or near tattoos results in favorable preservation of the tattoo appearance and high patient satisfaction.


The procedure is performed similarly for removal of allergenic tattoo pigment, with careful excision to the mid dermis. Since the areas affected by the cutaneous reaction may be relatively large, surgical precision is required to maintain a uniform depth to remove the tattoo pigment and preserve the deep dermis (Figure 2). Once removed, the defect can be left to granulate and heal by secondary intention. If the patient wants to have the tattoo revised in the future, it would be prudent to utilize pigment that the patient has responded favorably to. In our experience, this approach is effective and yields high patient satisfaction and minimizes morbidity.
Practice Implications
Tattoos often hold special meaning for patients; therefore, treatment of pathologies arising in or near tattooed skin should emphasize maintaining the appearance of the tattoo while still being effective. Conservative thickness layers in MMS and intradermal excisions for allergic reactions to tattoo pigment are an effective treatment strategy that clinicians may consider.
One shortcoming of using CTL for MMS is the need for subsequent layers to clear the tumor; however, data suggest that first-stage cure rates are extremely high even with CTL for appropriately selected patients, with clearance of nearly 80% of tumors on the first stage. Tumors that may be most responsive to CTL include exophytic NMSCs and those arising in areas with a thicker dermis, including the back, legs, and scalp, although other locations including the face, hands, shins, ankles, and feet also may be well suited for CTL.4 Another shortcoming of CTL is that skin cancers arising in tattoos may not be considered appropriate for MMS based on the 2012
Conservative thickness layers in MMS and intradermal excisions of tattoo pigment are both effective techniques of minimizing disruption of tattoos while effectively treating patients.
Practice Gap
Tattoos have become increasingly prevalent in Western culture, with approximately 1 in 4 Americans having at least 1 tattoo. Individuals invest money, time, and even pain in getting tattoos, many of which hold special personal, family, or religious significance.1 Various cutaneous pathologies may arise in areas of the skin with tattoos, including malignancies and inflammatory reactions to tattoo pigment, and in these cases, surgical management may be indicated.2,3
Nonmelanoma skin cancers (NMSCs) such as superficial basal cell carcinomas on broadly sun-damaged areas (eg, trunk, torso), squamous cell carcinomas, reactive keratoacanthomas, and reactive pseudoepitheliomatous squamous hyperplasia diagnosed as squamous cell carcinoma have been reported to occur in or near areas of the skin with tattoos.2 Mohs micrographic surgery (MMS) is the standard of care for removing NMSCs, particularly when they manifest in cosmetically sensitive areas.4 This treatment option allows for careful guided resection of tumors to minimize the risk for recurrence; it also preserves healthy tissue, which typically results in a smaller radial defect after the procedure is complete.
Chronic reactions to tattoo pigment may include granulomatous tattoo reactions and pseudolymphomas.3 Treatment options may include immunosuppressives such as intralesional triamcinolone as well as pigment destruction via lasers5; however, not all tattoos are responsive to these treatments. Surgical excision is an effective and definitive treatment in this context, as tattoo pigment resides in or above the mid dermis to a depth of approximately 400 μm. Intradermal excision effectively removes the antigenic pigment.5
In these clinical scenarios, patients may be hesitant to pursue surgical treatment due to concerns that it may alter tattoo appearance. Many clinicians and surgeons may consider definitive treatment and tattoo preservation to be mutually exclusive, but this is not always the case. We propose a technique that utilizes conservative thickness layers (CTL) to minimize disruption to the appearance of tattoos in MMS for treatment of cutaneous malignancies as well as intradermal excision of tattoo pigment in the setting of chronic inflammatory tattoo reactions.
The Technique
In the appropriate clinical context, CTL can effectively result in defects that heal well by secondary intention and minimize collateral tissue distortion.4 Lesions manifesting in or near tattooed skin often are responsive to treatment with CTL; furthermore, CTL may preserve some deeper tattoo pigment, resulting in only partial loss of the tattooed skin.
Conservative thickness layers are performed intradermally, similar to removing traditional layers in MMS. For treatment of NMSCs, a margin is scored around the lesion, and then the blade is passed carefully under the lesion nearly parallel to the skin through an intradermal plane. It is important to avoid entering the subcuticular fat (Figure 1). The tissue then is processed normally in the Mohs laboratory for complete circumferential margin evaluation. If necessary and possible, subsequent layers also can be performed in the intradermal plane. Once total circumferential margin control is obtained, the wound is allowed to granulate and heal by secondary intention. As these processes occur, we have found that wound contraction is less likely with the dermis intact, resulting in less impact on the overall appearance of the tattoo (Figures 1 and 2). For very thin lesions, resultant defects may retain some residual tattoo pigment. The residual scars also may be responsive to tattoo revision, although a period of monitoring for recurrence should be considered if there is concern that revising the tattoo could obscure early recurrent tumors. From our experience, utilizing CTL for NMSCs that arise within or near tattoos results in favorable preservation of the tattoo appearance and high patient satisfaction.


The procedure is performed similarly for removal of allergenic tattoo pigment, with careful excision to the mid dermis. Since the areas affected by the cutaneous reaction may be relatively large, surgical precision is required to maintain a uniform depth to remove the tattoo pigment and preserve the deep dermis (Figure 2). Once removed, the defect can be left to granulate and heal by secondary intention. If the patient wants to have the tattoo revised in the future, it would be prudent to utilize pigment that the patient has responded favorably to. In our experience, this approach is effective and yields high patient satisfaction and minimizes morbidity.
Practice Implications
Tattoos often hold special meaning for patients; therefore, treatment of pathologies arising in or near tattooed skin should emphasize maintaining the appearance of the tattoo while still being effective. Conservative thickness layers in MMS and intradermal excisions for allergic reactions to tattoo pigment are an effective treatment strategy that clinicians may consider.
One shortcoming of using CTL for MMS is the need for subsequent layers to clear the tumor; however, data suggest that first-stage cure rates are extremely high even with CTL for appropriately selected patients, with clearance of nearly 80% of tumors on the first stage. Tumors that may be most responsive to CTL include exophytic NMSCs and those arising in areas with a thicker dermis, including the back, legs, and scalp, although other locations including the face, hands, shins, ankles, and feet also may be well suited for CTL.4 Another shortcoming of CTL is that skin cancers arising in tattoos may not be considered appropriate for MMS based on the 2012
Conservative thickness layers in MMS and intradermal excisions of tattoo pigment are both effective techniques of minimizing disruption of tattoos while effectively treating patients.
- Roggenkamp H, Nicholls A, Pierre JM. Tattoos as a window to the psyche: how talking about skin art can inform psychiatric practice. World J Psychiatry. 2017;7:148-158. doi:10.5498/wjp.v7.i3.148
- Rubatto M, Gelato F, Mastorino L, et al. Nonmelanoma skin cancer arising on tattoos. Int J Dermatol. 2023;62:E155-E156. doi:10.1111/ijd.16381
- Atwater AR, Bembry R, Reeder M. Tattoo hypersensitivity reactions: inky business. Cutis. 2020;106:64-67. doi:10.12788/cutis.0028
- Tolkachjov SN, Cappel JA, Bryant EA, et al. Conservative thickness layers in Mohs micrographic surgery. Int J Dermatol. 2018;57:1128-1134. doi:10.1111/ijd.14043
- Sardana K, Ranjan R, Ghunawat S. Optimising laser tattoo removal. J Cutan Aesthet Surg. 2015;8:16-24. doi:10.4103/0974-2077.155068
- Connolly SM, Baker DR, Coldiron BM, et al. AAD/ACMS/ASDSA/ASMS 2012 appropriate use criteria for Mohs micrographic surgery: a report of the American Academy of Dermatology, American College of Mohs Surgery, American Society for Dermatologic Surgery Association, and the American Society for Mohs Surgery. J Am Acad Dermatol. 2012;67:531-550. doi:10.1016/j.jaad.2012.06.009.
- Amthor Croley JA. Current controversies in mohs micrographic surgery. Cutis. 2019;104:E29-E31.
- Roggenkamp H, Nicholls A, Pierre JM. Tattoos as a window to the psyche: how talking about skin art can inform psychiatric practice. World J Psychiatry. 2017;7:148-158. doi:10.5498/wjp.v7.i3.148
- Rubatto M, Gelato F, Mastorino L, et al. Nonmelanoma skin cancer arising on tattoos. Int J Dermatol. 2023;62:E155-E156. doi:10.1111/ijd.16381
- Atwater AR, Bembry R, Reeder M. Tattoo hypersensitivity reactions: inky business. Cutis. 2020;106:64-67. doi:10.12788/cutis.0028
- Tolkachjov SN, Cappel JA, Bryant EA, et al. Conservative thickness layers in Mohs micrographic surgery. Int J Dermatol. 2018;57:1128-1134. doi:10.1111/ijd.14043
- Sardana K, Ranjan R, Ghunawat S. Optimising laser tattoo removal. J Cutan Aesthet Surg. 2015;8:16-24. doi:10.4103/0974-2077.155068
- Connolly SM, Baker DR, Coldiron BM, et al. AAD/ACMS/ASDSA/ASMS 2012 appropriate use criteria for Mohs micrographic surgery: a report of the American Academy of Dermatology, American College of Mohs Surgery, American Society for Dermatologic Surgery Association, and the American Society for Mohs Surgery. J Am Acad Dermatol. 2012;67:531-550. doi:10.1016/j.jaad.2012.06.009.
- Amthor Croley JA. Current controversies in mohs micrographic surgery. Cutis. 2019;104:E29-E31.
Conservative Thickness Layers to Preserve Tattoo Appearance During Excisional Procedures
Conservative Thickness Layers to Preserve Tattoo Appearance During Excisional Procedures
Botulinum Toxin as a Tool to Reduce Hyperhidrosis in Amputees
Botulinum Toxin as a Tool to Reduce Hyperhidrosis in Amputees
Practice Gap
Hyperhidrosis poses a considerable challenge for many amputees who use prosthetic devices, particularly at the interface between the residual limb and the prosthetic socket. The enclosed environment of the socket often leads to excessive sweating, which can compromise suction fit and increase the risk for skin chafing, irritation, and slippage. Persistent moisture also promotes bacterial and fungal growth, raising the likelihood of infections and foul odors within the socket. Research has shown that skin complications are highly prevalent among amputees, affecting up to 73.9% of this population in the United States.1 Commonly reported complications include wounds, abscesses, and blisters, many of which can be triggered or worsened by hyperhidrosis.2 Current treatment options for residual limb sweating include topical antiperspirants, botulinum toxin (BTX) injections, iontophoresis, and liner-liner socks.
While BTX commonly is used to treat hyperhidrosis in areas such as the palms and axillae, it typically is not considered as a first-line therapy for residual limb sweating; however, both BTX type A and type B have shown safety and effectiveness in managing hyperhidrosis in amputees, enhancing prosthetic use, and improving overall quality of life.3 Despite these benefits, BTX remains relatively underutilized for
Tools and Techniques
A 64-year-old man initially presented to our dermatology clinic after undergoing an above-the-knee amputation of the left leg 1 year prior. The amputation had been performed due to chronic prosthetic joint infections with Escherichia coli. He reported persistent sweating of the residual limb, which severely limited his use of a prosthesis and led to frequent falls.
During the initial visit, treatment options for primary hyperhidrosis including topical and injectable therapies were discussed. Due to a fear of needles, the patient chose topical treatment, with the option to pursue BTX injections later if better control was needed. An aluminum chloride hexahydrate prosthetic antiperspirant was prescribed for nightly application on the anterior and posterior
Botulinum toxin injections were administered in a grid-like pattern across the surface area where the residual limb made contact with the prosthetic. Using a surgical marker, the patient assisted the medical team in identifying the areas where sweating occurred most frequently. The area was divided into 4 equal sections, with each section treated per weekly interval sequentially over 4 weeks. The targeted areas included the left anterior (extending from the anterior tensor fasciae latae band to the lateral thigh) and left posterior residual limb (Figure 1 and eFigure 1, respectively).
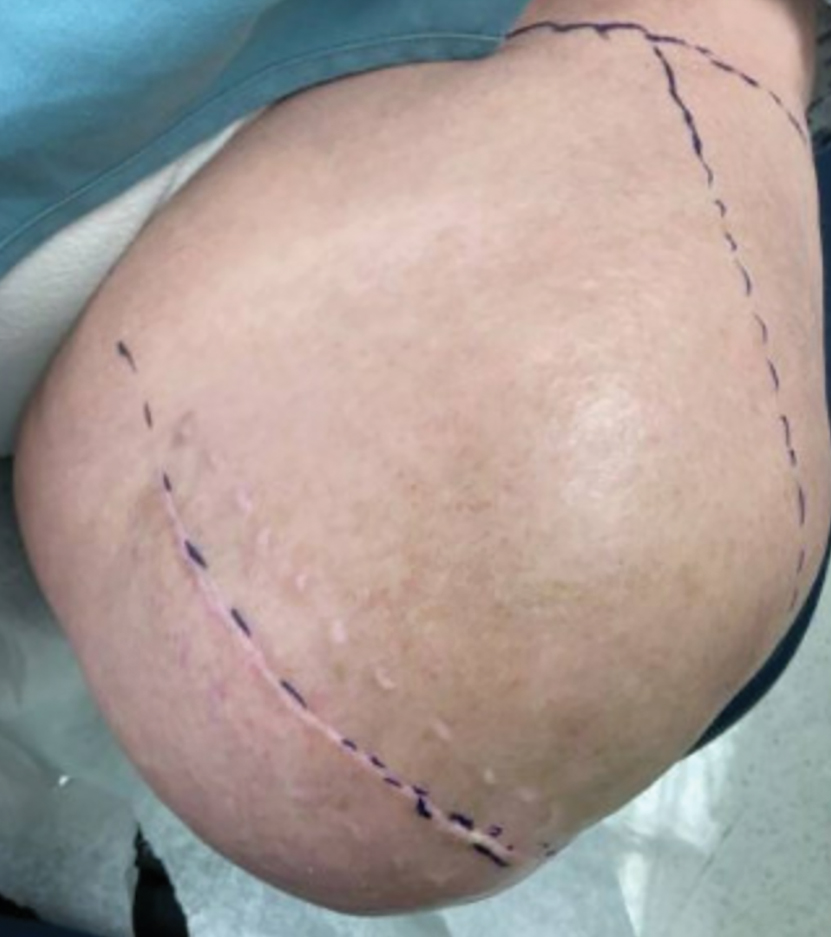
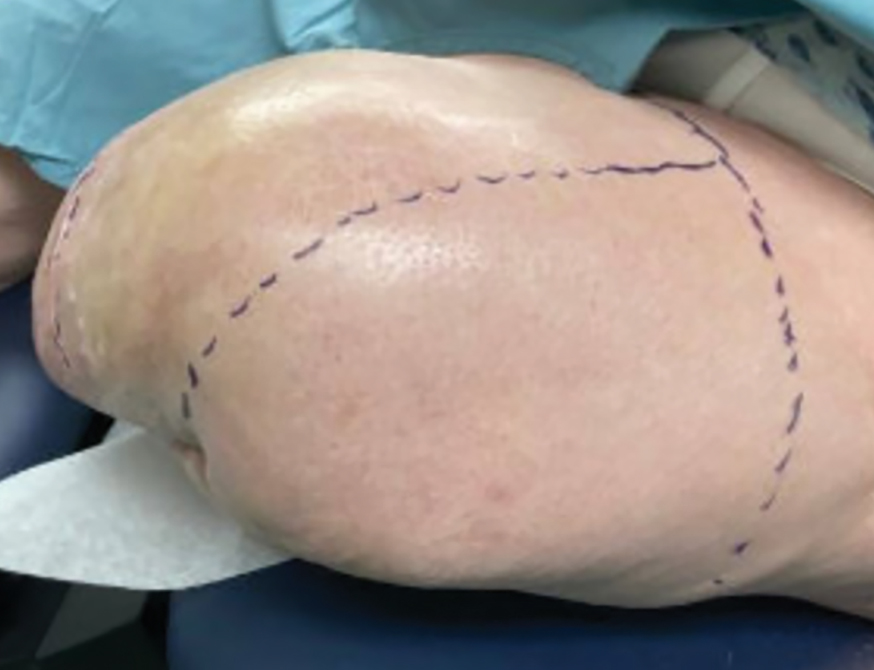
The treated section was cleaned with an alcohol wipe prior to each injection, and 50 units of BTX (diluted to 2.5 units per 0.1 mL in bacteriostatic saline) were injected intradermally into each section (Figure 2 and eFigure 2). The injections were administered in rows, with the needle inserted at evenly spaced intervals approximately 1 inch apart. A total of 100 units were administered per section at each weekly appointment. The patient tolerated the procedure well, and no complications were observed.
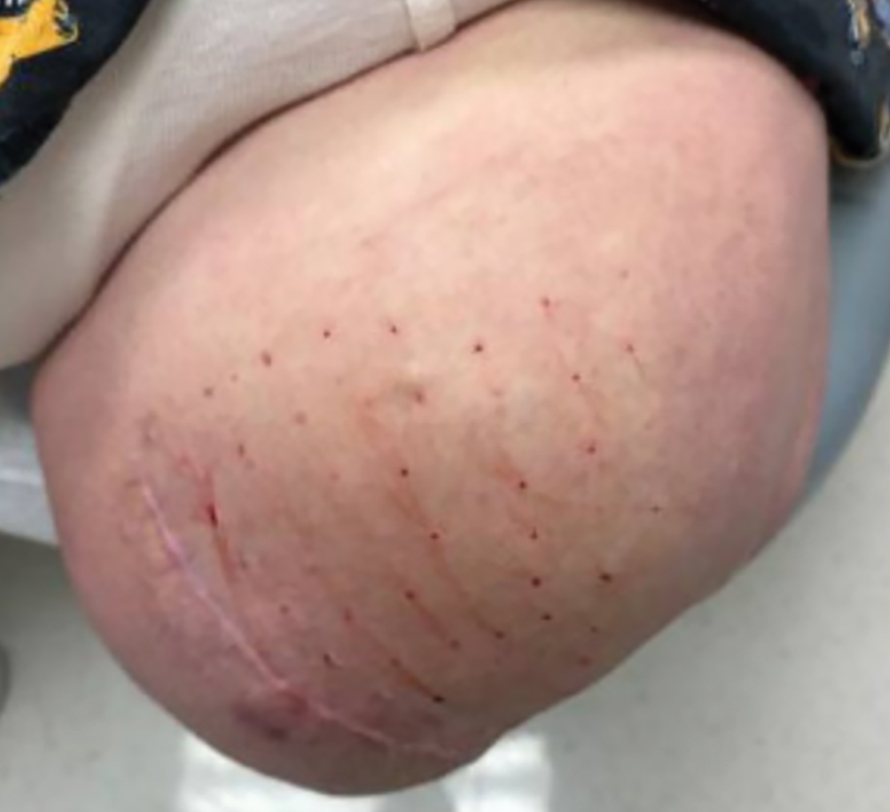
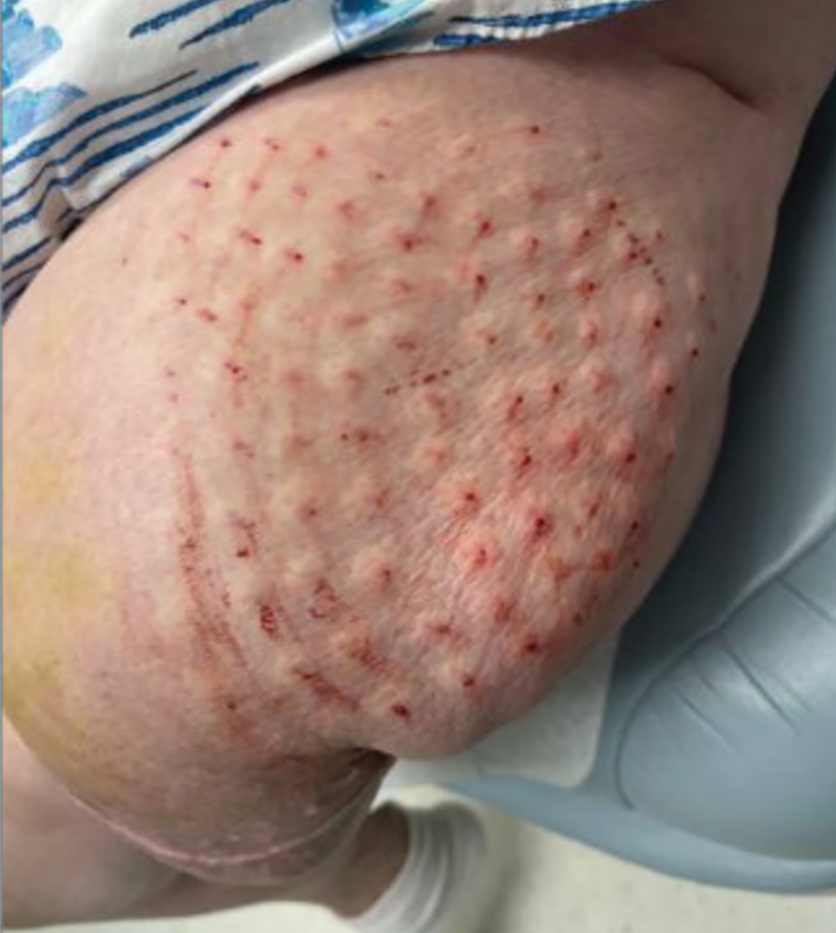
Practice Implications
This staged approach to administering BTX ensures even distribution of the injections, optimizes hyperhidrosis control, minimizes the risk for complications, and allows for precise targeting of the affected areas to maximize therapeutic benefit. Following the initial procedure, our patient was scheduled for follow-ups approximately every 3 to 4 months starting from the first set of injections for each area. Over 9 months, the patient successfully completed 3 treatment sessions using this method. The patient reported improved quality of life after starting the BTX injections.
After evaluating the initial treatment outcomes with 100 units per section, the dosage was increased to 200 units per section to reduce the number of visits from 4 every 3 months to cover the entire area to 2 visits every 3 months. This adjustment aimed to optimize results and better manage the patient’s ongoing symptoms. At about 1 to 2 weeks after beginning treatment, the patient noticed decreased sweating and discomfort during his daily activities and reduced friction with his prosthetic leg. No adverse effects were noted with the increased dosage during a clinical visit.
Our case highlights the importance of ensuring equitable access to hyperhidrosis treatment. Dermatologists should prioritize patient-centered care by factoring in financial constraints when recommending therapies. In this patient’s case, offering a range of options including over-the-counter antiperspirants and prescription treatments allowed for a management plan tailored to his individual needs and circumstances.
DaxibotulinumtoxinA, known for its longer duration of action compared to other BTX formulations, presents a promising alternative for treating hyperhidrosis.4 However, a gap in care emerged for our patient when prescription antiperspirant was not covered by his insurance, and daxibotulinumtoxinA, which could have offered a more durable solution, was not yet available at our clinic for hyperhidrosis management. Expanding insurance coverage for effective prescription treatments and improving access to newer treatment options are crucial for enhancing patient outcomes and ensuring more equitable care.
Focusing dermatologic care on amputees presents distinct challenges and opportunities for improving their care and decreasing discomfort. Amputees, particularly those with residual limb hyperhidrosis, often experience additional discomfort and difficulty while using prosthetics, as excessive sweating can interfere with fit and function.5,6 Dermatologists should proactively address these specific needs by tailoring treatment accordingly. Incorporating targeted therapies, such as BTX injections, in addition to education on lifestyle modifications and managing treatment expectations, ensures comprehensive care that enhances both quality of life and functional outcomes. Engaging patients in discussions about all available options, including emerging therapies, is essential for improving care for this underserved population.
- Koc E, Tunca M, Akar A, et al. Skin problems in amputees: a descriptive study. Int J Dermatol. 2008;47:463–466. doi:10.1111/j.1365-4632.2008.03604.x
- Bui KM, Raugi GJ, Nguyen VQ, et al. Skin problems in individuals with lower-limb loss: literature review and proposed classification system. J Rehabil Res Dev. 2009;46:1085-1090. doi:10.1682/jrrd.2009.04.0052
- Rocha Melo J, Rodrigues MA, Caetano M, et al. Botulinum toxin in the treatment of residual limb hyperhidrosis: a systematic review. Rehabilitacion (Madr). 2023;57:100754. doi:10.1016/j.rh.2022.07.003
- Hansen C, Godfrey B, Wixom J, et al. Incidence, severity, and impact of hyperhidrosis in people with lower-limb amputation. J Rehabil Res Dev. 2015;52:31-40. doi:10.1682/JRRD.2014.04.0108
- Lannan FM, Powell J, Kim GM, et al. Hyperhidrosis of the residual limb: a narrative review of the measurement and treatment of excess perspiration affecting individuals with amputation. Prosthet Orthot Int. 2021;45:477-486. doi:10.1097/PXR.0000000000000040
- Pace S, Kentosh J. Managing residual limb hyperhidrosis in wounded warriors. Cutis. 2016;97:401-403.
Practice Gap
Hyperhidrosis poses a considerable challenge for many amputees who use prosthetic devices, particularly at the interface between the residual limb and the prosthetic socket. The enclosed environment of the socket often leads to excessive sweating, which can compromise suction fit and increase the risk for skin chafing, irritation, and slippage. Persistent moisture also promotes bacterial and fungal growth, raising the likelihood of infections and foul odors within the socket. Research has shown that skin complications are highly prevalent among amputees, affecting up to 73.9% of this population in the United States.1 Commonly reported complications include wounds, abscesses, and blisters, many of which can be triggered or worsened by hyperhidrosis.2 Current treatment options for residual limb sweating include topical antiperspirants, botulinum toxin (BTX) injections, iontophoresis, and liner-liner socks.
While BTX commonly is used to treat hyperhidrosis in areas such as the palms and axillae, it typically is not considered as a first-line therapy for residual limb sweating; however, both BTX type A and type B have shown safety and effectiveness in managing hyperhidrosis in amputees, enhancing prosthetic use, and improving overall quality of life.3 Despite these benefits, BTX remains relatively underutilized for
Tools and Techniques
A 64-year-old man initially presented to our dermatology clinic after undergoing an above-the-knee amputation of the left leg 1 year prior. The amputation had been performed due to chronic prosthetic joint infections with Escherichia coli. He reported persistent sweating of the residual limb, which severely limited his use of a prosthesis and led to frequent falls.
During the initial visit, treatment options for primary hyperhidrosis including topical and injectable therapies were discussed. Due to a fear of needles, the patient chose topical treatment, with the option to pursue BTX injections later if better control was needed. An aluminum chloride hexahydrate prosthetic antiperspirant was prescribed for nightly application on the anterior and posterior
Botulinum toxin injections were administered in a grid-like pattern across the surface area where the residual limb made contact with the prosthetic. Using a surgical marker, the patient assisted the medical team in identifying the areas where sweating occurred most frequently. The area was divided into 4 equal sections, with each section treated per weekly interval sequentially over 4 weeks. The targeted areas included the left anterior (extending from the anterior tensor fasciae latae band to the lateral thigh) and left posterior residual limb (Figure 1 and eFigure 1, respectively).


The treated section was cleaned with an alcohol wipe prior to each injection, and 50 units of BTX (diluted to 2.5 units per 0.1 mL in bacteriostatic saline) were injected intradermally into each section (Figure 2 and eFigure 2). The injections were administered in rows, with the needle inserted at evenly spaced intervals approximately 1 inch apart. A total of 100 units were administered per section at each weekly appointment. The patient tolerated the procedure well, and no complications were observed.


Practice Implications
This staged approach to administering BTX ensures even distribution of the injections, optimizes hyperhidrosis control, minimizes the risk for complications, and allows for precise targeting of the affected areas to maximize therapeutic benefit. Following the initial procedure, our patient was scheduled for follow-ups approximately every 3 to 4 months starting from the first set of injections for each area. Over 9 months, the patient successfully completed 3 treatment sessions using this method. The patient reported improved quality of life after starting the BTX injections.
After evaluating the initial treatment outcomes with 100 units per section, the dosage was increased to 200 units per section to reduce the number of visits from 4 every 3 months to cover the entire area to 2 visits every 3 months. This adjustment aimed to optimize results and better manage the patient’s ongoing symptoms. At about 1 to 2 weeks after beginning treatment, the patient noticed decreased sweating and discomfort during his daily activities and reduced friction with his prosthetic leg. No adverse effects were noted with the increased dosage during a clinical visit.
Our case highlights the importance of ensuring equitable access to hyperhidrosis treatment. Dermatologists should prioritize patient-centered care by factoring in financial constraints when recommending therapies. In this patient’s case, offering a range of options including over-the-counter antiperspirants and prescription treatments allowed for a management plan tailored to his individual needs and circumstances.
DaxibotulinumtoxinA, known for its longer duration of action compared to other BTX formulations, presents a promising alternative for treating hyperhidrosis.4 However, a gap in care emerged for our patient when prescription antiperspirant was not covered by his insurance, and daxibotulinumtoxinA, which could have offered a more durable solution, was not yet available at our clinic for hyperhidrosis management. Expanding insurance coverage for effective prescription treatments and improving access to newer treatment options are crucial for enhancing patient outcomes and ensuring more equitable care.
Focusing dermatologic care on amputees presents distinct challenges and opportunities for improving their care and decreasing discomfort. Amputees, particularly those with residual limb hyperhidrosis, often experience additional discomfort and difficulty while using prosthetics, as excessive sweating can interfere with fit and function.5,6 Dermatologists should proactively address these specific needs by tailoring treatment accordingly. Incorporating targeted therapies, such as BTX injections, in addition to education on lifestyle modifications and managing treatment expectations, ensures comprehensive care that enhances both quality of life and functional outcomes. Engaging patients in discussions about all available options, including emerging therapies, is essential for improving care for this underserved population.
Practice Gap
Hyperhidrosis poses a considerable challenge for many amputees who use prosthetic devices, particularly at the interface between the residual limb and the prosthetic socket. The enclosed environment of the socket often leads to excessive sweating, which can compromise suction fit and increase the risk for skin chafing, irritation, and slippage. Persistent moisture also promotes bacterial and fungal growth, raising the likelihood of infections and foul odors within the socket. Research has shown that skin complications are highly prevalent among amputees, affecting up to 73.9% of this population in the United States.1 Commonly reported complications include wounds, abscesses, and blisters, many of which can be triggered or worsened by hyperhidrosis.2 Current treatment options for residual limb sweating include topical antiperspirants, botulinum toxin (BTX) injections, iontophoresis, and liner-liner socks.
While BTX commonly is used to treat hyperhidrosis in areas such as the palms and axillae, it typically is not considered as a first-line therapy for residual limb sweating; however, both BTX type A and type B have shown safety and effectiveness in managing hyperhidrosis in amputees, enhancing prosthetic use, and improving overall quality of life.3 Despite these benefits, BTX remains relatively underutilized for
Tools and Techniques
A 64-year-old man initially presented to our dermatology clinic after undergoing an above-the-knee amputation of the left leg 1 year prior. The amputation had been performed due to chronic prosthetic joint infections with Escherichia coli. He reported persistent sweating of the residual limb, which severely limited his use of a prosthesis and led to frequent falls.
During the initial visit, treatment options for primary hyperhidrosis including topical and injectable therapies were discussed. Due to a fear of needles, the patient chose topical treatment, with the option to pursue BTX injections later if better control was needed. An aluminum chloride hexahydrate prosthetic antiperspirant was prescribed for nightly application on the anterior and posterior
Botulinum toxin injections were administered in a grid-like pattern across the surface area where the residual limb made contact with the prosthetic. Using a surgical marker, the patient assisted the medical team in identifying the areas where sweating occurred most frequently. The area was divided into 4 equal sections, with each section treated per weekly interval sequentially over 4 weeks. The targeted areas included the left anterior (extending from the anterior tensor fasciae latae band to the lateral thigh) and left posterior residual limb (Figure 1 and eFigure 1, respectively).


The treated section was cleaned with an alcohol wipe prior to each injection, and 50 units of BTX (diluted to 2.5 units per 0.1 mL in bacteriostatic saline) were injected intradermally into each section (Figure 2 and eFigure 2). The injections were administered in rows, with the needle inserted at evenly spaced intervals approximately 1 inch apart. A total of 100 units were administered per section at each weekly appointment. The patient tolerated the procedure well, and no complications were observed.


Practice Implications
This staged approach to administering BTX ensures even distribution of the injections, optimizes hyperhidrosis control, minimizes the risk for complications, and allows for precise targeting of the affected areas to maximize therapeutic benefit. Following the initial procedure, our patient was scheduled for follow-ups approximately every 3 to 4 months starting from the first set of injections for each area. Over 9 months, the patient successfully completed 3 treatment sessions using this method. The patient reported improved quality of life after starting the BTX injections.
After evaluating the initial treatment outcomes with 100 units per section, the dosage was increased to 200 units per section to reduce the number of visits from 4 every 3 months to cover the entire area to 2 visits every 3 months. This adjustment aimed to optimize results and better manage the patient’s ongoing symptoms. At about 1 to 2 weeks after beginning treatment, the patient noticed decreased sweating and discomfort during his daily activities and reduced friction with his prosthetic leg. No adverse effects were noted with the increased dosage during a clinical visit.
Our case highlights the importance of ensuring equitable access to hyperhidrosis treatment. Dermatologists should prioritize patient-centered care by factoring in financial constraints when recommending therapies. In this patient’s case, offering a range of options including over-the-counter antiperspirants and prescription treatments allowed for a management plan tailored to his individual needs and circumstances.
DaxibotulinumtoxinA, known for its longer duration of action compared to other BTX formulations, presents a promising alternative for treating hyperhidrosis.4 However, a gap in care emerged for our patient when prescription antiperspirant was not covered by his insurance, and daxibotulinumtoxinA, which could have offered a more durable solution, was not yet available at our clinic for hyperhidrosis management. Expanding insurance coverage for effective prescription treatments and improving access to newer treatment options are crucial for enhancing patient outcomes and ensuring more equitable care.
Focusing dermatologic care on amputees presents distinct challenges and opportunities for improving their care and decreasing discomfort. Amputees, particularly those with residual limb hyperhidrosis, often experience additional discomfort and difficulty while using prosthetics, as excessive sweating can interfere with fit and function.5,6 Dermatologists should proactively address these specific needs by tailoring treatment accordingly. Incorporating targeted therapies, such as BTX injections, in addition to education on lifestyle modifications and managing treatment expectations, ensures comprehensive care that enhances both quality of life and functional outcomes. Engaging patients in discussions about all available options, including emerging therapies, is essential for improving care for this underserved population.
- Koc E, Tunca M, Akar A, et al. Skin problems in amputees: a descriptive study. Int J Dermatol. 2008;47:463–466. doi:10.1111/j.1365-4632.2008.03604.x
- Bui KM, Raugi GJ, Nguyen VQ, et al. Skin problems in individuals with lower-limb loss: literature review and proposed classification system. J Rehabil Res Dev. 2009;46:1085-1090. doi:10.1682/jrrd.2009.04.0052
- Rocha Melo J, Rodrigues MA, Caetano M, et al. Botulinum toxin in the treatment of residual limb hyperhidrosis: a systematic review. Rehabilitacion (Madr). 2023;57:100754. doi:10.1016/j.rh.2022.07.003
- Hansen C, Godfrey B, Wixom J, et al. Incidence, severity, and impact of hyperhidrosis in people with lower-limb amputation. J Rehabil Res Dev. 2015;52:31-40. doi:10.1682/JRRD.2014.04.0108
- Lannan FM, Powell J, Kim GM, et al. Hyperhidrosis of the residual limb: a narrative review of the measurement and treatment of excess perspiration affecting individuals with amputation. Prosthet Orthot Int. 2021;45:477-486. doi:10.1097/PXR.0000000000000040
- Pace S, Kentosh J. Managing residual limb hyperhidrosis in wounded warriors. Cutis. 2016;97:401-403.
- Koc E, Tunca M, Akar A, et al. Skin problems in amputees: a descriptive study. Int J Dermatol. 2008;47:463–466. doi:10.1111/j.1365-4632.2008.03604.x
- Bui KM, Raugi GJ, Nguyen VQ, et al. Skin problems in individuals with lower-limb loss: literature review and proposed classification system. J Rehabil Res Dev. 2009;46:1085-1090. doi:10.1682/jrrd.2009.04.0052
- Rocha Melo J, Rodrigues MA, Caetano M, et al. Botulinum toxin in the treatment of residual limb hyperhidrosis: a systematic review. Rehabilitacion (Madr). 2023;57:100754. doi:10.1016/j.rh.2022.07.003
- Hansen C, Godfrey B, Wixom J, et al. Incidence, severity, and impact of hyperhidrosis in people with lower-limb amputation. J Rehabil Res Dev. 2015;52:31-40. doi:10.1682/JRRD.2014.04.0108
- Lannan FM, Powell J, Kim GM, et al. Hyperhidrosis of the residual limb: a narrative review of the measurement and treatment of excess perspiration affecting individuals with amputation. Prosthet Orthot Int. 2021;45:477-486. doi:10.1097/PXR.0000000000000040
- Pace S, Kentosh J. Managing residual limb hyperhidrosis in wounded warriors. Cutis. 2016;97:401-403.
Botulinum Toxin as a Tool to Reduce Hyperhidrosis in Amputees
Botulinum Toxin as a Tool to Reduce Hyperhidrosis in Amputees
Tapping Into Relief: A Distraction Technique to Reduce Pain During Dermatologic Procedures
Tapping Into Relief: A Distraction Technique to Reduce Pain During Dermatologic Procedures
Practice Gap
Pain during minimally invasive dermatologic procedures such as lidocaine injections, cryotherapy, nail unit injections, and cosmetic procedures including neurotoxin injections can cause patient discomfort leading to procedural anxiety, poor compliance with treatment regimens, and avoidance of necessary care. Current solutions to manage pain during dermatologic procedures present several limitations; for example, topical anesthetics seldom alleviate procedural pain,1 particularly in sensitive areas (eg, nail unit, face) or for patients with a needle phobia. Additionally, topical anesthetics often require up to 2 hours to take effect, making them impractical for quick outpatient procedures. Other pain reduction strategies including vibration devices or cold sprays2,3 can be effective but are an added expense to the physician or clinic, which may preclude their use in resource-limited settings. Psychological distraction techniques such as deep breathing require active patient participation and might reinforce pain expectations and increase patient anxiety.4 Given these challenges, there is a need for effective, affordable, nonpharmacologic pain reduction strategies that can be integrated seamlessly into clinical practice to enhance the patient experience.
The Technique
Tapping is a simple noninvasive distraction technique that may alleviate procedural pain by exploiting the gate control theory of pain.5 According to this theory, tactile stimuli activate mechanoreceptors that send inhibitory signals to the spinal cord, effectively closing the gate to pain transmission. Unlike the Helfer skin tap technique,6 which involves 15 preinjection taps and 3 postinjection taps directly on the injection site, our approach targets distant bony prominences. This modification allows for immediate needle insertion without interfering with the sterile field or increasing the risk for needlestick injuries from tapping near the injection site. Bony sites such as the shoulder or knee are ideal for this technique due to their high density and rigidity that efficiently transmit tactile stimuli––similar to how sound travels faster through solids than through liquids or gases.7
To implement this technique in practice, we first stabilize the injection site to reduce movement from tapping. This can be done by stabilizing the injection site (eg, resting the hand on an instrument stand during a nail unit injection). A second person—such as a medical assistant, medical student, resident, or even the patient’s family member—taps at a distant site at least an arm’s length away from the injection site (Figure). The tapping pressure should be firm enough for the patient to feel the vibration but not forceful enough that it becomes unpleasant or disrupts the injection area. Tapping starts just before needle insertion and continues through the injection. No warning is given to the patient, as the surprise element may help distract them from pain. Varying the rhythm, intensity, or location of the tapping can enhance its distracting effect.
This tapping technique can be effectively combined with other pain reduction strategies in a multimodal approach; for example, when used concurrently with topical anesthetics, both the central (tapping) and peripheral (anesthetic) pain pathways are addressed, potentially yielding additive effects. For patients with a needle phobia, pairing tapping with cognitive distraction (eg, talkesthesia) may further reduce anxiety. In our nail specialty clinic at Weill Cornell Medicine (New York, New York), we often combine tapping with cold sprays and talkesthesia, which improves patient comfort without prolonging the visit. Importantly, the technique enables seamless integration with most pharmacologic and nonpharmacologic methods, eliminating the need for additional patient education or procedure time.
Practice Implications
The tapping technique described here is free, easy to implement, and requires no additional resources aside from another person to tap the patient during the procedure. It can be used for a wide range of dermatologic procedures, including biopsies, intralesional injections, and cosmetic treatments, including neurotoxin injections. The minimal learning curve and ease of integration into procedural workflows make this technique a valuable tool for dermatologists aiming to improve patient comfort without disrupting workflow. In our practice, we have observed that tapping reduces self-reported pain and helps ease anxiety, particularly in patients with a needle phobia. Its simplicity and accessibility make it a valuable addition to a wide range of dermatologic procedures. Prospective studies investigating patient-reported outcomes could help establish this technique’s role in clinical practice.
- Navarro-Rodriguez JM, Suarez-Serrano C, Martin-Valero R, et al. Effectiveness of topical anesthetics in pain management for dermal injuries: a systematic review. J Clin Med. 2021;10:2522. doi:10.3390/jcm10112522
- Lipner SR. Pain-minimizing strategies for nail surgery. Cutis. 2018;101:76-77.
- Ricardo JW, Lipner SR. Air cooling for improved analgesia during local anesthetic infiltration for nail surgery. J Am Acad Dermatol. 2021;84:e231-e232. doi:10.1016/j.jaad.2019.11.032
- Hill RC, Chernoff KA, Lipner SR. A breath of fresh air: use of deep breathing technique to minimize pain with nail injections. J Am Acad Dermatol. 2024;90:e163. doi:10.1016/j.jaad.2023.10.043
- Mendell LM. Constructing and deconstructing the gate theory of pain. Pain. 2014;155:210-216. doi:10.1016/j.pain.2013.12.010
- Jyoti G, Arora S, Sharma B. Helfer Skin Tap Tech Technique for the IM injection pain among adult patients. Nursing & Midwifery Research Journal. 2018;14:18-30. doi:10.1177/0974150X20180304
- Iowa State University. Nondestructive Evaluation Physics: Sound. Published 2021. Accessed July 31, 2025. https://www.nde-ed.org/Physics/Sound/speedinmaterials.xhtml
Practice Gap
Pain during minimally invasive dermatologic procedures such as lidocaine injections, cryotherapy, nail unit injections, and cosmetic procedures including neurotoxin injections can cause patient discomfort leading to procedural anxiety, poor compliance with treatment regimens, and avoidance of necessary care. Current solutions to manage pain during dermatologic procedures present several limitations; for example, topical anesthetics seldom alleviate procedural pain,1 particularly in sensitive areas (eg, nail unit, face) or for patients with a needle phobia. Additionally, topical anesthetics often require up to 2 hours to take effect, making them impractical for quick outpatient procedures. Other pain reduction strategies including vibration devices or cold sprays2,3 can be effective but are an added expense to the physician or clinic, which may preclude their use in resource-limited settings. Psychological distraction techniques such as deep breathing require active patient participation and might reinforce pain expectations and increase patient anxiety.4 Given these challenges, there is a need for effective, affordable, nonpharmacologic pain reduction strategies that can be integrated seamlessly into clinical practice to enhance the patient experience.
The Technique
Tapping is a simple noninvasive distraction technique that may alleviate procedural pain by exploiting the gate control theory of pain.5 According to this theory, tactile stimuli activate mechanoreceptors that send inhibitory signals to the spinal cord, effectively closing the gate to pain transmission. Unlike the Helfer skin tap technique,6 which involves 15 preinjection taps and 3 postinjection taps directly on the injection site, our approach targets distant bony prominences. This modification allows for immediate needle insertion without interfering with the sterile field or increasing the risk for needlestick injuries from tapping near the injection site. Bony sites such as the shoulder or knee are ideal for this technique due to their high density and rigidity that efficiently transmit tactile stimuli––similar to how sound travels faster through solids than through liquids or gases.7
To implement this technique in practice, we first stabilize the injection site to reduce movement from tapping. This can be done by stabilizing the injection site (eg, resting the hand on an instrument stand during a nail unit injection). A second person—such as a medical assistant, medical student, resident, or even the patient’s family member—taps at a distant site at least an arm’s length away from the injection site (Figure). The tapping pressure should be firm enough for the patient to feel the vibration but not forceful enough that it becomes unpleasant or disrupts the injection area. Tapping starts just before needle insertion and continues through the injection. No warning is given to the patient, as the surprise element may help distract them from pain. Varying the rhythm, intensity, or location of the tapping can enhance its distracting effect.

This tapping technique can be effectively combined with other pain reduction strategies in a multimodal approach; for example, when used concurrently with topical anesthetics, both the central (tapping) and peripheral (anesthetic) pain pathways are addressed, potentially yielding additive effects. For patients with a needle phobia, pairing tapping with cognitive distraction (eg, talkesthesia) may further reduce anxiety. In our nail specialty clinic at Weill Cornell Medicine (New York, New York), we often combine tapping with cold sprays and talkesthesia, which improves patient comfort without prolonging the visit. Importantly, the technique enables seamless integration with most pharmacologic and nonpharmacologic methods, eliminating the need for additional patient education or procedure time.
Practice Implications
The tapping technique described here is free, easy to implement, and requires no additional resources aside from another person to tap the patient during the procedure. It can be used for a wide range of dermatologic procedures, including biopsies, intralesional injections, and cosmetic treatments, including neurotoxin injections. The minimal learning curve and ease of integration into procedural workflows make this technique a valuable tool for dermatologists aiming to improve patient comfort without disrupting workflow. In our practice, we have observed that tapping reduces self-reported pain and helps ease anxiety, particularly in patients with a needle phobia. Its simplicity and accessibility make it a valuable addition to a wide range of dermatologic procedures. Prospective studies investigating patient-reported outcomes could help establish this technique’s role in clinical practice.
Practice Gap
Pain during minimally invasive dermatologic procedures such as lidocaine injections, cryotherapy, nail unit injections, and cosmetic procedures including neurotoxin injections can cause patient discomfort leading to procedural anxiety, poor compliance with treatment regimens, and avoidance of necessary care. Current solutions to manage pain during dermatologic procedures present several limitations; for example, topical anesthetics seldom alleviate procedural pain,1 particularly in sensitive areas (eg, nail unit, face) or for patients with a needle phobia. Additionally, topical anesthetics often require up to 2 hours to take effect, making them impractical for quick outpatient procedures. Other pain reduction strategies including vibration devices or cold sprays2,3 can be effective but are an added expense to the physician or clinic, which may preclude their use in resource-limited settings. Psychological distraction techniques such as deep breathing require active patient participation and might reinforce pain expectations and increase patient anxiety.4 Given these challenges, there is a need for effective, affordable, nonpharmacologic pain reduction strategies that can be integrated seamlessly into clinical practice to enhance the patient experience.
The Technique
Tapping is a simple noninvasive distraction technique that may alleviate procedural pain by exploiting the gate control theory of pain.5 According to this theory, tactile stimuli activate mechanoreceptors that send inhibitory signals to the spinal cord, effectively closing the gate to pain transmission. Unlike the Helfer skin tap technique,6 which involves 15 preinjection taps and 3 postinjection taps directly on the injection site, our approach targets distant bony prominences. This modification allows for immediate needle insertion without interfering with the sterile field or increasing the risk for needlestick injuries from tapping near the injection site. Bony sites such as the shoulder or knee are ideal for this technique due to their high density and rigidity that efficiently transmit tactile stimuli––similar to how sound travels faster through solids than through liquids or gases.7
To implement this technique in practice, we first stabilize the injection site to reduce movement from tapping. This can be done by stabilizing the injection site (eg, resting the hand on an instrument stand during a nail unit injection). A second person—such as a medical assistant, medical student, resident, or even the patient’s family member—taps at a distant site at least an arm’s length away from the injection site (Figure). The tapping pressure should be firm enough for the patient to feel the vibration but not forceful enough that it becomes unpleasant or disrupts the injection area. Tapping starts just before needle insertion and continues through the injection. No warning is given to the patient, as the surprise element may help distract them from pain. Varying the rhythm, intensity, or location of the tapping can enhance its distracting effect.

This tapping technique can be effectively combined with other pain reduction strategies in a multimodal approach; for example, when used concurrently with topical anesthetics, both the central (tapping) and peripheral (anesthetic) pain pathways are addressed, potentially yielding additive effects. For patients with a needle phobia, pairing tapping with cognitive distraction (eg, talkesthesia) may further reduce anxiety. In our nail specialty clinic at Weill Cornell Medicine (New York, New York), we often combine tapping with cold sprays and talkesthesia, which improves patient comfort without prolonging the visit. Importantly, the technique enables seamless integration with most pharmacologic and nonpharmacologic methods, eliminating the need for additional patient education or procedure time.
Practice Implications
The tapping technique described here is free, easy to implement, and requires no additional resources aside from another person to tap the patient during the procedure. It can be used for a wide range of dermatologic procedures, including biopsies, intralesional injections, and cosmetic treatments, including neurotoxin injections. The minimal learning curve and ease of integration into procedural workflows make this technique a valuable tool for dermatologists aiming to improve patient comfort without disrupting workflow. In our practice, we have observed that tapping reduces self-reported pain and helps ease anxiety, particularly in patients with a needle phobia. Its simplicity and accessibility make it a valuable addition to a wide range of dermatologic procedures. Prospective studies investigating patient-reported outcomes could help establish this technique’s role in clinical practice.
- Navarro-Rodriguez JM, Suarez-Serrano C, Martin-Valero R, et al. Effectiveness of topical anesthetics in pain management for dermal injuries: a systematic review. J Clin Med. 2021;10:2522. doi:10.3390/jcm10112522
- Lipner SR. Pain-minimizing strategies for nail surgery. Cutis. 2018;101:76-77.
- Ricardo JW, Lipner SR. Air cooling for improved analgesia during local anesthetic infiltration for nail surgery. J Am Acad Dermatol. 2021;84:e231-e232. doi:10.1016/j.jaad.2019.11.032
- Hill RC, Chernoff KA, Lipner SR. A breath of fresh air: use of deep breathing technique to minimize pain with nail injections. J Am Acad Dermatol. 2024;90:e163. doi:10.1016/j.jaad.2023.10.043
- Mendell LM. Constructing and deconstructing the gate theory of pain. Pain. 2014;155:210-216. doi:10.1016/j.pain.2013.12.010
- Jyoti G, Arora S, Sharma B. Helfer Skin Tap Tech Technique for the IM injection pain among adult patients. Nursing & Midwifery Research Journal. 2018;14:18-30. doi:10.1177/0974150X20180304
- Iowa State University. Nondestructive Evaluation Physics: Sound. Published 2021. Accessed July 31, 2025. https://www.nde-ed.org/Physics/Sound/speedinmaterials.xhtml
- Navarro-Rodriguez JM, Suarez-Serrano C, Martin-Valero R, et al. Effectiveness of topical anesthetics in pain management for dermal injuries: a systematic review. J Clin Med. 2021;10:2522. doi:10.3390/jcm10112522
- Lipner SR. Pain-minimizing strategies for nail surgery. Cutis. 2018;101:76-77.
- Ricardo JW, Lipner SR. Air cooling for improved analgesia during local anesthetic infiltration for nail surgery. J Am Acad Dermatol. 2021;84:e231-e232. doi:10.1016/j.jaad.2019.11.032
- Hill RC, Chernoff KA, Lipner SR. A breath of fresh air: use of deep breathing technique to minimize pain with nail injections. J Am Acad Dermatol. 2024;90:e163. doi:10.1016/j.jaad.2023.10.043
- Mendell LM. Constructing and deconstructing the gate theory of pain. Pain. 2014;155:210-216. doi:10.1016/j.pain.2013.12.010
- Jyoti G, Arora S, Sharma B. Helfer Skin Tap Tech Technique for the IM injection pain among adult patients. Nursing & Midwifery Research Journal. 2018;14:18-30. doi:10.1177/0974150X20180304
- Iowa State University. Nondestructive Evaluation Physics: Sound. Published 2021. Accessed July 31, 2025. https://www.nde-ed.org/Physics/Sound/speedinmaterials.xhtml
Tapping Into Relief: A Distraction Technique to Reduce Pain During Dermatologic Procedures
Tapping Into Relief: A Distraction Technique to Reduce Pain During Dermatologic Procedures
Choosing the Best Formalin-Resistant Ink for Biopsy Specimen Labeling
Choosing the Best Formalin-Resistant Ink for Biopsy Specimen Labeling
Practice Gap
Many dermatology practices utilize pens and markers to label biopsy specimen containers, but the ink may have variable susceptibility to fading and smearing when exposed to moisture before processing. Specimen containers often are placed in plastic bags for transport. If formalin accidentally spills into the bag during this time, the labels may be exposed to moisture for hours, overnight, or even over a weekend. Effective labeling with formalin-resistant ink is crucial for maintaining the clarity of anatomic location and planning treatment, especially when multiple samples are obtained.
The Technique
We tested 12 pens and markers commonly used when labeling specimen containers to determine their susceptibility to fading due to accidental formalin exposure (Figure). Various inks were allowed to dry on sample specimen labels for 5 minutes before a thin layer of 10% buffered formalin was evenly distributed over the dried ink. Photographs of the labels were taken at baseline as well as 15 minutes, 1 hour, 3 hours, and 24 hours after formalin exposure.
Fading was observed in both the skin marker and gel panes after 15 minutes and peaked after 1 hour. Gel pens were most susceptible to fading on exposure to formalin, and the level of fading varied by ink color, with certain colors disappearing almost entirely (Figure). The solvent-resistant marker had a robust defense to formalin, as did both ballpoint pens.
Practice Implications
Given our findings, dermatology practices should avoid using gel pens to label specimen containers. Solvent-resistant markers performed as expected; however, ballpoint pens appeared to withstand formalin exposure to a similar degree and often are more readily available. Labeling biopsy specimens with an appropriate ink ensures that each sample is clearly identified with the appropriate anatomic location and any other relevant patient information.
Practice Gap
Many dermatology practices utilize pens and markers to label biopsy specimen containers, but the ink may have variable susceptibility to fading and smearing when exposed to moisture before processing. Specimen containers often are placed in plastic bags for transport. If formalin accidentally spills into the bag during this time, the labels may be exposed to moisture for hours, overnight, or even over a weekend. Effective labeling with formalin-resistant ink is crucial for maintaining the clarity of anatomic location and planning treatment, especially when multiple samples are obtained.
The Technique
We tested 12 pens and markers commonly used when labeling specimen containers to determine their susceptibility to fading due to accidental formalin exposure (Figure). Various inks were allowed to dry on sample specimen labels for 5 minutes before a thin layer of 10% buffered formalin was evenly distributed over the dried ink. Photographs of the labels were taken at baseline as well as 15 minutes, 1 hour, 3 hours, and 24 hours after formalin exposure.

Fading was observed in both the skin marker and gel panes after 15 minutes and peaked after 1 hour. Gel pens were most susceptible to fading on exposure to formalin, and the level of fading varied by ink color, with certain colors disappearing almost entirely (Figure). The solvent-resistant marker had a robust defense to formalin, as did both ballpoint pens.
Practice Implications
Given our findings, dermatology practices should avoid using gel pens to label specimen containers. Solvent-resistant markers performed as expected; however, ballpoint pens appeared to withstand formalin exposure to a similar degree and often are more readily available. Labeling biopsy specimens with an appropriate ink ensures that each sample is clearly identified with the appropriate anatomic location and any other relevant patient information.
Practice Gap
Many dermatology practices utilize pens and markers to label biopsy specimen containers, but the ink may have variable susceptibility to fading and smearing when exposed to moisture before processing. Specimen containers often are placed in plastic bags for transport. If formalin accidentally spills into the bag during this time, the labels may be exposed to moisture for hours, overnight, or even over a weekend. Effective labeling with formalin-resistant ink is crucial for maintaining the clarity of anatomic location and planning treatment, especially when multiple samples are obtained.
The Technique
We tested 12 pens and markers commonly used when labeling specimen containers to determine their susceptibility to fading due to accidental formalin exposure (Figure). Various inks were allowed to dry on sample specimen labels for 5 minutes before a thin layer of 10% buffered formalin was evenly distributed over the dried ink. Photographs of the labels were taken at baseline as well as 15 minutes, 1 hour, 3 hours, and 24 hours after formalin exposure.

Fading was observed in both the skin marker and gel panes after 15 minutes and peaked after 1 hour. Gel pens were most susceptible to fading on exposure to formalin, and the level of fading varied by ink color, with certain colors disappearing almost entirely (Figure). The solvent-resistant marker had a robust defense to formalin, as did both ballpoint pens.
Practice Implications
Given our findings, dermatology practices should avoid using gel pens to label specimen containers. Solvent-resistant markers performed as expected; however, ballpoint pens appeared to withstand formalin exposure to a similar degree and often are more readily available. Labeling biopsy specimens with an appropriate ink ensures that each sample is clearly identified with the appropriate anatomic location and any other relevant patient information.
Choosing the Best Formalin-Resistant Ink for Biopsy Specimen Labeling
Choosing the Best Formalin-Resistant Ink for Biopsy Specimen Labeling