User login
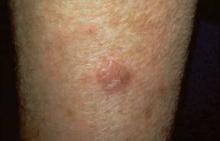
Bexarotene Confers No Survival Benefit for Mycosis Fungoides
SAN DIEGO – The median survival for patients with tumor stage mycosis fungoides who received oral bexarotene therapy was 3.3 years, compared with a median of 7.7 years for patients who did not receive the drug, results from a long-term, single-center study demonstrated.
The finding "was not intuitive, because approximately 54% of patients who took bexarotene responded to the drug, but it had a negative impact on survival," Dr. John A. Zic said in an interview following a poster session at the annual meeting of the American Academy of Dermatology, where the study was presented.
Mycosis fungoides accounts for the majority of cutaneous T-cell lymphoma cases, yet no randomized controlled trials exist to compare existing therapies head to head. "Therefore, it is important to gather long-term clinical data for retrospective analysis when these patient cohorts exist to analyze how different therapies contribute to patient outcome," Dr. Zic and his associates wrote in their poster abstract.
For the current study, the researchers reviewed data from 39 patients with tumor stage mycosis fungoides who were followed at the Vanderbilt University Cutaneous Lymphoma Clinic in Nashville, Tenn., between July of 1995 and July of 2010. They set out to determine if patients who received therapy with oral bexarotene for the treatment of tumor stage mycosis fungoides had improved outcome, compared with those who did not take the drug. Bexarotene is a synthetic retinoid approved by the Food and Drug Administration in 1999 for the treatment of refractory, advanced-stage cutaneous T-cell lymphoma, including mycosis fungoides.
Of the 39 patients 27 (69%) were male. More than half of patients (67%) received oral bexarotene while 33% did not. Patients in the bexarotene group were older than those who did not receive the drug (a mean of 61 vs. 56 years, respectively), and a higher proportion had late clinical stage disease at diagnosis (19 patients vs. 10 patinets). They were also more likely to have large cell transformation, "which is a negative prognostic indicator," said Dr. Zic, associate professor of dermatology at Vanderbilt University.
He went on to report that 54% of patients in the bexarotene group achieved durable response, which was defined as a greater than 50% clearing for at least 1 month. However, the median overall survival for patients in the bexarotene group was 3.3 years, compared with 7.7 years for patients who did not receive the drug.
The researchers also found that patients who were diagnosed with mycosis fungoides before the year 2000 "seemed to have a longer survival than patients who were diagnosed after 2000," Dr. Zic said. "You would think that with the introduction of newer therapies we might be able to impact survival in the past decade versus survival two decades ago. We didn’t find that, and we’re not sure why. However, the more recently enrolled patients appear to be sicker; they have higher stages of disease and more [large cell] transformation. That might explain the difference."
Dr. Zic acknowledged that a chief limitation of the study was its retrospective design. "There could be certain biases introduced such as selection bias and referral bias that might help to explain some of the differences that were seen," he said.
Further analyses of the patients are planned.
Dr. Zic said that he had no relevant financial disclosures.
SAN DIEGO – The median survival for patients with tumor stage mycosis fungoides who received oral bexarotene therapy was 3.3 years, compared with a median of 7.7 years for patients who did not receive the drug, results from a long-term, single-center study demonstrated.
The finding "was not intuitive, because approximately 54% of patients who took bexarotene responded to the drug, but it had a negative impact on survival," Dr. John A. Zic said in an interview following a poster session at the annual meeting of the American Academy of Dermatology, where the study was presented.
Mycosis fungoides accounts for the majority of cutaneous T-cell lymphoma cases, yet no randomized controlled trials exist to compare existing therapies head to head. "Therefore, it is important to gather long-term clinical data for retrospective analysis when these patient cohorts exist to analyze how different therapies contribute to patient outcome," Dr. Zic and his associates wrote in their poster abstract.
For the current study, the researchers reviewed data from 39 patients with tumor stage mycosis fungoides who were followed at the Vanderbilt University Cutaneous Lymphoma Clinic in Nashville, Tenn., between July of 1995 and July of 2010. They set out to determine if patients who received therapy with oral bexarotene for the treatment of tumor stage mycosis fungoides had improved outcome, compared with those who did not take the drug. Bexarotene is a synthetic retinoid approved by the Food and Drug Administration in 1999 for the treatment of refractory, advanced-stage cutaneous T-cell lymphoma, including mycosis fungoides.
Of the 39 patients 27 (69%) were male. More than half of patients (67%) received oral bexarotene while 33% did not. Patients in the bexarotene group were older than those who did not receive the drug (a mean of 61 vs. 56 years, respectively), and a higher proportion had late clinical stage disease at diagnosis (19 patients vs. 10 patinets). They were also more likely to have large cell transformation, "which is a negative prognostic indicator," said Dr. Zic, associate professor of dermatology at Vanderbilt University.
He went on to report that 54% of patients in the bexarotene group achieved durable response, which was defined as a greater than 50% clearing for at least 1 month. However, the median overall survival for patients in the bexarotene group was 3.3 years, compared with 7.7 years for patients who did not receive the drug.
The researchers also found that patients who were diagnosed with mycosis fungoides before the year 2000 "seemed to have a longer survival than patients who were diagnosed after 2000," Dr. Zic said. "You would think that with the introduction of newer therapies we might be able to impact survival in the past decade versus survival two decades ago. We didn’t find that, and we’re not sure why. However, the more recently enrolled patients appear to be sicker; they have higher stages of disease and more [large cell] transformation. That might explain the difference."
Dr. Zic acknowledged that a chief limitation of the study was its retrospective design. "There could be certain biases introduced such as selection bias and referral bias that might help to explain some of the differences that were seen," he said.
Further analyses of the patients are planned.
Dr. Zic said that he had no relevant financial disclosures.
SAN DIEGO – The median survival for patients with tumor stage mycosis fungoides who received oral bexarotene therapy was 3.3 years, compared with a median of 7.7 years for patients who did not receive the drug, results from a long-term, single-center study demonstrated.
The finding "was not intuitive, because approximately 54% of patients who took bexarotene responded to the drug, but it had a negative impact on survival," Dr. John A. Zic said in an interview following a poster session at the annual meeting of the American Academy of Dermatology, where the study was presented.
Mycosis fungoides accounts for the majority of cutaneous T-cell lymphoma cases, yet no randomized controlled trials exist to compare existing therapies head to head. "Therefore, it is important to gather long-term clinical data for retrospective analysis when these patient cohorts exist to analyze how different therapies contribute to patient outcome," Dr. Zic and his associates wrote in their poster abstract.
For the current study, the researchers reviewed data from 39 patients with tumor stage mycosis fungoides who were followed at the Vanderbilt University Cutaneous Lymphoma Clinic in Nashville, Tenn., between July of 1995 and July of 2010. They set out to determine if patients who received therapy with oral bexarotene for the treatment of tumor stage mycosis fungoides had improved outcome, compared with those who did not take the drug. Bexarotene is a synthetic retinoid approved by the Food and Drug Administration in 1999 for the treatment of refractory, advanced-stage cutaneous T-cell lymphoma, including mycosis fungoides.
Of the 39 patients 27 (69%) were male. More than half of patients (67%) received oral bexarotene while 33% did not. Patients in the bexarotene group were older than those who did not receive the drug (a mean of 61 vs. 56 years, respectively), and a higher proportion had late clinical stage disease at diagnosis (19 patients vs. 10 patinets). They were also more likely to have large cell transformation, "which is a negative prognostic indicator," said Dr. Zic, associate professor of dermatology at Vanderbilt University.
He went on to report that 54% of patients in the bexarotene group achieved durable response, which was defined as a greater than 50% clearing for at least 1 month. However, the median overall survival for patients in the bexarotene group was 3.3 years, compared with 7.7 years for patients who did not receive the drug.
The researchers also found that patients who were diagnosed with mycosis fungoides before the year 2000 "seemed to have a longer survival than patients who were diagnosed after 2000," Dr. Zic said. "You would think that with the introduction of newer therapies we might be able to impact survival in the past decade versus survival two decades ago. We didn’t find that, and we’re not sure why. However, the more recently enrolled patients appear to be sicker; they have higher stages of disease and more [large cell] transformation. That might explain the difference."
Dr. Zic acknowledged that a chief limitation of the study was its retrospective design. "There could be certain biases introduced such as selection bias and referral bias that might help to explain some of the differences that were seen," he said.
Further analyses of the patients are planned.
Dr. Zic said that he had no relevant financial disclosures.
FROM THE ANNUAL MEETING OF THE AMERICAN ACADEMY OF DERMATOLOGY
Major Finding: The median survival time for patients with tumor stage mycosis fungoides who received oral bexarotene therapy was 3.3 years, compared with a median of 7.7 years for patients who did not receive the drug.
Data Source: A retrospective analysis of 39 patients with tumor stage mycosis fungoides who were followed at the Vanderbilt University Cutaneous Lymphoma Clinic between July of 1995 and July of 2010.
Disclosures: Dr. Zic said that he had no relevant financial conflicts to disclose.
Study Results Support Ingenol Mebutate's AK Effectiveness
Topical ingenol mebutate gel effectively treated actinic keratoses when applied to the face, scalp, trunk, or extremities for 2-3 days, according to a report in the March 15 issue of the New England Journal of Medicine.
Compared with existing therapies for actinic keratoses, the chief advantage of ingenol mebutate gel is the short exposure time, reported Dr. Mark Lebwohl, professor and chairman of the department of dermatology at Mount Sinai School of Medicine, New York, and his associates. This allows for relatively rapid resolution of local reactions, and it will likely improve adherence to treatment, which in turn should improve the therapy’s effectiveness, they noted.
"Many patients find it difficult to adhere to the currently available regimens of topical treatment that last for periods of 1-4 months, which may result in ‘real-world’ effectiveness lower than that achieved in supervised and patient-compensated clinical trials," Dr. Lebwohl and his colleagues wrote.
The researchers evaluated the safety and effectiveness of ingenol mebutate gel compared with a placebo in a manufacturer-sponsored, randomized, double-blind trial of 1,005 patients treated at four medical centers. The majority of the patients had Fitzpatrick type I or II skin, and their mean age was 65 years.
Approximately half of the study patients had a history of skin cancer, and 75% had already had their keratosis treated with cryotherapy, imiquimod, or topical fluorouracil.
In each patient, a 25-cm contiguous field containing at least four to eight clinically typical and discrete actinic keratoses was selected for treatment. The study patients were divided into two groups according to the location of the treated area: on the head (face or scalp), and on the body (trunk or extremities).
Those with facial or scalp lesions were randomly assigned to self-apply either 0.015% active gel (277 patients) or placebo gel (270 patients) to the area once daily for 3 consecutive days. Those with trunk or extremity lesions were randomly assigned to self-apply 0.05% active gel (226 patients) or placebo gel (232 patients) to the area once daily for 2 consecutive days.
The primary end point was complete clearance of all clinically visible actinic keratoses in the treatment area on day 57.
Among patients with facial or scalp lesions, 42.2% of those who used active gel reached this end point, compared with only 3.7% of those who used placebo gel. Among patients with trunk or extremity lesions, 34.1% who used active gel reached this end point, compared with only 4.7% of those who used placebo gel.
In addition, patients with face or scalp lesions that were treated with ingenol mebutate showed a median reduction of 83% in the number of actinic keratoses, compared with a 0% reduction with placebo gel. "We calculated that the number of patients who needed to be treated with ingenol mebutate to obtain complete clearance in one patient was 2.6," the investigators reported (N. Engl. J. Med. 2012;366:1010-19).
Patients with trunk or extremity lesions treated with ingenol mebutate showed a median reduction of 75% in the number of actinic keratoses, compared with a 0% reduction with placebo gel. "We calculated that the number of patients who would need to be treated with ingenol mebutate to obtain complete clearance in one patient was 3.4," the researchers noted.
A secondary end point was partial (75% or more) clearance in the number of clinically visible actinic keratoses in the treatment area on day 57.
Among patients with facial or scalp lesions, 63.9% of those who used active gel reached this end point, compared with only 7.4% of those who used placebo gel. Among patients with trunk or extremity lesions, 49.1% of those who used active gel reached this end point, compared with only 6.9% of those who used placebo gel.
Patients who used ingenol mebutate showed minimal scarring or change in pigmentation. Local reactions such as erythema, crusting, swelling, vesiculation, or pustulation were common, but were mild to moderate in intensity; they peaked within a few days of treatment and resolved rapidly afterward, with no sequelae. One patient developed eye pain, burning, and periorbital edema related to ingenol mebutate and dropped out of the study, Dr. Lebwohl and his associates noted.
"Future studies are needed to assess the risks and benefits of treating larger areas of skin, using multiple treatments in the same area, and using combination therapies," they added.
This study was funded by LEO Pharma, maker of ingenol mebutate. Dr. Lebwohl reported ties to LEO Pharma, Graceway, PharmaDerm, and Peplin LTD.
Topical ingenol mebutate gel effectively treated actinic keratoses when applied to the face, scalp, trunk, or extremities for 2-3 days, according to a report in the March 15 issue of the New England Journal of Medicine.
Compared with existing therapies for actinic keratoses, the chief advantage of ingenol mebutate gel is the short exposure time, reported Dr. Mark Lebwohl, professor and chairman of the department of dermatology at Mount Sinai School of Medicine, New York, and his associates. This allows for relatively rapid resolution of local reactions, and it will likely improve adherence to treatment, which in turn should improve the therapy’s effectiveness, they noted.
"Many patients find it difficult to adhere to the currently available regimens of topical treatment that last for periods of 1-4 months, which may result in ‘real-world’ effectiveness lower than that achieved in supervised and patient-compensated clinical trials," Dr. Lebwohl and his colleagues wrote.
The researchers evaluated the safety and effectiveness of ingenol mebutate gel compared with a placebo in a manufacturer-sponsored, randomized, double-blind trial of 1,005 patients treated at four medical centers. The majority of the patients had Fitzpatrick type I or II skin, and their mean age was 65 years.
Approximately half of the study patients had a history of skin cancer, and 75% had already had their keratosis treated with cryotherapy, imiquimod, or topical fluorouracil.
In each patient, a 25-cm contiguous field containing at least four to eight clinically typical and discrete actinic keratoses was selected for treatment. The study patients were divided into two groups according to the location of the treated area: on the head (face or scalp), and on the body (trunk or extremities).
Those with facial or scalp lesions were randomly assigned to self-apply either 0.015% active gel (277 patients) or placebo gel (270 patients) to the area once daily for 3 consecutive days. Those with trunk or extremity lesions were randomly assigned to self-apply 0.05% active gel (226 patients) or placebo gel (232 patients) to the area once daily for 2 consecutive days.
The primary end point was complete clearance of all clinically visible actinic keratoses in the treatment area on day 57.
Among patients with facial or scalp lesions, 42.2% of those who used active gel reached this end point, compared with only 3.7% of those who used placebo gel. Among patients with trunk or extremity lesions, 34.1% who used active gel reached this end point, compared with only 4.7% of those who used placebo gel.
In addition, patients with face or scalp lesions that were treated with ingenol mebutate showed a median reduction of 83% in the number of actinic keratoses, compared with a 0% reduction with placebo gel. "We calculated that the number of patients who needed to be treated with ingenol mebutate to obtain complete clearance in one patient was 2.6," the investigators reported (N. Engl. J. Med. 2012;366:1010-19).
Patients with trunk or extremity lesions treated with ingenol mebutate showed a median reduction of 75% in the number of actinic keratoses, compared with a 0% reduction with placebo gel. "We calculated that the number of patients who would need to be treated with ingenol mebutate to obtain complete clearance in one patient was 3.4," the researchers noted.
A secondary end point was partial (75% or more) clearance in the number of clinically visible actinic keratoses in the treatment area on day 57.
Among patients with facial or scalp lesions, 63.9% of those who used active gel reached this end point, compared with only 7.4% of those who used placebo gel. Among patients with trunk or extremity lesions, 49.1% of those who used active gel reached this end point, compared with only 6.9% of those who used placebo gel.
Patients who used ingenol mebutate showed minimal scarring or change in pigmentation. Local reactions such as erythema, crusting, swelling, vesiculation, or pustulation were common, but were mild to moderate in intensity; they peaked within a few days of treatment and resolved rapidly afterward, with no sequelae. One patient developed eye pain, burning, and periorbital edema related to ingenol mebutate and dropped out of the study, Dr. Lebwohl and his associates noted.
"Future studies are needed to assess the risks and benefits of treating larger areas of skin, using multiple treatments in the same area, and using combination therapies," they added.
This study was funded by LEO Pharma, maker of ingenol mebutate. Dr. Lebwohl reported ties to LEO Pharma, Graceway, PharmaDerm, and Peplin LTD.
Topical ingenol mebutate gel effectively treated actinic keratoses when applied to the face, scalp, trunk, or extremities for 2-3 days, according to a report in the March 15 issue of the New England Journal of Medicine.
Compared with existing therapies for actinic keratoses, the chief advantage of ingenol mebutate gel is the short exposure time, reported Dr. Mark Lebwohl, professor and chairman of the department of dermatology at Mount Sinai School of Medicine, New York, and his associates. This allows for relatively rapid resolution of local reactions, and it will likely improve adherence to treatment, which in turn should improve the therapy’s effectiveness, they noted.
"Many patients find it difficult to adhere to the currently available regimens of topical treatment that last for periods of 1-4 months, which may result in ‘real-world’ effectiveness lower than that achieved in supervised and patient-compensated clinical trials," Dr. Lebwohl and his colleagues wrote.
The researchers evaluated the safety and effectiveness of ingenol mebutate gel compared with a placebo in a manufacturer-sponsored, randomized, double-blind trial of 1,005 patients treated at four medical centers. The majority of the patients had Fitzpatrick type I or II skin, and their mean age was 65 years.
Approximately half of the study patients had a history of skin cancer, and 75% had already had their keratosis treated with cryotherapy, imiquimod, or topical fluorouracil.
In each patient, a 25-cm contiguous field containing at least four to eight clinically typical and discrete actinic keratoses was selected for treatment. The study patients were divided into two groups according to the location of the treated area: on the head (face or scalp), and on the body (trunk or extremities).
Those with facial or scalp lesions were randomly assigned to self-apply either 0.015% active gel (277 patients) or placebo gel (270 patients) to the area once daily for 3 consecutive days. Those with trunk or extremity lesions were randomly assigned to self-apply 0.05% active gel (226 patients) or placebo gel (232 patients) to the area once daily for 2 consecutive days.
The primary end point was complete clearance of all clinically visible actinic keratoses in the treatment area on day 57.
Among patients with facial or scalp lesions, 42.2% of those who used active gel reached this end point, compared with only 3.7% of those who used placebo gel. Among patients with trunk or extremity lesions, 34.1% who used active gel reached this end point, compared with only 4.7% of those who used placebo gel.
In addition, patients with face or scalp lesions that were treated with ingenol mebutate showed a median reduction of 83% in the number of actinic keratoses, compared with a 0% reduction with placebo gel. "We calculated that the number of patients who needed to be treated with ingenol mebutate to obtain complete clearance in one patient was 2.6," the investigators reported (N. Engl. J. Med. 2012;366:1010-19).
Patients with trunk or extremity lesions treated with ingenol mebutate showed a median reduction of 75% in the number of actinic keratoses, compared with a 0% reduction with placebo gel. "We calculated that the number of patients who would need to be treated with ingenol mebutate to obtain complete clearance in one patient was 3.4," the researchers noted.
A secondary end point was partial (75% or more) clearance in the number of clinically visible actinic keratoses in the treatment area on day 57.
Among patients with facial or scalp lesions, 63.9% of those who used active gel reached this end point, compared with only 7.4% of those who used placebo gel. Among patients with trunk or extremity lesions, 49.1% of those who used active gel reached this end point, compared with only 6.9% of those who used placebo gel.
Patients who used ingenol mebutate showed minimal scarring or change in pigmentation. Local reactions such as erythema, crusting, swelling, vesiculation, or pustulation were common, but were mild to moderate in intensity; they peaked within a few days of treatment and resolved rapidly afterward, with no sequelae. One patient developed eye pain, burning, and periorbital edema related to ingenol mebutate and dropped out of the study, Dr. Lebwohl and his associates noted.
"Future studies are needed to assess the risks and benefits of treating larger areas of skin, using multiple treatments in the same area, and using combination therapies," they added.
This study was funded by LEO Pharma, maker of ingenol mebutate. Dr. Lebwohl reported ties to LEO Pharma, Graceway, PharmaDerm, and Peplin LTD.
FROM THE NEW ENGLAND JOURNAL OF MEDICINE
Major Finding: In patients with facial or scalp lesions, 42.2% who used ingenol mebutate gel achieved complete clearance at 2 months, compared with only 3.7% of those who used placebo gel; in patients with trunk or extremity lesions, 34.1% who used active gel achieved complete clearance, compared with only 4.7% of those who used placebo gel.
Data Source: Data are from a manufacturer-sponsored, randomized, double-blind trial of 1,005 patients treated at four medical centers.
Disclosures: This study was funded by LEO Pharma, maker of ingenol mebutate. Dr. Lebwohl reported ties to LEO Pharma, Graceway, PharmaDerm, and Peplin LTD.
Sunburns Still Common Despite Protective Efforts
WASHINGTON – While the use of sun protection measures became more common between 2000 and 2010, there was not a corresponding decrease in sunburns, according to an analysis of national data.
Overall among women, staying in the shade, using sunscreen, and wearing clothing to the ankles increased significantly over time by 5%, 6%, and 5%, respectively, between 2000 and 2010. Similarly, among men, staying in the shade, sunscreen use, and wearing clothing to the ankles increased by 7%, 2%, and 5%, respectively. However, the overall prevalence of sunburn did not change significantly over those years. In 2010, 51% of women and 49% of men reported having at least one sunburn in the past year. The results come from a poster presented at the annual meeting of the American Society of Preventive Oncology.
The rates of melanoma and nonmelanoma skin cancers have increased in recent years. Monitoring and reporting sun-protective behaviors and sunburns over time are important ways to measure the impact of skin cancer prevention activity and to track progress toward Healthy People 2020 objectives, noted lead author Dawn M. Holman and her coinvestigators at the Centers for Disease Control and Prevention.
To estimate how commonly people engage in these behaviors that protect against sun exposure, the researchers used data from the National Health Interview Survey – Cancer Control Supplement, for years 2000, 2003, 2005, 2008, and 2010. The survey assessed the following behaviors as related to sun protection and sunburn: use of sunscreen, staying in the shade, wearing a wide-brimmed hat, wearing a long-sleeved shirt, and wearing long clothing to the ankles. Respondents reported their use as being: always, most of the time, sometimes, rarely, or never for each item. Respondents were also asked about the number of sunburns they’ve had in the last year.
Data were weighted to produce nationally representative estimates. Analyses were limited to those aged 18-29 years – age-adjusted to the 2000 U.S. population. The researchers estimated the percentage who reported engaging in each behavior always or most of the time and the percentage reporting one or more sunburns in the past year overall, by gender and by race/ethnicity.
Among women, using sunscreen (37%) and staying in the shade (35%) were the most common reported protective behaviors in 2010. Wearing a long-sleeved shirt (5%) and wearing a wide-brimmed hat (4%) were the least common. Black women were significantly less likely to report sunscreen use than were other racial/ethnic groups.
Among men, wearing long clothing to the ankles (33%) and staying in the shade (26%) were the most commonly reported behaviors in 2010. Fewer men reported using sunscreen (16%), wearing a long-sleeved shirt (8%), and wearing a wide-brimmed hat (7%). Of note, sunburn was significantly more common among non-Hispanic whites, compared with other racial/ethnic groups.
The results point to the "need for continued public health efforts to facilitate sun protection by: creating environments that support protective behaviors and by changing social norms regarding tanning and tanned skin. Facilitating sun protection may prevent sunburns and future increases in the burden of skin cancer," the researchers wrote.
The authors did not report whether they had any relevant financial interests.
WASHINGTON – While the use of sun protection measures became more common between 2000 and 2010, there was not a corresponding decrease in sunburns, according to an analysis of national data.
Overall among women, staying in the shade, using sunscreen, and wearing clothing to the ankles increased significantly over time by 5%, 6%, and 5%, respectively, between 2000 and 2010. Similarly, among men, staying in the shade, sunscreen use, and wearing clothing to the ankles increased by 7%, 2%, and 5%, respectively. However, the overall prevalence of sunburn did not change significantly over those years. In 2010, 51% of women and 49% of men reported having at least one sunburn in the past year. The results come from a poster presented at the annual meeting of the American Society of Preventive Oncology.
The rates of melanoma and nonmelanoma skin cancers have increased in recent years. Monitoring and reporting sun-protective behaviors and sunburns over time are important ways to measure the impact of skin cancer prevention activity and to track progress toward Healthy People 2020 objectives, noted lead author Dawn M. Holman and her coinvestigators at the Centers for Disease Control and Prevention.
To estimate how commonly people engage in these behaviors that protect against sun exposure, the researchers used data from the National Health Interview Survey – Cancer Control Supplement, for years 2000, 2003, 2005, 2008, and 2010. The survey assessed the following behaviors as related to sun protection and sunburn: use of sunscreen, staying in the shade, wearing a wide-brimmed hat, wearing a long-sleeved shirt, and wearing long clothing to the ankles. Respondents reported their use as being: always, most of the time, sometimes, rarely, or never for each item. Respondents were also asked about the number of sunburns they’ve had in the last year.
Data were weighted to produce nationally representative estimates. Analyses were limited to those aged 18-29 years – age-adjusted to the 2000 U.S. population. The researchers estimated the percentage who reported engaging in each behavior always or most of the time and the percentage reporting one or more sunburns in the past year overall, by gender and by race/ethnicity.
Among women, using sunscreen (37%) and staying in the shade (35%) were the most common reported protective behaviors in 2010. Wearing a long-sleeved shirt (5%) and wearing a wide-brimmed hat (4%) were the least common. Black women were significantly less likely to report sunscreen use than were other racial/ethnic groups.
Among men, wearing long clothing to the ankles (33%) and staying in the shade (26%) were the most commonly reported behaviors in 2010. Fewer men reported using sunscreen (16%), wearing a long-sleeved shirt (8%), and wearing a wide-brimmed hat (7%). Of note, sunburn was significantly more common among non-Hispanic whites, compared with other racial/ethnic groups.
The results point to the "need for continued public health efforts to facilitate sun protection by: creating environments that support protective behaviors and by changing social norms regarding tanning and tanned skin. Facilitating sun protection may prevent sunburns and future increases in the burden of skin cancer," the researchers wrote.
The authors did not report whether they had any relevant financial interests.
WASHINGTON – While the use of sun protection measures became more common between 2000 and 2010, there was not a corresponding decrease in sunburns, according to an analysis of national data.
Overall among women, staying in the shade, using sunscreen, and wearing clothing to the ankles increased significantly over time by 5%, 6%, and 5%, respectively, between 2000 and 2010. Similarly, among men, staying in the shade, sunscreen use, and wearing clothing to the ankles increased by 7%, 2%, and 5%, respectively. However, the overall prevalence of sunburn did not change significantly over those years. In 2010, 51% of women and 49% of men reported having at least one sunburn in the past year. The results come from a poster presented at the annual meeting of the American Society of Preventive Oncology.
The rates of melanoma and nonmelanoma skin cancers have increased in recent years. Monitoring and reporting sun-protective behaviors and sunburns over time are important ways to measure the impact of skin cancer prevention activity and to track progress toward Healthy People 2020 objectives, noted lead author Dawn M. Holman and her coinvestigators at the Centers for Disease Control and Prevention.
To estimate how commonly people engage in these behaviors that protect against sun exposure, the researchers used data from the National Health Interview Survey – Cancer Control Supplement, for years 2000, 2003, 2005, 2008, and 2010. The survey assessed the following behaviors as related to sun protection and sunburn: use of sunscreen, staying in the shade, wearing a wide-brimmed hat, wearing a long-sleeved shirt, and wearing long clothing to the ankles. Respondents reported their use as being: always, most of the time, sometimes, rarely, or never for each item. Respondents were also asked about the number of sunburns they’ve had in the last year.
Data were weighted to produce nationally representative estimates. Analyses were limited to those aged 18-29 years – age-adjusted to the 2000 U.S. population. The researchers estimated the percentage who reported engaging in each behavior always or most of the time and the percentage reporting one or more sunburns in the past year overall, by gender and by race/ethnicity.
Among women, using sunscreen (37%) and staying in the shade (35%) were the most common reported protective behaviors in 2010. Wearing a long-sleeved shirt (5%) and wearing a wide-brimmed hat (4%) were the least common. Black women were significantly less likely to report sunscreen use than were other racial/ethnic groups.
Among men, wearing long clothing to the ankles (33%) and staying in the shade (26%) were the most commonly reported behaviors in 2010. Fewer men reported using sunscreen (16%), wearing a long-sleeved shirt (8%), and wearing a wide-brimmed hat (7%). Of note, sunburn was significantly more common among non-Hispanic whites, compared with other racial/ethnic groups.
The results point to the "need for continued public health efforts to facilitate sun protection by: creating environments that support protective behaviors and by changing social norms regarding tanning and tanned skin. Facilitating sun protection may prevent sunburns and future increases in the burden of skin cancer," the researchers wrote.
The authors did not report whether they had any relevant financial interests.
FROM THE ANNUAL MEETING OF THE AMERICAN SOCIETY OF PREVENTIVE ONCOLOGY
Major Findings: In 2010, 51% of women and 49% of men reported having at least one sunburn in the past year. Sunburn was significantly more common among non-Hispanic whites, compared with other racial/ethnic groups.
Data Source: Researchers used data from the National Health Interview Survey – Cancer Control Supplement for years 2000, 2003, 2005, 2008, and 2010.
Disclosures: The authors did not report whether they had any relevant financial interests.
Tanning Regulations Linger at FDA
Rep. Rosa DeLauro (D-Conn.) sent a letter on March 9 to FDA Commissioner Margaret Hamburg urging her to act on recommendations - made by the General and Plastic Surgery Devices Panel on March 25, 2010 - to enforce stricter tanning bed regulations.
Rep. DeLauro noted that it will soon be exactly 2 years since the panel met and asked whether there will be any action before the end of May.
She also included a letter from two melanoma experts: Dr. David Fisher, director of the Melanoma Program at Massachusetts General Hospital and Mr. Alan Geller of the Harvard School of Public Health.
In the letter, the two experts contend that during the 2 years since the FDA panel meeting, tanning bed use has led to more than 5,000 cases of new melanomas, and an estimated 750 unnecessary deaths. They also asked, "How can the established skin cancer risk from tanning beds be continuously permitted, in the face of so much scientific and clinical evidence?"
Rep. DeLauro, who is a cancer survivor, said, "I simply cannot accept this inaction." She added," When will we have honest and accurate regulations and labels in place to protect Americans and end these unnecessary deaths caused by an inappropriately-regulated device?"
Her letter comes about a month after Democrats on the House Energy and Commerce Committee released results of an investigation that showed tanning salons were misleading users about the potential health risks.
The American Academy of Dermatology supports tougher regulations of indoor tanning and has urged Congress to pass H.R. 1676, the Tanning Bed Cancer Control Act. The bill had only 12 cosponsors at press time and had not been the subject of any hearings.
Rep. Rosa DeLauro (D-Conn.) sent a letter on March 9 to FDA Commissioner Margaret Hamburg urging her to act on recommendations - made by the General and Plastic Surgery Devices Panel on March 25, 2010 - to enforce stricter tanning bed regulations.
Rep. DeLauro noted that it will soon be exactly 2 years since the panel met and asked whether there will be any action before the end of May.
She also included a letter from two melanoma experts: Dr. David Fisher, director of the Melanoma Program at Massachusetts General Hospital and Mr. Alan Geller of the Harvard School of Public Health.
In the letter, the two experts contend that during the 2 years since the FDA panel meeting, tanning bed use has led to more than 5,000 cases of new melanomas, and an estimated 750 unnecessary deaths. They also asked, "How can the established skin cancer risk from tanning beds be continuously permitted, in the face of so much scientific and clinical evidence?"
Rep. DeLauro, who is a cancer survivor, said, "I simply cannot accept this inaction." She added," When will we have honest and accurate regulations and labels in place to protect Americans and end these unnecessary deaths caused by an inappropriately-regulated device?"
Her letter comes about a month after Democrats on the House Energy and Commerce Committee released results of an investigation that showed tanning salons were misleading users about the potential health risks.
The American Academy of Dermatology supports tougher regulations of indoor tanning and has urged Congress to pass H.R. 1676, the Tanning Bed Cancer Control Act. The bill had only 12 cosponsors at press time and had not been the subject of any hearings.
Rep. Rosa DeLauro (D-Conn.) sent a letter on March 9 to FDA Commissioner Margaret Hamburg urging her to act on recommendations - made by the General and Plastic Surgery Devices Panel on March 25, 2010 - to enforce stricter tanning bed regulations.
Rep. DeLauro noted that it will soon be exactly 2 years since the panel met and asked whether there will be any action before the end of May.
She also included a letter from two melanoma experts: Dr. David Fisher, director of the Melanoma Program at Massachusetts General Hospital and Mr. Alan Geller of the Harvard School of Public Health.
In the letter, the two experts contend that during the 2 years since the FDA panel meeting, tanning bed use has led to more than 5,000 cases of new melanomas, and an estimated 750 unnecessary deaths. They also asked, "How can the established skin cancer risk from tanning beds be continuously permitted, in the face of so much scientific and clinical evidence?"
Rep. DeLauro, who is a cancer survivor, said, "I simply cannot accept this inaction." She added," When will we have honest and accurate regulations and labels in place to protect Americans and end these unnecessary deaths caused by an inappropriately-regulated device?"
Her letter comes about a month after Democrats on the House Energy and Commerce Committee released results of an investigation that showed tanning salons were misleading users about the potential health risks.
The American Academy of Dermatology supports tougher regulations of indoor tanning and has urged Congress to pass H.R. 1676, the Tanning Bed Cancer Control Act. The bill had only 12 cosponsors at press time and had not been the subject of any hearings.
Numbing Agents Cause More Pain in PDT
WAIKOLOA, HAWAII – Using EMLA or lidocaine cream in conjunction with topical 5-aminolevulinic acid photodynamic therapy may boost uptake of the photosensitizer, but the clinical outcomes can be unpredictable, according to Dr. E. Victor Ross.
"Our worst scenarios have been when we’ve used EMLA on the skin. We’ve stopped using EMLA or lidocaine cream on the skin in ALA-PDT because of the very pronounced, enhanced photodynamic effects. You get great long-term results, but such a bad short-term result that lots of people didn’t want to go through it," said Dr. Ross of Scripps Clinic Laser and Cosmetic Dermatology Center in Carmel Valley, Calif.
Using a topical numbing agent seemed to be an attractive option because patients often complain that photodynamic therapy (PDT) is painful. Plus, there seemed to be an added benefit: The anesthetic cream increased aminolevulanic acid (ALA) uptake by the skin, such that ALA incubation times before application of the light source could be greatly compressed. But clinical outcomes were too unpredictable, he said at the seminar sponsored by Skin Disease Education Foundation (SDEF). After several patients had unintended florid full photopeels involving 1½ weeks of downtime, it was time to abandon the practice.
When ALA-PDT first appeared about 10 years ago, the indication was for the treatment of actinic keratoses (AKs). Today this approved indication remains the No. 1 reason Dr. Ross utilizes the therapy, he said. But PDT’s role has expanded off label to include cosmetic procedures and the treatment of warts, nonmelanoma skin cancer, nevus sebaceous, as well as acne, which he considers "one of the great opportunities for PDT." In addition, in Asia, dermatologists are now refining the use of PDT with hematoporphyrin derivatives for the treatment of port wine stains and other vascular lesions.
Many U.S. dermatologists have incorporated PDT into their practices, with ALA being more widely used as a photosensitizer than methyl aminolevulinate (MAL). Yet PDT is a therapy that hasn’t been fully optimized; it is still fraught with side effects and suboptimal results, he said. Moreover, some fundamental issues regarding PDT remain unanswered: For example, the optimal duration of photosensitizer incubation time for various indications is still controversial.
Dr. Ross said he opts for what he considers a middle-of-the-road approach, with ALA application times of about 90-120 minutes, which he views as having an optimal balance between side effects and effectiveness. Even so, he noted that among the 8-10 patients per week he treats with ALA-PDT on average, 1 or 2 experience mild side effects.
"I don’t think we’re quite ‘there’ yet with PDT, although we’re getting closer. There are still so many tricks involved in making it work without side effects. We’ve still got some work to do," he said.
In addition to advising his colleagues to stay away from topical anesthetic creams, he offered additional tips for the use of ALA-PDT. Among them:
• Sending acne into long-term remission via PDT remains the Holy Grail, he said. The objective is to enhance the fluorescence of protoporphyrin 9 at the sebaceous gland while sparing the epidermis.
Some investigators are using low-intensity blue light at the skin surface while the photosensitizer is incubating in order to bleach it out of the epidermis, or, alternatively, warming and cooling the skin.
The best results Dr. Ross said he has seen have come through an arduous regimen involving three 3-hour-long ALA applications scheduled a month apart. There’s a delayed effect, with significant improvement coming at 3-6 months.
"It’s a tough, tough therapy to get through. There are lots of pustules and papules, and the acne invariably gets worse before it gets better. This doesn’t play into the hands of the typical teenager, who wants to get better right away," Dr. Ross said.
• A creamy solution of ALA will create more protoporphyrin 9 than an aqueous solution will.
• A red light source should be considered for deeper structures, such as basal cell carcinomas or sebaceous glands, and blue light for treating more superficial skin lesions. Although continuous blue light is 40 times more potent per photon than red light in exciting protoporphyrin 9, it doesn’t penetrate as deeply.
• Performing low-density fractional CO2 ablative laser therapy prior to application of the photosensitizing agent is "an exciting advance" in PDT, he said.
The innovation was developed by an international team led by investigators at the Wellman Center for Photomedicine at Massachusetts General Hospital, Boston, who published a split-face randomized study involving 15 patients with a total of 212 facial AKs. At the 3-month follow-up, the complete response rate of grade II-III lesions treated with MAL-PDT preceded by fractional ablative laser therapy was 87.5%, vs. 58.8% with conventional MAL-PDT. The complete response rate of grade I AKs was 100% with fractional laser/MAL-PDT, vs. 79% for MAL-PDT alone.
The fractional laser–pretreated areas also displayed significantly greater improvement in photoaging and fewer new AKs at follow-up: 3, compared with 11. But these superior outcomes came at a price: higher pain scores during illumination and significantly worse erythema and crusting post treatment (Br. J. Dermatol. 2012 Feb. 20 [doi:10.1111/j.1365-2133.2012.10893.x]).
Fractional CO2 laser pretreatment enhances conversion of the photosensitizing agent to protoporphyrin 9, Dr. Ross explained. He compared the tiny holes in the skin created by the fractional laser to the process of aerating a lawn. The holes create conduits for the photosensitizer to bypass the stratum corneum, which is the major obstacle to uptake of ALA or MAL. Once the photosensitizer skips past the stratum corneum, it quickly spreads laterally throughout the epidermis.
However, Dr. Ross offered a note of caution regarding this novel approach. He said that he performed the therapy recently and found that 30 minutes of ALA incubation was too much.
"If you do these procedures, I would say go very light and just leave the ALA for less than 30 minutes to start, because the response you’re going to get when using a fractional laser beforehand is profound. It’s a huge difference," he said.
Dr. Ross reported that he serves as a consultant to and receives research support from Palomar. He also disclosed receiving research support from Candela, Cutera, Lumenis, Sciton, and Ulthera.
SDEF and this news organization are owned by Elsevier.
WAIKOLOA, HAWAII – Using EMLA or lidocaine cream in conjunction with topical 5-aminolevulinic acid photodynamic therapy may boost uptake of the photosensitizer, but the clinical outcomes can be unpredictable, according to Dr. E. Victor Ross.
"Our worst scenarios have been when we’ve used EMLA on the skin. We’ve stopped using EMLA or lidocaine cream on the skin in ALA-PDT because of the very pronounced, enhanced photodynamic effects. You get great long-term results, but such a bad short-term result that lots of people didn’t want to go through it," said Dr. Ross of Scripps Clinic Laser and Cosmetic Dermatology Center in Carmel Valley, Calif.
Using a topical numbing agent seemed to be an attractive option because patients often complain that photodynamic therapy (PDT) is painful. Plus, there seemed to be an added benefit: The anesthetic cream increased aminolevulanic acid (ALA) uptake by the skin, such that ALA incubation times before application of the light source could be greatly compressed. But clinical outcomes were too unpredictable, he said at the seminar sponsored by Skin Disease Education Foundation (SDEF). After several patients had unintended florid full photopeels involving 1½ weeks of downtime, it was time to abandon the practice.
When ALA-PDT first appeared about 10 years ago, the indication was for the treatment of actinic keratoses (AKs). Today this approved indication remains the No. 1 reason Dr. Ross utilizes the therapy, he said. But PDT’s role has expanded off label to include cosmetic procedures and the treatment of warts, nonmelanoma skin cancer, nevus sebaceous, as well as acne, which he considers "one of the great opportunities for PDT." In addition, in Asia, dermatologists are now refining the use of PDT with hematoporphyrin derivatives for the treatment of port wine stains and other vascular lesions.
Many U.S. dermatologists have incorporated PDT into their practices, with ALA being more widely used as a photosensitizer than methyl aminolevulinate (MAL). Yet PDT is a therapy that hasn’t been fully optimized; it is still fraught with side effects and suboptimal results, he said. Moreover, some fundamental issues regarding PDT remain unanswered: For example, the optimal duration of photosensitizer incubation time for various indications is still controversial.
Dr. Ross said he opts for what he considers a middle-of-the-road approach, with ALA application times of about 90-120 minutes, which he views as having an optimal balance between side effects and effectiveness. Even so, he noted that among the 8-10 patients per week he treats with ALA-PDT on average, 1 or 2 experience mild side effects.
"I don’t think we’re quite ‘there’ yet with PDT, although we’re getting closer. There are still so many tricks involved in making it work without side effects. We’ve still got some work to do," he said.
In addition to advising his colleagues to stay away from topical anesthetic creams, he offered additional tips for the use of ALA-PDT. Among them:
• Sending acne into long-term remission via PDT remains the Holy Grail, he said. The objective is to enhance the fluorescence of protoporphyrin 9 at the sebaceous gland while sparing the epidermis.
Some investigators are using low-intensity blue light at the skin surface while the photosensitizer is incubating in order to bleach it out of the epidermis, or, alternatively, warming and cooling the skin.
The best results Dr. Ross said he has seen have come through an arduous regimen involving three 3-hour-long ALA applications scheduled a month apart. There’s a delayed effect, with significant improvement coming at 3-6 months.
"It’s a tough, tough therapy to get through. There are lots of pustules and papules, and the acne invariably gets worse before it gets better. This doesn’t play into the hands of the typical teenager, who wants to get better right away," Dr. Ross said.
• A creamy solution of ALA will create more protoporphyrin 9 than an aqueous solution will.
• A red light source should be considered for deeper structures, such as basal cell carcinomas or sebaceous glands, and blue light for treating more superficial skin lesions. Although continuous blue light is 40 times more potent per photon than red light in exciting protoporphyrin 9, it doesn’t penetrate as deeply.
• Performing low-density fractional CO2 ablative laser therapy prior to application of the photosensitizing agent is "an exciting advance" in PDT, he said.
The innovation was developed by an international team led by investigators at the Wellman Center for Photomedicine at Massachusetts General Hospital, Boston, who published a split-face randomized study involving 15 patients with a total of 212 facial AKs. At the 3-month follow-up, the complete response rate of grade II-III lesions treated with MAL-PDT preceded by fractional ablative laser therapy was 87.5%, vs. 58.8% with conventional MAL-PDT. The complete response rate of grade I AKs was 100% with fractional laser/MAL-PDT, vs. 79% for MAL-PDT alone.
The fractional laser–pretreated areas also displayed significantly greater improvement in photoaging and fewer new AKs at follow-up: 3, compared with 11. But these superior outcomes came at a price: higher pain scores during illumination and significantly worse erythema and crusting post treatment (Br. J. Dermatol. 2012 Feb. 20 [doi:10.1111/j.1365-2133.2012.10893.x]).
Fractional CO2 laser pretreatment enhances conversion of the photosensitizing agent to protoporphyrin 9, Dr. Ross explained. He compared the tiny holes in the skin created by the fractional laser to the process of aerating a lawn. The holes create conduits for the photosensitizer to bypass the stratum corneum, which is the major obstacle to uptake of ALA or MAL. Once the photosensitizer skips past the stratum corneum, it quickly spreads laterally throughout the epidermis.
However, Dr. Ross offered a note of caution regarding this novel approach. He said that he performed the therapy recently and found that 30 minutes of ALA incubation was too much.
"If you do these procedures, I would say go very light and just leave the ALA for less than 30 minutes to start, because the response you’re going to get when using a fractional laser beforehand is profound. It’s a huge difference," he said.
Dr. Ross reported that he serves as a consultant to and receives research support from Palomar. He also disclosed receiving research support from Candela, Cutera, Lumenis, Sciton, and Ulthera.
SDEF and this news organization are owned by Elsevier.
WAIKOLOA, HAWAII – Using EMLA or lidocaine cream in conjunction with topical 5-aminolevulinic acid photodynamic therapy may boost uptake of the photosensitizer, but the clinical outcomes can be unpredictable, according to Dr. E. Victor Ross.
"Our worst scenarios have been when we’ve used EMLA on the skin. We’ve stopped using EMLA or lidocaine cream on the skin in ALA-PDT because of the very pronounced, enhanced photodynamic effects. You get great long-term results, but such a bad short-term result that lots of people didn’t want to go through it," said Dr. Ross of Scripps Clinic Laser and Cosmetic Dermatology Center in Carmel Valley, Calif.
Using a topical numbing agent seemed to be an attractive option because patients often complain that photodynamic therapy (PDT) is painful. Plus, there seemed to be an added benefit: The anesthetic cream increased aminolevulanic acid (ALA) uptake by the skin, such that ALA incubation times before application of the light source could be greatly compressed. But clinical outcomes were too unpredictable, he said at the seminar sponsored by Skin Disease Education Foundation (SDEF). After several patients had unintended florid full photopeels involving 1½ weeks of downtime, it was time to abandon the practice.
When ALA-PDT first appeared about 10 years ago, the indication was for the treatment of actinic keratoses (AKs). Today this approved indication remains the No. 1 reason Dr. Ross utilizes the therapy, he said. But PDT’s role has expanded off label to include cosmetic procedures and the treatment of warts, nonmelanoma skin cancer, nevus sebaceous, as well as acne, which he considers "one of the great opportunities for PDT." In addition, in Asia, dermatologists are now refining the use of PDT with hematoporphyrin derivatives for the treatment of port wine stains and other vascular lesions.
Many U.S. dermatologists have incorporated PDT into their practices, with ALA being more widely used as a photosensitizer than methyl aminolevulinate (MAL). Yet PDT is a therapy that hasn’t been fully optimized; it is still fraught with side effects and suboptimal results, he said. Moreover, some fundamental issues regarding PDT remain unanswered: For example, the optimal duration of photosensitizer incubation time for various indications is still controversial.
Dr. Ross said he opts for what he considers a middle-of-the-road approach, with ALA application times of about 90-120 minutes, which he views as having an optimal balance between side effects and effectiveness. Even so, he noted that among the 8-10 patients per week he treats with ALA-PDT on average, 1 or 2 experience mild side effects.
"I don’t think we’re quite ‘there’ yet with PDT, although we’re getting closer. There are still so many tricks involved in making it work without side effects. We’ve still got some work to do," he said.
In addition to advising his colleagues to stay away from topical anesthetic creams, he offered additional tips for the use of ALA-PDT. Among them:
• Sending acne into long-term remission via PDT remains the Holy Grail, he said. The objective is to enhance the fluorescence of protoporphyrin 9 at the sebaceous gland while sparing the epidermis.
Some investigators are using low-intensity blue light at the skin surface while the photosensitizer is incubating in order to bleach it out of the epidermis, or, alternatively, warming and cooling the skin.
The best results Dr. Ross said he has seen have come through an arduous regimen involving three 3-hour-long ALA applications scheduled a month apart. There’s a delayed effect, with significant improvement coming at 3-6 months.
"It’s a tough, tough therapy to get through. There are lots of pustules and papules, and the acne invariably gets worse before it gets better. This doesn’t play into the hands of the typical teenager, who wants to get better right away," Dr. Ross said.
• A creamy solution of ALA will create more protoporphyrin 9 than an aqueous solution will.
• A red light source should be considered for deeper structures, such as basal cell carcinomas or sebaceous glands, and blue light for treating more superficial skin lesions. Although continuous blue light is 40 times more potent per photon than red light in exciting protoporphyrin 9, it doesn’t penetrate as deeply.
• Performing low-density fractional CO2 ablative laser therapy prior to application of the photosensitizing agent is "an exciting advance" in PDT, he said.
The innovation was developed by an international team led by investigators at the Wellman Center for Photomedicine at Massachusetts General Hospital, Boston, who published a split-face randomized study involving 15 patients with a total of 212 facial AKs. At the 3-month follow-up, the complete response rate of grade II-III lesions treated with MAL-PDT preceded by fractional ablative laser therapy was 87.5%, vs. 58.8% with conventional MAL-PDT. The complete response rate of grade I AKs was 100% with fractional laser/MAL-PDT, vs. 79% for MAL-PDT alone.
The fractional laser–pretreated areas also displayed significantly greater improvement in photoaging and fewer new AKs at follow-up: 3, compared with 11. But these superior outcomes came at a price: higher pain scores during illumination and significantly worse erythema and crusting post treatment (Br. J. Dermatol. 2012 Feb. 20 [doi:10.1111/j.1365-2133.2012.10893.x]).
Fractional CO2 laser pretreatment enhances conversion of the photosensitizing agent to protoporphyrin 9, Dr. Ross explained. He compared the tiny holes in the skin created by the fractional laser to the process of aerating a lawn. The holes create conduits for the photosensitizer to bypass the stratum corneum, which is the major obstacle to uptake of ALA or MAL. Once the photosensitizer skips past the stratum corneum, it quickly spreads laterally throughout the epidermis.
However, Dr. Ross offered a note of caution regarding this novel approach. He said that he performed the therapy recently and found that 30 minutes of ALA incubation was too much.
"If you do these procedures, I would say go very light and just leave the ALA for less than 30 minutes to start, because the response you’re going to get when using a fractional laser beforehand is profound. It’s a huge difference," he said.
Dr. Ross reported that he serves as a consultant to and receives research support from Palomar. He also disclosed receiving research support from Candela, Cutera, Lumenis, Sciton, and Ulthera.
SDEF and this news organization are owned by Elsevier.
EXPERT ANALYSIS FROM THE SDEF HAWAII DERMATOLOGY SEMINAR
Don't Delay Psoriasis Treatment: The Skinny Podcast
In this month's program, Dr. Alexa B. Kimball discusses why psoriasis treatment shouldn't be delayed.
Dr. James M. Spencer offers his insights on the pros and cons of bringing radiation treatment back to the dermatologist's office.
Then, a marketing guru offers tips on how to keep cosmetic patients coming back.
Cosmetic counter host Dr. Lily Talakoub explains what it really means when a product claims to be all "natural."
And finally, Dr. Alan Rockoff shares one of the more humorous places people have gone to read his new book.
In this month's program, Dr. Alexa B. Kimball discusses why psoriasis treatment shouldn't be delayed.
Dr. James M. Spencer offers his insights on the pros and cons of bringing radiation treatment back to the dermatologist's office.
Then, a marketing guru offers tips on how to keep cosmetic patients coming back.
Cosmetic counter host Dr. Lily Talakoub explains what it really means when a product claims to be all "natural."
And finally, Dr. Alan Rockoff shares one of the more humorous places people have gone to read his new book.
In this month's program, Dr. Alexa B. Kimball discusses why psoriasis treatment shouldn't be delayed.
Dr. James M. Spencer offers his insights on the pros and cons of bringing radiation treatment back to the dermatologist's office.
Then, a marketing guru offers tips on how to keep cosmetic patients coming back.
Cosmetic counter host Dr. Lily Talakoub explains what it really means when a product claims to be all "natural."
And finally, Dr. Alan Rockoff shares one of the more humorous places people have gone to read his new book.
Off-Label Use of Aprepitant Quells Refractory Pruritus
WAIKOLOA, HAWAII – Oral aprepitant has shown considerable promise as a novel therapy in patients with refractory pruritus, including those with severe itching induced by biologic therapies for cancer.
"I think it’s a tool we should consider using in our patients with pruritus who do not respond to the usual measures," Dr. Mario E. Lacouture said at the annual Hawaii Dermatology Seminar sponsored by Skin Disease Education Foundation (SDEF).
Aprepitant (Emend) is approved as an antiemetic agent for cancer patients undergoing chemotherapy. But a growing body of data, including two single-arm proof-of-concept studies, has shown a high response rate in patients with itching resistant to standard treatment, including corticosteroids, UV therapy, and antihistamines, noted Dr. Lacouture, a dermatologist at Memorial Sloan-Kettering Cancer Center, New York.
Severe itching is increasingly recognized as an important side effect of targeted biologic agents for cancer, including erlotinib (Tarceva) and cetuximab (Erbitux). At last year’s meeting of the American Society of Clinical Oncology, Dr. Daniele Santini and coworkers at the Università "Campus Bio-Medico" di Roma presented a study involving 22 patients with severe pruritus treated with aprepitant using a regimen of 125 mg on day 1 and 80 mg on days 3 and 5.
After the single 3-dose cycle of aprepitant, the patients’ median pruritus intensity plummeted from a baseline of 8 down to 1 on a 0-10 visual analog scale (VAS). Twenty of 22 patients experienced a greater than 50% reduction in pruritus. The median duration of benefit following a 3-dose cycle given over 5 days was 25 days.
In the second proof-of-concept study, dermatologists at the Westfälische Wilhelms-Universität Münster (Germany) reported on 20 patients with refractory chronic pruritus from nonmalignant underlying disorders placed on aprepitant at 80 mg/day for 1 week. Sixteen of the 20 patients experienced a marked reduction in itch intensity. From a baseline mean of 8.4 points on the VAS, the group as a whole averaged a 42% improvement to a post-treatment score of 4.3.
A significant reduction in itching was observed as early as 2 days after initiating aprepitant in the study population (PLoS One. 2010;5:e10968).
Patients with itching related to prurigo nodularis or atopic disease responded better than those without the conditions, and patients under age 60 obtained greater improvement than older patients. However, patients with chronic kidney disease as the underlying cause of their itching achieved only minimal benefit.
Side effects were confined to mild, non-treatment-limiting nausea, dizziness, and drowsiness in three patients.
Between the two proof-of-concept studies and smaller published case series, Dr. Lacouture said that he is aware of 63 reported patients with severe refractory pruritus, including 6 with Sézary syndrome and 2 with mycosis fungoides, who were treated with aprepitant on various dosing regimens. The result was an improvement from a mean baseline VAS of 8.4 to 2.3.
Aprepitant’s mechanism of benefit in cases of severe pruritus is believed to lie in the drug’s high affinity as an antagonist of neurokinin receptor 1, which is expressed in the skin and CNS, he explained. Substance P, an important mediator in the initiation and maintenance of pruritus, binds to this receptor.
Dry skin due to radiation therapy, chemotherapy, or cachexia is the most common cause of pruritus in oncology. But he added that pruritus is intrinsically often a prominent feature of a variety of paraneoplastic diseases and malignancies, and aprepitant is worthy of study in refractory cases.
Pruritic paraneoplastic dermatoses in which the skin findings are an indication of potential concurrent underlying malignancy include dermatomyositis, erythroderma, generalized granuloma annulare, and acrokeratosis paraneoplastica.
Hematologic malignancies where itch often figures prominently include Hodgkin’s disease, polycythemia vera, and Waldenström macroglobulinemia. The solid cancers most frequently accompanied by severe itching include malignancies of the brain, prostate, rectum, and vulva.
Dr. Lacouture reported serving as a consultant to more than a dozen pharmaceutical companies, although Merck, which markets aprepitant, is not among them.
SDEF and this news organization are owned by Elsevier.
WAIKOLOA, HAWAII – Oral aprepitant has shown considerable promise as a novel therapy in patients with refractory pruritus, including those with severe itching induced by biologic therapies for cancer.
"I think it’s a tool we should consider using in our patients with pruritus who do not respond to the usual measures," Dr. Mario E. Lacouture said at the annual Hawaii Dermatology Seminar sponsored by Skin Disease Education Foundation (SDEF).
Aprepitant (Emend) is approved as an antiemetic agent for cancer patients undergoing chemotherapy. But a growing body of data, including two single-arm proof-of-concept studies, has shown a high response rate in patients with itching resistant to standard treatment, including corticosteroids, UV therapy, and antihistamines, noted Dr. Lacouture, a dermatologist at Memorial Sloan-Kettering Cancer Center, New York.
Severe itching is increasingly recognized as an important side effect of targeted biologic agents for cancer, including erlotinib (Tarceva) and cetuximab (Erbitux). At last year’s meeting of the American Society of Clinical Oncology, Dr. Daniele Santini and coworkers at the Università "Campus Bio-Medico" di Roma presented a study involving 22 patients with severe pruritus treated with aprepitant using a regimen of 125 mg on day 1 and 80 mg on days 3 and 5.
After the single 3-dose cycle of aprepitant, the patients’ median pruritus intensity plummeted from a baseline of 8 down to 1 on a 0-10 visual analog scale (VAS). Twenty of 22 patients experienced a greater than 50% reduction in pruritus. The median duration of benefit following a 3-dose cycle given over 5 days was 25 days.
In the second proof-of-concept study, dermatologists at the Westfälische Wilhelms-Universität Münster (Germany) reported on 20 patients with refractory chronic pruritus from nonmalignant underlying disorders placed on aprepitant at 80 mg/day for 1 week. Sixteen of the 20 patients experienced a marked reduction in itch intensity. From a baseline mean of 8.4 points on the VAS, the group as a whole averaged a 42% improvement to a post-treatment score of 4.3.
A significant reduction in itching was observed as early as 2 days after initiating aprepitant in the study population (PLoS One. 2010;5:e10968).
Patients with itching related to prurigo nodularis or atopic disease responded better than those without the conditions, and patients under age 60 obtained greater improvement than older patients. However, patients with chronic kidney disease as the underlying cause of their itching achieved only minimal benefit.
Side effects were confined to mild, non-treatment-limiting nausea, dizziness, and drowsiness in three patients.
Between the two proof-of-concept studies and smaller published case series, Dr. Lacouture said that he is aware of 63 reported patients with severe refractory pruritus, including 6 with Sézary syndrome and 2 with mycosis fungoides, who were treated with aprepitant on various dosing regimens. The result was an improvement from a mean baseline VAS of 8.4 to 2.3.
Aprepitant’s mechanism of benefit in cases of severe pruritus is believed to lie in the drug’s high affinity as an antagonist of neurokinin receptor 1, which is expressed in the skin and CNS, he explained. Substance P, an important mediator in the initiation and maintenance of pruritus, binds to this receptor.
Dry skin due to radiation therapy, chemotherapy, or cachexia is the most common cause of pruritus in oncology. But he added that pruritus is intrinsically often a prominent feature of a variety of paraneoplastic diseases and malignancies, and aprepitant is worthy of study in refractory cases.
Pruritic paraneoplastic dermatoses in which the skin findings are an indication of potential concurrent underlying malignancy include dermatomyositis, erythroderma, generalized granuloma annulare, and acrokeratosis paraneoplastica.
Hematologic malignancies where itch often figures prominently include Hodgkin’s disease, polycythemia vera, and Waldenström macroglobulinemia. The solid cancers most frequently accompanied by severe itching include malignancies of the brain, prostate, rectum, and vulva.
Dr. Lacouture reported serving as a consultant to more than a dozen pharmaceutical companies, although Merck, which markets aprepitant, is not among them.
SDEF and this news organization are owned by Elsevier.
WAIKOLOA, HAWAII – Oral aprepitant has shown considerable promise as a novel therapy in patients with refractory pruritus, including those with severe itching induced by biologic therapies for cancer.
"I think it’s a tool we should consider using in our patients with pruritus who do not respond to the usual measures," Dr. Mario E. Lacouture said at the annual Hawaii Dermatology Seminar sponsored by Skin Disease Education Foundation (SDEF).
Aprepitant (Emend) is approved as an antiemetic agent for cancer patients undergoing chemotherapy. But a growing body of data, including two single-arm proof-of-concept studies, has shown a high response rate in patients with itching resistant to standard treatment, including corticosteroids, UV therapy, and antihistamines, noted Dr. Lacouture, a dermatologist at Memorial Sloan-Kettering Cancer Center, New York.
Severe itching is increasingly recognized as an important side effect of targeted biologic agents for cancer, including erlotinib (Tarceva) and cetuximab (Erbitux). At last year’s meeting of the American Society of Clinical Oncology, Dr. Daniele Santini and coworkers at the Università "Campus Bio-Medico" di Roma presented a study involving 22 patients with severe pruritus treated with aprepitant using a regimen of 125 mg on day 1 and 80 mg on days 3 and 5.
After the single 3-dose cycle of aprepitant, the patients’ median pruritus intensity plummeted from a baseline of 8 down to 1 on a 0-10 visual analog scale (VAS). Twenty of 22 patients experienced a greater than 50% reduction in pruritus. The median duration of benefit following a 3-dose cycle given over 5 days was 25 days.
In the second proof-of-concept study, dermatologists at the Westfälische Wilhelms-Universität Münster (Germany) reported on 20 patients with refractory chronic pruritus from nonmalignant underlying disorders placed on aprepitant at 80 mg/day for 1 week. Sixteen of the 20 patients experienced a marked reduction in itch intensity. From a baseline mean of 8.4 points on the VAS, the group as a whole averaged a 42% improvement to a post-treatment score of 4.3.
A significant reduction in itching was observed as early as 2 days after initiating aprepitant in the study population (PLoS One. 2010;5:e10968).
Patients with itching related to prurigo nodularis or atopic disease responded better than those without the conditions, and patients under age 60 obtained greater improvement than older patients. However, patients with chronic kidney disease as the underlying cause of their itching achieved only minimal benefit.
Side effects were confined to mild, non-treatment-limiting nausea, dizziness, and drowsiness in three patients.
Between the two proof-of-concept studies and smaller published case series, Dr. Lacouture said that he is aware of 63 reported patients with severe refractory pruritus, including 6 with Sézary syndrome and 2 with mycosis fungoides, who were treated with aprepitant on various dosing regimens. The result was an improvement from a mean baseline VAS of 8.4 to 2.3.
Aprepitant’s mechanism of benefit in cases of severe pruritus is believed to lie in the drug’s high affinity as an antagonist of neurokinin receptor 1, which is expressed in the skin and CNS, he explained. Substance P, an important mediator in the initiation and maintenance of pruritus, binds to this receptor.
Dry skin due to radiation therapy, chemotherapy, or cachexia is the most common cause of pruritus in oncology. But he added that pruritus is intrinsically often a prominent feature of a variety of paraneoplastic diseases and malignancies, and aprepitant is worthy of study in refractory cases.
Pruritic paraneoplastic dermatoses in which the skin findings are an indication of potential concurrent underlying malignancy include dermatomyositis, erythroderma, generalized granuloma annulare, and acrokeratosis paraneoplastica.
Hematologic malignancies where itch often figures prominently include Hodgkin’s disease, polycythemia vera, and Waldenström macroglobulinemia. The solid cancers most frequently accompanied by severe itching include malignancies of the brain, prostate, rectum, and vulva.
Dr. Lacouture reported serving as a consultant to more than a dozen pharmaceutical companies, although Merck, which markets aprepitant, is not among them.
SDEF and this news organization are owned by Elsevier.
EXPERT ANALYSIS FROM THE SDEF HAWAII DERMATOLOGY SEMINAR
What You and Your Patients Need to Know About Vitamin D
Jennifer G. Powers, MD, and Barbara A. Gilchrest, MD
“Vitamin D” is the term commonly used to denote the lipid-soluble hormone critical for calcium homeostasis and skeletal maintenance. A precursor to the active compound is found in many plants and animal tissues and can be absorbed from the gut; it can also be derived from cell membranes in the epidermis during ultraviolet B irradiation. This compound is then hydroxylated sequentially in the liver and kidney to produce the active hormone 1,25(OH)2D that binds its nuclear receptor to modulate gene expression. Recently, vitamin D hydroxylases and the nuclear receptor have been identified in many tissues, suggesting previously unrecognized roles for vitamin D. Some epidemiologic studies have also correlated low levels of the inactive storage form 25(OH)D with an increased incidence or prevalence of a variety of diseases, suggesting that large oral supplements and/or increased ultraviolet (UV) exposure might therefore improve individual health. However, randomized, prospective controlled trials comparing vitamin D supplements with placebo have not supported this belief. Moreover, current evidence supports the conclusion that protection from UV radiation does not compromise vitamin D status or lead to iatrogenic disease. In contrast, high vitamin D levels appear to incur a risk of kidney stones and other adverse effects. In the case of true vitamin D deficiency, supplements are a more reliable and quantifiable source of the vitamin than UV exposure.
*For a PDF of the full article, click on the link to the left of this introduction.
Jennifer G. Powers, MD, and Barbara A. Gilchrest, MD
“Vitamin D” is the term commonly used to denote the lipid-soluble hormone critical for calcium homeostasis and skeletal maintenance. A precursor to the active compound is found in many plants and animal tissues and can be absorbed from the gut; it can also be derived from cell membranes in the epidermis during ultraviolet B irradiation. This compound is then hydroxylated sequentially in the liver and kidney to produce the active hormone 1,25(OH)2D that binds its nuclear receptor to modulate gene expression. Recently, vitamin D hydroxylases and the nuclear receptor have been identified in many tissues, suggesting previously unrecognized roles for vitamin D. Some epidemiologic studies have also correlated low levels of the inactive storage form 25(OH)D with an increased incidence or prevalence of a variety of diseases, suggesting that large oral supplements and/or increased ultraviolet (UV) exposure might therefore improve individual health. However, randomized, prospective controlled trials comparing vitamin D supplements with placebo have not supported this belief. Moreover, current evidence supports the conclusion that protection from UV radiation does not compromise vitamin D status or lead to iatrogenic disease. In contrast, high vitamin D levels appear to incur a risk of kidney stones and other adverse effects. In the case of true vitamin D deficiency, supplements are a more reliable and quantifiable source of the vitamin than UV exposure.
*For a PDF of the full article, click on the link to the left of this introduction.
Jennifer G. Powers, MD, and Barbara A. Gilchrest, MD
“Vitamin D” is the term commonly used to denote the lipid-soluble hormone critical for calcium homeostasis and skeletal maintenance. A precursor to the active compound is found in many plants and animal tissues and can be absorbed from the gut; it can also be derived from cell membranes in the epidermis during ultraviolet B irradiation. This compound is then hydroxylated sequentially in the liver and kidney to produce the active hormone 1,25(OH)2D that binds its nuclear receptor to modulate gene expression. Recently, vitamin D hydroxylases and the nuclear receptor have been identified in many tissues, suggesting previously unrecognized roles for vitamin D. Some epidemiologic studies have also correlated low levels of the inactive storage form 25(OH)D with an increased incidence or prevalence of a variety of diseases, suggesting that large oral supplements and/or increased ultraviolet (UV) exposure might therefore improve individual health. However, randomized, prospective controlled trials comparing vitamin D supplements with placebo have not supported this belief. Moreover, current evidence supports the conclusion that protection from UV radiation does not compromise vitamin D status or lead to iatrogenic disease. In contrast, high vitamin D levels appear to incur a risk of kidney stones and other adverse effects. In the case of true vitamin D deficiency, supplements are a more reliable and quantifiable source of the vitamin than UV exposure.
*For a PDF of the full article, click on the link to the left of this introduction.
Vitamin A: History, Current Uses, and Controversies
M. Shane Chapman, MD
Vitamin A is required for the proper functioning of many important metabolic and physiologic activities, including vision, gene transcription, the immune system and skin cell differentiation. Both excessive and deficient levels of vitamin A lead to poor functioning of many human systems. The biologically active form, retinoic acid, binds to nuclear receptors that facilitate transcription that ultimately leads to it’s physiological effects. Retinoids are derivatives of vitamin A that are medications used to treat acne vulgaris, psoriasis, ichthyosis (and other disorders of keratinization), skin cancer prevention as well as several bone marrow derived neoplasias. Systemic retinoids are teratogenic and have to be prescribed with caution and close oversight. Other potential adverse events are controversial. These include the relationship of retinoid derivatives in sunscreens, their effects on bone mineral density, depression and suicidal ideation and inflammatory bowel disease. These controversies will be discussed in detail.
*For a PDF of the full article, click on the link to the left of this introduction.
M. Shane Chapman, MD
Vitamin A is required for the proper functioning of many important metabolic and physiologic activities, including vision, gene transcription, the immune system and skin cell differentiation. Both excessive and deficient levels of vitamin A lead to poor functioning of many human systems. The biologically active form, retinoic acid, binds to nuclear receptors that facilitate transcription that ultimately leads to it’s physiological effects. Retinoids are derivatives of vitamin A that are medications used to treat acne vulgaris, psoriasis, ichthyosis (and other disorders of keratinization), skin cancer prevention as well as several bone marrow derived neoplasias. Systemic retinoids are teratogenic and have to be prescribed with caution and close oversight. Other potential adverse events are controversial. These include the relationship of retinoid derivatives in sunscreens, their effects on bone mineral density, depression and suicidal ideation and inflammatory bowel disease. These controversies will be discussed in detail.
*For a PDF of the full article, click on the link to the left of this introduction.
M. Shane Chapman, MD
Vitamin A is required for the proper functioning of many important metabolic and physiologic activities, including vision, gene transcription, the immune system and skin cell differentiation. Both excessive and deficient levels of vitamin A lead to poor functioning of many human systems. The biologically active form, retinoic acid, binds to nuclear receptors that facilitate transcription that ultimately leads to it’s physiological effects. Retinoids are derivatives of vitamin A that are medications used to treat acne vulgaris, psoriasis, ichthyosis (and other disorders of keratinization), skin cancer prevention as well as several bone marrow derived neoplasias. Systemic retinoids are teratogenic and have to be prescribed with caution and close oversight. Other potential adverse events are controversial. These include the relationship of retinoid derivatives in sunscreens, their effects on bone mineral density, depression and suicidal ideation and inflammatory bowel disease. These controversies will be discussed in detail.
*For a PDF of the full article, click on the link to the left of this introduction.
Menus for Managing Patients With Cutaneous T-Cell Lymphoma
Brian Poligone, MD, PhD, and Peter Heald, MD
In the management of patients with cutaneous T-cell lymphoma (CTCL), there are numerous distinct therapy options. Each of these therapies is discussed in terms of when to use it, what factors limit the success of the treatment, and what to expect. A menu is defined as a list of items from which to choose. The treatments for CTCL are presented in various menus where they are options for a particular goal in a particular setting of CTCL. The best recognized clinical scenarios of CTCL are those recognized by the staging system: limited patch plaque (T1), disseminated patch plaque (T2), erythroderma (T4), and tumor (T3). Each phase of the disease will have the menu of therapy options presented for a given goal of management.
*For a PDF of the full article, click on the link to the left of this introduction.
Brian Poligone, MD, PhD, and Peter Heald, MD
In the management of patients with cutaneous T-cell lymphoma (CTCL), there are numerous distinct therapy options. Each of these therapies is discussed in terms of when to use it, what factors limit the success of the treatment, and what to expect. A menu is defined as a list of items from which to choose. The treatments for CTCL are presented in various menus where they are options for a particular goal in a particular setting of CTCL. The best recognized clinical scenarios of CTCL are those recognized by the staging system: limited patch plaque (T1), disseminated patch plaque (T2), erythroderma (T4), and tumor (T3). Each phase of the disease will have the menu of therapy options presented for a given goal of management.
*For a PDF of the full article, click on the link to the left of this introduction.
Brian Poligone, MD, PhD, and Peter Heald, MD
In the management of patients with cutaneous T-cell lymphoma (CTCL), there are numerous distinct therapy options. Each of these therapies is discussed in terms of when to use it, what factors limit the success of the treatment, and what to expect. A menu is defined as a list of items from which to choose. The treatments for CTCL are presented in various menus where they are options for a particular goal in a particular setting of CTCL. The best recognized clinical scenarios of CTCL are those recognized by the staging system: limited patch plaque (T1), disseminated patch plaque (T2), erythroderma (T4), and tumor (T3). Each phase of the disease will have the menu of therapy options presented for a given goal of management.
*For a PDF of the full article, click on the link to the left of this introduction.