User login
Part 1: Self-care for Diabetes Patients
Diabetes mellitus is prevalent in our society; 1 in 10 Americans has the condition and > 1 in 3 has prediabetes.1 Due to the widespread comorbidities and complications of this disease, the American Diabetes Association (ADA) recommends that diabetes management focus on evaluation and treatment of complications.2 Diabetes-related complications can be life-altering and challenging for patients because their quality of life suffers.
For providers, there are several evidence-based screening tools and preventive practices (in and beyond glycemic control) that reduce diabetes complications such as congestive heart failure, kidney failure, lower extremity amputation, and stroke.3 We as providers can treat patients by implementing appropriate goal-directed therapy.4-6
In this 5-part series, I will explore the evidence and recommendations for a multimodal approach in a patient with type 2 diabetes. Here—in Part 1—I explore the self-care behaviors our patients can adopt to improve their symptoms of diabetes.
Case Report
Mr. W is an overweight 64-year-old man with hypertension, hyperlipidemia, and type 2 diabetes mellitus. He visits the clinic for his yearly physical exam. He is concerned because his father, who had diabetes, developed renal failure and had multiple amputations near the end of his life. He is worried that he might face the same outcomes and asks you what he can do to avoid his father’s fate.
Advising Your Patient on Self-care
The cornerstone of diabetes management is appropriate self-care. Both the ADA and the American Association of Clinical Endocrinologists (AACE) recommend that treatment plans should encourage the patient to adopt healthy lifestyle behaviors, including a healthy diet, regular exercise, weight control, and avoidance of tobacco.2,7,8 These interventions have positive effects on blood pressure, glucose control, and lipid levels. They can also reduce the risk for diabetic complications, including atherosclerotic cardiovascular disease (ASCVD), which is the foremost cause of death among patients with diabetes. During a patient visit, clinicians can suggest the following self-care interventions for improving long-term outcomes.
Education sessions. The ADA recommends that individuals with diabetes participate in diabetes self-management education and support (DSMES) sessions.2 In these sessions, patients with diabetes are instructed on a variety of self-care behaviors, including lifestyle interventions, medication management, self-monitoring, and problem-solving.9 These programs—often paid for in part by health insurance—are taught by health care professionals such as registered dieticians, nutritionists, or certified diabetes educators.9,10 Evidence suggests DSMES increases patients’ sense of self-efficacy and may improve blood sugar management.10 Clinicians can help guide their patients through the Association of Diabetes Care & Education Specialists’ online database to identify a DSMES program near them (see www.diabeteseducator.org/living-with-diabetes/find-an-education-program).11
Diet. The AACE recommends a plant-based diet high in polyunsaturated and monounsaturated fatty acids and limited in trans fatty acids and saturated fats.7 Evidence strongly suggests that a Mediterranean diet with high vegetable intake and decreased saturated fats helps to reduce the risk for major cardiovascular events (myocardial infarction and stroke).12
Continue to: Exercise
Exercise. Both the ADA and AACE recommend that most adults with diabetes engage in at least 150 min/week of moderate-to-vigorous aerobic and strength-training exercises.2,7 Clinicians should evaluate patients with sedentary lifestyles prior to them engaging in vigorous physical activity beyond simple walking.2 The ADA also recommends that patients should avoid sitting for long periods of time by engaging in physical activity at least every 30 minutes.2 For adults who may not be able to participate in moderate-to-vigorous exercise, recommend alternative flexibility and balance-training activities, such as yoga or tai chi, 2 to 3 times per week.2
Weight management—a combined effort of diet, exercise, and behavioral therapy—is pivotal in the management of type 2 diabetes due to the potential benefits in insulin resistance, blood pressure, hyperlipidemia, and other factors.2 Weight loss may also improve glycemic control and reduce the need for glucose-lowering medications.2 For patients who struggle with weight loss, consider prescribing FDA-approved weight-loss medications (phentermine, orlistat, lorcaserin, naltrexone/bupropion, liraglutide) or, in some cases, referring for bariatric surgery.2,7
Sleep hygiene is an important element in any preventive treatment plan. This includes interventions as simple as going to bed at the same time every night, sleeping in a dark room, sleeping for at least 7 hours, and removing electronic devices from the bedroom.13 Patients should avoid alcohol, caffeine, and large meals before bedtime.13
Additionally, obstructive sleep apnea (OSA) is often underdiagnosed in patients with diabetes and contributes to insulin resistance, inflammation, and elevated blood pressure.7,14 For early identification of OSA, order a sleep study when appropriate and refer patients to sleep specialists if needed. Patients who are recommended for treatment should be monitored for increasing compliance with care and to ensure benefit from treatment.
In Part 2, we’ll check in with Mr. W as I discuss the role of blood pressure monitoring and antihypertensive medications in reducing cardiovascular risks in patients with diabetes.
1. Centers for Disease Control and Prevention. Diabetes incidence and prevalence. Diabetes Report Card 2017. www.cdc.gov/diabetes/library/reports/reportcard/incidence-2017.html. Published 2018. Accessed June 18, 2020.
2. Standards of Medical Care in Diabetes—2020 Abridged for Primary Care Providers. American Diabetes Association Clinical Diabetes. 2020;38(1):10-38.
3. Chen Y, Sloan FA, Yashkin AP. Adherence to diabetes guidelines for screening, physical activity and medication and onset of complications and death. J Diabetes Complications. 2015;29(8):1228-1233.
4. Mehta S, Mocarski M, Wisniewski T, et al. Primary care physicians’ utilization of type 2 diabetes screening guidelines and referrals to behavioral interventions: a survey-linked retrospective study. BMJ Open Diabetes Res Care. 2017;5(1):e000406.
5. Center for Disease Control and Prevention. Preventive care practices. Diabetes Report Card 2017. www.cdc.gov/diabetes/library/reports/reportcard/preventive-care.html. Published 2018. Accessed June 18, 2020.
6. Arnold SV, de Lemos JA, Rosenson RS, et al; GOULD Investigators. Use of guideline-recommended risk reduction strategies among patients with diabetes and atherosclerotic cardiovascular disease. Circulation. 2019;140(7):618-620.
7. Garber AJ, Handelsman Y, Grunberger G, et al. Consensus Statement by the American Association of Clinical Endocrinologists and American College of Endocrinology on the comprehensive type 2 diabetes management algorithm—2020 executive summary. Endocr Pract Endocr Pract. 2020;26(1):107-139.
8. American Diabetes Association. Comprehensive medical evaluation and assessment of comorbidities: standards of medical care in diabetes—2020. Diabetes Care. 2020;43(suppl 1):S37-S47.
9. Beck J, Greenwood DA, Blanton L, et al; 2017 Standards Revision Task Force. 2017 National Standards for diabetes self-management education and support. Diabetes Educ. 2017;43(5): 449-464.
10. Chrvala CA, Sherr D, Lipman RD. Diabetes self-management education for adults with type 2 diabetes mellitus: a systematic review of the effect on glycemic control. Patient Educ Couns. 2016;99(6):926-943.
11. Association of Diabetes Care & Education Specialists. Find a diabetes education program in your area. www.diabeteseducator.org/living-with-diabetes/find-an-education-program. Accessed June 15, 2020.
12. Estruch R, Ros E, Salas-Salvadó J, et al; PREDIMED Study Investigators. Primary prevention of cardiovascular disease with a Mediterranean diet supplemented with extra-virgin olive oil or nuts. NEJM. 2018;378(25):e34.
13. Centers for Disease Control and Prevention. Tips for better sleep. Sleep and sleep disorders. www.cdc.gov/sleep/about_sleep/sleep_hygiene.html. Reviewed July 15, 2016. Accessed June 18, 2020.
14. Doumit J, Prasad B. Sleep Apnea in Type 2 Diabetes. Diabetes Spectrum. 2016; 29(1): 14-19.
15. Marso SP, Daniels GH, Brown-Frandsen K, et al; LEADER Steering Committee on behalf of the LEADER Trial Investigators. Liraglutide and cardiovascular outcomes in type 2 diabetes. N Engl J Med. 2016;375:311-322.
16. Perkovic V, Jardine MJ, Neal B, et al; CREDENCE Trial Investigators. Canagliflozin and renal outcomes in type 2 diabetes and nephropathy. N Engl J Med. 2019;380(24):2295-2306.
17. Trends in Blood pressure control and treatment among type 2 diabetes with comorbid hypertension in the United States: 1988-2004. J Hypertens. 2009;27(9):1908-1916.
18. Emdin CA, Rahimi K, Neal B, et al. Blood pressure lowering in type 2 diabetes: a systematic review and meta-analysis. JAMA. 2015;313(6):603-615.
19. Vouri SM, Shaw RF, Waterbury NV, et al. Prevalence of achievement of A1c, blood pressure, and cholesterol (ABC) goal in veterans with diabetes. J Manag Care Pharm. 2011;17(4):304-312.
20. Kudo N, Yokokawa H, Fukuda H, et al. Achievement of target blood pressure levels among Japanese workers with hypertension and healthy lifestyle characteristics associated with therapeutic failure. Plos One. 2015;10(7):e0133641.
21. Carey RM, Whelton PK; 2017 ACC/AHA Hypertension Guideline Writing Committee. Prevention, detection, evaluation, and management of high blood pressure in adults: synopsis of the 2017 American College of Cardiology/American Heart Association Hypertension guideline. Ann Intern Med. 2018;168(5):351-358.
22. Deedwania PC. Blood pressure control in diabetes mellitus. Circulation. 2011;123:2776–2778.
23. Catalá-López F, Saint-Gerons DM, González-Bermejo D, et al. Cardiovascular and renal outcomes of renin-angiotensin system blockade in adult patients with diabetes mellitus: a systematic review with network meta-analyses. PLoS Med. 2016;13(3):e1001971.
24. Furberg CD, Wright JT Jr, Davis BR, et al; ALLHAT Officers and Coordinators for the ALLHAT Collaborative Research Group. Major outcomes in high-risk hypertensive patients randomized to angiotensin-converting enzyme inhibitor or calcium channel blocker vs diuretic: the Antihypertensive and Lipid-Lowering Treatment to Prevent Heart Attack Trial (ALLHAT). JAMA. 2002;288(23):2981-2997.
25. Sleight P. The HOPE Study (Heart Outcomes Prevention Evaluation). J Renin-Angiotensin-Aldosterone Syst. 2000;1(1):18-20.
26. Tatti P, Pahor M, Byington RP, et al. Outcome results of the Fosinopril Versus Amlodipine Cardiovascular Events Randomized Trial (FACET) in patients with hypertension and NIDDM. Diabetes Care. 1998;21(4):597-603.
27. Schrier RW, Estacio RO, Jeffers B. Appropriate Blood Pressure Control in NIDDM (ABCD) Trial. Diabetologia. 1996;39(12):1646-1654.
28. Hansson L, Zanchetti A, Carruthers SG, et al; HOT Study Group. Effects of intensive blood-pressure lowering and low-dose aspirin in patients with hypertension: principal results of the Hypertension Optimal Treatment (HOT) Randomised Trial. Lancet. 1998;351(9118):1755-1762.
29. Baigent C, Blackwell L, Emberson J, et al; Cholesterol Treatment Trialists’ (CTT) Collaboration. Efficacy and safety of more intensive lowering of LDL cholesterol: a meta-analysis of data from 170,000 participants in 26 randomised trials. Lancet. 2010;376(9753):1670-1681.
30. Fu AZ, Zhang Q, Davies MJ, et al. Underutilization of statins in patients with type 2 diabetes in US clinical practice: a retrospective cohort study. Curr Med Res Opin. 2011;27(5):1035-1040.
31. Cannon CP, Blazing MA, Giugliano RP, et al; IMPROVE-IT Investigators. Ezetimibe added to statin therapy after acute coronary syndromes. N Engl J Med. 2015; 372:2387-2397
32. Sabatine MS, Giugliano RP, Keech AC, et al; the FOURIER Steering Committee and Investigators. Evolocumab and clinical outcomes in patients with cardiovascular disease. N Engl J Med. 2017;376:1713-1722.
33. Schwartz GG, Steg PG, Szarek M, et al; ODYSSEY OUTCOMES Committees and Investigators. Alirocumab and Cardiovascular Outcomes after Acute Coronary Syndrome | NEJM. N Engl J Med. 2018;379:2097-2107.
34. Icosapent ethyl [package insert]. Bridgewater, NJ: Amarin Pharma, Inc.; 2019.
35. Bhatt DL, Steg PG, Miller M, et al; REDUCE-IT Investigators. Cardiovascular risk reduction with icosapent ethyl for hypertriglyceridemia. N Engl J Med. 2019;380:11-22
36. Bolton WK. Renal Physicians Association Clinical practice guideline: appropriate patient preparation for renal replacement therapy: guideline number 3. J Am Soc Nephrol. 2003;14(5):1406-1410.
37. American Diabetes Association. Pharmacologic Approaches to glycemic treatment: standards of medical care in diabetes—2020. Diabetes Care. 2020;43(suppl 1):S98-S110.
38. Qaseem A, Barry MJ, Humphrey LL, Forciea MA; Clinical Guidelines Committee of the American College of Physicians. Oral pharmacologic treatment of type 2 diabetes mellitus: a clinical practice guideline update from the American College of Physicians. Ann Intern Med. 2017;166(4):279-290.
39. Kidney Disease: Improving Global Outcomes (KDIGO) CKD-MBD Update Work Group. KDIGO 2017 Clinical Practice Guideline Update for the diagnosis, evaluation, prevention, and treatment of chronic kidney disease–mineral and bone disorder (CKD-MBD). Kidney Int Suppl (2011). 2017;7(1):1-59.
40. Pop-Busui R, Boulton AJM, Feldman EL, et al. Diabetic neuropathy: a position statement by the American Diabetes Association. Diabetes Care. 2017;40(1):136-154.
41. Gupta V, Bansal R, Gupta A, Bhansali A. The sensitivity and specificity of nonmydriatic digital stereoscopic retinal imaging in detecting diabetic retinopathy. Indian J Ophthalmol. 2014;62(8):851-856.
42. Pérez MA, Bruce BB, Newman NJ, Biousse V. The use of retinal photography in non-ophthalmic settings and its potential for neurology. The Neurologist. 2012;18(6):350-355.
Diabetes mellitus is prevalent in our society; 1 in 10 Americans has the condition and > 1 in 3 has prediabetes.1 Due to the widespread comorbidities and complications of this disease, the American Diabetes Association (ADA) recommends that diabetes management focus on evaluation and treatment of complications.2 Diabetes-related complications can be life-altering and challenging for patients because their quality of life suffers.
For providers, there are several evidence-based screening tools and preventive practices (in and beyond glycemic control) that reduce diabetes complications such as congestive heart failure, kidney failure, lower extremity amputation, and stroke.3 We as providers can treat patients by implementing appropriate goal-directed therapy.4-6
In this 5-part series, I will explore the evidence and recommendations for a multimodal approach in a patient with type 2 diabetes. Here—in Part 1—I explore the self-care behaviors our patients can adopt to improve their symptoms of diabetes.
Case Report
Mr. W is an overweight 64-year-old man with hypertension, hyperlipidemia, and type 2 diabetes mellitus. He visits the clinic for his yearly physical exam. He is concerned because his father, who had diabetes, developed renal failure and had multiple amputations near the end of his life. He is worried that he might face the same outcomes and asks you what he can do to avoid his father’s fate.
Advising Your Patient on Self-care
The cornerstone of diabetes management is appropriate self-care. Both the ADA and the American Association of Clinical Endocrinologists (AACE) recommend that treatment plans should encourage the patient to adopt healthy lifestyle behaviors, including a healthy diet, regular exercise, weight control, and avoidance of tobacco.2,7,8 These interventions have positive effects on blood pressure, glucose control, and lipid levels. They can also reduce the risk for diabetic complications, including atherosclerotic cardiovascular disease (ASCVD), which is the foremost cause of death among patients with diabetes. During a patient visit, clinicians can suggest the following self-care interventions for improving long-term outcomes.
Education sessions. The ADA recommends that individuals with diabetes participate in diabetes self-management education and support (DSMES) sessions.2 In these sessions, patients with diabetes are instructed on a variety of self-care behaviors, including lifestyle interventions, medication management, self-monitoring, and problem-solving.9 These programs—often paid for in part by health insurance—are taught by health care professionals such as registered dieticians, nutritionists, or certified diabetes educators.9,10 Evidence suggests DSMES increases patients’ sense of self-efficacy and may improve blood sugar management.10 Clinicians can help guide their patients through the Association of Diabetes Care & Education Specialists’ online database to identify a DSMES program near them (see www.diabeteseducator.org/living-with-diabetes/find-an-education-program).11
Diet. The AACE recommends a plant-based diet high in polyunsaturated and monounsaturated fatty acids and limited in trans fatty acids and saturated fats.7 Evidence strongly suggests that a Mediterranean diet with high vegetable intake and decreased saturated fats helps to reduce the risk for major cardiovascular events (myocardial infarction and stroke).12
Continue to: Exercise
Exercise. Both the ADA and AACE recommend that most adults with diabetes engage in at least 150 min/week of moderate-to-vigorous aerobic and strength-training exercises.2,7 Clinicians should evaluate patients with sedentary lifestyles prior to them engaging in vigorous physical activity beyond simple walking.2 The ADA also recommends that patients should avoid sitting for long periods of time by engaging in physical activity at least every 30 minutes.2 For adults who may not be able to participate in moderate-to-vigorous exercise, recommend alternative flexibility and balance-training activities, such as yoga or tai chi, 2 to 3 times per week.2
Weight management—a combined effort of diet, exercise, and behavioral therapy—is pivotal in the management of type 2 diabetes due to the potential benefits in insulin resistance, blood pressure, hyperlipidemia, and other factors.2 Weight loss may also improve glycemic control and reduce the need for glucose-lowering medications.2 For patients who struggle with weight loss, consider prescribing FDA-approved weight-loss medications (phentermine, orlistat, lorcaserin, naltrexone/bupropion, liraglutide) or, in some cases, referring for bariatric surgery.2,7
Sleep hygiene is an important element in any preventive treatment plan. This includes interventions as simple as going to bed at the same time every night, sleeping in a dark room, sleeping for at least 7 hours, and removing electronic devices from the bedroom.13 Patients should avoid alcohol, caffeine, and large meals before bedtime.13
Additionally, obstructive sleep apnea (OSA) is often underdiagnosed in patients with diabetes and contributes to insulin resistance, inflammation, and elevated blood pressure.7,14 For early identification of OSA, order a sleep study when appropriate and refer patients to sleep specialists if needed. Patients who are recommended for treatment should be monitored for increasing compliance with care and to ensure benefit from treatment.
In Part 2, we’ll check in with Mr. W as I discuss the role of blood pressure monitoring and antihypertensive medications in reducing cardiovascular risks in patients with diabetes.
Diabetes mellitus is prevalent in our society; 1 in 10 Americans has the condition and > 1 in 3 has prediabetes.1 Due to the widespread comorbidities and complications of this disease, the American Diabetes Association (ADA) recommends that diabetes management focus on evaluation and treatment of complications.2 Diabetes-related complications can be life-altering and challenging for patients because their quality of life suffers.
For providers, there are several evidence-based screening tools and preventive practices (in and beyond glycemic control) that reduce diabetes complications such as congestive heart failure, kidney failure, lower extremity amputation, and stroke.3 We as providers can treat patients by implementing appropriate goal-directed therapy.4-6
In this 5-part series, I will explore the evidence and recommendations for a multimodal approach in a patient with type 2 diabetes. Here—in Part 1—I explore the self-care behaviors our patients can adopt to improve their symptoms of diabetes.
Case Report
Mr. W is an overweight 64-year-old man with hypertension, hyperlipidemia, and type 2 diabetes mellitus. He visits the clinic for his yearly physical exam. He is concerned because his father, who had diabetes, developed renal failure and had multiple amputations near the end of his life. He is worried that he might face the same outcomes and asks you what he can do to avoid his father’s fate.
Advising Your Patient on Self-care
The cornerstone of diabetes management is appropriate self-care. Both the ADA and the American Association of Clinical Endocrinologists (AACE) recommend that treatment plans should encourage the patient to adopt healthy lifestyle behaviors, including a healthy diet, regular exercise, weight control, and avoidance of tobacco.2,7,8 These interventions have positive effects on blood pressure, glucose control, and lipid levels. They can also reduce the risk for diabetic complications, including atherosclerotic cardiovascular disease (ASCVD), which is the foremost cause of death among patients with diabetes. During a patient visit, clinicians can suggest the following self-care interventions for improving long-term outcomes.
Education sessions. The ADA recommends that individuals with diabetes participate in diabetes self-management education and support (DSMES) sessions.2 In these sessions, patients with diabetes are instructed on a variety of self-care behaviors, including lifestyle interventions, medication management, self-monitoring, and problem-solving.9 These programs—often paid for in part by health insurance—are taught by health care professionals such as registered dieticians, nutritionists, or certified diabetes educators.9,10 Evidence suggests DSMES increases patients’ sense of self-efficacy and may improve blood sugar management.10 Clinicians can help guide their patients through the Association of Diabetes Care & Education Specialists’ online database to identify a DSMES program near them (see www.diabeteseducator.org/living-with-diabetes/find-an-education-program).11
Diet. The AACE recommends a plant-based diet high in polyunsaturated and monounsaturated fatty acids and limited in trans fatty acids and saturated fats.7 Evidence strongly suggests that a Mediterranean diet with high vegetable intake and decreased saturated fats helps to reduce the risk for major cardiovascular events (myocardial infarction and stroke).12
Continue to: Exercise
Exercise. Both the ADA and AACE recommend that most adults with diabetes engage in at least 150 min/week of moderate-to-vigorous aerobic and strength-training exercises.2,7 Clinicians should evaluate patients with sedentary lifestyles prior to them engaging in vigorous physical activity beyond simple walking.2 The ADA also recommends that patients should avoid sitting for long periods of time by engaging in physical activity at least every 30 minutes.2 For adults who may not be able to participate in moderate-to-vigorous exercise, recommend alternative flexibility and balance-training activities, such as yoga or tai chi, 2 to 3 times per week.2
Weight management—a combined effort of diet, exercise, and behavioral therapy—is pivotal in the management of type 2 diabetes due to the potential benefits in insulin resistance, blood pressure, hyperlipidemia, and other factors.2 Weight loss may also improve glycemic control and reduce the need for glucose-lowering medications.2 For patients who struggle with weight loss, consider prescribing FDA-approved weight-loss medications (phentermine, orlistat, lorcaserin, naltrexone/bupropion, liraglutide) or, in some cases, referring for bariatric surgery.2,7
Sleep hygiene is an important element in any preventive treatment plan. This includes interventions as simple as going to bed at the same time every night, sleeping in a dark room, sleeping for at least 7 hours, and removing electronic devices from the bedroom.13 Patients should avoid alcohol, caffeine, and large meals before bedtime.13
Additionally, obstructive sleep apnea (OSA) is often underdiagnosed in patients with diabetes and contributes to insulin resistance, inflammation, and elevated blood pressure.7,14 For early identification of OSA, order a sleep study when appropriate and refer patients to sleep specialists if needed. Patients who are recommended for treatment should be monitored for increasing compliance with care and to ensure benefit from treatment.
In Part 2, we’ll check in with Mr. W as I discuss the role of blood pressure monitoring and antihypertensive medications in reducing cardiovascular risks in patients with diabetes.
1. Centers for Disease Control and Prevention. Diabetes incidence and prevalence. Diabetes Report Card 2017. www.cdc.gov/diabetes/library/reports/reportcard/incidence-2017.html. Published 2018. Accessed June 18, 2020.
2. Standards of Medical Care in Diabetes—2020 Abridged for Primary Care Providers. American Diabetes Association Clinical Diabetes. 2020;38(1):10-38.
3. Chen Y, Sloan FA, Yashkin AP. Adherence to diabetes guidelines for screening, physical activity and medication and onset of complications and death. J Diabetes Complications. 2015;29(8):1228-1233.
4. Mehta S, Mocarski M, Wisniewski T, et al. Primary care physicians’ utilization of type 2 diabetes screening guidelines and referrals to behavioral interventions: a survey-linked retrospective study. BMJ Open Diabetes Res Care. 2017;5(1):e000406.
5. Center for Disease Control and Prevention. Preventive care practices. Diabetes Report Card 2017. www.cdc.gov/diabetes/library/reports/reportcard/preventive-care.html. Published 2018. Accessed June 18, 2020.
6. Arnold SV, de Lemos JA, Rosenson RS, et al; GOULD Investigators. Use of guideline-recommended risk reduction strategies among patients with diabetes and atherosclerotic cardiovascular disease. Circulation. 2019;140(7):618-620.
7. Garber AJ, Handelsman Y, Grunberger G, et al. Consensus Statement by the American Association of Clinical Endocrinologists and American College of Endocrinology on the comprehensive type 2 diabetes management algorithm—2020 executive summary. Endocr Pract Endocr Pract. 2020;26(1):107-139.
8. American Diabetes Association. Comprehensive medical evaluation and assessment of comorbidities: standards of medical care in diabetes—2020. Diabetes Care. 2020;43(suppl 1):S37-S47.
9. Beck J, Greenwood DA, Blanton L, et al; 2017 Standards Revision Task Force. 2017 National Standards for diabetes self-management education and support. Diabetes Educ. 2017;43(5): 449-464.
10. Chrvala CA, Sherr D, Lipman RD. Diabetes self-management education for adults with type 2 diabetes mellitus: a systematic review of the effect on glycemic control. Patient Educ Couns. 2016;99(6):926-943.
11. Association of Diabetes Care & Education Specialists. Find a diabetes education program in your area. www.diabeteseducator.org/living-with-diabetes/find-an-education-program. Accessed June 15, 2020.
12. Estruch R, Ros E, Salas-Salvadó J, et al; PREDIMED Study Investigators. Primary prevention of cardiovascular disease with a Mediterranean diet supplemented with extra-virgin olive oil or nuts. NEJM. 2018;378(25):e34.
13. Centers for Disease Control and Prevention. Tips for better sleep. Sleep and sleep disorders. www.cdc.gov/sleep/about_sleep/sleep_hygiene.html. Reviewed July 15, 2016. Accessed June 18, 2020.
14. Doumit J, Prasad B. Sleep Apnea in Type 2 Diabetes. Diabetes Spectrum. 2016; 29(1): 14-19.
15. Marso SP, Daniels GH, Brown-Frandsen K, et al; LEADER Steering Committee on behalf of the LEADER Trial Investigators. Liraglutide and cardiovascular outcomes in type 2 diabetes. N Engl J Med. 2016;375:311-322.
16. Perkovic V, Jardine MJ, Neal B, et al; CREDENCE Trial Investigators. Canagliflozin and renal outcomes in type 2 diabetes and nephropathy. N Engl J Med. 2019;380(24):2295-2306.
17. Trends in Blood pressure control and treatment among type 2 diabetes with comorbid hypertension in the United States: 1988-2004. J Hypertens. 2009;27(9):1908-1916.
18. Emdin CA, Rahimi K, Neal B, et al. Blood pressure lowering in type 2 diabetes: a systematic review and meta-analysis. JAMA. 2015;313(6):603-615.
19. Vouri SM, Shaw RF, Waterbury NV, et al. Prevalence of achievement of A1c, blood pressure, and cholesterol (ABC) goal in veterans with diabetes. J Manag Care Pharm. 2011;17(4):304-312.
20. Kudo N, Yokokawa H, Fukuda H, et al. Achievement of target blood pressure levels among Japanese workers with hypertension and healthy lifestyle characteristics associated with therapeutic failure. Plos One. 2015;10(7):e0133641.
21. Carey RM, Whelton PK; 2017 ACC/AHA Hypertension Guideline Writing Committee. Prevention, detection, evaluation, and management of high blood pressure in adults: synopsis of the 2017 American College of Cardiology/American Heart Association Hypertension guideline. Ann Intern Med. 2018;168(5):351-358.
22. Deedwania PC. Blood pressure control in diabetes mellitus. Circulation. 2011;123:2776–2778.
23. Catalá-López F, Saint-Gerons DM, González-Bermejo D, et al. Cardiovascular and renal outcomes of renin-angiotensin system blockade in adult patients with diabetes mellitus: a systematic review with network meta-analyses. PLoS Med. 2016;13(3):e1001971.
24. Furberg CD, Wright JT Jr, Davis BR, et al; ALLHAT Officers and Coordinators for the ALLHAT Collaborative Research Group. Major outcomes in high-risk hypertensive patients randomized to angiotensin-converting enzyme inhibitor or calcium channel blocker vs diuretic: the Antihypertensive and Lipid-Lowering Treatment to Prevent Heart Attack Trial (ALLHAT). JAMA. 2002;288(23):2981-2997.
25. Sleight P. The HOPE Study (Heart Outcomes Prevention Evaluation). J Renin-Angiotensin-Aldosterone Syst. 2000;1(1):18-20.
26. Tatti P, Pahor M, Byington RP, et al. Outcome results of the Fosinopril Versus Amlodipine Cardiovascular Events Randomized Trial (FACET) in patients with hypertension and NIDDM. Diabetes Care. 1998;21(4):597-603.
27. Schrier RW, Estacio RO, Jeffers B. Appropriate Blood Pressure Control in NIDDM (ABCD) Trial. Diabetologia. 1996;39(12):1646-1654.
28. Hansson L, Zanchetti A, Carruthers SG, et al; HOT Study Group. Effects of intensive blood-pressure lowering and low-dose aspirin in patients with hypertension: principal results of the Hypertension Optimal Treatment (HOT) Randomised Trial. Lancet. 1998;351(9118):1755-1762.
29. Baigent C, Blackwell L, Emberson J, et al; Cholesterol Treatment Trialists’ (CTT) Collaboration. Efficacy and safety of more intensive lowering of LDL cholesterol: a meta-analysis of data from 170,000 participants in 26 randomised trials. Lancet. 2010;376(9753):1670-1681.
30. Fu AZ, Zhang Q, Davies MJ, et al. Underutilization of statins in patients with type 2 diabetes in US clinical practice: a retrospective cohort study. Curr Med Res Opin. 2011;27(5):1035-1040.
31. Cannon CP, Blazing MA, Giugliano RP, et al; IMPROVE-IT Investigators. Ezetimibe added to statin therapy after acute coronary syndromes. N Engl J Med. 2015; 372:2387-2397
32. Sabatine MS, Giugliano RP, Keech AC, et al; the FOURIER Steering Committee and Investigators. Evolocumab and clinical outcomes in patients with cardiovascular disease. N Engl J Med. 2017;376:1713-1722.
33. Schwartz GG, Steg PG, Szarek M, et al; ODYSSEY OUTCOMES Committees and Investigators. Alirocumab and Cardiovascular Outcomes after Acute Coronary Syndrome | NEJM. N Engl J Med. 2018;379:2097-2107.
34. Icosapent ethyl [package insert]. Bridgewater, NJ: Amarin Pharma, Inc.; 2019.
35. Bhatt DL, Steg PG, Miller M, et al; REDUCE-IT Investigators. Cardiovascular risk reduction with icosapent ethyl for hypertriglyceridemia. N Engl J Med. 2019;380:11-22
36. Bolton WK. Renal Physicians Association Clinical practice guideline: appropriate patient preparation for renal replacement therapy: guideline number 3. J Am Soc Nephrol. 2003;14(5):1406-1410.
37. American Diabetes Association. Pharmacologic Approaches to glycemic treatment: standards of medical care in diabetes—2020. Diabetes Care. 2020;43(suppl 1):S98-S110.
38. Qaseem A, Barry MJ, Humphrey LL, Forciea MA; Clinical Guidelines Committee of the American College of Physicians. Oral pharmacologic treatment of type 2 diabetes mellitus: a clinical practice guideline update from the American College of Physicians. Ann Intern Med. 2017;166(4):279-290.
39. Kidney Disease: Improving Global Outcomes (KDIGO) CKD-MBD Update Work Group. KDIGO 2017 Clinical Practice Guideline Update for the diagnosis, evaluation, prevention, and treatment of chronic kidney disease–mineral and bone disorder (CKD-MBD). Kidney Int Suppl (2011). 2017;7(1):1-59.
40. Pop-Busui R, Boulton AJM, Feldman EL, et al. Diabetic neuropathy: a position statement by the American Diabetes Association. Diabetes Care. 2017;40(1):136-154.
41. Gupta V, Bansal R, Gupta A, Bhansali A. The sensitivity and specificity of nonmydriatic digital stereoscopic retinal imaging in detecting diabetic retinopathy. Indian J Ophthalmol. 2014;62(8):851-856.
42. Pérez MA, Bruce BB, Newman NJ, Biousse V. The use of retinal photography in non-ophthalmic settings and its potential for neurology. The Neurologist. 2012;18(6):350-355.
1. Centers for Disease Control and Prevention. Diabetes incidence and prevalence. Diabetes Report Card 2017. www.cdc.gov/diabetes/library/reports/reportcard/incidence-2017.html. Published 2018. Accessed June 18, 2020.
2. Standards of Medical Care in Diabetes—2020 Abridged for Primary Care Providers. American Diabetes Association Clinical Diabetes. 2020;38(1):10-38.
3. Chen Y, Sloan FA, Yashkin AP. Adherence to diabetes guidelines for screening, physical activity and medication and onset of complications and death. J Diabetes Complications. 2015;29(8):1228-1233.
4. Mehta S, Mocarski M, Wisniewski T, et al. Primary care physicians’ utilization of type 2 diabetes screening guidelines and referrals to behavioral interventions: a survey-linked retrospective study. BMJ Open Diabetes Res Care. 2017;5(1):e000406.
5. Center for Disease Control and Prevention. Preventive care practices. Diabetes Report Card 2017. www.cdc.gov/diabetes/library/reports/reportcard/preventive-care.html. Published 2018. Accessed June 18, 2020.
6. Arnold SV, de Lemos JA, Rosenson RS, et al; GOULD Investigators. Use of guideline-recommended risk reduction strategies among patients with diabetes and atherosclerotic cardiovascular disease. Circulation. 2019;140(7):618-620.
7. Garber AJ, Handelsman Y, Grunberger G, et al. Consensus Statement by the American Association of Clinical Endocrinologists and American College of Endocrinology on the comprehensive type 2 diabetes management algorithm—2020 executive summary. Endocr Pract Endocr Pract. 2020;26(1):107-139.
8. American Diabetes Association. Comprehensive medical evaluation and assessment of comorbidities: standards of medical care in diabetes—2020. Diabetes Care. 2020;43(suppl 1):S37-S47.
9. Beck J, Greenwood DA, Blanton L, et al; 2017 Standards Revision Task Force. 2017 National Standards for diabetes self-management education and support. Diabetes Educ. 2017;43(5): 449-464.
10. Chrvala CA, Sherr D, Lipman RD. Diabetes self-management education for adults with type 2 diabetes mellitus: a systematic review of the effect on glycemic control. Patient Educ Couns. 2016;99(6):926-943.
11. Association of Diabetes Care & Education Specialists. Find a diabetes education program in your area. www.diabeteseducator.org/living-with-diabetes/find-an-education-program. Accessed June 15, 2020.
12. Estruch R, Ros E, Salas-Salvadó J, et al; PREDIMED Study Investigators. Primary prevention of cardiovascular disease with a Mediterranean diet supplemented with extra-virgin olive oil or nuts. NEJM. 2018;378(25):e34.
13. Centers for Disease Control and Prevention. Tips for better sleep. Sleep and sleep disorders. www.cdc.gov/sleep/about_sleep/sleep_hygiene.html. Reviewed July 15, 2016. Accessed June 18, 2020.
14. Doumit J, Prasad B. Sleep Apnea in Type 2 Diabetes. Diabetes Spectrum. 2016; 29(1): 14-19.
15. Marso SP, Daniels GH, Brown-Frandsen K, et al; LEADER Steering Committee on behalf of the LEADER Trial Investigators. Liraglutide and cardiovascular outcomes in type 2 diabetes. N Engl J Med. 2016;375:311-322.
16. Perkovic V, Jardine MJ, Neal B, et al; CREDENCE Trial Investigators. Canagliflozin and renal outcomes in type 2 diabetes and nephropathy. N Engl J Med. 2019;380(24):2295-2306.
17. Trends in Blood pressure control and treatment among type 2 diabetes with comorbid hypertension in the United States: 1988-2004. J Hypertens. 2009;27(9):1908-1916.
18. Emdin CA, Rahimi K, Neal B, et al. Blood pressure lowering in type 2 diabetes: a systematic review and meta-analysis. JAMA. 2015;313(6):603-615.
19. Vouri SM, Shaw RF, Waterbury NV, et al. Prevalence of achievement of A1c, blood pressure, and cholesterol (ABC) goal in veterans with diabetes. J Manag Care Pharm. 2011;17(4):304-312.
20. Kudo N, Yokokawa H, Fukuda H, et al. Achievement of target blood pressure levels among Japanese workers with hypertension and healthy lifestyle characteristics associated with therapeutic failure. Plos One. 2015;10(7):e0133641.
21. Carey RM, Whelton PK; 2017 ACC/AHA Hypertension Guideline Writing Committee. Prevention, detection, evaluation, and management of high blood pressure in adults: synopsis of the 2017 American College of Cardiology/American Heart Association Hypertension guideline. Ann Intern Med. 2018;168(5):351-358.
22. Deedwania PC. Blood pressure control in diabetes mellitus. Circulation. 2011;123:2776–2778.
23. Catalá-López F, Saint-Gerons DM, González-Bermejo D, et al. Cardiovascular and renal outcomes of renin-angiotensin system blockade in adult patients with diabetes mellitus: a systematic review with network meta-analyses. PLoS Med. 2016;13(3):e1001971.
24. Furberg CD, Wright JT Jr, Davis BR, et al; ALLHAT Officers and Coordinators for the ALLHAT Collaborative Research Group. Major outcomes in high-risk hypertensive patients randomized to angiotensin-converting enzyme inhibitor or calcium channel blocker vs diuretic: the Antihypertensive and Lipid-Lowering Treatment to Prevent Heart Attack Trial (ALLHAT). JAMA. 2002;288(23):2981-2997.
25. Sleight P. The HOPE Study (Heart Outcomes Prevention Evaluation). J Renin-Angiotensin-Aldosterone Syst. 2000;1(1):18-20.
26. Tatti P, Pahor M, Byington RP, et al. Outcome results of the Fosinopril Versus Amlodipine Cardiovascular Events Randomized Trial (FACET) in patients with hypertension and NIDDM. Diabetes Care. 1998;21(4):597-603.
27. Schrier RW, Estacio RO, Jeffers B. Appropriate Blood Pressure Control in NIDDM (ABCD) Trial. Diabetologia. 1996;39(12):1646-1654.
28. Hansson L, Zanchetti A, Carruthers SG, et al; HOT Study Group. Effects of intensive blood-pressure lowering and low-dose aspirin in patients with hypertension: principal results of the Hypertension Optimal Treatment (HOT) Randomised Trial. Lancet. 1998;351(9118):1755-1762.
29. Baigent C, Blackwell L, Emberson J, et al; Cholesterol Treatment Trialists’ (CTT) Collaboration. Efficacy and safety of more intensive lowering of LDL cholesterol: a meta-analysis of data from 170,000 participants in 26 randomised trials. Lancet. 2010;376(9753):1670-1681.
30. Fu AZ, Zhang Q, Davies MJ, et al. Underutilization of statins in patients with type 2 diabetes in US clinical practice: a retrospective cohort study. Curr Med Res Opin. 2011;27(5):1035-1040.
31. Cannon CP, Blazing MA, Giugliano RP, et al; IMPROVE-IT Investigators. Ezetimibe added to statin therapy after acute coronary syndromes. N Engl J Med. 2015; 372:2387-2397
32. Sabatine MS, Giugliano RP, Keech AC, et al; the FOURIER Steering Committee and Investigators. Evolocumab and clinical outcomes in patients with cardiovascular disease. N Engl J Med. 2017;376:1713-1722.
33. Schwartz GG, Steg PG, Szarek M, et al; ODYSSEY OUTCOMES Committees and Investigators. Alirocumab and Cardiovascular Outcomes after Acute Coronary Syndrome | NEJM. N Engl J Med. 2018;379:2097-2107.
34. Icosapent ethyl [package insert]. Bridgewater, NJ: Amarin Pharma, Inc.; 2019.
35. Bhatt DL, Steg PG, Miller M, et al; REDUCE-IT Investigators. Cardiovascular risk reduction with icosapent ethyl for hypertriglyceridemia. N Engl J Med. 2019;380:11-22
36. Bolton WK. Renal Physicians Association Clinical practice guideline: appropriate patient preparation for renal replacement therapy: guideline number 3. J Am Soc Nephrol. 2003;14(5):1406-1410.
37. American Diabetes Association. Pharmacologic Approaches to glycemic treatment: standards of medical care in diabetes—2020. Diabetes Care. 2020;43(suppl 1):S98-S110.
38. Qaseem A, Barry MJ, Humphrey LL, Forciea MA; Clinical Guidelines Committee of the American College of Physicians. Oral pharmacologic treatment of type 2 diabetes mellitus: a clinical practice guideline update from the American College of Physicians. Ann Intern Med. 2017;166(4):279-290.
39. Kidney Disease: Improving Global Outcomes (KDIGO) CKD-MBD Update Work Group. KDIGO 2017 Clinical Practice Guideline Update for the diagnosis, evaluation, prevention, and treatment of chronic kidney disease–mineral and bone disorder (CKD-MBD). Kidney Int Suppl (2011). 2017;7(1):1-59.
40. Pop-Busui R, Boulton AJM, Feldman EL, et al. Diabetic neuropathy: a position statement by the American Diabetes Association. Diabetes Care. 2017;40(1):136-154.
41. Gupta V, Bansal R, Gupta A, Bhansali A. The sensitivity and specificity of nonmydriatic digital stereoscopic retinal imaging in detecting diabetic retinopathy. Indian J Ophthalmol. 2014;62(8):851-856.
42. Pérez MA, Bruce BB, Newman NJ, Biousse V. The use of retinal photography in non-ophthalmic settings and its potential for neurology. The Neurologist. 2012;18(6):350-355.
Dapagliflozin benefits low-EF heart failure regardless of diuretic dose: DAPA-HF
The DAPA-HF trial has already changed cardiology in opening up a new class of drugs to patients with heart failure (HF), whether or not they have diabetes. Now the trial is yielding clues as to how it benefits them. For now, it’s doing so by process of elimination.
A new analysis suggests that dapagliflozin (Farxiga, AstraZeneca) didn’t need help from loop diuretics to cut the risk for clinical events in patients with HF with reduced ejection fraction (HFrEF), a benefit seen across the spectrum of glycosylated hemoglobin levels and without compromising renal function, said DAPA-HF investigators. Also, use of dapagliflozin and its clinical effects were not associated with changes in loop diuretic dosage. Those findings and others suggest the drug helps in HFrEF at least partly by some other mechanism than its own diuretic effect, the researchers say.
Such insights will likely be important to case-by-case decisions on whether to use the drug, a sodium-glucose cotransporter 2 (SGLT2) inhibitor once reserved for patients with diabetes, given the recently broader landscape of HF treatment options.
As previously reported from DAPA-HF, with more than 4,700 patients, those who received dapagliflozin showed significant reductions in the primary end point, a composite of cardiovascular (CV) death, HF hospitalization, and urgent HF visit requiring IV therapy over about 18 months. The 45% of patients with and 55% without type 2 diabetes enjoyed about equal benefit in the placebo-controlled trial for that end point, as well as for all-cause mortality.
SGLT2 inhibitors work in diabetes by promoting urinary glucose excretion. That had led some to speculate that its benefit in HFrEF comes primarily from a diuretic effect; the current findings largely put that question to rest.
“Our findings show that treatment with dapagliflozin was effective regardless of diuretic use or diuretic dose. They also show that dapagliflozin did not lead to an increase in renal adverse events or discontinuation of therapy in patients treated with a diuretic,” trialist Alice M. Jackson, MB, ChB, said in an interview.
“In fact, renal adverse events were generally less common in patients treated with dapagliflozin, across the diuretic categories,” said Dr. Jackson, from the University of Glasgow.
Dr. Jackson presented the new analysis at a Late-Breaking Science Session during the European Society of Cardiology Heart Failure Discoveries virtual meeting. The HFA sessions were conducted virtually this year due to the COVID-19 pandemic.
At baseline, 84% of patients were on conventional diuretics. The post hoc analysis broke out all patients by loop-diuretic dosage level: none; less than 40 mg furosemide equivalents (FE); 40 mg FE; or more than 40 mg FE. Clinical outcomes were similar across the four groups.
Clinicians in the trial “were not given specific advice about adjusting diuretic doses, but were encouraged to assess volume status and make changes to medical therapy based on this, if necessary,” Dr. Jackson said. “This suggests that, for most patients, starting dapagliflozin will not necessitate a change in diuretic dose.”
With the caveat that the event rate was low in the relatively few patients not prescribed loop diuretics, she said, “the magnitude of the benefit from dapagliflozin appeared to be larger in patients not treated with a diuretic.”
There was no suggestion of a diuretic dose–response effect or statistical interaction between diuretic use and clinical outcomes on dapagliflozin, Dr. Jackson observed in the interview.
Of note in the analysis, hematocrit levels shot up soon after patients started active therapy, but they didn’t rise much in the placebo group. The sustained hematocrit elevation on dapagliflozin, seen at all diuretic dosage levels, persisted even after dosage reductions at 6 months, she said.
“Dapagliflozin is effective in HFrEF irrespective of background diuretic therapy; therefore, it is almost certainly not purely acting as a diuretic,” Andrew J. Coats, MD, DSc, MBA, said in an interview.
The findings also “lessen the concern that dapagliflozin’s beneficial effects are only seen only in patients without effective diuretic dosing,” said Dr. Coats, from University of Warwick, Coventry, England.
“Altogether, these data give further reassurance that dapagliflozin can safely be used in heart failure, and has a beneficial effect independent of the use of diuretic drugs,” invited discussant Wolfram Doehner, MD, PhD, Charité-Universitätsmedizin Berlin, said after Dr. Jackson’s presentation of the analysis.
He made special mention of the sustained hematocrit elevation on dapagliflozin. “While this effect may likely relate to the mild reduction in plasma volume secondary to dapagliflozin therapy, it is noted that the increase in hematocrit was independent of any change of the diuretic dose,” Doehner said. “If additional mechanisms have a role for this observed increase in hematocrit, it may be of interest in further investigations.”
Dr. Jackson pointed to several observations that suggest the hematocrit finding isn’t explained by hemoconcentration from reduced plasma volume, at least not entirely.
For example, hematocrit levels rose “without any suggestion of a relationship between diuretic dose and degree of hematocrit elevation with dapagliflozin,” she said.
The elevations persisted even with diuretic dose reductions at 6 and 12 months, “which should have led to a decrease in hemoconcentration if it was caused by volume contraction.”
Also, she said, “among patients not taking a diuretic, volume depletion occurred less frequently in the dapagliflozin group than in the placebo group, but there was still a similar rise in hematocrit with dapagliflozin.”
Both Dr. Jackson and Dr. Coats said the sustained elevation in hematocrit on the drug is unlikely to pose a major hazard.
Dr. Coats said that, theoretically, “increased hematocrit could reduce peripheral vessel blood flow, making ischemia and thrombosis more likely. But the size of the effect is small and unlikely to be clinically important.”
A diuretic dose could not be determined for 128 of the trial’s 4,744 randomized patients with HFrEF, so the post hoc analysis was limited to the remaining 4,616. Of those, 746 were not on diuretics at baseline, 1,311 were on loop diuretics at less than 40 mg FE or on non-loop diuretics only, 1,365 were taking 40 mg FE, and 1,204 were on higher doses of loop diuretics.
The mean baseline dosage was 60 mg FE, which rose slightly throughout the trial. But the baseline dosage and the increases were both similar in the placebo and dapagliflozin groups. Dr. Jackson said 84% and 83% of patients on dapagliflozin and placebo, respectively, maintained their baseline dose at 6 months and about 77% in both groups at 12 months.
The overall trial’s significant primary endpoint reduction for dapagliflozin versus placebo applied similarly to patients not on a diuretics and to those on any dose of diuretic, with an interaction P value of .23 for the effect of diuretic use. The hazard ratios (95% confidence interval) were 0.57 (0.36-0.92) for patients not on diuretics, 0.78 (0.68-0.90) for patients on any diuretic dosage, and 0.74 (0.65-0.85) overall
Dr. Jackson said during her formal online presentation that patients on diuretics showed a “tendency toward slightly more volume depletion in those on dapagliflozin than in those on placebo, but the excess was small and not greater than approximately 3% in those taking 40 mg furosemide equivalent diuretic. And fortunately, this did not result in an increase in frequency in renal adverse events nor of discontinuation of study drug.”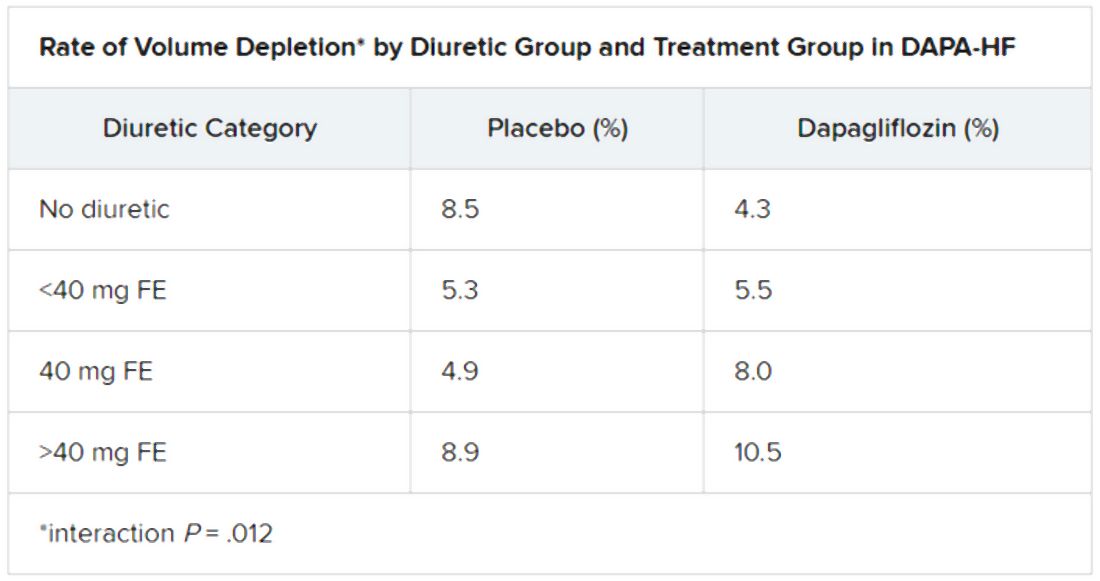
Renal adverse events were similarly prevalent in the two treatment groups, as were such events leading to treatment discontinuation. But serious renal events were less common in the dapagliflozin group (1.6% vs 2.7%; P = .009), as was investigator-reported serious acute kidney injury (1.0% vs 1.9%; P = .007).
“Overall, renal events were infrequent,” Dr. Jackson said, and “because of the small number of events, it is very difficult to draw conclusions about the impact of dapagliflozin on renal function according to diuretic-dose subgroups.”
Still, she said, worsening renal function was less common on dapagliflozin in three of the four groups by diuretic dosage; the exception was the less than 40 mg FE group, “but the absolute difference in this group was only two events.”
There seem to be dapagliflozin mechanisms “underneath the surface that need to be unraveled,” Dr. Doehner said as discussant, processes that are favorable for the treatment of HFrEF in which “diuretics play no big role.”
Dr. Jackson has no disclosures. Dr. Coats has disclosed receiving personal fees from Actimed, AstraZeneca, Faraday, WL Gore, Menarini, Novartis, Nutricia, Respicardia, Servier, Stealth Peptides, Verona, and Vifor. Dr. Doener has recently disclosed receiving grants and personal fees from Vifor, Pfizer, Boehringer Ingelheim, Sphingotec, ZS Pharma, Bayer, and Medtronic.
A version of this article originally appeared on Medscape.com.
The DAPA-HF trial has already changed cardiology in opening up a new class of drugs to patients with heart failure (HF), whether or not they have diabetes. Now the trial is yielding clues as to how it benefits them. For now, it’s doing so by process of elimination.
A new analysis suggests that dapagliflozin (Farxiga, AstraZeneca) didn’t need help from loop diuretics to cut the risk for clinical events in patients with HF with reduced ejection fraction (HFrEF), a benefit seen across the spectrum of glycosylated hemoglobin levels and without compromising renal function, said DAPA-HF investigators. Also, use of dapagliflozin and its clinical effects were not associated with changes in loop diuretic dosage. Those findings and others suggest the drug helps in HFrEF at least partly by some other mechanism than its own diuretic effect, the researchers say.
Such insights will likely be important to case-by-case decisions on whether to use the drug, a sodium-glucose cotransporter 2 (SGLT2) inhibitor once reserved for patients with diabetes, given the recently broader landscape of HF treatment options.
As previously reported from DAPA-HF, with more than 4,700 patients, those who received dapagliflozin showed significant reductions in the primary end point, a composite of cardiovascular (CV) death, HF hospitalization, and urgent HF visit requiring IV therapy over about 18 months. The 45% of patients with and 55% without type 2 diabetes enjoyed about equal benefit in the placebo-controlled trial for that end point, as well as for all-cause mortality.
SGLT2 inhibitors work in diabetes by promoting urinary glucose excretion. That had led some to speculate that its benefit in HFrEF comes primarily from a diuretic effect; the current findings largely put that question to rest.
“Our findings show that treatment with dapagliflozin was effective regardless of diuretic use or diuretic dose. They also show that dapagliflozin did not lead to an increase in renal adverse events or discontinuation of therapy in patients treated with a diuretic,” trialist Alice M. Jackson, MB, ChB, said in an interview.
“In fact, renal adverse events were generally less common in patients treated with dapagliflozin, across the diuretic categories,” said Dr. Jackson, from the University of Glasgow.
Dr. Jackson presented the new analysis at a Late-Breaking Science Session during the European Society of Cardiology Heart Failure Discoveries virtual meeting. The HFA sessions were conducted virtually this year due to the COVID-19 pandemic.
At baseline, 84% of patients were on conventional diuretics. The post hoc analysis broke out all patients by loop-diuretic dosage level: none; less than 40 mg furosemide equivalents (FE); 40 mg FE; or more than 40 mg FE. Clinical outcomes were similar across the four groups.
Clinicians in the trial “were not given specific advice about adjusting diuretic doses, but were encouraged to assess volume status and make changes to medical therapy based on this, if necessary,” Dr. Jackson said. “This suggests that, for most patients, starting dapagliflozin will not necessitate a change in diuretic dose.”
With the caveat that the event rate was low in the relatively few patients not prescribed loop diuretics, she said, “the magnitude of the benefit from dapagliflozin appeared to be larger in patients not treated with a diuretic.”
There was no suggestion of a diuretic dose–response effect or statistical interaction between diuretic use and clinical outcomes on dapagliflozin, Dr. Jackson observed in the interview.
Of note in the analysis, hematocrit levels shot up soon after patients started active therapy, but they didn’t rise much in the placebo group. The sustained hematocrit elevation on dapagliflozin, seen at all diuretic dosage levels, persisted even after dosage reductions at 6 months, she said.
“Dapagliflozin is effective in HFrEF irrespective of background diuretic therapy; therefore, it is almost certainly not purely acting as a diuretic,” Andrew J. Coats, MD, DSc, MBA, said in an interview.
The findings also “lessen the concern that dapagliflozin’s beneficial effects are only seen only in patients without effective diuretic dosing,” said Dr. Coats, from University of Warwick, Coventry, England.
“Altogether, these data give further reassurance that dapagliflozin can safely be used in heart failure, and has a beneficial effect independent of the use of diuretic drugs,” invited discussant Wolfram Doehner, MD, PhD, Charité-Universitätsmedizin Berlin, said after Dr. Jackson’s presentation of the analysis.
He made special mention of the sustained hematocrit elevation on dapagliflozin. “While this effect may likely relate to the mild reduction in plasma volume secondary to dapagliflozin therapy, it is noted that the increase in hematocrit was independent of any change of the diuretic dose,” Doehner said. “If additional mechanisms have a role for this observed increase in hematocrit, it may be of interest in further investigations.”
Dr. Jackson pointed to several observations that suggest the hematocrit finding isn’t explained by hemoconcentration from reduced plasma volume, at least not entirely.
For example, hematocrit levels rose “without any suggestion of a relationship between diuretic dose and degree of hematocrit elevation with dapagliflozin,” she said.
The elevations persisted even with diuretic dose reductions at 6 and 12 months, “which should have led to a decrease in hemoconcentration if it was caused by volume contraction.”
Also, she said, “among patients not taking a diuretic, volume depletion occurred less frequently in the dapagliflozin group than in the placebo group, but there was still a similar rise in hematocrit with dapagliflozin.”
Both Dr. Jackson and Dr. Coats said the sustained elevation in hematocrit on the drug is unlikely to pose a major hazard.
Dr. Coats said that, theoretically, “increased hematocrit could reduce peripheral vessel blood flow, making ischemia and thrombosis more likely. But the size of the effect is small and unlikely to be clinically important.”
A diuretic dose could not be determined for 128 of the trial’s 4,744 randomized patients with HFrEF, so the post hoc analysis was limited to the remaining 4,616. Of those, 746 were not on diuretics at baseline, 1,311 were on loop diuretics at less than 40 mg FE or on non-loop diuretics only, 1,365 were taking 40 mg FE, and 1,204 were on higher doses of loop diuretics.
The mean baseline dosage was 60 mg FE, which rose slightly throughout the trial. But the baseline dosage and the increases were both similar in the placebo and dapagliflozin groups. Dr. Jackson said 84% and 83% of patients on dapagliflozin and placebo, respectively, maintained their baseline dose at 6 months and about 77% in both groups at 12 months.
The overall trial’s significant primary endpoint reduction for dapagliflozin versus placebo applied similarly to patients not on a diuretics and to those on any dose of diuretic, with an interaction P value of .23 for the effect of diuretic use. The hazard ratios (95% confidence interval) were 0.57 (0.36-0.92) for patients not on diuretics, 0.78 (0.68-0.90) for patients on any diuretic dosage, and 0.74 (0.65-0.85) overall
Dr. Jackson said during her formal online presentation that patients on diuretics showed a “tendency toward slightly more volume depletion in those on dapagliflozin than in those on placebo, but the excess was small and not greater than approximately 3% in those taking 40 mg furosemide equivalent diuretic. And fortunately, this did not result in an increase in frequency in renal adverse events nor of discontinuation of study drug.”
Renal adverse events were similarly prevalent in the two treatment groups, as were such events leading to treatment discontinuation. But serious renal events were less common in the dapagliflozin group (1.6% vs 2.7%; P = .009), as was investigator-reported serious acute kidney injury (1.0% vs 1.9%; P = .007).
“Overall, renal events were infrequent,” Dr. Jackson said, and “because of the small number of events, it is very difficult to draw conclusions about the impact of dapagliflozin on renal function according to diuretic-dose subgroups.”
Still, she said, worsening renal function was less common on dapagliflozin in three of the four groups by diuretic dosage; the exception was the less than 40 mg FE group, “but the absolute difference in this group was only two events.”
There seem to be dapagliflozin mechanisms “underneath the surface that need to be unraveled,” Dr. Doehner said as discussant, processes that are favorable for the treatment of HFrEF in which “diuretics play no big role.”
Dr. Jackson has no disclosures. Dr. Coats has disclosed receiving personal fees from Actimed, AstraZeneca, Faraday, WL Gore, Menarini, Novartis, Nutricia, Respicardia, Servier, Stealth Peptides, Verona, and Vifor. Dr. Doener has recently disclosed receiving grants and personal fees from Vifor, Pfizer, Boehringer Ingelheim, Sphingotec, ZS Pharma, Bayer, and Medtronic.
A version of this article originally appeared on Medscape.com.
The DAPA-HF trial has already changed cardiology in opening up a new class of drugs to patients with heart failure (HF), whether or not they have diabetes. Now the trial is yielding clues as to how it benefits them. For now, it’s doing so by process of elimination.
A new analysis suggests that dapagliflozin (Farxiga, AstraZeneca) didn’t need help from loop diuretics to cut the risk for clinical events in patients with HF with reduced ejection fraction (HFrEF), a benefit seen across the spectrum of glycosylated hemoglobin levels and without compromising renal function, said DAPA-HF investigators. Also, use of dapagliflozin and its clinical effects were not associated with changes in loop diuretic dosage. Those findings and others suggest the drug helps in HFrEF at least partly by some other mechanism than its own diuretic effect, the researchers say.
Such insights will likely be important to case-by-case decisions on whether to use the drug, a sodium-glucose cotransporter 2 (SGLT2) inhibitor once reserved for patients with diabetes, given the recently broader landscape of HF treatment options.
As previously reported from DAPA-HF, with more than 4,700 patients, those who received dapagliflozin showed significant reductions in the primary end point, a composite of cardiovascular (CV) death, HF hospitalization, and urgent HF visit requiring IV therapy over about 18 months. The 45% of patients with and 55% without type 2 diabetes enjoyed about equal benefit in the placebo-controlled trial for that end point, as well as for all-cause mortality.
SGLT2 inhibitors work in diabetes by promoting urinary glucose excretion. That had led some to speculate that its benefit in HFrEF comes primarily from a diuretic effect; the current findings largely put that question to rest.
“Our findings show that treatment with dapagliflozin was effective regardless of diuretic use or diuretic dose. They also show that dapagliflozin did not lead to an increase in renal adverse events or discontinuation of therapy in patients treated with a diuretic,” trialist Alice M. Jackson, MB, ChB, said in an interview.
“In fact, renal adverse events were generally less common in patients treated with dapagliflozin, across the diuretic categories,” said Dr. Jackson, from the University of Glasgow.
Dr. Jackson presented the new analysis at a Late-Breaking Science Session during the European Society of Cardiology Heart Failure Discoveries virtual meeting. The HFA sessions were conducted virtually this year due to the COVID-19 pandemic.
At baseline, 84% of patients were on conventional diuretics. The post hoc analysis broke out all patients by loop-diuretic dosage level: none; less than 40 mg furosemide equivalents (FE); 40 mg FE; or more than 40 mg FE. Clinical outcomes were similar across the four groups.
Clinicians in the trial “were not given specific advice about adjusting diuretic doses, but were encouraged to assess volume status and make changes to medical therapy based on this, if necessary,” Dr. Jackson said. “This suggests that, for most patients, starting dapagliflozin will not necessitate a change in diuretic dose.”
With the caveat that the event rate was low in the relatively few patients not prescribed loop diuretics, she said, “the magnitude of the benefit from dapagliflozin appeared to be larger in patients not treated with a diuretic.”
There was no suggestion of a diuretic dose–response effect or statistical interaction between diuretic use and clinical outcomes on dapagliflozin, Dr. Jackson observed in the interview.
Of note in the analysis, hematocrit levels shot up soon after patients started active therapy, but they didn’t rise much in the placebo group. The sustained hematocrit elevation on dapagliflozin, seen at all diuretic dosage levels, persisted even after dosage reductions at 6 months, she said.
“Dapagliflozin is effective in HFrEF irrespective of background diuretic therapy; therefore, it is almost certainly not purely acting as a diuretic,” Andrew J. Coats, MD, DSc, MBA, said in an interview.
The findings also “lessen the concern that dapagliflozin’s beneficial effects are only seen only in patients without effective diuretic dosing,” said Dr. Coats, from University of Warwick, Coventry, England.
“Altogether, these data give further reassurance that dapagliflozin can safely be used in heart failure, and has a beneficial effect independent of the use of diuretic drugs,” invited discussant Wolfram Doehner, MD, PhD, Charité-Universitätsmedizin Berlin, said after Dr. Jackson’s presentation of the analysis.
He made special mention of the sustained hematocrit elevation on dapagliflozin. “While this effect may likely relate to the mild reduction in plasma volume secondary to dapagliflozin therapy, it is noted that the increase in hematocrit was independent of any change of the diuretic dose,” Doehner said. “If additional mechanisms have a role for this observed increase in hematocrit, it may be of interest in further investigations.”
Dr. Jackson pointed to several observations that suggest the hematocrit finding isn’t explained by hemoconcentration from reduced plasma volume, at least not entirely.
For example, hematocrit levels rose “without any suggestion of a relationship between diuretic dose and degree of hematocrit elevation with dapagliflozin,” she said.
The elevations persisted even with diuretic dose reductions at 6 and 12 months, “which should have led to a decrease in hemoconcentration if it was caused by volume contraction.”
Also, she said, “among patients not taking a diuretic, volume depletion occurred less frequently in the dapagliflozin group than in the placebo group, but there was still a similar rise in hematocrit with dapagliflozin.”
Both Dr. Jackson and Dr. Coats said the sustained elevation in hematocrit on the drug is unlikely to pose a major hazard.
Dr. Coats said that, theoretically, “increased hematocrit could reduce peripheral vessel blood flow, making ischemia and thrombosis more likely. But the size of the effect is small and unlikely to be clinically important.”
A diuretic dose could not be determined for 128 of the trial’s 4,744 randomized patients with HFrEF, so the post hoc analysis was limited to the remaining 4,616. Of those, 746 were not on diuretics at baseline, 1,311 were on loop diuretics at less than 40 mg FE or on non-loop diuretics only, 1,365 were taking 40 mg FE, and 1,204 were on higher doses of loop diuretics.
The mean baseline dosage was 60 mg FE, which rose slightly throughout the trial. But the baseline dosage and the increases were both similar in the placebo and dapagliflozin groups. Dr. Jackson said 84% and 83% of patients on dapagliflozin and placebo, respectively, maintained their baseline dose at 6 months and about 77% in both groups at 12 months.
The overall trial’s significant primary endpoint reduction for dapagliflozin versus placebo applied similarly to patients not on a diuretics and to those on any dose of diuretic, with an interaction P value of .23 for the effect of diuretic use. The hazard ratios (95% confidence interval) were 0.57 (0.36-0.92) for patients not on diuretics, 0.78 (0.68-0.90) for patients on any diuretic dosage, and 0.74 (0.65-0.85) overall
Dr. Jackson said during her formal online presentation that patients on diuretics showed a “tendency toward slightly more volume depletion in those on dapagliflozin than in those on placebo, but the excess was small and not greater than approximately 3% in those taking 40 mg furosemide equivalent diuretic. And fortunately, this did not result in an increase in frequency in renal adverse events nor of discontinuation of study drug.”
Renal adverse events were similarly prevalent in the two treatment groups, as were such events leading to treatment discontinuation. But serious renal events were less common in the dapagliflozin group (1.6% vs 2.7%; P = .009), as was investigator-reported serious acute kidney injury (1.0% vs 1.9%; P = .007).
“Overall, renal events were infrequent,” Dr. Jackson said, and “because of the small number of events, it is very difficult to draw conclusions about the impact of dapagliflozin on renal function according to diuretic-dose subgroups.”
Still, she said, worsening renal function was less common on dapagliflozin in three of the four groups by diuretic dosage; the exception was the less than 40 mg FE group, “but the absolute difference in this group was only two events.”
There seem to be dapagliflozin mechanisms “underneath the surface that need to be unraveled,” Dr. Doehner said as discussant, processes that are favorable for the treatment of HFrEF in which “diuretics play no big role.”
Dr. Jackson has no disclosures. Dr. Coats has disclosed receiving personal fees from Actimed, AstraZeneca, Faraday, WL Gore, Menarini, Novartis, Nutricia, Respicardia, Servier, Stealth Peptides, Verona, and Vifor. Dr. Doener has recently disclosed receiving grants and personal fees from Vifor, Pfizer, Boehringer Ingelheim, Sphingotec, ZS Pharma, Bayer, and Medtronic.
A version of this article originally appeared on Medscape.com.
FROM ESC HEART FAILURE 2020
Cortisol levels on COVID-19 admission may be a marker of severity
Patients with COVID-19 who have high levels of the steroid hormone cortisol on admission to hospital have a substantially increased risk of dying, U.K. researchers have discovered.
Waljit S. Dhillo, MBBS, PhD, head of the division of diabetes, endocrinology and metabolism at Imperial College London, and colleagues studied 535 patients admitted to major London hospitals. Their article was published online June 18 in Lancet Diabetes & Endocrinology.
“Our analyses show for the first time that patients with COVID-19 mount a marked and appropriate acute cortisol stress response,” said Dr. Dhillo and colleagues.
Moreover, “high cortisol concentrations were associated with increased mortality and a reduced median survival, probably because this is a marker of the severity of illness.”
So measuring cortisol on admission is potentially “another simple marker to use alongside oxygen saturation levels to help us identify which patients need to be admitted immediately, and which may not,” Dr. Dhillo noted in a statement from his institution.
“Having an early indicator of which patients may deteriorate more quickly will help us with providing the best level of care as quickly as possible. In addition, we can also take cortisol levels into account when we are working out how best to treat our patients,” he said.
However, it’s important to note that this means – particularly in the wake of the RECOVERY trial reported last week – that “in the early part of the disease you don’t need steroids,” he said.
In contrast to SARS, no adrenal insufficiency with COVID-19
Cortisol levels when healthy and resting are 100-200 nmol/L and nearly zero when sleeping, the researchers explained.
They decided to examine cortisol levels because, although physiological stress from critical illness normally increases levels of the hormone, the prior coronavirus, severe acute respiratory syndrome coronavirus (SARS-CoV), had the opposite effect and induced cortisol insufficiency in some patients.
“We would have said we’re not quite sure” what effect SARS-CoV-2 is having on cortisol levels, “so that’s why we collected the data,” Dr. Dhillo said in an interview.
The researchers studied patients admitted to three large London teaching hospitals between March 9 and April 22 with a clinical suspicion of SARS-CoV-2 infection. All patients had a standard set of blood tests, including full blood count, creatinine, C-reactive protein, D-dimer, and serum cortisol.
After exclusions, the team assessed 535 patients admitted over the study period who had baseline cortisol measured within 48 hours of admission.
Of these, 403 patients were diagnosed with COVID-19 based on a positive result on real-time polymerase chain reaction testing (88%) or a strong clinical and radiological suspicion, despite a negative test (12%).
In total, 132 (25%) individuals were not diagnosed with COVID-19.
Patients with COVID-19 were a mean age of 66.3 years, and 59.6% were men.
Mean cortisol concentrations in patients with COVID-19 were significantly higher than those not diagnosed with the virus (619 vs 519 nmol/L; P < .0001).
And by May 8, significantly more patients with COVID-19 died than those without (27.8% vs 6.8%; P < .0001).
Doubling of cortisol levels associated with 40% higher mortality
Multivariate analysis taking into account age, presence of comorbidities, and laboratory tests revealed that a doubling of cortisol concentrations among those with COVID-19 was associated with a significant increase in mortality, at a hazard ratio of 1.42 (P = .014).
And patients with COVID-19 whose baseline cortisol level was >744 nmol/L had a median survival of just 15 days, compared with those with a level ≤744 nmol/L, who had a median survival of 36 days (P < .0001).
The team notes that the cortisol stress responses in their patients with COVID-19 ranged up to 3,241 nmol/L, which is “a marked cortisol stress response, perhaps higher than is observed in patients undergoing major surgery.”
Of interest, there was no interaction between cortisol levels and ethnicity in their study; a subsequent analysis of the data stratified by black, Asian, and other minority ethnicities revealed no significant differences.
The team note that their results will need to be reproduced in other populations.
“Any potential role for cortisol measurement at baseline and later during an inpatient stay with COVID-19 as a prognostic biomarker, either by itself or in combination with other biomarkers, will require validation in a prospective study.”
Implications for treatment: Reserve dexamethasone for critically ill
Dr. Dhillo explained that, because their findings indicate that people initially infected with COVID-19 do mount an appropriate stress (cortisol) response, it is important that people properly understand this in the wake of the RECOVERY trial, reported last week.
The trial showed that the widely available steroid dexamethasone significantly reduced mortality among severely ill COVID-19 patients in the intensive care unit when given at a supraphysiologic dose of 6 mg.
But it would be hazardous for anyone to self-medicate with steroids at an early stage of COVID-19 because that would further increase cortisol levels and could suppress the immune system.
“For the average person on the street with COVID-19,” excess steroids will make their symptoms worse, Dr. Dhillo explained, adding this is important to emphasize because dexamethasone, and similar steroids, “are cheap and likely available on the Internet, and so misunderstanding of the RECOVERY trial could have serious implications.”
But once patients are very sick, with “inflammation in their lungs” and are in the intensive care unit, and often on ventilators – which is a very small subgroup of those with COVID-19 – it becomes a very different story, he stressed.
“RECOVERY shows clearly there seems to be a benefit once you need oxygen or are on a ventilator, and that makes sense because [dexamethasone] is going to be an anti-inflammatory,” in this instance when the “lungs are full of water.”
“But in the early days you definitely don’t need it and it could be harmful,” he reiterated.
The study is funded by the U.K. National Institute for Health Research and Medical Research Council. The authors have reported no relevant financial relationships.
This article first appeared on Medscape.com.
Patients with COVID-19 who have high levels of the steroid hormone cortisol on admission to hospital have a substantially increased risk of dying, U.K. researchers have discovered.
Waljit S. Dhillo, MBBS, PhD, head of the division of diabetes, endocrinology and metabolism at Imperial College London, and colleagues studied 535 patients admitted to major London hospitals. Their article was published online June 18 in Lancet Diabetes & Endocrinology.
“Our analyses show for the first time that patients with COVID-19 mount a marked and appropriate acute cortisol stress response,” said Dr. Dhillo and colleagues.
Moreover, “high cortisol concentrations were associated with increased mortality and a reduced median survival, probably because this is a marker of the severity of illness.”
So measuring cortisol on admission is potentially “another simple marker to use alongside oxygen saturation levels to help us identify which patients need to be admitted immediately, and which may not,” Dr. Dhillo noted in a statement from his institution.
“Having an early indicator of which patients may deteriorate more quickly will help us with providing the best level of care as quickly as possible. In addition, we can also take cortisol levels into account when we are working out how best to treat our patients,” he said.
However, it’s important to note that this means – particularly in the wake of the RECOVERY trial reported last week – that “in the early part of the disease you don’t need steroids,” he said.
In contrast to SARS, no adrenal insufficiency with COVID-19
Cortisol levels when healthy and resting are 100-200 nmol/L and nearly zero when sleeping, the researchers explained.
They decided to examine cortisol levels because, although physiological stress from critical illness normally increases levels of the hormone, the prior coronavirus, severe acute respiratory syndrome coronavirus (SARS-CoV), had the opposite effect and induced cortisol insufficiency in some patients.
“We would have said we’re not quite sure” what effect SARS-CoV-2 is having on cortisol levels, “so that’s why we collected the data,” Dr. Dhillo said in an interview.
The researchers studied patients admitted to three large London teaching hospitals between March 9 and April 22 with a clinical suspicion of SARS-CoV-2 infection. All patients had a standard set of blood tests, including full blood count, creatinine, C-reactive protein, D-dimer, and serum cortisol.
After exclusions, the team assessed 535 patients admitted over the study period who had baseline cortisol measured within 48 hours of admission.
Of these, 403 patients were diagnosed with COVID-19 based on a positive result on real-time polymerase chain reaction testing (88%) or a strong clinical and radiological suspicion, despite a negative test (12%).
In total, 132 (25%) individuals were not diagnosed with COVID-19.
Patients with COVID-19 were a mean age of 66.3 years, and 59.6% were men.
Mean cortisol concentrations in patients with COVID-19 were significantly higher than those not diagnosed with the virus (619 vs 519 nmol/L; P < .0001).
And by May 8, significantly more patients with COVID-19 died than those without (27.8% vs 6.8%; P < .0001).
Doubling of cortisol levels associated with 40% higher mortality
Multivariate analysis taking into account age, presence of comorbidities, and laboratory tests revealed that a doubling of cortisol concentrations among those with COVID-19 was associated with a significant increase in mortality, at a hazard ratio of 1.42 (P = .014).
And patients with COVID-19 whose baseline cortisol level was >744 nmol/L had a median survival of just 15 days, compared with those with a level ≤744 nmol/L, who had a median survival of 36 days (P < .0001).
The team notes that the cortisol stress responses in their patients with COVID-19 ranged up to 3,241 nmol/L, which is “a marked cortisol stress response, perhaps higher than is observed in patients undergoing major surgery.”
Of interest, there was no interaction between cortisol levels and ethnicity in their study; a subsequent analysis of the data stratified by black, Asian, and other minority ethnicities revealed no significant differences.
The team note that their results will need to be reproduced in other populations.
“Any potential role for cortisol measurement at baseline and later during an inpatient stay with COVID-19 as a prognostic biomarker, either by itself or in combination with other biomarkers, will require validation in a prospective study.”
Implications for treatment: Reserve dexamethasone for critically ill
Dr. Dhillo explained that, because their findings indicate that people initially infected with COVID-19 do mount an appropriate stress (cortisol) response, it is important that people properly understand this in the wake of the RECOVERY trial, reported last week.
The trial showed that the widely available steroid dexamethasone significantly reduced mortality among severely ill COVID-19 patients in the intensive care unit when given at a supraphysiologic dose of 6 mg.
But it would be hazardous for anyone to self-medicate with steroids at an early stage of COVID-19 because that would further increase cortisol levels and could suppress the immune system.
“For the average person on the street with COVID-19,” excess steroids will make their symptoms worse, Dr. Dhillo explained, adding this is important to emphasize because dexamethasone, and similar steroids, “are cheap and likely available on the Internet, and so misunderstanding of the RECOVERY trial could have serious implications.”
But once patients are very sick, with “inflammation in their lungs” and are in the intensive care unit, and often on ventilators – which is a very small subgroup of those with COVID-19 – it becomes a very different story, he stressed.
“RECOVERY shows clearly there seems to be a benefit once you need oxygen or are on a ventilator, and that makes sense because [dexamethasone] is going to be an anti-inflammatory,” in this instance when the “lungs are full of water.”
“But in the early days you definitely don’t need it and it could be harmful,” he reiterated.
The study is funded by the U.K. National Institute for Health Research and Medical Research Council. The authors have reported no relevant financial relationships.
This article first appeared on Medscape.com.
Patients with COVID-19 who have high levels of the steroid hormone cortisol on admission to hospital have a substantially increased risk of dying, U.K. researchers have discovered.
Waljit S. Dhillo, MBBS, PhD, head of the division of diabetes, endocrinology and metabolism at Imperial College London, and colleagues studied 535 patients admitted to major London hospitals. Their article was published online June 18 in Lancet Diabetes & Endocrinology.
“Our analyses show for the first time that patients with COVID-19 mount a marked and appropriate acute cortisol stress response,” said Dr. Dhillo and colleagues.
Moreover, “high cortisol concentrations were associated with increased mortality and a reduced median survival, probably because this is a marker of the severity of illness.”
So measuring cortisol on admission is potentially “another simple marker to use alongside oxygen saturation levels to help us identify which patients need to be admitted immediately, and which may not,” Dr. Dhillo noted in a statement from his institution.
“Having an early indicator of which patients may deteriorate more quickly will help us with providing the best level of care as quickly as possible. In addition, we can also take cortisol levels into account when we are working out how best to treat our patients,” he said.
However, it’s important to note that this means – particularly in the wake of the RECOVERY trial reported last week – that “in the early part of the disease you don’t need steroids,” he said.
In contrast to SARS, no adrenal insufficiency with COVID-19
Cortisol levels when healthy and resting are 100-200 nmol/L and nearly zero when sleeping, the researchers explained.
They decided to examine cortisol levels because, although physiological stress from critical illness normally increases levels of the hormone, the prior coronavirus, severe acute respiratory syndrome coronavirus (SARS-CoV), had the opposite effect and induced cortisol insufficiency in some patients.
“We would have said we’re not quite sure” what effect SARS-CoV-2 is having on cortisol levels, “so that’s why we collected the data,” Dr. Dhillo said in an interview.
The researchers studied patients admitted to three large London teaching hospitals between March 9 and April 22 with a clinical suspicion of SARS-CoV-2 infection. All patients had a standard set of blood tests, including full blood count, creatinine, C-reactive protein, D-dimer, and serum cortisol.
After exclusions, the team assessed 535 patients admitted over the study period who had baseline cortisol measured within 48 hours of admission.
Of these, 403 patients were diagnosed with COVID-19 based on a positive result on real-time polymerase chain reaction testing (88%) or a strong clinical and radiological suspicion, despite a negative test (12%).
In total, 132 (25%) individuals were not diagnosed with COVID-19.
Patients with COVID-19 were a mean age of 66.3 years, and 59.6% were men.
Mean cortisol concentrations in patients with COVID-19 were significantly higher than those not diagnosed with the virus (619 vs 519 nmol/L; P < .0001).
And by May 8, significantly more patients with COVID-19 died than those without (27.8% vs 6.8%; P < .0001).
Doubling of cortisol levels associated with 40% higher mortality
Multivariate analysis taking into account age, presence of comorbidities, and laboratory tests revealed that a doubling of cortisol concentrations among those with COVID-19 was associated with a significant increase in mortality, at a hazard ratio of 1.42 (P = .014).
And patients with COVID-19 whose baseline cortisol level was >744 nmol/L had a median survival of just 15 days, compared with those with a level ≤744 nmol/L, who had a median survival of 36 days (P < .0001).
The team notes that the cortisol stress responses in their patients with COVID-19 ranged up to 3,241 nmol/L, which is “a marked cortisol stress response, perhaps higher than is observed in patients undergoing major surgery.”
Of interest, there was no interaction between cortisol levels and ethnicity in their study; a subsequent analysis of the data stratified by black, Asian, and other minority ethnicities revealed no significant differences.
The team note that their results will need to be reproduced in other populations.
“Any potential role for cortisol measurement at baseline and later during an inpatient stay with COVID-19 as a prognostic biomarker, either by itself or in combination with other biomarkers, will require validation in a prospective study.”
Implications for treatment: Reserve dexamethasone for critically ill
Dr. Dhillo explained that, because their findings indicate that people initially infected with COVID-19 do mount an appropriate stress (cortisol) response, it is important that people properly understand this in the wake of the RECOVERY trial, reported last week.
The trial showed that the widely available steroid dexamethasone significantly reduced mortality among severely ill COVID-19 patients in the intensive care unit when given at a supraphysiologic dose of 6 mg.
But it would be hazardous for anyone to self-medicate with steroids at an early stage of COVID-19 because that would further increase cortisol levels and could suppress the immune system.
“For the average person on the street with COVID-19,” excess steroids will make their symptoms worse, Dr. Dhillo explained, adding this is important to emphasize because dexamethasone, and similar steroids, “are cheap and likely available on the Internet, and so misunderstanding of the RECOVERY trial could have serious implications.”
But once patients are very sick, with “inflammation in their lungs” and are in the intensive care unit, and often on ventilators – which is a very small subgroup of those with COVID-19 – it becomes a very different story, he stressed.
“RECOVERY shows clearly there seems to be a benefit once you need oxygen or are on a ventilator, and that makes sense because [dexamethasone] is going to be an anti-inflammatory,” in this instance when the “lungs are full of water.”
“But in the early days you definitely don’t need it and it could be harmful,” he reiterated.
The study is funded by the U.K. National Institute for Health Research and Medical Research Council. The authors have reported no relevant financial relationships.
This article first appeared on Medscape.com.
T2D plus heart failure packs a deadly punch
It’s bad news for patients with newly diagnosed type 2 diabetes when they then develop heart failure during the next few years.
Patients with incident type 2 diabetes (T2D) who soon after also had heart failure appear faced a dramatically elevated mortality risk, higher than the incremental risk from any other cardiovascular or renal comorbidity that appeared following diabetes onset, in an analysis of more than 150,000 Danish patients with incident type 2 diabetes during 1998-2015.
The 5-year risk of death in patients who developed heart failure during the first 5 years following an initial diagnosis of T2D was about 48%, about threefold higher than in patients with newly diagnosed T2D who remained free of heart failure or any of the other studied comorbidities, Bochra Zareini, MD, and associates reported in a study published in Circulation: Cardiovascular Quality and Outcomes. The studied patients had no known cardiovascular or renal disease at the time of their first T2D diagnosis.
“Our study reports not only on the absolute 5-year risk” of mortality, “but also takes into consideration when patients developed” a comorbidity. “What is surprising and worrying is the very high risk of death following heart failure and the potential life years lost when compared to T2D patients who do not develop heart failure,” said Dr. Zareini, a cardiologist at Herlev and Gentofte University Hospital in Copenhagen. “The implications of our study are to create awareness and highlight the importance of early detection of heart failure development in patients with T2D.” The results also showed that “heart failure is a common cardiovascular disease” in patients with newly diagnosed T2D, she added in an interview.
The data she and her associates reported came from a retrospective analysis of 153,403 Danish citizens in national health records who received a prescription for an antidiabetes drug for the first time during 1998-2015, excluding patients with a prior diagnosis of heart failure, ischemic heart disease (IHD), stroke, peripheral artery disease (PAD), chronic kidney disease (CKD), or gestational diabetes. They followed these patients for a median of just under 10 years, during which time 45% of the cohort had an incident diagnosis of at least one of these cardiovascular and renal conditions, based on medical-record entries from hospitalization discharges or ambulatory contacts.
Nearly two-thirds of the T2D patients with an incident comorbidity during follow-up had a single new diagnosis, a quarter had two new comorbidities appear during follow-up, and 13% developed at least three new comorbidities.
Heart failure, least common but deadliest comorbidity
The most common of the tracked comorbidities was IHD, which appeared in 8% of the T2D patients within 5 years and in 13% after 10 years. Next most common was stroke, affecting 3% of patients after 5 years and 5% after 10 years. CKD occurred in 2.2% after 5 years and in 4.0% after 10 years, PAD occurred in 2.1% after 5 years and in 3.0% at 10 years, and heart failure occurred in 1.6% at 5 years and in 2.2% after 10 years.
But despite being the least common of the studied comorbidities, heart failure was by far the most deadly, roughly tripling the 5-year mortality rate, compared with T2D patients with no comorbidities, regardless of exactly when it first appeared during the first 5 years after the initial T2D diagnosis. The next most deadly comorbidities were stroke and PAD, which each roughly doubled mortality, compared with the patients who remained free of any studied comorbidity. CKD boosted mortality by 70%-110%, depending on exactly when it appeared during the first 5 years of follow-up, and IHD, while the most frequent comorbidity was also the most benign, increasing mortality by about 30%.
The most deadly combinations of two comorbidities were when heart failure appeared with either CKD or with PAD; each of these combinations boosted mortality by 300%-400% when it occurred during the first few years after a T2D diagnosis.
The findings came from “a very big and unselected patient group of patients, making our results highly generalizable in terms of assessing the prognostic consequences of heart failure,” Dr. Zareini stressed.
Management implications
The dangerous combination of T2D and heart failure has been documented for several years, and prompted a focused statement in 2019 about best practices for managing these patients (Circulation. 2019 Aug 3;140[7]:e294-324). “Heart failure has been known for some time to predict poorer outcomes in patients with T2D. Not much surprising” in the new findings reported by Dr. Zareini and associates, commented Robert H. Eckel, MD, a cardiovascular endocrinologist at the University of Colorado at Denver, Aurora. Heart failure “rarely acts alone, but in combination with other forms of heart or renal disease,” he noted in an interview.
Earlier studies may have “overlooked” heart failure’s importance compared with other comorbidities because they often “only investigated one cardiovascular disease in patients with T2D,” Dr. Zareini noted. In recent years the importance of heart failure occurring in patients with T2D also gained heightened significance because of the growing role of the sodium-glucose cotransporter 2 (SGLT2) inhibitor drug class in treating patients with T2D and the documented ability of these drugs to significantly reduce hospitalizations for heart failure (J Am Coll Cardiol. 2020 Apr 28;75[16]:1956-74). Dr. Zareini and associates put it this way in their report: “Heart failure has in recent years been recognized as an important clinical endpoint ... in patients with T2D, in particular, after the results from randomized, controlled trials of SGLT2 inhibitors showed benefit on cardiovascular death and heart failure hospitalizations.”
Despite this, the new findings “do not address treatment with SGLT2 inhibitors in patients with T2D, nor can we use our data to address which patients should not be treated,” with this drug class, which instead should rely on “current evidence and expert consensus,” she said.
“Guidelines favor SGLT2 inhibitors or [glucagonlike peptide–1] receptor agonists in patients with a history of or high risk for major adverse coronary events,” and SGLT2 inhibitors are also “preferable in patients with renal disease,” Dr. Eckel noted.
Other avenues also exist for minimizing the onset of heart failure and other cardiovascular diseases in patients with T2D, Dr. Zareini said, citing modifiable risks that lead to heart failure that include hypertension, “diabetic cardiomyopathy,” and ISD. “Clinicians must treat all modifiable risk factors in patients with T2D in order to improve prognosis and limit development of cardiovascular and renal disease.”
The study received no commercial funding. Dr. Zareini and Dr. Eckel had no disclosures.
SOURCE: Zareini B et al. Circ Cardiovasc Qual Outcomes. 2020 Jun 23. doi: 10.1161/CIRCOUTCOMES.119.006260.
It’s bad news for patients with newly diagnosed type 2 diabetes when they then develop heart failure during the next few years.
Patients with incident type 2 diabetes (T2D) who soon after also had heart failure appear faced a dramatically elevated mortality risk, higher than the incremental risk from any other cardiovascular or renal comorbidity that appeared following diabetes onset, in an analysis of more than 150,000 Danish patients with incident type 2 diabetes during 1998-2015.
The 5-year risk of death in patients who developed heart failure during the first 5 years following an initial diagnosis of T2D was about 48%, about threefold higher than in patients with newly diagnosed T2D who remained free of heart failure or any of the other studied comorbidities, Bochra Zareini, MD, and associates reported in a study published in Circulation: Cardiovascular Quality and Outcomes. The studied patients had no known cardiovascular or renal disease at the time of their first T2D diagnosis.
“Our study reports not only on the absolute 5-year risk” of mortality, “but also takes into consideration when patients developed” a comorbidity. “What is surprising and worrying is the very high risk of death following heart failure and the potential life years lost when compared to T2D patients who do not develop heart failure,” said Dr. Zareini, a cardiologist at Herlev and Gentofte University Hospital in Copenhagen. “The implications of our study are to create awareness and highlight the importance of early detection of heart failure development in patients with T2D.” The results also showed that “heart failure is a common cardiovascular disease” in patients with newly diagnosed T2D, she added in an interview.
The data she and her associates reported came from a retrospective analysis of 153,403 Danish citizens in national health records who received a prescription for an antidiabetes drug for the first time during 1998-2015, excluding patients with a prior diagnosis of heart failure, ischemic heart disease (IHD), stroke, peripheral artery disease (PAD), chronic kidney disease (CKD), or gestational diabetes. They followed these patients for a median of just under 10 years, during which time 45% of the cohort had an incident diagnosis of at least one of these cardiovascular and renal conditions, based on medical-record entries from hospitalization discharges or ambulatory contacts.
Nearly two-thirds of the T2D patients with an incident comorbidity during follow-up had a single new diagnosis, a quarter had two new comorbidities appear during follow-up, and 13% developed at least three new comorbidities.
Heart failure, least common but deadliest comorbidity
The most common of the tracked comorbidities was IHD, which appeared in 8% of the T2D patients within 5 years and in 13% after 10 years. Next most common was stroke, affecting 3% of patients after 5 years and 5% after 10 years. CKD occurred in 2.2% after 5 years and in 4.0% after 10 years, PAD occurred in 2.1% after 5 years and in 3.0% at 10 years, and heart failure occurred in 1.6% at 5 years and in 2.2% after 10 years.
But despite being the least common of the studied comorbidities, heart failure was by far the most deadly, roughly tripling the 5-year mortality rate, compared with T2D patients with no comorbidities, regardless of exactly when it first appeared during the first 5 years after the initial T2D diagnosis. The next most deadly comorbidities were stroke and PAD, which each roughly doubled mortality, compared with the patients who remained free of any studied comorbidity. CKD boosted mortality by 70%-110%, depending on exactly when it appeared during the first 5 years of follow-up, and IHD, while the most frequent comorbidity was also the most benign, increasing mortality by about 30%.
The most deadly combinations of two comorbidities were when heart failure appeared with either CKD or with PAD; each of these combinations boosted mortality by 300%-400% when it occurred during the first few years after a T2D diagnosis.
The findings came from “a very big and unselected patient group of patients, making our results highly generalizable in terms of assessing the prognostic consequences of heart failure,” Dr. Zareini stressed.
Management implications
The dangerous combination of T2D and heart failure has been documented for several years, and prompted a focused statement in 2019 about best practices for managing these patients (Circulation. 2019 Aug 3;140[7]:e294-324). “Heart failure has been known for some time to predict poorer outcomes in patients with T2D. Not much surprising” in the new findings reported by Dr. Zareini and associates, commented Robert H. Eckel, MD, a cardiovascular endocrinologist at the University of Colorado at Denver, Aurora. Heart failure “rarely acts alone, but in combination with other forms of heart or renal disease,” he noted in an interview.
Earlier studies may have “overlooked” heart failure’s importance compared with other comorbidities because they often “only investigated one cardiovascular disease in patients with T2D,” Dr. Zareini noted. In recent years the importance of heart failure occurring in patients with T2D also gained heightened significance because of the growing role of the sodium-glucose cotransporter 2 (SGLT2) inhibitor drug class in treating patients with T2D and the documented ability of these drugs to significantly reduce hospitalizations for heart failure (J Am Coll Cardiol. 2020 Apr 28;75[16]:1956-74). Dr. Zareini and associates put it this way in their report: “Heart failure has in recent years been recognized as an important clinical endpoint ... in patients with T2D, in particular, after the results from randomized, controlled trials of SGLT2 inhibitors showed benefit on cardiovascular death and heart failure hospitalizations.”
Despite this, the new findings “do not address treatment with SGLT2 inhibitors in patients with T2D, nor can we use our data to address which patients should not be treated,” with this drug class, which instead should rely on “current evidence and expert consensus,” she said.
“Guidelines favor SGLT2 inhibitors or [glucagonlike peptide–1] receptor agonists in patients with a history of or high risk for major adverse coronary events,” and SGLT2 inhibitors are also “preferable in patients with renal disease,” Dr. Eckel noted.
Other avenues also exist for minimizing the onset of heart failure and other cardiovascular diseases in patients with T2D, Dr. Zareini said, citing modifiable risks that lead to heart failure that include hypertension, “diabetic cardiomyopathy,” and ISD. “Clinicians must treat all modifiable risk factors in patients with T2D in order to improve prognosis and limit development of cardiovascular and renal disease.”
The study received no commercial funding. Dr. Zareini and Dr. Eckel had no disclosures.
SOURCE: Zareini B et al. Circ Cardiovasc Qual Outcomes. 2020 Jun 23. doi: 10.1161/CIRCOUTCOMES.119.006260.
It’s bad news for patients with newly diagnosed type 2 diabetes when they then develop heart failure during the next few years.
Patients with incident type 2 diabetes (T2D) who soon after also had heart failure appear faced a dramatically elevated mortality risk, higher than the incremental risk from any other cardiovascular or renal comorbidity that appeared following diabetes onset, in an analysis of more than 150,000 Danish patients with incident type 2 diabetes during 1998-2015.
The 5-year risk of death in patients who developed heart failure during the first 5 years following an initial diagnosis of T2D was about 48%, about threefold higher than in patients with newly diagnosed T2D who remained free of heart failure or any of the other studied comorbidities, Bochra Zareini, MD, and associates reported in a study published in Circulation: Cardiovascular Quality and Outcomes. The studied patients had no known cardiovascular or renal disease at the time of their first T2D diagnosis.
“Our study reports not only on the absolute 5-year risk” of mortality, “but also takes into consideration when patients developed” a comorbidity. “What is surprising and worrying is the very high risk of death following heart failure and the potential life years lost when compared to T2D patients who do not develop heart failure,” said Dr. Zareini, a cardiologist at Herlev and Gentofte University Hospital in Copenhagen. “The implications of our study are to create awareness and highlight the importance of early detection of heart failure development in patients with T2D.” The results also showed that “heart failure is a common cardiovascular disease” in patients with newly diagnosed T2D, she added in an interview.
The data she and her associates reported came from a retrospective analysis of 153,403 Danish citizens in national health records who received a prescription for an antidiabetes drug for the first time during 1998-2015, excluding patients with a prior diagnosis of heart failure, ischemic heart disease (IHD), stroke, peripheral artery disease (PAD), chronic kidney disease (CKD), or gestational diabetes. They followed these patients for a median of just under 10 years, during which time 45% of the cohort had an incident diagnosis of at least one of these cardiovascular and renal conditions, based on medical-record entries from hospitalization discharges or ambulatory contacts.
Nearly two-thirds of the T2D patients with an incident comorbidity during follow-up had a single new diagnosis, a quarter had two new comorbidities appear during follow-up, and 13% developed at least three new comorbidities.
Heart failure, least common but deadliest comorbidity
The most common of the tracked comorbidities was IHD, which appeared in 8% of the T2D patients within 5 years and in 13% after 10 years. Next most common was stroke, affecting 3% of patients after 5 years and 5% after 10 years. CKD occurred in 2.2% after 5 years and in 4.0% after 10 years, PAD occurred in 2.1% after 5 years and in 3.0% at 10 years, and heart failure occurred in 1.6% at 5 years and in 2.2% after 10 years.
But despite being the least common of the studied comorbidities, heart failure was by far the most deadly, roughly tripling the 5-year mortality rate, compared with T2D patients with no comorbidities, regardless of exactly when it first appeared during the first 5 years after the initial T2D diagnosis. The next most deadly comorbidities were stroke and PAD, which each roughly doubled mortality, compared with the patients who remained free of any studied comorbidity. CKD boosted mortality by 70%-110%, depending on exactly when it appeared during the first 5 years of follow-up, and IHD, while the most frequent comorbidity was also the most benign, increasing mortality by about 30%.
The most deadly combinations of two comorbidities were when heart failure appeared with either CKD or with PAD; each of these combinations boosted mortality by 300%-400% when it occurred during the first few years after a T2D diagnosis.
The findings came from “a very big and unselected patient group of patients, making our results highly generalizable in terms of assessing the prognostic consequences of heart failure,” Dr. Zareini stressed.
Management implications
The dangerous combination of T2D and heart failure has been documented for several years, and prompted a focused statement in 2019 about best practices for managing these patients (Circulation. 2019 Aug 3;140[7]:e294-324). “Heart failure has been known for some time to predict poorer outcomes in patients with T2D. Not much surprising” in the new findings reported by Dr. Zareini and associates, commented Robert H. Eckel, MD, a cardiovascular endocrinologist at the University of Colorado at Denver, Aurora. Heart failure “rarely acts alone, but in combination with other forms of heart or renal disease,” he noted in an interview.
Earlier studies may have “overlooked” heart failure’s importance compared with other comorbidities because they often “only investigated one cardiovascular disease in patients with T2D,” Dr. Zareini noted. In recent years the importance of heart failure occurring in patients with T2D also gained heightened significance because of the growing role of the sodium-glucose cotransporter 2 (SGLT2) inhibitor drug class in treating patients with T2D and the documented ability of these drugs to significantly reduce hospitalizations for heart failure (J Am Coll Cardiol. 2020 Apr 28;75[16]:1956-74). Dr. Zareini and associates put it this way in their report: “Heart failure has in recent years been recognized as an important clinical endpoint ... in patients with T2D, in particular, after the results from randomized, controlled trials of SGLT2 inhibitors showed benefit on cardiovascular death and heart failure hospitalizations.”
Despite this, the new findings “do not address treatment with SGLT2 inhibitors in patients with T2D, nor can we use our data to address which patients should not be treated,” with this drug class, which instead should rely on “current evidence and expert consensus,” she said.
“Guidelines favor SGLT2 inhibitors or [glucagonlike peptide–1] receptor agonists in patients with a history of or high risk for major adverse coronary events,” and SGLT2 inhibitors are also “preferable in patients with renal disease,” Dr. Eckel noted.
Other avenues also exist for minimizing the onset of heart failure and other cardiovascular diseases in patients with T2D, Dr. Zareini said, citing modifiable risks that lead to heart failure that include hypertension, “diabetic cardiomyopathy,” and ISD. “Clinicians must treat all modifiable risk factors in patients with T2D in order to improve prognosis and limit development of cardiovascular and renal disease.”
The study received no commercial funding. Dr. Zareini and Dr. Eckel had no disclosures.
SOURCE: Zareini B et al. Circ Cardiovasc Qual Outcomes. 2020 Jun 23. doi: 10.1161/CIRCOUTCOMES.119.006260.
FROM CIRCULATION: CARDIOVASCULAR QUALITY AND OUTCOMES
Where does dexamethasone fit in with diabetic ketoacidosis in COVID-19?
A new article in the Journal of Clinical Endocrinology & Metabolism (JCEM) addresses unique concerns and considerations regarding diabetic ketoacidosis (DKA) in the setting of COVID-19.
Corresponding author Marie E. McDonnell, MD, director of the diabetes program at Brigham and Women’s Hospital, Boston, Massachusetts, discussed the recommendations with Medscape Medical News and also spoke about the news this week that the corticosteroid dexamethasone reduced death rates in severely ill patients with COVID-19.
The full JCEM article, by lead author Nadine E. Palermo, DO, Division of Endocrinology, Diabetes, and Hypertension, also at Brigham and Women’s Hospital, covers DKA diagnosis and triage, and emphasizes that usual hospital protocols for DKA management may need to be adjusted during COVID-19 to help preserve personal protective equipment and ICU beds.
“Hospitals and clinicians need to be able to quickly identify and manage DKA in COVID patients to save lives. This involves determining the options for management, including when less intensive subcutaneous insulin is indicated, and understanding how to guide patients on avoiding this serious complication,” McDonnell said in an Endocrine Society statement.
What about dexamethasone for severe COVID-19 in diabetes?
The new article briefly touches on the fact that upward adjustments to intensive intravenous insulin therapy for DKA may be necessary in patients with COVID-19 who are receiving concomitant corticosteroids or vasopressors.
But it was written prior to the June 16 announcement of the “RECOVERY” trial results with dexamethasone. The UK National Health Service immediately approved the drug’s use in the COVID-19 setting, despite the fact that there has been no published article on the findings yet.
McDonnell told Medscape Medical News that she would need to see formal results to better understand exactly which patients were studied and which ones benefited.
“The peer review will be critical. It looks as if it only benefits people who need respiratory support, but I want to understand that in much more detail,” she said. “If they all had acute respiratory distress syndrome [ARDS],” that’s different.
“There are already some data supporting steroid use in ARDS,” she noted, but added that not all of it suggests benefit.
She pointed to one of several studies now showing that diabetes, and hyperglycemia among people without a prior diabetes diagnosis, are both strong predictors of mortality in hospitalized patients with COVID-19.
“There was a very clear relationship between hyperglycemia and outcomes. We really shouldn’t put people at risk until we have clear data,” she said.
If, once the data are reviewed and appropriate dexamethasone becomes an established treatment for severe COVID-19, hyperglycemia would be a concern among all patients, not just those with previously diagnosed diabetes, she noted.
“We know a good number of people with prediabetes develop hyperglycemia when put on steroids. They can push people over the edge. We’re not going to miss anybody, but treating steroid-induced hyperglycemia is really hard,” McDonnell explained.
She also recommended 2014 guidance from Diabetes UK and the Association of British Clinical Diabetologists, which addresses management of inpatient steroid-induced DKA in patients with and without pre-existing diabetes.
Another major concern, she said, is “patients trying to get dexamethasone when they start to get sick” because this is not the right population to use this agent.
“We worry about people who do not need this drug. If they have diabetes, they put themselves at risk of hyperglycemia, which then increases the risk of severe COVID-19. And then they’re also putting themselves at risk of DKA. It would just be bad medicine,” she said.
Managing DKA in the face of COVID-19: Flexibility is key
In the JCEM article, Palermo and colleagues emphasize that the usual hospital protocols for DKA management may need to be adjusted during COVID-19 in the interest of reducing transmission risk and preserving scare resources.
They provide evidence for alternative treatment strategies, such as the use of subcutaneous rather than intravenous insulin when appropriate.
“We wanted to outline when exactly you should consider nonintensive management strategies for DKA,” McDonnell further explained to Medscape Medical News.
“That would include those with mild or some with moderate DKA. ... The idea is to remind our colleagues about that because hospitals tend to operate on a protocol-driven algorithmic methodology, they can forget to step off the usual care pathway even if evidence supports that,” she said.
But on the other hand, she also said that, in some very complex or severely ill patients with COVID-19, classical intravenous insulin therapy makes the most sense even if their DKA is mild.
The outpatient setting: Prevention and preparation
The new article also addresses several concerns regarding DKA prevention in the outpatient setting.
As with other guidelines, it includes a reminder that patients with diabetes should be advised to discontinue sodium-glucose cotransporter 2 (SGLT2) inhibitors if they become ill with COVID-19, especially if they’re not eating or drinking normally, because they raise the risk for DKA.
Also, for patients with type 1 diabetes, particularly those with a history of repeated DKA, “this is the time to make sure we reach out to patients to refill their insulin prescriptions and address issues related to cost and other access difficulties,” McDonnell said.
The authors also emphasize that insulin starts and education should not be postponed during the pandemic. “Patients identified as meeting criteria to start insulin should be referred for urgent education, either in person or, whenever possible and practical, via video teleconferencing,” they urge.
McDonnell has reported receiving research funding from Novo Nordisk. The other two authors have reported no relevant financial relationships.
This article first appeared on Medscape.com.
A new article in the Journal of Clinical Endocrinology & Metabolism (JCEM) addresses unique concerns and considerations regarding diabetic ketoacidosis (DKA) in the setting of COVID-19.
Corresponding author Marie E. McDonnell, MD, director of the diabetes program at Brigham and Women’s Hospital, Boston, Massachusetts, discussed the recommendations with Medscape Medical News and also spoke about the news this week that the corticosteroid dexamethasone reduced death rates in severely ill patients with COVID-19.
The full JCEM article, by lead author Nadine E. Palermo, DO, Division of Endocrinology, Diabetes, and Hypertension, also at Brigham and Women’s Hospital, covers DKA diagnosis and triage, and emphasizes that usual hospital protocols for DKA management may need to be adjusted during COVID-19 to help preserve personal protective equipment and ICU beds.
“Hospitals and clinicians need to be able to quickly identify and manage DKA in COVID patients to save lives. This involves determining the options for management, including when less intensive subcutaneous insulin is indicated, and understanding how to guide patients on avoiding this serious complication,” McDonnell said in an Endocrine Society statement.
What about dexamethasone for severe COVID-19 in diabetes?
The new article briefly touches on the fact that upward adjustments to intensive intravenous insulin therapy for DKA may be necessary in patients with COVID-19 who are receiving concomitant corticosteroids or vasopressors.
But it was written prior to the June 16 announcement of the “RECOVERY” trial results with dexamethasone. The UK National Health Service immediately approved the drug’s use in the COVID-19 setting, despite the fact that there has been no published article on the findings yet.
McDonnell told Medscape Medical News that she would need to see formal results to better understand exactly which patients were studied and which ones benefited.
“The peer review will be critical. It looks as if it only benefits people who need respiratory support, but I want to understand that in much more detail,” she said. “If they all had acute respiratory distress syndrome [ARDS],” that’s different.
“There are already some data supporting steroid use in ARDS,” she noted, but added that not all of it suggests benefit.
She pointed to one of several studies now showing that diabetes, and hyperglycemia among people without a prior diabetes diagnosis, are both strong predictors of mortality in hospitalized patients with COVID-19.
“There was a very clear relationship between hyperglycemia and outcomes. We really shouldn’t put people at risk until we have clear data,” she said.
If, once the data are reviewed and appropriate dexamethasone becomes an established treatment for severe COVID-19, hyperglycemia would be a concern among all patients, not just those with previously diagnosed diabetes, she noted.
“We know a good number of people with prediabetes develop hyperglycemia when put on steroids. They can push people over the edge. We’re not going to miss anybody, but treating steroid-induced hyperglycemia is really hard,” McDonnell explained.
She also recommended 2014 guidance from Diabetes UK and the Association of British Clinical Diabetologists, which addresses management of inpatient steroid-induced DKA in patients with and without pre-existing diabetes.
Another major concern, she said, is “patients trying to get dexamethasone when they start to get sick” because this is not the right population to use this agent.
“We worry about people who do not need this drug. If they have diabetes, they put themselves at risk of hyperglycemia, which then increases the risk of severe COVID-19. And then they’re also putting themselves at risk of DKA. It would just be bad medicine,” she said.
Managing DKA in the face of COVID-19: Flexibility is key
In the JCEM article, Palermo and colleagues emphasize that the usual hospital protocols for DKA management may need to be adjusted during COVID-19 in the interest of reducing transmission risk and preserving scare resources.
They provide evidence for alternative treatment strategies, such as the use of subcutaneous rather than intravenous insulin when appropriate.
“We wanted to outline when exactly you should consider nonintensive management strategies for DKA,” McDonnell further explained to Medscape Medical News.
“That would include those with mild or some with moderate DKA. ... The idea is to remind our colleagues about that because hospitals tend to operate on a protocol-driven algorithmic methodology, they can forget to step off the usual care pathway even if evidence supports that,” she said.
But on the other hand, she also said that, in some very complex or severely ill patients with COVID-19, classical intravenous insulin therapy makes the most sense even if their DKA is mild.
The outpatient setting: Prevention and preparation
The new article also addresses several concerns regarding DKA prevention in the outpatient setting.
As with other guidelines, it includes a reminder that patients with diabetes should be advised to discontinue sodium-glucose cotransporter 2 (SGLT2) inhibitors if they become ill with COVID-19, especially if they’re not eating or drinking normally, because they raise the risk for DKA.
Also, for patients with type 1 diabetes, particularly those with a history of repeated DKA, “this is the time to make sure we reach out to patients to refill their insulin prescriptions and address issues related to cost and other access difficulties,” McDonnell said.
The authors also emphasize that insulin starts and education should not be postponed during the pandemic. “Patients identified as meeting criteria to start insulin should be referred for urgent education, either in person or, whenever possible and practical, via video teleconferencing,” they urge.
McDonnell has reported receiving research funding from Novo Nordisk. The other two authors have reported no relevant financial relationships.
This article first appeared on Medscape.com.
A new article in the Journal of Clinical Endocrinology & Metabolism (JCEM) addresses unique concerns and considerations regarding diabetic ketoacidosis (DKA) in the setting of COVID-19.
Corresponding author Marie E. McDonnell, MD, director of the diabetes program at Brigham and Women’s Hospital, Boston, Massachusetts, discussed the recommendations with Medscape Medical News and also spoke about the news this week that the corticosteroid dexamethasone reduced death rates in severely ill patients with COVID-19.
The full JCEM article, by lead author Nadine E. Palermo, DO, Division of Endocrinology, Diabetes, and Hypertension, also at Brigham and Women’s Hospital, covers DKA diagnosis and triage, and emphasizes that usual hospital protocols for DKA management may need to be adjusted during COVID-19 to help preserve personal protective equipment and ICU beds.
“Hospitals and clinicians need to be able to quickly identify and manage DKA in COVID patients to save lives. This involves determining the options for management, including when less intensive subcutaneous insulin is indicated, and understanding how to guide patients on avoiding this serious complication,” McDonnell said in an Endocrine Society statement.
What about dexamethasone for severe COVID-19 in diabetes?
The new article briefly touches on the fact that upward adjustments to intensive intravenous insulin therapy for DKA may be necessary in patients with COVID-19 who are receiving concomitant corticosteroids or vasopressors.
But it was written prior to the June 16 announcement of the “RECOVERY” trial results with dexamethasone. The UK National Health Service immediately approved the drug’s use in the COVID-19 setting, despite the fact that there has been no published article on the findings yet.
McDonnell told Medscape Medical News that she would need to see formal results to better understand exactly which patients were studied and which ones benefited.
“The peer review will be critical. It looks as if it only benefits people who need respiratory support, but I want to understand that in much more detail,” she said. “If they all had acute respiratory distress syndrome [ARDS],” that’s different.
“There are already some data supporting steroid use in ARDS,” she noted, but added that not all of it suggests benefit.
She pointed to one of several studies now showing that diabetes, and hyperglycemia among people without a prior diabetes diagnosis, are both strong predictors of mortality in hospitalized patients with COVID-19.
“There was a very clear relationship between hyperglycemia and outcomes. We really shouldn’t put people at risk until we have clear data,” she said.
If, once the data are reviewed and appropriate dexamethasone becomes an established treatment for severe COVID-19, hyperglycemia would be a concern among all patients, not just those with previously diagnosed diabetes, she noted.
“We know a good number of people with prediabetes develop hyperglycemia when put on steroids. They can push people over the edge. We’re not going to miss anybody, but treating steroid-induced hyperglycemia is really hard,” McDonnell explained.
She also recommended 2014 guidance from Diabetes UK and the Association of British Clinical Diabetologists, which addresses management of inpatient steroid-induced DKA in patients with and without pre-existing diabetes.
Another major concern, she said, is “patients trying to get dexamethasone when they start to get sick” because this is not the right population to use this agent.
“We worry about people who do not need this drug. If they have diabetes, they put themselves at risk of hyperglycemia, which then increases the risk of severe COVID-19. And then they’re also putting themselves at risk of DKA. It would just be bad medicine,” she said.
Managing DKA in the face of COVID-19: Flexibility is key
In the JCEM article, Palermo and colleagues emphasize that the usual hospital protocols for DKA management may need to be adjusted during COVID-19 in the interest of reducing transmission risk and preserving scare resources.
They provide evidence for alternative treatment strategies, such as the use of subcutaneous rather than intravenous insulin when appropriate.
“We wanted to outline when exactly you should consider nonintensive management strategies for DKA,” McDonnell further explained to Medscape Medical News.
“That would include those with mild or some with moderate DKA. ... The idea is to remind our colleagues about that because hospitals tend to operate on a protocol-driven algorithmic methodology, they can forget to step off the usual care pathway even if evidence supports that,” she said.
But on the other hand, she also said that, in some very complex or severely ill patients with COVID-19, classical intravenous insulin therapy makes the most sense even if their DKA is mild.
The outpatient setting: Prevention and preparation
The new article also addresses several concerns regarding DKA prevention in the outpatient setting.
As with other guidelines, it includes a reminder that patients with diabetes should be advised to discontinue sodium-glucose cotransporter 2 (SGLT2) inhibitors if they become ill with COVID-19, especially if they’re not eating or drinking normally, because they raise the risk for DKA.
Also, for patients with type 1 diabetes, particularly those with a history of repeated DKA, “this is the time to make sure we reach out to patients to refill their insulin prescriptions and address issues related to cost and other access difficulties,” McDonnell said.
The authors also emphasize that insulin starts and education should not be postponed during the pandemic. “Patients identified as meeting criteria to start insulin should be referred for urgent education, either in person or, whenever possible and practical, via video teleconferencing,” they urge.
McDonnell has reported receiving research funding from Novo Nordisk. The other two authors have reported no relevant financial relationships.
This article first appeared on Medscape.com.
Increased hypothyroidism risk seen in young men with HS
Anna Figueiredo, MD, declared at the virtual annual meeting of the American Academy of Dermatology.
The surprise about this finding from a large retrospective case-control study stems from the fact that the elevated risk for hypothyroidism didn’t also extend to younger women with hidradenitis suppurativa (HS) nor to patients older than 40 years of either gender, explained Dr. Figueiredo of the department of dermatology at Northwestern University, Chicago.
She presented a retrospective case-control study based on information extracted from a medical records database of more than 8 million Midwestern adults. Among nearly 141,000 dermatology patients with follow-up in the database for at least 1 year, there were 405 HS patients aged 18-40 years and 327 aged 41-89.
In an age-matched comparison with the dermatology patients without HS, the younger HS cohort was at a significant 1.52-fold increased risk for comorbid hypothyroidism. Upon further stratification by sex, only the younger men with HS were at increased risk. Those patients were at 3.95-fold greater risk for having a diagnosis of hypothyroidism than were age-matched younger male dermatology patients.
Both younger and older HS patients were at numerically increased risk for being diagnosed with hyperthyroidism; however, this difference didn’t approach statistical significance because there were so few cases: a total of just eight in the HS population across the full age spectrum.
Hidradenitis suppurativa is a chronic inflammatory disease with an estimated prevalence of up to 4% in the United States. Growing evidence suggests it is an immune-mediated disorder because the tumor necrosis factor inhibitor adalimumab (Humira) has been approved for treatment of HS.
Thyroid disease is also often autoimmune-mediated, but its relationship with HS hasn’t been extensively examined. A recent meta-analysis of five case-control studies concluded that HS was associated with a 1.36-fold increased risk of thyroid disease; however, the Nepalese investigators didn’t distinguish between hypo- and hyperthyroidism (J Am Acad Dermatol. 2020 Feb;82[2]:491-3).
Dr. Figueiredo reported having no financial conflicts regarding her study, which was without commercial support.
Anna Figueiredo, MD, declared at the virtual annual meeting of the American Academy of Dermatology.
The surprise about this finding from a large retrospective case-control study stems from the fact that the elevated risk for hypothyroidism didn’t also extend to younger women with hidradenitis suppurativa (HS) nor to patients older than 40 years of either gender, explained Dr. Figueiredo of the department of dermatology at Northwestern University, Chicago.
She presented a retrospective case-control study based on information extracted from a medical records database of more than 8 million Midwestern adults. Among nearly 141,000 dermatology patients with follow-up in the database for at least 1 year, there were 405 HS patients aged 18-40 years and 327 aged 41-89.
In an age-matched comparison with the dermatology patients without HS, the younger HS cohort was at a significant 1.52-fold increased risk for comorbid hypothyroidism. Upon further stratification by sex, only the younger men with HS were at increased risk. Those patients were at 3.95-fold greater risk for having a diagnosis of hypothyroidism than were age-matched younger male dermatology patients.
Both younger and older HS patients were at numerically increased risk for being diagnosed with hyperthyroidism; however, this difference didn’t approach statistical significance because there were so few cases: a total of just eight in the HS population across the full age spectrum.
Hidradenitis suppurativa is a chronic inflammatory disease with an estimated prevalence of up to 4% in the United States. Growing evidence suggests it is an immune-mediated disorder because the tumor necrosis factor inhibitor adalimumab (Humira) has been approved for treatment of HS.
Thyroid disease is also often autoimmune-mediated, but its relationship with HS hasn’t been extensively examined. A recent meta-analysis of five case-control studies concluded that HS was associated with a 1.36-fold increased risk of thyroid disease; however, the Nepalese investigators didn’t distinguish between hypo- and hyperthyroidism (J Am Acad Dermatol. 2020 Feb;82[2]:491-3).
Dr. Figueiredo reported having no financial conflicts regarding her study, which was without commercial support.
Anna Figueiredo, MD, declared at the virtual annual meeting of the American Academy of Dermatology.
The surprise about this finding from a large retrospective case-control study stems from the fact that the elevated risk for hypothyroidism didn’t also extend to younger women with hidradenitis suppurativa (HS) nor to patients older than 40 years of either gender, explained Dr. Figueiredo of the department of dermatology at Northwestern University, Chicago.
She presented a retrospective case-control study based on information extracted from a medical records database of more than 8 million Midwestern adults. Among nearly 141,000 dermatology patients with follow-up in the database for at least 1 year, there were 405 HS patients aged 18-40 years and 327 aged 41-89.
In an age-matched comparison with the dermatology patients without HS, the younger HS cohort was at a significant 1.52-fold increased risk for comorbid hypothyroidism. Upon further stratification by sex, only the younger men with HS were at increased risk. Those patients were at 3.95-fold greater risk for having a diagnosis of hypothyroidism than were age-matched younger male dermatology patients.
Both younger and older HS patients were at numerically increased risk for being diagnosed with hyperthyroidism; however, this difference didn’t approach statistical significance because there were so few cases: a total of just eight in the HS population across the full age spectrum.
Hidradenitis suppurativa is a chronic inflammatory disease with an estimated prevalence of up to 4% in the United States. Growing evidence suggests it is an immune-mediated disorder because the tumor necrosis factor inhibitor adalimumab (Humira) has been approved for treatment of HS.
Thyroid disease is also often autoimmune-mediated, but its relationship with HS hasn’t been extensively examined. A recent meta-analysis of five case-control studies concluded that HS was associated with a 1.36-fold increased risk of thyroid disease; however, the Nepalese investigators didn’t distinguish between hypo- and hyperthyroidism (J Am Acad Dermatol. 2020 Feb;82[2]:491-3).
Dr. Figueiredo reported having no financial conflicts regarding her study, which was without commercial support.
FROM AAD 20
DAPA-HF: Dapagliflozin slows T2D onset in heart failure patients
Dapagliflozin treatment of patients with heart failure but without diabetes in the DAPA-HF trial led to a one-third cut in the relative incidence of new-onset diabetes over a median follow-up of 18 months in a prespecified analysis from the multicenter trial that included 2,605 heart failure patients without diabetes at baseline.
The findings represented the first evidence that a drug from dapagliflozin’s class, the sodium-glucose cotransporter 2 (SGLT2) inhibitors, could prevent or slow the onset of type 2 diabetes. It represents “an additional benefit” that dapagliflozin (Farxiga) offers to patients with heart failure with reduced ejection fraction (HFrEF) like those enrolled in the DAPA-HF trial, Silvio E. Inzucchi, MD, said at the virtual annual scientific sessions of the American Diabetes Association. DAPA-HF had previously proved that treatment with this drug significantly reduced the study’s primary endpoint of cardiovascular death or heart failure worsening.
During 18 months of follow-up, 7.1% of patients in the placebo arm developed type 2 diabetes, compared with 4.9% in those who received dapagliflozin, a 2.2% absolute difference and a 32% relative risk reduction that was statistically significant for this prespecified but “exploratory” endpoint, reported Dr. Inzucchi, an endocrinologist and professor of medicine at Yale University, New Haven, Conn.
For this analysis, a hemoglobin A1c level of at least 6.5% measured in two consecutive assessments was the criterion for diagnosing incident diabetes. The 2,605 enrolled patients without diabetes in the DAPA-HF trial represented 55% of the entire trial cohort of 4,744 patients with HFrEF.
The 32% relative risk reduction for incident diabetes was primarily relevant to enrolled patients with prediabetes at entry, who constituted 67% of the enrolled cohort based on the usual definition of prediabetes, an A1c of 5.7%-6.4%.
Among all 157 (6%) of the DAPA-HF patients who developed diabetes during the trial, 150 (96%) occurred in patients with prediabetes by the usual definition; 136 of the incident cases (87%) had prediabetes by a more stringent criterion of an A1c of 6.0%-6.4%.
To put the preventive efficacy of dapagliflozin into more context, Dr. Inzucchi cited the 31% relative protection rate exerted by metformin in the Diabetes Prevention Program study (N Engl J Med. 2002 Feb 7;346[6]:393-403).
The findings showed that “dapagliflozin is the first medication demonstrated to reduce both incident type 2 diabetes and mortality in a single trial,” as well as the first agent from the SGLT2 inhibitor class to show a diabetes prevention effect, Dr. Inzucchi noted. Patients with both heart failure and diabetes are known to have a substantially increased mortality risk, compared with patients with just one of these diseases, and the potent risk posed by the confluence of both was confirmed in the results Dr. Inzucchi reported.
The 157 HFrEF patients in the trial who developed diabetes had a statistically significant 70% increased incidence of all-cause mortality during the trial’s follow-up, compared with similar HFrEF patients who remained free from a diabetes diagnosis, and they also had a significant 77% relative increase in their incidence of cardiovascular death. This analysis failed to show that incident diabetes had a significant impact on hospitalizations for heart failure coupled with cardiovascular death, another endpoint of the trial.
“This is a tremendously important analysis. We recognize that diabetes is an important factor that can forecast heart failure risk, even over relatively short follow-up. A drug that targets both diseases can be quite beneficial,” commented Muthiah Vaduganathan, MD, a cardiologist at Brigham and Women’s Hospital in Boston.
The impact of dapagliflozin on average A1c levels during the DAPA-HF trial was minimal, reducing levels by an average of 0.04% among those who entered with prediabetes and by 0.05% among the other patients. This suggests that the mechanisms by which dapagliflozin reduced incident diabetes was by routes that did not involve simply reducing hyperglycemia, and the observed decrease in incident diabetes was not apparently caused by “masking” of hyperglycemia by dapagliflozin, said Dr. Inzucchi.
One possibility is that dapagliflozin, which also improved quality of life and reduced hospitalizations in the DAPA-HF trial, led to improved function and mobility among patients that had beneficial effects on their insulin sensitivity, Dr. Vaduganathan speculated in an interview.
The new finding of dapagliflozin’s benefit “is great news,” commented Yehuda Handelsman, MD, an endocrinologist and diabetes specialist who is medical director of the Metabolic Institute of America in Tarzana, Calif. “It’s an impressive and important result, and another reason to use dapagliflozin in patients with HFrEF, a group of patients whom you want to prevent from having worse outcomes” by developing diabetes.
The DAPA-HF (Dapagliflozin and Prevention of Adverse Outcomes in Heart Failure) trial enrolled HFrEF patients at 410 centers in 20 countries during February 2017–August 2018. The study’s primary endpoint was the composite incidence of cardiovascular death or worsening heart failure, which occurred in 16.3% of patients randomized to receive dapagliflozin and in 21.2% of control patients on standard care but on placebo instead of the study drug, a statistically significant relative risk reduction of 26% (N Engl J Med. 2019 Nov 21;381[21]:1995-2008). In the 2,605-patient subgroup without type 2 diabetes at baseline the primary endpoint fell by a statistically significant 27% with dapagliflozin treatment, the first time an SGLT2 inhibitor drug was shown effective for reducing this endpoint in patients with HFrEF but without diabetes. DAPA-HF did not enroll any patients with type 1 diabetes.
DAPA-HF was sponsored by AstraZeneca, the company that markets dapagliflozin (Farxiga). Dr. Inzucchi has been a consultant to AstraZeneca and to Abbott, Boehringer Ingelheim, Merck, Novo Nordisk, Sanofi/Lexicon, and vTv Therapeutics. Dr. Vaduganathan has been an adviser to AstraZeneca and to Amgen, Baxter, Bayer, Boehringer Ingelheim, Cytokinetics, and Relypsa. Dr. Handelsman has been a consultant to several drug companies including AstraZeneca.
SOURCE: Inzucchi SE et al. ADA 2020, abstract 271-OR.
Dapagliflozin treatment of patients with heart failure but without diabetes in the DAPA-HF trial led to a one-third cut in the relative incidence of new-onset diabetes over a median follow-up of 18 months in a prespecified analysis from the multicenter trial that included 2,605 heart failure patients without diabetes at baseline.
The findings represented the first evidence that a drug from dapagliflozin’s class, the sodium-glucose cotransporter 2 (SGLT2) inhibitors, could prevent or slow the onset of type 2 diabetes. It represents “an additional benefit” that dapagliflozin (Farxiga) offers to patients with heart failure with reduced ejection fraction (HFrEF) like those enrolled in the DAPA-HF trial, Silvio E. Inzucchi, MD, said at the virtual annual scientific sessions of the American Diabetes Association. DAPA-HF had previously proved that treatment with this drug significantly reduced the study’s primary endpoint of cardiovascular death or heart failure worsening.
During 18 months of follow-up, 7.1% of patients in the placebo arm developed type 2 diabetes, compared with 4.9% in those who received dapagliflozin, a 2.2% absolute difference and a 32% relative risk reduction that was statistically significant for this prespecified but “exploratory” endpoint, reported Dr. Inzucchi, an endocrinologist and professor of medicine at Yale University, New Haven, Conn.
For this analysis, a hemoglobin A1c level of at least 6.5% measured in two consecutive assessments was the criterion for diagnosing incident diabetes. The 2,605 enrolled patients without diabetes in the DAPA-HF trial represented 55% of the entire trial cohort of 4,744 patients with HFrEF.
The 32% relative risk reduction for incident diabetes was primarily relevant to enrolled patients with prediabetes at entry, who constituted 67% of the enrolled cohort based on the usual definition of prediabetes, an A1c of 5.7%-6.4%.
Among all 157 (6%) of the DAPA-HF patients who developed diabetes during the trial, 150 (96%) occurred in patients with prediabetes by the usual definition; 136 of the incident cases (87%) had prediabetes by a more stringent criterion of an A1c of 6.0%-6.4%.
To put the preventive efficacy of dapagliflozin into more context, Dr. Inzucchi cited the 31% relative protection rate exerted by metformin in the Diabetes Prevention Program study (N Engl J Med. 2002 Feb 7;346[6]:393-403).
The findings showed that “dapagliflozin is the first medication demonstrated to reduce both incident type 2 diabetes and mortality in a single trial,” as well as the first agent from the SGLT2 inhibitor class to show a diabetes prevention effect, Dr. Inzucchi noted. Patients with both heart failure and diabetes are known to have a substantially increased mortality risk, compared with patients with just one of these diseases, and the potent risk posed by the confluence of both was confirmed in the results Dr. Inzucchi reported.
The 157 HFrEF patients in the trial who developed diabetes had a statistically significant 70% increased incidence of all-cause mortality during the trial’s follow-up, compared with similar HFrEF patients who remained free from a diabetes diagnosis, and they also had a significant 77% relative increase in their incidence of cardiovascular death. This analysis failed to show that incident diabetes had a significant impact on hospitalizations for heart failure coupled with cardiovascular death, another endpoint of the trial.
“This is a tremendously important analysis. We recognize that diabetes is an important factor that can forecast heart failure risk, even over relatively short follow-up. A drug that targets both diseases can be quite beneficial,” commented Muthiah Vaduganathan, MD, a cardiologist at Brigham and Women’s Hospital in Boston.
The impact of dapagliflozin on average A1c levels during the DAPA-HF trial was minimal, reducing levels by an average of 0.04% among those who entered with prediabetes and by 0.05% among the other patients. This suggests that the mechanisms by which dapagliflozin reduced incident diabetes was by routes that did not involve simply reducing hyperglycemia, and the observed decrease in incident diabetes was not apparently caused by “masking” of hyperglycemia by dapagliflozin, said Dr. Inzucchi.
One possibility is that dapagliflozin, which also improved quality of life and reduced hospitalizations in the DAPA-HF trial, led to improved function and mobility among patients that had beneficial effects on their insulin sensitivity, Dr. Vaduganathan speculated in an interview.
The new finding of dapagliflozin’s benefit “is great news,” commented Yehuda Handelsman, MD, an endocrinologist and diabetes specialist who is medical director of the Metabolic Institute of America in Tarzana, Calif. “It’s an impressive and important result, and another reason to use dapagliflozin in patients with HFrEF, a group of patients whom you want to prevent from having worse outcomes” by developing diabetes.
The DAPA-HF (Dapagliflozin and Prevention of Adverse Outcomes in Heart Failure) trial enrolled HFrEF patients at 410 centers in 20 countries during February 2017–August 2018. The study’s primary endpoint was the composite incidence of cardiovascular death or worsening heart failure, which occurred in 16.3% of patients randomized to receive dapagliflozin and in 21.2% of control patients on standard care but on placebo instead of the study drug, a statistically significant relative risk reduction of 26% (N Engl J Med. 2019 Nov 21;381[21]:1995-2008). In the 2,605-patient subgroup without type 2 diabetes at baseline the primary endpoint fell by a statistically significant 27% with dapagliflozin treatment, the first time an SGLT2 inhibitor drug was shown effective for reducing this endpoint in patients with HFrEF but without diabetes. DAPA-HF did not enroll any patients with type 1 diabetes.
DAPA-HF was sponsored by AstraZeneca, the company that markets dapagliflozin (Farxiga). Dr. Inzucchi has been a consultant to AstraZeneca and to Abbott, Boehringer Ingelheim, Merck, Novo Nordisk, Sanofi/Lexicon, and vTv Therapeutics. Dr. Vaduganathan has been an adviser to AstraZeneca and to Amgen, Baxter, Bayer, Boehringer Ingelheim, Cytokinetics, and Relypsa. Dr. Handelsman has been a consultant to several drug companies including AstraZeneca.
SOURCE: Inzucchi SE et al. ADA 2020, abstract 271-OR.
Dapagliflozin treatment of patients with heart failure but without diabetes in the DAPA-HF trial led to a one-third cut in the relative incidence of new-onset diabetes over a median follow-up of 18 months in a prespecified analysis from the multicenter trial that included 2,605 heart failure patients without diabetes at baseline.
The findings represented the first evidence that a drug from dapagliflozin’s class, the sodium-glucose cotransporter 2 (SGLT2) inhibitors, could prevent or slow the onset of type 2 diabetes. It represents “an additional benefit” that dapagliflozin (Farxiga) offers to patients with heart failure with reduced ejection fraction (HFrEF) like those enrolled in the DAPA-HF trial, Silvio E. Inzucchi, MD, said at the virtual annual scientific sessions of the American Diabetes Association. DAPA-HF had previously proved that treatment with this drug significantly reduced the study’s primary endpoint of cardiovascular death or heart failure worsening.
During 18 months of follow-up, 7.1% of patients in the placebo arm developed type 2 diabetes, compared with 4.9% in those who received dapagliflozin, a 2.2% absolute difference and a 32% relative risk reduction that was statistically significant for this prespecified but “exploratory” endpoint, reported Dr. Inzucchi, an endocrinologist and professor of medicine at Yale University, New Haven, Conn.
For this analysis, a hemoglobin A1c level of at least 6.5% measured in two consecutive assessments was the criterion for diagnosing incident diabetes. The 2,605 enrolled patients without diabetes in the DAPA-HF trial represented 55% of the entire trial cohort of 4,744 patients with HFrEF.
The 32% relative risk reduction for incident diabetes was primarily relevant to enrolled patients with prediabetes at entry, who constituted 67% of the enrolled cohort based on the usual definition of prediabetes, an A1c of 5.7%-6.4%.
Among all 157 (6%) of the DAPA-HF patients who developed diabetes during the trial, 150 (96%) occurred in patients with prediabetes by the usual definition; 136 of the incident cases (87%) had prediabetes by a more stringent criterion of an A1c of 6.0%-6.4%.
To put the preventive efficacy of dapagliflozin into more context, Dr. Inzucchi cited the 31% relative protection rate exerted by metformin in the Diabetes Prevention Program study (N Engl J Med. 2002 Feb 7;346[6]:393-403).
The findings showed that “dapagliflozin is the first medication demonstrated to reduce both incident type 2 diabetes and mortality in a single trial,” as well as the first agent from the SGLT2 inhibitor class to show a diabetes prevention effect, Dr. Inzucchi noted. Patients with both heart failure and diabetes are known to have a substantially increased mortality risk, compared with patients with just one of these diseases, and the potent risk posed by the confluence of both was confirmed in the results Dr. Inzucchi reported.
The 157 HFrEF patients in the trial who developed diabetes had a statistically significant 70% increased incidence of all-cause mortality during the trial’s follow-up, compared with similar HFrEF patients who remained free from a diabetes diagnosis, and they also had a significant 77% relative increase in their incidence of cardiovascular death. This analysis failed to show that incident diabetes had a significant impact on hospitalizations for heart failure coupled with cardiovascular death, another endpoint of the trial.
“This is a tremendously important analysis. We recognize that diabetes is an important factor that can forecast heart failure risk, even over relatively short follow-up. A drug that targets both diseases can be quite beneficial,” commented Muthiah Vaduganathan, MD, a cardiologist at Brigham and Women’s Hospital in Boston.
The impact of dapagliflozin on average A1c levels during the DAPA-HF trial was minimal, reducing levels by an average of 0.04% among those who entered with prediabetes and by 0.05% among the other patients. This suggests that the mechanisms by which dapagliflozin reduced incident diabetes was by routes that did not involve simply reducing hyperglycemia, and the observed decrease in incident diabetes was not apparently caused by “masking” of hyperglycemia by dapagliflozin, said Dr. Inzucchi.
One possibility is that dapagliflozin, which also improved quality of life and reduced hospitalizations in the DAPA-HF trial, led to improved function and mobility among patients that had beneficial effects on their insulin sensitivity, Dr. Vaduganathan speculated in an interview.
The new finding of dapagliflozin’s benefit “is great news,” commented Yehuda Handelsman, MD, an endocrinologist and diabetes specialist who is medical director of the Metabolic Institute of America in Tarzana, Calif. “It’s an impressive and important result, and another reason to use dapagliflozin in patients with HFrEF, a group of patients whom you want to prevent from having worse outcomes” by developing diabetes.
The DAPA-HF (Dapagliflozin and Prevention of Adverse Outcomes in Heart Failure) trial enrolled HFrEF patients at 410 centers in 20 countries during February 2017–August 2018. The study’s primary endpoint was the composite incidence of cardiovascular death or worsening heart failure, which occurred in 16.3% of patients randomized to receive dapagliflozin and in 21.2% of control patients on standard care but on placebo instead of the study drug, a statistically significant relative risk reduction of 26% (N Engl J Med. 2019 Nov 21;381[21]:1995-2008). In the 2,605-patient subgroup without type 2 diabetes at baseline the primary endpoint fell by a statistically significant 27% with dapagliflozin treatment, the first time an SGLT2 inhibitor drug was shown effective for reducing this endpoint in patients with HFrEF but without diabetes. DAPA-HF did not enroll any patients with type 1 diabetes.
DAPA-HF was sponsored by AstraZeneca, the company that markets dapagliflozin (Farxiga). Dr. Inzucchi has been a consultant to AstraZeneca and to Abbott, Boehringer Ingelheim, Merck, Novo Nordisk, Sanofi/Lexicon, and vTv Therapeutics. Dr. Vaduganathan has been an adviser to AstraZeneca and to Amgen, Baxter, Bayer, Boehringer Ingelheim, Cytokinetics, and Relypsa. Dr. Handelsman has been a consultant to several drug companies including AstraZeneca.
SOURCE: Inzucchi SE et al. ADA 2020, abstract 271-OR.
FROM ADA 2020
Daily Recap: FDA revokes emergency use of hydroxychloroquine; Hardest hit specialties ranked in financial report
Here are the stories our MDedge editors across specialties think you need to know about today:
It’s official: COVID-19 is bad for your health care business
For the months of March and April 2020, use of medical professional services dropped by 65% and 68%, respectively, compared with last year, and estimated revenue fell by 45% and 48%, FAIR Health, a nonprofit organization that manages a database of 31 billion claim records, said in a new report.
Of the seven specialties included in the study, oral surgery was hit the hardest, followed by gastroenterology, cardiology, orthopedics, dermatology, adult primary care, and pediatric primary care, FAIR Health said.
“Even when medical practices have continued to function via telehealth, many have experienced lower reimbursements for telehealth visits than for in-person visits and more time educating patients on how to use the technology,” according to the report. Read more.
FDA revokes emergency use of hydroxychloroquine
The FDA revoked its decision from March 28 allowing use of hydroxychloroquine and chloroquine to treat people hospitalized with COVID-19 under an emergency use authorization (EUA).
"Based on its ongoing analysis of the EUA and emerging scientific data, the FDA determined that chloroquine and hydroxychloroquine are unlikely to be effective in treating COVID-19," the agency announced in a June 15 statement.
"In light of ongoing serious cardiac adverse events and other potential serious side effects, the known and potential benefits of chloroquine and hydroxychloroquine no longer outweigh the known and potential risks for the authorized use," noted the FDA. Read more.
Secondary infections common in COVID-19, implications unclear
Secondary respiratory infections appear to be highly prevalent among patients with severe COVID-19, but at this point, most physicians aren’t sure what to make of this understudied phenomenon.
“We really do not understand the implications of secondary infections on outcomes in COVID-19 patients,” David L. Bowton, MD, FCCP, said in an interview. “In most early reports the incidence of secondary infections was much higher in patients dying from COVID-19, compared to survivors, but it isn’t clear whether this indicates that the secondary infection itself led to excess mortality or was more a marker of the severity of the COVID-19 infection."
An early retrospective cohort study including 191 COVID-19 patients in Wuhan, China found that of the 54 who died in hospital, half had secondary bacterial lung infections (Lancet. 2020 Mar 28;395[10229]:1054-62). That comes as no surprise to U.S. physicians, who learned in training that many deaths during the so-called Spanish influenza epidemic were actually caused by secondary pneumonia involving Staphylococcus aureus, commented Daniel L. Ouellette, MD, FCCP. Read more.
Automated insulin delivery system ‘getting better and better’
Medtronic’s next-generation automated insulin delivery system offers significant improvements over the currently available model, particularly in young people with type 1 diabetes, new data suggest.
Data from three trials of such systems using Medtronic’s advanced hybrid closed-loop (AHCL) algorithm (trade name SmartGuard) were presented during the virtual American Diabetes Association (ADA) 80th Scientific Sessions.
Taken together, the data from the three trials showed that the AHCL-based system improved glycemic time-in-range with no increased risk for hypoglycemia, including in children and teenagers, with high patient-reported satisfaction.
“None of these devices is perfect, but they are a substantial improvement over what we’ve had ... They might make the quality of [patient] lives better. That’s really underappreciated,” session moderator Timothy S. Bailey, MD, commented. Read more.
Access more top news from the ADA virtual meeting.
For more on COVID-19, visit our Resource Center. All of our latest news is available on MDedge.com.
Here are the stories our MDedge editors across specialties think you need to know about today:
It’s official: COVID-19 is bad for your health care business
For the months of March and April 2020, use of medical professional services dropped by 65% and 68%, respectively, compared with last year, and estimated revenue fell by 45% and 48%, FAIR Health, a nonprofit organization that manages a database of 31 billion claim records, said in a new report.
Of the seven specialties included in the study, oral surgery was hit the hardest, followed by gastroenterology, cardiology, orthopedics, dermatology, adult primary care, and pediatric primary care, FAIR Health said.
“Even when medical practices have continued to function via telehealth, many have experienced lower reimbursements for telehealth visits than for in-person visits and more time educating patients on how to use the technology,” according to the report. Read more.
FDA revokes emergency use of hydroxychloroquine
The FDA revoked its decision from March 28 allowing use of hydroxychloroquine and chloroquine to treat people hospitalized with COVID-19 under an emergency use authorization (EUA).
"Based on its ongoing analysis of the EUA and emerging scientific data, the FDA determined that chloroquine and hydroxychloroquine are unlikely to be effective in treating COVID-19," the agency announced in a June 15 statement.
"In light of ongoing serious cardiac adverse events and other potential serious side effects, the known and potential benefits of chloroquine and hydroxychloroquine no longer outweigh the known and potential risks for the authorized use," noted the FDA. Read more.
Secondary infections common in COVID-19, implications unclear
Secondary respiratory infections appear to be highly prevalent among patients with severe COVID-19, but at this point, most physicians aren’t sure what to make of this understudied phenomenon.
“We really do not understand the implications of secondary infections on outcomes in COVID-19 patients,” David L. Bowton, MD, FCCP, said in an interview. “In most early reports the incidence of secondary infections was much higher in patients dying from COVID-19, compared to survivors, but it isn’t clear whether this indicates that the secondary infection itself led to excess mortality or was more a marker of the severity of the COVID-19 infection."
An early retrospective cohort study including 191 COVID-19 patients in Wuhan, China found that of the 54 who died in hospital, half had secondary bacterial lung infections (Lancet. 2020 Mar 28;395[10229]:1054-62). That comes as no surprise to U.S. physicians, who learned in training that many deaths during the so-called Spanish influenza epidemic were actually caused by secondary pneumonia involving Staphylococcus aureus, commented Daniel L. Ouellette, MD, FCCP. Read more.
Automated insulin delivery system ‘getting better and better’
Medtronic’s next-generation automated insulin delivery system offers significant improvements over the currently available model, particularly in young people with type 1 diabetes, new data suggest.
Data from three trials of such systems using Medtronic’s advanced hybrid closed-loop (AHCL) algorithm (trade name SmartGuard) were presented during the virtual American Diabetes Association (ADA) 80th Scientific Sessions.
Taken together, the data from the three trials showed that the AHCL-based system improved glycemic time-in-range with no increased risk for hypoglycemia, including in children and teenagers, with high patient-reported satisfaction.
“None of these devices is perfect, but they are a substantial improvement over what we’ve had ... They might make the quality of [patient] lives better. That’s really underappreciated,” session moderator Timothy S. Bailey, MD, commented. Read more.
Access more top news from the ADA virtual meeting.
For more on COVID-19, visit our Resource Center. All of our latest news is available on MDedge.com.
Here are the stories our MDedge editors across specialties think you need to know about today:
It’s official: COVID-19 is bad for your health care business
For the months of March and April 2020, use of medical professional services dropped by 65% and 68%, respectively, compared with last year, and estimated revenue fell by 45% and 48%, FAIR Health, a nonprofit organization that manages a database of 31 billion claim records, said in a new report.
Of the seven specialties included in the study, oral surgery was hit the hardest, followed by gastroenterology, cardiology, orthopedics, dermatology, adult primary care, and pediatric primary care, FAIR Health said.
“Even when medical practices have continued to function via telehealth, many have experienced lower reimbursements for telehealth visits than for in-person visits and more time educating patients on how to use the technology,” according to the report. Read more.
FDA revokes emergency use of hydroxychloroquine
The FDA revoked its decision from March 28 allowing use of hydroxychloroquine and chloroquine to treat people hospitalized with COVID-19 under an emergency use authorization (EUA).
"Based on its ongoing analysis of the EUA and emerging scientific data, the FDA determined that chloroquine and hydroxychloroquine are unlikely to be effective in treating COVID-19," the agency announced in a June 15 statement.
"In light of ongoing serious cardiac adverse events and other potential serious side effects, the known and potential benefits of chloroquine and hydroxychloroquine no longer outweigh the known and potential risks for the authorized use," noted the FDA. Read more.
Secondary infections common in COVID-19, implications unclear
Secondary respiratory infections appear to be highly prevalent among patients with severe COVID-19, but at this point, most physicians aren’t sure what to make of this understudied phenomenon.
“We really do not understand the implications of secondary infections on outcomes in COVID-19 patients,” David L. Bowton, MD, FCCP, said in an interview. “In most early reports the incidence of secondary infections was much higher in patients dying from COVID-19, compared to survivors, but it isn’t clear whether this indicates that the secondary infection itself led to excess mortality or was more a marker of the severity of the COVID-19 infection."
An early retrospective cohort study including 191 COVID-19 patients in Wuhan, China found that of the 54 who died in hospital, half had secondary bacterial lung infections (Lancet. 2020 Mar 28;395[10229]:1054-62). That comes as no surprise to U.S. physicians, who learned in training that many deaths during the so-called Spanish influenza epidemic were actually caused by secondary pneumonia involving Staphylococcus aureus, commented Daniel L. Ouellette, MD, FCCP. Read more.
Automated insulin delivery system ‘getting better and better’
Medtronic’s next-generation automated insulin delivery system offers significant improvements over the currently available model, particularly in young people with type 1 diabetes, new data suggest.
Data from three trials of such systems using Medtronic’s advanced hybrid closed-loop (AHCL) algorithm (trade name SmartGuard) were presented during the virtual American Diabetes Association (ADA) 80th Scientific Sessions.
Taken together, the data from the three trials showed that the AHCL-based system improved glycemic time-in-range with no increased risk for hypoglycemia, including in children and teenagers, with high patient-reported satisfaction.
“None of these devices is perfect, but they are a substantial improvement over what we’ve had ... They might make the quality of [patient] lives better. That’s really underappreciated,” session moderator Timothy S. Bailey, MD, commented. Read more.
Access more top news from the ADA virtual meeting.
For more on COVID-19, visit our Resource Center. All of our latest news is available on MDedge.com.
Cognitive-behavioral therapy a standout for better immune function
Psychosocial interventions, particularly cognitive-behavioral therapy (CBT), are associated with enhanced immune system function, new research suggests.
Results of a systematic review and meta-analysis that included 56 randomized controlled trials and more than 4,000 participants showed that over time, psychosocial interventions appeared to augment beneficial immune system function while concurrently decreasing harmful immune system function in comparison with control conditions.
“These associations were most reliable for cognitive-behavioral therapy and multiple or combined interventions and for studies that assessed proinflammatory cytokines or markers, which are key indicators of inflammation in the body,” study investigator George M. Slavich, PhD, said in an interview.
“The analysis helps address the question of which types of psychosocial interventions are most consistently associated with changes in immune system function, under what conditions, and for whom. This knowledge could, in turn, be used to inform research efforts and public policy aimed at using psychosocial interventions to improve immune-related health outcomes,” added Dr. Slavich, director of the Laboratory for Stress Assessment and Research, University of California, Los Angeles.
The study was published online June 3 in JAMA Psychiatry.
Link to serious physical, mental illnesses
There is substantial evidence that the immune system plays a role in a variety of mental and physical health problems. Such problems include anxiety disorders, depression, suicide, schizophrenia, cardiovascular disease, autoimmune disorders, and neurodegenerative diseases. It has been recently suggested that more than half of all deaths worldwide are attributable to inflammation-related conditions.
Although pharmacologic interventions can play a role in addressing inflammation, they are not without drawbacks, most notably, cost and adverse side effects.
The World Health Organization, the National Academy of Medicine, the National Institutes of Health, and other groups have emphasized the importance of addressing global disease burden through psychosocial interventions when possible.
Such recommendations are supported by scientific evidence. Previous research has shown that immune system processes are influenced by a variety of social, neurocognitive, and behavioral factors.
Given such findings, researchers have examined the effects of interventions that reduce stress or bolster psychological resources on immune system function.
However, such research has yielded conflicting findings. Some studies show that psychosocial interventions clearly enhance immunity, whereas others do not.
In addition, questions remain regarding which types of interventions reliably improve immune system function, under what conditions, and for whom.
“Research has shown that psychological factors – such as life stress, negative emotions, and social support – are associated with changes in immune system function,” Dr. Slavich noted.
“In addition, there is growing appreciation that immune system processes involved in inflammation may contribute to peoples’ risk for several major mental and physical health problems, including anxiety disorders, depression, heart disease, and autoimmune and neurodegenerative disorders.”
First study of its kind
To shed light on these potential links, the researchers conducted what they believe is the first systematic review and meta-analysis of randomized clinical trials of the effects of psychosocial interventions on immune system outcomes.
As part of the review, Dr. Slavich and colleagues estimated the associations between eight psychosocial interventions and seven markers of immune system function.
The eight psychosocial interventions were behavior therapy, cognitive therapy, CBT, CBT plus additive treatment or mode of delivery, bereavement or supportive therapy, multiple or combined interventions, other psychotherapy, and psychoeducation.
The seven immune outcomes that might be influenced by these interventions are proinflammatory cytokines and markers, anti-inflammatory cytokines, antibodies, immune cell counts, natural killer cell activity, viral load, and other immune outcomes.
The researchers also examined nine potential factors that might moderate the associations between psychosocial interventions and immune system function.
They searched a variety of databases for all relevant randomized controlled trials published through Dec. 31, 2018. Studies were eligible for inclusion if they included a psychosocial intervention and immune outcome, as well as preintervention and postintervention immunologic assessments.
The researchers identified 4,621 studies. Of these studies, 62 were eligible for inclusion; 56, which included 4,060 patients, were included in the final meta-analysis.
Results showed that psychosocial interventions were associated with enhanced immune system function (P < .001). There was relatively low heterogeneity between studies in these effect sizes, which, the investigators said, indicates that the association was relatively consistent across studies and conditions.
The meta-analysis showed that individuals who were assigned to a psychosocial intervention condition demonstrated a 14.7% improvement (95% confidence interval [CI], 5.7%–23.8%) in beneficial immune system function compared with their counterparts who were assigned to a control condition.
Similarly, participants who received psychosocial interventions demonstrated an 18.0% decrease (95% CI, 7.2%–28.8%) in harmful immune system function over time.
A standout
Regarding the effect of the type of intervention on the association, only CBT (31 studies; P < .001) and multiple or combined interventions (seven studies; P = .01) were significantly associated with changes in immune system outcomes.
The analysis also found that interventions that included a group component were more consistently associated with enhanced immune function than were those that did not include a group component. Nevertheless, this difference did not reach statistical significance (P = .06).
Contrary to the researchers’ expectations, the analysis also revealed that intervention length did not moderate the association between psychosocial interventions and immune system function (P = .93).
With respect to the type of immune marker studied, the meta-analysis found that psychosocial interventions had significantly different associations with the various immune markers studied. Of the seven immune outcomes investigated, only proinflammatory cytokine or marker levels (33 studies; P < .001) and immune cell counts (27 studies; P < .001) were significantly associated with the psychosocial interventions examined.
and were robust across age, sex, and intervention duration.
These results suggest that psychosocial interventions – particularly CBT and multiple or combined psychotherapeutic modalities – may play an important role in improving immune-related health outcomes.
Such interventions may not only be effective, they may also prove to be affordable alternatives to current therapeutic options. The mean length of a CBT intervention in the meta-analysis was 10.4 weeks, which the investigators equated with a total cost of $1,560 per patient.
“By comparison, the cost of using infliximab to reduce inflammation in persons with an autoimmune disorder is approximately $25,000 per patient per year,” they wrote.
“The results suggest the possibility that psychotherapy may be helpful for reducing inflammation and improving immune-related health in certain circumstances,” Dr. Slavich concluded. “However, the studies that we examined differed in terms of their quality, and we did not examine health outcomes in the present investigation.
“Therefore, more research needs to be done to determine how the present findings might be translated into treatment options or public policy.”
A path to better health
In an accompanying editorial, Veronika Engert, PhD, Joshua A. Grant, PhD, and Bernhard Strauss, PhD, noted that although infectious disease was once the primary cause of death in society, it has been supplanted by other complex and chronic illnesses, which often do not follow simple cause-and-effect associations.
“Rather,” they wrote, “these illnesses develop from a complex milieu of biological, psychological, and social factors that may also influence the disease progress and its prognosis. Against this backdrop, the meta-analysis by Shields and colleagues is an important confirmation of the biopsychosocial model.”
The editorialists explained that recent psychophysiological, neurobiological, and epigenetic research offers a glimpse into the relationship between psychological and social factors in pathogenesis. Nevertheless, the authors noted that a comprehensive examination of the potential effects of psychosocial interventions on immune parameters in various physical health conditions has been lacking.
“The evidence provided by Shields et al. is exactly what is needed to more fully shift treatment from an illness-centered to a patient-centered approach,” they wrote. “To that end, this meta-analysis may serve as a guide for policy makers aiming to improve immune-associated health.”
The research was supported by a Society in Science–Branco Weiss Fellowship, Brain and Behavior Research, and the National Institutes of Health. Dr. Slavich, Dr. Engert, Dr. Grant, and Dr. Strauss have disclosed no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
Psychosocial interventions, particularly cognitive-behavioral therapy (CBT), are associated with enhanced immune system function, new research suggests.
Results of a systematic review and meta-analysis that included 56 randomized controlled trials and more than 4,000 participants showed that over time, psychosocial interventions appeared to augment beneficial immune system function while concurrently decreasing harmful immune system function in comparison with control conditions.
“These associations were most reliable for cognitive-behavioral therapy and multiple or combined interventions and for studies that assessed proinflammatory cytokines or markers, which are key indicators of inflammation in the body,” study investigator George M. Slavich, PhD, said in an interview.
“The analysis helps address the question of which types of psychosocial interventions are most consistently associated with changes in immune system function, under what conditions, and for whom. This knowledge could, in turn, be used to inform research efforts and public policy aimed at using psychosocial interventions to improve immune-related health outcomes,” added Dr. Slavich, director of the Laboratory for Stress Assessment and Research, University of California, Los Angeles.
The study was published online June 3 in JAMA Psychiatry.
Link to serious physical, mental illnesses
There is substantial evidence that the immune system plays a role in a variety of mental and physical health problems. Such problems include anxiety disorders, depression, suicide, schizophrenia, cardiovascular disease, autoimmune disorders, and neurodegenerative diseases. It has been recently suggested that more than half of all deaths worldwide are attributable to inflammation-related conditions.
Although pharmacologic interventions can play a role in addressing inflammation, they are not without drawbacks, most notably, cost and adverse side effects.
The World Health Organization, the National Academy of Medicine, the National Institutes of Health, and other groups have emphasized the importance of addressing global disease burden through psychosocial interventions when possible.
Such recommendations are supported by scientific evidence. Previous research has shown that immune system processes are influenced by a variety of social, neurocognitive, and behavioral factors.
Given such findings, researchers have examined the effects of interventions that reduce stress or bolster psychological resources on immune system function.
However, such research has yielded conflicting findings. Some studies show that psychosocial interventions clearly enhance immunity, whereas others do not.
In addition, questions remain regarding which types of interventions reliably improve immune system function, under what conditions, and for whom.
“Research has shown that psychological factors – such as life stress, negative emotions, and social support – are associated with changes in immune system function,” Dr. Slavich noted.
“In addition, there is growing appreciation that immune system processes involved in inflammation may contribute to peoples’ risk for several major mental and physical health problems, including anxiety disorders, depression, heart disease, and autoimmune and neurodegenerative disorders.”
First study of its kind
To shed light on these potential links, the researchers conducted what they believe is the first systematic review and meta-analysis of randomized clinical trials of the effects of psychosocial interventions on immune system outcomes.
As part of the review, Dr. Slavich and colleagues estimated the associations between eight psychosocial interventions and seven markers of immune system function.
The eight psychosocial interventions were behavior therapy, cognitive therapy, CBT, CBT plus additive treatment or mode of delivery, bereavement or supportive therapy, multiple or combined interventions, other psychotherapy, and psychoeducation.
The seven immune outcomes that might be influenced by these interventions are proinflammatory cytokines and markers, anti-inflammatory cytokines, antibodies, immune cell counts, natural killer cell activity, viral load, and other immune outcomes.
The researchers also examined nine potential factors that might moderate the associations between psychosocial interventions and immune system function.
They searched a variety of databases for all relevant randomized controlled trials published through Dec. 31, 2018. Studies were eligible for inclusion if they included a psychosocial intervention and immune outcome, as well as preintervention and postintervention immunologic assessments.
The researchers identified 4,621 studies. Of these studies, 62 were eligible for inclusion; 56, which included 4,060 patients, were included in the final meta-analysis.
Results showed that psychosocial interventions were associated with enhanced immune system function (P < .001). There was relatively low heterogeneity between studies in these effect sizes, which, the investigators said, indicates that the association was relatively consistent across studies and conditions.
The meta-analysis showed that individuals who were assigned to a psychosocial intervention condition demonstrated a 14.7% improvement (95% confidence interval [CI], 5.7%–23.8%) in beneficial immune system function compared with their counterparts who were assigned to a control condition.
Similarly, participants who received psychosocial interventions demonstrated an 18.0% decrease (95% CI, 7.2%–28.8%) in harmful immune system function over time.
A standout
Regarding the effect of the type of intervention on the association, only CBT (31 studies; P < .001) and multiple or combined interventions (seven studies; P = .01) were significantly associated with changes in immune system outcomes.
The analysis also found that interventions that included a group component were more consistently associated with enhanced immune function than were those that did not include a group component. Nevertheless, this difference did not reach statistical significance (P = .06).
Contrary to the researchers’ expectations, the analysis also revealed that intervention length did not moderate the association between psychosocial interventions and immune system function (P = .93).
With respect to the type of immune marker studied, the meta-analysis found that psychosocial interventions had significantly different associations with the various immune markers studied. Of the seven immune outcomes investigated, only proinflammatory cytokine or marker levels (33 studies; P < .001) and immune cell counts (27 studies; P < .001) were significantly associated with the psychosocial interventions examined.
and were robust across age, sex, and intervention duration.
These results suggest that psychosocial interventions – particularly CBT and multiple or combined psychotherapeutic modalities – may play an important role in improving immune-related health outcomes.
Such interventions may not only be effective, they may also prove to be affordable alternatives to current therapeutic options. The mean length of a CBT intervention in the meta-analysis was 10.4 weeks, which the investigators equated with a total cost of $1,560 per patient.
“By comparison, the cost of using infliximab to reduce inflammation in persons with an autoimmune disorder is approximately $25,000 per patient per year,” they wrote.
“The results suggest the possibility that psychotherapy may be helpful for reducing inflammation and improving immune-related health in certain circumstances,” Dr. Slavich concluded. “However, the studies that we examined differed in terms of their quality, and we did not examine health outcomes in the present investigation.
“Therefore, more research needs to be done to determine how the present findings might be translated into treatment options or public policy.”
A path to better health
In an accompanying editorial, Veronika Engert, PhD, Joshua A. Grant, PhD, and Bernhard Strauss, PhD, noted that although infectious disease was once the primary cause of death in society, it has been supplanted by other complex and chronic illnesses, which often do not follow simple cause-and-effect associations.
“Rather,” they wrote, “these illnesses develop from a complex milieu of biological, psychological, and social factors that may also influence the disease progress and its prognosis. Against this backdrop, the meta-analysis by Shields and colleagues is an important confirmation of the biopsychosocial model.”
The editorialists explained that recent psychophysiological, neurobiological, and epigenetic research offers a glimpse into the relationship between psychological and social factors in pathogenesis. Nevertheless, the authors noted that a comprehensive examination of the potential effects of psychosocial interventions on immune parameters in various physical health conditions has been lacking.
“The evidence provided by Shields et al. is exactly what is needed to more fully shift treatment from an illness-centered to a patient-centered approach,” they wrote. “To that end, this meta-analysis may serve as a guide for policy makers aiming to improve immune-associated health.”
The research was supported by a Society in Science–Branco Weiss Fellowship, Brain and Behavior Research, and the National Institutes of Health. Dr. Slavich, Dr. Engert, Dr. Grant, and Dr. Strauss have disclosed no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
Psychosocial interventions, particularly cognitive-behavioral therapy (CBT), are associated with enhanced immune system function, new research suggests.
Results of a systematic review and meta-analysis that included 56 randomized controlled trials and more than 4,000 participants showed that over time, psychosocial interventions appeared to augment beneficial immune system function while concurrently decreasing harmful immune system function in comparison with control conditions.
“These associations were most reliable for cognitive-behavioral therapy and multiple or combined interventions and for studies that assessed proinflammatory cytokines or markers, which are key indicators of inflammation in the body,” study investigator George M. Slavich, PhD, said in an interview.
“The analysis helps address the question of which types of psychosocial interventions are most consistently associated with changes in immune system function, under what conditions, and for whom. This knowledge could, in turn, be used to inform research efforts and public policy aimed at using psychosocial interventions to improve immune-related health outcomes,” added Dr. Slavich, director of the Laboratory for Stress Assessment and Research, University of California, Los Angeles.
The study was published online June 3 in JAMA Psychiatry.
Link to serious physical, mental illnesses
There is substantial evidence that the immune system plays a role in a variety of mental and physical health problems. Such problems include anxiety disorders, depression, suicide, schizophrenia, cardiovascular disease, autoimmune disorders, and neurodegenerative diseases. It has been recently suggested that more than half of all deaths worldwide are attributable to inflammation-related conditions.
Although pharmacologic interventions can play a role in addressing inflammation, they are not without drawbacks, most notably, cost and adverse side effects.
The World Health Organization, the National Academy of Medicine, the National Institutes of Health, and other groups have emphasized the importance of addressing global disease burden through psychosocial interventions when possible.
Such recommendations are supported by scientific evidence. Previous research has shown that immune system processes are influenced by a variety of social, neurocognitive, and behavioral factors.
Given such findings, researchers have examined the effects of interventions that reduce stress or bolster psychological resources on immune system function.
However, such research has yielded conflicting findings. Some studies show that psychosocial interventions clearly enhance immunity, whereas others do not.
In addition, questions remain regarding which types of interventions reliably improve immune system function, under what conditions, and for whom.
“Research has shown that psychological factors – such as life stress, negative emotions, and social support – are associated with changes in immune system function,” Dr. Slavich noted.
“In addition, there is growing appreciation that immune system processes involved in inflammation may contribute to peoples’ risk for several major mental and physical health problems, including anxiety disorders, depression, heart disease, and autoimmune and neurodegenerative disorders.”
First study of its kind
To shed light on these potential links, the researchers conducted what they believe is the first systematic review and meta-analysis of randomized clinical trials of the effects of psychosocial interventions on immune system outcomes.
As part of the review, Dr. Slavich and colleagues estimated the associations between eight psychosocial interventions and seven markers of immune system function.
The eight psychosocial interventions were behavior therapy, cognitive therapy, CBT, CBT plus additive treatment or mode of delivery, bereavement or supportive therapy, multiple or combined interventions, other psychotherapy, and psychoeducation.
The seven immune outcomes that might be influenced by these interventions are proinflammatory cytokines and markers, anti-inflammatory cytokines, antibodies, immune cell counts, natural killer cell activity, viral load, and other immune outcomes.
The researchers also examined nine potential factors that might moderate the associations between psychosocial interventions and immune system function.
They searched a variety of databases for all relevant randomized controlled trials published through Dec. 31, 2018. Studies were eligible for inclusion if they included a psychosocial intervention and immune outcome, as well as preintervention and postintervention immunologic assessments.
The researchers identified 4,621 studies. Of these studies, 62 were eligible for inclusion; 56, which included 4,060 patients, were included in the final meta-analysis.
Results showed that psychosocial interventions were associated with enhanced immune system function (P < .001). There was relatively low heterogeneity between studies in these effect sizes, which, the investigators said, indicates that the association was relatively consistent across studies and conditions.
The meta-analysis showed that individuals who were assigned to a psychosocial intervention condition demonstrated a 14.7% improvement (95% confidence interval [CI], 5.7%–23.8%) in beneficial immune system function compared with their counterparts who were assigned to a control condition.
Similarly, participants who received psychosocial interventions demonstrated an 18.0% decrease (95% CI, 7.2%–28.8%) in harmful immune system function over time.
A standout
Regarding the effect of the type of intervention on the association, only CBT (31 studies; P < .001) and multiple or combined interventions (seven studies; P = .01) were significantly associated with changes in immune system outcomes.
The analysis also found that interventions that included a group component were more consistently associated with enhanced immune function than were those that did not include a group component. Nevertheless, this difference did not reach statistical significance (P = .06).
Contrary to the researchers’ expectations, the analysis also revealed that intervention length did not moderate the association between psychosocial interventions and immune system function (P = .93).
With respect to the type of immune marker studied, the meta-analysis found that psychosocial interventions had significantly different associations with the various immune markers studied. Of the seven immune outcomes investigated, only proinflammatory cytokine or marker levels (33 studies; P < .001) and immune cell counts (27 studies; P < .001) were significantly associated with the psychosocial interventions examined.
and were robust across age, sex, and intervention duration.
These results suggest that psychosocial interventions – particularly CBT and multiple or combined psychotherapeutic modalities – may play an important role in improving immune-related health outcomes.
Such interventions may not only be effective, they may also prove to be affordable alternatives to current therapeutic options. The mean length of a CBT intervention in the meta-analysis was 10.4 weeks, which the investigators equated with a total cost of $1,560 per patient.
“By comparison, the cost of using infliximab to reduce inflammation in persons with an autoimmune disorder is approximately $25,000 per patient per year,” they wrote.
“The results suggest the possibility that psychotherapy may be helpful for reducing inflammation and improving immune-related health in certain circumstances,” Dr. Slavich concluded. “However, the studies that we examined differed in terms of their quality, and we did not examine health outcomes in the present investigation.
“Therefore, more research needs to be done to determine how the present findings might be translated into treatment options or public policy.”
A path to better health
In an accompanying editorial, Veronika Engert, PhD, Joshua A. Grant, PhD, and Bernhard Strauss, PhD, noted that although infectious disease was once the primary cause of death in society, it has been supplanted by other complex and chronic illnesses, which often do not follow simple cause-and-effect associations.
“Rather,” they wrote, “these illnesses develop from a complex milieu of biological, psychological, and social factors that may also influence the disease progress and its prognosis. Against this backdrop, the meta-analysis by Shields and colleagues is an important confirmation of the biopsychosocial model.”
The editorialists explained that recent psychophysiological, neurobiological, and epigenetic research offers a glimpse into the relationship between psychological and social factors in pathogenesis. Nevertheless, the authors noted that a comprehensive examination of the potential effects of psychosocial interventions on immune parameters in various physical health conditions has been lacking.
“The evidence provided by Shields et al. is exactly what is needed to more fully shift treatment from an illness-centered to a patient-centered approach,” they wrote. “To that end, this meta-analysis may serve as a guide for policy makers aiming to improve immune-associated health.”
The research was supported by a Society in Science–Branco Weiss Fellowship, Brain and Behavior Research, and the National Institutes of Health. Dr. Slavich, Dr. Engert, Dr. Grant, and Dr. Strauss have disclosed no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
For COVID-19 plus diabetes, glycemic control tops treatment list
Optimizing glycemic control “is the key to overall treatment in people with diabetes and COVID-19,” said Antonio Ceriello, MD, during a June 5 webinar sponsored by Harvard Medical School, Boston.
Dr. Ceriello, a research consultant with the Italian Ministry of Health, IRCCS Multi-Medica, Milan, highlighted a recent study that examined the association of blood glucose control and outcomes in COVID-19 patients with preexisting type 2 diabetes.
Among 7,000 cases of COVID-19, type 2 diabetes correlated with a higher death rate. However, those with well-controlled blood glucose (upper limit ≤10 mmol/L) had a survival rate of 98.9%, compared with just 11% among those with poorly controlled blood glucose (upper limit >10 mmol/L), a reduction in risk of 86% (adjusted hazard ratio, 0.14; Cell Metab. 2020 May 1. doi: 10.1016/j.cmet.2020.04.021).
Clinicians should also consider the possible side effects of hypoglycemic agents in the evolution of this disease. This is true of all patients, not just diabetes patients, Dr. Ceriello said. “We have data showing that ... hyperglycemia contributes directly to worsening the prognosis of COVID-19 independent of the presence of diabetes.”
One study found that the glycosylation of ACE-2 played an important role in allowing cellular entry of the virus (Am J Physiol Endocrinol Metab. 2020 Mar 31;318:E736-41). “This is something that could be related to hyperglycemia,” he added.
Another risk factor is thrombosis, a clear contributor to death rates in COVID-19. Research on thrombosis incidence in COVID-19 patients with diabetes reported higher levels of D-dimer levels in people with diabetes, especially among those who couldn’t manage their disease.
Tying all of these factors together, Dr. Ceriello discussed how ACE-2 glycosylation, in combination with other factors in SARS-CoV-2 infection, could lead to hyperglycemia, thrombosis, and subsequently multiorgan damage in diabetes patients.
Other research has associated higher HbA1c levels (mean HbA1c, 7.5%) with higher mortality risk in COVID-19 patients, said another speaker, Linong Ji, MD, director for endocrinology and metabolism at Peking University People’s Hospital, Beijing, and director of Peking University’s Diabetes Center. Proper guidance is key to ensuring early detection of hyperglycemic crisis in people with diabetes, advised Dr. Ji.
Global management of diabetes in SARS-CoV-2 patients is “quite challenging,” given that most patients don’t have their diabetes under control, said host and moderator A. Enrique Caballero, MD, an endocrinologist/investigator in the division of endocrinology, diabetes, and hypertension and division of global health equity at Brigham and Women’s Hospital, Boston. “They are not meeting treatment targets for cholesterol or glucose control. So we’re not managing optimal care. And now on top of this, we have COVID-19.”
Optimizing glycemic control “is the key to overall treatment in people with diabetes and COVID-19,” said Antonio Ceriello, MD, during a June 5 webinar sponsored by Harvard Medical School, Boston.
Dr. Ceriello, a research consultant with the Italian Ministry of Health, IRCCS Multi-Medica, Milan, highlighted a recent study that examined the association of blood glucose control and outcomes in COVID-19 patients with preexisting type 2 diabetes.
Among 7,000 cases of COVID-19, type 2 diabetes correlated with a higher death rate. However, those with well-controlled blood glucose (upper limit ≤10 mmol/L) had a survival rate of 98.9%, compared with just 11% among those with poorly controlled blood glucose (upper limit >10 mmol/L), a reduction in risk of 86% (adjusted hazard ratio, 0.14; Cell Metab. 2020 May 1. doi: 10.1016/j.cmet.2020.04.021).
Clinicians should also consider the possible side effects of hypoglycemic agents in the evolution of this disease. This is true of all patients, not just diabetes patients, Dr. Ceriello said. “We have data showing that ... hyperglycemia contributes directly to worsening the prognosis of COVID-19 independent of the presence of diabetes.”
One study found that the glycosylation of ACE-2 played an important role in allowing cellular entry of the virus (Am J Physiol Endocrinol Metab. 2020 Mar 31;318:E736-41). “This is something that could be related to hyperglycemia,” he added.
Another risk factor is thrombosis, a clear contributor to death rates in COVID-19. Research on thrombosis incidence in COVID-19 patients with diabetes reported higher levels of D-dimer levels in people with diabetes, especially among those who couldn’t manage their disease.
Tying all of these factors together, Dr. Ceriello discussed how ACE-2 glycosylation, in combination with other factors in SARS-CoV-2 infection, could lead to hyperglycemia, thrombosis, and subsequently multiorgan damage in diabetes patients.
Other research has associated higher HbA1c levels (mean HbA1c, 7.5%) with higher mortality risk in COVID-19 patients, said another speaker, Linong Ji, MD, director for endocrinology and metabolism at Peking University People’s Hospital, Beijing, and director of Peking University’s Diabetes Center. Proper guidance is key to ensuring early detection of hyperglycemic crisis in people with diabetes, advised Dr. Ji.
Global management of diabetes in SARS-CoV-2 patients is “quite challenging,” given that most patients don’t have their diabetes under control, said host and moderator A. Enrique Caballero, MD, an endocrinologist/investigator in the division of endocrinology, diabetes, and hypertension and division of global health equity at Brigham and Women’s Hospital, Boston. “They are not meeting treatment targets for cholesterol or glucose control. So we’re not managing optimal care. And now on top of this, we have COVID-19.”
Optimizing glycemic control “is the key to overall treatment in people with diabetes and COVID-19,” said Antonio Ceriello, MD, during a June 5 webinar sponsored by Harvard Medical School, Boston.
Dr. Ceriello, a research consultant with the Italian Ministry of Health, IRCCS Multi-Medica, Milan, highlighted a recent study that examined the association of blood glucose control and outcomes in COVID-19 patients with preexisting type 2 diabetes.
Among 7,000 cases of COVID-19, type 2 diabetes correlated with a higher death rate. However, those with well-controlled blood glucose (upper limit ≤10 mmol/L) had a survival rate of 98.9%, compared with just 11% among those with poorly controlled blood glucose (upper limit >10 mmol/L), a reduction in risk of 86% (adjusted hazard ratio, 0.14; Cell Metab. 2020 May 1. doi: 10.1016/j.cmet.2020.04.021).
Clinicians should also consider the possible side effects of hypoglycemic agents in the evolution of this disease. This is true of all patients, not just diabetes patients, Dr. Ceriello said. “We have data showing that ... hyperglycemia contributes directly to worsening the prognosis of COVID-19 independent of the presence of diabetes.”
One study found that the glycosylation of ACE-2 played an important role in allowing cellular entry of the virus (Am J Physiol Endocrinol Metab. 2020 Mar 31;318:E736-41). “This is something that could be related to hyperglycemia,” he added.
Another risk factor is thrombosis, a clear contributor to death rates in COVID-19. Research on thrombosis incidence in COVID-19 patients with diabetes reported higher levels of D-dimer levels in people with diabetes, especially among those who couldn’t manage their disease.
Tying all of these factors together, Dr. Ceriello discussed how ACE-2 glycosylation, in combination with other factors in SARS-CoV-2 infection, could lead to hyperglycemia, thrombosis, and subsequently multiorgan damage in diabetes patients.
Other research has associated higher HbA1c levels (mean HbA1c, 7.5%) with higher mortality risk in COVID-19 patients, said another speaker, Linong Ji, MD, director for endocrinology and metabolism at Peking University People’s Hospital, Beijing, and director of Peking University’s Diabetes Center. Proper guidance is key to ensuring early detection of hyperglycemic crisis in people with diabetes, advised Dr. Ji.
Global management of diabetes in SARS-CoV-2 patients is “quite challenging,” given that most patients don’t have their diabetes under control, said host and moderator A. Enrique Caballero, MD, an endocrinologist/investigator in the division of endocrinology, diabetes, and hypertension and division of global health equity at Brigham and Women’s Hospital, Boston. “They are not meeting treatment targets for cholesterol or glucose control. So we’re not managing optimal care. And now on top of this, we have COVID-19.”










