User login
Click for Credit: PPI use & dementia; Weight loss after gastroplasty; more
Here are 5 articles from the December issue of Clinician Reviews (individual articles are valid for one year from date of publication—expiration dates below):
1. Sustainable weight loss seen 5 years after endoscopic sleeve gastroplasty
To take the posttest, go to: https://bit.ly/37lteRX
Expires May 16, 2020
2. PT beats steroid injections for knee OA
To take the posttest, go to: https://bit.ly/2KIWKY6
Expires May 17, 2020
3. Better screening needed to reduce pregnancy-related overdose, death
To take the posttest, go to: https://bit.ly/2XEZyuG
Expires May 17, 2020
4. Meta-analysis finds no link between PPI use and risk of dementia
To take the posttest, go to: https://bit.ly/2Xzs7JM
Expires June 3, 2020
5. Study: Cardiac biomarkers predicted CV events in CAP
To take the posttest, go to: https://bit.ly/33bAH2u
Expires August 13, 2020
Here are 5 articles from the December issue of Clinician Reviews (individual articles are valid for one year from date of publication—expiration dates below):
1. Sustainable weight loss seen 5 years after endoscopic sleeve gastroplasty
To take the posttest, go to: https://bit.ly/37lteRX
Expires May 16, 2020
2. PT beats steroid injections for knee OA
To take the posttest, go to: https://bit.ly/2KIWKY6
Expires May 17, 2020
3. Better screening needed to reduce pregnancy-related overdose, death
To take the posttest, go to: https://bit.ly/2XEZyuG
Expires May 17, 2020
4. Meta-analysis finds no link between PPI use and risk of dementia
To take the posttest, go to: https://bit.ly/2Xzs7JM
Expires June 3, 2020
5. Study: Cardiac biomarkers predicted CV events in CAP
To take the posttest, go to: https://bit.ly/33bAH2u
Expires August 13, 2020
Here are 5 articles from the December issue of Clinician Reviews (individual articles are valid for one year from date of publication—expiration dates below):
1. Sustainable weight loss seen 5 years after endoscopic sleeve gastroplasty
To take the posttest, go to: https://bit.ly/37lteRX
Expires May 16, 2020
2. PT beats steroid injections for knee OA
To take the posttest, go to: https://bit.ly/2KIWKY6
Expires May 17, 2020
3. Better screening needed to reduce pregnancy-related overdose, death
To take the posttest, go to: https://bit.ly/2XEZyuG
Expires May 17, 2020
4. Meta-analysis finds no link between PPI use and risk of dementia
To take the posttest, go to: https://bit.ly/2Xzs7JM
Expires June 3, 2020
5. Study: Cardiac biomarkers predicted CV events in CAP
To take the posttest, go to: https://bit.ly/33bAH2u
Expires August 13, 2020
Supplemental MRI found to benefit women with dense breast tissue
The use of supplemental MRI screening in women with extremely dense breast tissue and normal results on mammography led to the diagnosis of significantly fewer interval cancers, compared with mammography alone during a 2-year screening period, results from a randomized trial show.
“Women with extremely dense breast tissue have an increased risk of breast cancer, and their cancers are also less likely to be detected on mammography,” Dutch researchers led by Marije F. Bakker, PhD, of Utrecht (The Netherlands) University and colleagues wrote for the Dense Tissue and Early Breast Neoplasm Screening (DENSE) Trial Study Group in an article published in the New England Journal of Medicine.
“Such patients may benefit from a tailored breast-screening strategy, supplemented with more sensitive imaging methods. The benefit of supplemental imaging is the subject of a worldwide debate. In the United States, a federal law directs breast-density reporting, but supplemental screening is not recommended in American guidelines. Although supplemental imaging increases the rate of cancer detection in women with dense breasts, the question remains whether it improves health outcomes,” they said.
In the DENSE trial, researchers assigned 40,373 women with extremely dense breast tissue and negative results on screening mammography to a group that was invited to undergo supplemental MRI or to a group that received mammography screening only. The women were between the ages of 50 and 75 years and were enrolled between December 2011 and November 2015 as part of the Dutch population-based digital mammography screening program. The primary outcome was the between-group difference in the incidence of interval cancers during a 2-year screening period.
Dr. Bakker and associates found that the interval cancer rate was 2.5 per 1,000 screenings among 4,783 women in the MRI invitation group, compared with 5 per 1,000 among the 32,312 women in the mammography-only group, a difference of 2.5 per 1,000 screenings (P less than 0.001). Among the women who were invited to undergo MRI, 59% actually underwent the procedure. Of the 20 interval cancers diagnosed in the MRI-invitation group, 4 were diagnosed in the women who had undergone MRI, which translated to 0.8 per 1,000 screenings. The remaining 16 were diagnosed in those who had not undergone MRI, which translated into 4.9 per 1,000 screenings.
“Undergoing supplemental MRI was associated with a cancer-detection rate of 16.5 per 1,000 screenings and resulted in a false positive rate of 8.0% (79.8 per 1,000 screenings),” the researchers wrote. “Of the women who underwent a breast biopsy on the basis of an MRI indication, 26.3% had breast cancer and 73.7% did not.”
Dr. Bakker and coauthors acknowledged certain limitations of the trial, including the fact that it was not large enough to examine the effect of MRI screening on breast cancer–specific or overall mortality. “This outcome would require a much larger sample size and longer follow-up,” they wrote. “The lower rate of interval cancers that we found among participants who underwent MRI is indicative of and prerequisite for an effect on mortality. After that, a reduction in the number of advanced cancers would also be required to show a mortality benefit, which would require several years of follow-up.”
In an accompanying editorial, Dan L. Longo, MD, noted that the study provides high-quality data from a randomized trial where none existed (N Engl J Med. 2019 Nov 27. doi: 10.1056/NEJMe1912943). “It appears to show that among women with dense breasts, the risk of interval cancers is halved by following a negative mammogram with MRI screening,” wrote Dr. Longo, who is deputy editor of the New England Journal of Medicine, as well as professor of medicine at Harvard Medical School, Boston. “But is a reduction in interval cancers an appropriate surrogate for improved overall survival? It appears that most of the cancers that were detected on supplemental MRI screening were found at an early stage. Ductal carcinoma in situ was 10 times more frequent among patients undergoing MRI, and these diagnoses were likely to lead to treatments. What remains unclear is whether the tumors would never otherwise have been detected or threatened the patient’s survival.”
The trial was supported by the University Medical Center Utrecht (the Netherlands), the Netherlands Organization for Health Research and Development, the Dutch Cancer Society, the Dutch Pink Ribbon–A Sister’s Hope organization, Stichting Kankerpreventie Midden-West, and Bayer Pharmaceuticals, with an in-kind contribution from Volpara Health Technologies.
The researchers reported having no relevant financial disclosures other than the trial funding. Dr. Longo is employed by the New England Journal of Medicine as deputy editor.
SOURCE: Bakker MF et al. N Engl J Med. 2019 Nov 27. doi: 10.1056/NEJMoa1903986.
The use of supplemental MRI screening in women with extremely dense breast tissue and normal results on mammography led to the diagnosis of significantly fewer interval cancers, compared with mammography alone during a 2-year screening period, results from a randomized trial show.
“Women with extremely dense breast tissue have an increased risk of breast cancer, and their cancers are also less likely to be detected on mammography,” Dutch researchers led by Marije F. Bakker, PhD, of Utrecht (The Netherlands) University and colleagues wrote for the Dense Tissue and Early Breast Neoplasm Screening (DENSE) Trial Study Group in an article published in the New England Journal of Medicine.
“Such patients may benefit from a tailored breast-screening strategy, supplemented with more sensitive imaging methods. The benefit of supplemental imaging is the subject of a worldwide debate. In the United States, a federal law directs breast-density reporting, but supplemental screening is not recommended in American guidelines. Although supplemental imaging increases the rate of cancer detection in women with dense breasts, the question remains whether it improves health outcomes,” they said.
In the DENSE trial, researchers assigned 40,373 women with extremely dense breast tissue and negative results on screening mammography to a group that was invited to undergo supplemental MRI or to a group that received mammography screening only. The women were between the ages of 50 and 75 years and were enrolled between December 2011 and November 2015 as part of the Dutch population-based digital mammography screening program. The primary outcome was the between-group difference in the incidence of interval cancers during a 2-year screening period.
Dr. Bakker and associates found that the interval cancer rate was 2.5 per 1,000 screenings among 4,783 women in the MRI invitation group, compared with 5 per 1,000 among the 32,312 women in the mammography-only group, a difference of 2.5 per 1,000 screenings (P less than 0.001). Among the women who were invited to undergo MRI, 59% actually underwent the procedure. Of the 20 interval cancers diagnosed in the MRI-invitation group, 4 were diagnosed in the women who had undergone MRI, which translated to 0.8 per 1,000 screenings. The remaining 16 were diagnosed in those who had not undergone MRI, which translated into 4.9 per 1,000 screenings.
“Undergoing supplemental MRI was associated with a cancer-detection rate of 16.5 per 1,000 screenings and resulted in a false positive rate of 8.0% (79.8 per 1,000 screenings),” the researchers wrote. “Of the women who underwent a breast biopsy on the basis of an MRI indication, 26.3% had breast cancer and 73.7% did not.”
Dr. Bakker and coauthors acknowledged certain limitations of the trial, including the fact that it was not large enough to examine the effect of MRI screening on breast cancer–specific or overall mortality. “This outcome would require a much larger sample size and longer follow-up,” they wrote. “The lower rate of interval cancers that we found among participants who underwent MRI is indicative of and prerequisite for an effect on mortality. After that, a reduction in the number of advanced cancers would also be required to show a mortality benefit, which would require several years of follow-up.”
In an accompanying editorial, Dan L. Longo, MD, noted that the study provides high-quality data from a randomized trial where none existed (N Engl J Med. 2019 Nov 27. doi: 10.1056/NEJMe1912943). “It appears to show that among women with dense breasts, the risk of interval cancers is halved by following a negative mammogram with MRI screening,” wrote Dr. Longo, who is deputy editor of the New England Journal of Medicine, as well as professor of medicine at Harvard Medical School, Boston. “But is a reduction in interval cancers an appropriate surrogate for improved overall survival? It appears that most of the cancers that were detected on supplemental MRI screening were found at an early stage. Ductal carcinoma in situ was 10 times more frequent among patients undergoing MRI, and these diagnoses were likely to lead to treatments. What remains unclear is whether the tumors would never otherwise have been detected or threatened the patient’s survival.”
The trial was supported by the University Medical Center Utrecht (the Netherlands), the Netherlands Organization for Health Research and Development, the Dutch Cancer Society, the Dutch Pink Ribbon–A Sister’s Hope organization, Stichting Kankerpreventie Midden-West, and Bayer Pharmaceuticals, with an in-kind contribution from Volpara Health Technologies.
The researchers reported having no relevant financial disclosures other than the trial funding. Dr. Longo is employed by the New England Journal of Medicine as deputy editor.
SOURCE: Bakker MF et al. N Engl J Med. 2019 Nov 27. doi: 10.1056/NEJMoa1903986.
The use of supplemental MRI screening in women with extremely dense breast tissue and normal results on mammography led to the diagnosis of significantly fewer interval cancers, compared with mammography alone during a 2-year screening period, results from a randomized trial show.
“Women with extremely dense breast tissue have an increased risk of breast cancer, and their cancers are also less likely to be detected on mammography,” Dutch researchers led by Marije F. Bakker, PhD, of Utrecht (The Netherlands) University and colleagues wrote for the Dense Tissue and Early Breast Neoplasm Screening (DENSE) Trial Study Group in an article published in the New England Journal of Medicine.
“Such patients may benefit from a tailored breast-screening strategy, supplemented with more sensitive imaging methods. The benefit of supplemental imaging is the subject of a worldwide debate. In the United States, a federal law directs breast-density reporting, but supplemental screening is not recommended in American guidelines. Although supplemental imaging increases the rate of cancer detection in women with dense breasts, the question remains whether it improves health outcomes,” they said.
In the DENSE trial, researchers assigned 40,373 women with extremely dense breast tissue and negative results on screening mammography to a group that was invited to undergo supplemental MRI or to a group that received mammography screening only. The women were between the ages of 50 and 75 years and were enrolled between December 2011 and November 2015 as part of the Dutch population-based digital mammography screening program. The primary outcome was the between-group difference in the incidence of interval cancers during a 2-year screening period.
Dr. Bakker and associates found that the interval cancer rate was 2.5 per 1,000 screenings among 4,783 women in the MRI invitation group, compared with 5 per 1,000 among the 32,312 women in the mammography-only group, a difference of 2.5 per 1,000 screenings (P less than 0.001). Among the women who were invited to undergo MRI, 59% actually underwent the procedure. Of the 20 interval cancers diagnosed in the MRI-invitation group, 4 were diagnosed in the women who had undergone MRI, which translated to 0.8 per 1,000 screenings. The remaining 16 were diagnosed in those who had not undergone MRI, which translated into 4.9 per 1,000 screenings.
“Undergoing supplemental MRI was associated with a cancer-detection rate of 16.5 per 1,000 screenings and resulted in a false positive rate of 8.0% (79.8 per 1,000 screenings),” the researchers wrote. “Of the women who underwent a breast biopsy on the basis of an MRI indication, 26.3% had breast cancer and 73.7% did not.”
Dr. Bakker and coauthors acknowledged certain limitations of the trial, including the fact that it was not large enough to examine the effect of MRI screening on breast cancer–specific or overall mortality. “This outcome would require a much larger sample size and longer follow-up,” they wrote. “The lower rate of interval cancers that we found among participants who underwent MRI is indicative of and prerequisite for an effect on mortality. After that, a reduction in the number of advanced cancers would also be required to show a mortality benefit, which would require several years of follow-up.”
In an accompanying editorial, Dan L. Longo, MD, noted that the study provides high-quality data from a randomized trial where none existed (N Engl J Med. 2019 Nov 27. doi: 10.1056/NEJMe1912943). “It appears to show that among women with dense breasts, the risk of interval cancers is halved by following a negative mammogram with MRI screening,” wrote Dr. Longo, who is deputy editor of the New England Journal of Medicine, as well as professor of medicine at Harvard Medical School, Boston. “But is a reduction in interval cancers an appropriate surrogate for improved overall survival? It appears that most of the cancers that were detected on supplemental MRI screening were found at an early stage. Ductal carcinoma in situ was 10 times more frequent among patients undergoing MRI, and these diagnoses were likely to lead to treatments. What remains unclear is whether the tumors would never otherwise have been detected or threatened the patient’s survival.”
The trial was supported by the University Medical Center Utrecht (the Netherlands), the Netherlands Organization for Health Research and Development, the Dutch Cancer Society, the Dutch Pink Ribbon–A Sister’s Hope organization, Stichting Kankerpreventie Midden-West, and Bayer Pharmaceuticals, with an in-kind contribution from Volpara Health Technologies.
The researchers reported having no relevant financial disclosures other than the trial funding. Dr. Longo is employed by the New England Journal of Medicine as deputy editor.
SOURCE: Bakker MF et al. N Engl J Med. 2019 Nov 27. doi: 10.1056/NEJMoa1903986.
FROM THE NEW ENGLAND JOURNAL OF MEDICINE
Key clinical point:
Major finding: The interval cancer rate was 2.5 per 1,000 screenings among women in the MRI invitation group, compared with 5 per 1,000 among women in the mammography-only group, a difference of 2.5 per 1,000 screenings (P less than 0.001).
Study details: A multicenter, randomized study of 40,373 women between the ages of 50 and 75 years. One-quarter were offered supplemental MRI to the mammography all received.
Disclosures: The trial was supported by the University Medical Center Utrecht, the Netherlands Organization for Health Research and Development, the Dutch Cancer Society, the Dutch Pink Ribbon–A Sister’s Hope organization, Stichting Kankerpreventie Midden-West, and Bayer Pharmaceuticals, with an in-kind contribution from Volpara Health Technologies.
Source: Bakker MF et al. N Engl J Med. 2019 Nov 27. doi: 10.1056/NEJMoa1903986.
ART treatment at birth found to benefit neonates with HIV
Initiating antiretroviral therapy within an hour after birth, rather than waiting a few weeks, lowers the reservoir of HIV virus and improves immune response, early results from an ongoing study in Botswana, Africa, showed.
Despite advances in treatment programs during pregnancy that prevent mother to child HIV transmission, 300-500 pediatric HIV infections occur each day in sub-Saharan Africa, Roger Shapiro, MD, MPH, said during a media teleconference organized by the American Association for the Advancement of Science. “Most pediatric HIV diagnosis programs currently test children at 4-6 weeks of age to identify infections that occur either in pregnancy or during delivery,” said Dr. Shapiro, associate professor of immunology and infectious diseases at the Harvard T.H. Chan School of Public Health, Boston. “However, these programs miss the opportunity to begin immediate antiretroviral treatment for children who can be identified earlier. There are benefits to starting treatment and arresting HIV replication in the first week of life. These include limiting the viral reservoir or the population of infected cells, limiting potentially harmful immune responses to the virus, and preventing the rapid decline in health that can occur in the early weeks of HIV infection in infants. Without treatment, 50% of HIV-infected children regress to death by 2 years. Starting treatment in the first weeks or months of life has been shown to improve survival.”
With these benefits in mind, he and his associates initiated the Early Infant Treatment (EIT) study in 2015 to diagnose and treat HIV infected infants in Botswana in the first week of life or as early as possible after infection. They screened more than 10,000 children and identified 40 that were HIV infected. “This low transmission rate is a testament to the fact that most HIV-positive women in Botswana receive three-drug treatment in pregnancy, which is highly successful in blocking transmission,” Dr. Shapiro said. “When we identified an HIV-infected infant, we consented mothers to allow us to start treatment right away. We used a series of regimens because there are limited options. The available options include older drugs, some of which are no longer used for adults but which were the only options for children.”
The researchers initiated three initial drugs approved for newborns: nevirapine, zidovudine, and lamivudine, and then changed the regimen slightly after a few weeks, when they used ritonavir-boosted lopinavir, plus the lamivudine and zidovudine. “We followed the children weekly at first, then at monthly refill visits, and kept close track of how they were taking the medicines and the level of virus in each child’s blood,” Dr. Shapiro said.
In a manuscript published online in Science Translational Medicine on Nov. 27, 2019, he and his associates reported results of the first 10 children enrolled in the EIT study who reached about 96 weeks on treatment. For comparison, they also enrolled a group of children as controls, who started treatment later in the first year of life, after being identified at a more standard time of 4-6 weeks. Tests performed included droplet digital polymerase chain reaction, HIV near-full-genome sequencing, whole-genome amplification, and flow cytometry.
“What we wanted to focus on are the HIV reservoir cells that are persisting in the setting of antiretroviral treatment,” study coauthor Mathias Lichterfeld, MD, PhD, explained during the teleconference. “Those are the cells that would cause viral rebound if treatment were to be interrupted. We used complex technology to look at these cells, using next-generation sequencing, which allows us to identify those cells that harbor HIV that has the ability to initiate new viral replication.”
He and his colleagues observed that the number of reservoir cells was significantly smaller than in adults who were on ART for a median of 16 years. It also was smaller than in infected infants who started ART treatment weeks after birth.
In addition, immune activation was reduced in the cohort of infants who were treated immediately after birth.
“We are seeing a distinct advantage of early treatment initiation,” said Dr. Lichterfeld of the infectious disease division at Brigham and Women’s Hospital, Boston. “By doing these assays we see both virological benefits in terms of a very-low reservoir size, and we see immune system characteristics that are also associated with better abilities for antimicrobial immune defense and a lower level of immune activation.”
Another study coauthor, Daniel R. Kuritzkes, MD, chief of the infectious disease division at Brigham and Women’s Hospital, said the findings show “how critically important” it is to extend studies of HIV cure or long-term remission to infants and children. “Very-early intervention in neonates limits the size of the reservoir and offers us the best opportunity for future interventions aimed at cure and long-term drug-free remission of HIV infection,” he said. “We don’t think the current intervention is itself curative, but it sets the stage for the capacity to offer additional innovative interventions in the future. Beyond the importance of this work for cure research per se, this very early intervention in neonates also has the potential of conferring important clinical benefits to the children who participated in this study. Finally, our study demonstrates the feasibility and importance of doing this type of research in neonates in resource-limited settings, given the appropriate infrastructure.”
EIT is supported by the National Institutes of Health. Dr. Lichterfeld disclosed having received speaking and consulting honoraria from Merck and Gilead. Dr. Kuritzkes disclosed having received consulting honoraria and/or research support from Gilead, Merck, and ViiV.
SOURCE: Garcia-Broncano P et al. Sci Transl Med. 2019 Nov 27. eaax7350.
Initiating antiretroviral therapy within an hour after birth, rather than waiting a few weeks, lowers the reservoir of HIV virus and improves immune response, early results from an ongoing study in Botswana, Africa, showed.
Despite advances in treatment programs during pregnancy that prevent mother to child HIV transmission, 300-500 pediatric HIV infections occur each day in sub-Saharan Africa, Roger Shapiro, MD, MPH, said during a media teleconference organized by the American Association for the Advancement of Science. “Most pediatric HIV diagnosis programs currently test children at 4-6 weeks of age to identify infections that occur either in pregnancy or during delivery,” said Dr. Shapiro, associate professor of immunology and infectious diseases at the Harvard T.H. Chan School of Public Health, Boston. “However, these programs miss the opportunity to begin immediate antiretroviral treatment for children who can be identified earlier. There are benefits to starting treatment and arresting HIV replication in the first week of life. These include limiting the viral reservoir or the population of infected cells, limiting potentially harmful immune responses to the virus, and preventing the rapid decline in health that can occur in the early weeks of HIV infection in infants. Without treatment, 50% of HIV-infected children regress to death by 2 years. Starting treatment in the first weeks or months of life has been shown to improve survival.”
With these benefits in mind, he and his associates initiated the Early Infant Treatment (EIT) study in 2015 to diagnose and treat HIV infected infants in Botswana in the first week of life or as early as possible after infection. They screened more than 10,000 children and identified 40 that were HIV infected. “This low transmission rate is a testament to the fact that most HIV-positive women in Botswana receive three-drug treatment in pregnancy, which is highly successful in blocking transmission,” Dr. Shapiro said. “When we identified an HIV-infected infant, we consented mothers to allow us to start treatment right away. We used a series of regimens because there are limited options. The available options include older drugs, some of which are no longer used for adults but which were the only options for children.”
The researchers initiated three initial drugs approved for newborns: nevirapine, zidovudine, and lamivudine, and then changed the regimen slightly after a few weeks, when they used ritonavir-boosted lopinavir, plus the lamivudine and zidovudine. “We followed the children weekly at first, then at monthly refill visits, and kept close track of how they were taking the medicines and the level of virus in each child’s blood,” Dr. Shapiro said.
In a manuscript published online in Science Translational Medicine on Nov. 27, 2019, he and his associates reported results of the first 10 children enrolled in the EIT study who reached about 96 weeks on treatment. For comparison, they also enrolled a group of children as controls, who started treatment later in the first year of life, after being identified at a more standard time of 4-6 weeks. Tests performed included droplet digital polymerase chain reaction, HIV near-full-genome sequencing, whole-genome amplification, and flow cytometry.
“What we wanted to focus on are the HIV reservoir cells that are persisting in the setting of antiretroviral treatment,” study coauthor Mathias Lichterfeld, MD, PhD, explained during the teleconference. “Those are the cells that would cause viral rebound if treatment were to be interrupted. We used complex technology to look at these cells, using next-generation sequencing, which allows us to identify those cells that harbor HIV that has the ability to initiate new viral replication.”
He and his colleagues observed that the number of reservoir cells was significantly smaller than in adults who were on ART for a median of 16 years. It also was smaller than in infected infants who started ART treatment weeks after birth.
In addition, immune activation was reduced in the cohort of infants who were treated immediately after birth.
“We are seeing a distinct advantage of early treatment initiation,” said Dr. Lichterfeld of the infectious disease division at Brigham and Women’s Hospital, Boston. “By doing these assays we see both virological benefits in terms of a very-low reservoir size, and we see immune system characteristics that are also associated with better abilities for antimicrobial immune defense and a lower level of immune activation.”
Another study coauthor, Daniel R. Kuritzkes, MD, chief of the infectious disease division at Brigham and Women’s Hospital, said the findings show “how critically important” it is to extend studies of HIV cure or long-term remission to infants and children. “Very-early intervention in neonates limits the size of the reservoir and offers us the best opportunity for future interventions aimed at cure and long-term drug-free remission of HIV infection,” he said. “We don’t think the current intervention is itself curative, but it sets the stage for the capacity to offer additional innovative interventions in the future. Beyond the importance of this work for cure research per se, this very early intervention in neonates also has the potential of conferring important clinical benefits to the children who participated in this study. Finally, our study demonstrates the feasibility and importance of doing this type of research in neonates in resource-limited settings, given the appropriate infrastructure.”
EIT is supported by the National Institutes of Health. Dr. Lichterfeld disclosed having received speaking and consulting honoraria from Merck and Gilead. Dr. Kuritzkes disclosed having received consulting honoraria and/or research support from Gilead, Merck, and ViiV.
SOURCE: Garcia-Broncano P et al. Sci Transl Med. 2019 Nov 27. eaax7350.
Initiating antiretroviral therapy within an hour after birth, rather than waiting a few weeks, lowers the reservoir of HIV virus and improves immune response, early results from an ongoing study in Botswana, Africa, showed.
Despite advances in treatment programs during pregnancy that prevent mother to child HIV transmission, 300-500 pediatric HIV infections occur each day in sub-Saharan Africa, Roger Shapiro, MD, MPH, said during a media teleconference organized by the American Association for the Advancement of Science. “Most pediatric HIV diagnosis programs currently test children at 4-6 weeks of age to identify infections that occur either in pregnancy or during delivery,” said Dr. Shapiro, associate professor of immunology and infectious diseases at the Harvard T.H. Chan School of Public Health, Boston. “However, these programs miss the opportunity to begin immediate antiretroviral treatment for children who can be identified earlier. There are benefits to starting treatment and arresting HIV replication in the first week of life. These include limiting the viral reservoir or the population of infected cells, limiting potentially harmful immune responses to the virus, and preventing the rapid decline in health that can occur in the early weeks of HIV infection in infants. Without treatment, 50% of HIV-infected children regress to death by 2 years. Starting treatment in the first weeks or months of life has been shown to improve survival.”
With these benefits in mind, he and his associates initiated the Early Infant Treatment (EIT) study in 2015 to diagnose and treat HIV infected infants in Botswana in the first week of life or as early as possible after infection. They screened more than 10,000 children and identified 40 that were HIV infected. “This low transmission rate is a testament to the fact that most HIV-positive women in Botswana receive three-drug treatment in pregnancy, which is highly successful in blocking transmission,” Dr. Shapiro said. “When we identified an HIV-infected infant, we consented mothers to allow us to start treatment right away. We used a series of regimens because there are limited options. The available options include older drugs, some of which are no longer used for adults but which were the only options for children.”
The researchers initiated three initial drugs approved for newborns: nevirapine, zidovudine, and lamivudine, and then changed the regimen slightly after a few weeks, when they used ritonavir-boosted lopinavir, plus the lamivudine and zidovudine. “We followed the children weekly at first, then at monthly refill visits, and kept close track of how they were taking the medicines and the level of virus in each child’s blood,” Dr. Shapiro said.
In a manuscript published online in Science Translational Medicine on Nov. 27, 2019, he and his associates reported results of the first 10 children enrolled in the EIT study who reached about 96 weeks on treatment. For comparison, they also enrolled a group of children as controls, who started treatment later in the first year of life, after being identified at a more standard time of 4-6 weeks. Tests performed included droplet digital polymerase chain reaction, HIV near-full-genome sequencing, whole-genome amplification, and flow cytometry.
“What we wanted to focus on are the HIV reservoir cells that are persisting in the setting of antiretroviral treatment,” study coauthor Mathias Lichterfeld, MD, PhD, explained during the teleconference. “Those are the cells that would cause viral rebound if treatment were to be interrupted. We used complex technology to look at these cells, using next-generation sequencing, which allows us to identify those cells that harbor HIV that has the ability to initiate new viral replication.”
He and his colleagues observed that the number of reservoir cells was significantly smaller than in adults who were on ART for a median of 16 years. It also was smaller than in infected infants who started ART treatment weeks after birth.
In addition, immune activation was reduced in the cohort of infants who were treated immediately after birth.
“We are seeing a distinct advantage of early treatment initiation,” said Dr. Lichterfeld of the infectious disease division at Brigham and Women’s Hospital, Boston. “By doing these assays we see both virological benefits in terms of a very-low reservoir size, and we see immune system characteristics that are also associated with better abilities for antimicrobial immune defense and a lower level of immune activation.”
Another study coauthor, Daniel R. Kuritzkes, MD, chief of the infectious disease division at Brigham and Women’s Hospital, said the findings show “how critically important” it is to extend studies of HIV cure or long-term remission to infants and children. “Very-early intervention in neonates limits the size of the reservoir and offers us the best opportunity for future interventions aimed at cure and long-term drug-free remission of HIV infection,” he said. “We don’t think the current intervention is itself curative, but it sets the stage for the capacity to offer additional innovative interventions in the future. Beyond the importance of this work for cure research per se, this very early intervention in neonates also has the potential of conferring important clinical benefits to the children who participated in this study. Finally, our study demonstrates the feasibility and importance of doing this type of research in neonates in resource-limited settings, given the appropriate infrastructure.”
EIT is supported by the National Institutes of Health. Dr. Lichterfeld disclosed having received speaking and consulting honoraria from Merck and Gilead. Dr. Kuritzkes disclosed having received consulting honoraria and/or research support from Gilead, Merck, and ViiV.
SOURCE: Garcia-Broncano P et al. Sci Transl Med. 2019 Nov 27. eaax7350.
FROM SCIENCE TRANSLATIONAL MEDICINE
Key clinical point: Antiretroviral treatment initiation immediately after birth reduced HIV-1 viral reservoir size and alters innate immune responses in neonates.
Major finding: Very-early ART intervention in neonates infected with HIV limited the number of virally infected cells and improves immune response.
Study details: A cohort study of 10 infants infected with HIV who were born in Botswana, Africa.
Disclosures: The Early Infant Treatment study is supported by the National Institutes of Health. Dr. Lichterfeld disclosed having received speaking and consulting honoraria from Merck and Gilead. Dr. Kuritzkes disclosed having received consulting honoraria and/or research support from Gilead, Merck, and ViiV.
Source: Garcia-Broncano P et al. Sci Transl Med. 2019 Nov 27. eaax7350.
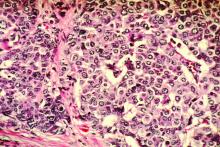
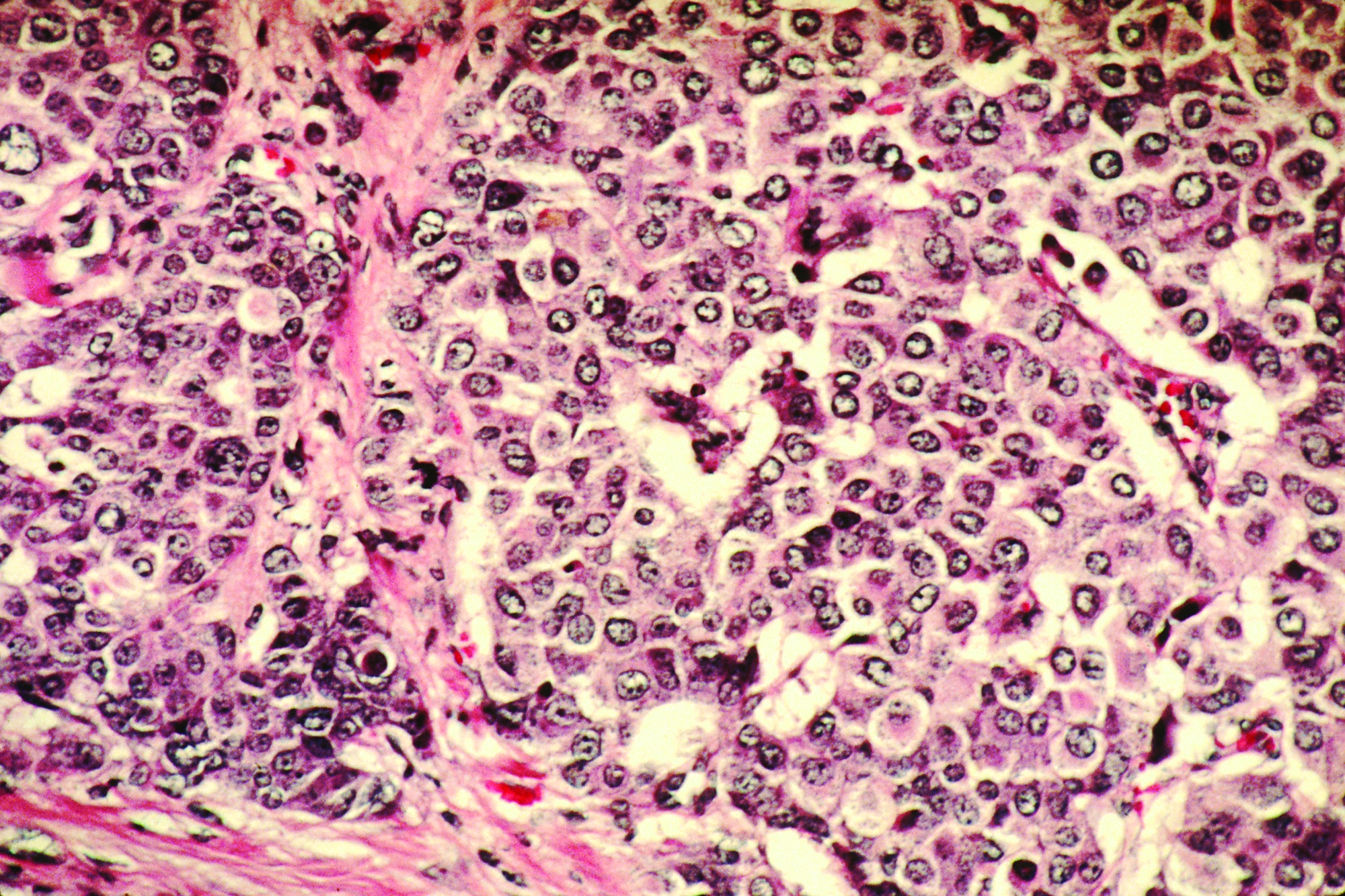
Could your patient benefit from a breast CA risk-reducing med?
References
1. Final update summary: breast cancer: medication use to reduce risk. U.S. Preventive Services Task Force Web Site. https://www.uspreventiveservicestaskforce.org/Page/Document/UpdateSummaryFinal/breast-cancer-medications-for-risk-reduction1 Updated October 2019. Accessed November 25, 2019.
2. Final update summary: BRCA-related cancer: risk assessment, genetic counseling, and genetic testing. U.S. Preventive Services Task Force Web Site. https://www.uspreventiveservicestaskforce.org/Page/Document/UpdateSummaryFinal/brca-related-cancer-risk-assessment-genetic-counseling-and-genetic-testing1. Updated August 2019. Accessed November 25, 2019.
3. Campos-Outcalt D. USPSTF BRCA testing recs: 2 more groups require attention. J Fam Pract. 2019;68:audio. https://www.mdedge.com/familymedicine/article/208085/womens-health/uspstf-brca-testing-recs-2-more-groups-require-attention?channel=60894. Accessed November 25, 2019.
References
1. Final update summary: breast cancer: medication use to reduce risk. U.S. Preventive Services Task Force Web Site. https://www.uspreventiveservicestaskforce.org/Page/Document/UpdateSummaryFinal/breast-cancer-medications-for-risk-reduction1 Updated October 2019. Accessed November 25, 2019.
2. Final update summary: BRCA-related cancer: risk assessment, genetic counseling, and genetic testing. U.S. Preventive Services Task Force Web Site. https://www.uspreventiveservicestaskforce.org/Page/Document/UpdateSummaryFinal/brca-related-cancer-risk-assessment-genetic-counseling-and-genetic-testing1. Updated August 2019. Accessed November 25, 2019.
3. Campos-Outcalt D. USPSTF BRCA testing recs: 2 more groups require attention. J Fam Pract. 2019;68:audio. https://www.mdedge.com/familymedicine/article/208085/womens-health/uspstf-brca-testing-recs-2-more-groups-require-attention?channel=60894. Accessed November 25, 2019.
References
1. Final update summary: breast cancer: medication use to reduce risk. U.S. Preventive Services Task Force Web Site. https://www.uspreventiveservicestaskforce.org/Page/Document/UpdateSummaryFinal/breast-cancer-medications-for-risk-reduction1 Updated October 2019. Accessed November 25, 2019.
2. Final update summary: BRCA-related cancer: risk assessment, genetic counseling, and genetic testing. U.S. Preventive Services Task Force Web Site. https://www.uspreventiveservicestaskforce.org/Page/Document/UpdateSummaryFinal/brca-related-cancer-risk-assessment-genetic-counseling-and-genetic-testing1. Updated August 2019. Accessed November 25, 2019.
3. Campos-Outcalt D. USPSTF BRCA testing recs: 2 more groups require attention. J Fam Pract. 2019;68:audio. https://www.mdedge.com/familymedicine/article/208085/womens-health/uspstf-brca-testing-recs-2-more-groups-require-attention?channel=60894. Accessed November 25, 2019.
Metformin after GDM: Lessons from landmark diabetes prevention trial
WASHINGTON – Metformin’s role in preventing or delaying the onset of type 2 diabetes in women with a history of gestational diabetes mellitus has been firmly established by the Diabetes Prevention Program (DPP) trial – most recently, by 15-year follow-up data reported this year – and the drug should be front and center for clinicians who hope to stave off the “remarkable” incidence of type 2 diabetes after GDM, Robert E. Ratner, MD, maintained at the biennial meeting of the Diabetes in Pregnancy Study Group of North America.
The DPP included “the single largest population of women with a history of GDM that’s been looked at in a randomized controlled trial,” and considering its multiethnic population, the trial offers a reliable representative sample to ponder today when evaluating long-term use of metformin after GDM, said Dr. Ratner, a principal investigator of the National Institutes of Health–sponsored DPP and the DPP Outcomes Study and a former chief scientific & medical officer for the American Diabetes Association.
The drug stacked up equally to lifestyle interventions among DPP participants who had a history of GDM, but it’s important to appreciate that these interventions were intensive and that metformin is inexpensive, well tolerated, and “has a long safety record,” he said.
Results of follow-up out to 15 years
Of the more than 3,000 men and women enrolled in the landmark DPP, conducted during 1996-2001, 350 were women with a documented history of GDM and over 1,400 were women who had deliveries but no history of GDM. All participants had impaired glucose tolerance – defined for the trial as having both a fasting plasma glucose value of 95-125 mg/dL and a 2-hour value of 140-199 mg/dL after a 75-g glucose load – and were randomized to placebo, metformin, or intensive lifestyle intervention.
Metformin therapy reduced the incidence of diabetes by approximately 50% in women with a history of GDM, compared with the placebo group – as did lifestyle – over 3 years. The number needed to treat to prevent one case of diabetes was five. Women without a history of GDM, on the other hand, saw only a 14% reduction with metformin when compared with placebo (and a 49% reduction with lifestyle).
“In women with a history of GDM ... one pill twice a day for $4 a month worked as well as intensive lifestyle [change],” Dr. Ratner said, referring to the initial GDM-specific analysis of DPP data published in 2008 (J Clin Endocrinol Metab. 2008;93[12]:4774-9).
In a 10-year postrandomization follow-up, published in 2015, both metformin and lifestyle continued to be equally effective for the GDM group, reducing the progression to diabetes by 40% and 35%, respectively (J Clin Endocrinol Metab. 2015;100:1646-53). The number needed to treat to prevent one case of diabetes was seven. (Among women without a history of GDM, metformin did not reduce progression to diabetes.)
A recent DPP Outcomes Study analysis of metformin’s impact on diabetes prevention at 15 years, moreover, showed a 41% risk reduction among women with a history of GDM (Diabetes Care. 2019;42[4]:601-8).
Advice on prescribing metformin prophylactically
Asked after his presentation whether women with a history of GDM and either an elevated fasting plasma glucose value or an elevated 2-hour oral glucose tolerance test (GTT) value – or neither of the two – would benefit from taking metformin, Dr. Ratner said that “we’re stuck with inclusion criteria of the DPP, in which they had to meet both criteria ... What I’d say, though, is that not everyone with a history of GDM needs to be on metformin prophylactically. But [for women who have] prediabetes as defined by the ADA, the cost-benefit analysis points toward metformin.”
And with respect to early initiation and long-term use of the drug, “I would have absolutely no qualms about medicating a 25-year-old who had developed GDM and who in the postpartum period has prediabetes,” Dr. Ratner said during an open discussion. “She’s actually at the highest risk for developing type 2 very early.”
Kim Boggess, MD, who also presented on long-term use of metformin after GDM, said in the discussion period that she is often quick to recommend metformin therapy to her patients who have an elevated fasting plasma glucose value in the postpartum period, even when a 75-g oral GTT has not yet been performed. (The ADA and the American College of Obstetricians and Gynecologists recommend completion of an oral GTT at 4-12 weeks postpartum after GDM.)
“I start them [on metformin] especially if they’ve had a cesarean section. Even 2, 3, 4 weeks of profound hyperglycemia could have potentially deleterious effects,” said Dr. Boggess, professor and maternal-fetal medicine program director at the University of North Carolina, Chapel Hill. “If someone comes in [shortly after] and looks like they have pristine control, then it might be worth stopping the metformin for 3-5 days (and retesting).”
Dr. Ratner said that, in this clinical scenario, he would first ensure that the fasting glucose value “is a true fasting glucose” and “if it’s substantially elevated – I’m talking 100, 105, 110 mg/dL – I’d start metformin, and I’m not even sure I’d do the GTT.” But, he advised, “if you’re going to do the GTT, I’d stop the metformin the day before.”
In her presentation, Dr. Boggess pointed out that metformin wasn’t shown to be superior to lifestyle interventions in the DPP for preventing progression to type 2 DM, and that some women are more motivated for intensive lifestyle change than others. The ADA recommends, in fact, that either metformin or lifestyle interventions be prescribed to women with a history of GDM who are found to have prediabetes.
There are no data to support the use of metformin either during or after pregnancy to improve weight loss or reduce weight retention following pregnancy, but at least several studies have shown that lifestyle interventions are effective, she noted.
What is needed, Dr. Boggess said, are more data on the effects of metformin on cardiovascular disease risk, as well as larger studies of metformin in the postpartum period “to help us determine the best dose.” Some research on metformin use in the postpartum period has reported gastrointestinal side effects and dissatisfaction, she noted.
Dr. Ratner said that metformin’s main drawback is the need for occasional testing of B12 levels. Regarding weight loss and what was observed in the DPP, he said, women with a history of GDM who were randomized to intensive lifestyle interventions did not lose as much weight as women without a history of GDM.
Women who entered the DPP with a GDM history, he noted in his presentation, were essentially a “cohort of survivors.” They had an average age of 43 (compared with 52 years in the parous women without GDM) and a mean interval from the index GDM pregnancy of 11 years, which means that women with the highest risk of diabetes conversion were excluded, Dr. Ratner said.
Age was the only significantly different baseline characteristic between parous women with and without GDM, he noted. Women with a history of GDM who were randomized to placebo had a 71% higher incidence of diabetes than women without such a history – a striking natural history, Dr. Ratner said.
He and Dr. Boggess each reported that they have no financial or other interests that pose a conflict of interest.
WASHINGTON – Metformin’s role in preventing or delaying the onset of type 2 diabetes in women with a history of gestational diabetes mellitus has been firmly established by the Diabetes Prevention Program (DPP) trial – most recently, by 15-year follow-up data reported this year – and the drug should be front and center for clinicians who hope to stave off the “remarkable” incidence of type 2 diabetes after GDM, Robert E. Ratner, MD, maintained at the biennial meeting of the Diabetes in Pregnancy Study Group of North America.
The DPP included “the single largest population of women with a history of GDM that’s been looked at in a randomized controlled trial,” and considering its multiethnic population, the trial offers a reliable representative sample to ponder today when evaluating long-term use of metformin after GDM, said Dr. Ratner, a principal investigator of the National Institutes of Health–sponsored DPP and the DPP Outcomes Study and a former chief scientific & medical officer for the American Diabetes Association.
The drug stacked up equally to lifestyle interventions among DPP participants who had a history of GDM, but it’s important to appreciate that these interventions were intensive and that metformin is inexpensive, well tolerated, and “has a long safety record,” he said.
Results of follow-up out to 15 years
Of the more than 3,000 men and women enrolled in the landmark DPP, conducted during 1996-2001, 350 were women with a documented history of GDM and over 1,400 were women who had deliveries but no history of GDM. All participants had impaired glucose tolerance – defined for the trial as having both a fasting plasma glucose value of 95-125 mg/dL and a 2-hour value of 140-199 mg/dL after a 75-g glucose load – and were randomized to placebo, metformin, or intensive lifestyle intervention.
Metformin therapy reduced the incidence of diabetes by approximately 50% in women with a history of GDM, compared with the placebo group – as did lifestyle – over 3 years. The number needed to treat to prevent one case of diabetes was five. Women without a history of GDM, on the other hand, saw only a 14% reduction with metformin when compared with placebo (and a 49% reduction with lifestyle).
“In women with a history of GDM ... one pill twice a day for $4 a month worked as well as intensive lifestyle [change],” Dr. Ratner said, referring to the initial GDM-specific analysis of DPP data published in 2008 (J Clin Endocrinol Metab. 2008;93[12]:4774-9).
In a 10-year postrandomization follow-up, published in 2015, both metformin and lifestyle continued to be equally effective for the GDM group, reducing the progression to diabetes by 40% and 35%, respectively (J Clin Endocrinol Metab. 2015;100:1646-53). The number needed to treat to prevent one case of diabetes was seven. (Among women without a history of GDM, metformin did not reduce progression to diabetes.)
A recent DPP Outcomes Study analysis of metformin’s impact on diabetes prevention at 15 years, moreover, showed a 41% risk reduction among women with a history of GDM (Diabetes Care. 2019;42[4]:601-8).
Advice on prescribing metformin prophylactically
Asked after his presentation whether women with a history of GDM and either an elevated fasting plasma glucose value or an elevated 2-hour oral glucose tolerance test (GTT) value – or neither of the two – would benefit from taking metformin, Dr. Ratner said that “we’re stuck with inclusion criteria of the DPP, in which they had to meet both criteria ... What I’d say, though, is that not everyone with a history of GDM needs to be on metformin prophylactically. But [for women who have] prediabetes as defined by the ADA, the cost-benefit analysis points toward metformin.”
And with respect to early initiation and long-term use of the drug, “I would have absolutely no qualms about medicating a 25-year-old who had developed GDM and who in the postpartum period has prediabetes,” Dr. Ratner said during an open discussion. “She’s actually at the highest risk for developing type 2 very early.”
Kim Boggess, MD, who also presented on long-term use of metformin after GDM, said in the discussion period that she is often quick to recommend metformin therapy to her patients who have an elevated fasting plasma glucose value in the postpartum period, even when a 75-g oral GTT has not yet been performed. (The ADA and the American College of Obstetricians and Gynecologists recommend completion of an oral GTT at 4-12 weeks postpartum after GDM.)
“I start them [on metformin] especially if they’ve had a cesarean section. Even 2, 3, 4 weeks of profound hyperglycemia could have potentially deleterious effects,” said Dr. Boggess, professor and maternal-fetal medicine program director at the University of North Carolina, Chapel Hill. “If someone comes in [shortly after] and looks like they have pristine control, then it might be worth stopping the metformin for 3-5 days (and retesting).”
Dr. Ratner said that, in this clinical scenario, he would first ensure that the fasting glucose value “is a true fasting glucose” and “if it’s substantially elevated – I’m talking 100, 105, 110 mg/dL – I’d start metformin, and I’m not even sure I’d do the GTT.” But, he advised, “if you’re going to do the GTT, I’d stop the metformin the day before.”
In her presentation, Dr. Boggess pointed out that metformin wasn’t shown to be superior to lifestyle interventions in the DPP for preventing progression to type 2 DM, and that some women are more motivated for intensive lifestyle change than others. The ADA recommends, in fact, that either metformin or lifestyle interventions be prescribed to women with a history of GDM who are found to have prediabetes.
There are no data to support the use of metformin either during or after pregnancy to improve weight loss or reduce weight retention following pregnancy, but at least several studies have shown that lifestyle interventions are effective, she noted.
What is needed, Dr. Boggess said, are more data on the effects of metformin on cardiovascular disease risk, as well as larger studies of metformin in the postpartum period “to help us determine the best dose.” Some research on metformin use in the postpartum period has reported gastrointestinal side effects and dissatisfaction, she noted.
Dr. Ratner said that metformin’s main drawback is the need for occasional testing of B12 levels. Regarding weight loss and what was observed in the DPP, he said, women with a history of GDM who were randomized to intensive lifestyle interventions did not lose as much weight as women without a history of GDM.
Women who entered the DPP with a GDM history, he noted in his presentation, were essentially a “cohort of survivors.” They had an average age of 43 (compared with 52 years in the parous women without GDM) and a mean interval from the index GDM pregnancy of 11 years, which means that women with the highest risk of diabetes conversion were excluded, Dr. Ratner said.
Age was the only significantly different baseline characteristic between parous women with and without GDM, he noted. Women with a history of GDM who were randomized to placebo had a 71% higher incidence of diabetes than women without such a history – a striking natural history, Dr. Ratner said.
He and Dr. Boggess each reported that they have no financial or other interests that pose a conflict of interest.
WASHINGTON – Metformin’s role in preventing or delaying the onset of type 2 diabetes in women with a history of gestational diabetes mellitus has been firmly established by the Diabetes Prevention Program (DPP) trial – most recently, by 15-year follow-up data reported this year – and the drug should be front and center for clinicians who hope to stave off the “remarkable” incidence of type 2 diabetes after GDM, Robert E. Ratner, MD, maintained at the biennial meeting of the Diabetes in Pregnancy Study Group of North America.
The DPP included “the single largest population of women with a history of GDM that’s been looked at in a randomized controlled trial,” and considering its multiethnic population, the trial offers a reliable representative sample to ponder today when evaluating long-term use of metformin after GDM, said Dr. Ratner, a principal investigator of the National Institutes of Health–sponsored DPP and the DPP Outcomes Study and a former chief scientific & medical officer for the American Diabetes Association.
The drug stacked up equally to lifestyle interventions among DPP participants who had a history of GDM, but it’s important to appreciate that these interventions were intensive and that metformin is inexpensive, well tolerated, and “has a long safety record,” he said.
Results of follow-up out to 15 years
Of the more than 3,000 men and women enrolled in the landmark DPP, conducted during 1996-2001, 350 were women with a documented history of GDM and over 1,400 were women who had deliveries but no history of GDM. All participants had impaired glucose tolerance – defined for the trial as having both a fasting plasma glucose value of 95-125 mg/dL and a 2-hour value of 140-199 mg/dL after a 75-g glucose load – and were randomized to placebo, metformin, or intensive lifestyle intervention.
Metformin therapy reduced the incidence of diabetes by approximately 50% in women with a history of GDM, compared with the placebo group – as did lifestyle – over 3 years. The number needed to treat to prevent one case of diabetes was five. Women without a history of GDM, on the other hand, saw only a 14% reduction with metformin when compared with placebo (and a 49% reduction with lifestyle).
“In women with a history of GDM ... one pill twice a day for $4 a month worked as well as intensive lifestyle [change],” Dr. Ratner said, referring to the initial GDM-specific analysis of DPP data published in 2008 (J Clin Endocrinol Metab. 2008;93[12]:4774-9).
In a 10-year postrandomization follow-up, published in 2015, both metformin and lifestyle continued to be equally effective for the GDM group, reducing the progression to diabetes by 40% and 35%, respectively (J Clin Endocrinol Metab. 2015;100:1646-53). The number needed to treat to prevent one case of diabetes was seven. (Among women without a history of GDM, metformin did not reduce progression to diabetes.)
A recent DPP Outcomes Study analysis of metformin’s impact on diabetes prevention at 15 years, moreover, showed a 41% risk reduction among women with a history of GDM (Diabetes Care. 2019;42[4]:601-8).
Advice on prescribing metformin prophylactically
Asked after his presentation whether women with a history of GDM and either an elevated fasting plasma glucose value or an elevated 2-hour oral glucose tolerance test (GTT) value – or neither of the two – would benefit from taking metformin, Dr. Ratner said that “we’re stuck with inclusion criteria of the DPP, in which they had to meet both criteria ... What I’d say, though, is that not everyone with a history of GDM needs to be on metformin prophylactically. But [for women who have] prediabetes as defined by the ADA, the cost-benefit analysis points toward metformin.”
And with respect to early initiation and long-term use of the drug, “I would have absolutely no qualms about medicating a 25-year-old who had developed GDM and who in the postpartum period has prediabetes,” Dr. Ratner said during an open discussion. “She’s actually at the highest risk for developing type 2 very early.”
Kim Boggess, MD, who also presented on long-term use of metformin after GDM, said in the discussion period that she is often quick to recommend metformin therapy to her patients who have an elevated fasting plasma glucose value in the postpartum period, even when a 75-g oral GTT has not yet been performed. (The ADA and the American College of Obstetricians and Gynecologists recommend completion of an oral GTT at 4-12 weeks postpartum after GDM.)
“I start them [on metformin] especially if they’ve had a cesarean section. Even 2, 3, 4 weeks of profound hyperglycemia could have potentially deleterious effects,” said Dr. Boggess, professor and maternal-fetal medicine program director at the University of North Carolina, Chapel Hill. “If someone comes in [shortly after] and looks like they have pristine control, then it might be worth stopping the metformin for 3-5 days (and retesting).”
Dr. Ratner said that, in this clinical scenario, he would first ensure that the fasting glucose value “is a true fasting glucose” and “if it’s substantially elevated – I’m talking 100, 105, 110 mg/dL – I’d start metformin, and I’m not even sure I’d do the GTT.” But, he advised, “if you’re going to do the GTT, I’d stop the metformin the day before.”
In her presentation, Dr. Boggess pointed out that metformin wasn’t shown to be superior to lifestyle interventions in the DPP for preventing progression to type 2 DM, and that some women are more motivated for intensive lifestyle change than others. The ADA recommends, in fact, that either metformin or lifestyle interventions be prescribed to women with a history of GDM who are found to have prediabetes.
There are no data to support the use of metformin either during or after pregnancy to improve weight loss or reduce weight retention following pregnancy, but at least several studies have shown that lifestyle interventions are effective, she noted.
What is needed, Dr. Boggess said, are more data on the effects of metformin on cardiovascular disease risk, as well as larger studies of metformin in the postpartum period “to help us determine the best dose.” Some research on metformin use in the postpartum period has reported gastrointestinal side effects and dissatisfaction, she noted.
Dr. Ratner said that metformin’s main drawback is the need for occasional testing of B12 levels. Regarding weight loss and what was observed in the DPP, he said, women with a history of GDM who were randomized to intensive lifestyle interventions did not lose as much weight as women without a history of GDM.
Women who entered the DPP with a GDM history, he noted in his presentation, were essentially a “cohort of survivors.” They had an average age of 43 (compared with 52 years in the parous women without GDM) and a mean interval from the index GDM pregnancy of 11 years, which means that women with the highest risk of diabetes conversion were excluded, Dr. Ratner said.
Age was the only significantly different baseline characteristic between parous women with and without GDM, he noted. Women with a history of GDM who were randomized to placebo had a 71% higher incidence of diabetes than women without such a history – a striking natural history, Dr. Ratner said.
He and Dr. Boggess each reported that they have no financial or other interests that pose a conflict of interest.
REPORTING FROM THE DPSG-NA 2019
Learning about and prescribing emergency contraception
As health care providers to children, we always are learning. And with new knowledge we sometimes can be taken out of our comfort zone. One of those areas are teenagers, contraception, safe-sex counseling, and now emergency contraception (EC). In residency you have your 1-month adolescent medicine rotation to try and absorb every bit of information like a sponge, but there also will be a level of discomfort and uncertainty. However, as medical providers we cannot let the above prevent us from giving well-rounded and informed care.
When our teens disclose the most private moment of their life, we have to be armed and ready to not only comfort them, but advise and guide them to making a decision so that they can ensure their safety. The answers regarding sexual activity are becoming more and more alarming, especially in our younger patients. Therefore, this is an important discussion to have at every visit (not just well-child checks), so that education opportunities are not missed and our patients feel a sense of normalcy about discussing reproductive health with their health care provider and or parents.
We all have our personal beliefs, but we cannot let that guide our decision on what care or education we give our patients. Unfortunately, I have heard many health care providers judge our patients for their promiscuity, when we need to educate them – not be their judge and jury. Our teens go through different stages of growth and development, and with these stages come experimentation and risk taking. So as their health care providers, we need to be up to date on the information out there.
With regards with EC, some of our patients think that they can get it only after having unprotected sex. However, they should know that the oral ECs can be given to them at any time, so should they be in the situation above, they have an immediate remedy. With the different options come different counseling and different instructions on administration and follow-up. In residency, we might not have learned the skill of inserting an IUD, which is another form of EC; that is why there are many resources available. These resources include hands-on workshops, videos on counseling, and your friendly neighborhood adolescent medicine physician or ob.gyn.
EC can give our patients that sense of relief, especially when they have unprotected sex. However, they also need to have a sense of responsibility for their actions because you do not want them to engage in high-risk behaviors. Just as we are responsible to provide up-to-date care, our patients must take ownership of their health and well-being. Also If they are engaging in unprotected sex, they are just as responsible; therefore, they should know everything about contraception as well as EC. They should feel comfortable talking to their partners about contraception. Health care providers should make them feel comfortable receiving EC that they can give to their female partner.
We need to become knowledgeable and comfortable prescribing EC, as well as incorporating it in our routine care. This is a policy that I strongly believe should be part of every pediatrician’s and family physician’s office, especially when there is a lack of resources. Of the different options that are available, the oral forms of EC – especially Ella or Plan B step 1 (levonorgestrel) – would be the easiest to prescribe and counsel on. I would not recommend the options where multiple pills need to be taken more than once a day, because compliance becomes a factor. Also knowing that these options are available over the counter also is helpful because our community pharmacist also can help with medication administration and counseling.
In summary, I strongly recommend the discussion of EC in the office, especially the general pediatrician’s office. I recommend that, for those physicians’ who may be uncomfortable, that they should start with the “easier” options of oral progestins (Ella or Plan B step 1). As you become more comfortable with the information and counseling, you can learn skills such as IUD insertions, so you then can offer more options.
As health care providers to children, we always are learning. And with new knowledge we sometimes can be taken out of our comfort zone. One of those areas are teenagers, contraception, safe-sex counseling, and now emergency contraception (EC). In residency you have your 1-month adolescent medicine rotation to try and absorb every bit of information like a sponge, but there also will be a level of discomfort and uncertainty. However, as medical providers we cannot let the above prevent us from giving well-rounded and informed care.
When our teens disclose the most private moment of their life, we have to be armed and ready to not only comfort them, but advise and guide them to making a decision so that they can ensure their safety. The answers regarding sexual activity are becoming more and more alarming, especially in our younger patients. Therefore, this is an important discussion to have at every visit (not just well-child checks), so that education opportunities are not missed and our patients feel a sense of normalcy about discussing reproductive health with their health care provider and or parents.
We all have our personal beliefs, but we cannot let that guide our decision on what care or education we give our patients. Unfortunately, I have heard many health care providers judge our patients for their promiscuity, when we need to educate them – not be their judge and jury. Our teens go through different stages of growth and development, and with these stages come experimentation and risk taking. So as their health care providers, we need to be up to date on the information out there.
With regards with EC, some of our patients think that they can get it only after having unprotected sex. However, they should know that the oral ECs can be given to them at any time, so should they be in the situation above, they have an immediate remedy. With the different options come different counseling and different instructions on administration and follow-up. In residency, we might not have learned the skill of inserting an IUD, which is another form of EC; that is why there are many resources available. These resources include hands-on workshops, videos on counseling, and your friendly neighborhood adolescent medicine physician or ob.gyn.
EC can give our patients that sense of relief, especially when they have unprotected sex. However, they also need to have a sense of responsibility for their actions because you do not want them to engage in high-risk behaviors. Just as we are responsible to provide up-to-date care, our patients must take ownership of their health and well-being. Also If they are engaging in unprotected sex, they are just as responsible; therefore, they should know everything about contraception as well as EC. They should feel comfortable talking to their partners about contraception. Health care providers should make them feel comfortable receiving EC that they can give to their female partner.
We need to become knowledgeable and comfortable prescribing EC, as well as incorporating it in our routine care. This is a policy that I strongly believe should be part of every pediatrician’s and family physician’s office, especially when there is a lack of resources. Of the different options that are available, the oral forms of EC – especially Ella or Plan B step 1 (levonorgestrel) – would be the easiest to prescribe and counsel on. I would not recommend the options where multiple pills need to be taken more than once a day, because compliance becomes a factor. Also knowing that these options are available over the counter also is helpful because our community pharmacist also can help with medication administration and counseling.
In summary, I strongly recommend the discussion of EC in the office, especially the general pediatrician’s office. I recommend that, for those physicians’ who may be uncomfortable, that they should start with the “easier” options of oral progestins (Ella or Plan B step 1). As you become more comfortable with the information and counseling, you can learn skills such as IUD insertions, so you then can offer more options.
As health care providers to children, we always are learning. And with new knowledge we sometimes can be taken out of our comfort zone. One of those areas are teenagers, contraception, safe-sex counseling, and now emergency contraception (EC). In residency you have your 1-month adolescent medicine rotation to try and absorb every bit of information like a sponge, but there also will be a level of discomfort and uncertainty. However, as medical providers we cannot let the above prevent us from giving well-rounded and informed care.
When our teens disclose the most private moment of their life, we have to be armed and ready to not only comfort them, but advise and guide them to making a decision so that they can ensure their safety. The answers regarding sexual activity are becoming more and more alarming, especially in our younger patients. Therefore, this is an important discussion to have at every visit (not just well-child checks), so that education opportunities are not missed and our patients feel a sense of normalcy about discussing reproductive health with their health care provider and or parents.
We all have our personal beliefs, but we cannot let that guide our decision on what care or education we give our patients. Unfortunately, I have heard many health care providers judge our patients for their promiscuity, when we need to educate them – not be their judge and jury. Our teens go through different stages of growth and development, and with these stages come experimentation and risk taking. So as their health care providers, we need to be up to date on the information out there.
With regards with EC, some of our patients think that they can get it only after having unprotected sex. However, they should know that the oral ECs can be given to them at any time, so should they be in the situation above, they have an immediate remedy. With the different options come different counseling and different instructions on administration and follow-up. In residency, we might not have learned the skill of inserting an IUD, which is another form of EC; that is why there are many resources available. These resources include hands-on workshops, videos on counseling, and your friendly neighborhood adolescent medicine physician or ob.gyn.
EC can give our patients that sense of relief, especially when they have unprotected sex. However, they also need to have a sense of responsibility for their actions because you do not want them to engage in high-risk behaviors. Just as we are responsible to provide up-to-date care, our patients must take ownership of their health and well-being. Also If they are engaging in unprotected sex, they are just as responsible; therefore, they should know everything about contraception as well as EC. They should feel comfortable talking to their partners about contraception. Health care providers should make them feel comfortable receiving EC that they can give to their female partner.
We need to become knowledgeable and comfortable prescribing EC, as well as incorporating it in our routine care. This is a policy that I strongly believe should be part of every pediatrician’s and family physician’s office, especially when there is a lack of resources. Of the different options that are available, the oral forms of EC – especially Ella or Plan B step 1 (levonorgestrel) – would be the easiest to prescribe and counsel on. I would not recommend the options where multiple pills need to be taken more than once a day, because compliance becomes a factor. Also knowing that these options are available over the counter also is helpful because our community pharmacist also can help with medication administration and counseling.
In summary, I strongly recommend the discussion of EC in the office, especially the general pediatrician’s office. I recommend that, for those physicians’ who may be uncomfortable, that they should start with the “easier” options of oral progestins (Ella or Plan B step 1). As you become more comfortable with the information and counseling, you can learn skills such as IUD insertions, so you then can offer more options.
Elagolix is effective second-tier treatment for endometriosis-associated dysmenorrhea
PHILADELPHIA – Charles E. Miller, MD, said at the annual meeting of the American Society for Reproductive Medicine.
Although clinicians need prior authorization and evidence of treatment failure before prescribing Elagolix, the drug is a viable option as a second-tier treatment for patients with endometriosis-associated dysmenorrhea, said Dr. Miller, director of minimally invasive gynecologic surgery at Advocate Lutheran General Hospital in Park Ridge, Ill. “We have a drug that is very effective, that has a very low adverse event profile, and is tolerated by the vast majority of our patients.”
First-line options
NSAIDs are first-line treatment for endometriosis-related dysmenorrhea, with acetaminophen used in cases where NSAIDs are contraindicated or cause side effects such as gastrointestinal issues. Hormonal contraceptives also can be used as first-line treatment, divided into estrogen/progestin and progestin-only options that can be combined. Evidence from the literature has shown oral pills decrease pain, compared with placebo, but the decrease is not dose dependent, said Dr. Miller.
“We also know that if you use it continuously or prolonged, we find that there is going to be greater success with dysmenorrhea, and that ultimately you would use a higher-dose pill because of the greater risk of breakthrough when using a lesser dose in a continuous fashion,” he said. “Obviously if you’re not having menses, you’re not going to have dysmenorrhea.”
Other estrogen/progestin hormonal contraception such as the vaginal ring or transdermal patch also have been shown to decrease dysmenorrhea from endometriosis, with one study showing a reduction from 17% to 6% in moderate to severe dysmenorrhea in patients using the vaginal ring, compared with patients receiving oral contraceptives. In a separate randomized, controlled trial, “dysmenorrhea was more common in patch users, so it doesn’t appear that the patch is quite as effective in terms of reducing dysmenorrhea,” said Dr. Miller (JAMA. 2001 May 9. doi: 10.1001/jama.285.18.2347).
Compared with combination hormone therapy, there has been less research conducted on progestin-only hormone contraceptives on reducing dysmenorrhea from endometriosis. For example, there is little evidence for depot medroxyprogesterone acetate in reducing dysmenorrhea, but rather with it causing amenorrhea; one study showed a 50% amenorrhea rate at 1 year. “The disadvantage, however, in our infertile population is ultimately getting the menses back,” said Dr. Miller.
IUDs using levonorgestrel appear comparable with gonadotropin-releasing hormone (GnRH) agonists in reducing endometriosis-related pain; in one study, most women treated with either of these had visual analogue scores of less than 3 at 6 months of treatment. Between 68% and 75% of women with dysmenorrhea who receive an implantable contraceptive device with etonogestrel report decreased pain, and one meta-analysis reported 75% of women had “complete resolution of dysmenorrhea.” Concerning progestin-only pills, they can be used for endometriosis-related dysmenorrhea, but they are “problematic in that there’s a lot of breakthrough bleeding, and often times that is associated with pain,” said Dr. Miller.
Second-tier options
Injectable GnRH agonists are effective options as second-tier treatments for endometriosis-related dysmenorrhea, but patients are at risk of developing postmenopausal symptoms such as hot flashes, insomnia, spotting, and decreased libido. “One advantage to that is, over the years and particularly something that I’ve done with my endometriosis-related dysmenorrhea, is to utilize add-back with these patients,” said Dr. Miller, who noted that patients on 2.5 mg of norethindrone acetate and 0.5 mg of ethinyl estradiol“do very well” with that combination of add-back therapy.
Elagolix is the most recent second-tier treatment option for these patients, and was studied in the Elaris EM-I and Elaris EM-II trials in a once-daily dose of 150 mg and a twice-daily dose of 200 mg. In Elaris EM-1, 76% of patients in the 200-mg elagolix group had a clinical response, compared with 46% in the 150-mg group and 20% in the placebo group (N Engl J Med. 2017 Jul 6. doi: 10.1056/NEJMoa1700089). However, patients should not be on elagolix at 200 mg for more than 6 months, while patients receiving elagolix at 150 mg can stay on the treatment for up to 2 years.
Patients taking elagolix also showed postmenopausal symptoms, with 24% in the 150-mg group and 46% in the 200-mg group experiencing hot flashes, compared with 9% of patients in the placebo group. While 6% of patients in the 150-mg group and 10% in the 200-mg group discontinued because of adverse events, 1% and 3% of patients in the 150-mg and 200-mg group discontinued because of hot flashes or night sweats, respectively. “Symptoms are well tolerated, far different than in comparison with leuprolide acetate and GnRH agonists,” said Dr. Miller.
There also is a benefit to how patients recover from bone mineral density (BMD) changes after remaining on elagolix, Dr. Miller noted. In patients who received elagolix for 12 months at doses of 150 mg and 200 mg, there was an increase in lumbar spine BMD recovered 6 months after discontinuation, with patients in the 150-mg group experiencing a recovery close to baseline BMD levels. Among patients who discontinued treatment, there also was a quick resumption in menses for both groups: 87% of patients in the 150 mg group and 88% of patients in the 200-mg group who discontinued treatment after 6 months had resumed menses by 2 months after discontinuation, while 95% of patients in the 150-mg and 91% in the 200-mg group who discontinued after 12 months resumed menses by 2 months after discontinuation.
Dr. Miller reported relationships with AbbVie, Allergan, Blue Seas Med Spa, Espiner Medical, Gynesonics, Halt Medical, Hologic, Karl Storz, Medtronic, and Richard Wolf in the form of consultancies, grants, speakers’ bureau appointments, stock options, royalties, and ownership interests.
PHILADELPHIA – Charles E. Miller, MD, said at the annual meeting of the American Society for Reproductive Medicine.
Although clinicians need prior authorization and evidence of treatment failure before prescribing Elagolix, the drug is a viable option as a second-tier treatment for patients with endometriosis-associated dysmenorrhea, said Dr. Miller, director of minimally invasive gynecologic surgery at Advocate Lutheran General Hospital in Park Ridge, Ill. “We have a drug that is very effective, that has a very low adverse event profile, and is tolerated by the vast majority of our patients.”
First-line options
NSAIDs are first-line treatment for endometriosis-related dysmenorrhea, with acetaminophen used in cases where NSAIDs are contraindicated or cause side effects such as gastrointestinal issues. Hormonal contraceptives also can be used as first-line treatment, divided into estrogen/progestin and progestin-only options that can be combined. Evidence from the literature has shown oral pills decrease pain, compared with placebo, but the decrease is not dose dependent, said Dr. Miller.
“We also know that if you use it continuously or prolonged, we find that there is going to be greater success with dysmenorrhea, and that ultimately you would use a higher-dose pill because of the greater risk of breakthrough when using a lesser dose in a continuous fashion,” he said. “Obviously if you’re not having menses, you’re not going to have dysmenorrhea.”
Other estrogen/progestin hormonal contraception such as the vaginal ring or transdermal patch also have been shown to decrease dysmenorrhea from endometriosis, with one study showing a reduction from 17% to 6% in moderate to severe dysmenorrhea in patients using the vaginal ring, compared with patients receiving oral contraceptives. In a separate randomized, controlled trial, “dysmenorrhea was more common in patch users, so it doesn’t appear that the patch is quite as effective in terms of reducing dysmenorrhea,” said Dr. Miller (JAMA. 2001 May 9. doi: 10.1001/jama.285.18.2347).
Compared with combination hormone therapy, there has been less research conducted on progestin-only hormone contraceptives on reducing dysmenorrhea from endometriosis. For example, there is little evidence for depot medroxyprogesterone acetate in reducing dysmenorrhea, but rather with it causing amenorrhea; one study showed a 50% amenorrhea rate at 1 year. “The disadvantage, however, in our infertile population is ultimately getting the menses back,” said Dr. Miller.
IUDs using levonorgestrel appear comparable with gonadotropin-releasing hormone (GnRH) agonists in reducing endometriosis-related pain; in one study, most women treated with either of these had visual analogue scores of less than 3 at 6 months of treatment. Between 68% and 75% of women with dysmenorrhea who receive an implantable contraceptive device with etonogestrel report decreased pain, and one meta-analysis reported 75% of women had “complete resolution of dysmenorrhea.” Concerning progestin-only pills, they can be used for endometriosis-related dysmenorrhea, but they are “problematic in that there’s a lot of breakthrough bleeding, and often times that is associated with pain,” said Dr. Miller.
Second-tier options
Injectable GnRH agonists are effective options as second-tier treatments for endometriosis-related dysmenorrhea, but patients are at risk of developing postmenopausal symptoms such as hot flashes, insomnia, spotting, and decreased libido. “One advantage to that is, over the years and particularly something that I’ve done with my endometriosis-related dysmenorrhea, is to utilize add-back with these patients,” said Dr. Miller, who noted that patients on 2.5 mg of norethindrone acetate and 0.5 mg of ethinyl estradiol“do very well” with that combination of add-back therapy.
Elagolix is the most recent second-tier treatment option for these patients, and was studied in the Elaris EM-I and Elaris EM-II trials in a once-daily dose of 150 mg and a twice-daily dose of 200 mg. In Elaris EM-1, 76% of patients in the 200-mg elagolix group had a clinical response, compared with 46% in the 150-mg group and 20% in the placebo group (N Engl J Med. 2017 Jul 6. doi: 10.1056/NEJMoa1700089). However, patients should not be on elagolix at 200 mg for more than 6 months, while patients receiving elagolix at 150 mg can stay on the treatment for up to 2 years.
Patients taking elagolix also showed postmenopausal symptoms, with 24% in the 150-mg group and 46% in the 200-mg group experiencing hot flashes, compared with 9% of patients in the placebo group. While 6% of patients in the 150-mg group and 10% in the 200-mg group discontinued because of adverse events, 1% and 3% of patients in the 150-mg and 200-mg group discontinued because of hot flashes or night sweats, respectively. “Symptoms are well tolerated, far different than in comparison with leuprolide acetate and GnRH agonists,” said Dr. Miller.
There also is a benefit to how patients recover from bone mineral density (BMD) changes after remaining on elagolix, Dr. Miller noted. In patients who received elagolix for 12 months at doses of 150 mg and 200 mg, there was an increase in lumbar spine BMD recovered 6 months after discontinuation, with patients in the 150-mg group experiencing a recovery close to baseline BMD levels. Among patients who discontinued treatment, there also was a quick resumption in menses for both groups: 87% of patients in the 150 mg group and 88% of patients in the 200-mg group who discontinued treatment after 6 months had resumed menses by 2 months after discontinuation, while 95% of patients in the 150-mg and 91% in the 200-mg group who discontinued after 12 months resumed menses by 2 months after discontinuation.
Dr. Miller reported relationships with AbbVie, Allergan, Blue Seas Med Spa, Espiner Medical, Gynesonics, Halt Medical, Hologic, Karl Storz, Medtronic, and Richard Wolf in the form of consultancies, grants, speakers’ bureau appointments, stock options, royalties, and ownership interests.
PHILADELPHIA – Charles E. Miller, MD, said at the annual meeting of the American Society for Reproductive Medicine.
Although clinicians need prior authorization and evidence of treatment failure before prescribing Elagolix, the drug is a viable option as a second-tier treatment for patients with endometriosis-associated dysmenorrhea, said Dr. Miller, director of minimally invasive gynecologic surgery at Advocate Lutheran General Hospital in Park Ridge, Ill. “We have a drug that is very effective, that has a very low adverse event profile, and is tolerated by the vast majority of our patients.”
First-line options
NSAIDs are first-line treatment for endometriosis-related dysmenorrhea, with acetaminophen used in cases where NSAIDs are contraindicated or cause side effects such as gastrointestinal issues. Hormonal contraceptives also can be used as first-line treatment, divided into estrogen/progestin and progestin-only options that can be combined. Evidence from the literature has shown oral pills decrease pain, compared with placebo, but the decrease is not dose dependent, said Dr. Miller.
“We also know that if you use it continuously or prolonged, we find that there is going to be greater success with dysmenorrhea, and that ultimately you would use a higher-dose pill because of the greater risk of breakthrough when using a lesser dose in a continuous fashion,” he said. “Obviously if you’re not having menses, you’re not going to have dysmenorrhea.”
Other estrogen/progestin hormonal contraception such as the vaginal ring or transdermal patch also have been shown to decrease dysmenorrhea from endometriosis, with one study showing a reduction from 17% to 6% in moderate to severe dysmenorrhea in patients using the vaginal ring, compared with patients receiving oral contraceptives. In a separate randomized, controlled trial, “dysmenorrhea was more common in patch users, so it doesn’t appear that the patch is quite as effective in terms of reducing dysmenorrhea,” said Dr. Miller (JAMA. 2001 May 9. doi: 10.1001/jama.285.18.2347).
Compared with combination hormone therapy, there has been less research conducted on progestin-only hormone contraceptives on reducing dysmenorrhea from endometriosis. For example, there is little evidence for depot medroxyprogesterone acetate in reducing dysmenorrhea, but rather with it causing amenorrhea; one study showed a 50% amenorrhea rate at 1 year. “The disadvantage, however, in our infertile population is ultimately getting the menses back,” said Dr. Miller.
IUDs using levonorgestrel appear comparable with gonadotropin-releasing hormone (GnRH) agonists in reducing endometriosis-related pain; in one study, most women treated with either of these had visual analogue scores of less than 3 at 6 months of treatment. Between 68% and 75% of women with dysmenorrhea who receive an implantable contraceptive device with etonogestrel report decreased pain, and one meta-analysis reported 75% of women had “complete resolution of dysmenorrhea.” Concerning progestin-only pills, they can be used for endometriosis-related dysmenorrhea, but they are “problematic in that there’s a lot of breakthrough bleeding, and often times that is associated with pain,” said Dr. Miller.
Second-tier options
Injectable GnRH agonists are effective options as second-tier treatments for endometriosis-related dysmenorrhea, but patients are at risk of developing postmenopausal symptoms such as hot flashes, insomnia, spotting, and decreased libido. “One advantage to that is, over the years and particularly something that I’ve done with my endometriosis-related dysmenorrhea, is to utilize add-back with these patients,” said Dr. Miller, who noted that patients on 2.5 mg of norethindrone acetate and 0.5 mg of ethinyl estradiol“do very well” with that combination of add-back therapy.
Elagolix is the most recent second-tier treatment option for these patients, and was studied in the Elaris EM-I and Elaris EM-II trials in a once-daily dose of 150 mg and a twice-daily dose of 200 mg. In Elaris EM-1, 76% of patients in the 200-mg elagolix group had a clinical response, compared with 46% in the 150-mg group and 20% in the placebo group (N Engl J Med. 2017 Jul 6. doi: 10.1056/NEJMoa1700089). However, patients should not be on elagolix at 200 mg for more than 6 months, while patients receiving elagolix at 150 mg can stay on the treatment for up to 2 years.
Patients taking elagolix also showed postmenopausal symptoms, with 24% in the 150-mg group and 46% in the 200-mg group experiencing hot flashes, compared with 9% of patients in the placebo group. While 6% of patients in the 150-mg group and 10% in the 200-mg group discontinued because of adverse events, 1% and 3% of patients in the 150-mg and 200-mg group discontinued because of hot flashes or night sweats, respectively. “Symptoms are well tolerated, far different than in comparison with leuprolide acetate and GnRH agonists,” said Dr. Miller.
There also is a benefit to how patients recover from bone mineral density (BMD) changes after remaining on elagolix, Dr. Miller noted. In patients who received elagolix for 12 months at doses of 150 mg and 200 mg, there was an increase in lumbar spine BMD recovered 6 months after discontinuation, with patients in the 150-mg group experiencing a recovery close to baseline BMD levels. Among patients who discontinued treatment, there also was a quick resumption in menses for both groups: 87% of patients in the 150 mg group and 88% of patients in the 200-mg group who discontinued treatment after 6 months had resumed menses by 2 months after discontinuation, while 95% of patients in the 150-mg and 91% in the 200-mg group who discontinued after 12 months resumed menses by 2 months after discontinuation.
Dr. Miller reported relationships with AbbVie, Allergan, Blue Seas Med Spa, Espiner Medical, Gynesonics, Halt Medical, Hologic, Karl Storz, Medtronic, and Richard Wolf in the form of consultancies, grants, speakers’ bureau appointments, stock options, royalties, and ownership interests.
EXPERT ANALYSIS FROM ASRM 2019
Cannabis frequently is used for endometriosis pain
VANCOUVER, B.C. – with over a third reporting either current or past use, according to a new survey.
The finding comes as more and more companies are marketing CBD-containing products to women, with unsubstantiated claims about efficacy, according to Anna Reinert, MD, who presented the research at a meeting sponsored by AAGL.
Women self-reported that marijuana use was moderately effective, while the median value for CBD corresponded to “slightly effective.”
To investigate use patterns, Dr. Reinert and colleagues created a questionnaire with 55-75 questions, which followed a branching logic tree. Topics included pain history, demographics, and experience with marijuana and CBD for the purpose of controlling pelvic pain. The survey was sent to two populations: an endometriosis association mailing list, and patients at a chronic pain center in Phoenix.
About 24,500 surveys were sent out; 366 were received and analyzed. The response rate was much different between the two populations, at 1% in the endometriosis association and 16% of the clinic population. Dr. Reinert attributed the low response rate in the association sample to the continuing stigma surrounding marijuana use, citing much higher response rates to other surveys sent out by the association around the same time.
Overall, 63% of respondents said they had never used marijuana; 37% reported past or present use; 65% said they had never used CBD; and 35% reported past or present use. About 45% of marijuana users reported that its use was very effective, and 25% said it was moderately effective. About 22% of CBD users said it was very effective, and about 33% said it was moderately effective. The median values lay in the moderately effective range for marijuana, and in the slightly effective range for CBD.
The findings suggest a need for more research into the potential benefit and limitations of cannabis for pelvic pain from endometriosis, said Dr. Reinert, an obstetrician/gynecologist the University of Southern California, Los Angeles.
Until this study, evidence of efficacy of marijuana for this indication has been sparse. A report from the National Academy of Sciences showed that there is evidence that cannabis and cannabinoids have a therapeutic effect on chronic pain in adults (National Academies Press (US) 2017 Jan 12), but the report made no mention of gynecological applications. Despite this lack of evidence, surveys have shown that women of reproductive age use marijuana, and an analysis by the Ameritox Laboratory in a pain management population found that 13% of women and 19% of men tested positive for marijuana in their urine.
Still, “there is not research looking at marijuana for women with chronic health pain,” Dr. Reinert said at the meeting.
But that doesn’t stop companies from developing CBD vaginal suppositories and marketing them for menstrual pelvic discomfort, pain during sex, and other issues. Lay press articles often boost these claims, although some skeptical takes address the lack of evidence. Still, “there’s a lot on the more positive side,” she said.
That leads to a lot of interest among patients in using marijuana or CBD for symptom relief, which is part of the reason that Dr. Reinert’s team decided to examine its use and perceived efficacy. Another reason is that there is some biological basis to believe that cannabis could be helpful. There is some evidence that women with endometriosis have changes in their endocannabinoid system (Cannabis Cannabinoid Res. 2017;2:72-80), and there are clinical trials examining the impact of non-CBD, non-tetrahydrocannabinol (THC) endocannabinoid ligands.
Dr. Reinert has no financial disclosures.
VANCOUVER, B.C. – with over a third reporting either current or past use, according to a new survey.
The finding comes as more and more companies are marketing CBD-containing products to women, with unsubstantiated claims about efficacy, according to Anna Reinert, MD, who presented the research at a meeting sponsored by AAGL.
Women self-reported that marijuana use was moderately effective, while the median value for CBD corresponded to “slightly effective.”
To investigate use patterns, Dr. Reinert and colleagues created a questionnaire with 55-75 questions, which followed a branching logic tree. Topics included pain history, demographics, and experience with marijuana and CBD for the purpose of controlling pelvic pain. The survey was sent to two populations: an endometriosis association mailing list, and patients at a chronic pain center in Phoenix.
About 24,500 surveys were sent out; 366 were received and analyzed. The response rate was much different between the two populations, at 1% in the endometriosis association and 16% of the clinic population. Dr. Reinert attributed the low response rate in the association sample to the continuing stigma surrounding marijuana use, citing much higher response rates to other surveys sent out by the association around the same time.
Overall, 63% of respondents said they had never used marijuana; 37% reported past or present use; 65% said they had never used CBD; and 35% reported past or present use. About 45% of marijuana users reported that its use was very effective, and 25% said it was moderately effective. About 22% of CBD users said it was very effective, and about 33% said it was moderately effective. The median values lay in the moderately effective range for marijuana, and in the slightly effective range for CBD.
The findings suggest a need for more research into the potential benefit and limitations of cannabis for pelvic pain from endometriosis, said Dr. Reinert, an obstetrician/gynecologist the University of Southern California, Los Angeles.
Until this study, evidence of efficacy of marijuana for this indication has been sparse. A report from the National Academy of Sciences showed that there is evidence that cannabis and cannabinoids have a therapeutic effect on chronic pain in adults (National Academies Press (US) 2017 Jan 12), but the report made no mention of gynecological applications. Despite this lack of evidence, surveys have shown that women of reproductive age use marijuana, and an analysis by the Ameritox Laboratory in a pain management population found that 13% of women and 19% of men tested positive for marijuana in their urine.
Still, “there is not research looking at marijuana for women with chronic health pain,” Dr. Reinert said at the meeting.
But that doesn’t stop companies from developing CBD vaginal suppositories and marketing them for menstrual pelvic discomfort, pain during sex, and other issues. Lay press articles often boost these claims, although some skeptical takes address the lack of evidence. Still, “there’s a lot on the more positive side,” she said.
That leads to a lot of interest among patients in using marijuana or CBD for symptom relief, which is part of the reason that Dr. Reinert’s team decided to examine its use and perceived efficacy. Another reason is that there is some biological basis to believe that cannabis could be helpful. There is some evidence that women with endometriosis have changes in their endocannabinoid system (Cannabis Cannabinoid Res. 2017;2:72-80), and there are clinical trials examining the impact of non-CBD, non-tetrahydrocannabinol (THC) endocannabinoid ligands.
Dr. Reinert has no financial disclosures.
VANCOUVER, B.C. – with over a third reporting either current or past use, according to a new survey.
The finding comes as more and more companies are marketing CBD-containing products to women, with unsubstantiated claims about efficacy, according to Anna Reinert, MD, who presented the research at a meeting sponsored by AAGL.
Women self-reported that marijuana use was moderately effective, while the median value for CBD corresponded to “slightly effective.”
To investigate use patterns, Dr. Reinert and colleagues created a questionnaire with 55-75 questions, which followed a branching logic tree. Topics included pain history, demographics, and experience with marijuana and CBD for the purpose of controlling pelvic pain. The survey was sent to two populations: an endometriosis association mailing list, and patients at a chronic pain center in Phoenix.
About 24,500 surveys were sent out; 366 were received and analyzed. The response rate was much different between the two populations, at 1% in the endometriosis association and 16% of the clinic population. Dr. Reinert attributed the low response rate in the association sample to the continuing stigma surrounding marijuana use, citing much higher response rates to other surveys sent out by the association around the same time.
Overall, 63% of respondents said they had never used marijuana; 37% reported past or present use; 65% said they had never used CBD; and 35% reported past or present use. About 45% of marijuana users reported that its use was very effective, and 25% said it was moderately effective. About 22% of CBD users said it was very effective, and about 33% said it was moderately effective. The median values lay in the moderately effective range for marijuana, and in the slightly effective range for CBD.
The findings suggest a need for more research into the potential benefit and limitations of cannabis for pelvic pain from endometriosis, said Dr. Reinert, an obstetrician/gynecologist the University of Southern California, Los Angeles.
Until this study, evidence of efficacy of marijuana for this indication has been sparse. A report from the National Academy of Sciences showed that there is evidence that cannabis and cannabinoids have a therapeutic effect on chronic pain in adults (National Academies Press (US) 2017 Jan 12), but the report made no mention of gynecological applications. Despite this lack of evidence, surveys have shown that women of reproductive age use marijuana, and an analysis by the Ameritox Laboratory in a pain management population found that 13% of women and 19% of men tested positive for marijuana in their urine.
Still, “there is not research looking at marijuana for women with chronic health pain,” Dr. Reinert said at the meeting.
But that doesn’t stop companies from developing CBD vaginal suppositories and marketing them for menstrual pelvic discomfort, pain during sex, and other issues. Lay press articles often boost these claims, although some skeptical takes address the lack of evidence. Still, “there’s a lot on the more positive side,” she said.
That leads to a lot of interest among patients in using marijuana or CBD for symptom relief, which is part of the reason that Dr. Reinert’s team decided to examine its use and perceived efficacy. Another reason is that there is some biological basis to believe that cannabis could be helpful. There is some evidence that women with endometriosis have changes in their endocannabinoid system (Cannabis Cannabinoid Res. 2017;2:72-80), and there are clinical trials examining the impact of non-CBD, non-tetrahydrocannabinol (THC) endocannabinoid ligands.
Dr. Reinert has no financial disclosures.
REPORTING FROM THE AAGL GLOBAL CONGRESS
Women with obesity need not boost calories during pregnancy
LAS VEGAS – Contrary to current U.S. dietary recommendations for pregnancy, women with obesity should not increase their energy intake during pregnancy to achieve the current recommended level of gestational weight gain, based on findings from an intensive assessment of 54 women with obesity during weeks 13-37 of pregnancy.
To achieve the gestational weight gain of 11-20 pounds (5-9.1 kg) recommended by the Institute of Medicine, women with obesity ‒ those with a body mass index of 30 kg/m2 or greater ‒ had an average energy intake during the second and third trimesters of 125 kcal/day less than their energy expenditure, Leanne M. Redman, PhD, said at a meeting presented by the Obesity Society and the American Society for Metabolic and Bariatric Surgery.
However, women in the study who had inadequate gestational weight gain had a daily calorie deficit that was only slightly larger, an average of 262 kcal/day below their energy expenditure. As a consequence, Dr. Redman believes the take-home message from her findings is that pregnant women with obesity should maintain their prepregnancy energy intake, though she also strongly recommended improvements in diet quality.
“Chasing a 100-kcal/day deficit in intake is extremely problematic,” Dr. Redman admitted, so she suggested that women with obesity be advised simply to not increase their calorie intake during pregnancy.
“The message is: Focus on improving diet quality rather than increasing calories,” she said in an interview. Pregnant women with obesity “do not need to increase calorie intake. They need to improve their diet quality,” with increased consumption of fruits and vegetables, said Dr. Redman, a professor and director of the Reproductive Endocrinology and Women’s Health Laboratory at Louisiana State University’s Pennington Biomedical Research Center in Baton Rouge.
The results she reported represent “the first time” researchers have examined energy expenditure and weight-gain trajectories in women with obesity throughout the second and third trimesters. Until now, dietary energy recommendations for women with obesity during pregnancy were based on observations made in women without obesity.
Those observations led the Institute of Medicine to call for a recommended pregnancy weight gain of 11-20 pounds in women with obesity, as well as gains of 25-35 pounds in women with a normal body mass index of 18.5-24.9 kg/m2 (Weight Gain During Pregnancy: Reexamining the Guidelines; May 2009). In that 2009 document, the IOM committee said that, in general, pregnant women should add 340 kcal/day to their prepregnancy intake during the second trimester and add 452 kcal/day during the third trimester without regard to their prepregnancy body mass index, a recommendation that clinicians continued to promote in subsequent years (Med Clin North Amer. 2016;100[6]:1199-215), and that was generally affirmed by the American College of Obstetricians and Gynecologists in 2013 and reaffirmed in 2018.*
The new evidence collected by Dr. Redman and associates “challenges current practice and argues that women with obesity should not be advised to consume additional energy during pregnancy as currently recommended,” they wrote in an article with their findings published a few days before Dr. Redman gave her talk (J Clin Invest. 2019;129[11]:4682-90).
The MomEE (Determinants of Gestational Weight Gain in Obese Pregnant Women) study enrolled 72 women with obesity during the first trimester of pregnancy and collected complete data through the end of the third trimester from 54 women. The researchers collected data on weight, body fat mass, and energy expenditure at multiple times during the second and third trimesters and calculated energy intake.
Based on body weights at the end of the third trimester, the researchers divided the 54 women into three subgroups: 10 women (19%) with inadequate weight gain by the IOM recommendations, 8 (15%) who had the IOM’s recommended weight gain of 11-20 pounds, and 36 women (67%; total is greater than 100% because of rounding) with excess weight gain, and within each group, they calculated the average level of energy intake relative to energy expenditure.
In addition to the daily calorie deficits associated with women who maintained recommended or inadequate weight, the researchers also found that women with excess weight gain averaged 186 more kcal/day than required to meet their daily energy expenditure.
The analyses showed that the increased energy demand of pregnancy and the fetus is compensated for by mobilization of the maternal fat mass in women with obesity, and that an imbalance between energy intake and expenditure is the main driver of weight gain during pregnancy. The results also highlighted how often pregnant women with obesity fail to follow a diet that results in the recommended weight gain of 11-20 pounds. In the MomEE cohort, two-thirds of enrolled women had excess weight gain.
The finding that women had the recommended weight gain on a diet that cut their daily calorie intake by about 100 kcal/day during the last two trimesters highlighted the nutritional challenge faced by women with obesity who are pregnant. “About three-quarters of women in the study had poor diet quality. There is an opportunity to improve diet with more fruits and vegetables to increase fullness, and [to reduce] energy-dense foods,” Dr. Redman said.
She is planning to collaborate on a study that will test the efficacy and safety of providing pregnant women with extreme obesity (class II-III) with defined meals to provide better control of energy intake and nutritional quality. Dr. Redman said she also hoped that the new findings she reported would be taken into account by the advisory committee assembled by the Department of Health & Human Services and the Department of Agriculture, which are currently preparing a revision of U.S. dietary guidelines for release in 2020.
The National Institutes of Health and the Clinical Research Cores at Pennington Biomedical Research Center funded the study. Dr. Redman had no disclosures.
SOURCE: Redman LM et al. Obesity Week 2019, Abstract T-OR-2079.
*This article was updated 2/7/2020.
The results reported by Dr. Redman from the MomEE study showed that women with obesity need not ingest surplus calories to gain weight during pregnancy. The findings indicate that pregnant women efficiently convert a portion of their accumulated fat mass to fat-free mass in the form of the fetus, uterus, blood volume, and other tissue. A deficit of about approximately 100 kcal/day effectively kept weight gain within the 11- to 20-pound target recommended by the Institute of Medicine in 2009.
But the weight gains recommended for women with obesity may be too high. The desire of the writers of the IOM recommendation to avoid negative perinatal outcomes for infants may instead lead to negative maternal outcomes, such as preeclampsia, gestational hypertension, and need for cesarean birth. Gestational weight gains below what the IOM recommended for women with obesity may be able to serve present-day standards and work better for these pregnant women by reducing their morbidity risk. Future studies should take into careful account overall nutrient values rather than just calorie intake, as well as physical activity.
The MomEE results showed that a striking two-thirds of women with obesity gained an excess of weight during pregnancy, beyond the 2009 recommendations. This finding highlights the need to identify strategies that can prevent excessive weight gain. Furthermore, results from several studies and systematic reviews suggest that the IOM recommendation for weight gain during pregnancy is too high for women with obesity, especially those with class II-III obesity, with a body mass index of 35 kg/m2 or greater. In my opinion, an appropriate weight-gain target to replace the current, blanket recommendation of 11-20 pounds gained for all women with obesity is a target of 5-15 pounds gained for women with class I obesity, less than 10 pounds for class II obesity, and no change in prepregnancy weight for women with class III obesity.
Sarah S. Comstock, PhD, is a nutrition researcher at Michigan State University, East Lansing. She is an inventor named on three patents that involve nutrition. She made these comments in an editorial that accompanied the MomEE report (J Clin Invest. 2019;129[11]:4567-9).
The results reported by Dr. Redman from the MomEE study showed that women with obesity need not ingest surplus calories to gain weight during pregnancy. The findings indicate that pregnant women efficiently convert a portion of their accumulated fat mass to fat-free mass in the form of the fetus, uterus, blood volume, and other tissue. A deficit of about approximately 100 kcal/day effectively kept weight gain within the 11- to 20-pound target recommended by the Institute of Medicine in 2009.
But the weight gains recommended for women with obesity may be too high. The desire of the writers of the IOM recommendation to avoid negative perinatal outcomes for infants may instead lead to negative maternal outcomes, such as preeclampsia, gestational hypertension, and need for cesarean birth. Gestational weight gains below what the IOM recommended for women with obesity may be able to serve present-day standards and work better for these pregnant women by reducing their morbidity risk. Future studies should take into careful account overall nutrient values rather than just calorie intake, as well as physical activity.
The MomEE results showed that a striking two-thirds of women with obesity gained an excess of weight during pregnancy, beyond the 2009 recommendations. This finding highlights the need to identify strategies that can prevent excessive weight gain. Furthermore, results from several studies and systematic reviews suggest that the IOM recommendation for weight gain during pregnancy is too high for women with obesity, especially those with class II-III obesity, with a body mass index of 35 kg/m2 or greater. In my opinion, an appropriate weight-gain target to replace the current, blanket recommendation of 11-20 pounds gained for all women with obesity is a target of 5-15 pounds gained for women with class I obesity, less than 10 pounds for class II obesity, and no change in prepregnancy weight for women with class III obesity.
Sarah S. Comstock, PhD, is a nutrition researcher at Michigan State University, East Lansing. She is an inventor named on three patents that involve nutrition. She made these comments in an editorial that accompanied the MomEE report (J Clin Invest. 2019;129[11]:4567-9).
The results reported by Dr. Redman from the MomEE study showed that women with obesity need not ingest surplus calories to gain weight during pregnancy. The findings indicate that pregnant women efficiently convert a portion of their accumulated fat mass to fat-free mass in the form of the fetus, uterus, blood volume, and other tissue. A deficit of about approximately 100 kcal/day effectively kept weight gain within the 11- to 20-pound target recommended by the Institute of Medicine in 2009.
But the weight gains recommended for women with obesity may be too high. The desire of the writers of the IOM recommendation to avoid negative perinatal outcomes for infants may instead lead to negative maternal outcomes, such as preeclampsia, gestational hypertension, and need for cesarean birth. Gestational weight gains below what the IOM recommended for women with obesity may be able to serve present-day standards and work better for these pregnant women by reducing their morbidity risk. Future studies should take into careful account overall nutrient values rather than just calorie intake, as well as physical activity.
The MomEE results showed that a striking two-thirds of women with obesity gained an excess of weight during pregnancy, beyond the 2009 recommendations. This finding highlights the need to identify strategies that can prevent excessive weight gain. Furthermore, results from several studies and systematic reviews suggest that the IOM recommendation for weight gain during pregnancy is too high for women with obesity, especially those with class II-III obesity, with a body mass index of 35 kg/m2 or greater. In my opinion, an appropriate weight-gain target to replace the current, blanket recommendation of 11-20 pounds gained for all women with obesity is a target of 5-15 pounds gained for women with class I obesity, less than 10 pounds for class II obesity, and no change in prepregnancy weight for women with class III obesity.
Sarah S. Comstock, PhD, is a nutrition researcher at Michigan State University, East Lansing. She is an inventor named on three patents that involve nutrition. She made these comments in an editorial that accompanied the MomEE report (J Clin Invest. 2019;129[11]:4567-9).
LAS VEGAS – Contrary to current U.S. dietary recommendations for pregnancy, women with obesity should not increase their energy intake during pregnancy to achieve the current recommended level of gestational weight gain, based on findings from an intensive assessment of 54 women with obesity during weeks 13-37 of pregnancy.
To achieve the gestational weight gain of 11-20 pounds (5-9.1 kg) recommended by the Institute of Medicine, women with obesity ‒ those with a body mass index of 30 kg/m2 or greater ‒ had an average energy intake during the second and third trimesters of 125 kcal/day less than their energy expenditure, Leanne M. Redman, PhD, said at a meeting presented by the Obesity Society and the American Society for Metabolic and Bariatric Surgery.
However, women in the study who had inadequate gestational weight gain had a daily calorie deficit that was only slightly larger, an average of 262 kcal/day below their energy expenditure. As a consequence, Dr. Redman believes the take-home message from her findings is that pregnant women with obesity should maintain their prepregnancy energy intake, though she also strongly recommended improvements in diet quality.
“Chasing a 100-kcal/day deficit in intake is extremely problematic,” Dr. Redman admitted, so she suggested that women with obesity be advised simply to not increase their calorie intake during pregnancy.
“The message is: Focus on improving diet quality rather than increasing calories,” she said in an interview. Pregnant women with obesity “do not need to increase calorie intake. They need to improve their diet quality,” with increased consumption of fruits and vegetables, said Dr. Redman, a professor and director of the Reproductive Endocrinology and Women’s Health Laboratory at Louisiana State University’s Pennington Biomedical Research Center in Baton Rouge.
The results she reported represent “the first time” researchers have examined energy expenditure and weight-gain trajectories in women with obesity throughout the second and third trimesters. Until now, dietary energy recommendations for women with obesity during pregnancy were based on observations made in women without obesity.
Those observations led the Institute of Medicine to call for a recommended pregnancy weight gain of 11-20 pounds in women with obesity, as well as gains of 25-35 pounds in women with a normal body mass index of 18.5-24.9 kg/m2 (Weight Gain During Pregnancy: Reexamining the Guidelines; May 2009). In that 2009 document, the IOM committee said that, in general, pregnant women should add 340 kcal/day to their prepregnancy intake during the second trimester and add 452 kcal/day during the third trimester without regard to their prepregnancy body mass index, a recommendation that clinicians continued to promote in subsequent years (Med Clin North Amer. 2016;100[6]:1199-215), and that was generally affirmed by the American College of Obstetricians and Gynecologists in 2013 and reaffirmed in 2018.*
The new evidence collected by Dr. Redman and associates “challenges current practice and argues that women with obesity should not be advised to consume additional energy during pregnancy as currently recommended,” they wrote in an article with their findings published a few days before Dr. Redman gave her talk (J Clin Invest. 2019;129[11]:4682-90).
The MomEE (Determinants of Gestational Weight Gain in Obese Pregnant Women) study enrolled 72 women with obesity during the first trimester of pregnancy and collected complete data through the end of the third trimester from 54 women. The researchers collected data on weight, body fat mass, and energy expenditure at multiple times during the second and third trimesters and calculated energy intake.
Based on body weights at the end of the third trimester, the researchers divided the 54 women into three subgroups: 10 women (19%) with inadequate weight gain by the IOM recommendations, 8 (15%) who had the IOM’s recommended weight gain of 11-20 pounds, and 36 women (67%; total is greater than 100% because of rounding) with excess weight gain, and within each group, they calculated the average level of energy intake relative to energy expenditure.
In addition to the daily calorie deficits associated with women who maintained recommended or inadequate weight, the researchers also found that women with excess weight gain averaged 186 more kcal/day than required to meet their daily energy expenditure.
The analyses showed that the increased energy demand of pregnancy and the fetus is compensated for by mobilization of the maternal fat mass in women with obesity, and that an imbalance between energy intake and expenditure is the main driver of weight gain during pregnancy. The results also highlighted how often pregnant women with obesity fail to follow a diet that results in the recommended weight gain of 11-20 pounds. In the MomEE cohort, two-thirds of enrolled women had excess weight gain.
The finding that women had the recommended weight gain on a diet that cut their daily calorie intake by about 100 kcal/day during the last two trimesters highlighted the nutritional challenge faced by women with obesity who are pregnant. “About three-quarters of women in the study had poor diet quality. There is an opportunity to improve diet with more fruits and vegetables to increase fullness, and [to reduce] energy-dense foods,” Dr. Redman said.
She is planning to collaborate on a study that will test the efficacy and safety of providing pregnant women with extreme obesity (class II-III) with defined meals to provide better control of energy intake and nutritional quality. Dr. Redman said she also hoped that the new findings she reported would be taken into account by the advisory committee assembled by the Department of Health & Human Services and the Department of Agriculture, which are currently preparing a revision of U.S. dietary guidelines for release in 2020.
The National Institutes of Health and the Clinical Research Cores at Pennington Biomedical Research Center funded the study. Dr. Redman had no disclosures.
SOURCE: Redman LM et al. Obesity Week 2019, Abstract T-OR-2079.
*This article was updated 2/7/2020.
LAS VEGAS – Contrary to current U.S. dietary recommendations for pregnancy, women with obesity should not increase their energy intake during pregnancy to achieve the current recommended level of gestational weight gain, based on findings from an intensive assessment of 54 women with obesity during weeks 13-37 of pregnancy.
To achieve the gestational weight gain of 11-20 pounds (5-9.1 kg) recommended by the Institute of Medicine, women with obesity ‒ those with a body mass index of 30 kg/m2 or greater ‒ had an average energy intake during the second and third trimesters of 125 kcal/day less than their energy expenditure, Leanne M. Redman, PhD, said at a meeting presented by the Obesity Society and the American Society for Metabolic and Bariatric Surgery.
However, women in the study who had inadequate gestational weight gain had a daily calorie deficit that was only slightly larger, an average of 262 kcal/day below their energy expenditure. As a consequence, Dr. Redman believes the take-home message from her findings is that pregnant women with obesity should maintain their prepregnancy energy intake, though she also strongly recommended improvements in diet quality.
“Chasing a 100-kcal/day deficit in intake is extremely problematic,” Dr. Redman admitted, so she suggested that women with obesity be advised simply to not increase their calorie intake during pregnancy.
“The message is: Focus on improving diet quality rather than increasing calories,” she said in an interview. Pregnant women with obesity “do not need to increase calorie intake. They need to improve their diet quality,” with increased consumption of fruits and vegetables, said Dr. Redman, a professor and director of the Reproductive Endocrinology and Women’s Health Laboratory at Louisiana State University’s Pennington Biomedical Research Center in Baton Rouge.
The results she reported represent “the first time” researchers have examined energy expenditure and weight-gain trajectories in women with obesity throughout the second and third trimesters. Until now, dietary energy recommendations for women with obesity during pregnancy were based on observations made in women without obesity.
Those observations led the Institute of Medicine to call for a recommended pregnancy weight gain of 11-20 pounds in women with obesity, as well as gains of 25-35 pounds in women with a normal body mass index of 18.5-24.9 kg/m2 (Weight Gain During Pregnancy: Reexamining the Guidelines; May 2009). In that 2009 document, the IOM committee said that, in general, pregnant women should add 340 kcal/day to their prepregnancy intake during the second trimester and add 452 kcal/day during the third trimester without regard to their prepregnancy body mass index, a recommendation that clinicians continued to promote in subsequent years (Med Clin North Amer. 2016;100[6]:1199-215), and that was generally affirmed by the American College of Obstetricians and Gynecologists in 2013 and reaffirmed in 2018.*
The new evidence collected by Dr. Redman and associates “challenges current practice and argues that women with obesity should not be advised to consume additional energy during pregnancy as currently recommended,” they wrote in an article with their findings published a few days before Dr. Redman gave her talk (J Clin Invest. 2019;129[11]:4682-90).
The MomEE (Determinants of Gestational Weight Gain in Obese Pregnant Women) study enrolled 72 women with obesity during the first trimester of pregnancy and collected complete data through the end of the third trimester from 54 women. The researchers collected data on weight, body fat mass, and energy expenditure at multiple times during the second and third trimesters and calculated energy intake.
Based on body weights at the end of the third trimester, the researchers divided the 54 women into three subgroups: 10 women (19%) with inadequate weight gain by the IOM recommendations, 8 (15%) who had the IOM’s recommended weight gain of 11-20 pounds, and 36 women (67%; total is greater than 100% because of rounding) with excess weight gain, and within each group, they calculated the average level of energy intake relative to energy expenditure.
In addition to the daily calorie deficits associated with women who maintained recommended or inadequate weight, the researchers also found that women with excess weight gain averaged 186 more kcal/day than required to meet their daily energy expenditure.
The analyses showed that the increased energy demand of pregnancy and the fetus is compensated for by mobilization of the maternal fat mass in women with obesity, and that an imbalance between energy intake and expenditure is the main driver of weight gain during pregnancy. The results also highlighted how often pregnant women with obesity fail to follow a diet that results in the recommended weight gain of 11-20 pounds. In the MomEE cohort, two-thirds of enrolled women had excess weight gain.
The finding that women had the recommended weight gain on a diet that cut their daily calorie intake by about 100 kcal/day during the last two trimesters highlighted the nutritional challenge faced by women with obesity who are pregnant. “About three-quarters of women in the study had poor diet quality. There is an opportunity to improve diet with more fruits and vegetables to increase fullness, and [to reduce] energy-dense foods,” Dr. Redman said.
She is planning to collaborate on a study that will test the efficacy and safety of providing pregnant women with extreme obesity (class II-III) with defined meals to provide better control of energy intake and nutritional quality. Dr. Redman said she also hoped that the new findings she reported would be taken into account by the advisory committee assembled by the Department of Health & Human Services and the Department of Agriculture, which are currently preparing a revision of U.S. dietary guidelines for release in 2020.
The National Institutes of Health and the Clinical Research Cores at Pennington Biomedical Research Center funded the study. Dr. Redman had no disclosures.
SOURCE: Redman LM et al. Obesity Week 2019, Abstract T-OR-2079.
*This article was updated 2/7/2020.
REPORTING FROM OBESITY WEEK 2019