User login
CagA-positive H. pylori patients at higher risk of osteoporosis, fracture
A new study has found that older patients who test positive for the cytotoxin associated gene-A (CagA) strain of Helicobacter pylori may be more at risk of both osteoporosis and fractures.
“Further studies will be required to replicate these findings in other cohorts and to better clarify the underlying pathogenetic mechanisms leading to increased bone fragility in subjects infected by CagA-positive H. pylori strains,” wrote Luigi Gennari, MD, PhD, of the University of Siena (Italy), and coauthors. The study was published in the Journal of Bone and Mineral Research.
To determine the effects of H. pylori on bone health and potential fracture risk, the researchers launched a population-based cohort study of 1,149 adults between the ages of 50 and 80 in Siena. The cohort comprised 174 males with an average (SD) age of 65.9 (plus or minus 6 years) and 975 females with an average age of 62.5 (plus or minus 6 years). All subjects were examined for H. pylori antibodies, and those who were infected were also examined for anti-CagA serum antibodies. As blood was sampled, bone mineral density (BMD) of the lumbar spine, femoral neck, total hip, and total body was measured via dual-energy x-ray absorptiometry.
In total, 53% of male participants and 49% of female participants tested positive for H. pylori, with CagA-positive strains found in 27% of males and 26% of females. No differences in infection rates were discovered in regard to socioeconomic status, age, weight, or height. Patients with normal BMD (45%), osteoporosis (51%), or osteopenia (49%) had similar prevalence of H. pylori infection, but CagA-positive strains were more frequently found in osteoporotic (30%) and osteopenic (26%) patients, compared to patients with normal BMD (21%, P < .01). CagA-positive female patients also had lower lumbar (0.950 g/cm2) and femoral (0.795 g/cm2) BMD, compared to CagA-negative (0.987 and 0.813 g/cm2) or H. pylori-negative women (0.997 and 0.821 g/cm2), respectively.
After an average follow-up period of 11.8 years, 199 nontraumatic fractures (72 vertebral and 127 nonvertebral) had occurred in 158 participants. Patients with CagA-positive strains of H. pylori had significantly increased risk of a clinical vertebral fracture (hazard ratio [HR], 5.27; 95% confidence interval, 2.23-12.63; P < .0001) or a nonvertebral incident fracture (HR, 2.09; 95% CI, 1.27-2.46; P < .01), compared to patients without H. pylori. After adjustment for age, sex, and body mass index, the risk among CagA-positive patients remained similarly significantly elevated for both vertebral (aHR, 4.78; 95% CI, 1.99-11.47; P < .0001) and nonvertebral fractures (aHR, 2.04; 95% CI, 1.22-3.41; P < .01).
The authors acknowledged their study’s limitations, including a cohort that was notably low in male participants, an inability to assess the effects of eradicating H. pylori on bone, and uncertainty as to which specific effects of H. pylori infection increase the risk of osteoporosis or fracture. Along those lines, they noted that an association between serum CagA antibody titer and gastric mucosal inflammation could lead to malabsorption of calcium, hypothesizing that antibody titer rather than antibody positivity “might be a more relevant marker for assessing the risk of bone fragility in patients affected by H. pylori infection.”
The study was supported in part by a grant from the Italian Association for Osteoporosis. The authors reported no potential conflicts of interest.
SOURCE: Gennari L et al. J Bone Miner Res. 2020 Aug 13. doi: 10.1002/jbmr.4162.
A new study has found that older patients who test positive for the cytotoxin associated gene-A (CagA) strain of Helicobacter pylori may be more at risk of both osteoporosis and fractures.
“Further studies will be required to replicate these findings in other cohorts and to better clarify the underlying pathogenetic mechanisms leading to increased bone fragility in subjects infected by CagA-positive H. pylori strains,” wrote Luigi Gennari, MD, PhD, of the University of Siena (Italy), and coauthors. The study was published in the Journal of Bone and Mineral Research.
To determine the effects of H. pylori on bone health and potential fracture risk, the researchers launched a population-based cohort study of 1,149 adults between the ages of 50 and 80 in Siena. The cohort comprised 174 males with an average (SD) age of 65.9 (plus or minus 6 years) and 975 females with an average age of 62.5 (plus or minus 6 years). All subjects were examined for H. pylori antibodies, and those who were infected were also examined for anti-CagA serum antibodies. As blood was sampled, bone mineral density (BMD) of the lumbar spine, femoral neck, total hip, and total body was measured via dual-energy x-ray absorptiometry.
In total, 53% of male participants and 49% of female participants tested positive for H. pylori, with CagA-positive strains found in 27% of males and 26% of females. No differences in infection rates were discovered in regard to socioeconomic status, age, weight, or height. Patients with normal BMD (45%), osteoporosis (51%), or osteopenia (49%) had similar prevalence of H. pylori infection, but CagA-positive strains were more frequently found in osteoporotic (30%) and osteopenic (26%) patients, compared to patients with normal BMD (21%, P < .01). CagA-positive female patients also had lower lumbar (0.950 g/cm2) and femoral (0.795 g/cm2) BMD, compared to CagA-negative (0.987 and 0.813 g/cm2) or H. pylori-negative women (0.997 and 0.821 g/cm2), respectively.
After an average follow-up period of 11.8 years, 199 nontraumatic fractures (72 vertebral and 127 nonvertebral) had occurred in 158 participants. Patients with CagA-positive strains of H. pylori had significantly increased risk of a clinical vertebral fracture (hazard ratio [HR], 5.27; 95% confidence interval, 2.23-12.63; P < .0001) or a nonvertebral incident fracture (HR, 2.09; 95% CI, 1.27-2.46; P < .01), compared to patients without H. pylori. After adjustment for age, sex, and body mass index, the risk among CagA-positive patients remained similarly significantly elevated for both vertebral (aHR, 4.78; 95% CI, 1.99-11.47; P < .0001) and nonvertebral fractures (aHR, 2.04; 95% CI, 1.22-3.41; P < .01).
The authors acknowledged their study’s limitations, including a cohort that was notably low in male participants, an inability to assess the effects of eradicating H. pylori on bone, and uncertainty as to which specific effects of H. pylori infection increase the risk of osteoporosis or fracture. Along those lines, they noted that an association between serum CagA antibody titer and gastric mucosal inflammation could lead to malabsorption of calcium, hypothesizing that antibody titer rather than antibody positivity “might be a more relevant marker for assessing the risk of bone fragility in patients affected by H. pylori infection.”
The study was supported in part by a grant from the Italian Association for Osteoporosis. The authors reported no potential conflicts of interest.
SOURCE: Gennari L et al. J Bone Miner Res. 2020 Aug 13. doi: 10.1002/jbmr.4162.
A new study has found that older patients who test positive for the cytotoxin associated gene-A (CagA) strain of Helicobacter pylori may be more at risk of both osteoporosis and fractures.
“Further studies will be required to replicate these findings in other cohorts and to better clarify the underlying pathogenetic mechanisms leading to increased bone fragility in subjects infected by CagA-positive H. pylori strains,” wrote Luigi Gennari, MD, PhD, of the University of Siena (Italy), and coauthors. The study was published in the Journal of Bone and Mineral Research.
To determine the effects of H. pylori on bone health and potential fracture risk, the researchers launched a population-based cohort study of 1,149 adults between the ages of 50 and 80 in Siena. The cohort comprised 174 males with an average (SD) age of 65.9 (plus or minus 6 years) and 975 females with an average age of 62.5 (plus or minus 6 years). All subjects were examined for H. pylori antibodies, and those who were infected were also examined for anti-CagA serum antibodies. As blood was sampled, bone mineral density (BMD) of the lumbar spine, femoral neck, total hip, and total body was measured via dual-energy x-ray absorptiometry.
In total, 53% of male participants and 49% of female participants tested positive for H. pylori, with CagA-positive strains found in 27% of males and 26% of females. No differences in infection rates were discovered in regard to socioeconomic status, age, weight, or height. Patients with normal BMD (45%), osteoporosis (51%), or osteopenia (49%) had similar prevalence of H. pylori infection, but CagA-positive strains were more frequently found in osteoporotic (30%) and osteopenic (26%) patients, compared to patients with normal BMD (21%, P < .01). CagA-positive female patients also had lower lumbar (0.950 g/cm2) and femoral (0.795 g/cm2) BMD, compared to CagA-negative (0.987 and 0.813 g/cm2) or H. pylori-negative women (0.997 and 0.821 g/cm2), respectively.
After an average follow-up period of 11.8 years, 199 nontraumatic fractures (72 vertebral and 127 nonvertebral) had occurred in 158 participants. Patients with CagA-positive strains of H. pylori had significantly increased risk of a clinical vertebral fracture (hazard ratio [HR], 5.27; 95% confidence interval, 2.23-12.63; P < .0001) or a nonvertebral incident fracture (HR, 2.09; 95% CI, 1.27-2.46; P < .01), compared to patients without H. pylori. After adjustment for age, sex, and body mass index, the risk among CagA-positive patients remained similarly significantly elevated for both vertebral (aHR, 4.78; 95% CI, 1.99-11.47; P < .0001) and nonvertebral fractures (aHR, 2.04; 95% CI, 1.22-3.41; P < .01).
The authors acknowledged their study’s limitations, including a cohort that was notably low in male participants, an inability to assess the effects of eradicating H. pylori on bone, and uncertainty as to which specific effects of H. pylori infection increase the risk of osteoporosis or fracture. Along those lines, they noted that an association between serum CagA antibody titer and gastric mucosal inflammation could lead to malabsorption of calcium, hypothesizing that antibody titer rather than antibody positivity “might be a more relevant marker for assessing the risk of bone fragility in patients affected by H. pylori infection.”
The study was supported in part by a grant from the Italian Association for Osteoporosis. The authors reported no potential conflicts of interest.
SOURCE: Gennari L et al. J Bone Miner Res. 2020 Aug 13. doi: 10.1002/jbmr.4162.
FROM THE JOURNAL OF BONE AND MINERAL RESEARCH
Atypical fractures with bisphosphonates highest in Asians, study confirms
The latest findings regarding the risk for atypical femur fracture (AFF) with use of bisphosphonates for osteoporosis show a significant increase in risk when treatment extends beyond 5 years. The risk is notably higher risk among Asian women, compared with White women. However, the benefits in fracture reduction still appear to far outweigh the risk for AFF.
The research, published in the New England Journal of Medicine, importantly adds to findings from smaller studies by showing effects in a population of nearly 200,000 women in a diverse cohort, said Angela M. Cheung, MD, PhD.
“This study answers some important questions – Kaiser Permanente Southern California is a large health maintenance organization with a diverse racial population,” said Dr. Cheung, director of the Center of Excellence in Skeletal Health Assessment and osteoporosis program at the University of Toronto.
“This is the first study that included a diverse population to definitively show that Asians are at a much higher risk of atypical femur fractures than Caucasians,” she emphasized.
Although AFFs are rare, concerns about them remain pressing in the treatment of osteoporosis, Dr. Cheung noted. “This is a big concern for clinicians – they want to do no harm.”
Risk for AFF increases with longer duration of bisphosphonate use
For the study, Dennis M. Black, PhD, of the departments of epidemiology and biostatistics and orthopedic surgery at the University of California, San Francisco, and colleagues identified women aged 50 years or older enrolled in the Kaiser Permanente Southern California system who were treated with bisphosphonates and were followed from January 2007 to November 2017.
Among the 196,129 women identified in the study, 277 AFFs occurred.
After multivariate adjustment, compared with those treated for less than 3 months, for women who were treated for 3-5 years, the hazard ratio for experiencing an AFF was 8.86. For therapy of 5-8 years, the HR increased to 19.88, and for those treated with bisphosphonates for 8 years or longer, the HR was 43.51.
The risk for AFF declined quickly upon bisphosphonate discontinuation; compared with current users, the HR dropped to 0.52 within 3-15 months after the last bisphosphonate use. It declined to 0.26 at more than 4 years after discontinuation.
The risk for AFF with bisphosphonate use was higher for Asian women than for White women (HR, 4.84); this did not apply to any other ethnic groups (HR, 0.99).
Other risk factors for AFF included shorter height (HR, 1.28 per 5-cm decrement), greater weight (HR, 1.15 per 5-kg increment), and glucocorticoid use (HR, 2.28 for glucocorticoid use of 1 or more years).
Among White women, the number of fractures prevented with bisphosphonate use far outweighed the risk for bisphosphonate-associated AFFs.
For example, among White women, during a 3-year treatment period, there were two bisphosphonate-associated AFFs, whereas 149 hip fractures and 541 clinical fractures were prevented, the authors wrote.
After 5 years, there were eight AFFs, but 286 hip fractures and 859 clinical fractures were prevented.
Although the risk-benefit ratio among Asian women still favored prevention of fractures, the difference was less pronounced – eight bisphosphonate-associated AFFs had occurred at 3 years, whereas 91 hip fractures and 330 clinical fractures were prevented.
The authors noted that previous studies have also shown Asian women to be at a disproportionately higher risk for AFF.
An earlier Kaiser Permanente Southern California case series showed that 49% of 142 AFFs occurred in Asian patients, despite the fact that those patients made up only 10% of the study population.
Various factors could cause higher risk in Asian women
The reasons for the increased risk among Asian women are likely multifactorial and could include greater medication adherence among Asian women, genetic differences in drug metabolism and bone turnover, and, notably, increased lateral stress caused by bowed Asian femora, the authors speculated.
Further questions include whether the risk is limited to Asians living outside of Asia and whether cultural differences in diet or physical activity are risk factors, they added.
“At this early stage, further research into the cause of the increased risk among women of Asian ancestry is warranted,” they wrote.
Although the risk for AFF may be higher among Asian women, the incidence of hip and other osteoporotic fractures is lower among Asians as well as other non-White persons, compared with White persons, they added.
The findings have important implications in how clinicians should discuss treatment options with different patient groups, Dr. Cheung said.
“I think this is one of the key findings of the study,” she added. “In this day and age of personalized medicine, we need to keep the individual patient in mind, and that includes their racial/ethnic background, genetic characteristics, sex, medical conditions and medications, etc. So it is important for physicians to pay attention to this. The risk-benefit ratio of these drugs for Asians will be quite different, compared to Caucasians.”
No link between traditional fracture risk factors and AFF, study shows
Interestingly, although older age, previous fractures, and lower bone mineral density are key risk factors for hip and other osteoporotic fractures in the general population, they do not significantly increase the risk for AFF with bisphosphonate use, the study also showed.
“In fact, the oldest women in our cohort, who are at highest risk for hip and other fractures, were at lowest risk for AFF,” the authors wrote.
The collective findings “add to the risk-benefit balance of bisphosphonate treatment in these populations and could directly affect decisions regarding treatment initiation and duration.”
Notable limitations of the study include the fact that most women were treated with one particular bisphosphonate, alendronate, and that other bisphosphonates were underrepresented, Dr. Cheung said.
“This study examined bisphosphonate therapy, but the vast majority of the women were exposed to alendronate, so whether women on risedronate or other bisphosphonates have similar risks is unclear,” she observed.
“In addition, because they can only capture bisphosphonate use using their database, any bisphosphonate exposure prior to joining Kaiser Permanente will not be captured. So the study may underestimate the total cumulative duration of bisphosphonate use,” she added.
The study received support from Kaiser Permanente and discretionary funds from the University of California, San Francisco. The study began with a pilot grant from Merck Sharp & Dohme, which had no role in the conduct of the study. Dr. Cheung has served as a consultant for Amgen. She chaired and led the 2019 International Society for Clinical Densitometry Position Development Conference on Detection of Atypical Femur Fractures and currently is on the Osteoporosis Canada Guidelines Committee.
A version of this article originally appeared on Medscape.com.
The latest findings regarding the risk for atypical femur fracture (AFF) with use of bisphosphonates for osteoporosis show a significant increase in risk when treatment extends beyond 5 years. The risk is notably higher risk among Asian women, compared with White women. However, the benefits in fracture reduction still appear to far outweigh the risk for AFF.
The research, published in the New England Journal of Medicine, importantly adds to findings from smaller studies by showing effects in a population of nearly 200,000 women in a diverse cohort, said Angela M. Cheung, MD, PhD.
“This study answers some important questions – Kaiser Permanente Southern California is a large health maintenance organization with a diverse racial population,” said Dr. Cheung, director of the Center of Excellence in Skeletal Health Assessment and osteoporosis program at the University of Toronto.
“This is the first study that included a diverse population to definitively show that Asians are at a much higher risk of atypical femur fractures than Caucasians,” she emphasized.
Although AFFs are rare, concerns about them remain pressing in the treatment of osteoporosis, Dr. Cheung noted. “This is a big concern for clinicians – they want to do no harm.”
Risk for AFF increases with longer duration of bisphosphonate use
For the study, Dennis M. Black, PhD, of the departments of epidemiology and biostatistics and orthopedic surgery at the University of California, San Francisco, and colleagues identified women aged 50 years or older enrolled in the Kaiser Permanente Southern California system who were treated with bisphosphonates and were followed from January 2007 to November 2017.
Among the 196,129 women identified in the study, 277 AFFs occurred.
After multivariate adjustment, compared with those treated for less than 3 months, for women who were treated for 3-5 years, the hazard ratio for experiencing an AFF was 8.86. For therapy of 5-8 years, the HR increased to 19.88, and for those treated with bisphosphonates for 8 years or longer, the HR was 43.51.
The risk for AFF declined quickly upon bisphosphonate discontinuation; compared with current users, the HR dropped to 0.52 within 3-15 months after the last bisphosphonate use. It declined to 0.26 at more than 4 years after discontinuation.
The risk for AFF with bisphosphonate use was higher for Asian women than for White women (HR, 4.84); this did not apply to any other ethnic groups (HR, 0.99).
Other risk factors for AFF included shorter height (HR, 1.28 per 5-cm decrement), greater weight (HR, 1.15 per 5-kg increment), and glucocorticoid use (HR, 2.28 for glucocorticoid use of 1 or more years).
Among White women, the number of fractures prevented with bisphosphonate use far outweighed the risk for bisphosphonate-associated AFFs.
For example, among White women, during a 3-year treatment period, there were two bisphosphonate-associated AFFs, whereas 149 hip fractures and 541 clinical fractures were prevented, the authors wrote.
After 5 years, there were eight AFFs, but 286 hip fractures and 859 clinical fractures were prevented.
Although the risk-benefit ratio among Asian women still favored prevention of fractures, the difference was less pronounced – eight bisphosphonate-associated AFFs had occurred at 3 years, whereas 91 hip fractures and 330 clinical fractures were prevented.
The authors noted that previous studies have also shown Asian women to be at a disproportionately higher risk for AFF.
An earlier Kaiser Permanente Southern California case series showed that 49% of 142 AFFs occurred in Asian patients, despite the fact that those patients made up only 10% of the study population.
Various factors could cause higher risk in Asian women
The reasons for the increased risk among Asian women are likely multifactorial and could include greater medication adherence among Asian women, genetic differences in drug metabolism and bone turnover, and, notably, increased lateral stress caused by bowed Asian femora, the authors speculated.
Further questions include whether the risk is limited to Asians living outside of Asia and whether cultural differences in diet or physical activity are risk factors, they added.
“At this early stage, further research into the cause of the increased risk among women of Asian ancestry is warranted,” they wrote.
Although the risk for AFF may be higher among Asian women, the incidence of hip and other osteoporotic fractures is lower among Asians as well as other non-White persons, compared with White persons, they added.
The findings have important implications in how clinicians should discuss treatment options with different patient groups, Dr. Cheung said.
“I think this is one of the key findings of the study,” she added. “In this day and age of personalized medicine, we need to keep the individual patient in mind, and that includes their racial/ethnic background, genetic characteristics, sex, medical conditions and medications, etc. So it is important for physicians to pay attention to this. The risk-benefit ratio of these drugs for Asians will be quite different, compared to Caucasians.”
No link between traditional fracture risk factors and AFF, study shows
Interestingly, although older age, previous fractures, and lower bone mineral density are key risk factors for hip and other osteoporotic fractures in the general population, they do not significantly increase the risk for AFF with bisphosphonate use, the study also showed.
“In fact, the oldest women in our cohort, who are at highest risk for hip and other fractures, were at lowest risk for AFF,” the authors wrote.
The collective findings “add to the risk-benefit balance of bisphosphonate treatment in these populations and could directly affect decisions regarding treatment initiation and duration.”
Notable limitations of the study include the fact that most women were treated with one particular bisphosphonate, alendronate, and that other bisphosphonates were underrepresented, Dr. Cheung said.
“This study examined bisphosphonate therapy, but the vast majority of the women were exposed to alendronate, so whether women on risedronate or other bisphosphonates have similar risks is unclear,” she observed.
“In addition, because they can only capture bisphosphonate use using their database, any bisphosphonate exposure prior to joining Kaiser Permanente will not be captured. So the study may underestimate the total cumulative duration of bisphosphonate use,” she added.
The study received support from Kaiser Permanente and discretionary funds from the University of California, San Francisco. The study began with a pilot grant from Merck Sharp & Dohme, which had no role in the conduct of the study. Dr. Cheung has served as a consultant for Amgen. She chaired and led the 2019 International Society for Clinical Densitometry Position Development Conference on Detection of Atypical Femur Fractures and currently is on the Osteoporosis Canada Guidelines Committee.
A version of this article originally appeared on Medscape.com.
The latest findings regarding the risk for atypical femur fracture (AFF) with use of bisphosphonates for osteoporosis show a significant increase in risk when treatment extends beyond 5 years. The risk is notably higher risk among Asian women, compared with White women. However, the benefits in fracture reduction still appear to far outweigh the risk for AFF.
The research, published in the New England Journal of Medicine, importantly adds to findings from smaller studies by showing effects in a population of nearly 200,000 women in a diverse cohort, said Angela M. Cheung, MD, PhD.
“This study answers some important questions – Kaiser Permanente Southern California is a large health maintenance organization with a diverse racial population,” said Dr. Cheung, director of the Center of Excellence in Skeletal Health Assessment and osteoporosis program at the University of Toronto.
“This is the first study that included a diverse population to definitively show that Asians are at a much higher risk of atypical femur fractures than Caucasians,” she emphasized.
Although AFFs are rare, concerns about them remain pressing in the treatment of osteoporosis, Dr. Cheung noted. “This is a big concern for clinicians – they want to do no harm.”
Risk for AFF increases with longer duration of bisphosphonate use
For the study, Dennis M. Black, PhD, of the departments of epidemiology and biostatistics and orthopedic surgery at the University of California, San Francisco, and colleagues identified women aged 50 years or older enrolled in the Kaiser Permanente Southern California system who were treated with bisphosphonates and were followed from January 2007 to November 2017.
Among the 196,129 women identified in the study, 277 AFFs occurred.
After multivariate adjustment, compared with those treated for less than 3 months, for women who were treated for 3-5 years, the hazard ratio for experiencing an AFF was 8.86. For therapy of 5-8 years, the HR increased to 19.88, and for those treated with bisphosphonates for 8 years or longer, the HR was 43.51.
The risk for AFF declined quickly upon bisphosphonate discontinuation; compared with current users, the HR dropped to 0.52 within 3-15 months after the last bisphosphonate use. It declined to 0.26 at more than 4 years after discontinuation.
The risk for AFF with bisphosphonate use was higher for Asian women than for White women (HR, 4.84); this did not apply to any other ethnic groups (HR, 0.99).
Other risk factors for AFF included shorter height (HR, 1.28 per 5-cm decrement), greater weight (HR, 1.15 per 5-kg increment), and glucocorticoid use (HR, 2.28 for glucocorticoid use of 1 or more years).
Among White women, the number of fractures prevented with bisphosphonate use far outweighed the risk for bisphosphonate-associated AFFs.
For example, among White women, during a 3-year treatment period, there were two bisphosphonate-associated AFFs, whereas 149 hip fractures and 541 clinical fractures were prevented, the authors wrote.
After 5 years, there were eight AFFs, but 286 hip fractures and 859 clinical fractures were prevented.
Although the risk-benefit ratio among Asian women still favored prevention of fractures, the difference was less pronounced – eight bisphosphonate-associated AFFs had occurred at 3 years, whereas 91 hip fractures and 330 clinical fractures were prevented.
The authors noted that previous studies have also shown Asian women to be at a disproportionately higher risk for AFF.
An earlier Kaiser Permanente Southern California case series showed that 49% of 142 AFFs occurred in Asian patients, despite the fact that those patients made up only 10% of the study population.
Various factors could cause higher risk in Asian women
The reasons for the increased risk among Asian women are likely multifactorial and could include greater medication adherence among Asian women, genetic differences in drug metabolism and bone turnover, and, notably, increased lateral stress caused by bowed Asian femora, the authors speculated.
Further questions include whether the risk is limited to Asians living outside of Asia and whether cultural differences in diet or physical activity are risk factors, they added.
“At this early stage, further research into the cause of the increased risk among women of Asian ancestry is warranted,” they wrote.
Although the risk for AFF may be higher among Asian women, the incidence of hip and other osteoporotic fractures is lower among Asians as well as other non-White persons, compared with White persons, they added.
The findings have important implications in how clinicians should discuss treatment options with different patient groups, Dr. Cheung said.
“I think this is one of the key findings of the study,” she added. “In this day and age of personalized medicine, we need to keep the individual patient in mind, and that includes their racial/ethnic background, genetic characteristics, sex, medical conditions and medications, etc. So it is important for physicians to pay attention to this. The risk-benefit ratio of these drugs for Asians will be quite different, compared to Caucasians.”
No link between traditional fracture risk factors and AFF, study shows
Interestingly, although older age, previous fractures, and lower bone mineral density are key risk factors for hip and other osteoporotic fractures in the general population, they do not significantly increase the risk for AFF with bisphosphonate use, the study also showed.
“In fact, the oldest women in our cohort, who are at highest risk for hip and other fractures, were at lowest risk for AFF,” the authors wrote.
The collective findings “add to the risk-benefit balance of bisphosphonate treatment in these populations and could directly affect decisions regarding treatment initiation and duration.”
Notable limitations of the study include the fact that most women were treated with one particular bisphosphonate, alendronate, and that other bisphosphonates were underrepresented, Dr. Cheung said.
“This study examined bisphosphonate therapy, but the vast majority of the women were exposed to alendronate, so whether women on risedronate or other bisphosphonates have similar risks is unclear,” she observed.
“In addition, because they can only capture bisphosphonate use using their database, any bisphosphonate exposure prior to joining Kaiser Permanente will not be captured. So the study may underestimate the total cumulative duration of bisphosphonate use,” she added.
The study received support from Kaiser Permanente and discretionary funds from the University of California, San Francisco. The study began with a pilot grant from Merck Sharp & Dohme, which had no role in the conduct of the study. Dr. Cheung has served as a consultant for Amgen. She chaired and led the 2019 International Society for Clinical Densitometry Position Development Conference on Detection of Atypical Femur Fractures and currently is on the Osteoporosis Canada Guidelines Committee.
A version of this article originally appeared on Medscape.com.
SGLT2 inhibitors with metformin look safe for bone
The combination of sodium-glucose transporter-2 (SGLT-2) inhibitors and metformin is not associated with an increase in fracture risk among patients with type 2 diabetes (T2D), according to a new meta-analysis of 25 randomized, controlled trials.
Researchers at The Second Clinical College of Dalian Medical University in Jiangsu, China, compared fracture risk associated with the metformin/SLGT2 combination to metformin alone as well as other T2D therapeutics, and found no differences in risk. The study was published online Aug. 11 in Osteoporosis International.
T2D is associated with an increased risk of fracture, though causative mechanisms remain uncertain. Some lines of evidence suggest multiple factors may contribute to fractures, including hyperglycemia, oxidative stress, toxic effects of advanced glycosylation end-products, altered insulin levels, and treatment-induced hypoglycemia, as well as an association between T2D and increased risk of falls.
Antidiabetes drugs can have positive or negative effects on bone. thiazolidinediones, insulin, and sulfonylureas may increase risk of fractures, while dipeptidyl peptidase-4 (DPP-4) inhibitors and glucagon-like peptide-2 (GLP-2) receptor agonists may be protective. Metformin may also reduce fracture risk.
SGLT-2 inhibitors interrupt glucose reabsorption in the kidney, leading to improved glycemic control. Other benefits include improved renal and cardiovascular outcomes, weight loss, and reduced blood pressure, liver fat, and serum uric acid levels.
These properties have made SGLT-2 inhibitors combined with metformin an important therapy for patients at high risk of atherosclerotic disease, or who have heart failure or chronic kidney disease.
But SGLT-2 inhibition increases osmotic diuresis, and this could alter the mineral balance within bone. Some studies also showed that SGLT-2 inhibitors led to changes in bone turnover markers, bone mineral density, and bone microarchitecture. Observational studies of the SGLT-2 inhibitor canagliflozin found associations with a higher rate of fracture risk in patients taking the drug.
Such studies carry the risk of confounding factors, so the researchers took advantage of the fact that many recent clinical trials have examined the impact of SGLT-2 inhibitors on T2D. They pooled data from 25 clinical trials with a total of 19,500 participants, 9,662 of whom received SGLT-2 inhibitors plus metformin; 9,838 received other active comparators.
The fracture rate was 0.91% in the SGLT-2 inhibitors/metformin group, and 0.80% among controls (odds ratio, 0.97; 95% CI, 0.71-1.32), with no heterogeneity. Metformin alone was not associated with a change in fracture rate (OR, 0.95; 95% CI, 0.44-2.08), nor were other forms of diabetes control (OR, 0.95; 95% CI, 0.69-1.31).
There were some differences in fracture risk among SGLT-2 inhibitors when studied individually, though none differed significantly from controls. The highest risk was associated with the canagliflozin/metformin (OR, 2.19; 95% CI, 0.66-7.27), followed by dapagliflozin/metformin (OR, 0.91; 95% CI, 0.50-1.64), empagliflozin/metformin (OR, 0.94; 95% CI, 0.59-1.50), and ertugliflozin/metformin (OR, 0.76; 95% CI, 0.38-1.54).
There were no differences with respect to hip or lumbar spine fractures, or other fractures. The researchers found no differences in bone mineral density or bone turnover markers.
The meta-analysis is limited by the relatively short average follow-up in the included studies, which was 61 weeks. Bone damage may occur over longer time periods. Bone fractures were also not a prespecified adverse event in most included studies.
The studies also did not provide detailed information on the types of fractures experienced, such as whether they were result of a fall, or the location of the fracture, or bone health parameters. Although the results support a belief that SGLT-2 inhibitors do not adversely affect bone health, “given limited information on bone health outcomes, further work is needed to validate this conclusion,” the authors wrote.
The authors did not disclose any funding and had no relevant conflicts of interest.
SOURCE: B-B Qian et al. Osteoporosis Int. 2020 Aug 11. doi: 10.1007/s00198-020-05590-y.
The combination of sodium-glucose transporter-2 (SGLT-2) inhibitors and metformin is not associated with an increase in fracture risk among patients with type 2 diabetes (T2D), according to a new meta-analysis of 25 randomized, controlled trials.
Researchers at The Second Clinical College of Dalian Medical University in Jiangsu, China, compared fracture risk associated with the metformin/SLGT2 combination to metformin alone as well as other T2D therapeutics, and found no differences in risk. The study was published online Aug. 11 in Osteoporosis International.
T2D is associated with an increased risk of fracture, though causative mechanisms remain uncertain. Some lines of evidence suggest multiple factors may contribute to fractures, including hyperglycemia, oxidative stress, toxic effects of advanced glycosylation end-products, altered insulin levels, and treatment-induced hypoglycemia, as well as an association between T2D and increased risk of falls.
Antidiabetes drugs can have positive or negative effects on bone. thiazolidinediones, insulin, and sulfonylureas may increase risk of fractures, while dipeptidyl peptidase-4 (DPP-4) inhibitors and glucagon-like peptide-2 (GLP-2) receptor agonists may be protective. Metformin may also reduce fracture risk.
SGLT-2 inhibitors interrupt glucose reabsorption in the kidney, leading to improved glycemic control. Other benefits include improved renal and cardiovascular outcomes, weight loss, and reduced blood pressure, liver fat, and serum uric acid levels.
These properties have made SGLT-2 inhibitors combined with metformin an important therapy for patients at high risk of atherosclerotic disease, or who have heart failure or chronic kidney disease.
But SGLT-2 inhibition increases osmotic diuresis, and this could alter the mineral balance within bone. Some studies also showed that SGLT-2 inhibitors led to changes in bone turnover markers, bone mineral density, and bone microarchitecture. Observational studies of the SGLT-2 inhibitor canagliflozin found associations with a higher rate of fracture risk in patients taking the drug.
Such studies carry the risk of confounding factors, so the researchers took advantage of the fact that many recent clinical trials have examined the impact of SGLT-2 inhibitors on T2D. They pooled data from 25 clinical trials with a total of 19,500 participants, 9,662 of whom received SGLT-2 inhibitors plus metformin; 9,838 received other active comparators.
The fracture rate was 0.91% in the SGLT-2 inhibitors/metformin group, and 0.80% among controls (odds ratio, 0.97; 95% CI, 0.71-1.32), with no heterogeneity. Metformin alone was not associated with a change in fracture rate (OR, 0.95; 95% CI, 0.44-2.08), nor were other forms of diabetes control (OR, 0.95; 95% CI, 0.69-1.31).
There were some differences in fracture risk among SGLT-2 inhibitors when studied individually, though none differed significantly from controls. The highest risk was associated with the canagliflozin/metformin (OR, 2.19; 95% CI, 0.66-7.27), followed by dapagliflozin/metformin (OR, 0.91; 95% CI, 0.50-1.64), empagliflozin/metformin (OR, 0.94; 95% CI, 0.59-1.50), and ertugliflozin/metformin (OR, 0.76; 95% CI, 0.38-1.54).
There were no differences with respect to hip or lumbar spine fractures, or other fractures. The researchers found no differences in bone mineral density or bone turnover markers.
The meta-analysis is limited by the relatively short average follow-up in the included studies, which was 61 weeks. Bone damage may occur over longer time periods. Bone fractures were also not a prespecified adverse event in most included studies.
The studies also did not provide detailed information on the types of fractures experienced, such as whether they were result of a fall, or the location of the fracture, or bone health parameters. Although the results support a belief that SGLT-2 inhibitors do not adversely affect bone health, “given limited information on bone health outcomes, further work is needed to validate this conclusion,” the authors wrote.
The authors did not disclose any funding and had no relevant conflicts of interest.
SOURCE: B-B Qian et al. Osteoporosis Int. 2020 Aug 11. doi: 10.1007/s00198-020-05590-y.
The combination of sodium-glucose transporter-2 (SGLT-2) inhibitors and metformin is not associated with an increase in fracture risk among patients with type 2 diabetes (T2D), according to a new meta-analysis of 25 randomized, controlled trials.
Researchers at The Second Clinical College of Dalian Medical University in Jiangsu, China, compared fracture risk associated with the metformin/SLGT2 combination to metformin alone as well as other T2D therapeutics, and found no differences in risk. The study was published online Aug. 11 in Osteoporosis International.
T2D is associated with an increased risk of fracture, though causative mechanisms remain uncertain. Some lines of evidence suggest multiple factors may contribute to fractures, including hyperglycemia, oxidative stress, toxic effects of advanced glycosylation end-products, altered insulin levels, and treatment-induced hypoglycemia, as well as an association between T2D and increased risk of falls.
Antidiabetes drugs can have positive or negative effects on bone. thiazolidinediones, insulin, and sulfonylureas may increase risk of fractures, while dipeptidyl peptidase-4 (DPP-4) inhibitors and glucagon-like peptide-2 (GLP-2) receptor agonists may be protective. Metformin may also reduce fracture risk.
SGLT-2 inhibitors interrupt glucose reabsorption in the kidney, leading to improved glycemic control. Other benefits include improved renal and cardiovascular outcomes, weight loss, and reduced blood pressure, liver fat, and serum uric acid levels.
These properties have made SGLT-2 inhibitors combined with metformin an important therapy for patients at high risk of atherosclerotic disease, or who have heart failure or chronic kidney disease.
But SGLT-2 inhibition increases osmotic diuresis, and this could alter the mineral balance within bone. Some studies also showed that SGLT-2 inhibitors led to changes in bone turnover markers, bone mineral density, and bone microarchitecture. Observational studies of the SGLT-2 inhibitor canagliflozin found associations with a higher rate of fracture risk in patients taking the drug.
Such studies carry the risk of confounding factors, so the researchers took advantage of the fact that many recent clinical trials have examined the impact of SGLT-2 inhibitors on T2D. They pooled data from 25 clinical trials with a total of 19,500 participants, 9,662 of whom received SGLT-2 inhibitors plus metformin; 9,838 received other active comparators.
The fracture rate was 0.91% in the SGLT-2 inhibitors/metformin group, and 0.80% among controls (odds ratio, 0.97; 95% CI, 0.71-1.32), with no heterogeneity. Metformin alone was not associated with a change in fracture rate (OR, 0.95; 95% CI, 0.44-2.08), nor were other forms of diabetes control (OR, 0.95; 95% CI, 0.69-1.31).
There were some differences in fracture risk among SGLT-2 inhibitors when studied individually, though none differed significantly from controls. The highest risk was associated with the canagliflozin/metformin (OR, 2.19; 95% CI, 0.66-7.27), followed by dapagliflozin/metformin (OR, 0.91; 95% CI, 0.50-1.64), empagliflozin/metformin (OR, 0.94; 95% CI, 0.59-1.50), and ertugliflozin/metformin (OR, 0.76; 95% CI, 0.38-1.54).
There were no differences with respect to hip or lumbar spine fractures, or other fractures. The researchers found no differences in bone mineral density or bone turnover markers.
The meta-analysis is limited by the relatively short average follow-up in the included studies, which was 61 weeks. Bone damage may occur over longer time periods. Bone fractures were also not a prespecified adverse event in most included studies.
The studies also did not provide detailed information on the types of fractures experienced, such as whether they were result of a fall, or the location of the fracture, or bone health parameters. Although the results support a belief that SGLT-2 inhibitors do not adversely affect bone health, “given limited information on bone health outcomes, further work is needed to validate this conclusion,” the authors wrote.
The authors did not disclose any funding and had no relevant conflicts of interest.
SOURCE: B-B Qian et al. Osteoporosis Int. 2020 Aug 11. doi: 10.1007/s00198-020-05590-y.
FROM OSTEOPOROSIS INTERNATIONAL
Fracture risk prediction: No benefit to repeat BMD testing in postmenopausal women
On the basis of the findings, published online in JAMA Internal Medicine, the authors recommend against routine repeat testing in postmenopausal women. Other experts, however, caution that the results may not be so broadly generalizable.
For the investigation, Carolyn J. Crandall, MD, of the division of general internal medicine and health services research at the University of California, Los Angeles, and colleagues analyzed data from 7,419 women enrolled in the prospective Women’s Health Initiative study and who underwent baseline and repeat dual-energy x-ray absorptiometry (DXA) between 1993 and 2010. The researchers excluded patients who reported using bisphosphonates, calcitonin, or selective estrogen-receptor modulators, those with a history of major osteoporotic fracture, or those who lacked follow-up visits. The mean body mass index (BMI) of the study population was 28.7 kg/m2, and the mean age was 66.1 years.
The mean follow-up after the repeat BMD test was 9.0 years, during which period 732 (9.9%) of the women experienced a major osteoporotic fracture, and 139 (1.9%) experienced hip fractures.
To determine whether repeat testing improved fracture risk discrimination, the researchers calculated area under the receiver operating characteristic curve (AUROC) for baseline BMD, absolute change in BMD, and the combination of baseline BMD and change in BMD.
With respect to any major osteoporotic fracture risk, the AUROC values for total hip BMD at baseline, change in total hip BMD at 3 years, and the combination of the two, respectively, were 0.61 (95% confidence interval, 0.59-0.63), 0.53 (95% CI, 0.51-0.55), and 0.61 (95% CI, 0.59-0.63). For hip fracture risk, the respective AUROC values were 0.71 (95% CI, 0.67-0.75), 0.61 (95% CI, 0.56-0.65), and 0.73 (95% CI, 0.69-0.77), the authors reported.
Similar results were observed for femoral neck and lumbar spine BMD measurements. The associations between BMD changes and fracture risk were consistent across age, race, ethnicity, BMI, and baseline BMD T-score subgroups.
Although baseline BMD and change in BMD were independently associated with incident fracture, the association was stronger for lower baseline BMD than the 3-year absolute change in BMD, the authors stated.
The findings, which are consistent with those of previous investigations that involved older adults, are notable because of the age range of the population, according to the authors. “To our knowledge, this is the first prospective study that addressed this issue in a study cohort that included younger postmenopausal U.S. women,” they wrote. “Forty-four percent of our study population was younger than 65 years.”
The authors wrote that, given the lack of benefit associated with repeat BMD testing, such tests should no longer be routinely performed. “Our findings further suggest that resources should be devoted to increasing the underuse of baseline BMD testing among women aged [between] 65 and 85 years, one-quarter of whom do not receive an initial BMD test.”
However, some experts are not comfortable with the broad recommendation to skip repeat testing in the general population. “This is a great study, and it gives important information. However, we know, even in the real world, that patients can lose BMD in this time frame and not really fracture. This does not mean that they will not fracture further down the road,” said Pauline Camacho, MD, director of Loyola University Medical Center’s Osteoporosis and Metabolic Bone Disease Center in Chicago,. “The value of doing BMD goes beyond predicting fracture risk. It also helps assess patient compliance and detect the presence of uncorrected secondary causes of osteoporosis that are limiting the response to therapy, including failure to absorb oral bisphosphonates, vitamin D deficiency, or hyperparathyroidism.”
In addition, patients for whom treatment is initiated would want to know whether it’s working. “Seeing the BMD response to therapy is helpful to both clinicians and patients,” Dr. Camacho said in an interview.
Another concern is the study population. “The study was designed to assess the clinical utility of repeating a screening BMD test in a population of low-risk women -- older postmenopausal women with remarkably good BMD on initial testing,” according to E. Michael Lewiecki, MD, vice president of the National Osteoporosis Foundation and director of the New Mexico Clinical Research and Osteoporosis Center in Albuquerque. “Not surprisingly, with what we know about the expected age-related rate of bone loss, there was only a modest decrease in BMD and little clinical utility in repeating DXA in 3 years. However, repeat testing is an important component in the care of many patients seen in clinical practice.”
There are numerous situations in clinical practice in which repeat BMD testing can enhance patient care and potentially improve outcomes, Dr. Lewiecki said in an interview. “Repeating BMD 1-2 years after starting osteoporosis therapy is a useful way to assess response and determine whether the patient is on a pathway to achieving an acceptable level of fracture risk with a strategy called treat to target.”
Additionally, patients starting high-dose glucocorticoids who are at high risk for rapid bone loss may benefit from undergoing baseline BMD testing and having a follow-up test 1 year later or even sooner, he said. Further, for early postmenopausal women, the rate of bone loss may be accelerated and may be faster than age-related bone loss later in life. For this reason, “close monitoring of BMD may be used to determine when a treatment threshold has been crossed and pharmacological therapy is indicated.”
The most important message from this study for clinicians and healthcare policymakers is not the relative value of the repeat BMD testing, Dr. Lewiecki stated. Rather, it is the call to action regarding the underuse of BMD testing. “There is a global crisis in the care of osteoporosis that is characterized by underdiagnosis and undertreatment of patients at risk for fracture. Many patients who could benefit from treatment to reduce fracture risk are not receiving it, resulting in disability and deaths from fractures that might have been prevented. We need more bone density testing in appropriately selected patients to identify high-risk patients and intervene to reduce fracture risk,” he said. “DXA is an inexpensive and highly versatile clinical tool with many applications in clinical practice. When used wisely, it can be extraordinarily useful to identify and monitor high-risk patients, with the goal of reducing the burden of osteoporotic fractures.”
The barriers to performing baseline BMD measurement in this population are poorly understood and not well researched, Dr. Crandall said in an interview. “I expect that they relate to the multiple competing demands on primary care physicians, who are, for example, trying to juggle hypertension, a sprained ankle, diabetes, and complex social situations simultaneously with identifying appropriate candidates for osteoporosis screening and considering numerous other screening guidelines.”
The Women’s Health Initiative is funded by the National Heart, Lung, and Blood Institute; National Institutes of Health; and the Department of Health & Human Services. The study authors reported relationships with multiple companies, including Amgen, Pfizer, Bayer, Mithra, Norton Rose Fulbright, TherapeuticsMD, AbbVie, Radius, and Allergan. Dr. Camacho reported relationships with Amgen and Shire. Dr. Lewiecki reported relationships with Amgen, Radius Health, Alexion, Samsung Bioepis, Sandoz, Mereo, and Bindex.
A version of this article originally appeared on Medscape.com.
On the basis of the findings, published online in JAMA Internal Medicine, the authors recommend against routine repeat testing in postmenopausal women. Other experts, however, caution that the results may not be so broadly generalizable.
For the investigation, Carolyn J. Crandall, MD, of the division of general internal medicine and health services research at the University of California, Los Angeles, and colleagues analyzed data from 7,419 women enrolled in the prospective Women’s Health Initiative study and who underwent baseline and repeat dual-energy x-ray absorptiometry (DXA) between 1993 and 2010. The researchers excluded patients who reported using bisphosphonates, calcitonin, or selective estrogen-receptor modulators, those with a history of major osteoporotic fracture, or those who lacked follow-up visits. The mean body mass index (BMI) of the study population was 28.7 kg/m2, and the mean age was 66.1 years.
The mean follow-up after the repeat BMD test was 9.0 years, during which period 732 (9.9%) of the women experienced a major osteoporotic fracture, and 139 (1.9%) experienced hip fractures.
To determine whether repeat testing improved fracture risk discrimination, the researchers calculated area under the receiver operating characteristic curve (AUROC) for baseline BMD, absolute change in BMD, and the combination of baseline BMD and change in BMD.
With respect to any major osteoporotic fracture risk, the AUROC values for total hip BMD at baseline, change in total hip BMD at 3 years, and the combination of the two, respectively, were 0.61 (95% confidence interval, 0.59-0.63), 0.53 (95% CI, 0.51-0.55), and 0.61 (95% CI, 0.59-0.63). For hip fracture risk, the respective AUROC values were 0.71 (95% CI, 0.67-0.75), 0.61 (95% CI, 0.56-0.65), and 0.73 (95% CI, 0.69-0.77), the authors reported.
Similar results were observed for femoral neck and lumbar spine BMD measurements. The associations between BMD changes and fracture risk were consistent across age, race, ethnicity, BMI, and baseline BMD T-score subgroups.
Although baseline BMD and change in BMD were independently associated with incident fracture, the association was stronger for lower baseline BMD than the 3-year absolute change in BMD, the authors stated.
The findings, which are consistent with those of previous investigations that involved older adults, are notable because of the age range of the population, according to the authors. “To our knowledge, this is the first prospective study that addressed this issue in a study cohort that included younger postmenopausal U.S. women,” they wrote. “Forty-four percent of our study population was younger than 65 years.”
The authors wrote that, given the lack of benefit associated with repeat BMD testing, such tests should no longer be routinely performed. “Our findings further suggest that resources should be devoted to increasing the underuse of baseline BMD testing among women aged [between] 65 and 85 years, one-quarter of whom do not receive an initial BMD test.”
However, some experts are not comfortable with the broad recommendation to skip repeat testing in the general population. “This is a great study, and it gives important information. However, we know, even in the real world, that patients can lose BMD in this time frame and not really fracture. This does not mean that they will not fracture further down the road,” said Pauline Camacho, MD, director of Loyola University Medical Center’s Osteoporosis and Metabolic Bone Disease Center in Chicago,. “The value of doing BMD goes beyond predicting fracture risk. It also helps assess patient compliance and detect the presence of uncorrected secondary causes of osteoporosis that are limiting the response to therapy, including failure to absorb oral bisphosphonates, vitamin D deficiency, or hyperparathyroidism.”
In addition, patients for whom treatment is initiated would want to know whether it’s working. “Seeing the BMD response to therapy is helpful to both clinicians and patients,” Dr. Camacho said in an interview.
Another concern is the study population. “The study was designed to assess the clinical utility of repeating a screening BMD test in a population of low-risk women -- older postmenopausal women with remarkably good BMD on initial testing,” according to E. Michael Lewiecki, MD, vice president of the National Osteoporosis Foundation and director of the New Mexico Clinical Research and Osteoporosis Center in Albuquerque. “Not surprisingly, with what we know about the expected age-related rate of bone loss, there was only a modest decrease in BMD and little clinical utility in repeating DXA in 3 years. However, repeat testing is an important component in the care of many patients seen in clinical practice.”
There are numerous situations in clinical practice in which repeat BMD testing can enhance patient care and potentially improve outcomes, Dr. Lewiecki said in an interview. “Repeating BMD 1-2 years after starting osteoporosis therapy is a useful way to assess response and determine whether the patient is on a pathway to achieving an acceptable level of fracture risk with a strategy called treat to target.”
Additionally, patients starting high-dose glucocorticoids who are at high risk for rapid bone loss may benefit from undergoing baseline BMD testing and having a follow-up test 1 year later or even sooner, he said. Further, for early postmenopausal women, the rate of bone loss may be accelerated and may be faster than age-related bone loss later in life. For this reason, “close monitoring of BMD may be used to determine when a treatment threshold has been crossed and pharmacological therapy is indicated.”
The most important message from this study for clinicians and healthcare policymakers is not the relative value of the repeat BMD testing, Dr. Lewiecki stated. Rather, it is the call to action regarding the underuse of BMD testing. “There is a global crisis in the care of osteoporosis that is characterized by underdiagnosis and undertreatment of patients at risk for fracture. Many patients who could benefit from treatment to reduce fracture risk are not receiving it, resulting in disability and deaths from fractures that might have been prevented. We need more bone density testing in appropriately selected patients to identify high-risk patients and intervene to reduce fracture risk,” he said. “DXA is an inexpensive and highly versatile clinical tool with many applications in clinical practice. When used wisely, it can be extraordinarily useful to identify and monitor high-risk patients, with the goal of reducing the burden of osteoporotic fractures.”
The barriers to performing baseline BMD measurement in this population are poorly understood and not well researched, Dr. Crandall said in an interview. “I expect that they relate to the multiple competing demands on primary care physicians, who are, for example, trying to juggle hypertension, a sprained ankle, diabetes, and complex social situations simultaneously with identifying appropriate candidates for osteoporosis screening and considering numerous other screening guidelines.”
The Women’s Health Initiative is funded by the National Heart, Lung, and Blood Institute; National Institutes of Health; and the Department of Health & Human Services. The study authors reported relationships with multiple companies, including Amgen, Pfizer, Bayer, Mithra, Norton Rose Fulbright, TherapeuticsMD, AbbVie, Radius, and Allergan. Dr. Camacho reported relationships with Amgen and Shire. Dr. Lewiecki reported relationships with Amgen, Radius Health, Alexion, Samsung Bioepis, Sandoz, Mereo, and Bindex.
A version of this article originally appeared on Medscape.com.
On the basis of the findings, published online in JAMA Internal Medicine, the authors recommend against routine repeat testing in postmenopausal women. Other experts, however, caution that the results may not be so broadly generalizable.
For the investigation, Carolyn J. Crandall, MD, of the division of general internal medicine and health services research at the University of California, Los Angeles, and colleagues analyzed data from 7,419 women enrolled in the prospective Women’s Health Initiative study and who underwent baseline and repeat dual-energy x-ray absorptiometry (DXA) between 1993 and 2010. The researchers excluded patients who reported using bisphosphonates, calcitonin, or selective estrogen-receptor modulators, those with a history of major osteoporotic fracture, or those who lacked follow-up visits. The mean body mass index (BMI) of the study population was 28.7 kg/m2, and the mean age was 66.1 years.
The mean follow-up after the repeat BMD test was 9.0 years, during which period 732 (9.9%) of the women experienced a major osteoporotic fracture, and 139 (1.9%) experienced hip fractures.
To determine whether repeat testing improved fracture risk discrimination, the researchers calculated area under the receiver operating characteristic curve (AUROC) for baseline BMD, absolute change in BMD, and the combination of baseline BMD and change in BMD.
With respect to any major osteoporotic fracture risk, the AUROC values for total hip BMD at baseline, change in total hip BMD at 3 years, and the combination of the two, respectively, were 0.61 (95% confidence interval, 0.59-0.63), 0.53 (95% CI, 0.51-0.55), and 0.61 (95% CI, 0.59-0.63). For hip fracture risk, the respective AUROC values were 0.71 (95% CI, 0.67-0.75), 0.61 (95% CI, 0.56-0.65), and 0.73 (95% CI, 0.69-0.77), the authors reported.
Similar results were observed for femoral neck and lumbar spine BMD measurements. The associations between BMD changes and fracture risk were consistent across age, race, ethnicity, BMI, and baseline BMD T-score subgroups.
Although baseline BMD and change in BMD were independently associated with incident fracture, the association was stronger for lower baseline BMD than the 3-year absolute change in BMD, the authors stated.
The findings, which are consistent with those of previous investigations that involved older adults, are notable because of the age range of the population, according to the authors. “To our knowledge, this is the first prospective study that addressed this issue in a study cohort that included younger postmenopausal U.S. women,” they wrote. “Forty-four percent of our study population was younger than 65 years.”
The authors wrote that, given the lack of benefit associated with repeat BMD testing, such tests should no longer be routinely performed. “Our findings further suggest that resources should be devoted to increasing the underuse of baseline BMD testing among women aged [between] 65 and 85 years, one-quarter of whom do not receive an initial BMD test.”
However, some experts are not comfortable with the broad recommendation to skip repeat testing in the general population. “This is a great study, and it gives important information. However, we know, even in the real world, that patients can lose BMD in this time frame and not really fracture. This does not mean that they will not fracture further down the road,” said Pauline Camacho, MD, director of Loyola University Medical Center’s Osteoporosis and Metabolic Bone Disease Center in Chicago,. “The value of doing BMD goes beyond predicting fracture risk. It also helps assess patient compliance and detect the presence of uncorrected secondary causes of osteoporosis that are limiting the response to therapy, including failure to absorb oral bisphosphonates, vitamin D deficiency, or hyperparathyroidism.”
In addition, patients for whom treatment is initiated would want to know whether it’s working. “Seeing the BMD response to therapy is helpful to both clinicians and patients,” Dr. Camacho said in an interview.
Another concern is the study population. “The study was designed to assess the clinical utility of repeating a screening BMD test in a population of low-risk women -- older postmenopausal women with remarkably good BMD on initial testing,” according to E. Michael Lewiecki, MD, vice president of the National Osteoporosis Foundation and director of the New Mexico Clinical Research and Osteoporosis Center in Albuquerque. “Not surprisingly, with what we know about the expected age-related rate of bone loss, there was only a modest decrease in BMD and little clinical utility in repeating DXA in 3 years. However, repeat testing is an important component in the care of many patients seen in clinical practice.”
There are numerous situations in clinical practice in which repeat BMD testing can enhance patient care and potentially improve outcomes, Dr. Lewiecki said in an interview. “Repeating BMD 1-2 years after starting osteoporosis therapy is a useful way to assess response and determine whether the patient is on a pathway to achieving an acceptable level of fracture risk with a strategy called treat to target.”
Additionally, patients starting high-dose glucocorticoids who are at high risk for rapid bone loss may benefit from undergoing baseline BMD testing and having a follow-up test 1 year later or even sooner, he said. Further, for early postmenopausal women, the rate of bone loss may be accelerated and may be faster than age-related bone loss later in life. For this reason, “close monitoring of BMD may be used to determine when a treatment threshold has been crossed and pharmacological therapy is indicated.”
The most important message from this study for clinicians and healthcare policymakers is not the relative value of the repeat BMD testing, Dr. Lewiecki stated. Rather, it is the call to action regarding the underuse of BMD testing. “There is a global crisis in the care of osteoporosis that is characterized by underdiagnosis and undertreatment of patients at risk for fracture. Many patients who could benefit from treatment to reduce fracture risk are not receiving it, resulting in disability and deaths from fractures that might have been prevented. We need more bone density testing in appropriately selected patients to identify high-risk patients and intervene to reduce fracture risk,” he said. “DXA is an inexpensive and highly versatile clinical tool with many applications in clinical practice. When used wisely, it can be extraordinarily useful to identify and monitor high-risk patients, with the goal of reducing the burden of osteoporotic fractures.”
The barriers to performing baseline BMD measurement in this population are poorly understood and not well researched, Dr. Crandall said in an interview. “I expect that they relate to the multiple competing demands on primary care physicians, who are, for example, trying to juggle hypertension, a sprained ankle, diabetes, and complex social situations simultaneously with identifying appropriate candidates for osteoporosis screening and considering numerous other screening guidelines.”
The Women’s Health Initiative is funded by the National Heart, Lung, and Blood Institute; National Institutes of Health; and the Department of Health & Human Services. The study authors reported relationships with multiple companies, including Amgen, Pfizer, Bayer, Mithra, Norton Rose Fulbright, TherapeuticsMD, AbbVie, Radius, and Allergan. Dr. Camacho reported relationships with Amgen and Shire. Dr. Lewiecki reported relationships with Amgen, Radius Health, Alexion, Samsung Bioepis, Sandoz, Mereo, and Bindex.
A version of this article originally appeared on Medscape.com.
Low vitamin D linked to increased COVID-19 risk
Low plasma vitamin D levels emerged as an independent risk factor for COVID-19 infection and hospitalization in a large, population-based study.
Participants positive for COVID-19 were 50% more likely to have low vs normal 25(OH)D levels in a multivariate analysis that controlled for other confounders, for example.
The take home message for physicians is to “test patients’ vitamin D levels and keep them optimal for the overall health – as well as for a better immunoresponse to COVID-19,” senior author Milana Frenkel-Morgenstern, PhD, head of the Cancer Genomics and BioComputing of Complex Diseases Lab at Bar-Ilan University in Ramat Gan, Israel, said in an interview.
The study was published online July 23 in The FEBS Journal.
Previous and ongoing studies are evaluating a potential role for vitamin D to prevent or minimize the severity of SARS-CoV-2 infection, building on years of research addressing vitamin D for other viral respiratory infections. The evidence to date regarding COVID-19, primarily observational studies, has yielded mixed results.
Multiple experts weighed in on the controversy in a previous report. Many point out the limitations of observational data, particularly when it comes to ruling out other factors that could affect the severity of COVID-19 infection. In addition, in a video report, JoAnn E. Manson, MD, DrPH, of Harvard Medical School in Boston, cited an observational study from three South Asian hospitals that found more severe COVID-19 patients had lower vitamin D levels, as well as other “compelling evidence” suggesting an association.
Dr. Frenkel-Morgenstern and colleagues studied data for 7,807 people, of whom 10.1% were COVID-19 positive. They assessed electronic health records for demographics, potential confounders, and outcomes between February 1 and April 30.
Participants positive for COVID-19 tended to be younger and were more likely to be men and live in a lower socioeconomic area, compared with the participants who were negative for COVID-19, in a univariate analysis.
Key findings
A higher proportion of COVID-19–positive patients had low plasma 25(OH)D concentrations, about 90% versus 85% of participants who were negative for COVID-19. The difference was statistically significant (P < .001). Furthermore, the increased likelihood for low vitamin D levels among those positive for COVID-19 held in a multivariate analysis that controlled for demographics and psychiatric and somatic disorders (adjusted odds ratio, 1.50). The difference remained statistically significant (P < .001).
The study also was noteworthy for what it did not find among participants with COVID-19. For example, the prevalence of dementia, cardiovascular disease, chronic lung disorders, and hypertension were significantly higher among the COVID-19 negative participants.
“Severe social contacts restrictions that were imposed on all the population and were even more emphasized in this highly vulnerable population” could explain these findings, the researchers noted.
“We assume that following the Israeli Ministry of Health instructions, patients with chronic medical conditions significantly reduced their social contacts” and thereby reduced their infection risk.
In contrast to previous reports, obesity was not a significant factor associated with increased likelihood for COVID-19 infection or hospitalization in the current study.
The researchers also linked low plasma 25(OH)D level to an increased likelihood of hospitalization for COVID-19 infection (crude OR, 2.09; P < .05).
After controlling for demographics and chronic disorders, the aOR decreased to 1.95 (P = .061) in a multivariate analysis. The only factor that remained statistically significant for hospitalization was age over 50 years (aOR, 2.71; P < .001).
Implications and future plans
The large number of participants and the “real world,” population-based design are strengths of the study. Considering potential confounders is another strength, the researchers noted. The retrospective database design was a limitation.
Going forward, Dr. Frenkel-Morgenstern and colleagues will “try to decipher the potential role of vitamin D in prevention and/or treatment of COVID-19” through three additional studies, she said. Also, they would like to conduct a meta-analysis to combine data from different countries to further explore the potential role of vitamin D in COVID-19.
“A compelling case”
“This is a strong study – large, adjusted for confounders, consistent with the biology and other clinical studies of vitamin D, infections, and COVID-19,” Wayne Jonas, MD, a practicing family physician and executive director of Samueli Integrative Health Programs, said in an interview.
Because the research was retrospective and observational, a causative link between vitamin D levels and COVID-19 risk cannot be interpreted from the findings. “That would need a prospective, randomized study,” said Dr. Jonas, who was not involved with the current study.
However, “the study makes a compelling case for possibly screening vitamin D levels for judging risk of COVID infection and hospitalization,” Dr. Jonas said, “and the compelling need for a large, randomized vitamin D supplement study to see if it can help prevent infection.”
“Given that vitamin D is largely safe, such a study could be done quickly and on healthy people with minimal risk for harm,” he added.
More confounders likely?
“I think the study is of interest,” Naveed Sattar, PhD, professor of metabolic medicine at the University of Glasgow, who also was not affiliated with the research, said in an interview.
“Whilst the authors adjusted for some confounders, there is a strong potential for residual confounding,” said Dr. Sattar, a coauthor of a UK Biobank study that did not find an association between vitamin D stages and COVID-19 infection in multivariate models.
For example, Dr. Sattar said, “Robust adjustment for social class is important since both Vitamin D levels and COVID-19 severity are both strongly associated with social class.” Further, it remains unknown when and what time of year the vitamin D concentrations were measured in the current study.
“In the end, only a robust randomized trial can tell us whether vitamin D supplementation helps lessen COVID-19 severity,” Dr. Sattar added. “I am not hopeful we will find this is the case – but I am glad some such trials are [ongoing].”
Dr. Frenkel-Morgenstern received a COVID-19 Data Sciences Institute grant to support this work. Dr. Frenkel-Morgenstern, Dr. Jonas, and Dr. Sattar have disclosed no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
Low plasma vitamin D levels emerged as an independent risk factor for COVID-19 infection and hospitalization in a large, population-based study.
Participants positive for COVID-19 were 50% more likely to have low vs normal 25(OH)D levels in a multivariate analysis that controlled for other confounders, for example.
The take home message for physicians is to “test patients’ vitamin D levels and keep them optimal for the overall health – as well as for a better immunoresponse to COVID-19,” senior author Milana Frenkel-Morgenstern, PhD, head of the Cancer Genomics and BioComputing of Complex Diseases Lab at Bar-Ilan University in Ramat Gan, Israel, said in an interview.
The study was published online July 23 in The FEBS Journal.
Previous and ongoing studies are evaluating a potential role for vitamin D to prevent or minimize the severity of SARS-CoV-2 infection, building on years of research addressing vitamin D for other viral respiratory infections. The evidence to date regarding COVID-19, primarily observational studies, has yielded mixed results.
Multiple experts weighed in on the controversy in a previous report. Many point out the limitations of observational data, particularly when it comes to ruling out other factors that could affect the severity of COVID-19 infection. In addition, in a video report, JoAnn E. Manson, MD, DrPH, of Harvard Medical School in Boston, cited an observational study from three South Asian hospitals that found more severe COVID-19 patients had lower vitamin D levels, as well as other “compelling evidence” suggesting an association.
Dr. Frenkel-Morgenstern and colleagues studied data for 7,807 people, of whom 10.1% were COVID-19 positive. They assessed electronic health records for demographics, potential confounders, and outcomes between February 1 and April 30.
Participants positive for COVID-19 tended to be younger and were more likely to be men and live in a lower socioeconomic area, compared with the participants who were negative for COVID-19, in a univariate analysis.
Key findings
A higher proportion of COVID-19–positive patients had low plasma 25(OH)D concentrations, about 90% versus 85% of participants who were negative for COVID-19. The difference was statistically significant (P < .001). Furthermore, the increased likelihood for low vitamin D levels among those positive for COVID-19 held in a multivariate analysis that controlled for demographics and psychiatric and somatic disorders (adjusted odds ratio, 1.50). The difference remained statistically significant (P < .001).
The study also was noteworthy for what it did not find among participants with COVID-19. For example, the prevalence of dementia, cardiovascular disease, chronic lung disorders, and hypertension were significantly higher among the COVID-19 negative participants.
“Severe social contacts restrictions that were imposed on all the population and were even more emphasized in this highly vulnerable population” could explain these findings, the researchers noted.
“We assume that following the Israeli Ministry of Health instructions, patients with chronic medical conditions significantly reduced their social contacts” and thereby reduced their infection risk.
In contrast to previous reports, obesity was not a significant factor associated with increased likelihood for COVID-19 infection or hospitalization in the current study.
The researchers also linked low plasma 25(OH)D level to an increased likelihood of hospitalization for COVID-19 infection (crude OR, 2.09; P < .05).
After controlling for demographics and chronic disorders, the aOR decreased to 1.95 (P = .061) in a multivariate analysis. The only factor that remained statistically significant for hospitalization was age over 50 years (aOR, 2.71; P < .001).
Implications and future plans
The large number of participants and the “real world,” population-based design are strengths of the study. Considering potential confounders is another strength, the researchers noted. The retrospective database design was a limitation.
Going forward, Dr. Frenkel-Morgenstern and colleagues will “try to decipher the potential role of vitamin D in prevention and/or treatment of COVID-19” through three additional studies, she said. Also, they would like to conduct a meta-analysis to combine data from different countries to further explore the potential role of vitamin D in COVID-19.
“A compelling case”
“This is a strong study – large, adjusted for confounders, consistent with the biology and other clinical studies of vitamin D, infections, and COVID-19,” Wayne Jonas, MD, a practicing family physician and executive director of Samueli Integrative Health Programs, said in an interview.
Because the research was retrospective and observational, a causative link between vitamin D levels and COVID-19 risk cannot be interpreted from the findings. “That would need a prospective, randomized study,” said Dr. Jonas, who was not involved with the current study.
However, “the study makes a compelling case for possibly screening vitamin D levels for judging risk of COVID infection and hospitalization,” Dr. Jonas said, “and the compelling need for a large, randomized vitamin D supplement study to see if it can help prevent infection.”
“Given that vitamin D is largely safe, such a study could be done quickly and on healthy people with minimal risk for harm,” he added.
More confounders likely?
“I think the study is of interest,” Naveed Sattar, PhD, professor of metabolic medicine at the University of Glasgow, who also was not affiliated with the research, said in an interview.
“Whilst the authors adjusted for some confounders, there is a strong potential for residual confounding,” said Dr. Sattar, a coauthor of a UK Biobank study that did not find an association between vitamin D stages and COVID-19 infection in multivariate models.
For example, Dr. Sattar said, “Robust adjustment for social class is important since both Vitamin D levels and COVID-19 severity are both strongly associated with social class.” Further, it remains unknown when and what time of year the vitamin D concentrations were measured in the current study.
“In the end, only a robust randomized trial can tell us whether vitamin D supplementation helps lessen COVID-19 severity,” Dr. Sattar added. “I am not hopeful we will find this is the case – but I am glad some such trials are [ongoing].”
Dr. Frenkel-Morgenstern received a COVID-19 Data Sciences Institute grant to support this work. Dr. Frenkel-Morgenstern, Dr. Jonas, and Dr. Sattar have disclosed no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
Low plasma vitamin D levels emerged as an independent risk factor for COVID-19 infection and hospitalization in a large, population-based study.
Participants positive for COVID-19 were 50% more likely to have low vs normal 25(OH)D levels in a multivariate analysis that controlled for other confounders, for example.
The take home message for physicians is to “test patients’ vitamin D levels and keep them optimal for the overall health – as well as for a better immunoresponse to COVID-19,” senior author Milana Frenkel-Morgenstern, PhD, head of the Cancer Genomics and BioComputing of Complex Diseases Lab at Bar-Ilan University in Ramat Gan, Israel, said in an interview.
The study was published online July 23 in The FEBS Journal.
Previous and ongoing studies are evaluating a potential role for vitamin D to prevent or minimize the severity of SARS-CoV-2 infection, building on years of research addressing vitamin D for other viral respiratory infections. The evidence to date regarding COVID-19, primarily observational studies, has yielded mixed results.
Multiple experts weighed in on the controversy in a previous report. Many point out the limitations of observational data, particularly when it comes to ruling out other factors that could affect the severity of COVID-19 infection. In addition, in a video report, JoAnn E. Manson, MD, DrPH, of Harvard Medical School in Boston, cited an observational study from three South Asian hospitals that found more severe COVID-19 patients had lower vitamin D levels, as well as other “compelling evidence” suggesting an association.
Dr. Frenkel-Morgenstern and colleagues studied data for 7,807 people, of whom 10.1% were COVID-19 positive. They assessed electronic health records for demographics, potential confounders, and outcomes between February 1 and April 30.
Participants positive for COVID-19 tended to be younger and were more likely to be men and live in a lower socioeconomic area, compared with the participants who were negative for COVID-19, in a univariate analysis.
Key findings
A higher proportion of COVID-19–positive patients had low plasma 25(OH)D concentrations, about 90% versus 85% of participants who were negative for COVID-19. The difference was statistically significant (P < .001). Furthermore, the increased likelihood for low vitamin D levels among those positive for COVID-19 held in a multivariate analysis that controlled for demographics and psychiatric and somatic disorders (adjusted odds ratio, 1.50). The difference remained statistically significant (P < .001).
The study also was noteworthy for what it did not find among participants with COVID-19. For example, the prevalence of dementia, cardiovascular disease, chronic lung disorders, and hypertension were significantly higher among the COVID-19 negative participants.
“Severe social contacts restrictions that were imposed on all the population and were even more emphasized in this highly vulnerable population” could explain these findings, the researchers noted.
“We assume that following the Israeli Ministry of Health instructions, patients with chronic medical conditions significantly reduced their social contacts” and thereby reduced their infection risk.
In contrast to previous reports, obesity was not a significant factor associated with increased likelihood for COVID-19 infection or hospitalization in the current study.
The researchers also linked low plasma 25(OH)D level to an increased likelihood of hospitalization for COVID-19 infection (crude OR, 2.09; P < .05).
After controlling for demographics and chronic disorders, the aOR decreased to 1.95 (P = .061) in a multivariate analysis. The only factor that remained statistically significant for hospitalization was age over 50 years (aOR, 2.71; P < .001).
Implications and future plans
The large number of participants and the “real world,” population-based design are strengths of the study. Considering potential confounders is another strength, the researchers noted. The retrospective database design was a limitation.
Going forward, Dr. Frenkel-Morgenstern and colleagues will “try to decipher the potential role of vitamin D in prevention and/or treatment of COVID-19” through three additional studies, she said. Also, they would like to conduct a meta-analysis to combine data from different countries to further explore the potential role of vitamin D in COVID-19.
“A compelling case”
“This is a strong study – large, adjusted for confounders, consistent with the biology and other clinical studies of vitamin D, infections, and COVID-19,” Wayne Jonas, MD, a practicing family physician and executive director of Samueli Integrative Health Programs, said in an interview.
Because the research was retrospective and observational, a causative link between vitamin D levels and COVID-19 risk cannot be interpreted from the findings. “That would need a prospective, randomized study,” said Dr. Jonas, who was not involved with the current study.
However, “the study makes a compelling case for possibly screening vitamin D levels for judging risk of COVID infection and hospitalization,” Dr. Jonas said, “and the compelling need for a large, randomized vitamin D supplement study to see if it can help prevent infection.”
“Given that vitamin D is largely safe, such a study could be done quickly and on healthy people with minimal risk for harm,” he added.
More confounders likely?
“I think the study is of interest,” Naveed Sattar, PhD, professor of metabolic medicine at the University of Glasgow, who also was not affiliated with the research, said in an interview.
“Whilst the authors adjusted for some confounders, there is a strong potential for residual confounding,” said Dr. Sattar, a coauthor of a UK Biobank study that did not find an association between vitamin D stages and COVID-19 infection in multivariate models.
For example, Dr. Sattar said, “Robust adjustment for social class is important since both Vitamin D levels and COVID-19 severity are both strongly associated with social class.” Further, it remains unknown when and what time of year the vitamin D concentrations were measured in the current study.
“In the end, only a robust randomized trial can tell us whether vitamin D supplementation helps lessen COVID-19 severity,” Dr. Sattar added. “I am not hopeful we will find this is the case – but I am glad some such trials are [ongoing].”
Dr. Frenkel-Morgenstern received a COVID-19 Data Sciences Institute grant to support this work. Dr. Frenkel-Morgenstern, Dr. Jonas, and Dr. Sattar have disclosed no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
Delaying denosumab dose boosts risk for vertebral fractures
a new study confirms. Physicians say they are especially concerned about the risk facing patients who are delaying the treatment during the coronavirus pandemic.
The recommended doses of denosumab are at 6-month intervals. Patients who delayed a dose by more than 16 weeks were nearly four times more likely to suffer vertebral fractures, compared with those who received on-time injections, according to the study, which was published in Annals of Internal Medicine.
“Because patients who used denosumab were at high risk for vertebral fracture, strategies to improve timely administration of denosumab in routine clinical settings are needed,” wrote the study authors, led by Houchen Lyu, MD, PhD, of National Clinical Research Center for Orthopedics, Sports Medicine & Rehabilitation at General Hospital of Chinese PLA in Beijing.
Denosumab, a human monoclonal antibody, is used to reduce bone loss in osteoporosis. The manufacturer of Prolia, a brand of the drug, recommends it be given every 6 months, but the study reports that it’s common for injections to be delayed.
Researchers have linked cessation of denosumab to higher risk of fractures, and Dr. Lyu led a study published earlier this year that linked less-frequent doses to less bone mineral density improvement. “However,” the authors of the new study wrote, “whether delaying subsequent injections beyond the recommended 6-month interval is associated with fractures is unknown.”
For their new study, researchers retrospectively analyzed data from 2,594 patients in the U.K. 45 years or older (mean age, 76; 94% female; 53% with a history of major osteoporotic fracture) who began taking denosumab between 2010 and 2019. They used a design that aimed to emulate a clinical trial, comparing three dosing intervals: “on time” (within 4 weeks of the recommended 6-month interval), “short delay” (within 4-16 weeks) and “long delay” (16 weeks to 6 months).
The study found that the risk of composite fracture over 6 months out of 1,000 was 27.3 for on-time dosing, 32.2 for short-delay dosing, and 42.4 for long-delay dosing. The hazard ratio for long-delay versus on-time was 1.44 (95% confidence interval, 0.96-2.17; P = .093).
Vertebral fractures were less likely, but delays boosted the risk significantly: Over 6 months, it grew from 2.2 in 1,000 (on time) to 3.6 in 1,000 (short delay) and 10.1 in 1,000 (long delay). The HR for long delay versus on time was 3.91 (95% CI, 1.62-9.45; P = .005).
“This study had limited statistical power for composite fracture and several secondary end points ... except for vertebral fracture. Thus, evidence was insufficient to conclude that fracture risk was increased at other anatomical sites.”
In an accompanying editorial, two physicians from the University of Minnesota, Minneapolis, noted that the study is “timely and relevant” since the coronavirus pandemic may disrupt dosage schedules more than usual. While the study has limitations, the “findings are consistent with known denosumab pharmacokinetics and prior studies of fracture incidence after denosumab treatment discontinuation, wrote Kristine E. Ensrud, MD, MPH, who is also of Minneapolis VA Health Care System, and John T. Schousboe, MD, PhD, who is also of HealthPartners Institute.
The editorial authors noted that, in light of the pandemic, “some organizations recommend temporary transition to an oral bisphosphonate in patients receiving denosumab treatment for whom continued treatment is not feasible within 7 to 8 months of their most recent injection.”
In an interview, endocrinologist and osteoporosis specialist Ethel Siris, MD, of Columbia University, New York, said many of her patients aren’t coming in for denosumab injections during the pandemic. “It’s hard enough to get people to show up every 6 months to get their shot when things are going nicely,” she said. “We’re talking older women who may be on a lot of other medications. People forget, and it’s difficult for the office to constantly remind some of them to get their shots at an infusion center.”
The lack of symptoms is another challenge to getting patients to return for doses, she said. “In osteoporosis, the only time something hurts is if you break it.”
Since the pandemic began, many patients have been avoiding medical offices because of fear of getting the coronavirus.
The new research is helpful because it shows that patients are “more likely to fracture if they delay,” Dr. Siris noted. The endocrinologist added that she has successfully convinced some patients to give themselves subcutaneous injections in the abdomen at home.
Dr. Siris said she has been able to watch patients do these injections on video to check their technique. Her patients have been impressed by “how easy it is and delighted to have accomplished it,” she said.
The study was funded by the National Institutes of Health China’s National Clinical Research Center for Orthopedics, Sports Medicine & Rehabilitation. The study authors, commentary authors, and Dr. Siris report no relevant disclosures.
SOURCE: Lyu H et al. Ann Intern Med. 2020 Jul 28. doi: 10.7326/M20-0882.
a new study confirms. Physicians say they are especially concerned about the risk facing patients who are delaying the treatment during the coronavirus pandemic.
The recommended doses of denosumab are at 6-month intervals. Patients who delayed a dose by more than 16 weeks were nearly four times more likely to suffer vertebral fractures, compared with those who received on-time injections, according to the study, which was published in Annals of Internal Medicine.
“Because patients who used denosumab were at high risk for vertebral fracture, strategies to improve timely administration of denosumab in routine clinical settings are needed,” wrote the study authors, led by Houchen Lyu, MD, PhD, of National Clinical Research Center for Orthopedics, Sports Medicine & Rehabilitation at General Hospital of Chinese PLA in Beijing.
Denosumab, a human monoclonal antibody, is used to reduce bone loss in osteoporosis. The manufacturer of Prolia, a brand of the drug, recommends it be given every 6 months, but the study reports that it’s common for injections to be delayed.
Researchers have linked cessation of denosumab to higher risk of fractures, and Dr. Lyu led a study published earlier this year that linked less-frequent doses to less bone mineral density improvement. “However,” the authors of the new study wrote, “whether delaying subsequent injections beyond the recommended 6-month interval is associated with fractures is unknown.”
For their new study, researchers retrospectively analyzed data from 2,594 patients in the U.K. 45 years or older (mean age, 76; 94% female; 53% with a history of major osteoporotic fracture) who began taking denosumab between 2010 and 2019. They used a design that aimed to emulate a clinical trial, comparing three dosing intervals: “on time” (within 4 weeks of the recommended 6-month interval), “short delay” (within 4-16 weeks) and “long delay” (16 weeks to 6 months).
The study found that the risk of composite fracture over 6 months out of 1,000 was 27.3 for on-time dosing, 32.2 for short-delay dosing, and 42.4 for long-delay dosing. The hazard ratio for long-delay versus on-time was 1.44 (95% confidence interval, 0.96-2.17; P = .093).
Vertebral fractures were less likely, but delays boosted the risk significantly: Over 6 months, it grew from 2.2 in 1,000 (on time) to 3.6 in 1,000 (short delay) and 10.1 in 1,000 (long delay). The HR for long delay versus on time was 3.91 (95% CI, 1.62-9.45; P = .005).
“This study had limited statistical power for composite fracture and several secondary end points ... except for vertebral fracture. Thus, evidence was insufficient to conclude that fracture risk was increased at other anatomical sites.”
In an accompanying editorial, two physicians from the University of Minnesota, Minneapolis, noted that the study is “timely and relevant” since the coronavirus pandemic may disrupt dosage schedules more than usual. While the study has limitations, the “findings are consistent with known denosumab pharmacokinetics and prior studies of fracture incidence after denosumab treatment discontinuation, wrote Kristine E. Ensrud, MD, MPH, who is also of Minneapolis VA Health Care System, and John T. Schousboe, MD, PhD, who is also of HealthPartners Institute.
The editorial authors noted that, in light of the pandemic, “some organizations recommend temporary transition to an oral bisphosphonate in patients receiving denosumab treatment for whom continued treatment is not feasible within 7 to 8 months of their most recent injection.”
In an interview, endocrinologist and osteoporosis specialist Ethel Siris, MD, of Columbia University, New York, said many of her patients aren’t coming in for denosumab injections during the pandemic. “It’s hard enough to get people to show up every 6 months to get their shot when things are going nicely,” she said. “We’re talking older women who may be on a lot of other medications. People forget, and it’s difficult for the office to constantly remind some of them to get their shots at an infusion center.”
The lack of symptoms is another challenge to getting patients to return for doses, she said. “In osteoporosis, the only time something hurts is if you break it.”
Since the pandemic began, many patients have been avoiding medical offices because of fear of getting the coronavirus.
The new research is helpful because it shows that patients are “more likely to fracture if they delay,” Dr. Siris noted. The endocrinologist added that she has successfully convinced some patients to give themselves subcutaneous injections in the abdomen at home.
Dr. Siris said she has been able to watch patients do these injections on video to check their technique. Her patients have been impressed by “how easy it is and delighted to have accomplished it,” she said.
The study was funded by the National Institutes of Health China’s National Clinical Research Center for Orthopedics, Sports Medicine & Rehabilitation. The study authors, commentary authors, and Dr. Siris report no relevant disclosures.
SOURCE: Lyu H et al. Ann Intern Med. 2020 Jul 28. doi: 10.7326/M20-0882.
a new study confirms. Physicians say they are especially concerned about the risk facing patients who are delaying the treatment during the coronavirus pandemic.
The recommended doses of denosumab are at 6-month intervals. Patients who delayed a dose by more than 16 weeks were nearly four times more likely to suffer vertebral fractures, compared with those who received on-time injections, according to the study, which was published in Annals of Internal Medicine.
“Because patients who used denosumab were at high risk for vertebral fracture, strategies to improve timely administration of denosumab in routine clinical settings are needed,” wrote the study authors, led by Houchen Lyu, MD, PhD, of National Clinical Research Center for Orthopedics, Sports Medicine & Rehabilitation at General Hospital of Chinese PLA in Beijing.
Denosumab, a human monoclonal antibody, is used to reduce bone loss in osteoporosis. The manufacturer of Prolia, a brand of the drug, recommends it be given every 6 months, but the study reports that it’s common for injections to be delayed.
Researchers have linked cessation of denosumab to higher risk of fractures, and Dr. Lyu led a study published earlier this year that linked less-frequent doses to less bone mineral density improvement. “However,” the authors of the new study wrote, “whether delaying subsequent injections beyond the recommended 6-month interval is associated with fractures is unknown.”
For their new study, researchers retrospectively analyzed data from 2,594 patients in the U.K. 45 years or older (mean age, 76; 94% female; 53% with a history of major osteoporotic fracture) who began taking denosumab between 2010 and 2019. They used a design that aimed to emulate a clinical trial, comparing three dosing intervals: “on time” (within 4 weeks of the recommended 6-month interval), “short delay” (within 4-16 weeks) and “long delay” (16 weeks to 6 months).
The study found that the risk of composite fracture over 6 months out of 1,000 was 27.3 for on-time dosing, 32.2 for short-delay dosing, and 42.4 for long-delay dosing. The hazard ratio for long-delay versus on-time was 1.44 (95% confidence interval, 0.96-2.17; P = .093).
Vertebral fractures were less likely, but delays boosted the risk significantly: Over 6 months, it grew from 2.2 in 1,000 (on time) to 3.6 in 1,000 (short delay) and 10.1 in 1,000 (long delay). The HR for long delay versus on time was 3.91 (95% CI, 1.62-9.45; P = .005).
“This study had limited statistical power for composite fracture and several secondary end points ... except for vertebral fracture. Thus, evidence was insufficient to conclude that fracture risk was increased at other anatomical sites.”
In an accompanying editorial, two physicians from the University of Minnesota, Minneapolis, noted that the study is “timely and relevant” since the coronavirus pandemic may disrupt dosage schedules more than usual. While the study has limitations, the “findings are consistent with known denosumab pharmacokinetics and prior studies of fracture incidence after denosumab treatment discontinuation, wrote Kristine E. Ensrud, MD, MPH, who is also of Minneapolis VA Health Care System, and John T. Schousboe, MD, PhD, who is also of HealthPartners Institute.
The editorial authors noted that, in light of the pandemic, “some organizations recommend temporary transition to an oral bisphosphonate in patients receiving denosumab treatment for whom continued treatment is not feasible within 7 to 8 months of their most recent injection.”
In an interview, endocrinologist and osteoporosis specialist Ethel Siris, MD, of Columbia University, New York, said many of her patients aren’t coming in for denosumab injections during the pandemic. “It’s hard enough to get people to show up every 6 months to get their shot when things are going nicely,” she said. “We’re talking older women who may be on a lot of other medications. People forget, and it’s difficult for the office to constantly remind some of them to get their shots at an infusion center.”
The lack of symptoms is another challenge to getting patients to return for doses, she said. “In osteoporosis, the only time something hurts is if you break it.”
Since the pandemic began, many patients have been avoiding medical offices because of fear of getting the coronavirus.
The new research is helpful because it shows that patients are “more likely to fracture if they delay,” Dr. Siris noted. The endocrinologist added that she has successfully convinced some patients to give themselves subcutaneous injections in the abdomen at home.
Dr. Siris said she has been able to watch patients do these injections on video to check their technique. Her patients have been impressed by “how easy it is and delighted to have accomplished it,” she said.
The study was funded by the National Institutes of Health China’s National Clinical Research Center for Orthopedics, Sports Medicine & Rehabilitation. The study authors, commentary authors, and Dr. Siris report no relevant disclosures.
SOURCE: Lyu H et al. Ann Intern Med. 2020 Jul 28. doi: 10.7326/M20-0882.
FROM ANNALS OF INTERNAL MEDICINE
Key clinical point: Patients with osteoporosis who delay denosumab doses are at much higher risk for vertebral fractures.
Major finding: Over 6 months, the risk of vertebral fractures grew from 2.2 in 1,000 (on-time doses) to 10.1 in 1,000 (delay of more than 16 weeks) – a hazard ratio of 3.91 (confidence interval, 1.62 to 9.45; P = .005).
Study details: Retrospective analysis of 2,594 patients in the U.K. 45 years or older who began taking denosumab between 2010 and 2019.
Disclosures: The study was funded by the National Institutes of Health China’s National Clinical Research Center for Orthopedics, Sports Medicine & Rehabilitation. The study authors report no relevant disclosures.
Source: Lyu H et al. Ann Intern Med. 2020 Jul 28. doi: 10.7326/M20-0882.
Bisphosphonates may have limited ‘protective’ effect against knee OA progression
New data from the National Institutes of Health–funded Osteoarthritis Initiative suggest that, in some women at least, taking bisphosphonates may help to reduce the chances that there will be radiographic progression of knee osteoarthritis (OA).
In a propensity-matched cohort analysis, women who had a Kellgren and Lawrence (KL) grade of less than 2 and who used bisphosphonates were half as likely as those who did not use bisphosphonates to have radiographic OA progression at 2 years (hazard ratio, 0.53; 95% confidence interval, 0.35-0.79). Radiographic OA progression has been defined as a one-step increase in the KL grade.
While the association appeared even stronger in women with a KL grade less than 2 and who were not overweight (HR, 0.49; 95% CI, 0.26-0.92), bisphosphonate use was not associated with radiographic OA progression in women with a higher (≥2) KL grade (HR, 1.06; 95% CI, 0.83-1.35).
“In all analyses, the effect of bisphosphonates was larger in radiographic-disease-naive individuals, suggesting protection using bisphosphonates may be more profound in those who do not already have evidence of knee damage or who have mild disease, and once damage occurs, bisphosphonate use may not have much effect,” Kaleen N. Hayes, PharmD, of the University of Toronto and her coauthors reported in the Journal of Bone and Mineral Research.
“Our study was the first to our knowledge to examine bisphosphonate exposure effects in different disease severity subgroups and obesity classifications using a rigorous, propensity-matched time-to-event analysis that uniquely addresses confounding by indication,” Dr. Hayes and her team wrote.
Furthermore, they noted that extensive sensitivity analyses, which included redoing the primary analyses to look at statin use, showed that their main conclusions were unchanged and that this helped account for any potential residual confounding, healthy-user bias, or exposure misclassification.
Study details
The Osteoarthritis Initiative is a 10-year longitudinal cohort study conducted at four clinical sites in the United States and recruited men and women aged 45-75 years over a 2-year period starting in 2004. Dr. Hayes and her coauthors restricted their analyses to women 50 years and older. Their study population consisted of 344 bisphosphonate users and 344 bisphosphonate nonusers.
The main bisphosphonate being taken was alendronate (69%), and the average duration of bisphosphonate use was 3.3 years, but no significant effect of duration of use on radiographic progression was found.
The women were followed until the first radiographic OA progression, or the first missed visit or end of the 2-year follow-up period.
Overall, 95 (13.8%) of the 688 women included in the analysis experienced radiographic OA progression. Of those, 27 (3.9%) had a KL grade of less than 2 and 68 (9.8%) had a KL grade of 2 or greater. Ten women with KL less than 2 and 27 women with KL or 2 or greater were taking bisphosphonates at their baseline visit.
“Kaplan-Meier analysis indicated that non-users and users with a baseline KL grade of 0 or 1 had 2-year risks of progression of 10.5% and 5.9%, respectively, whereas non-users and users with a baseline KL grade of 2 or 3 had 2-year of these women risks of progression of 23.0% and 23.5%, respectively,” reported the authors.
Before propensity score matching, Dr. Hayes and her colleagues observed that women taking bisphosphonates were older, had lower body weight and a higher prevalence of any fracture or hip and vertebral fractures, and were also more likely be White, compared with non-users. “In addition, bisphosphonate-users appeared to be healthier than non-users, as suggested by a lower smoking prevalence, lower average baseline KL grade, lower diabetes prevalence, and higher multivitamin use (a healthy-user proxy),” they acknowledged.
Results in perspective
“The key thing that I’m concerned about when I see something like bisphosphonates and osteoarthritis is just how well confounding has been addressed,” commented Tuhina Neogi, MD, PhD, professor of medicine and epidemiology at Boston University and chief of rheumatology at Boston Medical Center, in an interview.
“So are there factors other than the bisphosphonates themselves that might explain the findings? It looks like they’ve taken into account a lot of important things that one would consider for trying to get the two groups to look as similar as possible,” she added. Dr. Neogi queried, however, if body mass index had been suitably been adjusted for even after propensity score matching.
“The effect estimate is quite large, so I do think there is some confounding. So I would feel comfortable saying that there’s a signal here for bisphosphonates in reducing the risk of progression among those who do not have radiographic OA at baseline,” Dr. Neogi observed.
“The context of all this is that there have been large, well-designed, randomized control trials of oral bisphosphonates from years ago that did not find any benefit of bisphosphonates in [terms of] radiographic OA progression,” Dr. Neogi explained.
In the Knee OA Structural Arthritis (KOSTAR) study, now considered “quite a large landmark study,” the efficacy of risedronate in providing symptom relief and slowing disease progression was studied in almost 2,500 patients. “They saw some improvements in signs and symptoms, but risedronate did not significantly reduce radiographic progression. [However] there were some signals on biomarkers,” Dr. Neogi said.
One of the issues is that radiographs are too insensitive to pick up early bone changes in OA, a fact not missed by Dr. Hayes et al. More recent research has thus looked to using more sensitive imaging methods, such as CT and MRI, such as a recent study published in JAMA looking at the use of intravenous zoledronic acid on bone marrow lesions and cartilage volume. The results did not show any benefit of bisphosphonate use over 2 years.
“So even though we thought the MRI might provide a better way to detect a signal, it hasn’t panned out,” Dr. Neogi said.
But that’s not to say that there isn’t still a signal. Dr. Neogi’s most recent research has been using MRI to look at bone marrow lesion volume in women who were newly starting bisphosphonate therapy versus those who were not, and this has been just been accepted for publication.
“We found no difference in bone marrow lesion volume between the two groups. But in the women who had bone marrow lesions at baseline, there was a statistically significant greater proportion of women on bisphosphonates having a decrease in bone marrow lesion volume than the non-initiators,” she said.
So is there evidence that putting more women on bisphosphonates could prevent OA? “I’m not sure that you would be able to say that this should be something that all postmenopausal women should be on,” Dr. Neogi said.
“There’s a theoretical risk that has not been formally studied that, if you diminish bone turnover and you get more and more mineralization occurring, the bone potentially may have altered mechanical properties, become stiffer and, over the long term, that might not be good for OA.”
She added that, if there is already a clear clinical indication for bisphosphonate use, however, such as older women who have had a fracture and who should be on a bisphosphonate anyway, then “a bisphosphonate has the theoretical potential additional benefit for their osteoarthritis.”
The authors and Dr. Neogi had no conflicts of interest or relationships to disclose.
SOURCE: Hayes KN et al. J Bone Miner Res. 2020 July 14. doi: 10.1002/jbmr.4133.
New data from the National Institutes of Health–funded Osteoarthritis Initiative suggest that, in some women at least, taking bisphosphonates may help to reduce the chances that there will be radiographic progression of knee osteoarthritis (OA).
In a propensity-matched cohort analysis, women who had a Kellgren and Lawrence (KL) grade of less than 2 and who used bisphosphonates were half as likely as those who did not use bisphosphonates to have radiographic OA progression at 2 years (hazard ratio, 0.53; 95% confidence interval, 0.35-0.79). Radiographic OA progression has been defined as a one-step increase in the KL grade.
While the association appeared even stronger in women with a KL grade less than 2 and who were not overweight (HR, 0.49; 95% CI, 0.26-0.92), bisphosphonate use was not associated with radiographic OA progression in women with a higher (≥2) KL grade (HR, 1.06; 95% CI, 0.83-1.35).
“In all analyses, the effect of bisphosphonates was larger in radiographic-disease-naive individuals, suggesting protection using bisphosphonates may be more profound in those who do not already have evidence of knee damage or who have mild disease, and once damage occurs, bisphosphonate use may not have much effect,” Kaleen N. Hayes, PharmD, of the University of Toronto and her coauthors reported in the Journal of Bone and Mineral Research.
“Our study was the first to our knowledge to examine bisphosphonate exposure effects in different disease severity subgroups and obesity classifications using a rigorous, propensity-matched time-to-event analysis that uniquely addresses confounding by indication,” Dr. Hayes and her team wrote.
Furthermore, they noted that extensive sensitivity analyses, which included redoing the primary analyses to look at statin use, showed that their main conclusions were unchanged and that this helped account for any potential residual confounding, healthy-user bias, or exposure misclassification.
Study details
The Osteoarthritis Initiative is a 10-year longitudinal cohort study conducted at four clinical sites in the United States and recruited men and women aged 45-75 years over a 2-year period starting in 2004. Dr. Hayes and her coauthors restricted their analyses to women 50 years and older. Their study population consisted of 344 bisphosphonate users and 344 bisphosphonate nonusers.
The main bisphosphonate being taken was alendronate (69%), and the average duration of bisphosphonate use was 3.3 years, but no significant effect of duration of use on radiographic progression was found.
The women were followed until the first radiographic OA progression, or the first missed visit or end of the 2-year follow-up period.
Overall, 95 (13.8%) of the 688 women included in the analysis experienced radiographic OA progression. Of those, 27 (3.9%) had a KL grade of less than 2 and 68 (9.8%) had a KL grade of 2 or greater. Ten women with KL less than 2 and 27 women with KL or 2 or greater were taking bisphosphonates at their baseline visit.
“Kaplan-Meier analysis indicated that non-users and users with a baseline KL grade of 0 or 1 had 2-year risks of progression of 10.5% and 5.9%, respectively, whereas non-users and users with a baseline KL grade of 2 or 3 had 2-year of these women risks of progression of 23.0% and 23.5%, respectively,” reported the authors.
Before propensity score matching, Dr. Hayes and her colleagues observed that women taking bisphosphonates were older, had lower body weight and a higher prevalence of any fracture or hip and vertebral fractures, and were also more likely be White, compared with non-users. “In addition, bisphosphonate-users appeared to be healthier than non-users, as suggested by a lower smoking prevalence, lower average baseline KL grade, lower diabetes prevalence, and higher multivitamin use (a healthy-user proxy),” they acknowledged.
Results in perspective
“The key thing that I’m concerned about when I see something like bisphosphonates and osteoarthritis is just how well confounding has been addressed,” commented Tuhina Neogi, MD, PhD, professor of medicine and epidemiology at Boston University and chief of rheumatology at Boston Medical Center, in an interview.
“So are there factors other than the bisphosphonates themselves that might explain the findings? It looks like they’ve taken into account a lot of important things that one would consider for trying to get the two groups to look as similar as possible,” she added. Dr. Neogi queried, however, if body mass index had been suitably been adjusted for even after propensity score matching.
“The effect estimate is quite large, so I do think there is some confounding. So I would feel comfortable saying that there’s a signal here for bisphosphonates in reducing the risk of progression among those who do not have radiographic OA at baseline,” Dr. Neogi observed.
“The context of all this is that there have been large, well-designed, randomized control trials of oral bisphosphonates from years ago that did not find any benefit of bisphosphonates in [terms of] radiographic OA progression,” Dr. Neogi explained.
In the Knee OA Structural Arthritis (KOSTAR) study, now considered “quite a large landmark study,” the efficacy of risedronate in providing symptom relief and slowing disease progression was studied in almost 2,500 patients. “They saw some improvements in signs and symptoms, but risedronate did not significantly reduce radiographic progression. [However] there were some signals on biomarkers,” Dr. Neogi said.
One of the issues is that radiographs are too insensitive to pick up early bone changes in OA, a fact not missed by Dr. Hayes et al. More recent research has thus looked to using more sensitive imaging methods, such as CT and MRI, such as a recent study published in JAMA looking at the use of intravenous zoledronic acid on bone marrow lesions and cartilage volume. The results did not show any benefit of bisphosphonate use over 2 years.
“So even though we thought the MRI might provide a better way to detect a signal, it hasn’t panned out,” Dr. Neogi said.
But that’s not to say that there isn’t still a signal. Dr. Neogi’s most recent research has been using MRI to look at bone marrow lesion volume in women who were newly starting bisphosphonate therapy versus those who were not, and this has been just been accepted for publication.
“We found no difference in bone marrow lesion volume between the two groups. But in the women who had bone marrow lesions at baseline, there was a statistically significant greater proportion of women on bisphosphonates having a decrease in bone marrow lesion volume than the non-initiators,” she said.
So is there evidence that putting more women on bisphosphonates could prevent OA? “I’m not sure that you would be able to say that this should be something that all postmenopausal women should be on,” Dr. Neogi said.
“There’s a theoretical risk that has not been formally studied that, if you diminish bone turnover and you get more and more mineralization occurring, the bone potentially may have altered mechanical properties, become stiffer and, over the long term, that might not be good for OA.”
She added that, if there is already a clear clinical indication for bisphosphonate use, however, such as older women who have had a fracture and who should be on a bisphosphonate anyway, then “a bisphosphonate has the theoretical potential additional benefit for their osteoarthritis.”
The authors and Dr. Neogi had no conflicts of interest or relationships to disclose.
SOURCE: Hayes KN et al. J Bone Miner Res. 2020 July 14. doi: 10.1002/jbmr.4133.
New data from the National Institutes of Health–funded Osteoarthritis Initiative suggest that, in some women at least, taking bisphosphonates may help to reduce the chances that there will be radiographic progression of knee osteoarthritis (OA).
In a propensity-matched cohort analysis, women who had a Kellgren and Lawrence (KL) grade of less than 2 and who used bisphosphonates were half as likely as those who did not use bisphosphonates to have radiographic OA progression at 2 years (hazard ratio, 0.53; 95% confidence interval, 0.35-0.79). Radiographic OA progression has been defined as a one-step increase in the KL grade.
While the association appeared even stronger in women with a KL grade less than 2 and who were not overweight (HR, 0.49; 95% CI, 0.26-0.92), bisphosphonate use was not associated with radiographic OA progression in women with a higher (≥2) KL grade (HR, 1.06; 95% CI, 0.83-1.35).
“In all analyses, the effect of bisphosphonates was larger in radiographic-disease-naive individuals, suggesting protection using bisphosphonates may be more profound in those who do not already have evidence of knee damage or who have mild disease, and once damage occurs, bisphosphonate use may not have much effect,” Kaleen N. Hayes, PharmD, of the University of Toronto and her coauthors reported in the Journal of Bone and Mineral Research.
“Our study was the first to our knowledge to examine bisphosphonate exposure effects in different disease severity subgroups and obesity classifications using a rigorous, propensity-matched time-to-event analysis that uniquely addresses confounding by indication,” Dr. Hayes and her team wrote.
Furthermore, they noted that extensive sensitivity analyses, which included redoing the primary analyses to look at statin use, showed that their main conclusions were unchanged and that this helped account for any potential residual confounding, healthy-user bias, or exposure misclassification.
Study details
The Osteoarthritis Initiative is a 10-year longitudinal cohort study conducted at four clinical sites in the United States and recruited men and women aged 45-75 years over a 2-year period starting in 2004. Dr. Hayes and her coauthors restricted their analyses to women 50 years and older. Their study population consisted of 344 bisphosphonate users and 344 bisphosphonate nonusers.
The main bisphosphonate being taken was alendronate (69%), and the average duration of bisphosphonate use was 3.3 years, but no significant effect of duration of use on radiographic progression was found.
The women were followed until the first radiographic OA progression, or the first missed visit or end of the 2-year follow-up period.
Overall, 95 (13.8%) of the 688 women included in the analysis experienced radiographic OA progression. Of those, 27 (3.9%) had a KL grade of less than 2 and 68 (9.8%) had a KL grade of 2 or greater. Ten women with KL less than 2 and 27 women with KL or 2 or greater were taking bisphosphonates at their baseline visit.
“Kaplan-Meier analysis indicated that non-users and users with a baseline KL grade of 0 or 1 had 2-year risks of progression of 10.5% and 5.9%, respectively, whereas non-users and users with a baseline KL grade of 2 or 3 had 2-year of these women risks of progression of 23.0% and 23.5%, respectively,” reported the authors.
Before propensity score matching, Dr. Hayes and her colleagues observed that women taking bisphosphonates were older, had lower body weight and a higher prevalence of any fracture or hip and vertebral fractures, and were also more likely be White, compared with non-users. “In addition, bisphosphonate-users appeared to be healthier than non-users, as suggested by a lower smoking prevalence, lower average baseline KL grade, lower diabetes prevalence, and higher multivitamin use (a healthy-user proxy),” they acknowledged.
Results in perspective
“The key thing that I’m concerned about when I see something like bisphosphonates and osteoarthritis is just how well confounding has been addressed,” commented Tuhina Neogi, MD, PhD, professor of medicine and epidemiology at Boston University and chief of rheumatology at Boston Medical Center, in an interview.
“So are there factors other than the bisphosphonates themselves that might explain the findings? It looks like they’ve taken into account a lot of important things that one would consider for trying to get the two groups to look as similar as possible,” she added. Dr. Neogi queried, however, if body mass index had been suitably been adjusted for even after propensity score matching.
“The effect estimate is quite large, so I do think there is some confounding. So I would feel comfortable saying that there’s a signal here for bisphosphonates in reducing the risk of progression among those who do not have radiographic OA at baseline,” Dr. Neogi observed.
“The context of all this is that there have been large, well-designed, randomized control trials of oral bisphosphonates from years ago that did not find any benefit of bisphosphonates in [terms of] radiographic OA progression,” Dr. Neogi explained.
In the Knee OA Structural Arthritis (KOSTAR) study, now considered “quite a large landmark study,” the efficacy of risedronate in providing symptom relief and slowing disease progression was studied in almost 2,500 patients. “They saw some improvements in signs and symptoms, but risedronate did not significantly reduce radiographic progression. [However] there were some signals on biomarkers,” Dr. Neogi said.
One of the issues is that radiographs are too insensitive to pick up early bone changes in OA, a fact not missed by Dr. Hayes et al. More recent research has thus looked to using more sensitive imaging methods, such as CT and MRI, such as a recent study published in JAMA looking at the use of intravenous zoledronic acid on bone marrow lesions and cartilage volume. The results did not show any benefit of bisphosphonate use over 2 years.
“So even though we thought the MRI might provide a better way to detect a signal, it hasn’t panned out,” Dr. Neogi said.
But that’s not to say that there isn’t still a signal. Dr. Neogi’s most recent research has been using MRI to look at bone marrow lesion volume in women who were newly starting bisphosphonate therapy versus those who were not, and this has been just been accepted for publication.
“We found no difference in bone marrow lesion volume between the two groups. But in the women who had bone marrow lesions at baseline, there was a statistically significant greater proportion of women on bisphosphonates having a decrease in bone marrow lesion volume than the non-initiators,” she said.
So is there evidence that putting more women on bisphosphonates could prevent OA? “I’m not sure that you would be able to say that this should be something that all postmenopausal women should be on,” Dr. Neogi said.
“There’s a theoretical risk that has not been formally studied that, if you diminish bone turnover and you get more and more mineralization occurring, the bone potentially may have altered mechanical properties, become stiffer and, over the long term, that might not be good for OA.”
She added that, if there is already a clear clinical indication for bisphosphonate use, however, such as older women who have had a fracture and who should be on a bisphosphonate anyway, then “a bisphosphonate has the theoretical potential additional benefit for their osteoarthritis.”
The authors and Dr. Neogi had no conflicts of interest or relationships to disclose.
SOURCE: Hayes KN et al. J Bone Miner Res. 2020 July 14. doi: 10.1002/jbmr.4133.
FROM THE JOURNAL OF BONE AND MINERAL RESEARCH
New osteoporosis recommendations from AACE help therapy selection
Recommendations on use of the new dual-action anabolic agent romosozumab (Evenity, Amgen) and how to safely transition between osteoporosis agents are two of the issues addressed in the latest clinical practice guidelines for the diagnosis and treatment of postmenopausal osteoporosis from the American Association of Clinical Endocrinologists and American College of Endocrinology.
“This guideline is a practical tool for endocrinologists, physicians in general, regulatory bodies, health-related organizations, and interested laypersons regarding the diagnosis, evaluation, and treatment of postmenopausal osteoporosis,” the authors wrote.
The guidelines focus on 12 key clinical questions related to postmenopausal osteoporosis, with 52 specific recommendations, each graded according to the level of evidence.
They also include a treatment algorithm to help guide choice of therapy.
Reiterating role of FRAX in the diagnosis of patients with osteopenia
Among key updates is an emphasis on the role of the Fracture Risk Assessment Tool (FRAX) in the diagnosis of osteoporosis in patients with osteopenia.
While patients have traditionally been diagnosed with osteoporosis based on the presence of low bone mineral density (BMD) in the absence of fracture, the updated guidelines indicate that osteoporosis may be diagnosed in patients with osteopenia and an increased fracture risk using FRAX.
“The use of FRAX and osteopenia to diagnosis osteoporosis was first proposed by the National Bone Health Alliance years ago, and in the 2016 guideline, we agreed with it,” Pauline M. Camacho, MD, cochair of the guidelines task force, said in an interview.
“We reiterate in the 2020 guideline that we feel this is a valid diagnostic criteria,” said Dr. Camacho, professor of medicine and director of the Osteoporosis and Metabolic Bone Disease Center at Loyola University Chicago, Maywood, Ill. “It makes sense because when the thresholds are met by FRAX in patients with osteopenia, treatment is recommended. Therefore, why would they not fulfill treatment criteria for diagnosing osteoporosis?”
An increased risk of fracture based on a FRAX score may also be used to determine pharmacologic therapy, as can other traditional factors such as a low T score or a fragility fracture, the guidelines stated.
High risk vs. very high risk guides choice of first therapy
Another key update is the clarification of the risk stratification of patients who are high risk versus very high risk, which is key in determining the initial choice of agents and duration of therapy.
Specifically, patients should be considered at a very high fracture risk if they have the following criteria: a recent fracture (e.g., within the past 12 months), fractures while on approved osteoporosis therapy, multiple fractures, fractures while on drugs causing skeletal harm (e.g., long-term glucocorticoids), very low T score (e.g., less than −3.0), a high risk for falls or history of injurious falls, and a very high fracture probability by FRAX (e.g., major osteoporosis fracture >30%, hip fracture >4.5%) or other validated fracture risk algorithm.
Meanwhile, patients should be considered at high risk if they have been diagnosed with osteoporosis but do not meet the criteria for very high fracture risk.
Romosozumab brought into the mix
Another important update provides information on the role of one of the newest osteoporosis agents on the market, the anabolic drug romosozumab, a monoclonal antibody directed against sclerostin.
The drug’s approval by the Food and Drug Administration in 2019 for postmenopausal women at high risk of fracture was based on two large trials that showed dramatic increases in bone density through modeling as well as remodeling.
Those studies specifically showed significant reductions in radiographic vertebral fractures with romosozumab, compared with placebo and alendronate.
Dr. Camacho noted that romosozumab “will likely be for the very high risk group and those who have maxed out on teriparatide or abaloparatide.”
Romosozumab can safely be used in patients with prior radiation exposure, the guidelines noted.
Importantly, because of reports of a higher risk of serious cardiovascular events with romosozumab, compared with alendronate, romosozumab comes with a black-box warning that it should not be used in patients at high risk for cardiovascular events or who have had a recent myocardial infarction or stroke.
“Unfortunately, the very high risk group is often the older patients,” Dr. Camacho noted.
“The drug should not be given if there is a history of myocardial infarction or stroke in the past year,” she emphasized. “Clinical judgment is needed to decide who is at risk for cardiovascular complications.”
Notably, teriparatide and abaloparatide have black box warnings of their own regarding risk for osteosarcoma.
Switching therapies
Reflecting the evolving data on osteoporosis drug holidays, the guidelines also addressed the issue and the clinical challenges of switching therapies.
“In 2016, we said drug holidays are not recommended, and the treatment can be continued indefinitely, [however] in 2020, we felt that if some patients are no longer high risk, they can be transitioned off the drug,” Dr. Camacho said.
For teriparatide and abaloparatide, the FDA recommends treatment be limited to no more than 2 years, and for romosozumab, 1 year.
The updated guidelines recommend that upon discontinuation of an anabolic agent (e.g., abaloparatide, romosozumab, or teriparatide), a switch to therapy with an antiresorptive agent, such as denosumab or bisphosphonates, should be implemented to prevent loss of BMD and fracture efficacy.
Discontinuation of denosumab, however, can have notably negative effects. Clinical trials show rapid decreases in BMD when denosumab treatment is stopped after 2 or 8 years, as well as rapid loss of protection from vertebral fractures.
Therefore, if denosumab is going to be discontinued, there should be a proper transition to an antiresorptive agent for a limited time, such as one infusion of the bisphosphonate zoledronate.
Communicate the risks with and without treatment to patients
The authors underscored that, in addition to communicating the potential risk and expected benefits of osteoporosis treatments, clinicians should make sure patients fully appreciate the risk of fractures and their consequences, such as pain, disability, loss of independence, and death, when no treatment is given.
“It is incumbent on the clinician to provide this information to each patient in a manner that is fully understood, and it is equally important to learn from the patient about cultural beliefs, previous treatment experiences, fears, and concerns,” they wrote.
And in estimating patients’ fracture risk, T score must be combined with clinical risk factors, particularly advanced age and previous fracture, and clinicians should recognize that the absolute fracture risk is more useful than a risk ratio in developing treatment plans.
“Treatment recommendations may be quite different; an early postmenopausal woman with a T score of −2.5 has osteoporosis, although fracture risk is much lower than an 80-year-old woman with the same T score,” the authors explained.
Dr. Camacho reported financial relationships with Amgen and Shire. Disclosures for other task force members are detailed in the guidelines.
A version of this article originally appeared on Medscape.com.
Recommendations on use of the new dual-action anabolic agent romosozumab (Evenity, Amgen) and how to safely transition between osteoporosis agents are two of the issues addressed in the latest clinical practice guidelines for the diagnosis and treatment of postmenopausal osteoporosis from the American Association of Clinical Endocrinologists and American College of Endocrinology.
“This guideline is a practical tool for endocrinologists, physicians in general, regulatory bodies, health-related organizations, and interested laypersons regarding the diagnosis, evaluation, and treatment of postmenopausal osteoporosis,” the authors wrote.
The guidelines focus on 12 key clinical questions related to postmenopausal osteoporosis, with 52 specific recommendations, each graded according to the level of evidence.
They also include a treatment algorithm to help guide choice of therapy.
Reiterating role of FRAX in the diagnosis of patients with osteopenia
Among key updates is an emphasis on the role of the Fracture Risk Assessment Tool (FRAX) in the diagnosis of osteoporosis in patients with osteopenia.
While patients have traditionally been diagnosed with osteoporosis based on the presence of low bone mineral density (BMD) in the absence of fracture, the updated guidelines indicate that osteoporosis may be diagnosed in patients with osteopenia and an increased fracture risk using FRAX.
“The use of FRAX and osteopenia to diagnosis osteoporosis was first proposed by the National Bone Health Alliance years ago, and in the 2016 guideline, we agreed with it,” Pauline M. Camacho, MD, cochair of the guidelines task force, said in an interview.
“We reiterate in the 2020 guideline that we feel this is a valid diagnostic criteria,” said Dr. Camacho, professor of medicine and director of the Osteoporosis and Metabolic Bone Disease Center at Loyola University Chicago, Maywood, Ill. “It makes sense because when the thresholds are met by FRAX in patients with osteopenia, treatment is recommended. Therefore, why would they not fulfill treatment criteria for diagnosing osteoporosis?”
An increased risk of fracture based on a FRAX score may also be used to determine pharmacologic therapy, as can other traditional factors such as a low T score or a fragility fracture, the guidelines stated.
High risk vs. very high risk guides choice of first therapy
Another key update is the clarification of the risk stratification of patients who are high risk versus very high risk, which is key in determining the initial choice of agents and duration of therapy.
Specifically, patients should be considered at a very high fracture risk if they have the following criteria: a recent fracture (e.g., within the past 12 months), fractures while on approved osteoporosis therapy, multiple fractures, fractures while on drugs causing skeletal harm (e.g., long-term glucocorticoids), very low T score (e.g., less than −3.0), a high risk for falls or history of injurious falls, and a very high fracture probability by FRAX (e.g., major osteoporosis fracture >30%, hip fracture >4.5%) or other validated fracture risk algorithm.
Meanwhile, patients should be considered at high risk if they have been diagnosed with osteoporosis but do not meet the criteria for very high fracture risk.
Romosozumab brought into the mix
Another important update provides information on the role of one of the newest osteoporosis agents on the market, the anabolic drug romosozumab, a monoclonal antibody directed against sclerostin.
The drug’s approval by the Food and Drug Administration in 2019 for postmenopausal women at high risk of fracture was based on two large trials that showed dramatic increases in bone density through modeling as well as remodeling.
Those studies specifically showed significant reductions in radiographic vertebral fractures with romosozumab, compared with placebo and alendronate.
Dr. Camacho noted that romosozumab “will likely be for the very high risk group and those who have maxed out on teriparatide or abaloparatide.”
Romosozumab can safely be used in patients with prior radiation exposure, the guidelines noted.
Importantly, because of reports of a higher risk of serious cardiovascular events with romosozumab, compared with alendronate, romosozumab comes with a black-box warning that it should not be used in patients at high risk for cardiovascular events or who have had a recent myocardial infarction or stroke.
“Unfortunately, the very high risk group is often the older patients,” Dr. Camacho noted.
“The drug should not be given if there is a history of myocardial infarction or stroke in the past year,” she emphasized. “Clinical judgment is needed to decide who is at risk for cardiovascular complications.”
Notably, teriparatide and abaloparatide have black box warnings of their own regarding risk for osteosarcoma.
Switching therapies
Reflecting the evolving data on osteoporosis drug holidays, the guidelines also addressed the issue and the clinical challenges of switching therapies.
“In 2016, we said drug holidays are not recommended, and the treatment can be continued indefinitely, [however] in 2020, we felt that if some patients are no longer high risk, they can be transitioned off the drug,” Dr. Camacho said.
For teriparatide and abaloparatide, the FDA recommends treatment be limited to no more than 2 years, and for romosozumab, 1 year.
The updated guidelines recommend that upon discontinuation of an anabolic agent (e.g., abaloparatide, romosozumab, or teriparatide), a switch to therapy with an antiresorptive agent, such as denosumab or bisphosphonates, should be implemented to prevent loss of BMD and fracture efficacy.
Discontinuation of denosumab, however, can have notably negative effects. Clinical trials show rapid decreases in BMD when denosumab treatment is stopped after 2 or 8 years, as well as rapid loss of protection from vertebral fractures.
Therefore, if denosumab is going to be discontinued, there should be a proper transition to an antiresorptive agent for a limited time, such as one infusion of the bisphosphonate zoledronate.
Communicate the risks with and without treatment to patients
The authors underscored that, in addition to communicating the potential risk and expected benefits of osteoporosis treatments, clinicians should make sure patients fully appreciate the risk of fractures and their consequences, such as pain, disability, loss of independence, and death, when no treatment is given.
“It is incumbent on the clinician to provide this information to each patient in a manner that is fully understood, and it is equally important to learn from the patient about cultural beliefs, previous treatment experiences, fears, and concerns,” they wrote.
And in estimating patients’ fracture risk, T score must be combined with clinical risk factors, particularly advanced age and previous fracture, and clinicians should recognize that the absolute fracture risk is more useful than a risk ratio in developing treatment plans.
“Treatment recommendations may be quite different; an early postmenopausal woman with a T score of −2.5 has osteoporosis, although fracture risk is much lower than an 80-year-old woman with the same T score,” the authors explained.
Dr. Camacho reported financial relationships with Amgen and Shire. Disclosures for other task force members are detailed in the guidelines.
A version of this article originally appeared on Medscape.com.
Recommendations on use of the new dual-action anabolic agent romosozumab (Evenity, Amgen) and how to safely transition between osteoporosis agents are two of the issues addressed in the latest clinical practice guidelines for the diagnosis and treatment of postmenopausal osteoporosis from the American Association of Clinical Endocrinologists and American College of Endocrinology.
“This guideline is a practical tool for endocrinologists, physicians in general, regulatory bodies, health-related organizations, and interested laypersons regarding the diagnosis, evaluation, and treatment of postmenopausal osteoporosis,” the authors wrote.
The guidelines focus on 12 key clinical questions related to postmenopausal osteoporosis, with 52 specific recommendations, each graded according to the level of evidence.
They also include a treatment algorithm to help guide choice of therapy.
Reiterating role of FRAX in the diagnosis of patients with osteopenia
Among key updates is an emphasis on the role of the Fracture Risk Assessment Tool (FRAX) in the diagnosis of osteoporosis in patients with osteopenia.
While patients have traditionally been diagnosed with osteoporosis based on the presence of low bone mineral density (BMD) in the absence of fracture, the updated guidelines indicate that osteoporosis may be diagnosed in patients with osteopenia and an increased fracture risk using FRAX.
“The use of FRAX and osteopenia to diagnosis osteoporosis was first proposed by the National Bone Health Alliance years ago, and in the 2016 guideline, we agreed with it,” Pauline M. Camacho, MD, cochair of the guidelines task force, said in an interview.
“We reiterate in the 2020 guideline that we feel this is a valid diagnostic criteria,” said Dr. Camacho, professor of medicine and director of the Osteoporosis and Metabolic Bone Disease Center at Loyola University Chicago, Maywood, Ill. “It makes sense because when the thresholds are met by FRAX in patients with osteopenia, treatment is recommended. Therefore, why would they not fulfill treatment criteria for diagnosing osteoporosis?”
An increased risk of fracture based on a FRAX score may also be used to determine pharmacologic therapy, as can other traditional factors such as a low T score or a fragility fracture, the guidelines stated.
High risk vs. very high risk guides choice of first therapy
Another key update is the clarification of the risk stratification of patients who are high risk versus very high risk, which is key in determining the initial choice of agents and duration of therapy.
Specifically, patients should be considered at a very high fracture risk if they have the following criteria: a recent fracture (e.g., within the past 12 months), fractures while on approved osteoporosis therapy, multiple fractures, fractures while on drugs causing skeletal harm (e.g., long-term glucocorticoids), very low T score (e.g., less than −3.0), a high risk for falls or history of injurious falls, and a very high fracture probability by FRAX (e.g., major osteoporosis fracture >30%, hip fracture >4.5%) or other validated fracture risk algorithm.
Meanwhile, patients should be considered at high risk if they have been diagnosed with osteoporosis but do not meet the criteria for very high fracture risk.
Romosozumab brought into the mix
Another important update provides information on the role of one of the newest osteoporosis agents on the market, the anabolic drug romosozumab, a monoclonal antibody directed against sclerostin.
The drug’s approval by the Food and Drug Administration in 2019 for postmenopausal women at high risk of fracture was based on two large trials that showed dramatic increases in bone density through modeling as well as remodeling.
Those studies specifically showed significant reductions in radiographic vertebral fractures with romosozumab, compared with placebo and alendronate.
Dr. Camacho noted that romosozumab “will likely be for the very high risk group and those who have maxed out on teriparatide or abaloparatide.”
Romosozumab can safely be used in patients with prior radiation exposure, the guidelines noted.
Importantly, because of reports of a higher risk of serious cardiovascular events with romosozumab, compared with alendronate, romosozumab comes with a black-box warning that it should not be used in patients at high risk for cardiovascular events or who have had a recent myocardial infarction or stroke.
“Unfortunately, the very high risk group is often the older patients,” Dr. Camacho noted.
“The drug should not be given if there is a history of myocardial infarction or stroke in the past year,” she emphasized. “Clinical judgment is needed to decide who is at risk for cardiovascular complications.”
Notably, teriparatide and abaloparatide have black box warnings of their own regarding risk for osteosarcoma.
Switching therapies
Reflecting the evolving data on osteoporosis drug holidays, the guidelines also addressed the issue and the clinical challenges of switching therapies.
“In 2016, we said drug holidays are not recommended, and the treatment can be continued indefinitely, [however] in 2020, we felt that if some patients are no longer high risk, they can be transitioned off the drug,” Dr. Camacho said.
For teriparatide and abaloparatide, the FDA recommends treatment be limited to no more than 2 years, and for romosozumab, 1 year.
The updated guidelines recommend that upon discontinuation of an anabolic agent (e.g., abaloparatide, romosozumab, or teriparatide), a switch to therapy with an antiresorptive agent, such as denosumab or bisphosphonates, should be implemented to prevent loss of BMD and fracture efficacy.
Discontinuation of denosumab, however, can have notably negative effects. Clinical trials show rapid decreases in BMD when denosumab treatment is stopped after 2 or 8 years, as well as rapid loss of protection from vertebral fractures.
Therefore, if denosumab is going to be discontinued, there should be a proper transition to an antiresorptive agent for a limited time, such as one infusion of the bisphosphonate zoledronate.
Communicate the risks with and without treatment to patients
The authors underscored that, in addition to communicating the potential risk and expected benefits of osteoporosis treatments, clinicians should make sure patients fully appreciate the risk of fractures and their consequences, such as pain, disability, loss of independence, and death, when no treatment is given.
“It is incumbent on the clinician to provide this information to each patient in a manner that is fully understood, and it is equally important to learn from the patient about cultural beliefs, previous treatment experiences, fears, and concerns,” they wrote.
And in estimating patients’ fracture risk, T score must be combined with clinical risk factors, particularly advanced age and previous fracture, and clinicians should recognize that the absolute fracture risk is more useful than a risk ratio in developing treatment plans.
“Treatment recommendations may be quite different; an early postmenopausal woman with a T score of −2.5 has osteoporosis, although fracture risk is much lower than an 80-year-old woman with the same T score,” the authors explained.
Dr. Camacho reported financial relationships with Amgen and Shire. Disclosures for other task force members are detailed in the guidelines.
A version of this article originally appeared on Medscape.com.
Abaloparatide shows no effect on cardiovascular risk in postmenopausal women
Osteoporosis treatment with abaloparatide in postmenopausal women does not lead to increased cardiovascular risk, according to a post hoc analysis of the pivotal ACTIVE and ACTIVExtend trials.
“Neither treatment with abaloparatide or teriparatide was associated with an increase in serious cardiac [adverse events],” wrote Felicia Cosman, MD, of Columbia University, New York, and coauthors. The study was published in the Journal of Clinical Endocrinology.
To assess the cardiovascular safety profile of abaloparatide, a synthetic analogue of parathyroid hormone–related peptide, the researchers analyzed data on heart rate, blood pressure and cardiovascular-related adverse events (AEs) from patients taking part in the Abaloparatide Comparator Trial in Vertebral Endpoints (ACTIVE) trial and its ACTIVExtend extension study.
The 2,460 participants in the ACTIVE trial were postmenopausal women between the ages of 49 and 86 years with osteoporosis; they were given 80 mcg of daily subcutaneous abaloparatide, 20 mcg of open-label daily subcutaneous teriparatide, or placebo in roughly equal numbers for 18 months. After a 1-month treatment-free period, 1,133 eligible participants from either the abaloparatide or placebo groups were enrolled in ACTIVExtend and given 70 mg of open-label alendronate once a week for 24 months. Because heart rate was only assessed pre- and post dose in the ACTIVE trial, an additional pharmacology study of abaloparatide involving 55 healthy volunteers (32 men and 23 women) was undertaken. After a dose of either abaloparatide or placebo, heart rate was measured at 15, 30, and 45 minutes and 1, 1.5, 2, 2.5, 4, 6, 8, and 12 hours.
Overall, treatment-emergent AEs were higher in the abaloparatide (165, 20.1%) and teriparatide (106, 13%) groups, compared with placebo (74, 9%), as were AEs that led to discontinuation of the study and were potentially associated with changes in heart rate or BP (27 in abaloparatide, 11 in teriparatide, and 5 in placebo). However, the percentage of patients with serious cardiac AEs was similar across groups (1%, 1%, and 0.9%, respectively).
During the ACTIVE trial, major cardiac adverse events plus heart failure were more common in the placebo group (1.7%) than the abaloparatide (0.5%) or teriparatide (0.6%) groups. During ACTIVExtend, major cardiac adverse plus heart failure were similarly common in the abaloparatide/alendronate (1.6%) and the placebo/alendronate (1.6%) groups.
On day 1 of treatment during ACTIVE, the mean change in heart rate from pretreatment to an hour post treatment was 7.9 bpm, 5.3 bpm, and 1.2 bpm for abaloparatide, teriparatide, and placebo, respectively (P < .0001 for abaloparatide and teriparatide vs. placebo; P < .05 for abaloparatide vs. teriparatide).
Subsequent visits saw similar changes. The mean maximum heart rate at 1 hour post dose was 80.7 bpm for abaloparatide, 79.0 bpm for teriparatide, and 73.7 bpm for placebo (P < .0001 for abaloparatide and teriparatide vs. placebo; P < .01 for abaloparatide vs. teriparatide). In the study of healthy volunteers, HR peaked at 15 minutes after dosing and then declined, resolving within 2.5-4 hours.
From predose to 1 hour post dose, small but significant decreases were observed in mean supine systolic and diastolic BP across groups (–2.7/–3.6 mm Hg with abaloparatide, –2.0/–3.6 with teriparatide, –1.5/–2.3 with placebo). During the first year of ACTIVE, the mean maximal decrease in BP from predose to 1 hour post dose was slightly higher (1-2 mm Hg) in the abaloparatide and teriparatide groups, compared with the placebo group (P < .05).
The authors acknowledged their study’s limitations, including the analysis of major cardiac adverse plus heart failure in ACTIVE being limited because of a low number of events and the trial not being designed in that regard.
Abaloparatide was approved by the Food and Drug Administration in 2017 on the basis of results from the ACTIVE and ACTIVExtend trials showing significant reductions in new vertebral and nonvertebral fractures, compared with placebo.
The analysis was partially funded by Radius Health. Its authors acknowledged numerous potential conflicts of interest, including receiving grants and research support from various organizations and pharmaceutical companies.
SOURCE: Cosman F et al. J Clin Endocrinol Metab. 2020 Jul 13. doi: 10.1210/clinem/dgaa450.
Osteoporosis treatment with abaloparatide in postmenopausal women does not lead to increased cardiovascular risk, according to a post hoc analysis of the pivotal ACTIVE and ACTIVExtend trials.
“Neither treatment with abaloparatide or teriparatide was associated with an increase in serious cardiac [adverse events],” wrote Felicia Cosman, MD, of Columbia University, New York, and coauthors. The study was published in the Journal of Clinical Endocrinology.
To assess the cardiovascular safety profile of abaloparatide, a synthetic analogue of parathyroid hormone–related peptide, the researchers analyzed data on heart rate, blood pressure and cardiovascular-related adverse events (AEs) from patients taking part in the Abaloparatide Comparator Trial in Vertebral Endpoints (ACTIVE) trial and its ACTIVExtend extension study.
The 2,460 participants in the ACTIVE trial were postmenopausal women between the ages of 49 and 86 years with osteoporosis; they were given 80 mcg of daily subcutaneous abaloparatide, 20 mcg of open-label daily subcutaneous teriparatide, or placebo in roughly equal numbers for 18 months. After a 1-month treatment-free period, 1,133 eligible participants from either the abaloparatide or placebo groups were enrolled in ACTIVExtend and given 70 mg of open-label alendronate once a week for 24 months. Because heart rate was only assessed pre- and post dose in the ACTIVE trial, an additional pharmacology study of abaloparatide involving 55 healthy volunteers (32 men and 23 women) was undertaken. After a dose of either abaloparatide or placebo, heart rate was measured at 15, 30, and 45 minutes and 1, 1.5, 2, 2.5, 4, 6, 8, and 12 hours.
Overall, treatment-emergent AEs were higher in the abaloparatide (165, 20.1%) and teriparatide (106, 13%) groups, compared with placebo (74, 9%), as were AEs that led to discontinuation of the study and were potentially associated with changes in heart rate or BP (27 in abaloparatide, 11 in teriparatide, and 5 in placebo). However, the percentage of patients with serious cardiac AEs was similar across groups (1%, 1%, and 0.9%, respectively).
During the ACTIVE trial, major cardiac adverse events plus heart failure were more common in the placebo group (1.7%) than the abaloparatide (0.5%) or teriparatide (0.6%) groups. During ACTIVExtend, major cardiac adverse plus heart failure were similarly common in the abaloparatide/alendronate (1.6%) and the placebo/alendronate (1.6%) groups.
On day 1 of treatment during ACTIVE, the mean change in heart rate from pretreatment to an hour post treatment was 7.9 bpm, 5.3 bpm, and 1.2 bpm for abaloparatide, teriparatide, and placebo, respectively (P < .0001 for abaloparatide and teriparatide vs. placebo; P < .05 for abaloparatide vs. teriparatide).
Subsequent visits saw similar changes. The mean maximum heart rate at 1 hour post dose was 80.7 bpm for abaloparatide, 79.0 bpm for teriparatide, and 73.7 bpm for placebo (P < .0001 for abaloparatide and teriparatide vs. placebo; P < .01 for abaloparatide vs. teriparatide). In the study of healthy volunteers, HR peaked at 15 minutes after dosing and then declined, resolving within 2.5-4 hours.
From predose to 1 hour post dose, small but significant decreases were observed in mean supine systolic and diastolic BP across groups (–2.7/–3.6 mm Hg with abaloparatide, –2.0/–3.6 with teriparatide, –1.5/–2.3 with placebo). During the first year of ACTIVE, the mean maximal decrease in BP from predose to 1 hour post dose was slightly higher (1-2 mm Hg) in the abaloparatide and teriparatide groups, compared with the placebo group (P < .05).
The authors acknowledged their study’s limitations, including the analysis of major cardiac adverse plus heart failure in ACTIVE being limited because of a low number of events and the trial not being designed in that regard.
Abaloparatide was approved by the Food and Drug Administration in 2017 on the basis of results from the ACTIVE and ACTIVExtend trials showing significant reductions in new vertebral and nonvertebral fractures, compared with placebo.
The analysis was partially funded by Radius Health. Its authors acknowledged numerous potential conflicts of interest, including receiving grants and research support from various organizations and pharmaceutical companies.
SOURCE: Cosman F et al. J Clin Endocrinol Metab. 2020 Jul 13. doi: 10.1210/clinem/dgaa450.
Osteoporosis treatment with abaloparatide in postmenopausal women does not lead to increased cardiovascular risk, according to a post hoc analysis of the pivotal ACTIVE and ACTIVExtend trials.
“Neither treatment with abaloparatide or teriparatide was associated with an increase in serious cardiac [adverse events],” wrote Felicia Cosman, MD, of Columbia University, New York, and coauthors. The study was published in the Journal of Clinical Endocrinology.
To assess the cardiovascular safety profile of abaloparatide, a synthetic analogue of parathyroid hormone–related peptide, the researchers analyzed data on heart rate, blood pressure and cardiovascular-related adverse events (AEs) from patients taking part in the Abaloparatide Comparator Trial in Vertebral Endpoints (ACTIVE) trial and its ACTIVExtend extension study.
The 2,460 participants in the ACTIVE trial were postmenopausal women between the ages of 49 and 86 years with osteoporosis; they were given 80 mcg of daily subcutaneous abaloparatide, 20 mcg of open-label daily subcutaneous teriparatide, or placebo in roughly equal numbers for 18 months. After a 1-month treatment-free period, 1,133 eligible participants from either the abaloparatide or placebo groups were enrolled in ACTIVExtend and given 70 mg of open-label alendronate once a week for 24 months. Because heart rate was only assessed pre- and post dose in the ACTIVE trial, an additional pharmacology study of abaloparatide involving 55 healthy volunteers (32 men and 23 women) was undertaken. After a dose of either abaloparatide or placebo, heart rate was measured at 15, 30, and 45 minutes and 1, 1.5, 2, 2.5, 4, 6, 8, and 12 hours.
Overall, treatment-emergent AEs were higher in the abaloparatide (165, 20.1%) and teriparatide (106, 13%) groups, compared with placebo (74, 9%), as were AEs that led to discontinuation of the study and were potentially associated with changes in heart rate or BP (27 in abaloparatide, 11 in teriparatide, and 5 in placebo). However, the percentage of patients with serious cardiac AEs was similar across groups (1%, 1%, and 0.9%, respectively).
During the ACTIVE trial, major cardiac adverse events plus heart failure were more common in the placebo group (1.7%) than the abaloparatide (0.5%) or teriparatide (0.6%) groups. During ACTIVExtend, major cardiac adverse plus heart failure were similarly common in the abaloparatide/alendronate (1.6%) and the placebo/alendronate (1.6%) groups.
On day 1 of treatment during ACTIVE, the mean change in heart rate from pretreatment to an hour post treatment was 7.9 bpm, 5.3 bpm, and 1.2 bpm for abaloparatide, teriparatide, and placebo, respectively (P < .0001 for abaloparatide and teriparatide vs. placebo; P < .05 for abaloparatide vs. teriparatide).
Subsequent visits saw similar changes. The mean maximum heart rate at 1 hour post dose was 80.7 bpm for abaloparatide, 79.0 bpm for teriparatide, and 73.7 bpm for placebo (P < .0001 for abaloparatide and teriparatide vs. placebo; P < .01 for abaloparatide vs. teriparatide). In the study of healthy volunteers, HR peaked at 15 minutes after dosing and then declined, resolving within 2.5-4 hours.
From predose to 1 hour post dose, small but significant decreases were observed in mean supine systolic and diastolic BP across groups (–2.7/–3.6 mm Hg with abaloparatide, –2.0/–3.6 with teriparatide, –1.5/–2.3 with placebo). During the first year of ACTIVE, the mean maximal decrease in BP from predose to 1 hour post dose was slightly higher (1-2 mm Hg) in the abaloparatide and teriparatide groups, compared with the placebo group (P < .05).
The authors acknowledged their study’s limitations, including the analysis of major cardiac adverse plus heart failure in ACTIVE being limited because of a low number of events and the trial not being designed in that regard.
Abaloparatide was approved by the Food and Drug Administration in 2017 on the basis of results from the ACTIVE and ACTIVExtend trials showing significant reductions in new vertebral and nonvertebral fractures, compared with placebo.
The analysis was partially funded by Radius Health. Its authors acknowledged numerous potential conflicts of interest, including receiving grants and research support from various organizations and pharmaceutical companies.
SOURCE: Cosman F et al. J Clin Endocrinol Metab. 2020 Jul 13. doi: 10.1210/clinem/dgaa450.
FROM THE JOURNAL OF CLINICAL ENDOCRINOLOGY & METABOLISM
Move over supplements, here come medical foods
As the Food and Drug Administration focuses on other issues, companies, both big and small, are looking to boost physician and consumer interest in their “medical foods” – products that fall somewhere between drugs and supplements and promise to mitigate symptoms, or even address underlying pathologies, of a range of diseases.
Manufacturers now market an array of medical foods, ranging from powders and capsules for Alzheimer disease to low-protein spaghetti for chronic kidney disease (CKD). The FDA has not been completely absent; it takes a narrow view of what medical conditions qualify for treatment with food products and has warned some manufacturers that their misbranded products are acting more like unapproved drugs.
By the FDA’s definition, medical food is limited to products that provide crucial therapy for patients with inborn errors of metabolism (IEM). An example is specialized baby formula for infants with phenylketonuria. Unlike supplements, medical foods are supposed to be used under the supervision of a physician. This has prompted some sales reps to turn up in the clinic, and most manufacturers have online approval forms for doctors to sign. Manufacturers, advisers, and regulators were interviewed for a closer look at this burgeoning industry.
The market
The global market for medical foods – about $18 billion in 2019 – is expected to grow steadily in the near future. It is drawing more interest, especially in Europe, where medical foods are more accepted by physicians and consumers, Meghan Donnelly, MS, RDN, said in an interview. She is a registered dietitian who conducts physician outreach in the United States for Flavis, a division of Dr. Schär. That company, based in northern Italy, started out targeting IEMs but now also sells gluten-free foods for celiac disease and low-protein foods for CKD.
It is still a niche market in the United States – and isn’t likely to ever approach the size of the supplement market, according to Marcus Charuvastra, the managing director of Targeted Medical Pharma, which markets Theramine capsules for pain management, among many other products. But it could still be a big win for a manufacturer if they get a small slice of a big market, such as for Alzheimer disease.
Defining medical food
According to an update of the Orphan Drug Act in 1988, a medical food is “a food which is formulated to be consumed or administered enterally under the supervision of a physician and which is intended for the specific dietary management of a disease or condition for which distinctive nutritional requirements, based on recognized scientific principles, are established by medical evaluation.” The FDA issued regulations to accompany that law in 1993 but has since only issued a guidance document that is not legally binding.
Medical foods are not drugs and they are not supplements (the latter are intended only for healthy people). The FDA doesn’t require formal approval of a medical food, but, by law, the ingredients must be generally recognized as safe, and manufacturers must follow good manufacturing practices. However, the agency has taken a narrow view of what conditions require medical foods.
Policing medical foods hasn’t been a priority for the FDA, which is why there has been a proliferation of products that don’t meet the FDA’s view of the statutory definition of medical foods, according to Miriam Guggenheim, a food and drug law attorney in Washington, D.C. The FDA usually takes enforcement action when it sees a risk to the public’s health.
The agency’s stance has led to confusion – among manufacturers, physicians, consumers, and even regulators – making the market a kind of Wild West, according to Paul Hyman, a Washington, D.C.–based attorney who has represented medical food companies.
George A. Burdock, PhD, an Orlando-based regulatory consultant who has worked with medical food makers, believes the FDA will be forced to expand their narrow definition. He foresees a reconsideration of many medical food products in light of an October 2019 White House executive order prohibiting federal agencies from issuing guidance in lieu of rules.
Manufacturers and the FDA differ
One example of a product about which regulators and manufacturers differ is Theramine, which is described as “specially designed to supply the nervous system with the fuel it needs to meet the altered metabolic requirements of chronic pain and inflammatory disorders.”
It is not considered a medical food by the FDA, and the company has had numerous discussions with the agency about their diverging views, according to Mr. Charuvastra. “We’ve had our warning letters and we’ve had our sit downs, and we just had an inspection.”
Targeted Medical Pharma continues to market its products as medical foods but steers away from making any claims that they are like drugs, he said.
Confusion about medical foods has been exposed in the California Workers’ Compensation System by Leslie Wilson, PhD, and colleagues at the University of California, San Francisco. They found that physicians regularly wrote medical food prescriptions for non–FDA-approved uses and that the system reimbursed the majority of the products at a cost of $15.5 million from 2011 to 2013. More than half of these prescriptions were for Theramine.
Dr. Wilson reported that, for most products, no evidence supported effectiveness, and they were frequently mislabeled – for all 36 that were studied, submissions for reimbursement were made using a National Drug Code, an impossibility because medical foods are not drugs, and 14 were labeled “Rx only.”
Big-name companies joining in
The FDA does not keep a list of approved medical foods or manufacturers. Both small businesses and big food companies like Danone, Nestlé, and Abbott are players. Most products are sold online.
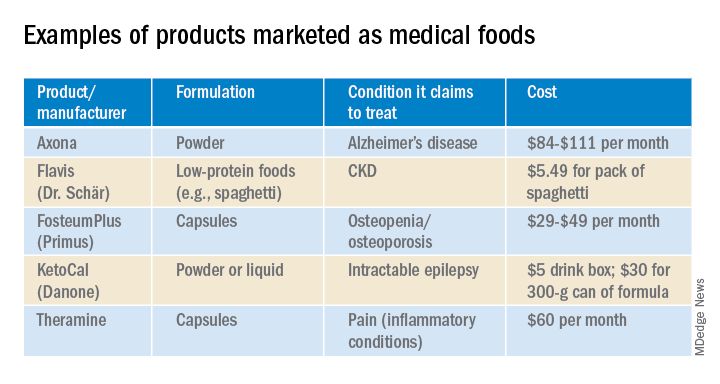
In the United States, Danone’s Nutricia division sells formulas and low-protein foods for IEMs. They also sell Ketocal, a powder or ready-to-drink liquid that is pitched as a balanced medical food to simplify and optimize the ketogenic diet for children with intractable epilepsy. Yet the FDA does not include epilepsy among the conditions that medical foods can treat.
Nestlé sells traditional medical foods for IEMs and also markets a range of what it calls nutritional therapies for such conditions as irritable bowel syndrome and dysphagia.
Nestlé is a minority shareholder in Axona, a product originally developed by Accera (Cerecin as of 2018). Jacquelyn Campo, senior director of global communications at Nestlé Health Sciences, said that the company is not actively involved in the operations management of Cerecin. However, on its website, Nestlé touts Axona, which is only available in the United States, as a “medical food” that “is intended for the clinical dietary management of mild to moderate Alzheimer disease.” The Axona site claims that the main ingredient, caprylic triglyceride, is broken down into ketones that provide fuel to treat cerebral hypometabolism, a precursor to Alzheimer disease. In a 2009 study, daily dosing of a preliminary formulation was associated with improved cognitive performance compared with placebo in patients with mild to moderate Alzheimer disease.
In 2013, the FDA warned Accera that it was misbranding Axona as a medical food and that the therapeutic claims the company was making would make the product an unapproved drug. Ms. Campo said Nestlé is aware of the agency’s warning, but added, “to our knowledge, Cerecin provided answers to the issues raised by the FDA.”
With the goal of getting drug approval, Accera went on to test a tweaked formulation in a 400-patient randomized, placebo-controlled trial called NOURISH AD that ultimately failed. Nevertheless, Axona is still marketed as a medical food. It costs about $100 for a month’s supply.
Repeated requests for comment from Cerecin were not answered. Danielle Schor, an FDA spokesperson, said the agency will not discuss the status of individual products.
More disputes and insurance coverage
Mary Ann DeMarco, executive director of sales and marketing for the Scottsdale, Ariz.–based medical food maker Primus Pharmaceuticals, said the company believes its products fit within the FDA’s medical foods rubric.
These include Fosteum Plus capsules, which it markets “for the clinical dietary management of the metabolic processes of osteopenia and osteoporosis.” The capsules contain a combination of genistein, zinc, calcium, phosphate, vitamin K2, and vitamin D. As proof of effectiveness, the company cites clinical data on some of the ingredients – not the product itself.
Primus has run afoul of the FDA before when it similarly positioned another product, called Limbrel, as a medical food for osteoarthritis. From 2007 to 2017, the FDA received 194 adverse event reports associated with Limbrel, including reports of drug-induced liver injury, pancreatitis, and hypersensitivity pneumonitis. In December 2017, the agency urged Primus to recall Limbrel, a move that it said was “necessary to protect the public health and welfare.” Primus withdrew the product but laid out a defense of Limbrel on a devoted website.
The FDA would not comment any further, said Ms. Schor. Ms. DeMarco said that Primus is working with the FDA to bring Limbrel back to market.
A lack of insurance coverage – even for approved medical foods for IEMs – has frustrated advocates, parents, and manufacturers. They are putting their weight behind the Medical Nutrition Equity Act, which would mandate public and private payer coverage of medical foods for IEMs and digestive conditions such as Crohn disease. That 2019 House bill has 56 cosponsors; there is no Senate companion bill.
“If you can get reimbursement, it really makes the market,” for Primus and the other manufacturers, Mr. Hyman said.
Primus Pharmaceuticals has launched its own campaign, Cover My Medical Foods, to enlist consumers and others to the cause.
Partnering with advocates
Although its low-protein breads, pastas, and baking products are not considered medical foods by the FDA, Dr. Schär is marketing them as such in the United States. They are trying to make a mark in CKD, according to Ms. Donnelly. She added that Dr. Schär has been successful in Europe, where nutrition therapy is more integrated in the health care system.
In 2019, Flavis and the National Kidney Foundation joined forces to raise awareness of nutritional interventions and to build enthusiasm for the Flavis products. The partnership has now ended, mostly because Flavis could no longer afford it, according to Ms. Donnelly.
“Information on diet and nutrition is the most requested subject matter from the NKF,” said Anthony Gucciardo, senior vice president of strategic partnerships at the foundation. The partnership “has never been necessarily about promoting their products per se; it’s promoting a healthy diet and really a diet specific for CKD.”
The NKF developed cobranded materials on low-protein foods for physicians and a teaching tool they could use with patients. Consumers could access nutrition information and a discount on Flavis products on a dedicated webpage. The foundation didn’t describe the low-protein products as medical foods, said Mr. Gucciardo, even if Flavis promoted them as such.
In patients with CKD, dietary management can help prevent the progression to end-stage renal disease. Although Medicare covers medical nutrition therapy – in which patients receive personalized assessments and dietary advice – uptake is abysmally low, according to a 2018 study.
Dr. Burdock thinks low-protein foods for CKD do meet the FDA’s criteria for a medical food but that the agency might not necessarily agree with him. The FDA would not comment.
Physician beware
When it comes to medical foods, the FDA has often looked the other way because the ingredients may already have been proven safe and the danger to an individual or to the public’s health is relatively low, according to Dr. Burdock and Mr. Hyman.
However, if the agency “feels that a medical food will prevent people from seeking medical care or there is potential to defraud the public, it is justified in taking action against the company,” said Dr. Burdock.
According to Dr. Wilson, the pharmacist who reported on the inappropriate medical food prescriptions in the California system, the FDA could help by creating a list of approved medical foods. Physicians should take time to learn about the difference between medical foods and supplements, she said, adding that they should also not hesitate to “question the veracity of the claims for them.”
Ms. Guggenheim believed doctors need to know that, for the most part, these are not FDA-approved products. She emphasized the importance of evaluating the products and looking at the data of their impact on a disease or condition.
“Many of these companies strongly believe that the products work and help people, so clinicians need to be very data driven,” she said.
A version of this article originally appeared on Medscape.com.
As the Food and Drug Administration focuses on other issues, companies, both big and small, are looking to boost physician and consumer interest in their “medical foods” – products that fall somewhere between drugs and supplements and promise to mitigate symptoms, or even address underlying pathologies, of a range of diseases.
Manufacturers now market an array of medical foods, ranging from powders and capsules for Alzheimer disease to low-protein spaghetti for chronic kidney disease (CKD). The FDA has not been completely absent; it takes a narrow view of what medical conditions qualify for treatment with food products and has warned some manufacturers that their misbranded products are acting more like unapproved drugs.
By the FDA’s definition, medical food is limited to products that provide crucial therapy for patients with inborn errors of metabolism (IEM). An example is specialized baby formula for infants with phenylketonuria. Unlike supplements, medical foods are supposed to be used under the supervision of a physician. This has prompted some sales reps to turn up in the clinic, and most manufacturers have online approval forms for doctors to sign. Manufacturers, advisers, and regulators were interviewed for a closer look at this burgeoning industry.
The market
The global market for medical foods – about $18 billion in 2019 – is expected to grow steadily in the near future. It is drawing more interest, especially in Europe, where medical foods are more accepted by physicians and consumers, Meghan Donnelly, MS, RDN, said in an interview. She is a registered dietitian who conducts physician outreach in the United States for Flavis, a division of Dr. Schär. That company, based in northern Italy, started out targeting IEMs but now also sells gluten-free foods for celiac disease and low-protein foods for CKD.
It is still a niche market in the United States – and isn’t likely to ever approach the size of the supplement market, according to Marcus Charuvastra, the managing director of Targeted Medical Pharma, which markets Theramine capsules for pain management, among many other products. But it could still be a big win for a manufacturer if they get a small slice of a big market, such as for Alzheimer disease.
Defining medical food
According to an update of the Orphan Drug Act in 1988, a medical food is “a food which is formulated to be consumed or administered enterally under the supervision of a physician and which is intended for the specific dietary management of a disease or condition for which distinctive nutritional requirements, based on recognized scientific principles, are established by medical evaluation.” The FDA issued regulations to accompany that law in 1993 but has since only issued a guidance document that is not legally binding.
Medical foods are not drugs and they are not supplements (the latter are intended only for healthy people). The FDA doesn’t require formal approval of a medical food, but, by law, the ingredients must be generally recognized as safe, and manufacturers must follow good manufacturing practices. However, the agency has taken a narrow view of what conditions require medical foods.
Policing medical foods hasn’t been a priority for the FDA, which is why there has been a proliferation of products that don’t meet the FDA’s view of the statutory definition of medical foods, according to Miriam Guggenheim, a food and drug law attorney in Washington, D.C. The FDA usually takes enforcement action when it sees a risk to the public’s health.
The agency’s stance has led to confusion – among manufacturers, physicians, consumers, and even regulators – making the market a kind of Wild West, according to Paul Hyman, a Washington, D.C.–based attorney who has represented medical food companies.
George A. Burdock, PhD, an Orlando-based regulatory consultant who has worked with medical food makers, believes the FDA will be forced to expand their narrow definition. He foresees a reconsideration of many medical food products in light of an October 2019 White House executive order prohibiting federal agencies from issuing guidance in lieu of rules.
Manufacturers and the FDA differ
One example of a product about which regulators and manufacturers differ is Theramine, which is described as “specially designed to supply the nervous system with the fuel it needs to meet the altered metabolic requirements of chronic pain and inflammatory disorders.”
It is not considered a medical food by the FDA, and the company has had numerous discussions with the agency about their diverging views, according to Mr. Charuvastra. “We’ve had our warning letters and we’ve had our sit downs, and we just had an inspection.”
Targeted Medical Pharma continues to market its products as medical foods but steers away from making any claims that they are like drugs, he said.
Confusion about medical foods has been exposed in the California Workers’ Compensation System by Leslie Wilson, PhD, and colleagues at the University of California, San Francisco. They found that physicians regularly wrote medical food prescriptions for non–FDA-approved uses and that the system reimbursed the majority of the products at a cost of $15.5 million from 2011 to 2013. More than half of these prescriptions were for Theramine.
Dr. Wilson reported that, for most products, no evidence supported effectiveness, and they were frequently mislabeled – for all 36 that were studied, submissions for reimbursement were made using a National Drug Code, an impossibility because medical foods are not drugs, and 14 were labeled “Rx only.”
Big-name companies joining in
The FDA does not keep a list of approved medical foods or manufacturers. Both small businesses and big food companies like Danone, Nestlé, and Abbott are players. Most products are sold online.

In the United States, Danone’s Nutricia division sells formulas and low-protein foods for IEMs. They also sell Ketocal, a powder or ready-to-drink liquid that is pitched as a balanced medical food to simplify and optimize the ketogenic diet for children with intractable epilepsy. Yet the FDA does not include epilepsy among the conditions that medical foods can treat.
Nestlé sells traditional medical foods for IEMs and also markets a range of what it calls nutritional therapies for such conditions as irritable bowel syndrome and dysphagia.
Nestlé is a minority shareholder in Axona, a product originally developed by Accera (Cerecin as of 2018). Jacquelyn Campo, senior director of global communications at Nestlé Health Sciences, said that the company is not actively involved in the operations management of Cerecin. However, on its website, Nestlé touts Axona, which is only available in the United States, as a “medical food” that “is intended for the clinical dietary management of mild to moderate Alzheimer disease.” The Axona site claims that the main ingredient, caprylic triglyceride, is broken down into ketones that provide fuel to treat cerebral hypometabolism, a precursor to Alzheimer disease. In a 2009 study, daily dosing of a preliminary formulation was associated with improved cognitive performance compared with placebo in patients with mild to moderate Alzheimer disease.
In 2013, the FDA warned Accera that it was misbranding Axona as a medical food and that the therapeutic claims the company was making would make the product an unapproved drug. Ms. Campo said Nestlé is aware of the agency’s warning, but added, “to our knowledge, Cerecin provided answers to the issues raised by the FDA.”
With the goal of getting drug approval, Accera went on to test a tweaked formulation in a 400-patient randomized, placebo-controlled trial called NOURISH AD that ultimately failed. Nevertheless, Axona is still marketed as a medical food. It costs about $100 for a month’s supply.
Repeated requests for comment from Cerecin were not answered. Danielle Schor, an FDA spokesperson, said the agency will not discuss the status of individual products.
More disputes and insurance coverage
Mary Ann DeMarco, executive director of sales and marketing for the Scottsdale, Ariz.–based medical food maker Primus Pharmaceuticals, said the company believes its products fit within the FDA’s medical foods rubric.
These include Fosteum Plus capsules, which it markets “for the clinical dietary management of the metabolic processes of osteopenia and osteoporosis.” The capsules contain a combination of genistein, zinc, calcium, phosphate, vitamin K2, and vitamin D. As proof of effectiveness, the company cites clinical data on some of the ingredients – not the product itself.
Primus has run afoul of the FDA before when it similarly positioned another product, called Limbrel, as a medical food for osteoarthritis. From 2007 to 2017, the FDA received 194 adverse event reports associated with Limbrel, including reports of drug-induced liver injury, pancreatitis, and hypersensitivity pneumonitis. In December 2017, the agency urged Primus to recall Limbrel, a move that it said was “necessary to protect the public health and welfare.” Primus withdrew the product but laid out a defense of Limbrel on a devoted website.
The FDA would not comment any further, said Ms. Schor. Ms. DeMarco said that Primus is working with the FDA to bring Limbrel back to market.
A lack of insurance coverage – even for approved medical foods for IEMs – has frustrated advocates, parents, and manufacturers. They are putting their weight behind the Medical Nutrition Equity Act, which would mandate public and private payer coverage of medical foods for IEMs and digestive conditions such as Crohn disease. That 2019 House bill has 56 cosponsors; there is no Senate companion bill.
“If you can get reimbursement, it really makes the market,” for Primus and the other manufacturers, Mr. Hyman said.
Primus Pharmaceuticals has launched its own campaign, Cover My Medical Foods, to enlist consumers and others to the cause.
Partnering with advocates
Although its low-protein breads, pastas, and baking products are not considered medical foods by the FDA, Dr. Schär is marketing them as such in the United States. They are trying to make a mark in CKD, according to Ms. Donnelly. She added that Dr. Schär has been successful in Europe, where nutrition therapy is more integrated in the health care system.
In 2019, Flavis and the National Kidney Foundation joined forces to raise awareness of nutritional interventions and to build enthusiasm for the Flavis products. The partnership has now ended, mostly because Flavis could no longer afford it, according to Ms. Donnelly.
“Information on diet and nutrition is the most requested subject matter from the NKF,” said Anthony Gucciardo, senior vice president of strategic partnerships at the foundation. The partnership “has never been necessarily about promoting their products per se; it’s promoting a healthy diet and really a diet specific for CKD.”
The NKF developed cobranded materials on low-protein foods for physicians and a teaching tool they could use with patients. Consumers could access nutrition information and a discount on Flavis products on a dedicated webpage. The foundation didn’t describe the low-protein products as medical foods, said Mr. Gucciardo, even if Flavis promoted them as such.
In patients with CKD, dietary management can help prevent the progression to end-stage renal disease. Although Medicare covers medical nutrition therapy – in which patients receive personalized assessments and dietary advice – uptake is abysmally low, according to a 2018 study.
Dr. Burdock thinks low-protein foods for CKD do meet the FDA’s criteria for a medical food but that the agency might not necessarily agree with him. The FDA would not comment.
Physician beware
When it comes to medical foods, the FDA has often looked the other way because the ingredients may already have been proven safe and the danger to an individual or to the public’s health is relatively low, according to Dr. Burdock and Mr. Hyman.
However, if the agency “feels that a medical food will prevent people from seeking medical care or there is potential to defraud the public, it is justified in taking action against the company,” said Dr. Burdock.
According to Dr. Wilson, the pharmacist who reported on the inappropriate medical food prescriptions in the California system, the FDA could help by creating a list of approved medical foods. Physicians should take time to learn about the difference between medical foods and supplements, she said, adding that they should also not hesitate to “question the veracity of the claims for them.”
Ms. Guggenheim believed doctors need to know that, for the most part, these are not FDA-approved products. She emphasized the importance of evaluating the products and looking at the data of their impact on a disease or condition.
“Many of these companies strongly believe that the products work and help people, so clinicians need to be very data driven,” she said.
A version of this article originally appeared on Medscape.com.
As the Food and Drug Administration focuses on other issues, companies, both big and small, are looking to boost physician and consumer interest in their “medical foods” – products that fall somewhere between drugs and supplements and promise to mitigate symptoms, or even address underlying pathologies, of a range of diseases.
Manufacturers now market an array of medical foods, ranging from powders and capsules for Alzheimer disease to low-protein spaghetti for chronic kidney disease (CKD). The FDA has not been completely absent; it takes a narrow view of what medical conditions qualify for treatment with food products and has warned some manufacturers that their misbranded products are acting more like unapproved drugs.
By the FDA’s definition, medical food is limited to products that provide crucial therapy for patients with inborn errors of metabolism (IEM). An example is specialized baby formula for infants with phenylketonuria. Unlike supplements, medical foods are supposed to be used under the supervision of a physician. This has prompted some sales reps to turn up in the clinic, and most manufacturers have online approval forms for doctors to sign. Manufacturers, advisers, and regulators were interviewed for a closer look at this burgeoning industry.
The market
The global market for medical foods – about $18 billion in 2019 – is expected to grow steadily in the near future. It is drawing more interest, especially in Europe, where medical foods are more accepted by physicians and consumers, Meghan Donnelly, MS, RDN, said in an interview. She is a registered dietitian who conducts physician outreach in the United States for Flavis, a division of Dr. Schär. That company, based in northern Italy, started out targeting IEMs but now also sells gluten-free foods for celiac disease and low-protein foods for CKD.
It is still a niche market in the United States – and isn’t likely to ever approach the size of the supplement market, according to Marcus Charuvastra, the managing director of Targeted Medical Pharma, which markets Theramine capsules for pain management, among many other products. But it could still be a big win for a manufacturer if they get a small slice of a big market, such as for Alzheimer disease.
Defining medical food
According to an update of the Orphan Drug Act in 1988, a medical food is “a food which is formulated to be consumed or administered enterally under the supervision of a physician and which is intended for the specific dietary management of a disease or condition for which distinctive nutritional requirements, based on recognized scientific principles, are established by medical evaluation.” The FDA issued regulations to accompany that law in 1993 but has since only issued a guidance document that is not legally binding.
Medical foods are not drugs and they are not supplements (the latter are intended only for healthy people). The FDA doesn’t require formal approval of a medical food, but, by law, the ingredients must be generally recognized as safe, and manufacturers must follow good manufacturing practices. However, the agency has taken a narrow view of what conditions require medical foods.
Policing medical foods hasn’t been a priority for the FDA, which is why there has been a proliferation of products that don’t meet the FDA’s view of the statutory definition of medical foods, according to Miriam Guggenheim, a food and drug law attorney in Washington, D.C. The FDA usually takes enforcement action when it sees a risk to the public’s health.
The agency’s stance has led to confusion – among manufacturers, physicians, consumers, and even regulators – making the market a kind of Wild West, according to Paul Hyman, a Washington, D.C.–based attorney who has represented medical food companies.
George A. Burdock, PhD, an Orlando-based regulatory consultant who has worked with medical food makers, believes the FDA will be forced to expand their narrow definition. He foresees a reconsideration of many medical food products in light of an October 2019 White House executive order prohibiting federal agencies from issuing guidance in lieu of rules.
Manufacturers and the FDA differ
One example of a product about which regulators and manufacturers differ is Theramine, which is described as “specially designed to supply the nervous system with the fuel it needs to meet the altered metabolic requirements of chronic pain and inflammatory disorders.”
It is not considered a medical food by the FDA, and the company has had numerous discussions with the agency about their diverging views, according to Mr. Charuvastra. “We’ve had our warning letters and we’ve had our sit downs, and we just had an inspection.”
Targeted Medical Pharma continues to market its products as medical foods but steers away from making any claims that they are like drugs, he said.
Confusion about medical foods has been exposed in the California Workers’ Compensation System by Leslie Wilson, PhD, and colleagues at the University of California, San Francisco. They found that physicians regularly wrote medical food prescriptions for non–FDA-approved uses and that the system reimbursed the majority of the products at a cost of $15.5 million from 2011 to 2013. More than half of these prescriptions were for Theramine.
Dr. Wilson reported that, for most products, no evidence supported effectiveness, and they were frequently mislabeled – for all 36 that were studied, submissions for reimbursement were made using a National Drug Code, an impossibility because medical foods are not drugs, and 14 were labeled “Rx only.”
Big-name companies joining in
The FDA does not keep a list of approved medical foods or manufacturers. Both small businesses and big food companies like Danone, Nestlé, and Abbott are players. Most products are sold online.

In the United States, Danone’s Nutricia division sells formulas and low-protein foods for IEMs. They also sell Ketocal, a powder or ready-to-drink liquid that is pitched as a balanced medical food to simplify and optimize the ketogenic diet for children with intractable epilepsy. Yet the FDA does not include epilepsy among the conditions that medical foods can treat.
Nestlé sells traditional medical foods for IEMs and also markets a range of what it calls nutritional therapies for such conditions as irritable bowel syndrome and dysphagia.
Nestlé is a minority shareholder in Axona, a product originally developed by Accera (Cerecin as of 2018). Jacquelyn Campo, senior director of global communications at Nestlé Health Sciences, said that the company is not actively involved in the operations management of Cerecin. However, on its website, Nestlé touts Axona, which is only available in the United States, as a “medical food” that “is intended for the clinical dietary management of mild to moderate Alzheimer disease.” The Axona site claims that the main ingredient, caprylic triglyceride, is broken down into ketones that provide fuel to treat cerebral hypometabolism, a precursor to Alzheimer disease. In a 2009 study, daily dosing of a preliminary formulation was associated with improved cognitive performance compared with placebo in patients with mild to moderate Alzheimer disease.
In 2013, the FDA warned Accera that it was misbranding Axona as a medical food and that the therapeutic claims the company was making would make the product an unapproved drug. Ms. Campo said Nestlé is aware of the agency’s warning, but added, “to our knowledge, Cerecin provided answers to the issues raised by the FDA.”
With the goal of getting drug approval, Accera went on to test a tweaked formulation in a 400-patient randomized, placebo-controlled trial called NOURISH AD that ultimately failed. Nevertheless, Axona is still marketed as a medical food. It costs about $100 for a month’s supply.
Repeated requests for comment from Cerecin were not answered. Danielle Schor, an FDA spokesperson, said the agency will not discuss the status of individual products.
More disputes and insurance coverage
Mary Ann DeMarco, executive director of sales and marketing for the Scottsdale, Ariz.–based medical food maker Primus Pharmaceuticals, said the company believes its products fit within the FDA’s medical foods rubric.
These include Fosteum Plus capsules, which it markets “for the clinical dietary management of the metabolic processes of osteopenia and osteoporosis.” The capsules contain a combination of genistein, zinc, calcium, phosphate, vitamin K2, and vitamin D. As proof of effectiveness, the company cites clinical data on some of the ingredients – not the product itself.
Primus has run afoul of the FDA before when it similarly positioned another product, called Limbrel, as a medical food for osteoarthritis. From 2007 to 2017, the FDA received 194 adverse event reports associated with Limbrel, including reports of drug-induced liver injury, pancreatitis, and hypersensitivity pneumonitis. In December 2017, the agency urged Primus to recall Limbrel, a move that it said was “necessary to protect the public health and welfare.” Primus withdrew the product but laid out a defense of Limbrel on a devoted website.
The FDA would not comment any further, said Ms. Schor. Ms. DeMarco said that Primus is working with the FDA to bring Limbrel back to market.
A lack of insurance coverage – even for approved medical foods for IEMs – has frustrated advocates, parents, and manufacturers. They are putting their weight behind the Medical Nutrition Equity Act, which would mandate public and private payer coverage of medical foods for IEMs and digestive conditions such as Crohn disease. That 2019 House bill has 56 cosponsors; there is no Senate companion bill.
“If you can get reimbursement, it really makes the market,” for Primus and the other manufacturers, Mr. Hyman said.
Primus Pharmaceuticals has launched its own campaign, Cover My Medical Foods, to enlist consumers and others to the cause.
Partnering with advocates
Although its low-protein breads, pastas, and baking products are not considered medical foods by the FDA, Dr. Schär is marketing them as such in the United States. They are trying to make a mark in CKD, according to Ms. Donnelly. She added that Dr. Schär has been successful in Europe, where nutrition therapy is more integrated in the health care system.
In 2019, Flavis and the National Kidney Foundation joined forces to raise awareness of nutritional interventions and to build enthusiasm for the Flavis products. The partnership has now ended, mostly because Flavis could no longer afford it, according to Ms. Donnelly.
“Information on diet and nutrition is the most requested subject matter from the NKF,” said Anthony Gucciardo, senior vice president of strategic partnerships at the foundation. The partnership “has never been necessarily about promoting their products per se; it’s promoting a healthy diet and really a diet specific for CKD.”
The NKF developed cobranded materials on low-protein foods for physicians and a teaching tool they could use with patients. Consumers could access nutrition information and a discount on Flavis products on a dedicated webpage. The foundation didn’t describe the low-protein products as medical foods, said Mr. Gucciardo, even if Flavis promoted them as such.
In patients with CKD, dietary management can help prevent the progression to end-stage renal disease. Although Medicare covers medical nutrition therapy – in which patients receive personalized assessments and dietary advice – uptake is abysmally low, according to a 2018 study.
Dr. Burdock thinks low-protein foods for CKD do meet the FDA’s criteria for a medical food but that the agency might not necessarily agree with him. The FDA would not comment.
Physician beware
When it comes to medical foods, the FDA has often looked the other way because the ingredients may already have been proven safe and the danger to an individual or to the public’s health is relatively low, according to Dr. Burdock and Mr. Hyman.
However, if the agency “feels that a medical food will prevent people from seeking medical care or there is potential to defraud the public, it is justified in taking action against the company,” said Dr. Burdock.
According to Dr. Wilson, the pharmacist who reported on the inappropriate medical food prescriptions in the California system, the FDA could help by creating a list of approved medical foods. Physicians should take time to learn about the difference between medical foods and supplements, she said, adding that they should also not hesitate to “question the veracity of the claims for them.”
Ms. Guggenheim believed doctors need to know that, for the most part, these are not FDA-approved products. She emphasized the importance of evaluating the products and looking at the data of their impact on a disease or condition.
“Many of these companies strongly believe that the products work and help people, so clinicians need to be very data driven,” she said.
A version of this article originally appeared on Medscape.com.