User login
A Pragmatic Approach to Melanoma Screening in Collaboration With Primary Care Providers
In 2009, the US Preventive Services Task Force issued an I statement for routine skin cancer screening, noting a lack of evidence to support the balance of benefits and harms from screening,1 a recommendation that is likely to be upheld this year. As dermatologists and melanoma specialists, we have abundant anecdotal evidence of the value of screening; however, population-based screening performed exclusively by dermatologists is not practical. There are approximately 170,000,000 adults 35 years and older and only 9600 practicing dermatologists in the United States, requiring each dermatologist to screen nearly 18,000 individuals per year to meet the needs of the population.
Only 8% to 15% of people in the United States report having received a recent skin examination by a physician.2,3 Partnering with our primary care provider (PCP) colleagues has the potential to reach more patients and to improve skin cancer screening rates more rapidly. The workforce in primary care is substantially larger than dermatology by approximately 30-fold, and PCPs are more likely than dermatologists to practice in rural areas, thus reaching patients with limited access to dermatologists. Skin cancer screening can be included in the routine PCP visit, reducing the need for an additional physician visit for the patient. Patients visit their PCP more frequently as they age, which parallels the risk for developing and dying from melanoma and also provides an opportunity to introduce skin cancer education and screening to a population at higher risk who may not otherwise seek it on their own.4 Providing PCPs with the training and tools to perform melanoma screening shifts the responsibility of initiating screening from the patient alone to a shared responsibility of patient and provider. Dermatologists, in turn, need to be available to examine those patients found to have a suspicious lesion, treat newly diagnosed skin cancer, and follow those patients at highest risk of developing skin cancer, including those who are immunosuppressed, have multiple atypical moles, or have a personal or family history of melanoma.
Evidence from the SCREEN (Skin Cancer Research to provide Evidence for Effectiveness of Screening in Northern Germany) project supports PCP-based screening. In the 5 years following a 1-year pilot screening program, there was nearly a 50% reduction in melanoma mortality.5 Unfortunately, these encouraging results were not confirmed once the pilot project was translated into a national skin cancer screening program.6 However, there are lessons to be learned from the German project and we propose that PCP-led screening is feasible and practical in the United States and we currently have a pilot program in our institution, the University of Pittsburgh Medical Center (Pittsburgh, Pennsylvania).
In the SCREEN project and in routine practice across the United States, screening is primarily driven by patients. Generally, higher-risk patients such as men and the elderly are the least likely group to seek skin cancer screening. In our program, PCPs are offered training in skin cancer screening using a validated web-based program and alerted through the electronic health record to offer skin cancer screening annually to patients 35 years and older who present for routine primary care visits.7 This approach reduces self-referral bias by promoting physician initiation rather than patient initiation of screening, which can occur while the patient is already in the PCP’s office.
Melanoma thickness can be measured among screened patients, unscreened patients, and historic controls and compared to determine if this approach is effective. Health care utilization data can help to inform us if this approach leads to more skin biopsies and procedures or to an increased rate of dermatology referrals. As health care payment and delivery models evolve, there is greater emphasis on outcomes and team-based care. We believe that this approach will allow us to form effective teams of PCPs, dermatologists, and other experts in melanoma, public health, and informatics to reduce melanoma mortality in a cost-effective manner.
- U.S. Preventive Services Task Force. Screening for skin cancer: U.S. Preventive Services Task Force recommendation statement. Ann Intern Med. 2009;150:188-193.
- Saraiya M, Hall HI, Thompson T, et al. Skin cancer screening among U.S. adults from 1992, 1998, and 2000 National Health Interview Surveys. Prev Med. 2004;39:308-314.
- Coups EJ, Geller AC, Weinstock MA, et al. Prevalence and correlates of skin cancer screening among middle-aged and older white adults in the United States. Am J Med. 2010;123:439-445.
- Centers for Disease Control and Prevention. Ambulatory care use and physician office visits. CDC website. http://www.cdc.gov/nchs/fastats/physician-visits.htm. Updated April 27, 2016. Accessed May 4, 2016.
- Katalinic A, Waldmann A, Weinstock MA, et al. Does skin cancer screening save lives? an observational study comparing trends in melanoma mortality in regions with and without screening. Cancer. 2012;118:5395-5402.
- Katalinic A, Eisemann N, Waldmann A. Skin cancer screening in Germany. documenting melanoma incidence and mortality from 2008 to 2013. Dtsch Arztebl Int. 2015;112:629-634.
- Weinstock M. INFORMED: melanoma and skin cancer early detection. Skinsight website. http://www.skinsight.com/info/for_professionals/skin-cancer-detection-informed/skin-cancer-education. Accessed May 12, 2016.
In 2009, the US Preventive Services Task Force issued an I statement for routine skin cancer screening, noting a lack of evidence to support the balance of benefits and harms from screening,1 a recommendation that is likely to be upheld this year. As dermatologists and melanoma specialists, we have abundant anecdotal evidence of the value of screening; however, population-based screening performed exclusively by dermatologists is not practical. There are approximately 170,000,000 adults 35 years and older and only 9600 practicing dermatologists in the United States, requiring each dermatologist to screen nearly 18,000 individuals per year to meet the needs of the population.
Only 8% to 15% of people in the United States report having received a recent skin examination by a physician.2,3 Partnering with our primary care provider (PCP) colleagues has the potential to reach more patients and to improve skin cancer screening rates more rapidly. The workforce in primary care is substantially larger than dermatology by approximately 30-fold, and PCPs are more likely than dermatologists to practice in rural areas, thus reaching patients with limited access to dermatologists. Skin cancer screening can be included in the routine PCP visit, reducing the need for an additional physician visit for the patient. Patients visit their PCP more frequently as they age, which parallels the risk for developing and dying from melanoma and also provides an opportunity to introduce skin cancer education and screening to a population at higher risk who may not otherwise seek it on their own.4 Providing PCPs with the training and tools to perform melanoma screening shifts the responsibility of initiating screening from the patient alone to a shared responsibility of patient and provider. Dermatologists, in turn, need to be available to examine those patients found to have a suspicious lesion, treat newly diagnosed skin cancer, and follow those patients at highest risk of developing skin cancer, including those who are immunosuppressed, have multiple atypical moles, or have a personal or family history of melanoma.
Evidence from the SCREEN (Skin Cancer Research to provide Evidence for Effectiveness of Screening in Northern Germany) project supports PCP-based screening. In the 5 years following a 1-year pilot screening program, there was nearly a 50% reduction in melanoma mortality.5 Unfortunately, these encouraging results were not confirmed once the pilot project was translated into a national skin cancer screening program.6 However, there are lessons to be learned from the German project and we propose that PCP-led screening is feasible and practical in the United States and we currently have a pilot program in our institution, the University of Pittsburgh Medical Center (Pittsburgh, Pennsylvania).
In the SCREEN project and in routine practice across the United States, screening is primarily driven by patients. Generally, higher-risk patients such as men and the elderly are the least likely group to seek skin cancer screening. In our program, PCPs are offered training in skin cancer screening using a validated web-based program and alerted through the electronic health record to offer skin cancer screening annually to patients 35 years and older who present for routine primary care visits.7 This approach reduces self-referral bias by promoting physician initiation rather than patient initiation of screening, which can occur while the patient is already in the PCP’s office.
Melanoma thickness can be measured among screened patients, unscreened patients, and historic controls and compared to determine if this approach is effective. Health care utilization data can help to inform us if this approach leads to more skin biopsies and procedures or to an increased rate of dermatology referrals. As health care payment and delivery models evolve, there is greater emphasis on outcomes and team-based care. We believe that this approach will allow us to form effective teams of PCPs, dermatologists, and other experts in melanoma, public health, and informatics to reduce melanoma mortality in a cost-effective manner.
In 2009, the US Preventive Services Task Force issued an I statement for routine skin cancer screening, noting a lack of evidence to support the balance of benefits and harms from screening,1 a recommendation that is likely to be upheld this year. As dermatologists and melanoma specialists, we have abundant anecdotal evidence of the value of screening; however, population-based screening performed exclusively by dermatologists is not practical. There are approximately 170,000,000 adults 35 years and older and only 9600 practicing dermatologists in the United States, requiring each dermatologist to screen nearly 18,000 individuals per year to meet the needs of the population.
Only 8% to 15% of people in the United States report having received a recent skin examination by a physician.2,3 Partnering with our primary care provider (PCP) colleagues has the potential to reach more patients and to improve skin cancer screening rates more rapidly. The workforce in primary care is substantially larger than dermatology by approximately 30-fold, and PCPs are more likely than dermatologists to practice in rural areas, thus reaching patients with limited access to dermatologists. Skin cancer screening can be included in the routine PCP visit, reducing the need for an additional physician visit for the patient. Patients visit their PCP more frequently as they age, which parallels the risk for developing and dying from melanoma and also provides an opportunity to introduce skin cancer education and screening to a population at higher risk who may not otherwise seek it on their own.4 Providing PCPs with the training and tools to perform melanoma screening shifts the responsibility of initiating screening from the patient alone to a shared responsibility of patient and provider. Dermatologists, in turn, need to be available to examine those patients found to have a suspicious lesion, treat newly diagnosed skin cancer, and follow those patients at highest risk of developing skin cancer, including those who are immunosuppressed, have multiple atypical moles, or have a personal or family history of melanoma.
Evidence from the SCREEN (Skin Cancer Research to provide Evidence for Effectiveness of Screening in Northern Germany) project supports PCP-based screening. In the 5 years following a 1-year pilot screening program, there was nearly a 50% reduction in melanoma mortality.5 Unfortunately, these encouraging results were not confirmed once the pilot project was translated into a national skin cancer screening program.6 However, there are lessons to be learned from the German project and we propose that PCP-led screening is feasible and practical in the United States and we currently have a pilot program in our institution, the University of Pittsburgh Medical Center (Pittsburgh, Pennsylvania).
In the SCREEN project and in routine practice across the United States, screening is primarily driven by patients. Generally, higher-risk patients such as men and the elderly are the least likely group to seek skin cancer screening. In our program, PCPs are offered training in skin cancer screening using a validated web-based program and alerted through the electronic health record to offer skin cancer screening annually to patients 35 years and older who present for routine primary care visits.7 This approach reduces self-referral bias by promoting physician initiation rather than patient initiation of screening, which can occur while the patient is already in the PCP’s office.
Melanoma thickness can be measured among screened patients, unscreened patients, and historic controls and compared to determine if this approach is effective. Health care utilization data can help to inform us if this approach leads to more skin biopsies and procedures or to an increased rate of dermatology referrals. As health care payment and delivery models evolve, there is greater emphasis on outcomes and team-based care. We believe that this approach will allow us to form effective teams of PCPs, dermatologists, and other experts in melanoma, public health, and informatics to reduce melanoma mortality in a cost-effective manner.
- U.S. Preventive Services Task Force. Screening for skin cancer: U.S. Preventive Services Task Force recommendation statement. Ann Intern Med. 2009;150:188-193.
- Saraiya M, Hall HI, Thompson T, et al. Skin cancer screening among U.S. adults from 1992, 1998, and 2000 National Health Interview Surveys. Prev Med. 2004;39:308-314.
- Coups EJ, Geller AC, Weinstock MA, et al. Prevalence and correlates of skin cancer screening among middle-aged and older white adults in the United States. Am J Med. 2010;123:439-445.
- Centers for Disease Control and Prevention. Ambulatory care use and physician office visits. CDC website. http://www.cdc.gov/nchs/fastats/physician-visits.htm. Updated April 27, 2016. Accessed May 4, 2016.
- Katalinic A, Waldmann A, Weinstock MA, et al. Does skin cancer screening save lives? an observational study comparing trends in melanoma mortality in regions with and without screening. Cancer. 2012;118:5395-5402.
- Katalinic A, Eisemann N, Waldmann A. Skin cancer screening in Germany. documenting melanoma incidence and mortality from 2008 to 2013. Dtsch Arztebl Int. 2015;112:629-634.
- Weinstock M. INFORMED: melanoma and skin cancer early detection. Skinsight website. http://www.skinsight.com/info/for_professionals/skin-cancer-detection-informed/skin-cancer-education. Accessed May 12, 2016.
- U.S. Preventive Services Task Force. Screening for skin cancer: U.S. Preventive Services Task Force recommendation statement. Ann Intern Med. 2009;150:188-193.
- Saraiya M, Hall HI, Thompson T, et al. Skin cancer screening among U.S. adults from 1992, 1998, and 2000 National Health Interview Surveys. Prev Med. 2004;39:308-314.
- Coups EJ, Geller AC, Weinstock MA, et al. Prevalence and correlates of skin cancer screening among middle-aged and older white adults in the United States. Am J Med. 2010;123:439-445.
- Centers for Disease Control and Prevention. Ambulatory care use and physician office visits. CDC website. http://www.cdc.gov/nchs/fastats/physician-visits.htm. Updated April 27, 2016. Accessed May 4, 2016.
- Katalinic A, Waldmann A, Weinstock MA, et al. Does skin cancer screening save lives? an observational study comparing trends in melanoma mortality in regions with and without screening. Cancer. 2012;118:5395-5402.
- Katalinic A, Eisemann N, Waldmann A. Skin cancer screening in Germany. documenting melanoma incidence and mortality from 2008 to 2013. Dtsch Arztebl Int. 2015;112:629-634.
- Weinstock M. INFORMED: melanoma and skin cancer early detection. Skinsight website. http://www.skinsight.com/info/for_professionals/skin-cancer-detection-informed/skin-cancer-education. Accessed May 12, 2016.
Novel Melanoma Therapies and Their Side Effects
In the last few years, melanoma treatment has been revolutionized by the development of immune checkpoint–blocking antibodies or immune checkpoint inhibitors. These drugs act through receptor or ligand blockades at certain points along the immunologic cascade to enhance the immune system’s ability to fight malignancies.1 In 2011, the US Food and Drug Administration approved ipilimumab, an inhibitor of cytotoxic T-lymphocyte antigen 4 (CTLA-4), for treatment of patients with unresectable or metastatic melanoma. Other immune-modulating agents followed thereafter. Vemurafenib and dabrafenib, 2 selective BRAF inhibitors, were approved in 2011 and 2013, respectively, and trametinib, a mitogen-activated extracellular signal-regulated kinase 1 (MEK1) and MEK2 blocker, was approved in 2013. These agents are being used to treat patients with activating BRAF or NRAS mutations.2,3 Nivolumab and pembrolizumab, which target programmed death receptor-1 (PD-1) and programmed death ligand 1 (PD-L1), respectively, were approved in 2014. Furthermore, phase 2 and 3 trials are ongoing for patients with unresectable stage III or IV melanomas harboring activating c-KIT mutations, which are rare and usually are found in acral or mucosal melanomas. The multikinase inhibitors imatinib, sunitinib, dasatinib, and nilotinib are being used in clinical trials for this purpose and are not yet approved.4
Although immune checkpoint inhibitors have shown promising results, they lack direct activity against malignant cells. The nonspecific enhanced immune system response promoted by these drugs has been shown to cause multiple adverse events (AEs). A subset of these side effects has been termed immune-related AEs (irAEs), which occur secondary to reduced tolerance to antigens previously recognized as self-antigens, leading to immune-related side effects.5 The majority of these AEs involve the skin and are mild to moderate in severity; however, other organ systems (eg, gastrointestinal, hepatic, endocrine, and neurologic systems) also may be affected. Most of the toxicities have been successfully treated with immunosuppressive agents such as corticosteroids, tumor necrosis factor α antagonists, and mycophenolate mofetil.6
Dermatologic Side Effects
The most common AEs associated with immune checkpoint inhibitors are cutaneous reactions, which commonly present after 2 to 3 weeks of treatment.7 Approximately 50% of patients receiving ipilimumab (CTLA-4 inhibitor) will experience cutaneous reactions, including erythematous, reticulated, or maculopapular rashes.8 Vitiligo and Sweet syndrome also have been observed.9,10
Antibodies against PD-1 and PD-L1 have been associated with oral mucositis and dry mouth.11 Most patients treated with BRAF, MEK, and KIT inhibitors also experience dermatologic AEs. Rashes caused by BRAF inhibitors commonly are maculopapular to verrucous and hyperkeratotic. Keratoacanthomas, squamous cell carcinomas, and other hyperkeratotic lesions such as verruca vulgaris, actinic keratoses, and milia have been reported, usually in sun-exposed areas.4,12,13 Other types of keratotic lesions have been observed, such as areolar hyperkeratosis with vemurafenib (BRAF inhibitor).14 Photosensitivity, panniculitis (eg, erythema nodosum), and mild alopecia also have been reported.15 Radiosensitization and radiation recall also have been reported in patients treated with BRAF inhibitors.16-19 Cutaneous reactions observed with MEK inhibitors are acneiform to papulopustular and appear in seborrheic areas such as the face and chest.4 In contrast to BRAF inhibitors, increased rates of squamous cell carcinomas and keratoacanthomas have not been reported with MEK inhibitors. Severe cutaneous effects such as toxic epidermal necrolysis and Stevens-Johnson syndrome may occur, and although rare, treatment should be discontinued in these cases.
Gastrointestinal Tract Side Effects
Gastrointestinal (GI) tract side effects commonly result from treatment with immunomodulators, usually occurring after 6 to 7 weeks.7 Most patients will experience mild to moderate GI adverse effects (eg, diarrhea), but a few patients have had episodes of colitis, some of which have been fatal.20 Diarrhea and other GI effects are more common in patients treated with ipilimumab, occurring in approximately 30% of patients,20 in comparison to 1% to 2% of those treated with PD-1 and PD-L1 inhibitors.11,21
Liver abnormalities and asymptomatic elevations in liver enzymes can occur with KIT, BRAF, CTLA-4, and PD-L1 inhibitors.11,20-23 More serious abnormalities such as symptomatic hepatitis and fever are mostly seen with CTLA-4 inhibitors.
Endocrinologic Side Effects
Immune-related AEs also can affect the pituitary, adrenal, and thyroid glands. These events occur after an average of 9 weeks and usually consist of nausea, headache, and/or fatigue.7 Hypophysitis and hypothyroidism are the most common endocrinopathies reported based on characteristic laboratory or radiographic findings and are observed most often with CTLA-4 inhibitors, though they also have been reported with PD-1/PD-L1 blockers.24,25 Ipilimumab-induced thyrotoxicosis also has been reported, though it is far less common than hypothyroidism.26
Other Side Effects
Other irAEs that are less common include neurologic side effects ranging from Bell palsy27 and Guillain-Barré syndrome20 to paresthesia, as well as pancreatitis,28 ophthalmologic reactions,29-33 nephritis,34,35 and hematologic side effects.36-38 One distinctive AE is lung toxicity, which has been reported with PD-1 inhibitors and presents as cough, dyspnea, or pneumonitis early in treatment.21
It is unclear whether immunomodulating agents exacerbate autoimmune diseases. Patients with autoimmune diseases were not included in the clinical trials but reportedly have been treated with ipilimumab without exacerbations. Nevertheless, there has been a report of worsening multiple sclerosis in a melanoma patient treated with ipilimumab.39
Conclusion
Immunomodulators have dramatically improved the survival and care of patients with unresectable melanomas. Because of their mechanism of action, they have the capability to produce substantial toxicity. Although most AEs are mild, lethal side effects can ensue. Therefore, all specialists treating patients with melanoma should be familiar with these side effects and their treatment options, as survival rates and survival times will be increasing over the next few years. Rapid AE identification and treatment can improve patient outcomes and optimize the therapeutic potential of these medications. Because immune checkpoint inhibitors are fairly new, further studies are needed to assess irAEs and the long-term impact in patients treated with immunomodulators.
- Ito A, Kondo S, Tada K, et al. Clinical development of immune checkpoint inhibitors. Biomed Res Int. 2015;2015:605478.
- Flaherty KT, Infante JR, Daud A, et al. Combined BRAF and MEK inhibition in melanoma with BRAF V600 mutations. N Engl J Med. 2012;367:1694-1703.
- Ascierto PA, Schadendorf D, Berking C, et al. MEK162 for patients with advanced melanoma harbouring NRAS or Val600 BRAF mutations: a non-randomised, open-label phase 2 study. Lancet Oncol. 2013;14:249-256.
- Livingstone E, Zimmer L, Vaubel J, et al. BRAF, MEK and KIT inhibitors for melanoma: adverse events and their management. Chin Clin Oncol. 2014;3:29.
- Schmerling RA. Toxicity of checkpoint inhibitors. Chin Clin Oncol. 2014;3:31.
- Yervoy [package insert]. Princeton, NJ: Bristol-Myers Squibb Company; 2011.
- Weber JS, Kähler KC, Hauschild A. Management of immune-related adverse events and kinetics of response with ipilimumab. J Clin Oncol. 2012;30:2691-2697.
- Lacouture ME, Wolchok JD, Yosipovitch G, et al. Ipilimumab in patients with cancer and the management of dermatologic adverse events. J Am Acad Dermatol. 2014;71:161-169.
- Wolchok JD, Neyns B, Linette G, et al. Ipilimumab monotherapy in patients with pretreated advanced melanoma: a randomised, double-blind, multicentre, phase 2, dose-ranging study [published online December 8, 2009]. Lancet Oncol. 2010;11:155-164.
- Pintova S, Sidhu H, Friedlander PA, et al. Sweet’s syndrome in a patient with metastatic melanoma after ipilimumab therapy. Melanoma Res. 2013;23:498-501.
- Topalian SL, Sznol M, McDermott DF, et al. Survival, durable tumor remission, and long-term safety in patients with advanced melanoma receiving nivolumab. J Clin Oncol. 2014;32:1020-1030.
- Chapman PB, Hauschild A, Robert C, et al. Improved survival with vemurafenib in melanoma with BRAF V600E mutation. N Engl J Med. 2011;364:2507-2516.
- Lacouture ME, Duvic M, Hauschild A, et al. Analysis of dermatologic events in vemurafenib-treated patients with melanoma. Oncologist. 2013;18:314-322.
- Martinez-Garcia E, Taibjee S, Koch D, et al. Vemurafenib-induced hyperkeratosis of the areola treated with topical adapalene [published online February 22, 2015]. Clin Exp Dermatol. 2016;41:148-151.
- Sanlorenzo M, Choudhry A, Vujic I, et al. Comparative profile of cutaneous adverse events: BRAF/MEK inhibitor combination therapy versus BRAF monotherapy in melanoma. J Am Acad Dermatol. 2014;71:1102-1109.
- Boussemart L, Boivin C, Claveau J, et al. Vemurafenib and radiosensitization. JAMA Dermatol. 2013;149:855-857.
- Ducassou A, David I, Delannes M, et al. Radiosensitization induced by vemurafenib. Cancer Radiother. 2013;17:304-307.
- Peuvrel L, Ruellan AL, Thillays F, et al. Severe radiotherapy-induced extracutaneous toxicity under vemurafenib. Eur J Dermatol. 2013;23:879-881.
- Satzger I, Degen A, Asper H, et al. Serious skin toxicity with the combination of BRAF inhibitors and radiotherapy. J Clin Oncol. 2013;31:e220-e222.
- Hodi FS, O’Day SJ, McDermott DF, et al. Improved survival with ipilimumab in patients with metastatic melanoma. N Engl J Med. 2010;363:711-723.
- Hamid O, Robert C, Daud A, et al. Safety and tumor responses with lambrolizumab (anti-PD-1) in melanoma. N Engl J Med. 2013;369:134-144.
- Topalian SL, Hodi FS, Brahmer JR, et al. Safety, activity, and immune correlates of anti-PD-1 antibody in cancer. N Engl J Med. 2012;366:2443-2454.
- Ribas A, Kefford R, Marshall MA, et al. Phase III randomized clinical trial comparing tremelimumab with standard-of-care chemotherapy in patients with advanced melanoma. J Clin Oncol. 2013;31:616-622.
- Corsello SM, Barnabei A, Marchetti P, et al. Endocrine side effects induced by immune checkpoint inhibitors. J Clin Endocrinol Metab. 2013;98:1361-1375.
- Ryder M, Callahan M, Postow MA, et al. Endocrine-related adverse events following ipilimumab in patients with advanced melanoma: a comprehensive retrospective review from a single institution. Endocr Relat Cancer. 2014;21:371-381.
- Yu C, Chopra IJ, Ha E. A novel melanoma therapy stirs up a storm: ipilimumab-induced thyrotoxicosis. Endocrinol Diabetes Metab Case Rep. 2015;2015:140092.
- Klein O, Ribas A, Chmielowski B, et al. Facial palsy as a side effect of vemurafenib treatment in patients with metastatic melanoma. J Clin Oncol. 2013;31:e215-e217.
- Muluneh B, Buie LW, Collichio F. Vemurafenib-associated pancreatitis: case report. Pharmacotherapy. 2013;33:e43-e44.
- Flaherty L, Hamid O, Linette G, et al. A single-arm, open-label, expanded access study of vemurafenib in patients with metastatic melanoma in the United States. Cancer J. 2014;20:18-24.
- Wolf SE, Meenken C, Moll AC, et al. Severe pan-uveitis in a patient treated with vemurafenib for metastatic melanoma. BMC Cancer. 2013;13:561.
- Sandhu SS, Ling C, Lim L, et al. Vemurafenib (B-RAF inhibitor) associated uveitis in patients with metastatic cutaneous melanoma. Clin Exp Ophthalmol. 2012;40:118.
- Joshi L, Karydis A, Gemenetzi M, et al. Uveitis as a result of MAP kinase pathway inhibition. Case Rep Ophthalmol. 2013;4:279-282.
- Robinson MR, Chan CC, Yang JC, et al. Cytotoxic T lymphocyte-associated antigen 4 blockade in patients with metastatic melanoma: a new cause of uveitis. J Immunother. 2004;27:478-479.
- Regnier-Rosencher E, Lazareth H, Gressier L, et al. Acute kidney injury in patients with severe rash on vemurafenib treatment for metastatic melanomas. Br J Dermatol. 2013;169:934-938.
- Izzedine H, Gueutin V, Gharbi C, et al. Kidney injuries related to ipilimumab [published online April 1, 2014]. Invest New Drugs. 2014;32:769-773.
- Akhtari M, Waller EK, Jaye DL, et al. Neutropenia in a patient treated with ipilimumab (anti-CTLA-4 antibody). J Immunother. 2009;32:322-324.
- Gordon IO, Wade T, Chin K, et al. Immune mediated red cell aplasia after anti-CTLA-4 immunotherapy for metastatic melanoma. Cancer Immunol Immunother. 2009;58:1351-1353.
- Kopecký J, Trojanová P, Kubeček O, et al. Treatment possibilities of ipilimumab-induced thrombocytopenia—case study and literature review. Jpn J Clin Oncol. 2015;45:381-384.
- Gettings EJ, Hackett CT, Scott TF. Severe relapse in a multiple sclerosis patient associated with ipilimumab treatment of melanoma. Mult Scler. 2015;21:670.
In the last few years, melanoma treatment has been revolutionized by the development of immune checkpoint–blocking antibodies or immune checkpoint inhibitors. These drugs act through receptor or ligand blockades at certain points along the immunologic cascade to enhance the immune system’s ability to fight malignancies.1 In 2011, the US Food and Drug Administration approved ipilimumab, an inhibitor of cytotoxic T-lymphocyte antigen 4 (CTLA-4), for treatment of patients with unresectable or metastatic melanoma. Other immune-modulating agents followed thereafter. Vemurafenib and dabrafenib, 2 selective BRAF inhibitors, were approved in 2011 and 2013, respectively, and trametinib, a mitogen-activated extracellular signal-regulated kinase 1 (MEK1) and MEK2 blocker, was approved in 2013. These agents are being used to treat patients with activating BRAF or NRAS mutations.2,3 Nivolumab and pembrolizumab, which target programmed death receptor-1 (PD-1) and programmed death ligand 1 (PD-L1), respectively, were approved in 2014. Furthermore, phase 2 and 3 trials are ongoing for patients with unresectable stage III or IV melanomas harboring activating c-KIT mutations, which are rare and usually are found in acral or mucosal melanomas. The multikinase inhibitors imatinib, sunitinib, dasatinib, and nilotinib are being used in clinical trials for this purpose and are not yet approved.4
Although immune checkpoint inhibitors have shown promising results, they lack direct activity against malignant cells. The nonspecific enhanced immune system response promoted by these drugs has been shown to cause multiple adverse events (AEs). A subset of these side effects has been termed immune-related AEs (irAEs), which occur secondary to reduced tolerance to antigens previously recognized as self-antigens, leading to immune-related side effects.5 The majority of these AEs involve the skin and are mild to moderate in severity; however, other organ systems (eg, gastrointestinal, hepatic, endocrine, and neurologic systems) also may be affected. Most of the toxicities have been successfully treated with immunosuppressive agents such as corticosteroids, tumor necrosis factor α antagonists, and mycophenolate mofetil.6
Dermatologic Side Effects
The most common AEs associated with immune checkpoint inhibitors are cutaneous reactions, which commonly present after 2 to 3 weeks of treatment.7 Approximately 50% of patients receiving ipilimumab (CTLA-4 inhibitor) will experience cutaneous reactions, including erythematous, reticulated, or maculopapular rashes.8 Vitiligo and Sweet syndrome also have been observed.9,10
Antibodies against PD-1 and PD-L1 have been associated with oral mucositis and dry mouth.11 Most patients treated with BRAF, MEK, and KIT inhibitors also experience dermatologic AEs. Rashes caused by BRAF inhibitors commonly are maculopapular to verrucous and hyperkeratotic. Keratoacanthomas, squamous cell carcinomas, and other hyperkeratotic lesions such as verruca vulgaris, actinic keratoses, and milia have been reported, usually in sun-exposed areas.4,12,13 Other types of keratotic lesions have been observed, such as areolar hyperkeratosis with vemurafenib (BRAF inhibitor).14 Photosensitivity, panniculitis (eg, erythema nodosum), and mild alopecia also have been reported.15 Radiosensitization and radiation recall also have been reported in patients treated with BRAF inhibitors.16-19 Cutaneous reactions observed with MEK inhibitors are acneiform to papulopustular and appear in seborrheic areas such as the face and chest.4 In contrast to BRAF inhibitors, increased rates of squamous cell carcinomas and keratoacanthomas have not been reported with MEK inhibitors. Severe cutaneous effects such as toxic epidermal necrolysis and Stevens-Johnson syndrome may occur, and although rare, treatment should be discontinued in these cases.
Gastrointestinal Tract Side Effects
Gastrointestinal (GI) tract side effects commonly result from treatment with immunomodulators, usually occurring after 6 to 7 weeks.7 Most patients will experience mild to moderate GI adverse effects (eg, diarrhea), but a few patients have had episodes of colitis, some of which have been fatal.20 Diarrhea and other GI effects are more common in patients treated with ipilimumab, occurring in approximately 30% of patients,20 in comparison to 1% to 2% of those treated with PD-1 and PD-L1 inhibitors.11,21
Liver abnormalities and asymptomatic elevations in liver enzymes can occur with KIT, BRAF, CTLA-4, and PD-L1 inhibitors.11,20-23 More serious abnormalities such as symptomatic hepatitis and fever are mostly seen with CTLA-4 inhibitors.
Endocrinologic Side Effects
Immune-related AEs also can affect the pituitary, adrenal, and thyroid glands. These events occur after an average of 9 weeks and usually consist of nausea, headache, and/or fatigue.7 Hypophysitis and hypothyroidism are the most common endocrinopathies reported based on characteristic laboratory or radiographic findings and are observed most often with CTLA-4 inhibitors, though they also have been reported with PD-1/PD-L1 blockers.24,25 Ipilimumab-induced thyrotoxicosis also has been reported, though it is far less common than hypothyroidism.26
Other Side Effects
Other irAEs that are less common include neurologic side effects ranging from Bell palsy27 and Guillain-Barré syndrome20 to paresthesia, as well as pancreatitis,28 ophthalmologic reactions,29-33 nephritis,34,35 and hematologic side effects.36-38 One distinctive AE is lung toxicity, which has been reported with PD-1 inhibitors and presents as cough, dyspnea, or pneumonitis early in treatment.21
It is unclear whether immunomodulating agents exacerbate autoimmune diseases. Patients with autoimmune diseases were not included in the clinical trials but reportedly have been treated with ipilimumab without exacerbations. Nevertheless, there has been a report of worsening multiple sclerosis in a melanoma patient treated with ipilimumab.39
Conclusion
Immunomodulators have dramatically improved the survival and care of patients with unresectable melanomas. Because of their mechanism of action, they have the capability to produce substantial toxicity. Although most AEs are mild, lethal side effects can ensue. Therefore, all specialists treating patients with melanoma should be familiar with these side effects and their treatment options, as survival rates and survival times will be increasing over the next few years. Rapid AE identification and treatment can improve patient outcomes and optimize the therapeutic potential of these medications. Because immune checkpoint inhibitors are fairly new, further studies are needed to assess irAEs and the long-term impact in patients treated with immunomodulators.
In the last few years, melanoma treatment has been revolutionized by the development of immune checkpoint–blocking antibodies or immune checkpoint inhibitors. These drugs act through receptor or ligand blockades at certain points along the immunologic cascade to enhance the immune system’s ability to fight malignancies.1 In 2011, the US Food and Drug Administration approved ipilimumab, an inhibitor of cytotoxic T-lymphocyte antigen 4 (CTLA-4), for treatment of patients with unresectable or metastatic melanoma. Other immune-modulating agents followed thereafter. Vemurafenib and dabrafenib, 2 selective BRAF inhibitors, were approved in 2011 and 2013, respectively, and trametinib, a mitogen-activated extracellular signal-regulated kinase 1 (MEK1) and MEK2 blocker, was approved in 2013. These agents are being used to treat patients with activating BRAF or NRAS mutations.2,3 Nivolumab and pembrolizumab, which target programmed death receptor-1 (PD-1) and programmed death ligand 1 (PD-L1), respectively, were approved in 2014. Furthermore, phase 2 and 3 trials are ongoing for patients with unresectable stage III or IV melanomas harboring activating c-KIT mutations, which are rare and usually are found in acral or mucosal melanomas. The multikinase inhibitors imatinib, sunitinib, dasatinib, and nilotinib are being used in clinical trials for this purpose and are not yet approved.4
Although immune checkpoint inhibitors have shown promising results, they lack direct activity against malignant cells. The nonspecific enhanced immune system response promoted by these drugs has been shown to cause multiple adverse events (AEs). A subset of these side effects has been termed immune-related AEs (irAEs), which occur secondary to reduced tolerance to antigens previously recognized as self-antigens, leading to immune-related side effects.5 The majority of these AEs involve the skin and are mild to moderate in severity; however, other organ systems (eg, gastrointestinal, hepatic, endocrine, and neurologic systems) also may be affected. Most of the toxicities have been successfully treated with immunosuppressive agents such as corticosteroids, tumor necrosis factor α antagonists, and mycophenolate mofetil.6
Dermatologic Side Effects
The most common AEs associated with immune checkpoint inhibitors are cutaneous reactions, which commonly present after 2 to 3 weeks of treatment.7 Approximately 50% of patients receiving ipilimumab (CTLA-4 inhibitor) will experience cutaneous reactions, including erythematous, reticulated, or maculopapular rashes.8 Vitiligo and Sweet syndrome also have been observed.9,10
Antibodies against PD-1 and PD-L1 have been associated with oral mucositis and dry mouth.11 Most patients treated with BRAF, MEK, and KIT inhibitors also experience dermatologic AEs. Rashes caused by BRAF inhibitors commonly are maculopapular to verrucous and hyperkeratotic. Keratoacanthomas, squamous cell carcinomas, and other hyperkeratotic lesions such as verruca vulgaris, actinic keratoses, and milia have been reported, usually in sun-exposed areas.4,12,13 Other types of keratotic lesions have been observed, such as areolar hyperkeratosis with vemurafenib (BRAF inhibitor).14 Photosensitivity, panniculitis (eg, erythema nodosum), and mild alopecia also have been reported.15 Radiosensitization and radiation recall also have been reported in patients treated with BRAF inhibitors.16-19 Cutaneous reactions observed with MEK inhibitors are acneiform to papulopustular and appear in seborrheic areas such as the face and chest.4 In contrast to BRAF inhibitors, increased rates of squamous cell carcinomas and keratoacanthomas have not been reported with MEK inhibitors. Severe cutaneous effects such as toxic epidermal necrolysis and Stevens-Johnson syndrome may occur, and although rare, treatment should be discontinued in these cases.
Gastrointestinal Tract Side Effects
Gastrointestinal (GI) tract side effects commonly result from treatment with immunomodulators, usually occurring after 6 to 7 weeks.7 Most patients will experience mild to moderate GI adverse effects (eg, diarrhea), but a few patients have had episodes of colitis, some of which have been fatal.20 Diarrhea and other GI effects are more common in patients treated with ipilimumab, occurring in approximately 30% of patients,20 in comparison to 1% to 2% of those treated with PD-1 and PD-L1 inhibitors.11,21
Liver abnormalities and asymptomatic elevations in liver enzymes can occur with KIT, BRAF, CTLA-4, and PD-L1 inhibitors.11,20-23 More serious abnormalities such as symptomatic hepatitis and fever are mostly seen with CTLA-4 inhibitors.
Endocrinologic Side Effects
Immune-related AEs also can affect the pituitary, adrenal, and thyroid glands. These events occur after an average of 9 weeks and usually consist of nausea, headache, and/or fatigue.7 Hypophysitis and hypothyroidism are the most common endocrinopathies reported based on characteristic laboratory or radiographic findings and are observed most often with CTLA-4 inhibitors, though they also have been reported with PD-1/PD-L1 blockers.24,25 Ipilimumab-induced thyrotoxicosis also has been reported, though it is far less common than hypothyroidism.26
Other Side Effects
Other irAEs that are less common include neurologic side effects ranging from Bell palsy27 and Guillain-Barré syndrome20 to paresthesia, as well as pancreatitis,28 ophthalmologic reactions,29-33 nephritis,34,35 and hematologic side effects.36-38 One distinctive AE is lung toxicity, which has been reported with PD-1 inhibitors and presents as cough, dyspnea, or pneumonitis early in treatment.21
It is unclear whether immunomodulating agents exacerbate autoimmune diseases. Patients with autoimmune diseases were not included in the clinical trials but reportedly have been treated with ipilimumab without exacerbations. Nevertheless, there has been a report of worsening multiple sclerosis in a melanoma patient treated with ipilimumab.39
Conclusion
Immunomodulators have dramatically improved the survival and care of patients with unresectable melanomas. Because of their mechanism of action, they have the capability to produce substantial toxicity. Although most AEs are mild, lethal side effects can ensue. Therefore, all specialists treating patients with melanoma should be familiar with these side effects and their treatment options, as survival rates and survival times will be increasing over the next few years. Rapid AE identification and treatment can improve patient outcomes and optimize the therapeutic potential of these medications. Because immune checkpoint inhibitors are fairly new, further studies are needed to assess irAEs and the long-term impact in patients treated with immunomodulators.
- Ito A, Kondo S, Tada K, et al. Clinical development of immune checkpoint inhibitors. Biomed Res Int. 2015;2015:605478.
- Flaherty KT, Infante JR, Daud A, et al. Combined BRAF and MEK inhibition in melanoma with BRAF V600 mutations. N Engl J Med. 2012;367:1694-1703.
- Ascierto PA, Schadendorf D, Berking C, et al. MEK162 for patients with advanced melanoma harbouring NRAS or Val600 BRAF mutations: a non-randomised, open-label phase 2 study. Lancet Oncol. 2013;14:249-256.
- Livingstone E, Zimmer L, Vaubel J, et al. BRAF, MEK and KIT inhibitors for melanoma: adverse events and their management. Chin Clin Oncol. 2014;3:29.
- Schmerling RA. Toxicity of checkpoint inhibitors. Chin Clin Oncol. 2014;3:31.
- Yervoy [package insert]. Princeton, NJ: Bristol-Myers Squibb Company; 2011.
- Weber JS, Kähler KC, Hauschild A. Management of immune-related adverse events and kinetics of response with ipilimumab. J Clin Oncol. 2012;30:2691-2697.
- Lacouture ME, Wolchok JD, Yosipovitch G, et al. Ipilimumab in patients with cancer and the management of dermatologic adverse events. J Am Acad Dermatol. 2014;71:161-169.
- Wolchok JD, Neyns B, Linette G, et al. Ipilimumab monotherapy in patients with pretreated advanced melanoma: a randomised, double-blind, multicentre, phase 2, dose-ranging study [published online December 8, 2009]. Lancet Oncol. 2010;11:155-164.
- Pintova S, Sidhu H, Friedlander PA, et al. Sweet’s syndrome in a patient with metastatic melanoma after ipilimumab therapy. Melanoma Res. 2013;23:498-501.
- Topalian SL, Sznol M, McDermott DF, et al. Survival, durable tumor remission, and long-term safety in patients with advanced melanoma receiving nivolumab. J Clin Oncol. 2014;32:1020-1030.
- Chapman PB, Hauschild A, Robert C, et al. Improved survival with vemurafenib in melanoma with BRAF V600E mutation. N Engl J Med. 2011;364:2507-2516.
- Lacouture ME, Duvic M, Hauschild A, et al. Analysis of dermatologic events in vemurafenib-treated patients with melanoma. Oncologist. 2013;18:314-322.
- Martinez-Garcia E, Taibjee S, Koch D, et al. Vemurafenib-induced hyperkeratosis of the areola treated with topical adapalene [published online February 22, 2015]. Clin Exp Dermatol. 2016;41:148-151.
- Sanlorenzo M, Choudhry A, Vujic I, et al. Comparative profile of cutaneous adverse events: BRAF/MEK inhibitor combination therapy versus BRAF monotherapy in melanoma. J Am Acad Dermatol. 2014;71:1102-1109.
- Boussemart L, Boivin C, Claveau J, et al. Vemurafenib and radiosensitization. JAMA Dermatol. 2013;149:855-857.
- Ducassou A, David I, Delannes M, et al. Radiosensitization induced by vemurafenib. Cancer Radiother. 2013;17:304-307.
- Peuvrel L, Ruellan AL, Thillays F, et al. Severe radiotherapy-induced extracutaneous toxicity under vemurafenib. Eur J Dermatol. 2013;23:879-881.
- Satzger I, Degen A, Asper H, et al. Serious skin toxicity with the combination of BRAF inhibitors and radiotherapy. J Clin Oncol. 2013;31:e220-e222.
- Hodi FS, O’Day SJ, McDermott DF, et al. Improved survival with ipilimumab in patients with metastatic melanoma. N Engl J Med. 2010;363:711-723.
- Hamid O, Robert C, Daud A, et al. Safety and tumor responses with lambrolizumab (anti-PD-1) in melanoma. N Engl J Med. 2013;369:134-144.
- Topalian SL, Hodi FS, Brahmer JR, et al. Safety, activity, and immune correlates of anti-PD-1 antibody in cancer. N Engl J Med. 2012;366:2443-2454.
- Ribas A, Kefford R, Marshall MA, et al. Phase III randomized clinical trial comparing tremelimumab with standard-of-care chemotherapy in patients with advanced melanoma. J Clin Oncol. 2013;31:616-622.
- Corsello SM, Barnabei A, Marchetti P, et al. Endocrine side effects induced by immune checkpoint inhibitors. J Clin Endocrinol Metab. 2013;98:1361-1375.
- Ryder M, Callahan M, Postow MA, et al. Endocrine-related adverse events following ipilimumab in patients with advanced melanoma: a comprehensive retrospective review from a single institution. Endocr Relat Cancer. 2014;21:371-381.
- Yu C, Chopra IJ, Ha E. A novel melanoma therapy stirs up a storm: ipilimumab-induced thyrotoxicosis. Endocrinol Diabetes Metab Case Rep. 2015;2015:140092.
- Klein O, Ribas A, Chmielowski B, et al. Facial palsy as a side effect of vemurafenib treatment in patients with metastatic melanoma. J Clin Oncol. 2013;31:e215-e217.
- Muluneh B, Buie LW, Collichio F. Vemurafenib-associated pancreatitis: case report. Pharmacotherapy. 2013;33:e43-e44.
- Flaherty L, Hamid O, Linette G, et al. A single-arm, open-label, expanded access study of vemurafenib in patients with metastatic melanoma in the United States. Cancer J. 2014;20:18-24.
- Wolf SE, Meenken C, Moll AC, et al. Severe pan-uveitis in a patient treated with vemurafenib for metastatic melanoma. BMC Cancer. 2013;13:561.
- Sandhu SS, Ling C, Lim L, et al. Vemurafenib (B-RAF inhibitor) associated uveitis in patients with metastatic cutaneous melanoma. Clin Exp Ophthalmol. 2012;40:118.
- Joshi L, Karydis A, Gemenetzi M, et al. Uveitis as a result of MAP kinase pathway inhibition. Case Rep Ophthalmol. 2013;4:279-282.
- Robinson MR, Chan CC, Yang JC, et al. Cytotoxic T lymphocyte-associated antigen 4 blockade in patients with metastatic melanoma: a new cause of uveitis. J Immunother. 2004;27:478-479.
- Regnier-Rosencher E, Lazareth H, Gressier L, et al. Acute kidney injury in patients with severe rash on vemurafenib treatment for metastatic melanomas. Br J Dermatol. 2013;169:934-938.
- Izzedine H, Gueutin V, Gharbi C, et al. Kidney injuries related to ipilimumab [published online April 1, 2014]. Invest New Drugs. 2014;32:769-773.
- Akhtari M, Waller EK, Jaye DL, et al. Neutropenia in a patient treated with ipilimumab (anti-CTLA-4 antibody). J Immunother. 2009;32:322-324.
- Gordon IO, Wade T, Chin K, et al. Immune mediated red cell aplasia after anti-CTLA-4 immunotherapy for metastatic melanoma. Cancer Immunol Immunother. 2009;58:1351-1353.
- Kopecký J, Trojanová P, Kubeček O, et al. Treatment possibilities of ipilimumab-induced thrombocytopenia—case study and literature review. Jpn J Clin Oncol. 2015;45:381-384.
- Gettings EJ, Hackett CT, Scott TF. Severe relapse in a multiple sclerosis patient associated with ipilimumab treatment of melanoma. Mult Scler. 2015;21:670.
- Ito A, Kondo S, Tada K, et al. Clinical development of immune checkpoint inhibitors. Biomed Res Int. 2015;2015:605478.
- Flaherty KT, Infante JR, Daud A, et al. Combined BRAF and MEK inhibition in melanoma with BRAF V600 mutations. N Engl J Med. 2012;367:1694-1703.
- Ascierto PA, Schadendorf D, Berking C, et al. MEK162 for patients with advanced melanoma harbouring NRAS or Val600 BRAF mutations: a non-randomised, open-label phase 2 study. Lancet Oncol. 2013;14:249-256.
- Livingstone E, Zimmer L, Vaubel J, et al. BRAF, MEK and KIT inhibitors for melanoma: adverse events and their management. Chin Clin Oncol. 2014;3:29.
- Schmerling RA. Toxicity of checkpoint inhibitors. Chin Clin Oncol. 2014;3:31.
- Yervoy [package insert]. Princeton, NJ: Bristol-Myers Squibb Company; 2011.
- Weber JS, Kähler KC, Hauschild A. Management of immune-related adverse events and kinetics of response with ipilimumab. J Clin Oncol. 2012;30:2691-2697.
- Lacouture ME, Wolchok JD, Yosipovitch G, et al. Ipilimumab in patients with cancer and the management of dermatologic adverse events. J Am Acad Dermatol. 2014;71:161-169.
- Wolchok JD, Neyns B, Linette G, et al. Ipilimumab monotherapy in patients with pretreated advanced melanoma: a randomised, double-blind, multicentre, phase 2, dose-ranging study [published online December 8, 2009]. Lancet Oncol. 2010;11:155-164.
- Pintova S, Sidhu H, Friedlander PA, et al. Sweet’s syndrome in a patient with metastatic melanoma after ipilimumab therapy. Melanoma Res. 2013;23:498-501.
- Topalian SL, Sznol M, McDermott DF, et al. Survival, durable tumor remission, and long-term safety in patients with advanced melanoma receiving nivolumab. J Clin Oncol. 2014;32:1020-1030.
- Chapman PB, Hauschild A, Robert C, et al. Improved survival with vemurafenib in melanoma with BRAF V600E mutation. N Engl J Med. 2011;364:2507-2516.
- Lacouture ME, Duvic M, Hauschild A, et al. Analysis of dermatologic events in vemurafenib-treated patients with melanoma. Oncologist. 2013;18:314-322.
- Martinez-Garcia E, Taibjee S, Koch D, et al. Vemurafenib-induced hyperkeratosis of the areola treated with topical adapalene [published online February 22, 2015]. Clin Exp Dermatol. 2016;41:148-151.
- Sanlorenzo M, Choudhry A, Vujic I, et al. Comparative profile of cutaneous adverse events: BRAF/MEK inhibitor combination therapy versus BRAF monotherapy in melanoma. J Am Acad Dermatol. 2014;71:1102-1109.
- Boussemart L, Boivin C, Claveau J, et al. Vemurafenib and radiosensitization. JAMA Dermatol. 2013;149:855-857.
- Ducassou A, David I, Delannes M, et al. Radiosensitization induced by vemurafenib. Cancer Radiother. 2013;17:304-307.
- Peuvrel L, Ruellan AL, Thillays F, et al. Severe radiotherapy-induced extracutaneous toxicity under vemurafenib. Eur J Dermatol. 2013;23:879-881.
- Satzger I, Degen A, Asper H, et al. Serious skin toxicity with the combination of BRAF inhibitors and radiotherapy. J Clin Oncol. 2013;31:e220-e222.
- Hodi FS, O’Day SJ, McDermott DF, et al. Improved survival with ipilimumab in patients with metastatic melanoma. N Engl J Med. 2010;363:711-723.
- Hamid O, Robert C, Daud A, et al. Safety and tumor responses with lambrolizumab (anti-PD-1) in melanoma. N Engl J Med. 2013;369:134-144.
- Topalian SL, Hodi FS, Brahmer JR, et al. Safety, activity, and immune correlates of anti-PD-1 antibody in cancer. N Engl J Med. 2012;366:2443-2454.
- Ribas A, Kefford R, Marshall MA, et al. Phase III randomized clinical trial comparing tremelimumab with standard-of-care chemotherapy in patients with advanced melanoma. J Clin Oncol. 2013;31:616-622.
- Corsello SM, Barnabei A, Marchetti P, et al. Endocrine side effects induced by immune checkpoint inhibitors. J Clin Endocrinol Metab. 2013;98:1361-1375.
- Ryder M, Callahan M, Postow MA, et al. Endocrine-related adverse events following ipilimumab in patients with advanced melanoma: a comprehensive retrospective review from a single institution. Endocr Relat Cancer. 2014;21:371-381.
- Yu C, Chopra IJ, Ha E. A novel melanoma therapy stirs up a storm: ipilimumab-induced thyrotoxicosis. Endocrinol Diabetes Metab Case Rep. 2015;2015:140092.
- Klein O, Ribas A, Chmielowski B, et al. Facial palsy as a side effect of vemurafenib treatment in patients with metastatic melanoma. J Clin Oncol. 2013;31:e215-e217.
- Muluneh B, Buie LW, Collichio F. Vemurafenib-associated pancreatitis: case report. Pharmacotherapy. 2013;33:e43-e44.
- Flaherty L, Hamid O, Linette G, et al. A single-arm, open-label, expanded access study of vemurafenib in patients with metastatic melanoma in the United States. Cancer J. 2014;20:18-24.
- Wolf SE, Meenken C, Moll AC, et al. Severe pan-uveitis in a patient treated with vemurafenib for metastatic melanoma. BMC Cancer. 2013;13:561.
- Sandhu SS, Ling C, Lim L, et al. Vemurafenib (B-RAF inhibitor) associated uveitis in patients with metastatic cutaneous melanoma. Clin Exp Ophthalmol. 2012;40:118.
- Joshi L, Karydis A, Gemenetzi M, et al. Uveitis as a result of MAP kinase pathway inhibition. Case Rep Ophthalmol. 2013;4:279-282.
- Robinson MR, Chan CC, Yang JC, et al. Cytotoxic T lymphocyte-associated antigen 4 blockade in patients with metastatic melanoma: a new cause of uveitis. J Immunother. 2004;27:478-479.
- Regnier-Rosencher E, Lazareth H, Gressier L, et al. Acute kidney injury in patients with severe rash on vemurafenib treatment for metastatic melanomas. Br J Dermatol. 2013;169:934-938.
- Izzedine H, Gueutin V, Gharbi C, et al. Kidney injuries related to ipilimumab [published online April 1, 2014]. Invest New Drugs. 2014;32:769-773.
- Akhtari M, Waller EK, Jaye DL, et al. Neutropenia in a patient treated with ipilimumab (anti-CTLA-4 antibody). J Immunother. 2009;32:322-324.
- Gordon IO, Wade T, Chin K, et al. Immune mediated red cell aplasia after anti-CTLA-4 immunotherapy for metastatic melanoma. Cancer Immunol Immunother. 2009;58:1351-1353.
- Kopecký J, Trojanová P, Kubeček O, et al. Treatment possibilities of ipilimumab-induced thrombocytopenia—case study and literature review. Jpn J Clin Oncol. 2015;45:381-384.
- Gettings EJ, Hackett CT, Scott TF. Severe relapse in a multiple sclerosis patient associated with ipilimumab treatment of melanoma. Mult Scler. 2015;21:670.
Practice Points
- Immune checkpoint inhibitors can cause immune-related adverse events (irAEs), which most commonly involve the skin but also involve the gastrointestinal, hepatic, endocrine, and neurologic systems.
- These irAEs can be treated with corticosteroids, tumor necrosis factor α antagonists, and mycopheno-late mofetil.
Could a Specific Dietary Intake Be a Risk Factor for Cutaneous Melanoma?
The incidence of cutaneous melanoma (CM) has increased, warranting further study of new risk factors.1,2 Hereditary risk factors for CM include light-colored eyes; fair skin; light brown, blonde, or red hair; tendency to burn; high density of freckles; history of other types of skin cancer; high number of common, atypical, and/or congenital nevi; and family history of skin cancer, as well as risks related to the presence of CDKN2A, BRAF, and MC1R gene mutations. Environmental risk factors include UV exposure from sunlight or tanning beds, among others.3-5
Nutritional factors also have been suggested as possible modifiable risk factors for CM.6 Evidence from epidemiological studies show that diets rich in fruits and vegetables are associated with lower risks for several types of cancer.7,8 A growing number of studies have assessed the effects of diet and the intake of nutrients on the prevention of cancer, specifically the use of dietary supplements to protect the skin from the adverse effects of UV light.6
Preformed vitamin A (ie, retinol) is necessary for the regulation of cell differentiation and also can reduce the incidence of skin tumors in animals exposed to UV light. Certain carotenoids such as α-carotene and β-carotene are metabolized to retinol. These retinol precursors, along with antioxidant nutrients, are important components of fruits and vegetables and may account for the observed anticancer effects of these foods.8
The aim of this study was to assess the relationship between dietary intake and the risk for CM.
Methods
Participants
A case-control study was carried out between 2012 and 2013 at 3 reference centers in Porto Alegre, Brazil—Universidade Federal de Ciências da Saúde de Porto Alegre, Pontifícia Universidade Católica do Rio Grande do Sul, and Hospital de Clínicas de Porto Alegre—for the treatment of patients with CM. Enrolled patients were 18 years and older with a diagnosis of primary CM confirmed by histology. Controls were selected from patients at the same centers, and they were enrolled and matched by institution. Controls were frequency matched to cases by sex and age (+/– 5 years). Exclusion criteria for controls were those presenting with suspicious lesions and those needing radiation therapy or chemotherapy due to other diseases. The study was approved by the ethics committees of the participating centers and informed consent was obtained from all participants. A total of 191 participants (95 cases; 96 controls) were enrolled in the study.
Data Collection
After informed consent was obtained, participants were interviewed and were clinically examined by an experienced dermatologist (C.B.H. and M.M.S.). The questionnaire included sociodemographic variables, medical history, phenotypic characteristics (ie, Fitzpatrick skin type, skin/hair/eye color), family history of skin cancer, history of sunlight exposure, history of sunburns, use of artificial tanning, sunscreen use, and detailed dietary intake. Physical examination included the assessment of several melanocytic lesions (nevi, freckles/ephelides, lentigines, and café au lait spots), actinic keratoses, solar elastosis, and nonmelanocytic tumors following the International Agency for Research on Cancer (IARC) protocol.9
Using a food frequency questionnaire, participants were asked to report their usual frequency of consumption of each food from a list of 36 foods. The frequency of intake of all groups of food and beverages was defined according to the following scale: never, rarely (less than once monthly), once or twice weekly, 3 to 4 times weekly, 5 to 7 times weekly, and more than 7 times weekly. Combination of categories was based on the overall distribution among controls. Therefore, for some items such as mussels and fresh herbs, only 2 categories were used.
Statistical Analysis
A descriptive statistical analysis of the results was performed using SPSS version 20.0 with absolute and relative frequencies for the categorical variables, and mean, SD, and median for the continuous variables. The symmetry of distributions was investigated using the Kolmogorov-Smirnov test.
A t test for independent groups was applied for the continuous variables, while the Pearson χ2 test was used for the categorical variables. The Fisher exact test was used in situations in which at least 25% of the values of the cells presented an expected frequency of less than 5. Monte Carlo simulation was used when at least 1 variable had a polytomic characteristic. Odds ratio (OR) was used to estimate the strength of the association between exposures and outcome. An unconditional binary logistic regression was used to study the association between dietary variables and the risk for CM. To obtain unbiased estimates, multivariate analyses were performed controlling for 1 or more confounding variables. Using low exposure as a base category, the risks and 95% CIs were calculated for the high-exposure categories. Based on the results of bivariate analyses, variables with P≤.25 or lower were included in the models. The likelihood ratio test was used to decide which covariates should be maintained in the model. To test the goodness of fit of the models, the Hosmer-Lemeshow statistic was used.
Potential confounding factors considered in the logistic regression model were sex; age; education level; skin, hair, and eye color; Fitzpatrick skin type; presence of freckles, solar lentigines, and actinic keratosis; history of nonmelanoma skin cancer; number of melanocytic nevi; family history of skin cancer; sunburns in adulthood (≥6 episodes a year); occupational sun exposure; and history of sunscreen use in adulthood.
Results
A total of 191 participants were enrolled in the study (95 [49.7%] cases; 96 [50.3%] controls). Most participants were female (60.0% of cases; 59.4% of controls). The mean age (SD) of cases and controls was 56.8 (13.9) years and 56.5 (13.2) years, respectively. Mean body mass index (SD) did not differ between cases (27.2 [4.6]) and controls (28.2 [6.5]). Education levels of 8 years or less predominated in both groups (64.2% of cases; 57.3% of controls). No statistical difference was found for sex, age, education, or body mass index. The most frequent anatomic sites of CM were the trunk (54.7%) and arms (20.0%), and the most frequent histological type was superficial spreading (62.8%). The median Breslow thickness was 0.90 mm. Ulceration was observed in 20.9% of the cases, and 67% of participants with CM had a high mitotic rate (≥1 mitosis per square millimeter).
Phenotypic characteristics associated with an increased risk for melanoma were light brown hair (OR, 6.73; 95% CI, 3.30-14.2), blonde/red hair (OR, 21.7; 95% CI, 7.51-63.1), light-colored eyes (eg, blue, gray, green)(OR, 13.2; 95% CI, 6.13-28.7), light brown eyes (OR, 5.01; 95% CI, 2.24-11.5), and Fitzpatrick skin types I and II (OR, 7.37; 95% CI, 2.90-26.1). Family history of skin cancer was associated with an increased risk for CM (OR, 4.31; 95% CI, 1.86-10.7) as well as sunburns in adulthood (OR, 1.64; 95% CI, 1.17-1.99). Regular sunscreen use in adulthood had a 5-fold increased risk for CM compared to not using sunscreen regularly (OR, 5.6; 95% CI, 2.85-10.7). Regarding pigmented lesions, the presence of solar lentigines (OR, 4.8; 95% CI, 2.2-11.2), 60 or more nevi (OR, 5.4; 95% CI, 2.4-12.7), and freckles (OR, 3.7; 95% CI, 1.82-7.64) were all associated with an increased risk for CM. Solar elastosis (OR, 2.5; 95% CI, 1.08-5.85), actinic keratosis (OR, 9.1, 95% CI, 3.97-20.84), and occupational exposure to sun (OR, 2.57; 95% CI, 1.23-5.38) also were associated with an increased risk for melanoma.
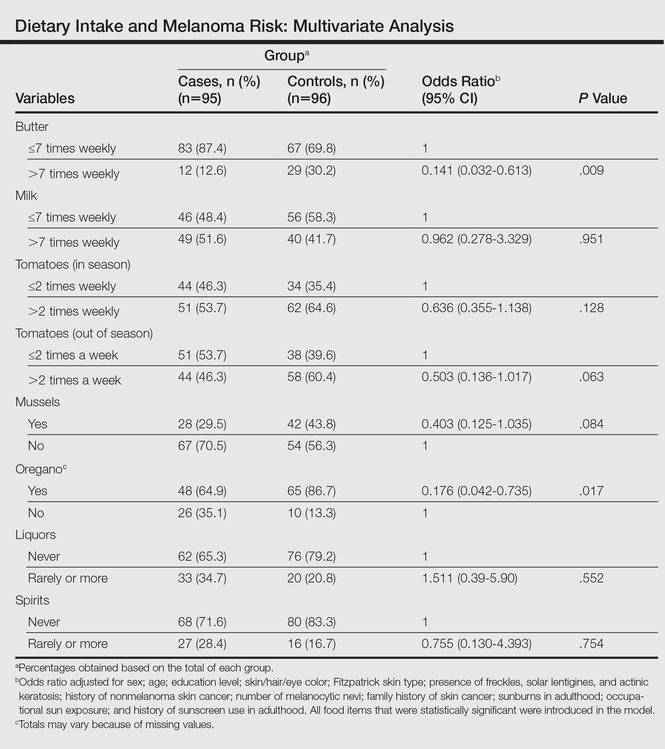
The intake of most of the foods and beverages included in the study showed no association with CM. High frequency of butter intake (more than daily) was a protective factor for CM (OR, 0.33; 95% CI, 0.16-0.70) compared to low-frequency consumption (daily and less than daily). Consumption of mussels (OR, 0.53; 95% CI, 0.29-0.97) and oregano (OR, 0.28; 95% CI, 0.12-0.66) also were shown to be protective against CM (OR, 0.53; 95% CI, 0.29-0.97). Regarding beverages, those in the highest categories of consumption—liquor (OR, 2.12; 95% CI, 1.09-4.12) and spirits (OR, 2.23; 95% CI, 1.16-4.68)—were associated with an increased risk for CM.
To identify the relationship between CM and the consumption of some foods that were relevant on bivariate analysis, we performed a multivariate model. When adjustments were made, the association remained for butter (OR, 0.141; 95% CI, 0.032-0.613) and oregano (OR, 0.176; 95% CI, 0.042-0.735), while the risk associated with the consumption of both liquor (OR, 1.511; 95% CI, 0.39-5.90) and spirits (OR, 0.755; 95% CI, 0.130-4.393) disappeared (Table).
Comment
Observational studies show that diets rich in fruits and vegetables are associated with a lower risk for different types of cancers.7,8 According to some studies, more than 30% of cancers in adulthood could be prevented or delayed by appropriate dietary intake and physical activity.10 However, there are still limited data on some specific cancers such as CM.
Substantial differences in the incidence of CM among different populations have suggested that environmental factors may play an etiological role in the development of CM and diet could be one of the modifiable risk factors.11-13
Initially, we assessed the already known risk factors for CM, and results showed a significantly increased risk for participants with light brown, blonde, or red hair (P<.0001); light-colored and light brown eyes (P<.0001); Fitzpatrick skin types I and II (P<.0001); positive family history of skin cancer (P=.001); the presence of solar lentigines (P<.001), freckles (P<.001), and actinic keratosis (P<.0001); and high number of nevi (P<.0001). Sunburns in adulthood (P<.001) were associated with an increased risk for CM, and our findings are in agreement with the literature.12
Besides confirming the well-known risk factors for CM, our study also showed that some foods (eg, butter, oregano) may act as important protective factors in CM. It could be argued that the increased risks associated with the well-known risk factors (eg, Fitzpatrick skin type, number of sunburns) might not be as strong and/or could be modulated by dietary factors. To further elucidate this critical issue, we analyzed our data by examining the joint relationship between dietary consumption, individual characteristics, sun exposure, and melanoma. We conducted a multivariable analysis controlling for the well-known risk factors and our findings suggest that both butter and oregano, foods that are rich in vitamins A and D, are independent and protective risk factors for melanoma.
Vitamin A (retinol) is a fat-soluble, organic compound that cannot be synthesized by humans but is necessary for normal physiological function and therefore is classified as an essential nutrient. The main source of vitamin A in the human diet is from retinyl esters, mostly from animal products such as dairy products (eg, butter) as well as from plant-based, provitamin A carotenoids (α-carotene, β-carotene) that can be converted to retinol in the intestines.14
Some case-control studies have investigated the association of vitamin A intake and CM risk, reporting mixed findings. Naldi et al15 found a notable inverse association between vitamin A intake and CM risk. Le Marchand et al16 found no inverse association for carotenoids or retinol. Kirkpatrick et al17 found no evidence of a protective effect for vitamin A or carotenoids on CM. However, the Nurses’ Health Study and the Nurses’ Health Study II reported inverse associations between CM and retinol from foods and dietary supplements.8
Dairy products such as butter contain several components considered to be potentially anticarcinogenic, such as calcium, vitamin D, butyric acid, conjugated linoleic acid, sphingolipids, and probiotic bacteria. Some studies found an inverted association between melanoma and high intake of dairy products or other dietary sources of vitamin D, while some investigators showed no association.6,18
Fortes et al18 assessed the role of diet on CM and found no protective effects of butter intake against the development of melanoma; however, a protective effect was found for carrots, which are rich in provitamin A (β-carotene) and for the regular intake of herbs rich in polyphenols (eg, rosemary). In our study, we found a protective effect against CM for butter but not for other dairy products. These findings could be explained by the high content of vitamin A in butter in comparison to other dairy products. Habitual intake of oregano also was associated with a protective effect for CM. Oregano is rich in polyphenols such as carvacrol, thymol, and rosmarinic acid, which are known for their antioxidant capacities and the inhibition of cyclooxygenase.19-21 At experimental levels, both carvacrol and thymol have been shown to inhibit the growth of melanoma cells.19,20 Rosmarinic acid, contained by both rosemary and oregano, have been shown at experimental levels to have photoprotective effects against melanoma.21
The relationship between dietary and nutritional intake and CM has a great potential that should be further explored. Tong and Young22 showed that proanthocyanidins found in grape seeds, epigallocatechin-3-gallate, resveratrol, rosmarinic acid, lycopene, and fig latex have demonstrated clear anticancer effects toward melanoma.
The strength of this study is the high response rate of both cases and controls and the use of incidence melanoma cases that decrease recall bias. A limitation of our study is that food portions were based on average portion size for each food item and therefore it can capture habitual consumption but not calculate actual nutrient intake. Misclassification of dietary exposure also could be a problem. Part of this misclassification is a result of a food frequency questionnaire being an imperfect measure of dietary history; however, we evaluated the reproducibility of the food frequency questionnaire used in this case-control study. Overall, there was a fair to good reproducibility between answers in 2 different periods (12 months apart). For example, agreement for frequency of intake of fresh herbs, tomatoes, and butter were 90.8%, 83.1%, and 83.3%, respectively.
Our sample size had sufficient statistical power to detect the effects of diet on CM.
Conclusion
Our study indicates that butter and oregano intake seem to have a protective role against the development of CM. Further studies are needed to confirm these findings.
- Gilchrest B, Eller MS, Geller AC, et al. The pathogenesis of melanoma induced by ultraviolet radiation. N Engl J Med. 1999;340:1341-1347.
- Lotti T, Bruscino N, Hercogova J, et al. Controversial issues on melanoma. Dermatol Ther. 2012;25:458-462.
- Ródenas JM, Delgado-Rodríguez M, Herranz MT, et al. Sun exposure, pigmentary traits, and risk of cutaneous malignant melanoma: a case-control study in a Mediterranean population. Cancer Causes Control. 1996;7:275-283.
- Autier P, Doré JF. Influence of sun exposures during childhood and during adulthood on melanoma risk. EEPIMEL and EORTC. Melanoma Cooperative Group. European Organization for research and treatment of cancer. Int J Cancer. 1998;77:533-537.
- Fortes C, Mastroeni S, Melchi F, et al. The association between residential pesticide use and cutaneous melanoma. Eur J Cancer. 2007;43:1066-1075.
- Jensen JD, Wing GJ, Dellavalle RP. Nutrition and melanoma prevention. Clin Dermatol. 2010;28:644-649.
- Millen AE, Tucker MA, Hartge P, et al. Diet and melanoma in a case-control study. Cancer Epidemiol Biomarkers Prev. 2004;13:1042-1051.
- Feskanich D, Willett WC, Hunter DJ, et al. Dietary intakes of vitamins A, C, and E and risk of melanoma in two cohorts of women. Br J Cancer. 2003;88:1381-1387.
- English DR, Mac Lennan R, Rivers J, et al. Epidemiological studies of melanocytic naevi: protocol for identifying and recording naevi. International Agency for Research on Cancer (IARC) internal report. No. 90/002. Lyon, France: IARC; 1990.
- Cancer preventability statistics. World Cancer Research Fund website. http://www.wcrf-uk.org/uk/preventing-cancer/cancer-preventability-statistics. Accessed May 24, 2016.
- Gandini S, Raimondi S, Gnagnarella P, et al. Vitamin D and skin cancer: a meta-analysis. Eur J Cancer. 2009;45:634-641.
- Gandini S, Sera F, Cattaruzza MS, et al. Meta-analysis of risk factors for cutaneous melanoma: II. sun exposure. Eur J Cancer. 2005;41:45-60.
- Volkovova K, Bilanicova D, Bartonova A, et al. Associations between environmental factors and incidence of cutaneous melanoma. review. Environ Health. 2012;11(11, suppl 1):S12.
- Asgari MM, Brasky TM, White E. Association of vitamin A and carotenoid intake with melanoma risk in a large prospective cohort. J Invest Dermatol. 2012;132:1573-1582.
- Naldi L, Gallus S, Tavani A, et al. Risk of melanoma and vitamin A, coffee and alcohol: a case-control study from Italy. Eur J Cancer Prev. 2004;13:503-508.
- Le Marchand L, Saltzman BS, Hankin JH, et al. Sun exposure, diet, and melanoma in Hawaii Caucasians. Am J Epidemiol. 2006;164:232-245.
- Kirkpatrick CS, White E, Lee JA. Case-control study of malignant melanoma in Washington State. II. diet, alcohol, and obesity. Am J Epidemiol. 1994;139:869-880.
- Fortes C, Mastroeni S, Melchi F, et al. A protective effect of the Mediterranean diet for cutaneous melanoma. Int J Epidemiol. 2008;37:1018-1029.
- Landa P, Kokoska L, Pribylova M, et al. In vitro anti-inflammatory activity of carvacrol: inhibitory effect on COX-2 catalyzed prostaglandin E(2) biosynthesis. Arch Pharm Res. 2009;32:75-78.
- He L, Mo H, Hadisusilo S, et al. Isoprenoids suppress the growth of murine B16 melanomas in vitro and in vivo. J Nutr. 1997;127:668-674.
- Sánchez-Campillo M, Gabaldon JA, Castillo J, et al. Rosmarinic acid, a photo-protective agent against UV and other ionizing radiations. Food Chem Toxicol. 2009;47:386-392.
- Tong LX, Young LC. Nutrition: the future of melanoma prevention? J Am Acad Dermatol. 2014;71:151-160.
The incidence of cutaneous melanoma (CM) has increased, warranting further study of new risk factors.1,2 Hereditary risk factors for CM include light-colored eyes; fair skin; light brown, blonde, or red hair; tendency to burn; high density of freckles; history of other types of skin cancer; high number of common, atypical, and/or congenital nevi; and family history of skin cancer, as well as risks related to the presence of CDKN2A, BRAF, and MC1R gene mutations. Environmental risk factors include UV exposure from sunlight or tanning beds, among others.3-5
Nutritional factors also have been suggested as possible modifiable risk factors for CM.6 Evidence from epidemiological studies show that diets rich in fruits and vegetables are associated with lower risks for several types of cancer.7,8 A growing number of studies have assessed the effects of diet and the intake of nutrients on the prevention of cancer, specifically the use of dietary supplements to protect the skin from the adverse effects of UV light.6
Preformed vitamin A (ie, retinol) is necessary for the regulation of cell differentiation and also can reduce the incidence of skin tumors in animals exposed to UV light. Certain carotenoids such as α-carotene and β-carotene are metabolized to retinol. These retinol precursors, along with antioxidant nutrients, are important components of fruits and vegetables and may account for the observed anticancer effects of these foods.8
The aim of this study was to assess the relationship between dietary intake and the risk for CM.
Methods
Participants
A case-control study was carried out between 2012 and 2013 at 3 reference centers in Porto Alegre, Brazil—Universidade Federal de Ciências da Saúde de Porto Alegre, Pontifícia Universidade Católica do Rio Grande do Sul, and Hospital de Clínicas de Porto Alegre—for the treatment of patients with CM. Enrolled patients were 18 years and older with a diagnosis of primary CM confirmed by histology. Controls were selected from patients at the same centers, and they were enrolled and matched by institution. Controls were frequency matched to cases by sex and age (+/– 5 years). Exclusion criteria for controls were those presenting with suspicious lesions and those needing radiation therapy or chemotherapy due to other diseases. The study was approved by the ethics committees of the participating centers and informed consent was obtained from all participants. A total of 191 participants (95 cases; 96 controls) were enrolled in the study.
Data Collection
After informed consent was obtained, participants were interviewed and were clinically examined by an experienced dermatologist (C.B.H. and M.M.S.). The questionnaire included sociodemographic variables, medical history, phenotypic characteristics (ie, Fitzpatrick skin type, skin/hair/eye color), family history of skin cancer, history of sunlight exposure, history of sunburns, use of artificial tanning, sunscreen use, and detailed dietary intake. Physical examination included the assessment of several melanocytic lesions (nevi, freckles/ephelides, lentigines, and café au lait spots), actinic keratoses, solar elastosis, and nonmelanocytic tumors following the International Agency for Research on Cancer (IARC) protocol.9
Using a food frequency questionnaire, participants were asked to report their usual frequency of consumption of each food from a list of 36 foods. The frequency of intake of all groups of food and beverages was defined according to the following scale: never, rarely (less than once monthly), once or twice weekly, 3 to 4 times weekly, 5 to 7 times weekly, and more than 7 times weekly. Combination of categories was based on the overall distribution among controls. Therefore, for some items such as mussels and fresh herbs, only 2 categories were used.
Statistical Analysis
A descriptive statistical analysis of the results was performed using SPSS version 20.0 with absolute and relative frequencies for the categorical variables, and mean, SD, and median for the continuous variables. The symmetry of distributions was investigated using the Kolmogorov-Smirnov test.
A t test for independent groups was applied for the continuous variables, while the Pearson χ2 test was used for the categorical variables. The Fisher exact test was used in situations in which at least 25% of the values of the cells presented an expected frequency of less than 5. Monte Carlo simulation was used when at least 1 variable had a polytomic characteristic. Odds ratio (OR) was used to estimate the strength of the association between exposures and outcome. An unconditional binary logistic regression was used to study the association between dietary variables and the risk for CM. To obtain unbiased estimates, multivariate analyses were performed controlling for 1 or more confounding variables. Using low exposure as a base category, the risks and 95% CIs were calculated for the high-exposure categories. Based on the results of bivariate analyses, variables with P≤.25 or lower were included in the models. The likelihood ratio test was used to decide which covariates should be maintained in the model. To test the goodness of fit of the models, the Hosmer-Lemeshow statistic was used.
Potential confounding factors considered in the logistic regression model were sex; age; education level; skin, hair, and eye color; Fitzpatrick skin type; presence of freckles, solar lentigines, and actinic keratosis; history of nonmelanoma skin cancer; number of melanocytic nevi; family history of skin cancer; sunburns in adulthood (≥6 episodes a year); occupational sun exposure; and history of sunscreen use in adulthood.
Results
A total of 191 participants were enrolled in the study (95 [49.7%] cases; 96 [50.3%] controls). Most participants were female (60.0% of cases; 59.4% of controls). The mean age (SD) of cases and controls was 56.8 (13.9) years and 56.5 (13.2) years, respectively. Mean body mass index (SD) did not differ between cases (27.2 [4.6]) and controls (28.2 [6.5]). Education levels of 8 years or less predominated in both groups (64.2% of cases; 57.3% of controls). No statistical difference was found for sex, age, education, or body mass index. The most frequent anatomic sites of CM were the trunk (54.7%) and arms (20.0%), and the most frequent histological type was superficial spreading (62.8%). The median Breslow thickness was 0.90 mm. Ulceration was observed in 20.9% of the cases, and 67% of participants with CM had a high mitotic rate (≥1 mitosis per square millimeter).
Phenotypic characteristics associated with an increased risk for melanoma were light brown hair (OR, 6.73; 95% CI, 3.30-14.2), blonde/red hair (OR, 21.7; 95% CI, 7.51-63.1), light-colored eyes (eg, blue, gray, green)(OR, 13.2; 95% CI, 6.13-28.7), light brown eyes (OR, 5.01; 95% CI, 2.24-11.5), and Fitzpatrick skin types I and II (OR, 7.37; 95% CI, 2.90-26.1). Family history of skin cancer was associated with an increased risk for CM (OR, 4.31; 95% CI, 1.86-10.7) as well as sunburns in adulthood (OR, 1.64; 95% CI, 1.17-1.99). Regular sunscreen use in adulthood had a 5-fold increased risk for CM compared to not using sunscreen regularly (OR, 5.6; 95% CI, 2.85-10.7). Regarding pigmented lesions, the presence of solar lentigines (OR, 4.8; 95% CI, 2.2-11.2), 60 or more nevi (OR, 5.4; 95% CI, 2.4-12.7), and freckles (OR, 3.7; 95% CI, 1.82-7.64) were all associated with an increased risk for CM. Solar elastosis (OR, 2.5; 95% CI, 1.08-5.85), actinic keratosis (OR, 9.1, 95% CI, 3.97-20.84), and occupational exposure to sun (OR, 2.57; 95% CI, 1.23-5.38) also were associated with an increased risk for melanoma.
The intake of most of the foods and beverages included in the study showed no association with CM. High frequency of butter intake (more than daily) was a protective factor for CM (OR, 0.33; 95% CI, 0.16-0.70) compared to low-frequency consumption (daily and less than daily). Consumption of mussels (OR, 0.53; 95% CI, 0.29-0.97) and oregano (OR, 0.28; 95% CI, 0.12-0.66) also were shown to be protective against CM (OR, 0.53; 95% CI, 0.29-0.97). Regarding beverages, those in the highest categories of consumption—liquor (OR, 2.12; 95% CI, 1.09-4.12) and spirits (OR, 2.23; 95% CI, 1.16-4.68)—were associated with an increased risk for CM.
To identify the relationship between CM and the consumption of some foods that were relevant on bivariate analysis, we performed a multivariate model. When adjustments were made, the association remained for butter (OR, 0.141; 95% CI, 0.032-0.613) and oregano (OR, 0.176; 95% CI, 0.042-0.735), while the risk associated with the consumption of both liquor (OR, 1.511; 95% CI, 0.39-5.90) and spirits (OR, 0.755; 95% CI, 0.130-4.393) disappeared (Table).

Comment
Observational studies show that diets rich in fruits and vegetables are associated with a lower risk for different types of cancers.7,8 According to some studies, more than 30% of cancers in adulthood could be prevented or delayed by appropriate dietary intake and physical activity.10 However, there are still limited data on some specific cancers such as CM.
Substantial differences in the incidence of CM among different populations have suggested that environmental factors may play an etiological role in the development of CM and diet could be one of the modifiable risk factors.11-13
Initially, we assessed the already known risk factors for CM, and results showed a significantly increased risk for participants with light brown, blonde, or red hair (P<.0001); light-colored and light brown eyes (P<.0001); Fitzpatrick skin types I and II (P<.0001); positive family history of skin cancer (P=.001); the presence of solar lentigines (P<.001), freckles (P<.001), and actinic keratosis (P<.0001); and high number of nevi (P<.0001). Sunburns in adulthood (P<.001) were associated with an increased risk for CM, and our findings are in agreement with the literature.12
Besides confirming the well-known risk factors for CM, our study also showed that some foods (eg, butter, oregano) may act as important protective factors in CM. It could be argued that the increased risks associated with the well-known risk factors (eg, Fitzpatrick skin type, number of sunburns) might not be as strong and/or could be modulated by dietary factors. To further elucidate this critical issue, we analyzed our data by examining the joint relationship between dietary consumption, individual characteristics, sun exposure, and melanoma. We conducted a multivariable analysis controlling for the well-known risk factors and our findings suggest that both butter and oregano, foods that are rich in vitamins A and D, are independent and protective risk factors for melanoma.
Vitamin A (retinol) is a fat-soluble, organic compound that cannot be synthesized by humans but is necessary for normal physiological function and therefore is classified as an essential nutrient. The main source of vitamin A in the human diet is from retinyl esters, mostly from animal products such as dairy products (eg, butter) as well as from plant-based, provitamin A carotenoids (α-carotene, β-carotene) that can be converted to retinol in the intestines.14
Some case-control studies have investigated the association of vitamin A intake and CM risk, reporting mixed findings. Naldi et al15 found a notable inverse association between vitamin A intake and CM risk. Le Marchand et al16 found no inverse association for carotenoids or retinol. Kirkpatrick et al17 found no evidence of a protective effect for vitamin A or carotenoids on CM. However, the Nurses’ Health Study and the Nurses’ Health Study II reported inverse associations between CM and retinol from foods and dietary supplements.8
Dairy products such as butter contain several components considered to be potentially anticarcinogenic, such as calcium, vitamin D, butyric acid, conjugated linoleic acid, sphingolipids, and probiotic bacteria. Some studies found an inverted association between melanoma and high intake of dairy products or other dietary sources of vitamin D, while some investigators showed no association.6,18
Fortes et al18 assessed the role of diet on CM and found no protective effects of butter intake against the development of melanoma; however, a protective effect was found for carrots, which are rich in provitamin A (β-carotene) and for the regular intake of herbs rich in polyphenols (eg, rosemary). In our study, we found a protective effect against CM for butter but not for other dairy products. These findings could be explained by the high content of vitamin A in butter in comparison to other dairy products. Habitual intake of oregano also was associated with a protective effect for CM. Oregano is rich in polyphenols such as carvacrol, thymol, and rosmarinic acid, which are known for their antioxidant capacities and the inhibition of cyclooxygenase.19-21 At experimental levels, both carvacrol and thymol have been shown to inhibit the growth of melanoma cells.19,20 Rosmarinic acid, contained by both rosemary and oregano, have been shown at experimental levels to have photoprotective effects against melanoma.21
The relationship between dietary and nutritional intake and CM has a great potential that should be further explored. Tong and Young22 showed that proanthocyanidins found in grape seeds, epigallocatechin-3-gallate, resveratrol, rosmarinic acid, lycopene, and fig latex have demonstrated clear anticancer effects toward melanoma.
The strength of this study is the high response rate of both cases and controls and the use of incidence melanoma cases that decrease recall bias. A limitation of our study is that food portions were based on average portion size for each food item and therefore it can capture habitual consumption but not calculate actual nutrient intake. Misclassification of dietary exposure also could be a problem. Part of this misclassification is a result of a food frequency questionnaire being an imperfect measure of dietary history; however, we evaluated the reproducibility of the food frequency questionnaire used in this case-control study. Overall, there was a fair to good reproducibility between answers in 2 different periods (12 months apart). For example, agreement for frequency of intake of fresh herbs, tomatoes, and butter were 90.8%, 83.1%, and 83.3%, respectively.
Our sample size had sufficient statistical power to detect the effects of diet on CM.
Conclusion
Our study indicates that butter and oregano intake seem to have a protective role against the development of CM. Further studies are needed to confirm these findings.
The incidence of cutaneous melanoma (CM) has increased, warranting further study of new risk factors.1,2 Hereditary risk factors for CM include light-colored eyes; fair skin; light brown, blonde, or red hair; tendency to burn; high density of freckles; history of other types of skin cancer; high number of common, atypical, and/or congenital nevi; and family history of skin cancer, as well as risks related to the presence of CDKN2A, BRAF, and MC1R gene mutations. Environmental risk factors include UV exposure from sunlight or tanning beds, among others.3-5
Nutritional factors also have been suggested as possible modifiable risk factors for CM.6 Evidence from epidemiological studies show that diets rich in fruits and vegetables are associated with lower risks for several types of cancer.7,8 A growing number of studies have assessed the effects of diet and the intake of nutrients on the prevention of cancer, specifically the use of dietary supplements to protect the skin from the adverse effects of UV light.6
Preformed vitamin A (ie, retinol) is necessary for the regulation of cell differentiation and also can reduce the incidence of skin tumors in animals exposed to UV light. Certain carotenoids such as α-carotene and β-carotene are metabolized to retinol. These retinol precursors, along with antioxidant nutrients, are important components of fruits and vegetables and may account for the observed anticancer effects of these foods.8
The aim of this study was to assess the relationship between dietary intake and the risk for CM.
Methods
Participants
A case-control study was carried out between 2012 and 2013 at 3 reference centers in Porto Alegre, Brazil—Universidade Federal de Ciências da Saúde de Porto Alegre, Pontifícia Universidade Católica do Rio Grande do Sul, and Hospital de Clínicas de Porto Alegre—for the treatment of patients with CM. Enrolled patients were 18 years and older with a diagnosis of primary CM confirmed by histology. Controls were selected from patients at the same centers, and they were enrolled and matched by institution. Controls were frequency matched to cases by sex and age (+/– 5 years). Exclusion criteria for controls were those presenting with suspicious lesions and those needing radiation therapy or chemotherapy due to other diseases. The study was approved by the ethics committees of the participating centers and informed consent was obtained from all participants. A total of 191 participants (95 cases; 96 controls) were enrolled in the study.
Data Collection
After informed consent was obtained, participants were interviewed and were clinically examined by an experienced dermatologist (C.B.H. and M.M.S.). The questionnaire included sociodemographic variables, medical history, phenotypic characteristics (ie, Fitzpatrick skin type, skin/hair/eye color), family history of skin cancer, history of sunlight exposure, history of sunburns, use of artificial tanning, sunscreen use, and detailed dietary intake. Physical examination included the assessment of several melanocytic lesions (nevi, freckles/ephelides, lentigines, and café au lait spots), actinic keratoses, solar elastosis, and nonmelanocytic tumors following the International Agency for Research on Cancer (IARC) protocol.9
Using a food frequency questionnaire, participants were asked to report their usual frequency of consumption of each food from a list of 36 foods. The frequency of intake of all groups of food and beverages was defined according to the following scale: never, rarely (less than once monthly), once or twice weekly, 3 to 4 times weekly, 5 to 7 times weekly, and more than 7 times weekly. Combination of categories was based on the overall distribution among controls. Therefore, for some items such as mussels and fresh herbs, only 2 categories were used.
Statistical Analysis
A descriptive statistical analysis of the results was performed using SPSS version 20.0 with absolute and relative frequencies for the categorical variables, and mean, SD, and median for the continuous variables. The symmetry of distributions was investigated using the Kolmogorov-Smirnov test.
A t test for independent groups was applied for the continuous variables, while the Pearson χ2 test was used for the categorical variables. The Fisher exact test was used in situations in which at least 25% of the values of the cells presented an expected frequency of less than 5. Monte Carlo simulation was used when at least 1 variable had a polytomic characteristic. Odds ratio (OR) was used to estimate the strength of the association between exposures and outcome. An unconditional binary logistic regression was used to study the association between dietary variables and the risk for CM. To obtain unbiased estimates, multivariate analyses were performed controlling for 1 or more confounding variables. Using low exposure as a base category, the risks and 95% CIs were calculated for the high-exposure categories. Based on the results of bivariate analyses, variables with P≤.25 or lower were included in the models. The likelihood ratio test was used to decide which covariates should be maintained in the model. To test the goodness of fit of the models, the Hosmer-Lemeshow statistic was used.
Potential confounding factors considered in the logistic regression model were sex; age; education level; skin, hair, and eye color; Fitzpatrick skin type; presence of freckles, solar lentigines, and actinic keratosis; history of nonmelanoma skin cancer; number of melanocytic nevi; family history of skin cancer; sunburns in adulthood (≥6 episodes a year); occupational sun exposure; and history of sunscreen use in adulthood.
Results
A total of 191 participants were enrolled in the study (95 [49.7%] cases; 96 [50.3%] controls). Most participants were female (60.0% of cases; 59.4% of controls). The mean age (SD) of cases and controls was 56.8 (13.9) years and 56.5 (13.2) years, respectively. Mean body mass index (SD) did not differ between cases (27.2 [4.6]) and controls (28.2 [6.5]). Education levels of 8 years or less predominated in both groups (64.2% of cases; 57.3% of controls). No statistical difference was found for sex, age, education, or body mass index. The most frequent anatomic sites of CM were the trunk (54.7%) and arms (20.0%), and the most frequent histological type was superficial spreading (62.8%). The median Breslow thickness was 0.90 mm. Ulceration was observed in 20.9% of the cases, and 67% of participants with CM had a high mitotic rate (≥1 mitosis per square millimeter).
Phenotypic characteristics associated with an increased risk for melanoma were light brown hair (OR, 6.73; 95% CI, 3.30-14.2), blonde/red hair (OR, 21.7; 95% CI, 7.51-63.1), light-colored eyes (eg, blue, gray, green)(OR, 13.2; 95% CI, 6.13-28.7), light brown eyes (OR, 5.01; 95% CI, 2.24-11.5), and Fitzpatrick skin types I and II (OR, 7.37; 95% CI, 2.90-26.1). Family history of skin cancer was associated with an increased risk for CM (OR, 4.31; 95% CI, 1.86-10.7) as well as sunburns in adulthood (OR, 1.64; 95% CI, 1.17-1.99). Regular sunscreen use in adulthood had a 5-fold increased risk for CM compared to not using sunscreen regularly (OR, 5.6; 95% CI, 2.85-10.7). Regarding pigmented lesions, the presence of solar lentigines (OR, 4.8; 95% CI, 2.2-11.2), 60 or more nevi (OR, 5.4; 95% CI, 2.4-12.7), and freckles (OR, 3.7; 95% CI, 1.82-7.64) were all associated with an increased risk for CM. Solar elastosis (OR, 2.5; 95% CI, 1.08-5.85), actinic keratosis (OR, 9.1, 95% CI, 3.97-20.84), and occupational exposure to sun (OR, 2.57; 95% CI, 1.23-5.38) also were associated with an increased risk for melanoma.
The intake of most of the foods and beverages included in the study showed no association with CM. High frequency of butter intake (more than daily) was a protective factor for CM (OR, 0.33; 95% CI, 0.16-0.70) compared to low-frequency consumption (daily and less than daily). Consumption of mussels (OR, 0.53; 95% CI, 0.29-0.97) and oregano (OR, 0.28; 95% CI, 0.12-0.66) also were shown to be protective against CM (OR, 0.53; 95% CI, 0.29-0.97). Regarding beverages, those in the highest categories of consumption—liquor (OR, 2.12; 95% CI, 1.09-4.12) and spirits (OR, 2.23; 95% CI, 1.16-4.68)—were associated with an increased risk for CM.
To identify the relationship between CM and the consumption of some foods that were relevant on bivariate analysis, we performed a multivariate model. When adjustments were made, the association remained for butter (OR, 0.141; 95% CI, 0.032-0.613) and oregano (OR, 0.176; 95% CI, 0.042-0.735), while the risk associated with the consumption of both liquor (OR, 1.511; 95% CI, 0.39-5.90) and spirits (OR, 0.755; 95% CI, 0.130-4.393) disappeared (Table).

Comment
Observational studies show that diets rich in fruits and vegetables are associated with a lower risk for different types of cancers.7,8 According to some studies, more than 30% of cancers in adulthood could be prevented or delayed by appropriate dietary intake and physical activity.10 However, there are still limited data on some specific cancers such as CM.
Substantial differences in the incidence of CM among different populations have suggested that environmental factors may play an etiological role in the development of CM and diet could be one of the modifiable risk factors.11-13
Initially, we assessed the already known risk factors for CM, and results showed a significantly increased risk for participants with light brown, blonde, or red hair (P<.0001); light-colored and light brown eyes (P<.0001); Fitzpatrick skin types I and II (P<.0001); positive family history of skin cancer (P=.001); the presence of solar lentigines (P<.001), freckles (P<.001), and actinic keratosis (P<.0001); and high number of nevi (P<.0001). Sunburns in adulthood (P<.001) were associated with an increased risk for CM, and our findings are in agreement with the literature.12
Besides confirming the well-known risk factors for CM, our study also showed that some foods (eg, butter, oregano) may act as important protective factors in CM. It could be argued that the increased risks associated with the well-known risk factors (eg, Fitzpatrick skin type, number of sunburns) might not be as strong and/or could be modulated by dietary factors. To further elucidate this critical issue, we analyzed our data by examining the joint relationship between dietary consumption, individual characteristics, sun exposure, and melanoma. We conducted a multivariable analysis controlling for the well-known risk factors and our findings suggest that both butter and oregano, foods that are rich in vitamins A and D, are independent and protective risk factors for melanoma.
Vitamin A (retinol) is a fat-soluble, organic compound that cannot be synthesized by humans but is necessary for normal physiological function and therefore is classified as an essential nutrient. The main source of vitamin A in the human diet is from retinyl esters, mostly from animal products such as dairy products (eg, butter) as well as from plant-based, provitamin A carotenoids (α-carotene, β-carotene) that can be converted to retinol in the intestines.14
Some case-control studies have investigated the association of vitamin A intake and CM risk, reporting mixed findings. Naldi et al15 found a notable inverse association between vitamin A intake and CM risk. Le Marchand et al16 found no inverse association for carotenoids or retinol. Kirkpatrick et al17 found no evidence of a protective effect for vitamin A or carotenoids on CM. However, the Nurses’ Health Study and the Nurses’ Health Study II reported inverse associations between CM and retinol from foods and dietary supplements.8
Dairy products such as butter contain several components considered to be potentially anticarcinogenic, such as calcium, vitamin D, butyric acid, conjugated linoleic acid, sphingolipids, and probiotic bacteria. Some studies found an inverted association between melanoma and high intake of dairy products or other dietary sources of vitamin D, while some investigators showed no association.6,18
Fortes et al18 assessed the role of diet on CM and found no protective effects of butter intake against the development of melanoma; however, a protective effect was found for carrots, which are rich in provitamin A (β-carotene) and for the regular intake of herbs rich in polyphenols (eg, rosemary). In our study, we found a protective effect against CM for butter but not for other dairy products. These findings could be explained by the high content of vitamin A in butter in comparison to other dairy products. Habitual intake of oregano also was associated with a protective effect for CM. Oregano is rich in polyphenols such as carvacrol, thymol, and rosmarinic acid, which are known for their antioxidant capacities and the inhibition of cyclooxygenase.19-21 At experimental levels, both carvacrol and thymol have been shown to inhibit the growth of melanoma cells.19,20 Rosmarinic acid, contained by both rosemary and oregano, have been shown at experimental levels to have photoprotective effects against melanoma.21
The relationship between dietary and nutritional intake and CM has a great potential that should be further explored. Tong and Young22 showed that proanthocyanidins found in grape seeds, epigallocatechin-3-gallate, resveratrol, rosmarinic acid, lycopene, and fig latex have demonstrated clear anticancer effects toward melanoma.
The strength of this study is the high response rate of both cases and controls and the use of incidence melanoma cases that decrease recall bias. A limitation of our study is that food portions were based on average portion size for each food item and therefore it can capture habitual consumption but not calculate actual nutrient intake. Misclassification of dietary exposure also could be a problem. Part of this misclassification is a result of a food frequency questionnaire being an imperfect measure of dietary history; however, we evaluated the reproducibility of the food frequency questionnaire used in this case-control study. Overall, there was a fair to good reproducibility between answers in 2 different periods (12 months apart). For example, agreement for frequency of intake of fresh herbs, tomatoes, and butter were 90.8%, 83.1%, and 83.3%, respectively.
Our sample size had sufficient statistical power to detect the effects of diet on CM.
Conclusion
Our study indicates that butter and oregano intake seem to have a protective role against the development of CM. Further studies are needed to confirm these findings.
- Gilchrest B, Eller MS, Geller AC, et al. The pathogenesis of melanoma induced by ultraviolet radiation. N Engl J Med. 1999;340:1341-1347.
- Lotti T, Bruscino N, Hercogova J, et al. Controversial issues on melanoma. Dermatol Ther. 2012;25:458-462.
- Ródenas JM, Delgado-Rodríguez M, Herranz MT, et al. Sun exposure, pigmentary traits, and risk of cutaneous malignant melanoma: a case-control study in a Mediterranean population. Cancer Causes Control. 1996;7:275-283.
- Autier P, Doré JF. Influence of sun exposures during childhood and during adulthood on melanoma risk. EEPIMEL and EORTC. Melanoma Cooperative Group. European Organization for research and treatment of cancer. Int J Cancer. 1998;77:533-537.
- Fortes C, Mastroeni S, Melchi F, et al. The association between residential pesticide use and cutaneous melanoma. Eur J Cancer. 2007;43:1066-1075.
- Jensen JD, Wing GJ, Dellavalle RP. Nutrition and melanoma prevention. Clin Dermatol. 2010;28:644-649.
- Millen AE, Tucker MA, Hartge P, et al. Diet and melanoma in a case-control study. Cancer Epidemiol Biomarkers Prev. 2004;13:1042-1051.
- Feskanich D, Willett WC, Hunter DJ, et al. Dietary intakes of vitamins A, C, and E and risk of melanoma in two cohorts of women. Br J Cancer. 2003;88:1381-1387.
- English DR, Mac Lennan R, Rivers J, et al. Epidemiological studies of melanocytic naevi: protocol for identifying and recording naevi. International Agency for Research on Cancer (IARC) internal report. No. 90/002. Lyon, France: IARC; 1990.
- Cancer preventability statistics. World Cancer Research Fund website. http://www.wcrf-uk.org/uk/preventing-cancer/cancer-preventability-statistics. Accessed May 24, 2016.
- Gandini S, Raimondi S, Gnagnarella P, et al. Vitamin D and skin cancer: a meta-analysis. Eur J Cancer. 2009;45:634-641.
- Gandini S, Sera F, Cattaruzza MS, et al. Meta-analysis of risk factors for cutaneous melanoma: II. sun exposure. Eur J Cancer. 2005;41:45-60.
- Volkovova K, Bilanicova D, Bartonova A, et al. Associations between environmental factors and incidence of cutaneous melanoma. review. Environ Health. 2012;11(11, suppl 1):S12.
- Asgari MM, Brasky TM, White E. Association of vitamin A and carotenoid intake with melanoma risk in a large prospective cohort. J Invest Dermatol. 2012;132:1573-1582.
- Naldi L, Gallus S, Tavani A, et al. Risk of melanoma and vitamin A, coffee and alcohol: a case-control study from Italy. Eur J Cancer Prev. 2004;13:503-508.
- Le Marchand L, Saltzman BS, Hankin JH, et al. Sun exposure, diet, and melanoma in Hawaii Caucasians. Am J Epidemiol. 2006;164:232-245.
- Kirkpatrick CS, White E, Lee JA. Case-control study of malignant melanoma in Washington State. II. diet, alcohol, and obesity. Am J Epidemiol. 1994;139:869-880.
- Fortes C, Mastroeni S, Melchi F, et al. A protective effect of the Mediterranean diet for cutaneous melanoma. Int J Epidemiol. 2008;37:1018-1029.
- Landa P, Kokoska L, Pribylova M, et al. In vitro anti-inflammatory activity of carvacrol: inhibitory effect on COX-2 catalyzed prostaglandin E(2) biosynthesis. Arch Pharm Res. 2009;32:75-78.
- He L, Mo H, Hadisusilo S, et al. Isoprenoids suppress the growth of murine B16 melanomas in vitro and in vivo. J Nutr. 1997;127:668-674.
- Sánchez-Campillo M, Gabaldon JA, Castillo J, et al. Rosmarinic acid, a photo-protective agent against UV and other ionizing radiations. Food Chem Toxicol. 2009;47:386-392.
- Tong LX, Young LC. Nutrition: the future of melanoma prevention? J Am Acad Dermatol. 2014;71:151-160.
- Gilchrest B, Eller MS, Geller AC, et al. The pathogenesis of melanoma induced by ultraviolet radiation. N Engl J Med. 1999;340:1341-1347.
- Lotti T, Bruscino N, Hercogova J, et al. Controversial issues on melanoma. Dermatol Ther. 2012;25:458-462.
- Ródenas JM, Delgado-Rodríguez M, Herranz MT, et al. Sun exposure, pigmentary traits, and risk of cutaneous malignant melanoma: a case-control study in a Mediterranean population. Cancer Causes Control. 1996;7:275-283.
- Autier P, Doré JF. Influence of sun exposures during childhood and during adulthood on melanoma risk. EEPIMEL and EORTC. Melanoma Cooperative Group. European Organization for research and treatment of cancer. Int J Cancer. 1998;77:533-537.
- Fortes C, Mastroeni S, Melchi F, et al. The association between residential pesticide use and cutaneous melanoma. Eur J Cancer. 2007;43:1066-1075.
- Jensen JD, Wing GJ, Dellavalle RP. Nutrition and melanoma prevention. Clin Dermatol. 2010;28:644-649.
- Millen AE, Tucker MA, Hartge P, et al. Diet and melanoma in a case-control study. Cancer Epidemiol Biomarkers Prev. 2004;13:1042-1051.
- Feskanich D, Willett WC, Hunter DJ, et al. Dietary intakes of vitamins A, C, and E and risk of melanoma in two cohorts of women. Br J Cancer. 2003;88:1381-1387.
- English DR, Mac Lennan R, Rivers J, et al. Epidemiological studies of melanocytic naevi: protocol for identifying and recording naevi. International Agency for Research on Cancer (IARC) internal report. No. 90/002. Lyon, France: IARC; 1990.
- Cancer preventability statistics. World Cancer Research Fund website. http://www.wcrf-uk.org/uk/preventing-cancer/cancer-preventability-statistics. Accessed May 24, 2016.
- Gandini S, Raimondi S, Gnagnarella P, et al. Vitamin D and skin cancer: a meta-analysis. Eur J Cancer. 2009;45:634-641.
- Gandini S, Sera F, Cattaruzza MS, et al. Meta-analysis of risk factors for cutaneous melanoma: II. sun exposure. Eur J Cancer. 2005;41:45-60.
- Volkovova K, Bilanicova D, Bartonova A, et al. Associations between environmental factors and incidence of cutaneous melanoma. review. Environ Health. 2012;11(11, suppl 1):S12.
- Asgari MM, Brasky TM, White E. Association of vitamin A and carotenoid intake with melanoma risk in a large prospective cohort. J Invest Dermatol. 2012;132:1573-1582.
- Naldi L, Gallus S, Tavani A, et al. Risk of melanoma and vitamin A, coffee and alcohol: a case-control study from Italy. Eur J Cancer Prev. 2004;13:503-508.
- Le Marchand L, Saltzman BS, Hankin JH, et al. Sun exposure, diet, and melanoma in Hawaii Caucasians. Am J Epidemiol. 2006;164:232-245.
- Kirkpatrick CS, White E, Lee JA. Case-control study of malignant melanoma in Washington State. II. diet, alcohol, and obesity. Am J Epidemiol. 1994;139:869-880.
- Fortes C, Mastroeni S, Melchi F, et al. A protective effect of the Mediterranean diet for cutaneous melanoma. Int J Epidemiol. 2008;37:1018-1029.
- Landa P, Kokoska L, Pribylova M, et al. In vitro anti-inflammatory activity of carvacrol: inhibitory effect on COX-2 catalyzed prostaglandin E(2) biosynthesis. Arch Pharm Res. 2009;32:75-78.
- He L, Mo H, Hadisusilo S, et al. Isoprenoids suppress the growth of murine B16 melanomas in vitro and in vivo. J Nutr. 1997;127:668-674.
- Sánchez-Campillo M, Gabaldon JA, Castillo J, et al. Rosmarinic acid, a photo-protective agent against UV and other ionizing radiations. Food Chem Toxicol. 2009;47:386-392.
- Tong LX, Young LC. Nutrition: the future of melanoma prevention? J Am Acad Dermatol. 2014;71:151-160.
Practice Points
- Hereditary and environmental risk factors have been identified for cutaneous melanoma (CM). Nutritional factors have been suggested as possible modifiable risk factors.
- Foods rich in vitamins A and D may be protective risk factors for CM.
Anxiety before Mohs surgery can be easily managed
ORLANDO – Assessment, education, and a judicious dose of medication can make a big difference to patients who are feeling anxious about undergoing Mohs surgery.
No studies or guidelines lay out a step-by-step management plan for anxious patients. But a little bit of common sense and empathy go a long way in easing the feeling, according to presenters at the annual meeting of the American College of Mohs Surgery.
“We don’t have an algorithm for reducing anxiety,” said Dr. Joseph Sobanko of the University of Pennsylvania, Philadelphia. “But we do have a lot of studies showing that better psychosocial health is related to better surgical outcomes.”
The lack of definitive guidance means that anxious patients might be identified and assessed by gut instinct, he continued. “Those of you who see patients probably have a gestalt technique of identifying the anxious ones. I would suggest this might not be the best way.”
Instead of guessing, he recommends assessing all patients with a validated screening tool, and dealing with anxiety proactively.
Assessing anxiety
Although anxiety assessment may not be part of a typical Mohs surgery intake, it probably should be, Dr. Sobanko said. There are a number of excellent, well-validated tools, and none of them require expertise in psychology to administer.
The Beck Anxiety Inventory is a 21-question index that takes about 10 minutes to complete. It assesses subjective, somatic, and panic-related symptoms of anxiety, and has been validated in a variety of clinical settings. It focuses quite a bit on strong physical symptoms, however, which Dr. Sobanko feels “may not be as relevant for our patients.”
The State-Trait Anxiety Inventory consists of 40 questions and takes about 15 minutes to complete. “I like it because it not only assesses how they generally feel, but how they feel at the moment,” he said. “We think it’s good and we do use it, but it takes a while to complete.”
The Hospital Anxiety and Depression Scale is short, with only 14 questions, and takes only about 5 minutes to complete. “It’s validated for hospital patients, but we are often working with patients who are less sick than that,” Dr. Sobanko said.
The Skin Cancer Index is his go-to anxiety screen for Mohs patients. It includes 15 questions that “really get to the heart of things that matter to our patients: emotional, social, and appearance issues,” he noted. Created in 2006, it was validated in a large cohort of Mohs surgery patients (Arch Facial Plast Surg. 2006;8[5]:314-318). The questions probe patients’ feelings about the seriousness of their skin cancer, its long-term health effect, and the impact the lesion – and its treatment – will have on appearance.
Tackling anxiety
Dr. Sobanko described his own, soon-to-be-published study of 104 Mohs surgery patients, who were randomized to receive a presurgical phone call to discuss anxiety, or the usual presurgical consultation. It was easy to implement the call, he said, noting that 70% were reached on the first try, and the interaction only took about 7 minutes.
Anxiety was common, with 43% reporting being anxious about the procedure. A frequent worry (25%) was whether their skin cancer would threaten their health over the long term. But both groups reported about the same reduction in anxiety after their discussion with the provider, whether it occurred over the phone or in person. After surgery, they expressed similar levels of satisfaction with the experience.
Clearly, the most effective method of dealing with patient anxiety has yet to be identified, Dr. Sobanko noted. Others are being explored, including music and educational videos.
In 2013, he and colleagues published a small study showing that music significantly reduced anxiety during Mohs surgery (Dermatol Surg. 2013 Feb;39[2]:298-305). It randomized 100 patients to surgery without music, or to listening to a playlist they had selected for themselves. Anxiety was measured using the State-Trait Anxiety Inventory and on a visual analog scale. Subjects in the music group experienced statistically significantly lower scores on both measures, Dr. Sobanko said.
A study presented at the meeting found that a preoperative education video helped as well. Dr. Sidney Smith of the Georgia Skin and Cancer Clinic, Savannah, examined the benefit of a 9-minute video created by the American College of Mohs Surgery. The video interviews patients about their experiences, describes the surgery and overall cure rates, and touches on reconstruction and follow-up.
The study comprised 200 patients; 100 saw the movie, and then completed a 24 question survey about their perception of the procedure. Almost all (94%) of those who viewed it said the video answered their questions; 85% said it relieved their fear about undergoing the surgery.
Treating anxiety
Anxiolytics can be easily employed to help ease day-of-surgery anxiety, Dr. Jerry Brewer said at the meeting. Generally speaking, the medications are safe, well-tolerated, and very effective.
“One thing we should remember, however, is that anxiolytics do not affect pain. They have no effect on pain receptors, although they may affect a patient’s memory of pain. For people who are anxious, though, this can be a really great help,” said Dr. Brewer of the Mayo Clinic, Rochester, Minn.
He favors the short-acting benzodiazepines, particularly midazolam. It has a peak concentration of 17-55 minutes, so it’s particularly well suited for shorter cases. It also has a very rapid metabolization profile, with an elimination half life of 3-7 hours.
Since midazolam has twice the affinity for the benzodiazepine receptors as does diazepam, it can be effective in relatively small doses – usually about 0.25 mg/kg. The dose should be reduced by half for elderly patients and for those with renal or hepatic failure. In those patients, the elimination half-life can be increased up to 13 hours.
The typical dose for both adults and children is 10-20 mg. “We should remember that patients who take narcotics and those who take a benzodiazepine as a sleep aide may be quite tolerant and need a higher dose,” Dr. Brewer said.
Diazepam has a peak concentration of about 2 hours, but a much longer elimination half-life – up to 48 hours in a healthy adult and up to 80 hours in an elderly person. “It’s important that patients know they’re going to have this drug in their system for a couple days. This should be part of the consenting process,” Dr. Brewer pointed out.
Lorazepam has a peak concentration of about 2 hours as well, but a shorter half-life of 12-18 hours. That can be prolonged by 75% in patients with renal problems.
With the right clinical supervision, these medications are very safe, he said. “We treat about 800 patients per year with these and have data on about 12,000. Of those, we have had very few problems. Two have fallen out of bed. One patient wrote and said he was discharged too early, as he was very tired. One person fell and hit his head in the bathroom. One was sedated enough to need a sternal rub to improve responsiveness. And one gentleman enjoyed the medication so much that when the nurse left the room for a moment he grabbed the rest of the dose and drank it.”
Safe discharge is crucial when using anxiolytics, he added. “They absolutely cannot drive themselves home and they cannot go back to work. We make sure there is a reliable person to stay with the patient for at least 4 hours after discharge.”
Dr. Brewer does not discharge any patient until that person displays a zero rating on the Richmond Agitation-Sedation Scale (RASS) sedation scale. “That means he is awake, alert, and calmly interacting with you.”
Neither Dr. Sobanko nor Dr. Brewer had any financial declarations.
ORLANDO – Assessment, education, and a judicious dose of medication can make a big difference to patients who are feeling anxious about undergoing Mohs surgery.
No studies or guidelines lay out a step-by-step management plan for anxious patients. But a little bit of common sense and empathy go a long way in easing the feeling, according to presenters at the annual meeting of the American College of Mohs Surgery.
“We don’t have an algorithm for reducing anxiety,” said Dr. Joseph Sobanko of the University of Pennsylvania, Philadelphia. “But we do have a lot of studies showing that better psychosocial health is related to better surgical outcomes.”
The lack of definitive guidance means that anxious patients might be identified and assessed by gut instinct, he continued. “Those of you who see patients probably have a gestalt technique of identifying the anxious ones. I would suggest this might not be the best way.”
Instead of guessing, he recommends assessing all patients with a validated screening tool, and dealing with anxiety proactively.
Assessing anxiety
Although anxiety assessment may not be part of a typical Mohs surgery intake, it probably should be, Dr. Sobanko said. There are a number of excellent, well-validated tools, and none of them require expertise in psychology to administer.
The Beck Anxiety Inventory is a 21-question index that takes about 10 minutes to complete. It assesses subjective, somatic, and panic-related symptoms of anxiety, and has been validated in a variety of clinical settings. It focuses quite a bit on strong physical symptoms, however, which Dr. Sobanko feels “may not be as relevant for our patients.”
The State-Trait Anxiety Inventory consists of 40 questions and takes about 15 minutes to complete. “I like it because it not only assesses how they generally feel, but how they feel at the moment,” he said. “We think it’s good and we do use it, but it takes a while to complete.”
The Hospital Anxiety and Depression Scale is short, with only 14 questions, and takes only about 5 minutes to complete. “It’s validated for hospital patients, but we are often working with patients who are less sick than that,” Dr. Sobanko said.

The Skin Cancer Index is his go-to anxiety screen for Mohs patients. It includes 15 questions that “really get to the heart of things that matter to our patients: emotional, social, and appearance issues,” he noted. Created in 2006, it was validated in a large cohort of Mohs surgery patients (Arch Facial Plast Surg. 2006;8[5]:314-318). The questions probe patients’ feelings about the seriousness of their skin cancer, its long-term health effect, and the impact the lesion – and its treatment – will have on appearance.
Tackling anxiety
Dr. Sobanko described his own, soon-to-be-published study of 104 Mohs surgery patients, who were randomized to receive a presurgical phone call to discuss anxiety, or the usual presurgical consultation. It was easy to implement the call, he said, noting that 70% were reached on the first try, and the interaction only took about 7 minutes.
Anxiety was common, with 43% reporting being anxious about the procedure. A frequent worry (25%) was whether their skin cancer would threaten their health over the long term. But both groups reported about the same reduction in anxiety after their discussion with the provider, whether it occurred over the phone or in person. After surgery, they expressed similar levels of satisfaction with the experience.
Clearly, the most effective method of dealing with patient anxiety has yet to be identified, Dr. Sobanko noted. Others are being explored, including music and educational videos.
In 2013, he and colleagues published a small study showing that music significantly reduced anxiety during Mohs surgery (Dermatol Surg. 2013 Feb;39[2]:298-305). It randomized 100 patients to surgery without music, or to listening to a playlist they had selected for themselves. Anxiety was measured using the State-Trait Anxiety Inventory and on a visual analog scale. Subjects in the music group experienced statistically significantly lower scores on both measures, Dr. Sobanko said.
A study presented at the meeting found that a preoperative education video helped as well. Dr. Sidney Smith of the Georgia Skin and Cancer Clinic, Savannah, examined the benefit of a 9-minute video created by the American College of Mohs Surgery. The video interviews patients about their experiences, describes the surgery and overall cure rates, and touches on reconstruction and follow-up.
The study comprised 200 patients; 100 saw the movie, and then completed a 24 question survey about their perception of the procedure. Almost all (94%) of those who viewed it said the video answered their questions; 85% said it relieved their fear about undergoing the surgery.
Treating anxiety
Anxiolytics can be easily employed to help ease day-of-surgery anxiety, Dr. Jerry Brewer said at the meeting. Generally speaking, the medications are safe, well-tolerated, and very effective.
“One thing we should remember, however, is that anxiolytics do not affect pain. They have no effect on pain receptors, although they may affect a patient’s memory of pain. For people who are anxious, though, this can be a really great help,” said Dr. Brewer of the Mayo Clinic, Rochester, Minn.
He favors the short-acting benzodiazepines, particularly midazolam. It has a peak concentration of 17-55 minutes, so it’s particularly well suited for shorter cases. It also has a very rapid metabolization profile, with an elimination half life of 3-7 hours.
Since midazolam has twice the affinity for the benzodiazepine receptors as does diazepam, it can be effective in relatively small doses – usually about 0.25 mg/kg. The dose should be reduced by half for elderly patients and for those with renal or hepatic failure. In those patients, the elimination half-life can be increased up to 13 hours.
The typical dose for both adults and children is 10-20 mg. “We should remember that patients who take narcotics and those who take a benzodiazepine as a sleep aide may be quite tolerant and need a higher dose,” Dr. Brewer said.
Diazepam has a peak concentration of about 2 hours, but a much longer elimination half-life – up to 48 hours in a healthy adult and up to 80 hours in an elderly person. “It’s important that patients know they’re going to have this drug in their system for a couple days. This should be part of the consenting process,” Dr. Brewer pointed out.
Lorazepam has a peak concentration of about 2 hours as well, but a shorter half-life of 12-18 hours. That can be prolonged by 75% in patients with renal problems.
With the right clinical supervision, these medications are very safe, he said. “We treat about 800 patients per year with these and have data on about 12,000. Of those, we have had very few problems. Two have fallen out of bed. One patient wrote and said he was discharged too early, as he was very tired. One person fell and hit his head in the bathroom. One was sedated enough to need a sternal rub to improve responsiveness. And one gentleman enjoyed the medication so much that when the nurse left the room for a moment he grabbed the rest of the dose and drank it.”
Safe discharge is crucial when using anxiolytics, he added. “They absolutely cannot drive themselves home and they cannot go back to work. We make sure there is a reliable person to stay with the patient for at least 4 hours after discharge.”
Dr. Brewer does not discharge any patient until that person displays a zero rating on the Richmond Agitation-Sedation Scale (RASS) sedation scale. “That means he is awake, alert, and calmly interacting with you.”
Neither Dr. Sobanko nor Dr. Brewer had any financial declarations.
ORLANDO – Assessment, education, and a judicious dose of medication can make a big difference to patients who are feeling anxious about undergoing Mohs surgery.
No studies or guidelines lay out a step-by-step management plan for anxious patients. But a little bit of common sense and empathy go a long way in easing the feeling, according to presenters at the annual meeting of the American College of Mohs Surgery.
“We don’t have an algorithm for reducing anxiety,” said Dr. Joseph Sobanko of the University of Pennsylvania, Philadelphia. “But we do have a lot of studies showing that better psychosocial health is related to better surgical outcomes.”
The lack of definitive guidance means that anxious patients might be identified and assessed by gut instinct, he continued. “Those of you who see patients probably have a gestalt technique of identifying the anxious ones. I would suggest this might not be the best way.”
Instead of guessing, he recommends assessing all patients with a validated screening tool, and dealing with anxiety proactively.
Assessing anxiety
Although anxiety assessment may not be part of a typical Mohs surgery intake, it probably should be, Dr. Sobanko said. There are a number of excellent, well-validated tools, and none of them require expertise in psychology to administer.
The Beck Anxiety Inventory is a 21-question index that takes about 10 minutes to complete. It assesses subjective, somatic, and panic-related symptoms of anxiety, and has been validated in a variety of clinical settings. It focuses quite a bit on strong physical symptoms, however, which Dr. Sobanko feels “may not be as relevant for our patients.”
The State-Trait Anxiety Inventory consists of 40 questions and takes about 15 minutes to complete. “I like it because it not only assesses how they generally feel, but how they feel at the moment,” he said. “We think it’s good and we do use it, but it takes a while to complete.”
The Hospital Anxiety and Depression Scale is short, with only 14 questions, and takes only about 5 minutes to complete. “It’s validated for hospital patients, but we are often working with patients who are less sick than that,” Dr. Sobanko said.

The Skin Cancer Index is his go-to anxiety screen for Mohs patients. It includes 15 questions that “really get to the heart of things that matter to our patients: emotional, social, and appearance issues,” he noted. Created in 2006, it was validated in a large cohort of Mohs surgery patients (Arch Facial Plast Surg. 2006;8[5]:314-318). The questions probe patients’ feelings about the seriousness of their skin cancer, its long-term health effect, and the impact the lesion – and its treatment – will have on appearance.
Tackling anxiety
Dr. Sobanko described his own, soon-to-be-published study of 104 Mohs surgery patients, who were randomized to receive a presurgical phone call to discuss anxiety, or the usual presurgical consultation. It was easy to implement the call, he said, noting that 70% were reached on the first try, and the interaction only took about 7 minutes.
Anxiety was common, with 43% reporting being anxious about the procedure. A frequent worry (25%) was whether their skin cancer would threaten their health over the long term. But both groups reported about the same reduction in anxiety after their discussion with the provider, whether it occurred over the phone or in person. After surgery, they expressed similar levels of satisfaction with the experience.
Clearly, the most effective method of dealing with patient anxiety has yet to be identified, Dr. Sobanko noted. Others are being explored, including music and educational videos.
In 2013, he and colleagues published a small study showing that music significantly reduced anxiety during Mohs surgery (Dermatol Surg. 2013 Feb;39[2]:298-305). It randomized 100 patients to surgery without music, or to listening to a playlist they had selected for themselves. Anxiety was measured using the State-Trait Anxiety Inventory and on a visual analog scale. Subjects in the music group experienced statistically significantly lower scores on both measures, Dr. Sobanko said.
A study presented at the meeting found that a preoperative education video helped as well. Dr. Sidney Smith of the Georgia Skin and Cancer Clinic, Savannah, examined the benefit of a 9-minute video created by the American College of Mohs Surgery. The video interviews patients about their experiences, describes the surgery and overall cure rates, and touches on reconstruction and follow-up.
The study comprised 200 patients; 100 saw the movie, and then completed a 24 question survey about their perception of the procedure. Almost all (94%) of those who viewed it said the video answered their questions; 85% said it relieved their fear about undergoing the surgery.
Treating anxiety
Anxiolytics can be easily employed to help ease day-of-surgery anxiety, Dr. Jerry Brewer said at the meeting. Generally speaking, the medications are safe, well-tolerated, and very effective.
“One thing we should remember, however, is that anxiolytics do not affect pain. They have no effect on pain receptors, although they may affect a patient’s memory of pain. For people who are anxious, though, this can be a really great help,” said Dr. Brewer of the Mayo Clinic, Rochester, Minn.
He favors the short-acting benzodiazepines, particularly midazolam. It has a peak concentration of 17-55 minutes, so it’s particularly well suited for shorter cases. It also has a very rapid metabolization profile, with an elimination half life of 3-7 hours.
Since midazolam has twice the affinity for the benzodiazepine receptors as does diazepam, it can be effective in relatively small doses – usually about 0.25 mg/kg. The dose should be reduced by half for elderly patients and for those with renal or hepatic failure. In those patients, the elimination half-life can be increased up to 13 hours.
The typical dose for both adults and children is 10-20 mg. “We should remember that patients who take narcotics and those who take a benzodiazepine as a sleep aide may be quite tolerant and need a higher dose,” Dr. Brewer said.
Diazepam has a peak concentration of about 2 hours, but a much longer elimination half-life – up to 48 hours in a healthy adult and up to 80 hours in an elderly person. “It’s important that patients know they’re going to have this drug in their system for a couple days. This should be part of the consenting process,” Dr. Brewer pointed out.
Lorazepam has a peak concentration of about 2 hours as well, but a shorter half-life of 12-18 hours. That can be prolonged by 75% in patients with renal problems.
With the right clinical supervision, these medications are very safe, he said. “We treat about 800 patients per year with these and have data on about 12,000. Of those, we have had very few problems. Two have fallen out of bed. One patient wrote and said he was discharged too early, as he was very tired. One person fell and hit his head in the bathroom. One was sedated enough to need a sternal rub to improve responsiveness. And one gentleman enjoyed the medication so much that when the nurse left the room for a moment he grabbed the rest of the dose and drank it.”
Safe discharge is crucial when using anxiolytics, he added. “They absolutely cannot drive themselves home and they cannot go back to work. We make sure there is a reliable person to stay with the patient for at least 4 hours after discharge.”
Dr. Brewer does not discharge any patient until that person displays a zero rating on the Richmond Agitation-Sedation Scale (RASS) sedation scale. “That means he is awake, alert, and calmly interacting with you.”
Neither Dr. Sobanko nor Dr. Brewer had any financial declarations.
EXPERT ANALYSIS FROM THE ACMS ANNUAL MEETING
Investigational CDK4/6 inhibitor shows activity, less toxicity
Abemaciclib, a CDK4/6 inhibitor, showed durable clinical activity when given as continuous single-agent therapy to patients with advanced cancer, including breast cancer and non–small-cell lung cancer, according to investigators.
Neutropenia was rarely observed in patients treated with abemaciclib, the toxicity observed in some patients who receive the only Food and Drug Administration–approved CDK4/6 inhibitor, palbociclib.
“Abemaciclib is a small-molecule inhibitor of CDK4 and CDK6 that is structurally distinct from other dual inhibitors (such as palbociclib and ribociclib) and notably exhibits greater selectivity for CDK4 compared with CDK6,” wrote Dr. Amita Patnaik of South Texas Accelerated Research Therapeutics and her associates (Cancer Discov. 2016 May 23. doi: 10.1158/2159-8290.CD-16-0095).
Furthermore, preclinical models indicate that the drug can cross the blood-brain barrier, suggesting potential efficacy against primary and metastatic tumors involving the central nervous system, they said.
A total of 225 patients with various types of advanced cancers were enrolled in this multicohort phase I study (dose escalation, n = 33; single-agent abemaciclib therapy for breast cancer, n = 47; non–small-cell lung cancer, n = 68; glioblastoma, n = 17; melanoma, n = 26; colorectal cancer, n = 15; abemaciclib plus fulvestrant combination therapy for hormone receptor–positive breast cancer, n = 19). Abemaciclib was given orally to all patients.
Neither dose-limiting toxicity nor maximum tolerated dose was reached in patients treated at levels of 50 mg, 100 mg, 150 mg, or 225 mg once daily. The maximum tolerated dose was 200 mg for patients treated with abemaciclib twice daily.
In the single-agent breast cancer cohort, the disease control rate was 81% for hormone receptor–positive (HR-positive) tumors, 33% for HR-negative tumors, 100% for HR-positive HER2-positive tumors, 72% for HR-positive HER2-negative tumors, and 70% overall. The response rate was 31% for HR-positive tumors, 0% for HR-negative tumors, 36% for HR-positive HER2-positive tumors, 28% for HR-positive HER2-negative tumors, and 23% overall.
The overall response rate was 21% for breast cancer patients receiving abemaciclib plus fulvestrant.
Among the 68 patients with non–small-cell lung cancer, 2 had a partial response and 31 had stable disease. Of the 26 patients with melanoma, 1 had a partial response and 6 had stable disease. Of the 17 patients with glioblastoma, 3 had stable disease.
Overall, there were no study-related deaths. Diarrhea, nausea, and fatigue were the most common adverse events; all were reversible.
Neutropenia was observed in 39 patients (23% of 173 patients in the single-agent tumor-specific cohort) – 2 were grade 4 events. Grade 3 neutropenia occurred in 6 patients (32% of 19 patients with HR-positive breast cancer receiving combination therapy with abemaciclib plus fulvestrant).
“Previous reports have identified neutropenia as an adverse event associated with dual inhibition of CDK4 and CDK6. However, abemaciclib given as a single agent on a continuous schedule in the tumor-specific cohorts was associated with an acceptable incidence of investigator-reported grade 3 (9%, 16 of 173 patients) or grade 4 (1%, 2 of 173 patients) neutropenia,” wrote the investigators.
“In summary, the results of this clinical trial demonstrate the safety and antitumor activity of abemaciclib as a single agent and support its further development both as monotherapy and in rational combinations. Furthermore, these findings validate CDK4 and CDK6 as anticancer drug targets and translate preclinical predictions regarding therapeutic targeting of cell-cycle derangements in cancer into clinical efficacy,”they wrote.
Eli Lilly funded the study. Eight investigators reported serving in advisory roles, having ownership or stock interest in, or receiving financial compensation from multiple companies including Eli Lilly.
On Twitter @JessCraig_OP
Abemaciclib, a CDK4/6 inhibitor, showed durable clinical activity when given as continuous single-agent therapy to patients with advanced cancer, including breast cancer and non–small-cell lung cancer, according to investigators.
Neutropenia was rarely observed in patients treated with abemaciclib, the toxicity observed in some patients who receive the only Food and Drug Administration–approved CDK4/6 inhibitor, palbociclib.
“Abemaciclib is a small-molecule inhibitor of CDK4 and CDK6 that is structurally distinct from other dual inhibitors (such as palbociclib and ribociclib) and notably exhibits greater selectivity for CDK4 compared with CDK6,” wrote Dr. Amita Patnaik of South Texas Accelerated Research Therapeutics and her associates (Cancer Discov. 2016 May 23. doi: 10.1158/2159-8290.CD-16-0095).
Furthermore, preclinical models indicate that the drug can cross the blood-brain barrier, suggesting potential efficacy against primary and metastatic tumors involving the central nervous system, they said.
A total of 225 patients with various types of advanced cancers were enrolled in this multicohort phase I study (dose escalation, n = 33; single-agent abemaciclib therapy for breast cancer, n = 47; non–small-cell lung cancer, n = 68; glioblastoma, n = 17; melanoma, n = 26; colorectal cancer, n = 15; abemaciclib plus fulvestrant combination therapy for hormone receptor–positive breast cancer, n = 19). Abemaciclib was given orally to all patients.
Neither dose-limiting toxicity nor maximum tolerated dose was reached in patients treated at levels of 50 mg, 100 mg, 150 mg, or 225 mg once daily. The maximum tolerated dose was 200 mg for patients treated with abemaciclib twice daily.
In the single-agent breast cancer cohort, the disease control rate was 81% for hormone receptor–positive (HR-positive) tumors, 33% for HR-negative tumors, 100% for HR-positive HER2-positive tumors, 72% for HR-positive HER2-negative tumors, and 70% overall. The response rate was 31% for HR-positive tumors, 0% for HR-negative tumors, 36% for HR-positive HER2-positive tumors, 28% for HR-positive HER2-negative tumors, and 23% overall.
The overall response rate was 21% for breast cancer patients receiving abemaciclib plus fulvestrant.
Among the 68 patients with non–small-cell lung cancer, 2 had a partial response and 31 had stable disease. Of the 26 patients with melanoma, 1 had a partial response and 6 had stable disease. Of the 17 patients with glioblastoma, 3 had stable disease.
Overall, there were no study-related deaths. Diarrhea, nausea, and fatigue were the most common adverse events; all were reversible.
Neutropenia was observed in 39 patients (23% of 173 patients in the single-agent tumor-specific cohort) – 2 were grade 4 events. Grade 3 neutropenia occurred in 6 patients (32% of 19 patients with HR-positive breast cancer receiving combination therapy with abemaciclib plus fulvestrant).
“Previous reports have identified neutropenia as an adverse event associated with dual inhibition of CDK4 and CDK6. However, abemaciclib given as a single agent on a continuous schedule in the tumor-specific cohorts was associated with an acceptable incidence of investigator-reported grade 3 (9%, 16 of 173 patients) or grade 4 (1%, 2 of 173 patients) neutropenia,” wrote the investigators.
“In summary, the results of this clinical trial demonstrate the safety and antitumor activity of abemaciclib as a single agent and support its further development both as monotherapy and in rational combinations. Furthermore, these findings validate CDK4 and CDK6 as anticancer drug targets and translate preclinical predictions regarding therapeutic targeting of cell-cycle derangements in cancer into clinical efficacy,”they wrote.
Eli Lilly funded the study. Eight investigators reported serving in advisory roles, having ownership or stock interest in, or receiving financial compensation from multiple companies including Eli Lilly.
On Twitter @JessCraig_OP
Abemaciclib, a CDK4/6 inhibitor, showed durable clinical activity when given as continuous single-agent therapy to patients with advanced cancer, including breast cancer and non–small-cell lung cancer, according to investigators.
Neutropenia was rarely observed in patients treated with abemaciclib, the toxicity observed in some patients who receive the only Food and Drug Administration–approved CDK4/6 inhibitor, palbociclib.
“Abemaciclib is a small-molecule inhibitor of CDK4 and CDK6 that is structurally distinct from other dual inhibitors (such as palbociclib and ribociclib) and notably exhibits greater selectivity for CDK4 compared with CDK6,” wrote Dr. Amita Patnaik of South Texas Accelerated Research Therapeutics and her associates (Cancer Discov. 2016 May 23. doi: 10.1158/2159-8290.CD-16-0095).
Furthermore, preclinical models indicate that the drug can cross the blood-brain barrier, suggesting potential efficacy against primary and metastatic tumors involving the central nervous system, they said.
A total of 225 patients with various types of advanced cancers were enrolled in this multicohort phase I study (dose escalation, n = 33; single-agent abemaciclib therapy for breast cancer, n = 47; non–small-cell lung cancer, n = 68; glioblastoma, n = 17; melanoma, n = 26; colorectal cancer, n = 15; abemaciclib plus fulvestrant combination therapy for hormone receptor–positive breast cancer, n = 19). Abemaciclib was given orally to all patients.
Neither dose-limiting toxicity nor maximum tolerated dose was reached in patients treated at levels of 50 mg, 100 mg, 150 mg, or 225 mg once daily. The maximum tolerated dose was 200 mg for patients treated with abemaciclib twice daily.
In the single-agent breast cancer cohort, the disease control rate was 81% for hormone receptor–positive (HR-positive) tumors, 33% for HR-negative tumors, 100% for HR-positive HER2-positive tumors, 72% for HR-positive HER2-negative tumors, and 70% overall. The response rate was 31% for HR-positive tumors, 0% for HR-negative tumors, 36% for HR-positive HER2-positive tumors, 28% for HR-positive HER2-negative tumors, and 23% overall.
The overall response rate was 21% for breast cancer patients receiving abemaciclib plus fulvestrant.
Among the 68 patients with non–small-cell lung cancer, 2 had a partial response and 31 had stable disease. Of the 26 patients with melanoma, 1 had a partial response and 6 had stable disease. Of the 17 patients with glioblastoma, 3 had stable disease.
Overall, there were no study-related deaths. Diarrhea, nausea, and fatigue were the most common adverse events; all were reversible.
Neutropenia was observed in 39 patients (23% of 173 patients in the single-agent tumor-specific cohort) – 2 were grade 4 events. Grade 3 neutropenia occurred in 6 patients (32% of 19 patients with HR-positive breast cancer receiving combination therapy with abemaciclib plus fulvestrant).
“Previous reports have identified neutropenia as an adverse event associated with dual inhibition of CDK4 and CDK6. However, abemaciclib given as a single agent on a continuous schedule in the tumor-specific cohorts was associated with an acceptable incidence of investigator-reported grade 3 (9%, 16 of 173 patients) or grade 4 (1%, 2 of 173 patients) neutropenia,” wrote the investigators.
“In summary, the results of this clinical trial demonstrate the safety and antitumor activity of abemaciclib as a single agent and support its further development both as monotherapy and in rational combinations. Furthermore, these findings validate CDK4 and CDK6 as anticancer drug targets and translate preclinical predictions regarding therapeutic targeting of cell-cycle derangements in cancer into clinical efficacy,”they wrote.
Eli Lilly funded the study. Eight investigators reported serving in advisory roles, having ownership or stock interest in, or receiving financial compensation from multiple companies including Eli Lilly.
On Twitter @JessCraig_OP
FROM CANCER DISCOVERY
Key clinical point: A phase I trial indicates that abemaciclib is safe and shows activity in treating patients with advanced breast and other cancers.
Major finding: In the single-agent breast cancer cohort, the overall disease control rate was 70%. Incidence of neutropenia was 9% for grade 3 and 1% for grade 4.
Data source: A multicenter phase I dose-escalation and tumor-specific cohort study of 225 patients with advanced cancers.
Disclosures: Eli Lilly funded the study. Eight investigators reported serving in advisory roles, having ownership or stock interest in, or receiving financial compensation from multiple companies including Eli Lilly.
Gene expression test predicts melanoma metastasis
ORLANDO – A commercially available melanoma gene expression test had a negative predictive value of 98% for metastasis, but also correctly identified patients at very high risk of disease spread.
In a retrospective study, the DecisionDx-Melanoma test, which examines 28 risk genes, showed that patients stratified as high risk with the test were 22 times more likely to develop metastatic disease than were those stratified as low risk, Dr. Bradley Greenhaw said at the annual meeting of the American College of Mohs Surgery. The test classifies patients with stage I and stage II melanoma as having a low risk (class 1) or a high risk (class 2) of metastasis within 5 years.
“Our mean follow-up time is less than half of the 5-year risk this test is able to predict,” said Dr. Greenhaw, a Mohs surgeon in Tupelo, Miss. “So if the test is performing as it’s supposed to, there would be even more events – you would see this gap widen even more as time passes. Seeing this with our limited follow-up period is pretty significant.”
The test also added valuable prognostic information to the American Joint Committee on Cancer (AJCC) staging method, he said. “For example, based on AJCC staging alone, we would expect to see a 5-year metastatic rate of 5%-10% for stage I melanomas. We saw less than a 1% rate. That is a pretty significant negative predictive value.”
The genes in the panel were selected from those examined in eight different genetic risk studies of cutaneous melanomas. The panel divides tumors into the high- and low-risk groups. A validation study published last year concluded that the 5-year disease-free survival rates were 97% for class 1 tumors and 31% for class 2 tumors (Clin Cancer Res 2015;21[1];175-83).
In the retrospective study, Dr. Greenhaw and his associates examined the test’s accuracy in a cohort of 256 patients treated at his center. The mean follow-up time was 23 months, although some patients have completed 5 years of follow-up.
Of the tumor blocks analyzed, 214 were class 1 and 42 were class 2. He noted significant differences between the class 1 and class 2 tumors, including mean patient age (66 years vs. 74 years, respectively), mean Breslow’s depth (0.7 mm vs. 2.3 mm), mitotic rate (0.8 vs. 3.3), and ulceration (4% vs. 41%).
Over the mean 2-year follow-up time, three class 1 tumors and 10 class 2 tumors metastasized (1.4% vs. 23.8%). The test’s negative predictive value was 98%; class 2 tumors were 22 times more likely to metastasize. “About 77% of metastatic tumors were correctly identified as high risk by this test,” Dr. Greenhaw said in an interview. “For comparison, more than 80% were correctly identified in the validation study, so our study was in line with it.”
The test showed a link between metastatic risk and ulceration at presentation. Of the class 1 tumors, 4% were ulcerated, compared to 41% of class 2 tumors; class 2 tumors were 16 times more likely to be ulcerated. Among the ulcerated lesions, metastasis occurred in 11% of class 1 and 41% of class 2 tumors.
Dr. Greenhaw also compared the gene test’s characterization of tumors to the AJCC staging.
Of the 214 class 1 tumors, 94% were stage IA or IB. Among these, there was no metastasis. Of the class 2 tumors, 17% were stage IA and 26% were stage IB. Interestingly, Dr. Greenhaw said, despite the high-risk genetic signature, none of these tumors metastasized. “This could have been because although the gene expression showed them to be associated with a high risk of metastases, the tumors were completely excised prior to the metastatic event, or because our follow-up is still relatively short,” he noted.
In the class 1 group, 4% of the tumors were stage IIA and 2% were stage IIB. Of these 13 tumors, two metastasized. “This 15% metastatic rate is a lower rate than we would predict by the AJCC staging,” Dr. Greenhaw said.
In the class 2 groups, 24% were stage IIA, 26% were stage IIB, and 7% were stage IIC. Of these 24 tumors, 10 metastasized. “This is a higher rate than the AJCC staging would predict, and it occurred rather quickly, with our abbreviated follow-up period,” Dr. Greenhaw noted.
Based on these findings, Dr. Greenhaw suggested a clinical management algorithm:
• All patients with invasive melanoma should be offered the gene expression profiling.
• Those with class 1 tumors have a high chance of cure and a low risk of metastasis. They can be reasonably managed with clinical skin and nodal exams every 6 months for 2 years, and then annually.
• Those with class 2 tumors have a much higher risk of metastatic disease. They should receive clinical skin and nodal exams every 3 months for 2 years, then every 6-12 months for 5 years. After that, a yearly exam should suffice.
Tests like this are an important advance in managing melanoma, Dr. Greenhaw said. “I think this is where the future of melanoma prognosis is. The keys are found in the genes and DNA of these tumors. These tests are being used in other types of cancer and I think it’s where we need to go in our field as well.”
Dr. Greenhaw has no financial disclosures to report.
ORLANDO – A commercially available melanoma gene expression test had a negative predictive value of 98% for metastasis, but also correctly identified patients at very high risk of disease spread.
In a retrospective study, the DecisionDx-Melanoma test, which examines 28 risk genes, showed that patients stratified as high risk with the test were 22 times more likely to develop metastatic disease than were those stratified as low risk, Dr. Bradley Greenhaw said at the annual meeting of the American College of Mohs Surgery. The test classifies patients with stage I and stage II melanoma as having a low risk (class 1) or a high risk (class 2) of metastasis within 5 years.
“Our mean follow-up time is less than half of the 5-year risk this test is able to predict,” said Dr. Greenhaw, a Mohs surgeon in Tupelo, Miss. “So if the test is performing as it’s supposed to, there would be even more events – you would see this gap widen even more as time passes. Seeing this with our limited follow-up period is pretty significant.”
The test also added valuable prognostic information to the American Joint Committee on Cancer (AJCC) staging method, he said. “For example, based on AJCC staging alone, we would expect to see a 5-year metastatic rate of 5%-10% for stage I melanomas. We saw less than a 1% rate. That is a pretty significant negative predictive value.”
The genes in the panel were selected from those examined in eight different genetic risk studies of cutaneous melanomas. The panel divides tumors into the high- and low-risk groups. A validation study published last year concluded that the 5-year disease-free survival rates were 97% for class 1 tumors and 31% for class 2 tumors (Clin Cancer Res 2015;21[1];175-83).
In the retrospective study, Dr. Greenhaw and his associates examined the test’s accuracy in a cohort of 256 patients treated at his center. The mean follow-up time was 23 months, although some patients have completed 5 years of follow-up.
Of the tumor blocks analyzed, 214 were class 1 and 42 were class 2. He noted significant differences between the class 1 and class 2 tumors, including mean patient age (66 years vs. 74 years, respectively), mean Breslow’s depth (0.7 mm vs. 2.3 mm), mitotic rate (0.8 vs. 3.3), and ulceration (4% vs. 41%).
Over the mean 2-year follow-up time, three class 1 tumors and 10 class 2 tumors metastasized (1.4% vs. 23.8%). The test’s negative predictive value was 98%; class 2 tumors were 22 times more likely to metastasize. “About 77% of metastatic tumors were correctly identified as high risk by this test,” Dr. Greenhaw said in an interview. “For comparison, more than 80% were correctly identified in the validation study, so our study was in line with it.”
The test showed a link between metastatic risk and ulceration at presentation. Of the class 1 tumors, 4% were ulcerated, compared to 41% of class 2 tumors; class 2 tumors were 16 times more likely to be ulcerated. Among the ulcerated lesions, metastasis occurred in 11% of class 1 and 41% of class 2 tumors.
Dr. Greenhaw also compared the gene test’s characterization of tumors to the AJCC staging.
Of the 214 class 1 tumors, 94% were stage IA or IB. Among these, there was no metastasis. Of the class 2 tumors, 17% were stage IA and 26% were stage IB. Interestingly, Dr. Greenhaw said, despite the high-risk genetic signature, none of these tumors metastasized. “This could have been because although the gene expression showed them to be associated with a high risk of metastases, the tumors were completely excised prior to the metastatic event, or because our follow-up is still relatively short,” he noted.
In the class 1 group, 4% of the tumors were stage IIA and 2% were stage IIB. Of these 13 tumors, two metastasized. “This 15% metastatic rate is a lower rate than we would predict by the AJCC staging,” Dr. Greenhaw said.
In the class 2 groups, 24% were stage IIA, 26% were stage IIB, and 7% were stage IIC. Of these 24 tumors, 10 metastasized. “This is a higher rate than the AJCC staging would predict, and it occurred rather quickly, with our abbreviated follow-up period,” Dr. Greenhaw noted.
Based on these findings, Dr. Greenhaw suggested a clinical management algorithm:
• All patients with invasive melanoma should be offered the gene expression profiling.
• Those with class 1 tumors have a high chance of cure and a low risk of metastasis. They can be reasonably managed with clinical skin and nodal exams every 6 months for 2 years, and then annually.
• Those with class 2 tumors have a much higher risk of metastatic disease. They should receive clinical skin and nodal exams every 3 months for 2 years, then every 6-12 months for 5 years. After that, a yearly exam should suffice.
Tests like this are an important advance in managing melanoma, Dr. Greenhaw said. “I think this is where the future of melanoma prognosis is. The keys are found in the genes and DNA of these tumors. These tests are being used in other types of cancer and I think it’s where we need to go in our field as well.”
Dr. Greenhaw has no financial disclosures to report.
ORLANDO – A commercially available melanoma gene expression test had a negative predictive value of 98% for metastasis, but also correctly identified patients at very high risk of disease spread.
In a retrospective study, the DecisionDx-Melanoma test, which examines 28 risk genes, showed that patients stratified as high risk with the test were 22 times more likely to develop metastatic disease than were those stratified as low risk, Dr. Bradley Greenhaw said at the annual meeting of the American College of Mohs Surgery. The test classifies patients with stage I and stage II melanoma as having a low risk (class 1) or a high risk (class 2) of metastasis within 5 years.
“Our mean follow-up time is less than half of the 5-year risk this test is able to predict,” said Dr. Greenhaw, a Mohs surgeon in Tupelo, Miss. “So if the test is performing as it’s supposed to, there would be even more events – you would see this gap widen even more as time passes. Seeing this with our limited follow-up period is pretty significant.”
The test also added valuable prognostic information to the American Joint Committee on Cancer (AJCC) staging method, he said. “For example, based on AJCC staging alone, we would expect to see a 5-year metastatic rate of 5%-10% for stage I melanomas. We saw less than a 1% rate. That is a pretty significant negative predictive value.”
The genes in the panel were selected from those examined in eight different genetic risk studies of cutaneous melanomas. The panel divides tumors into the high- and low-risk groups. A validation study published last year concluded that the 5-year disease-free survival rates were 97% for class 1 tumors and 31% for class 2 tumors (Clin Cancer Res 2015;21[1];175-83).
In the retrospective study, Dr. Greenhaw and his associates examined the test’s accuracy in a cohort of 256 patients treated at his center. The mean follow-up time was 23 months, although some patients have completed 5 years of follow-up.
Of the tumor blocks analyzed, 214 were class 1 and 42 were class 2. He noted significant differences between the class 1 and class 2 tumors, including mean patient age (66 years vs. 74 years, respectively), mean Breslow’s depth (0.7 mm vs. 2.3 mm), mitotic rate (0.8 vs. 3.3), and ulceration (4% vs. 41%).
Over the mean 2-year follow-up time, three class 1 tumors and 10 class 2 tumors metastasized (1.4% vs. 23.8%). The test’s negative predictive value was 98%; class 2 tumors were 22 times more likely to metastasize. “About 77% of metastatic tumors were correctly identified as high risk by this test,” Dr. Greenhaw said in an interview. “For comparison, more than 80% were correctly identified in the validation study, so our study was in line with it.”
The test showed a link between metastatic risk and ulceration at presentation. Of the class 1 tumors, 4% were ulcerated, compared to 41% of class 2 tumors; class 2 tumors were 16 times more likely to be ulcerated. Among the ulcerated lesions, metastasis occurred in 11% of class 1 and 41% of class 2 tumors.
Dr. Greenhaw also compared the gene test’s characterization of tumors to the AJCC staging.
Of the 214 class 1 tumors, 94% were stage IA or IB. Among these, there was no metastasis. Of the class 2 tumors, 17% were stage IA and 26% were stage IB. Interestingly, Dr. Greenhaw said, despite the high-risk genetic signature, none of these tumors metastasized. “This could have been because although the gene expression showed them to be associated with a high risk of metastases, the tumors were completely excised prior to the metastatic event, or because our follow-up is still relatively short,” he noted.
In the class 1 group, 4% of the tumors were stage IIA and 2% were stage IIB. Of these 13 tumors, two metastasized. “This 15% metastatic rate is a lower rate than we would predict by the AJCC staging,” Dr. Greenhaw said.
In the class 2 groups, 24% were stage IIA, 26% were stage IIB, and 7% were stage IIC. Of these 24 tumors, 10 metastasized. “This is a higher rate than the AJCC staging would predict, and it occurred rather quickly, with our abbreviated follow-up period,” Dr. Greenhaw noted.
Based on these findings, Dr. Greenhaw suggested a clinical management algorithm:
• All patients with invasive melanoma should be offered the gene expression profiling.
• Those with class 1 tumors have a high chance of cure and a low risk of metastasis. They can be reasonably managed with clinical skin and nodal exams every 6 months for 2 years, and then annually.
• Those with class 2 tumors have a much higher risk of metastatic disease. They should receive clinical skin and nodal exams every 3 months for 2 years, then every 6-12 months for 5 years. After that, a yearly exam should suffice.
Tests like this are an important advance in managing melanoma, Dr. Greenhaw said. “I think this is where the future of melanoma prognosis is. The keys are found in the genes and DNA of these tumors. These tests are being used in other types of cancer and I think it’s where we need to go in our field as well.”
Dr. Greenhaw has no financial disclosures to report.
AT THE ACMS ANNUAL MEETING
Key clinical point: A commercially available gene expression test can help predict metastasis in early melanoma cases.
Major finding: The test had a 98% negative predictive value.
Data source: A retrospective study that evaluated the gene expression test in 256 patients with melanoma.
Disclosures: Dr. Greenhaw has no financial interest in the test.
TBI prior to adoptive cell transfer added harm, no benefit
The addition of total body irradiation to nonmyeloablative chemotherapy prior to adoptive cell transfer does not provide any response or survival benefits but increases the risk of developing thrombotic microangiopathy (TMA), investigators reported.
Sequential clinical studies in the Surgery Branch of the National Cancer Institute had suggested that adding total body irradiation (TBI) to the preparative regimen for adoptive cell transfer could increase the complete response rate.
To evaluate in a randomized trial whether the addition of TBI to the nonmyeloablating (NMA) chemotherapy preparative regimen did indeed increase the complete response rate, 101 patients with metastatic melanoma with at least two measurable lesions were randomly assigned to receive only NMA chemotherapy or NMA chemotherapy and TBI. Patients in both treatment groups then underwent adoptive cell transfer and received intravenous infusions of tumor-infiltrating lymphocytes and high-dose IL-2. Only patients who received both NMA chemotherapy and TBI received CD34+ hematopoietic stem cells.
Complete response (CR) rate was 24% in both experimental arms (NMA: 12 of 51 patients, 95% CI 13% to 37%; NMA and TBI: 12 of 50 patients, 95% CI 13% to 38%). Progression-free survival and overall survival were similarly identical for the two experimental groups, Dr. Stephanie Goff of the National Cancer Institute and her associates reported (J Clin Oncol. 2016 May 23. doi: 10.1200/JCO.2016.66.7220).
The NMA plus TBI group had a high incidence of late-onset TMA (27%), complications that led to one treatment-related death.
“Addition of TBI did not increase CR rates or impact overall survival, but did introduce a new toxicity in the form of TMA. The nonmyeloablative chemotherapy regimen thus seemed to provide sufficient lymphodepletion for successful adoptive transfer without the need to add TBI,” Dr. Goff and associates concluded.
The Center for Cancer Research at the National Cancer Institute, the Dr. Miriam and Sheldon G. Adelson Medical Research Foundation, and Lion Biotechnologies funded the study. Four investigators disclosed financial ties with various companies. The other investigators had no disclosures to report.
On Twitter @JessCraig_OP
The addition of total body irradiation to nonmyeloablative chemotherapy prior to adoptive cell transfer does not provide any response or survival benefits but increases the risk of developing thrombotic microangiopathy (TMA), investigators reported.
Sequential clinical studies in the Surgery Branch of the National Cancer Institute had suggested that adding total body irradiation (TBI) to the preparative regimen for adoptive cell transfer could increase the complete response rate.
To evaluate in a randomized trial whether the addition of TBI to the nonmyeloablating (NMA) chemotherapy preparative regimen did indeed increase the complete response rate, 101 patients with metastatic melanoma with at least two measurable lesions were randomly assigned to receive only NMA chemotherapy or NMA chemotherapy and TBI. Patients in both treatment groups then underwent adoptive cell transfer and received intravenous infusions of tumor-infiltrating lymphocytes and high-dose IL-2. Only patients who received both NMA chemotherapy and TBI received CD34+ hematopoietic stem cells.
Complete response (CR) rate was 24% in both experimental arms (NMA: 12 of 51 patients, 95% CI 13% to 37%; NMA and TBI: 12 of 50 patients, 95% CI 13% to 38%). Progression-free survival and overall survival were similarly identical for the two experimental groups, Dr. Stephanie Goff of the National Cancer Institute and her associates reported (J Clin Oncol. 2016 May 23. doi: 10.1200/JCO.2016.66.7220).
The NMA plus TBI group had a high incidence of late-onset TMA (27%), complications that led to one treatment-related death.
“Addition of TBI did not increase CR rates or impact overall survival, but did introduce a new toxicity in the form of TMA. The nonmyeloablative chemotherapy regimen thus seemed to provide sufficient lymphodepletion for successful adoptive transfer without the need to add TBI,” Dr. Goff and associates concluded.
The Center for Cancer Research at the National Cancer Institute, the Dr. Miriam and Sheldon G. Adelson Medical Research Foundation, and Lion Biotechnologies funded the study. Four investigators disclosed financial ties with various companies. The other investigators had no disclosures to report.
On Twitter @JessCraig_OP
The addition of total body irradiation to nonmyeloablative chemotherapy prior to adoptive cell transfer does not provide any response or survival benefits but increases the risk of developing thrombotic microangiopathy (TMA), investigators reported.
Sequential clinical studies in the Surgery Branch of the National Cancer Institute had suggested that adding total body irradiation (TBI) to the preparative regimen for adoptive cell transfer could increase the complete response rate.
To evaluate in a randomized trial whether the addition of TBI to the nonmyeloablating (NMA) chemotherapy preparative regimen did indeed increase the complete response rate, 101 patients with metastatic melanoma with at least two measurable lesions were randomly assigned to receive only NMA chemotherapy or NMA chemotherapy and TBI. Patients in both treatment groups then underwent adoptive cell transfer and received intravenous infusions of tumor-infiltrating lymphocytes and high-dose IL-2. Only patients who received both NMA chemotherapy and TBI received CD34+ hematopoietic stem cells.
Complete response (CR) rate was 24% in both experimental arms (NMA: 12 of 51 patients, 95% CI 13% to 37%; NMA and TBI: 12 of 50 patients, 95% CI 13% to 38%). Progression-free survival and overall survival were similarly identical for the two experimental groups, Dr. Stephanie Goff of the National Cancer Institute and her associates reported (J Clin Oncol. 2016 May 23. doi: 10.1200/JCO.2016.66.7220).
The NMA plus TBI group had a high incidence of late-onset TMA (27%), complications that led to one treatment-related death.
“Addition of TBI did not increase CR rates or impact overall survival, but did introduce a new toxicity in the form of TMA. The nonmyeloablative chemotherapy regimen thus seemed to provide sufficient lymphodepletion for successful adoptive transfer without the need to add TBI,” Dr. Goff and associates concluded.
The Center for Cancer Research at the National Cancer Institute, the Dr. Miriam and Sheldon G. Adelson Medical Research Foundation, and Lion Biotechnologies funded the study. Four investigators disclosed financial ties with various companies. The other investigators had no disclosures to report.
On Twitter @JessCraig_OP
FROM JOURNAL OF CLINICAL ONCOLOGY
Key clinical point: No benefit but potential harm is observed when TBI is added to NMA chemotherapy prior to adoptive cell transfer.
Major finding: Complete response (CR) rate was 24% in both experimental arms (NMA: 12 of 51 patients, 95% CI 13% to 37%; NMA and TBI: 12 of 50 patients, 95% CI 13% to 38%). Progression-free survival and overall survival were also similar for the two experimental groups. A total of 27% of patients in the NMA plus TBI group experienced thrombotic microangiopathy.
Data source: Randomized clinical trial of 101 patients with metastatic melanoma.
Disclosures: The Center for Cancer Research at the National Cancer Institute, the Dr. Miriam and Sheldon G. Adelson Medical Research Foundation, and Lion Biotechnologies funded the study. Four investigators disclosed financial ties with various companies. The other investigators had no disclosures to report.
Sentinel node biopsies may be useful in head and neck squamous cell carcinoma
ORLANDO – Sentinel node biopsies may be a useful staging tool for patients with cutaneous squamous cell carcinomas of the head and neck.
These patients – especially those with compromised immune systems – appear to be at sufficiently high risk of metastasis to justify the procedure, Dr. Jonathan Lopez said at the annual meeting of the American College of Mohs Surgery.
“We found that sentinel lymph node biopsy in our clinic had a 91% negative predictive value for local recurrence, nodal recurrence, and disease-specific death. It provides valuable prognostic information for patients at increased risk of nodal metastasis,” said Dr. Lopez, a dermatology resident at the Mayo Clinic, Rochester, Minn.
He and his associates conducted a chart review of 24 patients treated at the Mayo Clinic from 2000 to 2014 for a cutaneous squamous cell carcinoma (SCC) of the head or neck. Of these, 11 patients were immunosuppressed. Five had undergone a kidney transplant and three a lung transplant. One patient had Hodgkin’s lymphoma, one had cutaneous lymphocytic leukemia, and one, metastatic urothelial carcinoma.
Before sentinel node biopsy, eight patients had a wide local excision; 12 were treated with Mohs micrographic surgery only; and four had a Mohs procedure followed by resection for better margins.
The biopsies identified two patients with nodal disease, but failed to identify a third who had it, Dr. Lopez said.
Patient No. 1 had a primary SCC on the nasal tip that was stage 2, according to the American Joint Committee on Cancer (AJCC) staging system, and 2b according to the Brigham and Women’s Hospital (BWH) system. He had undergone a prior double lung transplant and his lymph node dissection showed no nodal metastasis. He declined radiotherapy and died within 2 months of the biopsy, of unclear causes that were not related to his skin cancer.
Patient No. 2 had a primary lesion on the right cheek, and a history of kidney transplant. His cancer was stage 2 by the AJCC system and 2b by the BWH system. His lymph node dissection of the right parotid and neck was negative. At last follow-up of 3.5 years, he was cancer free. However, Dr. Lopez noted, the patient died at 4 years’ follow-up of unknown causes.
The final patient had a primary lesion on the right conchal bowl. It was a stage 2 cancer by the AJCC system and 2a by the BWH system. His sentinel node biopsy was negative. However, the otolaryngologist who performed the biopsy also took seven superficial parotid nodes and one of those was positive. This patient had no recurrence at the last visit, 1.5 years after the biopsy.
The sentinel node biopsies were negative in the 21 other patients. Of these, 14 had no evidence of recurrence at a mean of 3 years’ follow-up after the sentinel lymph node biopsy. Two developed local recurrence and two others, both of whom had a history of multiple squamous cell carcinomas, developed nodal spread and died of metastatic disease. Three have died of causes unrelated to their cancer.
Dr. Lopez had no financial disclosures.
ORLANDO – Sentinel node biopsies may be a useful staging tool for patients with cutaneous squamous cell carcinomas of the head and neck.
These patients – especially those with compromised immune systems – appear to be at sufficiently high risk of metastasis to justify the procedure, Dr. Jonathan Lopez said at the annual meeting of the American College of Mohs Surgery.
“We found that sentinel lymph node biopsy in our clinic had a 91% negative predictive value for local recurrence, nodal recurrence, and disease-specific death. It provides valuable prognostic information for patients at increased risk of nodal metastasis,” said Dr. Lopez, a dermatology resident at the Mayo Clinic, Rochester, Minn.
He and his associates conducted a chart review of 24 patients treated at the Mayo Clinic from 2000 to 2014 for a cutaneous squamous cell carcinoma (SCC) of the head or neck. Of these, 11 patients were immunosuppressed. Five had undergone a kidney transplant and three a lung transplant. One patient had Hodgkin’s lymphoma, one had cutaneous lymphocytic leukemia, and one, metastatic urothelial carcinoma.
Before sentinel node biopsy, eight patients had a wide local excision; 12 were treated with Mohs micrographic surgery only; and four had a Mohs procedure followed by resection for better margins.
The biopsies identified two patients with nodal disease, but failed to identify a third who had it, Dr. Lopez said.
Patient No. 1 had a primary SCC on the nasal tip that was stage 2, according to the American Joint Committee on Cancer (AJCC) staging system, and 2b according to the Brigham and Women’s Hospital (BWH) system. He had undergone a prior double lung transplant and his lymph node dissection showed no nodal metastasis. He declined radiotherapy and died within 2 months of the biopsy, of unclear causes that were not related to his skin cancer.
Patient No. 2 had a primary lesion on the right cheek, and a history of kidney transplant. His cancer was stage 2 by the AJCC system and 2b by the BWH system. His lymph node dissection of the right parotid and neck was negative. At last follow-up of 3.5 years, he was cancer free. However, Dr. Lopez noted, the patient died at 4 years’ follow-up of unknown causes.
The final patient had a primary lesion on the right conchal bowl. It was a stage 2 cancer by the AJCC system and 2a by the BWH system. His sentinel node biopsy was negative. However, the otolaryngologist who performed the biopsy also took seven superficial parotid nodes and one of those was positive. This patient had no recurrence at the last visit, 1.5 years after the biopsy.
The sentinel node biopsies were negative in the 21 other patients. Of these, 14 had no evidence of recurrence at a mean of 3 years’ follow-up after the sentinel lymph node biopsy. Two developed local recurrence and two others, both of whom had a history of multiple squamous cell carcinomas, developed nodal spread and died of metastatic disease. Three have died of causes unrelated to their cancer.
Dr. Lopez had no financial disclosures.
ORLANDO – Sentinel node biopsies may be a useful staging tool for patients with cutaneous squamous cell carcinomas of the head and neck.
These patients – especially those with compromised immune systems – appear to be at sufficiently high risk of metastasis to justify the procedure, Dr. Jonathan Lopez said at the annual meeting of the American College of Mohs Surgery.
“We found that sentinel lymph node biopsy in our clinic had a 91% negative predictive value for local recurrence, nodal recurrence, and disease-specific death. It provides valuable prognostic information for patients at increased risk of nodal metastasis,” said Dr. Lopez, a dermatology resident at the Mayo Clinic, Rochester, Minn.
He and his associates conducted a chart review of 24 patients treated at the Mayo Clinic from 2000 to 2014 for a cutaneous squamous cell carcinoma (SCC) of the head or neck. Of these, 11 patients were immunosuppressed. Five had undergone a kidney transplant and three a lung transplant. One patient had Hodgkin’s lymphoma, one had cutaneous lymphocytic leukemia, and one, metastatic urothelial carcinoma.
Before sentinel node biopsy, eight patients had a wide local excision; 12 were treated with Mohs micrographic surgery only; and four had a Mohs procedure followed by resection for better margins.
The biopsies identified two patients with nodal disease, but failed to identify a third who had it, Dr. Lopez said.
Patient No. 1 had a primary SCC on the nasal tip that was stage 2, according to the American Joint Committee on Cancer (AJCC) staging system, and 2b according to the Brigham and Women’s Hospital (BWH) system. He had undergone a prior double lung transplant and his lymph node dissection showed no nodal metastasis. He declined radiotherapy and died within 2 months of the biopsy, of unclear causes that were not related to his skin cancer.
Patient No. 2 had a primary lesion on the right cheek, and a history of kidney transplant. His cancer was stage 2 by the AJCC system and 2b by the BWH system. His lymph node dissection of the right parotid and neck was negative. At last follow-up of 3.5 years, he was cancer free. However, Dr. Lopez noted, the patient died at 4 years’ follow-up of unknown causes.
The final patient had a primary lesion on the right conchal bowl. It was a stage 2 cancer by the AJCC system and 2a by the BWH system. His sentinel node biopsy was negative. However, the otolaryngologist who performed the biopsy also took seven superficial parotid nodes and one of those was positive. This patient had no recurrence at the last visit, 1.5 years after the biopsy.
The sentinel node biopsies were negative in the 21 other patients. Of these, 14 had no evidence of recurrence at a mean of 3 years’ follow-up after the sentinel lymph node biopsy. Two developed local recurrence and two others, both of whom had a history of multiple squamous cell carcinomas, developed nodal spread and died of metastatic disease. Three have died of causes unrelated to their cancer.
Dr. Lopez had no financial disclosures.
AT THE ACMS ANNUAL MEETING
Key clinical point: Sentinel node biopsies identified nodal spread in some patients with cutaneous SCC of the head and neck
Major finding: The procedure had a 91% negative predictive value for nodal spread and disease-specific death.
Data source: The retrospective chart review comprised of 24 patients, treated at the Mayo Clinic for cutaneous SCC of the head and neck from 2000 to 2014.
Disclosures: Dr. Lopez had no financial disclosures.
Matrilin-2 protein distinguished BCCs from benign tumors in study
ORLANDO – Matrilin-2 – a matrix protein found in peritumoral stroma – reliably distinguished invasive basal cell carcinoma from the often difficult-to-distinguish basaloid follicular hamartoma (BFH), in a study that evaluated the protein as a marker in this setting.
The protein marked 41 of 42 cancers and none of the hamartomas, Dr. Renato Goreshi reported at the annual meeting of the American College of Mohs Surgery. The one cancer it failed to identify was a superficial basal cell tumor – a finding that makes sense, since dermal fibroblasts appear to secrete matrilin-2 as a response to invasive skin tumors, said Dr. Goreshi of the Roger Williams Cancer Center, Providence, R.I.
Mohs surgery typically employs hematoxylin and eosin staining to delineate tumor boundary. But, Dr. Goreshi said, that stain doesn’t always reliably differentiate adnexal tumors from basal cell carcinomas. “Basaloid follicular hamartoma can be particularly difficult to distinguish from basal cell carcinoma,” he said.
BFH typically presents as individual or linearly arranged, small skin-colored to brown papules or plaques, or as multiple lesions in a generalized distribution on the face, scalp, and occasionally, the trunk (Arch Pathol Lab Med. 2010 Aug;134[8]:1215-9). These are often stable for many years. The differential diagnosis includes basal cell carcinoma and trichoepithelioma.
BFH sometimes occurs near a BCC, although there are no data on how often this happens.
Dr. Goreshi cited a 2007 case report of a young woman that illustrates this problem. The patient presented with a basal cell carcinoma on the side of her nose. The adjacent BFH was unrecognized, however. She underwent a multiple-stage Mohs that was unnecessarily extended because tumor margins included sections of the BFH.
“The lesion was interpreted as malignancy by both the Mohs surgeon and the dermatopathologist, but was later determined to have been a hamartoma. This highlights the importance of finding an effective marker,” Dr. Goreshi said.
He and his fellowship director, Dr. Satori Iwamoto, chief of Mohs micrographic surgery at Roger Williams, looked for a reliable way to differentiate these tumors, capitalizing on the invasive nature of BCC. The peritumoral stroma plays a role in tumor growth and invasion. It involves fibroblasts, inflammatory and endothelial cells, and extracellular matrix proteins. Matrilin-2, which is involved in the formation of filamentous networks, was a promising candidate and the initial investigations looked good, said Dr. Goreshi said.
Their confirmatory study comprised 42 BCC and seven BFH sections that were obtained during Mohs surgery. All were stained for matrilin-2 and scored for location and intensity of staining by two reviewers. The investigators also conducted flow cytometry to determine the source of the protein.
The BCC set consisted of 11 morpheaform/infiltrative BCCs, 25 nodular BCCs, and 6 superficial BCCs. With the exception of one superficial lesion, all of these stained positive for matrilin-2 in the peritumoral stroma. None of the BFH sections stained positive for the protein, however. Flow cytometry determined that the protein was coming from dermal fibroblasts in the stroma.
This is actually a key point, Dr. Goreshi noted. “Matrilin-2 is not acting as a conventional tumor marker would, but as a marker of invasion.”
This was again played out in the variation of staining intensity in the tumor subtypes. It was most intense around the infiltrative subtypes. There was also adnexal staining, but it was significantly less than what was seen in the peritumoral stroma. There was virtually no staining in or around the hamartoma.
Staining was not as intense around the superficial BCC subtypes. In fact, it was not significantly different from what was seen in the adnexal structures. Again, however, there was no staining in or around the hamartoma.
“Now we are looking at the staining patterns of other lesions, including melanoma and squamous cell carcinoma, and trying to figure out why the dermal fibroblasts are secreting matrilin-2,” Dr. Goreshi said.
The study was the winner of the 2016 Theodore Tromovitch Award, presented for original research conducted by a fellow-in-training during his or her year of training.
Neither Dr. Goreshi nor Dr. Iwamoto had any relevant financial disclosures.
ORLANDO – Matrilin-2 – a matrix protein found in peritumoral stroma – reliably distinguished invasive basal cell carcinoma from the often difficult-to-distinguish basaloid follicular hamartoma (BFH), in a study that evaluated the protein as a marker in this setting.
The protein marked 41 of 42 cancers and none of the hamartomas, Dr. Renato Goreshi reported at the annual meeting of the American College of Mohs Surgery. The one cancer it failed to identify was a superficial basal cell tumor – a finding that makes sense, since dermal fibroblasts appear to secrete matrilin-2 as a response to invasive skin tumors, said Dr. Goreshi of the Roger Williams Cancer Center, Providence, R.I.
Mohs surgery typically employs hematoxylin and eosin staining to delineate tumor boundary. But, Dr. Goreshi said, that stain doesn’t always reliably differentiate adnexal tumors from basal cell carcinomas. “Basaloid follicular hamartoma can be particularly difficult to distinguish from basal cell carcinoma,” he said.
BFH typically presents as individual or linearly arranged, small skin-colored to brown papules or plaques, or as multiple lesions in a generalized distribution on the face, scalp, and occasionally, the trunk (Arch Pathol Lab Med. 2010 Aug;134[8]:1215-9). These are often stable for many years. The differential diagnosis includes basal cell carcinoma and trichoepithelioma.
BFH sometimes occurs near a BCC, although there are no data on how often this happens.
Dr. Goreshi cited a 2007 case report of a young woman that illustrates this problem. The patient presented with a basal cell carcinoma on the side of her nose. The adjacent BFH was unrecognized, however. She underwent a multiple-stage Mohs that was unnecessarily extended because tumor margins included sections of the BFH.
“The lesion was interpreted as malignancy by both the Mohs surgeon and the dermatopathologist, but was later determined to have been a hamartoma. This highlights the importance of finding an effective marker,” Dr. Goreshi said.
He and his fellowship director, Dr. Satori Iwamoto, chief of Mohs micrographic surgery at Roger Williams, looked for a reliable way to differentiate these tumors, capitalizing on the invasive nature of BCC. The peritumoral stroma plays a role in tumor growth and invasion. It involves fibroblasts, inflammatory and endothelial cells, and extracellular matrix proteins. Matrilin-2, which is involved in the formation of filamentous networks, was a promising candidate and the initial investigations looked good, said Dr. Goreshi said.
Their confirmatory study comprised 42 BCC and seven BFH sections that were obtained during Mohs surgery. All were stained for matrilin-2 and scored for location and intensity of staining by two reviewers. The investigators also conducted flow cytometry to determine the source of the protein.
The BCC set consisted of 11 morpheaform/infiltrative BCCs, 25 nodular BCCs, and 6 superficial BCCs. With the exception of one superficial lesion, all of these stained positive for matrilin-2 in the peritumoral stroma. None of the BFH sections stained positive for the protein, however. Flow cytometry determined that the protein was coming from dermal fibroblasts in the stroma.
This is actually a key point, Dr. Goreshi noted. “Matrilin-2 is not acting as a conventional tumor marker would, but as a marker of invasion.”
This was again played out in the variation of staining intensity in the tumor subtypes. It was most intense around the infiltrative subtypes. There was also adnexal staining, but it was significantly less than what was seen in the peritumoral stroma. There was virtually no staining in or around the hamartoma.
Staining was not as intense around the superficial BCC subtypes. In fact, it was not significantly different from what was seen in the adnexal structures. Again, however, there was no staining in or around the hamartoma.
“Now we are looking at the staining patterns of other lesions, including melanoma and squamous cell carcinoma, and trying to figure out why the dermal fibroblasts are secreting matrilin-2,” Dr. Goreshi said.
The study was the winner of the 2016 Theodore Tromovitch Award, presented for original research conducted by a fellow-in-training during his or her year of training.
Neither Dr. Goreshi nor Dr. Iwamoto had any relevant financial disclosures.
ORLANDO – Matrilin-2 – a matrix protein found in peritumoral stroma – reliably distinguished invasive basal cell carcinoma from the often difficult-to-distinguish basaloid follicular hamartoma (BFH), in a study that evaluated the protein as a marker in this setting.
The protein marked 41 of 42 cancers and none of the hamartomas, Dr. Renato Goreshi reported at the annual meeting of the American College of Mohs Surgery. The one cancer it failed to identify was a superficial basal cell tumor – a finding that makes sense, since dermal fibroblasts appear to secrete matrilin-2 as a response to invasive skin tumors, said Dr. Goreshi of the Roger Williams Cancer Center, Providence, R.I.
Mohs surgery typically employs hematoxylin and eosin staining to delineate tumor boundary. But, Dr. Goreshi said, that stain doesn’t always reliably differentiate adnexal tumors from basal cell carcinomas. “Basaloid follicular hamartoma can be particularly difficult to distinguish from basal cell carcinoma,” he said.
BFH typically presents as individual or linearly arranged, small skin-colored to brown papules or plaques, or as multiple lesions in a generalized distribution on the face, scalp, and occasionally, the trunk (Arch Pathol Lab Med. 2010 Aug;134[8]:1215-9). These are often stable for many years. The differential diagnosis includes basal cell carcinoma and trichoepithelioma.
BFH sometimes occurs near a BCC, although there are no data on how often this happens.
Dr. Goreshi cited a 2007 case report of a young woman that illustrates this problem. The patient presented with a basal cell carcinoma on the side of her nose. The adjacent BFH was unrecognized, however. She underwent a multiple-stage Mohs that was unnecessarily extended because tumor margins included sections of the BFH.
“The lesion was interpreted as malignancy by both the Mohs surgeon and the dermatopathologist, but was later determined to have been a hamartoma. This highlights the importance of finding an effective marker,” Dr. Goreshi said.
He and his fellowship director, Dr. Satori Iwamoto, chief of Mohs micrographic surgery at Roger Williams, looked for a reliable way to differentiate these tumors, capitalizing on the invasive nature of BCC. The peritumoral stroma plays a role in tumor growth and invasion. It involves fibroblasts, inflammatory and endothelial cells, and extracellular matrix proteins. Matrilin-2, which is involved in the formation of filamentous networks, was a promising candidate and the initial investigations looked good, said Dr. Goreshi said.
Their confirmatory study comprised 42 BCC and seven BFH sections that were obtained during Mohs surgery. All were stained for matrilin-2 and scored for location and intensity of staining by two reviewers. The investigators also conducted flow cytometry to determine the source of the protein.
The BCC set consisted of 11 morpheaform/infiltrative BCCs, 25 nodular BCCs, and 6 superficial BCCs. With the exception of one superficial lesion, all of these stained positive for matrilin-2 in the peritumoral stroma. None of the BFH sections stained positive for the protein, however. Flow cytometry determined that the protein was coming from dermal fibroblasts in the stroma.
This is actually a key point, Dr. Goreshi noted. “Matrilin-2 is not acting as a conventional tumor marker would, but as a marker of invasion.”
This was again played out in the variation of staining intensity in the tumor subtypes. It was most intense around the infiltrative subtypes. There was also adnexal staining, but it was significantly less than what was seen in the peritumoral stroma. There was virtually no staining in or around the hamartoma.
Staining was not as intense around the superficial BCC subtypes. In fact, it was not significantly different from what was seen in the adnexal structures. Again, however, there was no staining in or around the hamartoma.
“Now we are looking at the staining patterns of other lesions, including melanoma and squamous cell carcinoma, and trying to figure out why the dermal fibroblasts are secreting matrilin-2,” Dr. Goreshi said.
The study was the winner of the 2016 Theodore Tromovitch Award, presented for original research conducted by a fellow-in-training during his or her year of training.
Neither Dr. Goreshi nor Dr. Iwamoto had any relevant financial disclosures.
AT THE ACMS ANNUAL MEETING
Key clinical point: Matrilin-2 is the first marker of tumor invasion to be used in skin cancers.
Major finding: The protein bound to 41 of 42 BCCs, and to none of the hamartoma lesions studied, reliably distinguishing the two.
Data source: 42 frozen section BCCs and seven basaloid follicular hamartomas.
Disclosures: Neither Dr. Goreshi nor Dr. Iwamoto had any relevant financial disclosures.
For preventing AKs, 5-FU beats placebo for up to 3 years
SCOTTSDALE – A single course of topical 5-fluorouracil (5-FU) prevented 62% more actinic keratoses than placebo, and this chemopreventive effect persisted for up to 3 years, according to an analysis of the Veterans Affairs Keratinocyte Carcinoma Chemoprevention Trial (VAKCCT) trial.
Other studies have shown that 5-FU effectively treats precancerous AKs, but have not examined whether 5-FU can prevent AKs, Dr. Joanna Walker said in an interview at the annual meeting of the Society for Investigative Dermatology.
Clinicians should consider preventive 5-FU in patients who are at high risk for basal cell and squamous cell carcinomas, especially if a skin check reveals multiple AKs, said Dr. Walker of the department of dermatology, Brown University, Providence, RI.
The VAKCCT was a randomized, double-blind, placebo-controlled study conducted at 12 Veterans Affairs dermatology clinics. The 319 patients in the analysis were nearly all elderly men with extensive sun damage, with a total of 2,386 AKs at baseline, for an average of five lesions per patient. Patients also had a history of at least two keratinocyte carcinomas in the past 5 years, including at least one lesion on the face or ears, and no recent history of 5-FU exposure.
The clinically and demographically similar study arms were randomized to either 5% topical 5-FU cream or a vehicle control cream, applied twice daily for 2-4 weeks. Both groups received cryotherapy for existing AKs, and were given free SPF 30 sunscreen. At each 6-month follow-up visit, the researchers counted existing AKs and new lesions.
At month 6, the treatment group had 62% fewer new AKs than the placebo group (average per patient, 1.78 and 4.73, respectively), a statistically significant difference. At months 12, 18, 24, 30, and 36, respectively, the treatment group had 50%, 40%, 41%, 25%, and 35% fewer new AKs than the placebo group, and these differences all were statistically significant. Furthermore, at month 6, only 56% of treated patients had at least one new AK, compared with 78% of the control group (incidence rate ratio, 0.72; 95% confidence interval, 0.54-0.95).
This chemopreventive effect remained significant for 24 months, the investigators reported. “Individuals with at least five AKs at the time of 5-FU treatment had an even more dramatic reduction in new AKs,” Dr. Walker noted. “There is now high-quality evidence supporting the use of topical 5-FU for AK chemoprevention. I think this is important information that we can take back to the clinic when we are trying to convince our patients to go through a course of 5-FU.”
The rate of new AKs in the placebo group fell during the first 2.5 years of the study and then stabilized. “For both groups, there was a dramatic increase in the use of sunscreen during the trial, and we hypothesized that the decrease in AKs in the control group was due to increased use of sun-protective measures,” Dr. Walker said.
The research was funded by the Cooperative Studies Program of the U.S. Department of Veterans Affairs. Dr. Walker had no disclosures.
SCOTTSDALE – A single course of topical 5-fluorouracil (5-FU) prevented 62% more actinic keratoses than placebo, and this chemopreventive effect persisted for up to 3 years, according to an analysis of the Veterans Affairs Keratinocyte Carcinoma Chemoprevention Trial (VAKCCT) trial.
Other studies have shown that 5-FU effectively treats precancerous AKs, but have not examined whether 5-FU can prevent AKs, Dr. Joanna Walker said in an interview at the annual meeting of the Society for Investigative Dermatology.
Clinicians should consider preventive 5-FU in patients who are at high risk for basal cell and squamous cell carcinomas, especially if a skin check reveals multiple AKs, said Dr. Walker of the department of dermatology, Brown University, Providence, RI.
The VAKCCT was a randomized, double-blind, placebo-controlled study conducted at 12 Veterans Affairs dermatology clinics. The 319 patients in the analysis were nearly all elderly men with extensive sun damage, with a total of 2,386 AKs at baseline, for an average of five lesions per patient. Patients also had a history of at least two keratinocyte carcinomas in the past 5 years, including at least one lesion on the face or ears, and no recent history of 5-FU exposure.
The clinically and demographically similar study arms were randomized to either 5% topical 5-FU cream or a vehicle control cream, applied twice daily for 2-4 weeks. Both groups received cryotherapy for existing AKs, and were given free SPF 30 sunscreen. At each 6-month follow-up visit, the researchers counted existing AKs and new lesions.
At month 6, the treatment group had 62% fewer new AKs than the placebo group (average per patient, 1.78 and 4.73, respectively), a statistically significant difference. At months 12, 18, 24, 30, and 36, respectively, the treatment group had 50%, 40%, 41%, 25%, and 35% fewer new AKs than the placebo group, and these differences all were statistically significant. Furthermore, at month 6, only 56% of treated patients had at least one new AK, compared with 78% of the control group (incidence rate ratio, 0.72; 95% confidence interval, 0.54-0.95).
This chemopreventive effect remained significant for 24 months, the investigators reported. “Individuals with at least five AKs at the time of 5-FU treatment had an even more dramatic reduction in new AKs,” Dr. Walker noted. “There is now high-quality evidence supporting the use of topical 5-FU for AK chemoprevention. I think this is important information that we can take back to the clinic when we are trying to convince our patients to go through a course of 5-FU.”
The rate of new AKs in the placebo group fell during the first 2.5 years of the study and then stabilized. “For both groups, there was a dramatic increase in the use of sunscreen during the trial, and we hypothesized that the decrease in AKs in the control group was due to increased use of sun-protective measures,” Dr. Walker said.
The research was funded by the Cooperative Studies Program of the U.S. Department of Veterans Affairs. Dr. Walker had no disclosures.
SCOTTSDALE – A single course of topical 5-fluorouracil (5-FU) prevented 62% more actinic keratoses than placebo, and this chemopreventive effect persisted for up to 3 years, according to an analysis of the Veterans Affairs Keratinocyte Carcinoma Chemoprevention Trial (VAKCCT) trial.
Other studies have shown that 5-FU effectively treats precancerous AKs, but have not examined whether 5-FU can prevent AKs, Dr. Joanna Walker said in an interview at the annual meeting of the Society for Investigative Dermatology.
Clinicians should consider preventive 5-FU in patients who are at high risk for basal cell and squamous cell carcinomas, especially if a skin check reveals multiple AKs, said Dr. Walker of the department of dermatology, Brown University, Providence, RI.
The VAKCCT was a randomized, double-blind, placebo-controlled study conducted at 12 Veterans Affairs dermatology clinics. The 319 patients in the analysis were nearly all elderly men with extensive sun damage, with a total of 2,386 AKs at baseline, for an average of five lesions per patient. Patients also had a history of at least two keratinocyte carcinomas in the past 5 years, including at least one lesion on the face or ears, and no recent history of 5-FU exposure.
The clinically and demographically similar study arms were randomized to either 5% topical 5-FU cream or a vehicle control cream, applied twice daily for 2-4 weeks. Both groups received cryotherapy for existing AKs, and were given free SPF 30 sunscreen. At each 6-month follow-up visit, the researchers counted existing AKs and new lesions.
At month 6, the treatment group had 62% fewer new AKs than the placebo group (average per patient, 1.78 and 4.73, respectively), a statistically significant difference. At months 12, 18, 24, 30, and 36, respectively, the treatment group had 50%, 40%, 41%, 25%, and 35% fewer new AKs than the placebo group, and these differences all were statistically significant. Furthermore, at month 6, only 56% of treated patients had at least one new AK, compared with 78% of the control group (incidence rate ratio, 0.72; 95% confidence interval, 0.54-0.95).
This chemopreventive effect remained significant for 24 months, the investigators reported. “Individuals with at least five AKs at the time of 5-FU treatment had an even more dramatic reduction in new AKs,” Dr. Walker noted. “There is now high-quality evidence supporting the use of topical 5-FU for AK chemoprevention. I think this is important information that we can take back to the clinic when we are trying to convince our patients to go through a course of 5-FU.”
The rate of new AKs in the placebo group fell during the first 2.5 years of the study and then stabilized. “For both groups, there was a dramatic increase in the use of sunscreen during the trial, and we hypothesized that the decrease in AKs in the control group was due to increased use of sun-protective measures,” Dr. Walker said.
The research was funded by the Cooperative Studies Program of the U.S. Department of Veterans Affairs. Dr. Walker had no disclosures.
AT THE 2016 SID ANNUAL MEETING
Key clinical point: One course of topical 5-fluorouracil was effective and durable in preventing new actinic keratoses in high-risk patients.
Major finding: At month 6, the treatment group had 62% fewer new AKs than the placebo group, and the difference remained significant at month 36.
Data source: The double-blind controlled study evaluated 5-FU vs. a vehicle cream in 319 veterans, most of whom were elderly men.
Disclosures: The study was funded by the Cooperative Studies Program of the U.S. Department of Veterans Affairs. Dr. Walker had no disclosures.