User login
FDA OKs New Drug for Urinary Tract Infections
The Food and Drug Administration (FDA) has approved Orlynvah, a new oral treatment for uncomplicated urinary tract infections (UTIs) in women who have limited options for effective antibiotic therapy.
Uncomplicated UTIs are bladder infections that typically affect women who don’t have other issues like kidney disease or urinary tract abnormalities. These infections are common, affecting around half of all women at least once in their lives.
Treating UTIs can be difficult when standard antibiotics don’t work well, often because of antibiotic resistance or certain health conditions. Orlynvah offers a promising new option by combining two drugs, sulopenem etzadroxil and probenecid, in one oral tablet. This combination helps keep the antibiotic in the body longer, making it work better, especially against bacteria that resist traditional treatments. Orlynvah is specifically approved to target infections from bacteria like Escherichia coli, Klebsiella pneumoniae, and Proteus mirabilis, which can be harder to treat.
Marjorie Golden, MD, an infectious disease specialist at Yale New Haven Hospital in Connecticut, described Orlynvah as a much-needed alternative for women struggling with difficult-to-treat UTIs.
“Orlynvah has the potential to be an important treatment option for those who need it,” she said in a news release from Iterum Therapeutics, the drug’s maker.
The FDA approved Orlynvah based on two large clinical trials involving over 3,800 women. In these studies, Orlynvah worked as well as or better than antibiotics like ciprofloxacin and Augmentin. It was generally well-tolerated, though common side effects included diarrhea, nausea, yeast infections, and headaches.
The FDA advises people to discuss their medical history with their doctor before taking Orlynvah, especially if they have conditions like gout, kidney stones, or allergies to other antibiotics.
A version of this article first appeared on Medscape.com.
The Food and Drug Administration (FDA) has approved Orlynvah, a new oral treatment for uncomplicated urinary tract infections (UTIs) in women who have limited options for effective antibiotic therapy.
Uncomplicated UTIs are bladder infections that typically affect women who don’t have other issues like kidney disease or urinary tract abnormalities. These infections are common, affecting around half of all women at least once in their lives.
Treating UTIs can be difficult when standard antibiotics don’t work well, often because of antibiotic resistance or certain health conditions. Orlynvah offers a promising new option by combining two drugs, sulopenem etzadroxil and probenecid, in one oral tablet. This combination helps keep the antibiotic in the body longer, making it work better, especially against bacteria that resist traditional treatments. Orlynvah is specifically approved to target infections from bacteria like Escherichia coli, Klebsiella pneumoniae, and Proteus mirabilis, which can be harder to treat.
Marjorie Golden, MD, an infectious disease specialist at Yale New Haven Hospital in Connecticut, described Orlynvah as a much-needed alternative for women struggling with difficult-to-treat UTIs.
“Orlynvah has the potential to be an important treatment option for those who need it,” she said in a news release from Iterum Therapeutics, the drug’s maker.
The FDA approved Orlynvah based on two large clinical trials involving over 3,800 women. In these studies, Orlynvah worked as well as or better than antibiotics like ciprofloxacin and Augmentin. It was generally well-tolerated, though common side effects included diarrhea, nausea, yeast infections, and headaches.
The FDA advises people to discuss their medical history with their doctor before taking Orlynvah, especially if they have conditions like gout, kidney stones, or allergies to other antibiotics.
A version of this article first appeared on Medscape.com.
The Food and Drug Administration (FDA) has approved Orlynvah, a new oral treatment for uncomplicated urinary tract infections (UTIs) in women who have limited options for effective antibiotic therapy.
Uncomplicated UTIs are bladder infections that typically affect women who don’t have other issues like kidney disease or urinary tract abnormalities. These infections are common, affecting around half of all women at least once in their lives.
Treating UTIs can be difficult when standard antibiotics don’t work well, often because of antibiotic resistance or certain health conditions. Orlynvah offers a promising new option by combining two drugs, sulopenem etzadroxil and probenecid, in one oral tablet. This combination helps keep the antibiotic in the body longer, making it work better, especially against bacteria that resist traditional treatments. Orlynvah is specifically approved to target infections from bacteria like Escherichia coli, Klebsiella pneumoniae, and Proteus mirabilis, which can be harder to treat.
Marjorie Golden, MD, an infectious disease specialist at Yale New Haven Hospital in Connecticut, described Orlynvah as a much-needed alternative for women struggling with difficult-to-treat UTIs.
“Orlynvah has the potential to be an important treatment option for those who need it,” she said in a news release from Iterum Therapeutics, the drug’s maker.
The FDA approved Orlynvah based on two large clinical trials involving over 3,800 women. In these studies, Orlynvah worked as well as or better than antibiotics like ciprofloxacin and Augmentin. It was generally well-tolerated, though common side effects included diarrhea, nausea, yeast infections, and headaches.
The FDA advises people to discuss their medical history with their doctor before taking Orlynvah, especially if they have conditions like gout, kidney stones, or allergies to other antibiotics.
A version of this article first appeared on Medscape.com.
Increase in Troublesome Fungal Infections Requires All-Out Approach
As dermatologists, public health officials, and infectious disease specialists scramble to raise awareness about prevention and treatment, challenges ranging from a dearth of testing facilities and data to payer pushback over longer therapeutic courses remain.
Dermatophyte Discourse Changing
“Trichophyton indotineae is changing the way we talk about dermatophyte infections,” Avrom S. Caplan, MD, assistant professor in the Department of Dermatology at New York University, New York City, said in an interview. Called T mentagrophytes VIII (TMVIII) before a 2020 report in the journal Mycopathologia proposed the name T indotineae, this species requires clinicians to expand their conception of how tinea looks, acts, and responds to treatment.
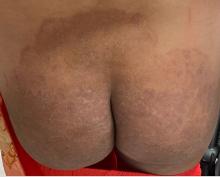
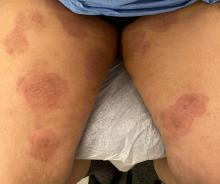
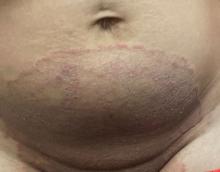
Boni E. Elewski, MD, professor and chair of dermatology, at The University of Alabama at Birmingham, saw her first case of probable T indotineae in a patient in early 2020. “He was covered with fine scale, and he itched all over. I thought he had atopic dermatitis. This didn’t look like any tinea. His face, arms, back, and legs were scaly.”
Nevertheless, KOH and biopsy confirmed dermatophytosis. Culture (performed at the Center for Medical Mycology [CMM] in Cleveland) identified T mentagrophytes. Back then, Elewski told this news organization, labs did not routinely go beyond genus and species. But based on the patient’s symptoms, history of unresponsiveness to terbinafine, borderline sensitivity to fluconazole, and travel to India and Spain, Elewski strongly suspected T indotineae.
The patient refused itraconazole, to which the fungus was sensitive, and did not respond to fluconazole 400 mg daily. Ultimately, he was lost to follow-up. “Last I saw him,” said Elewski, “he was not cured.”
Tracking Cases
Because T indotineae does not require reporting to public health agencies, said Jeremy Gold, MD, MS, a medical officer with the US Centers for Disease Control and Prevention (CDC) Mycotic Diseases Branch in Atlanta, “there is no official public health surveillance keeping track of exactly how many cases have occurred.”
The same is true for TMVII and terbinafine-resistant T rubrum, which are also on the rise. Regarding T indotineae, authors from the University of Texas Health Science Center at San Antonio retrospectively reported 21 terbinafine-resistant isolates from North America in the July 2023 Journal of Clinical Microbiology .
Caplan has seen approximately 12 T indotineae cases to date, including the first two confirmed US cases, which he and co-authors, including Gold, reported in the CDC’s Morbidity and Mortality Weekly Report in May 2023. T indotineae is likely underreported, he said, because it eludes standard culture-based techniques, and identifying it requires molecular testing, which is available at only a handful of labs nationally.
To help educate providers, in July, the American Academy of Dermatology (AAD) and the International League of Dermatological Societies unveiled an Emerging Diseases Resource Center, which includes resources for providers and a registry for reporting confirmed and suspected resistant dermatophytes.
“Our goal is to provide easy-to-access and easy-to-understand resources to healthcare providers,” Esther Freeman, MD, PhD, told this news organization. She is director of Global Health Dermatology at Massachusetts General Hospital, associate professor of dermatology at Harvard Medical School, both in Boston, and chair of the AAD’s Emerging Diseases Task Force.
“Our resources include an algorithm for when to suspect a drug-resistant case and how to think through treatment options. We cover issues related to diagnosis and treatment, as well as linking to our case registry reporting system,” said Freeman.
The new registry resides within the AAD’s existing COVID-19, Mpox, and Emerging Infections Registry. “Our registry efforts have already captured 2500 COVID-19 and mpox cases from 72 different countries,” Freeman said. For all these infections, she added, “we hope that real-time data analysis of cases worldwide will provide information that helps physicians recognize and treat cases.”
Consistent with the registry’s approach, said Caplan and Gold, there is no silver bullet for battling dermatophyte resistance. What is needed, said Gold, is a coordinated approach involving public health officials, dermatologists, primary care providers, infectious disease specialists, pharmacists, and patients. “It’s going to be a team effort to address the challenge of emerging complex dermatophytosis,” he said.
Resistant T rubrum
“The biggest difference with T rubrum resistance is you may not see that widespread infection that we see with T indotineae,” said Caplan. T rubrum is probably the most common dermatophyte that dermatologists see, added Elewski, who encounters a resistant case at least monthly. One such patient, featured in a January 2021 British Journal of Dermatology research letter, cleared on itraconazole and ciclopirox cream but subsequently returned with itraconazole-resistant T rubrum because he had been doctor-shopping for the drug intermittently for years, she said. He cleared on posaconazole 300 mg daily, then was lost to follow-up.
TMVII
A 2023 Emerging Infectious Diseases report highlighted the potential for this dermatophyte to spread among men who have sex with men (MSM), presenting as an itchy, scaly rash affecting the pubic, genital, and buttocks skin. “People don’t generally think of a fungal infection as something that could behave like a sexually transmitted infection (STI),” said Gold.
Caplan and coauthors recently reported the first confirmed US TMVII case in JAMA Dermatology. Many experts suspect that unreported US cases existed previously, he said. “When it circulates in Europe and there’s so much travel, it’s probably here too.”
The fact that T indotineae was formerly called TMVIII has created confusion, added Caplan. “I’ve had patients say, ‘I’m worried I have that resistant ringworm that’s spreading among MSM.’ Whenever we talk about STIs and introduce the word ‘resistant,’ that comes with the potential for stigma, anxiety, and concern.” Fortunately, he said, TMVII has shown no resistance to first-line antifungals.
Why the Rise
Gold said, “We don’t know for sure why we’re seeing these different drug-resistant species popping up.” One possibility, he said, is the common misuse and overuse of topical antifungals — especially those available overseas in combination with high-potency steroids, such as clobetasol. Consumers use these products for a few weeks until symptoms resolve, then reapply them off and on over years, fueling resistance, said Gold.
“We are worried that with warming temperatures, there’s potential to see expansion of the geographic range of epidemic fungi,” he added. “That could be part of what has fueled recent increases in resistant dermatophytes. But it’s hard to prove.”
Climate change may be behind the emergence of Candida auris, according to a 2022 article in The Lancet Regional Health – Americas. This potentially fatal multidrug-resistant infection spreads easily among sick patients in healthcare facilities, according to a CDC information page on C auris.
Confirming Dermatophyte Infection
“A biopsy will only confirm the presence of fungus,” said Elewski. “Here you will need a lab that knows how to do a fungal culture.” Most state laboratories can do this, she said, as can some hospitals and special labs such as CMM in Cleveland.
It takes a Clinical Laboratory Improvement Amendments–certified lab to perform KOH prep in-house, added Caplan, plus up-to-date gear and knowledge of where and how to scrape and what to look for microscopically. Moreover, identifying T indotineae requires molecular testing available at only a handful of laboratories — listed on the AAD Emerging Dermatophytes webpage — nationwide.
Nevertheless, said Caplan, nailing down a diagnosis can guide treatment, often supplanting empirically prescribed antifungal steroid creams. “Those are probably not going to help. And people may be using those on areas of the body they shouldn’t. Both the clinical clues and the steps to make the diagnosis need to come together. But that’s often easier said than done, especially in a busy practice.”
Identifying resistance requires antifungal sensitivity testing, he added, which few labs perform. “Practically speaking,” said Elewski, “if the patient failed terbinafine, I would try itraconazole. You don’t necessarily need proof” of resistance. But if a patient does not respond to itraconazole and terbinafine clinically, she said that she might consider fungal susceptibility testing.
Treatment Tips
To address any resistant dermatophyte, Elewski recommended getting comfortable with itraconazole. For decades, she said, dermatologists have avoided itraconazole because terbinafine typically costs patients $10 for 3 months. “Itraconazole could be $200 per month,” said Elewski. Because of potential drug-drug interactions and absorption issues — and a boxed warning regarding congestive heart failure — physicians historically reserved itraconazole for severe fungal infections.
Itraconazole labeled dosing for onychomycosis is 200 mg daily for 12 weeks. Elewski favors a two-pronged attack, often combining an -azole antifungal with topical ciclopirox.
Another element that emerging tinea pathogens share is slower response to treatment. For T indotineae, reports appearing in the Journal of the American Academy of Dermatology in 2022 and 2024 suggest duration from 6-8 weeks up to 20 weeks.
To avoid recurrences of resistant T rubrum, Elewski treats for a year. However, she has problems getting itraconazole approved, when often it is the only agent that works. “I’ve written more letters than I like to insurance companies” to document terbinafine failure, she said.
Rarely, said Gold, dermatophyte infections resist both terbinafine and itraconazole. Next-line agents such as voriconazole, which some dermatologists have used for resistant T indotineae, can be much harder to tolerate, with more drug interactions, he said.
And because itraconazole, voriconazole, and posaconazole are all triazoles, added Elewski, the latter two might not work better than the former. But because these drugs might outperform itraconazole in selected cases, she said, “that’s when you want to do fungal susceptibility testing.”
TMVII is so new, said Caplan, that optimal therapy duration remains unclear. “One of the challenges with TMVII is when it gets into the genital skin, it’s a hair-bearing area. And based on various grooming practices, there’s an opportunity for the tinea to get deeper into the hair follicle and dermis. That may also be true of T indotineae.”
Anemic Arsenal
Unfortunately, said Gold, the arsenal of antifungals available in the United States remains limited. “Depending on how you count, there are only three to four classes of antifungal drugs designed to treat severe or invasive infections. So whenever we hear about a new fungal pathogen that’s causing resistant infections, it causes public health concern.”
Promising drugs in development include olorofim (F2G) and fosmanogepix (Basilea), according to Gold. However, he said, the development of these drugs to date has targeted invasive fungal infections such as aspergillosis. In June 2023, the Food and Drug Administration rejected the new drug application for olorofim, requesting additional data and analyses. Regarding fosmanogepix, a double-blinded noninferiority phase 3 trial in invasive yeast infections was recently launched, according to a September 24 press release.
Gold, Caplan, and Elewski reported no relevant financial disclosures. Freeman is a COVID-19 co-author for UpToDate and chair of the AAD Emerging Diseases Task Force.
A version of this article appeared on Medscape.com.
As dermatologists, public health officials, and infectious disease specialists scramble to raise awareness about prevention and treatment, challenges ranging from a dearth of testing facilities and data to payer pushback over longer therapeutic courses remain.
Dermatophyte Discourse Changing
“Trichophyton indotineae is changing the way we talk about dermatophyte infections,” Avrom S. Caplan, MD, assistant professor in the Department of Dermatology at New York University, New York City, said in an interview. Called T mentagrophytes VIII (TMVIII) before a 2020 report in the journal Mycopathologia proposed the name T indotineae, this species requires clinicians to expand their conception of how tinea looks, acts, and responds to treatment.
Boni E. Elewski, MD, professor and chair of dermatology, at The University of Alabama at Birmingham, saw her first case of probable T indotineae in a patient in early 2020. “He was covered with fine scale, and he itched all over. I thought he had atopic dermatitis. This didn’t look like any tinea. His face, arms, back, and legs were scaly.”
Nevertheless, KOH and biopsy confirmed dermatophytosis. Culture (performed at the Center for Medical Mycology [CMM] in Cleveland) identified T mentagrophytes. Back then, Elewski told this news organization, labs did not routinely go beyond genus and species. But based on the patient’s symptoms, history of unresponsiveness to terbinafine, borderline sensitivity to fluconazole, and travel to India and Spain, Elewski strongly suspected T indotineae.
The patient refused itraconazole, to which the fungus was sensitive, and did not respond to fluconazole 400 mg daily. Ultimately, he was lost to follow-up. “Last I saw him,” said Elewski, “he was not cured.”
Tracking Cases
Because T indotineae does not require reporting to public health agencies, said Jeremy Gold, MD, MS, a medical officer with the US Centers for Disease Control and Prevention (CDC) Mycotic Diseases Branch in Atlanta, “there is no official public health surveillance keeping track of exactly how many cases have occurred.”
The same is true for TMVII and terbinafine-resistant T rubrum, which are also on the rise. Regarding T indotineae, authors from the University of Texas Health Science Center at San Antonio retrospectively reported 21 terbinafine-resistant isolates from North America in the July 2023 Journal of Clinical Microbiology .
Caplan has seen approximately 12 T indotineae cases to date, including the first two confirmed US cases, which he and co-authors, including Gold, reported in the CDC’s Morbidity and Mortality Weekly Report in May 2023. T indotineae is likely underreported, he said, because it eludes standard culture-based techniques, and identifying it requires molecular testing, which is available at only a handful of labs nationally.
To help educate providers, in July, the American Academy of Dermatology (AAD) and the International League of Dermatological Societies unveiled an Emerging Diseases Resource Center, which includes resources for providers and a registry for reporting confirmed and suspected resistant dermatophytes.
“Our goal is to provide easy-to-access and easy-to-understand resources to healthcare providers,” Esther Freeman, MD, PhD, told this news organization. She is director of Global Health Dermatology at Massachusetts General Hospital, associate professor of dermatology at Harvard Medical School, both in Boston, and chair of the AAD’s Emerging Diseases Task Force.
“Our resources include an algorithm for when to suspect a drug-resistant case and how to think through treatment options. We cover issues related to diagnosis and treatment, as well as linking to our case registry reporting system,” said Freeman.
The new registry resides within the AAD’s existing COVID-19, Mpox, and Emerging Infections Registry. “Our registry efforts have already captured 2500 COVID-19 and mpox cases from 72 different countries,” Freeman said. For all these infections, she added, “we hope that real-time data analysis of cases worldwide will provide information that helps physicians recognize and treat cases.”
Consistent with the registry’s approach, said Caplan and Gold, there is no silver bullet for battling dermatophyte resistance. What is needed, said Gold, is a coordinated approach involving public health officials, dermatologists, primary care providers, infectious disease specialists, pharmacists, and patients. “It’s going to be a team effort to address the challenge of emerging complex dermatophytosis,” he said.
Resistant T rubrum
“The biggest difference with T rubrum resistance is you may not see that widespread infection that we see with T indotineae,” said Caplan. T rubrum is probably the most common dermatophyte that dermatologists see, added Elewski, who encounters a resistant case at least monthly. One such patient, featured in a January 2021 British Journal of Dermatology research letter, cleared on itraconazole and ciclopirox cream but subsequently returned with itraconazole-resistant T rubrum because he had been doctor-shopping for the drug intermittently for years, she said. He cleared on posaconazole 300 mg daily, then was lost to follow-up.
TMVII
A 2023 Emerging Infectious Diseases report highlighted the potential for this dermatophyte to spread among men who have sex with men (MSM), presenting as an itchy, scaly rash affecting the pubic, genital, and buttocks skin. “People don’t generally think of a fungal infection as something that could behave like a sexually transmitted infection (STI),” said Gold.
Caplan and coauthors recently reported the first confirmed US TMVII case in JAMA Dermatology. Many experts suspect that unreported US cases existed previously, he said. “When it circulates in Europe and there’s so much travel, it’s probably here too.”
The fact that T indotineae was formerly called TMVIII has created confusion, added Caplan. “I’ve had patients say, ‘I’m worried I have that resistant ringworm that’s spreading among MSM.’ Whenever we talk about STIs and introduce the word ‘resistant,’ that comes with the potential for stigma, anxiety, and concern.” Fortunately, he said, TMVII has shown no resistance to first-line antifungals.
Why the Rise
Gold said, “We don’t know for sure why we’re seeing these different drug-resistant species popping up.” One possibility, he said, is the common misuse and overuse of topical antifungals — especially those available overseas in combination with high-potency steroids, such as clobetasol. Consumers use these products for a few weeks until symptoms resolve, then reapply them off and on over years, fueling resistance, said Gold.
“We are worried that with warming temperatures, there’s potential to see expansion of the geographic range of epidemic fungi,” he added. “That could be part of what has fueled recent increases in resistant dermatophytes. But it’s hard to prove.”
Climate change may be behind the emergence of Candida auris, according to a 2022 article in The Lancet Regional Health – Americas. This potentially fatal multidrug-resistant infection spreads easily among sick patients in healthcare facilities, according to a CDC information page on C auris.
Confirming Dermatophyte Infection
“A biopsy will only confirm the presence of fungus,” said Elewski. “Here you will need a lab that knows how to do a fungal culture.” Most state laboratories can do this, she said, as can some hospitals and special labs such as CMM in Cleveland.
It takes a Clinical Laboratory Improvement Amendments–certified lab to perform KOH prep in-house, added Caplan, plus up-to-date gear and knowledge of where and how to scrape and what to look for microscopically. Moreover, identifying T indotineae requires molecular testing available at only a handful of laboratories — listed on the AAD Emerging Dermatophytes webpage — nationwide.
Nevertheless, said Caplan, nailing down a diagnosis can guide treatment, often supplanting empirically prescribed antifungal steroid creams. “Those are probably not going to help. And people may be using those on areas of the body they shouldn’t. Both the clinical clues and the steps to make the diagnosis need to come together. But that’s often easier said than done, especially in a busy practice.”
Identifying resistance requires antifungal sensitivity testing, he added, which few labs perform. “Practically speaking,” said Elewski, “if the patient failed terbinafine, I would try itraconazole. You don’t necessarily need proof” of resistance. But if a patient does not respond to itraconazole and terbinafine clinically, she said that she might consider fungal susceptibility testing.
Treatment Tips
To address any resistant dermatophyte, Elewski recommended getting comfortable with itraconazole. For decades, she said, dermatologists have avoided itraconazole because terbinafine typically costs patients $10 for 3 months. “Itraconazole could be $200 per month,” said Elewski. Because of potential drug-drug interactions and absorption issues — and a boxed warning regarding congestive heart failure — physicians historically reserved itraconazole for severe fungal infections.
Itraconazole labeled dosing for onychomycosis is 200 mg daily for 12 weeks. Elewski favors a two-pronged attack, often combining an -azole antifungal with topical ciclopirox.
Another element that emerging tinea pathogens share is slower response to treatment. For T indotineae, reports appearing in the Journal of the American Academy of Dermatology in 2022 and 2024 suggest duration from 6-8 weeks up to 20 weeks.
To avoid recurrences of resistant T rubrum, Elewski treats for a year. However, she has problems getting itraconazole approved, when often it is the only agent that works. “I’ve written more letters than I like to insurance companies” to document terbinafine failure, she said.
Rarely, said Gold, dermatophyte infections resist both terbinafine and itraconazole. Next-line agents such as voriconazole, which some dermatologists have used for resistant T indotineae, can be much harder to tolerate, with more drug interactions, he said.
And because itraconazole, voriconazole, and posaconazole are all triazoles, added Elewski, the latter two might not work better than the former. But because these drugs might outperform itraconazole in selected cases, she said, “that’s when you want to do fungal susceptibility testing.”
TMVII is so new, said Caplan, that optimal therapy duration remains unclear. “One of the challenges with TMVII is when it gets into the genital skin, it’s a hair-bearing area. And based on various grooming practices, there’s an opportunity for the tinea to get deeper into the hair follicle and dermis. That may also be true of T indotineae.”
Anemic Arsenal
Unfortunately, said Gold, the arsenal of antifungals available in the United States remains limited. “Depending on how you count, there are only three to four classes of antifungal drugs designed to treat severe or invasive infections. So whenever we hear about a new fungal pathogen that’s causing resistant infections, it causes public health concern.”
Promising drugs in development include olorofim (F2G) and fosmanogepix (Basilea), according to Gold. However, he said, the development of these drugs to date has targeted invasive fungal infections such as aspergillosis. In June 2023, the Food and Drug Administration rejected the new drug application for olorofim, requesting additional data and analyses. Regarding fosmanogepix, a double-blinded noninferiority phase 3 trial in invasive yeast infections was recently launched, according to a September 24 press release.
Gold, Caplan, and Elewski reported no relevant financial disclosures. Freeman is a COVID-19 co-author for UpToDate and chair of the AAD Emerging Diseases Task Force.
A version of this article appeared on Medscape.com.
As dermatologists, public health officials, and infectious disease specialists scramble to raise awareness about prevention and treatment, challenges ranging from a dearth of testing facilities and data to payer pushback over longer therapeutic courses remain.
Dermatophyte Discourse Changing
“Trichophyton indotineae is changing the way we talk about dermatophyte infections,” Avrom S. Caplan, MD, assistant professor in the Department of Dermatology at New York University, New York City, said in an interview. Called T mentagrophytes VIII (TMVIII) before a 2020 report in the journal Mycopathologia proposed the name T indotineae, this species requires clinicians to expand their conception of how tinea looks, acts, and responds to treatment.
Boni E. Elewski, MD, professor and chair of dermatology, at The University of Alabama at Birmingham, saw her first case of probable T indotineae in a patient in early 2020. “He was covered with fine scale, and he itched all over. I thought he had atopic dermatitis. This didn’t look like any tinea. His face, arms, back, and legs were scaly.”
Nevertheless, KOH and biopsy confirmed dermatophytosis. Culture (performed at the Center for Medical Mycology [CMM] in Cleveland) identified T mentagrophytes. Back then, Elewski told this news organization, labs did not routinely go beyond genus and species. But based on the patient’s symptoms, history of unresponsiveness to terbinafine, borderline sensitivity to fluconazole, and travel to India and Spain, Elewski strongly suspected T indotineae.
The patient refused itraconazole, to which the fungus was sensitive, and did not respond to fluconazole 400 mg daily. Ultimately, he was lost to follow-up. “Last I saw him,” said Elewski, “he was not cured.”
Tracking Cases
Because T indotineae does not require reporting to public health agencies, said Jeremy Gold, MD, MS, a medical officer with the US Centers for Disease Control and Prevention (CDC) Mycotic Diseases Branch in Atlanta, “there is no official public health surveillance keeping track of exactly how many cases have occurred.”
The same is true for TMVII and terbinafine-resistant T rubrum, which are also on the rise. Regarding T indotineae, authors from the University of Texas Health Science Center at San Antonio retrospectively reported 21 terbinafine-resistant isolates from North America in the July 2023 Journal of Clinical Microbiology .
Caplan has seen approximately 12 T indotineae cases to date, including the first two confirmed US cases, which he and co-authors, including Gold, reported in the CDC’s Morbidity and Mortality Weekly Report in May 2023. T indotineae is likely underreported, he said, because it eludes standard culture-based techniques, and identifying it requires molecular testing, which is available at only a handful of labs nationally.
To help educate providers, in July, the American Academy of Dermatology (AAD) and the International League of Dermatological Societies unveiled an Emerging Diseases Resource Center, which includes resources for providers and a registry for reporting confirmed and suspected resistant dermatophytes.
“Our goal is to provide easy-to-access and easy-to-understand resources to healthcare providers,” Esther Freeman, MD, PhD, told this news organization. She is director of Global Health Dermatology at Massachusetts General Hospital, associate professor of dermatology at Harvard Medical School, both in Boston, and chair of the AAD’s Emerging Diseases Task Force.
“Our resources include an algorithm for when to suspect a drug-resistant case and how to think through treatment options. We cover issues related to diagnosis and treatment, as well as linking to our case registry reporting system,” said Freeman.
The new registry resides within the AAD’s existing COVID-19, Mpox, and Emerging Infections Registry. “Our registry efforts have already captured 2500 COVID-19 and mpox cases from 72 different countries,” Freeman said. For all these infections, she added, “we hope that real-time data analysis of cases worldwide will provide information that helps physicians recognize and treat cases.”
Consistent with the registry’s approach, said Caplan and Gold, there is no silver bullet for battling dermatophyte resistance. What is needed, said Gold, is a coordinated approach involving public health officials, dermatologists, primary care providers, infectious disease specialists, pharmacists, and patients. “It’s going to be a team effort to address the challenge of emerging complex dermatophytosis,” he said.
Resistant T rubrum
“The biggest difference with T rubrum resistance is you may not see that widespread infection that we see with T indotineae,” said Caplan. T rubrum is probably the most common dermatophyte that dermatologists see, added Elewski, who encounters a resistant case at least monthly. One such patient, featured in a January 2021 British Journal of Dermatology research letter, cleared on itraconazole and ciclopirox cream but subsequently returned with itraconazole-resistant T rubrum because he had been doctor-shopping for the drug intermittently for years, she said. He cleared on posaconazole 300 mg daily, then was lost to follow-up.
TMVII
A 2023 Emerging Infectious Diseases report highlighted the potential for this dermatophyte to spread among men who have sex with men (MSM), presenting as an itchy, scaly rash affecting the pubic, genital, and buttocks skin. “People don’t generally think of a fungal infection as something that could behave like a sexually transmitted infection (STI),” said Gold.
Caplan and coauthors recently reported the first confirmed US TMVII case in JAMA Dermatology. Many experts suspect that unreported US cases existed previously, he said. “When it circulates in Europe and there’s so much travel, it’s probably here too.”
The fact that T indotineae was formerly called TMVIII has created confusion, added Caplan. “I’ve had patients say, ‘I’m worried I have that resistant ringworm that’s spreading among MSM.’ Whenever we talk about STIs and introduce the word ‘resistant,’ that comes with the potential for stigma, anxiety, and concern.” Fortunately, he said, TMVII has shown no resistance to first-line antifungals.
Why the Rise
Gold said, “We don’t know for sure why we’re seeing these different drug-resistant species popping up.” One possibility, he said, is the common misuse and overuse of topical antifungals — especially those available overseas in combination with high-potency steroids, such as clobetasol. Consumers use these products for a few weeks until symptoms resolve, then reapply them off and on over years, fueling resistance, said Gold.
“We are worried that with warming temperatures, there’s potential to see expansion of the geographic range of epidemic fungi,” he added. “That could be part of what has fueled recent increases in resistant dermatophytes. But it’s hard to prove.”
Climate change may be behind the emergence of Candida auris, according to a 2022 article in The Lancet Regional Health – Americas. This potentially fatal multidrug-resistant infection spreads easily among sick patients in healthcare facilities, according to a CDC information page on C auris.
Confirming Dermatophyte Infection
“A biopsy will only confirm the presence of fungus,” said Elewski. “Here you will need a lab that knows how to do a fungal culture.” Most state laboratories can do this, she said, as can some hospitals and special labs such as CMM in Cleveland.
It takes a Clinical Laboratory Improvement Amendments–certified lab to perform KOH prep in-house, added Caplan, plus up-to-date gear and knowledge of where and how to scrape and what to look for microscopically. Moreover, identifying T indotineae requires molecular testing available at only a handful of laboratories — listed on the AAD Emerging Dermatophytes webpage — nationwide.
Nevertheless, said Caplan, nailing down a diagnosis can guide treatment, often supplanting empirically prescribed antifungal steroid creams. “Those are probably not going to help. And people may be using those on areas of the body they shouldn’t. Both the clinical clues and the steps to make the diagnosis need to come together. But that’s often easier said than done, especially in a busy practice.”
Identifying resistance requires antifungal sensitivity testing, he added, which few labs perform. “Practically speaking,” said Elewski, “if the patient failed terbinafine, I would try itraconazole. You don’t necessarily need proof” of resistance. But if a patient does not respond to itraconazole and terbinafine clinically, she said that she might consider fungal susceptibility testing.
Treatment Tips
To address any resistant dermatophyte, Elewski recommended getting comfortable with itraconazole. For decades, she said, dermatologists have avoided itraconazole because terbinafine typically costs patients $10 for 3 months. “Itraconazole could be $200 per month,” said Elewski. Because of potential drug-drug interactions and absorption issues — and a boxed warning regarding congestive heart failure — physicians historically reserved itraconazole for severe fungal infections.
Itraconazole labeled dosing for onychomycosis is 200 mg daily for 12 weeks. Elewski favors a two-pronged attack, often combining an -azole antifungal with topical ciclopirox.
Another element that emerging tinea pathogens share is slower response to treatment. For T indotineae, reports appearing in the Journal of the American Academy of Dermatology in 2022 and 2024 suggest duration from 6-8 weeks up to 20 weeks.
To avoid recurrences of resistant T rubrum, Elewski treats for a year. However, she has problems getting itraconazole approved, when often it is the only agent that works. “I’ve written more letters than I like to insurance companies” to document terbinafine failure, she said.
Rarely, said Gold, dermatophyte infections resist both terbinafine and itraconazole. Next-line agents such as voriconazole, which some dermatologists have used for resistant T indotineae, can be much harder to tolerate, with more drug interactions, he said.
And because itraconazole, voriconazole, and posaconazole are all triazoles, added Elewski, the latter two might not work better than the former. But because these drugs might outperform itraconazole in selected cases, she said, “that’s when you want to do fungal susceptibility testing.”
TMVII is so new, said Caplan, that optimal therapy duration remains unclear. “One of the challenges with TMVII is when it gets into the genital skin, it’s a hair-bearing area. And based on various grooming practices, there’s an opportunity for the tinea to get deeper into the hair follicle and dermis. That may also be true of T indotineae.”
Anemic Arsenal
Unfortunately, said Gold, the arsenal of antifungals available in the United States remains limited. “Depending on how you count, there are only three to four classes of antifungal drugs designed to treat severe or invasive infections. So whenever we hear about a new fungal pathogen that’s causing resistant infections, it causes public health concern.”
Promising drugs in development include olorofim (F2G) and fosmanogepix (Basilea), according to Gold. However, he said, the development of these drugs to date has targeted invasive fungal infections such as aspergillosis. In June 2023, the Food and Drug Administration rejected the new drug application for olorofim, requesting additional data and analyses. Regarding fosmanogepix, a double-blinded noninferiority phase 3 trial in invasive yeast infections was recently launched, according to a September 24 press release.
Gold, Caplan, and Elewski reported no relevant financial disclosures. Freeman is a COVID-19 co-author for UpToDate and chair of the AAD Emerging Diseases Task Force.
A version of this article appeared on Medscape.com.
H pylori: ACG Guideline Advises New Approaches to Treatment
Helicobacter pylori is one of the most common human bacterial chronic infections globally. Its prevalence has actually decreased in North America in recent years, although its current range of approximately 30%-40% remains substantial given the potential clinical implications of infection.
Standards have changed considerably regarding the testing, treatment, and follow-up of H pylori. This is made clear by the just-published clinical practice guideline from the American College of Gastroenterology (ACG), which provides several new recommendations based on recent scientific evidence that should change your clinical approach to managing this common infection.
This discussion aims to synthesize and highlight key concepts from the ACG’s comprehensive publication.
Who Should Be Tested and Treated?
The cardinal diseases caused by H pylori have traditionally included peptic ulcer disease, marginal zone B-cell lymphoma, gastric adenocarcinoma, and dyspepsia.
Additional associations have been made with idiopathic thrombocytopenic purpura and otherwise unexplained iron deficiency.
New evidence suggests that patients taking long-term nonsteroidal anti-inflammatory drugs, including low-dose aspirin, are relatively more susceptible to infection.
The ACG’s guideline also recommends testing persons at an increased risk for gastric adenocarcinoma (eg, those with autoimmune gastritis, current or history of premalignant conditions, or first-degree relative with gastric cancer), as well as household members of patients with a positive nonserologic test for H pylori.
The authors note that those with an indication for testing should be offered treatment if determined to have an infection. These patients should also undergo a posttreatment test-of-cure, which should occur at least 4 weeks afterwards using a urea breath test, fecal antigen test, or gastric biopsy.
Caveats to Treatment
Patients with H pylori infections are advised to undergo treatment for a duration of 14 days. Some of the commercial prepackaged H pylori treatment options (eg, Pylera, which contains bismuth subcitrate/metronidazole/tetracycline) are dispensed in regimens lasting only 10 days and currently are viewed as inadequate.
In the United States, the patterns of antibiotic resistance for the previously used standard drugs in the treatment of H pylori have increased considerably. They range from 32% for clarithromycin, 38% for levofloxacin, and 42% for metronidazole, in contrast to 3% for amoxicillin, 1% for tetracycline, and 0% for rifabutin.
Clarithromycin- and levofloxacin-containing treatments should be avoided in treatment-naive patients unless specifically directed following the results of susceptibility tests with either a phenotypic method (culture-based) or a molecular method (polymerase chain reaction or next-generation sequencing). Notably, the mutations responsible for both clarithromycin and levofloxacin resistance may be detectable by stool-based testing.
Maintenance of intragastric acid suppression is key to H pylori eradication, as elevated intragastric pH promotes active replication of H pylori and makes it more susceptible to bactericidal antibiotics.
Therefore, the use of histamine-2 receptors is not recommended, as they are inadequate for achieving acid suppression. Instead, a dual-based therapy of either the potassium-competitive acid blocker (PCAB) vonoprazan (20 mg) or a high-dose proton pump inhibitor (PPI) and amoxicillin, administered twice daily, is effective, although this finding is based on limited evidence.
Treatment-Naive Patients
In treatment-naive patients without penicillin allergy and for whom antibiotic susceptibility testing has not been obtained, the guideline offers its strongest recommendation for bismuth quadruple therapy. This therapy typically consists of a PPI, bismuth subcitrate or subsalicylate, tetracycline, and metronidazole.
Among those with a penicillin allergy, bismuth quadruple therapy is also the primary treatment choice. The authors suggest that patients with a suspected allergy are referred to an allergist for possible penicillin desensitization, given that less than 1% of the population is thought to present with a “true” allergy.
The guideline also presented conditional recommendations, based on low- to moderate-quality evidence, for using a rifabutin-based triple regimen of omeprazole, amoxicillin, and rifabutin (Talicia); a PCAB-based dual regimen of vonoprazan and amoxicillin (Voquezna Dual Pak); and a PCAB-based triple regimen of vonoprazan, clarithromycin, and amoxicillin (Voquezna Triple Pak). In patients with unknown clarithromycin susceptibility, the PCAB-based triple therapy is preferred over PPI-clarithromycin triple therapy.
Although probiotics have been suggested to possibly lead to increased effectiveness or tolerability for H pylori eradication, this was based on studies with significant heterogeneity in their designs. At present, no high-quality data support probiotic therapy.
Clinicians may substitute doxycycline for tetracycline due to availability or cost, and also may prescribe metronidazole at a lower dose than recommended (1.5-2 g/d) to limit side effects. Both modifications have been associated with lower rates of H pylori eradication and are not recommended.
Treatment-Experienced Patients
Quadruple bismuth therapy is the optimal approach among treatment-experienced patients with persistent H pylori infection who have not previously received this therapy. However, this recommendation was rated as conditional, given that it was based on a low quality of evidence.
The guideline offered other recommendations for treatment-experienced patients with persistent infection who had received bismuth quadruple therapy — also conditionally based on a low quality of evidence.
In such patients, it is recommended to consider the use of a rifabutin-based triple therapy (ie, a PPI standard to double dose, amoxicillin, and rifabutin) and a levofloxacin-based triple therapy (ie, a PPI standard dose, levofloxacin, and amoxicillin or metronidazole).
Although significant evidence gaps prevented the authors from providing formal recommendations, they included a PCAB-based triple therapy of vonoprazan, clarithromycin, and amoxicillin (Voquezna Triple Pak) and a high-dose dual therapy of either vonoprazan (20 mg) or PPI (double dose) and amoxicillin among their suggested salvage regimens for these patients.
A New Standard
We must recognize, however, that there are still substantial evidence gaps, particularly around the use of a PCAB-based regimen and its relative advantages over a standard or high-dose PPI-based regimen. This may be of particular importance based on the variable prevalence of cytochrome P450 2C19 (CYP2C19) polymorphisms in the specific patient populations, as PCABs are not metabolized by CYP2C19.
Reviewing the entirety of the ACG’s clinical guideline is encouraged for additional details about the management of H pylori beyond what is highlighted herein.
Dr. Johnson, Professor of Medicine, Chief of Gastroenterology, Eastern Virginia Medical School, Norfolk, Virginia, disclosed ties with ISOTHRIVE and Johnson & Johnson.
A version of this article appeared on Medscape.com.
Helicobacter pylori is one of the most common human bacterial chronic infections globally. Its prevalence has actually decreased in North America in recent years, although its current range of approximately 30%-40% remains substantial given the potential clinical implications of infection.
Standards have changed considerably regarding the testing, treatment, and follow-up of H pylori. This is made clear by the just-published clinical practice guideline from the American College of Gastroenterology (ACG), which provides several new recommendations based on recent scientific evidence that should change your clinical approach to managing this common infection.
This discussion aims to synthesize and highlight key concepts from the ACG’s comprehensive publication.
Who Should Be Tested and Treated?
The cardinal diseases caused by H pylori have traditionally included peptic ulcer disease, marginal zone B-cell lymphoma, gastric adenocarcinoma, and dyspepsia.
Additional associations have been made with idiopathic thrombocytopenic purpura and otherwise unexplained iron deficiency.
New evidence suggests that patients taking long-term nonsteroidal anti-inflammatory drugs, including low-dose aspirin, are relatively more susceptible to infection.
The ACG’s guideline also recommends testing persons at an increased risk for gastric adenocarcinoma (eg, those with autoimmune gastritis, current or history of premalignant conditions, or first-degree relative with gastric cancer), as well as household members of patients with a positive nonserologic test for H pylori.
The authors note that those with an indication for testing should be offered treatment if determined to have an infection. These patients should also undergo a posttreatment test-of-cure, which should occur at least 4 weeks afterwards using a urea breath test, fecal antigen test, or gastric biopsy.
Caveats to Treatment
Patients with H pylori infections are advised to undergo treatment for a duration of 14 days. Some of the commercial prepackaged H pylori treatment options (eg, Pylera, which contains bismuth subcitrate/metronidazole/tetracycline) are dispensed in regimens lasting only 10 days and currently are viewed as inadequate.
In the United States, the patterns of antibiotic resistance for the previously used standard drugs in the treatment of H pylori have increased considerably. They range from 32% for clarithromycin, 38% for levofloxacin, and 42% for metronidazole, in contrast to 3% for amoxicillin, 1% for tetracycline, and 0% for rifabutin.
Clarithromycin- and levofloxacin-containing treatments should be avoided in treatment-naive patients unless specifically directed following the results of susceptibility tests with either a phenotypic method (culture-based) or a molecular method (polymerase chain reaction or next-generation sequencing). Notably, the mutations responsible for both clarithromycin and levofloxacin resistance may be detectable by stool-based testing.
Maintenance of intragastric acid suppression is key to H pylori eradication, as elevated intragastric pH promotes active replication of H pylori and makes it more susceptible to bactericidal antibiotics.
Therefore, the use of histamine-2 receptors is not recommended, as they are inadequate for achieving acid suppression. Instead, a dual-based therapy of either the potassium-competitive acid blocker (PCAB) vonoprazan (20 mg) or a high-dose proton pump inhibitor (PPI) and amoxicillin, administered twice daily, is effective, although this finding is based on limited evidence.
Treatment-Naive Patients
In treatment-naive patients without penicillin allergy and for whom antibiotic susceptibility testing has not been obtained, the guideline offers its strongest recommendation for bismuth quadruple therapy. This therapy typically consists of a PPI, bismuth subcitrate or subsalicylate, tetracycline, and metronidazole.
Among those with a penicillin allergy, bismuth quadruple therapy is also the primary treatment choice. The authors suggest that patients with a suspected allergy are referred to an allergist for possible penicillin desensitization, given that less than 1% of the population is thought to present with a “true” allergy.
The guideline also presented conditional recommendations, based on low- to moderate-quality evidence, for using a rifabutin-based triple regimen of omeprazole, amoxicillin, and rifabutin (Talicia); a PCAB-based dual regimen of vonoprazan and amoxicillin (Voquezna Dual Pak); and a PCAB-based triple regimen of vonoprazan, clarithromycin, and amoxicillin (Voquezna Triple Pak). In patients with unknown clarithromycin susceptibility, the PCAB-based triple therapy is preferred over PPI-clarithromycin triple therapy.
Although probiotics have been suggested to possibly lead to increased effectiveness or tolerability for H pylori eradication, this was based on studies with significant heterogeneity in their designs. At present, no high-quality data support probiotic therapy.
Clinicians may substitute doxycycline for tetracycline due to availability or cost, and also may prescribe metronidazole at a lower dose than recommended (1.5-2 g/d) to limit side effects. Both modifications have been associated with lower rates of H pylori eradication and are not recommended.
Treatment-Experienced Patients
Quadruple bismuth therapy is the optimal approach among treatment-experienced patients with persistent H pylori infection who have not previously received this therapy. However, this recommendation was rated as conditional, given that it was based on a low quality of evidence.
The guideline offered other recommendations for treatment-experienced patients with persistent infection who had received bismuth quadruple therapy — also conditionally based on a low quality of evidence.
In such patients, it is recommended to consider the use of a rifabutin-based triple therapy (ie, a PPI standard to double dose, amoxicillin, and rifabutin) and a levofloxacin-based triple therapy (ie, a PPI standard dose, levofloxacin, and amoxicillin or metronidazole).
Although significant evidence gaps prevented the authors from providing formal recommendations, they included a PCAB-based triple therapy of vonoprazan, clarithromycin, and amoxicillin (Voquezna Triple Pak) and a high-dose dual therapy of either vonoprazan (20 mg) or PPI (double dose) and amoxicillin among their suggested salvage regimens for these patients.
A New Standard
We must recognize, however, that there are still substantial evidence gaps, particularly around the use of a PCAB-based regimen and its relative advantages over a standard or high-dose PPI-based regimen. This may be of particular importance based on the variable prevalence of cytochrome P450 2C19 (CYP2C19) polymorphisms in the specific patient populations, as PCABs are not metabolized by CYP2C19.
Reviewing the entirety of the ACG’s clinical guideline is encouraged for additional details about the management of H pylori beyond what is highlighted herein.
Dr. Johnson, Professor of Medicine, Chief of Gastroenterology, Eastern Virginia Medical School, Norfolk, Virginia, disclosed ties with ISOTHRIVE and Johnson & Johnson.
A version of this article appeared on Medscape.com.
Helicobacter pylori is one of the most common human bacterial chronic infections globally. Its prevalence has actually decreased in North America in recent years, although its current range of approximately 30%-40% remains substantial given the potential clinical implications of infection.
Standards have changed considerably regarding the testing, treatment, and follow-up of H pylori. This is made clear by the just-published clinical practice guideline from the American College of Gastroenterology (ACG), which provides several new recommendations based on recent scientific evidence that should change your clinical approach to managing this common infection.
This discussion aims to synthesize and highlight key concepts from the ACG’s comprehensive publication.
Who Should Be Tested and Treated?
The cardinal diseases caused by H pylori have traditionally included peptic ulcer disease, marginal zone B-cell lymphoma, gastric adenocarcinoma, and dyspepsia.
Additional associations have been made with idiopathic thrombocytopenic purpura and otherwise unexplained iron deficiency.
New evidence suggests that patients taking long-term nonsteroidal anti-inflammatory drugs, including low-dose aspirin, are relatively more susceptible to infection.
The ACG’s guideline also recommends testing persons at an increased risk for gastric adenocarcinoma (eg, those with autoimmune gastritis, current or history of premalignant conditions, or first-degree relative with gastric cancer), as well as household members of patients with a positive nonserologic test for H pylori.
The authors note that those with an indication for testing should be offered treatment if determined to have an infection. These patients should also undergo a posttreatment test-of-cure, which should occur at least 4 weeks afterwards using a urea breath test, fecal antigen test, or gastric biopsy.
Caveats to Treatment
Patients with H pylori infections are advised to undergo treatment for a duration of 14 days. Some of the commercial prepackaged H pylori treatment options (eg, Pylera, which contains bismuth subcitrate/metronidazole/tetracycline) are dispensed in regimens lasting only 10 days and currently are viewed as inadequate.
In the United States, the patterns of antibiotic resistance for the previously used standard drugs in the treatment of H pylori have increased considerably. They range from 32% for clarithromycin, 38% for levofloxacin, and 42% for metronidazole, in contrast to 3% for amoxicillin, 1% for tetracycline, and 0% for rifabutin.
Clarithromycin- and levofloxacin-containing treatments should be avoided in treatment-naive patients unless specifically directed following the results of susceptibility tests with either a phenotypic method (culture-based) or a molecular method (polymerase chain reaction or next-generation sequencing). Notably, the mutations responsible for both clarithromycin and levofloxacin resistance may be detectable by stool-based testing.
Maintenance of intragastric acid suppression is key to H pylori eradication, as elevated intragastric pH promotes active replication of H pylori and makes it more susceptible to bactericidal antibiotics.
Therefore, the use of histamine-2 receptors is not recommended, as they are inadequate for achieving acid suppression. Instead, a dual-based therapy of either the potassium-competitive acid blocker (PCAB) vonoprazan (20 mg) or a high-dose proton pump inhibitor (PPI) and amoxicillin, administered twice daily, is effective, although this finding is based on limited evidence.
Treatment-Naive Patients
In treatment-naive patients without penicillin allergy and for whom antibiotic susceptibility testing has not been obtained, the guideline offers its strongest recommendation for bismuth quadruple therapy. This therapy typically consists of a PPI, bismuth subcitrate or subsalicylate, tetracycline, and metronidazole.
Among those with a penicillin allergy, bismuth quadruple therapy is also the primary treatment choice. The authors suggest that patients with a suspected allergy are referred to an allergist for possible penicillin desensitization, given that less than 1% of the population is thought to present with a “true” allergy.
The guideline also presented conditional recommendations, based on low- to moderate-quality evidence, for using a rifabutin-based triple regimen of omeprazole, amoxicillin, and rifabutin (Talicia); a PCAB-based dual regimen of vonoprazan and amoxicillin (Voquezna Dual Pak); and a PCAB-based triple regimen of vonoprazan, clarithromycin, and amoxicillin (Voquezna Triple Pak). In patients with unknown clarithromycin susceptibility, the PCAB-based triple therapy is preferred over PPI-clarithromycin triple therapy.
Although probiotics have been suggested to possibly lead to increased effectiveness or tolerability for H pylori eradication, this was based on studies with significant heterogeneity in their designs. At present, no high-quality data support probiotic therapy.
Clinicians may substitute doxycycline for tetracycline due to availability or cost, and also may prescribe metronidazole at a lower dose than recommended (1.5-2 g/d) to limit side effects. Both modifications have been associated with lower rates of H pylori eradication and are not recommended.
Treatment-Experienced Patients
Quadruple bismuth therapy is the optimal approach among treatment-experienced patients with persistent H pylori infection who have not previously received this therapy. However, this recommendation was rated as conditional, given that it was based on a low quality of evidence.
The guideline offered other recommendations for treatment-experienced patients with persistent infection who had received bismuth quadruple therapy — also conditionally based on a low quality of evidence.
In such patients, it is recommended to consider the use of a rifabutin-based triple therapy (ie, a PPI standard to double dose, amoxicillin, and rifabutin) and a levofloxacin-based triple therapy (ie, a PPI standard dose, levofloxacin, and amoxicillin or metronidazole).
Although significant evidence gaps prevented the authors from providing formal recommendations, they included a PCAB-based triple therapy of vonoprazan, clarithromycin, and amoxicillin (Voquezna Triple Pak) and a high-dose dual therapy of either vonoprazan (20 mg) or PPI (double dose) and amoxicillin among their suggested salvage regimens for these patients.
A New Standard
We must recognize, however, that there are still substantial evidence gaps, particularly around the use of a PCAB-based regimen and its relative advantages over a standard or high-dose PPI-based regimen. This may be of particular importance based on the variable prevalence of cytochrome P450 2C19 (CYP2C19) polymorphisms in the specific patient populations, as PCABs are not metabolized by CYP2C19.
Reviewing the entirety of the ACG’s clinical guideline is encouraged for additional details about the management of H pylori beyond what is highlighted herein.
Dr. Johnson, Professor of Medicine, Chief of Gastroenterology, Eastern Virginia Medical School, Norfolk, Virginia, disclosed ties with ISOTHRIVE and Johnson & Johnson.
A version of this article appeared on Medscape.com.
ACIP Recommends Pneumococcal Vaccine for Adults 50 Years or Older
The US Centers for Disease Control and Prevention’s (CDC’s) Advisory Committee on Immunization Practices (ACIP) now recommends a pneumococcal conjugate vaccine (PCV) for all PCV-naive adults aged 50 years or older. The new recommendation, which passed with an ACIP member vote of 14 for and one against, expands the current age-based recommendations, which include children younger than 5 years and adults older than 65 years, as well as adults aged 19-64 years with underlying conditions or risk factors who have not received a PCV and certain adults who have received PCV13 but not PCV20.
The decision was based in part on economic analyses of the use of PCV in adults aged 50-64 years in the United States. Miwako Kobayashi, MD, presented the summary of the Pneumococcal Vaccines Work Group’s interpretation of the evidence and the proposed recommendation in a meeting of the ACIP on October 23, 2024, when the ACIP voting occurred.
Data from the CDC show an increase in the relative burden of pneumococcal disease in adults aged 50-64 years based in part on the success of the pediatric PCV program, she said.
Health equity was another main factor in the Work Group’s decision to recommend vaccination for adults aged 50 years or older. “Disparities in pneumococcal vaccine coverage by race and ethnicity exist for both age-based and risk-based indications,” Kobayashi noted in her presentation. The Work Group acknowledged that the overall effect of a vaccine recommendation on health equity is complex, but the majority agreed that the update would improve health equity by increasing vaccine coverage for those with known or unknown risk factors and providing protection at an earlier age when some populations already experience elevated disease rates, she said.
As for safety, the Work Group concluded that the undesirable anticipated effects of PCVs are minimal, despite the potential signal for Guillain-Barré Syndrome, and the CDC and US Food and Drug Administration will continue to monitor post-licensure safety of PCVs.
Support Not Universal
A majority of the ACIP Pneumococcal Vaccines Work Group supported the approved option, but agreed that a future booster dose may be needed, Work Group Chair James Loehr, MD, said in his introductory presentation.
Overall, key uncertainties remain, including indirect effects of new pediatric pneumococcal vaccines on adults, data on the duration of protection with adult vaccinations, and the impact new higher-valency vaccines have on adults, several of which are in development, Loehr said.
A new 21-valent PCV, known as PCV 21, was approved by the FDA for adults aged 18 years or older in June 2024, said Loehr. “PCV21 is not PCV20 with one additional serotype” and provides additional protection, he emphasized. The Work Group examined models involving PCV21 and the existing PCV20. However, a majority of the Work Group agreed that having age-based recommendations based on vaccine product would be more challenging to implement and that insurance coverage may be a factor given the recent approval of PCV21. Therefore, the proposal submitted to the full ACIP was not for a specific PCV.
Notably, Loehr said that, although as Work Group Chair he was tasked with making the motion in favor of the recommendation, he voted against it as a voting member because of his strong opinion that only the PCV21 vaccine is needed for vaccine-naive adults aged 50 or older. “I think that PCV21 is a better vaccine that targets more serotypes,” he said during the discussion. Data presented at the February 2024 ACIP meeting showed more than 80% coverage vs less than 60% coverage for invasive pneumococcal disease with PCV21 vs PCV20 among adults aged 65 years or older and those aged 19-64 years with a risk-based indication, Loehr said.
A version of this article appeared on Medscape.com.
The US Centers for Disease Control and Prevention’s (CDC’s) Advisory Committee on Immunization Practices (ACIP) now recommends a pneumococcal conjugate vaccine (PCV) for all PCV-naive adults aged 50 years or older. The new recommendation, which passed with an ACIP member vote of 14 for and one against, expands the current age-based recommendations, which include children younger than 5 years and adults older than 65 years, as well as adults aged 19-64 years with underlying conditions or risk factors who have not received a PCV and certain adults who have received PCV13 but not PCV20.
The decision was based in part on economic analyses of the use of PCV in adults aged 50-64 years in the United States. Miwako Kobayashi, MD, presented the summary of the Pneumococcal Vaccines Work Group’s interpretation of the evidence and the proposed recommendation in a meeting of the ACIP on October 23, 2024, when the ACIP voting occurred.
Data from the CDC show an increase in the relative burden of pneumococcal disease in adults aged 50-64 years based in part on the success of the pediatric PCV program, she said.
Health equity was another main factor in the Work Group’s decision to recommend vaccination for adults aged 50 years or older. “Disparities in pneumococcal vaccine coverage by race and ethnicity exist for both age-based and risk-based indications,” Kobayashi noted in her presentation. The Work Group acknowledged that the overall effect of a vaccine recommendation on health equity is complex, but the majority agreed that the update would improve health equity by increasing vaccine coverage for those with known or unknown risk factors and providing protection at an earlier age when some populations already experience elevated disease rates, she said.
As for safety, the Work Group concluded that the undesirable anticipated effects of PCVs are minimal, despite the potential signal for Guillain-Barré Syndrome, and the CDC and US Food and Drug Administration will continue to monitor post-licensure safety of PCVs.
Support Not Universal
A majority of the ACIP Pneumococcal Vaccines Work Group supported the approved option, but agreed that a future booster dose may be needed, Work Group Chair James Loehr, MD, said in his introductory presentation.
Overall, key uncertainties remain, including indirect effects of new pediatric pneumococcal vaccines on adults, data on the duration of protection with adult vaccinations, and the impact new higher-valency vaccines have on adults, several of which are in development, Loehr said.
A new 21-valent PCV, known as PCV 21, was approved by the FDA for adults aged 18 years or older in June 2024, said Loehr. “PCV21 is not PCV20 with one additional serotype” and provides additional protection, he emphasized. The Work Group examined models involving PCV21 and the existing PCV20. However, a majority of the Work Group agreed that having age-based recommendations based on vaccine product would be more challenging to implement and that insurance coverage may be a factor given the recent approval of PCV21. Therefore, the proposal submitted to the full ACIP was not for a specific PCV.
Notably, Loehr said that, although as Work Group Chair he was tasked with making the motion in favor of the recommendation, he voted against it as a voting member because of his strong opinion that only the PCV21 vaccine is needed for vaccine-naive adults aged 50 or older. “I think that PCV21 is a better vaccine that targets more serotypes,” he said during the discussion. Data presented at the February 2024 ACIP meeting showed more than 80% coverage vs less than 60% coverage for invasive pneumococcal disease with PCV21 vs PCV20 among adults aged 65 years or older and those aged 19-64 years with a risk-based indication, Loehr said.
A version of this article appeared on Medscape.com.
The US Centers for Disease Control and Prevention’s (CDC’s) Advisory Committee on Immunization Practices (ACIP) now recommends a pneumococcal conjugate vaccine (PCV) for all PCV-naive adults aged 50 years or older. The new recommendation, which passed with an ACIP member vote of 14 for and one against, expands the current age-based recommendations, which include children younger than 5 years and adults older than 65 years, as well as adults aged 19-64 years with underlying conditions or risk factors who have not received a PCV and certain adults who have received PCV13 but not PCV20.
The decision was based in part on economic analyses of the use of PCV in adults aged 50-64 years in the United States. Miwako Kobayashi, MD, presented the summary of the Pneumococcal Vaccines Work Group’s interpretation of the evidence and the proposed recommendation in a meeting of the ACIP on October 23, 2024, when the ACIP voting occurred.
Data from the CDC show an increase in the relative burden of pneumococcal disease in adults aged 50-64 years based in part on the success of the pediatric PCV program, she said.
Health equity was another main factor in the Work Group’s decision to recommend vaccination for adults aged 50 years or older. “Disparities in pneumococcal vaccine coverage by race and ethnicity exist for both age-based and risk-based indications,” Kobayashi noted in her presentation. The Work Group acknowledged that the overall effect of a vaccine recommendation on health equity is complex, but the majority agreed that the update would improve health equity by increasing vaccine coverage for those with known or unknown risk factors and providing protection at an earlier age when some populations already experience elevated disease rates, she said.
As for safety, the Work Group concluded that the undesirable anticipated effects of PCVs are minimal, despite the potential signal for Guillain-Barré Syndrome, and the CDC and US Food and Drug Administration will continue to monitor post-licensure safety of PCVs.
Support Not Universal
A majority of the ACIP Pneumococcal Vaccines Work Group supported the approved option, but agreed that a future booster dose may be needed, Work Group Chair James Loehr, MD, said in his introductory presentation.
Overall, key uncertainties remain, including indirect effects of new pediatric pneumococcal vaccines on adults, data on the duration of protection with adult vaccinations, and the impact new higher-valency vaccines have on adults, several of which are in development, Loehr said.
A new 21-valent PCV, known as PCV 21, was approved by the FDA for adults aged 18 years or older in June 2024, said Loehr. “PCV21 is not PCV20 with one additional serotype” and provides additional protection, he emphasized. The Work Group examined models involving PCV21 and the existing PCV20. However, a majority of the Work Group agreed that having age-based recommendations based on vaccine product would be more challenging to implement and that insurance coverage may be a factor given the recent approval of PCV21. Therefore, the proposal submitted to the full ACIP was not for a specific PCV.
Notably, Loehr said that, although as Work Group Chair he was tasked with making the motion in favor of the recommendation, he voted against it as a voting member because of his strong opinion that only the PCV21 vaccine is needed for vaccine-naive adults aged 50 or older. “I think that PCV21 is a better vaccine that targets more serotypes,” he said during the discussion. Data presented at the February 2024 ACIP meeting showed more than 80% coverage vs less than 60% coverage for invasive pneumococcal disease with PCV21 vs PCV20 among adults aged 65 years or older and those aged 19-64 years with a risk-based indication, Loehr said.
A version of this article appeared on Medscape.com.
Groups With Highest Unmet Need for PrEP Highlighted in Analysis
LOS ANGELES — Use of preexposure prophylaxis (PrEP) to prevent HIV is increasing overall, but both the rate of increase for starting PrEP and the rate of unmet need differ widely by demographic group, according to new data from a large study.
An analysis by Li Tao, MD, MS, PhD, director of real-world evidence at Gilead Sciences, and colleagues looked at statistical trends from 2019 to 2023 and found that Black, Hispanic, and Medicaid-insured populations continue to lack equitable access to PrEP.
Among the findings were that most new PrEP users were men with HIV risk factors who are commercially insured and live in predominantly non-Hispanic White areas (53% in 2019 and 43% in 2023). For comparison, men living in predominantly Black or Hispanic neighborhoods, or who are insured by Medicaid, saw lower proportions of PrEP use (16% in 2019 and 17% in 2023) despite higher annual increases in PrEP use (11% per year) and higher unmet needs.
Half a Million Real-World Participants
Tao presented her team’s findings at the Infectious Disease Week (IDWeek) 2024 Annual Meeting. The study included “more than half a million real-world PrEP users over the past 5 years,” she said.
The group with the lowest growth in initiation of PrEP in the study period (an annual percentage increase of 2%) and the lowest unmet need included men with HIV risk factors, who were using commercial insurance and living in White-dominant neighborhoods.
HIV risk factors included diagnosis of any sexually transmitted disease, contact with and exposure to communicable diseases, high-risk sexual behavior, contact with a hypodermic needle, long-term prophylaxis, HIV prevention counseling, and HIV screening.
Other men with HIV risk factors (those who were commercially insured, living in Black/Hispanic neighborhoods, or those on Medicaid across all neighborhoods) had a moderate increase in PrEP initiation (an annual percentage increase of 11%-16%) and higher unmet needs.
Researchers gathered data on PrEP prescriptions and new HIV diagnoses (from 2019 to 2023) through the IQVIA pharmacy claims database. PrEP-to-need ratio (PNR) is the number of individuals using PrEP in a year divided by new HIV diagnoses in the previous year. It was calculated for subgroups defined by five PNR-associated factors: Sex, insurance, recorded HIV risk factors (identified by diagnosis or procedure codes), “Ending the HIV Epidemic” jurisdictions, and neighborhood race/ethnicity mix.
Disparities Persist
While PrEP use improved across all the groups studied in the 5 years, “disparities still persist and the need remains very significant,” Tao said. “It’s very crucial for guiding the future HIV prevention options.”
“Long-acting PrEP options may help to address some social determinants structural factors in HIV acquisition,” she added.
What Programs Are Helping?
Some guidelines and programs are helping increase uptake, Tao said.
The United States Preventive Services Task Force (USPSTF) guidelines “reinforce more accessible PrEP programs to individuals like zero-cost sharing or same-day dispensing,” Tao said in a press briefing. “Those kinds of policies are really effective. We can see that after the implementation of the USPSTF guidelines, the copay sharing is really decreasing and is coinciding with the HIV rates declining.”
The Medicaid coverage expansion in 40 states “has been really effective” in PrEP uptake, she added.
Colleen Kelley, MD, MPH, with the Division of Infectious Diseases at the Rollins School of Public Health, Emory University, in Atlanta, who was not part of the research, said there has been a slow but improving uptake of PrEP across the board in the United States, “but the issue is that the uptake has been inequitable.”
Large Study With Recent Data
“This is an extremely large study with very recent data,” Kelley said. “Additionally, they were able to couple (the uptake) with unmet need. People who are at higher risk of acquiring HIV or who live in high-risk areas for HIV should have greater access to PrEP. They have a greater need for PrEP. What we really need to do from an equity perspective is match the PrEP use with the PrEP need and we have not been successful in doing that.”
Kelley added that the finding that the group that had the highest unmet need for PrEP in the study also had no recorded HIV risk factors. “It’s an interesting time to start thinking about beyond risk factor coverage for PrEP,” she said.
Another issue, Kelley said, is that “people are using (PrEP) but they’re also stopping it. People will need to take PrEP many years for protection, but about half discontinue in the first 6-12 months.
“We need to look at how people will persist on PrEP over the long term. That’s the next frontier,” she said. “We hope the long-acting injectables will help overcome some of the PrEP fatigue. But some may just tire of taking medication repeatedly for an infection they don’t have,” she said.
The study was funded by Gilead Sciences. Tao is employed by and is a shareholder of Gilead Sciences. All relevant financial disclosures have been mitigated, according to the paper. Kelley has research grants to her institution from Gilead, Moderna, Novavax, ViiV, and Humanigen.
A version of this article first appeared on Medscape.com.
LOS ANGELES — Use of preexposure prophylaxis (PrEP) to prevent HIV is increasing overall, but both the rate of increase for starting PrEP and the rate of unmet need differ widely by demographic group, according to new data from a large study.
An analysis by Li Tao, MD, MS, PhD, director of real-world evidence at Gilead Sciences, and colleagues looked at statistical trends from 2019 to 2023 and found that Black, Hispanic, and Medicaid-insured populations continue to lack equitable access to PrEP.
Among the findings were that most new PrEP users were men with HIV risk factors who are commercially insured and live in predominantly non-Hispanic White areas (53% in 2019 and 43% in 2023). For comparison, men living in predominantly Black or Hispanic neighborhoods, or who are insured by Medicaid, saw lower proportions of PrEP use (16% in 2019 and 17% in 2023) despite higher annual increases in PrEP use (11% per year) and higher unmet needs.
Half a Million Real-World Participants
Tao presented her team’s findings at the Infectious Disease Week (IDWeek) 2024 Annual Meeting. The study included “more than half a million real-world PrEP users over the past 5 years,” she said.
The group with the lowest growth in initiation of PrEP in the study period (an annual percentage increase of 2%) and the lowest unmet need included men with HIV risk factors, who were using commercial insurance and living in White-dominant neighborhoods.
HIV risk factors included diagnosis of any sexually transmitted disease, contact with and exposure to communicable diseases, high-risk sexual behavior, contact with a hypodermic needle, long-term prophylaxis, HIV prevention counseling, and HIV screening.
Other men with HIV risk factors (those who were commercially insured, living in Black/Hispanic neighborhoods, or those on Medicaid across all neighborhoods) had a moderate increase in PrEP initiation (an annual percentage increase of 11%-16%) and higher unmet needs.
Researchers gathered data on PrEP prescriptions and new HIV diagnoses (from 2019 to 2023) through the IQVIA pharmacy claims database. PrEP-to-need ratio (PNR) is the number of individuals using PrEP in a year divided by new HIV diagnoses in the previous year. It was calculated for subgroups defined by five PNR-associated factors: Sex, insurance, recorded HIV risk factors (identified by diagnosis or procedure codes), “Ending the HIV Epidemic” jurisdictions, and neighborhood race/ethnicity mix.
Disparities Persist
While PrEP use improved across all the groups studied in the 5 years, “disparities still persist and the need remains very significant,” Tao said. “It’s very crucial for guiding the future HIV prevention options.”
“Long-acting PrEP options may help to address some social determinants structural factors in HIV acquisition,” she added.
What Programs Are Helping?
Some guidelines and programs are helping increase uptake, Tao said.
The United States Preventive Services Task Force (USPSTF) guidelines “reinforce more accessible PrEP programs to individuals like zero-cost sharing or same-day dispensing,” Tao said in a press briefing. “Those kinds of policies are really effective. We can see that after the implementation of the USPSTF guidelines, the copay sharing is really decreasing and is coinciding with the HIV rates declining.”
The Medicaid coverage expansion in 40 states “has been really effective” in PrEP uptake, she added.
Colleen Kelley, MD, MPH, with the Division of Infectious Diseases at the Rollins School of Public Health, Emory University, in Atlanta, who was not part of the research, said there has been a slow but improving uptake of PrEP across the board in the United States, “but the issue is that the uptake has been inequitable.”
Large Study With Recent Data
“This is an extremely large study with very recent data,” Kelley said. “Additionally, they were able to couple (the uptake) with unmet need. People who are at higher risk of acquiring HIV or who live in high-risk areas for HIV should have greater access to PrEP. They have a greater need for PrEP. What we really need to do from an equity perspective is match the PrEP use with the PrEP need and we have not been successful in doing that.”
Kelley added that the finding that the group that had the highest unmet need for PrEP in the study also had no recorded HIV risk factors. “It’s an interesting time to start thinking about beyond risk factor coverage for PrEP,” she said.
Another issue, Kelley said, is that “people are using (PrEP) but they’re also stopping it. People will need to take PrEP many years for protection, but about half discontinue in the first 6-12 months.
“We need to look at how people will persist on PrEP over the long term. That’s the next frontier,” she said. “We hope the long-acting injectables will help overcome some of the PrEP fatigue. But some may just tire of taking medication repeatedly for an infection they don’t have,” she said.
The study was funded by Gilead Sciences. Tao is employed by and is a shareholder of Gilead Sciences. All relevant financial disclosures have been mitigated, according to the paper. Kelley has research grants to her institution from Gilead, Moderna, Novavax, ViiV, and Humanigen.
A version of this article first appeared on Medscape.com.
LOS ANGELES — Use of preexposure prophylaxis (PrEP) to prevent HIV is increasing overall, but both the rate of increase for starting PrEP and the rate of unmet need differ widely by demographic group, according to new data from a large study.
An analysis by Li Tao, MD, MS, PhD, director of real-world evidence at Gilead Sciences, and colleagues looked at statistical trends from 2019 to 2023 and found that Black, Hispanic, and Medicaid-insured populations continue to lack equitable access to PrEP.
Among the findings were that most new PrEP users were men with HIV risk factors who are commercially insured and live in predominantly non-Hispanic White areas (53% in 2019 and 43% in 2023). For comparison, men living in predominantly Black or Hispanic neighborhoods, or who are insured by Medicaid, saw lower proportions of PrEP use (16% in 2019 and 17% in 2023) despite higher annual increases in PrEP use (11% per year) and higher unmet needs.
Half a Million Real-World Participants
Tao presented her team’s findings at the Infectious Disease Week (IDWeek) 2024 Annual Meeting. The study included “more than half a million real-world PrEP users over the past 5 years,” she said.
The group with the lowest growth in initiation of PrEP in the study period (an annual percentage increase of 2%) and the lowest unmet need included men with HIV risk factors, who were using commercial insurance and living in White-dominant neighborhoods.
HIV risk factors included diagnosis of any sexually transmitted disease, contact with and exposure to communicable diseases, high-risk sexual behavior, contact with a hypodermic needle, long-term prophylaxis, HIV prevention counseling, and HIV screening.
Other men with HIV risk factors (those who were commercially insured, living in Black/Hispanic neighborhoods, or those on Medicaid across all neighborhoods) had a moderate increase in PrEP initiation (an annual percentage increase of 11%-16%) and higher unmet needs.
Researchers gathered data on PrEP prescriptions and new HIV diagnoses (from 2019 to 2023) through the IQVIA pharmacy claims database. PrEP-to-need ratio (PNR) is the number of individuals using PrEP in a year divided by new HIV diagnoses in the previous year. It was calculated for subgroups defined by five PNR-associated factors: Sex, insurance, recorded HIV risk factors (identified by diagnosis or procedure codes), “Ending the HIV Epidemic” jurisdictions, and neighborhood race/ethnicity mix.
Disparities Persist
While PrEP use improved across all the groups studied in the 5 years, “disparities still persist and the need remains very significant,” Tao said. “It’s very crucial for guiding the future HIV prevention options.”
“Long-acting PrEP options may help to address some social determinants structural factors in HIV acquisition,” she added.
What Programs Are Helping?
Some guidelines and programs are helping increase uptake, Tao said.
The United States Preventive Services Task Force (USPSTF) guidelines “reinforce more accessible PrEP programs to individuals like zero-cost sharing or same-day dispensing,” Tao said in a press briefing. “Those kinds of policies are really effective. We can see that after the implementation of the USPSTF guidelines, the copay sharing is really decreasing and is coinciding with the HIV rates declining.”
The Medicaid coverage expansion in 40 states “has been really effective” in PrEP uptake, she added.
Colleen Kelley, MD, MPH, with the Division of Infectious Diseases at the Rollins School of Public Health, Emory University, in Atlanta, who was not part of the research, said there has been a slow but improving uptake of PrEP across the board in the United States, “but the issue is that the uptake has been inequitable.”
Large Study With Recent Data
“This is an extremely large study with very recent data,” Kelley said. “Additionally, they were able to couple (the uptake) with unmet need. People who are at higher risk of acquiring HIV or who live in high-risk areas for HIV should have greater access to PrEP. They have a greater need for PrEP. What we really need to do from an equity perspective is match the PrEP use with the PrEP need and we have not been successful in doing that.”
Kelley added that the finding that the group that had the highest unmet need for PrEP in the study also had no recorded HIV risk factors. “It’s an interesting time to start thinking about beyond risk factor coverage for PrEP,” she said.
Another issue, Kelley said, is that “people are using (PrEP) but they’re also stopping it. People will need to take PrEP many years for protection, but about half discontinue in the first 6-12 months.
“We need to look at how people will persist on PrEP over the long term. That’s the next frontier,” she said. “We hope the long-acting injectables will help overcome some of the PrEP fatigue. But some may just tire of taking medication repeatedly for an infection they don’t have,” she said.
The study was funded by Gilead Sciences. Tao is employed by and is a shareholder of Gilead Sciences. All relevant financial disclosures have been mitigated, according to the paper. Kelley has research grants to her institution from Gilead, Moderna, Novavax, ViiV, and Humanigen.
A version of this article first appeared on Medscape.com.
FROM IDWEEK 2024
How Effective Is the High-Dose Flu Vaccine in Older Adults?
How can the immunogenicity and effectiveness of flu vaccines be improved in older adults? Several strategies are available, one being the addition of an adjuvant. For example, the MF59-adjuvanted vaccine has shown superior immunogenicity. However, “we do not have data from controlled and randomized clinical trials showing superior clinical effectiveness versus the standard dose,” Professor Odile Launay, an infectious disease specialist at Cochin Hospital in Paris, France, noted during a press conference. Another option is to increase the antigen dose in the vaccine, creating a high-dose (HD) flu vaccine.
Why is there a need for an HD vaccine? “The elderly population bears the greatest burden from the flu,” explained Launay. “This is due to three factors: An aging immune system, a higher number of comorbidities, and increased frailty.” Standard-dose flu vaccines are seen as offering suboptimal protection for those older than 65 years, which led to the development of a quadrivalent vaccine with four times the antigen dose of standard flu vaccines. This HD vaccine was introduced in France during the 2021/2022 flu season. A real-world cohort study has since been conducted to evaluate its effectiveness in the target population — those aged 65 years or older. The results were recently published in Clinical Microbiology and Infection.
Cohort Study
The study included 405,385 noninstitutionalized people aged 65 years or older matched with 1,621,540 individuals in a 1:4 ratio. The first group received the HD vaccine, while the second group received the standard-dose vaccine. Both the groups had an average age of 77 years, with 56% women, and 51% vaccinated in pharmacies. The majority had been previously vaccinated against flu (91%), and 97% had completed a full COVID-19 vaccination schedule. More than half had at least one chronic illness.
Hospitalization rates for flu — the study’s primary outcome — were 69.5 vs 90.5 per 100,000 person-years in the HD vs standard-dose group. This represented a 23.3% reduction (95% CI, 8.4-35.8; P = .003).
Strengths and Limitations
Among the strengths of the study, Launay highlighted the large number of vaccinated participants older than 65 years — more than 7 million — and the widespread use of polymerase chain reaction flu tests in cases of hospitalization for respiratory infections, which improved flu coding in the database used. Additionally, the results were consistent with those of previous studies.
However, limitations included the retrospective design, which did not randomize participants and introduced potential bias. For example, the HD vaccine may have been prioritized for the oldest people or those with multiple comorbidities. Additionally, the 2021/2022 flu season was atypical, with the simultaneous circulation of the flu virus and SARS-CoV-2, as noted by Launay.
Conclusion
In conclusion, this first evaluation of the HD flu vaccine’s effectiveness in France showed a 25% reduction in hospitalizations, consistent with existing data covering 12 flu seasons. The vaccine has been available for a longer period in the United States and Northern Europe.
“The latest unpublished data from the 2022/23 season show a 27% reduction in hospitalizations with the HD vaccine in people over 65,” added Launay.
Note: Due to a pricing disagreement with the French government, Sanofi’s HD flu vaccine Efluelda, intended for people older than 65 years, will not be available this year. (See: Withdrawal of the Efluelda Influenza Vaccine: The Academy of Medicine Reacts). However, the company has submitted a dossier for a trivalent form for a return in the 2025/2026 season and is working on developing mRNA vaccines. Additionally, a combined flu/COVID-19 vaccine is currently in development.
The study was funded by Sanofi. Several authors are Sanofi employees. Odile Launay reported conflicts of interest with Sanofi, MSD, Pfizer, GSK, and Moderna.
This story was translated from Medscape’s French edition using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article appeared on Medscape.com.
How can the immunogenicity and effectiveness of flu vaccines be improved in older adults? Several strategies are available, one being the addition of an adjuvant. For example, the MF59-adjuvanted vaccine has shown superior immunogenicity. However, “we do not have data from controlled and randomized clinical trials showing superior clinical effectiveness versus the standard dose,” Professor Odile Launay, an infectious disease specialist at Cochin Hospital in Paris, France, noted during a press conference. Another option is to increase the antigen dose in the vaccine, creating a high-dose (HD) flu vaccine.
Why is there a need for an HD vaccine? “The elderly population bears the greatest burden from the flu,” explained Launay. “This is due to three factors: An aging immune system, a higher number of comorbidities, and increased frailty.” Standard-dose flu vaccines are seen as offering suboptimal protection for those older than 65 years, which led to the development of a quadrivalent vaccine with four times the antigen dose of standard flu vaccines. This HD vaccine was introduced in France during the 2021/2022 flu season. A real-world cohort study has since been conducted to evaluate its effectiveness in the target population — those aged 65 years or older. The results were recently published in Clinical Microbiology and Infection.
Cohort Study
The study included 405,385 noninstitutionalized people aged 65 years or older matched with 1,621,540 individuals in a 1:4 ratio. The first group received the HD vaccine, while the second group received the standard-dose vaccine. Both the groups had an average age of 77 years, with 56% women, and 51% vaccinated in pharmacies. The majority had been previously vaccinated against flu (91%), and 97% had completed a full COVID-19 vaccination schedule. More than half had at least one chronic illness.
Hospitalization rates for flu — the study’s primary outcome — were 69.5 vs 90.5 per 100,000 person-years in the HD vs standard-dose group. This represented a 23.3% reduction (95% CI, 8.4-35.8; P = .003).
Strengths and Limitations
Among the strengths of the study, Launay highlighted the large number of vaccinated participants older than 65 years — more than 7 million — and the widespread use of polymerase chain reaction flu tests in cases of hospitalization for respiratory infections, which improved flu coding in the database used. Additionally, the results were consistent with those of previous studies.
However, limitations included the retrospective design, which did not randomize participants and introduced potential bias. For example, the HD vaccine may have been prioritized for the oldest people or those with multiple comorbidities. Additionally, the 2021/2022 flu season was atypical, with the simultaneous circulation of the flu virus and SARS-CoV-2, as noted by Launay.
Conclusion
In conclusion, this first evaluation of the HD flu vaccine’s effectiveness in France showed a 25% reduction in hospitalizations, consistent with existing data covering 12 flu seasons. The vaccine has been available for a longer period in the United States and Northern Europe.
“The latest unpublished data from the 2022/23 season show a 27% reduction in hospitalizations with the HD vaccine in people over 65,” added Launay.
Note: Due to a pricing disagreement with the French government, Sanofi’s HD flu vaccine Efluelda, intended for people older than 65 years, will not be available this year. (See: Withdrawal of the Efluelda Influenza Vaccine: The Academy of Medicine Reacts). However, the company has submitted a dossier for a trivalent form for a return in the 2025/2026 season and is working on developing mRNA vaccines. Additionally, a combined flu/COVID-19 vaccine is currently in development.
The study was funded by Sanofi. Several authors are Sanofi employees. Odile Launay reported conflicts of interest with Sanofi, MSD, Pfizer, GSK, and Moderna.
This story was translated from Medscape’s French edition using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article appeared on Medscape.com.
How can the immunogenicity and effectiveness of flu vaccines be improved in older adults? Several strategies are available, one being the addition of an adjuvant. For example, the MF59-adjuvanted vaccine has shown superior immunogenicity. However, “we do not have data from controlled and randomized clinical trials showing superior clinical effectiveness versus the standard dose,” Professor Odile Launay, an infectious disease specialist at Cochin Hospital in Paris, France, noted during a press conference. Another option is to increase the antigen dose in the vaccine, creating a high-dose (HD) flu vaccine.
Why is there a need for an HD vaccine? “The elderly population bears the greatest burden from the flu,” explained Launay. “This is due to three factors: An aging immune system, a higher number of comorbidities, and increased frailty.” Standard-dose flu vaccines are seen as offering suboptimal protection for those older than 65 years, which led to the development of a quadrivalent vaccine with four times the antigen dose of standard flu vaccines. This HD vaccine was introduced in France during the 2021/2022 flu season. A real-world cohort study has since been conducted to evaluate its effectiveness in the target population — those aged 65 years or older. The results were recently published in Clinical Microbiology and Infection.
Cohort Study
The study included 405,385 noninstitutionalized people aged 65 years or older matched with 1,621,540 individuals in a 1:4 ratio. The first group received the HD vaccine, while the second group received the standard-dose vaccine. Both the groups had an average age of 77 years, with 56% women, and 51% vaccinated in pharmacies. The majority had been previously vaccinated against flu (91%), and 97% had completed a full COVID-19 vaccination schedule. More than half had at least one chronic illness.
Hospitalization rates for flu — the study’s primary outcome — were 69.5 vs 90.5 per 100,000 person-years in the HD vs standard-dose group. This represented a 23.3% reduction (95% CI, 8.4-35.8; P = .003).
Strengths and Limitations
Among the strengths of the study, Launay highlighted the large number of vaccinated participants older than 65 years — more than 7 million — and the widespread use of polymerase chain reaction flu tests in cases of hospitalization for respiratory infections, which improved flu coding in the database used. Additionally, the results were consistent with those of previous studies.
However, limitations included the retrospective design, which did not randomize participants and introduced potential bias. For example, the HD vaccine may have been prioritized for the oldest people or those with multiple comorbidities. Additionally, the 2021/2022 flu season was atypical, with the simultaneous circulation of the flu virus and SARS-CoV-2, as noted by Launay.
Conclusion
In conclusion, this first evaluation of the HD flu vaccine’s effectiveness in France showed a 25% reduction in hospitalizations, consistent with existing data covering 12 flu seasons. The vaccine has been available for a longer period in the United States and Northern Europe.
“The latest unpublished data from the 2022/23 season show a 27% reduction in hospitalizations with the HD vaccine in people over 65,” added Launay.
Note: Due to a pricing disagreement with the French government, Sanofi’s HD flu vaccine Efluelda, intended for people older than 65 years, will not be available this year. (See: Withdrawal of the Efluelda Influenza Vaccine: The Academy of Medicine Reacts). However, the company has submitted a dossier for a trivalent form for a return in the 2025/2026 season and is working on developing mRNA vaccines. Additionally, a combined flu/COVID-19 vaccine is currently in development.
The study was funded by Sanofi. Several authors are Sanofi employees. Odile Launay reported conflicts of interest with Sanofi, MSD, Pfizer, GSK, and Moderna.
This story was translated from Medscape’s French edition using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article appeared on Medscape.com.
EHR Prompt Helped Cut Acute Otitis Media Antibiotic Use by Half
LOS ANGELES — Embedding a new discharge order set into electronic health records (EHRs) with a preselected 5-day antibiotic course for children aged 2 years or older diagnosed with acute otitis media (AOM) cut antibiotic duration sharply, according to new data presented at the Infectious Disease Week (IDWeek) 2024 Annual Meeting.
“We were effectively able to cut antibiotic use in half by shortening the duration of treatment,” said lead author Joana Dimo, DO, a Pediatric Infectious Diseases fellow at the University of Colorado Denver/Children’s Hospital Colorado.
In the United States, 80% of children will experience otitis media during their lifetime. Untreated ear infections can lead to symptoms ranging from mild ear discharge to life-threatening conditions such as mastoiditis and intracranial abscesses.
Most Cases Resolve Without Antibiotics
Ear infections “are the leading reason for antibiotic prescriptions in kids,” Dimo noted, adding that 24% of all pediatric antibiotic prescriptions are for AOM. Amoxicillin is the preferred first-line treatment. “Research supports that 75% of children get better on their own without antibiotics, and when needed, short courses of just 5 days are safe and effective.”
Antibiotics can cause side effects such as diarrhea and rashes. “Each additional day of antibiotics that are not needed leads to more side effects,” Dimo said, as well as contributing to antibiotic resistance.
Dimo’s team implemented new EHR order sets across the University of Colorado/Children’s Hospital Colorado health network’s four emergency departments and four urgent care centers and included 31,929 patients in the study.
Then they conducted a retrospective review of patients 61 days to 18 years old who entered those settings and had confirmed AOM between January 2019 through December 2023, before and after the April 2021 intervention. The researchers also developed a guideline on managing ear infections to support clinicians as part of the intervention in December 2022.
Compliance Grew From 3% to 83%
Dimo said they found very few clinicians in their study had been prescribing according to current guidelines. Their results showed a jump from 3% to 83% in providers prescribing 5-day durations of antibiotics for children aged 2 years or older after their intervention.
The intervention did not lead to increased treatment failures or complications, she added. The team looked for diagnostic codes for mastoiditis, subperiosteal abscess, petrositis, labyrinthitis, meningitis, and intracranial abscess, and “none of our patients” developed any of those complications, Dimo said.
Dimo said the overall rate of prescribing, however, increased. Finding out why prescribing rates remained high throughout the study, before and after their intervention, is a question they are investigating in future work, she said.
Cost-Effective and Scalable
“The benefit of this strategy to other institutions is that it’s not labor-intensive. It’s cost-effective, and it can result in dramatic changes in antibiotic use,” Dimo said.
“In the outpatient setting, there’s still a lot of antibiotics being given unnecessarily to children with acute otitis media,” said William Schaffner, MD, infectious disease specialist at Vanderbilt University School of Medicine in Nashville, Tennessee, who was not part of the research. “The American Academy of Pediatrics has been working on that for about a decade — to get pediatricians attuned to when you use them. Most of these episodes of acute otitis media — it’s now well-established — are due to viral infections.”
He said that some physicians may still be defaulting to the longer doses — up to 10 days — that they may have learned in medical school or residency.
“The data would indicate that 5 days of treatment — when treatment is appropriate — is, in the vast majority of instances, sufficient,” Schaffner said.
The researchers “were remarkably successful,” he said, adding that another question is ripe for research. “They still have to get to this issue of whether all of these antibiotic starts were necessary.”
Not knowing whether antibiotic prescriptions in this study were warranted is a limitation of the study, Dimo said, as was not being able to track whether patients presented to institutions outside their own for a return visit or for complications.
She said she thinks one of the reasons for such a sharp increase in compliance was that clinicians in their system routinely use order sets, so using the new order sets easily became part of their workflow.
Dimo and Schaffner reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
LOS ANGELES — Embedding a new discharge order set into electronic health records (EHRs) with a preselected 5-day antibiotic course for children aged 2 years or older diagnosed with acute otitis media (AOM) cut antibiotic duration sharply, according to new data presented at the Infectious Disease Week (IDWeek) 2024 Annual Meeting.
“We were effectively able to cut antibiotic use in half by shortening the duration of treatment,” said lead author Joana Dimo, DO, a Pediatric Infectious Diseases fellow at the University of Colorado Denver/Children’s Hospital Colorado.
In the United States, 80% of children will experience otitis media during their lifetime. Untreated ear infections can lead to symptoms ranging from mild ear discharge to life-threatening conditions such as mastoiditis and intracranial abscesses.
Most Cases Resolve Without Antibiotics
Ear infections “are the leading reason for antibiotic prescriptions in kids,” Dimo noted, adding that 24% of all pediatric antibiotic prescriptions are for AOM. Amoxicillin is the preferred first-line treatment. “Research supports that 75% of children get better on their own without antibiotics, and when needed, short courses of just 5 days are safe and effective.”
Antibiotics can cause side effects such as diarrhea and rashes. “Each additional day of antibiotics that are not needed leads to more side effects,” Dimo said, as well as contributing to antibiotic resistance.
Dimo’s team implemented new EHR order sets across the University of Colorado/Children’s Hospital Colorado health network’s four emergency departments and four urgent care centers and included 31,929 patients in the study.
Then they conducted a retrospective review of patients 61 days to 18 years old who entered those settings and had confirmed AOM between January 2019 through December 2023, before and after the April 2021 intervention. The researchers also developed a guideline on managing ear infections to support clinicians as part of the intervention in December 2022.
Compliance Grew From 3% to 83%
Dimo said they found very few clinicians in their study had been prescribing according to current guidelines. Their results showed a jump from 3% to 83% in providers prescribing 5-day durations of antibiotics for children aged 2 years or older after their intervention.
The intervention did not lead to increased treatment failures or complications, she added. The team looked for diagnostic codes for mastoiditis, subperiosteal abscess, petrositis, labyrinthitis, meningitis, and intracranial abscess, and “none of our patients” developed any of those complications, Dimo said.
Dimo said the overall rate of prescribing, however, increased. Finding out why prescribing rates remained high throughout the study, before and after their intervention, is a question they are investigating in future work, she said.
Cost-Effective and Scalable
“The benefit of this strategy to other institutions is that it’s not labor-intensive. It’s cost-effective, and it can result in dramatic changes in antibiotic use,” Dimo said.
“In the outpatient setting, there’s still a lot of antibiotics being given unnecessarily to children with acute otitis media,” said William Schaffner, MD, infectious disease specialist at Vanderbilt University School of Medicine in Nashville, Tennessee, who was not part of the research. “The American Academy of Pediatrics has been working on that for about a decade — to get pediatricians attuned to when you use them. Most of these episodes of acute otitis media — it’s now well-established — are due to viral infections.”
He said that some physicians may still be defaulting to the longer doses — up to 10 days — that they may have learned in medical school or residency.
“The data would indicate that 5 days of treatment — when treatment is appropriate — is, in the vast majority of instances, sufficient,” Schaffner said.
The researchers “were remarkably successful,” he said, adding that another question is ripe for research. “They still have to get to this issue of whether all of these antibiotic starts were necessary.”
Not knowing whether antibiotic prescriptions in this study were warranted is a limitation of the study, Dimo said, as was not being able to track whether patients presented to institutions outside their own for a return visit or for complications.
She said she thinks one of the reasons for such a sharp increase in compliance was that clinicians in their system routinely use order sets, so using the new order sets easily became part of their workflow.
Dimo and Schaffner reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
LOS ANGELES — Embedding a new discharge order set into electronic health records (EHRs) with a preselected 5-day antibiotic course for children aged 2 years or older diagnosed with acute otitis media (AOM) cut antibiotic duration sharply, according to new data presented at the Infectious Disease Week (IDWeek) 2024 Annual Meeting.
“We were effectively able to cut antibiotic use in half by shortening the duration of treatment,” said lead author Joana Dimo, DO, a Pediatric Infectious Diseases fellow at the University of Colorado Denver/Children’s Hospital Colorado.
In the United States, 80% of children will experience otitis media during their lifetime. Untreated ear infections can lead to symptoms ranging from mild ear discharge to life-threatening conditions such as mastoiditis and intracranial abscesses.
Most Cases Resolve Without Antibiotics
Ear infections “are the leading reason for antibiotic prescriptions in kids,” Dimo noted, adding that 24% of all pediatric antibiotic prescriptions are for AOM. Amoxicillin is the preferred first-line treatment. “Research supports that 75% of children get better on their own without antibiotics, and when needed, short courses of just 5 days are safe and effective.”
Antibiotics can cause side effects such as diarrhea and rashes. “Each additional day of antibiotics that are not needed leads to more side effects,” Dimo said, as well as contributing to antibiotic resistance.
Dimo’s team implemented new EHR order sets across the University of Colorado/Children’s Hospital Colorado health network’s four emergency departments and four urgent care centers and included 31,929 patients in the study.
Then they conducted a retrospective review of patients 61 days to 18 years old who entered those settings and had confirmed AOM between January 2019 through December 2023, before and after the April 2021 intervention. The researchers also developed a guideline on managing ear infections to support clinicians as part of the intervention in December 2022.
Compliance Grew From 3% to 83%
Dimo said they found very few clinicians in their study had been prescribing according to current guidelines. Their results showed a jump from 3% to 83% in providers prescribing 5-day durations of antibiotics for children aged 2 years or older after their intervention.
The intervention did not lead to increased treatment failures or complications, she added. The team looked for diagnostic codes for mastoiditis, subperiosteal abscess, petrositis, labyrinthitis, meningitis, and intracranial abscess, and “none of our patients” developed any of those complications, Dimo said.
Dimo said the overall rate of prescribing, however, increased. Finding out why prescribing rates remained high throughout the study, before and after their intervention, is a question they are investigating in future work, she said.
Cost-Effective and Scalable
“The benefit of this strategy to other institutions is that it’s not labor-intensive. It’s cost-effective, and it can result in dramatic changes in antibiotic use,” Dimo said.
“In the outpatient setting, there’s still a lot of antibiotics being given unnecessarily to children with acute otitis media,” said William Schaffner, MD, infectious disease specialist at Vanderbilt University School of Medicine in Nashville, Tennessee, who was not part of the research. “The American Academy of Pediatrics has been working on that for about a decade — to get pediatricians attuned to when you use them. Most of these episodes of acute otitis media — it’s now well-established — are due to viral infections.”
He said that some physicians may still be defaulting to the longer doses — up to 10 days — that they may have learned in medical school or residency.
“The data would indicate that 5 days of treatment — when treatment is appropriate — is, in the vast majority of instances, sufficient,” Schaffner said.
The researchers “were remarkably successful,” he said, adding that another question is ripe for research. “They still have to get to this issue of whether all of these antibiotic starts were necessary.”
Not knowing whether antibiotic prescriptions in this study were warranted is a limitation of the study, Dimo said, as was not being able to track whether patients presented to institutions outside their own for a return visit or for complications.
She said she thinks one of the reasons for such a sharp increase in compliance was that clinicians in their system routinely use order sets, so using the new order sets easily became part of their workflow.
Dimo and Schaffner reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FROM IDWEEK 2024
One-Dose HPV Vaccine Program Would Be Efficient in Canada
In Canada, switching to a one-dose, gender-neutral vaccination program for human papillomavirus (HPV) could use vaccine doses more efficiently and prevent a similar number of cervical cancer cases, compared with a two-dose program, according to a new modeling analysis.
If vaccine protection remains high during the ages of peak sexual activity, all one-dose vaccination options are projected to be “substantially more efficient” than two-dose programs, even in the most pessimistic scenarios, the study authors wrote.
In addition, the scenarios projected the elimination of cervical cancer in Canada between 2032 and 2040. HPV can also lead to oral, throat, and penile cancers, and most are preventable through vaccination.
“The COVID-19 pandemic has impacted HPV vaccination in Canada, particularly among vulnerable population subgroups,” said study author Chantal Sauvageau, MD, a consultant in infectious diseases at the National Institute of Public Health of Quebec and associate professor of social and preventive medicine at the University of Laval, Quebec City, Canada.
Switching to one-dose vaccination would offer potential economic savings and programmatic flexibility, she added. The change also could enable investments aimed at increasing vaccination rates in regions where coverage is suboptimal, as well as in subgroups with a high HPV burden. Such initiatives could mitigate the pandemic’s impact on health programs and reduce inequalities.
The study was published online in CMAJ.
Vaccination Program Changes
Globally, countries have been investigating whether to shift from a two-dose to a one-dose HPV vaccine strategy since the World Health Organization’s Strategic Advisory Group of Experts on Immunization issued a single-dose recommendation in 2022.
In July, Canada’s National Advisory Committee on Immunization (NACI) updated its guidelines to recommend the single-dose approach for ages 9-20 years. The change aligns Canada with 35 other countries, including Australia and the United Kingdom. Canada›s vaccine advisory group still recommends two doses for ages 21-26 years and three doses for patients who are immunocompromised or have HIV.
To help inform new NACI policies, Sauvageau and colleagues modeled several one-dose and two-dose strategies using HPV-ADVISE, an individual-based transmission-dynamic model of HPV infections and diseases. They looked at vaccination programs in Quebec, which has a high HPV vaccine coverage rate of around 85%, and Ontario, which has lower coverage of around 65%.
For one-dose programs, the researchers analyzed noninferior (98% efficacy) and pessimistic (90% efficacy) scenarios and different average vaccine duration periods, including lifelong, 30-year, and 25-year coverage. They compared the scenarios with a two-dose program with 98% efficacy and lifelong duration, estimating the relative reduction in HPV-16 infection and cervical cancer incidence and the number of doses needed to prevent one cervical cancer case.
Overall, the model projected that gender-neutral HPV vaccine programs with either two doses or a noninferior one dose would nearly eliminate HPV-16 infection by 2040-2045 in Quebec and reduce infection by more than 90% in Ontario. Under a one-dose strategy with 90% vaccine efficacy, rebounds in HPV-16 infection would start more than 25-30 years after a switch to a lower-dose strategy, thus providing time for officials to detect any signs of waning efficacy and change policies, if needed, the authors wrote.
In addition, the model projected that a noninferior one-dose, gender-neutral HPV vaccination program would avert a similar number of cervical cancer cases, compared with a two-dose program. The reduction would be about 60% in Quebec and 55% in Ontario, compared with no vaccination. Under the most pessimistic scenario with 25-year vaccine duration, a one-dose program would be slightly less effective in averting cancer: about 3% lower than a two-dose program over 100 years.
All one-dose scenarios were projected to lead to the elimination of cervical cancer in 8-16 years — at fewer than four cervical cancer cases per 100,000 female-years.
One-dose programs would also lead to more efficient use of vaccine doses, with about 800-1000 doses needed to prevent one cervical cancer case in a one-dose program and more than 10,000 incremental doses needed to prevent one additional cervical cancer case in a two-dose program.
What Next?
In Canada, the HPV vaccine is authorized for patients aged 9-45 years. Current immunization coverage among adolescents and young adults varies across provinces and falls below the national target of 90%. In its July 2024 update, NACI estimated that 76% of 14-year-olds of both genders received at least one vaccine dose and that 67% received two doses in 2023. Vaccine uptake was slightly higher among girls than boys.
To boost the coverage rate, shifting to a one-dose schedule could appeal to young people, as well as maintain vaccination efficacy.
“When you look at the studies that have been published worldwide, the effectiveness of one dose of the HPV vaccine is actually quite high,” said Caroline Quach-Thanh, MD, professor of microbiology, infectious diseases, immunology, and pediatrics at the University of Montreal, Quebec, Canada.
Quach-Thanh, who wasn’t involved with this study, previously served as NACI chair and now serves as chair of the Quebec Immunization Committee.
“In terms of prevention of HPV infections that may lead to cancer, whether you give one dose or two doses basically gives you the same amount of protection,” she said.
However, not all physicians agree about the switch in vaccination approaches. In early October, the Federation of Medical Women of Canada released a report with 12 recommendations to increase HPV vaccination rates, including a call for healthcare providers to continue with multidose immunization schedules for now.
“Vaccination is the most powerful action we can take in preventing HPV-related cancers. Canada is falling behind, but we can get back on track if we act quickly,” said Vivien Brown, MD, chair of the group’s HPV Immunization Task Force, chair and cofounder of HPV Prevention Week in Canada, and a past president of the federation.
After the NACI update in July, the task force evaluated the risks and benefits of a single-dose vaccine regimen, she said. They concluded that a multidose schedule should continue at this time because of its proven effectiveness.
“Until more research on the efficacy of a single-dose schedule becomes available, healthcare providers and public health agencies should continue to offer patients a multidose schedule,” said Brown. “This is the only way to ensure individuals are protected against HPV infection and cancer over the long term.”
The study was supported by the Public Health Agency of Canada, the Canadian Institutes of Health Research, the Bill & Melinda Gates Foundation, and Canadian Immunization Research Network. Sauvageau, Quach-Thanh, and Brown declared no relevant financial disclosures.
A version of this article first appeared on Medscape.com.
In Canada, switching to a one-dose, gender-neutral vaccination program for human papillomavirus (HPV) could use vaccine doses more efficiently and prevent a similar number of cervical cancer cases, compared with a two-dose program, according to a new modeling analysis.
If vaccine protection remains high during the ages of peak sexual activity, all one-dose vaccination options are projected to be “substantially more efficient” than two-dose programs, even in the most pessimistic scenarios, the study authors wrote.
In addition, the scenarios projected the elimination of cervical cancer in Canada between 2032 and 2040. HPV can also lead to oral, throat, and penile cancers, and most are preventable through vaccination.
“The COVID-19 pandemic has impacted HPV vaccination in Canada, particularly among vulnerable population subgroups,” said study author Chantal Sauvageau, MD, a consultant in infectious diseases at the National Institute of Public Health of Quebec and associate professor of social and preventive medicine at the University of Laval, Quebec City, Canada.
Switching to one-dose vaccination would offer potential economic savings and programmatic flexibility, she added. The change also could enable investments aimed at increasing vaccination rates in regions where coverage is suboptimal, as well as in subgroups with a high HPV burden. Such initiatives could mitigate the pandemic’s impact on health programs and reduce inequalities.
The study was published online in CMAJ.
Vaccination Program Changes
Globally, countries have been investigating whether to shift from a two-dose to a one-dose HPV vaccine strategy since the World Health Organization’s Strategic Advisory Group of Experts on Immunization issued a single-dose recommendation in 2022.
In July, Canada’s National Advisory Committee on Immunization (NACI) updated its guidelines to recommend the single-dose approach for ages 9-20 years. The change aligns Canada with 35 other countries, including Australia and the United Kingdom. Canada›s vaccine advisory group still recommends two doses for ages 21-26 years and three doses for patients who are immunocompromised or have HIV.
To help inform new NACI policies, Sauvageau and colleagues modeled several one-dose and two-dose strategies using HPV-ADVISE, an individual-based transmission-dynamic model of HPV infections and diseases. They looked at vaccination programs in Quebec, which has a high HPV vaccine coverage rate of around 85%, and Ontario, which has lower coverage of around 65%.
For one-dose programs, the researchers analyzed noninferior (98% efficacy) and pessimistic (90% efficacy) scenarios and different average vaccine duration periods, including lifelong, 30-year, and 25-year coverage. They compared the scenarios with a two-dose program with 98% efficacy and lifelong duration, estimating the relative reduction in HPV-16 infection and cervical cancer incidence and the number of doses needed to prevent one cervical cancer case.
Overall, the model projected that gender-neutral HPV vaccine programs with either two doses or a noninferior one dose would nearly eliminate HPV-16 infection by 2040-2045 in Quebec and reduce infection by more than 90% in Ontario. Under a one-dose strategy with 90% vaccine efficacy, rebounds in HPV-16 infection would start more than 25-30 years after a switch to a lower-dose strategy, thus providing time for officials to detect any signs of waning efficacy and change policies, if needed, the authors wrote.
In addition, the model projected that a noninferior one-dose, gender-neutral HPV vaccination program would avert a similar number of cervical cancer cases, compared with a two-dose program. The reduction would be about 60% in Quebec and 55% in Ontario, compared with no vaccination. Under the most pessimistic scenario with 25-year vaccine duration, a one-dose program would be slightly less effective in averting cancer: about 3% lower than a two-dose program over 100 years.
All one-dose scenarios were projected to lead to the elimination of cervical cancer in 8-16 years — at fewer than four cervical cancer cases per 100,000 female-years.
One-dose programs would also lead to more efficient use of vaccine doses, with about 800-1000 doses needed to prevent one cervical cancer case in a one-dose program and more than 10,000 incremental doses needed to prevent one additional cervical cancer case in a two-dose program.
What Next?
In Canada, the HPV vaccine is authorized for patients aged 9-45 years. Current immunization coverage among adolescents and young adults varies across provinces and falls below the national target of 90%. In its July 2024 update, NACI estimated that 76% of 14-year-olds of both genders received at least one vaccine dose and that 67% received two doses in 2023. Vaccine uptake was slightly higher among girls than boys.
To boost the coverage rate, shifting to a one-dose schedule could appeal to young people, as well as maintain vaccination efficacy.
“When you look at the studies that have been published worldwide, the effectiveness of one dose of the HPV vaccine is actually quite high,” said Caroline Quach-Thanh, MD, professor of microbiology, infectious diseases, immunology, and pediatrics at the University of Montreal, Quebec, Canada.
Quach-Thanh, who wasn’t involved with this study, previously served as NACI chair and now serves as chair of the Quebec Immunization Committee.
“In terms of prevention of HPV infections that may lead to cancer, whether you give one dose or two doses basically gives you the same amount of protection,” she said.
However, not all physicians agree about the switch in vaccination approaches. In early October, the Federation of Medical Women of Canada released a report with 12 recommendations to increase HPV vaccination rates, including a call for healthcare providers to continue with multidose immunization schedules for now.
“Vaccination is the most powerful action we can take in preventing HPV-related cancers. Canada is falling behind, but we can get back on track if we act quickly,” said Vivien Brown, MD, chair of the group’s HPV Immunization Task Force, chair and cofounder of HPV Prevention Week in Canada, and a past president of the federation.
After the NACI update in July, the task force evaluated the risks and benefits of a single-dose vaccine regimen, she said. They concluded that a multidose schedule should continue at this time because of its proven effectiveness.
“Until more research on the efficacy of a single-dose schedule becomes available, healthcare providers and public health agencies should continue to offer patients a multidose schedule,” said Brown. “This is the only way to ensure individuals are protected against HPV infection and cancer over the long term.”
The study was supported by the Public Health Agency of Canada, the Canadian Institutes of Health Research, the Bill & Melinda Gates Foundation, and Canadian Immunization Research Network. Sauvageau, Quach-Thanh, and Brown declared no relevant financial disclosures.
A version of this article first appeared on Medscape.com.
In Canada, switching to a one-dose, gender-neutral vaccination program for human papillomavirus (HPV) could use vaccine doses more efficiently and prevent a similar number of cervical cancer cases, compared with a two-dose program, according to a new modeling analysis.
If vaccine protection remains high during the ages of peak sexual activity, all one-dose vaccination options are projected to be “substantially more efficient” than two-dose programs, even in the most pessimistic scenarios, the study authors wrote.
In addition, the scenarios projected the elimination of cervical cancer in Canada between 2032 and 2040. HPV can also lead to oral, throat, and penile cancers, and most are preventable through vaccination.
“The COVID-19 pandemic has impacted HPV vaccination in Canada, particularly among vulnerable population subgroups,” said study author Chantal Sauvageau, MD, a consultant in infectious diseases at the National Institute of Public Health of Quebec and associate professor of social and preventive medicine at the University of Laval, Quebec City, Canada.
Switching to one-dose vaccination would offer potential economic savings and programmatic flexibility, she added. The change also could enable investments aimed at increasing vaccination rates in regions where coverage is suboptimal, as well as in subgroups with a high HPV burden. Such initiatives could mitigate the pandemic’s impact on health programs and reduce inequalities.
The study was published online in CMAJ.
Vaccination Program Changes
Globally, countries have been investigating whether to shift from a two-dose to a one-dose HPV vaccine strategy since the World Health Organization’s Strategic Advisory Group of Experts on Immunization issued a single-dose recommendation in 2022.
In July, Canada’s National Advisory Committee on Immunization (NACI) updated its guidelines to recommend the single-dose approach for ages 9-20 years. The change aligns Canada with 35 other countries, including Australia and the United Kingdom. Canada›s vaccine advisory group still recommends two doses for ages 21-26 years and three doses for patients who are immunocompromised or have HIV.
To help inform new NACI policies, Sauvageau and colleagues modeled several one-dose and two-dose strategies using HPV-ADVISE, an individual-based transmission-dynamic model of HPV infections and diseases. They looked at vaccination programs in Quebec, which has a high HPV vaccine coverage rate of around 85%, and Ontario, which has lower coverage of around 65%.
For one-dose programs, the researchers analyzed noninferior (98% efficacy) and pessimistic (90% efficacy) scenarios and different average vaccine duration periods, including lifelong, 30-year, and 25-year coverage. They compared the scenarios with a two-dose program with 98% efficacy and lifelong duration, estimating the relative reduction in HPV-16 infection and cervical cancer incidence and the number of doses needed to prevent one cervical cancer case.
Overall, the model projected that gender-neutral HPV vaccine programs with either two doses or a noninferior one dose would nearly eliminate HPV-16 infection by 2040-2045 in Quebec and reduce infection by more than 90% in Ontario. Under a one-dose strategy with 90% vaccine efficacy, rebounds in HPV-16 infection would start more than 25-30 years after a switch to a lower-dose strategy, thus providing time for officials to detect any signs of waning efficacy and change policies, if needed, the authors wrote.
In addition, the model projected that a noninferior one-dose, gender-neutral HPV vaccination program would avert a similar number of cervical cancer cases, compared with a two-dose program. The reduction would be about 60% in Quebec and 55% in Ontario, compared with no vaccination. Under the most pessimistic scenario with 25-year vaccine duration, a one-dose program would be slightly less effective in averting cancer: about 3% lower than a two-dose program over 100 years.
All one-dose scenarios were projected to lead to the elimination of cervical cancer in 8-16 years — at fewer than four cervical cancer cases per 100,000 female-years.
One-dose programs would also lead to more efficient use of vaccine doses, with about 800-1000 doses needed to prevent one cervical cancer case in a one-dose program and more than 10,000 incremental doses needed to prevent one additional cervical cancer case in a two-dose program.
What Next?
In Canada, the HPV vaccine is authorized for patients aged 9-45 years. Current immunization coverage among adolescents and young adults varies across provinces and falls below the national target of 90%. In its July 2024 update, NACI estimated that 76% of 14-year-olds of both genders received at least one vaccine dose and that 67% received two doses in 2023. Vaccine uptake was slightly higher among girls than boys.
To boost the coverage rate, shifting to a one-dose schedule could appeal to young people, as well as maintain vaccination efficacy.
“When you look at the studies that have been published worldwide, the effectiveness of one dose of the HPV vaccine is actually quite high,” said Caroline Quach-Thanh, MD, professor of microbiology, infectious diseases, immunology, and pediatrics at the University of Montreal, Quebec, Canada.
Quach-Thanh, who wasn’t involved with this study, previously served as NACI chair and now serves as chair of the Quebec Immunization Committee.
“In terms of prevention of HPV infections that may lead to cancer, whether you give one dose or two doses basically gives you the same amount of protection,” she said.
However, not all physicians agree about the switch in vaccination approaches. In early October, the Federation of Medical Women of Canada released a report with 12 recommendations to increase HPV vaccination rates, including a call for healthcare providers to continue with multidose immunization schedules for now.
“Vaccination is the most powerful action we can take in preventing HPV-related cancers. Canada is falling behind, but we can get back on track if we act quickly,” said Vivien Brown, MD, chair of the group’s HPV Immunization Task Force, chair and cofounder of HPV Prevention Week in Canada, and a past president of the federation.
After the NACI update in July, the task force evaluated the risks and benefits of a single-dose vaccine regimen, she said. They concluded that a multidose schedule should continue at this time because of its proven effectiveness.
“Until more research on the efficacy of a single-dose schedule becomes available, healthcare providers and public health agencies should continue to offer patients a multidose schedule,” said Brown. “This is the only way to ensure individuals are protected against HPV infection and cancer over the long term.”
The study was supported by the Public Health Agency of Canada, the Canadian Institutes of Health Research, the Bill & Melinda Gates Foundation, and Canadian Immunization Research Network. Sauvageau, Quach-Thanh, and Brown declared no relevant financial disclosures.
A version of this article first appeared on Medscape.com.
FROM CMAJ
Heart Attack, Stroke Survivors at High Risk for Long COVID
Primary care doctors and specialists should advise patients who have already experienced a heart attack or stroke that they are at a higher risk for long COVID and need to take steps to avoid contracting the virus, according to new research.
The study, led by researchers at Columbia University, New York City, suggests that anyone with cardiovascular disease (CVD) — defined as having experienced a heart attack or stroke — should consider getting the updated COVID vaccine boosters. They also suggest patients with CVD take other steps to avoid an acute infection, such as avoiding crowded indoor spaces.
There is no specific test or treatment for long COVID, which can become disabling and chronic. Long COVID is defined by the failure to recover from acute COVID-19 in 90 days.
The scientists used data from nearly 5000 people enrolled in 14 established, ongoing research programs, including the 76-year-old Framingham Heart Study. The results of the analysis of the “mega-cohort” were published in JAMA Network Open.
Most of the 14 studies already had 10-20 years of data on the cardiac health of thousands of enrollees, said Norrina B. Allen, one of the authors and a cardiac epidemiologist at Northwestern University Feinberg School of Medicine in Chicago, Illinois.
“This is a particularly strong study that looked at risk factors — or individual health — prior to developing COVID and their impact on the likely of recovering from COVID,” she said.
In addition to those with CVD, women and adults with preexisting chronic illnesses took longer to recover.
More than 20% of those in the large, racially and ethnically diverse US population–based study did not recover from COVID in 90 days. The researchers found that the median self-reported time to recovery from acute infection was 20 days.
While women and those with chronic illness had a higher risk for long COVID, vaccination and infection with the Omicron variant wave were associated with shorter recovery times.
These findings make sense, said Ziyad Al-Aly, MD, chief of research at Veterans Affairs St. Louis Health Care System and clinical epidemiologist at Washington University in St. Louis, Missouri.
“We also see that COVID-19 can lead to new-onset cardiovascular disease,” said Al-Aly, who was not involved in the study. “There is clearly a (link) between COVID and cardiovascular disease. These two seem to be intimately intertwined. In my view, this emphasizes the importance of targeting these individuals for vaccination and potentially antivirals (when they get infected) to help reduce their risk of adverse events and ameliorate their chance of full and fast recovery.”
The study used data from the Collaborative Cohort of Cohorts for COVID-19 Research. The long list of researchers contributing to this study includes epidemiologists, biostatisticians, neurologists, pulmonologists, and cardiologists. The data come from a list of cohorts like the Framingham Heart Study, which identified key risk factors for CVD, including cholesterol levels. Other studies include the Atherosclerosis Risk in Communities study, which began in the mid-1980s. Researchers there recruited a cohort of 15,792 men and women in rural North Carolina and Mississippi and suburban Minneapolis. They enrolled a high number of African American participants, who have been underrepresented in past studies. Other cohorts focused on young adults with CVD and Hispanics, while another focused on people with chronic obstructive pulmonary disease.
Lead author Elizabeth C. Oelsner, MD, of Columbia University Irving Medical Center in New York City, said she was not surprised by the CVD-long COVID link.
“We were aware that individuals with CVD were at higher risk of a more severe acute infection,” she said. “We were also seeing evidence that long and severe infection led to persistent symptoms.”
Oelsner noted that many patients still take more than 3 months to recover, even during the Omicron wave.
“While that has improved over the course of the pandemic, many individuals are taking a very long time to recover, and that can have a huge burden on the patient,” she said.
She encourages healthcare providers to tell patients at higher risk to take steps to avoid the virus, including vaccination and boosters.
A version of this article first appeared on Medscape.com.
Primary care doctors and specialists should advise patients who have already experienced a heart attack or stroke that they are at a higher risk for long COVID and need to take steps to avoid contracting the virus, according to new research.
The study, led by researchers at Columbia University, New York City, suggests that anyone with cardiovascular disease (CVD) — defined as having experienced a heart attack or stroke — should consider getting the updated COVID vaccine boosters. They also suggest patients with CVD take other steps to avoid an acute infection, such as avoiding crowded indoor spaces.
There is no specific test or treatment for long COVID, which can become disabling and chronic. Long COVID is defined by the failure to recover from acute COVID-19 in 90 days.
The scientists used data from nearly 5000 people enrolled in 14 established, ongoing research programs, including the 76-year-old Framingham Heart Study. The results of the analysis of the “mega-cohort” were published in JAMA Network Open.
Most of the 14 studies already had 10-20 years of data on the cardiac health of thousands of enrollees, said Norrina B. Allen, one of the authors and a cardiac epidemiologist at Northwestern University Feinberg School of Medicine in Chicago, Illinois.
“This is a particularly strong study that looked at risk factors — or individual health — prior to developing COVID and their impact on the likely of recovering from COVID,” she said.
In addition to those with CVD, women and adults with preexisting chronic illnesses took longer to recover.
More than 20% of those in the large, racially and ethnically diverse US population–based study did not recover from COVID in 90 days. The researchers found that the median self-reported time to recovery from acute infection was 20 days.
While women and those with chronic illness had a higher risk for long COVID, vaccination and infection with the Omicron variant wave were associated with shorter recovery times.
These findings make sense, said Ziyad Al-Aly, MD, chief of research at Veterans Affairs St. Louis Health Care System and clinical epidemiologist at Washington University in St. Louis, Missouri.
“We also see that COVID-19 can lead to new-onset cardiovascular disease,” said Al-Aly, who was not involved in the study. “There is clearly a (link) between COVID and cardiovascular disease. These two seem to be intimately intertwined. In my view, this emphasizes the importance of targeting these individuals for vaccination and potentially antivirals (when they get infected) to help reduce their risk of adverse events and ameliorate their chance of full and fast recovery.”
The study used data from the Collaborative Cohort of Cohorts for COVID-19 Research. The long list of researchers contributing to this study includes epidemiologists, biostatisticians, neurologists, pulmonologists, and cardiologists. The data come from a list of cohorts like the Framingham Heart Study, which identified key risk factors for CVD, including cholesterol levels. Other studies include the Atherosclerosis Risk in Communities study, which began in the mid-1980s. Researchers there recruited a cohort of 15,792 men and women in rural North Carolina and Mississippi and suburban Minneapolis. They enrolled a high number of African American participants, who have been underrepresented in past studies. Other cohorts focused on young adults with CVD and Hispanics, while another focused on people with chronic obstructive pulmonary disease.
Lead author Elizabeth C. Oelsner, MD, of Columbia University Irving Medical Center in New York City, said she was not surprised by the CVD-long COVID link.
“We were aware that individuals with CVD were at higher risk of a more severe acute infection,” she said. “We were also seeing evidence that long and severe infection led to persistent symptoms.”
Oelsner noted that many patients still take more than 3 months to recover, even during the Omicron wave.
“While that has improved over the course of the pandemic, many individuals are taking a very long time to recover, and that can have a huge burden on the patient,” she said.
She encourages healthcare providers to tell patients at higher risk to take steps to avoid the virus, including vaccination and boosters.
A version of this article first appeared on Medscape.com.
Primary care doctors and specialists should advise patients who have already experienced a heart attack or stroke that they are at a higher risk for long COVID and need to take steps to avoid contracting the virus, according to new research.
The study, led by researchers at Columbia University, New York City, suggests that anyone with cardiovascular disease (CVD) — defined as having experienced a heart attack or stroke — should consider getting the updated COVID vaccine boosters. They also suggest patients with CVD take other steps to avoid an acute infection, such as avoiding crowded indoor spaces.
There is no specific test or treatment for long COVID, which can become disabling and chronic. Long COVID is defined by the failure to recover from acute COVID-19 in 90 days.
The scientists used data from nearly 5000 people enrolled in 14 established, ongoing research programs, including the 76-year-old Framingham Heart Study. The results of the analysis of the “mega-cohort” were published in JAMA Network Open.
Most of the 14 studies already had 10-20 years of data on the cardiac health of thousands of enrollees, said Norrina B. Allen, one of the authors and a cardiac epidemiologist at Northwestern University Feinberg School of Medicine in Chicago, Illinois.
“This is a particularly strong study that looked at risk factors — or individual health — prior to developing COVID and their impact on the likely of recovering from COVID,” she said.
In addition to those with CVD, women and adults with preexisting chronic illnesses took longer to recover.
More than 20% of those in the large, racially and ethnically diverse US population–based study did not recover from COVID in 90 days. The researchers found that the median self-reported time to recovery from acute infection was 20 days.
While women and those with chronic illness had a higher risk for long COVID, vaccination and infection with the Omicron variant wave were associated with shorter recovery times.
These findings make sense, said Ziyad Al-Aly, MD, chief of research at Veterans Affairs St. Louis Health Care System and clinical epidemiologist at Washington University in St. Louis, Missouri.
“We also see that COVID-19 can lead to new-onset cardiovascular disease,” said Al-Aly, who was not involved in the study. “There is clearly a (link) between COVID and cardiovascular disease. These two seem to be intimately intertwined. In my view, this emphasizes the importance of targeting these individuals for vaccination and potentially antivirals (when they get infected) to help reduce their risk of adverse events and ameliorate their chance of full and fast recovery.”
The study used data from the Collaborative Cohort of Cohorts for COVID-19 Research. The long list of researchers contributing to this study includes epidemiologists, biostatisticians, neurologists, pulmonologists, and cardiologists. The data come from a list of cohorts like the Framingham Heart Study, which identified key risk factors for CVD, including cholesterol levels. Other studies include the Atherosclerosis Risk in Communities study, which began in the mid-1980s. Researchers there recruited a cohort of 15,792 men and women in rural North Carolina and Mississippi and suburban Minneapolis. They enrolled a high number of African American participants, who have been underrepresented in past studies. Other cohorts focused on young adults with CVD and Hispanics, while another focused on people with chronic obstructive pulmonary disease.
Lead author Elizabeth C. Oelsner, MD, of Columbia University Irving Medical Center in New York City, said she was not surprised by the CVD-long COVID link.
“We were aware that individuals with CVD were at higher risk of a more severe acute infection,” she said. “We were also seeing evidence that long and severe infection led to persistent symptoms.”
Oelsner noted that many patients still take more than 3 months to recover, even during the Omicron wave.
“While that has improved over the course of the pandemic, many individuals are taking a very long time to recover, and that can have a huge burden on the patient,” she said.
She encourages healthcare providers to tell patients at higher risk to take steps to avoid the virus, including vaccination and boosters.
A version of this article first appeared on Medscape.com.
Live Rotavirus Vaccine Safe for Newborns of Biologic-Treated Moms With IBD
No adverse events or impairment of the immune system emerged in babies at 7 days, 1 month, and 9 months post vaccination, in findings from a small Canadian study published in Clinical Gastroenterology and Hepatology.
The study found normal extended immune function testing in infants despite third-trimester maternal biologic therapy and regardless of circulating drug levels. The data provide reassurance about live rotavirus vaccination in this population and may also offer insights into the safety of other live vaccines in biologic-exposed individuals, wrote investigators led by gastroenterologist Cynthia H. Seow, MD, a professor in the Cumming School of Medicine at the University of Calgary in Alberta, Canada.
“Despite the well-established safety and effectiveness of non–live vaccination in individuals with IBD, including those on immunomodulators and biologic therapy, vaccine uptake in pregnant women with IBD and their infants remains suboptimal,” Seow said in an interview. This largely arises from maternal and physician concerns regarding transplacental transfer of IBD therapies and their impact on the safety of vaccination.
“These concerns were heightened after reports emerged of five fatal outcomes following the administration of the live Bacille Calmette-Guérin [BCG] vaccine in biologic-exposed infants. However, it had already been reported that inadvertent administration of the live oral rotavirus vaccine, a very different vaccine in terms of target and mechanism of action, in biologic-exposed individuals had not been associated with significant adverse effects,” she said.
They undertook their analysis with the hypothesis that vaccination would carry low risk, although the live oral vaccine is not currently recommended in biologic-exposed infants. “Yet rotavirus is a leading cause of severe, dehydrating diarrhea in children under the age of 5 years globally, and vaccination has led to significant reductions in hospitalizations and mortality,” Seow added.
Provision of the vaccine to anti–tumor necrosis factor (TNF)–exposed infants has been incorporated into the Canadian Public Health and Immunization guidelines, as the majority of the biologic-exposed infants were exposed to anti-TNF agents. “And with collection of further data, we expect that this will be extended to other biologic agent exposure. These data are important to pregnant women with IBD as they help to normalize their care. Pregnancy is difficult enough without having to remember exceptions to care,” Seow said.
“Before some of the studies came out, broad guidelines recommended that live vaccines should not be used in biologic-exposed infants, but this had been thought to be overly zealous and too conservative, and the risk was thought to be low,” said Elizabeth Spencer, MD, an assistant professor of pediatrics in the Division of Pediatric Gastroenterology at the Icahn School of Medicine at Mount Sinai in New York City, in an interview. Spencer was not involved in the Canadian study.
“At our center, we had some moms on biologics during pregnancy who forgot and had their babies vaccinated for rotavirus, and the babies were all fine,” she said.
The safety of this vaccine has been confirmed by several small studies and recently the PIANO Helmsley Global Consensus on Pregnancy and Inflammatory Bowel Disease, which was presented at Digestive Disease Week 2024. The consensus encompasses preconception counseling and the safety of IBD medications during pregnancy and lactation.
“Another concern, however, was that giving a live GI bug like rotavirus to babies might overstimulate their immune systems and provoke IBD,” Spencer added. “While a number of population-based studies in the US and Europe showed that was not the case, at least in the general population, there was a suggestion that, down the road, vaccination might be mildly protective against IBD in some cases.”
She added the caveat that these studies were not done in mothers and their babies with IBD, who might be inherently at greater risk for IBD. “So, a question for future research would be, ‘Is immune stimulation of the gut in IBD moms and their babies a good or a bad thing for their gut?’ ”
Spencer conceded that “the data present a bit of a blurry picture, but I think it’s always better just to vaccinate according to the regular schedule. The current data say there is no added risk, but it would be nice to look specifically at risk in moms with IBD and their children.”
The Study
The prospective cohort study is a substudy of a larger 2023 one that included biologic use in a range of maternal illnesses, not just IBD.
For the current study, Seow and colleagues identified 57 infants born to 52 mothers with IBD attending a pregnancy clinic at the University of Calgary in the period 2019-2023. Almost 81% of the mothers had Crohn’s disease, and the median duration of IBD was 10 years. The median gestational age at delivery was 39 weeks, and almost 60% of deliveries were vaginal. The infants had been exposed in utero to infliximab (n = 21), adalimumab (n = 19), vedolizumab (n = 10), and ustekinumab (n = 7) in the third trimester.
The 57 biologic-exposed infants underwent standardized clinical assessments, drug concentration, and immune function testing. The live oral rotavirus vaccine series was provided to 50 infants, with the first dose at a median of 13 weeks of age. Immunologic assessments validated for age were normal in all infants despite median infliximab concentrations of 6.1 μg/mL (range, 0.4-28.8 μg/mL), adalimumab concentrations of 1.7 μg/mL (range, 0.7-7.9 μg/mL), ustekinumab concentrations of 0.6 μg/mL (range, 0-1.1), and undetectable for vedolizumab at 10.7 weeks of age.
As anticipated, infant immune function was normal regardless of circulating drug levels.
The overall message, said Seow, is “healthy mum equals healthy baby. Be more concerned regarding active inflammation than active medications. In almost all circumstances, treat to target in pregnancy as you would in the nonpregnant state.” She added, however, that further studies are needed to determine the safety and optimal timing of other live vaccines, such as the BCG, in the presence of biologic therapy.
This study was funded by the Alberta Children’s Hospital Research Institute. Seow reported advisory/speaker’s fees for Janssen, AbbVie, Takeda, Pfizer, Fresenius Kabi, Bristol-Myers Squibb, Pharmascience, and Lilly, as well as funding from Alberta Children’s Hospital Research Institute, Crohn’s and Colitis Canada, the Canadian Institutes of Health Research, and Calgary Health Trust, and data safety monitoring from New South Wales Government Health, Australia. Multiple coauthors disclosed similar consulting or speaker relationships with private industry. Spencer had no competing interests with regard to her comments.
A version of this article first appeared on Medscape.com.
No adverse events or impairment of the immune system emerged in babies at 7 days, 1 month, and 9 months post vaccination, in findings from a small Canadian study published in Clinical Gastroenterology and Hepatology.
The study found normal extended immune function testing in infants despite third-trimester maternal biologic therapy and regardless of circulating drug levels. The data provide reassurance about live rotavirus vaccination in this population and may also offer insights into the safety of other live vaccines in biologic-exposed individuals, wrote investigators led by gastroenterologist Cynthia H. Seow, MD, a professor in the Cumming School of Medicine at the University of Calgary in Alberta, Canada.
“Despite the well-established safety and effectiveness of non–live vaccination in individuals with IBD, including those on immunomodulators and biologic therapy, vaccine uptake in pregnant women with IBD and their infants remains suboptimal,” Seow said in an interview. This largely arises from maternal and physician concerns regarding transplacental transfer of IBD therapies and their impact on the safety of vaccination.
“These concerns were heightened after reports emerged of five fatal outcomes following the administration of the live Bacille Calmette-Guérin [BCG] vaccine in biologic-exposed infants. However, it had already been reported that inadvertent administration of the live oral rotavirus vaccine, a very different vaccine in terms of target and mechanism of action, in biologic-exposed individuals had not been associated with significant adverse effects,” she said.
They undertook their analysis with the hypothesis that vaccination would carry low risk, although the live oral vaccine is not currently recommended in biologic-exposed infants. “Yet rotavirus is a leading cause of severe, dehydrating diarrhea in children under the age of 5 years globally, and vaccination has led to significant reductions in hospitalizations and mortality,” Seow added.
Provision of the vaccine to anti–tumor necrosis factor (TNF)–exposed infants has been incorporated into the Canadian Public Health and Immunization guidelines, as the majority of the biologic-exposed infants were exposed to anti-TNF agents. “And with collection of further data, we expect that this will be extended to other biologic agent exposure. These data are important to pregnant women with IBD as they help to normalize their care. Pregnancy is difficult enough without having to remember exceptions to care,” Seow said.
“Before some of the studies came out, broad guidelines recommended that live vaccines should not be used in biologic-exposed infants, but this had been thought to be overly zealous and too conservative, and the risk was thought to be low,” said Elizabeth Spencer, MD, an assistant professor of pediatrics in the Division of Pediatric Gastroenterology at the Icahn School of Medicine at Mount Sinai in New York City, in an interview. Spencer was not involved in the Canadian study.
“At our center, we had some moms on biologics during pregnancy who forgot and had their babies vaccinated for rotavirus, and the babies were all fine,” she said.
The safety of this vaccine has been confirmed by several small studies and recently the PIANO Helmsley Global Consensus on Pregnancy and Inflammatory Bowel Disease, which was presented at Digestive Disease Week 2024. The consensus encompasses preconception counseling and the safety of IBD medications during pregnancy and lactation.
“Another concern, however, was that giving a live GI bug like rotavirus to babies might overstimulate their immune systems and provoke IBD,” Spencer added. “While a number of population-based studies in the US and Europe showed that was not the case, at least in the general population, there was a suggestion that, down the road, vaccination might be mildly protective against IBD in some cases.”
She added the caveat that these studies were not done in mothers and their babies with IBD, who might be inherently at greater risk for IBD. “So, a question for future research would be, ‘Is immune stimulation of the gut in IBD moms and their babies a good or a bad thing for their gut?’ ”
Spencer conceded that “the data present a bit of a blurry picture, but I think it’s always better just to vaccinate according to the regular schedule. The current data say there is no added risk, but it would be nice to look specifically at risk in moms with IBD and their children.”
The Study
The prospective cohort study is a substudy of a larger 2023 one that included biologic use in a range of maternal illnesses, not just IBD.
For the current study, Seow and colleagues identified 57 infants born to 52 mothers with IBD attending a pregnancy clinic at the University of Calgary in the period 2019-2023. Almost 81% of the mothers had Crohn’s disease, and the median duration of IBD was 10 years. The median gestational age at delivery was 39 weeks, and almost 60% of deliveries were vaginal. The infants had been exposed in utero to infliximab (n = 21), adalimumab (n = 19), vedolizumab (n = 10), and ustekinumab (n = 7) in the third trimester.
The 57 biologic-exposed infants underwent standardized clinical assessments, drug concentration, and immune function testing. The live oral rotavirus vaccine series was provided to 50 infants, with the first dose at a median of 13 weeks of age. Immunologic assessments validated for age were normal in all infants despite median infliximab concentrations of 6.1 μg/mL (range, 0.4-28.8 μg/mL), adalimumab concentrations of 1.7 μg/mL (range, 0.7-7.9 μg/mL), ustekinumab concentrations of 0.6 μg/mL (range, 0-1.1), and undetectable for vedolizumab at 10.7 weeks of age.
As anticipated, infant immune function was normal regardless of circulating drug levels.
The overall message, said Seow, is “healthy mum equals healthy baby. Be more concerned regarding active inflammation than active medications. In almost all circumstances, treat to target in pregnancy as you would in the nonpregnant state.” She added, however, that further studies are needed to determine the safety and optimal timing of other live vaccines, such as the BCG, in the presence of biologic therapy.
This study was funded by the Alberta Children’s Hospital Research Institute. Seow reported advisory/speaker’s fees for Janssen, AbbVie, Takeda, Pfizer, Fresenius Kabi, Bristol-Myers Squibb, Pharmascience, and Lilly, as well as funding from Alberta Children’s Hospital Research Institute, Crohn’s and Colitis Canada, the Canadian Institutes of Health Research, and Calgary Health Trust, and data safety monitoring from New South Wales Government Health, Australia. Multiple coauthors disclosed similar consulting or speaker relationships with private industry. Spencer had no competing interests with regard to her comments.
A version of this article first appeared on Medscape.com.
No adverse events or impairment of the immune system emerged in babies at 7 days, 1 month, and 9 months post vaccination, in findings from a small Canadian study published in Clinical Gastroenterology and Hepatology.
The study found normal extended immune function testing in infants despite third-trimester maternal biologic therapy and regardless of circulating drug levels. The data provide reassurance about live rotavirus vaccination in this population and may also offer insights into the safety of other live vaccines in biologic-exposed individuals, wrote investigators led by gastroenterologist Cynthia H. Seow, MD, a professor in the Cumming School of Medicine at the University of Calgary in Alberta, Canada.
“Despite the well-established safety and effectiveness of non–live vaccination in individuals with IBD, including those on immunomodulators and biologic therapy, vaccine uptake in pregnant women with IBD and their infants remains suboptimal,” Seow said in an interview. This largely arises from maternal and physician concerns regarding transplacental transfer of IBD therapies and their impact on the safety of vaccination.
“These concerns were heightened after reports emerged of five fatal outcomes following the administration of the live Bacille Calmette-Guérin [BCG] vaccine in biologic-exposed infants. However, it had already been reported that inadvertent administration of the live oral rotavirus vaccine, a very different vaccine in terms of target and mechanism of action, in biologic-exposed individuals had not been associated with significant adverse effects,” she said.
They undertook their analysis with the hypothesis that vaccination would carry low risk, although the live oral vaccine is not currently recommended in biologic-exposed infants. “Yet rotavirus is a leading cause of severe, dehydrating diarrhea in children under the age of 5 years globally, and vaccination has led to significant reductions in hospitalizations and mortality,” Seow added.
Provision of the vaccine to anti–tumor necrosis factor (TNF)–exposed infants has been incorporated into the Canadian Public Health and Immunization guidelines, as the majority of the biologic-exposed infants were exposed to anti-TNF agents. “And with collection of further data, we expect that this will be extended to other biologic agent exposure. These data are important to pregnant women with IBD as they help to normalize their care. Pregnancy is difficult enough without having to remember exceptions to care,” Seow said.
“Before some of the studies came out, broad guidelines recommended that live vaccines should not be used in biologic-exposed infants, but this had been thought to be overly zealous and too conservative, and the risk was thought to be low,” said Elizabeth Spencer, MD, an assistant professor of pediatrics in the Division of Pediatric Gastroenterology at the Icahn School of Medicine at Mount Sinai in New York City, in an interview. Spencer was not involved in the Canadian study.
“At our center, we had some moms on biologics during pregnancy who forgot and had their babies vaccinated for rotavirus, and the babies were all fine,” she said.
The safety of this vaccine has been confirmed by several small studies and recently the PIANO Helmsley Global Consensus on Pregnancy and Inflammatory Bowel Disease, which was presented at Digestive Disease Week 2024. The consensus encompasses preconception counseling and the safety of IBD medications during pregnancy and lactation.
“Another concern, however, was that giving a live GI bug like rotavirus to babies might overstimulate their immune systems and provoke IBD,” Spencer added. “While a number of population-based studies in the US and Europe showed that was not the case, at least in the general population, there was a suggestion that, down the road, vaccination might be mildly protective against IBD in some cases.”
She added the caveat that these studies were not done in mothers and their babies with IBD, who might be inherently at greater risk for IBD. “So, a question for future research would be, ‘Is immune stimulation of the gut in IBD moms and their babies a good or a bad thing for their gut?’ ”
Spencer conceded that “the data present a bit of a blurry picture, but I think it’s always better just to vaccinate according to the regular schedule. The current data say there is no added risk, but it would be nice to look specifically at risk in moms with IBD and their children.”
The Study
The prospective cohort study is a substudy of a larger 2023 one that included biologic use in a range of maternal illnesses, not just IBD.
For the current study, Seow and colleagues identified 57 infants born to 52 mothers with IBD attending a pregnancy clinic at the University of Calgary in the period 2019-2023. Almost 81% of the mothers had Crohn’s disease, and the median duration of IBD was 10 years. The median gestational age at delivery was 39 weeks, and almost 60% of deliveries were vaginal. The infants had been exposed in utero to infliximab (n = 21), adalimumab (n = 19), vedolizumab (n = 10), and ustekinumab (n = 7) in the third trimester.
The 57 biologic-exposed infants underwent standardized clinical assessments, drug concentration, and immune function testing. The live oral rotavirus vaccine series was provided to 50 infants, with the first dose at a median of 13 weeks of age. Immunologic assessments validated for age were normal in all infants despite median infliximab concentrations of 6.1 μg/mL (range, 0.4-28.8 μg/mL), adalimumab concentrations of 1.7 μg/mL (range, 0.7-7.9 μg/mL), ustekinumab concentrations of 0.6 μg/mL (range, 0-1.1), and undetectable for vedolizumab at 10.7 weeks of age.
As anticipated, infant immune function was normal regardless of circulating drug levels.
The overall message, said Seow, is “healthy mum equals healthy baby. Be more concerned regarding active inflammation than active medications. In almost all circumstances, treat to target in pregnancy as you would in the nonpregnant state.” She added, however, that further studies are needed to determine the safety and optimal timing of other live vaccines, such as the BCG, in the presence of biologic therapy.
This study was funded by the Alberta Children’s Hospital Research Institute. Seow reported advisory/speaker’s fees for Janssen, AbbVie, Takeda, Pfizer, Fresenius Kabi, Bristol-Myers Squibb, Pharmascience, and Lilly, as well as funding from Alberta Children’s Hospital Research Institute, Crohn’s and Colitis Canada, the Canadian Institutes of Health Research, and Calgary Health Trust, and data safety monitoring from New South Wales Government Health, Australia. Multiple coauthors disclosed similar consulting or speaker relationships with private industry. Spencer had no competing interests with regard to her comments.
A version of this article first appeared on Medscape.com.
FROM CLINICAL GASTROENTEROLOGY AND HEPATOLOGY