User login
New Vemurafenib Data Highlight Long-Term Melanoma Survival
A phase-II clinical trial with the longest follow-up to date confirmed that vemurafenib induces a clinical response in more than half of patients with advanced melanoma expressing BRAFV600 mutations, according to a report in the Feb. 23 issue of the New England Journal of Medicine.
Median overall survival was 16 months in these patients, who had highly unfavorable characteristics at baseline. With follow-up as long as 20 months, this study "provides critical information on long-term survival" that was not available from previous studies of vemurafenib (Zelboraf) in metastatic melanoma, said Dr. Jeffrey A. Sosman of the Vanderbilt-Ingram Cancer Center, Nashville, Tenn., and his associates.
The Food and Drug Administration approved vemurafenib on Aug. 17, 2011 for patients with unresectable or metastatic melanoma with the BRAFV600E mutation as detected by an FDA-approved test. The FDA acted after a pivotal phase-III trial showed vemurafenib improved overall survival compared with dacarbazine. When the phase-III data were analyzed, median follow-up was just 6.2 months in the experimental arm, and median overall survival had not been reached for patients treated with vemurafenib.
The FDA also considered the currently reported phase-II trial, which included 132 patients with stage-IV melanoma that had progressed despite at least one systemic treatment. Tumor tissue from 56% of patients screened for the trial had a BRAFV600 mutation, which was required for study entry. Vemurafenib is a potent kinase inhibitor that was developed to target this mutation within cancer cells specifically.
Patients in the study were given oral vemurafenib (960 mg twice daily) and followed every 6 weeks for disease progression at 10 medical centers in the United States and 3 in Australia. Median follow-up for efficacy was 13 months (range, 0.6-20 months). Treatment response was assessed by both the investigative team and by a blinded independent review committee.
The committee found that 8 patients (6%) showed a complete response and 62 (47%) showed a partial response to vemurafenib, for an overall response rate of 53%. A total of 38 patients (29%) showed stable disease. Only 18 (14%) showed progressive disease; 6 patients could not be assessed.
Dr. Sosman and his colleagues found a 5% complete response rate and a 52% partial response rate, for an overall response rate of 57%, which generally accorded with the independent review committee’s findings.
Molecular sequencing showed 122 patients in the trial had BRAFV600E mutations and 10 patients had BRAFV600K mutations. Four of the 10 patients with the BRAFV600K mutation had a partial response and 3 had stable disease.
These response rates are higher than those associated with any previous treatments for advanced melanoma, the researchers noted. The lowest response occurred in patients with an LDL level more than 1.5 times the upper limit of normal.
The median duration of response was 6.7 months, and one-fourth of the subjects remained progression free for more than a year. Median overall survival was 16 months. At the time of the data cut-off (July 1, 2011), 62 patients (47% of the study population) were alive.
"Most objective responses were evident at the time of the first set of scans (week 6), but in some patients, responses did not appear until the patient had been receiving the drug for more than 6 months," the researchers said (N. Engl. J. Med. 2012;366:707-14).
The longer follow-up in this study thus permitted the investigators to observe these late responses.
Overall survival was 77% at 6 months and 58% at 12 months, and it was estimated to be 43% at 18 months, they noted.
Until now, median overall of metastatic melanoma has been 6-10 months, Dr. Sosman and his colleagues noted. In previously untreated patients who responded to ipilimumab (Yervoy) – an immunotherapy also approved last year – it reached 11 months.
Most patients had at least one adverse event related to vemurafenib, but most were not severe. Adverse effects chiefly were arthralgia, rash, photosensitivity, fatigue, and alopecia. Although many patients required dose interruptions (64%) or reductions (45%), "patients were able to receive most of their intended daily dose," the investigators wrote.
Four patients discontinued the study drug altogether, including one who developed a retinal-vein occlusion. Another patient died from rapidly progressive melanoma and renal failure that may have been related to the study drug.
Vemurafenib is known to have the paradoxical effect of promoting cutaneous squamous cell carcinomas or keratoacanthomas, so the study subjects were closely monitored for these. Thirty-four patients (26%) developed such neoplasms, usually at the start of therapy. All were managed with resection and vemurafenib was not discontinued. Another eight patients were found to have basal cell carcinomas.
"As with most targeted therapies that block a driver oncogene," vemurafenib will likely encounter resistance with long-term use, Dr. Sosman and his colleagues said. The mechanisms of resistance to vemurafenib are currently being investigated, so that strategies to prevent or overcome it can be devised.
This study was funded by Hoffman-LaRoche. Dr. Sosman and his associates reported ties to various companies, including Abraxis, Altor, Amgen, BMS, Celldex, Genentech, Genesis Biopharma, GlaxoSmithKline, Hoffman-La Roche, Merck, Novartis, Pfizer, Plexxikon, Prometheus, Roche, and Serametrix.
A phase-II clinical trial with the longest follow-up to date confirmed that vemurafenib induces a clinical response in more than half of patients with advanced melanoma expressing BRAFV600 mutations, according to a report in the Feb. 23 issue of the New England Journal of Medicine.
Median overall survival was 16 months in these patients, who had highly unfavorable characteristics at baseline. With follow-up as long as 20 months, this study "provides critical information on long-term survival" that was not available from previous studies of vemurafenib (Zelboraf) in metastatic melanoma, said Dr. Jeffrey A. Sosman of the Vanderbilt-Ingram Cancer Center, Nashville, Tenn., and his associates.
The Food and Drug Administration approved vemurafenib on Aug. 17, 2011 for patients with unresectable or metastatic melanoma with the BRAFV600E mutation as detected by an FDA-approved test. The FDA acted after a pivotal phase-III trial showed vemurafenib improved overall survival compared with dacarbazine. When the phase-III data were analyzed, median follow-up was just 6.2 months in the experimental arm, and median overall survival had not been reached for patients treated with vemurafenib.
The FDA also considered the currently reported phase-II trial, which included 132 patients with stage-IV melanoma that had progressed despite at least one systemic treatment. Tumor tissue from 56% of patients screened for the trial had a BRAFV600 mutation, which was required for study entry. Vemurafenib is a potent kinase inhibitor that was developed to target this mutation within cancer cells specifically.
Patients in the study were given oral vemurafenib (960 mg twice daily) and followed every 6 weeks for disease progression at 10 medical centers in the United States and 3 in Australia. Median follow-up for efficacy was 13 months (range, 0.6-20 months). Treatment response was assessed by both the investigative team and by a blinded independent review committee.
The committee found that 8 patients (6%) showed a complete response and 62 (47%) showed a partial response to vemurafenib, for an overall response rate of 53%. A total of 38 patients (29%) showed stable disease. Only 18 (14%) showed progressive disease; 6 patients could not be assessed.
Dr. Sosman and his colleagues found a 5% complete response rate and a 52% partial response rate, for an overall response rate of 57%, which generally accorded with the independent review committee’s findings.
Molecular sequencing showed 122 patients in the trial had BRAFV600E mutations and 10 patients had BRAFV600K mutations. Four of the 10 patients with the BRAFV600K mutation had a partial response and 3 had stable disease.
These response rates are higher than those associated with any previous treatments for advanced melanoma, the researchers noted. The lowest response occurred in patients with an LDL level more than 1.5 times the upper limit of normal.
The median duration of response was 6.7 months, and one-fourth of the subjects remained progression free for more than a year. Median overall survival was 16 months. At the time of the data cut-off (July 1, 2011), 62 patients (47% of the study population) were alive.
"Most objective responses were evident at the time of the first set of scans (week 6), but in some patients, responses did not appear until the patient had been receiving the drug for more than 6 months," the researchers said (N. Engl. J. Med. 2012;366:707-14).
The longer follow-up in this study thus permitted the investigators to observe these late responses.
Overall survival was 77% at 6 months and 58% at 12 months, and it was estimated to be 43% at 18 months, they noted.
Until now, median overall of metastatic melanoma has been 6-10 months, Dr. Sosman and his colleagues noted. In previously untreated patients who responded to ipilimumab (Yervoy) – an immunotherapy also approved last year – it reached 11 months.
Most patients had at least one adverse event related to vemurafenib, but most were not severe. Adverse effects chiefly were arthralgia, rash, photosensitivity, fatigue, and alopecia. Although many patients required dose interruptions (64%) or reductions (45%), "patients were able to receive most of their intended daily dose," the investigators wrote.
Four patients discontinued the study drug altogether, including one who developed a retinal-vein occlusion. Another patient died from rapidly progressive melanoma and renal failure that may have been related to the study drug.
Vemurafenib is known to have the paradoxical effect of promoting cutaneous squamous cell carcinomas or keratoacanthomas, so the study subjects were closely monitored for these. Thirty-four patients (26%) developed such neoplasms, usually at the start of therapy. All were managed with resection and vemurafenib was not discontinued. Another eight patients were found to have basal cell carcinomas.
"As with most targeted therapies that block a driver oncogene," vemurafenib will likely encounter resistance with long-term use, Dr. Sosman and his colleagues said. The mechanisms of resistance to vemurafenib are currently being investigated, so that strategies to prevent or overcome it can be devised.
This study was funded by Hoffman-LaRoche. Dr. Sosman and his associates reported ties to various companies, including Abraxis, Altor, Amgen, BMS, Celldex, Genentech, Genesis Biopharma, GlaxoSmithKline, Hoffman-La Roche, Merck, Novartis, Pfizer, Plexxikon, Prometheus, Roche, and Serametrix.
A phase-II clinical trial with the longest follow-up to date confirmed that vemurafenib induces a clinical response in more than half of patients with advanced melanoma expressing BRAFV600 mutations, according to a report in the Feb. 23 issue of the New England Journal of Medicine.
Median overall survival was 16 months in these patients, who had highly unfavorable characteristics at baseline. With follow-up as long as 20 months, this study "provides critical information on long-term survival" that was not available from previous studies of vemurafenib (Zelboraf) in metastatic melanoma, said Dr. Jeffrey A. Sosman of the Vanderbilt-Ingram Cancer Center, Nashville, Tenn., and his associates.
The Food and Drug Administration approved vemurafenib on Aug. 17, 2011 for patients with unresectable or metastatic melanoma with the BRAFV600E mutation as detected by an FDA-approved test. The FDA acted after a pivotal phase-III trial showed vemurafenib improved overall survival compared with dacarbazine. When the phase-III data were analyzed, median follow-up was just 6.2 months in the experimental arm, and median overall survival had not been reached for patients treated with vemurafenib.
The FDA also considered the currently reported phase-II trial, which included 132 patients with stage-IV melanoma that had progressed despite at least one systemic treatment. Tumor tissue from 56% of patients screened for the trial had a BRAFV600 mutation, which was required for study entry. Vemurafenib is a potent kinase inhibitor that was developed to target this mutation within cancer cells specifically.
Patients in the study were given oral vemurafenib (960 mg twice daily) and followed every 6 weeks for disease progression at 10 medical centers in the United States and 3 in Australia. Median follow-up for efficacy was 13 months (range, 0.6-20 months). Treatment response was assessed by both the investigative team and by a blinded independent review committee.
The committee found that 8 patients (6%) showed a complete response and 62 (47%) showed a partial response to vemurafenib, for an overall response rate of 53%. A total of 38 patients (29%) showed stable disease. Only 18 (14%) showed progressive disease; 6 patients could not be assessed.
Dr. Sosman and his colleagues found a 5% complete response rate and a 52% partial response rate, for an overall response rate of 57%, which generally accorded with the independent review committee’s findings.
Molecular sequencing showed 122 patients in the trial had BRAFV600E mutations and 10 patients had BRAFV600K mutations. Four of the 10 patients with the BRAFV600K mutation had a partial response and 3 had stable disease.
These response rates are higher than those associated with any previous treatments for advanced melanoma, the researchers noted. The lowest response occurred in patients with an LDL level more than 1.5 times the upper limit of normal.
The median duration of response was 6.7 months, and one-fourth of the subjects remained progression free for more than a year. Median overall survival was 16 months. At the time of the data cut-off (July 1, 2011), 62 patients (47% of the study population) were alive.
"Most objective responses were evident at the time of the first set of scans (week 6), but in some patients, responses did not appear until the patient had been receiving the drug for more than 6 months," the researchers said (N. Engl. J. Med. 2012;366:707-14).
The longer follow-up in this study thus permitted the investigators to observe these late responses.
Overall survival was 77% at 6 months and 58% at 12 months, and it was estimated to be 43% at 18 months, they noted.
Until now, median overall of metastatic melanoma has been 6-10 months, Dr. Sosman and his colleagues noted. In previously untreated patients who responded to ipilimumab (Yervoy) – an immunotherapy also approved last year – it reached 11 months.
Most patients had at least one adverse event related to vemurafenib, but most were not severe. Adverse effects chiefly were arthralgia, rash, photosensitivity, fatigue, and alopecia. Although many patients required dose interruptions (64%) or reductions (45%), "patients were able to receive most of their intended daily dose," the investigators wrote.
Four patients discontinued the study drug altogether, including one who developed a retinal-vein occlusion. Another patient died from rapidly progressive melanoma and renal failure that may have been related to the study drug.
Vemurafenib is known to have the paradoxical effect of promoting cutaneous squamous cell carcinomas or keratoacanthomas, so the study subjects were closely monitored for these. Thirty-four patients (26%) developed such neoplasms, usually at the start of therapy. All were managed with resection and vemurafenib was not discontinued. Another eight patients were found to have basal cell carcinomas.
"As with most targeted therapies that block a driver oncogene," vemurafenib will likely encounter resistance with long-term use, Dr. Sosman and his colleagues said. The mechanisms of resistance to vemurafenib are currently being investigated, so that strategies to prevent or overcome it can be devised.
This study was funded by Hoffman-LaRoche. Dr. Sosman and his associates reported ties to various companies, including Abraxis, Altor, Amgen, BMS, Celldex, Genentech, Genesis Biopharma, GlaxoSmithKline, Hoffman-La Roche, Merck, Novartis, Pfizer, Plexxikon, Prometheus, Roche, and Serametrix.
FROM THE NEW ENGLAND JOURNAL OF MEDICINE
Major Finding: The overall response rate to vemurafenib was 53%, and median overall survival was 16 months.
Data Source: The multicenter phase II clinical trial involved 132 patients who had advanced melanoma that expressed BRAFV600 mutations and were followed for as long as 20 months.
Disclosures: This study was funded by Hoffman-La Roche. Dr. Sosman and his associates reported ties to various companies, including Abraxis, Altor, Amgen, BMS, Celldex, Genentech, Genesis Biopharma, GlaxoSmithKline, Hoffman-La Roche, Merck, Novartis, Pfizer, Plexxikon, Prometheus, Roche, and Serametrix.
Identifying AK Variants Is Key to Management
The ability to distinguish between the two key variants of actinic keratosis is key to effective management, according to Dr. Theodore Rosen.
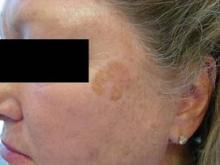
Pigmented actinic keratosis (AK) that spreads typically occurs in patients older than age 60 years and affects more women than men. The lesions are larger than 1.5 cm and have a history of lateral spread. "The lesions are flat, with a smooth, scaly surface, and they occur on sun-exposed skin, mostly the face, and near other manifestations of photodamage," noted Dr. Rosen at the annual Hawaii Dermatology Seminar sponsored by Skin Disease Education Foundation (SDEF).
Differential diagnosis includes solar lentigo, lentigo maligna, lentigo maligna melanoma, seborrheic keratosis, and pigmented squamous cell carcinoma in situ. "The presence of dots, globules, and asymmetry on dermatoscopy can help you distinguish spreading pigmented AKs from other lesions," said Dr. Rosen, professor of dermatology at Baylor College of Medicine, Houston.
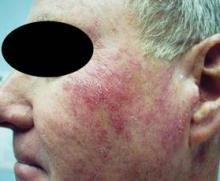
The other main variant, proliferating AK, also tends to affect patients older than age 60 but it occurs more commonly in men than in women. This variant is greater than 1.5 cm in size with a history of rapid lateral spread, and it is marked by a confluent scaly surface with some palpability. Affected areas include sun-exposed skin, mostly the face, and areas near other manifestations of photodamage. "This variant is resistant to usual topical therapy, and the differential diagnosis may include eczema, psoriasis, squamous cell carcinoma in situ, and superficial basal cell carcinoma," said Dr. Rosen, who is also chief of the dermatology clinic at the Houston-based Michael E. DeBakey VA Medical Center. Histology reveals fingerlike invasive projections into the dermis of atypical squamous cells.
The appearance of multiple AKs in a cosmetic unit indicates that the clinician should consider the entire photodamaged area to be diseased. This concept, known as field-directed therapy, "not only addresses apparent clinical lesions, but also reveals and may treat subclinical lesions," he said. "Field therapy may also offer the advantage of reversing basic cellular aberrations leading to transition into invasive cutaneous malignancy."
Another trend in the treatment of AKs is the development of new imiquimod formulations. Results from two placebo-controlled studies of daily application of 2.5% and 3.75% imiquimod creams to the face and balding scalp for two 3-week cycles revealed that both formulations were more effective than placebo and had an acceptable safety profile (J. Am. Acad. Dermatol. 2010;62:573-81). The final evaluation occurred 8 weeks after the final treatment, said Dr. Rosen, who was one of the study investigators. Results from this and other studies led to Food and Drug Administration approval of imiquimod 2.5% (Zyclara) for AKs on July 19, 2011, but the formulation is not yet commercially available.
In a more recent analysis, investigators assessed complete clearance rates in AK patients from four phase III trials of imiquimod who were followed for 1 year (J. Drugs Dermatol. 2011;10:165-70). For imiquimod 3.75% and 2.5% cream, respectively, complete clearance was sustained for 1 year in 40.5% and 33.3% of patients from the 2-week cycle studies, and in 47.9% and 43.2% of patients from the 3-week cycle studies.
Combination therapies for AKs have become increasingly popular. Treatments include cryosurgery followed by diclofenac, 5-fluorouracil followed by cryosurgery, cryosurgery followed by imiquimod 5% or 3.75%, photodynamic therapy (PTD) followed by imiquimod 5%, and tazarotene followed by PDT. "For all studies where field therapy is combined with lesion-directed therapy or with another field therapy, the combination proved superior to monotherapy," Dr. Rosen said.
He went on to note that there is considerable user variation – such as time, the distance the device is held from the skin, and the type of unit used – in the delivery of cryosurgery to treat AKs. The variation "may have more to do with skin temperature than any other variable," said Dr. Rosen. In one study, investigators used an infrared detector in a liquid nitrogen container to deliver cryotherapy to a temperature of –5° C in 36 patients with 180 thin AKs (Dermatol. Surg. 2010;36:1956-61). The clearance rates were 67% at 1 week and 100% at 6 weeks.
Treatment of AKs with radiation is another area of study. In one recent trial, 12 patients with refractory AK and 4 patients with AKs near a nonmelanoma skin cancer underwent external beam radiation therapy five times a week for 4-6 weeks. The total dose of radiation ranged from 4 to 60 Gy (J. Clin. Aesthet. Dermatol. 2011;4:47-50). Of the 16 patients, 13 had complete resolution of AKs, 1 noted a prompt recurrence, and 90% said they were "very satisfied" with the treatment results.
Treating AKs with methyl aminolevulinate photodynamic therapy (MAL-PDT) is also being investigated. In a multicenter study conducted in Scandinavia, 120 patients with 1,572 AKs underwent MAL-PDT followed by immediate exposure to the sun for 1.5-2.5 hours (Br. J. Dermatol. 2011;164:1083-90). At 3 months, 75%-77% of the lesions had cleared. This approach "has an advantage for the health care provider in that no equipment is needed," Dr. Rosen said. "The advantage for the patient is that the therapy is nearly pain free."
Dr. Rosen disclosed that he is a consultant and a member of the speakers bureau for Medicis, which makes imiquimod. He is also a consultant for LEO Pharma.
SDEF and this news organization are owned by Elsevier.
The ability to distinguish between the two key variants of actinic keratosis is key to effective management, according to Dr. Theodore Rosen.
Pigmented actinic keratosis (AK) that spreads typically occurs in patients older than age 60 years and affects more women than men. The lesions are larger than 1.5 cm and have a history of lateral spread. "The lesions are flat, with a smooth, scaly surface, and they occur on sun-exposed skin, mostly the face, and near other manifestations of photodamage," noted Dr. Rosen at the annual Hawaii Dermatology Seminar sponsored by Skin Disease Education Foundation (SDEF).
Differential diagnosis includes solar lentigo, lentigo maligna, lentigo maligna melanoma, seborrheic keratosis, and pigmented squamous cell carcinoma in situ. "The presence of dots, globules, and asymmetry on dermatoscopy can help you distinguish spreading pigmented AKs from other lesions," said Dr. Rosen, professor of dermatology at Baylor College of Medicine, Houston.
The other main variant, proliferating AK, also tends to affect patients older than age 60 but it occurs more commonly in men than in women. This variant is greater than 1.5 cm in size with a history of rapid lateral spread, and it is marked by a confluent scaly surface with some palpability. Affected areas include sun-exposed skin, mostly the face, and areas near other manifestations of photodamage. "This variant is resistant to usual topical therapy, and the differential diagnosis may include eczema, psoriasis, squamous cell carcinoma in situ, and superficial basal cell carcinoma," said Dr. Rosen, who is also chief of the dermatology clinic at the Houston-based Michael E. DeBakey VA Medical Center. Histology reveals fingerlike invasive projections into the dermis of atypical squamous cells.
The appearance of multiple AKs in a cosmetic unit indicates that the clinician should consider the entire photodamaged area to be diseased. This concept, known as field-directed therapy, "not only addresses apparent clinical lesions, but also reveals and may treat subclinical lesions," he said. "Field therapy may also offer the advantage of reversing basic cellular aberrations leading to transition into invasive cutaneous malignancy."
Another trend in the treatment of AKs is the development of new imiquimod formulations. Results from two placebo-controlled studies of daily application of 2.5% and 3.75% imiquimod creams to the face and balding scalp for two 3-week cycles revealed that both formulations were more effective than placebo and had an acceptable safety profile (J. Am. Acad. Dermatol. 2010;62:573-81). The final evaluation occurred 8 weeks after the final treatment, said Dr. Rosen, who was one of the study investigators. Results from this and other studies led to Food and Drug Administration approval of imiquimod 2.5% (Zyclara) for AKs on July 19, 2011, but the formulation is not yet commercially available.
In a more recent analysis, investigators assessed complete clearance rates in AK patients from four phase III trials of imiquimod who were followed for 1 year (J. Drugs Dermatol. 2011;10:165-70). For imiquimod 3.75% and 2.5% cream, respectively, complete clearance was sustained for 1 year in 40.5% and 33.3% of patients from the 2-week cycle studies, and in 47.9% and 43.2% of patients from the 3-week cycle studies.
Combination therapies for AKs have become increasingly popular. Treatments include cryosurgery followed by diclofenac, 5-fluorouracil followed by cryosurgery, cryosurgery followed by imiquimod 5% or 3.75%, photodynamic therapy (PTD) followed by imiquimod 5%, and tazarotene followed by PDT. "For all studies where field therapy is combined with lesion-directed therapy or with another field therapy, the combination proved superior to monotherapy," Dr. Rosen said.
He went on to note that there is considerable user variation – such as time, the distance the device is held from the skin, and the type of unit used – in the delivery of cryosurgery to treat AKs. The variation "may have more to do with skin temperature than any other variable," said Dr. Rosen. In one study, investigators used an infrared detector in a liquid nitrogen container to deliver cryotherapy to a temperature of –5° C in 36 patients with 180 thin AKs (Dermatol. Surg. 2010;36:1956-61). The clearance rates were 67% at 1 week and 100% at 6 weeks.
Treatment of AKs with radiation is another area of study. In one recent trial, 12 patients with refractory AK and 4 patients with AKs near a nonmelanoma skin cancer underwent external beam radiation therapy five times a week for 4-6 weeks. The total dose of radiation ranged from 4 to 60 Gy (J. Clin. Aesthet. Dermatol. 2011;4:47-50). Of the 16 patients, 13 had complete resolution of AKs, 1 noted a prompt recurrence, and 90% said they were "very satisfied" with the treatment results.
Treating AKs with methyl aminolevulinate photodynamic therapy (MAL-PDT) is also being investigated. In a multicenter study conducted in Scandinavia, 120 patients with 1,572 AKs underwent MAL-PDT followed by immediate exposure to the sun for 1.5-2.5 hours (Br. J. Dermatol. 2011;164:1083-90). At 3 months, 75%-77% of the lesions had cleared. This approach "has an advantage for the health care provider in that no equipment is needed," Dr. Rosen said. "The advantage for the patient is that the therapy is nearly pain free."
Dr. Rosen disclosed that he is a consultant and a member of the speakers bureau for Medicis, which makes imiquimod. He is also a consultant for LEO Pharma.
SDEF and this news organization are owned by Elsevier.
The ability to distinguish between the two key variants of actinic keratosis is key to effective management, according to Dr. Theodore Rosen.
Pigmented actinic keratosis (AK) that spreads typically occurs in patients older than age 60 years and affects more women than men. The lesions are larger than 1.5 cm and have a history of lateral spread. "The lesions are flat, with a smooth, scaly surface, and they occur on sun-exposed skin, mostly the face, and near other manifestations of photodamage," noted Dr. Rosen at the annual Hawaii Dermatology Seminar sponsored by Skin Disease Education Foundation (SDEF).
Differential diagnosis includes solar lentigo, lentigo maligna, lentigo maligna melanoma, seborrheic keratosis, and pigmented squamous cell carcinoma in situ. "The presence of dots, globules, and asymmetry on dermatoscopy can help you distinguish spreading pigmented AKs from other lesions," said Dr. Rosen, professor of dermatology at Baylor College of Medicine, Houston.
The other main variant, proliferating AK, also tends to affect patients older than age 60 but it occurs more commonly in men than in women. This variant is greater than 1.5 cm in size with a history of rapid lateral spread, and it is marked by a confluent scaly surface with some palpability. Affected areas include sun-exposed skin, mostly the face, and areas near other manifestations of photodamage. "This variant is resistant to usual topical therapy, and the differential diagnosis may include eczema, psoriasis, squamous cell carcinoma in situ, and superficial basal cell carcinoma," said Dr. Rosen, who is also chief of the dermatology clinic at the Houston-based Michael E. DeBakey VA Medical Center. Histology reveals fingerlike invasive projections into the dermis of atypical squamous cells.
The appearance of multiple AKs in a cosmetic unit indicates that the clinician should consider the entire photodamaged area to be diseased. This concept, known as field-directed therapy, "not only addresses apparent clinical lesions, but also reveals and may treat subclinical lesions," he said. "Field therapy may also offer the advantage of reversing basic cellular aberrations leading to transition into invasive cutaneous malignancy."
Another trend in the treatment of AKs is the development of new imiquimod formulations. Results from two placebo-controlled studies of daily application of 2.5% and 3.75% imiquimod creams to the face and balding scalp for two 3-week cycles revealed that both formulations were more effective than placebo and had an acceptable safety profile (J. Am. Acad. Dermatol. 2010;62:573-81). The final evaluation occurred 8 weeks after the final treatment, said Dr. Rosen, who was one of the study investigators. Results from this and other studies led to Food and Drug Administration approval of imiquimod 2.5% (Zyclara) for AKs on July 19, 2011, but the formulation is not yet commercially available.
In a more recent analysis, investigators assessed complete clearance rates in AK patients from four phase III trials of imiquimod who were followed for 1 year (J. Drugs Dermatol. 2011;10:165-70). For imiquimod 3.75% and 2.5% cream, respectively, complete clearance was sustained for 1 year in 40.5% and 33.3% of patients from the 2-week cycle studies, and in 47.9% and 43.2% of patients from the 3-week cycle studies.
Combination therapies for AKs have become increasingly popular. Treatments include cryosurgery followed by diclofenac, 5-fluorouracil followed by cryosurgery, cryosurgery followed by imiquimod 5% or 3.75%, photodynamic therapy (PTD) followed by imiquimod 5%, and tazarotene followed by PDT. "For all studies where field therapy is combined with lesion-directed therapy or with another field therapy, the combination proved superior to monotherapy," Dr. Rosen said.
He went on to note that there is considerable user variation – such as time, the distance the device is held from the skin, and the type of unit used – in the delivery of cryosurgery to treat AKs. The variation "may have more to do with skin temperature than any other variable," said Dr. Rosen. In one study, investigators used an infrared detector in a liquid nitrogen container to deliver cryotherapy to a temperature of –5° C in 36 patients with 180 thin AKs (Dermatol. Surg. 2010;36:1956-61). The clearance rates were 67% at 1 week and 100% at 6 weeks.
Treatment of AKs with radiation is another area of study. In one recent trial, 12 patients with refractory AK and 4 patients with AKs near a nonmelanoma skin cancer underwent external beam radiation therapy five times a week for 4-6 weeks. The total dose of radiation ranged from 4 to 60 Gy (J. Clin. Aesthet. Dermatol. 2011;4:47-50). Of the 16 patients, 13 had complete resolution of AKs, 1 noted a prompt recurrence, and 90% said they were "very satisfied" with the treatment results.
Treating AKs with methyl aminolevulinate photodynamic therapy (MAL-PDT) is also being investigated. In a multicenter study conducted in Scandinavia, 120 patients with 1,572 AKs underwent MAL-PDT followed by immediate exposure to the sun for 1.5-2.5 hours (Br. J. Dermatol. 2011;164:1083-90). At 3 months, 75%-77% of the lesions had cleared. This approach "has an advantage for the health care provider in that no equipment is needed," Dr. Rosen said. "The advantage for the patient is that the therapy is nearly pain free."
Dr. Rosen disclosed that he is a consultant and a member of the speakers bureau for Medicis, which makes imiquimod. He is also a consultant for LEO Pharma.
SDEF and this news organization are owned by Elsevier.
EXPERT ANALYSIS FROM THE SDEF HAWAII DERMATOLOGY SEMINAR
Posttransplant Head and Neck Tumors Tallied
MIAMI BEACH – Patients who have undergone solid organ transplantation are at greater risk for subsequent tumor development, and head and neck cancers can be particularly aggressive, according to results of a single-institution study.
Dr. Robert H. Deeb and his associates at Henry Ford Hospital in Detroit studied 3,639 patients who underwent solid organ transplantation between January 1990 and December 2011. By retrospectively searching electronic medical records, they identified 95 people who developed cutaneous, salivary gland, or mucosal malignancies.
They found a 2.1% incidence of cutaneous cancers and a 0.5% incidence of noncutaneous head and neck cancers in this population. Despite a relatively low overall 2.6% incidence, there are reasons for concern, Dr. Deeb said at the Triological Society’s Combined Sections Meeting, which was jointly sponsored by the Triological Society and the American College of Surgeons. When cutaneous cancers did occur, they were more aggressive and were associated with shorter 1-year survival rates.
Henry Ford Transplant Institute maintains a noncutaneous tumor registry. This allowed the researchers to compare survival and other factors. Compared with the "nontransplant tumor registry in our institution, we found significantly fewer patients were alive at 1 year," Dr. Deeb said. Specifically, 55% of posttransplant patients with these cancers survived to 1 year, compared with 81% of nontransplant patients. There were no significant differences between groups in terms of age, sex, stage at diagnosis, or 5-year survival.
More than half (52%) of the patients who developed skin cancers had multiple head and neck cutaneous malignancies, pointing to the aggressive nature of these cancers, said Dr. Deeb of the department of otolaryngology–head and neck surgery at Henry Ford Hospital.
"We believe these patients require aggressive screening, treatment, and follow-up," Dr. Deeb said.
Of the 78 cutaneous head and neck cancers, 51% were squamous cell carcinomas, 36% were basal cell carcinomas, and the remaining 13% were other skin cancer types. The cheek and scalp were the most common sites. The patients’ average age was 61 years at the time of diagnosis, and 74% were men. Skin cancer developed a mean 4 years after transplant surgery.
Four patients had a salivary gland cancer and 13 had an upper aerodigestive tract mucosal malignancy. The average patient age in this group was 60 years, and 94% were men. Cancer was diagnosed a mean 66 months post transplantation. All 13 of the mucosal malignancies in the study were squamous cell carcinomas, Dr. Deeb said.
Kidney, liver, and heart were the most commonly transplanted organs among patients who ultimately developed these head and neck malignancies.
Although immunosuppressants are universally prescribed to prevent organ rejection following transplant surgery, the exact mechanism for subsequent cancer development remains unknown, Dr. Deeb said. Loss of immunosurveillance is one possibility; uncontrolled cell proliferation from chronic and low-level antigenic stimulation throughout the body is another. In addition, he said, immunosuppressants might somehow activate oncogenic viruses.
Despite the findings of this study, Dr. Deeb said that the benefits of solid organ transplantation still generally outweigh the risk of subsequent cancer development.
Henry Ford Hospital funded the study. Dr. Deeb said that he had no relevant disclosures.
MIAMI BEACH – Patients who have undergone solid organ transplantation are at greater risk for subsequent tumor development, and head and neck cancers can be particularly aggressive, according to results of a single-institution study.
Dr. Robert H. Deeb and his associates at Henry Ford Hospital in Detroit studied 3,639 patients who underwent solid organ transplantation between January 1990 and December 2011. By retrospectively searching electronic medical records, they identified 95 people who developed cutaneous, salivary gland, or mucosal malignancies.
They found a 2.1% incidence of cutaneous cancers and a 0.5% incidence of noncutaneous head and neck cancers in this population. Despite a relatively low overall 2.6% incidence, there are reasons for concern, Dr. Deeb said at the Triological Society’s Combined Sections Meeting, which was jointly sponsored by the Triological Society and the American College of Surgeons. When cutaneous cancers did occur, they were more aggressive and were associated with shorter 1-year survival rates.
Henry Ford Transplant Institute maintains a noncutaneous tumor registry. This allowed the researchers to compare survival and other factors. Compared with the "nontransplant tumor registry in our institution, we found significantly fewer patients were alive at 1 year," Dr. Deeb said. Specifically, 55% of posttransplant patients with these cancers survived to 1 year, compared with 81% of nontransplant patients. There were no significant differences between groups in terms of age, sex, stage at diagnosis, or 5-year survival.
More than half (52%) of the patients who developed skin cancers had multiple head and neck cutaneous malignancies, pointing to the aggressive nature of these cancers, said Dr. Deeb of the department of otolaryngology–head and neck surgery at Henry Ford Hospital.
"We believe these patients require aggressive screening, treatment, and follow-up," Dr. Deeb said.
Of the 78 cutaneous head and neck cancers, 51% were squamous cell carcinomas, 36% were basal cell carcinomas, and the remaining 13% were other skin cancer types. The cheek and scalp were the most common sites. The patients’ average age was 61 years at the time of diagnosis, and 74% were men. Skin cancer developed a mean 4 years after transplant surgery.
Four patients had a salivary gland cancer and 13 had an upper aerodigestive tract mucosal malignancy. The average patient age in this group was 60 years, and 94% were men. Cancer was diagnosed a mean 66 months post transplantation. All 13 of the mucosal malignancies in the study were squamous cell carcinomas, Dr. Deeb said.
Kidney, liver, and heart were the most commonly transplanted organs among patients who ultimately developed these head and neck malignancies.
Although immunosuppressants are universally prescribed to prevent organ rejection following transplant surgery, the exact mechanism for subsequent cancer development remains unknown, Dr. Deeb said. Loss of immunosurveillance is one possibility; uncontrolled cell proliferation from chronic and low-level antigenic stimulation throughout the body is another. In addition, he said, immunosuppressants might somehow activate oncogenic viruses.
Despite the findings of this study, Dr. Deeb said that the benefits of solid organ transplantation still generally outweigh the risk of subsequent cancer development.
Henry Ford Hospital funded the study. Dr. Deeb said that he had no relevant disclosures.
MIAMI BEACH – Patients who have undergone solid organ transplantation are at greater risk for subsequent tumor development, and head and neck cancers can be particularly aggressive, according to results of a single-institution study.
Dr. Robert H. Deeb and his associates at Henry Ford Hospital in Detroit studied 3,639 patients who underwent solid organ transplantation between January 1990 and December 2011. By retrospectively searching electronic medical records, they identified 95 people who developed cutaneous, salivary gland, or mucosal malignancies.
They found a 2.1% incidence of cutaneous cancers and a 0.5% incidence of noncutaneous head and neck cancers in this population. Despite a relatively low overall 2.6% incidence, there are reasons for concern, Dr. Deeb said at the Triological Society’s Combined Sections Meeting, which was jointly sponsored by the Triological Society and the American College of Surgeons. When cutaneous cancers did occur, they were more aggressive and were associated with shorter 1-year survival rates.
Henry Ford Transplant Institute maintains a noncutaneous tumor registry. This allowed the researchers to compare survival and other factors. Compared with the "nontransplant tumor registry in our institution, we found significantly fewer patients were alive at 1 year," Dr. Deeb said. Specifically, 55% of posttransplant patients with these cancers survived to 1 year, compared with 81% of nontransplant patients. There were no significant differences between groups in terms of age, sex, stage at diagnosis, or 5-year survival.
More than half (52%) of the patients who developed skin cancers had multiple head and neck cutaneous malignancies, pointing to the aggressive nature of these cancers, said Dr. Deeb of the department of otolaryngology–head and neck surgery at Henry Ford Hospital.
"We believe these patients require aggressive screening, treatment, and follow-up," Dr. Deeb said.
Of the 78 cutaneous head and neck cancers, 51% were squamous cell carcinomas, 36% were basal cell carcinomas, and the remaining 13% were other skin cancer types. The cheek and scalp were the most common sites. The patients’ average age was 61 years at the time of diagnosis, and 74% were men. Skin cancer developed a mean 4 years after transplant surgery.
Four patients had a salivary gland cancer and 13 had an upper aerodigestive tract mucosal malignancy. The average patient age in this group was 60 years, and 94% were men. Cancer was diagnosed a mean 66 months post transplantation. All 13 of the mucosal malignancies in the study were squamous cell carcinomas, Dr. Deeb said.
Kidney, liver, and heart were the most commonly transplanted organs among patients who ultimately developed these head and neck malignancies.
Although immunosuppressants are universally prescribed to prevent organ rejection following transplant surgery, the exact mechanism for subsequent cancer development remains unknown, Dr. Deeb said. Loss of immunosurveillance is one possibility; uncontrolled cell proliferation from chronic and low-level antigenic stimulation throughout the body is another. In addition, he said, immunosuppressants might somehow activate oncogenic viruses.
Despite the findings of this study, Dr. Deeb said that the benefits of solid organ transplantation still generally outweigh the risk of subsequent cancer development.
Henry Ford Hospital funded the study. Dr. Deeb said that he had no relevant disclosures.
FROM THE TRIOLOGICAL SOCIETY'S COMBINED SECTIONS MEETING
Major Finding: A total of 95 people who underwent solid organ transplantation over 21 years developed subsequent head and neck cancer, for an incidence of 2.6%.
Data Source: Data are taken from a retrospective study of 3,639 solid organ transplant patients treated at the Henry Ford Hospital since 1990.
Disclosures: Henry Ford Hospital sponsored the study. Dr. Deeb reported having no financial disclosures.
Observation Okayed as Option for Some Skin Cancers
MIAMI BEACH – Observation or a "watchful waiting" approach is an acceptable option vs. re-excision for patients with positive margins remaining after removal of head and neck skin cancers, according to a retrospective study.
"A lot of times we take these lesions off and they appear to be benign, not really knowing what the preoperative histologic diagnosis is. [Then we] find out the diagnosis is cancer, and not only that, but they have a positive margin." Dr. Justin Douglas said at the Triological Society Combined Sections meeting.
The question is, he said, "Do you have to rush them back in [and re-excise] or can you sit on them for a while?"
"Dr. Douglas addresses an interesting question," said session moderator Dr. Jeffrey H. Spiegel, chief of the division of facial plastic and reconstructive surgery at Boston University Medical Center and otolaryngology faculty member at Boston University. "I see a lot of people who had squamous cells and basal cells excised sent back for re-excision ... and he’ll help us understand if I even need to be doing that."
Dr. Douglas and his colleagues studied 492 patients at the Clarksburg Veterans Affairs Hospital in Morgantown, W.Va. All had skin cancers removed from their head or neck, including 387 basal cell carcinomas and 105 squamous cell carcinomas, over 5 years.
"Do you have to rush them back in [and re-excise] or can you sit on them for a while?"
About 40% or 197 lesions had a negative margin after wide local excision. The remaining 295 lesions featured a positive margin on permanent section analysis. The positive lesions included 232 basal cell carcinomas (BCCs) and 63 squamous cell carcinomas (SCCs).
"In the group of 232 positive basal cell lesions, everybody was offered re-excision. A total 105 chose re-excision and 127 decided to just watch it, despite our best judgment," said Dr. Douglas, an otolaryngologist at the Morgantown V.A.
"Even of the squamous cell patients, 26 still said they wanted to watch it," Dr. Douglas said at the meeting, which was cosponsored by the Triological Society and the American College of Surgeons.
Of the positive margin group, none of the BCC and four (3.8%) of the SCC lesions recurred within a median follow-up of 3.7 years. This finding suggests a role for clinical observation of low-risk, nonmelanomatous cancers, Dr. Douglas said.
The four SCC lesions that recurred included one lesion in the observation-only group and three in the re-excision group. Some of these patients likely had a more aggressive form of cancer, Dr. Douglas said.
"Essentially, we feel what we learned from this study is that ... given the fact that you can handle any recurrence of these lesions if they occur, you can watch the basal cells, even with positive margins," Dr. Douglas said. "We still feel all squamous cell lesions should be taken out until you get a clear margin."
Why the majority of positive margin patients who chose observation did not experience recurrence is poorly understood, Dr. Douglas said. However, it could be the patient’s immune system, secondary to the wound healing process, comes in and cleans up any residual cells. Another possibility is that patients die from other causes before recurrence can occur. "The truth of the matter is: We don’t know. We need more studies to figure this out."
The nose was the most common site for the T1, nonmelanoma lesions. The average size was 1.2 cm. Mean patient age was 61 years and 484 (98%) were men. Patients were excluded from the study if their lesion was previously treated or they chose an option other than re-excision or observation.
Although initial excision by a Mohs surgeon would be ideal, Dr. Douglas said, in his study dermatologists and otolaryngologists removed the lesions at one of the hospital ambulatory clinics. This strategy reflects some real-world limitations. "The problem is, at least in West Virginia, Mohs surgeons are very sparse. There are probably only a handful in the state." Operating room time and scheduling (particularly for patients with many lesions), availability of a pathologist to read frozen sections, and costs are additional challenges.
Dr. Douglas and Dr. Spiegel said that they had no relevant financial disclosures.
MIAMI BEACH – Observation or a "watchful waiting" approach is an acceptable option vs. re-excision for patients with positive margins remaining after removal of head and neck skin cancers, according to a retrospective study.
"A lot of times we take these lesions off and they appear to be benign, not really knowing what the preoperative histologic diagnosis is. [Then we] find out the diagnosis is cancer, and not only that, but they have a positive margin." Dr. Justin Douglas said at the Triological Society Combined Sections meeting.
The question is, he said, "Do you have to rush them back in [and re-excise] or can you sit on them for a while?"
"Dr. Douglas addresses an interesting question," said session moderator Dr. Jeffrey H. Spiegel, chief of the division of facial plastic and reconstructive surgery at Boston University Medical Center and otolaryngology faculty member at Boston University. "I see a lot of people who had squamous cells and basal cells excised sent back for re-excision ... and he’ll help us understand if I even need to be doing that."
Dr. Douglas and his colleagues studied 492 patients at the Clarksburg Veterans Affairs Hospital in Morgantown, W.Va. All had skin cancers removed from their head or neck, including 387 basal cell carcinomas and 105 squamous cell carcinomas, over 5 years.
"Do you have to rush them back in [and re-excise] or can you sit on them for a while?"
About 40% or 197 lesions had a negative margin after wide local excision. The remaining 295 lesions featured a positive margin on permanent section analysis. The positive lesions included 232 basal cell carcinomas (BCCs) and 63 squamous cell carcinomas (SCCs).
"In the group of 232 positive basal cell lesions, everybody was offered re-excision. A total 105 chose re-excision and 127 decided to just watch it, despite our best judgment," said Dr. Douglas, an otolaryngologist at the Morgantown V.A.
"Even of the squamous cell patients, 26 still said they wanted to watch it," Dr. Douglas said at the meeting, which was cosponsored by the Triological Society and the American College of Surgeons.
Of the positive margin group, none of the BCC and four (3.8%) of the SCC lesions recurred within a median follow-up of 3.7 years. This finding suggests a role for clinical observation of low-risk, nonmelanomatous cancers, Dr. Douglas said.
The four SCC lesions that recurred included one lesion in the observation-only group and three in the re-excision group. Some of these patients likely had a more aggressive form of cancer, Dr. Douglas said.
"Essentially, we feel what we learned from this study is that ... given the fact that you can handle any recurrence of these lesions if they occur, you can watch the basal cells, even with positive margins," Dr. Douglas said. "We still feel all squamous cell lesions should be taken out until you get a clear margin."
Why the majority of positive margin patients who chose observation did not experience recurrence is poorly understood, Dr. Douglas said. However, it could be the patient’s immune system, secondary to the wound healing process, comes in and cleans up any residual cells. Another possibility is that patients die from other causes before recurrence can occur. "The truth of the matter is: We don’t know. We need more studies to figure this out."
The nose was the most common site for the T1, nonmelanoma lesions. The average size was 1.2 cm. Mean patient age was 61 years and 484 (98%) were men. Patients were excluded from the study if their lesion was previously treated or they chose an option other than re-excision or observation.
Although initial excision by a Mohs surgeon would be ideal, Dr. Douglas said, in his study dermatologists and otolaryngologists removed the lesions at one of the hospital ambulatory clinics. This strategy reflects some real-world limitations. "The problem is, at least in West Virginia, Mohs surgeons are very sparse. There are probably only a handful in the state." Operating room time and scheduling (particularly for patients with many lesions), availability of a pathologist to read frozen sections, and costs are additional challenges.
Dr. Douglas and Dr. Spiegel said that they had no relevant financial disclosures.
MIAMI BEACH – Observation or a "watchful waiting" approach is an acceptable option vs. re-excision for patients with positive margins remaining after removal of head and neck skin cancers, according to a retrospective study.
"A lot of times we take these lesions off and they appear to be benign, not really knowing what the preoperative histologic diagnosis is. [Then we] find out the diagnosis is cancer, and not only that, but they have a positive margin." Dr. Justin Douglas said at the Triological Society Combined Sections meeting.
The question is, he said, "Do you have to rush them back in [and re-excise] or can you sit on them for a while?"
"Dr. Douglas addresses an interesting question," said session moderator Dr. Jeffrey H. Spiegel, chief of the division of facial plastic and reconstructive surgery at Boston University Medical Center and otolaryngology faculty member at Boston University. "I see a lot of people who had squamous cells and basal cells excised sent back for re-excision ... and he’ll help us understand if I even need to be doing that."
Dr. Douglas and his colleagues studied 492 patients at the Clarksburg Veterans Affairs Hospital in Morgantown, W.Va. All had skin cancers removed from their head or neck, including 387 basal cell carcinomas and 105 squamous cell carcinomas, over 5 years.
"Do you have to rush them back in [and re-excise] or can you sit on them for a while?"
About 40% or 197 lesions had a negative margin after wide local excision. The remaining 295 lesions featured a positive margin on permanent section analysis. The positive lesions included 232 basal cell carcinomas (BCCs) and 63 squamous cell carcinomas (SCCs).
"In the group of 232 positive basal cell lesions, everybody was offered re-excision. A total 105 chose re-excision and 127 decided to just watch it, despite our best judgment," said Dr. Douglas, an otolaryngologist at the Morgantown V.A.
"Even of the squamous cell patients, 26 still said they wanted to watch it," Dr. Douglas said at the meeting, which was cosponsored by the Triological Society and the American College of Surgeons.
Of the positive margin group, none of the BCC and four (3.8%) of the SCC lesions recurred within a median follow-up of 3.7 years. This finding suggests a role for clinical observation of low-risk, nonmelanomatous cancers, Dr. Douglas said.
The four SCC lesions that recurred included one lesion in the observation-only group and three in the re-excision group. Some of these patients likely had a more aggressive form of cancer, Dr. Douglas said.
"Essentially, we feel what we learned from this study is that ... given the fact that you can handle any recurrence of these lesions if they occur, you can watch the basal cells, even with positive margins," Dr. Douglas said. "We still feel all squamous cell lesions should be taken out until you get a clear margin."
Why the majority of positive margin patients who chose observation did not experience recurrence is poorly understood, Dr. Douglas said. However, it could be the patient’s immune system, secondary to the wound healing process, comes in and cleans up any residual cells. Another possibility is that patients die from other causes before recurrence can occur. "The truth of the matter is: We don’t know. We need more studies to figure this out."
The nose was the most common site for the T1, nonmelanoma lesions. The average size was 1.2 cm. Mean patient age was 61 years and 484 (98%) were men. Patients were excluded from the study if their lesion was previously treated or they chose an option other than re-excision or observation.
Although initial excision by a Mohs surgeon would be ideal, Dr. Douglas said, in his study dermatologists and otolaryngologists removed the lesions at one of the hospital ambulatory clinics. This strategy reflects some real-world limitations. "The problem is, at least in West Virginia, Mohs surgeons are very sparse. There are probably only a handful in the state." Operating room time and scheduling (particularly for patients with many lesions), availability of a pathologist to read frozen sections, and costs are additional challenges.
Dr. Douglas and Dr. Spiegel said that they had no relevant financial disclosures.
FROM THE TRIOLOGICAL SOCIETY COMBINED SECTIONS MEETING
Major Finding: None of 232 basal cell carcinoma lesions and 4 (3.8%) of 63 positive squamous cell carcinoma lesions with an initial positive margin recurred over a median follow-up of 3.7 years.
Data Source: Retrospective, single-center study of 492 patients diagnosed with head and neck skin cancers over 5 years.
Disclosures: Dr. Douglas reported having no financial disclosures.
Skin of Color: Melasma Education for Patients
Melasma can be a distressing condition for our darker skinned patients. When educating them about sun protection, remind them that:
1. SPF only refers to protection against UVB radiation; it has no implication on the amount of protection against UVA. UVA is highly implicated in the progression of melasma. UVA even penetrates window glass so, if your patients drive, sit near a window, and/or are "never in the sun," remind them that they still need UVA and UVB protection every day.
2. Sunscreen needs to be applied 365 days a year. Ultraviolet light is present on cloudy, snowy, and rainy days.
3. "Broad spectrum" does not mean complete coverage. The two sunscreens that offer complete coverage against both UVA and UVB are Anthelios with Mexoryl and Neutrogena with Helioplex technology.
4. Heat can worsen melasma. If your patients work around heat, such as cooking by a hot stove or being around hot air, the heat can contribute to their melasma.
5. Computer monitors emit a small amount of UV. Suggest that melasma patients purchase a UV shield for their screens.
6. UV bracelets or beads help monitor the amount of UV in a given area. With the help of the devices, patients can monitor the amount of UV at home, at work, and in their car. Consider demonstrating the technology to your patients in the office to teach them about UV exposures in their daily environments.
7. Purchasing sunscreens with high SPF and broad spectrum coverage can be difficult. Most sunscreens leave a white or ashy residue on darker skin. Sunscreens with micronized titanium dioxide or zinc oxide can minimize the white residue and are more cosmetically appealing. Similarly, newer foundations and makeup products on the market have been developed that contain high SPF sunscreens in a tinted base. Some of my favorites include Laura Mercier tinted moisturizer and Revision Intellishade. Both have a small amount of tint to counteract the white appearance on darker skin.
8. Practice aggressive sun avoidance and protection before medical management. I don’t treat any patient with melasma unless they are vigilant about sun protection. The lasers, bleaching creams, medications (such as retinoids), and peels we use to treat melasma can make the skin more susceptible to UV radiation which can make melasma worse.
- Lily Talakoub, M.D.
Melasma can be a distressing condition for our darker skinned patients. When educating them about sun protection, remind them that:
1. SPF only refers to protection against UVB radiation; it has no implication on the amount of protection against UVA. UVA is highly implicated in the progression of melasma. UVA even penetrates window glass so, if your patients drive, sit near a window, and/or are "never in the sun," remind them that they still need UVA and UVB protection every day.
2. Sunscreen needs to be applied 365 days a year. Ultraviolet light is present on cloudy, snowy, and rainy days.
3. "Broad spectrum" does not mean complete coverage. The two sunscreens that offer complete coverage against both UVA and UVB are Anthelios with Mexoryl and Neutrogena with Helioplex technology.
4. Heat can worsen melasma. If your patients work around heat, such as cooking by a hot stove or being around hot air, the heat can contribute to their melasma.
5. Computer monitors emit a small amount of UV. Suggest that melasma patients purchase a UV shield for their screens.
6. UV bracelets or beads help monitor the amount of UV in a given area. With the help of the devices, patients can monitor the amount of UV at home, at work, and in their car. Consider demonstrating the technology to your patients in the office to teach them about UV exposures in their daily environments.
7. Purchasing sunscreens with high SPF and broad spectrum coverage can be difficult. Most sunscreens leave a white or ashy residue on darker skin. Sunscreens with micronized titanium dioxide or zinc oxide can minimize the white residue and are more cosmetically appealing. Similarly, newer foundations and makeup products on the market have been developed that contain high SPF sunscreens in a tinted base. Some of my favorites include Laura Mercier tinted moisturizer and Revision Intellishade. Both have a small amount of tint to counteract the white appearance on darker skin.
8. Practice aggressive sun avoidance and protection before medical management. I don’t treat any patient with melasma unless they are vigilant about sun protection. The lasers, bleaching creams, medications (such as retinoids), and peels we use to treat melasma can make the skin more susceptible to UV radiation which can make melasma worse.
- Lily Talakoub, M.D.
Melasma can be a distressing condition for our darker skinned patients. When educating them about sun protection, remind them that:
1. SPF only refers to protection against UVB radiation; it has no implication on the amount of protection against UVA. UVA is highly implicated in the progression of melasma. UVA even penetrates window glass so, if your patients drive, sit near a window, and/or are "never in the sun," remind them that they still need UVA and UVB protection every day.
2. Sunscreen needs to be applied 365 days a year. Ultraviolet light is present on cloudy, snowy, and rainy days.
3. "Broad spectrum" does not mean complete coverage. The two sunscreens that offer complete coverage against both UVA and UVB are Anthelios with Mexoryl and Neutrogena with Helioplex technology.
4. Heat can worsen melasma. If your patients work around heat, such as cooking by a hot stove or being around hot air, the heat can contribute to their melasma.
5. Computer monitors emit a small amount of UV. Suggest that melasma patients purchase a UV shield for their screens.
6. UV bracelets or beads help monitor the amount of UV in a given area. With the help of the devices, patients can monitor the amount of UV at home, at work, and in their car. Consider demonstrating the technology to your patients in the office to teach them about UV exposures in their daily environments.
7. Purchasing sunscreens with high SPF and broad spectrum coverage can be difficult. Most sunscreens leave a white or ashy residue on darker skin. Sunscreens with micronized titanium dioxide or zinc oxide can minimize the white residue and are more cosmetically appealing. Similarly, newer foundations and makeup products on the market have been developed that contain high SPF sunscreens in a tinted base. Some of my favorites include Laura Mercier tinted moisturizer and Revision Intellishade. Both have a small amount of tint to counteract the white appearance on darker skin.
8. Practice aggressive sun avoidance and protection before medical management. I don’t treat any patient with melasma unless they are vigilant about sun protection. The lasers, bleaching creams, medications (such as retinoids), and peels we use to treat melasma can make the skin more susceptible to UV radiation which can make melasma worse.
- Lily Talakoub, M.D.
Tanning Salons Mislead Teens, Congressional Probe Finds
Many tanning salons are downplaying the health risks associated with indoor tanning while claiming that time in a tanning bed offers an array of health benefits, according to an investigative report from Democrats on the House Energy and Commerce Committee.
Committee investigators, posing as fair-skinned teenage girls, called 300 tanning salons across the country, and found that 90% said that the use of tanning beds did not pose a health risk. When pressed about skin cancer risks, some salon employees said the link to indoor tanning was "hype" or "a big myth."
"I think it validates what we’ve been saying as dermatologists for many years," said Dr. Bruce A. Brod of the University of Pennsylvania, Philadelphia. "Unfortunately, the indoor tanning industry does not responsibly regulate [itself]."
Dr. Brod said he’s not surprised by the report’s findings. "I hear those stories all the time" from patients who have used tanning salons, he said.
The American Academy of Dermatology Association called on Congress and the states to better regulate indoor tanning. In a statement, the AADA said states should follow California’s example and ban indoor tanning among young adults under the age of 18. The organization also urged Congress to pass H.R. 1676, the Tanning Bed Cancer Control Act, which instructs the Food and Drug Administration to reexamine the way it regulates tanning beds.
The congressional investigation was requested by Rep. Carolyn Maloney (D- N.Y.), sponsor of H.R. 1676, along with other top Democrats on the House Energy and Commerce Committee.
The Indoor Tanning Association (ITA) criticized the way the investigation was conducted. Association spokesman John Overstreet said the study was flawed because it was constructed by people who already have an unfavorable opinion of the industry. It was also "unfair" for committee investigators to rely on information given over the phone to undercover investigators.
The experience would be far different if a teenager came into a tanning salon accompanied by a parent, he said, adding that in such a situation the information provided likely would be much more comprehensive and would include a discussion about potential risks.
But while the ITA disputes the findings, the organization acknowledged the need to reevaluate how the tanning salon industry could better ensure that salons provide accurate and consistent information to all consumers.
Congressional investigators also found that 78% of the tanning salons they contacted touted some type of health benefits from indoor tanning, ranging from the treatment of depression to weight loss. Salon employees also told investigators that young people were not at risk for developing skin cancer and that the rising rates of cancer were linked to the increased use of sunscreen.
The report also took the tanning industry to task for targeting teenage girls in their marketing materials. The investigators found that tanning salons often offered student discounts or other promotions for prom, homecoming, and back-to-school.
Many tanning salons are downplaying the health risks associated with indoor tanning while claiming that time in a tanning bed offers an array of health benefits, according to an investigative report from Democrats on the House Energy and Commerce Committee.
Committee investigators, posing as fair-skinned teenage girls, called 300 tanning salons across the country, and found that 90% said that the use of tanning beds did not pose a health risk. When pressed about skin cancer risks, some salon employees said the link to indoor tanning was "hype" or "a big myth."
"I think it validates what we’ve been saying as dermatologists for many years," said Dr. Bruce A. Brod of the University of Pennsylvania, Philadelphia. "Unfortunately, the indoor tanning industry does not responsibly regulate [itself]."
Dr. Brod said he’s not surprised by the report’s findings. "I hear those stories all the time" from patients who have used tanning salons, he said.
The American Academy of Dermatology Association called on Congress and the states to better regulate indoor tanning. In a statement, the AADA said states should follow California’s example and ban indoor tanning among young adults under the age of 18. The organization also urged Congress to pass H.R. 1676, the Tanning Bed Cancer Control Act, which instructs the Food and Drug Administration to reexamine the way it regulates tanning beds.
The congressional investigation was requested by Rep. Carolyn Maloney (D- N.Y.), sponsor of H.R. 1676, along with other top Democrats on the House Energy and Commerce Committee.
The Indoor Tanning Association (ITA) criticized the way the investigation was conducted. Association spokesman John Overstreet said the study was flawed because it was constructed by people who already have an unfavorable opinion of the industry. It was also "unfair" for committee investigators to rely on information given over the phone to undercover investigators.
The experience would be far different if a teenager came into a tanning salon accompanied by a parent, he said, adding that in such a situation the information provided likely would be much more comprehensive and would include a discussion about potential risks.
But while the ITA disputes the findings, the organization acknowledged the need to reevaluate how the tanning salon industry could better ensure that salons provide accurate and consistent information to all consumers.
Congressional investigators also found that 78% of the tanning salons they contacted touted some type of health benefits from indoor tanning, ranging from the treatment of depression to weight loss. Salon employees also told investigators that young people were not at risk for developing skin cancer and that the rising rates of cancer were linked to the increased use of sunscreen.
The report also took the tanning industry to task for targeting teenage girls in their marketing materials. The investigators found that tanning salons often offered student discounts or other promotions for prom, homecoming, and back-to-school.
Many tanning salons are downplaying the health risks associated with indoor tanning while claiming that time in a tanning bed offers an array of health benefits, according to an investigative report from Democrats on the House Energy and Commerce Committee.
Committee investigators, posing as fair-skinned teenage girls, called 300 tanning salons across the country, and found that 90% said that the use of tanning beds did not pose a health risk. When pressed about skin cancer risks, some salon employees said the link to indoor tanning was "hype" or "a big myth."
"I think it validates what we’ve been saying as dermatologists for many years," said Dr. Bruce A. Brod of the University of Pennsylvania, Philadelphia. "Unfortunately, the indoor tanning industry does not responsibly regulate [itself]."
Dr. Brod said he’s not surprised by the report’s findings. "I hear those stories all the time" from patients who have used tanning salons, he said.
The American Academy of Dermatology Association called on Congress and the states to better regulate indoor tanning. In a statement, the AADA said states should follow California’s example and ban indoor tanning among young adults under the age of 18. The organization also urged Congress to pass H.R. 1676, the Tanning Bed Cancer Control Act, which instructs the Food and Drug Administration to reexamine the way it regulates tanning beds.
The congressional investigation was requested by Rep. Carolyn Maloney (D- N.Y.), sponsor of H.R. 1676, along with other top Democrats on the House Energy and Commerce Committee.
The Indoor Tanning Association (ITA) criticized the way the investigation was conducted. Association spokesman John Overstreet said the study was flawed because it was constructed by people who already have an unfavorable opinion of the industry. It was also "unfair" for committee investigators to rely on information given over the phone to undercover investigators.
The experience would be far different if a teenager came into a tanning salon accompanied by a parent, he said, adding that in such a situation the information provided likely would be much more comprehensive and would include a discussion about potential risks.
But while the ITA disputes the findings, the organization acknowledged the need to reevaluate how the tanning salon industry could better ensure that salons provide accurate and consistent information to all consumers.
Congressional investigators also found that 78% of the tanning salons they contacted touted some type of health benefits from indoor tanning, ranging from the treatment of depression to weight loss. Salon employees also told investigators that young people were not at risk for developing skin cancer and that the rising rates of cancer were linked to the increased use of sunscreen.
The report also took the tanning industry to task for targeting teenage girls in their marketing materials. The investigators found that tanning salons often offered student discounts or other promotions for prom, homecoming, and back-to-school.
Mole Picking Lorikeets: The Skinny Podcast
In this month's podcast, Reporter Naseem Miller talks with Dr. Terry Cronin about a research review of medical malpractice claims filed in Florida. The findings may surprise you.
Reporter Heidi Splete chats with Dr. Doris Hexel about why when it comes to injections, less is more in the lower face.
And Reporter Jeff Evans covers a shocking finding: many melanoma survivors aren't using sunscreen.
In this month's Cosmetic Counter segment, Dr. Lily Talakoub discusses the effectiveness of anti-aging products.
And last but not least, Dr. Alan Rockoff shares a story about mole picking lorikeets.
Don't miss another episode of The Skinny Podcast; subscribe on iTunes!
In this month's podcast, Reporter Naseem Miller talks with Dr. Terry Cronin about a research review of medical malpractice claims filed in Florida. The findings may surprise you.
Reporter Heidi Splete chats with Dr. Doris Hexel about why when it comes to injections, less is more in the lower face.
And Reporter Jeff Evans covers a shocking finding: many melanoma survivors aren't using sunscreen.
In this month's Cosmetic Counter segment, Dr. Lily Talakoub discusses the effectiveness of anti-aging products.
And last but not least, Dr. Alan Rockoff shares a story about mole picking lorikeets.
Don't miss another episode of The Skinny Podcast; subscribe on iTunes!
In this month's podcast, Reporter Naseem Miller talks with Dr. Terry Cronin about a research review of medical malpractice claims filed in Florida. The findings may surprise you.
Reporter Heidi Splete chats with Dr. Doris Hexel about why when it comes to injections, less is more in the lower face.
And Reporter Jeff Evans covers a shocking finding: many melanoma survivors aren't using sunscreen.
In this month's Cosmetic Counter segment, Dr. Lily Talakoub discusses the effectiveness of anti-aging products.
And last but not least, Dr. Alan Rockoff shares a story about mole picking lorikeets.
Don't miss another episode of The Skinny Podcast; subscribe on iTunes!
Vismodegib Receives FDA Approval for Metastatic BCC
The Food and Drug Administration has approved vismodegib, an oral, once-daily medication for adults with locally advanced and metastatic advanced basal cell carcinoma on Jan. 30.
Vismodegib (Erivedge) is the first medication approved for metastatic BCC and was approved ahead of its March 8 statutory review date, according to a statement from the FDA.
The drug works by inhibiting the hedgehog pathway, which is active in most BCCs. "Our understanding of molecular pathways involved in cancer, such as the hedgehog pathway, has enabled the development of targeted drugs for specific diseases," said Dr. Richard Pazdur, director of the FDA’s Office of Hematology and Oncology Products, in a statement.
"Today’s approval provides a new treatment for people with advanced basal cell carcinoma who, until now, had no approved medicines to help shrink disfiguring or potentially life-threatening lesions," Dr. Hal Barron, Genentech’s chief medical officer and head of global product development, said in a statement.
The FDA based its approval on a single-arm, multicenter phase II study, ERIVANCE BCC. The open-label study enrolled 104 patients with locally advanced or metastatic BCC but, according to the FDA, approval was based on evaluations of 96 patients. Study participants took 150 mg vismodegib once daily.
The primary end point was objective response rate, that is, the percentage of patients who experienced complete and partial shrinkage or disappearance of the cancerous lesions after treatment. A total of 43% of patients with locally advanced BCC had partial or complete response (27 of 63) and 30% with metastatic disease had a partial response (10 of 33), according to Genentech. The median duration of response was 7.6 months.
The most common side effects were muscle spasms, hair loss, weight loss, nausea, diarrhea, fatigue, distorted sense of taste or loss of taste, decreased appetite, constipation, vomiting, and aching joints.
Vismodegib will carry a boxed warning about a potential risk of death or severe birth effects to a fetus. The FDA is not requiring a birth defects risk management program for vismodegib, but Genentech is advising female patients to use "highly effective" birth control before, during, and for 7 months after the last dose of treatment. Men should use a condom with spermicide, even if they have had a vasectomy, during sex with female partners during treatment and for 2 months after the last dose.
The company said that pregnant women are being encouraged to participate in the Erivedge pregnancy pharmacovigilance program, which collects information about exposure to that drug during pregnancy and the effects on the mother and her unborn child.
Vismodegib will be available in the United States within a few weeks and will be distributed through specialty pharmacies, according to Genentech.
The Food and Drug Administration has approved vismodegib, an oral, once-daily medication for adults with locally advanced and metastatic advanced basal cell carcinoma on Jan. 30.
Vismodegib (Erivedge) is the first medication approved for metastatic BCC and was approved ahead of its March 8 statutory review date, according to a statement from the FDA.
The drug works by inhibiting the hedgehog pathway, which is active in most BCCs. "Our understanding of molecular pathways involved in cancer, such as the hedgehog pathway, has enabled the development of targeted drugs for specific diseases," said Dr. Richard Pazdur, director of the FDA’s Office of Hematology and Oncology Products, in a statement.
"Today’s approval provides a new treatment for people with advanced basal cell carcinoma who, until now, had no approved medicines to help shrink disfiguring or potentially life-threatening lesions," Dr. Hal Barron, Genentech’s chief medical officer and head of global product development, said in a statement.
The FDA based its approval on a single-arm, multicenter phase II study, ERIVANCE BCC. The open-label study enrolled 104 patients with locally advanced or metastatic BCC but, according to the FDA, approval was based on evaluations of 96 patients. Study participants took 150 mg vismodegib once daily.
The primary end point was objective response rate, that is, the percentage of patients who experienced complete and partial shrinkage or disappearance of the cancerous lesions after treatment. A total of 43% of patients with locally advanced BCC had partial or complete response (27 of 63) and 30% with metastatic disease had a partial response (10 of 33), according to Genentech. The median duration of response was 7.6 months.
The most common side effects were muscle spasms, hair loss, weight loss, nausea, diarrhea, fatigue, distorted sense of taste or loss of taste, decreased appetite, constipation, vomiting, and aching joints.
Vismodegib will carry a boxed warning about a potential risk of death or severe birth effects to a fetus. The FDA is not requiring a birth defects risk management program for vismodegib, but Genentech is advising female patients to use "highly effective" birth control before, during, and for 7 months after the last dose of treatment. Men should use a condom with spermicide, even if they have had a vasectomy, during sex with female partners during treatment and for 2 months after the last dose.
The company said that pregnant women are being encouraged to participate in the Erivedge pregnancy pharmacovigilance program, which collects information about exposure to that drug during pregnancy and the effects on the mother and her unborn child.
Vismodegib will be available in the United States within a few weeks and will be distributed through specialty pharmacies, according to Genentech.
The Food and Drug Administration has approved vismodegib, an oral, once-daily medication for adults with locally advanced and metastatic advanced basal cell carcinoma on Jan. 30.
Vismodegib (Erivedge) is the first medication approved for metastatic BCC and was approved ahead of its March 8 statutory review date, according to a statement from the FDA.
The drug works by inhibiting the hedgehog pathway, which is active in most BCCs. "Our understanding of molecular pathways involved in cancer, such as the hedgehog pathway, has enabled the development of targeted drugs for specific diseases," said Dr. Richard Pazdur, director of the FDA’s Office of Hematology and Oncology Products, in a statement.
"Today’s approval provides a new treatment for people with advanced basal cell carcinoma who, until now, had no approved medicines to help shrink disfiguring or potentially life-threatening lesions," Dr. Hal Barron, Genentech’s chief medical officer and head of global product development, said in a statement.
The FDA based its approval on a single-arm, multicenter phase II study, ERIVANCE BCC. The open-label study enrolled 104 patients with locally advanced or metastatic BCC but, according to the FDA, approval was based on evaluations of 96 patients. Study participants took 150 mg vismodegib once daily.
The primary end point was objective response rate, that is, the percentage of patients who experienced complete and partial shrinkage or disappearance of the cancerous lesions after treatment. A total of 43% of patients with locally advanced BCC had partial or complete response (27 of 63) and 30% with metastatic disease had a partial response (10 of 33), according to Genentech. The median duration of response was 7.6 months.
The most common side effects were muscle spasms, hair loss, weight loss, nausea, diarrhea, fatigue, distorted sense of taste or loss of taste, decreased appetite, constipation, vomiting, and aching joints.
Vismodegib will carry a boxed warning about a potential risk of death or severe birth effects to a fetus. The FDA is not requiring a birth defects risk management program for vismodegib, but Genentech is advising female patients to use "highly effective" birth control before, during, and for 7 months after the last dose of treatment. Men should use a condom with spermicide, even if they have had a vasectomy, during sex with female partners during treatment and for 2 months after the last dose.
The company said that pregnant women are being encouraged to participate in the Erivedge pregnancy pharmacovigilance program, which collects information about exposure to that drug during pregnancy and the effects on the mother and her unborn child.
Vismodegib will be available in the United States within a few weeks and will be distributed through specialty pharmacies, according to Genentech.
Picato Gel Approved to Treat Actinic Keratoses
The Food and Drug Administration approved two strengths of ingenol mebutate gel for the treatment of actinic keratoses Jan. 23.
The 0.015% formulation of ingenol mebutate gel (Picato) was approved for the treatment of lesions on the face and scalp once daily for 3 consecutive days; the 0.05% gel was cleared for once-daily treatment of the trunk and extremities for 3 consecutive days. The gel may be applied to the affected area, up to one contiguous skin area of about 25 cm2.
Ingenol mebutate is taken from the milky sap of a plant called radium weed – or Euphorbia peplus – which has been used traditionally as a treatment for skin conditions. It is thought that the treatment induces cell death, but the mechanism of action isn’t fully understood.
Approval was supported by four double-blind, vehicle (placebo)-controlled trials of about 1,000 patients, two of which were done in the trunk and extremities and the other two in the neck and scalp.
In the trunk and extremity studies, ingenol mebutate gel had a complete clearance rate (100% of lesions gone) of 28% and 42%, compared with 5% for the vehicle control in both studies. Partial clearance rates (at least 75% of lesions cleared) were 44% and 55%, vs. 7% for the vehicle control in both studies.
In the face and scalp studies, the complete clearance rates were 37% and 47%, vs. 2% and 5% for controls. Partial clearance rates came in at 60% and 68%, vs. 7% and 8% for vehicle controls.
The most common adverse events were local skin reactions, application site pain, and application site pruritus.
Manufacturer LEO Pharmaceuticals is planning to launch in the United States at the end of the first quarter or the beginning of the second quarter, President and CEO John Koconis said in an interview.
"The Pink Sheet" and this news organization are owned by Elsevier.
The Food and Drug Administration approved two strengths of ingenol mebutate gel for the treatment of actinic keratoses Jan. 23.
The 0.015% formulation of ingenol mebutate gel (Picato) was approved for the treatment of lesions on the face and scalp once daily for 3 consecutive days; the 0.05% gel was cleared for once-daily treatment of the trunk and extremities for 3 consecutive days. The gel may be applied to the affected area, up to one contiguous skin area of about 25 cm2.
Ingenol mebutate is taken from the milky sap of a plant called radium weed – or Euphorbia peplus – which has been used traditionally as a treatment for skin conditions. It is thought that the treatment induces cell death, but the mechanism of action isn’t fully understood.
Approval was supported by four double-blind, vehicle (placebo)-controlled trials of about 1,000 patients, two of which were done in the trunk and extremities and the other two in the neck and scalp.
In the trunk and extremity studies, ingenol mebutate gel had a complete clearance rate (100% of lesions gone) of 28% and 42%, compared with 5% for the vehicle control in both studies. Partial clearance rates (at least 75% of lesions cleared) were 44% and 55%, vs. 7% for the vehicle control in both studies.
In the face and scalp studies, the complete clearance rates were 37% and 47%, vs. 2% and 5% for controls. Partial clearance rates came in at 60% and 68%, vs. 7% and 8% for vehicle controls.
The most common adverse events were local skin reactions, application site pain, and application site pruritus.
Manufacturer LEO Pharmaceuticals is planning to launch in the United States at the end of the first quarter or the beginning of the second quarter, President and CEO John Koconis said in an interview.
"The Pink Sheet" and this news organization are owned by Elsevier.
The Food and Drug Administration approved two strengths of ingenol mebutate gel for the treatment of actinic keratoses Jan. 23.
The 0.015% formulation of ingenol mebutate gel (Picato) was approved for the treatment of lesions on the face and scalp once daily for 3 consecutive days; the 0.05% gel was cleared for once-daily treatment of the trunk and extremities for 3 consecutive days. The gel may be applied to the affected area, up to one contiguous skin area of about 25 cm2.
Ingenol mebutate is taken from the milky sap of a plant called radium weed – or Euphorbia peplus – which has been used traditionally as a treatment for skin conditions. It is thought that the treatment induces cell death, but the mechanism of action isn’t fully understood.
Approval was supported by four double-blind, vehicle (placebo)-controlled trials of about 1,000 patients, two of which were done in the trunk and extremities and the other two in the neck and scalp.
In the trunk and extremity studies, ingenol mebutate gel had a complete clearance rate (100% of lesions gone) of 28% and 42%, compared with 5% for the vehicle control in both studies. Partial clearance rates (at least 75% of lesions cleared) were 44% and 55%, vs. 7% for the vehicle control in both studies.
In the face and scalp studies, the complete clearance rates were 37% and 47%, vs. 2% and 5% for controls. Partial clearance rates came in at 60% and 68%, vs. 7% and 8% for vehicle controls.
The most common adverse events were local skin reactions, application site pain, and application site pruritus.
Manufacturer LEO Pharmaceuticals is planning to launch in the United States at the end of the first quarter or the beginning of the second quarter, President and CEO John Koconis said in an interview.
"The Pink Sheet" and this news organization are owned by Elsevier.
Malignancy Rates Remain Stable in Ustekinumab-Treated Patients
RIO GRANDE, P.R. – Malignancy rates in psoriasis patients treated with ustekinumab did not increase significantly over 4 years of follow-up, based on pooled data from 3,117 patients enrolled in ustekinumab clinical trials.
Ustekinumab has shown effectiveness for treating moderate to severe psoriasis, but due to the potential of increased risk for cancer associated with its use, patients from several clinical trials (including PHOENIX I, PHOENIX II, and ACCEPT) are still being followed, said Dr. Kim A. Papp, director of research at Probity Medical Research, Waterloo, Ont., and colleagues.
The cumulative rates for nonmelanoma skin cancer in patients treated with ustekinumab for psoriasis remained low and stable throughout the follow-up period. A total of 0.57 cancer events per 100 person-years were reported in 2009 and 0.62 cancer events per 100 person-years were reported in 2010. The findings were presented at the annual Caribbean Dermatology Symposium.
In the complete analysis that included 6,791 patient-years of follow-up, 41 patients treated with any dose of ustekinumab developed at least one nonmelanoma skin cancer, and 3 patients developed both basal cell carcinoma and squamous cell carcinoma.
Another 42 patients developed at least one other malignancy, including 4 patients with melanoma in situ. However, no cases of invasive melanoma were observed during the study period. The other most common malignancies were prostate cancer (12 patients), colorectal cancer (4 patients) and breast cancer (3 patients).
By comparison, 39 individuals in the general population (based on the National Cancer Institute’s Surveillance, Epidemiology, and End Results database) developed at least one malignancy.
The findings were limited by the inclusion of several studies of varying lengths and by the inclusion criteria that can make comparison with the general population difficult, the researchers noted. The results suggest that rates of nonmelanoma skin cancer and other malignancies in psoriasis patients taking ustekinumab do not increase over time.
However, "additional long term data from clinical trials, observational registries, and postmarketing reporting databases will continue to define the ustekinumab malignancy risk profile," they wrote.
The study was sponsored by Centocor, which manufactures ustekinmab.
RIO GRANDE, P.R. – Malignancy rates in psoriasis patients treated with ustekinumab did not increase significantly over 4 years of follow-up, based on pooled data from 3,117 patients enrolled in ustekinumab clinical trials.
Ustekinumab has shown effectiveness for treating moderate to severe psoriasis, but due to the potential of increased risk for cancer associated with its use, patients from several clinical trials (including PHOENIX I, PHOENIX II, and ACCEPT) are still being followed, said Dr. Kim A. Papp, director of research at Probity Medical Research, Waterloo, Ont., and colleagues.
The cumulative rates for nonmelanoma skin cancer in patients treated with ustekinumab for psoriasis remained low and stable throughout the follow-up period. A total of 0.57 cancer events per 100 person-years were reported in 2009 and 0.62 cancer events per 100 person-years were reported in 2010. The findings were presented at the annual Caribbean Dermatology Symposium.
In the complete analysis that included 6,791 patient-years of follow-up, 41 patients treated with any dose of ustekinumab developed at least one nonmelanoma skin cancer, and 3 patients developed both basal cell carcinoma and squamous cell carcinoma.
Another 42 patients developed at least one other malignancy, including 4 patients with melanoma in situ. However, no cases of invasive melanoma were observed during the study period. The other most common malignancies were prostate cancer (12 patients), colorectal cancer (4 patients) and breast cancer (3 patients).
By comparison, 39 individuals in the general population (based on the National Cancer Institute’s Surveillance, Epidemiology, and End Results database) developed at least one malignancy.
The findings were limited by the inclusion of several studies of varying lengths and by the inclusion criteria that can make comparison with the general population difficult, the researchers noted. The results suggest that rates of nonmelanoma skin cancer and other malignancies in psoriasis patients taking ustekinumab do not increase over time.
However, "additional long term data from clinical trials, observational registries, and postmarketing reporting databases will continue to define the ustekinumab malignancy risk profile," they wrote.
The study was sponsored by Centocor, which manufactures ustekinmab.
RIO GRANDE, P.R. – Malignancy rates in psoriasis patients treated with ustekinumab did not increase significantly over 4 years of follow-up, based on pooled data from 3,117 patients enrolled in ustekinumab clinical trials.
Ustekinumab has shown effectiveness for treating moderate to severe psoriasis, but due to the potential of increased risk for cancer associated with its use, patients from several clinical trials (including PHOENIX I, PHOENIX II, and ACCEPT) are still being followed, said Dr. Kim A. Papp, director of research at Probity Medical Research, Waterloo, Ont., and colleagues.
The cumulative rates for nonmelanoma skin cancer in patients treated with ustekinumab for psoriasis remained low and stable throughout the follow-up period. A total of 0.57 cancer events per 100 person-years were reported in 2009 and 0.62 cancer events per 100 person-years were reported in 2010. The findings were presented at the annual Caribbean Dermatology Symposium.
In the complete analysis that included 6,791 patient-years of follow-up, 41 patients treated with any dose of ustekinumab developed at least one nonmelanoma skin cancer, and 3 patients developed both basal cell carcinoma and squamous cell carcinoma.
Another 42 patients developed at least one other malignancy, including 4 patients with melanoma in situ. However, no cases of invasive melanoma were observed during the study period. The other most common malignancies were prostate cancer (12 patients), colorectal cancer (4 patients) and breast cancer (3 patients).
By comparison, 39 individuals in the general population (based on the National Cancer Institute’s Surveillance, Epidemiology, and End Results database) developed at least one malignancy.
The findings were limited by the inclusion of several studies of varying lengths and by the inclusion criteria that can make comparison with the general population difficult, the researchers noted. The results suggest that rates of nonmelanoma skin cancer and other malignancies in psoriasis patients taking ustekinumab do not increase over time.
However, "additional long term data from clinical trials, observational registries, and postmarketing reporting databases will continue to define the ustekinumab malignancy risk profile," they wrote.
The study was sponsored by Centocor, which manufactures ustekinmab.
FROM THE ANNUAL CARIBBEAN DERMATOLOGY SYMPOSIUM
Major Finding: A total of 0.57 cancer events per 100 person-years were reported in 2009 and 0.62 cancer events per 100 person-years were reported in 2010.
Data Source: A review of 3,117 adults with psoriasis who were enrolled in ustekinumab clinical trials.
Disclosures: The study was sponsored by Centocor, which manufactures ustekinumab.