User login
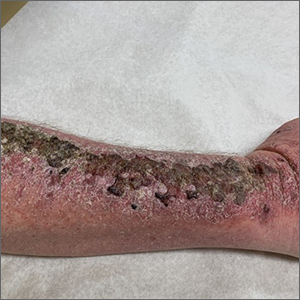
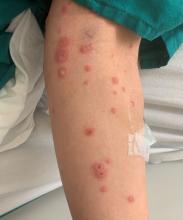
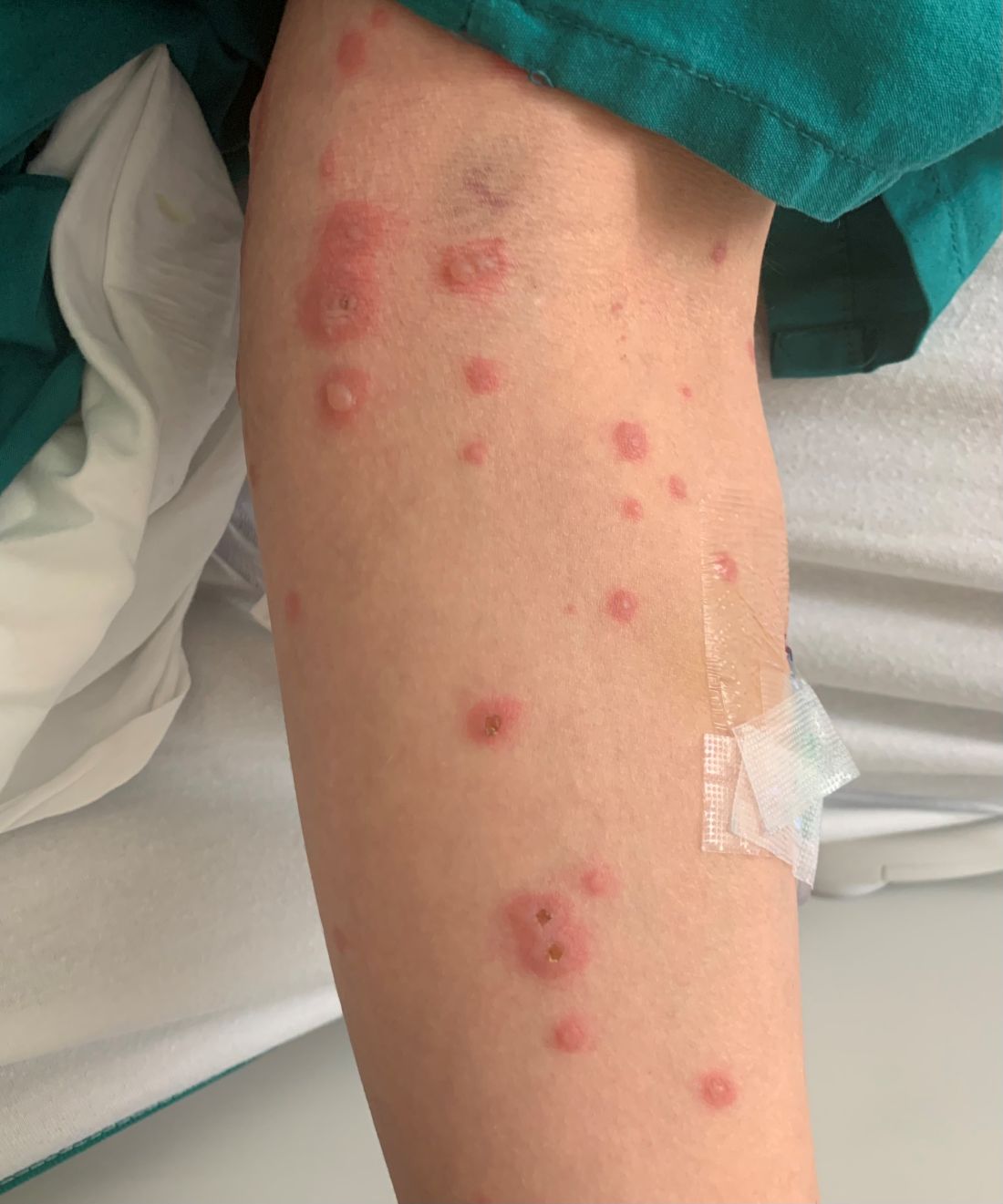
Right arm rash
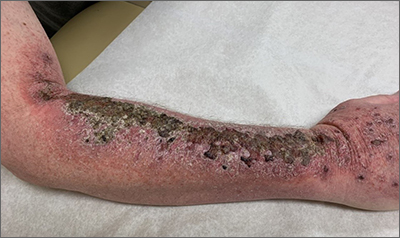
Common skin reactions to treatment with topical 5-FU for actinic keratosis include erythema, ulceration, and burning. In this case, however, the skin disruption opened the door to secondary impetigo.
Secondary skin infections are a known risk of treatment with topical 5-FU. The agent inhibits thymidylate synthetase, an enzyme involved in the synthesis and repair of DNA. The inhibition of this enzyme can lead to skin disruption with erosion, desquamation, and the risk of superimposed skin infections, as was seen with this patient.1
Impetigo is a common skin infection affecting the superficial layers of the epidermis and is most commonly caused by gram-positive bacteria, such as Staphylococcus aureus or Streptococcus pyogenes.2 Secondary impetigo, also known as impetiginization, is an infection of previously disrupted skin due to eczema, trauma, insect bites, and other conditions. This contrasts with primary impetigo, which results from a direct bacterial invasion of intact healthy skin. While impetigo predominantly affects children between the ages of 2 and 5 years, people of any age can be affected.2 Impetigo characteristically manifests with painful erosions, classically covered by honey-colored crusts. Thin-walled vesicles often appear and subsequently rupture.
Treatment options for impetigo include both topical and systemic antibiotics. Topical therapy is preferred for patients with limited skin involvement, while systemic therapy is indicated for patients with numerous lesions. Mupirocin and retapamulin are first-line topical treatments. Systemic antibiotic therapy should provide coverage for both S. aureus and streptococcal infections; cephalexin and dicloxacillin are preferred. Doxycycline, trimethoprim-sulfamethoxazole, or clindamycin can be used if methicillin-resistant Staphylococcus aureus is suspected.3
This patient was advised to try warm soaks (to reduce the crusting) and to follow that with the application of white petrolatum bid. The patient was also prescribed doxycycline 100 mg orally bid for 10 days. At the 1-month follow-up, there was some residual erythema, but the impetigo and crusting had resolved. The actinic keratoses had resolved, as well.
Image courtesy of Daniel Stulberg, MD. Text courtesy of Tess Pei Lemon, BA, University of New Mexico School of Medicine and Daniel Stulberg, MD, FAAFP, Department of Family and Community Medicine, University of New Mexico School of Medicine, Albuquerque
1. Chughtai K, Gupta R, Upadhaya S, et al. Topical 5-Fluorouracil associated skin reaction. Oxf Med Case Rep. 2017;2017(8):omx043. doi:10.1093/omcr/omx043
2. Hartman-Adams H, Banvard C, Juckett G. Impetigo: diagnosis and treatment. Am Fam Physician. 2014;90:229-235.
3. Stevens DL, Bisno AL, Chambers HF, et al. Practice guidelines for the diagnosis and management of skin and soft tissue infections: 2014 update by the Infectious Diseases Society of America. Clin Infect Dis. 2014;59:147-159. doi:10.1093/cid/ciu296

Common skin reactions to treatment with topical 5-FU for actinic keratosis include erythema, ulceration, and burning. In this case, however, the skin disruption opened the door to secondary impetigo.
Secondary skin infections are a known risk of treatment with topical 5-FU. The agent inhibits thymidylate synthetase, an enzyme involved in the synthesis and repair of DNA. The inhibition of this enzyme can lead to skin disruption with erosion, desquamation, and the risk of superimposed skin infections, as was seen with this patient.1
Impetigo is a common skin infection affecting the superficial layers of the epidermis and is most commonly caused by gram-positive bacteria, such as Staphylococcus aureus or Streptococcus pyogenes.2 Secondary impetigo, also known as impetiginization, is an infection of previously disrupted skin due to eczema, trauma, insect bites, and other conditions. This contrasts with primary impetigo, which results from a direct bacterial invasion of intact healthy skin. While impetigo predominantly affects children between the ages of 2 and 5 years, people of any age can be affected.2 Impetigo characteristically manifests with painful erosions, classically covered by honey-colored crusts. Thin-walled vesicles often appear and subsequently rupture.
Treatment options for impetigo include both topical and systemic antibiotics. Topical therapy is preferred for patients with limited skin involvement, while systemic therapy is indicated for patients with numerous lesions. Mupirocin and retapamulin are first-line topical treatments. Systemic antibiotic therapy should provide coverage for both S. aureus and streptococcal infections; cephalexin and dicloxacillin are preferred. Doxycycline, trimethoprim-sulfamethoxazole, or clindamycin can be used if methicillin-resistant Staphylococcus aureus is suspected.3
This patient was advised to try warm soaks (to reduce the crusting) and to follow that with the application of white petrolatum bid. The patient was also prescribed doxycycline 100 mg orally bid for 10 days. At the 1-month follow-up, there was some residual erythema, but the impetigo and crusting had resolved. The actinic keratoses had resolved, as well.
Image courtesy of Daniel Stulberg, MD. Text courtesy of Tess Pei Lemon, BA, University of New Mexico School of Medicine and Daniel Stulberg, MD, FAAFP, Department of Family and Community Medicine, University of New Mexico School of Medicine, Albuquerque

Common skin reactions to treatment with topical 5-FU for actinic keratosis include erythema, ulceration, and burning. In this case, however, the skin disruption opened the door to secondary impetigo.
Secondary skin infections are a known risk of treatment with topical 5-FU. The agent inhibits thymidylate synthetase, an enzyme involved in the synthesis and repair of DNA. The inhibition of this enzyme can lead to skin disruption with erosion, desquamation, and the risk of superimposed skin infections, as was seen with this patient.1
Impetigo is a common skin infection affecting the superficial layers of the epidermis and is most commonly caused by gram-positive bacteria, such as Staphylococcus aureus or Streptococcus pyogenes.2 Secondary impetigo, also known as impetiginization, is an infection of previously disrupted skin due to eczema, trauma, insect bites, and other conditions. This contrasts with primary impetigo, which results from a direct bacterial invasion of intact healthy skin. While impetigo predominantly affects children between the ages of 2 and 5 years, people of any age can be affected.2 Impetigo characteristically manifests with painful erosions, classically covered by honey-colored crusts. Thin-walled vesicles often appear and subsequently rupture.
Treatment options for impetigo include both topical and systemic antibiotics. Topical therapy is preferred for patients with limited skin involvement, while systemic therapy is indicated for patients with numerous lesions. Mupirocin and retapamulin are first-line topical treatments. Systemic antibiotic therapy should provide coverage for both S. aureus and streptococcal infections; cephalexin and dicloxacillin are preferred. Doxycycline, trimethoprim-sulfamethoxazole, or clindamycin can be used if methicillin-resistant Staphylococcus aureus is suspected.3
This patient was advised to try warm soaks (to reduce the crusting) and to follow that with the application of white petrolatum bid. The patient was also prescribed doxycycline 100 mg orally bid for 10 days. At the 1-month follow-up, there was some residual erythema, but the impetigo and crusting had resolved. The actinic keratoses had resolved, as well.
Image courtesy of Daniel Stulberg, MD. Text courtesy of Tess Pei Lemon, BA, University of New Mexico School of Medicine and Daniel Stulberg, MD, FAAFP, Department of Family and Community Medicine, University of New Mexico School of Medicine, Albuquerque
1. Chughtai K, Gupta R, Upadhaya S, et al. Topical 5-Fluorouracil associated skin reaction. Oxf Med Case Rep. 2017;2017(8):omx043. doi:10.1093/omcr/omx043
2. Hartman-Adams H, Banvard C, Juckett G. Impetigo: diagnosis and treatment. Am Fam Physician. 2014;90:229-235.
3. Stevens DL, Bisno AL, Chambers HF, et al. Practice guidelines for the diagnosis and management of skin and soft tissue infections: 2014 update by the Infectious Diseases Society of America. Clin Infect Dis. 2014;59:147-159. doi:10.1093/cid/ciu296
1. Chughtai K, Gupta R, Upadhaya S, et al. Topical 5-Fluorouracil associated skin reaction. Oxf Med Case Rep. 2017;2017(8):omx043. doi:10.1093/omcr/omx043
2. Hartman-Adams H, Banvard C, Juckett G. Impetigo: diagnosis and treatment. Am Fam Physician. 2014;90:229-235.
3. Stevens DL, Bisno AL, Chambers HF, et al. Practice guidelines for the diagnosis and management of skin and soft tissue infections: 2014 update by the Infectious Diseases Society of America. Clin Infect Dis. 2014;59:147-159. doi:10.1093/cid/ciu296
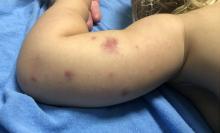
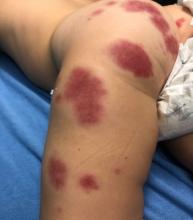
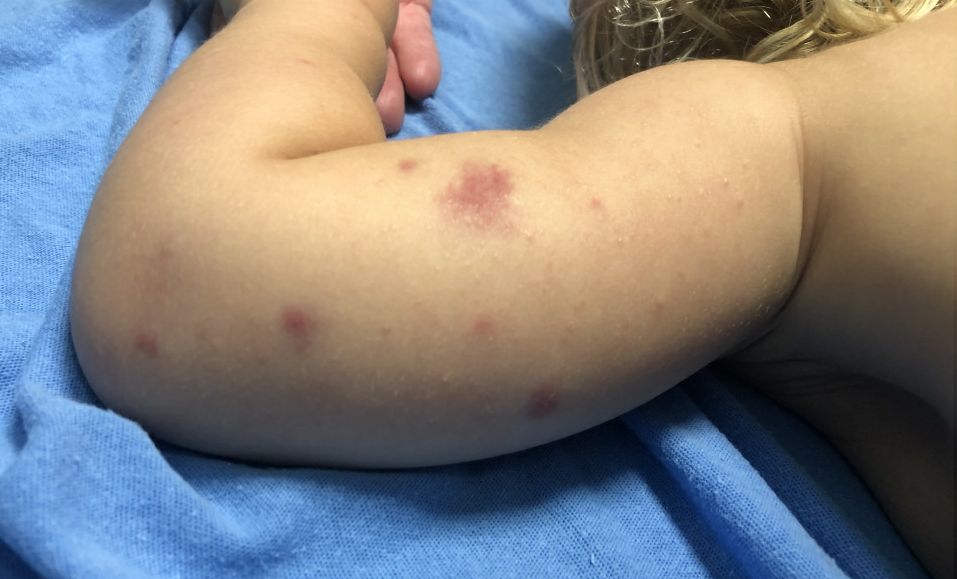
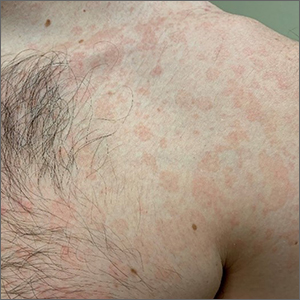
A 19-month-old vaccinated female with 2 days of rash
Acute hemorrhagic edema of infancy (AHEI) is a leukocytoclastic vasculitis that typically affects children between 4 months and 2 years of age.1 Etiology is unknown but the majority of cases are preceded by infections, vaccinations, or certain medications.2
AHEI is a self-limited disease that runs a benign course with spontaneous resolution within days to 3 weeks.3 Classic presentation involves acute onset of fever, purpura, ecchymosis, and inflammatory edema. Edema is often the first sign, and may involve the face, ears, scrotum, or extremities. Hemorrhagic lesions may vary in size but often coalesce and present in a distinctive “cockade” or rosette pattern with scalloped borders. Systemic manifestations are rare, but renal and joint involvement may occur.4 Despite the dramatic and sometimes extensive appearance of the dermatologic manifestations, patients with AHEI are usually not in significant distress.
Diagnosis is clinical, but skin biopsy may show leukocytoclastic vasculitis of the superficial small vessels with infiltrations of neutrophils, extravasation of red blood cells, and fibrinoid necrosis.5 In most cases, immunofluorescence is negative for perivascular IgA deposition. Treatment is symptomatic as the disease resolves spontaneously. Recurrence is uncommon but may occur, and usually occurs early.
What is on the differential?
Kawasaki disease. Similar to AHEI, patients with Kawasaki disease also may present with facial and extremity edema. However, patients with Kawasaki disease appear sicker, have associated lymphadenopathy, conjunctivitis, and fever longer than 5 days. The lack of elevated inflammatory markers, acute-onset, classic dermatologic lesions, and nontoxic appearance in our patient rule out Kawasaki disease and make AHEI more likely.
IgA vasculitis/Henoch-Schönlein purpura. The distinction between AHEI and Henoch-Schönlein purpura is among the most challenging. AHEI commonly afflicts younger children ranging from 4 months to 2 years, whereas Henoch-Schönlein purpura occurs in older children from 3 to 6 years of age. Visceral involvement is rare in AHEI, but frequently presents in Henoch-Schönlein purpura with gastrointestinal and renal complications. Although our patient had both mild renal involvement and a distribution primarily on the buttocks and lower limbs, similar to the classic distribution of Henoch-Schönlein purpura, the younger age and lack of gastrointestinal and arthritic manifestations make AHEI more likely.
Gianotti-Crosti syndrome. Gianotti-Crosti syndrome, also known as papulovesicular acrodermatitis of childhood, mainly affects children between the ages of 6 months and 12 years. Like AHEI, Gianotti-Crosti is a self-limiting condition likely triggered by viral infection or immunization. However, Gianotti-Crosti is characterized by a papular rash that may last for several weeks. Neither AHEI nor Gianotti-Crosti are pruritic, but patients with Gianotti-Crosti tend to have either inguinal or axillary lymphadenopathy. Our patient’s large, coalescing dusky red patches and edematous plaques without lymphadenopathy are more consistent with AHEI.
Erythema multiforme. Erythema multiforme is an acute, immune-mediated condition characterized by distinctive target-like lesions on the skin often accompanied by erosions or bullae. Unlike AHEI, erythema multiforme can involve the oral, genital, and/or ocular mucosae. Erythema multiforme is rare before the age of 4 years. Although the targetoid or annular purpuric configuration of erythema multiforme may present similarly to AHEI in some cases, the young age of our patient and the lack of mucosal involvement make AHEI more likely.
Dr. Matiz is a pediatric dermatologist at Southern California Permanente Medical Group, San Diego. Ms. Kleinman is a pediatric dermatology research associate at the University of California, San Diego, and Rady Children’s Hospital, San Diego. Neither Dr. Matiz nor Ms. Kleinman has any relevant financial disclosures.
References
1. Savino F et al. Pediatr Dermatol. 2013;30(6):e149-e152.
2. Carboni E et al. F1000Res. 2019;8:1771. 2019 Oct 17.
3. Fiore E et al. J Am Acad Dermatol. 2008;59(4):684-95.
4. Watanabe T and Sato Y. Pediatr Nephrol. 2007;22(11):1979-81.
5. Cunha DF et al. Autops Case Rep. 2015;5(3):37-41.
Acute hemorrhagic edema of infancy (AHEI) is a leukocytoclastic vasculitis that typically affects children between 4 months and 2 years of age.1 Etiology is unknown but the majority of cases are preceded by infections, vaccinations, or certain medications.2
AHEI is a self-limited disease that runs a benign course with spontaneous resolution within days to 3 weeks.3 Classic presentation involves acute onset of fever, purpura, ecchymosis, and inflammatory edema. Edema is often the first sign, and may involve the face, ears, scrotum, or extremities. Hemorrhagic lesions may vary in size but often coalesce and present in a distinctive “cockade” or rosette pattern with scalloped borders. Systemic manifestations are rare, but renal and joint involvement may occur.4 Despite the dramatic and sometimes extensive appearance of the dermatologic manifestations, patients with AHEI are usually not in significant distress.
Diagnosis is clinical, but skin biopsy may show leukocytoclastic vasculitis of the superficial small vessels with infiltrations of neutrophils, extravasation of red blood cells, and fibrinoid necrosis.5 In most cases, immunofluorescence is negative for perivascular IgA deposition. Treatment is symptomatic as the disease resolves spontaneously. Recurrence is uncommon but may occur, and usually occurs early.
What is on the differential?
Kawasaki disease. Similar to AHEI, patients with Kawasaki disease also may present with facial and extremity edema. However, patients with Kawasaki disease appear sicker, have associated lymphadenopathy, conjunctivitis, and fever longer than 5 days. The lack of elevated inflammatory markers, acute-onset, classic dermatologic lesions, and nontoxic appearance in our patient rule out Kawasaki disease and make AHEI more likely.
IgA vasculitis/Henoch-Schönlein purpura. The distinction between AHEI and Henoch-Schönlein purpura is among the most challenging. AHEI commonly afflicts younger children ranging from 4 months to 2 years, whereas Henoch-Schönlein purpura occurs in older children from 3 to 6 years of age. Visceral involvement is rare in AHEI, but frequently presents in Henoch-Schönlein purpura with gastrointestinal and renal complications. Although our patient had both mild renal involvement and a distribution primarily on the buttocks and lower limbs, similar to the classic distribution of Henoch-Schönlein purpura, the younger age and lack of gastrointestinal and arthritic manifestations make AHEI more likely.
Gianotti-Crosti syndrome. Gianotti-Crosti syndrome, also known as papulovesicular acrodermatitis of childhood, mainly affects children between the ages of 6 months and 12 years. Like AHEI, Gianotti-Crosti is a self-limiting condition likely triggered by viral infection or immunization. However, Gianotti-Crosti is characterized by a papular rash that may last for several weeks. Neither AHEI nor Gianotti-Crosti are pruritic, but patients with Gianotti-Crosti tend to have either inguinal or axillary lymphadenopathy. Our patient’s large, coalescing dusky red patches and edematous plaques without lymphadenopathy are more consistent with AHEI.
Erythema multiforme. Erythema multiforme is an acute, immune-mediated condition characterized by distinctive target-like lesions on the skin often accompanied by erosions or bullae. Unlike AHEI, erythema multiforme can involve the oral, genital, and/or ocular mucosae. Erythema multiforme is rare before the age of 4 years. Although the targetoid or annular purpuric configuration of erythema multiforme may present similarly to AHEI in some cases, the young age of our patient and the lack of mucosal involvement make AHEI more likely.
Dr. Matiz is a pediatric dermatologist at Southern California Permanente Medical Group, San Diego. Ms. Kleinman is a pediatric dermatology research associate at the University of California, San Diego, and Rady Children’s Hospital, San Diego. Neither Dr. Matiz nor Ms. Kleinman has any relevant financial disclosures.
References
1. Savino F et al. Pediatr Dermatol. 2013;30(6):e149-e152.
2. Carboni E et al. F1000Res. 2019;8:1771. 2019 Oct 17.
3. Fiore E et al. J Am Acad Dermatol. 2008;59(4):684-95.
4. Watanabe T and Sato Y. Pediatr Nephrol. 2007;22(11):1979-81.
5. Cunha DF et al. Autops Case Rep. 2015;5(3):37-41.
Acute hemorrhagic edema of infancy (AHEI) is a leukocytoclastic vasculitis that typically affects children between 4 months and 2 years of age.1 Etiology is unknown but the majority of cases are preceded by infections, vaccinations, or certain medications.2
AHEI is a self-limited disease that runs a benign course with spontaneous resolution within days to 3 weeks.3 Classic presentation involves acute onset of fever, purpura, ecchymosis, and inflammatory edema. Edema is often the first sign, and may involve the face, ears, scrotum, or extremities. Hemorrhagic lesions may vary in size but often coalesce and present in a distinctive “cockade” or rosette pattern with scalloped borders. Systemic manifestations are rare, but renal and joint involvement may occur.4 Despite the dramatic and sometimes extensive appearance of the dermatologic manifestations, patients with AHEI are usually not in significant distress.
Diagnosis is clinical, but skin biopsy may show leukocytoclastic vasculitis of the superficial small vessels with infiltrations of neutrophils, extravasation of red blood cells, and fibrinoid necrosis.5 In most cases, immunofluorescence is negative for perivascular IgA deposition. Treatment is symptomatic as the disease resolves spontaneously. Recurrence is uncommon but may occur, and usually occurs early.
What is on the differential?
Kawasaki disease. Similar to AHEI, patients with Kawasaki disease also may present with facial and extremity edema. However, patients with Kawasaki disease appear sicker, have associated lymphadenopathy, conjunctivitis, and fever longer than 5 days. The lack of elevated inflammatory markers, acute-onset, classic dermatologic lesions, and nontoxic appearance in our patient rule out Kawasaki disease and make AHEI more likely.
IgA vasculitis/Henoch-Schönlein purpura. The distinction between AHEI and Henoch-Schönlein purpura is among the most challenging. AHEI commonly afflicts younger children ranging from 4 months to 2 years, whereas Henoch-Schönlein purpura occurs in older children from 3 to 6 years of age. Visceral involvement is rare in AHEI, but frequently presents in Henoch-Schönlein purpura with gastrointestinal and renal complications. Although our patient had both mild renal involvement and a distribution primarily on the buttocks and lower limbs, similar to the classic distribution of Henoch-Schönlein purpura, the younger age and lack of gastrointestinal and arthritic manifestations make AHEI more likely.
Gianotti-Crosti syndrome. Gianotti-Crosti syndrome, also known as papulovesicular acrodermatitis of childhood, mainly affects children between the ages of 6 months and 12 years. Like AHEI, Gianotti-Crosti is a self-limiting condition likely triggered by viral infection or immunization. However, Gianotti-Crosti is characterized by a papular rash that may last for several weeks. Neither AHEI nor Gianotti-Crosti are pruritic, but patients with Gianotti-Crosti tend to have either inguinal or axillary lymphadenopathy. Our patient’s large, coalescing dusky red patches and edematous plaques without lymphadenopathy are more consistent with AHEI.
Erythema multiforme. Erythema multiforme is an acute, immune-mediated condition characterized by distinctive target-like lesions on the skin often accompanied by erosions or bullae. Unlike AHEI, erythema multiforme can involve the oral, genital, and/or ocular mucosae. Erythema multiforme is rare before the age of 4 years. Although the targetoid or annular purpuric configuration of erythema multiforme may present similarly to AHEI in some cases, the young age of our patient and the lack of mucosal involvement make AHEI more likely.
Dr. Matiz is a pediatric dermatologist at Southern California Permanente Medical Group, San Diego. Ms. Kleinman is a pediatric dermatology research associate at the University of California, San Diego, and Rady Children’s Hospital, San Diego. Neither Dr. Matiz nor Ms. Kleinman has any relevant financial disclosures.
References
1. Savino F et al. Pediatr Dermatol. 2013;30(6):e149-e152.
2. Carboni E et al. F1000Res. 2019;8:1771. 2019 Oct 17.
3. Fiore E et al. J Am Acad Dermatol. 2008;59(4):684-95.
4. Watanabe T and Sato Y. Pediatr Nephrol. 2007;22(11):1979-81.
5. Cunha DF et al. Autops Case Rep. 2015;5(3):37-41.
A 34-year-old male presented with 10 days of a pruritic rash
Less frequently observable infectious agents associated with EM are Mycoplasma pneumoniae, Histoplasma capsulatum, and parapoxvirus (orf). Rarely, EM is triggered by drug eruption or systemic disease. Individuals of all age groups and races can be affected by EM. However, it is predominantly observed in young adult patients (20-40 years of age), and there is a male predominance.
Patients typically present with the abrupt onset of symmetrical red papules that evolve into typical and atypical targetoid lesions. Lesions evolve in 48-72 hours, favoring acrofacial sites that then spread down towards the trunk. Systemic symptoms such as fever and arthralgia may accompany the skin lesions.1-3
Erythema multiforme is recognized in two forms: EM minor and EM major. Both forms share the same characteristic of target lesions. However, the presence of mucosal involvement distinguishes the two. Mucosal involvement is absent or mild in EM minor, while mucosal involvement in EM major is often severe.2,3 Painful bullous lesions are commonly present in the mouth, genital, and ocular mucous membranes. Severe symptoms can often result in difficulty eating and drinking.
Diagnosis is largely clinical. Further histologic study may accompany diagnoses to exclude differential diagnosis. In EM, direct immunofluorescence (DIF) is negative. Histopathology reveals apoptosis of individual keratinocytes.1,2
Therapeutic treatment for painful bullous lesions in the mouth involve antiseptic rinses and anesthetic solutions. Preventive treatment for patients with HSV-associated EM recurrence includes oral acyclovir or valacyclovir.2
In this patient, a punch biopsy was performed, confirming the diagnosis. A DIF was negative, and a chest x-ray was negative. Treatment was initiated with oral acyclovir, doxycycline, and a prednisone taper. In addition, topical clobetasol propionate and magic mouthwash (Maalox/lidocaine/nystatin) was prescribed. The patient was placed on daily suppressive valacyclovir to prevent frequent recurrence of EM.
This case and photo were submitted by Ms. Pham, the University of California, Los Angeles, and Dr. Sateesh, San Diego Family Dermatology. Dr. Bilu-Martin edited the column.
Dr. Bilu Martin is a board-certified dermatologist in private practice at Premier Dermatology, MD, in Aventura, Fla. More diagnostic cases are available at mdedge.com/dermatology. To submit a case for possible publication, send an email to [email protected].
References
1. Hafsi W and Badri T. Erythema Multiforme, in “StatPearls [Internet].” Treasure Island, Fla.: StatPearls Publishing, 2022 Jan.
2. Bolognia J et al. Dermatology. St. Louis: Mosby/Elsevier, 2008.
3. Oakley A. Erythema Multiforme. DermNet NZ. 2015 Oct.
Less frequently observable infectious agents associated with EM are Mycoplasma pneumoniae, Histoplasma capsulatum, and parapoxvirus (orf). Rarely, EM is triggered by drug eruption or systemic disease. Individuals of all age groups and races can be affected by EM. However, it is predominantly observed in young adult patients (20-40 years of age), and there is a male predominance.
Patients typically present with the abrupt onset of symmetrical red papules that evolve into typical and atypical targetoid lesions. Lesions evolve in 48-72 hours, favoring acrofacial sites that then spread down towards the trunk. Systemic symptoms such as fever and arthralgia may accompany the skin lesions.1-3
Erythema multiforme is recognized in two forms: EM minor and EM major. Both forms share the same characteristic of target lesions. However, the presence of mucosal involvement distinguishes the two. Mucosal involvement is absent or mild in EM minor, while mucosal involvement in EM major is often severe.2,3 Painful bullous lesions are commonly present in the mouth, genital, and ocular mucous membranes. Severe symptoms can often result in difficulty eating and drinking.
Diagnosis is largely clinical. Further histologic study may accompany diagnoses to exclude differential diagnosis. In EM, direct immunofluorescence (DIF) is negative. Histopathology reveals apoptosis of individual keratinocytes.1,2
Therapeutic treatment for painful bullous lesions in the mouth involve antiseptic rinses and anesthetic solutions. Preventive treatment for patients with HSV-associated EM recurrence includes oral acyclovir or valacyclovir.2
In this patient, a punch biopsy was performed, confirming the diagnosis. A DIF was negative, and a chest x-ray was negative. Treatment was initiated with oral acyclovir, doxycycline, and a prednisone taper. In addition, topical clobetasol propionate and magic mouthwash (Maalox/lidocaine/nystatin) was prescribed. The patient was placed on daily suppressive valacyclovir to prevent frequent recurrence of EM.
This case and photo were submitted by Ms. Pham, the University of California, Los Angeles, and Dr. Sateesh, San Diego Family Dermatology. Dr. Bilu-Martin edited the column.
Dr. Bilu Martin is a board-certified dermatologist in private practice at Premier Dermatology, MD, in Aventura, Fla. More diagnostic cases are available at mdedge.com/dermatology. To submit a case for possible publication, send an email to [email protected].
References
1. Hafsi W and Badri T. Erythema Multiforme, in “StatPearls [Internet].” Treasure Island, Fla.: StatPearls Publishing, 2022 Jan.
2. Bolognia J et al. Dermatology. St. Louis: Mosby/Elsevier, 2008.
3. Oakley A. Erythema Multiforme. DermNet NZ. 2015 Oct.
Less frequently observable infectious agents associated with EM are Mycoplasma pneumoniae, Histoplasma capsulatum, and parapoxvirus (orf). Rarely, EM is triggered by drug eruption or systemic disease. Individuals of all age groups and races can be affected by EM. However, it is predominantly observed in young adult patients (20-40 years of age), and there is a male predominance.
Patients typically present with the abrupt onset of symmetrical red papules that evolve into typical and atypical targetoid lesions. Lesions evolve in 48-72 hours, favoring acrofacial sites that then spread down towards the trunk. Systemic symptoms such as fever and arthralgia may accompany the skin lesions.1-3
Erythema multiforme is recognized in two forms: EM minor and EM major. Both forms share the same characteristic of target lesions. However, the presence of mucosal involvement distinguishes the two. Mucosal involvement is absent or mild in EM minor, while mucosal involvement in EM major is often severe.2,3 Painful bullous lesions are commonly present in the mouth, genital, and ocular mucous membranes. Severe symptoms can often result in difficulty eating and drinking.
Diagnosis is largely clinical. Further histologic study may accompany diagnoses to exclude differential diagnosis. In EM, direct immunofluorescence (DIF) is negative. Histopathology reveals apoptosis of individual keratinocytes.1,2
Therapeutic treatment for painful bullous lesions in the mouth involve antiseptic rinses and anesthetic solutions. Preventive treatment for patients with HSV-associated EM recurrence includes oral acyclovir or valacyclovir.2
In this patient, a punch biopsy was performed, confirming the diagnosis. A DIF was negative, and a chest x-ray was negative. Treatment was initiated with oral acyclovir, doxycycline, and a prednisone taper. In addition, topical clobetasol propionate and magic mouthwash (Maalox/lidocaine/nystatin) was prescribed. The patient was placed on daily suppressive valacyclovir to prevent frequent recurrence of EM.
This case and photo were submitted by Ms. Pham, the University of California, Los Angeles, and Dr. Sateesh, San Diego Family Dermatology. Dr. Bilu-Martin edited the column.
Dr. Bilu Martin is a board-certified dermatologist in private practice at Premier Dermatology, MD, in Aventura, Fla. More diagnostic cases are available at mdedge.com/dermatology. To submit a case for possible publication, send an email to [email protected].
References
1. Hafsi W and Badri T. Erythema Multiforme, in “StatPearls [Internet].” Treasure Island, Fla.: StatPearls Publishing, 2022 Jan.
2. Bolognia J et al. Dermatology. St. Louis: Mosby/Elsevier, 2008.
3. Oakley A. Erythema Multiforme. DermNet NZ. 2015 Oct.
Review finds anti-staphylococcus treatments have little impact on eczema
published in Clinical and Experimental Allergy.
Eczema remains a huge disease burden worldwide, and colonization with S. aureus in eczema patients is common, but no standard intervention exists to relieve symptoms, wrote Nandini Banerjee, MD, of Addenbrooke’s Hospital, Cambridge, England. “While antibiotic treatment of clinically obvious infections such as cellulitis is beneficial, it is not clear whether antibiotic treatment of eczema influences eczema severity,” Dr. Banerjee noted.
The 41 studies included 1,753 participants and 10 treatment categories. Most of the studies were conducted in secondary care centers in Western Europe, North America, and the Far East. Twelve studies included children, four included only adults, 19 included children and adults, and in six studies, the participant age range was unclear. Among the studies with reported ages, the mean age ranged from 1.1 to 34.6 years. Eczema severity ranged from mild to severe, and treatment durations ranged from 10 minutes to 3 months.
The review presented comparisons of topical steroid/antibiotic combinations, oral antibiotics, and bleach baths. In 14 studies that compared topical steroid/antibiotic combinations to topical steroids alone, patients showed slightly greater global improvement in symptoms with the combination, but the impact on quality of life was not significantly different. Severe adverse events, including flare of dermatitis, worsening of eczema, and folliculitis, were reported by the patients who received the combination and the topical steroid–only patients. One study reported similar rates of antibiotic resistance in children treated with steroid only and with an antibiotic/steroid combination at 3 months’ follow-up.
In four studies, oral antibiotics “may make no difference in terms of good or excellent global improvement in infants and children at 14 to 28 days follow-up compared to placebo,” according to the review. The reviewers said that there was likely little or no difference in quality of life for infants and children given oral antibiotics, although they noted the low quality of evidence on this topic.
Five studies evaluated the impact of bleach baths on eczema patients with and without S. aureus infections. These studies showed no difference in global improvement measures compared with placebo and little or no difference in quality of life. Also, patients who underwent bleach baths compared with placebo patients reported similar adverse events of burning/stinging or dry skin at 2 months’ follow-up.
“Low-quality evidence, due to risk of bias, imprecise effect estimates, and heterogeneity, made pooling of results difficult,” Dr. Banerjee wrote. “Topical steroid/antibiotic combinations may be associated with possible small improvements in good or excellent signs/symptoms compared with topical steroid alone. High-quality trials evaluating efficacy, QOL, and antibiotic resistance are required,” she concluded.
In a commentary section after the review, Dr. Banerjee and colleagues noted that the United Kingdom’s NICE guidelines for managing atopic eczema in children younger than 12 years of age, published in March 2021, include evidence from the current updated Cochrane Review. The NICE guidelines emphasize that “in people who are not systemically unwell, clinicians should not routinely offer either a topical or oral antibiotic for secondary bacterial infection of eczema,” the Cochrane authors said. They added in their commentary that the use of antibiotics in cases of nonsevere infections can worsen eczema. Also, “the risk of antimicrobial resistance is high with topical antibiotics, and therefore extended doses of the same antibiotics should be avoided to prevent resistance,” they said. However, the authors acknowledged a role for antibiotics in certain situations. “In patients with systemic signs of infection such as cellulitis, systemic antibiotics have an important role in helping clear infection,” they noted.
Reasons for varying disease severity elude research
The current study is important because of the abundance of preclinical and clinical data that implicate S. aureus in atopic dermatitis pathogenesis, Brian Kim, MD, of the Icahn School of Medicine at Mount Sinai, New York, said in an interview.
Dr. Kim said that he was surprised by some of the study findings but not others. “On the one hand, I thought there would be data supporting antimicrobial therapy, albeit not strong support,” he said. “However, AD is a very complex disease, and understanding what a disease modifier does to it is hard to capture across studies of various different designs,” he said.
“The data supporting antimicrobial therapy for S. aureus in AD is not as clear as our clinical impressions may indicate,” said Dr. Kim. “We need to understand the relationship better, perhaps in particular subsets of patients,” he emphasized. In addition, “We need a better understanding of why some people are colonized with S. aureus, yet with little effect on AD itself, while others experience severe exacerbation of disease,” said Dr. Kim. Therefore, a key research question for future studies is whether the exacerbation is caused by the particular strain of the bug, the host susceptibility, or both, he said.
The review received no outside funding. Dr. Banerjee and Dr. Kim have disclosed that they had no relevant financial relationships.
A version of this article first appeared on Medscape.com.
published in Clinical and Experimental Allergy.
Eczema remains a huge disease burden worldwide, and colonization with S. aureus in eczema patients is common, but no standard intervention exists to relieve symptoms, wrote Nandini Banerjee, MD, of Addenbrooke’s Hospital, Cambridge, England. “While antibiotic treatment of clinically obvious infections such as cellulitis is beneficial, it is not clear whether antibiotic treatment of eczema influences eczema severity,” Dr. Banerjee noted.
The 41 studies included 1,753 participants and 10 treatment categories. Most of the studies were conducted in secondary care centers in Western Europe, North America, and the Far East. Twelve studies included children, four included only adults, 19 included children and adults, and in six studies, the participant age range was unclear. Among the studies with reported ages, the mean age ranged from 1.1 to 34.6 years. Eczema severity ranged from mild to severe, and treatment durations ranged from 10 minutes to 3 months.
The review presented comparisons of topical steroid/antibiotic combinations, oral antibiotics, and bleach baths. In 14 studies that compared topical steroid/antibiotic combinations to topical steroids alone, patients showed slightly greater global improvement in symptoms with the combination, but the impact on quality of life was not significantly different. Severe adverse events, including flare of dermatitis, worsening of eczema, and folliculitis, were reported by the patients who received the combination and the topical steroid–only patients. One study reported similar rates of antibiotic resistance in children treated with steroid only and with an antibiotic/steroid combination at 3 months’ follow-up.
In four studies, oral antibiotics “may make no difference in terms of good or excellent global improvement in infants and children at 14 to 28 days follow-up compared to placebo,” according to the review. The reviewers said that there was likely little or no difference in quality of life for infants and children given oral antibiotics, although they noted the low quality of evidence on this topic.
Five studies evaluated the impact of bleach baths on eczema patients with and without S. aureus infections. These studies showed no difference in global improvement measures compared with placebo and little or no difference in quality of life. Also, patients who underwent bleach baths compared with placebo patients reported similar adverse events of burning/stinging or dry skin at 2 months’ follow-up.
“Low-quality evidence, due to risk of bias, imprecise effect estimates, and heterogeneity, made pooling of results difficult,” Dr. Banerjee wrote. “Topical steroid/antibiotic combinations may be associated with possible small improvements in good or excellent signs/symptoms compared with topical steroid alone. High-quality trials evaluating efficacy, QOL, and antibiotic resistance are required,” she concluded.
In a commentary section after the review, Dr. Banerjee and colleagues noted that the United Kingdom’s NICE guidelines for managing atopic eczema in children younger than 12 years of age, published in March 2021, include evidence from the current updated Cochrane Review. The NICE guidelines emphasize that “in people who are not systemically unwell, clinicians should not routinely offer either a topical or oral antibiotic for secondary bacterial infection of eczema,” the Cochrane authors said. They added in their commentary that the use of antibiotics in cases of nonsevere infections can worsen eczema. Also, “the risk of antimicrobial resistance is high with topical antibiotics, and therefore extended doses of the same antibiotics should be avoided to prevent resistance,” they said. However, the authors acknowledged a role for antibiotics in certain situations. “In patients with systemic signs of infection such as cellulitis, systemic antibiotics have an important role in helping clear infection,” they noted.
Reasons for varying disease severity elude research
The current study is important because of the abundance of preclinical and clinical data that implicate S. aureus in atopic dermatitis pathogenesis, Brian Kim, MD, of the Icahn School of Medicine at Mount Sinai, New York, said in an interview.
Dr. Kim said that he was surprised by some of the study findings but not others. “On the one hand, I thought there would be data supporting antimicrobial therapy, albeit not strong support,” he said. “However, AD is a very complex disease, and understanding what a disease modifier does to it is hard to capture across studies of various different designs,” he said.
“The data supporting antimicrobial therapy for S. aureus in AD is not as clear as our clinical impressions may indicate,” said Dr. Kim. “We need to understand the relationship better, perhaps in particular subsets of patients,” he emphasized. In addition, “We need a better understanding of why some people are colonized with S. aureus, yet with little effect on AD itself, while others experience severe exacerbation of disease,” said Dr. Kim. Therefore, a key research question for future studies is whether the exacerbation is caused by the particular strain of the bug, the host susceptibility, or both, he said.
The review received no outside funding. Dr. Banerjee and Dr. Kim have disclosed that they had no relevant financial relationships.
A version of this article first appeared on Medscape.com.
published in Clinical and Experimental Allergy.
Eczema remains a huge disease burden worldwide, and colonization with S. aureus in eczema patients is common, but no standard intervention exists to relieve symptoms, wrote Nandini Banerjee, MD, of Addenbrooke’s Hospital, Cambridge, England. “While antibiotic treatment of clinically obvious infections such as cellulitis is beneficial, it is not clear whether antibiotic treatment of eczema influences eczema severity,” Dr. Banerjee noted.
The 41 studies included 1,753 participants and 10 treatment categories. Most of the studies were conducted in secondary care centers in Western Europe, North America, and the Far East. Twelve studies included children, four included only adults, 19 included children and adults, and in six studies, the participant age range was unclear. Among the studies with reported ages, the mean age ranged from 1.1 to 34.6 years. Eczema severity ranged from mild to severe, and treatment durations ranged from 10 minutes to 3 months.
The review presented comparisons of topical steroid/antibiotic combinations, oral antibiotics, and bleach baths. In 14 studies that compared topical steroid/antibiotic combinations to topical steroids alone, patients showed slightly greater global improvement in symptoms with the combination, but the impact on quality of life was not significantly different. Severe adverse events, including flare of dermatitis, worsening of eczema, and folliculitis, were reported by the patients who received the combination and the topical steroid–only patients. One study reported similar rates of antibiotic resistance in children treated with steroid only and with an antibiotic/steroid combination at 3 months’ follow-up.
In four studies, oral antibiotics “may make no difference in terms of good or excellent global improvement in infants and children at 14 to 28 days follow-up compared to placebo,” according to the review. The reviewers said that there was likely little or no difference in quality of life for infants and children given oral antibiotics, although they noted the low quality of evidence on this topic.
Five studies evaluated the impact of bleach baths on eczema patients with and without S. aureus infections. These studies showed no difference in global improvement measures compared with placebo and little or no difference in quality of life. Also, patients who underwent bleach baths compared with placebo patients reported similar adverse events of burning/stinging or dry skin at 2 months’ follow-up.
“Low-quality evidence, due to risk of bias, imprecise effect estimates, and heterogeneity, made pooling of results difficult,” Dr. Banerjee wrote. “Topical steroid/antibiotic combinations may be associated with possible small improvements in good or excellent signs/symptoms compared with topical steroid alone. High-quality trials evaluating efficacy, QOL, and antibiotic resistance are required,” she concluded.
In a commentary section after the review, Dr. Banerjee and colleagues noted that the United Kingdom’s NICE guidelines for managing atopic eczema in children younger than 12 years of age, published in March 2021, include evidence from the current updated Cochrane Review. The NICE guidelines emphasize that “in people who are not systemically unwell, clinicians should not routinely offer either a topical or oral antibiotic for secondary bacterial infection of eczema,” the Cochrane authors said. They added in their commentary that the use of antibiotics in cases of nonsevere infections can worsen eczema. Also, “the risk of antimicrobial resistance is high with topical antibiotics, and therefore extended doses of the same antibiotics should be avoided to prevent resistance,” they said. However, the authors acknowledged a role for antibiotics in certain situations. “In patients with systemic signs of infection such as cellulitis, systemic antibiotics have an important role in helping clear infection,” they noted.
Reasons for varying disease severity elude research
The current study is important because of the abundance of preclinical and clinical data that implicate S. aureus in atopic dermatitis pathogenesis, Brian Kim, MD, of the Icahn School of Medicine at Mount Sinai, New York, said in an interview.
Dr. Kim said that he was surprised by some of the study findings but not others. “On the one hand, I thought there would be data supporting antimicrobial therapy, albeit not strong support,” he said. “However, AD is a very complex disease, and understanding what a disease modifier does to it is hard to capture across studies of various different designs,” he said.
“The data supporting antimicrobial therapy for S. aureus in AD is not as clear as our clinical impressions may indicate,” said Dr. Kim. “We need to understand the relationship better, perhaps in particular subsets of patients,” he emphasized. In addition, “We need a better understanding of why some people are colonized with S. aureus, yet with little effect on AD itself, while others experience severe exacerbation of disease,” said Dr. Kim. Therefore, a key research question for future studies is whether the exacerbation is caused by the particular strain of the bug, the host susceptibility, or both, he said.
The review received no outside funding. Dr. Banerjee and Dr. Kim have disclosed that they had no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FROM CLINICAL AND EXPERIMENTAL ALLERGY
Male alopecia agents ranked by efficacy in meta-analysis
While up to 90% of men experience AGA in their lifetime, only three therapies are currently approved for treatment of the condition by the Food and Drug Administration – topical minoxidil, oral finasteride 1 mg, and low-level light therapy.
However, with common use of off-label oral minoxidil, as well as oral dutasteride and higher doses of oral finasteride, the latter two being 5-alpha reductase inhibitors, Aditya K. Gupta, MD, PhD, of Mediprobe Research, in London, Ont., and colleagues sought to compare the data on the three agents. Their results were published in JAMA Dermatology.
They note that, while there have been recent comparisons between oral and topical minoxidil, “to our knowledge no study has determined the comparative effectiveness of these 2 [formulations] with that of local and systemic dutasteride and finasteride.”
For the meta-analysis, the authors identified 23 studies meeting their criteria, involving patients with mean ages ranging from 22.8 to 41.8 years.
For the primary endpoint of the greatest increases in total hair count at 24 weeks, the analysis showed the 0.5-mg/day dose of dutasteride topped the list, with significantly greater efficacy, compared with 1 mg/day of finasteride (mean difference, 7.1 hairs per cm2).
The 0.5-mg/d dutasteride dose also showed higher efficacy than oral minoxidil at 0.25 mg/day (mean difference, 23.7 hairs per cm2) and 5 mg/day (mean difference, 15.0 hairs per cm2) and topical minoxidil at 2% (mean difference, 8.5 hairs per cm2).
For the secondary endpoint of the greatest increase in terminal hair count at 24 weeks, the 5-mg/day dose of minoxidil had significantly greater efficacy compared with the 0.25-mg/day dose of the drug, as well as with minoxidil’s 2% and 5% topical formulations.
The minoxidil 5-mg/day dose was also significantly more effective than 1 mg/day of finasteride for terminal hair count at 24 weeks.
In longer-term outcomes at 48 weeks, the greatest increase in total hair count at 48 weeks was observed with 5 mg/day of finasteride, which was significantly more effective, compared with 2% topical minoxidil.
And the greatest increase in terminal hair count at 48 weeks was observed with 1 mg/day of oral finasteride, which was significantly more effective than 2% as well as 5% topical minoxidil.
Based on the results, the authors ranked the agents in a decreasing order of efficacy: 0.5 mg/day of oral dutasteride, 5 mg/day of oral finasteride, 5 mg/day of oral minoxidil, 1 mg/day of oral finasteride, 5% topical minoxidil, 2% topical minoxidil, and 0.25 mg/day of oral minoxidil.
Commenting on the analysis in an accompanying editorial, Kathie P. Huang, MD, of the department of dermatology, Brigham and Women’s Hospital, Boston, and Maryanne M. Senna, MD, of the department of dermatology, Massachusetts General Hospital, Boston, said the results, in general, are consistent with their experiences, noting that 2% minoxidil is typically not used in men.
They noted that, “although topical minoxidil ranked higher than the very-low-dose 0.25 mg oral minoxidil, our personal experience is that oral minoxidil at doses of 1.25 mg to 5 mg are far superior to topical minoxidil for treating AGA.”
Adverse event considerations important
Importantly, however, strong consideration needs to be given to adverse-event profiles, as well as patient comorbidities in selecting agents, the editorial authors asserted.
With 1 mg finasteride, for instance, potential adverse events include decreased libido, erectile dysfunction, decreased ejaculatory volume, reduction in sperm count, testicular pain, depression, and gynecomastia, they noted.
And while finasteride appears to be associated with a decreased risk of prostate cancer, those receiving the drug who do develop prostate cancer may be diagnosed with higher-grade prostate cancer; however, that “might be related to tissue sampling artifact,” the editorial authors said.
Less has been published on dutasteride’s adverse-event profile, and that, in itself, is a concern.
Overall, “as more direct-to-consumer companies treating male AGA emerge, it is especially important that the potential risks of these medications be made clear to patients,” they added.
Further commenting on the analysis to this news organization, Antonella Tosti, MD, the Fredric Brandt Endowed Professor of Dermatology and Cutaneous Surgery at the University of Miami, said the study offers some important insights – and caveats.
“I think this is a very interesting study, but you have to consider what works for your patients,” she said.
Dr. Tosti noted that the 5-mg dose of minoxidil is a concern in terms of side effects. “That dose is pretty high and could feasibly cause some hypertrichosis, which can be a concern to men as well as women.”
She agrees that the lack of data on side effects with dutasteride is also a concern, especially in light of some of the known side effects with other agents.
“That’s why I don’t use it very much in younger patients – because I’m afraid it could potentially affect their fertility,” Dr. Tosti said.
In general, Dr. Tosti said she finds a combination of agents provides the best results, as many clinicians use.
“I find dutasteride (0.5 mg/day) plus oral minoxidil (1-2.5 mg/day) plus topical 5% minoxidil is the best combination,” she said.
The authors and Dr. Tosti disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
While up to 90% of men experience AGA in their lifetime, only three therapies are currently approved for treatment of the condition by the Food and Drug Administration – topical minoxidil, oral finasteride 1 mg, and low-level light therapy.
However, with common use of off-label oral minoxidil, as well as oral dutasteride and higher doses of oral finasteride, the latter two being 5-alpha reductase inhibitors, Aditya K. Gupta, MD, PhD, of Mediprobe Research, in London, Ont., and colleagues sought to compare the data on the three agents. Their results were published in JAMA Dermatology.
They note that, while there have been recent comparisons between oral and topical minoxidil, “to our knowledge no study has determined the comparative effectiveness of these 2 [formulations] with that of local and systemic dutasteride and finasteride.”
For the meta-analysis, the authors identified 23 studies meeting their criteria, involving patients with mean ages ranging from 22.8 to 41.8 years.
For the primary endpoint of the greatest increases in total hair count at 24 weeks, the analysis showed the 0.5-mg/day dose of dutasteride topped the list, with significantly greater efficacy, compared with 1 mg/day of finasteride (mean difference, 7.1 hairs per cm2).
The 0.5-mg/d dutasteride dose also showed higher efficacy than oral minoxidil at 0.25 mg/day (mean difference, 23.7 hairs per cm2) and 5 mg/day (mean difference, 15.0 hairs per cm2) and topical minoxidil at 2% (mean difference, 8.5 hairs per cm2).
For the secondary endpoint of the greatest increase in terminal hair count at 24 weeks, the 5-mg/day dose of minoxidil had significantly greater efficacy compared with the 0.25-mg/day dose of the drug, as well as with minoxidil’s 2% and 5% topical formulations.
The minoxidil 5-mg/day dose was also significantly more effective than 1 mg/day of finasteride for terminal hair count at 24 weeks.
In longer-term outcomes at 48 weeks, the greatest increase in total hair count at 48 weeks was observed with 5 mg/day of finasteride, which was significantly more effective, compared with 2% topical minoxidil.
And the greatest increase in terminal hair count at 48 weeks was observed with 1 mg/day of oral finasteride, which was significantly more effective than 2% as well as 5% topical minoxidil.
Based on the results, the authors ranked the agents in a decreasing order of efficacy: 0.5 mg/day of oral dutasteride, 5 mg/day of oral finasteride, 5 mg/day of oral minoxidil, 1 mg/day of oral finasteride, 5% topical minoxidil, 2% topical minoxidil, and 0.25 mg/day of oral minoxidil.
Commenting on the analysis in an accompanying editorial, Kathie P. Huang, MD, of the department of dermatology, Brigham and Women’s Hospital, Boston, and Maryanne M. Senna, MD, of the department of dermatology, Massachusetts General Hospital, Boston, said the results, in general, are consistent with their experiences, noting that 2% minoxidil is typically not used in men.
They noted that, “although topical minoxidil ranked higher than the very-low-dose 0.25 mg oral minoxidil, our personal experience is that oral minoxidil at doses of 1.25 mg to 5 mg are far superior to topical minoxidil for treating AGA.”
Adverse event considerations important
Importantly, however, strong consideration needs to be given to adverse-event profiles, as well as patient comorbidities in selecting agents, the editorial authors asserted.
With 1 mg finasteride, for instance, potential adverse events include decreased libido, erectile dysfunction, decreased ejaculatory volume, reduction in sperm count, testicular pain, depression, and gynecomastia, they noted.
And while finasteride appears to be associated with a decreased risk of prostate cancer, those receiving the drug who do develop prostate cancer may be diagnosed with higher-grade prostate cancer; however, that “might be related to tissue sampling artifact,” the editorial authors said.
Less has been published on dutasteride’s adverse-event profile, and that, in itself, is a concern.
Overall, “as more direct-to-consumer companies treating male AGA emerge, it is especially important that the potential risks of these medications be made clear to patients,” they added.
Further commenting on the analysis to this news organization, Antonella Tosti, MD, the Fredric Brandt Endowed Professor of Dermatology and Cutaneous Surgery at the University of Miami, said the study offers some important insights – and caveats.
“I think this is a very interesting study, but you have to consider what works for your patients,” she said.
Dr. Tosti noted that the 5-mg dose of minoxidil is a concern in terms of side effects. “That dose is pretty high and could feasibly cause some hypertrichosis, which can be a concern to men as well as women.”
She agrees that the lack of data on side effects with dutasteride is also a concern, especially in light of some of the known side effects with other agents.
“That’s why I don’t use it very much in younger patients – because I’m afraid it could potentially affect their fertility,” Dr. Tosti said.
In general, Dr. Tosti said she finds a combination of agents provides the best results, as many clinicians use.
“I find dutasteride (0.5 mg/day) plus oral minoxidil (1-2.5 mg/day) plus topical 5% minoxidil is the best combination,” she said.
The authors and Dr. Tosti disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
While up to 90% of men experience AGA in their lifetime, only three therapies are currently approved for treatment of the condition by the Food and Drug Administration – topical minoxidil, oral finasteride 1 mg, and low-level light therapy.
However, with common use of off-label oral minoxidil, as well as oral dutasteride and higher doses of oral finasteride, the latter two being 5-alpha reductase inhibitors, Aditya K. Gupta, MD, PhD, of Mediprobe Research, in London, Ont., and colleagues sought to compare the data on the three agents. Their results were published in JAMA Dermatology.
They note that, while there have been recent comparisons between oral and topical minoxidil, “to our knowledge no study has determined the comparative effectiveness of these 2 [formulations] with that of local and systemic dutasteride and finasteride.”
For the meta-analysis, the authors identified 23 studies meeting their criteria, involving patients with mean ages ranging from 22.8 to 41.8 years.
For the primary endpoint of the greatest increases in total hair count at 24 weeks, the analysis showed the 0.5-mg/day dose of dutasteride topped the list, with significantly greater efficacy, compared with 1 mg/day of finasteride (mean difference, 7.1 hairs per cm2).
The 0.5-mg/d dutasteride dose also showed higher efficacy than oral minoxidil at 0.25 mg/day (mean difference, 23.7 hairs per cm2) and 5 mg/day (mean difference, 15.0 hairs per cm2) and topical minoxidil at 2% (mean difference, 8.5 hairs per cm2).
For the secondary endpoint of the greatest increase in terminal hair count at 24 weeks, the 5-mg/day dose of minoxidil had significantly greater efficacy compared with the 0.25-mg/day dose of the drug, as well as with minoxidil’s 2% and 5% topical formulations.
The minoxidil 5-mg/day dose was also significantly more effective than 1 mg/day of finasteride for terminal hair count at 24 weeks.
In longer-term outcomes at 48 weeks, the greatest increase in total hair count at 48 weeks was observed with 5 mg/day of finasteride, which was significantly more effective, compared with 2% topical minoxidil.
And the greatest increase in terminal hair count at 48 weeks was observed with 1 mg/day of oral finasteride, which was significantly more effective than 2% as well as 5% topical minoxidil.
Based on the results, the authors ranked the agents in a decreasing order of efficacy: 0.5 mg/day of oral dutasteride, 5 mg/day of oral finasteride, 5 mg/day of oral minoxidil, 1 mg/day of oral finasteride, 5% topical minoxidil, 2% topical minoxidil, and 0.25 mg/day of oral minoxidil.
Commenting on the analysis in an accompanying editorial, Kathie P. Huang, MD, of the department of dermatology, Brigham and Women’s Hospital, Boston, and Maryanne M. Senna, MD, of the department of dermatology, Massachusetts General Hospital, Boston, said the results, in general, are consistent with their experiences, noting that 2% minoxidil is typically not used in men.
They noted that, “although topical minoxidil ranked higher than the very-low-dose 0.25 mg oral minoxidil, our personal experience is that oral minoxidil at doses of 1.25 mg to 5 mg are far superior to topical minoxidil for treating AGA.”
Adverse event considerations important
Importantly, however, strong consideration needs to be given to adverse-event profiles, as well as patient comorbidities in selecting agents, the editorial authors asserted.
With 1 mg finasteride, for instance, potential adverse events include decreased libido, erectile dysfunction, decreased ejaculatory volume, reduction in sperm count, testicular pain, depression, and gynecomastia, they noted.
And while finasteride appears to be associated with a decreased risk of prostate cancer, those receiving the drug who do develop prostate cancer may be diagnosed with higher-grade prostate cancer; however, that “might be related to tissue sampling artifact,” the editorial authors said.
Less has been published on dutasteride’s adverse-event profile, and that, in itself, is a concern.
Overall, “as more direct-to-consumer companies treating male AGA emerge, it is especially important that the potential risks of these medications be made clear to patients,” they added.
Further commenting on the analysis to this news organization, Antonella Tosti, MD, the Fredric Brandt Endowed Professor of Dermatology and Cutaneous Surgery at the University of Miami, said the study offers some important insights – and caveats.
“I think this is a very interesting study, but you have to consider what works for your patients,” she said.
Dr. Tosti noted that the 5-mg dose of minoxidil is a concern in terms of side effects. “That dose is pretty high and could feasibly cause some hypertrichosis, which can be a concern to men as well as women.”
She agrees that the lack of data on side effects with dutasteride is also a concern, especially in light of some of the known side effects with other agents.
“That’s why I don’t use it very much in younger patients – because I’m afraid it could potentially affect their fertility,” Dr. Tosti said.
In general, Dr. Tosti said she finds a combination of agents provides the best results, as many clinicians use.
“I find dutasteride (0.5 mg/day) plus oral minoxidil (1-2.5 mg/day) plus topical 5% minoxidil is the best combination,” she said.
The authors and Dr. Tosti disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FROM JAMA DERMATOLOGY
Dupilumab under FDA review for atopic dermatitis in children aged 6 months to 5 years
The and Sanofi.
If approved, dupilumab would be the first biologic approved for children in this age group in the United States, according to the statement. The proposed indication is as add-on therapy for children with moderate to severe AD not adequately controlled with topical prescription therapies or for whom topical therapies are not advised. The FDA granted breakthrough therapy designation for dupilumab for the treatment of severe AD in children aged 6 months to 11 years in 2016.
Approximately 85%-95% of atopic dermatitis patients develop symptoms before 5 years of age, and these symptoms often continue into adulthood, with an increased risk of skin infections and a significant impact on quality of life, according to the statement.
The sBLA is based on data from a phase 3 pivotal study of 162 children aged 6 months to 5 years in which dupilumab was added to standard-of-care topical corticosteroids, presented in December 2021. In the study, dupilumab plus standard of care significantly improved skin clearance and reduced overall disease severity and itch at 16 weeks compared with standard of care alone. Overall, 28% of the children randomized to dupilumab achieved the primary endpoint of clear or almost-clear skin, compared with 4% with those on standard of care alone (P < .0001), according to the manufacturers. Patients in the dupilumab group received either 200 mg (for children weighing ≥ 5 to < 15 kg) or 300 mg (for children weighing ≥ 15 to < 30 kg) every 4 weeks. Safety results were similar to those seen with dupilumab for children aged 6 years and older.
Conjunctivitis and herpes infections were among the most common adverse events associated with dupilumab in the study, according to the statement.
The target action date for the FDA decision on this application is June 9, 2022.
The and Sanofi.
If approved, dupilumab would be the first biologic approved for children in this age group in the United States, according to the statement. The proposed indication is as add-on therapy for children with moderate to severe AD not adequately controlled with topical prescription therapies or for whom topical therapies are not advised. The FDA granted breakthrough therapy designation for dupilumab for the treatment of severe AD in children aged 6 months to 11 years in 2016.
Approximately 85%-95% of atopic dermatitis patients develop symptoms before 5 years of age, and these symptoms often continue into adulthood, with an increased risk of skin infections and a significant impact on quality of life, according to the statement.
The sBLA is based on data from a phase 3 pivotal study of 162 children aged 6 months to 5 years in which dupilumab was added to standard-of-care topical corticosteroids, presented in December 2021. In the study, dupilumab plus standard of care significantly improved skin clearance and reduced overall disease severity and itch at 16 weeks compared with standard of care alone. Overall, 28% of the children randomized to dupilumab achieved the primary endpoint of clear or almost-clear skin, compared with 4% with those on standard of care alone (P < .0001), according to the manufacturers. Patients in the dupilumab group received either 200 mg (for children weighing ≥ 5 to < 15 kg) or 300 mg (for children weighing ≥ 15 to < 30 kg) every 4 weeks. Safety results were similar to those seen with dupilumab for children aged 6 years and older.
Conjunctivitis and herpes infections were among the most common adverse events associated with dupilumab in the study, according to the statement.
The target action date for the FDA decision on this application is June 9, 2022.
The and Sanofi.
If approved, dupilumab would be the first biologic approved for children in this age group in the United States, according to the statement. The proposed indication is as add-on therapy for children with moderate to severe AD not adequately controlled with topical prescription therapies or for whom topical therapies are not advised. The FDA granted breakthrough therapy designation for dupilumab for the treatment of severe AD in children aged 6 months to 11 years in 2016.
Approximately 85%-95% of atopic dermatitis patients develop symptoms before 5 years of age, and these symptoms often continue into adulthood, with an increased risk of skin infections and a significant impact on quality of life, according to the statement.
The sBLA is based on data from a phase 3 pivotal study of 162 children aged 6 months to 5 years in which dupilumab was added to standard-of-care topical corticosteroids, presented in December 2021. In the study, dupilumab plus standard of care significantly improved skin clearance and reduced overall disease severity and itch at 16 weeks compared with standard of care alone. Overall, 28% of the children randomized to dupilumab achieved the primary endpoint of clear or almost-clear skin, compared with 4% with those on standard of care alone (P < .0001), according to the manufacturers. Patients in the dupilumab group received either 200 mg (for children weighing ≥ 5 to < 15 kg) or 300 mg (for children weighing ≥ 15 to < 30 kg) every 4 weeks. Safety results were similar to those seen with dupilumab for children aged 6 years and older.
Conjunctivitis and herpes infections were among the most common adverse events associated with dupilumab in the study, according to the statement.
The target action date for the FDA decision on this application is June 9, 2022.
FROM THE FDA
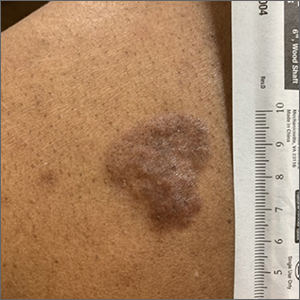
Chronic truncal rash
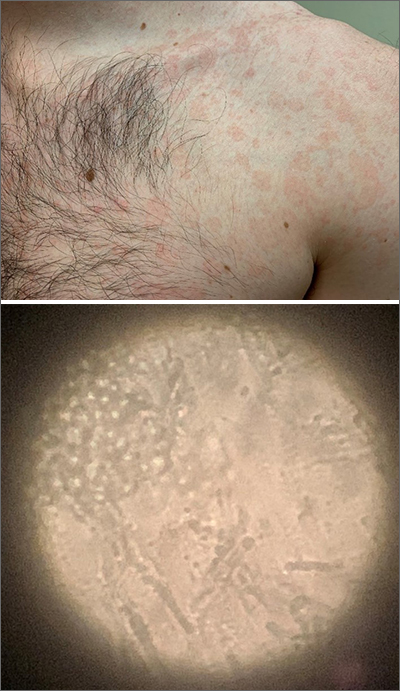
The patient was given a diagnosis of tinea versicolor (TV), also known as pityriasis versicolor, after a potassium hydroxide (KOH) prep test on a skin scraping confirmed the spaghetti and meatballs pattern of Malassezia Furfur (see image above). Note that in most cases, a KOH prep is not required for the diagnosis. Experienced clinicians will usually make the diagnosis based on the appearance of hyper- or hypopigmented macules or patches with fine scale on the trunk of adults. KOH prep is useful if the diagnosis is uncertain.
TV is a common fungal infection that’s seen more frequently in tropical climates and occurs equally in men and women.1 M. Furfur thrives on the lipids in the skin of sebum-rich areas, which explains its truncal distribution and rare occurrence in children (who have much lower sebum production).
Usually, topical antifungal medications are considered first-line treatment, but since large areas of skin are involved, adequate amounts need to be used. One of the most common and inexpensive treatments is to apply selenium sulfide shampoo (Selsun Blue) undiluted to the entire trunk, then allow to dry and remain in place overnight before showering. A repeat application should be done 1 week later. Topical terbinafine cream applied bid for 2 weeks is another option, as is oral itraconazole in a single 400 mg dose.
This patient declined the selenium sulfide topical treatment and requested systemic therapy, so he was prescribed itraconazole 400 mg orally as a single dose. He was advised that it might take a few weeks to clear up, and to use the selenium sulfide application if the itraconazole was not effective. Follow-up was not planned due to the high success rate of these therapies.
Image courtesy of Daniel Stulberg, MD. Text courtesy of Daniel Stulberg, MD, FAAFP, Department of Family and Community Medicine, University of New Mexico School of Medicine, Albuquerque.
1. Saunte DML, Gaitanis G, Hay RJ. Malassezia-associated skin diseases, the use of diagnostics and treatment. Front Cell Infect Microbiol. 2020;10:112. doi: 10.3389/fcimb.2020.00112

The patient was given a diagnosis of tinea versicolor (TV), also known as pityriasis versicolor, after a potassium hydroxide (KOH) prep test on a skin scraping confirmed the spaghetti and meatballs pattern of Malassezia Furfur (see image above). Note that in most cases, a KOH prep is not required for the diagnosis. Experienced clinicians will usually make the diagnosis based on the appearance of hyper- or hypopigmented macules or patches with fine scale on the trunk of adults. KOH prep is useful if the diagnosis is uncertain.
TV is a common fungal infection that’s seen more frequently in tropical climates and occurs equally in men and women.1 M. Furfur thrives on the lipids in the skin of sebum-rich areas, which explains its truncal distribution and rare occurrence in children (who have much lower sebum production).
Usually, topical antifungal medications are considered first-line treatment, but since large areas of skin are involved, adequate amounts need to be used. One of the most common and inexpensive treatments is to apply selenium sulfide shampoo (Selsun Blue) undiluted to the entire trunk, then allow to dry and remain in place overnight before showering. A repeat application should be done 1 week later. Topical terbinafine cream applied bid for 2 weeks is another option, as is oral itraconazole in a single 400 mg dose.
This patient declined the selenium sulfide topical treatment and requested systemic therapy, so he was prescribed itraconazole 400 mg orally as a single dose. He was advised that it might take a few weeks to clear up, and to use the selenium sulfide application if the itraconazole was not effective. Follow-up was not planned due to the high success rate of these therapies.
Image courtesy of Daniel Stulberg, MD. Text courtesy of Daniel Stulberg, MD, FAAFP, Department of Family and Community Medicine, University of New Mexico School of Medicine, Albuquerque.

The patient was given a diagnosis of tinea versicolor (TV), also known as pityriasis versicolor, after a potassium hydroxide (KOH) prep test on a skin scraping confirmed the spaghetti and meatballs pattern of Malassezia Furfur (see image above). Note that in most cases, a KOH prep is not required for the diagnosis. Experienced clinicians will usually make the diagnosis based on the appearance of hyper- or hypopigmented macules or patches with fine scale on the trunk of adults. KOH prep is useful if the diagnosis is uncertain.
TV is a common fungal infection that’s seen more frequently in tropical climates and occurs equally in men and women.1 M. Furfur thrives on the lipids in the skin of sebum-rich areas, which explains its truncal distribution and rare occurrence in children (who have much lower sebum production).
Usually, topical antifungal medications are considered first-line treatment, but since large areas of skin are involved, adequate amounts need to be used. One of the most common and inexpensive treatments is to apply selenium sulfide shampoo (Selsun Blue) undiluted to the entire trunk, then allow to dry and remain in place overnight before showering. A repeat application should be done 1 week later. Topical terbinafine cream applied bid for 2 weeks is another option, as is oral itraconazole in a single 400 mg dose.
This patient declined the selenium sulfide topical treatment and requested systemic therapy, so he was prescribed itraconazole 400 mg orally as a single dose. He was advised that it might take a few weeks to clear up, and to use the selenium sulfide application if the itraconazole was not effective. Follow-up was not planned due to the high success rate of these therapies.
Image courtesy of Daniel Stulberg, MD. Text courtesy of Daniel Stulberg, MD, FAAFP, Department of Family and Community Medicine, University of New Mexico School of Medicine, Albuquerque.
1. Saunte DML, Gaitanis G, Hay RJ. Malassezia-associated skin diseases, the use of diagnostics and treatment. Front Cell Infect Microbiol. 2020;10:112. doi: 10.3389/fcimb.2020.00112
1. Saunte DML, Gaitanis G, Hay RJ. Malassezia-associated skin diseases, the use of diagnostics and treatment. Front Cell Infect Microbiol. 2020;10:112. doi: 10.3389/fcimb.2020.00112
Expert shares workup pearls for children with severe atopic dermatitis
“Many patients who have failed topical steroids have never had adequate treatment,” Anna Yasmine Kirkorian, MD, chief of dermatology at National Children’s Hospital in Washington, said during the ODAC Dermatology, Aesthetic & Surgical Conference. “There is no lower age limit on the use of topical corticosteroids, and low potency corticosteroids are inadequate to treat severe eczema. The idea that only over-the-counter 2.5% hydrocortisone cream is necessary is not true,” she added.
“You also want to scrutinize the vehicle,” she said, noting that children are often prescribed cream formulations that hurt when applied, so parents stop applying them. “Ointments are generally the vehicles of choice in childhood,” she added.
It is generally not advised to use topical and oral antibiotics in children with AD, unless there are clear signs of infection. “If they’re just slightly oozy, don’t use them,” she continued. “Of course, every child or adult with eczema has Staph aureus on them, but most of the time, what you need to do is repair the barrier. We know that from data and common sense. When we repair their barrier, their rates of infection decrease.”
A focal area with pustules and pus should be cultured and treated, Dr. Kirkorian said. “Monotherapy with antibiotics is going to do nothing for you.” In cases of children with failure to thrive, she recommends referral to pediatric dermatology, allergy/immunology, GI, or genetics, as appropriate.
For children with severe AD, Dr. Kirkorian favors a rescue plan with a one-pound jar of triamcinolone ointment 0.1%. She recommends application of the ointment to all areas, including the face and scalp once nightly for 2 weeks, with a follow-up appointment at the end of that time. “If you just give people medicine and ask them to come back in 6 months, they are not able to comply with that and they don’t have faith that it’s going to work,” explained Dr. Kirkorian, associate professor of dermatology and pediatrics at George Washington University, Washington. At the end of 2 weeks, “the majority will have improved dramatically, and then you can implement maintenance therapy with topical calcineurin inhibitors, crisaborole, or possibly topical ruxolitinib.”
Some clinicians prescribe oral antihistamines for AD, but Dr. Kirkorian said that data supporting their use are limited and antihistamines are not approved for use in children younger than 6 months of age. Sedating antihistamines will induce sleep, “but do not provide durable night-long sleep,” and routine use may have an impact on learning and school performance. In addition, exposure to antihistamines in children under age 2 may be associated with development of ADHD at school age.
The interleukin-4 receptor alpha antagonist dupilumab (Dupixent) is approved by the Food and Drug Administration for moderate to severe AD in patients ages 6 and older. But obtaining it for patients can be tricky, she said, as this requires documented failure of corticosteroids, calcineurin inhibitors, crisaborole ointment, and phototherapy (if prescribed). Patients are often obligated to do step therapy with an off-label drug such as cyclosporine or methotrexate for 3 months, and they need to demonstrate responses with objective measures of severity such as the SCORAD (SCORing Atopic Dermatitis) and the validated Investigator Global Assessment.
“Most of my patients carry insurance that does not approve dupilumab without failure of a prior off-label systemic immunosuppressant medication,” Dr. Kirkorian said. Cyclosporine is her first choice for a systemic immunosuppressant “because it has a fast onset of action, it’s effective for treatment of atopic dermatitis, and safe for short-term use,” she said. “I don’t think that methotrexate works well for eczema. It can take weeks and weeks to work.”
She typically starts patients on a 5 mg/kg dose of cyclosporine. Baseline tests include CBC, CMP (comprehensive metabolic panel), lipids, and vitals. She repeats the labs at 1 month, and includes a blood pressure check. Potential adverse effects of cyclosporine include infections (including opportunistic infections), cytopenias, hypertension, nephrotoxicity, hepatotoxicity, neurotoxicity (including posterior reversible encephalopathy syndrome), electrolyte disturbance, lymphoma, and cutaneous malignancy.
“The good news is that we generally don’t see the adverse effects with short-term use,” Dr. Kirkorian said. “We will see some hypertrichosis and gingival hypertrophy, which resolves with cessation of therapy. There are serious side effects if you use it for long enough.”
As for methotrexate, “it is still a very important drug in pediatric dermatology, particularly in other conditions such as psoriasis,” she said. “The problem is that weekly dosing of methotrexate poses a greater risk of dosing errors. People aren’t really triggered to think of a once-weekly medication. If you do use it, give them a short supply to make sure that they come back, and that they don’t give it daily accidentally.”
Practical tips she offered for prescribing cyclosporine include supplying a patient handout with information on all adverse effects, dosing information, vaccination information, and pregnancy precautions, with contact information (a patient portal or on-call number) for the treating clinician in case a patient develops adverse effects. Administration of live vaccines while patients are on cyclosporine is not recommended.
When transitioning patients from cyclosporine or methotrexate to dupilumab, Dr. Kirkorian recommends tapering the immunosuppressant dose by half every 2 weeks to complete cessation by week 8 of treatment. For patients who experience a severe baseline flare once the immunosuppressant is tapered, despite the switch to dupilumab, she recommends restarting methotrexate at a full dose and then reducing the dose every 2 weeks until the lowest effective dose (2.5-5 mg weekly) is reached.
“Waning efficacy is real,” she said. “We can add methotrexate to recapture efficacy. Check for superimposed allergic contact dermatitis.”
With upadacitinib (Rinvoq), an oral Janus kinase (JAK) inhibitor recently approved for treating refractory, moderate to severe AD in patients 12 years of age and older, is the risk profile acceptable to parents and physicians? “I think the answer is yes,” Dr. Kirkorian said. “But we’re going to have to think through that very carefully. It’s going to be exciting to see how this drug changes management in our patients.”
Dr. Kirkorian disclosed that she is a member of the advisory board for Verrica Pharmaceuticals.
“Many patients who have failed topical steroids have never had adequate treatment,” Anna Yasmine Kirkorian, MD, chief of dermatology at National Children’s Hospital in Washington, said during the ODAC Dermatology, Aesthetic & Surgical Conference. “There is no lower age limit on the use of topical corticosteroids, and low potency corticosteroids are inadequate to treat severe eczema. The idea that only over-the-counter 2.5% hydrocortisone cream is necessary is not true,” she added.
“You also want to scrutinize the vehicle,” she said, noting that children are often prescribed cream formulations that hurt when applied, so parents stop applying them. “Ointments are generally the vehicles of choice in childhood,” she added.
It is generally not advised to use topical and oral antibiotics in children with AD, unless there are clear signs of infection. “If they’re just slightly oozy, don’t use them,” she continued. “Of course, every child or adult with eczema has Staph aureus on them, but most of the time, what you need to do is repair the barrier. We know that from data and common sense. When we repair their barrier, their rates of infection decrease.”
A focal area with pustules and pus should be cultured and treated, Dr. Kirkorian said. “Monotherapy with antibiotics is going to do nothing for you.” In cases of children with failure to thrive, she recommends referral to pediatric dermatology, allergy/immunology, GI, or genetics, as appropriate.
For children with severe AD, Dr. Kirkorian favors a rescue plan with a one-pound jar of triamcinolone ointment 0.1%. She recommends application of the ointment to all areas, including the face and scalp once nightly for 2 weeks, with a follow-up appointment at the end of that time. “If you just give people medicine and ask them to come back in 6 months, they are not able to comply with that and they don’t have faith that it’s going to work,” explained Dr. Kirkorian, associate professor of dermatology and pediatrics at George Washington University, Washington. At the end of 2 weeks, “the majority will have improved dramatically, and then you can implement maintenance therapy with topical calcineurin inhibitors, crisaborole, or possibly topical ruxolitinib.”
Some clinicians prescribe oral antihistamines for AD, but Dr. Kirkorian said that data supporting their use are limited and antihistamines are not approved for use in children younger than 6 months of age. Sedating antihistamines will induce sleep, “but do not provide durable night-long sleep,” and routine use may have an impact on learning and school performance. In addition, exposure to antihistamines in children under age 2 may be associated with development of ADHD at school age.
The interleukin-4 receptor alpha antagonist dupilumab (Dupixent) is approved by the Food and Drug Administration for moderate to severe AD in patients ages 6 and older. But obtaining it for patients can be tricky, she said, as this requires documented failure of corticosteroids, calcineurin inhibitors, crisaborole ointment, and phototherapy (if prescribed). Patients are often obligated to do step therapy with an off-label drug such as cyclosporine or methotrexate for 3 months, and they need to demonstrate responses with objective measures of severity such as the SCORAD (SCORing Atopic Dermatitis) and the validated Investigator Global Assessment.
“Most of my patients carry insurance that does not approve dupilumab without failure of a prior off-label systemic immunosuppressant medication,” Dr. Kirkorian said. Cyclosporine is her first choice for a systemic immunosuppressant “because it has a fast onset of action, it’s effective for treatment of atopic dermatitis, and safe for short-term use,” she said. “I don’t think that methotrexate works well for eczema. It can take weeks and weeks to work.”
She typically starts patients on a 5 mg/kg dose of cyclosporine. Baseline tests include CBC, CMP (comprehensive metabolic panel), lipids, and vitals. She repeats the labs at 1 month, and includes a blood pressure check. Potential adverse effects of cyclosporine include infections (including opportunistic infections), cytopenias, hypertension, nephrotoxicity, hepatotoxicity, neurotoxicity (including posterior reversible encephalopathy syndrome), electrolyte disturbance, lymphoma, and cutaneous malignancy.
“The good news is that we generally don’t see the adverse effects with short-term use,” Dr. Kirkorian said. “We will see some hypertrichosis and gingival hypertrophy, which resolves with cessation of therapy. There are serious side effects if you use it for long enough.”
As for methotrexate, “it is still a very important drug in pediatric dermatology, particularly in other conditions such as psoriasis,” she said. “The problem is that weekly dosing of methotrexate poses a greater risk of dosing errors. People aren’t really triggered to think of a once-weekly medication. If you do use it, give them a short supply to make sure that they come back, and that they don’t give it daily accidentally.”
Practical tips she offered for prescribing cyclosporine include supplying a patient handout with information on all adverse effects, dosing information, vaccination information, and pregnancy precautions, with contact information (a patient portal or on-call number) for the treating clinician in case a patient develops adverse effects. Administration of live vaccines while patients are on cyclosporine is not recommended.
When transitioning patients from cyclosporine or methotrexate to dupilumab, Dr. Kirkorian recommends tapering the immunosuppressant dose by half every 2 weeks to complete cessation by week 8 of treatment. For patients who experience a severe baseline flare once the immunosuppressant is tapered, despite the switch to dupilumab, she recommends restarting methotrexate at a full dose and then reducing the dose every 2 weeks until the lowest effective dose (2.5-5 mg weekly) is reached.
“Waning efficacy is real,” she said. “We can add methotrexate to recapture efficacy. Check for superimposed allergic contact dermatitis.”
With upadacitinib (Rinvoq), an oral Janus kinase (JAK) inhibitor recently approved for treating refractory, moderate to severe AD in patients 12 years of age and older, is the risk profile acceptable to parents and physicians? “I think the answer is yes,” Dr. Kirkorian said. “But we’re going to have to think through that very carefully. It’s going to be exciting to see how this drug changes management in our patients.”
Dr. Kirkorian disclosed that she is a member of the advisory board for Verrica Pharmaceuticals.
“Many patients who have failed topical steroids have never had adequate treatment,” Anna Yasmine Kirkorian, MD, chief of dermatology at National Children’s Hospital in Washington, said during the ODAC Dermatology, Aesthetic & Surgical Conference. “There is no lower age limit on the use of topical corticosteroids, and low potency corticosteroids are inadequate to treat severe eczema. The idea that only over-the-counter 2.5% hydrocortisone cream is necessary is not true,” she added.
“You also want to scrutinize the vehicle,” she said, noting that children are often prescribed cream formulations that hurt when applied, so parents stop applying them. “Ointments are generally the vehicles of choice in childhood,” she added.
It is generally not advised to use topical and oral antibiotics in children with AD, unless there are clear signs of infection. “If they’re just slightly oozy, don’t use them,” she continued. “Of course, every child or adult with eczema has Staph aureus on them, but most of the time, what you need to do is repair the barrier. We know that from data and common sense. When we repair their barrier, their rates of infection decrease.”
A focal area with pustules and pus should be cultured and treated, Dr. Kirkorian said. “Monotherapy with antibiotics is going to do nothing for you.” In cases of children with failure to thrive, she recommends referral to pediatric dermatology, allergy/immunology, GI, or genetics, as appropriate.
For children with severe AD, Dr. Kirkorian favors a rescue plan with a one-pound jar of triamcinolone ointment 0.1%. She recommends application of the ointment to all areas, including the face and scalp once nightly for 2 weeks, with a follow-up appointment at the end of that time. “If you just give people medicine and ask them to come back in 6 months, they are not able to comply with that and they don’t have faith that it’s going to work,” explained Dr. Kirkorian, associate professor of dermatology and pediatrics at George Washington University, Washington. At the end of 2 weeks, “the majority will have improved dramatically, and then you can implement maintenance therapy with topical calcineurin inhibitors, crisaborole, or possibly topical ruxolitinib.”
Some clinicians prescribe oral antihistamines for AD, but Dr. Kirkorian said that data supporting their use are limited and antihistamines are not approved for use in children younger than 6 months of age. Sedating antihistamines will induce sleep, “but do not provide durable night-long sleep,” and routine use may have an impact on learning and school performance. In addition, exposure to antihistamines in children under age 2 may be associated with development of ADHD at school age.
The interleukin-4 receptor alpha antagonist dupilumab (Dupixent) is approved by the Food and Drug Administration for moderate to severe AD in patients ages 6 and older. But obtaining it for patients can be tricky, she said, as this requires documented failure of corticosteroids, calcineurin inhibitors, crisaborole ointment, and phototherapy (if prescribed). Patients are often obligated to do step therapy with an off-label drug such as cyclosporine or methotrexate for 3 months, and they need to demonstrate responses with objective measures of severity such as the SCORAD (SCORing Atopic Dermatitis) and the validated Investigator Global Assessment.
“Most of my patients carry insurance that does not approve dupilumab without failure of a prior off-label systemic immunosuppressant medication,” Dr. Kirkorian said. Cyclosporine is her first choice for a systemic immunosuppressant “because it has a fast onset of action, it’s effective for treatment of atopic dermatitis, and safe for short-term use,” she said. “I don’t think that methotrexate works well for eczema. It can take weeks and weeks to work.”
She typically starts patients on a 5 mg/kg dose of cyclosporine. Baseline tests include CBC, CMP (comprehensive metabolic panel), lipids, and vitals. She repeats the labs at 1 month, and includes a blood pressure check. Potential adverse effects of cyclosporine include infections (including opportunistic infections), cytopenias, hypertension, nephrotoxicity, hepatotoxicity, neurotoxicity (including posterior reversible encephalopathy syndrome), electrolyte disturbance, lymphoma, and cutaneous malignancy.
“The good news is that we generally don’t see the adverse effects with short-term use,” Dr. Kirkorian said. “We will see some hypertrichosis and gingival hypertrophy, which resolves with cessation of therapy. There are serious side effects if you use it for long enough.”
As for methotrexate, “it is still a very important drug in pediatric dermatology, particularly in other conditions such as psoriasis,” she said. “The problem is that weekly dosing of methotrexate poses a greater risk of dosing errors. People aren’t really triggered to think of a once-weekly medication. If you do use it, give them a short supply to make sure that they come back, and that they don’t give it daily accidentally.”
Practical tips she offered for prescribing cyclosporine include supplying a patient handout with information on all adverse effects, dosing information, vaccination information, and pregnancy precautions, with contact information (a patient portal or on-call number) for the treating clinician in case a patient develops adverse effects. Administration of live vaccines while patients are on cyclosporine is not recommended.
When transitioning patients from cyclosporine or methotrexate to dupilumab, Dr. Kirkorian recommends tapering the immunosuppressant dose by half every 2 weeks to complete cessation by week 8 of treatment. For patients who experience a severe baseline flare once the immunosuppressant is tapered, despite the switch to dupilumab, she recommends restarting methotrexate at a full dose and then reducing the dose every 2 weeks until the lowest effective dose (2.5-5 mg weekly) is reached.
“Waning efficacy is real,” she said. “We can add methotrexate to recapture efficacy. Check for superimposed allergic contact dermatitis.”
With upadacitinib (Rinvoq), an oral Janus kinase (JAK) inhibitor recently approved for treating refractory, moderate to severe AD in patients 12 years of age and older, is the risk profile acceptable to parents and physicians? “I think the answer is yes,” Dr. Kirkorian said. “But we’re going to have to think through that very carefully. It’s going to be exciting to see how this drug changes management in our patients.”
Dr. Kirkorian disclosed that she is a member of the advisory board for Verrica Pharmaceuticals.
FROM ODAC 2022
Questions about optimal dosing of isotretinoin persist, expert says
Although the to this day, according to Diane M. Thiboutot, MD.
These include, what is the ideal daily dose of isotretinoin? What is the ideal cumulative dose of isotretinoin to minimize relapse of acne? What is the ideal duration of isotretinoin therapy? How do you define relapse?
“Initially, it was recommended as 1–mg/kg per day dosing,” Dr. Thiboutot, professor of dermatology at Penn State University, Hershey, said during the Orlando Dermatology Aesthetic and Clinical Conference. “As time went on and we became familiar with flares that some patients can have on that dose, it was recommended to start treatment at a dose of 0.5 mg/kg per day. Then there were trends for low dosing, intermittent dosing, and some of the more recent medical literature is talking about high dosing.”
Clinical studies
A multicenter study of 150 patients published in 1984 found that rates of relapse (retreatment needed) were 42% of patients of patients treated with 0.1 mg/kg daily, 20% in patients treated with 0.5 mg/kg daily, and 10% in patients treated with 1.0 mg/kg daily.
In a later study, researchers who followed 299 patients for 5 years post isotretinoin treatment found that there were more relapses in the 0.5-mg/kg group versus those treated with 1.0 mg/kg. Factors that contributed to the need for more treatment courses included lower-dose regimens (0.1 mg/kg and 0.5 mg/kg), having severe acne, being a female over the age of 25 at the onset of therapy, and having a prolonged history of acne.
More recently, investigators who conducted a single-center study of 1,453 patients treated with isotretinoin defined relapse as the need for a second course of isotretinoin. “They found that neither daily nor cumulative dosages influenced relapse of acne vulgaris in patients treated with varying doses of isotretinoin as long as treatment was continued for 2 or more months after the acne had completely resolved,” said Dr. Thiboutot, a past president of the American Acne and Rosacea Society.
“The current evidence underpinning the 120-150 mg/kg cumulative threshold–dosing regimen is equivocal and is based on two low-grade studies,” she noted. “Cumulative doses required for clearance appear lower for acne of mild to moderate severity and higher for more severe acne. Future investigations should use clinically relevant endpoints as end of treatment criteria and define treatment success in acne accurately.”
Other studies have looked at whether higher doses of isotretinoin could reduce treatment failures in patients with acne, Dr. Thiboutot said. In a retrospective chart review of 102 patients with acne who had been treated with isotretinoin for at least 4 consecutive months and followed for over a year, 45.1% required further treatment and 15.7% received a second course of isotretinoin. Cumulative dose (mg/kg), follow-up period, duration of treatment, and daily dose during the last month of treatment were not significantly different between those who relapsed and those who did not relapse.
However, “while the cumulative dose of isotretinoin did not significantly impact acne relapse, patients who received a higher cumulative dose were less likely to require a second course of treatment,” the authors wrote, adding that “female patients had a higher risk of needing retrial regardless of their cumulative dose.” They commented that “prescribing a higher dose per weight may result in less severe acne recurrences and the need for further isotretinoin therapy.”
Another study evaluated 116 patients who were treated to clearance with dosing at the discretion of the provider and defined relapse as subsequent treatment with an oral or topical agent. In the lower-dose treatment (less than 220 mg/kg; mean of 170 mg/kg) group, the relapse rate was 47.4%, compared with 26.9% in the higher-dose (greater than 220 mg/kg) group (P = .03). Cheilitis and xerosis during treatment was reported in nearly all patients in both treatment groups, but retinoid dermatitis was significantly more common among those in the higher-dose group (53.8% vs. 31.6%; P = .02). There were no significant differences in other adverse events between the two groups.
According to Dr. Thiboutot, variables to consider in selection of a daily dose include the presence of intense inflammation, cysts, nodules, potential difference in side-effect profiles with ethnicity, and an individual’s degree of side effects and their level of comfort. “What are some of the concerns with higher doses? Those of us who have treated a lot of acne patients have had the unfortunate occurrence where you start someone on isotretinoin and their acne explodes, as do their cysts and nodules. Once that happens it’s a bad situation and it takes you a while to get past it.”
Acne fulminans
Dr. Thiboutot was part of a panel of experts who assembled guidelines on the classification, management, and prevention of acne fulminans, published in 2017. “Acne fulminans can be induced by isotretinoin or it can occur on its own. If you have someone who has a lot of inflammation, a lot of cysts and nodules, it’s probably best to start them on a lower dose with or without prednisone,” she said.
Age at treatment is another variable that can affect the duration of response to isotretinoin. “If the patient is very young when they need isotretinoin, it’s highly likely that they will need it again,” Dr. Thiboutot said. “Also, adult females often need a repeat dose of isotretinoin. The cumulative dose is important, and the presence of truncal acne is a factor affecting duration of response. Truncal acne takes longer to clear, and it oftentimes needs a longer treatment course.”
Data from the iPledge database indicate that 37% of 10- to 11-year-olds needed a repeat course of isotretinoin, “and as you get older, the need for retreatment is less,” she said.
High-dose versus low-dose isotretinoin
A slow, low-dose approach to the use of isotretinoin in practice can minimize side effects and cystic flares, according to Dr. Thiboutot. “The cons are that you could have a longer treatment duration, you might need more patient visits, and it creates more prolonged drug exposure in patients who can become pregnant,” she said.
“The pros of a high-dose strategy include a shorter treatment duration and potential costs savings. The cons are that there is an increased risk of side effects and a risk of cystic flare. It seems that the general agreement is to treat until clearance and then treat for another 2 months. A clear definition of acne relapse is needed as well as prospective studies to optimize dosing.”
Dr. Thiboutot disclosed that she is a consultant Galderma and Novartis. She has also performed clinical trials for Galderma, Cassiopea, and Foamix.
Commentary by Robert Sidbury, MD, MPH
Isotretinoin has been around for a long time – 40-plus years, to be exact. Despite this cumulative experience, ideal dosing and duration remain uncertain. Many providers have evolved from a “one size fits all” approach (for example, 1 mg/kg of body weight per day for 4-6 months) to a more tailored and nuanced strategy. Dr. Thiboutot validates the need to individualize therapy and helps identify patients at higher risk for relapse. Younger patients are at greater risk for repeat courses of isotretinoin – Dr. Thiboutot cites a study showing 37% of 10- to 11-year-olds required a second course of isotretinoin. This could simply be a proxy for severity, which also augurs a more protracted course. Adult women and those with truncal acne also trend toward longer courses. Knowing these risk factors will help practitioners counsel proper expectations, tailor treatment courses, and thread the always challenging isotretinoin needle: Should one dose “low and slow” and court a longer risk window, or higher and faster, sometimes leading to a dryer disaster?
Dr. Sidbury is chief of dermatology at Seattle Children's Hospital and professor, department of pediatrics, University of Washington, Seattle. He is a site principal investigator for dupilumab trials, for which the hospital has a contract with Regeneron.
This article was updated 6/10/22.
Although the to this day, according to Diane M. Thiboutot, MD.
These include, what is the ideal daily dose of isotretinoin? What is the ideal cumulative dose of isotretinoin to minimize relapse of acne? What is the ideal duration of isotretinoin therapy? How do you define relapse?
“Initially, it was recommended as 1–mg/kg per day dosing,” Dr. Thiboutot, professor of dermatology at Penn State University, Hershey, said during the Orlando Dermatology Aesthetic and Clinical Conference. “As time went on and we became familiar with flares that some patients can have on that dose, it was recommended to start treatment at a dose of 0.5 mg/kg per day. Then there were trends for low dosing, intermittent dosing, and some of the more recent medical literature is talking about high dosing.”
Clinical studies
A multicenter study of 150 patients published in 1984 found that rates of relapse (retreatment needed) were 42% of patients of patients treated with 0.1 mg/kg daily, 20% in patients treated with 0.5 mg/kg daily, and 10% in patients treated with 1.0 mg/kg daily.
In a later study, researchers who followed 299 patients for 5 years post isotretinoin treatment found that there were more relapses in the 0.5-mg/kg group versus those treated with 1.0 mg/kg. Factors that contributed to the need for more treatment courses included lower-dose regimens (0.1 mg/kg and 0.5 mg/kg), having severe acne, being a female over the age of 25 at the onset of therapy, and having a prolonged history of acne.
More recently, investigators who conducted a single-center study of 1,453 patients treated with isotretinoin defined relapse as the need for a second course of isotretinoin. “They found that neither daily nor cumulative dosages influenced relapse of acne vulgaris in patients treated with varying doses of isotretinoin as long as treatment was continued for 2 or more months after the acne had completely resolved,” said Dr. Thiboutot, a past president of the American Acne and Rosacea Society.
“The current evidence underpinning the 120-150 mg/kg cumulative threshold–dosing regimen is equivocal and is based on two low-grade studies,” she noted. “Cumulative doses required for clearance appear lower for acne of mild to moderate severity and higher for more severe acne. Future investigations should use clinically relevant endpoints as end of treatment criteria and define treatment success in acne accurately.”
Other studies have looked at whether higher doses of isotretinoin could reduce treatment failures in patients with acne, Dr. Thiboutot said. In a retrospective chart review of 102 patients with acne who had been treated with isotretinoin for at least 4 consecutive months and followed for over a year, 45.1% required further treatment and 15.7% received a second course of isotretinoin. Cumulative dose (mg/kg), follow-up period, duration of treatment, and daily dose during the last month of treatment were not significantly different between those who relapsed and those who did not relapse.
However, “while the cumulative dose of isotretinoin did not significantly impact acne relapse, patients who received a higher cumulative dose were less likely to require a second course of treatment,” the authors wrote, adding that “female patients had a higher risk of needing retrial regardless of their cumulative dose.” They commented that “prescribing a higher dose per weight may result in less severe acne recurrences and the need for further isotretinoin therapy.”
Another study evaluated 116 patients who were treated to clearance with dosing at the discretion of the provider and defined relapse as subsequent treatment with an oral or topical agent. In the lower-dose treatment (less than 220 mg/kg; mean of 170 mg/kg) group, the relapse rate was 47.4%, compared with 26.9% in the higher-dose (greater than 220 mg/kg) group (P = .03). Cheilitis and xerosis during treatment was reported in nearly all patients in both treatment groups, but retinoid dermatitis was significantly more common among those in the higher-dose group (53.8% vs. 31.6%; P = .02). There were no significant differences in other adverse events between the two groups.
According to Dr. Thiboutot, variables to consider in selection of a daily dose include the presence of intense inflammation, cysts, nodules, potential difference in side-effect profiles with ethnicity, and an individual’s degree of side effects and their level of comfort. “What are some of the concerns with higher doses? Those of us who have treated a lot of acne patients have had the unfortunate occurrence where you start someone on isotretinoin and their acne explodes, as do their cysts and nodules. Once that happens it’s a bad situation and it takes you a while to get past it.”
Acne fulminans
Dr. Thiboutot was part of a panel of experts who assembled guidelines on the classification, management, and prevention of acne fulminans, published in 2017. “Acne fulminans can be induced by isotretinoin or it can occur on its own. If you have someone who has a lot of inflammation, a lot of cysts and nodules, it’s probably best to start them on a lower dose with or without prednisone,” she said.
Age at treatment is another variable that can affect the duration of response to isotretinoin. “If the patient is very young when they need isotretinoin, it’s highly likely that they will need it again,” Dr. Thiboutot said. “Also, adult females often need a repeat dose of isotretinoin. The cumulative dose is important, and the presence of truncal acne is a factor affecting duration of response. Truncal acne takes longer to clear, and it oftentimes needs a longer treatment course.”
Data from the iPledge database indicate that 37% of 10- to 11-year-olds needed a repeat course of isotretinoin, “and as you get older, the need for retreatment is less,” she said.
High-dose versus low-dose isotretinoin
A slow, low-dose approach to the use of isotretinoin in practice can minimize side effects and cystic flares, according to Dr. Thiboutot. “The cons are that you could have a longer treatment duration, you might need more patient visits, and it creates more prolonged drug exposure in patients who can become pregnant,” she said.
“The pros of a high-dose strategy include a shorter treatment duration and potential costs savings. The cons are that there is an increased risk of side effects and a risk of cystic flare. It seems that the general agreement is to treat until clearance and then treat for another 2 months. A clear definition of acne relapse is needed as well as prospective studies to optimize dosing.”
Dr. Thiboutot disclosed that she is a consultant Galderma and Novartis. She has also performed clinical trials for Galderma, Cassiopea, and Foamix.
Commentary by Robert Sidbury, MD, MPH
Isotretinoin has been around for a long time – 40-plus years, to be exact. Despite this cumulative experience, ideal dosing and duration remain uncertain. Many providers have evolved from a “one size fits all” approach (for example, 1 mg/kg of body weight per day for 4-6 months) to a more tailored and nuanced strategy. Dr. Thiboutot validates the need to individualize therapy and helps identify patients at higher risk for relapse. Younger patients are at greater risk for repeat courses of isotretinoin – Dr. Thiboutot cites a study showing 37% of 10- to 11-year-olds required a second course of isotretinoin. This could simply be a proxy for severity, which also augurs a more protracted course. Adult women and those with truncal acne also trend toward longer courses. Knowing these risk factors will help practitioners counsel proper expectations, tailor treatment courses, and thread the always challenging isotretinoin needle: Should one dose “low and slow” and court a longer risk window, or higher and faster, sometimes leading to a dryer disaster?
Dr. Sidbury is chief of dermatology at Seattle Children's Hospital and professor, department of pediatrics, University of Washington, Seattle. He is a site principal investigator for dupilumab trials, for which the hospital has a contract with Regeneron.
This article was updated 6/10/22.
Although the to this day, according to Diane M. Thiboutot, MD.
These include, what is the ideal daily dose of isotretinoin? What is the ideal cumulative dose of isotretinoin to minimize relapse of acne? What is the ideal duration of isotretinoin therapy? How do you define relapse?
“Initially, it was recommended as 1–mg/kg per day dosing,” Dr. Thiboutot, professor of dermatology at Penn State University, Hershey, said during the Orlando Dermatology Aesthetic and Clinical Conference. “As time went on and we became familiar with flares that some patients can have on that dose, it was recommended to start treatment at a dose of 0.5 mg/kg per day. Then there were trends for low dosing, intermittent dosing, and some of the more recent medical literature is talking about high dosing.”
Clinical studies
A multicenter study of 150 patients published in 1984 found that rates of relapse (retreatment needed) were 42% of patients of patients treated with 0.1 mg/kg daily, 20% in patients treated with 0.5 mg/kg daily, and 10% in patients treated with 1.0 mg/kg daily.
In a later study, researchers who followed 299 patients for 5 years post isotretinoin treatment found that there were more relapses in the 0.5-mg/kg group versus those treated with 1.0 mg/kg. Factors that contributed to the need for more treatment courses included lower-dose regimens (0.1 mg/kg and 0.5 mg/kg), having severe acne, being a female over the age of 25 at the onset of therapy, and having a prolonged history of acne.
More recently, investigators who conducted a single-center study of 1,453 patients treated with isotretinoin defined relapse as the need for a second course of isotretinoin. “They found that neither daily nor cumulative dosages influenced relapse of acne vulgaris in patients treated with varying doses of isotretinoin as long as treatment was continued for 2 or more months after the acne had completely resolved,” said Dr. Thiboutot, a past president of the American Acne and Rosacea Society.
“The current evidence underpinning the 120-150 mg/kg cumulative threshold–dosing regimen is equivocal and is based on two low-grade studies,” she noted. “Cumulative doses required for clearance appear lower for acne of mild to moderate severity and higher for more severe acne. Future investigations should use clinically relevant endpoints as end of treatment criteria and define treatment success in acne accurately.”
Other studies have looked at whether higher doses of isotretinoin could reduce treatment failures in patients with acne, Dr. Thiboutot said. In a retrospective chart review of 102 patients with acne who had been treated with isotretinoin for at least 4 consecutive months and followed for over a year, 45.1% required further treatment and 15.7% received a second course of isotretinoin. Cumulative dose (mg/kg), follow-up period, duration of treatment, and daily dose during the last month of treatment were not significantly different between those who relapsed and those who did not relapse.
However, “while the cumulative dose of isotretinoin did not significantly impact acne relapse, patients who received a higher cumulative dose were less likely to require a second course of treatment,” the authors wrote, adding that “female patients had a higher risk of needing retrial regardless of their cumulative dose.” They commented that “prescribing a higher dose per weight may result in less severe acne recurrences and the need for further isotretinoin therapy.”
Another study evaluated 116 patients who were treated to clearance with dosing at the discretion of the provider and defined relapse as subsequent treatment with an oral or topical agent. In the lower-dose treatment (less than 220 mg/kg; mean of 170 mg/kg) group, the relapse rate was 47.4%, compared with 26.9% in the higher-dose (greater than 220 mg/kg) group (P = .03). Cheilitis and xerosis during treatment was reported in nearly all patients in both treatment groups, but retinoid dermatitis was significantly more common among those in the higher-dose group (53.8% vs. 31.6%; P = .02). There were no significant differences in other adverse events between the two groups.
According to Dr. Thiboutot, variables to consider in selection of a daily dose include the presence of intense inflammation, cysts, nodules, potential difference in side-effect profiles with ethnicity, and an individual’s degree of side effects and their level of comfort. “What are some of the concerns with higher doses? Those of us who have treated a lot of acne patients have had the unfortunate occurrence where you start someone on isotretinoin and their acne explodes, as do their cysts and nodules. Once that happens it’s a bad situation and it takes you a while to get past it.”
Acne fulminans
Dr. Thiboutot was part of a panel of experts who assembled guidelines on the classification, management, and prevention of acne fulminans, published in 2017. “Acne fulminans can be induced by isotretinoin or it can occur on its own. If you have someone who has a lot of inflammation, a lot of cysts and nodules, it’s probably best to start them on a lower dose with or without prednisone,” she said.
Age at treatment is another variable that can affect the duration of response to isotretinoin. “If the patient is very young when they need isotretinoin, it’s highly likely that they will need it again,” Dr. Thiboutot said. “Also, adult females often need a repeat dose of isotretinoin. The cumulative dose is important, and the presence of truncal acne is a factor affecting duration of response. Truncal acne takes longer to clear, and it oftentimes needs a longer treatment course.”
Data from the iPledge database indicate that 37% of 10- to 11-year-olds needed a repeat course of isotretinoin, “and as you get older, the need for retreatment is less,” she said.
High-dose versus low-dose isotretinoin
A slow, low-dose approach to the use of isotretinoin in practice can minimize side effects and cystic flares, according to Dr. Thiboutot. “The cons are that you could have a longer treatment duration, you might need more patient visits, and it creates more prolonged drug exposure in patients who can become pregnant,” she said.
“The pros of a high-dose strategy include a shorter treatment duration and potential costs savings. The cons are that there is an increased risk of side effects and a risk of cystic flare. It seems that the general agreement is to treat until clearance and then treat for another 2 months. A clear definition of acne relapse is needed as well as prospective studies to optimize dosing.”
Dr. Thiboutot disclosed that she is a consultant Galderma and Novartis. She has also performed clinical trials for Galderma, Cassiopea, and Foamix.
Commentary by Robert Sidbury, MD, MPH
Isotretinoin has been around for a long time – 40-plus years, to be exact. Despite this cumulative experience, ideal dosing and duration remain uncertain. Many providers have evolved from a “one size fits all” approach (for example, 1 mg/kg of body weight per day for 4-6 months) to a more tailored and nuanced strategy. Dr. Thiboutot validates the need to individualize therapy and helps identify patients at higher risk for relapse. Younger patients are at greater risk for repeat courses of isotretinoin – Dr. Thiboutot cites a study showing 37% of 10- to 11-year-olds required a second course of isotretinoin. This could simply be a proxy for severity, which also augurs a more protracted course. Adult women and those with truncal acne also trend toward longer courses. Knowing these risk factors will help practitioners counsel proper expectations, tailor treatment courses, and thread the always challenging isotretinoin needle: Should one dose “low and slow” and court a longer risk window, or higher and faster, sometimes leading to a dryer disaster?
Dr. Sidbury is chief of dermatology at Seattle Children's Hospital and professor, department of pediatrics, University of Washington, Seattle. He is a site principal investigator for dupilumab trials, for which the hospital has a contract with Regeneron.
This article was updated 6/10/22.
FROM ODAC 2022
Hyperpigmented plaque on back
Based on the thickness and size of this irregular lesion, a punch biopsy was performed and confirmed the diagnosis of dermatofibrosarcoma protuberans (DFSP).
DFSPs are usually found on the trunk and proximal extremities; they are most often located on the chest and shoulders. The lesion usually manifests as an asymptomatic, firm, and sometimes nodular plaque that may go undiagnosed for years. DFSP is an uncommon mesenchymal tumor with uncertain etiology. It is thought that prior injury to the affected skin may result in a translocation of chromosomes 17 and 22 in skin cells, as this molecular change characterizes the vast majority of DFSPs.
Black patients are more likely than other ethnic populations to develop DFSP and its variants; there is also a slight female predominance.1 Known variants of DFSP include violaceous plaques with telangiectatic atrophic skin, and plaques with dark brown pigmentation called Bednar tumors.1,2
DFSPs are rarely metastatic, but can be locally invasive, so primary treatment consists of wide local excision or Mohs micrographic surgery (MMS). There is a higher probability for cure when MMS is utilized to treat DFSPs with the added benefit of minimizing surgical margins and preserving healthy surrounding skin. In the rarer cases of advanced local, unresectable, or metastatic disease, inhibitor therapy with imatinib or radiation therapy may be considered.2 Due to the risk of local recurrence, patients with DFSP should have regular clinical follow-up every 6 months for 5 years, followed by annual lifelong surveillance.1
This patient was referred for MMS and has not yet returned for follow-up evaluation.
Photo courtesy of Daniel Stulberg, MD. Text courtesy of Morgan Haynes, BS, University of New Mexico School of Medicine and Daniel Stulberg, MD, FAAFP, Department of Family and Community Medicine, University of New Mexico School of Medicine, Albuquerque.
1. Mendenhall WM, Scarborough MT, Flowers FP. Dermatofibrosarcoma protuberans: epidemiology, pathogenesis, clinical presentation, diagnosis, and staging. UpToDate. Updated March 31, 2021. Accessed February 2, 2022. www.uptodate.com/contents/dermatofibrosarcoma-protuberans-epidemiology-pathogenesis-clinical-presentation-diagnosis-and-staging
2. Brooks J, Ramsey ML. Dermatofibrosarcoma Protuberans. StatPearls. Updated November 14, 2021. Accessed January 27, 2022. www.ncbi.nlm.nih.gov/books/NBK513305

Based on the thickness and size of this irregular lesion, a punch biopsy was performed and confirmed the diagnosis of dermatofibrosarcoma protuberans (DFSP).
DFSPs are usually found on the trunk and proximal extremities; they are most often located on the chest and shoulders. The lesion usually manifests as an asymptomatic, firm, and sometimes nodular plaque that may go undiagnosed for years. DFSP is an uncommon mesenchymal tumor with uncertain etiology. It is thought that prior injury to the affected skin may result in a translocation of chromosomes 17 and 22 in skin cells, as this molecular change characterizes the vast majority of DFSPs.
Black patients are more likely than other ethnic populations to develop DFSP and its variants; there is also a slight female predominance.1 Known variants of DFSP include violaceous plaques with telangiectatic atrophic skin, and plaques with dark brown pigmentation called Bednar tumors.1,2
DFSPs are rarely metastatic, but can be locally invasive, so primary treatment consists of wide local excision or Mohs micrographic surgery (MMS). There is a higher probability for cure when MMS is utilized to treat DFSPs with the added benefit of minimizing surgical margins and preserving healthy surrounding skin. In the rarer cases of advanced local, unresectable, or metastatic disease, inhibitor therapy with imatinib or radiation therapy may be considered.2 Due to the risk of local recurrence, patients with DFSP should have regular clinical follow-up every 6 months for 5 years, followed by annual lifelong surveillance.1
This patient was referred for MMS and has not yet returned for follow-up evaluation.
Photo courtesy of Daniel Stulberg, MD. Text courtesy of Morgan Haynes, BS, University of New Mexico School of Medicine and Daniel Stulberg, MD, FAAFP, Department of Family and Community Medicine, University of New Mexico School of Medicine, Albuquerque.

Based on the thickness and size of this irregular lesion, a punch biopsy was performed and confirmed the diagnosis of dermatofibrosarcoma protuberans (DFSP).
DFSPs are usually found on the trunk and proximal extremities; they are most often located on the chest and shoulders. The lesion usually manifests as an asymptomatic, firm, and sometimes nodular plaque that may go undiagnosed for years. DFSP is an uncommon mesenchymal tumor with uncertain etiology. It is thought that prior injury to the affected skin may result in a translocation of chromosomes 17 and 22 in skin cells, as this molecular change characterizes the vast majority of DFSPs.
Black patients are more likely than other ethnic populations to develop DFSP and its variants; there is also a slight female predominance.1 Known variants of DFSP include violaceous plaques with telangiectatic atrophic skin, and plaques with dark brown pigmentation called Bednar tumors.1,2
DFSPs are rarely metastatic, but can be locally invasive, so primary treatment consists of wide local excision or Mohs micrographic surgery (MMS). There is a higher probability for cure when MMS is utilized to treat DFSPs with the added benefit of minimizing surgical margins and preserving healthy surrounding skin. In the rarer cases of advanced local, unresectable, or metastatic disease, inhibitor therapy with imatinib or radiation therapy may be considered.2 Due to the risk of local recurrence, patients with DFSP should have regular clinical follow-up every 6 months for 5 years, followed by annual lifelong surveillance.1
This patient was referred for MMS and has not yet returned for follow-up evaluation.
Photo courtesy of Daniel Stulberg, MD. Text courtesy of Morgan Haynes, BS, University of New Mexico School of Medicine and Daniel Stulberg, MD, FAAFP, Department of Family and Community Medicine, University of New Mexico School of Medicine, Albuquerque.
1. Mendenhall WM, Scarborough MT, Flowers FP. Dermatofibrosarcoma protuberans: epidemiology, pathogenesis, clinical presentation, diagnosis, and staging. UpToDate. Updated March 31, 2021. Accessed February 2, 2022. www.uptodate.com/contents/dermatofibrosarcoma-protuberans-epidemiology-pathogenesis-clinical-presentation-diagnosis-and-staging
2. Brooks J, Ramsey ML. Dermatofibrosarcoma Protuberans. StatPearls. Updated November 14, 2021. Accessed January 27, 2022. www.ncbi.nlm.nih.gov/books/NBK513305
1. Mendenhall WM, Scarborough MT, Flowers FP. Dermatofibrosarcoma protuberans: epidemiology, pathogenesis, clinical presentation, diagnosis, and staging. UpToDate. Updated March 31, 2021. Accessed February 2, 2022. www.uptodate.com/contents/dermatofibrosarcoma-protuberans-epidemiology-pathogenesis-clinical-presentation-diagnosis-and-staging
2. Brooks J, Ramsey ML. Dermatofibrosarcoma Protuberans. StatPearls. Updated November 14, 2021. Accessed January 27, 2022. www.ncbi.nlm.nih.gov/books/NBK513305