User login
Oncologists need to advocate for scarce COVID-19 resources: ASCO
both at the institutional and regional level, according to new recommendations from the American Society for Clinical Oncology (ASCO).
“There was a lot of concern from the oncology community that if a patient had cancer, they would be arbitrarily excluded from consideration for critical care resources,” said Jonathan M. Marron, MD, chair-elect of ASCO’s Ethics Committee and lead author of the recommendations.
“The hope is that we’ll never have to make any of these decisions ... but the primary reason for putting together these recommendations was that if such decisions have to be made, we hope to inform them,” he told Medscape Medical News.
Marron, who is a pediatric hematologist at Boston Children’s Hospital, says ASCO’s main recommendation is that decisions about the allocation of resources must be separated from bedside clinical care, meaning that clinicians who are caring for individual patients should not also be the ones making the allocation decisions.
“Those dueling responsibilities are a conflict of interest and make that physician unable to make an unbiased decision,” he said.
“It’s also just an unbearable burden to try and do those two things simultaneously,” he added. “It’s an incredible burden to do them individually, but it’s multifold worse to try to do them both simultaneously.”
He said the vital role of oncologists who provide treatment is to offer the kind of personalized information that triage committees need in order to make appropriate decisions.
“They should be asked – maybe even must be asked – to provide the most high-quality evidence-based data about their patients’ diagnosis and prognosis,” Marron commented. “Because oncology is evolving so rapidly, and cancer is so many different diseases, it’s impossible for someone making these decisions to know everything they would need to know about why this patient is likely to survive their cancer and this patient is not.”
He says that during the COVID-19 pandemic, concerns regarding public health transcend the well-being of individual patients and that consideration must be given to providing the maximum benefit to the greatest number of people.
“That makes perfect sense and is the appropriate and laudable goal during a public health emergency like this ... but one of the challenges is that there is this belief that a diagnosis of cancer is uniformly fatal,” Marron said.
“It’s certainly conceivable that it would be a better use of resources to give the last ventilator to a young, otherwise healthy patient rather than a patient with multiply recurrent progressive metastatic cancer,” he continued. “However, we want to ensure that there is at least a discussion where that information is made available, rather than just saying, ‘She’s got cancer. She’s a lost cause.’ ”
Cancer patients are doing very well
Concerns about cancer misconceptions have been circulating in the oncology community since the start of the pandemic. “It’s really important that people understand that cancer patients are doing very well nowadays, and even with a diagnosis of cancer, they can potentially live for many years,” Anne Chiang, MD, PhD, from the Smilow Cancer Network, New Haven, Connecticut, told Medscape Medical News in a recent interview.
Thus far, even in hard-hit New York City, fears that cancer patients may not be receiving appropriate care have not materialized, according to Mark Robson, MD, a medical oncologist at Memorial Sloan Kettering Cancer Center (MSKCC). “I would emphasize that cancer patients are ABSOLUTELY getting the care they need, including patients with metastatic disease,” he recently tweeted. “NOONE at @sloan_kettering (or anywhere else) is being ‘triaged’ because of advanced cancer. Period.”
Robson told Medscape Medical News that although MSKCC continues to provide oncology care to patients with cancer, “we are [also] treating them if they develop COVID. ... I am trying to help pivot the institution towards care in this setting.”
He said he agrees with Craig Spencer, MD, MPH, director of global health in emergency medicine at the New York–Presbyterian/Columbia University Medical Center, who recently tweeted, “If you need a ventilator, you get a ventilator. Let’s be clear – this isn’t being ‘rationed.’ ”
Marron emphasized that an important safeguard against uninformed decision making is appropriate planning. For hospitalized patients, this means oncologists who provide treatment should offer information even before it is requested. But he said the “duty to plan” begins long before that.
“Clinicians haven’t always been great at talking about death and long-term outcomes with their patients, but this really cranks up the importance of having those conversations, and having them early, even though it’s incredibly hard. If someone has expressed that they would never want to be put on a ventilator, it’s important now even more so that is made clear,” he said.
He said early responses to the ASCO statement suggest that it has calmed some concerns in the oncology community, “but it still remains to be seen whether individual institutions will take this to heart, because this unto itself cannot enforce anything – it is up to individual institutions. I am hopeful this will get to the people it needs to get to.”
This article first appeared on Medscape.com.
both at the institutional and regional level, according to new recommendations from the American Society for Clinical Oncology (ASCO).
“There was a lot of concern from the oncology community that if a patient had cancer, they would be arbitrarily excluded from consideration for critical care resources,” said Jonathan M. Marron, MD, chair-elect of ASCO’s Ethics Committee and lead author of the recommendations.
“The hope is that we’ll never have to make any of these decisions ... but the primary reason for putting together these recommendations was that if such decisions have to be made, we hope to inform them,” he told Medscape Medical News.
Marron, who is a pediatric hematologist at Boston Children’s Hospital, says ASCO’s main recommendation is that decisions about the allocation of resources must be separated from bedside clinical care, meaning that clinicians who are caring for individual patients should not also be the ones making the allocation decisions.
“Those dueling responsibilities are a conflict of interest and make that physician unable to make an unbiased decision,” he said.
“It’s also just an unbearable burden to try and do those two things simultaneously,” he added. “It’s an incredible burden to do them individually, but it’s multifold worse to try to do them both simultaneously.”
He said the vital role of oncologists who provide treatment is to offer the kind of personalized information that triage committees need in order to make appropriate decisions.
“They should be asked – maybe even must be asked – to provide the most high-quality evidence-based data about their patients’ diagnosis and prognosis,” Marron commented. “Because oncology is evolving so rapidly, and cancer is so many different diseases, it’s impossible for someone making these decisions to know everything they would need to know about why this patient is likely to survive their cancer and this patient is not.”
He says that during the COVID-19 pandemic, concerns regarding public health transcend the well-being of individual patients and that consideration must be given to providing the maximum benefit to the greatest number of people.
“That makes perfect sense and is the appropriate and laudable goal during a public health emergency like this ... but one of the challenges is that there is this belief that a diagnosis of cancer is uniformly fatal,” Marron said.
“It’s certainly conceivable that it would be a better use of resources to give the last ventilator to a young, otherwise healthy patient rather than a patient with multiply recurrent progressive metastatic cancer,” he continued. “However, we want to ensure that there is at least a discussion where that information is made available, rather than just saying, ‘She’s got cancer. She’s a lost cause.’ ”
Cancer patients are doing very well
Concerns about cancer misconceptions have been circulating in the oncology community since the start of the pandemic. “It’s really important that people understand that cancer patients are doing very well nowadays, and even with a diagnosis of cancer, they can potentially live for many years,” Anne Chiang, MD, PhD, from the Smilow Cancer Network, New Haven, Connecticut, told Medscape Medical News in a recent interview.
Thus far, even in hard-hit New York City, fears that cancer patients may not be receiving appropriate care have not materialized, according to Mark Robson, MD, a medical oncologist at Memorial Sloan Kettering Cancer Center (MSKCC). “I would emphasize that cancer patients are ABSOLUTELY getting the care they need, including patients with metastatic disease,” he recently tweeted. “NOONE at @sloan_kettering (or anywhere else) is being ‘triaged’ because of advanced cancer. Period.”
Robson told Medscape Medical News that although MSKCC continues to provide oncology care to patients with cancer, “we are [also] treating them if they develop COVID. ... I am trying to help pivot the institution towards care in this setting.”
He said he agrees with Craig Spencer, MD, MPH, director of global health in emergency medicine at the New York–Presbyterian/Columbia University Medical Center, who recently tweeted, “If you need a ventilator, you get a ventilator. Let’s be clear – this isn’t being ‘rationed.’ ”
Marron emphasized that an important safeguard against uninformed decision making is appropriate planning. For hospitalized patients, this means oncologists who provide treatment should offer information even before it is requested. But he said the “duty to plan” begins long before that.
“Clinicians haven’t always been great at talking about death and long-term outcomes with their patients, but this really cranks up the importance of having those conversations, and having them early, even though it’s incredibly hard. If someone has expressed that they would never want to be put on a ventilator, it’s important now even more so that is made clear,” he said.
He said early responses to the ASCO statement suggest that it has calmed some concerns in the oncology community, “but it still remains to be seen whether individual institutions will take this to heart, because this unto itself cannot enforce anything – it is up to individual institutions. I am hopeful this will get to the people it needs to get to.”
This article first appeared on Medscape.com.
both at the institutional and regional level, according to new recommendations from the American Society for Clinical Oncology (ASCO).
“There was a lot of concern from the oncology community that if a patient had cancer, they would be arbitrarily excluded from consideration for critical care resources,” said Jonathan M. Marron, MD, chair-elect of ASCO’s Ethics Committee and lead author of the recommendations.
“The hope is that we’ll never have to make any of these decisions ... but the primary reason for putting together these recommendations was that if such decisions have to be made, we hope to inform them,” he told Medscape Medical News.
Marron, who is a pediatric hematologist at Boston Children’s Hospital, says ASCO’s main recommendation is that decisions about the allocation of resources must be separated from bedside clinical care, meaning that clinicians who are caring for individual patients should not also be the ones making the allocation decisions.
“Those dueling responsibilities are a conflict of interest and make that physician unable to make an unbiased decision,” he said.
“It’s also just an unbearable burden to try and do those two things simultaneously,” he added. “It’s an incredible burden to do them individually, but it’s multifold worse to try to do them both simultaneously.”
He said the vital role of oncologists who provide treatment is to offer the kind of personalized information that triage committees need in order to make appropriate decisions.
“They should be asked – maybe even must be asked – to provide the most high-quality evidence-based data about their patients’ diagnosis and prognosis,” Marron commented. “Because oncology is evolving so rapidly, and cancer is so many different diseases, it’s impossible for someone making these decisions to know everything they would need to know about why this patient is likely to survive their cancer and this patient is not.”
He says that during the COVID-19 pandemic, concerns regarding public health transcend the well-being of individual patients and that consideration must be given to providing the maximum benefit to the greatest number of people.
“That makes perfect sense and is the appropriate and laudable goal during a public health emergency like this ... but one of the challenges is that there is this belief that a diagnosis of cancer is uniformly fatal,” Marron said.
“It’s certainly conceivable that it would be a better use of resources to give the last ventilator to a young, otherwise healthy patient rather than a patient with multiply recurrent progressive metastatic cancer,” he continued. “However, we want to ensure that there is at least a discussion where that information is made available, rather than just saying, ‘She’s got cancer. She’s a lost cause.’ ”
Cancer patients are doing very well
Concerns about cancer misconceptions have been circulating in the oncology community since the start of the pandemic. “It’s really important that people understand that cancer patients are doing very well nowadays, and even with a diagnosis of cancer, they can potentially live for many years,” Anne Chiang, MD, PhD, from the Smilow Cancer Network, New Haven, Connecticut, told Medscape Medical News in a recent interview.
Thus far, even in hard-hit New York City, fears that cancer patients may not be receiving appropriate care have not materialized, according to Mark Robson, MD, a medical oncologist at Memorial Sloan Kettering Cancer Center (MSKCC). “I would emphasize that cancer patients are ABSOLUTELY getting the care they need, including patients with metastatic disease,” he recently tweeted. “NOONE at @sloan_kettering (or anywhere else) is being ‘triaged’ because of advanced cancer. Period.”
Robson told Medscape Medical News that although MSKCC continues to provide oncology care to patients with cancer, “we are [also] treating them if they develop COVID. ... I am trying to help pivot the institution towards care in this setting.”
He said he agrees with Craig Spencer, MD, MPH, director of global health in emergency medicine at the New York–Presbyterian/Columbia University Medical Center, who recently tweeted, “If you need a ventilator, you get a ventilator. Let’s be clear – this isn’t being ‘rationed.’ ”
Marron emphasized that an important safeguard against uninformed decision making is appropriate planning. For hospitalized patients, this means oncologists who provide treatment should offer information even before it is requested. But he said the “duty to plan” begins long before that.
“Clinicians haven’t always been great at talking about death and long-term outcomes with their patients, but this really cranks up the importance of having those conversations, and having them early, even though it’s incredibly hard. If someone has expressed that they would never want to be put on a ventilator, it’s important now even more so that is made clear,” he said.
He said early responses to the ASCO statement suggest that it has calmed some concerns in the oncology community, “but it still remains to be seen whether individual institutions will take this to heart, because this unto itself cannot enforce anything – it is up to individual institutions. I am hopeful this will get to the people it needs to get to.”
This article first appeared on Medscape.com.
ASH tackles COVID-19 with hematology-related FAQ, promotes new registries
The American Society of Hematology has committed a portion of its website to providing continually updated information addressing specific hematologic disorders in relation to COVID-19.
“As the world grapples with the novel coronavirus, ASH believes that we can help each other be as knowledgeable and prepared as possible,” wrote the society’s president, Stephanie J. Lee, MD, MPH.
On its website, ASH provides relevant COVID-19 information in a series of FAQ divided into malignant and nonmalignant hematologic diseases and disorders. In the malignant category, the various lymphomas and leukemias are individually addressed, as well as other conditions such as myelodysplastic syndromes, myeloproliferative neoplasms, and multiple myeloma. In the nonmalignant category, ASH has provided FAQ on aplastic anemia, thalassemia, sickle cell disease, pulmonary embolism, venous thromboembolism/anticoagulation, coagulopathy, and immune as well as thrombotic thrombocytopenic purpura.
In addition to the continually updated series of relevant FAQ, as part of its response to the pandemic ASH is promoting two unique COVID-19 registries for physicians: the ASH Research Collaborative’s (ASH RC) Data Hub COVID-19 Registry and the Surveillance Epidemiology of Coronavirus (COVID-19) Under Research Exclusion Sickle Cell Disease (SECURE-SCD) Registry.
“The ASH Research Collaborative’s (ASH RC) Data Hub launched the COVID-19 Registry and is currently capturing data on people who test positive for COVID-19 and have been or are currently being treated for hematologic malignancy,” according to the website. The intention is to provide “near real-time observational data summaries,” which will hopefully provide useful information to clinicians treating hematologic malignancies in patients in the midst of the COVID-19 pandemic.
The registry allows clinicians to enter their own cases in a specified format to allow data analysis on clinical practice and patient outcomes that will be aggregated to provide rapid insights for clinicians to help them care for their patients, according to ASH.
The second registry specifically deals with COVID-19 cases in patients with sickle cell disease. It also allows clinicians to add cases with a similar intention of aggregating data to provide near real-time insights into patient care. “We are asking providers caring for these patients to report all of their cases of COVID-19 to this registry,” according to the registry website. The registry is for reporting COVID-19 cases in sickle cell disease patients “after sufficient time has passed to observe the disease course through resolution of acute illness and/or death.”
ASH also provides more generalized information for hematology practitioners dealing with COVID-19 on the topics of conducting their practice and using telemedicine, among others.
Correction, April 15, 2020: This story originally said incorrectly that ASH developed the 2 new registries. The registries are merely being promoted on the ASH website.
The American Society of Hematology has committed a portion of its website to providing continually updated information addressing specific hematologic disorders in relation to COVID-19.
“As the world grapples with the novel coronavirus, ASH believes that we can help each other be as knowledgeable and prepared as possible,” wrote the society’s president, Stephanie J. Lee, MD, MPH.
On its website, ASH provides relevant COVID-19 information in a series of FAQ divided into malignant and nonmalignant hematologic diseases and disorders. In the malignant category, the various lymphomas and leukemias are individually addressed, as well as other conditions such as myelodysplastic syndromes, myeloproliferative neoplasms, and multiple myeloma. In the nonmalignant category, ASH has provided FAQ on aplastic anemia, thalassemia, sickle cell disease, pulmonary embolism, venous thromboembolism/anticoagulation, coagulopathy, and immune as well as thrombotic thrombocytopenic purpura.
In addition to the continually updated series of relevant FAQ, as part of its response to the pandemic ASH is promoting two unique COVID-19 registries for physicians: the ASH Research Collaborative’s (ASH RC) Data Hub COVID-19 Registry and the Surveillance Epidemiology of Coronavirus (COVID-19) Under Research Exclusion Sickle Cell Disease (SECURE-SCD) Registry.
“The ASH Research Collaborative’s (ASH RC) Data Hub launched the COVID-19 Registry and is currently capturing data on people who test positive for COVID-19 and have been or are currently being treated for hematologic malignancy,” according to the website. The intention is to provide “near real-time observational data summaries,” which will hopefully provide useful information to clinicians treating hematologic malignancies in patients in the midst of the COVID-19 pandemic.
The registry allows clinicians to enter their own cases in a specified format to allow data analysis on clinical practice and patient outcomes that will be aggregated to provide rapid insights for clinicians to help them care for their patients, according to ASH.
The second registry specifically deals with COVID-19 cases in patients with sickle cell disease. It also allows clinicians to add cases with a similar intention of aggregating data to provide near real-time insights into patient care. “We are asking providers caring for these patients to report all of their cases of COVID-19 to this registry,” according to the registry website. The registry is for reporting COVID-19 cases in sickle cell disease patients “after sufficient time has passed to observe the disease course through resolution of acute illness and/or death.”
ASH also provides more generalized information for hematology practitioners dealing with COVID-19 on the topics of conducting their practice and using telemedicine, among others.
Correction, April 15, 2020: This story originally said incorrectly that ASH developed the 2 new registries. The registries are merely being promoted on the ASH website.
The American Society of Hematology has committed a portion of its website to providing continually updated information addressing specific hematologic disorders in relation to COVID-19.
“As the world grapples with the novel coronavirus, ASH believes that we can help each other be as knowledgeable and prepared as possible,” wrote the society’s president, Stephanie J. Lee, MD, MPH.
On its website, ASH provides relevant COVID-19 information in a series of FAQ divided into malignant and nonmalignant hematologic diseases and disorders. In the malignant category, the various lymphomas and leukemias are individually addressed, as well as other conditions such as myelodysplastic syndromes, myeloproliferative neoplasms, and multiple myeloma. In the nonmalignant category, ASH has provided FAQ on aplastic anemia, thalassemia, sickle cell disease, pulmonary embolism, venous thromboembolism/anticoagulation, coagulopathy, and immune as well as thrombotic thrombocytopenic purpura.
In addition to the continually updated series of relevant FAQ, as part of its response to the pandemic ASH is promoting two unique COVID-19 registries for physicians: the ASH Research Collaborative’s (ASH RC) Data Hub COVID-19 Registry and the Surveillance Epidemiology of Coronavirus (COVID-19) Under Research Exclusion Sickle Cell Disease (SECURE-SCD) Registry.
“The ASH Research Collaborative’s (ASH RC) Data Hub launched the COVID-19 Registry and is currently capturing data on people who test positive for COVID-19 and have been or are currently being treated for hematologic malignancy,” according to the website. The intention is to provide “near real-time observational data summaries,” which will hopefully provide useful information to clinicians treating hematologic malignancies in patients in the midst of the COVID-19 pandemic.
The registry allows clinicians to enter their own cases in a specified format to allow data analysis on clinical practice and patient outcomes that will be aggregated to provide rapid insights for clinicians to help them care for their patients, according to ASH.
The second registry specifically deals with COVID-19 cases in patients with sickle cell disease. It also allows clinicians to add cases with a similar intention of aggregating data to provide near real-time insights into patient care. “We are asking providers caring for these patients to report all of their cases of COVID-19 to this registry,” according to the registry website. The registry is for reporting COVID-19 cases in sickle cell disease patients “after sufficient time has passed to observe the disease course through resolution of acute illness and/or death.”
ASH also provides more generalized information for hematology practitioners dealing with COVID-19 on the topics of conducting their practice and using telemedicine, among others.
Correction, April 15, 2020: This story originally said incorrectly that ASH developed the 2 new registries. The registries are merely being promoted on the ASH website.
Managing gynecologic cancers during the COVID-19 pandemic
To manage patients with gynecologic cancers, oncologists in the United States and Europe are recommending reducing outpatient visits, delaying surgeries, prolonging chemotherapy regimens, and generally trying to keep cancer patients away from those who have tested positive for COVID-19.
“We recognize that, in this special situation, we must continue to provide our gynecologic oncology patients with the highest quality of medical services,” Pedro T. Ramirez, MD, of the University of Texas MD Anderson Cancer Center in Houston and associates wrote in an editorial published in the International Journal of Gynecological Cancer.
At the same time, the authors added, the safety of patients, their families, and medical staff needs to be assured.
Dr. Ramirez and colleagues’ editorial includes recommendations on how to optimize the care of patients with gynecologic cancers while prioritizing safety and minimizing the burden to the healthcare system. The group’s recommendations outline when surgery, radiotherapy, and other treatments might be safely postponed and when they need to proceed out of urgency.
Some authors of the editorial also described their experiences with COVID-19 during a webinar on managing patients with advanced ovarian cancer, which was hosted by the European Society of Gynaecological Oncology (ESGO).
A lack of resources
In Spain, health resources “are collapsed” by the pandemic, editorial author Luis Chiva, MD, said during the webinar.
At his institution, the Clínica Universidad de Navarra in Madrid, 98% of the 1,500 intensive care beds were occupied by COVID-19 patients at the end of March. So the hope was to be able to refer their patients to other communities where there may still be some capacity.
Another problem in Spain is the high percentage of health workers infected with SARS-CoV-2, the virus behind COVID-19. More than 15,000 health workers were recently reported to be sick or self-isolating, which is around 14% of the health care workforce in the country.
Dr. Chiva noted that this puts those treating gynecologic cancers in a difficult position. On the one hand, surgery to remove a high-risk ovarian mass should not be delayed, but the majority of hospitals in Spain simply cannot perform this type of surgery during the pandemic.
“Unfortunately, due to this specific situation, almost, I would say in 80%-90% of hospitals, we are only able to carry out emergency surgical procedures,” Dr. Chiva said. That’s general emergency procedures such as appendectomies, removing blockages, and dealing with hemorrhages, not gynecologic surgeries. “It’s almost impossible to schedule the typical oncological cases,” he said.
Even with the Hospital IFEMA now set up at the Feria de Madrid, which is usually used to host large-scale events, there are “minimal options for performing standard oncological surgery,” Dr. Chiva said. He estimated that just 5% of hospitals in Spain are able to perform oncologic surgeries as normal, with maybe 15% able to offer surgery without the backup of postsurgical intensive care.
‘Ring-fencing’
“This is really an unusual time for us,” commented Jonathan Ledermann, MD, vice president of ESGO and a professor of medical oncology at University College London, who moderated the webinar.
“This is affecting the way in which we diagnose our patients and have access to care,” he said. “It compromises the way in which we treat patients. We have to adjust our treatment pathways. We have to look at the risks of coronavirus infection in cancer patients and how we manage patients in a socially distancing environment. We also need to think about managing gynecological oncology departments in the face of disease amongst staff, the risks of transmission, and the reduced clinical service.”
Dr. Ledermann noted that “ring-fencing” a few hospitals to deal only with patients free of COVID-19 might be a way forward. This approach has been used in Northern Italy and was recently started in London.
“We try to divide and have separate access between COVID-positive and -negative patients,” said Anna Fagotti, MD, an assistant professor at Fondazione Policlinico Universitario Agostino Gemelli IRCCS in Rome and another coauthor of the editorial.
“We are trying to divide the work flow of patients and try to ensure treatment to cancer patients as much as we can,” she explained. “This means that it’s a very difficult situation, and, every time, you have to deal with the number of places available as some places have been taken by other patients from the emergency room. We are still trying to have a number of beds and intensive care unit beds available for our patients.”
Setting up dedicated hospitals is a good idea, but it has to be done before the “tsunami” of cases hits and there are no more intensive care beds or ventilators, according to Antonio González-Martín, MD, of Clínica Universidad de Navarra in Madrid, another coauthor of the editorial.
Limiting hospital visits
Strategies to limit the number of times patients need to come into hospital for appointments and treatment is key to getting through the pandemic, Sandro Pignata, MD, of Instituto Nazionale Tumori IRCCS Fondazione G. Pascale in Naples, Italy, said during the webinar.
“It will be imperative to explore options that reduce the number of procedures or surgical interventions that may be associated with prolonged operative time, risk of major blood loss, necessitating blood products, risk of infection to the medical personnel, or admission to intensive care units,” Dr. Ramirez and colleagues wrote in their editorial.
“In considering management of disease, we must recognize that, in many centers, access to routine visits and surgery may be either completely restricted or significantly reduced. We must, therefore, consider options that may still offer our patients a treatment plan that addresses their disease while at the same time limiting risk of exposure,” the authors wrote.
The authors declared no competing interests or specific funding in relation to their work, and the webinar participants had no conflicts of interest.
SOURCE: Ramirez PT et al. Int J Gynecol Cancer. 2020 Mar 27. doi: 10.1136/ijgc-2020-001419.
To manage patients with gynecologic cancers, oncologists in the United States and Europe are recommending reducing outpatient visits, delaying surgeries, prolonging chemotherapy regimens, and generally trying to keep cancer patients away from those who have tested positive for COVID-19.
“We recognize that, in this special situation, we must continue to provide our gynecologic oncology patients with the highest quality of medical services,” Pedro T. Ramirez, MD, of the University of Texas MD Anderson Cancer Center in Houston and associates wrote in an editorial published in the International Journal of Gynecological Cancer.
At the same time, the authors added, the safety of patients, their families, and medical staff needs to be assured.
Dr. Ramirez and colleagues’ editorial includes recommendations on how to optimize the care of patients with gynecologic cancers while prioritizing safety and minimizing the burden to the healthcare system. The group’s recommendations outline when surgery, radiotherapy, and other treatments might be safely postponed and when they need to proceed out of urgency.
Some authors of the editorial also described their experiences with COVID-19 during a webinar on managing patients with advanced ovarian cancer, which was hosted by the European Society of Gynaecological Oncology (ESGO).
A lack of resources
In Spain, health resources “are collapsed” by the pandemic, editorial author Luis Chiva, MD, said during the webinar.
At his institution, the Clínica Universidad de Navarra in Madrid, 98% of the 1,500 intensive care beds were occupied by COVID-19 patients at the end of March. So the hope was to be able to refer their patients to other communities where there may still be some capacity.
Another problem in Spain is the high percentage of health workers infected with SARS-CoV-2, the virus behind COVID-19. More than 15,000 health workers were recently reported to be sick or self-isolating, which is around 14% of the health care workforce in the country.
Dr. Chiva noted that this puts those treating gynecologic cancers in a difficult position. On the one hand, surgery to remove a high-risk ovarian mass should not be delayed, but the majority of hospitals in Spain simply cannot perform this type of surgery during the pandemic.
“Unfortunately, due to this specific situation, almost, I would say in 80%-90% of hospitals, we are only able to carry out emergency surgical procedures,” Dr. Chiva said. That’s general emergency procedures such as appendectomies, removing blockages, and dealing with hemorrhages, not gynecologic surgeries. “It’s almost impossible to schedule the typical oncological cases,” he said.
Even with the Hospital IFEMA now set up at the Feria de Madrid, which is usually used to host large-scale events, there are “minimal options for performing standard oncological surgery,” Dr. Chiva said. He estimated that just 5% of hospitals in Spain are able to perform oncologic surgeries as normal, with maybe 15% able to offer surgery without the backup of postsurgical intensive care.
‘Ring-fencing’
“This is really an unusual time for us,” commented Jonathan Ledermann, MD, vice president of ESGO and a professor of medical oncology at University College London, who moderated the webinar.
“This is affecting the way in which we diagnose our patients and have access to care,” he said. “It compromises the way in which we treat patients. We have to adjust our treatment pathways. We have to look at the risks of coronavirus infection in cancer patients and how we manage patients in a socially distancing environment. We also need to think about managing gynecological oncology departments in the face of disease amongst staff, the risks of transmission, and the reduced clinical service.”
Dr. Ledermann noted that “ring-fencing” a few hospitals to deal only with patients free of COVID-19 might be a way forward. This approach has been used in Northern Italy and was recently started in London.
“We try to divide and have separate access between COVID-positive and -negative patients,” said Anna Fagotti, MD, an assistant professor at Fondazione Policlinico Universitario Agostino Gemelli IRCCS in Rome and another coauthor of the editorial.
“We are trying to divide the work flow of patients and try to ensure treatment to cancer patients as much as we can,” she explained. “This means that it’s a very difficult situation, and, every time, you have to deal with the number of places available as some places have been taken by other patients from the emergency room. We are still trying to have a number of beds and intensive care unit beds available for our patients.”
Setting up dedicated hospitals is a good idea, but it has to be done before the “tsunami” of cases hits and there are no more intensive care beds or ventilators, according to Antonio González-Martín, MD, of Clínica Universidad de Navarra in Madrid, another coauthor of the editorial.
Limiting hospital visits
Strategies to limit the number of times patients need to come into hospital for appointments and treatment is key to getting through the pandemic, Sandro Pignata, MD, of Instituto Nazionale Tumori IRCCS Fondazione G. Pascale in Naples, Italy, said during the webinar.
“It will be imperative to explore options that reduce the number of procedures or surgical interventions that may be associated with prolonged operative time, risk of major blood loss, necessitating blood products, risk of infection to the medical personnel, or admission to intensive care units,” Dr. Ramirez and colleagues wrote in their editorial.
“In considering management of disease, we must recognize that, in many centers, access to routine visits and surgery may be either completely restricted or significantly reduced. We must, therefore, consider options that may still offer our patients a treatment plan that addresses their disease while at the same time limiting risk of exposure,” the authors wrote.
The authors declared no competing interests or specific funding in relation to their work, and the webinar participants had no conflicts of interest.
SOURCE: Ramirez PT et al. Int J Gynecol Cancer. 2020 Mar 27. doi: 10.1136/ijgc-2020-001419.
To manage patients with gynecologic cancers, oncologists in the United States and Europe are recommending reducing outpatient visits, delaying surgeries, prolonging chemotherapy regimens, and generally trying to keep cancer patients away from those who have tested positive for COVID-19.
“We recognize that, in this special situation, we must continue to provide our gynecologic oncology patients with the highest quality of medical services,” Pedro T. Ramirez, MD, of the University of Texas MD Anderson Cancer Center in Houston and associates wrote in an editorial published in the International Journal of Gynecological Cancer.
At the same time, the authors added, the safety of patients, their families, and medical staff needs to be assured.
Dr. Ramirez and colleagues’ editorial includes recommendations on how to optimize the care of patients with gynecologic cancers while prioritizing safety and minimizing the burden to the healthcare system. The group’s recommendations outline when surgery, radiotherapy, and other treatments might be safely postponed and when they need to proceed out of urgency.
Some authors of the editorial also described their experiences with COVID-19 during a webinar on managing patients with advanced ovarian cancer, which was hosted by the European Society of Gynaecological Oncology (ESGO).
A lack of resources
In Spain, health resources “are collapsed” by the pandemic, editorial author Luis Chiva, MD, said during the webinar.
At his institution, the Clínica Universidad de Navarra in Madrid, 98% of the 1,500 intensive care beds were occupied by COVID-19 patients at the end of March. So the hope was to be able to refer their patients to other communities where there may still be some capacity.
Another problem in Spain is the high percentage of health workers infected with SARS-CoV-2, the virus behind COVID-19. More than 15,000 health workers were recently reported to be sick or self-isolating, which is around 14% of the health care workforce in the country.
Dr. Chiva noted that this puts those treating gynecologic cancers in a difficult position. On the one hand, surgery to remove a high-risk ovarian mass should not be delayed, but the majority of hospitals in Spain simply cannot perform this type of surgery during the pandemic.
“Unfortunately, due to this specific situation, almost, I would say in 80%-90% of hospitals, we are only able to carry out emergency surgical procedures,” Dr. Chiva said. That’s general emergency procedures such as appendectomies, removing blockages, and dealing with hemorrhages, not gynecologic surgeries. “It’s almost impossible to schedule the typical oncological cases,” he said.
Even with the Hospital IFEMA now set up at the Feria de Madrid, which is usually used to host large-scale events, there are “minimal options for performing standard oncological surgery,” Dr. Chiva said. He estimated that just 5% of hospitals in Spain are able to perform oncologic surgeries as normal, with maybe 15% able to offer surgery without the backup of postsurgical intensive care.
‘Ring-fencing’
“This is really an unusual time for us,” commented Jonathan Ledermann, MD, vice president of ESGO and a professor of medical oncology at University College London, who moderated the webinar.
“This is affecting the way in which we diagnose our patients and have access to care,” he said. “It compromises the way in which we treat patients. We have to adjust our treatment pathways. We have to look at the risks of coronavirus infection in cancer patients and how we manage patients in a socially distancing environment. We also need to think about managing gynecological oncology departments in the face of disease amongst staff, the risks of transmission, and the reduced clinical service.”
Dr. Ledermann noted that “ring-fencing” a few hospitals to deal only with patients free of COVID-19 might be a way forward. This approach has been used in Northern Italy and was recently started in London.
“We try to divide and have separate access between COVID-positive and -negative patients,” said Anna Fagotti, MD, an assistant professor at Fondazione Policlinico Universitario Agostino Gemelli IRCCS in Rome and another coauthor of the editorial.
“We are trying to divide the work flow of patients and try to ensure treatment to cancer patients as much as we can,” she explained. “This means that it’s a very difficult situation, and, every time, you have to deal with the number of places available as some places have been taken by other patients from the emergency room. We are still trying to have a number of beds and intensive care unit beds available for our patients.”
Setting up dedicated hospitals is a good idea, but it has to be done before the “tsunami” of cases hits and there are no more intensive care beds or ventilators, according to Antonio González-Martín, MD, of Clínica Universidad de Navarra in Madrid, another coauthor of the editorial.
Limiting hospital visits
Strategies to limit the number of times patients need to come into hospital for appointments and treatment is key to getting through the pandemic, Sandro Pignata, MD, of Instituto Nazionale Tumori IRCCS Fondazione G. Pascale in Naples, Italy, said during the webinar.
“It will be imperative to explore options that reduce the number of procedures or surgical interventions that may be associated with prolonged operative time, risk of major blood loss, necessitating blood products, risk of infection to the medical personnel, or admission to intensive care units,” Dr. Ramirez and colleagues wrote in their editorial.
“In considering management of disease, we must recognize that, in many centers, access to routine visits and surgery may be either completely restricted or significantly reduced. We must, therefore, consider options that may still offer our patients a treatment plan that addresses their disease while at the same time limiting risk of exposure,” the authors wrote.
The authors declared no competing interests or specific funding in relation to their work, and the webinar participants had no conflicts of interest.
SOURCE: Ramirez PT et al. Int J Gynecol Cancer. 2020 Mar 27. doi: 10.1136/ijgc-2020-001419.
FROM THE INTERNATIONAL JOURNAL OF GYNECOLOGICAL CANCER
ASCO announces its own COVID-19 and cancer registry
Data will not be commercialized, unlike CancerLinQ
The American Society of Clinical Oncology (ASCO) has launched a registry to collect data on cancer patients with COVID-19 and is asking oncology practices across the United States to share information about their patients with the infection for educational purposes.
The new registry joins at least two other cancer and COVID-19 patient registries already underway in the U.S.
In a statement, ASCO President Howard “Skip” Burris III, MD said there is a need to know “how the virus is impacting our patients, their cancer treatment, and outcomes to inform current cancer care” and future care.
The web-based registry, known as the American Society of Clinical Oncology (ASCO) Survey on COVID-19 in Oncology Registry, is open to all U.S. oncology practices. Participating practices will receive an unspecified “nominal” payment for their data entry efforts.
The registry patient information will be stored on ASCO’s “Big Data” platform, known as CancerLinQ, but is being held apart from that pool of data. The registry information will not be available for commercial purposes, ASCO spokesperson Rachel Martin recently told Medscape Medical News.
Separately, CancerLinQ, which is a wholly owned subsidiary of ASCO, will continue to collect data from its participant oncology practices (as usual), including COVID-19 information.
CancerLinQ has been criticized by ethicists for allowing partner companies to sell access to its data (after stripping off patient identifiers), but without asking for patients’ permission, as reported last year by Medscape Medical News.
Eleven practices, including academic enterprises, have so far expressed interested in participating in the ASCO COVID-19 Registry.
Participating practices are requested to send in details about cancer patients with a confirmed COVID-19 diagnosis. As well as a baseline data capture form, they will need to provide details of subsequent status, treatment, and outcomes. Some patient-identifying data, including zip code, date of birth, gender, race, ethnicity, type of cancer, and comorbidities, will be collected for the purposes of analysis.
ASCO hopes to learn about characteristics of patients with cancer most impacted by COVID-19; estimates of disease severity; treatment modifications or delays; implementation of telemedicine in the cancer treatment setting; and clinical outcomes related to both COVID-19 and cancer.
ASCO says it will deliver periodic reports to the cancer community and the broader public on these and other “key learnings.” It also says that the registry is designed to capture point-in-time data as well as longitudinal data on how the virus will impact care and outcomes into 2021.
ASCO is not alone in its data collection efforts.
The COVID-19 and Cancer Consortium is already collecting information from more than 50 cancer centers and organizations on COVID-19 in patients with cancer. The American Society of Hematology (ASH) Research Collaborative COVID-19 Registry for Hematologic Malignancy is doing the same but with a focus on hematologic malignancies.
This article first appeared on Medscape.com.
Data will not be commercialized, unlike CancerLinQ
Data will not be commercialized, unlike CancerLinQ
The American Society of Clinical Oncology (ASCO) has launched a registry to collect data on cancer patients with COVID-19 and is asking oncology practices across the United States to share information about their patients with the infection for educational purposes.
The new registry joins at least two other cancer and COVID-19 patient registries already underway in the U.S.
In a statement, ASCO President Howard “Skip” Burris III, MD said there is a need to know “how the virus is impacting our patients, their cancer treatment, and outcomes to inform current cancer care” and future care.
The web-based registry, known as the American Society of Clinical Oncology (ASCO) Survey on COVID-19 in Oncology Registry, is open to all U.S. oncology practices. Participating practices will receive an unspecified “nominal” payment for their data entry efforts.
The registry patient information will be stored on ASCO’s “Big Data” platform, known as CancerLinQ, but is being held apart from that pool of data. The registry information will not be available for commercial purposes, ASCO spokesperson Rachel Martin recently told Medscape Medical News.
Separately, CancerLinQ, which is a wholly owned subsidiary of ASCO, will continue to collect data from its participant oncology practices (as usual), including COVID-19 information.
CancerLinQ has been criticized by ethicists for allowing partner companies to sell access to its data (after stripping off patient identifiers), but without asking for patients’ permission, as reported last year by Medscape Medical News.
Eleven practices, including academic enterprises, have so far expressed interested in participating in the ASCO COVID-19 Registry.
Participating practices are requested to send in details about cancer patients with a confirmed COVID-19 diagnosis. As well as a baseline data capture form, they will need to provide details of subsequent status, treatment, and outcomes. Some patient-identifying data, including zip code, date of birth, gender, race, ethnicity, type of cancer, and comorbidities, will be collected for the purposes of analysis.
ASCO hopes to learn about characteristics of patients with cancer most impacted by COVID-19; estimates of disease severity; treatment modifications or delays; implementation of telemedicine in the cancer treatment setting; and clinical outcomes related to both COVID-19 and cancer.
ASCO says it will deliver periodic reports to the cancer community and the broader public on these and other “key learnings.” It also says that the registry is designed to capture point-in-time data as well as longitudinal data on how the virus will impact care and outcomes into 2021.
ASCO is not alone in its data collection efforts.
The COVID-19 and Cancer Consortium is already collecting information from more than 50 cancer centers and organizations on COVID-19 in patients with cancer. The American Society of Hematology (ASH) Research Collaborative COVID-19 Registry for Hematologic Malignancy is doing the same but with a focus on hematologic malignancies.
This article first appeared on Medscape.com.
The American Society of Clinical Oncology (ASCO) has launched a registry to collect data on cancer patients with COVID-19 and is asking oncology practices across the United States to share information about their patients with the infection for educational purposes.
The new registry joins at least two other cancer and COVID-19 patient registries already underway in the U.S.
In a statement, ASCO President Howard “Skip” Burris III, MD said there is a need to know “how the virus is impacting our patients, their cancer treatment, and outcomes to inform current cancer care” and future care.
The web-based registry, known as the American Society of Clinical Oncology (ASCO) Survey on COVID-19 in Oncology Registry, is open to all U.S. oncology practices. Participating practices will receive an unspecified “nominal” payment for their data entry efforts.
The registry patient information will be stored on ASCO’s “Big Data” platform, known as CancerLinQ, but is being held apart from that pool of data. The registry information will not be available for commercial purposes, ASCO spokesperson Rachel Martin recently told Medscape Medical News.
Separately, CancerLinQ, which is a wholly owned subsidiary of ASCO, will continue to collect data from its participant oncology practices (as usual), including COVID-19 information.
CancerLinQ has been criticized by ethicists for allowing partner companies to sell access to its data (after stripping off patient identifiers), but without asking for patients’ permission, as reported last year by Medscape Medical News.
Eleven practices, including academic enterprises, have so far expressed interested in participating in the ASCO COVID-19 Registry.
Participating practices are requested to send in details about cancer patients with a confirmed COVID-19 diagnosis. As well as a baseline data capture form, they will need to provide details of subsequent status, treatment, and outcomes. Some patient-identifying data, including zip code, date of birth, gender, race, ethnicity, type of cancer, and comorbidities, will be collected for the purposes of analysis.
ASCO hopes to learn about characteristics of patients with cancer most impacted by COVID-19; estimates of disease severity; treatment modifications or delays; implementation of telemedicine in the cancer treatment setting; and clinical outcomes related to both COVID-19 and cancer.
ASCO says it will deliver periodic reports to the cancer community and the broader public on these and other “key learnings.” It also says that the registry is designed to capture point-in-time data as well as longitudinal data on how the virus will impact care and outcomes into 2021.
ASCO is not alone in its data collection efforts.
The COVID-19 and Cancer Consortium is already collecting information from more than 50 cancer centers and organizations on COVID-19 in patients with cancer. The American Society of Hematology (ASH) Research Collaborative COVID-19 Registry for Hematologic Malignancy is doing the same but with a focus on hematologic malignancies.
This article first appeared on Medscape.com.
Treating rectal cancer in the COVID-19 era: Expert guidance
As the COVID-19 pandemic continues, minimizing risks of infection to patients with cancer while maintaining good outcomes remains a priority. An international panel of experts has now issued recommendations for treating patients with rectal cancer, which includes using a short pre-operative course of radiotherapy (SCRT) and then delaying surgery.
Using SCRT translates to fewer hospital appointments, which will keep patients safer and allow them to maintain social distancing. The panel also found that surgery can be safely delayed by up to 12 weeks, and thus will allow procedures to be rescheduled after the pandemic peaks.
“The COVID-19 pandemic is a global emergency and we needed to work very quickly to identify changes that would benefit patients,” said David Sebag-Montefiore, MD, a professor of clinical oncology at the University of Leeds and honorary clinical oncologist with the Leeds Teaching Hospitals NHS Trust, who led the 15 member panel. “Our recommendations were published 20 days after our first meeting.”
“This process normally takes many months, if not years,” he said in a statement.
The recommendations were published online April 2 in Radiotherapy and Oncology.
The panel used the European Society for Medical Oncology (ESMO) rectal cancer guidelines as a framework to describe these new recommendations.
Recommendations by Stage
The recommendations were categorized into four subgroups based on cancer stage.
Early stage
- The ESMO guidelines recommend total mesorectal excision (TME) surgery without pre-operative radiotherapy for most cases.
- Panel recommendation also strongly supports the use of TME without pre-operative radiotherapy.
Intermediate stage
- The ESMO guidelines recommend TME alone or combined with SCRT or conventional radiotherapy (CRT) if there is uncertainty that a good quality mesorectal excision can be achieved.
- The panel strongly recommends TME alone in regions where high quality surgery is performed. The use of radiotherapy in this subgroup requires careful discussion, as the benefits of preoperative radiotherapy are likely to be small. If radiotherapy is used, then the preferred option should be SCRT.
Locally advanced
- The ESMO guideline recommends either pre-operative SCRT or CRT.
- The panel strongly recommends the use of SCRT and notes two phase 3 trials have compared SCRT and CRT and showed comparable outcomes for local recurrence, disease-free survival, overall survival, and late toxicity. In the COVID-19 setting, the panel points out that SCRT has many advantages over CRT, namely that there is less acute toxicity, fewer treatments which translate to less travel and contact with other patients and staff, and a significantly reduced risk of COVID-19 infection during treatment.
Timing of surgery after SCRT
- The ESMO guideline does not have any recommendations as they were issued before the Stockholm III trial (Lancet Oncol. 2017;18:336-46).
- The panel notes that the use of SCRT and delaying surgery has advantages that can be beneficial in both routine clinical practice and the COVID-19 setting. Several clinical trials have recommended that surgery should be performed within 3-7 days of completing radiotherapy, but the Stockholm III trial reported no difference in outcomes when surgery was delayed. It compared surgery performed within 1 week versus 4-8 weeks following SCRT and there was no difference in any survival endpoints. In addition, a longer delay to surgery was associated with a reduction in post-operative and surgical morbidity although no differences in severe complications or re-operations.
Advanced subgroup
- The ESMO guidelines recommend the use of pre-operative CRT or SCRT followed by neoadjuvant chemotherapy. CRT should be given as a fluoropyrimidine (usually capecitabine) combined with radiotherapy of 45-50.4 Gy over 5-5.5 weeks. Adjuvant chemotherapy should be considered but there is wide international variation in its use.
- The panel recommends that two options be considered based on the current evidence. The first is pre-op CRT, which is the most established standard of care, with the duration of concurrent capecitabine chemotherapy limited to 5-5.5 weeks. The second option is SCRT with or without neoadjuvant chemotherapy. In this case, the duration of radiotherapy is substantially less and has advantages versus CRT. “We consider both options to be acceptable but note the advantages of using SCRT in the COVID-19 setting,” the authors write. “The decision to use neoadjuvant chemotherapy in option 2 will reflect the attitudes to neoadjuvant and adjuvant chemotherapy in each country, the assessment of the risk-benefit ratio, considering the risk factors for COVID-19 increased mortality, and the capacity and prioritization of chemotherapy delivery.”
Organ Preservation
Organ preservation is being increasingly considered when a complete clinical response is achieved after CRT or SCRT, the panel points out. “An organ preservation approach may be considered during the COVID-19 period providing that resources for an adequate surveillance including imaging and endoscopy are available to detect local failures that require salvage surgery,” they write.
This article first appeared on Medscape.com.
As the COVID-19 pandemic continues, minimizing risks of infection to patients with cancer while maintaining good outcomes remains a priority. An international panel of experts has now issued recommendations for treating patients with rectal cancer, which includes using a short pre-operative course of radiotherapy (SCRT) and then delaying surgery.
Using SCRT translates to fewer hospital appointments, which will keep patients safer and allow them to maintain social distancing. The panel also found that surgery can be safely delayed by up to 12 weeks, and thus will allow procedures to be rescheduled after the pandemic peaks.
“The COVID-19 pandemic is a global emergency and we needed to work very quickly to identify changes that would benefit patients,” said David Sebag-Montefiore, MD, a professor of clinical oncology at the University of Leeds and honorary clinical oncologist with the Leeds Teaching Hospitals NHS Trust, who led the 15 member panel. “Our recommendations were published 20 days after our first meeting.”
“This process normally takes many months, if not years,” he said in a statement.
The recommendations were published online April 2 in Radiotherapy and Oncology.
The panel used the European Society for Medical Oncology (ESMO) rectal cancer guidelines as a framework to describe these new recommendations.
Recommendations by Stage
The recommendations were categorized into four subgroups based on cancer stage.
Early stage
- The ESMO guidelines recommend total mesorectal excision (TME) surgery without pre-operative radiotherapy for most cases.
- Panel recommendation also strongly supports the use of TME without pre-operative radiotherapy.
Intermediate stage
- The ESMO guidelines recommend TME alone or combined with SCRT or conventional radiotherapy (CRT) if there is uncertainty that a good quality mesorectal excision can be achieved.
- The panel strongly recommends TME alone in regions where high quality surgery is performed. The use of radiotherapy in this subgroup requires careful discussion, as the benefits of preoperative radiotherapy are likely to be small. If radiotherapy is used, then the preferred option should be SCRT.
Locally advanced
- The ESMO guideline recommends either pre-operative SCRT or CRT.
- The panel strongly recommends the use of SCRT and notes two phase 3 trials have compared SCRT and CRT and showed comparable outcomes for local recurrence, disease-free survival, overall survival, and late toxicity. In the COVID-19 setting, the panel points out that SCRT has many advantages over CRT, namely that there is less acute toxicity, fewer treatments which translate to less travel and contact with other patients and staff, and a significantly reduced risk of COVID-19 infection during treatment.
Timing of surgery after SCRT
- The ESMO guideline does not have any recommendations as they were issued before the Stockholm III trial (Lancet Oncol. 2017;18:336-46).
- The panel notes that the use of SCRT and delaying surgery has advantages that can be beneficial in both routine clinical practice and the COVID-19 setting. Several clinical trials have recommended that surgery should be performed within 3-7 days of completing radiotherapy, but the Stockholm III trial reported no difference in outcomes when surgery was delayed. It compared surgery performed within 1 week versus 4-8 weeks following SCRT and there was no difference in any survival endpoints. In addition, a longer delay to surgery was associated with a reduction in post-operative and surgical morbidity although no differences in severe complications or re-operations.
Advanced subgroup
- The ESMO guidelines recommend the use of pre-operative CRT or SCRT followed by neoadjuvant chemotherapy. CRT should be given as a fluoropyrimidine (usually capecitabine) combined with radiotherapy of 45-50.4 Gy over 5-5.5 weeks. Adjuvant chemotherapy should be considered but there is wide international variation in its use.
- The panel recommends that two options be considered based on the current evidence. The first is pre-op CRT, which is the most established standard of care, with the duration of concurrent capecitabine chemotherapy limited to 5-5.5 weeks. The second option is SCRT with or without neoadjuvant chemotherapy. In this case, the duration of radiotherapy is substantially less and has advantages versus CRT. “We consider both options to be acceptable but note the advantages of using SCRT in the COVID-19 setting,” the authors write. “The decision to use neoadjuvant chemotherapy in option 2 will reflect the attitudes to neoadjuvant and adjuvant chemotherapy in each country, the assessment of the risk-benefit ratio, considering the risk factors for COVID-19 increased mortality, and the capacity and prioritization of chemotherapy delivery.”
Organ Preservation
Organ preservation is being increasingly considered when a complete clinical response is achieved after CRT or SCRT, the panel points out. “An organ preservation approach may be considered during the COVID-19 period providing that resources for an adequate surveillance including imaging and endoscopy are available to detect local failures that require salvage surgery,” they write.
This article first appeared on Medscape.com.
As the COVID-19 pandemic continues, minimizing risks of infection to patients with cancer while maintaining good outcomes remains a priority. An international panel of experts has now issued recommendations for treating patients with rectal cancer, which includes using a short pre-operative course of radiotherapy (SCRT) and then delaying surgery.
Using SCRT translates to fewer hospital appointments, which will keep patients safer and allow them to maintain social distancing. The panel also found that surgery can be safely delayed by up to 12 weeks, and thus will allow procedures to be rescheduled after the pandemic peaks.
“The COVID-19 pandemic is a global emergency and we needed to work very quickly to identify changes that would benefit patients,” said David Sebag-Montefiore, MD, a professor of clinical oncology at the University of Leeds and honorary clinical oncologist with the Leeds Teaching Hospitals NHS Trust, who led the 15 member panel. “Our recommendations were published 20 days after our first meeting.”
“This process normally takes many months, if not years,” he said in a statement.
The recommendations were published online April 2 in Radiotherapy and Oncology.
The panel used the European Society for Medical Oncology (ESMO) rectal cancer guidelines as a framework to describe these new recommendations.
Recommendations by Stage
The recommendations were categorized into four subgroups based on cancer stage.
Early stage
- The ESMO guidelines recommend total mesorectal excision (TME) surgery without pre-operative radiotherapy for most cases.
- Panel recommendation also strongly supports the use of TME without pre-operative radiotherapy.
Intermediate stage
- The ESMO guidelines recommend TME alone or combined with SCRT or conventional radiotherapy (CRT) if there is uncertainty that a good quality mesorectal excision can be achieved.
- The panel strongly recommends TME alone in regions where high quality surgery is performed. The use of radiotherapy in this subgroup requires careful discussion, as the benefits of preoperative radiotherapy are likely to be small. If radiotherapy is used, then the preferred option should be SCRT.
Locally advanced
- The ESMO guideline recommends either pre-operative SCRT or CRT.
- The panel strongly recommends the use of SCRT and notes two phase 3 trials have compared SCRT and CRT and showed comparable outcomes for local recurrence, disease-free survival, overall survival, and late toxicity. In the COVID-19 setting, the panel points out that SCRT has many advantages over CRT, namely that there is less acute toxicity, fewer treatments which translate to less travel and contact with other patients and staff, and a significantly reduced risk of COVID-19 infection during treatment.
Timing of surgery after SCRT
- The ESMO guideline does not have any recommendations as they were issued before the Stockholm III trial (Lancet Oncol. 2017;18:336-46).
- The panel notes that the use of SCRT and delaying surgery has advantages that can be beneficial in both routine clinical practice and the COVID-19 setting. Several clinical trials have recommended that surgery should be performed within 3-7 days of completing radiotherapy, but the Stockholm III trial reported no difference in outcomes when surgery was delayed. It compared surgery performed within 1 week versus 4-8 weeks following SCRT and there was no difference in any survival endpoints. In addition, a longer delay to surgery was associated with a reduction in post-operative and surgical morbidity although no differences in severe complications or re-operations.
Advanced subgroup
- The ESMO guidelines recommend the use of pre-operative CRT or SCRT followed by neoadjuvant chemotherapy. CRT should be given as a fluoropyrimidine (usually capecitabine) combined with radiotherapy of 45-50.4 Gy over 5-5.5 weeks. Adjuvant chemotherapy should be considered but there is wide international variation in its use.
- The panel recommends that two options be considered based on the current evidence. The first is pre-op CRT, which is the most established standard of care, with the duration of concurrent capecitabine chemotherapy limited to 5-5.5 weeks. The second option is SCRT with or without neoadjuvant chemotherapy. In this case, the duration of radiotherapy is substantially less and has advantages versus CRT. “We consider both options to be acceptable but note the advantages of using SCRT in the COVID-19 setting,” the authors write. “The decision to use neoadjuvant chemotherapy in option 2 will reflect the attitudes to neoadjuvant and adjuvant chemotherapy in each country, the assessment of the risk-benefit ratio, considering the risk factors for COVID-19 increased mortality, and the capacity and prioritization of chemotherapy delivery.”
Organ Preservation
Organ preservation is being increasingly considered when a complete clinical response is achieved after CRT or SCRT, the panel points out. “An organ preservation approach may be considered during the COVID-19 period providing that resources for an adequate surveillance including imaging and endoscopy are available to detect local failures that require salvage surgery,” they write.
This article first appeared on Medscape.com.
Cancer prevalence among COVID-19 patients may be higher than previously reported
An early report pegged the prevalence of cancer among COVID-19 patients at 1%, but authors of a recent meta-analysis found an overall prevalence of 2% and up to 3% depending on the subset of data they reviewed.
However, those findings are limited by the retrospective nature of the studies published to date, according to the authors of the meta-analysis, led by Aakash Desai, MBBS, of the University of Connecticut, Farmington.
Nevertheless, the results do confirm that cancer patients and survivors are an important at-risk population for COVID-19, according to Dr. Desai and colleagues.
“We hope that additional data from China and Italy will provide information on the characteristics of patients with cancer at risk, types of cancer that confer higher risk, and systemic regimens that may increase COVID-19 infection complications,” the authors wrote in JCO Global Oncology.
More than 15 million individuals with cancer and many more cancer survivors are at increased risk of COVID-19 because of compromised immune systems, according to the authors.
Exactly how many individuals with cancer are among the COVID-19 cases remains unclear, though a previous report suggested the prevalence of cancer was 1% (95% confidence interval, 0.61%-1.65%) among COVID-19 patients in China (Lancet Oncol. 2020 Mar;21[3]:335-7). This “seems to be higher” than the 0.29% prevalence of cancer in the overall Chinese population, the investigators noted at the time.
That study revealed 18 cancer patients among 1,590 COVID-19 cases, though it was “hypothesis generating,” according to Dr. Desai and colleagues, who rolled that data into their meta-analysis of 11 reports including 3,661 COVID-19 cases.
Overall, Dr. Desai and colleagues found the pooled prevalence of cancer was 2.0% (95% CI, 2.0%-3.0%) in that population. In a subgroup analysis of five studies with sample sizes of less than 100 COVID-19 patients, the researchers found a “slightly higher” prevalence of 3.0% (95% CI, 1.0%-6.0%).
However, even that data wasn’t robust enough for Dr. Desai and colleagues to make any pronouncements on cancer prevalence. “Overall, current evidence on the association between cancer and COVID-19 remains inconclusive,” they wrote.
Though inconclusive, the findings raise questions about whether treatments or interventions might need to be postponed in certain patients, whether cancer patients and survivors need stronger personal protection, and how to deal with potential delays in cancer clinical trials, according to Dr. Desai and colleagues.
“As the evidence continues to rise, we must strive to answer the unanswered clinical questions,” the authors wrote.
Dr. Desai and colleagues reported no potential conflicts of interest related to the study.
SOURCE: Desai A et al. JCO Glob Oncol. 2020 Apr 6. doi: 10.1200/GO.20.00097.
An early report pegged the prevalence of cancer among COVID-19 patients at 1%, but authors of a recent meta-analysis found an overall prevalence of 2% and up to 3% depending on the subset of data they reviewed.
However, those findings are limited by the retrospective nature of the studies published to date, according to the authors of the meta-analysis, led by Aakash Desai, MBBS, of the University of Connecticut, Farmington.
Nevertheless, the results do confirm that cancer patients and survivors are an important at-risk population for COVID-19, according to Dr. Desai and colleagues.
“We hope that additional data from China and Italy will provide information on the characteristics of patients with cancer at risk, types of cancer that confer higher risk, and systemic regimens that may increase COVID-19 infection complications,” the authors wrote in JCO Global Oncology.
More than 15 million individuals with cancer and many more cancer survivors are at increased risk of COVID-19 because of compromised immune systems, according to the authors.
Exactly how many individuals with cancer are among the COVID-19 cases remains unclear, though a previous report suggested the prevalence of cancer was 1% (95% confidence interval, 0.61%-1.65%) among COVID-19 patients in China (Lancet Oncol. 2020 Mar;21[3]:335-7). This “seems to be higher” than the 0.29% prevalence of cancer in the overall Chinese population, the investigators noted at the time.
That study revealed 18 cancer patients among 1,590 COVID-19 cases, though it was “hypothesis generating,” according to Dr. Desai and colleagues, who rolled that data into their meta-analysis of 11 reports including 3,661 COVID-19 cases.
Overall, Dr. Desai and colleagues found the pooled prevalence of cancer was 2.0% (95% CI, 2.0%-3.0%) in that population. In a subgroup analysis of five studies with sample sizes of less than 100 COVID-19 patients, the researchers found a “slightly higher” prevalence of 3.0% (95% CI, 1.0%-6.0%).
However, even that data wasn’t robust enough for Dr. Desai and colleagues to make any pronouncements on cancer prevalence. “Overall, current evidence on the association between cancer and COVID-19 remains inconclusive,” they wrote.
Though inconclusive, the findings raise questions about whether treatments or interventions might need to be postponed in certain patients, whether cancer patients and survivors need stronger personal protection, and how to deal with potential delays in cancer clinical trials, according to Dr. Desai and colleagues.
“As the evidence continues to rise, we must strive to answer the unanswered clinical questions,” the authors wrote.
Dr. Desai and colleagues reported no potential conflicts of interest related to the study.
SOURCE: Desai A et al. JCO Glob Oncol. 2020 Apr 6. doi: 10.1200/GO.20.00097.
An early report pegged the prevalence of cancer among COVID-19 patients at 1%, but authors of a recent meta-analysis found an overall prevalence of 2% and up to 3% depending on the subset of data they reviewed.
However, those findings are limited by the retrospective nature of the studies published to date, according to the authors of the meta-analysis, led by Aakash Desai, MBBS, of the University of Connecticut, Farmington.
Nevertheless, the results do confirm that cancer patients and survivors are an important at-risk population for COVID-19, according to Dr. Desai and colleagues.
“We hope that additional data from China and Italy will provide information on the characteristics of patients with cancer at risk, types of cancer that confer higher risk, and systemic regimens that may increase COVID-19 infection complications,” the authors wrote in JCO Global Oncology.
More than 15 million individuals with cancer and many more cancer survivors are at increased risk of COVID-19 because of compromised immune systems, according to the authors.
Exactly how many individuals with cancer are among the COVID-19 cases remains unclear, though a previous report suggested the prevalence of cancer was 1% (95% confidence interval, 0.61%-1.65%) among COVID-19 patients in China (Lancet Oncol. 2020 Mar;21[3]:335-7). This “seems to be higher” than the 0.29% prevalence of cancer in the overall Chinese population, the investigators noted at the time.
That study revealed 18 cancer patients among 1,590 COVID-19 cases, though it was “hypothesis generating,” according to Dr. Desai and colleagues, who rolled that data into their meta-analysis of 11 reports including 3,661 COVID-19 cases.
Overall, Dr. Desai and colleagues found the pooled prevalence of cancer was 2.0% (95% CI, 2.0%-3.0%) in that population. In a subgroup analysis of five studies with sample sizes of less than 100 COVID-19 patients, the researchers found a “slightly higher” prevalence of 3.0% (95% CI, 1.0%-6.0%).
However, even that data wasn’t robust enough for Dr. Desai and colleagues to make any pronouncements on cancer prevalence. “Overall, current evidence on the association between cancer and COVID-19 remains inconclusive,” they wrote.
Though inconclusive, the findings raise questions about whether treatments or interventions might need to be postponed in certain patients, whether cancer patients and survivors need stronger personal protection, and how to deal with potential delays in cancer clinical trials, according to Dr. Desai and colleagues.
“As the evidence continues to rise, we must strive to answer the unanswered clinical questions,” the authors wrote.
Dr. Desai and colleagues reported no potential conflicts of interest related to the study.
SOURCE: Desai A et al. JCO Glob Oncol. 2020 Apr 6. doi: 10.1200/GO.20.00097.
FROM JCO GLOBAL ONCOLOGY
When to treat, delay, or omit breast cancer therapy in the face of COVID-19
Nothing is business as usual during the COVID-19 pandemic, and that includes breast cancer therapy. That’s why two groups have released guidance documents on treating breast cancer patients during the pandemic.
A guidance on surgery, drug therapy, and radiotherapy was created by the COVID-19 Pandemic Breast Cancer Consortium. This guidance is set to be published in Breast Cancer Research and Treatment and can be downloaded from the American College of Surgeons website.
A group from Memorial Sloan Kettering Cancer Center (MSKCC) created a guidance document on radiotherapy for breast cancer patients, and that guidance was recently published in Advances in Radiation Oncology.
Prioritizing certain patients and treatments
As hospital beds and clinics fill with coronavirus-infected patients, oncologists must balance the need for timely therapy for their patients with the imperative to protect vulnerable, immunosuppressed patients from exposure and keep clinical resources as free as possible.
“As we’re taking care of breast cancer patients during this unprecedented pandemic, what we’re all trying to do is balance the most effective treatments for our patients against the risk of additional exposures, either from other patients [or] from being outside, and considerations about the safety of our staff,” said Steven Isakoff, MD, PhD, of Massachusetts General Hospital Cancer Center in Boston, who is an author of the COVID-19 Pandemic Breast Cancer Consortium guidance.
The consortium’s guidance recommends prioritizing treatment according to patient needs and the disease type and stage. The three basic categories for considering when to treat are:
- Priority A: Patients who have immediately life-threatening conditions, are clinically unstable, or would experience a significant change in prognosis with even a short delay in treatment.
- Priority B: Deferring treatment for a short time (6-12 weeks) would not impact overall outcomes in these patients.
- Priority C: These patients are stable enough that treatment can be delayed for the duration of the COVID-19 pandemic.
“The consortium highly recommends multidisciplinary discussion regarding priority for elective surgery and adjuvant treatments for your breast cancer patients,” the guidance authors wrote. “The COVID-19 pandemic may vary in severity over time, and these recommendations are subject to change with changing COVID-19 pandemic severity.”
For example, depending on local circumstances, the guidance recommends limiting immediate outpatient visits to patients with potentially unstable conditions such as infection or hematoma. Established patients with new problems or patients with a new diagnosis of noninvasive cancer might be managed with telemedicine visits, and patients who are on follow-up with no new issues or who have benign lesions might have their visits safely postponed.
Surgery and drug recommendations
High-priority surgical procedures include operative drainage of a breast abscess in a septic patient and evacuation of expanding hematoma in a hemodynamically unstable patient, according to the consortium guidance.
Other surgical situations are more nuanced. For example, for patients with triple-negative breast cancer (TNBC) or HER2-positive disease, the guidance recommends neoadjuvant chemotherapy or HER2-targeted chemotherapy in some cases. In other cases, institutions may proceed with surgery before chemotherapy, but “these decisions will depend on institutional resources and patient factors,” according to the authors.
The guidance states that chemotherapy and other drug treatments should not be delayed in patients with oncologic emergencies, such as febrile neutropenia, hypercalcemia, intolerable pain, symptomatic pleural effusions, or brain metastases.
In addition, patients with inflammatory breast cancer, TNBC, or HER2-positive breast cancer should receive neoadjuvant/adjuvant chemotherapy. Patients with metastatic disease that is likely to benefit from therapy should start chemotherapy, endocrine therapy, or targeted therapy. And patients who have already started neoadjuvant/adjuvant chemotherapy or oral adjuvant endocrine therapy should continue on these treatments.
Radiation therapy recommendations
The consortium guidance recommends administering radiation to patients with bleeding or painful inoperable locoregional disease, those with symptomatic metastatic disease, and patients who progress on neoadjuvant chemotherapy.
In contrast, older patients (aged 65-70 years) with lower-risk, stage I, hormone receptor–positive, HER2-negative cancers who are on adjuvant endocrine therapy can safely defer or omit radiation without affecting their overall survival, according to the guidance. Patients with ductal carcinoma in situ, especially those with estrogen receptor–positive disease on endocrine therapy, can safely omit radiation.
“There are clearly conditions where radiation might reduce the risk of recurrence but not improve overall survival, where a delay in treatment really will have minimal or no impact,” Dr. Isakoff said.
The MSKCC guidance recommends omitting radiation for some patients with favorable-risk disease and truncating or accelerating regimens using hypofractionation for others who require whole-breast radiation or post-mastectomy treatment.
The MSKCC guidance also contains recommendations for prioritization of patients according to disease state and the urgency of care. It divides cases into high, intermediate, and low priority for breast radiotherapy, as follows:
- Tier 1 (high priority): Patients with inflammatory breast cancer, residual node-positive disease after neoadjuvant chemotherapy, four or more positive nodes (N2), recurrent disease, node-positive TNBC, or extensive lymphovascular invasion.
- Tier 2 (intermediate priority): Patients with estrogen receptor–positive disease with one to three positive nodes (N1a), pathologic stage N0 after neoadjuvant chemotherapy, lymphovascular invasion not otherwise specified, or node-negative TNBC.
- Tier 3 (low priority): Patients with early-stage estrogen receptor-positive breast cancer (especially patients of advanced age), patients with ductal carcinoma in situ, or those who otherwise do not meet the criteria for tiers 1 or 2.
The MSKCC guidance also contains recommended hypofractionated or accelerated radiotherapy regimens for partial and whole-breast irradiation, post-mastectomy treatment, and breast and regional node irradiation, including recommended techniques (for example, 3-D conformal or intensity modulated approaches).
The authors of the MSKCC guidance disclosed relationships with eContour, Volastra Therapeutics, Sanofi, the Prostate Cancer Foundation, and Cancer Research UK. The authors of the COVID-19 Pandemic Breast Cancer Consortium guidance did not disclose any conflicts and said there was no funding source for the guidance.
SOURCES: Braunstein LZ et al. Adv Radiat Oncol. 2020 Apr 1. doi:10.1016/j.adro.2020.03.013; Dietz JR et al. 2020 Apr. Recommendations for prioritization, treatment and triage of breast cancer patients during the COVID-19 pandemic. Accepted for publication in Breast Cancer Research and Treatment.
Nothing is business as usual during the COVID-19 pandemic, and that includes breast cancer therapy. That’s why two groups have released guidance documents on treating breast cancer patients during the pandemic.
A guidance on surgery, drug therapy, and radiotherapy was created by the COVID-19 Pandemic Breast Cancer Consortium. This guidance is set to be published in Breast Cancer Research and Treatment and can be downloaded from the American College of Surgeons website.
A group from Memorial Sloan Kettering Cancer Center (MSKCC) created a guidance document on radiotherapy for breast cancer patients, and that guidance was recently published in Advances in Radiation Oncology.
Prioritizing certain patients and treatments
As hospital beds and clinics fill with coronavirus-infected patients, oncologists must balance the need for timely therapy for their patients with the imperative to protect vulnerable, immunosuppressed patients from exposure and keep clinical resources as free as possible.
“As we’re taking care of breast cancer patients during this unprecedented pandemic, what we’re all trying to do is balance the most effective treatments for our patients against the risk of additional exposures, either from other patients [or] from being outside, and considerations about the safety of our staff,” said Steven Isakoff, MD, PhD, of Massachusetts General Hospital Cancer Center in Boston, who is an author of the COVID-19 Pandemic Breast Cancer Consortium guidance.
The consortium’s guidance recommends prioritizing treatment according to patient needs and the disease type and stage. The three basic categories for considering when to treat are:
- Priority A: Patients who have immediately life-threatening conditions, are clinically unstable, or would experience a significant change in prognosis with even a short delay in treatment.
- Priority B: Deferring treatment for a short time (6-12 weeks) would not impact overall outcomes in these patients.
- Priority C: These patients are stable enough that treatment can be delayed for the duration of the COVID-19 pandemic.
“The consortium highly recommends multidisciplinary discussion regarding priority for elective surgery and adjuvant treatments for your breast cancer patients,” the guidance authors wrote. “The COVID-19 pandemic may vary in severity over time, and these recommendations are subject to change with changing COVID-19 pandemic severity.”
For example, depending on local circumstances, the guidance recommends limiting immediate outpatient visits to patients with potentially unstable conditions such as infection or hematoma. Established patients with new problems or patients with a new diagnosis of noninvasive cancer might be managed with telemedicine visits, and patients who are on follow-up with no new issues or who have benign lesions might have their visits safely postponed.
Surgery and drug recommendations
High-priority surgical procedures include operative drainage of a breast abscess in a septic patient and evacuation of expanding hematoma in a hemodynamically unstable patient, according to the consortium guidance.
Other surgical situations are more nuanced. For example, for patients with triple-negative breast cancer (TNBC) or HER2-positive disease, the guidance recommends neoadjuvant chemotherapy or HER2-targeted chemotherapy in some cases. In other cases, institutions may proceed with surgery before chemotherapy, but “these decisions will depend on institutional resources and patient factors,” according to the authors.
The guidance states that chemotherapy and other drug treatments should not be delayed in patients with oncologic emergencies, such as febrile neutropenia, hypercalcemia, intolerable pain, symptomatic pleural effusions, or brain metastases.
In addition, patients with inflammatory breast cancer, TNBC, or HER2-positive breast cancer should receive neoadjuvant/adjuvant chemotherapy. Patients with metastatic disease that is likely to benefit from therapy should start chemotherapy, endocrine therapy, or targeted therapy. And patients who have already started neoadjuvant/adjuvant chemotherapy or oral adjuvant endocrine therapy should continue on these treatments.
Radiation therapy recommendations
The consortium guidance recommends administering radiation to patients with bleeding or painful inoperable locoregional disease, those with symptomatic metastatic disease, and patients who progress on neoadjuvant chemotherapy.
In contrast, older patients (aged 65-70 years) with lower-risk, stage I, hormone receptor–positive, HER2-negative cancers who are on adjuvant endocrine therapy can safely defer or omit radiation without affecting their overall survival, according to the guidance. Patients with ductal carcinoma in situ, especially those with estrogen receptor–positive disease on endocrine therapy, can safely omit radiation.
“There are clearly conditions where radiation might reduce the risk of recurrence but not improve overall survival, where a delay in treatment really will have minimal or no impact,” Dr. Isakoff said.
The MSKCC guidance recommends omitting radiation for some patients with favorable-risk disease and truncating or accelerating regimens using hypofractionation for others who require whole-breast radiation or post-mastectomy treatment.
The MSKCC guidance also contains recommendations for prioritization of patients according to disease state and the urgency of care. It divides cases into high, intermediate, and low priority for breast radiotherapy, as follows:
- Tier 1 (high priority): Patients with inflammatory breast cancer, residual node-positive disease after neoadjuvant chemotherapy, four or more positive nodes (N2), recurrent disease, node-positive TNBC, or extensive lymphovascular invasion.
- Tier 2 (intermediate priority): Patients with estrogen receptor–positive disease with one to three positive nodes (N1a), pathologic stage N0 after neoadjuvant chemotherapy, lymphovascular invasion not otherwise specified, or node-negative TNBC.
- Tier 3 (low priority): Patients with early-stage estrogen receptor-positive breast cancer (especially patients of advanced age), patients with ductal carcinoma in situ, or those who otherwise do not meet the criteria for tiers 1 or 2.
The MSKCC guidance also contains recommended hypofractionated or accelerated radiotherapy regimens for partial and whole-breast irradiation, post-mastectomy treatment, and breast and regional node irradiation, including recommended techniques (for example, 3-D conformal or intensity modulated approaches).
The authors of the MSKCC guidance disclosed relationships with eContour, Volastra Therapeutics, Sanofi, the Prostate Cancer Foundation, and Cancer Research UK. The authors of the COVID-19 Pandemic Breast Cancer Consortium guidance did not disclose any conflicts and said there was no funding source for the guidance.
SOURCES: Braunstein LZ et al. Adv Radiat Oncol. 2020 Apr 1. doi:10.1016/j.adro.2020.03.013; Dietz JR et al. 2020 Apr. Recommendations for prioritization, treatment and triage of breast cancer patients during the COVID-19 pandemic. Accepted for publication in Breast Cancer Research and Treatment.
Nothing is business as usual during the COVID-19 pandemic, and that includes breast cancer therapy. That’s why two groups have released guidance documents on treating breast cancer patients during the pandemic.
A guidance on surgery, drug therapy, and radiotherapy was created by the COVID-19 Pandemic Breast Cancer Consortium. This guidance is set to be published in Breast Cancer Research and Treatment and can be downloaded from the American College of Surgeons website.
A group from Memorial Sloan Kettering Cancer Center (MSKCC) created a guidance document on radiotherapy for breast cancer patients, and that guidance was recently published in Advances in Radiation Oncology.
Prioritizing certain patients and treatments
As hospital beds and clinics fill with coronavirus-infected patients, oncologists must balance the need for timely therapy for their patients with the imperative to protect vulnerable, immunosuppressed patients from exposure and keep clinical resources as free as possible.
“As we’re taking care of breast cancer patients during this unprecedented pandemic, what we’re all trying to do is balance the most effective treatments for our patients against the risk of additional exposures, either from other patients [or] from being outside, and considerations about the safety of our staff,” said Steven Isakoff, MD, PhD, of Massachusetts General Hospital Cancer Center in Boston, who is an author of the COVID-19 Pandemic Breast Cancer Consortium guidance.
The consortium’s guidance recommends prioritizing treatment according to patient needs and the disease type and stage. The three basic categories for considering when to treat are:
- Priority A: Patients who have immediately life-threatening conditions, are clinically unstable, or would experience a significant change in prognosis with even a short delay in treatment.
- Priority B: Deferring treatment for a short time (6-12 weeks) would not impact overall outcomes in these patients.
- Priority C: These patients are stable enough that treatment can be delayed for the duration of the COVID-19 pandemic.
“The consortium highly recommends multidisciplinary discussion regarding priority for elective surgery and adjuvant treatments for your breast cancer patients,” the guidance authors wrote. “The COVID-19 pandemic may vary in severity over time, and these recommendations are subject to change with changing COVID-19 pandemic severity.”
For example, depending on local circumstances, the guidance recommends limiting immediate outpatient visits to patients with potentially unstable conditions such as infection or hematoma. Established patients with new problems or patients with a new diagnosis of noninvasive cancer might be managed with telemedicine visits, and patients who are on follow-up with no new issues or who have benign lesions might have their visits safely postponed.
Surgery and drug recommendations
High-priority surgical procedures include operative drainage of a breast abscess in a septic patient and evacuation of expanding hematoma in a hemodynamically unstable patient, according to the consortium guidance.
Other surgical situations are more nuanced. For example, for patients with triple-negative breast cancer (TNBC) or HER2-positive disease, the guidance recommends neoadjuvant chemotherapy or HER2-targeted chemotherapy in some cases. In other cases, institutions may proceed with surgery before chemotherapy, but “these decisions will depend on institutional resources and patient factors,” according to the authors.
The guidance states that chemotherapy and other drug treatments should not be delayed in patients with oncologic emergencies, such as febrile neutropenia, hypercalcemia, intolerable pain, symptomatic pleural effusions, or brain metastases.
In addition, patients with inflammatory breast cancer, TNBC, or HER2-positive breast cancer should receive neoadjuvant/adjuvant chemotherapy. Patients with metastatic disease that is likely to benefit from therapy should start chemotherapy, endocrine therapy, or targeted therapy. And patients who have already started neoadjuvant/adjuvant chemotherapy or oral adjuvant endocrine therapy should continue on these treatments.
Radiation therapy recommendations
The consortium guidance recommends administering radiation to patients with bleeding or painful inoperable locoregional disease, those with symptomatic metastatic disease, and patients who progress on neoadjuvant chemotherapy.
In contrast, older patients (aged 65-70 years) with lower-risk, stage I, hormone receptor–positive, HER2-negative cancers who are on adjuvant endocrine therapy can safely defer or omit radiation without affecting their overall survival, according to the guidance. Patients with ductal carcinoma in situ, especially those with estrogen receptor–positive disease on endocrine therapy, can safely omit radiation.
“There are clearly conditions where radiation might reduce the risk of recurrence but not improve overall survival, where a delay in treatment really will have minimal or no impact,” Dr. Isakoff said.
The MSKCC guidance recommends omitting radiation for some patients with favorable-risk disease and truncating or accelerating regimens using hypofractionation for others who require whole-breast radiation or post-mastectomy treatment.
The MSKCC guidance also contains recommendations for prioritization of patients according to disease state and the urgency of care. It divides cases into high, intermediate, and low priority for breast radiotherapy, as follows:
- Tier 1 (high priority): Patients with inflammatory breast cancer, residual node-positive disease after neoadjuvant chemotherapy, four or more positive nodes (N2), recurrent disease, node-positive TNBC, or extensive lymphovascular invasion.
- Tier 2 (intermediate priority): Patients with estrogen receptor–positive disease with one to three positive nodes (N1a), pathologic stage N0 after neoadjuvant chemotherapy, lymphovascular invasion not otherwise specified, or node-negative TNBC.
- Tier 3 (low priority): Patients with early-stage estrogen receptor-positive breast cancer (especially patients of advanced age), patients with ductal carcinoma in situ, or those who otherwise do not meet the criteria for tiers 1 or 2.
The MSKCC guidance also contains recommended hypofractionated or accelerated radiotherapy regimens for partial and whole-breast irradiation, post-mastectomy treatment, and breast and regional node irradiation, including recommended techniques (for example, 3-D conformal or intensity modulated approaches).
The authors of the MSKCC guidance disclosed relationships with eContour, Volastra Therapeutics, Sanofi, the Prostate Cancer Foundation, and Cancer Research UK. The authors of the COVID-19 Pandemic Breast Cancer Consortium guidance did not disclose any conflicts and said there was no funding source for the guidance.
SOURCES: Braunstein LZ et al. Adv Radiat Oncol. 2020 Apr 1. doi:10.1016/j.adro.2020.03.013; Dietz JR et al. 2020 Apr. Recommendations for prioritization, treatment and triage of breast cancer patients during the COVID-19 pandemic. Accepted for publication in Breast Cancer Research and Treatment.
Home-based chemo skyrockets at one U.S. center
Major organization opposes concept
In the fall of 2019, the University of Pennsylvania in Philadelphia started a pilot program of home-based chemotherapy for two treatment regimens (one via infusion and one via injection). Six months later, the Cancer Care at Home program had treated 40 patients.
The uptake within the university’s large regional health system was acceptable but not rapid, admitted Amy Laughlin, MD, a hematology-oncology fellow involved with the program.
Then COVID-19 arrived, along with related travel restrictions.
Suddenly, in a 5-week period (March to April 7), 175 patients had been treated – a 300% increase from the first half year. Program staff jumped from 12 to 80 employees. The list of chemotherapies delivered went from two to seven, with more coming.
“We’re not the pilot anymore – we’re the standard of care,” Laughlin told Medscape Medical News.
“The impact [on patients] is amazing,” she said. “As long as you are selecting the right patients and right therapy, it is feasible and even preferable for a lot of patients.”
For example, patients with hormone-positive breast cancer who receive leuprolide (to shut down the ovaries and suppress estrogen production) ordinarily would have to visit a Penn facility for an injection every month, potentially for years. Now, a nurse can meet patients at home (or before the COVID-19 pandemic, even at their place of work) and administer the injection, saving the patient travel time and associated costs.
This home-based chemotherapy service does not appear to be offered elsewhere in the United States, and a major oncology organization – the Community Oncology Alliance – is opposed to the practice because of patient safety concerns.
The service is not offered at a sample of cancer centers queried by Medscape Medical News, including the Dana-Farber Cancer Institute in Boston, the Moffitt Cancer Center in Tampa, the Huntsman Cancer Institute in Salt Lake City, Utah, and Moores Cancer Center, the University of California, San Diego.
Opposition because of safety concerns
On April 9, the Community Oncology Alliance (COA) issued a statement saying it “fundamentally opposes home infusion of chemotherapy, cancer immunotherapy, and cancer treatment supportive drugs because of serious patient safety concerns.”
The COA warned that “many of the side effects caused by cancer treatment can have a rapid, unpredictable onset that places patients in incredible jeopardy and can even be life-threatening.”
In contrast, in a recent communication related to COVID-19, the National Comprehensive Cancer Network tacitly endorsed the concept, stating that a number of chemotherapies may potentially be administered at home, but it did not include guidelines for doing so.
The American Society of Clinical Oncology said that chemotherapy at home is “an issue [we] are monitoring closely,” according to a spokesperson.
What’s involved
Criteria for home-based chemotherapy at Penn include use of anticancer therapies that a patient has previously tolerated and low toxicity (that can be readily managed in the home setting). In addition, patients must be capable of following a med chart.
The chemotherapy is reconstituted at a Penn facility in a Philadelphia suburb. A courier then delivers the drug to the patient’s home, where it is administered by an oncology-trained nurse. Drugs must be stable for at least a few hours to qualify for the program.
The Penn program started with two regimens: EPOCH (etoposide, vincristine, doxorubicin, cyclophosphamide, and prednisone) for lymphoma, and leuprolide acetate injections for either breast or prostate cancer.
The two treatments are polar opposites in terms of complexity, common usage, and time required, which was intended, said Laughlin.
Time to deliver the chemo varies from a matter of minutes with leuprolide to more than 2 hours for rituximab, a lymphoma drug that may be added to EPOCH.
The current list of at-home chemo agents in the Penn program also includes bortezomib, lanreotide, zoledronic acid, and denosumab. Soon to come are rituximab and pembrolizumab for lung cancer and head and neck cancer.
Already practiced in some European countries
Home-based chemotherapy dates from at least the 1980s in the medical literature and is practiced in some European countries.
A 2018 randomized study of adjuvant treatment with capecitabine and oxaliplatin for stage II/III colon cancer in Denmark, where home-based care has been practiced for the past 2 years and is growing in use, concluded that “it might be a valuable alternative to treatment at an outpatient clinic.”
However, in the study, there was no difference in quality of life between the home and outpatient settings, which is somewhat surprising, inasmuch as a major appeal to receiving chemotherapy at home is that it is less disruptive compared to receiving it in a hospital or clinic, which requires travel.
Also, chemo at home “may be resource intensive” and have a “lower throughput of patients due to transportation time,” cautioned the Danish investigators, who were from Herlev and Gentofte Hospital.
A 2015 review called home chemo “a safe and patient‐centered alternative to hospital‐ and outpatient‐based service.” Jenna Evans, PhD, McMaster University, Toronto, Canada, and lead author of that review, says there are two major barriers to infusion chemotherapy in homes.
One is inadequate resources in the community, such as oncology-trained nurses to deliver treatment, and the other is perceptions of safety and quality, including among healthcare providers.
COVID-19 might prompt more chemo at home, said Evans, a health policy expert, in an email to Medscape Medical News. “It is not unusual for change of this type and scale to require a seismic event to become more mainstream,” she argued.
Reimbursement for home-based chemo is usually the same as for chemo in a free-standing infusion suite, says Cassandra Redmond, PharmD, MBA, director of pharmacy, Penn Home Infusion Therapy.
Private insurers and Medicare cover a subset of infused medications at home, but coverage is limited. “The opportunity now is to expand these initiatives ... to include other cancer therapies,” she said about coverage.
This article first appeared on Medscape.com.
Major organization opposes concept
Major organization opposes concept
In the fall of 2019, the University of Pennsylvania in Philadelphia started a pilot program of home-based chemotherapy for two treatment regimens (one via infusion and one via injection). Six months later, the Cancer Care at Home program had treated 40 patients.
The uptake within the university’s large regional health system was acceptable but not rapid, admitted Amy Laughlin, MD, a hematology-oncology fellow involved with the program.
Then COVID-19 arrived, along with related travel restrictions.
Suddenly, in a 5-week period (March to April 7), 175 patients had been treated – a 300% increase from the first half year. Program staff jumped from 12 to 80 employees. The list of chemotherapies delivered went from two to seven, with more coming.
“We’re not the pilot anymore – we’re the standard of care,” Laughlin told Medscape Medical News.
“The impact [on patients] is amazing,” she said. “As long as you are selecting the right patients and right therapy, it is feasible and even preferable for a lot of patients.”
For example, patients with hormone-positive breast cancer who receive leuprolide (to shut down the ovaries and suppress estrogen production) ordinarily would have to visit a Penn facility for an injection every month, potentially for years. Now, a nurse can meet patients at home (or before the COVID-19 pandemic, even at their place of work) and administer the injection, saving the patient travel time and associated costs.
This home-based chemotherapy service does not appear to be offered elsewhere in the United States, and a major oncology organization – the Community Oncology Alliance – is opposed to the practice because of patient safety concerns.
The service is not offered at a sample of cancer centers queried by Medscape Medical News, including the Dana-Farber Cancer Institute in Boston, the Moffitt Cancer Center in Tampa, the Huntsman Cancer Institute in Salt Lake City, Utah, and Moores Cancer Center, the University of California, San Diego.
Opposition because of safety concerns
On April 9, the Community Oncology Alliance (COA) issued a statement saying it “fundamentally opposes home infusion of chemotherapy, cancer immunotherapy, and cancer treatment supportive drugs because of serious patient safety concerns.”
The COA warned that “many of the side effects caused by cancer treatment can have a rapid, unpredictable onset that places patients in incredible jeopardy and can even be life-threatening.”
In contrast, in a recent communication related to COVID-19, the National Comprehensive Cancer Network tacitly endorsed the concept, stating that a number of chemotherapies may potentially be administered at home, but it did not include guidelines for doing so.
The American Society of Clinical Oncology said that chemotherapy at home is “an issue [we] are monitoring closely,” according to a spokesperson.
What’s involved
Criteria for home-based chemotherapy at Penn include use of anticancer therapies that a patient has previously tolerated and low toxicity (that can be readily managed in the home setting). In addition, patients must be capable of following a med chart.
The chemotherapy is reconstituted at a Penn facility in a Philadelphia suburb. A courier then delivers the drug to the patient’s home, where it is administered by an oncology-trained nurse. Drugs must be stable for at least a few hours to qualify for the program.
The Penn program started with two regimens: EPOCH (etoposide, vincristine, doxorubicin, cyclophosphamide, and prednisone) for lymphoma, and leuprolide acetate injections for either breast or prostate cancer.
The two treatments are polar opposites in terms of complexity, common usage, and time required, which was intended, said Laughlin.
Time to deliver the chemo varies from a matter of minutes with leuprolide to more than 2 hours for rituximab, a lymphoma drug that may be added to EPOCH.
The current list of at-home chemo agents in the Penn program also includes bortezomib, lanreotide, zoledronic acid, and denosumab. Soon to come are rituximab and pembrolizumab for lung cancer and head and neck cancer.
Already practiced in some European countries
Home-based chemotherapy dates from at least the 1980s in the medical literature and is practiced in some European countries.
A 2018 randomized study of adjuvant treatment with capecitabine and oxaliplatin for stage II/III colon cancer in Denmark, where home-based care has been practiced for the past 2 years and is growing in use, concluded that “it might be a valuable alternative to treatment at an outpatient clinic.”
However, in the study, there was no difference in quality of life between the home and outpatient settings, which is somewhat surprising, inasmuch as a major appeal to receiving chemotherapy at home is that it is less disruptive compared to receiving it in a hospital or clinic, which requires travel.
Also, chemo at home “may be resource intensive” and have a “lower throughput of patients due to transportation time,” cautioned the Danish investigators, who were from Herlev and Gentofte Hospital.
A 2015 review called home chemo “a safe and patient‐centered alternative to hospital‐ and outpatient‐based service.” Jenna Evans, PhD, McMaster University, Toronto, Canada, and lead author of that review, says there are two major barriers to infusion chemotherapy in homes.
One is inadequate resources in the community, such as oncology-trained nurses to deliver treatment, and the other is perceptions of safety and quality, including among healthcare providers.
COVID-19 might prompt more chemo at home, said Evans, a health policy expert, in an email to Medscape Medical News. “It is not unusual for change of this type and scale to require a seismic event to become more mainstream,” she argued.
Reimbursement for home-based chemo is usually the same as for chemo in a free-standing infusion suite, says Cassandra Redmond, PharmD, MBA, director of pharmacy, Penn Home Infusion Therapy.
Private insurers and Medicare cover a subset of infused medications at home, but coverage is limited. “The opportunity now is to expand these initiatives ... to include other cancer therapies,” she said about coverage.
This article first appeared on Medscape.com.
In the fall of 2019, the University of Pennsylvania in Philadelphia started a pilot program of home-based chemotherapy for two treatment regimens (one via infusion and one via injection). Six months later, the Cancer Care at Home program had treated 40 patients.
The uptake within the university’s large regional health system was acceptable but not rapid, admitted Amy Laughlin, MD, a hematology-oncology fellow involved with the program.
Then COVID-19 arrived, along with related travel restrictions.
Suddenly, in a 5-week period (March to April 7), 175 patients had been treated – a 300% increase from the first half year. Program staff jumped from 12 to 80 employees. The list of chemotherapies delivered went from two to seven, with more coming.
“We’re not the pilot anymore – we’re the standard of care,” Laughlin told Medscape Medical News.
“The impact [on patients] is amazing,” she said. “As long as you are selecting the right patients and right therapy, it is feasible and even preferable for a lot of patients.”
For example, patients with hormone-positive breast cancer who receive leuprolide (to shut down the ovaries and suppress estrogen production) ordinarily would have to visit a Penn facility for an injection every month, potentially for years. Now, a nurse can meet patients at home (or before the COVID-19 pandemic, even at their place of work) and administer the injection, saving the patient travel time and associated costs.
This home-based chemotherapy service does not appear to be offered elsewhere in the United States, and a major oncology organization – the Community Oncology Alliance – is opposed to the practice because of patient safety concerns.
The service is not offered at a sample of cancer centers queried by Medscape Medical News, including the Dana-Farber Cancer Institute in Boston, the Moffitt Cancer Center in Tampa, the Huntsman Cancer Institute in Salt Lake City, Utah, and Moores Cancer Center, the University of California, San Diego.
Opposition because of safety concerns
On April 9, the Community Oncology Alliance (COA) issued a statement saying it “fundamentally opposes home infusion of chemotherapy, cancer immunotherapy, and cancer treatment supportive drugs because of serious patient safety concerns.”
The COA warned that “many of the side effects caused by cancer treatment can have a rapid, unpredictable onset that places patients in incredible jeopardy and can even be life-threatening.”
In contrast, in a recent communication related to COVID-19, the National Comprehensive Cancer Network tacitly endorsed the concept, stating that a number of chemotherapies may potentially be administered at home, but it did not include guidelines for doing so.
The American Society of Clinical Oncology said that chemotherapy at home is “an issue [we] are monitoring closely,” according to a spokesperson.
What’s involved
Criteria for home-based chemotherapy at Penn include use of anticancer therapies that a patient has previously tolerated and low toxicity (that can be readily managed in the home setting). In addition, patients must be capable of following a med chart.
The chemotherapy is reconstituted at a Penn facility in a Philadelphia suburb. A courier then delivers the drug to the patient’s home, where it is administered by an oncology-trained nurse. Drugs must be stable for at least a few hours to qualify for the program.
The Penn program started with two regimens: EPOCH (etoposide, vincristine, doxorubicin, cyclophosphamide, and prednisone) for lymphoma, and leuprolide acetate injections for either breast or prostate cancer.
The two treatments are polar opposites in terms of complexity, common usage, and time required, which was intended, said Laughlin.
Time to deliver the chemo varies from a matter of minutes with leuprolide to more than 2 hours for rituximab, a lymphoma drug that may be added to EPOCH.
The current list of at-home chemo agents in the Penn program also includes bortezomib, lanreotide, zoledronic acid, and denosumab. Soon to come are rituximab and pembrolizumab for lung cancer and head and neck cancer.
Already practiced in some European countries
Home-based chemotherapy dates from at least the 1980s in the medical literature and is practiced in some European countries.
A 2018 randomized study of adjuvant treatment with capecitabine and oxaliplatin for stage II/III colon cancer in Denmark, where home-based care has been practiced for the past 2 years and is growing in use, concluded that “it might be a valuable alternative to treatment at an outpatient clinic.”
However, in the study, there was no difference in quality of life between the home and outpatient settings, which is somewhat surprising, inasmuch as a major appeal to receiving chemotherapy at home is that it is less disruptive compared to receiving it in a hospital or clinic, which requires travel.
Also, chemo at home “may be resource intensive” and have a “lower throughput of patients due to transportation time,” cautioned the Danish investigators, who were from Herlev and Gentofte Hospital.
A 2015 review called home chemo “a safe and patient‐centered alternative to hospital‐ and outpatient‐based service.” Jenna Evans, PhD, McMaster University, Toronto, Canada, and lead author of that review, says there are two major barriers to infusion chemotherapy in homes.
One is inadequate resources in the community, such as oncology-trained nurses to deliver treatment, and the other is perceptions of safety and quality, including among healthcare providers.
COVID-19 might prompt more chemo at home, said Evans, a health policy expert, in an email to Medscape Medical News. “It is not unusual for change of this type and scale to require a seismic event to become more mainstream,” she argued.
Reimbursement for home-based chemo is usually the same as for chemo in a free-standing infusion suite, says Cassandra Redmond, PharmD, MBA, director of pharmacy, Penn Home Infusion Therapy.
Private insurers and Medicare cover a subset of infused medications at home, but coverage is limited. “The opportunity now is to expand these initiatives ... to include other cancer therapies,” she said about coverage.
This article first appeared on Medscape.com.
Managing pediatric heme/onc departments during the pandemic
Given the possibility that children with hematologic malignancies may have increased susceptibility to coronavirus disease 2019 (COVID-19), clinicians from China and the United States have proposed a plan for preventing and managing outbreaks in hospitals’ pediatric hematology and oncology departments.
The plan is focused primarily on infection prevention and control strategies, Yulei He, MD, of Chengdu (China) Women’s and Children’s Central Hospital and colleagues explained in an article published in The Lancet Haematology.
The authors noted that close contact with COVID-19 patients is thought to be the main route of transmission, and a retrospective study indicated that 41.3% of initial COVID-19 cases were caused by hospital-related transmission.
“Children with hematological malignancies might have increased susceptibility to infection with SARS-CoV-2 because of immunodeficiency; therefore, procedures are needed to avoid hospital-related transmission and infection for these patients,” the authors wrote.
Preventing the spread of infection
Dr. He and colleagues advised that medical staff be kept up-to-date with the latest information about COVID-19 and perform assessments regularly to identify cases in their departments.
The authors also recommended establishing a COVID-19 expert committee – consisting of infectious disease physicians, hematologists, oncologists, radiologists, pharmacists, and hospital infection control staff – to make medical decisions in multidisciplinary consultation meetings. In addition, the authors recommended regional management strategies be adopted to minimize cross infection within the hospital. Specifically, the authors proposed creating the following four zones:
1. A surveillance and screening zone for patients potentially infected with SARS-CoV-2
2. A suspected-case quarantine zone where patients thought to have COVID-19 are isolated in single rooms
3. A confirmed-case quarantine zone where patients are treated for COVID-19
4. A hematology/oncology ward for treating non–COVID-19 patients with malignancies.
Dr. He and colleagues also stressed the importance of providing personal protective equipment for all zones, along with instructions for proper use and disposal. The authors recommended developing and following specific protocols for outpatient visits in the hematology/oncology ward, and providing COVID-19 prevention and control information to families and health care workers.
Managing cancer treatment
For patients with acute leukemias who have induction chemotherapy planned, Dr. He and colleagues argued that scheduled chemotherapy should not be interrupted unless COVID-19 is suspected or diagnosed. The authors said treatment should not be delayed more than 7 days during induction, consolidation, or the intermediate phase of chemotherapy because the virus has an incubation period of 2-7 days. This will allow a short period of observation to screen for potential infection.
The authors recommended that patients with lymphoma and solid tumors first undergo COVID-19 screening and then receive treatment in hematology/oncology wards “according to their chemotherapy schedule, and without delay, until they are in complete remission.”
“If the patient is in complete remission, we recommend a treatment delay of no more than 7 days to allow a short period of observation to screen for COVID-19,” the authors added.
Maintenance chemotherapy should not be delayed for more than 14 days, Dr. He and colleagues wrote. “This increase in the maximum delay before chemotherapy strikes a balance between the potential risk of SARS-CoV-2 infection and tumor recurrence, since pediatric patients in this phase of treatment have a reduced risk of tumor recurrence,” the authors added.
Caring for patients with COVID-19
For inpatients diagnosed with COVID-19, Dr. He and colleagues recommended the following:
- Prioritize COVID-19 treatment for children with primary disease remission.
- For children not in remission, prioritize treatment for critical patients.
- Isolated patients should be treated for COVID-19, and their chemotherapy should be temporarily suspended or reduced in intensity..
Dr. He and colleagues noted that, by following these recommendations for infection prevention, they had no cases of COVID-19 among children in their hematology/oncology departments. However, the authors said the recommendations “could fail to some extent” based on “differences in medical resources, health care settings, and the policy of the specific government.”
The authors said their recommendations should be updated continuously as new information and clinical evidence emerges.
Dr. He and colleagues reported having no conflicts of interest.
SOURCE: He Y et al. Lancet Haematol. doi: 10/1016/s2352-3026(20)30104-6.
Given the possibility that children with hematologic malignancies may have increased susceptibility to coronavirus disease 2019 (COVID-19), clinicians from China and the United States have proposed a plan for preventing and managing outbreaks in hospitals’ pediatric hematology and oncology departments.
The plan is focused primarily on infection prevention and control strategies, Yulei He, MD, of Chengdu (China) Women’s and Children’s Central Hospital and colleagues explained in an article published in The Lancet Haematology.
The authors noted that close contact with COVID-19 patients is thought to be the main route of transmission, and a retrospective study indicated that 41.3% of initial COVID-19 cases were caused by hospital-related transmission.
“Children with hematological malignancies might have increased susceptibility to infection with SARS-CoV-2 because of immunodeficiency; therefore, procedures are needed to avoid hospital-related transmission and infection for these patients,” the authors wrote.
Preventing the spread of infection
Dr. He and colleagues advised that medical staff be kept up-to-date with the latest information about COVID-19 and perform assessments regularly to identify cases in their departments.
The authors also recommended establishing a COVID-19 expert committee – consisting of infectious disease physicians, hematologists, oncologists, radiologists, pharmacists, and hospital infection control staff – to make medical decisions in multidisciplinary consultation meetings. In addition, the authors recommended regional management strategies be adopted to minimize cross infection within the hospital. Specifically, the authors proposed creating the following four zones:
1. A surveillance and screening zone for patients potentially infected with SARS-CoV-2
2. A suspected-case quarantine zone where patients thought to have COVID-19 are isolated in single rooms
3. A confirmed-case quarantine zone where patients are treated for COVID-19
4. A hematology/oncology ward for treating non–COVID-19 patients with malignancies.
Dr. He and colleagues also stressed the importance of providing personal protective equipment for all zones, along with instructions for proper use and disposal. The authors recommended developing and following specific protocols for outpatient visits in the hematology/oncology ward, and providing COVID-19 prevention and control information to families and health care workers.
Managing cancer treatment
For patients with acute leukemias who have induction chemotherapy planned, Dr. He and colleagues argued that scheduled chemotherapy should not be interrupted unless COVID-19 is suspected or diagnosed. The authors said treatment should not be delayed more than 7 days during induction, consolidation, or the intermediate phase of chemotherapy because the virus has an incubation period of 2-7 days. This will allow a short period of observation to screen for potential infection.
The authors recommended that patients with lymphoma and solid tumors first undergo COVID-19 screening and then receive treatment in hematology/oncology wards “according to their chemotherapy schedule, and without delay, until they are in complete remission.”
“If the patient is in complete remission, we recommend a treatment delay of no more than 7 days to allow a short period of observation to screen for COVID-19,” the authors added.
Maintenance chemotherapy should not be delayed for more than 14 days, Dr. He and colleagues wrote. “This increase in the maximum delay before chemotherapy strikes a balance between the potential risk of SARS-CoV-2 infection and tumor recurrence, since pediatric patients in this phase of treatment have a reduced risk of tumor recurrence,” the authors added.
Caring for patients with COVID-19
For inpatients diagnosed with COVID-19, Dr. He and colleagues recommended the following:
- Prioritize COVID-19 treatment for children with primary disease remission.
- For children not in remission, prioritize treatment for critical patients.
- Isolated patients should be treated for COVID-19, and their chemotherapy should be temporarily suspended or reduced in intensity..
Dr. He and colleagues noted that, by following these recommendations for infection prevention, they had no cases of COVID-19 among children in their hematology/oncology departments. However, the authors said the recommendations “could fail to some extent” based on “differences in medical resources, health care settings, and the policy of the specific government.”
The authors said their recommendations should be updated continuously as new information and clinical evidence emerges.
Dr. He and colleagues reported having no conflicts of interest.
SOURCE: He Y et al. Lancet Haematol. doi: 10/1016/s2352-3026(20)30104-6.
Given the possibility that children with hematologic malignancies may have increased susceptibility to coronavirus disease 2019 (COVID-19), clinicians from China and the United States have proposed a plan for preventing and managing outbreaks in hospitals’ pediatric hematology and oncology departments.
The plan is focused primarily on infection prevention and control strategies, Yulei He, MD, of Chengdu (China) Women’s and Children’s Central Hospital and colleagues explained in an article published in The Lancet Haematology.
The authors noted that close contact with COVID-19 patients is thought to be the main route of transmission, and a retrospective study indicated that 41.3% of initial COVID-19 cases were caused by hospital-related transmission.
“Children with hematological malignancies might have increased susceptibility to infection with SARS-CoV-2 because of immunodeficiency; therefore, procedures are needed to avoid hospital-related transmission and infection for these patients,” the authors wrote.
Preventing the spread of infection
Dr. He and colleagues advised that medical staff be kept up-to-date with the latest information about COVID-19 and perform assessments regularly to identify cases in their departments.
The authors also recommended establishing a COVID-19 expert committee – consisting of infectious disease physicians, hematologists, oncologists, radiologists, pharmacists, and hospital infection control staff – to make medical decisions in multidisciplinary consultation meetings. In addition, the authors recommended regional management strategies be adopted to minimize cross infection within the hospital. Specifically, the authors proposed creating the following four zones:
1. A surveillance and screening zone for patients potentially infected with SARS-CoV-2
2. A suspected-case quarantine zone where patients thought to have COVID-19 are isolated in single rooms
3. A confirmed-case quarantine zone where patients are treated for COVID-19
4. A hematology/oncology ward for treating non–COVID-19 patients with malignancies.
Dr. He and colleagues also stressed the importance of providing personal protective equipment for all zones, along with instructions for proper use and disposal. The authors recommended developing and following specific protocols for outpatient visits in the hematology/oncology ward, and providing COVID-19 prevention and control information to families and health care workers.
Managing cancer treatment
For patients with acute leukemias who have induction chemotherapy planned, Dr. He and colleagues argued that scheduled chemotherapy should not be interrupted unless COVID-19 is suspected or diagnosed. The authors said treatment should not be delayed more than 7 days during induction, consolidation, or the intermediate phase of chemotherapy because the virus has an incubation period of 2-7 days. This will allow a short period of observation to screen for potential infection.
The authors recommended that patients with lymphoma and solid tumors first undergo COVID-19 screening and then receive treatment in hematology/oncology wards “according to their chemotherapy schedule, and without delay, until they are in complete remission.”
“If the patient is in complete remission, we recommend a treatment delay of no more than 7 days to allow a short period of observation to screen for COVID-19,” the authors added.
Maintenance chemotherapy should not be delayed for more than 14 days, Dr. He and colleagues wrote. “This increase in the maximum delay before chemotherapy strikes a balance between the potential risk of SARS-CoV-2 infection and tumor recurrence, since pediatric patients in this phase of treatment have a reduced risk of tumor recurrence,” the authors added.
Caring for patients with COVID-19
For inpatients diagnosed with COVID-19, Dr. He and colleagues recommended the following:
- Prioritize COVID-19 treatment for children with primary disease remission.
- For children not in remission, prioritize treatment for critical patients.
- Isolated patients should be treated for COVID-19, and their chemotherapy should be temporarily suspended or reduced in intensity..
Dr. He and colleagues noted that, by following these recommendations for infection prevention, they had no cases of COVID-19 among children in their hematology/oncology departments. However, the authors said the recommendations “could fail to some extent” based on “differences in medical resources, health care settings, and the policy of the specific government.”
The authors said their recommendations should be updated continuously as new information and clinical evidence emerges.
Dr. He and colleagues reported having no conflicts of interest.
SOURCE: He Y et al. Lancet Haematol. doi: 10/1016/s2352-3026(20)30104-6.
FROM THE LANCET HAEMATOLOGY
Colorectal cancer: Proposed treatment guidelines for the COVID-19 era
In light of the rapid changes affecting cancer clinics due to the COVID-19 pandemic, Dr. David Kerr and Dr. Rachel Kerr, both specialists in gastrointestinal cancers at the University of Oxford in Oxford, United Kingdom, drafted these guidelines for the use of chemotherapy in colorectal cancer patients. Dr. Kerr and Dr. Kerr are putting forth this guidance as a topic for discussion and debate.
Our aim in developing these recommendations for the care of colorectal cancer patients in areas affected by the COVID-19 outbreak is to reduce the comorbidity of chemotherapy and decrease the risk of patients dying from COVID-19, weighed against the potential benefits of receiving chemotherapy. These recommendations are also designed to reduce the burden on chemotherapy units during a time of great pressure.
We have modified the guidelines in such a way that, we believe, will decrease the total number of patients receiving chemotherapy – particularly in the adjuvant setting – and reduce the overall immune impact of chemotherapy on these patients. Specifically, we suggest changing doublet chemotherapy to single-agent chemotherapy for some groups; changing to combinations involving capecitabine rather than bolus and infusional 5-FU for other patients; and, finally, making reasonable dose reductions upfront to reduce the risk for cycle 1 complications.
By changing from push-and-pump 5-FU to capecitabine for the vast majority of patients, we will both reduce the rates of neutropenia and decrease throughput in chemotherapy outpatient units, reducing requirements for weekly line flushing, pump disconnections, and other routine maintenance.
We continue to recommend the use of ToxNav germline genetic testing as a genetic screen for DPYD/ENOSF1 single-nucleotide polymorphisms (SNPs) to identify patients at high risk for fluoropyrimidine toxicity.
Use of biomarkers to sharpen prognosis should also be considered to refine therapeutic decisions.
Recommendations for stage II-III colorectal cancer
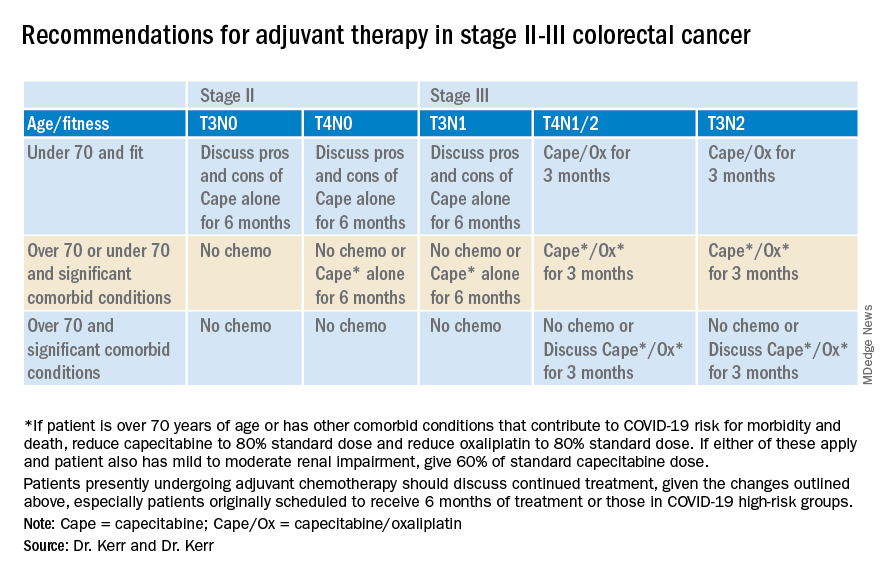
Recommendations for advanced colorectal cancer
Which regimen? Capecitabine/oxaliplatin should be the default backbone chemotherapy (rather than FOLFOX) in order to decrease the stress on infusion units.
Capecitabine plus irinotecan should be considered rather than FOLFIRI. However, in order to increase safety, reduce the dose of the capecitabine and the irinotecan, both to 80%, in all patient groups; and perhaps reduce the capecitabine dose further to 60% in those over the age of 70 or with significant comorbid conditions.
Treatment breaks. Full treatment breaks should be considered after 3 months of treatment in most patients with lower-volume, more indolent disease.
Treatment deintensification to capecitabine alone should be used in those with higher-volume disease (for example, more than 50% of liver replaced by tumor) at the beginning of treatment.
Deferring the start of any chemotherapy. Some older patients, or those with significant other comorbidities (that is, those who will be at increased risk for COVID-19 complications and death); who have low-volume disease, such as a couple of small lung metastases or a single liver metastasis; or who were diagnosed more than 12 months since adjuvant chemotherapy may decide to defer any chemotherapy for a period of time.
In these cases, we suggest rescanning at 3 months and discussing further treatment at that point. Some of these patients will be eligible for other interventions, such as resection, ablation, or stereotactic body radiation therapy. However, it will be important to consider the pressures on these other services during this unprecedented time.
Chemotherapy after resection of metastases. Given the lack of evidence and the present extenuating circumstances, we would not recommend any chemotherapy in this setting.
David J. Kerr, MD, CBE, MD, DSc, is a professor of cancer medicine at the University of Oxford. He is recognized internationally for his work in the research and treatment of colorectal cancer, and has founded three university spin-out companies: COBRA Therapeutics, Celleron Therapeutics, and Oxford Cancer Biomarkers. In 2002, he was appointed Commander of the British Empire by Queen Elizabeth. Rachel S. Kerr, MBChB, is a medical oncologist and associate professor of gastrointestinal oncology at the University of Oxford. She holds a UK Department of Health Fellowship, where she is clinical director of phase 3 trials in the oncology clinical trials office.
This article first appeared on Medscape.com.
In light of the rapid changes affecting cancer clinics due to the COVID-19 pandemic, Dr. David Kerr and Dr. Rachel Kerr, both specialists in gastrointestinal cancers at the University of Oxford in Oxford, United Kingdom, drafted these guidelines for the use of chemotherapy in colorectal cancer patients. Dr. Kerr and Dr. Kerr are putting forth this guidance as a topic for discussion and debate.
Our aim in developing these recommendations for the care of colorectal cancer patients in areas affected by the COVID-19 outbreak is to reduce the comorbidity of chemotherapy and decrease the risk of patients dying from COVID-19, weighed against the potential benefits of receiving chemotherapy. These recommendations are also designed to reduce the burden on chemotherapy units during a time of great pressure.
We have modified the guidelines in such a way that, we believe, will decrease the total number of patients receiving chemotherapy – particularly in the adjuvant setting – and reduce the overall immune impact of chemotherapy on these patients. Specifically, we suggest changing doublet chemotherapy to single-agent chemotherapy for some groups; changing to combinations involving capecitabine rather than bolus and infusional 5-FU for other patients; and, finally, making reasonable dose reductions upfront to reduce the risk for cycle 1 complications.
By changing from push-and-pump 5-FU to capecitabine for the vast majority of patients, we will both reduce the rates of neutropenia and decrease throughput in chemotherapy outpatient units, reducing requirements for weekly line flushing, pump disconnections, and other routine maintenance.
We continue to recommend the use of ToxNav germline genetic testing as a genetic screen for DPYD/ENOSF1 single-nucleotide polymorphisms (SNPs) to identify patients at high risk for fluoropyrimidine toxicity.
Use of biomarkers to sharpen prognosis should also be considered to refine therapeutic decisions.
Recommendations for stage II-III colorectal cancer

Recommendations for advanced colorectal cancer
Which regimen? Capecitabine/oxaliplatin should be the default backbone chemotherapy (rather than FOLFOX) in order to decrease the stress on infusion units.
Capecitabine plus irinotecan should be considered rather than FOLFIRI. However, in order to increase safety, reduce the dose of the capecitabine and the irinotecan, both to 80%, in all patient groups; and perhaps reduce the capecitabine dose further to 60% in those over the age of 70 or with significant comorbid conditions.
Treatment breaks. Full treatment breaks should be considered after 3 months of treatment in most patients with lower-volume, more indolent disease.
Treatment deintensification to capecitabine alone should be used in those with higher-volume disease (for example, more than 50% of liver replaced by tumor) at the beginning of treatment.
Deferring the start of any chemotherapy. Some older patients, or those with significant other comorbidities (that is, those who will be at increased risk for COVID-19 complications and death); who have low-volume disease, such as a couple of small lung metastases or a single liver metastasis; or who were diagnosed more than 12 months since adjuvant chemotherapy may decide to defer any chemotherapy for a period of time.
In these cases, we suggest rescanning at 3 months and discussing further treatment at that point. Some of these patients will be eligible for other interventions, such as resection, ablation, or stereotactic body radiation therapy. However, it will be important to consider the pressures on these other services during this unprecedented time.
Chemotherapy after resection of metastases. Given the lack of evidence and the present extenuating circumstances, we would not recommend any chemotherapy in this setting.
David J. Kerr, MD, CBE, MD, DSc, is a professor of cancer medicine at the University of Oxford. He is recognized internationally for his work in the research and treatment of colorectal cancer, and has founded three university spin-out companies: COBRA Therapeutics, Celleron Therapeutics, and Oxford Cancer Biomarkers. In 2002, he was appointed Commander of the British Empire by Queen Elizabeth. Rachel S. Kerr, MBChB, is a medical oncologist and associate professor of gastrointestinal oncology at the University of Oxford. She holds a UK Department of Health Fellowship, where she is clinical director of phase 3 trials in the oncology clinical trials office.
This article first appeared on Medscape.com.
In light of the rapid changes affecting cancer clinics due to the COVID-19 pandemic, Dr. David Kerr and Dr. Rachel Kerr, both specialists in gastrointestinal cancers at the University of Oxford in Oxford, United Kingdom, drafted these guidelines for the use of chemotherapy in colorectal cancer patients. Dr. Kerr and Dr. Kerr are putting forth this guidance as a topic for discussion and debate.
Our aim in developing these recommendations for the care of colorectal cancer patients in areas affected by the COVID-19 outbreak is to reduce the comorbidity of chemotherapy and decrease the risk of patients dying from COVID-19, weighed against the potential benefits of receiving chemotherapy. These recommendations are also designed to reduce the burden on chemotherapy units during a time of great pressure.
We have modified the guidelines in such a way that, we believe, will decrease the total number of patients receiving chemotherapy – particularly in the adjuvant setting – and reduce the overall immune impact of chemotherapy on these patients. Specifically, we suggest changing doublet chemotherapy to single-agent chemotherapy for some groups; changing to combinations involving capecitabine rather than bolus and infusional 5-FU for other patients; and, finally, making reasonable dose reductions upfront to reduce the risk for cycle 1 complications.
By changing from push-and-pump 5-FU to capecitabine for the vast majority of patients, we will both reduce the rates of neutropenia and decrease throughput in chemotherapy outpatient units, reducing requirements for weekly line flushing, pump disconnections, and other routine maintenance.
We continue to recommend the use of ToxNav germline genetic testing as a genetic screen for DPYD/ENOSF1 single-nucleotide polymorphisms (SNPs) to identify patients at high risk for fluoropyrimidine toxicity.
Use of biomarkers to sharpen prognosis should also be considered to refine therapeutic decisions.
Recommendations for stage II-III colorectal cancer

Recommendations for advanced colorectal cancer
Which regimen? Capecitabine/oxaliplatin should be the default backbone chemotherapy (rather than FOLFOX) in order to decrease the stress on infusion units.
Capecitabine plus irinotecan should be considered rather than FOLFIRI. However, in order to increase safety, reduce the dose of the capecitabine and the irinotecan, both to 80%, in all patient groups; and perhaps reduce the capecitabine dose further to 60% in those over the age of 70 or with significant comorbid conditions.
Treatment breaks. Full treatment breaks should be considered after 3 months of treatment in most patients with lower-volume, more indolent disease.
Treatment deintensification to capecitabine alone should be used in those with higher-volume disease (for example, more than 50% of liver replaced by tumor) at the beginning of treatment.
Deferring the start of any chemotherapy. Some older patients, or those with significant other comorbidities (that is, those who will be at increased risk for COVID-19 complications and death); who have low-volume disease, such as a couple of small lung metastases or a single liver metastasis; or who were diagnosed more than 12 months since adjuvant chemotherapy may decide to defer any chemotherapy for a period of time.
In these cases, we suggest rescanning at 3 months and discussing further treatment at that point. Some of these patients will be eligible for other interventions, such as resection, ablation, or stereotactic body radiation therapy. However, it will be important to consider the pressures on these other services during this unprecedented time.
Chemotherapy after resection of metastases. Given the lack of evidence and the present extenuating circumstances, we would not recommend any chemotherapy in this setting.
David J. Kerr, MD, CBE, MD, DSc, is a professor of cancer medicine at the University of Oxford. He is recognized internationally for his work in the research and treatment of colorectal cancer, and has founded three university spin-out companies: COBRA Therapeutics, Celleron Therapeutics, and Oxford Cancer Biomarkers. In 2002, he was appointed Commander of the British Empire by Queen Elizabeth. Rachel S. Kerr, MBChB, is a medical oncologist and associate professor of gastrointestinal oncology at the University of Oxford. She holds a UK Department of Health Fellowship, where she is clinical director of phase 3 trials in the oncology clinical trials office.
This article first appeared on Medscape.com.