User login
Food Insecurity Among Veterans: Resources to Screen and Intervene
Nearly 1 in 8 households—and 1 in 6 households with children—experienced food insecurity in 2017, defined as limited or uncertain availability of nutritionally adequate and safe foods.1 Food insecurity is often even more pronounced among households with individuals with acute or chronic medical conditions.2-6 Moreover, food insecurity is independently associated with a range of adverse health outcomes, including poorer control of diabetes mellitus, hypertension, depression and other major psychiatric disorders, HIV, and chronic lung and kidney disease, as well as poorer overall health status.7-14 Food insecurity also has been associated with increased health care costs and acute care utilization as well as increased probability of delayed or missed care.15-19
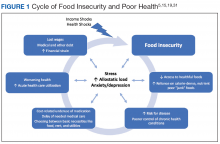
The relationship between food insecurity and poor health outcomes is a complex and often cyclic phenomenon (Figure 1). Poor nutritional status is fueled by limited access to healthful foods as well as increased reliance on calorie-dense and nutrient-poor “junk” foods, which are less expensive and often more readily available in low-income neighborhoods.5,20-24 These compensatory dietary patterns place individuals at higher risk for developing cardiometabolic conditions and for poor control of these conditions.5,8,9,12,25,26 Additionally, the physiological and psychological stressors of food insecurity may precipitate depression and anxiety or worsen existing mental health conditions, resulting in feelings of overwhelm and decreased self-management capacity.5,8,27-31 Food insecurity has further been associated with poor sleep, declines in cognitive function, and increased falls, particularly among the frail and elderly.32-34
Individuals experiencing food insecurity often report having to make trade-offs between food and other necessities, such as paying rent or utilities. Additional strategies to stretch limited resources include cost-related underuse of medication and delays in needed medical care.4,17,31,35 In a nationally representative survey among adults with at least 1 chronic medical condition, 1 in 3 reported having to choose between food and medicine; 11% were unable to afford either.3 Furthermore, the inability to reliably adhere to medication regimens that need to be taken with food can result in potentially life-threatening hypoglycemia (as can lack of food regardless of medication use).5,26,36 In addition to the more obvious risks of glucose-lowering medications, such as insulin and long-acting sulfonylureas in patients experiencing food insecurity, many drugs commonly used among nondiabetic adults such as ACE-inhibitors, β blockers, quinolones, and salicylates can also precipitate hypoglycemia, and food insecurity has been associated with experiences of hypoglycemia even among individuals without diabetes mellitus.32,37 In one study the risk for hospital admissions for hypoglycemia among low-income populations increased by 27% at the end of the month when food budgets were more likely to be exhausted.38 Worsening health status and increased emergency department visits and hospitalizations may then result in lost wages and mounting medical bills, contributing to further financial strain and worsening food insecurity.
Prevalence and Importance of Food Insecurity Among US Veterans
Nearly 1.5 million veterans in the US are living below the federal poverty level (FPL).39 An additional 2.4 million veterans are living paycheck to paycheck at < 200% of the FPL.40 Veterans living in poverty are at even higher risk than nonveterans for food insecurity, homelessness, and other material hardship.41
Estimates of food insecurity among veterans vary widely, ranging from 6% to 24%—nearly twice that of the general US population.8,42-45 Higher rates of food insecurity have been reported among certain high-risk subgroups, including veterans who served in Iraq and Afghanistan (27%), female veterans (28%), homeless and formerly homeless veterans (49%), and veterans with serious mental illness (35%).6,32,43,46 Additional risk factors for food insecurity specific to veteran populations include younger age, having recently left active-duty military service, and lower final military paygrade.42,45-47 As in the general population, veteran food insecurity is associated with a range of adverse health outcomes, including poorer overall health status as well as increased probability of delayed or missed care.6,8,32,42-44,46
Even among veterans enrolled in federal food assistance programs, many still struggle to afford nutritionally adequate foods. As one example, in a study of mostly male homeless and formerly homeless veterans, O’Toole and colleagues found that nearly half of those reporting food insecurity were already receiving federal food assistance benefits, and 22% relied on emergency food resources.32 Of households served by Feeding America food pantries and meal programs, 20% have a member who has served in the US military.48
Federal Programs To Address Food Insecurity
There are several important federal food assistance programs designed to help alleviate food insecurity. The Supplemental Nutrition Assistance Program (SNAP, formerly the Food Stamp program) is the largest federal food assistance program and provides low-income Americans with cash benefits to purchase food. SNAP has been shown to substantially reduce food insecurity.7,49 The program also is associated with significant decreases in cost-related medication nonadherence as well as reductions in health care costs and both acute care and nursing home utilization.16,50-54 Although nearly 1.4 million veterans live in SNAP-enrolled households, 59% of eligible veterans are not enrolled.43,55 Closing this SNAP eligibility-enrollment gap, has been a focus of recent efforts to improve long-term food security among veterans. There also are several federal food assistance programs for households with children, including the Special Supplemental Nutrition Program for Women, Infants, and Children (WIC) and school meals programs. Among federal nutrition programs for seniors, the Older American’s Act contains designated funding to support nutrition services for older adults, including congregate meal programs in community settings like senior centers, places of worship, and housing communities, and home-delivered meals through programs like Meals on Wheels.56
VHA Response to Food Insecurity
The Veterans Health Administration (VHA) is the country’s largest integrated, federally funded health care system.57 In November 2015, congressional briefings on veteran food insecurity organized by the national non-profit organization MAZON: A Jewish Response to Hunger and hosted with bipartisan support were provided to the US House and Senate. As a result of these briefings, VHA chartered the national Ensuring Veteran Food Security Workgroup with a mandate to partner with governmental and nonprofit agencies to “focus on the issue of food insecurity, the identification of veterans at risk, the needed training of VHA staff and the coordination of resources and initiatives to support the veterans for whom we care.” Building off a pilot in US Department of Veterans Affairs (VA) Homeless Patient Aligned Care Teams (H-PACTs),32 VHA subsequently integrated a single-item food insecurity screening tool into the VA electronic health record (EHR) clinical reminder system (Figure 2). The clinical reminder, which was rolled out across VA medical centers nationally in October 2017, provides an alert to screen all noninstitutionalized veterans for food insecurity. To date, nearly 5 million veterans have been screened. When a veteran endorses food insecurity based on the initial screening question, a prompt appears to offer the veteran a referral to a social worker and/or dietitian. Positive screening results also should be communicated to the patient’s primary care provider. Depending on site-specific clinical flow, the reminders are typically completed in the outpatient setting either by nurses or medical assistants during intake or by providers as part of the clinical visit. However, any member of the health care team can complete the clinical reminder at any time. As of September 2019, approximately 74,000 veterans have been identified as food insecure.58
Addressing Food Insecurity
VHA has been a recognized leader in addressing homelessness and other social determinants of health through its integrated care and PACT delivery models.59-61 The food insecurity clinical reminder was designed to facilitate a tailored, interdisciplinary approach to identify and address food insecurity. Interdisciplinary care team members—including medical assistants, clinicians, social workers, registered dietitians, nurse care managers, occupational or physical therapists, and pharmacists—are uniquely positioned to identify veterans impacted by food insecurity, assess for associated clinical and/or social risk factors, and offer appropriate medical and nutrition interventions and resource referrals.
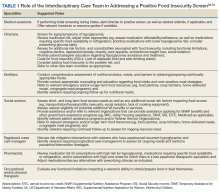
This interdisciplinary team-based model is essential given the range of potential drivers underlying veteran experiences of food insecurity and subsequent health outcomes. It is critically important for clinicians to review the medication list with veterans screening positive for food insecurity to assess for risk of hypoglycemia and/or cost-related nonadherence, make any necessary adjustments to therapeutic regimens, and assess for additional risk factors associated with food insecurity. Examples of tailored nutrition counseling that clinical dietitians may provide include meal preparation strategies for veterans who only have access to a microwave or hotplate, or recommendations for how veterans on medically restricted diets can best navigate food selection at soup kitchens or food pantries. Resource referrals provided by social workers or other care team members may include both emergency food resources to address immediate shortages (eg, food pantries, soup kitchens, or vouchers for free lunch) as well as resources focused on improving longer term food security (eg, federal food assistance programs or home delivered meal programs). Importantly, although providing a list of food resources may be helpful for some patients, such lists are often insufficient.62,63 Many patients require active assistance with program enrollment either onsite the day of their clinic visit or through connection with a partnering community-based organization that can, in turn, identify appropriate resources and facilitate program enrollment.63,64 Planned follow-up is also crucial to determine whether referrals are successful and to assess for ongoing need. Proposed roles for interdisciplinary care team members in addressing a positive food insecurity screen are outlined in Table 1.
VHA-Community Partnerships
In addition to services offered within VA, public and private sector partnerships can greatly enhance the range of resources available to food insecure veterans. Several VA facilities have developed formal community partnerships, such as the Veterans Pantry Pilot (VPP) program, a national partnership between Feeding America food banks and VA medical centers to establish onsite or mobile food pantries. There are currently 17 active Feeding America VPP sites, with a number of additional sites under development. Several of the VPP sites also include other “wraparound services,” such as SNAP application assistance.65,66
State Veterans Affairs offices67 and Veterans Service Organizations (VSOs)68 also can serve as valuable partners for connecting veterans with needed resources. VSOs offer a range of services, including assistancewith benefit claims, employment and housing assistance, emergency food assistance, and transportation to medical appointments. Some VSOs also have established local affiliations with Meals on Wheels focused on veteran outreach and providing hot meals for low-income, homebound, and disabled veterans.
Additional Resources
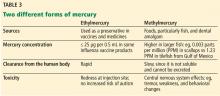
Although resources vary by regional setting, several key governmental and community-based food assistance programs are summarized in Table 2. Local community partners and online/phone-based directories, such as United Way’s 2-1-1 can help identify additional local resources. For older adults and individuals with disabilities, local Aging and Disability Resources Centers can provide information and assistance connecting to needed resources.69 Finally, there are a number of online resources available for clinicians interested in learning more about the impact of food insecurity on health and tools to use in the clinical setting (Table 3).
Conclusion
The VA has recognized food insecurity as a critical concern for the well-being of our nation’s veterans. Use of the EHR clinical reminder represents a crucial first step toward increasing provider awareness about veteran food insecurity and improving clinical efforts to address food insecurity once identified. Through the reminder, health care teams can connect veterans to needed resources and create both the individual and population-level data necessary to inform VHA and community efforts to address veteran food insecurity. Clinical reminder data are currently being used for local quality improvement efforts and have established the need nationally for formalized partnerships between VHA Social Work Services and Nutrition and Food Services to connect veterans with food and provide them with strategies to best use available food resources.
Moving forward, the Ensuring Veteran Food Security Workgroup continues to work with agencies and organizations across the country to improve food insecure veterans’ access to needed services. In addition to existing VA partnerships with Feeding America for the VPP, memorandums of understanding are currently underway to formalize partnerships with both the Food Research and Action Center (FRAC) and MAZON. Additional research is needed both to formally validate the current food insecurity clinical reminder screening question and to identify best practices and potential models for how to most effectively use VHA-community partnerships to address the unique needs of the veteran population.
Ensuring the food security of our nation’s veterans is essential to VA’s commitment to providing integrated, veteran-centered, whole person care. Toward that goal, VA health care teams are urged to use the clinical reminder and help connect food insecure veterans with relevant resources both within and outside of the VA health care system.
1. Coleman-Jensen A, Rabbitt MP, Gregory CA, Singh A. Household food security in the United States in 2017. http://www.ers.usda.gov/publications/pub-details/?pubid=90022. Published September 2018. Accessed December 9, 2019.
2. Berkowitz SA, Meigs JB, DeWalt D, et al. Material need insecurities, control of diabetes mellitus, and use of health care resources: results of the Measuring Economic Insecurity in Diabetes study. JAMA Intern Med. 2015;175(2):257-265.
3. Berkowitz SA, Seligman HK, Choudhry NK. Treat or eat: food insecurity, cost-related medication underuse, and unmet needs. Am J Med. 2014;127(4):303-310.e3.
4. Lyles CR, Seligman HK, Parker MM, et al. Financial strain and medication adherence among diabetes patients in an integrated health care delivery system: The Diabetes Study of Northern California (DISTANCE). Health Serv Res. 2016;51(2):610-624.
5. Seligman HK, Schillinger D. Hunger and socioeconomic disparities in chronic disease. N Engl J Med. 2010;363(1):6-9.
6. Narain K, Bean-Mayberry B, Washington DL, Canelo IA, Darling JE, Yano EM. Access to care and health outcomes among women veterans using veterans administration health care: association with food insufficiency. Womens Health Issues. 2018;28(3):267-272.
7. Gundersen C, Ziliak JP. Food insecurity and health outcomes. Health Aff. 2015;34(11):1830-1839.
8. Wang EA, McGinnis KA, Goulet J, et al; Veterans Aging Cohort Study Project Team. Food insecurity and health: data from the Veterans Aging Cohort Study. Public Health Rep. 2015;130(3):261-268.
9. Berkowitz SA, Berkowitz TSZ, Meigs JB, Wexler DJ. Trends in food insecurity for adults with cardiometabolic disease in the United States: 2005-2012. PloS One. 2017;12(6):e0179172.
10. Seligman HK, Laraia BA, Kushel MB. Food insecurity is associated with chronic disease among low-income NHANES participants. J Nutr. 2010;140(2):304-310.
11. Berkowitz SA, Baggett TP, Wexler DJ, Huskey KW, Wee CC. Food insecurity and metabolic control among U.S. adults with diabetes. Diabetes Care. 2013;36(10):3093-3099.
12. Seligman HK, Jacobs EA, López A, Tschann J, Fernandez A. Food insecurity and glycemic control among low-income patients with type 2 diabetes. Diabetes Care. 2012;35(2):233-238.
13. Banerjee T, Crews DC, Wesson DE, et al; CDC CKD Surveillance Team. Food insecurity, CKD, and subsequent ESRD in US adults. Am J Kidney Dis. 2017;70(1):38-47.
14. Bruening M, Dinour LM, Chavez JBR. Food insecurity and emotional health in the USA: a systematic narrative review of longitudinal research. Public Health Nutr. 2017;20(17):3200-3208.
15. Berkowitz SA, Basu S, Meigs JB, Seligman HK. Food insecurity and health care expenditures in the United States, 2011-2013. Health Serv Res. 2018;53(3):1600-1620.
16. Berkowitz SA, Seligman HK, Basu S. Impact of food insecurity and SNAP participation on healthcare utilization and expenditures. http://www.ukcpr.org/research/discussion-papers. Published 2017. Accessed December 9, 2019.
17. Kushel MB, Gupta R, Gee L, Haas JS. Housing instability and food insecurity as barriers to health care among low-income Americans. J Gen Intern Med. 2006;21(1):71-77.
18. Garcia SP, Haddix A, Barnett K. Incremental health care costs associated with food insecurity and chronic conditions among older adults. Chronic Dis. 2018;15:180058.
19. Berkowitz SA, Seligman HK, Meigs JB, Basu S. Food insecurity, healthcare utilization, and high cost: a longitudinal cohort study. Am J Manag Care. 2018;24(9):399-404.
20. Larson NI, Story MT, Nelson MC. Neighborhood environments: disparities in access to healthy foods in the U.S. Am J Prev Med. 2009;36(1):74-81.
21. Darmon N, Drewnowski A. Contribution of food prices and diet cost to socioeconomic disparities in diet quality and health: a systematic review and analysis. Nutr Rev. 2015;73(10):643-660.
22. Darmon N, Drewnowski A. Does social class predict diet quality? Am J Clin Nutr. 2008;87(5):1107-1117.
23. Drewnowski A. The cost of US foods as related to their nutritive value. Am J Clin Nutr. 2010;92(5):1181-1188.
24. Lucan SC, Maroko AR, Seitchik JL, Yoon DH, Sperry LE, Schechter CB. Unexpected neighborhood sources of food and drink: implications for research and community health. Am J Prev Med. 2018;55(2):e29-e38.
25. Castillo DC, Ramsey NL, Yu SS, Ricks M, Courville AB, Sumner AE. Inconsistent access to food and cardiometabolic disease: the effect of food insecurity. Curr Cardiovasc Risk Rep. 2012;6(3):245-250.
26. Seligman HK, Davis TC, Schillinger D, Wolf MS. Food insecurity is associated with hypoglycemia and poor diabetes self-management in a low-income sample with diabetes. J Health Care Poor Underserved. 2010;21(4):1227-1233.
27. Siefert K, Heflin CM, Corcoran ME, Williams DR. Food insufficiency and physical and mental health in a longitudinal survey of welfare recipients. J Health Soc Behav. 2004;45(2):171-186.
28. Mangurian C, Sreshta N, Seligman H. Food insecurity among adults with severe mental illness. Psychiatr Serv. 2013;64(9):931-932.
29. Melchior M, Caspi A, Howard LM, et al. Mental health context of food insecurity: a representative cohort of families with young children. Pediatrics. 2009;124(4):e564-e572.
30. Brostow DP, Gunzburger E, Abbate LM, Brenner LA, Thomas KS. Mental illness, not obesity status, is associated with food insecurity among the elderly in the health and retirement study. J Nutr Gerontol Geriatr. 2019;38(2):149-172.
31. Higashi RT, Craddock Lee SJ, Pezzia C, Quirk L, Leonard T, Pruitt SL. Family and social context contributes to the interplay of economic insecurity, food insecurity, and health. Ann Anthropol Pract. 2017;41(2):67-77.
32. O’Toole TP, Roberts CB, Johnson EE. Screening for food insecurity in six Veterans Administration clinics for the homeless, June-December 2015. Prev Chronic Dis. 2017;14:160375.
33. Feil DG, Pogach LM. Cognitive impairment is a major risk factor for serious hypoglycaemia; public health intervention is warranted. Evid Based Med. 2014;19(2):77.
34. Frith E, Loprinzi PD. Food insecurity and cognitive function in older adults: Brief report. Clin Nutr. 2018;37(5):1765-1768.
35. Herman D, Afulani P, Coleman-Jensen A, Harrison GG. Food insecurity and cost-related medication underuse among nonelderly adults in a nationally representative sample. Am J Public Health. 2015;105(10):e48-e59.
36. Tseng C-L, Soroka O, Maney M, Aron DC, Pogach LM. Assessing potential glycemic overtreatment in persons at hypoglycemic risk. JAMA Intern Med. 2014;174(2):259-268.
37. Vue MH, Setter SM. Drug-induced glucose alterations part 1: drug-induced hypoglycemia. Diabetes Spectr. 2011;24(3):171-177.
38. Seligman HK, Bolger AF, Guzman D, López A, Bibbins-Domingo K. Exhaustion of food budgets at month’s end and hospital admissions for hypoglycemia. Health Aff (Millwood). 2014;33(1):116-123.
39. US Department of Veterans Affairs, National Center for Veterans Analysis and Statistics. Veteran poverty trends. https://www.va.gov/vetdata/docs/specialreports/veteran_poverty_trends.pdf. Published May 2015. Accessed December 9, 2019.
40. Robbins KG, Ravi A. Veterans living paycheck to paycheck are under threat during budget debates. https://www.americanprogress.org/issues/poverty/news/2017/09/19/439023/veterans-living-paycheck-paycheck-threat-budget-debates. Published September 19, 2017. Accessed December 9, 2019.
41. Wilmoth JM, London AS, Heflin CM. Economic well-being among older-adult households: variation by veteran and disability status. J Gerontol Soc Work. 2015;58(4):399-419.
42. Brostow DP, Gunzburger E, Thomas KS. Food insecurity among veterans: findings from the health and retirement study. J Nutr Health Aging. 2017;21(10):1358-1364.
43. Pooler J, Mian P, Srinivasan M, Miller Z. Veterans and food insecurity. https://www.impaqint.com/sites/default/files/issue-briefs/VeteransFoodInsecurity_IssueBrief_V1.3.pdf. Published November 2018. Accessed December 9, 2019.
44. Schure MB, Katon JG, Wong E, Liu C-F. Food and housing insecurity and health status among U.S. adults with and without prior military service. SSM Popul Health. 2016;29(2):244-248.
45. Miller DP, Larson MJ, Byrne T, DeVoe E. Food insecurity in veteran households: findings from nationally representative data. Public Health Nutr. 2016;19(10):1731-1740.
46. Widome R, Jensen A, Bangerter A, Fu SS. Food insecurity among veterans of the US wars in Iraq and Afghanistan. Public Health Nutr. 2015;18(5):844-849.
47. London AS, Heflin CM. Supplemental Nutrition Assistance Program (SNAP) use among active-duty military personnel, veterans, and reservists. Popul Res Policy Rev. 2015;34(6):805-826.
48. Weinfield NS, Mills G, Borger C, et al. Hunger in America 2014. Natl rep prepared for Feeding America. https://www.feedingamerica.org/research/hunger-in-america. Published 2014. Accessed December 9, 2019.
49. Mabli J, Ohls J, Dragoset L, Castner L, Santos B. Measuring the Effect of Supplemental Nutrition Assistance Program (SNAP) Participation on Food Security. Washington, DC: US Department of Agriculture, Food and Nutrition Service; 2013.
50. Srinivasan M, Pooler JA. Cost-related medication nonadherence for older adults participating in SNAP, 2013–2015. Am J Public Health. 2017;108(2):224-230.
51. Heflin C, Hodges L, Mueser P. Supplemental Nutrition Assistance Progam benefits and emergency room visits for hypoglycaemia. Public Health Nutr. 2017;20(7):1314-1321.
52. Samuel LJ, Szanton SL, Cahill R, et al. Does the Supplemental Nutrition Assistance Program affect hospital utilization among older adults? The case of Maryland. Popul Health Manag. 2018;21(2):88-95.
53. Szanton SL, Samuel LJ, Cahill R, et al. Food assistance is associated with decreased nursing home admissions for Maryland’s dually eligible older adults. BMC Geriatr. 2017;17(1):162.
54. Carlson S, Keith-Jennings B. SNAP is linked with improved nutritional outcomes and lower health care costs. https://www.cbpp.org/research/food-assistance/snap-is-linked-with-improved-nutritional-outcomes-and-lower-health-care. Published January 17, 2018. Accessed December 10, 2019.
55. Keith-Jennings B, Cai L. SNAP helps almost 1.4 million low-income veterans, including thousands in every state. https://www.cbpp.org/research/food-assistance/snap-helps-almost-14-million-low-income-veterans-including-thousands-in. Updated November 8, 2018. Accessed December 10, 2019.
56. US Department of Health and Human Services. Older Americans Act nutrition programs. https://acl.gov/sites/default/files/news%202017-03/OAA-Nutrition_Programs_Fact_Sheet.pdf. Accessed December 10, 2019.
57. US Department of Veterans Affairs. About VHA. https://www.va.gov/health/aboutvha.asp. Accessed December 10, 2019.
58. US Department of Veterans Affairs. VA Corporate Data Warehouse.
59. Yano EM, Bair MJ, Carrasquillo O, Krein SL, Rubenstein LV. Patient aligned care teams (PACT): VA’s journey to implement patient-centered medical homes. J Gen Intern Med. 2014;29(suppl 2):S547-s549.
60. O’Toole TP, Pape L. Innovative efforts to address homelessness among veterans. N C Med J. 2015;76(5):311-314.
61. O’Toole TP, Johnson EE, Aiello R, Kane V, Pape L. Tailoring care to vulnerable populations by incorporating social determinants of health: the Veterans Health Administration’s “Homeless Patient Aligned Care Team” Program. Prev Chronic Dis. 2016;13:150567.
62. Marpadga S, Fernandez A, Leung J, Tang A, Seligman H, Murphy EJ. Challenges and successes with food resource referrals for food-insecure patients with diabetes. Perm J. 2019;23.
63. Stenmark SH, Steiner JF, Marpadga S, Debor M, Underhill K, Seligman H. Lessons learned from implementation of the food insecurity screening and referral program at Kaiser Permanente Colorado. Perm J. 2018;22.
64. Martel ML, Klein LR, Hager KA, Cutts DB. Emergency department experience with novel electronic medical record order for referral to food resources. West J Emerg Med. 2018;19(2):232-237.
65. Going C, Cohen AJ, Bares M, Christensen M. Interdisciplinary approaches to addressing the food insecure veteran. Veterans Health Administration Employee Education System webinar; October 30, 2018.
66. Feeding America Announces New Partnership With U.S. Department Of Veterans Affairs. https://www.prnewswire.com/news-releases/feeding-america-announces-new-partnership-with-us-department-of-veterans-affairs-300481891.html. Published June 29, 2017. Accessed December 10, 2019.
67. US Department of Veterans Affairs. State Veterans Affairs offices. https://www.va.gov/statedva.htm. Updated March 20, 2019. Accessed December 10, 2019.
68. US Department of Veterans Affairs. Directory of veterans service organizations. https://www.va.gov/vso. Updated December 24, 2013. Accessed December 10, 2019.
69. ACL Administration for Community Living. Aging and disability resource centers. https://acl.gov/programs/aging-and-disability-networks/aging-and-disability-resource-centers. Updated December 13, 2017. Accessed December 10, 2019.
70. Nutrition and Obesity Policy Research and Evaluation Network (NOPREN). Clinical screening algorithms. https://nopren.org/resource/download-food-insecurity-screening-and-referral-algorithms-for-adults-patients-living-with-diabetes-and-pediatric-patients. Accessed December 10, 2019.
Nearly 1 in 8 households—and 1 in 6 households with children—experienced food insecurity in 2017, defined as limited or uncertain availability of nutritionally adequate and safe foods.1 Food insecurity is often even more pronounced among households with individuals with acute or chronic medical conditions.2-6 Moreover, food insecurity is independently associated with a range of adverse health outcomes, including poorer control of diabetes mellitus, hypertension, depression and other major psychiatric disorders, HIV, and chronic lung and kidney disease, as well as poorer overall health status.7-14 Food insecurity also has been associated with increased health care costs and acute care utilization as well as increased probability of delayed or missed care.15-19
The relationship between food insecurity and poor health outcomes is a complex and often cyclic phenomenon (Figure 1). Poor nutritional status is fueled by limited access to healthful foods as well as increased reliance on calorie-dense and nutrient-poor “junk” foods, which are less expensive and often more readily available in low-income neighborhoods.5,20-24 These compensatory dietary patterns place individuals at higher risk for developing cardiometabolic conditions and for poor control of these conditions.5,8,9,12,25,26 Additionally, the physiological and psychological stressors of food insecurity may precipitate depression and anxiety or worsen existing mental health conditions, resulting in feelings of overwhelm and decreased self-management capacity.5,8,27-31 Food insecurity has further been associated with poor sleep, declines in cognitive function, and increased falls, particularly among the frail and elderly.32-34
Individuals experiencing food insecurity often report having to make trade-offs between food and other necessities, such as paying rent or utilities. Additional strategies to stretch limited resources include cost-related underuse of medication and delays in needed medical care.4,17,31,35 In a nationally representative survey among adults with at least 1 chronic medical condition, 1 in 3 reported having to choose between food and medicine; 11% were unable to afford either.3 Furthermore, the inability to reliably adhere to medication regimens that need to be taken with food can result in potentially life-threatening hypoglycemia (as can lack of food regardless of medication use).5,26,36 In addition to the more obvious risks of glucose-lowering medications, such as insulin and long-acting sulfonylureas in patients experiencing food insecurity, many drugs commonly used among nondiabetic adults such as ACE-inhibitors, β blockers, quinolones, and salicylates can also precipitate hypoglycemia, and food insecurity has been associated with experiences of hypoglycemia even among individuals without diabetes mellitus.32,37 In one study the risk for hospital admissions for hypoglycemia among low-income populations increased by 27% at the end of the month when food budgets were more likely to be exhausted.38 Worsening health status and increased emergency department visits and hospitalizations may then result in lost wages and mounting medical bills, contributing to further financial strain and worsening food insecurity.
Prevalence and Importance of Food Insecurity Among US Veterans
Nearly 1.5 million veterans in the US are living below the federal poverty level (FPL).39 An additional 2.4 million veterans are living paycheck to paycheck at < 200% of the FPL.40 Veterans living in poverty are at even higher risk than nonveterans for food insecurity, homelessness, and other material hardship.41
Estimates of food insecurity among veterans vary widely, ranging from 6% to 24%—nearly twice that of the general US population.8,42-45 Higher rates of food insecurity have been reported among certain high-risk subgroups, including veterans who served in Iraq and Afghanistan (27%), female veterans (28%), homeless and formerly homeless veterans (49%), and veterans with serious mental illness (35%).6,32,43,46 Additional risk factors for food insecurity specific to veteran populations include younger age, having recently left active-duty military service, and lower final military paygrade.42,45-47 As in the general population, veteran food insecurity is associated with a range of adverse health outcomes, including poorer overall health status as well as increased probability of delayed or missed care.6,8,32,42-44,46
Even among veterans enrolled in federal food assistance programs, many still struggle to afford nutritionally adequate foods. As one example, in a study of mostly male homeless and formerly homeless veterans, O’Toole and colleagues found that nearly half of those reporting food insecurity were already receiving federal food assistance benefits, and 22% relied on emergency food resources.32 Of households served by Feeding America food pantries and meal programs, 20% have a member who has served in the US military.48
Federal Programs To Address Food Insecurity
There are several important federal food assistance programs designed to help alleviate food insecurity. The Supplemental Nutrition Assistance Program (SNAP, formerly the Food Stamp program) is the largest federal food assistance program and provides low-income Americans with cash benefits to purchase food. SNAP has been shown to substantially reduce food insecurity.7,49 The program also is associated with significant decreases in cost-related medication nonadherence as well as reductions in health care costs and both acute care and nursing home utilization.16,50-54 Although nearly 1.4 million veterans live in SNAP-enrolled households, 59% of eligible veterans are not enrolled.43,55 Closing this SNAP eligibility-enrollment gap, has been a focus of recent efforts to improve long-term food security among veterans. There also are several federal food assistance programs for households with children, including the Special Supplemental Nutrition Program for Women, Infants, and Children (WIC) and school meals programs. Among federal nutrition programs for seniors, the Older American’s Act contains designated funding to support nutrition services for older adults, including congregate meal programs in community settings like senior centers, places of worship, and housing communities, and home-delivered meals through programs like Meals on Wheels.56
VHA Response to Food Insecurity
The Veterans Health Administration (VHA) is the country’s largest integrated, federally funded health care system.57 In November 2015, congressional briefings on veteran food insecurity organized by the national non-profit organization MAZON: A Jewish Response to Hunger and hosted with bipartisan support were provided to the US House and Senate. As a result of these briefings, VHA chartered the national Ensuring Veteran Food Security Workgroup with a mandate to partner with governmental and nonprofit agencies to “focus on the issue of food insecurity, the identification of veterans at risk, the needed training of VHA staff and the coordination of resources and initiatives to support the veterans for whom we care.” Building off a pilot in US Department of Veterans Affairs (VA) Homeless Patient Aligned Care Teams (H-PACTs),32 VHA subsequently integrated a single-item food insecurity screening tool into the VA electronic health record (EHR) clinical reminder system (Figure 2). The clinical reminder, which was rolled out across VA medical centers nationally in October 2017, provides an alert to screen all noninstitutionalized veterans for food insecurity. To date, nearly 5 million veterans have been screened. When a veteran endorses food insecurity based on the initial screening question, a prompt appears to offer the veteran a referral to a social worker and/or dietitian. Positive screening results also should be communicated to the patient’s primary care provider. Depending on site-specific clinical flow, the reminders are typically completed in the outpatient setting either by nurses or medical assistants during intake or by providers as part of the clinical visit. However, any member of the health care team can complete the clinical reminder at any time. As of September 2019, approximately 74,000 veterans have been identified as food insecure.58
Addressing Food Insecurity
VHA has been a recognized leader in addressing homelessness and other social determinants of health through its integrated care and PACT delivery models.59-61 The food insecurity clinical reminder was designed to facilitate a tailored, interdisciplinary approach to identify and address food insecurity. Interdisciplinary care team members—including medical assistants, clinicians, social workers, registered dietitians, nurse care managers, occupational or physical therapists, and pharmacists—are uniquely positioned to identify veterans impacted by food insecurity, assess for associated clinical and/or social risk factors, and offer appropriate medical and nutrition interventions and resource referrals.
This interdisciplinary team-based model is essential given the range of potential drivers underlying veteran experiences of food insecurity and subsequent health outcomes. It is critically important for clinicians to review the medication list with veterans screening positive for food insecurity to assess for risk of hypoglycemia and/or cost-related nonadherence, make any necessary adjustments to therapeutic regimens, and assess for additional risk factors associated with food insecurity. Examples of tailored nutrition counseling that clinical dietitians may provide include meal preparation strategies for veterans who only have access to a microwave or hotplate, or recommendations for how veterans on medically restricted diets can best navigate food selection at soup kitchens or food pantries. Resource referrals provided by social workers or other care team members may include both emergency food resources to address immediate shortages (eg, food pantries, soup kitchens, or vouchers for free lunch) as well as resources focused on improving longer term food security (eg, federal food assistance programs or home delivered meal programs). Importantly, although providing a list of food resources may be helpful for some patients, such lists are often insufficient.62,63 Many patients require active assistance with program enrollment either onsite the day of their clinic visit or through connection with a partnering community-based organization that can, in turn, identify appropriate resources and facilitate program enrollment.63,64 Planned follow-up is also crucial to determine whether referrals are successful and to assess for ongoing need. Proposed roles for interdisciplinary care team members in addressing a positive food insecurity screen are outlined in Table 1.
VHA-Community Partnerships
In addition to services offered within VA, public and private sector partnerships can greatly enhance the range of resources available to food insecure veterans. Several VA facilities have developed formal community partnerships, such as the Veterans Pantry Pilot (VPP) program, a national partnership between Feeding America food banks and VA medical centers to establish onsite or mobile food pantries. There are currently 17 active Feeding America VPP sites, with a number of additional sites under development. Several of the VPP sites also include other “wraparound services,” such as SNAP application assistance.65,66
State Veterans Affairs offices67 and Veterans Service Organizations (VSOs)68 also can serve as valuable partners for connecting veterans with needed resources. VSOs offer a range of services, including assistancewith benefit claims, employment and housing assistance, emergency food assistance, and transportation to medical appointments. Some VSOs also have established local affiliations with Meals on Wheels focused on veteran outreach and providing hot meals for low-income, homebound, and disabled veterans.
Additional Resources
Although resources vary by regional setting, several key governmental and community-based food assistance programs are summarized in Table 2. Local community partners and online/phone-based directories, such as United Way’s 2-1-1 can help identify additional local resources. For older adults and individuals with disabilities, local Aging and Disability Resources Centers can provide information and assistance connecting to needed resources.69 Finally, there are a number of online resources available for clinicians interested in learning more about the impact of food insecurity on health and tools to use in the clinical setting (Table 3).
Conclusion
The VA has recognized food insecurity as a critical concern for the well-being of our nation’s veterans. Use of the EHR clinical reminder represents a crucial first step toward increasing provider awareness about veteran food insecurity and improving clinical efforts to address food insecurity once identified. Through the reminder, health care teams can connect veterans to needed resources and create both the individual and population-level data necessary to inform VHA and community efforts to address veteran food insecurity. Clinical reminder data are currently being used for local quality improvement efforts and have established the need nationally for formalized partnerships between VHA Social Work Services and Nutrition and Food Services to connect veterans with food and provide them with strategies to best use available food resources.
Moving forward, the Ensuring Veteran Food Security Workgroup continues to work with agencies and organizations across the country to improve food insecure veterans’ access to needed services. In addition to existing VA partnerships with Feeding America for the VPP, memorandums of understanding are currently underway to formalize partnerships with both the Food Research and Action Center (FRAC) and MAZON. Additional research is needed both to formally validate the current food insecurity clinical reminder screening question and to identify best practices and potential models for how to most effectively use VHA-community partnerships to address the unique needs of the veteran population.
Ensuring the food security of our nation’s veterans is essential to VA’s commitment to providing integrated, veteran-centered, whole person care. Toward that goal, VA health care teams are urged to use the clinical reminder and help connect food insecure veterans with relevant resources both within and outside of the VA health care system.
Nearly 1 in 8 households—and 1 in 6 households with children—experienced food insecurity in 2017, defined as limited or uncertain availability of nutritionally adequate and safe foods.1 Food insecurity is often even more pronounced among households with individuals with acute or chronic medical conditions.2-6 Moreover, food insecurity is independently associated with a range of adverse health outcomes, including poorer control of diabetes mellitus, hypertension, depression and other major psychiatric disorders, HIV, and chronic lung and kidney disease, as well as poorer overall health status.7-14 Food insecurity also has been associated with increased health care costs and acute care utilization as well as increased probability of delayed or missed care.15-19
The relationship between food insecurity and poor health outcomes is a complex and often cyclic phenomenon (Figure 1). Poor nutritional status is fueled by limited access to healthful foods as well as increased reliance on calorie-dense and nutrient-poor “junk” foods, which are less expensive and often more readily available in low-income neighborhoods.5,20-24 These compensatory dietary patterns place individuals at higher risk for developing cardiometabolic conditions and for poor control of these conditions.5,8,9,12,25,26 Additionally, the physiological and psychological stressors of food insecurity may precipitate depression and anxiety or worsen existing mental health conditions, resulting in feelings of overwhelm and decreased self-management capacity.5,8,27-31 Food insecurity has further been associated with poor sleep, declines in cognitive function, and increased falls, particularly among the frail and elderly.32-34
Individuals experiencing food insecurity often report having to make trade-offs between food and other necessities, such as paying rent or utilities. Additional strategies to stretch limited resources include cost-related underuse of medication and delays in needed medical care.4,17,31,35 In a nationally representative survey among adults with at least 1 chronic medical condition, 1 in 3 reported having to choose between food and medicine; 11% were unable to afford either.3 Furthermore, the inability to reliably adhere to medication regimens that need to be taken with food can result in potentially life-threatening hypoglycemia (as can lack of food regardless of medication use).5,26,36 In addition to the more obvious risks of glucose-lowering medications, such as insulin and long-acting sulfonylureas in patients experiencing food insecurity, many drugs commonly used among nondiabetic adults such as ACE-inhibitors, β blockers, quinolones, and salicylates can also precipitate hypoglycemia, and food insecurity has been associated with experiences of hypoglycemia even among individuals without diabetes mellitus.32,37 In one study the risk for hospital admissions for hypoglycemia among low-income populations increased by 27% at the end of the month when food budgets were more likely to be exhausted.38 Worsening health status and increased emergency department visits and hospitalizations may then result in lost wages and mounting medical bills, contributing to further financial strain and worsening food insecurity.
Prevalence and Importance of Food Insecurity Among US Veterans
Nearly 1.5 million veterans in the US are living below the federal poverty level (FPL).39 An additional 2.4 million veterans are living paycheck to paycheck at < 200% of the FPL.40 Veterans living in poverty are at even higher risk than nonveterans for food insecurity, homelessness, and other material hardship.41
Estimates of food insecurity among veterans vary widely, ranging from 6% to 24%—nearly twice that of the general US population.8,42-45 Higher rates of food insecurity have been reported among certain high-risk subgroups, including veterans who served in Iraq and Afghanistan (27%), female veterans (28%), homeless and formerly homeless veterans (49%), and veterans with serious mental illness (35%).6,32,43,46 Additional risk factors for food insecurity specific to veteran populations include younger age, having recently left active-duty military service, and lower final military paygrade.42,45-47 As in the general population, veteran food insecurity is associated with a range of adverse health outcomes, including poorer overall health status as well as increased probability of delayed or missed care.6,8,32,42-44,46
Even among veterans enrolled in federal food assistance programs, many still struggle to afford nutritionally adequate foods. As one example, in a study of mostly male homeless and formerly homeless veterans, O’Toole and colleagues found that nearly half of those reporting food insecurity were already receiving federal food assistance benefits, and 22% relied on emergency food resources.32 Of households served by Feeding America food pantries and meal programs, 20% have a member who has served in the US military.48
Federal Programs To Address Food Insecurity
There are several important federal food assistance programs designed to help alleviate food insecurity. The Supplemental Nutrition Assistance Program (SNAP, formerly the Food Stamp program) is the largest federal food assistance program and provides low-income Americans with cash benefits to purchase food. SNAP has been shown to substantially reduce food insecurity.7,49 The program also is associated with significant decreases in cost-related medication nonadherence as well as reductions in health care costs and both acute care and nursing home utilization.16,50-54 Although nearly 1.4 million veterans live in SNAP-enrolled households, 59% of eligible veterans are not enrolled.43,55 Closing this SNAP eligibility-enrollment gap, has been a focus of recent efforts to improve long-term food security among veterans. There also are several federal food assistance programs for households with children, including the Special Supplemental Nutrition Program for Women, Infants, and Children (WIC) and school meals programs. Among federal nutrition programs for seniors, the Older American’s Act contains designated funding to support nutrition services for older adults, including congregate meal programs in community settings like senior centers, places of worship, and housing communities, and home-delivered meals through programs like Meals on Wheels.56
VHA Response to Food Insecurity
The Veterans Health Administration (VHA) is the country’s largest integrated, federally funded health care system.57 In November 2015, congressional briefings on veteran food insecurity organized by the national non-profit organization MAZON: A Jewish Response to Hunger and hosted with bipartisan support were provided to the US House and Senate. As a result of these briefings, VHA chartered the national Ensuring Veteran Food Security Workgroup with a mandate to partner with governmental and nonprofit agencies to “focus on the issue of food insecurity, the identification of veterans at risk, the needed training of VHA staff and the coordination of resources and initiatives to support the veterans for whom we care.” Building off a pilot in US Department of Veterans Affairs (VA) Homeless Patient Aligned Care Teams (H-PACTs),32 VHA subsequently integrated a single-item food insecurity screening tool into the VA electronic health record (EHR) clinical reminder system (Figure 2). The clinical reminder, which was rolled out across VA medical centers nationally in October 2017, provides an alert to screen all noninstitutionalized veterans for food insecurity. To date, nearly 5 million veterans have been screened. When a veteran endorses food insecurity based on the initial screening question, a prompt appears to offer the veteran a referral to a social worker and/or dietitian. Positive screening results also should be communicated to the patient’s primary care provider. Depending on site-specific clinical flow, the reminders are typically completed in the outpatient setting either by nurses or medical assistants during intake or by providers as part of the clinical visit. However, any member of the health care team can complete the clinical reminder at any time. As of September 2019, approximately 74,000 veterans have been identified as food insecure.58
Addressing Food Insecurity
VHA has been a recognized leader in addressing homelessness and other social determinants of health through its integrated care and PACT delivery models.59-61 The food insecurity clinical reminder was designed to facilitate a tailored, interdisciplinary approach to identify and address food insecurity. Interdisciplinary care team members—including medical assistants, clinicians, social workers, registered dietitians, nurse care managers, occupational or physical therapists, and pharmacists—are uniquely positioned to identify veterans impacted by food insecurity, assess for associated clinical and/or social risk factors, and offer appropriate medical and nutrition interventions and resource referrals.
This interdisciplinary team-based model is essential given the range of potential drivers underlying veteran experiences of food insecurity and subsequent health outcomes. It is critically important for clinicians to review the medication list with veterans screening positive for food insecurity to assess for risk of hypoglycemia and/or cost-related nonadherence, make any necessary adjustments to therapeutic regimens, and assess for additional risk factors associated with food insecurity. Examples of tailored nutrition counseling that clinical dietitians may provide include meal preparation strategies for veterans who only have access to a microwave or hotplate, or recommendations for how veterans on medically restricted diets can best navigate food selection at soup kitchens or food pantries. Resource referrals provided by social workers or other care team members may include both emergency food resources to address immediate shortages (eg, food pantries, soup kitchens, or vouchers for free lunch) as well as resources focused on improving longer term food security (eg, federal food assistance programs or home delivered meal programs). Importantly, although providing a list of food resources may be helpful for some patients, such lists are often insufficient.62,63 Many patients require active assistance with program enrollment either onsite the day of their clinic visit or through connection with a partnering community-based organization that can, in turn, identify appropriate resources and facilitate program enrollment.63,64 Planned follow-up is also crucial to determine whether referrals are successful and to assess for ongoing need. Proposed roles for interdisciplinary care team members in addressing a positive food insecurity screen are outlined in Table 1.
VHA-Community Partnerships
In addition to services offered within VA, public and private sector partnerships can greatly enhance the range of resources available to food insecure veterans. Several VA facilities have developed formal community partnerships, such as the Veterans Pantry Pilot (VPP) program, a national partnership between Feeding America food banks and VA medical centers to establish onsite or mobile food pantries. There are currently 17 active Feeding America VPP sites, with a number of additional sites under development. Several of the VPP sites also include other “wraparound services,” such as SNAP application assistance.65,66
State Veterans Affairs offices67 and Veterans Service Organizations (VSOs)68 also can serve as valuable partners for connecting veterans with needed resources. VSOs offer a range of services, including assistancewith benefit claims, employment and housing assistance, emergency food assistance, and transportation to medical appointments. Some VSOs also have established local affiliations with Meals on Wheels focused on veteran outreach and providing hot meals for low-income, homebound, and disabled veterans.
Additional Resources
Although resources vary by regional setting, several key governmental and community-based food assistance programs are summarized in Table 2. Local community partners and online/phone-based directories, such as United Way’s 2-1-1 can help identify additional local resources. For older adults and individuals with disabilities, local Aging and Disability Resources Centers can provide information and assistance connecting to needed resources.69 Finally, there are a number of online resources available for clinicians interested in learning more about the impact of food insecurity on health and tools to use in the clinical setting (Table 3).
Conclusion
The VA has recognized food insecurity as a critical concern for the well-being of our nation’s veterans. Use of the EHR clinical reminder represents a crucial first step toward increasing provider awareness about veteran food insecurity and improving clinical efforts to address food insecurity once identified. Through the reminder, health care teams can connect veterans to needed resources and create both the individual and population-level data necessary to inform VHA and community efforts to address veteran food insecurity. Clinical reminder data are currently being used for local quality improvement efforts and have established the need nationally for formalized partnerships between VHA Social Work Services and Nutrition and Food Services to connect veterans with food and provide them with strategies to best use available food resources.
Moving forward, the Ensuring Veteran Food Security Workgroup continues to work with agencies and organizations across the country to improve food insecure veterans’ access to needed services. In addition to existing VA partnerships with Feeding America for the VPP, memorandums of understanding are currently underway to formalize partnerships with both the Food Research and Action Center (FRAC) and MAZON. Additional research is needed both to formally validate the current food insecurity clinical reminder screening question and to identify best practices and potential models for how to most effectively use VHA-community partnerships to address the unique needs of the veteran population.
Ensuring the food security of our nation’s veterans is essential to VA’s commitment to providing integrated, veteran-centered, whole person care. Toward that goal, VA health care teams are urged to use the clinical reminder and help connect food insecure veterans with relevant resources both within and outside of the VA health care system.
1. Coleman-Jensen A, Rabbitt MP, Gregory CA, Singh A. Household food security in the United States in 2017. http://www.ers.usda.gov/publications/pub-details/?pubid=90022. Published September 2018. Accessed December 9, 2019.
2. Berkowitz SA, Meigs JB, DeWalt D, et al. Material need insecurities, control of diabetes mellitus, and use of health care resources: results of the Measuring Economic Insecurity in Diabetes study. JAMA Intern Med. 2015;175(2):257-265.
3. Berkowitz SA, Seligman HK, Choudhry NK. Treat or eat: food insecurity, cost-related medication underuse, and unmet needs. Am J Med. 2014;127(4):303-310.e3.
4. Lyles CR, Seligman HK, Parker MM, et al. Financial strain and medication adherence among diabetes patients in an integrated health care delivery system: The Diabetes Study of Northern California (DISTANCE). Health Serv Res. 2016;51(2):610-624.
5. Seligman HK, Schillinger D. Hunger and socioeconomic disparities in chronic disease. N Engl J Med. 2010;363(1):6-9.
6. Narain K, Bean-Mayberry B, Washington DL, Canelo IA, Darling JE, Yano EM. Access to care and health outcomes among women veterans using veterans administration health care: association with food insufficiency. Womens Health Issues. 2018;28(3):267-272.
7. Gundersen C, Ziliak JP. Food insecurity and health outcomes. Health Aff. 2015;34(11):1830-1839.
8. Wang EA, McGinnis KA, Goulet J, et al; Veterans Aging Cohort Study Project Team. Food insecurity and health: data from the Veterans Aging Cohort Study. Public Health Rep. 2015;130(3):261-268.
9. Berkowitz SA, Berkowitz TSZ, Meigs JB, Wexler DJ. Trends in food insecurity for adults with cardiometabolic disease in the United States: 2005-2012. PloS One. 2017;12(6):e0179172.
10. Seligman HK, Laraia BA, Kushel MB. Food insecurity is associated with chronic disease among low-income NHANES participants. J Nutr. 2010;140(2):304-310.
11. Berkowitz SA, Baggett TP, Wexler DJ, Huskey KW, Wee CC. Food insecurity and metabolic control among U.S. adults with diabetes. Diabetes Care. 2013;36(10):3093-3099.
12. Seligman HK, Jacobs EA, López A, Tschann J, Fernandez A. Food insecurity and glycemic control among low-income patients with type 2 diabetes. Diabetes Care. 2012;35(2):233-238.
13. Banerjee T, Crews DC, Wesson DE, et al; CDC CKD Surveillance Team. Food insecurity, CKD, and subsequent ESRD in US adults. Am J Kidney Dis. 2017;70(1):38-47.
14. Bruening M, Dinour LM, Chavez JBR. Food insecurity and emotional health in the USA: a systematic narrative review of longitudinal research. Public Health Nutr. 2017;20(17):3200-3208.
15. Berkowitz SA, Basu S, Meigs JB, Seligman HK. Food insecurity and health care expenditures in the United States, 2011-2013. Health Serv Res. 2018;53(3):1600-1620.
16. Berkowitz SA, Seligman HK, Basu S. Impact of food insecurity and SNAP participation on healthcare utilization and expenditures. http://www.ukcpr.org/research/discussion-papers. Published 2017. Accessed December 9, 2019.
17. Kushel MB, Gupta R, Gee L, Haas JS. Housing instability and food insecurity as barriers to health care among low-income Americans. J Gen Intern Med. 2006;21(1):71-77.
18. Garcia SP, Haddix A, Barnett K. Incremental health care costs associated with food insecurity and chronic conditions among older adults. Chronic Dis. 2018;15:180058.
19. Berkowitz SA, Seligman HK, Meigs JB, Basu S. Food insecurity, healthcare utilization, and high cost: a longitudinal cohort study. Am J Manag Care. 2018;24(9):399-404.
20. Larson NI, Story MT, Nelson MC. Neighborhood environments: disparities in access to healthy foods in the U.S. Am J Prev Med. 2009;36(1):74-81.
21. Darmon N, Drewnowski A. Contribution of food prices and diet cost to socioeconomic disparities in diet quality and health: a systematic review and analysis. Nutr Rev. 2015;73(10):643-660.
22. Darmon N, Drewnowski A. Does social class predict diet quality? Am J Clin Nutr. 2008;87(5):1107-1117.
23. Drewnowski A. The cost of US foods as related to their nutritive value. Am J Clin Nutr. 2010;92(5):1181-1188.
24. Lucan SC, Maroko AR, Seitchik JL, Yoon DH, Sperry LE, Schechter CB. Unexpected neighborhood sources of food and drink: implications for research and community health. Am J Prev Med. 2018;55(2):e29-e38.
25. Castillo DC, Ramsey NL, Yu SS, Ricks M, Courville AB, Sumner AE. Inconsistent access to food and cardiometabolic disease: the effect of food insecurity. Curr Cardiovasc Risk Rep. 2012;6(3):245-250.
26. Seligman HK, Davis TC, Schillinger D, Wolf MS. Food insecurity is associated with hypoglycemia and poor diabetes self-management in a low-income sample with diabetes. J Health Care Poor Underserved. 2010;21(4):1227-1233.
27. Siefert K, Heflin CM, Corcoran ME, Williams DR. Food insufficiency and physical and mental health in a longitudinal survey of welfare recipients. J Health Soc Behav. 2004;45(2):171-186.
28. Mangurian C, Sreshta N, Seligman H. Food insecurity among adults with severe mental illness. Psychiatr Serv. 2013;64(9):931-932.
29. Melchior M, Caspi A, Howard LM, et al. Mental health context of food insecurity: a representative cohort of families with young children. Pediatrics. 2009;124(4):e564-e572.
30. Brostow DP, Gunzburger E, Abbate LM, Brenner LA, Thomas KS. Mental illness, not obesity status, is associated with food insecurity among the elderly in the health and retirement study. J Nutr Gerontol Geriatr. 2019;38(2):149-172.
31. Higashi RT, Craddock Lee SJ, Pezzia C, Quirk L, Leonard T, Pruitt SL. Family and social context contributes to the interplay of economic insecurity, food insecurity, and health. Ann Anthropol Pract. 2017;41(2):67-77.
32. O’Toole TP, Roberts CB, Johnson EE. Screening for food insecurity in six Veterans Administration clinics for the homeless, June-December 2015. Prev Chronic Dis. 2017;14:160375.
33. Feil DG, Pogach LM. Cognitive impairment is a major risk factor for serious hypoglycaemia; public health intervention is warranted. Evid Based Med. 2014;19(2):77.
34. Frith E, Loprinzi PD. Food insecurity and cognitive function in older adults: Brief report. Clin Nutr. 2018;37(5):1765-1768.
35. Herman D, Afulani P, Coleman-Jensen A, Harrison GG. Food insecurity and cost-related medication underuse among nonelderly adults in a nationally representative sample. Am J Public Health. 2015;105(10):e48-e59.
36. Tseng C-L, Soroka O, Maney M, Aron DC, Pogach LM. Assessing potential glycemic overtreatment in persons at hypoglycemic risk. JAMA Intern Med. 2014;174(2):259-268.
37. Vue MH, Setter SM. Drug-induced glucose alterations part 1: drug-induced hypoglycemia. Diabetes Spectr. 2011;24(3):171-177.
38. Seligman HK, Bolger AF, Guzman D, López A, Bibbins-Domingo K. Exhaustion of food budgets at month’s end and hospital admissions for hypoglycemia. Health Aff (Millwood). 2014;33(1):116-123.
39. US Department of Veterans Affairs, National Center for Veterans Analysis and Statistics. Veteran poverty trends. https://www.va.gov/vetdata/docs/specialreports/veteran_poverty_trends.pdf. Published May 2015. Accessed December 9, 2019.
40. Robbins KG, Ravi A. Veterans living paycheck to paycheck are under threat during budget debates. https://www.americanprogress.org/issues/poverty/news/2017/09/19/439023/veterans-living-paycheck-paycheck-threat-budget-debates. Published September 19, 2017. Accessed December 9, 2019.
41. Wilmoth JM, London AS, Heflin CM. Economic well-being among older-adult households: variation by veteran and disability status. J Gerontol Soc Work. 2015;58(4):399-419.
42. Brostow DP, Gunzburger E, Thomas KS. Food insecurity among veterans: findings from the health and retirement study. J Nutr Health Aging. 2017;21(10):1358-1364.
43. Pooler J, Mian P, Srinivasan M, Miller Z. Veterans and food insecurity. https://www.impaqint.com/sites/default/files/issue-briefs/VeteransFoodInsecurity_IssueBrief_V1.3.pdf. Published November 2018. Accessed December 9, 2019.
44. Schure MB, Katon JG, Wong E, Liu C-F. Food and housing insecurity and health status among U.S. adults with and without prior military service. SSM Popul Health. 2016;29(2):244-248.
45. Miller DP, Larson MJ, Byrne T, DeVoe E. Food insecurity in veteran households: findings from nationally representative data. Public Health Nutr. 2016;19(10):1731-1740.
46. Widome R, Jensen A, Bangerter A, Fu SS. Food insecurity among veterans of the US wars in Iraq and Afghanistan. Public Health Nutr. 2015;18(5):844-849.
47. London AS, Heflin CM. Supplemental Nutrition Assistance Program (SNAP) use among active-duty military personnel, veterans, and reservists. Popul Res Policy Rev. 2015;34(6):805-826.
48. Weinfield NS, Mills G, Borger C, et al. Hunger in America 2014. Natl rep prepared for Feeding America. https://www.feedingamerica.org/research/hunger-in-america. Published 2014. Accessed December 9, 2019.
49. Mabli J, Ohls J, Dragoset L, Castner L, Santos B. Measuring the Effect of Supplemental Nutrition Assistance Program (SNAP) Participation on Food Security. Washington, DC: US Department of Agriculture, Food and Nutrition Service; 2013.
50. Srinivasan M, Pooler JA. Cost-related medication nonadherence for older adults participating in SNAP, 2013–2015. Am J Public Health. 2017;108(2):224-230.
51. Heflin C, Hodges L, Mueser P. Supplemental Nutrition Assistance Progam benefits and emergency room visits for hypoglycaemia. Public Health Nutr. 2017;20(7):1314-1321.
52. Samuel LJ, Szanton SL, Cahill R, et al. Does the Supplemental Nutrition Assistance Program affect hospital utilization among older adults? The case of Maryland. Popul Health Manag. 2018;21(2):88-95.
53. Szanton SL, Samuel LJ, Cahill R, et al. Food assistance is associated with decreased nursing home admissions for Maryland’s dually eligible older adults. BMC Geriatr. 2017;17(1):162.
54. Carlson S, Keith-Jennings B. SNAP is linked with improved nutritional outcomes and lower health care costs. https://www.cbpp.org/research/food-assistance/snap-is-linked-with-improved-nutritional-outcomes-and-lower-health-care. Published January 17, 2018. Accessed December 10, 2019.
55. Keith-Jennings B, Cai L. SNAP helps almost 1.4 million low-income veterans, including thousands in every state. https://www.cbpp.org/research/food-assistance/snap-helps-almost-14-million-low-income-veterans-including-thousands-in. Updated November 8, 2018. Accessed December 10, 2019.
56. US Department of Health and Human Services. Older Americans Act nutrition programs. https://acl.gov/sites/default/files/news%202017-03/OAA-Nutrition_Programs_Fact_Sheet.pdf. Accessed December 10, 2019.
57. US Department of Veterans Affairs. About VHA. https://www.va.gov/health/aboutvha.asp. Accessed December 10, 2019.
58. US Department of Veterans Affairs. VA Corporate Data Warehouse.
59. Yano EM, Bair MJ, Carrasquillo O, Krein SL, Rubenstein LV. Patient aligned care teams (PACT): VA’s journey to implement patient-centered medical homes. J Gen Intern Med. 2014;29(suppl 2):S547-s549.
60. O’Toole TP, Pape L. Innovative efforts to address homelessness among veterans. N C Med J. 2015;76(5):311-314.
61. O’Toole TP, Johnson EE, Aiello R, Kane V, Pape L. Tailoring care to vulnerable populations by incorporating social determinants of health: the Veterans Health Administration’s “Homeless Patient Aligned Care Team” Program. Prev Chronic Dis. 2016;13:150567.
62. Marpadga S, Fernandez A, Leung J, Tang A, Seligman H, Murphy EJ. Challenges and successes with food resource referrals for food-insecure patients with diabetes. Perm J. 2019;23.
63. Stenmark SH, Steiner JF, Marpadga S, Debor M, Underhill K, Seligman H. Lessons learned from implementation of the food insecurity screening and referral program at Kaiser Permanente Colorado. Perm J. 2018;22.
64. Martel ML, Klein LR, Hager KA, Cutts DB. Emergency department experience with novel electronic medical record order for referral to food resources. West J Emerg Med. 2018;19(2):232-237.
65. Going C, Cohen AJ, Bares M, Christensen M. Interdisciplinary approaches to addressing the food insecure veteran. Veterans Health Administration Employee Education System webinar; October 30, 2018.
66. Feeding America Announces New Partnership With U.S. Department Of Veterans Affairs. https://www.prnewswire.com/news-releases/feeding-america-announces-new-partnership-with-us-department-of-veterans-affairs-300481891.html. Published June 29, 2017. Accessed December 10, 2019.
67. US Department of Veterans Affairs. State Veterans Affairs offices. https://www.va.gov/statedva.htm. Updated March 20, 2019. Accessed December 10, 2019.
68. US Department of Veterans Affairs. Directory of veterans service organizations. https://www.va.gov/vso. Updated December 24, 2013. Accessed December 10, 2019.
69. ACL Administration for Community Living. Aging and disability resource centers. https://acl.gov/programs/aging-and-disability-networks/aging-and-disability-resource-centers. Updated December 13, 2017. Accessed December 10, 2019.
70. Nutrition and Obesity Policy Research and Evaluation Network (NOPREN). Clinical screening algorithms. https://nopren.org/resource/download-food-insecurity-screening-and-referral-algorithms-for-adults-patients-living-with-diabetes-and-pediatric-patients. Accessed December 10, 2019.
1. Coleman-Jensen A, Rabbitt MP, Gregory CA, Singh A. Household food security in the United States in 2017. http://www.ers.usda.gov/publications/pub-details/?pubid=90022. Published September 2018. Accessed December 9, 2019.
2. Berkowitz SA, Meigs JB, DeWalt D, et al. Material need insecurities, control of diabetes mellitus, and use of health care resources: results of the Measuring Economic Insecurity in Diabetes study. JAMA Intern Med. 2015;175(2):257-265.
3. Berkowitz SA, Seligman HK, Choudhry NK. Treat or eat: food insecurity, cost-related medication underuse, and unmet needs. Am J Med. 2014;127(4):303-310.e3.
4. Lyles CR, Seligman HK, Parker MM, et al. Financial strain and medication adherence among diabetes patients in an integrated health care delivery system: The Diabetes Study of Northern California (DISTANCE). Health Serv Res. 2016;51(2):610-624.
5. Seligman HK, Schillinger D. Hunger and socioeconomic disparities in chronic disease. N Engl J Med. 2010;363(1):6-9.
6. Narain K, Bean-Mayberry B, Washington DL, Canelo IA, Darling JE, Yano EM. Access to care and health outcomes among women veterans using veterans administration health care: association with food insufficiency. Womens Health Issues. 2018;28(3):267-272.
7. Gundersen C, Ziliak JP. Food insecurity and health outcomes. Health Aff. 2015;34(11):1830-1839.
8. Wang EA, McGinnis KA, Goulet J, et al; Veterans Aging Cohort Study Project Team. Food insecurity and health: data from the Veterans Aging Cohort Study. Public Health Rep. 2015;130(3):261-268.
9. Berkowitz SA, Berkowitz TSZ, Meigs JB, Wexler DJ. Trends in food insecurity for adults with cardiometabolic disease in the United States: 2005-2012. PloS One. 2017;12(6):e0179172.
10. Seligman HK, Laraia BA, Kushel MB. Food insecurity is associated with chronic disease among low-income NHANES participants. J Nutr. 2010;140(2):304-310.
11. Berkowitz SA, Baggett TP, Wexler DJ, Huskey KW, Wee CC. Food insecurity and metabolic control among U.S. adults with diabetes. Diabetes Care. 2013;36(10):3093-3099.
12. Seligman HK, Jacobs EA, López A, Tschann J, Fernandez A. Food insecurity and glycemic control among low-income patients with type 2 diabetes. Diabetes Care. 2012;35(2):233-238.
13. Banerjee T, Crews DC, Wesson DE, et al; CDC CKD Surveillance Team. Food insecurity, CKD, and subsequent ESRD in US adults. Am J Kidney Dis. 2017;70(1):38-47.
14. Bruening M, Dinour LM, Chavez JBR. Food insecurity and emotional health in the USA: a systematic narrative review of longitudinal research. Public Health Nutr. 2017;20(17):3200-3208.
15. Berkowitz SA, Basu S, Meigs JB, Seligman HK. Food insecurity and health care expenditures in the United States, 2011-2013. Health Serv Res. 2018;53(3):1600-1620.
16. Berkowitz SA, Seligman HK, Basu S. Impact of food insecurity and SNAP participation on healthcare utilization and expenditures. http://www.ukcpr.org/research/discussion-papers. Published 2017. Accessed December 9, 2019.
17. Kushel MB, Gupta R, Gee L, Haas JS. Housing instability and food insecurity as barriers to health care among low-income Americans. J Gen Intern Med. 2006;21(1):71-77.
18. Garcia SP, Haddix A, Barnett K. Incremental health care costs associated with food insecurity and chronic conditions among older adults. Chronic Dis. 2018;15:180058.
19. Berkowitz SA, Seligman HK, Meigs JB, Basu S. Food insecurity, healthcare utilization, and high cost: a longitudinal cohort study. Am J Manag Care. 2018;24(9):399-404.
20. Larson NI, Story MT, Nelson MC. Neighborhood environments: disparities in access to healthy foods in the U.S. Am J Prev Med. 2009;36(1):74-81.
21. Darmon N, Drewnowski A. Contribution of food prices and diet cost to socioeconomic disparities in diet quality and health: a systematic review and analysis. Nutr Rev. 2015;73(10):643-660.
22. Darmon N, Drewnowski A. Does social class predict diet quality? Am J Clin Nutr. 2008;87(5):1107-1117.
23. Drewnowski A. The cost of US foods as related to their nutritive value. Am J Clin Nutr. 2010;92(5):1181-1188.
24. Lucan SC, Maroko AR, Seitchik JL, Yoon DH, Sperry LE, Schechter CB. Unexpected neighborhood sources of food and drink: implications for research and community health. Am J Prev Med. 2018;55(2):e29-e38.
25. Castillo DC, Ramsey NL, Yu SS, Ricks M, Courville AB, Sumner AE. Inconsistent access to food and cardiometabolic disease: the effect of food insecurity. Curr Cardiovasc Risk Rep. 2012;6(3):245-250.
26. Seligman HK, Davis TC, Schillinger D, Wolf MS. Food insecurity is associated with hypoglycemia and poor diabetes self-management in a low-income sample with diabetes. J Health Care Poor Underserved. 2010;21(4):1227-1233.
27. Siefert K, Heflin CM, Corcoran ME, Williams DR. Food insufficiency and physical and mental health in a longitudinal survey of welfare recipients. J Health Soc Behav. 2004;45(2):171-186.
28. Mangurian C, Sreshta N, Seligman H. Food insecurity among adults with severe mental illness. Psychiatr Serv. 2013;64(9):931-932.
29. Melchior M, Caspi A, Howard LM, et al. Mental health context of food insecurity: a representative cohort of families with young children. Pediatrics. 2009;124(4):e564-e572.
30. Brostow DP, Gunzburger E, Abbate LM, Brenner LA, Thomas KS. Mental illness, not obesity status, is associated with food insecurity among the elderly in the health and retirement study. J Nutr Gerontol Geriatr. 2019;38(2):149-172.
31. Higashi RT, Craddock Lee SJ, Pezzia C, Quirk L, Leonard T, Pruitt SL. Family and social context contributes to the interplay of economic insecurity, food insecurity, and health. Ann Anthropol Pract. 2017;41(2):67-77.
32. O’Toole TP, Roberts CB, Johnson EE. Screening for food insecurity in six Veterans Administration clinics for the homeless, June-December 2015. Prev Chronic Dis. 2017;14:160375.
33. Feil DG, Pogach LM. Cognitive impairment is a major risk factor for serious hypoglycaemia; public health intervention is warranted. Evid Based Med. 2014;19(2):77.
34. Frith E, Loprinzi PD. Food insecurity and cognitive function in older adults: Brief report. Clin Nutr. 2018;37(5):1765-1768.
35. Herman D, Afulani P, Coleman-Jensen A, Harrison GG. Food insecurity and cost-related medication underuse among nonelderly adults in a nationally representative sample. Am J Public Health. 2015;105(10):e48-e59.
36. Tseng C-L, Soroka O, Maney M, Aron DC, Pogach LM. Assessing potential glycemic overtreatment in persons at hypoglycemic risk. JAMA Intern Med. 2014;174(2):259-268.
37. Vue MH, Setter SM. Drug-induced glucose alterations part 1: drug-induced hypoglycemia. Diabetes Spectr. 2011;24(3):171-177.
38. Seligman HK, Bolger AF, Guzman D, López A, Bibbins-Domingo K. Exhaustion of food budgets at month’s end and hospital admissions for hypoglycemia. Health Aff (Millwood). 2014;33(1):116-123.
39. US Department of Veterans Affairs, National Center for Veterans Analysis and Statistics. Veteran poverty trends. https://www.va.gov/vetdata/docs/specialreports/veteran_poverty_trends.pdf. Published May 2015. Accessed December 9, 2019.
40. Robbins KG, Ravi A. Veterans living paycheck to paycheck are under threat during budget debates. https://www.americanprogress.org/issues/poverty/news/2017/09/19/439023/veterans-living-paycheck-paycheck-threat-budget-debates. Published September 19, 2017. Accessed December 9, 2019.
41. Wilmoth JM, London AS, Heflin CM. Economic well-being among older-adult households: variation by veteran and disability status. J Gerontol Soc Work. 2015;58(4):399-419.
42. Brostow DP, Gunzburger E, Thomas KS. Food insecurity among veterans: findings from the health and retirement study. J Nutr Health Aging. 2017;21(10):1358-1364.
43. Pooler J, Mian P, Srinivasan M, Miller Z. Veterans and food insecurity. https://www.impaqint.com/sites/default/files/issue-briefs/VeteransFoodInsecurity_IssueBrief_V1.3.pdf. Published November 2018. Accessed December 9, 2019.
44. Schure MB, Katon JG, Wong E, Liu C-F. Food and housing insecurity and health status among U.S. adults with and without prior military service. SSM Popul Health. 2016;29(2):244-248.
45. Miller DP, Larson MJ, Byrne T, DeVoe E. Food insecurity in veteran households: findings from nationally representative data. Public Health Nutr. 2016;19(10):1731-1740.
46. Widome R, Jensen A, Bangerter A, Fu SS. Food insecurity among veterans of the US wars in Iraq and Afghanistan. Public Health Nutr. 2015;18(5):844-849.
47. London AS, Heflin CM. Supplemental Nutrition Assistance Program (SNAP) use among active-duty military personnel, veterans, and reservists. Popul Res Policy Rev. 2015;34(6):805-826.
48. Weinfield NS, Mills G, Borger C, et al. Hunger in America 2014. Natl rep prepared for Feeding America. https://www.feedingamerica.org/research/hunger-in-america. Published 2014. Accessed December 9, 2019.
49. Mabli J, Ohls J, Dragoset L, Castner L, Santos B. Measuring the Effect of Supplemental Nutrition Assistance Program (SNAP) Participation on Food Security. Washington, DC: US Department of Agriculture, Food and Nutrition Service; 2013.
50. Srinivasan M, Pooler JA. Cost-related medication nonadherence for older adults participating in SNAP, 2013–2015. Am J Public Health. 2017;108(2):224-230.
51. Heflin C, Hodges L, Mueser P. Supplemental Nutrition Assistance Progam benefits and emergency room visits for hypoglycaemia. Public Health Nutr. 2017;20(7):1314-1321.
52. Samuel LJ, Szanton SL, Cahill R, et al. Does the Supplemental Nutrition Assistance Program affect hospital utilization among older adults? The case of Maryland. Popul Health Manag. 2018;21(2):88-95.
53. Szanton SL, Samuel LJ, Cahill R, et al. Food assistance is associated with decreased nursing home admissions for Maryland’s dually eligible older adults. BMC Geriatr. 2017;17(1):162.
54. Carlson S, Keith-Jennings B. SNAP is linked with improved nutritional outcomes and lower health care costs. https://www.cbpp.org/research/food-assistance/snap-is-linked-with-improved-nutritional-outcomes-and-lower-health-care. Published January 17, 2018. Accessed December 10, 2019.
55. Keith-Jennings B, Cai L. SNAP helps almost 1.4 million low-income veterans, including thousands in every state. https://www.cbpp.org/research/food-assistance/snap-helps-almost-14-million-low-income-veterans-including-thousands-in. Updated November 8, 2018. Accessed December 10, 2019.
56. US Department of Health and Human Services. Older Americans Act nutrition programs. https://acl.gov/sites/default/files/news%202017-03/OAA-Nutrition_Programs_Fact_Sheet.pdf. Accessed December 10, 2019.
57. US Department of Veterans Affairs. About VHA. https://www.va.gov/health/aboutvha.asp. Accessed December 10, 2019.
58. US Department of Veterans Affairs. VA Corporate Data Warehouse.
59. Yano EM, Bair MJ, Carrasquillo O, Krein SL, Rubenstein LV. Patient aligned care teams (PACT): VA’s journey to implement patient-centered medical homes. J Gen Intern Med. 2014;29(suppl 2):S547-s549.
60. O’Toole TP, Pape L. Innovative efforts to address homelessness among veterans. N C Med J. 2015;76(5):311-314.
61. O’Toole TP, Johnson EE, Aiello R, Kane V, Pape L. Tailoring care to vulnerable populations by incorporating social determinants of health: the Veterans Health Administration’s “Homeless Patient Aligned Care Team” Program. Prev Chronic Dis. 2016;13:150567.
62. Marpadga S, Fernandez A, Leung J, Tang A, Seligman H, Murphy EJ. Challenges and successes with food resource referrals for food-insecure patients with diabetes. Perm J. 2019;23.
63. Stenmark SH, Steiner JF, Marpadga S, Debor M, Underhill K, Seligman H. Lessons learned from implementation of the food insecurity screening and referral program at Kaiser Permanente Colorado. Perm J. 2018;22.
64. Martel ML, Klein LR, Hager KA, Cutts DB. Emergency department experience with novel electronic medical record order for referral to food resources. West J Emerg Med. 2018;19(2):232-237.
65. Going C, Cohen AJ, Bares M, Christensen M. Interdisciplinary approaches to addressing the food insecure veteran. Veterans Health Administration Employee Education System webinar; October 30, 2018.
66. Feeding America Announces New Partnership With U.S. Department Of Veterans Affairs. https://www.prnewswire.com/news-releases/feeding-america-announces-new-partnership-with-us-department-of-veterans-affairs-300481891.html. Published June 29, 2017. Accessed December 10, 2019.
67. US Department of Veterans Affairs. State Veterans Affairs offices. https://www.va.gov/statedva.htm. Updated March 20, 2019. Accessed December 10, 2019.
68. US Department of Veterans Affairs. Directory of veterans service organizations. https://www.va.gov/vso. Updated December 24, 2013. Accessed December 10, 2019.
69. ACL Administration for Community Living. Aging and disability resource centers. https://acl.gov/programs/aging-and-disability-networks/aging-and-disability-resource-centers. Updated December 13, 2017. Accessed December 10, 2019.
70. Nutrition and Obesity Policy Research and Evaluation Network (NOPREN). Clinical screening algorithms. https://nopren.org/resource/download-food-insecurity-screening-and-referral-algorithms-for-adults-patients-living-with-diabetes-and-pediatric-patients. Accessed December 10, 2019.
Biofeedback corrects dyssynergic constipation in elderly
SAN ANTONIO – Biofeedback for treatment of dyssynergic constipation is highly effective in the elderly, just as it is in younger patients, Samantha Spilman, MD, reported at the annual meeting of the American College of Gastroenterology.
“I think the main point of this study is that older adults have a profound burden of constipation with dyssynergic defecation, and we propose that biofeedback be given strong consideration as first-line therapy for this population, in whom overall we’re trying to reduce medication use,” said Dr. Spilman, a gastroenterology fellow at the University of California, San Diego.
The prevalence of constipation in older patients is estimated to be up to 40%. Yet few prior studies have scrutinized how well older patients with constipation actually respond to biofeedback. It’s a legitimate question, since biofeedback training involves operant conditioning and requires learning new techniques. For this reason, she and her coinvestigators conducted a retrospective analysis of 58 patients over age 65 referred from the university’s gastrointestinal motility and physiology program to the biofeedback program for treatment of dyssynergic defection. The patients’ mean age was 74 years, with a 9.5-year history of constipation. The oldest patient was 88. Most of the subjects were high school graduates. Thirteen of the 58 carried a diagnosis of irritable bowel syndrome.
Numerous studies have demonstrated that 70%-80% of younger adults with dyssynergic constipation experience marked improvement in response to biofeedback training, which typically utilizes an inflated rectal balloon to simulate retained stool. The key finding in Dr. Spilman’s study was that the elderly patients did comparably well in terms of both self-reported outcomes and objective high-resolution anorectal manometric parameters upon completing an average of three biofeedback sessions.
Mean global bowel satisfaction on a 1-10 scale nearly doubled from 2.77 at baseline to 5.01 with biofeedback. Moreover, 79% of seniors demonstrated resolution of their dyssynergia on high-resolution anorectal manometry performed with sensors in the rectum and anal canal. The proportion of patients who reported a feeling of incomplete evacuation after stooling – a sensation individuals with constipation find highly bothersome – improved from 95% to 24% with biofeedback.
The strongest response in terms of the defecation index was observed in older patients with type 2 dyssynergia, characterized by defective propulsion coupled with a paradoxical contraction of the sphincter muscles during defecation. Their defecation index score, derived by dividing intrarectal pressure by residual intra-anal pressure during simulated defection, showed a robust improvement from 0.307 at baseline to 0.793. Patients with types 1 and 3 dyssynergia showed lesser improvements on this objective measure.
Dr. Spilman noted as a study limitation that baseline cognitive status wasn’t formally assessed, so the investigators don’t know how many of the older patients had minimal cognitive impairment. However, baseline quality of life assessment via the Short Form-36 indicated that patients scored average or above for physical and social functioning as well as emotional well-being.
Dr. Spilman reported having no financial conflicts regarding her study, conducted free of commercial support.
SOURCE: Spilman S. ACG 2019. Abstract 45.
SAN ANTONIO – Biofeedback for treatment of dyssynergic constipation is highly effective in the elderly, just as it is in younger patients, Samantha Spilman, MD, reported at the annual meeting of the American College of Gastroenterology.
“I think the main point of this study is that older adults have a profound burden of constipation with dyssynergic defecation, and we propose that biofeedback be given strong consideration as first-line therapy for this population, in whom overall we’re trying to reduce medication use,” said Dr. Spilman, a gastroenterology fellow at the University of California, San Diego.
The prevalence of constipation in older patients is estimated to be up to 40%. Yet few prior studies have scrutinized how well older patients with constipation actually respond to biofeedback. It’s a legitimate question, since biofeedback training involves operant conditioning and requires learning new techniques. For this reason, she and her coinvestigators conducted a retrospective analysis of 58 patients over age 65 referred from the university’s gastrointestinal motility and physiology program to the biofeedback program for treatment of dyssynergic defection. The patients’ mean age was 74 years, with a 9.5-year history of constipation. The oldest patient was 88. Most of the subjects were high school graduates. Thirteen of the 58 carried a diagnosis of irritable bowel syndrome.
Numerous studies have demonstrated that 70%-80% of younger adults with dyssynergic constipation experience marked improvement in response to biofeedback training, which typically utilizes an inflated rectal balloon to simulate retained stool. The key finding in Dr. Spilman’s study was that the elderly patients did comparably well in terms of both self-reported outcomes and objective high-resolution anorectal manometric parameters upon completing an average of three biofeedback sessions.
Mean global bowel satisfaction on a 1-10 scale nearly doubled from 2.77 at baseline to 5.01 with biofeedback. Moreover, 79% of seniors demonstrated resolution of their dyssynergia on high-resolution anorectal manometry performed with sensors in the rectum and anal canal. The proportion of patients who reported a feeling of incomplete evacuation after stooling – a sensation individuals with constipation find highly bothersome – improved from 95% to 24% with biofeedback.
The strongest response in terms of the defecation index was observed in older patients with type 2 dyssynergia, characterized by defective propulsion coupled with a paradoxical contraction of the sphincter muscles during defecation. Their defecation index score, derived by dividing intrarectal pressure by residual intra-anal pressure during simulated defection, showed a robust improvement from 0.307 at baseline to 0.793. Patients with types 1 and 3 dyssynergia showed lesser improvements on this objective measure.
Dr. Spilman noted as a study limitation that baseline cognitive status wasn’t formally assessed, so the investigators don’t know how many of the older patients had minimal cognitive impairment. However, baseline quality of life assessment via the Short Form-36 indicated that patients scored average or above for physical and social functioning as well as emotional well-being.
Dr. Spilman reported having no financial conflicts regarding her study, conducted free of commercial support.
SOURCE: Spilman S. ACG 2019. Abstract 45.
SAN ANTONIO – Biofeedback for treatment of dyssynergic constipation is highly effective in the elderly, just as it is in younger patients, Samantha Spilman, MD, reported at the annual meeting of the American College of Gastroenterology.
“I think the main point of this study is that older adults have a profound burden of constipation with dyssynergic defecation, and we propose that biofeedback be given strong consideration as first-line therapy for this population, in whom overall we’re trying to reduce medication use,” said Dr. Spilman, a gastroenterology fellow at the University of California, San Diego.
The prevalence of constipation in older patients is estimated to be up to 40%. Yet few prior studies have scrutinized how well older patients with constipation actually respond to biofeedback. It’s a legitimate question, since biofeedback training involves operant conditioning and requires learning new techniques. For this reason, she and her coinvestigators conducted a retrospective analysis of 58 patients over age 65 referred from the university’s gastrointestinal motility and physiology program to the biofeedback program for treatment of dyssynergic defection. The patients’ mean age was 74 years, with a 9.5-year history of constipation. The oldest patient was 88. Most of the subjects were high school graduates. Thirteen of the 58 carried a diagnosis of irritable bowel syndrome.
Numerous studies have demonstrated that 70%-80% of younger adults with dyssynergic constipation experience marked improvement in response to biofeedback training, which typically utilizes an inflated rectal balloon to simulate retained stool. The key finding in Dr. Spilman’s study was that the elderly patients did comparably well in terms of both self-reported outcomes and objective high-resolution anorectal manometric parameters upon completing an average of three biofeedback sessions.
Mean global bowel satisfaction on a 1-10 scale nearly doubled from 2.77 at baseline to 5.01 with biofeedback. Moreover, 79% of seniors demonstrated resolution of their dyssynergia on high-resolution anorectal manometry performed with sensors in the rectum and anal canal. The proportion of patients who reported a feeling of incomplete evacuation after stooling – a sensation individuals with constipation find highly bothersome – improved from 95% to 24% with biofeedback.
The strongest response in terms of the defecation index was observed in older patients with type 2 dyssynergia, characterized by defective propulsion coupled with a paradoxical contraction of the sphincter muscles during defecation. Their defecation index score, derived by dividing intrarectal pressure by residual intra-anal pressure during simulated defection, showed a robust improvement from 0.307 at baseline to 0.793. Patients with types 1 and 3 dyssynergia showed lesser improvements on this objective measure.
Dr. Spilman noted as a study limitation that baseline cognitive status wasn’t formally assessed, so the investigators don’t know how many of the older patients had minimal cognitive impairment. However, baseline quality of life assessment via the Short Form-36 indicated that patients scored average or above for physical and social functioning as well as emotional well-being.
Dr. Spilman reported having no financial conflicts regarding her study, conducted free of commercial support.
SOURCE: Spilman S. ACG 2019. Abstract 45.
REPORTING FROM ACG 2019
Could preventing dementia be as simple as following your mom’s advice?
SAN DIEGO – To prevent dementia, follow Mom’s advice: Get up off the couch, go play with your friends, and eat your vegetables.
After 15 years of disappointing drug trials, strong new evidence says the best way to attack Alzheimer’s disease is not to treat it once it develops, but to prevent it in the first place, Laura D. Baker, PhD, said at the Clinical Trials on Alzheimer’s Disease conference. Studies of exercise, cognitive and social stimulation, and diet show that each one can reduce the risk of dementia, and that a combination of all three may have even a more powerful and synergistic effect.
“We have become absolutely phobic of exercise,” said Dr. Baker of Wake Forest University, Winston-Salem, N.C. And it’s not just structured exercise we shirk. “We take the closest parking space, sit for hours on end, don’t even take the stairs. Yet we know from years of work that exercise has a powerful benefit on cardiovascular disease, lipid profiles, metabolic disease, stress, and mood. Now we are seeing that exercise also promotes brain health in normal aging and protects against cognitive decline and prevention.”
Get off the couch
The general benefits of exercise – chiefly aerobic exercise – are myriad, Dr. Baker said.
“Exercise increases effective neurorepair. It reduces oxidative stress. It improves insulin sensitivity and helps with maintaining normal weight. It reduces inflammation and increases normal clearance of amyloid-beta.”
A 2017 meta-analysis reviewed some of these findings. “The current review [of 16 studies] suggests that aerobic exercise may have positive effects on the right hippocampus and potentially beneficial effects on the overall and other parts of the hippocampus, the cingulate cortex, and the medial temporal areas. ... Moreover, aerobic exercise may increase functional connectivity or activation in the hippocampus, cingulate cortex, and parahippocampal gyrus regions,” wrote Mo-yi Li, PhD, of Fujian University of Traditional Chinese Medicine, Fuzhou, China, and colleagues.
Exercise increases brain-derived neurotrophic factor (BDNF), which in turn increases neuronal potentiation and synaptic plasticity. BDNF is also important in hippocampal neurogenesis; mice, after just one aerobic session, showed dramatic boosts in BDNF. A 2018 review elaborates on these findings.
Eat right
Diet mediates dementia risk through less direct, but very effective, pathways, Dr. Baker said. Diets rich in vegetables, berries, nuts, fish, lean proteins, and healthy fats improves virtually all metabolic measures. These, in turn, reduce the risk cerebrovascular disease – an important driver of vascular dementias and a contributor to Alzheimer’s disease risk as well.
The MIND diet study (Mediterranean-DASH Diet Intervention for Neurodegenerative Delay), reported in 2015 was a very successful demonstration of this concept. A combination of the Mediterranean diet and the DASH diet (Dietary Approaches to Stop Hypertension), the MIND diet stresses frequent consumption of vegetables – especially leafy greens – as well as nuts, berries, whole grains, fish, poultry, and wine or grape juice. In the large, nearly 5-year study of 923 subjects aged 58-98 years, the MIND diet was associated with significant gains in cognition – equivalent to a 7-year reversal of age. After 4.5 years, those who strictly adhered to the diet had a 53% reduction in risk for Alzheimer’s disease, and those who adhered moderately had a 35% reduction. And in a more recent Australian longitudinal study, the MIND diet was associated with a 53% reduced risk of cognitive impairment over 12 years.
Ketogenic diets also may exert a benefit. Theoretically, a state of ketosis forces the brain to burn ketones as an alternative fuel to glucose, thus boosting brain function in glucose-starved brains. A small pilot study with exploratory cognitive endpoints determined that diet-compliant subjects with mild to moderate Alzheimer’s experienced a mean 5-point improvement in the Alzheimer’s Disease Assessment Score–Cognition. They reverted to baseline scores within a month of ending the study.
Recent initial work into the gut microbiome provides some additional speculative, but interesting, data. A dysregulated microbiome can shift microbial populations toward a more inflammatory profile. Some work suggests that inflammatory cytokines then travel to the brain and induce a hyperresponse of neuron-damaging immune cells. A comprehensive review article discusses the complicated mechanisms that may be in play.
Play with your friends
Cognitive stimulation and social interaction also appear to modify dementia risk, although the data are a little more limited. But personal interaction is a key element of Dr. Baker’s ongoing EXERT trial.
The ongoing phase 3 trial randomized 300 adults with amnestic mild cognitive impairment to moderate to high intensity aerobic exercise plus one-on-one support at local YMCA gyms or a low-intensity stretching, balance, and range of motion program. In additional to cognitive testing, the trial includes brain imaging, cerebrospinal fluid sampling for biomarkers of Alzheimer’s disease, and a sleep study.
A key component is personal interaction with a trainer. “They spend a lot of one-on-one time with each person,” Dr. Baker said. “For me, that’s the crucial ingredient – that personal touch. It’s what helps people move from Point A to Point B in their behaviors.”
Virtual cognitive stimulation is also a burgeoning area of dementia prevention research right now. Numerous studies are ongoing to test whether virtual reality or other computer-based games might keep the mind sharp or even improve cognition in people at risk.
The power of three
If one lifestyle change can reduce dementia risk, what happens when all three work together?
That’s the newest question, first successfully explored in the mid-2000s, with the FINGER study (Finnish Geriatric Intervention Study to Prevent Cognitive Impairment and Disability). In FINGER, the triad of exercise, personal support at the gym, and a modified Mediterranean diet reduced Alzheimer’s disease risk and improved cognition relative to the control group.
FINGER showed that the intervention was feasible and that it was associated with cognitive preservation and reduced Alzheimer’s disease risk in a group of at-risk subjects. The active group also had a 25% greater improvement on a neuropsychological test battery relative to the control group. They also performed 150% better in processing speed, 83% better in executive function, and 40% better in short-term memory. They showed no increased risk of cognitive decline relative to the control group, which experienced a 30% increase in risk, according to lead investigator Miia Kivipelto, PhD, of the Karolinska Institute, Stockholm.
So successful was FINGER that it launched a global consortium of related studies called World Wide FINGERS. Active in six countries now, including the United States, the studies aim to discover whether such combinations of lifestyle interventions are workable across countries and cultures. World Wide FINGERS is largely supported by the Alzheimer’s Association.
Global enthusiasm for lifestyle interventions
In recognition of the importance of lifestyle changes for dementia prevention, the World Health Organization recently published “Risk reduction of cognitive decline and dementia.” The document reviews many studies and makes recommendations regarding not only exercise, diet, and cognitive stimulation, but also smoking and alcohol.
Research interest in these areas is surging, Dr. Baker said. “The [U.S.] National Institute on Aging now has 29 ongoing trials. There’s a strong commitment to investigations into how lifestyle interventions could protect brain health as we get older. Certainly, many fit and healthy people do develop Alzheimer’s. But for some, it could be medicine.”
But no matter how compliant people are, lifestyle changes will never completely rid the world of Alzheimer’s and other dementias. The view of Dr. Baker – and most other Alzheimer’s researchers – is to employ lifestyle changes to reduce risk as much as possible and not to stop when cognitive problems do present.
“We need to understand how lifestyle interventions might work in combination with pharmaceuticals,” she said. “If we can support the health of the body and the health of the mind, lifestyle interventions can be the fertilizer that would help drug therapy have its maximum effect.”
SAN DIEGO – To prevent dementia, follow Mom’s advice: Get up off the couch, go play with your friends, and eat your vegetables.
After 15 years of disappointing drug trials, strong new evidence says the best way to attack Alzheimer’s disease is not to treat it once it develops, but to prevent it in the first place, Laura D. Baker, PhD, said at the Clinical Trials on Alzheimer’s Disease conference. Studies of exercise, cognitive and social stimulation, and diet show that each one can reduce the risk of dementia, and that a combination of all three may have even a more powerful and synergistic effect.
“We have become absolutely phobic of exercise,” said Dr. Baker of Wake Forest University, Winston-Salem, N.C. And it’s not just structured exercise we shirk. “We take the closest parking space, sit for hours on end, don’t even take the stairs. Yet we know from years of work that exercise has a powerful benefit on cardiovascular disease, lipid profiles, metabolic disease, stress, and mood. Now we are seeing that exercise also promotes brain health in normal aging and protects against cognitive decline and prevention.”
Get off the couch
The general benefits of exercise – chiefly aerobic exercise – are myriad, Dr. Baker said.
“Exercise increases effective neurorepair. It reduces oxidative stress. It improves insulin sensitivity and helps with maintaining normal weight. It reduces inflammation and increases normal clearance of amyloid-beta.”
A 2017 meta-analysis reviewed some of these findings. “The current review [of 16 studies] suggests that aerobic exercise may have positive effects on the right hippocampus and potentially beneficial effects on the overall and other parts of the hippocampus, the cingulate cortex, and the medial temporal areas. ... Moreover, aerobic exercise may increase functional connectivity or activation in the hippocampus, cingulate cortex, and parahippocampal gyrus regions,” wrote Mo-yi Li, PhD, of Fujian University of Traditional Chinese Medicine, Fuzhou, China, and colleagues.
Exercise increases brain-derived neurotrophic factor (BDNF), which in turn increases neuronal potentiation and synaptic plasticity. BDNF is also important in hippocampal neurogenesis; mice, after just one aerobic session, showed dramatic boosts in BDNF. A 2018 review elaborates on these findings.
Eat right
Diet mediates dementia risk through less direct, but very effective, pathways, Dr. Baker said. Diets rich in vegetables, berries, nuts, fish, lean proteins, and healthy fats improves virtually all metabolic measures. These, in turn, reduce the risk cerebrovascular disease – an important driver of vascular dementias and a contributor to Alzheimer’s disease risk as well.
The MIND diet study (Mediterranean-DASH Diet Intervention for Neurodegenerative Delay), reported in 2015 was a very successful demonstration of this concept. A combination of the Mediterranean diet and the DASH diet (Dietary Approaches to Stop Hypertension), the MIND diet stresses frequent consumption of vegetables – especially leafy greens – as well as nuts, berries, whole grains, fish, poultry, and wine or grape juice. In the large, nearly 5-year study of 923 subjects aged 58-98 years, the MIND diet was associated with significant gains in cognition – equivalent to a 7-year reversal of age. After 4.5 years, those who strictly adhered to the diet had a 53% reduction in risk for Alzheimer’s disease, and those who adhered moderately had a 35% reduction. And in a more recent Australian longitudinal study, the MIND diet was associated with a 53% reduced risk of cognitive impairment over 12 years.
Ketogenic diets also may exert a benefit. Theoretically, a state of ketosis forces the brain to burn ketones as an alternative fuel to glucose, thus boosting brain function in glucose-starved brains. A small pilot study with exploratory cognitive endpoints determined that diet-compliant subjects with mild to moderate Alzheimer’s experienced a mean 5-point improvement in the Alzheimer’s Disease Assessment Score–Cognition. They reverted to baseline scores within a month of ending the study.
Recent initial work into the gut microbiome provides some additional speculative, but interesting, data. A dysregulated microbiome can shift microbial populations toward a more inflammatory profile. Some work suggests that inflammatory cytokines then travel to the brain and induce a hyperresponse of neuron-damaging immune cells. A comprehensive review article discusses the complicated mechanisms that may be in play.
Play with your friends
Cognitive stimulation and social interaction also appear to modify dementia risk, although the data are a little more limited. But personal interaction is a key element of Dr. Baker’s ongoing EXERT trial.
The ongoing phase 3 trial randomized 300 adults with amnestic mild cognitive impairment to moderate to high intensity aerobic exercise plus one-on-one support at local YMCA gyms or a low-intensity stretching, balance, and range of motion program. In additional to cognitive testing, the trial includes brain imaging, cerebrospinal fluid sampling for biomarkers of Alzheimer’s disease, and a sleep study.
A key component is personal interaction with a trainer. “They spend a lot of one-on-one time with each person,” Dr. Baker said. “For me, that’s the crucial ingredient – that personal touch. It’s what helps people move from Point A to Point B in their behaviors.”
Virtual cognitive stimulation is also a burgeoning area of dementia prevention research right now. Numerous studies are ongoing to test whether virtual reality or other computer-based games might keep the mind sharp or even improve cognition in people at risk.
The power of three
If one lifestyle change can reduce dementia risk, what happens when all three work together?
That’s the newest question, first successfully explored in the mid-2000s, with the FINGER study (Finnish Geriatric Intervention Study to Prevent Cognitive Impairment and Disability). In FINGER, the triad of exercise, personal support at the gym, and a modified Mediterranean diet reduced Alzheimer’s disease risk and improved cognition relative to the control group.
FINGER showed that the intervention was feasible and that it was associated with cognitive preservation and reduced Alzheimer’s disease risk in a group of at-risk subjects. The active group also had a 25% greater improvement on a neuropsychological test battery relative to the control group. They also performed 150% better in processing speed, 83% better in executive function, and 40% better in short-term memory. They showed no increased risk of cognitive decline relative to the control group, which experienced a 30% increase in risk, according to lead investigator Miia Kivipelto, PhD, of the Karolinska Institute, Stockholm.
So successful was FINGER that it launched a global consortium of related studies called World Wide FINGERS. Active in six countries now, including the United States, the studies aim to discover whether such combinations of lifestyle interventions are workable across countries and cultures. World Wide FINGERS is largely supported by the Alzheimer’s Association.
Global enthusiasm for lifestyle interventions
In recognition of the importance of lifestyle changes for dementia prevention, the World Health Organization recently published “Risk reduction of cognitive decline and dementia.” The document reviews many studies and makes recommendations regarding not only exercise, diet, and cognitive stimulation, but also smoking and alcohol.
Research interest in these areas is surging, Dr. Baker said. “The [U.S.] National Institute on Aging now has 29 ongoing trials. There’s a strong commitment to investigations into how lifestyle interventions could protect brain health as we get older. Certainly, many fit and healthy people do develop Alzheimer’s. But for some, it could be medicine.”
But no matter how compliant people are, lifestyle changes will never completely rid the world of Alzheimer’s and other dementias. The view of Dr. Baker – and most other Alzheimer’s researchers – is to employ lifestyle changes to reduce risk as much as possible and not to stop when cognitive problems do present.
“We need to understand how lifestyle interventions might work in combination with pharmaceuticals,” she said. “If we can support the health of the body and the health of the mind, lifestyle interventions can be the fertilizer that would help drug therapy have its maximum effect.”
SAN DIEGO – To prevent dementia, follow Mom’s advice: Get up off the couch, go play with your friends, and eat your vegetables.
After 15 years of disappointing drug trials, strong new evidence says the best way to attack Alzheimer’s disease is not to treat it once it develops, but to prevent it in the first place, Laura D. Baker, PhD, said at the Clinical Trials on Alzheimer’s Disease conference. Studies of exercise, cognitive and social stimulation, and diet show that each one can reduce the risk of dementia, and that a combination of all three may have even a more powerful and synergistic effect.
“We have become absolutely phobic of exercise,” said Dr. Baker of Wake Forest University, Winston-Salem, N.C. And it’s not just structured exercise we shirk. “We take the closest parking space, sit for hours on end, don’t even take the stairs. Yet we know from years of work that exercise has a powerful benefit on cardiovascular disease, lipid profiles, metabolic disease, stress, and mood. Now we are seeing that exercise also promotes brain health in normal aging and protects against cognitive decline and prevention.”
Get off the couch
The general benefits of exercise – chiefly aerobic exercise – are myriad, Dr. Baker said.
“Exercise increases effective neurorepair. It reduces oxidative stress. It improves insulin sensitivity and helps with maintaining normal weight. It reduces inflammation and increases normal clearance of amyloid-beta.”
A 2017 meta-analysis reviewed some of these findings. “The current review [of 16 studies] suggests that aerobic exercise may have positive effects on the right hippocampus and potentially beneficial effects on the overall and other parts of the hippocampus, the cingulate cortex, and the medial temporal areas. ... Moreover, aerobic exercise may increase functional connectivity or activation in the hippocampus, cingulate cortex, and parahippocampal gyrus regions,” wrote Mo-yi Li, PhD, of Fujian University of Traditional Chinese Medicine, Fuzhou, China, and colleagues.
Exercise increases brain-derived neurotrophic factor (BDNF), which in turn increases neuronal potentiation and synaptic plasticity. BDNF is also important in hippocampal neurogenesis; mice, after just one aerobic session, showed dramatic boosts in BDNF. A 2018 review elaborates on these findings.
Eat right
Diet mediates dementia risk through less direct, but very effective, pathways, Dr. Baker said. Diets rich in vegetables, berries, nuts, fish, lean proteins, and healthy fats improves virtually all metabolic measures. These, in turn, reduce the risk cerebrovascular disease – an important driver of vascular dementias and a contributor to Alzheimer’s disease risk as well.
The MIND diet study (Mediterranean-DASH Diet Intervention for Neurodegenerative Delay), reported in 2015 was a very successful demonstration of this concept. A combination of the Mediterranean diet and the DASH diet (Dietary Approaches to Stop Hypertension), the MIND diet stresses frequent consumption of vegetables – especially leafy greens – as well as nuts, berries, whole grains, fish, poultry, and wine or grape juice. In the large, nearly 5-year study of 923 subjects aged 58-98 years, the MIND diet was associated with significant gains in cognition – equivalent to a 7-year reversal of age. After 4.5 years, those who strictly adhered to the diet had a 53% reduction in risk for Alzheimer’s disease, and those who adhered moderately had a 35% reduction. And in a more recent Australian longitudinal study, the MIND diet was associated with a 53% reduced risk of cognitive impairment over 12 years.
Ketogenic diets also may exert a benefit. Theoretically, a state of ketosis forces the brain to burn ketones as an alternative fuel to glucose, thus boosting brain function in glucose-starved brains. A small pilot study with exploratory cognitive endpoints determined that diet-compliant subjects with mild to moderate Alzheimer’s experienced a mean 5-point improvement in the Alzheimer’s Disease Assessment Score–Cognition. They reverted to baseline scores within a month of ending the study.
Recent initial work into the gut microbiome provides some additional speculative, but interesting, data. A dysregulated microbiome can shift microbial populations toward a more inflammatory profile. Some work suggests that inflammatory cytokines then travel to the brain and induce a hyperresponse of neuron-damaging immune cells. A comprehensive review article discusses the complicated mechanisms that may be in play.
Play with your friends
Cognitive stimulation and social interaction also appear to modify dementia risk, although the data are a little more limited. But personal interaction is a key element of Dr. Baker’s ongoing EXERT trial.
The ongoing phase 3 trial randomized 300 adults with amnestic mild cognitive impairment to moderate to high intensity aerobic exercise plus one-on-one support at local YMCA gyms or a low-intensity stretching, balance, and range of motion program. In additional to cognitive testing, the trial includes brain imaging, cerebrospinal fluid sampling for biomarkers of Alzheimer’s disease, and a sleep study.
A key component is personal interaction with a trainer. “They spend a lot of one-on-one time with each person,” Dr. Baker said. “For me, that’s the crucial ingredient – that personal touch. It’s what helps people move from Point A to Point B in their behaviors.”
Virtual cognitive stimulation is also a burgeoning area of dementia prevention research right now. Numerous studies are ongoing to test whether virtual reality or other computer-based games might keep the mind sharp or even improve cognition in people at risk.
The power of three
If one lifestyle change can reduce dementia risk, what happens when all three work together?
That’s the newest question, first successfully explored in the mid-2000s, with the FINGER study (Finnish Geriatric Intervention Study to Prevent Cognitive Impairment and Disability). In FINGER, the triad of exercise, personal support at the gym, and a modified Mediterranean diet reduced Alzheimer’s disease risk and improved cognition relative to the control group.
FINGER showed that the intervention was feasible and that it was associated with cognitive preservation and reduced Alzheimer’s disease risk in a group of at-risk subjects. The active group also had a 25% greater improvement on a neuropsychological test battery relative to the control group. They also performed 150% better in processing speed, 83% better in executive function, and 40% better in short-term memory. They showed no increased risk of cognitive decline relative to the control group, which experienced a 30% increase in risk, according to lead investigator Miia Kivipelto, PhD, of the Karolinska Institute, Stockholm.
So successful was FINGER that it launched a global consortium of related studies called World Wide FINGERS. Active in six countries now, including the United States, the studies aim to discover whether such combinations of lifestyle interventions are workable across countries and cultures. World Wide FINGERS is largely supported by the Alzheimer’s Association.
Global enthusiasm for lifestyle interventions
In recognition of the importance of lifestyle changes for dementia prevention, the World Health Organization recently published “Risk reduction of cognitive decline and dementia.” The document reviews many studies and makes recommendations regarding not only exercise, diet, and cognitive stimulation, but also smoking and alcohol.
Research interest in these areas is surging, Dr. Baker said. “The [U.S.] National Institute on Aging now has 29 ongoing trials. There’s a strong commitment to investigations into how lifestyle interventions could protect brain health as we get older. Certainly, many fit and healthy people do develop Alzheimer’s. But for some, it could be medicine.”
But no matter how compliant people are, lifestyle changes will never completely rid the world of Alzheimer’s and other dementias. The view of Dr. Baker – and most other Alzheimer’s researchers – is to employ lifestyle changes to reduce risk as much as possible and not to stop when cognitive problems do present.
“We need to understand how lifestyle interventions might work in combination with pharmaceuticals,” she said. “If we can support the health of the body and the health of the mind, lifestyle interventions can be the fertilizer that would help drug therapy have its maximum effect.”
EXPERT ANALYSIS FROM CTAD 2019
Positive functional results reported for aducanumab in a pooled, post hoc analysis
SAN DIEGO – Positive findings from a post hoc subanalysis of two unsuccessful studies represent “a major step forward in Alzheimer’s disease research” and could set the antiamyloid antibody up as a “foothold” in slowing disease progression, study investigators said at the Clinical Trials on Alzheimer’s Disease conference.
After full follow-up of 78 weeks, patients with mild Alzheimer’s disease (AD) who took the highest 10-mg/kg dose for a full 14 doses experienced up to a 53% slowing of functional decline on the Clinical Dementia Rating–Sum of Boxes (CDR-SB) in one study and a 48% slowing in the other study – relative to placebo – a result that might give them “an extra year or 2” of independence; they might perhaps retain the ability to drive and even stay employed, said Sharon Cohen, MD, a panelist at the meeting’s aducanumab presentation session and a clinical investigator in EMERGE, one of two phase 3 studies from which the data were derived.
Samantha Budd Haeberlein, PhD, Biogen’s vice president and head of late-stage clinical development in Alzheimer’s disease, presented the new data. They “are complex” and require much more study before investigators, clinicians, and federal regulators can fully embrace them, said the panelists who discussed the results. Nevertheless, Biogen, which is codeveloping the antibody with partner Eisai, said in October it will put aducanumab forward to the Food and Drug Administration in a new drug application for the first-ever AD disease-modifying agent. FDA regulators have said they will review the data.
The new subanalysis comprised 570 of 3,285 patients in two identical studies with negative primary endpoint results. One, ENGAGE, failed to reach both its primary and secondary endpoints; the other, EMERGE, was halted last spring after a futility analysis determined that aducanumab was unlikely to confer significant benefit. The post hoc subanalysis looked at a combined subset of those who received the highest 10-mg/kg dose for the full 78 weeks of each trial. The statistically significant functional endpoints occurred in this group, comprised largely of apolipoprotein E epsilon-4 (APOE4) allele carriers.
“The futility analysis of EMERGE was highly unfortunate,” said panelist Paul Aisen, MD, founding director of the Alzheimer’s Therapeutic Research Institute at the University of Southern California, Los Angeles. “Clearly in the final analysis, EMERGE was positive in the primary endpoints, and now the secondary analysis of both studies is positive and consistent.” The diverging trajectory of placebo and treatment groups continued to the end of follow-up in both studies, a finding that at least suggests continuing improvement, he added.
Biogen undertook the pooled analysis after ENGAGE’s futility analysis. Early in the development program, concern about amyloid-related imaging abnormalities (ARIA) in APOE4 carriers led Biogen to stratify doses in that group.
“When we started [creating aducanumab trials], we stratified the dose so that e4 carriers had the lowest dose, but in PRIME [the phase 1b study], we saw the best result from the 10-mg/kg dose, so we believed that was important for efficacy. However, we didn’t have sufficient evidence to believe that it was safe to put carriers on that dose. In EMERGE, we saw that carriers could safely take it until the end of the study.”
Since the trials were running almost synchronously, a new version of randomization ensued. This allowed more e4 carriers to go forward on the 10-mg/kg dose.
“I would not normally recommend changing dose in the middle of a phase 3 trial, but it did have a real impact in the high-dose group,” Dr. Haeberlein said. Additionally, by the time of data lock after the futility analysis, more patients had completed the entire 78 weeks at the 10-mg/kg dose. Cumulative dosing ended up being quite different in the APOE4 carriers after this new version ensued. Before, the median cumulative dose for both carriers and noncarriers was 116 mg/kg. After the change, the median cumulative dose was 153 mg/kg. And before the alteration, 21% in EMERGE and 15% in ENGAGE received the full 14 possible 10-mg/kg doses. After the change, 51% in EMERGE and 47% in ENGAGE received the full 14 doses of 10 mg/kg.
The pooled analysis comprised this combined group, which was then largely composed of APOE4 carriers.
Imaging confirmed such dose-driven reductions in both brain amyloid plaques and phosphorylated tau. Although amyloid reduction has never been tied to cognitive or functional benefits, tau reduction has been associated with nonsignificant cognitive benefits in prior studies.
In the primary analysis of ENGAGE, aducanumab conferred no cognitive or functional benefit. In EMERGE, there were significant cognitive improvements on both the Mini Mental State Exam score (an 18% slowing of decline relative to placebo) and the Alzheimer’s Disease Assessment Scale cognitive portion (a 27% slowing).
However, the functional improvements seen in the pooled post hoc data “are a big deal,” and probably more meaningful to patients and families than the memory improvements, Dr. Cohen said.
“Those of us who know this disease well know what it means to lose yourself slice by slice, and anything you can hang onto is a triumph,” said Dr. Cohen, medical director and principal investigator of the Toronto Memory Program, an independent medical facility for dementia care and research. “I am pleased with a 27% slowing of cognitive decline, but a 40% slowing of functional decline is what will be really meaningful to patients. This is a long, slow disease, and if we can slow it at all, we’re winning out.”
Safety endpoints, especially ARIA, were not unexpected considering past studies. ARIA occurred in 41% of patients treated with the high aducanumab dose in EMERGE and in 40% in ENGAGE. It was largely asymptomatic (80% in EMERGE and 71% in ENGAGE). Headache was the next most common adverse event, followed by dizziness, visual disturbance, and nausea and vomiting. ARIA generally resolved within 4-6 weeks, and most patients continued their 10-mg/kg dose.
Biogen intends to begin a new study, an open-label nonrandomized trial that will offer the 10-mg/kg dose to all patients in both trials, including those who took placebo. This may provide interesting data regarding redosing patients who were off their successful 10-mg/kg dose for an extended period of time, said Laurie Ryan, PhD, chief of the Dementias of Aging Branch in the Division of Neuroscience at the National Institute on Aging.
“If those in the high-dose group had a regression of their improvements and then improved again when restarted, that would certainly tell us something,” she said in an interview. Likewise, researchers will be carefully looking at any placebo group response. “But we have to remember that this will not be a randomized study,” and will bring with it all the issues that such a study typically carries.
“I agree it’s unfortunate that they had to stop the EMERGE trial,” she said. “It really did complicate the results, even though they are certainly trending in the right way. But we have had a number of post hoc analyses that show APOE4-positive benefiting, or e4-negative benefiting, and these haven’t panned out.”
SAN DIEGO – Positive findings from a post hoc subanalysis of two unsuccessful studies represent “a major step forward in Alzheimer’s disease research” and could set the antiamyloid antibody up as a “foothold” in slowing disease progression, study investigators said at the Clinical Trials on Alzheimer’s Disease conference.
After full follow-up of 78 weeks, patients with mild Alzheimer’s disease (AD) who took the highest 10-mg/kg dose for a full 14 doses experienced up to a 53% slowing of functional decline on the Clinical Dementia Rating–Sum of Boxes (CDR-SB) in one study and a 48% slowing in the other study – relative to placebo – a result that might give them “an extra year or 2” of independence; they might perhaps retain the ability to drive and even stay employed, said Sharon Cohen, MD, a panelist at the meeting’s aducanumab presentation session and a clinical investigator in EMERGE, one of two phase 3 studies from which the data were derived.
Samantha Budd Haeberlein, PhD, Biogen’s vice president and head of late-stage clinical development in Alzheimer’s disease, presented the new data. They “are complex” and require much more study before investigators, clinicians, and federal regulators can fully embrace them, said the panelists who discussed the results. Nevertheless, Biogen, which is codeveloping the antibody with partner Eisai, said in October it will put aducanumab forward to the Food and Drug Administration in a new drug application for the first-ever AD disease-modifying agent. FDA regulators have said they will review the data.
The new subanalysis comprised 570 of 3,285 patients in two identical studies with negative primary endpoint results. One, ENGAGE, failed to reach both its primary and secondary endpoints; the other, EMERGE, was halted last spring after a futility analysis determined that aducanumab was unlikely to confer significant benefit. The post hoc subanalysis looked at a combined subset of those who received the highest 10-mg/kg dose for the full 78 weeks of each trial. The statistically significant functional endpoints occurred in this group, comprised largely of apolipoprotein E epsilon-4 (APOE4) allele carriers.
“The futility analysis of EMERGE was highly unfortunate,” said panelist Paul Aisen, MD, founding director of the Alzheimer’s Therapeutic Research Institute at the University of Southern California, Los Angeles. “Clearly in the final analysis, EMERGE was positive in the primary endpoints, and now the secondary analysis of both studies is positive and consistent.” The diverging trajectory of placebo and treatment groups continued to the end of follow-up in both studies, a finding that at least suggests continuing improvement, he added.
Biogen undertook the pooled analysis after ENGAGE’s futility analysis. Early in the development program, concern about amyloid-related imaging abnormalities (ARIA) in APOE4 carriers led Biogen to stratify doses in that group.
“When we started [creating aducanumab trials], we stratified the dose so that e4 carriers had the lowest dose, but in PRIME [the phase 1b study], we saw the best result from the 10-mg/kg dose, so we believed that was important for efficacy. However, we didn’t have sufficient evidence to believe that it was safe to put carriers on that dose. In EMERGE, we saw that carriers could safely take it until the end of the study.”
Since the trials were running almost synchronously, a new version of randomization ensued. This allowed more e4 carriers to go forward on the 10-mg/kg dose.
“I would not normally recommend changing dose in the middle of a phase 3 trial, but it did have a real impact in the high-dose group,” Dr. Haeberlein said. Additionally, by the time of data lock after the futility analysis, more patients had completed the entire 78 weeks at the 10-mg/kg dose. Cumulative dosing ended up being quite different in the APOE4 carriers after this new version ensued. Before, the median cumulative dose for both carriers and noncarriers was 116 mg/kg. After the change, the median cumulative dose was 153 mg/kg. And before the alteration, 21% in EMERGE and 15% in ENGAGE received the full 14 possible 10-mg/kg doses. After the change, 51% in EMERGE and 47% in ENGAGE received the full 14 doses of 10 mg/kg.
The pooled analysis comprised this combined group, which was then largely composed of APOE4 carriers.
Imaging confirmed such dose-driven reductions in both brain amyloid plaques and phosphorylated tau. Although amyloid reduction has never been tied to cognitive or functional benefits, tau reduction has been associated with nonsignificant cognitive benefits in prior studies.
In the primary analysis of ENGAGE, aducanumab conferred no cognitive or functional benefit. In EMERGE, there were significant cognitive improvements on both the Mini Mental State Exam score (an 18% slowing of decline relative to placebo) and the Alzheimer’s Disease Assessment Scale cognitive portion (a 27% slowing).
However, the functional improvements seen in the pooled post hoc data “are a big deal,” and probably more meaningful to patients and families than the memory improvements, Dr. Cohen said.
“Those of us who know this disease well know what it means to lose yourself slice by slice, and anything you can hang onto is a triumph,” said Dr. Cohen, medical director and principal investigator of the Toronto Memory Program, an independent medical facility for dementia care and research. “I am pleased with a 27% slowing of cognitive decline, but a 40% slowing of functional decline is what will be really meaningful to patients. This is a long, slow disease, and if we can slow it at all, we’re winning out.”
Safety endpoints, especially ARIA, were not unexpected considering past studies. ARIA occurred in 41% of patients treated with the high aducanumab dose in EMERGE and in 40% in ENGAGE. It was largely asymptomatic (80% in EMERGE and 71% in ENGAGE). Headache was the next most common adverse event, followed by dizziness, visual disturbance, and nausea and vomiting. ARIA generally resolved within 4-6 weeks, and most patients continued their 10-mg/kg dose.
Biogen intends to begin a new study, an open-label nonrandomized trial that will offer the 10-mg/kg dose to all patients in both trials, including those who took placebo. This may provide interesting data regarding redosing patients who were off their successful 10-mg/kg dose for an extended period of time, said Laurie Ryan, PhD, chief of the Dementias of Aging Branch in the Division of Neuroscience at the National Institute on Aging.
“If those in the high-dose group had a regression of their improvements and then improved again when restarted, that would certainly tell us something,” she said in an interview. Likewise, researchers will be carefully looking at any placebo group response. “But we have to remember that this will not be a randomized study,” and will bring with it all the issues that such a study typically carries.
“I agree it’s unfortunate that they had to stop the EMERGE trial,” she said. “It really did complicate the results, even though they are certainly trending in the right way. But we have had a number of post hoc analyses that show APOE4-positive benefiting, or e4-negative benefiting, and these haven’t panned out.”
SAN DIEGO – Positive findings from a post hoc subanalysis of two unsuccessful studies represent “a major step forward in Alzheimer’s disease research” and could set the antiamyloid antibody up as a “foothold” in slowing disease progression, study investigators said at the Clinical Trials on Alzheimer’s Disease conference.
After full follow-up of 78 weeks, patients with mild Alzheimer’s disease (AD) who took the highest 10-mg/kg dose for a full 14 doses experienced up to a 53% slowing of functional decline on the Clinical Dementia Rating–Sum of Boxes (CDR-SB) in one study and a 48% slowing in the other study – relative to placebo – a result that might give them “an extra year or 2” of independence; they might perhaps retain the ability to drive and even stay employed, said Sharon Cohen, MD, a panelist at the meeting’s aducanumab presentation session and a clinical investigator in EMERGE, one of two phase 3 studies from which the data were derived.
Samantha Budd Haeberlein, PhD, Biogen’s vice president and head of late-stage clinical development in Alzheimer’s disease, presented the new data. They “are complex” and require much more study before investigators, clinicians, and federal regulators can fully embrace them, said the panelists who discussed the results. Nevertheless, Biogen, which is codeveloping the antibody with partner Eisai, said in October it will put aducanumab forward to the Food and Drug Administration in a new drug application for the first-ever AD disease-modifying agent. FDA regulators have said they will review the data.
The new subanalysis comprised 570 of 3,285 patients in two identical studies with negative primary endpoint results. One, ENGAGE, failed to reach both its primary and secondary endpoints; the other, EMERGE, was halted last spring after a futility analysis determined that aducanumab was unlikely to confer significant benefit. The post hoc subanalysis looked at a combined subset of those who received the highest 10-mg/kg dose for the full 78 weeks of each trial. The statistically significant functional endpoints occurred in this group, comprised largely of apolipoprotein E epsilon-4 (APOE4) allele carriers.
“The futility analysis of EMERGE was highly unfortunate,” said panelist Paul Aisen, MD, founding director of the Alzheimer’s Therapeutic Research Institute at the University of Southern California, Los Angeles. “Clearly in the final analysis, EMERGE was positive in the primary endpoints, and now the secondary analysis of both studies is positive and consistent.” The diverging trajectory of placebo and treatment groups continued to the end of follow-up in both studies, a finding that at least suggests continuing improvement, he added.
Biogen undertook the pooled analysis after ENGAGE’s futility analysis. Early in the development program, concern about amyloid-related imaging abnormalities (ARIA) in APOE4 carriers led Biogen to stratify doses in that group.
“When we started [creating aducanumab trials], we stratified the dose so that e4 carriers had the lowest dose, but in PRIME [the phase 1b study], we saw the best result from the 10-mg/kg dose, so we believed that was important for efficacy. However, we didn’t have sufficient evidence to believe that it was safe to put carriers on that dose. In EMERGE, we saw that carriers could safely take it until the end of the study.”
Since the trials were running almost synchronously, a new version of randomization ensued. This allowed more e4 carriers to go forward on the 10-mg/kg dose.
“I would not normally recommend changing dose in the middle of a phase 3 trial, but it did have a real impact in the high-dose group,” Dr. Haeberlein said. Additionally, by the time of data lock after the futility analysis, more patients had completed the entire 78 weeks at the 10-mg/kg dose. Cumulative dosing ended up being quite different in the APOE4 carriers after this new version ensued. Before, the median cumulative dose for both carriers and noncarriers was 116 mg/kg. After the change, the median cumulative dose was 153 mg/kg. And before the alteration, 21% in EMERGE and 15% in ENGAGE received the full 14 possible 10-mg/kg doses. After the change, 51% in EMERGE and 47% in ENGAGE received the full 14 doses of 10 mg/kg.
The pooled analysis comprised this combined group, which was then largely composed of APOE4 carriers.
Imaging confirmed such dose-driven reductions in both brain amyloid plaques and phosphorylated tau. Although amyloid reduction has never been tied to cognitive or functional benefits, tau reduction has been associated with nonsignificant cognitive benefits in prior studies.
In the primary analysis of ENGAGE, aducanumab conferred no cognitive or functional benefit. In EMERGE, there were significant cognitive improvements on both the Mini Mental State Exam score (an 18% slowing of decline relative to placebo) and the Alzheimer’s Disease Assessment Scale cognitive portion (a 27% slowing).
However, the functional improvements seen in the pooled post hoc data “are a big deal,” and probably more meaningful to patients and families than the memory improvements, Dr. Cohen said.
“Those of us who know this disease well know what it means to lose yourself slice by slice, and anything you can hang onto is a triumph,” said Dr. Cohen, medical director and principal investigator of the Toronto Memory Program, an independent medical facility for dementia care and research. “I am pleased with a 27% slowing of cognitive decline, but a 40% slowing of functional decline is what will be really meaningful to patients. This is a long, slow disease, and if we can slow it at all, we’re winning out.”
Safety endpoints, especially ARIA, were not unexpected considering past studies. ARIA occurred in 41% of patients treated with the high aducanumab dose in EMERGE and in 40% in ENGAGE. It was largely asymptomatic (80% in EMERGE and 71% in ENGAGE). Headache was the next most common adverse event, followed by dizziness, visual disturbance, and nausea and vomiting. ARIA generally resolved within 4-6 weeks, and most patients continued their 10-mg/kg dose.
Biogen intends to begin a new study, an open-label nonrandomized trial that will offer the 10-mg/kg dose to all patients in both trials, including those who took placebo. This may provide interesting data regarding redosing patients who were off their successful 10-mg/kg dose for an extended period of time, said Laurie Ryan, PhD, chief of the Dementias of Aging Branch in the Division of Neuroscience at the National Institute on Aging.
“If those in the high-dose group had a regression of their improvements and then improved again when restarted, that would certainly tell us something,” she said in an interview. Likewise, researchers will be carefully looking at any placebo group response. “But we have to remember that this will not be a randomized study,” and will bring with it all the issues that such a study typically carries.
“I agree it’s unfortunate that they had to stop the EMERGE trial,” she said. “It really did complicate the results, even though they are certainly trending in the right way. But we have had a number of post hoc analyses that show APOE4-positive benefiting, or e4-negative benefiting, and these haven’t panned out.”
REPORTING FROM CTAD 2019
Key clinical point: A pooled posthoc subanalysis of two unsuccessful phase 3 trials, found that the antiamyloid antibody aducanumab conferred significant functional benefits in patients with mild Alzheimer’s disease who took the highest 10-mg/kg dose for a full 78 weeks.
Major finding: Aducanumab conferred a 53% slowing of functional decline on the Clinical Dementia Rating–Sum of Boxes (CDR-SB) in one study, ENGAGE, and a 48% slowing in the other, EMERGE, relative to placebo.
Study details: The pooled group comprised 570 of 3,285 patients in the two identical ENGAGE and EMERGE studies.
Disclosures: Biogen and Eisai sponsored the studies and are codeveloping aducanumab.
Source: Budd SH et al. CTAD 2019, OC 1-4.
Understanding Principles of High Reliability Organizations Through the Eyes of VIONE, A Clinical Program to Improve Patient Safety by Deprescribing Potentially Inappropriate Medications and Reducing Polypharmacy
High reliability organizations (HROs) incorporate continuous process improvement through leadership commitment to create a safety culture that works toward creating a zero-harm environment.1 The Veterans Health Administration (VHA) has set transformational goals for becoming an HRO. In this article, we describe VIONE, an expanding medication deprescribing clinical program, which exemplifies the translation of HRO principles into health care system models. Both VIONE and HRO are globally relevant.
Reducing medication errors and related adverse drug events are important for achieving zero harm. Preventable medical errors rank behind heart disease and cancer as the third leading cause of death in the US.2 The simultaneous use of multiple medications can lead to dangerous drug interactions, adverse outcomes, and challenges with adherence. When a person is taking multiple medicines, known as polypharmacy, it is more likely that some are potentially inappropriate medications (PIM). Current literature highlights the prevalence and dangers of polypharmacy, which ranks among the top 10 common causes of death in the US, as well as suggestions to address preventable adverse outcomes from polypharmacy and PIM.3-5
Deprescribing of PIM frequently results in better disease management with improved health outcomes and quality of life.4 Many health care settings lack standardized approaches or set expectations to proactively deprescribe PIM. There has been insufficient emphasis on how to make decisions for deprescribing medications when therapeutic benefits are not clear and/or when the adverse effects may outweigh the therapeutic benefits.5
It is imperative to provide practice guidance for deprescribing nonessential medications along with systems-based infrastructure to enable integrated and effective assessments during opportune moments in the health care continuum. Multimodal approaches that include education, risk stratification, population health management interventions, research and resource allocation can help transform organizational culture in health care facilities toward HRO models of care, aiming at zero harm to patients.
The practical lessons learned from VIONE implementation science experiences on various scales and under diverse circumstances, cumulative wisdom from hindsight, foresight and critical insights gathered during nationwide spread of VIONE over the past 3 years continues to propel us toward the desirable direction and core concepts of an HRO.
The VIONE program facilitates practical, real-time interventions that could be tailored to various health care settings, organizational needs, and available resources. VIONE implements an electronic Computerized Patient Record System (CPRS) tool to enable planned cessation of nonessential medications that are potentially harmful, inappropriate, not indicated, or not necessary. The VIONE tool supports systematic, individualized assessment and adjustment through 5 filters (Figure 1). It prompts providers to assign 1 of these filters intuitively and objectively. VIONE combines clinical evidence for best practices, an interprofessional team approach, patient engagement, adapted use of existing medical records systems, and HRO principles for effective implementation.
As a tool to support safer prescribing practices, VIONE aligns closely with HRO principles (Table 1) and core pillars (Table 2).6-8 A zero-harm safety culture necessitates that medications be used for correct reasons, over a correct duration of time, and following a correct schedule while monitoring for adverse outcomes. However, reality generally falls significantly short of this for a myriad of reasons, such as compromised health literacy, functional limitations, affordability, communication gaps, patients seen by multiple providers, and an accumulation of prescriptions due to comorbidities, symptom progression, and management of adverse effects. Through a sharpened focus on both precision medicine and competent prescription management, VIONE is a viable opportunity for investing in the zero-harm philosophy that is integral to an HRO.
Design and Implementation
Initially launched in 2016 in a 15-bed inpatient, subacute rehabilitation unit within a VHA tertiary care facility, VIONE has been sustained and gradually expanded to 38 other VHA facility programs (Figure 2). Recognizing the potential value if adopted into widespread use, VIONE was a Gold Status winner in the VHA Under Secretary for Health Shark Tank-style competition in 2017 and was selected by the VHA Diffusion of Excellence as an innovation worthy of scale and spread through national dissemination.9 A toolkit for VIONE implementation, patient and provider brochures, VIONE vignette, and National Dialog template also have been created.10
Implementing VIONE in a new facility requires an actively engaged core team committed to patient safety and reduction of polypharmacy and PIM, interest and availability to lead project implementation strategies, along with meaningful local organizational support. The current structure for VIONE spread is as follows:
- Interested VHA participants review information and contact [email protected].
- The VIONE team orients implementing champions, mainly pharmacists, physicians, nurse practitioners, and physician assistants at a facility program level, offering guidance and available resources.
- Clinical Application Coordinators at Central Arkansas VA Healthcare System and participating facilities collaborate to add deprescribing menu options in CPRS and install the VIONE Polypharmacy Reminder Dialog template.
- Through close and ongoing collaborations, medical providers and clinical pharmacists proceed with deprescribing, aiming at planned cessation of nonessential and PIM, using the mnemonic prompt of VIONE. Vital and Important medications are continued and consolidated while a methodical plan is developed to deprescribe any medications that could lead to more harm than benefit and qualify based on the filters of Optional, Not indicated, and Every medicine has a diagnosis/reason. They select the proper discontinuation reasons in the CPRS medication menu (Figure 3) and document the rationale in the progress notes. It is highly encouraged that the collaborating pharmacists and health care providers add each other as cosigners and communicate effectively. Clinical pharmacy specialists also use the VIONE Polypharmacy Reminder Dialog Template (RDT) to document complete medication reviews with veterans to include deprescribing rationale and document shared decision making.
- A VIONE national dashboard captures deprescribing data in real time and automates reporting with daily updates that are readily accessible to all implementing facilities. Minimum data captured include the number of unique veterans impacted, number of medications deprescribed, cumulative cost avoidance to date, and number of prescriptions deprescribed per veteran. The dashboard facilitates real-time use of individual patient data and has also been designed to capture data from VHA administrative data portals and Corporate Data Warehouse.
Results
As of October 31, 2019, the assessment of polypharmacy using the VIONE tool across VHA sites has benefited > 60,000 unique veterans, of whom 49.2% were in urban areas, 47.7% in rural areas, and 3.1% in highly rural areas. Elderly male veterans comprised a clear majority. More than 128,000 medications have been deprescribed. The top classes of medications deprescribed are antihypertensives, over-the-counter medications, and antidiabetic medications. An annualized cost avoidance of > $4.0 million has been achieved. Cost avoidance is the cost of medications that otherwise would have continued to be filled and paid for by the VHA if they had not been deprescribed, projected for a maximum of 365 days. The calculation methodology can be summarized as follows:
The calculations reported in Table 3 and Figure 4 are conservative and include only chronic outpatient prescriptions and do not account for medications deprescribed in inpatient units, nursing home, community living centers, or domiciliary populations. Data tracked separately from inpatient and community living center patient populations indicated an additional 25,536 deprescribed medications, across 28 VA facilities, impacting 7,076 veterans with an average 2.15 medications deprescribed per veteran. The additional achieved cost avoidance was $370,272 (based on $14.50 average cost per prescription). Medications restarted within 30 days of deprescribing are not included in these calculations.
The cost avoidance calculation further excludes the effects of VIONE implementation on many other types of interventions. These interventions include, but are not limited to, changing from aggressive care to end of life, comfort care when strongly indicated; reduced emergency department visits or invasive diagnostic and therapeutic approaches, when not indicated; medical supplies, antimicrobial preparations; labor costs related to packaging, mailing, and administering prescriptions; reduced/prevented clinical waste; reduced decompensation of systemic illnesses and subsequent health care needs precipitated by iatrogenic disturbances and prolonged convalescence; and overall changes to prescribing practices through purposeful and targeted interactions with colleagues across various disciplines and various hierarchical levels.
Discussion
The VIONE clinical program exemplifies the translation of HRO principles into health care system practices. VIONE offers a systematic approach to improve medication management with an emphasis on deprescribing nonessential medications across various health care settings, facilitating VHA efforts toward zero harm. It demonstrates close alignment with the key building blocks of an HRO. Effective VIONE incorporation into an organizational culture reflects leadership commitment to safety and reliability in their vision and actions. By empowering staff to proactively reduce inappropriate medications and thereby prevent patient harm, VIONE contributes to enhancing an enterprise-wide culture of safety, with fewer errors and greater reliability. As a standardized decision support tool for the ongoing practice of assessment and planned cessation of potentially inappropriate medications, VIONE illustrates how continuous process improvement can be a part of staff-engaged, veteran-centered, highly reliable care. The standardization of the VIONE tool promotes achievement and sustainment of desired HRO principles and practices within health care delivery systems.
Conclusions
The VIONE program was launched not as a cost savings or research program but as a practical, real-time bedside or ambulatory care intervention to improve patient safety. Its value is reflected in the overwhelming response from scholarly and well-engaged colleagues expressing serious interests in expanding collaborations and tailoring efforts to add more depth and breadth to VIONE related efforts.
Acknowledgments
The authors express their gratitude to Central Arkansas VA Healthcare System leadership, Clinical Applications Coordinators, and colleagues for their unconditional support, to the Diffusion of Excellence programs at US Department of Veterans Affairs Central Office for their endorsement, and to the many VHA participants who renew our optimism and energy as we continue this exciting journey. We also thank Bridget B. Kelly for her assistance in writing and editing of the manuscript.
1. Chassin MR, Jerod ML. High-reliability health care: getting there from here. The Joint Commission. Milbank Q. 2013;91(3):459-490.
2. Makary MA, Daniel M. Medical error—the third leading cause of death in the US. BMJ. 2016;353:i2139.
3. Quinn KJ, Shah NH. A dataset quantifying polypharmacy in the United States. Sci Data. 2017;4:170167.
4. Scott IA, Hilmer SN, Reeve E, et al. Reducing inappropriate polypharmacy: the process of deprescribing. JAMA Intern Med. 2015;175(5):827-834.
5. Steinman MA. Polypharmacy—time to get beyond numbers. JAMA Intern Med. 2016;176(4):482-483.
6. US Department of Veterans Affairs. High reliability. https://dvagov.sharepoint.com/sites/OHT-PMO/high-reliability/Pages/default.aspx. [Nonpublic source, not verified.]
7. Gordon S, Mendenhall P, O’Connor BB. Beyond the Checklist: What Else Health Care Can Learn from Aviation Teamwork and Safety. Ithaca, NY: Cornell University Press; 2013.
8. Institute of Medicine (US) Committee on Quality of Health Care in America; Kohn LT, Corrigan JM, Donaldson MS, eds. To Err Is Human: Building a Safer Health System. Washington, DC: The National Academies Press; 2000.
9. US Department of Veterans Affairs. Diffusion of Excellence. https://www.va.gov/HEALTHCAREEXCELLENCE/diffusion-of-excellence/. Updated August 10, 2018. Accessed June 26, 2019.
10. US Department of Veterans Affairs. VIONE program toolkit. https://www.vapulse.net/docs/DOC-259375. [Nonpublic source, not verified.]
High reliability organizations (HROs) incorporate continuous process improvement through leadership commitment to create a safety culture that works toward creating a zero-harm environment.1 The Veterans Health Administration (VHA) has set transformational goals for becoming an HRO. In this article, we describe VIONE, an expanding medication deprescribing clinical program, which exemplifies the translation of HRO principles into health care system models. Both VIONE and HRO are globally relevant.
Reducing medication errors and related adverse drug events are important for achieving zero harm. Preventable medical errors rank behind heart disease and cancer as the third leading cause of death in the US.2 The simultaneous use of multiple medications can lead to dangerous drug interactions, adverse outcomes, and challenges with adherence. When a person is taking multiple medicines, known as polypharmacy, it is more likely that some are potentially inappropriate medications (PIM). Current literature highlights the prevalence and dangers of polypharmacy, which ranks among the top 10 common causes of death in the US, as well as suggestions to address preventable adverse outcomes from polypharmacy and PIM.3-5
Deprescribing of PIM frequently results in better disease management with improved health outcomes and quality of life.4 Many health care settings lack standardized approaches or set expectations to proactively deprescribe PIM. There has been insufficient emphasis on how to make decisions for deprescribing medications when therapeutic benefits are not clear and/or when the adverse effects may outweigh the therapeutic benefits.5
It is imperative to provide practice guidance for deprescribing nonessential medications along with systems-based infrastructure to enable integrated and effective assessments during opportune moments in the health care continuum. Multimodal approaches that include education, risk stratification, population health management interventions, research and resource allocation can help transform organizational culture in health care facilities toward HRO models of care, aiming at zero harm to patients.
The practical lessons learned from VIONE implementation science experiences on various scales and under diverse circumstances, cumulative wisdom from hindsight, foresight and critical insights gathered during nationwide spread of VIONE over the past 3 years continues to propel us toward the desirable direction and core concepts of an HRO.
The VIONE program facilitates practical, real-time interventions that could be tailored to various health care settings, organizational needs, and available resources. VIONE implements an electronic Computerized Patient Record System (CPRS) tool to enable planned cessation of nonessential medications that are potentially harmful, inappropriate, not indicated, or not necessary. The VIONE tool supports systematic, individualized assessment and adjustment through 5 filters (Figure 1). It prompts providers to assign 1 of these filters intuitively and objectively. VIONE combines clinical evidence for best practices, an interprofessional team approach, patient engagement, adapted use of existing medical records systems, and HRO principles for effective implementation.
As a tool to support safer prescribing practices, VIONE aligns closely with HRO principles (Table 1) and core pillars (Table 2).6-8 A zero-harm safety culture necessitates that medications be used for correct reasons, over a correct duration of time, and following a correct schedule while monitoring for adverse outcomes. However, reality generally falls significantly short of this for a myriad of reasons, such as compromised health literacy, functional limitations, affordability, communication gaps, patients seen by multiple providers, and an accumulation of prescriptions due to comorbidities, symptom progression, and management of adverse effects. Through a sharpened focus on both precision medicine and competent prescription management, VIONE is a viable opportunity for investing in the zero-harm philosophy that is integral to an HRO.
Design and Implementation
Initially launched in 2016 in a 15-bed inpatient, subacute rehabilitation unit within a VHA tertiary care facility, VIONE has been sustained and gradually expanded to 38 other VHA facility programs (Figure 2). Recognizing the potential value if adopted into widespread use, VIONE was a Gold Status winner in the VHA Under Secretary for Health Shark Tank-style competition in 2017 and was selected by the VHA Diffusion of Excellence as an innovation worthy of scale and spread through national dissemination.9 A toolkit for VIONE implementation, patient and provider brochures, VIONE vignette, and National Dialog template also have been created.10
Implementing VIONE in a new facility requires an actively engaged core team committed to patient safety and reduction of polypharmacy and PIM, interest and availability to lead project implementation strategies, along with meaningful local organizational support. The current structure for VIONE spread is as follows:
- Interested VHA participants review information and contact [email protected].
- The VIONE team orients implementing champions, mainly pharmacists, physicians, nurse practitioners, and physician assistants at a facility program level, offering guidance and available resources.
- Clinical Application Coordinators at Central Arkansas VA Healthcare System and participating facilities collaborate to add deprescribing menu options in CPRS and install the VIONE Polypharmacy Reminder Dialog template.
- Through close and ongoing collaborations, medical providers and clinical pharmacists proceed with deprescribing, aiming at planned cessation of nonessential and PIM, using the mnemonic prompt of VIONE. Vital and Important medications are continued and consolidated while a methodical plan is developed to deprescribe any medications that could lead to more harm than benefit and qualify based on the filters of Optional, Not indicated, and Every medicine has a diagnosis/reason. They select the proper discontinuation reasons in the CPRS medication menu (Figure 3) and document the rationale in the progress notes. It is highly encouraged that the collaborating pharmacists and health care providers add each other as cosigners and communicate effectively. Clinical pharmacy specialists also use the VIONE Polypharmacy Reminder Dialog Template (RDT) to document complete medication reviews with veterans to include deprescribing rationale and document shared decision making.
- A VIONE national dashboard captures deprescribing data in real time and automates reporting with daily updates that are readily accessible to all implementing facilities. Minimum data captured include the number of unique veterans impacted, number of medications deprescribed, cumulative cost avoidance to date, and number of prescriptions deprescribed per veteran. The dashboard facilitates real-time use of individual patient data and has also been designed to capture data from VHA administrative data portals and Corporate Data Warehouse.
Results
As of October 31, 2019, the assessment of polypharmacy using the VIONE tool across VHA sites has benefited > 60,000 unique veterans, of whom 49.2% were in urban areas, 47.7% in rural areas, and 3.1% in highly rural areas. Elderly male veterans comprised a clear majority. More than 128,000 medications have been deprescribed. The top classes of medications deprescribed are antihypertensives, over-the-counter medications, and antidiabetic medications. An annualized cost avoidance of > $4.0 million has been achieved. Cost avoidance is the cost of medications that otherwise would have continued to be filled and paid for by the VHA if they had not been deprescribed, projected for a maximum of 365 days. The calculation methodology can be summarized as follows:
The calculations reported in Table 3 and Figure 4 are conservative and include only chronic outpatient prescriptions and do not account for medications deprescribed in inpatient units, nursing home, community living centers, or domiciliary populations. Data tracked separately from inpatient and community living center patient populations indicated an additional 25,536 deprescribed medications, across 28 VA facilities, impacting 7,076 veterans with an average 2.15 medications deprescribed per veteran. The additional achieved cost avoidance was $370,272 (based on $14.50 average cost per prescription). Medications restarted within 30 days of deprescribing are not included in these calculations.
The cost avoidance calculation further excludes the effects of VIONE implementation on many other types of interventions. These interventions include, but are not limited to, changing from aggressive care to end of life, comfort care when strongly indicated; reduced emergency department visits or invasive diagnostic and therapeutic approaches, when not indicated; medical supplies, antimicrobial preparations; labor costs related to packaging, mailing, and administering prescriptions; reduced/prevented clinical waste; reduced decompensation of systemic illnesses and subsequent health care needs precipitated by iatrogenic disturbances and prolonged convalescence; and overall changes to prescribing practices through purposeful and targeted interactions with colleagues across various disciplines and various hierarchical levels.
Discussion
The VIONE clinical program exemplifies the translation of HRO principles into health care system practices. VIONE offers a systematic approach to improve medication management with an emphasis on deprescribing nonessential medications across various health care settings, facilitating VHA efforts toward zero harm. It demonstrates close alignment with the key building blocks of an HRO. Effective VIONE incorporation into an organizational culture reflects leadership commitment to safety and reliability in their vision and actions. By empowering staff to proactively reduce inappropriate medications and thereby prevent patient harm, VIONE contributes to enhancing an enterprise-wide culture of safety, with fewer errors and greater reliability. As a standardized decision support tool for the ongoing practice of assessment and planned cessation of potentially inappropriate medications, VIONE illustrates how continuous process improvement can be a part of staff-engaged, veteran-centered, highly reliable care. The standardization of the VIONE tool promotes achievement and sustainment of desired HRO principles and practices within health care delivery systems.
Conclusions
The VIONE program was launched not as a cost savings or research program but as a practical, real-time bedside or ambulatory care intervention to improve patient safety. Its value is reflected in the overwhelming response from scholarly and well-engaged colleagues expressing serious interests in expanding collaborations and tailoring efforts to add more depth and breadth to VIONE related efforts.
Acknowledgments
The authors express their gratitude to Central Arkansas VA Healthcare System leadership, Clinical Applications Coordinators, and colleagues for their unconditional support, to the Diffusion of Excellence programs at US Department of Veterans Affairs Central Office for their endorsement, and to the many VHA participants who renew our optimism and energy as we continue this exciting journey. We also thank Bridget B. Kelly for her assistance in writing and editing of the manuscript.
High reliability organizations (HROs) incorporate continuous process improvement through leadership commitment to create a safety culture that works toward creating a zero-harm environment.1 The Veterans Health Administration (VHA) has set transformational goals for becoming an HRO. In this article, we describe VIONE, an expanding medication deprescribing clinical program, which exemplifies the translation of HRO principles into health care system models. Both VIONE and HRO are globally relevant.
Reducing medication errors and related adverse drug events are important for achieving zero harm. Preventable medical errors rank behind heart disease and cancer as the third leading cause of death in the US.2 The simultaneous use of multiple medications can lead to dangerous drug interactions, adverse outcomes, and challenges with adherence. When a person is taking multiple medicines, known as polypharmacy, it is more likely that some are potentially inappropriate medications (PIM). Current literature highlights the prevalence and dangers of polypharmacy, which ranks among the top 10 common causes of death in the US, as well as suggestions to address preventable adverse outcomes from polypharmacy and PIM.3-5
Deprescribing of PIM frequently results in better disease management with improved health outcomes and quality of life.4 Many health care settings lack standardized approaches or set expectations to proactively deprescribe PIM. There has been insufficient emphasis on how to make decisions for deprescribing medications when therapeutic benefits are not clear and/or when the adverse effects may outweigh the therapeutic benefits.5
It is imperative to provide practice guidance for deprescribing nonessential medications along with systems-based infrastructure to enable integrated and effective assessments during opportune moments in the health care continuum. Multimodal approaches that include education, risk stratification, population health management interventions, research and resource allocation can help transform organizational culture in health care facilities toward HRO models of care, aiming at zero harm to patients.
The practical lessons learned from VIONE implementation science experiences on various scales and under diverse circumstances, cumulative wisdom from hindsight, foresight and critical insights gathered during nationwide spread of VIONE over the past 3 years continues to propel us toward the desirable direction and core concepts of an HRO.
The VIONE program facilitates practical, real-time interventions that could be tailored to various health care settings, organizational needs, and available resources. VIONE implements an electronic Computerized Patient Record System (CPRS) tool to enable planned cessation of nonessential medications that are potentially harmful, inappropriate, not indicated, or not necessary. The VIONE tool supports systematic, individualized assessment and adjustment through 5 filters (Figure 1). It prompts providers to assign 1 of these filters intuitively and objectively. VIONE combines clinical evidence for best practices, an interprofessional team approach, patient engagement, adapted use of existing medical records systems, and HRO principles for effective implementation.
As a tool to support safer prescribing practices, VIONE aligns closely with HRO principles (Table 1) and core pillars (Table 2).6-8 A zero-harm safety culture necessitates that medications be used for correct reasons, over a correct duration of time, and following a correct schedule while monitoring for adverse outcomes. However, reality generally falls significantly short of this for a myriad of reasons, such as compromised health literacy, functional limitations, affordability, communication gaps, patients seen by multiple providers, and an accumulation of prescriptions due to comorbidities, symptom progression, and management of adverse effects. Through a sharpened focus on both precision medicine and competent prescription management, VIONE is a viable opportunity for investing in the zero-harm philosophy that is integral to an HRO.
Design and Implementation
Initially launched in 2016 in a 15-bed inpatient, subacute rehabilitation unit within a VHA tertiary care facility, VIONE has been sustained and gradually expanded to 38 other VHA facility programs (Figure 2). Recognizing the potential value if adopted into widespread use, VIONE was a Gold Status winner in the VHA Under Secretary for Health Shark Tank-style competition in 2017 and was selected by the VHA Diffusion of Excellence as an innovation worthy of scale and spread through national dissemination.9 A toolkit for VIONE implementation, patient and provider brochures, VIONE vignette, and National Dialog template also have been created.10
Implementing VIONE in a new facility requires an actively engaged core team committed to patient safety and reduction of polypharmacy and PIM, interest and availability to lead project implementation strategies, along with meaningful local organizational support. The current structure for VIONE spread is as follows:
- Interested VHA participants review information and contact [email protected].
- The VIONE team orients implementing champions, mainly pharmacists, physicians, nurse practitioners, and physician assistants at a facility program level, offering guidance and available resources.
- Clinical Application Coordinators at Central Arkansas VA Healthcare System and participating facilities collaborate to add deprescribing menu options in CPRS and install the VIONE Polypharmacy Reminder Dialog template.
- Through close and ongoing collaborations, medical providers and clinical pharmacists proceed with deprescribing, aiming at planned cessation of nonessential and PIM, using the mnemonic prompt of VIONE. Vital and Important medications are continued and consolidated while a methodical plan is developed to deprescribe any medications that could lead to more harm than benefit and qualify based on the filters of Optional, Not indicated, and Every medicine has a diagnosis/reason. They select the proper discontinuation reasons in the CPRS medication menu (Figure 3) and document the rationale in the progress notes. It is highly encouraged that the collaborating pharmacists and health care providers add each other as cosigners and communicate effectively. Clinical pharmacy specialists also use the VIONE Polypharmacy Reminder Dialog Template (RDT) to document complete medication reviews with veterans to include deprescribing rationale and document shared decision making.
- A VIONE national dashboard captures deprescribing data in real time and automates reporting with daily updates that are readily accessible to all implementing facilities. Minimum data captured include the number of unique veterans impacted, number of medications deprescribed, cumulative cost avoidance to date, and number of prescriptions deprescribed per veteran. The dashboard facilitates real-time use of individual patient data and has also been designed to capture data from VHA administrative data portals and Corporate Data Warehouse.
Results
As of October 31, 2019, the assessment of polypharmacy using the VIONE tool across VHA sites has benefited > 60,000 unique veterans, of whom 49.2% were in urban areas, 47.7% in rural areas, and 3.1% in highly rural areas. Elderly male veterans comprised a clear majority. More than 128,000 medications have been deprescribed. The top classes of medications deprescribed are antihypertensives, over-the-counter medications, and antidiabetic medications. An annualized cost avoidance of > $4.0 million has been achieved. Cost avoidance is the cost of medications that otherwise would have continued to be filled and paid for by the VHA if they had not been deprescribed, projected for a maximum of 365 days. The calculation methodology can be summarized as follows:
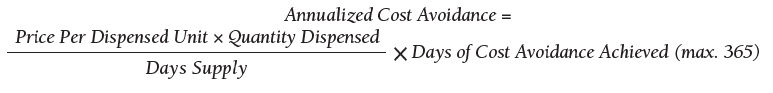
The calculations reported in Table 3 and Figure 4 are conservative and include only chronic outpatient prescriptions and do not account for medications deprescribed in inpatient units, nursing home, community living centers, or domiciliary populations. Data tracked separately from inpatient and community living center patient populations indicated an additional 25,536 deprescribed medications, across 28 VA facilities, impacting 7,076 veterans with an average 2.15 medications deprescribed per veteran. The additional achieved cost avoidance was $370,272 (based on $14.50 average cost per prescription). Medications restarted within 30 days of deprescribing are not included in these calculations.
The cost avoidance calculation further excludes the effects of VIONE implementation on many other types of interventions. These interventions include, but are not limited to, changing from aggressive care to end of life, comfort care when strongly indicated; reduced emergency department visits or invasive diagnostic and therapeutic approaches, when not indicated; medical supplies, antimicrobial preparations; labor costs related to packaging, mailing, and administering prescriptions; reduced/prevented clinical waste; reduced decompensation of systemic illnesses and subsequent health care needs precipitated by iatrogenic disturbances and prolonged convalescence; and overall changes to prescribing practices through purposeful and targeted interactions with colleagues across various disciplines and various hierarchical levels.
Discussion
The VIONE clinical program exemplifies the translation of HRO principles into health care system practices. VIONE offers a systematic approach to improve medication management with an emphasis on deprescribing nonessential medications across various health care settings, facilitating VHA efforts toward zero harm. It demonstrates close alignment with the key building blocks of an HRO. Effective VIONE incorporation into an organizational culture reflects leadership commitment to safety and reliability in their vision and actions. By empowering staff to proactively reduce inappropriate medications and thereby prevent patient harm, VIONE contributes to enhancing an enterprise-wide culture of safety, with fewer errors and greater reliability. As a standardized decision support tool for the ongoing practice of assessment and planned cessation of potentially inappropriate medications, VIONE illustrates how continuous process improvement can be a part of staff-engaged, veteran-centered, highly reliable care. The standardization of the VIONE tool promotes achievement and sustainment of desired HRO principles and practices within health care delivery systems.
Conclusions
The VIONE program was launched not as a cost savings or research program but as a practical, real-time bedside or ambulatory care intervention to improve patient safety. Its value is reflected in the overwhelming response from scholarly and well-engaged colleagues expressing serious interests in expanding collaborations and tailoring efforts to add more depth and breadth to VIONE related efforts.
Acknowledgments
The authors express their gratitude to Central Arkansas VA Healthcare System leadership, Clinical Applications Coordinators, and colleagues for their unconditional support, to the Diffusion of Excellence programs at US Department of Veterans Affairs Central Office for their endorsement, and to the many VHA participants who renew our optimism and energy as we continue this exciting journey. We also thank Bridget B. Kelly for her assistance in writing and editing of the manuscript.
1. Chassin MR, Jerod ML. High-reliability health care: getting there from here. The Joint Commission. Milbank Q. 2013;91(3):459-490.
2. Makary MA, Daniel M. Medical error—the third leading cause of death in the US. BMJ. 2016;353:i2139.
3. Quinn KJ, Shah NH. A dataset quantifying polypharmacy in the United States. Sci Data. 2017;4:170167.
4. Scott IA, Hilmer SN, Reeve E, et al. Reducing inappropriate polypharmacy: the process of deprescribing. JAMA Intern Med. 2015;175(5):827-834.
5. Steinman MA. Polypharmacy—time to get beyond numbers. JAMA Intern Med. 2016;176(4):482-483.
6. US Department of Veterans Affairs. High reliability. https://dvagov.sharepoint.com/sites/OHT-PMO/high-reliability/Pages/default.aspx. [Nonpublic source, not verified.]
7. Gordon S, Mendenhall P, O’Connor BB. Beyond the Checklist: What Else Health Care Can Learn from Aviation Teamwork and Safety. Ithaca, NY: Cornell University Press; 2013.
8. Institute of Medicine (US) Committee on Quality of Health Care in America; Kohn LT, Corrigan JM, Donaldson MS, eds. To Err Is Human: Building a Safer Health System. Washington, DC: The National Academies Press; 2000.
9. US Department of Veterans Affairs. Diffusion of Excellence. https://www.va.gov/HEALTHCAREEXCELLENCE/diffusion-of-excellence/. Updated August 10, 2018. Accessed June 26, 2019.
10. US Department of Veterans Affairs. VIONE program toolkit. https://www.vapulse.net/docs/DOC-259375. [Nonpublic source, not verified.]
1. Chassin MR, Jerod ML. High-reliability health care: getting there from here. The Joint Commission. Milbank Q. 2013;91(3):459-490.
2. Makary MA, Daniel M. Medical error—the third leading cause of death in the US. BMJ. 2016;353:i2139.
3. Quinn KJ, Shah NH. A dataset quantifying polypharmacy in the United States. Sci Data. 2017;4:170167.
4. Scott IA, Hilmer SN, Reeve E, et al. Reducing inappropriate polypharmacy: the process of deprescribing. JAMA Intern Med. 2015;175(5):827-834.
5. Steinman MA. Polypharmacy—time to get beyond numbers. JAMA Intern Med. 2016;176(4):482-483.
6. US Department of Veterans Affairs. High reliability. https://dvagov.sharepoint.com/sites/OHT-PMO/high-reliability/Pages/default.aspx. [Nonpublic source, not verified.]
7. Gordon S, Mendenhall P, O’Connor BB. Beyond the Checklist: What Else Health Care Can Learn from Aviation Teamwork and Safety. Ithaca, NY: Cornell University Press; 2013.
8. Institute of Medicine (US) Committee on Quality of Health Care in America; Kohn LT, Corrigan JM, Donaldson MS, eds. To Err Is Human: Building a Safer Health System. Washington, DC: The National Academies Press; 2000.
9. US Department of Veterans Affairs. Diffusion of Excellence. https://www.va.gov/HEALTHCAREEXCELLENCE/diffusion-of-excellence/. Updated August 10, 2018. Accessed June 26, 2019.
10. US Department of Veterans Affairs. VIONE program toolkit. https://www.vapulse.net/docs/DOC-259375. [Nonpublic source, not verified.]
Aging and Trauma: Post Traumatic Stress Disorder Among Korean War Veterans
The Korean War lasted from June 25, 1950 through July 27, 1953. Although many veterans of the Korean War experienced traumas during extremely stressful combat conditions. However, they would not have been diagnosed with posttraumatic stress disorder (PTSD) at the time because the latter did not exist as a formal diagnosis until the publication of the third edition of the Diagnostic and Statistical Manual (DSM) in 1980.1 Prior to 1980, psychiatric syndromes resulting from war and combat exposure where known by numerous other terms including shell shock, chronic traumatic war neurosis, and combat fatigue/combat exhaustion.2,3 Military psychiatrists attended to combat fatigue during the course of the Korean War, but as was true of World War I and II, the focus was on returning soldiers to duty. Combat fatigue was generally viewed as a transient condition.4-8
Although now octo- and nonagenarians, in 2019 there are 1.2 million living Korean War veterans in the US, representing 6.7% of all current veterans.9 Understanding their war experiences and the nature of their current and past presentation of PTSD is relevant not only in formal mental health settings, but in primary care settings, including home-based primary care, as well as community living centers, skilled nursing facilities and assisted living facilities. Older adults with PTSD often present with somatic concerns rather than spontaneously reporting mental health symptoms.10 Beyond the short-term clinical management of Korean War veterans with PTSD, consideration of their experiences also has long-term relevance for the appropriate treatment of other veteran cohorts as they age in coming decades.
The purpose of this article is to provide a clinically focused overview of PTSD in Korean War veterans, to help promote understanding of this often-forgotten group of veterans, and to foster optimized personalized care. This overview will include a description of the Korean War veteran population and the Korean War itself, the manifestations and identification of PTSD among Korean War veterans, and treatment approaches using evidence-based psychotherapies and pharmacotherapies. Finally, we provide recommendations for future research to address present empirical gaps in the understanding and treatment of Korean War veterans with PTSD.
Causes and Course of the Korean War
When working with Korean War veterans it is important to consider the special nature of that specific conflict. Space considerations limit our ability to do justice to the complex history and numerous battles of the Korean War, but information in the following summary was gleaned from several excellent histories.11-13
The Korean War has been referred to as The Forgotten War, a concern expressed even during the latter parts of the war.14,15 But the war and its veterans warrant remembering. The root and proximal causes of the Korean War are complex and not fully agreed upon by the main participants.16-19 In part this may reflect the fact that there was no clear victor in the Korean War, so that the different protagonists have developed their own versions of the history of the conflict. Also, US involvement and the public reaction to the war must be viewed within the larger historical context of that time. This context included the recent end of 4 years of US involvement in World War II (1941-1945) and the subsequent rapid rise of Cold War tensions between the US and the Soviet Union. The latter also included a worldwide fear of nuclear war and the US fear of the global spread of communism. These fears were fueled by the Soviet-led Berlin Blockade from June 1948 through May 1949, the Soviet Union’s successful atomic bomb test in August 1949, the founding of the People’s Republic of China in October 1949, and the February 1950 Sino-Soviet Treaty of Friendship and Alliance.13
In the closing days of World War II, the US and Soviet Union agreed to a temporary division of Korea along the 38th parallel to facilitate timely and efficient surrender of Japanese troops. But as Cold War tensions rose, the temporary division became permanent, and Soviet- and US-backed governments of the north and south, respectively, were officially established on the Korean peninsula in 1948. Although by 1949 the Soviets and US had withdrawn most troops from the peninsula, tensions between the north and south continued to mount and hostilities increased. To this day the exact causes of the eruption of war remain disputed, although it is clear that ideological as well as economic factors played a role, and both leaders of North and South Korea were pledging to reunite the peninsula under their respective leadership.16-19 The tension culminated on June 25, 1950, when North Korean troops crossed the 38th parallel and invaded South Korea. On June 27, 1950, President Truman ordered US naval and air forces to support South Korea and then ordered the involvement of ground troops on June 30.16,17,19
Although several other member countries of the United Nations (UN) provided troops, 90% of the troops were from the US. About 5.7 million US military personnel served during the war, including about 1.8 million in Korea itself. The US forces experienced approximately 34,000 battle-related deaths, 103,000 were wounded, and 7,000 were prisoners of war (POWs).11,20-22 The nature and events of the Korean War made it particularly stressful and traumatizing for the soldiers, sailors, and marines involved throughout its entire course. These included near defeat in the early months, a widely alternating war front along the north/south axis during the first year, and subsequently, not only intense constant battles on the fronts, but also a demanding and exhausting guerrilla war in the south, which lasted throughout the remainder of the conflict.11,15 The US troops during the initial months of the war have been described as outnumbered and underprepared, as many in the initial phase were reassigned from peace-time occupation duty in Japan.7
The first year of war was characterized by a repeated north-to-south/south-to-north shifts in control of territory. During the first 3 months, the North Korean forces overwhelmed the South and captured control of all but 2 South Korean cities in the far southeastern region (Pusan, now Busan; and Daegu), and US and UN forces were forced to retreat to the perimeter around Pusan. The intense Battle of Pusan Perimeter lasted from August 4, 1950 to September 18, 1950, and resulted in massive causalities as well as a flood of civilian refugees.
The course of the war began to change in early September 1950 with the landing of amphibious US/UN forces at Inchon, behind North Korean lines, which cut off southern supply routes for the North Korean troops.11 US/UN forces soon crossed to the north of the 38th parallel and captured the North Korean capital, Pyongyang, on October 19, 1950. They continued to push north and approached the Yalu River border with China by late November 1950, but then the Chinese introduced their own troops forcing a southward retreat of US/UN troops during which there were again numerous US/UN casualties. Chinese troops retook Seoul in late December 1950/early January 1951. However, the US/UN forces soon recaptured Seoul and advanced back to the 38th parallel. This back-and-forth across the 38th parallel continued until July 1951 when the front line of battle stabilized there. Although the line stabilized, intense battles and casualties continued for 2 more years. During this period US/UN troops also had to deal with guerrilla warfare behind the front lines due to the actions of communist partisans and isolated North Korean troops. This situation continued until the armistice was signed July 27, 1953.
Trauma and Characteristic Stresses of the War
There were many factors that made the Korean War experience different from previous wars, particularly World War II. For example, in contrast to the strong public support during and after World War II, public support for the Korean War in the US was low, particularly during its final year.23 In public opinion polls from October 1952 through April 1953, only 23% to 39% reported feeling that the war was worth fighting.23 A retrospective 1985 survey also found that 70% of World War II veterans, but only 33% of Korean War veterans reported feeling appreciated by the US public on their return from the war.24
Those fighting in the initial months of the war faced a particularly grim situation. According to LTC Philip Smith, who served as Division Psychiatrist on the Masan Front (Pusan Perimeter) during August and September of 1950, “Fighting was almost continuous and all available troops were on the fighting front… For the most part these soldiers were soft from occupation duty, many had not received adequate combat basic training, no refresher combat training in Korea had as yet been instituted,” he reported.7 “The extremes of climate coupled with the generally rugged mountainous terrain in Korea were physical factors of importance…These men were psychologically unprepared for the horrors and isolation of war.” LTC Smith noted that the change in status from civilian or occupation life to the marked deprivation of the war in Korea had been “too abrupt to allow as yet for a reasonable adjustment to the new setting” and that as a result “the highest rate of wounded and neuropsychiatric casualties in the Korean campaign resulted.”7
Even after this initial period, the nature of the shifting war, the challenging terrain, the high military casualty rate, and the high rate of civilian casualties and displacement continued throughout the war.
PTSD in Korean War Veterans
It is clear that Korean War combat veterans were exposed to traumatic events. It is unknown how many developed PTSD. While notions of psychological distress and disability related to combat trauma exposure have existed for centuries, Korean War and World War II veterans are a remaining link to pre-DSM PTSD mental health in the military. Military/forward psychiatry—psychiatric services near the battle zone rather than requiring evacuation of patients—was present in Korea from the early months of the war, but the focus of forward psychiatry was to reduce psychiatric causalities from combat fatigue and maximize rapid return-to-duty.4-6 With no real conception of PTSD, there were limited treatments available, and evidenced-based trauma-focused treatments for PTSD would not be introduced for at least another 4 decades.27-29
Skinner and Kaplick conducted a historical review of case descriptions of trauma-related conditions from World War I through the Vietnam War and noted the consistent inclusion of hyperarousal and intrusive symptoms, although there also was a greater emphasis on somatic conversion or hysteria symptoms in the earlier descriptions.30 By the Korean War, descriptions of combat fatigue included a number of symptoms that overlap with PTSD, including preoccupation with the traumatic stressor, nightmares, irritability/anger, increased startle, and hyperarousal.31 But following the acute phases, attention to any chronic problems associated with these conditions waned. As was acknowledged by a military psychiatrist in a 1954 talk, studies of the long-term adjustment of those who had “broken down in combat” were sorely needed.6 In a small 1965 study reported by Archibald and Tuddenham, persistent symptoms of combat fatigue among Korean War veterans were definitely present, and there was even a suggestion that the symptoms had increased over the decade since the war.32
Given the stoicism that typified cultural expectations for military men during this period, Korean War veterans may also have been reluctant to seek mental health treatment either at the time or later. In short, it is likely that a nontrivial proportion of Korean War veterans with PTSD were underdiagnosed and received suboptimal or no mental health treatment for decades following their war experiences.33 Although the nature of the war, deployment, and public support were distinct in World War II vs the Korean War, the absence of attention to the long-term effects of disorders related to combat trauma and the cultural expectations for stoicism suggest that PTSD among aging World War II veterans may also have gone underrecognized and undertreated.
Apart from the lack of interest in chronic effects of stressors, another problem that has plagued the limited empirical research on Korean War veterans has been the propensity to combine Korean War with World War II veteran samples in studies. Because World War II veterans have outnumbered Korean War veterans until recently, combined samples tended to have relatively few Korean War veterans. Nevertheless, from those studies that have been reported in which 2 groups were compared, important differences have been revealed. Specifically, although precise estimates of the prevalence of PTSD among Korean War combat veterans have varied depending on sampling and method, studies from the 1990s and early 2000s suggested that the prevalence of PTSD and other mental health concerns as well as the severity of symptoms, suicide risk, and psychosocial adjustment difficulties were worse among Korean War combat veterans relative to those among World War II combat veterans; however, both groups had lower prevalence than did Vietnam War combat veterans.21,34-37 Several authors speculated that these differences in outcome were at least partially due to differences in public support for the respective wars.36,37
Although there has been a paucity of research on psychiatric issues and PTSD in Korean War veterans, POWs who were very likely to have been exposed to extreme psychological traumas have received some attention. There have been comparisons of mortality and morbidity among POWs from the Korean War (PWK), World War II Pacific Theater (PWJ), and Europe (PWE).38 Among measures that were administered to the former POWs, the overall pattern seen from survey data in the mid-1960s revealed significantly worse health and functioning among the PWK and PWJ groups relative to the PWE group, with psychiatric difficulties being the most commonly reported impairments among the former 2 groups. This pattern was found most strongly with regards to objective measures, such as hospitalizations for “psychoneuroses,” and US Department of Veterans Affairs (VA) disability records, as well as based on self-reported psychosocial/recreational difficulties measured using the Cornell Medical Index (CMI).38
Gold and colleagues reported a follow-up study of more than 700 former POWs who were reinterviewed between 1989 and 1992.39 Although there was no scale of PTSD symptoms prior to formulation of the diagnosis in 1980, the CMI was a self-reported checklist that included a large range of both medical as well as behavioral and psychiatric symptoms. Thus, using CMI survey responses from 1965, the authors examined the factor structure (ie, the correlational relationships between multiple scale items and subgroupings of items) of the CMI relative to diagnosis of PTSD in 1989 to 1992 based on results from the Structured Clinical Interview for the DSM-III-R (SCID). The intent was to help discern whether the component domains of PTSD were present and intercorrelated in a pattern similar to that of the contemporary diagnosis. The investigators examined the factor structure of 20 psychological items from the CMI that appeared relevant to PTSD criteria using the 1965 data. Three factors (subgroups of highly intercorrelated items) were found: irritability (31% of variance), fearfulness/anxiousness (9% of the variance), and social withdrawal (7% of the variance). Although these did not directly correspond to, or fully cover, DSM PTSD domains or criteria, there does appear to be a thematic resemblance of the CMI findings with PTSD, including alterations in arousal and mood, vigilance, and startle.
Identification and Treatment of PTSD in Older Veterans
Of the 1.2 million living Korean War veterans in the US, 36.3% use VA provided health care.40 There are a number of complicating factors to consider in the current identification and treatment of PTSD in this cohort, including their advanced age; physical, cognitive, and social changes associated with normal aging; the associated medical and cognitive comorbidities; and the specific social-contextual factors in that age cohort. Any combination of these factors may complicate recognition, diagnosis, and treatment. It is also important to be cognizant of the additional stressors that may have been experienced by ethnic minorites and women serving in Korea, which are poorly documented and studied. Racial integration of the US military began during the Korean War, but the general pattern was for African American soldiers to be assigned to all-white units, rather than the reverse.14,41,42 And although the majority of military personnel serving in Korea were male, there were women serving in health care positions at mobile army surgical hospital (MASH) units, medical air evacuation (Medevac) aircraft, and off-shore hospital ships.
The clinical presentation of PTSD in older adults has varied, which may partially relate to the time elapsed since the index trauma. For example, older veterans in general may show less avoidance behavior as a part of PTSD, but in those who experience trauma later in life there may actually be greater avoidance.43,44 There have also been discrepant reports of intrusion or reexperiencing of symptoms, with these also potentially reduced in older veterans.43,44 However, sleep disturbances seem to be very common among elderly combat veterans, and attention should be paid to the possible presence of sleep apnea, which may be more common in veterans with PTSD in general.43,45,46
PTSD symptoms may reemerge after decades of remission or quiescence during retirement and/or with the emergence of neurocognitive impairment, such as Alzheimer disease or dementia. These individuals may have more difficulty engaging in distracting activities and work and spend more time engaging in reminiscence about the past, which can include increased focus on traumatic memories.45,47 Davison and colleagues have suggested a concept they call later-adulthood trauma reengagement (LATR) where later in life combat veterans may “confront and rework their wartime memories in an effort to find meaning and build coherence.”48 This process can be a double-edged sword, leading at times positively to enhanced personal growth or negatively to increased symptoms; preventive interventions may be able foster a more positive outcome.48
There is some evidence supporting the validity of the Clinician Administered PTSD Scale (CAPS) for the evaluation of PTSD in older adults, although this was based on the DSM-III-revised criteria for PTSD and an earlier version of CAPS.49 Bhattarai and colleagues examined responses to the 35-item Mississippi Scale for Combat-Related PTSD (M-PTSD) using VA clinical data collected between 2008 and 2015 on veterans of each combat era from World War II through the post-9/11.50 Strong internal consistency and test-retest reliability of the M-PTSD was observed within each veteran era sample. However, using chart diagnosis of PTSD as the criterion standard, the cut-scores for optimal balance of sensitivity and specificity of the M-PTSD scores were substantially lower for the older cohorts (World War II and Korean War veterans) relative to those for Vietnam and more recent veteran cohorts. The authors concluded that M-PTSD can be validly used to screen for PTSD in veterans within each of these cohorts but recommended using lower than standard cut-scores for Korean War and World War II veterans.50
This is also consistent with reports that suggest the use of lower cut-scores on self-administered PTSD symptom screens.43,44 For the clinician interested in quantifying the severity of PTSD, the most recent tools available are the CAPS-5 and the PCL-5, which have both been created in accordance with the DSM-5. The CAPS-5 is a rater-administered tool, and the PCL-5 is self-administered by the veteran. Although there has been little research using these newer tools in geriatric populations, they can currently serve as a means of tracking the severity of PTSD while we await measures that are better validated in Korean War and other older veterans.
Beyond specific empirical guidance, VA clinicians must presently rely on clinical observations and experience. Patients from the Korean War cohort often present at the insistence of a family member for changes in sleep, mood, behavior, or cognition. When the veterans themselves present, older adults with PTSD often focus more on somatic concerns (including pain, sleep, and gastrointestinal disturbance) than psychiatric problems per se. The latter tendency may in part be due to the salience of such symptoms for them, but perhaps also due to considerable stigma of mental health care that is still largely present in this group.43,44
Psychotherapy
Current VA treatment guidelines recommend trauma-focused therapies, with the strongest evidence base for prolonged exposure (PE), cognitive processing therapy (CPT), and eye movement desensitization and reprocessing (EMDR) therapies.51
There have been several excellent prior reviews discussing treatment of PTSD in older adults generally.10,43,44,52 These reviews have invariably expressed concern about the lack of sufficient empirical studies, but based on evidence from studies and case reports, there seems to be tentative support that trauma-focused therapies are acceptable and efficacious for use with older adults with PTSD. In their recent scoping review, Pless Kaiser and colleagues made several recommendations for trauma-focused therapy with older adults, including slow/careful pacing and use of compensatory aids for cognitive and sensory deficits.44 When cognitive impairment has exacerbated PTSD symptoms, they suggest therapists consider using an adapted form of CPT completed without a trauma narrative. For PE they recommend extending content across sessions and involving spouse or caregivers to assist with in vivo exposure and homework completion.44
Recent studies suggest that PTSD may be a risk factor for the later development of neurodegenerative disorders, and it is often during assessments for dementia that a revelation of PTSD occurs.10,43,47,55 Cognitive impairment may also be of relevance in deciding on the type of psychotherapy to be implemented, as it may have more adverse effects on the effectiveness of CPT than of exposure-based treatments (PE or EMDR). It may be useful to perform a cognitive assessment prior to initiation of a cognitive-based therapy, although extensive cognitive testing may not be practical or may be contraindicated because of fatigue. A brief screening tool such as the Montreal Cognitive Assessment or the Mini-Mental State Examinationmay be helpful.56, 57
Prolonged exposure has been reported by many clinicians to be effective in older adults with PTSD; however, due consideration should be given to the needs of individuals, as many have functioned for decades by suppressing memories.
Apart from the treatment needs for specific PTSD symptoms, the decades-long effects of poor sleep, irritability, hypervigilance, and dissociation also have social consequences for patients, including marital discord and divorce, and social and family isolation that should be addressed in therapy when appropriate. In addition, many Korean War veterans, like all veterans, sought postmilitary employment in professions that are associated with higher rates of exposure to psychological trauma, such as police or fire departments, and this may have an exacerbating effect on PTSD.58
Pharmacotherapy
There is very little empirical evidence guiding pharmacologic approaches to PTSD in older veterans. This population is at increased risk for many comorbidities, and pharmacologic treatments many require dosage adjustments, as is the case for any geriatric patient. Selective serotonin reuptake inhibitor (SSRI) and serotonin norepinephrine reuptake inhibitor (SNRI) medications have been proposed for some cases of PTSD.59,60 Health care providers may consider the SSRIs escitalopram or sertraline preferentially given their decreased potential for drug-drug interactions, anticholinergic effects, or cardiac toxicity compared with that of other drugs in this class.60,61 As venlafaxine can increase blood pressure, especially at higher doses, prescribers may choose duloxetine as an alternative if a SNRI is indicated.60 For veterans when prazosin is being considered for nightmare control, monitoring for hypotension, orthostasis, and the administration of other antihypertensives or prostatic hypertrophy medications is necessary.61 The use of benzodiazepines, while not recommended for PTSD, should be viewed with even greater trepidation in a geriatric population given enhanced risk of falls and confusion in the geriatric veteran population.60,62
Conclusions
Many of the oldest veterans (aged > 80 years) are from the Korean War era. The harsh and unique nature of the war, as well as the differences in context and support from the US public, and the outcome of the war, may have all contributed to and elevation of “combat fatigue” and PTSD among combat veterans from the Korean War. As the “forgotten war” cohort also has been forgotten by researchers, relatively little is known about posttraumatic stress sequelae of these veterans in the decades following the war.
From available evidence, we can readily surmise that problems were underrecognized and suboptimally diagnosed and treated. There is tentative evidence supporting the use of standard interviews and rating scales, such as the CAPS, M-PTSD, and PCL, but lower cut-scores than applied with Vietnam and later veteran cohorts are generally recommended to avoid excessive false negative errors. In terms of psychotherapy treatment, there is again a stark paucity of systematic research, but the limited evidence from studies of PTSD treatment in older adults from the general population tentatively support the acceptability and potential efficacy of recognized evidence-based trauma-focused psychotherapies for PTSD. Research on medication treatment is similarly lacking, but the general recommendations for the use of SSRI or SNRI medications seem to be valid, at least in our clinical experience, and the general rules for geriatric psychopharmacology definitely apply here—start low, go slow.
There are several important avenues for future research. Most pressing among these are establishing the effectiveness of existing treatments, and the modifications that may be needed in the broader context of the above factors, as well as the physical and cognitive changes associated with advanced age. Further research on the phenomenologic aspects of PTSD among Korean War and subsequent cohorts are also needed, as the information obtained will not only guide more effective personalized treatment of the Korean War veterans who remain with us, but also inform future generations of care in terms of the degree and dimensions of variability that may present between cohorts and within cohorts over the life span.
1. American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders, 3rd ed. Arlington VA: American Psychiatric Association; 1980.
2. Friedman MJ, Schnurr PP, McDonagh-Coyle A. Posttraumatic stress disorder in the military veteran. Psychiatr Clin North Am. 1994;17(2):265-277.
3. Salmon TW. The Care and Treatment of Mental Diseases and War Neuroses (“Shell Shock”) in the British Army. New York: War Work Committee of the National Committee for Mental Hygiene, Inc; 1917.
4. Jones E, Wessely S. “Forward psychiatry” in the military: its origins and effectiveness. J Trauma Stress. 2003;16(4):411-419.
5. Newman RA. Combat fatigue: a review to the Korean conflict. Mil Med. 1964;129:921-928.
6. Harris FG. Some comments on the differential diagnosis and treatment of psychiatric breakdowns in Korea. https://history.amedd.army.mil/booksdocs/korea/recad2/ch9-2.html. Published April 30, 1954. Accessed November 8, 2019.
7. Smith PB. Psychiatric experiences during the Korean conflict. Am Pract Dig Treat. 1955;6(2):183-189.
8. Koontz AR. Psychiatry in the Korean War. Military Surg.
1950;107(6):444-445.
9. US Department of Veterans Affairs. National Center for Veterans Analysis and Statistics. Population Tables - Table 2L: VETPOP2016 Living Veterans by period of service, gender, 2015-2045. https://www.va.gov/vetdata/docs/Demographics/New_Vetpop_Model/2L_VetPop2016
_POS_National.xlsx. Accessed November 8, 2019.
10. Cook JM, McCarthy E, Thorp SR. Older adults with PTSD: brief state of research and evidence-based psychotherapy case illustration. Am J Geriatr Psychiatry. 2017;25(5):522-530.
11. Millett AR. Korean War: 1950-1953. Encylopaedia Britannica. https://www.britannica.com/event/Korean-War#accordion-article-history. Updated Nov 7, 2019. Accessed November 8, 2019.
12. Stack L. Korean War, a ‘forgotten’ conflict that shaped the modern world. The New York Times. January 2, 2018. https://www.nytimes.com/2018/01/01/world/asia/korean-war-history.html. Accessed November 8, 2019.
13. Westad OA. The Cold War: A World History. New York: Basic Books; 2018.
14. Young C, Conard PL, Armstrong ML, Lacy D. Older military veteran care: many still believe they are forgotten. J Holist Nurs. 2018;36(3):291-300.
15. Huebner AJ. Kilroy is back, 1950-1953. In: The Warrior Image: Soldiers in American Culture From the Second World War to the Vietnam Era. Chapel Hill, NC: The University of North Carolina Press; 2008:97-131.
16. The annexation of Korea (editorial). Japan Times. https://www.japantimes.co.jp/opinion/2010/08/29/editorials/the-annexation-of-korea/#.XPgvJvlKhhE. Published August 29, 2010. Accessed November 8, 2019.
17. Gupta K. How did the Korean war begin? China Q. 1972;52:699-716.
18. Lin L, Zhao Y, Ogawa M, Hoge J, Kim BY. Whose history? An analysis of the Korean War in history textbooks from the United States, South Korea, Japan, and China. Social Studies. 2009;100(5):222-232.
19. Weathersby K. The Korean War revisited. Wilson Q. 1999;23(3):91.
20. US Department of Veterans Affairs, Office of Program and Data Analyses, Assistant Secretary for Planning and Analysis. Data on veterans of the Korean War. https://www.va.gov/vetdata/docs/SpecialReports/KW2000.pdf. Published June 2000. Accessed November 8, 2019.
21. Brooks MS, Fulton L. Evidence of poorer life-course mental health outcomes among veterans of the Korean War cohort. Aging Ment Health. 2010;14(2):177-183.
22. US Department of Veterans Affairs, Office of Public Affairs. America’s wars. https://www.va.gov/opa/publications/factsheets/fs_americas_wars.pdf. Accessed November 8, 2019.
23. Memorandum on recent polls on Korea. https://www.eisenhowerlibrary.gov/sites/default/files/research/online-documents/korean-war/public-opinion-1953-06-02.pdf. Published June 2, 1953. Accessed November 8, 2019.
24. Elder GH Jr, Clipp EC. Combat experience and emotional health: impairment and resilience in later life. J Pers. 1989;57(2):311-341.
25. US Department of Veterans Affairs. Public health: cold injuries. https://www.publichealth.va.gov/PUBLICHEALTH/exposures/cold-injuries/index.asp. Updated July 31, 2019. Accessed November 8, 2019.
26. US Department of Veterans Affairs. Korean War veterans health issues. https://www.va.gov/health-care/health-needs-conditions/health-issues-related-to-service-era
/korean-war/. Updated June 14, 2019. Accessed November 8, 2019.
27. Shapiro F. Efficacy of the eye movement desensitization procedure in the treatment of traumatic memories. J Trauma Stress. 1989;2(2):199-223.
28. Resick PA, Schnicke MK. Cognitive processing therapy for sexual assault victims. J Consul Clin Psychol. 1992;60(5):748-756.
29. Foa EB, Rothbaum BO. Treating Trauma of Rape: Cognitive-Behavioral Therapy for PTSD. New York: Guilford; 2001.
30. Skinner R, Kaplick PM. Cultural shift in mental illness: a comparison of stress responses in World War I and the Vietnam War. JRSM Open. 2017;8(12):2054270417746061.
31. Kardiner A, Spiegel H. War Stress and Neurotic Illness. New York: Hoeber; 1947.
32. Archibald HC, Tuddenham RD. Persistent stress reaction after combat: a 20-year follow-up. Arch Gen Psychiatry. 1965;12:475-481.
33. Cook JM, Simiola V. Trauma and aging. Curr Psychiatry Rep. 2018;20(10):93.
34. Rosenheck R, Fontana A. Long-term sequelae of combat in World War II, Korea and Vietnam: a comparative study. In: McCaughey BG, Fullerton CS, Ursano RJ, eds. Individual
and Community Responses to Trauma and Disaster: The Structure of Human Chaos. New York: Cambridge University Press; 1994:330-359.
35. Blake DD, Keane TM, Wine PR, Mora C, Taylor KL, Lyons JA. Prevalence of PTSD symptoms in combat veterans seeking medical treatment. J Trauma Stress. 1990;3(1):15-27.
36. McCranie EW, Hyer LA. Posttraumatic stress disorder symptoms in Korean conflict and World War II combat veterans seeking outpatient treatment. J Trauma Stress. 2000;13(3):427-439.
37. Fontana A, Rosenheck R. Traumatic war stressors and psychiatric symptoms among World War II, Korean, and Vietnam War veterans. Psychology Aging. 1994;9(1):27-33.
38. Beebe GW. Follow-up studies of World War II and Korean war prisoners. II. Morbidity, disability, and maladjustments. Am J Epidemiol. 1975;101(5):400-422.
39. Gold PB, Engdahl BE, Eberly RE, Blake RJ, Page WF, Frueh BC. Trauma exposure, resilience, social support, and PTSD construct validity among former prisoners of war. Social Psychiatry Psychiatr Epidemiol. 2000;35(1):36-42.
40. US Department of Veterans Affairs. Key statistics by veteran status and period of service. https://www.va.gov/vetdata/docs/SpecialReports/KeyStats.pdf. Accessed November 11, 2019.
41. Bowers WT, Hammond WM, MacGarrigle GL. Black Soldier, White Army. Washington DC: US Army Center of Military History; 1996.
42. Black HK. Three generations, three wars: African American veterans. Gerontologist. 2016;56(1):33-41.
43. Thorp SR, Sones HM, Cook JM. Posttraumatic stress disorder among older adults. In: Sorocco KH, Lauderdale S, eds. Cognitive Behavior Therapy With Older Adults: Innovations Across Care Settings. New York: Springer; 2011:189-217.
44. Pless Kaiser A, Cook JM, Glick DM, Moye J. Posttraumatic stress disorder in older adults: a conceptual review. Clinical Gerontol. 2019;42(4):359-376.
45. Sadavoy J. Survivors. A review of the late-life effects of prior psychological trauma. Am J Geriatr Psychiatry. 1997;5(4):287-301.
46. Tamanna S, Parker JD, Lyons J, Ullah MI. The effect of continuous positive air pressure (CPAP) on nightmares in patients with posttraumatic stress disorder (PTSD) and obstructive sleep apnea (OSA). J Clin Sleep Med. 2014;10(6):631-636.
47. Mota N, Tsai J, Kirwin PD, et al. Late-life exacerbation of PTSD symptoms in US veterans: results from the National Health and Resilience in Veterans Study. J Clin Psychiatry. 2016;77(3):348-354.
48. Davison EH, Kaiser AP, Spiro A 3rd, Moye J, King LA, King DW. From Late-onset stress symptomatology to later-adulthood trauma reengagement in aging combat veterans: taking a broader view. Gerontologist. 2016;56(1):14-21.
49. Hyer L, Summers MN, Boyd S, Litaker M, Boudewyns P. Assessment of older combat veterans with the clinician-administered PTSD scale. J Trauma Stress. 1996;9(3):587-593.
50. Bhattarai JJ, Oehlert ME, Weber DK. Psychometric properties of the Mississippi Scale for combat-related posttraumatic stress disorder based on veterans’ period of service. Psychol Serv. 2018. [Epub ahead of print]
51. US Department of Veterans Affairs, US Department of Defense. VA/DOD Clinical practice guideline for the management of posttraumatic stress disorder and acute stress disorder. Version 3.0. https://www.healthquality.va.gov/guidelines/MH/ptsd/VADoDPTSDCPGFinal012418.pdf.
Updated 2017. Accessed November 11, 2019.
52. Dinnen S, Simiola V, Cook JM. Post-traumatic stress disorder in older adults: a systematic review of the psychotherapy treatment literature. Aging Ment Health. 2015;19(2):144-150.
53. Jakel RJ. Posttraumatic Stress Disorder in the Elderly. Psychiatr Clin North Am. 2018;41(1):165-175.
54. Thorp SR, Glassman LH, Wells SY, et al. A randomized controlled trial of prolonged exposure therapy versus relaxation training for older veterans with military-related PTSD. J Anxiety Disord. 2019;64:45-54.
55. Kang B, Xu H, McConnell ES. Neurocognitive and psychiatric comorbidities of posttraumatic stress disorder among older veterans: a systematic review. Int J Geriatr Psychiatry. 2019;34(4):522-538.
56. Nasreddine ZS, Phillips NA, Bédirian V, et al. The Montreal Cognitive Assessment, MoCA: a brief screening tool for mild cognitive impairment. J Am Geriatr Soc. 2005;53(4):695-699.
57. Folstein MF, Folstein SE, McHugh PR. “Mini-mental state”. A practical method for grading the cognitive state of patients for the clinician. J Psychiatr Res. 1975;12(3):189-198.
58. Paton D. Traumatic Stress in Police Officers a Career-Length Assessment From Recruitment to Retirement. Springfield, IL: Charles C. Thomas; 2009.
59. Alexander W. Pharmacotherapy for post-traumatic stress disorder in combat veterans: focus on antidepressants and atypical antipsychotic agents. P T. 2012;37(1):32-38.
60. Beck JG, Sloan DM, Friedman MJ. Pharmacotherapy for PTSD. In: The Oxford Handbook of Traumatic Stress Disorders. Oxford University Press; 2012.
61. Waltman SH, Shearer D, Moore BA. Management of posttraumatic nightmares: a review of pharmacologic and nonpharmacologic treatments since 2013. Curr Psychiatry Rep. 2018;20(12):108.
62. Díaz-Gutiérrez MJ, Martínez-Cengotitabengoa M, Sáez de Adana E, et al. Relationship between the use of benzodiazepines and falls in older adults: a systematic review. Maturitas. 2017;101:17-22.
The Korean War lasted from June 25, 1950 through July 27, 1953. Although many veterans of the Korean War experienced traumas during extremely stressful combat conditions. However, they would not have been diagnosed with posttraumatic stress disorder (PTSD) at the time because the latter did not exist as a formal diagnosis until the publication of the third edition of the Diagnostic and Statistical Manual (DSM) in 1980.1 Prior to 1980, psychiatric syndromes resulting from war and combat exposure where known by numerous other terms including shell shock, chronic traumatic war neurosis, and combat fatigue/combat exhaustion.2,3 Military psychiatrists attended to combat fatigue during the course of the Korean War, but as was true of World War I and II, the focus was on returning soldiers to duty. Combat fatigue was generally viewed as a transient condition.4-8
Although now octo- and nonagenarians, in 2019 there are 1.2 million living Korean War veterans in the US, representing 6.7% of all current veterans.9 Understanding their war experiences and the nature of their current and past presentation of PTSD is relevant not only in formal mental health settings, but in primary care settings, including home-based primary care, as well as community living centers, skilled nursing facilities and assisted living facilities. Older adults with PTSD often present with somatic concerns rather than spontaneously reporting mental health symptoms.10 Beyond the short-term clinical management of Korean War veterans with PTSD, consideration of their experiences also has long-term relevance for the appropriate treatment of other veteran cohorts as they age in coming decades.
The purpose of this article is to provide a clinically focused overview of PTSD in Korean War veterans, to help promote understanding of this often-forgotten group of veterans, and to foster optimized personalized care. This overview will include a description of the Korean War veteran population and the Korean War itself, the manifestations and identification of PTSD among Korean War veterans, and treatment approaches using evidence-based psychotherapies and pharmacotherapies. Finally, we provide recommendations for future research to address present empirical gaps in the understanding and treatment of Korean War veterans with PTSD.
Causes and Course of the Korean War
When working with Korean War veterans it is important to consider the special nature of that specific conflict. Space considerations limit our ability to do justice to the complex history and numerous battles of the Korean War, but information in the following summary was gleaned from several excellent histories.11-13
The Korean War has been referred to as The Forgotten War, a concern expressed even during the latter parts of the war.14,15 But the war and its veterans warrant remembering. The root and proximal causes of the Korean War are complex and not fully agreed upon by the main participants.16-19 In part this may reflect the fact that there was no clear victor in the Korean War, so that the different protagonists have developed their own versions of the history of the conflict. Also, US involvement and the public reaction to the war must be viewed within the larger historical context of that time. This context included the recent end of 4 years of US involvement in World War II (1941-1945) and the subsequent rapid rise of Cold War tensions between the US and the Soviet Union. The latter also included a worldwide fear of nuclear war and the US fear of the global spread of communism. These fears were fueled by the Soviet-led Berlin Blockade from June 1948 through May 1949, the Soviet Union’s successful atomic bomb test in August 1949, the founding of the People’s Republic of China in October 1949, and the February 1950 Sino-Soviet Treaty of Friendship and Alliance.13
In the closing days of World War II, the US and Soviet Union agreed to a temporary division of Korea along the 38th parallel to facilitate timely and efficient surrender of Japanese troops. But as Cold War tensions rose, the temporary division became permanent, and Soviet- and US-backed governments of the north and south, respectively, were officially established on the Korean peninsula in 1948. Although by 1949 the Soviets and US had withdrawn most troops from the peninsula, tensions between the north and south continued to mount and hostilities increased. To this day the exact causes of the eruption of war remain disputed, although it is clear that ideological as well as economic factors played a role, and both leaders of North and South Korea were pledging to reunite the peninsula under their respective leadership.16-19 The tension culminated on June 25, 1950, when North Korean troops crossed the 38th parallel and invaded South Korea. On June 27, 1950, President Truman ordered US naval and air forces to support South Korea and then ordered the involvement of ground troops on June 30.16,17,19
Although several other member countries of the United Nations (UN) provided troops, 90% of the troops were from the US. About 5.7 million US military personnel served during the war, including about 1.8 million in Korea itself. The US forces experienced approximately 34,000 battle-related deaths, 103,000 were wounded, and 7,000 were prisoners of war (POWs).11,20-22 The nature and events of the Korean War made it particularly stressful and traumatizing for the soldiers, sailors, and marines involved throughout its entire course. These included near defeat in the early months, a widely alternating war front along the north/south axis during the first year, and subsequently, not only intense constant battles on the fronts, but also a demanding and exhausting guerrilla war in the south, which lasted throughout the remainder of the conflict.11,15 The US troops during the initial months of the war have been described as outnumbered and underprepared, as many in the initial phase were reassigned from peace-time occupation duty in Japan.7
The first year of war was characterized by a repeated north-to-south/south-to-north shifts in control of territory. During the first 3 months, the North Korean forces overwhelmed the South and captured control of all but 2 South Korean cities in the far southeastern region (Pusan, now Busan; and Daegu), and US and UN forces were forced to retreat to the perimeter around Pusan. The intense Battle of Pusan Perimeter lasted from August 4, 1950 to September 18, 1950, and resulted in massive causalities as well as a flood of civilian refugees.
The course of the war began to change in early September 1950 with the landing of amphibious US/UN forces at Inchon, behind North Korean lines, which cut off southern supply routes for the North Korean troops.11 US/UN forces soon crossed to the north of the 38th parallel and captured the North Korean capital, Pyongyang, on October 19, 1950. They continued to push north and approached the Yalu River border with China by late November 1950, but then the Chinese introduced their own troops forcing a southward retreat of US/UN troops during which there were again numerous US/UN casualties. Chinese troops retook Seoul in late December 1950/early January 1951. However, the US/UN forces soon recaptured Seoul and advanced back to the 38th parallel. This back-and-forth across the 38th parallel continued until July 1951 when the front line of battle stabilized there. Although the line stabilized, intense battles and casualties continued for 2 more years. During this period US/UN troops also had to deal with guerrilla warfare behind the front lines due to the actions of communist partisans and isolated North Korean troops. This situation continued until the armistice was signed July 27, 1953.
Trauma and Characteristic Stresses of the War
There were many factors that made the Korean War experience different from previous wars, particularly World War II. For example, in contrast to the strong public support during and after World War II, public support for the Korean War in the US was low, particularly during its final year.23 In public opinion polls from October 1952 through April 1953, only 23% to 39% reported feeling that the war was worth fighting.23 A retrospective 1985 survey also found that 70% of World War II veterans, but only 33% of Korean War veterans reported feeling appreciated by the US public on their return from the war.24
Those fighting in the initial months of the war faced a particularly grim situation. According to LTC Philip Smith, who served as Division Psychiatrist on the Masan Front (Pusan Perimeter) during August and September of 1950, “Fighting was almost continuous and all available troops were on the fighting front… For the most part these soldiers were soft from occupation duty, many had not received adequate combat basic training, no refresher combat training in Korea had as yet been instituted,” he reported.7 “The extremes of climate coupled with the generally rugged mountainous terrain in Korea were physical factors of importance…These men were psychologically unprepared for the horrors and isolation of war.” LTC Smith noted that the change in status from civilian or occupation life to the marked deprivation of the war in Korea had been “too abrupt to allow as yet for a reasonable adjustment to the new setting” and that as a result “the highest rate of wounded and neuropsychiatric casualties in the Korean campaign resulted.”7
Even after this initial period, the nature of the shifting war, the challenging terrain, the high military casualty rate, and the high rate of civilian casualties and displacement continued throughout the war.
PTSD in Korean War Veterans
It is clear that Korean War combat veterans were exposed to traumatic events. It is unknown how many developed PTSD. While notions of psychological distress and disability related to combat trauma exposure have existed for centuries, Korean War and World War II veterans are a remaining link to pre-DSM PTSD mental health in the military. Military/forward psychiatry—psychiatric services near the battle zone rather than requiring evacuation of patients—was present in Korea from the early months of the war, but the focus of forward psychiatry was to reduce psychiatric causalities from combat fatigue and maximize rapid return-to-duty.4-6 With no real conception of PTSD, there were limited treatments available, and evidenced-based trauma-focused treatments for PTSD would not be introduced for at least another 4 decades.27-29
Skinner and Kaplick conducted a historical review of case descriptions of trauma-related conditions from World War I through the Vietnam War and noted the consistent inclusion of hyperarousal and intrusive symptoms, although there also was a greater emphasis on somatic conversion or hysteria symptoms in the earlier descriptions.30 By the Korean War, descriptions of combat fatigue included a number of symptoms that overlap with PTSD, including preoccupation with the traumatic stressor, nightmares, irritability/anger, increased startle, and hyperarousal.31 But following the acute phases, attention to any chronic problems associated with these conditions waned. As was acknowledged by a military psychiatrist in a 1954 talk, studies of the long-term adjustment of those who had “broken down in combat” were sorely needed.6 In a small 1965 study reported by Archibald and Tuddenham, persistent symptoms of combat fatigue among Korean War veterans were definitely present, and there was even a suggestion that the symptoms had increased over the decade since the war.32
Given the stoicism that typified cultural expectations for military men during this period, Korean War veterans may also have been reluctant to seek mental health treatment either at the time or later. In short, it is likely that a nontrivial proportion of Korean War veterans with PTSD were underdiagnosed and received suboptimal or no mental health treatment for decades following their war experiences.33 Although the nature of the war, deployment, and public support were distinct in World War II vs the Korean War, the absence of attention to the long-term effects of disorders related to combat trauma and the cultural expectations for stoicism suggest that PTSD among aging World War II veterans may also have gone underrecognized and undertreated.
Apart from the lack of interest in chronic effects of stressors, another problem that has plagued the limited empirical research on Korean War veterans has been the propensity to combine Korean War with World War II veteran samples in studies. Because World War II veterans have outnumbered Korean War veterans until recently, combined samples tended to have relatively few Korean War veterans. Nevertheless, from those studies that have been reported in which 2 groups were compared, important differences have been revealed. Specifically, although precise estimates of the prevalence of PTSD among Korean War combat veterans have varied depending on sampling and method, studies from the 1990s and early 2000s suggested that the prevalence of PTSD and other mental health concerns as well as the severity of symptoms, suicide risk, and psychosocial adjustment difficulties were worse among Korean War combat veterans relative to those among World War II combat veterans; however, both groups had lower prevalence than did Vietnam War combat veterans.21,34-37 Several authors speculated that these differences in outcome were at least partially due to differences in public support for the respective wars.36,37
Although there has been a paucity of research on psychiatric issues and PTSD in Korean War veterans, POWs who were very likely to have been exposed to extreme psychological traumas have received some attention. There have been comparisons of mortality and morbidity among POWs from the Korean War (PWK), World War II Pacific Theater (PWJ), and Europe (PWE).38 Among measures that were administered to the former POWs, the overall pattern seen from survey data in the mid-1960s revealed significantly worse health and functioning among the PWK and PWJ groups relative to the PWE group, with psychiatric difficulties being the most commonly reported impairments among the former 2 groups. This pattern was found most strongly with regards to objective measures, such as hospitalizations for “psychoneuroses,” and US Department of Veterans Affairs (VA) disability records, as well as based on self-reported psychosocial/recreational difficulties measured using the Cornell Medical Index (CMI).38
Gold and colleagues reported a follow-up study of more than 700 former POWs who were reinterviewed between 1989 and 1992.39 Although there was no scale of PTSD symptoms prior to formulation of the diagnosis in 1980, the CMI was a self-reported checklist that included a large range of both medical as well as behavioral and psychiatric symptoms. Thus, using CMI survey responses from 1965, the authors examined the factor structure (ie, the correlational relationships between multiple scale items and subgroupings of items) of the CMI relative to diagnosis of PTSD in 1989 to 1992 based on results from the Structured Clinical Interview for the DSM-III-R (SCID). The intent was to help discern whether the component domains of PTSD were present and intercorrelated in a pattern similar to that of the contemporary diagnosis. The investigators examined the factor structure of 20 psychological items from the CMI that appeared relevant to PTSD criteria using the 1965 data. Three factors (subgroups of highly intercorrelated items) were found: irritability (31% of variance), fearfulness/anxiousness (9% of the variance), and social withdrawal (7% of the variance). Although these did not directly correspond to, or fully cover, DSM PTSD domains or criteria, there does appear to be a thematic resemblance of the CMI findings with PTSD, including alterations in arousal and mood, vigilance, and startle.
Identification and Treatment of PTSD in Older Veterans
Of the 1.2 million living Korean War veterans in the US, 36.3% use VA provided health care.40 There are a number of complicating factors to consider in the current identification and treatment of PTSD in this cohort, including their advanced age; physical, cognitive, and social changes associated with normal aging; the associated medical and cognitive comorbidities; and the specific social-contextual factors in that age cohort. Any combination of these factors may complicate recognition, diagnosis, and treatment. It is also important to be cognizant of the additional stressors that may have been experienced by ethnic minorites and women serving in Korea, which are poorly documented and studied. Racial integration of the US military began during the Korean War, but the general pattern was for African American soldiers to be assigned to all-white units, rather than the reverse.14,41,42 And although the majority of military personnel serving in Korea were male, there were women serving in health care positions at mobile army surgical hospital (MASH) units, medical air evacuation (Medevac) aircraft, and off-shore hospital ships.
The clinical presentation of PTSD in older adults has varied, which may partially relate to the time elapsed since the index trauma. For example, older veterans in general may show less avoidance behavior as a part of PTSD, but in those who experience trauma later in life there may actually be greater avoidance.43,44 There have also been discrepant reports of intrusion or reexperiencing of symptoms, with these also potentially reduced in older veterans.43,44 However, sleep disturbances seem to be very common among elderly combat veterans, and attention should be paid to the possible presence of sleep apnea, which may be more common in veterans with PTSD in general.43,45,46
PTSD symptoms may reemerge after decades of remission or quiescence during retirement and/or with the emergence of neurocognitive impairment, such as Alzheimer disease or dementia. These individuals may have more difficulty engaging in distracting activities and work and spend more time engaging in reminiscence about the past, which can include increased focus on traumatic memories.45,47 Davison and colleagues have suggested a concept they call later-adulthood trauma reengagement (LATR) where later in life combat veterans may “confront and rework their wartime memories in an effort to find meaning and build coherence.”48 This process can be a double-edged sword, leading at times positively to enhanced personal growth or negatively to increased symptoms; preventive interventions may be able foster a more positive outcome.48
There is some evidence supporting the validity of the Clinician Administered PTSD Scale (CAPS) for the evaluation of PTSD in older adults, although this was based on the DSM-III-revised criteria for PTSD and an earlier version of CAPS.49 Bhattarai and colleagues examined responses to the 35-item Mississippi Scale for Combat-Related PTSD (M-PTSD) using VA clinical data collected between 2008 and 2015 on veterans of each combat era from World War II through the post-9/11.50 Strong internal consistency and test-retest reliability of the M-PTSD was observed within each veteran era sample. However, using chart diagnosis of PTSD as the criterion standard, the cut-scores for optimal balance of sensitivity and specificity of the M-PTSD scores were substantially lower for the older cohorts (World War II and Korean War veterans) relative to those for Vietnam and more recent veteran cohorts. The authors concluded that M-PTSD can be validly used to screen for PTSD in veterans within each of these cohorts but recommended using lower than standard cut-scores for Korean War and World War II veterans.50
This is also consistent with reports that suggest the use of lower cut-scores on self-administered PTSD symptom screens.43,44 For the clinician interested in quantifying the severity of PTSD, the most recent tools available are the CAPS-5 and the PCL-5, which have both been created in accordance with the DSM-5. The CAPS-5 is a rater-administered tool, and the PCL-5 is self-administered by the veteran. Although there has been little research using these newer tools in geriatric populations, they can currently serve as a means of tracking the severity of PTSD while we await measures that are better validated in Korean War and other older veterans.
Beyond specific empirical guidance, VA clinicians must presently rely on clinical observations and experience. Patients from the Korean War cohort often present at the insistence of a family member for changes in sleep, mood, behavior, or cognition. When the veterans themselves present, older adults with PTSD often focus more on somatic concerns (including pain, sleep, and gastrointestinal disturbance) than psychiatric problems per se. The latter tendency may in part be due to the salience of such symptoms for them, but perhaps also due to considerable stigma of mental health care that is still largely present in this group.43,44
Psychotherapy
Current VA treatment guidelines recommend trauma-focused therapies, with the strongest evidence base for prolonged exposure (PE), cognitive processing therapy (CPT), and eye movement desensitization and reprocessing (EMDR) therapies.51
There have been several excellent prior reviews discussing treatment of PTSD in older adults generally.10,43,44,52 These reviews have invariably expressed concern about the lack of sufficient empirical studies, but based on evidence from studies and case reports, there seems to be tentative support that trauma-focused therapies are acceptable and efficacious for use with older adults with PTSD. In their recent scoping review, Pless Kaiser and colleagues made several recommendations for trauma-focused therapy with older adults, including slow/careful pacing and use of compensatory aids for cognitive and sensory deficits.44 When cognitive impairment has exacerbated PTSD symptoms, they suggest therapists consider using an adapted form of CPT completed without a trauma narrative. For PE they recommend extending content across sessions and involving spouse or caregivers to assist with in vivo exposure and homework completion.44
Recent studies suggest that PTSD may be a risk factor for the later development of neurodegenerative disorders, and it is often during assessments for dementia that a revelation of PTSD occurs.10,43,47,55 Cognitive impairment may also be of relevance in deciding on the type of psychotherapy to be implemented, as it may have more adverse effects on the effectiveness of CPT than of exposure-based treatments (PE or EMDR). It may be useful to perform a cognitive assessment prior to initiation of a cognitive-based therapy, although extensive cognitive testing may not be practical or may be contraindicated because of fatigue. A brief screening tool such as the Montreal Cognitive Assessment or the Mini-Mental State Examinationmay be helpful.56, 57
Prolonged exposure has been reported by many clinicians to be effective in older adults with PTSD; however, due consideration should be given to the needs of individuals, as many have functioned for decades by suppressing memories.
Apart from the treatment needs for specific PTSD symptoms, the decades-long effects of poor sleep, irritability, hypervigilance, and dissociation also have social consequences for patients, including marital discord and divorce, and social and family isolation that should be addressed in therapy when appropriate. In addition, many Korean War veterans, like all veterans, sought postmilitary employment in professions that are associated with higher rates of exposure to psychological trauma, such as police or fire departments, and this may have an exacerbating effect on PTSD.58
Pharmacotherapy
There is very little empirical evidence guiding pharmacologic approaches to PTSD in older veterans. This population is at increased risk for many comorbidities, and pharmacologic treatments many require dosage adjustments, as is the case for any geriatric patient. Selective serotonin reuptake inhibitor (SSRI) and serotonin norepinephrine reuptake inhibitor (SNRI) medications have been proposed for some cases of PTSD.59,60 Health care providers may consider the SSRIs escitalopram or sertraline preferentially given their decreased potential for drug-drug interactions, anticholinergic effects, or cardiac toxicity compared with that of other drugs in this class.60,61 As venlafaxine can increase blood pressure, especially at higher doses, prescribers may choose duloxetine as an alternative if a SNRI is indicated.60 For veterans when prazosin is being considered for nightmare control, monitoring for hypotension, orthostasis, and the administration of other antihypertensives or prostatic hypertrophy medications is necessary.61 The use of benzodiazepines, while not recommended for PTSD, should be viewed with even greater trepidation in a geriatric population given enhanced risk of falls and confusion in the geriatric veteran population.60,62
Conclusions
Many of the oldest veterans (aged > 80 years) are from the Korean War era. The harsh and unique nature of the war, as well as the differences in context and support from the US public, and the outcome of the war, may have all contributed to and elevation of “combat fatigue” and PTSD among combat veterans from the Korean War. As the “forgotten war” cohort also has been forgotten by researchers, relatively little is known about posttraumatic stress sequelae of these veterans in the decades following the war.
From available evidence, we can readily surmise that problems were underrecognized and suboptimally diagnosed and treated. There is tentative evidence supporting the use of standard interviews and rating scales, such as the CAPS, M-PTSD, and PCL, but lower cut-scores than applied with Vietnam and later veteran cohorts are generally recommended to avoid excessive false negative errors. In terms of psychotherapy treatment, there is again a stark paucity of systematic research, but the limited evidence from studies of PTSD treatment in older adults from the general population tentatively support the acceptability and potential efficacy of recognized evidence-based trauma-focused psychotherapies for PTSD. Research on medication treatment is similarly lacking, but the general recommendations for the use of SSRI or SNRI medications seem to be valid, at least in our clinical experience, and the general rules for geriatric psychopharmacology definitely apply here—start low, go slow.
There are several important avenues for future research. Most pressing among these are establishing the effectiveness of existing treatments, and the modifications that may be needed in the broader context of the above factors, as well as the physical and cognitive changes associated with advanced age. Further research on the phenomenologic aspects of PTSD among Korean War and subsequent cohorts are also needed, as the information obtained will not only guide more effective personalized treatment of the Korean War veterans who remain with us, but also inform future generations of care in terms of the degree and dimensions of variability that may present between cohorts and within cohorts over the life span.
The Korean War lasted from June 25, 1950 through July 27, 1953. Although many veterans of the Korean War experienced traumas during extremely stressful combat conditions. However, they would not have been diagnosed with posttraumatic stress disorder (PTSD) at the time because the latter did not exist as a formal diagnosis until the publication of the third edition of the Diagnostic and Statistical Manual (DSM) in 1980.1 Prior to 1980, psychiatric syndromes resulting from war and combat exposure where known by numerous other terms including shell shock, chronic traumatic war neurosis, and combat fatigue/combat exhaustion.2,3 Military psychiatrists attended to combat fatigue during the course of the Korean War, but as was true of World War I and II, the focus was on returning soldiers to duty. Combat fatigue was generally viewed as a transient condition.4-8
Although now octo- and nonagenarians, in 2019 there are 1.2 million living Korean War veterans in the US, representing 6.7% of all current veterans.9 Understanding their war experiences and the nature of their current and past presentation of PTSD is relevant not only in formal mental health settings, but in primary care settings, including home-based primary care, as well as community living centers, skilled nursing facilities and assisted living facilities. Older adults with PTSD often present with somatic concerns rather than spontaneously reporting mental health symptoms.10 Beyond the short-term clinical management of Korean War veterans with PTSD, consideration of their experiences also has long-term relevance for the appropriate treatment of other veteran cohorts as they age in coming decades.
The purpose of this article is to provide a clinically focused overview of PTSD in Korean War veterans, to help promote understanding of this often-forgotten group of veterans, and to foster optimized personalized care. This overview will include a description of the Korean War veteran population and the Korean War itself, the manifestations and identification of PTSD among Korean War veterans, and treatment approaches using evidence-based psychotherapies and pharmacotherapies. Finally, we provide recommendations for future research to address present empirical gaps in the understanding and treatment of Korean War veterans with PTSD.
Causes and Course of the Korean War
When working with Korean War veterans it is important to consider the special nature of that specific conflict. Space considerations limit our ability to do justice to the complex history and numerous battles of the Korean War, but information in the following summary was gleaned from several excellent histories.11-13
The Korean War has been referred to as The Forgotten War, a concern expressed even during the latter parts of the war.14,15 But the war and its veterans warrant remembering. The root and proximal causes of the Korean War are complex and not fully agreed upon by the main participants.16-19 In part this may reflect the fact that there was no clear victor in the Korean War, so that the different protagonists have developed their own versions of the history of the conflict. Also, US involvement and the public reaction to the war must be viewed within the larger historical context of that time. This context included the recent end of 4 years of US involvement in World War II (1941-1945) and the subsequent rapid rise of Cold War tensions between the US and the Soviet Union. The latter also included a worldwide fear of nuclear war and the US fear of the global spread of communism. These fears were fueled by the Soviet-led Berlin Blockade from June 1948 through May 1949, the Soviet Union’s successful atomic bomb test in August 1949, the founding of the People’s Republic of China in October 1949, and the February 1950 Sino-Soviet Treaty of Friendship and Alliance.13
In the closing days of World War II, the US and Soviet Union agreed to a temporary division of Korea along the 38th parallel to facilitate timely and efficient surrender of Japanese troops. But as Cold War tensions rose, the temporary division became permanent, and Soviet- and US-backed governments of the north and south, respectively, were officially established on the Korean peninsula in 1948. Although by 1949 the Soviets and US had withdrawn most troops from the peninsula, tensions between the north and south continued to mount and hostilities increased. To this day the exact causes of the eruption of war remain disputed, although it is clear that ideological as well as economic factors played a role, and both leaders of North and South Korea were pledging to reunite the peninsula under their respective leadership.16-19 The tension culminated on June 25, 1950, when North Korean troops crossed the 38th parallel and invaded South Korea. On June 27, 1950, President Truman ordered US naval and air forces to support South Korea and then ordered the involvement of ground troops on June 30.16,17,19
Although several other member countries of the United Nations (UN) provided troops, 90% of the troops were from the US. About 5.7 million US military personnel served during the war, including about 1.8 million in Korea itself. The US forces experienced approximately 34,000 battle-related deaths, 103,000 were wounded, and 7,000 were prisoners of war (POWs).11,20-22 The nature and events of the Korean War made it particularly stressful and traumatizing for the soldiers, sailors, and marines involved throughout its entire course. These included near defeat in the early months, a widely alternating war front along the north/south axis during the first year, and subsequently, not only intense constant battles on the fronts, but also a demanding and exhausting guerrilla war in the south, which lasted throughout the remainder of the conflict.11,15 The US troops during the initial months of the war have been described as outnumbered and underprepared, as many in the initial phase were reassigned from peace-time occupation duty in Japan.7
The first year of war was characterized by a repeated north-to-south/south-to-north shifts in control of territory. During the first 3 months, the North Korean forces overwhelmed the South and captured control of all but 2 South Korean cities in the far southeastern region (Pusan, now Busan; and Daegu), and US and UN forces were forced to retreat to the perimeter around Pusan. The intense Battle of Pusan Perimeter lasted from August 4, 1950 to September 18, 1950, and resulted in massive causalities as well as a flood of civilian refugees.
The course of the war began to change in early September 1950 with the landing of amphibious US/UN forces at Inchon, behind North Korean lines, which cut off southern supply routes for the North Korean troops.11 US/UN forces soon crossed to the north of the 38th parallel and captured the North Korean capital, Pyongyang, on October 19, 1950. They continued to push north and approached the Yalu River border with China by late November 1950, but then the Chinese introduced their own troops forcing a southward retreat of US/UN troops during which there were again numerous US/UN casualties. Chinese troops retook Seoul in late December 1950/early January 1951. However, the US/UN forces soon recaptured Seoul and advanced back to the 38th parallel. This back-and-forth across the 38th parallel continued until July 1951 when the front line of battle stabilized there. Although the line stabilized, intense battles and casualties continued for 2 more years. During this period US/UN troops also had to deal with guerrilla warfare behind the front lines due to the actions of communist partisans and isolated North Korean troops. This situation continued until the armistice was signed July 27, 1953.
Trauma and Characteristic Stresses of the War
There were many factors that made the Korean War experience different from previous wars, particularly World War II. For example, in contrast to the strong public support during and after World War II, public support for the Korean War in the US was low, particularly during its final year.23 In public opinion polls from October 1952 through April 1953, only 23% to 39% reported feeling that the war was worth fighting.23 A retrospective 1985 survey also found that 70% of World War II veterans, but only 33% of Korean War veterans reported feeling appreciated by the US public on their return from the war.24
Those fighting in the initial months of the war faced a particularly grim situation. According to LTC Philip Smith, who served as Division Psychiatrist on the Masan Front (Pusan Perimeter) during August and September of 1950, “Fighting was almost continuous and all available troops were on the fighting front… For the most part these soldiers were soft from occupation duty, many had not received adequate combat basic training, no refresher combat training in Korea had as yet been instituted,” he reported.7 “The extremes of climate coupled with the generally rugged mountainous terrain in Korea were physical factors of importance…These men were psychologically unprepared for the horrors and isolation of war.” LTC Smith noted that the change in status from civilian or occupation life to the marked deprivation of the war in Korea had been “too abrupt to allow as yet for a reasonable adjustment to the new setting” and that as a result “the highest rate of wounded and neuropsychiatric casualties in the Korean campaign resulted.”7
Even after this initial period, the nature of the shifting war, the challenging terrain, the high military casualty rate, and the high rate of civilian casualties and displacement continued throughout the war.
PTSD in Korean War Veterans
It is clear that Korean War combat veterans were exposed to traumatic events. It is unknown how many developed PTSD. While notions of psychological distress and disability related to combat trauma exposure have existed for centuries, Korean War and World War II veterans are a remaining link to pre-DSM PTSD mental health in the military. Military/forward psychiatry—psychiatric services near the battle zone rather than requiring evacuation of patients—was present in Korea from the early months of the war, but the focus of forward psychiatry was to reduce psychiatric causalities from combat fatigue and maximize rapid return-to-duty.4-6 With no real conception of PTSD, there were limited treatments available, and evidenced-based trauma-focused treatments for PTSD would not be introduced for at least another 4 decades.27-29
Skinner and Kaplick conducted a historical review of case descriptions of trauma-related conditions from World War I through the Vietnam War and noted the consistent inclusion of hyperarousal and intrusive symptoms, although there also was a greater emphasis on somatic conversion or hysteria symptoms in the earlier descriptions.30 By the Korean War, descriptions of combat fatigue included a number of symptoms that overlap with PTSD, including preoccupation with the traumatic stressor, nightmares, irritability/anger, increased startle, and hyperarousal.31 But following the acute phases, attention to any chronic problems associated with these conditions waned. As was acknowledged by a military psychiatrist in a 1954 talk, studies of the long-term adjustment of those who had “broken down in combat” were sorely needed.6 In a small 1965 study reported by Archibald and Tuddenham, persistent symptoms of combat fatigue among Korean War veterans were definitely present, and there was even a suggestion that the symptoms had increased over the decade since the war.32
Given the stoicism that typified cultural expectations for military men during this period, Korean War veterans may also have been reluctant to seek mental health treatment either at the time or later. In short, it is likely that a nontrivial proportion of Korean War veterans with PTSD were underdiagnosed and received suboptimal or no mental health treatment for decades following their war experiences.33 Although the nature of the war, deployment, and public support were distinct in World War II vs the Korean War, the absence of attention to the long-term effects of disorders related to combat trauma and the cultural expectations for stoicism suggest that PTSD among aging World War II veterans may also have gone underrecognized and undertreated.
Apart from the lack of interest in chronic effects of stressors, another problem that has plagued the limited empirical research on Korean War veterans has been the propensity to combine Korean War with World War II veteran samples in studies. Because World War II veterans have outnumbered Korean War veterans until recently, combined samples tended to have relatively few Korean War veterans. Nevertheless, from those studies that have been reported in which 2 groups were compared, important differences have been revealed. Specifically, although precise estimates of the prevalence of PTSD among Korean War combat veterans have varied depending on sampling and method, studies from the 1990s and early 2000s suggested that the prevalence of PTSD and other mental health concerns as well as the severity of symptoms, suicide risk, and psychosocial adjustment difficulties were worse among Korean War combat veterans relative to those among World War II combat veterans; however, both groups had lower prevalence than did Vietnam War combat veterans.21,34-37 Several authors speculated that these differences in outcome were at least partially due to differences in public support for the respective wars.36,37
Although there has been a paucity of research on psychiatric issues and PTSD in Korean War veterans, POWs who were very likely to have been exposed to extreme psychological traumas have received some attention. There have been comparisons of mortality and morbidity among POWs from the Korean War (PWK), World War II Pacific Theater (PWJ), and Europe (PWE).38 Among measures that were administered to the former POWs, the overall pattern seen from survey data in the mid-1960s revealed significantly worse health and functioning among the PWK and PWJ groups relative to the PWE group, with psychiatric difficulties being the most commonly reported impairments among the former 2 groups. This pattern was found most strongly with regards to objective measures, such as hospitalizations for “psychoneuroses,” and US Department of Veterans Affairs (VA) disability records, as well as based on self-reported psychosocial/recreational difficulties measured using the Cornell Medical Index (CMI).38
Gold and colleagues reported a follow-up study of more than 700 former POWs who were reinterviewed between 1989 and 1992.39 Although there was no scale of PTSD symptoms prior to formulation of the diagnosis in 1980, the CMI was a self-reported checklist that included a large range of both medical as well as behavioral and psychiatric symptoms. Thus, using CMI survey responses from 1965, the authors examined the factor structure (ie, the correlational relationships between multiple scale items and subgroupings of items) of the CMI relative to diagnosis of PTSD in 1989 to 1992 based on results from the Structured Clinical Interview for the DSM-III-R (SCID). The intent was to help discern whether the component domains of PTSD were present and intercorrelated in a pattern similar to that of the contemporary diagnosis. The investigators examined the factor structure of 20 psychological items from the CMI that appeared relevant to PTSD criteria using the 1965 data. Three factors (subgroups of highly intercorrelated items) were found: irritability (31% of variance), fearfulness/anxiousness (9% of the variance), and social withdrawal (7% of the variance). Although these did not directly correspond to, or fully cover, DSM PTSD domains or criteria, there does appear to be a thematic resemblance of the CMI findings with PTSD, including alterations in arousal and mood, vigilance, and startle.
Identification and Treatment of PTSD in Older Veterans
Of the 1.2 million living Korean War veterans in the US, 36.3% use VA provided health care.40 There are a number of complicating factors to consider in the current identification and treatment of PTSD in this cohort, including their advanced age; physical, cognitive, and social changes associated with normal aging; the associated medical and cognitive comorbidities; and the specific social-contextual factors in that age cohort. Any combination of these factors may complicate recognition, diagnosis, and treatment. It is also important to be cognizant of the additional stressors that may have been experienced by ethnic minorites and women serving in Korea, which are poorly documented and studied. Racial integration of the US military began during the Korean War, but the general pattern was for African American soldiers to be assigned to all-white units, rather than the reverse.14,41,42 And although the majority of military personnel serving in Korea were male, there were women serving in health care positions at mobile army surgical hospital (MASH) units, medical air evacuation (Medevac) aircraft, and off-shore hospital ships.
The clinical presentation of PTSD in older adults has varied, which may partially relate to the time elapsed since the index trauma. For example, older veterans in general may show less avoidance behavior as a part of PTSD, but in those who experience trauma later in life there may actually be greater avoidance.43,44 There have also been discrepant reports of intrusion or reexperiencing of symptoms, with these also potentially reduced in older veterans.43,44 However, sleep disturbances seem to be very common among elderly combat veterans, and attention should be paid to the possible presence of sleep apnea, which may be more common in veterans with PTSD in general.43,45,46
PTSD symptoms may reemerge after decades of remission or quiescence during retirement and/or with the emergence of neurocognitive impairment, such as Alzheimer disease or dementia. These individuals may have more difficulty engaging in distracting activities and work and spend more time engaging in reminiscence about the past, which can include increased focus on traumatic memories.45,47 Davison and colleagues have suggested a concept they call later-adulthood trauma reengagement (LATR) where later in life combat veterans may “confront and rework their wartime memories in an effort to find meaning and build coherence.”48 This process can be a double-edged sword, leading at times positively to enhanced personal growth or negatively to increased symptoms; preventive interventions may be able foster a more positive outcome.48
There is some evidence supporting the validity of the Clinician Administered PTSD Scale (CAPS) for the evaluation of PTSD in older adults, although this was based on the DSM-III-revised criteria for PTSD and an earlier version of CAPS.49 Bhattarai and colleagues examined responses to the 35-item Mississippi Scale for Combat-Related PTSD (M-PTSD) using VA clinical data collected between 2008 and 2015 on veterans of each combat era from World War II through the post-9/11.50 Strong internal consistency and test-retest reliability of the M-PTSD was observed within each veteran era sample. However, using chart diagnosis of PTSD as the criterion standard, the cut-scores for optimal balance of sensitivity and specificity of the M-PTSD scores were substantially lower for the older cohorts (World War II and Korean War veterans) relative to those for Vietnam and more recent veteran cohorts. The authors concluded that M-PTSD can be validly used to screen for PTSD in veterans within each of these cohorts but recommended using lower than standard cut-scores for Korean War and World War II veterans.50
This is also consistent with reports that suggest the use of lower cut-scores on self-administered PTSD symptom screens.43,44 For the clinician interested in quantifying the severity of PTSD, the most recent tools available are the CAPS-5 and the PCL-5, which have both been created in accordance with the DSM-5. The CAPS-5 is a rater-administered tool, and the PCL-5 is self-administered by the veteran. Although there has been little research using these newer tools in geriatric populations, they can currently serve as a means of tracking the severity of PTSD while we await measures that are better validated in Korean War and other older veterans.
Beyond specific empirical guidance, VA clinicians must presently rely on clinical observations and experience. Patients from the Korean War cohort often present at the insistence of a family member for changes in sleep, mood, behavior, or cognition. When the veterans themselves present, older adults with PTSD often focus more on somatic concerns (including pain, sleep, and gastrointestinal disturbance) than psychiatric problems per se. The latter tendency may in part be due to the salience of such symptoms for them, but perhaps also due to considerable stigma of mental health care that is still largely present in this group.43,44
Psychotherapy
Current VA treatment guidelines recommend trauma-focused therapies, with the strongest evidence base for prolonged exposure (PE), cognitive processing therapy (CPT), and eye movement desensitization and reprocessing (EMDR) therapies.51
There have been several excellent prior reviews discussing treatment of PTSD in older adults generally.10,43,44,52 These reviews have invariably expressed concern about the lack of sufficient empirical studies, but based on evidence from studies and case reports, there seems to be tentative support that trauma-focused therapies are acceptable and efficacious for use with older adults with PTSD. In their recent scoping review, Pless Kaiser and colleagues made several recommendations for trauma-focused therapy with older adults, including slow/careful pacing and use of compensatory aids for cognitive and sensory deficits.44 When cognitive impairment has exacerbated PTSD symptoms, they suggest therapists consider using an adapted form of CPT completed without a trauma narrative. For PE they recommend extending content across sessions and involving spouse or caregivers to assist with in vivo exposure and homework completion.44
Recent studies suggest that PTSD may be a risk factor for the later development of neurodegenerative disorders, and it is often during assessments for dementia that a revelation of PTSD occurs.10,43,47,55 Cognitive impairment may also be of relevance in deciding on the type of psychotherapy to be implemented, as it may have more adverse effects on the effectiveness of CPT than of exposure-based treatments (PE or EMDR). It may be useful to perform a cognitive assessment prior to initiation of a cognitive-based therapy, although extensive cognitive testing may not be practical or may be contraindicated because of fatigue. A brief screening tool such as the Montreal Cognitive Assessment or the Mini-Mental State Examinationmay be helpful.56, 57
Prolonged exposure has been reported by many clinicians to be effective in older adults with PTSD; however, due consideration should be given to the needs of individuals, as many have functioned for decades by suppressing memories.
Apart from the treatment needs for specific PTSD symptoms, the decades-long effects of poor sleep, irritability, hypervigilance, and dissociation also have social consequences for patients, including marital discord and divorce, and social and family isolation that should be addressed in therapy when appropriate. In addition, many Korean War veterans, like all veterans, sought postmilitary employment in professions that are associated with higher rates of exposure to psychological trauma, such as police or fire departments, and this may have an exacerbating effect on PTSD.58
Pharmacotherapy
There is very little empirical evidence guiding pharmacologic approaches to PTSD in older veterans. This population is at increased risk for many comorbidities, and pharmacologic treatments many require dosage adjustments, as is the case for any geriatric patient. Selective serotonin reuptake inhibitor (SSRI) and serotonin norepinephrine reuptake inhibitor (SNRI) medications have been proposed for some cases of PTSD.59,60 Health care providers may consider the SSRIs escitalopram or sertraline preferentially given their decreased potential for drug-drug interactions, anticholinergic effects, or cardiac toxicity compared with that of other drugs in this class.60,61 As venlafaxine can increase blood pressure, especially at higher doses, prescribers may choose duloxetine as an alternative if a SNRI is indicated.60 For veterans when prazosin is being considered for nightmare control, monitoring for hypotension, orthostasis, and the administration of other antihypertensives or prostatic hypertrophy medications is necessary.61 The use of benzodiazepines, while not recommended for PTSD, should be viewed with even greater trepidation in a geriatric population given enhanced risk of falls and confusion in the geriatric veteran population.60,62
Conclusions
Many of the oldest veterans (aged > 80 years) are from the Korean War era. The harsh and unique nature of the war, as well as the differences in context and support from the US public, and the outcome of the war, may have all contributed to and elevation of “combat fatigue” and PTSD among combat veterans from the Korean War. As the “forgotten war” cohort also has been forgotten by researchers, relatively little is known about posttraumatic stress sequelae of these veterans in the decades following the war.
From available evidence, we can readily surmise that problems were underrecognized and suboptimally diagnosed and treated. There is tentative evidence supporting the use of standard interviews and rating scales, such as the CAPS, M-PTSD, and PCL, but lower cut-scores than applied with Vietnam and later veteran cohorts are generally recommended to avoid excessive false negative errors. In terms of psychotherapy treatment, there is again a stark paucity of systematic research, but the limited evidence from studies of PTSD treatment in older adults from the general population tentatively support the acceptability and potential efficacy of recognized evidence-based trauma-focused psychotherapies for PTSD. Research on medication treatment is similarly lacking, but the general recommendations for the use of SSRI or SNRI medications seem to be valid, at least in our clinical experience, and the general rules for geriatric psychopharmacology definitely apply here—start low, go slow.
There are several important avenues for future research. Most pressing among these are establishing the effectiveness of existing treatments, and the modifications that may be needed in the broader context of the above factors, as well as the physical and cognitive changes associated with advanced age. Further research on the phenomenologic aspects of PTSD among Korean War and subsequent cohorts are also needed, as the information obtained will not only guide more effective personalized treatment of the Korean War veterans who remain with us, but also inform future generations of care in terms of the degree and dimensions of variability that may present between cohorts and within cohorts over the life span.
1. American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders, 3rd ed. Arlington VA: American Psychiatric Association; 1980.
2. Friedman MJ, Schnurr PP, McDonagh-Coyle A. Posttraumatic stress disorder in the military veteran. Psychiatr Clin North Am. 1994;17(2):265-277.
3. Salmon TW. The Care and Treatment of Mental Diseases and War Neuroses (“Shell Shock”) in the British Army. New York: War Work Committee of the National Committee for Mental Hygiene, Inc; 1917.
4. Jones E, Wessely S. “Forward psychiatry” in the military: its origins and effectiveness. J Trauma Stress. 2003;16(4):411-419.
5. Newman RA. Combat fatigue: a review to the Korean conflict. Mil Med. 1964;129:921-928.
6. Harris FG. Some comments on the differential diagnosis and treatment of psychiatric breakdowns in Korea. https://history.amedd.army.mil/booksdocs/korea/recad2/ch9-2.html. Published April 30, 1954. Accessed November 8, 2019.
7. Smith PB. Psychiatric experiences during the Korean conflict. Am Pract Dig Treat. 1955;6(2):183-189.
8. Koontz AR. Psychiatry in the Korean War. Military Surg.
1950;107(6):444-445.
9. US Department of Veterans Affairs. National Center for Veterans Analysis and Statistics. Population Tables - Table 2L: VETPOP2016 Living Veterans by period of service, gender, 2015-2045. https://www.va.gov/vetdata/docs/Demographics/New_Vetpop_Model/2L_VetPop2016
_POS_National.xlsx. Accessed November 8, 2019.
10. Cook JM, McCarthy E, Thorp SR. Older adults with PTSD: brief state of research and evidence-based psychotherapy case illustration. Am J Geriatr Psychiatry. 2017;25(5):522-530.
11. Millett AR. Korean War: 1950-1953. Encylopaedia Britannica. https://www.britannica.com/event/Korean-War#accordion-article-history. Updated Nov 7, 2019. Accessed November 8, 2019.
12. Stack L. Korean War, a ‘forgotten’ conflict that shaped the modern world. The New York Times. January 2, 2018. https://www.nytimes.com/2018/01/01/world/asia/korean-war-history.html. Accessed November 8, 2019.
13. Westad OA. The Cold War: A World History. New York: Basic Books; 2018.
14. Young C, Conard PL, Armstrong ML, Lacy D. Older military veteran care: many still believe they are forgotten. J Holist Nurs. 2018;36(3):291-300.
15. Huebner AJ. Kilroy is back, 1950-1953. In: The Warrior Image: Soldiers in American Culture From the Second World War to the Vietnam Era. Chapel Hill, NC: The University of North Carolina Press; 2008:97-131.
16. The annexation of Korea (editorial). Japan Times. https://www.japantimes.co.jp/opinion/2010/08/29/editorials/the-annexation-of-korea/#.XPgvJvlKhhE. Published August 29, 2010. Accessed November 8, 2019.
17. Gupta K. How did the Korean war begin? China Q. 1972;52:699-716.
18. Lin L, Zhao Y, Ogawa M, Hoge J, Kim BY. Whose history? An analysis of the Korean War in history textbooks from the United States, South Korea, Japan, and China. Social Studies. 2009;100(5):222-232.
19. Weathersby K. The Korean War revisited. Wilson Q. 1999;23(3):91.
20. US Department of Veterans Affairs, Office of Program and Data Analyses, Assistant Secretary for Planning and Analysis. Data on veterans of the Korean War. https://www.va.gov/vetdata/docs/SpecialReports/KW2000.pdf. Published June 2000. Accessed November 8, 2019.
21. Brooks MS, Fulton L. Evidence of poorer life-course mental health outcomes among veterans of the Korean War cohort. Aging Ment Health. 2010;14(2):177-183.
22. US Department of Veterans Affairs, Office of Public Affairs. America’s wars. https://www.va.gov/opa/publications/factsheets/fs_americas_wars.pdf. Accessed November 8, 2019.
23. Memorandum on recent polls on Korea. https://www.eisenhowerlibrary.gov/sites/default/files/research/online-documents/korean-war/public-opinion-1953-06-02.pdf. Published June 2, 1953. Accessed November 8, 2019.
24. Elder GH Jr, Clipp EC. Combat experience and emotional health: impairment and resilience in later life. J Pers. 1989;57(2):311-341.
25. US Department of Veterans Affairs. Public health: cold injuries. https://www.publichealth.va.gov/PUBLICHEALTH/exposures/cold-injuries/index.asp. Updated July 31, 2019. Accessed November 8, 2019.
26. US Department of Veterans Affairs. Korean War veterans health issues. https://www.va.gov/health-care/health-needs-conditions/health-issues-related-to-service-era
/korean-war/. Updated June 14, 2019. Accessed November 8, 2019.
27. Shapiro F. Efficacy of the eye movement desensitization procedure in the treatment of traumatic memories. J Trauma Stress. 1989;2(2):199-223.
28. Resick PA, Schnicke MK. Cognitive processing therapy for sexual assault victims. J Consul Clin Psychol. 1992;60(5):748-756.
29. Foa EB, Rothbaum BO. Treating Trauma of Rape: Cognitive-Behavioral Therapy for PTSD. New York: Guilford; 2001.
30. Skinner R, Kaplick PM. Cultural shift in mental illness: a comparison of stress responses in World War I and the Vietnam War. JRSM Open. 2017;8(12):2054270417746061.
31. Kardiner A, Spiegel H. War Stress and Neurotic Illness. New York: Hoeber; 1947.
32. Archibald HC, Tuddenham RD. Persistent stress reaction after combat: a 20-year follow-up. Arch Gen Psychiatry. 1965;12:475-481.
33. Cook JM, Simiola V. Trauma and aging. Curr Psychiatry Rep. 2018;20(10):93.
34. Rosenheck R, Fontana A. Long-term sequelae of combat in World War II, Korea and Vietnam: a comparative study. In: McCaughey BG, Fullerton CS, Ursano RJ, eds. Individual
and Community Responses to Trauma and Disaster: The Structure of Human Chaos. New York: Cambridge University Press; 1994:330-359.
35. Blake DD, Keane TM, Wine PR, Mora C, Taylor KL, Lyons JA. Prevalence of PTSD symptoms in combat veterans seeking medical treatment. J Trauma Stress. 1990;3(1):15-27.
36. McCranie EW, Hyer LA. Posttraumatic stress disorder symptoms in Korean conflict and World War II combat veterans seeking outpatient treatment. J Trauma Stress. 2000;13(3):427-439.
37. Fontana A, Rosenheck R. Traumatic war stressors and psychiatric symptoms among World War II, Korean, and Vietnam War veterans. Psychology Aging. 1994;9(1):27-33.
38. Beebe GW. Follow-up studies of World War II and Korean war prisoners. II. Morbidity, disability, and maladjustments. Am J Epidemiol. 1975;101(5):400-422.
39. Gold PB, Engdahl BE, Eberly RE, Blake RJ, Page WF, Frueh BC. Trauma exposure, resilience, social support, and PTSD construct validity among former prisoners of war. Social Psychiatry Psychiatr Epidemiol. 2000;35(1):36-42.
40. US Department of Veterans Affairs. Key statistics by veteran status and period of service. https://www.va.gov/vetdata/docs/SpecialReports/KeyStats.pdf. Accessed November 11, 2019.
41. Bowers WT, Hammond WM, MacGarrigle GL. Black Soldier, White Army. Washington DC: US Army Center of Military History; 1996.
42. Black HK. Three generations, three wars: African American veterans. Gerontologist. 2016;56(1):33-41.
43. Thorp SR, Sones HM, Cook JM. Posttraumatic stress disorder among older adults. In: Sorocco KH, Lauderdale S, eds. Cognitive Behavior Therapy With Older Adults: Innovations Across Care Settings. New York: Springer; 2011:189-217.
44. Pless Kaiser A, Cook JM, Glick DM, Moye J. Posttraumatic stress disorder in older adults: a conceptual review. Clinical Gerontol. 2019;42(4):359-376.
45. Sadavoy J. Survivors. A review of the late-life effects of prior psychological trauma. Am J Geriatr Psychiatry. 1997;5(4):287-301.
46. Tamanna S, Parker JD, Lyons J, Ullah MI. The effect of continuous positive air pressure (CPAP) on nightmares in patients with posttraumatic stress disorder (PTSD) and obstructive sleep apnea (OSA). J Clin Sleep Med. 2014;10(6):631-636.
47. Mota N, Tsai J, Kirwin PD, et al. Late-life exacerbation of PTSD symptoms in US veterans: results from the National Health and Resilience in Veterans Study. J Clin Psychiatry. 2016;77(3):348-354.
48. Davison EH, Kaiser AP, Spiro A 3rd, Moye J, King LA, King DW. From Late-onset stress symptomatology to later-adulthood trauma reengagement in aging combat veterans: taking a broader view. Gerontologist. 2016;56(1):14-21.
49. Hyer L, Summers MN, Boyd S, Litaker M, Boudewyns P. Assessment of older combat veterans with the clinician-administered PTSD scale. J Trauma Stress. 1996;9(3):587-593.
50. Bhattarai JJ, Oehlert ME, Weber DK. Psychometric properties of the Mississippi Scale for combat-related posttraumatic stress disorder based on veterans’ period of service. Psychol Serv. 2018. [Epub ahead of print]
51. US Department of Veterans Affairs, US Department of Defense. VA/DOD Clinical practice guideline for the management of posttraumatic stress disorder and acute stress disorder. Version 3.0. https://www.healthquality.va.gov/guidelines/MH/ptsd/VADoDPTSDCPGFinal012418.pdf.
Updated 2017. Accessed November 11, 2019.
52. Dinnen S, Simiola V, Cook JM. Post-traumatic stress disorder in older adults: a systematic review of the psychotherapy treatment literature. Aging Ment Health. 2015;19(2):144-150.
53. Jakel RJ. Posttraumatic Stress Disorder in the Elderly. Psychiatr Clin North Am. 2018;41(1):165-175.
54. Thorp SR, Glassman LH, Wells SY, et al. A randomized controlled trial of prolonged exposure therapy versus relaxation training for older veterans with military-related PTSD. J Anxiety Disord. 2019;64:45-54.
55. Kang B, Xu H, McConnell ES. Neurocognitive and psychiatric comorbidities of posttraumatic stress disorder among older veterans: a systematic review. Int J Geriatr Psychiatry. 2019;34(4):522-538.
56. Nasreddine ZS, Phillips NA, Bédirian V, et al. The Montreal Cognitive Assessment, MoCA: a brief screening tool for mild cognitive impairment. J Am Geriatr Soc. 2005;53(4):695-699.
57. Folstein MF, Folstein SE, McHugh PR. “Mini-mental state”. A practical method for grading the cognitive state of patients for the clinician. J Psychiatr Res. 1975;12(3):189-198.
58. Paton D. Traumatic Stress in Police Officers a Career-Length Assessment From Recruitment to Retirement. Springfield, IL: Charles C. Thomas; 2009.
59. Alexander W. Pharmacotherapy for post-traumatic stress disorder in combat veterans: focus on antidepressants and atypical antipsychotic agents. P T. 2012;37(1):32-38.
60. Beck JG, Sloan DM, Friedman MJ. Pharmacotherapy for PTSD. In: The Oxford Handbook of Traumatic Stress Disorders. Oxford University Press; 2012.
61. Waltman SH, Shearer D, Moore BA. Management of posttraumatic nightmares: a review of pharmacologic and nonpharmacologic treatments since 2013. Curr Psychiatry Rep. 2018;20(12):108.
62. Díaz-Gutiérrez MJ, Martínez-Cengotitabengoa M, Sáez de Adana E, et al. Relationship between the use of benzodiazepines and falls in older adults: a systematic review. Maturitas. 2017;101:17-22.
1. American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders, 3rd ed. Arlington VA: American Psychiatric Association; 1980.
2. Friedman MJ, Schnurr PP, McDonagh-Coyle A. Posttraumatic stress disorder in the military veteran. Psychiatr Clin North Am. 1994;17(2):265-277.
3. Salmon TW. The Care and Treatment of Mental Diseases and War Neuroses (“Shell Shock”) in the British Army. New York: War Work Committee of the National Committee for Mental Hygiene, Inc; 1917.
4. Jones E, Wessely S. “Forward psychiatry” in the military: its origins and effectiveness. J Trauma Stress. 2003;16(4):411-419.
5. Newman RA. Combat fatigue: a review to the Korean conflict. Mil Med. 1964;129:921-928.
6. Harris FG. Some comments on the differential diagnosis and treatment of psychiatric breakdowns in Korea. https://history.amedd.army.mil/booksdocs/korea/recad2/ch9-2.html. Published April 30, 1954. Accessed November 8, 2019.
7. Smith PB. Psychiatric experiences during the Korean conflict. Am Pract Dig Treat. 1955;6(2):183-189.
8. Koontz AR. Psychiatry in the Korean War. Military Surg.
1950;107(6):444-445.
9. US Department of Veterans Affairs. National Center for Veterans Analysis and Statistics. Population Tables - Table 2L: VETPOP2016 Living Veterans by period of service, gender, 2015-2045. https://www.va.gov/vetdata/docs/Demographics/New_Vetpop_Model/2L_VetPop2016
_POS_National.xlsx. Accessed November 8, 2019.
10. Cook JM, McCarthy E, Thorp SR. Older adults with PTSD: brief state of research and evidence-based psychotherapy case illustration. Am J Geriatr Psychiatry. 2017;25(5):522-530.
11. Millett AR. Korean War: 1950-1953. Encylopaedia Britannica. https://www.britannica.com/event/Korean-War#accordion-article-history. Updated Nov 7, 2019. Accessed November 8, 2019.
12. Stack L. Korean War, a ‘forgotten’ conflict that shaped the modern world. The New York Times. January 2, 2018. https://www.nytimes.com/2018/01/01/world/asia/korean-war-history.html. Accessed November 8, 2019.
13. Westad OA. The Cold War: A World History. New York: Basic Books; 2018.
14. Young C, Conard PL, Armstrong ML, Lacy D. Older military veteran care: many still believe they are forgotten. J Holist Nurs. 2018;36(3):291-300.
15. Huebner AJ. Kilroy is back, 1950-1953. In: The Warrior Image: Soldiers in American Culture From the Second World War to the Vietnam Era. Chapel Hill, NC: The University of North Carolina Press; 2008:97-131.
16. The annexation of Korea (editorial). Japan Times. https://www.japantimes.co.jp/opinion/2010/08/29/editorials/the-annexation-of-korea/#.XPgvJvlKhhE. Published August 29, 2010. Accessed November 8, 2019.
17. Gupta K. How did the Korean war begin? China Q. 1972;52:699-716.
18. Lin L, Zhao Y, Ogawa M, Hoge J, Kim BY. Whose history? An analysis of the Korean War in history textbooks from the United States, South Korea, Japan, and China. Social Studies. 2009;100(5):222-232.
19. Weathersby K. The Korean War revisited. Wilson Q. 1999;23(3):91.
20. US Department of Veterans Affairs, Office of Program and Data Analyses, Assistant Secretary for Planning and Analysis. Data on veterans of the Korean War. https://www.va.gov/vetdata/docs/SpecialReports/KW2000.pdf. Published June 2000. Accessed November 8, 2019.
21. Brooks MS, Fulton L. Evidence of poorer life-course mental health outcomes among veterans of the Korean War cohort. Aging Ment Health. 2010;14(2):177-183.
22. US Department of Veterans Affairs, Office of Public Affairs. America’s wars. https://www.va.gov/opa/publications/factsheets/fs_americas_wars.pdf. Accessed November 8, 2019.
23. Memorandum on recent polls on Korea. https://www.eisenhowerlibrary.gov/sites/default/files/research/online-documents/korean-war/public-opinion-1953-06-02.pdf. Published June 2, 1953. Accessed November 8, 2019.
24. Elder GH Jr, Clipp EC. Combat experience and emotional health: impairment and resilience in later life. J Pers. 1989;57(2):311-341.
25. US Department of Veterans Affairs. Public health: cold injuries. https://www.publichealth.va.gov/PUBLICHEALTH/exposures/cold-injuries/index.asp. Updated July 31, 2019. Accessed November 8, 2019.
26. US Department of Veterans Affairs. Korean War veterans health issues. https://www.va.gov/health-care/health-needs-conditions/health-issues-related-to-service-era
/korean-war/. Updated June 14, 2019. Accessed November 8, 2019.
27. Shapiro F. Efficacy of the eye movement desensitization procedure in the treatment of traumatic memories. J Trauma Stress. 1989;2(2):199-223.
28. Resick PA, Schnicke MK. Cognitive processing therapy for sexual assault victims. J Consul Clin Psychol. 1992;60(5):748-756.
29. Foa EB, Rothbaum BO. Treating Trauma of Rape: Cognitive-Behavioral Therapy for PTSD. New York: Guilford; 2001.
30. Skinner R, Kaplick PM. Cultural shift in mental illness: a comparison of stress responses in World War I and the Vietnam War. JRSM Open. 2017;8(12):2054270417746061.
31. Kardiner A, Spiegel H. War Stress and Neurotic Illness. New York: Hoeber; 1947.
32. Archibald HC, Tuddenham RD. Persistent stress reaction after combat: a 20-year follow-up. Arch Gen Psychiatry. 1965;12:475-481.
33. Cook JM, Simiola V. Trauma and aging. Curr Psychiatry Rep. 2018;20(10):93.
34. Rosenheck R, Fontana A. Long-term sequelae of combat in World War II, Korea and Vietnam: a comparative study. In: McCaughey BG, Fullerton CS, Ursano RJ, eds. Individual
and Community Responses to Trauma and Disaster: The Structure of Human Chaos. New York: Cambridge University Press; 1994:330-359.
35. Blake DD, Keane TM, Wine PR, Mora C, Taylor KL, Lyons JA. Prevalence of PTSD symptoms in combat veterans seeking medical treatment. J Trauma Stress. 1990;3(1):15-27.
36. McCranie EW, Hyer LA. Posttraumatic stress disorder symptoms in Korean conflict and World War II combat veterans seeking outpatient treatment. J Trauma Stress. 2000;13(3):427-439.
37. Fontana A, Rosenheck R. Traumatic war stressors and psychiatric symptoms among World War II, Korean, and Vietnam War veterans. Psychology Aging. 1994;9(1):27-33.
38. Beebe GW. Follow-up studies of World War II and Korean war prisoners. II. Morbidity, disability, and maladjustments. Am J Epidemiol. 1975;101(5):400-422.
39. Gold PB, Engdahl BE, Eberly RE, Blake RJ, Page WF, Frueh BC. Trauma exposure, resilience, social support, and PTSD construct validity among former prisoners of war. Social Psychiatry Psychiatr Epidemiol. 2000;35(1):36-42.
40. US Department of Veterans Affairs. Key statistics by veteran status and period of service. https://www.va.gov/vetdata/docs/SpecialReports/KeyStats.pdf. Accessed November 11, 2019.
41. Bowers WT, Hammond WM, MacGarrigle GL. Black Soldier, White Army. Washington DC: US Army Center of Military History; 1996.
42. Black HK. Three generations, three wars: African American veterans. Gerontologist. 2016;56(1):33-41.
43. Thorp SR, Sones HM, Cook JM. Posttraumatic stress disorder among older adults. In: Sorocco KH, Lauderdale S, eds. Cognitive Behavior Therapy With Older Adults: Innovations Across Care Settings. New York: Springer; 2011:189-217.
44. Pless Kaiser A, Cook JM, Glick DM, Moye J. Posttraumatic stress disorder in older adults: a conceptual review. Clinical Gerontol. 2019;42(4):359-376.
45. Sadavoy J. Survivors. A review of the late-life effects of prior psychological trauma. Am J Geriatr Psychiatry. 1997;5(4):287-301.
46. Tamanna S, Parker JD, Lyons J, Ullah MI. The effect of continuous positive air pressure (CPAP) on nightmares in patients with posttraumatic stress disorder (PTSD) and obstructive sleep apnea (OSA). J Clin Sleep Med. 2014;10(6):631-636.
47. Mota N, Tsai J, Kirwin PD, et al. Late-life exacerbation of PTSD symptoms in US veterans: results from the National Health and Resilience in Veterans Study. J Clin Psychiatry. 2016;77(3):348-354.
48. Davison EH, Kaiser AP, Spiro A 3rd, Moye J, King LA, King DW. From Late-onset stress symptomatology to later-adulthood trauma reengagement in aging combat veterans: taking a broader view. Gerontologist. 2016;56(1):14-21.
49. Hyer L, Summers MN, Boyd S, Litaker M, Boudewyns P. Assessment of older combat veterans with the clinician-administered PTSD scale. J Trauma Stress. 1996;9(3):587-593.
50. Bhattarai JJ, Oehlert ME, Weber DK. Psychometric properties of the Mississippi Scale for combat-related posttraumatic stress disorder based on veterans’ period of service. Psychol Serv. 2018. [Epub ahead of print]
51. US Department of Veterans Affairs, US Department of Defense. VA/DOD Clinical practice guideline for the management of posttraumatic stress disorder and acute stress disorder. Version 3.0. https://www.healthquality.va.gov/guidelines/MH/ptsd/VADoDPTSDCPGFinal012418.pdf.
Updated 2017. Accessed November 11, 2019.
52. Dinnen S, Simiola V, Cook JM. Post-traumatic stress disorder in older adults: a systematic review of the psychotherapy treatment literature. Aging Ment Health. 2015;19(2):144-150.
53. Jakel RJ. Posttraumatic Stress Disorder in the Elderly. Psychiatr Clin North Am. 2018;41(1):165-175.
54. Thorp SR, Glassman LH, Wells SY, et al. A randomized controlled trial of prolonged exposure therapy versus relaxation training for older veterans with military-related PTSD. J Anxiety Disord. 2019;64:45-54.
55. Kang B, Xu H, McConnell ES. Neurocognitive and psychiatric comorbidities of posttraumatic stress disorder among older veterans: a systematic review. Int J Geriatr Psychiatry. 2019;34(4):522-538.
56. Nasreddine ZS, Phillips NA, Bédirian V, et al. The Montreal Cognitive Assessment, MoCA: a brief screening tool for mild cognitive impairment. J Am Geriatr Soc. 2005;53(4):695-699.
57. Folstein MF, Folstein SE, McHugh PR. “Mini-mental state”. A practical method for grading the cognitive state of patients for the clinician. J Psychiatr Res. 1975;12(3):189-198.
58. Paton D. Traumatic Stress in Police Officers a Career-Length Assessment From Recruitment to Retirement. Springfield, IL: Charles C. Thomas; 2009.
59. Alexander W. Pharmacotherapy for post-traumatic stress disorder in combat veterans: focus on antidepressants and atypical antipsychotic agents. P T. 2012;37(1):32-38.
60. Beck JG, Sloan DM, Friedman MJ. Pharmacotherapy for PTSD. In: The Oxford Handbook of Traumatic Stress Disorders. Oxford University Press; 2012.
61. Waltman SH, Shearer D, Moore BA. Management of posttraumatic nightmares: a review of pharmacologic and nonpharmacologic treatments since 2013. Curr Psychiatry Rep. 2018;20(12):108.
62. Díaz-Gutiérrez MJ, Martínez-Cengotitabengoa M, Sáez de Adana E, et al. Relationship between the use of benzodiazepines and falls in older adults: a systematic review. Maturitas. 2017;101:17-22.
The Worst and the Best of 2019
Readers may recall that at the end of each calendar as opposed to fiscal year—I know it is hard to believe time exists outside the Federal system—Federal Practitioner publishes my ethics-focused version of the familiar year-end roundup. This year I am reversing the typical order of most annual rankings by putting the worst first for 2 morally salient reasons.
The first is that, sadly, it is almost always easier to identify multiple incidents that compete ignominiously for the “worst” of federal health care. Even more disappointing, it is comparatively difficult to find stories for the “best” that are of the same scale and scope as the bad news. This is not to say that every day there are not individual narratives of courage and compassion reported in US Department of Defense, US Public Health Service, and US Department of Veterans Affairs (VA), and hundreds more unsung heroes.
The second reason is that as human beings our psychology is such that we gravitate toward the worst things more powerfully and persistently than we do the best. This is in part why it is more difficult to find uplifting stories and why the demoralizing ones affect us so strongly. In an exhaustive review of the subject, psychologists Roy Baumeister and colleagues conclude that,
When equal measures of good and bad are present, however, the psychological effects of bad ones outweigh those of the good ones. This may in fact be a general principle or law of psychological phenomena, possibly reflecting the innate predispositions of the psyche or at least reflecting the almost inevitable adaptation of each individual to the exigencies of daily life.2
I am thus saving the best for last in the hope that it will be more memorable and impactful than the worst.
Unique to this year’s look-back, both the negative and the positive accounts come from the domain of end-of-life care. And unlike prior reviews where the lack of administrative vigilance and professional competence affected hundreds of patients, families, and staff, each of this year’s incidents involve a single patient.
An incident that occurred in September 2019 at a VA Community Living Center (CLC) in Georgia stood out in infamy apart from all others. It was the report of a veteran in a VA nursing home who had been bitten more than 100 times by ants crawling all over his room. He died shortly afterward. In a scene out of a horror movie tapping into the most primeval human fears, his daughter Laquana Ross described her father, a Vietnam Air Force veteran with cancer, to media and VA officials in graphic terms. “I understand mistakes happen,” she said. “I’ve had ants. But he was bit by ants two days in a row. They feasted on him.”3
In this new era of holding its senior executive service accountable, the outraged chair of the Senate Veterans Affairs Committee demanded that heads roll, and the VA acted rapidly to comply.4 The VA Central Office placed the network director on administrative leave, reassigned the chief medical officer, and initiated quality and safety reviews as well as an administrative investigative board to scrutinize how the parent Atlanta VA medical center managed the situation. In total, 9 officials connected to the incident were placed on leave. The VA apologized, with VA Secretary Robert Wilke zeroing in on the core values involved in the tragedy, “This is about basic humanity and dignity,” he said. “I don’t care what steps were taken to address the issues. We did not treat a vet with the dignity that he and his family deserved.”5 Yet it was the veteran’s daughter, with unbelievable charity, who asked the most crucial question that must be answered within the framework of a just culture if similar tragedies are not to occur in the future, “I know the staff, without a shadow of doubt, respected my dad and even loved him,” Ross said. “But what’s their ability to assess situations and fix things?”3
To begin to give Ms. Ross the answer she deserves, we must understand that the antithesis of love is not hate but indifference; of compassion, it is not cruelty but coldness. A true just culture reserves individual blame for those who have ill-will and adopts a systems perspective of organizational improvement toward most other types of errors.6 This means that the deplorable conditions in the CLC cannot be charged to the failure of a single staff member to fulfil their obligations but to collective collapse at many levels of the organization. Just culture is ethically laudable and far superior to the history in federal service of capricious punishment or institutional apathy that far too often were the default reactions to media exposures or congressional ire. Justice, though necessary, is not sufficient to achieve virtue. Those who work in health care also must be inspired to offer mercy, kindness, and compassion, especially in our most sacred privilege to provide care of the dying.
The best of 2019 illustrates this distinction movingly. This account also involves a Vietnam veteran, this time a Marine also dying of cancer, which happened just about a month after the earlier report. To be transparent it occurred at my home VA medical center in New Mexico. I was peripherally involved in the case as a consultant but had no role in the wondrous things that transpired. The last wish of a patient dying in the hospice unit on campus was to see his beloved dog who had been taken to the local city animal shelter when he was hospitalized because he had no friends or family to look after the companion animal. A social worker on the palliative care team called the animal shelter and explained the patient did not have much time left but wanted to see his dog before he died. Working together with support from facility leadership, shelter workers brought the dog to visit with the patient for an entire day while hospice staff cried with joy and sadness.7
As the epigraph for this editorial from Dame Cicely Saunders, the founder of the modern hospice movement, says, the difference between unspeakable pain and meaningful suffering can be measured in the depth of compassion caregivers show to the dying. It is this quality of mercy that in one case condemns, and in the other praises, us all as health care and administrative professionals in the service of our country. Baumeister and colleagues suggest that the human tendency to magnify the bad and minimize the good in everyday myopia may in a wider vision actually be a reason for hope:
It may be that humans and animals show heightened awareness of and responded more quickly to negative information because it signals a need for change. Hence, the adaptiveness of self-regulation partly lies in the organism’s ability to detect when response modifications are necessary and when they are unnecessary. Moreover, the lessons learned from bad events should ideally be retained permanently so that the same dangers or costs are not encountered repeatedly. Meanwhile, good events (such as those that provide a feeling of satisfaction and contentment) should ideally wear off so that the organism is motivated to continue searching for more and better outcomes.2
Let us all take this lesson into our work in 2020 so that when it comes time to write this column next year in the chilling cold of late autumn there will be more stories of light than darkness from which to choose.
1. Saunders C. The management of patients in the terminal stage. In: Raven R, ed. Cancer, Vol. 6. London: Butterworth and Company; 1960:403-417.
2. Baumeister RF, Bratslavasky E, Finkenauer C, Vohs KD. Bad is stronger than good. Rev General Psychol. 2001;5(4);323-370.
3. Knowles H. ‘They feasted on him’: Ants at VA nursing home bite a veteran 100 times before his death, daughter says. Washington Post. September 17, 2019. https://www.washingtonpost.com/health/2019/09/13/they-feasted-him-ants-va-nursing-home-bit-veteran-times-before-his-death-daughter-says. Accessed November 25, 2019.
4. Axelrod T. GOP senator presses VA after veteran reportedly bitten by ants in nursing home. https://thehill.com/homenews/senate/461196-gop-senator-presses-va-after-veteran-reportedly-bitten-by-ants-at-nursing. Published September 12, 2019. Accessed November 25, 2019.
5. Kime P. Nine VA leaders, staff placed on leave amid anti-bite scandal. https://www.military.com/daily-news/2019/09/17/nine-va-leaders-staff-placed-leave-amid-ant-bite-scandal.html. Published September 17, 2019. Accessed November 22, 2019.
6. Sculli GL, Hemphill R. Culture of safety and just culture. https://www.patientsafety.va.gov/docs/joe/just_culture_2013_tagged.pdf. Accessed November 22, 2019.
7. Hughes M. A Vietnam veteran in hospice care got to see his beloved dog one last time. https://www.cnn.com/2019/10/21/us/veteran-dying-wish-dog-trnd/index.html. Published October 21, 2019. Accessed November 22, 2019.
Readers may recall that at the end of each calendar as opposed to fiscal year—I know it is hard to believe time exists outside the Federal system—Federal Practitioner publishes my ethics-focused version of the familiar year-end roundup. This year I am reversing the typical order of most annual rankings by putting the worst first for 2 morally salient reasons.
The first is that, sadly, it is almost always easier to identify multiple incidents that compete ignominiously for the “worst” of federal health care. Even more disappointing, it is comparatively difficult to find stories for the “best” that are of the same scale and scope as the bad news. This is not to say that every day there are not individual narratives of courage and compassion reported in US Department of Defense, US Public Health Service, and US Department of Veterans Affairs (VA), and hundreds more unsung heroes.
The second reason is that as human beings our psychology is such that we gravitate toward the worst things more powerfully and persistently than we do the best. This is in part why it is more difficult to find uplifting stories and why the demoralizing ones affect us so strongly. In an exhaustive review of the subject, psychologists Roy Baumeister and colleagues conclude that,
When equal measures of good and bad are present, however, the psychological effects of bad ones outweigh those of the good ones. This may in fact be a general principle or law of psychological phenomena, possibly reflecting the innate predispositions of the psyche or at least reflecting the almost inevitable adaptation of each individual to the exigencies of daily life.2
I am thus saving the best for last in the hope that it will be more memorable and impactful than the worst.
Unique to this year’s look-back, both the negative and the positive accounts come from the domain of end-of-life care. And unlike prior reviews where the lack of administrative vigilance and professional competence affected hundreds of patients, families, and staff, each of this year’s incidents involve a single patient.
An incident that occurred in September 2019 at a VA Community Living Center (CLC) in Georgia stood out in infamy apart from all others. It was the report of a veteran in a VA nursing home who had been bitten more than 100 times by ants crawling all over his room. He died shortly afterward. In a scene out of a horror movie tapping into the most primeval human fears, his daughter Laquana Ross described her father, a Vietnam Air Force veteran with cancer, to media and VA officials in graphic terms. “I understand mistakes happen,” she said. “I’ve had ants. But he was bit by ants two days in a row. They feasted on him.”3
In this new era of holding its senior executive service accountable, the outraged chair of the Senate Veterans Affairs Committee demanded that heads roll, and the VA acted rapidly to comply.4 The VA Central Office placed the network director on administrative leave, reassigned the chief medical officer, and initiated quality and safety reviews as well as an administrative investigative board to scrutinize how the parent Atlanta VA medical center managed the situation. In total, 9 officials connected to the incident were placed on leave. The VA apologized, with VA Secretary Robert Wilke zeroing in on the core values involved in the tragedy, “This is about basic humanity and dignity,” he said. “I don’t care what steps were taken to address the issues. We did not treat a vet with the dignity that he and his family deserved.”5 Yet it was the veteran’s daughter, with unbelievable charity, who asked the most crucial question that must be answered within the framework of a just culture if similar tragedies are not to occur in the future, “I know the staff, without a shadow of doubt, respected my dad and even loved him,” Ross said. “But what’s their ability to assess situations and fix things?”3
To begin to give Ms. Ross the answer she deserves, we must understand that the antithesis of love is not hate but indifference; of compassion, it is not cruelty but coldness. A true just culture reserves individual blame for those who have ill-will and adopts a systems perspective of organizational improvement toward most other types of errors.6 This means that the deplorable conditions in the CLC cannot be charged to the failure of a single staff member to fulfil their obligations but to collective collapse at many levels of the organization. Just culture is ethically laudable and far superior to the history in federal service of capricious punishment or institutional apathy that far too often were the default reactions to media exposures or congressional ire. Justice, though necessary, is not sufficient to achieve virtue. Those who work in health care also must be inspired to offer mercy, kindness, and compassion, especially in our most sacred privilege to provide care of the dying.
The best of 2019 illustrates this distinction movingly. This account also involves a Vietnam veteran, this time a Marine also dying of cancer, which happened just about a month after the earlier report. To be transparent it occurred at my home VA medical center in New Mexico. I was peripherally involved in the case as a consultant but had no role in the wondrous things that transpired. The last wish of a patient dying in the hospice unit on campus was to see his beloved dog who had been taken to the local city animal shelter when he was hospitalized because he had no friends or family to look after the companion animal. A social worker on the palliative care team called the animal shelter and explained the patient did not have much time left but wanted to see his dog before he died. Working together with support from facility leadership, shelter workers brought the dog to visit with the patient for an entire day while hospice staff cried with joy and sadness.7
As the epigraph for this editorial from Dame Cicely Saunders, the founder of the modern hospice movement, says, the difference between unspeakable pain and meaningful suffering can be measured in the depth of compassion caregivers show to the dying. It is this quality of mercy that in one case condemns, and in the other praises, us all as health care and administrative professionals in the service of our country. Baumeister and colleagues suggest that the human tendency to magnify the bad and minimize the good in everyday myopia may in a wider vision actually be a reason for hope:
It may be that humans and animals show heightened awareness of and responded more quickly to negative information because it signals a need for change. Hence, the adaptiveness of self-regulation partly lies in the organism’s ability to detect when response modifications are necessary and when they are unnecessary. Moreover, the lessons learned from bad events should ideally be retained permanently so that the same dangers or costs are not encountered repeatedly. Meanwhile, good events (such as those that provide a feeling of satisfaction and contentment) should ideally wear off so that the organism is motivated to continue searching for more and better outcomes.2
Let us all take this lesson into our work in 2020 so that when it comes time to write this column next year in the chilling cold of late autumn there will be more stories of light than darkness from which to choose.
Readers may recall that at the end of each calendar as opposed to fiscal year—I know it is hard to believe time exists outside the Federal system—Federal Practitioner publishes my ethics-focused version of the familiar year-end roundup. This year I am reversing the typical order of most annual rankings by putting the worst first for 2 morally salient reasons.
The first is that, sadly, it is almost always easier to identify multiple incidents that compete ignominiously for the “worst” of federal health care. Even more disappointing, it is comparatively difficult to find stories for the “best” that are of the same scale and scope as the bad news. This is not to say that every day there are not individual narratives of courage and compassion reported in US Department of Defense, US Public Health Service, and US Department of Veterans Affairs (VA), and hundreds more unsung heroes.
The second reason is that as human beings our psychology is such that we gravitate toward the worst things more powerfully and persistently than we do the best. This is in part why it is more difficult to find uplifting stories and why the demoralizing ones affect us so strongly. In an exhaustive review of the subject, psychologists Roy Baumeister and colleagues conclude that,
When equal measures of good and bad are present, however, the psychological effects of bad ones outweigh those of the good ones. This may in fact be a general principle or law of psychological phenomena, possibly reflecting the innate predispositions of the psyche or at least reflecting the almost inevitable adaptation of each individual to the exigencies of daily life.2
I am thus saving the best for last in the hope that it will be more memorable and impactful than the worst.
Unique to this year’s look-back, both the negative and the positive accounts come from the domain of end-of-life care. And unlike prior reviews where the lack of administrative vigilance and professional competence affected hundreds of patients, families, and staff, each of this year’s incidents involve a single patient.
An incident that occurred in September 2019 at a VA Community Living Center (CLC) in Georgia stood out in infamy apart from all others. It was the report of a veteran in a VA nursing home who had been bitten more than 100 times by ants crawling all over his room. He died shortly afterward. In a scene out of a horror movie tapping into the most primeval human fears, his daughter Laquana Ross described her father, a Vietnam Air Force veteran with cancer, to media and VA officials in graphic terms. “I understand mistakes happen,” she said. “I’ve had ants. But he was bit by ants two days in a row. They feasted on him.”3
In this new era of holding its senior executive service accountable, the outraged chair of the Senate Veterans Affairs Committee demanded that heads roll, and the VA acted rapidly to comply.4 The VA Central Office placed the network director on administrative leave, reassigned the chief medical officer, and initiated quality and safety reviews as well as an administrative investigative board to scrutinize how the parent Atlanta VA medical center managed the situation. In total, 9 officials connected to the incident were placed on leave. The VA apologized, with VA Secretary Robert Wilke zeroing in on the core values involved in the tragedy, “This is about basic humanity and dignity,” he said. “I don’t care what steps were taken to address the issues. We did not treat a vet with the dignity that he and his family deserved.”5 Yet it was the veteran’s daughter, with unbelievable charity, who asked the most crucial question that must be answered within the framework of a just culture if similar tragedies are not to occur in the future, “I know the staff, without a shadow of doubt, respected my dad and even loved him,” Ross said. “But what’s their ability to assess situations and fix things?”3
To begin to give Ms. Ross the answer she deserves, we must understand that the antithesis of love is not hate but indifference; of compassion, it is not cruelty but coldness. A true just culture reserves individual blame for those who have ill-will and adopts a systems perspective of organizational improvement toward most other types of errors.6 This means that the deplorable conditions in the CLC cannot be charged to the failure of a single staff member to fulfil their obligations but to collective collapse at many levels of the organization. Just culture is ethically laudable and far superior to the history in federal service of capricious punishment or institutional apathy that far too often were the default reactions to media exposures or congressional ire. Justice, though necessary, is not sufficient to achieve virtue. Those who work in health care also must be inspired to offer mercy, kindness, and compassion, especially in our most sacred privilege to provide care of the dying.
The best of 2019 illustrates this distinction movingly. This account also involves a Vietnam veteran, this time a Marine also dying of cancer, which happened just about a month after the earlier report. To be transparent it occurred at my home VA medical center in New Mexico. I was peripherally involved in the case as a consultant but had no role in the wondrous things that transpired. The last wish of a patient dying in the hospice unit on campus was to see his beloved dog who had been taken to the local city animal shelter when he was hospitalized because he had no friends or family to look after the companion animal. A social worker on the palliative care team called the animal shelter and explained the patient did not have much time left but wanted to see his dog before he died. Working together with support from facility leadership, shelter workers brought the dog to visit with the patient for an entire day while hospice staff cried with joy and sadness.7
As the epigraph for this editorial from Dame Cicely Saunders, the founder of the modern hospice movement, says, the difference between unspeakable pain and meaningful suffering can be measured in the depth of compassion caregivers show to the dying. It is this quality of mercy that in one case condemns, and in the other praises, us all as health care and administrative professionals in the service of our country. Baumeister and colleagues suggest that the human tendency to magnify the bad and minimize the good in everyday myopia may in a wider vision actually be a reason for hope:
It may be that humans and animals show heightened awareness of and responded more quickly to negative information because it signals a need for change. Hence, the adaptiveness of self-regulation partly lies in the organism’s ability to detect when response modifications are necessary and when they are unnecessary. Moreover, the lessons learned from bad events should ideally be retained permanently so that the same dangers or costs are not encountered repeatedly. Meanwhile, good events (such as those that provide a feeling of satisfaction and contentment) should ideally wear off so that the organism is motivated to continue searching for more and better outcomes.2
Let us all take this lesson into our work in 2020 so that when it comes time to write this column next year in the chilling cold of late autumn there will be more stories of light than darkness from which to choose.
1. Saunders C. The management of patients in the terminal stage. In: Raven R, ed. Cancer, Vol. 6. London: Butterworth and Company; 1960:403-417.
2. Baumeister RF, Bratslavasky E, Finkenauer C, Vohs KD. Bad is stronger than good. Rev General Psychol. 2001;5(4);323-370.
3. Knowles H. ‘They feasted on him’: Ants at VA nursing home bite a veteran 100 times before his death, daughter says. Washington Post. September 17, 2019. https://www.washingtonpost.com/health/2019/09/13/they-feasted-him-ants-va-nursing-home-bit-veteran-times-before-his-death-daughter-says. Accessed November 25, 2019.
4. Axelrod T. GOP senator presses VA after veteran reportedly bitten by ants in nursing home. https://thehill.com/homenews/senate/461196-gop-senator-presses-va-after-veteran-reportedly-bitten-by-ants-at-nursing. Published September 12, 2019. Accessed November 25, 2019.
5. Kime P. Nine VA leaders, staff placed on leave amid anti-bite scandal. https://www.military.com/daily-news/2019/09/17/nine-va-leaders-staff-placed-leave-amid-ant-bite-scandal.html. Published September 17, 2019. Accessed November 22, 2019.
6. Sculli GL, Hemphill R. Culture of safety and just culture. https://www.patientsafety.va.gov/docs/joe/just_culture_2013_tagged.pdf. Accessed November 22, 2019.
7. Hughes M. A Vietnam veteran in hospice care got to see his beloved dog one last time. https://www.cnn.com/2019/10/21/us/veteran-dying-wish-dog-trnd/index.html. Published October 21, 2019. Accessed November 22, 2019.
1. Saunders C. The management of patients in the terminal stage. In: Raven R, ed. Cancer, Vol. 6. London: Butterworth and Company; 1960:403-417.
2. Baumeister RF, Bratslavasky E, Finkenauer C, Vohs KD. Bad is stronger than good. Rev General Psychol. 2001;5(4);323-370.
3. Knowles H. ‘They feasted on him’: Ants at VA nursing home bite a veteran 100 times before his death, daughter says. Washington Post. September 17, 2019. https://www.washingtonpost.com/health/2019/09/13/they-feasted-him-ants-va-nursing-home-bit-veteran-times-before-his-death-daughter-says. Accessed November 25, 2019.
4. Axelrod T. GOP senator presses VA after veteran reportedly bitten by ants in nursing home. https://thehill.com/homenews/senate/461196-gop-senator-presses-va-after-veteran-reportedly-bitten-by-ants-at-nursing. Published September 12, 2019. Accessed November 25, 2019.
5. Kime P. Nine VA leaders, staff placed on leave amid anti-bite scandal. https://www.military.com/daily-news/2019/09/17/nine-va-leaders-staff-placed-leave-amid-ant-bite-scandal.html. Published September 17, 2019. Accessed November 22, 2019.
6. Sculli GL, Hemphill R. Culture of safety and just culture. https://www.patientsafety.va.gov/docs/joe/just_culture_2013_tagged.pdf. Accessed November 22, 2019.
7. Hughes M. A Vietnam veteran in hospice care got to see his beloved dog one last time. https://www.cnn.com/2019/10/21/us/veteran-dying-wish-dog-trnd/index.html. Published October 21, 2019. Accessed November 22, 2019.
Simple prevention strategies can lessen postoperative delirium after orthopedic surgery
A new study has found that and a prevention program can help improve staff education and outcomes.
“In an aging society, it is very important to develop and implement a strategy for POD prevention to ensure that aging patients are treated as safely and effectively as possible,” wrote Jung-Yeon Choi of Seoul (South Korea) National University Bundang Hospital and coauthors. The study was published in BMC Geriatrics.
To determine how to better identify and treat high-risk patients for POD after orthopedic surgery, the researchers led a retrospective cohort study that included an intervention group of participants who were aged at least 65 years (n = 275) and a control group from a year prior (n = 274). Patients in the intervention group had their risk of delirium assessed and categorized using a simple screening tool, and those deemed at risk were entered into a multicomponent delirium prevention program.
Of the 275 patients in the intervention group, 144 required screening for delirium. Ninety-nine were classified as low risk, 29 were classified as high risk, and 16 missed the screening. Fifty-three additional patients were classified as high risk because they were aged 80 years or older. During the study, 17 participants experienced POD, 16 of whom were classified as high risk. In regard to estimating POD risk, the sensitivity and specificity of the delirium screening tool were 94.1% and 72.7%, respectively. Incidence rates of POD were 10.2% in the control group and 6.2% in the intervention group.
The authors noted their study’s limitations, including its design as a retrospective review of medical records rather than a prospective randomized controlled trial. In addition, because it was conducted in just one teaching hospital, they deemed it “not possible to determine the generalizability and long-term effect of our findings.”
The authors reported no conflicts of interest.
SOURCE: Choi JY et al. BMC Geriatr. 2019 Oct 26. doi: 10.1186/s12877-019-1303-z.
A new study has found that and a prevention program can help improve staff education and outcomes.
“In an aging society, it is very important to develop and implement a strategy for POD prevention to ensure that aging patients are treated as safely and effectively as possible,” wrote Jung-Yeon Choi of Seoul (South Korea) National University Bundang Hospital and coauthors. The study was published in BMC Geriatrics.
To determine how to better identify and treat high-risk patients for POD after orthopedic surgery, the researchers led a retrospective cohort study that included an intervention group of participants who were aged at least 65 years (n = 275) and a control group from a year prior (n = 274). Patients in the intervention group had their risk of delirium assessed and categorized using a simple screening tool, and those deemed at risk were entered into a multicomponent delirium prevention program.
Of the 275 patients in the intervention group, 144 required screening for delirium. Ninety-nine were classified as low risk, 29 were classified as high risk, and 16 missed the screening. Fifty-three additional patients were classified as high risk because they were aged 80 years or older. During the study, 17 participants experienced POD, 16 of whom were classified as high risk. In regard to estimating POD risk, the sensitivity and specificity of the delirium screening tool were 94.1% and 72.7%, respectively. Incidence rates of POD were 10.2% in the control group and 6.2% in the intervention group.
The authors noted their study’s limitations, including its design as a retrospective review of medical records rather than a prospective randomized controlled trial. In addition, because it was conducted in just one teaching hospital, they deemed it “not possible to determine the generalizability and long-term effect of our findings.”
The authors reported no conflicts of interest.
SOURCE: Choi JY et al. BMC Geriatr. 2019 Oct 26. doi: 10.1186/s12877-019-1303-z.
A new study has found that and a prevention program can help improve staff education and outcomes.
“In an aging society, it is very important to develop and implement a strategy for POD prevention to ensure that aging patients are treated as safely and effectively as possible,” wrote Jung-Yeon Choi of Seoul (South Korea) National University Bundang Hospital and coauthors. The study was published in BMC Geriatrics.
To determine how to better identify and treat high-risk patients for POD after orthopedic surgery, the researchers led a retrospective cohort study that included an intervention group of participants who were aged at least 65 years (n = 275) and a control group from a year prior (n = 274). Patients in the intervention group had their risk of delirium assessed and categorized using a simple screening tool, and those deemed at risk were entered into a multicomponent delirium prevention program.
Of the 275 patients in the intervention group, 144 required screening for delirium. Ninety-nine were classified as low risk, 29 were classified as high risk, and 16 missed the screening. Fifty-three additional patients were classified as high risk because they were aged 80 years or older. During the study, 17 participants experienced POD, 16 of whom were classified as high risk. In regard to estimating POD risk, the sensitivity and specificity of the delirium screening tool were 94.1% and 72.7%, respectively. Incidence rates of POD were 10.2% in the control group and 6.2% in the intervention group.
The authors noted their study’s limitations, including its design as a retrospective review of medical records rather than a prospective randomized controlled trial. In addition, because it was conducted in just one teaching hospital, they deemed it “not possible to determine the generalizability and long-term effect of our findings.”
The authors reported no conflicts of interest.
SOURCE: Choi JY et al. BMC Geriatr. 2019 Oct 26. doi: 10.1186/s12877-019-1303-z.
FROM BMC GERIATRICS
How to respond to flu vaccine doubters
The benefits of influenza vaccination are clear to those in the medical community. Yet misinformation and unfounded fears continue to discourage some people from getting a flu shot. During the 2018–2019 influenza season, only 45% of US adults and 63% of children were vaccinated.1
‘IT DOESN’T WORK FOR MANY PEOPLE’
Multiple studies have shown that the flu vaccine prevents millions of flu cases and flu-related doctor’s visits each year. During the 2016–2017 flu season, flu vaccine prevented an estimated 5.3 million influenza cases, 2.6 million influenza-associated medical visits, and 85,000 influenza-associated hospitalizations.2
Several viral and host factors affect vaccine effectiveness. In seasons when the vaccine viruses have matched circulating strains, flu vaccine has been shown to reduce the following:
- The risk of having to go to the doctor with flu by 40% to 60%
- Children’s risk of flu-related death and intensive care unit (ICU) admission by 74%
- The risk in adults of flu-associated hospitalizations by 40% and ICU admission by 82%
- The rate of cardiac events in people with heart disease
- Hospitalizations in people with diabetes or underlying chronic lung disease.3
In people hospitalized with influenza despite receiving the flu vaccine for the season, studies have shown that receiving the flu vaccine shortens the average duration of hospitalization, reduces the chance of ICU admission by 59%, shortens the duration of ICU stay by 4 days, and reduces deaths.3
‘IT TARGETS THE WRONG VIRUS’
Selecting an effective influenza vaccine is a challenge. Every year, the World Health Organization and the CDC decide on the influenza strains expected to circulate in the upcoming flu season in the Northern Hemisphere, based on data for circulating strains in the Southern Hemisphere. This decision takes place about 7 months before the expected onset of the flu season. Flu viruses may mutate between the time the decision is made and the time the vaccine is administered (as well as after the flu season starts). Also, vaccine production in eggs needs time, which is why this decision must be made several months ahead of the flu season.
Vaccine effectiveness varies by virus serotype. Vaccines are typically less effective against influenza A H3N2 viruses than against influenza A H1N1 and influenza B viruses. Effectiveness also varies from season to season depending on how close the vaccine serotypes match the circulating serotypes, but some effectiveness is retained even in seasons when some of the serotypes don’t match circulating viruses. For example, in the 2017–2018 season, when the influenza A H3N2 vaccine serotype did not match the circulating serotype, the overall effectiveness in preventing medically attended, laboratory-confirmed influenza virus infection was 36%.5
A universal flu vaccine that does not need to be updated annually is the ultimate solution, but according to the National Institute of Allergy and Infectious Diseases, such a vaccine is likely several years away.6
‘IT MAKES PEOPLE SICK’
Pain at the injection site of a flu shot occurs in 10% to 65% of people, lasts less than 2 days, and does not usually interfere with daily activities.7
Systemic symptoms such as fever, malaise, and myalgia may occur in people who have had no previous exposure to the influenza virus antigens in the vaccine, particularly in children. In adults, the frequency of systemic symptoms after the flu shot is similar to that with placebo.
The Vaccine Adverse Event Reporting System, which has been capturing data since 1990, shows that the influenza vaccine accounted for 5.7% of people who developed malaise after receiving any vaccine.8
The injectable inactivated influenza vaccine cannot biologically cause an influenza virus-related illness, since the inactivated vaccine viruses can elicit a protective immune response but cannot replicate. The nasal live-attenuated flu vaccine can in theory cause acute illness in the person receiving it, but because it is cold-adapted, it multiplies only in the colder environment of the nasal epithelium, not in the lower airways where the temperature is higher. Consequently, the vaccine virus triggers immunity by multiplying in the nose, but doesn’t infect the lungs.
From 10% to 50% of people who receive the nasal live-attenuated vaccine develop runny nose, wheezing, headache, vomiting, muscle aches, fever, sore throat, or cough shortly after receiving the vaccine, but these symptoms are usually mild and short-lived.
The most common reactions people have to flu vaccines are considerably less severe than the symptoms caused by actual flu illness.
While influenza illness results in natural immunity to the specific viral serotype causing it, this illness results in hospitalization in 2% and is fatal in 0.16% of people. Influenza vaccine results in immunity to the serotypes included in the vaccine, and multiple studies have not found a causal relationship between vaccination and death.9
‘IT CAUSES GUILLAIN-BARRÉ SYNDROME’
In the United States, 3,000 to 6,000 people per year develop Guillain-Barré syndrome, or 1 to 2 of every 100,000, which translates to 80 to 160 cases per week.10 While the exact cause of Guillain-Barré syndrome is unknown, about two-thirds of people have an acute diarrheal or respiratory illness within 3 months before the onset of symptoms. In 1976, the estimated attributable risk of influenza vaccine-related Guillain-Barré syndrome in the US adult population was 1 case per 100,000 in the 6 weeks after vaccination.11 Studies in subsequent influenza seasons have not shown similar findings.12 In fact, one study showed that the risk of developing Guillain-Barré syndrome was 15 times higher after influenza illness than after influenza vaccination.13
Since 5% to 15% of the US population develop symptomatic influenza annually,14 the decision to vaccinate with respect to the risk of Guillain-Barré syndrome should be obvious: vaccinate. The correct question to ask before influenza vaccination should be, “Have you previously developed Guillain-Barré syndrome within 6 weeks after receiving the flu vaccine?” If the answer is yes, the CDC considers this a caution, not a contraindication against receiving the influenza vaccine, since the benefit may still outweigh the risk.
‘I GOT THE FLU SHOT AND STILL GOT SICK’
The flu vaccine does not prevent illnesses caused by other viruses or bacteria that can make people sick during flu season. Influenza, the common cold, and streptococcal pharyngitis can have similar symptoms that make it difficult for patients—and, frequently, even healthcare providers—to distinguish between these illnesses with certainty.
One study suggested that influenza vaccine recipients had an increased risk of virologically confirmed noninfluenza respiratory viral infections,15 citing the phenomenon of virus interference that was described in the 1940s16 as a potential explanation. In essence, people protected against influenza by the vaccine may lack temporary nonspecific immunity against other respiratory viruses. However, these findings have not been replicated in subsequent studies.17
Viral gastroenteritis, mistakenly called “stomach flu,” is also not prevented by influenza vaccination.
‘I’M ALLERGIC TO EGGS’
The prevalence of egg allergy in US children is 0.5% to 2.5%.18 Most outgrow it by school age, but in one-third, the allergy persists into adulthood.
In general, people who can eat lightly cooked eggs (eg, scrambled eggs) without a reaction are unlikely to be allergic. On the other hand, the fact that egg-allergic people may tolerate egg included in baked products does not exclude the possibility of egg allergy. Egg allergy can be confirmed by a consistent medical history of adverse reaction to eggs and egg-containing foods, in addition to skin or blood testing for immunoglobulin E directed against egg proteins.19
Most currently available influenza vaccines are prepared by propagation of virus in embryonated eggs and so may contain trace amounts of egg proteins such as ovalbumin, with the exception of the inactivated quadrivalent recombinant influenza vaccine (Flublok) and the inactivated quadrivalent cell culture-based vaccine (Flucelvax).
The ACIP recommends that persons with a history of urticaria (hives) after exposure to eggs should receive any licensed, recommended influenza vaccine that is otherwise appropriate for their age and health status. Persons who report having angioedema, respiratory distress, lightheadedness, or recurrent vomiting, or who required epinephrine or another emergency medical intervention after exposure to eggs, should receive the influenza vaccine in an inpatient or outpatient medical setting under the supervision of a healthcare provider who is able to recognize and manage severe allergic reactions.
A history of severe allergic reaction such as anaphylaxis to a previous dose of any influenza vaccine, regardless of the vaccine component (including eggs) suspected of being responsible for the reaction, is a contraindication to influenza vaccination. The ACIP recommends that vaccine providers consider observing patients for 15 minutes after administration of any vaccine (regardless of history of egg allergy) to decrease the risk of injury should syncope occur.20
‘I DON’T WANT TO PUT POISONOUS MERCURY IN MY BODY’
A process of biomagnification of methylmercury occurs when humans eat large fish that have eaten smaller fish. Thus, larger fish such as shark can be hazardous for women who are or may become pregnant, for nursing mothers, and for young children, while smaller fish such as herring are relatively safe.
As a precautionary measure, thimerosal was taken out of childhood vaccines in the United States in 2001. Thimerosal-free influenza vaccine formulations include the nasal live-attenuated flu vaccine, the inactivated quadrivalent recombinant influenza vaccine, and the inactivated quadrivalent cell culture-based vaccine.
‘I DON’T LIKE NEEDLES’
At least 10% of US adults have aichmophobia, the fear of sharp objects including needles.22 Vasovagal syncope is the most common manifestation. Behavioral therapy, topical anesthetics, and systemic anxiolytics have variable efficacy in treating needle phobia. For those who are absolutely averse to needles, the nasal flu vaccine is an appropriate alternative.
‘I DON’T WANT TO TAKE ANYTHING THAT CAN MESS WITH MY OTHER MEDICATIONS’
Some immunosuppressive medications may decrease influenza vaccine immunogenicity. Concomitant administration of the inactivated influenza vaccine with other vaccines is safe and does not alter immunogenicity of other vaccines.1 The live-attenuated influenza vaccine is contraindicated in children and adolescents taking aspirin or other salicylates due to the risk of Reye syndrome.
‘I’M AFRAID IT WILL TRIGGER AN IMMUNE RESPONSE THAT WILL MAKE MY ASTHMA WORSE’
A recent systematic review and meta-analysis showed that the inactivated influenza vaccine is not associated with asthma exacerbation.23 However, the nasal live-attenuated influenza vaccine is contraindicated in children 2 to 4 years old who have asthma and should be used with caution in persons with asthma 5 years old and older. In the systematic review, influenza vaccine prevented 59% to 78% of asthma attacks leading to emergency visits or hospitalization.23 In other immune-mediated diseases such as rheumatoid arthritis, influenza vaccine does not precipitate exacerbations.24
‘I HAD AN ORGAN TRANSPLANT, AND I’M AFRAID THE FLU SHOT WILL CAUSE ORGAN REJECTION’
A study of 51,730 kidney transplant recipients found that receipt of the inactivated influenza vaccine in the first year after transplant was associated with a lower risk of subsequent allograft loss (adjusted hazard ratio 0.77; 95% confidence interval 0.69–0.85; P < .001) and death (adjusted hazard ratio 0.82; 95% confidence interval 0.76–0.89; P < .001).25 In the same study, although acute rejection in the first year was not associated with influenza vaccination, influenza infection in the first year was associated with rejection (odds ratio 1.58; 95% confidence interval 1.10–2.26; P < 0.001), but not with graft loss or death. Solid organ transplant recipients should receive the inactivated influenza vaccine starting 3 months after transplant.26
Influenza vaccination has not been shown to precipitate graft-vs-host disease in hematopoietic stem cell transplant recipients. These patients should also receive the inactivated influenza vaccine starting 3 to 6 months after transplant.27
The nasal live-attenuated influenza vaccine is contraindicated in these immunocompromised patients.
‘I’M PREGNANT, AND I DON’T WANT TO EXPOSE MY UNBORN BABY TO ANYTHING POTENTIALLY HARMFUL’
The morbidity and mortality risk from influenza is high in children under 2 years old because of low immunogenicity to flu vaccine. This is particularly true in children younger than 6 months, but the vaccine is not recommended in this population. The best way to protect infants is for all household members to be vaccinated against the flu.
Equally important, morbidity and mortality risk from influenza is much higher in pregnant women than in the general population. Many studies have shown the value of influenza vaccination during pregnancy for both mothers and their infants. A recently published study showed that 18% of infants who developed influenza required hospitalization.28 In that study, prenatal and postpartum maternal influenza vaccination decreased the odds of influenza in infants by 61% and 53%, respectively. Another study showed that vaccine effectiveness did not vary by gestational age at vaccination.29 A post hoc analysis of an influenza vaccination study in pregnant women suggested that the vaccine was also associated with decreased rates of pertussis in these women.30
Healthcare providers should try to understand the public’s misconceptions31 about seasonal influenza and influenza vaccines in order to best address them.
- Centers for Disease Control and Prevention (CDC). Flu vaccination coverage, United States, 2018–19 influenza season. www.cdc.gov/flu/fluvaxview/coverage-1819estimates.htm. Accessed November 13, 2019.
- Centers for Disease Control and Prevention (CDC). Immunogenicity, efficacy, and effectiveness of influenza vaccines. www.cdc.gov/flu/professionals/acip/immunogenicity.htm. Accessed November 13, 2019.
- Centers for Disease Control and Prevention (CDC). What are the benefits of flu vaccination? www.cdc.gov/flu/prevent/vaccine-benefits.htm. Accessed November 13, 2019.
- Grohskopf LA, Alyanak E, Broder KR, Walter EB, Fry AM, Jernigan DB. Prevention and control of seasonal influenza with vaccines: recommendations of the Advisory Committee on Immunization Practices—United States, 2019–20 influenza season. MMWR Recomm Rep 2019; 68(3):1–21. doi:10.15585/mmwr.rr6803a1
- Flannery B, Chung JR, Belongia EA, et al. Interim estimates of 2017–18 seasonal influenza vaccine effectiveness—United States, February 2018. MMWR Morb Mortal Wkly Rep 2018; 67(6):180–185. doi:10.15585/mmwr.mm6706a2
- Erbelding EJ, Post DJ, Stemmy EJ, et al. A universal influenza vaccine: the strategic plan for the National Institute of Allergy and Infectious Diseases. J Infect Dis 2018; 218(3):347–354. doi:10.1093/infdis/jiy103
- Centers for Disease Control and Prevention (CDC). Seasonal influenza vaccine safety: a summary for clinicians. www.cdc.gov/flu/professionals/vaccination/vaccine_safety.htm. Accessed November 13, 2019.
- Centers for Disease Control and Prevention (CDC). About the Vaccine Adverse Event Reporting System (VAERS). https://wonder.cdc.gov/vaers.html. Accessed November 13, 2019.
- Miller ER, Moro PL, Cano M, Shimabukuro TT. Deaths following vaccination: what does the evidence show? Vaccine 2015; 33(29):3288–3292. doi:10.1016/j.vaccine.2015.05.023
- Centers for Disease Control and Prevention (CDC). Guillain-Barré syndrome and flu vaccine. www.cdc.gov/flu/prevent/guillainbarre.htm. Accessed November 13, 2019.
- Schonberger LB, Bregman DJ, Sullivan-Bolyai JZ, et al. Guillain-Barre syndrome following vaccination in the national influenza immunization program, United States, 1976–1977. Am J Epidemiol 1979; 110(2):105–123. doi:10.1093/oxfordjournals.aje.a112795
- Baxter R, Bakshi N, Fireman B, et al. Lack of association of Guillain-Barré syndrome with vaccinations. Clin Infect Dis 2013; 57(2):197–204. doi:10.1093/cid/cit222
- Kwong JC, Vasa PP, Campitelli MA, et al. Risk of Guillain-Barré syndrome after seasonal influenza vaccination and influenza health-care encounters: a self-controlled study. Lancet Infect Dis 2013; 13(9):769–776. doi:10.1016/S1473-3099(13)70104-X
- Centers for Disease Control and Prevention (CDC). Disease burden of influenza. www.cdc.gov/flu/about/burden/index.html. Accessed November 13, 2019.
- Cowling BJ, Fang VJ, Nishiura H, et al. Increased risk of noninfluenza respiratory virus infections associated with receipt of inactivated influenza vaccine. Clin Infect Dis 2012; 54(12):1778–1783. doi:10.1093/cid/cis307
- Henle W, Henle G. Interference of inactive virus with the propagation of virus of influenza. Science 1943; 98(2534):87–89. doi:10.1126/science.98.2534.87
- Sundaram ME, McClure DL, VanWormer JJ, Friedrich TC, Meece JK, Belongia EA. Influenza vaccination is not associated with detection of noninfluenza respiratory viruses in seasonal studies of influenza vaccine effectiveness. Clin Infect Dis 2013; 57(6):789–793. doi:10.1093/cid/cit379
- Caubet JC, Wang J. Current understanding of egg allergy. Pediatr Clin North Am 2011; 58(2):427–443. doi:10.1016/j.pcl.2011.02.014
- Erlewyn-Lajeunesse M, Brathwaite N, Lucas JS, Warner JO. Recommendations for the administration of influenza vaccine in children allergic to egg. BMJ 2009; 339:b3680. doi:10.1136/bmj.b3680
- Ezeanolue E, Harriman K, Hunter P, Kroger A, Pellegrini C. General Best Practice Guidelines for Immunization. Best Practices Guidance of the Advisory Committee on Immunization Practices (ACIP). https://www.cdc.gov/vaccines/hcp/acip-recs/general-recs/downloads/general-recs.pdf. Accessed November 13, 2019.
- Centers for Disease Control and Prevention (CDC). Thimerosal in vaccines. www.cdc.gov/vaccinesafety/concerns/thimerosal/index.html. Accessed November 13, 2019.
- Hamilton JG. Needle phobia: a neglected diagnosis. J Fam Pract 1995; 41(2):169–175. pmid:7636457
- Vasileiou E, Sheikh A, Butler C, et al. Effectiveness of influenza vaccines in asthma: a systematic review and meta-analysis. Clin Infect Dis 2017; 65(8):1388–1395. doi:10.1093/cid/cix524
- Fomin I, Caspi D, Levy V, et al. Vaccination against influenza in rheumatoid arthritis: the effect of disease modifying drugs, including TNF alpha blockers. Ann Rheum Dis 2006; 65(2):191–194. doi:10.1136/ard.2005.036434
- Hurst FP, Lee JJ, Jindal RM, Agodoa LY, Abbott KC. Outcomes associated with influenza vaccination in the first year after kidney transplantation. Clin J Am Soc Nephrol 2011; 6(5):1192–1197. doi:10.2215/CJN.05430610
- Chong PP, Handler L, Weber DJ. A systematic review of safety and immunogenicity of influenza vaccination strategies in solid organ transplant recipients. Clin Infect Dis 2018; 66(11):1802–1811. doi:10.1093/cid/cix1081
- Ljungman P, Avetisyan G. Influenza vaccination in hematopoietic SCT recipients. Bone Marrow Transplant 2008; 42(10):637–641. doi:10.1038/bmt.2008.264
- Ohfuji S, Deguchi M, Tachibana D, et al; Osaka Pregnant Women Influenza Study Group. Protective effect of maternal influenza vaccination on influenza in their infants: a prospective cohort study. J Infect Dis 2018; 217(6):878–886. doi:10.1093/infdis/jix629
- Katz J, Englund JA, Steinhoff MC, et al. Impact of timing of influenza vaccination in pregnancy on transplacental antibody transfer, influenza incidence, and birth outcomes: a randomized trial in rural Nepal. Clin Infect Dis 2018; 67(3):334–340. doi:10.1093/cid/ciy090
- Nunes MC, Cutland CL, Madhi SA. Influenza vaccination during pregnancy and protection against pertussis. N Engl J Med 2018; 378(13):1257–1258. doi:10.1056/NEJMc1705208
- Centers for Disease Control and Prevention (CDC). Misconceptions about seasonal flu and flu vaccines. www.cdc.gov/flu/prevent/misconceptions.htm. Accessed November 13, 2019.
The benefits of influenza vaccination are clear to those in the medical community. Yet misinformation and unfounded fears continue to discourage some people from getting a flu shot. During the 2018–2019 influenza season, only 45% of US adults and 63% of children were vaccinated.1
‘IT DOESN’T WORK FOR MANY PEOPLE’
Multiple studies have shown that the flu vaccine prevents millions of flu cases and flu-related doctor’s visits each year. During the 2016–2017 flu season, flu vaccine prevented an estimated 5.3 million influenza cases, 2.6 million influenza-associated medical visits, and 85,000 influenza-associated hospitalizations.2
Several viral and host factors affect vaccine effectiveness. In seasons when the vaccine viruses have matched circulating strains, flu vaccine has been shown to reduce the following:
- The risk of having to go to the doctor with flu by 40% to 60%
- Children’s risk of flu-related death and intensive care unit (ICU) admission by 74%
- The risk in adults of flu-associated hospitalizations by 40% and ICU admission by 82%
- The rate of cardiac events in people with heart disease
- Hospitalizations in people with diabetes or underlying chronic lung disease.3
In people hospitalized with influenza despite receiving the flu vaccine for the season, studies have shown that receiving the flu vaccine shortens the average duration of hospitalization, reduces the chance of ICU admission by 59%, shortens the duration of ICU stay by 4 days, and reduces deaths.3
‘IT TARGETS THE WRONG VIRUS’
Selecting an effective influenza vaccine is a challenge. Every year, the World Health Organization and the CDC decide on the influenza strains expected to circulate in the upcoming flu season in the Northern Hemisphere, based on data for circulating strains in the Southern Hemisphere. This decision takes place about 7 months before the expected onset of the flu season. Flu viruses may mutate between the time the decision is made and the time the vaccine is administered (as well as after the flu season starts). Also, vaccine production in eggs needs time, which is why this decision must be made several months ahead of the flu season.
Vaccine effectiveness varies by virus serotype. Vaccines are typically less effective against influenza A H3N2 viruses than against influenza A H1N1 and influenza B viruses. Effectiveness also varies from season to season depending on how close the vaccine serotypes match the circulating serotypes, but some effectiveness is retained even in seasons when some of the serotypes don’t match circulating viruses. For example, in the 2017–2018 season, when the influenza A H3N2 vaccine serotype did not match the circulating serotype, the overall effectiveness in preventing medically attended, laboratory-confirmed influenza virus infection was 36%.5
A universal flu vaccine that does not need to be updated annually is the ultimate solution, but according to the National Institute of Allergy and Infectious Diseases, such a vaccine is likely several years away.6
‘IT MAKES PEOPLE SICK’
Pain at the injection site of a flu shot occurs in 10% to 65% of people, lasts less than 2 days, and does not usually interfere with daily activities.7
Systemic symptoms such as fever, malaise, and myalgia may occur in people who have had no previous exposure to the influenza virus antigens in the vaccine, particularly in children. In adults, the frequency of systemic symptoms after the flu shot is similar to that with placebo.
The Vaccine Adverse Event Reporting System, which has been capturing data since 1990, shows that the influenza vaccine accounted for 5.7% of people who developed malaise after receiving any vaccine.8
The injectable inactivated influenza vaccine cannot biologically cause an influenza virus-related illness, since the inactivated vaccine viruses can elicit a protective immune response but cannot replicate. The nasal live-attenuated flu vaccine can in theory cause acute illness in the person receiving it, but because it is cold-adapted, it multiplies only in the colder environment of the nasal epithelium, not in the lower airways where the temperature is higher. Consequently, the vaccine virus triggers immunity by multiplying in the nose, but doesn’t infect the lungs.
From 10% to 50% of people who receive the nasal live-attenuated vaccine develop runny nose, wheezing, headache, vomiting, muscle aches, fever, sore throat, or cough shortly after receiving the vaccine, but these symptoms are usually mild and short-lived.
The most common reactions people have to flu vaccines are considerably less severe than the symptoms caused by actual flu illness.
While influenza illness results in natural immunity to the specific viral serotype causing it, this illness results in hospitalization in 2% and is fatal in 0.16% of people. Influenza vaccine results in immunity to the serotypes included in the vaccine, and multiple studies have not found a causal relationship between vaccination and death.9
‘IT CAUSES GUILLAIN-BARRÉ SYNDROME’
In the United States, 3,000 to 6,000 people per year develop Guillain-Barré syndrome, or 1 to 2 of every 100,000, which translates to 80 to 160 cases per week.10 While the exact cause of Guillain-Barré syndrome is unknown, about two-thirds of people have an acute diarrheal or respiratory illness within 3 months before the onset of symptoms. In 1976, the estimated attributable risk of influenza vaccine-related Guillain-Barré syndrome in the US adult population was 1 case per 100,000 in the 6 weeks after vaccination.11 Studies in subsequent influenza seasons have not shown similar findings.12 In fact, one study showed that the risk of developing Guillain-Barré syndrome was 15 times higher after influenza illness than after influenza vaccination.13
Since 5% to 15% of the US population develop symptomatic influenza annually,14 the decision to vaccinate with respect to the risk of Guillain-Barré syndrome should be obvious: vaccinate. The correct question to ask before influenza vaccination should be, “Have you previously developed Guillain-Barré syndrome within 6 weeks after receiving the flu vaccine?” If the answer is yes, the CDC considers this a caution, not a contraindication against receiving the influenza vaccine, since the benefit may still outweigh the risk.
‘I GOT THE FLU SHOT AND STILL GOT SICK’
The flu vaccine does not prevent illnesses caused by other viruses or bacteria that can make people sick during flu season. Influenza, the common cold, and streptococcal pharyngitis can have similar symptoms that make it difficult for patients—and, frequently, even healthcare providers—to distinguish between these illnesses with certainty.
One study suggested that influenza vaccine recipients had an increased risk of virologically confirmed noninfluenza respiratory viral infections,15 citing the phenomenon of virus interference that was described in the 1940s16 as a potential explanation. In essence, people protected against influenza by the vaccine may lack temporary nonspecific immunity against other respiratory viruses. However, these findings have not been replicated in subsequent studies.17
Viral gastroenteritis, mistakenly called “stomach flu,” is also not prevented by influenza vaccination.
‘I’M ALLERGIC TO EGGS’
The prevalence of egg allergy in US children is 0.5% to 2.5%.18 Most outgrow it by school age, but in one-third, the allergy persists into adulthood.
In general, people who can eat lightly cooked eggs (eg, scrambled eggs) without a reaction are unlikely to be allergic. On the other hand, the fact that egg-allergic people may tolerate egg included in baked products does not exclude the possibility of egg allergy. Egg allergy can be confirmed by a consistent medical history of adverse reaction to eggs and egg-containing foods, in addition to skin or blood testing for immunoglobulin E directed against egg proteins.19
Most currently available influenza vaccines are prepared by propagation of virus in embryonated eggs and so may contain trace amounts of egg proteins such as ovalbumin, with the exception of the inactivated quadrivalent recombinant influenza vaccine (Flublok) and the inactivated quadrivalent cell culture-based vaccine (Flucelvax).
The ACIP recommends that persons with a history of urticaria (hives) after exposure to eggs should receive any licensed, recommended influenza vaccine that is otherwise appropriate for their age and health status. Persons who report having angioedema, respiratory distress, lightheadedness, or recurrent vomiting, or who required epinephrine or another emergency medical intervention after exposure to eggs, should receive the influenza vaccine in an inpatient or outpatient medical setting under the supervision of a healthcare provider who is able to recognize and manage severe allergic reactions.
A history of severe allergic reaction such as anaphylaxis to a previous dose of any influenza vaccine, regardless of the vaccine component (including eggs) suspected of being responsible for the reaction, is a contraindication to influenza vaccination. The ACIP recommends that vaccine providers consider observing patients for 15 minutes after administration of any vaccine (regardless of history of egg allergy) to decrease the risk of injury should syncope occur.20
‘I DON’T WANT TO PUT POISONOUS MERCURY IN MY BODY’
A process of biomagnification of methylmercury occurs when humans eat large fish that have eaten smaller fish. Thus, larger fish such as shark can be hazardous for women who are or may become pregnant, for nursing mothers, and for young children, while smaller fish such as herring are relatively safe.
As a precautionary measure, thimerosal was taken out of childhood vaccines in the United States in 2001. Thimerosal-free influenza vaccine formulations include the nasal live-attenuated flu vaccine, the inactivated quadrivalent recombinant influenza vaccine, and the inactivated quadrivalent cell culture-based vaccine.
‘I DON’T LIKE NEEDLES’
At least 10% of US adults have aichmophobia, the fear of sharp objects including needles.22 Vasovagal syncope is the most common manifestation. Behavioral therapy, topical anesthetics, and systemic anxiolytics have variable efficacy in treating needle phobia. For those who are absolutely averse to needles, the nasal flu vaccine is an appropriate alternative.
‘I DON’T WANT TO TAKE ANYTHING THAT CAN MESS WITH MY OTHER MEDICATIONS’
Some immunosuppressive medications may decrease influenza vaccine immunogenicity. Concomitant administration of the inactivated influenza vaccine with other vaccines is safe and does not alter immunogenicity of other vaccines.1 The live-attenuated influenza vaccine is contraindicated in children and adolescents taking aspirin or other salicylates due to the risk of Reye syndrome.
‘I’M AFRAID IT WILL TRIGGER AN IMMUNE RESPONSE THAT WILL MAKE MY ASTHMA WORSE’
A recent systematic review and meta-analysis showed that the inactivated influenza vaccine is not associated with asthma exacerbation.23 However, the nasal live-attenuated influenza vaccine is contraindicated in children 2 to 4 years old who have asthma and should be used with caution in persons with asthma 5 years old and older. In the systematic review, influenza vaccine prevented 59% to 78% of asthma attacks leading to emergency visits or hospitalization.23 In other immune-mediated diseases such as rheumatoid arthritis, influenza vaccine does not precipitate exacerbations.24
‘I HAD AN ORGAN TRANSPLANT, AND I’M AFRAID THE FLU SHOT WILL CAUSE ORGAN REJECTION’
A study of 51,730 kidney transplant recipients found that receipt of the inactivated influenza vaccine in the first year after transplant was associated with a lower risk of subsequent allograft loss (adjusted hazard ratio 0.77; 95% confidence interval 0.69–0.85; P < .001) and death (adjusted hazard ratio 0.82; 95% confidence interval 0.76–0.89; P < .001).25 In the same study, although acute rejection in the first year was not associated with influenza vaccination, influenza infection in the first year was associated with rejection (odds ratio 1.58; 95% confidence interval 1.10–2.26; P < 0.001), but not with graft loss or death. Solid organ transplant recipients should receive the inactivated influenza vaccine starting 3 months after transplant.26
Influenza vaccination has not been shown to precipitate graft-vs-host disease in hematopoietic stem cell transplant recipients. These patients should also receive the inactivated influenza vaccine starting 3 to 6 months after transplant.27
The nasal live-attenuated influenza vaccine is contraindicated in these immunocompromised patients.
‘I’M PREGNANT, AND I DON’T WANT TO EXPOSE MY UNBORN BABY TO ANYTHING POTENTIALLY HARMFUL’
The morbidity and mortality risk from influenza is high in children under 2 years old because of low immunogenicity to flu vaccine. This is particularly true in children younger than 6 months, but the vaccine is not recommended in this population. The best way to protect infants is for all household members to be vaccinated against the flu.
Equally important, morbidity and mortality risk from influenza is much higher in pregnant women than in the general population. Many studies have shown the value of influenza vaccination during pregnancy for both mothers and their infants. A recently published study showed that 18% of infants who developed influenza required hospitalization.28 In that study, prenatal and postpartum maternal influenza vaccination decreased the odds of influenza in infants by 61% and 53%, respectively. Another study showed that vaccine effectiveness did not vary by gestational age at vaccination.29 A post hoc analysis of an influenza vaccination study in pregnant women suggested that the vaccine was also associated with decreased rates of pertussis in these women.30
Healthcare providers should try to understand the public’s misconceptions31 about seasonal influenza and influenza vaccines in order to best address them.
The benefits of influenza vaccination are clear to those in the medical community. Yet misinformation and unfounded fears continue to discourage some people from getting a flu shot. During the 2018–2019 influenza season, only 45% of US adults and 63% of children were vaccinated.1
‘IT DOESN’T WORK FOR MANY PEOPLE’
Multiple studies have shown that the flu vaccine prevents millions of flu cases and flu-related doctor’s visits each year. During the 2016–2017 flu season, flu vaccine prevented an estimated 5.3 million influenza cases, 2.6 million influenza-associated medical visits, and 85,000 influenza-associated hospitalizations.2
Several viral and host factors affect vaccine effectiveness. In seasons when the vaccine viruses have matched circulating strains, flu vaccine has been shown to reduce the following:
- The risk of having to go to the doctor with flu by 40% to 60%
- Children’s risk of flu-related death and intensive care unit (ICU) admission by 74%
- The risk in adults of flu-associated hospitalizations by 40% and ICU admission by 82%
- The rate of cardiac events in people with heart disease
- Hospitalizations in people with diabetes or underlying chronic lung disease.3
In people hospitalized with influenza despite receiving the flu vaccine for the season, studies have shown that receiving the flu vaccine shortens the average duration of hospitalization, reduces the chance of ICU admission by 59%, shortens the duration of ICU stay by 4 days, and reduces deaths.3
‘IT TARGETS THE WRONG VIRUS’
Selecting an effective influenza vaccine is a challenge. Every year, the World Health Organization and the CDC decide on the influenza strains expected to circulate in the upcoming flu season in the Northern Hemisphere, based on data for circulating strains in the Southern Hemisphere. This decision takes place about 7 months before the expected onset of the flu season. Flu viruses may mutate between the time the decision is made and the time the vaccine is administered (as well as after the flu season starts). Also, vaccine production in eggs needs time, which is why this decision must be made several months ahead of the flu season.
Vaccine effectiveness varies by virus serotype. Vaccines are typically less effective against influenza A H3N2 viruses than against influenza A H1N1 and influenza B viruses. Effectiveness also varies from season to season depending on how close the vaccine serotypes match the circulating serotypes, but some effectiveness is retained even in seasons when some of the serotypes don’t match circulating viruses. For example, in the 2017–2018 season, when the influenza A H3N2 vaccine serotype did not match the circulating serotype, the overall effectiveness in preventing medically attended, laboratory-confirmed influenza virus infection was 36%.5
A universal flu vaccine that does not need to be updated annually is the ultimate solution, but according to the National Institute of Allergy and Infectious Diseases, such a vaccine is likely several years away.6
‘IT MAKES PEOPLE SICK’
Pain at the injection site of a flu shot occurs in 10% to 65% of people, lasts less than 2 days, and does not usually interfere with daily activities.7
Systemic symptoms such as fever, malaise, and myalgia may occur in people who have had no previous exposure to the influenza virus antigens in the vaccine, particularly in children. In adults, the frequency of systemic symptoms after the flu shot is similar to that with placebo.
The Vaccine Adverse Event Reporting System, which has been capturing data since 1990, shows that the influenza vaccine accounted for 5.7% of people who developed malaise after receiving any vaccine.8
The injectable inactivated influenza vaccine cannot biologically cause an influenza virus-related illness, since the inactivated vaccine viruses can elicit a protective immune response but cannot replicate. The nasal live-attenuated flu vaccine can in theory cause acute illness in the person receiving it, but because it is cold-adapted, it multiplies only in the colder environment of the nasal epithelium, not in the lower airways where the temperature is higher. Consequently, the vaccine virus triggers immunity by multiplying in the nose, but doesn’t infect the lungs.
From 10% to 50% of people who receive the nasal live-attenuated vaccine develop runny nose, wheezing, headache, vomiting, muscle aches, fever, sore throat, or cough shortly after receiving the vaccine, but these symptoms are usually mild and short-lived.
The most common reactions people have to flu vaccines are considerably less severe than the symptoms caused by actual flu illness.
While influenza illness results in natural immunity to the specific viral serotype causing it, this illness results in hospitalization in 2% and is fatal in 0.16% of people. Influenza vaccine results in immunity to the serotypes included in the vaccine, and multiple studies have not found a causal relationship between vaccination and death.9
‘IT CAUSES GUILLAIN-BARRÉ SYNDROME’
In the United States, 3,000 to 6,000 people per year develop Guillain-Barré syndrome, or 1 to 2 of every 100,000, which translates to 80 to 160 cases per week.10 While the exact cause of Guillain-Barré syndrome is unknown, about two-thirds of people have an acute diarrheal or respiratory illness within 3 months before the onset of symptoms. In 1976, the estimated attributable risk of influenza vaccine-related Guillain-Barré syndrome in the US adult population was 1 case per 100,000 in the 6 weeks after vaccination.11 Studies in subsequent influenza seasons have not shown similar findings.12 In fact, one study showed that the risk of developing Guillain-Barré syndrome was 15 times higher after influenza illness than after influenza vaccination.13
Since 5% to 15% of the US population develop symptomatic influenza annually,14 the decision to vaccinate with respect to the risk of Guillain-Barré syndrome should be obvious: vaccinate. The correct question to ask before influenza vaccination should be, “Have you previously developed Guillain-Barré syndrome within 6 weeks after receiving the flu vaccine?” If the answer is yes, the CDC considers this a caution, not a contraindication against receiving the influenza vaccine, since the benefit may still outweigh the risk.
‘I GOT THE FLU SHOT AND STILL GOT SICK’
The flu vaccine does not prevent illnesses caused by other viruses or bacteria that can make people sick during flu season. Influenza, the common cold, and streptococcal pharyngitis can have similar symptoms that make it difficult for patients—and, frequently, even healthcare providers—to distinguish between these illnesses with certainty.
One study suggested that influenza vaccine recipients had an increased risk of virologically confirmed noninfluenza respiratory viral infections,15 citing the phenomenon of virus interference that was described in the 1940s16 as a potential explanation. In essence, people protected against influenza by the vaccine may lack temporary nonspecific immunity against other respiratory viruses. However, these findings have not been replicated in subsequent studies.17
Viral gastroenteritis, mistakenly called “stomach flu,” is also not prevented by influenza vaccination.
‘I’M ALLERGIC TO EGGS’
The prevalence of egg allergy in US children is 0.5% to 2.5%.18 Most outgrow it by school age, but in one-third, the allergy persists into adulthood.
In general, people who can eat lightly cooked eggs (eg, scrambled eggs) without a reaction are unlikely to be allergic. On the other hand, the fact that egg-allergic people may tolerate egg included in baked products does not exclude the possibility of egg allergy. Egg allergy can be confirmed by a consistent medical history of adverse reaction to eggs and egg-containing foods, in addition to skin or blood testing for immunoglobulin E directed against egg proteins.19
Most currently available influenza vaccines are prepared by propagation of virus in embryonated eggs and so may contain trace amounts of egg proteins such as ovalbumin, with the exception of the inactivated quadrivalent recombinant influenza vaccine (Flublok) and the inactivated quadrivalent cell culture-based vaccine (Flucelvax).
The ACIP recommends that persons with a history of urticaria (hives) after exposure to eggs should receive any licensed, recommended influenza vaccine that is otherwise appropriate for their age and health status. Persons who report having angioedema, respiratory distress, lightheadedness, or recurrent vomiting, or who required epinephrine or another emergency medical intervention after exposure to eggs, should receive the influenza vaccine in an inpatient or outpatient medical setting under the supervision of a healthcare provider who is able to recognize and manage severe allergic reactions.
A history of severe allergic reaction such as anaphylaxis to a previous dose of any influenza vaccine, regardless of the vaccine component (including eggs) suspected of being responsible for the reaction, is a contraindication to influenza vaccination. The ACIP recommends that vaccine providers consider observing patients for 15 minutes after administration of any vaccine (regardless of history of egg allergy) to decrease the risk of injury should syncope occur.20
‘I DON’T WANT TO PUT POISONOUS MERCURY IN MY BODY’
A process of biomagnification of methylmercury occurs when humans eat large fish that have eaten smaller fish. Thus, larger fish such as shark can be hazardous for women who are or may become pregnant, for nursing mothers, and for young children, while smaller fish such as herring are relatively safe.
As a precautionary measure, thimerosal was taken out of childhood vaccines in the United States in 2001. Thimerosal-free influenza vaccine formulations include the nasal live-attenuated flu vaccine, the inactivated quadrivalent recombinant influenza vaccine, and the inactivated quadrivalent cell culture-based vaccine.
‘I DON’T LIKE NEEDLES’
At least 10% of US adults have aichmophobia, the fear of sharp objects including needles.22 Vasovagal syncope is the most common manifestation. Behavioral therapy, topical anesthetics, and systemic anxiolytics have variable efficacy in treating needle phobia. For those who are absolutely averse to needles, the nasal flu vaccine is an appropriate alternative.
‘I DON’T WANT TO TAKE ANYTHING THAT CAN MESS WITH MY OTHER MEDICATIONS’
Some immunosuppressive medications may decrease influenza vaccine immunogenicity. Concomitant administration of the inactivated influenza vaccine with other vaccines is safe and does not alter immunogenicity of other vaccines.1 The live-attenuated influenza vaccine is contraindicated in children and adolescents taking aspirin or other salicylates due to the risk of Reye syndrome.
‘I’M AFRAID IT WILL TRIGGER AN IMMUNE RESPONSE THAT WILL MAKE MY ASTHMA WORSE’
A recent systematic review and meta-analysis showed that the inactivated influenza vaccine is not associated with asthma exacerbation.23 However, the nasal live-attenuated influenza vaccine is contraindicated in children 2 to 4 years old who have asthma and should be used with caution in persons with asthma 5 years old and older. In the systematic review, influenza vaccine prevented 59% to 78% of asthma attacks leading to emergency visits or hospitalization.23 In other immune-mediated diseases such as rheumatoid arthritis, influenza vaccine does not precipitate exacerbations.24
‘I HAD AN ORGAN TRANSPLANT, AND I’M AFRAID THE FLU SHOT WILL CAUSE ORGAN REJECTION’
A study of 51,730 kidney transplant recipients found that receipt of the inactivated influenza vaccine in the first year after transplant was associated with a lower risk of subsequent allograft loss (adjusted hazard ratio 0.77; 95% confidence interval 0.69–0.85; P < .001) and death (adjusted hazard ratio 0.82; 95% confidence interval 0.76–0.89; P < .001).25 In the same study, although acute rejection in the first year was not associated with influenza vaccination, influenza infection in the first year was associated with rejection (odds ratio 1.58; 95% confidence interval 1.10–2.26; P < 0.001), but not with graft loss or death. Solid organ transplant recipients should receive the inactivated influenza vaccine starting 3 months after transplant.26
Influenza vaccination has not been shown to precipitate graft-vs-host disease in hematopoietic stem cell transplant recipients. These patients should also receive the inactivated influenza vaccine starting 3 to 6 months after transplant.27
The nasal live-attenuated influenza vaccine is contraindicated in these immunocompromised patients.
‘I’M PREGNANT, AND I DON’T WANT TO EXPOSE MY UNBORN BABY TO ANYTHING POTENTIALLY HARMFUL’
The morbidity and mortality risk from influenza is high in children under 2 years old because of low immunogenicity to flu vaccine. This is particularly true in children younger than 6 months, but the vaccine is not recommended in this population. The best way to protect infants is for all household members to be vaccinated against the flu.
Equally important, morbidity and mortality risk from influenza is much higher in pregnant women than in the general population. Many studies have shown the value of influenza vaccination during pregnancy for both mothers and their infants. A recently published study showed that 18% of infants who developed influenza required hospitalization.28 In that study, prenatal and postpartum maternal influenza vaccination decreased the odds of influenza in infants by 61% and 53%, respectively. Another study showed that vaccine effectiveness did not vary by gestational age at vaccination.29 A post hoc analysis of an influenza vaccination study in pregnant women suggested that the vaccine was also associated with decreased rates of pertussis in these women.30
Healthcare providers should try to understand the public’s misconceptions31 about seasonal influenza and influenza vaccines in order to best address them.
- Centers for Disease Control and Prevention (CDC). Flu vaccination coverage, United States, 2018–19 influenza season. www.cdc.gov/flu/fluvaxview/coverage-1819estimates.htm. Accessed November 13, 2019.
- Centers for Disease Control and Prevention (CDC). Immunogenicity, efficacy, and effectiveness of influenza vaccines. www.cdc.gov/flu/professionals/acip/immunogenicity.htm. Accessed November 13, 2019.
- Centers for Disease Control and Prevention (CDC). What are the benefits of flu vaccination? www.cdc.gov/flu/prevent/vaccine-benefits.htm. Accessed November 13, 2019.
- Grohskopf LA, Alyanak E, Broder KR, Walter EB, Fry AM, Jernigan DB. Prevention and control of seasonal influenza with vaccines: recommendations of the Advisory Committee on Immunization Practices—United States, 2019–20 influenza season. MMWR Recomm Rep 2019; 68(3):1–21. doi:10.15585/mmwr.rr6803a1
- Flannery B, Chung JR, Belongia EA, et al. Interim estimates of 2017–18 seasonal influenza vaccine effectiveness—United States, February 2018. MMWR Morb Mortal Wkly Rep 2018; 67(6):180–185. doi:10.15585/mmwr.mm6706a2
- Erbelding EJ, Post DJ, Stemmy EJ, et al. A universal influenza vaccine: the strategic plan for the National Institute of Allergy and Infectious Diseases. J Infect Dis 2018; 218(3):347–354. doi:10.1093/infdis/jiy103
- Centers for Disease Control and Prevention (CDC). Seasonal influenza vaccine safety: a summary for clinicians. www.cdc.gov/flu/professionals/vaccination/vaccine_safety.htm. Accessed November 13, 2019.
- Centers for Disease Control and Prevention (CDC). About the Vaccine Adverse Event Reporting System (VAERS). https://wonder.cdc.gov/vaers.html. Accessed November 13, 2019.
- Miller ER, Moro PL, Cano M, Shimabukuro TT. Deaths following vaccination: what does the evidence show? Vaccine 2015; 33(29):3288–3292. doi:10.1016/j.vaccine.2015.05.023
- Centers for Disease Control and Prevention (CDC). Guillain-Barré syndrome and flu vaccine. www.cdc.gov/flu/prevent/guillainbarre.htm. Accessed November 13, 2019.
- Schonberger LB, Bregman DJ, Sullivan-Bolyai JZ, et al. Guillain-Barre syndrome following vaccination in the national influenza immunization program, United States, 1976–1977. Am J Epidemiol 1979; 110(2):105–123. doi:10.1093/oxfordjournals.aje.a112795
- Baxter R, Bakshi N, Fireman B, et al. Lack of association of Guillain-Barré syndrome with vaccinations. Clin Infect Dis 2013; 57(2):197–204. doi:10.1093/cid/cit222
- Kwong JC, Vasa PP, Campitelli MA, et al. Risk of Guillain-Barré syndrome after seasonal influenza vaccination and influenza health-care encounters: a self-controlled study. Lancet Infect Dis 2013; 13(9):769–776. doi:10.1016/S1473-3099(13)70104-X
- Centers for Disease Control and Prevention (CDC). Disease burden of influenza. www.cdc.gov/flu/about/burden/index.html. Accessed November 13, 2019.
- Cowling BJ, Fang VJ, Nishiura H, et al. Increased risk of noninfluenza respiratory virus infections associated with receipt of inactivated influenza vaccine. Clin Infect Dis 2012; 54(12):1778–1783. doi:10.1093/cid/cis307
- Henle W, Henle G. Interference of inactive virus with the propagation of virus of influenza. Science 1943; 98(2534):87–89. doi:10.1126/science.98.2534.87
- Sundaram ME, McClure DL, VanWormer JJ, Friedrich TC, Meece JK, Belongia EA. Influenza vaccination is not associated with detection of noninfluenza respiratory viruses in seasonal studies of influenza vaccine effectiveness. Clin Infect Dis 2013; 57(6):789–793. doi:10.1093/cid/cit379
- Caubet JC, Wang J. Current understanding of egg allergy. Pediatr Clin North Am 2011; 58(2):427–443. doi:10.1016/j.pcl.2011.02.014
- Erlewyn-Lajeunesse M, Brathwaite N, Lucas JS, Warner JO. Recommendations for the administration of influenza vaccine in children allergic to egg. BMJ 2009; 339:b3680. doi:10.1136/bmj.b3680
- Ezeanolue E, Harriman K, Hunter P, Kroger A, Pellegrini C. General Best Practice Guidelines for Immunization. Best Practices Guidance of the Advisory Committee on Immunization Practices (ACIP). https://www.cdc.gov/vaccines/hcp/acip-recs/general-recs/downloads/general-recs.pdf. Accessed November 13, 2019.
- Centers for Disease Control and Prevention (CDC). Thimerosal in vaccines. www.cdc.gov/vaccinesafety/concerns/thimerosal/index.html. Accessed November 13, 2019.
- Hamilton JG. Needle phobia: a neglected diagnosis. J Fam Pract 1995; 41(2):169–175. pmid:7636457
- Vasileiou E, Sheikh A, Butler C, et al. Effectiveness of influenza vaccines in asthma: a systematic review and meta-analysis. Clin Infect Dis 2017; 65(8):1388–1395. doi:10.1093/cid/cix524
- Fomin I, Caspi D, Levy V, et al. Vaccination against influenza in rheumatoid arthritis: the effect of disease modifying drugs, including TNF alpha blockers. Ann Rheum Dis 2006; 65(2):191–194. doi:10.1136/ard.2005.036434
- Hurst FP, Lee JJ, Jindal RM, Agodoa LY, Abbott KC. Outcomes associated with influenza vaccination in the first year after kidney transplantation. Clin J Am Soc Nephrol 2011; 6(5):1192–1197. doi:10.2215/CJN.05430610
- Chong PP, Handler L, Weber DJ. A systematic review of safety and immunogenicity of influenza vaccination strategies in solid organ transplant recipients. Clin Infect Dis 2018; 66(11):1802–1811. doi:10.1093/cid/cix1081
- Ljungman P, Avetisyan G. Influenza vaccination in hematopoietic SCT recipients. Bone Marrow Transplant 2008; 42(10):637–641. doi:10.1038/bmt.2008.264
- Ohfuji S, Deguchi M, Tachibana D, et al; Osaka Pregnant Women Influenza Study Group. Protective effect of maternal influenza vaccination on influenza in their infants: a prospective cohort study. J Infect Dis 2018; 217(6):878–886. doi:10.1093/infdis/jix629
- Katz J, Englund JA, Steinhoff MC, et al. Impact of timing of influenza vaccination in pregnancy on transplacental antibody transfer, influenza incidence, and birth outcomes: a randomized trial in rural Nepal. Clin Infect Dis 2018; 67(3):334–340. doi:10.1093/cid/ciy090
- Nunes MC, Cutland CL, Madhi SA. Influenza vaccination during pregnancy and protection against pertussis. N Engl J Med 2018; 378(13):1257–1258. doi:10.1056/NEJMc1705208
- Centers for Disease Control and Prevention (CDC). Misconceptions about seasonal flu and flu vaccines. www.cdc.gov/flu/prevent/misconceptions.htm. Accessed November 13, 2019.
- Centers for Disease Control and Prevention (CDC). Flu vaccination coverage, United States, 2018–19 influenza season. www.cdc.gov/flu/fluvaxview/coverage-1819estimates.htm. Accessed November 13, 2019.
- Centers for Disease Control and Prevention (CDC). Immunogenicity, efficacy, and effectiveness of influenza vaccines. www.cdc.gov/flu/professionals/acip/immunogenicity.htm. Accessed November 13, 2019.
- Centers for Disease Control and Prevention (CDC). What are the benefits of flu vaccination? www.cdc.gov/flu/prevent/vaccine-benefits.htm. Accessed November 13, 2019.
- Grohskopf LA, Alyanak E, Broder KR, Walter EB, Fry AM, Jernigan DB. Prevention and control of seasonal influenza with vaccines: recommendations of the Advisory Committee on Immunization Practices—United States, 2019–20 influenza season. MMWR Recomm Rep 2019; 68(3):1–21. doi:10.15585/mmwr.rr6803a1
- Flannery B, Chung JR, Belongia EA, et al. Interim estimates of 2017–18 seasonal influenza vaccine effectiveness—United States, February 2018. MMWR Morb Mortal Wkly Rep 2018; 67(6):180–185. doi:10.15585/mmwr.mm6706a2
- Erbelding EJ, Post DJ, Stemmy EJ, et al. A universal influenza vaccine: the strategic plan for the National Institute of Allergy and Infectious Diseases. J Infect Dis 2018; 218(3):347–354. doi:10.1093/infdis/jiy103
- Centers for Disease Control and Prevention (CDC). Seasonal influenza vaccine safety: a summary for clinicians. www.cdc.gov/flu/professionals/vaccination/vaccine_safety.htm. Accessed November 13, 2019.
- Centers for Disease Control and Prevention (CDC). About the Vaccine Adverse Event Reporting System (VAERS). https://wonder.cdc.gov/vaers.html. Accessed November 13, 2019.
- Miller ER, Moro PL, Cano M, Shimabukuro TT. Deaths following vaccination: what does the evidence show? Vaccine 2015; 33(29):3288–3292. doi:10.1016/j.vaccine.2015.05.023
- Centers for Disease Control and Prevention (CDC). Guillain-Barré syndrome and flu vaccine. www.cdc.gov/flu/prevent/guillainbarre.htm. Accessed November 13, 2019.
- Schonberger LB, Bregman DJ, Sullivan-Bolyai JZ, et al. Guillain-Barre syndrome following vaccination in the national influenza immunization program, United States, 1976–1977. Am J Epidemiol 1979; 110(2):105–123. doi:10.1093/oxfordjournals.aje.a112795
- Baxter R, Bakshi N, Fireman B, et al. Lack of association of Guillain-Barré syndrome with vaccinations. Clin Infect Dis 2013; 57(2):197–204. doi:10.1093/cid/cit222
- Kwong JC, Vasa PP, Campitelli MA, et al. Risk of Guillain-Barré syndrome after seasonal influenza vaccination and influenza health-care encounters: a self-controlled study. Lancet Infect Dis 2013; 13(9):769–776. doi:10.1016/S1473-3099(13)70104-X
- Centers for Disease Control and Prevention (CDC). Disease burden of influenza. www.cdc.gov/flu/about/burden/index.html. Accessed November 13, 2019.
- Cowling BJ, Fang VJ, Nishiura H, et al. Increased risk of noninfluenza respiratory virus infections associated with receipt of inactivated influenza vaccine. Clin Infect Dis 2012; 54(12):1778–1783. doi:10.1093/cid/cis307
- Henle W, Henle G. Interference of inactive virus with the propagation of virus of influenza. Science 1943; 98(2534):87–89. doi:10.1126/science.98.2534.87
- Sundaram ME, McClure DL, VanWormer JJ, Friedrich TC, Meece JK, Belongia EA. Influenza vaccination is not associated with detection of noninfluenza respiratory viruses in seasonal studies of influenza vaccine effectiveness. Clin Infect Dis 2013; 57(6):789–793. doi:10.1093/cid/cit379
- Caubet JC, Wang J. Current understanding of egg allergy. Pediatr Clin North Am 2011; 58(2):427–443. doi:10.1016/j.pcl.2011.02.014
- Erlewyn-Lajeunesse M, Brathwaite N, Lucas JS, Warner JO. Recommendations for the administration of influenza vaccine in children allergic to egg. BMJ 2009; 339:b3680. doi:10.1136/bmj.b3680
- Ezeanolue E, Harriman K, Hunter P, Kroger A, Pellegrini C. General Best Practice Guidelines for Immunization. Best Practices Guidance of the Advisory Committee on Immunization Practices (ACIP). https://www.cdc.gov/vaccines/hcp/acip-recs/general-recs/downloads/general-recs.pdf. Accessed November 13, 2019.
- Centers for Disease Control and Prevention (CDC). Thimerosal in vaccines. www.cdc.gov/vaccinesafety/concerns/thimerosal/index.html. Accessed November 13, 2019.
- Hamilton JG. Needle phobia: a neglected diagnosis. J Fam Pract 1995; 41(2):169–175. pmid:7636457
- Vasileiou E, Sheikh A, Butler C, et al. Effectiveness of influenza vaccines in asthma: a systematic review and meta-analysis. Clin Infect Dis 2017; 65(8):1388–1395. doi:10.1093/cid/cix524
- Fomin I, Caspi D, Levy V, et al. Vaccination against influenza in rheumatoid arthritis: the effect of disease modifying drugs, including TNF alpha blockers. Ann Rheum Dis 2006; 65(2):191–194. doi:10.1136/ard.2005.036434
- Hurst FP, Lee JJ, Jindal RM, Agodoa LY, Abbott KC. Outcomes associated with influenza vaccination in the first year after kidney transplantation. Clin J Am Soc Nephrol 2011; 6(5):1192–1197. doi:10.2215/CJN.05430610
- Chong PP, Handler L, Weber DJ. A systematic review of safety and immunogenicity of influenza vaccination strategies in solid organ transplant recipients. Clin Infect Dis 2018; 66(11):1802–1811. doi:10.1093/cid/cix1081
- Ljungman P, Avetisyan G. Influenza vaccination in hematopoietic SCT recipients. Bone Marrow Transplant 2008; 42(10):637–641. doi:10.1038/bmt.2008.264
- Ohfuji S, Deguchi M, Tachibana D, et al; Osaka Pregnant Women Influenza Study Group. Protective effect of maternal influenza vaccination on influenza in their infants: a prospective cohort study. J Infect Dis 2018; 217(6):878–886. doi:10.1093/infdis/jix629
- Katz J, Englund JA, Steinhoff MC, et al. Impact of timing of influenza vaccination in pregnancy on transplacental antibody transfer, influenza incidence, and birth outcomes: a randomized trial in rural Nepal. Clin Infect Dis 2018; 67(3):334–340. doi:10.1093/cid/ciy090
- Nunes MC, Cutland CL, Madhi SA. Influenza vaccination during pregnancy and protection against pertussis. N Engl J Med 2018; 378(13):1257–1258. doi:10.1056/NEJMc1705208
- Centers for Disease Control and Prevention (CDC). Misconceptions about seasonal flu and flu vaccines. www.cdc.gov/flu/prevent/misconceptions.htm. Accessed November 13, 2019.
Click for Credit: PPI use & dementia; Weight loss after gastroplasty; more
Here are 5 articles from the December issue of Clinician Reviews (individual articles are valid for one year from date of publication—expiration dates below):
1. Sustainable weight loss seen 5 years after endoscopic sleeve gastroplasty
To take the posttest, go to: https://bit.ly/37lteRX
Expires May 16, 2020
2. PT beats steroid injections for knee OA
To take the posttest, go to: https://bit.ly/2KIWKY6
Expires May 17, 2020
3. Better screening needed to reduce pregnancy-related overdose, death
To take the posttest, go to: https://bit.ly/2XEZyuG
Expires May 17, 2020
4. Meta-analysis finds no link between PPI use and risk of dementia
To take the posttest, go to: https://bit.ly/2Xzs7JM
Expires June 3, 2020
5. Study: Cardiac biomarkers predicted CV events in CAP
To take the posttest, go to: https://bit.ly/33bAH2u
Expires August 13, 2020
Here are 5 articles from the December issue of Clinician Reviews (individual articles are valid for one year from date of publication—expiration dates below):
1. Sustainable weight loss seen 5 years after endoscopic sleeve gastroplasty
To take the posttest, go to: https://bit.ly/37lteRX
Expires May 16, 2020
2. PT beats steroid injections for knee OA
To take the posttest, go to: https://bit.ly/2KIWKY6
Expires May 17, 2020
3. Better screening needed to reduce pregnancy-related overdose, death
To take the posttest, go to: https://bit.ly/2XEZyuG
Expires May 17, 2020
4. Meta-analysis finds no link between PPI use and risk of dementia
To take the posttest, go to: https://bit.ly/2Xzs7JM
Expires June 3, 2020
5. Study: Cardiac biomarkers predicted CV events in CAP
To take the posttest, go to: https://bit.ly/33bAH2u
Expires August 13, 2020
Here are 5 articles from the December issue of Clinician Reviews (individual articles are valid for one year from date of publication—expiration dates below):
1. Sustainable weight loss seen 5 years after endoscopic sleeve gastroplasty
To take the posttest, go to: https://bit.ly/37lteRX
Expires May 16, 2020
2. PT beats steroid injections for knee OA
To take the posttest, go to: https://bit.ly/2KIWKY6
Expires May 17, 2020
3. Better screening needed to reduce pregnancy-related overdose, death
To take the posttest, go to: https://bit.ly/2XEZyuG
Expires May 17, 2020
4. Meta-analysis finds no link between PPI use and risk of dementia
To take the posttest, go to: https://bit.ly/2Xzs7JM
Expires June 3, 2020
5. Study: Cardiac biomarkers predicted CV events in CAP
To take the posttest, go to: https://bit.ly/33bAH2u
Expires August 13, 2020