User login
Labor induction at 39 weeks reduced cesarean rate for low-risk, first-time mothers
Nulliparous women who were induced at 39 weeks had the same relative risk of adverse perinatal outcomes but a lower risk of a cesarean delivery, compared with women who received expectant management, results that researchers say contrast traditional recommendations for perinatal care, according to study from the New England Journal of Medicine.
“These findings contradict the conclusions of multiple observational studies that have suggested that labor induction is associated with an increased risk of adverse maternal and perinatal outcomes,” William A. Grobman, MD, the Arthur Hale Curtis, MD, Professor of Obstetrics and Gynecology at Northwestern University in Chicago, and his colleagues wrote. “These studies, however, compared women who underwent labor induction with those who had spontaneous labor, which is not a comparison that is useful to guide clinical decision making.”
Dr. Grobman and his colleagues evaluated the deliveries of 3,062 women who underwent labor induction between 39 weeks of gestation and 39 weeks and 4 days of gestation, and compared them with outcomes of 3,044 women who received expectant management until 40 weeks and 5 days of gestation. Women in both groups had a singleton fetus, no indication of early delivery, and did not plan on delivering by C-section. The participants were assessed again at about 38 weeks of gestation and randomly assigned to receive labor induction or expectant management as part of a multicenter randomized, controlled, parallel-group, unmasked trial in 41 maternal-fetal medicine departments in hospitals participating in the Eunice Kennedy Shriver National Institute of Child Health and Human Development network screened between March 2014, and August 2017.
Primary perinatal outcomes and components were defined as perinatal death, respiratory support, an Apgar score of 3 or less at 5 minutes, hypoxic-ischemic encephalopathy, seizure, infection, meconium aspiration syndrome, birth trauma, intracranial or subgaleal hemorrhage, or hypotension that requires vasopressor support. The principal secondary outcome was cesarean delivery, but other secondary outcomes included neonatal or intensive care, infection, postpartum hospital stay, and hypertension, among others.
Dr. Grobman and his colleagues found 132 (4.3%) of neonates in the induction group and 164 (5.4%) in the expectant-management group experienced a primary composite outcome (relative risk, 0.80; 95% confidence interval, 0.64-1.00; P = .049).
Regarding secondary outcomes, there was a significantly lower risk of cesarean delivery in the induction group, with 18.6% of women undergoing a cesarean delivery, compared with 22.2% of women in the expectant-management group (RR, 0.84; 95% CI, 0.76-0.93; P less than .001). Women in the labor induction group had a significantly lower relative risk of hypertensive disorders of pregnancy (9.1%), compared with the expectant-management (14.1%) group (RR, 0.64; 95% CI, 0.56-0.74; P less than .001). The investigators said women who underwent induced labor had lower 10-point Likert scale scores, were more likely to have “extensions of the uterine incision during cesarean delivery,” perceived they had “more control” during delivery, and had a shorter postpartum stay in the hospital, compared with women who received expectant management. However, women in the induced labor group also had a longer stay in the labor and delivery units, they said.
The researchers noted the limitations in this study, which included its unmasked design, lack of power to detect infrequent outcome differences, and the lack of information surrounding labor induction at 39 weeks in low-risk nulliparous women.
“These results suggest that policies aimed at the avoidance of elective labor induction among low-risk nulliparous women at 39 weeks of gestation are unlikely to reduce the rate of cesarean delivery on a population level; the trial provides information that can be incorporated into discussions that rely on principles of shared decision making,” Dr. Grobman and his colleagues wrote.
This study was funded by the Eunice Kennedy Shriver National Institute of Child Health and Human Development. Dr. Silver reports receiving personal fees from Gestavision. The other authors report no relevant financial disclosures.
SOURCE: Grobman WA et al. N Engl J Med. 2018 Aug 9. doi: 10.1056/NEJMoa1800566.
Of the more than 50,000 women screened for the study by Grobman et al., there were more than 44,000 women excluded and more than 16,000 did not participate in the trial. Further, the study participants tended to be younger and comprised more black or Hispanic women than the general population of mothers in the United States, Michael F. Greene, MD, said in a related editorial.
“Readers can only speculate as to why so many women declined to participate in the trial and what implications the demographics of the participants may have for the generalizability of the trial results and the acceptability of elective induction of labor at 39 weeks among women in the United States more generally,” Dr. Greene said. “If induction at 39 weeks becomes a widely popular option, busy obstetrical centers will need to find new ways to accommodate larger numbers of women with longer lengths of stay in the labor and delivery unit.”
Nevertheless, the study reflects a “public preference for a less interventionist approach” to delivery, Dr. Greene said, and the interest is backed by available data. He cited a meta-analysis of 20 randomized trials that found inducing labor at 39 weeks may reduce perinatal morality while not increasing the risk of operative deliveries. Specifically, he noted a randomized trial from the United Kingdom found induction of labor among 619 women at 39 weeks who were at least 35 years old did not affect the participants’ perception of delivery or increase the number of operative deliveries.
“These results across multiple obstetrical centers in the United States, however, should reassure women that elective induction of labor at 39 weeks is a reasonable choice that is very unlikely to result in poorer obstetrical outcomes,” he said.
Dr. Greene is chief of obstetrics and gynecology at Massachusetts General Hospital in Boston. He reported no relevant conflicts of interest. These comments summarize his editorial accompanying the article by Dr. Grobman and his associates ( N Engl J Med. 2018 Aug 9;379[6]:580-1 ).
Of the more than 50,000 women screened for the study by Grobman et al., there were more than 44,000 women excluded and more than 16,000 did not participate in the trial. Further, the study participants tended to be younger and comprised more black or Hispanic women than the general population of mothers in the United States, Michael F. Greene, MD, said in a related editorial.
“Readers can only speculate as to why so many women declined to participate in the trial and what implications the demographics of the participants may have for the generalizability of the trial results and the acceptability of elective induction of labor at 39 weeks among women in the United States more generally,” Dr. Greene said. “If induction at 39 weeks becomes a widely popular option, busy obstetrical centers will need to find new ways to accommodate larger numbers of women with longer lengths of stay in the labor and delivery unit.”
Nevertheless, the study reflects a “public preference for a less interventionist approach” to delivery, Dr. Greene said, and the interest is backed by available data. He cited a meta-analysis of 20 randomized trials that found inducing labor at 39 weeks may reduce perinatal morality while not increasing the risk of operative deliveries. Specifically, he noted a randomized trial from the United Kingdom found induction of labor among 619 women at 39 weeks who were at least 35 years old did not affect the participants’ perception of delivery or increase the number of operative deliveries.
“These results across multiple obstetrical centers in the United States, however, should reassure women that elective induction of labor at 39 weeks is a reasonable choice that is very unlikely to result in poorer obstetrical outcomes,” he said.
Dr. Greene is chief of obstetrics and gynecology at Massachusetts General Hospital in Boston. He reported no relevant conflicts of interest. These comments summarize his editorial accompanying the article by Dr. Grobman and his associates ( N Engl J Med. 2018 Aug 9;379[6]:580-1 ).
Of the more than 50,000 women screened for the study by Grobman et al., there were more than 44,000 women excluded and more than 16,000 did not participate in the trial. Further, the study participants tended to be younger and comprised more black or Hispanic women than the general population of mothers in the United States, Michael F. Greene, MD, said in a related editorial.
“Readers can only speculate as to why so many women declined to participate in the trial and what implications the demographics of the participants may have for the generalizability of the trial results and the acceptability of elective induction of labor at 39 weeks among women in the United States more generally,” Dr. Greene said. “If induction at 39 weeks becomes a widely popular option, busy obstetrical centers will need to find new ways to accommodate larger numbers of women with longer lengths of stay in the labor and delivery unit.”
Nevertheless, the study reflects a “public preference for a less interventionist approach” to delivery, Dr. Greene said, and the interest is backed by available data. He cited a meta-analysis of 20 randomized trials that found inducing labor at 39 weeks may reduce perinatal morality while not increasing the risk of operative deliveries. Specifically, he noted a randomized trial from the United Kingdom found induction of labor among 619 women at 39 weeks who were at least 35 years old did not affect the participants’ perception of delivery or increase the number of operative deliveries.
“These results across multiple obstetrical centers in the United States, however, should reassure women that elective induction of labor at 39 weeks is a reasonable choice that is very unlikely to result in poorer obstetrical outcomes,” he said.
Dr. Greene is chief of obstetrics and gynecology at Massachusetts General Hospital in Boston. He reported no relevant conflicts of interest. These comments summarize his editorial accompanying the article by Dr. Grobman and his associates ( N Engl J Med. 2018 Aug 9;379[6]:580-1 ).
Nulliparous women who were induced at 39 weeks had the same relative risk of adverse perinatal outcomes but a lower risk of a cesarean delivery, compared with women who received expectant management, results that researchers say contrast traditional recommendations for perinatal care, according to study from the New England Journal of Medicine.
“These findings contradict the conclusions of multiple observational studies that have suggested that labor induction is associated with an increased risk of adverse maternal and perinatal outcomes,” William A. Grobman, MD, the Arthur Hale Curtis, MD, Professor of Obstetrics and Gynecology at Northwestern University in Chicago, and his colleagues wrote. “These studies, however, compared women who underwent labor induction with those who had spontaneous labor, which is not a comparison that is useful to guide clinical decision making.”
Dr. Grobman and his colleagues evaluated the deliveries of 3,062 women who underwent labor induction between 39 weeks of gestation and 39 weeks and 4 days of gestation, and compared them with outcomes of 3,044 women who received expectant management until 40 weeks and 5 days of gestation. Women in both groups had a singleton fetus, no indication of early delivery, and did not plan on delivering by C-section. The participants were assessed again at about 38 weeks of gestation and randomly assigned to receive labor induction or expectant management as part of a multicenter randomized, controlled, parallel-group, unmasked trial in 41 maternal-fetal medicine departments in hospitals participating in the Eunice Kennedy Shriver National Institute of Child Health and Human Development network screened between March 2014, and August 2017.
Primary perinatal outcomes and components were defined as perinatal death, respiratory support, an Apgar score of 3 or less at 5 minutes, hypoxic-ischemic encephalopathy, seizure, infection, meconium aspiration syndrome, birth trauma, intracranial or subgaleal hemorrhage, or hypotension that requires vasopressor support. The principal secondary outcome was cesarean delivery, but other secondary outcomes included neonatal or intensive care, infection, postpartum hospital stay, and hypertension, among others.
Dr. Grobman and his colleagues found 132 (4.3%) of neonates in the induction group and 164 (5.4%) in the expectant-management group experienced a primary composite outcome (relative risk, 0.80; 95% confidence interval, 0.64-1.00; P = .049).
Regarding secondary outcomes, there was a significantly lower risk of cesarean delivery in the induction group, with 18.6% of women undergoing a cesarean delivery, compared with 22.2% of women in the expectant-management group (RR, 0.84; 95% CI, 0.76-0.93; P less than .001). Women in the labor induction group had a significantly lower relative risk of hypertensive disorders of pregnancy (9.1%), compared with the expectant-management (14.1%) group (RR, 0.64; 95% CI, 0.56-0.74; P less than .001). The investigators said women who underwent induced labor had lower 10-point Likert scale scores, were more likely to have “extensions of the uterine incision during cesarean delivery,” perceived they had “more control” during delivery, and had a shorter postpartum stay in the hospital, compared with women who received expectant management. However, women in the induced labor group also had a longer stay in the labor and delivery units, they said.
The researchers noted the limitations in this study, which included its unmasked design, lack of power to detect infrequent outcome differences, and the lack of information surrounding labor induction at 39 weeks in low-risk nulliparous women.
“These results suggest that policies aimed at the avoidance of elective labor induction among low-risk nulliparous women at 39 weeks of gestation are unlikely to reduce the rate of cesarean delivery on a population level; the trial provides information that can be incorporated into discussions that rely on principles of shared decision making,” Dr. Grobman and his colleagues wrote.
This study was funded by the Eunice Kennedy Shriver National Institute of Child Health and Human Development. Dr. Silver reports receiving personal fees from Gestavision. The other authors report no relevant financial disclosures.
SOURCE: Grobman WA et al. N Engl J Med. 2018 Aug 9. doi: 10.1056/NEJMoa1800566.
Nulliparous women who were induced at 39 weeks had the same relative risk of adverse perinatal outcomes but a lower risk of a cesarean delivery, compared with women who received expectant management, results that researchers say contrast traditional recommendations for perinatal care, according to study from the New England Journal of Medicine.
“These findings contradict the conclusions of multiple observational studies that have suggested that labor induction is associated with an increased risk of adverse maternal and perinatal outcomes,” William A. Grobman, MD, the Arthur Hale Curtis, MD, Professor of Obstetrics and Gynecology at Northwestern University in Chicago, and his colleagues wrote. “These studies, however, compared women who underwent labor induction with those who had spontaneous labor, which is not a comparison that is useful to guide clinical decision making.”
Dr. Grobman and his colleagues evaluated the deliveries of 3,062 women who underwent labor induction between 39 weeks of gestation and 39 weeks and 4 days of gestation, and compared them with outcomes of 3,044 women who received expectant management until 40 weeks and 5 days of gestation. Women in both groups had a singleton fetus, no indication of early delivery, and did not plan on delivering by C-section. The participants were assessed again at about 38 weeks of gestation and randomly assigned to receive labor induction or expectant management as part of a multicenter randomized, controlled, parallel-group, unmasked trial in 41 maternal-fetal medicine departments in hospitals participating in the Eunice Kennedy Shriver National Institute of Child Health and Human Development network screened between March 2014, and August 2017.
Primary perinatal outcomes and components were defined as perinatal death, respiratory support, an Apgar score of 3 or less at 5 minutes, hypoxic-ischemic encephalopathy, seizure, infection, meconium aspiration syndrome, birth trauma, intracranial or subgaleal hemorrhage, or hypotension that requires vasopressor support. The principal secondary outcome was cesarean delivery, but other secondary outcomes included neonatal or intensive care, infection, postpartum hospital stay, and hypertension, among others.
Dr. Grobman and his colleagues found 132 (4.3%) of neonates in the induction group and 164 (5.4%) in the expectant-management group experienced a primary composite outcome (relative risk, 0.80; 95% confidence interval, 0.64-1.00; P = .049).
Regarding secondary outcomes, there was a significantly lower risk of cesarean delivery in the induction group, with 18.6% of women undergoing a cesarean delivery, compared with 22.2% of women in the expectant-management group (RR, 0.84; 95% CI, 0.76-0.93; P less than .001). Women in the labor induction group had a significantly lower relative risk of hypertensive disorders of pregnancy (9.1%), compared with the expectant-management (14.1%) group (RR, 0.64; 95% CI, 0.56-0.74; P less than .001). The investigators said women who underwent induced labor had lower 10-point Likert scale scores, were more likely to have “extensions of the uterine incision during cesarean delivery,” perceived they had “more control” during delivery, and had a shorter postpartum stay in the hospital, compared with women who received expectant management. However, women in the induced labor group also had a longer stay in the labor and delivery units, they said.
The researchers noted the limitations in this study, which included its unmasked design, lack of power to detect infrequent outcome differences, and the lack of information surrounding labor induction at 39 weeks in low-risk nulliparous women.
“These results suggest that policies aimed at the avoidance of elective labor induction among low-risk nulliparous women at 39 weeks of gestation are unlikely to reduce the rate of cesarean delivery on a population level; the trial provides information that can be incorporated into discussions that rely on principles of shared decision making,” Dr. Grobman and his colleagues wrote.
This study was funded by the Eunice Kennedy Shriver National Institute of Child Health and Human Development. Dr. Silver reports receiving personal fees from Gestavision. The other authors report no relevant financial disclosures.
SOURCE: Grobman WA et al. N Engl J Med. 2018 Aug 9. doi: 10.1056/NEJMoa1800566.
FROM NEW ENGLAND JOURNAL OF MEDICINE
Key clinical point:
Major finding: 18.6% of women in the induced labor group underwent cesarean delivery, compared with 22.2% in the expectant management group.
Study details: A multicenter randomized, controlled, parallel-group, unmasked trial of 6,106 women from 41 maternal-fetal medicine departments in hospitals participating in the Eunice Kennedy Shriver National Institute of Child Health and Human Development network screened between March 2014 and August 2017.
Disclosures: This study was funded by the Eunice Kennedy Shriver National Institute of Child Health and Human Development. Dr. Silver reports receiving personal fees from Gestavision. The other authors report no relevant financial disclosures..
Source: Grobman WA et al. N Engl J Med. 2018 Aug 9. doi: 10.1056/NEJMoa1800566.
Maternal obesity plus diabetes lead to psychiatric disorders in offspring
Women who were obese and had diabetes before becoming pregnant were sixfold more likely to have children with psychiatric and neurodevelopmental disorders by age 11 years, as compared to women with normal body mass indexes (BMIs), based on results of a large, prospective, population-based, cohort study published in Pediatrics.
The risks to offspring whose mothers were obese and had pregestational diabetes mellitus (PGDM) were far greater than the risks seen when mothers had either condition alone or had gestational diabetes mellitus (GDM) in the study, reported Linghua Kong of the Karolinska Institute, Stockholm, and colleagues. The study is based on data from various national registries in Finland regarding 649,043 live births during 2004-2014 and data regarding psychiatric diagnoses from the Finnish Care Registers for Health Care.
Of the children in the cohort, 7.67% had mothers who were obese and 3.66% had mothers who were severely obese based on standard World Health Organization criteria; mothers had PGDM in 0.62% of the births and GDM in 15.7% of the births.
Overall, 5.4% of the children were diagnosed with a psychiatric disorder by age 11 years.
Compared with children born to mothers of normal weight (BMI less than 25 kg/m2), those born to mothers with severe maternal obesity alone (BMI greater than 35) had higher rates of developmental disorders or speech, language, motor, and scholastic skills (hazard ratio, 1.69; 95% confidence interval 1.54-1.86); ADHD and/or conduct disorder (HR, 1.88; 95% CI, 1.58-2.23); and psychosis and mood and anxiety disorders (HR, 1.67; 95% CI, 1.31-2.13). Increased risk of psychiatric disorders were only slightly statistically significant in the offspring of women with severe obesity and GDM.
The risks were significantly elevated, however, for children born to obese women who also had PGDM. The hazard ratio for autism spectrum disorder was 6.49 (95% CI, 3.08-13.69), and the HR for ADHD and/or conduct disorder was 6.03 (95% CI, 3.23-11.24). The risks were fourfold higher for mixed disorders of emotions and conduct, disorders of social function, and tics (HR, 4.29; 95% CI, 2.14-8.60).
Limitations of the study included basing results on shorter follow-up times for those born later in the study period, grouping of offspring’s disorder diagnoses, basing the definition of PGDM on insulin prescription, and using BMI measurements taken at only one time point during pregnancy.
The researchers were supported by the National Institute for Health and Welfare: Drugs and Pregnancy project, the Swedish Research Council, the regional agreement on medical training and clinical research between Stockholm County Council and Karolinska Institutet Stockholm County Council, the China Scholarship Council, and the Swedish Brain Foundation.
SOURCE: Kong L et al. Pediatrics. 2018 Sep;142(3):1-11.
Women who were obese and had diabetes before becoming pregnant were sixfold more likely to have children with psychiatric and neurodevelopmental disorders by age 11 years, as compared to women with normal body mass indexes (BMIs), based on results of a large, prospective, population-based, cohort study published in Pediatrics.
The risks to offspring whose mothers were obese and had pregestational diabetes mellitus (PGDM) were far greater than the risks seen when mothers had either condition alone or had gestational diabetes mellitus (GDM) in the study, reported Linghua Kong of the Karolinska Institute, Stockholm, and colleagues. The study is based on data from various national registries in Finland regarding 649,043 live births during 2004-2014 and data regarding psychiatric diagnoses from the Finnish Care Registers for Health Care.
Of the children in the cohort, 7.67% had mothers who were obese and 3.66% had mothers who were severely obese based on standard World Health Organization criteria; mothers had PGDM in 0.62% of the births and GDM in 15.7% of the births.
Overall, 5.4% of the children were diagnosed with a psychiatric disorder by age 11 years.
Compared with children born to mothers of normal weight (BMI less than 25 kg/m2), those born to mothers with severe maternal obesity alone (BMI greater than 35) had higher rates of developmental disorders or speech, language, motor, and scholastic skills (hazard ratio, 1.69; 95% confidence interval 1.54-1.86); ADHD and/or conduct disorder (HR, 1.88; 95% CI, 1.58-2.23); and psychosis and mood and anxiety disorders (HR, 1.67; 95% CI, 1.31-2.13). Increased risk of psychiatric disorders were only slightly statistically significant in the offspring of women with severe obesity and GDM.
The risks were significantly elevated, however, for children born to obese women who also had PGDM. The hazard ratio for autism spectrum disorder was 6.49 (95% CI, 3.08-13.69), and the HR for ADHD and/or conduct disorder was 6.03 (95% CI, 3.23-11.24). The risks were fourfold higher for mixed disorders of emotions and conduct, disorders of social function, and tics (HR, 4.29; 95% CI, 2.14-8.60).
Limitations of the study included basing results on shorter follow-up times for those born later in the study period, grouping of offspring’s disorder diagnoses, basing the definition of PGDM on insulin prescription, and using BMI measurements taken at only one time point during pregnancy.
The researchers were supported by the National Institute for Health and Welfare: Drugs and Pregnancy project, the Swedish Research Council, the regional agreement on medical training and clinical research between Stockholm County Council and Karolinska Institutet Stockholm County Council, the China Scholarship Council, and the Swedish Brain Foundation.
SOURCE: Kong L et al. Pediatrics. 2018 Sep;142(3):1-11.
Women who were obese and had diabetes before becoming pregnant were sixfold more likely to have children with psychiatric and neurodevelopmental disorders by age 11 years, as compared to women with normal body mass indexes (BMIs), based on results of a large, prospective, population-based, cohort study published in Pediatrics.
The risks to offspring whose mothers were obese and had pregestational diabetes mellitus (PGDM) were far greater than the risks seen when mothers had either condition alone or had gestational diabetes mellitus (GDM) in the study, reported Linghua Kong of the Karolinska Institute, Stockholm, and colleagues. The study is based on data from various national registries in Finland regarding 649,043 live births during 2004-2014 and data regarding psychiatric diagnoses from the Finnish Care Registers for Health Care.
Of the children in the cohort, 7.67% had mothers who were obese and 3.66% had mothers who were severely obese based on standard World Health Organization criteria; mothers had PGDM in 0.62% of the births and GDM in 15.7% of the births.
Overall, 5.4% of the children were diagnosed with a psychiatric disorder by age 11 years.
Compared with children born to mothers of normal weight (BMI less than 25 kg/m2), those born to mothers with severe maternal obesity alone (BMI greater than 35) had higher rates of developmental disorders or speech, language, motor, and scholastic skills (hazard ratio, 1.69; 95% confidence interval 1.54-1.86); ADHD and/or conduct disorder (HR, 1.88; 95% CI, 1.58-2.23); and psychosis and mood and anxiety disorders (HR, 1.67; 95% CI, 1.31-2.13). Increased risk of psychiatric disorders were only slightly statistically significant in the offspring of women with severe obesity and GDM.
The risks were significantly elevated, however, for children born to obese women who also had PGDM. The hazard ratio for autism spectrum disorder was 6.49 (95% CI, 3.08-13.69), and the HR for ADHD and/or conduct disorder was 6.03 (95% CI, 3.23-11.24). The risks were fourfold higher for mixed disorders of emotions and conduct, disorders of social function, and tics (HR, 4.29; 95% CI, 2.14-8.60).
Limitations of the study included basing results on shorter follow-up times for those born later in the study period, grouping of offspring’s disorder diagnoses, basing the definition of PGDM on insulin prescription, and using BMI measurements taken at only one time point during pregnancy.
The researchers were supported by the National Institute for Health and Welfare: Drugs and Pregnancy project, the Swedish Research Council, the regional agreement on medical training and clinical research between Stockholm County Council and Karolinska Institutet Stockholm County Council, the China Scholarship Council, and the Swedish Brain Foundation.
SOURCE: Kong L et al. Pediatrics. 2018 Sep;142(3):1-11.
FROM PEDIATRICS
1 in 7 Zika-exposed babies have at least one health problem related to the virus
About 14% of 1-year-olds with prenatal Zika virus exposure show at least one health problem probably related to the virus, according to a study published in Morbidity and Mortality Weekly Report.
Many of the problems are brain and eye abnormalities, which occurred at 30 times the 0.16% background rate among unexposed babies, Margaret Honein, PhD, and colleagues reported.
In a press briefing, Dr. Honein, chief of the Birth Defects Branch at the National Center on Birth Defects and Developmental Disabilities, described findings from the U.S. Zika Pregnancy and Infant Registry (USZPIR).
“Today’s report is the largest to date with long-term outcomes of babies born to mothers with [lab-confirmed] Zika infections, and the first published data on children 1 year or older from the ongoing surveillance network,” Dr. Honein said. It “clearly shows that the Zika story is not over, especially for the children and families who are affected by it.”
USZPIR is monitoring the outcomes of 7,300 pregnancies with lab-confirmed Zika infection. From these, 4,800 babies were born in the U.S. territories and freely associated states, had reached the age of 1 year by Feb. 1, 2018, and were included in the study.
In addition to clinical outcomes, the investigators looked at how many babies received the recommended evaluations, including neuroimaging, hearing screens, opthalmologic exams, developmental screening, and physical exams.
Almost all (95%) had at least one exam in the first 2 weeks of life; 76% had at least one developmental screening; 60% had postnatal neuroimaging; 48% at least one hearing exam; and 36% at least one eye exam by a specialist, the investigators found.
Findings that many didn’t get all the recommended health screenings are concerning, CDC Director Robert Redfield, MD. said during the briefing. “We are still learning about the full range of long-term health problems these babies could face. We thank clinicians for their continued commitment to conduct all necessary tests and evaluations to ensure appropriate care.”
Zika-associated birth defects occurred 203 babies (14%). Another 136 (9%) had at least one neurodevelopmental abnormality possibly associated with congenital Zika virus infection, and 20 (1%) had both. Most babies (1,386; 96%) did not have microcephaly detected at birth. But there was some “misclassification” of the condition, the investigators found. “Five infants had microcephaly at birth with brain or eye anomalies identified at birth; 59 had microcephaly at birth with no brain or eye anomalies identified at birth; and 20 infants did not have microcephaly identified at birth but had postnatal identification of microcephaly.”
Neurodevelopmental abnormalities possibly associated with Zika occurred in 136 (9%) of the cohort; 116 (8%) had no Zika-associated birth defects. Among these, half (58) had only possible developmental delay.
Zika transmission appears to be slowing, Lyle Peterson, MD, said during the press briefing, with no cases in the continental U.S. since 2017. That year, there were two cases in Florida and five in Texas. However, it is now endemic in many regions. Everyone should continue to take precautions against mosquito bites, he urged.
The MMWR also included updated guidance for men who are planning a pregnancy with a partner and may have been exposed to Zika.
CDC now recommends that these men wait at least 3 months after onset of Zika symptoms or any possible exposure, including travel to or living in a risk area. Past guidance recommended a 6-month waiting period. The new recommendation reflects emerging data suggesting that the risk of infectious Zika in semen declines during the 3 months after symptom onset.
Men who want to avoid passing Zika through sex should abstain for 3 months, or use a condom every time they have sex, the new recommendation said.
“All other Zika guidance remains unchanged,” the guidelines note. “Men with possible Zika virus exposure whose partner is pregnant should use condoms or the couple should not have sex for the entire pregnancy to reduce the risk of transmission.”
SOURCE: Honein, MA et al. MMWR 2018; 67: 1-10.
About 14% of 1-year-olds with prenatal Zika virus exposure show at least one health problem probably related to the virus, according to a study published in Morbidity and Mortality Weekly Report.
Many of the problems are brain and eye abnormalities, which occurred at 30 times the 0.16% background rate among unexposed babies, Margaret Honein, PhD, and colleagues reported.
In a press briefing, Dr. Honein, chief of the Birth Defects Branch at the National Center on Birth Defects and Developmental Disabilities, described findings from the U.S. Zika Pregnancy and Infant Registry (USZPIR).
“Today’s report is the largest to date with long-term outcomes of babies born to mothers with [lab-confirmed] Zika infections, and the first published data on children 1 year or older from the ongoing surveillance network,” Dr. Honein said. It “clearly shows that the Zika story is not over, especially for the children and families who are affected by it.”
USZPIR is monitoring the outcomes of 7,300 pregnancies with lab-confirmed Zika infection. From these, 4,800 babies were born in the U.S. territories and freely associated states, had reached the age of 1 year by Feb. 1, 2018, and were included in the study.
In addition to clinical outcomes, the investigators looked at how many babies received the recommended evaluations, including neuroimaging, hearing screens, opthalmologic exams, developmental screening, and physical exams.
Almost all (95%) had at least one exam in the first 2 weeks of life; 76% had at least one developmental screening; 60% had postnatal neuroimaging; 48% at least one hearing exam; and 36% at least one eye exam by a specialist, the investigators found.
Findings that many didn’t get all the recommended health screenings are concerning, CDC Director Robert Redfield, MD. said during the briefing. “We are still learning about the full range of long-term health problems these babies could face. We thank clinicians for their continued commitment to conduct all necessary tests and evaluations to ensure appropriate care.”
Zika-associated birth defects occurred 203 babies (14%). Another 136 (9%) had at least one neurodevelopmental abnormality possibly associated with congenital Zika virus infection, and 20 (1%) had both. Most babies (1,386; 96%) did not have microcephaly detected at birth. But there was some “misclassification” of the condition, the investigators found. “Five infants had microcephaly at birth with brain or eye anomalies identified at birth; 59 had microcephaly at birth with no brain or eye anomalies identified at birth; and 20 infants did not have microcephaly identified at birth but had postnatal identification of microcephaly.”
Neurodevelopmental abnormalities possibly associated with Zika occurred in 136 (9%) of the cohort; 116 (8%) had no Zika-associated birth defects. Among these, half (58) had only possible developmental delay.
Zika transmission appears to be slowing, Lyle Peterson, MD, said during the press briefing, with no cases in the continental U.S. since 2017. That year, there were two cases in Florida and five in Texas. However, it is now endemic in many regions. Everyone should continue to take precautions against mosquito bites, he urged.
The MMWR also included updated guidance for men who are planning a pregnancy with a partner and may have been exposed to Zika.
CDC now recommends that these men wait at least 3 months after onset of Zika symptoms or any possible exposure, including travel to or living in a risk area. Past guidance recommended a 6-month waiting period. The new recommendation reflects emerging data suggesting that the risk of infectious Zika in semen declines during the 3 months after symptom onset.
Men who want to avoid passing Zika through sex should abstain for 3 months, or use a condom every time they have sex, the new recommendation said.
“All other Zika guidance remains unchanged,” the guidelines note. “Men with possible Zika virus exposure whose partner is pregnant should use condoms or the couple should not have sex for the entire pregnancy to reduce the risk of transmission.”
SOURCE: Honein, MA et al. MMWR 2018; 67: 1-10.
About 14% of 1-year-olds with prenatal Zika virus exposure show at least one health problem probably related to the virus, according to a study published in Morbidity and Mortality Weekly Report.
Many of the problems are brain and eye abnormalities, which occurred at 30 times the 0.16% background rate among unexposed babies, Margaret Honein, PhD, and colleagues reported.
In a press briefing, Dr. Honein, chief of the Birth Defects Branch at the National Center on Birth Defects and Developmental Disabilities, described findings from the U.S. Zika Pregnancy and Infant Registry (USZPIR).
“Today’s report is the largest to date with long-term outcomes of babies born to mothers with [lab-confirmed] Zika infections, and the first published data on children 1 year or older from the ongoing surveillance network,” Dr. Honein said. It “clearly shows that the Zika story is not over, especially for the children and families who are affected by it.”
USZPIR is monitoring the outcomes of 7,300 pregnancies with lab-confirmed Zika infection. From these, 4,800 babies were born in the U.S. territories and freely associated states, had reached the age of 1 year by Feb. 1, 2018, and were included in the study.
In addition to clinical outcomes, the investigators looked at how many babies received the recommended evaluations, including neuroimaging, hearing screens, opthalmologic exams, developmental screening, and physical exams.
Almost all (95%) had at least one exam in the first 2 weeks of life; 76% had at least one developmental screening; 60% had postnatal neuroimaging; 48% at least one hearing exam; and 36% at least one eye exam by a specialist, the investigators found.
Findings that many didn’t get all the recommended health screenings are concerning, CDC Director Robert Redfield, MD. said during the briefing. “We are still learning about the full range of long-term health problems these babies could face. We thank clinicians for their continued commitment to conduct all necessary tests and evaluations to ensure appropriate care.”
Zika-associated birth defects occurred 203 babies (14%). Another 136 (9%) had at least one neurodevelopmental abnormality possibly associated with congenital Zika virus infection, and 20 (1%) had both. Most babies (1,386; 96%) did not have microcephaly detected at birth. But there was some “misclassification” of the condition, the investigators found. “Five infants had microcephaly at birth with brain or eye anomalies identified at birth; 59 had microcephaly at birth with no brain or eye anomalies identified at birth; and 20 infants did not have microcephaly identified at birth but had postnatal identification of microcephaly.”
Neurodevelopmental abnormalities possibly associated with Zika occurred in 136 (9%) of the cohort; 116 (8%) had no Zika-associated birth defects. Among these, half (58) had only possible developmental delay.
Zika transmission appears to be slowing, Lyle Peterson, MD, said during the press briefing, with no cases in the continental U.S. since 2017. That year, there were two cases in Florida and five in Texas. However, it is now endemic in many regions. Everyone should continue to take precautions against mosquito bites, he urged.
The MMWR also included updated guidance for men who are planning a pregnancy with a partner and may have been exposed to Zika.
CDC now recommends that these men wait at least 3 months after onset of Zika symptoms or any possible exposure, including travel to or living in a risk area. Past guidance recommended a 6-month waiting period. The new recommendation reflects emerging data suggesting that the risk of infectious Zika in semen declines during the 3 months after symptom onset.
Men who want to avoid passing Zika through sex should abstain for 3 months, or use a condom every time they have sex, the new recommendation said.
“All other Zika guidance remains unchanged,” the guidelines note. “Men with possible Zika virus exposure whose partner is pregnant should use condoms or the couple should not have sex for the entire pregnancy to reduce the risk of transmission.”
SOURCE: Honein, MA et al. MMWR 2018; 67: 1-10.
FROM MMWR
Key clinical point: Zika-related health problems are present in a substantial number of prenatally exposed babies.
Major finding: Problems occurred in 14% of 4,800 included in a national registry.
Study details: USZPIR is monitoring the outcomes of 7,300 pregnancies with lab-confirmed Zika infection. Disclosures: No relevant conflicts of interest were disclosed.
Source: Honein, MA et al. MMWR 2018; 67: 1-10.
FDA warning shines light on vaginal rejuvenation
The Food and Drug Administration has issued a stern warning to several manufacturers and a statement of caution to the public concerning “vaginal rejuvenation,” an umbrella term for a host of procedures to alter vaginal tissue for therapeutic or cosmetic purposes.
Lasers and other energy-based devices have been approved to treat abnormal or precancerous cervical or vaginal tissue and general warts, but the FDA has not approved any to treat vaginal atrophy, urinary incontinence, or reduced sexual function.
Device manufacturers claim lasers can address these conditions despite limited scientific evidence for their safety or efficacy. Insurers do not reimburse the procedures, considering them to be cosmetic.
In a July 30 statement, FDA Commissioner Scott Gottlieb, MD, slammed “deceptive” marketing practices on the part of manufacturers.
The FDA has reviewed 12 complaints since December 2015 of adverse effects related to vaginal procedures using the devices. Two were from manufacturers reporting pain and bleeding in patients following treatment, FDA spokeswoman Deborah Kotz said in an interview. “The FDA has also received voluntary MedWatch reports from individual patients who experienced significant pain and discomfort from procedures performed with these devices.”
The agency has targeted seven firms: Alma Lasers, BTL Aesthetics, BTL Industries, Cynosure, InMode, Sciton, and ThermiGen, with letters demanding evidence of FDA approval, clearance, or intent to seek clearance for use of their products on female genitalia. They also asked for evidence backing specific claims.
In a July 26 letter to BTL Industries, for example, the FDA demanded to know why the firm was marketing its Exilis laser device, approved for the treatment of facial wrinkles, as “Ultra Femme 360,” which it called “a whole new approach to women’s intimate health.” The device, according to the manufacturer, “provides the shortest noninvasive radio-frequency treatment available for female intimate parts” and “is proven to increase elastin and collagen in the treatment area.”
The FDA asked Cynosure, the maker of the Mona Lisa Touch, a system marketed as an FDA-approved treatment for vaginal atrophy, for evidence to support its claims that Mona Lisa Touch “is the only technology for vaginal and vulvar health with over 18+ published clinical studies” and is clinically proven to treat “painful symptoms of menopause, including intimacy.” They also asked for information about a modification to the originally approved device that was not brought to the FDA’s attention.
In a letter to Alma Lasers, whose Pixel CO2 Laser System was approved for use in a broad use of surgical applications including gynecologic surgery, the FDA noted that the device was being marketed as “FEMILIFT,” a laser procedure designed to “improve vaginal irregularities” and to assist “in vaginal mucosa revitalization.” The FDA demanded evidence for those claims.
The manufacturers have 30 days to respond to the FDA, which has not ruled out seeking enforcement action against firms with unsatisfactory responses.
For more than a decade, researchers have shown that healthy vaginal morphology is exceptionally wide ranging, including a recent study in more than 650 women, the largest to date (BJOG. 2018 Jun 25. doi: 10.1111/1471-0528.15387). Nonetheless, interest in elective vaginal procedures has only increased, with an industry report from the International Society of Aesthetic and Plastic Surgery describing a 45% increase in the use of one surgery, vaginal labiaplasty, between 2014 and 2016. Most procedures were performed in Brazil and the United States.
While plastic surgery societies support vaginal rejuvenation procedures, the American College of Obstetricians and Gynecologists has long frowned on them, with its first critique issued in a 2007 committee opinion. “No adequate studies have been published assessing the long-term satisfaction, safety, and complication rates for these procedures,” the association said last year in its most recent update on the subject.
Gynecologist David M. Jaspan, DO, of the Einstein Healthcare Network in Philadelphia, echoed ACOG’s views and said he welcomed FDA interest in vaginal rejuvenation.
The practice has “never been endorsed by the College or a board. It’s been considered a cosmetic procedure and it’s been under scrutiny for at least a decade,” Dr. Jaspan said. “I have reservations about the clinical outcomes and the training surrounds these procedures and I anxiously await randomized controlled trials to further evaluate them.”
Gynecologists who offer the procedures caution that they may have a role, and that randomized trials are underway to determine which groups of women might be best helped.
Marie Paraiso, MD, professor of obstetrics and gynecology at the Cleveland Clinic, said she uses the Mona Lisa Touch, a CO2 fractionated laser, to treat patients with genitourinary syndrome of menopause (GSM). These patients, Dr. Paraiso said, “complain of vaginal dryness and are unable to have intercourse or experience significant pain during or after intercourse. Some of them also may have irritative voiding, urinary frequency and urgency, or mild stress incontinence.”
Dr. Paraiso’s group has performed some 300 treatments with the laser and “we have fortunately not had patients complaining of persistent vaginal pain or scarring.” About 80%-90% of patients respond, she said, with some 20%-25% seeking retreatment within a year. “I believe for women who have contraindications to hormonal therapy or do not tolerate or cannot afford prolonged hormonal therapy, the CO2 fractional vaginal laser has been effective.”
Dr. Paraiso is also leading a multisite clinical trial randomizing about 200 patients with GSM to the laser treatment or estrogen-based vaginal creams, and following them for 6 months; thus far, she said, 6 of 89 patients, half in the laser arm, have reported mild to moderate adverse events.
Dr. Paraiso said she does not have a financial relationship with the manufacturer of Mona Lisa Touch, and that the trial was funded by the Foundation for Female Health Awareness, which receives unrestricted research grants from some device makers. “Our institute owns the laser and I have never been paid to train anyone to perform these procedures,” Dr. Paraiso added. “Our onus was to study the laser in order to improve the lives of our patients.”
Other trials comparing vaginal lasers with sham treatment are currently underway or in planning.
The North American Menopause Society struck a cautious note in response to the FDA criticism. In a statement issued August 1, JoAnn Pinkerton, MD, the society’s executive director, said the field needed prospective, randomized, sham-controlled trials of the laser and energy therapies. The therapies “may turn out to be an appropriate choice for many women, particularly for those concerned about breast cancer risk” associated with hormonal treatments. But until more robust data are available, doctors should “discuss the benefits and risks of all available treatment options for vaginal symptoms, including over-the-counter lubricants, vaginal moisturizers, and FDA-approved vaginal therapies such as vaginal estrogen and intravaginal dehydroepiandrosterone and oral therapies such as hormone therapy and ospemifene to determine the best treatment for women with GSM.”
Any discussion of vaginal energy-based therapies, should include the disclosure that these have not been approved for the specific indication, Dr. Pinkerton cautioned.
The term “vaginal rejuvenation,” coined by cosmetic gynecologists, incorporates surgeries designed to modify the appearance of the vulva, reduce the redundancy of vaginal tissue, and improve vaginal tone.
Endorsed by some well-known academic gynecologists, these devices have been promoted as “safe and effective” without any prospective, randomized studies and without accountability for conflicts of interest including “educational stipends” from device manufacturers and clinicians’ need to recoup the high cost of the devices themselves.
Studies have generally been limited to fewer than 100 patients followed for 12 weeks or less. Companies are not informing doctors that the devices may not be FDA approved for the purposes advertised, nor are they providing adverse effects reports. Laser and radio-frequency procedures at best demonstrate temporary, marginal improvement in vaginal tone and dyspareunia, and at worst are associated with increased pelvic pain and dyspareunia, as well as vaginal, rectal, and bladder thermal burns. For those of us who specialize in cosmetic surgery, they have very limited benefit with a significant risk of injury to the patient even when properly used.
Julio Cesar Novoa, MD, is a private practice ob.gyn from El Paso, Tex. He reported no relevant conflicts of interest.
The term “vaginal rejuvenation,” coined by cosmetic gynecologists, incorporates surgeries designed to modify the appearance of the vulva, reduce the redundancy of vaginal tissue, and improve vaginal tone.
Endorsed by some well-known academic gynecologists, these devices have been promoted as “safe and effective” without any prospective, randomized studies and without accountability for conflicts of interest including “educational stipends” from device manufacturers and clinicians’ need to recoup the high cost of the devices themselves.
Studies have generally been limited to fewer than 100 patients followed for 12 weeks or less. Companies are not informing doctors that the devices may not be FDA approved for the purposes advertised, nor are they providing adverse effects reports. Laser and radio-frequency procedures at best demonstrate temporary, marginal improvement in vaginal tone and dyspareunia, and at worst are associated with increased pelvic pain and dyspareunia, as well as vaginal, rectal, and bladder thermal burns. For those of us who specialize in cosmetic surgery, they have very limited benefit with a significant risk of injury to the patient even when properly used.
Julio Cesar Novoa, MD, is a private practice ob.gyn from El Paso, Tex. He reported no relevant conflicts of interest.
The term “vaginal rejuvenation,” coined by cosmetic gynecologists, incorporates surgeries designed to modify the appearance of the vulva, reduce the redundancy of vaginal tissue, and improve vaginal tone.
Endorsed by some well-known academic gynecologists, these devices have been promoted as “safe and effective” without any prospective, randomized studies and without accountability for conflicts of interest including “educational stipends” from device manufacturers and clinicians’ need to recoup the high cost of the devices themselves.
Studies have generally been limited to fewer than 100 patients followed for 12 weeks or less. Companies are not informing doctors that the devices may not be FDA approved for the purposes advertised, nor are they providing adverse effects reports. Laser and radio-frequency procedures at best demonstrate temporary, marginal improvement in vaginal tone and dyspareunia, and at worst are associated with increased pelvic pain and dyspareunia, as well as vaginal, rectal, and bladder thermal burns. For those of us who specialize in cosmetic surgery, they have very limited benefit with a significant risk of injury to the patient even when properly used.
Julio Cesar Novoa, MD, is a private practice ob.gyn from El Paso, Tex. He reported no relevant conflicts of interest.
The Food and Drug Administration has issued a stern warning to several manufacturers and a statement of caution to the public concerning “vaginal rejuvenation,” an umbrella term for a host of procedures to alter vaginal tissue for therapeutic or cosmetic purposes.
Lasers and other energy-based devices have been approved to treat abnormal or precancerous cervical or vaginal tissue and general warts, but the FDA has not approved any to treat vaginal atrophy, urinary incontinence, or reduced sexual function.
Device manufacturers claim lasers can address these conditions despite limited scientific evidence for their safety or efficacy. Insurers do not reimburse the procedures, considering them to be cosmetic.
In a July 30 statement, FDA Commissioner Scott Gottlieb, MD, slammed “deceptive” marketing practices on the part of manufacturers.
The FDA has reviewed 12 complaints since December 2015 of adverse effects related to vaginal procedures using the devices. Two were from manufacturers reporting pain and bleeding in patients following treatment, FDA spokeswoman Deborah Kotz said in an interview. “The FDA has also received voluntary MedWatch reports from individual patients who experienced significant pain and discomfort from procedures performed with these devices.”
The agency has targeted seven firms: Alma Lasers, BTL Aesthetics, BTL Industries, Cynosure, InMode, Sciton, and ThermiGen, with letters demanding evidence of FDA approval, clearance, or intent to seek clearance for use of their products on female genitalia. They also asked for evidence backing specific claims.
In a July 26 letter to BTL Industries, for example, the FDA demanded to know why the firm was marketing its Exilis laser device, approved for the treatment of facial wrinkles, as “Ultra Femme 360,” which it called “a whole new approach to women’s intimate health.” The device, according to the manufacturer, “provides the shortest noninvasive radio-frequency treatment available for female intimate parts” and “is proven to increase elastin and collagen in the treatment area.”
The FDA asked Cynosure, the maker of the Mona Lisa Touch, a system marketed as an FDA-approved treatment for vaginal atrophy, for evidence to support its claims that Mona Lisa Touch “is the only technology for vaginal and vulvar health with over 18+ published clinical studies” and is clinically proven to treat “painful symptoms of menopause, including intimacy.” They also asked for information about a modification to the originally approved device that was not brought to the FDA’s attention.
In a letter to Alma Lasers, whose Pixel CO2 Laser System was approved for use in a broad use of surgical applications including gynecologic surgery, the FDA noted that the device was being marketed as “FEMILIFT,” a laser procedure designed to “improve vaginal irregularities” and to assist “in vaginal mucosa revitalization.” The FDA demanded evidence for those claims.
The manufacturers have 30 days to respond to the FDA, which has not ruled out seeking enforcement action against firms with unsatisfactory responses.
For more than a decade, researchers have shown that healthy vaginal morphology is exceptionally wide ranging, including a recent study in more than 650 women, the largest to date (BJOG. 2018 Jun 25. doi: 10.1111/1471-0528.15387). Nonetheless, interest in elective vaginal procedures has only increased, with an industry report from the International Society of Aesthetic and Plastic Surgery describing a 45% increase in the use of one surgery, vaginal labiaplasty, between 2014 and 2016. Most procedures were performed in Brazil and the United States.
While plastic surgery societies support vaginal rejuvenation procedures, the American College of Obstetricians and Gynecologists has long frowned on them, with its first critique issued in a 2007 committee opinion. “No adequate studies have been published assessing the long-term satisfaction, safety, and complication rates for these procedures,” the association said last year in its most recent update on the subject.
Gynecologist David M. Jaspan, DO, of the Einstein Healthcare Network in Philadelphia, echoed ACOG’s views and said he welcomed FDA interest in vaginal rejuvenation.
The practice has “never been endorsed by the College or a board. It’s been considered a cosmetic procedure and it’s been under scrutiny for at least a decade,” Dr. Jaspan said. “I have reservations about the clinical outcomes and the training surrounds these procedures and I anxiously await randomized controlled trials to further evaluate them.”
Gynecologists who offer the procedures caution that they may have a role, and that randomized trials are underway to determine which groups of women might be best helped.
Marie Paraiso, MD, professor of obstetrics and gynecology at the Cleveland Clinic, said she uses the Mona Lisa Touch, a CO2 fractionated laser, to treat patients with genitourinary syndrome of menopause (GSM). These patients, Dr. Paraiso said, “complain of vaginal dryness and are unable to have intercourse or experience significant pain during or after intercourse. Some of them also may have irritative voiding, urinary frequency and urgency, or mild stress incontinence.”
Dr. Paraiso’s group has performed some 300 treatments with the laser and “we have fortunately not had patients complaining of persistent vaginal pain or scarring.” About 80%-90% of patients respond, she said, with some 20%-25% seeking retreatment within a year. “I believe for women who have contraindications to hormonal therapy or do not tolerate or cannot afford prolonged hormonal therapy, the CO2 fractional vaginal laser has been effective.”
Dr. Paraiso is also leading a multisite clinical trial randomizing about 200 patients with GSM to the laser treatment or estrogen-based vaginal creams, and following them for 6 months; thus far, she said, 6 of 89 patients, half in the laser arm, have reported mild to moderate adverse events.
Dr. Paraiso said she does not have a financial relationship with the manufacturer of Mona Lisa Touch, and that the trial was funded by the Foundation for Female Health Awareness, which receives unrestricted research grants from some device makers. “Our institute owns the laser and I have never been paid to train anyone to perform these procedures,” Dr. Paraiso added. “Our onus was to study the laser in order to improve the lives of our patients.”
Other trials comparing vaginal lasers with sham treatment are currently underway or in planning.
The North American Menopause Society struck a cautious note in response to the FDA criticism. In a statement issued August 1, JoAnn Pinkerton, MD, the society’s executive director, said the field needed prospective, randomized, sham-controlled trials of the laser and energy therapies. The therapies “may turn out to be an appropriate choice for many women, particularly for those concerned about breast cancer risk” associated with hormonal treatments. But until more robust data are available, doctors should “discuss the benefits and risks of all available treatment options for vaginal symptoms, including over-the-counter lubricants, vaginal moisturizers, and FDA-approved vaginal therapies such as vaginal estrogen and intravaginal dehydroepiandrosterone and oral therapies such as hormone therapy and ospemifene to determine the best treatment for women with GSM.”
Any discussion of vaginal energy-based therapies, should include the disclosure that these have not been approved for the specific indication, Dr. Pinkerton cautioned.
The Food and Drug Administration has issued a stern warning to several manufacturers and a statement of caution to the public concerning “vaginal rejuvenation,” an umbrella term for a host of procedures to alter vaginal tissue for therapeutic or cosmetic purposes.
Lasers and other energy-based devices have been approved to treat abnormal or precancerous cervical or vaginal tissue and general warts, but the FDA has not approved any to treat vaginal atrophy, urinary incontinence, or reduced sexual function.
Device manufacturers claim lasers can address these conditions despite limited scientific evidence for their safety or efficacy. Insurers do not reimburse the procedures, considering them to be cosmetic.
In a July 30 statement, FDA Commissioner Scott Gottlieb, MD, slammed “deceptive” marketing practices on the part of manufacturers.
The FDA has reviewed 12 complaints since December 2015 of adverse effects related to vaginal procedures using the devices. Two were from manufacturers reporting pain and bleeding in patients following treatment, FDA spokeswoman Deborah Kotz said in an interview. “The FDA has also received voluntary MedWatch reports from individual patients who experienced significant pain and discomfort from procedures performed with these devices.”
The agency has targeted seven firms: Alma Lasers, BTL Aesthetics, BTL Industries, Cynosure, InMode, Sciton, and ThermiGen, with letters demanding evidence of FDA approval, clearance, or intent to seek clearance for use of their products on female genitalia. They also asked for evidence backing specific claims.
In a July 26 letter to BTL Industries, for example, the FDA demanded to know why the firm was marketing its Exilis laser device, approved for the treatment of facial wrinkles, as “Ultra Femme 360,” which it called “a whole new approach to women’s intimate health.” The device, according to the manufacturer, “provides the shortest noninvasive radio-frequency treatment available for female intimate parts” and “is proven to increase elastin and collagen in the treatment area.”
The FDA asked Cynosure, the maker of the Mona Lisa Touch, a system marketed as an FDA-approved treatment for vaginal atrophy, for evidence to support its claims that Mona Lisa Touch “is the only technology for vaginal and vulvar health with over 18+ published clinical studies” and is clinically proven to treat “painful symptoms of menopause, including intimacy.” They also asked for information about a modification to the originally approved device that was not brought to the FDA’s attention.
In a letter to Alma Lasers, whose Pixel CO2 Laser System was approved for use in a broad use of surgical applications including gynecologic surgery, the FDA noted that the device was being marketed as “FEMILIFT,” a laser procedure designed to “improve vaginal irregularities” and to assist “in vaginal mucosa revitalization.” The FDA demanded evidence for those claims.
The manufacturers have 30 days to respond to the FDA, which has not ruled out seeking enforcement action against firms with unsatisfactory responses.
For more than a decade, researchers have shown that healthy vaginal morphology is exceptionally wide ranging, including a recent study in more than 650 women, the largest to date (BJOG. 2018 Jun 25. doi: 10.1111/1471-0528.15387). Nonetheless, interest in elective vaginal procedures has only increased, with an industry report from the International Society of Aesthetic and Plastic Surgery describing a 45% increase in the use of one surgery, vaginal labiaplasty, between 2014 and 2016. Most procedures were performed in Brazil and the United States.
While plastic surgery societies support vaginal rejuvenation procedures, the American College of Obstetricians and Gynecologists has long frowned on them, with its first critique issued in a 2007 committee opinion. “No adequate studies have been published assessing the long-term satisfaction, safety, and complication rates for these procedures,” the association said last year in its most recent update on the subject.
Gynecologist David M. Jaspan, DO, of the Einstein Healthcare Network in Philadelphia, echoed ACOG’s views and said he welcomed FDA interest in vaginal rejuvenation.
The practice has “never been endorsed by the College or a board. It’s been considered a cosmetic procedure and it’s been under scrutiny for at least a decade,” Dr. Jaspan said. “I have reservations about the clinical outcomes and the training surrounds these procedures and I anxiously await randomized controlled trials to further evaluate them.”
Gynecologists who offer the procedures caution that they may have a role, and that randomized trials are underway to determine which groups of women might be best helped.
Marie Paraiso, MD, professor of obstetrics and gynecology at the Cleveland Clinic, said she uses the Mona Lisa Touch, a CO2 fractionated laser, to treat patients with genitourinary syndrome of menopause (GSM). These patients, Dr. Paraiso said, “complain of vaginal dryness and are unable to have intercourse or experience significant pain during or after intercourse. Some of them also may have irritative voiding, urinary frequency and urgency, or mild stress incontinence.”
Dr. Paraiso’s group has performed some 300 treatments with the laser and “we have fortunately not had patients complaining of persistent vaginal pain or scarring.” About 80%-90% of patients respond, she said, with some 20%-25% seeking retreatment within a year. “I believe for women who have contraindications to hormonal therapy or do not tolerate or cannot afford prolonged hormonal therapy, the CO2 fractional vaginal laser has been effective.”
Dr. Paraiso is also leading a multisite clinical trial randomizing about 200 patients with GSM to the laser treatment or estrogen-based vaginal creams, and following them for 6 months; thus far, she said, 6 of 89 patients, half in the laser arm, have reported mild to moderate adverse events.
Dr. Paraiso said she does not have a financial relationship with the manufacturer of Mona Lisa Touch, and that the trial was funded by the Foundation for Female Health Awareness, which receives unrestricted research grants from some device makers. “Our institute owns the laser and I have never been paid to train anyone to perform these procedures,” Dr. Paraiso added. “Our onus was to study the laser in order to improve the lives of our patients.”
Other trials comparing vaginal lasers with sham treatment are currently underway or in planning.
The North American Menopause Society struck a cautious note in response to the FDA criticism. In a statement issued August 1, JoAnn Pinkerton, MD, the society’s executive director, said the field needed prospective, randomized, sham-controlled trials of the laser and energy therapies. The therapies “may turn out to be an appropriate choice for many women, particularly for those concerned about breast cancer risk” associated with hormonal treatments. But until more robust data are available, doctors should “discuss the benefits and risks of all available treatment options for vaginal symptoms, including over-the-counter lubricants, vaginal moisturizers, and FDA-approved vaginal therapies such as vaginal estrogen and intravaginal dehydroepiandrosterone and oral therapies such as hormone therapy and ospemifene to determine the best treatment for women with GSM.”
Any discussion of vaginal energy-based therapies, should include the disclosure that these have not been approved for the specific indication, Dr. Pinkerton cautioned.
Medical associations want withdrawal of Title X changes
Leading medical societies are calling on the Trump administration to withdraw its proposed changes to the federal Title X family planning program, calling the modifications a threat to essential health care for women.
In late May, the Department of Health & Human Services proposed broad changes to Title X, including no longer allowing staff at Title X clinics to counsel, refer, or provide information to women about abortions and mandating that Title X clinics that offer abortions maintain a separate facility for abortion services. The proposed changes aim to “refocus” the Title X program and ensure that all Title X services align with its family planning mission, according to the proposed rule published June 1.
In a July 31 letter to HHS, the American Medical Association requested that HHS withdraw the proposal, citing concerns from the medical community.
“We are very concerned that the proposed changes, if implemented, would undermine patients’ access to high-quality medical care and information, dangerously interfere with the patient-physician relationship and conflict with physicians’ ethical obligations, exclude qualified providers, and jeopardize public health,” James L. Madara, MD, chief executive officer and vice president of the AMA, wrote in a letter. “We urge HHS to withdraw this [proposal].”
The American College of Obstetricians and Gynecologists, the American College of Physicians, the American Academy of Pediatrics, the American Psychiatric Association, and 13 other health care associations also have called on the HHS to rescind its proposed rule. According to a statement from these associations, the proposal endangers women’s lives by restricting access to medically accurate information and preventive health care.
Title X is a long-standing federal program that provides funding for women’s health care and comprehensive family planning services, primarily to low-income and uninsured patients. Federal law prohibits the use of Title X funds to pay for abortions.
Under the proposed regulations, the Trump administration would define “family planning” as the voluntary process of identifying goals and developing a plan for the number and spacing of children and the means by which those goals may be achieved. This includes planning methods and services “to limit or enhance the likelihood of conception, including contraceptive methods and natural family planning or other fertility awareness-based methods,” according to the proposal. HHS specifies that family planning does not include postconception care, obstetric or prenatal care, or abortion as a method of family planning. HHS has proposed that, if a woman comes to a Title X–funded clinic and is pregnant, she be referred externally for pregnancy services. However, the proposed rule would no longer allow Title X programs to provide abortion counseling and/or referral.
According to HHS, requiring separate facilities for abortion-related care would ensure that Title X funds are used for the purposes expressly mandated by Congress – to offer family planning methods and services – and that any infrastructure built with Title X funds would not be used for impermissible purposes.
More than 100,000 comments have been submitted on the proposed rule since June. Antiabortion organizations, such as the Susan B. Anthony List, have expressed strong support for the proposed rule.
“The American people have repeatedly expressed their predominant policy preferences by supporting Congressional enactments designed to distinguish and separate abortion from family planning,” SBA List President Marjorie Dannenfelser wrote in a comment. “Abortion is not health care, nor is abortion family planning. The Clinton administration and subsequent presidential administrations have erroneously allowed the blatant distribution of Title X funding to abortion centers and abortion-referral facilities for years and in direct violation of the original purpose of Title X funding.”
A group of 14 state governors, meanwhile, has threatened legal action if the Trump administration moves forward with finalizing its rule. In a May 31 letter, the 14 Democratic governors urged HHS to halt its changes to the Title X program and said they would explore all options, including legal avenues, to protect patients’ access to care. More recently, Democratic governors in Washington, Oregon, Hawaii, and New York have said they will refuse all Title X funding if the Trump administration does not rescind its proposed changes to the program.
“This is not an issue about life or choices. This is an issue about the rights of millions of individuals who deserve the best health care available,” Hawaii governor David Ige said in a July 30 statement. “Hawaii will not accept federal funds for these programs if the proposed rules are implemented.”
Public comment on the proposed rule closed on July 31.
Leading medical societies are calling on the Trump administration to withdraw its proposed changes to the federal Title X family planning program, calling the modifications a threat to essential health care for women.
In late May, the Department of Health & Human Services proposed broad changes to Title X, including no longer allowing staff at Title X clinics to counsel, refer, or provide information to women about abortions and mandating that Title X clinics that offer abortions maintain a separate facility for abortion services. The proposed changes aim to “refocus” the Title X program and ensure that all Title X services align with its family planning mission, according to the proposed rule published June 1.
In a July 31 letter to HHS, the American Medical Association requested that HHS withdraw the proposal, citing concerns from the medical community.
“We are very concerned that the proposed changes, if implemented, would undermine patients’ access to high-quality medical care and information, dangerously interfere with the patient-physician relationship and conflict with physicians’ ethical obligations, exclude qualified providers, and jeopardize public health,” James L. Madara, MD, chief executive officer and vice president of the AMA, wrote in a letter. “We urge HHS to withdraw this [proposal].”
The American College of Obstetricians and Gynecologists, the American College of Physicians, the American Academy of Pediatrics, the American Psychiatric Association, and 13 other health care associations also have called on the HHS to rescind its proposed rule. According to a statement from these associations, the proposal endangers women’s lives by restricting access to medically accurate information and preventive health care.
Title X is a long-standing federal program that provides funding for women’s health care and comprehensive family planning services, primarily to low-income and uninsured patients. Federal law prohibits the use of Title X funds to pay for abortions.
Under the proposed regulations, the Trump administration would define “family planning” as the voluntary process of identifying goals and developing a plan for the number and spacing of children and the means by which those goals may be achieved. This includes planning methods and services “to limit or enhance the likelihood of conception, including contraceptive methods and natural family planning or other fertility awareness-based methods,” according to the proposal. HHS specifies that family planning does not include postconception care, obstetric or prenatal care, or abortion as a method of family planning. HHS has proposed that, if a woman comes to a Title X–funded clinic and is pregnant, she be referred externally for pregnancy services. However, the proposed rule would no longer allow Title X programs to provide abortion counseling and/or referral.
According to HHS, requiring separate facilities for abortion-related care would ensure that Title X funds are used for the purposes expressly mandated by Congress – to offer family planning methods and services – and that any infrastructure built with Title X funds would not be used for impermissible purposes.
More than 100,000 comments have been submitted on the proposed rule since June. Antiabortion organizations, such as the Susan B. Anthony List, have expressed strong support for the proposed rule.
“The American people have repeatedly expressed their predominant policy preferences by supporting Congressional enactments designed to distinguish and separate abortion from family planning,” SBA List President Marjorie Dannenfelser wrote in a comment. “Abortion is not health care, nor is abortion family planning. The Clinton administration and subsequent presidential administrations have erroneously allowed the blatant distribution of Title X funding to abortion centers and abortion-referral facilities for years and in direct violation of the original purpose of Title X funding.”
A group of 14 state governors, meanwhile, has threatened legal action if the Trump administration moves forward with finalizing its rule. In a May 31 letter, the 14 Democratic governors urged HHS to halt its changes to the Title X program and said they would explore all options, including legal avenues, to protect patients’ access to care. More recently, Democratic governors in Washington, Oregon, Hawaii, and New York have said they will refuse all Title X funding if the Trump administration does not rescind its proposed changes to the program.
“This is not an issue about life or choices. This is an issue about the rights of millions of individuals who deserve the best health care available,” Hawaii governor David Ige said in a July 30 statement. “Hawaii will not accept federal funds for these programs if the proposed rules are implemented.”
Public comment on the proposed rule closed on July 31.
Leading medical societies are calling on the Trump administration to withdraw its proposed changes to the federal Title X family planning program, calling the modifications a threat to essential health care for women.
In late May, the Department of Health & Human Services proposed broad changes to Title X, including no longer allowing staff at Title X clinics to counsel, refer, or provide information to women about abortions and mandating that Title X clinics that offer abortions maintain a separate facility for abortion services. The proposed changes aim to “refocus” the Title X program and ensure that all Title X services align with its family planning mission, according to the proposed rule published June 1.
In a July 31 letter to HHS, the American Medical Association requested that HHS withdraw the proposal, citing concerns from the medical community.
“We are very concerned that the proposed changes, if implemented, would undermine patients’ access to high-quality medical care and information, dangerously interfere with the patient-physician relationship and conflict with physicians’ ethical obligations, exclude qualified providers, and jeopardize public health,” James L. Madara, MD, chief executive officer and vice president of the AMA, wrote in a letter. “We urge HHS to withdraw this [proposal].”
The American College of Obstetricians and Gynecologists, the American College of Physicians, the American Academy of Pediatrics, the American Psychiatric Association, and 13 other health care associations also have called on the HHS to rescind its proposed rule. According to a statement from these associations, the proposal endangers women’s lives by restricting access to medically accurate information and preventive health care.
Title X is a long-standing federal program that provides funding for women’s health care and comprehensive family planning services, primarily to low-income and uninsured patients. Federal law prohibits the use of Title X funds to pay for abortions.
Under the proposed regulations, the Trump administration would define “family planning” as the voluntary process of identifying goals and developing a plan for the number and spacing of children and the means by which those goals may be achieved. This includes planning methods and services “to limit or enhance the likelihood of conception, including contraceptive methods and natural family planning or other fertility awareness-based methods,” according to the proposal. HHS specifies that family planning does not include postconception care, obstetric or prenatal care, or abortion as a method of family planning. HHS has proposed that, if a woman comes to a Title X–funded clinic and is pregnant, she be referred externally for pregnancy services. However, the proposed rule would no longer allow Title X programs to provide abortion counseling and/or referral.
According to HHS, requiring separate facilities for abortion-related care would ensure that Title X funds are used for the purposes expressly mandated by Congress – to offer family planning methods and services – and that any infrastructure built with Title X funds would not be used for impermissible purposes.
More than 100,000 comments have been submitted on the proposed rule since June. Antiabortion organizations, such as the Susan B. Anthony List, have expressed strong support for the proposed rule.
“The American people have repeatedly expressed their predominant policy preferences by supporting Congressional enactments designed to distinguish and separate abortion from family planning,” SBA List President Marjorie Dannenfelser wrote in a comment. “Abortion is not health care, nor is abortion family planning. The Clinton administration and subsequent presidential administrations have erroneously allowed the blatant distribution of Title X funding to abortion centers and abortion-referral facilities for years and in direct violation of the original purpose of Title X funding.”
A group of 14 state governors, meanwhile, has threatened legal action if the Trump administration moves forward with finalizing its rule. In a May 31 letter, the 14 Democratic governors urged HHS to halt its changes to the Title X program and said they would explore all options, including legal avenues, to protect patients’ access to care. More recently, Democratic governors in Washington, Oregon, Hawaii, and New York have said they will refuse all Title X funding if the Trump administration does not rescind its proposed changes to the program.
“This is not an issue about life or choices. This is an issue about the rights of millions of individuals who deserve the best health care available,” Hawaii governor David Ige said in a July 30 statement. “Hawaii will not accept federal funds for these programs if the proposed rules are implemented.”
Public comment on the proposed rule closed on July 31.
The Mother of All Skin Problems
Each time this 32-year-old woman has a baby—she’s had four to date—she notices that sections of her face darken. Early on, she observed a pattern in which the coming of winter coincided with a lightening of these affected areas—but now the effect lasts year-round, with progressive darkening. She has not tried any products (OTC or prescription) for this problem.
Growing up in the South, the patient and her family spent most summers boating, swimming, and fishing. Her use of sunscreen was sporadic, but she would tan easily regardless.
Her health is good, aside from a 15-year history of smoking.
EXAMINATION
There is excessive hyperpigmentation (brown) on the patient’s face. It follows a mask-like pattern, including her maxilla and the periphery of her face.
Elsewhere, there is abundant evidence of excessive sun exposure, with focal hyperpigmentation and telangiectasias on her arms. She has type IV skin, consistent with her Native American ancestry.
What is the diagnosis?
DISCUSSION
Melasma, also known as chloasma and dubbed the “mask of pregnancy,” is an extremely common problem that results from a combination of naturally dark skin, lots of sun exposure, and increased levels of estrogenic hormones. The latter can result from pregnancy or from oral contraceptive or estrogen replacement therapy use. Another precipitating factor is thyroid disease, certain types of which lead to an increase in melanocytic stimulating hormone.
Melasma, as one might expect, is seen almost exclusively in women, though a rare male is affected. It is especially common among Latina, Native American, and African-American women, whose melanocytes are especially able to produce pigment.
There are several treatments for melasma, none of them perfect, including tretinoin, azelaic acid, chemical peels, dermabrasion, and lasers. The most common treatment is hydroquinone cream, available in the US in both OTC (2%) and prescription (4%) strengths. However, hydroquinone is available OTC in stronger formulations (15% to 20%) in many Central and South American countries; unfortunately, many women who obtain and use these products experience exogenous ochronosis—a worsening or even precipitation of melasma, resulting from excessive production of tyrosinase.
Any treatment must be used in conjunction with rigorous sunscreen application. A full-spectrum product, with titanium dioxide and zinc oxide as the only active ingredients, must be used, because chemical-laden sunscreens don’t do as good a job covering UVA, UVB, and visible light. Convincing women who are unaccustomed to needing sunscreen to use it religiously is part of what makes treating melasma difficult.
The differential for melasma includes postinflammatory hyperpigmentation (eg, following an episode of contact dermatitis) and simple solar lentigines.
This patient was treated with hydroquinone 4% cream bid, plus sunscreen. She was also given information about other treatment options, such as laser and dermabrasion.
TAKE-HOME LEARNING POINTS
- Melasma, also known at chloasma, is quite common, especially among women with darker skin who live in sunny parts of the world.
- It results from a combination of dark skin, an increased level of estrogenic hormones (eg, with pregnancy, birth control pills, or estrogen replacement therapy), and excessive exposure to UV light.
- While hydroquinone cream can be an effective treatment, the maximum strength should be 4%; overuse of stronger concentrations (available in other countries, such as Mexico), can actually cause melasma to worsen.
Each time this 32-year-old woman has a baby—she’s had four to date—she notices that sections of her face darken. Early on, she observed a pattern in which the coming of winter coincided with a lightening of these affected areas—but now the effect lasts year-round, with progressive darkening. She has not tried any products (OTC or prescription) for this problem.
Growing up in the South, the patient and her family spent most summers boating, swimming, and fishing. Her use of sunscreen was sporadic, but she would tan easily regardless.
Her health is good, aside from a 15-year history of smoking.
EXAMINATION
There is excessive hyperpigmentation (brown) on the patient’s face. It follows a mask-like pattern, including her maxilla and the periphery of her face.

Elsewhere, there is abundant evidence of excessive sun exposure, with focal hyperpigmentation and telangiectasias on her arms. She has type IV skin, consistent with her Native American ancestry.
What is the diagnosis?
DISCUSSION
Melasma, also known as chloasma and dubbed the “mask of pregnancy,” is an extremely common problem that results from a combination of naturally dark skin, lots of sun exposure, and increased levels of estrogenic hormones. The latter can result from pregnancy or from oral contraceptive or estrogen replacement therapy use. Another precipitating factor is thyroid disease, certain types of which lead to an increase in melanocytic stimulating hormone.
Melasma, as one might expect, is seen almost exclusively in women, though a rare male is affected. It is especially common among Latina, Native American, and African-American women, whose melanocytes are especially able to produce pigment.
There are several treatments for melasma, none of them perfect, including tretinoin, azelaic acid, chemical peels, dermabrasion, and lasers. The most common treatment is hydroquinone cream, available in the US in both OTC (2%) and prescription (4%) strengths. However, hydroquinone is available OTC in stronger formulations (15% to 20%) in many Central and South American countries; unfortunately, many women who obtain and use these products experience exogenous ochronosis—a worsening or even precipitation of melasma, resulting from excessive production of tyrosinase.
Any treatment must be used in conjunction with rigorous sunscreen application. A full-spectrum product, with titanium dioxide and zinc oxide as the only active ingredients, must be used, because chemical-laden sunscreens don’t do as good a job covering UVA, UVB, and visible light. Convincing women who are unaccustomed to needing sunscreen to use it religiously is part of what makes treating melasma difficult.
The differential for melasma includes postinflammatory hyperpigmentation (eg, following an episode of contact dermatitis) and simple solar lentigines.
This patient was treated with hydroquinone 4% cream bid, plus sunscreen. She was also given information about other treatment options, such as laser and dermabrasion.
TAKE-HOME LEARNING POINTS
- Melasma, also known at chloasma, is quite common, especially among women with darker skin who live in sunny parts of the world.
- It results from a combination of dark skin, an increased level of estrogenic hormones (eg, with pregnancy, birth control pills, or estrogen replacement therapy), and excessive exposure to UV light.
- While hydroquinone cream can be an effective treatment, the maximum strength should be 4%; overuse of stronger concentrations (available in other countries, such as Mexico), can actually cause melasma to worsen.
Each time this 32-year-old woman has a baby—she’s had four to date—she notices that sections of her face darken. Early on, she observed a pattern in which the coming of winter coincided with a lightening of these affected areas—but now the effect lasts year-round, with progressive darkening. She has not tried any products (OTC or prescription) for this problem.
Growing up in the South, the patient and her family spent most summers boating, swimming, and fishing. Her use of sunscreen was sporadic, but she would tan easily regardless.
Her health is good, aside from a 15-year history of smoking.
EXAMINATION
There is excessive hyperpigmentation (brown) on the patient’s face. It follows a mask-like pattern, including her maxilla and the periphery of her face.

Elsewhere, there is abundant evidence of excessive sun exposure, with focal hyperpigmentation and telangiectasias on her arms. She has type IV skin, consistent with her Native American ancestry.
What is the diagnosis?
DISCUSSION
Melasma, also known as chloasma and dubbed the “mask of pregnancy,” is an extremely common problem that results from a combination of naturally dark skin, lots of sun exposure, and increased levels of estrogenic hormones. The latter can result from pregnancy or from oral contraceptive or estrogen replacement therapy use. Another precipitating factor is thyroid disease, certain types of which lead to an increase in melanocytic stimulating hormone.
Melasma, as one might expect, is seen almost exclusively in women, though a rare male is affected. It is especially common among Latina, Native American, and African-American women, whose melanocytes are especially able to produce pigment.
There are several treatments for melasma, none of them perfect, including tretinoin, azelaic acid, chemical peels, dermabrasion, and lasers. The most common treatment is hydroquinone cream, available in the US in both OTC (2%) and prescription (4%) strengths. However, hydroquinone is available OTC in stronger formulations (15% to 20%) in many Central and South American countries; unfortunately, many women who obtain and use these products experience exogenous ochronosis—a worsening or even precipitation of melasma, resulting from excessive production of tyrosinase.
Any treatment must be used in conjunction with rigorous sunscreen application. A full-spectrum product, with titanium dioxide and zinc oxide as the only active ingredients, must be used, because chemical-laden sunscreens don’t do as good a job covering UVA, UVB, and visible light. Convincing women who are unaccustomed to needing sunscreen to use it religiously is part of what makes treating melasma difficult.
The differential for melasma includes postinflammatory hyperpigmentation (eg, following an episode of contact dermatitis) and simple solar lentigines.
This patient was treated with hydroquinone 4% cream bid, plus sunscreen. She was also given information about other treatment options, such as laser and dermabrasion.
TAKE-HOME LEARNING POINTS
- Melasma, also known at chloasma, is quite common, especially among women with darker skin who live in sunny parts of the world.
- It results from a combination of dark skin, an increased level of estrogenic hormones (eg, with pregnancy, birth control pills, or estrogen replacement therapy), and excessive exposure to UV light.
- While hydroquinone cream can be an effective treatment, the maximum strength should be 4%; overuse of stronger concentrations (available in other countries, such as Mexico), can actually cause melasma to worsen.
Angular cheilitis induced by iron deficiency anemia
A 20-year-old woman had a 4-month history of painful red erosions around the mouth. She had no dysphagia or fatigue and no history of diarrhea, gluten intolerance, or diabetes mellitus. An antifungal-antibacterial ointment prescribed by her dentist had provided no relief.
- Hemoglobin 8.0 g/dL (reference range for females 12.3–15.3)
- Mean corpuscular volume 62 fL (80–100)
- Serum ferritin 1.3 ng/mL (15–200)
- Reticulocyte count 0.86% (0.5–1.5)
- White blood cell count 9.8 × 109/L (4.5–11.0)
- Platelet count 450 × 109/L (150–400).
Vitamin B12 and folate levels were normal, and tests for antitissue transglutaminase and antinuclear antibodies were negative.
Based on these results, the diagnosis was angular cheilitis from iron deficiency anemia. Treatment with oral ferrous gluconate 300 mg twice daily cleared the cheilitis, and after 4 weeks of this treatment, the hemoglobin level increased to 9.8 g/dL, the serum ferritin increased to 7 ng/mL, and the reticulocyte count increased to 2.6%. She was advised to continue taking oral iron tablets for another 3 months until the hemoglobin level reached 12.0 g/dL.
During 2 years of follow-up, she had no recurrence of angular cheilitis, and her hemoglobin and serum ferritin levels remained normal. Ferrous gluconate was her only medication from the time of her diagnosis.
A BROAD DIFFERENTIAL DIAGNOSIS
Angular cheilitis (perlèche) is an inflammatory condition characterized by erosive inflammation at one or both angles of the mouth. It typically presents as erythema, scaling, fissuring, and ulceration. A wide variety of factors, including nutritional deficiencies, local and systemic factors, and drug side effects, may produce cheilitis.1,2
Nutritional deficiencies account for 25% of all cases of angular cheilitis3 and include iron deficiency and deficiencies of the B vitamins riboflavin (B2), niacin (B3), pyridoxine (B6), and cyanocobalamin (B12).1
Local causes include infection with Candida albicans or Staphylococcus aureus and allergic contact dermatitis. Common causes of allergic contact dermatitis include lipstick, toothpaste, mouthwash, cosmetics, sunscreen, fragrance, metals such as nickel, and dental appliances.1
Systemic diseases associated with angular cheilitis include xerostomia, inflammatory bowel disease, Sjögren syndrome, glucagonoma, and human immunodeficiency virus.1
Drugs that cause angular cheilitis include isotretinoin, sorafenib (antineoplastic kinase inhibitor), and ointments or creams such as neomycin sulfate–polymyxin B sulfate, bacitracin, idoxuridine, and steroids.1,4
Conditions that mimic angular cheilitis include herpes simplex type 1 (herpes labialis) and actinic cheilitis. Herpes labialis, characterized by burning sensation, itching, or paresthesia, usually precedes a recurrence of vesicles that eventually ulcerate or form a crust and heal without a crust. Herpes labialis often recurs, affecting the vermilion border and lasting approximately 1 week.
Actinic cheilitis, a premalignant condition that commonly involves the lower lip with sparing of the corners of the mouth, is caused by excessive sun exposure. Patients often have persistent dryness and cracking of the lips.
In our patient, angular cheilitis was the main clinical manifestation of iron deficiency anemia, highlighting the importance of looking for iron deficiency in affected patients without a more obvious cause.
- Park KK, Brodell RT, Helms SE. Angular cheilitis, part 2: nutritional, systemic, and drug-related causes and treatment. Cutis 2011; 88(1):27–32. pmid:21877503
- Park KK, Brodell RT, Helms SE. Angular cheilitis, part 1: local etiologies. Cutis 2011; 87(6):289–295. pmid:21838086
- Konstantinidis AB, Hatziotis JH. Angular cheilosis: an analysis of 156 cases. J Oral Med 1984; 39(4):199–206. pmid:6594458
- Yang CH, Lin WC, Chuang CK, et al. Hand-foot skin reaction in patients treated with sorafenib: a clinicopathological study of cutaneous manifestations due to multitargeted kinase inhibitor therapy. Br J Dermatol 2008; 158(3):592–596. doi:10.1111/j.1365-2133.2007.08357.x
A 20-year-old woman had a 4-month history of painful red erosions around the mouth. She had no dysphagia or fatigue and no history of diarrhea, gluten intolerance, or diabetes mellitus. An antifungal-antibacterial ointment prescribed by her dentist had provided no relief.
- Hemoglobin 8.0 g/dL (reference range for females 12.3–15.3)
- Mean corpuscular volume 62 fL (80–100)
- Serum ferritin 1.3 ng/mL (15–200)
- Reticulocyte count 0.86% (0.5–1.5)
- White blood cell count 9.8 × 109/L (4.5–11.0)
- Platelet count 450 × 109/L (150–400).
Vitamin B12 and folate levels were normal, and tests for antitissue transglutaminase and antinuclear antibodies were negative.
Based on these results, the diagnosis was angular cheilitis from iron deficiency anemia. Treatment with oral ferrous gluconate 300 mg twice daily cleared the cheilitis, and after 4 weeks of this treatment, the hemoglobin level increased to 9.8 g/dL, the serum ferritin increased to 7 ng/mL, and the reticulocyte count increased to 2.6%. She was advised to continue taking oral iron tablets for another 3 months until the hemoglobin level reached 12.0 g/dL.
During 2 years of follow-up, she had no recurrence of angular cheilitis, and her hemoglobin and serum ferritin levels remained normal. Ferrous gluconate was her only medication from the time of her diagnosis.
A BROAD DIFFERENTIAL DIAGNOSIS
Angular cheilitis (perlèche) is an inflammatory condition characterized by erosive inflammation at one or both angles of the mouth. It typically presents as erythema, scaling, fissuring, and ulceration. A wide variety of factors, including nutritional deficiencies, local and systemic factors, and drug side effects, may produce cheilitis.1,2
Nutritional deficiencies account for 25% of all cases of angular cheilitis3 and include iron deficiency and deficiencies of the B vitamins riboflavin (B2), niacin (B3), pyridoxine (B6), and cyanocobalamin (B12).1
Local causes include infection with Candida albicans or Staphylococcus aureus and allergic contact dermatitis. Common causes of allergic contact dermatitis include lipstick, toothpaste, mouthwash, cosmetics, sunscreen, fragrance, metals such as nickel, and dental appliances.1
Systemic diseases associated with angular cheilitis include xerostomia, inflammatory bowel disease, Sjögren syndrome, glucagonoma, and human immunodeficiency virus.1
Drugs that cause angular cheilitis include isotretinoin, sorafenib (antineoplastic kinase inhibitor), and ointments or creams such as neomycin sulfate–polymyxin B sulfate, bacitracin, idoxuridine, and steroids.1,4
Conditions that mimic angular cheilitis include herpes simplex type 1 (herpes labialis) and actinic cheilitis. Herpes labialis, characterized by burning sensation, itching, or paresthesia, usually precedes a recurrence of vesicles that eventually ulcerate or form a crust and heal without a crust. Herpes labialis often recurs, affecting the vermilion border and lasting approximately 1 week.
Actinic cheilitis, a premalignant condition that commonly involves the lower lip with sparing of the corners of the mouth, is caused by excessive sun exposure. Patients often have persistent dryness and cracking of the lips.
In our patient, angular cheilitis was the main clinical manifestation of iron deficiency anemia, highlighting the importance of looking for iron deficiency in affected patients without a more obvious cause.
A 20-year-old woman had a 4-month history of painful red erosions around the mouth. She had no dysphagia or fatigue and no history of diarrhea, gluten intolerance, or diabetes mellitus. An antifungal-antibacterial ointment prescribed by her dentist had provided no relief.
- Hemoglobin 8.0 g/dL (reference range for females 12.3–15.3)
- Mean corpuscular volume 62 fL (80–100)
- Serum ferritin 1.3 ng/mL (15–200)
- Reticulocyte count 0.86% (0.5–1.5)
- White blood cell count 9.8 × 109/L (4.5–11.0)
- Platelet count 450 × 109/L (150–400).
Vitamin B12 and folate levels were normal, and tests for antitissue transglutaminase and antinuclear antibodies were negative.
Based on these results, the diagnosis was angular cheilitis from iron deficiency anemia. Treatment with oral ferrous gluconate 300 mg twice daily cleared the cheilitis, and after 4 weeks of this treatment, the hemoglobin level increased to 9.8 g/dL, the serum ferritin increased to 7 ng/mL, and the reticulocyte count increased to 2.6%. She was advised to continue taking oral iron tablets for another 3 months until the hemoglobin level reached 12.0 g/dL.
During 2 years of follow-up, she had no recurrence of angular cheilitis, and her hemoglobin and serum ferritin levels remained normal. Ferrous gluconate was her only medication from the time of her diagnosis.
A BROAD DIFFERENTIAL DIAGNOSIS
Angular cheilitis (perlèche) is an inflammatory condition characterized by erosive inflammation at one or both angles of the mouth. It typically presents as erythema, scaling, fissuring, and ulceration. A wide variety of factors, including nutritional deficiencies, local and systemic factors, and drug side effects, may produce cheilitis.1,2
Nutritional deficiencies account for 25% of all cases of angular cheilitis3 and include iron deficiency and deficiencies of the B vitamins riboflavin (B2), niacin (B3), pyridoxine (B6), and cyanocobalamin (B12).1
Local causes include infection with Candida albicans or Staphylococcus aureus and allergic contact dermatitis. Common causes of allergic contact dermatitis include lipstick, toothpaste, mouthwash, cosmetics, sunscreen, fragrance, metals such as nickel, and dental appliances.1
Systemic diseases associated with angular cheilitis include xerostomia, inflammatory bowel disease, Sjögren syndrome, glucagonoma, and human immunodeficiency virus.1
Drugs that cause angular cheilitis include isotretinoin, sorafenib (antineoplastic kinase inhibitor), and ointments or creams such as neomycin sulfate–polymyxin B sulfate, bacitracin, idoxuridine, and steroids.1,4
Conditions that mimic angular cheilitis include herpes simplex type 1 (herpes labialis) and actinic cheilitis. Herpes labialis, characterized by burning sensation, itching, or paresthesia, usually precedes a recurrence of vesicles that eventually ulcerate or form a crust and heal without a crust. Herpes labialis often recurs, affecting the vermilion border and lasting approximately 1 week.
Actinic cheilitis, a premalignant condition that commonly involves the lower lip with sparing of the corners of the mouth, is caused by excessive sun exposure. Patients often have persistent dryness and cracking of the lips.
In our patient, angular cheilitis was the main clinical manifestation of iron deficiency anemia, highlighting the importance of looking for iron deficiency in affected patients without a more obvious cause.
- Park KK, Brodell RT, Helms SE. Angular cheilitis, part 2: nutritional, systemic, and drug-related causes and treatment. Cutis 2011; 88(1):27–32. pmid:21877503
- Park KK, Brodell RT, Helms SE. Angular cheilitis, part 1: local etiologies. Cutis 2011; 87(6):289–295. pmid:21838086
- Konstantinidis AB, Hatziotis JH. Angular cheilosis: an analysis of 156 cases. J Oral Med 1984; 39(4):199–206. pmid:6594458
- Yang CH, Lin WC, Chuang CK, et al. Hand-foot skin reaction in patients treated with sorafenib: a clinicopathological study of cutaneous manifestations due to multitargeted kinase inhibitor therapy. Br J Dermatol 2008; 158(3):592–596. doi:10.1111/j.1365-2133.2007.08357.x
- Park KK, Brodell RT, Helms SE. Angular cheilitis, part 2: nutritional, systemic, and drug-related causes and treatment. Cutis 2011; 88(1):27–32. pmid:21877503
- Park KK, Brodell RT, Helms SE. Angular cheilitis, part 1: local etiologies. Cutis 2011; 87(6):289–295. pmid:21838086
- Konstantinidis AB, Hatziotis JH. Angular cheilosis: an analysis of 156 cases. J Oral Med 1984; 39(4):199–206. pmid:6594458
- Yang CH, Lin WC, Chuang CK, et al. Hand-foot skin reaction in patients treated with sorafenib: a clinicopathological study of cutaneous manifestations due to multitargeted kinase inhibitor therapy. Br J Dermatol 2008; 158(3):592–596. doi:10.1111/j.1365-2133.2007.08357.x
Diabetes and pregnancy: Risks and opportunities
A 29-year-old nulliparous woman presents for a routine checkup. She has hypertension and type 2 diabetes mellitus. Her current medications are chlorpropamide 500 mg daily, metformin 500 mg twice a day, lisinopril 40 mg daily, simvastatin 40 mg daily, and aspirin 81 mg daily. Her body mass index is 37 kg/m2 and her blood pressure is 130/80 mm Hg. Her hemoglobin A1c level is 7.8% and her low-density lipoprotein cholesterol 90 mg/dL.
She is considering pregnancy. How would you counsel her?
DEFINING DIABETES IN PREGNANCY
Diabetes in pregnant women, both gestational and pregestational, is the most common medical complication associated with pregnancy.1
- Gestational diabetes is defined as diabetes that is diagnosed during the second or third trimester of pregnancy and that is not clearly pregestational.2
- Pregestational diabetes exists before pregnancy and can be either type 1 or type 2.
Most cases of diabetes diagnosed during the first trimester reflect pregestational diabetes, as gestational diabetes occurs when insulin resistance increases in the later trimesters.
Type 1 diabetes involves autoimmune destruction of pancreatic islet cells, leading to insulin deficiency and the need for insulin therapy. Type 2 diabetes is characterized by insulin resistance rather than overall insulin deficiency. Type 2 diabetes tends to be associated with comorbidities such as obesity and hypertension, which are independent risk factors for adverse perinatal outcomes.3,4
Gestational diabetes accounts for most cases of diabetes during pregnancy. Although both pregestational and gestational diabetes increase the risk of maternal and fetal complications, pregestational diabetes is associated with significantly greater risks.1
IMPACT OF DIABETES ON THE MOTHER
Pregnancy increases the risk of maternal hypoglycemia, especially during the first trimester in patients with type 1 diabetes, as insulin sensitivity increases in early pregnancy.1 Pregnant women with diabetes may also have an altered counterregulatory response and less hypoglycemic awareness.1 Insulin resistance rises during the second and early third trimesters, increasing the risk of hyperglycemia in women with diabetes.1
Glycemic control during pregnancy is usually easier to achieve in patients with type 2 diabetes than with type 1, but it may require much higher insulin doses.
Because pregnancy is inherently a ketogenic state, women with type 1 diabetes are at higher risk of diabetic ketoacidosis, particularly during the second and third trimesters.1 There are reports of euglycemic diabetic ketoacidosis in pregnant women with either gestational or pregestational diabetes.5
Diabetes is associated with a risk of preeclampsia 4 times higher than in nondiabetic women.6 Other potential pregnancy-related complications include infections, polyhydramnios, spontaneous abortion, and cesarean delivery.1,7 The risk of pregnancy loss is similar in women with either type 1 or type 2 diabetes (2.6% and 3.7%, respectively), but the causes are different.8 Although preexisting diabetic complications such as retinopathy, nephropathy, and gastroparesis can be exacerbated during pregnancy,1 only severe gastroparesis and advanced renal disease are considered relative contraindications to pregnancy.
IMPACT OF DIABETES ON THE FETUS
Fetal complications of maternal diabetes include embryopathy (fetal malformations) and fetopathy (overgrowth, ie, fetus large for gestational age, and increased risk of fetal death or distress). Maternal hyperglycemia is associated with diabetic embryopathy, resulting in major birth defects in 5% to 25% of pregnancies and spontaneous abortions in 15% to 20%.9,10 There is a 2- to 6-fold increase in risk of congenital malformations.6
The most common diabetes-associated congenital malformations affect the cardiovascular system. Congenital heart disease includes tetralogy of Fallot, transposition of the great vessels, septal defects, and anomalous pulmonary venous return. Other relatively common defects involve the fetal central nervous system, spine, orofacial system, kidneys, urogenital system, gastrointestinal tract, and skeleton.11
The risk of fetopathy is proportional to the degree of maternal hyperglycemia. Excess maternal glucose and fatty acid levels can lead to fetal hyperglycemia and overgrowth, which increases fetal oxygen requirements. Erythropoietin levels rise, causing an increase in red cell mass, with subsequent hyperviscosity within the placenta and higher risk of fetal death.
Other complications include intrauterine growth restriction, prematurity, and preterm delivery. Fetal macrosomia (birth weight > 90th percentile or 4 kg, approximately 8 lb, 13 oz) occurs in 27% to 62% of children born to mothers with diabetes, a rate 10 times higher than in patients without diabetes. It contributes to shoulder dystocia (risk 2 to 4 times higher in diabetic pregnancies) and cesarean delivery.6 Infants born to mothers with diabetes also have higher risks of neonatal hypoglycemia, erythrocytosis, hyperbilirubinemia, hypocalcemia, respiratory distress, cardiomyopathy, and death, as well as for developing diabetes, obesity, and other adverse cardiometabolic outcomes later in life.11
GET GLUCOSE UNDER CONTROL BEFORE PREGNANCY
Nearly half of pregnancies in the general population are unplanned,15 so preconception diabetes assessment needs to be part of routine medical care for all reproductive-age women. Because most organogenesis occurs during the first 5 to 8 weeks after fertilization—potentially before a woman realizes she is pregnant—achieving optimal glycemic control before conception is necessary to improve pregnancy outcomes.1
EVERY VISIT IS AN OPPORTUNITY
Every medical visit with a reproductive-age woman with diabetes is an opportunity for counseling about pregnancy. Topics that need to be discussed include the risks of unplanned pregnancy and of poor metabolic control, and the benefits of improved maternal and fetal outcomes with appropriate pregnancy planning and diabetes management.
Referral to a registered dietitian for individualized counseling about proper nutrition, particularly during pregnancy, has been associated with positive outcomes.16 Patients with diabetes and at high risk of pregnancy complications should be referred to a clinic that specializes in high-risk pregnancies.
Practitioners also should emphasize the importance of regular exercise and encourage patients to maintain or achieve a medically optimal weight before conception. Ideally, this would be a normal body mass index; however, this is not always possible.
In women who are planning pregnancy or are not on effective contraception, medications should be reviewed for potential teratogenicity. If needed, discuss alternative medications or switch to safer ones. However, these changes should not interrupt diabetes treatment.
In addition, ensure that the patient is up to date on age- and disease-appropriate preventive care (eg, immunizations, screening for sexually transmitted disease and malignancy). Counseling and intervention for use of tobacco, alcohol, and recreational drugs are also important. As with any preconception counseling, the patient (and her partner, if possible) should be advised to avoid travel to areas where Zika virus is endemic, and informed about the availability of expanded carrier genetic screening through her obstetric provider.
Finally, pregnant women with diabetes benefit from screening for diabetic complications including hypertension, retinopathy, cardiovascular disease, neuropathy, and nephropathy.
ASSESSING RISKS
Blood pressure
Chronic (preexisting) hypertension is defined as a systolic pressure 140 mm Hg or higher or a diastolic pressure 90 mm Hg or higher, or both, that antedates pregnancy or is present before the 20th week of pregnancy.3 Chronic hypertension has been reported in up to 5% of pregnant women and is associated with increased risk of preterm delivery, superimposed preeclampsia, low birth weight, and perinatal death.3
Reproductive-age women with diabetes and high blood pressure benefit from lifestyle and behavioral modifications.17 If drug therapy is needed, antihypertensive drugs that are safe for the fetus should be used. Treatment of mild or moderate hypertension during pregnancy reduces the risk of progression to severe hypertension but may not improve obstetric outcomes.
Diabetic retinopathy
Diabetic retinopathy can significantly worsen during pregnancy: the risk of progression is double that in the nonpregnant state.18 Women with diabetes who are contemplating pregnancy should have a comprehensive eye examination before conception, and any active proliferative retinopathy needs to be treated. These patients may require ophthalmologic monitoring and treatment during pregnancy. (Note: laser photocoagulation is not contraindicated during pregnancy.)
Cardiovascular disease
Cardiovascular physiology changes dramatically during pregnancy. Cardiovascular disease, especially when superimposed on diabetes, can increase the risk of maternal death. Thus, evaluation for cardiovascular risk factors as well as cardiovascular system integrity before conception is important. Listen for arterial bruits and murmurs, and assess peripheral pulses. Consideration should be given to obtaining a preconception resting electrocardiogram in women with diabetes who are over age 35 or who are suspected of having cardiovascular disease.16
Neurologic disorders
Peripheral neuropathy, the most common neurologic complication of diabetes, is associated with injury and infection.19
Autonomic neuropathy is associated with decreased cardiac responsiveness and orthostatic hypotension.19 Diabetic gastroparesis alone can precipitate serious complications during pregnancy, including extreme hypoglycemia and hyperglycemia, increased risk of diabetic ketoacidosis, weight loss, malnutrition, frequent hospitalizations, and increased requirement for parenteral nutrition.20
Although diabetic neuropathy does not significantly worsen during pregnancy, women with preexisting gastroparesis should be counseled on the substantial risks associated with pregnancy. Screening for neuropathy should be part of all diabetic preconception examinations.
Renal complications
Pregnancy in women with diabetes and preexisting renal dysfunction increases their risk of accelerated progression of diabetic kidney disease.21 Preexisting renal dysfunction also increases the risk of pregnancy-related complications, such as stillbirth, intrauterine growth restriction, gestational hypertension, preeclampsia, and preterm delivery.19,21,22 Further, the risk of pregnancy complications correlates directly with the severity of renal dysfunction.22
Psychiatric disorders
Emotional wellness is essential for optimal diabetes management. It is important to recognize the emotional impact of diabetes in pregnant women and to conduct routine screening for depression, anxiety, stress, and eating disorders.16
LABORATORY TESTS TO CONSIDER
Hemoglobin A1c. The general consensus is to achieve the lowest hemoglobin A1c level possible that does not increase the risk of hypoglycemia. The American Diabetes Association (ADA) recommends that, before attempting to conceive, women should lower their hemoglobin A1c to below 6.5%.1
Thyroid measures. Autoimmune thyroid disease is the most common autoimmune disorder associated with diabetes and has been reported in 35% to 40% of women with type 1 diabetes.23 Recommendations are to check thyroid-stimulating hormone and thyroid peroxidase antibody levels before conception or early in pregnancy in all women with diabetes.1,24 Overt hypothyroidism should be treated before conception, given that early fetal brain development depends on maternal thyroxine.
Renal function testing. Preconception assessment of renal function is important for counseling and risk stratification. This assessment should include serum creatinine level, estimated glomerular filtration rate, and urinary albumin excretion.21
Celiac screening. Because women with type 1 diabetes are more susceptible to autoimmune diseases, they should be screened for celiac disease before conception, with testing for immunoglobulin A (IgA) and tissue transglutaminase antibodies, with or without IgA endomysial antibodies.16,25,26 An estimated 6% of patients with type 1 diabetes have celiac disease vs 1% of the general population.25 Celiac disease is 2 to 3 times more common in women, and asymptomatic people with type 1 diabetes are considered at increased risk for celiac disease.26
The association between type 1 diabetes and celiac disease most likely relates to the overlap in human leukocyte antigens of the diseases. There is no established link between type 2 diabetes and celiac disease.25
Undiagnosed celiac disease increases a woman’s risk of obstetric complications such as preterm birth, low birth weight, and stillbirth.26 The most likely explanation for these adverse effects is nutrient malabsorption, which is characteristic of celiac disease. Adherence to a gluten-free diet before and during gestation may reduce the risk of preterm delivery by as much as 20%.26
Vitamin B12 level. Celiac disease interferes with the absorption of vitamin B12-instrinsic factor in the ileum, which can lead to vitamin B12 deficiency. Therefore, baseline vitamin B12 levels should be checked before conception in women with celiac disease. Levels should also be checked in women taking metformin, which also decreases vitamin B12 absorption. Of note, increased folate levels due to taking supplements can potentially mask vitamin B12 deficiency.
MEDICATIONS TO REVIEW FOR PREGNANCY INTERACTIONS
Diabetic medications
Insulin is the first-line pharmacotherapy for pregnant patients with type 1, type 2, or gestational diabetes. Insulin does not cross the placenta to a measurable extent, and most insulin preparations have been classified as category B,1 meaning no risks to the fetus have been found in humans.
Insulin dosing during pregnancy is not static. Beginning around mid-gestation, insulin requirements increase,28,29 but after 32 weeks the need may decrease. These changes require practitioners to closely monitor blood glucose throughout pregnancy.
Both basal-bolus injections and continuous subcutaneous infusion are reasonable options during pregnancy.30 However, the need for multiple and potentially painful insulin injections daily can lead to poor compliance. This inconvenience has led to studies using oral hypoglycemic medications instead of insulin for patients with gestational and type 2 diabetes.
Metformin is an oral biguanide that decreases hepatic gluconeogenesis and intestinal glucose absorption while peripherally increasing glucose utilization and uptake. Metformin does not pose a risk of hypoglycemia because its mechanism of action does not involve increased insulin production.7
Metformin does cross the placenta, resulting in umbilical cord blood levels higher than maternal levels. Nevertheless, studies support the efficacy and short-term safety of metformin use during a pregnancy complicated by gestational or type 2 diabetes.7,31 Moreover, metformin has been associated with a lower risk of neonatal hypoglycemia and maternal weight gain than insulin.32 However, this agent should be used with caution, as long-term data are not yet available, and it may slightly increase the risk of premature delivery.
Glyburide is another oral hypoglycemic medication that has been used during pregnancy. This second-generation sulfonylurea enhances the release of insulin from the pancreas by binding beta islet cell ATP-calcium channel receptors. Compared with other sulfonylureas, glyburide has the lowest rate of maternal-to-fetal transfer, with umbilical cord plasma concentrations 70% of maternal levels.33 Although some trials support the efficacy and short-term safety of glyburide treatment for gestational diabetes,34 recent studies have associated glyburide use during pregnancy with a higher rate of neonatal hypoglycemia, neonatal respiratory distress, macrosomia, and neonatal intensive care unit admissions than insulin and metformin.1,35
Patients treated with oral agents should be informed that these drugs cross the placenta, and that although no adverse effects on the fetus have been demonstrated, long-term safety data are lacking. In addition, oral agents are ineffective in type 1 diabetes and may be insufficient to overcome the insulin resistance in type 2 diabetes.
Antihypertensive drugs
All antihypertensive drugs cross the placenta, but several have an acceptable safety profile in pregnancy, including methyldopa, labetalol, clonidine, prazosin, and nifedipine. Hydralazine and labetalol are short-acting, come in intravenous formulations, and can be used for urgent blood pressure control during pregnancy. Diltiazem may be used for heart rate control during pregnancy, and it has been shown to lower blood pressure and proteinuria in pregnant patients with underlying renal disease.36,37 The ADA recommends against chronic use of diuretics during pregnancy because of potential reductions in maternal plasma volume and uteroplacental perfusion.1
Angiotensin-converting enzyme (ACE) inhibitors, angiotensin II receptor blockers (ARBs), and direct renin inhibitors are contraindicated during pregnancy because of the risk of fetal defects, particularly in the renal system.21,38 Although there is evidence to question the association between first semester exposure and fetotoxicity,39 we avoid these drugs during pregnancy and switch to a different agent in women planning pregnancy.
Other drugs
Statins are contraindicated in pregnancy because they interfere with the development of the fetal nervous system.21 Although preliminary data from a small study did not identify safety risks associated with pravastatin use after 12 weeks of gestation,40 we recommend discontinuing statins in women attempting pregnancy.
Aspirin. The US Preventive Services Task Force41 recommends low-dose aspirin (81 mg/day) after 12 weeks of gestation for women with type 1 or type 2 diabetes, as well as those with renal disease or chronic hypertension, to prevent preeclampsia. Of note, higher doses need to be used with caution during pregnancy because fetal abnormalities have been reported, such as disruption of fetal vasculature (mesenteric vessels), gastroschisis, and small intestinal atresia.16
Folate supplementation (0.6–4 mg/day) is recommended in women with celiac disease to prevent neural tube defects in the offspring, and the US Preventive Services Task Force recommends 0.4 mg daily of folic acid supplementation for all women planning or capable of pregnancy.42–44 Higher doses, ranging from 0.6 to 5 mg/day, have been proposed for patients with diabetes,13 and we recommend at least 1 mg for this group, based on data suggesting that higher doses further reduce the risk of neural tube defects.43
IS BREASTFEEDING AFFECTED?
Maternal diabetes, insulin therapy, and oral hypoglycemic agents are not contraindications to breastfeeding. The US Preventive Services Task Force recommends interventions by primary care physicians to promote and support breastfeeding.45 Breastfeeding is encouraged based on various short- and long-term health benefits for both breastfed infants and breastfeeding mothers. Breastfeeding decreases a woman’s insulin requirements and increases the risk for hypoglycemia, especially in patients with insulin-dependent type 1 diabetes.1
Additionally, insulin sensitivity increases immediately following delivery of the placenta.1 Therefore, it is prudent to adjust insulin doses postpartum, especially while a patient is breastfeeding, or to suggest high-carbohydrate snacks before feeds.9,29
Antihypertensive drugs considered safe to use during lactation include captopril, enalapril, quinapril, labetalol, propranolol, nifedipine, and hydralazine.21,46 Methyldopa is not contraindicated, but it causes fatigue and worsened postpartum depression and should not be used as first-line therapy. Diuretics and ARBs are not recommended during lactation.21 Both metformin and glyburide enter breast milk in small enough amounts that they are not contraindicated during breastfeeding.16 The Lactmed database (www.toxnet.nlm.nih.gov) provides information about drugs and breastfeeding.
WHAT ABOUT CONTRACEPTIVES?
The ADA recommends contraception for women with diabetes because, just as in women without diabetes, the risks of unplanned pregnancy outweigh those of contraceptives.1
We recommend low-dose combination estrogen-progestin oral contraceptives to normotensive women under age 35 with diabetes but without underlying microvascular disease. For women over age 35 or for those with microvascular disease, additional options include intrauterine devices or progestin implants. We prefer not to use injectable depot medroxyprogesterone acetate because of its side effects of insulin resistance and weight gain.47
CASE DISCUSSION: NEXT STEPS
Our patient’s interest in family planning presents an opportunity for preconception counseling. We recommend a prenatal folic acid supplement, diet and regular exercise for weight loss, and screening tests including a comprehensive metabolic panel, hemoglobin A1c, thyroid-stimulating hormone, and dilated eye examination. We make sure she is up to date on her indicated health maintenance (eg, immunizations, disease screening), and we review her medications for potential teratogens. She denies any recreational drug use. Also, she has no plans for long-distance travel.
Our counseling includes discussions of pregnancy risks associated with pregestational diabetes and suboptimal glycemic control. We encourage her to use effective contraception until she is “medically optimized” for pregnancy—ie, until her hemoglobin A1c is lower than 6.5% and she has achieved a medically optimal weight. If feasible, a reduction of weight (7% or so) through lifestyle modification should be attempted, and if her hemoglobin A1c remains elevated, adding insulin would be recommended.
Pregnant patients or patients contemplating pregnancy are usually motivated to modify their behavior, making this a good time to reinforce lifestyle modifications. Many patients benefit from individualized counseling by a registered dietitian to help achieve the recommended weight and glycemic control.
Our physical examination in this patient includes screening for micro- and macrovascular complications of diabetes, and the test results are negative. Patients with active proliferative retinopathy should be referred to an ophthalmologist for assessment and treatment.
We review her medications for potential teratogenic effects and stop her ACE inhibitor (lisinopril) and statin (simvastatin). We switch her from a first-generation sulfonylurea (chlorpropamide) to glyburide, a second-generation sulfonylurea. Second-generation sulfonylureas are considered more “fetus-friendly” because first-generation sulfonylureas cross the placenta more easily and can cause fetal hyperinsulinemia, leading to macrosomia and neonatal hypoglycemia.7
The management of diabetes during pregnancy leans toward insulin use, given the lack of information regarding long-term outcomes with oral agents. If insulin is needed, it is best to initiate it before the patient conceives, and then to stop other diabetes medications. We would not make any changes to her aspirin or metformin use.
Educating the patient and her family about prevention, recognition, and treatment of hypoglycemia is important to prevent and manage the increased risk of hypoglycemia with insulin therapy and in early pregnancy.1 Consideration should be given to providing ketone strips as well as education on diabetic ketoacidosis prevention and detection.1 If the patient conceives, begin prenatal care early to allow adequate planning for care of her disease and evaluation of the fetus. Because of the complexity of insulin management in pregnancy, the ADA recommends referral, if possible, to a center offering team-based care, including an obstetrician specialized in high-risk pregnancies, an endocrinologist, and a dietitian.1
- American Diabetes Association. 13. Management of diabetes in pregnancy: standards of medical care in diabetes—2018. Diabetes Care 2018; 41(suppl 1):S137–S143. doi:10.2337/dc18-S013
- American Diabetes Association. 2. Classification and diagnosis of diabetes: standards of medical care in diabetes—2018. Diabetes Care 2018; 41(suppl 1):S13–S27. doi:10.2337/dc18-S002
- Lawler J, Osman M, Shelton JA, Yeh J. Population-based analysis of hypertensive disorders in pregnancy. Hypertens Pregnancy 2007; 26(1):67–76. doi:10.1080/10641950601147945
- Marchi J, Berg M, Dencker A, Olander EK, Begley C. Risks associated with obesity in pregnancy, for the mother and baby: a systematic review of reviews. Obes Rev 2015; 16(8):621–638. doi:10.1111/obr.12288
- Garrison EA, Jagasia S. Inpatient management of women with gestational and pregestational diabetes in pregnancy. Curr Diab Rep 2014; 14(2):457. doi:10.1007/s11892-013-0457-x
- Ballas J, Moore TR, Ramos GA. Management of diabetes in pregnancy. Curr Diab Rep 2012; 12(1):33–42. doi:10.1007/s11892-011-0249-0
- Ryu RJ, Hays KE, Hebert MF. Gestational diabetes mellitus management with oral hypoglycemic agents. Semin Perinatol 2014; 38(8):508–515. doi:10.1053/j.semperi.2014.08.012
- Cundy T, Gamble G, Neale L, et al. Differing causes of pregnancy loss in type 1 and type 2 diabetes. Diabetes Care 2007; 30(10):2603–2607. doi:10.2337/dc07-0555
- Castorino K, Jovanovic L. Pregnancy and diabetes management: advances and controversies. Clin Chem 2011; 57(2):221–230. doi:10.1373/clinchem.2010.155382
- Hammouda SA, Hakeem R. Role of HbA1c in predicting risk for congenital malformations. Prim Care Diabetes 2015; 9(6):458–464. doi:10.1016/j.pcd.2015.01.004
- Chen CP. Congenital malformations associated with maternal diabetes. Taiwanese J Obstet Gynecol 2005; 44(1):1–7. doi:10.1016/S1028-4559(09)60099-1
- International Association of Diabetes and Pregnancy Study Groups Consensus Panel, Metzger BE, Gabbe SG, Persson B, et al. International Association of Diabetes and Pregnancy study groups recommendations on the diagnosis and classification of hyperglycemia in pregnancy. Diabetes Care 2010; 33(3):676–682. doi:10.2337/dc09-1848
- Seaquist ER, Anderson J, Childs B, et al. Hypoglycemia and diabetes: a report of a workgroup of the American Diabetes Association and the Endocrine Society. Diabetes Care 2013; 36(5):1384–1395. doi:10.2337/dc12-2480
- HAPO Study Cooperative Research Group; Metzger BE, Lowe LP, Dyer AR, et al. Hyperglycemia and adverse pregnancy outcomes. N Engl J Med 2008; 358(19):1991–2002. doi:10.1056/NEJMoa0707943
- Finer LB, Zolna MR. Shifts in intended and unintended pregnancies in the United States, 2001–2008. Am J Public Health 2014; 104(suppl 1):S43–S48. doi:10.2105/AJPH.2013.301416
- Kitzmiller JL, Block JM, Brown FM, et al. Managing preexisting diabetes for pregnancy: summary of evidence and consensus recommendations for care. Diabetes Care 2008; 31(5):1060–1079. doi:10.2337/dc08-9020
- Webster LM, Conti-Ramsden F, Seed PT, Webb AJ, Nelson-Piercy C, Chappell LC. Impact of antihypertensive treatment on maternal and perinatal outcomes in pregnancy complicated by chronic hypertension: a systematic review and meta-analysis. J Am Heart Assoc 2017; 6(5).pii:e005526. doi:10.1161/JAHA.117.005526
- Chew EY, Mills JL, Metzger BE, et al. Metabolic control and progression of retinopathy: the Diabetes in Early Pregnancy Study. Diabetes Care 1995; 18(5):631–637. pmid:8586000
- American Diabetes Association. Standards of medical care in diabetes—2016. Diabetes Care 2016; 39 (suppl 1):S1–S109.
- Hawthorne, G. Maternal complications in diabetic pregnancy. Best Pract Res Clin Obstet Gynaecol 2011; 25(1):77–90. doi:10.1016/j.bpobgyn.2010.10.015
- Ringholm L, Damm JA, Vestgaard M, Damm P, Mathiesen ER. Diabetic nephropathy in women with preexisting diabetes: from pregnancy planning to breastfeeding. Curr Diab Rep 2016; 16(2):12. doi:10.1007/s11892-015-0705-3
- Zhang JJ, Ma XX, Hao L, Liu LJ, Lv JC, Zhang H. A systematic review and meta-analysis of outcomes of pregnancy in CKD and CKD outcomes in pregnancy. Clin J Am Soc Nephrol 2015; 10(11):1964–1978. doi:10.2215/CJN.09250914
- Umpierrez GE, Latif KA, Murphy MB, et al. Thyroid dysfunction in patients with type 1 diabetes: a longitudinal study. Diabetes Care 2003; 26(4):1181–1185. pmid:12663594
- Alexander EK, Pearce EN, Brent GA, et al. 2017 Guidelines of the American Thyroid Association for the Diagnosis and Management of Thyroid Disease During Pregnancy and the Postpartum. Thyroid 2017; 27(3):315–389. doi:10.1089/thy.2016.0457
- Akirov A, Pinhas-Hamiel O. Co-occurrence of type 1 diabetes mellitus and celiac disease. World J Diabetes 2015; 6(5):707–714. doi:10.4239/wjd.v6.i5.707
- Saccone G, Berghella V, Sarno L, et al. Celiac disease and obstetric complications: a systematic review and metaanalysis. Am J Obstet Gynecol 2016; 214(2):225–234. doi:10.1016/j.ajog.2015.09.080
- Feghali M, Venkataramanan R, Caritis S. Pharmacokinetics of drugs in pregnancy. Semin Perinatol 2015; 39(7):512–519. doi:10.1053/j.semperi.2015.08.003
- de Valk HW, Visser GH. Insulin during pregnancy, labour and delivery. Best Pract Res Clin Obstet Gynaecol 2011; 25(1):65–76. doi:10.1016/j.bpobgyn.2010.10.002
- Morello CM. Pharmacokinetics and pharmacodynamics of insulin analogs in special populations with type 2 diabetes mellitus. Int J Gen Med 2011; 4:827–835. doi:10.2147/IJGM.S26889
- Farrar D, Tuffnell DJ, West J, West HM. Continuous subcutaneous insulin infusion versus multiple daily injections of insulin for pregnant women with diabetes. Cochrane Database Syst Rev 2016; (6):CD005542. doi:10.1002/14651858.CD005542.pub2
- Charles B, Norris R, Xiao X, Hague W. Population pharmacokinetics of metformin in late pregnancy. Ther Drug Monit 2006; 28(1):67–72. pmid:16418696
- Balsells M, García-Patterson A, Solà I, Roqué M, Gich I, Corcoy R. Glibenclamide, metformin, and insulin for the treatment of gestational diabetes: a systematic review and meta-analysis. BMJ 2015; 350:h102. doi:10.1136/bmj.h102
- Hebert MF, Ma X, Naraharisetti SB, et al; Obstetric-Fetal Pharmacology Research Unit Network. Are we optimizing gestational diabetes treatment with glyburide? The pharmacologic basis for better clinical practice. Clin Pharmacol Ther 2009; 85(6):607–614. doi:10.1038/clpt.2009.5
- Langer O, Conway DL, Berkus MD, Xenakis EM, Gonzales O. A comparison of glyburide and insulin in women with gestational diabetes mellitus. N Engl J Med 2000; 343(16):1134–1138. doi:10.1056/NEJM200010193431601
- Camelo Castillo W, Boggess K, Stürmer T, Brookhart MA, Benjamin DK Jr, Jonsson Funk M. Association of adverse pregnancy outcomes with glyburide vs insulin in women with gestational diabetes. JAMA Pediatr 2015; 169:452–458. doi:10.1001/jamapediatrics.2015.74
- Gowda RM, Khan IA, Mehta NJ, Vasavada BC, Sacchi TJ. Cardiac arrhythmias in pregnancy: clinical and therapeutic considerations. Int J Cardiol 2003; 88(2):129–133. pmid:12714190
- Khandelwal M, Kumanova M, Gaughan JP, Reece EA. Role of diltiazem in pregnant women with chronic renal disease. J Matern Fetal Neonatal Med 2002; 12(6):408–412. doi:10.1080/jmf.12.6.408.412
- Magee LA, Abalos E, von Dadelszen P, Sibai B, Easterling T, Walkinshaw S; CHIPS Study Group. How to manage hypertension in pregnancy effectively. Br J Clin Pharmacol 2011; 72(3):394–401. doi:10.1111/j.1365-2125.2011.04002.x
- Cooper WO, Hernandez-Diaz S, Arbogast PG, et al. Major congenital malformations after first-trimester exposure to ACE inhibitors. N Engl J Med 2006; 354(23):2443–2451. doi:10.1056/NEJMoa055202
- Costantine MM, Cleary K, Hebert MF, et al; Eunice Kennedy Shriver National Institute of Child Health and Human Development Obstetric-Fetal Pharmacology Research Units Network. Safety and pharmacokinetics of pravastatin used for the prevention of preeclampsia in high-risk pregnant women: a pilot randomized controlled trial. Am J Obstet Gynecol 2016; 214(6):720.e1–720.e17. doi:10.1016/j.ajog.2015.12.038
- LeFevre ML; US Preventive Services Task Force. Low-dose aspirin use for the prevention of morbidity and mortality from preeclampsia: US Preventive Services Task Force recommendation statement. Ann Intern Med 2014; 161(11):819–826. doi:10.7326/M14-1884
- Curry SJ, Grossman DC, Whitlock EP, Cantu A. Behavioral counseling research and evidence-based practice recommendations: US Preventive Services Task Force perspectives. Ann Intern Med 2014; 160(6):407–413. doi:10.7326/M13-2128
- Wald N, Law M, Morris J, Wald D. Quantifying the effect of folic acid. Lancet 2001; 358(9298):2069–2073. pmid:11755633
- US Preventive Services Task Force; Bibbins-Domingo K, Grossman DC, Curry SJ, et al. Folic acid supplementation for the prevention of neural tube defects: US Preventive Services Task Force recommendation statement. JAMA 2017; 317(2):183–189. doi:10.1001/jama.2016.19438
- US Preventive Services Task Force; Bibbins-Domingo K, Grossman DC, Curry SJ, et al. Primary care interventions to support breastfeeding: US Preventive Services Task Force recommendation statement. JAMA 2016; 316(16):1688–1693. doi:10.1001/jama.2016.14697
- Newton ER, Hale TW. Drugs in breast milk. Clin Obstet Gynecol 2015; 58(4):868–884. doi:10.1097/GRF.0000000000000142
- Xiang AH, Kawakubo M, Kjos SL, Buchanan TA. Long-acting injectable progestin contraception and risk of type 2 diabetes in Latino women with prior gestational diabetes mellitus. Diabetes Care 2006; 29(3):613–617. pmid:16505515
A 29-year-old nulliparous woman presents for a routine checkup. She has hypertension and type 2 diabetes mellitus. Her current medications are chlorpropamide 500 mg daily, metformin 500 mg twice a day, lisinopril 40 mg daily, simvastatin 40 mg daily, and aspirin 81 mg daily. Her body mass index is 37 kg/m2 and her blood pressure is 130/80 mm Hg. Her hemoglobin A1c level is 7.8% and her low-density lipoprotein cholesterol 90 mg/dL.
She is considering pregnancy. How would you counsel her?
DEFINING DIABETES IN PREGNANCY
Diabetes in pregnant women, both gestational and pregestational, is the most common medical complication associated with pregnancy.1
- Gestational diabetes is defined as diabetes that is diagnosed during the second or third trimester of pregnancy and that is not clearly pregestational.2
- Pregestational diabetes exists before pregnancy and can be either type 1 or type 2.
Most cases of diabetes diagnosed during the first trimester reflect pregestational diabetes, as gestational diabetes occurs when insulin resistance increases in the later trimesters.
Type 1 diabetes involves autoimmune destruction of pancreatic islet cells, leading to insulin deficiency and the need for insulin therapy. Type 2 diabetes is characterized by insulin resistance rather than overall insulin deficiency. Type 2 diabetes tends to be associated with comorbidities such as obesity and hypertension, which are independent risk factors for adverse perinatal outcomes.3,4
Gestational diabetes accounts for most cases of diabetes during pregnancy. Although both pregestational and gestational diabetes increase the risk of maternal and fetal complications, pregestational diabetes is associated with significantly greater risks.1
IMPACT OF DIABETES ON THE MOTHER
Pregnancy increases the risk of maternal hypoglycemia, especially during the first trimester in patients with type 1 diabetes, as insulin sensitivity increases in early pregnancy.1 Pregnant women with diabetes may also have an altered counterregulatory response and less hypoglycemic awareness.1 Insulin resistance rises during the second and early third trimesters, increasing the risk of hyperglycemia in women with diabetes.1
Glycemic control during pregnancy is usually easier to achieve in patients with type 2 diabetes than with type 1, but it may require much higher insulin doses.
Because pregnancy is inherently a ketogenic state, women with type 1 diabetes are at higher risk of diabetic ketoacidosis, particularly during the second and third trimesters.1 There are reports of euglycemic diabetic ketoacidosis in pregnant women with either gestational or pregestational diabetes.5
Diabetes is associated with a risk of preeclampsia 4 times higher than in nondiabetic women.6 Other potential pregnancy-related complications include infections, polyhydramnios, spontaneous abortion, and cesarean delivery.1,7 The risk of pregnancy loss is similar in women with either type 1 or type 2 diabetes (2.6% and 3.7%, respectively), but the causes are different.8 Although preexisting diabetic complications such as retinopathy, nephropathy, and gastroparesis can be exacerbated during pregnancy,1 only severe gastroparesis and advanced renal disease are considered relative contraindications to pregnancy.
IMPACT OF DIABETES ON THE FETUS
Fetal complications of maternal diabetes include embryopathy (fetal malformations) and fetopathy (overgrowth, ie, fetus large for gestational age, and increased risk of fetal death or distress). Maternal hyperglycemia is associated with diabetic embryopathy, resulting in major birth defects in 5% to 25% of pregnancies and spontaneous abortions in 15% to 20%.9,10 There is a 2- to 6-fold increase in risk of congenital malformations.6
The most common diabetes-associated congenital malformations affect the cardiovascular system. Congenital heart disease includes tetralogy of Fallot, transposition of the great vessels, septal defects, and anomalous pulmonary venous return. Other relatively common defects involve the fetal central nervous system, spine, orofacial system, kidneys, urogenital system, gastrointestinal tract, and skeleton.11
The risk of fetopathy is proportional to the degree of maternal hyperglycemia. Excess maternal glucose and fatty acid levels can lead to fetal hyperglycemia and overgrowth, which increases fetal oxygen requirements. Erythropoietin levels rise, causing an increase in red cell mass, with subsequent hyperviscosity within the placenta and higher risk of fetal death.
Other complications include intrauterine growth restriction, prematurity, and preterm delivery. Fetal macrosomia (birth weight > 90th percentile or 4 kg, approximately 8 lb, 13 oz) occurs in 27% to 62% of children born to mothers with diabetes, a rate 10 times higher than in patients without diabetes. It contributes to shoulder dystocia (risk 2 to 4 times higher in diabetic pregnancies) and cesarean delivery.6 Infants born to mothers with diabetes also have higher risks of neonatal hypoglycemia, erythrocytosis, hyperbilirubinemia, hypocalcemia, respiratory distress, cardiomyopathy, and death, as well as for developing diabetes, obesity, and other adverse cardiometabolic outcomes later in life.11
GET GLUCOSE UNDER CONTROL BEFORE PREGNANCY
Nearly half of pregnancies in the general population are unplanned,15 so preconception diabetes assessment needs to be part of routine medical care for all reproductive-age women. Because most organogenesis occurs during the first 5 to 8 weeks after fertilization—potentially before a woman realizes she is pregnant—achieving optimal glycemic control before conception is necessary to improve pregnancy outcomes.1
EVERY VISIT IS AN OPPORTUNITY
Every medical visit with a reproductive-age woman with diabetes is an opportunity for counseling about pregnancy. Topics that need to be discussed include the risks of unplanned pregnancy and of poor metabolic control, and the benefits of improved maternal and fetal outcomes with appropriate pregnancy planning and diabetes management.
Referral to a registered dietitian for individualized counseling about proper nutrition, particularly during pregnancy, has been associated with positive outcomes.16 Patients with diabetes and at high risk of pregnancy complications should be referred to a clinic that specializes in high-risk pregnancies.
Practitioners also should emphasize the importance of regular exercise and encourage patients to maintain or achieve a medically optimal weight before conception. Ideally, this would be a normal body mass index; however, this is not always possible.
In women who are planning pregnancy or are not on effective contraception, medications should be reviewed for potential teratogenicity. If needed, discuss alternative medications or switch to safer ones. However, these changes should not interrupt diabetes treatment.
In addition, ensure that the patient is up to date on age- and disease-appropriate preventive care (eg, immunizations, screening for sexually transmitted disease and malignancy). Counseling and intervention for use of tobacco, alcohol, and recreational drugs are also important. As with any preconception counseling, the patient (and her partner, if possible) should be advised to avoid travel to areas where Zika virus is endemic, and informed about the availability of expanded carrier genetic screening through her obstetric provider.
Finally, pregnant women with diabetes benefit from screening for diabetic complications including hypertension, retinopathy, cardiovascular disease, neuropathy, and nephropathy.
ASSESSING RISKS
Blood pressure
Chronic (preexisting) hypertension is defined as a systolic pressure 140 mm Hg or higher or a diastolic pressure 90 mm Hg or higher, or both, that antedates pregnancy or is present before the 20th week of pregnancy.3 Chronic hypertension has been reported in up to 5% of pregnant women and is associated with increased risk of preterm delivery, superimposed preeclampsia, low birth weight, and perinatal death.3
Reproductive-age women with diabetes and high blood pressure benefit from lifestyle and behavioral modifications.17 If drug therapy is needed, antihypertensive drugs that are safe for the fetus should be used. Treatment of mild or moderate hypertension during pregnancy reduces the risk of progression to severe hypertension but may not improve obstetric outcomes.
Diabetic retinopathy
Diabetic retinopathy can significantly worsen during pregnancy: the risk of progression is double that in the nonpregnant state.18 Women with diabetes who are contemplating pregnancy should have a comprehensive eye examination before conception, and any active proliferative retinopathy needs to be treated. These patients may require ophthalmologic monitoring and treatment during pregnancy. (Note: laser photocoagulation is not contraindicated during pregnancy.)
Cardiovascular disease
Cardiovascular physiology changes dramatically during pregnancy. Cardiovascular disease, especially when superimposed on diabetes, can increase the risk of maternal death. Thus, evaluation for cardiovascular risk factors as well as cardiovascular system integrity before conception is important. Listen for arterial bruits and murmurs, and assess peripheral pulses. Consideration should be given to obtaining a preconception resting electrocardiogram in women with diabetes who are over age 35 or who are suspected of having cardiovascular disease.16
Neurologic disorders
Peripheral neuropathy, the most common neurologic complication of diabetes, is associated with injury and infection.19
Autonomic neuropathy is associated with decreased cardiac responsiveness and orthostatic hypotension.19 Diabetic gastroparesis alone can precipitate serious complications during pregnancy, including extreme hypoglycemia and hyperglycemia, increased risk of diabetic ketoacidosis, weight loss, malnutrition, frequent hospitalizations, and increased requirement for parenteral nutrition.20
Although diabetic neuropathy does not significantly worsen during pregnancy, women with preexisting gastroparesis should be counseled on the substantial risks associated with pregnancy. Screening for neuropathy should be part of all diabetic preconception examinations.
Renal complications
Pregnancy in women with diabetes and preexisting renal dysfunction increases their risk of accelerated progression of diabetic kidney disease.21 Preexisting renal dysfunction also increases the risk of pregnancy-related complications, such as stillbirth, intrauterine growth restriction, gestational hypertension, preeclampsia, and preterm delivery.19,21,22 Further, the risk of pregnancy complications correlates directly with the severity of renal dysfunction.22
Psychiatric disorders
Emotional wellness is essential for optimal diabetes management. It is important to recognize the emotional impact of diabetes in pregnant women and to conduct routine screening for depression, anxiety, stress, and eating disorders.16
LABORATORY TESTS TO CONSIDER
Hemoglobin A1c. The general consensus is to achieve the lowest hemoglobin A1c level possible that does not increase the risk of hypoglycemia. The American Diabetes Association (ADA) recommends that, before attempting to conceive, women should lower their hemoglobin A1c to below 6.5%.1
Thyroid measures. Autoimmune thyroid disease is the most common autoimmune disorder associated with diabetes and has been reported in 35% to 40% of women with type 1 diabetes.23 Recommendations are to check thyroid-stimulating hormone and thyroid peroxidase antibody levels before conception or early in pregnancy in all women with diabetes.1,24 Overt hypothyroidism should be treated before conception, given that early fetal brain development depends on maternal thyroxine.
Renal function testing. Preconception assessment of renal function is important for counseling and risk stratification. This assessment should include serum creatinine level, estimated glomerular filtration rate, and urinary albumin excretion.21
Celiac screening. Because women with type 1 diabetes are more susceptible to autoimmune diseases, they should be screened for celiac disease before conception, with testing for immunoglobulin A (IgA) and tissue transglutaminase antibodies, with or without IgA endomysial antibodies.16,25,26 An estimated 6% of patients with type 1 diabetes have celiac disease vs 1% of the general population.25 Celiac disease is 2 to 3 times more common in women, and asymptomatic people with type 1 diabetes are considered at increased risk for celiac disease.26
The association between type 1 diabetes and celiac disease most likely relates to the overlap in human leukocyte antigens of the diseases. There is no established link between type 2 diabetes and celiac disease.25
Undiagnosed celiac disease increases a woman’s risk of obstetric complications such as preterm birth, low birth weight, and stillbirth.26 The most likely explanation for these adverse effects is nutrient malabsorption, which is characteristic of celiac disease. Adherence to a gluten-free diet before and during gestation may reduce the risk of preterm delivery by as much as 20%.26
Vitamin B12 level. Celiac disease interferes with the absorption of vitamin B12-instrinsic factor in the ileum, which can lead to vitamin B12 deficiency. Therefore, baseline vitamin B12 levels should be checked before conception in women with celiac disease. Levels should also be checked in women taking metformin, which also decreases vitamin B12 absorption. Of note, increased folate levels due to taking supplements can potentially mask vitamin B12 deficiency.
MEDICATIONS TO REVIEW FOR PREGNANCY INTERACTIONS
Diabetic medications
Insulin is the first-line pharmacotherapy for pregnant patients with type 1, type 2, or gestational diabetes. Insulin does not cross the placenta to a measurable extent, and most insulin preparations have been classified as category B,1 meaning no risks to the fetus have been found in humans.
Insulin dosing during pregnancy is not static. Beginning around mid-gestation, insulin requirements increase,28,29 but after 32 weeks the need may decrease. These changes require practitioners to closely monitor blood glucose throughout pregnancy.
Both basal-bolus injections and continuous subcutaneous infusion are reasonable options during pregnancy.30 However, the need for multiple and potentially painful insulin injections daily can lead to poor compliance. This inconvenience has led to studies using oral hypoglycemic medications instead of insulin for patients with gestational and type 2 diabetes.
Metformin is an oral biguanide that decreases hepatic gluconeogenesis and intestinal glucose absorption while peripherally increasing glucose utilization and uptake. Metformin does not pose a risk of hypoglycemia because its mechanism of action does not involve increased insulin production.7
Metformin does cross the placenta, resulting in umbilical cord blood levels higher than maternal levels. Nevertheless, studies support the efficacy and short-term safety of metformin use during a pregnancy complicated by gestational or type 2 diabetes.7,31 Moreover, metformin has been associated with a lower risk of neonatal hypoglycemia and maternal weight gain than insulin.32 However, this agent should be used with caution, as long-term data are not yet available, and it may slightly increase the risk of premature delivery.
Glyburide is another oral hypoglycemic medication that has been used during pregnancy. This second-generation sulfonylurea enhances the release of insulin from the pancreas by binding beta islet cell ATP-calcium channel receptors. Compared with other sulfonylureas, glyburide has the lowest rate of maternal-to-fetal transfer, with umbilical cord plasma concentrations 70% of maternal levels.33 Although some trials support the efficacy and short-term safety of glyburide treatment for gestational diabetes,34 recent studies have associated glyburide use during pregnancy with a higher rate of neonatal hypoglycemia, neonatal respiratory distress, macrosomia, and neonatal intensive care unit admissions than insulin and metformin.1,35
Patients treated with oral agents should be informed that these drugs cross the placenta, and that although no adverse effects on the fetus have been demonstrated, long-term safety data are lacking. In addition, oral agents are ineffective in type 1 diabetes and may be insufficient to overcome the insulin resistance in type 2 diabetes.
Antihypertensive drugs
All antihypertensive drugs cross the placenta, but several have an acceptable safety profile in pregnancy, including methyldopa, labetalol, clonidine, prazosin, and nifedipine. Hydralazine and labetalol are short-acting, come in intravenous formulations, and can be used for urgent blood pressure control during pregnancy. Diltiazem may be used for heart rate control during pregnancy, and it has been shown to lower blood pressure and proteinuria in pregnant patients with underlying renal disease.36,37 The ADA recommends against chronic use of diuretics during pregnancy because of potential reductions in maternal plasma volume and uteroplacental perfusion.1
Angiotensin-converting enzyme (ACE) inhibitors, angiotensin II receptor blockers (ARBs), and direct renin inhibitors are contraindicated during pregnancy because of the risk of fetal defects, particularly in the renal system.21,38 Although there is evidence to question the association between first semester exposure and fetotoxicity,39 we avoid these drugs during pregnancy and switch to a different agent in women planning pregnancy.
Other drugs
Statins are contraindicated in pregnancy because they interfere with the development of the fetal nervous system.21 Although preliminary data from a small study did not identify safety risks associated with pravastatin use after 12 weeks of gestation,40 we recommend discontinuing statins in women attempting pregnancy.
Aspirin. The US Preventive Services Task Force41 recommends low-dose aspirin (81 mg/day) after 12 weeks of gestation for women with type 1 or type 2 diabetes, as well as those with renal disease or chronic hypertension, to prevent preeclampsia. Of note, higher doses need to be used with caution during pregnancy because fetal abnormalities have been reported, such as disruption of fetal vasculature (mesenteric vessels), gastroschisis, and small intestinal atresia.16
Folate supplementation (0.6–4 mg/day) is recommended in women with celiac disease to prevent neural tube defects in the offspring, and the US Preventive Services Task Force recommends 0.4 mg daily of folic acid supplementation for all women planning or capable of pregnancy.42–44 Higher doses, ranging from 0.6 to 5 mg/day, have been proposed for patients with diabetes,13 and we recommend at least 1 mg for this group, based on data suggesting that higher doses further reduce the risk of neural tube defects.43
IS BREASTFEEDING AFFECTED?
Maternal diabetes, insulin therapy, and oral hypoglycemic agents are not contraindications to breastfeeding. The US Preventive Services Task Force recommends interventions by primary care physicians to promote and support breastfeeding.45 Breastfeeding is encouraged based on various short- and long-term health benefits for both breastfed infants and breastfeeding mothers. Breastfeeding decreases a woman’s insulin requirements and increases the risk for hypoglycemia, especially in patients with insulin-dependent type 1 diabetes.1
Additionally, insulin sensitivity increases immediately following delivery of the placenta.1 Therefore, it is prudent to adjust insulin doses postpartum, especially while a patient is breastfeeding, or to suggest high-carbohydrate snacks before feeds.9,29
Antihypertensive drugs considered safe to use during lactation include captopril, enalapril, quinapril, labetalol, propranolol, nifedipine, and hydralazine.21,46 Methyldopa is not contraindicated, but it causes fatigue and worsened postpartum depression and should not be used as first-line therapy. Diuretics and ARBs are not recommended during lactation.21 Both metformin and glyburide enter breast milk in small enough amounts that they are not contraindicated during breastfeeding.16 The Lactmed database (www.toxnet.nlm.nih.gov) provides information about drugs and breastfeeding.
WHAT ABOUT CONTRACEPTIVES?
The ADA recommends contraception for women with diabetes because, just as in women without diabetes, the risks of unplanned pregnancy outweigh those of contraceptives.1
We recommend low-dose combination estrogen-progestin oral contraceptives to normotensive women under age 35 with diabetes but without underlying microvascular disease. For women over age 35 or for those with microvascular disease, additional options include intrauterine devices or progestin implants. We prefer not to use injectable depot medroxyprogesterone acetate because of its side effects of insulin resistance and weight gain.47
CASE DISCUSSION: NEXT STEPS
Our patient’s interest in family planning presents an opportunity for preconception counseling. We recommend a prenatal folic acid supplement, diet and regular exercise for weight loss, and screening tests including a comprehensive metabolic panel, hemoglobin A1c, thyroid-stimulating hormone, and dilated eye examination. We make sure she is up to date on her indicated health maintenance (eg, immunizations, disease screening), and we review her medications for potential teratogens. She denies any recreational drug use. Also, she has no plans for long-distance travel.
Our counseling includes discussions of pregnancy risks associated with pregestational diabetes and suboptimal glycemic control. We encourage her to use effective contraception until she is “medically optimized” for pregnancy—ie, until her hemoglobin A1c is lower than 6.5% and she has achieved a medically optimal weight. If feasible, a reduction of weight (7% or so) through lifestyle modification should be attempted, and if her hemoglobin A1c remains elevated, adding insulin would be recommended.
Pregnant patients or patients contemplating pregnancy are usually motivated to modify their behavior, making this a good time to reinforce lifestyle modifications. Many patients benefit from individualized counseling by a registered dietitian to help achieve the recommended weight and glycemic control.
Our physical examination in this patient includes screening for micro- and macrovascular complications of diabetes, and the test results are negative. Patients with active proliferative retinopathy should be referred to an ophthalmologist for assessment and treatment.
We review her medications for potential teratogenic effects and stop her ACE inhibitor (lisinopril) and statin (simvastatin). We switch her from a first-generation sulfonylurea (chlorpropamide) to glyburide, a second-generation sulfonylurea. Second-generation sulfonylureas are considered more “fetus-friendly” because first-generation sulfonylureas cross the placenta more easily and can cause fetal hyperinsulinemia, leading to macrosomia and neonatal hypoglycemia.7
The management of diabetes during pregnancy leans toward insulin use, given the lack of information regarding long-term outcomes with oral agents. If insulin is needed, it is best to initiate it before the patient conceives, and then to stop other diabetes medications. We would not make any changes to her aspirin or metformin use.
Educating the patient and her family about prevention, recognition, and treatment of hypoglycemia is important to prevent and manage the increased risk of hypoglycemia with insulin therapy and in early pregnancy.1 Consideration should be given to providing ketone strips as well as education on diabetic ketoacidosis prevention and detection.1 If the patient conceives, begin prenatal care early to allow adequate planning for care of her disease and evaluation of the fetus. Because of the complexity of insulin management in pregnancy, the ADA recommends referral, if possible, to a center offering team-based care, including an obstetrician specialized in high-risk pregnancies, an endocrinologist, and a dietitian.1
A 29-year-old nulliparous woman presents for a routine checkup. She has hypertension and type 2 diabetes mellitus. Her current medications are chlorpropamide 500 mg daily, metformin 500 mg twice a day, lisinopril 40 mg daily, simvastatin 40 mg daily, and aspirin 81 mg daily. Her body mass index is 37 kg/m2 and her blood pressure is 130/80 mm Hg. Her hemoglobin A1c level is 7.8% and her low-density lipoprotein cholesterol 90 mg/dL.
She is considering pregnancy. How would you counsel her?
DEFINING DIABETES IN PREGNANCY
Diabetes in pregnant women, both gestational and pregestational, is the most common medical complication associated with pregnancy.1
- Gestational diabetes is defined as diabetes that is diagnosed during the second or third trimester of pregnancy and that is not clearly pregestational.2
- Pregestational diabetes exists before pregnancy and can be either type 1 or type 2.
Most cases of diabetes diagnosed during the first trimester reflect pregestational diabetes, as gestational diabetes occurs when insulin resistance increases in the later trimesters.
Type 1 diabetes involves autoimmune destruction of pancreatic islet cells, leading to insulin deficiency and the need for insulin therapy. Type 2 diabetes is characterized by insulin resistance rather than overall insulin deficiency. Type 2 diabetes tends to be associated with comorbidities such as obesity and hypertension, which are independent risk factors for adverse perinatal outcomes.3,4
Gestational diabetes accounts for most cases of diabetes during pregnancy. Although both pregestational and gestational diabetes increase the risk of maternal and fetal complications, pregestational diabetes is associated with significantly greater risks.1
IMPACT OF DIABETES ON THE MOTHER
Pregnancy increases the risk of maternal hypoglycemia, especially during the first trimester in patients with type 1 diabetes, as insulin sensitivity increases in early pregnancy.1 Pregnant women with diabetes may also have an altered counterregulatory response and less hypoglycemic awareness.1 Insulin resistance rises during the second and early third trimesters, increasing the risk of hyperglycemia in women with diabetes.1
Glycemic control during pregnancy is usually easier to achieve in patients with type 2 diabetes than with type 1, but it may require much higher insulin doses.
Because pregnancy is inherently a ketogenic state, women with type 1 diabetes are at higher risk of diabetic ketoacidosis, particularly during the second and third trimesters.1 There are reports of euglycemic diabetic ketoacidosis in pregnant women with either gestational or pregestational diabetes.5
Diabetes is associated with a risk of preeclampsia 4 times higher than in nondiabetic women.6 Other potential pregnancy-related complications include infections, polyhydramnios, spontaneous abortion, and cesarean delivery.1,7 The risk of pregnancy loss is similar in women with either type 1 or type 2 diabetes (2.6% and 3.7%, respectively), but the causes are different.8 Although preexisting diabetic complications such as retinopathy, nephropathy, and gastroparesis can be exacerbated during pregnancy,1 only severe gastroparesis and advanced renal disease are considered relative contraindications to pregnancy.
IMPACT OF DIABETES ON THE FETUS
Fetal complications of maternal diabetes include embryopathy (fetal malformations) and fetopathy (overgrowth, ie, fetus large for gestational age, and increased risk of fetal death or distress). Maternal hyperglycemia is associated with diabetic embryopathy, resulting in major birth defects in 5% to 25% of pregnancies and spontaneous abortions in 15% to 20%.9,10 There is a 2- to 6-fold increase in risk of congenital malformations.6
The most common diabetes-associated congenital malformations affect the cardiovascular system. Congenital heart disease includes tetralogy of Fallot, transposition of the great vessels, septal defects, and anomalous pulmonary venous return. Other relatively common defects involve the fetal central nervous system, spine, orofacial system, kidneys, urogenital system, gastrointestinal tract, and skeleton.11
The risk of fetopathy is proportional to the degree of maternal hyperglycemia. Excess maternal glucose and fatty acid levels can lead to fetal hyperglycemia and overgrowth, which increases fetal oxygen requirements. Erythropoietin levels rise, causing an increase in red cell mass, with subsequent hyperviscosity within the placenta and higher risk of fetal death.
Other complications include intrauterine growth restriction, prematurity, and preterm delivery. Fetal macrosomia (birth weight > 90th percentile or 4 kg, approximately 8 lb, 13 oz) occurs in 27% to 62% of children born to mothers with diabetes, a rate 10 times higher than in patients without diabetes. It contributes to shoulder dystocia (risk 2 to 4 times higher in diabetic pregnancies) and cesarean delivery.6 Infants born to mothers with diabetes also have higher risks of neonatal hypoglycemia, erythrocytosis, hyperbilirubinemia, hypocalcemia, respiratory distress, cardiomyopathy, and death, as well as for developing diabetes, obesity, and other adverse cardiometabolic outcomes later in life.11
GET GLUCOSE UNDER CONTROL BEFORE PREGNANCY
Nearly half of pregnancies in the general population are unplanned,15 so preconception diabetes assessment needs to be part of routine medical care for all reproductive-age women. Because most organogenesis occurs during the first 5 to 8 weeks after fertilization—potentially before a woman realizes she is pregnant—achieving optimal glycemic control before conception is necessary to improve pregnancy outcomes.1
EVERY VISIT IS AN OPPORTUNITY
Every medical visit with a reproductive-age woman with diabetes is an opportunity for counseling about pregnancy. Topics that need to be discussed include the risks of unplanned pregnancy and of poor metabolic control, and the benefits of improved maternal and fetal outcomes with appropriate pregnancy planning and diabetes management.
Referral to a registered dietitian for individualized counseling about proper nutrition, particularly during pregnancy, has been associated with positive outcomes.16 Patients with diabetes and at high risk of pregnancy complications should be referred to a clinic that specializes in high-risk pregnancies.
Practitioners also should emphasize the importance of regular exercise and encourage patients to maintain or achieve a medically optimal weight before conception. Ideally, this would be a normal body mass index; however, this is not always possible.
In women who are planning pregnancy or are not on effective contraception, medications should be reviewed for potential teratogenicity. If needed, discuss alternative medications or switch to safer ones. However, these changes should not interrupt diabetes treatment.
In addition, ensure that the patient is up to date on age- and disease-appropriate preventive care (eg, immunizations, screening for sexually transmitted disease and malignancy). Counseling and intervention for use of tobacco, alcohol, and recreational drugs are also important. As with any preconception counseling, the patient (and her partner, if possible) should be advised to avoid travel to areas where Zika virus is endemic, and informed about the availability of expanded carrier genetic screening through her obstetric provider.
Finally, pregnant women with diabetes benefit from screening for diabetic complications including hypertension, retinopathy, cardiovascular disease, neuropathy, and nephropathy.
ASSESSING RISKS
Blood pressure
Chronic (preexisting) hypertension is defined as a systolic pressure 140 mm Hg or higher or a diastolic pressure 90 mm Hg or higher, or both, that antedates pregnancy or is present before the 20th week of pregnancy.3 Chronic hypertension has been reported in up to 5% of pregnant women and is associated with increased risk of preterm delivery, superimposed preeclampsia, low birth weight, and perinatal death.3
Reproductive-age women with diabetes and high blood pressure benefit from lifestyle and behavioral modifications.17 If drug therapy is needed, antihypertensive drugs that are safe for the fetus should be used. Treatment of mild or moderate hypertension during pregnancy reduces the risk of progression to severe hypertension but may not improve obstetric outcomes.
Diabetic retinopathy
Diabetic retinopathy can significantly worsen during pregnancy: the risk of progression is double that in the nonpregnant state.18 Women with diabetes who are contemplating pregnancy should have a comprehensive eye examination before conception, and any active proliferative retinopathy needs to be treated. These patients may require ophthalmologic monitoring and treatment during pregnancy. (Note: laser photocoagulation is not contraindicated during pregnancy.)
Cardiovascular disease
Cardiovascular physiology changes dramatically during pregnancy. Cardiovascular disease, especially when superimposed on diabetes, can increase the risk of maternal death. Thus, evaluation for cardiovascular risk factors as well as cardiovascular system integrity before conception is important. Listen for arterial bruits and murmurs, and assess peripheral pulses. Consideration should be given to obtaining a preconception resting electrocardiogram in women with diabetes who are over age 35 or who are suspected of having cardiovascular disease.16
Neurologic disorders
Peripheral neuropathy, the most common neurologic complication of diabetes, is associated with injury and infection.19
Autonomic neuropathy is associated with decreased cardiac responsiveness and orthostatic hypotension.19 Diabetic gastroparesis alone can precipitate serious complications during pregnancy, including extreme hypoglycemia and hyperglycemia, increased risk of diabetic ketoacidosis, weight loss, malnutrition, frequent hospitalizations, and increased requirement for parenteral nutrition.20
Although diabetic neuropathy does not significantly worsen during pregnancy, women with preexisting gastroparesis should be counseled on the substantial risks associated with pregnancy. Screening for neuropathy should be part of all diabetic preconception examinations.
Renal complications
Pregnancy in women with diabetes and preexisting renal dysfunction increases their risk of accelerated progression of diabetic kidney disease.21 Preexisting renal dysfunction also increases the risk of pregnancy-related complications, such as stillbirth, intrauterine growth restriction, gestational hypertension, preeclampsia, and preterm delivery.19,21,22 Further, the risk of pregnancy complications correlates directly with the severity of renal dysfunction.22
Psychiatric disorders
Emotional wellness is essential for optimal diabetes management. It is important to recognize the emotional impact of diabetes in pregnant women and to conduct routine screening for depression, anxiety, stress, and eating disorders.16
LABORATORY TESTS TO CONSIDER
Hemoglobin A1c. The general consensus is to achieve the lowest hemoglobin A1c level possible that does not increase the risk of hypoglycemia. The American Diabetes Association (ADA) recommends that, before attempting to conceive, women should lower their hemoglobin A1c to below 6.5%.1
Thyroid measures. Autoimmune thyroid disease is the most common autoimmune disorder associated with diabetes and has been reported in 35% to 40% of women with type 1 diabetes.23 Recommendations are to check thyroid-stimulating hormone and thyroid peroxidase antibody levels before conception or early in pregnancy in all women with diabetes.1,24 Overt hypothyroidism should be treated before conception, given that early fetal brain development depends on maternal thyroxine.
Renal function testing. Preconception assessment of renal function is important for counseling and risk stratification. This assessment should include serum creatinine level, estimated glomerular filtration rate, and urinary albumin excretion.21
Celiac screening. Because women with type 1 diabetes are more susceptible to autoimmune diseases, they should be screened for celiac disease before conception, with testing for immunoglobulin A (IgA) and tissue transglutaminase antibodies, with or without IgA endomysial antibodies.16,25,26 An estimated 6% of patients with type 1 diabetes have celiac disease vs 1% of the general population.25 Celiac disease is 2 to 3 times more common in women, and asymptomatic people with type 1 diabetes are considered at increased risk for celiac disease.26
The association between type 1 diabetes and celiac disease most likely relates to the overlap in human leukocyte antigens of the diseases. There is no established link between type 2 diabetes and celiac disease.25
Undiagnosed celiac disease increases a woman’s risk of obstetric complications such as preterm birth, low birth weight, and stillbirth.26 The most likely explanation for these adverse effects is nutrient malabsorption, which is characteristic of celiac disease. Adherence to a gluten-free diet before and during gestation may reduce the risk of preterm delivery by as much as 20%.26
Vitamin B12 level. Celiac disease interferes with the absorption of vitamin B12-instrinsic factor in the ileum, which can lead to vitamin B12 deficiency. Therefore, baseline vitamin B12 levels should be checked before conception in women with celiac disease. Levels should also be checked in women taking metformin, which also decreases vitamin B12 absorption. Of note, increased folate levels due to taking supplements can potentially mask vitamin B12 deficiency.
MEDICATIONS TO REVIEW FOR PREGNANCY INTERACTIONS
Diabetic medications
Insulin is the first-line pharmacotherapy for pregnant patients with type 1, type 2, or gestational diabetes. Insulin does not cross the placenta to a measurable extent, and most insulin preparations have been classified as category B,1 meaning no risks to the fetus have been found in humans.
Insulin dosing during pregnancy is not static. Beginning around mid-gestation, insulin requirements increase,28,29 but after 32 weeks the need may decrease. These changes require practitioners to closely monitor blood glucose throughout pregnancy.
Both basal-bolus injections and continuous subcutaneous infusion are reasonable options during pregnancy.30 However, the need for multiple and potentially painful insulin injections daily can lead to poor compliance. This inconvenience has led to studies using oral hypoglycemic medications instead of insulin for patients with gestational and type 2 diabetes.
Metformin is an oral biguanide that decreases hepatic gluconeogenesis and intestinal glucose absorption while peripherally increasing glucose utilization and uptake. Metformin does not pose a risk of hypoglycemia because its mechanism of action does not involve increased insulin production.7
Metformin does cross the placenta, resulting in umbilical cord blood levels higher than maternal levels. Nevertheless, studies support the efficacy and short-term safety of metformin use during a pregnancy complicated by gestational or type 2 diabetes.7,31 Moreover, metformin has been associated with a lower risk of neonatal hypoglycemia and maternal weight gain than insulin.32 However, this agent should be used with caution, as long-term data are not yet available, and it may slightly increase the risk of premature delivery.
Glyburide is another oral hypoglycemic medication that has been used during pregnancy. This second-generation sulfonylurea enhances the release of insulin from the pancreas by binding beta islet cell ATP-calcium channel receptors. Compared with other sulfonylureas, glyburide has the lowest rate of maternal-to-fetal transfer, with umbilical cord plasma concentrations 70% of maternal levels.33 Although some trials support the efficacy and short-term safety of glyburide treatment for gestational diabetes,34 recent studies have associated glyburide use during pregnancy with a higher rate of neonatal hypoglycemia, neonatal respiratory distress, macrosomia, and neonatal intensive care unit admissions than insulin and metformin.1,35
Patients treated with oral agents should be informed that these drugs cross the placenta, and that although no adverse effects on the fetus have been demonstrated, long-term safety data are lacking. In addition, oral agents are ineffective in type 1 diabetes and may be insufficient to overcome the insulin resistance in type 2 diabetes.
Antihypertensive drugs
All antihypertensive drugs cross the placenta, but several have an acceptable safety profile in pregnancy, including methyldopa, labetalol, clonidine, prazosin, and nifedipine. Hydralazine and labetalol are short-acting, come in intravenous formulations, and can be used for urgent blood pressure control during pregnancy. Diltiazem may be used for heart rate control during pregnancy, and it has been shown to lower blood pressure and proteinuria in pregnant patients with underlying renal disease.36,37 The ADA recommends against chronic use of diuretics during pregnancy because of potential reductions in maternal plasma volume and uteroplacental perfusion.1
Angiotensin-converting enzyme (ACE) inhibitors, angiotensin II receptor blockers (ARBs), and direct renin inhibitors are contraindicated during pregnancy because of the risk of fetal defects, particularly in the renal system.21,38 Although there is evidence to question the association between first semester exposure and fetotoxicity,39 we avoid these drugs during pregnancy and switch to a different agent in women planning pregnancy.
Other drugs
Statins are contraindicated in pregnancy because they interfere with the development of the fetal nervous system.21 Although preliminary data from a small study did not identify safety risks associated with pravastatin use after 12 weeks of gestation,40 we recommend discontinuing statins in women attempting pregnancy.
Aspirin. The US Preventive Services Task Force41 recommends low-dose aspirin (81 mg/day) after 12 weeks of gestation for women with type 1 or type 2 diabetes, as well as those with renal disease or chronic hypertension, to prevent preeclampsia. Of note, higher doses need to be used with caution during pregnancy because fetal abnormalities have been reported, such as disruption of fetal vasculature (mesenteric vessels), gastroschisis, and small intestinal atresia.16
Folate supplementation (0.6–4 mg/day) is recommended in women with celiac disease to prevent neural tube defects in the offspring, and the US Preventive Services Task Force recommends 0.4 mg daily of folic acid supplementation for all women planning or capable of pregnancy.42–44 Higher doses, ranging from 0.6 to 5 mg/day, have been proposed for patients with diabetes,13 and we recommend at least 1 mg for this group, based on data suggesting that higher doses further reduce the risk of neural tube defects.43
IS BREASTFEEDING AFFECTED?
Maternal diabetes, insulin therapy, and oral hypoglycemic agents are not contraindications to breastfeeding. The US Preventive Services Task Force recommends interventions by primary care physicians to promote and support breastfeeding.45 Breastfeeding is encouraged based on various short- and long-term health benefits for both breastfed infants and breastfeeding mothers. Breastfeeding decreases a woman’s insulin requirements and increases the risk for hypoglycemia, especially in patients with insulin-dependent type 1 diabetes.1
Additionally, insulin sensitivity increases immediately following delivery of the placenta.1 Therefore, it is prudent to adjust insulin doses postpartum, especially while a patient is breastfeeding, or to suggest high-carbohydrate snacks before feeds.9,29
Antihypertensive drugs considered safe to use during lactation include captopril, enalapril, quinapril, labetalol, propranolol, nifedipine, and hydralazine.21,46 Methyldopa is not contraindicated, but it causes fatigue and worsened postpartum depression and should not be used as first-line therapy. Diuretics and ARBs are not recommended during lactation.21 Both metformin and glyburide enter breast milk in small enough amounts that they are not contraindicated during breastfeeding.16 The Lactmed database (www.toxnet.nlm.nih.gov) provides information about drugs and breastfeeding.
WHAT ABOUT CONTRACEPTIVES?
The ADA recommends contraception for women with diabetes because, just as in women without diabetes, the risks of unplanned pregnancy outweigh those of contraceptives.1
We recommend low-dose combination estrogen-progestin oral contraceptives to normotensive women under age 35 with diabetes but without underlying microvascular disease. For women over age 35 or for those with microvascular disease, additional options include intrauterine devices or progestin implants. We prefer not to use injectable depot medroxyprogesterone acetate because of its side effects of insulin resistance and weight gain.47
CASE DISCUSSION: NEXT STEPS
Our patient’s interest in family planning presents an opportunity for preconception counseling. We recommend a prenatal folic acid supplement, diet and regular exercise for weight loss, and screening tests including a comprehensive metabolic panel, hemoglobin A1c, thyroid-stimulating hormone, and dilated eye examination. We make sure she is up to date on her indicated health maintenance (eg, immunizations, disease screening), and we review her medications for potential teratogens. She denies any recreational drug use. Also, she has no plans for long-distance travel.
Our counseling includes discussions of pregnancy risks associated with pregestational diabetes and suboptimal glycemic control. We encourage her to use effective contraception until she is “medically optimized” for pregnancy—ie, until her hemoglobin A1c is lower than 6.5% and she has achieved a medically optimal weight. If feasible, a reduction of weight (7% or so) through lifestyle modification should be attempted, and if her hemoglobin A1c remains elevated, adding insulin would be recommended.
Pregnant patients or patients contemplating pregnancy are usually motivated to modify their behavior, making this a good time to reinforce lifestyle modifications. Many patients benefit from individualized counseling by a registered dietitian to help achieve the recommended weight and glycemic control.
Our physical examination in this patient includes screening for micro- and macrovascular complications of diabetes, and the test results are negative. Patients with active proliferative retinopathy should be referred to an ophthalmologist for assessment and treatment.
We review her medications for potential teratogenic effects and stop her ACE inhibitor (lisinopril) and statin (simvastatin). We switch her from a first-generation sulfonylurea (chlorpropamide) to glyburide, a second-generation sulfonylurea. Second-generation sulfonylureas are considered more “fetus-friendly” because first-generation sulfonylureas cross the placenta more easily and can cause fetal hyperinsulinemia, leading to macrosomia and neonatal hypoglycemia.7
The management of diabetes during pregnancy leans toward insulin use, given the lack of information regarding long-term outcomes with oral agents. If insulin is needed, it is best to initiate it before the patient conceives, and then to stop other diabetes medications. We would not make any changes to her aspirin or metformin use.
Educating the patient and her family about prevention, recognition, and treatment of hypoglycemia is important to prevent and manage the increased risk of hypoglycemia with insulin therapy and in early pregnancy.1 Consideration should be given to providing ketone strips as well as education on diabetic ketoacidosis prevention and detection.1 If the patient conceives, begin prenatal care early to allow adequate planning for care of her disease and evaluation of the fetus. Because of the complexity of insulin management in pregnancy, the ADA recommends referral, if possible, to a center offering team-based care, including an obstetrician specialized in high-risk pregnancies, an endocrinologist, and a dietitian.1
- American Diabetes Association. 13. Management of diabetes in pregnancy: standards of medical care in diabetes—2018. Diabetes Care 2018; 41(suppl 1):S137–S143. doi:10.2337/dc18-S013
- American Diabetes Association. 2. Classification and diagnosis of diabetes: standards of medical care in diabetes—2018. Diabetes Care 2018; 41(suppl 1):S13–S27. doi:10.2337/dc18-S002
- Lawler J, Osman M, Shelton JA, Yeh J. Population-based analysis of hypertensive disorders in pregnancy. Hypertens Pregnancy 2007; 26(1):67–76. doi:10.1080/10641950601147945
- Marchi J, Berg M, Dencker A, Olander EK, Begley C. Risks associated with obesity in pregnancy, for the mother and baby: a systematic review of reviews. Obes Rev 2015; 16(8):621–638. doi:10.1111/obr.12288
- Garrison EA, Jagasia S. Inpatient management of women with gestational and pregestational diabetes in pregnancy. Curr Diab Rep 2014; 14(2):457. doi:10.1007/s11892-013-0457-x
- Ballas J, Moore TR, Ramos GA. Management of diabetes in pregnancy. Curr Diab Rep 2012; 12(1):33–42. doi:10.1007/s11892-011-0249-0
- Ryu RJ, Hays KE, Hebert MF. Gestational diabetes mellitus management with oral hypoglycemic agents. Semin Perinatol 2014; 38(8):508–515. doi:10.1053/j.semperi.2014.08.012
- Cundy T, Gamble G, Neale L, et al. Differing causes of pregnancy loss in type 1 and type 2 diabetes. Diabetes Care 2007; 30(10):2603–2607. doi:10.2337/dc07-0555
- Castorino K, Jovanovic L. Pregnancy and diabetes management: advances and controversies. Clin Chem 2011; 57(2):221–230. doi:10.1373/clinchem.2010.155382
- Hammouda SA, Hakeem R. Role of HbA1c in predicting risk for congenital malformations. Prim Care Diabetes 2015; 9(6):458–464. doi:10.1016/j.pcd.2015.01.004
- Chen CP. Congenital malformations associated with maternal diabetes. Taiwanese J Obstet Gynecol 2005; 44(1):1–7. doi:10.1016/S1028-4559(09)60099-1
- International Association of Diabetes and Pregnancy Study Groups Consensus Panel, Metzger BE, Gabbe SG, Persson B, et al. International Association of Diabetes and Pregnancy study groups recommendations on the diagnosis and classification of hyperglycemia in pregnancy. Diabetes Care 2010; 33(3):676–682. doi:10.2337/dc09-1848
- Seaquist ER, Anderson J, Childs B, et al. Hypoglycemia and diabetes: a report of a workgroup of the American Diabetes Association and the Endocrine Society. Diabetes Care 2013; 36(5):1384–1395. doi:10.2337/dc12-2480
- HAPO Study Cooperative Research Group; Metzger BE, Lowe LP, Dyer AR, et al. Hyperglycemia and adverse pregnancy outcomes. N Engl J Med 2008; 358(19):1991–2002. doi:10.1056/NEJMoa0707943
- Finer LB, Zolna MR. Shifts in intended and unintended pregnancies in the United States, 2001–2008. Am J Public Health 2014; 104(suppl 1):S43–S48. doi:10.2105/AJPH.2013.301416
- Kitzmiller JL, Block JM, Brown FM, et al. Managing preexisting diabetes for pregnancy: summary of evidence and consensus recommendations for care. Diabetes Care 2008; 31(5):1060–1079. doi:10.2337/dc08-9020
- Webster LM, Conti-Ramsden F, Seed PT, Webb AJ, Nelson-Piercy C, Chappell LC. Impact of antihypertensive treatment on maternal and perinatal outcomes in pregnancy complicated by chronic hypertension: a systematic review and meta-analysis. J Am Heart Assoc 2017; 6(5).pii:e005526. doi:10.1161/JAHA.117.005526
- Chew EY, Mills JL, Metzger BE, et al. Metabolic control and progression of retinopathy: the Diabetes in Early Pregnancy Study. Diabetes Care 1995; 18(5):631–637. pmid:8586000
- American Diabetes Association. Standards of medical care in diabetes—2016. Diabetes Care 2016; 39 (suppl 1):S1–S109.
- Hawthorne, G. Maternal complications in diabetic pregnancy. Best Pract Res Clin Obstet Gynaecol 2011; 25(1):77–90. doi:10.1016/j.bpobgyn.2010.10.015
- Ringholm L, Damm JA, Vestgaard M, Damm P, Mathiesen ER. Diabetic nephropathy in women with preexisting diabetes: from pregnancy planning to breastfeeding. Curr Diab Rep 2016; 16(2):12. doi:10.1007/s11892-015-0705-3
- Zhang JJ, Ma XX, Hao L, Liu LJ, Lv JC, Zhang H. A systematic review and meta-analysis of outcomes of pregnancy in CKD and CKD outcomes in pregnancy. Clin J Am Soc Nephrol 2015; 10(11):1964–1978. doi:10.2215/CJN.09250914
- Umpierrez GE, Latif KA, Murphy MB, et al. Thyroid dysfunction in patients with type 1 diabetes: a longitudinal study. Diabetes Care 2003; 26(4):1181–1185. pmid:12663594
- Alexander EK, Pearce EN, Brent GA, et al. 2017 Guidelines of the American Thyroid Association for the Diagnosis and Management of Thyroid Disease During Pregnancy and the Postpartum. Thyroid 2017; 27(3):315–389. doi:10.1089/thy.2016.0457
- Akirov A, Pinhas-Hamiel O. Co-occurrence of type 1 diabetes mellitus and celiac disease. World J Diabetes 2015; 6(5):707–714. doi:10.4239/wjd.v6.i5.707
- Saccone G, Berghella V, Sarno L, et al. Celiac disease and obstetric complications: a systematic review and metaanalysis. Am J Obstet Gynecol 2016; 214(2):225–234. doi:10.1016/j.ajog.2015.09.080
- Feghali M, Venkataramanan R, Caritis S. Pharmacokinetics of drugs in pregnancy. Semin Perinatol 2015; 39(7):512–519. doi:10.1053/j.semperi.2015.08.003
- de Valk HW, Visser GH. Insulin during pregnancy, labour and delivery. Best Pract Res Clin Obstet Gynaecol 2011; 25(1):65–76. doi:10.1016/j.bpobgyn.2010.10.002
- Morello CM. Pharmacokinetics and pharmacodynamics of insulin analogs in special populations with type 2 diabetes mellitus. Int J Gen Med 2011; 4:827–835. doi:10.2147/IJGM.S26889
- Farrar D, Tuffnell DJ, West J, West HM. Continuous subcutaneous insulin infusion versus multiple daily injections of insulin for pregnant women with diabetes. Cochrane Database Syst Rev 2016; (6):CD005542. doi:10.1002/14651858.CD005542.pub2
- Charles B, Norris R, Xiao X, Hague W. Population pharmacokinetics of metformin in late pregnancy. Ther Drug Monit 2006; 28(1):67–72. pmid:16418696
- Balsells M, García-Patterson A, Solà I, Roqué M, Gich I, Corcoy R. Glibenclamide, metformin, and insulin for the treatment of gestational diabetes: a systematic review and meta-analysis. BMJ 2015; 350:h102. doi:10.1136/bmj.h102
- Hebert MF, Ma X, Naraharisetti SB, et al; Obstetric-Fetal Pharmacology Research Unit Network. Are we optimizing gestational diabetes treatment with glyburide? The pharmacologic basis for better clinical practice. Clin Pharmacol Ther 2009; 85(6):607–614. doi:10.1038/clpt.2009.5
- Langer O, Conway DL, Berkus MD, Xenakis EM, Gonzales O. A comparison of glyburide and insulin in women with gestational diabetes mellitus. N Engl J Med 2000; 343(16):1134–1138. doi:10.1056/NEJM200010193431601
- Camelo Castillo W, Boggess K, Stürmer T, Brookhart MA, Benjamin DK Jr, Jonsson Funk M. Association of adverse pregnancy outcomes with glyburide vs insulin in women with gestational diabetes. JAMA Pediatr 2015; 169:452–458. doi:10.1001/jamapediatrics.2015.74
- Gowda RM, Khan IA, Mehta NJ, Vasavada BC, Sacchi TJ. Cardiac arrhythmias in pregnancy: clinical and therapeutic considerations. Int J Cardiol 2003; 88(2):129–133. pmid:12714190
- Khandelwal M, Kumanova M, Gaughan JP, Reece EA. Role of diltiazem in pregnant women with chronic renal disease. J Matern Fetal Neonatal Med 2002; 12(6):408–412. doi:10.1080/jmf.12.6.408.412
- Magee LA, Abalos E, von Dadelszen P, Sibai B, Easterling T, Walkinshaw S; CHIPS Study Group. How to manage hypertension in pregnancy effectively. Br J Clin Pharmacol 2011; 72(3):394–401. doi:10.1111/j.1365-2125.2011.04002.x
- Cooper WO, Hernandez-Diaz S, Arbogast PG, et al. Major congenital malformations after first-trimester exposure to ACE inhibitors. N Engl J Med 2006; 354(23):2443–2451. doi:10.1056/NEJMoa055202
- Costantine MM, Cleary K, Hebert MF, et al; Eunice Kennedy Shriver National Institute of Child Health and Human Development Obstetric-Fetal Pharmacology Research Units Network. Safety and pharmacokinetics of pravastatin used for the prevention of preeclampsia in high-risk pregnant women: a pilot randomized controlled trial. Am J Obstet Gynecol 2016; 214(6):720.e1–720.e17. doi:10.1016/j.ajog.2015.12.038
- LeFevre ML; US Preventive Services Task Force. Low-dose aspirin use for the prevention of morbidity and mortality from preeclampsia: US Preventive Services Task Force recommendation statement. Ann Intern Med 2014; 161(11):819–826. doi:10.7326/M14-1884
- Curry SJ, Grossman DC, Whitlock EP, Cantu A. Behavioral counseling research and evidence-based practice recommendations: US Preventive Services Task Force perspectives. Ann Intern Med 2014; 160(6):407–413. doi:10.7326/M13-2128
- Wald N, Law M, Morris J, Wald D. Quantifying the effect of folic acid. Lancet 2001; 358(9298):2069–2073. pmid:11755633
- US Preventive Services Task Force; Bibbins-Domingo K, Grossman DC, Curry SJ, et al. Folic acid supplementation for the prevention of neural tube defects: US Preventive Services Task Force recommendation statement. JAMA 2017; 317(2):183–189. doi:10.1001/jama.2016.19438
- US Preventive Services Task Force; Bibbins-Domingo K, Grossman DC, Curry SJ, et al. Primary care interventions to support breastfeeding: US Preventive Services Task Force recommendation statement. JAMA 2016; 316(16):1688–1693. doi:10.1001/jama.2016.14697
- Newton ER, Hale TW. Drugs in breast milk. Clin Obstet Gynecol 2015; 58(4):868–884. doi:10.1097/GRF.0000000000000142
- Xiang AH, Kawakubo M, Kjos SL, Buchanan TA. Long-acting injectable progestin contraception and risk of type 2 diabetes in Latino women with prior gestational diabetes mellitus. Diabetes Care 2006; 29(3):613–617. pmid:16505515
- American Diabetes Association. 13. Management of diabetes in pregnancy: standards of medical care in diabetes—2018. Diabetes Care 2018; 41(suppl 1):S137–S143. doi:10.2337/dc18-S013
- American Diabetes Association. 2. Classification and diagnosis of diabetes: standards of medical care in diabetes—2018. Diabetes Care 2018; 41(suppl 1):S13–S27. doi:10.2337/dc18-S002
- Lawler J, Osman M, Shelton JA, Yeh J. Population-based analysis of hypertensive disorders in pregnancy. Hypertens Pregnancy 2007; 26(1):67–76. doi:10.1080/10641950601147945
- Marchi J, Berg M, Dencker A, Olander EK, Begley C. Risks associated with obesity in pregnancy, for the mother and baby: a systematic review of reviews. Obes Rev 2015; 16(8):621–638. doi:10.1111/obr.12288
- Garrison EA, Jagasia S. Inpatient management of women with gestational and pregestational diabetes in pregnancy. Curr Diab Rep 2014; 14(2):457. doi:10.1007/s11892-013-0457-x
- Ballas J, Moore TR, Ramos GA. Management of diabetes in pregnancy. Curr Diab Rep 2012; 12(1):33–42. doi:10.1007/s11892-011-0249-0
- Ryu RJ, Hays KE, Hebert MF. Gestational diabetes mellitus management with oral hypoglycemic agents. Semin Perinatol 2014; 38(8):508–515. doi:10.1053/j.semperi.2014.08.012
- Cundy T, Gamble G, Neale L, et al. Differing causes of pregnancy loss in type 1 and type 2 diabetes. Diabetes Care 2007; 30(10):2603–2607. doi:10.2337/dc07-0555
- Castorino K, Jovanovic L. Pregnancy and diabetes management: advances and controversies. Clin Chem 2011; 57(2):221–230. doi:10.1373/clinchem.2010.155382
- Hammouda SA, Hakeem R. Role of HbA1c in predicting risk for congenital malformations. Prim Care Diabetes 2015; 9(6):458–464. doi:10.1016/j.pcd.2015.01.004
- Chen CP. Congenital malformations associated with maternal diabetes. Taiwanese J Obstet Gynecol 2005; 44(1):1–7. doi:10.1016/S1028-4559(09)60099-1
- International Association of Diabetes and Pregnancy Study Groups Consensus Panel, Metzger BE, Gabbe SG, Persson B, et al. International Association of Diabetes and Pregnancy study groups recommendations on the diagnosis and classification of hyperglycemia in pregnancy. Diabetes Care 2010; 33(3):676–682. doi:10.2337/dc09-1848
- Seaquist ER, Anderson J, Childs B, et al. Hypoglycemia and diabetes: a report of a workgroup of the American Diabetes Association and the Endocrine Society. Diabetes Care 2013; 36(5):1384–1395. doi:10.2337/dc12-2480
- HAPO Study Cooperative Research Group; Metzger BE, Lowe LP, Dyer AR, et al. Hyperglycemia and adverse pregnancy outcomes. N Engl J Med 2008; 358(19):1991–2002. doi:10.1056/NEJMoa0707943
- Finer LB, Zolna MR. Shifts in intended and unintended pregnancies in the United States, 2001–2008. Am J Public Health 2014; 104(suppl 1):S43–S48. doi:10.2105/AJPH.2013.301416
- Kitzmiller JL, Block JM, Brown FM, et al. Managing preexisting diabetes for pregnancy: summary of evidence and consensus recommendations for care. Diabetes Care 2008; 31(5):1060–1079. doi:10.2337/dc08-9020
- Webster LM, Conti-Ramsden F, Seed PT, Webb AJ, Nelson-Piercy C, Chappell LC. Impact of antihypertensive treatment on maternal and perinatal outcomes in pregnancy complicated by chronic hypertension: a systematic review and meta-analysis. J Am Heart Assoc 2017; 6(5).pii:e005526. doi:10.1161/JAHA.117.005526
- Chew EY, Mills JL, Metzger BE, et al. Metabolic control and progression of retinopathy: the Diabetes in Early Pregnancy Study. Diabetes Care 1995; 18(5):631–637. pmid:8586000
- American Diabetes Association. Standards of medical care in diabetes—2016. Diabetes Care 2016; 39 (suppl 1):S1–S109.
- Hawthorne, G. Maternal complications in diabetic pregnancy. Best Pract Res Clin Obstet Gynaecol 2011; 25(1):77–90. doi:10.1016/j.bpobgyn.2010.10.015
- Ringholm L, Damm JA, Vestgaard M, Damm P, Mathiesen ER. Diabetic nephropathy in women with preexisting diabetes: from pregnancy planning to breastfeeding. Curr Diab Rep 2016; 16(2):12. doi:10.1007/s11892-015-0705-3
- Zhang JJ, Ma XX, Hao L, Liu LJ, Lv JC, Zhang H. A systematic review and meta-analysis of outcomes of pregnancy in CKD and CKD outcomes in pregnancy. Clin J Am Soc Nephrol 2015; 10(11):1964–1978. doi:10.2215/CJN.09250914
- Umpierrez GE, Latif KA, Murphy MB, et al. Thyroid dysfunction in patients with type 1 diabetes: a longitudinal study. Diabetes Care 2003; 26(4):1181–1185. pmid:12663594
- Alexander EK, Pearce EN, Brent GA, et al. 2017 Guidelines of the American Thyroid Association for the Diagnosis and Management of Thyroid Disease During Pregnancy and the Postpartum. Thyroid 2017; 27(3):315–389. doi:10.1089/thy.2016.0457
- Akirov A, Pinhas-Hamiel O. Co-occurrence of type 1 diabetes mellitus and celiac disease. World J Diabetes 2015; 6(5):707–714. doi:10.4239/wjd.v6.i5.707
- Saccone G, Berghella V, Sarno L, et al. Celiac disease and obstetric complications: a systematic review and metaanalysis. Am J Obstet Gynecol 2016; 214(2):225–234. doi:10.1016/j.ajog.2015.09.080
- Feghali M, Venkataramanan R, Caritis S. Pharmacokinetics of drugs in pregnancy. Semin Perinatol 2015; 39(7):512–519. doi:10.1053/j.semperi.2015.08.003
- de Valk HW, Visser GH. Insulin during pregnancy, labour and delivery. Best Pract Res Clin Obstet Gynaecol 2011; 25(1):65–76. doi:10.1016/j.bpobgyn.2010.10.002
- Morello CM. Pharmacokinetics and pharmacodynamics of insulin analogs in special populations with type 2 diabetes mellitus. Int J Gen Med 2011; 4:827–835. doi:10.2147/IJGM.S26889
- Farrar D, Tuffnell DJ, West J, West HM. Continuous subcutaneous insulin infusion versus multiple daily injections of insulin for pregnant women with diabetes. Cochrane Database Syst Rev 2016; (6):CD005542. doi:10.1002/14651858.CD005542.pub2
- Charles B, Norris R, Xiao X, Hague W. Population pharmacokinetics of metformin in late pregnancy. Ther Drug Monit 2006; 28(1):67–72. pmid:16418696
- Balsells M, García-Patterson A, Solà I, Roqué M, Gich I, Corcoy R. Glibenclamide, metformin, and insulin for the treatment of gestational diabetes: a systematic review and meta-analysis. BMJ 2015; 350:h102. doi:10.1136/bmj.h102
- Hebert MF, Ma X, Naraharisetti SB, et al; Obstetric-Fetal Pharmacology Research Unit Network. Are we optimizing gestational diabetes treatment with glyburide? The pharmacologic basis for better clinical practice. Clin Pharmacol Ther 2009; 85(6):607–614. doi:10.1038/clpt.2009.5
- Langer O, Conway DL, Berkus MD, Xenakis EM, Gonzales O. A comparison of glyburide and insulin in women with gestational diabetes mellitus. N Engl J Med 2000; 343(16):1134–1138. doi:10.1056/NEJM200010193431601
- Camelo Castillo W, Boggess K, Stürmer T, Brookhart MA, Benjamin DK Jr, Jonsson Funk M. Association of adverse pregnancy outcomes with glyburide vs insulin in women with gestational diabetes. JAMA Pediatr 2015; 169:452–458. doi:10.1001/jamapediatrics.2015.74
- Gowda RM, Khan IA, Mehta NJ, Vasavada BC, Sacchi TJ. Cardiac arrhythmias in pregnancy: clinical and therapeutic considerations. Int J Cardiol 2003; 88(2):129–133. pmid:12714190
- Khandelwal M, Kumanova M, Gaughan JP, Reece EA. Role of diltiazem in pregnant women with chronic renal disease. J Matern Fetal Neonatal Med 2002; 12(6):408–412. doi:10.1080/jmf.12.6.408.412
- Magee LA, Abalos E, von Dadelszen P, Sibai B, Easterling T, Walkinshaw S; CHIPS Study Group. How to manage hypertension in pregnancy effectively. Br J Clin Pharmacol 2011; 72(3):394–401. doi:10.1111/j.1365-2125.2011.04002.x
- Cooper WO, Hernandez-Diaz S, Arbogast PG, et al. Major congenital malformations after first-trimester exposure to ACE inhibitors. N Engl J Med 2006; 354(23):2443–2451. doi:10.1056/NEJMoa055202
- Costantine MM, Cleary K, Hebert MF, et al; Eunice Kennedy Shriver National Institute of Child Health and Human Development Obstetric-Fetal Pharmacology Research Units Network. Safety and pharmacokinetics of pravastatin used for the prevention of preeclampsia in high-risk pregnant women: a pilot randomized controlled trial. Am J Obstet Gynecol 2016; 214(6):720.e1–720.e17. doi:10.1016/j.ajog.2015.12.038
- LeFevre ML; US Preventive Services Task Force. Low-dose aspirin use for the prevention of morbidity and mortality from preeclampsia: US Preventive Services Task Force recommendation statement. Ann Intern Med 2014; 161(11):819–826. doi:10.7326/M14-1884
- Curry SJ, Grossman DC, Whitlock EP, Cantu A. Behavioral counseling research and evidence-based practice recommendations: US Preventive Services Task Force perspectives. Ann Intern Med 2014; 160(6):407–413. doi:10.7326/M13-2128
- Wald N, Law M, Morris J, Wald D. Quantifying the effect of folic acid. Lancet 2001; 358(9298):2069–2073. pmid:11755633
- US Preventive Services Task Force; Bibbins-Domingo K, Grossman DC, Curry SJ, et al. Folic acid supplementation for the prevention of neural tube defects: US Preventive Services Task Force recommendation statement. JAMA 2017; 317(2):183–189. doi:10.1001/jama.2016.19438
- US Preventive Services Task Force; Bibbins-Domingo K, Grossman DC, Curry SJ, et al. Primary care interventions to support breastfeeding: US Preventive Services Task Force recommendation statement. JAMA 2016; 316(16):1688–1693. doi:10.1001/jama.2016.14697
- Newton ER, Hale TW. Drugs in breast milk. Clin Obstet Gynecol 2015; 58(4):868–884. doi:10.1097/GRF.0000000000000142
- Xiang AH, Kawakubo M, Kjos SL, Buchanan TA. Long-acting injectable progestin contraception and risk of type 2 diabetes in Latino women with prior gestational diabetes mellitus. Diabetes Care 2006; 29(3):613–617. pmid:16505515
KEY POINTS
- Aim for a hemoglobin A1c of 6.5% or lower, if it is attainable without increasing the risk of hypoglycemia.
- Avoid teratogenic drugs in sexually active women of childbearing age unless the patient uses effective contraception.
- Because about half of pregnancies are unplanned, it is important to routinely discuss family planning and provide preconception counseling that includes reducing risks associated with pregnancy.
- Screen for diabetic end-organ damage, especially retinopathy and nephropathy.
13 weeks' gestation • heart palpitations • chest tightness • Dx?
THE CASE
A 29-year-old G1P0 woman at 13 weeks’ gestation came in for a routine prenatal visit complaining of sudden-onset heart palpitations that were occurring about once a week. Each episode lasted between 15 and 60 minutes and was accompanied by chest tightness, with no identifiable cause. The patient could inconsistently terminate the episodes with Valsalva maneuvers. She reported having had 2 similar incidents of palpitations within the past year. Her family history was significant for sudden cardiac death of her father and paternal grandfather in their fifth decades of life.
A cardiovascular exam was normal; heart auscultation revealed a regular rate and rhythm without murmurs, rubs, or gallops, and the peripheral pulses were normal. A thyroid-stimulating hormone (TSH) level, basic metabolic panel (BMP), and complete blood count (CBC) were within normal limits. A transthoracic echocardiogram was negative for structural heart disease.
THE DIAGNOSIS
An initial Holter monitor study failed to capture an episode of her palpitations. The frequency of her palpitations increased as her pregnancy progressed, occurring almost daily by the second half of the third trimester, and a repeat Holter monitor study in the third trimester was significant for a 3-minute episode of supraventricular tachycardia (SVT) that correlated with patient-recorded symptoms (FIGURE).
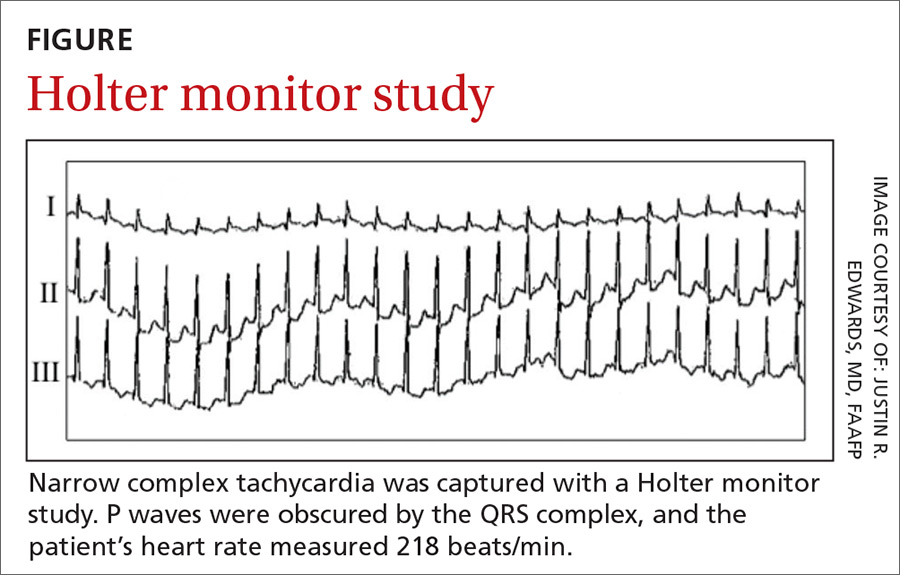
Based on these results, we diagnosed the patient with an atrioventricular nodal reentry tachycardia (AVNRT). Although atrioventricular reciprocating tachycardia (AVRT) remained a remote possibility, it is far less common, and a 12-lead electrocardiogram (EKG) showed no evidence of pre-excitation.
DISCUSSION
AVNRT is the most common form of paroxysmal supraventricular tachycardia (PSVT). It occurs more frequently in women and typically manifests in the second to fourth decades of life.1 AVNRT is a narrow complex tachycardia characterized by a heart rate of 120 to >200 beats/min.
Hemodynamic changes in pregnancy can trigger arrhythmias
During pregnancy, hemodynamic changes (including increased blood volume and cardiac output) are thought to stimulate stretch-activated ion channels within the walls of the heart.2-4 Such changes may exacerbate previously existing cardiac arrhythmias or (less commonly) cause new-onset arrhythmias.3,4 A family history positive for arrhythmias or sudden cardiac death increases the likelihood of developing tachyarrhythmia during pregnancy.3 Women with a known history of PSVT might experience symptom exacerbation despite being on prophylactic therapy.4
Detection and diagnosis
While AVNRT is relatively benign in pregnancy, other cardiac arrhythmias (eg, atrial fibrillation/flutter, ventricular tachycardia) carry a greater risk for fetal and maternal complications, underscoring the need to correctly identify the type of arrhythmia.2,3
Continue to: Physical exam findings
Physical exam findings are often unremarkable unless the patient is actively experiencing SVT in the office, in which case prominent jugular pulsations may be seen due to simultaneous contraction of the atria and ventricles.
The initial evaluation of a pregnant patient presenting with tachycardia should include a BMP, TSH, 12-lead EKG, and transthoracic echocardiography.3,5 In most patients with AVNRT, the results of these tests will be normal. A Holter monitor can be used to document an arrhythmia if the episodes are relatively frequent or an event monitor can be used if the episodes are infrequent.5
EKG findings. When patients are actively experiencing SVT, EKG findings include a P wave obscured by the QRS complex, sometimes manifesting as a pseudo-R wave in the V1 lead and a pseudo-S wave in leads II, III, and AVF. The QRS complex is narrow and the R-R interval is regular.6
Types of treatment
Valsalva maneuvers. Treatment of AVNRT in pregnancy should first involve addressing any precipitating causes, including metabolic and endocrine abnormalities.3 As virtually all antiarrhythmic drugs cross the placenta and are traceable in breast milk,2,3 patients should be counseled to try to stop episodes using Valsalva maneuvers before moving to pharmacologic treatment.
Antiarrhythmics. First-line pharmacologic treatment for the prevention of AVNRT in pregnancy is metoprolol or verapamil.2,5 Neither drug has been associated with adverse outcomes in infants, although there is a large body of evidence suggesting that low levels of metoprolol are present in breast milk.7
Continue to: Acute episodes of SVT that are refractory to...
Acute episodes of SVT that are refractory to vagal maneuvers or occur despite medical management can be treated acutely in pregnancy with adenosine, which effectively stops episodes about 90% of the time.2 (See the TABLE8,9 for a list of antiarrhythmics that may be used to treat AVNRT.)
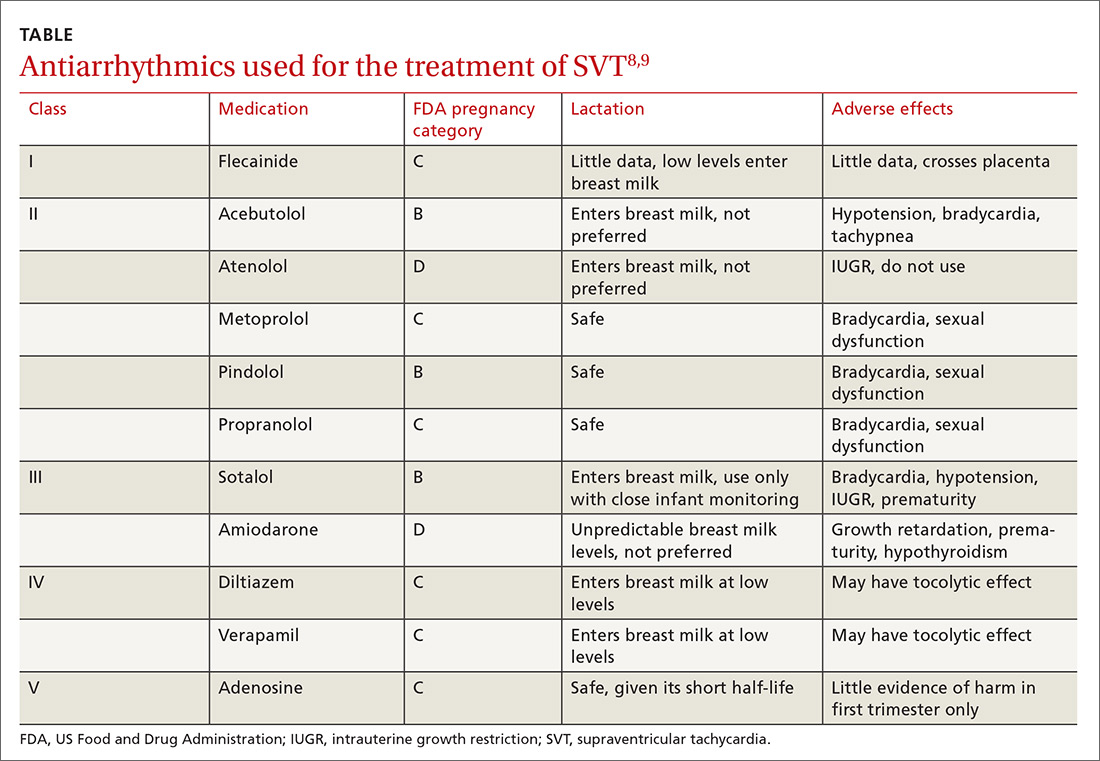
Catheter ablation is first-line treatment for AVNRT in nonpregnant patients.1,5 The risks of undergoing ablation during pregnancy include fetal exposure to radiation and anesthetic drugs.2,3 Therefore, this treatment should be used only when pharmacologic treatment is unsuccessful and risks to the mother and fetus due to the arrhythmia outweigh the risks of the procedure. Ablation can be offered postpartum as more definitive therapy.
Our patient was started on metoprolol tartrate 12.5 mg bid at 35 weeks’ gestation due to increasingly common and persistent palpitations. This helped control the episodes for 2 weeks, at which point they increased again in frequency. These were terminated using Valsalva maneuvers; increasing the metoprolol dosage was prohibitive due to patient intolerance.
Following an uncomplicated delivery, and discontinuation of metoprolol, the patient reported a decrease in both the number of episodes and the duration of SVT. Ultimately, she opted for a catheter ablation to prevent SVT exacerbation during subsequent pregnancies.
THE TAKEAWAY
AVNRT (and other tachyarrhythmias) may worsen or manifest with physiologic changes that occur during pregnancy. After establishing the diagnosis, effort should be made to manage the condition conservatively with Valsalva maneuvers and medication. Catheter ablation should be offered postpartum as a more definitive treatment option.
CORRESPONDENCE
Joseph Lane Wilson, MD, ECU Brody School of Medicine, Department of Family Medicine Medical Director, 101 Heart Drive, Greenville, NC 27834; [email protected].
1. Kwaku KF, Josephson ME. Typical AVNRT—an update on mechanisms and therapy. Card Electrophysiol Rev. 2002;6:414-421.
2. Enriquez AD, Economy KE, Tedrow UB. Contemporary management of arrhythmias during pregnancy. Circ Arrhythm Electrophysiol. 2014;7:961-967.
3. Knotts RJ, Garan H. Cardiac arrhythmias in pregnancy. Semin Perinatol. 2014;38:285-288.
4. Silversides CK, Harris L, Haberer K, et al. Recurrence rates of arrhythmias during pregnancy in women with previous tacharrhythmias and impact on fetal and neonatal outcomes. Am J Cardiol. 2006;97:1206-1212.
5. Page RL, Joglar JA, Caldwell MA, et al. 2015 ACC/AHA/HRS guideline for the management of adult patients with supraventricular tachycardia: a report of the American College of Cardiology/American Heart Association Task Force on Clinical Practice Guidelines and the Heart Rhythm Society. Circulation. 2016;133:e471-e505.
6. Di Biase L, Gianni C, Bagliani G, et. al. Arrhythmias involving the atrioventricular junction. Card Electrophysiol Clin. 2017;9:435-452.
7. Fitzpatrick RB. LactMed: drugs and lactation database. J Electron Resour Med Libr. 2007;4:155.
8. Yaksh A, van der Does LJ, Lanters EA, et al. Pharmacological therapy of tachyarrhythmias during pregnancy. Arrhythm Electrophysiol Rev. 2016;5:41-44.
9. US National Library of Medicine. Drugs and lactation database (LactMed). Available at: toxnet.nlm.nih.gov/newtoxnet/lactmed.htm. Accessed July 3, 2018.
THE CASE
A 29-year-old G1P0 woman at 13 weeks’ gestation came in for a routine prenatal visit complaining of sudden-onset heart palpitations that were occurring about once a week. Each episode lasted between 15 and 60 minutes and was accompanied by chest tightness, with no identifiable cause. The patient could inconsistently terminate the episodes with Valsalva maneuvers. She reported having had 2 similar incidents of palpitations within the past year. Her family history was significant for sudden cardiac death of her father and paternal grandfather in their fifth decades of life.
A cardiovascular exam was normal; heart auscultation revealed a regular rate and rhythm without murmurs, rubs, or gallops, and the peripheral pulses were normal. A thyroid-stimulating hormone (TSH) level, basic metabolic panel (BMP), and complete blood count (CBC) were within normal limits. A transthoracic echocardiogram was negative for structural heart disease.
THE DIAGNOSIS
An initial Holter monitor study failed to capture an episode of her palpitations. The frequency of her palpitations increased as her pregnancy progressed, occurring almost daily by the second half of the third trimester, and a repeat Holter monitor study in the third trimester was significant for a 3-minute episode of supraventricular tachycardia (SVT) that correlated with patient-recorded symptoms (FIGURE).

Based on these results, we diagnosed the patient with an atrioventricular nodal reentry tachycardia (AVNRT). Although atrioventricular reciprocating tachycardia (AVRT) remained a remote possibility, it is far less common, and a 12-lead electrocardiogram (EKG) showed no evidence of pre-excitation.
DISCUSSION
AVNRT is the most common form of paroxysmal supraventricular tachycardia (PSVT). It occurs more frequently in women and typically manifests in the second to fourth decades of life.1 AVNRT is a narrow complex tachycardia characterized by a heart rate of 120 to >200 beats/min.
Hemodynamic changes in pregnancy can trigger arrhythmias
During pregnancy, hemodynamic changes (including increased blood volume and cardiac output) are thought to stimulate stretch-activated ion channels within the walls of the heart.2-4 Such changes may exacerbate previously existing cardiac arrhythmias or (less commonly) cause new-onset arrhythmias.3,4 A family history positive for arrhythmias or sudden cardiac death increases the likelihood of developing tachyarrhythmia during pregnancy.3 Women with a known history of PSVT might experience symptom exacerbation despite being on prophylactic therapy.4
Detection and diagnosis
While AVNRT is relatively benign in pregnancy, other cardiac arrhythmias (eg, atrial fibrillation/flutter, ventricular tachycardia) carry a greater risk for fetal and maternal complications, underscoring the need to correctly identify the type of arrhythmia.2,3
Continue to: Physical exam findings
Physical exam findings are often unremarkable unless the patient is actively experiencing SVT in the office, in which case prominent jugular pulsations may be seen due to simultaneous contraction of the atria and ventricles.
The initial evaluation of a pregnant patient presenting with tachycardia should include a BMP, TSH, 12-lead EKG, and transthoracic echocardiography.3,5 In most patients with AVNRT, the results of these tests will be normal. A Holter monitor can be used to document an arrhythmia if the episodes are relatively frequent or an event monitor can be used if the episodes are infrequent.5
EKG findings. When patients are actively experiencing SVT, EKG findings include a P wave obscured by the QRS complex, sometimes manifesting as a pseudo-R wave in the V1 lead and a pseudo-S wave in leads II, III, and AVF. The QRS complex is narrow and the R-R interval is regular.6
Types of treatment
Valsalva maneuvers. Treatment of AVNRT in pregnancy should first involve addressing any precipitating causes, including metabolic and endocrine abnormalities.3 As virtually all antiarrhythmic drugs cross the placenta and are traceable in breast milk,2,3 patients should be counseled to try to stop episodes using Valsalva maneuvers before moving to pharmacologic treatment.
Antiarrhythmics. First-line pharmacologic treatment for the prevention of AVNRT in pregnancy is metoprolol or verapamil.2,5 Neither drug has been associated with adverse outcomes in infants, although there is a large body of evidence suggesting that low levels of metoprolol are present in breast milk.7
Continue to: Acute episodes of SVT that are refractory to...
Acute episodes of SVT that are refractory to vagal maneuvers or occur despite medical management can be treated acutely in pregnancy with adenosine, which effectively stops episodes about 90% of the time.2 (See the TABLE8,9 for a list of antiarrhythmics that may be used to treat AVNRT.)

Catheter ablation is first-line treatment for AVNRT in nonpregnant patients.1,5 The risks of undergoing ablation during pregnancy include fetal exposure to radiation and anesthetic drugs.2,3 Therefore, this treatment should be used only when pharmacologic treatment is unsuccessful and risks to the mother and fetus due to the arrhythmia outweigh the risks of the procedure. Ablation can be offered postpartum as more definitive therapy.
Our patient was started on metoprolol tartrate 12.5 mg bid at 35 weeks’ gestation due to increasingly common and persistent palpitations. This helped control the episodes for 2 weeks, at which point they increased again in frequency. These were terminated using Valsalva maneuvers; increasing the metoprolol dosage was prohibitive due to patient intolerance.
Following an uncomplicated delivery, and discontinuation of metoprolol, the patient reported a decrease in both the number of episodes and the duration of SVT. Ultimately, she opted for a catheter ablation to prevent SVT exacerbation during subsequent pregnancies.
THE TAKEAWAY
AVNRT (and other tachyarrhythmias) may worsen or manifest with physiologic changes that occur during pregnancy. After establishing the diagnosis, effort should be made to manage the condition conservatively with Valsalva maneuvers and medication. Catheter ablation should be offered postpartum as a more definitive treatment option.
CORRESPONDENCE
Joseph Lane Wilson, MD, ECU Brody School of Medicine, Department of Family Medicine Medical Director, 101 Heart Drive, Greenville, NC 27834; [email protected].
THE CASE
A 29-year-old G1P0 woman at 13 weeks’ gestation came in for a routine prenatal visit complaining of sudden-onset heart palpitations that were occurring about once a week. Each episode lasted between 15 and 60 minutes and was accompanied by chest tightness, with no identifiable cause. The patient could inconsistently terminate the episodes with Valsalva maneuvers. She reported having had 2 similar incidents of palpitations within the past year. Her family history was significant for sudden cardiac death of her father and paternal grandfather in their fifth decades of life.
A cardiovascular exam was normal; heart auscultation revealed a regular rate and rhythm without murmurs, rubs, or gallops, and the peripheral pulses were normal. A thyroid-stimulating hormone (TSH) level, basic metabolic panel (BMP), and complete blood count (CBC) were within normal limits. A transthoracic echocardiogram was negative for structural heart disease.
THE DIAGNOSIS
An initial Holter monitor study failed to capture an episode of her palpitations. The frequency of her palpitations increased as her pregnancy progressed, occurring almost daily by the second half of the third trimester, and a repeat Holter monitor study in the third trimester was significant for a 3-minute episode of supraventricular tachycardia (SVT) that correlated with patient-recorded symptoms (FIGURE).

Based on these results, we diagnosed the patient with an atrioventricular nodal reentry tachycardia (AVNRT). Although atrioventricular reciprocating tachycardia (AVRT) remained a remote possibility, it is far less common, and a 12-lead electrocardiogram (EKG) showed no evidence of pre-excitation.
DISCUSSION
AVNRT is the most common form of paroxysmal supraventricular tachycardia (PSVT). It occurs more frequently in women and typically manifests in the second to fourth decades of life.1 AVNRT is a narrow complex tachycardia characterized by a heart rate of 120 to >200 beats/min.
Hemodynamic changes in pregnancy can trigger arrhythmias
During pregnancy, hemodynamic changes (including increased blood volume and cardiac output) are thought to stimulate stretch-activated ion channels within the walls of the heart.2-4 Such changes may exacerbate previously existing cardiac arrhythmias or (less commonly) cause new-onset arrhythmias.3,4 A family history positive for arrhythmias or sudden cardiac death increases the likelihood of developing tachyarrhythmia during pregnancy.3 Women with a known history of PSVT might experience symptom exacerbation despite being on prophylactic therapy.4
Detection and diagnosis
While AVNRT is relatively benign in pregnancy, other cardiac arrhythmias (eg, atrial fibrillation/flutter, ventricular tachycardia) carry a greater risk for fetal and maternal complications, underscoring the need to correctly identify the type of arrhythmia.2,3
Continue to: Physical exam findings
Physical exam findings are often unremarkable unless the patient is actively experiencing SVT in the office, in which case prominent jugular pulsations may be seen due to simultaneous contraction of the atria and ventricles.
The initial evaluation of a pregnant patient presenting with tachycardia should include a BMP, TSH, 12-lead EKG, and transthoracic echocardiography.3,5 In most patients with AVNRT, the results of these tests will be normal. A Holter monitor can be used to document an arrhythmia if the episodes are relatively frequent or an event monitor can be used if the episodes are infrequent.5
EKG findings. When patients are actively experiencing SVT, EKG findings include a P wave obscured by the QRS complex, sometimes manifesting as a pseudo-R wave in the V1 lead and a pseudo-S wave in leads II, III, and AVF. The QRS complex is narrow and the R-R interval is regular.6
Types of treatment
Valsalva maneuvers. Treatment of AVNRT in pregnancy should first involve addressing any precipitating causes, including metabolic and endocrine abnormalities.3 As virtually all antiarrhythmic drugs cross the placenta and are traceable in breast milk,2,3 patients should be counseled to try to stop episodes using Valsalva maneuvers before moving to pharmacologic treatment.
Antiarrhythmics. First-line pharmacologic treatment for the prevention of AVNRT in pregnancy is metoprolol or verapamil.2,5 Neither drug has been associated with adverse outcomes in infants, although there is a large body of evidence suggesting that low levels of metoprolol are present in breast milk.7
Continue to: Acute episodes of SVT that are refractory to...
Acute episodes of SVT that are refractory to vagal maneuvers or occur despite medical management can be treated acutely in pregnancy with adenosine, which effectively stops episodes about 90% of the time.2 (See the TABLE8,9 for a list of antiarrhythmics that may be used to treat AVNRT.)

Catheter ablation is first-line treatment for AVNRT in nonpregnant patients.1,5 The risks of undergoing ablation during pregnancy include fetal exposure to radiation and anesthetic drugs.2,3 Therefore, this treatment should be used only when pharmacologic treatment is unsuccessful and risks to the mother and fetus due to the arrhythmia outweigh the risks of the procedure. Ablation can be offered postpartum as more definitive therapy.
Our patient was started on metoprolol tartrate 12.5 mg bid at 35 weeks’ gestation due to increasingly common and persistent palpitations. This helped control the episodes for 2 weeks, at which point they increased again in frequency. These were terminated using Valsalva maneuvers; increasing the metoprolol dosage was prohibitive due to patient intolerance.
Following an uncomplicated delivery, and discontinuation of metoprolol, the patient reported a decrease in both the number of episodes and the duration of SVT. Ultimately, she opted for a catheter ablation to prevent SVT exacerbation during subsequent pregnancies.
THE TAKEAWAY
AVNRT (and other tachyarrhythmias) may worsen or manifest with physiologic changes that occur during pregnancy. After establishing the diagnosis, effort should be made to manage the condition conservatively with Valsalva maneuvers and medication. Catheter ablation should be offered postpartum as a more definitive treatment option.
CORRESPONDENCE
Joseph Lane Wilson, MD, ECU Brody School of Medicine, Department of Family Medicine Medical Director, 101 Heart Drive, Greenville, NC 27834; [email protected].
1. Kwaku KF, Josephson ME. Typical AVNRT—an update on mechanisms and therapy. Card Electrophysiol Rev. 2002;6:414-421.
2. Enriquez AD, Economy KE, Tedrow UB. Contemporary management of arrhythmias during pregnancy. Circ Arrhythm Electrophysiol. 2014;7:961-967.
3. Knotts RJ, Garan H. Cardiac arrhythmias in pregnancy. Semin Perinatol. 2014;38:285-288.
4. Silversides CK, Harris L, Haberer K, et al. Recurrence rates of arrhythmias during pregnancy in women with previous tacharrhythmias and impact on fetal and neonatal outcomes. Am J Cardiol. 2006;97:1206-1212.
5. Page RL, Joglar JA, Caldwell MA, et al. 2015 ACC/AHA/HRS guideline for the management of adult patients with supraventricular tachycardia: a report of the American College of Cardiology/American Heart Association Task Force on Clinical Practice Guidelines and the Heart Rhythm Society. Circulation. 2016;133:e471-e505.
6. Di Biase L, Gianni C, Bagliani G, et. al. Arrhythmias involving the atrioventricular junction. Card Electrophysiol Clin. 2017;9:435-452.
7. Fitzpatrick RB. LactMed: drugs and lactation database. J Electron Resour Med Libr. 2007;4:155.
8. Yaksh A, van der Does LJ, Lanters EA, et al. Pharmacological therapy of tachyarrhythmias during pregnancy. Arrhythm Electrophysiol Rev. 2016;5:41-44.
9. US National Library of Medicine. Drugs and lactation database (LactMed). Available at: toxnet.nlm.nih.gov/newtoxnet/lactmed.htm. Accessed July 3, 2018.
1. Kwaku KF, Josephson ME. Typical AVNRT—an update on mechanisms and therapy. Card Electrophysiol Rev. 2002;6:414-421.
2. Enriquez AD, Economy KE, Tedrow UB. Contemporary management of arrhythmias during pregnancy. Circ Arrhythm Electrophysiol. 2014;7:961-967.
3. Knotts RJ, Garan H. Cardiac arrhythmias in pregnancy. Semin Perinatol. 2014;38:285-288.
4. Silversides CK, Harris L, Haberer K, et al. Recurrence rates of arrhythmias during pregnancy in women with previous tacharrhythmias and impact on fetal and neonatal outcomes. Am J Cardiol. 2006;97:1206-1212.
5. Page RL, Joglar JA, Caldwell MA, et al. 2015 ACC/AHA/HRS guideline for the management of adult patients with supraventricular tachycardia: a report of the American College of Cardiology/American Heart Association Task Force on Clinical Practice Guidelines and the Heart Rhythm Society. Circulation. 2016;133:e471-e505.
6. Di Biase L, Gianni C, Bagliani G, et. al. Arrhythmias involving the atrioventricular junction. Card Electrophysiol Clin. 2017;9:435-452.
7. Fitzpatrick RB. LactMed: drugs and lactation database. J Electron Resour Med Libr. 2007;4:155.
8. Yaksh A, van der Does LJ, Lanters EA, et al. Pharmacological therapy of tachyarrhythmias during pregnancy. Arrhythm Electrophysiol Rev. 2016;5:41-44.
9. US National Library of Medicine. Drugs and lactation database (LactMed). Available at: toxnet.nlm.nih.gov/newtoxnet/lactmed.htm. Accessed July 3, 2018.
New York City launches initiative to eliminate racial disparities in maternal death
In response to alarming racial disparities, New York City announced a new initiative last week to reduce maternal deaths and complications among women of color. Under the new plan, the city will improve the data collection on maternal deaths and complications, fund implicit bias training for medical staff at private and public hospitals, and launch a public awareness campaign.
Over the next three years, the city will spend $12.8 million on the initiative, with the goal of eliminating the black-white racial disparity in deaths related to pregnancy and childbirth and cutting the number of complications in half within five years.
“We recognize these are ambitious goals, but they are not unrealistic,” said Dr. Herminia Palacio, New York City’s deputy mayor for health and human services. “It’s an explicit recognition of the urgency of this issue and puts the goal posts in front of us.”
The city’s health department is targeting nearly two dozen public and private hospitals over four years, focusing on neighborhoods with the highest complication rates, including the South Bronx, North and Central Brooklyn, and East and Central Harlem. Hospital officials will study data from cases that led to bad outcomes, and staff will participate in drills aimed at helping them recognize and treat those complications.
Health department officials approached SUNY Downstate Medical Center in May to serve as a pilot site for many of the new measures.
The Central Brooklyn hospital was featured in the “Lost Mothers” series published by ProPublica and NPR last year as one of the starkest examples of racial disparities among hospitals in three states, according to our analysis of over 1 million births in Florida, Illinois and New York. In the second half of last year, two women, both black, died shortly after delivering at SUNY Downstate from causes that experts have said are preventable. The public, state-run hospital has one of the highest complication rates for hemorrhage in the city.
“We look forward to working with all of our partners to provide quality maternal health care for expectant mothers,” said hospital spokesperson Dawn Skeete-Walker.
“SUNY Downstate serves a unique and diverse population in Brooklyn where many of our expectant mothers are from a variety of different backgrounds, beliefs, and cultures.”
The city will also specifically target its own public hospitals, which are run by NYC Health + Hospitals, training staff on how to better identify and treat hemorrhage and blood clots, two leading causes of maternal death.
The initiative is “aimed at using an approach that encourages folks to have a sense of accountability without finger pointing or blame, and that encourages hospitals to be active participants to identify practices that would benefit from improvement,” said Palacio.
In addition to training, the city’s public hospitals will hire maternal care coordinators who will assist high-risk pregnant women with their appointments, prescriptions and public health benefits. Public hospitals will also work to strengthen prenatal and postpartum care, including conducting hemorrhage assessments, establishing care plans, and providing contraceptive counselling, breastfeeding support and screening for maternal depression.
Starting in 2019, the health department plans to launch a maternal safety public awareness campaign in partnership with grassroots organizations.
“This is a positive first step in really being able to address the concerns of women of color and pregnant women,” said Chanel Porchia-Albert, founder and executive director of Ancient Song Doula Services, which is based in New York City. “There need to be accountability measures that are put in place that stress the community as an active participant and stakeholder.”
The city’s initiative is the latest in a wave of maternal health reforms following the “Lost Mothers” series. Over the past few months, the U.S. Senate has proposed $50 million in funding to reduce maternal deaths, and several states have launched review committees to examine birth outcomes.
As ProPublica and NPR reported, between 700 and 900 women die from causes related to pregnancy and childbirth in the United States every year, and tens of thousands more experience severe complications. The rate of maternal death is substantially higher in the United States than in other affluent nations, and has climbed over the past decade, mostly driven by the outcomes of women of color.
While poverty and inadequate access to health care explain part of the racial disparity in maternal deaths, research has shown that the quality of care at hospitals where black women deliver plays a significant role as well. ProPublica added to research that has found that women who deliver at disproportionately “black-serving” hospitals are more likely to experience serious complications — from emergency hysterectomies to birth-related blood clots — than mothers who deliver at institutions that serve fewer black women.
In New York City, the racial disparity in maternal outcomes is among the largest in the nation, and it’s growing. According to a recent report from New York City’s Department of Health and Mental Hygiene, even as the overall maternal mortality rate across the city has decreased, the gap between black and white mothers has widened.
Regardless of their education, obesity or poverty level, black mothers in New York City are at a higher risk of harm than their white counterparts. Black mothers with a college education fare worse than women of all other races who dropped out of high school. Black women of normal weight have higher rates of harm than obese women of all other races. And black women who reside in the wealthiest neighborhoods have worse outcomes than white, Asian and Hispanic mothers in the poorest ones.
“If you are a poor black woman, you don’t have access to quality OBGYN care, and if you are a wealthy black women, like Serena Williams, you get providers who don’t listen to you when you say you can’t breathe,” said Patricia Loftman, a member of the American College of Nurse Midwives Board of Directors who worked for 30 years as a certified nurse-midwife in Harlem. “The components of this initiative are very aggressive and laudable to the extent that they are forcing hospital departments to talk about implicit bias.”
ProPublica is a Pulitzer Prize-winning investigative newsroom.
In response to alarming racial disparities, New York City announced a new initiative last week to reduce maternal deaths and complications among women of color. Under the new plan, the city will improve the data collection on maternal deaths and complications, fund implicit bias training for medical staff at private and public hospitals, and launch a public awareness campaign.
Over the next three years, the city will spend $12.8 million on the initiative, with the goal of eliminating the black-white racial disparity in deaths related to pregnancy and childbirth and cutting the number of complications in half within five years.
“We recognize these are ambitious goals, but they are not unrealistic,” said Dr. Herminia Palacio, New York City’s deputy mayor for health and human services. “It’s an explicit recognition of the urgency of this issue and puts the goal posts in front of us.”
The city’s health department is targeting nearly two dozen public and private hospitals over four years, focusing on neighborhoods with the highest complication rates, including the South Bronx, North and Central Brooklyn, and East and Central Harlem. Hospital officials will study data from cases that led to bad outcomes, and staff will participate in drills aimed at helping them recognize and treat those complications.
Health department officials approached SUNY Downstate Medical Center in May to serve as a pilot site for many of the new measures.
The Central Brooklyn hospital was featured in the “Lost Mothers” series published by ProPublica and NPR last year as one of the starkest examples of racial disparities among hospitals in three states, according to our analysis of over 1 million births in Florida, Illinois and New York. In the second half of last year, two women, both black, died shortly after delivering at SUNY Downstate from causes that experts have said are preventable. The public, state-run hospital has one of the highest complication rates for hemorrhage in the city.
“We look forward to working with all of our partners to provide quality maternal health care for expectant mothers,” said hospital spokesperson Dawn Skeete-Walker.
“SUNY Downstate serves a unique and diverse population in Brooklyn where many of our expectant mothers are from a variety of different backgrounds, beliefs, and cultures.”
The city will also specifically target its own public hospitals, which are run by NYC Health + Hospitals, training staff on how to better identify and treat hemorrhage and blood clots, two leading causes of maternal death.
The initiative is “aimed at using an approach that encourages folks to have a sense of accountability without finger pointing or blame, and that encourages hospitals to be active participants to identify practices that would benefit from improvement,” said Palacio.
In addition to training, the city’s public hospitals will hire maternal care coordinators who will assist high-risk pregnant women with their appointments, prescriptions and public health benefits. Public hospitals will also work to strengthen prenatal and postpartum care, including conducting hemorrhage assessments, establishing care plans, and providing contraceptive counselling, breastfeeding support and screening for maternal depression.
Starting in 2019, the health department plans to launch a maternal safety public awareness campaign in partnership with grassroots organizations.
“This is a positive first step in really being able to address the concerns of women of color and pregnant women,” said Chanel Porchia-Albert, founder and executive director of Ancient Song Doula Services, which is based in New York City. “There need to be accountability measures that are put in place that stress the community as an active participant and stakeholder.”
The city’s initiative is the latest in a wave of maternal health reforms following the “Lost Mothers” series. Over the past few months, the U.S. Senate has proposed $50 million in funding to reduce maternal deaths, and several states have launched review committees to examine birth outcomes.
As ProPublica and NPR reported, between 700 and 900 women die from causes related to pregnancy and childbirth in the United States every year, and tens of thousands more experience severe complications. The rate of maternal death is substantially higher in the United States than in other affluent nations, and has climbed over the past decade, mostly driven by the outcomes of women of color.
While poverty and inadequate access to health care explain part of the racial disparity in maternal deaths, research has shown that the quality of care at hospitals where black women deliver plays a significant role as well. ProPublica added to research that has found that women who deliver at disproportionately “black-serving” hospitals are more likely to experience serious complications — from emergency hysterectomies to birth-related blood clots — than mothers who deliver at institutions that serve fewer black women.
In New York City, the racial disparity in maternal outcomes is among the largest in the nation, and it’s growing. According to a recent report from New York City’s Department of Health and Mental Hygiene, even as the overall maternal mortality rate across the city has decreased, the gap between black and white mothers has widened.
Regardless of their education, obesity or poverty level, black mothers in New York City are at a higher risk of harm than their white counterparts. Black mothers with a college education fare worse than women of all other races who dropped out of high school. Black women of normal weight have higher rates of harm than obese women of all other races. And black women who reside in the wealthiest neighborhoods have worse outcomes than white, Asian and Hispanic mothers in the poorest ones.
“If you are a poor black woman, you don’t have access to quality OBGYN care, and if you are a wealthy black women, like Serena Williams, you get providers who don’t listen to you when you say you can’t breathe,” said Patricia Loftman, a member of the American College of Nurse Midwives Board of Directors who worked for 30 years as a certified nurse-midwife in Harlem. “The components of this initiative are very aggressive and laudable to the extent that they are forcing hospital departments to talk about implicit bias.”
ProPublica is a Pulitzer Prize-winning investigative newsroom.
In response to alarming racial disparities, New York City announced a new initiative last week to reduce maternal deaths and complications among women of color. Under the new plan, the city will improve the data collection on maternal deaths and complications, fund implicit bias training for medical staff at private and public hospitals, and launch a public awareness campaign.
Over the next three years, the city will spend $12.8 million on the initiative, with the goal of eliminating the black-white racial disparity in deaths related to pregnancy and childbirth and cutting the number of complications in half within five years.
“We recognize these are ambitious goals, but they are not unrealistic,” said Dr. Herminia Palacio, New York City’s deputy mayor for health and human services. “It’s an explicit recognition of the urgency of this issue and puts the goal posts in front of us.”
The city’s health department is targeting nearly two dozen public and private hospitals over four years, focusing on neighborhoods with the highest complication rates, including the South Bronx, North and Central Brooklyn, and East and Central Harlem. Hospital officials will study data from cases that led to bad outcomes, and staff will participate in drills aimed at helping them recognize and treat those complications.
Health department officials approached SUNY Downstate Medical Center in May to serve as a pilot site for many of the new measures.
The Central Brooklyn hospital was featured in the “Lost Mothers” series published by ProPublica and NPR last year as one of the starkest examples of racial disparities among hospitals in three states, according to our analysis of over 1 million births in Florida, Illinois and New York. In the second half of last year, two women, both black, died shortly after delivering at SUNY Downstate from causes that experts have said are preventable. The public, state-run hospital has one of the highest complication rates for hemorrhage in the city.
“We look forward to working with all of our partners to provide quality maternal health care for expectant mothers,” said hospital spokesperson Dawn Skeete-Walker.
“SUNY Downstate serves a unique and diverse population in Brooklyn where many of our expectant mothers are from a variety of different backgrounds, beliefs, and cultures.”
The city will also specifically target its own public hospitals, which are run by NYC Health + Hospitals, training staff on how to better identify and treat hemorrhage and blood clots, two leading causes of maternal death.
The initiative is “aimed at using an approach that encourages folks to have a sense of accountability without finger pointing or blame, and that encourages hospitals to be active participants to identify practices that would benefit from improvement,” said Palacio.
In addition to training, the city’s public hospitals will hire maternal care coordinators who will assist high-risk pregnant women with their appointments, prescriptions and public health benefits. Public hospitals will also work to strengthen prenatal and postpartum care, including conducting hemorrhage assessments, establishing care plans, and providing contraceptive counselling, breastfeeding support and screening for maternal depression.
Starting in 2019, the health department plans to launch a maternal safety public awareness campaign in partnership with grassroots organizations.
“This is a positive first step in really being able to address the concerns of women of color and pregnant women,” said Chanel Porchia-Albert, founder and executive director of Ancient Song Doula Services, which is based in New York City. “There need to be accountability measures that are put in place that stress the community as an active participant and stakeholder.”
The city’s initiative is the latest in a wave of maternal health reforms following the “Lost Mothers” series. Over the past few months, the U.S. Senate has proposed $50 million in funding to reduce maternal deaths, and several states have launched review committees to examine birth outcomes.
As ProPublica and NPR reported, between 700 and 900 women die from causes related to pregnancy and childbirth in the United States every year, and tens of thousands more experience severe complications. The rate of maternal death is substantially higher in the United States than in other affluent nations, and has climbed over the past decade, mostly driven by the outcomes of women of color.
While poverty and inadequate access to health care explain part of the racial disparity in maternal deaths, research has shown that the quality of care at hospitals where black women deliver plays a significant role as well. ProPublica added to research that has found that women who deliver at disproportionately “black-serving” hospitals are more likely to experience serious complications — from emergency hysterectomies to birth-related blood clots — than mothers who deliver at institutions that serve fewer black women.
In New York City, the racial disparity in maternal outcomes is among the largest in the nation, and it’s growing. According to a recent report from New York City’s Department of Health and Mental Hygiene, even as the overall maternal mortality rate across the city has decreased, the gap between black and white mothers has widened.
Regardless of their education, obesity or poverty level, black mothers in New York City are at a higher risk of harm than their white counterparts. Black mothers with a college education fare worse than women of all other races who dropped out of high school. Black women of normal weight have higher rates of harm than obese women of all other races. And black women who reside in the wealthiest neighborhoods have worse outcomes than white, Asian and Hispanic mothers in the poorest ones.
“If you are a poor black woman, you don’t have access to quality OBGYN care, and if you are a wealthy black women, like Serena Williams, you get providers who don’t listen to you when you say you can’t breathe,” said Patricia Loftman, a member of the American College of Nurse Midwives Board of Directors who worked for 30 years as a certified nurse-midwife in Harlem. “The components of this initiative are very aggressive and laudable to the extent that they are forcing hospital departments to talk about implicit bias.”
ProPublica is a Pulitzer Prize-winning investigative newsroom.