User login
Higher teen pregnancy risk in girls with ADHD
Teenage girls with ADHD may be at greater risk of pregnancy than their unaffected peers, which suggests they may benefit from targeted interventions to prevent teen pregnancy.
A Swedish nationwide cohort study published in JAMA Network Open examined data from 384,103 nulliparous women and girls who gave birth between 2007-2014, of whom, 6,410 (1.7%) had received treatment for ADHD.
While the overall rate of teenage births was 3%, the rate among women and girls with ADHD was 15.3%, which represents a greater than sixfold higher odds of giving birth below the age of 20 years (odds ratio, 6.23; 95% confidence interval, 5.80-6.68).
“Becoming a mother at such early age is associated with long-term adverse outcomes for both women and their children,” wrote Charlotte Skoglund, PhD, of the department of clinical neuroscience at the Karolinska Institute in Stockholm and coauthors. “Consequently, our findings argue for an improvement in the standard of care for women and girls with ADHD, including active efforts to prevent teenage pregnancies and address comorbid medical and psychiatric conditions.”
The study also found women and girls with ADHD were significantly more likely to be underweight (OR, 1.29; 95% CI, 1.12-1.49) or have a body mass index greater than 40 kg/m2 (OR, 2.01; 95% CI, 1.60-2.52) when compared with those without ADHD.
They were also six times more likely to smoke, were nearly seven times more likely to continue smoking into their third trimester of pregnancy, and had a 20-fold higher odds of alcohol and substance use disorder. Among individuals who had been diagnosed with ADHD, 7.6% continued to use stimulant and nonstimulant ADHD medication during pregnancy, and 16.4% used antidepressants during pregnancy.
Psychiatric comorbidities were also significantly more common among individuals with ADHD in the year preceding pregnancy, compared with those without ADHD. The authors saw a 17-fold higher odds of receiving a diagnosis of bipolar disorder, nearly 8-fold higher odds of a diagnosis of schizophrenia or other psychotic disorder, and 22-fold higher odds of being diagnosed with emotionally unstable personality disorder among women and girls with ADHD versus those without.
The authors commented that antenatal care should focus on trying to reduce such obstetric risk factors in these women, but also pointed out that ADHD in women and girls was still underdiagnosed and undertreated.
Commenting on the association between ADHD and teenage pregnancy, the authors noted that women and girls with ADHD may be less likely to receive adequate contraceptive counseling and less likely to access, respond to, and act on counseling. They may also experience more adverse effects from hormonal contraceptives.
While Swedish youth clinics enable easier and low-cost access to counseling and contraception, the authors called for greater collaboration between psychiatric care clinics and specialized youth clinics to provide adequate care for women and girls with ADHD.
Three authors declared advisory board positions, grants, personal fees, and speakers’ fees from the pharmaceutical sector. No other conflicts of interest were declared.
SOURCE: Skoglund C et al. JAMA Netw Open. 2019 Oct 2. doi: 10.1001/jamanetworkopen.2019.12463
Teenage girls with ADHD may be at greater risk of pregnancy than their unaffected peers, which suggests they may benefit from targeted interventions to prevent teen pregnancy.
A Swedish nationwide cohort study published in JAMA Network Open examined data from 384,103 nulliparous women and girls who gave birth between 2007-2014, of whom, 6,410 (1.7%) had received treatment for ADHD.
While the overall rate of teenage births was 3%, the rate among women and girls with ADHD was 15.3%, which represents a greater than sixfold higher odds of giving birth below the age of 20 years (odds ratio, 6.23; 95% confidence interval, 5.80-6.68).
“Becoming a mother at such early age is associated with long-term adverse outcomes for both women and their children,” wrote Charlotte Skoglund, PhD, of the department of clinical neuroscience at the Karolinska Institute in Stockholm and coauthors. “Consequently, our findings argue for an improvement in the standard of care for women and girls with ADHD, including active efforts to prevent teenage pregnancies and address comorbid medical and psychiatric conditions.”
The study also found women and girls with ADHD were significantly more likely to be underweight (OR, 1.29; 95% CI, 1.12-1.49) or have a body mass index greater than 40 kg/m2 (OR, 2.01; 95% CI, 1.60-2.52) when compared with those without ADHD.
They were also six times more likely to smoke, were nearly seven times more likely to continue smoking into their third trimester of pregnancy, and had a 20-fold higher odds of alcohol and substance use disorder. Among individuals who had been diagnosed with ADHD, 7.6% continued to use stimulant and nonstimulant ADHD medication during pregnancy, and 16.4% used antidepressants during pregnancy.
Psychiatric comorbidities were also significantly more common among individuals with ADHD in the year preceding pregnancy, compared with those without ADHD. The authors saw a 17-fold higher odds of receiving a diagnosis of bipolar disorder, nearly 8-fold higher odds of a diagnosis of schizophrenia or other psychotic disorder, and 22-fold higher odds of being diagnosed with emotionally unstable personality disorder among women and girls with ADHD versus those without.
The authors commented that antenatal care should focus on trying to reduce such obstetric risk factors in these women, but also pointed out that ADHD in women and girls was still underdiagnosed and undertreated.
Commenting on the association between ADHD and teenage pregnancy, the authors noted that women and girls with ADHD may be less likely to receive adequate contraceptive counseling and less likely to access, respond to, and act on counseling. They may also experience more adverse effects from hormonal contraceptives.
While Swedish youth clinics enable easier and low-cost access to counseling and contraception, the authors called for greater collaboration between psychiatric care clinics and specialized youth clinics to provide adequate care for women and girls with ADHD.
Three authors declared advisory board positions, grants, personal fees, and speakers’ fees from the pharmaceutical sector. No other conflicts of interest were declared.
SOURCE: Skoglund C et al. JAMA Netw Open. 2019 Oct 2. doi: 10.1001/jamanetworkopen.2019.12463
Teenage girls with ADHD may be at greater risk of pregnancy than their unaffected peers, which suggests they may benefit from targeted interventions to prevent teen pregnancy.
A Swedish nationwide cohort study published in JAMA Network Open examined data from 384,103 nulliparous women and girls who gave birth between 2007-2014, of whom, 6,410 (1.7%) had received treatment for ADHD.
While the overall rate of teenage births was 3%, the rate among women and girls with ADHD was 15.3%, which represents a greater than sixfold higher odds of giving birth below the age of 20 years (odds ratio, 6.23; 95% confidence interval, 5.80-6.68).
“Becoming a mother at such early age is associated with long-term adverse outcomes for both women and their children,” wrote Charlotte Skoglund, PhD, of the department of clinical neuroscience at the Karolinska Institute in Stockholm and coauthors. “Consequently, our findings argue for an improvement in the standard of care for women and girls with ADHD, including active efforts to prevent teenage pregnancies and address comorbid medical and psychiatric conditions.”
The study also found women and girls with ADHD were significantly more likely to be underweight (OR, 1.29; 95% CI, 1.12-1.49) or have a body mass index greater than 40 kg/m2 (OR, 2.01; 95% CI, 1.60-2.52) when compared with those without ADHD.
They were also six times more likely to smoke, were nearly seven times more likely to continue smoking into their third trimester of pregnancy, and had a 20-fold higher odds of alcohol and substance use disorder. Among individuals who had been diagnosed with ADHD, 7.6% continued to use stimulant and nonstimulant ADHD medication during pregnancy, and 16.4% used antidepressants during pregnancy.
Psychiatric comorbidities were also significantly more common among individuals with ADHD in the year preceding pregnancy, compared with those without ADHD. The authors saw a 17-fold higher odds of receiving a diagnosis of bipolar disorder, nearly 8-fold higher odds of a diagnosis of schizophrenia or other psychotic disorder, and 22-fold higher odds of being diagnosed with emotionally unstable personality disorder among women and girls with ADHD versus those without.
The authors commented that antenatal care should focus on trying to reduce such obstetric risk factors in these women, but also pointed out that ADHD in women and girls was still underdiagnosed and undertreated.
Commenting on the association between ADHD and teenage pregnancy, the authors noted that women and girls with ADHD may be less likely to receive adequate contraceptive counseling and less likely to access, respond to, and act on counseling. They may also experience more adverse effects from hormonal contraceptives.
While Swedish youth clinics enable easier and low-cost access to counseling and contraception, the authors called for greater collaboration between psychiatric care clinics and specialized youth clinics to provide adequate care for women and girls with ADHD.
Three authors declared advisory board positions, grants, personal fees, and speakers’ fees from the pharmaceutical sector. No other conflicts of interest were declared.
SOURCE: Skoglund C et al. JAMA Netw Open. 2019 Oct 2. doi: 10.1001/jamanetworkopen.2019.12463
FROM JAMA NETWORK OPEN
An overview of endoscopy in neurologic surgery
Over the last 3 decades, the endoscope has become a highly valued visualization tool in neurosurgery, applicable to a broad range of neurosurgical procedures. Following the pace of technological innovations, the quality of the instrumentation has greatly improved along with the status of endoscopy in the neurosurgical field. The use of the endoscope in interdisciplinary extended transnasal approaches revolutionized skull-base surgery.1 Transcranial neurosurgery took advantage of the endoscope for inspection, endoscope-assisted, and endoscope-controlled procedures, although the main visualization tool during these interventions remains the operating microscope.
At present, endoscopy has applications in a variety of neurosurgical procedures including transnasal approaches for pituitary and other skull-base tumors, third ventriculostomy, and resection of intraventricular tumors. The range of application is expanding to include extracranial procedures such as peripheral nerve and spine surgery.
optics and instruments are passed through a rigid, multiport chamber. This technique is ideal when performing surgery within the ventricular system using only a standard bur hole craniotomy.
CURRENT CONCEPTS
Hopf and Perneczky2 defined the terminology regarding endoscopic procedures and divided them into 3 categories:
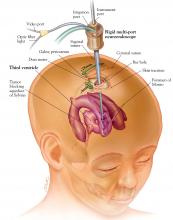
Pure endoscopic neurosurgery, ie, procedures performed through working channels under complete endoscopic visualization and with endoscopic instrumentation (Figure 1).3
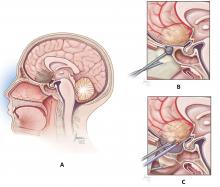
Endoscope-controlled microsurgery, ie, operations performed with standard microsurgical instruments under endoscopic visualization—the microscope is not used (Figure 2).
Endoscope-assisted neurosurgery, ie, the use of both microscope and endoscope during the same intervention. In endoscopic inspection the endoscope is solely used as an adjunctive tool for visualization and not for surgical manipulations.
Enhanced area and surgical dissection
Technical innovations are probably the major reason for the growing role of endoscopy in neurosurgery over the last 3 decades.4 High-definition imaging, neuronavigation, new instruments, an interdisciplinary approach mostly with ear, nose, and throat (ENT) surgeons, and detailed anatomic studies led to the breakthrough of endoscopic endonasal extended approaches in skull-base surgery.5
These endoscopic techniques allow the neurosurgeon to optimize tumor resection, increasing the area of surgical dissection without increasing the size of the surgical approach, thereby limiting perioperative morbidity due to surgical manipulation of eloquent brain structures. Endoscopy offers direct illumination of the operative field, magnification, and the ability to look around corners with angled optics.
However, while angled endoscopic optics provide various visual perspectives, the surgical issue is not only to see but also to work on and around remote structures. Microsurgical endoscope-assisted manipulations require optimal working angles that are guaranteed only by a sufficiently large craniotomy. As an example, a dissection study by Chaynes et al6 highlights that a craniotomy that is too narrow often hinders a sufficient exploration of the entire cerebellopontine angle. Most neurosurgeons are familiar with the operating microscope. The microscopic field of inspection is 3-dimensional (3D) and of high quality. However, the light stream is straight and thus limited in the narrow and angled corridor of the cerebellopontine angle or in the perimesencephalic cisterns. In these situations, the angled optic of the endoscope offers the advantage of being able to look around the corner with the appropriate amount of direct illumination.7
Peripheral nerve surgery
Minimally invasive endoscopic approaches are also being used in peripheral nerve surgery, especially carpal tunnel decompression. The first carpal tunnel release treated endoscopically was performed by Okutsu et al in the late 1980s.8 Since that time, endoscopic carpal tunnel decompression has become very common and is the preferred method for many surgeons, using either single-portal or dual-portal techniques. Although the superiority of endoscopic over conventional minimally invasive microsurgical peripheral nerve surgeries has not been proven, large series of endoscopic carpal tunnel decompressions have reported low complication rates and excellent success rates with high patient satisfaction scores.8,9
Visualization of the spinal canal
Expanding the use of the endoscope to spine surgery, endoscopic explorations of the interlaminar spaces after having completed open surgical laminectomies have been reported since the early 1980s,10 while endoscope-assisted interlaminar procedures started in the late 1990s.11–13 The development of fully endoscopic transforaminal or interlaminar approaches for lumbar stenosis or lumbar disk herniation has been ongoing in the last 2 decades. The rationale for direct endoscopic visualization of the spinal canal is to reduce scarring of the epidural space, which might affect the outcome of possible revision surgeries (recurrent disk herniation), and to reduce injury to the paraspinal muscles, which may reduce postoperative incisional pain and length of hospital stay. Major limiting factors for fully endoscopic spine surgeries such as the narrow working channels (which are limited by the osseous perimeter of the neuroforamina, as well as the pelvis and abdominal structures) and the learning curve for the surgeons are, however, still matters of debate and restrict the use of endoscopy to very carefully selected cases.14,15
Pediatric craniosynostosis
Recently, the use of the endoscope has extended to treatment of craniosynostosis in pediatric patients, historically treated with large and occasionally staged craniotomic approaches. A meta-analysis of the literature showed statistically significant reductions in blood loss and rates of perioperative complications, reoperation, and transfusion compared with open approaches.16
Technical limitations
While neurosurgeons increasingly advocate the use of the endoscope in their practice, the development of instruments for endoscopic surgery does not always follow the same pace. There are technical problems with current rigid endoscopes and ergonomic limitations of the endoscope-assisted techniques in transcranial neurosurgery. The endoscope itself occupies space in an already limited surgical corridor like the posterior fossa, the parasellar space, or the intraventricular region. The ideal endoscope is thin and sturdy, does not generate heat, and provides high-resolution images. In addition, a self-irrigating feature could minimize the need to remove and reinsert the endoscope for cleaning. Finally, most intracranial surgery is extremely delicate and requires bimanual dissection. The ideal endoscope should also be easily integrated with a holder that allows the surgeon to easily transition between static and dynamic endoscope movements.
Newer flexible fiberscopes with even smaller diameters are likely to be launched on the market in the near future. When working in a surgical corridor less than 10 mm wide, this difference could be substantial.
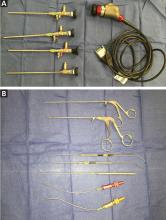
In addition, surgical instruments specifically designed for endoscopic endonasal procedures are needed for microdissection in these regions, which were previously only visible but not reachable endoscopically. These include tools such as malleable suctions and curettes, rotatable back-biting microscissors, and malleable bipolar instruments (Figure 3).
IMPACT OF NEUROENDOSCOPY IN CURRENT CLINICAL PRACTICE
The introduction of endoscopy in neurosurgery changed many treatment paradigms and had an important impact on morbidity and outcomes. In this section, we discuss the specific indications, contraindications, and expected benefit of endoscopic vs open surgical approaches applied to neurosurgical pathology at the present time.
Skull-base tumors and CSF leaks
The use of the endoscope in skull-base surgery was originally applied to purely midline intrasellar tumors without suprasellar or lateral extension beyond the carotid cave. Ideal cases were intrasellar pituitary microadenomas not responding to medical treatment or Rathke cleft cysts.
These pathologies were traditionally addressed via microscopic craniotomic approaches and later through sublabial or transnasal transsphenoidal approaches. Traditional transsphenoidal approaches were highly invasive for the oral mucosa, causing delayed healing, oral dysesthesia, and, in some cases, loss of the superior dental arch (sublabial) or limited visualization and surgical maneuverability (microscopic endonasal).
The endoscope offered better visualization and surgical freedom, thus allowing higher resection rates to be achieved. Resection of purely intrasellar pathology with preservation of the diaphragma sellae as a barrier to the subarachnoid cysterns and third ventricle guaranteed a lower incidence of cerebrospinal fluid (CSF) leaks.
New endoscope optics with varied angles, together with dedicated long surgical instruments with low steric volume, offered a large variety of new endonasal surgical corridors, so-called expanded endonasal approaches on the sagittal and coronal planes, as discussed in detail by Kassam et al.17–19 These allowed endoscopic treatment of invasive tumors extending on the coronary plane into the suprasellar region or invading the cavernous sinuses (pituitary macroadenomas, craniopharyngiomas).
Highly specialized centers with expertise in endoscopic skull-base surgery can now also offer pure endoscopic treatment for some selected cases of lesions located far laterally to the cavernous sinus, such as trigeminal schwannomas, or along the sagittal plane like olfactory groove or tuberculum sellae meningiomas and clival lesions (chordomas, chondrosarcomas).
As one might expect, the increase in surgical complexity corresponded to an increase in complication rates. For example, the incidence of CSF leaks varied from 5% for standard midline transsphenoidal approaches to 11% for expanded endonasal approaches.20,21 The consolidation of the use of the endoscope and the cooperation with ENT surgeons led to the development of surgical strategies to prevent and reduce the incidence of CSF leaks, such as the use of “rescue flaps,” nasoseptal flaps, or temporoparietal fascia flaps.21–23
The development of such techniques allowed endoscopic endonasal approaches to be used in treatment of other pathologies, such as spontaneous CSF leaks, treated in the past with large transcranial repairs that carried high morbidity rates due to the surgical frontal lobe retraction and injury to the olfactory mucosa.24,25 Progress in the field of neuroendoscopy therefore led to the creation of specialized endoscopic skull-base surgery centers, including neurosurgery, ENT, ophthalmology, and endocrinology services.
In clinical practice, when evaluating a patient with intracranial skull-base pathology amenable to endoscopic resection, one should consider referring the patient not only to a neurosurgeon, but also to an ENT surgeon for preoperative assessment of the sinonasal cavities. The same concept applies to postsurgical follow-up, which is mostly performed by the ENT physician to assess nasal mucosa healing and nasal hygiene.
Ventricular neuroendoscopy
The introduction of endoscopic third ventriculostomy created the opportunity to offer a more physiologic treatment in selected patients with obstructive hydrocephalus by creating an internal CSF diversion through the basal cisterns. Two advantages of this procedure are that it does not create dependence on a CSF shunt, and it eliminates the related risks of shunt infection and malfunction. Its drawback is the recurrence rate of hydrocephalus (around 58% at 2 years of follow-up) due to formation of scarring in the perforated Liliequist membrane, which may require repeat surgery or conversion to CSF shunting.26,27
Neuroendoscopic approaches are also used in cases of purely intraventricular pathology such as colloid cyst or choroid plexus papillomas. The concept behind neuroendoscopy is to achieve maximal resection in a minimally invasive way, using the natural cavity of the cerebral ventricles and reducing the need for brain retraction and, in particular, the risk of injury of the fornix (therefore causing memory deficits) of open transventricular approaches and of the corpus callosum necessary in interhemispheric approaches. Large tumor size and inability to tolerate a longer surgical procedure can be relative contraindications to a pure endoscopic approach to these lesions.
Degenerative spine disease
In recent years there has been a growing interest in the use of endoscopy for selected cases of degenerative lumbar spondylosis (generally, lateral disk herniation above the L5-S1 level or spinal canal stenosis). This approach has been shown to reduce postoperative incisional pain, scarring of the epidural space affecting the outcome of possible revision surgeries (recurrent disc herniation), and length of hospital stay.14,15 Information on surgical nuances should be provided when consulting on selected patients with lumbar degenerative disease resistant to conservative treatment.
Carpal tunnel syndrome
Although endoscopic carpal tunnel release is controversial, its supporters report smaller incision size and lower recurrence rates due to better visualization of the entire carpal ligament compared with open surgery, with high patient satisfaction scores.8,9,28
Craniosynostosis
Increasing data from specialized centers show that early endoscopic suturectomy is an effective treatment option alone or when combined with open surgeries for patients with syndromic and nonsyndromic craniosynostosis. The aesthetic advantage of small incisions (which can also be achieved with some open techniques) is accompanied by significant reductions in blood loss (median 162.4 mL), operative time (median 112.38 minutes), length of stay (median 2.56 days), and rates of perioperative complications (odds ratio 0.58), reoperation (odds ratio 0.37), and transfusion (odds ratio 0.09) compared with open approaches.16
SURGICAL TRAINING
Today’s patients expect high-quality healthcare, and they approach their surgeons with an enormous amount of information collected through unlimited Web-based access or peer-group blogs. In this respect, the pressure on young surgeons to achieve excellent results is high and growing from the very beginning of their careers.
Residency training programs differ in each country, and surgical standards usually focus on open microscopic procedures rather than newly developed endoscopic techniques. Endoscopic pituitary adenoma surgery, the most frequent neuroendoscopic procedure, is still performed mostly by experienced neurosurgeons, not trainees. Moreover, many training institutions might not offer pediatric neurosurgery care, limiting exposure to endoscopic third ventriculostomy procedures. The European Union of Medical Specialists, responsible for harmonizing and improving the quality of training of medical specialists in Europe, set low neuroendoscopic surgical requirements for trainees to complete their residency programs (minimum of 0 to optimum of 5 total transcranial or transsphenoidal pituitary adenoma resections as first operator, 10 procedures as assistant, and a minimum of 2 to an optimum of 4 endoscopic third ventriculostomies as first operator).29
The need to develop training programs in neuroendoscopy is especially urgent because endoscopic surgery has a steeper learning curve than conventional microneurosurgery. In particular, endoscopy requires a good deal of dexterity and hand-eye coordination, which surgeons consider the main pitfall of neuroendoscopy. For such reasons, many accredited clinical fellowship programs have been developed inside and outside North America that offer intensive training in endoscopic skull-base surgery and pediatric neurosurgery after residency.
Some clinical studies have shown that the complication rate of neuroendoscopy is 15% to 18%.27,30 In view of this statistic, it is ethically questionable to perform a randomized study to prospectively compare microscopic and endoscopic procedures. Surgeons specialize in one technique or the other, experience their own learning curve, and do not randomly decide which tool to use. Furthermore, every intracranial surgical exploration is unique and somewhat difficult to compare with each other without the risk of bias.
FURTHER DEVELOPMENTS
Multivariable rigid endoscopes like the EndoCAMeleon (Karl Storz, Tuttlingen, Germany) or the EndActive (Karl Storz, Tuttlingen, Germany) for cerebellopontine angle surgery represent a starting point to overcome some of the aforementioned limitations.31,32 They are inserted in the surgical field with a direct 0° angulation view into the operative site beyond neurovascular structures that need to be preserved and that obstruct the microscopic view. Once the final position is reached, the field of view is directed toward the region of interest without moving the endoscope tip.
The EndoCAMeleon is a rigid rod-lens endoscope, steerable in one plane from –10° to +120° by a fine optomechanical mechanism. Anatomic laboratory testing found it to be superior in terms of usability and visualization compared with rigid fixed-angle endoscopes.31 The first clinical experiences have been promising; however, ergonomics and the limited perspective of a single plane of rotation leave room for improvement.
The EndActive endoscope might overcome such limitations.33 This device is a rigid videoendoscope connected to a laptop (video data) and USB port (control and power supply); thus, it weighs less and can be held in one hand like a microsurgical instrument. The endoscopic imaging system allows the operator to simultaneously see a 160° wide-angle view of the site and an inset of a specific region of interest. The surgeon can hold the device like a microsurgical instrument in one hand and control movements precisely due to its reduced weight and ergonomic shape.
The multiplanar variable-view rigid endoscope has proven to be useful for working on diverse anatomic structures such as intracranial vessels and cranial nerves. The device is effective in narrow working spaces where even small movements can jeopardize the delicate surrounding structures. The multiplanar variable-viewing mechanism in a compact device offers advantages in terms of safety and ergonomics. Improving the usability will probably optimize the applicability of those endoscopic devices in neurosurgery. A major drawback of the current prototype is poor image resolution, which will probably soon be overcome with the ongoing progress in electronic microchip technology.
The addition of laser technology to endoscopic techniques offers a huge potential to neurosurgery but has achieved little acceptance to date. The reasons include concern regarding heat production, uncontrollable and distant penetration, and tissue interaction. Experiences with a 2-micron continuous- wave laser (RevoLix Jr, LISA Laser Products, Katlenburg-Lindau, Germany) for neuroendoscopic intraventricular procedures proved this laser to be a valuable and useful tool with safe applicability for endoscopic intracranial procedures in patients of all ages.34
Parallel to the launch of video screens for other uses with higher image definition, the image quality on the 2D endoscope cameras has been constantly improving over the last years. At the same time, the introduction of modern 3D endoscopic monitors is promising. However, 3D endoscopes have some disadvantages compared with the 2D endoscopes. First, the smallest 3D endoscopes are 4 mm in diameter, compared with 2.7 mm for 2D endoscopes. Moreover, the field of view with the 3D endoscope is less than half of that with conventional 2D endoscopes.34 When working in and around a region with critical neurovascular structures in close proximity, this loss of field of view can result in an increase in iatrogenic injury from the endoscope. In addition, 3D endoscopes require special glasses, generating a potential obstacle to the seamless integration of visual information from the microscope and endoscope. Finally, some surgeons experience vertigo when looking at the 3D picture through the glasses, which limits its universal applicability.
CONCLUSIONS
Using the endoscope and microscope as complementary and not competing tools allows surgeons to benefit from both technologies at the same time.35,36 The intraoperative combination of these 2 powerful visualization tools expands the effectiveness of microsurgical procedures and has the potential to further improve surgical results and reduce surgical risks. With endoscope-assisted microsurgery, visualization is often far superior to surgical maneuverability.
Endoscopic neurosurgery will likely be influenced by further innovations in optical physics, electronics, and robotics. Specific implementations in endoscopic systems are likely to pave the way for remarkable progress in minimally invasive surgery, such as robotic surgical technology, further miniaturization of devices, improvements in 3D endoscopy, multiport endoscopy, and new designs for surgical instruments. Future progress in flexible endoscopes and wireless capsule or camera technology may reduce our dependence on rigid rod lens systems. Rigid variable-view endoscopes will bring endoscopes closer to ideal attributes utilizing newer instrumentation that is tailored to specific indications and techniques.37,38 Extension of the visual field by the feature of a movable optic lens may allow the neurosurgeon to use tailored keyhole approaches to treat pathologies in smaller surgical corridors with less trauma and greater efficacy.
- Kassam AB, Gardner P, Snyderman C, Mintz A, Carrau R. Expanded endonasal approach: fully endoscopic, completely transnasal approach to the middle third of the clivus, petrous bone, middle cranial fossa, and infratemporal fossa. Neurosurg Focus 2005; 19(1):E6. pmid:16078820
- Hopf NJ, Perneczky A. Endoscopic neurosurgery and endoscope-assisted microneurosurgery for the treatment of intracranial cysts. Neurosurgery 1998; 43(6):1330–1336. doi:10.1097/00006123-199812000-00037
- Li KW, Nelson C, Suk I, Jallo GI. Neuroendoscopy: past, present, and future. Neurosurg Focus 2005; 19(6):E1. doi:10.3171/foc.2005.19.6.2
- Prevedello DM, Doglietto F, Jane JA Jr, Jagannathan J, Han J, Laws ER Jr. History of endoscopic skull base surgery: its evolution and current reality. J Neurosurg 2007; 107(1):206–213. doi:10.3171/JNS-07/07/0206
- Schroeder HW, Nehlsen M. Value of high-definition imaging in neuroendoscopy. Neurosurg Rev 2009; 32(3):303–308. doi:10.1007/s10143-009-0200-x
- Chaynes P, Deguine O, Moscovici J, Fraysse B, Becue J, Lazorthes Y. Endoscopic anatomy of the cerebellopontine angle: a study in cadaver brains. Neurosurg Focus 1998; 5(3):e8.
- Setty P, Volkov AA, D'Andrea KP, Pieper DR. Endoscopic vascular decompression for the treatment of trigeminal neuralgia: clinical outcomes and technical note. World Neurosurg 2014; 81(3–4):603–608. doi:10.1016/j.wneu.2013.10.036
- Okutsu I, Hamanaka I, Yoshida A. Retrospective analysis of five-year and longer clinical and electrophysiological results of the world's first endoscopic management for carpal tunnel syndrome. Hand Surg 2013; 18(3):317–323. doi:10.1142/S0218810413500330
- Zuo D, Zhou Z, Wang H, et al. Endoscopic versus open carpal tunnel release for idiopathic carpal tunnel syndrome: a meta-analysis of randomized controlled trials. J Orthop Surg Res 2015; 10:12. doi:10.1186/s13018-014-0148-6
- Forst R, Hausmann B. Nucleoscopy—a new examination technique. Arch Orthop Trauma Surg 1983; 101(3):219–221. pmid:6870510
- Brayda-Bruno M, Cinnella P. Posterior endoscopic discectomy (and other procedures). Eur Spine J 2000; 9(suppl 1):S24–S29. pmid:10766054
- Destandau J. A special device for endoscopic surgery of lumbar disc herniation. Neurol Res 1999; 21(1):39–42. pmid:10048052
- Perez-Cruet MJ, Foley KT, Isaacs RE, et al. Microendoscopic lumbar discectomy: technical note. Neurosurgery 2002; 51(5 suppl):S129–S136. pmid:12234440
- Ruetten S, Komp M, Merk H, Godolias G. Full-endoscopic interlaminar and transforaminal lumbar discectomy versus conventional microsurgical technique: a prospective, randomized, controlled study. Spine (Phila Pa 1976) 2008; 33(9):931–939. doi:10.1097/BRS.0b013e31816c8af7
- Komp M, Hahn P, Merk H, Godolias G, Ruetten S. Bilateral operation of lumbar degenerative central spinal stenosis in full-endoscopic interlaminar technique with unilateral approach: prospective 2-year results of 74 patients. J Spinal Disord Tech 2011; 24(5):281–287. doi:10.1097/BSD.0b013e3181f9f55e
- Goyal A, Lu VM, Yolcu YU, Elminawy M, Daniels DJ. Endoscopic versus open approach in craniosynostosis repair: a systematic review and meta-analysis of perioperative outcomes. Childs Nerv Syst 2018; 34(9):1627–1637. doi:10.1007/s00381-018-3852-4
- Kassam AB, Gardner P, Snyderman C, Mintz A, Carrau R. Expanded endonasal approach: fully endoscopic, completely transnasal approach to the middle third of the clivus, petrous bone, middle cranial fossa, and infratemporal fossa. Neurosurg Focus 2005; 19(1):E6. pmid:16078820
- Kassam A, Snyderman CH, Mintz A, Gardner P, Carrau RL. Expanded endonasal approach: the rostrocaudal axis. Part II. Posterior clinoids to the foramen magnum. Neurosurg Focus 2005; 19(1):E4. pmid:16078818
- Kassam A, Snyderman CH, Mintz A, Gardner P, Carrau RL. Expanded endonasal approach: the rostrocaudal axis. Part I. Crista galli to the sella turcica. Neurosurg Focus 2005; 19(1):E3. pmid:16078817
- Kassam A, Carrau RL, Snyderman CH, Gardner P, Mintz A. Evolution of reconstructive techniques following endoscopic expanded endonasal approaches. Neurosurg Focus 2005; 19(1):E8. pmid:16078822
- Kassam AB, Thomas A, Carrau RL, et al. Endoscopic reconstruction of the cranial base using a pedicled nasoseptal flap. Neurosurgery 2008; 63(1 suppl 1):ONS44–ONS52. doi:10.1227/01.NEU.0000297074.13423.F5
- Hadad G, Bassagasteguy L, Carrau RL, et al. A novel reconstructive technique after endoscopic expanded endonasal approaches: vascular pedicle nasoseptal flap. Laryngoscope 2006; 116(10):1882–1886. doi:10.1097/01.mlg.0000234933.37779.e4
- Fortes FS, Carrau RL, Snyderman CH, et al. Transpterygoid transposition of a temporoparietal fascia flap: a new method for skull base reconstruction after endoscopic expanded endonasal approaches. Laryngoscope 2007; 117(6):970–976. doi:10.1097/MLG.0b013e3180471482
- Carrau RL, Snyderman CH, Kassam AB. The management of cerebrospinal fluid leaks in patients at risk for high-pressure hydrocephalus. Laryngoscope 2005; 115(2):205–212. doi:10.1097/01.mlg.0000154719.62668.70
- Zweig JL, Carrau RL, Celin SE, et al. Endoscopic repair of cerebrospinal fluid leaks to the sinonasal tract: predictors of success. Otolaryngol Head Neck Surg 2000; 123(3):195–201. doi:10.1067/mhn.2000.107452
- Kulkarni AV, Riva-Cambrin J, Holubkov R, et al. Endoscopic third ventriculostomy in children: prospective, multicenter results from the Hydrocephalus Clinical Research Network. J Neurosurg Pediatr 2016; 18(4):423–429. doi:10.3171/2016.4.PEDS163
- Ersahin Y, Arslan D. Complications of endoscopic third ventriculostomy. Childs Nerv Syst 2008; 24(8):943–948. doi:10.1007/s00381-008-0589-5
- Martínez-Catasús A, Lobo-Escolar L, García-Bonet J, Corrales-Rodríguez M, Pasarín-Martínez A, Berlanga-de-Mingo D. Comparison between single portal endoscopic, 1-cm open carpal tunnel release. Hand Surg Rehabil 2019. pii:S2468-1229(19)30027-1. doi:10.1016/j.hansur.2019.02.003
- Steers J, Reulen HJ, Lindsay K; European Union of Medical Specialists; Joint Residency Advisory and Accreditation Committee. UEMS charter on training of medical specialists in the EU—the new neurosurgical training charter. Acta Neurochir Suppl 2004; 90:3–11. pmid:15553111
- Mori H, Nishiyama K, Yoshimura J, Tanaka R. Current status of neuroendoscopic surgery in Japan and discussion on the training system. Childs Nerv Syst 2007; 23(6):673–676. doi:10.1007/s00381-007-0329-2
- Aryan HE, Hoeg HD, Marshall LF, Levy ML. Multidirectional projectional rigid neuro-endoscopy: prototype and initial experience. Minim Invasive Neurosurg 2005; 48(5):293–296. doi:10.1055/s-2005-915602
- Ebner FH, Marquardt JS, Hirt B, Tatagiba M, Schuhmann MU. Visualization of the anterior cerebral artery complex with a continuously variable-view rigid endoscope: new options in aneurysm surgery. Neurosurgery 2010; 67(2 suppl operative):321–324. doi:10.1227/NEU.0b013e3181f74548
- Ebner FH, Hirt B, Marquardt JS, Herlan S, Tatagiba M, Schuhmann MU. Actual state of EndActive ventricular endoscopy. Childs Nerv Syst 2012; 28(1):87–91. doi:10.1007/s00381-011-1537-3
- Ebner FH, Nagel C, Tatagiba M, Schuhmann MU. Efficacy and versatility of the 2-micron continuous wave laser in neuroendoscopic procedures. Acta Neurochir Suppl 2012; 113:143–147. doi:10.1007/978-3-7091-0923-6_29
- Van Gompel JJ, Tabor MH, Youssef AS, et al. Field of view comparison between two-dimensional and three-dimensional endoscopy. Laryngoscope 2014; 124(2):387–390. doi:10.1002/lary.24222
- Ebner FH, Roser F, Thaher F, Schittenhelm J, Tatagiba M. Balancing the shortcomings of microscope and endoscope: endoscope-assisted technique in microsurgical removal of recurrent epidermoid cysts in the posterior fossa. Minim Invasive Neurosurg 2010 ;53(5–6):218–222. doi:10.1055/s-0030-1267973
- Perneczky A, Fries G. Endoscope-assisted brain surgery: part 1—evolution, basic concept, and current technique. Neurosurgery 1998; 42(2):219–224. doi:10.1097/00006123-199802000-00001
- Ebner FH, Marquardt JS, Hirt B, Feigl GC, Tatagiba M, Schuhmann MU. Broadening horizons of neuroendoscopy with a variable-view rigid endoscope: an anatomical study. Eur J Surg Oncol 2010; 36(2):195–200. doi:10.1016/j.ejso.2009.07.185
meningioma, peripheral nerve, spinal canal, minimally invasive, carpal tunnel, ventricular neuroendoscopy, craniosynostosis, degenerative spine disease, Luigi Rigante, Hamid Borghei-Razavi, Pablo Recinos, Florian Roser
Over the last 3 decades, the endoscope has become a highly valued visualization tool in neurosurgery, applicable to a broad range of neurosurgical procedures. Following the pace of technological innovations, the quality of the instrumentation has greatly improved along with the status of endoscopy in the neurosurgical field. The use of the endoscope in interdisciplinary extended transnasal approaches revolutionized skull-base surgery.1 Transcranial neurosurgery took advantage of the endoscope for inspection, endoscope-assisted, and endoscope-controlled procedures, although the main visualization tool during these interventions remains the operating microscope.
At present, endoscopy has applications in a variety of neurosurgical procedures including transnasal approaches for pituitary and other skull-base tumors, third ventriculostomy, and resection of intraventricular tumors. The range of application is expanding to include extracranial procedures such as peripheral nerve and spine surgery.
optics and instruments are passed through a rigid, multiport chamber. This technique is ideal when performing surgery within the ventricular system using only a standard bur hole craniotomy.
CURRENT CONCEPTS
Hopf and Perneczky2 defined the terminology regarding endoscopic procedures and divided them into 3 categories:
Pure endoscopic neurosurgery, ie, procedures performed through working channels under complete endoscopic visualization and with endoscopic instrumentation (Figure 1).3
Endoscope-controlled microsurgery, ie, operations performed with standard microsurgical instruments under endoscopic visualization—the microscope is not used (Figure 2).
Endoscope-assisted neurosurgery, ie, the use of both microscope and endoscope during the same intervention. In endoscopic inspection the endoscope is solely used as an adjunctive tool for visualization and not for surgical manipulations.
Enhanced area and surgical dissection
Technical innovations are probably the major reason for the growing role of endoscopy in neurosurgery over the last 3 decades.4 High-definition imaging, neuronavigation, new instruments, an interdisciplinary approach mostly with ear, nose, and throat (ENT) surgeons, and detailed anatomic studies led to the breakthrough of endoscopic endonasal extended approaches in skull-base surgery.5
These endoscopic techniques allow the neurosurgeon to optimize tumor resection, increasing the area of surgical dissection without increasing the size of the surgical approach, thereby limiting perioperative morbidity due to surgical manipulation of eloquent brain structures. Endoscopy offers direct illumination of the operative field, magnification, and the ability to look around corners with angled optics.
However, while angled endoscopic optics provide various visual perspectives, the surgical issue is not only to see but also to work on and around remote structures. Microsurgical endoscope-assisted manipulations require optimal working angles that are guaranteed only by a sufficiently large craniotomy. As an example, a dissection study by Chaynes et al6 highlights that a craniotomy that is too narrow often hinders a sufficient exploration of the entire cerebellopontine angle. Most neurosurgeons are familiar with the operating microscope. The microscopic field of inspection is 3-dimensional (3D) and of high quality. However, the light stream is straight and thus limited in the narrow and angled corridor of the cerebellopontine angle or in the perimesencephalic cisterns. In these situations, the angled optic of the endoscope offers the advantage of being able to look around the corner with the appropriate amount of direct illumination.7
Peripheral nerve surgery
Minimally invasive endoscopic approaches are also being used in peripheral nerve surgery, especially carpal tunnel decompression. The first carpal tunnel release treated endoscopically was performed by Okutsu et al in the late 1980s.8 Since that time, endoscopic carpal tunnel decompression has become very common and is the preferred method for many surgeons, using either single-portal or dual-portal techniques. Although the superiority of endoscopic over conventional minimally invasive microsurgical peripheral nerve surgeries has not been proven, large series of endoscopic carpal tunnel decompressions have reported low complication rates and excellent success rates with high patient satisfaction scores.8,9
Visualization of the spinal canal
Expanding the use of the endoscope to spine surgery, endoscopic explorations of the interlaminar spaces after having completed open surgical laminectomies have been reported since the early 1980s,10 while endoscope-assisted interlaminar procedures started in the late 1990s.11–13 The development of fully endoscopic transforaminal or interlaminar approaches for lumbar stenosis or lumbar disk herniation has been ongoing in the last 2 decades. The rationale for direct endoscopic visualization of the spinal canal is to reduce scarring of the epidural space, which might affect the outcome of possible revision surgeries (recurrent disk herniation), and to reduce injury to the paraspinal muscles, which may reduce postoperative incisional pain and length of hospital stay. Major limiting factors for fully endoscopic spine surgeries such as the narrow working channels (which are limited by the osseous perimeter of the neuroforamina, as well as the pelvis and abdominal structures) and the learning curve for the surgeons are, however, still matters of debate and restrict the use of endoscopy to very carefully selected cases.14,15
Pediatric craniosynostosis
Recently, the use of the endoscope has extended to treatment of craniosynostosis in pediatric patients, historically treated with large and occasionally staged craniotomic approaches. A meta-analysis of the literature showed statistically significant reductions in blood loss and rates of perioperative complications, reoperation, and transfusion compared with open approaches.16
Technical limitations
While neurosurgeons increasingly advocate the use of the endoscope in their practice, the development of instruments for endoscopic surgery does not always follow the same pace. There are technical problems with current rigid endoscopes and ergonomic limitations of the endoscope-assisted techniques in transcranial neurosurgery. The endoscope itself occupies space in an already limited surgical corridor like the posterior fossa, the parasellar space, or the intraventricular region. The ideal endoscope is thin and sturdy, does not generate heat, and provides high-resolution images. In addition, a self-irrigating feature could minimize the need to remove and reinsert the endoscope for cleaning. Finally, most intracranial surgery is extremely delicate and requires bimanual dissection. The ideal endoscope should also be easily integrated with a holder that allows the surgeon to easily transition between static and dynamic endoscope movements.
Newer flexible fiberscopes with even smaller diameters are likely to be launched on the market in the near future. When working in a surgical corridor less than 10 mm wide, this difference could be substantial.
In addition, surgical instruments specifically designed for endoscopic endonasal procedures are needed for microdissection in these regions, which were previously only visible but not reachable endoscopically. These include tools such as malleable suctions and curettes, rotatable back-biting microscissors, and malleable bipolar instruments (Figure 3).
IMPACT OF NEUROENDOSCOPY IN CURRENT CLINICAL PRACTICE
The introduction of endoscopy in neurosurgery changed many treatment paradigms and had an important impact on morbidity and outcomes. In this section, we discuss the specific indications, contraindications, and expected benefit of endoscopic vs open surgical approaches applied to neurosurgical pathology at the present time.
Skull-base tumors and CSF leaks
The use of the endoscope in skull-base surgery was originally applied to purely midline intrasellar tumors without suprasellar or lateral extension beyond the carotid cave. Ideal cases were intrasellar pituitary microadenomas not responding to medical treatment or Rathke cleft cysts.
These pathologies were traditionally addressed via microscopic craniotomic approaches and later through sublabial or transnasal transsphenoidal approaches. Traditional transsphenoidal approaches were highly invasive for the oral mucosa, causing delayed healing, oral dysesthesia, and, in some cases, loss of the superior dental arch (sublabial) or limited visualization and surgical maneuverability (microscopic endonasal).
The endoscope offered better visualization and surgical freedom, thus allowing higher resection rates to be achieved. Resection of purely intrasellar pathology with preservation of the diaphragma sellae as a barrier to the subarachnoid cysterns and third ventricle guaranteed a lower incidence of cerebrospinal fluid (CSF) leaks.
New endoscope optics with varied angles, together with dedicated long surgical instruments with low steric volume, offered a large variety of new endonasal surgical corridors, so-called expanded endonasal approaches on the sagittal and coronal planes, as discussed in detail by Kassam et al.17–19 These allowed endoscopic treatment of invasive tumors extending on the coronary plane into the suprasellar region or invading the cavernous sinuses (pituitary macroadenomas, craniopharyngiomas).
Highly specialized centers with expertise in endoscopic skull-base surgery can now also offer pure endoscopic treatment for some selected cases of lesions located far laterally to the cavernous sinus, such as trigeminal schwannomas, or along the sagittal plane like olfactory groove or tuberculum sellae meningiomas and clival lesions (chordomas, chondrosarcomas).
As one might expect, the increase in surgical complexity corresponded to an increase in complication rates. For example, the incidence of CSF leaks varied from 5% for standard midline transsphenoidal approaches to 11% for expanded endonasal approaches.20,21 The consolidation of the use of the endoscope and the cooperation with ENT surgeons led to the development of surgical strategies to prevent and reduce the incidence of CSF leaks, such as the use of “rescue flaps,” nasoseptal flaps, or temporoparietal fascia flaps.21–23
The development of such techniques allowed endoscopic endonasal approaches to be used in treatment of other pathologies, such as spontaneous CSF leaks, treated in the past with large transcranial repairs that carried high morbidity rates due to the surgical frontal lobe retraction and injury to the olfactory mucosa.24,25 Progress in the field of neuroendoscopy therefore led to the creation of specialized endoscopic skull-base surgery centers, including neurosurgery, ENT, ophthalmology, and endocrinology services.
In clinical practice, when evaluating a patient with intracranial skull-base pathology amenable to endoscopic resection, one should consider referring the patient not only to a neurosurgeon, but also to an ENT surgeon for preoperative assessment of the sinonasal cavities. The same concept applies to postsurgical follow-up, which is mostly performed by the ENT physician to assess nasal mucosa healing and nasal hygiene.
Ventricular neuroendoscopy
The introduction of endoscopic third ventriculostomy created the opportunity to offer a more physiologic treatment in selected patients with obstructive hydrocephalus by creating an internal CSF diversion through the basal cisterns. Two advantages of this procedure are that it does not create dependence on a CSF shunt, and it eliminates the related risks of shunt infection and malfunction. Its drawback is the recurrence rate of hydrocephalus (around 58% at 2 years of follow-up) due to formation of scarring in the perforated Liliequist membrane, which may require repeat surgery or conversion to CSF shunting.26,27
Neuroendoscopic approaches are also used in cases of purely intraventricular pathology such as colloid cyst or choroid plexus papillomas. The concept behind neuroendoscopy is to achieve maximal resection in a minimally invasive way, using the natural cavity of the cerebral ventricles and reducing the need for brain retraction and, in particular, the risk of injury of the fornix (therefore causing memory deficits) of open transventricular approaches and of the corpus callosum necessary in interhemispheric approaches. Large tumor size and inability to tolerate a longer surgical procedure can be relative contraindications to a pure endoscopic approach to these lesions.
Degenerative spine disease
In recent years there has been a growing interest in the use of endoscopy for selected cases of degenerative lumbar spondylosis (generally, lateral disk herniation above the L5-S1 level or spinal canal stenosis). This approach has been shown to reduce postoperative incisional pain, scarring of the epidural space affecting the outcome of possible revision surgeries (recurrent disc herniation), and length of hospital stay.14,15 Information on surgical nuances should be provided when consulting on selected patients with lumbar degenerative disease resistant to conservative treatment.
Carpal tunnel syndrome
Although endoscopic carpal tunnel release is controversial, its supporters report smaller incision size and lower recurrence rates due to better visualization of the entire carpal ligament compared with open surgery, with high patient satisfaction scores.8,9,28
Craniosynostosis
Increasing data from specialized centers show that early endoscopic suturectomy is an effective treatment option alone or when combined with open surgeries for patients with syndromic and nonsyndromic craniosynostosis. The aesthetic advantage of small incisions (which can also be achieved with some open techniques) is accompanied by significant reductions in blood loss (median 162.4 mL), operative time (median 112.38 minutes), length of stay (median 2.56 days), and rates of perioperative complications (odds ratio 0.58), reoperation (odds ratio 0.37), and transfusion (odds ratio 0.09) compared with open approaches.16
SURGICAL TRAINING
Today’s patients expect high-quality healthcare, and they approach their surgeons with an enormous amount of information collected through unlimited Web-based access or peer-group blogs. In this respect, the pressure on young surgeons to achieve excellent results is high and growing from the very beginning of their careers.
Residency training programs differ in each country, and surgical standards usually focus on open microscopic procedures rather than newly developed endoscopic techniques. Endoscopic pituitary adenoma surgery, the most frequent neuroendoscopic procedure, is still performed mostly by experienced neurosurgeons, not trainees. Moreover, many training institutions might not offer pediatric neurosurgery care, limiting exposure to endoscopic third ventriculostomy procedures. The European Union of Medical Specialists, responsible for harmonizing and improving the quality of training of medical specialists in Europe, set low neuroendoscopic surgical requirements for trainees to complete their residency programs (minimum of 0 to optimum of 5 total transcranial or transsphenoidal pituitary adenoma resections as first operator, 10 procedures as assistant, and a minimum of 2 to an optimum of 4 endoscopic third ventriculostomies as first operator).29
The need to develop training programs in neuroendoscopy is especially urgent because endoscopic surgery has a steeper learning curve than conventional microneurosurgery. In particular, endoscopy requires a good deal of dexterity and hand-eye coordination, which surgeons consider the main pitfall of neuroendoscopy. For such reasons, many accredited clinical fellowship programs have been developed inside and outside North America that offer intensive training in endoscopic skull-base surgery and pediatric neurosurgery after residency.
Some clinical studies have shown that the complication rate of neuroendoscopy is 15% to 18%.27,30 In view of this statistic, it is ethically questionable to perform a randomized study to prospectively compare microscopic and endoscopic procedures. Surgeons specialize in one technique or the other, experience their own learning curve, and do not randomly decide which tool to use. Furthermore, every intracranial surgical exploration is unique and somewhat difficult to compare with each other without the risk of bias.
FURTHER DEVELOPMENTS
Multivariable rigid endoscopes like the EndoCAMeleon (Karl Storz, Tuttlingen, Germany) or the EndActive (Karl Storz, Tuttlingen, Germany) for cerebellopontine angle surgery represent a starting point to overcome some of the aforementioned limitations.31,32 They are inserted in the surgical field with a direct 0° angulation view into the operative site beyond neurovascular structures that need to be preserved and that obstruct the microscopic view. Once the final position is reached, the field of view is directed toward the region of interest without moving the endoscope tip.
The EndoCAMeleon is a rigid rod-lens endoscope, steerable in one plane from –10° to +120° by a fine optomechanical mechanism. Anatomic laboratory testing found it to be superior in terms of usability and visualization compared with rigid fixed-angle endoscopes.31 The first clinical experiences have been promising; however, ergonomics and the limited perspective of a single plane of rotation leave room for improvement.
The EndActive endoscope might overcome such limitations.33 This device is a rigid videoendoscope connected to a laptop (video data) and USB port (control and power supply); thus, it weighs less and can be held in one hand like a microsurgical instrument. The endoscopic imaging system allows the operator to simultaneously see a 160° wide-angle view of the site and an inset of a specific region of interest. The surgeon can hold the device like a microsurgical instrument in one hand and control movements precisely due to its reduced weight and ergonomic shape.
The multiplanar variable-view rigid endoscope has proven to be useful for working on diverse anatomic structures such as intracranial vessels and cranial nerves. The device is effective in narrow working spaces where even small movements can jeopardize the delicate surrounding structures. The multiplanar variable-viewing mechanism in a compact device offers advantages in terms of safety and ergonomics. Improving the usability will probably optimize the applicability of those endoscopic devices in neurosurgery. A major drawback of the current prototype is poor image resolution, which will probably soon be overcome with the ongoing progress in electronic microchip technology.
The addition of laser technology to endoscopic techniques offers a huge potential to neurosurgery but has achieved little acceptance to date. The reasons include concern regarding heat production, uncontrollable and distant penetration, and tissue interaction. Experiences with a 2-micron continuous- wave laser (RevoLix Jr, LISA Laser Products, Katlenburg-Lindau, Germany) for neuroendoscopic intraventricular procedures proved this laser to be a valuable and useful tool with safe applicability for endoscopic intracranial procedures in patients of all ages.34
Parallel to the launch of video screens for other uses with higher image definition, the image quality on the 2D endoscope cameras has been constantly improving over the last years. At the same time, the introduction of modern 3D endoscopic monitors is promising. However, 3D endoscopes have some disadvantages compared with the 2D endoscopes. First, the smallest 3D endoscopes are 4 mm in diameter, compared with 2.7 mm for 2D endoscopes. Moreover, the field of view with the 3D endoscope is less than half of that with conventional 2D endoscopes.34 When working in and around a region with critical neurovascular structures in close proximity, this loss of field of view can result in an increase in iatrogenic injury from the endoscope. In addition, 3D endoscopes require special glasses, generating a potential obstacle to the seamless integration of visual information from the microscope and endoscope. Finally, some surgeons experience vertigo when looking at the 3D picture through the glasses, which limits its universal applicability.
CONCLUSIONS
Using the endoscope and microscope as complementary and not competing tools allows surgeons to benefit from both technologies at the same time.35,36 The intraoperative combination of these 2 powerful visualization tools expands the effectiveness of microsurgical procedures and has the potential to further improve surgical results and reduce surgical risks. With endoscope-assisted microsurgery, visualization is often far superior to surgical maneuverability.
Endoscopic neurosurgery will likely be influenced by further innovations in optical physics, electronics, and robotics. Specific implementations in endoscopic systems are likely to pave the way for remarkable progress in minimally invasive surgery, such as robotic surgical technology, further miniaturization of devices, improvements in 3D endoscopy, multiport endoscopy, and new designs for surgical instruments. Future progress in flexible endoscopes and wireless capsule or camera technology may reduce our dependence on rigid rod lens systems. Rigid variable-view endoscopes will bring endoscopes closer to ideal attributes utilizing newer instrumentation that is tailored to specific indications and techniques.37,38 Extension of the visual field by the feature of a movable optic lens may allow the neurosurgeon to use tailored keyhole approaches to treat pathologies in smaller surgical corridors with less trauma and greater efficacy.
Over the last 3 decades, the endoscope has become a highly valued visualization tool in neurosurgery, applicable to a broad range of neurosurgical procedures. Following the pace of technological innovations, the quality of the instrumentation has greatly improved along with the status of endoscopy in the neurosurgical field. The use of the endoscope in interdisciplinary extended transnasal approaches revolutionized skull-base surgery.1 Transcranial neurosurgery took advantage of the endoscope for inspection, endoscope-assisted, and endoscope-controlled procedures, although the main visualization tool during these interventions remains the operating microscope.
At present, endoscopy has applications in a variety of neurosurgical procedures including transnasal approaches for pituitary and other skull-base tumors, third ventriculostomy, and resection of intraventricular tumors. The range of application is expanding to include extracranial procedures such as peripheral nerve and spine surgery.
optics and instruments are passed through a rigid, multiport chamber. This technique is ideal when performing surgery within the ventricular system using only a standard bur hole craniotomy.
CURRENT CONCEPTS
Hopf and Perneczky2 defined the terminology regarding endoscopic procedures and divided them into 3 categories:
Pure endoscopic neurosurgery, ie, procedures performed through working channels under complete endoscopic visualization and with endoscopic instrumentation (Figure 1).3
Endoscope-controlled microsurgery, ie, operations performed with standard microsurgical instruments under endoscopic visualization—the microscope is not used (Figure 2).
Endoscope-assisted neurosurgery, ie, the use of both microscope and endoscope during the same intervention. In endoscopic inspection the endoscope is solely used as an adjunctive tool for visualization and not for surgical manipulations.
Enhanced area and surgical dissection
Technical innovations are probably the major reason for the growing role of endoscopy in neurosurgery over the last 3 decades.4 High-definition imaging, neuronavigation, new instruments, an interdisciplinary approach mostly with ear, nose, and throat (ENT) surgeons, and detailed anatomic studies led to the breakthrough of endoscopic endonasal extended approaches in skull-base surgery.5
These endoscopic techniques allow the neurosurgeon to optimize tumor resection, increasing the area of surgical dissection without increasing the size of the surgical approach, thereby limiting perioperative morbidity due to surgical manipulation of eloquent brain structures. Endoscopy offers direct illumination of the operative field, magnification, and the ability to look around corners with angled optics.
However, while angled endoscopic optics provide various visual perspectives, the surgical issue is not only to see but also to work on and around remote structures. Microsurgical endoscope-assisted manipulations require optimal working angles that are guaranteed only by a sufficiently large craniotomy. As an example, a dissection study by Chaynes et al6 highlights that a craniotomy that is too narrow often hinders a sufficient exploration of the entire cerebellopontine angle. Most neurosurgeons are familiar with the operating microscope. The microscopic field of inspection is 3-dimensional (3D) and of high quality. However, the light stream is straight and thus limited in the narrow and angled corridor of the cerebellopontine angle or in the perimesencephalic cisterns. In these situations, the angled optic of the endoscope offers the advantage of being able to look around the corner with the appropriate amount of direct illumination.7
Peripheral nerve surgery
Minimally invasive endoscopic approaches are also being used in peripheral nerve surgery, especially carpal tunnel decompression. The first carpal tunnel release treated endoscopically was performed by Okutsu et al in the late 1980s.8 Since that time, endoscopic carpal tunnel decompression has become very common and is the preferred method for many surgeons, using either single-portal or dual-portal techniques. Although the superiority of endoscopic over conventional minimally invasive microsurgical peripheral nerve surgeries has not been proven, large series of endoscopic carpal tunnel decompressions have reported low complication rates and excellent success rates with high patient satisfaction scores.8,9
Visualization of the spinal canal
Expanding the use of the endoscope to spine surgery, endoscopic explorations of the interlaminar spaces after having completed open surgical laminectomies have been reported since the early 1980s,10 while endoscope-assisted interlaminar procedures started in the late 1990s.11–13 The development of fully endoscopic transforaminal or interlaminar approaches for lumbar stenosis or lumbar disk herniation has been ongoing in the last 2 decades. The rationale for direct endoscopic visualization of the spinal canal is to reduce scarring of the epidural space, which might affect the outcome of possible revision surgeries (recurrent disk herniation), and to reduce injury to the paraspinal muscles, which may reduce postoperative incisional pain and length of hospital stay. Major limiting factors for fully endoscopic spine surgeries such as the narrow working channels (which are limited by the osseous perimeter of the neuroforamina, as well as the pelvis and abdominal structures) and the learning curve for the surgeons are, however, still matters of debate and restrict the use of endoscopy to very carefully selected cases.14,15
Pediatric craniosynostosis
Recently, the use of the endoscope has extended to treatment of craniosynostosis in pediatric patients, historically treated with large and occasionally staged craniotomic approaches. A meta-analysis of the literature showed statistically significant reductions in blood loss and rates of perioperative complications, reoperation, and transfusion compared with open approaches.16
Technical limitations
While neurosurgeons increasingly advocate the use of the endoscope in their practice, the development of instruments for endoscopic surgery does not always follow the same pace. There are technical problems with current rigid endoscopes and ergonomic limitations of the endoscope-assisted techniques in transcranial neurosurgery. The endoscope itself occupies space in an already limited surgical corridor like the posterior fossa, the parasellar space, or the intraventricular region. The ideal endoscope is thin and sturdy, does not generate heat, and provides high-resolution images. In addition, a self-irrigating feature could minimize the need to remove and reinsert the endoscope for cleaning. Finally, most intracranial surgery is extremely delicate and requires bimanual dissection. The ideal endoscope should also be easily integrated with a holder that allows the surgeon to easily transition between static and dynamic endoscope movements.
Newer flexible fiberscopes with even smaller diameters are likely to be launched on the market in the near future. When working in a surgical corridor less than 10 mm wide, this difference could be substantial.
In addition, surgical instruments specifically designed for endoscopic endonasal procedures are needed for microdissection in these regions, which were previously only visible but not reachable endoscopically. These include tools such as malleable suctions and curettes, rotatable back-biting microscissors, and malleable bipolar instruments (Figure 3).
IMPACT OF NEUROENDOSCOPY IN CURRENT CLINICAL PRACTICE
The introduction of endoscopy in neurosurgery changed many treatment paradigms and had an important impact on morbidity and outcomes. In this section, we discuss the specific indications, contraindications, and expected benefit of endoscopic vs open surgical approaches applied to neurosurgical pathology at the present time.
Skull-base tumors and CSF leaks
The use of the endoscope in skull-base surgery was originally applied to purely midline intrasellar tumors without suprasellar or lateral extension beyond the carotid cave. Ideal cases were intrasellar pituitary microadenomas not responding to medical treatment or Rathke cleft cysts.
These pathologies were traditionally addressed via microscopic craniotomic approaches and later through sublabial or transnasal transsphenoidal approaches. Traditional transsphenoidal approaches were highly invasive for the oral mucosa, causing delayed healing, oral dysesthesia, and, in some cases, loss of the superior dental arch (sublabial) or limited visualization and surgical maneuverability (microscopic endonasal).
The endoscope offered better visualization and surgical freedom, thus allowing higher resection rates to be achieved. Resection of purely intrasellar pathology with preservation of the diaphragma sellae as a barrier to the subarachnoid cysterns and third ventricle guaranteed a lower incidence of cerebrospinal fluid (CSF) leaks.
New endoscope optics with varied angles, together with dedicated long surgical instruments with low steric volume, offered a large variety of new endonasal surgical corridors, so-called expanded endonasal approaches on the sagittal and coronal planes, as discussed in detail by Kassam et al.17–19 These allowed endoscopic treatment of invasive tumors extending on the coronary plane into the suprasellar region or invading the cavernous sinuses (pituitary macroadenomas, craniopharyngiomas).
Highly specialized centers with expertise in endoscopic skull-base surgery can now also offer pure endoscopic treatment for some selected cases of lesions located far laterally to the cavernous sinus, such as trigeminal schwannomas, or along the sagittal plane like olfactory groove or tuberculum sellae meningiomas and clival lesions (chordomas, chondrosarcomas).
As one might expect, the increase in surgical complexity corresponded to an increase in complication rates. For example, the incidence of CSF leaks varied from 5% for standard midline transsphenoidal approaches to 11% for expanded endonasal approaches.20,21 The consolidation of the use of the endoscope and the cooperation with ENT surgeons led to the development of surgical strategies to prevent and reduce the incidence of CSF leaks, such as the use of “rescue flaps,” nasoseptal flaps, or temporoparietal fascia flaps.21–23
The development of such techniques allowed endoscopic endonasal approaches to be used in treatment of other pathologies, such as spontaneous CSF leaks, treated in the past with large transcranial repairs that carried high morbidity rates due to the surgical frontal lobe retraction and injury to the olfactory mucosa.24,25 Progress in the field of neuroendoscopy therefore led to the creation of specialized endoscopic skull-base surgery centers, including neurosurgery, ENT, ophthalmology, and endocrinology services.
In clinical practice, when evaluating a patient with intracranial skull-base pathology amenable to endoscopic resection, one should consider referring the patient not only to a neurosurgeon, but also to an ENT surgeon for preoperative assessment of the sinonasal cavities. The same concept applies to postsurgical follow-up, which is mostly performed by the ENT physician to assess nasal mucosa healing and nasal hygiene.
Ventricular neuroendoscopy
The introduction of endoscopic third ventriculostomy created the opportunity to offer a more physiologic treatment in selected patients with obstructive hydrocephalus by creating an internal CSF diversion through the basal cisterns. Two advantages of this procedure are that it does not create dependence on a CSF shunt, and it eliminates the related risks of shunt infection and malfunction. Its drawback is the recurrence rate of hydrocephalus (around 58% at 2 years of follow-up) due to formation of scarring in the perforated Liliequist membrane, which may require repeat surgery or conversion to CSF shunting.26,27
Neuroendoscopic approaches are also used in cases of purely intraventricular pathology such as colloid cyst or choroid plexus papillomas. The concept behind neuroendoscopy is to achieve maximal resection in a minimally invasive way, using the natural cavity of the cerebral ventricles and reducing the need for brain retraction and, in particular, the risk of injury of the fornix (therefore causing memory deficits) of open transventricular approaches and of the corpus callosum necessary in interhemispheric approaches. Large tumor size and inability to tolerate a longer surgical procedure can be relative contraindications to a pure endoscopic approach to these lesions.
Degenerative spine disease
In recent years there has been a growing interest in the use of endoscopy for selected cases of degenerative lumbar spondylosis (generally, lateral disk herniation above the L5-S1 level or spinal canal stenosis). This approach has been shown to reduce postoperative incisional pain, scarring of the epidural space affecting the outcome of possible revision surgeries (recurrent disc herniation), and length of hospital stay.14,15 Information on surgical nuances should be provided when consulting on selected patients with lumbar degenerative disease resistant to conservative treatment.
Carpal tunnel syndrome
Although endoscopic carpal tunnel release is controversial, its supporters report smaller incision size and lower recurrence rates due to better visualization of the entire carpal ligament compared with open surgery, with high patient satisfaction scores.8,9,28
Craniosynostosis
Increasing data from specialized centers show that early endoscopic suturectomy is an effective treatment option alone or when combined with open surgeries for patients with syndromic and nonsyndromic craniosynostosis. The aesthetic advantage of small incisions (which can also be achieved with some open techniques) is accompanied by significant reductions in blood loss (median 162.4 mL), operative time (median 112.38 minutes), length of stay (median 2.56 days), and rates of perioperative complications (odds ratio 0.58), reoperation (odds ratio 0.37), and transfusion (odds ratio 0.09) compared with open approaches.16
SURGICAL TRAINING
Today’s patients expect high-quality healthcare, and they approach their surgeons with an enormous amount of information collected through unlimited Web-based access or peer-group blogs. In this respect, the pressure on young surgeons to achieve excellent results is high and growing from the very beginning of their careers.
Residency training programs differ in each country, and surgical standards usually focus on open microscopic procedures rather than newly developed endoscopic techniques. Endoscopic pituitary adenoma surgery, the most frequent neuroendoscopic procedure, is still performed mostly by experienced neurosurgeons, not trainees. Moreover, many training institutions might not offer pediatric neurosurgery care, limiting exposure to endoscopic third ventriculostomy procedures. The European Union of Medical Specialists, responsible for harmonizing and improving the quality of training of medical specialists in Europe, set low neuroendoscopic surgical requirements for trainees to complete their residency programs (minimum of 0 to optimum of 5 total transcranial or transsphenoidal pituitary adenoma resections as first operator, 10 procedures as assistant, and a minimum of 2 to an optimum of 4 endoscopic third ventriculostomies as first operator).29
The need to develop training programs in neuroendoscopy is especially urgent because endoscopic surgery has a steeper learning curve than conventional microneurosurgery. In particular, endoscopy requires a good deal of dexterity and hand-eye coordination, which surgeons consider the main pitfall of neuroendoscopy. For such reasons, many accredited clinical fellowship programs have been developed inside and outside North America that offer intensive training in endoscopic skull-base surgery and pediatric neurosurgery after residency.
Some clinical studies have shown that the complication rate of neuroendoscopy is 15% to 18%.27,30 In view of this statistic, it is ethically questionable to perform a randomized study to prospectively compare microscopic and endoscopic procedures. Surgeons specialize in one technique or the other, experience their own learning curve, and do not randomly decide which tool to use. Furthermore, every intracranial surgical exploration is unique and somewhat difficult to compare with each other without the risk of bias.
FURTHER DEVELOPMENTS
Multivariable rigid endoscopes like the EndoCAMeleon (Karl Storz, Tuttlingen, Germany) or the EndActive (Karl Storz, Tuttlingen, Germany) for cerebellopontine angle surgery represent a starting point to overcome some of the aforementioned limitations.31,32 They are inserted in the surgical field with a direct 0° angulation view into the operative site beyond neurovascular structures that need to be preserved and that obstruct the microscopic view. Once the final position is reached, the field of view is directed toward the region of interest without moving the endoscope tip.
The EndoCAMeleon is a rigid rod-lens endoscope, steerable in one plane from –10° to +120° by a fine optomechanical mechanism. Anatomic laboratory testing found it to be superior in terms of usability and visualization compared with rigid fixed-angle endoscopes.31 The first clinical experiences have been promising; however, ergonomics and the limited perspective of a single plane of rotation leave room for improvement.
The EndActive endoscope might overcome such limitations.33 This device is a rigid videoendoscope connected to a laptop (video data) and USB port (control and power supply); thus, it weighs less and can be held in one hand like a microsurgical instrument. The endoscopic imaging system allows the operator to simultaneously see a 160° wide-angle view of the site and an inset of a specific region of interest. The surgeon can hold the device like a microsurgical instrument in one hand and control movements precisely due to its reduced weight and ergonomic shape.
The multiplanar variable-view rigid endoscope has proven to be useful for working on diverse anatomic structures such as intracranial vessels and cranial nerves. The device is effective in narrow working spaces where even small movements can jeopardize the delicate surrounding structures. The multiplanar variable-viewing mechanism in a compact device offers advantages in terms of safety and ergonomics. Improving the usability will probably optimize the applicability of those endoscopic devices in neurosurgery. A major drawback of the current prototype is poor image resolution, which will probably soon be overcome with the ongoing progress in electronic microchip technology.
The addition of laser technology to endoscopic techniques offers a huge potential to neurosurgery but has achieved little acceptance to date. The reasons include concern regarding heat production, uncontrollable and distant penetration, and tissue interaction. Experiences with a 2-micron continuous- wave laser (RevoLix Jr, LISA Laser Products, Katlenburg-Lindau, Germany) for neuroendoscopic intraventricular procedures proved this laser to be a valuable and useful tool with safe applicability for endoscopic intracranial procedures in patients of all ages.34
Parallel to the launch of video screens for other uses with higher image definition, the image quality on the 2D endoscope cameras has been constantly improving over the last years. At the same time, the introduction of modern 3D endoscopic monitors is promising. However, 3D endoscopes have some disadvantages compared with the 2D endoscopes. First, the smallest 3D endoscopes are 4 mm in diameter, compared with 2.7 mm for 2D endoscopes. Moreover, the field of view with the 3D endoscope is less than half of that with conventional 2D endoscopes.34 When working in and around a region with critical neurovascular structures in close proximity, this loss of field of view can result in an increase in iatrogenic injury from the endoscope. In addition, 3D endoscopes require special glasses, generating a potential obstacle to the seamless integration of visual information from the microscope and endoscope. Finally, some surgeons experience vertigo when looking at the 3D picture through the glasses, which limits its universal applicability.
CONCLUSIONS
Using the endoscope and microscope as complementary and not competing tools allows surgeons to benefit from both technologies at the same time.35,36 The intraoperative combination of these 2 powerful visualization tools expands the effectiveness of microsurgical procedures and has the potential to further improve surgical results and reduce surgical risks. With endoscope-assisted microsurgery, visualization is often far superior to surgical maneuverability.
Endoscopic neurosurgery will likely be influenced by further innovations in optical physics, electronics, and robotics. Specific implementations in endoscopic systems are likely to pave the way for remarkable progress in minimally invasive surgery, such as robotic surgical technology, further miniaturization of devices, improvements in 3D endoscopy, multiport endoscopy, and new designs for surgical instruments. Future progress in flexible endoscopes and wireless capsule or camera technology may reduce our dependence on rigid rod lens systems. Rigid variable-view endoscopes will bring endoscopes closer to ideal attributes utilizing newer instrumentation that is tailored to specific indications and techniques.37,38 Extension of the visual field by the feature of a movable optic lens may allow the neurosurgeon to use tailored keyhole approaches to treat pathologies in smaller surgical corridors with less trauma and greater efficacy.
- Kassam AB, Gardner P, Snyderman C, Mintz A, Carrau R. Expanded endonasal approach: fully endoscopic, completely transnasal approach to the middle third of the clivus, petrous bone, middle cranial fossa, and infratemporal fossa. Neurosurg Focus 2005; 19(1):E6. pmid:16078820
- Hopf NJ, Perneczky A. Endoscopic neurosurgery and endoscope-assisted microneurosurgery for the treatment of intracranial cysts. Neurosurgery 1998; 43(6):1330–1336. doi:10.1097/00006123-199812000-00037
- Li KW, Nelson C, Suk I, Jallo GI. Neuroendoscopy: past, present, and future. Neurosurg Focus 2005; 19(6):E1. doi:10.3171/foc.2005.19.6.2
- Prevedello DM, Doglietto F, Jane JA Jr, Jagannathan J, Han J, Laws ER Jr. History of endoscopic skull base surgery: its evolution and current reality. J Neurosurg 2007; 107(1):206–213. doi:10.3171/JNS-07/07/0206
- Schroeder HW, Nehlsen M. Value of high-definition imaging in neuroendoscopy. Neurosurg Rev 2009; 32(3):303–308. doi:10.1007/s10143-009-0200-x
- Chaynes P, Deguine O, Moscovici J, Fraysse B, Becue J, Lazorthes Y. Endoscopic anatomy of the cerebellopontine angle: a study in cadaver brains. Neurosurg Focus 1998; 5(3):e8.
- Setty P, Volkov AA, D'Andrea KP, Pieper DR. Endoscopic vascular decompression for the treatment of trigeminal neuralgia: clinical outcomes and technical note. World Neurosurg 2014; 81(3–4):603–608. doi:10.1016/j.wneu.2013.10.036
- Okutsu I, Hamanaka I, Yoshida A. Retrospective analysis of five-year and longer clinical and electrophysiological results of the world's first endoscopic management for carpal tunnel syndrome. Hand Surg 2013; 18(3):317–323. doi:10.1142/S0218810413500330
- Zuo D, Zhou Z, Wang H, et al. Endoscopic versus open carpal tunnel release for idiopathic carpal tunnel syndrome: a meta-analysis of randomized controlled trials. J Orthop Surg Res 2015; 10:12. doi:10.1186/s13018-014-0148-6
- Forst R, Hausmann B. Nucleoscopy—a new examination technique. Arch Orthop Trauma Surg 1983; 101(3):219–221. pmid:6870510
- Brayda-Bruno M, Cinnella P. Posterior endoscopic discectomy (and other procedures). Eur Spine J 2000; 9(suppl 1):S24–S29. pmid:10766054
- Destandau J. A special device for endoscopic surgery of lumbar disc herniation. Neurol Res 1999; 21(1):39–42. pmid:10048052
- Perez-Cruet MJ, Foley KT, Isaacs RE, et al. Microendoscopic lumbar discectomy: technical note. Neurosurgery 2002; 51(5 suppl):S129–S136. pmid:12234440
- Ruetten S, Komp M, Merk H, Godolias G. Full-endoscopic interlaminar and transforaminal lumbar discectomy versus conventional microsurgical technique: a prospective, randomized, controlled study. Spine (Phila Pa 1976) 2008; 33(9):931–939. doi:10.1097/BRS.0b013e31816c8af7
- Komp M, Hahn P, Merk H, Godolias G, Ruetten S. Bilateral operation of lumbar degenerative central spinal stenosis in full-endoscopic interlaminar technique with unilateral approach: prospective 2-year results of 74 patients. J Spinal Disord Tech 2011; 24(5):281–287. doi:10.1097/BSD.0b013e3181f9f55e
- Goyal A, Lu VM, Yolcu YU, Elminawy M, Daniels DJ. Endoscopic versus open approach in craniosynostosis repair: a systematic review and meta-analysis of perioperative outcomes. Childs Nerv Syst 2018; 34(9):1627–1637. doi:10.1007/s00381-018-3852-4
- Kassam AB, Gardner P, Snyderman C, Mintz A, Carrau R. Expanded endonasal approach: fully endoscopic, completely transnasal approach to the middle third of the clivus, petrous bone, middle cranial fossa, and infratemporal fossa. Neurosurg Focus 2005; 19(1):E6. pmid:16078820
- Kassam A, Snyderman CH, Mintz A, Gardner P, Carrau RL. Expanded endonasal approach: the rostrocaudal axis. Part II. Posterior clinoids to the foramen magnum. Neurosurg Focus 2005; 19(1):E4. pmid:16078818
- Kassam A, Snyderman CH, Mintz A, Gardner P, Carrau RL. Expanded endonasal approach: the rostrocaudal axis. Part I. Crista galli to the sella turcica. Neurosurg Focus 2005; 19(1):E3. pmid:16078817
- Kassam A, Carrau RL, Snyderman CH, Gardner P, Mintz A. Evolution of reconstructive techniques following endoscopic expanded endonasal approaches. Neurosurg Focus 2005; 19(1):E8. pmid:16078822
- Kassam AB, Thomas A, Carrau RL, et al. Endoscopic reconstruction of the cranial base using a pedicled nasoseptal flap. Neurosurgery 2008; 63(1 suppl 1):ONS44–ONS52. doi:10.1227/01.NEU.0000297074.13423.F5
- Hadad G, Bassagasteguy L, Carrau RL, et al. A novel reconstructive technique after endoscopic expanded endonasal approaches: vascular pedicle nasoseptal flap. Laryngoscope 2006; 116(10):1882–1886. doi:10.1097/01.mlg.0000234933.37779.e4
- Fortes FS, Carrau RL, Snyderman CH, et al. Transpterygoid transposition of a temporoparietal fascia flap: a new method for skull base reconstruction after endoscopic expanded endonasal approaches. Laryngoscope 2007; 117(6):970–976. doi:10.1097/MLG.0b013e3180471482
- Carrau RL, Snyderman CH, Kassam AB. The management of cerebrospinal fluid leaks in patients at risk for high-pressure hydrocephalus. Laryngoscope 2005; 115(2):205–212. doi:10.1097/01.mlg.0000154719.62668.70
- Zweig JL, Carrau RL, Celin SE, et al. Endoscopic repair of cerebrospinal fluid leaks to the sinonasal tract: predictors of success. Otolaryngol Head Neck Surg 2000; 123(3):195–201. doi:10.1067/mhn.2000.107452
- Kulkarni AV, Riva-Cambrin J, Holubkov R, et al. Endoscopic third ventriculostomy in children: prospective, multicenter results from the Hydrocephalus Clinical Research Network. J Neurosurg Pediatr 2016; 18(4):423–429. doi:10.3171/2016.4.PEDS163
- Ersahin Y, Arslan D. Complications of endoscopic third ventriculostomy. Childs Nerv Syst 2008; 24(8):943–948. doi:10.1007/s00381-008-0589-5
- Martínez-Catasús A, Lobo-Escolar L, García-Bonet J, Corrales-Rodríguez M, Pasarín-Martínez A, Berlanga-de-Mingo D. Comparison between single portal endoscopic, 1-cm open carpal tunnel release. Hand Surg Rehabil 2019. pii:S2468-1229(19)30027-1. doi:10.1016/j.hansur.2019.02.003
- Steers J, Reulen HJ, Lindsay K; European Union of Medical Specialists; Joint Residency Advisory and Accreditation Committee. UEMS charter on training of medical specialists in the EU—the new neurosurgical training charter. Acta Neurochir Suppl 2004; 90:3–11. pmid:15553111
- Mori H, Nishiyama K, Yoshimura J, Tanaka R. Current status of neuroendoscopic surgery in Japan and discussion on the training system. Childs Nerv Syst 2007; 23(6):673–676. doi:10.1007/s00381-007-0329-2
- Aryan HE, Hoeg HD, Marshall LF, Levy ML. Multidirectional projectional rigid neuro-endoscopy: prototype and initial experience. Minim Invasive Neurosurg 2005; 48(5):293–296. doi:10.1055/s-2005-915602
- Ebner FH, Marquardt JS, Hirt B, Tatagiba M, Schuhmann MU. Visualization of the anterior cerebral artery complex with a continuously variable-view rigid endoscope: new options in aneurysm surgery. Neurosurgery 2010; 67(2 suppl operative):321–324. doi:10.1227/NEU.0b013e3181f74548
- Ebner FH, Hirt B, Marquardt JS, Herlan S, Tatagiba M, Schuhmann MU. Actual state of EndActive ventricular endoscopy. Childs Nerv Syst 2012; 28(1):87–91. doi:10.1007/s00381-011-1537-3
- Ebner FH, Nagel C, Tatagiba M, Schuhmann MU. Efficacy and versatility of the 2-micron continuous wave laser in neuroendoscopic procedures. Acta Neurochir Suppl 2012; 113:143–147. doi:10.1007/978-3-7091-0923-6_29
- Van Gompel JJ, Tabor MH, Youssef AS, et al. Field of view comparison between two-dimensional and three-dimensional endoscopy. Laryngoscope 2014; 124(2):387–390. doi:10.1002/lary.24222
- Ebner FH, Roser F, Thaher F, Schittenhelm J, Tatagiba M. Balancing the shortcomings of microscope and endoscope: endoscope-assisted technique in microsurgical removal of recurrent epidermoid cysts in the posterior fossa. Minim Invasive Neurosurg 2010 ;53(5–6):218–222. doi:10.1055/s-0030-1267973
- Perneczky A, Fries G. Endoscope-assisted brain surgery: part 1—evolution, basic concept, and current technique. Neurosurgery 1998; 42(2):219–224. doi:10.1097/00006123-199802000-00001
- Ebner FH, Marquardt JS, Hirt B, Feigl GC, Tatagiba M, Schuhmann MU. Broadening horizons of neuroendoscopy with a variable-view rigid endoscope: an anatomical study. Eur J Surg Oncol 2010; 36(2):195–200. doi:10.1016/j.ejso.2009.07.185
- Kassam AB, Gardner P, Snyderman C, Mintz A, Carrau R. Expanded endonasal approach: fully endoscopic, completely transnasal approach to the middle third of the clivus, petrous bone, middle cranial fossa, and infratemporal fossa. Neurosurg Focus 2005; 19(1):E6. pmid:16078820
- Hopf NJ, Perneczky A. Endoscopic neurosurgery and endoscope-assisted microneurosurgery for the treatment of intracranial cysts. Neurosurgery 1998; 43(6):1330–1336. doi:10.1097/00006123-199812000-00037
- Li KW, Nelson C, Suk I, Jallo GI. Neuroendoscopy: past, present, and future. Neurosurg Focus 2005; 19(6):E1. doi:10.3171/foc.2005.19.6.2
- Prevedello DM, Doglietto F, Jane JA Jr, Jagannathan J, Han J, Laws ER Jr. History of endoscopic skull base surgery: its evolution and current reality. J Neurosurg 2007; 107(1):206–213. doi:10.3171/JNS-07/07/0206
- Schroeder HW, Nehlsen M. Value of high-definition imaging in neuroendoscopy. Neurosurg Rev 2009; 32(3):303–308. doi:10.1007/s10143-009-0200-x
- Chaynes P, Deguine O, Moscovici J, Fraysse B, Becue J, Lazorthes Y. Endoscopic anatomy of the cerebellopontine angle: a study in cadaver brains. Neurosurg Focus 1998; 5(3):e8.
- Setty P, Volkov AA, D'Andrea KP, Pieper DR. Endoscopic vascular decompression for the treatment of trigeminal neuralgia: clinical outcomes and technical note. World Neurosurg 2014; 81(3–4):603–608. doi:10.1016/j.wneu.2013.10.036
- Okutsu I, Hamanaka I, Yoshida A. Retrospective analysis of five-year and longer clinical and electrophysiological results of the world's first endoscopic management for carpal tunnel syndrome. Hand Surg 2013; 18(3):317–323. doi:10.1142/S0218810413500330
- Zuo D, Zhou Z, Wang H, et al. Endoscopic versus open carpal tunnel release for idiopathic carpal tunnel syndrome: a meta-analysis of randomized controlled trials. J Orthop Surg Res 2015; 10:12. doi:10.1186/s13018-014-0148-6
- Forst R, Hausmann B. Nucleoscopy—a new examination technique. Arch Orthop Trauma Surg 1983; 101(3):219–221. pmid:6870510
- Brayda-Bruno M, Cinnella P. Posterior endoscopic discectomy (and other procedures). Eur Spine J 2000; 9(suppl 1):S24–S29. pmid:10766054
- Destandau J. A special device for endoscopic surgery of lumbar disc herniation. Neurol Res 1999; 21(1):39–42. pmid:10048052
- Perez-Cruet MJ, Foley KT, Isaacs RE, et al. Microendoscopic lumbar discectomy: technical note. Neurosurgery 2002; 51(5 suppl):S129–S136. pmid:12234440
- Ruetten S, Komp M, Merk H, Godolias G. Full-endoscopic interlaminar and transforaminal lumbar discectomy versus conventional microsurgical technique: a prospective, randomized, controlled study. Spine (Phila Pa 1976) 2008; 33(9):931–939. doi:10.1097/BRS.0b013e31816c8af7
- Komp M, Hahn P, Merk H, Godolias G, Ruetten S. Bilateral operation of lumbar degenerative central spinal stenosis in full-endoscopic interlaminar technique with unilateral approach: prospective 2-year results of 74 patients. J Spinal Disord Tech 2011; 24(5):281–287. doi:10.1097/BSD.0b013e3181f9f55e
- Goyal A, Lu VM, Yolcu YU, Elminawy M, Daniels DJ. Endoscopic versus open approach in craniosynostosis repair: a systematic review and meta-analysis of perioperative outcomes. Childs Nerv Syst 2018; 34(9):1627–1637. doi:10.1007/s00381-018-3852-4
- Kassam AB, Gardner P, Snyderman C, Mintz A, Carrau R. Expanded endonasal approach: fully endoscopic, completely transnasal approach to the middle third of the clivus, petrous bone, middle cranial fossa, and infratemporal fossa. Neurosurg Focus 2005; 19(1):E6. pmid:16078820
- Kassam A, Snyderman CH, Mintz A, Gardner P, Carrau RL. Expanded endonasal approach: the rostrocaudal axis. Part II. Posterior clinoids to the foramen magnum. Neurosurg Focus 2005; 19(1):E4. pmid:16078818
- Kassam A, Snyderman CH, Mintz A, Gardner P, Carrau RL. Expanded endonasal approach: the rostrocaudal axis. Part I. Crista galli to the sella turcica. Neurosurg Focus 2005; 19(1):E3. pmid:16078817
- Kassam A, Carrau RL, Snyderman CH, Gardner P, Mintz A. Evolution of reconstructive techniques following endoscopic expanded endonasal approaches. Neurosurg Focus 2005; 19(1):E8. pmid:16078822
- Kassam AB, Thomas A, Carrau RL, et al. Endoscopic reconstruction of the cranial base using a pedicled nasoseptal flap. Neurosurgery 2008; 63(1 suppl 1):ONS44–ONS52. doi:10.1227/01.NEU.0000297074.13423.F5
- Hadad G, Bassagasteguy L, Carrau RL, et al. A novel reconstructive technique after endoscopic expanded endonasal approaches: vascular pedicle nasoseptal flap. Laryngoscope 2006; 116(10):1882–1886. doi:10.1097/01.mlg.0000234933.37779.e4
- Fortes FS, Carrau RL, Snyderman CH, et al. Transpterygoid transposition of a temporoparietal fascia flap: a new method for skull base reconstruction after endoscopic expanded endonasal approaches. Laryngoscope 2007; 117(6):970–976. doi:10.1097/MLG.0b013e3180471482
- Carrau RL, Snyderman CH, Kassam AB. The management of cerebrospinal fluid leaks in patients at risk for high-pressure hydrocephalus. Laryngoscope 2005; 115(2):205–212. doi:10.1097/01.mlg.0000154719.62668.70
- Zweig JL, Carrau RL, Celin SE, et al. Endoscopic repair of cerebrospinal fluid leaks to the sinonasal tract: predictors of success. Otolaryngol Head Neck Surg 2000; 123(3):195–201. doi:10.1067/mhn.2000.107452
- Kulkarni AV, Riva-Cambrin J, Holubkov R, et al. Endoscopic third ventriculostomy in children: prospective, multicenter results from the Hydrocephalus Clinical Research Network. J Neurosurg Pediatr 2016; 18(4):423–429. doi:10.3171/2016.4.PEDS163
- Ersahin Y, Arslan D. Complications of endoscopic third ventriculostomy. Childs Nerv Syst 2008; 24(8):943–948. doi:10.1007/s00381-008-0589-5
- Martínez-Catasús A, Lobo-Escolar L, García-Bonet J, Corrales-Rodríguez M, Pasarín-Martínez A, Berlanga-de-Mingo D. Comparison between single portal endoscopic, 1-cm open carpal tunnel release. Hand Surg Rehabil 2019. pii:S2468-1229(19)30027-1. doi:10.1016/j.hansur.2019.02.003
- Steers J, Reulen HJ, Lindsay K; European Union of Medical Specialists; Joint Residency Advisory and Accreditation Committee. UEMS charter on training of medical specialists in the EU—the new neurosurgical training charter. Acta Neurochir Suppl 2004; 90:3–11. pmid:15553111
- Mori H, Nishiyama K, Yoshimura J, Tanaka R. Current status of neuroendoscopic surgery in Japan and discussion on the training system. Childs Nerv Syst 2007; 23(6):673–676. doi:10.1007/s00381-007-0329-2
- Aryan HE, Hoeg HD, Marshall LF, Levy ML. Multidirectional projectional rigid neuro-endoscopy: prototype and initial experience. Minim Invasive Neurosurg 2005; 48(5):293–296. doi:10.1055/s-2005-915602
- Ebner FH, Marquardt JS, Hirt B, Tatagiba M, Schuhmann MU. Visualization of the anterior cerebral artery complex with a continuously variable-view rigid endoscope: new options in aneurysm surgery. Neurosurgery 2010; 67(2 suppl operative):321–324. doi:10.1227/NEU.0b013e3181f74548
- Ebner FH, Hirt B, Marquardt JS, Herlan S, Tatagiba M, Schuhmann MU. Actual state of EndActive ventricular endoscopy. Childs Nerv Syst 2012; 28(1):87–91. doi:10.1007/s00381-011-1537-3
- Ebner FH, Nagel C, Tatagiba M, Schuhmann MU. Efficacy and versatility of the 2-micron continuous wave laser in neuroendoscopic procedures. Acta Neurochir Suppl 2012; 113:143–147. doi:10.1007/978-3-7091-0923-6_29
- Van Gompel JJ, Tabor MH, Youssef AS, et al. Field of view comparison between two-dimensional and three-dimensional endoscopy. Laryngoscope 2014; 124(2):387–390. doi:10.1002/lary.24222
- Ebner FH, Roser F, Thaher F, Schittenhelm J, Tatagiba M. Balancing the shortcomings of microscope and endoscope: endoscope-assisted technique in microsurgical removal of recurrent epidermoid cysts in the posterior fossa. Minim Invasive Neurosurg 2010 ;53(5–6):218–222. doi:10.1055/s-0030-1267973
- Perneczky A, Fries G. Endoscope-assisted brain surgery: part 1—evolution, basic concept, and current technique. Neurosurgery 1998; 42(2):219–224. doi:10.1097/00006123-199802000-00001
- Ebner FH, Marquardt JS, Hirt B, Feigl GC, Tatagiba M, Schuhmann MU. Broadening horizons of neuroendoscopy with a variable-view rigid endoscope: an anatomical study. Eur J Surg Oncol 2010; 36(2):195–200. doi:10.1016/j.ejso.2009.07.185
meningioma, peripheral nerve, spinal canal, minimally invasive, carpal tunnel, ventricular neuroendoscopy, craniosynostosis, degenerative spine disease, Luigi Rigante, Hamid Borghei-Razavi, Pablo Recinos, Florian Roser
meningioma, peripheral nerve, spinal canal, minimally invasive, carpal tunnel, ventricular neuroendoscopy, craniosynostosis, degenerative spine disease, Luigi Rigante, Hamid Borghei-Razavi, Pablo Recinos, Florian Roser
KEY POINTS
- An increasing number of neurosurgical patients are undergoing endoscopic surgeries of the brain, spine, and peripheral nerves. Familiarization with these techniques provides medical specialists with important knowledge regarding appropriate patient care.
- The combination of classic microscopic and endoscopic procedures improves surgical outcomes by increasing surgical maneuverability and reducing manipulation of eloquent structures.
- Further innovations in optical physics, electronics, and robotics will dramatically improve the potential of endoscopic neurosurgery in the next decades.
A few pearls can help prepare the mind
We need to recognize the diverse problems that patients with potential multisystem disease can develop, lobby when necessary for them to be seen promptly by the relevant specialists, and initiate appropriate diagnostic testing and management in less-urgent scenarios. Most of us need frequent refreshers on the clinical manifestations of these disorders so that we can recognize them when they appear unannounced in our exam rooms.
The caregiver with a prepared mind is more likely to experience the diagnostic epiphany, and then use point-of-care references to hone in on the details. With many patients and clinical conundrums, the basics matter.
Dr. Chester Oddis, in this issue of the Journal, reviews the basics of several primary muscle disorders. He discusses, in a case-based format extracted from his recent Medicine Grand Rounds presentation at Cleveland Clinic, nuances of specific diagnoses and the clinical progression of diseases that are critical to be aware of in order to recognize and manage them, and expeditiously refer the patient to our appropriate subspecialty colleagues.
Major challenges exist in recognizing the inflammatory myopathies and their mimics early in their course. These are serious but uncommon entities, and in part because patients and physicians often attribute their early symptoms to more-common causes, diagnosis can be elusive—until the possibility is considered. We hope that Dr. Oddis’s article will make it easier to rapidly recognize these muscle disorders.
Patients often struggle to explain their symptoms of early muscle dysfunction. Since patients often verbalize their fatigue as “feeling weak,” we often misconstrue complaints of true muscle weakness (like difficulty walking up steps) as being due to fatigue. Add in some anemia from chronic inflammation and some “liver test” abnormalities, and it is easy to see how the recognition of true muscle weakness can be delayed.
We can tease muscle weakness from fatigue or dyspnea by asking the patient to specifically and functionally describe their “weakness,” and then by asking pointed questions: “Do you have difficulty getting up from the toilet without using your arms? Do you have trouble brushing your hair or teeth?” Physical examination can clearly help here, but without routine examination of muscle strength in normal fragile elderly patients, the degree of muscle weakness can be difficult to assess. Likewise challenging is detecting the early onset of weakness by examination in a 280-lb power-lifter.
Obtaining an accurate functional and behavioral history is often critical to the early recognition of muscle disease. Muscle pain, as Dr. Oddis notes, is not a characteristic feature of many myopathies, whereas, paradoxically, the coexistence of new-onset symmetrical small-joint pain (especially with arthritis) along with muscle weakness can be a powerful clue to the diagnosis of an inflammatory myopathy.
An elevated creatine kinase (CK) level generally points directly to a muscle disease, although some neurologic disorders are associated with elevations in CK, and the entity of benign “hyperCKemia” must be recognized and not overmanaged. The latter becomes a problem when laboratory tests are allowed to drive the diagnostic evaluation in a vacuum of clinical details.
A more common scenario is the misinterpretation of common laboratory test abnormalities in the setting of a patient with “fatigue” or generalized weakness who has elevations in aspartate aminotransferase (AST) and alanine aminotransferase (ALT). Although AST and ALT are often called “liver function tests,” these enzymes are also abundant in skeletal muscle, and since they are included on routine biochemical panels, their elevation often leads to liver imaging and sometimes even biopsy before anyone recognizes muscle disease as the cause of the patient’s symptoms and laboratory test abnormalities. Hence, a muscle source (or hemolysis) should at least be considered when AST and ALT are elevated in the absence of elevated alkaline phosphatase or gamma-glutamyl transferase.
When evaluating innumerable clinical scenarios, experienced clinicians can most certainly generate similar principles of diagnostic reasoning, based on having a few fundamental facts at their fingertips. Increasing the chances of having a prepared mind when confronted with a patient with a less-than-straightforward set of symptoms is one of my major arguments in support of continuing to read and generate internal medicine teaching literature and to attend and participate in clinical teaching conferences such as Medicine Grand Rounds. It is also why we will continue to appreciate and publish presentations like this one in the Journal.
I don’t expect to retain all the details from these and similar papers, and I know we all carry virtually infinite databases in our pockets. But keeping a few clinical pearls outside of my specialty in my head comes in handy. Having a prepared mind makes it much easier to converse with patients, to promptly initiate appropriate testing, plans, and consultations, and to then decide what to search for on my smartphone between patients.
We need to recognize the diverse problems that patients with potential multisystem disease can develop, lobby when necessary for them to be seen promptly by the relevant specialists, and initiate appropriate diagnostic testing and management in less-urgent scenarios. Most of us need frequent refreshers on the clinical manifestations of these disorders so that we can recognize them when they appear unannounced in our exam rooms.
The caregiver with a prepared mind is more likely to experience the diagnostic epiphany, and then use point-of-care references to hone in on the details. With many patients and clinical conundrums, the basics matter.
Dr. Chester Oddis, in this issue of the Journal, reviews the basics of several primary muscle disorders. He discusses, in a case-based format extracted from his recent Medicine Grand Rounds presentation at Cleveland Clinic, nuances of specific diagnoses and the clinical progression of diseases that are critical to be aware of in order to recognize and manage them, and expeditiously refer the patient to our appropriate subspecialty colleagues.
Major challenges exist in recognizing the inflammatory myopathies and their mimics early in their course. These are serious but uncommon entities, and in part because patients and physicians often attribute their early symptoms to more-common causes, diagnosis can be elusive—until the possibility is considered. We hope that Dr. Oddis’s article will make it easier to rapidly recognize these muscle disorders.
Patients often struggle to explain their symptoms of early muscle dysfunction. Since patients often verbalize their fatigue as “feeling weak,” we often misconstrue complaints of true muscle weakness (like difficulty walking up steps) as being due to fatigue. Add in some anemia from chronic inflammation and some “liver test” abnormalities, and it is easy to see how the recognition of true muscle weakness can be delayed.
We can tease muscle weakness from fatigue or dyspnea by asking the patient to specifically and functionally describe their “weakness,” and then by asking pointed questions: “Do you have difficulty getting up from the toilet without using your arms? Do you have trouble brushing your hair or teeth?” Physical examination can clearly help here, but without routine examination of muscle strength in normal fragile elderly patients, the degree of muscle weakness can be difficult to assess. Likewise challenging is detecting the early onset of weakness by examination in a 280-lb power-lifter.
Obtaining an accurate functional and behavioral history is often critical to the early recognition of muscle disease. Muscle pain, as Dr. Oddis notes, is not a characteristic feature of many myopathies, whereas, paradoxically, the coexistence of new-onset symmetrical small-joint pain (especially with arthritis) along with muscle weakness can be a powerful clue to the diagnosis of an inflammatory myopathy.
An elevated creatine kinase (CK) level generally points directly to a muscle disease, although some neurologic disorders are associated with elevations in CK, and the entity of benign “hyperCKemia” must be recognized and not overmanaged. The latter becomes a problem when laboratory tests are allowed to drive the diagnostic evaluation in a vacuum of clinical details.
A more common scenario is the misinterpretation of common laboratory test abnormalities in the setting of a patient with “fatigue” or generalized weakness who has elevations in aspartate aminotransferase (AST) and alanine aminotransferase (ALT). Although AST and ALT are often called “liver function tests,” these enzymes are also abundant in skeletal muscle, and since they are included on routine biochemical panels, their elevation often leads to liver imaging and sometimes even biopsy before anyone recognizes muscle disease as the cause of the patient’s symptoms and laboratory test abnormalities. Hence, a muscle source (or hemolysis) should at least be considered when AST and ALT are elevated in the absence of elevated alkaline phosphatase or gamma-glutamyl transferase.
When evaluating innumerable clinical scenarios, experienced clinicians can most certainly generate similar principles of diagnostic reasoning, based on having a few fundamental facts at their fingertips. Increasing the chances of having a prepared mind when confronted with a patient with a less-than-straightforward set of symptoms is one of my major arguments in support of continuing to read and generate internal medicine teaching literature and to attend and participate in clinical teaching conferences such as Medicine Grand Rounds. It is also why we will continue to appreciate and publish presentations like this one in the Journal.
I don’t expect to retain all the details from these and similar papers, and I know we all carry virtually infinite databases in our pockets. But keeping a few clinical pearls outside of my specialty in my head comes in handy. Having a prepared mind makes it much easier to converse with patients, to promptly initiate appropriate testing, plans, and consultations, and to then decide what to search for on my smartphone between patients.
We need to recognize the diverse problems that patients with potential multisystem disease can develop, lobby when necessary for them to be seen promptly by the relevant specialists, and initiate appropriate diagnostic testing and management in less-urgent scenarios. Most of us need frequent refreshers on the clinical manifestations of these disorders so that we can recognize them when they appear unannounced in our exam rooms.
The caregiver with a prepared mind is more likely to experience the diagnostic epiphany, and then use point-of-care references to hone in on the details. With many patients and clinical conundrums, the basics matter.
Dr. Chester Oddis, in this issue of the Journal, reviews the basics of several primary muscle disorders. He discusses, in a case-based format extracted from his recent Medicine Grand Rounds presentation at Cleveland Clinic, nuances of specific diagnoses and the clinical progression of diseases that are critical to be aware of in order to recognize and manage them, and expeditiously refer the patient to our appropriate subspecialty colleagues.
Major challenges exist in recognizing the inflammatory myopathies and their mimics early in their course. These are serious but uncommon entities, and in part because patients and physicians often attribute their early symptoms to more-common causes, diagnosis can be elusive—until the possibility is considered. We hope that Dr. Oddis’s article will make it easier to rapidly recognize these muscle disorders.
Patients often struggle to explain their symptoms of early muscle dysfunction. Since patients often verbalize their fatigue as “feeling weak,” we often misconstrue complaints of true muscle weakness (like difficulty walking up steps) as being due to fatigue. Add in some anemia from chronic inflammation and some “liver test” abnormalities, and it is easy to see how the recognition of true muscle weakness can be delayed.
We can tease muscle weakness from fatigue or dyspnea by asking the patient to specifically and functionally describe their “weakness,” and then by asking pointed questions: “Do you have difficulty getting up from the toilet without using your arms? Do you have trouble brushing your hair or teeth?” Physical examination can clearly help here, but without routine examination of muscle strength in normal fragile elderly patients, the degree of muscle weakness can be difficult to assess. Likewise challenging is detecting the early onset of weakness by examination in a 280-lb power-lifter.
Obtaining an accurate functional and behavioral history is often critical to the early recognition of muscle disease. Muscle pain, as Dr. Oddis notes, is not a characteristic feature of many myopathies, whereas, paradoxically, the coexistence of new-onset symmetrical small-joint pain (especially with arthritis) along with muscle weakness can be a powerful clue to the diagnosis of an inflammatory myopathy.
An elevated creatine kinase (CK) level generally points directly to a muscle disease, although some neurologic disorders are associated with elevations in CK, and the entity of benign “hyperCKemia” must be recognized and not overmanaged. The latter becomes a problem when laboratory tests are allowed to drive the diagnostic evaluation in a vacuum of clinical details.
A more common scenario is the misinterpretation of common laboratory test abnormalities in the setting of a patient with “fatigue” or generalized weakness who has elevations in aspartate aminotransferase (AST) and alanine aminotransferase (ALT). Although AST and ALT are often called “liver function tests,” these enzymes are also abundant in skeletal muscle, and since they are included on routine biochemical panels, their elevation often leads to liver imaging and sometimes even biopsy before anyone recognizes muscle disease as the cause of the patient’s symptoms and laboratory test abnormalities. Hence, a muscle source (or hemolysis) should at least be considered when AST and ALT are elevated in the absence of elevated alkaline phosphatase or gamma-glutamyl transferase.
When evaluating innumerable clinical scenarios, experienced clinicians can most certainly generate similar principles of diagnostic reasoning, based on having a few fundamental facts at their fingertips. Increasing the chances of having a prepared mind when confronted with a patient with a less-than-straightforward set of symptoms is one of my major arguments in support of continuing to read and generate internal medicine teaching literature and to attend and participate in clinical teaching conferences such as Medicine Grand Rounds. It is also why we will continue to appreciate and publish presentations like this one in the Journal.
I don’t expect to retain all the details from these and similar papers, and I know we all carry virtually infinite databases in our pockets. But keeping a few clinical pearls outside of my specialty in my head comes in handy. Having a prepared mind makes it much easier to converse with patients, to promptly initiate appropriate testing, plans, and consultations, and to then decide what to search for on my smartphone between patients.
Myopathy for the general internist: Statins and much more
Myopathies can present with a wide variety of symptoms, so patients with muscle weakness are often seen initially by a general practitioner. Nonrheumatologists should be able to evaluate a patient presenting with muscle weakness or myalgia and be aware of red flags indicating potentially dangerous syndromes that require a prompt, thorough investigation.
This article reviews selected causes of muscle weakness, such as statin-induced and autoimmune disorders, and systemic features of inflammatory myopathies beyond myositis, such as dermatologic and pulmonary manifestations.
FOCUSING THE EVALUATION
The evaluation of a patient presenting with muscle weakness should include several assessments:
Temporal progression. Was the onset of symptoms rapid or insidious? Patterns of onset may give clues to etiology, including the possibility of an associated autoimmune condition.
Location of muscle weakness. Are symptoms global or localized? And if localized, are they proximal or distal? Proximal weakness can be manifested by difficulty rising from a chair (hip muscles) or combing one’s hair (shoulder muscles), whereas distal weakness can involve difficulty standing on toes (gastrocnemius and soleus muscles) or performing fine motor activities (intrinsic hand muscles).
Symmetry. A focal or asymmetric pattern often has a neurologic etiology, but this could also be consistent with inclusion body myositis.
Other symptoms. Arthritis, rash, and swallowing problems point to a possible underlying rheumatologic disease. Weight gain or loss may indicate a thyroid disorder.
Family history. Some patients report that others in their family have this pattern of weakness, indicating a likely genetic myopathy. If the patient reports a relative with multiple sclerosis, lupus erythematosus, rheumatoid arthritis, or another autoimmune disease, then an immune-mediated myopathy should be considered.
Medications should be reviewed, particularly statins.
CASE 1: SLOWLY PROGRESSIVE WEAKNESS
A 65-year-old man presented with the insidious onset of muscle weakness and episodes of falling. On review of his medical record, his serum creatine kinase (CK) levels were elevated at various periods at 2 to 4 times the upper limit of normal. Electromyography (EMG) previously showed a myopathic pattern, and a muscle biopsy was abnormal, consistent with endomysial inflammation (term is consistent with “polymyositis”). He was treated for polymyositis for several years with prednisone alone, with steroids plus methotrexate, and with combined immunosuppression including methotrexate and azathioprine, but with no improvement. Eventually, another muscle biopsy revealed inclusion bodies with rimmed vacuoles, consistent with inclusion body myositis.
Inclusion body myositis
Inclusion body myositis is the most common myopathy in middle-aged to elderly people, especially men. These patients are often told “You are just getting old,” but they have a defined condition. It should also be considered in patients failing to respond to treatment or with those with “refractory” polymyositis.
The onset of muscle weakness is insidious and painless, and the weakness progresses slowly. The pattern is distal and asymmetric (eg, foot drop), and muscle atrophy typically affects the forearm flexors, quadriceps, and intrinsic muscles of the hands.1
Magnetic resonance imaging may show marked muscle atrophy. Unfortunately, no treatment has shown efficacy, and most neuromuscular and rheumatology experts do not treat inclusion body myositis with immunosuppressive drugs.
CASE 2: MILD MYALGIA WITHOUT WEAKNESS
A black 52-year-old man was referred because of myalgia and a CK level of 862 U/L (reference range < 200). His physician wanted to start him on a statin but was hesitant to do so without first consulting a rheumatologist.
The patient had a long history of mild arthralgias and myalgias without muscle weakness. He had dyslipidemia and hypertension. He reported no family history of myopathy and no illicit drug use. He was formerly an athlete. Medications included a thiazide diuretic and a beta-blocker. On examination, his muscles were strong (rated 5 on a scale of 5) in the upper and lower extremities, without atrophy.
His records showed that his CK levels had risen and fallen repeatedly over the past few years, ranging from 600 to 1,100 U/L. On further questioning, he reported that when he had joined the army 30 years previously, a physician had recommended he undergo a liver biopsy in view of elevated liver function tests, but that he had refused because he felt fine.
Currently, his gamma-glutamyl transpeptidase levels were normal.
Idiopathic ‘hyperCKemia’
So-called idiopathic hyperCKemia is not a form of myositis but merely a laboratory result outside the “normal” range. Reference ranges are based predominantly on measurements in white people and on an assumption that the distribution is Gaussian (bell-shaped). A normal CK level is usually defined as less than 200 U/L. Using this standard, up to 20% of men and 5% of women have hyperCKemia.2
However, CK levels vary by sex and ethnicity, with mean levels highest in black men, followed by black women, white men, and white women. The mean level in black men is higher than the standard cutoff point for normal, and especially in this population, there is wide fluctuation around the mean, leading to hyperCKemia quite frequently in black men. Exercise and manual labor also drive up CK levels.3–5
Idiopathic hyperCKemia is benign. D’Adda et al6 followed 55 patients for a mean of 7.5 years. CK levels normalized in 12 patients or at least decreased in 24. Most remained symptom-free or had minimal symptoms.
Idiopathic hyperCKemia: Bottom line
Before prescribing a statin, determine the baseline CK level. If slightly elevated (ie, up to 3 to 5 times the upper limit of normal, or even higher) in the setting of normal muscle strength, there is no need for electromyography or muscle biopsy, and the patient can certainly receive a statin. Most of these patients do not need to see a rheumatologist but can simply have their CK and muscle strength monitored.
CLASSIFYING MYOSITIS
Myositis (idiopathic inflammatory myopathy) is a heterogeneous group of autoimmune syndromes of unknown cause characterized by chronic muscle weakness and inflammation of striated muscle. These syndromes likely arise as a result of genetic predisposition and an environmental or infectious “hit.”
Myositis is rare, with an incidence of 5 to 10 cases per million per year and an estimated prevalence of 50 to 90 cases per million. It has 2 incidence peaks: 1 in childhood (age 5–15) and another in adult midlife (age 30–50). Women are affected 2 to 3 times more often than men, with black women most commonly affected.
Myositis is traditionally classified as follows:
- Adult polymyositis
- Adult dermatomyositis
- Juvenile myositis (dermatomyositis much more frequent than polymyositis)
- Malignancy-associated myositis (usually dermatomyositis)
- Myositis overlapping with another autoimmune disease
- Inclusion body myositis.
However, polymyositis is less common than we originally thought, and the term necrotizing myopathy is now used in many patients, as noted in the case studies below. Further, myositis overlap syndromes are being increasingly diagnosed, likely related to the emergence of autoantibodies and clinical “syndromes” associated with these autoantibody subsets (discussed in cases below).
Dermatomyositis
Dermatomyositis is characterized by muscle weakness and a rash that can be obvious or subtle. Classic skin lesions are Gottron papules, which are raised, flat-topped red or purplish lesions over the knuckles, elbows, or knees.
Lesions may be confused with those of psoriasis. There can also be a V-neck rash over the anterior chest or upper back (“shawl sign”) or a rash over the lateral thigh (“holster sign”). A facial rash may occur, but unlike lupus, dermatomyositis does not spare the nasolabial area. However, the V-neck rash can be similar to that seen in lupus.
Dermatomyositis may cause muscle pain, perhaps related to muscle ischemia, whereas polymyositis and necrotizing myopathy are often painless. However, pain is also associated with fibromyalgia, which may be seen in many autoimmune conditions. It is important not to overtreat rheumatologic diseases with immunosuppression to try to control pain if the pain is actually caused by fibromyalgia.
Polymyositis mimics
Hypothyroid myopathy can present as classic polymyositis. The serum CK may be elevated, and there may be myalgias, muscle hypertrophy with stiffness, weakness, cramps, and even features of a proximal myopathy, and rhabdomyolysis. The electromyogram can be normal or myopathic. Results of muscle biopsy are often normal but may show focal necrosis and mild inflammatory infiltrates, thus mimicking that seen with inflammatory myopathy.7
Drug-induced or toxic myopathies can also mimic polymyositis. Statins are among the most commonly prescribed drugs in the United States, with more than 35 million people taking them. Statins are generally well tolerated but have a broad spectrum of toxicity, ranging from myalgias to life-threatening rhabdomyolysis. Myalgias lead to about 5% to 10% of patients refusing to take a statin or stopping it on their own.
Myalgias affect up to 20% of statin users in clinical practice.8,9 A small cross-sectional study10 of 1,000 patients in a primary care setting found that the risk of muscle complaints in statin users was 1.5 times higher than in nonstatin users, similar to findings in other studies.
My strategy for managing a patient with possible statin-induced myopathy is illustrated in Figure 1.
CASE 3: WEAKNESS, VERY HIGH CK ON A STATIN
In March 2010, a 67-year-old woman presented with muscle weakness. She had a history of hypertension, hyperlipidemia, and, more than 10 years previously, uterine cancer. In 2004, she was given atorvastatin for dyslipidemia. Four years later, she developed lower-extremity weakness, which her doctor attributed to normal aging. A year after that, she found it difficult to walk up steps and lift her arms overhead. In June 2009, she stopped taking the atorvastatin on her own, but the weakness did not improve.
In September 2009, she returned to her doctor, who found her CK level was 6,473 U/L but believed it to be an error, so the test was repeated, with a result of 9,375 U/L. She had no rash or joint involvement.
She was admitted to the hospital and underwent muscle biopsy, which showed myonecrosis with no inflammation or vasculitis. She was treated with prednisone 60 mg/day, and her elevated CK level and weakness improved.
Immune-mediated necrotizing myopathy associated with statins
The hallmark of necrotizing myopathy is myonecrosis without significant inflammation.12 This pattern contrasts with that of polymyositis, which is characterized by lymphocytic inflammation.
Although statins became available in the United States in 1987, immune-mediated necrotizing myopathy associated with statins was first described only in 2010. In that report, Grable-Esposito et al13 described 25 patients from 2 neuromuscular centers seen between 2000 and 2008 who had elevated CK and proximal weakness during or after statin use, both of which persisted despite stopping the statin. Patients improved with immunosuppressive agents but had a relapse when steroids were stopped or tapered, a pattern typical in autoimmune disease.
Autoantibody defines subgroup of necrotizing myopathy
Also in 2010, Christopher-Stine et al14 reported an antibody associated with necrotizing myopathy. Of 38 patients with the condition, 16 were found to have an abnormal “doublet” autoantibody recognizing 200- and 100-kDa proteins. All patients had weakness and a high CK level, and 63% had statin exposure before the weakness (this percentage increased to 83% in patients older than 50). All responded to immunosuppressive therapy, and many had a relapse when it was withdrawn.
Statins lower cholesterol by inhibiting 3-hydroxy-3-methylglutaryl-Co A reductase (HMGCR), and paradoxically, they also upregulate it. HMGCR has a molecular weight of 97 kDa. Mammen et al15 identified HMGCR as the 100-kDa target of the identified antibody and developed an enzyme-linked immunosorbent assay for it. Of 750 patients presenting to one center, only 45 (6%) had anti-HMGCR autoantibodies, but all 16 patients who had the abnormal doublet antibody tested positive for anti-HMGCR. Regenerating muscle cells express high levels of HMGCR, which may sustain the immune response after statins are discontinued.
Case 3 continued: Intravenous immunoglobulin brings improvement
In March 2010, when the 67-year-old patient presented to our myositis center, her CK level was 5,800 U/L, which increased as prednisone was tapered. She still felt weak. On examination, her muscle strength findings were deltoids 4+/5, neck flexors 4/5, and iliopsoas 3+/5. She was treated with methotrexate and azathioprine without benefit. She was next treated with intravenous immunoglobulin, and after 3 months, her strength normalized for the first time in years. Her CK level decreased but did not normalize. Testing showed that she was positive for anti-HMGCR autoantibody, as this test had become commercially available.
In 2015, Mammen and Tiniakou16 suggested using intravenous immunoglobulin as first-line therapy for statin-associated autoimmune necrotizing myopathy, based on experience at a single center with 3 patients who declined glucocorticoid treatment.
Necrotizing myopathy: Bottom line
Myositis overlap syndromes
Heterogeneity is the rule in myositis, and it can present with a wide variety of signs and symptoms as outlined in Table 2.
CASE 4: FEVER, NEW ‘RHEUMATOID ARTHRITIS,’ AND LUNG DISEASE
A 52-year-old woman with knee osteoarthritis saw her primary care physician in November 2013 for dyspnea and low-grade fever. The next month, she presented with polyarthritis, muscle weakness, and Raynaud phenomenon.
In January 2014, she developed acrocyanosis of her fingers. Examination revealed hyperkeratotic, cracked areas of her fingers. Her oxygen saturation by pulse oximetry was low. She was admitted to the hospital. Her doctor suspected new onset of rheumatoid arthritis, but blood tests revealed a negative antinuclear antibody, so an autoimmune condition was deemed unlikely. Her CK was mildly elevated at 350 U/L.
Because of her dyspnea, an open-lung biopsy was performed. High-resolution computed tomography (CT) revealed infiltrates and ground-glass opacities, leading to the diagnosis of nonspecific interstitial pneumonia. A rheumatologist was consulted and recommended pulse methylprednisolone, followed by prednisone 60 mg/day and mycophenolate mofetil. Testing for Jo-1 antibodies was positive.
Antisynthetase syndrome
The antisynthetase syndrome is a clinically heterogeneous condition that can occur with any or all of the following:
- Fever
- Myositis
- Arthritis (often misdiagnosed as rheumatoid arthritis)
- Raynaud phenomenon
- Mechanic’s hands (hyperkeratotic roughness with fissures on the lateral aspects of the fingers and finger pads)
- Interstitial lung disease.
The skin rashes and myositis may be subtle, making the presentation “lung-dominant,” and nonrheumatologists should be aware of this syndrome. Although in our patient the condition developed in a classic manner, with all of the aforementioned features of the antisynthetase syndrome, some patients will manifest one or a few of the features.
Clinically, patients with the Jo-1 antisynthetase syndrome often present differently than those with non-Jo-1 antisynthetase autoantibodies. When we compared 122 patients with Jo-1 vs 80 patients with a non-Jo-1 antisynthetase autoantibody, patients with Jo-1 antibodies were more likely to have initially received a diagnosis of myositis (83%), while myositis was the original diagnosis in only 17% of those possessing non-Jo-1 antisynthetase autoantibodies. In fact, many patients (approximately 50%) were diagnosed as having undifferentiated connective tissue disease or an overlap syndrome, and 13% had scleroderma as their first diagnosis.17
We also found that the survival rate was higher in patients with Jo-1 syndrome compared with patients with non-Jo-1 antisynthetase syndromes. We attributed the difference in survival rates to a delayed diagnosis in the non-Jo-1 group, perhaps due to their “nonclassic” presentations of the antisynthetase syndrome, delaying appropriate treatment. Patients received a diagnosis of Jo-1 antibody syndrome after a mean of 0.4 year (range 0.2–0.8), while those with a non-Jo-1 antisynthetase autoantibody had a delay in diagnosis of 1.0 year (range 0.4–5.1) (P < .01).17
In nearly half the cases in this cohort, pulmonary fibrosis was the cause of death, with primary pulmonary hypertension being the second leading cause (11%).
Antisynthetase syndrome: Bottom line
Antisynthetase syndrome is an often fatal disease that does not always present in a typical fashion with symptoms of myositis, as lung disease may be the predominant feature. A negative antinuclear antibody test result does not imply antibody negativity, as the autoantigen in these diseases is not located in the nucleus. Prompt diagnosis and appropriate immunosuppressive therapy are critical to improving outcomes.
CASE 5: FEVER, UNDIAGNOSED LUNG DISEASE, NO MYOSITIS
In January 2001, a 39-year-old woman was admitted to the hospital after 5 weeks of fever (temperatures 103°–104°F) and myalgias. An extensive workup was negative except for low-titer antinuclear antibody and for mild basilar fibrosis noted on chest radiography. She left the hospital against medical advice because of frustration with a lack of a specific diagnosis (“fever of unknown origin”).
Two months later, at a follow-up rheumatology consult, she reported more myalgias and arthralgias, as well as fever. Chest radiography now showed pleural effusions. Her fingers had color changes consistent with Raynaud phenomenon. At that time, I diagnosed an undifferentiated connective tissue disease and told her that I suspected an autoimmune condition that would need time to reveal itself. In the meantime, I treated her empirically with prednisone.
In April, she returned, much more short of breath and with more prominent diffuse pulmonary infiltrates. Physical examination revealed subtle Gottron changes. Testing revealed poor pulmonary function: forced vital capacity (FVC) 56%, forced expiratory volume in 1 second (FEV1) 52%, and diffusing capacity for carbon monoxide (Dlco) 40%. Blood testing was positive for anti-PL-12 antibody, one of the non-Jo-1 antisynthetase antibodies. At this time, we treated her with glucocorticoids and tacrolimus.
More than 15 years later, this patient is doing well. Her skin rash, joint symptoms, and fever have not returned, and interestingly, she never developed myositis. Her Raynaud symptoms are mild. Her most recent pulmonary function test results (January 2018) were FVC 75%, FEV1 87%, and Dlco 78%. Although these results are not normal, they are much improved and allow her to be completely functional without supplemental oxygen. Echocardiography showed normal pulmonary artery systolic pressure (25 mm Hg). She was still taking tacrolimus and prednisone. When we tried to stop tacrolimus after she had done well for many years, her condition flared.
Non-Jo-1 antisynthetase syndrome: Bottom line
Patients with a non-Jo-1 antisynthetase syndrome often present without myositis symptoms and may never manifest myositis symptoms. Likely because of this presentation, diagnosis of a specific connective tissue disorder is delayed, perhaps leading to increased mortality risk from pulmonary disease. Chronic immunosuppression is often required for these autoimmune conditions.
CASE 6: DERMATOMYOSITIS, RAPIDLY PROGRESSIVE INTERSTITIAL LUNG DISEASE
A 58-year-old woman presented in the summer of 2012 with a photosensitive rash. The following January, she returned with polyarthritis, mild muscle weakness, and a dermatomyositis-pattern rash. Her CK level was normal, and her antinuclear antibody and Sjögren syndrome antibody test results were negative. She improved on low-dose prednisone and methotrexate.
She was originally referred to me in May of that year for worsening rash and mild weakness. She denied pulmonary symptoms, but examination revealed faint basilar crackles. I increased her prednisone dosage to 20 mg/day and started mycophenolate mofetil mainly for the mild cutaneous and myositis features. I also recommended high-resolution CT of the lungs and pulmonary function tests, which she underwent in early June. High-resolution CT showed nonspecific mild infiltrates with minimal ground-glass opacities.
On July 1, she presented to her local emergency department with severe shortness of breath, requiring oxygen 12 L/min. She had a palmar rash. Repeat high-resolution CT showed dramatic worsening compared with the scan the previous month. Because of continued inadequate oxygenation, she was transferred to our center. A blood test later was positive for antimelanoma differentiation-associated gene 5 (MDA-5) autoantibody, previously known as anticlinically amyopathic dermatomyositis (anti-CADM)-140 antibody (based on immunoprecipitation results).
She died on the third day after transfer, just 2 months after I had originally seen her, at which time she had had no pulmonary symptoms.
Clinically amyopathic dermatomyositis
Anti-CADM-140, first reported from Asia,18–20 is an autoantibody-associated disease but not an antisynthetase. It is associated with dermatomyositis; patients often have a “vasculopathy” with cutaneous ulcerations and palmar papules.
MDA-5 is a cytoplasmic protein that “senses” viral RNA and induces production of type 1 interferon. It is involved in the innate immune defense against viruses.
Anti-MDA-5 positivity is associated with a poor pulmonary outcome.21 In our cohort from the University of Pittsburgh, many patients died within 3 years, compared with about a 40% survival rate in patients with dermatomyositis who tested negative for this antibody. That being said, many patients with anti-MDA-5 do not develop rapidly progressive interstitial lung disease.
Autoimmune interstitial lung disease: Bottom line
Autoimmune interstitial lung disease is easy to miss, especially in the case of a non-Jo-1 syndrome, for 3 important reasons:
- The autoimmune features may initially be subtle (eg, Raynaud phenomena, mild dermatomyositis rash, undifferentiated connective tissue disease)
- Autoantibody testing is not often ordered, is not standardized, or may be unavailable
- Providers are mistakenly reassured that a patient who tests negative for antinuclear antibody does not have an autoimmune condition.
To emphasize the last point, in a cohort of 202 patients who tested positive for an antisynthetase antibody, only half were antinuclear antibody-positive, but nearly three-quarters demonstrated anticytoplasmic staining on indirect immunofluorescence (due to the location of the autoantigen in the cytoplasm), making the latter a better screening test for an antisynthetase antibody. For scleroderma, 99% were antinculear antibody-positive, but for myositis, this test is much less sensitive.22
- Felice KJ, North WA. Inclusion body myositis in Connecticut: observations in 35 patients during an 8-year period. Medicine (Baltimore) 2001; 80(5):320–327. doi:10.1097/00005792-200109000-00006
- Lev EI, Tur-Kaspa I, Ashkenazy I, et al. Distribution of serum creatine kinase activity in young healthy persons. Clin Chim Acta 1999; 279(1-2):107–115. doi:10.1016/S0009-8981(98)00180-6
- Lilleng H, Abeler K, Johnsen SH, et al. Variation of serum creatine kinase (CK) levels and prevalence of persistent hyperCKemia in a Norwegian normal population. The Tromsø Study. Neuromuscul Disord 2011; 21(7):494–500. doi:10.1016/j.nmd.2011.04.007
- Johnston JD, Lloyd M, Mathews JA, Hawthorne SW. Racial variation in serum creatine kinase levels. J R Soc Med 1996; 89(8):462-464. pmid:8795501
- Prelle A, Tancredi L, Sciacco M, et al. Retrospective study of a large population of patients with asymptomatic or minimally symptomatic raised serum creatine kinase levels. J Neurol 2002; 249(3):305–311. pmid:11993531
- D’Adda E, Sciacco M, Fruguglietti ME, et al. Follow-up of a large population of asymptomatic/oligosymptomatic hyperckemic subjects. J Neurol 2006; 253(11):1399–1403. doi:10.1007/s00415-006-0223-y
- Madariaga MG. Polymyositis-like syndrome in hypothyroidism: review of cases reported over the past twenty-five years. Thyroid 2002; 12(4):331–336. doi:10.1089/10507250252949478
- de Sauvage Nolting PR, Buirma RJ, Hutten BA, Kastelein JJ; Dutch ExPRESS Investigator Group. Two-year efficacy and safety of simvastatin 80 mg in familial hypercholesterolemia (the Examination of Probands and Relatives in Statin Studies With Familial Hypercholesterolemia [ExPRESS FH]). Am J Cardiol 2002; 90(2):181–184. doi:10.1016/s0002-9149(02)02449-9
- Bruckert E, Hayem G, Dejager S, Yau C, Bégaud B. Mild to moderate muscular symptoms with high-dosage statin therapy in hyperlipidemic patients--the PRIMO study. Cardiovasc Drugs Ther 2005; 19(6):403–414. doi:10.1007/s10557-005-5686-z
- Mosshammer D, Lorenz G, Meznaric S, Schwarz J, Muche R, Mörike K. Statin use and its association with musculoskeletal symptoms—a cross-sectional study in primary care settings. Fam Pract 2009; 26(2):88–95. doi:10.1093/fampra/cmp006
- Nichols GA, Koro CE. Does statin therapy initiation increase the risk for myopathy? An observational study of 32,225 diabetic and nondiabetic patients. Clin Ther 2007; 29(8):1761–1770. doi:10.1016/j.clinthera.2007.08.022
- Kassardjian CD, Lennon VA, Alfugham NB, Mahler M, Milone M. Clinical features and treatment outcomes of necrotizing autoimmune myopathy. JAMA Neurol 2015; 72(9):996–1003. doi:10.1001/jamaneurol.2015.1207
- Grable-Esposito P, Katzberg HD, Greenberg SA, Srinivasan J, Katz J, Amato AA. Immune-mediated necrotizing myopathy associated with statins. Muscle Nerve 2010; 41(2):185–190. doi:10.1002/mus.21486
- Christopher-Stine L, Casciola-Rosen LA, Hong G, Chung T, Corse AM, Mammen AL. A novel autoantibody recognizing 200-kd and 100-kd proteins is associated with an immune-mediated necrotizing myopathy. Arthritis Rheum 2010; 62(9):2757–2766. doi:10.1002/art.27572
- Mammen AL, Chung T, Christopher-Stine L, et al. Autoantibodies against 3-hydroxy-3-methylglutaryl-coenzyme A reductase in patients with statin-associated autoimmune myopathy. Arthritis Rheum 2011; 63(3):713–721. doi:10.1002/art.30156
- Mammen AL, Tiniakou E. Intravenous immune globulin for statin-triggered autoimmune myopathy. N Engl J Med 2015; 373(17):1680–1682. doi:10.1056/NEJMc1506163
- Aggarwal R, Cassidy E, Fertig N, et al. Patients with non-Jo-1 anti-tRNA-synthetase autoantibodies have worse survival than Jo-1 positive patients. Ann Rheum Dis 2014; 73(1):227–232. doi:10.1136/annrheumdis-2012-201800
- Sato S, Hirakata M, Kuwana M, et al. Autoantibodies to a 140-kd polypeptide, CADM-140, in Japanese patients with clinically amyopathic dermatomyositis. Arthritis Rheum 2005; 52(5):1571–1576. doi:10.1002/art.21023
- Sato S, Hoshino K, Satoh T, et al. RNA helicase encoded by melanoma differentiation-associated gene 5 is a major autoantigen in patients with clinically amyopathic dermatomyositis: association with rapidly progressive interstitial lung disease. Arthritis Rheum 2009; 60(7):2193–2200. doi:10.1002/art.24621
- Chen F, Wang D, Shu X, Nakashima R, Wang G. Anti-MDA5 antibody is associated with A/SIP and decreased T cells in peripheral blood and predicts poor prognosis of ILD in Chinese patients with dermatomyositis. Rheumatol Int 2012; 32(12):3909–3915. doi:10.1007/s00296-011-2323-y
- Moghadam-Kia S, Oddis CV, Sato S, Kuwana M, Aggarwal R. Anti-melanoma differentiation-associated gene 5 is associated with rapidly progressive lung disease and poor survival in US patients with amyopathic and myopathic dermatomyositis. Arthritis Care Res (Hoboken) 2016; 68(5):689–694. doi:10.1002/acr.22728
- Aggarwal R, Dhillon N, Fertig N, Koontz D, Qi Z, Oddis CV. A negative antinuclear antibody does not indicate autoantibody negativity in myositis: role of anticytoplasmic antibody as a screening test for antisynthetase syndrome. J Rheumatol 2017; 44(2):223–229. doi:10.3899/jrheum.160618
Myopathies can present with a wide variety of symptoms, so patients with muscle weakness are often seen initially by a general practitioner. Nonrheumatologists should be able to evaluate a patient presenting with muscle weakness or myalgia and be aware of red flags indicating potentially dangerous syndromes that require a prompt, thorough investigation.
This article reviews selected causes of muscle weakness, such as statin-induced and autoimmune disorders, and systemic features of inflammatory myopathies beyond myositis, such as dermatologic and pulmonary manifestations.
FOCUSING THE EVALUATION
The evaluation of a patient presenting with muscle weakness should include several assessments:
Temporal progression. Was the onset of symptoms rapid or insidious? Patterns of onset may give clues to etiology, including the possibility of an associated autoimmune condition.
Location of muscle weakness. Are symptoms global or localized? And if localized, are they proximal or distal? Proximal weakness can be manifested by difficulty rising from a chair (hip muscles) or combing one’s hair (shoulder muscles), whereas distal weakness can involve difficulty standing on toes (gastrocnemius and soleus muscles) or performing fine motor activities (intrinsic hand muscles).
Symmetry. A focal or asymmetric pattern often has a neurologic etiology, but this could also be consistent with inclusion body myositis.
Other symptoms. Arthritis, rash, and swallowing problems point to a possible underlying rheumatologic disease. Weight gain or loss may indicate a thyroid disorder.
Family history. Some patients report that others in their family have this pattern of weakness, indicating a likely genetic myopathy. If the patient reports a relative with multiple sclerosis, lupus erythematosus, rheumatoid arthritis, or another autoimmune disease, then an immune-mediated myopathy should be considered.
Medications should be reviewed, particularly statins.
CASE 1: SLOWLY PROGRESSIVE WEAKNESS
A 65-year-old man presented with the insidious onset of muscle weakness and episodes of falling. On review of his medical record, his serum creatine kinase (CK) levels were elevated at various periods at 2 to 4 times the upper limit of normal. Electromyography (EMG) previously showed a myopathic pattern, and a muscle biopsy was abnormal, consistent with endomysial inflammation (term is consistent with “polymyositis”). He was treated for polymyositis for several years with prednisone alone, with steroids plus methotrexate, and with combined immunosuppression including methotrexate and azathioprine, but with no improvement. Eventually, another muscle biopsy revealed inclusion bodies with rimmed vacuoles, consistent with inclusion body myositis.
Inclusion body myositis
Inclusion body myositis is the most common myopathy in middle-aged to elderly people, especially men. These patients are often told “You are just getting old,” but they have a defined condition. It should also be considered in patients failing to respond to treatment or with those with “refractory” polymyositis.
The onset of muscle weakness is insidious and painless, and the weakness progresses slowly. The pattern is distal and asymmetric (eg, foot drop), and muscle atrophy typically affects the forearm flexors, quadriceps, and intrinsic muscles of the hands.1
Magnetic resonance imaging may show marked muscle atrophy. Unfortunately, no treatment has shown efficacy, and most neuromuscular and rheumatology experts do not treat inclusion body myositis with immunosuppressive drugs.
CASE 2: MILD MYALGIA WITHOUT WEAKNESS
A black 52-year-old man was referred because of myalgia and a CK level of 862 U/L (reference range < 200). His physician wanted to start him on a statin but was hesitant to do so without first consulting a rheumatologist.
The patient had a long history of mild arthralgias and myalgias without muscle weakness. He had dyslipidemia and hypertension. He reported no family history of myopathy and no illicit drug use. He was formerly an athlete. Medications included a thiazide diuretic and a beta-blocker. On examination, his muscles were strong (rated 5 on a scale of 5) in the upper and lower extremities, without atrophy.
His records showed that his CK levels had risen and fallen repeatedly over the past few years, ranging from 600 to 1,100 U/L. On further questioning, he reported that when he had joined the army 30 years previously, a physician had recommended he undergo a liver biopsy in view of elevated liver function tests, but that he had refused because he felt fine.
Currently, his gamma-glutamyl transpeptidase levels were normal.
Idiopathic ‘hyperCKemia’
So-called idiopathic hyperCKemia is not a form of myositis but merely a laboratory result outside the “normal” range. Reference ranges are based predominantly on measurements in white people and on an assumption that the distribution is Gaussian (bell-shaped). A normal CK level is usually defined as less than 200 U/L. Using this standard, up to 20% of men and 5% of women have hyperCKemia.2
However, CK levels vary by sex and ethnicity, with mean levels highest in black men, followed by black women, white men, and white women. The mean level in black men is higher than the standard cutoff point for normal, and especially in this population, there is wide fluctuation around the mean, leading to hyperCKemia quite frequently in black men. Exercise and manual labor also drive up CK levels.3–5
Idiopathic hyperCKemia is benign. D’Adda et al6 followed 55 patients for a mean of 7.5 years. CK levels normalized in 12 patients or at least decreased in 24. Most remained symptom-free or had minimal symptoms.
Idiopathic hyperCKemia: Bottom line
Before prescribing a statin, determine the baseline CK level. If slightly elevated (ie, up to 3 to 5 times the upper limit of normal, or even higher) in the setting of normal muscle strength, there is no need for electromyography or muscle biopsy, and the patient can certainly receive a statin. Most of these patients do not need to see a rheumatologist but can simply have their CK and muscle strength monitored.
CLASSIFYING MYOSITIS
Myositis (idiopathic inflammatory myopathy) is a heterogeneous group of autoimmune syndromes of unknown cause characterized by chronic muscle weakness and inflammation of striated muscle. These syndromes likely arise as a result of genetic predisposition and an environmental or infectious “hit.”
Myositis is rare, with an incidence of 5 to 10 cases per million per year and an estimated prevalence of 50 to 90 cases per million. It has 2 incidence peaks: 1 in childhood (age 5–15) and another in adult midlife (age 30–50). Women are affected 2 to 3 times more often than men, with black women most commonly affected.
Myositis is traditionally classified as follows:
- Adult polymyositis
- Adult dermatomyositis
- Juvenile myositis (dermatomyositis much more frequent than polymyositis)
- Malignancy-associated myositis (usually dermatomyositis)
- Myositis overlapping with another autoimmune disease
- Inclusion body myositis.
However, polymyositis is less common than we originally thought, and the term necrotizing myopathy is now used in many patients, as noted in the case studies below. Further, myositis overlap syndromes are being increasingly diagnosed, likely related to the emergence of autoantibodies and clinical “syndromes” associated with these autoantibody subsets (discussed in cases below).
Dermatomyositis
Dermatomyositis is characterized by muscle weakness and a rash that can be obvious or subtle. Classic skin lesions are Gottron papules, which are raised, flat-topped red or purplish lesions over the knuckles, elbows, or knees.
Lesions may be confused with those of psoriasis. There can also be a V-neck rash over the anterior chest or upper back (“shawl sign”) or a rash over the lateral thigh (“holster sign”). A facial rash may occur, but unlike lupus, dermatomyositis does not spare the nasolabial area. However, the V-neck rash can be similar to that seen in lupus.
Dermatomyositis may cause muscle pain, perhaps related to muscle ischemia, whereas polymyositis and necrotizing myopathy are often painless. However, pain is also associated with fibromyalgia, which may be seen in many autoimmune conditions. It is important not to overtreat rheumatologic diseases with immunosuppression to try to control pain if the pain is actually caused by fibromyalgia.
Polymyositis mimics
Hypothyroid myopathy can present as classic polymyositis. The serum CK may be elevated, and there may be myalgias, muscle hypertrophy with stiffness, weakness, cramps, and even features of a proximal myopathy, and rhabdomyolysis. The electromyogram can be normal or myopathic. Results of muscle biopsy are often normal but may show focal necrosis and mild inflammatory infiltrates, thus mimicking that seen with inflammatory myopathy.7
Drug-induced or toxic myopathies can also mimic polymyositis. Statins are among the most commonly prescribed drugs in the United States, with more than 35 million people taking them. Statins are generally well tolerated but have a broad spectrum of toxicity, ranging from myalgias to life-threatening rhabdomyolysis. Myalgias lead to about 5% to 10% of patients refusing to take a statin or stopping it on their own.
Myalgias affect up to 20% of statin users in clinical practice.8,9 A small cross-sectional study10 of 1,000 patients in a primary care setting found that the risk of muscle complaints in statin users was 1.5 times higher than in nonstatin users, similar to findings in other studies.
My strategy for managing a patient with possible statin-induced myopathy is illustrated in Figure 1.
CASE 3: WEAKNESS, VERY HIGH CK ON A STATIN
In March 2010, a 67-year-old woman presented with muscle weakness. She had a history of hypertension, hyperlipidemia, and, more than 10 years previously, uterine cancer. In 2004, she was given atorvastatin for dyslipidemia. Four years later, she developed lower-extremity weakness, which her doctor attributed to normal aging. A year after that, she found it difficult to walk up steps and lift her arms overhead. In June 2009, she stopped taking the atorvastatin on her own, but the weakness did not improve.
In September 2009, she returned to her doctor, who found her CK level was 6,473 U/L but believed it to be an error, so the test was repeated, with a result of 9,375 U/L. She had no rash or joint involvement.
She was admitted to the hospital and underwent muscle biopsy, which showed myonecrosis with no inflammation or vasculitis. She was treated with prednisone 60 mg/day, and her elevated CK level and weakness improved.
Immune-mediated necrotizing myopathy associated with statins
The hallmark of necrotizing myopathy is myonecrosis without significant inflammation.12 This pattern contrasts with that of polymyositis, which is characterized by lymphocytic inflammation.
Although statins became available in the United States in 1987, immune-mediated necrotizing myopathy associated with statins was first described only in 2010. In that report, Grable-Esposito et al13 described 25 patients from 2 neuromuscular centers seen between 2000 and 2008 who had elevated CK and proximal weakness during or after statin use, both of which persisted despite stopping the statin. Patients improved with immunosuppressive agents but had a relapse when steroids were stopped or tapered, a pattern typical in autoimmune disease.
Autoantibody defines subgroup of necrotizing myopathy
Also in 2010, Christopher-Stine et al14 reported an antibody associated with necrotizing myopathy. Of 38 patients with the condition, 16 were found to have an abnormal “doublet” autoantibody recognizing 200- and 100-kDa proteins. All patients had weakness and a high CK level, and 63% had statin exposure before the weakness (this percentage increased to 83% in patients older than 50). All responded to immunosuppressive therapy, and many had a relapse when it was withdrawn.
Statins lower cholesterol by inhibiting 3-hydroxy-3-methylglutaryl-Co A reductase (HMGCR), and paradoxically, they also upregulate it. HMGCR has a molecular weight of 97 kDa. Mammen et al15 identified HMGCR as the 100-kDa target of the identified antibody and developed an enzyme-linked immunosorbent assay for it. Of 750 patients presenting to one center, only 45 (6%) had anti-HMGCR autoantibodies, but all 16 patients who had the abnormal doublet antibody tested positive for anti-HMGCR. Regenerating muscle cells express high levels of HMGCR, which may sustain the immune response after statins are discontinued.
Case 3 continued: Intravenous immunoglobulin brings improvement
In March 2010, when the 67-year-old patient presented to our myositis center, her CK level was 5,800 U/L, which increased as prednisone was tapered. She still felt weak. On examination, her muscle strength findings were deltoids 4+/5, neck flexors 4/5, and iliopsoas 3+/5. She was treated with methotrexate and azathioprine without benefit. She was next treated with intravenous immunoglobulin, and after 3 months, her strength normalized for the first time in years. Her CK level decreased but did not normalize. Testing showed that she was positive for anti-HMGCR autoantibody, as this test had become commercially available.
In 2015, Mammen and Tiniakou16 suggested using intravenous immunoglobulin as first-line therapy for statin-associated autoimmune necrotizing myopathy, based on experience at a single center with 3 patients who declined glucocorticoid treatment.
Necrotizing myopathy: Bottom line
Myositis overlap syndromes
Heterogeneity is the rule in myositis, and it can present with a wide variety of signs and symptoms as outlined in Table 2.
CASE 4: FEVER, NEW ‘RHEUMATOID ARTHRITIS,’ AND LUNG DISEASE
A 52-year-old woman with knee osteoarthritis saw her primary care physician in November 2013 for dyspnea and low-grade fever. The next month, she presented with polyarthritis, muscle weakness, and Raynaud phenomenon.
In January 2014, she developed acrocyanosis of her fingers. Examination revealed hyperkeratotic, cracked areas of her fingers. Her oxygen saturation by pulse oximetry was low. She was admitted to the hospital. Her doctor suspected new onset of rheumatoid arthritis, but blood tests revealed a negative antinuclear antibody, so an autoimmune condition was deemed unlikely. Her CK was mildly elevated at 350 U/L.
Because of her dyspnea, an open-lung biopsy was performed. High-resolution computed tomography (CT) revealed infiltrates and ground-glass opacities, leading to the diagnosis of nonspecific interstitial pneumonia. A rheumatologist was consulted and recommended pulse methylprednisolone, followed by prednisone 60 mg/day and mycophenolate mofetil. Testing for Jo-1 antibodies was positive.
Antisynthetase syndrome
The antisynthetase syndrome is a clinically heterogeneous condition that can occur with any or all of the following:
- Fever
- Myositis
- Arthritis (often misdiagnosed as rheumatoid arthritis)
- Raynaud phenomenon
- Mechanic’s hands (hyperkeratotic roughness with fissures on the lateral aspects of the fingers and finger pads)
- Interstitial lung disease.
The skin rashes and myositis may be subtle, making the presentation “lung-dominant,” and nonrheumatologists should be aware of this syndrome. Although in our patient the condition developed in a classic manner, with all of the aforementioned features of the antisynthetase syndrome, some patients will manifest one or a few of the features.
Clinically, patients with the Jo-1 antisynthetase syndrome often present differently than those with non-Jo-1 antisynthetase autoantibodies. When we compared 122 patients with Jo-1 vs 80 patients with a non-Jo-1 antisynthetase autoantibody, patients with Jo-1 antibodies were more likely to have initially received a diagnosis of myositis (83%), while myositis was the original diagnosis in only 17% of those possessing non-Jo-1 antisynthetase autoantibodies. In fact, many patients (approximately 50%) were diagnosed as having undifferentiated connective tissue disease or an overlap syndrome, and 13% had scleroderma as their first diagnosis.17
We also found that the survival rate was higher in patients with Jo-1 syndrome compared with patients with non-Jo-1 antisynthetase syndromes. We attributed the difference in survival rates to a delayed diagnosis in the non-Jo-1 group, perhaps due to their “nonclassic” presentations of the antisynthetase syndrome, delaying appropriate treatment. Patients received a diagnosis of Jo-1 antibody syndrome after a mean of 0.4 year (range 0.2–0.8), while those with a non-Jo-1 antisynthetase autoantibody had a delay in diagnosis of 1.0 year (range 0.4–5.1) (P < .01).17
In nearly half the cases in this cohort, pulmonary fibrosis was the cause of death, with primary pulmonary hypertension being the second leading cause (11%).
Antisynthetase syndrome: Bottom line
Antisynthetase syndrome is an often fatal disease that does not always present in a typical fashion with symptoms of myositis, as lung disease may be the predominant feature. A negative antinuclear antibody test result does not imply antibody negativity, as the autoantigen in these diseases is not located in the nucleus. Prompt diagnosis and appropriate immunosuppressive therapy are critical to improving outcomes.
CASE 5: FEVER, UNDIAGNOSED LUNG DISEASE, NO MYOSITIS
In January 2001, a 39-year-old woman was admitted to the hospital after 5 weeks of fever (temperatures 103°–104°F) and myalgias. An extensive workup was negative except for low-titer antinuclear antibody and for mild basilar fibrosis noted on chest radiography. She left the hospital against medical advice because of frustration with a lack of a specific diagnosis (“fever of unknown origin”).
Two months later, at a follow-up rheumatology consult, she reported more myalgias and arthralgias, as well as fever. Chest radiography now showed pleural effusions. Her fingers had color changes consistent with Raynaud phenomenon. At that time, I diagnosed an undifferentiated connective tissue disease and told her that I suspected an autoimmune condition that would need time to reveal itself. In the meantime, I treated her empirically with prednisone.
In April, she returned, much more short of breath and with more prominent diffuse pulmonary infiltrates. Physical examination revealed subtle Gottron changes. Testing revealed poor pulmonary function: forced vital capacity (FVC) 56%, forced expiratory volume in 1 second (FEV1) 52%, and diffusing capacity for carbon monoxide (Dlco) 40%. Blood testing was positive for anti-PL-12 antibody, one of the non-Jo-1 antisynthetase antibodies. At this time, we treated her with glucocorticoids and tacrolimus.
More than 15 years later, this patient is doing well. Her skin rash, joint symptoms, and fever have not returned, and interestingly, she never developed myositis. Her Raynaud symptoms are mild. Her most recent pulmonary function test results (January 2018) were FVC 75%, FEV1 87%, and Dlco 78%. Although these results are not normal, they are much improved and allow her to be completely functional without supplemental oxygen. Echocardiography showed normal pulmonary artery systolic pressure (25 mm Hg). She was still taking tacrolimus and prednisone. When we tried to stop tacrolimus after she had done well for many years, her condition flared.
Non-Jo-1 antisynthetase syndrome: Bottom line
Patients with a non-Jo-1 antisynthetase syndrome often present without myositis symptoms and may never manifest myositis symptoms. Likely because of this presentation, diagnosis of a specific connective tissue disorder is delayed, perhaps leading to increased mortality risk from pulmonary disease. Chronic immunosuppression is often required for these autoimmune conditions.
CASE 6: DERMATOMYOSITIS, RAPIDLY PROGRESSIVE INTERSTITIAL LUNG DISEASE
A 58-year-old woman presented in the summer of 2012 with a photosensitive rash. The following January, she returned with polyarthritis, mild muscle weakness, and a dermatomyositis-pattern rash. Her CK level was normal, and her antinuclear antibody and Sjögren syndrome antibody test results were negative. She improved on low-dose prednisone and methotrexate.
She was originally referred to me in May of that year for worsening rash and mild weakness. She denied pulmonary symptoms, but examination revealed faint basilar crackles. I increased her prednisone dosage to 20 mg/day and started mycophenolate mofetil mainly for the mild cutaneous and myositis features. I also recommended high-resolution CT of the lungs and pulmonary function tests, which she underwent in early June. High-resolution CT showed nonspecific mild infiltrates with minimal ground-glass opacities.
On July 1, she presented to her local emergency department with severe shortness of breath, requiring oxygen 12 L/min. She had a palmar rash. Repeat high-resolution CT showed dramatic worsening compared with the scan the previous month. Because of continued inadequate oxygenation, she was transferred to our center. A blood test later was positive for antimelanoma differentiation-associated gene 5 (MDA-5) autoantibody, previously known as anticlinically amyopathic dermatomyositis (anti-CADM)-140 antibody (based on immunoprecipitation results).
She died on the third day after transfer, just 2 months after I had originally seen her, at which time she had had no pulmonary symptoms.
Clinically amyopathic dermatomyositis
Anti-CADM-140, first reported from Asia,18–20 is an autoantibody-associated disease but not an antisynthetase. It is associated with dermatomyositis; patients often have a “vasculopathy” with cutaneous ulcerations and palmar papules.
MDA-5 is a cytoplasmic protein that “senses” viral RNA and induces production of type 1 interferon. It is involved in the innate immune defense against viruses.
Anti-MDA-5 positivity is associated with a poor pulmonary outcome.21 In our cohort from the University of Pittsburgh, many patients died within 3 years, compared with about a 40% survival rate in patients with dermatomyositis who tested negative for this antibody. That being said, many patients with anti-MDA-5 do not develop rapidly progressive interstitial lung disease.
Autoimmune interstitial lung disease: Bottom line
Autoimmune interstitial lung disease is easy to miss, especially in the case of a non-Jo-1 syndrome, for 3 important reasons:
- The autoimmune features may initially be subtle (eg, Raynaud phenomena, mild dermatomyositis rash, undifferentiated connective tissue disease)
- Autoantibody testing is not often ordered, is not standardized, or may be unavailable
- Providers are mistakenly reassured that a patient who tests negative for antinuclear antibody does not have an autoimmune condition.
To emphasize the last point, in a cohort of 202 patients who tested positive for an antisynthetase antibody, only half were antinuclear antibody-positive, but nearly three-quarters demonstrated anticytoplasmic staining on indirect immunofluorescence (due to the location of the autoantigen in the cytoplasm), making the latter a better screening test for an antisynthetase antibody. For scleroderma, 99% were antinculear antibody-positive, but for myositis, this test is much less sensitive.22
Myopathies can present with a wide variety of symptoms, so patients with muscle weakness are often seen initially by a general practitioner. Nonrheumatologists should be able to evaluate a patient presenting with muscle weakness or myalgia and be aware of red flags indicating potentially dangerous syndromes that require a prompt, thorough investigation.
This article reviews selected causes of muscle weakness, such as statin-induced and autoimmune disorders, and systemic features of inflammatory myopathies beyond myositis, such as dermatologic and pulmonary manifestations.
FOCUSING THE EVALUATION
The evaluation of a patient presenting with muscle weakness should include several assessments:
Temporal progression. Was the onset of symptoms rapid or insidious? Patterns of onset may give clues to etiology, including the possibility of an associated autoimmune condition.
Location of muscle weakness. Are symptoms global or localized? And if localized, are they proximal or distal? Proximal weakness can be manifested by difficulty rising from a chair (hip muscles) or combing one’s hair (shoulder muscles), whereas distal weakness can involve difficulty standing on toes (gastrocnemius and soleus muscles) or performing fine motor activities (intrinsic hand muscles).
Symmetry. A focal or asymmetric pattern often has a neurologic etiology, but this could also be consistent with inclusion body myositis.
Other symptoms. Arthritis, rash, and swallowing problems point to a possible underlying rheumatologic disease. Weight gain or loss may indicate a thyroid disorder.
Family history. Some patients report that others in their family have this pattern of weakness, indicating a likely genetic myopathy. If the patient reports a relative with multiple sclerosis, lupus erythematosus, rheumatoid arthritis, or another autoimmune disease, then an immune-mediated myopathy should be considered.
Medications should be reviewed, particularly statins.
CASE 1: SLOWLY PROGRESSIVE WEAKNESS
A 65-year-old man presented with the insidious onset of muscle weakness and episodes of falling. On review of his medical record, his serum creatine kinase (CK) levels were elevated at various periods at 2 to 4 times the upper limit of normal. Electromyography (EMG) previously showed a myopathic pattern, and a muscle biopsy was abnormal, consistent with endomysial inflammation (term is consistent with “polymyositis”). He was treated for polymyositis for several years with prednisone alone, with steroids plus methotrexate, and with combined immunosuppression including methotrexate and azathioprine, but with no improvement. Eventually, another muscle biopsy revealed inclusion bodies with rimmed vacuoles, consistent with inclusion body myositis.
Inclusion body myositis
Inclusion body myositis is the most common myopathy in middle-aged to elderly people, especially men. These patients are often told “You are just getting old,” but they have a defined condition. It should also be considered in patients failing to respond to treatment or with those with “refractory” polymyositis.
The onset of muscle weakness is insidious and painless, and the weakness progresses slowly. The pattern is distal and asymmetric (eg, foot drop), and muscle atrophy typically affects the forearm flexors, quadriceps, and intrinsic muscles of the hands.1
Magnetic resonance imaging may show marked muscle atrophy. Unfortunately, no treatment has shown efficacy, and most neuromuscular and rheumatology experts do not treat inclusion body myositis with immunosuppressive drugs.
CASE 2: MILD MYALGIA WITHOUT WEAKNESS
A black 52-year-old man was referred because of myalgia and a CK level of 862 U/L (reference range < 200). His physician wanted to start him on a statin but was hesitant to do so without first consulting a rheumatologist.
The patient had a long history of mild arthralgias and myalgias without muscle weakness. He had dyslipidemia and hypertension. He reported no family history of myopathy and no illicit drug use. He was formerly an athlete. Medications included a thiazide diuretic and a beta-blocker. On examination, his muscles were strong (rated 5 on a scale of 5) in the upper and lower extremities, without atrophy.
His records showed that his CK levels had risen and fallen repeatedly over the past few years, ranging from 600 to 1,100 U/L. On further questioning, he reported that when he had joined the army 30 years previously, a physician had recommended he undergo a liver biopsy in view of elevated liver function tests, but that he had refused because he felt fine.
Currently, his gamma-glutamyl transpeptidase levels were normal.
Idiopathic ‘hyperCKemia’
So-called idiopathic hyperCKemia is not a form of myositis but merely a laboratory result outside the “normal” range. Reference ranges are based predominantly on measurements in white people and on an assumption that the distribution is Gaussian (bell-shaped). A normal CK level is usually defined as less than 200 U/L. Using this standard, up to 20% of men and 5% of women have hyperCKemia.2
However, CK levels vary by sex and ethnicity, with mean levels highest in black men, followed by black women, white men, and white women. The mean level in black men is higher than the standard cutoff point for normal, and especially in this population, there is wide fluctuation around the mean, leading to hyperCKemia quite frequently in black men. Exercise and manual labor also drive up CK levels.3–5
Idiopathic hyperCKemia is benign. D’Adda et al6 followed 55 patients for a mean of 7.5 years. CK levels normalized in 12 patients or at least decreased in 24. Most remained symptom-free or had minimal symptoms.
Idiopathic hyperCKemia: Bottom line
Before prescribing a statin, determine the baseline CK level. If slightly elevated (ie, up to 3 to 5 times the upper limit of normal, or even higher) in the setting of normal muscle strength, there is no need for electromyography or muscle biopsy, and the patient can certainly receive a statin. Most of these patients do not need to see a rheumatologist but can simply have their CK and muscle strength monitored.
CLASSIFYING MYOSITIS
Myositis (idiopathic inflammatory myopathy) is a heterogeneous group of autoimmune syndromes of unknown cause characterized by chronic muscle weakness and inflammation of striated muscle. These syndromes likely arise as a result of genetic predisposition and an environmental or infectious “hit.”
Myositis is rare, with an incidence of 5 to 10 cases per million per year and an estimated prevalence of 50 to 90 cases per million. It has 2 incidence peaks: 1 in childhood (age 5–15) and another in adult midlife (age 30–50). Women are affected 2 to 3 times more often than men, with black women most commonly affected.
Myositis is traditionally classified as follows:
- Adult polymyositis
- Adult dermatomyositis
- Juvenile myositis (dermatomyositis much more frequent than polymyositis)
- Malignancy-associated myositis (usually dermatomyositis)
- Myositis overlapping with another autoimmune disease
- Inclusion body myositis.
However, polymyositis is less common than we originally thought, and the term necrotizing myopathy is now used in many patients, as noted in the case studies below. Further, myositis overlap syndromes are being increasingly diagnosed, likely related to the emergence of autoantibodies and clinical “syndromes” associated with these autoantibody subsets (discussed in cases below).
Dermatomyositis
Dermatomyositis is characterized by muscle weakness and a rash that can be obvious or subtle. Classic skin lesions are Gottron papules, which are raised, flat-topped red or purplish lesions over the knuckles, elbows, or knees.
Lesions may be confused with those of psoriasis. There can also be a V-neck rash over the anterior chest or upper back (“shawl sign”) or a rash over the lateral thigh (“holster sign”). A facial rash may occur, but unlike lupus, dermatomyositis does not spare the nasolabial area. However, the V-neck rash can be similar to that seen in lupus.
Dermatomyositis may cause muscle pain, perhaps related to muscle ischemia, whereas polymyositis and necrotizing myopathy are often painless. However, pain is also associated with fibromyalgia, which may be seen in many autoimmune conditions. It is important not to overtreat rheumatologic diseases with immunosuppression to try to control pain if the pain is actually caused by fibromyalgia.
Polymyositis mimics
Hypothyroid myopathy can present as classic polymyositis. The serum CK may be elevated, and there may be myalgias, muscle hypertrophy with stiffness, weakness, cramps, and even features of a proximal myopathy, and rhabdomyolysis. The electromyogram can be normal or myopathic. Results of muscle biopsy are often normal but may show focal necrosis and mild inflammatory infiltrates, thus mimicking that seen with inflammatory myopathy.7
Drug-induced or toxic myopathies can also mimic polymyositis. Statins are among the most commonly prescribed drugs in the United States, with more than 35 million people taking them. Statins are generally well tolerated but have a broad spectrum of toxicity, ranging from myalgias to life-threatening rhabdomyolysis. Myalgias lead to about 5% to 10% of patients refusing to take a statin or stopping it on their own.
Myalgias affect up to 20% of statin users in clinical practice.8,9 A small cross-sectional study10 of 1,000 patients in a primary care setting found that the risk of muscle complaints in statin users was 1.5 times higher than in nonstatin users, similar to findings in other studies.
My strategy for managing a patient with possible statin-induced myopathy is illustrated in Figure 1.
CASE 3: WEAKNESS, VERY HIGH CK ON A STATIN
In March 2010, a 67-year-old woman presented with muscle weakness. She had a history of hypertension, hyperlipidemia, and, more than 10 years previously, uterine cancer. In 2004, she was given atorvastatin for dyslipidemia. Four years later, she developed lower-extremity weakness, which her doctor attributed to normal aging. A year after that, she found it difficult to walk up steps and lift her arms overhead. In June 2009, she stopped taking the atorvastatin on her own, but the weakness did not improve.
In September 2009, she returned to her doctor, who found her CK level was 6,473 U/L but believed it to be an error, so the test was repeated, with a result of 9,375 U/L. She had no rash or joint involvement.
She was admitted to the hospital and underwent muscle biopsy, which showed myonecrosis with no inflammation or vasculitis. She was treated with prednisone 60 mg/day, and her elevated CK level and weakness improved.
Immune-mediated necrotizing myopathy associated with statins
The hallmark of necrotizing myopathy is myonecrosis without significant inflammation.12 This pattern contrasts with that of polymyositis, which is characterized by lymphocytic inflammation.
Although statins became available in the United States in 1987, immune-mediated necrotizing myopathy associated with statins was first described only in 2010. In that report, Grable-Esposito et al13 described 25 patients from 2 neuromuscular centers seen between 2000 and 2008 who had elevated CK and proximal weakness during or after statin use, both of which persisted despite stopping the statin. Patients improved with immunosuppressive agents but had a relapse when steroids were stopped or tapered, a pattern typical in autoimmune disease.
Autoantibody defines subgroup of necrotizing myopathy
Also in 2010, Christopher-Stine et al14 reported an antibody associated with necrotizing myopathy. Of 38 patients with the condition, 16 were found to have an abnormal “doublet” autoantibody recognizing 200- and 100-kDa proteins. All patients had weakness and a high CK level, and 63% had statin exposure before the weakness (this percentage increased to 83% in patients older than 50). All responded to immunosuppressive therapy, and many had a relapse when it was withdrawn.
Statins lower cholesterol by inhibiting 3-hydroxy-3-methylglutaryl-Co A reductase (HMGCR), and paradoxically, they also upregulate it. HMGCR has a molecular weight of 97 kDa. Mammen et al15 identified HMGCR as the 100-kDa target of the identified antibody and developed an enzyme-linked immunosorbent assay for it. Of 750 patients presenting to one center, only 45 (6%) had anti-HMGCR autoantibodies, but all 16 patients who had the abnormal doublet antibody tested positive for anti-HMGCR. Regenerating muscle cells express high levels of HMGCR, which may sustain the immune response after statins are discontinued.
Case 3 continued: Intravenous immunoglobulin brings improvement
In March 2010, when the 67-year-old patient presented to our myositis center, her CK level was 5,800 U/L, which increased as prednisone was tapered. She still felt weak. On examination, her muscle strength findings were deltoids 4+/5, neck flexors 4/5, and iliopsoas 3+/5. She was treated with methotrexate and azathioprine without benefit. She was next treated with intravenous immunoglobulin, and after 3 months, her strength normalized for the first time in years. Her CK level decreased but did not normalize. Testing showed that she was positive for anti-HMGCR autoantibody, as this test had become commercially available.
In 2015, Mammen and Tiniakou16 suggested using intravenous immunoglobulin as first-line therapy for statin-associated autoimmune necrotizing myopathy, based on experience at a single center with 3 patients who declined glucocorticoid treatment.
Necrotizing myopathy: Bottom line
Myositis overlap syndromes
Heterogeneity is the rule in myositis, and it can present with a wide variety of signs and symptoms as outlined in Table 2.
CASE 4: FEVER, NEW ‘RHEUMATOID ARTHRITIS,’ AND LUNG DISEASE
A 52-year-old woman with knee osteoarthritis saw her primary care physician in November 2013 for dyspnea and low-grade fever. The next month, she presented with polyarthritis, muscle weakness, and Raynaud phenomenon.
In January 2014, she developed acrocyanosis of her fingers. Examination revealed hyperkeratotic, cracked areas of her fingers. Her oxygen saturation by pulse oximetry was low. She was admitted to the hospital. Her doctor suspected new onset of rheumatoid arthritis, but blood tests revealed a negative antinuclear antibody, so an autoimmune condition was deemed unlikely. Her CK was mildly elevated at 350 U/L.
Because of her dyspnea, an open-lung biopsy was performed. High-resolution computed tomography (CT) revealed infiltrates and ground-glass opacities, leading to the diagnosis of nonspecific interstitial pneumonia. A rheumatologist was consulted and recommended pulse methylprednisolone, followed by prednisone 60 mg/day and mycophenolate mofetil. Testing for Jo-1 antibodies was positive.
Antisynthetase syndrome
The antisynthetase syndrome is a clinically heterogeneous condition that can occur with any or all of the following:
- Fever
- Myositis
- Arthritis (often misdiagnosed as rheumatoid arthritis)
- Raynaud phenomenon
- Mechanic’s hands (hyperkeratotic roughness with fissures on the lateral aspects of the fingers and finger pads)
- Interstitial lung disease.
The skin rashes and myositis may be subtle, making the presentation “lung-dominant,” and nonrheumatologists should be aware of this syndrome. Although in our patient the condition developed in a classic manner, with all of the aforementioned features of the antisynthetase syndrome, some patients will manifest one or a few of the features.
Clinically, patients with the Jo-1 antisynthetase syndrome often present differently than those with non-Jo-1 antisynthetase autoantibodies. When we compared 122 patients with Jo-1 vs 80 patients with a non-Jo-1 antisynthetase autoantibody, patients with Jo-1 antibodies were more likely to have initially received a diagnosis of myositis (83%), while myositis was the original diagnosis in only 17% of those possessing non-Jo-1 antisynthetase autoantibodies. In fact, many patients (approximately 50%) were diagnosed as having undifferentiated connective tissue disease or an overlap syndrome, and 13% had scleroderma as their first diagnosis.17
We also found that the survival rate was higher in patients with Jo-1 syndrome compared with patients with non-Jo-1 antisynthetase syndromes. We attributed the difference in survival rates to a delayed diagnosis in the non-Jo-1 group, perhaps due to their “nonclassic” presentations of the antisynthetase syndrome, delaying appropriate treatment. Patients received a diagnosis of Jo-1 antibody syndrome after a mean of 0.4 year (range 0.2–0.8), while those with a non-Jo-1 antisynthetase autoantibody had a delay in diagnosis of 1.0 year (range 0.4–5.1) (P < .01).17
In nearly half the cases in this cohort, pulmonary fibrosis was the cause of death, with primary pulmonary hypertension being the second leading cause (11%).
Antisynthetase syndrome: Bottom line
Antisynthetase syndrome is an often fatal disease that does not always present in a typical fashion with symptoms of myositis, as lung disease may be the predominant feature. A negative antinuclear antibody test result does not imply antibody negativity, as the autoantigen in these diseases is not located in the nucleus. Prompt diagnosis and appropriate immunosuppressive therapy are critical to improving outcomes.
CASE 5: FEVER, UNDIAGNOSED LUNG DISEASE, NO MYOSITIS
In January 2001, a 39-year-old woman was admitted to the hospital after 5 weeks of fever (temperatures 103°–104°F) and myalgias. An extensive workup was negative except for low-titer antinuclear antibody and for mild basilar fibrosis noted on chest radiography. She left the hospital against medical advice because of frustration with a lack of a specific diagnosis (“fever of unknown origin”).
Two months later, at a follow-up rheumatology consult, she reported more myalgias and arthralgias, as well as fever. Chest radiography now showed pleural effusions. Her fingers had color changes consistent with Raynaud phenomenon. At that time, I diagnosed an undifferentiated connective tissue disease and told her that I suspected an autoimmune condition that would need time to reveal itself. In the meantime, I treated her empirically with prednisone.
In April, she returned, much more short of breath and with more prominent diffuse pulmonary infiltrates. Physical examination revealed subtle Gottron changes. Testing revealed poor pulmonary function: forced vital capacity (FVC) 56%, forced expiratory volume in 1 second (FEV1) 52%, and diffusing capacity for carbon monoxide (Dlco) 40%. Blood testing was positive for anti-PL-12 antibody, one of the non-Jo-1 antisynthetase antibodies. At this time, we treated her with glucocorticoids and tacrolimus.
More than 15 years later, this patient is doing well. Her skin rash, joint symptoms, and fever have not returned, and interestingly, she never developed myositis. Her Raynaud symptoms are mild. Her most recent pulmonary function test results (January 2018) were FVC 75%, FEV1 87%, and Dlco 78%. Although these results are not normal, they are much improved and allow her to be completely functional without supplemental oxygen. Echocardiography showed normal pulmonary artery systolic pressure (25 mm Hg). She was still taking tacrolimus and prednisone. When we tried to stop tacrolimus after she had done well for many years, her condition flared.
Non-Jo-1 antisynthetase syndrome: Bottom line
Patients with a non-Jo-1 antisynthetase syndrome often present without myositis symptoms and may never manifest myositis symptoms. Likely because of this presentation, diagnosis of a specific connective tissue disorder is delayed, perhaps leading to increased mortality risk from pulmonary disease. Chronic immunosuppression is often required for these autoimmune conditions.
CASE 6: DERMATOMYOSITIS, RAPIDLY PROGRESSIVE INTERSTITIAL LUNG DISEASE
A 58-year-old woman presented in the summer of 2012 with a photosensitive rash. The following January, she returned with polyarthritis, mild muscle weakness, and a dermatomyositis-pattern rash. Her CK level was normal, and her antinuclear antibody and Sjögren syndrome antibody test results were negative. She improved on low-dose prednisone and methotrexate.
She was originally referred to me in May of that year for worsening rash and mild weakness. She denied pulmonary symptoms, but examination revealed faint basilar crackles. I increased her prednisone dosage to 20 mg/day and started mycophenolate mofetil mainly for the mild cutaneous and myositis features. I also recommended high-resolution CT of the lungs and pulmonary function tests, which she underwent in early June. High-resolution CT showed nonspecific mild infiltrates with minimal ground-glass opacities.
On July 1, she presented to her local emergency department with severe shortness of breath, requiring oxygen 12 L/min. She had a palmar rash. Repeat high-resolution CT showed dramatic worsening compared with the scan the previous month. Because of continued inadequate oxygenation, she was transferred to our center. A blood test later was positive for antimelanoma differentiation-associated gene 5 (MDA-5) autoantibody, previously known as anticlinically amyopathic dermatomyositis (anti-CADM)-140 antibody (based on immunoprecipitation results).
She died on the third day after transfer, just 2 months after I had originally seen her, at which time she had had no pulmonary symptoms.
Clinically amyopathic dermatomyositis
Anti-CADM-140, first reported from Asia,18–20 is an autoantibody-associated disease but not an antisynthetase. It is associated with dermatomyositis; patients often have a “vasculopathy” with cutaneous ulcerations and palmar papules.
MDA-5 is a cytoplasmic protein that “senses” viral RNA and induces production of type 1 interferon. It is involved in the innate immune defense against viruses.
Anti-MDA-5 positivity is associated with a poor pulmonary outcome.21 In our cohort from the University of Pittsburgh, many patients died within 3 years, compared with about a 40% survival rate in patients with dermatomyositis who tested negative for this antibody. That being said, many patients with anti-MDA-5 do not develop rapidly progressive interstitial lung disease.
Autoimmune interstitial lung disease: Bottom line
Autoimmune interstitial lung disease is easy to miss, especially in the case of a non-Jo-1 syndrome, for 3 important reasons:
- The autoimmune features may initially be subtle (eg, Raynaud phenomena, mild dermatomyositis rash, undifferentiated connective tissue disease)
- Autoantibody testing is not often ordered, is not standardized, or may be unavailable
- Providers are mistakenly reassured that a patient who tests negative for antinuclear antibody does not have an autoimmune condition.
To emphasize the last point, in a cohort of 202 patients who tested positive for an antisynthetase antibody, only half were antinuclear antibody-positive, but nearly three-quarters demonstrated anticytoplasmic staining on indirect immunofluorescence (due to the location of the autoantigen in the cytoplasm), making the latter a better screening test for an antisynthetase antibody. For scleroderma, 99% were antinculear antibody-positive, but for myositis, this test is much less sensitive.22
- Felice KJ, North WA. Inclusion body myositis in Connecticut: observations in 35 patients during an 8-year period. Medicine (Baltimore) 2001; 80(5):320–327. doi:10.1097/00005792-200109000-00006
- Lev EI, Tur-Kaspa I, Ashkenazy I, et al. Distribution of serum creatine kinase activity in young healthy persons. Clin Chim Acta 1999; 279(1-2):107–115. doi:10.1016/S0009-8981(98)00180-6
- Lilleng H, Abeler K, Johnsen SH, et al. Variation of serum creatine kinase (CK) levels and prevalence of persistent hyperCKemia in a Norwegian normal population. The Tromsø Study. Neuromuscul Disord 2011; 21(7):494–500. doi:10.1016/j.nmd.2011.04.007
- Johnston JD, Lloyd M, Mathews JA, Hawthorne SW. Racial variation in serum creatine kinase levels. J R Soc Med 1996; 89(8):462-464. pmid:8795501
- Prelle A, Tancredi L, Sciacco M, et al. Retrospective study of a large population of patients with asymptomatic or minimally symptomatic raised serum creatine kinase levels. J Neurol 2002; 249(3):305–311. pmid:11993531
- D’Adda E, Sciacco M, Fruguglietti ME, et al. Follow-up of a large population of asymptomatic/oligosymptomatic hyperckemic subjects. J Neurol 2006; 253(11):1399–1403. doi:10.1007/s00415-006-0223-y
- Madariaga MG. Polymyositis-like syndrome in hypothyroidism: review of cases reported over the past twenty-five years. Thyroid 2002; 12(4):331–336. doi:10.1089/10507250252949478
- de Sauvage Nolting PR, Buirma RJ, Hutten BA, Kastelein JJ; Dutch ExPRESS Investigator Group. Two-year efficacy and safety of simvastatin 80 mg in familial hypercholesterolemia (the Examination of Probands and Relatives in Statin Studies With Familial Hypercholesterolemia [ExPRESS FH]). Am J Cardiol 2002; 90(2):181–184. doi:10.1016/s0002-9149(02)02449-9
- Bruckert E, Hayem G, Dejager S, Yau C, Bégaud B. Mild to moderate muscular symptoms with high-dosage statin therapy in hyperlipidemic patients--the PRIMO study. Cardiovasc Drugs Ther 2005; 19(6):403–414. doi:10.1007/s10557-005-5686-z
- Mosshammer D, Lorenz G, Meznaric S, Schwarz J, Muche R, Mörike K. Statin use and its association with musculoskeletal symptoms—a cross-sectional study in primary care settings. Fam Pract 2009; 26(2):88–95. doi:10.1093/fampra/cmp006
- Nichols GA, Koro CE. Does statin therapy initiation increase the risk for myopathy? An observational study of 32,225 diabetic and nondiabetic patients. Clin Ther 2007; 29(8):1761–1770. doi:10.1016/j.clinthera.2007.08.022
- Kassardjian CD, Lennon VA, Alfugham NB, Mahler M, Milone M. Clinical features and treatment outcomes of necrotizing autoimmune myopathy. JAMA Neurol 2015; 72(9):996–1003. doi:10.1001/jamaneurol.2015.1207
- Grable-Esposito P, Katzberg HD, Greenberg SA, Srinivasan J, Katz J, Amato AA. Immune-mediated necrotizing myopathy associated with statins. Muscle Nerve 2010; 41(2):185–190. doi:10.1002/mus.21486
- Christopher-Stine L, Casciola-Rosen LA, Hong G, Chung T, Corse AM, Mammen AL. A novel autoantibody recognizing 200-kd and 100-kd proteins is associated with an immune-mediated necrotizing myopathy. Arthritis Rheum 2010; 62(9):2757–2766. doi:10.1002/art.27572
- Mammen AL, Chung T, Christopher-Stine L, et al. Autoantibodies against 3-hydroxy-3-methylglutaryl-coenzyme A reductase in patients with statin-associated autoimmune myopathy. Arthritis Rheum 2011; 63(3):713–721. doi:10.1002/art.30156
- Mammen AL, Tiniakou E. Intravenous immune globulin for statin-triggered autoimmune myopathy. N Engl J Med 2015; 373(17):1680–1682. doi:10.1056/NEJMc1506163
- Aggarwal R, Cassidy E, Fertig N, et al. Patients with non-Jo-1 anti-tRNA-synthetase autoantibodies have worse survival than Jo-1 positive patients. Ann Rheum Dis 2014; 73(1):227–232. doi:10.1136/annrheumdis-2012-201800
- Sato S, Hirakata M, Kuwana M, et al. Autoantibodies to a 140-kd polypeptide, CADM-140, in Japanese patients with clinically amyopathic dermatomyositis. Arthritis Rheum 2005; 52(5):1571–1576. doi:10.1002/art.21023
- Sato S, Hoshino K, Satoh T, et al. RNA helicase encoded by melanoma differentiation-associated gene 5 is a major autoantigen in patients with clinically amyopathic dermatomyositis: association with rapidly progressive interstitial lung disease. Arthritis Rheum 2009; 60(7):2193–2200. doi:10.1002/art.24621
- Chen F, Wang D, Shu X, Nakashima R, Wang G. Anti-MDA5 antibody is associated with A/SIP and decreased T cells in peripheral blood and predicts poor prognosis of ILD in Chinese patients with dermatomyositis. Rheumatol Int 2012; 32(12):3909–3915. doi:10.1007/s00296-011-2323-y
- Moghadam-Kia S, Oddis CV, Sato S, Kuwana M, Aggarwal R. Anti-melanoma differentiation-associated gene 5 is associated with rapidly progressive lung disease and poor survival in US patients with amyopathic and myopathic dermatomyositis. Arthritis Care Res (Hoboken) 2016; 68(5):689–694. doi:10.1002/acr.22728
- Aggarwal R, Dhillon N, Fertig N, Koontz D, Qi Z, Oddis CV. A negative antinuclear antibody does not indicate autoantibody negativity in myositis: role of anticytoplasmic antibody as a screening test for antisynthetase syndrome. J Rheumatol 2017; 44(2):223–229. doi:10.3899/jrheum.160618
- Felice KJ, North WA. Inclusion body myositis in Connecticut: observations in 35 patients during an 8-year period. Medicine (Baltimore) 2001; 80(5):320–327. doi:10.1097/00005792-200109000-00006
- Lev EI, Tur-Kaspa I, Ashkenazy I, et al. Distribution of serum creatine kinase activity in young healthy persons. Clin Chim Acta 1999; 279(1-2):107–115. doi:10.1016/S0009-8981(98)00180-6
- Lilleng H, Abeler K, Johnsen SH, et al. Variation of serum creatine kinase (CK) levels and prevalence of persistent hyperCKemia in a Norwegian normal population. The Tromsø Study. Neuromuscul Disord 2011; 21(7):494–500. doi:10.1016/j.nmd.2011.04.007
- Johnston JD, Lloyd M, Mathews JA, Hawthorne SW. Racial variation in serum creatine kinase levels. J R Soc Med 1996; 89(8):462-464. pmid:8795501
- Prelle A, Tancredi L, Sciacco M, et al. Retrospective study of a large population of patients with asymptomatic or minimally symptomatic raised serum creatine kinase levels. J Neurol 2002; 249(3):305–311. pmid:11993531
- D’Adda E, Sciacco M, Fruguglietti ME, et al. Follow-up of a large population of asymptomatic/oligosymptomatic hyperckemic subjects. J Neurol 2006; 253(11):1399–1403. doi:10.1007/s00415-006-0223-y
- Madariaga MG. Polymyositis-like syndrome in hypothyroidism: review of cases reported over the past twenty-five years. Thyroid 2002; 12(4):331–336. doi:10.1089/10507250252949478
- de Sauvage Nolting PR, Buirma RJ, Hutten BA, Kastelein JJ; Dutch ExPRESS Investigator Group. Two-year efficacy and safety of simvastatin 80 mg in familial hypercholesterolemia (the Examination of Probands and Relatives in Statin Studies With Familial Hypercholesterolemia [ExPRESS FH]). Am J Cardiol 2002; 90(2):181–184. doi:10.1016/s0002-9149(02)02449-9
- Bruckert E, Hayem G, Dejager S, Yau C, Bégaud B. Mild to moderate muscular symptoms with high-dosage statin therapy in hyperlipidemic patients--the PRIMO study. Cardiovasc Drugs Ther 2005; 19(6):403–414. doi:10.1007/s10557-005-5686-z
- Mosshammer D, Lorenz G, Meznaric S, Schwarz J, Muche R, Mörike K. Statin use and its association with musculoskeletal symptoms—a cross-sectional study in primary care settings. Fam Pract 2009; 26(2):88–95. doi:10.1093/fampra/cmp006
- Nichols GA, Koro CE. Does statin therapy initiation increase the risk for myopathy? An observational study of 32,225 diabetic and nondiabetic patients. Clin Ther 2007; 29(8):1761–1770. doi:10.1016/j.clinthera.2007.08.022
- Kassardjian CD, Lennon VA, Alfugham NB, Mahler M, Milone M. Clinical features and treatment outcomes of necrotizing autoimmune myopathy. JAMA Neurol 2015; 72(9):996–1003. doi:10.1001/jamaneurol.2015.1207
- Grable-Esposito P, Katzberg HD, Greenberg SA, Srinivasan J, Katz J, Amato AA. Immune-mediated necrotizing myopathy associated with statins. Muscle Nerve 2010; 41(2):185–190. doi:10.1002/mus.21486
- Christopher-Stine L, Casciola-Rosen LA, Hong G, Chung T, Corse AM, Mammen AL. A novel autoantibody recognizing 200-kd and 100-kd proteins is associated with an immune-mediated necrotizing myopathy. Arthritis Rheum 2010; 62(9):2757–2766. doi:10.1002/art.27572
- Mammen AL, Chung T, Christopher-Stine L, et al. Autoantibodies against 3-hydroxy-3-methylglutaryl-coenzyme A reductase in patients with statin-associated autoimmune myopathy. Arthritis Rheum 2011; 63(3):713–721. doi:10.1002/art.30156
- Mammen AL, Tiniakou E. Intravenous immune globulin for statin-triggered autoimmune myopathy. N Engl J Med 2015; 373(17):1680–1682. doi:10.1056/NEJMc1506163
- Aggarwal R, Cassidy E, Fertig N, et al. Patients with non-Jo-1 anti-tRNA-synthetase autoantibodies have worse survival than Jo-1 positive patients. Ann Rheum Dis 2014; 73(1):227–232. doi:10.1136/annrheumdis-2012-201800
- Sato S, Hirakata M, Kuwana M, et al. Autoantibodies to a 140-kd polypeptide, CADM-140, in Japanese patients with clinically amyopathic dermatomyositis. Arthritis Rheum 2005; 52(5):1571–1576. doi:10.1002/art.21023
- Sato S, Hoshino K, Satoh T, et al. RNA helicase encoded by melanoma differentiation-associated gene 5 is a major autoantigen in patients with clinically amyopathic dermatomyositis: association with rapidly progressive interstitial lung disease. Arthritis Rheum 2009; 60(7):2193–2200. doi:10.1002/art.24621
- Chen F, Wang D, Shu X, Nakashima R, Wang G. Anti-MDA5 antibody is associated with A/SIP and decreased T cells in peripheral blood and predicts poor prognosis of ILD in Chinese patients with dermatomyositis. Rheumatol Int 2012; 32(12):3909–3915. doi:10.1007/s00296-011-2323-y
- Moghadam-Kia S, Oddis CV, Sato S, Kuwana M, Aggarwal R. Anti-melanoma differentiation-associated gene 5 is associated with rapidly progressive lung disease and poor survival in US patients with amyopathic and myopathic dermatomyositis. Arthritis Care Res (Hoboken) 2016; 68(5):689–694. doi:10.1002/acr.22728
- Aggarwal R, Dhillon N, Fertig N, Koontz D, Qi Z, Oddis CV. A negative antinuclear antibody does not indicate autoantibody negativity in myositis: role of anticytoplasmic antibody as a screening test for antisynthetase syndrome. J Rheumatol 2017; 44(2):223–229. doi:10.3899/jrheum.160618
KEY POINTS
- Inclusion body myositis affects older men more than women and is characterized by slowly progressive, asymmetric, distal and proximal weakness and atrophy.
- Statin-associated muscle complaints are common, whereas necrotizing myopathy, characterized by a very high CK plus weakness, is rare but must be recognized.
- Elevated CK does not necessarily indicate myositis, especially in African Americans or after heavy exercise.
- Dermatomyositis is characterized by muscle weakness and raised red or purple Gottron papules over the knuckles, elbows, or knees.
- Autoimmune interstitial lung disease may be caused by a variety of antibodies, the most common being anti-Jo-1 (directed against histidyl tRNA synthetase).
- The rarer non-Jo-1 antisynthetase autoantibodies may be associated with rapidly progressive interstitial lung disease, which is a challenge to recognize because associated rheumatologic symptoms may be minimal.
Cannabis-using MS patients improve cognition with 28 days of abstinence
STOCKHOLM – The good news about cognitive impairment in patients with multiple sclerosis who’ve been using cannabis heavily for symptom relief – even for many years – is that their memory, executive function, and information processing speed will improve significantly once they’ve been off the drug for just 28 days, according to the results of a randomized trial presented at the annual congress of the European Committee for Treatment and Research in Multiple Sclerosis.
“It’s good for neurologists to know that, if they prescribe cannabis or their patient is self-medicating and chooses to stop, their cognition will improve considerably,” observed Cecilia Meza, a coinvestigator in the study led by Anthony Feinstein, MD, professor of psychiatry at the University of Toronto.
But there’s a surprise twist to this study, she explained in an interview: “We showed patients their results, and they also felt that their cognition was doing a lot better, but despite that, they would rather be using cannabis to feel better than to have their memory intact. The pain was that bad,” said Ms. Meza, a research coordinator at the university’s Sunnybrook Research Institute.
It’s known that cognitive impairment in healthy long-term cannabis users, provided they started as adults, is fully reversed after 28 days of abstinence. But disease-related cognitive dysfunction affects 40%-80% of patients with MS, and cannabis use may compound this impairment.
The study included 40 MS patients with global impairment of cognition, none of whom were cannabis users prior to their diagnosis. They typically started using it for MS symptom relief 2-3 years after receiving their diagnosis. By the time they were approached for study participation, they had been using cannabis four to five times per day or more for an average of 7 years for relief of symptoms, including incontinence, spasticity, poor sleep, headaches, and difficulties in eating.
All participants were willing to try 28 days of abstinence; half were randomized to do so, while the others stayed the course. Study endpoints included change from baseline to day 28 in the Brief Repeatable Neuropsychological Battery, functional MRI done while taking the Symbol Digit Modalities Test, and urine testing to assure compliance with abstinence.
By day 28, the abstinence group – and with one exception, urine testing confirmed they were bona fide cannabis quitters for the study duration – performed significantly better on the neuropsychological test battery than at baseline, with an associated significant increase in brain activation in the bilateral inferior frontal gyri, as well as the caudate and declive cerebellum while executing the Symbol Digit Modalities Test. The control group who kept on using cannabis showed no such improvements.
The full study details were published in conjunction with Ms. Meza’s presentation (Brain. 2019 Sep 1;142[9]:2800-12).
She reported having no financial conflicts regarding the study, funded by the Multiple Sclerosis Society of Canada.
SOURCE: Meza C. ECTRIMS 2019, Abstract P542.
STOCKHOLM – The good news about cognitive impairment in patients with multiple sclerosis who’ve been using cannabis heavily for symptom relief – even for many years – is that their memory, executive function, and information processing speed will improve significantly once they’ve been off the drug for just 28 days, according to the results of a randomized trial presented at the annual congress of the European Committee for Treatment and Research in Multiple Sclerosis.
“It’s good for neurologists to know that, if they prescribe cannabis or their patient is self-medicating and chooses to stop, their cognition will improve considerably,” observed Cecilia Meza, a coinvestigator in the study led by Anthony Feinstein, MD, professor of psychiatry at the University of Toronto.
But there’s a surprise twist to this study, she explained in an interview: “We showed patients their results, and they also felt that their cognition was doing a lot better, but despite that, they would rather be using cannabis to feel better than to have their memory intact. The pain was that bad,” said Ms. Meza, a research coordinator at the university’s Sunnybrook Research Institute.
It’s known that cognitive impairment in healthy long-term cannabis users, provided they started as adults, is fully reversed after 28 days of abstinence. But disease-related cognitive dysfunction affects 40%-80% of patients with MS, and cannabis use may compound this impairment.
The study included 40 MS patients with global impairment of cognition, none of whom were cannabis users prior to their diagnosis. They typically started using it for MS symptom relief 2-3 years after receiving their diagnosis. By the time they were approached for study participation, they had been using cannabis four to five times per day or more for an average of 7 years for relief of symptoms, including incontinence, spasticity, poor sleep, headaches, and difficulties in eating.
All participants were willing to try 28 days of abstinence; half were randomized to do so, while the others stayed the course. Study endpoints included change from baseline to day 28 in the Brief Repeatable Neuropsychological Battery, functional MRI done while taking the Symbol Digit Modalities Test, and urine testing to assure compliance with abstinence.
By day 28, the abstinence group – and with one exception, urine testing confirmed they were bona fide cannabis quitters for the study duration – performed significantly better on the neuropsychological test battery than at baseline, with an associated significant increase in brain activation in the bilateral inferior frontal gyri, as well as the caudate and declive cerebellum while executing the Symbol Digit Modalities Test. The control group who kept on using cannabis showed no such improvements.
The full study details were published in conjunction with Ms. Meza’s presentation (Brain. 2019 Sep 1;142[9]:2800-12).
She reported having no financial conflicts regarding the study, funded by the Multiple Sclerosis Society of Canada.
SOURCE: Meza C. ECTRIMS 2019, Abstract P542.
STOCKHOLM – The good news about cognitive impairment in patients with multiple sclerosis who’ve been using cannabis heavily for symptom relief – even for many years – is that their memory, executive function, and information processing speed will improve significantly once they’ve been off the drug for just 28 days, according to the results of a randomized trial presented at the annual congress of the European Committee for Treatment and Research in Multiple Sclerosis.
“It’s good for neurologists to know that, if they prescribe cannabis or their patient is self-medicating and chooses to stop, their cognition will improve considerably,” observed Cecilia Meza, a coinvestigator in the study led by Anthony Feinstein, MD, professor of psychiatry at the University of Toronto.
But there’s a surprise twist to this study, she explained in an interview: “We showed patients their results, and they also felt that their cognition was doing a lot better, but despite that, they would rather be using cannabis to feel better than to have their memory intact. The pain was that bad,” said Ms. Meza, a research coordinator at the university’s Sunnybrook Research Institute.
It’s known that cognitive impairment in healthy long-term cannabis users, provided they started as adults, is fully reversed after 28 days of abstinence. But disease-related cognitive dysfunction affects 40%-80% of patients with MS, and cannabis use may compound this impairment.
The study included 40 MS patients with global impairment of cognition, none of whom were cannabis users prior to their diagnosis. They typically started using it for MS symptom relief 2-3 years after receiving their diagnosis. By the time they were approached for study participation, they had been using cannabis four to five times per day or more for an average of 7 years for relief of symptoms, including incontinence, spasticity, poor sleep, headaches, and difficulties in eating.
All participants were willing to try 28 days of abstinence; half were randomized to do so, while the others stayed the course. Study endpoints included change from baseline to day 28 in the Brief Repeatable Neuropsychological Battery, functional MRI done while taking the Symbol Digit Modalities Test, and urine testing to assure compliance with abstinence.
By day 28, the abstinence group – and with one exception, urine testing confirmed they were bona fide cannabis quitters for the study duration – performed significantly better on the neuropsychological test battery than at baseline, with an associated significant increase in brain activation in the bilateral inferior frontal gyri, as well as the caudate and declive cerebellum while executing the Symbol Digit Modalities Test. The control group who kept on using cannabis showed no such improvements.
The full study details were published in conjunction with Ms. Meza’s presentation (Brain. 2019 Sep 1;142[9]:2800-12).
She reported having no financial conflicts regarding the study, funded by the Multiple Sclerosis Society of Canada.
SOURCE: Meza C. ECTRIMS 2019, Abstract P542.
REPORTING FROM ECTRIMS 2019
How does alcohol intake affect dementia risk in older adults?
Mild cognitive impairment (MCI) may influence the relationship between alcohol consumption and dementia risk, a study of more than 3,000 adults suggests. In addition, , according to the study, which was published in JAMA Network Open.
“The associations of self-reported alcohol consumption with dementia risk and cognitive decline were more consistently adverse among individuals with MCI than those with normal cognition,” reported Manja Koch, PhD, a researcher in the department of nutrition at Harvard T.H. Chan School of Public Health in Boston and colleagues. “This was particularly true for the subset of individuals [with MCI] who drank more than 14.0 servings per week, whose rate of cognitive decline and risk of dementia were the highest of any subgroup.”
Among older adults with normal cognition, the results generally were consistent with those of a recent meta-analysis that found a U-shaped relationship between drinking and dementia, the researchers said (Eur J Epidemiol. 2017 Jan;32[1]:31-42.).
“Our results did not show significant associations and clearly do not suffice to suggest a clinical benefit from even limited alcohol use,” said Dr. Koch and colleagues. “Nonetheless, our findings provide some reassurance that alcohol consumed within recommended limits was not associated with an increased risk of dementia among older adults with normal baseline cognition.”
GEMS data
To study whether alcohol consumption is associated with the risk of dementia and cognitive decline in older adults with and without MCI, the investigators analyzed data from the Ginkgo Evaluation of Memory Study (GEMS). GEMS was a randomized controlled trial conducted between 2000 and 2008 that found no overall association between ginkgo biloba and dementia prevention. During the trial, participants completed the Modified Mini-Mental State Examination, the Clinical Dementia Rating scale, and the cognitive portion of the Alzheimer’s Disease Assessment Scale.
In the present study, the investigators analyzed data from 3,021 participants aged 72 years and older who were free of dementia at baseline and had provided information about their alcohol intake. Their median age was 78 years, and 46.2% were female. Fifty-eight percent consumed alcohol, including 45% of the participants with MCI at baseline.
During follow-up, 512 cases of dementia occurred. Among the 473 participants with MCI at baseline, the adjusted hazard ratio (HR) for dementia was 1.72 for those who consumed more than 14 drinks per week, compared with light drinkers who consumed less than 1 drink per week. For participants who consumed between 7 and 14 drinks per week, the adjusted HR for dementia was 0.63 among those without MCI and 0.93 among those with MCI, relative to light drinkers who consumed less than 1 drink per week.
Among adults with normal cognition at baseline, daily low-quantity drinking was associated with lower dementia risk, compared with infrequent higher-quantity drinking (HR, 0.45).
Trial excluded adults with excessive alcohol use
Limitations of the study include a lack of data about any changes in alcohol consumption over time. In addition, the original trial excluded people with a known history of excessive alcohol use. Furthermore, it is possible that the “long preclinical phase of dementia” and other health issues affect drinking behavior, the authors said. “At present, our findings cannot be directly translated into clinical recommendations,” the authors said. Nevertheless, the results “suggest that, while caring for older adults, physicians should carefully assess the full dimensions of drinking behavior and cognition when providing guidance to patients about alcohol consumption,” they said.
The study was supported by grants from the National Center for Complementary and Alternative Medicine; the National Institute of Neurological Disorders and Stroke; the Office of Dietary Supplements of the National Institute on Aging; the National Heart, Lung, and Blood Institute; the University of Pittsburgh Alzheimer’s Disease Research Center; the Roena Kulynych Center for Memory and Cognition Research; and Wake Forest University School of Medicine. In addition, the researchers used plasma samples from the National Cell Repository for Alzheimer’s Disease, which receives support from the National Institute on Aging. Dr. Koch had no conflicts of interest. Coauthors disclosed university and government grants and personal fees from pharmaceutical companies outside the study. One author was an employee of Genentech at the time of publication, but Genentech did not contribute to the study.
SOURCE: Koch M et al. JAMA Network Open. 2019 Sep 27. doi: 10.1001/jamanetworkopen.2019.10319.
Mild cognitive impairment (MCI) may influence the relationship between alcohol consumption and dementia risk, a study of more than 3,000 adults suggests. In addition, , according to the study, which was published in JAMA Network Open.
“The associations of self-reported alcohol consumption with dementia risk and cognitive decline were more consistently adverse among individuals with MCI than those with normal cognition,” reported Manja Koch, PhD, a researcher in the department of nutrition at Harvard T.H. Chan School of Public Health in Boston and colleagues. “This was particularly true for the subset of individuals [with MCI] who drank more than 14.0 servings per week, whose rate of cognitive decline and risk of dementia were the highest of any subgroup.”
Among older adults with normal cognition, the results generally were consistent with those of a recent meta-analysis that found a U-shaped relationship between drinking and dementia, the researchers said (Eur J Epidemiol. 2017 Jan;32[1]:31-42.).
“Our results did not show significant associations and clearly do not suffice to suggest a clinical benefit from even limited alcohol use,” said Dr. Koch and colleagues. “Nonetheless, our findings provide some reassurance that alcohol consumed within recommended limits was not associated with an increased risk of dementia among older adults with normal baseline cognition.”
GEMS data
To study whether alcohol consumption is associated with the risk of dementia and cognitive decline in older adults with and without MCI, the investigators analyzed data from the Ginkgo Evaluation of Memory Study (GEMS). GEMS was a randomized controlled trial conducted between 2000 and 2008 that found no overall association between ginkgo biloba and dementia prevention. During the trial, participants completed the Modified Mini-Mental State Examination, the Clinical Dementia Rating scale, and the cognitive portion of the Alzheimer’s Disease Assessment Scale.
In the present study, the investigators analyzed data from 3,021 participants aged 72 years and older who were free of dementia at baseline and had provided information about their alcohol intake. Their median age was 78 years, and 46.2% were female. Fifty-eight percent consumed alcohol, including 45% of the participants with MCI at baseline.
During follow-up, 512 cases of dementia occurred. Among the 473 participants with MCI at baseline, the adjusted hazard ratio (HR) for dementia was 1.72 for those who consumed more than 14 drinks per week, compared with light drinkers who consumed less than 1 drink per week. For participants who consumed between 7 and 14 drinks per week, the adjusted HR for dementia was 0.63 among those without MCI and 0.93 among those with MCI, relative to light drinkers who consumed less than 1 drink per week.
Among adults with normal cognition at baseline, daily low-quantity drinking was associated with lower dementia risk, compared with infrequent higher-quantity drinking (HR, 0.45).
Trial excluded adults with excessive alcohol use
Limitations of the study include a lack of data about any changes in alcohol consumption over time. In addition, the original trial excluded people with a known history of excessive alcohol use. Furthermore, it is possible that the “long preclinical phase of dementia” and other health issues affect drinking behavior, the authors said. “At present, our findings cannot be directly translated into clinical recommendations,” the authors said. Nevertheless, the results “suggest that, while caring for older adults, physicians should carefully assess the full dimensions of drinking behavior and cognition when providing guidance to patients about alcohol consumption,” they said.
The study was supported by grants from the National Center for Complementary and Alternative Medicine; the National Institute of Neurological Disorders and Stroke; the Office of Dietary Supplements of the National Institute on Aging; the National Heart, Lung, and Blood Institute; the University of Pittsburgh Alzheimer’s Disease Research Center; the Roena Kulynych Center for Memory and Cognition Research; and Wake Forest University School of Medicine. In addition, the researchers used plasma samples from the National Cell Repository for Alzheimer’s Disease, which receives support from the National Institute on Aging. Dr. Koch had no conflicts of interest. Coauthors disclosed university and government grants and personal fees from pharmaceutical companies outside the study. One author was an employee of Genentech at the time of publication, but Genentech did not contribute to the study.
SOURCE: Koch M et al. JAMA Network Open. 2019 Sep 27. doi: 10.1001/jamanetworkopen.2019.10319.
Mild cognitive impairment (MCI) may influence the relationship between alcohol consumption and dementia risk, a study of more than 3,000 adults suggests. In addition, , according to the study, which was published in JAMA Network Open.
“The associations of self-reported alcohol consumption with dementia risk and cognitive decline were more consistently adverse among individuals with MCI than those with normal cognition,” reported Manja Koch, PhD, a researcher in the department of nutrition at Harvard T.H. Chan School of Public Health in Boston and colleagues. “This was particularly true for the subset of individuals [with MCI] who drank more than 14.0 servings per week, whose rate of cognitive decline and risk of dementia were the highest of any subgroup.”
Among older adults with normal cognition, the results generally were consistent with those of a recent meta-analysis that found a U-shaped relationship between drinking and dementia, the researchers said (Eur J Epidemiol. 2017 Jan;32[1]:31-42.).
“Our results did not show significant associations and clearly do not suffice to suggest a clinical benefit from even limited alcohol use,” said Dr. Koch and colleagues. “Nonetheless, our findings provide some reassurance that alcohol consumed within recommended limits was not associated with an increased risk of dementia among older adults with normal baseline cognition.”
GEMS data
To study whether alcohol consumption is associated with the risk of dementia and cognitive decline in older adults with and without MCI, the investigators analyzed data from the Ginkgo Evaluation of Memory Study (GEMS). GEMS was a randomized controlled trial conducted between 2000 and 2008 that found no overall association between ginkgo biloba and dementia prevention. During the trial, participants completed the Modified Mini-Mental State Examination, the Clinical Dementia Rating scale, and the cognitive portion of the Alzheimer’s Disease Assessment Scale.
In the present study, the investigators analyzed data from 3,021 participants aged 72 years and older who were free of dementia at baseline and had provided information about their alcohol intake. Their median age was 78 years, and 46.2% were female. Fifty-eight percent consumed alcohol, including 45% of the participants with MCI at baseline.
During follow-up, 512 cases of dementia occurred. Among the 473 participants with MCI at baseline, the adjusted hazard ratio (HR) for dementia was 1.72 for those who consumed more than 14 drinks per week, compared with light drinkers who consumed less than 1 drink per week. For participants who consumed between 7 and 14 drinks per week, the adjusted HR for dementia was 0.63 among those without MCI and 0.93 among those with MCI, relative to light drinkers who consumed less than 1 drink per week.
Among adults with normal cognition at baseline, daily low-quantity drinking was associated with lower dementia risk, compared with infrequent higher-quantity drinking (HR, 0.45).
Trial excluded adults with excessive alcohol use
Limitations of the study include a lack of data about any changes in alcohol consumption over time. In addition, the original trial excluded people with a known history of excessive alcohol use. Furthermore, it is possible that the “long preclinical phase of dementia” and other health issues affect drinking behavior, the authors said. “At present, our findings cannot be directly translated into clinical recommendations,” the authors said. Nevertheless, the results “suggest that, while caring for older adults, physicians should carefully assess the full dimensions of drinking behavior and cognition when providing guidance to patients about alcohol consumption,” they said.
The study was supported by grants from the National Center for Complementary and Alternative Medicine; the National Institute of Neurological Disorders and Stroke; the Office of Dietary Supplements of the National Institute on Aging; the National Heart, Lung, and Blood Institute; the University of Pittsburgh Alzheimer’s Disease Research Center; the Roena Kulynych Center for Memory and Cognition Research; and Wake Forest University School of Medicine. In addition, the researchers used plasma samples from the National Cell Repository for Alzheimer’s Disease, which receives support from the National Institute on Aging. Dr. Koch had no conflicts of interest. Coauthors disclosed university and government grants and personal fees from pharmaceutical companies outside the study. One author was an employee of Genentech at the time of publication, but Genentech did not contribute to the study.
SOURCE: Koch M et al. JAMA Network Open. 2019 Sep 27. doi: 10.1001/jamanetworkopen.2019.10319.
FROM JAMA NETWORK OPEN
FDA expands Dysport’s upper-limb spasticity indication to children
The Food and Drug Administration has expanded the indication of abobotulinumtoxinA (Dysport) for upper-limb spasticity to include patients aged 2 years and older, according to a release from Ipsen. This botulinum toxin product received approval for this indication in adults in 2015 and approval for lower-limb spasticity in patients aged 2 years and older in 2016. Notably, Orphan Drug Exclusivity prevents it from being indicated for patients with cerebral palsy because another botulinum toxin product, onabotulinumtoxinA (Botox), already was approved for the indication in June 2019.
Spasticity affects the muscles and joints of extremities, especially in growing children, and is usually caused by nerve damage, such as head trauma or spinal cord injury. The degree of spasticity can vary from mild muscle stiffness to severe, painful, and uncontrollable muscle spasms.
AbobotulinumtoxinA was evaluated for upper-limb spasticity in a phase 3, randomized, double-blind, low-dose controlled, multicenter study; the study enrolled 210 children aged 2-17 years with the condition and a Modified Ashworth Scale grade 2 or greater for elbow and wrist flexors. The children were randomized 1:1:1 to injections of either 8 units/kg, 16 units/kg, or 2 units/kg into the elbow flexors and wrist flexors. At 6 weeks, there were statistically significant improvements in Modified Ashworth Scale grade, the primary endpoint, with least-square mean changes from baseline of –2.0, –2.3, and –1.6, respectively.
AbobotulinumtoxinA and all other botulinum toxin products carry a boxed warning, the most serious warning the FDA issues. This warning refers to risk of botulism-like symptoms caused by the botulinum toxin spreading away from the injection area; these symptoms can included sometimes life-threatening difficulty swallowing or breathing. AbobotulinumtoxinA is contraindicated in patients with known hypersensitivity to any botulinum toxin or any of the components, those with presence of infection at proposed injection site(s), and those with known allergy to cow’s milk protein. It is also important to note that botulinum toxin preparations are not interchangeable; the potency units of one are not the same as those of another. Full prescribing information can be found on the Ipsen website.
The Food and Drug Administration has expanded the indication of abobotulinumtoxinA (Dysport) for upper-limb spasticity to include patients aged 2 years and older, according to a release from Ipsen. This botulinum toxin product received approval for this indication in adults in 2015 and approval for lower-limb spasticity in patients aged 2 years and older in 2016. Notably, Orphan Drug Exclusivity prevents it from being indicated for patients with cerebral palsy because another botulinum toxin product, onabotulinumtoxinA (Botox), already was approved for the indication in June 2019.
Spasticity affects the muscles and joints of extremities, especially in growing children, and is usually caused by nerve damage, such as head trauma or spinal cord injury. The degree of spasticity can vary from mild muscle stiffness to severe, painful, and uncontrollable muscle spasms.
AbobotulinumtoxinA was evaluated for upper-limb spasticity in a phase 3, randomized, double-blind, low-dose controlled, multicenter study; the study enrolled 210 children aged 2-17 years with the condition and a Modified Ashworth Scale grade 2 or greater for elbow and wrist flexors. The children were randomized 1:1:1 to injections of either 8 units/kg, 16 units/kg, or 2 units/kg into the elbow flexors and wrist flexors. At 6 weeks, there were statistically significant improvements in Modified Ashworth Scale grade, the primary endpoint, with least-square mean changes from baseline of –2.0, –2.3, and –1.6, respectively.
AbobotulinumtoxinA and all other botulinum toxin products carry a boxed warning, the most serious warning the FDA issues. This warning refers to risk of botulism-like symptoms caused by the botulinum toxin spreading away from the injection area; these symptoms can included sometimes life-threatening difficulty swallowing or breathing. AbobotulinumtoxinA is contraindicated in patients with known hypersensitivity to any botulinum toxin or any of the components, those with presence of infection at proposed injection site(s), and those with known allergy to cow’s milk protein. It is also important to note that botulinum toxin preparations are not interchangeable; the potency units of one are not the same as those of another. Full prescribing information can be found on the Ipsen website.
The Food and Drug Administration has expanded the indication of abobotulinumtoxinA (Dysport) for upper-limb spasticity to include patients aged 2 years and older, according to a release from Ipsen. This botulinum toxin product received approval for this indication in adults in 2015 and approval for lower-limb spasticity in patients aged 2 years and older in 2016. Notably, Orphan Drug Exclusivity prevents it from being indicated for patients with cerebral palsy because another botulinum toxin product, onabotulinumtoxinA (Botox), already was approved for the indication in June 2019.
Spasticity affects the muscles and joints of extremities, especially in growing children, and is usually caused by nerve damage, such as head trauma or spinal cord injury. The degree of spasticity can vary from mild muscle stiffness to severe, painful, and uncontrollable muscle spasms.
AbobotulinumtoxinA was evaluated for upper-limb spasticity in a phase 3, randomized, double-blind, low-dose controlled, multicenter study; the study enrolled 210 children aged 2-17 years with the condition and a Modified Ashworth Scale grade 2 or greater for elbow and wrist flexors. The children were randomized 1:1:1 to injections of either 8 units/kg, 16 units/kg, or 2 units/kg into the elbow flexors and wrist flexors. At 6 weeks, there were statistically significant improvements in Modified Ashworth Scale grade, the primary endpoint, with least-square mean changes from baseline of –2.0, –2.3, and –1.6, respectively.
AbobotulinumtoxinA and all other botulinum toxin products carry a boxed warning, the most serious warning the FDA issues. This warning refers to risk of botulism-like symptoms caused by the botulinum toxin spreading away from the injection area; these symptoms can included sometimes life-threatening difficulty swallowing or breathing. AbobotulinumtoxinA is contraindicated in patients with known hypersensitivity to any botulinum toxin or any of the components, those with presence of infection at proposed injection site(s), and those with known allergy to cow’s milk protein. It is also important to note that botulinum toxin preparations are not interchangeable; the potency units of one are not the same as those of another. Full prescribing information can be found on the Ipsen website.
Chronic Subjective Tinnitus
Don’t Take the Fall With Head Injuries
In the early morning hours of June 10, 2009, a 77-year-old man who had been undergoing chemotherapy for multiple myeloma took sleep medication. He then fell down a flight of stairs in his split-level home.
The patient sustained a laceration to his scalp but returned to bed and waited until later that morning to call his internist for an appointment. Later that day, the physician placed 11 sutures for the scalp laceration and performed a neurologic examination; he did not note any abnormalities. The patient complained of back pain, so the physician ordered a back x-ray, which revealed a TI2 fracture that had occurred from the fall. No further treatment was provided for the scalp injury, except removal of the stitches about a week later.
Six days after the fall and doctor visit, the patient’s condition began to deteriorate rapidly, with noted slurred speech and loss of consciousness. He was transported to an emergency department, where CT revealed a massive subdural hematoma. An immediate craniotomy was performed. However, on June 27, 2009, the patient died as a result of the brain bleed.
His estate filed suit against the physician and his practice, alleging medical malpractice and violations in the standard of care. The estate alleged that the standard of care required the physician to obtain a CT scan and that, had one been performed, it would have revealed a small subdural hematoma in time for it to have been successfully treated (ie, before the massive second related bleed). The estate’s theory of the case did not rest on the presentation of clinical symptoms. A medical expert who testified for the estate stated that the subdural hematoma began at the time of the fall.
The defense denied any violations in the standard of care. The physician contended that the patient had presented with no symptoms other than a head laceration, and there were no criteria for ordering CT. Further, the defense asserted that the patient was symptom free for 6 days post-fall. According to the defense, the patient experienced a sudden arterial bleed that was not caused by the fall and would not have been revealed on CT ordered at the time of initial presentation, because it did not occur until 6 days later.
VERDICT
After a 10-day trial and 25 minutes’ deliberation, the jury returned a defense verdict.
COMMENTARY
The 25-minute deliberation suggests that terms such as “bridging veins” and “shearing injury” were unlikely bandied about in the jury room. The jury was likely dismissive of the plaintiff’s claim owing to his cancer diagnosis, and perhaps rightly so. But if we eliminate the multiple myeloma diagnosis, the jury might have decided differently.
Continue to: The defendant physician...
The defendant physician did a good job of documenting a negative neurologic exam, which helped him convince the jury that the patient did not have any signs or symptoms when first evaluated. But in this patient, was imaging to rule out intracranial bleeding indicated?
As an oversimplification, we tend to think of intracranial hemorrhage in 2 varieties: the insidious and the bold. Subdural hematomas are stealthy, they are sneaky, and they prey on the old. They step out of the shadows to cause symptoms. They are the ninjas of intracranial hemorrhage. Beware.
Epidural hematomas and subarachnoid hemorrhage (SAH) are the opposite. They classically present with a sudden and severe symptom complex: with epidural hematoma, the loss of consciousness, lucid interval, and final loss of consciousness; with SAH, the “worst in your life” thunder-clap headache, which may be heralded by a sentinel headache.1 When manifesting this way, they are brash, direct, and unsubtle to the point of being obnoxious—the Steven Stifler of intracranial bleeding.
This generalization is made to highlight the potentially sneaky nature of subdural hemorrhage. There are circumstances in which the clinical presentation of epidural hematoma and SAH will be more challenging. The question here is whether a negative initial neurologic exam can adequately screen for a potentially stealthy subdural hematoma.
Subdural hemorrhage is caused by rapidly changing velocity that may stretch and tear small bridging veins.2,3 Subdural hematoma is more common in the elderly, those who abuse alcohol, and those with a prior history of head trauma.4 As the brain shrinks with age or atrophy, the subdural space enlarges and traversing veins are stretched to cover a wider distance—rendering them vulnerable to rupture.5 These structures may also weaken as a result of low cerebrospinal fluid (intracranial hypotension); as pressure decreases (eg, from a leak), the brain’s buoyancy is reduced, causing traction on anchoring and supporting structures (eg, bridging veins).5 Injury to bridging veins can even occur as a result of a coup-contrecoup mechanism in the absence of direct physical impact.6,7 Bottom line: the injury itself may be subtle, requiring an index of suspicion to make the diagnosis.
Continue to: The case patient was...
The case patient was elderly. He had a chronic malignancy and sustained a fall down the stairs. He was taking sleeping pills, which may have slowed reflexive protective mechanisms after he started to fall (resulting in greater force imparted to his head). Multiple myeloma can predispose a patient to coagulopathy, and we don’t know in this case if this patient’s multiple myeloma made him more susceptible to bleeding—but it certainly didn’t help.8 The patient’s age, the mechanism of injury, and the history of malignancy made this a setup for hemorrhage.
Interestingly, we are not given details about how the patient looked during his suture removal. We are told the time between the initial fall and deterioration was 6 days. Scalp sutures were removed “about a week later,” which was after the deterioration—so this can’t be correct. Removing scalp sutures after 5 days seems premature, but that is the only possibility if 6 days elapsed between the fall and the deterioration.
In short, these are difficult cases. If intracranial bleeding can be subtle and delayed, how can we be sure a patient is not experiencing a bleed? We can only apply the relevant standard of care using all the clinical information we have. The Canadian CT Head Rule and New Orleans Criteria are clinical tools designed to help providers determine when to image (see Table for details).9
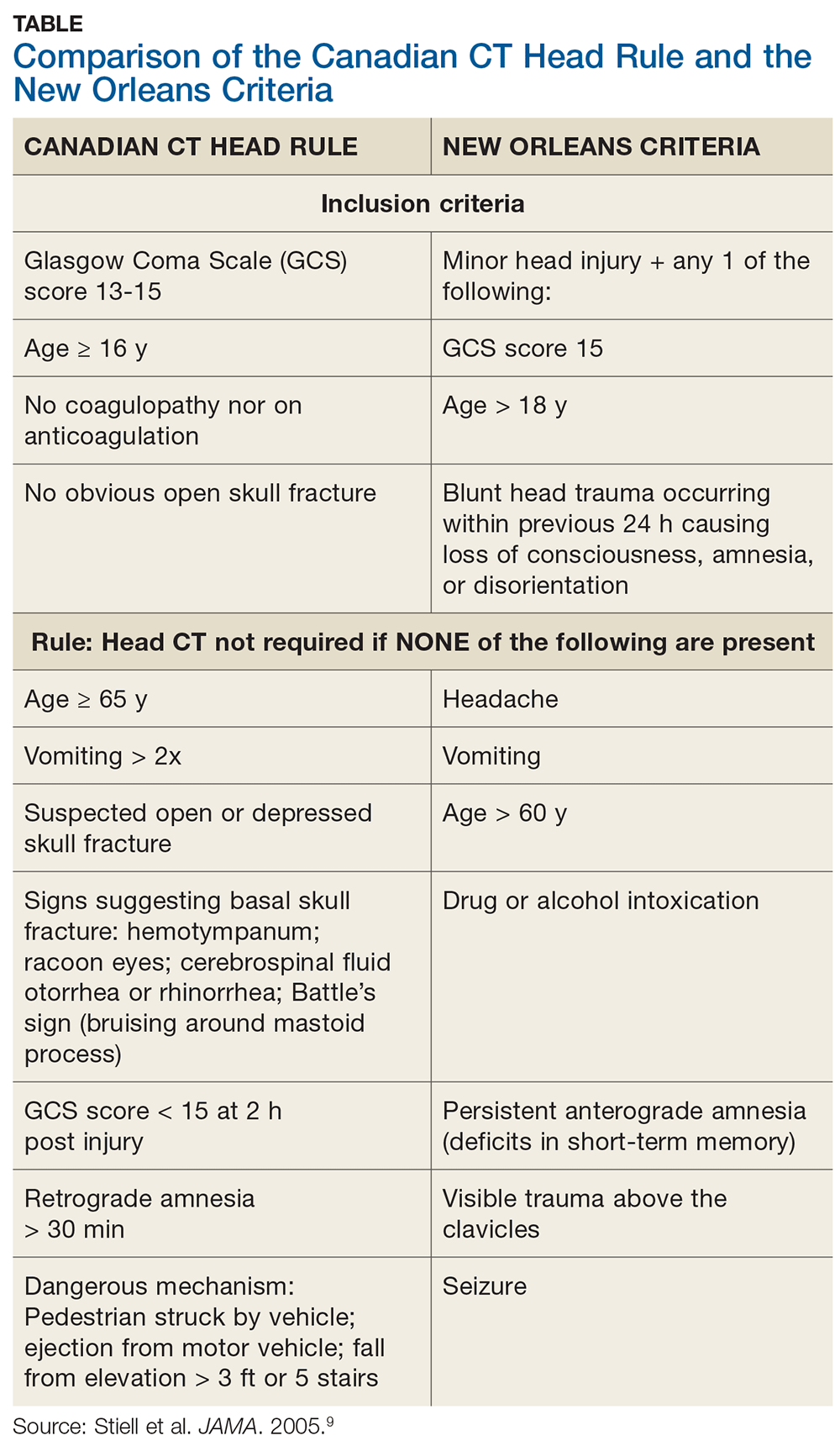
Applying the Canadian CT Head Rule to the facts of this case, we would image the patient because he fell down a “flight” of stairs (which is > 5 stairs) and he is 77 years old (older than 65). The New Orleans Criteria require head CT for minor injury with any positive findings.9 Because the patient is older than 60, he would be scanned according this rule.
In this case, the tools indicate scanning would have been appropriate. The patient’s multiple myeloma might have further impelled a decision to image. However, the jury was persuaded that the defendant’s negative neurologic exam was reasonable under the circumstances. This was likely made possible by the physician’s good recordkeeping and demonstrated genuine concern for the patient’s well-being—as well as a differing viewpoint of the patient’s age and health status.
Continue to: Finally, a word about...
Finally, a word about falls and the elderly: We’ve all heard the 80s advertising catchphrase (which lives on as a present-day meme) “I’ve fallen, and I can’t get up!” The problem is, many don’t. It would be more clinically accurate to say, “I’ve fallen, and I’ll be hospitalized for an extended period of time, then transferred to a skilled nursing facility, but I won’t survive to discharge.” The reality is that falls kill, and the severity is underestimated.10 If it were a “brain-eating amoeba,” the media would be all over it. With falls, not so much. We tend to pay less attention.
Risk factors for a fall include postural hypotension; use of benzodiazepines or other sedative-hypnotic drugs; use of ≥ 4 medications; environmental hazards for tripping; impairment in balance and transfer skills; and gait impairment.11 Home setup also contributes—loose throw rugs, uneven carpet edges, cracked sidewalks, clutter and furniture, cables and wires and cords, oh my.
Do your older patients a favor by reinforcing fall risk. Instruct them to rise slowly from seated or recumbent positions; always consider central nervous system sedation and/or the coordination-hampering properties of medications, particularly in combination. Raise the issue of home safety. A brief 10-second comment from you may plant a seed in a family member’s head to do what you cannot: scan and make safe the patient’s living environment.
1. de Falco FA. Sentinel headache. Neurol Sci. 2004;25(suppl 3):S215-S217.
2. Miller JD, Nader R. Acute subdural hematoma from bridging vein rupture: a potential mechanism for growth. J Neurosurg. 2014;120(6):1378-1384.
3. Victor M, Ropper A. Craniocerebral trauma. In: Victor M, Ropper A, eds. Adams and Victor’s Principles of Neurology. 7th ed. New York, NY: McGraw-Hill; 2001:925.
4. McBridde W. Subdural hematoma in adults: etiology, clinical features, and diagnosis. UpToDate website. www.uptodate.com/contents/subdural-hematoma-in-adults-etiology-clinical-features-and-diagnosis? search=subdural%20hematoma. Published December 10, 2018. Accessed September 23, 2019.
5. US National Library of Medicine. Subdural hematoma. Medline Plus website. https://medlineplus.gov/ency/article/000713.htm. Accessed September 23, 2019.
6. Besenski N. Traumatic injuries: imaging of head injuries. Eur Radiol. 2002;12(6):1237-1252.
7. Mayer S, Rowland L. Head injury. In: Rowland L, ed. Merritt’s Neurology. Philadelphia, PA: Lippincott Williams & Wilkins; 2000:401.
8. Saif MW, Allegra CJ, Greenberg B. Bleeding diathesis in multiple myeloma. J Hematother Stem Cell Res. 2001;10(5):657-660.
9. Stiell IG, Clement CM, Rowe BH, et al. Comparison of the Canadian CT Head Rule and the New Orleans Criteria in patients with minor head injury. JAMA. 2005;294(12):1511-1518.
10. Abdelrahman H, Almadani A, El-Menyar A, et al. Home-related falls: an underestimated mechanism of injury. J Family Community Med. 2018; 25(1):48-51.
11. Fuller GF. Falls in the elderly. Am Fam Physician. 2000;61(7):2159-2168.
In the early morning hours of June 10, 2009, a 77-year-old man who had been undergoing chemotherapy for multiple myeloma took sleep medication. He then fell down a flight of stairs in his split-level home.
The patient sustained a laceration to his scalp but returned to bed and waited until later that morning to call his internist for an appointment. Later that day, the physician placed 11 sutures for the scalp laceration and performed a neurologic examination; he did not note any abnormalities. The patient complained of back pain, so the physician ordered a back x-ray, which revealed a TI2 fracture that had occurred from the fall. No further treatment was provided for the scalp injury, except removal of the stitches about a week later.
Six days after the fall and doctor visit, the patient’s condition began to deteriorate rapidly, with noted slurred speech and loss of consciousness. He was transported to an emergency department, where CT revealed a massive subdural hematoma. An immediate craniotomy was performed. However, on June 27, 2009, the patient died as a result of the brain bleed.
His estate filed suit against the physician and his practice, alleging medical malpractice and violations in the standard of care. The estate alleged that the standard of care required the physician to obtain a CT scan and that, had one been performed, it would have revealed a small subdural hematoma in time for it to have been successfully treated (ie, before the massive second related bleed). The estate’s theory of the case did not rest on the presentation of clinical symptoms. A medical expert who testified for the estate stated that the subdural hematoma began at the time of the fall.
The defense denied any violations in the standard of care. The physician contended that the patient had presented with no symptoms other than a head laceration, and there were no criteria for ordering CT. Further, the defense asserted that the patient was symptom free for 6 days post-fall. According to the defense, the patient experienced a sudden arterial bleed that was not caused by the fall and would not have been revealed on CT ordered at the time of initial presentation, because it did not occur until 6 days later.
VERDICT
After a 10-day trial and 25 minutes’ deliberation, the jury returned a defense verdict.
COMMENTARY
The 25-minute deliberation suggests that terms such as “bridging veins” and “shearing injury” were unlikely bandied about in the jury room. The jury was likely dismissive of the plaintiff’s claim owing to his cancer diagnosis, and perhaps rightly so. But if we eliminate the multiple myeloma diagnosis, the jury might have decided differently.
Continue to: The defendant physician...
The defendant physician did a good job of documenting a negative neurologic exam, which helped him convince the jury that the patient did not have any signs or symptoms when first evaluated. But in this patient, was imaging to rule out intracranial bleeding indicated?
As an oversimplification, we tend to think of intracranial hemorrhage in 2 varieties: the insidious and the bold. Subdural hematomas are stealthy, they are sneaky, and they prey on the old. They step out of the shadows to cause symptoms. They are the ninjas of intracranial hemorrhage. Beware.
Epidural hematomas and subarachnoid hemorrhage (SAH) are the opposite. They classically present with a sudden and severe symptom complex: with epidural hematoma, the loss of consciousness, lucid interval, and final loss of consciousness; with SAH, the “worst in your life” thunder-clap headache, which may be heralded by a sentinel headache.1 When manifesting this way, they are brash, direct, and unsubtle to the point of being obnoxious—the Steven Stifler of intracranial bleeding.
This generalization is made to highlight the potentially sneaky nature of subdural hemorrhage. There are circumstances in which the clinical presentation of epidural hematoma and SAH will be more challenging. The question here is whether a negative initial neurologic exam can adequately screen for a potentially stealthy subdural hematoma.
Subdural hemorrhage is caused by rapidly changing velocity that may stretch and tear small bridging veins.2,3 Subdural hematoma is more common in the elderly, those who abuse alcohol, and those with a prior history of head trauma.4 As the brain shrinks with age or atrophy, the subdural space enlarges and traversing veins are stretched to cover a wider distance—rendering them vulnerable to rupture.5 These structures may also weaken as a result of low cerebrospinal fluid (intracranial hypotension); as pressure decreases (eg, from a leak), the brain’s buoyancy is reduced, causing traction on anchoring and supporting structures (eg, bridging veins).5 Injury to bridging veins can even occur as a result of a coup-contrecoup mechanism in the absence of direct physical impact.6,7 Bottom line: the injury itself may be subtle, requiring an index of suspicion to make the diagnosis.
Continue to: The case patient was...
The case patient was elderly. He had a chronic malignancy and sustained a fall down the stairs. He was taking sleeping pills, which may have slowed reflexive protective mechanisms after he started to fall (resulting in greater force imparted to his head). Multiple myeloma can predispose a patient to coagulopathy, and we don’t know in this case if this patient’s multiple myeloma made him more susceptible to bleeding—but it certainly didn’t help.8 The patient’s age, the mechanism of injury, and the history of malignancy made this a setup for hemorrhage.
Interestingly, we are not given details about how the patient looked during his suture removal. We are told the time between the initial fall and deterioration was 6 days. Scalp sutures were removed “about a week later,” which was after the deterioration—so this can’t be correct. Removing scalp sutures after 5 days seems premature, but that is the only possibility if 6 days elapsed between the fall and the deterioration.
In short, these are difficult cases. If intracranial bleeding can be subtle and delayed, how can we be sure a patient is not experiencing a bleed? We can only apply the relevant standard of care using all the clinical information we have. The Canadian CT Head Rule and New Orleans Criteria are clinical tools designed to help providers determine when to image (see Table for details).9

Applying the Canadian CT Head Rule to the facts of this case, we would image the patient because he fell down a “flight” of stairs (which is > 5 stairs) and he is 77 years old (older than 65). The New Orleans Criteria require head CT for minor injury with any positive findings.9 Because the patient is older than 60, he would be scanned according this rule.
In this case, the tools indicate scanning would have been appropriate. The patient’s multiple myeloma might have further impelled a decision to image. However, the jury was persuaded that the defendant’s negative neurologic exam was reasonable under the circumstances. This was likely made possible by the physician’s good recordkeeping and demonstrated genuine concern for the patient’s well-being—as well as a differing viewpoint of the patient’s age and health status.
Continue to: Finally, a word about...
Finally, a word about falls and the elderly: We’ve all heard the 80s advertising catchphrase (which lives on as a present-day meme) “I’ve fallen, and I can’t get up!” The problem is, many don’t. It would be more clinically accurate to say, “I’ve fallen, and I’ll be hospitalized for an extended period of time, then transferred to a skilled nursing facility, but I won’t survive to discharge.” The reality is that falls kill, and the severity is underestimated.10 If it were a “brain-eating amoeba,” the media would be all over it. With falls, not so much. We tend to pay less attention.
Risk factors for a fall include postural hypotension; use of benzodiazepines or other sedative-hypnotic drugs; use of ≥ 4 medications; environmental hazards for tripping; impairment in balance and transfer skills; and gait impairment.11 Home setup also contributes—loose throw rugs, uneven carpet edges, cracked sidewalks, clutter and furniture, cables and wires and cords, oh my.
Do your older patients a favor by reinforcing fall risk. Instruct them to rise slowly from seated or recumbent positions; always consider central nervous system sedation and/or the coordination-hampering properties of medications, particularly in combination. Raise the issue of home safety. A brief 10-second comment from you may plant a seed in a family member’s head to do what you cannot: scan and make safe the patient’s living environment.
In the early morning hours of June 10, 2009, a 77-year-old man who had been undergoing chemotherapy for multiple myeloma took sleep medication. He then fell down a flight of stairs in his split-level home.
The patient sustained a laceration to his scalp but returned to bed and waited until later that morning to call his internist for an appointment. Later that day, the physician placed 11 sutures for the scalp laceration and performed a neurologic examination; he did not note any abnormalities. The patient complained of back pain, so the physician ordered a back x-ray, which revealed a TI2 fracture that had occurred from the fall. No further treatment was provided for the scalp injury, except removal of the stitches about a week later.
Six days after the fall and doctor visit, the patient’s condition began to deteriorate rapidly, with noted slurred speech and loss of consciousness. He was transported to an emergency department, where CT revealed a massive subdural hematoma. An immediate craniotomy was performed. However, on June 27, 2009, the patient died as a result of the brain bleed.
His estate filed suit against the physician and his practice, alleging medical malpractice and violations in the standard of care. The estate alleged that the standard of care required the physician to obtain a CT scan and that, had one been performed, it would have revealed a small subdural hematoma in time for it to have been successfully treated (ie, before the massive second related bleed). The estate’s theory of the case did not rest on the presentation of clinical symptoms. A medical expert who testified for the estate stated that the subdural hematoma began at the time of the fall.
The defense denied any violations in the standard of care. The physician contended that the patient had presented with no symptoms other than a head laceration, and there were no criteria for ordering CT. Further, the defense asserted that the patient was symptom free for 6 days post-fall. According to the defense, the patient experienced a sudden arterial bleed that was not caused by the fall and would not have been revealed on CT ordered at the time of initial presentation, because it did not occur until 6 days later.
VERDICT
After a 10-day trial and 25 minutes’ deliberation, the jury returned a defense verdict.
COMMENTARY
The 25-minute deliberation suggests that terms such as “bridging veins” and “shearing injury” were unlikely bandied about in the jury room. The jury was likely dismissive of the plaintiff’s claim owing to his cancer diagnosis, and perhaps rightly so. But if we eliminate the multiple myeloma diagnosis, the jury might have decided differently.
Continue to: The defendant physician...
The defendant physician did a good job of documenting a negative neurologic exam, which helped him convince the jury that the patient did not have any signs or symptoms when first evaluated. But in this patient, was imaging to rule out intracranial bleeding indicated?
As an oversimplification, we tend to think of intracranial hemorrhage in 2 varieties: the insidious and the bold. Subdural hematomas are stealthy, they are sneaky, and they prey on the old. They step out of the shadows to cause symptoms. They are the ninjas of intracranial hemorrhage. Beware.
Epidural hematomas and subarachnoid hemorrhage (SAH) are the opposite. They classically present with a sudden and severe symptom complex: with epidural hematoma, the loss of consciousness, lucid interval, and final loss of consciousness; with SAH, the “worst in your life” thunder-clap headache, which may be heralded by a sentinel headache.1 When manifesting this way, they are brash, direct, and unsubtle to the point of being obnoxious—the Steven Stifler of intracranial bleeding.
This generalization is made to highlight the potentially sneaky nature of subdural hemorrhage. There are circumstances in which the clinical presentation of epidural hematoma and SAH will be more challenging. The question here is whether a negative initial neurologic exam can adequately screen for a potentially stealthy subdural hematoma.
Subdural hemorrhage is caused by rapidly changing velocity that may stretch and tear small bridging veins.2,3 Subdural hematoma is more common in the elderly, those who abuse alcohol, and those with a prior history of head trauma.4 As the brain shrinks with age or atrophy, the subdural space enlarges and traversing veins are stretched to cover a wider distance—rendering them vulnerable to rupture.5 These structures may also weaken as a result of low cerebrospinal fluid (intracranial hypotension); as pressure decreases (eg, from a leak), the brain’s buoyancy is reduced, causing traction on anchoring and supporting structures (eg, bridging veins).5 Injury to bridging veins can even occur as a result of a coup-contrecoup mechanism in the absence of direct physical impact.6,7 Bottom line: the injury itself may be subtle, requiring an index of suspicion to make the diagnosis.
Continue to: The case patient was...
The case patient was elderly. He had a chronic malignancy and sustained a fall down the stairs. He was taking sleeping pills, which may have slowed reflexive protective mechanisms after he started to fall (resulting in greater force imparted to his head). Multiple myeloma can predispose a patient to coagulopathy, and we don’t know in this case if this patient’s multiple myeloma made him more susceptible to bleeding—but it certainly didn’t help.8 The patient’s age, the mechanism of injury, and the history of malignancy made this a setup for hemorrhage.
Interestingly, we are not given details about how the patient looked during his suture removal. We are told the time between the initial fall and deterioration was 6 days. Scalp sutures were removed “about a week later,” which was after the deterioration—so this can’t be correct. Removing scalp sutures after 5 days seems premature, but that is the only possibility if 6 days elapsed between the fall and the deterioration.
In short, these are difficult cases. If intracranial bleeding can be subtle and delayed, how can we be sure a patient is not experiencing a bleed? We can only apply the relevant standard of care using all the clinical information we have. The Canadian CT Head Rule and New Orleans Criteria are clinical tools designed to help providers determine when to image (see Table for details).9

Applying the Canadian CT Head Rule to the facts of this case, we would image the patient because he fell down a “flight” of stairs (which is > 5 stairs) and he is 77 years old (older than 65). The New Orleans Criteria require head CT for minor injury with any positive findings.9 Because the patient is older than 60, he would be scanned according this rule.
In this case, the tools indicate scanning would have been appropriate. The patient’s multiple myeloma might have further impelled a decision to image. However, the jury was persuaded that the defendant’s negative neurologic exam was reasonable under the circumstances. This was likely made possible by the physician’s good recordkeeping and demonstrated genuine concern for the patient’s well-being—as well as a differing viewpoint of the patient’s age and health status.
Continue to: Finally, a word about...
Finally, a word about falls and the elderly: We’ve all heard the 80s advertising catchphrase (which lives on as a present-day meme) “I’ve fallen, and I can’t get up!” The problem is, many don’t. It would be more clinically accurate to say, “I’ve fallen, and I’ll be hospitalized for an extended period of time, then transferred to a skilled nursing facility, but I won’t survive to discharge.” The reality is that falls kill, and the severity is underestimated.10 If it were a “brain-eating amoeba,” the media would be all over it. With falls, not so much. We tend to pay less attention.
Risk factors for a fall include postural hypotension; use of benzodiazepines or other sedative-hypnotic drugs; use of ≥ 4 medications; environmental hazards for tripping; impairment in balance and transfer skills; and gait impairment.11 Home setup also contributes—loose throw rugs, uneven carpet edges, cracked sidewalks, clutter and furniture, cables and wires and cords, oh my.
Do your older patients a favor by reinforcing fall risk. Instruct them to rise slowly from seated or recumbent positions; always consider central nervous system sedation and/or the coordination-hampering properties of medications, particularly in combination. Raise the issue of home safety. A brief 10-second comment from you may plant a seed in a family member’s head to do what you cannot: scan and make safe the patient’s living environment.
1. de Falco FA. Sentinel headache. Neurol Sci. 2004;25(suppl 3):S215-S217.
2. Miller JD, Nader R. Acute subdural hematoma from bridging vein rupture: a potential mechanism for growth. J Neurosurg. 2014;120(6):1378-1384.
3. Victor M, Ropper A. Craniocerebral trauma. In: Victor M, Ropper A, eds. Adams and Victor’s Principles of Neurology. 7th ed. New York, NY: McGraw-Hill; 2001:925.
4. McBridde W. Subdural hematoma in adults: etiology, clinical features, and diagnosis. UpToDate website. www.uptodate.com/contents/subdural-hematoma-in-adults-etiology-clinical-features-and-diagnosis? search=subdural%20hematoma. Published December 10, 2018. Accessed September 23, 2019.
5. US National Library of Medicine. Subdural hematoma. Medline Plus website. https://medlineplus.gov/ency/article/000713.htm. Accessed September 23, 2019.
6. Besenski N. Traumatic injuries: imaging of head injuries. Eur Radiol. 2002;12(6):1237-1252.
7. Mayer S, Rowland L. Head injury. In: Rowland L, ed. Merritt’s Neurology. Philadelphia, PA: Lippincott Williams & Wilkins; 2000:401.
8. Saif MW, Allegra CJ, Greenberg B. Bleeding diathesis in multiple myeloma. J Hematother Stem Cell Res. 2001;10(5):657-660.
9. Stiell IG, Clement CM, Rowe BH, et al. Comparison of the Canadian CT Head Rule and the New Orleans Criteria in patients with minor head injury. JAMA. 2005;294(12):1511-1518.
10. Abdelrahman H, Almadani A, El-Menyar A, et al. Home-related falls: an underestimated mechanism of injury. J Family Community Med. 2018; 25(1):48-51.
11. Fuller GF. Falls in the elderly. Am Fam Physician. 2000;61(7):2159-2168.
1. de Falco FA. Sentinel headache. Neurol Sci. 2004;25(suppl 3):S215-S217.
2. Miller JD, Nader R. Acute subdural hematoma from bridging vein rupture: a potential mechanism for growth. J Neurosurg. 2014;120(6):1378-1384.
3. Victor M, Ropper A. Craniocerebral trauma. In: Victor M, Ropper A, eds. Adams and Victor’s Principles of Neurology. 7th ed. New York, NY: McGraw-Hill; 2001:925.
4. McBridde W. Subdural hematoma in adults: etiology, clinical features, and diagnosis. UpToDate website. www.uptodate.com/contents/subdural-hematoma-in-adults-etiology-clinical-features-and-diagnosis? search=subdural%20hematoma. Published December 10, 2018. Accessed September 23, 2019.
5. US National Library of Medicine. Subdural hematoma. Medline Plus website. https://medlineplus.gov/ency/article/000713.htm. Accessed September 23, 2019.
6. Besenski N. Traumatic injuries: imaging of head injuries. Eur Radiol. 2002;12(6):1237-1252.
7. Mayer S, Rowland L. Head injury. In: Rowland L, ed. Merritt’s Neurology. Philadelphia, PA: Lippincott Williams & Wilkins; 2000:401.
8. Saif MW, Allegra CJ, Greenberg B. Bleeding diathesis in multiple myeloma. J Hematother Stem Cell Res. 2001;10(5):657-660.
9. Stiell IG, Clement CM, Rowe BH, et al. Comparison of the Canadian CT Head Rule and the New Orleans Criteria in patients with minor head injury. JAMA. 2005;294(12):1511-1518.
10. Abdelrahman H, Almadani A, El-Menyar A, et al. Home-related falls: an underestimated mechanism of injury. J Family Community Med. 2018; 25(1):48-51.
11. Fuller GF. Falls in the elderly. Am Fam Physician. 2000;61(7):2159-2168.
Judicious EEG use identifies pseudosyncope during tilt-table testing
NEW ORLEANS – , according to an investigation from Vanderbilt University, Nashville.
Pseudosyncope, also known as nonsyncopal fainting (NSF), is a conversion disorder where people appear to faint, but don’t lose consciousness. It’s generally thought to be a physical manifestation of traumatic stress. Patients “aren’t faking it; they believe they are fainting,” said senior investigator Italo Biaggioni, MD, a professor of medicine and pharmacology at the university and director of the Vanderbilt Autonomic Dysfunction Center.
It’s “not benign. These patients stop working, stop driving, and need to depend on other people. They can have very dramatic episodes and hurt themselves” when they fall. “They are very disabled,” he said.
NSF is often misdiagnosed and mistreated, and sometimes unrecognized for years. Almost a third of the 39 NSF cases in Dr. Biaggioni’s series, for instance, were on anticonvulsants, and several had undergone cardiac catheterization. Often, NSF is treated as vasovagal syncope, but patients don’t respond to medications. A better way to identify it is needed. “By the time we get them, they’ve been through a lot.” Dr. Biaggioni said at the joint scientific sessions of the American Heart Association Council on Hypertension, AHA Council on Kidney in Cardiovascular Disease, and American Society of Hypertension.
He and his team had a hunch that judicious use of EEG would help, so they limited EEG to suspected NSF cases in a series of 107 refractory syncope patients referred to Vanderbilt for head-up tilt-table testing; 39 (36%) had normal EEGs during an apparent loss of consciousness, as opposed to the slow-wave pattern of true loss of consciousness, and were diagnosed with NSF.
The 36% identified was a marked increase in incidence over more common approaches. Among 64 patients who had tilt-table testing without EEG at Vanderbilt, for instance, three (5%) were diagnosed with NSF. Historically, tilt table plus EEG in all comers has a diagnostic yield of around 18% for the condition. In short, “elective EEG monitoring during tilt-table testing” better “distinguishes between syncope” and NSF, the team concluded.
NSF is suggested by a history of more than 20 episodes of apparent fainting; episodes once a week or more; or losing consciousness for more than 5 minutes. Fainting with eyes closed, or while supine, is also suggestive.
The elective approach prevents inappropriate treatment but is also therapeutic in itself. “When we document with EEG that patients are not really fainting, and explain that to them, it automatically reduces the number of episodes,” Dr. Biaggioni said.
It also saves NSF patients from a nitroglycerin challenge and repeat tilt testing, which is the default in many places when the first round of testing doesn’t trigger an episode. Challenge testing provokes vasovagal syncope in around 10% of even healthy people, so it puts NSF patients at risk for a false positive. As a rule, “we don’t use provocative agents when we [suspect NSF],” he said.
In addition to the 39 NSF cases, 11 patients in the series were diagnosed with vasovagal syncope, and testing didn’t provoke an event in 57 (53%), which isn’t uncommon.
Baseline blood pressure and heart rates were similar across the three groups, and there were more women than men in each. Subjects were in their early 40s, on average.
NSF patients were more likely to be taking anxiety and depression medications. One NSF patient had posttraumatic stress disorder, and two had sexual abuse histories, compared with none in the nondiagnostic and vasovagal groups. The NSF group had a shorter time to an event: 9 minutes versus 19 minutes among vasovagal patients.
Tilt-table testing was done after 6 hours of fasting, and the team used standard 22-channel EEG. Cognitive-behavioral therapy is the go-to treatment for NSF, Dr. Biaggioni said.
There was no industry funding, and the authors didn’t have any disclosures.
SOURCE: Muldowney JA et al. Joint Hypertension 2019, Abstract P3061.
NEW ORLEANS – , according to an investigation from Vanderbilt University, Nashville.
Pseudosyncope, also known as nonsyncopal fainting (NSF), is a conversion disorder where people appear to faint, but don’t lose consciousness. It’s generally thought to be a physical manifestation of traumatic stress. Patients “aren’t faking it; they believe they are fainting,” said senior investigator Italo Biaggioni, MD, a professor of medicine and pharmacology at the university and director of the Vanderbilt Autonomic Dysfunction Center.
It’s “not benign. These patients stop working, stop driving, and need to depend on other people. They can have very dramatic episodes and hurt themselves” when they fall. “They are very disabled,” he said.
NSF is often misdiagnosed and mistreated, and sometimes unrecognized for years. Almost a third of the 39 NSF cases in Dr. Biaggioni’s series, for instance, were on anticonvulsants, and several had undergone cardiac catheterization. Often, NSF is treated as vasovagal syncope, but patients don’t respond to medications. A better way to identify it is needed. “By the time we get them, they’ve been through a lot.” Dr. Biaggioni said at the joint scientific sessions of the American Heart Association Council on Hypertension, AHA Council on Kidney in Cardiovascular Disease, and American Society of Hypertension.
He and his team had a hunch that judicious use of EEG would help, so they limited EEG to suspected NSF cases in a series of 107 refractory syncope patients referred to Vanderbilt for head-up tilt-table testing; 39 (36%) had normal EEGs during an apparent loss of consciousness, as opposed to the slow-wave pattern of true loss of consciousness, and were diagnosed with NSF.
The 36% identified was a marked increase in incidence over more common approaches. Among 64 patients who had tilt-table testing without EEG at Vanderbilt, for instance, three (5%) were diagnosed with NSF. Historically, tilt table plus EEG in all comers has a diagnostic yield of around 18% for the condition. In short, “elective EEG monitoring during tilt-table testing” better “distinguishes between syncope” and NSF, the team concluded.
NSF is suggested by a history of more than 20 episodes of apparent fainting; episodes once a week or more; or losing consciousness for more than 5 minutes. Fainting with eyes closed, or while supine, is also suggestive.
The elective approach prevents inappropriate treatment but is also therapeutic in itself. “When we document with EEG that patients are not really fainting, and explain that to them, it automatically reduces the number of episodes,” Dr. Biaggioni said.
It also saves NSF patients from a nitroglycerin challenge and repeat tilt testing, which is the default in many places when the first round of testing doesn’t trigger an episode. Challenge testing provokes vasovagal syncope in around 10% of even healthy people, so it puts NSF patients at risk for a false positive. As a rule, “we don’t use provocative agents when we [suspect NSF],” he said.
In addition to the 39 NSF cases, 11 patients in the series were diagnosed with vasovagal syncope, and testing didn’t provoke an event in 57 (53%), which isn’t uncommon.
Baseline blood pressure and heart rates were similar across the three groups, and there were more women than men in each. Subjects were in their early 40s, on average.
NSF patients were more likely to be taking anxiety and depression medications. One NSF patient had posttraumatic stress disorder, and two had sexual abuse histories, compared with none in the nondiagnostic and vasovagal groups. The NSF group had a shorter time to an event: 9 minutes versus 19 minutes among vasovagal patients.
Tilt-table testing was done after 6 hours of fasting, and the team used standard 22-channel EEG. Cognitive-behavioral therapy is the go-to treatment for NSF, Dr. Biaggioni said.
There was no industry funding, and the authors didn’t have any disclosures.
SOURCE: Muldowney JA et al. Joint Hypertension 2019, Abstract P3061.
NEW ORLEANS – , according to an investigation from Vanderbilt University, Nashville.
Pseudosyncope, also known as nonsyncopal fainting (NSF), is a conversion disorder where people appear to faint, but don’t lose consciousness. It’s generally thought to be a physical manifestation of traumatic stress. Patients “aren’t faking it; they believe they are fainting,” said senior investigator Italo Biaggioni, MD, a professor of medicine and pharmacology at the university and director of the Vanderbilt Autonomic Dysfunction Center.
It’s “not benign. These patients stop working, stop driving, and need to depend on other people. They can have very dramatic episodes and hurt themselves” when they fall. “They are very disabled,” he said.
NSF is often misdiagnosed and mistreated, and sometimes unrecognized for years. Almost a third of the 39 NSF cases in Dr. Biaggioni’s series, for instance, were on anticonvulsants, and several had undergone cardiac catheterization. Often, NSF is treated as vasovagal syncope, but patients don’t respond to medications. A better way to identify it is needed. “By the time we get them, they’ve been through a lot.” Dr. Biaggioni said at the joint scientific sessions of the American Heart Association Council on Hypertension, AHA Council on Kidney in Cardiovascular Disease, and American Society of Hypertension.
He and his team had a hunch that judicious use of EEG would help, so they limited EEG to suspected NSF cases in a series of 107 refractory syncope patients referred to Vanderbilt for head-up tilt-table testing; 39 (36%) had normal EEGs during an apparent loss of consciousness, as opposed to the slow-wave pattern of true loss of consciousness, and were diagnosed with NSF.
The 36% identified was a marked increase in incidence over more common approaches. Among 64 patients who had tilt-table testing without EEG at Vanderbilt, for instance, three (5%) were diagnosed with NSF. Historically, tilt table plus EEG in all comers has a diagnostic yield of around 18% for the condition. In short, “elective EEG monitoring during tilt-table testing” better “distinguishes between syncope” and NSF, the team concluded.
NSF is suggested by a history of more than 20 episodes of apparent fainting; episodes once a week or more; or losing consciousness for more than 5 minutes. Fainting with eyes closed, or while supine, is also suggestive.
The elective approach prevents inappropriate treatment but is also therapeutic in itself. “When we document with EEG that patients are not really fainting, and explain that to them, it automatically reduces the number of episodes,” Dr. Biaggioni said.
It also saves NSF patients from a nitroglycerin challenge and repeat tilt testing, which is the default in many places when the first round of testing doesn’t trigger an episode. Challenge testing provokes vasovagal syncope in around 10% of even healthy people, so it puts NSF patients at risk for a false positive. As a rule, “we don’t use provocative agents when we [suspect NSF],” he said.
In addition to the 39 NSF cases, 11 patients in the series were diagnosed with vasovagal syncope, and testing didn’t provoke an event in 57 (53%), which isn’t uncommon.
Baseline blood pressure and heart rates were similar across the three groups, and there were more women than men in each. Subjects were in their early 40s, on average.
NSF patients were more likely to be taking anxiety and depression medications. One NSF patient had posttraumatic stress disorder, and two had sexual abuse histories, compared with none in the nondiagnostic and vasovagal groups. The NSF group had a shorter time to an event: 9 minutes versus 19 minutes among vasovagal patients.
Tilt-table testing was done after 6 hours of fasting, and the team used standard 22-channel EEG. Cognitive-behavioral therapy is the go-to treatment for NSF, Dr. Biaggioni said.
There was no industry funding, and the authors didn’t have any disclosures.
SOURCE: Muldowney JA et al. Joint Hypertension 2019, Abstract P3061.
REPORTING FROM JOINT HYPERTENSION 2019