User login
Official Newspaper of the American College of Surgeons
Most Medicaid enrollees exempt from work requirements
Only 6% of the Medicaid population would be unlikely to qualify for an exemption from work requirements for “able-bodied adults” that states are in the process of being implementing, according to a new report from the Kaiser Family Foundation.
Another 15% were in fair or poor health or didn’t work because of illness or disability, 11% didn’t work because they were providing care for family members, and 6% were attending school, Kaiser wrote in an issue brief.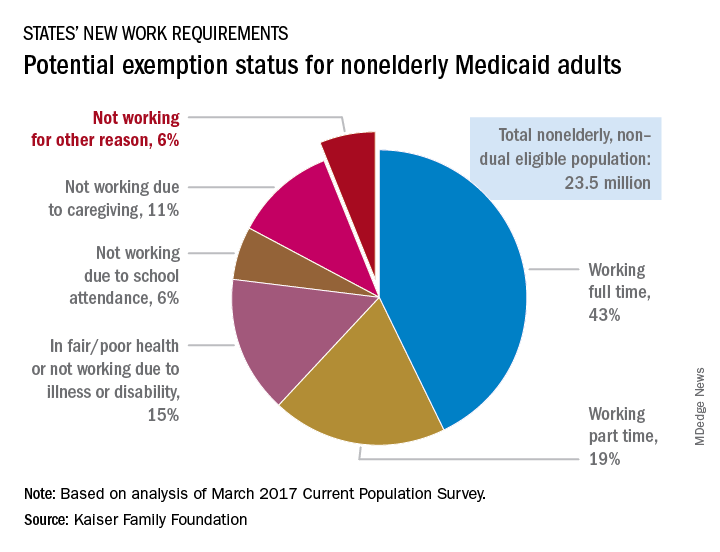
“This target population is much smaller than the groups of enrollees who are already working but would need to comply with new reporting requirements and those who could be exempt and would have to navigate an exemption process,” the Kaiser investigators said.
States will need to set up systems to deal with these issues, but many enrollees face barriers to complying. The waiver program in Arkansas – one of the first four states to receive permission to impose work requirements – “requires beneficiaries to set up an online account and use this account as the sole means of periodic reporting related to work requirements and exemptions,” they noted, but 30% of all nonelderly Medicaid adults say that they have never used a computer, 21% do not use the Internet, and 41% do not use email, based on analysis of 2016 National Health Interview Survey data.
Only 6% of the Medicaid population would be unlikely to qualify for an exemption from work requirements for “able-bodied adults” that states are in the process of being implementing, according to a new report from the Kaiser Family Foundation.
Another 15% were in fair or poor health or didn’t work because of illness or disability, 11% didn’t work because they were providing care for family members, and 6% were attending school, Kaiser wrote in an issue brief.
“This target population is much smaller than the groups of enrollees who are already working but would need to comply with new reporting requirements and those who could be exempt and would have to navigate an exemption process,” the Kaiser investigators said.
States will need to set up systems to deal with these issues, but many enrollees face barriers to complying. The waiver program in Arkansas – one of the first four states to receive permission to impose work requirements – “requires beneficiaries to set up an online account and use this account as the sole means of periodic reporting related to work requirements and exemptions,” they noted, but 30% of all nonelderly Medicaid adults say that they have never used a computer, 21% do not use the Internet, and 41% do not use email, based on analysis of 2016 National Health Interview Survey data.
Only 6% of the Medicaid population would be unlikely to qualify for an exemption from work requirements for “able-bodied adults” that states are in the process of being implementing, according to a new report from the Kaiser Family Foundation.
Another 15% were in fair or poor health or didn’t work because of illness or disability, 11% didn’t work because they were providing care for family members, and 6% were attending school, Kaiser wrote in an issue brief.
“This target population is much smaller than the groups of enrollees who are already working but would need to comply with new reporting requirements and those who could be exempt and would have to navigate an exemption process,” the Kaiser investigators said.
States will need to set up systems to deal with these issues, but many enrollees face barriers to complying. The waiver program in Arkansas – one of the first four states to receive permission to impose work requirements – “requires beneficiaries to set up an online account and use this account as the sole means of periodic reporting related to work requirements and exemptions,” they noted, but 30% of all nonelderly Medicaid adults say that they have never used a computer, 21% do not use the Internet, and 41% do not use email, based on analysis of 2016 National Health Interview Survey data.
Lung volume reduction procedures on the rise since 2011
SAN DIEGO – The number of has been increasing since 2011, according to a large national analysis.
Lung volume–reduction surgery (LVRS) has been available for decades, but results from the National Emphysema Treatment Trial (NETT) in 2003 (N Engl J Med 2003;348:2059-73) demonstrated that certain COPD patients benefited from the surgery, Amy Attaway, MD, said at an international conference of the American Thoracic Society.
In that trial, overall mortality at 30 days was 3.6% for the surgical group. “If you excluded high-risk patients, the 30-day mortality was only 2.2%,” said Dr. Attaway, a staff physician at the Cleveland Clinic Respiratory Institute. “If you look at the upper lobe–predominant, low-exercise group, their mortality at 30 days was 1.4%.”
Subsequent studies that evaluated NETT patients over time showed continued improvements in their mortality. For example, one study found that in the upper lobe–predominant patients who received surgery in the NETT trial, their survival at 3 years was 81% vs. 74% in the medical group (P = .05), while survival at 5 years was 70% in the surgery group vs. 60% in the medical group (P = .02; J Thorac Cardiovasc Surg. 2010;140[3]:564-72). Despite this improved mortality, other studies have shown that LVRS remains underutilized. Once such analysis of the Nationwide Inpatient Sample showed a logarithmic drop from 2000 to 2010 (Chest 2014;146[6]:e228-9). The authors also noted that the overall mortality was 6% and that the need for a tracheostomy was 7.9%. Age greater than 65 years was associated with increased mortality (odds ratio 2.8).
Dr. Attaway and her associates hypothesized that availability of the long-term survival data from the NETT, support from GOLD guidelines, and the lack of a Food and Drug Administration–approved alternative may have increased utilization of this surgery from 2007 through 2013. With data from the Nationwide Inpatient Sample for 2007-2013, the researchers performed a retrospective cohort analysis of 2,805 patients who underwent LVRS. The composite primary outcome was mortality or need for tracheostomy. Logistic regression was performed to analyze factors associated with the composite primary outcome.
The average patient age was 59 years, and 64% were male. Medicare was the payer in nearly half of cases, in-hospital mortality and need for tracheostomy were both 5.5%, and the risk for tracheostomy or mortality was 10.5%. Linear regression analysis showed a significant increase in LVRS over time, with the 320 surgeries in 2007 and 605 in 2013 (P = .0016).
On univariate analysis, the following factors were found to be significantly associated with the composite primary outcome: in-hospital mortality or the need for tracheostomy (P less than .001), respiratory failure (P less than .001), septicemia (P = .01), shock (P less than .001), acute kidney injury (P less than .001), secondary pulmonary hypertension (P less than .001), and a higher mean number of diagnoses or number of chronic conditions on admission (P less than .001 and P = .017, respectively).
On multivariate logistic regression, only two factors were found to be significantly associated with the composite primary outcome: a higher number of diagnoses (adjusted OR of 1.17 per additional diagnosis), and the presence of secondary pulmonary hypertension (adjusted OR 4.4).
“Availability of long-term survival data from NETT, support from the GOLD guidelines, and lack of current FDA-approved alternatives are potential reasons for increased utilization [of LVRS],” Dr. Attaway said. “However, our study showed that in-hospital mortality and morbidity is high, compared to the NETT results. We also found that secondary pulmonary hypertension and comorbidities are associated with poor outcomes. This is important to keep in mind for patient selection.”
Dr. Attaway and her associates reported having no financial disclosures.
SOURCE: Attaway A et al. ATS 2018, Abstract 4436.
SAN DIEGO – The number of has been increasing since 2011, according to a large national analysis.
Lung volume–reduction surgery (LVRS) has been available for decades, but results from the National Emphysema Treatment Trial (NETT) in 2003 (N Engl J Med 2003;348:2059-73) demonstrated that certain COPD patients benefited from the surgery, Amy Attaway, MD, said at an international conference of the American Thoracic Society.
In that trial, overall mortality at 30 days was 3.6% for the surgical group. “If you excluded high-risk patients, the 30-day mortality was only 2.2%,” said Dr. Attaway, a staff physician at the Cleveland Clinic Respiratory Institute. “If you look at the upper lobe–predominant, low-exercise group, their mortality at 30 days was 1.4%.”
Subsequent studies that evaluated NETT patients over time showed continued improvements in their mortality. For example, one study found that in the upper lobe–predominant patients who received surgery in the NETT trial, their survival at 3 years was 81% vs. 74% in the medical group (P = .05), while survival at 5 years was 70% in the surgery group vs. 60% in the medical group (P = .02; J Thorac Cardiovasc Surg. 2010;140[3]:564-72). Despite this improved mortality, other studies have shown that LVRS remains underutilized. Once such analysis of the Nationwide Inpatient Sample showed a logarithmic drop from 2000 to 2010 (Chest 2014;146[6]:e228-9). The authors also noted that the overall mortality was 6% and that the need for a tracheostomy was 7.9%. Age greater than 65 years was associated with increased mortality (odds ratio 2.8).
Dr. Attaway and her associates hypothesized that availability of the long-term survival data from the NETT, support from GOLD guidelines, and the lack of a Food and Drug Administration–approved alternative may have increased utilization of this surgery from 2007 through 2013. With data from the Nationwide Inpatient Sample for 2007-2013, the researchers performed a retrospective cohort analysis of 2,805 patients who underwent LVRS. The composite primary outcome was mortality or need for tracheostomy. Logistic regression was performed to analyze factors associated with the composite primary outcome.
The average patient age was 59 years, and 64% were male. Medicare was the payer in nearly half of cases, in-hospital mortality and need for tracheostomy were both 5.5%, and the risk for tracheostomy or mortality was 10.5%. Linear regression analysis showed a significant increase in LVRS over time, with the 320 surgeries in 2007 and 605 in 2013 (P = .0016).
On univariate analysis, the following factors were found to be significantly associated with the composite primary outcome: in-hospital mortality or the need for tracheostomy (P less than .001), respiratory failure (P less than .001), septicemia (P = .01), shock (P less than .001), acute kidney injury (P less than .001), secondary pulmonary hypertension (P less than .001), and a higher mean number of diagnoses or number of chronic conditions on admission (P less than .001 and P = .017, respectively).
On multivariate logistic regression, only two factors were found to be significantly associated with the composite primary outcome: a higher number of diagnoses (adjusted OR of 1.17 per additional diagnosis), and the presence of secondary pulmonary hypertension (adjusted OR 4.4).
“Availability of long-term survival data from NETT, support from the GOLD guidelines, and lack of current FDA-approved alternatives are potential reasons for increased utilization [of LVRS],” Dr. Attaway said. “However, our study showed that in-hospital mortality and morbidity is high, compared to the NETT results. We also found that secondary pulmonary hypertension and comorbidities are associated with poor outcomes. This is important to keep in mind for patient selection.”
Dr. Attaway and her associates reported having no financial disclosures.
SOURCE: Attaway A et al. ATS 2018, Abstract 4436.
SAN DIEGO – The number of has been increasing since 2011, according to a large national analysis.
Lung volume–reduction surgery (LVRS) has been available for decades, but results from the National Emphysema Treatment Trial (NETT) in 2003 (N Engl J Med 2003;348:2059-73) demonstrated that certain COPD patients benefited from the surgery, Amy Attaway, MD, said at an international conference of the American Thoracic Society.
In that trial, overall mortality at 30 days was 3.6% for the surgical group. “If you excluded high-risk patients, the 30-day mortality was only 2.2%,” said Dr. Attaway, a staff physician at the Cleveland Clinic Respiratory Institute. “If you look at the upper lobe–predominant, low-exercise group, their mortality at 30 days was 1.4%.”
Subsequent studies that evaluated NETT patients over time showed continued improvements in their mortality. For example, one study found that in the upper lobe–predominant patients who received surgery in the NETT trial, their survival at 3 years was 81% vs. 74% in the medical group (P = .05), while survival at 5 years was 70% in the surgery group vs. 60% in the medical group (P = .02; J Thorac Cardiovasc Surg. 2010;140[3]:564-72). Despite this improved mortality, other studies have shown that LVRS remains underutilized. Once such analysis of the Nationwide Inpatient Sample showed a logarithmic drop from 2000 to 2010 (Chest 2014;146[6]:e228-9). The authors also noted that the overall mortality was 6% and that the need for a tracheostomy was 7.9%. Age greater than 65 years was associated with increased mortality (odds ratio 2.8).
Dr. Attaway and her associates hypothesized that availability of the long-term survival data from the NETT, support from GOLD guidelines, and the lack of a Food and Drug Administration–approved alternative may have increased utilization of this surgery from 2007 through 2013. With data from the Nationwide Inpatient Sample for 2007-2013, the researchers performed a retrospective cohort analysis of 2,805 patients who underwent LVRS. The composite primary outcome was mortality or need for tracheostomy. Logistic regression was performed to analyze factors associated with the composite primary outcome.
The average patient age was 59 years, and 64% were male. Medicare was the payer in nearly half of cases, in-hospital mortality and need for tracheostomy were both 5.5%, and the risk for tracheostomy or mortality was 10.5%. Linear regression analysis showed a significant increase in LVRS over time, with the 320 surgeries in 2007 and 605 in 2013 (P = .0016).
On univariate analysis, the following factors were found to be significantly associated with the composite primary outcome: in-hospital mortality or the need for tracheostomy (P less than .001), respiratory failure (P less than .001), septicemia (P = .01), shock (P less than .001), acute kidney injury (P less than .001), secondary pulmonary hypertension (P less than .001), and a higher mean number of diagnoses or number of chronic conditions on admission (P less than .001 and P = .017, respectively).
On multivariate logistic regression, only two factors were found to be significantly associated with the composite primary outcome: a higher number of diagnoses (adjusted OR of 1.17 per additional diagnosis), and the presence of secondary pulmonary hypertension (adjusted OR 4.4).
“Availability of long-term survival data from NETT, support from the GOLD guidelines, and lack of current FDA-approved alternatives are potential reasons for increased utilization [of LVRS],” Dr. Attaway said. “However, our study showed that in-hospital mortality and morbidity is high, compared to the NETT results. We also found that secondary pulmonary hypertension and comorbidities are associated with poor outcomes. This is important to keep in mind for patient selection.”
Dr. Attaway and her associates reported having no financial disclosures.
SOURCE: Attaway A et al. ATS 2018, Abstract 4436.
AT ATS 2018
Key clinical point: Lung volume–reduction surgery remains an evidence-based therapy for a cohort of severe COPD patients.
Major finding: Linear regression analysis showed a significant increase in LVRS over time, with the 320 surgeries in 2007 and 605 in 2013 (P = .0016).
Study details: A retrospective cohort analysis of 2,805 patients who underwent LVRS.
Disclosures: The researchers reported having no financial disclosures.
Source: Attaway A et al. Abstract 4436, ATS 2018.
PREOPANC-1: Early findings suggest benefit with preop chemo in pancreatic cancer
CHICAGO – Preoperative chemotherapy improves outcomes in patients with resectable or borderline resectable pancreatic cancer, preliminary findings from the phase 3 PREOPANC-1 trial suggest.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
Overall survival in 127 patients randomized to immediate surgery followed by adjuvant chemotherapy was 13.7 months vs. 17.1 months in 119 patients randomized to receive preoperative chemoradiotherapy and postoperative adjuvant chemotherapy, Geertjan van Tienhoven, MD, PhD, reported at the annual meeting of the American Society of Clinical Oncology.
The difference did not quite reach statistical significance, but final analysis requires an additional 26 events, Dr. van Tienhoven of Academic Medical Center, Amsterdam explained in a video interview at the meeting.
Other differences between the groups, which included disease-free survival, local control, and metastasis-free survival, did differ significantly in favor of preoperative chemotherapy, he said.
Of note, 72% and 62% of patients in the immediate surgery and preoperative chemoradiotherapy groups, respectively, underwent resection and a greater proportion of patients in the latter group achieved microscopically complete resection, he said (63% vs. 31%).
Should these results hold up in the final analysis, particularly if the difference in overall survival reaches statistical significance, “then this is a proof of principle and practice-changing trial,” Dr. van Tienhoven said.
Dr. van Tienhoven reported having no disclosures.
SOURCE: van Tienhoven et al. ASCO 2108, Abstract LBA4002.
CHICAGO – Preoperative chemotherapy improves outcomes in patients with resectable or borderline resectable pancreatic cancer, preliminary findings from the phase 3 PREOPANC-1 trial suggest.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
Overall survival in 127 patients randomized to immediate surgery followed by adjuvant chemotherapy was 13.7 months vs. 17.1 months in 119 patients randomized to receive preoperative chemoradiotherapy and postoperative adjuvant chemotherapy, Geertjan van Tienhoven, MD, PhD, reported at the annual meeting of the American Society of Clinical Oncology.
The difference did not quite reach statistical significance, but final analysis requires an additional 26 events, Dr. van Tienhoven of Academic Medical Center, Amsterdam explained in a video interview at the meeting.
Other differences between the groups, which included disease-free survival, local control, and metastasis-free survival, did differ significantly in favor of preoperative chemotherapy, he said.
Of note, 72% and 62% of patients in the immediate surgery and preoperative chemoradiotherapy groups, respectively, underwent resection and a greater proportion of patients in the latter group achieved microscopically complete resection, he said (63% vs. 31%).
Should these results hold up in the final analysis, particularly if the difference in overall survival reaches statistical significance, “then this is a proof of principle and practice-changing trial,” Dr. van Tienhoven said.
Dr. van Tienhoven reported having no disclosures.
SOURCE: van Tienhoven et al. ASCO 2108, Abstract LBA4002.
CHICAGO – Preoperative chemotherapy improves outcomes in patients with resectable or borderline resectable pancreatic cancer, preliminary findings from the phase 3 PREOPANC-1 trial suggest.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
Overall survival in 127 patients randomized to immediate surgery followed by adjuvant chemotherapy was 13.7 months vs. 17.1 months in 119 patients randomized to receive preoperative chemoradiotherapy and postoperative adjuvant chemotherapy, Geertjan van Tienhoven, MD, PhD, reported at the annual meeting of the American Society of Clinical Oncology.
The difference did not quite reach statistical significance, but final analysis requires an additional 26 events, Dr. van Tienhoven of Academic Medical Center, Amsterdam explained in a video interview at the meeting.
Other differences between the groups, which included disease-free survival, local control, and metastasis-free survival, did differ significantly in favor of preoperative chemotherapy, he said.
Of note, 72% and 62% of patients in the immediate surgery and preoperative chemoradiotherapy groups, respectively, underwent resection and a greater proportion of patients in the latter group achieved microscopically complete resection, he said (63% vs. 31%).
Should these results hold up in the final analysis, particularly if the difference in overall survival reaches statistical significance, “then this is a proof of principle and practice-changing trial,” Dr. van Tienhoven said.
Dr. van Tienhoven reported having no disclosures.
SOURCE: van Tienhoven et al. ASCO 2108, Abstract LBA4002.
REPORTING FROM ASCO 2018
What underlies post–bariatric surgery bone fragility?
BOSTON – Charting a healthy path for patients after bariatric surgery can be complicated and addressing bone health is an important part of the endocrinologist’s role in keeping patients safe from postsurgical fractures, according to John Bilezikian, MD.
said Dr. Bilezikian, speaking during a bariatric surgery–focused session at the annual scientific & clinical congress of the American Academy of Clinical Endocrinologists.
It’s not easy to assess bone health, even before surgery, said Dr. Bilezikian. Even objective measures of bone density, such as dual-energy x-ray absorptiometry (DXA), may be skewed: very high fat mass causes artifact that interferes with accurate measurement of bone density, and DXA can’t distinguish between cortical and trabecular bone. The latter is a particular issue in high body mass index patients, since obesity is known to be associated with a more fragile bone microarchitecture, said Dr. Bilezikian, the Dorothy L. and Daniel H. Silberberg Professor of Medicine and director of the metabolic bone diseases unit at Columbia University, New York.
With these caveats in mind, Dr. Bilezikian said, there are some lessons to be learned from existing research to better manage bone health in bariatric patients.
After Roux-en-Y gastric bypass surgery (RYGB), bone turnover soon increases, with bone resorption markers increasing by up to 200% in the first 12-18 months after surgery. Bone formation markers also are elevated but to a lesser extent, said Dr. Bilezikian. Over time, the weight loss from RYGB is associated with a significant drop in bone mineral density (BMD) at weight-bearing sites. Weight loss was associated with bone loss at the total hip (r = 0.70; P less than .0003) and femoral neck (r = 0.47; P = .03 (J Clin Endocrinol Metab. 2013 Feb;98[2] 541-9).
A newer-technology, high-resolution peripheral quantitative CT (HR-pQCT) offers a noninvasive look not just at bone size and density but also at microarchitecture, including cortical thickness and details of trabecular structure. This technology “can help elucidate the structural basis for fragility,” said Dr. Bilezikian.
HR-pQCT was used in a recent study (J Bone Min Res. 2017 Dec. 27. doi: 10.1002/jbmr.3371) that followed 48 patients for 1 year after RYGB. Using HR-pQCT, DXA, and serum markers of bone turnover, the researchers found significant decrease in BMD and estimated decrease in bone strength after RYGB. Bone cortex became increasingly porous as well. Taken together, these changes may indicate an increased fracture risk, concluded the investigators.
A longer study that followed RYGB recipients for 2 years and used similar imaging and serum parameters also found that participants had decreased BMD. Tellingly, these investigators saw more marked increase in cortical porosity in the second year after bypass. Estimated bone strength continued to decline during the study period, even after weight loss had stopped.
All of these findings, said Dr. Bilezikian, point to a pathogenetic process other than weight loss that promotes the deteriorating bone microarchitecture seen years after RYGB. “Loss of bone mass and skeletal deterioration after gastric bypass surgery cannot be explained by weight loss alone,” said Dr. Bilezikian.
Another recent study was able to follow a small cohort of patients for a full 5 years, using DXA, lumbar CT, and Hr-pQCT. Though weight loss stabilized after 2 years and 25-OH D and calcium levels were unchanged from presurgical baseline, bone density continued to drop, and bone microarchitecture further deteriorated, said Dr. Bilezikian (Greenblatt L et al. ASBMR 2017, Abstract 1125).
Initially, post–bariatric surgery weight loss may induce bone changes because of skeletal unloading; further down the road, estrogen production by adipose tissue is decreased with ongoing fat loss, and sarcopenia may have an adverse effect on bone microarchitecture. Postsurgical malabsorption may also be an early mechanism of bone loss.
Other hormonal changes can include secondary hyperparathyroidism. Leptin, adiponectin, and peptide YY levels also may be altered.
Do these changes in BMD and bone architecture result in increased fracture risk? This question is difficult to answer, for the same reasons that other bariatric surgery research can be challenging, said Dr. Bilezikian. There is heterogeneity of procedures and supplement regimens, sample sizes can be small, follow-up times short, and adherence often is not tracked.
However, there are some clues that RYGB may be associated with an increased risk of all fractures and of fragility fractures, with appendicular fractures seen most frequently (Osteoporos Int. 2014 Jan; 25[1]:151-8). A larger study that tracked 12,676 patients receiving bariatric surgery, 38,028 patients with obesity, and 126,760 nonobese participants found that the bariatric patients had a 4.1% risk of fracture at 4 years post surgery, compared with 2.7% and 2.4% fracture rates in the participants with and without obesity, respectively (BMJ. 2016;354:i3794).
Other retrospective studies have found “a time-dependent increase in nonvertebral fractures with Roux-en-Y gastric bypass compared to gastric banding,” said Dr. Bilezikian.
How can these risks be managed after gastric bypass surgery? “Strive for nutritional adequacy” as the first step, said Dr. Bilezikian, meaning that calcium and vitamin D should be prescribed – and adherence encouraged – as indicated. Levels of 25-OH D should be checked regularly, with supplementation managed to keep levels over 30 ng/mL, he said.
All patients should be encouraged to develop and maintain an appropriate exercise regimen, and BMD should be followed over time. Those caring for post–gastric bypass patients can still use a bisphosphonate or other bone-health medication, if indicated using standard parameters. However, “You probably shouldn’t use an oral bisphosphonate in this population,” said Dr. Bilezikian.
Dr. Bilezikian reported that he has consulting or advisory relationships with Amgen, Radius Pharmaceuticals, Shire Pharmaceuticals, and Ultragenyx, and serves on a data safety monitoring board for Regeneron.
BOSTON – Charting a healthy path for patients after bariatric surgery can be complicated and addressing bone health is an important part of the endocrinologist’s role in keeping patients safe from postsurgical fractures, according to John Bilezikian, MD.
said Dr. Bilezikian, speaking during a bariatric surgery–focused session at the annual scientific & clinical congress of the American Academy of Clinical Endocrinologists.
It’s not easy to assess bone health, even before surgery, said Dr. Bilezikian. Even objective measures of bone density, such as dual-energy x-ray absorptiometry (DXA), may be skewed: very high fat mass causes artifact that interferes with accurate measurement of bone density, and DXA can’t distinguish between cortical and trabecular bone. The latter is a particular issue in high body mass index patients, since obesity is known to be associated with a more fragile bone microarchitecture, said Dr. Bilezikian, the Dorothy L. and Daniel H. Silberberg Professor of Medicine and director of the metabolic bone diseases unit at Columbia University, New York.
With these caveats in mind, Dr. Bilezikian said, there are some lessons to be learned from existing research to better manage bone health in bariatric patients.
After Roux-en-Y gastric bypass surgery (RYGB), bone turnover soon increases, with bone resorption markers increasing by up to 200% in the first 12-18 months after surgery. Bone formation markers also are elevated but to a lesser extent, said Dr. Bilezikian. Over time, the weight loss from RYGB is associated with a significant drop in bone mineral density (BMD) at weight-bearing sites. Weight loss was associated with bone loss at the total hip (r = 0.70; P less than .0003) and femoral neck (r = 0.47; P = .03 (J Clin Endocrinol Metab. 2013 Feb;98[2] 541-9).
A newer-technology, high-resolution peripheral quantitative CT (HR-pQCT) offers a noninvasive look not just at bone size and density but also at microarchitecture, including cortical thickness and details of trabecular structure. This technology “can help elucidate the structural basis for fragility,” said Dr. Bilezikian.
HR-pQCT was used in a recent study (J Bone Min Res. 2017 Dec. 27. doi: 10.1002/jbmr.3371) that followed 48 patients for 1 year after RYGB. Using HR-pQCT, DXA, and serum markers of bone turnover, the researchers found significant decrease in BMD and estimated decrease in bone strength after RYGB. Bone cortex became increasingly porous as well. Taken together, these changes may indicate an increased fracture risk, concluded the investigators.
A longer study that followed RYGB recipients for 2 years and used similar imaging and serum parameters also found that participants had decreased BMD. Tellingly, these investigators saw more marked increase in cortical porosity in the second year after bypass. Estimated bone strength continued to decline during the study period, even after weight loss had stopped.
All of these findings, said Dr. Bilezikian, point to a pathogenetic process other than weight loss that promotes the deteriorating bone microarchitecture seen years after RYGB. “Loss of bone mass and skeletal deterioration after gastric bypass surgery cannot be explained by weight loss alone,” said Dr. Bilezikian.
Another recent study was able to follow a small cohort of patients for a full 5 years, using DXA, lumbar CT, and Hr-pQCT. Though weight loss stabilized after 2 years and 25-OH D and calcium levels were unchanged from presurgical baseline, bone density continued to drop, and bone microarchitecture further deteriorated, said Dr. Bilezikian (Greenblatt L et al. ASBMR 2017, Abstract 1125).
Initially, post–bariatric surgery weight loss may induce bone changes because of skeletal unloading; further down the road, estrogen production by adipose tissue is decreased with ongoing fat loss, and sarcopenia may have an adverse effect on bone microarchitecture. Postsurgical malabsorption may also be an early mechanism of bone loss.
Other hormonal changes can include secondary hyperparathyroidism. Leptin, adiponectin, and peptide YY levels also may be altered.
Do these changes in BMD and bone architecture result in increased fracture risk? This question is difficult to answer, for the same reasons that other bariatric surgery research can be challenging, said Dr. Bilezikian. There is heterogeneity of procedures and supplement regimens, sample sizes can be small, follow-up times short, and adherence often is not tracked.
However, there are some clues that RYGB may be associated with an increased risk of all fractures and of fragility fractures, with appendicular fractures seen most frequently (Osteoporos Int. 2014 Jan; 25[1]:151-8). A larger study that tracked 12,676 patients receiving bariatric surgery, 38,028 patients with obesity, and 126,760 nonobese participants found that the bariatric patients had a 4.1% risk of fracture at 4 years post surgery, compared with 2.7% and 2.4% fracture rates in the participants with and without obesity, respectively (BMJ. 2016;354:i3794).
Other retrospective studies have found “a time-dependent increase in nonvertebral fractures with Roux-en-Y gastric bypass compared to gastric banding,” said Dr. Bilezikian.
How can these risks be managed after gastric bypass surgery? “Strive for nutritional adequacy” as the first step, said Dr. Bilezikian, meaning that calcium and vitamin D should be prescribed – and adherence encouraged – as indicated. Levels of 25-OH D should be checked regularly, with supplementation managed to keep levels over 30 ng/mL, he said.
All patients should be encouraged to develop and maintain an appropriate exercise regimen, and BMD should be followed over time. Those caring for post–gastric bypass patients can still use a bisphosphonate or other bone-health medication, if indicated using standard parameters. However, “You probably shouldn’t use an oral bisphosphonate in this population,” said Dr. Bilezikian.
Dr. Bilezikian reported that he has consulting or advisory relationships with Amgen, Radius Pharmaceuticals, Shire Pharmaceuticals, and Ultragenyx, and serves on a data safety monitoring board for Regeneron.
BOSTON – Charting a healthy path for patients after bariatric surgery can be complicated and addressing bone health is an important part of the endocrinologist’s role in keeping patients safe from postsurgical fractures, according to John Bilezikian, MD.
said Dr. Bilezikian, speaking during a bariatric surgery–focused session at the annual scientific & clinical congress of the American Academy of Clinical Endocrinologists.
It’s not easy to assess bone health, even before surgery, said Dr. Bilezikian. Even objective measures of bone density, such as dual-energy x-ray absorptiometry (DXA), may be skewed: very high fat mass causes artifact that interferes with accurate measurement of bone density, and DXA can’t distinguish between cortical and trabecular bone. The latter is a particular issue in high body mass index patients, since obesity is known to be associated with a more fragile bone microarchitecture, said Dr. Bilezikian, the Dorothy L. and Daniel H. Silberberg Professor of Medicine and director of the metabolic bone diseases unit at Columbia University, New York.
With these caveats in mind, Dr. Bilezikian said, there are some lessons to be learned from existing research to better manage bone health in bariatric patients.
After Roux-en-Y gastric bypass surgery (RYGB), bone turnover soon increases, with bone resorption markers increasing by up to 200% in the first 12-18 months after surgery. Bone formation markers also are elevated but to a lesser extent, said Dr. Bilezikian. Over time, the weight loss from RYGB is associated with a significant drop in bone mineral density (BMD) at weight-bearing sites. Weight loss was associated with bone loss at the total hip (r = 0.70; P less than .0003) and femoral neck (r = 0.47; P = .03 (J Clin Endocrinol Metab. 2013 Feb;98[2] 541-9).
A newer-technology, high-resolution peripheral quantitative CT (HR-pQCT) offers a noninvasive look not just at bone size and density but also at microarchitecture, including cortical thickness and details of trabecular structure. This technology “can help elucidate the structural basis for fragility,” said Dr. Bilezikian.
HR-pQCT was used in a recent study (J Bone Min Res. 2017 Dec. 27. doi: 10.1002/jbmr.3371) that followed 48 patients for 1 year after RYGB. Using HR-pQCT, DXA, and serum markers of bone turnover, the researchers found significant decrease in BMD and estimated decrease in bone strength after RYGB. Bone cortex became increasingly porous as well. Taken together, these changes may indicate an increased fracture risk, concluded the investigators.
A longer study that followed RYGB recipients for 2 years and used similar imaging and serum parameters also found that participants had decreased BMD. Tellingly, these investigators saw more marked increase in cortical porosity in the second year after bypass. Estimated bone strength continued to decline during the study period, even after weight loss had stopped.
All of these findings, said Dr. Bilezikian, point to a pathogenetic process other than weight loss that promotes the deteriorating bone microarchitecture seen years after RYGB. “Loss of bone mass and skeletal deterioration after gastric bypass surgery cannot be explained by weight loss alone,” said Dr. Bilezikian.
Another recent study was able to follow a small cohort of patients for a full 5 years, using DXA, lumbar CT, and Hr-pQCT. Though weight loss stabilized after 2 years and 25-OH D and calcium levels were unchanged from presurgical baseline, bone density continued to drop, and bone microarchitecture further deteriorated, said Dr. Bilezikian (Greenblatt L et al. ASBMR 2017, Abstract 1125).
Initially, post–bariatric surgery weight loss may induce bone changes because of skeletal unloading; further down the road, estrogen production by adipose tissue is decreased with ongoing fat loss, and sarcopenia may have an adverse effect on bone microarchitecture. Postsurgical malabsorption may also be an early mechanism of bone loss.
Other hormonal changes can include secondary hyperparathyroidism. Leptin, adiponectin, and peptide YY levels also may be altered.
Do these changes in BMD and bone architecture result in increased fracture risk? This question is difficult to answer, for the same reasons that other bariatric surgery research can be challenging, said Dr. Bilezikian. There is heterogeneity of procedures and supplement regimens, sample sizes can be small, follow-up times short, and adherence often is not tracked.
However, there are some clues that RYGB may be associated with an increased risk of all fractures and of fragility fractures, with appendicular fractures seen most frequently (Osteoporos Int. 2014 Jan; 25[1]:151-8). A larger study that tracked 12,676 patients receiving bariatric surgery, 38,028 patients with obesity, and 126,760 nonobese participants found that the bariatric patients had a 4.1% risk of fracture at 4 years post surgery, compared with 2.7% and 2.4% fracture rates in the participants with and without obesity, respectively (BMJ. 2016;354:i3794).
Other retrospective studies have found “a time-dependent increase in nonvertebral fractures with Roux-en-Y gastric bypass compared to gastric banding,” said Dr. Bilezikian.
How can these risks be managed after gastric bypass surgery? “Strive for nutritional adequacy” as the first step, said Dr. Bilezikian, meaning that calcium and vitamin D should be prescribed – and adherence encouraged – as indicated. Levels of 25-OH D should be checked regularly, with supplementation managed to keep levels over 30 ng/mL, he said.
All patients should be encouraged to develop and maintain an appropriate exercise regimen, and BMD should be followed over time. Those caring for post–gastric bypass patients can still use a bisphosphonate or other bone-health medication, if indicated using standard parameters. However, “You probably shouldn’t use an oral bisphosphonate in this population,” said Dr. Bilezikian.
Dr. Bilezikian reported that he has consulting or advisory relationships with Amgen, Radius Pharmaceuticals, Shire Pharmaceuticals, and Ultragenyx, and serves on a data safety monitoring board for Regeneron.
REPORTING FROM AACE 2018
Hemostatic clipping cuts bleeds after large polyp removal
WASHINGTON –
“The benefit appears limited to proximal polyps,” Heiko Pohl, MD, said at the annual Digestive Disease Week®. In that prespecified subgroup, which included two-thirds of enrolled patients, placement of hemostatic clips on defects left after removing polyps 20 mm in diameter or larger cut the rate of delayed, severe bleeding by two-thirds, compared with patients with large defects not treated with clips. This result represented a number needed to treat with clips of 15 patients with large proximal polyps to prevent one episode of delayed severe bleeding, said Dr. Pohl, a gastroenterologist at the VA Medical Center in White River Junction, Vt.
Although the results that Dr. Pohl reported came from a trial that originally had been designed to generate data for Food and Drug Administration approval for using the clips to close defects following large polyp removal, the clips received approval for this indication from the agency in 2016 while the study was still in progress.
But Dr. Pohl maintained that the new evidence for efficacy that he reported will provide further impetus for gastroenterologists to use clips when they remove larger polyps in proximal locations. “I think this study will help standardize treatment of mucosal resections and change clip use,” he said in an interview.
“This was a terrific study, and one that needed to be done,” commented John R. Saltzman, MD, professor of medicine at Harvard Medical School and director of endoscopy at Brigham and Women’s Hospital in Boston. But Dr. Saltzman, who spoke from the floor during discussion of Dr. Pohl’s report, added that data on the average number of clips required to close defects were needed to assess the cost-effectiveness of the treatment, data that Dr. Pohl said were available but still being analyzed.
“We have to know how many clips to use and how to close the polyp,” Dr. Saltzman said. Dr. Pohl estimated that roughly four or five clips had been used per defect, but he cautioned that this estimate was preliminary pending his complete analysis of the data.
The CLIP (Clip Closure After Endoscopic Resection of Large Polyps) study enrolled patients with at least one nonpedunculated colonic polyp that was at least 20 mm in diameter at 16 U.S. centers, as well as one center in Montreal and one in Barcelona. The patients averaged 65 years of age, and 6%-7% of patients had more than one large polyp removed during their procedure. Randomization produced one important imbalance in assignment: 25% of the 454 patients in the clipped arm were on an antithrombotic drug (either an anticoagulant or antiplatelet drug) at the time of their endoscopy, compared with 33% of the 464 patients in the control arm.
The study’s primary endpoint was the incidence of “severe” bleeding within 30 days after the procedure. The study defined severe bleeding as an event that required hospitalization, need for repeat endoscopy, need for a blood transfusion, or need for any other major intervention, explained Dr. Pohl, who is also on the staff of Dartmouth-Hitchcock Medical Center in Lebanon, N.H.
Such events occurred in 3.5% of the patients who underwent clipping and in 7.3% of control patients who received no clipping, a statistically significant difference (P = .01). Among patients with proximal polyps, the bleeding rates were 3.3% among clipped patients and 9.9% among controls, also a statistically significant difference. Among patients with distal polyps the bleeding rates were 4.0% among clipped patients and 1.4% among controls, a difference that was not statistically significant.
Dr. Pohl and his associates ran three other prespecified, secondary analyses that divided the enrolled patients into subgroups. These analyses showed no significant effect on outcome by polyp size when comparing 20-39 mm polyps with polyps 40 mm or larger, treatment with an antithrombotic drug, or method of cauterization. The median time to severe bleeding was 1 day among the controls and 7 days among the clipped patients.
Aside from the difference in rates of delayed bleeding, the two study arms showed no significant differences in the incidence of any other serious postprocedure events. The rates of these nonbleeding events were 1.3% among clipped patients and 2.4% among the controls.
The researchers ran all these analyses based on the intention-to-treat assignment of patients. However, during the study, 9% of patients assigned to the control arm crossed over ended up receiving clips during their procedure after all, a rate that Dr. Pohl called “surprisingly high,” whereas 14% of patients assigned to the clip arm never received clips. A per-protocol analysis that censored patients who did not receive their assigned treatment showed that, among the remaining patients who underwent their assigned treatment, the rate of delayed, severe bleeds was 2.3% among the 390 patients actually treated with clips and 7.2% among the 419 controls who never received clips, a statistically significant difference, he reported.
Dr. Pohl also noted that it was “somewhat surprising” that clipping appeared to result in complete closure in “only” 68% of patients who underwent clipping and that it produced partial closure in an additional 20% of patients, with the remaining patients having mucosal defects that were not considered closed by clipping.
The study was funded by Boston Scientific, the company that markets the hemostatic clip (Resolution 360) tested in the study. Dr. Pohl had no additional disclosures. Dr. Saltzman had no disclosures.
SOURCE: Pohl H et al. Digestive Disease Week, Presentation 886.
WASHINGTON –
“The benefit appears limited to proximal polyps,” Heiko Pohl, MD, said at the annual Digestive Disease Week®. In that prespecified subgroup, which included two-thirds of enrolled patients, placement of hemostatic clips on defects left after removing polyps 20 mm in diameter or larger cut the rate of delayed, severe bleeding by two-thirds, compared with patients with large defects not treated with clips. This result represented a number needed to treat with clips of 15 patients with large proximal polyps to prevent one episode of delayed severe bleeding, said Dr. Pohl, a gastroenterologist at the VA Medical Center in White River Junction, Vt.
Although the results that Dr. Pohl reported came from a trial that originally had been designed to generate data for Food and Drug Administration approval for using the clips to close defects following large polyp removal, the clips received approval for this indication from the agency in 2016 while the study was still in progress.
But Dr. Pohl maintained that the new evidence for efficacy that he reported will provide further impetus for gastroenterologists to use clips when they remove larger polyps in proximal locations. “I think this study will help standardize treatment of mucosal resections and change clip use,” he said in an interview.
“This was a terrific study, and one that needed to be done,” commented John R. Saltzman, MD, professor of medicine at Harvard Medical School and director of endoscopy at Brigham and Women’s Hospital in Boston. But Dr. Saltzman, who spoke from the floor during discussion of Dr. Pohl’s report, added that data on the average number of clips required to close defects were needed to assess the cost-effectiveness of the treatment, data that Dr. Pohl said were available but still being analyzed.
“We have to know how many clips to use and how to close the polyp,” Dr. Saltzman said. Dr. Pohl estimated that roughly four or five clips had been used per defect, but he cautioned that this estimate was preliminary pending his complete analysis of the data.
The CLIP (Clip Closure After Endoscopic Resection of Large Polyps) study enrolled patients with at least one nonpedunculated colonic polyp that was at least 20 mm in diameter at 16 U.S. centers, as well as one center in Montreal and one in Barcelona. The patients averaged 65 years of age, and 6%-7% of patients had more than one large polyp removed during their procedure. Randomization produced one important imbalance in assignment: 25% of the 454 patients in the clipped arm were on an antithrombotic drug (either an anticoagulant or antiplatelet drug) at the time of their endoscopy, compared with 33% of the 464 patients in the control arm.
The study’s primary endpoint was the incidence of “severe” bleeding within 30 days after the procedure. The study defined severe bleeding as an event that required hospitalization, need for repeat endoscopy, need for a blood transfusion, or need for any other major intervention, explained Dr. Pohl, who is also on the staff of Dartmouth-Hitchcock Medical Center in Lebanon, N.H.
Such events occurred in 3.5% of the patients who underwent clipping and in 7.3% of control patients who received no clipping, a statistically significant difference (P = .01). Among patients with proximal polyps, the bleeding rates were 3.3% among clipped patients and 9.9% among controls, also a statistically significant difference. Among patients with distal polyps the bleeding rates were 4.0% among clipped patients and 1.4% among controls, a difference that was not statistically significant.
Dr. Pohl and his associates ran three other prespecified, secondary analyses that divided the enrolled patients into subgroups. These analyses showed no significant effect on outcome by polyp size when comparing 20-39 mm polyps with polyps 40 mm or larger, treatment with an antithrombotic drug, or method of cauterization. The median time to severe bleeding was 1 day among the controls and 7 days among the clipped patients.
Aside from the difference in rates of delayed bleeding, the two study arms showed no significant differences in the incidence of any other serious postprocedure events. The rates of these nonbleeding events were 1.3% among clipped patients and 2.4% among the controls.
The researchers ran all these analyses based on the intention-to-treat assignment of patients. However, during the study, 9% of patients assigned to the control arm crossed over ended up receiving clips during their procedure after all, a rate that Dr. Pohl called “surprisingly high,” whereas 14% of patients assigned to the clip arm never received clips. A per-protocol analysis that censored patients who did not receive their assigned treatment showed that, among the remaining patients who underwent their assigned treatment, the rate of delayed, severe bleeds was 2.3% among the 390 patients actually treated with clips and 7.2% among the 419 controls who never received clips, a statistically significant difference, he reported.
Dr. Pohl also noted that it was “somewhat surprising” that clipping appeared to result in complete closure in “only” 68% of patients who underwent clipping and that it produced partial closure in an additional 20% of patients, with the remaining patients having mucosal defects that were not considered closed by clipping.
The study was funded by Boston Scientific, the company that markets the hemostatic clip (Resolution 360) tested in the study. Dr. Pohl had no additional disclosures. Dr. Saltzman had no disclosures.
SOURCE: Pohl H et al. Digestive Disease Week, Presentation 886.
WASHINGTON –
“The benefit appears limited to proximal polyps,” Heiko Pohl, MD, said at the annual Digestive Disease Week®. In that prespecified subgroup, which included two-thirds of enrolled patients, placement of hemostatic clips on defects left after removing polyps 20 mm in diameter or larger cut the rate of delayed, severe bleeding by two-thirds, compared with patients with large defects not treated with clips. This result represented a number needed to treat with clips of 15 patients with large proximal polyps to prevent one episode of delayed severe bleeding, said Dr. Pohl, a gastroenterologist at the VA Medical Center in White River Junction, Vt.
Although the results that Dr. Pohl reported came from a trial that originally had been designed to generate data for Food and Drug Administration approval for using the clips to close defects following large polyp removal, the clips received approval for this indication from the agency in 2016 while the study was still in progress.
But Dr. Pohl maintained that the new evidence for efficacy that he reported will provide further impetus for gastroenterologists to use clips when they remove larger polyps in proximal locations. “I think this study will help standardize treatment of mucosal resections and change clip use,” he said in an interview.
“This was a terrific study, and one that needed to be done,” commented John R. Saltzman, MD, professor of medicine at Harvard Medical School and director of endoscopy at Brigham and Women’s Hospital in Boston. But Dr. Saltzman, who spoke from the floor during discussion of Dr. Pohl’s report, added that data on the average number of clips required to close defects were needed to assess the cost-effectiveness of the treatment, data that Dr. Pohl said were available but still being analyzed.
“We have to know how many clips to use and how to close the polyp,” Dr. Saltzman said. Dr. Pohl estimated that roughly four or five clips had been used per defect, but he cautioned that this estimate was preliminary pending his complete analysis of the data.
The CLIP (Clip Closure After Endoscopic Resection of Large Polyps) study enrolled patients with at least one nonpedunculated colonic polyp that was at least 20 mm in diameter at 16 U.S. centers, as well as one center in Montreal and one in Barcelona. The patients averaged 65 years of age, and 6%-7% of patients had more than one large polyp removed during their procedure. Randomization produced one important imbalance in assignment: 25% of the 454 patients in the clipped arm were on an antithrombotic drug (either an anticoagulant or antiplatelet drug) at the time of their endoscopy, compared with 33% of the 464 patients in the control arm.
The study’s primary endpoint was the incidence of “severe” bleeding within 30 days after the procedure. The study defined severe bleeding as an event that required hospitalization, need for repeat endoscopy, need for a blood transfusion, or need for any other major intervention, explained Dr. Pohl, who is also on the staff of Dartmouth-Hitchcock Medical Center in Lebanon, N.H.
Such events occurred in 3.5% of the patients who underwent clipping and in 7.3% of control patients who received no clipping, a statistically significant difference (P = .01). Among patients with proximal polyps, the bleeding rates were 3.3% among clipped patients and 9.9% among controls, also a statistically significant difference. Among patients with distal polyps the bleeding rates were 4.0% among clipped patients and 1.4% among controls, a difference that was not statistically significant.
Dr. Pohl and his associates ran three other prespecified, secondary analyses that divided the enrolled patients into subgroups. These analyses showed no significant effect on outcome by polyp size when comparing 20-39 mm polyps with polyps 40 mm or larger, treatment with an antithrombotic drug, or method of cauterization. The median time to severe bleeding was 1 day among the controls and 7 days among the clipped patients.
Aside from the difference in rates of delayed bleeding, the two study arms showed no significant differences in the incidence of any other serious postprocedure events. The rates of these nonbleeding events were 1.3% among clipped patients and 2.4% among the controls.
The researchers ran all these analyses based on the intention-to-treat assignment of patients. However, during the study, 9% of patients assigned to the control arm crossed over ended up receiving clips during their procedure after all, a rate that Dr. Pohl called “surprisingly high,” whereas 14% of patients assigned to the clip arm never received clips. A per-protocol analysis that censored patients who did not receive their assigned treatment showed that, among the remaining patients who underwent their assigned treatment, the rate of delayed, severe bleeds was 2.3% among the 390 patients actually treated with clips and 7.2% among the 419 controls who never received clips, a statistically significant difference, he reported.
Dr. Pohl also noted that it was “somewhat surprising” that clipping appeared to result in complete closure in “only” 68% of patients who underwent clipping and that it produced partial closure in an additional 20% of patients, with the remaining patients having mucosal defects that were not considered closed by clipping.
The study was funded by Boston Scientific, the company that markets the hemostatic clip (Resolution 360) tested in the study. Dr. Pohl had no additional disclosures. Dr. Saltzman had no disclosures.
SOURCE: Pohl H et al. Digestive Disease Week, Presentation 886.
REPORTING FROM DDW 2018
Key clinical point: Hemostatic wound clipping after large polyp removal cuts delayed bleeding, especially for proximal polyps.
Major finding: The incidence of severe, delayed bleeds was 3.5% among clipped patients and 7.3% among controls.
Study details: The CLIP study, a multicenter, randomized trial with 918 patients.
Disclosures: The study was funded by Boston Scientific, the company that markets the hemostatic clip (Resolution 360) tested in the study. Dr. Pohl had no additional disclosures. Dr. Saltzman had no disclosures.
Source: Pohl H et al. Digestive Disease Week, Presentation 886.
A new way to classify endometrial cancer
We classify endometrial cancer so that we can communicate and define each patient’s disease status, the potential for harm, and the likelihood that adjuvant therapies might provide help. Traditional forms of classification have clearly fallen short in achieving this aim, as we all know of patients with apparent low-risk disease (such as stage IA grade 1 endometrioid carcinoma) who have had recurrences and died from their disease, and we know that many patients have been subjected to overtreatment for their cancer and have acquired lifelong toxicities of therapy. This column will explore the newer, more sophisticated molecular-based classifications that are being validated for endometrial cancer, and the ways in which this promises to personalize the treatment of endometrial cancer.
Breast cancer and melanoma are examples of the inclusion of molecular data such as hormone receptor status, HER2/neu status, or BRAF positivity resulting in advancements in personalizing therapeutics. We are now moving toward this for endometrial cancer.
What is the Cancer Genome Atlas?
In 2006 the National Institutes of Health announced an initiative to coordinate work between the National Cancer Institute and the National Human Genome Research Institute taking information about the human genome and analyzing it for key genomic alterations found in 33 common cancers. These data were combined with clinical information (such as survival) to classify the behaviors of those cancers with respect to their individual genomic alternations, in order to look for patterns in mutations and behaviors. The goal of this analysis was to shift the paradigm of cancer classification from being centered around primary organ site toward tumors’ shared genomic patterns.
In 2013 the Cancer Genome Atlas published their results of complete gene sequencing in endometrial cancer.3 The authors identified four discrete subgroups of endometrial cancer with distinct molecular mutational profiles and distinct clinical outcomes: polymerase epsilon (POLE, pronounced “pole-ee”) ultramutated, microsatellite instability (MSI) high, copy number high, and copy number low.
POLE ultramutated
An important subgroup identified in the Cancer Genome Atlas was a group of patients with a POLE ultramutated state. POLE encodes for a subunit of DNA polymerase, the enzyme responsible for replicating the leading DNA strand. Nonfunctioning POLE results in proofreading errors and a subsequent ultramutated cellular state with a predominance of single nucleotide variants. POLE proofreading domain mutations in endometrial cancer and colon cancer are associated with excellent prognosis, likely secondary to the immune response that is elicited by this ultramutated state from creation of “antigenic neoepitopes” that stimulate T-cell response. Effectively, the very mutated cell is seen as “more foreign” to the body’s immune system.
Approximately 10% of patients with endometrial cancer have a POLE ultramutated state, and, as stated above, prognosis is excellent, even if coexisting with a histologic cell type (such as serous) that is normally associated with adverse outcomes. These women tend to be younger, with a lower body mass index, higher-grade endometrioid cell type, the presence of lymphovascular space invasion, and low stage.
MSI high
MSI (microsatellite instability) is a result of epigenetic/hypermethylations or loss of expression in mismatch repair genes (such as MLH1, MSH2, MSH6, PMS2). These genes code for proteins critical in the repair of mismatches in short repeated sequences of DNA. Loss of their function results in an accumulation of errors in these sequences: MSI. It is a feature of the Lynch syndrome inherited state, but is also found sporadically in endometrial tumors. These tumors accumulate a number of mutations during cell replication that, as in POLE hypermutated tumors, are associated with eliciting an immune response.
These tumors tend to be associated with a higher-grade endometrioid cell type, the presence of lymphovascular space invasion, and an advanced stage. Patients with tumors that have been described as MSI high are candidates for “immune therapy” with the PDL1 inhibitor pembrolizumab because of their proinflammatory state and observed favorable responses in clinical trials.4
Copy number high/low
Copy number (CN) high and low refers to the results of microarrays in which hierarchical clustering was applied to identify reoccurring amplification or deletion regions. The CN-high group was associated with the poorest outcomes (recurrence and survival). There is significant overlap with mutations in TP53. Most serous carcinomas were CN high; however, 25% of patients with high-grade endometrioid cell type shared the CN-high classification. These tumors shared great molecular similarity to high-grade serous ovarian cancers and basal-like breast cancer.
Those patients who did not possess mutations that classified them as POLE hypermutated, MSI high, or CN high were classified as CN low. This group included predominantly grades 1 and 2 endometrioid adenocarcinomas of an early stage and had a favorable prognostic profile, though less favorable than those with a POLE ultramutated state, which appears to be somewhat protective.
Molecular/metabolic interactions
While molecular data are clearly important in driving a cancer cell’s behavior, other clinical and metabolic factors influence cancer behavior. For example, body mass index, adiposity, glucose, and lipid metabolism have been shown to be important drivers of cellular behavior and responsiveness to targeted therapies.5,6 Additionally age, race, and other metabolic states contribute to oncologic behavior. Future classifications of endometrial cancer are unlikely to use molecular profiles in isolation but will need to incorporate these additional patient-specific data to better predict and prognosticate outcomes.
Clinical applications
If researchers can better define and describe a patient’s endometrial cancer from the time of their biopsy, important clinical decisions might be able to be tackled. For example, in a premenopausal patient with an endometrial cancer who is considering fertility-sparing treatments, preoperative knowledge of a POLE ultramutated state (and therefore an anticipated good prognosis) might favor fertility preservation or avoid comprehensive staging which may be of limited value. Similarly, if an MSI-high profile is identified leading to a Lynch syndrome diagnosis, she may be more inclined to undergo a hysterectomy with bilateral salpingo-oophorectomy and staging as she is at known increased risk for a more advanced endometrial cancer, as well as the potential for ovarian cancer.
Postoperative incorporation of molecular data promises to be particularly helpful in guiding adjuvant therapies and sparing some women from unnecessary treatments. For example, women with high-grade endometrioid tumors who are CN high were historically treated with radiotherapy but might do better treated with systemic adjuvant therapies traditionally reserved for nonendometrioid carcinomas. Costly therapies such as immunotherapy can be directed toward those with MSI-high tumors, and the rare patient with a POLE ultramutated state who has a recurrence or advanced disease. Clinical trials will be able to cluster enrollment of patients with CN-high, serouslike cancers with those with serous cancers, rather than combining them with patients whose cancers predictably behave much differently.
Much work is still needed to validate this molecular profiling in endometrial cancer and define the algorithms associated with treatment decisions; however, it is likely that the way we describe endometrial cancer in the near future will be quite different.
Dr. Rossi is an assistant professor in the division of gynecologic oncology at the University of North Carolina at Chapel Hill. She has no disclosures.
References
1. Bokhman JV. Two pathogenetic types of endometrial carcinoma. Gynecol Oncol. 1983;15(1):10-7.
2. Clarke BA et al. Endometrial carcinoma: controversies in histopathological assessment of grade and tumour cell type. J Clin Pathol. 2010;63(5):410-5.
3. Cancer Genome Atlas Research Network. Integrated genomic characterization of endometrial carcinoma. Nature. 2013;497(7447):67-73.
4. Ott PA et al. Pembrolizumab in advanced endometrial cancer: Preliminary results from the phase Ib KEYNOTE-028 study. J Clin Oncol. 2016;34(suppl):Abstract 5581.
5. Roque DR et al. Association between differential gene expression and body mass index among endometrial cancers from the Cancer Genome Atlas Project. Gynecol Oncol. 2016;142(2):317-22.
6. Talhouk A et al. New classification of endometrial cancers: The development and potential applications of genomic-based classification in research and clinical care. Gynecol Oncol Res Pract. 2016 Dec;3:14.
We classify endometrial cancer so that we can communicate and define each patient’s disease status, the potential for harm, and the likelihood that adjuvant therapies might provide help. Traditional forms of classification have clearly fallen short in achieving this aim, as we all know of patients with apparent low-risk disease (such as stage IA grade 1 endometrioid carcinoma) who have had recurrences and died from their disease, and we know that many patients have been subjected to overtreatment for their cancer and have acquired lifelong toxicities of therapy. This column will explore the newer, more sophisticated molecular-based classifications that are being validated for endometrial cancer, and the ways in which this promises to personalize the treatment of endometrial cancer.
Breast cancer and melanoma are examples of the inclusion of molecular data such as hormone receptor status, HER2/neu status, or BRAF positivity resulting in advancements in personalizing therapeutics. We are now moving toward this for endometrial cancer.
What is the Cancer Genome Atlas?
In 2006 the National Institutes of Health announced an initiative to coordinate work between the National Cancer Institute and the National Human Genome Research Institute taking information about the human genome and analyzing it for key genomic alterations found in 33 common cancers. These data were combined with clinical information (such as survival) to classify the behaviors of those cancers with respect to their individual genomic alternations, in order to look for patterns in mutations and behaviors. The goal of this analysis was to shift the paradigm of cancer classification from being centered around primary organ site toward tumors’ shared genomic patterns.
In 2013 the Cancer Genome Atlas published their results of complete gene sequencing in endometrial cancer.3 The authors identified four discrete subgroups of endometrial cancer with distinct molecular mutational profiles and distinct clinical outcomes: polymerase epsilon (POLE, pronounced “pole-ee”) ultramutated, microsatellite instability (MSI) high, copy number high, and copy number low.
POLE ultramutated
An important subgroup identified in the Cancer Genome Atlas was a group of patients with a POLE ultramutated state. POLE encodes for a subunit of DNA polymerase, the enzyme responsible for replicating the leading DNA strand. Nonfunctioning POLE results in proofreading errors and a subsequent ultramutated cellular state with a predominance of single nucleotide variants. POLE proofreading domain mutations in endometrial cancer and colon cancer are associated with excellent prognosis, likely secondary to the immune response that is elicited by this ultramutated state from creation of “antigenic neoepitopes” that stimulate T-cell response. Effectively, the very mutated cell is seen as “more foreign” to the body’s immune system.
Approximately 10% of patients with endometrial cancer have a POLE ultramutated state, and, as stated above, prognosis is excellent, even if coexisting with a histologic cell type (such as serous) that is normally associated with adverse outcomes. These women tend to be younger, with a lower body mass index, higher-grade endometrioid cell type, the presence of lymphovascular space invasion, and low stage.
MSI high
MSI (microsatellite instability) is a result of epigenetic/hypermethylations or loss of expression in mismatch repair genes (such as MLH1, MSH2, MSH6, PMS2). These genes code for proteins critical in the repair of mismatches in short repeated sequences of DNA. Loss of their function results in an accumulation of errors in these sequences: MSI. It is a feature of the Lynch syndrome inherited state, but is also found sporadically in endometrial tumors. These tumors accumulate a number of mutations during cell replication that, as in POLE hypermutated tumors, are associated with eliciting an immune response.
These tumors tend to be associated with a higher-grade endometrioid cell type, the presence of lymphovascular space invasion, and an advanced stage. Patients with tumors that have been described as MSI high are candidates for “immune therapy” with the PDL1 inhibitor pembrolizumab because of their proinflammatory state and observed favorable responses in clinical trials.4
Copy number high/low
Copy number (CN) high and low refers to the results of microarrays in which hierarchical clustering was applied to identify reoccurring amplification or deletion regions. The CN-high group was associated with the poorest outcomes (recurrence and survival). There is significant overlap with mutations in TP53. Most serous carcinomas were CN high; however, 25% of patients with high-grade endometrioid cell type shared the CN-high classification. These tumors shared great molecular similarity to high-grade serous ovarian cancers and basal-like breast cancer.
Those patients who did not possess mutations that classified them as POLE hypermutated, MSI high, or CN high were classified as CN low. This group included predominantly grades 1 and 2 endometrioid adenocarcinomas of an early stage and had a favorable prognostic profile, though less favorable than those with a POLE ultramutated state, which appears to be somewhat protective.
Molecular/metabolic interactions
While molecular data are clearly important in driving a cancer cell’s behavior, other clinical and metabolic factors influence cancer behavior. For example, body mass index, adiposity, glucose, and lipid metabolism have been shown to be important drivers of cellular behavior and responsiveness to targeted therapies.5,6 Additionally age, race, and other metabolic states contribute to oncologic behavior. Future classifications of endometrial cancer are unlikely to use molecular profiles in isolation but will need to incorporate these additional patient-specific data to better predict and prognosticate outcomes.
Clinical applications
If researchers can better define and describe a patient’s endometrial cancer from the time of their biopsy, important clinical decisions might be able to be tackled. For example, in a premenopausal patient with an endometrial cancer who is considering fertility-sparing treatments, preoperative knowledge of a POLE ultramutated state (and therefore an anticipated good prognosis) might favor fertility preservation or avoid comprehensive staging which may be of limited value. Similarly, if an MSI-high profile is identified leading to a Lynch syndrome diagnosis, she may be more inclined to undergo a hysterectomy with bilateral salpingo-oophorectomy and staging as she is at known increased risk for a more advanced endometrial cancer, as well as the potential for ovarian cancer.
Postoperative incorporation of molecular data promises to be particularly helpful in guiding adjuvant therapies and sparing some women from unnecessary treatments. For example, women with high-grade endometrioid tumors who are CN high were historically treated with radiotherapy but might do better treated with systemic adjuvant therapies traditionally reserved for nonendometrioid carcinomas. Costly therapies such as immunotherapy can be directed toward those with MSI-high tumors, and the rare patient with a POLE ultramutated state who has a recurrence or advanced disease. Clinical trials will be able to cluster enrollment of patients with CN-high, serouslike cancers with those with serous cancers, rather than combining them with patients whose cancers predictably behave much differently.
Much work is still needed to validate this molecular profiling in endometrial cancer and define the algorithms associated with treatment decisions; however, it is likely that the way we describe endometrial cancer in the near future will be quite different.
Dr. Rossi is an assistant professor in the division of gynecologic oncology at the University of North Carolina at Chapel Hill. She has no disclosures.
References
1. Bokhman JV. Two pathogenetic types of endometrial carcinoma. Gynecol Oncol. 1983;15(1):10-7.
2. Clarke BA et al. Endometrial carcinoma: controversies in histopathological assessment of grade and tumour cell type. J Clin Pathol. 2010;63(5):410-5.
3. Cancer Genome Atlas Research Network. Integrated genomic characterization of endometrial carcinoma. Nature. 2013;497(7447):67-73.
4. Ott PA et al. Pembrolizumab in advanced endometrial cancer: Preliminary results from the phase Ib KEYNOTE-028 study. J Clin Oncol. 2016;34(suppl):Abstract 5581.
5. Roque DR et al. Association between differential gene expression and body mass index among endometrial cancers from the Cancer Genome Atlas Project. Gynecol Oncol. 2016;142(2):317-22.
6. Talhouk A et al. New classification of endometrial cancers: The development and potential applications of genomic-based classification in research and clinical care. Gynecol Oncol Res Pract. 2016 Dec;3:14.
We classify endometrial cancer so that we can communicate and define each patient’s disease status, the potential for harm, and the likelihood that adjuvant therapies might provide help. Traditional forms of classification have clearly fallen short in achieving this aim, as we all know of patients with apparent low-risk disease (such as stage IA grade 1 endometrioid carcinoma) who have had recurrences and died from their disease, and we know that many patients have been subjected to overtreatment for their cancer and have acquired lifelong toxicities of therapy. This column will explore the newer, more sophisticated molecular-based classifications that are being validated for endometrial cancer, and the ways in which this promises to personalize the treatment of endometrial cancer.
Breast cancer and melanoma are examples of the inclusion of molecular data such as hormone receptor status, HER2/neu status, or BRAF positivity resulting in advancements in personalizing therapeutics. We are now moving toward this for endometrial cancer.
What is the Cancer Genome Atlas?
In 2006 the National Institutes of Health announced an initiative to coordinate work between the National Cancer Institute and the National Human Genome Research Institute taking information about the human genome and analyzing it for key genomic alterations found in 33 common cancers. These data were combined with clinical information (such as survival) to classify the behaviors of those cancers with respect to their individual genomic alternations, in order to look for patterns in mutations and behaviors. The goal of this analysis was to shift the paradigm of cancer classification from being centered around primary organ site toward tumors’ shared genomic patterns.
In 2013 the Cancer Genome Atlas published their results of complete gene sequencing in endometrial cancer.3 The authors identified four discrete subgroups of endometrial cancer with distinct molecular mutational profiles and distinct clinical outcomes: polymerase epsilon (POLE, pronounced “pole-ee”) ultramutated, microsatellite instability (MSI) high, copy number high, and copy number low.
POLE ultramutated
An important subgroup identified in the Cancer Genome Atlas was a group of patients with a POLE ultramutated state. POLE encodes for a subunit of DNA polymerase, the enzyme responsible for replicating the leading DNA strand. Nonfunctioning POLE results in proofreading errors and a subsequent ultramutated cellular state with a predominance of single nucleotide variants. POLE proofreading domain mutations in endometrial cancer and colon cancer are associated with excellent prognosis, likely secondary to the immune response that is elicited by this ultramutated state from creation of “antigenic neoepitopes” that stimulate T-cell response. Effectively, the very mutated cell is seen as “more foreign” to the body’s immune system.
Approximately 10% of patients with endometrial cancer have a POLE ultramutated state, and, as stated above, prognosis is excellent, even if coexisting with a histologic cell type (such as serous) that is normally associated with adverse outcomes. These women tend to be younger, with a lower body mass index, higher-grade endometrioid cell type, the presence of lymphovascular space invasion, and low stage.
MSI high
MSI (microsatellite instability) is a result of epigenetic/hypermethylations or loss of expression in mismatch repair genes (such as MLH1, MSH2, MSH6, PMS2). These genes code for proteins critical in the repair of mismatches in short repeated sequences of DNA. Loss of their function results in an accumulation of errors in these sequences: MSI. It is a feature of the Lynch syndrome inherited state, but is also found sporadically in endometrial tumors. These tumors accumulate a number of mutations during cell replication that, as in POLE hypermutated tumors, are associated with eliciting an immune response.
These tumors tend to be associated with a higher-grade endometrioid cell type, the presence of lymphovascular space invasion, and an advanced stage. Patients with tumors that have been described as MSI high are candidates for “immune therapy” with the PDL1 inhibitor pembrolizumab because of their proinflammatory state and observed favorable responses in clinical trials.4
Copy number high/low
Copy number (CN) high and low refers to the results of microarrays in which hierarchical clustering was applied to identify reoccurring amplification or deletion regions. The CN-high group was associated with the poorest outcomes (recurrence and survival). There is significant overlap with mutations in TP53. Most serous carcinomas were CN high; however, 25% of patients with high-grade endometrioid cell type shared the CN-high classification. These tumors shared great molecular similarity to high-grade serous ovarian cancers and basal-like breast cancer.
Those patients who did not possess mutations that classified them as POLE hypermutated, MSI high, or CN high were classified as CN low. This group included predominantly grades 1 and 2 endometrioid adenocarcinomas of an early stage and had a favorable prognostic profile, though less favorable than those with a POLE ultramutated state, which appears to be somewhat protective.
Molecular/metabolic interactions
While molecular data are clearly important in driving a cancer cell’s behavior, other clinical and metabolic factors influence cancer behavior. For example, body mass index, adiposity, glucose, and lipid metabolism have been shown to be important drivers of cellular behavior and responsiveness to targeted therapies.5,6 Additionally age, race, and other metabolic states contribute to oncologic behavior. Future classifications of endometrial cancer are unlikely to use molecular profiles in isolation but will need to incorporate these additional patient-specific data to better predict and prognosticate outcomes.
Clinical applications
If researchers can better define and describe a patient’s endometrial cancer from the time of their biopsy, important clinical decisions might be able to be tackled. For example, in a premenopausal patient with an endometrial cancer who is considering fertility-sparing treatments, preoperative knowledge of a POLE ultramutated state (and therefore an anticipated good prognosis) might favor fertility preservation or avoid comprehensive staging which may be of limited value. Similarly, if an MSI-high profile is identified leading to a Lynch syndrome diagnosis, she may be more inclined to undergo a hysterectomy with bilateral salpingo-oophorectomy and staging as she is at known increased risk for a more advanced endometrial cancer, as well as the potential for ovarian cancer.
Postoperative incorporation of molecular data promises to be particularly helpful in guiding adjuvant therapies and sparing some women from unnecessary treatments. For example, women with high-grade endometrioid tumors who are CN high were historically treated with radiotherapy but might do better treated with systemic adjuvant therapies traditionally reserved for nonendometrioid carcinomas. Costly therapies such as immunotherapy can be directed toward those with MSI-high tumors, and the rare patient with a POLE ultramutated state who has a recurrence or advanced disease. Clinical trials will be able to cluster enrollment of patients with CN-high, serouslike cancers with those with serous cancers, rather than combining them with patients whose cancers predictably behave much differently.
Much work is still needed to validate this molecular profiling in endometrial cancer and define the algorithms associated with treatment decisions; however, it is likely that the way we describe endometrial cancer in the near future will be quite different.
Dr. Rossi is an assistant professor in the division of gynecologic oncology at the University of North Carolina at Chapel Hill. She has no disclosures.
References
1. Bokhman JV. Two pathogenetic types of endometrial carcinoma. Gynecol Oncol. 1983;15(1):10-7.
2. Clarke BA et al. Endometrial carcinoma: controversies in histopathological assessment of grade and tumour cell type. J Clin Pathol. 2010;63(5):410-5.
3. Cancer Genome Atlas Research Network. Integrated genomic characterization of endometrial carcinoma. Nature. 2013;497(7447):67-73.
4. Ott PA et al. Pembrolizumab in advanced endometrial cancer: Preliminary results from the phase Ib KEYNOTE-028 study. J Clin Oncol. 2016;34(suppl):Abstract 5581.
5. Roque DR et al. Association between differential gene expression and body mass index among endometrial cancers from the Cancer Genome Atlas Project. Gynecol Oncol. 2016;142(2):317-22.
6. Talhouk A et al. New classification of endometrial cancers: The development and potential applications of genomic-based classification in research and clinical care. Gynecol Oncol Res Pract. 2016 Dec;3:14.
NIH launches HEAL Initiative to combat opioid crisis
The NIH HEAL (Helping to End Addiction Long-term) Initiative aims to bring together agencies across the federal government, as well as academic institutions, private industry, and patient advocates to find new solutions to address the current national health emergency.
“There are 15 initiatives altogether that are being put out that we think are pretty bold and should make a big difference in our understanding of what to do about this national public health crisis,” NIH Director Francis Collins, MD, said in an interview.
HEAL will investigate ways to reformulate existing treatments for opioid use disorder (OUD), to improve efficacy and extend their availability to more patients.
“Although there are effective medications for OUD (methadone, buprenorphine, and naltrexone), only a small percentage of individuals in the United States who would benefit receive these medications,” according to an editorial introducing the NIH HEAL Initiative published in JAMA (doi:10.1001/jama.2018.8826). “Even among those who have initiated these medications, about half will relapse within 6 months.”
The editorial was authored by Dr. Collins, Walter J. Koroshetz, MD, director of the National Institute of Neurological Disorders and Stroke, and Nora Volkow, MD, director of the National Institute on Drug Abuse.
For example, the current formulation of naltrexone lasts about a month within the body, Dr. Collins said in an interview. “If we had a 6-month version of that, I think it would be much more effective because oftentimes the relapses happen after a month or so, before people have fully gotten themselves on the ground.”
Better overdose antidotes are needed as well, he said, particularly for fentanyl overdose. “Narcan may not be strong enough for those long-lasting and very potent opioids like fentanyl,” he said.
HEAL also will seek a better understanding of neonatal opioid withdrawal syndrome (NOWS), also referred to as neonatal abstinence syndrome, which has become alarmingly common as more women of childbearing potential struggle with opioid addiction.
“Innovative methods to identify and treat newborns exposed to opioids, often along with other drugs, have the potential to improve both short- and long-term developmental outcomes in such children,” Dr. Collins and colleagues noted. “To determine better approaches, HEAL will expand Advancing Clinical Trials in Neonatal Opioid Withdrawal Syndrome (ACT NOW). This pilot study is designed to assess the prevalence of NOWS, understand current approaches to managing NOWS , and develop common approaches for larger-scale studies that will determine best practices for clinical care of infants with NOWS throughout the country.”
HEAL efforts also seek to find integrated approaches to OUD treatment.
“One particularly bold element is to put together a number of pilot projects that enable bringing together all of the ways in which we are trying to turn this epidemic around by making it possible to assess whether individuals who are addicted can be successfully treated and maintained in abstinence for long periods of time,” Dr. Collins said. “Right now, the success is not so great.
“Suppose we brought together all of the treatment programs – the primary care facilities, the emergency rooms, the fire departments, the social work experts, the health departments in the states, the local communities, the criminal justice system. We brought together all of those players in a research design where we can really see what was working. Could we do a lot more to turn this around than basically doing one of those at a time? There is this multisite idea of a national research effort, still somewhat in development, but to do integration of all of these efforts. I am pretty excited about that one.”
In looking for better ways to treat pain safely and effectively, “we need to understand how it is that people transition from acute pain to chronic pain … and what can we do increase the likelihood of recovery from acute pain without making that transition,” Dr. Collins said. “Then we need to identify additional novel targets for developing pain therapies, both devices and pharmaceuticals. We need better means of testing those ideas.”
In addition to gaining a better understanding of chronic pain, HEAL aims to investigate new nonaddictive pain treatments and find ways to expedite those treatments through the clinical pipeline, according to Dr. Collins and colleagues.
HEAL “lays the foundation for an innovative therapy-development pipeline through a planned new public-private partnership. In collaboration with biopharmaceutical groups, the Food and Drug Administration, and the Foundation for the NIH, the NIH will collect and evaluate treatment assets from academia and biopharmaceutical and device companies to coordinate and accelerate the development of effective treatments for pain and addiction,” they wrote.
SOURCE: Collins F et al, JAMA doi: 10.1001/jama.2018.8826.
The NIH HEAL (Helping to End Addiction Long-term) Initiative aims to bring together agencies across the federal government, as well as academic institutions, private industry, and patient advocates to find new solutions to address the current national health emergency.
“There are 15 initiatives altogether that are being put out that we think are pretty bold and should make a big difference in our understanding of what to do about this national public health crisis,” NIH Director Francis Collins, MD, said in an interview.
HEAL will investigate ways to reformulate existing treatments for opioid use disorder (OUD), to improve efficacy and extend their availability to more patients.
“Although there are effective medications for OUD (methadone, buprenorphine, and naltrexone), only a small percentage of individuals in the United States who would benefit receive these medications,” according to an editorial introducing the NIH HEAL Initiative published in JAMA (doi:10.1001/jama.2018.8826). “Even among those who have initiated these medications, about half will relapse within 6 months.”
The editorial was authored by Dr. Collins, Walter J. Koroshetz, MD, director of the National Institute of Neurological Disorders and Stroke, and Nora Volkow, MD, director of the National Institute on Drug Abuse.
For example, the current formulation of naltrexone lasts about a month within the body, Dr. Collins said in an interview. “If we had a 6-month version of that, I think it would be much more effective because oftentimes the relapses happen after a month or so, before people have fully gotten themselves on the ground.”
Better overdose antidotes are needed as well, he said, particularly for fentanyl overdose. “Narcan may not be strong enough for those long-lasting and very potent opioids like fentanyl,” he said.
HEAL also will seek a better understanding of neonatal opioid withdrawal syndrome (NOWS), also referred to as neonatal abstinence syndrome, which has become alarmingly common as more women of childbearing potential struggle with opioid addiction.
“Innovative methods to identify and treat newborns exposed to opioids, often along with other drugs, have the potential to improve both short- and long-term developmental outcomes in such children,” Dr. Collins and colleagues noted. “To determine better approaches, HEAL will expand Advancing Clinical Trials in Neonatal Opioid Withdrawal Syndrome (ACT NOW). This pilot study is designed to assess the prevalence of NOWS, understand current approaches to managing NOWS , and develop common approaches for larger-scale studies that will determine best practices for clinical care of infants with NOWS throughout the country.”
HEAL efforts also seek to find integrated approaches to OUD treatment.
“One particularly bold element is to put together a number of pilot projects that enable bringing together all of the ways in which we are trying to turn this epidemic around by making it possible to assess whether individuals who are addicted can be successfully treated and maintained in abstinence for long periods of time,” Dr. Collins said. “Right now, the success is not so great.
“Suppose we brought together all of the treatment programs – the primary care facilities, the emergency rooms, the fire departments, the social work experts, the health departments in the states, the local communities, the criminal justice system. We brought together all of those players in a research design where we can really see what was working. Could we do a lot more to turn this around than basically doing one of those at a time? There is this multisite idea of a national research effort, still somewhat in development, but to do integration of all of these efforts. I am pretty excited about that one.”
In looking for better ways to treat pain safely and effectively, “we need to understand how it is that people transition from acute pain to chronic pain … and what can we do increase the likelihood of recovery from acute pain without making that transition,” Dr. Collins said. “Then we need to identify additional novel targets for developing pain therapies, both devices and pharmaceuticals. We need better means of testing those ideas.”
In addition to gaining a better understanding of chronic pain, HEAL aims to investigate new nonaddictive pain treatments and find ways to expedite those treatments through the clinical pipeline, according to Dr. Collins and colleagues.
HEAL “lays the foundation for an innovative therapy-development pipeline through a planned new public-private partnership. In collaboration with biopharmaceutical groups, the Food and Drug Administration, and the Foundation for the NIH, the NIH will collect and evaluate treatment assets from academia and biopharmaceutical and device companies to coordinate and accelerate the development of effective treatments for pain and addiction,” they wrote.
SOURCE: Collins F et al, JAMA doi: 10.1001/jama.2018.8826.
The NIH HEAL (Helping to End Addiction Long-term) Initiative aims to bring together agencies across the federal government, as well as academic institutions, private industry, and patient advocates to find new solutions to address the current national health emergency.
“There are 15 initiatives altogether that are being put out that we think are pretty bold and should make a big difference in our understanding of what to do about this national public health crisis,” NIH Director Francis Collins, MD, said in an interview.
HEAL will investigate ways to reformulate existing treatments for opioid use disorder (OUD), to improve efficacy and extend their availability to more patients.
“Although there are effective medications for OUD (methadone, buprenorphine, and naltrexone), only a small percentage of individuals in the United States who would benefit receive these medications,” according to an editorial introducing the NIH HEAL Initiative published in JAMA (doi:10.1001/jama.2018.8826). “Even among those who have initiated these medications, about half will relapse within 6 months.”
The editorial was authored by Dr. Collins, Walter J. Koroshetz, MD, director of the National Institute of Neurological Disorders and Stroke, and Nora Volkow, MD, director of the National Institute on Drug Abuse.
For example, the current formulation of naltrexone lasts about a month within the body, Dr. Collins said in an interview. “If we had a 6-month version of that, I think it would be much more effective because oftentimes the relapses happen after a month or so, before people have fully gotten themselves on the ground.”
Better overdose antidotes are needed as well, he said, particularly for fentanyl overdose. “Narcan may not be strong enough for those long-lasting and very potent opioids like fentanyl,” he said.
HEAL also will seek a better understanding of neonatal opioid withdrawal syndrome (NOWS), also referred to as neonatal abstinence syndrome, which has become alarmingly common as more women of childbearing potential struggle with opioid addiction.
“Innovative methods to identify and treat newborns exposed to opioids, often along with other drugs, have the potential to improve both short- and long-term developmental outcomes in such children,” Dr. Collins and colleagues noted. “To determine better approaches, HEAL will expand Advancing Clinical Trials in Neonatal Opioid Withdrawal Syndrome (ACT NOW). This pilot study is designed to assess the prevalence of NOWS, understand current approaches to managing NOWS , and develop common approaches for larger-scale studies that will determine best practices for clinical care of infants with NOWS throughout the country.”
HEAL efforts also seek to find integrated approaches to OUD treatment.
“One particularly bold element is to put together a number of pilot projects that enable bringing together all of the ways in which we are trying to turn this epidemic around by making it possible to assess whether individuals who are addicted can be successfully treated and maintained in abstinence for long periods of time,” Dr. Collins said. “Right now, the success is not so great.
“Suppose we brought together all of the treatment programs – the primary care facilities, the emergency rooms, the fire departments, the social work experts, the health departments in the states, the local communities, the criminal justice system. We brought together all of those players in a research design where we can really see what was working. Could we do a lot more to turn this around than basically doing one of those at a time? There is this multisite idea of a national research effort, still somewhat in development, but to do integration of all of these efforts. I am pretty excited about that one.”
In looking for better ways to treat pain safely and effectively, “we need to understand how it is that people transition from acute pain to chronic pain … and what can we do increase the likelihood of recovery from acute pain without making that transition,” Dr. Collins said. “Then we need to identify additional novel targets for developing pain therapies, both devices and pharmaceuticals. We need better means of testing those ideas.”
In addition to gaining a better understanding of chronic pain, HEAL aims to investigate new nonaddictive pain treatments and find ways to expedite those treatments through the clinical pipeline, according to Dr. Collins and colleagues.
HEAL “lays the foundation for an innovative therapy-development pipeline through a planned new public-private partnership. In collaboration with biopharmaceutical groups, the Food and Drug Administration, and the Foundation for the NIH, the NIH will collect and evaluate treatment assets from academia and biopharmaceutical and device companies to coordinate and accelerate the development of effective treatments for pain and addiction,” they wrote.
SOURCE: Collins F et al, JAMA doi: 10.1001/jama.2018.8826.
FROM JAMA
Bariatric revision mortality linked to age, comorbidities
WASHINGTON – and appears to be rising in recent years, according to two studies presented at the annual Digestive Disease Week®.
Violeta B. Popov, MD, of New York University, and a team of researchers used the Nationwide Inpatient Sample (NIS) to look at mortality risk, costs, and risk factors for complications in revisional bariatric procedures.
In one presentation, Dr. Popov noted that revision after bariatric surgery occurred in approximately 8% of cases for a variety of reasons including lap band adjustment, weight regain, gastric reflux problems, and rarely, because of staple-line leaks. Referring to findings based on the Bariatric Outcomes Longitudinal Database (BOLD), Dr. Popov said that mortality after primary bariatric surgery is estimated at around 0.2% and revisional procedures carry nearly the same low level of mortality risk. BOLD was developed by the American Society of Metabolic and Bariatric Surgery and reflects outcomes from certified Bariatric Centers of Excellence from 2007 to 2012. However, Dr. Popov noted, the outcomes derived from BOLD may well be better than those from noncertified centers (Gastrointest Surg. 2015 Jan;19[1]:171-8).
Dr. Popov reported that the number of revisional procedures has doubled over recent years, from 6% of all bariatric procedures in 2011 to 13% in 2015. The reasons behind the increase could be related to the number of patients switching to a different bariatric approach, the removal of lap bands, and possibly the increase in the number of primary bariatric surgeries performed by less-skilled operators, Dr. Popov said.
The investigators aimed to determine the mortality trends for these procedures in addition to evaluating costs and risk factors for complications. They conducted a retrospective cohort study using the 2014 NIS, comprising 14,280 patients who underwent revisional bariatric surgery. The primary outcome was postoperative in-hospital mortality, with secondary outcomes of cost, length of hospital stay (LOS), and ICU stay. The variables included a variety of comorbidities, alcohol use, smoking, income, and insurance status.
The mean age of this sample was 68 years and 58.8% were female. Outcomes for revisional bariatric surgery were worse in several categories than were found in the BOLD study in terms of LOS, costs, and mortality, and postoperative in-hospital mortality was unexpectedly high at 2.1% (290 patients). A total of 3.3% of the patients had an ICU stay, one-quarter of whom died.
On univariate analysis, comorbidities (age, coagulopathy, chronic kidney disease, anemia, and chronic heart failure) and the combined number of chronic conditions were all significant predictors of mortality. Multivariate analysis identified age (odds ratio, 1.08; 95% confidence interval, 1.04-1.20; P less than .001), alcohol use (OR, 4.0; 95% CI, 1.3-11.7; P = .01), coagulopathy (OR, 5.4; 95% CI, 2.2-13.3; P less than .001), and insurance status (Medicaid vs. private; OR, 4.0; 95% CI, 1.7-9.9; P = .002) as the most significant predictors of mortality after a revisional bariatric procedure.
In a poster, Dr. Popov and her colleagues presented data from the NIS database looking at 10-year mortality and outcome trends for revisional surgery versus primary Roux-en-Y gastric bypass (RYGB) surgery. Inpatient mortality for RYGB decreased from 2.54% in 2003 to 1.80% in 2014, but was still substantially higher than the BOLD findings. But mortality for revisional surgery increased: 1.90% versus 2.03%. LOS for RYGB decreased from 5.9 days to 5.4 but increased for revisional surgery from 4.6 to 5.4 days. Cost for both procedures, adjusted for inflation, more than doubled between 2003 and 2014. And patients requiring ICU admission for both procedures went from 1% in 2003 to 3% in 2014.
The limitations of both analyses are their retrospective design, the NIS bias inferred by the inclusion of only inpatient procedures, and the lack of laboratory data or data on body mass index. In addition, during the study period, primary bariatric surgery began to be performed as an outpatient procedure. “Low-risk procedures performed in outpatient facilities will not be captured in the database and thus the higher mortality for these higher risk patients is expected,” Dr. Popov said. These patients are likely to be sicker and have more comorbidities. Revisional procedures are typically done in the hospital, but there are some low-risk revisional procedures such as lap band removal that could be done as outpatient procedures. Dr. Popov had confidence that the NIS database reflects real-world outcomes for revisional bariatric procedures.
She concluded that the explanation for the increase in mortality risk for revisional bariatric surgery may be because of more of these procedures being done outside centers of excellence and more, older patients with comorbidities having the surgery, and that nonsurgical alternatives should be explored for the older sicker patients.
Dr. Popova disclosed ownership of shares in Embarcadero Technologies but no conflicts of interest.
SOURCE: Popov VB et al. DDW 2018, Abstract 324.
WASHINGTON – and appears to be rising in recent years, according to two studies presented at the annual Digestive Disease Week®.
Violeta B. Popov, MD, of New York University, and a team of researchers used the Nationwide Inpatient Sample (NIS) to look at mortality risk, costs, and risk factors for complications in revisional bariatric procedures.
In one presentation, Dr. Popov noted that revision after bariatric surgery occurred in approximately 8% of cases for a variety of reasons including lap band adjustment, weight regain, gastric reflux problems, and rarely, because of staple-line leaks. Referring to findings based on the Bariatric Outcomes Longitudinal Database (BOLD), Dr. Popov said that mortality after primary bariatric surgery is estimated at around 0.2% and revisional procedures carry nearly the same low level of mortality risk. BOLD was developed by the American Society of Metabolic and Bariatric Surgery and reflects outcomes from certified Bariatric Centers of Excellence from 2007 to 2012. However, Dr. Popov noted, the outcomes derived from BOLD may well be better than those from noncertified centers (Gastrointest Surg. 2015 Jan;19[1]:171-8).
Dr. Popov reported that the number of revisional procedures has doubled over recent years, from 6% of all bariatric procedures in 2011 to 13% in 2015. The reasons behind the increase could be related to the number of patients switching to a different bariatric approach, the removal of lap bands, and possibly the increase in the number of primary bariatric surgeries performed by less-skilled operators, Dr. Popov said.
The investigators aimed to determine the mortality trends for these procedures in addition to evaluating costs and risk factors for complications. They conducted a retrospective cohort study using the 2014 NIS, comprising 14,280 patients who underwent revisional bariatric surgery. The primary outcome was postoperative in-hospital mortality, with secondary outcomes of cost, length of hospital stay (LOS), and ICU stay. The variables included a variety of comorbidities, alcohol use, smoking, income, and insurance status.
The mean age of this sample was 68 years and 58.8% were female. Outcomes for revisional bariatric surgery were worse in several categories than were found in the BOLD study in terms of LOS, costs, and mortality, and postoperative in-hospital mortality was unexpectedly high at 2.1% (290 patients). A total of 3.3% of the patients had an ICU stay, one-quarter of whom died.
On univariate analysis, comorbidities (age, coagulopathy, chronic kidney disease, anemia, and chronic heart failure) and the combined number of chronic conditions were all significant predictors of mortality. Multivariate analysis identified age (odds ratio, 1.08; 95% confidence interval, 1.04-1.20; P less than .001), alcohol use (OR, 4.0; 95% CI, 1.3-11.7; P = .01), coagulopathy (OR, 5.4; 95% CI, 2.2-13.3; P less than .001), and insurance status (Medicaid vs. private; OR, 4.0; 95% CI, 1.7-9.9; P = .002) as the most significant predictors of mortality after a revisional bariatric procedure.
In a poster, Dr. Popov and her colleagues presented data from the NIS database looking at 10-year mortality and outcome trends for revisional surgery versus primary Roux-en-Y gastric bypass (RYGB) surgery. Inpatient mortality for RYGB decreased from 2.54% in 2003 to 1.80% in 2014, but was still substantially higher than the BOLD findings. But mortality for revisional surgery increased: 1.90% versus 2.03%. LOS for RYGB decreased from 5.9 days to 5.4 but increased for revisional surgery from 4.6 to 5.4 days. Cost for both procedures, adjusted for inflation, more than doubled between 2003 and 2014. And patients requiring ICU admission for both procedures went from 1% in 2003 to 3% in 2014.
The limitations of both analyses are their retrospective design, the NIS bias inferred by the inclusion of only inpatient procedures, and the lack of laboratory data or data on body mass index. In addition, during the study period, primary bariatric surgery began to be performed as an outpatient procedure. “Low-risk procedures performed in outpatient facilities will not be captured in the database and thus the higher mortality for these higher risk patients is expected,” Dr. Popov said. These patients are likely to be sicker and have more comorbidities. Revisional procedures are typically done in the hospital, but there are some low-risk revisional procedures such as lap band removal that could be done as outpatient procedures. Dr. Popov had confidence that the NIS database reflects real-world outcomes for revisional bariatric procedures.
She concluded that the explanation for the increase in mortality risk for revisional bariatric surgery may be because of more of these procedures being done outside centers of excellence and more, older patients with comorbidities having the surgery, and that nonsurgical alternatives should be explored for the older sicker patients.
Dr. Popova disclosed ownership of shares in Embarcadero Technologies but no conflicts of interest.
SOURCE: Popov VB et al. DDW 2018, Abstract 324.
WASHINGTON – and appears to be rising in recent years, according to two studies presented at the annual Digestive Disease Week®.
Violeta B. Popov, MD, of New York University, and a team of researchers used the Nationwide Inpatient Sample (NIS) to look at mortality risk, costs, and risk factors for complications in revisional bariatric procedures.
In one presentation, Dr. Popov noted that revision after bariatric surgery occurred in approximately 8% of cases for a variety of reasons including lap band adjustment, weight regain, gastric reflux problems, and rarely, because of staple-line leaks. Referring to findings based on the Bariatric Outcomes Longitudinal Database (BOLD), Dr. Popov said that mortality after primary bariatric surgery is estimated at around 0.2% and revisional procedures carry nearly the same low level of mortality risk. BOLD was developed by the American Society of Metabolic and Bariatric Surgery and reflects outcomes from certified Bariatric Centers of Excellence from 2007 to 2012. However, Dr. Popov noted, the outcomes derived from BOLD may well be better than those from noncertified centers (Gastrointest Surg. 2015 Jan;19[1]:171-8).
Dr. Popov reported that the number of revisional procedures has doubled over recent years, from 6% of all bariatric procedures in 2011 to 13% in 2015. The reasons behind the increase could be related to the number of patients switching to a different bariatric approach, the removal of lap bands, and possibly the increase in the number of primary bariatric surgeries performed by less-skilled operators, Dr. Popov said.
The investigators aimed to determine the mortality trends for these procedures in addition to evaluating costs and risk factors for complications. They conducted a retrospective cohort study using the 2014 NIS, comprising 14,280 patients who underwent revisional bariatric surgery. The primary outcome was postoperative in-hospital mortality, with secondary outcomes of cost, length of hospital stay (LOS), and ICU stay. The variables included a variety of comorbidities, alcohol use, smoking, income, and insurance status.
The mean age of this sample was 68 years and 58.8% were female. Outcomes for revisional bariatric surgery were worse in several categories than were found in the BOLD study in terms of LOS, costs, and mortality, and postoperative in-hospital mortality was unexpectedly high at 2.1% (290 patients). A total of 3.3% of the patients had an ICU stay, one-quarter of whom died.
On univariate analysis, comorbidities (age, coagulopathy, chronic kidney disease, anemia, and chronic heart failure) and the combined number of chronic conditions were all significant predictors of mortality. Multivariate analysis identified age (odds ratio, 1.08; 95% confidence interval, 1.04-1.20; P less than .001), alcohol use (OR, 4.0; 95% CI, 1.3-11.7; P = .01), coagulopathy (OR, 5.4; 95% CI, 2.2-13.3; P less than .001), and insurance status (Medicaid vs. private; OR, 4.0; 95% CI, 1.7-9.9; P = .002) as the most significant predictors of mortality after a revisional bariatric procedure.
In a poster, Dr. Popov and her colleagues presented data from the NIS database looking at 10-year mortality and outcome trends for revisional surgery versus primary Roux-en-Y gastric bypass (RYGB) surgery. Inpatient mortality for RYGB decreased from 2.54% in 2003 to 1.80% in 2014, but was still substantially higher than the BOLD findings. But mortality for revisional surgery increased: 1.90% versus 2.03%. LOS for RYGB decreased from 5.9 days to 5.4 but increased for revisional surgery from 4.6 to 5.4 days. Cost for both procedures, adjusted for inflation, more than doubled between 2003 and 2014. And patients requiring ICU admission for both procedures went from 1% in 2003 to 3% in 2014.
The limitations of both analyses are their retrospective design, the NIS bias inferred by the inclusion of only inpatient procedures, and the lack of laboratory data or data on body mass index. In addition, during the study period, primary bariatric surgery began to be performed as an outpatient procedure. “Low-risk procedures performed in outpatient facilities will not be captured in the database and thus the higher mortality for these higher risk patients is expected,” Dr. Popov said. These patients are likely to be sicker and have more comorbidities. Revisional procedures are typically done in the hospital, but there are some low-risk revisional procedures such as lap band removal that could be done as outpatient procedures. Dr. Popov had confidence that the NIS database reflects real-world outcomes for revisional bariatric procedures.
She concluded that the explanation for the increase in mortality risk for revisional bariatric surgery may be because of more of these procedures being done outside centers of excellence and more, older patients with comorbidities having the surgery, and that nonsurgical alternatives should be explored for the older sicker patients.
Dr. Popova disclosed ownership of shares in Embarcadero Technologies but no conflicts of interest.
SOURCE: Popov VB et al. DDW 2018, Abstract 324.
REPORTING FROM DDW 2018
Key clinical point: Revisional bariatric procedures may carry a greater mortality risk than previous studies have suggested.
Major finding: The mortality rate in the sample was 2.1%.
Study details: The 2014 Nationwide Inpatient Sample database, comprising 14,280 patients who underwent revisional bariatric surgery.
Disclosures: Dr. Popova disclosed ownership of shares in Embarcadero Technologies but no conflicts of interest.
Source: Popov VB et al. DDW 2018, Abstract 324.
Malnourished U.S. inpatients often go untreated
WASHINGTON – Hospital staffs often fail to treat .
A retrospective review of more than 150,000 patients admitted during a single year at any center within a large, multicenter U.S. hospital system found that even when patients receive oral nutritional supplementation, there is often a substantial delay to its onset.
The data also suggested potential benefits from treating malnutrition with oral nutritional supplementation (ONS). Patients who received ONS had a 10% relative reduction in their rate of 30-day readmission, compared with malnourished patients who did not receive supplements after adjusting for several baseline demographic and clinical variables, Gerard Mullin, MD, said at the annual Digestive Disease Week. His analysis also showed that every doubling of the time from hospital admission to an order for ONS significantly linked with a 6% rise in hospital length of stay.
The findings “highlight the importance of malnutrition screening on admission, starting a nutrition intervention as soon as malnutrition is confirmed, and treating with appropriate ONS,” said Dr. Mullin, a gastroenterologist at Johns Hopkins Medicine in Baltimore and director of the Celiac Disease Clinic. A standard formulation of Ensure was the ONS routinely used at the Johns Hopkins hospitals
“We’re missing malnutrition,” Dr. Mullin said in an interview. The hospital accreditation standards of the Joint Commission call for assessment of the nutritional status of hospitalized patients within 24 hours of admission (Jt Comm J Qual Patient Saf. 2015 Oct;41[10]:469-73). Screening is “not uniformly done,” and when malnutrition is identified, the finding must usually pass through several layers of a hospital’s medico-bureaucratic process before treatment actually starts, he noted. Plus, there’s often dismissal of the importance of intervention. “It’s important to treat patients with ONS sooner than later,” he said.
Dr. Mullin and his associates studied hospital records for 153,161 people admitted to any of the Baltimore-area hospitals in the Johns Hopkins system during October 2016 through the end of September 2017. The hospital staff routinely assessed nutritional status of patients after admission with a two-question screen based on the Malnutrition Screening Tool (Nutrition. 1999 June;15[6]:458-64): Have you had unplanned weight loss of 10 pounds or more during the past 6 months? Have you had decreased oral intake over the past 5 days? This identified 30,284 (20%) who qualified as possibly malnourished by either criterion. The researchers also retrospectively applied a more detailed screen to the patient records using the criteria set by an international consensus guideline committee in 2010 (J Parenter Enteraal Nutr. 2010 Mar-Apr;34[2]:156-9). This identified 8,713 of the hospitalized patients (6%) as malnourished soon after admission. Despite these numbers a scant 274 patients among these 8,713 (3%) actually received ONS, Dr. Mullin reported. In addition, it took an average of 85 hours from the time of each malnourished patient’s admission to when the ONS order appeared in their record.
Dr. Mullin conceded that both the association his group found between treatment with ONS and a reduced rate of 30-day readmission to any of the hospitals in the Johns Hopkins system, and the association between a delay in the time to the start of ONS and length of stay may have been confounded by factors not accounted for in the adjustments they applied. But he maintained that the links are consistent with results from prior studies, and warrant running prospective, randomized studies to better document the impact of ONS on newly admitted patients identified as malnourished.
“We need more of these types of studies and interventional trials to show that ONS makes a difference,” Dr. Mullin said.
The study was sponsored by Abbott, which markets the oral nutritional supplement Ensure. Dr. Mullin had no additional disclosures.
[email protected]
On Twitter @mitchelzoler
SOURCE: Source: Mullin G et al. DDW 2018 presentation 883.
WASHINGTON – Hospital staffs often fail to treat .
A retrospective review of more than 150,000 patients admitted during a single year at any center within a large, multicenter U.S. hospital system found that even when patients receive oral nutritional supplementation, there is often a substantial delay to its onset.
The data also suggested potential benefits from treating malnutrition with oral nutritional supplementation (ONS). Patients who received ONS had a 10% relative reduction in their rate of 30-day readmission, compared with malnourished patients who did not receive supplements after adjusting for several baseline demographic and clinical variables, Gerard Mullin, MD, said at the annual Digestive Disease Week. His analysis also showed that every doubling of the time from hospital admission to an order for ONS significantly linked with a 6% rise in hospital length of stay.
The findings “highlight the importance of malnutrition screening on admission, starting a nutrition intervention as soon as malnutrition is confirmed, and treating with appropriate ONS,” said Dr. Mullin, a gastroenterologist at Johns Hopkins Medicine in Baltimore and director of the Celiac Disease Clinic. A standard formulation of Ensure was the ONS routinely used at the Johns Hopkins hospitals
“We’re missing malnutrition,” Dr. Mullin said in an interview. The hospital accreditation standards of the Joint Commission call for assessment of the nutritional status of hospitalized patients within 24 hours of admission (Jt Comm J Qual Patient Saf. 2015 Oct;41[10]:469-73). Screening is “not uniformly done,” and when malnutrition is identified, the finding must usually pass through several layers of a hospital’s medico-bureaucratic process before treatment actually starts, he noted. Plus, there’s often dismissal of the importance of intervention. “It’s important to treat patients with ONS sooner than later,” he said.
Dr. Mullin and his associates studied hospital records for 153,161 people admitted to any of the Baltimore-area hospitals in the Johns Hopkins system during October 2016 through the end of September 2017. The hospital staff routinely assessed nutritional status of patients after admission with a two-question screen based on the Malnutrition Screening Tool (Nutrition. 1999 June;15[6]:458-64): Have you had unplanned weight loss of 10 pounds or more during the past 6 months? Have you had decreased oral intake over the past 5 days? This identified 30,284 (20%) who qualified as possibly malnourished by either criterion. The researchers also retrospectively applied a more detailed screen to the patient records using the criteria set by an international consensus guideline committee in 2010 (J Parenter Enteraal Nutr. 2010 Mar-Apr;34[2]:156-9). This identified 8,713 of the hospitalized patients (6%) as malnourished soon after admission. Despite these numbers a scant 274 patients among these 8,713 (3%) actually received ONS, Dr. Mullin reported. In addition, it took an average of 85 hours from the time of each malnourished patient’s admission to when the ONS order appeared in their record.
Dr. Mullin conceded that both the association his group found between treatment with ONS and a reduced rate of 30-day readmission to any of the hospitals in the Johns Hopkins system, and the association between a delay in the time to the start of ONS and length of stay may have been confounded by factors not accounted for in the adjustments they applied. But he maintained that the links are consistent with results from prior studies, and warrant running prospective, randomized studies to better document the impact of ONS on newly admitted patients identified as malnourished.
“We need more of these types of studies and interventional trials to show that ONS makes a difference,” Dr. Mullin said.
The study was sponsored by Abbott, which markets the oral nutritional supplement Ensure. Dr. Mullin had no additional disclosures.
[email protected]
On Twitter @mitchelzoler
SOURCE: Source: Mullin G et al. DDW 2018 presentation 883.
WASHINGTON – Hospital staffs often fail to treat .
A retrospective review of more than 150,000 patients admitted during a single year at any center within a large, multicenter U.S. hospital system found that even when patients receive oral nutritional supplementation, there is often a substantial delay to its onset.
The data also suggested potential benefits from treating malnutrition with oral nutritional supplementation (ONS). Patients who received ONS had a 10% relative reduction in their rate of 30-day readmission, compared with malnourished patients who did not receive supplements after adjusting for several baseline demographic and clinical variables, Gerard Mullin, MD, said at the annual Digestive Disease Week. His analysis also showed that every doubling of the time from hospital admission to an order for ONS significantly linked with a 6% rise in hospital length of stay.
The findings “highlight the importance of malnutrition screening on admission, starting a nutrition intervention as soon as malnutrition is confirmed, and treating with appropriate ONS,” said Dr. Mullin, a gastroenterologist at Johns Hopkins Medicine in Baltimore and director of the Celiac Disease Clinic. A standard formulation of Ensure was the ONS routinely used at the Johns Hopkins hospitals
“We’re missing malnutrition,” Dr. Mullin said in an interview. The hospital accreditation standards of the Joint Commission call for assessment of the nutritional status of hospitalized patients within 24 hours of admission (Jt Comm J Qual Patient Saf. 2015 Oct;41[10]:469-73). Screening is “not uniformly done,” and when malnutrition is identified, the finding must usually pass through several layers of a hospital’s medico-bureaucratic process before treatment actually starts, he noted. Plus, there’s often dismissal of the importance of intervention. “It’s important to treat patients with ONS sooner than later,” he said.
Dr. Mullin and his associates studied hospital records for 153,161 people admitted to any of the Baltimore-area hospitals in the Johns Hopkins system during October 2016 through the end of September 2017. The hospital staff routinely assessed nutritional status of patients after admission with a two-question screen based on the Malnutrition Screening Tool (Nutrition. 1999 June;15[6]:458-64): Have you had unplanned weight loss of 10 pounds or more during the past 6 months? Have you had decreased oral intake over the past 5 days? This identified 30,284 (20%) who qualified as possibly malnourished by either criterion. The researchers also retrospectively applied a more detailed screen to the patient records using the criteria set by an international consensus guideline committee in 2010 (J Parenter Enteraal Nutr. 2010 Mar-Apr;34[2]:156-9). This identified 8,713 of the hospitalized patients (6%) as malnourished soon after admission. Despite these numbers a scant 274 patients among these 8,713 (3%) actually received ONS, Dr. Mullin reported. In addition, it took an average of 85 hours from the time of each malnourished patient’s admission to when the ONS order appeared in their record.
Dr. Mullin conceded that both the association his group found between treatment with ONS and a reduced rate of 30-day readmission to any of the hospitals in the Johns Hopkins system, and the association between a delay in the time to the start of ONS and length of stay may have been confounded by factors not accounted for in the adjustments they applied. But he maintained that the links are consistent with results from prior studies, and warrant running prospective, randomized studies to better document the impact of ONS on newly admitted patients identified as malnourished.
“We need more of these types of studies and interventional trials to show that ONS makes a difference,” Dr. Mullin said.
The study was sponsored by Abbott, which markets the oral nutritional supplement Ensure. Dr. Mullin had no additional disclosures.
[email protected]
On Twitter @mitchelzoler
SOURCE: Source: Mullin G et al. DDW 2018 presentation 883.
REPORTING FROM DDW 2018
Key clinical point: Malnourished U.S. hospital inpatients often go untreated.
Major finding: Three percent of patients retrospectively identified as malnourished soon after hospital admission received oral nutritional supplementation.
Study details: Retrospective review of 153,161 patients admitted to a large U.S. hospital network during 2016-2017.
Disclosures: The study was sponsored by Abbott, which markets the oral nutritional supplement Ensure. Dr. Mullin had no additional disclosures.
Source: Mullin G et al. Digestive Disease Week presentation 883.
Beyond the sleeve and RYGB: The frontier of bariatric procedures
BOSTON – Though are the bariatric procedures most patients will receive, other surgical approaches to weight loss are occasionally performed. Knowing these various techniques and their likely efficacy and safety can help physicians who care for patients with obesity, whether a patient is considering a less common option, or whether a post-vagal blockade patient shows up on the schedule with long-term issues.
A common theme among many of these procedures is that overall numbers are low, long-term follow-up may be lacking, and research quality is variable, said Travis McKenzie, MD, speaking at a bariatric surgery-focused session of the annual clinical and scientific meeting of the American Association of Clinical Endocrinologists.
One minimally invasive approach targets stomach functions and the appetite and satiety signaling system. In vagal blockade via an electronic implant (vBloc), an indwelling, removable device produces electronically-induced intermittent blockade of the vagal nerve.
In one randomized controlled trial, excess weight loss in patients receiving this procedure was 24%, significantly more than the 16% seen in the group that received a sham procedure (P = .002); both groups received regular follow-up and counseling, according to the study protocol. Overall, at 1 year, 52% of those in the treatment group had seen at least 20% reduction in excess weight; just 3.7% of vBloc recipients had adverse events, mostly some dyspepsia and pain at the implant site, said Dr. McKenzie, an endocrine surgeon at the Mayo Clinic, Rochester, Minn.
The vBloc procedure, said Dr. McKenzie, “demonstrated modest weight loss at 2 years, with a reasonable risk profile.”
A variation of the duodenal switch is known as single anastomosis duodeno-ileal bypass with sleeve gastrectomy, or SADI-S. This procedure both resects the greater curve of the stomach to create a gastric sleeve, and uses a single intestinal anastomosis to create a common channel of 200, 250 or 300 cm, bypassing most of the small intestine.
In this procedure, also known as the one-anastomosis duodenal switch (OADS), weight loss occurs both because of intestinal malabsorption and because of the reduced stomach volume.
Parsing safety and efficacy of this procedure isn’t easy, given the studies at hand, said Dr. McKenzie: “The data are plagued by short follow-up, low numbers, and inconsistent quality.” Of the 14 case series following 1,045 patients, none include randomized controlled data, he said.
The data that are available show total body weight loss in the 34%-39% range, with little difference between losses seen at 1 year and 2 years.
However, said Dr. McKenzie, one 100-patient case series showed that SADI-S patients averaged 2.5 bowel movements daily after the procedure, and two patients needed surgical revision because they were experiencing malnutrition. Anemia, vitamin B12 and D deficiencies, and folate deficiency are all commonly seen two years after SADI-S procedures, he said.
“The OADS procedure is very effective, although better data are needed before drawing conclusions,” said Dr. McKenzie.
A gastric bypass variation known as the “mini” bypass, or the one anastomosis gastric bypass (OAGB), is another less common bariatric technique. In this procedure, a small gastric pouch is created that forms the working stomach, which is then connected to the duodenum with bypassing of a significant portion (up to 200 cm) of the small intestine. This procedure causes both restrictive and malabsorptive weight loss, and is usually performed using minimally invasive surgery.
Four randomized controlled trials exist, said Dr. McKenzie, that compare OAGB variously to Roux-en-Y gastric bypass (RYGB) and to sleeve gastrectomy. In an 80-patient study that compared OAGB with RYGB at two years post-procedure, excess weight loss was similar, at 60% for OAGB versus 64% for RYGB ( Ann Surg. 2005;24[1]20-8). However, morbidity was less for OAGB recipients (8% vs 20%, P less than .05).
Another study looked at OAGB and sleeve gastrectomy in 60 patients, following them for 5 years. Total body weight loss was similar between groups at 20%-23%, said Dr. McKenzie (Obes Surg. 2014;24[9]1552-62).
“But what about bile reflux?” asked Dr. McKenzie. He pointed out that in OAGB, digestive juices enter the digestive path very close to the outlet of the new, surgically created stomach, affording the potential for significant reflux. Calling for further study of the frequency of bile reflux and potential long-term sequelae, he advised caution with this otherwise attractive procedure.
Those caring for bariatric patients may also see the consequences of “rogue” procedures on occasion: “Interest in metabolic surgery has led to some ‘original’ procedures, many of which are not based on firm science,” said Dr. McKenzie. An exemplar of an understudied procedure is the sleeve gastrectomy with a loop bipartition, with results that have been published in case reports, but whose longer-term outcomes are unknown.
“Caution is advised regarding operations that are devised outside of study protocols,” said Dr. McKenzie.
Dr. McKenzie reported that he had no relevant financial disclosures.
SOURCE: McKenzie, T. AACE 2018, presentation SGS4.
BOSTON – Though are the bariatric procedures most patients will receive, other surgical approaches to weight loss are occasionally performed. Knowing these various techniques and their likely efficacy and safety can help physicians who care for patients with obesity, whether a patient is considering a less common option, or whether a post-vagal blockade patient shows up on the schedule with long-term issues.
A common theme among many of these procedures is that overall numbers are low, long-term follow-up may be lacking, and research quality is variable, said Travis McKenzie, MD, speaking at a bariatric surgery-focused session of the annual clinical and scientific meeting of the American Association of Clinical Endocrinologists.
One minimally invasive approach targets stomach functions and the appetite and satiety signaling system. In vagal blockade via an electronic implant (vBloc), an indwelling, removable device produces electronically-induced intermittent blockade of the vagal nerve.
In one randomized controlled trial, excess weight loss in patients receiving this procedure was 24%, significantly more than the 16% seen in the group that received a sham procedure (P = .002); both groups received regular follow-up and counseling, according to the study protocol. Overall, at 1 year, 52% of those in the treatment group had seen at least 20% reduction in excess weight; just 3.7% of vBloc recipients had adverse events, mostly some dyspepsia and pain at the implant site, said Dr. McKenzie, an endocrine surgeon at the Mayo Clinic, Rochester, Minn.
The vBloc procedure, said Dr. McKenzie, “demonstrated modest weight loss at 2 years, with a reasonable risk profile.”
A variation of the duodenal switch is known as single anastomosis duodeno-ileal bypass with sleeve gastrectomy, or SADI-S. This procedure both resects the greater curve of the stomach to create a gastric sleeve, and uses a single intestinal anastomosis to create a common channel of 200, 250 or 300 cm, bypassing most of the small intestine.
In this procedure, also known as the one-anastomosis duodenal switch (OADS), weight loss occurs both because of intestinal malabsorption and because of the reduced stomach volume.
Parsing safety and efficacy of this procedure isn’t easy, given the studies at hand, said Dr. McKenzie: “The data are plagued by short follow-up, low numbers, and inconsistent quality.” Of the 14 case series following 1,045 patients, none include randomized controlled data, he said.
The data that are available show total body weight loss in the 34%-39% range, with little difference between losses seen at 1 year and 2 years.
However, said Dr. McKenzie, one 100-patient case series showed that SADI-S patients averaged 2.5 bowel movements daily after the procedure, and two patients needed surgical revision because they were experiencing malnutrition. Anemia, vitamin B12 and D deficiencies, and folate deficiency are all commonly seen two years after SADI-S procedures, he said.
“The OADS procedure is very effective, although better data are needed before drawing conclusions,” said Dr. McKenzie.
A gastric bypass variation known as the “mini” bypass, or the one anastomosis gastric bypass (OAGB), is another less common bariatric technique. In this procedure, a small gastric pouch is created that forms the working stomach, which is then connected to the duodenum with bypassing of a significant portion (up to 200 cm) of the small intestine. This procedure causes both restrictive and malabsorptive weight loss, and is usually performed using minimally invasive surgery.
Four randomized controlled trials exist, said Dr. McKenzie, that compare OAGB variously to Roux-en-Y gastric bypass (RYGB) and to sleeve gastrectomy. In an 80-patient study that compared OAGB with RYGB at two years post-procedure, excess weight loss was similar, at 60% for OAGB versus 64% for RYGB ( Ann Surg. 2005;24[1]20-8). However, morbidity was less for OAGB recipients (8% vs 20%, P less than .05).
Another study looked at OAGB and sleeve gastrectomy in 60 patients, following them for 5 years. Total body weight loss was similar between groups at 20%-23%, said Dr. McKenzie (Obes Surg. 2014;24[9]1552-62).
“But what about bile reflux?” asked Dr. McKenzie. He pointed out that in OAGB, digestive juices enter the digestive path very close to the outlet of the new, surgically created stomach, affording the potential for significant reflux. Calling for further study of the frequency of bile reflux and potential long-term sequelae, he advised caution with this otherwise attractive procedure.
Those caring for bariatric patients may also see the consequences of “rogue” procedures on occasion: “Interest in metabolic surgery has led to some ‘original’ procedures, many of which are not based on firm science,” said Dr. McKenzie. An exemplar of an understudied procedure is the sleeve gastrectomy with a loop bipartition, with results that have been published in case reports, but whose longer-term outcomes are unknown.
“Caution is advised regarding operations that are devised outside of study protocols,” said Dr. McKenzie.
Dr. McKenzie reported that he had no relevant financial disclosures.
SOURCE: McKenzie, T. AACE 2018, presentation SGS4.
BOSTON – Though are the bariatric procedures most patients will receive, other surgical approaches to weight loss are occasionally performed. Knowing these various techniques and their likely efficacy and safety can help physicians who care for patients with obesity, whether a patient is considering a less common option, or whether a post-vagal blockade patient shows up on the schedule with long-term issues.
A common theme among many of these procedures is that overall numbers are low, long-term follow-up may be lacking, and research quality is variable, said Travis McKenzie, MD, speaking at a bariatric surgery-focused session of the annual clinical and scientific meeting of the American Association of Clinical Endocrinologists.
One minimally invasive approach targets stomach functions and the appetite and satiety signaling system. In vagal blockade via an electronic implant (vBloc), an indwelling, removable device produces electronically-induced intermittent blockade of the vagal nerve.
In one randomized controlled trial, excess weight loss in patients receiving this procedure was 24%, significantly more than the 16% seen in the group that received a sham procedure (P = .002); both groups received regular follow-up and counseling, according to the study protocol. Overall, at 1 year, 52% of those in the treatment group had seen at least 20% reduction in excess weight; just 3.7% of vBloc recipients had adverse events, mostly some dyspepsia and pain at the implant site, said Dr. McKenzie, an endocrine surgeon at the Mayo Clinic, Rochester, Minn.
The vBloc procedure, said Dr. McKenzie, “demonstrated modest weight loss at 2 years, with a reasonable risk profile.”
A variation of the duodenal switch is known as single anastomosis duodeno-ileal bypass with sleeve gastrectomy, or SADI-S. This procedure both resects the greater curve of the stomach to create a gastric sleeve, and uses a single intestinal anastomosis to create a common channel of 200, 250 or 300 cm, bypassing most of the small intestine.
In this procedure, also known as the one-anastomosis duodenal switch (OADS), weight loss occurs both because of intestinal malabsorption and because of the reduced stomach volume.
Parsing safety and efficacy of this procedure isn’t easy, given the studies at hand, said Dr. McKenzie: “The data are plagued by short follow-up, low numbers, and inconsistent quality.” Of the 14 case series following 1,045 patients, none include randomized controlled data, he said.
The data that are available show total body weight loss in the 34%-39% range, with little difference between losses seen at 1 year and 2 years.
However, said Dr. McKenzie, one 100-patient case series showed that SADI-S patients averaged 2.5 bowel movements daily after the procedure, and two patients needed surgical revision because they were experiencing malnutrition. Anemia, vitamin B12 and D deficiencies, and folate deficiency are all commonly seen two years after SADI-S procedures, he said.
“The OADS procedure is very effective, although better data are needed before drawing conclusions,” said Dr. McKenzie.
A gastric bypass variation known as the “mini” bypass, or the one anastomosis gastric bypass (OAGB), is another less common bariatric technique. In this procedure, a small gastric pouch is created that forms the working stomach, which is then connected to the duodenum with bypassing of a significant portion (up to 200 cm) of the small intestine. This procedure causes both restrictive and malabsorptive weight loss, and is usually performed using minimally invasive surgery.
Four randomized controlled trials exist, said Dr. McKenzie, that compare OAGB variously to Roux-en-Y gastric bypass (RYGB) and to sleeve gastrectomy. In an 80-patient study that compared OAGB with RYGB at two years post-procedure, excess weight loss was similar, at 60% for OAGB versus 64% for RYGB ( Ann Surg. 2005;24[1]20-8). However, morbidity was less for OAGB recipients (8% vs 20%, P less than .05).
Another study looked at OAGB and sleeve gastrectomy in 60 patients, following them for 5 years. Total body weight loss was similar between groups at 20%-23%, said Dr. McKenzie (Obes Surg. 2014;24[9]1552-62).
“But what about bile reflux?” asked Dr. McKenzie. He pointed out that in OAGB, digestive juices enter the digestive path very close to the outlet of the new, surgically created stomach, affording the potential for significant reflux. Calling for further study of the frequency of bile reflux and potential long-term sequelae, he advised caution with this otherwise attractive procedure.
Those caring for bariatric patients may also see the consequences of “rogue” procedures on occasion: “Interest in metabolic surgery has led to some ‘original’ procedures, many of which are not based on firm science,” said Dr. McKenzie. An exemplar of an understudied procedure is the sleeve gastrectomy with a loop bipartition, with results that have been published in case reports, but whose longer-term outcomes are unknown.
“Caution is advised regarding operations that are devised outside of study protocols,” said Dr. McKenzie.
Dr. McKenzie reported that he had no relevant financial disclosures.
SOURCE: McKenzie, T. AACE 2018, presentation SGS4.
EXPERT ANALYSIS FROM AACE 2018










