User login
Aerobic exercise may mitigate age-related cognitive decline
published in Neurology.
“The effect of aerobic exercise on executive function was more pronounced as age increased, suggesting that it may mitigate age-related declines,” wrote Yaakov Stern, PhD, chief of cognitive neuroscience in the department of neurology at Columbia University, New York, and his research colleagues.
Research indicates that aerobic exercise provides cognitive benefits across the lifespan, but controlled exercise studies have been limited to elderly individuals, the researchers wrote. To examine the effects of aerobic exercise on cognitive function in younger, healthy adults, they conducted a randomized, parallel-group, observer-masked, community-based clinical trial. The investigators enrolled 132 cognitively normal people aged 20-67 years with aerobic capacity below the median. About 70% were women, and participants’ mean age was about 40 years.
“We hypothesized that aerobic exercise would have cognitive benefits, even in this younger age range, but that age might moderate the nature or degree of the benefit,” Dr. Stern and his colleagues wrote.
Participants were nonsmoking, habitual nonexercisers with below-average fitness by American Heart Association standards. The investigators used baseline aerobic capacity testing to establish safe exercise measures and heart rate targets.
The investigators randomly assigned participants to a group that performed aerobic exercise or to a control group that performed stretching and toning four times per week for 6 months. Outcome measures included domains of cognitive function (such as executive function, episodic memory, processing speed, language, and attention), everyday function, aerobic capacity, body mass index, and cortical thickness.
During a 2-week run-in period, participants went to their choice of five YMCA of New York City fitness centers three times per week. They had to attend at least five of these sessions to stay in the study. In both study arms, training sessions consisted of 10-15 minutes of warm-up and cooldown and 30-40 minutes of workout. Coaches contacted participants weekly to monitor their progress, and participants wore heart rate monitors during each session. Exercises in the control group were designed to promote flexibility and improve core strength. In the aerobic exercise group, participants had a choice of exercises such as walking on a treadmill, cycling on a stationary bike, or using an elliptical machine, and they gradually increased their exercise intensity to 75% of maximum heart rate by week 5. A total of 94 participants – 50 in the control group and 44 in the aerobic exercise group – completed the 6-month trial.
Executive function, but not other cognitive measures, improved significantly in the aerobic exercise group. The effect on executive function was greater in older participants. For example, at age 40 years, the executive function measure increased by 0.228 standard deviation units from baseline; at age 60, it increased by 0.596 standard deviation units.
In addition, cortical thickness increased significantly in the aerobic exercise group in the left caudal middle frontal cortex Brodmann area; this effect did not differ by age. Improvement on executive function in the aerobic exercise group was greater among participants without an APOE E4 allele, contrasting with the findings of prior studies.
“Since a difference of 0.5 standard deviations is equivalent to 20 years of age-related difference in performance on these tests, the people who exercised were testing as if they were about 10 years younger at age 40 and about 20 years younger at age 60,” Dr. Stern said in a press release. “Since thinking skills at the start of the study were poorer for participants who were older, our findings suggest that aerobic exercise is more likely to improve age-related declines in thinking skills rather than improve performance in those without a decline.”
Furthermore, aerobic exercise significantly increased aerobic capacity and significantly decreased body mass index, whereas stretching and toning did not.
“Participants in this trial scheduled their exercise sessions on their own and exercised by themselves,” the authors noted. “In addition, they were allowed to choose whatever aerobic exercise modality they preferred, so long as they reached target heart rates, enhancing the flexibility of the intervention.” Limitations of the study include its relatively small sample size and the large number of participants who dropped out of the study between consenting to participate and randomization.
The trial was funded by the National Institutes of Health. Dr. Stern reported receiving a grant from the California Walnut Commission and consulting with Eli Lilly, Axovant Sciences, Takeda, and AbbVie. A coauthor reported grant support from AposTherapy, LIH Medical, and the Everest Foundation.
SOURCE: Stern Y et al. Neurology. 2019 Jan 30. doi: 10.1212/WNL.0000000000007003.
published in Neurology.
“The effect of aerobic exercise on executive function was more pronounced as age increased, suggesting that it may mitigate age-related declines,” wrote Yaakov Stern, PhD, chief of cognitive neuroscience in the department of neurology at Columbia University, New York, and his research colleagues.
Research indicates that aerobic exercise provides cognitive benefits across the lifespan, but controlled exercise studies have been limited to elderly individuals, the researchers wrote. To examine the effects of aerobic exercise on cognitive function in younger, healthy adults, they conducted a randomized, parallel-group, observer-masked, community-based clinical trial. The investigators enrolled 132 cognitively normal people aged 20-67 years with aerobic capacity below the median. About 70% were women, and participants’ mean age was about 40 years.
“We hypothesized that aerobic exercise would have cognitive benefits, even in this younger age range, but that age might moderate the nature or degree of the benefit,” Dr. Stern and his colleagues wrote.
Participants were nonsmoking, habitual nonexercisers with below-average fitness by American Heart Association standards. The investigators used baseline aerobic capacity testing to establish safe exercise measures and heart rate targets.
The investigators randomly assigned participants to a group that performed aerobic exercise or to a control group that performed stretching and toning four times per week for 6 months. Outcome measures included domains of cognitive function (such as executive function, episodic memory, processing speed, language, and attention), everyday function, aerobic capacity, body mass index, and cortical thickness.
During a 2-week run-in period, participants went to their choice of five YMCA of New York City fitness centers three times per week. They had to attend at least five of these sessions to stay in the study. In both study arms, training sessions consisted of 10-15 minutes of warm-up and cooldown and 30-40 minutes of workout. Coaches contacted participants weekly to monitor their progress, and participants wore heart rate monitors during each session. Exercises in the control group were designed to promote flexibility and improve core strength. In the aerobic exercise group, participants had a choice of exercises such as walking on a treadmill, cycling on a stationary bike, or using an elliptical machine, and they gradually increased their exercise intensity to 75% of maximum heart rate by week 5. A total of 94 participants – 50 in the control group and 44 in the aerobic exercise group – completed the 6-month trial.
Executive function, but not other cognitive measures, improved significantly in the aerobic exercise group. The effect on executive function was greater in older participants. For example, at age 40 years, the executive function measure increased by 0.228 standard deviation units from baseline; at age 60, it increased by 0.596 standard deviation units.
In addition, cortical thickness increased significantly in the aerobic exercise group in the left caudal middle frontal cortex Brodmann area; this effect did not differ by age. Improvement on executive function in the aerobic exercise group was greater among participants without an APOE E4 allele, contrasting with the findings of prior studies.
“Since a difference of 0.5 standard deviations is equivalent to 20 years of age-related difference in performance on these tests, the people who exercised were testing as if they were about 10 years younger at age 40 and about 20 years younger at age 60,” Dr. Stern said in a press release. “Since thinking skills at the start of the study were poorer for participants who were older, our findings suggest that aerobic exercise is more likely to improve age-related declines in thinking skills rather than improve performance in those without a decline.”
Furthermore, aerobic exercise significantly increased aerobic capacity and significantly decreased body mass index, whereas stretching and toning did not.
“Participants in this trial scheduled their exercise sessions on their own and exercised by themselves,” the authors noted. “In addition, they were allowed to choose whatever aerobic exercise modality they preferred, so long as they reached target heart rates, enhancing the flexibility of the intervention.” Limitations of the study include its relatively small sample size and the large number of participants who dropped out of the study between consenting to participate and randomization.
The trial was funded by the National Institutes of Health. Dr. Stern reported receiving a grant from the California Walnut Commission and consulting with Eli Lilly, Axovant Sciences, Takeda, and AbbVie. A coauthor reported grant support from AposTherapy, LIH Medical, and the Everest Foundation.
SOURCE: Stern Y et al. Neurology. 2019 Jan 30. doi: 10.1212/WNL.0000000000007003.
published in Neurology.
“The effect of aerobic exercise on executive function was more pronounced as age increased, suggesting that it may mitigate age-related declines,” wrote Yaakov Stern, PhD, chief of cognitive neuroscience in the department of neurology at Columbia University, New York, and his research colleagues.
Research indicates that aerobic exercise provides cognitive benefits across the lifespan, but controlled exercise studies have been limited to elderly individuals, the researchers wrote. To examine the effects of aerobic exercise on cognitive function in younger, healthy adults, they conducted a randomized, parallel-group, observer-masked, community-based clinical trial. The investigators enrolled 132 cognitively normal people aged 20-67 years with aerobic capacity below the median. About 70% were women, and participants’ mean age was about 40 years.
“We hypothesized that aerobic exercise would have cognitive benefits, even in this younger age range, but that age might moderate the nature or degree of the benefit,” Dr. Stern and his colleagues wrote.
Participants were nonsmoking, habitual nonexercisers with below-average fitness by American Heart Association standards. The investigators used baseline aerobic capacity testing to establish safe exercise measures and heart rate targets.
The investigators randomly assigned participants to a group that performed aerobic exercise or to a control group that performed stretching and toning four times per week for 6 months. Outcome measures included domains of cognitive function (such as executive function, episodic memory, processing speed, language, and attention), everyday function, aerobic capacity, body mass index, and cortical thickness.
During a 2-week run-in period, participants went to their choice of five YMCA of New York City fitness centers three times per week. They had to attend at least five of these sessions to stay in the study. In both study arms, training sessions consisted of 10-15 minutes of warm-up and cooldown and 30-40 minutes of workout. Coaches contacted participants weekly to monitor their progress, and participants wore heart rate monitors during each session. Exercises in the control group were designed to promote flexibility and improve core strength. In the aerobic exercise group, participants had a choice of exercises such as walking on a treadmill, cycling on a stationary bike, or using an elliptical machine, and they gradually increased their exercise intensity to 75% of maximum heart rate by week 5. A total of 94 participants – 50 in the control group and 44 in the aerobic exercise group – completed the 6-month trial.
Executive function, but not other cognitive measures, improved significantly in the aerobic exercise group. The effect on executive function was greater in older participants. For example, at age 40 years, the executive function measure increased by 0.228 standard deviation units from baseline; at age 60, it increased by 0.596 standard deviation units.
In addition, cortical thickness increased significantly in the aerobic exercise group in the left caudal middle frontal cortex Brodmann area; this effect did not differ by age. Improvement on executive function in the aerobic exercise group was greater among participants without an APOE E4 allele, contrasting with the findings of prior studies.
“Since a difference of 0.5 standard deviations is equivalent to 20 years of age-related difference in performance on these tests, the people who exercised were testing as if they were about 10 years younger at age 40 and about 20 years younger at age 60,” Dr. Stern said in a press release. “Since thinking skills at the start of the study were poorer for participants who were older, our findings suggest that aerobic exercise is more likely to improve age-related declines in thinking skills rather than improve performance in those without a decline.”
Furthermore, aerobic exercise significantly increased aerobic capacity and significantly decreased body mass index, whereas stretching and toning did not.
“Participants in this trial scheduled their exercise sessions on their own and exercised by themselves,” the authors noted. “In addition, they were allowed to choose whatever aerobic exercise modality they preferred, so long as they reached target heart rates, enhancing the flexibility of the intervention.” Limitations of the study include its relatively small sample size and the large number of participants who dropped out of the study between consenting to participate and randomization.
The trial was funded by the National Institutes of Health. Dr. Stern reported receiving a grant from the California Walnut Commission and consulting with Eli Lilly, Axovant Sciences, Takeda, and AbbVie. A coauthor reported grant support from AposTherapy, LIH Medical, and the Everest Foundation.
SOURCE: Stern Y et al. Neurology. 2019 Jan 30. doi: 10.1212/WNL.0000000000007003.
FROM NEUROLOGY
Key clinical point: Among adults with below-average fitness, a 6-month aerobic exercise program significantly improves executive function.
Major finding: The effect is more pronounced as age increases.
Study details: A randomized, parallel-group, observer-masked, community-based clinical trial of 132 cognitively normal adults aged 20-67 years.
Disclosures: The study was funded by the National Institutes of Health. Dr. Stern reported receiving a grant from the California Walnut Commission and consulted with Eli Lilly, Axovant Sciences, Takeda, and AbbVie. Another reported grant support from AposTherapy, LIH Medical, and the Everest Foundation.
Source: Stern Y et al. Neurology. 2019 Jan 30. doi: 10.1212/WNL.0000000000007003.
Antidepressants for chronic pain
Approximately 55 years ago, tricyclic antidepressants (TCAs) began to be used to treat neuropathic pain.1 Eventually, clinical trials emerged suggesting the utility of TCAs for other chronic pain conditions, such as fibromyalgia (FM) and migraine prophylaxis. However, despite TCAs’ effectiveness in mitigating painful conditions, their adverse effects limited their use.
Pharmacologic advancements have led to the development of other antidepressant classes, including selective serotonin reuptake inhibitors (SSRIs) and serotonin-norepinephrine reuptake inhibitors (SNRIs), and the use of these agents has come to eclipse that of TCAs. In the realm of pain management, such developments have raised the hope of possible alternative co-analgesic agents that could avoid the adverse effects associated with TCAs. Some of these agents have demonstrated efficacy for managing chronic pain states, while others have demonstrated only limited utility.
This article provides a synopsis of systematic reviews and meta-analyses examining the role of antidepressant therapy for managing several chronic pain conditions, including pain associated with neuropathy, FM, headache, and irritable bowel syndrome (IBS). Because the literature base is rapidly evolving, it is necessary to revisit the information gleaned from clinical data with respect to treatment effectiveness, and to clarify how antidepressants might be positioned in the management of chronic pain.
The effectiveness of antidepressants for pain
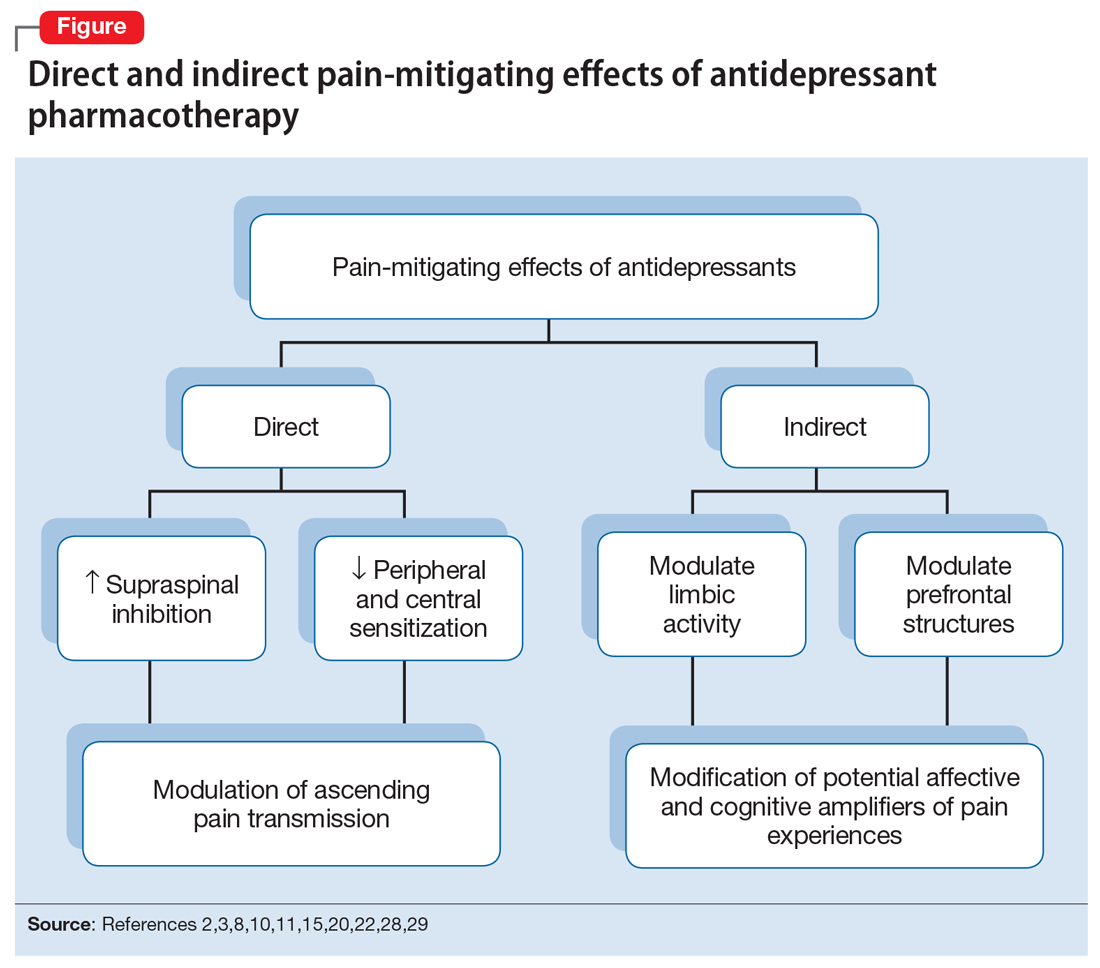
The pathophysiologic processes that precipitate and maintain chronic pain conditions are complex (Box 12-10). The pain-mitigating effects of antidepressants can be thought of in terms of direct analgesic effects and indirect effects (Box 22,3,8,10,11-33).
Box 1
The pathophysiologic processes precipitating and maintaining chronic pain conditions are complex. Persistent and chronic pain results from changes in sensitivity within both ascending pathways (relaying pain information from the periphery to the spinal cord and brain) and descending pain pathways (functioning to modulate ascending pain information).2,3 Tissue damage or peripheral nerve injury can lead to a cascade of neuroplastic changes within the CNS, resulting in hyperexcitability within the ascending pain pathways.
The descending pain pathways consist of the midbrain periaqueductal gray area (PGA), the rostroventral medulla (RVM), and the dorsolateral pontomesencephalic tegmentum (DLPT). The axons of the RVM (the outflow of which is serotonergic) and DLPT (the outflow of which is noradrenergic) terminate in the dorsal horn of the spinal cord,4 and thereby dampen pain signals arising from the periphery. Diminished output from descending pain pathways can heighten the pain experience. Input from the cortex, hypothalamus, and amygdala (among other structures) converges upon the PGA, RVM and DLPT, and can influence the degree of pain modulation emerging from descending pathways. In this way, thoughts, appraisals, and mood are believed to comprise cognitive and affective modifiers of pain experiences.
Devising effective chronic pain treatment becomes challenging; multimodal treatment approaches often are advocated, including pharmacologic treatment with analgesics in combination with co-analgesic medications such as antidepressants. Although a description of multimodal treatment is beyond the scope of this article, such treatment also would encompass physical therapy, occupational therapy, and psychotherapeutic interventions to augment rehabilitative efforts and the functional capabilities of patients who struggle with persisting pain.
Although the direct pain-mitigating effects of antidepressants are not fully understood, it is believed that augmentation of monoamine neurotransmission from supraspinal nuclei (ie, the RVM and DLPT) modulate pain transmission from the periphery.5,6 In addition, there is evidence that some effects of tricyclic antidepressants can modulate several other functions that impact peripheral and central sensitization.7-10
During the last several decades, antidepressants have been used to address—and have demonstrated clinical utility for—a variety of chronic pain states. However, antidepressants are not a panacea; some chronic pain conditions are more responsive to antidepressants than are others. The chronic painful states most amenable to antidepressants are those that result primarily from a process of neural sensitization, as opposed to acute somatic or visceral nociception. Hence, several meta-analyses and evidence-based reviews have long suggested the usefulness of antidepressants for mitigating pain associated with neuropathy,34,35 FM,36,37 headache,38 and IBS.39,40
Box 2
The pain-mitigating effects of antidepressants can be thought of in terms of direct analgesic effects (impacting neurotransmission of descending pathways independent of influences on mood) and indirect effects (presumably impacting cortical and limbic output to the periaqueductal gray area, the rostroventral medulla, and the dorsolateral pontomesencephalic tegmentum brought about by improvement in mood and/or cognitive appraisals) (Figure2,3,8,10,11,15,20,22,28,29). Support for the direct analgesic effects has been garnered from initial empirical work that demonstrated pain relief among patients with pain who are not depressed. Additionally, among patients who have depression and experience pain, analgesia reportedly often occurs within 2 weeks, which is before antidepressant effects are appreciated,11-15 and, at least for some antidepressants, occurs at doses far lower than those required to produce mood-elevating effects.11,12,16
On the other hand, it is well established that significant comorbidities exist between chronic pain states and psychiatric disorders (eg, depression and somatic symptom and related disorders).17-21 There may be common physiological substrates underlying chronic pain and depression.20,22 There are bidirectional influences of limbic (affective) systems and those CNS structures involved in pain processing and integration. The effects of pain and depression are reciprocal; the presence of one makes the management of the other more challenging.23-27 Mood disturbances can, therefore, impact pain processing by acting as affective and cognitive amplifiers of pain by leading to catastrophizing, pain severity augmentation, poor coping with pain-related stress, etc.28,29 It is plausible that the mood-elevating effects of antidepressants can improve pain by indirect effects, by modulating limbic activity, which in turn, impacts coping, cognitive appraisals of pain, etc.
Patients with somatoform disorders (using pre-DSM-5 terminology) frequently present with chronic pain, often in multiple sites.19 Such patients are characterized by hypervigilance for, and a predisposition to focus on, physical sensations and to appraise these sensations as reflecting a pathological state.30 Neuroimaging studies have begun to identify those neural circuits involved in somatoform disorders, many of which act as cognitive and affective amplifiers of visceral-somatic sensory processing. Many of these neural circuits overlap, and interact with, those involved in pain processing.31 Antidepressants can mitigate the severity of unexplained physical complaints, including pain, among patients who somatize32,33; however, due to the heterogeneity of studies upon which this claim is based, the quality of the evidence is reportedly low.33 There is uncertainty whether, or to what extent, antidepressant benefits among patients who somatize are due to a direct impact on pain modulation, or indirect effects on mood or cognitive appraisals/perceptions.
Despite the uncertainties about the exact mechanisms through which antidepressants exert analgesic effects, antidepressants can be appropriately used to treat patients with selected chronic pain syndromes, regardless of whether or not the patient has a psychiatric comorbidity. For those patients with pain and psychiatric comorbidities, the benefits may be brought about via direct mechanisms, indirect mechanisms, or a combination of both.
Continue to: Neuropathic pain
Neuropathic pain
Several treatment guidelines advocate for the use of antidepressants for neuropathic pain.41-44 For decades, TCAs have been employed off-label to successfully treat many patients with neuropathic pain states. Early investigations suggested that TCAs were robustly efficacious in managing patients with neuropathy.45-48 Calculated number-needed-to-treat (NNT) values for TCAs were quite low (ie, reflecting that few patients would need to be treated to yield a positive response in one patient compared with placebo), and were comparable to, if not slightly better than, the NNTs generated for anticonvulsants and α2-δ ligands, such as gabapentin or pregabalin.45-48
Unfortunately, early studies involving TCAs conducted many years ago do not meet contemporary standards of methodological rigor; they featured relatively small samples of patients assessed for brief post-treatment intervals with variable outcome measures. Thus, the NNT values obtained in meta-analyses based on these studies may overestimate treatment benefits.49 Further, NNT values derived from meta-analyses tended to combine all drugs within a particular antidepressant class (eg, amitriptyline, nortriptyline, desipramine, and imipramine among the TCAs) employed at diverse doses. Taken together, these limitations raise questions about the results of those meta-analyses.
Subsequent meta-analyses, which employed strict criteria to eliminate data from studies with potential sources of bias and used a primary outcome of frequencies of patients reporting at least 30% pain reduction compared with a placebo-controlled sample, suggest that the effectiveness of TCAs as a class for treating neuropathic pain is not as compelling as once was thought. Meta-analyses of studies employing specific TCAs revealed that there was little evidence to support the use of desipramine,50 imipramine,51 or nortriptyline52 in managing diabetic neuropathy or postherpetic neuralgia. Studies evaluating amitriptyline (dose range 12.5 to 150 mg/d), found low-level evidence of effectiveness; the benefit was expected to be present for a small subset (approximately 25%) of patients with neuropathic pain.53
There is moderate-quality evidence that duloxetine (60 to 120 mg/d) can produce a ≥50% improvement in pain severity ratings among patients with diabetic peripheral neuropathy.54 Although head-to-head studies with other antidepressants are limited, it appears that duloxetine and amitriptyline have comparable efficacy, even though the NNTs for amitriptyline were derived from lower-quality studies than those for duloxetine. Duloxetine is the only antidepressant to receive FDA approval for managing diabetic neuropathy. By contrast, studies assessing the utility of venlafaxine in neuropathic pain comprised small samples for brief durations, which limits the ability to draw clear (unbiased) support for its usefulness.55
Given the diversity of pathophysiologic processes underlying the disturbances that cause neuropathic pain disorders, it is unsurprising that the effectiveness of amitriptyline and duloxetine were not generalizable to all neuropathic pain states. Although amitriptyline produced pain-mitigating effects in patients with diabetic neuropathy and post-herpetic neuralgia, and duloxetine mitigated pain among patients with diabetic neuropathy, there was no evidence to suggest their effectiveness in phantom limb pain or human immunodeficiency virus-related and spinal cord-related neuropathies.35,53,54,56-58
Continue to: Fibromyalgia
Fibromyalgia
As with the issues encountered in interpreting the effectiveness of antidepressants in neuropathic pain, interpreting results gleaned from clinical trials of antidepressants for treating FM are fraught with similar difficulties. Although amitriptyline has been a first-line treatment for FM for many years, the evidence upon which such recommendations were based consisted of low-level studies that had a significant potential for bias.59 Large randomized trials would offer more compelling data regarding the efficacy of amitriptyline, but the prohibitive costs of such studies makes it unlikely they will be conducted. Amitriptyline (25 to 50 mg/d) was effective in mitigating FM-related pain in a small percentage of patients studied, with an estimated NNT of 4.59 Adverse effects, often contributing to treatment discontinuation, were encountered more frequently among patients who received amitriptyline compared with placebo.
Selective serotonin reuptake inhibitors failed to demonstrate significant pain relief (estimated NNT of 10), or improvement in fatigue or sleep problems, even though the studies upon which such conclusions were based were low-level studies with a high potential for bias.60 Although SSRIs have limited utility for mitigating pain, they are still quite useful for reducing depression among patients with FM.60
By contrast, the SNRIs duloxetine and milnacipran provided clinically relevant benefit over placebo in the frequency of patients reporting pain relief of ≥30%, as well as patients’ global impression of change.61 These agents, however, failed to provide clinically relevant benefit over placebo in improving health-related quality of life, reducing sleep problems, or improving fatigue. Nonetheless, duloxetine and milnacipran are FDA-approved for managing pain in FM. Studies assessing the efficacy of venlafaxine in the treatment of FM to date have been limited by small sample sizes, inconsistent dosing, lack of a placebo control, and lack of blinding, which limits the ability to clearly delineate the role of venlafaxine in managing FM.62
Mirtazapine (15 to 45 mg/d) showed a clinically relevant benefit compared with placebo for participant-reported pain relief of ≥30% and sleep disturbances. There was no benefit in terms of participant-reported improvement of quality of life, fatigue, or negative mood.63 The evidence was considered to be of low quality overall.
Headache
Amitriptyline has been employed off-label to address headache prophylaxis since 1964.64 Compared with placebo, it is efficacious in ameliorating migraine frequency and intensity as well as the frequency of tension headache.65,66 However, SSRIs and SNRIs (venlafaxine) failed to produce significant reductions in migraine frequency or severity or the frequencies of tension headache when compared with placebo.67,68
Continue to: Irritable bowel syndrome
Irritable bowel syndrome
Early studies addressing antidepressant efficacy in IBS reveal inconsistencies. For example, whereas some suggest that TCAs are effective in mitigating chronic, severe abdominal pain,39,40 others concluded that TCAs failed to demonstrate a significant analgesic benefit.69 A recent meta-analysis that restricted analysis of efficacy to randomized controlled trials (RCTs) with more rigorous methodological adherence found that pain relief in IBS is possible with both TCAs as well as SSRIs. However, adverse effects were more commonly encountered with TCAs than with SSRIs. Some of the inconsistencies in treatment efficacy reported in early studies may be due to variations in responsiveness of subsets of IBS patients. Specifically, the utility of TCAs appears to be best among patients with diarrheal-type (as opposed to constipation-type) IBS, presumably due to TCAs’ anticholinergic effects, whereas SSRIs may provide more of a benefit for patients with predominantly constipation-type IBS.40,70
Other chronic pain conditions
Antidepressants have been used to assist in the management of several other pain conditions, including oral-facial pain, interstitial cystitis, non-cardiac chest pain, and others. The role of antidepressants for such conditions remains unclear due to limitations in the prevailing empirical work, such as few trials, small sample sizes, variations in outcome measures, and insufficient randomization and blinding.71-76 The interpretation of results from systematic reviews and meta-analyses is limited because of these shortcomings.77 Hence, it has not always been possible to determine whether, and to what extent, patients with such conditions may benefit from antidepressants.
Neuromodulatory effects and efficacy for pain
The interplay of norepinephrine (NE) and serotonin (5-HT) neurotransmitter systems and cellular mechanisms involved in the descending modulation of pain pathways is complex. Experimental animal models of pain modulation suggest that 5-HT can both inhibit as well as promote pain perception by different physiological mechanisms, in contrast to NE, which is predominately inhibitory. While 5-HT in the descending modulating system can inhibit pain transmission ascending to the brain from the periphery, it appears that an intact noradrenergic system is necessary for the inhibitory influences of the serotonergic system to be appreciated.16,78,79 Deficiencies in one or both of these neurotransmitter systems may contribute to hyperactive pain processing, and thereby precipitate or maintain chronic pain.
Pain mitigation may be achieved best by enhancing both neurotransmitters simultaneously, less so by enhancing NE alone, and least by enhancing 5-HT alone.6 The ability to impact pain modulation would, therefore, depend on the degree to which an antidepressant capitalizes on both noradrenergic and serotonergic neurotransmission. Antidepressants commonly employed to manage pain are presented in Table 147,60,68,80-88 according to their primary neurotransmitter effects. Thus, the literature summarized above suggests that antidepressants that influence both NE and 5-HT transmission have greater analgesic effects than antidepressants with more specific effects, such as influencing 5-HT reuptake alone.80-85 It is unsurprising, therefore, that the SSRIs have not been demonstrated to be as consistently analgesic.47,60,68,80,86-88
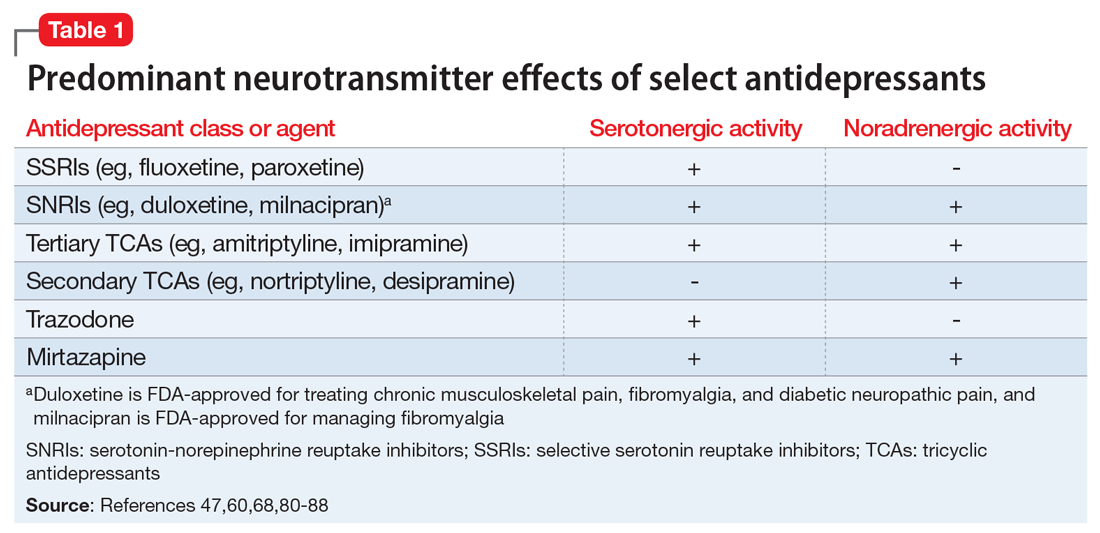
Similarly, pharmacodynamic and pharmacokinetic differences within antidepressant classes may influence analgesic effectiveness. Simultaneous effects on NE and 5-HT are achieved at low doses with duloxetine and milnacipran. By contrast, 5-HT effects predominate at low doses for venlafaxine. To achieve pain-mitigating effects, higher doses of venlafaxine generally are required.89 Therefore, inconsistencies across studies regarding the analgesic benefits of venlafaxine may be attributable to variability in dosing; patients treated with lower doses may not have experienced sufficient NE effects to garner positive results.
Continue to: The differences in analgesic efficacy...
The differences in analgesic efficacy among specific TCAs may be understood in a similar fashion. Specifically, tertiary TCAs (imipramine and amitriptyline) inhibit both 5-HT and NE reuptake.6,90 Secondary amines (desipramine and nortriptyline) predominantly impact NE reuptake, possibly accounting for the lesser pain-mitigating benefit achieved with these agents, such as for treating neuropathic pain. Further, in vivo imipramine and amitriptyline are rapidly metabolized to secondary amines that are potent and selective NE reuptake inhibitors. In this way, the secondary amines may substantially lose the ability to modulate pain transmission because of the loss of concurrent 5-HT influences.90
Clinical pearls
The following practical points can help guide clinicians regarding the usefulness of antidepressants for pain management:
- Antidepressants can alleviate symptoms of depression and pain. The pain-mitigating effects of antidepressants are possible even among chronic pain patients who are not depressed. Antidepressants may confer benefits for chronic pain patients with depression and other comorbid conditions, such as somatic symptom and related disorders.
- Antidepressants are useful for select chronic pain states. Although the noradrenergic and serotonergic antidepressants (SNRIs and, to some extent, amitriptyline) appear to have efficacy for neuropathic pain and FM, the benefits of SSRIs appear to be less robust. On the other hand, SSRIs and TCAs may have potential benefit for patients with IBS. However, the results of meta-analyses are limited in the ability to provide information about which patients will best respond to which specific antidepressant or how well. Future research directed at identifying characteristics that can predict which patients are likely to benefit from one antidepressant vs another would help inform how best to tailor treatment to individual needs.
- The pain-mitigating effects of antidepressants often emerge early in the course of treatment (often before mood-elevating effects are observed). For example, in the case of amitriptyline, pain relief may be possible for some patients at doses generally lower than those required for mood-elevating effects. To date, there is limited information in the literature to determine what constitutes a sufficient duration of treatment, or when treatment should be modified.
- Failure to reduce pain should raise questions about whether the dose should be increased, an alternative agent should be tried, or combinations with other analgesic agents should be considered. Failure to achieve pain-mitigating effects with one antidepressant does not mean failure with others. Hence, failure to achieve desired effects with one agent might warrant an empirical trial with another agent. Presently, too few double-blind RCTs have been conducted to assess the pain-mitigating effects of other antidepressants (eg, bupropion and newer SNRIs such as desvenlafaxine and levomilnacipran). Meta-analysis of the analgesic effectiveness of these agents or comparisons to the efficacy of other antidepressant classes is, therefore, impossible at this time.
Because many chronic pain states are complex, patients will seldom experience clinically relevant benefit from any one intervention.53 The bigger implication for clinical research is to determine whether there is a sequence or combination of medication use that will provide overall better clinical effectiveness.53 Only limited data are available exploring the utility of combining pharmacologic approaches to address pain.91 For example, preliminary evidence suggests that combinations of complementary strategies, such as duloxetine combined with pregabalin, may result in significantly greater numbers of FM patients achieving ≥30% pain reduction compared with monotherapy with either agent alone or placebo.92
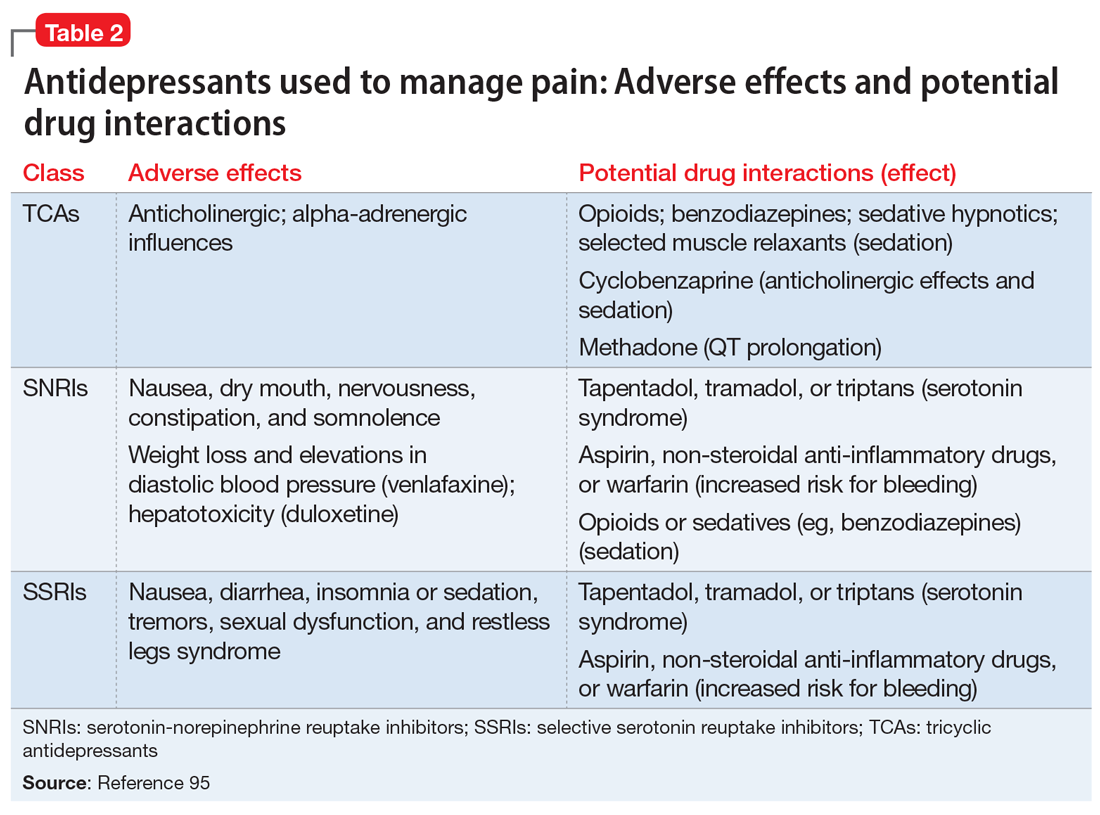
- Antidepressant selection may need to be based on medication-related adverse effect profiles and the potential for drug interactions. These factors are useful to consider in delineating multimodal treatment regimens for chronic pain in light of patients’ comorbidities and co-medication regimen. For example, the adverse effects of TCAs (anticholinergic and alpha-adrenergic influences) limit their utility for treating pain. Some of these effects can be more problematic in select populations, such as older adults or those with orthostatic difficulties, among others. TCAs are contraindicated in patients with closed-angle glaucoma, recent myocardial infarction, cardiac arrhythmias, poorly controlled seizures, or severe benign prostatic hypertrophy. Although the pain-mitigating effects of SNRIs have not been demonstrated to significantly exceed those of TCAs,68,93,94 SNRIs would offer an advantage of greater tolerability of adverse effects and relative safety in patients with comorbid medical conditions that would otherwise preclude TCA use. The adverse effects and common drug interactions associated with antidepressants are summarized in Table 295.
Conclusion
Chronic, nonmalignant pain conditions afflict many patients and significantly impair their ability to function. Because of heightened concerns related to the appropriateness of, and restricting inordinate access to, long-term opioid analgesics, clinicians need to explore the usefulness of co-analgesic agents, such as antidepressants. Significant comorbidities exist between psychiatric disorders and chronic pain, and psychiatrists are uniquely positioned to diagnose and treat psychiatric comorbidities, as well as pain, among their patients, especially since they understand the kinetics and dynamics of antidepressants.
Bottom Line
Antidepressants can alleviate symptoms of depression and pain. Noradrenergic and serotonergic antidepressants appear to have efficacy for pain associated with neuropathy and fibromyalgia, while selective serotonin reuptake inhibitors and tricyclic antidepressants may have benefit for patients with irritable bowel syndrome. However, evidence regarding which patients will best respond to which specific antidepressant is limited.
Continue to: Related Resources
Related Resources
- Williams AM, Knox ED. When to prescribe antidepressants to treat comorbid depression and pain disorders. Current Psychiatry. 2017;16(1):55-58.
- Maletic V, Demuri B. Chronic pain and depression: treatment of 2 culprits in common. Current Psychiatry. 2016;15(3):41,47-50,52.
Drug Brand Names
Amitriptyline • Elavil, Endep
Bupropion • Wellbutrin, Zyban
Carisoprodol • Rela, Soma
Cyclobenzaprine • Amrix, Flexeril
Desipramine • Norpramin
Desvenlafaxine • Pristiq
Duloxetine • Cymbalta
Fluoxetine • Prozac
Gabapentin • Horizant, Neurontin
Imipramine • Tofranil
Levomilnacipran • Fetzima
Methadone • Dolophine, Methadose
Milnacipran • Savella
Mirtazapine • Remeron
Nortriptyline • Pamelor
Paroxetine • Paxil
Pregabalin • Lyrica, Lyrica CR
Tapentadol • Nucynta
Tramadol • Ultram
Trazodone • Desyrel, Oleptro
Venlafaxine • Effexor
Warfarin • Coumadin, Jantoven
1. Paoli F, Darcourt G, Cossa P. Preliminary note on the action of imipramine in painful states [in French]. Rev Neurol (Paris). 1960;102:503-504.
2. Fields HL, Heinricher MM, Mason P. Neurotransmitters in nociceptive modulatory circuits. Annu Rev Neurosci. 1991;14:219-245.
3. Hunt SP, Mantyh PW. The molecular dynamics of pain control. Nat Rev Neurosci. 2001;2(2):83-91.
4. Lamont LA, Tranquilli WJ, Grimm KA. Physiology of pain. Vet Clin North Am Small Anim Pract. 2000;30(4):703-728, v.
5. Fields HL, Basbaum AI, Heinricher MM. Central nervous system mechanisms of pain modulation. In: McMahon S, Koltzenburg M, eds. Wall and Melzack’s Textbook of Pain. 5th ed. Burlington, MA: Elsevier Health Sciences; 2005:125-142.
6. Marks DM, Shah MJ, Patkar AA, et al. Serotonin-norepinephrine reuptake inhibitors for pain control: premise and promise. Curr Neuropharmacol. 2009;7(4):331-336.
7. Baba H, Shimoji K, Yoshimura M. Norepinephrine facilitates inhibitory transmission in substantia gelatinosa of adult rat spinal cord (part 1): effects on axon terminals of GABAergic and glycinergic neurons. Anesthesiology. 2000;92(2):473-484.
8. Carter GT, Sullivan MD. Antidepressants in pain management. Curr Opin Investig Drugs. 2002;3(3):454-458.
9. Kawasaki Y, Kumamoto E, Furue H, et al. Alpha 2 adrenoceptor-mediated presynaptic inhibition of primary afferent glutamatergic transmission in rat substantia gelatinosa neurons. Anesthesiology. 2003;98(3):682-689.
10. McCleane G. Antidepressants as analgesics. CNS Drugs. 2008;22(2):139-156.
11. Ansari A. The efficacy of newer antidepressants in the treatment of chronic pain: a review of current literature. Harv Rev Psychiatry. 2000;7(5):257-277.
12. Egbunike IG, Chaffee BJ. Antidepressants in the management of chronic pain syndromes. Pharmacotherapy. 1990;10(4):262-270.
13. Fishbain DA. Evidence-based data on pain relief with antidepressants. Ann Med. 2000;32(5):305-316.
14. Fishbain DA, Detke MJ, Wernicke J, et al. The relationship between antidepressant and analgesic responses: findings from six placebo-controlled trials assessing the efficacy of duloxetine in patients with major depressive disorder. Curr Med Res Opin. 2008;24(11):3105-3115.
15. Harada E, Tokuoka H, Fujikoshi S, et al. Is duloxetine’s effect on painful physical symptoms in depression an indirect result of improvement of depressive symptoms? Pooled analyses of three randomized controlled trials. Pain. 2016;157(3):577-584.
16. Kehoe WA. Antidepressants for chronic pain: selection and dosing considerations. Am J Pain Med. 1993;3(4):161-165.
17. Damush TM, Kroenke K, Bair MJ, et al. Pain self-management training increases self-efficacy, self-management behaviours and pain and depression outcomes. Eur J Pain. 2016;20(2):1070-1078.
18. DeVeaugh-Geiss AM, West SL, Miller WC, et al. The adverse effects of comorbid pain on depression outcomes in primary care patients: results from the ARTIST trial. Pain Medicine. 2010;11(5):732-741.
19. Egloff N, Cámara RJ, von Känel R, et al. Hypersensitivity and hyperalgesia in somatoform pain disorders. Gen Hosp Psychiatry. 2014;36(3):284-290.
20. Goesling J, Clauw DW, Hassett AL. Pain and depression: an integrative review of neurobiological and psychological factors. Curr Psych Reports. 2013;15(12):421.
21. Kroenke K, Wu J, Bair MJ, et al. Reciprocal relationship between pain and depression: a 12-Month longitudinal analysis in primary care. J Pain. 2011;12(9):964-973.
22. Leo RJ. Chronic pain and comorbid depression. Curr Treat Options Neurol. 2005;7(5):403-412.
23. Bair MJ, Robinson RL, Eckert GJ, et al. Impact of pain on depression treatment response in primary care. Psychosom Med. 2004;66(1):17-22.
24. Karp JF, Scott J, Houck P, et al. Pain predicts longer time to remission during treatment of recurrent depression. J Clin Psychiatry. 2005;66(5):591-597.
25. Kroenke K, Shen J, Oxman TE, et al. Impact of pain on the outcomes of depression treatment: results from the RESPECT trial. Pain. 2008;134(1-2):209-215.
26. Mavandadi S, Ten Have TR, Katz IR, et al. Effect of depression treatment on depressive symptoms in older adulthood: the moderating role of pain. J Am Geriatr Soc. 2007;55(2):202-211.
27. Thielke SM, Fan MY, Sullivan M, et al. Pain limits the effectiveness of collaborative care for depression. Am J Geriatr Psychiatry. 2007;15(8):699-707.
28. Arnow BA, Hunkeler EM, Blasey CM, et al. Comorbid depression, chronic pain, and disability in primary care. Psychosom Med. 2006;68(2):262-268.
29. Demyttenaere K, Bonnewyn A, Bruffaerts R, et al. Comorbid painful physical symptoms and depression: Prevalence, work loss, and help seeking. J Affect Disord. 2006;92(2-3):185-193.
30. Nakao M, Barsky AJ. Clinical application of somatosensory amplification in psychosomatic medicine. Biopsychosoc Med. 2007;1:17.
31. Perez DL, Barsky AJ, Vago DR, et al. A neural circuit framework for somatosensory amplification in somatoform disorders. J Neuropsychiatry Clin Neurosci. 2015;27(1):e40-e50.
32. Fishbain DA, Cutler RB, Rosomoff HL, et al. Do antidepressants have an analgesic effect in psychogenic pain and somatoform pain disorder? A meta-analysis. Psychosom Med. 1998;60(4):503-509.
33. Kleinstäuber M, Witthöft M, Steffanowski A, et al. Pharmacological interventions for somatoform disorders in adults. Cochrane Database Syst Rev. 2014;(11):CD010628.
34. Collins SL, Moore RA, McQuay HJ, et al. Antidepressants and anticonvulsants for diabetic neuropathy and postherpetic neuralgia: a quantitative systematic review. J Pain Symptom Manage. 2000;20(6):449-458.
35. Saarto T, Wiffen PJ. Antidepressants for neuropathic pain: a Cochrane review. J Neurol Neurosurg Psychiatry. 2010;81(12):1372-1373.
36. Arnold LM, Keck PE, Welge JA. Antidepressant treatment of fibromyalgia. A meta-analysis and review. Psychosomatics. 2000;41(2):104-113.
37. O’Malley PG, Balden E, Tomkins G, et al. Treatment of fibromyalgia with antidepressants: a meta-analysis. J Gen Intern Med. 2000;15(9):659-666.
38. Tomkins GE, Jackson JL, O’Malley PG, et al. Treatment of chronic headache with antidepressants: a meta-analysis. Am J Med. 2001;111(1):54-63.
39. Jackson JL, O’Malley PG, Tomkins G, et al. Treatment of functional gastrointestinal disorders with antidepressant medications: a meta-analysis. Am J Med. 2000;108(1):65-72.
40. Lesbros-Pantoflickova D, Michetti P, Fried M et al. Meta-analysis: the treatment of irritable bowel syndrome. Aliment Pharmacol Ther. 2004;20(11-12):1253-1269.
41. Centre for Clinical Practice at NICE (UK). Neuropathic pain: the pharmacological management of neuropathic pain in adults in non-specialist settings. London, UK: National Institute for Health and Care Excellence, (UK); 2013.
42. O’Connor AB, Dworkin RH. Treatment of neuropathic pain: an overview of recent guidelines. Am J Med. 2009;122(suppl 10):S22-S32.
43. Moulin D, Boulanger A, Clark AJ, et al; Canadian Pain Society. Pharmacological management of chronic neuropathic pain: revised consensus statement from the Canadian Pain Society. Pain Res Manag. 2014;19(6):328-35.
44. Mu A, Weinberg E, Moulin DE, et al. Pharmacologic management of chronic neuropathic pain: Review of the Canadian Pain Society consensus statement. Can Fam Physician. 2017;63(11):844-852.
45. Finnerup NB, Otto M, McQuay HJ, et al. Algorithm for neuropathic pain treatment: an evidence based proposal. Pain. 2005;118(3):289-305.
46. Hempenstall K, Nurmikko TJ, Johnson RW, et al. Analgesic therapy in postherpetic neuralgia: a quantitative systematic review. PLoS Med. 2005;2(7):e164.
47. Sindrup SH, Jensen TS. Efficacy of pharmacological treatments of neuropathic pain: an update and effect related to mechanism of drug action. Pain. 1999;83(3):389-400.
48. Wu CL, Raja SN. An update on the treatment of postherpetic neuralgia. J Pain. 2008;9(suppl 1):S19-S30.
49. Kroenke K, Krebs EE, Bair MJ. Pharmacotherapy of chronic pain: a synthesis of recommendations from systematic reviews. Gen Hosp Psychiatry. 2009;31(3):206-219.
50. Hearn L, Moore RA, Derry S, et al. Desipramine for neuropathic pain in adults. Cochrane Database Syst Rev. 2014;(9):CD011003.
51. Hearn L, Derry S, Phillips T, et al. Imipramine for neuropathic pain in adults. Cochrane Database Syst Rev. 2014;(5):CD010769.
52. Derry S, Wiffen PJ, Aldington D, et al. Nortriptyline for neuropathic pain in adults. Cochrane Database Syst Rev. 2015;1:CD011209.
53. Moore R, Derry S, Aldington D, et al. Amitriptyline for neuropathic pain in adults. Cochrane Database Syst Rev. 2015;(7):CD008242.
54. Lunn MP, Hughes RA, Wiffen PJ. Duloxetine for treating painful neuropathy, chronic pain or fibromyalgia. Cochrane Database Syst Rev. 2014;(1):CD007115.
55. Gallagher HC, Gallagher RM, Butler M, et al. Venlafaxine for neuropathic pain in adults. Cochrane Database Syst Rev. 2015;(8):CD011091.
56. Alviar MJ, Hale T, Dungca M. Pharmacologic interventions for treating phantom limb pain. Cochrane Database Syst Rev. 2016;10:CD006380.
57. Dinat N, Marinda E, Moch S, et al. Randomized, Double-Blind, Crossover Trial of Amitriptyline for Analgesia in Painful HIV-Associated Sensory Neuropathy. PLoS One. 2015;10(5):e0126297. doi: 10.1371/journal.pone.0126297.eCollection 2015.
58. Mehta S, McIntyre A, Janzen S, et al; Spinal Cord Injury Rehabilitation Evidence Team. Systematic review of pharmacologic treatments of pain after spinal cord injury: an update. Arch Phys Med Rehabil. 2016;97(8):1381-1391.e1.
59. Moore RA, Derry S, Aldington D, et al. Amitriptyline for neuropathic pain and fibromyalgia in adults. Cochrane Database Syst Rev. 2012;(12):CD008242..
60. Walitt B, Urrútia G, Nishishinya MB, et al. Selective serotonin reuptake inhibitors for fibromyalgia syndrome. Cochrane Database Syst Rev. 2015;(6):CD011735.
61. Welsch P, Üçeyler N, Klose P, et al. Serotonin and noradrenaline reuptake inhibitors (SNRIs) for fibromyalgia. Cochrane Database Syst Rev. 2018;(2):CD010292.
62. VanderWeide LA, Smith SM, Trinkley KE. A systematic review of the efficacy of venlafaxine for the treatment of fibromyalgia. J Clin Pharm Ther. 2015;40(1):1-6.
63. Welsch P, Bernardy K, Derry S, et al. Mirtazapine for fibromyalgia in adults. Cochrane Database Syst Rev. 2018;(8):CD012708.
64. Lance JW, Curran DA. Treatment of chronic tension headache. Lancet. 1964;283(7345):1236-1239.
65. Jackson JL, William S, Laura S, et al. Tricyclic antidepressants and headaches: systematic review and meta-analysis. BMJ. 2010;341:c5222. doi: https://doi.org/10.1136/bmj.c5222
66. Xu XM, Liu Y, Dong MX, et al. Tricyclic antidepressants for preventing migraine in adults. Medicine. 2017;96(22):e6989. doi: 10.1097/MD.0000000000006989.
67. Banzi R, Cusi C, Randazzo C, et al. Selective serotonin reuptake inhibitors (SSRIs) and serotonin-norepinephrine reuptake inhibitors (SNRIs) for the prevention of migraine in adults. Cochrane Database Syst Rev. 2015;(4):CD002919.
68. Banzi R, Cusi C, Randazzo C, et al. Selective serotonin reuptake inhibitors (SSRIs) and serotonin-norepinephrine reuptake inhibitors (SNRIs) for the prevention of tension-type headache in adults. Cochrane Database Syst Rev. 2015;(5):CD011681.
69. Quartero AO, Meineche-Schmidt V, Muris J, et al. Bulking agents, antispasmodic and antidepressant medication for the treatment of irritable bowel syndrome. Cochrane Database Syst Rev. 2005;(2):CD003460.
70. Ford AC, Talley NJ, Schoenfeld PS, et al. Efficacy of antidepressants and psychological therapies in irritable bowel syndrome: systematic review and meta-analysis. Gut. 2009;58(3):367-378.
71. Coss-Adame E, Erdogan A, Rao SS. Treatment of esophageal (noncardiac) chest pain: an expert review. Clin Gastroenterol Hepatol. 2014;12(8):1224-1245.
72. Kelada E, Jones A. Interstitial cystitis. Arch Gynecol Obstet. 2007;275(4):223-229.
73. Leo RJ, Dewani S. A systematic review of the utility of antidepressant pharmacotherapy in the treatment of vulvodynia pain. J Sex Med. 2013;10(10):2497-2505.
74. McMillan R, Forssell H, Buchanan JA, et al. Interventions for treating burning mouth syndrome. Cochrane Database Syst Rev. 2016;11:CD002779.
75. Patel DN. Inconclusive results of a systematic review of efficacy of antidepressants on orofacial pain disorders. Evid Based Dent. 2013;14(2):55-56.
76. Wang W, Sun YH, Wang YY, et al. Treatment of functional chest pain with antidepressants: a meta-analysis. Pain Physician. 2012;15(2):E131-E142.
77. Lavis JN. How can we support the use of systematic reviews in policymaking? PLoS Med. 2009;6(11):e1000141. doi: 10.1371/journal.pmed.1000141.
78. Sorkin L. Nociceptive transmission within the spinal cord. Mt Sinai J Med. 1991;58(3):208-216.
79. Yokogawa F, Kiuchi Y, Ishikawa Y, et al. An investigation of monoamine receptors involved in antinociceptive effects of antidepressants. Anesth Analg. 2002;95(1):163-168, table of contents.
80. Lynch ME. Antidepressants as analgesics: a review of randomized controlled trials. J Psychiatry Neurosci. 2001;26(1):30-36.
81. Max MB. Treatment of post-herpetic neuralgia: antidepressants. Ann Neurol. 1994;35(suppl):S50-S53.
82. Max MB, Lynch SA, Muir J, et al. Effects of desipramine, amitriptyline, and fluoxetine on pain in diabetic neuropathy. N Engl J Med. 1992;326(19):1250-1256.
83. McQuay HJ, Tramèr M, Nye BA, et al. A systematic review of antidepressants in neuropathic pain. Pain. 1996;68(2-3):217-227.
84. Mochizucki D. Serotonin and noradrenaline reuptake inhibitors in animal models of pain. Hum Psychopharmacol Clin Exp. 2004;19(suppl 1):15-19.
85. Sussman N. SNRIs versus SSRIs: mechanisms of action in treating depression and painful physical symptoms. Primary Care Companion J Clin Psychiatry. 2003;5(suppl 7):19-26.
86. Bundeff AW, Woodis CB. Selective serotonin reuptake inhibitors for the treatment of irritable bowel syndrome. Ann Pharmacother. 2014;48(6):777-784.
87. Jung AC, Staiger T, Sullivan M. The efficacy of selective serotonin reuptake inhibitors for the management of chronic pain. J Gen Intern Med. 1997;12(6):384-389.
88. Xie C, Tang Y, Wang Y, et al. Efficacy and safety of antidepressants for the treatment of irritable bowel syndrome: a meta-analysis. PLoS One. 2015;10(8):e0127815. doi: 10.1371/journal.pone.0127815. eCollection 2015.
89. Zijlstra TR , Barendregt PJ , van de Laar MA. Venlafaxine in fibromyalgia: results of a randomized, placebo-controlled, double-blind trial. Arthritis Rheum. 2002;46(suppl 9):S105.
90. Bymaster FP, Dreshfield-Ahmad LJ, Threlkeld PG. Comparative affinity of duloxetine and venlafaxine for serotonin and norepinephrine transporters in vitro and in vivo, human serotonin receptor subtypes, and other neuronal receptors. Neuropsychopharmacology. 2001;25(6):871-880.
91. Thorpe J, Shum B, Moore RA, et al. Combination pharmacotherapy for the treatment of fibromyalgia in adults. Cochrane Database Syst Rev. 2018;(2):CD010585.
92. Gilron I, Chaparro LE, Tu D, et al. Combination of pregabalin with duloxetine for fibromyalgia: a randomized controlled trial. Pain. 2016;157(7):1532-1540.
93. Häuser W, Petzke F, Üçeyler N, et al. Comparative efficacy and acceptability of amitriptyline, duloxetine and milnacipran in fibromyalgia syndrome: a systematic review with meta-analysis. Rheumatology (Oxford). 2011;50(3):532-543.
94. Hossain SM, Hussain SM, Ekram AR. Duloxetine in painful diabetic neuropathy: a systematic review. Clin J Pain. 2016;32(11):1005-1010.
95. Riediger C, Schuster T, Barlinn K, et al. Adverse effects of antidepressants for chronic pain: a systematic review and meta-analysis. Front Neurol. 2017;8:307.
Approximately 55 years ago, tricyclic antidepressants (TCAs) began to be used to treat neuropathic pain.1 Eventually, clinical trials emerged suggesting the utility of TCAs for other chronic pain conditions, such as fibromyalgia (FM) and migraine prophylaxis. However, despite TCAs’ effectiveness in mitigating painful conditions, their adverse effects limited their use.
Pharmacologic advancements have led to the development of other antidepressant classes, including selective serotonin reuptake inhibitors (SSRIs) and serotonin-norepinephrine reuptake inhibitors (SNRIs), and the use of these agents has come to eclipse that of TCAs. In the realm of pain management, such developments have raised the hope of possible alternative co-analgesic agents that could avoid the adverse effects associated with TCAs. Some of these agents have demonstrated efficacy for managing chronic pain states, while others have demonstrated only limited utility.
This article provides a synopsis of systematic reviews and meta-analyses examining the role of antidepressant therapy for managing several chronic pain conditions, including pain associated with neuropathy, FM, headache, and irritable bowel syndrome (IBS). Because the literature base is rapidly evolving, it is necessary to revisit the information gleaned from clinical data with respect to treatment effectiveness, and to clarify how antidepressants might be positioned in the management of chronic pain.
The effectiveness of antidepressants for pain
The pathophysiologic processes that precipitate and maintain chronic pain conditions are complex (Box 12-10). The pain-mitigating effects of antidepressants can be thought of in terms of direct analgesic effects and indirect effects (Box 22,3,8,10,11-33).
Box 1
The pathophysiologic processes precipitating and maintaining chronic pain conditions are complex. Persistent and chronic pain results from changes in sensitivity within both ascending pathways (relaying pain information from the periphery to the spinal cord and brain) and descending pain pathways (functioning to modulate ascending pain information).2,3 Tissue damage or peripheral nerve injury can lead to a cascade of neuroplastic changes within the CNS, resulting in hyperexcitability within the ascending pain pathways.
The descending pain pathways consist of the midbrain periaqueductal gray area (PGA), the rostroventral medulla (RVM), and the dorsolateral pontomesencephalic tegmentum (DLPT). The axons of the RVM (the outflow of which is serotonergic) and DLPT (the outflow of which is noradrenergic) terminate in the dorsal horn of the spinal cord,4 and thereby dampen pain signals arising from the periphery. Diminished output from descending pain pathways can heighten the pain experience. Input from the cortex, hypothalamus, and amygdala (among other structures) converges upon the PGA, RVM and DLPT, and can influence the degree of pain modulation emerging from descending pathways. In this way, thoughts, appraisals, and mood are believed to comprise cognitive and affective modifiers of pain experiences.
Devising effective chronic pain treatment becomes challenging; multimodal treatment approaches often are advocated, including pharmacologic treatment with analgesics in combination with co-analgesic medications such as antidepressants. Although a description of multimodal treatment is beyond the scope of this article, such treatment also would encompass physical therapy, occupational therapy, and psychotherapeutic interventions to augment rehabilitative efforts and the functional capabilities of patients who struggle with persisting pain.
Although the direct pain-mitigating effects of antidepressants are not fully understood, it is believed that augmentation of monoamine neurotransmission from supraspinal nuclei (ie, the RVM and DLPT) modulate pain transmission from the periphery.5,6 In addition, there is evidence that some effects of tricyclic antidepressants can modulate several other functions that impact peripheral and central sensitization.7-10
During the last several decades, antidepressants have been used to address—and have demonstrated clinical utility for—a variety of chronic pain states. However, antidepressants are not a panacea; some chronic pain conditions are more responsive to antidepressants than are others. The chronic painful states most amenable to antidepressants are those that result primarily from a process of neural sensitization, as opposed to acute somatic or visceral nociception. Hence, several meta-analyses and evidence-based reviews have long suggested the usefulness of antidepressants for mitigating pain associated with neuropathy,34,35 FM,36,37 headache,38 and IBS.39,40
Box 2
The pain-mitigating effects of antidepressants can be thought of in terms of direct analgesic effects (impacting neurotransmission of descending pathways independent of influences on mood) and indirect effects (presumably impacting cortical and limbic output to the periaqueductal gray area, the rostroventral medulla, and the dorsolateral pontomesencephalic tegmentum brought about by improvement in mood and/or cognitive appraisals) (Figure2,3,8,10,11,15,20,22,28,29). Support for the direct analgesic effects has been garnered from initial empirical work that demonstrated pain relief among patients with pain who are not depressed. Additionally, among patients who have depression and experience pain, analgesia reportedly often occurs within 2 weeks, which is before antidepressant effects are appreciated,11-15 and, at least for some antidepressants, occurs at doses far lower than those required to produce mood-elevating effects.11,12,16
On the other hand, it is well established that significant comorbidities exist between chronic pain states and psychiatric disorders (eg, depression and somatic symptom and related disorders).17-21 There may be common physiological substrates underlying chronic pain and depression.20,22 There are bidirectional influences of limbic (affective) systems and those CNS structures involved in pain processing and integration. The effects of pain and depression are reciprocal; the presence of one makes the management of the other more challenging.23-27 Mood disturbances can, therefore, impact pain processing by acting as affective and cognitive amplifiers of pain by leading to catastrophizing, pain severity augmentation, poor coping with pain-related stress, etc.28,29 It is plausible that the mood-elevating effects of antidepressants can improve pain by indirect effects, by modulating limbic activity, which in turn, impacts coping, cognitive appraisals of pain, etc.
Patients with somatoform disorders (using pre-DSM-5 terminology) frequently present with chronic pain, often in multiple sites.19 Such patients are characterized by hypervigilance for, and a predisposition to focus on, physical sensations and to appraise these sensations as reflecting a pathological state.30 Neuroimaging studies have begun to identify those neural circuits involved in somatoform disorders, many of which act as cognitive and affective amplifiers of visceral-somatic sensory processing. Many of these neural circuits overlap, and interact with, those involved in pain processing.31 Antidepressants can mitigate the severity of unexplained physical complaints, including pain, among patients who somatize32,33; however, due to the heterogeneity of studies upon which this claim is based, the quality of the evidence is reportedly low.33 There is uncertainty whether, or to what extent, antidepressant benefits among patients who somatize are due to a direct impact on pain modulation, or indirect effects on mood or cognitive appraisals/perceptions.
Despite the uncertainties about the exact mechanisms through which antidepressants exert analgesic effects, antidepressants can be appropriately used to treat patients with selected chronic pain syndromes, regardless of whether or not the patient has a psychiatric comorbidity. For those patients with pain and psychiatric comorbidities, the benefits may be brought about via direct mechanisms, indirect mechanisms, or a combination of both.

Continue to: Neuropathic pain
Neuropathic pain
Several treatment guidelines advocate for the use of antidepressants for neuropathic pain.41-44 For decades, TCAs have been employed off-label to successfully treat many patients with neuropathic pain states. Early investigations suggested that TCAs were robustly efficacious in managing patients with neuropathy.45-48 Calculated number-needed-to-treat (NNT) values for TCAs were quite low (ie, reflecting that few patients would need to be treated to yield a positive response in one patient compared with placebo), and were comparable to, if not slightly better than, the NNTs generated for anticonvulsants and α2-δ ligands, such as gabapentin or pregabalin.45-48
Unfortunately, early studies involving TCAs conducted many years ago do not meet contemporary standards of methodological rigor; they featured relatively small samples of patients assessed for brief post-treatment intervals with variable outcome measures. Thus, the NNT values obtained in meta-analyses based on these studies may overestimate treatment benefits.49 Further, NNT values derived from meta-analyses tended to combine all drugs within a particular antidepressant class (eg, amitriptyline, nortriptyline, desipramine, and imipramine among the TCAs) employed at diverse doses. Taken together, these limitations raise questions about the results of those meta-analyses.
Subsequent meta-analyses, which employed strict criteria to eliminate data from studies with potential sources of bias and used a primary outcome of frequencies of patients reporting at least 30% pain reduction compared with a placebo-controlled sample, suggest that the effectiveness of TCAs as a class for treating neuropathic pain is not as compelling as once was thought. Meta-analyses of studies employing specific TCAs revealed that there was little evidence to support the use of desipramine,50 imipramine,51 or nortriptyline52 in managing diabetic neuropathy or postherpetic neuralgia. Studies evaluating amitriptyline (dose range 12.5 to 150 mg/d), found low-level evidence of effectiveness; the benefit was expected to be present for a small subset (approximately 25%) of patients with neuropathic pain.53
There is moderate-quality evidence that duloxetine (60 to 120 mg/d) can produce a ≥50% improvement in pain severity ratings among patients with diabetic peripheral neuropathy.54 Although head-to-head studies with other antidepressants are limited, it appears that duloxetine and amitriptyline have comparable efficacy, even though the NNTs for amitriptyline were derived from lower-quality studies than those for duloxetine. Duloxetine is the only antidepressant to receive FDA approval for managing diabetic neuropathy. By contrast, studies assessing the utility of venlafaxine in neuropathic pain comprised small samples for brief durations, which limits the ability to draw clear (unbiased) support for its usefulness.55
Given the diversity of pathophysiologic processes underlying the disturbances that cause neuropathic pain disorders, it is unsurprising that the effectiveness of amitriptyline and duloxetine were not generalizable to all neuropathic pain states. Although amitriptyline produced pain-mitigating effects in patients with diabetic neuropathy and post-herpetic neuralgia, and duloxetine mitigated pain among patients with diabetic neuropathy, there was no evidence to suggest their effectiveness in phantom limb pain or human immunodeficiency virus-related and spinal cord-related neuropathies.35,53,54,56-58
Continue to: Fibromyalgia
Fibromyalgia
As with the issues encountered in interpreting the effectiveness of antidepressants in neuropathic pain, interpreting results gleaned from clinical trials of antidepressants for treating FM are fraught with similar difficulties. Although amitriptyline has been a first-line treatment for FM for many years, the evidence upon which such recommendations were based consisted of low-level studies that had a significant potential for bias.59 Large randomized trials would offer more compelling data regarding the efficacy of amitriptyline, but the prohibitive costs of such studies makes it unlikely they will be conducted. Amitriptyline (25 to 50 mg/d) was effective in mitigating FM-related pain in a small percentage of patients studied, with an estimated NNT of 4.59 Adverse effects, often contributing to treatment discontinuation, were encountered more frequently among patients who received amitriptyline compared with placebo.
Selective serotonin reuptake inhibitors failed to demonstrate significant pain relief (estimated NNT of 10), or improvement in fatigue or sleep problems, even though the studies upon which such conclusions were based were low-level studies with a high potential for bias.60 Although SSRIs have limited utility for mitigating pain, they are still quite useful for reducing depression among patients with FM.60
By contrast, the SNRIs duloxetine and milnacipran provided clinically relevant benefit over placebo in the frequency of patients reporting pain relief of ≥30%, as well as patients’ global impression of change.61 These agents, however, failed to provide clinically relevant benefit over placebo in improving health-related quality of life, reducing sleep problems, or improving fatigue. Nonetheless, duloxetine and milnacipran are FDA-approved for managing pain in FM. Studies assessing the efficacy of venlafaxine in the treatment of FM to date have been limited by small sample sizes, inconsistent dosing, lack of a placebo control, and lack of blinding, which limits the ability to clearly delineate the role of venlafaxine in managing FM.62
Mirtazapine (15 to 45 mg/d) showed a clinically relevant benefit compared with placebo for participant-reported pain relief of ≥30% and sleep disturbances. There was no benefit in terms of participant-reported improvement of quality of life, fatigue, or negative mood.63 The evidence was considered to be of low quality overall.
Headache
Amitriptyline has been employed off-label to address headache prophylaxis since 1964.64 Compared with placebo, it is efficacious in ameliorating migraine frequency and intensity as well as the frequency of tension headache.65,66 However, SSRIs and SNRIs (venlafaxine) failed to produce significant reductions in migraine frequency or severity or the frequencies of tension headache when compared with placebo.67,68
Continue to: Irritable bowel syndrome
Irritable bowel syndrome
Early studies addressing antidepressant efficacy in IBS reveal inconsistencies. For example, whereas some suggest that TCAs are effective in mitigating chronic, severe abdominal pain,39,40 others concluded that TCAs failed to demonstrate a significant analgesic benefit.69 A recent meta-analysis that restricted analysis of efficacy to randomized controlled trials (RCTs) with more rigorous methodological adherence found that pain relief in IBS is possible with both TCAs as well as SSRIs. However, adverse effects were more commonly encountered with TCAs than with SSRIs. Some of the inconsistencies in treatment efficacy reported in early studies may be due to variations in responsiveness of subsets of IBS patients. Specifically, the utility of TCAs appears to be best among patients with diarrheal-type (as opposed to constipation-type) IBS, presumably due to TCAs’ anticholinergic effects, whereas SSRIs may provide more of a benefit for patients with predominantly constipation-type IBS.40,70
Other chronic pain conditions
Antidepressants have been used to assist in the management of several other pain conditions, including oral-facial pain, interstitial cystitis, non-cardiac chest pain, and others. The role of antidepressants for such conditions remains unclear due to limitations in the prevailing empirical work, such as few trials, small sample sizes, variations in outcome measures, and insufficient randomization and blinding.71-76 The interpretation of results from systematic reviews and meta-analyses is limited because of these shortcomings.77 Hence, it has not always been possible to determine whether, and to what extent, patients with such conditions may benefit from antidepressants.
Neuromodulatory effects and efficacy for pain
The interplay of norepinephrine (NE) and serotonin (5-HT) neurotransmitter systems and cellular mechanisms involved in the descending modulation of pain pathways is complex. Experimental animal models of pain modulation suggest that 5-HT can both inhibit as well as promote pain perception by different physiological mechanisms, in contrast to NE, which is predominately inhibitory. While 5-HT in the descending modulating system can inhibit pain transmission ascending to the brain from the periphery, it appears that an intact noradrenergic system is necessary for the inhibitory influences of the serotonergic system to be appreciated.16,78,79 Deficiencies in one or both of these neurotransmitter systems may contribute to hyperactive pain processing, and thereby precipitate or maintain chronic pain.
Pain mitigation may be achieved best by enhancing both neurotransmitters simultaneously, less so by enhancing NE alone, and least by enhancing 5-HT alone.6 The ability to impact pain modulation would, therefore, depend on the degree to which an antidepressant capitalizes on both noradrenergic and serotonergic neurotransmission. Antidepressants commonly employed to manage pain are presented in Table 147,60,68,80-88 according to their primary neurotransmitter effects. Thus, the literature summarized above suggests that antidepressants that influence both NE and 5-HT transmission have greater analgesic effects than antidepressants with more specific effects, such as influencing 5-HT reuptake alone.80-85 It is unsurprising, therefore, that the SSRIs have not been demonstrated to be as consistently analgesic.47,60,68,80,86-88

Similarly, pharmacodynamic and pharmacokinetic differences within antidepressant classes may influence analgesic effectiveness. Simultaneous effects on NE and 5-HT are achieved at low doses with duloxetine and milnacipran. By contrast, 5-HT effects predominate at low doses for venlafaxine. To achieve pain-mitigating effects, higher doses of venlafaxine generally are required.89 Therefore, inconsistencies across studies regarding the analgesic benefits of venlafaxine may be attributable to variability in dosing; patients treated with lower doses may not have experienced sufficient NE effects to garner positive results.
Continue to: The differences in analgesic efficacy...
The differences in analgesic efficacy among specific TCAs may be understood in a similar fashion. Specifically, tertiary TCAs (imipramine and amitriptyline) inhibit both 5-HT and NE reuptake.6,90 Secondary amines (desipramine and nortriptyline) predominantly impact NE reuptake, possibly accounting for the lesser pain-mitigating benefit achieved with these agents, such as for treating neuropathic pain. Further, in vivo imipramine and amitriptyline are rapidly metabolized to secondary amines that are potent and selective NE reuptake inhibitors. In this way, the secondary amines may substantially lose the ability to modulate pain transmission because of the loss of concurrent 5-HT influences.90
Clinical pearls
The following practical points can help guide clinicians regarding the usefulness of antidepressants for pain management:
- Antidepressants can alleviate symptoms of depression and pain. The pain-mitigating effects of antidepressants are possible even among chronic pain patients who are not depressed. Antidepressants may confer benefits for chronic pain patients with depression and other comorbid conditions, such as somatic symptom and related disorders.
- Antidepressants are useful for select chronic pain states. Although the noradrenergic and serotonergic antidepressants (SNRIs and, to some extent, amitriptyline) appear to have efficacy for neuropathic pain and FM, the benefits of SSRIs appear to be less robust. On the other hand, SSRIs and TCAs may have potential benefit for patients with IBS. However, the results of meta-analyses are limited in the ability to provide information about which patients will best respond to which specific antidepressant or how well. Future research directed at identifying characteristics that can predict which patients are likely to benefit from one antidepressant vs another would help inform how best to tailor treatment to individual needs.
- The pain-mitigating effects of antidepressants often emerge early in the course of treatment (often before mood-elevating effects are observed). For example, in the case of amitriptyline, pain relief may be possible for some patients at doses generally lower than those required for mood-elevating effects. To date, there is limited information in the literature to determine what constitutes a sufficient duration of treatment, or when treatment should be modified.
- Failure to reduce pain should raise questions about whether the dose should be increased, an alternative agent should be tried, or combinations with other analgesic agents should be considered. Failure to achieve pain-mitigating effects with one antidepressant does not mean failure with others. Hence, failure to achieve desired effects with one agent might warrant an empirical trial with another agent. Presently, too few double-blind RCTs have been conducted to assess the pain-mitigating effects of other antidepressants (eg, bupropion and newer SNRIs such as desvenlafaxine and levomilnacipran). Meta-analysis of the analgesic effectiveness of these agents or comparisons to the efficacy of other antidepressant classes is, therefore, impossible at this time.
Because many chronic pain states are complex, patients will seldom experience clinically relevant benefit from any one intervention.53 The bigger implication for clinical research is to determine whether there is a sequence or combination of medication use that will provide overall better clinical effectiveness.53 Only limited data are available exploring the utility of combining pharmacologic approaches to address pain.91 For example, preliminary evidence suggests that combinations of complementary strategies, such as duloxetine combined with pregabalin, may result in significantly greater numbers of FM patients achieving ≥30% pain reduction compared with monotherapy with either agent alone or placebo.92
- Antidepressant selection may need to be based on medication-related adverse effect profiles and the potential for drug interactions. These factors are useful to consider in delineating multimodal treatment regimens for chronic pain in light of patients’ comorbidities and co-medication regimen. For example, the adverse effects of TCAs (anticholinergic and alpha-adrenergic influences) limit their utility for treating pain. Some of these effects can be more problematic in select populations, such as older adults or those with orthostatic difficulties, among others. TCAs are contraindicated in patients with closed-angle glaucoma, recent myocardial infarction, cardiac arrhythmias, poorly controlled seizures, or severe benign prostatic hypertrophy. Although the pain-mitigating effects of SNRIs have not been demonstrated to significantly exceed those of TCAs,68,93,94 SNRIs would offer an advantage of greater tolerability of adverse effects and relative safety in patients with comorbid medical conditions that would otherwise preclude TCA use. The adverse effects and common drug interactions associated with antidepressants are summarized in Table 295.

Conclusion
Chronic, nonmalignant pain conditions afflict many patients and significantly impair their ability to function. Because of heightened concerns related to the appropriateness of, and restricting inordinate access to, long-term opioid analgesics, clinicians need to explore the usefulness of co-analgesic agents, such as antidepressants. Significant comorbidities exist between psychiatric disorders and chronic pain, and psychiatrists are uniquely positioned to diagnose and treat psychiatric comorbidities, as well as pain, among their patients, especially since they understand the kinetics and dynamics of antidepressants.
Bottom Line
Antidepressants can alleviate symptoms of depression and pain. Noradrenergic and serotonergic antidepressants appear to have efficacy for pain associated with neuropathy and fibromyalgia, while selective serotonin reuptake inhibitors and tricyclic antidepressants may have benefit for patients with irritable bowel syndrome. However, evidence regarding which patients will best respond to which specific antidepressant is limited.
Continue to: Related Resources
Related Resources
- Williams AM, Knox ED. When to prescribe antidepressants to treat comorbid depression and pain disorders. Current Psychiatry. 2017;16(1):55-58.
- Maletic V, Demuri B. Chronic pain and depression: treatment of 2 culprits in common. Current Psychiatry. 2016;15(3):41,47-50,52.
Drug Brand Names
Amitriptyline • Elavil, Endep
Bupropion • Wellbutrin, Zyban
Carisoprodol • Rela, Soma
Cyclobenzaprine • Amrix, Flexeril
Desipramine • Norpramin
Desvenlafaxine • Pristiq
Duloxetine • Cymbalta
Fluoxetine • Prozac
Gabapentin • Horizant, Neurontin
Imipramine • Tofranil
Levomilnacipran • Fetzima
Methadone • Dolophine, Methadose
Milnacipran • Savella
Mirtazapine • Remeron
Nortriptyline • Pamelor
Paroxetine • Paxil
Pregabalin • Lyrica, Lyrica CR
Tapentadol • Nucynta
Tramadol • Ultram
Trazodone • Desyrel, Oleptro
Venlafaxine • Effexor
Warfarin • Coumadin, Jantoven
Approximately 55 years ago, tricyclic antidepressants (TCAs) began to be used to treat neuropathic pain.1 Eventually, clinical trials emerged suggesting the utility of TCAs for other chronic pain conditions, such as fibromyalgia (FM) and migraine prophylaxis. However, despite TCAs’ effectiveness in mitigating painful conditions, their adverse effects limited their use.
Pharmacologic advancements have led to the development of other antidepressant classes, including selective serotonin reuptake inhibitors (SSRIs) and serotonin-norepinephrine reuptake inhibitors (SNRIs), and the use of these agents has come to eclipse that of TCAs. In the realm of pain management, such developments have raised the hope of possible alternative co-analgesic agents that could avoid the adverse effects associated with TCAs. Some of these agents have demonstrated efficacy for managing chronic pain states, while others have demonstrated only limited utility.
This article provides a synopsis of systematic reviews and meta-analyses examining the role of antidepressant therapy for managing several chronic pain conditions, including pain associated with neuropathy, FM, headache, and irritable bowel syndrome (IBS). Because the literature base is rapidly evolving, it is necessary to revisit the information gleaned from clinical data with respect to treatment effectiveness, and to clarify how antidepressants might be positioned in the management of chronic pain.
The effectiveness of antidepressants for pain
The pathophysiologic processes that precipitate and maintain chronic pain conditions are complex (Box 12-10). The pain-mitigating effects of antidepressants can be thought of in terms of direct analgesic effects and indirect effects (Box 22,3,8,10,11-33).
Box 1
The pathophysiologic processes precipitating and maintaining chronic pain conditions are complex. Persistent and chronic pain results from changes in sensitivity within both ascending pathways (relaying pain information from the periphery to the spinal cord and brain) and descending pain pathways (functioning to modulate ascending pain information).2,3 Tissue damage or peripheral nerve injury can lead to a cascade of neuroplastic changes within the CNS, resulting in hyperexcitability within the ascending pain pathways.
The descending pain pathways consist of the midbrain periaqueductal gray area (PGA), the rostroventral medulla (RVM), and the dorsolateral pontomesencephalic tegmentum (DLPT). The axons of the RVM (the outflow of which is serotonergic) and DLPT (the outflow of which is noradrenergic) terminate in the dorsal horn of the spinal cord,4 and thereby dampen pain signals arising from the periphery. Diminished output from descending pain pathways can heighten the pain experience. Input from the cortex, hypothalamus, and amygdala (among other structures) converges upon the PGA, RVM and DLPT, and can influence the degree of pain modulation emerging from descending pathways. In this way, thoughts, appraisals, and mood are believed to comprise cognitive and affective modifiers of pain experiences.
Devising effective chronic pain treatment becomes challenging; multimodal treatment approaches often are advocated, including pharmacologic treatment with analgesics in combination with co-analgesic medications such as antidepressants. Although a description of multimodal treatment is beyond the scope of this article, such treatment also would encompass physical therapy, occupational therapy, and psychotherapeutic interventions to augment rehabilitative efforts and the functional capabilities of patients who struggle with persisting pain.
Although the direct pain-mitigating effects of antidepressants are not fully understood, it is believed that augmentation of monoamine neurotransmission from supraspinal nuclei (ie, the RVM and DLPT) modulate pain transmission from the periphery.5,6 In addition, there is evidence that some effects of tricyclic antidepressants can modulate several other functions that impact peripheral and central sensitization.7-10
During the last several decades, antidepressants have been used to address—and have demonstrated clinical utility for—a variety of chronic pain states. However, antidepressants are not a panacea; some chronic pain conditions are more responsive to antidepressants than are others. The chronic painful states most amenable to antidepressants are those that result primarily from a process of neural sensitization, as opposed to acute somatic or visceral nociception. Hence, several meta-analyses and evidence-based reviews have long suggested the usefulness of antidepressants for mitigating pain associated with neuropathy,34,35 FM,36,37 headache,38 and IBS.39,40
Box 2
The pain-mitigating effects of antidepressants can be thought of in terms of direct analgesic effects (impacting neurotransmission of descending pathways independent of influences on mood) and indirect effects (presumably impacting cortical and limbic output to the periaqueductal gray area, the rostroventral medulla, and the dorsolateral pontomesencephalic tegmentum brought about by improvement in mood and/or cognitive appraisals) (Figure2,3,8,10,11,15,20,22,28,29). Support for the direct analgesic effects has been garnered from initial empirical work that demonstrated pain relief among patients with pain who are not depressed. Additionally, among patients who have depression and experience pain, analgesia reportedly often occurs within 2 weeks, which is before antidepressant effects are appreciated,11-15 and, at least for some antidepressants, occurs at doses far lower than those required to produce mood-elevating effects.11,12,16
On the other hand, it is well established that significant comorbidities exist between chronic pain states and psychiatric disorders (eg, depression and somatic symptom and related disorders).17-21 There may be common physiological substrates underlying chronic pain and depression.20,22 There are bidirectional influences of limbic (affective) systems and those CNS structures involved in pain processing and integration. The effects of pain and depression are reciprocal; the presence of one makes the management of the other more challenging.23-27 Mood disturbances can, therefore, impact pain processing by acting as affective and cognitive amplifiers of pain by leading to catastrophizing, pain severity augmentation, poor coping with pain-related stress, etc.28,29 It is plausible that the mood-elevating effects of antidepressants can improve pain by indirect effects, by modulating limbic activity, which in turn, impacts coping, cognitive appraisals of pain, etc.
Patients with somatoform disorders (using pre-DSM-5 terminology) frequently present with chronic pain, often in multiple sites.19 Such patients are characterized by hypervigilance for, and a predisposition to focus on, physical sensations and to appraise these sensations as reflecting a pathological state.30 Neuroimaging studies have begun to identify those neural circuits involved in somatoform disorders, many of which act as cognitive and affective amplifiers of visceral-somatic sensory processing. Many of these neural circuits overlap, and interact with, those involved in pain processing.31 Antidepressants can mitigate the severity of unexplained physical complaints, including pain, among patients who somatize32,33; however, due to the heterogeneity of studies upon which this claim is based, the quality of the evidence is reportedly low.33 There is uncertainty whether, or to what extent, antidepressant benefits among patients who somatize are due to a direct impact on pain modulation, or indirect effects on mood or cognitive appraisals/perceptions.
Despite the uncertainties about the exact mechanisms through which antidepressants exert analgesic effects, antidepressants can be appropriately used to treat patients with selected chronic pain syndromes, regardless of whether or not the patient has a psychiatric comorbidity. For those patients with pain and psychiatric comorbidities, the benefits may be brought about via direct mechanisms, indirect mechanisms, or a combination of both.

Continue to: Neuropathic pain
Neuropathic pain
Several treatment guidelines advocate for the use of antidepressants for neuropathic pain.41-44 For decades, TCAs have been employed off-label to successfully treat many patients with neuropathic pain states. Early investigations suggested that TCAs were robustly efficacious in managing patients with neuropathy.45-48 Calculated number-needed-to-treat (NNT) values for TCAs were quite low (ie, reflecting that few patients would need to be treated to yield a positive response in one patient compared with placebo), and were comparable to, if not slightly better than, the NNTs generated for anticonvulsants and α2-δ ligands, such as gabapentin or pregabalin.45-48
Unfortunately, early studies involving TCAs conducted many years ago do not meet contemporary standards of methodological rigor; they featured relatively small samples of patients assessed for brief post-treatment intervals with variable outcome measures. Thus, the NNT values obtained in meta-analyses based on these studies may overestimate treatment benefits.49 Further, NNT values derived from meta-analyses tended to combine all drugs within a particular antidepressant class (eg, amitriptyline, nortriptyline, desipramine, and imipramine among the TCAs) employed at diverse doses. Taken together, these limitations raise questions about the results of those meta-analyses.
Subsequent meta-analyses, which employed strict criteria to eliminate data from studies with potential sources of bias and used a primary outcome of frequencies of patients reporting at least 30% pain reduction compared with a placebo-controlled sample, suggest that the effectiveness of TCAs as a class for treating neuropathic pain is not as compelling as once was thought. Meta-analyses of studies employing specific TCAs revealed that there was little evidence to support the use of desipramine,50 imipramine,51 or nortriptyline52 in managing diabetic neuropathy or postherpetic neuralgia. Studies evaluating amitriptyline (dose range 12.5 to 150 mg/d), found low-level evidence of effectiveness; the benefit was expected to be present for a small subset (approximately 25%) of patients with neuropathic pain.53
There is moderate-quality evidence that duloxetine (60 to 120 mg/d) can produce a ≥50% improvement in pain severity ratings among patients with diabetic peripheral neuropathy.54 Although head-to-head studies with other antidepressants are limited, it appears that duloxetine and amitriptyline have comparable efficacy, even though the NNTs for amitriptyline were derived from lower-quality studies than those for duloxetine. Duloxetine is the only antidepressant to receive FDA approval for managing diabetic neuropathy. By contrast, studies assessing the utility of venlafaxine in neuropathic pain comprised small samples for brief durations, which limits the ability to draw clear (unbiased) support for its usefulness.55
Given the diversity of pathophysiologic processes underlying the disturbances that cause neuropathic pain disorders, it is unsurprising that the effectiveness of amitriptyline and duloxetine were not generalizable to all neuropathic pain states. Although amitriptyline produced pain-mitigating effects in patients with diabetic neuropathy and post-herpetic neuralgia, and duloxetine mitigated pain among patients with diabetic neuropathy, there was no evidence to suggest their effectiveness in phantom limb pain or human immunodeficiency virus-related and spinal cord-related neuropathies.35,53,54,56-58
Continue to: Fibromyalgia
Fibromyalgia
As with the issues encountered in interpreting the effectiveness of antidepressants in neuropathic pain, interpreting results gleaned from clinical trials of antidepressants for treating FM are fraught with similar difficulties. Although amitriptyline has been a first-line treatment for FM for many years, the evidence upon which such recommendations were based consisted of low-level studies that had a significant potential for bias.59 Large randomized trials would offer more compelling data regarding the efficacy of amitriptyline, but the prohibitive costs of such studies makes it unlikely they will be conducted. Amitriptyline (25 to 50 mg/d) was effective in mitigating FM-related pain in a small percentage of patients studied, with an estimated NNT of 4.59 Adverse effects, often contributing to treatment discontinuation, were encountered more frequently among patients who received amitriptyline compared with placebo.
Selective serotonin reuptake inhibitors failed to demonstrate significant pain relief (estimated NNT of 10), or improvement in fatigue or sleep problems, even though the studies upon which such conclusions were based were low-level studies with a high potential for bias.60 Although SSRIs have limited utility for mitigating pain, they are still quite useful for reducing depression among patients with FM.60
By contrast, the SNRIs duloxetine and milnacipran provided clinically relevant benefit over placebo in the frequency of patients reporting pain relief of ≥30%, as well as patients’ global impression of change.61 These agents, however, failed to provide clinically relevant benefit over placebo in improving health-related quality of life, reducing sleep problems, or improving fatigue. Nonetheless, duloxetine and milnacipran are FDA-approved for managing pain in FM. Studies assessing the efficacy of venlafaxine in the treatment of FM to date have been limited by small sample sizes, inconsistent dosing, lack of a placebo control, and lack of blinding, which limits the ability to clearly delineate the role of venlafaxine in managing FM.62
Mirtazapine (15 to 45 mg/d) showed a clinically relevant benefit compared with placebo for participant-reported pain relief of ≥30% and sleep disturbances. There was no benefit in terms of participant-reported improvement of quality of life, fatigue, or negative mood.63 The evidence was considered to be of low quality overall.
Headache
Amitriptyline has been employed off-label to address headache prophylaxis since 1964.64 Compared with placebo, it is efficacious in ameliorating migraine frequency and intensity as well as the frequency of tension headache.65,66 However, SSRIs and SNRIs (venlafaxine) failed to produce significant reductions in migraine frequency or severity or the frequencies of tension headache when compared with placebo.67,68
Continue to: Irritable bowel syndrome
Irritable bowel syndrome
Early studies addressing antidepressant efficacy in IBS reveal inconsistencies. For example, whereas some suggest that TCAs are effective in mitigating chronic, severe abdominal pain,39,40 others concluded that TCAs failed to demonstrate a significant analgesic benefit.69 A recent meta-analysis that restricted analysis of efficacy to randomized controlled trials (RCTs) with more rigorous methodological adherence found that pain relief in IBS is possible with both TCAs as well as SSRIs. However, adverse effects were more commonly encountered with TCAs than with SSRIs. Some of the inconsistencies in treatment efficacy reported in early studies may be due to variations in responsiveness of subsets of IBS patients. Specifically, the utility of TCAs appears to be best among patients with diarrheal-type (as opposed to constipation-type) IBS, presumably due to TCAs’ anticholinergic effects, whereas SSRIs may provide more of a benefit for patients with predominantly constipation-type IBS.40,70
Other chronic pain conditions
Antidepressants have been used to assist in the management of several other pain conditions, including oral-facial pain, interstitial cystitis, non-cardiac chest pain, and others. The role of antidepressants for such conditions remains unclear due to limitations in the prevailing empirical work, such as few trials, small sample sizes, variations in outcome measures, and insufficient randomization and blinding.71-76 The interpretation of results from systematic reviews and meta-analyses is limited because of these shortcomings.77 Hence, it has not always been possible to determine whether, and to what extent, patients with such conditions may benefit from antidepressants.
Neuromodulatory effects and efficacy for pain
The interplay of norepinephrine (NE) and serotonin (5-HT) neurotransmitter systems and cellular mechanisms involved in the descending modulation of pain pathways is complex. Experimental animal models of pain modulation suggest that 5-HT can both inhibit as well as promote pain perception by different physiological mechanisms, in contrast to NE, which is predominately inhibitory. While 5-HT in the descending modulating system can inhibit pain transmission ascending to the brain from the periphery, it appears that an intact noradrenergic system is necessary for the inhibitory influences of the serotonergic system to be appreciated.16,78,79 Deficiencies in one or both of these neurotransmitter systems may contribute to hyperactive pain processing, and thereby precipitate or maintain chronic pain.
Pain mitigation may be achieved best by enhancing both neurotransmitters simultaneously, less so by enhancing NE alone, and least by enhancing 5-HT alone.6 The ability to impact pain modulation would, therefore, depend on the degree to which an antidepressant capitalizes on both noradrenergic and serotonergic neurotransmission. Antidepressants commonly employed to manage pain are presented in Table 147,60,68,80-88 according to their primary neurotransmitter effects. Thus, the literature summarized above suggests that antidepressants that influence both NE and 5-HT transmission have greater analgesic effects than antidepressants with more specific effects, such as influencing 5-HT reuptake alone.80-85 It is unsurprising, therefore, that the SSRIs have not been demonstrated to be as consistently analgesic.47,60,68,80,86-88

Similarly, pharmacodynamic and pharmacokinetic differences within antidepressant classes may influence analgesic effectiveness. Simultaneous effects on NE and 5-HT are achieved at low doses with duloxetine and milnacipran. By contrast, 5-HT effects predominate at low doses for venlafaxine. To achieve pain-mitigating effects, higher doses of venlafaxine generally are required.89 Therefore, inconsistencies across studies regarding the analgesic benefits of venlafaxine may be attributable to variability in dosing; patients treated with lower doses may not have experienced sufficient NE effects to garner positive results.
Continue to: The differences in analgesic efficacy...
The differences in analgesic efficacy among specific TCAs may be understood in a similar fashion. Specifically, tertiary TCAs (imipramine and amitriptyline) inhibit both 5-HT and NE reuptake.6,90 Secondary amines (desipramine and nortriptyline) predominantly impact NE reuptake, possibly accounting for the lesser pain-mitigating benefit achieved with these agents, such as for treating neuropathic pain. Further, in vivo imipramine and amitriptyline are rapidly metabolized to secondary amines that are potent and selective NE reuptake inhibitors. In this way, the secondary amines may substantially lose the ability to modulate pain transmission because of the loss of concurrent 5-HT influences.90
Clinical pearls
The following practical points can help guide clinicians regarding the usefulness of antidepressants for pain management:
- Antidepressants can alleviate symptoms of depression and pain. The pain-mitigating effects of antidepressants are possible even among chronic pain patients who are not depressed. Antidepressants may confer benefits for chronic pain patients with depression and other comorbid conditions, such as somatic symptom and related disorders.
- Antidepressants are useful for select chronic pain states. Although the noradrenergic and serotonergic antidepressants (SNRIs and, to some extent, amitriptyline) appear to have efficacy for neuropathic pain and FM, the benefits of SSRIs appear to be less robust. On the other hand, SSRIs and TCAs may have potential benefit for patients with IBS. However, the results of meta-analyses are limited in the ability to provide information about which patients will best respond to which specific antidepressant or how well. Future research directed at identifying characteristics that can predict which patients are likely to benefit from one antidepressant vs another would help inform how best to tailor treatment to individual needs.
- The pain-mitigating effects of antidepressants often emerge early in the course of treatment (often before mood-elevating effects are observed). For example, in the case of amitriptyline, pain relief may be possible for some patients at doses generally lower than those required for mood-elevating effects. To date, there is limited information in the literature to determine what constitutes a sufficient duration of treatment, or when treatment should be modified.
- Failure to reduce pain should raise questions about whether the dose should be increased, an alternative agent should be tried, or combinations with other analgesic agents should be considered. Failure to achieve pain-mitigating effects with one antidepressant does not mean failure with others. Hence, failure to achieve desired effects with one agent might warrant an empirical trial with another agent. Presently, too few double-blind RCTs have been conducted to assess the pain-mitigating effects of other antidepressants (eg, bupropion and newer SNRIs such as desvenlafaxine and levomilnacipran). Meta-analysis of the analgesic effectiveness of these agents or comparisons to the efficacy of other antidepressant classes is, therefore, impossible at this time.
Because many chronic pain states are complex, patients will seldom experience clinically relevant benefit from any one intervention.53 The bigger implication for clinical research is to determine whether there is a sequence or combination of medication use that will provide overall better clinical effectiveness.53 Only limited data are available exploring the utility of combining pharmacologic approaches to address pain.91 For example, preliminary evidence suggests that combinations of complementary strategies, such as duloxetine combined with pregabalin, may result in significantly greater numbers of FM patients achieving ≥30% pain reduction compared with monotherapy with either agent alone or placebo.92
- Antidepressant selection may need to be based on medication-related adverse effect profiles and the potential for drug interactions. These factors are useful to consider in delineating multimodal treatment regimens for chronic pain in light of patients’ comorbidities and co-medication regimen. For example, the adverse effects of TCAs (anticholinergic and alpha-adrenergic influences) limit their utility for treating pain. Some of these effects can be more problematic in select populations, such as older adults or those with orthostatic difficulties, among others. TCAs are contraindicated in patients with closed-angle glaucoma, recent myocardial infarction, cardiac arrhythmias, poorly controlled seizures, or severe benign prostatic hypertrophy. Although the pain-mitigating effects of SNRIs have not been demonstrated to significantly exceed those of TCAs,68,93,94 SNRIs would offer an advantage of greater tolerability of adverse effects and relative safety in patients with comorbid medical conditions that would otherwise preclude TCA use. The adverse effects and common drug interactions associated with antidepressants are summarized in Table 295.

Conclusion
Chronic, nonmalignant pain conditions afflict many patients and significantly impair their ability to function. Because of heightened concerns related to the appropriateness of, and restricting inordinate access to, long-term opioid analgesics, clinicians need to explore the usefulness of co-analgesic agents, such as antidepressants. Significant comorbidities exist between psychiatric disorders and chronic pain, and psychiatrists are uniquely positioned to diagnose and treat psychiatric comorbidities, as well as pain, among their patients, especially since they understand the kinetics and dynamics of antidepressants.
Bottom Line
Antidepressants can alleviate symptoms of depression and pain. Noradrenergic and serotonergic antidepressants appear to have efficacy for pain associated with neuropathy and fibromyalgia, while selective serotonin reuptake inhibitors and tricyclic antidepressants may have benefit for patients with irritable bowel syndrome. However, evidence regarding which patients will best respond to which specific antidepressant is limited.
Continue to: Related Resources
Related Resources
- Williams AM, Knox ED. When to prescribe antidepressants to treat comorbid depression and pain disorders. Current Psychiatry. 2017;16(1):55-58.
- Maletic V, Demuri B. Chronic pain and depression: treatment of 2 culprits in common. Current Psychiatry. 2016;15(3):41,47-50,52.
Drug Brand Names
Amitriptyline • Elavil, Endep
Bupropion • Wellbutrin, Zyban
Carisoprodol • Rela, Soma
Cyclobenzaprine • Amrix, Flexeril
Desipramine • Norpramin
Desvenlafaxine • Pristiq
Duloxetine • Cymbalta
Fluoxetine • Prozac
Gabapentin • Horizant, Neurontin
Imipramine • Tofranil
Levomilnacipran • Fetzima
Methadone • Dolophine, Methadose
Milnacipran • Savella
Mirtazapine • Remeron
Nortriptyline • Pamelor
Paroxetine • Paxil
Pregabalin • Lyrica, Lyrica CR
Tapentadol • Nucynta
Tramadol • Ultram
Trazodone • Desyrel, Oleptro
Venlafaxine • Effexor
Warfarin • Coumadin, Jantoven
1. Paoli F, Darcourt G, Cossa P. Preliminary note on the action of imipramine in painful states [in French]. Rev Neurol (Paris). 1960;102:503-504.
2. Fields HL, Heinricher MM, Mason P. Neurotransmitters in nociceptive modulatory circuits. Annu Rev Neurosci. 1991;14:219-245.
3. Hunt SP, Mantyh PW. The molecular dynamics of pain control. Nat Rev Neurosci. 2001;2(2):83-91.
4. Lamont LA, Tranquilli WJ, Grimm KA. Physiology of pain. Vet Clin North Am Small Anim Pract. 2000;30(4):703-728, v.
5. Fields HL, Basbaum AI, Heinricher MM. Central nervous system mechanisms of pain modulation. In: McMahon S, Koltzenburg M, eds. Wall and Melzack’s Textbook of Pain. 5th ed. Burlington, MA: Elsevier Health Sciences; 2005:125-142.
6. Marks DM, Shah MJ, Patkar AA, et al. Serotonin-norepinephrine reuptake inhibitors for pain control: premise and promise. Curr Neuropharmacol. 2009;7(4):331-336.
7. Baba H, Shimoji K, Yoshimura M. Norepinephrine facilitates inhibitory transmission in substantia gelatinosa of adult rat spinal cord (part 1): effects on axon terminals of GABAergic and glycinergic neurons. Anesthesiology. 2000;92(2):473-484.
8. Carter GT, Sullivan MD. Antidepressants in pain management. Curr Opin Investig Drugs. 2002;3(3):454-458.
9. Kawasaki Y, Kumamoto E, Furue H, et al. Alpha 2 adrenoceptor-mediated presynaptic inhibition of primary afferent glutamatergic transmission in rat substantia gelatinosa neurons. Anesthesiology. 2003;98(3):682-689.
10. McCleane G. Antidepressants as analgesics. CNS Drugs. 2008;22(2):139-156.
11. Ansari A. The efficacy of newer antidepressants in the treatment of chronic pain: a review of current literature. Harv Rev Psychiatry. 2000;7(5):257-277.
12. Egbunike IG, Chaffee BJ. Antidepressants in the management of chronic pain syndromes. Pharmacotherapy. 1990;10(4):262-270.
13. Fishbain DA. Evidence-based data on pain relief with antidepressants. Ann Med. 2000;32(5):305-316.
14. Fishbain DA, Detke MJ, Wernicke J, et al. The relationship between antidepressant and analgesic responses: findings from six placebo-controlled trials assessing the efficacy of duloxetine in patients with major depressive disorder. Curr Med Res Opin. 2008;24(11):3105-3115.
15. Harada E, Tokuoka H, Fujikoshi S, et al. Is duloxetine’s effect on painful physical symptoms in depression an indirect result of improvement of depressive symptoms? Pooled analyses of three randomized controlled trials. Pain. 2016;157(3):577-584.
16. Kehoe WA. Antidepressants for chronic pain: selection and dosing considerations. Am J Pain Med. 1993;3(4):161-165.
17. Damush TM, Kroenke K, Bair MJ, et al. Pain self-management training increases self-efficacy, self-management behaviours and pain and depression outcomes. Eur J Pain. 2016;20(2):1070-1078.
18. DeVeaugh-Geiss AM, West SL, Miller WC, et al. The adverse effects of comorbid pain on depression outcomes in primary care patients: results from the ARTIST trial. Pain Medicine. 2010;11(5):732-741.
19. Egloff N, Cámara RJ, von Känel R, et al. Hypersensitivity and hyperalgesia in somatoform pain disorders. Gen Hosp Psychiatry. 2014;36(3):284-290.
20. Goesling J, Clauw DW, Hassett AL. Pain and depression: an integrative review of neurobiological and psychological factors. Curr Psych Reports. 2013;15(12):421.
21. Kroenke K, Wu J, Bair MJ, et al. Reciprocal relationship between pain and depression: a 12-Month longitudinal analysis in primary care. J Pain. 2011;12(9):964-973.
22. Leo RJ. Chronic pain and comorbid depression. Curr Treat Options Neurol. 2005;7(5):403-412.
23. Bair MJ, Robinson RL, Eckert GJ, et al. Impact of pain on depression treatment response in primary care. Psychosom Med. 2004;66(1):17-22.
24. Karp JF, Scott J, Houck P, et al. Pain predicts longer time to remission during treatment of recurrent depression. J Clin Psychiatry. 2005;66(5):591-597.
25. Kroenke K, Shen J, Oxman TE, et al. Impact of pain on the outcomes of depression treatment: results from the RESPECT trial. Pain. 2008;134(1-2):209-215.
26. Mavandadi S, Ten Have TR, Katz IR, et al. Effect of depression treatment on depressive symptoms in older adulthood: the moderating role of pain. J Am Geriatr Soc. 2007;55(2):202-211.
27. Thielke SM, Fan MY, Sullivan M, et al. Pain limits the effectiveness of collaborative care for depression. Am J Geriatr Psychiatry. 2007;15(8):699-707.
28. Arnow BA, Hunkeler EM, Blasey CM, et al. Comorbid depression, chronic pain, and disability in primary care. Psychosom Med. 2006;68(2):262-268.
29. Demyttenaere K, Bonnewyn A, Bruffaerts R, et al. Comorbid painful physical symptoms and depression: Prevalence, work loss, and help seeking. J Affect Disord. 2006;92(2-3):185-193.
30. Nakao M, Barsky AJ. Clinical application of somatosensory amplification in psychosomatic medicine. Biopsychosoc Med. 2007;1:17.
31. Perez DL, Barsky AJ, Vago DR, et al. A neural circuit framework for somatosensory amplification in somatoform disorders. J Neuropsychiatry Clin Neurosci. 2015;27(1):e40-e50.
32. Fishbain DA, Cutler RB, Rosomoff HL, et al. Do antidepressants have an analgesic effect in psychogenic pain and somatoform pain disorder? A meta-analysis. Psychosom Med. 1998;60(4):503-509.
33. Kleinstäuber M, Witthöft M, Steffanowski A, et al. Pharmacological interventions for somatoform disorders in adults. Cochrane Database Syst Rev. 2014;(11):CD010628.
34. Collins SL, Moore RA, McQuay HJ, et al. Antidepressants and anticonvulsants for diabetic neuropathy and postherpetic neuralgia: a quantitative systematic review. J Pain Symptom Manage. 2000;20(6):449-458.
35. Saarto T, Wiffen PJ. Antidepressants for neuropathic pain: a Cochrane review. J Neurol Neurosurg Psychiatry. 2010;81(12):1372-1373.
36. Arnold LM, Keck PE, Welge JA. Antidepressant treatment of fibromyalgia. A meta-analysis and review. Psychosomatics. 2000;41(2):104-113.
37. O’Malley PG, Balden E, Tomkins G, et al. Treatment of fibromyalgia with antidepressants: a meta-analysis. J Gen Intern Med. 2000;15(9):659-666.
38. Tomkins GE, Jackson JL, O’Malley PG, et al. Treatment of chronic headache with antidepressants: a meta-analysis. Am J Med. 2001;111(1):54-63.
39. Jackson JL, O’Malley PG, Tomkins G, et al. Treatment of functional gastrointestinal disorders with antidepressant medications: a meta-analysis. Am J Med. 2000;108(1):65-72.
40. Lesbros-Pantoflickova D, Michetti P, Fried M et al. Meta-analysis: the treatment of irritable bowel syndrome. Aliment Pharmacol Ther. 2004;20(11-12):1253-1269.
41. Centre for Clinical Practice at NICE (UK). Neuropathic pain: the pharmacological management of neuropathic pain in adults in non-specialist settings. London, UK: National Institute for Health and Care Excellence, (UK); 2013.
42. O’Connor AB, Dworkin RH. Treatment of neuropathic pain: an overview of recent guidelines. Am J Med. 2009;122(suppl 10):S22-S32.
43. Moulin D, Boulanger A, Clark AJ, et al; Canadian Pain Society. Pharmacological management of chronic neuropathic pain: revised consensus statement from the Canadian Pain Society. Pain Res Manag. 2014;19(6):328-35.
44. Mu A, Weinberg E, Moulin DE, et al. Pharmacologic management of chronic neuropathic pain: Review of the Canadian Pain Society consensus statement. Can Fam Physician. 2017;63(11):844-852.
45. Finnerup NB, Otto M, McQuay HJ, et al. Algorithm for neuropathic pain treatment: an evidence based proposal. Pain. 2005;118(3):289-305.
46. Hempenstall K, Nurmikko TJ, Johnson RW, et al. Analgesic therapy in postherpetic neuralgia: a quantitative systematic review. PLoS Med. 2005;2(7):e164.
47. Sindrup SH, Jensen TS. Efficacy of pharmacological treatments of neuropathic pain: an update and effect related to mechanism of drug action. Pain. 1999;83(3):389-400.
48. Wu CL, Raja SN. An update on the treatment of postherpetic neuralgia. J Pain. 2008;9(suppl 1):S19-S30.
49. Kroenke K, Krebs EE, Bair MJ. Pharmacotherapy of chronic pain: a synthesis of recommendations from systematic reviews. Gen Hosp Psychiatry. 2009;31(3):206-219.
50. Hearn L, Moore RA, Derry S, et al. Desipramine for neuropathic pain in adults. Cochrane Database Syst Rev. 2014;(9):CD011003.
51. Hearn L, Derry S, Phillips T, et al. Imipramine for neuropathic pain in adults. Cochrane Database Syst Rev. 2014;(5):CD010769.
52. Derry S, Wiffen PJ, Aldington D, et al. Nortriptyline for neuropathic pain in adults. Cochrane Database Syst Rev. 2015;1:CD011209.
53. Moore R, Derry S, Aldington D, et al. Amitriptyline for neuropathic pain in adults. Cochrane Database Syst Rev. 2015;(7):CD008242.
54. Lunn MP, Hughes RA, Wiffen PJ. Duloxetine for treating painful neuropathy, chronic pain or fibromyalgia. Cochrane Database Syst Rev. 2014;(1):CD007115.
55. Gallagher HC, Gallagher RM, Butler M, et al. Venlafaxine for neuropathic pain in adults. Cochrane Database Syst Rev. 2015;(8):CD011091.
56. Alviar MJ, Hale T, Dungca M. Pharmacologic interventions for treating phantom limb pain. Cochrane Database Syst Rev. 2016;10:CD006380.
57. Dinat N, Marinda E, Moch S, et al. Randomized, Double-Blind, Crossover Trial of Amitriptyline for Analgesia in Painful HIV-Associated Sensory Neuropathy. PLoS One. 2015;10(5):e0126297. doi: 10.1371/journal.pone.0126297.eCollection 2015.
58. Mehta S, McIntyre A, Janzen S, et al; Spinal Cord Injury Rehabilitation Evidence Team. Systematic review of pharmacologic treatments of pain after spinal cord injury: an update. Arch Phys Med Rehabil. 2016;97(8):1381-1391.e1.
59. Moore RA, Derry S, Aldington D, et al. Amitriptyline for neuropathic pain and fibromyalgia in adults. Cochrane Database Syst Rev. 2012;(12):CD008242..
60. Walitt B, Urrútia G, Nishishinya MB, et al. Selective serotonin reuptake inhibitors for fibromyalgia syndrome. Cochrane Database Syst Rev. 2015;(6):CD011735.
61. Welsch P, Üçeyler N, Klose P, et al. Serotonin and noradrenaline reuptake inhibitors (SNRIs) for fibromyalgia. Cochrane Database Syst Rev. 2018;(2):CD010292.
62. VanderWeide LA, Smith SM, Trinkley KE. A systematic review of the efficacy of venlafaxine for the treatment of fibromyalgia. J Clin Pharm Ther. 2015;40(1):1-6.
63. Welsch P, Bernardy K, Derry S, et al. Mirtazapine for fibromyalgia in adults. Cochrane Database Syst Rev. 2018;(8):CD012708.
64. Lance JW, Curran DA. Treatment of chronic tension headache. Lancet. 1964;283(7345):1236-1239.
65. Jackson JL, William S, Laura S, et al. Tricyclic antidepressants and headaches: systematic review and meta-analysis. BMJ. 2010;341:c5222. doi: https://doi.org/10.1136/bmj.c5222
66. Xu XM, Liu Y, Dong MX, et al. Tricyclic antidepressants for preventing migraine in adults. Medicine. 2017;96(22):e6989. doi: 10.1097/MD.0000000000006989.
67. Banzi R, Cusi C, Randazzo C, et al. Selective serotonin reuptake inhibitors (SSRIs) and serotonin-norepinephrine reuptake inhibitors (SNRIs) for the prevention of migraine in adults. Cochrane Database Syst Rev. 2015;(4):CD002919.
68. Banzi R, Cusi C, Randazzo C, et al. Selective serotonin reuptake inhibitors (SSRIs) and serotonin-norepinephrine reuptake inhibitors (SNRIs) for the prevention of tension-type headache in adults. Cochrane Database Syst Rev. 2015;(5):CD011681.
69. Quartero AO, Meineche-Schmidt V, Muris J, et al. Bulking agents, antispasmodic and antidepressant medication for the treatment of irritable bowel syndrome. Cochrane Database Syst Rev. 2005;(2):CD003460.
70. Ford AC, Talley NJ, Schoenfeld PS, et al. Efficacy of antidepressants and psychological therapies in irritable bowel syndrome: systematic review and meta-analysis. Gut. 2009;58(3):367-378.
71. Coss-Adame E, Erdogan A, Rao SS. Treatment of esophageal (noncardiac) chest pain: an expert review. Clin Gastroenterol Hepatol. 2014;12(8):1224-1245.
72. Kelada E, Jones A. Interstitial cystitis. Arch Gynecol Obstet. 2007;275(4):223-229.
73. Leo RJ, Dewani S. A systematic review of the utility of antidepressant pharmacotherapy in the treatment of vulvodynia pain. J Sex Med. 2013;10(10):2497-2505.
74. McMillan R, Forssell H, Buchanan JA, et al. Interventions for treating burning mouth syndrome. Cochrane Database Syst Rev. 2016;11:CD002779.
75. Patel DN. Inconclusive results of a systematic review of efficacy of antidepressants on orofacial pain disorders. Evid Based Dent. 2013;14(2):55-56.
76. Wang W, Sun YH, Wang YY, et al. Treatment of functional chest pain with antidepressants: a meta-analysis. Pain Physician. 2012;15(2):E131-E142.
77. Lavis JN. How can we support the use of systematic reviews in policymaking? PLoS Med. 2009;6(11):e1000141. doi: 10.1371/journal.pmed.1000141.
78. Sorkin L. Nociceptive transmission within the spinal cord. Mt Sinai J Med. 1991;58(3):208-216.
79. Yokogawa F, Kiuchi Y, Ishikawa Y, et al. An investigation of monoamine receptors involved in antinociceptive effects of antidepressants. Anesth Analg. 2002;95(1):163-168, table of contents.
80. Lynch ME. Antidepressants as analgesics: a review of randomized controlled trials. J Psychiatry Neurosci. 2001;26(1):30-36.
81. Max MB. Treatment of post-herpetic neuralgia: antidepressants. Ann Neurol. 1994;35(suppl):S50-S53.
82. Max MB, Lynch SA, Muir J, et al. Effects of desipramine, amitriptyline, and fluoxetine on pain in diabetic neuropathy. N Engl J Med. 1992;326(19):1250-1256.
83. McQuay HJ, Tramèr M, Nye BA, et al. A systematic review of antidepressants in neuropathic pain. Pain. 1996;68(2-3):217-227.
84. Mochizucki D. Serotonin and noradrenaline reuptake inhibitors in animal models of pain. Hum Psychopharmacol Clin Exp. 2004;19(suppl 1):15-19.
85. Sussman N. SNRIs versus SSRIs: mechanisms of action in treating depression and painful physical symptoms. Primary Care Companion J Clin Psychiatry. 2003;5(suppl 7):19-26.
86. Bundeff AW, Woodis CB. Selective serotonin reuptake inhibitors for the treatment of irritable bowel syndrome. Ann Pharmacother. 2014;48(6):777-784.
87. Jung AC, Staiger T, Sullivan M. The efficacy of selective serotonin reuptake inhibitors for the management of chronic pain. J Gen Intern Med. 1997;12(6):384-389.
88. Xie C, Tang Y, Wang Y, et al. Efficacy and safety of antidepressants for the treatment of irritable bowel syndrome: a meta-analysis. PLoS One. 2015;10(8):e0127815. doi: 10.1371/journal.pone.0127815. eCollection 2015.
89. Zijlstra TR , Barendregt PJ , van de Laar MA. Venlafaxine in fibromyalgia: results of a randomized, placebo-controlled, double-blind trial. Arthritis Rheum. 2002;46(suppl 9):S105.
90. Bymaster FP, Dreshfield-Ahmad LJ, Threlkeld PG. Comparative affinity of duloxetine and venlafaxine for serotonin and norepinephrine transporters in vitro and in vivo, human serotonin receptor subtypes, and other neuronal receptors. Neuropsychopharmacology. 2001;25(6):871-880.
91. Thorpe J, Shum B, Moore RA, et al. Combination pharmacotherapy for the treatment of fibromyalgia in adults. Cochrane Database Syst Rev. 2018;(2):CD010585.
92. Gilron I, Chaparro LE, Tu D, et al. Combination of pregabalin with duloxetine for fibromyalgia: a randomized controlled trial. Pain. 2016;157(7):1532-1540.
93. Häuser W, Petzke F, Üçeyler N, et al. Comparative efficacy and acceptability of amitriptyline, duloxetine and milnacipran in fibromyalgia syndrome: a systematic review with meta-analysis. Rheumatology (Oxford). 2011;50(3):532-543.
94. Hossain SM, Hussain SM, Ekram AR. Duloxetine in painful diabetic neuropathy: a systematic review. Clin J Pain. 2016;32(11):1005-1010.
95. Riediger C, Schuster T, Barlinn K, et al. Adverse effects of antidepressants for chronic pain: a systematic review and meta-analysis. Front Neurol. 2017;8:307.
1. Paoli F, Darcourt G, Cossa P. Preliminary note on the action of imipramine in painful states [in French]. Rev Neurol (Paris). 1960;102:503-504.
2. Fields HL, Heinricher MM, Mason P. Neurotransmitters in nociceptive modulatory circuits. Annu Rev Neurosci. 1991;14:219-245.
3. Hunt SP, Mantyh PW. The molecular dynamics of pain control. Nat Rev Neurosci. 2001;2(2):83-91.
4. Lamont LA, Tranquilli WJ, Grimm KA. Physiology of pain. Vet Clin North Am Small Anim Pract. 2000;30(4):703-728, v.
5. Fields HL, Basbaum AI, Heinricher MM. Central nervous system mechanisms of pain modulation. In: McMahon S, Koltzenburg M, eds. Wall and Melzack’s Textbook of Pain. 5th ed. Burlington, MA: Elsevier Health Sciences; 2005:125-142.
6. Marks DM, Shah MJ, Patkar AA, et al. Serotonin-norepinephrine reuptake inhibitors for pain control: premise and promise. Curr Neuropharmacol. 2009;7(4):331-336.
7. Baba H, Shimoji K, Yoshimura M. Norepinephrine facilitates inhibitory transmission in substantia gelatinosa of adult rat spinal cord (part 1): effects on axon terminals of GABAergic and glycinergic neurons. Anesthesiology. 2000;92(2):473-484.
8. Carter GT, Sullivan MD. Antidepressants in pain management. Curr Opin Investig Drugs. 2002;3(3):454-458.
9. Kawasaki Y, Kumamoto E, Furue H, et al. Alpha 2 adrenoceptor-mediated presynaptic inhibition of primary afferent glutamatergic transmission in rat substantia gelatinosa neurons. Anesthesiology. 2003;98(3):682-689.
10. McCleane G. Antidepressants as analgesics. CNS Drugs. 2008;22(2):139-156.
11. Ansari A. The efficacy of newer antidepressants in the treatment of chronic pain: a review of current literature. Harv Rev Psychiatry. 2000;7(5):257-277.
12. Egbunike IG, Chaffee BJ. Antidepressants in the management of chronic pain syndromes. Pharmacotherapy. 1990;10(4):262-270.
13. Fishbain DA. Evidence-based data on pain relief with antidepressants. Ann Med. 2000;32(5):305-316.
14. Fishbain DA, Detke MJ, Wernicke J, et al. The relationship between antidepressant and analgesic responses: findings from six placebo-controlled trials assessing the efficacy of duloxetine in patients with major depressive disorder. Curr Med Res Opin. 2008;24(11):3105-3115.
15. Harada E, Tokuoka H, Fujikoshi S, et al. Is duloxetine’s effect on painful physical symptoms in depression an indirect result of improvement of depressive symptoms? Pooled analyses of three randomized controlled trials. Pain. 2016;157(3):577-584.
16. Kehoe WA. Antidepressants for chronic pain: selection and dosing considerations. Am J Pain Med. 1993;3(4):161-165.
17. Damush TM, Kroenke K, Bair MJ, et al. Pain self-management training increases self-efficacy, self-management behaviours and pain and depression outcomes. Eur J Pain. 2016;20(2):1070-1078.
18. DeVeaugh-Geiss AM, West SL, Miller WC, et al. The adverse effects of comorbid pain on depression outcomes in primary care patients: results from the ARTIST trial. Pain Medicine. 2010;11(5):732-741.
19. Egloff N, Cámara RJ, von Känel R, et al. Hypersensitivity and hyperalgesia in somatoform pain disorders. Gen Hosp Psychiatry. 2014;36(3):284-290.
20. Goesling J, Clauw DW, Hassett AL. Pain and depression: an integrative review of neurobiological and psychological factors. Curr Psych Reports. 2013;15(12):421.
21. Kroenke K, Wu J, Bair MJ, et al. Reciprocal relationship between pain and depression: a 12-Month longitudinal analysis in primary care. J Pain. 2011;12(9):964-973.
22. Leo RJ. Chronic pain and comorbid depression. Curr Treat Options Neurol. 2005;7(5):403-412.
23. Bair MJ, Robinson RL, Eckert GJ, et al. Impact of pain on depression treatment response in primary care. Psychosom Med. 2004;66(1):17-22.
24. Karp JF, Scott J, Houck P, et al. Pain predicts longer time to remission during treatment of recurrent depression. J Clin Psychiatry. 2005;66(5):591-597.
25. Kroenke K, Shen J, Oxman TE, et al. Impact of pain on the outcomes of depression treatment: results from the RESPECT trial. Pain. 2008;134(1-2):209-215.
26. Mavandadi S, Ten Have TR, Katz IR, et al. Effect of depression treatment on depressive symptoms in older adulthood: the moderating role of pain. J Am Geriatr Soc. 2007;55(2):202-211.
27. Thielke SM, Fan MY, Sullivan M, et al. Pain limits the effectiveness of collaborative care for depression. Am J Geriatr Psychiatry. 2007;15(8):699-707.
28. Arnow BA, Hunkeler EM, Blasey CM, et al. Comorbid depression, chronic pain, and disability in primary care. Psychosom Med. 2006;68(2):262-268.
29. Demyttenaere K, Bonnewyn A, Bruffaerts R, et al. Comorbid painful physical symptoms and depression: Prevalence, work loss, and help seeking. J Affect Disord. 2006;92(2-3):185-193.
30. Nakao M, Barsky AJ. Clinical application of somatosensory amplification in psychosomatic medicine. Biopsychosoc Med. 2007;1:17.
31. Perez DL, Barsky AJ, Vago DR, et al. A neural circuit framework for somatosensory amplification in somatoform disorders. J Neuropsychiatry Clin Neurosci. 2015;27(1):e40-e50.
32. Fishbain DA, Cutler RB, Rosomoff HL, et al. Do antidepressants have an analgesic effect in psychogenic pain and somatoform pain disorder? A meta-analysis. Psychosom Med. 1998;60(4):503-509.
33. Kleinstäuber M, Witthöft M, Steffanowski A, et al. Pharmacological interventions for somatoform disorders in adults. Cochrane Database Syst Rev. 2014;(11):CD010628.
34. Collins SL, Moore RA, McQuay HJ, et al. Antidepressants and anticonvulsants for diabetic neuropathy and postherpetic neuralgia: a quantitative systematic review. J Pain Symptom Manage. 2000;20(6):449-458.
35. Saarto T, Wiffen PJ. Antidepressants for neuropathic pain: a Cochrane review. J Neurol Neurosurg Psychiatry. 2010;81(12):1372-1373.
36. Arnold LM, Keck PE, Welge JA. Antidepressant treatment of fibromyalgia. A meta-analysis and review. Psychosomatics. 2000;41(2):104-113.
37. O’Malley PG, Balden E, Tomkins G, et al. Treatment of fibromyalgia with antidepressants: a meta-analysis. J Gen Intern Med. 2000;15(9):659-666.
38. Tomkins GE, Jackson JL, O’Malley PG, et al. Treatment of chronic headache with antidepressants: a meta-analysis. Am J Med. 2001;111(1):54-63.
39. Jackson JL, O’Malley PG, Tomkins G, et al. Treatment of functional gastrointestinal disorders with antidepressant medications: a meta-analysis. Am J Med. 2000;108(1):65-72.
40. Lesbros-Pantoflickova D, Michetti P, Fried M et al. Meta-analysis: the treatment of irritable bowel syndrome. Aliment Pharmacol Ther. 2004;20(11-12):1253-1269.
41. Centre for Clinical Practice at NICE (UK). Neuropathic pain: the pharmacological management of neuropathic pain in adults in non-specialist settings. London, UK: National Institute for Health and Care Excellence, (UK); 2013.
42. O’Connor AB, Dworkin RH. Treatment of neuropathic pain: an overview of recent guidelines. Am J Med. 2009;122(suppl 10):S22-S32.
43. Moulin D, Boulanger A, Clark AJ, et al; Canadian Pain Society. Pharmacological management of chronic neuropathic pain: revised consensus statement from the Canadian Pain Society. Pain Res Manag. 2014;19(6):328-35.
44. Mu A, Weinberg E, Moulin DE, et al. Pharmacologic management of chronic neuropathic pain: Review of the Canadian Pain Society consensus statement. Can Fam Physician. 2017;63(11):844-852.
45. Finnerup NB, Otto M, McQuay HJ, et al. Algorithm for neuropathic pain treatment: an evidence based proposal. Pain. 2005;118(3):289-305.
46. Hempenstall K, Nurmikko TJ, Johnson RW, et al. Analgesic therapy in postherpetic neuralgia: a quantitative systematic review. PLoS Med. 2005;2(7):e164.
47. Sindrup SH, Jensen TS. Efficacy of pharmacological treatments of neuropathic pain: an update and effect related to mechanism of drug action. Pain. 1999;83(3):389-400.
48. Wu CL, Raja SN. An update on the treatment of postherpetic neuralgia. J Pain. 2008;9(suppl 1):S19-S30.
49. Kroenke K, Krebs EE, Bair MJ. Pharmacotherapy of chronic pain: a synthesis of recommendations from systematic reviews. Gen Hosp Psychiatry. 2009;31(3):206-219.
50. Hearn L, Moore RA, Derry S, et al. Desipramine for neuropathic pain in adults. Cochrane Database Syst Rev. 2014;(9):CD011003.
51. Hearn L, Derry S, Phillips T, et al. Imipramine for neuropathic pain in adults. Cochrane Database Syst Rev. 2014;(5):CD010769.
52. Derry S, Wiffen PJ, Aldington D, et al. Nortriptyline for neuropathic pain in adults. Cochrane Database Syst Rev. 2015;1:CD011209.
53. Moore R, Derry S, Aldington D, et al. Amitriptyline for neuropathic pain in adults. Cochrane Database Syst Rev. 2015;(7):CD008242.
54. Lunn MP, Hughes RA, Wiffen PJ. Duloxetine for treating painful neuropathy, chronic pain or fibromyalgia. Cochrane Database Syst Rev. 2014;(1):CD007115.
55. Gallagher HC, Gallagher RM, Butler M, et al. Venlafaxine for neuropathic pain in adults. Cochrane Database Syst Rev. 2015;(8):CD011091.
56. Alviar MJ, Hale T, Dungca M. Pharmacologic interventions for treating phantom limb pain. Cochrane Database Syst Rev. 2016;10:CD006380.
57. Dinat N, Marinda E, Moch S, et al. Randomized, Double-Blind, Crossover Trial of Amitriptyline for Analgesia in Painful HIV-Associated Sensory Neuropathy. PLoS One. 2015;10(5):e0126297. doi: 10.1371/journal.pone.0126297.eCollection 2015.
58. Mehta S, McIntyre A, Janzen S, et al; Spinal Cord Injury Rehabilitation Evidence Team. Systematic review of pharmacologic treatments of pain after spinal cord injury: an update. Arch Phys Med Rehabil. 2016;97(8):1381-1391.e1.
59. Moore RA, Derry S, Aldington D, et al. Amitriptyline for neuropathic pain and fibromyalgia in adults. Cochrane Database Syst Rev. 2012;(12):CD008242..
60. Walitt B, Urrútia G, Nishishinya MB, et al. Selective serotonin reuptake inhibitors for fibromyalgia syndrome. Cochrane Database Syst Rev. 2015;(6):CD011735.
61. Welsch P, Üçeyler N, Klose P, et al. Serotonin and noradrenaline reuptake inhibitors (SNRIs) for fibromyalgia. Cochrane Database Syst Rev. 2018;(2):CD010292.
62. VanderWeide LA, Smith SM, Trinkley KE. A systematic review of the efficacy of venlafaxine for the treatment of fibromyalgia. J Clin Pharm Ther. 2015;40(1):1-6.
63. Welsch P, Bernardy K, Derry S, et al. Mirtazapine for fibromyalgia in adults. Cochrane Database Syst Rev. 2018;(8):CD012708.
64. Lance JW, Curran DA. Treatment of chronic tension headache. Lancet. 1964;283(7345):1236-1239.
65. Jackson JL, William S, Laura S, et al. Tricyclic antidepressants and headaches: systematic review and meta-analysis. BMJ. 2010;341:c5222. doi: https://doi.org/10.1136/bmj.c5222
66. Xu XM, Liu Y, Dong MX, et al. Tricyclic antidepressants for preventing migraine in adults. Medicine. 2017;96(22):e6989. doi: 10.1097/MD.0000000000006989.
67. Banzi R, Cusi C, Randazzo C, et al. Selective serotonin reuptake inhibitors (SSRIs) and serotonin-norepinephrine reuptake inhibitors (SNRIs) for the prevention of migraine in adults. Cochrane Database Syst Rev. 2015;(4):CD002919.
68. Banzi R, Cusi C, Randazzo C, et al. Selective serotonin reuptake inhibitors (SSRIs) and serotonin-norepinephrine reuptake inhibitors (SNRIs) for the prevention of tension-type headache in adults. Cochrane Database Syst Rev. 2015;(5):CD011681.
69. Quartero AO, Meineche-Schmidt V, Muris J, et al. Bulking agents, antispasmodic and antidepressant medication for the treatment of irritable bowel syndrome. Cochrane Database Syst Rev. 2005;(2):CD003460.
70. Ford AC, Talley NJ, Schoenfeld PS, et al. Efficacy of antidepressants and psychological therapies in irritable bowel syndrome: systematic review and meta-analysis. Gut. 2009;58(3):367-378.
71. Coss-Adame E, Erdogan A, Rao SS. Treatment of esophageal (noncardiac) chest pain: an expert review. Clin Gastroenterol Hepatol. 2014;12(8):1224-1245.
72. Kelada E, Jones A. Interstitial cystitis. Arch Gynecol Obstet. 2007;275(4):223-229.
73. Leo RJ, Dewani S. A systematic review of the utility of antidepressant pharmacotherapy in the treatment of vulvodynia pain. J Sex Med. 2013;10(10):2497-2505.
74. McMillan R, Forssell H, Buchanan JA, et al. Interventions for treating burning mouth syndrome. Cochrane Database Syst Rev. 2016;11:CD002779.
75. Patel DN. Inconclusive results of a systematic review of efficacy of antidepressants on orofacial pain disorders. Evid Based Dent. 2013;14(2):55-56.
76. Wang W, Sun YH, Wang YY, et al. Treatment of functional chest pain with antidepressants: a meta-analysis. Pain Physician. 2012;15(2):E131-E142.
77. Lavis JN. How can we support the use of systematic reviews in policymaking? PLoS Med. 2009;6(11):e1000141. doi: 10.1371/journal.pmed.1000141.
78. Sorkin L. Nociceptive transmission within the spinal cord. Mt Sinai J Med. 1991;58(3):208-216.
79. Yokogawa F, Kiuchi Y, Ishikawa Y, et al. An investigation of monoamine receptors involved in antinociceptive effects of antidepressants. Anesth Analg. 2002;95(1):163-168, table of contents.
80. Lynch ME. Antidepressants as analgesics: a review of randomized controlled trials. J Psychiatry Neurosci. 2001;26(1):30-36.
81. Max MB. Treatment of post-herpetic neuralgia: antidepressants. Ann Neurol. 1994;35(suppl):S50-S53.
82. Max MB, Lynch SA, Muir J, et al. Effects of desipramine, amitriptyline, and fluoxetine on pain in diabetic neuropathy. N Engl J Med. 1992;326(19):1250-1256.
83. McQuay HJ, Tramèr M, Nye BA, et al. A systematic review of antidepressants in neuropathic pain. Pain. 1996;68(2-3):217-227.
84. Mochizucki D. Serotonin and noradrenaline reuptake inhibitors in animal models of pain. Hum Psychopharmacol Clin Exp. 2004;19(suppl 1):15-19.
85. Sussman N. SNRIs versus SSRIs: mechanisms of action in treating depression and painful physical symptoms. Primary Care Companion J Clin Psychiatry. 2003;5(suppl 7):19-26.
86. Bundeff AW, Woodis CB. Selective serotonin reuptake inhibitors for the treatment of irritable bowel syndrome. Ann Pharmacother. 2014;48(6):777-784.
87. Jung AC, Staiger T, Sullivan M. The efficacy of selective serotonin reuptake inhibitors for the management of chronic pain. J Gen Intern Med. 1997;12(6):384-389.
88. Xie C, Tang Y, Wang Y, et al. Efficacy and safety of antidepressants for the treatment of irritable bowel syndrome: a meta-analysis. PLoS One. 2015;10(8):e0127815. doi: 10.1371/journal.pone.0127815. eCollection 2015.
89. Zijlstra TR , Barendregt PJ , van de Laar MA. Venlafaxine in fibromyalgia: results of a randomized, placebo-controlled, double-blind trial. Arthritis Rheum. 2002;46(suppl 9):S105.
90. Bymaster FP, Dreshfield-Ahmad LJ, Threlkeld PG. Comparative affinity of duloxetine and venlafaxine for serotonin and norepinephrine transporters in vitro and in vivo, human serotonin receptor subtypes, and other neuronal receptors. Neuropsychopharmacology. 2001;25(6):871-880.
91. Thorpe J, Shum B, Moore RA, et al. Combination pharmacotherapy for the treatment of fibromyalgia in adults. Cochrane Database Syst Rev. 2018;(2):CD010585.
92. Gilron I, Chaparro LE, Tu D, et al. Combination of pregabalin with duloxetine for fibromyalgia: a randomized controlled trial. Pain. 2016;157(7):1532-1540.
93. Häuser W, Petzke F, Üçeyler N, et al. Comparative efficacy and acceptability of amitriptyline, duloxetine and milnacipran in fibromyalgia syndrome: a systematic review with meta-analysis. Rheumatology (Oxford). 2011;50(3):532-543.
94. Hossain SM, Hussain SM, Ekram AR. Duloxetine in painful diabetic neuropathy: a systematic review. Clin J Pain. 2016;32(11):1005-1010.
95. Riediger C, Schuster T, Barlinn K, et al. Adverse effects of antidepressants for chronic pain: a systematic review and meta-analysis. Front Neurol. 2017;8:307.
Clinical benefits persist 5 years after thymectomy for myasthenia gravis
Thymectomy may continue to benefit patients with myasthenia gravis 5 years after the procedure, according to an extension study published in Lancet Neurology.
The study evaluated the clinical status, medication requirements, and adverse events of patients with myasthenia gravis who completed a randomized controlled trial of thymectomy plus prednisone versus prednisone alone and agreed to participate in a rater-blinded 2-year extension.
“Thymectomy within the first few years of the disease course in addition to prednisone therapy confers benefits that persist for 5 years ... in patients with generalized nonthymomatous myasthenia gravis,” said lead study author Gil I. Wolfe, MD, chair of the department of neurology at the University at Buffalo in New York, and his research colleagues. “Results from the extension study provide further support for the use of thymectomy in management of myasthenia gravis and should encourage serious consideration of this treatment option in discussions between clinicians and their patients,” they wrote. “Our results should lead to revision of clinical guidelines in favor of thymectomy and could potentially reverse downward trends in the use of thymectomy in overall management of myasthenia gravis.”
The main 3-year results of the Thymectomy Trial in Nonthymomatous Myasthenia Gravis Patients Receiving Prednisone (MGTX) were reported in 2016; the international trial found that thymectomy plus prednisone was superior to prednisone alone at 3 years (N Engl J Med. 2016 Aug 11;375[6]:511-22). The extension study aimed to assess the durability of the treatment response.
MGTX enrolled patients aged 18-65 years who had generalized nonthymomatous myasthenia gravis of less than 5 years’ duration and Myasthenia Gravis Foundation of America Clinical Classification Class II-IV disease. Of 111 patients who completed MGTX, 68 entered the extension study, and 50 completed the 60-month assessment (24 patients in the prednisone alone group and 26 patients in the prednisone plus thymectomy group).
At 5 years, patients in the thymectomy plus prednisone group had significantly lower time-weighted average Quantitative Myasthenia Gravis (QMG) scores (5.47 vs. 9.34) and mean alternate-day prednisone doses (24 mg vs. 48 mg), compared with patients who received prednisone alone. Twelve of 35 patients in the thymectomy group and 14 of 33 patients in the prednisone group had at least one adverse event by month 60. No treatment-related deaths occurred in the extension phase.
At 5 years, significantly more patients who underwent thymectomy had minimal manifestation status (i.e., no functional limitations from the disease other than some muscle weakness) – 88% versus 58%. The corresponding figures at 3 years were 67% and 47%.
In addition, 3-year and 5-year data indicate that the need for hospitalization is reduced after surgery, compared with medical therapy alone, Dr. Wolfe said.
Two patients in each treatment arm had an increase of 2 points or more in the QMG score, indicating clinical worsening.
“Our current findings reinforce the benefit of thymectomy seen in [MGTX], dispelling doubts about the procedure’s benefits and how long those benefits last,” said Dr. Wolfe. “We do hope that the new findings help reverse the apparent reluctance to do thymectomy and that the proportion of patients with myasthenia gravis who undergo thymectomy will increase.”
The authors noted that the small sample size of the extension study may limit its generalizability.
The study received funding from the National Institutes of Health. Dr. Wolfe reported grants from the NIH, the Muscular Dystrophy Association, the Myasthenia Gravis Foundation of America, CSL-Behring, and ArgenX, as well as personal fees from Grifols, Shire, and Alexion Pharmaceuticals. Coauthors reported working with and receiving funds from agencies, foundations, and pharmaceutical companies.
SOURCE: Wolfe GI et al. Lancet Neurol. 2019 Jan 25. doi: 10.1016/S1474-4422(18)30392-2.
Thymectomy may continue to benefit patients with myasthenia gravis 5 years after the procedure, according to an extension study published in Lancet Neurology.
The study evaluated the clinical status, medication requirements, and adverse events of patients with myasthenia gravis who completed a randomized controlled trial of thymectomy plus prednisone versus prednisone alone and agreed to participate in a rater-blinded 2-year extension.
“Thymectomy within the first few years of the disease course in addition to prednisone therapy confers benefits that persist for 5 years ... in patients with generalized nonthymomatous myasthenia gravis,” said lead study author Gil I. Wolfe, MD, chair of the department of neurology at the University at Buffalo in New York, and his research colleagues. “Results from the extension study provide further support for the use of thymectomy in management of myasthenia gravis and should encourage serious consideration of this treatment option in discussions between clinicians and their patients,” they wrote. “Our results should lead to revision of clinical guidelines in favor of thymectomy and could potentially reverse downward trends in the use of thymectomy in overall management of myasthenia gravis.”
The main 3-year results of the Thymectomy Trial in Nonthymomatous Myasthenia Gravis Patients Receiving Prednisone (MGTX) were reported in 2016; the international trial found that thymectomy plus prednisone was superior to prednisone alone at 3 years (N Engl J Med. 2016 Aug 11;375[6]:511-22). The extension study aimed to assess the durability of the treatment response.
MGTX enrolled patients aged 18-65 years who had generalized nonthymomatous myasthenia gravis of less than 5 years’ duration and Myasthenia Gravis Foundation of America Clinical Classification Class II-IV disease. Of 111 patients who completed MGTX, 68 entered the extension study, and 50 completed the 60-month assessment (24 patients in the prednisone alone group and 26 patients in the prednisone plus thymectomy group).
At 5 years, patients in the thymectomy plus prednisone group had significantly lower time-weighted average Quantitative Myasthenia Gravis (QMG) scores (5.47 vs. 9.34) and mean alternate-day prednisone doses (24 mg vs. 48 mg), compared with patients who received prednisone alone. Twelve of 35 patients in the thymectomy group and 14 of 33 patients in the prednisone group had at least one adverse event by month 60. No treatment-related deaths occurred in the extension phase.
At 5 years, significantly more patients who underwent thymectomy had minimal manifestation status (i.e., no functional limitations from the disease other than some muscle weakness) – 88% versus 58%. The corresponding figures at 3 years were 67% and 47%.
In addition, 3-year and 5-year data indicate that the need for hospitalization is reduced after surgery, compared with medical therapy alone, Dr. Wolfe said.
Two patients in each treatment arm had an increase of 2 points or more in the QMG score, indicating clinical worsening.
“Our current findings reinforce the benefit of thymectomy seen in [MGTX], dispelling doubts about the procedure’s benefits and how long those benefits last,” said Dr. Wolfe. “We do hope that the new findings help reverse the apparent reluctance to do thymectomy and that the proportion of patients with myasthenia gravis who undergo thymectomy will increase.”
The authors noted that the small sample size of the extension study may limit its generalizability.
The study received funding from the National Institutes of Health. Dr. Wolfe reported grants from the NIH, the Muscular Dystrophy Association, the Myasthenia Gravis Foundation of America, CSL-Behring, and ArgenX, as well as personal fees from Grifols, Shire, and Alexion Pharmaceuticals. Coauthors reported working with and receiving funds from agencies, foundations, and pharmaceutical companies.
SOURCE: Wolfe GI et al. Lancet Neurol. 2019 Jan 25. doi: 10.1016/S1474-4422(18)30392-2.
Thymectomy may continue to benefit patients with myasthenia gravis 5 years after the procedure, according to an extension study published in Lancet Neurology.
The study evaluated the clinical status, medication requirements, and adverse events of patients with myasthenia gravis who completed a randomized controlled trial of thymectomy plus prednisone versus prednisone alone and agreed to participate in a rater-blinded 2-year extension.
“Thymectomy within the first few years of the disease course in addition to prednisone therapy confers benefits that persist for 5 years ... in patients with generalized nonthymomatous myasthenia gravis,” said lead study author Gil I. Wolfe, MD, chair of the department of neurology at the University at Buffalo in New York, and his research colleagues. “Results from the extension study provide further support for the use of thymectomy in management of myasthenia gravis and should encourage serious consideration of this treatment option in discussions between clinicians and their patients,” they wrote. “Our results should lead to revision of clinical guidelines in favor of thymectomy and could potentially reverse downward trends in the use of thymectomy in overall management of myasthenia gravis.”
The main 3-year results of the Thymectomy Trial in Nonthymomatous Myasthenia Gravis Patients Receiving Prednisone (MGTX) were reported in 2016; the international trial found that thymectomy plus prednisone was superior to prednisone alone at 3 years (N Engl J Med. 2016 Aug 11;375[6]:511-22). The extension study aimed to assess the durability of the treatment response.
MGTX enrolled patients aged 18-65 years who had generalized nonthymomatous myasthenia gravis of less than 5 years’ duration and Myasthenia Gravis Foundation of America Clinical Classification Class II-IV disease. Of 111 patients who completed MGTX, 68 entered the extension study, and 50 completed the 60-month assessment (24 patients in the prednisone alone group and 26 patients in the prednisone plus thymectomy group).
At 5 years, patients in the thymectomy plus prednisone group had significantly lower time-weighted average Quantitative Myasthenia Gravis (QMG) scores (5.47 vs. 9.34) and mean alternate-day prednisone doses (24 mg vs. 48 mg), compared with patients who received prednisone alone. Twelve of 35 patients in the thymectomy group and 14 of 33 patients in the prednisone group had at least one adverse event by month 60. No treatment-related deaths occurred in the extension phase.
At 5 years, significantly more patients who underwent thymectomy had minimal manifestation status (i.e., no functional limitations from the disease other than some muscle weakness) – 88% versus 58%. The corresponding figures at 3 years were 67% and 47%.
In addition, 3-year and 5-year data indicate that the need for hospitalization is reduced after surgery, compared with medical therapy alone, Dr. Wolfe said.
Two patients in each treatment arm had an increase of 2 points or more in the QMG score, indicating clinical worsening.
“Our current findings reinforce the benefit of thymectomy seen in [MGTX], dispelling doubts about the procedure’s benefits and how long those benefits last,” said Dr. Wolfe. “We do hope that the new findings help reverse the apparent reluctance to do thymectomy and that the proportion of patients with myasthenia gravis who undergo thymectomy will increase.”
The authors noted that the small sample size of the extension study may limit its generalizability.
The study received funding from the National Institutes of Health. Dr. Wolfe reported grants from the NIH, the Muscular Dystrophy Association, the Myasthenia Gravis Foundation of America, CSL-Behring, and ArgenX, as well as personal fees from Grifols, Shire, and Alexion Pharmaceuticals. Coauthors reported working with and receiving funds from agencies, foundations, and pharmaceutical companies.
SOURCE: Wolfe GI et al. Lancet Neurol. 2019 Jan 25. doi: 10.1016/S1474-4422(18)30392-2.
FROM LANCET NEUROLOGY
Key clinical point: The benefits of thymectomy for myasthenia gravis persist 5 years after the procedure.
Major finding: Patients who undergo thymectomy and receive prednisone have lower time-weighted average Quantitative Myasthenia Gravis scores (5.47 vs. 9.34) and mean alternate-day prednisone doses (24 mg vs. 48 mg), compared with patients who receive prednisone alone.
Study details: A rater-blinded 2-year extension study that enrolled 68 patients who had completed a 3-year randomized controlled trial.
Disclosures: The study received funding from the National Institutes of Health. Dr. Wolfe reported grants from the NIH, the Muscular Dystrophy Association, the Myasthenia Gravis Foundation of America, CSL-Behring, and ArgenX, as well as personal fees from Grifols, Shire, and Alexion Pharmaceuticals. Other authors reported working with and receiving funds from various agencies, foundations, and pharmaceutical companies.
Source: Wolfe GI et al. Lancet Neurol. 2019 Jan 25. doi: 10.1016/S1474-4422(18)30392-2.
Phase 3 studies of antiamyloid Alzheimer’s drug crenezumab stopped
After a disappointing interim analysis, Roche and its collaborator AC Immune are halting two phase 3 trials of the antiamyloid antibody crenezumab.
CREAD 1 and CREAD 2 enrolled patients with prodromal-to-mild sporadic Alzheimer’s disease. The preplanned interim safety and efficacy analysis determined that neither study was likely to meet the primary endpoint of change from baseline on the Clinical Dementia Rating-sum of boxes score.
There were no unexpected safety signals associated with the drug, despite a quadrupling of the phase 3 dose from that used in phase 2. The company in its press release said that it will continue to conduct the Autosomal Dominant Alzheimer’s Disease (ADAD) trial as part of the Alzheimer’s Prevention Initiative (API). ADAD is a large South American trial of crenezumab in Colombian families with familial Alzheimer’s caused by mutations in the presenilin-1 gene (PSEN1).
Roche did not release any data but said the trial results will be discussed at an upcoming scientific meeting.
“While the results with crenezumab are disappointing, they meaningfully contribute to our understanding of Alzheimer’s disease,” Sandra Horning, MD, Roche’s chief medical officer and executive vice president for global development, said in an interview. “We gratefully acknowledge the participants in the CREAD trials and the efforts of everyone involved in this important program.”
The decision was not a surprise to researchers who have followed the antibody’s development. It advanced into phase 3 with lackluster phase 2 cognitive, imaging, and biomarker data. Its selection as the therapeutic agent for the ADAD trial was a key driver in its continued development, securing Roche $100 million in federal funds to help launch ADAD, the first-ever Alzheimer’s primary prevention study.
Despite its failure in sporadic Alzheimer’s, there is still some hope that crenezumab might benefit people with the PSEN1 mutation, said Richard Caselli, MD, professor of neurology at the Mayo Clinic Arizona in Scottsdale and associate director and clinical core director of the Arizona Alzheimer’s Disease Center.
“The Colombian trial is aimed at dominantly-inherited AD due to a PSEN1 mutation, so it is different enough to imagine it still might make a difference in patients in whom amyloid metabolism is actually defective due to functionally altered amyloid precursor protein or gamma secretase,” he said in an interview. “Possibly some might argue that many of the patients in the crenezumab trial likely had additional pathologies so that even if the AD component responded, the overall clinical picture might not reflect it due to the other components. That would be interesting if proven and could even argue against equating young-onset with late-onset AD, at least for clinical purposes, as is currently envisioned.”
Michael Wolfe, PhD, had a different take on the matter.
“Although amyloid-beta [Abeta] production is not necessarily altered in sporadic AD, there is essentially the same pathology, presentation, and progression with familial and sporadic AD, suggesting a common molecular mechanism,” said Dr. Wolfe, who is the Mathias P. Mertes Professor of Medicinal Chemistry at the University of Kansas, Lawrence. “It’s hard to say Abeta is the pathogenic species in familial but not sporadic AD.
“To me, the failures of the antiamyloid approaches are because the drugs are given too late, are targeting the wrong form of Abeta, or are targeting an enzyme [for example, beta secretase1] that has other important functions. Most likely it’s a combination of these reasons. One could argue that even if some form of Abeta is the pathogenic entity, it is not a practical target because intervention may need to be initiated many years before the onset of symptoms.”
Despite the long string of failed antiamyloid antibodies, it’s not yet time to give up on the approach, said James Kupiec, MD, chief medical officer at ProMIS Neurosciences of Toronto.
“I understand where the pessimism [around antiamyloid antibodies] is coming from, and I also understand the enthusiasm from these companies to pursue them,” said Dr. Kupiec, who formerly headed Pfizer’s neuroscience research unit. “Targeting plaque is clearly not going to do the job. But in my opinion, the deeper pathophysiologic questions have not been adequately addressed. I’m not willing to throw in the towel. The correct molecular species [of amyloid] has not been appropriately or adequately tested in studies with monoclonal antibodies.”
The antibodies that have been failing for 5 years now were designed in the early 2000s, Dr. Kupiec pointed out, when knowledge of the various amyloid species was still immature. Newer candidates can target specific conformations of the protein – monomers and oligomers – before they aggregate into insoluble sheets. “Solanezumab was the first of these, paving the way for this new generation of antibodies,” Dr. Kupiec said.
Because they target soluble Abeta, not amyloid plaques, these domain-specific antibodies are less likely to elicit ARIA (amyloid-related imaging abnormalities), the inflammatory reaction that’s been associated with plaque dissolution in other antibody trials. ARIA has been a dose-limiting step for antiamyloid antibodies – one that conformationally targeted antibodies could avoid, Dr. Kupiec said.
“There may be some limited success with the these, and there may be enough of a treatment effect to secure approval,” he said. “The question is: Can we generate a higher effect size with an antibody that is more selective to the toxic forms of Abeta?”
PMN310 is ProMIS’ attempt to thread this needle. In preclinical studies, the antibody did not bind to amyloid monomers, plaques, or vascular Abeta aggregates. The company expects to take this antibody into phase 1 trials later this year.
“If we have a molecule that doesn’t bind to monomers or to plaques, but only to the toxic oligomer, then that is an something well worth testing in the clinic,” he said.
Dr. Caselli and Dr. Wolfe have no financial disclosures.
On behalf of the millions of people living with Alzheimer’s disease and their families that we serve and represent, the Alzheimer’s Association is disappointed to learn that these trials have been stopped.
We learn something from every Alzheimer’s clinical trial. The Alzheimer’s Association looks forward to hearing details of these studies at an upcoming scientific meeting.
More important, we must redouble our efforts to better understand the causes of the disease, and to discover additional therapeutic targets. No stone can be left unturned in the pursuit of better treatments and effective preventions.
The Alzheimer’s Association is investing in research looking at a variety of novel targets for treatment and prevention, including brain inflammation, the life and death cycle of brain cells, how brain cells use different energy sources, and the impact of lifestyle.
• Lifestyle interventions include leading the U.S. POINTER Study.
• To further the study of blood pressure control on reducing risk of mild cognitive impairment and dementia, the Alzheimer’s Association recently announced seed funding of SPRINT MIND 2.0.
• Part The Cloud Translational Research program fills a gap in Alzheimer’s drug development by supporting more than 30 early phase clinical studies.
• The Association is also funding research into the causes of the disease.
The emotional and financial cost of Alzheimer’s is enormous. At the Alzheimer’s Association, we will not stop. We will not slow down in our fight against this terrible disease.
Maria Carrillo, PhD , is the Alzheimer’s Association’s chief science officer.
On behalf of the millions of people living with Alzheimer’s disease and their families that we serve and represent, the Alzheimer’s Association is disappointed to learn that these trials have been stopped.
We learn something from every Alzheimer’s clinical trial. The Alzheimer’s Association looks forward to hearing details of these studies at an upcoming scientific meeting.
More important, we must redouble our efforts to better understand the causes of the disease, and to discover additional therapeutic targets. No stone can be left unturned in the pursuit of better treatments and effective preventions.
The Alzheimer’s Association is investing in research looking at a variety of novel targets for treatment and prevention, including brain inflammation, the life and death cycle of brain cells, how brain cells use different energy sources, and the impact of lifestyle.
• Lifestyle interventions include leading the U.S. POINTER Study.
• To further the study of blood pressure control on reducing risk of mild cognitive impairment and dementia, the Alzheimer’s Association recently announced seed funding of SPRINT MIND 2.0.
• Part The Cloud Translational Research program fills a gap in Alzheimer’s drug development by supporting more than 30 early phase clinical studies.
• The Association is also funding research into the causes of the disease.
The emotional and financial cost of Alzheimer’s is enormous. At the Alzheimer’s Association, we will not stop. We will not slow down in our fight against this terrible disease.
Maria Carrillo, PhD , is the Alzheimer’s Association’s chief science officer.
On behalf of the millions of people living with Alzheimer’s disease and their families that we serve and represent, the Alzheimer’s Association is disappointed to learn that these trials have been stopped.
We learn something from every Alzheimer’s clinical trial. The Alzheimer’s Association looks forward to hearing details of these studies at an upcoming scientific meeting.
More important, we must redouble our efforts to better understand the causes of the disease, and to discover additional therapeutic targets. No stone can be left unturned in the pursuit of better treatments and effective preventions.
The Alzheimer’s Association is investing in research looking at a variety of novel targets for treatment and prevention, including brain inflammation, the life and death cycle of brain cells, how brain cells use different energy sources, and the impact of lifestyle.
• Lifestyle interventions include leading the U.S. POINTER Study.
• To further the study of blood pressure control on reducing risk of mild cognitive impairment and dementia, the Alzheimer’s Association recently announced seed funding of SPRINT MIND 2.0.
• Part The Cloud Translational Research program fills a gap in Alzheimer’s drug development by supporting more than 30 early phase clinical studies.
• The Association is also funding research into the causes of the disease.
The emotional and financial cost of Alzheimer’s is enormous. At the Alzheimer’s Association, we will not stop. We will not slow down in our fight against this terrible disease.
Maria Carrillo, PhD , is the Alzheimer’s Association’s chief science officer.
After a disappointing interim analysis, Roche and its collaborator AC Immune are halting two phase 3 trials of the antiamyloid antibody crenezumab.
CREAD 1 and CREAD 2 enrolled patients with prodromal-to-mild sporadic Alzheimer’s disease. The preplanned interim safety and efficacy analysis determined that neither study was likely to meet the primary endpoint of change from baseline on the Clinical Dementia Rating-sum of boxes score.
There were no unexpected safety signals associated with the drug, despite a quadrupling of the phase 3 dose from that used in phase 2. The company in its press release said that it will continue to conduct the Autosomal Dominant Alzheimer’s Disease (ADAD) trial as part of the Alzheimer’s Prevention Initiative (API). ADAD is a large South American trial of crenezumab in Colombian families with familial Alzheimer’s caused by mutations in the presenilin-1 gene (PSEN1).
Roche did not release any data but said the trial results will be discussed at an upcoming scientific meeting.
“While the results with crenezumab are disappointing, they meaningfully contribute to our understanding of Alzheimer’s disease,” Sandra Horning, MD, Roche’s chief medical officer and executive vice president for global development, said in an interview. “We gratefully acknowledge the participants in the CREAD trials and the efforts of everyone involved in this important program.”
The decision was not a surprise to researchers who have followed the antibody’s development. It advanced into phase 3 with lackluster phase 2 cognitive, imaging, and biomarker data. Its selection as the therapeutic agent for the ADAD trial was a key driver in its continued development, securing Roche $100 million in federal funds to help launch ADAD, the first-ever Alzheimer’s primary prevention study.
Despite its failure in sporadic Alzheimer’s, there is still some hope that crenezumab might benefit people with the PSEN1 mutation, said Richard Caselli, MD, professor of neurology at the Mayo Clinic Arizona in Scottsdale and associate director and clinical core director of the Arizona Alzheimer’s Disease Center.
“The Colombian trial is aimed at dominantly-inherited AD due to a PSEN1 mutation, so it is different enough to imagine it still might make a difference in patients in whom amyloid metabolism is actually defective due to functionally altered amyloid precursor protein or gamma secretase,” he said in an interview. “Possibly some might argue that many of the patients in the crenezumab trial likely had additional pathologies so that even if the AD component responded, the overall clinical picture might not reflect it due to the other components. That would be interesting if proven and could even argue against equating young-onset with late-onset AD, at least for clinical purposes, as is currently envisioned.”
Michael Wolfe, PhD, had a different take on the matter.
“Although amyloid-beta [Abeta] production is not necessarily altered in sporadic AD, there is essentially the same pathology, presentation, and progression with familial and sporadic AD, suggesting a common molecular mechanism,” said Dr. Wolfe, who is the Mathias P. Mertes Professor of Medicinal Chemistry at the University of Kansas, Lawrence. “It’s hard to say Abeta is the pathogenic species in familial but not sporadic AD.
“To me, the failures of the antiamyloid approaches are because the drugs are given too late, are targeting the wrong form of Abeta, or are targeting an enzyme [for example, beta secretase1] that has other important functions. Most likely it’s a combination of these reasons. One could argue that even if some form of Abeta is the pathogenic entity, it is not a practical target because intervention may need to be initiated many years before the onset of symptoms.”
Despite the long string of failed antiamyloid antibodies, it’s not yet time to give up on the approach, said James Kupiec, MD, chief medical officer at ProMIS Neurosciences of Toronto.
“I understand where the pessimism [around antiamyloid antibodies] is coming from, and I also understand the enthusiasm from these companies to pursue them,” said Dr. Kupiec, who formerly headed Pfizer’s neuroscience research unit. “Targeting plaque is clearly not going to do the job. But in my opinion, the deeper pathophysiologic questions have not been adequately addressed. I’m not willing to throw in the towel. The correct molecular species [of amyloid] has not been appropriately or adequately tested in studies with monoclonal antibodies.”
The antibodies that have been failing for 5 years now were designed in the early 2000s, Dr. Kupiec pointed out, when knowledge of the various amyloid species was still immature. Newer candidates can target specific conformations of the protein – monomers and oligomers – before they aggregate into insoluble sheets. “Solanezumab was the first of these, paving the way for this new generation of antibodies,” Dr. Kupiec said.
Because they target soluble Abeta, not amyloid plaques, these domain-specific antibodies are less likely to elicit ARIA (amyloid-related imaging abnormalities), the inflammatory reaction that’s been associated with plaque dissolution in other antibody trials. ARIA has been a dose-limiting step for antiamyloid antibodies – one that conformationally targeted antibodies could avoid, Dr. Kupiec said.
“There may be some limited success with the these, and there may be enough of a treatment effect to secure approval,” he said. “The question is: Can we generate a higher effect size with an antibody that is more selective to the toxic forms of Abeta?”
PMN310 is ProMIS’ attempt to thread this needle. In preclinical studies, the antibody did not bind to amyloid monomers, plaques, or vascular Abeta aggregates. The company expects to take this antibody into phase 1 trials later this year.
“If we have a molecule that doesn’t bind to monomers or to plaques, but only to the toxic oligomer, then that is an something well worth testing in the clinic,” he said.
Dr. Caselli and Dr. Wolfe have no financial disclosures.
After a disappointing interim analysis, Roche and its collaborator AC Immune are halting two phase 3 trials of the antiamyloid antibody crenezumab.
CREAD 1 and CREAD 2 enrolled patients with prodromal-to-mild sporadic Alzheimer’s disease. The preplanned interim safety and efficacy analysis determined that neither study was likely to meet the primary endpoint of change from baseline on the Clinical Dementia Rating-sum of boxes score.
There were no unexpected safety signals associated with the drug, despite a quadrupling of the phase 3 dose from that used in phase 2. The company in its press release said that it will continue to conduct the Autosomal Dominant Alzheimer’s Disease (ADAD) trial as part of the Alzheimer’s Prevention Initiative (API). ADAD is a large South American trial of crenezumab in Colombian families with familial Alzheimer’s caused by mutations in the presenilin-1 gene (PSEN1).
Roche did not release any data but said the trial results will be discussed at an upcoming scientific meeting.
“While the results with crenezumab are disappointing, they meaningfully contribute to our understanding of Alzheimer’s disease,” Sandra Horning, MD, Roche’s chief medical officer and executive vice president for global development, said in an interview. “We gratefully acknowledge the participants in the CREAD trials and the efforts of everyone involved in this important program.”
The decision was not a surprise to researchers who have followed the antibody’s development. It advanced into phase 3 with lackluster phase 2 cognitive, imaging, and biomarker data. Its selection as the therapeutic agent for the ADAD trial was a key driver in its continued development, securing Roche $100 million in federal funds to help launch ADAD, the first-ever Alzheimer’s primary prevention study.
Despite its failure in sporadic Alzheimer’s, there is still some hope that crenezumab might benefit people with the PSEN1 mutation, said Richard Caselli, MD, professor of neurology at the Mayo Clinic Arizona in Scottsdale and associate director and clinical core director of the Arizona Alzheimer’s Disease Center.
“The Colombian trial is aimed at dominantly-inherited AD due to a PSEN1 mutation, so it is different enough to imagine it still might make a difference in patients in whom amyloid metabolism is actually defective due to functionally altered amyloid precursor protein or gamma secretase,” he said in an interview. “Possibly some might argue that many of the patients in the crenezumab trial likely had additional pathologies so that even if the AD component responded, the overall clinical picture might not reflect it due to the other components. That would be interesting if proven and could even argue against equating young-onset with late-onset AD, at least for clinical purposes, as is currently envisioned.”
Michael Wolfe, PhD, had a different take on the matter.
“Although amyloid-beta [Abeta] production is not necessarily altered in sporadic AD, there is essentially the same pathology, presentation, and progression with familial and sporadic AD, suggesting a common molecular mechanism,” said Dr. Wolfe, who is the Mathias P. Mertes Professor of Medicinal Chemistry at the University of Kansas, Lawrence. “It’s hard to say Abeta is the pathogenic species in familial but not sporadic AD.
“To me, the failures of the antiamyloid approaches are because the drugs are given too late, are targeting the wrong form of Abeta, or are targeting an enzyme [for example, beta secretase1] that has other important functions. Most likely it’s a combination of these reasons. One could argue that even if some form of Abeta is the pathogenic entity, it is not a practical target because intervention may need to be initiated many years before the onset of symptoms.”
Despite the long string of failed antiamyloid antibodies, it’s not yet time to give up on the approach, said James Kupiec, MD, chief medical officer at ProMIS Neurosciences of Toronto.
“I understand where the pessimism [around antiamyloid antibodies] is coming from, and I also understand the enthusiasm from these companies to pursue them,” said Dr. Kupiec, who formerly headed Pfizer’s neuroscience research unit. “Targeting plaque is clearly not going to do the job. But in my opinion, the deeper pathophysiologic questions have not been adequately addressed. I’m not willing to throw in the towel. The correct molecular species [of amyloid] has not been appropriately or adequately tested in studies with monoclonal antibodies.”
The antibodies that have been failing for 5 years now were designed in the early 2000s, Dr. Kupiec pointed out, when knowledge of the various amyloid species was still immature. Newer candidates can target specific conformations of the protein – monomers and oligomers – before they aggregate into insoluble sheets. “Solanezumab was the first of these, paving the way for this new generation of antibodies,” Dr. Kupiec said.
Because they target soluble Abeta, not amyloid plaques, these domain-specific antibodies are less likely to elicit ARIA (amyloid-related imaging abnormalities), the inflammatory reaction that’s been associated with plaque dissolution in other antibody trials. ARIA has been a dose-limiting step for antiamyloid antibodies – one that conformationally targeted antibodies could avoid, Dr. Kupiec said.
“There may be some limited success with the these, and there may be enough of a treatment effect to secure approval,” he said. “The question is: Can we generate a higher effect size with an antibody that is more selective to the toxic forms of Abeta?”
PMN310 is ProMIS’ attempt to thread this needle. In preclinical studies, the antibody did not bind to amyloid monomers, plaques, or vascular Abeta aggregates. The company expects to take this antibody into phase 1 trials later this year.
“If we have a molecule that doesn’t bind to monomers or to plaques, but only to the toxic oligomer, then that is an something well worth testing in the clinic,” he said.
Dr. Caselli and Dr. Wolfe have no financial disclosures.
Keeping Your Brain in Shape
Every year, thousands of us vow to “get in shape” by eating right and exercising. (Whether we keep that resolution is another story.) But while we view physical exercise as a way to lose or maintain weight, reduce stress, or even hone athletic skills, we seldom think about exercising one of the most important muscles in our body: the brain.
“What?” you say. “The brain is not like other muscles.” No, it’s not … and yet, it isn’t as different as we used to think. Historically (maybe histologically?), it was believed that if nerve cells in the adult brain were damaged or had died, they, unlike other cells in t
But since the late 1990s, scientists have been debunking the negative myths about our brains as we age. They are not as static and unable to change as we have been led to fear! In fact, in 1998, American and Swedish scientists demonstrated that adult humans can generate new brain cells.1,2 Moreover, the brain does replicate neurons in the hippocampus, the area in our brains that is central to learning and memory. Neurons continue to grow and change beyond the first years of development and well into adulthood.
So learning (and teaching) movements to encourage the rebuilding of our neurons is key to keeping our minds sharp. In his work, Ratey found that “our physical movements can directly influence our ability to learn, think, and remember.”3 He also tells us that exercise enhances circulation to the brain, “priming it for improved function, including mental health as well as cognitive ability.”4
No, you can’t put your brain on a treadmill to get, and help keep, it “in shape.” But you can do something to maintain mental sharpness and delay decline in mental agility. And these exercises don’t require a health club membership or special equipment. They can be done anytime, anywhere … and no one knows you are doing them!
I’m talking about neurobics, a term coined to describe exercises that keep us mentally fit.5 The purpose of these activities is to work our brains in nonroutine or unexpected ways, using all of our senses to experience, or re-experience, a common activity.
Not sure what that means? Here are some examples:
Spend time in a new environment. Go to a different park or a new store. Travel, by the way, seems to slow age-related mental decline.
Continue to: Smell new odors in the morning
Smell new odors in the morning. Have new scents, like a bottle of mint extract, ready to smell first thing in the morning, to “wake up” your brain.
Take a shower with your eyes closed. Other senses become more active when you cannot see, and a shower engages several.
Try brushing your teeth with your nondominant hand. This may be difficult for some of us—and it definitely requires full attention the first time you try it!
Learn to read braille. This is a tough one, but learning to read with your fingers definitely involves one of your senses in a new context. Or, you could try learning American Sign Language, which also uses your fingers to communicate.
Respond to a situation differently. Catch yourself in a normal, unconscious response to a situation, and choose to respond in an alternate (and preferably better) way.
Continue to: Find a new route to work
Find a new route to work. It doesn’t have to be longer, just different. You may even find a faster way to work once you break your routine.
Act confidently. In a situation you are unsure about, choose to act confidently. You’ll notice that your mind gets very active once you adopt the assumption that you will know what to do.
Distinguish coins using only your sense of touch. This brain exercise can be used to kill time while waiting for an appointment. If you really want a challenge, see if you can distinguish paper currency denominations by touch.
Leave the lights off in the house. Get around your home by memory and feel. This certainly fully engages your attention—but be careful, of course!
If you give neurobics a try, let me know what you think! Or if you have other tips for staying mentally “fit,” please share them. I can be reached at [email protected]. And thank you to my friend Gail, who suggested this topic to me!
1. Kempermann G, Gage FH. New nerve cells for the adult brain. Scientific American. 1999;280(5):38-44.
2. Eriksson PS, Perfilieva E, Björk-Eriksson T, et al. Neurogenesis in the adult human hippocampus. Nature Medicine. 1998;4(11):1313-1317.
3. Ratey J. A User’s Guide to the Brain: Perception, Attention, and the Four Theaters of the Brain. New York, NY: Vintage Books; 2002.
4. Ratey J. SPARK: The Revolutionary New Science of Exercise and the Brain. New York, NY: Little, Brown and Company; 2008.
5. Katz LC, Rubin M. Keep Your Brain Alive: 83 Neurobic Exercises. New York, NY: Workman Publishing Company; 1999.
Every year, thousands of us vow to “get in shape” by eating right and exercising. (Whether we keep that resolution is another story.) But while we view physical exercise as a way to lose or maintain weight, reduce stress, or even hone athletic skills, we seldom think about exercising one of the most important muscles in our body: the brain.
“What?” you say. “The brain is not like other muscles.” No, it’s not … and yet, it isn’t as different as we used to think. Historically (maybe histologically?), it was believed that if nerve cells in the adult brain were damaged or had died, they, unlike other cells in t
But since the late 1990s, scientists have been debunking the negative myths about our brains as we age. They are not as static and unable to change as we have been led to fear! In fact, in 1998, American and Swedish scientists demonstrated that adult humans can generate new brain cells.1,2 Moreover, the brain does replicate neurons in the hippocampus, the area in our brains that is central to learning and memory. Neurons continue to grow and change beyond the first years of development and well into adulthood.
So learning (and teaching) movements to encourage the rebuilding of our neurons is key to keeping our minds sharp. In his work, Ratey found that “our physical movements can directly influence our ability to learn, think, and remember.”3 He also tells us that exercise enhances circulation to the brain, “priming it for improved function, including mental health as well as cognitive ability.”4
No, you can’t put your brain on a treadmill to get, and help keep, it “in shape.” But you can do something to maintain mental sharpness and delay decline in mental agility. And these exercises don’t require a health club membership or special equipment. They can be done anytime, anywhere … and no one knows you are doing them!
I’m talking about neurobics, a term coined to describe exercises that keep us mentally fit.5 The purpose of these activities is to work our brains in nonroutine or unexpected ways, using all of our senses to experience, or re-experience, a common activity.
Not sure what that means? Here are some examples:
Spend time in a new environment. Go to a different park or a new store. Travel, by the way, seems to slow age-related mental decline.
Continue to: Smell new odors in the morning
Smell new odors in the morning. Have new scents, like a bottle of mint extract, ready to smell first thing in the morning, to “wake up” your brain.
Take a shower with your eyes closed. Other senses become more active when you cannot see, and a shower engages several.
Try brushing your teeth with your nondominant hand. This may be difficult for some of us—and it definitely requires full attention the first time you try it!
Learn to read braille. This is a tough one, but learning to read with your fingers definitely involves one of your senses in a new context. Or, you could try learning American Sign Language, which also uses your fingers to communicate.
Respond to a situation differently. Catch yourself in a normal, unconscious response to a situation, and choose to respond in an alternate (and preferably better) way.
Continue to: Find a new route to work
Find a new route to work. It doesn’t have to be longer, just different. You may even find a faster way to work once you break your routine.
Act confidently. In a situation you are unsure about, choose to act confidently. You’ll notice that your mind gets very active once you adopt the assumption that you will know what to do.
Distinguish coins using only your sense of touch. This brain exercise can be used to kill time while waiting for an appointment. If you really want a challenge, see if you can distinguish paper currency denominations by touch.
Leave the lights off in the house. Get around your home by memory and feel. This certainly fully engages your attention—but be careful, of course!
If you give neurobics a try, let me know what you think! Or if you have other tips for staying mentally “fit,” please share them. I can be reached at [email protected]. And thank you to my friend Gail, who suggested this topic to me!
Every year, thousands of us vow to “get in shape” by eating right and exercising. (Whether we keep that resolution is another story.) But while we view physical exercise as a way to lose or maintain weight, reduce stress, or even hone athletic skills, we seldom think about exercising one of the most important muscles in our body: the brain.
“What?” you say. “The brain is not like other muscles.” No, it’s not … and yet, it isn’t as different as we used to think. Historically (maybe histologically?), it was believed that if nerve cells in the adult brain were damaged or had died, they, unlike other cells in t
But since the late 1990s, scientists have been debunking the negative myths about our brains as we age. They are not as static and unable to change as we have been led to fear! In fact, in 1998, American and Swedish scientists demonstrated that adult humans can generate new brain cells.1,2 Moreover, the brain does replicate neurons in the hippocampus, the area in our brains that is central to learning and memory. Neurons continue to grow and change beyond the first years of development and well into adulthood.
So learning (and teaching) movements to encourage the rebuilding of our neurons is key to keeping our minds sharp. In his work, Ratey found that “our physical movements can directly influence our ability to learn, think, and remember.”3 He also tells us that exercise enhances circulation to the brain, “priming it for improved function, including mental health as well as cognitive ability.”4
No, you can’t put your brain on a treadmill to get, and help keep, it “in shape.” But you can do something to maintain mental sharpness and delay decline in mental agility. And these exercises don’t require a health club membership or special equipment. They can be done anytime, anywhere … and no one knows you are doing them!
I’m talking about neurobics, a term coined to describe exercises that keep us mentally fit.5 The purpose of these activities is to work our brains in nonroutine or unexpected ways, using all of our senses to experience, or re-experience, a common activity.
Not sure what that means? Here are some examples:
Spend time in a new environment. Go to a different park or a new store. Travel, by the way, seems to slow age-related mental decline.
Continue to: Smell new odors in the morning
Smell new odors in the morning. Have new scents, like a bottle of mint extract, ready to smell first thing in the morning, to “wake up” your brain.
Take a shower with your eyes closed. Other senses become more active when you cannot see, and a shower engages several.
Try brushing your teeth with your nondominant hand. This may be difficult for some of us—and it definitely requires full attention the first time you try it!
Learn to read braille. This is a tough one, but learning to read with your fingers definitely involves one of your senses in a new context. Or, you could try learning American Sign Language, which also uses your fingers to communicate.
Respond to a situation differently. Catch yourself in a normal, unconscious response to a situation, and choose to respond in an alternate (and preferably better) way.
Continue to: Find a new route to work
Find a new route to work. It doesn’t have to be longer, just different. You may even find a faster way to work once you break your routine.
Act confidently. In a situation you are unsure about, choose to act confidently. You’ll notice that your mind gets very active once you adopt the assumption that you will know what to do.
Distinguish coins using only your sense of touch. This brain exercise can be used to kill time while waiting for an appointment. If you really want a challenge, see if you can distinguish paper currency denominations by touch.
Leave the lights off in the house. Get around your home by memory and feel. This certainly fully engages your attention—but be careful, of course!
If you give neurobics a try, let me know what you think! Or if you have other tips for staying mentally “fit,” please share them. I can be reached at [email protected]. And thank you to my friend Gail, who suggested this topic to me!
1. Kempermann G, Gage FH. New nerve cells for the adult brain. Scientific American. 1999;280(5):38-44.
2. Eriksson PS, Perfilieva E, Björk-Eriksson T, et al. Neurogenesis in the adult human hippocampus. Nature Medicine. 1998;4(11):1313-1317.
3. Ratey J. A User’s Guide to the Brain: Perception, Attention, and the Four Theaters of the Brain. New York, NY: Vintage Books; 2002.
4. Ratey J. SPARK: The Revolutionary New Science of Exercise and the Brain. New York, NY: Little, Brown and Company; 2008.
5. Katz LC, Rubin M. Keep Your Brain Alive: 83 Neurobic Exercises. New York, NY: Workman Publishing Company; 1999.
1. Kempermann G, Gage FH. New nerve cells for the adult brain. Scientific American. 1999;280(5):38-44.
2. Eriksson PS, Perfilieva E, Björk-Eriksson T, et al. Neurogenesis in the adult human hippocampus. Nature Medicine. 1998;4(11):1313-1317.
3. Ratey J. A User’s Guide to the Brain: Perception, Attention, and the Four Theaters of the Brain. New York, NY: Vintage Books; 2002.
4. Ratey J. SPARK: The Revolutionary New Science of Exercise and the Brain. New York, NY: Little, Brown and Company; 2008.
5. Katz LC, Rubin M. Keep Your Brain Alive: 83 Neurobic Exercises. New York, NY: Workman Publishing Company; 1999.
Defeating the opioid epidemic
The U.S. Surgeon General weighs in.
Vice Admiral Jerome M. Adams, MD, MPH, is the 20th Surgeon General of the United States, a post created in 1871.
Dr. Adams holds degrees in biochemistry and psychology from the University of Maryland, Baltimore County; a master’s degree in public health from the University of California, Berkeley; and a medical degree from the Indiana University, Indianapolis. He is a board-certified anesthesiologist and associate clinical professor of anesthesia at Indiana University.
At the 2018 Executive Advisory Board meeting of the Doctors Company, Richard E. Anderson, MD, FACP, chairman and chief executive officer of the Doctors Company, spoke with Dr. Adams about the opioid epidemic’s enormous impact on communities and health services in the United States.
Dr. Anderson: Dr. Adams, you’ve been busy since taking over as Surgeon General of the United States. What are some of the key challenges that you’re facing in this office?
Dr. Adams: You know, there are many challenges facing our country, but it boils down to a lack of wellness. We know that only 10% of health is due to health care, 20% of health is genetics, and the rest is a combination of behavior and environment.
My motto is “better health through better partnerships,” because I firmly believe that if we break out of our silos and reach across the traditional barriers that have been put up by funding, by reimbursement, and by infrastructure, then we can ultimately achieve wellness in our communities.
You asked what I’ve been focused on as Surgeon General. Well, I’m focused on three main areas right now.
No. 1 is the opioid epidemic. It is a scourge across our country. A person dies every 12½ minutes from an opioid overdose and that’s far too many. Especially when we know that many of those deaths can be prevented.
Another area I’m focused on is demonstrating the link between community health and economic prosperity. We want folks to invest in health because we know that not only will it achieve better health for individuals and communities but it will create a more prosperous nation, also.
And finally, I’m raising awareness about the links between our nation’s health and our safety and security – particularly our national security. Unfortunately, 7 out of 10 young people between the ages of 18 and 24 years old in our country are ineligible for military service. That’s because they can’t pass the physical, they can’t meet the educational requirements, or they have a criminal record.
So, our nation’s poor health is not just a matter of diabetes or heart disease 20 or 30 years down the road. We are literally a less-safe country right now because we’re an unhealthy country.
Dr. Anderson: Regarding the opioid epidemic, what are some of the programs that are available today that you find effective? What would you like to see us do as a nation to respond to the epidemic?
Dr. Adams: Recently, I was at a hospital in Alaska where they have implemented a neonatal abstinence syndrome protocol and program that is being looked at around the country – and others are attempting to replicate it.
We know that if you keep mom and baby together, baby does better, mom does better, hospital stays are shorter, costs go down, and you’re keeping that family unit intact. This prevents future problems for both the baby and the mother. That’s just one small example.
I’m also very happy to see that the prescribing of opioids is going down 20%-25% across the country. And there are even larger decreases in the military and veteran communities. That’s really a testament to doctors and the medical profession finally waking up. And I say this as a physician myself, as an anesthesiologist, as someone who is involved in acute and chronic pain management.
Four out of five people with substance use disorder say they started with a prescription opioid. Many physicians will say, “Those aren’t my patients,” but unfortunately when we look at the PDMP [prescription drug monitoring program] data across the country we do a poor job of predicting who is and who isn’t going to divert. It may not be your patient, but it could be their son or the babysitter who is diverting those overprescribed opioids.
One thing that I really think we need to lean into as health care practitioners is providing medication-assisted treatment, or MAT. We know that the gold standard for treatment and recovery is medication-assisted treatment of some form. But we also know it’s not nearly available enough and that there are barriers on the federal and state levels.
We need you to continue to talk to your congressional representatives and let them know which barriers you perceive because the data waiver comes directly from Congress.
Still, any ER can prescribe up to 3 days of MAT to someone. I’d much rather have our ER doctors putting patients on MAT and then connecting them to treatment, than sending them back out into the arms of a drug dealer after they put them into acute withdrawal with naloxone.
We also have too many pregnant women who want help but can’t find any treatment because no one out there will take care of pregnant moms. We need folks to step up to the plate and get that data waiver in our ob/gyn and primary care sectors.
Ultimately, we need hospitals and health care leaders to create an environment that makes providers feel comfortable providing that service by giving them the training and the support to be able to do it.
We also need to make sure we’re co-prescribing naloxone for those who are at risk for opioid overdose.
Dr. Anderson: Just so we are clear, are you in favor of regular prescribing of naloxone, along with prescriptions for opioids? Is that correct?
Dr. Adams: I issued the first Surgeon General’s advisory from more than 10 years earlier this year to help folks understand that over half of our opioid overdoses occur in a home setting. We all know that an anoxic brain injury occurs in 4-5 minutes. We also know that most ambulances and first responders aren’t going to show up in 4-5 minutes.
If we want to make a dent in this overdose epidemic, we need everyone to consider themselves a first responder. We need to look at it the same as we look at CPR; we need everyone carrying naloxone. That was one of the big pushes from my Surgeon General’s advisory.
How can providers help? Well, they can coprescribe naloxone to folks on high morphine milligram equivalents (MME) who are at risk. If grandma has naloxone at home and her grandson overdoses in the garage, then at least it’s in the same house. Naloxone is not the treatment for the opioid epidemic. But we can’t get someone who is dead into treatment.
I have no illusions that simply making naloxone available is going to turn the tide, but it certainly is an important part of it.
Dr. Anderson: From your unique viewpoint, how much progress do you see in relation to the opioid epidemic? Do you think we’re approaching an inflection point, or do you think there’s a long way to go before this starts to turn around?
Dr. Adams: When I talk about the opioid epidemic, I have two angles. No.1, I want to raise awareness about the opioid epidemic – the severity of it, and how everyone can lean into it in their own way. Whether it’s community citizens, providers, law enforcement, the business community, whomever.
But in addition to raising awareness, I want to instill hope.
I was in Huntington, West Virginia, just a few weeks ago at the epicenter of the opioid epidemic. They’ve been able to turn their opioid overdose rates around by providing peer recovery coaches to individuals and making sure naloxone is available throughout the community. You save the life and then you connect them to care.
We know that the folks who are at highest risk for overdose deaths are the ones who just overdosed. They come out of the ER where we’ve watched them for a few hours and then we send them right back out into the arms of the drug dealer to do exactly what we know they will do medically because we’ve thrown them into withdrawal and they try to get their next fix.
If we can partner with law enforcement, then we can turn our opioid overdose rates around.
A story of recovery that I want to share with you is about a guy named Jonathan, who I met when I was in Rhode Island.
Jonathan overdosed, but his roommate had access to naloxone, which he administered. Jonathan was taken to the ER and then connected with a peer recovery coach. He is now in recovery and has actually become a peer recovery coach himself. Saving this one life will now enable us to save many more.
Yet we still prescribe more than 80% of the world’s opioids to less than 5% of the world’s population. So, we still have an overprescribing epidemic, but we’ve surpassed the inflection point there. Prescribing is coming down.
But another part of this epidemic was that we squeezed the balloon in one place and, as prescribing opioids went down, lots of people switched over to heroin. That’s when we really first started to see overdose rates go up.
Well, it’s important for folks to know that, through law enforcement, through partnerships with the public health community, through an increase in syringe service programs, and through other touch points, heroin use is now going down in most places.
Unfortunately, now we’re seeing the third wave of the epidemic, and that’s fentanyl and carfentanil.
The U.S. Surgeon General weighs in.
The U.S. Surgeon General weighs in.
Vice Admiral Jerome M. Adams, MD, MPH, is the 20th Surgeon General of the United States, a post created in 1871.
Dr. Adams holds degrees in biochemistry and psychology from the University of Maryland, Baltimore County; a master’s degree in public health from the University of California, Berkeley; and a medical degree from the Indiana University, Indianapolis. He is a board-certified anesthesiologist and associate clinical professor of anesthesia at Indiana University.
At the 2018 Executive Advisory Board meeting of the Doctors Company, Richard E. Anderson, MD, FACP, chairman and chief executive officer of the Doctors Company, spoke with Dr. Adams about the opioid epidemic’s enormous impact on communities and health services in the United States.
Dr. Anderson: Dr. Adams, you’ve been busy since taking over as Surgeon General of the United States. What are some of the key challenges that you’re facing in this office?
Dr. Adams: You know, there are many challenges facing our country, but it boils down to a lack of wellness. We know that only 10% of health is due to health care, 20% of health is genetics, and the rest is a combination of behavior and environment.
My motto is “better health through better partnerships,” because I firmly believe that if we break out of our silos and reach across the traditional barriers that have been put up by funding, by reimbursement, and by infrastructure, then we can ultimately achieve wellness in our communities.
You asked what I’ve been focused on as Surgeon General. Well, I’m focused on three main areas right now.
No. 1 is the opioid epidemic. It is a scourge across our country. A person dies every 12½ minutes from an opioid overdose and that’s far too many. Especially when we know that many of those deaths can be prevented.
Another area I’m focused on is demonstrating the link between community health and economic prosperity. We want folks to invest in health because we know that not only will it achieve better health for individuals and communities but it will create a more prosperous nation, also.
And finally, I’m raising awareness about the links between our nation’s health and our safety and security – particularly our national security. Unfortunately, 7 out of 10 young people between the ages of 18 and 24 years old in our country are ineligible for military service. That’s because they can’t pass the physical, they can’t meet the educational requirements, or they have a criminal record.
So, our nation’s poor health is not just a matter of diabetes or heart disease 20 or 30 years down the road. We are literally a less-safe country right now because we’re an unhealthy country.
Dr. Anderson: Regarding the opioid epidemic, what are some of the programs that are available today that you find effective? What would you like to see us do as a nation to respond to the epidemic?
Dr. Adams: Recently, I was at a hospital in Alaska where they have implemented a neonatal abstinence syndrome protocol and program that is being looked at around the country – and others are attempting to replicate it.
We know that if you keep mom and baby together, baby does better, mom does better, hospital stays are shorter, costs go down, and you’re keeping that family unit intact. This prevents future problems for both the baby and the mother. That’s just one small example.
I’m also very happy to see that the prescribing of opioids is going down 20%-25% across the country. And there are even larger decreases in the military and veteran communities. That’s really a testament to doctors and the medical profession finally waking up. And I say this as a physician myself, as an anesthesiologist, as someone who is involved in acute and chronic pain management.
Four out of five people with substance use disorder say they started with a prescription opioid. Many physicians will say, “Those aren’t my patients,” but unfortunately when we look at the PDMP [prescription drug monitoring program] data across the country we do a poor job of predicting who is and who isn’t going to divert. It may not be your patient, but it could be their son or the babysitter who is diverting those overprescribed opioids.
One thing that I really think we need to lean into as health care practitioners is providing medication-assisted treatment, or MAT. We know that the gold standard for treatment and recovery is medication-assisted treatment of some form. But we also know it’s not nearly available enough and that there are barriers on the federal and state levels.
We need you to continue to talk to your congressional representatives and let them know which barriers you perceive because the data waiver comes directly from Congress.
Still, any ER can prescribe up to 3 days of MAT to someone. I’d much rather have our ER doctors putting patients on MAT and then connecting them to treatment, than sending them back out into the arms of a drug dealer after they put them into acute withdrawal with naloxone.
We also have too many pregnant women who want help but can’t find any treatment because no one out there will take care of pregnant moms. We need folks to step up to the plate and get that data waiver in our ob/gyn and primary care sectors.
Ultimately, we need hospitals and health care leaders to create an environment that makes providers feel comfortable providing that service by giving them the training and the support to be able to do it.
We also need to make sure we’re co-prescribing naloxone for those who are at risk for opioid overdose.
Dr. Anderson: Just so we are clear, are you in favor of regular prescribing of naloxone, along with prescriptions for opioids? Is that correct?
Dr. Adams: I issued the first Surgeon General’s advisory from more than 10 years earlier this year to help folks understand that over half of our opioid overdoses occur in a home setting. We all know that an anoxic brain injury occurs in 4-5 minutes. We also know that most ambulances and first responders aren’t going to show up in 4-5 minutes.
If we want to make a dent in this overdose epidemic, we need everyone to consider themselves a first responder. We need to look at it the same as we look at CPR; we need everyone carrying naloxone. That was one of the big pushes from my Surgeon General’s advisory.
How can providers help? Well, they can coprescribe naloxone to folks on high morphine milligram equivalents (MME) who are at risk. If grandma has naloxone at home and her grandson overdoses in the garage, then at least it’s in the same house. Naloxone is not the treatment for the opioid epidemic. But we can’t get someone who is dead into treatment.
I have no illusions that simply making naloxone available is going to turn the tide, but it certainly is an important part of it.
Dr. Anderson: From your unique viewpoint, how much progress do you see in relation to the opioid epidemic? Do you think we’re approaching an inflection point, or do you think there’s a long way to go before this starts to turn around?
Dr. Adams: When I talk about the opioid epidemic, I have two angles. No.1, I want to raise awareness about the opioid epidemic – the severity of it, and how everyone can lean into it in their own way. Whether it’s community citizens, providers, law enforcement, the business community, whomever.
But in addition to raising awareness, I want to instill hope.
I was in Huntington, West Virginia, just a few weeks ago at the epicenter of the opioid epidemic. They’ve been able to turn their opioid overdose rates around by providing peer recovery coaches to individuals and making sure naloxone is available throughout the community. You save the life and then you connect them to care.
We know that the folks who are at highest risk for overdose deaths are the ones who just overdosed. They come out of the ER where we’ve watched them for a few hours and then we send them right back out into the arms of the drug dealer to do exactly what we know they will do medically because we’ve thrown them into withdrawal and they try to get their next fix.
If we can partner with law enforcement, then we can turn our opioid overdose rates around.
A story of recovery that I want to share with you is about a guy named Jonathan, who I met when I was in Rhode Island.
Jonathan overdosed, but his roommate had access to naloxone, which he administered. Jonathan was taken to the ER and then connected with a peer recovery coach. He is now in recovery and has actually become a peer recovery coach himself. Saving this one life will now enable us to save many more.
Yet we still prescribe more than 80% of the world’s opioids to less than 5% of the world’s population. So, we still have an overprescribing epidemic, but we’ve surpassed the inflection point there. Prescribing is coming down.
But another part of this epidemic was that we squeezed the balloon in one place and, as prescribing opioids went down, lots of people switched over to heroin. That’s when we really first started to see overdose rates go up.
Well, it’s important for folks to know that, through law enforcement, through partnerships with the public health community, through an increase in syringe service programs, and through other touch points, heroin use is now going down in most places.
Unfortunately, now we’re seeing the third wave of the epidemic, and that’s fentanyl and carfentanil.
Vice Admiral Jerome M. Adams, MD, MPH, is the 20th Surgeon General of the United States, a post created in 1871.
Dr. Adams holds degrees in biochemistry and psychology from the University of Maryland, Baltimore County; a master’s degree in public health from the University of California, Berkeley; and a medical degree from the Indiana University, Indianapolis. He is a board-certified anesthesiologist and associate clinical professor of anesthesia at Indiana University.
At the 2018 Executive Advisory Board meeting of the Doctors Company, Richard E. Anderson, MD, FACP, chairman and chief executive officer of the Doctors Company, spoke with Dr. Adams about the opioid epidemic’s enormous impact on communities and health services in the United States.
Dr. Anderson: Dr. Adams, you’ve been busy since taking over as Surgeon General of the United States. What are some of the key challenges that you’re facing in this office?
Dr. Adams: You know, there are many challenges facing our country, but it boils down to a lack of wellness. We know that only 10% of health is due to health care, 20% of health is genetics, and the rest is a combination of behavior and environment.
My motto is “better health through better partnerships,” because I firmly believe that if we break out of our silos and reach across the traditional barriers that have been put up by funding, by reimbursement, and by infrastructure, then we can ultimately achieve wellness in our communities.
You asked what I’ve been focused on as Surgeon General. Well, I’m focused on three main areas right now.
No. 1 is the opioid epidemic. It is a scourge across our country. A person dies every 12½ minutes from an opioid overdose and that’s far too many. Especially when we know that many of those deaths can be prevented.
Another area I’m focused on is demonstrating the link between community health and economic prosperity. We want folks to invest in health because we know that not only will it achieve better health for individuals and communities but it will create a more prosperous nation, also.
And finally, I’m raising awareness about the links between our nation’s health and our safety and security – particularly our national security. Unfortunately, 7 out of 10 young people between the ages of 18 and 24 years old in our country are ineligible for military service. That’s because they can’t pass the physical, they can’t meet the educational requirements, or they have a criminal record.
So, our nation’s poor health is not just a matter of diabetes or heart disease 20 or 30 years down the road. We are literally a less-safe country right now because we’re an unhealthy country.
Dr. Anderson: Regarding the opioid epidemic, what are some of the programs that are available today that you find effective? What would you like to see us do as a nation to respond to the epidemic?
Dr. Adams: Recently, I was at a hospital in Alaska where they have implemented a neonatal abstinence syndrome protocol and program that is being looked at around the country – and others are attempting to replicate it.
We know that if you keep mom and baby together, baby does better, mom does better, hospital stays are shorter, costs go down, and you’re keeping that family unit intact. This prevents future problems for both the baby and the mother. That’s just one small example.
I’m also very happy to see that the prescribing of opioids is going down 20%-25% across the country. And there are even larger decreases in the military and veteran communities. That’s really a testament to doctors and the medical profession finally waking up. And I say this as a physician myself, as an anesthesiologist, as someone who is involved in acute and chronic pain management.
Four out of five people with substance use disorder say they started with a prescription opioid. Many physicians will say, “Those aren’t my patients,” but unfortunately when we look at the PDMP [prescription drug monitoring program] data across the country we do a poor job of predicting who is and who isn’t going to divert. It may not be your patient, but it could be their son or the babysitter who is diverting those overprescribed opioids.
One thing that I really think we need to lean into as health care practitioners is providing medication-assisted treatment, or MAT. We know that the gold standard for treatment and recovery is medication-assisted treatment of some form. But we also know it’s not nearly available enough and that there are barriers on the federal and state levels.
We need you to continue to talk to your congressional representatives and let them know which barriers you perceive because the data waiver comes directly from Congress.
Still, any ER can prescribe up to 3 days of MAT to someone. I’d much rather have our ER doctors putting patients on MAT and then connecting them to treatment, than sending them back out into the arms of a drug dealer after they put them into acute withdrawal with naloxone.
We also have too many pregnant women who want help but can’t find any treatment because no one out there will take care of pregnant moms. We need folks to step up to the plate and get that data waiver in our ob/gyn and primary care sectors.
Ultimately, we need hospitals and health care leaders to create an environment that makes providers feel comfortable providing that service by giving them the training and the support to be able to do it.
We also need to make sure we’re co-prescribing naloxone for those who are at risk for opioid overdose.
Dr. Anderson: Just so we are clear, are you in favor of regular prescribing of naloxone, along with prescriptions for opioids? Is that correct?
Dr. Adams: I issued the first Surgeon General’s advisory from more than 10 years earlier this year to help folks understand that over half of our opioid overdoses occur in a home setting. We all know that an anoxic brain injury occurs in 4-5 minutes. We also know that most ambulances and first responders aren’t going to show up in 4-5 minutes.
If we want to make a dent in this overdose epidemic, we need everyone to consider themselves a first responder. We need to look at it the same as we look at CPR; we need everyone carrying naloxone. That was one of the big pushes from my Surgeon General’s advisory.
How can providers help? Well, they can coprescribe naloxone to folks on high morphine milligram equivalents (MME) who are at risk. If grandma has naloxone at home and her grandson overdoses in the garage, then at least it’s in the same house. Naloxone is not the treatment for the opioid epidemic. But we can’t get someone who is dead into treatment.
I have no illusions that simply making naloxone available is going to turn the tide, but it certainly is an important part of it.
Dr. Anderson: From your unique viewpoint, how much progress do you see in relation to the opioid epidemic? Do you think we’re approaching an inflection point, or do you think there’s a long way to go before this starts to turn around?
Dr. Adams: When I talk about the opioid epidemic, I have two angles. No.1, I want to raise awareness about the opioid epidemic – the severity of it, and how everyone can lean into it in their own way. Whether it’s community citizens, providers, law enforcement, the business community, whomever.
But in addition to raising awareness, I want to instill hope.
I was in Huntington, West Virginia, just a few weeks ago at the epicenter of the opioid epidemic. They’ve been able to turn their opioid overdose rates around by providing peer recovery coaches to individuals and making sure naloxone is available throughout the community. You save the life and then you connect them to care.
We know that the folks who are at highest risk for overdose deaths are the ones who just overdosed. They come out of the ER where we’ve watched them for a few hours and then we send them right back out into the arms of the drug dealer to do exactly what we know they will do medically because we’ve thrown them into withdrawal and they try to get their next fix.
If we can partner with law enforcement, then we can turn our opioid overdose rates around.
A story of recovery that I want to share with you is about a guy named Jonathan, who I met when I was in Rhode Island.
Jonathan overdosed, but his roommate had access to naloxone, which he administered. Jonathan was taken to the ER and then connected with a peer recovery coach. He is now in recovery and has actually become a peer recovery coach himself. Saving this one life will now enable us to save many more.
Yet we still prescribe more than 80% of the world’s opioids to less than 5% of the world’s population. So, we still have an overprescribing epidemic, but we’ve surpassed the inflection point there. Prescribing is coming down.
But another part of this epidemic was that we squeezed the balloon in one place and, as prescribing opioids went down, lots of people switched over to heroin. That’s when we really first started to see overdose rates go up.
Well, it’s important for folks to know that, through law enforcement, through partnerships with the public health community, through an increase in syringe service programs, and through other touch points, heroin use is now going down in most places.
Unfortunately, now we’re seeing the third wave of the epidemic, and that’s fentanyl and carfentanil.
Race/ethnicity, other factors predict PTSD and depression after mild TBI
Civilian patients with mild traumatic brain injury (TBI) who are black, have psychiatric history or lower education, or whose injury was caused by assault might be at greater risk of developing posttraumatic stress disorder or major depression, a longitudinal study suggests.
“Our findings may have implications for surveillance and treatment of mental disorders after TBI,” wrote Murray B. Stein, MD, MPH, and his associates. The study was published Jan. 30 in JAMA Psychiatry.
The researchers looked at the risk factors for and prevalence of posttraumatic stress disorder (PTSD) and major depressive disorder among 1,155 patients. The patients were enrolled at 11 level 1 trauma centers across the United States after they were evaluated for mild TBI in emergency departments as part of a prospective study called Transforming Research and Clinical Knowledge in Traumatic Brain Injury, or TRACK-TBI. The comparison group was 230 patients with nonhead orthopedic trauma injuries, wrote Dr. Stein, distinguished professor of psychiatry and family medicine and public health at the University of California, San Diego, and his associates.
They found that each additional year of education was associated with a significant 11% reduction in the risk of developing PTSD after mild TBI (P = .005). Also, black patients had a greater than fivefold higher risk of PTSD (P less than.001) than that of individuals who were not black.
Among patients with a history of mental illness and those who had experienced their injury as a result of assault or violence – as opposed to a motor vehicle accident or fall, for example – both had a greater than threefold higher risk of developing PTSD (odds ratio, 3.57 and 3.43 respectively). A prior TBI was nonsignificantly associated with an increased risk of developing PTSD.
Lower education duration, being black, or a history of mental illness also were all significantly associated with an increased risk of developing major depressive disorder after mild TBI.
However, duration of lost consciousness or posttraumatic amnesia, evidence of brain injury on CT, or hospitalization did not predict an increased risk of PTSD or major depression.
“Although MDD and PTSD are prevalent after TBI, little is known about which patients are at risk for developing them,” Dr. Stein and his associates wrote.
Noting that having a prior mental health problem was an “exceptionally strong” risk factor for PTSD and MDD after TBI, the authors said this could represent continuation or exacerbation of the prior mental health issue, or the triggering of a new episode in a person with a past history who had recovered.
“However, in either case this finding underscores the importance of clinicians being aware of the mental health history of their patients with [mild TBI], as this information is central to expectations regarding both short-term and long-term outcome,” they wrote.
Dr. Stein and his associates cited as a limitation their reliance on patient or family report. In addition, they said, the elevated risk for mental disorders among black individuals after mild TBI, which was independent of socioeconomic status or cause of injury, was not understood. “Unmeasured covariates may be part of the explanation; this is a topic needing further study,” they wrote.
The study was supported by the National Institutes of Health, the U.S. Department of Defense, Abbott Laboratories, and One Mind. Four authors declared consultancies, advisory board positions, speaking fees, and shares or stock options with the pharmaceutical and private industry. Two authors declared grants from the study sponsors.
SOURCE: Stein MB et al. JAMA Psychiatry. 2019. Jan 30. doi: 10.1001/jamapsychiatry.2018.4288.
Civilian patients with mild traumatic brain injury (TBI) who are black, have psychiatric history or lower education, or whose injury was caused by assault might be at greater risk of developing posttraumatic stress disorder or major depression, a longitudinal study suggests.
“Our findings may have implications for surveillance and treatment of mental disorders after TBI,” wrote Murray B. Stein, MD, MPH, and his associates. The study was published Jan. 30 in JAMA Psychiatry.
The researchers looked at the risk factors for and prevalence of posttraumatic stress disorder (PTSD) and major depressive disorder among 1,155 patients. The patients were enrolled at 11 level 1 trauma centers across the United States after they were evaluated for mild TBI in emergency departments as part of a prospective study called Transforming Research and Clinical Knowledge in Traumatic Brain Injury, or TRACK-TBI. The comparison group was 230 patients with nonhead orthopedic trauma injuries, wrote Dr. Stein, distinguished professor of psychiatry and family medicine and public health at the University of California, San Diego, and his associates.
They found that each additional year of education was associated with a significant 11% reduction in the risk of developing PTSD after mild TBI (P = .005). Also, black patients had a greater than fivefold higher risk of PTSD (P less than.001) than that of individuals who were not black.
Among patients with a history of mental illness and those who had experienced their injury as a result of assault or violence – as opposed to a motor vehicle accident or fall, for example – both had a greater than threefold higher risk of developing PTSD (odds ratio, 3.57 and 3.43 respectively). A prior TBI was nonsignificantly associated with an increased risk of developing PTSD.
Lower education duration, being black, or a history of mental illness also were all significantly associated with an increased risk of developing major depressive disorder after mild TBI.
However, duration of lost consciousness or posttraumatic amnesia, evidence of brain injury on CT, or hospitalization did not predict an increased risk of PTSD or major depression.
“Although MDD and PTSD are prevalent after TBI, little is known about which patients are at risk for developing them,” Dr. Stein and his associates wrote.
Noting that having a prior mental health problem was an “exceptionally strong” risk factor for PTSD and MDD after TBI, the authors said this could represent continuation or exacerbation of the prior mental health issue, or the triggering of a new episode in a person with a past history who had recovered.
“However, in either case this finding underscores the importance of clinicians being aware of the mental health history of their patients with [mild TBI], as this information is central to expectations regarding both short-term and long-term outcome,” they wrote.
Dr. Stein and his associates cited as a limitation their reliance on patient or family report. In addition, they said, the elevated risk for mental disorders among black individuals after mild TBI, which was independent of socioeconomic status or cause of injury, was not understood. “Unmeasured covariates may be part of the explanation; this is a topic needing further study,” they wrote.
The study was supported by the National Institutes of Health, the U.S. Department of Defense, Abbott Laboratories, and One Mind. Four authors declared consultancies, advisory board positions, speaking fees, and shares or stock options with the pharmaceutical and private industry. Two authors declared grants from the study sponsors.
SOURCE: Stein MB et al. JAMA Psychiatry. 2019. Jan 30. doi: 10.1001/jamapsychiatry.2018.4288.
Civilian patients with mild traumatic brain injury (TBI) who are black, have psychiatric history or lower education, or whose injury was caused by assault might be at greater risk of developing posttraumatic stress disorder or major depression, a longitudinal study suggests.
“Our findings may have implications for surveillance and treatment of mental disorders after TBI,” wrote Murray B. Stein, MD, MPH, and his associates. The study was published Jan. 30 in JAMA Psychiatry.
The researchers looked at the risk factors for and prevalence of posttraumatic stress disorder (PTSD) and major depressive disorder among 1,155 patients. The patients were enrolled at 11 level 1 trauma centers across the United States after they were evaluated for mild TBI in emergency departments as part of a prospective study called Transforming Research and Clinical Knowledge in Traumatic Brain Injury, or TRACK-TBI. The comparison group was 230 patients with nonhead orthopedic trauma injuries, wrote Dr. Stein, distinguished professor of psychiatry and family medicine and public health at the University of California, San Diego, and his associates.
They found that each additional year of education was associated with a significant 11% reduction in the risk of developing PTSD after mild TBI (P = .005). Also, black patients had a greater than fivefold higher risk of PTSD (P less than.001) than that of individuals who were not black.
Among patients with a history of mental illness and those who had experienced their injury as a result of assault or violence – as opposed to a motor vehicle accident or fall, for example – both had a greater than threefold higher risk of developing PTSD (odds ratio, 3.57 and 3.43 respectively). A prior TBI was nonsignificantly associated with an increased risk of developing PTSD.
Lower education duration, being black, or a history of mental illness also were all significantly associated with an increased risk of developing major depressive disorder after mild TBI.
However, duration of lost consciousness or posttraumatic amnesia, evidence of brain injury on CT, or hospitalization did not predict an increased risk of PTSD or major depression.
“Although MDD and PTSD are prevalent after TBI, little is known about which patients are at risk for developing them,” Dr. Stein and his associates wrote.
Noting that having a prior mental health problem was an “exceptionally strong” risk factor for PTSD and MDD after TBI, the authors said this could represent continuation or exacerbation of the prior mental health issue, or the triggering of a new episode in a person with a past history who had recovered.
“However, in either case this finding underscores the importance of clinicians being aware of the mental health history of their patients with [mild TBI], as this information is central to expectations regarding both short-term and long-term outcome,” they wrote.
Dr. Stein and his associates cited as a limitation their reliance on patient or family report. In addition, they said, the elevated risk for mental disorders among black individuals after mild TBI, which was independent of socioeconomic status or cause of injury, was not understood. “Unmeasured covariates may be part of the explanation; this is a topic needing further study,” they wrote.
The study was supported by the National Institutes of Health, the U.S. Department of Defense, Abbott Laboratories, and One Mind. Four authors declared consultancies, advisory board positions, speaking fees, and shares or stock options with the pharmaceutical and private industry. Two authors declared grants from the study sponsors.
SOURCE: Stein MB et al. JAMA Psychiatry. 2019. Jan 30. doi: 10.1001/jamapsychiatry.2018.4288.
FROM JAMA PSYCHIATRY
Key clinical point: The findings underscore “the importance of clinicians being aware of the mental health history of their patients with [mild TBI], as this information is central to expectations regarding both short-term and long-term outcome.”
Major finding: Black patients have fivefold higher risk of PTSD after brain injury.
Study details: Longitudinal cohort study of 1,155 patients with mild traumatic brain injury.
Disclosures: The study was supported by the National Institutes of Health, the U.S. Department of Defense, Abbott Laboratories, and One Mind. Four authors declared consultancies, advisory board positions, and speaking fees, shares, or stock options with the pharmaceutical and private industry. Two authors declared grants from the study sponsors.
Source: Stein MB et al. JAMA Psychiatry 2019. Jan 30. doi: 10.1001/jamapsychiatry.2018.4288.
The fog may be lifting
One of the common symptoms described by postconcussion patients is that their heads feel a bit foggy. It may not be simply by chance that “foggy” is the best word to describe the atmosphere surrounding the entire field of concussion diagnosis and management.
Back in the Dark Ages, when the diagnosis of concussion was a simpler binary call, the issue of management seldom created much discussion. If the patient lost consciousness or was amnesic, he (it was less frequently she) could return to activity when his headache was gone and he could remember what he was supposed to do when the quarterback called for a “Red 34, Drive Right Smash” play. That may have even been during the second half of the game in which he was injured.
As it became more widely understood that the diagnosis of concussion didn’t require loss of consciousness and that repeated concussions could have serious sequelae, management became a bit fuzzier. No one had thought much about the recuperative process. Into this vacuum came a wide variety of researchers and providers. Not surprisingly, much of their advice was based on unproven assumptions, including the concept of “brain rest.”
It has taken time, but fortunately, folks with patience and wisdom have questioned these assumptions and begun collecting data. The result of these investigations and others has prompted the American Academy of Pediatrics to publish an updated set of guidelines on concussion management that includes the observation that extended school absence may slow the rehabilitation process (Pediatrics. 2018 Dec. doi: 10.1542/peds.2018-3074).
It is becoming clear that management of concussion can be rather complex and must be individualized to each patient. In my experience, the postconcussion period can unmask behavioral, cognitive, and emotional problems that were preexisting but had received little or no attention. For example, the trauma of the event may trigger anxiety about further injury or exacerbate depression that had been building for years. The student who “couldn’t do algebra” following a head injury may have had a lifelong learning disability that had gone unnoticed. The student athlete with prolonged postconcussion symptoms may indeed have another more serious problem. Hopefully, the new guidelines from the AAP will be a first step toward a more thoughtful and scientifically driven approach to concussion management.
It would be nice if that approach could filter down to the management of the more common but less dramatic pediatric injuries. There is hope. Choosing Wisely – a patient/parent–targeted initiative by the American Board of Internal Medicine Foundation in cooperation with the AAP – points out that, although half of the pediatric head injury patients seen in emergency departments received CT scan, only a third of those studies were indicated. Parents are encouraged to learn more about the risks of CT scans and question the physician when one is recommended.
But, doctors’ habits and old wives’ tales die slowly. I hope that you no longer recommend that parents keep their children awake after a head injury, or wake them every hour to check their pupils. Those counterproductive recommendations make about as much sense as staying out of the swimming pool for an hour after eating a chocolate chip cookie.
Dr. Wilkoff practiced primary care pediatrics in Brunswick, Maine, for nearly 40 years. He has authored several books on behavioral pediatrics, including “How to Say No to Your Toddler.” Email him at [email protected].
One of the common symptoms described by postconcussion patients is that their heads feel a bit foggy. It may not be simply by chance that “foggy” is the best word to describe the atmosphere surrounding the entire field of concussion diagnosis and management.
Back in the Dark Ages, when the diagnosis of concussion was a simpler binary call, the issue of management seldom created much discussion. If the patient lost consciousness or was amnesic, he (it was less frequently she) could return to activity when his headache was gone and he could remember what he was supposed to do when the quarterback called for a “Red 34, Drive Right Smash” play. That may have even been during the second half of the game in which he was injured.
As it became more widely understood that the diagnosis of concussion didn’t require loss of consciousness and that repeated concussions could have serious sequelae, management became a bit fuzzier. No one had thought much about the recuperative process. Into this vacuum came a wide variety of researchers and providers. Not surprisingly, much of their advice was based on unproven assumptions, including the concept of “brain rest.”
It has taken time, but fortunately, folks with patience and wisdom have questioned these assumptions and begun collecting data. The result of these investigations and others has prompted the American Academy of Pediatrics to publish an updated set of guidelines on concussion management that includes the observation that extended school absence may slow the rehabilitation process (Pediatrics. 2018 Dec. doi: 10.1542/peds.2018-3074).
It is becoming clear that management of concussion can be rather complex and must be individualized to each patient. In my experience, the postconcussion period can unmask behavioral, cognitive, and emotional problems that were preexisting but had received little or no attention. For example, the trauma of the event may trigger anxiety about further injury or exacerbate depression that had been building for years. The student who “couldn’t do algebra” following a head injury may have had a lifelong learning disability that had gone unnoticed. The student athlete with prolonged postconcussion symptoms may indeed have another more serious problem. Hopefully, the new guidelines from the AAP will be a first step toward a more thoughtful and scientifically driven approach to concussion management.
It would be nice if that approach could filter down to the management of the more common but less dramatic pediatric injuries. There is hope. Choosing Wisely – a patient/parent–targeted initiative by the American Board of Internal Medicine Foundation in cooperation with the AAP – points out that, although half of the pediatric head injury patients seen in emergency departments received CT scan, only a third of those studies were indicated. Parents are encouraged to learn more about the risks of CT scans and question the physician when one is recommended.
But, doctors’ habits and old wives’ tales die slowly. I hope that you no longer recommend that parents keep their children awake after a head injury, or wake them every hour to check their pupils. Those counterproductive recommendations make about as much sense as staying out of the swimming pool for an hour after eating a chocolate chip cookie.
Dr. Wilkoff practiced primary care pediatrics in Brunswick, Maine, for nearly 40 years. He has authored several books on behavioral pediatrics, including “How to Say No to Your Toddler.” Email him at [email protected].
One of the common symptoms described by postconcussion patients is that their heads feel a bit foggy. It may not be simply by chance that “foggy” is the best word to describe the atmosphere surrounding the entire field of concussion diagnosis and management.
Back in the Dark Ages, when the diagnosis of concussion was a simpler binary call, the issue of management seldom created much discussion. If the patient lost consciousness or was amnesic, he (it was less frequently she) could return to activity when his headache was gone and he could remember what he was supposed to do when the quarterback called for a “Red 34, Drive Right Smash” play. That may have even been during the second half of the game in which he was injured.
As it became more widely understood that the diagnosis of concussion didn’t require loss of consciousness and that repeated concussions could have serious sequelae, management became a bit fuzzier. No one had thought much about the recuperative process. Into this vacuum came a wide variety of researchers and providers. Not surprisingly, much of their advice was based on unproven assumptions, including the concept of “brain rest.”
It has taken time, but fortunately, folks with patience and wisdom have questioned these assumptions and begun collecting data. The result of these investigations and others has prompted the American Academy of Pediatrics to publish an updated set of guidelines on concussion management that includes the observation that extended school absence may slow the rehabilitation process (Pediatrics. 2018 Dec. doi: 10.1542/peds.2018-3074).
It is becoming clear that management of concussion can be rather complex and must be individualized to each patient. In my experience, the postconcussion period can unmask behavioral, cognitive, and emotional problems that were preexisting but had received little or no attention. For example, the trauma of the event may trigger anxiety about further injury or exacerbate depression that had been building for years. The student who “couldn’t do algebra” following a head injury may have had a lifelong learning disability that had gone unnoticed. The student athlete with prolonged postconcussion symptoms may indeed have another more serious problem. Hopefully, the new guidelines from the AAP will be a first step toward a more thoughtful and scientifically driven approach to concussion management.
It would be nice if that approach could filter down to the management of the more common but less dramatic pediatric injuries. There is hope. Choosing Wisely – a patient/parent–targeted initiative by the American Board of Internal Medicine Foundation in cooperation with the AAP – points out that, although half of the pediatric head injury patients seen in emergency departments received CT scan, only a third of those studies were indicated. Parents are encouraged to learn more about the risks of CT scans and question the physician when one is recommended.
But, doctors’ habits and old wives’ tales die slowly. I hope that you no longer recommend that parents keep their children awake after a head injury, or wake them every hour to check their pupils. Those counterproductive recommendations make about as much sense as staying out of the swimming pool for an hour after eating a chocolate chip cookie.
Dr. Wilkoff practiced primary care pediatrics in Brunswick, Maine, for nearly 40 years. He has authored several books on behavioral pediatrics, including “How to Say No to Your Toddler.” Email him at [email protected].
SPRINT MIND published: Extension trial to add 2 years’ follow-up
A new iteration of the SPRINT MIND hypertension trial will seek to prove conclusively the original study’s tantalizing suggestion: that intensive blood pressure control decreases the risk of developing mild cognitive impairment (MCI) and, eventually, dementia.
SPRINT MIND 2.0 will re-recruit SPRINT MIND subjects and enable another follow-up cognitive assessment and other clinical tests as they remain on their standard of care blood pressure regimen. It is largely funded by an $800,000 grant from the Alzheimer’s Association.
Initially released last July at the Alzheimer’s Association International Conference, the results of the SPRINT MIND have now appeared online in JAMA. Although it failed to meet its primary endpoint of reducing dementia incidence, the study did score on two secondary endpoints. Patients who reduced their systolic blood pressure to less than 120 mm Hg were 19% less likely to develop MCI and 17% less likely to be diagnosed with all-cause dementia than were those who achieved a hypertension target of less than 140 mm Hg.
The secondary results, and positive movement in the primary results, sparked excitement in the dementia research community last summer. They have suggested that the median 5-year follow-up just wasn’t long enough to show any significant effects on dementia, which can take years to fully manifest. Adding 2 more years with SPRINT MIND 2.0 should be long enough to discern those benefits, if indeed they exist.
“SPRINT MIND 2.0 and the work leading up to it offers genuine, concrete hope,” Maria C. Carrillo, PhD, chief science officer for the Alzheimer’s Association, said in a press statement. “MCI is a known risk factor for dementia, and everyone who experiences dementia passes through MCI. When you prevent new cases of MCI, you are preventing new cases of dementia. The Alzheimer’s Association finds these data to be compelling and is committed to getting clarity and certainty on the dementia outcome by following participants for a longer period of time.”
The study strengthens the new and energetic push to find ways to prevent dementia, which has proven itself intractable in every drug study to date.
“This study is in line with where the field of dementia research is going: preventing memory loss earlier,” said Laurie Ryan, PhD, chief of the dementias of aging branch in the National Institute on Aging. “Much like we have research-based interventions for heart health and cancer prevention, we hope to have guidance based on this and subsequent studies that will more definitively show how to slow or even stop dementia well before symptoms appear.”
NIA director Richard J. Hodes, MD, agreed.
“Dementia continues to be a large public health challenge, and based on the primary results of this study, we still have yet to find an intervention strategy proven to reduce the risk of dementia,” he said in a press statement. “Nevertheless, the secondary results showing that intensive lowering of blood pressure may reduce risk for MCI, a known risk factor for dementia, gives us additional avenues to explore on the path to prevention.”
SPRINT MIND was a substudy of the Systolic Blood Pressure Intervention Trial (SPRINT). It compared two strategies for managing hypertension in older adults. The intensive strategy had a target of less than 120 mm Hg, while standard care had a target of less than 140 mm Hg. SPRINT showed that more intensive blood pressure control produced a 25% reduction in the composite primary composite endpoint of cardiovascular events, stroke, and cardiovascular death. The intensive arm was so successful that SPRINT helped inform the 2017 high blood pressure clinical guidelines from the American Heart Association and American College of Cardiology.
The SPRINT MIND substudy, headed by Jeff D. Williamson, MD, of Wake Forest University, Winston-Salem, NC, asked whether intensive management had any effect on probable all-cause dementia or MCI, as well as imaging evidence of changes in white matter lesions and brain volume. It followed patients for up to 7 years and comprised 9,361 SPRINT subjects at least 50 years old (mean, 68 years) with at least one cardiovascular risk factor. Nearly a third (30%) were black, and 10% Hispanic. The primary outcome was incident probable dementia. Secondary outcomes were MCI and a composite of MCI and/or probable dementia. About a third had a SBP of 132 mm Hg or less, another third had a systolic pressure of 132-145 mm Hg, and the remainder had a systolic pressure greater than 145 mm Hg.
Physicians could use their choice of antihypertensive treatments. The study protocol encouraged, but did not mandate, thiazide-type diuretics as a first-line agent, followed by loop diuretics and beta-adrenergic blockers. Chlorthalidone was encouraged as the primary thiazide-type diuretic, and amlodipine as the preferred calcium-channel blocker.
The interventions did successfully control blood pressure, with a significant difference between the treatment groups. The mean SBP was 121.6 mm Hg in the intensive therapy group and 134.8 mm Hg in the standard group – a statistically significant difference of 13.3 mm Hg.
Dementia developed in 149 in the aggressive control group and 176 in the standard group – a nonsignificant difference of 17% (hazard ratio, 0.83). MCI developed in 287 in the intensive group and 353 in the standard treatment group. This amounted to a statistically significant 19% reduction. There was also a significant 15% reduction in the composite outcome of MCI or probable dementia in favor of intensive treatment.
As evidenced by the Alzheimer’s Association grant, dementia researchers chose to focus on SPRINT MIND’s positive secondary endpoints. At the AAIC meeting, Dr. Williamson even suggested that antihypertensive medications could be seen as disease-modifying agents for cognitive decline. Data support his claim: No dementia intervention yet tested has approached this level of success.
“I think we can say this is the first disease-modifying strategy to reduce the risk of MCI,” Dr. Williamson said during a press briefing. And although the primary endpoint – the 17% relative risk reduction for probable all-cause dementia – didn’t meet statistical significance, “It’s comforting to see that the benefit went in the same direction and was of the same magnitude..”
SOURCE: Williamson JD et al. JAMA 2019 Jan 28. doi:10.1001/jama.2018.21442.
SPRINT MIND offers hope that a very achievable blood pressure goal can dramatically alter the trajectory from mild cognitive impairment to dementia, Kristine Yaffe, MD, wrote in an accompanying editorial. But at this point, it’s impossible to make specific clinical recommendations.
Additionally it is not possible, right now, to know which hypertension treatment regimens were most effective in improved cognitive outcomes.
“Information necessary to compare the effects of classes of antihypertensive agents on cognitive outcomes is also not provided. SPRINT used a quasi-pragmatic approach with suggestions for treatment choice, but practitioners approached SBP control individually, and most participants were taking multiple drugs.”
Nevertheless, the positive secondary findings and the encouraging trajectory on dementia risk should fix blood pressure management squarely into a cornerstone of dementia prevention algorithms.
“The SPRINT MIND study may not be the final approach for prevention of AD or other cognitive impairment, but it represents a major leap forward in what has emerged as a marathon journey.”
Dr. Kristine Yaffe is professor of psychiatry, neurology and epidemiology and the Roy and Marie Scola Endowed Chair at the University of California, San Francisco.
SPRINT MIND offers hope that a very achievable blood pressure goal can dramatically alter the trajectory from mild cognitive impairment to dementia, Kristine Yaffe, MD, wrote in an accompanying editorial. But at this point, it’s impossible to make specific clinical recommendations.
Additionally it is not possible, right now, to know which hypertension treatment regimens were most effective in improved cognitive outcomes.
“Information necessary to compare the effects of classes of antihypertensive agents on cognitive outcomes is also not provided. SPRINT used a quasi-pragmatic approach with suggestions for treatment choice, but practitioners approached SBP control individually, and most participants were taking multiple drugs.”
Nevertheless, the positive secondary findings and the encouraging trajectory on dementia risk should fix blood pressure management squarely into a cornerstone of dementia prevention algorithms.
“The SPRINT MIND study may not be the final approach for prevention of AD or other cognitive impairment, but it represents a major leap forward in what has emerged as a marathon journey.”
Dr. Kristine Yaffe is professor of psychiatry, neurology and epidemiology and the Roy and Marie Scola Endowed Chair at the University of California, San Francisco.
SPRINT MIND offers hope that a very achievable blood pressure goal can dramatically alter the trajectory from mild cognitive impairment to dementia, Kristine Yaffe, MD, wrote in an accompanying editorial. But at this point, it’s impossible to make specific clinical recommendations.
Additionally it is not possible, right now, to know which hypertension treatment regimens were most effective in improved cognitive outcomes.
“Information necessary to compare the effects of classes of antihypertensive agents on cognitive outcomes is also not provided. SPRINT used a quasi-pragmatic approach with suggestions for treatment choice, but practitioners approached SBP control individually, and most participants were taking multiple drugs.”
Nevertheless, the positive secondary findings and the encouraging trajectory on dementia risk should fix blood pressure management squarely into a cornerstone of dementia prevention algorithms.
“The SPRINT MIND study may not be the final approach for prevention of AD or other cognitive impairment, but it represents a major leap forward in what has emerged as a marathon journey.”
Dr. Kristine Yaffe is professor of psychiatry, neurology and epidemiology and the Roy and Marie Scola Endowed Chair at the University of California, San Francisco.
A new iteration of the SPRINT MIND hypertension trial will seek to prove conclusively the original study’s tantalizing suggestion: that intensive blood pressure control decreases the risk of developing mild cognitive impairment (MCI) and, eventually, dementia.
SPRINT MIND 2.0 will re-recruit SPRINT MIND subjects and enable another follow-up cognitive assessment and other clinical tests as they remain on their standard of care blood pressure regimen. It is largely funded by an $800,000 grant from the Alzheimer’s Association.
Initially released last July at the Alzheimer’s Association International Conference, the results of the SPRINT MIND have now appeared online in JAMA. Although it failed to meet its primary endpoint of reducing dementia incidence, the study did score on two secondary endpoints. Patients who reduced their systolic blood pressure to less than 120 mm Hg were 19% less likely to develop MCI and 17% less likely to be diagnosed with all-cause dementia than were those who achieved a hypertension target of less than 140 mm Hg.
The secondary results, and positive movement in the primary results, sparked excitement in the dementia research community last summer. They have suggested that the median 5-year follow-up just wasn’t long enough to show any significant effects on dementia, which can take years to fully manifest. Adding 2 more years with SPRINT MIND 2.0 should be long enough to discern those benefits, if indeed they exist.
“SPRINT MIND 2.0 and the work leading up to it offers genuine, concrete hope,” Maria C. Carrillo, PhD, chief science officer for the Alzheimer’s Association, said in a press statement. “MCI is a known risk factor for dementia, and everyone who experiences dementia passes through MCI. When you prevent new cases of MCI, you are preventing new cases of dementia. The Alzheimer’s Association finds these data to be compelling and is committed to getting clarity and certainty on the dementia outcome by following participants for a longer period of time.”
The study strengthens the new and energetic push to find ways to prevent dementia, which has proven itself intractable in every drug study to date.
“This study is in line with where the field of dementia research is going: preventing memory loss earlier,” said Laurie Ryan, PhD, chief of the dementias of aging branch in the National Institute on Aging. “Much like we have research-based interventions for heart health and cancer prevention, we hope to have guidance based on this and subsequent studies that will more definitively show how to slow or even stop dementia well before symptoms appear.”
NIA director Richard J. Hodes, MD, agreed.
“Dementia continues to be a large public health challenge, and based on the primary results of this study, we still have yet to find an intervention strategy proven to reduce the risk of dementia,” he said in a press statement. “Nevertheless, the secondary results showing that intensive lowering of blood pressure may reduce risk for MCI, a known risk factor for dementia, gives us additional avenues to explore on the path to prevention.”
SPRINT MIND was a substudy of the Systolic Blood Pressure Intervention Trial (SPRINT). It compared two strategies for managing hypertension in older adults. The intensive strategy had a target of less than 120 mm Hg, while standard care had a target of less than 140 mm Hg. SPRINT showed that more intensive blood pressure control produced a 25% reduction in the composite primary composite endpoint of cardiovascular events, stroke, and cardiovascular death. The intensive arm was so successful that SPRINT helped inform the 2017 high blood pressure clinical guidelines from the American Heart Association and American College of Cardiology.
The SPRINT MIND substudy, headed by Jeff D. Williamson, MD, of Wake Forest University, Winston-Salem, NC, asked whether intensive management had any effect on probable all-cause dementia or MCI, as well as imaging evidence of changes in white matter lesions and brain volume. It followed patients for up to 7 years and comprised 9,361 SPRINT subjects at least 50 years old (mean, 68 years) with at least one cardiovascular risk factor. Nearly a third (30%) were black, and 10% Hispanic. The primary outcome was incident probable dementia. Secondary outcomes were MCI and a composite of MCI and/or probable dementia. About a third had a SBP of 132 mm Hg or less, another third had a systolic pressure of 132-145 mm Hg, and the remainder had a systolic pressure greater than 145 mm Hg.
Physicians could use their choice of antihypertensive treatments. The study protocol encouraged, but did not mandate, thiazide-type diuretics as a first-line agent, followed by loop diuretics and beta-adrenergic blockers. Chlorthalidone was encouraged as the primary thiazide-type diuretic, and amlodipine as the preferred calcium-channel blocker.
The interventions did successfully control blood pressure, with a significant difference between the treatment groups. The mean SBP was 121.6 mm Hg in the intensive therapy group and 134.8 mm Hg in the standard group – a statistically significant difference of 13.3 mm Hg.
Dementia developed in 149 in the aggressive control group and 176 in the standard group – a nonsignificant difference of 17% (hazard ratio, 0.83). MCI developed in 287 in the intensive group and 353 in the standard treatment group. This amounted to a statistically significant 19% reduction. There was also a significant 15% reduction in the composite outcome of MCI or probable dementia in favor of intensive treatment.
As evidenced by the Alzheimer’s Association grant, dementia researchers chose to focus on SPRINT MIND’s positive secondary endpoints. At the AAIC meeting, Dr. Williamson even suggested that antihypertensive medications could be seen as disease-modifying agents for cognitive decline. Data support his claim: No dementia intervention yet tested has approached this level of success.
“I think we can say this is the first disease-modifying strategy to reduce the risk of MCI,” Dr. Williamson said during a press briefing. And although the primary endpoint – the 17% relative risk reduction for probable all-cause dementia – didn’t meet statistical significance, “It’s comforting to see that the benefit went in the same direction and was of the same magnitude..”
SOURCE: Williamson JD et al. JAMA 2019 Jan 28. doi:10.1001/jama.2018.21442.
A new iteration of the SPRINT MIND hypertension trial will seek to prove conclusively the original study’s tantalizing suggestion: that intensive blood pressure control decreases the risk of developing mild cognitive impairment (MCI) and, eventually, dementia.
SPRINT MIND 2.0 will re-recruit SPRINT MIND subjects and enable another follow-up cognitive assessment and other clinical tests as they remain on their standard of care blood pressure regimen. It is largely funded by an $800,000 grant from the Alzheimer’s Association.
Initially released last July at the Alzheimer’s Association International Conference, the results of the SPRINT MIND have now appeared online in JAMA. Although it failed to meet its primary endpoint of reducing dementia incidence, the study did score on two secondary endpoints. Patients who reduced their systolic blood pressure to less than 120 mm Hg were 19% less likely to develop MCI and 17% less likely to be diagnosed with all-cause dementia than were those who achieved a hypertension target of less than 140 mm Hg.
The secondary results, and positive movement in the primary results, sparked excitement in the dementia research community last summer. They have suggested that the median 5-year follow-up just wasn’t long enough to show any significant effects on dementia, which can take years to fully manifest. Adding 2 more years with SPRINT MIND 2.0 should be long enough to discern those benefits, if indeed they exist.
“SPRINT MIND 2.0 and the work leading up to it offers genuine, concrete hope,” Maria C. Carrillo, PhD, chief science officer for the Alzheimer’s Association, said in a press statement. “MCI is a known risk factor for dementia, and everyone who experiences dementia passes through MCI. When you prevent new cases of MCI, you are preventing new cases of dementia. The Alzheimer’s Association finds these data to be compelling and is committed to getting clarity and certainty on the dementia outcome by following participants for a longer period of time.”
The study strengthens the new and energetic push to find ways to prevent dementia, which has proven itself intractable in every drug study to date.
“This study is in line with where the field of dementia research is going: preventing memory loss earlier,” said Laurie Ryan, PhD, chief of the dementias of aging branch in the National Institute on Aging. “Much like we have research-based interventions for heart health and cancer prevention, we hope to have guidance based on this and subsequent studies that will more definitively show how to slow or even stop dementia well before symptoms appear.”
NIA director Richard J. Hodes, MD, agreed.
“Dementia continues to be a large public health challenge, and based on the primary results of this study, we still have yet to find an intervention strategy proven to reduce the risk of dementia,” he said in a press statement. “Nevertheless, the secondary results showing that intensive lowering of blood pressure may reduce risk for MCI, a known risk factor for dementia, gives us additional avenues to explore on the path to prevention.”
SPRINT MIND was a substudy of the Systolic Blood Pressure Intervention Trial (SPRINT). It compared two strategies for managing hypertension in older adults. The intensive strategy had a target of less than 120 mm Hg, while standard care had a target of less than 140 mm Hg. SPRINT showed that more intensive blood pressure control produced a 25% reduction in the composite primary composite endpoint of cardiovascular events, stroke, and cardiovascular death. The intensive arm was so successful that SPRINT helped inform the 2017 high blood pressure clinical guidelines from the American Heart Association and American College of Cardiology.
The SPRINT MIND substudy, headed by Jeff D. Williamson, MD, of Wake Forest University, Winston-Salem, NC, asked whether intensive management had any effect on probable all-cause dementia or MCI, as well as imaging evidence of changes in white matter lesions and brain volume. It followed patients for up to 7 years and comprised 9,361 SPRINT subjects at least 50 years old (mean, 68 years) with at least one cardiovascular risk factor. Nearly a third (30%) were black, and 10% Hispanic. The primary outcome was incident probable dementia. Secondary outcomes were MCI and a composite of MCI and/or probable dementia. About a third had a SBP of 132 mm Hg or less, another third had a systolic pressure of 132-145 mm Hg, and the remainder had a systolic pressure greater than 145 mm Hg.
Physicians could use their choice of antihypertensive treatments. The study protocol encouraged, but did not mandate, thiazide-type diuretics as a first-line agent, followed by loop diuretics and beta-adrenergic blockers. Chlorthalidone was encouraged as the primary thiazide-type diuretic, and amlodipine as the preferred calcium-channel blocker.
The interventions did successfully control blood pressure, with a significant difference between the treatment groups. The mean SBP was 121.6 mm Hg in the intensive therapy group and 134.8 mm Hg in the standard group – a statistically significant difference of 13.3 mm Hg.
Dementia developed in 149 in the aggressive control group and 176 in the standard group – a nonsignificant difference of 17% (hazard ratio, 0.83). MCI developed in 287 in the intensive group and 353 in the standard treatment group. This amounted to a statistically significant 19% reduction. There was also a significant 15% reduction in the composite outcome of MCI or probable dementia in favor of intensive treatment.
As evidenced by the Alzheimer’s Association grant, dementia researchers chose to focus on SPRINT MIND’s positive secondary endpoints. At the AAIC meeting, Dr. Williamson even suggested that antihypertensive medications could be seen as disease-modifying agents for cognitive decline. Data support his claim: No dementia intervention yet tested has approached this level of success.
“I think we can say this is the first disease-modifying strategy to reduce the risk of MCI,” Dr. Williamson said during a press briefing. And although the primary endpoint – the 17% relative risk reduction for probable all-cause dementia – didn’t meet statistical significance, “It’s comforting to see that the benefit went in the same direction and was of the same magnitude..”
SOURCE: Williamson JD et al. JAMA 2019 Jan 28. doi:10.1001/jama.2018.21442.
FROM JAMA
Key clinical point: Keeping systolic blood pressure lower than 120 mm Hg did not significantly reduce the risk of all-cause dementia in patients with hypertension, but it did lower the risk of mild cognitive impairment and probable dementia.
Major finding: The intensively treated group had a nonsignificant 17% lower risk of dementia, and significant reductions in the risk of MCI (19%) and probable dementia (15%).
Study details: SPRINT MIND was a substudy of the SPRINT antihypertension trial.
Source: Williamson JD et al. JAMA 2019 Jan 28. doi:10.1001/jama.2018.21442.
Benefit of thrombectomy may be universal
The location of the arterial occlusive lesion and the imaging technique used to select patients for the procedure also do not influence the therapy’s benefits, the researchers said. Although the proportional benefit of thrombectomy plus medical management is uniform across subgroups, compared with medical management alone, patients may have different amounts of absolute benefit.
The results of the DEFUSE 3 (Endovascular Therapy Following Imaging Evaluation for Ischemic Stroke) trial, which were published in 2018, indicated that endovascular thrombectomy provided clinical benefits for patients with acute ischemic stroke if administered at 6-16 hours after stroke onset. As part of the trial’s prespecified analyses, Maarten G. Lansberg, MD, PhD, associate professor of neurology and neurological sciences at Stanford (Calif.) University Medical Center in California, and his colleagues sought to determine whether thrombectomy had uniform benefit among various patient subgroups (e.g., elderly people, patients with mild symptoms, and those who present late after onset).
A total of 296 patients were enrolled in the randomized, open-label, blinded-endpoint DEFUSE 3 trial at 38 sites in the United States. Eligible participants had acute ischemic stroke resulting from an occlusion of the internal carotid artery or middle cerebral artery and evidence of salvageable tissue on perfusion CT or MRI. In all, 182 patients met these criteria and were randomized and included in the intention-to-treat analysis. The researchers stopped DEFUSE 3 early because of efficacy.
The study’s primary endpoint was functional outcome at day 90, as measured with the modified Rankin Scale. Dr. Lansberg and his colleagues performed multivariate ordinal logistic regression to calculate the adjusted proportional association between endovascular treatment and clinical outcome among participants of various ages, baseline stroke severities, periods between onset and treatment, locations of the arterial occlusion, and imaging modalities, such as CT or MRI, used to identify salvageable tissue.
The population’s median age was 70 years, and 51% of participants were women. The median National Institutes of Health Stroke Scale score was 16. When the researchers considered the whole sample, they found that younger age, lower baseline NIHSS score, and lower serum glucose level independently predicted better functional outcome. The common odds ratio for improved functional outcome with endovascular therapy, adjusted for these variables, was 3.1. Age, NIHSS score, time to randomization, imaging modality, and location of the arterial occlusion did not interact significantly with treatment effect.
“Our results indicate that advanced age, up to 90 years, should not be considered a contraindication to thrombectomy, provided that the patient is fully independent prior to stroke onset,” said the researchers. “Although age did not modify the treatment effect, it was a strong independent predictor of 90-day disability, which is consistent with prior studies of both tissue plasminogen activator and endovascular therapy.”
The trial’s small sample size may have allowed small differences between groups to pass unnoticed, said the reseachers. Other trials of late-window thrombectomy will be required to validate these results, they concluded.
The National Institute for Neurological Disorders and Stroke supported the study through grants. Several investigators received consulting fees from and hold shares in iSchemaView, which manufactures the software that the investigators used for postprocessing of CT and MRI perfusion studies. Other authors received consulting fees from various pharmaceutical and medical device companies, including Genentech, Medtronic, Pfizer, and Stryker Neurovascular.
SOURCE: Lansberg MG et al. JAMA Neurol. 2019 Jan 28. doi: 10.1001/jamaneurol.2018.4587.
The location of the arterial occlusive lesion and the imaging technique used to select patients for the procedure also do not influence the therapy’s benefits, the researchers said. Although the proportional benefit of thrombectomy plus medical management is uniform across subgroups, compared with medical management alone, patients may have different amounts of absolute benefit.
The results of the DEFUSE 3 (Endovascular Therapy Following Imaging Evaluation for Ischemic Stroke) trial, which were published in 2018, indicated that endovascular thrombectomy provided clinical benefits for patients with acute ischemic stroke if administered at 6-16 hours after stroke onset. As part of the trial’s prespecified analyses, Maarten G. Lansberg, MD, PhD, associate professor of neurology and neurological sciences at Stanford (Calif.) University Medical Center in California, and his colleagues sought to determine whether thrombectomy had uniform benefit among various patient subgroups (e.g., elderly people, patients with mild symptoms, and those who present late after onset).
A total of 296 patients were enrolled in the randomized, open-label, blinded-endpoint DEFUSE 3 trial at 38 sites in the United States. Eligible participants had acute ischemic stroke resulting from an occlusion of the internal carotid artery or middle cerebral artery and evidence of salvageable tissue on perfusion CT or MRI. In all, 182 patients met these criteria and were randomized and included in the intention-to-treat analysis. The researchers stopped DEFUSE 3 early because of efficacy.
The study’s primary endpoint was functional outcome at day 90, as measured with the modified Rankin Scale. Dr. Lansberg and his colleagues performed multivariate ordinal logistic regression to calculate the adjusted proportional association between endovascular treatment and clinical outcome among participants of various ages, baseline stroke severities, periods between onset and treatment, locations of the arterial occlusion, and imaging modalities, such as CT or MRI, used to identify salvageable tissue.
The population’s median age was 70 years, and 51% of participants were women. The median National Institutes of Health Stroke Scale score was 16. When the researchers considered the whole sample, they found that younger age, lower baseline NIHSS score, and lower serum glucose level independently predicted better functional outcome. The common odds ratio for improved functional outcome with endovascular therapy, adjusted for these variables, was 3.1. Age, NIHSS score, time to randomization, imaging modality, and location of the arterial occlusion did not interact significantly with treatment effect.
“Our results indicate that advanced age, up to 90 years, should not be considered a contraindication to thrombectomy, provided that the patient is fully independent prior to stroke onset,” said the researchers. “Although age did not modify the treatment effect, it was a strong independent predictor of 90-day disability, which is consistent with prior studies of both tissue plasminogen activator and endovascular therapy.”
The trial’s small sample size may have allowed small differences between groups to pass unnoticed, said the reseachers. Other trials of late-window thrombectomy will be required to validate these results, they concluded.
The National Institute for Neurological Disorders and Stroke supported the study through grants. Several investigators received consulting fees from and hold shares in iSchemaView, which manufactures the software that the investigators used for postprocessing of CT and MRI perfusion studies. Other authors received consulting fees from various pharmaceutical and medical device companies, including Genentech, Medtronic, Pfizer, and Stryker Neurovascular.
SOURCE: Lansberg MG et al. JAMA Neurol. 2019 Jan 28. doi: 10.1001/jamaneurol.2018.4587.
The location of the arterial occlusive lesion and the imaging technique used to select patients for the procedure also do not influence the therapy’s benefits, the researchers said. Although the proportional benefit of thrombectomy plus medical management is uniform across subgroups, compared with medical management alone, patients may have different amounts of absolute benefit.
The results of the DEFUSE 3 (Endovascular Therapy Following Imaging Evaluation for Ischemic Stroke) trial, which were published in 2018, indicated that endovascular thrombectomy provided clinical benefits for patients with acute ischemic stroke if administered at 6-16 hours after stroke onset. As part of the trial’s prespecified analyses, Maarten G. Lansberg, MD, PhD, associate professor of neurology and neurological sciences at Stanford (Calif.) University Medical Center in California, and his colleagues sought to determine whether thrombectomy had uniform benefit among various patient subgroups (e.g., elderly people, patients with mild symptoms, and those who present late after onset).
A total of 296 patients were enrolled in the randomized, open-label, blinded-endpoint DEFUSE 3 trial at 38 sites in the United States. Eligible participants had acute ischemic stroke resulting from an occlusion of the internal carotid artery or middle cerebral artery and evidence of salvageable tissue on perfusion CT or MRI. In all, 182 patients met these criteria and were randomized and included in the intention-to-treat analysis. The researchers stopped DEFUSE 3 early because of efficacy.
The study’s primary endpoint was functional outcome at day 90, as measured with the modified Rankin Scale. Dr. Lansberg and his colleagues performed multivariate ordinal logistic regression to calculate the adjusted proportional association between endovascular treatment and clinical outcome among participants of various ages, baseline stroke severities, periods between onset and treatment, locations of the arterial occlusion, and imaging modalities, such as CT or MRI, used to identify salvageable tissue.
The population’s median age was 70 years, and 51% of participants were women. The median National Institutes of Health Stroke Scale score was 16. When the researchers considered the whole sample, they found that younger age, lower baseline NIHSS score, and lower serum glucose level independently predicted better functional outcome. The common odds ratio for improved functional outcome with endovascular therapy, adjusted for these variables, was 3.1. Age, NIHSS score, time to randomization, imaging modality, and location of the arterial occlusion did not interact significantly with treatment effect.
“Our results indicate that advanced age, up to 90 years, should not be considered a contraindication to thrombectomy, provided that the patient is fully independent prior to stroke onset,” said the researchers. “Although age did not modify the treatment effect, it was a strong independent predictor of 90-day disability, which is consistent with prior studies of both tissue plasminogen activator and endovascular therapy.”
The trial’s small sample size may have allowed small differences between groups to pass unnoticed, said the reseachers. Other trials of late-window thrombectomy will be required to validate these results, they concluded.
The National Institute for Neurological Disorders and Stroke supported the study through grants. Several investigators received consulting fees from and hold shares in iSchemaView, which manufactures the software that the investigators used for postprocessing of CT and MRI perfusion studies. Other authors received consulting fees from various pharmaceutical and medical device companies, including Genentech, Medtronic, Pfizer, and Stryker Neurovascular.
SOURCE: Lansberg MG et al. JAMA Neurol. 2019 Jan 28. doi: 10.1001/jamaneurol.2018.4587.
FROM JAMA NEUROLOGY
Key clinical point: Age, symptom severity, and serum glucose do not influence the benefit of thrombectomy for acute ischemic stroke.
Major finding: The adjusted common odds ratio for improved functional outcome with endovascular therapy was 3.1.
Study details: The randomized, open-label, blinded-end-point DEFUSE 3 trial included 182 patients.
Disclosures: The National Institute for Neurological Disorders and Stroke funded the study through grants.
Source: Lansberg MG et al. JAMA Neurol. 2019 Jan 28. doi: 10.1001/jamaneurol.2018.4587.