User login
Geriatric IBD hospitalization carries steep inpatient mortality
SAN ANTONIO – Jeffrey Schwartz, MD, reported at the annual meeting of the American College of Gastroenterology.

The magnitude of the age-related increased risk highlighted in this large national study was strikingly larger than the differential inpatient mortality between geriatric and nongeriatric patients hospitalized for conditions other than inflammatory bowel disease (IBD). It’s a finding that reveals a major unmet need for improved systems of care for elderly hospitalized IBD patients, according to Dr. Schwartz, an internal medicine resident at Beth Israel Deaconess Medical Center, Boston.
“Given the high prevalence of IBD patients that require inpatient admission, as well as the rapidly aging nature of the U.S. population, it’s our hope that this study will provide some insight to drive efforts to improve standardized guideline-directed therapy and propose interventions to help close what I think is a very important gap in clinical care,” he said.
It’s well established that a second peak of IBD diagnoses occurs in 50- to 70-year-olds. At present, roughly 30% of all individuals carrying the diagnosis of IBD are over age 65, and with the graying of the baby-boomer population, this proportion is climbing.
Dr. Schwartz presented a study of the National Inpatient Sample for 2016, which is a representative sample comprising 20% of all U.S. hospital discharges for that year, the most recent year for which the data are available. The study population included all 71,040 patients hospitalized for acute management of Crohn’s disease or its immediate complications, of whom 10,095 were aged over age 75 years, as well as the 35,950 patients hospitalized for ulcerative colitis, 8,285 of whom were over 75.
Inpatient mortality occurred in 1.5% of the geriatric admissions, compared with 0.2% of nongeriatric admissions for Crohn’s disease. Similarly, the inpatient mortality rate in geriatric patients with ulcerative colitis was 1.0% versus 0.1% in patients under age 75 hospitalized for ulcerative colitis.
There are lots of reasons why the management of geriatric patients with IBD is particularly challenging, Dr. Schwartz noted. They have a higher burden of comorbid conditions, worse nutritional status, and increased risks of infection and cancer. In a regression analysis that attempted to control for such confounders using the Elixhauser mortality index, the nongeriatric Crohn’s disease patients were an adjusted 75% less likely to die in the hospital than those who were older. Nongeriatric ulcerative colitis patients were 81% less likely to die than geriatric patients with the disease. In contrast, nongeriatric patients admitted for reasons other than IBD had only an adjusted 50% lower risk of inpatient mortality than those who were older than 75.
Of note, in this analysis adjusted for confounders, there was no difference between geriatric and nongeriatric IBD patients in terms of resource utilization as reflected in average length of stay and hospital charges, Dr. Schwartz continued.
Asked if he could shed light on any specific complications that drove the age-related disparity in inpatient mortality in the IBD population, the physician replied that he and his coinvestigators were thwarted in their effort to do so because the inpatient mortality of 1.0%-1.5% was so low that further breakdown as to causes of death would have been statistically unreliable. It might be possible to do so successfully by combining several years of National Inpatient Sample data. That being said, it’s reasonable to hypothesize that cardiovascular complications are an important contributor, he added.
Dr. Schwartz reported having no financial conflicts regarding his study, conducted free of commercial support.
SAN ANTONIO – Jeffrey Schwartz, MD, reported at the annual meeting of the American College of Gastroenterology.

The magnitude of the age-related increased risk highlighted in this large national study was strikingly larger than the differential inpatient mortality between geriatric and nongeriatric patients hospitalized for conditions other than inflammatory bowel disease (IBD). It’s a finding that reveals a major unmet need for improved systems of care for elderly hospitalized IBD patients, according to Dr. Schwartz, an internal medicine resident at Beth Israel Deaconess Medical Center, Boston.
“Given the high prevalence of IBD patients that require inpatient admission, as well as the rapidly aging nature of the U.S. population, it’s our hope that this study will provide some insight to drive efforts to improve standardized guideline-directed therapy and propose interventions to help close what I think is a very important gap in clinical care,” he said.
It’s well established that a second peak of IBD diagnoses occurs in 50- to 70-year-olds. At present, roughly 30% of all individuals carrying the diagnosis of IBD are over age 65, and with the graying of the baby-boomer population, this proportion is climbing.
Dr. Schwartz presented a study of the National Inpatient Sample for 2016, which is a representative sample comprising 20% of all U.S. hospital discharges for that year, the most recent year for which the data are available. The study population included all 71,040 patients hospitalized for acute management of Crohn’s disease or its immediate complications, of whom 10,095 were aged over age 75 years, as well as the 35,950 patients hospitalized for ulcerative colitis, 8,285 of whom were over 75.
Inpatient mortality occurred in 1.5% of the geriatric admissions, compared with 0.2% of nongeriatric admissions for Crohn’s disease. Similarly, the inpatient mortality rate in geriatric patients with ulcerative colitis was 1.0% versus 0.1% in patients under age 75 hospitalized for ulcerative colitis.
There are lots of reasons why the management of geriatric patients with IBD is particularly challenging, Dr. Schwartz noted. They have a higher burden of comorbid conditions, worse nutritional status, and increased risks of infection and cancer. In a regression analysis that attempted to control for such confounders using the Elixhauser mortality index, the nongeriatric Crohn’s disease patients were an adjusted 75% less likely to die in the hospital than those who were older. Nongeriatric ulcerative colitis patients were 81% less likely to die than geriatric patients with the disease. In contrast, nongeriatric patients admitted for reasons other than IBD had only an adjusted 50% lower risk of inpatient mortality than those who were older than 75.
Of note, in this analysis adjusted for confounders, there was no difference between geriatric and nongeriatric IBD patients in terms of resource utilization as reflected in average length of stay and hospital charges, Dr. Schwartz continued.
Asked if he could shed light on any specific complications that drove the age-related disparity in inpatient mortality in the IBD population, the physician replied that he and his coinvestigators were thwarted in their effort to do so because the inpatient mortality of 1.0%-1.5% was so low that further breakdown as to causes of death would have been statistically unreliable. It might be possible to do so successfully by combining several years of National Inpatient Sample data. That being said, it’s reasonable to hypothesize that cardiovascular complications are an important contributor, he added.
Dr. Schwartz reported having no financial conflicts regarding his study, conducted free of commercial support.
SAN ANTONIO – Jeffrey Schwartz, MD, reported at the annual meeting of the American College of Gastroenterology.

The magnitude of the age-related increased risk highlighted in this large national study was strikingly larger than the differential inpatient mortality between geriatric and nongeriatric patients hospitalized for conditions other than inflammatory bowel disease (IBD). It’s a finding that reveals a major unmet need for improved systems of care for elderly hospitalized IBD patients, according to Dr. Schwartz, an internal medicine resident at Beth Israel Deaconess Medical Center, Boston.
“Given the high prevalence of IBD patients that require inpatient admission, as well as the rapidly aging nature of the U.S. population, it’s our hope that this study will provide some insight to drive efforts to improve standardized guideline-directed therapy and propose interventions to help close what I think is a very important gap in clinical care,” he said.
It’s well established that a second peak of IBD diagnoses occurs in 50- to 70-year-olds. At present, roughly 30% of all individuals carrying the diagnosis of IBD are over age 65, and with the graying of the baby-boomer population, this proportion is climbing.
Dr. Schwartz presented a study of the National Inpatient Sample for 2016, which is a representative sample comprising 20% of all U.S. hospital discharges for that year, the most recent year for which the data are available. The study population included all 71,040 patients hospitalized for acute management of Crohn’s disease or its immediate complications, of whom 10,095 were aged over age 75 years, as well as the 35,950 patients hospitalized for ulcerative colitis, 8,285 of whom were over 75.
Inpatient mortality occurred in 1.5% of the geriatric admissions, compared with 0.2% of nongeriatric admissions for Crohn’s disease. Similarly, the inpatient mortality rate in geriatric patients with ulcerative colitis was 1.0% versus 0.1% in patients under age 75 hospitalized for ulcerative colitis.
There are lots of reasons why the management of geriatric patients with IBD is particularly challenging, Dr. Schwartz noted. They have a higher burden of comorbid conditions, worse nutritional status, and increased risks of infection and cancer. In a regression analysis that attempted to control for such confounders using the Elixhauser mortality index, the nongeriatric Crohn’s disease patients were an adjusted 75% less likely to die in the hospital than those who were older. Nongeriatric ulcerative colitis patients were 81% less likely to die than geriatric patients with the disease. In contrast, nongeriatric patients admitted for reasons other than IBD had only an adjusted 50% lower risk of inpatient mortality than those who were older than 75.
Of note, in this analysis adjusted for confounders, there was no difference between geriatric and nongeriatric IBD patients in terms of resource utilization as reflected in average length of stay and hospital charges, Dr. Schwartz continued.
Asked if he could shed light on any specific complications that drove the age-related disparity in inpatient mortality in the IBD population, the physician replied that he and his coinvestigators were thwarted in their effort to do so because the inpatient mortality of 1.0%-1.5% was so low that further breakdown as to causes of death would have been statistically unreliable. It might be possible to do so successfully by combining several years of National Inpatient Sample data. That being said, it’s reasonable to hypothesize that cardiovascular complications are an important contributor, he added.
Dr. Schwartz reported having no financial conflicts regarding his study, conducted free of commercial support.
REPORTING FROM ACG 2019
Key clinical point: A major unmet need exists for better guideline-directed management of geriatric patients hospitalized for inflammatory bowel disease.
Major finding: The inpatient mortality rate among patients aged over age 75 years hospitalized for management of inflammatory bowel disease is four to five times higher than in those who are younger.
Study details: This was a retrospective analysis of all 106,990 hospital admissions for management of inflammatory bowel disease included in the 2016 National Inpatient Sample.
Disclosures: The presenter reported having no financial conflicts regarding his study, conducted free of commercial support.
Source: Schwartz J. ACG 2019, Abstract 42.
Survey asks adults: How likely are you to develop dementia?
Donovan T. Maust, MD, and colleagues reported in a research letter published in JAMA Neurology.
More than half of study participants used crossword puzzles as a memory exercise, but only 5% said they spoke to their physician about how to reduce risk. Ironically, this lack of communication was also associated with buying unproven over-the-counter memory supplements, while still remaining ignorant of proven ways to head off dementia and other contributing chronic conditions, wrote Dr. Maust of the University of Michigan, Ann Arbor, and coauthors.
Their analysis of the Michigan National Poll on Healthy Aging found that close to half of respondents (48.5%) reported that they were at least somewhat likely to develop dementia. Another 4.2% thought dementia was “very likely” in their future.
The study comprised survey responses from 1,019 adults aged 50-64 years. Most rated their physical health either excellent (445 respondents) or good (413 respondents). Most also reported excellent or very good mental health (721 respondents); 234 reported good mental health. Many (678) were affluent, with annual incomes of $60,000 or higher. They tended to be well educated; only 337 were without at least some college education. More than half were white (753); there were 101 Hispanic respondents and 93 black respondents. Other groups made up the remainder.
A multivariate analysis found that black respondents were about half as likely to believe they would develop dementia, compared with whites – an assumption contrary to epidemiologic findings that blacks are more likely than whites to develop dementia.
People who reported fair or poor mental health were more than twice as likely to feel dementia was in their future (odds ratio, 2.3). But fair or poor physical health was not significantly associated with that concern.
“Those with fair to poor physical health did not accurately perceive that their likelihood of developing dementia was potentially higher than respondents with very good or excellent physical health,” the authors wrote. “In contrast, fair to poor mental health had the largest association with perceived likelihood of dementia, even though less evidence suggests that poor mental health is causally linked with dementia.”
Despite the concerns, just 5% of respondents said that they had spoken to their physician. Those who believed they had a high likelihood of dementia were more likely to talk with their clinician (7.1%) than those who believed they had a low risk (3.6%).
Many more, however, were using non–evidence-based compounds touted as memory supporting. These included fish oil or omega-3 fatty acids (31.6%) and vitamins or supplements (32.9%). Crossword puzzles were a very popular prevention strategy, employed by about 55% in both belief groups.
“While managing chronic medical conditions, such as diabetes or cardiovascular disease, could reduce dementia risk, few respondents appear to have discussed this with their physician. Given repeated failures of disease-preventing or disease-modifying treatments for dementia, interest in treatment and prevention has shifted earlier in the disease process. Adults in middle age may not accurately estimate their risk of developing dementia, which could lead to both overuse and underuse if preclinical dementia treatments become available. Policy and physicians should emphasize current evidence-based strategies of managing lifestyle and chronic medical conditions to reduce the risk of dementia,” the investigators wrote.
Dr. Maust had no financial disclosures.
SOURCE: Maust D et al. JAMA Neurol. 2019 Nov 15. doi: 10.1001/jamaneurol.2019.3946
I do not find it surprising that older adults fear dementia. Since they correctly perceive that there is no disease-modifying therapy (and maybe also that “getting caught with memory loss” would lead to a loss of driving privileges and other restrictions), they may be trying not to focus on it. As for asking about strategies to “prevent” dementia, that question implies unwarranted optimism about the effectiveness of any such strategy, especially in an older adult. I think we can say that a lifetime of healthy habits (regular physical exercise and careful control of any chronic conditions like diabetes being particularly important) may reduce our risk of dementia a bit, but the idea that anything a 75-year-old does is going to prevent it at that point is probably wishful thinking. Supplements and the like seem to have their own followers. It amazes me how many people suspect what they are taking probably does no good but they do it anyway out of blind hope. Sometimes we can talk them out of spending their money on such things – but not always.
Richard Caselli, MD, is associate director and clinical core director of the Alzheimer’s Disease Center at the Mayo Clinic in Scottsdale, Ariz.
I do not find it surprising that older adults fear dementia. Since they correctly perceive that there is no disease-modifying therapy (and maybe also that “getting caught with memory loss” would lead to a loss of driving privileges and other restrictions), they may be trying not to focus on it. As for asking about strategies to “prevent” dementia, that question implies unwarranted optimism about the effectiveness of any such strategy, especially in an older adult. I think we can say that a lifetime of healthy habits (regular physical exercise and careful control of any chronic conditions like diabetes being particularly important) may reduce our risk of dementia a bit, but the idea that anything a 75-year-old does is going to prevent it at that point is probably wishful thinking. Supplements and the like seem to have their own followers. It amazes me how many people suspect what they are taking probably does no good but they do it anyway out of blind hope. Sometimes we can talk them out of spending their money on such things – but not always.
Richard Caselli, MD, is associate director and clinical core director of the Alzheimer’s Disease Center at the Mayo Clinic in Scottsdale, Ariz.
I do not find it surprising that older adults fear dementia. Since they correctly perceive that there is no disease-modifying therapy (and maybe also that “getting caught with memory loss” would lead to a loss of driving privileges and other restrictions), they may be trying not to focus on it. As for asking about strategies to “prevent” dementia, that question implies unwarranted optimism about the effectiveness of any such strategy, especially in an older adult. I think we can say that a lifetime of healthy habits (regular physical exercise and careful control of any chronic conditions like diabetes being particularly important) may reduce our risk of dementia a bit, but the idea that anything a 75-year-old does is going to prevent it at that point is probably wishful thinking. Supplements and the like seem to have their own followers. It amazes me how many people suspect what they are taking probably does no good but they do it anyway out of blind hope. Sometimes we can talk them out of spending their money on such things – but not always.
Richard Caselli, MD, is associate director and clinical core director of the Alzheimer’s Disease Center at the Mayo Clinic in Scottsdale, Ariz.
Donovan T. Maust, MD, and colleagues reported in a research letter published in JAMA Neurology.
More than half of study participants used crossword puzzles as a memory exercise, but only 5% said they spoke to their physician about how to reduce risk. Ironically, this lack of communication was also associated with buying unproven over-the-counter memory supplements, while still remaining ignorant of proven ways to head off dementia and other contributing chronic conditions, wrote Dr. Maust of the University of Michigan, Ann Arbor, and coauthors.
Their analysis of the Michigan National Poll on Healthy Aging found that close to half of respondents (48.5%) reported that they were at least somewhat likely to develop dementia. Another 4.2% thought dementia was “very likely” in their future.
The study comprised survey responses from 1,019 adults aged 50-64 years. Most rated their physical health either excellent (445 respondents) or good (413 respondents). Most also reported excellent or very good mental health (721 respondents); 234 reported good mental health. Many (678) were affluent, with annual incomes of $60,000 or higher. They tended to be well educated; only 337 were without at least some college education. More than half were white (753); there were 101 Hispanic respondents and 93 black respondents. Other groups made up the remainder.
A multivariate analysis found that black respondents were about half as likely to believe they would develop dementia, compared with whites – an assumption contrary to epidemiologic findings that blacks are more likely than whites to develop dementia.
People who reported fair or poor mental health were more than twice as likely to feel dementia was in their future (odds ratio, 2.3). But fair or poor physical health was not significantly associated with that concern.
“Those with fair to poor physical health did not accurately perceive that their likelihood of developing dementia was potentially higher than respondents with very good or excellent physical health,” the authors wrote. “In contrast, fair to poor mental health had the largest association with perceived likelihood of dementia, even though less evidence suggests that poor mental health is causally linked with dementia.”
Despite the concerns, just 5% of respondents said that they had spoken to their physician. Those who believed they had a high likelihood of dementia were more likely to talk with their clinician (7.1%) than those who believed they had a low risk (3.6%).
Many more, however, were using non–evidence-based compounds touted as memory supporting. These included fish oil or omega-3 fatty acids (31.6%) and vitamins or supplements (32.9%). Crossword puzzles were a very popular prevention strategy, employed by about 55% in both belief groups.
“While managing chronic medical conditions, such as diabetes or cardiovascular disease, could reduce dementia risk, few respondents appear to have discussed this with their physician. Given repeated failures of disease-preventing or disease-modifying treatments for dementia, interest in treatment and prevention has shifted earlier in the disease process. Adults in middle age may not accurately estimate their risk of developing dementia, which could lead to both overuse and underuse if preclinical dementia treatments become available. Policy and physicians should emphasize current evidence-based strategies of managing lifestyle and chronic medical conditions to reduce the risk of dementia,” the investigators wrote.
Dr. Maust had no financial disclosures.
SOURCE: Maust D et al. JAMA Neurol. 2019 Nov 15. doi: 10.1001/jamaneurol.2019.3946
Donovan T. Maust, MD, and colleagues reported in a research letter published in JAMA Neurology.
More than half of study participants used crossword puzzles as a memory exercise, but only 5% said they spoke to their physician about how to reduce risk. Ironically, this lack of communication was also associated with buying unproven over-the-counter memory supplements, while still remaining ignorant of proven ways to head off dementia and other contributing chronic conditions, wrote Dr. Maust of the University of Michigan, Ann Arbor, and coauthors.
Their analysis of the Michigan National Poll on Healthy Aging found that close to half of respondents (48.5%) reported that they were at least somewhat likely to develop dementia. Another 4.2% thought dementia was “very likely” in their future.
The study comprised survey responses from 1,019 adults aged 50-64 years. Most rated their physical health either excellent (445 respondents) or good (413 respondents). Most also reported excellent or very good mental health (721 respondents); 234 reported good mental health. Many (678) were affluent, with annual incomes of $60,000 or higher. They tended to be well educated; only 337 were without at least some college education. More than half were white (753); there were 101 Hispanic respondents and 93 black respondents. Other groups made up the remainder.
A multivariate analysis found that black respondents were about half as likely to believe they would develop dementia, compared with whites – an assumption contrary to epidemiologic findings that blacks are more likely than whites to develop dementia.
People who reported fair or poor mental health were more than twice as likely to feel dementia was in their future (odds ratio, 2.3). But fair or poor physical health was not significantly associated with that concern.
“Those with fair to poor physical health did not accurately perceive that their likelihood of developing dementia was potentially higher than respondents with very good or excellent physical health,” the authors wrote. “In contrast, fair to poor mental health had the largest association with perceived likelihood of dementia, even though less evidence suggests that poor mental health is causally linked with dementia.”
Despite the concerns, just 5% of respondents said that they had spoken to their physician. Those who believed they had a high likelihood of dementia were more likely to talk with their clinician (7.1%) than those who believed they had a low risk (3.6%).
Many more, however, were using non–evidence-based compounds touted as memory supporting. These included fish oil or omega-3 fatty acids (31.6%) and vitamins or supplements (32.9%). Crossword puzzles were a very popular prevention strategy, employed by about 55% in both belief groups.
“While managing chronic medical conditions, such as diabetes or cardiovascular disease, could reduce dementia risk, few respondents appear to have discussed this with their physician. Given repeated failures of disease-preventing or disease-modifying treatments for dementia, interest in treatment and prevention has shifted earlier in the disease process. Adults in middle age may not accurately estimate their risk of developing dementia, which could lead to both overuse and underuse if preclinical dementia treatments become available. Policy and physicians should emphasize current evidence-based strategies of managing lifestyle and chronic medical conditions to reduce the risk of dementia,” the investigators wrote.
Dr. Maust had no financial disclosures.
SOURCE: Maust D et al. JAMA Neurol. 2019 Nov 15. doi: 10.1001/jamaneurol.2019.3946
FROM JAMA NEUROLOGY
Storytelling tool can assist elderly in the ICU
SAN FRANCISCO – A “Best Case/Worst Case” (BCWC) framework tool has been adapted for use with geriatric trauma patients in the ICU, where it can help track a patient’s progress and enable better communication with patients and loved ones. The tool relies on a combination of graphics and text that surgeons update daily during rounds, and creates a longitudinal view of a patient’s trajectory during their stay in the ICU.
– for example, after a complication has arisen.
“Each day during rounds, the ICU team records important events on the graphic aid that change the patient’s course. The team draws a star to represent the best case, and a line to represent prognostic uncertainty. The attending trauma surgeon then uses the geriatric trauma outcome score, their knowledge of the health state of the patient, and their own clinical experience to tell a story about treatments, recovery, and outcomes if everything goes as well as we might hope. This story is written down in the best-case scenario box,” Christopher Zimmerman, MD, a general surgery resident at the University of Wisconsin–Madison, said during a presentation about the BCWC tool at the annual clinical congress of the American College of Surgeons
“We often like to talk to patients and their families [about best- and worst-case scenarios] anyway, but [the research team] have tried to formalize it,” said Tam Pham, MD, professor of surgery at the University of Washington, in an interview. Dr. Pham comoderated the session where the research was presented.
“When we’re able to communicate where the uncertainty is and where the boundaries are around the course of care and possible outcomes, we can build an alliance with patients and families that will be helpful when there is a big decision to make, say about a laparotomy for a perforated viscus,” said Dr. Zimmerman.
Dr. Zimmerman gave an example of a patient who came into the ICU after suffering multiple fractures from falling down a set of stairs. The team created an initial BCWC with a hoped-for best-case scenario. Later, the patient developed hypoxemic respiratory failure and had to be intubated overnight. “This event is recorded on the graphic, and her star representing the best case has changed position, the line representing uncertainty has shortened, and the contents of her best-case scenario has changed. Each day in rounds, this process is repeated,” said Dr. Zimmerman.
Palliative care physicians, education experts, and surgeons at the University of Wisconsin–Madison developed the tool in an effort to reduce unwanted care at the end of life, in the context of high-risk surgeries. The researchers adapted the tool to the trauma setting by gathering six focus groups of trauma practitioners at the University of Wisconsin; University of Texas, Dallas; and Oregon Health & Science University, Portland. They modified the tool after incorporating comments, and then iteratively modified it through tasks carried out in the ICU as part of a qualitative improvement initiative at the University of Wisconsin–Madison. They generated a change to the tool, implemented it in the ICU during subsequent rounds, then collected observations and field notes, then revised and repeated the process, streamlining it to fit into the ICU environment, according to Dr. Zimmerman.
The back side of the tool is available for family members to write important details about their loved ones, leading insight into the patient’s personality and desires, such as favorite music or affection for a family pet.
The work was supported by the National Institutes of Health. Dr. Zimmerman and Dr. Pham have no relevant financial disclosures.
SOURCE: Zimmerman C et al. Clinical Congress 2019, Abstract.
SAN FRANCISCO – A “Best Case/Worst Case” (BCWC) framework tool has been adapted for use with geriatric trauma patients in the ICU, where it can help track a patient’s progress and enable better communication with patients and loved ones. The tool relies on a combination of graphics and text that surgeons update daily during rounds, and creates a longitudinal view of a patient’s trajectory during their stay in the ICU.
– for example, after a complication has arisen.
“Each day during rounds, the ICU team records important events on the graphic aid that change the patient’s course. The team draws a star to represent the best case, and a line to represent prognostic uncertainty. The attending trauma surgeon then uses the geriatric trauma outcome score, their knowledge of the health state of the patient, and their own clinical experience to tell a story about treatments, recovery, and outcomes if everything goes as well as we might hope. This story is written down in the best-case scenario box,” Christopher Zimmerman, MD, a general surgery resident at the University of Wisconsin–Madison, said during a presentation about the BCWC tool at the annual clinical congress of the American College of Surgeons
“We often like to talk to patients and their families [about best- and worst-case scenarios] anyway, but [the research team] have tried to formalize it,” said Tam Pham, MD, professor of surgery at the University of Washington, in an interview. Dr. Pham comoderated the session where the research was presented.
“When we’re able to communicate where the uncertainty is and where the boundaries are around the course of care and possible outcomes, we can build an alliance with patients and families that will be helpful when there is a big decision to make, say about a laparotomy for a perforated viscus,” said Dr. Zimmerman.
Dr. Zimmerman gave an example of a patient who came into the ICU after suffering multiple fractures from falling down a set of stairs. The team created an initial BCWC with a hoped-for best-case scenario. Later, the patient developed hypoxemic respiratory failure and had to be intubated overnight. “This event is recorded on the graphic, and her star representing the best case has changed position, the line representing uncertainty has shortened, and the contents of her best-case scenario has changed. Each day in rounds, this process is repeated,” said Dr. Zimmerman.
Palliative care physicians, education experts, and surgeons at the University of Wisconsin–Madison developed the tool in an effort to reduce unwanted care at the end of life, in the context of high-risk surgeries. The researchers adapted the tool to the trauma setting by gathering six focus groups of trauma practitioners at the University of Wisconsin; University of Texas, Dallas; and Oregon Health & Science University, Portland. They modified the tool after incorporating comments, and then iteratively modified it through tasks carried out in the ICU as part of a qualitative improvement initiative at the University of Wisconsin–Madison. They generated a change to the tool, implemented it in the ICU during subsequent rounds, then collected observations and field notes, then revised and repeated the process, streamlining it to fit into the ICU environment, according to Dr. Zimmerman.
The back side of the tool is available for family members to write important details about their loved ones, leading insight into the patient’s personality and desires, such as favorite music or affection for a family pet.
The work was supported by the National Institutes of Health. Dr. Zimmerman and Dr. Pham have no relevant financial disclosures.
SOURCE: Zimmerman C et al. Clinical Congress 2019, Abstract.
SAN FRANCISCO – A “Best Case/Worst Case” (BCWC) framework tool has been adapted for use with geriatric trauma patients in the ICU, where it can help track a patient’s progress and enable better communication with patients and loved ones. The tool relies on a combination of graphics and text that surgeons update daily during rounds, and creates a longitudinal view of a patient’s trajectory during their stay in the ICU.
– for example, after a complication has arisen.
“Each day during rounds, the ICU team records important events on the graphic aid that change the patient’s course. The team draws a star to represent the best case, and a line to represent prognostic uncertainty. The attending trauma surgeon then uses the geriatric trauma outcome score, their knowledge of the health state of the patient, and their own clinical experience to tell a story about treatments, recovery, and outcomes if everything goes as well as we might hope. This story is written down in the best-case scenario box,” Christopher Zimmerman, MD, a general surgery resident at the University of Wisconsin–Madison, said during a presentation about the BCWC tool at the annual clinical congress of the American College of Surgeons
“We often like to talk to patients and their families [about best- and worst-case scenarios] anyway, but [the research team] have tried to formalize it,” said Tam Pham, MD, professor of surgery at the University of Washington, in an interview. Dr. Pham comoderated the session where the research was presented.
“When we’re able to communicate where the uncertainty is and where the boundaries are around the course of care and possible outcomes, we can build an alliance with patients and families that will be helpful when there is a big decision to make, say about a laparotomy for a perforated viscus,” said Dr. Zimmerman.
Dr. Zimmerman gave an example of a patient who came into the ICU after suffering multiple fractures from falling down a set of stairs. The team created an initial BCWC with a hoped-for best-case scenario. Later, the patient developed hypoxemic respiratory failure and had to be intubated overnight. “This event is recorded on the graphic, and her star representing the best case has changed position, the line representing uncertainty has shortened, and the contents of her best-case scenario has changed. Each day in rounds, this process is repeated,” said Dr. Zimmerman.
Palliative care physicians, education experts, and surgeons at the University of Wisconsin–Madison developed the tool in an effort to reduce unwanted care at the end of life, in the context of high-risk surgeries. The researchers adapted the tool to the trauma setting by gathering six focus groups of trauma practitioners at the University of Wisconsin; University of Texas, Dallas; and Oregon Health & Science University, Portland. They modified the tool after incorporating comments, and then iteratively modified it through tasks carried out in the ICU as part of a qualitative improvement initiative at the University of Wisconsin–Madison. They generated a change to the tool, implemented it in the ICU during subsequent rounds, then collected observations and field notes, then revised and repeated the process, streamlining it to fit into the ICU environment, according to Dr. Zimmerman.
The back side of the tool is available for family members to write important details about their loved ones, leading insight into the patient’s personality and desires, such as favorite music or affection for a family pet.
The work was supported by the National Institutes of Health. Dr. Zimmerman and Dr. Pham have no relevant financial disclosures.
SOURCE: Zimmerman C et al. Clinical Congress 2019, Abstract.
REPORTING FROM CLINICAL CONGRESS 2019
Worse air quality linked to premature deaths
, according to a working paper issued by the National Bureau of Economic Research.
The increase in air pollution, defined as the amount of fine particulate matter (PM2.5) in the air, was associated with an additional 9,700 premature deaths from 2016 to 2018, representing damages totaling $89 billion, wrote Karen Clay and Nicholas C. Miller of Carnegie Mellon University, Pittsburgh. The increase may reflect in part the impact of the a major wildfire that occurred in the fall of 2018.
“These increases are worrisome, because previous studies have shown that PM2.5 increases premature mortality risk,” the researchers wrote. To assess the changes in air quality, they reviewed data from the Air Quality System (AQS) database including total PM2.5 and three PM2.5 species: ammonium nitrate, sulfate, and elemental carbon.
To examine the impact of pollution on public health, the researchers used data from the damage function approach used in the Environmental Protection Agency’s Benefit-Cost Analysis of the Clean Air Act, the Regulatory Impact Analysis for PM2.5, and multiple academic studies.
The number of premature deaths linked to PM2.5 increased by approximately 4,900 between 2016 and 2017 and by 9,700 from 2016 to 2018 in U.S. counties with monitors.
Elderly individuals are especially vulnerable to particulate matter exposure and experience approximately 80% of the burden of disease related to pollution, the researchers said.
“While some deaths among the elderly are shifted by days or weeks, recent research suggests that the burden is ‘concentrated among the elderly with 5-10 years of remaining life expectancy, followed by those with 2-5 years remaining, because these groups represent a large fraction of the Medicare population and are also vulnerable to acute particulate matter exposure,’” they said.
Overall, pollution levels across the United States stopped declining in 2016. When broken down by four Census regions, no change in PM2.5 levels occurred in the Northeast and South between 2016 and 2018; the Midwest and West showed increases in PM2.5 of 9.3% and 11.5%, respectively.
The researchers suggested three possible factors affecting the increase in pollution: economic activity, wildfires, and air quality enforcement. They noted that increases in PM2.5 were especially high in California, and that California accounted for 43% of the increase in pollution-related premature deaths nationwide between 2016 and 2018. When the researchers examined PM2.5 month by month, “November 2018 had an outsized effect on our mortality calculations,” largely because the devastating Camp Fire occurred in California at that time, they said.
With regard to the impact of economic activity on pollution, the researchers reviewed data from the National Highway Administration and Energy Information Administration that showed increased use of natural gas and increased vehicle travel as contributing to higher levels of nitrate and elemental carbon in the air.
Finally, the researchers reported that enforcement of the Clean Air Act appeared to have declined since 2013, and this decline, although it might reflect increased compliance in some areas “is concerning in light of the increases in air pollution in both attainment and nonattainment counties after 2016,” they said.
The researchers had no financial conflicts to disclose.
SOURCE: Clay K, Miller NZ. NBER 2019. Working Paper 26381. doi: 10.3386/w26381.
, according to a working paper issued by the National Bureau of Economic Research.
The increase in air pollution, defined as the amount of fine particulate matter (PM2.5) in the air, was associated with an additional 9,700 premature deaths from 2016 to 2018, representing damages totaling $89 billion, wrote Karen Clay and Nicholas C. Miller of Carnegie Mellon University, Pittsburgh. The increase may reflect in part the impact of the a major wildfire that occurred in the fall of 2018.
“These increases are worrisome, because previous studies have shown that PM2.5 increases premature mortality risk,” the researchers wrote. To assess the changes in air quality, they reviewed data from the Air Quality System (AQS) database including total PM2.5 and three PM2.5 species: ammonium nitrate, sulfate, and elemental carbon.
To examine the impact of pollution on public health, the researchers used data from the damage function approach used in the Environmental Protection Agency’s Benefit-Cost Analysis of the Clean Air Act, the Regulatory Impact Analysis for PM2.5, and multiple academic studies.
The number of premature deaths linked to PM2.5 increased by approximately 4,900 between 2016 and 2017 and by 9,700 from 2016 to 2018 in U.S. counties with monitors.
Elderly individuals are especially vulnerable to particulate matter exposure and experience approximately 80% of the burden of disease related to pollution, the researchers said.
“While some deaths among the elderly are shifted by days or weeks, recent research suggests that the burden is ‘concentrated among the elderly with 5-10 years of remaining life expectancy, followed by those with 2-5 years remaining, because these groups represent a large fraction of the Medicare population and are also vulnerable to acute particulate matter exposure,’” they said.
Overall, pollution levels across the United States stopped declining in 2016. When broken down by four Census regions, no change in PM2.5 levels occurred in the Northeast and South between 2016 and 2018; the Midwest and West showed increases in PM2.5 of 9.3% and 11.5%, respectively.
The researchers suggested three possible factors affecting the increase in pollution: economic activity, wildfires, and air quality enforcement. They noted that increases in PM2.5 were especially high in California, and that California accounted for 43% of the increase in pollution-related premature deaths nationwide between 2016 and 2018. When the researchers examined PM2.5 month by month, “November 2018 had an outsized effect on our mortality calculations,” largely because the devastating Camp Fire occurred in California at that time, they said.
With regard to the impact of economic activity on pollution, the researchers reviewed data from the National Highway Administration and Energy Information Administration that showed increased use of natural gas and increased vehicle travel as contributing to higher levels of nitrate and elemental carbon in the air.
Finally, the researchers reported that enforcement of the Clean Air Act appeared to have declined since 2013, and this decline, although it might reflect increased compliance in some areas “is concerning in light of the increases in air pollution in both attainment and nonattainment counties after 2016,” they said.
The researchers had no financial conflicts to disclose.
SOURCE: Clay K, Miller NZ. NBER 2019. Working Paper 26381. doi: 10.3386/w26381.
, according to a working paper issued by the National Bureau of Economic Research.
The increase in air pollution, defined as the amount of fine particulate matter (PM2.5) in the air, was associated with an additional 9,700 premature deaths from 2016 to 2018, representing damages totaling $89 billion, wrote Karen Clay and Nicholas C. Miller of Carnegie Mellon University, Pittsburgh. The increase may reflect in part the impact of the a major wildfire that occurred in the fall of 2018.
“These increases are worrisome, because previous studies have shown that PM2.5 increases premature mortality risk,” the researchers wrote. To assess the changes in air quality, they reviewed data from the Air Quality System (AQS) database including total PM2.5 and three PM2.5 species: ammonium nitrate, sulfate, and elemental carbon.
To examine the impact of pollution on public health, the researchers used data from the damage function approach used in the Environmental Protection Agency’s Benefit-Cost Analysis of the Clean Air Act, the Regulatory Impact Analysis for PM2.5, and multiple academic studies.
The number of premature deaths linked to PM2.5 increased by approximately 4,900 between 2016 and 2017 and by 9,700 from 2016 to 2018 in U.S. counties with monitors.
Elderly individuals are especially vulnerable to particulate matter exposure and experience approximately 80% of the burden of disease related to pollution, the researchers said.
“While some deaths among the elderly are shifted by days or weeks, recent research suggests that the burden is ‘concentrated among the elderly with 5-10 years of remaining life expectancy, followed by those with 2-5 years remaining, because these groups represent a large fraction of the Medicare population and are also vulnerable to acute particulate matter exposure,’” they said.
Overall, pollution levels across the United States stopped declining in 2016. When broken down by four Census regions, no change in PM2.5 levels occurred in the Northeast and South between 2016 and 2018; the Midwest and West showed increases in PM2.5 of 9.3% and 11.5%, respectively.
The researchers suggested three possible factors affecting the increase in pollution: economic activity, wildfires, and air quality enforcement. They noted that increases in PM2.5 were especially high in California, and that California accounted for 43% of the increase in pollution-related premature deaths nationwide between 2016 and 2018. When the researchers examined PM2.5 month by month, “November 2018 had an outsized effect on our mortality calculations,” largely because the devastating Camp Fire occurred in California at that time, they said.
With regard to the impact of economic activity on pollution, the researchers reviewed data from the National Highway Administration and Energy Information Administration that showed increased use of natural gas and increased vehicle travel as contributing to higher levels of nitrate and elemental carbon in the air.
Finally, the researchers reported that enforcement of the Clean Air Act appeared to have declined since 2013, and this decline, although it might reflect increased compliance in some areas “is concerning in light of the increases in air pollution in both attainment and nonattainment counties after 2016,” they said.
The researchers had no financial conflicts to disclose.
SOURCE: Clay K, Miller NZ. NBER 2019. Working Paper 26381. doi: 10.3386/w26381.
FROM AN NBER AIR QUALITY STUDY
Probiotics with Lactobacillus reduce loss in spine BMD for postmenopausal women
, according to recent research published in The Lancet Rheumatology.
“The menopausal and early postmenopausal lumbar spine bone loss is substantial in women, and by using a prevention therapy with bacteria naturally occurring in the human gut microbiota we observed a close to complete protection against lumbar spine bone loss in healthy postmenopausal women,” Per-Anders Jansson, MD, chief physician at the University of Gothenburg (Sweden), and colleagues wrote in their study.
Dr. Jansson and colleagues performed a double-blind trial at four centers in Sweden in which 249 postmenopausal women were randomized during April-November 2016 to receive probiotics consisting of three Lactobacillus strains or placebo once per day for 12 months. Participants were healthy women, neither underweight nor overweight, and were postmenopausal, which was defined as being 2-12 years or less from last menstruation. The Lactobacillus strains, L. paracasei 8700:2 (DSM 13434), L. plantarum Heal 9 (DSM 15312), and L. plantarum Heal 19 (DSM 15313), were equally represented in a capsule at a dose of 1 x 1010 colony-forming unit per capsule. The researchers measured the lumbar spine bone mineral density (LS-BMD) at baseline and at 12 months, and also evaluated the safety profile of participants in both the probiotic and placebo groups.
Overall, 234 participants (94%) had data available for analysis at the end of the study. There was a significant reduction in LS-BMD loss for participants who received the probiotic treatment, compared with women in the control group (mean difference, 0.71%; 95% confidence interval, 0.06%-1.35%), while there was a significant loss in LS-BMD for participants in the placebo group (percentage change, –0.72%; 95% CI, –1.22% to –0.22%) compared with loss in the probiotic group (percentage change, –0.01%; 95% CI, –0.50% to 0.48%). Using analysis of covariance, the researchers found the probiotic group had reduced LS-BMD loss after adjustment for factors such as study site, age at baseline, BMD at baseline, and number of years from menopause (mean difference, 7.44 mg/cm2; 95% CI, 0.38 to 14.50).
In a subgroup analysis of women above and below the median time since menopause at baseline (6 years), participants in the probiotic group who were below the median time saw a significant protective effect of Lactobacillus treatment (mean difference, 1.08%; 95% CI, 0.20%-1.96%), compared with women above the median time (mean difference, 0.31%; 95% CI, –0.62% to 1.23%).
Researchers also examined the effects of probiotic treatment on total hip and femoral neck BMD as secondary endpoints. Lactobacillus treatment did not appear to affect total hip (–1.01%; 95% CI, –1.65% to –0.37%) or trochanter BMD (–1.13%; 95% CI, –2.27% to 0.20%), but femoral neck BMD was reduced in the probiotic group (–1.34%; 95% CI, –2.09% to –0.58%), compared with the placebo group (–0.88%; 95% CI, –1.64% to –0.13%).
Limitations of the study included examining only one dose of Lactobacillus treatment and no analysis of the effect of short-chain fatty acids on LS-BMD. The researchers noted that “recent studies have shown that short-chain fatty acids, which are generated by fermentation of complex carbohydrates by the gut microbiota, are important regulators of both bone formation and resorption.”
The researchers also acknowledged that the LS-BMD effect size for the probiotic treatment over the 12 months was a lower magnitude, compared with first-line treatments for osteoporosis in postmenopausal women using bisphosphonates. “Further long-term studies should be done to evaluate if the bone-protective effect becomes more pronounced with prolonged treatment with the Lactobacillus strains used in the present study,” they said.
In a related editorial, Shivani Sahni, PhD, of Harvard Medical School, Boston, and Connie M. Weaver, PhD, of Purdue University, West Lafayette, Ind., reiterated that the effect size of probiotics is “of far less magnitude” than such treatments as bisphosphonates and expressed concern about the reduction of femoral neck BMD in the probiotic group, which was not explained in the study (Lancet Rheumatol. 2019 Nov;1[3]:e135-e137. doi: 10.1016/S2665-9913(19)30073-6). There is a need to learn the optimum dose of probiotics as well as which Lactobacillus strains should be used in future studies, as the strains chosen by Jansson et al. were based on results in mice.
In the meantime, patients might be better off choosing dietary interventions with proven bone protection and no documented negative effects on the hip, such as prebiotics like soluble corn fiber and dried prunes, in tandem with drug therapies, Dr. Sahni and Dr. Weaver said.
“Although Jansson and colleagues’ results are important, more work is needed before such probiotics are ready for consumers,” they concluded.
This study was funded by Probi, which employs two of the study’s authors. Three authors reported being coinventors of a patent involving the effects of probiotics in osteoporosis treatment, and one author is listed as an inventor on a pending patent application on probiotic compositions and uses. Dr. Sahni reported receiving grants from Dairy Management. Dr. Weaver reported no relevant conflicts of interest.
SOURCE: Jansson P-A et al. Lancet Rheumatol. 2019 Nov;1(3):e154-e162. doi: 10.1016/S2665-9913(19)30068-2
, according to recent research published in The Lancet Rheumatology.
“The menopausal and early postmenopausal lumbar spine bone loss is substantial in women, and by using a prevention therapy with bacteria naturally occurring in the human gut microbiota we observed a close to complete protection against lumbar spine bone loss in healthy postmenopausal women,” Per-Anders Jansson, MD, chief physician at the University of Gothenburg (Sweden), and colleagues wrote in their study.
Dr. Jansson and colleagues performed a double-blind trial at four centers in Sweden in which 249 postmenopausal women were randomized during April-November 2016 to receive probiotics consisting of three Lactobacillus strains or placebo once per day for 12 months. Participants were healthy women, neither underweight nor overweight, and were postmenopausal, which was defined as being 2-12 years or less from last menstruation. The Lactobacillus strains, L. paracasei 8700:2 (DSM 13434), L. plantarum Heal 9 (DSM 15312), and L. plantarum Heal 19 (DSM 15313), were equally represented in a capsule at a dose of 1 x 1010 colony-forming unit per capsule. The researchers measured the lumbar spine bone mineral density (LS-BMD) at baseline and at 12 months, and also evaluated the safety profile of participants in both the probiotic and placebo groups.
Overall, 234 participants (94%) had data available for analysis at the end of the study. There was a significant reduction in LS-BMD loss for participants who received the probiotic treatment, compared with women in the control group (mean difference, 0.71%; 95% confidence interval, 0.06%-1.35%), while there was a significant loss in LS-BMD for participants in the placebo group (percentage change, –0.72%; 95% CI, –1.22% to –0.22%) compared with loss in the probiotic group (percentage change, –0.01%; 95% CI, –0.50% to 0.48%). Using analysis of covariance, the researchers found the probiotic group had reduced LS-BMD loss after adjustment for factors such as study site, age at baseline, BMD at baseline, and number of years from menopause (mean difference, 7.44 mg/cm2; 95% CI, 0.38 to 14.50).
In a subgroup analysis of women above and below the median time since menopause at baseline (6 years), participants in the probiotic group who were below the median time saw a significant protective effect of Lactobacillus treatment (mean difference, 1.08%; 95% CI, 0.20%-1.96%), compared with women above the median time (mean difference, 0.31%; 95% CI, –0.62% to 1.23%).
Researchers also examined the effects of probiotic treatment on total hip and femoral neck BMD as secondary endpoints. Lactobacillus treatment did not appear to affect total hip (–1.01%; 95% CI, –1.65% to –0.37%) or trochanter BMD (–1.13%; 95% CI, –2.27% to 0.20%), but femoral neck BMD was reduced in the probiotic group (–1.34%; 95% CI, –2.09% to –0.58%), compared with the placebo group (–0.88%; 95% CI, –1.64% to –0.13%).
Limitations of the study included examining only one dose of Lactobacillus treatment and no analysis of the effect of short-chain fatty acids on LS-BMD. The researchers noted that “recent studies have shown that short-chain fatty acids, which are generated by fermentation of complex carbohydrates by the gut microbiota, are important regulators of both bone formation and resorption.”
The researchers also acknowledged that the LS-BMD effect size for the probiotic treatment over the 12 months was a lower magnitude, compared with first-line treatments for osteoporosis in postmenopausal women using bisphosphonates. “Further long-term studies should be done to evaluate if the bone-protective effect becomes more pronounced with prolonged treatment with the Lactobacillus strains used in the present study,” they said.
In a related editorial, Shivani Sahni, PhD, of Harvard Medical School, Boston, and Connie M. Weaver, PhD, of Purdue University, West Lafayette, Ind., reiterated that the effect size of probiotics is “of far less magnitude” than such treatments as bisphosphonates and expressed concern about the reduction of femoral neck BMD in the probiotic group, which was not explained in the study (Lancet Rheumatol. 2019 Nov;1[3]:e135-e137. doi: 10.1016/S2665-9913(19)30073-6). There is a need to learn the optimum dose of probiotics as well as which Lactobacillus strains should be used in future studies, as the strains chosen by Jansson et al. were based on results in mice.
In the meantime, patients might be better off choosing dietary interventions with proven bone protection and no documented negative effects on the hip, such as prebiotics like soluble corn fiber and dried prunes, in tandem with drug therapies, Dr. Sahni and Dr. Weaver said.
“Although Jansson and colleagues’ results are important, more work is needed before such probiotics are ready for consumers,” they concluded.
This study was funded by Probi, which employs two of the study’s authors. Three authors reported being coinventors of a patent involving the effects of probiotics in osteoporosis treatment, and one author is listed as an inventor on a pending patent application on probiotic compositions and uses. Dr. Sahni reported receiving grants from Dairy Management. Dr. Weaver reported no relevant conflicts of interest.
SOURCE: Jansson P-A et al. Lancet Rheumatol. 2019 Nov;1(3):e154-e162. doi: 10.1016/S2665-9913(19)30068-2
, according to recent research published in The Lancet Rheumatology.
“The menopausal and early postmenopausal lumbar spine bone loss is substantial in women, and by using a prevention therapy with bacteria naturally occurring in the human gut microbiota we observed a close to complete protection against lumbar spine bone loss in healthy postmenopausal women,” Per-Anders Jansson, MD, chief physician at the University of Gothenburg (Sweden), and colleagues wrote in their study.
Dr. Jansson and colleagues performed a double-blind trial at four centers in Sweden in which 249 postmenopausal women were randomized during April-November 2016 to receive probiotics consisting of three Lactobacillus strains or placebo once per day for 12 months. Participants were healthy women, neither underweight nor overweight, and were postmenopausal, which was defined as being 2-12 years or less from last menstruation. The Lactobacillus strains, L. paracasei 8700:2 (DSM 13434), L. plantarum Heal 9 (DSM 15312), and L. plantarum Heal 19 (DSM 15313), were equally represented in a capsule at a dose of 1 x 1010 colony-forming unit per capsule. The researchers measured the lumbar spine bone mineral density (LS-BMD) at baseline and at 12 months, and also evaluated the safety profile of participants in both the probiotic and placebo groups.
Overall, 234 participants (94%) had data available for analysis at the end of the study. There was a significant reduction in LS-BMD loss for participants who received the probiotic treatment, compared with women in the control group (mean difference, 0.71%; 95% confidence interval, 0.06%-1.35%), while there was a significant loss in LS-BMD for participants in the placebo group (percentage change, –0.72%; 95% CI, –1.22% to –0.22%) compared with loss in the probiotic group (percentage change, –0.01%; 95% CI, –0.50% to 0.48%). Using analysis of covariance, the researchers found the probiotic group had reduced LS-BMD loss after adjustment for factors such as study site, age at baseline, BMD at baseline, and number of years from menopause (mean difference, 7.44 mg/cm2; 95% CI, 0.38 to 14.50).
In a subgroup analysis of women above and below the median time since menopause at baseline (6 years), participants in the probiotic group who were below the median time saw a significant protective effect of Lactobacillus treatment (mean difference, 1.08%; 95% CI, 0.20%-1.96%), compared with women above the median time (mean difference, 0.31%; 95% CI, –0.62% to 1.23%).
Researchers also examined the effects of probiotic treatment on total hip and femoral neck BMD as secondary endpoints. Lactobacillus treatment did not appear to affect total hip (–1.01%; 95% CI, –1.65% to –0.37%) or trochanter BMD (–1.13%; 95% CI, –2.27% to 0.20%), but femoral neck BMD was reduced in the probiotic group (–1.34%; 95% CI, –2.09% to –0.58%), compared with the placebo group (–0.88%; 95% CI, –1.64% to –0.13%).
Limitations of the study included examining only one dose of Lactobacillus treatment and no analysis of the effect of short-chain fatty acids on LS-BMD. The researchers noted that “recent studies have shown that short-chain fatty acids, which are generated by fermentation of complex carbohydrates by the gut microbiota, are important regulators of both bone formation and resorption.”
The researchers also acknowledged that the LS-BMD effect size for the probiotic treatment over the 12 months was a lower magnitude, compared with first-line treatments for osteoporosis in postmenopausal women using bisphosphonates. “Further long-term studies should be done to evaluate if the bone-protective effect becomes more pronounced with prolonged treatment with the Lactobacillus strains used in the present study,” they said.
In a related editorial, Shivani Sahni, PhD, of Harvard Medical School, Boston, and Connie M. Weaver, PhD, of Purdue University, West Lafayette, Ind., reiterated that the effect size of probiotics is “of far less magnitude” than such treatments as bisphosphonates and expressed concern about the reduction of femoral neck BMD in the probiotic group, which was not explained in the study (Lancet Rheumatol. 2019 Nov;1[3]:e135-e137. doi: 10.1016/S2665-9913(19)30073-6). There is a need to learn the optimum dose of probiotics as well as which Lactobacillus strains should be used in future studies, as the strains chosen by Jansson et al. were based on results in mice.
In the meantime, patients might be better off choosing dietary interventions with proven bone protection and no documented negative effects on the hip, such as prebiotics like soluble corn fiber and dried prunes, in tandem with drug therapies, Dr. Sahni and Dr. Weaver said.
“Although Jansson and colleagues’ results are important, more work is needed before such probiotics are ready for consumers,” they concluded.
This study was funded by Probi, which employs two of the study’s authors. Three authors reported being coinventors of a patent involving the effects of probiotics in osteoporosis treatment, and one author is listed as an inventor on a pending patent application on probiotic compositions and uses. Dr. Sahni reported receiving grants from Dairy Management. Dr. Weaver reported no relevant conflicts of interest.
SOURCE: Jansson P-A et al. Lancet Rheumatol. 2019 Nov;1(3):e154-e162. doi: 10.1016/S2665-9913(19)30068-2
FROM THE LANCET RHEUMATOLOGY
Evaluating a Veterans Affairs Home-Based Primary Care Population for Patients at High Risk of Osteoporosis
Osteoporosis is a disease characterized by the loss of bone density.1 Bone is normally porous and is in a state of flux due to changes in regeneration caused by osteoclast or osteoblast activity. However, age and other factors can accelerate loss in bone density and lead to decreased bone strength and an increased risk of fracture. In men, bone mineral density (BMD) can begin to decline as early as age 30 to 40 years. By age 80 years, 25% of total bone mass may be lost.2
Of the 44 million Americans with low BMD or osteoporosis, 20% are men.1 This group accounts for up to 40% of all osteoporotic fractures. About 1 in 4 men aged ≥ 50 years may experience a lifetime fracture. Fractures may lead to chronic pain, disability, increased dependence, and potentially death. These complications cause expenditures upward of $4.1 billion annually in North America alone.3,4 About 80,000 US men will experience a hip fracture each year, one-third of whom will die within that year. This constitutes a mortality rate 2 to 3 times higher than that of women. Osteoporosis often goes undiagnosed and untreated due to a lack of symptoms until a fracture occurs, underlining the potential benefit of preemptive screening.
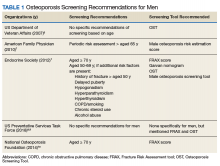
In 2007, Shekell and colleagues outlined how the US Department of Veterans Affairs (VA) screened men for osteoporosis.5 At the time, 95% of the VA population was male, though it has since dropped to 91%.6 Shekell and colleagues estimated that about 200,0000 to 400,0000 male veterans had osteoporosis.5 Osteoporotic risk factors deemed specific to veterans were excessive alcohol use, spinal cord injury and lack of weight-bearing exercise, prolonged corticosteroid use, and androgen deprivation therapy in prostate cancer. Different screening techniques were assessed, and the VA recommended the Osteoporosis Self-Assessment Tool (OST).5 Many organizations have developed clinical guidance, including who should be screened; however, screening for men remains a controversial area due to a lack of any strong recommendations (Table 1).
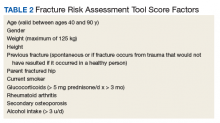
Endocrine Society screening guidelines for men are the most specific: testing BMD in men aged ≥ 70 years, or if aged 50 to 69 years with an additional risk factor (eg, low body weight, smoking, chronic obstructive pulmonary disease, chronic steroid use).1 The Fracture Risk Assessment tool (FRAX) score is often cited as a common screening tool. It is a free online questionnaire that provides a 10-year probability risk of hip or major osteoporotic fracture.11 However, this tool is limited by age, weight, and the assumption that all questions are answered accurately. Some of the information required includes the presence of a number of risk factors, such as alcohol use, glucocorticoids, and medical history of rheumatoid arthritis, among others (Table 2). The OST score, on the other hand, is a calculation that does not take into account other risk factors (Figure 1). This tool categorizes the patient into low, moderate, or high risk for osteoporosis.8
In a study of 4,000 men aged ≥ 70 years,
A 2017 VA Office of Rural Health study examined the utility of OST to screen referred patients aged > 50 years to receive DEXA scans in patient aligned care team (PACT) clinics at 3 different VA locations.13 The study excluded patients who had been screened previously or treated for osteoporosis, were receiving hospice care; 1 site excluded patients aged > 88 years. Two of the sites also reviewed the patient’s medications to screen for agents that may contribute to increased fracture risk. Veterans identified as high risk were referred for education and offered a DEXA scan and treatment. In total, 867 veterans were screened; 19% (168) were deemed high risk, and 6% (53) underwent DEXA scans. The study noted that only 15 patients had reportable DEXA scans and 10 were positive for bone disease.
As there has been documented success in the PACT setting in implementing standardized protocols for screening and treating veterans, it is reasonable to extend the concept into other VA services. The home-based primary care (HBPC) population is especially vulnerable due to the age of patients, limited weight-bearing exercise to improve bone strength, and limited access to DEXA scans due to difficulty traveling outside of the home. Despite these issues, a goal of the HBPC service is to provide continual care for veterans and improve their health so they may return to the community setting. As a result, patients are followed frequently, providing many opportunities for interventions. This study aims to determine the proportion of HBPC patients who are at high risk for osteoporosis and can receive a DEXA scan for evaluation.
Methods
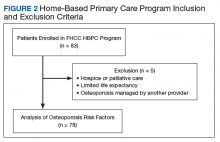
This study was a retrospective chart analysis using descriptive statistics. It was reviewed and approved by the institutional review board at Captain James A. Lovell Federal Health Care Center (FHCC). Patients were included in the study if they were enrolled in the HBPC program at FHCC. Patients were excluded if they were receiving hospice or palliative care, had a limited life expectancy per the HBPC provider, or had a diagnosis of osteoporosis that was being managed by a VA endocrinologist, rheumatologist, or non-VA provider.
The study was conducted from February 1, 2018, through November 30, 2018. All chart reviews were done through the FHCC electronic health record. A minimum of 80 and maximum of 150 charts were reviewed as this was the typical patient volume in the HBPC program. Basic demographic information was collected and analyzed by calculating FRAX and OST scores. With the results, patients were classified as low or high risk of developing osteoporosis, and whether a DEXA scan should be recommended.
Results
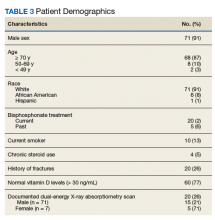
After chart review, 83 patients were enrolled in the FHCC HBPC program during the study period. Out of these, 5 patients were excluded due to hospice or palliative care status, limited life expectancy, or had their osteoporosis managed by another non-HBPC provider. As a result, 78 patients were analyzed to determine their risk of osteoporosis (Figure 2). Most of the patients were white males with a median age of 82 years. A majority of the patients did not have any current or previous treatment with bisphosphonates, 77% had normal vitamin D levels, and only 13% (10) were current smokers; of the male patients only 21% (15) had a previous DEXA scan (Table 3).
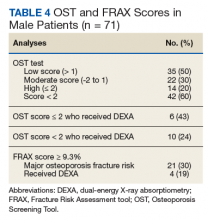
The FRAX and OST scores for each male patient were calculated (Table 4). Half the patients were low risk for osteoporosis. Just 20% (14) of the patients were at high risk for osteoporosis, and only 6 of those had DEXA scans. However, if expanding the criteria to OST scores of < 2, then only 24% (10) received DEXA scans. When calculating FRAX scores, 30% (21) had ≥ 9.3% for major osteoporotic fracture risk, and only 19% (4) had received a DEXA scan.
Discussion
Based on the collected data, many of the male HBPC patients have not had an evaluation for osteoporosis despite being in a high-risk population and meeting some of the screening guidelines by various organizations.1 Based on Diem and colleagues and the 2007 VA report, utilizing OST scores could help capture a subset of patients that would be referred for DEXA scans.5,12 Of the 60% (42) of patients that met OST scores of < 2, 76% (32) of them could have been referred for DEXA scans for osteoporosis evaluation. However, at the time of publication of this article, 50% (16) of the patients have been discharged from the service without interventions. Of the remaining 16 patients, only 2 were referred for a DEXA scan, and 1 patient had confirmed osteoporosis. Currently, these results have been reviewed by the HBPC provider, and plans are in place for DEXA scan referrals for the remaining patients. In addition, for new patients admitted to the program and during annual reviews, the plan is to use OST scores to help screen for osteoporosis.
Limitations
The HBPC population is often in flux due to discharges as patients pass away, become eligible for long-term care, advance to hospice or palliative care status, or see an improvement in their condition to transition back into the community. Along with patients who are bed-bound, have poor prognosis, and barriers to access (eg, transportation issues), interventions for DEXA scan referrals are often not clinically indicated. During calculations of the FRAX score, documentation is often missing from a patient’s medical chart, making it difficult to answer all questions on the questionnaire. This does increase the utility of the OST score as the calculation is much easier and does not rely on other osteoporotic factors. Despite these restrictions for offering DEXA scans, the HBPC service has a high standard of excellence in preventing falls, a major contributor to fractures. Physical therapy services are readily available, nursing visits are frequent and as clinically indicated, vitamin D levels are maintained within normal limits via supplementation, and medication management is performed at least quarterly among other interventions.
Conclusions
The retrospective chart review of patients in the HBPC program suggests that there may be a lack of standardized screening for osteoporosis in the male patient population. As seen within the data, there is great potential for interventions as many of the patients would be candidates for screening based on the OST score. The tool is easy to use and readily accessible to all health care providers and staff. By increasing screening of eligible patients, it also increases the identification of those who would benefit from osteoporosis treatment. While the HBPC population has access limitations (eg, homebound, limited life expectancy), the implementation of a protocol and extension of concepts from this study can be extrapolated into other PACT clinics at VA facilities. Osteoporosis in the male population is often overlooked, but screening procedures can help reduce health care expenditures.
1. Watts NB, Adler RA, Bilezikian JP, et al; Endocrine Society. Osteoporosis in men: an Endocrine Society clinical practice guideline. J Clin Endocrinol Metab. 2012;97(6):1802-1822.
2. Holt G, Smith R, Duncan K, Hutchison JD, Gregori A. Gender differences in epidemiology and outcome after hip fracture: evidence from the Scottish Hip Fracture Audit. J Bone Joint Surg Br. 2008;90(4):480-483.
3. Ackman JM, Lata PF, Schuna AA, Elliott ME. Bone health evaluation in a veteran population: a need for the Fracture Risk Assessment tool (FRAX). Ann Pharmacother. 2014;48(10):1288-1293.
4. International Osteoporosis Foundation. Osteoporosis in men: why change needs to happen. http://share.iofbone-health.org/WOD/2014/thematic-report/WOD14-Report.pdf. Published 2014. Accessed September 16, 2019.
5. Shekell P, Munjas B, Liu H, et al. Screening Men for Osteoporosis: Who & How. Evidence-based Synthesis Program. Washington, DC: Department of Veterans Affairs; 2007.
6. US Department of Veterans Affairs, National Center for Veterans Analysis and Statistics. Veteran population. https://www.va.gov/vetdata/Veteran_Population.asp. Accessed September 16, 2019.
7. Rao SS, Budhwar N, Ashfaque A. Osteoporosis in men. Am Fam Physician. 2010;82(5):503-508.
8. US Preventive Services Task Force, Curry SJ, Krist AH, et al. Screening for osteoporosis to prevent fractures: US Preventive Services Task Force recommendation statement. JAMA. 2018;319(24):2521-2531.
9. Viswanathan M, Reddy S, Berkman N, et al. Screening to prevent osteoporotic fractures updated evidence report and systematic review for the US Preventive Services Task Force. JAMA. 2018;319(24):2532-2551.
10. Cosman F, de Beur SJ, LeBoff MS, et al; National Osteoporosis Foundation. Clinician’s guide to prevention and treatment of osteoporosis. Osteoporos Int. 2014;25(10):2359-2381.
11. Centre for Metabolic Bone Diseases, University of Sheffield, UK. FRAX Fracture Risk Assessment Tool. http://www.sheffield.ac.uk/FRAX/tool.aspx?country=9. Accessed September 16, 2019.
12. Diem SJ, Peters KW, Gourlay ML, et al; Osteoporotic Fractures in Men Research Group. Screening for osteoporosis in older men: operating characteristics of proposed strategies for selecting men for BMD testing. J Gen Intern Med. 2017;32(11):1235-1241.
13. US Department of Veterans Affairs, Office of Rural Health. Osteoporosis risk assessment using Osteoporosis Self-Assessment Tool (OST) and other interventions at rural facilities. https://www.ruralhealth.va.gov/docs/promise/2017_02_01_OST_Issue%20Brief_v2.pdf. Published February 7, 2019. Accessed September 16, 2019.
Osteoporosis is a disease characterized by the loss of bone density.1 Bone is normally porous and is in a state of flux due to changes in regeneration caused by osteoclast or osteoblast activity. However, age and other factors can accelerate loss in bone density and lead to decreased bone strength and an increased risk of fracture. In men, bone mineral density (BMD) can begin to decline as early as age 30 to 40 years. By age 80 years, 25% of total bone mass may be lost.2
Of the 44 million Americans with low BMD or osteoporosis, 20% are men.1 This group accounts for up to 40% of all osteoporotic fractures. About 1 in 4 men aged ≥ 50 years may experience a lifetime fracture. Fractures may lead to chronic pain, disability, increased dependence, and potentially death. These complications cause expenditures upward of $4.1 billion annually in North America alone.3,4 About 80,000 US men will experience a hip fracture each year, one-third of whom will die within that year. This constitutes a mortality rate 2 to 3 times higher than that of women. Osteoporosis often goes undiagnosed and untreated due to a lack of symptoms until a fracture occurs, underlining the potential benefit of preemptive screening.
In 2007, Shekell and colleagues outlined how the US Department of Veterans Affairs (VA) screened men for osteoporosis.5 At the time, 95% of the VA population was male, though it has since dropped to 91%.6 Shekell and colleagues estimated that about 200,0000 to 400,0000 male veterans had osteoporosis.5 Osteoporotic risk factors deemed specific to veterans were excessive alcohol use, spinal cord injury and lack of weight-bearing exercise, prolonged corticosteroid use, and androgen deprivation therapy in prostate cancer. Different screening techniques were assessed, and the VA recommended the Osteoporosis Self-Assessment Tool (OST).5 Many organizations have developed clinical guidance, including who should be screened; however, screening for men remains a controversial area due to a lack of any strong recommendations (Table 1).
Endocrine Society screening guidelines for men are the most specific: testing BMD in men aged ≥ 70 years, or if aged 50 to 69 years with an additional risk factor (eg, low body weight, smoking, chronic obstructive pulmonary disease, chronic steroid use).1 The Fracture Risk Assessment tool (FRAX) score is often cited as a common screening tool. It is a free online questionnaire that provides a 10-year probability risk of hip or major osteoporotic fracture.11 However, this tool is limited by age, weight, and the assumption that all questions are answered accurately. Some of the information required includes the presence of a number of risk factors, such as alcohol use, glucocorticoids, and medical history of rheumatoid arthritis, among others (Table 2). The OST score, on the other hand, is a calculation that does not take into account other risk factors (Figure 1). This tool categorizes the patient into low, moderate, or high risk for osteoporosis.8
In a study of 4,000 men aged ≥ 70 years,
A 2017 VA Office of Rural Health study examined the utility of OST to screen referred patients aged > 50 years to receive DEXA scans in patient aligned care team (PACT) clinics at 3 different VA locations.13 The study excluded patients who had been screened previously or treated for osteoporosis, were receiving hospice care; 1 site excluded patients aged > 88 years. Two of the sites also reviewed the patient’s medications to screen for agents that may contribute to increased fracture risk. Veterans identified as high risk were referred for education and offered a DEXA scan and treatment. In total, 867 veterans were screened; 19% (168) were deemed high risk, and 6% (53) underwent DEXA scans. The study noted that only 15 patients had reportable DEXA scans and 10 were positive for bone disease.
As there has been documented success in the PACT setting in implementing standardized protocols for screening and treating veterans, it is reasonable to extend the concept into other VA services. The home-based primary care (HBPC) population is especially vulnerable due to the age of patients, limited weight-bearing exercise to improve bone strength, and limited access to DEXA scans due to difficulty traveling outside of the home. Despite these issues, a goal of the HBPC service is to provide continual care for veterans and improve their health so they may return to the community setting. As a result, patients are followed frequently, providing many opportunities for interventions. This study aims to determine the proportion of HBPC patients who are at high risk for osteoporosis and can receive a DEXA scan for evaluation.
Methods
This study was a retrospective chart analysis using descriptive statistics. It was reviewed and approved by the institutional review board at Captain James A. Lovell Federal Health Care Center (FHCC). Patients were included in the study if they were enrolled in the HBPC program at FHCC. Patients were excluded if they were receiving hospice or palliative care, had a limited life expectancy per the HBPC provider, or had a diagnosis of osteoporosis that was being managed by a VA endocrinologist, rheumatologist, or non-VA provider.
The study was conducted from February 1, 2018, through November 30, 2018. All chart reviews were done through the FHCC electronic health record. A minimum of 80 and maximum of 150 charts were reviewed as this was the typical patient volume in the HBPC program. Basic demographic information was collected and analyzed by calculating FRAX and OST scores. With the results, patients were classified as low or high risk of developing osteoporosis, and whether a DEXA scan should be recommended.
Results
After chart review, 83 patients were enrolled in the FHCC HBPC program during the study period. Out of these, 5 patients were excluded due to hospice or palliative care status, limited life expectancy, or had their osteoporosis managed by another non-HBPC provider. As a result, 78 patients were analyzed to determine their risk of osteoporosis (Figure 2). Most of the patients were white males with a median age of 82 years. A majority of the patients did not have any current or previous treatment with bisphosphonates, 77% had normal vitamin D levels, and only 13% (10) were current smokers; of the male patients only 21% (15) had a previous DEXA scan (Table 3).
The FRAX and OST scores for each male patient were calculated (Table 4). Half the patients were low risk for osteoporosis. Just 20% (14) of the patients were at high risk for osteoporosis, and only 6 of those had DEXA scans. However, if expanding the criteria to OST scores of < 2, then only 24% (10) received DEXA scans. When calculating FRAX scores, 30% (21) had ≥ 9.3% for major osteoporotic fracture risk, and only 19% (4) had received a DEXA scan.
Discussion
Based on the collected data, many of the male HBPC patients have not had an evaluation for osteoporosis despite being in a high-risk population and meeting some of the screening guidelines by various organizations.1 Based on Diem and colleagues and the 2007 VA report, utilizing OST scores could help capture a subset of patients that would be referred for DEXA scans.5,12 Of the 60% (42) of patients that met OST scores of < 2, 76% (32) of them could have been referred for DEXA scans for osteoporosis evaluation. However, at the time of publication of this article, 50% (16) of the patients have been discharged from the service without interventions. Of the remaining 16 patients, only 2 were referred for a DEXA scan, and 1 patient had confirmed osteoporosis. Currently, these results have been reviewed by the HBPC provider, and plans are in place for DEXA scan referrals for the remaining patients. In addition, for new patients admitted to the program and during annual reviews, the plan is to use OST scores to help screen for osteoporosis.
Limitations
The HBPC population is often in flux due to discharges as patients pass away, become eligible for long-term care, advance to hospice or palliative care status, or see an improvement in their condition to transition back into the community. Along with patients who are bed-bound, have poor prognosis, and barriers to access (eg, transportation issues), interventions for DEXA scan referrals are often not clinically indicated. During calculations of the FRAX score, documentation is often missing from a patient’s medical chart, making it difficult to answer all questions on the questionnaire. This does increase the utility of the OST score as the calculation is much easier and does not rely on other osteoporotic factors. Despite these restrictions for offering DEXA scans, the HBPC service has a high standard of excellence in preventing falls, a major contributor to fractures. Physical therapy services are readily available, nursing visits are frequent and as clinically indicated, vitamin D levels are maintained within normal limits via supplementation, and medication management is performed at least quarterly among other interventions.
Conclusions
The retrospective chart review of patients in the HBPC program suggests that there may be a lack of standardized screening for osteoporosis in the male patient population. As seen within the data, there is great potential for interventions as many of the patients would be candidates for screening based on the OST score. The tool is easy to use and readily accessible to all health care providers and staff. By increasing screening of eligible patients, it also increases the identification of those who would benefit from osteoporosis treatment. While the HBPC population has access limitations (eg, homebound, limited life expectancy), the implementation of a protocol and extension of concepts from this study can be extrapolated into other PACT clinics at VA facilities. Osteoporosis in the male population is often overlooked, but screening procedures can help reduce health care expenditures.
Osteoporosis is a disease characterized by the loss of bone density.1 Bone is normally porous and is in a state of flux due to changes in regeneration caused by osteoclast or osteoblast activity. However, age and other factors can accelerate loss in bone density and lead to decreased bone strength and an increased risk of fracture. In men, bone mineral density (BMD) can begin to decline as early as age 30 to 40 years. By age 80 years, 25% of total bone mass may be lost.2
Of the 44 million Americans with low BMD or osteoporosis, 20% are men.1 This group accounts for up to 40% of all osteoporotic fractures. About 1 in 4 men aged ≥ 50 years may experience a lifetime fracture. Fractures may lead to chronic pain, disability, increased dependence, and potentially death. These complications cause expenditures upward of $4.1 billion annually in North America alone.3,4 About 80,000 US men will experience a hip fracture each year, one-third of whom will die within that year. This constitutes a mortality rate 2 to 3 times higher than that of women. Osteoporosis often goes undiagnosed and untreated due to a lack of symptoms until a fracture occurs, underlining the potential benefit of preemptive screening.
In 2007, Shekell and colleagues outlined how the US Department of Veterans Affairs (VA) screened men for osteoporosis.5 At the time, 95% of the VA population was male, though it has since dropped to 91%.6 Shekell and colleagues estimated that about 200,0000 to 400,0000 male veterans had osteoporosis.5 Osteoporotic risk factors deemed specific to veterans were excessive alcohol use, spinal cord injury and lack of weight-bearing exercise, prolonged corticosteroid use, and androgen deprivation therapy in prostate cancer. Different screening techniques were assessed, and the VA recommended the Osteoporosis Self-Assessment Tool (OST).5 Many organizations have developed clinical guidance, including who should be screened; however, screening for men remains a controversial area due to a lack of any strong recommendations (Table 1).
Endocrine Society screening guidelines for men are the most specific: testing BMD in men aged ≥ 70 years, or if aged 50 to 69 years with an additional risk factor (eg, low body weight, smoking, chronic obstructive pulmonary disease, chronic steroid use).1 The Fracture Risk Assessment tool (FRAX) score is often cited as a common screening tool. It is a free online questionnaire that provides a 10-year probability risk of hip or major osteoporotic fracture.11 However, this tool is limited by age, weight, and the assumption that all questions are answered accurately. Some of the information required includes the presence of a number of risk factors, such as alcohol use, glucocorticoids, and medical history of rheumatoid arthritis, among others (Table 2). The OST score, on the other hand, is a calculation that does not take into account other risk factors (Figure 1). This tool categorizes the patient into low, moderate, or high risk for osteoporosis.8
In a study of 4,000 men aged ≥ 70 years,
A 2017 VA Office of Rural Health study examined the utility of OST to screen referred patients aged > 50 years to receive DEXA scans in patient aligned care team (PACT) clinics at 3 different VA locations.13 The study excluded patients who had been screened previously or treated for osteoporosis, were receiving hospice care; 1 site excluded patients aged > 88 years. Two of the sites also reviewed the patient’s medications to screen for agents that may contribute to increased fracture risk. Veterans identified as high risk were referred for education and offered a DEXA scan and treatment. In total, 867 veterans were screened; 19% (168) were deemed high risk, and 6% (53) underwent DEXA scans. The study noted that only 15 patients had reportable DEXA scans and 10 were positive for bone disease.
As there has been documented success in the PACT setting in implementing standardized protocols for screening and treating veterans, it is reasonable to extend the concept into other VA services. The home-based primary care (HBPC) population is especially vulnerable due to the age of patients, limited weight-bearing exercise to improve bone strength, and limited access to DEXA scans due to difficulty traveling outside of the home. Despite these issues, a goal of the HBPC service is to provide continual care for veterans and improve their health so they may return to the community setting. As a result, patients are followed frequently, providing many opportunities for interventions. This study aims to determine the proportion of HBPC patients who are at high risk for osteoporosis and can receive a DEXA scan for evaluation.
Methods
This study was a retrospective chart analysis using descriptive statistics. It was reviewed and approved by the institutional review board at Captain James A. Lovell Federal Health Care Center (FHCC). Patients were included in the study if they were enrolled in the HBPC program at FHCC. Patients were excluded if they were receiving hospice or palliative care, had a limited life expectancy per the HBPC provider, or had a diagnosis of osteoporosis that was being managed by a VA endocrinologist, rheumatologist, or non-VA provider.
The study was conducted from February 1, 2018, through November 30, 2018. All chart reviews were done through the FHCC electronic health record. A minimum of 80 and maximum of 150 charts were reviewed as this was the typical patient volume in the HBPC program. Basic demographic information was collected and analyzed by calculating FRAX and OST scores. With the results, patients were classified as low or high risk of developing osteoporosis, and whether a DEXA scan should be recommended.
Results
After chart review, 83 patients were enrolled in the FHCC HBPC program during the study period. Out of these, 5 patients were excluded due to hospice or palliative care status, limited life expectancy, or had their osteoporosis managed by another non-HBPC provider. As a result, 78 patients were analyzed to determine their risk of osteoporosis (Figure 2). Most of the patients were white males with a median age of 82 years. A majority of the patients did not have any current or previous treatment with bisphosphonates, 77% had normal vitamin D levels, and only 13% (10) were current smokers; of the male patients only 21% (15) had a previous DEXA scan (Table 3).
The FRAX and OST scores for each male patient were calculated (Table 4). Half the patients were low risk for osteoporosis. Just 20% (14) of the patients were at high risk for osteoporosis, and only 6 of those had DEXA scans. However, if expanding the criteria to OST scores of < 2, then only 24% (10) received DEXA scans. When calculating FRAX scores, 30% (21) had ≥ 9.3% for major osteoporotic fracture risk, and only 19% (4) had received a DEXA scan.
Discussion
Based on the collected data, many of the male HBPC patients have not had an evaluation for osteoporosis despite being in a high-risk population and meeting some of the screening guidelines by various organizations.1 Based on Diem and colleagues and the 2007 VA report, utilizing OST scores could help capture a subset of patients that would be referred for DEXA scans.5,12 Of the 60% (42) of patients that met OST scores of < 2, 76% (32) of them could have been referred for DEXA scans for osteoporosis evaluation. However, at the time of publication of this article, 50% (16) of the patients have been discharged from the service without interventions. Of the remaining 16 patients, only 2 were referred for a DEXA scan, and 1 patient had confirmed osteoporosis. Currently, these results have been reviewed by the HBPC provider, and plans are in place for DEXA scan referrals for the remaining patients. In addition, for new patients admitted to the program and during annual reviews, the plan is to use OST scores to help screen for osteoporosis.
Limitations
The HBPC population is often in flux due to discharges as patients pass away, become eligible for long-term care, advance to hospice or palliative care status, or see an improvement in their condition to transition back into the community. Along with patients who are bed-bound, have poor prognosis, and barriers to access (eg, transportation issues), interventions for DEXA scan referrals are often not clinically indicated. During calculations of the FRAX score, documentation is often missing from a patient’s medical chart, making it difficult to answer all questions on the questionnaire. This does increase the utility of the OST score as the calculation is much easier and does not rely on other osteoporotic factors. Despite these restrictions for offering DEXA scans, the HBPC service has a high standard of excellence in preventing falls, a major contributor to fractures. Physical therapy services are readily available, nursing visits are frequent and as clinically indicated, vitamin D levels are maintained within normal limits via supplementation, and medication management is performed at least quarterly among other interventions.
Conclusions
The retrospective chart review of patients in the HBPC program suggests that there may be a lack of standardized screening for osteoporosis in the male patient population. As seen within the data, there is great potential for interventions as many of the patients would be candidates for screening based on the OST score. The tool is easy to use and readily accessible to all health care providers and staff. By increasing screening of eligible patients, it also increases the identification of those who would benefit from osteoporosis treatment. While the HBPC population has access limitations (eg, homebound, limited life expectancy), the implementation of a protocol and extension of concepts from this study can be extrapolated into other PACT clinics at VA facilities. Osteoporosis in the male population is often overlooked, but screening procedures can help reduce health care expenditures.
1. Watts NB, Adler RA, Bilezikian JP, et al; Endocrine Society. Osteoporosis in men: an Endocrine Society clinical practice guideline. J Clin Endocrinol Metab. 2012;97(6):1802-1822.
2. Holt G, Smith R, Duncan K, Hutchison JD, Gregori A. Gender differences in epidemiology and outcome after hip fracture: evidence from the Scottish Hip Fracture Audit. J Bone Joint Surg Br. 2008;90(4):480-483.
3. Ackman JM, Lata PF, Schuna AA, Elliott ME. Bone health evaluation in a veteran population: a need for the Fracture Risk Assessment tool (FRAX). Ann Pharmacother. 2014;48(10):1288-1293.
4. International Osteoporosis Foundation. Osteoporosis in men: why change needs to happen. http://share.iofbone-health.org/WOD/2014/thematic-report/WOD14-Report.pdf. Published 2014. Accessed September 16, 2019.
5. Shekell P, Munjas B, Liu H, et al. Screening Men for Osteoporosis: Who & How. Evidence-based Synthesis Program. Washington, DC: Department of Veterans Affairs; 2007.
6. US Department of Veterans Affairs, National Center for Veterans Analysis and Statistics. Veteran population. https://www.va.gov/vetdata/Veteran_Population.asp. Accessed September 16, 2019.
7. Rao SS, Budhwar N, Ashfaque A. Osteoporosis in men. Am Fam Physician. 2010;82(5):503-508.
8. US Preventive Services Task Force, Curry SJ, Krist AH, et al. Screening for osteoporosis to prevent fractures: US Preventive Services Task Force recommendation statement. JAMA. 2018;319(24):2521-2531.
9. Viswanathan M, Reddy S, Berkman N, et al. Screening to prevent osteoporotic fractures updated evidence report and systematic review for the US Preventive Services Task Force. JAMA. 2018;319(24):2532-2551.
10. Cosman F, de Beur SJ, LeBoff MS, et al; National Osteoporosis Foundation. Clinician’s guide to prevention and treatment of osteoporosis. Osteoporos Int. 2014;25(10):2359-2381.
11. Centre for Metabolic Bone Diseases, University of Sheffield, UK. FRAX Fracture Risk Assessment Tool. http://www.sheffield.ac.uk/FRAX/tool.aspx?country=9. Accessed September 16, 2019.
12. Diem SJ, Peters KW, Gourlay ML, et al; Osteoporotic Fractures in Men Research Group. Screening for osteoporosis in older men: operating characteristics of proposed strategies for selecting men for BMD testing. J Gen Intern Med. 2017;32(11):1235-1241.
13. US Department of Veterans Affairs, Office of Rural Health. Osteoporosis risk assessment using Osteoporosis Self-Assessment Tool (OST) and other interventions at rural facilities. https://www.ruralhealth.va.gov/docs/promise/2017_02_01_OST_Issue%20Brief_v2.pdf. Published February 7, 2019. Accessed September 16, 2019.
1. Watts NB, Adler RA, Bilezikian JP, et al; Endocrine Society. Osteoporosis in men: an Endocrine Society clinical practice guideline. J Clin Endocrinol Metab. 2012;97(6):1802-1822.
2. Holt G, Smith R, Duncan K, Hutchison JD, Gregori A. Gender differences in epidemiology and outcome after hip fracture: evidence from the Scottish Hip Fracture Audit. J Bone Joint Surg Br. 2008;90(4):480-483.
3. Ackman JM, Lata PF, Schuna AA, Elliott ME. Bone health evaluation in a veteran population: a need for the Fracture Risk Assessment tool (FRAX). Ann Pharmacother. 2014;48(10):1288-1293.
4. International Osteoporosis Foundation. Osteoporosis in men: why change needs to happen. http://share.iofbone-health.org/WOD/2014/thematic-report/WOD14-Report.pdf. Published 2014. Accessed September 16, 2019.
5. Shekell P, Munjas B, Liu H, et al. Screening Men for Osteoporosis: Who & How. Evidence-based Synthesis Program. Washington, DC: Department of Veterans Affairs; 2007.
6. US Department of Veterans Affairs, National Center for Veterans Analysis and Statistics. Veteran population. https://www.va.gov/vetdata/Veteran_Population.asp. Accessed September 16, 2019.
7. Rao SS, Budhwar N, Ashfaque A. Osteoporosis in men. Am Fam Physician. 2010;82(5):503-508.
8. US Preventive Services Task Force, Curry SJ, Krist AH, et al. Screening for osteoporosis to prevent fractures: US Preventive Services Task Force recommendation statement. JAMA. 2018;319(24):2521-2531.
9. Viswanathan M, Reddy S, Berkman N, et al. Screening to prevent osteoporotic fractures updated evidence report and systematic review for the US Preventive Services Task Force. JAMA. 2018;319(24):2532-2551.
10. Cosman F, de Beur SJ, LeBoff MS, et al; National Osteoporosis Foundation. Clinician’s guide to prevention and treatment of osteoporosis. Osteoporos Int. 2014;25(10):2359-2381.
11. Centre for Metabolic Bone Diseases, University of Sheffield, UK. FRAX Fracture Risk Assessment Tool. http://www.sheffield.ac.uk/FRAX/tool.aspx?country=9. Accessed September 16, 2019.
12. Diem SJ, Peters KW, Gourlay ML, et al; Osteoporotic Fractures in Men Research Group. Screening for osteoporosis in older men: operating characteristics of proposed strategies for selecting men for BMD testing. J Gen Intern Med. 2017;32(11):1235-1241.
13. US Department of Veterans Affairs, Office of Rural Health. Osteoporosis risk assessment using Osteoporosis Self-Assessment Tool (OST) and other interventions at rural facilities. https://www.ruralhealth.va.gov/docs/promise/2017_02_01_OST_Issue%20Brief_v2.pdf. Published February 7, 2019. Accessed September 16, 2019.
Get ready for changes in polypharmacy quality ratings
NATIONAL HARBOR, MD. – and managed care organizations should start preparing now for the shift.
Panelists at an Oct. 30 session at the annual meeting of the Academy of Managed Care Pharmacy presented strategies for addressing the three areas of polypharmacy that will be tracked in the new rating system, which will replace the current high-risk medication measurement that is being retired this year.
Anticholinergic medications
The first area presented by the panelists was polypharmacy use of multiple anticholinergic medications in older adults (Poly-ACH). The new quality measure will examine the percentage of members aged 65 years or older who are using two or more anticholinergic medications concurrently.
“We know that anticholinergic burden increases the risk of cognitive decline in particular, but it’s also associated with a higher risk of falls, an increased number of hospitalizations, and [diminished] physical function,” said Marti Groeneweg, PharmD, supervisor of clinical pharmacy services at Kaiser Permanente.
Dr. Groeneweg noted that, in addition to using multiple drugs in this class, patients can also benefit from a decrease in the dosage of their drugs, so that should also be considered in managing the medication of beneficiaries.
She highlighted a program Kaiser Permanente started in the Northwest United States to reduce the concurrent use of these drugs. The program targeted tricyclic antidepressants – nortriptyline, in particular.
The company instituted a multipronged approach that included provider detailing of the risks of using multiple drugs and how they could taper schedules, as well as providing them with other supporting resources and a list of safer, alternative drugs. It also reached out to patients to educate them about the risks of their medications and why it was important for them to taper their medications. The third part of the approach was to use the EHR to provide doctors with the best-available information at the point of prescribing. And finally, there was a pharmacist review process put in place for more complex cases.
Dr. Groeneweg emphasized that this information was incorporated into existing programs.
The intervention, which is fairly new, has not been in place long enough to know exactly how well it is working, but early indicators suggest “we are on the right track,” she said, noting that to date there has been a decrease of 28% in the number of tricyclic antidepressant prescriptions per 1,000 Medicare members per month.
CNS medications
The second area the panelists addressed was the polypharmacy use of multiple CNS-active medications in older adults (Poly-CNS).
Rainelle Gaddy, PharmD, Rx clinical programs pharmacy lead at Humana Pharmacy Solutions, , noted that the clinical rationale for this measure was the “increased risk of falls and fractures when these medications are taken concurrently.”
She pointed out that taking one or more of the CNS medications can result in a 1.5-fold increase in the risk for falls, and that risk increases to 2.5-fold if two or more drugs are taken. In addition, a high-dose of these medications can lead to a threefold increase in risk of recurrent falls.
Dr. Gaddy highlighted a number of interventions that could be implemented when the managed care organization is not integrated in the way Kaiser Permanente is.
“Pharmacists can pay a pivotal role [in helping] patients who are receiving these Poly-CNS medications because they are able to interact and talk through the actual patient picture for all their medications ... because pharmacists have always been seen as being a trusted source,” she said.
Dr. Gaddy added that health plans can take a more direct role in reaching out to patients, for example, through telephone outreach, as well as direct mail, email, and newsletters.
“We want to make sure that members have as much information as possible,” she said.
She added that it is very important to include physicians and other prescribers in this process through faxes and information included in EHRs.
Opioids and benzodiazepines
The final measure highlighted during the session was the one measuring the concurrent use of opioids and benzodiazepines.
Dr. Gaddy noted that taking the two concurrently is associated with a fourfold increase in risk of opioid overdose and death, compared with opioid use without a benzodiazepine.
She noted that a black box warning on the risks of concurrent use was added to both opioids and benzodiazepines in August 2016 and that resulted in a 10% decrease in the concurrent use.
“This new measure is intended to ensure that the downward trend continues. CMS has indicated as such,” Dr. Gaddy said.
Most of the intervention strategies she highlighted were similar to those for the Poly-CNS category, including the use of medication therapy management programs and targeted interventions, telephone outreach to members, and provider detailing and outreach.
“Provider detailing is really key,” Dr. Gaddy said. “On any given day, it’s so easy for physicians to see 30 patients. The great thing about the provider detailing is that you are able to give the provider a ‘packet’ of their members, you can identify and/or aid in showing them the risk assessment associated with members taking these medications, and then equip them with pocket guides and [materials so they can] streamline the medications.”
NATIONAL HARBOR, MD. – and managed care organizations should start preparing now for the shift.
Panelists at an Oct. 30 session at the annual meeting of the Academy of Managed Care Pharmacy presented strategies for addressing the three areas of polypharmacy that will be tracked in the new rating system, which will replace the current high-risk medication measurement that is being retired this year.
Anticholinergic medications
The first area presented by the panelists was polypharmacy use of multiple anticholinergic medications in older adults (Poly-ACH). The new quality measure will examine the percentage of members aged 65 years or older who are using two or more anticholinergic medications concurrently.
“We know that anticholinergic burden increases the risk of cognitive decline in particular, but it’s also associated with a higher risk of falls, an increased number of hospitalizations, and [diminished] physical function,” said Marti Groeneweg, PharmD, supervisor of clinical pharmacy services at Kaiser Permanente.
Dr. Groeneweg noted that, in addition to using multiple drugs in this class, patients can also benefit from a decrease in the dosage of their drugs, so that should also be considered in managing the medication of beneficiaries.
She highlighted a program Kaiser Permanente started in the Northwest United States to reduce the concurrent use of these drugs. The program targeted tricyclic antidepressants – nortriptyline, in particular.
The company instituted a multipronged approach that included provider detailing of the risks of using multiple drugs and how they could taper schedules, as well as providing them with other supporting resources and a list of safer, alternative drugs. It also reached out to patients to educate them about the risks of their medications and why it was important for them to taper their medications. The third part of the approach was to use the EHR to provide doctors with the best-available information at the point of prescribing. And finally, there was a pharmacist review process put in place for more complex cases.
Dr. Groeneweg emphasized that this information was incorporated into existing programs.
The intervention, which is fairly new, has not been in place long enough to know exactly how well it is working, but early indicators suggest “we are on the right track,” she said, noting that to date there has been a decrease of 28% in the number of tricyclic antidepressant prescriptions per 1,000 Medicare members per month.
CNS medications
The second area the panelists addressed was the polypharmacy use of multiple CNS-active medications in older adults (Poly-CNS).
Rainelle Gaddy, PharmD, Rx clinical programs pharmacy lead at Humana Pharmacy Solutions, , noted that the clinical rationale for this measure was the “increased risk of falls and fractures when these medications are taken concurrently.”
She pointed out that taking one or more of the CNS medications can result in a 1.5-fold increase in the risk for falls, and that risk increases to 2.5-fold if two or more drugs are taken. In addition, a high-dose of these medications can lead to a threefold increase in risk of recurrent falls.
Dr. Gaddy highlighted a number of interventions that could be implemented when the managed care organization is not integrated in the way Kaiser Permanente is.
“Pharmacists can pay a pivotal role [in helping] patients who are receiving these Poly-CNS medications because they are able to interact and talk through the actual patient picture for all their medications ... because pharmacists have always been seen as being a trusted source,” she said.
Dr. Gaddy added that health plans can take a more direct role in reaching out to patients, for example, through telephone outreach, as well as direct mail, email, and newsletters.
“We want to make sure that members have as much information as possible,” she said.
She added that it is very important to include physicians and other prescribers in this process through faxes and information included in EHRs.
Opioids and benzodiazepines
The final measure highlighted during the session was the one measuring the concurrent use of opioids and benzodiazepines.
Dr. Gaddy noted that taking the two concurrently is associated with a fourfold increase in risk of opioid overdose and death, compared with opioid use without a benzodiazepine.
She noted that a black box warning on the risks of concurrent use was added to both opioids and benzodiazepines in August 2016 and that resulted in a 10% decrease in the concurrent use.
“This new measure is intended to ensure that the downward trend continues. CMS has indicated as such,” Dr. Gaddy said.
Most of the intervention strategies she highlighted were similar to those for the Poly-CNS category, including the use of medication therapy management programs and targeted interventions, telephone outreach to members, and provider detailing and outreach.
“Provider detailing is really key,” Dr. Gaddy said. “On any given day, it’s so easy for physicians to see 30 patients. The great thing about the provider detailing is that you are able to give the provider a ‘packet’ of their members, you can identify and/or aid in showing them the risk assessment associated with members taking these medications, and then equip them with pocket guides and [materials so they can] streamline the medications.”
NATIONAL HARBOR, MD. – and managed care organizations should start preparing now for the shift.
Panelists at an Oct. 30 session at the annual meeting of the Academy of Managed Care Pharmacy presented strategies for addressing the three areas of polypharmacy that will be tracked in the new rating system, which will replace the current high-risk medication measurement that is being retired this year.
Anticholinergic medications
The first area presented by the panelists was polypharmacy use of multiple anticholinergic medications in older adults (Poly-ACH). The new quality measure will examine the percentage of members aged 65 years or older who are using two or more anticholinergic medications concurrently.
“We know that anticholinergic burden increases the risk of cognitive decline in particular, but it’s also associated with a higher risk of falls, an increased number of hospitalizations, and [diminished] physical function,” said Marti Groeneweg, PharmD, supervisor of clinical pharmacy services at Kaiser Permanente.
Dr. Groeneweg noted that, in addition to using multiple drugs in this class, patients can also benefit from a decrease in the dosage of their drugs, so that should also be considered in managing the medication of beneficiaries.
She highlighted a program Kaiser Permanente started in the Northwest United States to reduce the concurrent use of these drugs. The program targeted tricyclic antidepressants – nortriptyline, in particular.
The company instituted a multipronged approach that included provider detailing of the risks of using multiple drugs and how they could taper schedules, as well as providing them with other supporting resources and a list of safer, alternative drugs. It also reached out to patients to educate them about the risks of their medications and why it was important for them to taper their medications. The third part of the approach was to use the EHR to provide doctors with the best-available information at the point of prescribing. And finally, there was a pharmacist review process put in place for more complex cases.
Dr. Groeneweg emphasized that this information was incorporated into existing programs.
The intervention, which is fairly new, has not been in place long enough to know exactly how well it is working, but early indicators suggest “we are on the right track,” she said, noting that to date there has been a decrease of 28% in the number of tricyclic antidepressant prescriptions per 1,000 Medicare members per month.
CNS medications
The second area the panelists addressed was the polypharmacy use of multiple CNS-active medications in older adults (Poly-CNS).
Rainelle Gaddy, PharmD, Rx clinical programs pharmacy lead at Humana Pharmacy Solutions, , noted that the clinical rationale for this measure was the “increased risk of falls and fractures when these medications are taken concurrently.”
She pointed out that taking one or more of the CNS medications can result in a 1.5-fold increase in the risk for falls, and that risk increases to 2.5-fold if two or more drugs are taken. In addition, a high-dose of these medications can lead to a threefold increase in risk of recurrent falls.
Dr. Gaddy highlighted a number of interventions that could be implemented when the managed care organization is not integrated in the way Kaiser Permanente is.
“Pharmacists can pay a pivotal role [in helping] patients who are receiving these Poly-CNS medications because they are able to interact and talk through the actual patient picture for all their medications ... because pharmacists have always been seen as being a trusted source,” she said.
Dr. Gaddy added that health plans can take a more direct role in reaching out to patients, for example, through telephone outreach, as well as direct mail, email, and newsletters.
“We want to make sure that members have as much information as possible,” she said.
She added that it is very important to include physicians and other prescribers in this process through faxes and information included in EHRs.
Opioids and benzodiazepines
The final measure highlighted during the session was the one measuring the concurrent use of opioids and benzodiazepines.
Dr. Gaddy noted that taking the two concurrently is associated with a fourfold increase in risk of opioid overdose and death, compared with opioid use without a benzodiazepine.
She noted that a black box warning on the risks of concurrent use was added to both opioids and benzodiazepines in August 2016 and that resulted in a 10% decrease in the concurrent use.
“This new measure is intended to ensure that the downward trend continues. CMS has indicated as such,” Dr. Gaddy said.
Most of the intervention strategies she highlighted were similar to those for the Poly-CNS category, including the use of medication therapy management programs and targeted interventions, telephone outreach to members, and provider detailing and outreach.
“Provider detailing is really key,” Dr. Gaddy said. “On any given day, it’s so easy for physicians to see 30 patients. The great thing about the provider detailing is that you are able to give the provider a ‘packet’ of their members, you can identify and/or aid in showing them the risk assessment associated with members taking these medications, and then equip them with pocket guides and [materials so they can] streamline the medications.”
REPORTING FROM AMCP NEXUS 2019
Geriatric Nutritional Risk Index predicts long-term outcomes in PAD
PARIS – The Geriatric Nutritional Risk Index proved to be an independent predictor of 5-year overall survival as well as the composite of major adverse cardiovascular and limb events in a prospective cohort study of 1,219 patients with peripheral artery disease, Yae Matsuo, MD, reported at the annual congress of the European Society of Cardiology.
The Geriatric Nutritional Risk Index (GNRI) is a score calculated with a formula based upon a patient’s height, serum albumin, and the ratio between ideal and actual body weight (Am J Clin Nutr. 2005 Oct;82(4):777-83). The GNRI tool has been shown to be an accurate prognosticator for clinical outcomes in patients on hemodialysis and those with heart failure. However, it’s predictive accuracy hasn’t been evaluated in patients with PAD, according to Dr. Matsuo, a cardiologist at Kitakanto Cardiovascular Hospital in Shibukawa, Japan.
“The Geriatric Nutritional Risk Index is simple to calculate – so easy – and I think it’s a better predictor than BMI,” she said.
Fifty-six percent of the PAD patients had a GNRI score greater than 98, indicative of no increased risk of malnutrition and nutritional deficiencies. Their 5-year overall survival rate was 81%, compared with 62% in patients with a score of 92-98, 40% in those with a score of 82-91, and 23% with a score of less than 82. Other independent predictors of overall survival in multivariate analysis were age, estimated glomerular filtration rate, ankle brachial index, and C-reactive protein level.
A GNRI score above 98 was also predictive of significantly lower 5-year risk of both major adverse cardiovascular events and the composite of major adverse cardiovascular and limb events than in patients with a score of 98 or less.
The key remaining unanswered question is whether providing timely nutritional support to PAD patients with a low GNRI score will result in improved overall and limb survival and other outcomes.
Dr. Matsuo reported having no financial conflicts.
SOURCE: Matsuo Y. ESC CONGRESS 2019. Abstract P1956.
PARIS – The Geriatric Nutritional Risk Index proved to be an independent predictor of 5-year overall survival as well as the composite of major adverse cardiovascular and limb events in a prospective cohort study of 1,219 patients with peripheral artery disease, Yae Matsuo, MD, reported at the annual congress of the European Society of Cardiology.
The Geriatric Nutritional Risk Index (GNRI) is a score calculated with a formula based upon a patient’s height, serum albumin, and the ratio between ideal and actual body weight (Am J Clin Nutr. 2005 Oct;82(4):777-83). The GNRI tool has been shown to be an accurate prognosticator for clinical outcomes in patients on hemodialysis and those with heart failure. However, it’s predictive accuracy hasn’t been evaluated in patients with PAD, according to Dr. Matsuo, a cardiologist at Kitakanto Cardiovascular Hospital in Shibukawa, Japan.
“The Geriatric Nutritional Risk Index is simple to calculate – so easy – and I think it’s a better predictor than BMI,” she said.
Fifty-six percent of the PAD patients had a GNRI score greater than 98, indicative of no increased risk of malnutrition and nutritional deficiencies. Their 5-year overall survival rate was 81%, compared with 62% in patients with a score of 92-98, 40% in those with a score of 82-91, and 23% with a score of less than 82. Other independent predictors of overall survival in multivariate analysis were age, estimated glomerular filtration rate, ankle brachial index, and C-reactive protein level.
A GNRI score above 98 was also predictive of significantly lower 5-year risk of both major adverse cardiovascular events and the composite of major adverse cardiovascular and limb events than in patients with a score of 98 or less.
The key remaining unanswered question is whether providing timely nutritional support to PAD patients with a low GNRI score will result in improved overall and limb survival and other outcomes.
Dr. Matsuo reported having no financial conflicts.
SOURCE: Matsuo Y. ESC CONGRESS 2019. Abstract P1956.
PARIS – The Geriatric Nutritional Risk Index proved to be an independent predictor of 5-year overall survival as well as the composite of major adverse cardiovascular and limb events in a prospective cohort study of 1,219 patients with peripheral artery disease, Yae Matsuo, MD, reported at the annual congress of the European Society of Cardiology.
The Geriatric Nutritional Risk Index (GNRI) is a score calculated with a formula based upon a patient’s height, serum albumin, and the ratio between ideal and actual body weight (Am J Clin Nutr. 2005 Oct;82(4):777-83). The GNRI tool has been shown to be an accurate prognosticator for clinical outcomes in patients on hemodialysis and those with heart failure. However, it’s predictive accuracy hasn’t been evaluated in patients with PAD, according to Dr. Matsuo, a cardiologist at Kitakanto Cardiovascular Hospital in Shibukawa, Japan.
“The Geriatric Nutritional Risk Index is simple to calculate – so easy – and I think it’s a better predictor than BMI,” she said.
Fifty-six percent of the PAD patients had a GNRI score greater than 98, indicative of no increased risk of malnutrition and nutritional deficiencies. Their 5-year overall survival rate was 81%, compared with 62% in patients with a score of 92-98, 40% in those with a score of 82-91, and 23% with a score of less than 82. Other independent predictors of overall survival in multivariate analysis were age, estimated glomerular filtration rate, ankle brachial index, and C-reactive protein level.
A GNRI score above 98 was also predictive of significantly lower 5-year risk of both major adverse cardiovascular events and the composite of major adverse cardiovascular and limb events than in patients with a score of 98 or less.
The key remaining unanswered question is whether providing timely nutritional support to PAD patients with a low GNRI score will result in improved overall and limb survival and other outcomes.
Dr. Matsuo reported having no financial conflicts.
SOURCE: Matsuo Y. ESC CONGRESS 2019. Abstract P1956.
REPORTING FROM THE ESC CONGRESS 2019
Severe hypoglycemia, poor glycemic control fuels fracture risk in older diabetic patients
Patients with type 2 diabetes and poor glycemic control or severe hypoglycemia may be at greater risk for fracture, according to recent research from a Japanese cohort of older men and postmenopausal women.
“The impacts of severe hypoglycemia and poor glycemic control on fractures appeared to be independent,” noted Yuji Komorita, MD, PhD, of the department of medicine and clinical science, Graduate School of Medical Sciences at Kyushu University, and colleagues. “This study suggests that the glycemic target to prevent fractures may be HbA1c <75 mmol/mol, which is far higher than that used to prevent microvascular complications, and higher than that for older adults with type 2 diabetes.”
Dr. Komorita and colleagues performed a prospective analysis of fracture incidence for 2,755 men and 1,951 postmenopausal women with type 2 diabetes in the Fukuoka Diabetes Registry who were mean 66 years old between April 2008 and October 2010. At the start of the study, the researchers assessed patient diabetes duration, previous fracture history, physical activity, alcohol and smoking status, whether patients were treated for diabetic retinopathy with laser photocoagulation, and their history of coronary artery disease or stroke. Patients were followed for a median 5.3 years, during which fractures were assessed through an annual self-administered questionnaire, with the results stratified by glycemic control and hypoglycemia.
Overall, there were 249 men and 413 women who experienced fractures during the study period, with a follow-up rate of 97.6%. In a multivariate analysis, patients with a higher risk of fracture included those with two or more episodes of severe hypoglycemia (hazard ratio, 2.25; 95% confidence interval, 1.57-3.22) and one episode of severe hypoglycemia (HR, 1.57; 95% CI, 1.11-2.20). In patients without severe hypoglycemic episodes, there was an increased risk of fracture in those with baseline hemoglobin A1c (HbA1c) level of 53 to less than 64 mmol/mol (7% to less than 8%; HR, 1.14; 0.94-1.39), 64 to less than 75 mmol/mol (8% to less than 9%; HR, 1.11; 95% CI, 0.86-1.43), and at least 75 mmol/mol (at least 9%; HR, 1.45; 95% CI, 1.06-1.98).
Compared with postmenopausal women, the unadjusted risk of fracture was higher in men with multiple severe hypoglycemic episodes (HR, 3.46; 95% CI, 2.05-5.85) and one episode of hypoglycemia (HR, 2.81; 95% CI, 1.74-4.56). These higher risks in older men persisted after adjustment for age, multivariate factors, and HbA1c.
“The association between severe hypoglycemia, poor glycemic control, and fracture risk at any anatomic site seems to be stronger in men than in postmenopausal women, although the interaction between men and postmenopausal women for fracture risk was not significant,” the researchers said. “The higher incidence rate of fractures in postmenopausal women, compared with men, was attributed to drastic changes in sex hormones after menopause, which may reduce the apparent impacts of hyperglycemia and severe hypoglycemia on postmenopausal women.”
Researchers said they did not consider potential external factors for fracture incidence, nor did they measure incident falls or other markers of bone health, such as bone mineral density and 25-hydroxyvitamin D levels. They also noted among the limitations of the study the self-reported nature of fracture reporting, and the lack of generalizability of the results.
This study was funded in part by grants from The Japan Society for the Promotion of Science KAKENHI from the Ministry of Education, Culture, Sports, Science and Technology of Japan; the Junior Scientist Development Grant supported by the Japan Diabetes Society; and the Lilly Research Grant Program for Bone & Mineral Research. The authors reported no relevant conflicts of interest.
SOURCE: Komorita Y et al. Diabet Med. 2019 Sep 25. doi: 10.1111/dme.14142.
Patients with type 2 diabetes and poor glycemic control or severe hypoglycemia may be at greater risk for fracture, according to recent research from a Japanese cohort of older men and postmenopausal women.
“The impacts of severe hypoglycemia and poor glycemic control on fractures appeared to be independent,” noted Yuji Komorita, MD, PhD, of the department of medicine and clinical science, Graduate School of Medical Sciences at Kyushu University, and colleagues. “This study suggests that the glycemic target to prevent fractures may be HbA1c <75 mmol/mol, which is far higher than that used to prevent microvascular complications, and higher than that for older adults with type 2 diabetes.”
Dr. Komorita and colleagues performed a prospective analysis of fracture incidence for 2,755 men and 1,951 postmenopausal women with type 2 diabetes in the Fukuoka Diabetes Registry who were mean 66 years old between April 2008 and October 2010. At the start of the study, the researchers assessed patient diabetes duration, previous fracture history, physical activity, alcohol and smoking status, whether patients were treated for diabetic retinopathy with laser photocoagulation, and their history of coronary artery disease or stroke. Patients were followed for a median 5.3 years, during which fractures were assessed through an annual self-administered questionnaire, with the results stratified by glycemic control and hypoglycemia.
Overall, there were 249 men and 413 women who experienced fractures during the study period, with a follow-up rate of 97.6%. In a multivariate analysis, patients with a higher risk of fracture included those with two or more episodes of severe hypoglycemia (hazard ratio, 2.25; 95% confidence interval, 1.57-3.22) and one episode of severe hypoglycemia (HR, 1.57; 95% CI, 1.11-2.20). In patients without severe hypoglycemic episodes, there was an increased risk of fracture in those with baseline hemoglobin A1c (HbA1c) level of 53 to less than 64 mmol/mol (7% to less than 8%; HR, 1.14; 0.94-1.39), 64 to less than 75 mmol/mol (8% to less than 9%; HR, 1.11; 95% CI, 0.86-1.43), and at least 75 mmol/mol (at least 9%; HR, 1.45; 95% CI, 1.06-1.98).
Compared with postmenopausal women, the unadjusted risk of fracture was higher in men with multiple severe hypoglycemic episodes (HR, 3.46; 95% CI, 2.05-5.85) and one episode of hypoglycemia (HR, 2.81; 95% CI, 1.74-4.56). These higher risks in older men persisted after adjustment for age, multivariate factors, and HbA1c.
“The association between severe hypoglycemia, poor glycemic control, and fracture risk at any anatomic site seems to be stronger in men than in postmenopausal women, although the interaction between men and postmenopausal women for fracture risk was not significant,” the researchers said. “The higher incidence rate of fractures in postmenopausal women, compared with men, was attributed to drastic changes in sex hormones after menopause, which may reduce the apparent impacts of hyperglycemia and severe hypoglycemia on postmenopausal women.”
Researchers said they did not consider potential external factors for fracture incidence, nor did they measure incident falls or other markers of bone health, such as bone mineral density and 25-hydroxyvitamin D levels. They also noted among the limitations of the study the self-reported nature of fracture reporting, and the lack of generalizability of the results.
This study was funded in part by grants from The Japan Society for the Promotion of Science KAKENHI from the Ministry of Education, Culture, Sports, Science and Technology of Japan; the Junior Scientist Development Grant supported by the Japan Diabetes Society; and the Lilly Research Grant Program for Bone & Mineral Research. The authors reported no relevant conflicts of interest.
SOURCE: Komorita Y et al. Diabet Med. 2019 Sep 25. doi: 10.1111/dme.14142.
Patients with type 2 diabetes and poor glycemic control or severe hypoglycemia may be at greater risk for fracture, according to recent research from a Japanese cohort of older men and postmenopausal women.
“The impacts of severe hypoglycemia and poor glycemic control on fractures appeared to be independent,” noted Yuji Komorita, MD, PhD, of the department of medicine and clinical science, Graduate School of Medical Sciences at Kyushu University, and colleagues. “This study suggests that the glycemic target to prevent fractures may be HbA1c <75 mmol/mol, which is far higher than that used to prevent microvascular complications, and higher than that for older adults with type 2 diabetes.”
Dr. Komorita and colleagues performed a prospective analysis of fracture incidence for 2,755 men and 1,951 postmenopausal women with type 2 diabetes in the Fukuoka Diabetes Registry who were mean 66 years old between April 2008 and October 2010. At the start of the study, the researchers assessed patient diabetes duration, previous fracture history, physical activity, alcohol and smoking status, whether patients were treated for diabetic retinopathy with laser photocoagulation, and their history of coronary artery disease or stroke. Patients were followed for a median 5.3 years, during which fractures were assessed through an annual self-administered questionnaire, with the results stratified by glycemic control and hypoglycemia.
Overall, there were 249 men and 413 women who experienced fractures during the study period, with a follow-up rate of 97.6%. In a multivariate analysis, patients with a higher risk of fracture included those with two or more episodes of severe hypoglycemia (hazard ratio, 2.25; 95% confidence interval, 1.57-3.22) and one episode of severe hypoglycemia (HR, 1.57; 95% CI, 1.11-2.20). In patients without severe hypoglycemic episodes, there was an increased risk of fracture in those with baseline hemoglobin A1c (HbA1c) level of 53 to less than 64 mmol/mol (7% to less than 8%; HR, 1.14; 0.94-1.39), 64 to less than 75 mmol/mol (8% to less than 9%; HR, 1.11; 95% CI, 0.86-1.43), and at least 75 mmol/mol (at least 9%; HR, 1.45; 95% CI, 1.06-1.98).
Compared with postmenopausal women, the unadjusted risk of fracture was higher in men with multiple severe hypoglycemic episodes (HR, 3.46; 95% CI, 2.05-5.85) and one episode of hypoglycemia (HR, 2.81; 95% CI, 1.74-4.56). These higher risks in older men persisted after adjustment for age, multivariate factors, and HbA1c.
“The association between severe hypoglycemia, poor glycemic control, and fracture risk at any anatomic site seems to be stronger in men than in postmenopausal women, although the interaction between men and postmenopausal women for fracture risk was not significant,” the researchers said. “The higher incidence rate of fractures in postmenopausal women, compared with men, was attributed to drastic changes in sex hormones after menopause, which may reduce the apparent impacts of hyperglycemia and severe hypoglycemia on postmenopausal women.”
Researchers said they did not consider potential external factors for fracture incidence, nor did they measure incident falls or other markers of bone health, such as bone mineral density and 25-hydroxyvitamin D levels. They also noted among the limitations of the study the self-reported nature of fracture reporting, and the lack of generalizability of the results.
This study was funded in part by grants from The Japan Society for the Promotion of Science KAKENHI from the Ministry of Education, Culture, Sports, Science and Technology of Japan; the Junior Scientist Development Grant supported by the Japan Diabetes Society; and the Lilly Research Grant Program for Bone & Mineral Research. The authors reported no relevant conflicts of interest.
SOURCE: Komorita Y et al. Diabet Med. 2019 Sep 25. doi: 10.1111/dme.14142.
FROM DIABETIC MEDICINE
Dr. Paul Aisen Q&A: Aducanumab for Alzheimer’s
In the wake of Biogen and Eisai’s Oct. 22 announcement about plans to apply to the Food and Drug Administration next year for the regulatory approval of the investigational monoclonal antibody aducanumab as a treatment for Alzheimer’s disease, we spoke with Paul Aisen, MD, the founding director of the Alzheimer’s Therapy Research Institute at the University of Southern California, Los Angeles, for his views on the news. He has been a consultant for Biogen and is a member of the aducanumab steering committee.
Q: What was your first reaction when you heard about the plan to submit an application for aducanumab to the FDA?
A: My initial reaction is that this provides terrific support for the amyloid hypothesis, and is consistent with the early aducanumab studies showing significant reductions in brain amyloid with resulting clinical improvement.
My next thought was that these data are going to be very, very challenging to analyze because both of these trials were stopped early, and one was clearly negative. We really need to scrutinize the data, but even at this point I would say this strongly supports targeting amyloid. The scrutiny will begin in detail at the Clinical Trials in Alzheimer’s Disease conference in December, when Biogen will likely release detailed data. A lot of people will analyze it, and I think that’s great. It’s beneficial to bring different perspectives.
We have had a terribly frustrating series of disappointments in the field. After the futility analysis of aducanumab and the multiple failures of BACE [beta-secretase] inhibitors, many were convinced we were barking up the wrong tree. I think these results, although complicated, should resurrect the enthusiasm for targeting amyloid.
Q: What is different about aducanumab from other antibodies tested – and rejected – in Alzheimer’s drug development?
A: There are lots of antibodies that have been tested in clinical trials. They all differ in terms of their affinity for amyloid beta. Some target monomers of the protein. Some target dimers. Some target fibrils. Some tie up amyloid and some reduce it. Aducanumab directly attacks brain plaques, reducing the plaque load in the brain. It carries a liability of amyloid-related imaging abnormalities [ARIA], but it also allows us to assess the impact that removing plaques might have on downstream events, including biomarkers. Overall, these data show that aducanumab did remove brain plaques and that removing them had a beneficial effect on cognition and function, and also a favorable effect on downstream biomarkers.
But again, we must be cautious because this is a complex data set taken from a post hoc analysis of two different terminated trials.
Q: We see some statistically significant differences in cognitive and functional outcomes. What would that mean for patients on an everyday basis?
A: Well, everyone is different, so that’s hard to say. A 25% slowing of functional decline on the Clinical Dementia Rating Scale sum of boxes (CDR-SB) might mean that, at the end of a year, there’s not a significant change in memory, or that there’s better social function. If both trials had been completed and if people had 18 months of high-dose aducanumab, the slowing of functional decline on the CDR-SB might in fact be greater than reported. Again, we’re having to draw conclusions from interrupted trials.
Q: This suggestion you make of a potentially continuous slowing of decline – are you suggesting that aducanumab might slow decline to the point of stopping it altogether? If an elderly patient has little or no progression until death would that, in effect, be considered a “cure?”
A: I don’t think it is possible to cure AD once the disease is clinically evident. These are studies of people with early AD, late mild cognitive impairment, and mild dementia. At that stage, there’s already a loss of synapses that won’t come back, and these studies don’t suggest that aducanumab can cure that. But what if people took it earlier, when the brain is still functioning normally? Some of us have argued for many years that earlier intervention is the way to go. And since we can now identify people [with brain plaques] before they become symptomatic, there is the possibility that if we removed them, we could stop progression.
Q: Are there any plans to study aducanumab as a preventive agent?
A: A grant has been awarded for this, but it was put on hold after the futility analysis. I don’t know when or if that will go forward.
(Editor’s note: The National Institutes of Health previously awarded Banner Health a $32 million, 5-year grant to examine this. The 2-year prevention study of aducanumab is aimed at cognitively unimpaired 65- to 80-year-old patients with PET-confirmed amyloid brain plaques. It was to be a multicenter, double-blind, placebo-controlled trial using Alzheimer’s biomarker endpoints as primary outcomes, along with cognitive and clinical changes, safety, and tolerability. The study was put on hold after Biogen discontinued the aducanumab development program in March. Investigators are considering whether to resurrect plans considering the new data. The study is intended to be a public-private partnership, with additional unspecified funding from Biogen plus $10 million from philanthropic sources. It has three intended goals: To find an approved prevention therapy as early as 2023, ahead of the National Plan to Address Alzheimer’s Disease’s goal of an effective prevention strategy by 2025; to advance the use of surrogate biomarkers to rapidly test and support accelerated approval of prevention therapies in almost everyone at biomarker or genetic risk, even in earlier preclinical Alzheimer’s stages when some treatments may have their greatest benefit; and to help make it possible to conduct prevention trials in at-risk persons even before they have extensive amyloid plaques, when some treatments may have their greatest benefit.)
Q: It seems like rolling this out to an enormous population of patients is going to be difficult, if not impossible. Are people really going to be able to commit to what could be a lifetime of monthly intravenous infusions of a medicine that could be expensive, as therapeutic antibodies generally are?
A: I would say, nothing about this disease is easy. It’s devastating and horrible. And if someone is diagnosed at this stage, I would think that individual would embrace any opportunity to treat it. My hope is that we will be able to prescreen people with an effective blood test for amyloid that would be part of a regular testing protocol once they reach a certain age. Those with positive results would be referred for more testing, including amyloid brain imaging.
In the wake of Biogen and Eisai’s Oct. 22 announcement about plans to apply to the Food and Drug Administration next year for the regulatory approval of the investigational monoclonal antibody aducanumab as a treatment for Alzheimer’s disease, we spoke with Paul Aisen, MD, the founding director of the Alzheimer’s Therapy Research Institute at the University of Southern California, Los Angeles, for his views on the news. He has been a consultant for Biogen and is a member of the aducanumab steering committee.
Q: What was your first reaction when you heard about the plan to submit an application for aducanumab to the FDA?
A: My initial reaction is that this provides terrific support for the amyloid hypothesis, and is consistent with the early aducanumab studies showing significant reductions in brain amyloid with resulting clinical improvement.
My next thought was that these data are going to be very, very challenging to analyze because both of these trials were stopped early, and one was clearly negative. We really need to scrutinize the data, but even at this point I would say this strongly supports targeting amyloid. The scrutiny will begin in detail at the Clinical Trials in Alzheimer’s Disease conference in December, when Biogen will likely release detailed data. A lot of people will analyze it, and I think that’s great. It’s beneficial to bring different perspectives.
We have had a terribly frustrating series of disappointments in the field. After the futility analysis of aducanumab and the multiple failures of BACE [beta-secretase] inhibitors, many were convinced we were barking up the wrong tree. I think these results, although complicated, should resurrect the enthusiasm for targeting amyloid.
Q: What is different about aducanumab from other antibodies tested – and rejected – in Alzheimer’s drug development?
A: There are lots of antibodies that have been tested in clinical trials. They all differ in terms of their affinity for amyloid beta. Some target monomers of the protein. Some target dimers. Some target fibrils. Some tie up amyloid and some reduce it. Aducanumab directly attacks brain plaques, reducing the plaque load in the brain. It carries a liability of amyloid-related imaging abnormalities [ARIA], but it also allows us to assess the impact that removing plaques might have on downstream events, including biomarkers. Overall, these data show that aducanumab did remove brain plaques and that removing them had a beneficial effect on cognition and function, and also a favorable effect on downstream biomarkers.
But again, we must be cautious because this is a complex data set taken from a post hoc analysis of two different terminated trials.
Q: We see some statistically significant differences in cognitive and functional outcomes. What would that mean for patients on an everyday basis?
A: Well, everyone is different, so that’s hard to say. A 25% slowing of functional decline on the Clinical Dementia Rating Scale sum of boxes (CDR-SB) might mean that, at the end of a year, there’s not a significant change in memory, or that there’s better social function. If both trials had been completed and if people had 18 months of high-dose aducanumab, the slowing of functional decline on the CDR-SB might in fact be greater than reported. Again, we’re having to draw conclusions from interrupted trials.
Q: This suggestion you make of a potentially continuous slowing of decline – are you suggesting that aducanumab might slow decline to the point of stopping it altogether? If an elderly patient has little or no progression until death would that, in effect, be considered a “cure?”
A: I don’t think it is possible to cure AD once the disease is clinically evident. These are studies of people with early AD, late mild cognitive impairment, and mild dementia. At that stage, there’s already a loss of synapses that won’t come back, and these studies don’t suggest that aducanumab can cure that. But what if people took it earlier, when the brain is still functioning normally? Some of us have argued for many years that earlier intervention is the way to go. And since we can now identify people [with brain plaques] before they become symptomatic, there is the possibility that if we removed them, we could stop progression.
Q: Are there any plans to study aducanumab as a preventive agent?
A: A grant has been awarded for this, but it was put on hold after the futility analysis. I don’t know when or if that will go forward.
(Editor’s note: The National Institutes of Health previously awarded Banner Health a $32 million, 5-year grant to examine this. The 2-year prevention study of aducanumab is aimed at cognitively unimpaired 65- to 80-year-old patients with PET-confirmed amyloid brain plaques. It was to be a multicenter, double-blind, placebo-controlled trial using Alzheimer’s biomarker endpoints as primary outcomes, along with cognitive and clinical changes, safety, and tolerability. The study was put on hold after Biogen discontinued the aducanumab development program in March. Investigators are considering whether to resurrect plans considering the new data. The study is intended to be a public-private partnership, with additional unspecified funding from Biogen plus $10 million from philanthropic sources. It has three intended goals: To find an approved prevention therapy as early as 2023, ahead of the National Plan to Address Alzheimer’s Disease’s goal of an effective prevention strategy by 2025; to advance the use of surrogate biomarkers to rapidly test and support accelerated approval of prevention therapies in almost everyone at biomarker or genetic risk, even in earlier preclinical Alzheimer’s stages when some treatments may have their greatest benefit; and to help make it possible to conduct prevention trials in at-risk persons even before they have extensive amyloid plaques, when some treatments may have their greatest benefit.)
Q: It seems like rolling this out to an enormous population of patients is going to be difficult, if not impossible. Are people really going to be able to commit to what could be a lifetime of monthly intravenous infusions of a medicine that could be expensive, as therapeutic antibodies generally are?
A: I would say, nothing about this disease is easy. It’s devastating and horrible. And if someone is diagnosed at this stage, I would think that individual would embrace any opportunity to treat it. My hope is that we will be able to prescreen people with an effective blood test for amyloid that would be part of a regular testing protocol once they reach a certain age. Those with positive results would be referred for more testing, including amyloid brain imaging.
In the wake of Biogen and Eisai’s Oct. 22 announcement about plans to apply to the Food and Drug Administration next year for the regulatory approval of the investigational monoclonal antibody aducanumab as a treatment for Alzheimer’s disease, we spoke with Paul Aisen, MD, the founding director of the Alzheimer’s Therapy Research Institute at the University of Southern California, Los Angeles, for his views on the news. He has been a consultant for Biogen and is a member of the aducanumab steering committee.
Q: What was your first reaction when you heard about the plan to submit an application for aducanumab to the FDA?
A: My initial reaction is that this provides terrific support for the amyloid hypothesis, and is consistent with the early aducanumab studies showing significant reductions in brain amyloid with resulting clinical improvement.
My next thought was that these data are going to be very, very challenging to analyze because both of these trials were stopped early, and one was clearly negative. We really need to scrutinize the data, but even at this point I would say this strongly supports targeting amyloid. The scrutiny will begin in detail at the Clinical Trials in Alzheimer’s Disease conference in December, when Biogen will likely release detailed data. A lot of people will analyze it, and I think that’s great. It’s beneficial to bring different perspectives.
We have had a terribly frustrating series of disappointments in the field. After the futility analysis of aducanumab and the multiple failures of BACE [beta-secretase] inhibitors, many were convinced we were barking up the wrong tree. I think these results, although complicated, should resurrect the enthusiasm for targeting amyloid.
Q: What is different about aducanumab from other antibodies tested – and rejected – in Alzheimer’s drug development?
A: There are lots of antibodies that have been tested in clinical trials. They all differ in terms of their affinity for amyloid beta. Some target monomers of the protein. Some target dimers. Some target fibrils. Some tie up amyloid and some reduce it. Aducanumab directly attacks brain plaques, reducing the plaque load in the brain. It carries a liability of amyloid-related imaging abnormalities [ARIA], but it also allows us to assess the impact that removing plaques might have on downstream events, including biomarkers. Overall, these data show that aducanumab did remove brain plaques and that removing them had a beneficial effect on cognition and function, and also a favorable effect on downstream biomarkers.
But again, we must be cautious because this is a complex data set taken from a post hoc analysis of two different terminated trials.
Q: We see some statistically significant differences in cognitive and functional outcomes. What would that mean for patients on an everyday basis?
A: Well, everyone is different, so that’s hard to say. A 25% slowing of functional decline on the Clinical Dementia Rating Scale sum of boxes (CDR-SB) might mean that, at the end of a year, there’s not a significant change in memory, or that there’s better social function. If both trials had been completed and if people had 18 months of high-dose aducanumab, the slowing of functional decline on the CDR-SB might in fact be greater than reported. Again, we’re having to draw conclusions from interrupted trials.
Q: This suggestion you make of a potentially continuous slowing of decline – are you suggesting that aducanumab might slow decline to the point of stopping it altogether? If an elderly patient has little or no progression until death would that, in effect, be considered a “cure?”
A: I don’t think it is possible to cure AD once the disease is clinically evident. These are studies of people with early AD, late mild cognitive impairment, and mild dementia. At that stage, there’s already a loss of synapses that won’t come back, and these studies don’t suggest that aducanumab can cure that. But what if people took it earlier, when the brain is still functioning normally? Some of us have argued for many years that earlier intervention is the way to go. And since we can now identify people [with brain plaques] before they become symptomatic, there is the possibility that if we removed them, we could stop progression.
Q: Are there any plans to study aducanumab as a preventive agent?
A: A grant has been awarded for this, but it was put on hold after the futility analysis. I don’t know when or if that will go forward.
(Editor’s note: The National Institutes of Health previously awarded Banner Health a $32 million, 5-year grant to examine this. The 2-year prevention study of aducanumab is aimed at cognitively unimpaired 65- to 80-year-old patients with PET-confirmed amyloid brain plaques. It was to be a multicenter, double-blind, placebo-controlled trial using Alzheimer’s biomarker endpoints as primary outcomes, along with cognitive and clinical changes, safety, and tolerability. The study was put on hold after Biogen discontinued the aducanumab development program in March. Investigators are considering whether to resurrect plans considering the new data. The study is intended to be a public-private partnership, with additional unspecified funding from Biogen plus $10 million from philanthropic sources. It has three intended goals: To find an approved prevention therapy as early as 2023, ahead of the National Plan to Address Alzheimer’s Disease’s goal of an effective prevention strategy by 2025; to advance the use of surrogate biomarkers to rapidly test and support accelerated approval of prevention therapies in almost everyone at biomarker or genetic risk, even in earlier preclinical Alzheimer’s stages when some treatments may have their greatest benefit; and to help make it possible to conduct prevention trials in at-risk persons even before they have extensive amyloid plaques, when some treatments may have their greatest benefit.)
Q: It seems like rolling this out to an enormous population of patients is going to be difficult, if not impossible. Are people really going to be able to commit to what could be a lifetime of monthly intravenous infusions of a medicine that could be expensive, as therapeutic antibodies generally are?
A: I would say, nothing about this disease is easy. It’s devastating and horrible. And if someone is diagnosed at this stage, I would think that individual would embrace any opportunity to treat it. My hope is that we will be able to prescreen people with an effective blood test for amyloid that would be part of a regular testing protocol once they reach a certain age. Those with positive results would be referred for more testing, including amyloid brain imaging.