User login
IV fluid weaning unnecessary after gastroenteritis rehydration
SEATTLE – Intravenous fluids can simply be stopped after children with acute viral gastroenteritis are rehydrated in the hospital; there’s no need for a slow wean, according to a review at the Connecticut Children’s Medical Center, Hartford.
Researchers found that children leave the hospital hours sooner, with no ill effects. “This study suggests that slowly weaning IV fluids may not be necessary,” said lead investigator Danielle Klima, DO, a University of Connecticut pediatrics resident.
The team at Connecticut Children’s noticed that weaning practices after gastroenteritis rehydration varied widely on the pediatric floors, and appeared to be largely provider dependent, with “much subjective decision making.” The team wanted to see if it made a difference one way or the other, Dr. Klima said at Pediatric Hospital Medicine.
During respiratory season, “our pediatric floors are surging. Saving even a couple hours to get these kids out” quicker matters, she said, noting that it’s likely the first time the issue has been studied.
The team reviewed 153 children aged 2 months to 18 years, 95 of whom had IV fluids stopped once physicians deemed they were fluid resuscitated and ready for an oral feeding trial; the other 58 were weaned, with at least two reductions by half before final discontinuation.
There were no significant differences in age, gender, race, or insurance type between the two groups. The mean age was 2.6 years, and there were slightly more boys. The ED triage level was a mean of 3.2 points in both groups on a scale of 1-5, with 1 being the most urgent. Children with serious comorbidities, chronic diarrhea, feeding tubes, severe electrolyte abnormalities, or feeding problems were among those excluded.
Overall length of stay was 36 hours in the stop group versus 40.5 hours in the weaning group (P = .004). Children left the hospital about 6 hours after IV fluids were discontinued, versus 26 hours after weaning was started (P less than .001).
Electrolyte abnormalities on admission were more common in the weaning group (65% versus 57%), but not significantly so (P = .541). Electrolyte abnormalities were also more common at the end of fluid resuscitation in the weaning arm, but again not significantly (65% 42%, P = .077).
Fluid resuscitation needed to be restarted in 15 children in the stop group (16%), versus 11 (19%) in the wean arm (P = .459). One child in the stop group (1%) versus four (7%) who were weaned were readmitted to the hospital within a week for acute viral gastroenteritis (P = .067).
“I expected we were taking a more conservative weaning approach in younger infants,” but age didn’t seem to affect whether patients were weaned or not, Dr. Klima said.
With the results in hand, “our group is taking a closer look at exactly what we are doing,” perhaps with an eye toward standardization or even a randomized trial, she said.
She noted that weaning still makes sense for a fussy toddler who refuses to take anything by mouth.
There was no external funding, and Dr. Klima had no disclosures. The conference was sponsored by the Society of Hospital Medicine, the American Academy of Pediatrics, and the Academic Pediatric Association.
SEATTLE – Intravenous fluids can simply be stopped after children with acute viral gastroenteritis are rehydrated in the hospital; there’s no need for a slow wean, according to a review at the Connecticut Children’s Medical Center, Hartford.
Researchers found that children leave the hospital hours sooner, with no ill effects. “This study suggests that slowly weaning IV fluids may not be necessary,” said lead investigator Danielle Klima, DO, a University of Connecticut pediatrics resident.
The team at Connecticut Children’s noticed that weaning practices after gastroenteritis rehydration varied widely on the pediatric floors, and appeared to be largely provider dependent, with “much subjective decision making.” The team wanted to see if it made a difference one way or the other, Dr. Klima said at Pediatric Hospital Medicine.
During respiratory season, “our pediatric floors are surging. Saving even a couple hours to get these kids out” quicker matters, she said, noting that it’s likely the first time the issue has been studied.
The team reviewed 153 children aged 2 months to 18 years, 95 of whom had IV fluids stopped once physicians deemed they were fluid resuscitated and ready for an oral feeding trial; the other 58 were weaned, with at least two reductions by half before final discontinuation.
There were no significant differences in age, gender, race, or insurance type between the two groups. The mean age was 2.6 years, and there were slightly more boys. The ED triage level was a mean of 3.2 points in both groups on a scale of 1-5, with 1 being the most urgent. Children with serious comorbidities, chronic diarrhea, feeding tubes, severe electrolyte abnormalities, or feeding problems were among those excluded.
Overall length of stay was 36 hours in the stop group versus 40.5 hours in the weaning group (P = .004). Children left the hospital about 6 hours after IV fluids were discontinued, versus 26 hours after weaning was started (P less than .001).
Electrolyte abnormalities on admission were more common in the weaning group (65% versus 57%), but not significantly so (P = .541). Electrolyte abnormalities were also more common at the end of fluid resuscitation in the weaning arm, but again not significantly (65% 42%, P = .077).
Fluid resuscitation needed to be restarted in 15 children in the stop group (16%), versus 11 (19%) in the wean arm (P = .459). One child in the stop group (1%) versus four (7%) who were weaned were readmitted to the hospital within a week for acute viral gastroenteritis (P = .067).
“I expected we were taking a more conservative weaning approach in younger infants,” but age didn’t seem to affect whether patients were weaned or not, Dr. Klima said.
With the results in hand, “our group is taking a closer look at exactly what we are doing,” perhaps with an eye toward standardization or even a randomized trial, she said.
She noted that weaning still makes sense for a fussy toddler who refuses to take anything by mouth.
There was no external funding, and Dr. Klima had no disclosures. The conference was sponsored by the Society of Hospital Medicine, the American Academy of Pediatrics, and the Academic Pediatric Association.
SEATTLE – Intravenous fluids can simply be stopped after children with acute viral gastroenteritis are rehydrated in the hospital; there’s no need for a slow wean, according to a review at the Connecticut Children’s Medical Center, Hartford.
Researchers found that children leave the hospital hours sooner, with no ill effects. “This study suggests that slowly weaning IV fluids may not be necessary,” said lead investigator Danielle Klima, DO, a University of Connecticut pediatrics resident.
The team at Connecticut Children’s noticed that weaning practices after gastroenteritis rehydration varied widely on the pediatric floors, and appeared to be largely provider dependent, with “much subjective decision making.” The team wanted to see if it made a difference one way or the other, Dr. Klima said at Pediatric Hospital Medicine.
During respiratory season, “our pediatric floors are surging. Saving even a couple hours to get these kids out” quicker matters, she said, noting that it’s likely the first time the issue has been studied.
The team reviewed 153 children aged 2 months to 18 years, 95 of whom had IV fluids stopped once physicians deemed they were fluid resuscitated and ready for an oral feeding trial; the other 58 were weaned, with at least two reductions by half before final discontinuation.
There were no significant differences in age, gender, race, or insurance type between the two groups. The mean age was 2.6 years, and there were slightly more boys. The ED triage level was a mean of 3.2 points in both groups on a scale of 1-5, with 1 being the most urgent. Children with serious comorbidities, chronic diarrhea, feeding tubes, severe electrolyte abnormalities, or feeding problems were among those excluded.
Overall length of stay was 36 hours in the stop group versus 40.5 hours in the weaning group (P = .004). Children left the hospital about 6 hours after IV fluids were discontinued, versus 26 hours after weaning was started (P less than .001).
Electrolyte abnormalities on admission were more common in the weaning group (65% versus 57%), but not significantly so (P = .541). Electrolyte abnormalities were also more common at the end of fluid resuscitation in the weaning arm, but again not significantly (65% 42%, P = .077).
Fluid resuscitation needed to be restarted in 15 children in the stop group (16%), versus 11 (19%) in the wean arm (P = .459). One child in the stop group (1%) versus four (7%) who were weaned were readmitted to the hospital within a week for acute viral gastroenteritis (P = .067).
“I expected we were taking a more conservative weaning approach in younger infants,” but age didn’t seem to affect whether patients were weaned or not, Dr. Klima said.
With the results in hand, “our group is taking a closer look at exactly what we are doing,” perhaps with an eye toward standardization or even a randomized trial, she said.
She noted that weaning still makes sense for a fussy toddler who refuses to take anything by mouth.
There was no external funding, and Dr. Klima had no disclosures. The conference was sponsored by the Society of Hospital Medicine, the American Academy of Pediatrics, and the Academic Pediatric Association.
REPORTING FROM PHM 2019
Are your patients ready for the transition to adult care?
AUSTIN, TEX. – All too often, children and adolescents stumble on their way to adult health care – if they make it at all.
In fact, data from an ongoing survey by the Department of Health & Human Services and the Health Resources and Services Administration indicate that only 40% of children with special health care needs aged 12-17 years receive the services necessary to make transitions to adult health care.
“Most fall off the proverbial cliff and do not engage with adult providers,” Cynthia Peacock, MD, said at the annual meeting of the Society for Pediatric Dermatology. She recalled meeting with one individual who, after being a patient at the children’s hospital for 13 years, was told over the phone to transition to an adult provider with not so much as a promised letter of introduction being sent on. “She did not know about the adult health care culture, which is fractured in care, relies on the patient to be their own advocate, and wants patients to be able to do their visit in 10-15 minutes.”
Dr. Peacock, medical director of the Transition Medicine Clinic at Baylor College of Medicine, Houston, described “Starting at age 12 is not too early,” she said. “It gives you time so that you can keep introducing the concept when that individual keeps coming back to see you, especially if it’s a chronic condition.”
She recommended that clinicians ask several questions to assess transition readiness of pediatric patients to adult care, including, Do you know your medications? Do you know how to take them? Do you know how to refill them? Do you know how to discuss them? Can you discuss your medical condition with the adult doctor? Can you call for a doctor’s appointment or get a prescription filled? “Adolescents are notorious for calling [the doctor’s office], and if they’re told they can’t make an appointment, that’s it; they stop right there,” Dr. Peacock said. “They don’t tend to problem solve. They don’t engage.”
Studies have suggested that the transfer of care is more likely to be successful if a formal transition program is in place to prepare the patient and to facilitate the change in health care providers. “There is a growing evidence base in the literature that skills training for young people with chronic illnesses can be associated with positive outcomes,” she said. “This can be as easy as telling the individual, ‘Do a book report about your condition. Talk to a friend. Tell a friend what your condition is. Or, do school science fair project and talk to your class about what you have.’ Get them past that uncomfortable feeling of having to talk about it.”
The earlier this happens, the better. “We know from research that if you get them to be their own [health care] advocate, that’s one less thing they have to do in the adult health care system,” she said. “They will move on to other things, such as getting a job or going to college.”
Dr. Peacock, who is board certified in pediatrics and internal medicine, added that providing adolescents with the option of being seen by professionals without their parents is considered best practice. “You could start by introducing the concept at age 12, but say at age 13, ‘I want to spend a minute with you alone without your parent. I want you to bring in questions that you want to ask me that you may not want to ask in front of your parents,’” she said. “I guarantee you that on that third visit the adolescent will ask you a question.”
Optimistic messaging is another component of effective planning. “You may see someone in your office with a disease that you know has high mortality and high morbidity, and you’re trying to help the family cope,” Dr. Peacock said. “That young person needs to be asked, ‘What are your plans for your future?’ Think about it: In 10 or 20 years when you’re transitioning that individual out of your health care system, what medical miracles have happened?” She recalled visiting with the mother of a patient with Down syndrome who had significant congenital heart disease. He was in his 30s and struggled to keep his behavior in check. “We were trying to develop a behavior plan, but his past care team had never put one in place for him,” Dr. Peacock said. “The mother looked at me and said, accusingly, ‘It’s your fault. It’s all of your doctors’ faults because they never told me that this would happen. They told me to take him home and spoil him because he would not be around at this age.’”
Effective transition handoffs are collaborative, she continued, with care plans built around what is likely to happen with the patient over time. “At Baylor College of Medicine, pediatric dermatologists follow patients for life, but I take over everything else adult care related,” Dr. Peacock said. “The pediatric dermatologist has me come over to the hospital when we’re talking about quality-of-life issues – about advance care, advanced directives, those types of things.”
She recommends not transferring care to an adult provider during pregnancy, hospitalization, during active disease, or during changes to a patient’s medical therapy. “When pediatricians call me from the hospital and they want an urgent transfer, I tell them, ‘Your emergency is not my urgency. Get everything ready; get them discharged. Get them followed up and back on their chronic care management, and I’ll be happy to do that transition for you,’ ” she said. “It’s also good to leave that door open for the adult provider to call you. Give them your cell phone number because it may be just one question, like, ‘Can you tell me why his liver enzymes are elevated? We can’t figure it out.’ ”
Dr. Peacock advises pediatric providers to develop processes within their own practice that facilitates transfer, “even if it just means sharing information with the adult provider at the end of the time you’re seeing that young adult. Know your systems and your resources. Get that medical summary done, even if it’s making the patient do the medical summary.” More information for clinicians and for patients and their families can be found at www.gottransition.org.
She reported having no relevant financial disclosures.
AUSTIN, TEX. – All too often, children and adolescents stumble on their way to adult health care – if they make it at all.
In fact, data from an ongoing survey by the Department of Health & Human Services and the Health Resources and Services Administration indicate that only 40% of children with special health care needs aged 12-17 years receive the services necessary to make transitions to adult health care.
“Most fall off the proverbial cliff and do not engage with adult providers,” Cynthia Peacock, MD, said at the annual meeting of the Society for Pediatric Dermatology. She recalled meeting with one individual who, after being a patient at the children’s hospital for 13 years, was told over the phone to transition to an adult provider with not so much as a promised letter of introduction being sent on. “She did not know about the adult health care culture, which is fractured in care, relies on the patient to be their own advocate, and wants patients to be able to do their visit in 10-15 minutes.”
Dr. Peacock, medical director of the Transition Medicine Clinic at Baylor College of Medicine, Houston, described “Starting at age 12 is not too early,” she said. “It gives you time so that you can keep introducing the concept when that individual keeps coming back to see you, especially if it’s a chronic condition.”
She recommended that clinicians ask several questions to assess transition readiness of pediatric patients to adult care, including, Do you know your medications? Do you know how to take them? Do you know how to refill them? Do you know how to discuss them? Can you discuss your medical condition with the adult doctor? Can you call for a doctor’s appointment or get a prescription filled? “Adolescents are notorious for calling [the doctor’s office], and if they’re told they can’t make an appointment, that’s it; they stop right there,” Dr. Peacock said. “They don’t tend to problem solve. They don’t engage.”
Studies have suggested that the transfer of care is more likely to be successful if a formal transition program is in place to prepare the patient and to facilitate the change in health care providers. “There is a growing evidence base in the literature that skills training for young people with chronic illnesses can be associated with positive outcomes,” she said. “This can be as easy as telling the individual, ‘Do a book report about your condition. Talk to a friend. Tell a friend what your condition is. Or, do school science fair project and talk to your class about what you have.’ Get them past that uncomfortable feeling of having to talk about it.”
The earlier this happens, the better. “We know from research that if you get them to be their own [health care] advocate, that’s one less thing they have to do in the adult health care system,” she said. “They will move on to other things, such as getting a job or going to college.”
Dr. Peacock, who is board certified in pediatrics and internal medicine, added that providing adolescents with the option of being seen by professionals without their parents is considered best practice. “You could start by introducing the concept at age 12, but say at age 13, ‘I want to spend a minute with you alone without your parent. I want you to bring in questions that you want to ask me that you may not want to ask in front of your parents,’” she said. “I guarantee you that on that third visit the adolescent will ask you a question.”
Optimistic messaging is another component of effective planning. “You may see someone in your office with a disease that you know has high mortality and high morbidity, and you’re trying to help the family cope,” Dr. Peacock said. “That young person needs to be asked, ‘What are your plans for your future?’ Think about it: In 10 or 20 years when you’re transitioning that individual out of your health care system, what medical miracles have happened?” She recalled visiting with the mother of a patient with Down syndrome who had significant congenital heart disease. He was in his 30s and struggled to keep his behavior in check. “We were trying to develop a behavior plan, but his past care team had never put one in place for him,” Dr. Peacock said. “The mother looked at me and said, accusingly, ‘It’s your fault. It’s all of your doctors’ faults because they never told me that this would happen. They told me to take him home and spoil him because he would not be around at this age.’”
Effective transition handoffs are collaborative, she continued, with care plans built around what is likely to happen with the patient over time. “At Baylor College of Medicine, pediatric dermatologists follow patients for life, but I take over everything else adult care related,” Dr. Peacock said. “The pediatric dermatologist has me come over to the hospital when we’re talking about quality-of-life issues – about advance care, advanced directives, those types of things.”
She recommends not transferring care to an adult provider during pregnancy, hospitalization, during active disease, or during changes to a patient’s medical therapy. “When pediatricians call me from the hospital and they want an urgent transfer, I tell them, ‘Your emergency is not my urgency. Get everything ready; get them discharged. Get them followed up and back on their chronic care management, and I’ll be happy to do that transition for you,’ ” she said. “It’s also good to leave that door open for the adult provider to call you. Give them your cell phone number because it may be just one question, like, ‘Can you tell me why his liver enzymes are elevated? We can’t figure it out.’ ”
Dr. Peacock advises pediatric providers to develop processes within their own practice that facilitates transfer, “even if it just means sharing information with the adult provider at the end of the time you’re seeing that young adult. Know your systems and your resources. Get that medical summary done, even if it’s making the patient do the medical summary.” More information for clinicians and for patients and their families can be found at www.gottransition.org.
She reported having no relevant financial disclosures.
AUSTIN, TEX. – All too often, children and adolescents stumble on their way to adult health care – if they make it at all.
In fact, data from an ongoing survey by the Department of Health & Human Services and the Health Resources and Services Administration indicate that only 40% of children with special health care needs aged 12-17 years receive the services necessary to make transitions to adult health care.
“Most fall off the proverbial cliff and do not engage with adult providers,” Cynthia Peacock, MD, said at the annual meeting of the Society for Pediatric Dermatology. She recalled meeting with one individual who, after being a patient at the children’s hospital for 13 years, was told over the phone to transition to an adult provider with not so much as a promised letter of introduction being sent on. “She did not know about the adult health care culture, which is fractured in care, relies on the patient to be their own advocate, and wants patients to be able to do their visit in 10-15 minutes.”
Dr. Peacock, medical director of the Transition Medicine Clinic at Baylor College of Medicine, Houston, described “Starting at age 12 is not too early,” she said. “It gives you time so that you can keep introducing the concept when that individual keeps coming back to see you, especially if it’s a chronic condition.”
She recommended that clinicians ask several questions to assess transition readiness of pediatric patients to adult care, including, Do you know your medications? Do you know how to take them? Do you know how to refill them? Do you know how to discuss them? Can you discuss your medical condition with the adult doctor? Can you call for a doctor’s appointment or get a prescription filled? “Adolescents are notorious for calling [the doctor’s office], and if they’re told they can’t make an appointment, that’s it; they stop right there,” Dr. Peacock said. “They don’t tend to problem solve. They don’t engage.”
Studies have suggested that the transfer of care is more likely to be successful if a formal transition program is in place to prepare the patient and to facilitate the change in health care providers. “There is a growing evidence base in the literature that skills training for young people with chronic illnesses can be associated with positive outcomes,” she said. “This can be as easy as telling the individual, ‘Do a book report about your condition. Talk to a friend. Tell a friend what your condition is. Or, do school science fair project and talk to your class about what you have.’ Get them past that uncomfortable feeling of having to talk about it.”
The earlier this happens, the better. “We know from research that if you get them to be their own [health care] advocate, that’s one less thing they have to do in the adult health care system,” she said. “They will move on to other things, such as getting a job or going to college.”
Dr. Peacock, who is board certified in pediatrics and internal medicine, added that providing adolescents with the option of being seen by professionals without their parents is considered best practice. “You could start by introducing the concept at age 12, but say at age 13, ‘I want to spend a minute with you alone without your parent. I want you to bring in questions that you want to ask me that you may not want to ask in front of your parents,’” she said. “I guarantee you that on that third visit the adolescent will ask you a question.”
Optimistic messaging is another component of effective planning. “You may see someone in your office with a disease that you know has high mortality and high morbidity, and you’re trying to help the family cope,” Dr. Peacock said. “That young person needs to be asked, ‘What are your plans for your future?’ Think about it: In 10 or 20 years when you’re transitioning that individual out of your health care system, what medical miracles have happened?” She recalled visiting with the mother of a patient with Down syndrome who had significant congenital heart disease. He was in his 30s and struggled to keep his behavior in check. “We were trying to develop a behavior plan, but his past care team had never put one in place for him,” Dr. Peacock said. “The mother looked at me and said, accusingly, ‘It’s your fault. It’s all of your doctors’ faults because they never told me that this would happen. They told me to take him home and spoil him because he would not be around at this age.’”
Effective transition handoffs are collaborative, she continued, with care plans built around what is likely to happen with the patient over time. “At Baylor College of Medicine, pediatric dermatologists follow patients for life, but I take over everything else adult care related,” Dr. Peacock said. “The pediatric dermatologist has me come over to the hospital when we’re talking about quality-of-life issues – about advance care, advanced directives, those types of things.”
She recommends not transferring care to an adult provider during pregnancy, hospitalization, during active disease, or during changes to a patient’s medical therapy. “When pediatricians call me from the hospital and they want an urgent transfer, I tell them, ‘Your emergency is not my urgency. Get everything ready; get them discharged. Get them followed up and back on their chronic care management, and I’ll be happy to do that transition for you,’ ” she said. “It’s also good to leave that door open for the adult provider to call you. Give them your cell phone number because it may be just one question, like, ‘Can you tell me why his liver enzymes are elevated? We can’t figure it out.’ ”
Dr. Peacock advises pediatric providers to develop processes within their own practice that facilitates transfer, “even if it just means sharing information with the adult provider at the end of the time you’re seeing that young adult. Know your systems and your resources. Get that medical summary done, even if it’s making the patient do the medical summary.” More information for clinicians and for patients and their families can be found at www.gottransition.org.
She reported having no relevant financial disclosures.
EXPERT ANALYSIS FROM SPD 2019
Antiepileptic drug outcomes have remained flat for 3 decades
BANGKOK – Since founding the Epilepsy Unit at Glasgow’s Western Infirmary 37 years ago, Martin J. Brodie, MD, has seen many changes in the field, including the introduction of more than a dozen new antiepileptic drugs (AEDs) in the past 2 decades.
And based upon this vast clinical experience coupled with his leadership of landmark studies, he has a message for his physician colleagues and their epilepsy patients. And it’s not pretty.
“Has the probability of achieving seizure freedom increased significantly in the last 3 decades? Regrettably, the answer is no,” he declared at the International Epilepsy Congress.
“Over all these years, in terms of seizure freedom there has been no real difference in outcome. There’s really quite a long way to go before we can say that we are doing all that well for people,” he said at the congress sponsored by the International League Against Epilepsy.
In the year 2000, he and his coinvestigators published a prospective, longitudinal, observational cohort study of 470 newly diagnosed patients with epilepsy treated at the Western Infirmary during 1982-1997, all with a minimum of 2 years’ follow-up. Sixty-one percent achieved complete freedom from seizures for at least 1 year on monotherapy, and another 3% did so on polytherapy, for a total rate of 64% (N Engl J Med. 2000 Feb 3;342[5]:314-19).
But these were patients who by and large were treated with older AEDs such as carbamazepine, which has since fallen by the wayside because of toxicities. Scottish neurologists now generally turn to lamotrigine (Lamictal), levetiracetam (Spritam), and other, newer AEDs. So Dr. Brodie and his coworkers recently published a follow-up study, this one featuring 30 years of longitudinal follow-up of 1,795 patients newly treated for epilepsy with AEDs, new and old, during 1982-2012. The investigators demonstrated that the seizure-free survival curves over time were virtually superimposable. In the larger, more recent study, remission was achieved in 55% of patients with AED monotherapy and in another 9% with polytherapy, for a total rate of 64%, identical to the rate in the 2000 study, and as was the case in the earlier study, 36% of patients remained uncontrolled (JAMA Neurol. 2018 Mar 1;75[3]:279-86).
“Overall, the way this population behaves, there’s no difference in efficacy and no difference in tolerability whether you’re using old drugs used properly or new drugs used properly,” said Dr. Brodie, professor of neurology at the University of Glasgow (Scotland).
It’s noteworthy that Sir William R. Gowers, the Londoner who has been called the greatest neurologist of all time, reported a 70% seizure-free rate in 1881, while Dr. Brodie and workers achieved a 64% rate in their 30-year study. “It’s interesting that the numbers are so bad, really, I suppose,” Dr. Brodie commented.
How about outcomes in pediatric epilepsy?
Dr. Brodie and coworkers recently published a 30-year prospective cohort study of 332 adolescent epilepsy patients newly diagnosed and treated at the Western Infirmary during 1982-2012. At the end of the study, 67% were seizure-free for at least the past year, a feat accomplished via monotherapy in 83% of cases. The seizure-free rate was 72% in those with generalized epilepsy, significantly better than the 60% figure in those with focal epilepsy. The efficacy rate was 74% with newer AED monotherapy and similar at 77% with monotherapy older drugs. Adverse event rates ranged from a low of 12% with lamotrigine to 56% with topiramate (Topamax), according to the findings published in Epilepsia (2019 Jun;60[6]:1083-90).
Roughly similar outcomes have been reported from Norway in a study of 600 children with epilepsy, median age 7 years, with a median follow-up of 5.8 years that is considerably shorter than that in the Glasgow pediatric study. Overall, 59% of the Norwegian children remained seizure free for at least 1 year, 30% developed drug-resistant epilepsy, and 11% followed an intermediate remitting/relapsing course (Pediatrics. 2018 Jun. doi: 10.1542/peds.2017-4016).
Why the decades of flat pharmacologic outcomes?
The consistently suboptimal seizure-free outcomes obtained over the past 30 years shouldn’t really be surprising, according to Dr. Brodie.
“Although we think we have lots of mechanisms of action and lots of differences between the drugs, they’re arguably all antiseizure drugs and not antiepilepsy drugs. We don’t treat the whale; we treat the spout. We don’t treat what we cannot see; we treat what we can see, which is the seizures, but we’re not influencing the long-term outcome,” the neurologist explained.
The compelling case for early epilepsy surgery
Epilepsy surgery remains underutilized, according to Dr. Brodie and other experts.
The International League Against Epilepsy defines drug-resistant epilepsy as failure to achieve sustained seizure freedom after adequate trials of two tolerated and appropriately chosen and used AED schedules. Dr. Brodie’s work was influential in creating that definition because his data demonstrated the sharply diminishing returns of additional drug trials.
“When do we consider epilepsy surgery? Arguably, the earlier, the better. After two drugs have failed appropriately, I don’t think anybody in this room would argue about that, although people in some of the other rooms might,” he said at the congress.
Influential in his thinking on this score were the impressive results of an early study, the first-ever randomized trial of surgery for epilepsy. In 80 patients with a 21-year history of drug-refractory temporal lobe epilepsy who were randomized to surgery or 1 year of AED therapy, at 1 year of follow-up blinded epileptologists rated 58% of surgically treated patients as free from seizures that impair awareness of self and surroundings, compared with just 8% in the AED group (N Engl J Med. 2001 Aug 2;345[5]:311-8).
“That’s a big outcome, and I’m very keen to ensure that my data continue to drive the push for early surgery,” according to the neurologist.
A Cochrane review of 177 studies totaling more than 16,000 patients concluded that 65% of epilepsy patients had good outcomes following surgery. Prognostic factors associated with better surgical outcomes included complete surgical resection of the epileptogenic focus, the presence of mesial temporal sclerosis, concordance of MRI and EEG findings, and an absence of cortical dysplasia (Cochrane Database Syst Rev. 2019;6:CD010541. doi: 10.1002/14651858.CD010541.pub3).
In addition, a systematic review and meta-analysis by Canadian investigators found that 72% of adults with lesional epilepsy identified by MRI or histopathology were seizure-free after surgery, compared with 36% of those with nonlesional epilepsy. The disparity in outcomes was similar in pediatric epilepsy patients, with seizure freedom after surgery in 74% of those with lesional disease versus 45% with nonlesional epilepsy (Epilepsy Res. 2010 May;89[2-3]:310-8).
Whither are neurostimulatory device therapies headed?
Dr. Brodie was quick to admit that as a pharmacologic researcher, device modalities including vagus nerve stimulation, responsive neurostimulation, and deep brain stimulation are outside his area of expertise. But he’s been following developments in the field with interest.
“These device therapies have shown efficacy in short-term randomized trials, but very few patients attain long-term seizure freedom. I think these are largely palliative techniques. I gave up on these techniques a long time ago because I felt it was a very costly way of reducing seizures by a relatively small margin, and really we need to go a little bit further than that. But I know there’s a lot of work going on at the moment,” he said.
Dr. Brodie reported serving on the scientific advisory boards of more than a half dozen pharmaceutical companies.
BANGKOK – Since founding the Epilepsy Unit at Glasgow’s Western Infirmary 37 years ago, Martin J. Brodie, MD, has seen many changes in the field, including the introduction of more than a dozen new antiepileptic drugs (AEDs) in the past 2 decades.
And based upon this vast clinical experience coupled with his leadership of landmark studies, he has a message for his physician colleagues and their epilepsy patients. And it’s not pretty.
“Has the probability of achieving seizure freedom increased significantly in the last 3 decades? Regrettably, the answer is no,” he declared at the International Epilepsy Congress.
“Over all these years, in terms of seizure freedom there has been no real difference in outcome. There’s really quite a long way to go before we can say that we are doing all that well for people,” he said at the congress sponsored by the International League Against Epilepsy.
In the year 2000, he and his coinvestigators published a prospective, longitudinal, observational cohort study of 470 newly diagnosed patients with epilepsy treated at the Western Infirmary during 1982-1997, all with a minimum of 2 years’ follow-up. Sixty-one percent achieved complete freedom from seizures for at least 1 year on monotherapy, and another 3% did so on polytherapy, for a total rate of 64% (N Engl J Med. 2000 Feb 3;342[5]:314-19).
But these were patients who by and large were treated with older AEDs such as carbamazepine, which has since fallen by the wayside because of toxicities. Scottish neurologists now generally turn to lamotrigine (Lamictal), levetiracetam (Spritam), and other, newer AEDs. So Dr. Brodie and his coworkers recently published a follow-up study, this one featuring 30 years of longitudinal follow-up of 1,795 patients newly treated for epilepsy with AEDs, new and old, during 1982-2012. The investigators demonstrated that the seizure-free survival curves over time were virtually superimposable. In the larger, more recent study, remission was achieved in 55% of patients with AED monotherapy and in another 9% with polytherapy, for a total rate of 64%, identical to the rate in the 2000 study, and as was the case in the earlier study, 36% of patients remained uncontrolled (JAMA Neurol. 2018 Mar 1;75[3]:279-86).
“Overall, the way this population behaves, there’s no difference in efficacy and no difference in tolerability whether you’re using old drugs used properly or new drugs used properly,” said Dr. Brodie, professor of neurology at the University of Glasgow (Scotland).
It’s noteworthy that Sir William R. Gowers, the Londoner who has been called the greatest neurologist of all time, reported a 70% seizure-free rate in 1881, while Dr. Brodie and workers achieved a 64% rate in their 30-year study. “It’s interesting that the numbers are so bad, really, I suppose,” Dr. Brodie commented.
How about outcomes in pediatric epilepsy?
Dr. Brodie and coworkers recently published a 30-year prospective cohort study of 332 adolescent epilepsy patients newly diagnosed and treated at the Western Infirmary during 1982-2012. At the end of the study, 67% were seizure-free for at least the past year, a feat accomplished via monotherapy in 83% of cases. The seizure-free rate was 72% in those with generalized epilepsy, significantly better than the 60% figure in those with focal epilepsy. The efficacy rate was 74% with newer AED monotherapy and similar at 77% with monotherapy older drugs. Adverse event rates ranged from a low of 12% with lamotrigine to 56% with topiramate (Topamax), according to the findings published in Epilepsia (2019 Jun;60[6]:1083-90).
Roughly similar outcomes have been reported from Norway in a study of 600 children with epilepsy, median age 7 years, with a median follow-up of 5.8 years that is considerably shorter than that in the Glasgow pediatric study. Overall, 59% of the Norwegian children remained seizure free for at least 1 year, 30% developed drug-resistant epilepsy, and 11% followed an intermediate remitting/relapsing course (Pediatrics. 2018 Jun. doi: 10.1542/peds.2017-4016).
Why the decades of flat pharmacologic outcomes?
The consistently suboptimal seizure-free outcomes obtained over the past 30 years shouldn’t really be surprising, according to Dr. Brodie.
“Although we think we have lots of mechanisms of action and lots of differences between the drugs, they’re arguably all antiseizure drugs and not antiepilepsy drugs. We don’t treat the whale; we treat the spout. We don’t treat what we cannot see; we treat what we can see, which is the seizures, but we’re not influencing the long-term outcome,” the neurologist explained.
The compelling case for early epilepsy surgery
Epilepsy surgery remains underutilized, according to Dr. Brodie and other experts.
The International League Against Epilepsy defines drug-resistant epilepsy as failure to achieve sustained seizure freedom after adequate trials of two tolerated and appropriately chosen and used AED schedules. Dr. Brodie’s work was influential in creating that definition because his data demonstrated the sharply diminishing returns of additional drug trials.
“When do we consider epilepsy surgery? Arguably, the earlier, the better. After two drugs have failed appropriately, I don’t think anybody in this room would argue about that, although people in some of the other rooms might,” he said at the congress.
Influential in his thinking on this score were the impressive results of an early study, the first-ever randomized trial of surgery for epilepsy. In 80 patients with a 21-year history of drug-refractory temporal lobe epilepsy who were randomized to surgery or 1 year of AED therapy, at 1 year of follow-up blinded epileptologists rated 58% of surgically treated patients as free from seizures that impair awareness of self and surroundings, compared with just 8% in the AED group (N Engl J Med. 2001 Aug 2;345[5]:311-8).
“That’s a big outcome, and I’m very keen to ensure that my data continue to drive the push for early surgery,” according to the neurologist.
A Cochrane review of 177 studies totaling more than 16,000 patients concluded that 65% of epilepsy patients had good outcomes following surgery. Prognostic factors associated with better surgical outcomes included complete surgical resection of the epileptogenic focus, the presence of mesial temporal sclerosis, concordance of MRI and EEG findings, and an absence of cortical dysplasia (Cochrane Database Syst Rev. 2019;6:CD010541. doi: 10.1002/14651858.CD010541.pub3).
In addition, a systematic review and meta-analysis by Canadian investigators found that 72% of adults with lesional epilepsy identified by MRI or histopathology were seizure-free after surgery, compared with 36% of those with nonlesional epilepsy. The disparity in outcomes was similar in pediatric epilepsy patients, with seizure freedom after surgery in 74% of those with lesional disease versus 45% with nonlesional epilepsy (Epilepsy Res. 2010 May;89[2-3]:310-8).
Whither are neurostimulatory device therapies headed?
Dr. Brodie was quick to admit that as a pharmacologic researcher, device modalities including vagus nerve stimulation, responsive neurostimulation, and deep brain stimulation are outside his area of expertise. But he’s been following developments in the field with interest.
“These device therapies have shown efficacy in short-term randomized trials, but very few patients attain long-term seizure freedom. I think these are largely palliative techniques. I gave up on these techniques a long time ago because I felt it was a very costly way of reducing seizures by a relatively small margin, and really we need to go a little bit further than that. But I know there’s a lot of work going on at the moment,” he said.
Dr. Brodie reported serving on the scientific advisory boards of more than a half dozen pharmaceutical companies.
BANGKOK – Since founding the Epilepsy Unit at Glasgow’s Western Infirmary 37 years ago, Martin J. Brodie, MD, has seen many changes in the field, including the introduction of more than a dozen new antiepileptic drugs (AEDs) in the past 2 decades.
And based upon this vast clinical experience coupled with his leadership of landmark studies, he has a message for his physician colleagues and their epilepsy patients. And it’s not pretty.
“Has the probability of achieving seizure freedom increased significantly in the last 3 decades? Regrettably, the answer is no,” he declared at the International Epilepsy Congress.
“Over all these years, in terms of seizure freedom there has been no real difference in outcome. There’s really quite a long way to go before we can say that we are doing all that well for people,” he said at the congress sponsored by the International League Against Epilepsy.
In the year 2000, he and his coinvestigators published a prospective, longitudinal, observational cohort study of 470 newly diagnosed patients with epilepsy treated at the Western Infirmary during 1982-1997, all with a minimum of 2 years’ follow-up. Sixty-one percent achieved complete freedom from seizures for at least 1 year on monotherapy, and another 3% did so on polytherapy, for a total rate of 64% (N Engl J Med. 2000 Feb 3;342[5]:314-19).
But these were patients who by and large were treated with older AEDs such as carbamazepine, which has since fallen by the wayside because of toxicities. Scottish neurologists now generally turn to lamotrigine (Lamictal), levetiracetam (Spritam), and other, newer AEDs. So Dr. Brodie and his coworkers recently published a follow-up study, this one featuring 30 years of longitudinal follow-up of 1,795 patients newly treated for epilepsy with AEDs, new and old, during 1982-2012. The investigators demonstrated that the seizure-free survival curves over time were virtually superimposable. In the larger, more recent study, remission was achieved in 55% of patients with AED monotherapy and in another 9% with polytherapy, for a total rate of 64%, identical to the rate in the 2000 study, and as was the case in the earlier study, 36% of patients remained uncontrolled (JAMA Neurol. 2018 Mar 1;75[3]:279-86).
“Overall, the way this population behaves, there’s no difference in efficacy and no difference in tolerability whether you’re using old drugs used properly or new drugs used properly,” said Dr. Brodie, professor of neurology at the University of Glasgow (Scotland).
It’s noteworthy that Sir William R. Gowers, the Londoner who has been called the greatest neurologist of all time, reported a 70% seizure-free rate in 1881, while Dr. Brodie and workers achieved a 64% rate in their 30-year study. “It’s interesting that the numbers are so bad, really, I suppose,” Dr. Brodie commented.
How about outcomes in pediatric epilepsy?
Dr. Brodie and coworkers recently published a 30-year prospective cohort study of 332 adolescent epilepsy patients newly diagnosed and treated at the Western Infirmary during 1982-2012. At the end of the study, 67% were seizure-free for at least the past year, a feat accomplished via monotherapy in 83% of cases. The seizure-free rate was 72% in those with generalized epilepsy, significantly better than the 60% figure in those with focal epilepsy. The efficacy rate was 74% with newer AED monotherapy and similar at 77% with monotherapy older drugs. Adverse event rates ranged from a low of 12% with lamotrigine to 56% with topiramate (Topamax), according to the findings published in Epilepsia (2019 Jun;60[6]:1083-90).
Roughly similar outcomes have been reported from Norway in a study of 600 children with epilepsy, median age 7 years, with a median follow-up of 5.8 years that is considerably shorter than that in the Glasgow pediatric study. Overall, 59% of the Norwegian children remained seizure free for at least 1 year, 30% developed drug-resistant epilepsy, and 11% followed an intermediate remitting/relapsing course (Pediatrics. 2018 Jun. doi: 10.1542/peds.2017-4016).
Why the decades of flat pharmacologic outcomes?
The consistently suboptimal seizure-free outcomes obtained over the past 30 years shouldn’t really be surprising, according to Dr. Brodie.
“Although we think we have lots of mechanisms of action and lots of differences between the drugs, they’re arguably all antiseizure drugs and not antiepilepsy drugs. We don’t treat the whale; we treat the spout. We don’t treat what we cannot see; we treat what we can see, which is the seizures, but we’re not influencing the long-term outcome,” the neurologist explained.
The compelling case for early epilepsy surgery
Epilepsy surgery remains underutilized, according to Dr. Brodie and other experts.
The International League Against Epilepsy defines drug-resistant epilepsy as failure to achieve sustained seizure freedom after adequate trials of two tolerated and appropriately chosen and used AED schedules. Dr. Brodie’s work was influential in creating that definition because his data demonstrated the sharply diminishing returns of additional drug trials.
“When do we consider epilepsy surgery? Arguably, the earlier, the better. After two drugs have failed appropriately, I don’t think anybody in this room would argue about that, although people in some of the other rooms might,” he said at the congress.
Influential in his thinking on this score were the impressive results of an early study, the first-ever randomized trial of surgery for epilepsy. In 80 patients with a 21-year history of drug-refractory temporal lobe epilepsy who were randomized to surgery or 1 year of AED therapy, at 1 year of follow-up blinded epileptologists rated 58% of surgically treated patients as free from seizures that impair awareness of self and surroundings, compared with just 8% in the AED group (N Engl J Med. 2001 Aug 2;345[5]:311-8).
“That’s a big outcome, and I’m very keen to ensure that my data continue to drive the push for early surgery,” according to the neurologist.
A Cochrane review of 177 studies totaling more than 16,000 patients concluded that 65% of epilepsy patients had good outcomes following surgery. Prognostic factors associated with better surgical outcomes included complete surgical resection of the epileptogenic focus, the presence of mesial temporal sclerosis, concordance of MRI and EEG findings, and an absence of cortical dysplasia (Cochrane Database Syst Rev. 2019;6:CD010541. doi: 10.1002/14651858.CD010541.pub3).
In addition, a systematic review and meta-analysis by Canadian investigators found that 72% of adults with lesional epilepsy identified by MRI or histopathology were seizure-free after surgery, compared with 36% of those with nonlesional epilepsy. The disparity in outcomes was similar in pediatric epilepsy patients, with seizure freedom after surgery in 74% of those with lesional disease versus 45% with nonlesional epilepsy (Epilepsy Res. 2010 May;89[2-3]:310-8).
Whither are neurostimulatory device therapies headed?
Dr. Brodie was quick to admit that as a pharmacologic researcher, device modalities including vagus nerve stimulation, responsive neurostimulation, and deep brain stimulation are outside his area of expertise. But he’s been following developments in the field with interest.
“These device therapies have shown efficacy in short-term randomized trials, but very few patients attain long-term seizure freedom. I think these are largely palliative techniques. I gave up on these techniques a long time ago because I felt it was a very costly way of reducing seizures by a relatively small margin, and really we need to go a little bit further than that. But I know there’s a lot of work going on at the moment,” he said.
Dr. Brodie reported serving on the scientific advisory boards of more than a half dozen pharmaceutical companies.
EXPERT ANALYSIS FROM IEC 2019
Too many blood cultures ordered for pediatric SSTIs
SEATTLE – Blood cultures were ordered for over half of pediatric skin infection encounters across 38 children’s hospitals, with rates varying from about 20% to 80% between hospitals, according to a review of almost 50,000 encounters in the Pediatric Health Information System database.
It was a surprising finding, because current guidelines from the Infectious Diseases Society of America do not recommend blood cultures as part of the routine evaluation of uncomplicated pediatric skin and soft-tissue infections (SSTIs), meaning infections in children who are otherwise healthy without neutropenia or other complicating factors.
Just 0.6% of the cultures were positive in the review, and it’s likely some of those were caused by contamination. After adjustment for demographics, complex chronic conditions, and severity of illness, culture draws were associated with a 20% increase in hospital length of stay (LOS), hospital costs, and 30-day readmission rates.
“Our data provide more evidence that [routine] blood cultures for children with SSTI represents low-value practice and should be avoided,” said lead investigator John Stephens, MD, a pediatrics professor and hospitalist at the University of North Carolina at Chapel Hill.
Dr. Stephens became curious about how common the practice was across hospitals after he and a friend penned an article about the issue for the Journal of Hospital Medicine’s “Things We Do for No Reason” series. The single-center studies they reviewed showed similarly high rates of both testing and negative cultures (J Hosp Med. 2018 Jul;13[7]:496-9).
Dr. Stephens and his team queried the Pediatric Health Information System database for encounters in children aged 2 months to 18 years with the diagnostic code 383, “cellulitis and other skin infections,” from 2012 to 2017, during which time “there really wasn’t a change” in IDSA guidance, he noted. Transfers, encounters with ICU care, and immunocompromised children were excluded.
Hospital admissions were included in the review if they had an additional code for erysipelas, cellulitis, impetigo, or other localized skin infection. The rate of positive cultures was inferred from subsequent codes for bacteremia or septicemia.
Across 49,291 encounters, the median rate of blood culture for skin infection was 51.6%, with tremendous variation between hospitals. With blood cultures, the hospital LOS was about 1.9 days, the hospital cost was $4,030, and the 30-day readmission rate was 1.3%. Without cultures, LOS was 1.6 days, the cost was $3,291, and the readmission rate was 1%.
Although infrequent, it’s likely that positive cultures triggered additional work-up, time in the hospital, and other measures, which might help account for the increase in LOS and costs.
As for why blood testing was so common, especially in some hospitals, “I think it’s just institutional culture. No amount of clinical variation in patient population could explain” a 20%-80% “variation across hospitals. It’s really just ingrained habits,” Dr. Stephens said at Pediatric Hospital Medicine.
“The rate of positive blood culture was really low, and the association was for higher cost and utilization. I think this really reinforces the IDSA guidelines. We need to focus on quality improvement efforts to do this better,” he said, noting that he hopes to do so at his own institution.
“I’d also like to know more on the positives. In the single center studies, we know more than half of them are contaminants. Often, there’s more contamination than true positives,” he said at the meeting sponsored by the Society of Hospital Medicine, the American Academy of Pediatrics, and the Academic Pediatric Association.
Instead of routine blood culture, Dr. Stephens recommended in his article to send pus for a Gram stain and culture and sensitivity, while noting that blood cultures remain reasonable for complicated infections, immunocompromised patients, and neonates.
There was no external funding, and Dr. Stephens didn’t report any disclosures.
SEATTLE – Blood cultures were ordered for over half of pediatric skin infection encounters across 38 children’s hospitals, with rates varying from about 20% to 80% between hospitals, according to a review of almost 50,000 encounters in the Pediatric Health Information System database.
It was a surprising finding, because current guidelines from the Infectious Diseases Society of America do not recommend blood cultures as part of the routine evaluation of uncomplicated pediatric skin and soft-tissue infections (SSTIs), meaning infections in children who are otherwise healthy without neutropenia or other complicating factors.
Just 0.6% of the cultures were positive in the review, and it’s likely some of those were caused by contamination. After adjustment for demographics, complex chronic conditions, and severity of illness, culture draws were associated with a 20% increase in hospital length of stay (LOS), hospital costs, and 30-day readmission rates.
“Our data provide more evidence that [routine] blood cultures for children with SSTI represents low-value practice and should be avoided,” said lead investigator John Stephens, MD, a pediatrics professor and hospitalist at the University of North Carolina at Chapel Hill.
Dr. Stephens became curious about how common the practice was across hospitals after he and a friend penned an article about the issue for the Journal of Hospital Medicine’s “Things We Do for No Reason” series. The single-center studies they reviewed showed similarly high rates of both testing and negative cultures (J Hosp Med. 2018 Jul;13[7]:496-9).
Dr. Stephens and his team queried the Pediatric Health Information System database for encounters in children aged 2 months to 18 years with the diagnostic code 383, “cellulitis and other skin infections,” from 2012 to 2017, during which time “there really wasn’t a change” in IDSA guidance, he noted. Transfers, encounters with ICU care, and immunocompromised children were excluded.
Hospital admissions were included in the review if they had an additional code for erysipelas, cellulitis, impetigo, or other localized skin infection. The rate of positive cultures was inferred from subsequent codes for bacteremia or septicemia.
Across 49,291 encounters, the median rate of blood culture for skin infection was 51.6%, with tremendous variation between hospitals. With blood cultures, the hospital LOS was about 1.9 days, the hospital cost was $4,030, and the 30-day readmission rate was 1.3%. Without cultures, LOS was 1.6 days, the cost was $3,291, and the readmission rate was 1%.
Although infrequent, it’s likely that positive cultures triggered additional work-up, time in the hospital, and other measures, which might help account for the increase in LOS and costs.
As for why blood testing was so common, especially in some hospitals, “I think it’s just institutional culture. No amount of clinical variation in patient population could explain” a 20%-80% “variation across hospitals. It’s really just ingrained habits,” Dr. Stephens said at Pediatric Hospital Medicine.
“The rate of positive blood culture was really low, and the association was for higher cost and utilization. I think this really reinforces the IDSA guidelines. We need to focus on quality improvement efforts to do this better,” he said, noting that he hopes to do so at his own institution.
“I’d also like to know more on the positives. In the single center studies, we know more than half of them are contaminants. Often, there’s more contamination than true positives,” he said at the meeting sponsored by the Society of Hospital Medicine, the American Academy of Pediatrics, and the Academic Pediatric Association.
Instead of routine blood culture, Dr. Stephens recommended in his article to send pus for a Gram stain and culture and sensitivity, while noting that blood cultures remain reasonable for complicated infections, immunocompromised patients, and neonates.
There was no external funding, and Dr. Stephens didn’t report any disclosures.
SEATTLE – Blood cultures were ordered for over half of pediatric skin infection encounters across 38 children’s hospitals, with rates varying from about 20% to 80% between hospitals, according to a review of almost 50,000 encounters in the Pediatric Health Information System database.
It was a surprising finding, because current guidelines from the Infectious Diseases Society of America do not recommend blood cultures as part of the routine evaluation of uncomplicated pediatric skin and soft-tissue infections (SSTIs), meaning infections in children who are otherwise healthy without neutropenia or other complicating factors.
Just 0.6% of the cultures were positive in the review, and it’s likely some of those were caused by contamination. After adjustment for demographics, complex chronic conditions, and severity of illness, culture draws were associated with a 20% increase in hospital length of stay (LOS), hospital costs, and 30-day readmission rates.
“Our data provide more evidence that [routine] blood cultures for children with SSTI represents low-value practice and should be avoided,” said lead investigator John Stephens, MD, a pediatrics professor and hospitalist at the University of North Carolina at Chapel Hill.
Dr. Stephens became curious about how common the practice was across hospitals after he and a friend penned an article about the issue for the Journal of Hospital Medicine’s “Things We Do for No Reason” series. The single-center studies they reviewed showed similarly high rates of both testing and negative cultures (J Hosp Med. 2018 Jul;13[7]:496-9).
Dr. Stephens and his team queried the Pediatric Health Information System database for encounters in children aged 2 months to 18 years with the diagnostic code 383, “cellulitis and other skin infections,” from 2012 to 2017, during which time “there really wasn’t a change” in IDSA guidance, he noted. Transfers, encounters with ICU care, and immunocompromised children were excluded.
Hospital admissions were included in the review if they had an additional code for erysipelas, cellulitis, impetigo, or other localized skin infection. The rate of positive cultures was inferred from subsequent codes for bacteremia or septicemia.
Across 49,291 encounters, the median rate of blood culture for skin infection was 51.6%, with tremendous variation between hospitals. With blood cultures, the hospital LOS was about 1.9 days, the hospital cost was $4,030, and the 30-day readmission rate was 1.3%. Without cultures, LOS was 1.6 days, the cost was $3,291, and the readmission rate was 1%.
Although infrequent, it’s likely that positive cultures triggered additional work-up, time in the hospital, and other measures, which might help account for the increase in LOS and costs.
As for why blood testing was so common, especially in some hospitals, “I think it’s just institutional culture. No amount of clinical variation in patient population could explain” a 20%-80% “variation across hospitals. It’s really just ingrained habits,” Dr. Stephens said at Pediatric Hospital Medicine.
“The rate of positive blood culture was really low, and the association was for higher cost and utilization. I think this really reinforces the IDSA guidelines. We need to focus on quality improvement efforts to do this better,” he said, noting that he hopes to do so at his own institution.
“I’d also like to know more on the positives. In the single center studies, we know more than half of them are contaminants. Often, there’s more contamination than true positives,” he said at the meeting sponsored by the Society of Hospital Medicine, the American Academy of Pediatrics, and the Academic Pediatric Association.
Instead of routine blood culture, Dr. Stephens recommended in his article to send pus for a Gram stain and culture and sensitivity, while noting that blood cultures remain reasonable for complicated infections, immunocompromised patients, and neonates.
There was no external funding, and Dr. Stephens didn’t report any disclosures.
REPORTING FROM PHM 2019
Enteral feeding is safe during bronchiolitis HFNC
SEATTLE – There were no cases of aspiration with enteric feeds of 60 children aged up to 2 years on high flow nasal cannula (HFNC) for bronchiolitis at the University of Oklahoma Children’s Hospital, Oklahoma City, according to research presented at the 2019 Pediatric Hospital Medicine Conference.
HFNC has become common for bronchiolitis management; it often saves infants from intubation. However, many providers opt for total parenteral nutrition during therapy instead of enteral feeding because of concerns about aspiration pneumonia.
Pediatricians at the children’s hospital began to wonder if the concern was really necessary. There have been reports of safe feeding during HFNC, and “clinical care literature has shown that feeding the gut throughout illness improves outcomes,” said lead investigator, Sarah Walter, MD, a third-year pediatrics resident at the hospital.
So her team took a leap of faith. They consulted the HFNC literature, asked their fellow providers what they would be comfortable with, and instituted a pediatric HFNC enteral feeding protocol at the children’s hospital for use on inpatient floors, pediatric ICUs, and elsewhere.
Feedings – formula or breast milk – are triggered by stable respiratory Tal scores over 8 hours, meaning that respiratory rates, breath sounds, and accessory muscle use were stable or improving. Children on a flow of 6 L/min or less, with a respiratory rate below 60 breaths per minute, are started on oral feeds, and those on higher flows on nasogastric (NG) tube feeds.
Feeds are started at 1 mL/kg per hour and advanced by the same amount every 3 hours until volume goals are reached; IV fluids are tapered accordingly. It’s a standing order, so nurses are able to initiate and advance feeding as indicated, any time of day.
Feeding was temporarily suspended in only 17 children: 6 for emesis, 6 for worsening respiratory scores, and the rest for dislodged NG tubes, procedures, or other issues. Enteric feeds were restarted with two stable scores below 7 points, at half the rate at which they were stopped.
NG tubes were used in over half of the 478 nursing shifts during which the 60 children – the majority aged 4-24 months – were fed; oral feeds in more than a third; and gastric tubes and other options in the rest. IV nutrition was used during just 1.8% of the shifts.
Enteric feeds were given up to a flow rate of 3.5 L/kg. There were no aspirations, even when children vomited. “We have seen good results so far that feeding is safe in these children,” Dr. Walters said.
“Our hospitalist team has been very receptive; they have been using the order set pretty continuously.” Parents also feel better when they know their children were “getting food in their belly,” even if by NG tube. “It’s important for family satisfaction,” she said.
The next step is to assess impact on length of stay, and education efforts to encourage broader use of the order set.
There was no external funding, and Dr. Walter had no disclosures. The meeting was sponsored by the Society of Hospital Medicine, the American Academy of Pediatrics, and the Academic Pediatric Association.
SEATTLE – There were no cases of aspiration with enteric feeds of 60 children aged up to 2 years on high flow nasal cannula (HFNC) for bronchiolitis at the University of Oklahoma Children’s Hospital, Oklahoma City, according to research presented at the 2019 Pediatric Hospital Medicine Conference.
HFNC has become common for bronchiolitis management; it often saves infants from intubation. However, many providers opt for total parenteral nutrition during therapy instead of enteral feeding because of concerns about aspiration pneumonia.
Pediatricians at the children’s hospital began to wonder if the concern was really necessary. There have been reports of safe feeding during HFNC, and “clinical care literature has shown that feeding the gut throughout illness improves outcomes,” said lead investigator, Sarah Walter, MD, a third-year pediatrics resident at the hospital.
So her team took a leap of faith. They consulted the HFNC literature, asked their fellow providers what they would be comfortable with, and instituted a pediatric HFNC enteral feeding protocol at the children’s hospital for use on inpatient floors, pediatric ICUs, and elsewhere.
Feedings – formula or breast milk – are triggered by stable respiratory Tal scores over 8 hours, meaning that respiratory rates, breath sounds, and accessory muscle use were stable or improving. Children on a flow of 6 L/min or less, with a respiratory rate below 60 breaths per minute, are started on oral feeds, and those on higher flows on nasogastric (NG) tube feeds.
Feeds are started at 1 mL/kg per hour and advanced by the same amount every 3 hours until volume goals are reached; IV fluids are tapered accordingly. It’s a standing order, so nurses are able to initiate and advance feeding as indicated, any time of day.
Feeding was temporarily suspended in only 17 children: 6 for emesis, 6 for worsening respiratory scores, and the rest for dislodged NG tubes, procedures, or other issues. Enteric feeds were restarted with two stable scores below 7 points, at half the rate at which they were stopped.
NG tubes were used in over half of the 478 nursing shifts during which the 60 children – the majority aged 4-24 months – were fed; oral feeds in more than a third; and gastric tubes and other options in the rest. IV nutrition was used during just 1.8% of the shifts.
Enteric feeds were given up to a flow rate of 3.5 L/kg. There were no aspirations, even when children vomited. “We have seen good results so far that feeding is safe in these children,” Dr. Walters said.
“Our hospitalist team has been very receptive; they have been using the order set pretty continuously.” Parents also feel better when they know their children were “getting food in their belly,” even if by NG tube. “It’s important for family satisfaction,” she said.
The next step is to assess impact on length of stay, and education efforts to encourage broader use of the order set.
There was no external funding, and Dr. Walter had no disclosures. The meeting was sponsored by the Society of Hospital Medicine, the American Academy of Pediatrics, and the Academic Pediatric Association.
SEATTLE – There were no cases of aspiration with enteric feeds of 60 children aged up to 2 years on high flow nasal cannula (HFNC) for bronchiolitis at the University of Oklahoma Children’s Hospital, Oklahoma City, according to research presented at the 2019 Pediatric Hospital Medicine Conference.
HFNC has become common for bronchiolitis management; it often saves infants from intubation. However, many providers opt for total parenteral nutrition during therapy instead of enteral feeding because of concerns about aspiration pneumonia.
Pediatricians at the children’s hospital began to wonder if the concern was really necessary. There have been reports of safe feeding during HFNC, and “clinical care literature has shown that feeding the gut throughout illness improves outcomes,” said lead investigator, Sarah Walter, MD, a third-year pediatrics resident at the hospital.
So her team took a leap of faith. They consulted the HFNC literature, asked their fellow providers what they would be comfortable with, and instituted a pediatric HFNC enteral feeding protocol at the children’s hospital for use on inpatient floors, pediatric ICUs, and elsewhere.
Feedings – formula or breast milk – are triggered by stable respiratory Tal scores over 8 hours, meaning that respiratory rates, breath sounds, and accessory muscle use were stable or improving. Children on a flow of 6 L/min or less, with a respiratory rate below 60 breaths per minute, are started on oral feeds, and those on higher flows on nasogastric (NG) tube feeds.
Feeds are started at 1 mL/kg per hour and advanced by the same amount every 3 hours until volume goals are reached; IV fluids are tapered accordingly. It’s a standing order, so nurses are able to initiate and advance feeding as indicated, any time of day.
Feeding was temporarily suspended in only 17 children: 6 for emesis, 6 for worsening respiratory scores, and the rest for dislodged NG tubes, procedures, or other issues. Enteric feeds were restarted with two stable scores below 7 points, at half the rate at which they were stopped.
NG tubes were used in over half of the 478 nursing shifts during which the 60 children – the majority aged 4-24 months – were fed; oral feeds in more than a third; and gastric tubes and other options in the rest. IV nutrition was used during just 1.8% of the shifts.
Enteric feeds were given up to a flow rate of 3.5 L/kg. There were no aspirations, even when children vomited. “We have seen good results so far that feeding is safe in these children,” Dr. Walters said.
“Our hospitalist team has been very receptive; they have been using the order set pretty continuously.” Parents also feel better when they know their children were “getting food in their belly,” even if by NG tube. “It’s important for family satisfaction,” she said.
The next step is to assess impact on length of stay, and education efforts to encourage broader use of the order set.
There was no external funding, and Dr. Walter had no disclosures. The meeting was sponsored by the Society of Hospital Medicine, the American Academy of Pediatrics, and the Academic Pediatric Association.
REPORTING FROM PHM 2019
U.S. infant mortality continued slow decline in 2017
according to data released Aug. 1 by the National Center for Health Statistics, based on data from the National Vital Statistics System.
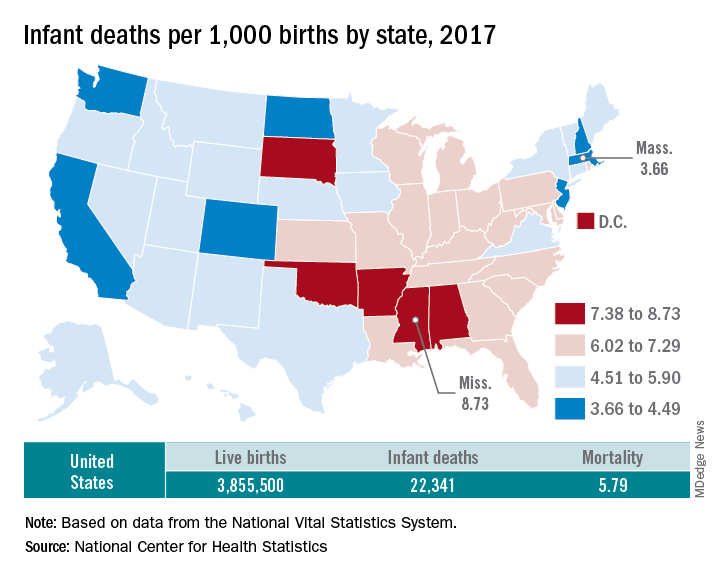
The rate for 2017 was 5.79 deaths per 1,000 live births, which was not statistically different from the rate of 5.87 in 2016, the National Center for Health Statistics said in a new report. Neonatal and postneonatal mortality – 3.85 and 1.94 per 1,000, respectively – both showed the same nonsignificant drop from 2016 to 2017.
About two-thirds of the infants who died in 2017 were children born preterm (less than 37 weeks’ gestation), the NCHS said, and “the mortality rate for infants born before 28 weeks of gestation [389.4 per 1,000] was 183 times the rate for term infants” born at 37-41 weeks.
Rates at the state level in 2017 ranged from a low of 3.66 deaths/1,000 live births in Massachusetts to a high of 8.73/1,000 in Mississippi. Washington (3.88) was the only other state with a rate below 4.0, while Arkansas (8.10) was the only other state above 8.0 (The District of Columbia had a rate of 8.16.). Infant mortality was significantly lower than the national rate in 11 states and significantly higher in 15 states and D.C., according to the report.
Overall, in 2017, 3,855,500 live births occurred, with 22,341 infants having died before the age of 1 year, data from the National Vital Statistics System’s linked birth/infant death file show. In 1995, the first year that the linked file was available, the corresponding numbers were 3,899,589 births and 29,505 deaths, for a rate of 7.57 deaths/1,000 live births.
according to data released Aug. 1 by the National Center for Health Statistics, based on data from the National Vital Statistics System.

The rate for 2017 was 5.79 deaths per 1,000 live births, which was not statistically different from the rate of 5.87 in 2016, the National Center for Health Statistics said in a new report. Neonatal and postneonatal mortality – 3.85 and 1.94 per 1,000, respectively – both showed the same nonsignificant drop from 2016 to 2017.
About two-thirds of the infants who died in 2017 were children born preterm (less than 37 weeks’ gestation), the NCHS said, and “the mortality rate for infants born before 28 weeks of gestation [389.4 per 1,000] was 183 times the rate for term infants” born at 37-41 weeks.
Rates at the state level in 2017 ranged from a low of 3.66 deaths/1,000 live births in Massachusetts to a high of 8.73/1,000 in Mississippi. Washington (3.88) was the only other state with a rate below 4.0, while Arkansas (8.10) was the only other state above 8.0 (The District of Columbia had a rate of 8.16.). Infant mortality was significantly lower than the national rate in 11 states and significantly higher in 15 states and D.C., according to the report.
Overall, in 2017, 3,855,500 live births occurred, with 22,341 infants having died before the age of 1 year, data from the National Vital Statistics System’s linked birth/infant death file show. In 1995, the first year that the linked file was available, the corresponding numbers were 3,899,589 births and 29,505 deaths, for a rate of 7.57 deaths/1,000 live births.
according to data released Aug. 1 by the National Center for Health Statistics, based on data from the National Vital Statistics System.

The rate for 2017 was 5.79 deaths per 1,000 live births, which was not statistically different from the rate of 5.87 in 2016, the National Center for Health Statistics said in a new report. Neonatal and postneonatal mortality – 3.85 and 1.94 per 1,000, respectively – both showed the same nonsignificant drop from 2016 to 2017.
About two-thirds of the infants who died in 2017 were children born preterm (less than 37 weeks’ gestation), the NCHS said, and “the mortality rate for infants born before 28 weeks of gestation [389.4 per 1,000] was 183 times the rate for term infants” born at 37-41 weeks.
Rates at the state level in 2017 ranged from a low of 3.66 deaths/1,000 live births in Massachusetts to a high of 8.73/1,000 in Mississippi. Washington (3.88) was the only other state with a rate below 4.0, while Arkansas (8.10) was the only other state above 8.0 (The District of Columbia had a rate of 8.16.). Infant mortality was significantly lower than the national rate in 11 states and significantly higher in 15 states and D.C., according to the report.
Overall, in 2017, 3,855,500 live births occurred, with 22,341 infants having died before the age of 1 year, data from the National Vital Statistics System’s linked birth/infant death file show. In 1995, the first year that the linked file was available, the corresponding numbers were 3,899,589 births and 29,505 deaths, for a rate of 7.57 deaths/1,000 live births.
Strategies for improving ADHD medication adherence
Attention-deficit/hyperactivity disorder (ADHD) is the most common childhood neurodevelopmental disorder, affecting 8% to 12% of school-aged children in the United States1-3 with significant impairments that often persist into adulthood.4-8 Current guidelines recommend stimulant medication and/or behavioral therapies as first-line treatments for ADHD.9,10 There is a wealth of evidence on the efficacy of stimulants in ADHD, with the most significant effects noted on core ADHD symptoms.11,12 Additional evidence links stimulants to decreased long-term negative outcomes, including reduced school absences and grade retention,13 as well as modestly but significantly improved reading and math scores.14 Other studies have reported that individuals with ADHD who receive medication have decreased criminality,15,16 motor vehicle accidents,17,18 injuries,19 substance abuse,20-22 and risk for subsequent and concurrent depression.23 Therefore, the evidence suggests that consistent medication treatment helps improve outcomes for individuals with ADHD.
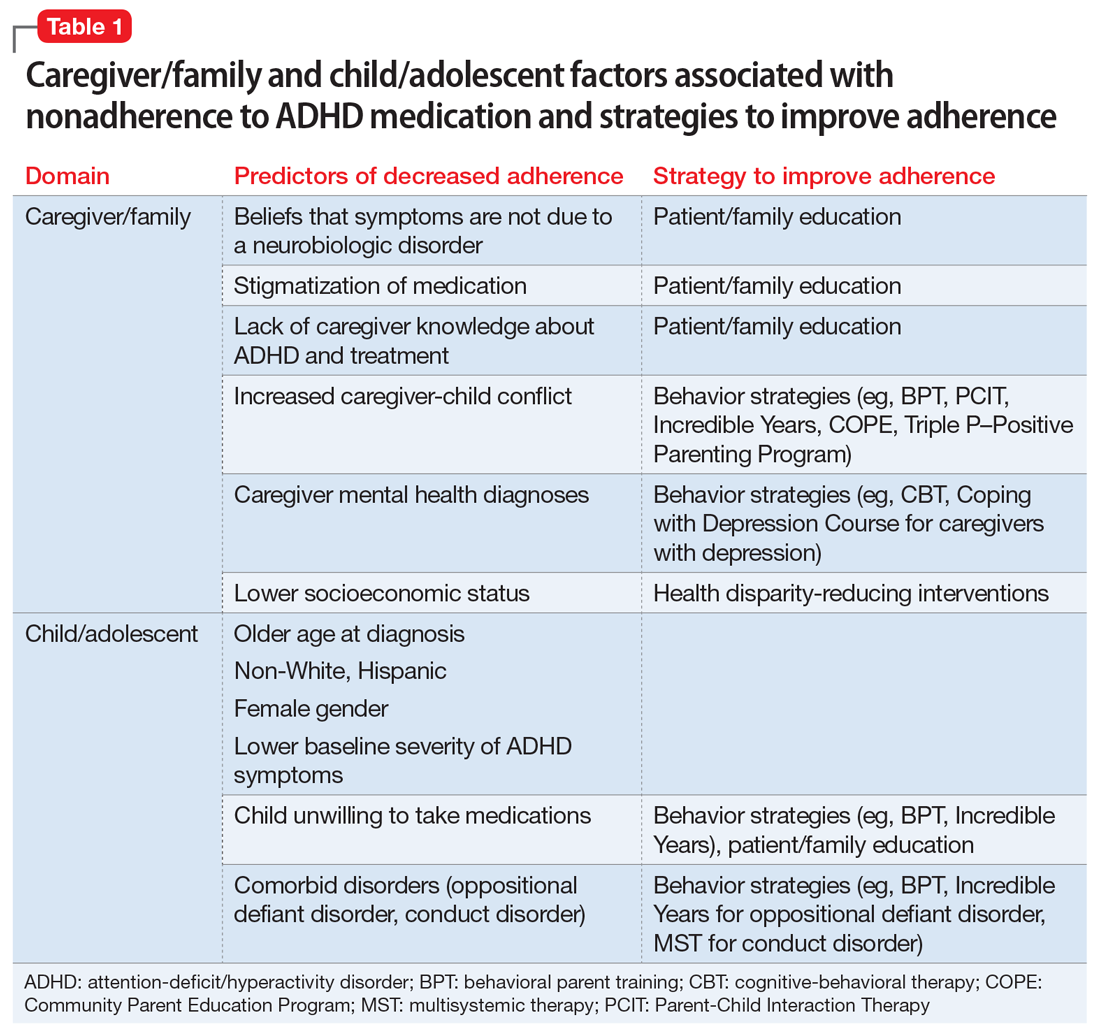
Adherence is defined as “the extent to which a person’s behavior (eg, taking medication) corresponds with agreed recommendations from a clinician.”24 Unfortunately, pediatric ADHD medication adherence has been found to be poor (approximately 64%).25-30 Nonadherence to ADHD medication has been linked to multiple factors, including caregiver/family and child/adolescent factors (Table 1), medication-related factors (Table 2), and health care/system factors (Table 3). Understanding and addressing these factors is essential to maximizing long-term outcomes. In this article, we review the factors associated with nonadherence to ADHD medication, and outline strategies to improve adherence.

Caregiver/family characteristics
Caregiver beliefs about ADHD and their attitudes toward treatment have been associated with the initiation of and adherence to ADHD medication. For example, caregivers who view a child’s difficulties as a medical disorder that requires a biologic intervention are more likely to accept and adhere to medication.31 Similarly, caregivers who perceive ADHD medication as safe, effective, and socially acceptable are more likely to be treatment-adherent.32-35
- increased caregiver knowledge about ADHD33
- receiving an ADHD diagnosis based on a thorough diagnostic process (ie, comprehensive psychological testing)36
- satisfaction with information about medicine
- comfort with the treatment plan.34
Socioeconomic status, family functioning, and caregiver mental health diagnoses (eg, ADHD, depression) have also been linked to ADHD medication adherence. Several studies, including the Multimodal Treatment Study of Children with ADHD,11 a landmark study of stimulant medication for children with ADHD, have found an association between low income and decreased likelihood of receiving ADHD medication.2,37-39 Further, Gau et al40 found that negative caregiver-child relationships and family dysfunction were associated with poor medication adherence in children with ADHD.9 Prior studies have also shown that mothers of children with ADHD are more likely to have depression and/or anxiety,41,42 and that caregivers with a history of mental health diagnoses are more accepting of initiating medication treatment for their children.43 However, additional studies have found that caregiver mental health diagnoses decreased the likelihood of ADHD medication adherence.40,44

Child characteristics
Child characteristics associated with decreased ADHD medication adherence include older age (eg, adolescents vs school-aged children),29,30,34,40,45-47 non-White race, Hispanic ethnicity,29,33,48-51 female gender,29,33,52 lower baseline ADHD symptom severity,30,37 and child unwillingness to take medications.34 However, prior studies have not been completely consistent about the relationship between child comorbid conditions (eg, oppositional defiant disorder [ODD], conduct disorder) and ADHD medication adherence. A few studies found that child comorbid conditions, especially ODD, mediate poor ADHD medication adherence, possibly secondary to an increased caregiver-child conflict.30,53,54 However, other studies have reported that the presence of comorbid ODD, depression, and anxiety predicted increased adherence to ADHD medications.37,46
Medication-related factors
Adverse effects of medications are the most commonly cited reason for ADHD medication nonadherence
On the other hand, increased ADHD medication effectiveness has been associated with improved medication adherence.5,34,54-56 Medication titration and dosing factors have also been shown to affect adherence. Specifically, adherence has been improved when ADHD medications are titrated in a systematic manner soon after starting treatment, and when families have an early first contact with a physician after starting medication (within 3 months).28 In addition, use of a simplified dose regimen has been linked to better adherence: patients who are prescribed long-acting stimulants are more likely to adhere to treatment compared with patients who take short-acting formulations.26,40,49,61-63 It is possible that long-acting stimulants increase adherence because they produce more even and sustained effects on ADHD symptoms throughout the day, compared with short-acting formulations.64 Furthermore, the inconvenience of taking multiple doses throughout the day, as well as the potential social stigma of mid-school day dosing, may negatively impact adherence to short-acting formulations.10
Continue to: Health care/system factors
Health care/system factors
Several studies have investigated the influence of health services factors on ADHD medication adherence. Specifically, limited transportation services and lack of mental health providers in the community have been linked to decreased ADHD medication adherence.47,65,66 Furthermore, limited insurance coverage and higher costs of ADHD medications, which lead to substantial out-of-pocket payments for families, have been associated with decreased likelihood of ADHD medication adherence.29,67
Clinician-related factors also can affect ADHD medication adherence. For example, a clinician’s lack knowledge of ADHD care can negatively impact ADHD medication adherence.68 Two studies have documented improved ADHD medication adherence when treatment is provided by specialists (eg, child psychiatrists) rather than by community primary care providers, possibly because specialists are more likely to provide close stimulant titration and monitoring (ie, ≥ 3 visits in the first 90 days) and use higher maximum doses.62,69 Furthermore, ADHD medication initiation and adherence are increased when patients have a strong working alliance with their clinician and trust the health care system,31,34,35 as well as when there is a match between the caregiver’s and clinician’s perception of the cause, course, and best treatment practices for a child’s ADHD.65
Strategies to improve medication adherence
A number of strategies to improve ADHD medication adherence can be derived from our knowledge of the factors that influence adherence.
Patient/family education. Unanswered questions about ADHD diagnosis, etiology, and medication adverse effects can negatively impact the ADHD treatment process. Therefore, patient/family education regarding ADHD and its management is necessary to improve medication adherence, because it helps families attain the knowledge, confidence, and motivation to manage their child’s condition.
Clinicians have an important role in educating patients about70:
- the medications they are taking
- why they are taking them
- what the medications look like
- the time of medication administration
- the potential adverse effects
- what to do if adverse effects occur
- what regular testing/monitoring is necessary.
Clinicians can provide appropriate psychoeducation by sharing written materials and trusted websites with families (see Related Resources).
Behavioral strategies. Behavioral interventions have been among the most effective strategies for improving medication adherence in other chronic conditions.71 Behavioral strategies are likely to be particularly important for families of children with ADHD and comorbid conditions such as ODD because these families experience considerable caregiver-child conflict.72 Moreover, parents of children with ADHD are at higher risk for having ADHD and depression themselves,73 both of which may interfere with a parent’s ability to obtain and administer medications consistently. Thus, for these families, using a combination of psychoeducation and behavioral strategies will be necessary to affect change in attitude and behavior. Behavioral strategies that can be used to improve medication adherence include:
- Technology-based interventions can reduce the impact of environmental barriers to adherence. For example, pharmacy automatic prescription renewal systems can reduce the likelihood of families failing to obtain ADHD medication refills. Pill reminder boxes, smartphone alerts, and setting various alarms can effectively prompt caregivers/patients to administer medication. In particular, these methods can be crucial in families for which multiple members have ADHD and its attendant difficulties with organization and task completion.
- Caregiver training may assist families in developing specific behavioral management skills that support adherence. This training can be as straightforward as instructing caregivers on the use of positive reinforcement when teaching their children to swallow pills. It may also encompass structured behavioral interventions aimed at training caregivers to utilize rewards and consequences in order to maximize medication adherence.74
Continue to: Clinician interventions
Clinician interventions. Clinicians can use decision aids to help inform families about treatment options, promote shared decision making, and decrease uncertainty about the treatment plan75 (see Related Resources). Early titration of ADHD medications and early first contact (within months of starting medication treatment) between caregivers and clinicians, whether via in-person visit, telephone, or email, have also been related to improved adherence.28 Furthermore, clinicians can improve adherence by prescribing a simplified medication regimen (ie, long-acting formulations that provide full-day coverage). To address the negative impact of high out-of-pocket ADHD medication costs on adherence, clinicians can also prescribe generic preparations and/or “preferred” medications options on an individual patient’s formulary.
Because clinician knowledge and expertise in ADHD care has been linked to improved patient medication adherence,68 clinicians are encouraged to use the American Academy of Pediatrics (AAP) guideline for diagnosis and treatment of ADHD, which includes a supplemental process of care algorithm (last published in 2011,10 with an updated guideline anticipated in 2019), as well as the AAP/National Institute for Children’s Health Quality (NICHQ) ADHD Toolkit,76 which includes items helpful for ADHD diagnosis and treatment. The Society for Developmental and Behavioral Pediatrics is also developing a clinical practice guideline for the diagnosis and treatment of complex ADHD (ie, ADHD complicated by coexisting mental health, developmental, and/or psychosocial conditions or issues), with publication anticipated in 2019. Primary care providers can also improve their expertise in ADHD care by pursuing additional mental health–related trainings (such as those conducted by the REACH Institute).77
Because receiving ADHD care from a specialist has been shown to improve medication initiation and adherence,62,69 other strategies to address the short supply of child psychiatrists include offering incentives to medical students to pursue a career in child psychiatry (eg, loan forgiveness). Telepsychiatry and co-location of mental health specialists and primary care providers are additional innovative ways in which ADHD specialty care can be delivered to more patients.64
Finally, providing culturally-sensitive care can strengthen the clinician-caregiver relationship and promote adherence to treatment. For example, clinicians can partner with local groups to increase their understanding of how different racial/ethnic groups perceive ADHD and its treatment.64
Peer support models. Peers are credible role models who have a valued role in facilitating the use of mental health services by empowering families and enhancing service satisfaction.78 In several communities in the United States, peer models using family advocates have been introduced.79 Family advocates are typically caregivers of children who have special needs or have been involved in the mental health system. Their perspective—as peers and first-hand consumers of the health care and/or mental health system—can make them powerful and effective coaches to families of children with ADHD. By helping families to navigate ADHD care systems successfully, family advocates can play an important role in enhancing ADHD medication adherence, although further investigation is needed. In addition, the stigma around ADHD medication use, which adversely impacts adherence, can be mitigated if caregivers participate in organized ADHD-related support groups (eg, Children and Adults with ADHD [CHADD]).
Continue to: Health disparity-reducing interventions
Health disparity-reducing interventions. Successful health disparity-reducing interventions—such as those developed to enhance care of other chronic disorders including asthma and diabetes—can be applied to improve ADHD care. These interventions, which include medical-legal partnerships (eg, between clinicians, social workers, legal advocates, and community partners) in primary care centers, have been shown to improve health insurance coverage and therefore health care access.80,81 Although some hardships linked to nonadherence (eg, low socioeconomic status) may not be amenable to health care–related interventions, screening for these hardships can identify children who are most at risk for poor adherence. This would alert clinicians to proactively identify barriers to adherence and implement mitigation strategies. This might include developing more streamlined, easier-to-follow management plans for these patients, such as those that can be delivered through pharmacist-physician collaborative programs82 and school-based therapy programs.83-85
Bottom Line
Suboptimal adherence to medications for attention-deficit/hyperactivity disorder (ADHD) can be addressed through patient/family education, behavioral strategies, clinician interventions, peer support models, and health disparity-reducing interventions. By improving ADHD treatment adherence, these interventions have the potential to maximize long-term outcomes.
Related Resources
- Cohen Children’s Medical Center Northwell Health. The ADHD Medication Guide. www.ADHDMedicationGuide.com. Revised December 31, 2017.
- Cincinnati Children’s Hospital Medical Center. Decision aids to facilitate shared decision making in practice. www.cincinnatichildrens.org/service/j/anderson-center/ evidence-based-care/decision-aids.
- CHADD. Children and Adults with attention-deficit/ hyperactivity disorder. www.chadd.org.
Drug Brand Name
Methylphenidate • Concerta, Ritalin
1. Froehlich TE, Lanphear BP, Epstein JN, et al. Prevalence, recognition, and treatment of attention-deficit/hyperactivity disorder in a national sample of US children. Arch Pediatr Adolesc Med. 2007;161(9):857-864.
2. Visser SN, Lesesne CA, Perou R. National estimates and factors associated with medication treatment for childhood attention-deficit/hyperactivity disorder. Pediatrics. 2007;119 (Suppl 1):S99-S106.
3. Danielson ML, Bitsko RH, Ghandour RM, et al. Prevalence of parent-reported ADHD diagnosis and associated treatment among U.S. children and adolescents, 2016. J Clin Child Adolesc Psychol. 2018;47(2):199-212.
4. Molina BS, Hinshaw SP, Swanson JM, et al. The MTA at 8 years: prospective follow-up of children treated for combined-type ADHD in a multisite study. J Am Acad Child Adolesc Psychiatry. 2009;48(5):484-500.
5. Charach A, Dashti B, Carson P, et al. Attention deficit hyperactivity disorder: effectiveness of treatment in at-risk preschoolers; long-term effectiveness in all ages; and variability in prevalence, diagnosis, and treatment. Rockville, MD: Agency for Healthcare Research and Quality; 2011. http://www.ncbi.nlm.nih.gov/books/NBK82368/.
6. Wehmeier PM, Schacht A, Barkley RA. Social and emotional impairment in children and adolescents with ADHD and the impact on quality of life. J Adolesc Health. 2010;46(3):209-217.
7. Barkley RA, Fischer M, Smallish L, et al. Young adult outcome of hyperactive children: adaptive functioning in major life activities. J Am Acad Child Adolesc Psychiatry. 2006;45(2):192-202.
8. Spencer TJ, Biederman J, Mick E. Attention-deficit/hyperactivity disorder: diagnosis, lifespan, comorbidities, and neurobiology. J Pediatr Psychol. 2007;32(6):631-642.
9. Pliszka S, the AACAP Work Group on Quality Issues. Practice parameter for the assessment and treatment of children and adolescents with attention-deficit/hyperactivity disorder. J Am Acad Child Adolesc Psychiatry. 2007;46(7):894-921.
10. Subcommittee on Attention-Deficit/Hyperactivity Disorder; Steering Committee on Quality Improvement and Management. ADHD: clinical practice guideline for the diagnosis, evaluation, and treatment of attention-deficit/hyperactivity disorder in children and adolescents. Pediatrics. 2011;128(5):1007-1022.
11. A 14-month randomized clinical trial of treatment strategies for attention-deficit/hyperactivity disorder. The MTA Cooperative Group. Multimodal Treatment Study of Children with ADHD. Arch Gen Psychiatry. 1999;56(12):1073-1086.
12. Abikoff H, Hechtman L, Klein RG, et al. Symptomatic improvement in children with ADHD treated with long-term methylphenidate and multimodal psychosocial treatment. J Am Acad Child Adolesc Psychiatry. 2004;43(7):802-811.
13. Barbaresi WJ, Katusic SK, Colligan RC, et al. Long-term school outcomes for children with attention-deficit/hyperactivity disorder: a population-based perspective. J Dev Behav Pediatr. 2007;28(4):265-273.
14. Scheffler RM, Brown TT, Fulton BD, et al. Positive association between attention-deficit/ hyperactivity disorder medication use and academic achievement during elementary school. Pediatrics. 2009;123(5):1273-1279.
15. Dalsgaard S, Nielsen HS, Simonsen M. Five-fold increase in national prevalence rates of attention-deficit/hyperactivity disorder medications for children and adolescents with autism spectrum disorder, attention-deficit/hyperactivity disorder, and other psychiatric disorders: a Danish register-based study. J Child Adolesc Psychopharmacol. 2013;23(7):432-439.
16. Lichtenstein P, Halldner L, Zetterqvist J, et al. Medication for attention deficit-hyperactivity disorder and criminality. N Engl J Med. 2012;367(21):2006-2014.
17. Chang Z, Lichtenstein P, D’Onofrio BM, et al. Serious transport accidents in adults with attention-deficit/hyperactivity disorder and the effect of medication: a population-based study. JAMA Psychiatry. 2014;71(3):319-325.
18. Chang Z, Quinn PD, Hur K, et al. Association between medication use for attention-deficit/hyperactivity disorder and risk of motor vehicle crashes. JAMA Psychiatry. 2017;74(6):597-603.
19. Dalsgaard S, Leckman JF, Mortensen PB, et al. Effect of drugs on the risk of injuries in children with attention deficit hyperactivity disorder: a prospective cohort study. Lancet Psychiatry. 2015;2(8):702-709.
20. Chang Z, Lichtenstein P, Halldner L, et al. Stimulant ADHD medication and risk for substance abuse. J Child Psychol Psychiatry. 2014;55(8):878-885.
21. Fischer M, Barkley RA. Childhood stimulant treatment and risk for later substance abuse. J Clin Psychiatry. 2003;64(Suppl 11):19-23.
22. Biederman J. Pharmacotherapy for attention-deficit/hyperactivity disorder (ADHD) decreases the risk for substance abuse: findings from a longitudinal follow-up of youths with and without ADHD. J Clin Psychiatry. 2003;64(Suppl 11):3-8.
23. Chang Z, D’Onofrio BM, Quinn PD, et al. Medicationfor attention-deficit/hyperactivity disorder and risk for depression: a nationwide longitudinal cohort study. Biol Psychiatry. 2016;80(12):916-922.
24. World Health Organization. Adherence to long-term therapies: evidence for action. https://www.who.int/chp/knowledge/publications/adherence_full_report.pdf?ua=1. Published 2003. Accessed July 22, 2019.
25. Perwien A, Hall J, Swensen A, et al. Stimulant treatment patterns and compliance in children and adults with newly treated attention-deficit/hyperactivity disorder. J Manag Care Pharm. 2004;10(2):122-129.
26. Faraone SV, Biederman J, Zimmerman B. An analysis of patient adherence to treatment during a 1-year, open-label study of OROS methylphenidate in children with ADHD. J Atten Disord. 2007;11(2):157-166.
27. Barner JC, Khoza S, Oladapo A. ADHD medication use, adherence, persistence and cost among Texas Medicaid children. Curr Med Res Opin. 2011;27(Suppl 2):13-22.
28. Brinkman WB, Baum R, Kelleher KJ, et al. Relationship between attention-deficit/hyperactivity disorder care and medication continuity. J Am Acad Child Adolesc Psychiatry. 2016;55(4):289-294.
29. Bokhari FAS, Heiland F, Levine P, et al. Risk factors for discontinuing drug therapy among children with ADHD. Health Services and Outcomes Research Methodology. 2008;8(3):134-158.
30. Thiruchelvam D, Charach A, Schachar RJ. Moderators and mediators of long-term adherence to stimulant treatment in children with ADHD. J Am Acad Child Adolesc Psychiatry. 2001;40(8):922-928.
31. DosReis S, Mychailyszyn MP, Evans-Lacko SE, et al. The meaning of attention-deficit/hyperactivity disorder medication and parents’ initiation and continuity of treatment for their child. J Child Adolesc Psychopharmacol. 2009;19(4):377-383.
32. dosReis S, Myers MA. Parental attitudes and involvement in psychopharmacological treatment for ADHD: a conceptual model. Int Rev Psychiatry. 2008;20(2):135-141.
33. Bussing R, Koro-Ljungberg M, Noguchi K, et al. Willingness to use ADHD treatments: a mixed methods study of perceptions by adolescents, parents, health professionals and teachers. Soc Sci Med. 2012;74(1):92-100.
34. Brinkman WB, Sucharew H, Majcher JH, et al. Predictors of medication continuity in children with ADHD. Pediatrics. 2018;141(6). doi: 10.1542/peds.2017-2580.
35. Coletti DJ, Pappadopulos E, Katsiotas NJ, et al. Parent perspectives on the decision to initiate medication treatment of attention-deficit/hyperactivity disorder. J Child Adolesc Psychopharmacol. 2012;22(3):226-237.
36. Bussing R, Gary FA. Practice guidelines and parental ADHD treatment evaluations: friends or foes? Harv Rev Psychiatry. 2001;9(5):223-233.
37. Charach A, Gajaria A. Improving psychostimulant adherence in children with ADHD. Expert Rev Neurother. 2008;8(10):1563-1571.
38. Rieppi R, Greenhill LL, Ford RE, et al. Socioeconomic status as a moderator of ADHD treatment outcomes. J Am Acad Child Adolesc Psychiatry. 2002;41(3):269-277.
39. Swanson JM, Hinshaw SP, Arnold LE, et al. Secondary evaluations of MTA 36-month outcomes: propensity score and growth mixture model analyses. J Am Acad Child Adolesc Psychiatry. 2007;46(8):1003-1014.
40. Gau SS, Shen HY, Chou MC, et al. Determinants of adherence to methylphenidate and the impact of poor adherence on maternal and family measures. J Child Adolesc Psychopharmacol. 2006;16(3):286-297.
41. Barkley RA, Fischer M, Edelbrock C, et al. The adolescent outcome of hyperactive children diagnosed by research criteria--III. Mother-child interactions, family conflicts and maternal psychopathology. J Child Psychol Psychiatry. 1991;32(2):233-255.
42. Kashdan TB, Jacob RG, Pelham WE, et al. Depression and anxiety in parents of children with ADHD and varying levels of oppositional defiant behaviors: modeling relationships with family functioning. J Clin Child Adolesc Psychol. 2004;33(1):169-181.
43. Chavira DA, Stein MB, Bailey K, et al. Parental opinions regarding treatment for social anxiety disorder in youth. J Dev Behav Pediatr. 2003;24(5):315-322.
44. Leslie LK, Aarons GA, Haine RA, et al. Caregiver depression and medication use by youths with ADHD who receive services in the public sector. Psychiatr Serv. 2007;58(1):131-134.
45. Barbaresi WJ, Katusic SK, Colligan RC, et al. Long-term stimulant medication treatment of attention-deficit/hyperactivity disorder: results from a population-based study. J Dev Behav Pediatr. 2006;27(1):1-10.
46. Atzori P, Usala T, Carucci S, et al. Predictive factors for persistent use and compliance of immediate-release methylphenidate: a 36-month naturalistic study. J Child Adolesc Psychopharmacol. 2009;19(6):673-681.
47. Chen CY, Yeh HH, Chen KH, et al. Differential effects of predictors on methylphenidate initiation and discontinuation among young people with newly diagnosed attention-deficit/hyperactivity disorder. J Child Adolesc Psychopharmacol. 2011;21(3):265-273.
48. Winterstein AG, Gerhard T, Shuster J, et al. Utilization of pharmacologic treatment in youths with attention deficit/hyperactivity disorder in Medicaid database. Ann Pharmacother. 2008;42(1):24-31.
49. Marcus SC, Wan GJ, Kemner JE, et al. Continuity of methylphenidate treatment for attention-deficit/hyperactivity disorder. Arch Pediatr Adolesc Med. 2005;159(6):572-578.
50. Cummings JR JX, Allen L, Lally C, et al. Racial and ethnic differences in ADHD treatment quality among Medicaid-enrolled youth. Pediatrics. 2017;139(6):e2016-e2044.
51. Hudson JL, Miller GE, Kirby JB. Explaining racial and ethnic differences in children’s use of stimulant medications. Med Care. 2007;45(11):1068-1075.
52. van den Ban E, Souverein PC, Swaab H, et al. Less discontinuation of ADHD drug use since the availability of long-acting ADHD medication in children, adolescents and adults under the age of 45 years in the Netherlands. Atten Defic Hyperact Disord. 2010;2(4):213-220.
53. Charach A, Ickowicz A, Schachar R. Stimulant treatment over five years: adherence, effectiveness, and adverse effects. J Am Acad Child Adolesc Psychiatry. 2004;43(5):559-567.
54. Toomey SL, Sox CM, Rusinak D, et al. Why do children with ADHD discontinue their medication? Clin Pediatr (Phila). 2012;51(8):763-769.
55. Brinkman WB, Simon JO, Epstein JN. Reasons why children and adolescents with attention-deficit/hyperactivity disorder stop and restart taking medicine. Acad Pediatr. 2018;18(3):273-280.
56. Wehmeier PM, Dittmann RW, Banaschewski T. Treatment compliance or medication adherence in children and adolescents on ADHD medication in clinical practice: results from the COMPLY observational study. Atten Defic Hyperact Disord. 2015;7(2):165-174.
57. Frank E, Ozon C, Nair V, et al. Examining why patients with attention-deficit/hyperactivity disorder lack adherence to medication over the long term: a review and analysis. J Clin Psychiatry. 2015;76(11):e1459-e1468.
58. Pozzi M, Carnovale C, Peeters G, et al. Adverse drug events related to mood and emotion in paediatric patients treated for ADHD: a meta-analysis. J Affect Disord. 2018;238:161-178.
59. Stuckelman ZD, Mulqueen JM, Ferracioli-Oda E, et al. Risk of irritability with psychostimulant treatment in children with ADHD: a meta-analysis. J Clin Psychiatry. 2017;78(6):e648-e655.
60. Cortese S, Adamo N, Del Giovane C, et al. Comparative efficacy and tolerability of medications for attention-deficit hyperactivity disorder in children, adolescents, and adults: a systematic review and network meta-analysis. Lancet Psychiatry. 2018;5(9):727-738.
61. Lawson KA, Johnsrud M, Hodgkins P, et al. Utilization patterns of stimulants in ADHD in the Medicaid population: a retrospective analysis of data from the Texas Medicaid program. Clin Ther. 2012;34(4):944-956 e944.
62. Olfson M, Marcus S, Wan G. Stimulant dosing for children with ADHD: a medical claims analysis. J Am Acad Child Adolesc Psychiatry. 2009;48(1):51-59.
63. Jensen PS, Arnold LE, Swanson JM, et al. 3-year follow-up of the NIMH MTA study. J Am Acad Child Adolesc Psychiatry. 2007;46(8):989-1002.
64. Van Cleave J, Leslie LK. Approaching ADHD as a chronic condition: implications for long-term adherence. Pediatr Ann. 2008;37(1):19-26.
65. Leslie LK, Plemmons D, Monn AR, et al. Investigating ADHD treatment trajectories: listening to families’ stories about medication use. J Dev Behav Pediatr. 2007;28(3):179-188.
66. Fiks AG, Mayne S, Localio AR, et al. Shared decision making and behavioral impairment: a national study among children with special health care needs. BMC Pediatr. 2012;12:153.
67. Stevens J, Harman JS, Kelleher KJ. Race/ethnicity and insurance status as factors associated with ADHD treatment patterns. J Child Adolesc Psychopharmacol. 2005;15(1):88-96.
68. Charach A, Skyba A, Cook L, et al. Using stimulant medication for children with ADHD: what do parents say? A brief report. J Can Acad Child Adolesc Psychiatry. 2006;15(2):75-83.
69. Chen CY, Gerhard T, Winterstein AG. Determinants of initial pharmacological treatment for youths with attention-deficit/hyperactivity disorder. J Child Adolescent Psychopharmacol. 2009;19(2):187-195.
70. National Council on Patient Information and Education. Enhancing prescription medication adherence: a national action plan. http://www.bemedwise.org/docs/enhancingprescriptionmedicineadherence.pdf. Published August 2007. Accessed July 22, 2019.
71. Kahana S, Drotar D, Frazier T. Meta-analysis of psychological interventions to promote adherence to treatment in pediatric chronic health conditions. J Pediatr Psychol. 2008;33(6):590-611.
72. Johnston C, Mash EJ. Families of children with attention-deficit/hyperactivity disorder: review and recommendations for future research. Clin Child Fam Psychol Rev. 2001;4(3):183-207.
73. Chronis AM, Lahey BB, Pelham WE Jr., et al. Psychopathology and substance abuse in parents of young children with attention-deficit/hyperactivity disorder. J Am Acad Child Adolesc Psychiatry. 2003;42(12):1424-1432.
74. Chacko A, Newcorn JH, Feirsen N, et al. Improving medication adherence in chronic pediatric health conditions: a focus on ADHD in youth. Curr Pharm Des. 2010;16(22):2416-2423.
75. Brinkman WB, Hartl Majcher J, Polling LM, et al. Shared decision-making to improve attention-deficit hyperactivity disorder care. Patient Educ Couns. 2013;93(1):95-101.
76. American Academy of Pediatrics. Caring for children with ADHD: a resource toolkit for clinicians. 2nd ed. https://www.aap.org/en-us/pubserv/adhd2/Pages/default.aspx. Published 2011. Accessed July 22, 2019.
77. The REACH Institute. Course dates and registration. http://www.thereachinstitute.org/services/for-primary-care-practitioners/training-dates-and-registration. Accessed July 22, 2019.
78. Sells D, Davidson L, Jewell C, et al. The treatment relationship in peer-based and regular case management for clients with severe mental illness. Psychiatr Serv. 2006;57(8):1179-1184.
79. Hoagwood KE, Green E, Kelleher K, et al. Family advocacy, support and education in children’s mental health: results of a national survey. Adm Policy Ment Health. 2008;35(1-2):73-83.
80. Klein MD, Beck AF, Henize AW, et al. Doctors and lawyers collaborating to HeLP children—outcomes from a successful partnership between professions. J Health Care Poor Underserved. 2013;24(3):1063-1073.
81. Weintraub D, Rodgers MA, Botcheva L, et al. Pilot study of medical-legal partnership to address social and legal needs of patients. J Health Care Poor Underserved. 2010;21(Suppl 2):157-168.
82. Bradley CL, Luder HR, Beck AF, et al. Pediatric asthma medication therapy management through community pharmacy and primary care collaboration. J Am Pharm Assoc (2003). 2016;56(4):455-460.
83. Noyes K, Bajorska A, Fisher S, et al. Cost-effectiveness of the school-based asthma therapy (SBAT) program. Pediatrics. 2013;131(3):e709-e717.
84. Halterman JS, Fagnano M, Montes G, et al. The school-based preventive asthma care trial: results of a pilot study. J Pediatr. 2012;161(6):1109-1115.
85. Halterman JS, Szilagyi PG, Fisher SG, et al. Randomized controlled trial to improve care for urban children with asthma: results of the school-based asthma therapy trial. Arch Pediatr Adolesc Med. 2011;165(3):262-268.
Attention-deficit/hyperactivity disorder (ADHD) is the most common childhood neurodevelopmental disorder, affecting 8% to 12% of school-aged children in the United States1-3 with significant impairments that often persist into adulthood.4-8 Current guidelines recommend stimulant medication and/or behavioral therapies as first-line treatments for ADHD.9,10 There is a wealth of evidence on the efficacy of stimulants in ADHD, with the most significant effects noted on core ADHD symptoms.11,12 Additional evidence links stimulants to decreased long-term negative outcomes, including reduced school absences and grade retention,13 as well as modestly but significantly improved reading and math scores.14 Other studies have reported that individuals with ADHD who receive medication have decreased criminality,15,16 motor vehicle accidents,17,18 injuries,19 substance abuse,20-22 and risk for subsequent and concurrent depression.23 Therefore, the evidence suggests that consistent medication treatment helps improve outcomes for individuals with ADHD.

Adherence is defined as “the extent to which a person’s behavior (eg, taking medication) corresponds with agreed recommendations from a clinician.”24 Unfortunately, pediatric ADHD medication adherence has been found to be poor (approximately 64%).25-30 Nonadherence to ADHD medication has been linked to multiple factors, including caregiver/family and child/adolescent factors (Table 1), medication-related factors (Table 2), and health care/system factors (Table 3). Understanding and addressing these factors is essential to maximizing long-term outcomes. In this article, we review the factors associated with nonadherence to ADHD medication, and outline strategies to improve adherence.

Caregiver/family characteristics
Caregiver beliefs about ADHD and their attitudes toward treatment have been associated with the initiation of and adherence to ADHD medication. For example, caregivers who view a child’s difficulties as a medical disorder that requires a biologic intervention are more likely to accept and adhere to medication.31 Similarly, caregivers who perceive ADHD medication as safe, effective, and socially acceptable are more likely to be treatment-adherent.32-35
- increased caregiver knowledge about ADHD33
- receiving an ADHD diagnosis based on a thorough diagnostic process (ie, comprehensive psychological testing)36
- satisfaction with information about medicine
- comfort with the treatment plan.34
Socioeconomic status, family functioning, and caregiver mental health diagnoses (eg, ADHD, depression) have also been linked to ADHD medication adherence. Several studies, including the Multimodal Treatment Study of Children with ADHD,11 a landmark study of stimulant medication for children with ADHD, have found an association between low income and decreased likelihood of receiving ADHD medication.2,37-39 Further, Gau et al40 found that negative caregiver-child relationships and family dysfunction were associated with poor medication adherence in children with ADHD.9 Prior studies have also shown that mothers of children with ADHD are more likely to have depression and/or anxiety,41,42 and that caregivers with a history of mental health diagnoses are more accepting of initiating medication treatment for their children.43 However, additional studies have found that caregiver mental health diagnoses decreased the likelihood of ADHD medication adherence.40,44

Child characteristics
Child characteristics associated with decreased ADHD medication adherence include older age (eg, adolescents vs school-aged children),29,30,34,40,45-47 non-White race, Hispanic ethnicity,29,33,48-51 female gender,29,33,52 lower baseline ADHD symptom severity,30,37 and child unwillingness to take medications.34 However, prior studies have not been completely consistent about the relationship between child comorbid conditions (eg, oppositional defiant disorder [ODD], conduct disorder) and ADHD medication adherence. A few studies found that child comorbid conditions, especially ODD, mediate poor ADHD medication adherence, possibly secondary to an increased caregiver-child conflict.30,53,54 However, other studies have reported that the presence of comorbid ODD, depression, and anxiety predicted increased adherence to ADHD medications.37,46
Medication-related factors
Adverse effects of medications are the most commonly cited reason for ADHD medication nonadherence
On the other hand, increased ADHD medication effectiveness has been associated with improved medication adherence.5,34,54-56 Medication titration and dosing factors have also been shown to affect adherence. Specifically, adherence has been improved when ADHD medications are titrated in a systematic manner soon after starting treatment, and when families have an early first contact with a physician after starting medication (within 3 months).28 In addition, use of a simplified dose regimen has been linked to better adherence: patients who are prescribed long-acting stimulants are more likely to adhere to treatment compared with patients who take short-acting formulations.26,40,49,61-63 It is possible that long-acting stimulants increase adherence because they produce more even and sustained effects on ADHD symptoms throughout the day, compared with short-acting formulations.64 Furthermore, the inconvenience of taking multiple doses throughout the day, as well as the potential social stigma of mid-school day dosing, may negatively impact adherence to short-acting formulations.10
Continue to: Health care/system factors
Health care/system factors
Several studies have investigated the influence of health services factors on ADHD medication adherence. Specifically, limited transportation services and lack of mental health providers in the community have been linked to decreased ADHD medication adherence.47,65,66 Furthermore, limited insurance coverage and higher costs of ADHD medications, which lead to substantial out-of-pocket payments for families, have been associated with decreased likelihood of ADHD medication adherence.29,67
Clinician-related factors also can affect ADHD medication adherence. For example, a clinician’s lack knowledge of ADHD care can negatively impact ADHD medication adherence.68 Two studies have documented improved ADHD medication adherence when treatment is provided by specialists (eg, child psychiatrists) rather than by community primary care providers, possibly because specialists are more likely to provide close stimulant titration and monitoring (ie, ≥ 3 visits in the first 90 days) and use higher maximum doses.62,69 Furthermore, ADHD medication initiation and adherence are increased when patients have a strong working alliance with their clinician and trust the health care system,31,34,35 as well as when there is a match between the caregiver’s and clinician’s perception of the cause, course, and best treatment practices for a child’s ADHD.65
Strategies to improve medication adherence
A number of strategies to improve ADHD medication adherence can be derived from our knowledge of the factors that influence adherence.
Patient/family education. Unanswered questions about ADHD diagnosis, etiology, and medication adverse effects can negatively impact the ADHD treatment process. Therefore, patient/family education regarding ADHD and its management is necessary to improve medication adherence, because it helps families attain the knowledge, confidence, and motivation to manage their child’s condition.
Clinicians have an important role in educating patients about70:
- the medications they are taking
- why they are taking them
- what the medications look like
- the time of medication administration
- the potential adverse effects
- what to do if adverse effects occur
- what regular testing/monitoring is necessary.
Clinicians can provide appropriate psychoeducation by sharing written materials and trusted websites with families (see Related Resources).
Behavioral strategies. Behavioral interventions have been among the most effective strategies for improving medication adherence in other chronic conditions.71 Behavioral strategies are likely to be particularly important for families of children with ADHD and comorbid conditions such as ODD because these families experience considerable caregiver-child conflict.72 Moreover, parents of children with ADHD are at higher risk for having ADHD and depression themselves,73 both of which may interfere with a parent’s ability to obtain and administer medications consistently. Thus, for these families, using a combination of psychoeducation and behavioral strategies will be necessary to affect change in attitude and behavior. Behavioral strategies that can be used to improve medication adherence include:
- Technology-based interventions can reduce the impact of environmental barriers to adherence. For example, pharmacy automatic prescription renewal systems can reduce the likelihood of families failing to obtain ADHD medication refills. Pill reminder boxes, smartphone alerts, and setting various alarms can effectively prompt caregivers/patients to administer medication. In particular, these methods can be crucial in families for which multiple members have ADHD and its attendant difficulties with organization and task completion.
- Caregiver training may assist families in developing specific behavioral management skills that support adherence. This training can be as straightforward as instructing caregivers on the use of positive reinforcement when teaching their children to swallow pills. It may also encompass structured behavioral interventions aimed at training caregivers to utilize rewards and consequences in order to maximize medication adherence.74
Continue to: Clinician interventions
Clinician interventions. Clinicians can use decision aids to help inform families about treatment options, promote shared decision making, and decrease uncertainty about the treatment plan75 (see Related Resources). Early titration of ADHD medications and early first contact (within months of starting medication treatment) between caregivers and clinicians, whether via in-person visit, telephone, or email, have also been related to improved adherence.28 Furthermore, clinicians can improve adherence by prescribing a simplified medication regimen (ie, long-acting formulations that provide full-day coverage). To address the negative impact of high out-of-pocket ADHD medication costs on adherence, clinicians can also prescribe generic preparations and/or “preferred” medications options on an individual patient’s formulary.
Because clinician knowledge and expertise in ADHD care has been linked to improved patient medication adherence,68 clinicians are encouraged to use the American Academy of Pediatrics (AAP) guideline for diagnosis and treatment of ADHD, which includes a supplemental process of care algorithm (last published in 2011,10 with an updated guideline anticipated in 2019), as well as the AAP/National Institute for Children’s Health Quality (NICHQ) ADHD Toolkit,76 which includes items helpful for ADHD diagnosis and treatment. The Society for Developmental and Behavioral Pediatrics is also developing a clinical practice guideline for the diagnosis and treatment of complex ADHD (ie, ADHD complicated by coexisting mental health, developmental, and/or psychosocial conditions or issues), with publication anticipated in 2019. Primary care providers can also improve their expertise in ADHD care by pursuing additional mental health–related trainings (such as those conducted by the REACH Institute).77
Because receiving ADHD care from a specialist has been shown to improve medication initiation and adherence,62,69 other strategies to address the short supply of child psychiatrists include offering incentives to medical students to pursue a career in child psychiatry (eg, loan forgiveness). Telepsychiatry and co-location of mental health specialists and primary care providers are additional innovative ways in which ADHD specialty care can be delivered to more patients.64
Finally, providing culturally-sensitive care can strengthen the clinician-caregiver relationship and promote adherence to treatment. For example, clinicians can partner with local groups to increase their understanding of how different racial/ethnic groups perceive ADHD and its treatment.64
Peer support models. Peers are credible role models who have a valued role in facilitating the use of mental health services by empowering families and enhancing service satisfaction.78 In several communities in the United States, peer models using family advocates have been introduced.79 Family advocates are typically caregivers of children who have special needs or have been involved in the mental health system. Their perspective—as peers and first-hand consumers of the health care and/or mental health system—can make them powerful and effective coaches to families of children with ADHD. By helping families to navigate ADHD care systems successfully, family advocates can play an important role in enhancing ADHD medication adherence, although further investigation is needed. In addition, the stigma around ADHD medication use, which adversely impacts adherence, can be mitigated if caregivers participate in organized ADHD-related support groups (eg, Children and Adults with ADHD [CHADD]).
Continue to: Health disparity-reducing interventions
Health disparity-reducing interventions. Successful health disparity-reducing interventions—such as those developed to enhance care of other chronic disorders including asthma and diabetes—can be applied to improve ADHD care. These interventions, which include medical-legal partnerships (eg, between clinicians, social workers, legal advocates, and community partners) in primary care centers, have been shown to improve health insurance coverage and therefore health care access.80,81 Although some hardships linked to nonadherence (eg, low socioeconomic status) may not be amenable to health care–related interventions, screening for these hardships can identify children who are most at risk for poor adherence. This would alert clinicians to proactively identify barriers to adherence and implement mitigation strategies. This might include developing more streamlined, easier-to-follow management plans for these patients, such as those that can be delivered through pharmacist-physician collaborative programs82 and school-based therapy programs.83-85
Bottom Line
Suboptimal adherence to medications for attention-deficit/hyperactivity disorder (ADHD) can be addressed through patient/family education, behavioral strategies, clinician interventions, peer support models, and health disparity-reducing interventions. By improving ADHD treatment adherence, these interventions have the potential to maximize long-term outcomes.
Related Resources
- Cohen Children’s Medical Center Northwell Health. The ADHD Medication Guide. www.ADHDMedicationGuide.com. Revised December 31, 2017.
- Cincinnati Children’s Hospital Medical Center. Decision aids to facilitate shared decision making in practice. www.cincinnatichildrens.org/service/j/anderson-center/ evidence-based-care/decision-aids.
- CHADD. Children and Adults with attention-deficit/ hyperactivity disorder. www.chadd.org.
Drug Brand Name
Methylphenidate • Concerta, Ritalin
Attention-deficit/hyperactivity disorder (ADHD) is the most common childhood neurodevelopmental disorder, affecting 8% to 12% of school-aged children in the United States1-3 with significant impairments that often persist into adulthood.4-8 Current guidelines recommend stimulant medication and/or behavioral therapies as first-line treatments for ADHD.9,10 There is a wealth of evidence on the efficacy of stimulants in ADHD, with the most significant effects noted on core ADHD symptoms.11,12 Additional evidence links stimulants to decreased long-term negative outcomes, including reduced school absences and grade retention,13 as well as modestly but significantly improved reading and math scores.14 Other studies have reported that individuals with ADHD who receive medication have decreased criminality,15,16 motor vehicle accidents,17,18 injuries,19 substance abuse,20-22 and risk for subsequent and concurrent depression.23 Therefore, the evidence suggests that consistent medication treatment helps improve outcomes for individuals with ADHD.

Adherence is defined as “the extent to which a person’s behavior (eg, taking medication) corresponds with agreed recommendations from a clinician.”24 Unfortunately, pediatric ADHD medication adherence has been found to be poor (approximately 64%).25-30 Nonadherence to ADHD medication has been linked to multiple factors, including caregiver/family and child/adolescent factors (Table 1), medication-related factors (Table 2), and health care/system factors (Table 3). Understanding and addressing these factors is essential to maximizing long-term outcomes. In this article, we review the factors associated with nonadherence to ADHD medication, and outline strategies to improve adherence.

Caregiver/family characteristics
Caregiver beliefs about ADHD and their attitudes toward treatment have been associated with the initiation of and adherence to ADHD medication. For example, caregivers who view a child’s difficulties as a medical disorder that requires a biologic intervention are more likely to accept and adhere to medication.31 Similarly, caregivers who perceive ADHD medication as safe, effective, and socially acceptable are more likely to be treatment-adherent.32-35
- increased caregiver knowledge about ADHD33
- receiving an ADHD diagnosis based on a thorough diagnostic process (ie, comprehensive psychological testing)36
- satisfaction with information about medicine
- comfort with the treatment plan.34
Socioeconomic status, family functioning, and caregiver mental health diagnoses (eg, ADHD, depression) have also been linked to ADHD medication adherence. Several studies, including the Multimodal Treatment Study of Children with ADHD,11 a landmark study of stimulant medication for children with ADHD, have found an association between low income and decreased likelihood of receiving ADHD medication.2,37-39 Further, Gau et al40 found that negative caregiver-child relationships and family dysfunction were associated with poor medication adherence in children with ADHD.9 Prior studies have also shown that mothers of children with ADHD are more likely to have depression and/or anxiety,41,42 and that caregivers with a history of mental health diagnoses are more accepting of initiating medication treatment for their children.43 However, additional studies have found that caregiver mental health diagnoses decreased the likelihood of ADHD medication adherence.40,44

Child characteristics
Child characteristics associated with decreased ADHD medication adherence include older age (eg, adolescents vs school-aged children),29,30,34,40,45-47 non-White race, Hispanic ethnicity,29,33,48-51 female gender,29,33,52 lower baseline ADHD symptom severity,30,37 and child unwillingness to take medications.34 However, prior studies have not been completely consistent about the relationship between child comorbid conditions (eg, oppositional defiant disorder [ODD], conduct disorder) and ADHD medication adherence. A few studies found that child comorbid conditions, especially ODD, mediate poor ADHD medication adherence, possibly secondary to an increased caregiver-child conflict.30,53,54 However, other studies have reported that the presence of comorbid ODD, depression, and anxiety predicted increased adherence to ADHD medications.37,46
Medication-related factors
Adverse effects of medications are the most commonly cited reason for ADHD medication nonadherence
On the other hand, increased ADHD medication effectiveness has been associated with improved medication adherence.5,34,54-56 Medication titration and dosing factors have also been shown to affect adherence. Specifically, adherence has been improved when ADHD medications are titrated in a systematic manner soon after starting treatment, and when families have an early first contact with a physician after starting medication (within 3 months).28 In addition, use of a simplified dose regimen has been linked to better adherence: patients who are prescribed long-acting stimulants are more likely to adhere to treatment compared with patients who take short-acting formulations.26,40,49,61-63 It is possible that long-acting stimulants increase adherence because they produce more even and sustained effects on ADHD symptoms throughout the day, compared with short-acting formulations.64 Furthermore, the inconvenience of taking multiple doses throughout the day, as well as the potential social stigma of mid-school day dosing, may negatively impact adherence to short-acting formulations.10
Continue to: Health care/system factors
Health care/system factors
Several studies have investigated the influence of health services factors on ADHD medication adherence. Specifically, limited transportation services and lack of mental health providers in the community have been linked to decreased ADHD medication adherence.47,65,66 Furthermore, limited insurance coverage and higher costs of ADHD medications, which lead to substantial out-of-pocket payments for families, have been associated with decreased likelihood of ADHD medication adherence.29,67
Clinician-related factors also can affect ADHD medication adherence. For example, a clinician’s lack knowledge of ADHD care can negatively impact ADHD medication adherence.68 Two studies have documented improved ADHD medication adherence when treatment is provided by specialists (eg, child psychiatrists) rather than by community primary care providers, possibly because specialists are more likely to provide close stimulant titration and monitoring (ie, ≥ 3 visits in the first 90 days) and use higher maximum doses.62,69 Furthermore, ADHD medication initiation and adherence are increased when patients have a strong working alliance with their clinician and trust the health care system,31,34,35 as well as when there is a match between the caregiver’s and clinician’s perception of the cause, course, and best treatment practices for a child’s ADHD.65
Strategies to improve medication adherence
A number of strategies to improve ADHD medication adherence can be derived from our knowledge of the factors that influence adherence.
Patient/family education. Unanswered questions about ADHD diagnosis, etiology, and medication adverse effects can negatively impact the ADHD treatment process. Therefore, patient/family education regarding ADHD and its management is necessary to improve medication adherence, because it helps families attain the knowledge, confidence, and motivation to manage their child’s condition.
Clinicians have an important role in educating patients about70:
- the medications they are taking
- why they are taking them
- what the medications look like
- the time of medication administration
- the potential adverse effects
- what to do if adverse effects occur
- what regular testing/monitoring is necessary.
Clinicians can provide appropriate psychoeducation by sharing written materials and trusted websites with families (see Related Resources).
Behavioral strategies. Behavioral interventions have been among the most effective strategies for improving medication adherence in other chronic conditions.71 Behavioral strategies are likely to be particularly important for families of children with ADHD and comorbid conditions such as ODD because these families experience considerable caregiver-child conflict.72 Moreover, parents of children with ADHD are at higher risk for having ADHD and depression themselves,73 both of which may interfere with a parent’s ability to obtain and administer medications consistently. Thus, for these families, using a combination of psychoeducation and behavioral strategies will be necessary to affect change in attitude and behavior. Behavioral strategies that can be used to improve medication adherence include:
- Technology-based interventions can reduce the impact of environmental barriers to adherence. For example, pharmacy automatic prescription renewal systems can reduce the likelihood of families failing to obtain ADHD medication refills. Pill reminder boxes, smartphone alerts, and setting various alarms can effectively prompt caregivers/patients to administer medication. In particular, these methods can be crucial in families for which multiple members have ADHD and its attendant difficulties with organization and task completion.
- Caregiver training may assist families in developing specific behavioral management skills that support adherence. This training can be as straightforward as instructing caregivers on the use of positive reinforcement when teaching their children to swallow pills. It may also encompass structured behavioral interventions aimed at training caregivers to utilize rewards and consequences in order to maximize medication adherence.74
Continue to: Clinician interventions
Clinician interventions. Clinicians can use decision aids to help inform families about treatment options, promote shared decision making, and decrease uncertainty about the treatment plan75 (see Related Resources). Early titration of ADHD medications and early first contact (within months of starting medication treatment) between caregivers and clinicians, whether via in-person visit, telephone, or email, have also been related to improved adherence.28 Furthermore, clinicians can improve adherence by prescribing a simplified medication regimen (ie, long-acting formulations that provide full-day coverage). To address the negative impact of high out-of-pocket ADHD medication costs on adherence, clinicians can also prescribe generic preparations and/or “preferred” medications options on an individual patient’s formulary.
Because clinician knowledge and expertise in ADHD care has been linked to improved patient medication adherence,68 clinicians are encouraged to use the American Academy of Pediatrics (AAP) guideline for diagnosis and treatment of ADHD, which includes a supplemental process of care algorithm (last published in 2011,10 with an updated guideline anticipated in 2019), as well as the AAP/National Institute for Children’s Health Quality (NICHQ) ADHD Toolkit,76 which includes items helpful for ADHD diagnosis and treatment. The Society for Developmental and Behavioral Pediatrics is also developing a clinical practice guideline for the diagnosis and treatment of complex ADHD (ie, ADHD complicated by coexisting mental health, developmental, and/or psychosocial conditions or issues), with publication anticipated in 2019. Primary care providers can also improve their expertise in ADHD care by pursuing additional mental health–related trainings (such as those conducted by the REACH Institute).77
Because receiving ADHD care from a specialist has been shown to improve medication initiation and adherence,62,69 other strategies to address the short supply of child psychiatrists include offering incentives to medical students to pursue a career in child psychiatry (eg, loan forgiveness). Telepsychiatry and co-location of mental health specialists and primary care providers are additional innovative ways in which ADHD specialty care can be delivered to more patients.64
Finally, providing culturally-sensitive care can strengthen the clinician-caregiver relationship and promote adherence to treatment. For example, clinicians can partner with local groups to increase their understanding of how different racial/ethnic groups perceive ADHD and its treatment.64
Peer support models. Peers are credible role models who have a valued role in facilitating the use of mental health services by empowering families and enhancing service satisfaction.78 In several communities in the United States, peer models using family advocates have been introduced.79 Family advocates are typically caregivers of children who have special needs or have been involved in the mental health system. Their perspective—as peers and first-hand consumers of the health care and/or mental health system—can make them powerful and effective coaches to families of children with ADHD. By helping families to navigate ADHD care systems successfully, family advocates can play an important role in enhancing ADHD medication adherence, although further investigation is needed. In addition, the stigma around ADHD medication use, which adversely impacts adherence, can be mitigated if caregivers participate in organized ADHD-related support groups (eg, Children and Adults with ADHD [CHADD]).
Continue to: Health disparity-reducing interventions
Health disparity-reducing interventions. Successful health disparity-reducing interventions—such as those developed to enhance care of other chronic disorders including asthma and diabetes—can be applied to improve ADHD care. These interventions, which include medical-legal partnerships (eg, between clinicians, social workers, legal advocates, and community partners) in primary care centers, have been shown to improve health insurance coverage and therefore health care access.80,81 Although some hardships linked to nonadherence (eg, low socioeconomic status) may not be amenable to health care–related interventions, screening for these hardships can identify children who are most at risk for poor adherence. This would alert clinicians to proactively identify barriers to adherence and implement mitigation strategies. This might include developing more streamlined, easier-to-follow management plans for these patients, such as those that can be delivered through pharmacist-physician collaborative programs82 and school-based therapy programs.83-85
Bottom Line
Suboptimal adherence to medications for attention-deficit/hyperactivity disorder (ADHD) can be addressed through patient/family education, behavioral strategies, clinician interventions, peer support models, and health disparity-reducing interventions. By improving ADHD treatment adherence, these interventions have the potential to maximize long-term outcomes.
Related Resources
- Cohen Children’s Medical Center Northwell Health. The ADHD Medication Guide. www.ADHDMedicationGuide.com. Revised December 31, 2017.
- Cincinnati Children’s Hospital Medical Center. Decision aids to facilitate shared decision making in practice. www.cincinnatichildrens.org/service/j/anderson-center/ evidence-based-care/decision-aids.
- CHADD. Children and Adults with attention-deficit/ hyperactivity disorder. www.chadd.org.
Drug Brand Name
Methylphenidate • Concerta, Ritalin
1. Froehlich TE, Lanphear BP, Epstein JN, et al. Prevalence, recognition, and treatment of attention-deficit/hyperactivity disorder in a national sample of US children. Arch Pediatr Adolesc Med. 2007;161(9):857-864.
2. Visser SN, Lesesne CA, Perou R. National estimates and factors associated with medication treatment for childhood attention-deficit/hyperactivity disorder. Pediatrics. 2007;119 (Suppl 1):S99-S106.
3. Danielson ML, Bitsko RH, Ghandour RM, et al. Prevalence of parent-reported ADHD diagnosis and associated treatment among U.S. children and adolescents, 2016. J Clin Child Adolesc Psychol. 2018;47(2):199-212.
4. Molina BS, Hinshaw SP, Swanson JM, et al. The MTA at 8 years: prospective follow-up of children treated for combined-type ADHD in a multisite study. J Am Acad Child Adolesc Psychiatry. 2009;48(5):484-500.
5. Charach A, Dashti B, Carson P, et al. Attention deficit hyperactivity disorder: effectiveness of treatment in at-risk preschoolers; long-term effectiveness in all ages; and variability in prevalence, diagnosis, and treatment. Rockville, MD: Agency for Healthcare Research and Quality; 2011. http://www.ncbi.nlm.nih.gov/books/NBK82368/.
6. Wehmeier PM, Schacht A, Barkley RA. Social and emotional impairment in children and adolescents with ADHD and the impact on quality of life. J Adolesc Health. 2010;46(3):209-217.
7. Barkley RA, Fischer M, Smallish L, et al. Young adult outcome of hyperactive children: adaptive functioning in major life activities. J Am Acad Child Adolesc Psychiatry. 2006;45(2):192-202.
8. Spencer TJ, Biederman J, Mick E. Attention-deficit/hyperactivity disorder: diagnosis, lifespan, comorbidities, and neurobiology. J Pediatr Psychol. 2007;32(6):631-642.
9. Pliszka S, the AACAP Work Group on Quality Issues. Practice parameter for the assessment and treatment of children and adolescents with attention-deficit/hyperactivity disorder. J Am Acad Child Adolesc Psychiatry. 2007;46(7):894-921.
10. Subcommittee on Attention-Deficit/Hyperactivity Disorder; Steering Committee on Quality Improvement and Management. ADHD: clinical practice guideline for the diagnosis, evaluation, and treatment of attention-deficit/hyperactivity disorder in children and adolescents. Pediatrics. 2011;128(5):1007-1022.
11. A 14-month randomized clinical trial of treatment strategies for attention-deficit/hyperactivity disorder. The MTA Cooperative Group. Multimodal Treatment Study of Children with ADHD. Arch Gen Psychiatry. 1999;56(12):1073-1086.
12. Abikoff H, Hechtman L, Klein RG, et al. Symptomatic improvement in children with ADHD treated with long-term methylphenidate and multimodal psychosocial treatment. J Am Acad Child Adolesc Psychiatry. 2004;43(7):802-811.
13. Barbaresi WJ, Katusic SK, Colligan RC, et al. Long-term school outcomes for children with attention-deficit/hyperactivity disorder: a population-based perspective. J Dev Behav Pediatr. 2007;28(4):265-273.
14. Scheffler RM, Brown TT, Fulton BD, et al. Positive association between attention-deficit/ hyperactivity disorder medication use and academic achievement during elementary school. Pediatrics. 2009;123(5):1273-1279.
15. Dalsgaard S, Nielsen HS, Simonsen M. Five-fold increase in national prevalence rates of attention-deficit/hyperactivity disorder medications for children and adolescents with autism spectrum disorder, attention-deficit/hyperactivity disorder, and other psychiatric disorders: a Danish register-based study. J Child Adolesc Psychopharmacol. 2013;23(7):432-439.
16. Lichtenstein P, Halldner L, Zetterqvist J, et al. Medication for attention deficit-hyperactivity disorder and criminality. N Engl J Med. 2012;367(21):2006-2014.
17. Chang Z, Lichtenstein P, D’Onofrio BM, et al. Serious transport accidents in adults with attention-deficit/hyperactivity disorder and the effect of medication: a population-based study. JAMA Psychiatry. 2014;71(3):319-325.
18. Chang Z, Quinn PD, Hur K, et al. Association between medication use for attention-deficit/hyperactivity disorder and risk of motor vehicle crashes. JAMA Psychiatry. 2017;74(6):597-603.
19. Dalsgaard S, Leckman JF, Mortensen PB, et al. Effect of drugs on the risk of injuries in children with attention deficit hyperactivity disorder: a prospective cohort study. Lancet Psychiatry. 2015;2(8):702-709.
20. Chang Z, Lichtenstein P, Halldner L, et al. Stimulant ADHD medication and risk for substance abuse. J Child Psychol Psychiatry. 2014;55(8):878-885.
21. Fischer M, Barkley RA. Childhood stimulant treatment and risk for later substance abuse. J Clin Psychiatry. 2003;64(Suppl 11):19-23.
22. Biederman J. Pharmacotherapy for attention-deficit/hyperactivity disorder (ADHD) decreases the risk for substance abuse: findings from a longitudinal follow-up of youths with and without ADHD. J Clin Psychiatry. 2003;64(Suppl 11):3-8.
23. Chang Z, D’Onofrio BM, Quinn PD, et al. Medicationfor attention-deficit/hyperactivity disorder and risk for depression: a nationwide longitudinal cohort study. Biol Psychiatry. 2016;80(12):916-922.
24. World Health Organization. Adherence to long-term therapies: evidence for action. https://www.who.int/chp/knowledge/publications/adherence_full_report.pdf?ua=1. Published 2003. Accessed July 22, 2019.
25. Perwien A, Hall J, Swensen A, et al. Stimulant treatment patterns and compliance in children and adults with newly treated attention-deficit/hyperactivity disorder. J Manag Care Pharm. 2004;10(2):122-129.
26. Faraone SV, Biederman J, Zimmerman B. An analysis of patient adherence to treatment during a 1-year, open-label study of OROS methylphenidate in children with ADHD. J Atten Disord. 2007;11(2):157-166.
27. Barner JC, Khoza S, Oladapo A. ADHD medication use, adherence, persistence and cost among Texas Medicaid children. Curr Med Res Opin. 2011;27(Suppl 2):13-22.
28. Brinkman WB, Baum R, Kelleher KJ, et al. Relationship between attention-deficit/hyperactivity disorder care and medication continuity. J Am Acad Child Adolesc Psychiatry. 2016;55(4):289-294.
29. Bokhari FAS, Heiland F, Levine P, et al. Risk factors for discontinuing drug therapy among children with ADHD. Health Services and Outcomes Research Methodology. 2008;8(3):134-158.
30. Thiruchelvam D, Charach A, Schachar RJ. Moderators and mediators of long-term adherence to stimulant treatment in children with ADHD. J Am Acad Child Adolesc Psychiatry. 2001;40(8):922-928.
31. DosReis S, Mychailyszyn MP, Evans-Lacko SE, et al. The meaning of attention-deficit/hyperactivity disorder medication and parents’ initiation and continuity of treatment for their child. J Child Adolesc Psychopharmacol. 2009;19(4):377-383.
32. dosReis S, Myers MA. Parental attitudes and involvement in psychopharmacological treatment for ADHD: a conceptual model. Int Rev Psychiatry. 2008;20(2):135-141.
33. Bussing R, Koro-Ljungberg M, Noguchi K, et al. Willingness to use ADHD treatments: a mixed methods study of perceptions by adolescents, parents, health professionals and teachers. Soc Sci Med. 2012;74(1):92-100.
34. Brinkman WB, Sucharew H, Majcher JH, et al. Predictors of medication continuity in children with ADHD. Pediatrics. 2018;141(6). doi: 10.1542/peds.2017-2580.
35. Coletti DJ, Pappadopulos E, Katsiotas NJ, et al. Parent perspectives on the decision to initiate medication treatment of attention-deficit/hyperactivity disorder. J Child Adolesc Psychopharmacol. 2012;22(3):226-237.
36. Bussing R, Gary FA. Practice guidelines and parental ADHD treatment evaluations: friends or foes? Harv Rev Psychiatry. 2001;9(5):223-233.
37. Charach A, Gajaria A. Improving psychostimulant adherence in children with ADHD. Expert Rev Neurother. 2008;8(10):1563-1571.
38. Rieppi R, Greenhill LL, Ford RE, et al. Socioeconomic status as a moderator of ADHD treatment outcomes. J Am Acad Child Adolesc Psychiatry. 2002;41(3):269-277.
39. Swanson JM, Hinshaw SP, Arnold LE, et al. Secondary evaluations of MTA 36-month outcomes: propensity score and growth mixture model analyses. J Am Acad Child Adolesc Psychiatry. 2007;46(8):1003-1014.
40. Gau SS, Shen HY, Chou MC, et al. Determinants of adherence to methylphenidate and the impact of poor adherence on maternal and family measures. J Child Adolesc Psychopharmacol. 2006;16(3):286-297.
41. Barkley RA, Fischer M, Edelbrock C, et al. The adolescent outcome of hyperactive children diagnosed by research criteria--III. Mother-child interactions, family conflicts and maternal psychopathology. J Child Psychol Psychiatry. 1991;32(2):233-255.
42. Kashdan TB, Jacob RG, Pelham WE, et al. Depression and anxiety in parents of children with ADHD and varying levels of oppositional defiant behaviors: modeling relationships with family functioning. J Clin Child Adolesc Psychol. 2004;33(1):169-181.
43. Chavira DA, Stein MB, Bailey K, et al. Parental opinions regarding treatment for social anxiety disorder in youth. J Dev Behav Pediatr. 2003;24(5):315-322.
44. Leslie LK, Aarons GA, Haine RA, et al. Caregiver depression and medication use by youths with ADHD who receive services in the public sector. Psychiatr Serv. 2007;58(1):131-134.
45. Barbaresi WJ, Katusic SK, Colligan RC, et al. Long-term stimulant medication treatment of attention-deficit/hyperactivity disorder: results from a population-based study. J Dev Behav Pediatr. 2006;27(1):1-10.
46. Atzori P, Usala T, Carucci S, et al. Predictive factors for persistent use and compliance of immediate-release methylphenidate: a 36-month naturalistic study. J Child Adolesc Psychopharmacol. 2009;19(6):673-681.
47. Chen CY, Yeh HH, Chen KH, et al. Differential effects of predictors on methylphenidate initiation and discontinuation among young people with newly diagnosed attention-deficit/hyperactivity disorder. J Child Adolesc Psychopharmacol. 2011;21(3):265-273.
48. Winterstein AG, Gerhard T, Shuster J, et al. Utilization of pharmacologic treatment in youths with attention deficit/hyperactivity disorder in Medicaid database. Ann Pharmacother. 2008;42(1):24-31.
49. Marcus SC, Wan GJ, Kemner JE, et al. Continuity of methylphenidate treatment for attention-deficit/hyperactivity disorder. Arch Pediatr Adolesc Med. 2005;159(6):572-578.
50. Cummings JR JX, Allen L, Lally C, et al. Racial and ethnic differences in ADHD treatment quality among Medicaid-enrolled youth. Pediatrics. 2017;139(6):e2016-e2044.
51. Hudson JL, Miller GE, Kirby JB. Explaining racial and ethnic differences in children’s use of stimulant medications. Med Care. 2007;45(11):1068-1075.
52. van den Ban E, Souverein PC, Swaab H, et al. Less discontinuation of ADHD drug use since the availability of long-acting ADHD medication in children, adolescents and adults under the age of 45 years in the Netherlands. Atten Defic Hyperact Disord. 2010;2(4):213-220.
53. Charach A, Ickowicz A, Schachar R. Stimulant treatment over five years: adherence, effectiveness, and adverse effects. J Am Acad Child Adolesc Psychiatry. 2004;43(5):559-567.
54. Toomey SL, Sox CM, Rusinak D, et al. Why do children with ADHD discontinue their medication? Clin Pediatr (Phila). 2012;51(8):763-769.
55. Brinkman WB, Simon JO, Epstein JN. Reasons why children and adolescents with attention-deficit/hyperactivity disorder stop and restart taking medicine. Acad Pediatr. 2018;18(3):273-280.
56. Wehmeier PM, Dittmann RW, Banaschewski T. Treatment compliance or medication adherence in children and adolescents on ADHD medication in clinical practice: results from the COMPLY observational study. Atten Defic Hyperact Disord. 2015;7(2):165-174.
57. Frank E, Ozon C, Nair V, et al. Examining why patients with attention-deficit/hyperactivity disorder lack adherence to medication over the long term: a review and analysis. J Clin Psychiatry. 2015;76(11):e1459-e1468.
58. Pozzi M, Carnovale C, Peeters G, et al. Adverse drug events related to mood and emotion in paediatric patients treated for ADHD: a meta-analysis. J Affect Disord. 2018;238:161-178.
59. Stuckelman ZD, Mulqueen JM, Ferracioli-Oda E, et al. Risk of irritability with psychostimulant treatment in children with ADHD: a meta-analysis. J Clin Psychiatry. 2017;78(6):e648-e655.
60. Cortese S, Adamo N, Del Giovane C, et al. Comparative efficacy and tolerability of medications for attention-deficit hyperactivity disorder in children, adolescents, and adults: a systematic review and network meta-analysis. Lancet Psychiatry. 2018;5(9):727-738.
61. Lawson KA, Johnsrud M, Hodgkins P, et al. Utilization patterns of stimulants in ADHD in the Medicaid population: a retrospective analysis of data from the Texas Medicaid program. Clin Ther. 2012;34(4):944-956 e944.
62. Olfson M, Marcus S, Wan G. Stimulant dosing for children with ADHD: a medical claims analysis. J Am Acad Child Adolesc Psychiatry. 2009;48(1):51-59.
63. Jensen PS, Arnold LE, Swanson JM, et al. 3-year follow-up of the NIMH MTA study. J Am Acad Child Adolesc Psychiatry. 2007;46(8):989-1002.
64. Van Cleave J, Leslie LK. Approaching ADHD as a chronic condition: implications for long-term adherence. Pediatr Ann. 2008;37(1):19-26.
65. Leslie LK, Plemmons D, Monn AR, et al. Investigating ADHD treatment trajectories: listening to families’ stories about medication use. J Dev Behav Pediatr. 2007;28(3):179-188.
66. Fiks AG, Mayne S, Localio AR, et al. Shared decision making and behavioral impairment: a national study among children with special health care needs. BMC Pediatr. 2012;12:153.
67. Stevens J, Harman JS, Kelleher KJ. Race/ethnicity and insurance status as factors associated with ADHD treatment patterns. J Child Adolesc Psychopharmacol. 2005;15(1):88-96.
68. Charach A, Skyba A, Cook L, et al. Using stimulant medication for children with ADHD: what do parents say? A brief report. J Can Acad Child Adolesc Psychiatry. 2006;15(2):75-83.
69. Chen CY, Gerhard T, Winterstein AG. Determinants of initial pharmacological treatment for youths with attention-deficit/hyperactivity disorder. J Child Adolescent Psychopharmacol. 2009;19(2):187-195.
70. National Council on Patient Information and Education. Enhancing prescription medication adherence: a national action plan. http://www.bemedwise.org/docs/enhancingprescriptionmedicineadherence.pdf. Published August 2007. Accessed July 22, 2019.
71. Kahana S, Drotar D, Frazier T. Meta-analysis of psychological interventions to promote adherence to treatment in pediatric chronic health conditions. J Pediatr Psychol. 2008;33(6):590-611.
72. Johnston C, Mash EJ. Families of children with attention-deficit/hyperactivity disorder: review and recommendations for future research. Clin Child Fam Psychol Rev. 2001;4(3):183-207.
73. Chronis AM, Lahey BB, Pelham WE Jr., et al. Psychopathology and substance abuse in parents of young children with attention-deficit/hyperactivity disorder. J Am Acad Child Adolesc Psychiatry. 2003;42(12):1424-1432.
74. Chacko A, Newcorn JH, Feirsen N, et al. Improving medication adherence in chronic pediatric health conditions: a focus on ADHD in youth. Curr Pharm Des. 2010;16(22):2416-2423.
75. Brinkman WB, Hartl Majcher J, Polling LM, et al. Shared decision-making to improve attention-deficit hyperactivity disorder care. Patient Educ Couns. 2013;93(1):95-101.
76. American Academy of Pediatrics. Caring for children with ADHD: a resource toolkit for clinicians. 2nd ed. https://www.aap.org/en-us/pubserv/adhd2/Pages/default.aspx. Published 2011. Accessed July 22, 2019.
77. The REACH Institute. Course dates and registration. http://www.thereachinstitute.org/services/for-primary-care-practitioners/training-dates-and-registration. Accessed July 22, 2019.
78. Sells D, Davidson L, Jewell C, et al. The treatment relationship in peer-based and regular case management for clients with severe mental illness. Psychiatr Serv. 2006;57(8):1179-1184.
79. Hoagwood KE, Green E, Kelleher K, et al. Family advocacy, support and education in children’s mental health: results of a national survey. Adm Policy Ment Health. 2008;35(1-2):73-83.
80. Klein MD, Beck AF, Henize AW, et al. Doctors and lawyers collaborating to HeLP children—outcomes from a successful partnership between professions. J Health Care Poor Underserved. 2013;24(3):1063-1073.
81. Weintraub D, Rodgers MA, Botcheva L, et al. Pilot study of medical-legal partnership to address social and legal needs of patients. J Health Care Poor Underserved. 2010;21(Suppl 2):157-168.
82. Bradley CL, Luder HR, Beck AF, et al. Pediatric asthma medication therapy management through community pharmacy and primary care collaboration. J Am Pharm Assoc (2003). 2016;56(4):455-460.
83. Noyes K, Bajorska A, Fisher S, et al. Cost-effectiveness of the school-based asthma therapy (SBAT) program. Pediatrics. 2013;131(3):e709-e717.
84. Halterman JS, Fagnano M, Montes G, et al. The school-based preventive asthma care trial: results of a pilot study. J Pediatr. 2012;161(6):1109-1115.
85. Halterman JS, Szilagyi PG, Fisher SG, et al. Randomized controlled trial to improve care for urban children with asthma: results of the school-based asthma therapy trial. Arch Pediatr Adolesc Med. 2011;165(3):262-268.
1. Froehlich TE, Lanphear BP, Epstein JN, et al. Prevalence, recognition, and treatment of attention-deficit/hyperactivity disorder in a national sample of US children. Arch Pediatr Adolesc Med. 2007;161(9):857-864.
2. Visser SN, Lesesne CA, Perou R. National estimates and factors associated with medication treatment for childhood attention-deficit/hyperactivity disorder. Pediatrics. 2007;119 (Suppl 1):S99-S106.
3. Danielson ML, Bitsko RH, Ghandour RM, et al. Prevalence of parent-reported ADHD diagnosis and associated treatment among U.S. children and adolescents, 2016. J Clin Child Adolesc Psychol. 2018;47(2):199-212.
4. Molina BS, Hinshaw SP, Swanson JM, et al. The MTA at 8 years: prospective follow-up of children treated for combined-type ADHD in a multisite study. J Am Acad Child Adolesc Psychiatry. 2009;48(5):484-500.
5. Charach A, Dashti B, Carson P, et al. Attention deficit hyperactivity disorder: effectiveness of treatment in at-risk preschoolers; long-term effectiveness in all ages; and variability in prevalence, diagnosis, and treatment. Rockville, MD: Agency for Healthcare Research and Quality; 2011. http://www.ncbi.nlm.nih.gov/books/NBK82368/.
6. Wehmeier PM, Schacht A, Barkley RA. Social and emotional impairment in children and adolescents with ADHD and the impact on quality of life. J Adolesc Health. 2010;46(3):209-217.
7. Barkley RA, Fischer M, Smallish L, et al. Young adult outcome of hyperactive children: adaptive functioning in major life activities. J Am Acad Child Adolesc Psychiatry. 2006;45(2):192-202.
8. Spencer TJ, Biederman J, Mick E. Attention-deficit/hyperactivity disorder: diagnosis, lifespan, comorbidities, and neurobiology. J Pediatr Psychol. 2007;32(6):631-642.
9. Pliszka S, the AACAP Work Group on Quality Issues. Practice parameter for the assessment and treatment of children and adolescents with attention-deficit/hyperactivity disorder. J Am Acad Child Adolesc Psychiatry. 2007;46(7):894-921.
10. Subcommittee on Attention-Deficit/Hyperactivity Disorder; Steering Committee on Quality Improvement and Management. ADHD: clinical practice guideline for the diagnosis, evaluation, and treatment of attention-deficit/hyperactivity disorder in children and adolescents. Pediatrics. 2011;128(5):1007-1022.
11. A 14-month randomized clinical trial of treatment strategies for attention-deficit/hyperactivity disorder. The MTA Cooperative Group. Multimodal Treatment Study of Children with ADHD. Arch Gen Psychiatry. 1999;56(12):1073-1086.
12. Abikoff H, Hechtman L, Klein RG, et al. Symptomatic improvement in children with ADHD treated with long-term methylphenidate and multimodal psychosocial treatment. J Am Acad Child Adolesc Psychiatry. 2004;43(7):802-811.
13. Barbaresi WJ, Katusic SK, Colligan RC, et al. Long-term school outcomes for children with attention-deficit/hyperactivity disorder: a population-based perspective. J Dev Behav Pediatr. 2007;28(4):265-273.
14. Scheffler RM, Brown TT, Fulton BD, et al. Positive association between attention-deficit/ hyperactivity disorder medication use and academic achievement during elementary school. Pediatrics. 2009;123(5):1273-1279.
15. Dalsgaard S, Nielsen HS, Simonsen M. Five-fold increase in national prevalence rates of attention-deficit/hyperactivity disorder medications for children and adolescents with autism spectrum disorder, attention-deficit/hyperactivity disorder, and other psychiatric disorders: a Danish register-based study. J Child Adolesc Psychopharmacol. 2013;23(7):432-439.
16. Lichtenstein P, Halldner L, Zetterqvist J, et al. Medication for attention deficit-hyperactivity disorder and criminality. N Engl J Med. 2012;367(21):2006-2014.
17. Chang Z, Lichtenstein P, D’Onofrio BM, et al. Serious transport accidents in adults with attention-deficit/hyperactivity disorder and the effect of medication: a population-based study. JAMA Psychiatry. 2014;71(3):319-325.
18. Chang Z, Quinn PD, Hur K, et al. Association between medication use for attention-deficit/hyperactivity disorder and risk of motor vehicle crashes. JAMA Psychiatry. 2017;74(6):597-603.
19. Dalsgaard S, Leckman JF, Mortensen PB, et al. Effect of drugs on the risk of injuries in children with attention deficit hyperactivity disorder: a prospective cohort study. Lancet Psychiatry. 2015;2(8):702-709.
20. Chang Z, Lichtenstein P, Halldner L, et al. Stimulant ADHD medication and risk for substance abuse. J Child Psychol Psychiatry. 2014;55(8):878-885.
21. Fischer M, Barkley RA. Childhood stimulant treatment and risk for later substance abuse. J Clin Psychiatry. 2003;64(Suppl 11):19-23.
22. Biederman J. Pharmacotherapy for attention-deficit/hyperactivity disorder (ADHD) decreases the risk for substance abuse: findings from a longitudinal follow-up of youths with and without ADHD. J Clin Psychiatry. 2003;64(Suppl 11):3-8.
23. Chang Z, D’Onofrio BM, Quinn PD, et al. Medicationfor attention-deficit/hyperactivity disorder and risk for depression: a nationwide longitudinal cohort study. Biol Psychiatry. 2016;80(12):916-922.
24. World Health Organization. Adherence to long-term therapies: evidence for action. https://www.who.int/chp/knowledge/publications/adherence_full_report.pdf?ua=1. Published 2003. Accessed July 22, 2019.
25. Perwien A, Hall J, Swensen A, et al. Stimulant treatment patterns and compliance in children and adults with newly treated attention-deficit/hyperactivity disorder. J Manag Care Pharm. 2004;10(2):122-129.
26. Faraone SV, Biederman J, Zimmerman B. An analysis of patient adherence to treatment during a 1-year, open-label study of OROS methylphenidate in children with ADHD. J Atten Disord. 2007;11(2):157-166.
27. Barner JC, Khoza S, Oladapo A. ADHD medication use, adherence, persistence and cost among Texas Medicaid children. Curr Med Res Opin. 2011;27(Suppl 2):13-22.
28. Brinkman WB, Baum R, Kelleher KJ, et al. Relationship between attention-deficit/hyperactivity disorder care and medication continuity. J Am Acad Child Adolesc Psychiatry. 2016;55(4):289-294.
29. Bokhari FAS, Heiland F, Levine P, et al. Risk factors for discontinuing drug therapy among children with ADHD. Health Services and Outcomes Research Methodology. 2008;8(3):134-158.
30. Thiruchelvam D, Charach A, Schachar RJ. Moderators and mediators of long-term adherence to stimulant treatment in children with ADHD. J Am Acad Child Adolesc Psychiatry. 2001;40(8):922-928.
31. DosReis S, Mychailyszyn MP, Evans-Lacko SE, et al. The meaning of attention-deficit/hyperactivity disorder medication and parents’ initiation and continuity of treatment for their child. J Child Adolesc Psychopharmacol. 2009;19(4):377-383.
32. dosReis S, Myers MA. Parental attitudes and involvement in psychopharmacological treatment for ADHD: a conceptual model. Int Rev Psychiatry. 2008;20(2):135-141.
33. Bussing R, Koro-Ljungberg M, Noguchi K, et al. Willingness to use ADHD treatments: a mixed methods study of perceptions by adolescents, parents, health professionals and teachers. Soc Sci Med. 2012;74(1):92-100.
34. Brinkman WB, Sucharew H, Majcher JH, et al. Predictors of medication continuity in children with ADHD. Pediatrics. 2018;141(6). doi: 10.1542/peds.2017-2580.
35. Coletti DJ, Pappadopulos E, Katsiotas NJ, et al. Parent perspectives on the decision to initiate medication treatment of attention-deficit/hyperactivity disorder. J Child Adolesc Psychopharmacol. 2012;22(3):226-237.
36. Bussing R, Gary FA. Practice guidelines and parental ADHD treatment evaluations: friends or foes? Harv Rev Psychiatry. 2001;9(5):223-233.
37. Charach A, Gajaria A. Improving psychostimulant adherence in children with ADHD. Expert Rev Neurother. 2008;8(10):1563-1571.
38. Rieppi R, Greenhill LL, Ford RE, et al. Socioeconomic status as a moderator of ADHD treatment outcomes. J Am Acad Child Adolesc Psychiatry. 2002;41(3):269-277.
39. Swanson JM, Hinshaw SP, Arnold LE, et al. Secondary evaluations of MTA 36-month outcomes: propensity score and growth mixture model analyses. J Am Acad Child Adolesc Psychiatry. 2007;46(8):1003-1014.
40. Gau SS, Shen HY, Chou MC, et al. Determinants of adherence to methylphenidate and the impact of poor adherence on maternal and family measures. J Child Adolesc Psychopharmacol. 2006;16(3):286-297.
41. Barkley RA, Fischer M, Edelbrock C, et al. The adolescent outcome of hyperactive children diagnosed by research criteria--III. Mother-child interactions, family conflicts and maternal psychopathology. J Child Psychol Psychiatry. 1991;32(2):233-255.
42. Kashdan TB, Jacob RG, Pelham WE, et al. Depression and anxiety in parents of children with ADHD and varying levels of oppositional defiant behaviors: modeling relationships with family functioning. J Clin Child Adolesc Psychol. 2004;33(1):169-181.
43. Chavira DA, Stein MB, Bailey K, et al. Parental opinions regarding treatment for social anxiety disorder in youth. J Dev Behav Pediatr. 2003;24(5):315-322.
44. Leslie LK, Aarons GA, Haine RA, et al. Caregiver depression and medication use by youths with ADHD who receive services in the public sector. Psychiatr Serv. 2007;58(1):131-134.
45. Barbaresi WJ, Katusic SK, Colligan RC, et al. Long-term stimulant medication treatment of attention-deficit/hyperactivity disorder: results from a population-based study. J Dev Behav Pediatr. 2006;27(1):1-10.
46. Atzori P, Usala T, Carucci S, et al. Predictive factors for persistent use and compliance of immediate-release methylphenidate: a 36-month naturalistic study. J Child Adolesc Psychopharmacol. 2009;19(6):673-681.
47. Chen CY, Yeh HH, Chen KH, et al. Differential effects of predictors on methylphenidate initiation and discontinuation among young people with newly diagnosed attention-deficit/hyperactivity disorder. J Child Adolesc Psychopharmacol. 2011;21(3):265-273.
48. Winterstein AG, Gerhard T, Shuster J, et al. Utilization of pharmacologic treatment in youths with attention deficit/hyperactivity disorder in Medicaid database. Ann Pharmacother. 2008;42(1):24-31.
49. Marcus SC, Wan GJ, Kemner JE, et al. Continuity of methylphenidate treatment for attention-deficit/hyperactivity disorder. Arch Pediatr Adolesc Med. 2005;159(6):572-578.
50. Cummings JR JX, Allen L, Lally C, et al. Racial and ethnic differences in ADHD treatment quality among Medicaid-enrolled youth. Pediatrics. 2017;139(6):e2016-e2044.
51. Hudson JL, Miller GE, Kirby JB. Explaining racial and ethnic differences in children’s use of stimulant medications. Med Care. 2007;45(11):1068-1075.
52. van den Ban E, Souverein PC, Swaab H, et al. Less discontinuation of ADHD drug use since the availability of long-acting ADHD medication in children, adolescents and adults under the age of 45 years in the Netherlands. Atten Defic Hyperact Disord. 2010;2(4):213-220.
53. Charach A, Ickowicz A, Schachar R. Stimulant treatment over five years: adherence, effectiveness, and adverse effects. J Am Acad Child Adolesc Psychiatry. 2004;43(5):559-567.
54. Toomey SL, Sox CM, Rusinak D, et al. Why do children with ADHD discontinue their medication? Clin Pediatr (Phila). 2012;51(8):763-769.
55. Brinkman WB, Simon JO, Epstein JN. Reasons why children and adolescents with attention-deficit/hyperactivity disorder stop and restart taking medicine. Acad Pediatr. 2018;18(3):273-280.
56. Wehmeier PM, Dittmann RW, Banaschewski T. Treatment compliance or medication adherence in children and adolescents on ADHD medication in clinical practice: results from the COMPLY observational study. Atten Defic Hyperact Disord. 2015;7(2):165-174.
57. Frank E, Ozon C, Nair V, et al. Examining why patients with attention-deficit/hyperactivity disorder lack adherence to medication over the long term: a review and analysis. J Clin Psychiatry. 2015;76(11):e1459-e1468.
58. Pozzi M, Carnovale C, Peeters G, et al. Adverse drug events related to mood and emotion in paediatric patients treated for ADHD: a meta-analysis. J Affect Disord. 2018;238:161-178.
59. Stuckelman ZD, Mulqueen JM, Ferracioli-Oda E, et al. Risk of irritability with psychostimulant treatment in children with ADHD: a meta-analysis. J Clin Psychiatry. 2017;78(6):e648-e655.
60. Cortese S, Adamo N, Del Giovane C, et al. Comparative efficacy and tolerability of medications for attention-deficit hyperactivity disorder in children, adolescents, and adults: a systematic review and network meta-analysis. Lancet Psychiatry. 2018;5(9):727-738.
61. Lawson KA, Johnsrud M, Hodgkins P, et al. Utilization patterns of stimulants in ADHD in the Medicaid population: a retrospective analysis of data from the Texas Medicaid program. Clin Ther. 2012;34(4):944-956 e944.
62. Olfson M, Marcus S, Wan G. Stimulant dosing for children with ADHD: a medical claims analysis. J Am Acad Child Adolesc Psychiatry. 2009;48(1):51-59.
63. Jensen PS, Arnold LE, Swanson JM, et al. 3-year follow-up of the NIMH MTA study. J Am Acad Child Adolesc Psychiatry. 2007;46(8):989-1002.
64. Van Cleave J, Leslie LK. Approaching ADHD as a chronic condition: implications for long-term adherence. Pediatr Ann. 2008;37(1):19-26.
65. Leslie LK, Plemmons D, Monn AR, et al. Investigating ADHD treatment trajectories: listening to families’ stories about medication use. J Dev Behav Pediatr. 2007;28(3):179-188.
66. Fiks AG, Mayne S, Localio AR, et al. Shared decision making and behavioral impairment: a national study among children with special health care needs. BMC Pediatr. 2012;12:153.
67. Stevens J, Harman JS, Kelleher KJ. Race/ethnicity and insurance status as factors associated with ADHD treatment patterns. J Child Adolesc Psychopharmacol. 2005;15(1):88-96.
68. Charach A, Skyba A, Cook L, et al. Using stimulant medication for children with ADHD: what do parents say? A brief report. J Can Acad Child Adolesc Psychiatry. 2006;15(2):75-83.
69. Chen CY, Gerhard T, Winterstein AG. Determinants of initial pharmacological treatment for youths with attention-deficit/hyperactivity disorder. J Child Adolescent Psychopharmacol. 2009;19(2):187-195.
70. National Council on Patient Information and Education. Enhancing prescription medication adherence: a national action plan. http://www.bemedwise.org/docs/enhancingprescriptionmedicineadherence.pdf. Published August 2007. Accessed July 22, 2019.
71. Kahana S, Drotar D, Frazier T. Meta-analysis of psychological interventions to promote adherence to treatment in pediatric chronic health conditions. J Pediatr Psychol. 2008;33(6):590-611.
72. Johnston C, Mash EJ. Families of children with attention-deficit/hyperactivity disorder: review and recommendations for future research. Clin Child Fam Psychol Rev. 2001;4(3):183-207.
73. Chronis AM, Lahey BB, Pelham WE Jr., et al. Psychopathology and substance abuse in parents of young children with attention-deficit/hyperactivity disorder. J Am Acad Child Adolesc Psychiatry. 2003;42(12):1424-1432.
74. Chacko A, Newcorn JH, Feirsen N, et al. Improving medication adherence in chronic pediatric health conditions: a focus on ADHD in youth. Curr Pharm Des. 2010;16(22):2416-2423.
75. Brinkman WB, Hartl Majcher J, Polling LM, et al. Shared decision-making to improve attention-deficit hyperactivity disorder care. Patient Educ Couns. 2013;93(1):95-101.
76. American Academy of Pediatrics. Caring for children with ADHD: a resource toolkit for clinicians. 2nd ed. https://www.aap.org/en-us/pubserv/adhd2/Pages/default.aspx. Published 2011. Accessed July 22, 2019.
77. The REACH Institute. Course dates and registration. http://www.thereachinstitute.org/services/for-primary-care-practitioners/training-dates-and-registration. Accessed July 22, 2019.
78. Sells D, Davidson L, Jewell C, et al. The treatment relationship in peer-based and regular case management for clients with severe mental illness. Psychiatr Serv. 2006;57(8):1179-1184.
79. Hoagwood KE, Green E, Kelleher K, et al. Family advocacy, support and education in children’s mental health: results of a national survey. Adm Policy Ment Health. 2008;35(1-2):73-83.
80. Klein MD, Beck AF, Henize AW, et al. Doctors and lawyers collaborating to HeLP children—outcomes from a successful partnership between professions. J Health Care Poor Underserved. 2013;24(3):1063-1073.
81. Weintraub D, Rodgers MA, Botcheva L, et al. Pilot study of medical-legal partnership to address social and legal needs of patients. J Health Care Poor Underserved. 2010;21(Suppl 2):157-168.
82. Bradley CL, Luder HR, Beck AF, et al. Pediatric asthma medication therapy management through community pharmacy and primary care collaboration. J Am Pharm Assoc (2003). 2016;56(4):455-460.
83. Noyes K, Bajorska A, Fisher S, et al. Cost-effectiveness of the school-based asthma therapy (SBAT) program. Pediatrics. 2013;131(3):e709-e717.
84. Halterman JS, Fagnano M, Montes G, et al. The school-based preventive asthma care trial: results of a pilot study. J Pediatr. 2012;161(6):1109-1115.
85. Halterman JS, Szilagyi PG, Fisher SG, et al. Randomized controlled trial to improve care for urban children with asthma: results of the school-based asthma therapy trial. Arch Pediatr Adolesc Med. 2011;165(3):262-268.
Seizure-like episodes, but is it really epilepsy?
CASE Increasingly frequent paroxysmal episodes
Ms. N, age 12, comes to the hospital for evaluation of paroxysmal episodes of pain, weakness, and muscle spasms. A neurologist who evaluated her as an outpatient had recommended a routine electroencephalogram (EEG); after those results were inconclusive, Ms. N’s mother brought her to the hospital for a 24-hour video EEG.
Ms. N has a history of asthma. She has no history of seizures or headache, but her mother has an unspecified seizure disorder that has been stable with antiepileptic medication for many years. Ms. N has no other family history of autoimmune or neurologic disorders.
Ms. N’s episodes began 6 months ago and have progressively increased in frequency from 5 to 12 episodes a day. She says that before she has an episode, she “ feels tingling in her fingers and mouth, and butterflies in her belly,” and then her “whole body clenches up.” She denies experiencing tongue biting, facial or extremity weakness, incontinence, or loss of consciousness during these episodes.
Shortly before her hospitalization, Ms. N had won a scholarship to attend an overnight art camp. Because her episodes were becoming more frequent and their etiology remained unclear, Ms. N and her mother decided it would be unsafe for her to attend, and that she should go to the hospital for evaluation instead.
EVALUATION Tough questions reveal answers
The pediatric team evaluates Ms. N. Her physical exam, laboratory values, and imaging are all within normal limits. Her neurologic exam demonstrates full strength, tone, and sensation in all extremities. All cranial nerves and reflexes are intact. No dysmorphic features or gait abnormalities are noted. All laboratory and imaging tests are normal, including complete blood cell count, electrolytes, calcium, magnesium, phosphorus, glucose, creatine kinase, liver enzymes, urine drug screen, human chorionic gonadotropin (hCG) urine test, and h
After the initial workup, the pediatric team consults the child and adolescent psychiatry team for a complete assessment of Ms. N due to concerns that a psychological component is contributing to her episodes. According to the psychosocial history obtained from Ms. N and her mother, Ms. N had experienced disrupted attachment, trauma, and loss. At age 5, Ms. N was temporarily removed from her mother’s custody after a fight between her mother and brother. At age 9, Ms. N’s stepfather, her primary father figure, died of a brain tumor.
Ms. N also has significant trauma stemming from her relationship with her biological father. Ms. N’s mother reports that her daughter was conceived during nonconsensual sexual intercourse. Ms. N did not have much contact with her biological father until 6 months ago, when he started picking her up at school and taking her to his home for several hours without permission or supervision. Afterwards, Ms. N confided to her mother and a teacher that her father sexually assaulted her during those visits.
Continue to: Ms. N and her mother...
Ms. N and her mother reported the assault to the police and were awaiting legal action.
During the interview with the psychiatry team, Ms. N denies that any thoughts or actions trigger the episodes and reports that she cannot control when they happen. Because she cannot anticipate the episodes, she says she is afraid to leave her house. She does not know why the episodes are happening and feels frustrated that they are getting worse. Ms. N says, “I have been feeling down lately,” but she denies hopelessness, worthlessness, suicidal ideation, homicidal ideation, delusions, or hallucinations.
In the hospital, when the psychiatry team asks Ms. N about her visits with her father, she says that they are “too painful to talk about,” and fears that discussing them will trigger an episode. However, her mother suggests that her daughter’s sexual trauma, as well as ongoing frustrations with the legal system, are influencing her mood; she has had low energy, poor appetite, and is spending more time in bed. Her mother also reports that Ms. N “avoids going out in the sun and spending time with her friends outside. She doesn’t seem to enjoy shopping and art like she used to.” Ms. N told her mother that she was having nightmares about the trauma and “could not stop thinking about some of the bad stuff that happened during the day.”
Ten minutes into the interview, while being questioned about her father, Ms. N experiences a spastic episode. She curls up in bed on her left side, clenches her entire body, and shuts her eyes. Her mother quickly runs to her bedside and counts the seconds until the end of the episode. After 25 seconds, Ms. N awakes with full recollection of the episode. On review of the video EEG during the episode, no ictal patterns are seen.
[polldaddy:10375873]
The authors’ observations
Paroxysmal episodes of weakness, numbness, and muscle spasms in a young female are suggestive of either epilepsy or nonepileptic seizure (NES).1,2 The negative EEG and physical features are inconsistent with epileptiform seizure, and Ms. N’s history and evaluation are suggestive of NES. Nonepileptic seizures are a type of a conversion disorder, or functional neurologic symptom disorder, in which a patient experiences weakness, abnormal movements, or seizure-like episodes that are inconsistent with organic neurologic disease.3 When a diagnosis of conversion disorder is suspected, a clinician must always consider other pathology that can explain the symptoms, such as migraine, vasovagal syncope, or intracranial mass. If a patient has focal neurologic deficits, head imaging should be pursued. Additionally, the clinician must screen for malingering and factitious disorder before establishing a definitive diagnosis. However, conversion disorder is not a diagnosis of exclusion. For example, a negative EEG does not rule out epilepsy, and patients can have both epilepsy and concomitant NES.
Continue to: Although NES is a common...
Although NES is a common type of conversion disorder, it is often difficult to diagnose, manage, and treat. Patients often receive antiepileptic medications but continue to have worsening events that are refractory to treatment. Various clinical features can suggest NES instead of epilepsy. Forced eye closure on video recording is a specific finding suggestive of NES, yet this feature is not sufficient to make the diagnosis.4 A video EEG must be performed to assess for epilepsy. The diagnosis of NES does not exclude the possibility that a patient has epilepsy, as NES can occur in up to 40% of patients with epilepsy.5 A video EEG without ictal patterns before, during, and after an observed episode is diagnostic of NES.6
[polldaddy:10375874]
The authors’ observations
Conversion disorders such as NES are a presentation of neurologic symptoms that cannot be readily accounted for by other conditions and are often associated with antecedent trauma. Multiple factors in Ms. N’s history increase her risk of NES, including loss of multiple loved ones, ongoing legal involvement, and alleged sexual abuse by her father.
Victims of sexual abuse are more likely than the general population to demonstrate symptoms of conversion disorder, especially NES.7,8 The onset of paroxysmal episodes after incestuous abuse in a teenage girl is characteristic of NES. Compared with patients with complex partial epilepsy (CPE), patients with NES are 3 times more likely to report sexual trauma.9,10 Children who report sexual abuse that precedes NES are more likely to have been victimized by a first-degree relative than patients with CPE who report sexual abuse.11 Risk factors for victims developing NES may be related to the severity of adversity, stress sensitivity, and decreased hippocampal volume.12,13
Ms. N endorsed many psychiatric symptoms that accompany her paroxysmal episodes; this is similar to findings in other patients with NES.14 One study found that depression is 3 times more prevalent and PTSD is 8 times more prevalent in patients with NES.12 During the evaluation, Ms. N’s mother said her daughter had low energy, poor appetite, lethargy, and anhedonia for the preceding 5 months, which is consistent with adjustment disorder.3 Her flashbacks, nightmares, difficulty sleeping, and agoraphobia, along with her trouble engaging with the people and activities that used to bring her joy, are symptoms of PTSD. Nonepileptic seizure is often associated with PTSD and can be viewed as an expression of a dissociated subtype.15
In a literature review, Durrant et al16 isolated prognostic indicators for NES (Table16). This study found that 70% of children and 40% of adults achieve remission from NES. Ms. N’s case has multiple concerning features, such as her comorbid psychiatric conditions, ongoing involvement in a legal case, and sexual trauma; this last factor is associated with the most severe symptoms and worse outcomes.16,17 Despite this somber reality, Ms. N has the support of her mother and is relatively young, which play a vital role in recovery.

Continue to: TREATMENT A strategy for minimizing the episodes
TREATMENT A strategy for minimizing the episodes
Ms. N’s medical workup remains unremarkable throughout the rest of her hospital stay. The psychiatry and pediatric teams discuss their assessments and agree that NES is the most likely diagnosis. The psychiatry team counsels Ms. N and her mother on the diagnosis and etiology of NES.
[polldaddy:10375876]
The authors’ observations
Cognitive-behavioral therapy is currently the treatment of choice for reducing seizure frequency in patients with NES.18,19 The use of CBT was suggested due to the theory that NES represents a dissociative response to trauma. Therapy focuses on changing a patient’s beliefs and perceptions associated with attacks.5 A randomized study of 66 patients with NES compared the use of CBT plus standard medical care with standard medical care alone.18 The standard medical care consisted of supportive treatment, an explanation of NES from a neuropsychiatrist, and supervised withdrawal of antiepileptic drugs. The CBT treatment group was offered weekly hour-long sessions for 12 weeks, accompanied by CBT homework and journaling the frequency and nature of seizure episodes (the CBT techniques are outlined in the Figure18). After 4 months, the CBT treatment group had fewer seizures, and after a 6-month follow-up, they were more likely to be seizure-free. However, in this study, CBT treatment did not improve mood or employment status.
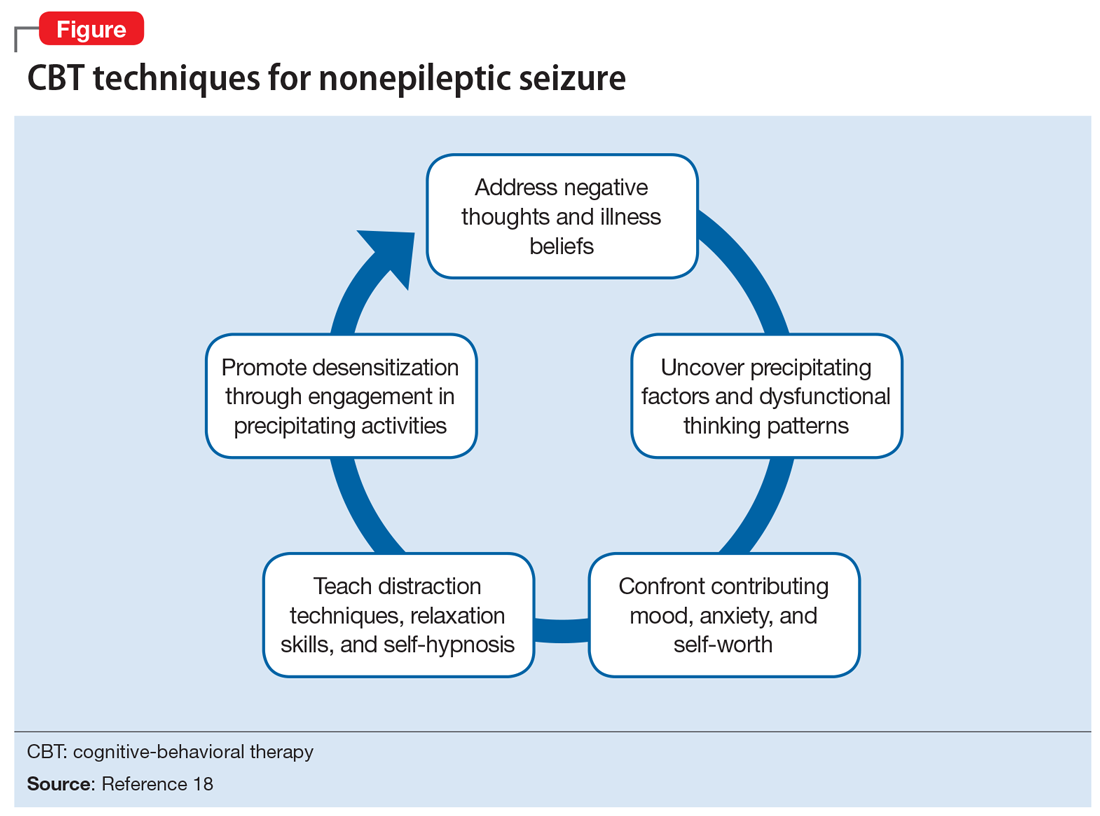
A later investigation looked at using selective serotonin reuptake inhibitors to treat NES in adults.19 This study divided participants into 4 treatment groups: CBT with informed psychotherapy (CBT-ip), CBT-ip plus sertraline, sertraline alone, and treatment as usual. Sertraline was titrated up to a dose of 200 mg/d as tolerated. After 16 weeks of sertraline alone, seizure frequency did not decrease. Although both CBT groups showed a reduction in symptoms of up to 60%, the CBT-ip group reported fewer psychiatric symptoms with better social interactions, quality of life, and global functioning compared with patients treated with CBT-ip plus sertraline. The authors suggested that this may be due to the somatic adverse effects associated with sertraline. This study suggests that CBT without medication is the treatment of choice.
In addition to CBT, studies of psychodynamic psychotherapy for NES have had promising findings.20 Psychodynamic psychotherapy focuses on addressing conscious and unconscious anger, loss, feelings of isolation, and trauma. Through improving emotional processing, insight, coping skills and self-regulation, patients often benefit from an improvement in seizures, psychosocial functioning and health care utilization.
Metin et al21 found that group therapy alongside a family-centered approach elicited a strong and durable reduction in seizures in patients with NES. At enrollment, investigators distributed information on NES to patients and families. Psychoeducation and psychoanalysis with behavior modification techniques were provided in 90-minute weekly group sessions over 3 months. Participants also underwent monthly individualized sessions for standard psychiatric care for 9 months. During the group sessions, operant conditioning techniques were used to prevent secondary gain from seizure-like activity. Families met 4 times for 1 hour each to discuss seizures, receive psychoeducation on a subconscious etiology of NES, and learn behavior modification techniques. All 9 participants who completed group and individual therapy reported a significant and sustained reduction in seizure frequency by at least 50% at 12-month follow-up. Patients also demonstrated improvements in mood, anxiety, and quality of life.
Continue to: A meta-analysis...
A meta-analysis by Carlson and Perry22 that included 13 studies and 228 participants, examined different treatment modalities and their effectiveness for NES. They found that patients who received psychological intervention had a 47% remission rate and 82% improvement in seizure frequency compared with only 14% to 23% of those who did not receive therapy. They postulated that therapy for this illness must be flexible to properly address the socially, psychologically, and functionally heterogenous patient population. Although there are few randomized controlled trials for NES to determine the best evidence-based intervention, there is now consensus that NES has a favorable prognosis when barriers to psychological care are eliminated.
OUTCOME Referral for CBT
The treatment team advises Ms. N to engage in outpatient therapy after discharge from the hospital. Ms. N and her mother agree to the treatment plan, and leave the hospital with a referral for CBT the next day.
Bottom Line
Nonepileptic seizure (NES) is a type of conversion disorder characterized by seizure-like episodes without ictal qualities. Risk factors for NES include concomitant epilepsy, psychiatric disorders, unstable psychosocial situations, and antecedent trauma. Patients with a history of incestuous sexual abuse are most at risk for developing NES. A normal EEG that fully captures a seizure-like episode is diagnostic of NES. Cognitive-behavioral therapy can minimize seizure frequency and intensity.
Related Resources
- Marsh P, Benbadis S, Fernandez F. Psychogenic nonepileptic seizures: ways to win over skeptical patients. Current Psychiatry. 2008;7(1):21-24, 32-35.
- LaFrance WC Jr. Eye-opening behaviors help diagnose nonepileptic seizures. Current Psychiatry. 2006;5(11):121-122, 124, 130.
- LaFrance WC Jr, Kanner AM, Barry JJ. Treating patients with psychological nonepileptic seizures. In: Ettinger AB, Kanner AM, eds. Psychiatric issues in epilepsy: a practical guide to diagnosis and treatment. 2nd ed. Philadelphia, PA: Wolters Kluwer Health/ Lippincott Williams & Wilkins; 2007:461-488.
Drug Brand Name
Sertraline • Zoloft
1. Lesser R. Psychogenic seizures. Neurology. 1996;46(6):1499-1507.
2. Stone J, LaFrance W, Brown R, et al. Conversion disorder: current problems and potential solutions for DSM-5. J Psychosom Res. 2011;71(6):369-376.
3. Diagnostic and statistical manual of mental disorders, 5th ed. Washington, DC: American Psychiatric Association; 2013.
4. Syed T, Arozullah A, Suciu G, et al. Do observer and self-reports of ictal eye closure predict psychogenic nonepileptic seizures? Epilepsia. 2008;49(5):898-904.
5. Vega-Zelaya L, Alvarez M, Ezquiaga E, et al. Psychogenic non-epileptic seizures in a surgical epilepsy unit: experience and a comprehensive review. Epilepsy Topics. 2014. doi: 10.5772/57439.
6. LaFrance W, Baker G, Duncan R, et al. Minimum requirements for the diagnosis of psychogenic nonepileptic seizures: a staged approach. Epilepsia. 2013;54(11):2005-2018.
7. Roeloes K, Pasman J. Stress, childhood trauma, and cognitive functions in functional neurologic disorders. In: Hallett M, Stone J, Carson A, eds. Handbook of clinical neurology: functional neurologic disorders. 3rd ed. New York, NY: Elsevier; 2017:139-155.
8. Paras M, Murad M, Chen L, et al. Sexual abuse and lifetime diagnosis of somatic disorders. JAMA. 2009;302(5):550.
9. Fiszman A, Alves-Leon SV, Nunes RG, et al. Traumatic events and posttraumatic stress disorder in patients with psychogenic nonepileptic seizures: a critical review. Epilepsy Behav. 2004;5(6):818-825.
10. Sharpe D, Faye C. Non-epileptic seizures and child sexual abuse: a critical review of the literature. Clin Psychol Rev. 2006;26(8):1020-1040.
11. Alper K, Devinsky O, Perrine K, et al. Nonepileptic seizures and childhood sexual and physical abuse. Neurology. 1993;43(10):1950-1953.
12. Plioplys S, Doss J, Siddarth P et al. A multisite controlled study of risk factors in pediatric psychogenic nonepileptic seizures. Epilepsia. 2014;55(11):1739-1747.
13. Andersen S, Tomada A, Vincow E, et al. Preliminary evidence for sensitive periods in the effect of childhood sexual abuse on regional brain development. J Neuropsychiatry Clin Neurosci. 2008;20(3):292-301.
14. Sar V. Childhood trauma, dissociation, and psychiatric comorbidity in patients with conversion disorder. Am J Psychiatry. 2004;161(12):2271-2276.
15. Rosenberg HJ, Rosenberg SD, Williamson PD, et al. A comparative study of trauma and posttraumatic stress disorder prevalence in epilepsy patients and psychogenic nonepileptic seizure patients. Epilepsia. 2000;41(4):447-452.
16. Durrant J, Rickards H, Cavanna A. Prognosis and outcome predictors in psychogenic nonepileptic seizures. Epilepsy Res Treat. 2011;2011:1-7.
17. Selkirk M, Duncan R, Oto M, et al. Clinical differences between patients with nonepileptic seizures who report antecedent sexual abuse and those who do not. Epilepsia. 2008;49(8):1446-1450.
18. Goldstein L, Chalder T, Chigwedere C, et al. Cognitive-behavioral therapy for psychogenic nonepileptic seizures: a pilot RCT. Neurology. 2010;74(24):1986-1994.
19. LaFrance W, Baird G, Barry J, et al. Multicenter pilot treatment trial for psychogenic nonepileptic seizures. JAMA Psychiatry. 2014;71(9):997.
20. Howlett S, Reuber M. An augmented model of brief psychodynamic interpersonal therapy for patients with nonepileptic seizures. Psychotherapy (Chic). 2009;46(1):125-138.
21. Metin SZ, Ozmen M, Metin B, et al. Treatment with group psychotherapy for chronic psychogenic nonepileptic seizures. Epilepsy Behav. 2013;28(1):91-94.
22. Carlson P, Perry KN. Psychological interventions for psychogenic non-epileptic seizures: a meta-analysis. Seizure. 2017;45:142-150.
CASE Increasingly frequent paroxysmal episodes
Ms. N, age 12, comes to the hospital for evaluation of paroxysmal episodes of pain, weakness, and muscle spasms. A neurologist who evaluated her as an outpatient had recommended a routine electroencephalogram (EEG); after those results were inconclusive, Ms. N’s mother brought her to the hospital for a 24-hour video EEG.
Ms. N has a history of asthma. She has no history of seizures or headache, but her mother has an unspecified seizure disorder that has been stable with antiepileptic medication for many years. Ms. N has no other family history of autoimmune or neurologic disorders.
Ms. N’s episodes began 6 months ago and have progressively increased in frequency from 5 to 12 episodes a day. She says that before she has an episode, she “ feels tingling in her fingers and mouth, and butterflies in her belly,” and then her “whole body clenches up.” She denies experiencing tongue biting, facial or extremity weakness, incontinence, or loss of consciousness during these episodes.
Shortly before her hospitalization, Ms. N had won a scholarship to attend an overnight art camp. Because her episodes were becoming more frequent and their etiology remained unclear, Ms. N and her mother decided it would be unsafe for her to attend, and that she should go to the hospital for evaluation instead.
EVALUATION Tough questions reveal answers
The pediatric team evaluates Ms. N. Her physical exam, laboratory values, and imaging are all within normal limits. Her neurologic exam demonstrates full strength, tone, and sensation in all extremities. All cranial nerves and reflexes are intact. No dysmorphic features or gait abnormalities are noted. All laboratory and imaging tests are normal, including complete blood cell count, electrolytes, calcium, magnesium, phosphorus, glucose, creatine kinase, liver enzymes, urine drug screen, human chorionic gonadotropin (hCG) urine test, and h
After the initial workup, the pediatric team consults the child and adolescent psychiatry team for a complete assessment of Ms. N due to concerns that a psychological component is contributing to her episodes. According to the psychosocial history obtained from Ms. N and her mother, Ms. N had experienced disrupted attachment, trauma, and loss. At age 5, Ms. N was temporarily removed from her mother’s custody after a fight between her mother and brother. At age 9, Ms. N’s stepfather, her primary father figure, died of a brain tumor.
Ms. N also has significant trauma stemming from her relationship with her biological father. Ms. N’s mother reports that her daughter was conceived during nonconsensual sexual intercourse. Ms. N did not have much contact with her biological father until 6 months ago, when he started picking her up at school and taking her to his home for several hours without permission or supervision. Afterwards, Ms. N confided to her mother and a teacher that her father sexually assaulted her during those visits.
Continue to: Ms. N and her mother...
Ms. N and her mother reported the assault to the police and were awaiting legal action.
During the interview with the psychiatry team, Ms. N denies that any thoughts or actions trigger the episodes and reports that she cannot control when they happen. Because she cannot anticipate the episodes, she says she is afraid to leave her house. She does not know why the episodes are happening and feels frustrated that they are getting worse. Ms. N says, “I have been feeling down lately,” but she denies hopelessness, worthlessness, suicidal ideation, homicidal ideation, delusions, or hallucinations.
In the hospital, when the psychiatry team asks Ms. N about her visits with her father, she says that they are “too painful to talk about,” and fears that discussing them will trigger an episode. However, her mother suggests that her daughter’s sexual trauma, as well as ongoing frustrations with the legal system, are influencing her mood; she has had low energy, poor appetite, and is spending more time in bed. Her mother also reports that Ms. N “avoids going out in the sun and spending time with her friends outside. She doesn’t seem to enjoy shopping and art like she used to.” Ms. N told her mother that she was having nightmares about the trauma and “could not stop thinking about some of the bad stuff that happened during the day.”
Ten minutes into the interview, while being questioned about her father, Ms. N experiences a spastic episode. She curls up in bed on her left side, clenches her entire body, and shuts her eyes. Her mother quickly runs to her bedside and counts the seconds until the end of the episode. After 25 seconds, Ms. N awakes with full recollection of the episode. On review of the video EEG during the episode, no ictal patterns are seen.
[polldaddy:10375873]
The authors’ observations
Paroxysmal episodes of weakness, numbness, and muscle spasms in a young female are suggestive of either epilepsy or nonepileptic seizure (NES).1,2 The negative EEG and physical features are inconsistent with epileptiform seizure, and Ms. N’s history and evaluation are suggestive of NES. Nonepileptic seizures are a type of a conversion disorder, or functional neurologic symptom disorder, in which a patient experiences weakness, abnormal movements, or seizure-like episodes that are inconsistent with organic neurologic disease.3 When a diagnosis of conversion disorder is suspected, a clinician must always consider other pathology that can explain the symptoms, such as migraine, vasovagal syncope, or intracranial mass. If a patient has focal neurologic deficits, head imaging should be pursued. Additionally, the clinician must screen for malingering and factitious disorder before establishing a definitive diagnosis. However, conversion disorder is not a diagnosis of exclusion. For example, a negative EEG does not rule out epilepsy, and patients can have both epilepsy and concomitant NES.
Continue to: Although NES is a common...
Although NES is a common type of conversion disorder, it is often difficult to diagnose, manage, and treat. Patients often receive antiepileptic medications but continue to have worsening events that are refractory to treatment. Various clinical features can suggest NES instead of epilepsy. Forced eye closure on video recording is a specific finding suggestive of NES, yet this feature is not sufficient to make the diagnosis.4 A video EEG must be performed to assess for epilepsy. The diagnosis of NES does not exclude the possibility that a patient has epilepsy, as NES can occur in up to 40% of patients with epilepsy.5 A video EEG without ictal patterns before, during, and after an observed episode is diagnostic of NES.6
[polldaddy:10375874]
The authors’ observations
Conversion disorders such as NES are a presentation of neurologic symptoms that cannot be readily accounted for by other conditions and are often associated with antecedent trauma. Multiple factors in Ms. N’s history increase her risk of NES, including loss of multiple loved ones, ongoing legal involvement, and alleged sexual abuse by her father.
Victims of sexual abuse are more likely than the general population to demonstrate symptoms of conversion disorder, especially NES.7,8 The onset of paroxysmal episodes after incestuous abuse in a teenage girl is characteristic of NES. Compared with patients with complex partial epilepsy (CPE), patients with NES are 3 times more likely to report sexual trauma.9,10 Children who report sexual abuse that precedes NES are more likely to have been victimized by a first-degree relative than patients with CPE who report sexual abuse.11 Risk factors for victims developing NES may be related to the severity of adversity, stress sensitivity, and decreased hippocampal volume.12,13
Ms. N endorsed many psychiatric symptoms that accompany her paroxysmal episodes; this is similar to findings in other patients with NES.14 One study found that depression is 3 times more prevalent and PTSD is 8 times more prevalent in patients with NES.12 During the evaluation, Ms. N’s mother said her daughter had low energy, poor appetite, lethargy, and anhedonia for the preceding 5 months, which is consistent with adjustment disorder.3 Her flashbacks, nightmares, difficulty sleeping, and agoraphobia, along with her trouble engaging with the people and activities that used to bring her joy, are symptoms of PTSD. Nonepileptic seizure is often associated with PTSD and can be viewed as an expression of a dissociated subtype.15
In a literature review, Durrant et al16 isolated prognostic indicators for NES (Table16). This study found that 70% of children and 40% of adults achieve remission from NES. Ms. N’s case has multiple concerning features, such as her comorbid psychiatric conditions, ongoing involvement in a legal case, and sexual trauma; this last factor is associated with the most severe symptoms and worse outcomes.16,17 Despite this somber reality, Ms. N has the support of her mother and is relatively young, which play a vital role in recovery.

Continue to: TREATMENT A strategy for minimizing the episodes
TREATMENT A strategy for minimizing the episodes
Ms. N’s medical workup remains unremarkable throughout the rest of her hospital stay. The psychiatry and pediatric teams discuss their assessments and agree that NES is the most likely diagnosis. The psychiatry team counsels Ms. N and her mother on the diagnosis and etiology of NES.
[polldaddy:10375876]
The authors’ observations
Cognitive-behavioral therapy is currently the treatment of choice for reducing seizure frequency in patients with NES.18,19 The use of CBT was suggested due to the theory that NES represents a dissociative response to trauma. Therapy focuses on changing a patient’s beliefs and perceptions associated with attacks.5 A randomized study of 66 patients with NES compared the use of CBT plus standard medical care with standard medical care alone.18 The standard medical care consisted of supportive treatment, an explanation of NES from a neuropsychiatrist, and supervised withdrawal of antiepileptic drugs. The CBT treatment group was offered weekly hour-long sessions for 12 weeks, accompanied by CBT homework and journaling the frequency and nature of seizure episodes (the CBT techniques are outlined in the Figure18). After 4 months, the CBT treatment group had fewer seizures, and after a 6-month follow-up, they were more likely to be seizure-free. However, in this study, CBT treatment did not improve mood or employment status.

A later investigation looked at using selective serotonin reuptake inhibitors to treat NES in adults.19 This study divided participants into 4 treatment groups: CBT with informed psychotherapy (CBT-ip), CBT-ip plus sertraline, sertraline alone, and treatment as usual. Sertraline was titrated up to a dose of 200 mg/d as tolerated. After 16 weeks of sertraline alone, seizure frequency did not decrease. Although both CBT groups showed a reduction in symptoms of up to 60%, the CBT-ip group reported fewer psychiatric symptoms with better social interactions, quality of life, and global functioning compared with patients treated with CBT-ip plus sertraline. The authors suggested that this may be due to the somatic adverse effects associated with sertraline. This study suggests that CBT without medication is the treatment of choice.
In addition to CBT, studies of psychodynamic psychotherapy for NES have had promising findings.20 Psychodynamic psychotherapy focuses on addressing conscious and unconscious anger, loss, feelings of isolation, and trauma. Through improving emotional processing, insight, coping skills and self-regulation, patients often benefit from an improvement in seizures, psychosocial functioning and health care utilization.
Metin et al21 found that group therapy alongside a family-centered approach elicited a strong and durable reduction in seizures in patients with NES. At enrollment, investigators distributed information on NES to patients and families. Psychoeducation and psychoanalysis with behavior modification techniques were provided in 90-minute weekly group sessions over 3 months. Participants also underwent monthly individualized sessions for standard psychiatric care for 9 months. During the group sessions, operant conditioning techniques were used to prevent secondary gain from seizure-like activity. Families met 4 times for 1 hour each to discuss seizures, receive psychoeducation on a subconscious etiology of NES, and learn behavior modification techniques. All 9 participants who completed group and individual therapy reported a significant and sustained reduction in seizure frequency by at least 50% at 12-month follow-up. Patients also demonstrated improvements in mood, anxiety, and quality of life.
Continue to: A meta-analysis...
A meta-analysis by Carlson and Perry22 that included 13 studies and 228 participants, examined different treatment modalities and their effectiveness for NES. They found that patients who received psychological intervention had a 47% remission rate and 82% improvement in seizure frequency compared with only 14% to 23% of those who did not receive therapy. They postulated that therapy for this illness must be flexible to properly address the socially, psychologically, and functionally heterogenous patient population. Although there are few randomized controlled trials for NES to determine the best evidence-based intervention, there is now consensus that NES has a favorable prognosis when barriers to psychological care are eliminated.
OUTCOME Referral for CBT
The treatment team advises Ms. N to engage in outpatient therapy after discharge from the hospital. Ms. N and her mother agree to the treatment plan, and leave the hospital with a referral for CBT the next day.
Bottom Line
Nonepileptic seizure (NES) is a type of conversion disorder characterized by seizure-like episodes without ictal qualities. Risk factors for NES include concomitant epilepsy, psychiatric disorders, unstable psychosocial situations, and antecedent trauma. Patients with a history of incestuous sexual abuse are most at risk for developing NES. A normal EEG that fully captures a seizure-like episode is diagnostic of NES. Cognitive-behavioral therapy can minimize seizure frequency and intensity.
Related Resources
- Marsh P, Benbadis S, Fernandez F. Psychogenic nonepileptic seizures: ways to win over skeptical patients. Current Psychiatry. 2008;7(1):21-24, 32-35.
- LaFrance WC Jr. Eye-opening behaviors help diagnose nonepileptic seizures. Current Psychiatry. 2006;5(11):121-122, 124, 130.
- LaFrance WC Jr, Kanner AM, Barry JJ. Treating patients with psychological nonepileptic seizures. In: Ettinger AB, Kanner AM, eds. Psychiatric issues in epilepsy: a practical guide to diagnosis and treatment. 2nd ed. Philadelphia, PA: Wolters Kluwer Health/ Lippincott Williams & Wilkins; 2007:461-488.
Drug Brand Name
Sertraline • Zoloft
CASE Increasingly frequent paroxysmal episodes
Ms. N, age 12, comes to the hospital for evaluation of paroxysmal episodes of pain, weakness, and muscle spasms. A neurologist who evaluated her as an outpatient had recommended a routine electroencephalogram (EEG); after those results were inconclusive, Ms. N’s mother brought her to the hospital for a 24-hour video EEG.
Ms. N has a history of asthma. She has no history of seizures or headache, but her mother has an unspecified seizure disorder that has been stable with antiepileptic medication for many years. Ms. N has no other family history of autoimmune or neurologic disorders.
Ms. N’s episodes began 6 months ago and have progressively increased in frequency from 5 to 12 episodes a day. She says that before she has an episode, she “ feels tingling in her fingers and mouth, and butterflies in her belly,” and then her “whole body clenches up.” She denies experiencing tongue biting, facial or extremity weakness, incontinence, or loss of consciousness during these episodes.
Shortly before her hospitalization, Ms. N had won a scholarship to attend an overnight art camp. Because her episodes were becoming more frequent and their etiology remained unclear, Ms. N and her mother decided it would be unsafe for her to attend, and that she should go to the hospital for evaluation instead.
EVALUATION Tough questions reveal answers
The pediatric team evaluates Ms. N. Her physical exam, laboratory values, and imaging are all within normal limits. Her neurologic exam demonstrates full strength, tone, and sensation in all extremities. All cranial nerves and reflexes are intact. No dysmorphic features or gait abnormalities are noted. All laboratory and imaging tests are normal, including complete blood cell count, electrolytes, calcium, magnesium, phosphorus, glucose, creatine kinase, liver enzymes, urine drug screen, human chorionic gonadotropin (hCG) urine test, and h
After the initial workup, the pediatric team consults the child and adolescent psychiatry team for a complete assessment of Ms. N due to concerns that a psychological component is contributing to her episodes. According to the psychosocial history obtained from Ms. N and her mother, Ms. N had experienced disrupted attachment, trauma, and loss. At age 5, Ms. N was temporarily removed from her mother’s custody after a fight between her mother and brother. At age 9, Ms. N’s stepfather, her primary father figure, died of a brain tumor.
Ms. N also has significant trauma stemming from her relationship with her biological father. Ms. N’s mother reports that her daughter was conceived during nonconsensual sexual intercourse. Ms. N did not have much contact with her biological father until 6 months ago, when he started picking her up at school and taking her to his home for several hours without permission or supervision. Afterwards, Ms. N confided to her mother and a teacher that her father sexually assaulted her during those visits.
Continue to: Ms. N and her mother...
Ms. N and her mother reported the assault to the police and were awaiting legal action.
During the interview with the psychiatry team, Ms. N denies that any thoughts or actions trigger the episodes and reports that she cannot control when they happen. Because she cannot anticipate the episodes, she says she is afraid to leave her house. She does not know why the episodes are happening and feels frustrated that they are getting worse. Ms. N says, “I have been feeling down lately,” but she denies hopelessness, worthlessness, suicidal ideation, homicidal ideation, delusions, or hallucinations.
In the hospital, when the psychiatry team asks Ms. N about her visits with her father, she says that they are “too painful to talk about,” and fears that discussing them will trigger an episode. However, her mother suggests that her daughter’s sexual trauma, as well as ongoing frustrations with the legal system, are influencing her mood; she has had low energy, poor appetite, and is spending more time in bed. Her mother also reports that Ms. N “avoids going out in the sun and spending time with her friends outside. She doesn’t seem to enjoy shopping and art like she used to.” Ms. N told her mother that she was having nightmares about the trauma and “could not stop thinking about some of the bad stuff that happened during the day.”
Ten minutes into the interview, while being questioned about her father, Ms. N experiences a spastic episode. She curls up in bed on her left side, clenches her entire body, and shuts her eyes. Her mother quickly runs to her bedside and counts the seconds until the end of the episode. After 25 seconds, Ms. N awakes with full recollection of the episode. On review of the video EEG during the episode, no ictal patterns are seen.
[polldaddy:10375873]
The authors’ observations
Paroxysmal episodes of weakness, numbness, and muscle spasms in a young female are suggestive of either epilepsy or nonepileptic seizure (NES).1,2 The negative EEG and physical features are inconsistent with epileptiform seizure, and Ms. N’s history and evaluation are suggestive of NES. Nonepileptic seizures are a type of a conversion disorder, or functional neurologic symptom disorder, in which a patient experiences weakness, abnormal movements, or seizure-like episodes that are inconsistent with organic neurologic disease.3 When a diagnosis of conversion disorder is suspected, a clinician must always consider other pathology that can explain the symptoms, such as migraine, vasovagal syncope, or intracranial mass. If a patient has focal neurologic deficits, head imaging should be pursued. Additionally, the clinician must screen for malingering and factitious disorder before establishing a definitive diagnosis. However, conversion disorder is not a diagnosis of exclusion. For example, a negative EEG does not rule out epilepsy, and patients can have both epilepsy and concomitant NES.
Continue to: Although NES is a common...
Although NES is a common type of conversion disorder, it is often difficult to diagnose, manage, and treat. Patients often receive antiepileptic medications but continue to have worsening events that are refractory to treatment. Various clinical features can suggest NES instead of epilepsy. Forced eye closure on video recording is a specific finding suggestive of NES, yet this feature is not sufficient to make the diagnosis.4 A video EEG must be performed to assess for epilepsy. The diagnosis of NES does not exclude the possibility that a patient has epilepsy, as NES can occur in up to 40% of patients with epilepsy.5 A video EEG without ictal patterns before, during, and after an observed episode is diagnostic of NES.6
[polldaddy:10375874]
The authors’ observations
Conversion disorders such as NES are a presentation of neurologic symptoms that cannot be readily accounted for by other conditions and are often associated with antecedent trauma. Multiple factors in Ms. N’s history increase her risk of NES, including loss of multiple loved ones, ongoing legal involvement, and alleged sexual abuse by her father.
Victims of sexual abuse are more likely than the general population to demonstrate symptoms of conversion disorder, especially NES.7,8 The onset of paroxysmal episodes after incestuous abuse in a teenage girl is characteristic of NES. Compared with patients with complex partial epilepsy (CPE), patients with NES are 3 times more likely to report sexual trauma.9,10 Children who report sexual abuse that precedes NES are more likely to have been victimized by a first-degree relative than patients with CPE who report sexual abuse.11 Risk factors for victims developing NES may be related to the severity of adversity, stress sensitivity, and decreased hippocampal volume.12,13
Ms. N endorsed many psychiatric symptoms that accompany her paroxysmal episodes; this is similar to findings in other patients with NES.14 One study found that depression is 3 times more prevalent and PTSD is 8 times more prevalent in patients with NES.12 During the evaluation, Ms. N’s mother said her daughter had low energy, poor appetite, lethargy, and anhedonia for the preceding 5 months, which is consistent with adjustment disorder.3 Her flashbacks, nightmares, difficulty sleeping, and agoraphobia, along with her trouble engaging with the people and activities that used to bring her joy, are symptoms of PTSD. Nonepileptic seizure is often associated with PTSD and can be viewed as an expression of a dissociated subtype.15
In a literature review, Durrant et al16 isolated prognostic indicators for NES (Table16). This study found that 70% of children and 40% of adults achieve remission from NES. Ms. N’s case has multiple concerning features, such as her comorbid psychiatric conditions, ongoing involvement in a legal case, and sexual trauma; this last factor is associated with the most severe symptoms and worse outcomes.16,17 Despite this somber reality, Ms. N has the support of her mother and is relatively young, which play a vital role in recovery.

Continue to: TREATMENT A strategy for minimizing the episodes
TREATMENT A strategy for minimizing the episodes
Ms. N’s medical workup remains unremarkable throughout the rest of her hospital stay. The psychiatry and pediatric teams discuss their assessments and agree that NES is the most likely diagnosis. The psychiatry team counsels Ms. N and her mother on the diagnosis and etiology of NES.
[polldaddy:10375876]
The authors’ observations
Cognitive-behavioral therapy is currently the treatment of choice for reducing seizure frequency in patients with NES.18,19 The use of CBT was suggested due to the theory that NES represents a dissociative response to trauma. Therapy focuses on changing a patient’s beliefs and perceptions associated with attacks.5 A randomized study of 66 patients with NES compared the use of CBT plus standard medical care with standard medical care alone.18 The standard medical care consisted of supportive treatment, an explanation of NES from a neuropsychiatrist, and supervised withdrawal of antiepileptic drugs. The CBT treatment group was offered weekly hour-long sessions for 12 weeks, accompanied by CBT homework and journaling the frequency and nature of seizure episodes (the CBT techniques are outlined in the Figure18). After 4 months, the CBT treatment group had fewer seizures, and after a 6-month follow-up, they were more likely to be seizure-free. However, in this study, CBT treatment did not improve mood or employment status.

A later investigation looked at using selective serotonin reuptake inhibitors to treat NES in adults.19 This study divided participants into 4 treatment groups: CBT with informed psychotherapy (CBT-ip), CBT-ip plus sertraline, sertraline alone, and treatment as usual. Sertraline was titrated up to a dose of 200 mg/d as tolerated. After 16 weeks of sertraline alone, seizure frequency did not decrease. Although both CBT groups showed a reduction in symptoms of up to 60%, the CBT-ip group reported fewer psychiatric symptoms with better social interactions, quality of life, and global functioning compared with patients treated with CBT-ip plus sertraline. The authors suggested that this may be due to the somatic adverse effects associated with sertraline. This study suggests that CBT without medication is the treatment of choice.
In addition to CBT, studies of psychodynamic psychotherapy for NES have had promising findings.20 Psychodynamic psychotherapy focuses on addressing conscious and unconscious anger, loss, feelings of isolation, and trauma. Through improving emotional processing, insight, coping skills and self-regulation, patients often benefit from an improvement in seizures, psychosocial functioning and health care utilization.
Metin et al21 found that group therapy alongside a family-centered approach elicited a strong and durable reduction in seizures in patients with NES. At enrollment, investigators distributed information on NES to patients and families. Psychoeducation and psychoanalysis with behavior modification techniques were provided in 90-minute weekly group sessions over 3 months. Participants also underwent monthly individualized sessions for standard psychiatric care for 9 months. During the group sessions, operant conditioning techniques were used to prevent secondary gain from seizure-like activity. Families met 4 times for 1 hour each to discuss seizures, receive psychoeducation on a subconscious etiology of NES, and learn behavior modification techniques. All 9 participants who completed group and individual therapy reported a significant and sustained reduction in seizure frequency by at least 50% at 12-month follow-up. Patients also demonstrated improvements in mood, anxiety, and quality of life.
Continue to: A meta-analysis...
A meta-analysis by Carlson and Perry22 that included 13 studies and 228 participants, examined different treatment modalities and their effectiveness for NES. They found that patients who received psychological intervention had a 47% remission rate and 82% improvement in seizure frequency compared with only 14% to 23% of those who did not receive therapy. They postulated that therapy for this illness must be flexible to properly address the socially, psychologically, and functionally heterogenous patient population. Although there are few randomized controlled trials for NES to determine the best evidence-based intervention, there is now consensus that NES has a favorable prognosis when barriers to psychological care are eliminated.
OUTCOME Referral for CBT
The treatment team advises Ms. N to engage in outpatient therapy after discharge from the hospital. Ms. N and her mother agree to the treatment plan, and leave the hospital with a referral for CBT the next day.
Bottom Line
Nonepileptic seizure (NES) is a type of conversion disorder characterized by seizure-like episodes without ictal qualities. Risk factors for NES include concomitant epilepsy, psychiatric disorders, unstable psychosocial situations, and antecedent trauma. Patients with a history of incestuous sexual abuse are most at risk for developing NES. A normal EEG that fully captures a seizure-like episode is diagnostic of NES. Cognitive-behavioral therapy can minimize seizure frequency and intensity.
Related Resources
- Marsh P, Benbadis S, Fernandez F. Psychogenic nonepileptic seizures: ways to win over skeptical patients. Current Psychiatry. 2008;7(1):21-24, 32-35.
- LaFrance WC Jr. Eye-opening behaviors help diagnose nonepileptic seizures. Current Psychiatry. 2006;5(11):121-122, 124, 130.
- LaFrance WC Jr, Kanner AM, Barry JJ. Treating patients with psychological nonepileptic seizures. In: Ettinger AB, Kanner AM, eds. Psychiatric issues in epilepsy: a practical guide to diagnosis and treatment. 2nd ed. Philadelphia, PA: Wolters Kluwer Health/ Lippincott Williams & Wilkins; 2007:461-488.
Drug Brand Name
Sertraline • Zoloft
1. Lesser R. Psychogenic seizures. Neurology. 1996;46(6):1499-1507.
2. Stone J, LaFrance W, Brown R, et al. Conversion disorder: current problems and potential solutions for DSM-5. J Psychosom Res. 2011;71(6):369-376.
3. Diagnostic and statistical manual of mental disorders, 5th ed. Washington, DC: American Psychiatric Association; 2013.
4. Syed T, Arozullah A, Suciu G, et al. Do observer and self-reports of ictal eye closure predict psychogenic nonepileptic seizures? Epilepsia. 2008;49(5):898-904.
5. Vega-Zelaya L, Alvarez M, Ezquiaga E, et al. Psychogenic non-epileptic seizures in a surgical epilepsy unit: experience and a comprehensive review. Epilepsy Topics. 2014. doi: 10.5772/57439.
6. LaFrance W, Baker G, Duncan R, et al. Minimum requirements for the diagnosis of psychogenic nonepileptic seizures: a staged approach. Epilepsia. 2013;54(11):2005-2018.
7. Roeloes K, Pasman J. Stress, childhood trauma, and cognitive functions in functional neurologic disorders. In: Hallett M, Stone J, Carson A, eds. Handbook of clinical neurology: functional neurologic disorders. 3rd ed. New York, NY: Elsevier; 2017:139-155.
8. Paras M, Murad M, Chen L, et al. Sexual abuse and lifetime diagnosis of somatic disorders. JAMA. 2009;302(5):550.
9. Fiszman A, Alves-Leon SV, Nunes RG, et al. Traumatic events and posttraumatic stress disorder in patients with psychogenic nonepileptic seizures: a critical review. Epilepsy Behav. 2004;5(6):818-825.
10. Sharpe D, Faye C. Non-epileptic seizures and child sexual abuse: a critical review of the literature. Clin Psychol Rev. 2006;26(8):1020-1040.
11. Alper K, Devinsky O, Perrine K, et al. Nonepileptic seizures and childhood sexual and physical abuse. Neurology. 1993;43(10):1950-1953.
12. Plioplys S, Doss J, Siddarth P et al. A multisite controlled study of risk factors in pediatric psychogenic nonepileptic seizures. Epilepsia. 2014;55(11):1739-1747.
13. Andersen S, Tomada A, Vincow E, et al. Preliminary evidence for sensitive periods in the effect of childhood sexual abuse on regional brain development. J Neuropsychiatry Clin Neurosci. 2008;20(3):292-301.
14. Sar V. Childhood trauma, dissociation, and psychiatric comorbidity in patients with conversion disorder. Am J Psychiatry. 2004;161(12):2271-2276.
15. Rosenberg HJ, Rosenberg SD, Williamson PD, et al. A comparative study of trauma and posttraumatic stress disorder prevalence in epilepsy patients and psychogenic nonepileptic seizure patients. Epilepsia. 2000;41(4):447-452.
16. Durrant J, Rickards H, Cavanna A. Prognosis and outcome predictors in psychogenic nonepileptic seizures. Epilepsy Res Treat. 2011;2011:1-7.
17. Selkirk M, Duncan R, Oto M, et al. Clinical differences between patients with nonepileptic seizures who report antecedent sexual abuse and those who do not. Epilepsia. 2008;49(8):1446-1450.
18. Goldstein L, Chalder T, Chigwedere C, et al. Cognitive-behavioral therapy for psychogenic nonepileptic seizures: a pilot RCT. Neurology. 2010;74(24):1986-1994.
19. LaFrance W, Baird G, Barry J, et al. Multicenter pilot treatment trial for psychogenic nonepileptic seizures. JAMA Psychiatry. 2014;71(9):997.
20. Howlett S, Reuber M. An augmented model of brief psychodynamic interpersonal therapy for patients with nonepileptic seizures. Psychotherapy (Chic). 2009;46(1):125-138.
21. Metin SZ, Ozmen M, Metin B, et al. Treatment with group psychotherapy for chronic psychogenic nonepileptic seizures. Epilepsy Behav. 2013;28(1):91-94.
22. Carlson P, Perry KN. Psychological interventions for psychogenic non-epileptic seizures: a meta-analysis. Seizure. 2017;45:142-150.
1. Lesser R. Psychogenic seizures. Neurology. 1996;46(6):1499-1507.
2. Stone J, LaFrance W, Brown R, et al. Conversion disorder: current problems and potential solutions for DSM-5. J Psychosom Res. 2011;71(6):369-376.
3. Diagnostic and statistical manual of mental disorders, 5th ed. Washington, DC: American Psychiatric Association; 2013.
4. Syed T, Arozullah A, Suciu G, et al. Do observer and self-reports of ictal eye closure predict psychogenic nonepileptic seizures? Epilepsia. 2008;49(5):898-904.
5. Vega-Zelaya L, Alvarez M, Ezquiaga E, et al. Psychogenic non-epileptic seizures in a surgical epilepsy unit: experience and a comprehensive review. Epilepsy Topics. 2014. doi: 10.5772/57439.
6. LaFrance W, Baker G, Duncan R, et al. Minimum requirements for the diagnosis of psychogenic nonepileptic seizures: a staged approach. Epilepsia. 2013;54(11):2005-2018.
7. Roeloes K, Pasman J. Stress, childhood trauma, and cognitive functions in functional neurologic disorders. In: Hallett M, Stone J, Carson A, eds. Handbook of clinical neurology: functional neurologic disorders. 3rd ed. New York, NY: Elsevier; 2017:139-155.
8. Paras M, Murad M, Chen L, et al. Sexual abuse and lifetime diagnosis of somatic disorders. JAMA. 2009;302(5):550.
9. Fiszman A, Alves-Leon SV, Nunes RG, et al. Traumatic events and posttraumatic stress disorder in patients with psychogenic nonepileptic seizures: a critical review. Epilepsy Behav. 2004;5(6):818-825.
10. Sharpe D, Faye C. Non-epileptic seizures and child sexual abuse: a critical review of the literature. Clin Psychol Rev. 2006;26(8):1020-1040.
11. Alper K, Devinsky O, Perrine K, et al. Nonepileptic seizures and childhood sexual and physical abuse. Neurology. 1993;43(10):1950-1953.
12. Plioplys S, Doss J, Siddarth P et al. A multisite controlled study of risk factors in pediatric psychogenic nonepileptic seizures. Epilepsia. 2014;55(11):1739-1747.
13. Andersen S, Tomada A, Vincow E, et al. Preliminary evidence for sensitive periods in the effect of childhood sexual abuse on regional brain development. J Neuropsychiatry Clin Neurosci. 2008;20(3):292-301.
14. Sar V. Childhood trauma, dissociation, and psychiatric comorbidity in patients with conversion disorder. Am J Psychiatry. 2004;161(12):2271-2276.
15. Rosenberg HJ, Rosenberg SD, Williamson PD, et al. A comparative study of trauma and posttraumatic stress disorder prevalence in epilepsy patients and psychogenic nonepileptic seizure patients. Epilepsia. 2000;41(4):447-452.
16. Durrant J, Rickards H, Cavanna A. Prognosis and outcome predictors in psychogenic nonepileptic seizures. Epilepsy Res Treat. 2011;2011:1-7.
17. Selkirk M, Duncan R, Oto M, et al. Clinical differences between patients with nonepileptic seizures who report antecedent sexual abuse and those who do not. Epilepsia. 2008;49(8):1446-1450.
18. Goldstein L, Chalder T, Chigwedere C, et al. Cognitive-behavioral therapy for psychogenic nonepileptic seizures: a pilot RCT. Neurology. 2010;74(24):1986-1994.
19. LaFrance W, Baird G, Barry J, et al. Multicenter pilot treatment trial for psychogenic nonepileptic seizures. JAMA Psychiatry. 2014;71(9):997.
20. Howlett S, Reuber M. An augmented model of brief psychodynamic interpersonal therapy for patients with nonepileptic seizures. Psychotherapy (Chic). 2009;46(1):125-138.
21. Metin SZ, Ozmen M, Metin B, et al. Treatment with group psychotherapy for chronic psychogenic nonepileptic seizures. Epilepsy Behav. 2013;28(1):91-94.
22. Carlson P, Perry KN. Psychological interventions for psychogenic non-epileptic seizures: a meta-analysis. Seizure. 2017;45:142-150.
Child trafficking: How to recognize the signs
Child trafficking—a modern-day form of slavery that continues to destroy many lives—often is hidden, even from the clinicians who see its victims. Traffickers typically exploit children for labor or commercial sexual work. The signs and symptoms that suggest a child is being trafficked may be less clear than those of the psychiatric illnesses we usually diagnose and treat. In this article, I summarize characteristics that could be helpful to note when you suspect a child is being trafficked, and offer some resources for helping victims.
How to identify possible victims
Children can be trafficked anywhere. The concept of a child being picked up off a street corner is outdated. Trafficking occurs in cities, suburbs, and rural areas. It happens in hotel rooms, at truck stops, on quiet residential streets, and in expensive homes. The internet has made it easier for traffickers to find victims.
Traffickers typically target youth who are emotionally and physically vulnerable. They often seek out teenagers who are undergoing financial hardships, experiencing family conflict, or have survived natural disasters. Many victims are runaways. In 2016, 1 in 6 child runaways reported to the National Center for Missing and Exploited Children were likely victims of trafficking.1 Of those children, 86% were receiving social services support or living in foster homes.
Traffickers are adept at emotional manipulation, which may explain why a child or adolescent might minimize the abuse during a clinical visit. Traffickers shroud the realities of trafficking with notions of love and inclusion. They use several physical and mental schemes to keep children and adolescents in their grip, such as withholding food, sleep, or medical care. Therefore, we should check for signs and symptoms of chronic medical conditions that have gone untreated, malnutrition, or bruises in various stages of healing.
Connecting risk factors for trafficking to dramatic changes in a young patient’s behavior is challenging. These youth often have dropped out of school, lack consistent family support, and spend their nights in search of a warm place to sleep. Their lives are upended. A child who once was more social may be forced into isolation and make excuses for why she no longer spends time with her friends.
In a study of 106 survivors of domestic sex trafficking, approximately 89% of respondents reported depression during depression. Many respondents reported experiencing anxiety (76.4%), nightmares (73.6%), flashbacks (68%), low self-esteem (81.1%), or feelings of shame or guilt (82.1%).2 Almost 88% of respondents said that they saw a doctor or other clinician while being trafficked, but their clinicians were unable to recognize the signs of trafficking. Part of the challenge is that many children and adolescents are not comfortable discussing their situations with clinicians because they may struggle with shame and guilt. Their traffickers also might have convinced them that they are criminals, not victims. These patients also may have an overwhelming fear of their trafficker, being reported to child welfare authorities, being arrested, being deported, or having their traffickers retaliate against their families. Gaining the trust of a patient who is being trafficked is critical, but not easy, because children may be skeptical of a clinician’s promise of confidentiality.
Some signs of trafficking overlap with the psychiatric presentations with which we are more familiar. These patients may abuse drugs or alcohol as means of escape or because their traffickers force them to use substances.2 They may show symptoms of depression or posttraumatic stress disorder (PTSD) and may be disoriented. Other indicators may be more telling, such as if a child or adolescent describes:
- having no control of their schedules or forms of identification
- having to work excessively long hours, often to pay off an overwhelming debt
- having high security measures installed in their place of residence (such as cameras or barred windows).
Continue to: Also, they may be...
Also, they may be dressed inappropriately for the weather.
We should be concerned when patients’ responses seem coached, if they say they are isolated from their family and community, or if they are submissive or overly timid. In addition, our suspicions should be raised if an accompanying adult guardian insists on sitting in on the appointment or translating for the child. In such instances, we may request that the guardian remain in the waiting area during the appointment so the child will have the opportunity to speak freely.2
How to help a suspected victim
Several local and national organizations help trafficking victims. These organizations provide educational materials and training opportunities for clinicians, as well as direct support for victims. The Homeland Security Blue Campaign advises against confronting a suspected trafficker directly and encourages clinicians to instead report suspected cases to 1-866-347-2423.3
Clinicians can better help children who are trafficked by taking the following 5 steps:
- Learn about the risk factors and signs of child trafficking.
- Post the National Human Trafficking Hotline (1-888-373-7888) in your waiting room.
- Determine if your patient is in danger and needs to be moved to a safe place.
- Connect the patient to social service agencies that can provide financial support and housing assistance so he/she doesn’t feel trapped by financial burdens.
- Work to rebuild their emotional and physical well-being while treating depression, PTSD, substance abuse, or any other mental illness.
1. National Center for Missing and Exploited Childr en. Missing children, state care, and child sex trafficking. http://www.missingkids.com/content/dam/missingkids/pdfs/publications/missingchildrenstatecare.pdf. Accessed June 10, 2019.
2. Lederer LJ, Wetzel CA. The health consequences of sex trafficking and their implications for identifying victims in healthcare facilities. Ann Health Law. 2014;23(1):61-91.
3. Blue Campaign. Identify a victim. US Department of Homeland Security. https://www.dhs.gov/blue-campaign/identify-victim. Accessed June 10, 2019.
Child trafficking—a modern-day form of slavery that continues to destroy many lives—often is hidden, even from the clinicians who see its victims. Traffickers typically exploit children for labor or commercial sexual work. The signs and symptoms that suggest a child is being trafficked may be less clear than those of the psychiatric illnesses we usually diagnose and treat. In this article, I summarize characteristics that could be helpful to note when you suspect a child is being trafficked, and offer some resources for helping victims.
How to identify possible victims
Children can be trafficked anywhere. The concept of a child being picked up off a street corner is outdated. Trafficking occurs in cities, suburbs, and rural areas. It happens in hotel rooms, at truck stops, on quiet residential streets, and in expensive homes. The internet has made it easier for traffickers to find victims.
Traffickers typically target youth who are emotionally and physically vulnerable. They often seek out teenagers who are undergoing financial hardships, experiencing family conflict, or have survived natural disasters. Many victims are runaways. In 2016, 1 in 6 child runaways reported to the National Center for Missing and Exploited Children were likely victims of trafficking.1 Of those children, 86% were receiving social services support or living in foster homes.
Traffickers are adept at emotional manipulation, which may explain why a child or adolescent might minimize the abuse during a clinical visit. Traffickers shroud the realities of trafficking with notions of love and inclusion. They use several physical and mental schemes to keep children and adolescents in their grip, such as withholding food, sleep, or medical care. Therefore, we should check for signs and symptoms of chronic medical conditions that have gone untreated, malnutrition, or bruises in various stages of healing.
Connecting risk factors for trafficking to dramatic changes in a young patient’s behavior is challenging. These youth often have dropped out of school, lack consistent family support, and spend their nights in search of a warm place to sleep. Their lives are upended. A child who once was more social may be forced into isolation and make excuses for why she no longer spends time with her friends.
In a study of 106 survivors of domestic sex trafficking, approximately 89% of respondents reported depression during depression. Many respondents reported experiencing anxiety (76.4%), nightmares (73.6%), flashbacks (68%), low self-esteem (81.1%), or feelings of shame or guilt (82.1%).2 Almost 88% of respondents said that they saw a doctor or other clinician while being trafficked, but their clinicians were unable to recognize the signs of trafficking. Part of the challenge is that many children and adolescents are not comfortable discussing their situations with clinicians because they may struggle with shame and guilt. Their traffickers also might have convinced them that they are criminals, not victims. These patients also may have an overwhelming fear of their trafficker, being reported to child welfare authorities, being arrested, being deported, or having their traffickers retaliate against their families. Gaining the trust of a patient who is being trafficked is critical, but not easy, because children may be skeptical of a clinician’s promise of confidentiality.
Some signs of trafficking overlap with the psychiatric presentations with which we are more familiar. These patients may abuse drugs or alcohol as means of escape or because their traffickers force them to use substances.2 They may show symptoms of depression or posttraumatic stress disorder (PTSD) and may be disoriented. Other indicators may be more telling, such as if a child or adolescent describes:
- having no control of their schedules or forms of identification
- having to work excessively long hours, often to pay off an overwhelming debt
- having high security measures installed in their place of residence (such as cameras or barred windows).
Continue to: Also, they may be...
Also, they may be dressed inappropriately for the weather.
We should be concerned when patients’ responses seem coached, if they say they are isolated from their family and community, or if they are submissive or overly timid. In addition, our suspicions should be raised if an accompanying adult guardian insists on sitting in on the appointment or translating for the child. In such instances, we may request that the guardian remain in the waiting area during the appointment so the child will have the opportunity to speak freely.2
How to help a suspected victim
Several local and national organizations help trafficking victims. These organizations provide educational materials and training opportunities for clinicians, as well as direct support for victims. The Homeland Security Blue Campaign advises against confronting a suspected trafficker directly and encourages clinicians to instead report suspected cases to 1-866-347-2423.3
Clinicians can better help children who are trafficked by taking the following 5 steps:
- Learn about the risk factors and signs of child trafficking.
- Post the National Human Trafficking Hotline (1-888-373-7888) in your waiting room.
- Determine if your patient is in danger and needs to be moved to a safe place.
- Connect the patient to social service agencies that can provide financial support and housing assistance so he/she doesn’t feel trapped by financial burdens.
- Work to rebuild their emotional and physical well-being while treating depression, PTSD, substance abuse, or any other mental illness.
Child trafficking—a modern-day form of slavery that continues to destroy many lives—often is hidden, even from the clinicians who see its victims. Traffickers typically exploit children for labor or commercial sexual work. The signs and symptoms that suggest a child is being trafficked may be less clear than those of the psychiatric illnesses we usually diagnose and treat. In this article, I summarize characteristics that could be helpful to note when you suspect a child is being trafficked, and offer some resources for helping victims.
How to identify possible victims
Children can be trafficked anywhere. The concept of a child being picked up off a street corner is outdated. Trafficking occurs in cities, suburbs, and rural areas. It happens in hotel rooms, at truck stops, on quiet residential streets, and in expensive homes. The internet has made it easier for traffickers to find victims.
Traffickers typically target youth who are emotionally and physically vulnerable. They often seek out teenagers who are undergoing financial hardships, experiencing family conflict, or have survived natural disasters. Many victims are runaways. In 2016, 1 in 6 child runaways reported to the National Center for Missing and Exploited Children were likely victims of trafficking.1 Of those children, 86% were receiving social services support or living in foster homes.
Traffickers are adept at emotional manipulation, which may explain why a child or adolescent might minimize the abuse during a clinical visit. Traffickers shroud the realities of trafficking with notions of love and inclusion. They use several physical and mental schemes to keep children and adolescents in their grip, such as withholding food, sleep, or medical care. Therefore, we should check for signs and symptoms of chronic medical conditions that have gone untreated, malnutrition, or bruises in various stages of healing.
Connecting risk factors for trafficking to dramatic changes in a young patient’s behavior is challenging. These youth often have dropped out of school, lack consistent family support, and spend their nights in search of a warm place to sleep. Their lives are upended. A child who once was more social may be forced into isolation and make excuses for why she no longer spends time with her friends.
In a study of 106 survivors of domestic sex trafficking, approximately 89% of respondents reported depression during depression. Many respondents reported experiencing anxiety (76.4%), nightmares (73.6%), flashbacks (68%), low self-esteem (81.1%), or feelings of shame or guilt (82.1%).2 Almost 88% of respondents said that they saw a doctor or other clinician while being trafficked, but their clinicians were unable to recognize the signs of trafficking. Part of the challenge is that many children and adolescents are not comfortable discussing their situations with clinicians because they may struggle with shame and guilt. Their traffickers also might have convinced them that they are criminals, not victims. These patients also may have an overwhelming fear of their trafficker, being reported to child welfare authorities, being arrested, being deported, or having their traffickers retaliate against their families. Gaining the trust of a patient who is being trafficked is critical, but not easy, because children may be skeptical of a clinician’s promise of confidentiality.
Some signs of trafficking overlap with the psychiatric presentations with which we are more familiar. These patients may abuse drugs or alcohol as means of escape or because their traffickers force them to use substances.2 They may show symptoms of depression or posttraumatic stress disorder (PTSD) and may be disoriented. Other indicators may be more telling, such as if a child or adolescent describes:
- having no control of their schedules or forms of identification
- having to work excessively long hours, often to pay off an overwhelming debt
- having high security measures installed in their place of residence (such as cameras or barred windows).
Continue to: Also, they may be...
Also, they may be dressed inappropriately for the weather.
We should be concerned when patients’ responses seem coached, if they say they are isolated from their family and community, or if they are submissive or overly timid. In addition, our suspicions should be raised if an accompanying adult guardian insists on sitting in on the appointment or translating for the child. In such instances, we may request that the guardian remain in the waiting area during the appointment so the child will have the opportunity to speak freely.2
How to help a suspected victim
Several local and national organizations help trafficking victims. These organizations provide educational materials and training opportunities for clinicians, as well as direct support for victims. The Homeland Security Blue Campaign advises against confronting a suspected trafficker directly and encourages clinicians to instead report suspected cases to 1-866-347-2423.3
Clinicians can better help children who are trafficked by taking the following 5 steps:
- Learn about the risk factors and signs of child trafficking.
- Post the National Human Trafficking Hotline (1-888-373-7888) in your waiting room.
- Determine if your patient is in danger and needs to be moved to a safe place.
- Connect the patient to social service agencies that can provide financial support and housing assistance so he/she doesn’t feel trapped by financial burdens.
- Work to rebuild their emotional and physical well-being while treating depression, PTSD, substance abuse, or any other mental illness.
1. National Center for Missing and Exploited Childr en. Missing children, state care, and child sex trafficking. http://www.missingkids.com/content/dam/missingkids/pdfs/publications/missingchildrenstatecare.pdf. Accessed June 10, 2019.
2. Lederer LJ, Wetzel CA. The health consequences of sex trafficking and their implications for identifying victims in healthcare facilities. Ann Health Law. 2014;23(1):61-91.
3. Blue Campaign. Identify a victim. US Department of Homeland Security. https://www.dhs.gov/blue-campaign/identify-victim. Accessed June 10, 2019.
1. National Center for Missing and Exploited Childr en. Missing children, state care, and child sex trafficking. http://www.missingkids.com/content/dam/missingkids/pdfs/publications/missingchildrenstatecare.pdf. Accessed June 10, 2019.
2. Lederer LJ, Wetzel CA. The health consequences of sex trafficking and their implications for identifying victims in healthcare facilities. Ann Health Law. 2014;23(1):61-91.
3. Blue Campaign. Identify a victim. US Department of Homeland Security. https://www.dhs.gov/blue-campaign/identify-victim. Accessed June 10, 2019.
Teens & tobacco use: USPSTF issues draft recs on prevention, cessation
Reference
1. US Preventive Services Task Force. Draft Evidence Review for Prevention and Cessation of Tobacco Use in Children and Adolescents: Primary Care Interventions. https://www.uspreventiveservicestaskforce.org/Page/Document/draft-evidence-review/tobacco-and-nicotine-use-prevention-in-children-and-adolescents-primary-care-interventions. Accessed July 8, 2019.
Reference
1. US Preventive Services Task Force. Draft Evidence Review for Prevention and Cessation of Tobacco Use in Children and Adolescents: Primary Care Interventions. https://www.uspreventiveservicestaskforce.org/Page/Document/draft-evidence-review/tobacco-and-nicotine-use-prevention-in-children-and-adolescents-primary-care-interventions. Accessed July 8, 2019.
Reference
1. US Preventive Services Task Force. Draft Evidence Review for Prevention and Cessation of Tobacco Use in Children and Adolescents: Primary Care Interventions. https://www.uspreventiveservicestaskforce.org/Page/Document/draft-evidence-review/tobacco-and-nicotine-use-prevention-in-children-and-adolescents-primary-care-interventions. Accessed July 8, 2019.











