User login
T-VEC plus ipilimumab safe, effective in advanced melanoma
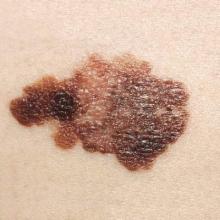
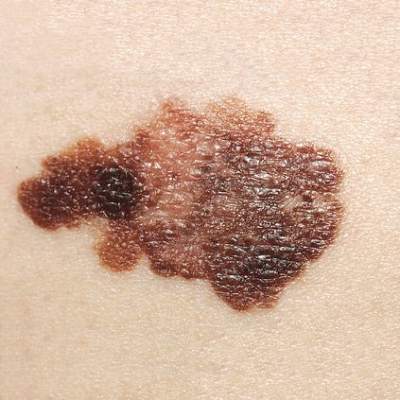
Talimogene laherparepvec (T-VEC) combined with ipilimumab was more effective in treating advanced melanoma than either treatment alone, report Igor Puzanov, MD, of Vanderbilt University Medical Center in Nashville, Tenn., and coauthors.
In a phase Ib trial of 19 patients with advanced melanoma, the results showed that 50% had positive responses to the combined immunotherapy, and 44% had durable responses lasting 6 months or longer. At 18 months, 50% of patients showed no progression, and overall patient survival was 67%, the authors reported.
The study’s safety analysis showed no new signs of safety concerns, or of any dose-limiting toxicities.
Read the full study in The Journal of Clinical Oncology.
Talimogene laherparepvec (T-VEC) combined with ipilimumab was more effective in treating advanced melanoma than either treatment alone, report Igor Puzanov, MD, of Vanderbilt University Medical Center in Nashville, Tenn., and coauthors.
In a phase Ib trial of 19 patients with advanced melanoma, the results showed that 50% had positive responses to the combined immunotherapy, and 44% had durable responses lasting 6 months or longer. At 18 months, 50% of patients showed no progression, and overall patient survival was 67%, the authors reported.
The study’s safety analysis showed no new signs of safety concerns, or of any dose-limiting toxicities.
Read the full study in The Journal of Clinical Oncology.
Talimogene laherparepvec (T-VEC) combined with ipilimumab was more effective in treating advanced melanoma than either treatment alone, report Igor Puzanov, MD, of Vanderbilt University Medical Center in Nashville, Tenn., and coauthors.
In a phase Ib trial of 19 patients with advanced melanoma, the results showed that 50% had positive responses to the combined immunotherapy, and 44% had durable responses lasting 6 months or longer. At 18 months, 50% of patients showed no progression, and overall patient survival was 67%, the authors reported.
The study’s safety analysis showed no new signs of safety concerns, or of any dose-limiting toxicities.
Read the full study in The Journal of Clinical Oncology.
FROM THE JOURNAL OF CLINICAL ONCOLOGY
Evolving therapeutic strategies maintain clinical momentum in melanoma
The past 5 years have witnessed a watershed moment in the management of metastatic melanoma. The successes of molecularly targeted and immune-based therapies have transformed it from an aggressively lethal malignancy into one that is readily treatable. Here, we discuss continued efforts to find new therapies and broaden the clinical impact of existing options to maintain the unprecedented momentum of improving patient outcomes.
Click on the PDF icon at the top of this introduction to read the full article.
The past 5 years have witnessed a watershed moment in the management of metastatic melanoma. The successes of molecularly targeted and immune-based therapies have transformed it from an aggressively lethal malignancy into one that is readily treatable. Here, we discuss continued efforts to find new therapies and broaden the clinical impact of existing options to maintain the unprecedented momentum of improving patient outcomes.
Click on the PDF icon at the top of this introduction to read the full article.
The past 5 years have witnessed a watershed moment in the management of metastatic melanoma. The successes of molecularly targeted and immune-based therapies have transformed it from an aggressively lethal malignancy into one that is readily treatable. Here, we discuss continued efforts to find new therapies and broaden the clinical impact of existing options to maintain the unprecedented momentum of improving patient outcomes.
Click on the PDF icon at the top of this introduction to read the full article.
Implementation of ipilimumab therapy in a private practice oncology group: overcoming start-up and reimbursement issues related to expensive new cancer drugs
The monoclonal antibody ipilimumab was the first treatment in more than 30 years to improve long-term survival in metastatic melanoma patients. Offering expensive ipilimumab treatment presented significant business challenges and potential financial risks for our private oncology practice and for patients because of the high acquisition cost of this agent. There was initial uncertainty about the willingness of insurance companies to reimburse for this new drug based on previous experiences in our practice with other expensive new drugs. Here we describe how our multiphysician practice methodically introduced ipilimumab treatment into the practice.
Click on the PDF icon at the top of this introduction to read the full article.
The monoclonal antibody ipilimumab was the first treatment in more than 30 years to improve long-term survival in metastatic melanoma patients. Offering expensive ipilimumab treatment presented significant business challenges and potential financial risks for our private oncology practice and for patients because of the high acquisition cost of this agent. There was initial uncertainty about the willingness of insurance companies to reimburse for this new drug based on previous experiences in our practice with other expensive new drugs. Here we describe how our multiphysician practice methodically introduced ipilimumab treatment into the practice.
Click on the PDF icon at the top of this introduction to read the full article.
The monoclonal antibody ipilimumab was the first treatment in more than 30 years to improve long-term survival in metastatic melanoma patients. Offering expensive ipilimumab treatment presented significant business challenges and potential financial risks for our private oncology practice and for patients because of the high acquisition cost of this agent. There was initial uncertainty about the willingness of insurance companies to reimburse for this new drug based on previous experiences in our practice with other expensive new drugs. Here we describe how our multiphysician practice methodically introduced ipilimumab treatment into the practice.
Click on the PDF icon at the top of this introduction to read the full article.
Checkpoint-blocking antibodies appear safe, beneficial in elderly melanoma patients
CHICAGO – Programmed cell death protein 1 (PD-1) inhibitors and combination ipilimumab and nivolumab prolonged survival, compared with ipilimumab alone in patients aged 80 and older with advanced melanoma, and the patients experienced no more adverse events than did patients of all age groups in prior published phase III studies, according to a retrospective case review.
Of 106 patients included in the current review, 74 received at least 1 dose of the anti–cytotoxic T-lymphocyte antigen 4 agent ipilimumab, 13 received at least 1 dose of an anti–PD-1 agent (pembrolizumab or nivolumab), and 8 received at least 1 dose of combination ipilimumab and nivolumab. Median survival was 7.5 months, 14.2 months, and 23.5 months for ipilimumab, anti–PD-1 agents, and combination therapy groups, respectively. The survival rates at 1 year were 40%, 56%, and 63%, respectively, Dr. Claire Frances Friedman reported at the annual meeting of the American Society of Clinical Oncology.
The differences between the groups did not reach statistical significance, said Dr. Friedman of Memorial Sloan Kettering Cancer Center, New York
Of note, a graphic representation of overall survival by treatment type shows a plateau after 24 months of treatment with ipilimumab (Ipi).
“About 20% of patients treated with Ipi have had a durable survival lasting many years,” she said, noting that this finding is similar to that based on pooled data from a previous study showing a survival rate of 22% with Ipi.
The findings are important because the highest incidence of advanced melanoma is among patients aged 80-84 years, and those over age 75 have a much higher mortality from melanoma, compared with younger patients, she said, adding that “the treatment of advanced melanoma has benefited from great strides in the area of immunotherapy, specifically the use of checkpoint-blocking antibodies.”
These antibodies work by blocking negative regulators of T-cell function, she explained.
The three used in this study are currently approved for the treatment of advanced melanoma. The combination of ipilimumab and nivolumab was approved when it was shown to improve progression-free survival vs. ipilimumab alone.
“Of note, responses to this class of drugs tend to be durable,” Dr. Friedman said.
In a prior study of nivolumab, the 5-year survival rate was 34%, she said.
However, the immune-related adverse events (irAEs) – “a unique set of side effects” associated with checkpoint-blocking antibodies – are concerning. The incidence of high-grade irAEs can vary with the type of checkpoint-blocking antibody – ranging from 16.3% with single-agent anti–PD-1 treatment to 55% with combination treatment in previous reports. The type of irAEs experienced tend to be similar across treatment types, and in a study published in 2015, were associated with a relatively high rate of treatment discontinuation (36.4% for combination treatment, 14.8% with ipilimumab, and 7.7% with nivolumab).
Given the increased mortality and morbidity in older patients, Dr. Friedman and her colleagues were interested in examining the toxicity and efficacy of checkpoint-blocking antibodies in patients aged 80 years and older. They performed the current review involving all patients aged 80 years and older at Memorial Sloan Kettering Cancer Center who were started on checkpoint blockade for stage 3 or 4 unresected melanoma between January 2008 and December 2015.
All patients had at least one infusion visit and one follow-up visit, and 12 of those in the Ipi group received subsequent anti–PD-1 therapy and were included in the toxicity analyses for both agents.
An irAE occurred in 88% of the 74 patients in the Ipi group, and 30% experienced a grade 3 or 4 irAE. The most common high-grade irAEs in this group were diarrhea, transaminitis, and rash.
Of the 25 patients receiving pembrolizumab or nivolumab, 84% experienced any irAE, and 16% experienced a grade 3 event. The most common adverse events in this group were pruritus, rash, fatigue, and musculoskeletal complaints, and one case each of grade 3 lipase, diarrhea, anemia, and nausea occurred.
Of the eight patients who received combination ipilimumab/nivolumab, 88% experienced any irAE and 63% experienced a high-grade 3 or 4 irAE. The most common high-grade irAEs in this group were elevated lipase, diarrhea, and transaminitis, occurring in 38%, 25%, and 25% of patients, respectively.
No deaths occurred in any of the groups, Dr. Friedman said.
The use of systemic steroids to manage irAEs was similar in the Ipi and anti–PD-1 monotherapy groups (28% in both groups), but 75% of the combination therapy patients were treated with systemic steroids. Infliximab was required for diarrhea in 7% of the Ipi patients and 38% of the combination therapy patients. None of the patients in any of the three groups required mycophenolate mofetil for steroid-refractory transaminitis, Dr. Friedman noted.
The findings suggest that when it comes to selecting the appropriate treatment for older patients, there are several things to consider, she said.
For example, in the elderly population, diarrhea and colitis are associated with an increased risk of dehydration and subsequent renal failure. Also, administering prednisone could lead to delirium or altered mental status, and can be of concern due to the increased incidence of comorbid conditions such as high blood pressure and diabetes, which may be worsened by steroid therapy.
Another concern is the fact that data from the rheumatology literature suggest that infliximab in the elderly may lead to increased risk of infections; a caveat is that this was in cases involving repeated doses, Dr. Friedman noted.
Treatment discontinuation was high across the groups, with 41%, 72%, and 63% of those in the Ipi, anti–PD-1, and combination therapy groups stopping treatment early. Toxicity was the reason for discontinuation in 31%, 20%, and 50%, respectively. Progression of disease or other unrelated issues were the reasons for discontinuation in the remaining patients.
“Patients aged 80 and older treated with ipilimumab had durable survival at a rate similar to that of the general population,” Dr. Friedman said.
Further, median overall survival is prolonged in older patients treated with anti–PD-1 drugs and with combination nivolumab/ipilimumab, but not significantly.
Importantly, irAEs occur more frequently with ipilimumab-based regimens in this population, she said, noting that discontinuation rates for toxicity are also higher in those aged 80 and older.
“So given these data, as clinicians we need to start thinking about individualizing treatment for our patients – using more than just the age. Physicians should consider incorporating assessments of functional status, such as the Mini-Cog or the Get Up and Go Test when deciding which of the checkpoint antibodies is appropriate for any individual patients,” she said, adding that as the population ages, more data in the elderly population will be needed.
“Clinicians should encourage older folks to participate in immuno-oncology clinical trials, and I hope that these data will encourage physicians to consider the use of checkpoint-blocking antibodies in the treatment of other malignancy types such as non–small cell lung cancer and bladder cancer, which also have a large proportion of adults over the age of 75,” she concluded.
Dr. Friedman reported receiving funds for travel, accommodations, and expenses from Bristol-Myers Squibb.
CHICAGO – Programmed cell death protein 1 (PD-1) inhibitors and combination ipilimumab and nivolumab prolonged survival, compared with ipilimumab alone in patients aged 80 and older with advanced melanoma, and the patients experienced no more adverse events than did patients of all age groups in prior published phase III studies, according to a retrospective case review.
Of 106 patients included in the current review, 74 received at least 1 dose of the anti–cytotoxic T-lymphocyte antigen 4 agent ipilimumab, 13 received at least 1 dose of an anti–PD-1 agent (pembrolizumab or nivolumab), and 8 received at least 1 dose of combination ipilimumab and nivolumab. Median survival was 7.5 months, 14.2 months, and 23.5 months for ipilimumab, anti–PD-1 agents, and combination therapy groups, respectively. The survival rates at 1 year were 40%, 56%, and 63%, respectively, Dr. Claire Frances Friedman reported at the annual meeting of the American Society of Clinical Oncology.
The differences between the groups did not reach statistical significance, said Dr. Friedman of Memorial Sloan Kettering Cancer Center, New York
Of note, a graphic representation of overall survival by treatment type shows a plateau after 24 months of treatment with ipilimumab (Ipi).
“About 20% of patients treated with Ipi have had a durable survival lasting many years,” she said, noting that this finding is similar to that based on pooled data from a previous study showing a survival rate of 22% with Ipi.
The findings are important because the highest incidence of advanced melanoma is among patients aged 80-84 years, and those over age 75 have a much higher mortality from melanoma, compared with younger patients, she said, adding that “the treatment of advanced melanoma has benefited from great strides in the area of immunotherapy, specifically the use of checkpoint-blocking antibodies.”
These antibodies work by blocking negative regulators of T-cell function, she explained.
The three used in this study are currently approved for the treatment of advanced melanoma. The combination of ipilimumab and nivolumab was approved when it was shown to improve progression-free survival vs. ipilimumab alone.
“Of note, responses to this class of drugs tend to be durable,” Dr. Friedman said.
In a prior study of nivolumab, the 5-year survival rate was 34%, she said.
However, the immune-related adverse events (irAEs) – “a unique set of side effects” associated with checkpoint-blocking antibodies – are concerning. The incidence of high-grade irAEs can vary with the type of checkpoint-blocking antibody – ranging from 16.3% with single-agent anti–PD-1 treatment to 55% with combination treatment in previous reports. The type of irAEs experienced tend to be similar across treatment types, and in a study published in 2015, were associated with a relatively high rate of treatment discontinuation (36.4% for combination treatment, 14.8% with ipilimumab, and 7.7% with nivolumab).
Given the increased mortality and morbidity in older patients, Dr. Friedman and her colleagues were interested in examining the toxicity and efficacy of checkpoint-blocking antibodies in patients aged 80 years and older. They performed the current review involving all patients aged 80 years and older at Memorial Sloan Kettering Cancer Center who were started on checkpoint blockade for stage 3 or 4 unresected melanoma between January 2008 and December 2015.
All patients had at least one infusion visit and one follow-up visit, and 12 of those in the Ipi group received subsequent anti–PD-1 therapy and were included in the toxicity analyses for both agents.
An irAE occurred in 88% of the 74 patients in the Ipi group, and 30% experienced a grade 3 or 4 irAE. The most common high-grade irAEs in this group were diarrhea, transaminitis, and rash.
Of the 25 patients receiving pembrolizumab or nivolumab, 84% experienced any irAE, and 16% experienced a grade 3 event. The most common adverse events in this group were pruritus, rash, fatigue, and musculoskeletal complaints, and one case each of grade 3 lipase, diarrhea, anemia, and nausea occurred.
Of the eight patients who received combination ipilimumab/nivolumab, 88% experienced any irAE and 63% experienced a high-grade 3 or 4 irAE. The most common high-grade irAEs in this group were elevated lipase, diarrhea, and transaminitis, occurring in 38%, 25%, and 25% of patients, respectively.
No deaths occurred in any of the groups, Dr. Friedman said.
The use of systemic steroids to manage irAEs was similar in the Ipi and anti–PD-1 monotherapy groups (28% in both groups), but 75% of the combination therapy patients were treated with systemic steroids. Infliximab was required for diarrhea in 7% of the Ipi patients and 38% of the combination therapy patients. None of the patients in any of the three groups required mycophenolate mofetil for steroid-refractory transaminitis, Dr. Friedman noted.
The findings suggest that when it comes to selecting the appropriate treatment for older patients, there are several things to consider, she said.
For example, in the elderly population, diarrhea and colitis are associated with an increased risk of dehydration and subsequent renal failure. Also, administering prednisone could lead to delirium or altered mental status, and can be of concern due to the increased incidence of comorbid conditions such as high blood pressure and diabetes, which may be worsened by steroid therapy.
Another concern is the fact that data from the rheumatology literature suggest that infliximab in the elderly may lead to increased risk of infections; a caveat is that this was in cases involving repeated doses, Dr. Friedman noted.
Treatment discontinuation was high across the groups, with 41%, 72%, and 63% of those in the Ipi, anti–PD-1, and combination therapy groups stopping treatment early. Toxicity was the reason for discontinuation in 31%, 20%, and 50%, respectively. Progression of disease or other unrelated issues were the reasons for discontinuation in the remaining patients.
“Patients aged 80 and older treated with ipilimumab had durable survival at a rate similar to that of the general population,” Dr. Friedman said.
Further, median overall survival is prolonged in older patients treated with anti–PD-1 drugs and with combination nivolumab/ipilimumab, but not significantly.
Importantly, irAEs occur more frequently with ipilimumab-based regimens in this population, she said, noting that discontinuation rates for toxicity are also higher in those aged 80 and older.
“So given these data, as clinicians we need to start thinking about individualizing treatment for our patients – using more than just the age. Physicians should consider incorporating assessments of functional status, such as the Mini-Cog or the Get Up and Go Test when deciding which of the checkpoint antibodies is appropriate for any individual patients,” she said, adding that as the population ages, more data in the elderly population will be needed.
“Clinicians should encourage older folks to participate in immuno-oncology clinical trials, and I hope that these data will encourage physicians to consider the use of checkpoint-blocking antibodies in the treatment of other malignancy types such as non–small cell lung cancer and bladder cancer, which also have a large proportion of adults over the age of 75,” she concluded.
Dr. Friedman reported receiving funds for travel, accommodations, and expenses from Bristol-Myers Squibb.
CHICAGO – Programmed cell death protein 1 (PD-1) inhibitors and combination ipilimumab and nivolumab prolonged survival, compared with ipilimumab alone in patients aged 80 and older with advanced melanoma, and the patients experienced no more adverse events than did patients of all age groups in prior published phase III studies, according to a retrospective case review.
Of 106 patients included in the current review, 74 received at least 1 dose of the anti–cytotoxic T-lymphocyte antigen 4 agent ipilimumab, 13 received at least 1 dose of an anti–PD-1 agent (pembrolizumab or nivolumab), and 8 received at least 1 dose of combination ipilimumab and nivolumab. Median survival was 7.5 months, 14.2 months, and 23.5 months for ipilimumab, anti–PD-1 agents, and combination therapy groups, respectively. The survival rates at 1 year were 40%, 56%, and 63%, respectively, Dr. Claire Frances Friedman reported at the annual meeting of the American Society of Clinical Oncology.
The differences between the groups did not reach statistical significance, said Dr. Friedman of Memorial Sloan Kettering Cancer Center, New York
Of note, a graphic representation of overall survival by treatment type shows a plateau after 24 months of treatment with ipilimumab (Ipi).
“About 20% of patients treated with Ipi have had a durable survival lasting many years,” she said, noting that this finding is similar to that based on pooled data from a previous study showing a survival rate of 22% with Ipi.
The findings are important because the highest incidence of advanced melanoma is among patients aged 80-84 years, and those over age 75 have a much higher mortality from melanoma, compared with younger patients, she said, adding that “the treatment of advanced melanoma has benefited from great strides in the area of immunotherapy, specifically the use of checkpoint-blocking antibodies.”
These antibodies work by blocking negative regulators of T-cell function, she explained.
The three used in this study are currently approved for the treatment of advanced melanoma. The combination of ipilimumab and nivolumab was approved when it was shown to improve progression-free survival vs. ipilimumab alone.
“Of note, responses to this class of drugs tend to be durable,” Dr. Friedman said.
In a prior study of nivolumab, the 5-year survival rate was 34%, she said.
However, the immune-related adverse events (irAEs) – “a unique set of side effects” associated with checkpoint-blocking antibodies – are concerning. The incidence of high-grade irAEs can vary with the type of checkpoint-blocking antibody – ranging from 16.3% with single-agent anti–PD-1 treatment to 55% with combination treatment in previous reports. The type of irAEs experienced tend to be similar across treatment types, and in a study published in 2015, were associated with a relatively high rate of treatment discontinuation (36.4% for combination treatment, 14.8% with ipilimumab, and 7.7% with nivolumab).
Given the increased mortality and morbidity in older patients, Dr. Friedman and her colleagues were interested in examining the toxicity and efficacy of checkpoint-blocking antibodies in patients aged 80 years and older. They performed the current review involving all patients aged 80 years and older at Memorial Sloan Kettering Cancer Center who were started on checkpoint blockade for stage 3 or 4 unresected melanoma between January 2008 and December 2015.
All patients had at least one infusion visit and one follow-up visit, and 12 of those in the Ipi group received subsequent anti–PD-1 therapy and were included in the toxicity analyses for both agents.
An irAE occurred in 88% of the 74 patients in the Ipi group, and 30% experienced a grade 3 or 4 irAE. The most common high-grade irAEs in this group were diarrhea, transaminitis, and rash.
Of the 25 patients receiving pembrolizumab or nivolumab, 84% experienced any irAE, and 16% experienced a grade 3 event. The most common adverse events in this group were pruritus, rash, fatigue, and musculoskeletal complaints, and one case each of grade 3 lipase, diarrhea, anemia, and nausea occurred.
Of the eight patients who received combination ipilimumab/nivolumab, 88% experienced any irAE and 63% experienced a high-grade 3 or 4 irAE. The most common high-grade irAEs in this group were elevated lipase, diarrhea, and transaminitis, occurring in 38%, 25%, and 25% of patients, respectively.
No deaths occurred in any of the groups, Dr. Friedman said.
The use of systemic steroids to manage irAEs was similar in the Ipi and anti–PD-1 monotherapy groups (28% in both groups), but 75% of the combination therapy patients were treated with systemic steroids. Infliximab was required for diarrhea in 7% of the Ipi patients and 38% of the combination therapy patients. None of the patients in any of the three groups required mycophenolate mofetil for steroid-refractory transaminitis, Dr. Friedman noted.
The findings suggest that when it comes to selecting the appropriate treatment for older patients, there are several things to consider, she said.
For example, in the elderly population, diarrhea and colitis are associated with an increased risk of dehydration and subsequent renal failure. Also, administering prednisone could lead to delirium or altered mental status, and can be of concern due to the increased incidence of comorbid conditions such as high blood pressure and diabetes, which may be worsened by steroid therapy.
Another concern is the fact that data from the rheumatology literature suggest that infliximab in the elderly may lead to increased risk of infections; a caveat is that this was in cases involving repeated doses, Dr. Friedman noted.
Treatment discontinuation was high across the groups, with 41%, 72%, and 63% of those in the Ipi, anti–PD-1, and combination therapy groups stopping treatment early. Toxicity was the reason for discontinuation in 31%, 20%, and 50%, respectively. Progression of disease or other unrelated issues were the reasons for discontinuation in the remaining patients.
“Patients aged 80 and older treated with ipilimumab had durable survival at a rate similar to that of the general population,” Dr. Friedman said.
Further, median overall survival is prolonged in older patients treated with anti–PD-1 drugs and with combination nivolumab/ipilimumab, but not significantly.
Importantly, irAEs occur more frequently with ipilimumab-based regimens in this population, she said, noting that discontinuation rates for toxicity are also higher in those aged 80 and older.
“So given these data, as clinicians we need to start thinking about individualizing treatment for our patients – using more than just the age. Physicians should consider incorporating assessments of functional status, such as the Mini-Cog or the Get Up and Go Test when deciding which of the checkpoint antibodies is appropriate for any individual patients,” she said, adding that as the population ages, more data in the elderly population will be needed.
“Clinicians should encourage older folks to participate in immuno-oncology clinical trials, and I hope that these data will encourage physicians to consider the use of checkpoint-blocking antibodies in the treatment of other malignancy types such as non–small cell lung cancer and bladder cancer, which also have a large proportion of adults over the age of 75,” she concluded.
Dr. Friedman reported receiving funds for travel, accommodations, and expenses from Bristol-Myers Squibb.
AT THE 2016 ASCO ANNUAL MEETING
Key clinical point: PD-1 inhibitors and combination ipilimumab/nivolumab prolonged survival vs. ipilimumab alone in patients aged 80 and older with advanced melanoma, and had an adverse event profile similar to that in all age groups.
Major finding: Median survival was 7.5 months, 14.2 months, and 23.5 months for ipilimumab, anti–PD-1 agents, and combination therapy groups, respectively. The survival rates at 1 year were 40%, 56%, and 63%, respectively,
Data source: A retrospective review of 106 cases.
Disclosures: Dr Friedman reported receiving funds for travel, accommodations, and expenses from Bristol-Myers Squibb.
SEER data underscore mortality associated with thin melanomas
SCOTTSDALE, ARIZ. – Thin melanomas account for most melanoma deaths, even though T4 lesions have the worst prognosis, according to an analysis of melanoma data from the National Cancer Institute’s Surveillance, Epidemiology, and End Results (SEER) program.
In 2015, a study from Queensland, Australia, reported that more patients died from T1 melanomas than did from T4 melanomas (J Invest Dermatol. 2015 Apr;135:1190-3). But a similar analysis in the United States was not available, so Dr. Shoshana Landow, a dermatologist at the Providence (R.I.) Veterans Affairs Medical Center,and her associates extracted SEER 13 data for invasive melanomas between 1992 through 2003. These registries covered Atlanta, Connecticut, Detroit, rural Georgia, Hawaii, Iowa, Los Angeles, New Mexico, San Francisco-Oakland, San Jose-Monterey, Seattle-Puget Sound, Utah, and the Alaska Native Tumor Registry, according to the SEER website.
Among 105,264 recorded melanomas during this period, 37,210 tumors were in situ, the investigators reported in a poster presented at the annual meeting of the Society for Investigative Dermatology. They excluded those cases, as well as 4,789 cases that involved more than one primary melanoma, 8,637 that extended beyond the skin at diagnosis, 5,308 with no record of thickness, and one that was not known to be primary.
That left 49,319 invasive melanomas, of which 35,509 (72%) were T1 (0.01-1.00mm), 7,879 (16%) were T2 (1.01-2.00 mm), 3,948 (8%) were T3 (2.01-4.00 mm), and 1,983 (4%) were T4 (more than 4.00 mm). A total of 17% of the T1 lesions were 0.01-0.25 mm thick at diagnosis, while 42% were 0.26-0.50 mm thick, 25% were 0.51-0.75 mm thick, and 16% were 0.76-1.00 mm thick.
Ten years after diagnosis, 3,660 (7.4%) patients had died of melanoma, according to the study. These deaths included 1,072 T1 patients, 974 T2 patients, 985 T3 patients, and 629 T4 patients. But while T4 lesions accounted for the fewest number of deaths and made up only 4% of all invasive melanomas, the 10-year mortality rate for T4 lesions was nearly 32%, vs. 25% for T3 lesions, 12% for T2 lesions, and 3% for T1 lesions.
Most (42%) T1 lesions were 0.26-0.50 mm thick, 25% of T1 lesions were 0.51-0.75 mm thick, and the remaining T1 lesions were nearly evenly split between the thinnest (0.01-0.25 mm) and the thickest (0.76-1.00mm) categories, the researchers also reported. When considering only the T1 lesions, 10-year mortality rates were 3% for the 0.01-0.25 mm category, 1.9% for the 0.26-0.5 mm category, 3.7% for the 0.51-0.75 mm category, and 5.8% for the 0.76-1.00 mm category.
Thus, the thinnest invasive melanomas had a higher 10-year death rate than did the next-thinnest category, a finding that “demands explanation” and was not caused by ulceration, the researchers noted.
“The greatest total number of deaths was from T1 melanomas and the smallest number from T4, despite the fact that prognosis worsened as melanoma thickened from T1 to T4,” they concluded in their poster. “Our findings highlight the heavy death toll of thin melanomas.”
The authors did not specify funding sources, and had no disclosures.
SCOTTSDALE, ARIZ. – Thin melanomas account for most melanoma deaths, even though T4 lesions have the worst prognosis, according to an analysis of melanoma data from the National Cancer Institute’s Surveillance, Epidemiology, and End Results (SEER) program.
In 2015, a study from Queensland, Australia, reported that more patients died from T1 melanomas than did from T4 melanomas (J Invest Dermatol. 2015 Apr;135:1190-3). But a similar analysis in the United States was not available, so Dr. Shoshana Landow, a dermatologist at the Providence (R.I.) Veterans Affairs Medical Center,and her associates extracted SEER 13 data for invasive melanomas between 1992 through 2003. These registries covered Atlanta, Connecticut, Detroit, rural Georgia, Hawaii, Iowa, Los Angeles, New Mexico, San Francisco-Oakland, San Jose-Monterey, Seattle-Puget Sound, Utah, and the Alaska Native Tumor Registry, according to the SEER website.
Among 105,264 recorded melanomas during this period, 37,210 tumors were in situ, the investigators reported in a poster presented at the annual meeting of the Society for Investigative Dermatology. They excluded those cases, as well as 4,789 cases that involved more than one primary melanoma, 8,637 that extended beyond the skin at diagnosis, 5,308 with no record of thickness, and one that was not known to be primary.
That left 49,319 invasive melanomas, of which 35,509 (72%) were T1 (0.01-1.00mm), 7,879 (16%) were T2 (1.01-2.00 mm), 3,948 (8%) were T3 (2.01-4.00 mm), and 1,983 (4%) were T4 (more than 4.00 mm). A total of 17% of the T1 lesions were 0.01-0.25 mm thick at diagnosis, while 42% were 0.26-0.50 mm thick, 25% were 0.51-0.75 mm thick, and 16% were 0.76-1.00 mm thick.
Ten years after diagnosis, 3,660 (7.4%) patients had died of melanoma, according to the study. These deaths included 1,072 T1 patients, 974 T2 patients, 985 T3 patients, and 629 T4 patients. But while T4 lesions accounted for the fewest number of deaths and made up only 4% of all invasive melanomas, the 10-year mortality rate for T4 lesions was nearly 32%, vs. 25% for T3 lesions, 12% for T2 lesions, and 3% for T1 lesions.
Most (42%) T1 lesions were 0.26-0.50 mm thick, 25% of T1 lesions were 0.51-0.75 mm thick, and the remaining T1 lesions were nearly evenly split between the thinnest (0.01-0.25 mm) and the thickest (0.76-1.00mm) categories, the researchers also reported. When considering only the T1 lesions, 10-year mortality rates were 3% for the 0.01-0.25 mm category, 1.9% for the 0.26-0.5 mm category, 3.7% for the 0.51-0.75 mm category, and 5.8% for the 0.76-1.00 mm category.
Thus, the thinnest invasive melanomas had a higher 10-year death rate than did the next-thinnest category, a finding that “demands explanation” and was not caused by ulceration, the researchers noted.
“The greatest total number of deaths was from T1 melanomas and the smallest number from T4, despite the fact that prognosis worsened as melanoma thickened from T1 to T4,” they concluded in their poster. “Our findings highlight the heavy death toll of thin melanomas.”
The authors did not specify funding sources, and had no disclosures.
SCOTTSDALE, ARIZ. – Thin melanomas account for most melanoma deaths, even though T4 lesions have the worst prognosis, according to an analysis of melanoma data from the National Cancer Institute’s Surveillance, Epidemiology, and End Results (SEER) program.
In 2015, a study from Queensland, Australia, reported that more patients died from T1 melanomas than did from T4 melanomas (J Invest Dermatol. 2015 Apr;135:1190-3). But a similar analysis in the United States was not available, so Dr. Shoshana Landow, a dermatologist at the Providence (R.I.) Veterans Affairs Medical Center,and her associates extracted SEER 13 data for invasive melanomas between 1992 through 2003. These registries covered Atlanta, Connecticut, Detroit, rural Georgia, Hawaii, Iowa, Los Angeles, New Mexico, San Francisco-Oakland, San Jose-Monterey, Seattle-Puget Sound, Utah, and the Alaska Native Tumor Registry, according to the SEER website.
Among 105,264 recorded melanomas during this period, 37,210 tumors were in situ, the investigators reported in a poster presented at the annual meeting of the Society for Investigative Dermatology. They excluded those cases, as well as 4,789 cases that involved more than one primary melanoma, 8,637 that extended beyond the skin at diagnosis, 5,308 with no record of thickness, and one that was not known to be primary.
That left 49,319 invasive melanomas, of which 35,509 (72%) were T1 (0.01-1.00mm), 7,879 (16%) were T2 (1.01-2.00 mm), 3,948 (8%) were T3 (2.01-4.00 mm), and 1,983 (4%) were T4 (more than 4.00 mm). A total of 17% of the T1 lesions were 0.01-0.25 mm thick at diagnosis, while 42% were 0.26-0.50 mm thick, 25% were 0.51-0.75 mm thick, and 16% were 0.76-1.00 mm thick.
Ten years after diagnosis, 3,660 (7.4%) patients had died of melanoma, according to the study. These deaths included 1,072 T1 patients, 974 T2 patients, 985 T3 patients, and 629 T4 patients. But while T4 lesions accounted for the fewest number of deaths and made up only 4% of all invasive melanomas, the 10-year mortality rate for T4 lesions was nearly 32%, vs. 25% for T3 lesions, 12% for T2 lesions, and 3% for T1 lesions.
Most (42%) T1 lesions were 0.26-0.50 mm thick, 25% of T1 lesions were 0.51-0.75 mm thick, and the remaining T1 lesions were nearly evenly split between the thinnest (0.01-0.25 mm) and the thickest (0.76-1.00mm) categories, the researchers also reported. When considering only the T1 lesions, 10-year mortality rates were 3% for the 0.01-0.25 mm category, 1.9% for the 0.26-0.5 mm category, 3.7% for the 0.51-0.75 mm category, and 5.8% for the 0.76-1.00 mm category.
Thus, the thinnest invasive melanomas had a higher 10-year death rate than did the next-thinnest category, a finding that “demands explanation” and was not caused by ulceration, the researchers noted.
“The greatest total number of deaths was from T1 melanomas and the smallest number from T4, despite the fact that prognosis worsened as melanoma thickened from T1 to T4,” they concluded in their poster. “Our findings highlight the heavy death toll of thin melanomas.”
The authors did not specify funding sources, and had no disclosures.
AT THE 2016 SID ANNUAL MEETING
Key clinical point: Thin (T1) melanomas accounted for the greatest number of deaths from melanoma in a large study.
Major finding: Among 3,660 deaths from invasive melanoma, most (1,072) were in people with T1 lesions. While T4 lesions made up only 4% of invasive melanomas, their 10-year mortality rate was nearly 32%.
Data source: A study of 49,139 invasive melanomas recorded in the NCI’s SEER database.
Disclosures: The investigators did not specify funding sources and had no disclosures.
Red Alert: Can Topical Skin Care Products Promote Melanoma Metastasis?

Le Gal et al (Sci Transl Med. 2015;7:308re8) discovered that antioxidant administration in mice not only increased lymph node metastases but also increased the migration and invasive properties of human melanoma cells. However, the antioxidant N-acetylcysteine (NAC) had no impact on the number and size of the primary tumors (in mice), and neither NAC nor Trolox (6-hydroxy-2,5,7,8-tetramethylchroman-2-carboxylic acid), a structurally unrelated antioxidant and soluble vitamin E analogue, affected the proliferation of human melanoma cells. Hence, the progression of malignant melanoma (MM), a cancer that is sensitive to changes in reduction-oxidation status, may be influenced by exposure to antioxidants and vitamin E.
What’s the issue?
Healthy individuals and oncology patients commonly use supplements containing antioxidants to prevent cancer and fight malignancy, respectively. However, animal studies and human clinical trials have shown that antioxidants increase cancer risk and accelerate the progression of primary lung tumors. Le Gal et al’s study regarding progression of melanoma metastases following exposure to antioxidants extends the observations demonstrated for lung neoplasms. N-acetylcysteine was added to the drinking water of mice, whereas NAC and Trolox were added to a panel of human MM cell lines. N-acetylcysteine increased lymph node metastases in the endogenous mouse model of MM, and both NAC and Trolox markedly increased the migrations and invasive properties of human MM cells.
Cancers may be caused or exacerbated by free radicals. It has been assumed that antioxidants may protect against malignancy by destroying free radicals. Although prior studies have concluded that antioxidants prevent healthy cells from transforming into cancer after exposure to free radicals, Le Gal et al’s research suggests that antioxidants may not only protect but also enhance tumor progression once a cancer has developed.
If one extends the results of animal and tissue culture studies to humans, exposure to antioxidants may potentially influence the course of metastatic disease in patients who have already developed melanoma. In addition to systemic exposure after receiving oral antioxidants, melanoma patients also can be topically exposed to antioxidants. For example, nonprescription skin care products such as cutaneous rejuvenation treatments, emollients, and sunscreens can contain β-carotene, vitamin E, and other antioxidants. It remains to be determined whether topical exposure to antioxidants can cause the same observations that have occurred following systemic absorption in mice or tissue culture studies in human cell lines. Should we caution our melanoma patients with regards to what they apply to their skin?

Le Gal et al (Sci Transl Med. 2015;7:308re8) discovered that antioxidant administration in mice not only increased lymph node metastases but also increased the migration and invasive properties of human melanoma cells. However, the antioxidant N-acetylcysteine (NAC) had no impact on the number and size of the primary tumors (in mice), and neither NAC nor Trolox (6-hydroxy-2,5,7,8-tetramethylchroman-2-carboxylic acid), a structurally unrelated antioxidant and soluble vitamin E analogue, affected the proliferation of human melanoma cells. Hence, the progression of malignant melanoma (MM), a cancer that is sensitive to changes in reduction-oxidation status, may be influenced by exposure to antioxidants and vitamin E.
What’s the issue?
Healthy individuals and oncology patients commonly use supplements containing antioxidants to prevent cancer and fight malignancy, respectively. However, animal studies and human clinical trials have shown that antioxidants increase cancer risk and accelerate the progression of primary lung tumors. Le Gal et al’s study regarding progression of melanoma metastases following exposure to antioxidants extends the observations demonstrated for lung neoplasms. N-acetylcysteine was added to the drinking water of mice, whereas NAC and Trolox were added to a panel of human MM cell lines. N-acetylcysteine increased lymph node metastases in the endogenous mouse model of MM, and both NAC and Trolox markedly increased the migrations and invasive properties of human MM cells.
Cancers may be caused or exacerbated by free radicals. It has been assumed that antioxidants may protect against malignancy by destroying free radicals. Although prior studies have concluded that antioxidants prevent healthy cells from transforming into cancer after exposure to free radicals, Le Gal et al’s research suggests that antioxidants may not only protect but also enhance tumor progression once a cancer has developed.
If one extends the results of animal and tissue culture studies to humans, exposure to antioxidants may potentially influence the course of metastatic disease in patients who have already developed melanoma. In addition to systemic exposure after receiving oral antioxidants, melanoma patients also can be topically exposed to antioxidants. For example, nonprescription skin care products such as cutaneous rejuvenation treatments, emollients, and sunscreens can contain β-carotene, vitamin E, and other antioxidants. It remains to be determined whether topical exposure to antioxidants can cause the same observations that have occurred following systemic absorption in mice or tissue culture studies in human cell lines. Should we caution our melanoma patients with regards to what they apply to their skin?

Le Gal et al (Sci Transl Med. 2015;7:308re8) discovered that antioxidant administration in mice not only increased lymph node metastases but also increased the migration and invasive properties of human melanoma cells. However, the antioxidant N-acetylcysteine (NAC) had no impact on the number and size of the primary tumors (in mice), and neither NAC nor Trolox (6-hydroxy-2,5,7,8-tetramethylchroman-2-carboxylic acid), a structurally unrelated antioxidant and soluble vitamin E analogue, affected the proliferation of human melanoma cells. Hence, the progression of malignant melanoma (MM), a cancer that is sensitive to changes in reduction-oxidation status, may be influenced by exposure to antioxidants and vitamin E.
What’s the issue?
Healthy individuals and oncology patients commonly use supplements containing antioxidants to prevent cancer and fight malignancy, respectively. However, animal studies and human clinical trials have shown that antioxidants increase cancer risk and accelerate the progression of primary lung tumors. Le Gal et al’s study regarding progression of melanoma metastases following exposure to antioxidants extends the observations demonstrated for lung neoplasms. N-acetylcysteine was added to the drinking water of mice, whereas NAC and Trolox were added to a panel of human MM cell lines. N-acetylcysteine increased lymph node metastases in the endogenous mouse model of MM, and both NAC and Trolox markedly increased the migrations and invasive properties of human MM cells.
Cancers may be caused or exacerbated by free radicals. It has been assumed that antioxidants may protect against malignancy by destroying free radicals. Although prior studies have concluded that antioxidants prevent healthy cells from transforming into cancer after exposure to free radicals, Le Gal et al’s research suggests that antioxidants may not only protect but also enhance tumor progression once a cancer has developed.
If one extends the results of animal and tissue culture studies to humans, exposure to antioxidants may potentially influence the course of metastatic disease in patients who have already developed melanoma. In addition to systemic exposure after receiving oral antioxidants, melanoma patients also can be topically exposed to antioxidants. For example, nonprescription skin care products such as cutaneous rejuvenation treatments, emollients, and sunscreens can contain β-carotene, vitamin E, and other antioxidants. It remains to be determined whether topical exposure to antioxidants can cause the same observations that have occurred following systemic absorption in mice or tissue culture studies in human cell lines. Should we caution our melanoma patients with regards to what they apply to their skin?
Immune agonist, checkpoint inhibitor combo shows good tolerability
CHICAGO – Combining two immunotherapies, one inhibiting immune suppression and the other stimulating immune activation, is well tolerated and shows activity for a variety of solid tumor types, according to a phase I trial presented at the annual meeting of the American Association of Clinical Oncology.
Investigators enrolled 51 patients with locally advanced or metastatic solid tumors of any type after progression on standard therapy to a phase Ib dose-escalation study using atezolizumab, a monoclonal antibody checkpoint inhibitor that targets PD-L1, in combination with MOXR0916 (MOXR), an agonist IgG1 monoclonal antibody targeting OX40, a costimulatory receptor. Atezolizumab received Food and Drug Administration approval in May 2016 for use in certain patients with urothelial carcinoma. There were 28 patients in a dose-escalation cohort of the study and 23 in a serial biopsy cohort. The dose of the drug combination was started at 12 mg and escalated to understand pharmacodynamic changes in the tumors.
“The pharmacokinetics of both MOXR0916 and atezolizumab were similar to their single-agent data, suggesting no interaction,” reported Dr. Jeffrey Infante of the Sarah Cannon Research Institute in Nashville, Tenn.
The drug combination was well tolerated through the entire escalation range of MOXR. There were no dose-limiting toxicities, and no maximal tolerated dose was reached. There were also no drug-related deaths or grade 4 toxicities or drug-related treatment discontinuations. One case of grade 3 pneumonitis, successfully managed with methylprednisolone and antibiotics, occurred at the MOXR 40-mg dose on cycle 4 of treatment in a patient with non–small-cell lung cancer, he said.
About half the patients (53%) experienced any form of adverse event on the drug combination, and only 8% were grade 2 or 3. There were very few adverse events of any one type, and they did not appear to cluster among patients on the higher MOXR doses. The most prevalent adverse events were nausea, fever, fatigue, and rash, and each was in the 8%-14% range and almost always grade 1.
Many patients showed efficacy of the regimens out to 6-7 cycles regardless of tumor type, and 8 of the 51 patients were still receiving the therapy past cycle 7 with partial responses.
The stimulatory molecule OX40 is not normally expressed on T cells, but it is expressed when antigen interacts with the T-cell receptor, and it can then interact with its ligand, OX40L. The result is production of inflammatory cytokines such as gamma-interferon, activation and survival of effector T cells, and production of memory T cells. At the same time, OX40 activity blocks the suppressive function of regulatory T cells.
“So a molecule that can be a cancer therapeutic such as an OX40 agonist has dual mechanisms of action,” Dr. Infante said. “It can costimulate effector T cells and at the same time inhibit regulatory T cells. Furthermore, there is a reduced risk of toxicity, potentially, as its activity is linked to antigen recognition.”
There is good rationale for using an OX40 agonist such as MOXR, either for its immune stimulatory function or to deactivate immune suppression by regulatory T cells, or both, said discussant Dr. Jedd Wolchok, chief, melanoma and immunotherapeutics service, Memorial Sloan-Kettering Cancer Center, New York. Dr. Infante’s dose-escalation study was “very nicely designed and showed quite good safety,” Dr. Wolchok said, though one thing he would have liked to have seen was a quantification of regulatory T cells in tumor biopsies.
“This [study] is very important considering that this is an agonist antibody, and the agonist agents need to be dosed very deliberatively, as was done here, to ensure safety of patients,” Dr. Wolchok said, adding that further research needs to target “optimal combinatorial partners” and explore other mechanistic biomarkers.
MOXR was given in this trial at escalating doses on a 3+3 design (0.8-1,200 mg) on the same day as atezolizumab 1,200 mg IV once every 3 weeks with a 21-day window for assessment of MOXR dose-limiting toxicities. MOXR doses of 300 mg maintained trough concentrations sufficient to saturate OX40 receptors. An expansion regimen using 300 mg MOXR with atezolizumab 1,200 mg every 3 weeks is underway and will assess efficacy in the treatment of melanoma, renal cell carcinoma, non–small-cell lung cancer, urothelial carcinoma, and triple-negative breast cancer.
The study was sponsored by Roche. Dr. Infante reported having no relevant financial disclosures. Dr. Wolchok owns stock in Potenza Therapeutics and Vesuvius Pharmaceuticals, has received travel expenses and/or has an advisory role with several other companies, and is a coinventor on an issued patent for DNA vaccines for the treatment of cancer in companion animals.
CHICAGO – Combining two immunotherapies, one inhibiting immune suppression and the other stimulating immune activation, is well tolerated and shows activity for a variety of solid tumor types, according to a phase I trial presented at the annual meeting of the American Association of Clinical Oncology.
Investigators enrolled 51 patients with locally advanced or metastatic solid tumors of any type after progression on standard therapy to a phase Ib dose-escalation study using atezolizumab, a monoclonal antibody checkpoint inhibitor that targets PD-L1, in combination with MOXR0916 (MOXR), an agonist IgG1 monoclonal antibody targeting OX40, a costimulatory receptor. Atezolizumab received Food and Drug Administration approval in May 2016 for use in certain patients with urothelial carcinoma. There were 28 patients in a dose-escalation cohort of the study and 23 in a serial biopsy cohort. The dose of the drug combination was started at 12 mg and escalated to understand pharmacodynamic changes in the tumors.
“The pharmacokinetics of both MOXR0916 and atezolizumab were similar to their single-agent data, suggesting no interaction,” reported Dr. Jeffrey Infante of the Sarah Cannon Research Institute in Nashville, Tenn.
The drug combination was well tolerated through the entire escalation range of MOXR. There were no dose-limiting toxicities, and no maximal tolerated dose was reached. There were also no drug-related deaths or grade 4 toxicities or drug-related treatment discontinuations. One case of grade 3 pneumonitis, successfully managed with methylprednisolone and antibiotics, occurred at the MOXR 40-mg dose on cycle 4 of treatment in a patient with non–small-cell lung cancer, he said.
About half the patients (53%) experienced any form of adverse event on the drug combination, and only 8% were grade 2 or 3. There were very few adverse events of any one type, and they did not appear to cluster among patients on the higher MOXR doses. The most prevalent adverse events were nausea, fever, fatigue, and rash, and each was in the 8%-14% range and almost always grade 1.
Many patients showed efficacy of the regimens out to 6-7 cycles regardless of tumor type, and 8 of the 51 patients were still receiving the therapy past cycle 7 with partial responses.
The stimulatory molecule OX40 is not normally expressed on T cells, but it is expressed when antigen interacts with the T-cell receptor, and it can then interact with its ligand, OX40L. The result is production of inflammatory cytokines such as gamma-interferon, activation and survival of effector T cells, and production of memory T cells. At the same time, OX40 activity blocks the suppressive function of regulatory T cells.
“So a molecule that can be a cancer therapeutic such as an OX40 agonist has dual mechanisms of action,” Dr. Infante said. “It can costimulate effector T cells and at the same time inhibit regulatory T cells. Furthermore, there is a reduced risk of toxicity, potentially, as its activity is linked to antigen recognition.”
There is good rationale for using an OX40 agonist such as MOXR, either for its immune stimulatory function or to deactivate immune suppression by regulatory T cells, or both, said discussant Dr. Jedd Wolchok, chief, melanoma and immunotherapeutics service, Memorial Sloan-Kettering Cancer Center, New York. Dr. Infante’s dose-escalation study was “very nicely designed and showed quite good safety,” Dr. Wolchok said, though one thing he would have liked to have seen was a quantification of regulatory T cells in tumor biopsies.
“This [study] is very important considering that this is an agonist antibody, and the agonist agents need to be dosed very deliberatively, as was done here, to ensure safety of patients,” Dr. Wolchok said, adding that further research needs to target “optimal combinatorial partners” and explore other mechanistic biomarkers.
MOXR was given in this trial at escalating doses on a 3+3 design (0.8-1,200 mg) on the same day as atezolizumab 1,200 mg IV once every 3 weeks with a 21-day window for assessment of MOXR dose-limiting toxicities. MOXR doses of 300 mg maintained trough concentrations sufficient to saturate OX40 receptors. An expansion regimen using 300 mg MOXR with atezolizumab 1,200 mg every 3 weeks is underway and will assess efficacy in the treatment of melanoma, renal cell carcinoma, non–small-cell lung cancer, urothelial carcinoma, and triple-negative breast cancer.
The study was sponsored by Roche. Dr. Infante reported having no relevant financial disclosures. Dr. Wolchok owns stock in Potenza Therapeutics and Vesuvius Pharmaceuticals, has received travel expenses and/or has an advisory role with several other companies, and is a coinventor on an issued patent for DNA vaccines for the treatment of cancer in companion animals.
CHICAGO – Combining two immunotherapies, one inhibiting immune suppression and the other stimulating immune activation, is well tolerated and shows activity for a variety of solid tumor types, according to a phase I trial presented at the annual meeting of the American Association of Clinical Oncology.
Investigators enrolled 51 patients with locally advanced or metastatic solid tumors of any type after progression on standard therapy to a phase Ib dose-escalation study using atezolizumab, a monoclonal antibody checkpoint inhibitor that targets PD-L1, in combination with MOXR0916 (MOXR), an agonist IgG1 monoclonal antibody targeting OX40, a costimulatory receptor. Atezolizumab received Food and Drug Administration approval in May 2016 for use in certain patients with urothelial carcinoma. There were 28 patients in a dose-escalation cohort of the study and 23 in a serial biopsy cohort. The dose of the drug combination was started at 12 mg and escalated to understand pharmacodynamic changes in the tumors.
“The pharmacokinetics of both MOXR0916 and atezolizumab were similar to their single-agent data, suggesting no interaction,” reported Dr. Jeffrey Infante of the Sarah Cannon Research Institute in Nashville, Tenn.
The drug combination was well tolerated through the entire escalation range of MOXR. There were no dose-limiting toxicities, and no maximal tolerated dose was reached. There were also no drug-related deaths or grade 4 toxicities or drug-related treatment discontinuations. One case of grade 3 pneumonitis, successfully managed with methylprednisolone and antibiotics, occurred at the MOXR 40-mg dose on cycle 4 of treatment in a patient with non–small-cell lung cancer, he said.
About half the patients (53%) experienced any form of adverse event on the drug combination, and only 8% were grade 2 or 3. There were very few adverse events of any one type, and they did not appear to cluster among patients on the higher MOXR doses. The most prevalent adverse events were nausea, fever, fatigue, and rash, and each was in the 8%-14% range and almost always grade 1.
Many patients showed efficacy of the regimens out to 6-7 cycles regardless of tumor type, and 8 of the 51 patients were still receiving the therapy past cycle 7 with partial responses.
The stimulatory molecule OX40 is not normally expressed on T cells, but it is expressed when antigen interacts with the T-cell receptor, and it can then interact with its ligand, OX40L. The result is production of inflammatory cytokines such as gamma-interferon, activation and survival of effector T cells, and production of memory T cells. At the same time, OX40 activity blocks the suppressive function of regulatory T cells.
“So a molecule that can be a cancer therapeutic such as an OX40 agonist has dual mechanisms of action,” Dr. Infante said. “It can costimulate effector T cells and at the same time inhibit regulatory T cells. Furthermore, there is a reduced risk of toxicity, potentially, as its activity is linked to antigen recognition.”
There is good rationale for using an OX40 agonist such as MOXR, either for its immune stimulatory function or to deactivate immune suppression by regulatory T cells, or both, said discussant Dr. Jedd Wolchok, chief, melanoma and immunotherapeutics service, Memorial Sloan-Kettering Cancer Center, New York. Dr. Infante’s dose-escalation study was “very nicely designed and showed quite good safety,” Dr. Wolchok said, though one thing he would have liked to have seen was a quantification of regulatory T cells in tumor biopsies.
“This [study] is very important considering that this is an agonist antibody, and the agonist agents need to be dosed very deliberatively, as was done here, to ensure safety of patients,” Dr. Wolchok said, adding that further research needs to target “optimal combinatorial partners” and explore other mechanistic biomarkers.
MOXR was given in this trial at escalating doses on a 3+3 design (0.8-1,200 mg) on the same day as atezolizumab 1,200 mg IV once every 3 weeks with a 21-day window for assessment of MOXR dose-limiting toxicities. MOXR doses of 300 mg maintained trough concentrations sufficient to saturate OX40 receptors. An expansion regimen using 300 mg MOXR with atezolizumab 1,200 mg every 3 weeks is underway and will assess efficacy in the treatment of melanoma, renal cell carcinoma, non–small-cell lung cancer, urothelial carcinoma, and triple-negative breast cancer.
The study was sponsored by Roche. Dr. Infante reported having no relevant financial disclosures. Dr. Wolchok owns stock in Potenza Therapeutics and Vesuvius Pharmaceuticals, has received travel expenses and/or has an advisory role with several other companies, and is a coinventor on an issued patent for DNA vaccines for the treatment of cancer in companion animals.
AT THE 2016 ASCO ANNUAL MEETING
Key clinical point: Combining an immune agonist and a checkpoint inhibitor shows good tolerability.
Major finding: Eighty-five percent of adverse effects were grade 1; the rest were grade 2/3.
Data source: A phase Ib, open-label multicenter study of 51 patients.
Disclosures: The study was sponsored by Roche. Dr. Infante reported having no relevant financial disclosures. Dr. Wolchok owns stock in Potenza Therapeutics and Vesuvius Pharmaceuticals, has received travel expenses and/or has an advisory role with several other companies, and is a coinventor on an issued patent for DNA vaccines for the treatment of cancer in companion animals.
MyPathway: Targeted therapies show promise in nonindicated tumors
CHICAGO – Agents that target the HER2, BRAF, Hedgehog, or EGFR pathways show promise in nonindicated tumor types that harbor these molecular alterations, according to early findings from the MyPathway study.
Of 129 patients enrolled in the multicenter, open-label, phase IIa study, 29 had a major response, defined as tumor shrinkage of at least 30%, to such treatment. One of those patients had a complete response, and 28 had a partial response. An additional 40 patients had stable disease on treatment. Fourteen of the 29 patients progressed after a median of 6 months’ follow-up, and 15 responses were ongoing at up to 11 months, Dr. John D. Hainsworth reported at the annual meeting of the American Society of Clinical Oncology.
No new safety signals were observed, said Dr. Hainsworth of Sarah Cannon Research Institute in Nashville, Tenn.
Treatments evaluated in MyPathway included:
• Trastuzumab + pertuzumab, which targets the HER2 pathway and is currently indicated for breast cancer.
• Vemurafenib, which targets the BRAF pathway and is currently indicated for melanoma.
• Vismodegib, which targets the Hedgehog pathway and is currently indicated for basal cell carcinoma of the skin.
• Erlotinib, which targets the EGFR pathway and is indicated for non–small-cell lung cancer.
Responses have been seen with all four of the treatments, but the best responses were seen among patients with HER2 and BRAF abnormalities.
Among 61 cancers with HER2 amplification/overexpression, trastuzumab + pertuzumab provided a benefit for colorectal, bladder, biliary, non–small-cell lung, pancreas, and head/neck cancers.
Of 20 colorectal tumors, 7 (35%) showed complete or partial response, and 3 (15%) remained stable for at least 120 days (clinical benefit rate, 50%). Complete/partial responses and stable disease, respectively, were also seen in three and two of eight bladder tumors (clinical benefit rate, 63%), in three and three of six biliary tumors (clinical benefit rate, 100%), in two and zero of seven non–small-cell lung tumors (clinical benefit rate, 29%), one and zero of six pancreas tumors (clinical benefit rate, 17%), and one and zero of three head and neck tumors (34%). One of 11 other types of tumors showed disease stability at 120 days (clinical benefit rate, 9%). The overall clinical benefit rate in the study was 43%, Dr. Hainsworth said.
Among 33 cancers with the BRAF mutation, vemurafenib showed activity for non–small-cell lung, ovary, unknown primary, colorectal, pancreas, and head/neck tumors. Of 15 non–small-cell lung tumors, 3 (20%) showed complete or partial responses and 2 (13%) remained stable for at least 120 days (clinical benefit rate, 33%). Complete/partial responses and stable disease, respectively, were also seen in one and two of four ovary tumors (clinical benefit rate, 75%), and complete or partial responses were seen in one each of three unknown primary tumors, two colorectal tumors, two pancreas tumors, and one head/neck tumor (clinical benefit rates of 33%, 50%, 50%, and 100%, respectively). No benefit was seen with tumors at other sites (total clinical benefit rate, 36%), Dr. Hainsworth said.
“Of interest in this group [of patients with BRAF mutations], seven of the eight responses were in V600E mutations, and as you know, that’s the mutation that’s been specifically correlated with high response to BRAF inhibition in melanoma where this treatment is now approved,” he said, adding that the response rate in those patients was 38%.
Based on these early results, enrollment of patients with HER2 abnormalities and colorectal, bladder, or biliary cancer, and of patients with BRAF mutations and lung cancer, will be expanded, he said.
Subjects enrolled in MyPathway have advanced cancer showing abnormalities in any of the pathways of interest. The first 129 received a mean of three prior therapies, and in the 29 who responded, 12 different types of cancer responded to the targeted treatment.
“An increasing number of targeted agents for advanced cancer are in use now based on the presence of molecular abnormalities in the cancer. … We’ve known that the same mutations that are in those cancers are found in a wide variety of other cancers, although at a lower incidence, and it’s been difficult to test how effective these same treatments are for the other cancers due to the difficulty in identifying the patient population,” he said, explaining that an increase in comprehensive genomic profiling in recent years has allowed for identification of more and more of these mutations in other cancers.
“I think we’ve shown now that this trial design is feasible, where patients are selected on the basis of molecular abnormalities in their cancers rather than on their primary tumor type or primary site, and certainly offers opportunities for patients with these molecular abnormalities,” Dr. Hainsworth concluded.
Thus far, MyPathway has enrolled more than 200 patients, and is designed to accrue up to 500, with adjustment of treatment groups based on response rates. Emerging new regimens that target these pathways, such as the MEK inhibitor cobemetinib, will also be added, as will new agents targeting additional molecular abnormalities.
The study design, using this “tumor-agnostic approach,” mirrors that of the ASCO-led TAPUR trial, according to ASCO spokesperson Dr. Sumanta Kumar Pal.
The findings of these and other precision medicine trials may ultimately shift the longstanding cancer treatment paradigm, Dr. Pal said.
MyPathway received funding from Genentech. Dr. Hainsworth reported that his institution has received research funding from Astellas Pharma, AstraZeneca, Celgene, Genentech, Johnson & Johnson, Lilly, and Novartis.
CHICAGO – Agents that target the HER2, BRAF, Hedgehog, or EGFR pathways show promise in nonindicated tumor types that harbor these molecular alterations, according to early findings from the MyPathway study.
Of 129 patients enrolled in the multicenter, open-label, phase IIa study, 29 had a major response, defined as tumor shrinkage of at least 30%, to such treatment. One of those patients had a complete response, and 28 had a partial response. An additional 40 patients had stable disease on treatment. Fourteen of the 29 patients progressed after a median of 6 months’ follow-up, and 15 responses were ongoing at up to 11 months, Dr. John D. Hainsworth reported at the annual meeting of the American Society of Clinical Oncology.
No new safety signals were observed, said Dr. Hainsworth of Sarah Cannon Research Institute in Nashville, Tenn.
Treatments evaluated in MyPathway included:
• Trastuzumab + pertuzumab, which targets the HER2 pathway and is currently indicated for breast cancer.
• Vemurafenib, which targets the BRAF pathway and is currently indicated for melanoma.
• Vismodegib, which targets the Hedgehog pathway and is currently indicated for basal cell carcinoma of the skin.
• Erlotinib, which targets the EGFR pathway and is indicated for non–small-cell lung cancer.
Responses have been seen with all four of the treatments, but the best responses were seen among patients with HER2 and BRAF abnormalities.
Among 61 cancers with HER2 amplification/overexpression, trastuzumab + pertuzumab provided a benefit for colorectal, bladder, biliary, non–small-cell lung, pancreas, and head/neck cancers.
Of 20 colorectal tumors, 7 (35%) showed complete or partial response, and 3 (15%) remained stable for at least 120 days (clinical benefit rate, 50%). Complete/partial responses and stable disease, respectively, were also seen in three and two of eight bladder tumors (clinical benefit rate, 63%), in three and three of six biliary tumors (clinical benefit rate, 100%), in two and zero of seven non–small-cell lung tumors (clinical benefit rate, 29%), one and zero of six pancreas tumors (clinical benefit rate, 17%), and one and zero of three head and neck tumors (34%). One of 11 other types of tumors showed disease stability at 120 days (clinical benefit rate, 9%). The overall clinical benefit rate in the study was 43%, Dr. Hainsworth said.
Among 33 cancers with the BRAF mutation, vemurafenib showed activity for non–small-cell lung, ovary, unknown primary, colorectal, pancreas, and head/neck tumors. Of 15 non–small-cell lung tumors, 3 (20%) showed complete or partial responses and 2 (13%) remained stable for at least 120 days (clinical benefit rate, 33%). Complete/partial responses and stable disease, respectively, were also seen in one and two of four ovary tumors (clinical benefit rate, 75%), and complete or partial responses were seen in one each of three unknown primary tumors, two colorectal tumors, two pancreas tumors, and one head/neck tumor (clinical benefit rates of 33%, 50%, 50%, and 100%, respectively). No benefit was seen with tumors at other sites (total clinical benefit rate, 36%), Dr. Hainsworth said.
“Of interest in this group [of patients with BRAF mutations], seven of the eight responses were in V600E mutations, and as you know, that’s the mutation that’s been specifically correlated with high response to BRAF inhibition in melanoma where this treatment is now approved,” he said, adding that the response rate in those patients was 38%.
Based on these early results, enrollment of patients with HER2 abnormalities and colorectal, bladder, or biliary cancer, and of patients with BRAF mutations and lung cancer, will be expanded, he said.
Subjects enrolled in MyPathway have advanced cancer showing abnormalities in any of the pathways of interest. The first 129 received a mean of three prior therapies, and in the 29 who responded, 12 different types of cancer responded to the targeted treatment.
“An increasing number of targeted agents for advanced cancer are in use now based on the presence of molecular abnormalities in the cancer. … We’ve known that the same mutations that are in those cancers are found in a wide variety of other cancers, although at a lower incidence, and it’s been difficult to test how effective these same treatments are for the other cancers due to the difficulty in identifying the patient population,” he said, explaining that an increase in comprehensive genomic profiling in recent years has allowed for identification of more and more of these mutations in other cancers.
“I think we’ve shown now that this trial design is feasible, where patients are selected on the basis of molecular abnormalities in their cancers rather than on their primary tumor type or primary site, and certainly offers opportunities for patients with these molecular abnormalities,” Dr. Hainsworth concluded.
Thus far, MyPathway has enrolled more than 200 patients, and is designed to accrue up to 500, with adjustment of treatment groups based on response rates. Emerging new regimens that target these pathways, such as the MEK inhibitor cobemetinib, will also be added, as will new agents targeting additional molecular abnormalities.
The study design, using this “tumor-agnostic approach,” mirrors that of the ASCO-led TAPUR trial, according to ASCO spokesperson Dr. Sumanta Kumar Pal.
The findings of these and other precision medicine trials may ultimately shift the longstanding cancer treatment paradigm, Dr. Pal said.
MyPathway received funding from Genentech. Dr. Hainsworth reported that his institution has received research funding from Astellas Pharma, AstraZeneca, Celgene, Genentech, Johnson & Johnson, Lilly, and Novartis.
CHICAGO – Agents that target the HER2, BRAF, Hedgehog, or EGFR pathways show promise in nonindicated tumor types that harbor these molecular alterations, according to early findings from the MyPathway study.
Of 129 patients enrolled in the multicenter, open-label, phase IIa study, 29 had a major response, defined as tumor shrinkage of at least 30%, to such treatment. One of those patients had a complete response, and 28 had a partial response. An additional 40 patients had stable disease on treatment. Fourteen of the 29 patients progressed after a median of 6 months’ follow-up, and 15 responses were ongoing at up to 11 months, Dr. John D. Hainsworth reported at the annual meeting of the American Society of Clinical Oncology.
No new safety signals were observed, said Dr. Hainsworth of Sarah Cannon Research Institute in Nashville, Tenn.
Treatments evaluated in MyPathway included:
• Trastuzumab + pertuzumab, which targets the HER2 pathway and is currently indicated for breast cancer.
• Vemurafenib, which targets the BRAF pathway and is currently indicated for melanoma.
• Vismodegib, which targets the Hedgehog pathway and is currently indicated for basal cell carcinoma of the skin.
• Erlotinib, which targets the EGFR pathway and is indicated for non–small-cell lung cancer.
Responses have been seen with all four of the treatments, but the best responses were seen among patients with HER2 and BRAF abnormalities.
Among 61 cancers with HER2 amplification/overexpression, trastuzumab + pertuzumab provided a benefit for colorectal, bladder, biliary, non–small-cell lung, pancreas, and head/neck cancers.
Of 20 colorectal tumors, 7 (35%) showed complete or partial response, and 3 (15%) remained stable for at least 120 days (clinical benefit rate, 50%). Complete/partial responses and stable disease, respectively, were also seen in three and two of eight bladder tumors (clinical benefit rate, 63%), in three and three of six biliary tumors (clinical benefit rate, 100%), in two and zero of seven non–small-cell lung tumors (clinical benefit rate, 29%), one and zero of six pancreas tumors (clinical benefit rate, 17%), and one and zero of three head and neck tumors (34%). One of 11 other types of tumors showed disease stability at 120 days (clinical benefit rate, 9%). The overall clinical benefit rate in the study was 43%, Dr. Hainsworth said.
Among 33 cancers with the BRAF mutation, vemurafenib showed activity for non–small-cell lung, ovary, unknown primary, colorectal, pancreas, and head/neck tumors. Of 15 non–small-cell lung tumors, 3 (20%) showed complete or partial responses and 2 (13%) remained stable for at least 120 days (clinical benefit rate, 33%). Complete/partial responses and stable disease, respectively, were also seen in one and two of four ovary tumors (clinical benefit rate, 75%), and complete or partial responses were seen in one each of three unknown primary tumors, two colorectal tumors, two pancreas tumors, and one head/neck tumor (clinical benefit rates of 33%, 50%, 50%, and 100%, respectively). No benefit was seen with tumors at other sites (total clinical benefit rate, 36%), Dr. Hainsworth said.
“Of interest in this group [of patients with BRAF mutations], seven of the eight responses were in V600E mutations, and as you know, that’s the mutation that’s been specifically correlated with high response to BRAF inhibition in melanoma where this treatment is now approved,” he said, adding that the response rate in those patients was 38%.
Based on these early results, enrollment of patients with HER2 abnormalities and colorectal, bladder, or biliary cancer, and of patients with BRAF mutations and lung cancer, will be expanded, he said.
Subjects enrolled in MyPathway have advanced cancer showing abnormalities in any of the pathways of interest. The first 129 received a mean of three prior therapies, and in the 29 who responded, 12 different types of cancer responded to the targeted treatment.
“An increasing number of targeted agents for advanced cancer are in use now based on the presence of molecular abnormalities in the cancer. … We’ve known that the same mutations that are in those cancers are found in a wide variety of other cancers, although at a lower incidence, and it’s been difficult to test how effective these same treatments are for the other cancers due to the difficulty in identifying the patient population,” he said, explaining that an increase in comprehensive genomic profiling in recent years has allowed for identification of more and more of these mutations in other cancers.
“I think we’ve shown now that this trial design is feasible, where patients are selected on the basis of molecular abnormalities in their cancers rather than on their primary tumor type or primary site, and certainly offers opportunities for patients with these molecular abnormalities,” Dr. Hainsworth concluded.
Thus far, MyPathway has enrolled more than 200 patients, and is designed to accrue up to 500, with adjustment of treatment groups based on response rates. Emerging new regimens that target these pathways, such as the MEK inhibitor cobemetinib, will also be added, as will new agents targeting additional molecular abnormalities.
The study design, using this “tumor-agnostic approach,” mirrors that of the ASCO-led TAPUR trial, according to ASCO spokesperson Dr. Sumanta Kumar Pal.
The findings of these and other precision medicine trials may ultimately shift the longstanding cancer treatment paradigm, Dr. Pal said.
MyPathway received funding from Genentech. Dr. Hainsworth reported that his institution has received research funding from Astellas Pharma, AstraZeneca, Celgene, Genentech, Johnson & Johnson, Lilly, and Novartis.
AT THE 2016 ASCO ANNUAL MEETING
Key clinical point: Agents that target the HER2, BRAF, Hedgehog, or EGFR pathways show promise in nonindicated tumor types that harbor these molecular alterations, according to early findings from the MyPathway study.
Major finding: Twenty-nine patients had a major response, and an additional 40 remained stable on treatment.
Data source: The ongoing open-label, phase IIa MyPathway study, including results from the first 129 patients.
Disclosures: MyPathway received funding from Genentech. Dr. Hainsworth reported that his institution has received research funding from Astellas Pharma, AstraZeneca, Celgene, Genentech, Johnson & Johnson, Lilly, and Novartis.
Tips and Tools for Melanoma Diagnosis

What does your patient need to know at the first visit? Does it apply to all patients?
All patients should have a total-body skin examination at least once per year; however, the frequency may change based on a prior history of melanoma or skin cancer, number of nevi or dysplastic nevi, and a family history of melanoma.
Patients should be completely undressed, and all nail polish or artificial nails should be removed prior to the examination. A complete cutaneous examination involves inspecting all skin surfaces, scalp, ocular and oral mucosa, fingernails/toenails, and genitalia if the patient agrees. Melanoma can occur in non–UV-exposed areas and the patient should be educated. Explain the ABCDEs of melanoma diagnosis to all patients and discuss concerns of any new or changing lesions, pigmented or not.
The patient should be made aware that a series of digital images will be taken for any suspicious lesions for possible short-term monitoring. The patient also may be offered full-body photography or 3D body imaging if the number of nevi warrants it.
Different patient populations have different risks for melanoma. Although melanoma predominately afflicts patients with a light skin type, there are certain types of melanoma, such as acral melanoma, that can be more common in darker skin types.
If a patient has a history of cutaneous melanoma, then the site should be checked for any local recurrence as well as palpation of the draining lymph nodes and regional lymph nodes.
I also let patients know that I will be using tools such as dermoscopy and/or reflectance confocal microscopy to better diagnose equivocal lesions before pursuing a biopsy. A biopsy may be done if there is a level of suspicion for atypia.
The use of dermoscopy, digital imaging, and reflectance confocal microscopy has changed the way we can detect, monitor, and evaluate atypical nevi. These tools can augment practice and possibly cut down on the rate of biopsies. They also are great for equivocal lesions or lesions that are in cosmetically sensitive areas. I use these tools in my everyday practice.
How do you keep patients compliant?
Empowering patients to perform self-examinations as well as examinations with his/her partner may help to reinforce monitoring by a dermatologist.
Provide patients with reading materials on self-examination while they wait in the office for your examination.
What do you do if they refuse treatment?
If patients defer a full-body skin examination, then I try to educate them about risks for UV exposure and the risk factors for both melanoma and nonmelanoma skin cancer. I also provide information on self-examinations so they can check at home for any irregularly shaped or changing moles.
What resources do you recommend to patients for more information?
It is important for patients to understand the risk factors for melanoma and the long-term prognosis of melanoma. I direct them to the American Academy of Dermatology’s website (http://www.AAD.org) for education and background about melanoma. Also, the Skin Cancer Foundation has inspiring patient stories (http://www.SkinCancer.org).

What does your patient need to know at the first visit? Does it apply to all patients?
All patients should have a total-body skin examination at least once per year; however, the frequency may change based on a prior history of melanoma or skin cancer, number of nevi or dysplastic nevi, and a family history of melanoma.
Patients should be completely undressed, and all nail polish or artificial nails should be removed prior to the examination. A complete cutaneous examination involves inspecting all skin surfaces, scalp, ocular and oral mucosa, fingernails/toenails, and genitalia if the patient agrees. Melanoma can occur in non–UV-exposed areas and the patient should be educated. Explain the ABCDEs of melanoma diagnosis to all patients and discuss concerns of any new or changing lesions, pigmented or not.
The patient should be made aware that a series of digital images will be taken for any suspicious lesions for possible short-term monitoring. The patient also may be offered full-body photography or 3D body imaging if the number of nevi warrants it.
Different patient populations have different risks for melanoma. Although melanoma predominately afflicts patients with a light skin type, there are certain types of melanoma, such as acral melanoma, that can be more common in darker skin types.
If a patient has a history of cutaneous melanoma, then the site should be checked for any local recurrence as well as palpation of the draining lymph nodes and regional lymph nodes.
I also let patients know that I will be using tools such as dermoscopy and/or reflectance confocal microscopy to better diagnose equivocal lesions before pursuing a biopsy. A biopsy may be done if there is a level of suspicion for atypia.
The use of dermoscopy, digital imaging, and reflectance confocal microscopy has changed the way we can detect, monitor, and evaluate atypical nevi. These tools can augment practice and possibly cut down on the rate of biopsies. They also are great for equivocal lesions or lesions that are in cosmetically sensitive areas. I use these tools in my everyday practice.
How do you keep patients compliant?
Empowering patients to perform self-examinations as well as examinations with his/her partner may help to reinforce monitoring by a dermatologist.
Provide patients with reading materials on self-examination while they wait in the office for your examination.
What do you do if they refuse treatment?
If patients defer a full-body skin examination, then I try to educate them about risks for UV exposure and the risk factors for both melanoma and nonmelanoma skin cancer. I also provide information on self-examinations so they can check at home for any irregularly shaped or changing moles.
What resources do you recommend to patients for more information?
It is important for patients to understand the risk factors for melanoma and the long-term prognosis of melanoma. I direct them to the American Academy of Dermatology’s website (http://www.AAD.org) for education and background about melanoma. Also, the Skin Cancer Foundation has inspiring patient stories (http://www.SkinCancer.org).

What does your patient need to know at the first visit? Does it apply to all patients?
All patients should have a total-body skin examination at least once per year; however, the frequency may change based on a prior history of melanoma or skin cancer, number of nevi or dysplastic nevi, and a family history of melanoma.
Patients should be completely undressed, and all nail polish or artificial nails should be removed prior to the examination. A complete cutaneous examination involves inspecting all skin surfaces, scalp, ocular and oral mucosa, fingernails/toenails, and genitalia if the patient agrees. Melanoma can occur in non–UV-exposed areas and the patient should be educated. Explain the ABCDEs of melanoma diagnosis to all patients and discuss concerns of any new or changing lesions, pigmented or not.
The patient should be made aware that a series of digital images will be taken for any suspicious lesions for possible short-term monitoring. The patient also may be offered full-body photography or 3D body imaging if the number of nevi warrants it.
Different patient populations have different risks for melanoma. Although melanoma predominately afflicts patients with a light skin type, there are certain types of melanoma, such as acral melanoma, that can be more common in darker skin types.
If a patient has a history of cutaneous melanoma, then the site should be checked for any local recurrence as well as palpation of the draining lymph nodes and regional lymph nodes.
I also let patients know that I will be using tools such as dermoscopy and/or reflectance confocal microscopy to better diagnose equivocal lesions before pursuing a biopsy. A biopsy may be done if there is a level of suspicion for atypia.
The use of dermoscopy, digital imaging, and reflectance confocal microscopy has changed the way we can detect, monitor, and evaluate atypical nevi. These tools can augment practice and possibly cut down on the rate of biopsies. They also are great for equivocal lesions or lesions that are in cosmetically sensitive areas. I use these tools in my everyday practice.
How do you keep patients compliant?
Empowering patients to perform self-examinations as well as examinations with his/her partner may help to reinforce monitoring by a dermatologist.
Provide patients with reading materials on self-examination while they wait in the office for your examination.
What do you do if they refuse treatment?
If patients defer a full-body skin examination, then I try to educate them about risks for UV exposure and the risk factors for both melanoma and nonmelanoma skin cancer. I also provide information on self-examinations so they can check at home for any irregularly shaped or changing moles.
What resources do you recommend to patients for more information?
It is important for patients to understand the risk factors for melanoma and the long-term prognosis of melanoma. I direct them to the American Academy of Dermatology’s website (http://www.AAD.org) for education and background about melanoma. Also, the Skin Cancer Foundation has inspiring patient stories (http://www.SkinCancer.org).
Optical Imaging to Detect Lentigo Maligna

In an article published online on January 26 in the Journal of the American Academy of Dermatology, my colleagues and I (Menge et al) reported on the use of reflectance confocal microscopy (RCM) for challenging facial lesions. We studied the diagnosis of lentigo maligna (LM) based on RCM versus the histopathologic diagnosis after biopsy.
In this study 17 patients were seen for evaluation of known or suspected LM at Memorial Sloan Kettering Cancer Center (New York, New York). Among these patients, a total of 63 sites on the skin were evaluated using RCM and a presumptive diagnosis was made. These sites were then biopsied to compare the diagnosis using RCM with that made by histopathology. When LM was present as determined by biopsy, RCM also was able to detect it 100% of the time (sensitivity). When LM was absent as determined by biopsy, RCM also indicated it was absent 71% of the time (specificity).
What’s the issue?
Lentigo maligna is a form of melanoma in situ occurring on sun-damaged skin. It can be quite subtle to detect clinically and therefore may go undiagnosed for a while. Lentigo maligna also has been shown to have notable subclinical extension with which traditional surgical margins for truncal melanoma may be too narrow to clear LM on the head and neck. Therefore, presurgical consultation may be difficult due to the amorphous borders. Random blind biopsies also are discouraged because of sampling error.
Additionally, repetitive biopsies over time, which may be frequently needed in individuals with heavy sun exposure, can be costly and cause adverse effects.
This study showed the usefulness and reliability of using RCM for challenging facial lesions that are suspicious for LM. The sensitivity and specificity of RCM in this study indicated that this technology performs well in detecting LM when present; however, false-positives were noted in this study. False-positives included pigmented actinic keratosis and melanocytosis. Dermatologists who are advanced in RCM technology and interpretation also were utilized in this study. More research is needed to understand how to best utilize this technology, but overall the ability of RCM to accurately identify LM without biopsy represents an exciting new development in how dermatologists can better diagnose, manage, and treat melanoma.
How will you adopt advances in cutaneous noninvasive imaging?

In an article published online on January 26 in the Journal of the American Academy of Dermatology, my colleagues and I (Menge et al) reported on the use of reflectance confocal microscopy (RCM) for challenging facial lesions. We studied the diagnosis of lentigo maligna (LM) based on RCM versus the histopathologic diagnosis after biopsy.
In this study 17 patients were seen for evaluation of known or suspected LM at Memorial Sloan Kettering Cancer Center (New York, New York). Among these patients, a total of 63 sites on the skin were evaluated using RCM and a presumptive diagnosis was made. These sites were then biopsied to compare the diagnosis using RCM with that made by histopathology. When LM was present as determined by biopsy, RCM also was able to detect it 100% of the time (sensitivity). When LM was absent as determined by biopsy, RCM also indicated it was absent 71% of the time (specificity).
What’s the issue?
Lentigo maligna is a form of melanoma in situ occurring on sun-damaged skin. It can be quite subtle to detect clinically and therefore may go undiagnosed for a while. Lentigo maligna also has been shown to have notable subclinical extension with which traditional surgical margins for truncal melanoma may be too narrow to clear LM on the head and neck. Therefore, presurgical consultation may be difficult due to the amorphous borders. Random blind biopsies also are discouraged because of sampling error.
Additionally, repetitive biopsies over time, which may be frequently needed in individuals with heavy sun exposure, can be costly and cause adverse effects.
This study showed the usefulness and reliability of using RCM for challenging facial lesions that are suspicious for LM. The sensitivity and specificity of RCM in this study indicated that this technology performs well in detecting LM when present; however, false-positives were noted in this study. False-positives included pigmented actinic keratosis and melanocytosis. Dermatologists who are advanced in RCM technology and interpretation also were utilized in this study. More research is needed to understand how to best utilize this technology, but overall the ability of RCM to accurately identify LM without biopsy represents an exciting new development in how dermatologists can better diagnose, manage, and treat melanoma.
How will you adopt advances in cutaneous noninvasive imaging?

In an article published online on January 26 in the Journal of the American Academy of Dermatology, my colleagues and I (Menge et al) reported on the use of reflectance confocal microscopy (RCM) for challenging facial lesions. We studied the diagnosis of lentigo maligna (LM) based on RCM versus the histopathologic diagnosis after biopsy.
In this study 17 patients were seen for evaluation of known or suspected LM at Memorial Sloan Kettering Cancer Center (New York, New York). Among these patients, a total of 63 sites on the skin were evaluated using RCM and a presumptive diagnosis was made. These sites were then biopsied to compare the diagnosis using RCM with that made by histopathology. When LM was present as determined by biopsy, RCM also was able to detect it 100% of the time (sensitivity). When LM was absent as determined by biopsy, RCM also indicated it was absent 71% of the time (specificity).
What’s the issue?
Lentigo maligna is a form of melanoma in situ occurring on sun-damaged skin. It can be quite subtle to detect clinically and therefore may go undiagnosed for a while. Lentigo maligna also has been shown to have notable subclinical extension with which traditional surgical margins for truncal melanoma may be too narrow to clear LM on the head and neck. Therefore, presurgical consultation may be difficult due to the amorphous borders. Random blind biopsies also are discouraged because of sampling error.
Additionally, repetitive biopsies over time, which may be frequently needed in individuals with heavy sun exposure, can be costly and cause adverse effects.
This study showed the usefulness and reliability of using RCM for challenging facial lesions that are suspicious for LM. The sensitivity and specificity of RCM in this study indicated that this technology performs well in detecting LM when present; however, false-positives were noted in this study. False-positives included pigmented actinic keratosis and melanocytosis. Dermatologists who are advanced in RCM technology and interpretation also were utilized in this study. More research is needed to understand how to best utilize this technology, but overall the ability of RCM to accurately identify LM without biopsy represents an exciting new development in how dermatologists can better diagnose, manage, and treat melanoma.
How will you adopt advances in cutaneous noninvasive imaging?