User login
NIH seeks gene-based cures for HIV, sickle cell disease
The National Institutes of Health and the Bill & Melinda Gates Foundation have announced that they plan to invest $100 million each over the next 4 years to develop affordable, gene-based cures for sickle cell disease (SCD) and HIV.
The initiative follows an announcement from President Trump that set a goal of ending the HIV epidemic in the United States in the next 10 years, seeking to reduce the number of diagnoses by 90% by 2030. The Trump administration has also identified SCD as an “intractable health challenge with the potential for dramatic advances in the coming years,” the NIH said in a statement.
Gene-based therapy has become a reality in recent years thanks to dramatic advances, but the cost is prohibitive in many parts of the world. “The collaboration between the NIH and the Gates Foundation sets out a bold goal of advancing safe, effective, and durable gene-based cures to clinical trials in the United States and relevant countries in sub-Saharan Africa within the next 7-10 years. The ultimate goal is to scale and implement these treatments globally in areas hardest hit by these diseases,” the NIH said.
Both diseases are a significant burden on low- and middle-income countries, as 95% of the 38 million people living with HIV globally are in the developing world, with 67% living in sub-Saharan Africa; about half of the HIV-infected population receives no treatment for the disease. An estimated 15 million children will be born with SCD over the next 30 years, with three-quarters of those births occurring in sub-Saharan Africa. About 50%-90% of children born with SCD will die before age 5 years.
The collaboration will focus on coordination in two areas: identifying potential candidate cures for SCD and HIV for preclinical and clinical evaluation, and defining long-term opportunities to work together and with African partners on advancing promising candidates to late-phase clinical trials, with funding to be determined as candidates progress.
“In recent years, gene-based treatments have been groundbreaking for rare genetic disorders and infectious diseases. While these treatments are exciting, people in low- and middle-income countries do not have access to these breakthroughs. By working with the NIH and scientists across Africa, we aim to ensure these approaches will improve the lives of those most in need and bring the incredible promise of gene-based treatments to the world of public health,” said Trevor Mundel, MD, PhD, president of the global health program at the Bill & Melinda Gates Foundation.
The National Institutes of Health and the Bill & Melinda Gates Foundation have announced that they plan to invest $100 million each over the next 4 years to develop affordable, gene-based cures for sickle cell disease (SCD) and HIV.
The initiative follows an announcement from President Trump that set a goal of ending the HIV epidemic in the United States in the next 10 years, seeking to reduce the number of diagnoses by 90% by 2030. The Trump administration has also identified SCD as an “intractable health challenge with the potential for dramatic advances in the coming years,” the NIH said in a statement.
Gene-based therapy has become a reality in recent years thanks to dramatic advances, but the cost is prohibitive in many parts of the world. “The collaboration between the NIH and the Gates Foundation sets out a bold goal of advancing safe, effective, and durable gene-based cures to clinical trials in the United States and relevant countries in sub-Saharan Africa within the next 7-10 years. The ultimate goal is to scale and implement these treatments globally in areas hardest hit by these diseases,” the NIH said.
Both diseases are a significant burden on low- and middle-income countries, as 95% of the 38 million people living with HIV globally are in the developing world, with 67% living in sub-Saharan Africa; about half of the HIV-infected population receives no treatment for the disease. An estimated 15 million children will be born with SCD over the next 30 years, with three-quarters of those births occurring in sub-Saharan Africa. About 50%-90% of children born with SCD will die before age 5 years.
The collaboration will focus on coordination in two areas: identifying potential candidate cures for SCD and HIV for preclinical and clinical evaluation, and defining long-term opportunities to work together and with African partners on advancing promising candidates to late-phase clinical trials, with funding to be determined as candidates progress.
“In recent years, gene-based treatments have been groundbreaking for rare genetic disorders and infectious diseases. While these treatments are exciting, people in low- and middle-income countries do not have access to these breakthroughs. By working with the NIH and scientists across Africa, we aim to ensure these approaches will improve the lives of those most in need and bring the incredible promise of gene-based treatments to the world of public health,” said Trevor Mundel, MD, PhD, president of the global health program at the Bill & Melinda Gates Foundation.
The National Institutes of Health and the Bill & Melinda Gates Foundation have announced that they plan to invest $100 million each over the next 4 years to develop affordable, gene-based cures for sickle cell disease (SCD) and HIV.
The initiative follows an announcement from President Trump that set a goal of ending the HIV epidemic in the United States in the next 10 years, seeking to reduce the number of diagnoses by 90% by 2030. The Trump administration has also identified SCD as an “intractable health challenge with the potential for dramatic advances in the coming years,” the NIH said in a statement.
Gene-based therapy has become a reality in recent years thanks to dramatic advances, but the cost is prohibitive in many parts of the world. “The collaboration between the NIH and the Gates Foundation sets out a bold goal of advancing safe, effective, and durable gene-based cures to clinical trials in the United States and relevant countries in sub-Saharan Africa within the next 7-10 years. The ultimate goal is to scale and implement these treatments globally in areas hardest hit by these diseases,” the NIH said.
Both diseases are a significant burden on low- and middle-income countries, as 95% of the 38 million people living with HIV globally are in the developing world, with 67% living in sub-Saharan Africa; about half of the HIV-infected population receives no treatment for the disease. An estimated 15 million children will be born with SCD over the next 30 years, with three-quarters of those births occurring in sub-Saharan Africa. About 50%-90% of children born with SCD will die before age 5 years.
The collaboration will focus on coordination in two areas: identifying potential candidate cures for SCD and HIV for preclinical and clinical evaluation, and defining long-term opportunities to work together and with African partners on advancing promising candidates to late-phase clinical trials, with funding to be determined as candidates progress.
“In recent years, gene-based treatments have been groundbreaking for rare genetic disorders and infectious diseases. While these treatments are exciting, people in low- and middle-income countries do not have access to these breakthroughs. By working with the NIH and scientists across Africa, we aim to ensure these approaches will improve the lives of those most in need and bring the incredible promise of gene-based treatments to the world of public health,” said Trevor Mundel, MD, PhD, president of the global health program at the Bill & Melinda Gates Foundation.
Fibrinogen concentrate effective, safe for postop bleeding
SAN ANTONIO – Fibrinogen concentrate was noninferior to cryoprecipitate for controlling bleeding following cardiac surgery in the randomized FIBRES trial, Canadian investigators reported.
Among 827 patients undergoing cardiopulmonary bypass, there were no significant differences in the use of allogenenic transfusion products within 24 hours of surgery for patients assigned to receive fibrinogen concentrate for control of bleeding, compared with patients who received cryoprecipitate, reported Jeannie Callum, MD, from Sunnybrook Health Sciences Centre in Toronto, on behalf of coinvestigators in the FIBRES trial.
Fibrinogen concentrate, commonly used to control postoperative bleeding in Europe, was associated with numerically, but not statistically, lower incidence of both adverse events and serious adverse events than cryoprecipitate, the current standard of care in North America.
“Given its safety and logistical advantages, fibrinogen concentrate may be considered in bleeding patients with acquired hypofibrinogenemia,” Dr. Callum said at the annual meeting of the AABB, the group formerly known as the American Association of Blood Banks.
Results of the FIBRES trial were published simultaneously in JAMA (2019 Oct 21. doi: 10.1001/jama.2019.17312).
Acquired hypofibrinogenemia, defined as a fibrinogen level below the range of 1.5-2.0 g/L, is a major cause of excess bleeding after cardiac surgery. European guidelines on the management of bleeding following trauma or cardiac surgery recommend the use of either cryoprecipitate or fibrinogen concentrate to control excessive bleeding in patients with acquired hypofibrinogenemia, Dr. Callum noted.
Cryoprecipitate is a pooled plasma–derived product that contains fibrinogen, but also fibronectin, platelet microparticles, coagulation factors VIII and XIII, and von Willebrand factor.
Additionally, fibrinogen levels in cryoprecipitate can range from as low as 3 g/L to as high as 30 g/L, and the product is normally kept and shipped frozen, and is then thawed for use and pooled prior to administration, with a shelf life of just 4-6 hours.
In contrast, fibrinogen concentrates “are pathogen-reduced and purified; have standardized fibrinogen content (20 g/L); are lyophilized, allowing for easy storage, reconstitution, and administration; and have longer shelf life after reconstitution (up to 24 hours),” Dr. Callum and her colleagues reported.
Despite the North American preference for cryoprecipitate and the European preference for fibrinogen concentrate, there have been few studies directly comparing the two products, which prompted the FIBRES investigators to design a head-to-head trial.
The randomized trial was conducted in 11 Canadian hospitals with adults undergoing cardiac surgery with cardiopulmonary bypass for whom fibrinogen supplementation was ordered in accordance with accepted clinical standards.
Patients were randomly assigned to received either 4 g of fibrinogen concentrate or 10 units of cryoprecipitate for 24 hours, with all patients receiving additional cryoprecipitate as needed after the first day.
Of 15,412 cardiac patients treated at the participating sites, 827 patients met the trial criteria and were randomized. Because the trial met the prespecified stopping criterion for noninferiority of fibrinogen at the interim analysis, the trial was halted, leaving the 827 patients as the final analysis population.
The mean number of allogeneic blood component units administered – the primary outcome – was 16.3 units in the fibrinogen concentrate group and 17.0 units in the cryoprecipitate group (mean ratio, 0.96; P for noninferiority less than .001; P for superiority = .50).
Fibrinogen was also noninferior for the secondary outcomes of individual 24-hour and cumulative 7-day blood component transfusions, and in a post-hoc analysis of cumulative transfusions measured from product administration to 24 hours after termination of cardiopulmonary bypass. These endpoints should be interpreted with caution, however, because they were not corrected for type 1 error, the investigators noted.
Fibrinogen concentrate also appeared to be noninferior for all defined subgroups, except for patients who underwent nonelective procedures, which included all patients in critical state before surgery.
Adverse events (AEs) of any kind occurred in 66.7% of patients with fibrinogen concentrate vs. 72.7% of those on cryoprecipitate. Serious AEs occurred in 31.5% vs. 34.7%, respectively.
Thromboembolic events – stroke or transient ischemic attack, amaurosis fugax (temporary vision loss), myocardial infarction, deep-vein thrombosis, pulmonary embolism, other-vessel thrombosis, disseminated intravascular coagulation, or thrombophlebitis – occurred in 7% vs. 9.6%, respectively.
The investigators acknowledged that the study was limited by the inability to blind the clinical team to the product used, by the adult-only population, and by the likelihood of variable dosing in the cryoprecipitate group.
Advantages of fibrinogen concentrate over cryoprecipitate are that the former is pathogen reduced and is easier to deliver, the investigators said.
“One important consideration is the cost differential that currently favors cryoprecipitate, but this varies across regions, and the most recent economic analysis failed to include the costs of future emerging pathogens and did not include comprehensive activity-based costing,” the investigators wrote in JAMA.
The trial was sponsored by Octapharma AG, which also provided fibrinogen concentrate. Cryoprecipitate was provided by the Canadian Blood Services and Héma-Québec. Dr. Callum reported receiving grants from Canadian Blood Services, Octapharma, and CSL Behring during the conduct of the study. Multiple coauthors had similar disclosures.
SOURCE: Callum J et al. JAMA. 2019 Oct 21. doi:10.1001/jama.2019.17312.
SAN ANTONIO – Fibrinogen concentrate was noninferior to cryoprecipitate for controlling bleeding following cardiac surgery in the randomized FIBRES trial, Canadian investigators reported.
Among 827 patients undergoing cardiopulmonary bypass, there were no significant differences in the use of allogenenic transfusion products within 24 hours of surgery for patients assigned to receive fibrinogen concentrate for control of bleeding, compared with patients who received cryoprecipitate, reported Jeannie Callum, MD, from Sunnybrook Health Sciences Centre in Toronto, on behalf of coinvestigators in the FIBRES trial.
Fibrinogen concentrate, commonly used to control postoperative bleeding in Europe, was associated with numerically, but not statistically, lower incidence of both adverse events and serious adverse events than cryoprecipitate, the current standard of care in North America.
“Given its safety and logistical advantages, fibrinogen concentrate may be considered in bleeding patients with acquired hypofibrinogenemia,” Dr. Callum said at the annual meeting of the AABB, the group formerly known as the American Association of Blood Banks.
Results of the FIBRES trial were published simultaneously in JAMA (2019 Oct 21. doi: 10.1001/jama.2019.17312).
Acquired hypofibrinogenemia, defined as a fibrinogen level below the range of 1.5-2.0 g/L, is a major cause of excess bleeding after cardiac surgery. European guidelines on the management of bleeding following trauma or cardiac surgery recommend the use of either cryoprecipitate or fibrinogen concentrate to control excessive bleeding in patients with acquired hypofibrinogenemia, Dr. Callum noted.
Cryoprecipitate is a pooled plasma–derived product that contains fibrinogen, but also fibronectin, platelet microparticles, coagulation factors VIII and XIII, and von Willebrand factor.
Additionally, fibrinogen levels in cryoprecipitate can range from as low as 3 g/L to as high as 30 g/L, and the product is normally kept and shipped frozen, and is then thawed for use and pooled prior to administration, with a shelf life of just 4-6 hours.
In contrast, fibrinogen concentrates “are pathogen-reduced and purified; have standardized fibrinogen content (20 g/L); are lyophilized, allowing for easy storage, reconstitution, and administration; and have longer shelf life after reconstitution (up to 24 hours),” Dr. Callum and her colleagues reported.
Despite the North American preference for cryoprecipitate and the European preference for fibrinogen concentrate, there have been few studies directly comparing the two products, which prompted the FIBRES investigators to design a head-to-head trial.
The randomized trial was conducted in 11 Canadian hospitals with adults undergoing cardiac surgery with cardiopulmonary bypass for whom fibrinogen supplementation was ordered in accordance with accepted clinical standards.
Patients were randomly assigned to received either 4 g of fibrinogen concentrate or 10 units of cryoprecipitate for 24 hours, with all patients receiving additional cryoprecipitate as needed after the first day.
Of 15,412 cardiac patients treated at the participating sites, 827 patients met the trial criteria and were randomized. Because the trial met the prespecified stopping criterion for noninferiority of fibrinogen at the interim analysis, the trial was halted, leaving the 827 patients as the final analysis population.
The mean number of allogeneic blood component units administered – the primary outcome – was 16.3 units in the fibrinogen concentrate group and 17.0 units in the cryoprecipitate group (mean ratio, 0.96; P for noninferiority less than .001; P for superiority = .50).
Fibrinogen was also noninferior for the secondary outcomes of individual 24-hour and cumulative 7-day blood component transfusions, and in a post-hoc analysis of cumulative transfusions measured from product administration to 24 hours after termination of cardiopulmonary bypass. These endpoints should be interpreted with caution, however, because they were not corrected for type 1 error, the investigators noted.
Fibrinogen concentrate also appeared to be noninferior for all defined subgroups, except for patients who underwent nonelective procedures, which included all patients in critical state before surgery.
Adverse events (AEs) of any kind occurred in 66.7% of patients with fibrinogen concentrate vs. 72.7% of those on cryoprecipitate. Serious AEs occurred in 31.5% vs. 34.7%, respectively.
Thromboembolic events – stroke or transient ischemic attack, amaurosis fugax (temporary vision loss), myocardial infarction, deep-vein thrombosis, pulmonary embolism, other-vessel thrombosis, disseminated intravascular coagulation, or thrombophlebitis – occurred in 7% vs. 9.6%, respectively.
The investigators acknowledged that the study was limited by the inability to blind the clinical team to the product used, by the adult-only population, and by the likelihood of variable dosing in the cryoprecipitate group.
Advantages of fibrinogen concentrate over cryoprecipitate are that the former is pathogen reduced and is easier to deliver, the investigators said.
“One important consideration is the cost differential that currently favors cryoprecipitate, but this varies across regions, and the most recent economic analysis failed to include the costs of future emerging pathogens and did not include comprehensive activity-based costing,” the investigators wrote in JAMA.
The trial was sponsored by Octapharma AG, which also provided fibrinogen concentrate. Cryoprecipitate was provided by the Canadian Blood Services and Héma-Québec. Dr. Callum reported receiving grants from Canadian Blood Services, Octapharma, and CSL Behring during the conduct of the study. Multiple coauthors had similar disclosures.
SOURCE: Callum J et al. JAMA. 2019 Oct 21. doi:10.1001/jama.2019.17312.
SAN ANTONIO – Fibrinogen concentrate was noninferior to cryoprecipitate for controlling bleeding following cardiac surgery in the randomized FIBRES trial, Canadian investigators reported.
Among 827 patients undergoing cardiopulmonary bypass, there were no significant differences in the use of allogenenic transfusion products within 24 hours of surgery for patients assigned to receive fibrinogen concentrate for control of bleeding, compared with patients who received cryoprecipitate, reported Jeannie Callum, MD, from Sunnybrook Health Sciences Centre in Toronto, on behalf of coinvestigators in the FIBRES trial.
Fibrinogen concentrate, commonly used to control postoperative bleeding in Europe, was associated with numerically, but not statistically, lower incidence of both adverse events and serious adverse events than cryoprecipitate, the current standard of care in North America.
“Given its safety and logistical advantages, fibrinogen concentrate may be considered in bleeding patients with acquired hypofibrinogenemia,” Dr. Callum said at the annual meeting of the AABB, the group formerly known as the American Association of Blood Banks.
Results of the FIBRES trial were published simultaneously in JAMA (2019 Oct 21. doi: 10.1001/jama.2019.17312).
Acquired hypofibrinogenemia, defined as a fibrinogen level below the range of 1.5-2.0 g/L, is a major cause of excess bleeding after cardiac surgery. European guidelines on the management of bleeding following trauma or cardiac surgery recommend the use of either cryoprecipitate or fibrinogen concentrate to control excessive bleeding in patients with acquired hypofibrinogenemia, Dr. Callum noted.
Cryoprecipitate is a pooled plasma–derived product that contains fibrinogen, but also fibronectin, platelet microparticles, coagulation factors VIII and XIII, and von Willebrand factor.
Additionally, fibrinogen levels in cryoprecipitate can range from as low as 3 g/L to as high as 30 g/L, and the product is normally kept and shipped frozen, and is then thawed for use and pooled prior to administration, with a shelf life of just 4-6 hours.
In contrast, fibrinogen concentrates “are pathogen-reduced and purified; have standardized fibrinogen content (20 g/L); are lyophilized, allowing for easy storage, reconstitution, and administration; and have longer shelf life after reconstitution (up to 24 hours),” Dr. Callum and her colleagues reported.
Despite the North American preference for cryoprecipitate and the European preference for fibrinogen concentrate, there have been few studies directly comparing the two products, which prompted the FIBRES investigators to design a head-to-head trial.
The randomized trial was conducted in 11 Canadian hospitals with adults undergoing cardiac surgery with cardiopulmonary bypass for whom fibrinogen supplementation was ordered in accordance with accepted clinical standards.
Patients were randomly assigned to received either 4 g of fibrinogen concentrate or 10 units of cryoprecipitate for 24 hours, with all patients receiving additional cryoprecipitate as needed after the first day.
Of 15,412 cardiac patients treated at the participating sites, 827 patients met the trial criteria and were randomized. Because the trial met the prespecified stopping criterion for noninferiority of fibrinogen at the interim analysis, the trial was halted, leaving the 827 patients as the final analysis population.
The mean number of allogeneic blood component units administered – the primary outcome – was 16.3 units in the fibrinogen concentrate group and 17.0 units in the cryoprecipitate group (mean ratio, 0.96; P for noninferiority less than .001; P for superiority = .50).
Fibrinogen was also noninferior for the secondary outcomes of individual 24-hour and cumulative 7-day blood component transfusions, and in a post-hoc analysis of cumulative transfusions measured from product administration to 24 hours after termination of cardiopulmonary bypass. These endpoints should be interpreted with caution, however, because they were not corrected for type 1 error, the investigators noted.
Fibrinogen concentrate also appeared to be noninferior for all defined subgroups, except for patients who underwent nonelective procedures, which included all patients in critical state before surgery.
Adverse events (AEs) of any kind occurred in 66.7% of patients with fibrinogen concentrate vs. 72.7% of those on cryoprecipitate. Serious AEs occurred in 31.5% vs. 34.7%, respectively.
Thromboembolic events – stroke or transient ischemic attack, amaurosis fugax (temporary vision loss), myocardial infarction, deep-vein thrombosis, pulmonary embolism, other-vessel thrombosis, disseminated intravascular coagulation, or thrombophlebitis – occurred in 7% vs. 9.6%, respectively.
The investigators acknowledged that the study was limited by the inability to blind the clinical team to the product used, by the adult-only population, and by the likelihood of variable dosing in the cryoprecipitate group.
Advantages of fibrinogen concentrate over cryoprecipitate are that the former is pathogen reduced and is easier to deliver, the investigators said.
“One important consideration is the cost differential that currently favors cryoprecipitate, but this varies across regions, and the most recent economic analysis failed to include the costs of future emerging pathogens and did not include comprehensive activity-based costing,” the investigators wrote in JAMA.
The trial was sponsored by Octapharma AG, which also provided fibrinogen concentrate. Cryoprecipitate was provided by the Canadian Blood Services and Héma-Québec. Dr. Callum reported receiving grants from Canadian Blood Services, Octapharma, and CSL Behring during the conduct of the study. Multiple coauthors had similar disclosures.
SOURCE: Callum J et al. JAMA. 2019 Oct 21. doi:10.1001/jama.2019.17312.
REPORTING FROM AABB 2019
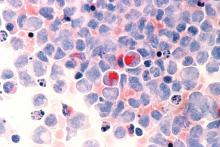
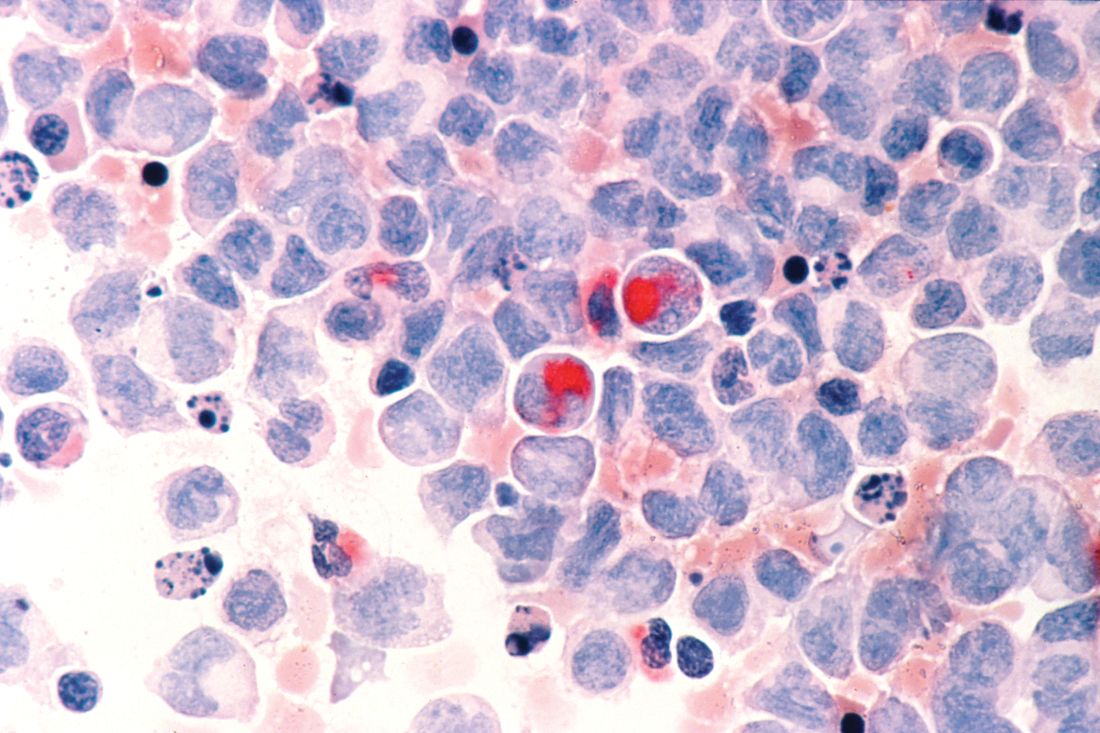
Hematopoietic cell transplant offers realistic cure in secondary AML
yielding significantly better survival outcomes, according to findings from an observational study.
Although secondary AML has been identified as an independent predictor of poor prognosis, it is not included in current risk classifications that provide the basis of deciding when to perform HCT.
Christer Nilsson, MD, of Karolinska Institute, Stockholm, and colleagues, used two nationwide Swedish registries – the Swedish AML Registry and the Swedish Cancer Registry – to characterize how often HCT is performed in these patients and to evaluate its impact in a real-world setting. The registries include all patients with AML diagnosed between 1997 and 2013.
Their findings are in Biology of Blood and Marrow Transplantation.
The analysis included 3,337 adult patients with AML who were intensively treated and did not have acute promyelocytic leukemia. More than three-quarters of the patients had de novo AML and the remainder had secondary AML that was either therapy related or developed after an antecedent myeloid disease. In total, 100 patients with secondary AML underwent HCT while in first complete remission.
In terms of crude survival at 5 years after diagnosis, patients with secondary AML who did not undergo HCT did very poorly. The survival rate was 0% in those with AML preceded by myeloproliferative neoplasm (MPN-AML), 2% in patients with AML preceded by myelodysplastic syndrome (MDS-AML), and 4% in patients with therapy-related AML (t-AML). In contrast, the 5-year overall survival in patients who underwent HCT at any time point or disease stage was 32% for patients with MPN-AML, 18% for patients with MDS-AML, and 25% for patients t-AML.
These crude survival figures suggest that “HCT is the sole realistic curable treatment option for [secondary] AML,” the researchers wrote.
The researchers also performed a propensity score matching analysis of HCT versus chemotherapy consolidation in patients with secondary AML who had been in first complete remission for more than 90 days. The model matched 45 patients who underwent HCT with 66 patients treated with chemotherapy consolidation. The projected 5-year overall survival was 48% in the HCT group, compared with 20% in the consolidation group (P = .01). Similarly, 5-year relapse-free survival was also higher in the HCT group, compared with the consolidation group (43% vs. 21%, P = .02).
“Ideally, the role of transplantation in [secondary] AML should be evaluated in a prospective randomized trial, minimizing the risk of any bias,” the researchers wrote. “However, such a trial is lacking and most likely will never be performed.”
The researchers concluded that HCT should be considered for all patients with secondary AML who are eligible and fit for transplantation.
The study was supported by the Swedish Cancer Foundation, Swedish Research Council, Stockholm County Council, Gothenberg Medical Society, and Assar Gabrielsson Foundation. The researchers reported having no conflicts of interest.
SOURCE: Nilson C et al. Biol Blood Marrow Tranplant. 2019;25:1770-8.
yielding significantly better survival outcomes, according to findings from an observational study.
Although secondary AML has been identified as an independent predictor of poor prognosis, it is not included in current risk classifications that provide the basis of deciding when to perform HCT.
Christer Nilsson, MD, of Karolinska Institute, Stockholm, and colleagues, used two nationwide Swedish registries – the Swedish AML Registry and the Swedish Cancer Registry – to characterize how often HCT is performed in these patients and to evaluate its impact in a real-world setting. The registries include all patients with AML diagnosed between 1997 and 2013.
Their findings are in Biology of Blood and Marrow Transplantation.
The analysis included 3,337 adult patients with AML who were intensively treated and did not have acute promyelocytic leukemia. More than three-quarters of the patients had de novo AML and the remainder had secondary AML that was either therapy related or developed after an antecedent myeloid disease. In total, 100 patients with secondary AML underwent HCT while in first complete remission.
In terms of crude survival at 5 years after diagnosis, patients with secondary AML who did not undergo HCT did very poorly. The survival rate was 0% in those with AML preceded by myeloproliferative neoplasm (MPN-AML), 2% in patients with AML preceded by myelodysplastic syndrome (MDS-AML), and 4% in patients with therapy-related AML (t-AML). In contrast, the 5-year overall survival in patients who underwent HCT at any time point or disease stage was 32% for patients with MPN-AML, 18% for patients with MDS-AML, and 25% for patients t-AML.
These crude survival figures suggest that “HCT is the sole realistic curable treatment option for [secondary] AML,” the researchers wrote.
The researchers also performed a propensity score matching analysis of HCT versus chemotherapy consolidation in patients with secondary AML who had been in first complete remission for more than 90 days. The model matched 45 patients who underwent HCT with 66 patients treated with chemotherapy consolidation. The projected 5-year overall survival was 48% in the HCT group, compared with 20% in the consolidation group (P = .01). Similarly, 5-year relapse-free survival was also higher in the HCT group, compared with the consolidation group (43% vs. 21%, P = .02).
“Ideally, the role of transplantation in [secondary] AML should be evaluated in a prospective randomized trial, minimizing the risk of any bias,” the researchers wrote. “However, such a trial is lacking and most likely will never be performed.”
The researchers concluded that HCT should be considered for all patients with secondary AML who are eligible and fit for transplantation.
The study was supported by the Swedish Cancer Foundation, Swedish Research Council, Stockholm County Council, Gothenberg Medical Society, and Assar Gabrielsson Foundation. The researchers reported having no conflicts of interest.
SOURCE: Nilson C et al. Biol Blood Marrow Tranplant. 2019;25:1770-8.
yielding significantly better survival outcomes, according to findings from an observational study.
Although secondary AML has been identified as an independent predictor of poor prognosis, it is not included in current risk classifications that provide the basis of deciding when to perform HCT.
Christer Nilsson, MD, of Karolinska Institute, Stockholm, and colleagues, used two nationwide Swedish registries – the Swedish AML Registry and the Swedish Cancer Registry – to characterize how often HCT is performed in these patients and to evaluate its impact in a real-world setting. The registries include all patients with AML diagnosed between 1997 and 2013.
Their findings are in Biology of Blood and Marrow Transplantation.
The analysis included 3,337 adult patients with AML who were intensively treated and did not have acute promyelocytic leukemia. More than three-quarters of the patients had de novo AML and the remainder had secondary AML that was either therapy related or developed after an antecedent myeloid disease. In total, 100 patients with secondary AML underwent HCT while in first complete remission.
In terms of crude survival at 5 years after diagnosis, patients with secondary AML who did not undergo HCT did very poorly. The survival rate was 0% in those with AML preceded by myeloproliferative neoplasm (MPN-AML), 2% in patients with AML preceded by myelodysplastic syndrome (MDS-AML), and 4% in patients with therapy-related AML (t-AML). In contrast, the 5-year overall survival in patients who underwent HCT at any time point or disease stage was 32% for patients with MPN-AML, 18% for patients with MDS-AML, and 25% for patients t-AML.
These crude survival figures suggest that “HCT is the sole realistic curable treatment option for [secondary] AML,” the researchers wrote.
The researchers also performed a propensity score matching analysis of HCT versus chemotherapy consolidation in patients with secondary AML who had been in first complete remission for more than 90 days. The model matched 45 patients who underwent HCT with 66 patients treated with chemotherapy consolidation. The projected 5-year overall survival was 48% in the HCT group, compared with 20% in the consolidation group (P = .01). Similarly, 5-year relapse-free survival was also higher in the HCT group, compared with the consolidation group (43% vs. 21%, P = .02).
“Ideally, the role of transplantation in [secondary] AML should be evaluated in a prospective randomized trial, minimizing the risk of any bias,” the researchers wrote. “However, such a trial is lacking and most likely will never be performed.”
The researchers concluded that HCT should be considered for all patients with secondary AML who are eligible and fit for transplantation.
The study was supported by the Swedish Cancer Foundation, Swedish Research Council, Stockholm County Council, Gothenberg Medical Society, and Assar Gabrielsson Foundation. The researchers reported having no conflicts of interest.
SOURCE: Nilson C et al. Biol Blood Marrow Tranplant. 2019;25:1770-8.
FROM BIOLOGY OF BLOOD AND MARROW TRANSPLANTATION
Updated international consensus recommendations on management of acute upper GI bleeding
Guidelines on the management of acute upper gastrointestinal bleeding have been updated, including recommendations on managing patients on antiplatelet or anticoagulant therapy and on use of endoscopy and new therapeutic approaches.
Writing in Annals of Internal Medicine, an international group of experts published an update to the 2010 International Consensus Recommendations on the Management of Patients With Nonvariceal Upper Gastrointestinal Bleeding, with a focus on resuscitation and risk assessment; pre-endoscopic, endoscopic, and pharmacologic management; and secondary prophylaxis.
Alan N. Barkun, MDCM, MSc, from McGill University, Montreal, and coauthors first recommended that fluid resuscitation should be initiated in patients with acute upper gastrointestinal bleeding and hemodynamic instability to avoid hemorrhagic shock and restore end-organ perfusion and tissue oxygenation while the bleeding is brought under control.
They acknowledged the uncertainty around whether colloid or crystalloid fluid should be used, but suggested routine use of colloids was not justified because they were more expensive and did not appear to increase survival.
On the question of whether the resuscitation should be aggressive or restrictive in its timing and rate, the group said there was not enough evidence to support a recommendation on this. “The important issue in patients with hemorrhagic shock due to trauma or UGIB [upper gastrointestinal bleeding] is to stop the bleeding while minimizing hemodynamic compromise,” they wrote.
They also advised blood transfusions in patients with a hemoglobin level below 80 g/L who did not have underlying cardiovascular disease, but suggested a higher hemoglobin threshold for those with underlying cardiovascular disease.
The second recommendation was that patients with a Glasgow Blatchford score of 1 or less were at very low risk for rebleeding and mortality, and these patients may therefore not need hospitalization or inpatient endoscopy. They advised against using the AIMS65 prognostic score for this purpose because it was designed to identify patients at high risk of death, not those at low risk for safe discharge.
In regard to endoscopic management, they advocated that all patients with acute upper gastrointestinal bleeding – whether low or high risk – undergo endoscopy within 24 hours of presentation. This was even more urgent in patients being treated with anticoagulants. “Because of the recognized benefits of early endoscopy, coagulopathy should be treated as necessary but endoscopy should not be delayed,” they wrote.
Patients with acutely bleeding ulcers with high-risk stigmata should undergo endoscopic therapy preferably with thermocoagulation or sclerosant injection, or with hemoclips depending on the bleeding location and patient characteristics.
The group also included two conditional recommendations, based on very-low-quality evidence, that patients with actively bleeding ulcers receive TC-325 hemostatic powder as temporizing therapy to stop the bleeding if conventional endoscopic therapies aren’t available or fail. However, they stressed that TC-325 should not be used as a single therapeutic strategy.
Because of a lack of efficacy data and low availability of expertise in the technology, the authors said they could not make a recommendation for or against using a Doppler endoscopic probe (DEP) to assess the need for further endoscopic therapy.
“The group generally agreed that although making a recommendation for or against using DEP to manage UGIB is premature, DEP has the potential to alter the usual approach to visually assessing bleeding lesion risk when evaluating the need for, and adequacy of, endoscopic hemostasis.”
The guidelines also addressed the issue of pharmacologic management of acute upper gastrointestinal bleeding. They strongly recommended that patients with bleeding ulcers and high-risk stigmata who have undergone successful endoscopic therapy should then receive an intravenous loading dose of proton pump inhibitor (PPI) therapy, followed by continuous intravenous infusion.
“Cost-effectiveness studies have suggested that high-dose intravenous PPIs after successful endoscopic hemostasis improve outcomes at a modest cost increase relative to non–high-dose intravenous or oral PPI strategies,” they wrote.
A second conditional recommendation, based on very-low-quality evidence, was that patients with a bleeding ulcer who were at high risk for rebleeding be also treated twice-daily with oral PPIs for 2 weeks, then once-daily. They also recommended patients on cardiovascular prophylaxis with single or dual antiplatelet therapy or anticoagulant therapy be given PPIs.
“The consensus group concluded that, for high-risk patients with an ongoing need for anticoagulants, the evidence suggests that the benefits of secondary prophylaxis outweigh the risks.”
The group was supported by a grant from CIHR Institute of Nutrition, Metabolism and Diabetes and from the Saudi Gastroenterology Association. Nine authors declared grants, personal fees, honoraria and other funding from the pharmaceutical and medical device sector outside the submitted work. No other conflicts of interest were declared.
SOURCE: Barkun A et al. Ann Intern Med 2019, October 22. doi: 10.7326/M19-1795.
These updated consensus guidelines provide a rigorous review of evidence on managing nonvariceal upper gastrointestinal bleeding. The recommendations for patients on anticoagulant or antiplatelet therapy will be particularly helpful because of increasing use of these medications. The advice on proton pump inhibitor therapy in patients on these drugs who have had previous ulcer bleeding can help allay concerns about possible integrations between PPIs and clopidogrel.
While the guidelines recommend using the Glasgow Blatchford scale to guide hospitalization decisions, prognostic scores are not commonly used in the emergency department, and many patients present with a Glasgow Blatchford score greater than 1, so this tool may have little impact on hospitalization rates. More studies are needed to compare clinical judgment with these prognostic scores.
Angel Lanas, MD, is from the University Clinic Hospital at the University of Zaragoza (Spain). These comments are adapted from an accompanying editorial (Ann Intern Med 2019, October 22. doi: 10.7326/M19-2789). Dr. Lanas declared unrelated personal fees from the pharmaceutical sector.
These updated consensus guidelines provide a rigorous review of evidence on managing nonvariceal upper gastrointestinal bleeding. The recommendations for patients on anticoagulant or antiplatelet therapy will be particularly helpful because of increasing use of these medications. The advice on proton pump inhibitor therapy in patients on these drugs who have had previous ulcer bleeding can help allay concerns about possible integrations between PPIs and clopidogrel.
While the guidelines recommend using the Glasgow Blatchford scale to guide hospitalization decisions, prognostic scores are not commonly used in the emergency department, and many patients present with a Glasgow Blatchford score greater than 1, so this tool may have little impact on hospitalization rates. More studies are needed to compare clinical judgment with these prognostic scores.
Angel Lanas, MD, is from the University Clinic Hospital at the University of Zaragoza (Spain). These comments are adapted from an accompanying editorial (Ann Intern Med 2019, October 22. doi: 10.7326/M19-2789). Dr. Lanas declared unrelated personal fees from the pharmaceutical sector.
These updated consensus guidelines provide a rigorous review of evidence on managing nonvariceal upper gastrointestinal bleeding. The recommendations for patients on anticoagulant or antiplatelet therapy will be particularly helpful because of increasing use of these medications. The advice on proton pump inhibitor therapy in patients on these drugs who have had previous ulcer bleeding can help allay concerns about possible integrations between PPIs and clopidogrel.
While the guidelines recommend using the Glasgow Blatchford scale to guide hospitalization decisions, prognostic scores are not commonly used in the emergency department, and many patients present with a Glasgow Blatchford score greater than 1, so this tool may have little impact on hospitalization rates. More studies are needed to compare clinical judgment with these prognostic scores.
Angel Lanas, MD, is from the University Clinic Hospital at the University of Zaragoza (Spain). These comments are adapted from an accompanying editorial (Ann Intern Med 2019, October 22. doi: 10.7326/M19-2789). Dr. Lanas declared unrelated personal fees from the pharmaceutical sector.
Guidelines on the management of acute upper gastrointestinal bleeding have been updated, including recommendations on managing patients on antiplatelet or anticoagulant therapy and on use of endoscopy and new therapeutic approaches.
Writing in Annals of Internal Medicine, an international group of experts published an update to the 2010 International Consensus Recommendations on the Management of Patients With Nonvariceal Upper Gastrointestinal Bleeding, with a focus on resuscitation and risk assessment; pre-endoscopic, endoscopic, and pharmacologic management; and secondary prophylaxis.
Alan N. Barkun, MDCM, MSc, from McGill University, Montreal, and coauthors first recommended that fluid resuscitation should be initiated in patients with acute upper gastrointestinal bleeding and hemodynamic instability to avoid hemorrhagic shock and restore end-organ perfusion and tissue oxygenation while the bleeding is brought under control.
They acknowledged the uncertainty around whether colloid or crystalloid fluid should be used, but suggested routine use of colloids was not justified because they were more expensive and did not appear to increase survival.
On the question of whether the resuscitation should be aggressive or restrictive in its timing and rate, the group said there was not enough evidence to support a recommendation on this. “The important issue in patients with hemorrhagic shock due to trauma or UGIB [upper gastrointestinal bleeding] is to stop the bleeding while minimizing hemodynamic compromise,” they wrote.
They also advised blood transfusions in patients with a hemoglobin level below 80 g/L who did not have underlying cardiovascular disease, but suggested a higher hemoglobin threshold for those with underlying cardiovascular disease.
The second recommendation was that patients with a Glasgow Blatchford score of 1 or less were at very low risk for rebleeding and mortality, and these patients may therefore not need hospitalization or inpatient endoscopy. They advised against using the AIMS65 prognostic score for this purpose because it was designed to identify patients at high risk of death, not those at low risk for safe discharge.
In regard to endoscopic management, they advocated that all patients with acute upper gastrointestinal bleeding – whether low or high risk – undergo endoscopy within 24 hours of presentation. This was even more urgent in patients being treated with anticoagulants. “Because of the recognized benefits of early endoscopy, coagulopathy should be treated as necessary but endoscopy should not be delayed,” they wrote.
Patients with acutely bleeding ulcers with high-risk stigmata should undergo endoscopic therapy preferably with thermocoagulation or sclerosant injection, or with hemoclips depending on the bleeding location and patient characteristics.
The group also included two conditional recommendations, based on very-low-quality evidence, that patients with actively bleeding ulcers receive TC-325 hemostatic powder as temporizing therapy to stop the bleeding if conventional endoscopic therapies aren’t available or fail. However, they stressed that TC-325 should not be used as a single therapeutic strategy.
Because of a lack of efficacy data and low availability of expertise in the technology, the authors said they could not make a recommendation for or against using a Doppler endoscopic probe (DEP) to assess the need for further endoscopic therapy.
“The group generally agreed that although making a recommendation for or against using DEP to manage UGIB is premature, DEP has the potential to alter the usual approach to visually assessing bleeding lesion risk when evaluating the need for, and adequacy of, endoscopic hemostasis.”
The guidelines also addressed the issue of pharmacologic management of acute upper gastrointestinal bleeding. They strongly recommended that patients with bleeding ulcers and high-risk stigmata who have undergone successful endoscopic therapy should then receive an intravenous loading dose of proton pump inhibitor (PPI) therapy, followed by continuous intravenous infusion.
“Cost-effectiveness studies have suggested that high-dose intravenous PPIs after successful endoscopic hemostasis improve outcomes at a modest cost increase relative to non–high-dose intravenous or oral PPI strategies,” they wrote.
A second conditional recommendation, based on very-low-quality evidence, was that patients with a bleeding ulcer who were at high risk for rebleeding be also treated twice-daily with oral PPIs for 2 weeks, then once-daily. They also recommended patients on cardiovascular prophylaxis with single or dual antiplatelet therapy or anticoagulant therapy be given PPIs.
“The consensus group concluded that, for high-risk patients with an ongoing need for anticoagulants, the evidence suggests that the benefits of secondary prophylaxis outweigh the risks.”
The group was supported by a grant from CIHR Institute of Nutrition, Metabolism and Diabetes and from the Saudi Gastroenterology Association. Nine authors declared grants, personal fees, honoraria and other funding from the pharmaceutical and medical device sector outside the submitted work. No other conflicts of interest were declared.
SOURCE: Barkun A et al. Ann Intern Med 2019, October 22. doi: 10.7326/M19-1795.
Guidelines on the management of acute upper gastrointestinal bleeding have been updated, including recommendations on managing patients on antiplatelet or anticoagulant therapy and on use of endoscopy and new therapeutic approaches.
Writing in Annals of Internal Medicine, an international group of experts published an update to the 2010 International Consensus Recommendations on the Management of Patients With Nonvariceal Upper Gastrointestinal Bleeding, with a focus on resuscitation and risk assessment; pre-endoscopic, endoscopic, and pharmacologic management; and secondary prophylaxis.
Alan N. Barkun, MDCM, MSc, from McGill University, Montreal, and coauthors first recommended that fluid resuscitation should be initiated in patients with acute upper gastrointestinal bleeding and hemodynamic instability to avoid hemorrhagic shock and restore end-organ perfusion and tissue oxygenation while the bleeding is brought under control.
They acknowledged the uncertainty around whether colloid or crystalloid fluid should be used, but suggested routine use of colloids was not justified because they were more expensive and did not appear to increase survival.
On the question of whether the resuscitation should be aggressive or restrictive in its timing and rate, the group said there was not enough evidence to support a recommendation on this. “The important issue in patients with hemorrhagic shock due to trauma or UGIB [upper gastrointestinal bleeding] is to stop the bleeding while minimizing hemodynamic compromise,” they wrote.
They also advised blood transfusions in patients with a hemoglobin level below 80 g/L who did not have underlying cardiovascular disease, but suggested a higher hemoglobin threshold for those with underlying cardiovascular disease.
The second recommendation was that patients with a Glasgow Blatchford score of 1 or less were at very low risk for rebleeding and mortality, and these patients may therefore not need hospitalization or inpatient endoscopy. They advised against using the AIMS65 prognostic score for this purpose because it was designed to identify patients at high risk of death, not those at low risk for safe discharge.
In regard to endoscopic management, they advocated that all patients with acute upper gastrointestinal bleeding – whether low or high risk – undergo endoscopy within 24 hours of presentation. This was even more urgent in patients being treated with anticoagulants. “Because of the recognized benefits of early endoscopy, coagulopathy should be treated as necessary but endoscopy should not be delayed,” they wrote.
Patients with acutely bleeding ulcers with high-risk stigmata should undergo endoscopic therapy preferably with thermocoagulation or sclerosant injection, or with hemoclips depending on the bleeding location and patient characteristics.
The group also included two conditional recommendations, based on very-low-quality evidence, that patients with actively bleeding ulcers receive TC-325 hemostatic powder as temporizing therapy to stop the bleeding if conventional endoscopic therapies aren’t available or fail. However, they stressed that TC-325 should not be used as a single therapeutic strategy.
Because of a lack of efficacy data and low availability of expertise in the technology, the authors said they could not make a recommendation for or against using a Doppler endoscopic probe (DEP) to assess the need for further endoscopic therapy.
“The group generally agreed that although making a recommendation for or against using DEP to manage UGIB is premature, DEP has the potential to alter the usual approach to visually assessing bleeding lesion risk when evaluating the need for, and adequacy of, endoscopic hemostasis.”
The guidelines also addressed the issue of pharmacologic management of acute upper gastrointestinal bleeding. They strongly recommended that patients with bleeding ulcers and high-risk stigmata who have undergone successful endoscopic therapy should then receive an intravenous loading dose of proton pump inhibitor (PPI) therapy, followed by continuous intravenous infusion.
“Cost-effectiveness studies have suggested that high-dose intravenous PPIs after successful endoscopic hemostasis improve outcomes at a modest cost increase relative to non–high-dose intravenous or oral PPI strategies,” they wrote.
A second conditional recommendation, based on very-low-quality evidence, was that patients with a bleeding ulcer who were at high risk for rebleeding be also treated twice-daily with oral PPIs for 2 weeks, then once-daily. They also recommended patients on cardiovascular prophylaxis with single or dual antiplatelet therapy or anticoagulant therapy be given PPIs.
“The consensus group concluded that, for high-risk patients with an ongoing need for anticoagulants, the evidence suggests that the benefits of secondary prophylaxis outweigh the risks.”
The group was supported by a grant from CIHR Institute of Nutrition, Metabolism and Diabetes and from the Saudi Gastroenterology Association. Nine authors declared grants, personal fees, honoraria and other funding from the pharmaceutical and medical device sector outside the submitted work. No other conflicts of interest were declared.
SOURCE: Barkun A et al. Ann Intern Med 2019, October 22. doi: 10.7326/M19-1795.
FROM ANNALS OF INTERNAL MEDICINE
FDA gives nod to earlier use of Nplate in adults with ITP
Romiplostim (Nplate) earned a new indication from the Food and Drug Administration, allowing for earlier usage in adult patients with immune thrombocytopenia (ITP) who have had an insufficient response to corticosteroids, immunoglobulins, or splenectomy.
The new indication is for newly diagnosed and persistent adult patients.
Romiplostim is already approved for the treatment of pediatric patients aged 1 year and older who have had ITP for at least 6 months and had an insufficient response to other treatments, as well as for adults patients with chronic ITP who had insufficient response to other therapies.
The latest approval is based on positive results from a single-arm, phase 2 trial in adults with ITP who had an insufficient response to first-line treatment. The study met its primary endpoint – platelet response (50 x 109/L or greater). The median number of months with platelet response was 11 months during a 12-month treatment period. The median time to first platelet response was just over 2 weeks.
Adverse events of at least 5% incidence in patients taking romiplostim with an ITP duration up to 12 months included bronchitis, sinusitis, vomiting, arthralgia, myalgia, headache, dizziness, diarrhea, upper respiratory tract infection, cough, nausea, and oropharyngeal pain. There was a 2% incidence of thrombocytosis among adults with an ITP duration up to 12 months.
Romiplostim (Nplate) earned a new indication from the Food and Drug Administration, allowing for earlier usage in adult patients with immune thrombocytopenia (ITP) who have had an insufficient response to corticosteroids, immunoglobulins, or splenectomy.
The new indication is for newly diagnosed and persistent adult patients.
Romiplostim is already approved for the treatment of pediatric patients aged 1 year and older who have had ITP for at least 6 months and had an insufficient response to other treatments, as well as for adults patients with chronic ITP who had insufficient response to other therapies.
The latest approval is based on positive results from a single-arm, phase 2 trial in adults with ITP who had an insufficient response to first-line treatment. The study met its primary endpoint – platelet response (50 x 109/L or greater). The median number of months with platelet response was 11 months during a 12-month treatment period. The median time to first platelet response was just over 2 weeks.
Adverse events of at least 5% incidence in patients taking romiplostim with an ITP duration up to 12 months included bronchitis, sinusitis, vomiting, arthralgia, myalgia, headache, dizziness, diarrhea, upper respiratory tract infection, cough, nausea, and oropharyngeal pain. There was a 2% incidence of thrombocytosis among adults with an ITP duration up to 12 months.
Romiplostim (Nplate) earned a new indication from the Food and Drug Administration, allowing for earlier usage in adult patients with immune thrombocytopenia (ITP) who have had an insufficient response to corticosteroids, immunoglobulins, or splenectomy.
The new indication is for newly diagnosed and persistent adult patients.
Romiplostim is already approved for the treatment of pediatric patients aged 1 year and older who have had ITP for at least 6 months and had an insufficient response to other treatments, as well as for adults patients with chronic ITP who had insufficient response to other therapies.
The latest approval is based on positive results from a single-arm, phase 2 trial in adults with ITP who had an insufficient response to first-line treatment. The study met its primary endpoint – platelet response (50 x 109/L or greater). The median number of months with platelet response was 11 months during a 12-month treatment period. The median time to first platelet response was just over 2 weeks.
Adverse events of at least 5% incidence in patients taking romiplostim with an ITP duration up to 12 months included bronchitis, sinusitis, vomiting, arthralgia, myalgia, headache, dizziness, diarrhea, upper respiratory tract infection, cough, nausea, and oropharyngeal pain. There was a 2% incidence of thrombocytosis among adults with an ITP duration up to 12 months.
Global blood supply runs low
Nearly two-thirds of countries worldwide have an insufficient supply of blood for transfusion, according to findings from a recent modeling study including 195 countries and territories.
Low- and middle-income countries are most often affected, reported lead author Nicholas Roberts of the University of Washington in Seattle and colleagues. Among all countries in Oceania, south Asia, and eastern, central, and western sub-Saharan Africa, not one had enough blood to meet estimated needs, they noted.
Shortages are attributable to a variety of challenges, including resource constraints, insufficient component production, and a high prevalence of infectious diseases, according to investigators. And existing guidelines, which call for 10-20 donations per 1,000 people in the population, may need to be revised, investigators suggested.
“These estimates are important, as they can be used to guide further investments in blood transfusion services, for analysis of current transfusion practices, and to highlight need for alternatives to transfusions such as antifibrinolytics, blood saving devices, and implementation of blood management systems,” the investigators wrote in Lancet Haematology.
Blood availability was calculated using the 2016 WHO Global Status Report on Blood Safety and Availability, in which 92% of countries participated. Blood needs were calculated using multiple databases, including the (U.S.) National Inpatient Sample datasets from 2000-2014, the State Inpatient Databases from 2003-2007, and the Global Burden of Disease 2017 study.
The global blood need was almost 305 million blood product units, while the supply totaled approximately 272 million units. These shortages, however, were not distributed equally across the globe. Out of 195 countries, 119 (61%) had an insufficient supply of blood to meet anticipated demand. Within this group, the shortage equated to about 102 million blood product units.
Denmark had the greatest supply of blood products (red blood cell products, platelets, and plasma), at 14,704 units per 100,000 population, compared with South Sudan, the least prepared to meet transfusion needs, with just 46 blood product units available per 100,000 population. This pattern was echoed across the globe; high-income countries were usually better stocked than those with low or middle income.
To meet demands, some of the most affected countries would need to raise their collection goals from single-digit figures to as high as 40 donations per 1,000 population, the investigators advised.
“Many countries face critical undersupply of transfusions, which will become more pronounced as access to care improves,” the investigators wrote.
The study was funded by the National Institutes of Health. The investigators reported having no other financial disclosures.
SOURCE: Roberts N et al. Lancet Haematol. 2019 Oct 17. doi: 10.1016/ S2352-3026(19)30200-5.
Nearly two-thirds of countries worldwide have an insufficient supply of blood for transfusion, according to findings from a recent modeling study including 195 countries and territories.
Low- and middle-income countries are most often affected, reported lead author Nicholas Roberts of the University of Washington in Seattle and colleagues. Among all countries in Oceania, south Asia, and eastern, central, and western sub-Saharan Africa, not one had enough blood to meet estimated needs, they noted.
Shortages are attributable to a variety of challenges, including resource constraints, insufficient component production, and a high prevalence of infectious diseases, according to investigators. And existing guidelines, which call for 10-20 donations per 1,000 people in the population, may need to be revised, investigators suggested.
“These estimates are important, as they can be used to guide further investments in blood transfusion services, for analysis of current transfusion practices, and to highlight need for alternatives to transfusions such as antifibrinolytics, blood saving devices, and implementation of blood management systems,” the investigators wrote in Lancet Haematology.
Blood availability was calculated using the 2016 WHO Global Status Report on Blood Safety and Availability, in which 92% of countries participated. Blood needs were calculated using multiple databases, including the (U.S.) National Inpatient Sample datasets from 2000-2014, the State Inpatient Databases from 2003-2007, and the Global Burden of Disease 2017 study.
The global blood need was almost 305 million blood product units, while the supply totaled approximately 272 million units. These shortages, however, were not distributed equally across the globe. Out of 195 countries, 119 (61%) had an insufficient supply of blood to meet anticipated demand. Within this group, the shortage equated to about 102 million blood product units.
Denmark had the greatest supply of blood products (red blood cell products, platelets, and plasma), at 14,704 units per 100,000 population, compared with South Sudan, the least prepared to meet transfusion needs, with just 46 blood product units available per 100,000 population. This pattern was echoed across the globe; high-income countries were usually better stocked than those with low or middle income.
To meet demands, some of the most affected countries would need to raise their collection goals from single-digit figures to as high as 40 donations per 1,000 population, the investigators advised.
“Many countries face critical undersupply of transfusions, which will become more pronounced as access to care improves,” the investigators wrote.
The study was funded by the National Institutes of Health. The investigators reported having no other financial disclosures.
SOURCE: Roberts N et al. Lancet Haematol. 2019 Oct 17. doi: 10.1016/ S2352-3026(19)30200-5.
Nearly two-thirds of countries worldwide have an insufficient supply of blood for transfusion, according to findings from a recent modeling study including 195 countries and territories.
Low- and middle-income countries are most often affected, reported lead author Nicholas Roberts of the University of Washington in Seattle and colleagues. Among all countries in Oceania, south Asia, and eastern, central, and western sub-Saharan Africa, not one had enough blood to meet estimated needs, they noted.
Shortages are attributable to a variety of challenges, including resource constraints, insufficient component production, and a high prevalence of infectious diseases, according to investigators. And existing guidelines, which call for 10-20 donations per 1,000 people in the population, may need to be revised, investigators suggested.
“These estimates are important, as they can be used to guide further investments in blood transfusion services, for analysis of current transfusion practices, and to highlight need for alternatives to transfusions such as antifibrinolytics, blood saving devices, and implementation of blood management systems,” the investigators wrote in Lancet Haematology.
Blood availability was calculated using the 2016 WHO Global Status Report on Blood Safety and Availability, in which 92% of countries participated. Blood needs were calculated using multiple databases, including the (U.S.) National Inpatient Sample datasets from 2000-2014, the State Inpatient Databases from 2003-2007, and the Global Burden of Disease 2017 study.
The global blood need was almost 305 million blood product units, while the supply totaled approximately 272 million units. These shortages, however, were not distributed equally across the globe. Out of 195 countries, 119 (61%) had an insufficient supply of blood to meet anticipated demand. Within this group, the shortage equated to about 102 million blood product units.
Denmark had the greatest supply of blood products (red blood cell products, platelets, and plasma), at 14,704 units per 100,000 population, compared with South Sudan, the least prepared to meet transfusion needs, with just 46 blood product units available per 100,000 population. This pattern was echoed across the globe; high-income countries were usually better stocked than those with low or middle income.
To meet demands, some of the most affected countries would need to raise their collection goals from single-digit figures to as high as 40 donations per 1,000 population, the investigators advised.
“Many countries face critical undersupply of transfusions, which will become more pronounced as access to care improves,” the investigators wrote.
The study was funded by the National Institutes of Health. The investigators reported having no other financial disclosures.
SOURCE: Roberts N et al. Lancet Haematol. 2019 Oct 17. doi: 10.1016/ S2352-3026(19)30200-5.
FROM LANCET HAEMATOLOGY
FDA approves rivaroxaban for VTE prevention in hospitalized, acutely ill patients
The Food and Drug Administration has approved rivaroxaban (Xarelto) for the prevention of venous thromboembolism (VTE) in hospitalized, acutely ill patients at risk for thromboembolic complications who do not have a high bleeding risk, according to a release from Janssen.
FDA approval for the new indication is based on results from the phase 3 MAGELLAN and MARINER trials, which included more than 20,000 hospitalized, acutely ill patients. In MAGELLAN, rivaroxaban demonstrated noninferiority to enoxaparin, a low-molecular-weight heparin, in short-term usage, and it was superior over the long term, compared with short-term enoxaparin followed by placebo.
While VTE and VTE-related deaths were not reduced in MARINER, compared with placebo, patients who received rivaroxaban did see a significantly reduction in symptomatic VTE with a favorable safety profile.
According to the indication, rivaroxaban can be administered to patients during hospitalization and can be continued after discharge for 31-39 days. The safety profile in MAGELLAN and MARINER was consistent with that already seen, with the most common adverse event being bleeding.
The new indication is the eighth for rivaroxaban, the most of any direct oral anticoagulant; six of these are specifically for the treatment, prevention, and reduction in the risk of VTE recurrence.
“With this new approval, Xarelto as an oral-only option now has the potential to change how acutely ill medical patients are managed for the prevention of blood clots, both in the hospital and for an extended period after discharge,” said Alex C. Spyropoulos, MD, of Northwell Health at Lenox Hill Hospital, New York, and a member of the steering committee of the MAGELLAN trial.
Find the full press release on the Janssen website.
The Food and Drug Administration has approved rivaroxaban (Xarelto) for the prevention of venous thromboembolism (VTE) in hospitalized, acutely ill patients at risk for thromboembolic complications who do not have a high bleeding risk, according to a release from Janssen.
FDA approval for the new indication is based on results from the phase 3 MAGELLAN and MARINER trials, which included more than 20,000 hospitalized, acutely ill patients. In MAGELLAN, rivaroxaban demonstrated noninferiority to enoxaparin, a low-molecular-weight heparin, in short-term usage, and it was superior over the long term, compared with short-term enoxaparin followed by placebo.
While VTE and VTE-related deaths were not reduced in MARINER, compared with placebo, patients who received rivaroxaban did see a significantly reduction in symptomatic VTE with a favorable safety profile.
According to the indication, rivaroxaban can be administered to patients during hospitalization and can be continued after discharge for 31-39 days. The safety profile in MAGELLAN and MARINER was consistent with that already seen, with the most common adverse event being bleeding.
The new indication is the eighth for rivaroxaban, the most of any direct oral anticoagulant; six of these are specifically for the treatment, prevention, and reduction in the risk of VTE recurrence.
“With this new approval, Xarelto as an oral-only option now has the potential to change how acutely ill medical patients are managed for the prevention of blood clots, both in the hospital and for an extended period after discharge,” said Alex C. Spyropoulos, MD, of Northwell Health at Lenox Hill Hospital, New York, and a member of the steering committee of the MAGELLAN trial.
Find the full press release on the Janssen website.
The Food and Drug Administration has approved rivaroxaban (Xarelto) for the prevention of venous thromboembolism (VTE) in hospitalized, acutely ill patients at risk for thromboembolic complications who do not have a high bleeding risk, according to a release from Janssen.
FDA approval for the new indication is based on results from the phase 3 MAGELLAN and MARINER trials, which included more than 20,000 hospitalized, acutely ill patients. In MAGELLAN, rivaroxaban demonstrated noninferiority to enoxaparin, a low-molecular-weight heparin, in short-term usage, and it was superior over the long term, compared with short-term enoxaparin followed by placebo.
While VTE and VTE-related deaths were not reduced in MARINER, compared with placebo, patients who received rivaroxaban did see a significantly reduction in symptomatic VTE with a favorable safety profile.
According to the indication, rivaroxaban can be administered to patients during hospitalization and can be continued after discharge for 31-39 days. The safety profile in MAGELLAN and MARINER was consistent with that already seen, with the most common adverse event being bleeding.
The new indication is the eighth for rivaroxaban, the most of any direct oral anticoagulant; six of these are specifically for the treatment, prevention, and reduction in the risk of VTE recurrence.
“With this new approval, Xarelto as an oral-only option now has the potential to change how acutely ill medical patients are managed for the prevention of blood clots, both in the hospital and for an extended period after discharge,” said Alex C. Spyropoulos, MD, of Northwell Health at Lenox Hill Hospital, New York, and a member of the steering committee of the MAGELLAN trial.
Find the full press release on the Janssen website.
Rivaroxaban trends toward higher thrombotic risk than vitamin K antagonists in APS
suggests a recent trial conducted in Spain.
Stroke was also more common among those taking rivaroxaban, while major bleeding was slightly less common, reported lead author Josep Ordi-Ros, MD, PhD, of Vall d’Hebrón University Hospital Research Institute in Barcelona, and colleagues in Annals of Internal Medicine.
“Two randomized, controlled trials comparing rivaroxaban with warfarin suggested that rivaroxaban may be efficacious in patients with previous venous thromboembolism who are receiving standard-intensity anticoagulation but showed an increased thrombotic risk in those with triple-positive antiphospholipid antibodies,” the investigators wrote. However, they also noted that these findings required a cautious interpretation because of study limitations, such as premature termination caused by an excess of study events and the use of a laboratory surrogate marker as a primary outcome.
To learn more, the investigators performed an open-label, phase 3 trial involving 190 patients with thrombotic APS. Patients were randomized in a 1:1 ratio to receive either rivaroxaban (20 mg per day, or 15 mg per day for patients with a creatinine clearance of 30-49 mL/min per 1.73 m2) or an adjusted dosage of vitamin K antagonists (target international normalized ratio of 2.0-3.0, or 3.1-4.0 for those with a history of recurrent thrombosis).
Patients underwent evaluations every month for the first 3 months and then every 3 months thereafter, each of which involved a variety of laboratory diagnostics such as checks for antinuclear antibodies and lupus anticoagulant, among others. Statistical analyses aimed to determine if rivaroxaban was noninferior to therapy with vitamin K antagonists based on parameters drawn from previous meta-analyses, as no studies had compared the two types of treatment when the present study was designed.
After 3 years of follow-up, almost twice as many patients in the rivaroxaban group had experienced recurrent thrombosis (11.6% vs. 6.3%), although this finding lacked statistical significance for both noninferiority of rivaroxaban (P = .29) and superiority of vitamin K antagonists (P = .20). Still, supporting a similar trend toward differences in efficacy, stroke was more common in the rivaroxaban group, in which nine events occurred, compared with none in the vitamin K antagonist group. In contrast, major bleeding was slightly less common with rivaroxaban than vitamin K antagonists (6.3% vs. 7.4%).
“In conclusion, rivaroxaban did not demonstrate noninferiority to dose-adjusted vitamin K antagonists for secondary thromboprophylaxis in patients with thrombotic APS,” the investigators wrote. “Instead, our results indicate a recurrent thrombotic rate that is nearly double, albeit without statistical significance.”
The study was funded by Bayer Hispania. One coauthor reported additional relationships with Pfizer, Lilly, Janssen, and others.
SOURCE: Ordi-Ros J et al. Ann Intern Med. 2019 Oct 15. doi: 10.7326/M19-0291.
The recent trial by Ordi-Ros et al. revealed similar findings to a previous trial, TRAPS, by Pengo et al., which compared rivaroxaban with warfarin among patients with thrombotic antiphospholipid syndrome and triple positivity for antiphospholipid antibodies. Despite the caveat that TRAPS was prematurely terminated, in both studies, a higher proportion of patients in the rivaroxaban group than the vitamin K antagonist group had thrombotic events, most of which were arterial, whether considering MI or stroke. Furthermore, both studies did not show noninferiority of rivaroxaban versus dose-adjusted vitamin K antagonists.
The reasons for this failure of noninferiority remain unclear.
Denis Wahl, MD, PhD, and Virginie Dufrost, MD, are with the University of Lorraine, Nancy, France, and the Centre Hospitalier Universitaire de Nancy. No conflicts of interest were reported. His remarks are adapted from an accompanying editorial (Ann Intern Med. 2019 Oct 15. doi: 10.7326/M19-2815).
The recent trial by Ordi-Ros et al. revealed similar findings to a previous trial, TRAPS, by Pengo et al., which compared rivaroxaban with warfarin among patients with thrombotic antiphospholipid syndrome and triple positivity for antiphospholipid antibodies. Despite the caveat that TRAPS was prematurely terminated, in both studies, a higher proportion of patients in the rivaroxaban group than the vitamin K antagonist group had thrombotic events, most of which were arterial, whether considering MI or stroke. Furthermore, both studies did not show noninferiority of rivaroxaban versus dose-adjusted vitamin K antagonists.
The reasons for this failure of noninferiority remain unclear.
Denis Wahl, MD, PhD, and Virginie Dufrost, MD, are with the University of Lorraine, Nancy, France, and the Centre Hospitalier Universitaire de Nancy. No conflicts of interest were reported. His remarks are adapted from an accompanying editorial (Ann Intern Med. 2019 Oct 15. doi: 10.7326/M19-2815).
The recent trial by Ordi-Ros et al. revealed similar findings to a previous trial, TRAPS, by Pengo et al., which compared rivaroxaban with warfarin among patients with thrombotic antiphospholipid syndrome and triple positivity for antiphospholipid antibodies. Despite the caveat that TRAPS was prematurely terminated, in both studies, a higher proportion of patients in the rivaroxaban group than the vitamin K antagonist group had thrombotic events, most of which were arterial, whether considering MI or stroke. Furthermore, both studies did not show noninferiority of rivaroxaban versus dose-adjusted vitamin K antagonists.
The reasons for this failure of noninferiority remain unclear.
Denis Wahl, MD, PhD, and Virginie Dufrost, MD, are with the University of Lorraine, Nancy, France, and the Centre Hospitalier Universitaire de Nancy. No conflicts of interest were reported. His remarks are adapted from an accompanying editorial (Ann Intern Med. 2019 Oct 15. doi: 10.7326/M19-2815).
suggests a recent trial conducted in Spain.
Stroke was also more common among those taking rivaroxaban, while major bleeding was slightly less common, reported lead author Josep Ordi-Ros, MD, PhD, of Vall d’Hebrón University Hospital Research Institute in Barcelona, and colleagues in Annals of Internal Medicine.
“Two randomized, controlled trials comparing rivaroxaban with warfarin suggested that rivaroxaban may be efficacious in patients with previous venous thromboembolism who are receiving standard-intensity anticoagulation but showed an increased thrombotic risk in those with triple-positive antiphospholipid antibodies,” the investigators wrote. However, they also noted that these findings required a cautious interpretation because of study limitations, such as premature termination caused by an excess of study events and the use of a laboratory surrogate marker as a primary outcome.
To learn more, the investigators performed an open-label, phase 3 trial involving 190 patients with thrombotic APS. Patients were randomized in a 1:1 ratio to receive either rivaroxaban (20 mg per day, or 15 mg per day for patients with a creatinine clearance of 30-49 mL/min per 1.73 m2) or an adjusted dosage of vitamin K antagonists (target international normalized ratio of 2.0-3.0, or 3.1-4.0 for those with a history of recurrent thrombosis).
Patients underwent evaluations every month for the first 3 months and then every 3 months thereafter, each of which involved a variety of laboratory diagnostics such as checks for antinuclear antibodies and lupus anticoagulant, among others. Statistical analyses aimed to determine if rivaroxaban was noninferior to therapy with vitamin K antagonists based on parameters drawn from previous meta-analyses, as no studies had compared the two types of treatment when the present study was designed.
After 3 years of follow-up, almost twice as many patients in the rivaroxaban group had experienced recurrent thrombosis (11.6% vs. 6.3%), although this finding lacked statistical significance for both noninferiority of rivaroxaban (P = .29) and superiority of vitamin K antagonists (P = .20). Still, supporting a similar trend toward differences in efficacy, stroke was more common in the rivaroxaban group, in which nine events occurred, compared with none in the vitamin K antagonist group. In contrast, major bleeding was slightly less common with rivaroxaban than vitamin K antagonists (6.3% vs. 7.4%).
“In conclusion, rivaroxaban did not demonstrate noninferiority to dose-adjusted vitamin K antagonists for secondary thromboprophylaxis in patients with thrombotic APS,” the investigators wrote. “Instead, our results indicate a recurrent thrombotic rate that is nearly double, albeit without statistical significance.”
The study was funded by Bayer Hispania. One coauthor reported additional relationships with Pfizer, Lilly, Janssen, and others.
SOURCE: Ordi-Ros J et al. Ann Intern Med. 2019 Oct 15. doi: 10.7326/M19-0291.
suggests a recent trial conducted in Spain.
Stroke was also more common among those taking rivaroxaban, while major bleeding was slightly less common, reported lead author Josep Ordi-Ros, MD, PhD, of Vall d’Hebrón University Hospital Research Institute in Barcelona, and colleagues in Annals of Internal Medicine.
“Two randomized, controlled trials comparing rivaroxaban with warfarin suggested that rivaroxaban may be efficacious in patients with previous venous thromboembolism who are receiving standard-intensity anticoagulation but showed an increased thrombotic risk in those with triple-positive antiphospholipid antibodies,” the investigators wrote. However, they also noted that these findings required a cautious interpretation because of study limitations, such as premature termination caused by an excess of study events and the use of a laboratory surrogate marker as a primary outcome.
To learn more, the investigators performed an open-label, phase 3 trial involving 190 patients with thrombotic APS. Patients were randomized in a 1:1 ratio to receive either rivaroxaban (20 mg per day, or 15 mg per day for patients with a creatinine clearance of 30-49 mL/min per 1.73 m2) or an adjusted dosage of vitamin K antagonists (target international normalized ratio of 2.0-3.0, or 3.1-4.0 for those with a history of recurrent thrombosis).
Patients underwent evaluations every month for the first 3 months and then every 3 months thereafter, each of which involved a variety of laboratory diagnostics such as checks for antinuclear antibodies and lupus anticoagulant, among others. Statistical analyses aimed to determine if rivaroxaban was noninferior to therapy with vitamin K antagonists based on parameters drawn from previous meta-analyses, as no studies had compared the two types of treatment when the present study was designed.
After 3 years of follow-up, almost twice as many patients in the rivaroxaban group had experienced recurrent thrombosis (11.6% vs. 6.3%), although this finding lacked statistical significance for both noninferiority of rivaroxaban (P = .29) and superiority of vitamin K antagonists (P = .20). Still, supporting a similar trend toward differences in efficacy, stroke was more common in the rivaroxaban group, in which nine events occurred, compared with none in the vitamin K antagonist group. In contrast, major bleeding was slightly less common with rivaroxaban than vitamin K antagonists (6.3% vs. 7.4%).
“In conclusion, rivaroxaban did not demonstrate noninferiority to dose-adjusted vitamin K antagonists for secondary thromboprophylaxis in patients with thrombotic APS,” the investigators wrote. “Instead, our results indicate a recurrent thrombotic rate that is nearly double, albeit without statistical significance.”
The study was funded by Bayer Hispania. One coauthor reported additional relationships with Pfizer, Lilly, Janssen, and others.
SOURCE: Ordi-Ros J et al. Ann Intern Med. 2019 Oct 15. doi: 10.7326/M19-0291.
FROM ANNALS OF INTERNAL MEDICINE
Readmission burden high for those with sickle cell disease
, according to the Agency for Healthcare Research and Quality.
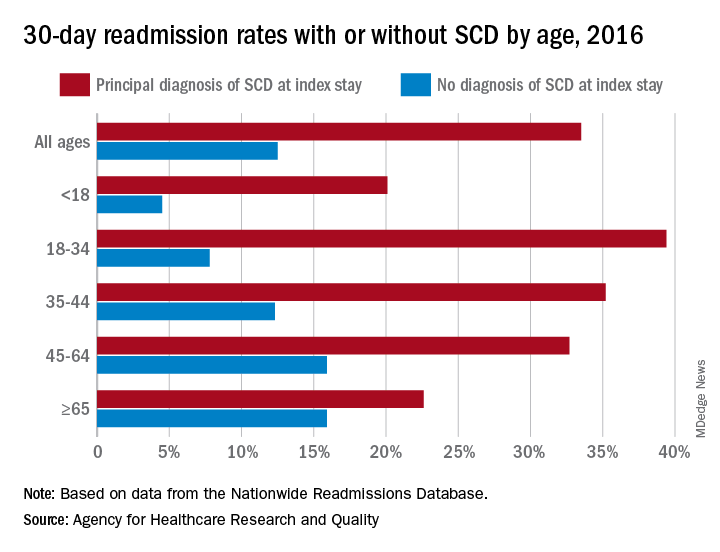
The 30-day all-cause readmission rate for index stays with a principal diagnosis of SCD was 33.5% in 2016, compared with 12.5% for non-SCD hospital stays. Patients with a secondary diagnosis of SCD had readmission rates of 32.9% with a pain crisis and 21.0% without one, and the overall readmission rate for an index stay with any SCD diagnosis was 31.1%, Kathryn R. Fingar, PhD, MPH, of IBM Watson Health, Sacramento, Calif., and associates wrote in an AHRQ statistical brief.
When age is factored in, the readmission gap between a principal SCD diagnosis and non-SCD becomes even greater – and smaller. The difference was greatest for patients aged 18-34 years – 39.4% with a principal diagnosis of SCD versus 7.8% without any SCD – and then narrowed as patients got older. For those aged 65 years and older, the rates were 22.6% with a principal diagnosis of SCD and 15.9% without, the investigators reported.
The approximately 100,000 Americans with SCD accounted for 134,000 admissions in 2016, and more than three-quarters of those stays involved a pain crisis. A principal diagnosis of SCD was recorded for almost 96,000 of those visits, and nearly all (96%) of those stays involved a pain crisis. For those with a secondary diagnosis of SCD, the most common reasons for hospitalization were diseases of the respiratory system (14.3% of those stays) and infectious and parasitic diseases (13.2%).
Patients with SCD were more likely than non-SCD patients to be admitted from the ED (79.6% vs. 51.3%), and they were more likely to discharged against medical advice (4.1% vs. 1.2%). Among those who left the hospital against medical advice, patients with SCD were much more likely to be readmitted than those without SCD (46.6% vs. 26.5%), based on data from the AHRQ’s Nationwide Readmissions Database.
Improved treatment of complications has “reduced mortality rates so that nearly 95% of individuals born with SCD in the United States reach 18 years of age [but] limited knowledge of SCD treatment guidelines among healthcare professionals continues to pose a barrier to effective patient-provider relationships, and this barrier contributes to lower quality of life,” Dr. Fingar and associates wrote.
, according to the Agency for Healthcare Research and Quality.

The 30-day all-cause readmission rate for index stays with a principal diagnosis of SCD was 33.5% in 2016, compared with 12.5% for non-SCD hospital stays. Patients with a secondary diagnosis of SCD had readmission rates of 32.9% with a pain crisis and 21.0% without one, and the overall readmission rate for an index stay with any SCD diagnosis was 31.1%, Kathryn R. Fingar, PhD, MPH, of IBM Watson Health, Sacramento, Calif., and associates wrote in an AHRQ statistical brief.
When age is factored in, the readmission gap between a principal SCD diagnosis and non-SCD becomes even greater – and smaller. The difference was greatest for patients aged 18-34 years – 39.4% with a principal diagnosis of SCD versus 7.8% without any SCD – and then narrowed as patients got older. For those aged 65 years and older, the rates were 22.6% with a principal diagnosis of SCD and 15.9% without, the investigators reported.
The approximately 100,000 Americans with SCD accounted for 134,000 admissions in 2016, and more than three-quarters of those stays involved a pain crisis. A principal diagnosis of SCD was recorded for almost 96,000 of those visits, and nearly all (96%) of those stays involved a pain crisis. For those with a secondary diagnosis of SCD, the most common reasons for hospitalization were diseases of the respiratory system (14.3% of those stays) and infectious and parasitic diseases (13.2%).
Patients with SCD were more likely than non-SCD patients to be admitted from the ED (79.6% vs. 51.3%), and they were more likely to discharged against medical advice (4.1% vs. 1.2%). Among those who left the hospital against medical advice, patients with SCD were much more likely to be readmitted than those without SCD (46.6% vs. 26.5%), based on data from the AHRQ’s Nationwide Readmissions Database.
Improved treatment of complications has “reduced mortality rates so that nearly 95% of individuals born with SCD in the United States reach 18 years of age [but] limited knowledge of SCD treatment guidelines among healthcare professionals continues to pose a barrier to effective patient-provider relationships, and this barrier contributes to lower quality of life,” Dr. Fingar and associates wrote.
, according to the Agency for Healthcare Research and Quality.

The 30-day all-cause readmission rate for index stays with a principal diagnosis of SCD was 33.5% in 2016, compared with 12.5% for non-SCD hospital stays. Patients with a secondary diagnosis of SCD had readmission rates of 32.9% with a pain crisis and 21.0% without one, and the overall readmission rate for an index stay with any SCD diagnosis was 31.1%, Kathryn R. Fingar, PhD, MPH, of IBM Watson Health, Sacramento, Calif., and associates wrote in an AHRQ statistical brief.
When age is factored in, the readmission gap between a principal SCD diagnosis and non-SCD becomes even greater – and smaller. The difference was greatest for patients aged 18-34 years – 39.4% with a principal diagnosis of SCD versus 7.8% without any SCD – and then narrowed as patients got older. For those aged 65 years and older, the rates were 22.6% with a principal diagnosis of SCD and 15.9% without, the investigators reported.
The approximately 100,000 Americans with SCD accounted for 134,000 admissions in 2016, and more than three-quarters of those stays involved a pain crisis. A principal diagnosis of SCD was recorded for almost 96,000 of those visits, and nearly all (96%) of those stays involved a pain crisis. For those with a secondary diagnosis of SCD, the most common reasons for hospitalization were diseases of the respiratory system (14.3% of those stays) and infectious and parasitic diseases (13.2%).
Patients with SCD were more likely than non-SCD patients to be admitted from the ED (79.6% vs. 51.3%), and they were more likely to discharged against medical advice (4.1% vs. 1.2%). Among those who left the hospital against medical advice, patients with SCD were much more likely to be readmitted than those without SCD (46.6% vs. 26.5%), based on data from the AHRQ’s Nationwide Readmissions Database.
Improved treatment of complications has “reduced mortality rates so that nearly 95% of individuals born with SCD in the United States reach 18 years of age [but] limited knowledge of SCD treatment guidelines among healthcare professionals continues to pose a barrier to effective patient-provider relationships, and this barrier contributes to lower quality of life,” Dr. Fingar and associates wrote.
Monthly and twice monthly emicizumab dosing safe for children with severe hemophilia A
Administration of twice-monthly or monthly emicizumab appears safe and effective for children with severe hemophilia A without inhibitors, according to a small cohort study.
After 24 weeks of treatment, only one moderate-intensity injection site reaction was reported, but no thrombotic microangiopathy or thromboembolic complications were observed.
The researchers evaluated the efficacy, safety, and pharmacokinetics of emicizumab in Japanese pediatric patients aged less than 12 years with severe hemophilia A without factor VIII inhibitors, wrote Midori Shima, MD, PhD, of Nara Medical University, Kashihara, Japan, and colleagues. The results were published in Haemophilia.
The open-label, nonrandomized study included 13 children who initially received weekly loading doses (3 mg/kg) of subcutaneous emicizumab for 4 weeks. Subsequently, patients received maintenance doses of 3 mg/kg every 2 weeks or 6 mg/kg every 4 weeks until week 24.
At baseline, the median age of patients in the 2- and 4-week dosing cohorts were 6.6 and 4.1 years, respectively. All participants had received factor VIII prophylaxis prior to starting emicizumab, with the exception of one patient.
Among six patients in the twice-monthly dosing cohort, two had no treated bleeding episodes, with an annualized bleeding rate for treated bleeding episodes of 1.3 (95% confidence interval, 0.6-2.9).
Among seven patients in the monthly dosing cohort, five had no treated bleeding episodes, with an annualized bleeding rate for treated bleeding episodes of 0.7 (95% CI, 0.2-2.6).
Caregivers completed a preference survey after the first 16 weeks of treatment, and “all reported a preference for emicizumab prophylaxis over the patient’s previous haemophilia treatment.” They cited the lower frequency of treatment and easier route of administration for favoring emicizumab.
With respect to pharmacokinetics, mean steady-state trough levels were within acceptable limits based on previous studies. No patients tested positive for anti-emicizumab antibodies.
The small sample size and nonrandomized design were key limitations of the study.
The results “confirm the appropriateness” of applying the every 2-week and every 4-week regimens of emicizumab in pediatric patients with hemophilia A without inhibitors, the researchers wrote.
The authors reported having financial affiliations with Chugai Pharmaceutical Co., which funded the study, and other companies.
SOURCE: Shima M et al. Haemophilia. 2019 Sep 12. doi: 10.1111/hae.13848.
Administration of twice-monthly or monthly emicizumab appears safe and effective for children with severe hemophilia A without inhibitors, according to a small cohort study.
After 24 weeks of treatment, only one moderate-intensity injection site reaction was reported, but no thrombotic microangiopathy or thromboembolic complications were observed.
The researchers evaluated the efficacy, safety, and pharmacokinetics of emicizumab in Japanese pediatric patients aged less than 12 years with severe hemophilia A without factor VIII inhibitors, wrote Midori Shima, MD, PhD, of Nara Medical University, Kashihara, Japan, and colleagues. The results were published in Haemophilia.
The open-label, nonrandomized study included 13 children who initially received weekly loading doses (3 mg/kg) of subcutaneous emicizumab for 4 weeks. Subsequently, patients received maintenance doses of 3 mg/kg every 2 weeks or 6 mg/kg every 4 weeks until week 24.
At baseline, the median age of patients in the 2- and 4-week dosing cohorts were 6.6 and 4.1 years, respectively. All participants had received factor VIII prophylaxis prior to starting emicizumab, with the exception of one patient.
Among six patients in the twice-monthly dosing cohort, two had no treated bleeding episodes, with an annualized bleeding rate for treated bleeding episodes of 1.3 (95% confidence interval, 0.6-2.9).
Among seven patients in the monthly dosing cohort, five had no treated bleeding episodes, with an annualized bleeding rate for treated bleeding episodes of 0.7 (95% CI, 0.2-2.6).
Caregivers completed a preference survey after the first 16 weeks of treatment, and “all reported a preference for emicizumab prophylaxis over the patient’s previous haemophilia treatment.” They cited the lower frequency of treatment and easier route of administration for favoring emicizumab.
With respect to pharmacokinetics, mean steady-state trough levels were within acceptable limits based on previous studies. No patients tested positive for anti-emicizumab antibodies.
The small sample size and nonrandomized design were key limitations of the study.
The results “confirm the appropriateness” of applying the every 2-week and every 4-week regimens of emicizumab in pediatric patients with hemophilia A without inhibitors, the researchers wrote.
The authors reported having financial affiliations with Chugai Pharmaceutical Co., which funded the study, and other companies.
SOURCE: Shima M et al. Haemophilia. 2019 Sep 12. doi: 10.1111/hae.13848.
Administration of twice-monthly or monthly emicizumab appears safe and effective for children with severe hemophilia A without inhibitors, according to a small cohort study.
After 24 weeks of treatment, only one moderate-intensity injection site reaction was reported, but no thrombotic microangiopathy or thromboembolic complications were observed.
The researchers evaluated the efficacy, safety, and pharmacokinetics of emicizumab in Japanese pediatric patients aged less than 12 years with severe hemophilia A without factor VIII inhibitors, wrote Midori Shima, MD, PhD, of Nara Medical University, Kashihara, Japan, and colleagues. The results were published in Haemophilia.
The open-label, nonrandomized study included 13 children who initially received weekly loading doses (3 mg/kg) of subcutaneous emicizumab for 4 weeks. Subsequently, patients received maintenance doses of 3 mg/kg every 2 weeks or 6 mg/kg every 4 weeks until week 24.
At baseline, the median age of patients in the 2- and 4-week dosing cohorts were 6.6 and 4.1 years, respectively. All participants had received factor VIII prophylaxis prior to starting emicizumab, with the exception of one patient.
Among six patients in the twice-monthly dosing cohort, two had no treated bleeding episodes, with an annualized bleeding rate for treated bleeding episodes of 1.3 (95% confidence interval, 0.6-2.9).
Among seven patients in the monthly dosing cohort, five had no treated bleeding episodes, with an annualized bleeding rate for treated bleeding episodes of 0.7 (95% CI, 0.2-2.6).
Caregivers completed a preference survey after the first 16 weeks of treatment, and “all reported a preference for emicizumab prophylaxis over the patient’s previous haemophilia treatment.” They cited the lower frequency of treatment and easier route of administration for favoring emicizumab.
With respect to pharmacokinetics, mean steady-state trough levels were within acceptable limits based on previous studies. No patients tested positive for anti-emicizumab antibodies.
The small sample size and nonrandomized design were key limitations of the study.
The results “confirm the appropriateness” of applying the every 2-week and every 4-week regimens of emicizumab in pediatric patients with hemophilia A without inhibitors, the researchers wrote.
The authors reported having financial affiliations with Chugai Pharmaceutical Co., which funded the study, and other companies.
SOURCE: Shima M et al. Haemophilia. 2019 Sep 12. doi: 10.1111/hae.13848.
FROM HAEMOPHILIA