User login
JAK-1 inhibitors heading for validation in phase III trials
LONDON – ABT-494 and filgotinib – two investigational and highly selective oral Janus kinase-1 inhibitors – are both showing promise in the treatment of patients with rheumatoid arthritis, according to the results of two separate phase II studies presented at the European Congress of Rheumatology.
In the BALANCE-2 study, 62%-80% patients who had an inadequate response to methotrexate alone achieved the primary endpoint of an ACR20 response after 12 weeks of combination treatment with methotrexate and ABT-494, depending on the dose used, versus 46% for placebo plus methotrexate. The secondary endpoint of ACR50 was reached by a respective 38%-50% vs. 18%, and ACR70 response was achieved by 16%-28% vs. 6%.
And in the DARWIN-1 study, 56%-79% of patients treated with different doses of filgotinib plus methotrexate achieved the trial’s primary endpoint, which was again ACR20 at 12 weeks, versus 44% for placebo plus methotrexate. ACR50 and ACR70 responses were also similarly high and maintained up to 24 weeks of follow-up.
Both drugs had safety and tolerability data that supported their further development, the respective study investigators said.
“I think the results are rather straightforward. There was significant improvement in signs and symptoms of RA with fast onset,” said René Westhovens, MD, PhD, of the University of Leuven (Belgium), who presented the data from the DARWIN-1 study. “These robust data support the future development of filgotinib in RA,” he said.
Mark Genovese, MD, of Stanford (Calif.) University, who presented the findings of the BALANCE-2 study, said: “ABT-494 has been shown to have significant improvements in symptoms and signs [of RA] based on our endpoints of ACR [response], DAS[28], and CDAI [clinical disease activity index].” Like ACR50 and ACR70, DAS28 and CDAI were secondary efficacy endpoints and showed significantly greater changes from baseline versus placebo, started from around 2 weeks.
In an interview, Peter Taylor, PhD, who chaired the session at the meeting where the findings were presented, said: “We’ve seen a lot of data about JAK inhibitors at various stages of development at EULAR 2016, with varying selectivity, and the clinical data unequivocally validates Janus kinases as a therapeutic target.”
Dr. Taylor, the Norman Collisson Professor of Musculoskeletal Sciences at the University of Oxford (England), added: “[JAK inhibitors] show very significant promise with favorable safety data overall, but there are subtle differences between the drugs which need further detailed analysis to understand what it means in a clinical context.”
BALANCE-2 was a double-blind, placebo-controlled, dose-ranging phase IIB study designed to look at the safety and efficacy of ABT-494 in adult patients with moderately to severely active rheumatoid arthritis who had an inadequate response to methotrexate. Five doses of ABT-494 were tested: four given twice-daily (3, 6, 12, and 18 mg) and one given once-daily (24 mg). A total of 300 patients were enrolled and 299 were randomized, 50 to placebo, 50 each to the once-daily doses, and 49 to the twice-daily dose group. The mean weekly methotrexate dose at baseline was 14-16 mg across the groups.
“In general, the safety and tolerability of ABT-494 was satisfactory at the doses tested, consistent with what would have been expected,” Dr. Genovese said.
There was a numerically higher rate of any adverse event in the groups treated with ABT-494, at 40%, 46%, 58%, and 50% for the twice-daily regimens of 3, 6, 12, and 18 mg, as well as 35% for the once-daily 24-mg dose. The rate was 26% for placebo plus methotrexate.
Infections occurred in a respective 20%, 14%, 24%, 22% across the twice-daily dosing groups, respectively, compared with 18% for the once-daily 24-mg dose and 14% for placebo plus methotrexate, he noted. While there were some grade 2-3 abnormalities in liver enzymes and dose-related decreases in hemoglobin seen at higher doses, these did not appear to have significant clinical impact. The ratio of high-density lipoprotein cholesterol (HDL-C) to low-density lipoprotein cholesterol (LDL-C) was also affected slightly.
DARWIN-1 involved a total of 599 enrolled and 594 randomized and exposed patients with RA treated with placebo plus methotrexate or methotrexate plus one of six dosing regimens of filgotinib: 50, 100, or 200 mg once daily, or 25, 50, or 100 mg twice daily, for 24 weeks, with around 85 patients in each group. Each patient previously had an inadequate response to methotrexate alone. At the 12-week halfway point, patients taking placebo and the 50-mg dose could be reassigned to filgotinib 100 mg once daily or 50 mg twice daily if their tender or swollen joint counts had not improved. The mean weekly dose of methotrexate at baseline was 16.4-17.5 mg across the groups.
In addition to the improved ACR responses, significant improvements with filgotinib versus placebo were seen in the secondary endpoints of DAS28 (including DAS28 based on C-reactive protein), CDAI, and the Health Assessment Questionnaire-Disability Index.
There were infrequent serious adverse events, which included serious infections, and adverse events leading to discontinuations, Dr. Westhovens observed, and nothing that would not have been expected or different from placebo. There was a small decrease in neutrophil counts and increase in creatinine, but neither had any clinical consequences. Interestingly, there was a dose-dependent increase in hemoglobin but no reduction in lymphocyte counts, he said. HDL-C increased more than LDL-C.
Five phase III trials with ABT-494 are currently underway in patients with RA:
• SELECT-COMPARE will enroll an estimated 1,500 RA patients who have had an inadequate response to a stable dose of methotrexate and will compare additional treatment with ABT-494 against additional treatment with adalimumab (Humira) or placebo.
• SELECT-NEXT will enroll an estimated 600 RA patients who have had an inadequate response to stable doses of conventional synthetic disease-modifying antirheumatic drugs (csDMARDs) and are then given ABT-494 or placebo on top.
• SELECT-BEYOND will enroll around 450 RA patients on stable csDMARDs who have an inadequate response or intolerance to biologic DMARDs and compare adding ABT-494 or placebo.
• SELECT-MONOTHERAPY will enroll 600 RA patients who have had an inadequate methotrexate response and compare ABT-494 monotherapy to methotrexate monotherapy.
• SELECT-EARLY will enroll 975 methotrexate-naive, moderately-to-severely active RA patients and compare giving ABT-494 monotherapy to methotrexate monotherapy.
Most of these trials should have primary endpoint data available for analysis by mid to late 2017 or 2018 and be finished by 2020 or 2021.
Filgotinib, formerly known as GLPG0634, is also about to enter phase III trials, but the details of these trials have not yet been revealed other than that they will begin mid-2016.
The BALANCE-2 study was funded by AbbVie. Dr. Genovese is a consultant for, and has received grants from AbbVie, Eli Lilly, Astellas, Vertex, Pfizer, Galapagos, and Gilead.
The DARWIN-1 study was funded by Galapagos. Dr. Westhovens is the principal investigator for the study. He also disclosed receiving research funding from Roche and speaker’s honoraria from Bristol-Myers Squibb.
Dr. Taylor was not involved in either study but has consulted for Eli Lilly, Pfizer, and Galapagos.
LONDON – ABT-494 and filgotinib – two investigational and highly selective oral Janus kinase-1 inhibitors – are both showing promise in the treatment of patients with rheumatoid arthritis, according to the results of two separate phase II studies presented at the European Congress of Rheumatology.
In the BALANCE-2 study, 62%-80% patients who had an inadequate response to methotrexate alone achieved the primary endpoint of an ACR20 response after 12 weeks of combination treatment with methotrexate and ABT-494, depending on the dose used, versus 46% for placebo plus methotrexate. The secondary endpoint of ACR50 was reached by a respective 38%-50% vs. 18%, and ACR70 response was achieved by 16%-28% vs. 6%.
And in the DARWIN-1 study, 56%-79% of patients treated with different doses of filgotinib plus methotrexate achieved the trial’s primary endpoint, which was again ACR20 at 12 weeks, versus 44% for placebo plus methotrexate. ACR50 and ACR70 responses were also similarly high and maintained up to 24 weeks of follow-up.
Both drugs had safety and tolerability data that supported their further development, the respective study investigators said.
“I think the results are rather straightforward. There was significant improvement in signs and symptoms of RA with fast onset,” said René Westhovens, MD, PhD, of the University of Leuven (Belgium), who presented the data from the DARWIN-1 study. “These robust data support the future development of filgotinib in RA,” he said.
Mark Genovese, MD, of Stanford (Calif.) University, who presented the findings of the BALANCE-2 study, said: “ABT-494 has been shown to have significant improvements in symptoms and signs [of RA] based on our endpoints of ACR [response], DAS[28], and CDAI [clinical disease activity index].” Like ACR50 and ACR70, DAS28 and CDAI were secondary efficacy endpoints and showed significantly greater changes from baseline versus placebo, started from around 2 weeks.
In an interview, Peter Taylor, PhD, who chaired the session at the meeting where the findings were presented, said: “We’ve seen a lot of data about JAK inhibitors at various stages of development at EULAR 2016, with varying selectivity, and the clinical data unequivocally validates Janus kinases as a therapeutic target.”
Dr. Taylor, the Norman Collisson Professor of Musculoskeletal Sciences at the University of Oxford (England), added: “[JAK inhibitors] show very significant promise with favorable safety data overall, but there are subtle differences between the drugs which need further detailed analysis to understand what it means in a clinical context.”
BALANCE-2 was a double-blind, placebo-controlled, dose-ranging phase IIB study designed to look at the safety and efficacy of ABT-494 in adult patients with moderately to severely active rheumatoid arthritis who had an inadequate response to methotrexate. Five doses of ABT-494 were tested: four given twice-daily (3, 6, 12, and 18 mg) and one given once-daily (24 mg). A total of 300 patients were enrolled and 299 were randomized, 50 to placebo, 50 each to the once-daily doses, and 49 to the twice-daily dose group. The mean weekly methotrexate dose at baseline was 14-16 mg across the groups.
“In general, the safety and tolerability of ABT-494 was satisfactory at the doses tested, consistent with what would have been expected,” Dr. Genovese said.
There was a numerically higher rate of any adverse event in the groups treated with ABT-494, at 40%, 46%, 58%, and 50% for the twice-daily regimens of 3, 6, 12, and 18 mg, as well as 35% for the once-daily 24-mg dose. The rate was 26% for placebo plus methotrexate.
Infections occurred in a respective 20%, 14%, 24%, 22% across the twice-daily dosing groups, respectively, compared with 18% for the once-daily 24-mg dose and 14% for placebo plus methotrexate, he noted. While there were some grade 2-3 abnormalities in liver enzymes and dose-related decreases in hemoglobin seen at higher doses, these did not appear to have significant clinical impact. The ratio of high-density lipoprotein cholesterol (HDL-C) to low-density lipoprotein cholesterol (LDL-C) was also affected slightly.
DARWIN-1 involved a total of 599 enrolled and 594 randomized and exposed patients with RA treated with placebo plus methotrexate or methotrexate plus one of six dosing regimens of filgotinib: 50, 100, or 200 mg once daily, or 25, 50, or 100 mg twice daily, for 24 weeks, with around 85 patients in each group. Each patient previously had an inadequate response to methotrexate alone. At the 12-week halfway point, patients taking placebo and the 50-mg dose could be reassigned to filgotinib 100 mg once daily or 50 mg twice daily if their tender or swollen joint counts had not improved. The mean weekly dose of methotrexate at baseline was 16.4-17.5 mg across the groups.
In addition to the improved ACR responses, significant improvements with filgotinib versus placebo were seen in the secondary endpoints of DAS28 (including DAS28 based on C-reactive protein), CDAI, and the Health Assessment Questionnaire-Disability Index.
There were infrequent serious adverse events, which included serious infections, and adverse events leading to discontinuations, Dr. Westhovens observed, and nothing that would not have been expected or different from placebo. There was a small decrease in neutrophil counts and increase in creatinine, but neither had any clinical consequences. Interestingly, there was a dose-dependent increase in hemoglobin but no reduction in lymphocyte counts, he said. HDL-C increased more than LDL-C.
Five phase III trials with ABT-494 are currently underway in patients with RA:
• SELECT-COMPARE will enroll an estimated 1,500 RA patients who have had an inadequate response to a stable dose of methotrexate and will compare additional treatment with ABT-494 against additional treatment with adalimumab (Humira) or placebo.
• SELECT-NEXT will enroll an estimated 600 RA patients who have had an inadequate response to stable doses of conventional synthetic disease-modifying antirheumatic drugs (csDMARDs) and are then given ABT-494 or placebo on top.
• SELECT-BEYOND will enroll around 450 RA patients on stable csDMARDs who have an inadequate response or intolerance to biologic DMARDs and compare adding ABT-494 or placebo.
• SELECT-MONOTHERAPY will enroll 600 RA patients who have had an inadequate methotrexate response and compare ABT-494 monotherapy to methotrexate monotherapy.
• SELECT-EARLY will enroll 975 methotrexate-naive, moderately-to-severely active RA patients and compare giving ABT-494 monotherapy to methotrexate monotherapy.
Most of these trials should have primary endpoint data available for analysis by mid to late 2017 or 2018 and be finished by 2020 or 2021.
Filgotinib, formerly known as GLPG0634, is also about to enter phase III trials, but the details of these trials have not yet been revealed other than that they will begin mid-2016.
The BALANCE-2 study was funded by AbbVie. Dr. Genovese is a consultant for, and has received grants from AbbVie, Eli Lilly, Astellas, Vertex, Pfizer, Galapagos, and Gilead.
The DARWIN-1 study was funded by Galapagos. Dr. Westhovens is the principal investigator for the study. He also disclosed receiving research funding from Roche and speaker’s honoraria from Bristol-Myers Squibb.
Dr. Taylor was not involved in either study but has consulted for Eli Lilly, Pfizer, and Galapagos.
LONDON – ABT-494 and filgotinib – two investigational and highly selective oral Janus kinase-1 inhibitors – are both showing promise in the treatment of patients with rheumatoid arthritis, according to the results of two separate phase II studies presented at the European Congress of Rheumatology.
In the BALANCE-2 study, 62%-80% patients who had an inadequate response to methotrexate alone achieved the primary endpoint of an ACR20 response after 12 weeks of combination treatment with methotrexate and ABT-494, depending on the dose used, versus 46% for placebo plus methotrexate. The secondary endpoint of ACR50 was reached by a respective 38%-50% vs. 18%, and ACR70 response was achieved by 16%-28% vs. 6%.
And in the DARWIN-1 study, 56%-79% of patients treated with different doses of filgotinib plus methotrexate achieved the trial’s primary endpoint, which was again ACR20 at 12 weeks, versus 44% for placebo plus methotrexate. ACR50 and ACR70 responses were also similarly high and maintained up to 24 weeks of follow-up.
Both drugs had safety and tolerability data that supported their further development, the respective study investigators said.
“I think the results are rather straightforward. There was significant improvement in signs and symptoms of RA with fast onset,” said René Westhovens, MD, PhD, of the University of Leuven (Belgium), who presented the data from the DARWIN-1 study. “These robust data support the future development of filgotinib in RA,” he said.
Mark Genovese, MD, of Stanford (Calif.) University, who presented the findings of the BALANCE-2 study, said: “ABT-494 has been shown to have significant improvements in symptoms and signs [of RA] based on our endpoints of ACR [response], DAS[28], and CDAI [clinical disease activity index].” Like ACR50 and ACR70, DAS28 and CDAI were secondary efficacy endpoints and showed significantly greater changes from baseline versus placebo, started from around 2 weeks.
In an interview, Peter Taylor, PhD, who chaired the session at the meeting where the findings were presented, said: “We’ve seen a lot of data about JAK inhibitors at various stages of development at EULAR 2016, with varying selectivity, and the clinical data unequivocally validates Janus kinases as a therapeutic target.”
Dr. Taylor, the Norman Collisson Professor of Musculoskeletal Sciences at the University of Oxford (England), added: “[JAK inhibitors] show very significant promise with favorable safety data overall, but there are subtle differences between the drugs which need further detailed analysis to understand what it means in a clinical context.”
BALANCE-2 was a double-blind, placebo-controlled, dose-ranging phase IIB study designed to look at the safety and efficacy of ABT-494 in adult patients with moderately to severely active rheumatoid arthritis who had an inadequate response to methotrexate. Five doses of ABT-494 were tested: four given twice-daily (3, 6, 12, and 18 mg) and one given once-daily (24 mg). A total of 300 patients were enrolled and 299 were randomized, 50 to placebo, 50 each to the once-daily doses, and 49 to the twice-daily dose group. The mean weekly methotrexate dose at baseline was 14-16 mg across the groups.
“In general, the safety and tolerability of ABT-494 was satisfactory at the doses tested, consistent with what would have been expected,” Dr. Genovese said.
There was a numerically higher rate of any adverse event in the groups treated with ABT-494, at 40%, 46%, 58%, and 50% for the twice-daily regimens of 3, 6, 12, and 18 mg, as well as 35% for the once-daily 24-mg dose. The rate was 26% for placebo plus methotrexate.
Infections occurred in a respective 20%, 14%, 24%, 22% across the twice-daily dosing groups, respectively, compared with 18% for the once-daily 24-mg dose and 14% for placebo plus methotrexate, he noted. While there were some grade 2-3 abnormalities in liver enzymes and dose-related decreases in hemoglobin seen at higher doses, these did not appear to have significant clinical impact. The ratio of high-density lipoprotein cholesterol (HDL-C) to low-density lipoprotein cholesterol (LDL-C) was also affected slightly.
DARWIN-1 involved a total of 599 enrolled and 594 randomized and exposed patients with RA treated with placebo plus methotrexate or methotrexate plus one of six dosing regimens of filgotinib: 50, 100, or 200 mg once daily, or 25, 50, or 100 mg twice daily, for 24 weeks, with around 85 patients in each group. Each patient previously had an inadequate response to methotrexate alone. At the 12-week halfway point, patients taking placebo and the 50-mg dose could be reassigned to filgotinib 100 mg once daily or 50 mg twice daily if their tender or swollen joint counts had not improved. The mean weekly dose of methotrexate at baseline was 16.4-17.5 mg across the groups.
In addition to the improved ACR responses, significant improvements with filgotinib versus placebo were seen in the secondary endpoints of DAS28 (including DAS28 based on C-reactive protein), CDAI, and the Health Assessment Questionnaire-Disability Index.
There were infrequent serious adverse events, which included serious infections, and adverse events leading to discontinuations, Dr. Westhovens observed, and nothing that would not have been expected or different from placebo. There was a small decrease in neutrophil counts and increase in creatinine, but neither had any clinical consequences. Interestingly, there was a dose-dependent increase in hemoglobin but no reduction in lymphocyte counts, he said. HDL-C increased more than LDL-C.
Five phase III trials with ABT-494 are currently underway in patients with RA:
• SELECT-COMPARE will enroll an estimated 1,500 RA patients who have had an inadequate response to a stable dose of methotrexate and will compare additional treatment with ABT-494 against additional treatment with adalimumab (Humira) or placebo.
• SELECT-NEXT will enroll an estimated 600 RA patients who have had an inadequate response to stable doses of conventional synthetic disease-modifying antirheumatic drugs (csDMARDs) and are then given ABT-494 or placebo on top.
• SELECT-BEYOND will enroll around 450 RA patients on stable csDMARDs who have an inadequate response or intolerance to biologic DMARDs and compare adding ABT-494 or placebo.
• SELECT-MONOTHERAPY will enroll 600 RA patients who have had an inadequate methotrexate response and compare ABT-494 monotherapy to methotrexate monotherapy.
• SELECT-EARLY will enroll 975 methotrexate-naive, moderately-to-severely active RA patients and compare giving ABT-494 monotherapy to methotrexate monotherapy.
Most of these trials should have primary endpoint data available for analysis by mid to late 2017 or 2018 and be finished by 2020 or 2021.
Filgotinib, formerly known as GLPG0634, is also about to enter phase III trials, but the details of these trials have not yet been revealed other than that they will begin mid-2016.
The BALANCE-2 study was funded by AbbVie. Dr. Genovese is a consultant for, and has received grants from AbbVie, Eli Lilly, Astellas, Vertex, Pfizer, Galapagos, and Gilead.
The DARWIN-1 study was funded by Galapagos. Dr. Westhovens is the principal investigator for the study. He also disclosed receiving research funding from Roche and speaker’s honoraria from Bristol-Myers Squibb.
Dr. Taylor was not involved in either study but has consulted for Eli Lilly, Pfizer, and Galapagos.
Key clinical point: Two new oral Janus kinase inhibitors selective for JAK-1 have shown promising efficacy and safety in separate phase II trials.
Major finding: ACR20 responses were achieved by 62%-80% of patients treated with different dosing regimens of ABT-494 plus methotrexate (vs. 46% with placebo plus methotrexate) in one trial and by 56%-79% of patients treated with different dosing regimens of filgotinib plus methotrexate (vs. 44% with placebo and methotrexate) in the other.
Data source: Two phase II studies addressing the efficacy and safety of the selective JAK-1 inhibitors ABT-494 and filgotinib in patients with rheumatoid arthritis and an inadequate response to methotrexate.
Disclosures: The BALANCE-2 study was funded by AbbVie. Dr. Genovese is a consultant for, and has received grants from AbbVie, Eli Lilly, Astellas, Vertex, Pfizer, Galapagos, and Gilead. The DARWIN-1 study was funded by Galapagos. Dr. Westhovens is the principal investigator for the study. He also disclosed receiving research funding from Roche and speaker’s honoraria from Bristol-Myers Squibb. Dr. Taylor has consulted for Eli Lilly, Pfizer, and Galapagos.
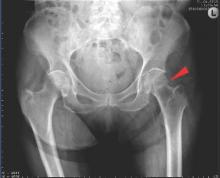
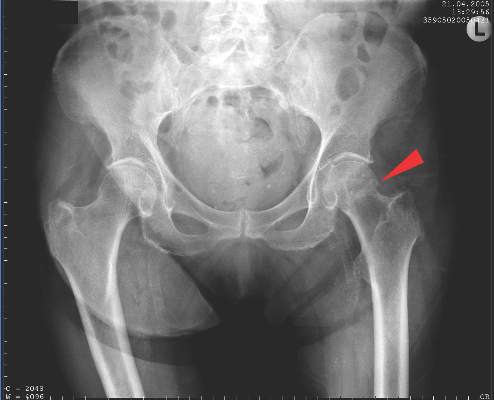
New fragility fracture recommendations emphasize coordination of care
LONDON – The European League Against Rheumatism and the European Federation of National Associations of Orthopaedics and Traumatology have joined forces to develop recommendations for the prevention and management of fragility fractures.
Such fractures are common in men and women over the age of 50 years and can lead to repeat fracture in some patients. The recommendations are unique as they are the first to consider both acute orthopedic and postfracture rheumatologic care, said Willem F. Lems, MD, PhD, of the Amsterdam Rheumatology and Immunology Centre.
At the European Congress of Rheumatology, Dr. Lems provided an overview of the draft recommendations, noting that there would be several overarching principles, one of which recognized the multidisciplinary nature of caring for someone with a fragility fracture. An important point is not who is taking care of the patient, but that the patient is given the best possible care within the multidisciplinary framework.
What constitutes optimal care of course depends on the clinical situation, notably the type of fracture and the age of the patient, and optimal care in all phases of presentation (pre-, peri- and postoperative) can have an important effect on a patient’s outcome. The prevention of subsequent fractures is a key focus, with the recommendation that all patients should be investigated systematically and those deemed at high risk for another fracture should be prescribed both pharmacologic and nonpharmacologic interventions as appropriate. Patient education is also considered important.
As for all EULAR-developed recommendations, standard procedures were followed that involved convening an expert scientific advisory committee and using the Delphi technique to come up with the most important research questions that would be used to formulate the final 10 recommendations. Four of the recommendations cover the acute care setting and six provide advice on postfracture care.
The first of the acute care recommendations looks at pre- and perioperative management of a fragility fracture and highlights that, within 24-48 hours of admission, patients should receive adequate pain and fluid management and treatment, including early surgery if appropriate. This is based on evidence that better outcomes can be achieved in terms of both morbidity and mortality if patients can be seen and managed quickly.
Another of the acute care recommendations focuses on orthogeriatric care, noting that the orthopedic surgeon and a dedicated orthogeriatric team should work together, particularly for elderly patients who have suffered a hip fracture. Key elements here are the management of and prevention of delirium, deep vein thrombosis, pressure sores, and malnutrition.
As for actual fracture treatment, a balanced approach is advised when deciding upon a surgical or nonsurgical approach, especially because this is likely to be an older population with other comorbidities. Only one in three vertebral fractures are symptomatic and only about 10% of patients will be hospitalized for pain. Analgesics, modifying activities, and bracing can be options here. Surgical options for distal radial fracture, hip fracture, and trochanteric and femoral neck fractures are included.
The fourth recommendation looks at the organization of postfracture care and the need for a systematic approach to identify those who may be at risk for subsequent fractures, starting with the suggestion that any patient older than 50 years with a recent fracture should be assessed. The fifth recommendation addresses ways to evaluate this risk, such as looking at the clinical risk factors, performing bone scans and imaging, and screening for underlying osteoporosis or metabolic disorders.
Implementation is the next step, and the sixth recommendation suggests ways these recommendations could be integrated into routine practice. Often one of the biggest barriers to effective postfracture care is the lack of patient, and sometimes clinician, awareness of the risk for a subsequent fracture. This recommendation looks at the role of a possible local fracture liaison service or facilitator to coordinate between the various members of the multidisciplinary team from secondary (orthopedic surgeons, rheumatologists, endocrinologists, and geriatricians) to primary care.
The seventh recommendation addresses rehabilitation and the need to initiate physical training and muscle strengthening as early as possible after the initial fracture, with long-term continuation of balance training and fall prevention.
The final three recommendations focus on how to educate patients about their risk factors, need for follow-up, and the duration of any pharmacologic or nonpharmacologic therapy that they may need. Nonpharmacologic options might include stopping smoking, limiting alcohol intake, as well as taking supplements such as calcium or vitamin D. There will be specific guidance on the use of calcium and vitamin D, which have both pros and cons, but the optimal dosage appears to be 1,000–1,200 mg/day for calcium and 800 IU/day for vitamin D.
Pharmacologic options to prevent subsequent fragility fractures include the bisphosphonates alendronate, risedronate, and zoledronic acid (Reclast), and also the monoclonal antibody denosumab (Prolia). These are the only drugs that have been shown to reduced the risk for vertebral, nonvertebral, and hip fractures in primary analyses. Adherence, tolerance, and regular monitoring are key, and a five-step plan is suggested to aid clinical decision making that covers case finding, risk evaluation, differential diagnosis, treatment, and follow-up.
The recommendations are being finalized and should be available for publication later this year. The recommendations task force also plans to propose a research agenda.
Dr. Lems had no relevant disclosures.
LONDON – The European League Against Rheumatism and the European Federation of National Associations of Orthopaedics and Traumatology have joined forces to develop recommendations for the prevention and management of fragility fractures.
Such fractures are common in men and women over the age of 50 years and can lead to repeat fracture in some patients. The recommendations are unique as they are the first to consider both acute orthopedic and postfracture rheumatologic care, said Willem F. Lems, MD, PhD, of the Amsterdam Rheumatology and Immunology Centre.
At the European Congress of Rheumatology, Dr. Lems provided an overview of the draft recommendations, noting that there would be several overarching principles, one of which recognized the multidisciplinary nature of caring for someone with a fragility fracture. An important point is not who is taking care of the patient, but that the patient is given the best possible care within the multidisciplinary framework.
What constitutes optimal care of course depends on the clinical situation, notably the type of fracture and the age of the patient, and optimal care in all phases of presentation (pre-, peri- and postoperative) can have an important effect on a patient’s outcome. The prevention of subsequent fractures is a key focus, with the recommendation that all patients should be investigated systematically and those deemed at high risk for another fracture should be prescribed both pharmacologic and nonpharmacologic interventions as appropriate. Patient education is also considered important.
As for all EULAR-developed recommendations, standard procedures were followed that involved convening an expert scientific advisory committee and using the Delphi technique to come up with the most important research questions that would be used to formulate the final 10 recommendations. Four of the recommendations cover the acute care setting and six provide advice on postfracture care.
The first of the acute care recommendations looks at pre- and perioperative management of a fragility fracture and highlights that, within 24-48 hours of admission, patients should receive adequate pain and fluid management and treatment, including early surgery if appropriate. This is based on evidence that better outcomes can be achieved in terms of both morbidity and mortality if patients can be seen and managed quickly.
Another of the acute care recommendations focuses on orthogeriatric care, noting that the orthopedic surgeon and a dedicated orthogeriatric team should work together, particularly for elderly patients who have suffered a hip fracture. Key elements here are the management of and prevention of delirium, deep vein thrombosis, pressure sores, and malnutrition.
As for actual fracture treatment, a balanced approach is advised when deciding upon a surgical or nonsurgical approach, especially because this is likely to be an older population with other comorbidities. Only one in three vertebral fractures are symptomatic and only about 10% of patients will be hospitalized for pain. Analgesics, modifying activities, and bracing can be options here. Surgical options for distal radial fracture, hip fracture, and trochanteric and femoral neck fractures are included.
The fourth recommendation looks at the organization of postfracture care and the need for a systematic approach to identify those who may be at risk for subsequent fractures, starting with the suggestion that any patient older than 50 years with a recent fracture should be assessed. The fifth recommendation addresses ways to evaluate this risk, such as looking at the clinical risk factors, performing bone scans and imaging, and screening for underlying osteoporosis or metabolic disorders.
Implementation is the next step, and the sixth recommendation suggests ways these recommendations could be integrated into routine practice. Often one of the biggest barriers to effective postfracture care is the lack of patient, and sometimes clinician, awareness of the risk for a subsequent fracture. This recommendation looks at the role of a possible local fracture liaison service or facilitator to coordinate between the various members of the multidisciplinary team from secondary (orthopedic surgeons, rheumatologists, endocrinologists, and geriatricians) to primary care.
The seventh recommendation addresses rehabilitation and the need to initiate physical training and muscle strengthening as early as possible after the initial fracture, with long-term continuation of balance training and fall prevention.
The final three recommendations focus on how to educate patients about their risk factors, need for follow-up, and the duration of any pharmacologic or nonpharmacologic therapy that they may need. Nonpharmacologic options might include stopping smoking, limiting alcohol intake, as well as taking supplements such as calcium or vitamin D. There will be specific guidance on the use of calcium and vitamin D, which have both pros and cons, but the optimal dosage appears to be 1,000–1,200 mg/day for calcium and 800 IU/day for vitamin D.
Pharmacologic options to prevent subsequent fragility fractures include the bisphosphonates alendronate, risedronate, and zoledronic acid (Reclast), and also the monoclonal antibody denosumab (Prolia). These are the only drugs that have been shown to reduced the risk for vertebral, nonvertebral, and hip fractures in primary analyses. Adherence, tolerance, and regular monitoring are key, and a five-step plan is suggested to aid clinical decision making that covers case finding, risk evaluation, differential diagnosis, treatment, and follow-up.
The recommendations are being finalized and should be available for publication later this year. The recommendations task force also plans to propose a research agenda.
Dr. Lems had no relevant disclosures.
LONDON – The European League Against Rheumatism and the European Federation of National Associations of Orthopaedics and Traumatology have joined forces to develop recommendations for the prevention and management of fragility fractures.
Such fractures are common in men and women over the age of 50 years and can lead to repeat fracture in some patients. The recommendations are unique as they are the first to consider both acute orthopedic and postfracture rheumatologic care, said Willem F. Lems, MD, PhD, of the Amsterdam Rheumatology and Immunology Centre.
At the European Congress of Rheumatology, Dr. Lems provided an overview of the draft recommendations, noting that there would be several overarching principles, one of which recognized the multidisciplinary nature of caring for someone with a fragility fracture. An important point is not who is taking care of the patient, but that the patient is given the best possible care within the multidisciplinary framework.
What constitutes optimal care of course depends on the clinical situation, notably the type of fracture and the age of the patient, and optimal care in all phases of presentation (pre-, peri- and postoperative) can have an important effect on a patient’s outcome. The prevention of subsequent fractures is a key focus, with the recommendation that all patients should be investigated systematically and those deemed at high risk for another fracture should be prescribed both pharmacologic and nonpharmacologic interventions as appropriate. Patient education is also considered important.
As for all EULAR-developed recommendations, standard procedures were followed that involved convening an expert scientific advisory committee and using the Delphi technique to come up with the most important research questions that would be used to formulate the final 10 recommendations. Four of the recommendations cover the acute care setting and six provide advice on postfracture care.
The first of the acute care recommendations looks at pre- and perioperative management of a fragility fracture and highlights that, within 24-48 hours of admission, patients should receive adequate pain and fluid management and treatment, including early surgery if appropriate. This is based on evidence that better outcomes can be achieved in terms of both morbidity and mortality if patients can be seen and managed quickly.
Another of the acute care recommendations focuses on orthogeriatric care, noting that the orthopedic surgeon and a dedicated orthogeriatric team should work together, particularly for elderly patients who have suffered a hip fracture. Key elements here are the management of and prevention of delirium, deep vein thrombosis, pressure sores, and malnutrition.
As for actual fracture treatment, a balanced approach is advised when deciding upon a surgical or nonsurgical approach, especially because this is likely to be an older population with other comorbidities. Only one in three vertebral fractures are symptomatic and only about 10% of patients will be hospitalized for pain. Analgesics, modifying activities, and bracing can be options here. Surgical options for distal radial fracture, hip fracture, and trochanteric and femoral neck fractures are included.
The fourth recommendation looks at the organization of postfracture care and the need for a systematic approach to identify those who may be at risk for subsequent fractures, starting with the suggestion that any patient older than 50 years with a recent fracture should be assessed. The fifth recommendation addresses ways to evaluate this risk, such as looking at the clinical risk factors, performing bone scans and imaging, and screening for underlying osteoporosis or metabolic disorders.
Implementation is the next step, and the sixth recommendation suggests ways these recommendations could be integrated into routine practice. Often one of the biggest barriers to effective postfracture care is the lack of patient, and sometimes clinician, awareness of the risk for a subsequent fracture. This recommendation looks at the role of a possible local fracture liaison service or facilitator to coordinate between the various members of the multidisciplinary team from secondary (orthopedic surgeons, rheumatologists, endocrinologists, and geriatricians) to primary care.
The seventh recommendation addresses rehabilitation and the need to initiate physical training and muscle strengthening as early as possible after the initial fracture, with long-term continuation of balance training and fall prevention.
The final three recommendations focus on how to educate patients about their risk factors, need for follow-up, and the duration of any pharmacologic or nonpharmacologic therapy that they may need. Nonpharmacologic options might include stopping smoking, limiting alcohol intake, as well as taking supplements such as calcium or vitamin D. There will be specific guidance on the use of calcium and vitamin D, which have both pros and cons, but the optimal dosage appears to be 1,000–1,200 mg/day for calcium and 800 IU/day for vitamin D.
Pharmacologic options to prevent subsequent fragility fractures include the bisphosphonates alendronate, risedronate, and zoledronic acid (Reclast), and also the monoclonal antibody denosumab (Prolia). These are the only drugs that have been shown to reduced the risk for vertebral, nonvertebral, and hip fractures in primary analyses. Adherence, tolerance, and regular monitoring are key, and a five-step plan is suggested to aid clinical decision making that covers case finding, risk evaluation, differential diagnosis, treatment, and follow-up.
The recommendations are being finalized and should be available for publication later this year. The recommendations task force also plans to propose a research agenda.
Dr. Lems had no relevant disclosures.
AT THE EULAR 2016 CONGRESS
HCQ eye toxicity needs experience to assess
LONDON – Retinopathy in patients taking long-term hydroxychloroquine for rheumatic conditions requires assessment by those experienced with specialized ophthalmic imaging, according to study findings presented at the European Congress of Rheumatology.
Nonspecific abnormalities, which often are unrelated to hydroxychloroquine (HCQ), can be seen with many of the tests recommended by current ophthalmology guidelines. These changes need “careful interpretation by retina specialists,” the study’s investigators wrote in a poster presentation.
HCQ is used widely for the treatment of systemic lupus erythematosus (SLE), rheumatoid arthritis, and many other inflammatory or autoimmune conditions, but it can cause irreversible eye damage and is often associated with prolonged (greater than 5 years) use. Specifically, it can cause a type of end-stage retinopathy called bull’s-eye maculopathy, which is where the fovea becomes hyperpigmented, much like the bull’s-eye on a dartboard. This can lead to substantial vision loss (blind spots) if not caught early.
Although it is reasonably rare to develop end-stage retinopathy, there is currently no treatment for HCQ-induced retinopathy. Stopping the drug may not necessarily stop the retinal damage, and drug withdrawal may not be an option in many patients given the lack of alternative options to treat the symptoms of SLE, study author and ophthalmologist Syed Mahmood Ali Shah, MBBS, MD, said in an interview.
Dr. Shah and his associates at Johns Hopkins University in Baltimore reported on applying the 2011 American Academy of Ophthalmology (AAO) guidelines on screening for HCQ retinopathy (Ophthalmology. 2011;118:415-22) to an academic practice. They also estimated the prevalence of HCQ retinopathy among 135 consecutively treated patients with SLE using recommended tests. The mean duration of HCQ use was 12.5 years.
The 2011 AAO guidelines – which in March 2016 were updated (Ophthalmology 2016 Jun;123:1386-94) – recommended the use of three “ancillary” tests in addition to the usual clinical ophthalmic examination and assessment of visual fields: optical coherence tomography (OCT), fundus autofluorescence (FAF), and multifocal electroretinography (mfERG). Dr. Shah and his colleagues used these three tests together with eye-tracking microperimetry (MP) as a substitute for Humphrey Visual Fields (HVF), which is a common visual field test used in the United States.
One difference between the 2011 guidelines and 2016 revision is that “the baseline exam can now be performed relying [only] on the fundus exam, with additional imaging required only for abnormal patients,” Dr. Shah said. “Overall, the guidelines have not changed on how often and how much you follow up,” he added. “The change is that there is no need to do these tests at baseline unless changes of the fundus are present.” However, OCT has become more widely used in many offices and has been recognized as the most useful objective test and shall be performed if there are any abnormal findings of the fundus.
A total of 266 eyes were examined using these imaging methods and interpreted by experienced retina specialists. Overall, HCQ-related abnormalities were noted in 14 (5%) eyes using OCT, 18 (7%) using FAF, 27 (10%) eyes using mfERG, and 20 (7%) using MP.
MP had the lowest discrepancy between the overall number of eyes with abnormalities (72 [27%] of 266) detected and the number of eyes with abnormalities related to HCQ (20 [28%] of 72), followed by OCT (21% and 25%, respectively), FAF (19% and 35%) and mfERG (37% and 28%). Only four patients (3%) showed changes in all four tests suggestive of HCQ retinopathy.
In the absence of baseline data from the AAO recommended ancillary tests before the use of HCQ, “it may be difficult to interpret changes seen on these tests since most of the screenings are done by regular ophthalmologists who lack the equipment and experience with specialized testing such as mfERG, FAF, and OCT,” Dr. Shah and his coauthors noted. “We found a substantial number of cases with abnormalities unrelated to HCQ.”
Giving some practical advice, Dr. Shah noted that “before a patient starts treatment with HCQ, they should undergo a baseline ophthalmic assessment. Then if the patient complains of any vision changes, even if they have been taking the drug for less than 5 years, they should be reassessed.”
While repeat follow-up is, of course, necessary, he intimated that it is necessary to find a balance of risk and cost in regard to the frequency of screening for drug-related damage. “The American Academy of Ophthalmology currently recommends that a baseline fundus exam be performed shortly after starting HCQ. Ancillary OCT and visual fields shall only be performed if the fundus is abnormal at this baseline exam. However, since most retina specialists get OCT and visual field testing anyway it is wise to look at these as well,” he suggested. After 5 years of using the drug, they must be seen more regularly, and this is the point when ophthalmologists can decide if this should be every 6 months or annually, with the latter recommended by the AAO guidelines for patients with no additional risk factors.
The study was supported by noncommercial grants. Dr. Shah had no conflicts of interest to disclose.
LONDON – Retinopathy in patients taking long-term hydroxychloroquine for rheumatic conditions requires assessment by those experienced with specialized ophthalmic imaging, according to study findings presented at the European Congress of Rheumatology.
Nonspecific abnormalities, which often are unrelated to hydroxychloroquine (HCQ), can be seen with many of the tests recommended by current ophthalmology guidelines. These changes need “careful interpretation by retina specialists,” the study’s investigators wrote in a poster presentation.
HCQ is used widely for the treatment of systemic lupus erythematosus (SLE), rheumatoid arthritis, and many other inflammatory or autoimmune conditions, but it can cause irreversible eye damage and is often associated with prolonged (greater than 5 years) use. Specifically, it can cause a type of end-stage retinopathy called bull’s-eye maculopathy, which is where the fovea becomes hyperpigmented, much like the bull’s-eye on a dartboard. This can lead to substantial vision loss (blind spots) if not caught early.
Although it is reasonably rare to develop end-stage retinopathy, there is currently no treatment for HCQ-induced retinopathy. Stopping the drug may not necessarily stop the retinal damage, and drug withdrawal may not be an option in many patients given the lack of alternative options to treat the symptoms of SLE, study author and ophthalmologist Syed Mahmood Ali Shah, MBBS, MD, said in an interview.
Dr. Shah and his associates at Johns Hopkins University in Baltimore reported on applying the 2011 American Academy of Ophthalmology (AAO) guidelines on screening for HCQ retinopathy (Ophthalmology. 2011;118:415-22) to an academic practice. They also estimated the prevalence of HCQ retinopathy among 135 consecutively treated patients with SLE using recommended tests. The mean duration of HCQ use was 12.5 years.
The 2011 AAO guidelines – which in March 2016 were updated (Ophthalmology 2016 Jun;123:1386-94) – recommended the use of three “ancillary” tests in addition to the usual clinical ophthalmic examination and assessment of visual fields: optical coherence tomography (OCT), fundus autofluorescence (FAF), and multifocal electroretinography (mfERG). Dr. Shah and his colleagues used these three tests together with eye-tracking microperimetry (MP) as a substitute for Humphrey Visual Fields (HVF), which is a common visual field test used in the United States.
One difference between the 2011 guidelines and 2016 revision is that “the baseline exam can now be performed relying [only] on the fundus exam, with additional imaging required only for abnormal patients,” Dr. Shah said. “Overall, the guidelines have not changed on how often and how much you follow up,” he added. “The change is that there is no need to do these tests at baseline unless changes of the fundus are present.” However, OCT has become more widely used in many offices and has been recognized as the most useful objective test and shall be performed if there are any abnormal findings of the fundus.
A total of 266 eyes were examined using these imaging methods and interpreted by experienced retina specialists. Overall, HCQ-related abnormalities were noted in 14 (5%) eyes using OCT, 18 (7%) using FAF, 27 (10%) eyes using mfERG, and 20 (7%) using MP.
MP had the lowest discrepancy between the overall number of eyes with abnormalities (72 [27%] of 266) detected and the number of eyes with abnormalities related to HCQ (20 [28%] of 72), followed by OCT (21% and 25%, respectively), FAF (19% and 35%) and mfERG (37% and 28%). Only four patients (3%) showed changes in all four tests suggestive of HCQ retinopathy.
In the absence of baseline data from the AAO recommended ancillary tests before the use of HCQ, “it may be difficult to interpret changes seen on these tests since most of the screenings are done by regular ophthalmologists who lack the equipment and experience with specialized testing such as mfERG, FAF, and OCT,” Dr. Shah and his coauthors noted. “We found a substantial number of cases with abnormalities unrelated to HCQ.”
Giving some practical advice, Dr. Shah noted that “before a patient starts treatment with HCQ, they should undergo a baseline ophthalmic assessment. Then if the patient complains of any vision changes, even if they have been taking the drug for less than 5 years, they should be reassessed.”
While repeat follow-up is, of course, necessary, he intimated that it is necessary to find a balance of risk and cost in regard to the frequency of screening for drug-related damage. “The American Academy of Ophthalmology currently recommends that a baseline fundus exam be performed shortly after starting HCQ. Ancillary OCT and visual fields shall only be performed if the fundus is abnormal at this baseline exam. However, since most retina specialists get OCT and visual field testing anyway it is wise to look at these as well,” he suggested. After 5 years of using the drug, they must be seen more regularly, and this is the point when ophthalmologists can decide if this should be every 6 months or annually, with the latter recommended by the AAO guidelines for patients with no additional risk factors.
The study was supported by noncommercial grants. Dr. Shah had no conflicts of interest to disclose.
LONDON – Retinopathy in patients taking long-term hydroxychloroquine for rheumatic conditions requires assessment by those experienced with specialized ophthalmic imaging, according to study findings presented at the European Congress of Rheumatology.
Nonspecific abnormalities, which often are unrelated to hydroxychloroquine (HCQ), can be seen with many of the tests recommended by current ophthalmology guidelines. These changes need “careful interpretation by retina specialists,” the study’s investigators wrote in a poster presentation.
HCQ is used widely for the treatment of systemic lupus erythematosus (SLE), rheumatoid arthritis, and many other inflammatory or autoimmune conditions, but it can cause irreversible eye damage and is often associated with prolonged (greater than 5 years) use. Specifically, it can cause a type of end-stage retinopathy called bull’s-eye maculopathy, which is where the fovea becomes hyperpigmented, much like the bull’s-eye on a dartboard. This can lead to substantial vision loss (blind spots) if not caught early.
Although it is reasonably rare to develop end-stage retinopathy, there is currently no treatment for HCQ-induced retinopathy. Stopping the drug may not necessarily stop the retinal damage, and drug withdrawal may not be an option in many patients given the lack of alternative options to treat the symptoms of SLE, study author and ophthalmologist Syed Mahmood Ali Shah, MBBS, MD, said in an interview.
Dr. Shah and his associates at Johns Hopkins University in Baltimore reported on applying the 2011 American Academy of Ophthalmology (AAO) guidelines on screening for HCQ retinopathy (Ophthalmology. 2011;118:415-22) to an academic practice. They also estimated the prevalence of HCQ retinopathy among 135 consecutively treated patients with SLE using recommended tests. The mean duration of HCQ use was 12.5 years.
The 2011 AAO guidelines – which in March 2016 were updated (Ophthalmology 2016 Jun;123:1386-94) – recommended the use of three “ancillary” tests in addition to the usual clinical ophthalmic examination and assessment of visual fields: optical coherence tomography (OCT), fundus autofluorescence (FAF), and multifocal electroretinography (mfERG). Dr. Shah and his colleagues used these three tests together with eye-tracking microperimetry (MP) as a substitute for Humphrey Visual Fields (HVF), which is a common visual field test used in the United States.
One difference between the 2011 guidelines and 2016 revision is that “the baseline exam can now be performed relying [only] on the fundus exam, with additional imaging required only for abnormal patients,” Dr. Shah said. “Overall, the guidelines have not changed on how often and how much you follow up,” he added. “The change is that there is no need to do these tests at baseline unless changes of the fundus are present.” However, OCT has become more widely used in many offices and has been recognized as the most useful objective test and shall be performed if there are any abnormal findings of the fundus.
A total of 266 eyes were examined using these imaging methods and interpreted by experienced retina specialists. Overall, HCQ-related abnormalities were noted in 14 (5%) eyes using OCT, 18 (7%) using FAF, 27 (10%) eyes using mfERG, and 20 (7%) using MP.
MP had the lowest discrepancy between the overall number of eyes with abnormalities (72 [27%] of 266) detected and the number of eyes with abnormalities related to HCQ (20 [28%] of 72), followed by OCT (21% and 25%, respectively), FAF (19% and 35%) and mfERG (37% and 28%). Only four patients (3%) showed changes in all four tests suggestive of HCQ retinopathy.
In the absence of baseline data from the AAO recommended ancillary tests before the use of HCQ, “it may be difficult to interpret changes seen on these tests since most of the screenings are done by regular ophthalmologists who lack the equipment and experience with specialized testing such as mfERG, FAF, and OCT,” Dr. Shah and his coauthors noted. “We found a substantial number of cases with abnormalities unrelated to HCQ.”
Giving some practical advice, Dr. Shah noted that “before a patient starts treatment with HCQ, they should undergo a baseline ophthalmic assessment. Then if the patient complains of any vision changes, even if they have been taking the drug for less than 5 years, they should be reassessed.”
While repeat follow-up is, of course, necessary, he intimated that it is necessary to find a balance of risk and cost in regard to the frequency of screening for drug-related damage. “The American Academy of Ophthalmology currently recommends that a baseline fundus exam be performed shortly after starting HCQ. Ancillary OCT and visual fields shall only be performed if the fundus is abnormal at this baseline exam. However, since most retina specialists get OCT and visual field testing anyway it is wise to look at these as well,” he suggested. After 5 years of using the drug, they must be seen more regularly, and this is the point when ophthalmologists can decide if this should be every 6 months or annually, with the latter recommended by the AAO guidelines for patients with no additional risk factors.
The study was supported by noncommercial grants. Dr. Shah had no conflicts of interest to disclose.
AT THE EULAR 2016 CONGRESS
Key clinical point: Several eye abnormalities can be mistaken for hydroxychloroquine-related eye toxicity, making specialist ophthalmic assessment paramount.
Major finding: Only four patients (3%) showed changes in all four tests suggestive of HCQ retinopathy.
Data source: Observational study of 135 patients with SLE being seen for suspected hydroxychloroquine-related retinopathy at an academic practice
Disclosures: The study was supported by noncommercial grants. Dr. Shah had no conflicts of interest to disclose.
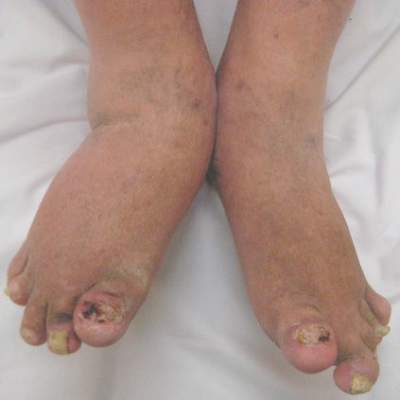
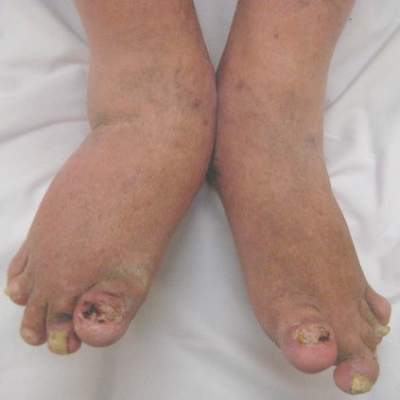
Actemra may reduce scleroderma skin thickening
The interleukin-6 inhibitor tocilizumab (Actemra) substantially reduced skin thickening in patients with systemic sclerosis in the “proof-of concept” faSScinate trial, the results of which have now been published in the Lancet.
The mean change in the primary endpoint of a modified Rodnan skin score at 24 weeks was –3.92 in the tocilizumab group and –1.22 in the placebo group, with an overall difference of –2.70 (95% confidence interval, –5.85-0.45; P = .0915). The effect was sustained at 48 weeks’ follow-up (–6.33 vs. –2.77; 95% CI, –7.2-0.12; P = .0579).
“This study is the first placebo-controlled trial in patients with early systemic sclerosis to show a clinically significant – albeit not statistically significant – amelioration of skin sclerosis, and clinically relevant improvement in lung function in patients treated with tocilizumab,” Dr. Dinesh Khanna, director of the University of Michigan Scleroderma Program, Ann Arbor, and his associates report (Lancet. 2016 May 5;387:2630-40).
Assessment of lung function was one of several exploratory analyses conducted and showed fewer tocilizumab- than placebo-treated patients had worsening of the percent predicted forced vital capacity at 48 weeks’ follow-up (P = .0373).
“We were very interested to see the potential impact of tocilizumab on lung function,” said study author Christopher P. Denton, Ph.D. of University College London at the recent British Society of Rheumatology Meeting in Glasgow. It perhaps signals “that IL-6 might be key in the pathogenesis of scleroderma lung fibrosis.”
FaSScinate was a double-blind, placebo-controlled phase II trial conducted at 35 hospitals in Canada, France, Germany, the United Kingdom, and the United States. A total of 87 adults were recruited, all had progressive systemic sclerosis for a duration of 5 years or less from the first appearance of a non–Raynaud’s sign or symptom. Tocilizumab and matched placebo were given subcutaneously at a dose of 162 mg/day.
“Given the lack of disease-modifying treatment options for patients with systemic sclerosis, combined with the morbidity and mortality associated with this disease, data from our trial provide hope for a potential future treatment,” Dr. Khanna and his associates observed in their article. “The safety profile was consistent with complications of systemic sclerosis, including osteomyelitis, and with the safety profile of tocilizumab,” they add.
The U.S. Food and Drug Administration gave tocilizumab a breakthrough therapy designation in systemic sclerosis following the preliminary release of the data in 2015. A phase III trial is now underway.
F Hoffmann-La Roche and Genentech funded the study. The authors disclosed financial relationships with multiple pharmaceutical companies, including the study sponsor.
The interleukin-6 inhibitor tocilizumab (Actemra) substantially reduced skin thickening in patients with systemic sclerosis in the “proof-of concept” faSScinate trial, the results of which have now been published in the Lancet.
The mean change in the primary endpoint of a modified Rodnan skin score at 24 weeks was –3.92 in the tocilizumab group and –1.22 in the placebo group, with an overall difference of –2.70 (95% confidence interval, –5.85-0.45; P = .0915). The effect was sustained at 48 weeks’ follow-up (–6.33 vs. –2.77; 95% CI, –7.2-0.12; P = .0579).
“This study is the first placebo-controlled trial in patients with early systemic sclerosis to show a clinically significant – albeit not statistically significant – amelioration of skin sclerosis, and clinically relevant improvement in lung function in patients treated with tocilizumab,” Dr. Dinesh Khanna, director of the University of Michigan Scleroderma Program, Ann Arbor, and his associates report (Lancet. 2016 May 5;387:2630-40).
Assessment of lung function was one of several exploratory analyses conducted and showed fewer tocilizumab- than placebo-treated patients had worsening of the percent predicted forced vital capacity at 48 weeks’ follow-up (P = .0373).
“We were very interested to see the potential impact of tocilizumab on lung function,” said study author Christopher P. Denton, Ph.D. of University College London at the recent British Society of Rheumatology Meeting in Glasgow. It perhaps signals “that IL-6 might be key in the pathogenesis of scleroderma lung fibrosis.”
FaSScinate was a double-blind, placebo-controlled phase II trial conducted at 35 hospitals in Canada, France, Germany, the United Kingdom, and the United States. A total of 87 adults were recruited, all had progressive systemic sclerosis for a duration of 5 years or less from the first appearance of a non–Raynaud’s sign or symptom. Tocilizumab and matched placebo were given subcutaneously at a dose of 162 mg/day.
“Given the lack of disease-modifying treatment options for patients with systemic sclerosis, combined with the morbidity and mortality associated with this disease, data from our trial provide hope for a potential future treatment,” Dr. Khanna and his associates observed in their article. “The safety profile was consistent with complications of systemic sclerosis, including osteomyelitis, and with the safety profile of tocilizumab,” they add.
The U.S. Food and Drug Administration gave tocilizumab a breakthrough therapy designation in systemic sclerosis following the preliminary release of the data in 2015. A phase III trial is now underway.
F Hoffmann-La Roche and Genentech funded the study. The authors disclosed financial relationships with multiple pharmaceutical companies, including the study sponsor.
The interleukin-6 inhibitor tocilizumab (Actemra) substantially reduced skin thickening in patients with systemic sclerosis in the “proof-of concept” faSScinate trial, the results of which have now been published in the Lancet.
The mean change in the primary endpoint of a modified Rodnan skin score at 24 weeks was –3.92 in the tocilizumab group and –1.22 in the placebo group, with an overall difference of –2.70 (95% confidence interval, –5.85-0.45; P = .0915). The effect was sustained at 48 weeks’ follow-up (–6.33 vs. –2.77; 95% CI, –7.2-0.12; P = .0579).
“This study is the first placebo-controlled trial in patients with early systemic sclerosis to show a clinically significant – albeit not statistically significant – amelioration of skin sclerosis, and clinically relevant improvement in lung function in patients treated with tocilizumab,” Dr. Dinesh Khanna, director of the University of Michigan Scleroderma Program, Ann Arbor, and his associates report (Lancet. 2016 May 5;387:2630-40).
Assessment of lung function was one of several exploratory analyses conducted and showed fewer tocilizumab- than placebo-treated patients had worsening of the percent predicted forced vital capacity at 48 weeks’ follow-up (P = .0373).
“We were very interested to see the potential impact of tocilizumab on lung function,” said study author Christopher P. Denton, Ph.D. of University College London at the recent British Society of Rheumatology Meeting in Glasgow. It perhaps signals “that IL-6 might be key in the pathogenesis of scleroderma lung fibrosis.”
FaSScinate was a double-blind, placebo-controlled phase II trial conducted at 35 hospitals in Canada, France, Germany, the United Kingdom, and the United States. A total of 87 adults were recruited, all had progressive systemic sclerosis for a duration of 5 years or less from the first appearance of a non–Raynaud’s sign or symptom. Tocilizumab and matched placebo were given subcutaneously at a dose of 162 mg/day.
“Given the lack of disease-modifying treatment options for patients with systemic sclerosis, combined with the morbidity and mortality associated with this disease, data from our trial provide hope for a potential future treatment,” Dr. Khanna and his associates observed in their article. “The safety profile was consistent with complications of systemic sclerosis, including osteomyelitis, and with the safety profile of tocilizumab,” they add.
The U.S. Food and Drug Administration gave tocilizumab a breakthrough therapy designation in systemic sclerosis following the preliminary release of the data in 2015. A phase III trial is now underway.
F Hoffmann-La Roche and Genentech funded the study. The authors disclosed financial relationships with multiple pharmaceutical companies, including the study sponsor.
FROM THE LANCET
Key clinical point: Tocilizumab reduced skin thickening and improved lung function in patients with systemic scleroderma.
Major finding: Mean change in a modified Rodnan skin score in the tocilizumab and placebo-treated patients at 24 weeks were a respective –3.92 and –1.22 (P = .0915).
Data source: Randomized, double blind, placebo-controlled multicenter phase II study of 87 adults with systemic sclerosis treated with tocilizumab 162 mg or placebo for 24 weeks.
Disclosures: F. Hoffmann-La Roche and Genentech funded the study. The authors disclosed financial relationships with multiple pharmaceutical companies, including the study sponsor.
July 2016: Click for Credit
Here are 4 articles in the July issue of Clinician Reviews (individual articles are valid for one year from date of publication—expiration dates below):
1. Pregnancy Alters Pharmacodynamics of Anti-TNF Agents in Women With IBD
To take the posttest, go to: http://bit.ly/1VQFIHf
Expires May 24, 2017
VITALS
Key clinical point: Blood levels of infliximab rose during pregnancy, while adalimumab levels remained stable, even after researchers accounted for changes in albumin, body mass index, and C-reactive protein levels.
Major finding: Median infliximab concentrations rose from 8.5 mcg/mL in the first trimester to a peak of 21 mcg/mL during the middle of the third trimester (P = .04). Median adalimumab levels ranged between 8.6 and 12.2 mcg/mL during pregnancy.
Data source: A prospective study of 25 pregnant women with ulcerative colitis or Crohn's disease.
Disclosures: Dr. Seow disclosed ties with Janssen, AbbVie, Takeda, Shire, and Actavis.
2. Vascular Disease Linked to Sight Loss in Giant Cell Arteritis
To take the posttest, go to: http://bit.ly/1UqLuu5
Expires May 10, 2017
VITALS
Key clinical point: Patients with vascular disease who develop giant cell arteritis may require careful monitoring for sight loss.
Major finding: Overall, 42.9% of patients had some visual disturbance at first clinic review; 7.9% were blind at 6 months.
Data source: Analysis of 433 patients newly diagnosed with GCA participating in the Diagnostic and Classification Criteria in Vasculitis Study (DCVAS).
Disclosures: The DCVAS study is supported by the American College of Rheumatology and is funded by the European League Against Rheumatism and the Vasculitis Foundation. Dr. Yates reported that he had no relevant disclosures.
3. Pediatric and Adolescent Mental Health
Part 1: Diagnoses, drug prescribing vary widely
To take the posttest, go to: http://bit.ly/24FHTxY
Expires April 1, 2017
VITALS
Key clinical point: A lack of psychiatrists only partially accounted for substantial variations in rates of mental illness diagnosis and prescriptions for psychotropic medications given in practices nationwide, a study has shown.
Major finding: Nationwide, 15% of pediatric patients received a mental health diagnosis, and 14% were prescribed psychotropic medications in primary care, regardless of colocated mental health services.
Data source: A retrospective study of electronic health records for 294,748 patients aged 4-18 years.
Disclosures: Dr. Alexander G. Fiks is an investigator for Pfizer; the other researchers said they had no relevant financial disclosures. This study was funded by the National Institutes of Health and the National Institute of Child Health and Human Development under the Best Pharmaceuticals for Children Act.
Part 2: Disorders prevalent in young transgender women
To take the posttest, go to: http://bit.ly/24FCDdq
Expires March 21, 2017
VITALS
Key clinical point: Young transgender women have a high prevalence of psychiatric disorders that is two to four times higher than that in the general population.
Major finding: 41.5% of the study participants had at least one psychiatric disorder, such as major depressive disorder, suicidality, generalized anxiety, PTSD, and alcohol or substance dependence.
Data source: An observational cohort study involving 298 transgender women aged 16-29 years residing in Chicago and Boston.
Disclosures: This study was supported by the National Institute of Mental Health. Dr. Reisner and his associates reported having no relevant financial disclosures.
Here are 4 articles in the July issue of Clinician Reviews (individual articles are valid for one year from date of publication—expiration dates below):
1. Pregnancy Alters Pharmacodynamics of Anti-TNF Agents in Women With IBD
To take the posttest, go to: http://bit.ly/1VQFIHf
Expires May 24, 2017
VITALS
Key clinical point: Blood levels of infliximab rose during pregnancy, while adalimumab levels remained stable, even after researchers accounted for changes in albumin, body mass index, and C-reactive protein levels.
Major finding: Median infliximab concentrations rose from 8.5 mcg/mL in the first trimester to a peak of 21 mcg/mL during the middle of the third trimester (P = .04). Median adalimumab levels ranged between 8.6 and 12.2 mcg/mL during pregnancy.
Data source: A prospective study of 25 pregnant women with ulcerative colitis or Crohn's disease.
Disclosures: Dr. Seow disclosed ties with Janssen, AbbVie, Takeda, Shire, and Actavis.
2. Vascular Disease Linked to Sight Loss in Giant Cell Arteritis
To take the posttest, go to: http://bit.ly/1UqLuu5
Expires May 10, 2017
VITALS
Key clinical point: Patients with vascular disease who develop giant cell arteritis may require careful monitoring for sight loss.
Major finding: Overall, 42.9% of patients had some visual disturbance at first clinic review; 7.9% were blind at 6 months.
Data source: Analysis of 433 patients newly diagnosed with GCA participating in the Diagnostic and Classification Criteria in Vasculitis Study (DCVAS).
Disclosures: The DCVAS study is supported by the American College of Rheumatology and is funded by the European League Against Rheumatism and the Vasculitis Foundation. Dr. Yates reported that he had no relevant disclosures.
3. Pediatric and Adolescent Mental Health
Part 1: Diagnoses, drug prescribing vary widely
To take the posttest, go to: http://bit.ly/24FHTxY
Expires April 1, 2017
VITALS
Key clinical point: A lack of psychiatrists only partially accounted for substantial variations in rates of mental illness diagnosis and prescriptions for psychotropic medications given in practices nationwide, a study has shown.
Major finding: Nationwide, 15% of pediatric patients received a mental health diagnosis, and 14% were prescribed psychotropic medications in primary care, regardless of colocated mental health services.
Data source: A retrospective study of electronic health records for 294,748 patients aged 4-18 years.
Disclosures: Dr. Alexander G. Fiks is an investigator for Pfizer; the other researchers said they had no relevant financial disclosures. This study was funded by the National Institutes of Health and the National Institute of Child Health and Human Development under the Best Pharmaceuticals for Children Act.
Part 2: Disorders prevalent in young transgender women
To take the posttest, go to: http://bit.ly/24FCDdq
Expires March 21, 2017
VITALS
Key clinical point: Young transgender women have a high prevalence of psychiatric disorders that is two to four times higher than that in the general population.
Major finding: 41.5% of the study participants had at least one psychiatric disorder, such as major depressive disorder, suicidality, generalized anxiety, PTSD, and alcohol or substance dependence.
Data source: An observational cohort study involving 298 transgender women aged 16-29 years residing in Chicago and Boston.
Disclosures: This study was supported by the National Institute of Mental Health. Dr. Reisner and his associates reported having no relevant financial disclosures.
Here are 4 articles in the July issue of Clinician Reviews (individual articles are valid for one year from date of publication—expiration dates below):
1. Pregnancy Alters Pharmacodynamics of Anti-TNF Agents in Women With IBD
To take the posttest, go to: http://bit.ly/1VQFIHf
Expires May 24, 2017
VITALS
Key clinical point: Blood levels of infliximab rose during pregnancy, while adalimumab levels remained stable, even after researchers accounted for changes in albumin, body mass index, and C-reactive protein levels.
Major finding: Median infliximab concentrations rose from 8.5 mcg/mL in the first trimester to a peak of 21 mcg/mL during the middle of the third trimester (P = .04). Median adalimumab levels ranged between 8.6 and 12.2 mcg/mL during pregnancy.
Data source: A prospective study of 25 pregnant women with ulcerative colitis or Crohn's disease.
Disclosures: Dr. Seow disclosed ties with Janssen, AbbVie, Takeda, Shire, and Actavis.
2. Vascular Disease Linked to Sight Loss in Giant Cell Arteritis
To take the posttest, go to: http://bit.ly/1UqLuu5
Expires May 10, 2017
VITALS
Key clinical point: Patients with vascular disease who develop giant cell arteritis may require careful monitoring for sight loss.
Major finding: Overall, 42.9% of patients had some visual disturbance at first clinic review; 7.9% were blind at 6 months.
Data source: Analysis of 433 patients newly diagnosed with GCA participating in the Diagnostic and Classification Criteria in Vasculitis Study (DCVAS).
Disclosures: The DCVAS study is supported by the American College of Rheumatology and is funded by the European League Against Rheumatism and the Vasculitis Foundation. Dr. Yates reported that he had no relevant disclosures.
3. Pediatric and Adolescent Mental Health
Part 1: Diagnoses, drug prescribing vary widely
To take the posttest, go to: http://bit.ly/24FHTxY
Expires April 1, 2017
VITALS
Key clinical point: A lack of psychiatrists only partially accounted for substantial variations in rates of mental illness diagnosis and prescriptions for psychotropic medications given in practices nationwide, a study has shown.
Major finding: Nationwide, 15% of pediatric patients received a mental health diagnosis, and 14% were prescribed psychotropic medications in primary care, regardless of colocated mental health services.
Data source: A retrospective study of electronic health records for 294,748 patients aged 4-18 years.
Disclosures: Dr. Alexander G. Fiks is an investigator for Pfizer; the other researchers said they had no relevant financial disclosures. This study was funded by the National Institutes of Health and the National Institute of Child Health and Human Development under the Best Pharmaceuticals for Children Act.
Part 2: Disorders prevalent in young transgender women
To take the posttest, go to: http://bit.ly/24FCDdq
Expires March 21, 2017
VITALS
Key clinical point: Young transgender women have a high prevalence of psychiatric disorders that is two to four times higher than that in the general population.
Major finding: 41.5% of the study participants had at least one psychiatric disorder, such as major depressive disorder, suicidality, generalized anxiety, PTSD, and alcohol or substance dependence.
Data source: An observational cohort study involving 298 transgender women aged 16-29 years residing in Chicago and Boston.
Disclosures: This study was supported by the National Institute of Mental Health. Dr. Reisner and his associates reported having no relevant financial disclosures.
Obesity May Attenuate Anti-TNF Response in Psoriatic Arthritis
LONDON – Patients with psoriatic arthritis appear less likely to achieve a good response to their first anti–tumor necrosis factor (anti-TNF) therapy if they are obese, according to data taken from two Nordic registries.
In a large observational cohort study, obese individuals with psoriatic arthritis (PsA) were significantly less likely than their nonobese counterparts to achieve a European League Against Rheumatism (EULAR) good or moderate response at 6 months (55% vs. 65%, P = .02). The overall odds ratio for achieving a good or moderate response was 0.47 when comparing obese with nonobese individuals.
The findings are potentially important because, with the exception of infliximab, anti-TNF therapy is not currently adjusted according to body weight, said presenting study author Pil Højgaard in an interview at the European Congress of Rheumatology.
Ms. Højgaard, who is an M.D. Ph.D. student at the department of rheumatology, Copenhagen University Hospital Gentofte, Rigshospitalet and the Parker Institute in Copenhagen, noted that obesity was a frequent comorbid condition in patients with PsA and that it is a known proinflammatory condition. As such, obesity could potentially affect immunologic processes, the pharmacokinetics of treatments, and ultimately patient outcomes.
Since TNF-alpha inhibitor (TNFi) treatment fails in around half of all patients with PsA treated in routine care, Ms. Højgaard noted that the aim of the cohort study was to investigate whether obesity could be having any influence on this.
Data on baseline characteristics, EULAR response rates, and drug adherence were obtained for 1,943 patients with PsA prescribed their anti-TNF therapy from two nationwide registries of disease-modifying therapies being used to treat rheumatic conditions in Denmark and Iceland, DANBIO (Rheumatology. 2011;50:69–77) and ICEBIO, respectively.
At baseline, body mass index (BMI) data were available for 1,271 patients and 408 (32%) of these had a BMI of 30 kg/m2 or more and were classed as being obese. The majority (39%) had received a first prescription for adalimumab, with around a quarter each prescribed etanercept (26%) or infliximab (24%), and the remainder prescribed golimumab (7%) and certolizumab (4%).
Compared to the 863 (68%) nonobese individuals, the obese patients were older (47 vs. 49 years, P = .01), less likely to smoke (30% vs. 23%, P = .01), and had higher disease activity measured on the Disease Activity Score 28 (DAS28) (4.4 vs. 4.6, P = .01). Health Assessment Questionnaire scores were also higher in obese than in nonobese individuals (1.1 vs. 0.9, P less than .01), and there were higher tender joint counts (6 vs. 5, P = .01), and higher pain levels assessed on a visual analog scale (VAS). Obese patients also had higher scores on a VAS patient global scale. The median follow-up time was 1.5 years.
Patients who were obese were found to adhere to TNFi treatment for shorter periods of time than nonobese patients, with median durations of 1.76 and 3.08 years, respectively (P less than .001). This discrepancy was most pronounced among men, a finding that may account for the fact that they were less likely to achieve a good EULAR response than their nonobese counterparts (OR = 0.5).
Being obese versus not being obese independently predicted TNFi withdrawal overall (hazard ratio, 1.6), especially in men (HR, 1.8; HR, 1.5 in women). TNFi withdrawal was more likely in obese than in nonobese patients even when individual treatments were considered; adalimumab: HR, 1.6; etanercept: HR, 2.0; infliximab: HR, 1.6.
An association between obesity and reduced response to anti-TNF therapy has also been observed in patients with rheumatoid arthritis, Ms. Højgaard acknowledged. There have also been a few studies of PsA and psoriasis “but to my knowledge, I think in the field of psoriatic arthritis, we are one of the few that have been looking at long-time drug survival,” she said. “We also include quite a lot of patients.”
“Of course this is not a randomized clinical study, so there could be residual confounding factors,” Ms. Højgaard cautioned. “It is always a bit difficult to say something about causality when it is a database study,” she added. “I think what we can see here is that there is an association, but in order to recommend weight loss we need some prospective studies.”
She noted that there was one published clinical study (Ann Rheum Dis. 2014;73:1157–62) that had looked at the benefit of a weight reduction program started at the same time as TNFi initiation in patients with PsA. This found there was a benefit of weight loss on response to TNFis, regardless of the type of diet.
DANBIO is supported by unrestricted grants from Abbott, Pfizer, MSD, Bristol-Myers Squibb, Roche, and UCB-Nordic. The sponsors have had no influence on data collection, analysis, or publication. ICEBIO is part of the electronic medical record system held by the University of Reykjavik and receives no industrial funding. Ms. Højgaard has received speaking fees from Celgene and UCB not related to this work.
LONDON – Patients with psoriatic arthritis appear less likely to achieve a good response to their first anti–tumor necrosis factor (anti-TNF) therapy if they are obese, according to data taken from two Nordic registries.
In a large observational cohort study, obese individuals with psoriatic arthritis (PsA) were significantly less likely than their nonobese counterparts to achieve a European League Against Rheumatism (EULAR) good or moderate response at 6 months (55% vs. 65%, P = .02). The overall odds ratio for achieving a good or moderate response was 0.47 when comparing obese with nonobese individuals.
The findings are potentially important because, with the exception of infliximab, anti-TNF therapy is not currently adjusted according to body weight, said presenting study author Pil Højgaard in an interview at the European Congress of Rheumatology.
Ms. Højgaard, who is an M.D. Ph.D. student at the department of rheumatology, Copenhagen University Hospital Gentofte, Rigshospitalet and the Parker Institute in Copenhagen, noted that obesity was a frequent comorbid condition in patients with PsA and that it is a known proinflammatory condition. As such, obesity could potentially affect immunologic processes, the pharmacokinetics of treatments, and ultimately patient outcomes.
Since TNF-alpha inhibitor (TNFi) treatment fails in around half of all patients with PsA treated in routine care, Ms. Højgaard noted that the aim of the cohort study was to investigate whether obesity could be having any influence on this.
Data on baseline characteristics, EULAR response rates, and drug adherence were obtained for 1,943 patients with PsA prescribed their anti-TNF therapy from two nationwide registries of disease-modifying therapies being used to treat rheumatic conditions in Denmark and Iceland, DANBIO (Rheumatology. 2011;50:69–77) and ICEBIO, respectively.
At baseline, body mass index (BMI) data were available for 1,271 patients and 408 (32%) of these had a BMI of 30 kg/m2 or more and were classed as being obese. The majority (39%) had received a first prescription for adalimumab, with around a quarter each prescribed etanercept (26%) or infliximab (24%), and the remainder prescribed golimumab (7%) and certolizumab (4%).
Compared to the 863 (68%) nonobese individuals, the obese patients were older (47 vs. 49 years, P = .01), less likely to smoke (30% vs. 23%, P = .01), and had higher disease activity measured on the Disease Activity Score 28 (DAS28) (4.4 vs. 4.6, P = .01). Health Assessment Questionnaire scores were also higher in obese than in nonobese individuals (1.1 vs. 0.9, P less than .01), and there were higher tender joint counts (6 vs. 5, P = .01), and higher pain levels assessed on a visual analog scale (VAS). Obese patients also had higher scores on a VAS patient global scale. The median follow-up time was 1.5 years.
Patients who were obese were found to adhere to TNFi treatment for shorter periods of time than nonobese patients, with median durations of 1.76 and 3.08 years, respectively (P less than .001). This discrepancy was most pronounced among men, a finding that may account for the fact that they were less likely to achieve a good EULAR response than their nonobese counterparts (OR = 0.5).
Being obese versus not being obese independently predicted TNFi withdrawal overall (hazard ratio, 1.6), especially in men (HR, 1.8; HR, 1.5 in women). TNFi withdrawal was more likely in obese than in nonobese patients even when individual treatments were considered; adalimumab: HR, 1.6; etanercept: HR, 2.0; infliximab: HR, 1.6.
An association between obesity and reduced response to anti-TNF therapy has also been observed in patients with rheumatoid arthritis, Ms. Højgaard acknowledged. There have also been a few studies of PsA and psoriasis “but to my knowledge, I think in the field of psoriatic arthritis, we are one of the few that have been looking at long-time drug survival,” she said. “We also include quite a lot of patients.”
“Of course this is not a randomized clinical study, so there could be residual confounding factors,” Ms. Højgaard cautioned. “It is always a bit difficult to say something about causality when it is a database study,” she added. “I think what we can see here is that there is an association, but in order to recommend weight loss we need some prospective studies.”
She noted that there was one published clinical study (Ann Rheum Dis. 2014;73:1157–62) that had looked at the benefit of a weight reduction program started at the same time as TNFi initiation in patients with PsA. This found there was a benefit of weight loss on response to TNFis, regardless of the type of diet.
DANBIO is supported by unrestricted grants from Abbott, Pfizer, MSD, Bristol-Myers Squibb, Roche, and UCB-Nordic. The sponsors have had no influence on data collection, analysis, or publication. ICEBIO is part of the electronic medical record system held by the University of Reykjavik and receives no industrial funding. Ms. Højgaard has received speaking fees from Celgene and UCB not related to this work.
LONDON – Patients with psoriatic arthritis appear less likely to achieve a good response to their first anti–tumor necrosis factor (anti-TNF) therapy if they are obese, according to data taken from two Nordic registries.
In a large observational cohort study, obese individuals with psoriatic arthritis (PsA) were significantly less likely than their nonobese counterparts to achieve a European League Against Rheumatism (EULAR) good or moderate response at 6 months (55% vs. 65%, P = .02). The overall odds ratio for achieving a good or moderate response was 0.47 when comparing obese with nonobese individuals.
The findings are potentially important because, with the exception of infliximab, anti-TNF therapy is not currently adjusted according to body weight, said presenting study author Pil Højgaard in an interview at the European Congress of Rheumatology.
Ms. Højgaard, who is an M.D. Ph.D. student at the department of rheumatology, Copenhagen University Hospital Gentofte, Rigshospitalet and the Parker Institute in Copenhagen, noted that obesity was a frequent comorbid condition in patients with PsA and that it is a known proinflammatory condition. As such, obesity could potentially affect immunologic processes, the pharmacokinetics of treatments, and ultimately patient outcomes.
Since TNF-alpha inhibitor (TNFi) treatment fails in around half of all patients with PsA treated in routine care, Ms. Højgaard noted that the aim of the cohort study was to investigate whether obesity could be having any influence on this.
Data on baseline characteristics, EULAR response rates, and drug adherence were obtained for 1,943 patients with PsA prescribed their anti-TNF therapy from two nationwide registries of disease-modifying therapies being used to treat rheumatic conditions in Denmark and Iceland, DANBIO (Rheumatology. 2011;50:69–77) and ICEBIO, respectively.
At baseline, body mass index (BMI) data were available for 1,271 patients and 408 (32%) of these had a BMI of 30 kg/m2 or more and were classed as being obese. The majority (39%) had received a first prescription for adalimumab, with around a quarter each prescribed etanercept (26%) or infliximab (24%), and the remainder prescribed golimumab (7%) and certolizumab (4%).
Compared to the 863 (68%) nonobese individuals, the obese patients were older (47 vs. 49 years, P = .01), less likely to smoke (30% vs. 23%, P = .01), and had higher disease activity measured on the Disease Activity Score 28 (DAS28) (4.4 vs. 4.6, P = .01). Health Assessment Questionnaire scores were also higher in obese than in nonobese individuals (1.1 vs. 0.9, P less than .01), and there were higher tender joint counts (6 vs. 5, P = .01), and higher pain levels assessed on a visual analog scale (VAS). Obese patients also had higher scores on a VAS patient global scale. The median follow-up time was 1.5 years.
Patients who were obese were found to adhere to TNFi treatment for shorter periods of time than nonobese patients, with median durations of 1.76 and 3.08 years, respectively (P less than .001). This discrepancy was most pronounced among men, a finding that may account for the fact that they were less likely to achieve a good EULAR response than their nonobese counterparts (OR = 0.5).
Being obese versus not being obese independently predicted TNFi withdrawal overall (hazard ratio, 1.6), especially in men (HR, 1.8; HR, 1.5 in women). TNFi withdrawal was more likely in obese than in nonobese patients even when individual treatments were considered; adalimumab: HR, 1.6; etanercept: HR, 2.0; infliximab: HR, 1.6.
An association between obesity and reduced response to anti-TNF therapy has also been observed in patients with rheumatoid arthritis, Ms. Højgaard acknowledged. There have also been a few studies of PsA and psoriasis “but to my knowledge, I think in the field of psoriatic arthritis, we are one of the few that have been looking at long-time drug survival,” she said. “We also include quite a lot of patients.”
“Of course this is not a randomized clinical study, so there could be residual confounding factors,” Ms. Højgaard cautioned. “It is always a bit difficult to say something about causality when it is a database study,” she added. “I think what we can see here is that there is an association, but in order to recommend weight loss we need some prospective studies.”
She noted that there was one published clinical study (Ann Rheum Dis. 2014;73:1157–62) that had looked at the benefit of a weight reduction program started at the same time as TNFi initiation in patients with PsA. This found there was a benefit of weight loss on response to TNFis, regardless of the type of diet.
DANBIO is supported by unrestricted grants from Abbott, Pfizer, MSD, Bristol-Myers Squibb, Roche, and UCB-Nordic. The sponsors have had no influence on data collection, analysis, or publication. ICEBIO is part of the electronic medical record system held by the University of Reykjavik and receives no industrial funding. Ms. Højgaard has received speaking fees from Celgene and UCB not related to this work.
Obesity may attenuate anti-TNF response in psoriatic arthritis
LONDON – Patients with psoriatic arthritis appear less likely to achieve a good response to their first anti–tumor necrosis factor (anti-TNF) therapy if they are obese, according to data taken from two Nordic registries.
In a large observational cohort study, obese individuals with psoriatic arthritis (PsA) were significantly less likely than their nonobese counterparts to achieve a European League Against Rheumatism (EULAR) good or moderate response at 6 months (55% vs. 65%, P = .02). The overall odds ratio for achieving a good or moderate response was 0.47 when comparing obese with nonobese individuals.
The findings are potentially important because, with the exception of infliximab, anti-TNF therapy is not currently adjusted according to body weight, said presenting study author Pil Højgaard in an interview at the European Congress of Rheumatology.
Ms. Højgaard, who is an M.D. Ph.D. student at the department of rheumatology, Copenhagen University Hospital Gentofte, Rigshospitalet and the Parker Institute in Copenhagen, noted that obesity was a frequent comorbid condition in patients with PsA and that it is a known proinflammatory condition. As such, obesity could potentially affect immunologic processes, the pharmacokinetics of treatments, and ultimately patient outcomes.
Since TNF-alpha inhibitor (TNFi) treatment fails in around half of all patients with PsA treated in routine care, Ms. Højgaard noted that the aim of the cohort study was to investigate whether obesity could be having any influence on this.
Data on baseline characteristics, EULAR response rates, and drug adherence were obtained for 1,943 patients with PsA prescribed their anti-TNF therapy from two nationwide registries of disease-modifying therapies being used to treat rheumatic conditions in Denmark and Iceland, DANBIO (Rheumatology. 2011;50:69–77) and ICEBIO, respectively.
At baseline, body mass index (BMI) data were available for 1,271 patients and 408 (32%) of these had a BMI of 30 kg/m2 or more and were classed as being obese. The majority (39%) had received a first prescription for adalimumab, with around a quarter each prescribed etanercept (26%) or infliximab (24%), and the remainder prescribed golimumab (7%) and certolizumab (4%).
Compared to the 863 (68%) nonobese individuals, the obese patients were older (47 vs. 49 years, P = .01), less likely to smoke (30% vs. 23%, P = .01), and had higher disease activity measured on the Disease Activity Score 28 (DAS28) (4.4 vs. 4.6, P = .01). Health Assessment Questionnaire scores were also higher in obese than in nonobese individuals (1.1 vs. 0.9, P less than .01), and there were higher tender joint counts (6 vs. 5, P = .01), and higher pain levels assessed on a visual analog scale (VAS). Obese patients also had higher scores on a VAS patient global scale. The median follow-up time was 1.5 years.
Patients who were obese were found to adhere to TNFi treatment for shorter periods of time than nonobese patients, with median durations of 1.76 and 3.08 years, respectively (P less than .001). This discrepancy was most pronounced among men, a finding that may account for the fact that they were less likely to achieve a good EULAR response than their nonobese counterparts (OR = 0.5).
Being obese versus not being obese independently predicted TNFi withdrawal overall (hazard ratio, 1.6), especially in men (HR, 1.8; HR, 1.5 in women). TNFi withdrawal was more likely in obese than in nonobese patients even when individual treatments were considered; adalimumab: HR, 1.6; etanercept: HR, 2.0; infliximab: HR, 1.6.
An association between obesity and reduced response to anti-TNF therapy has also been observed in patients with rheumatoid arthritis, Ms. Højgaard acknowledged. There have also been a few studies of PsA and psoriasis “but to my knowledge, I think in the field of psoriatic arthritis, we are one of the few that have been looking at long-time drug survival,” she said. “We also include quite a lot of patients.”
“Of course this is not a randomized clinical study, so there could be residual confounding factors,” Ms. Højgaard cautioned. “It is always a bit difficult to say something about causality when it is a database study,” she added. “I think what we can see here is that there is an association, but in order to recommend weight loss we need some prospective studies.”
She noted that there was one published clinical study (Ann Rheum Dis. 2014;73:1157–62) that had looked at the benefit of a weight reduction program started at the same time as TNFi initiation in patients with PsA. This found there was a benefit of weight loss on response to TNFis, regardless of the type of diet.
DANBIO is supported by unrestricted grants from Abbott, Pfizer, MSD, Bristol-Myers Squibb, Roche, and UCB-Nordic. The sponsors have had no influence on data collection, analysis, or publication. ICEBIO is part of the electronic medical record system held by the University of Reykjavik and receives no industrial funding. Ms. Højgaard has received speaking fees from Celgene and UCB not related to this work.
LONDON – Patients with psoriatic arthritis appear less likely to achieve a good response to their first anti–tumor necrosis factor (anti-TNF) therapy if they are obese, according to data taken from two Nordic registries.
In a large observational cohort study, obese individuals with psoriatic arthritis (PsA) were significantly less likely than their nonobese counterparts to achieve a European League Against Rheumatism (EULAR) good or moderate response at 6 months (55% vs. 65%, P = .02). The overall odds ratio for achieving a good or moderate response was 0.47 when comparing obese with nonobese individuals.
The findings are potentially important because, with the exception of infliximab, anti-TNF therapy is not currently adjusted according to body weight, said presenting study author Pil Højgaard in an interview at the European Congress of Rheumatology.
Ms. Højgaard, who is an M.D. Ph.D. student at the department of rheumatology, Copenhagen University Hospital Gentofte, Rigshospitalet and the Parker Institute in Copenhagen, noted that obesity was a frequent comorbid condition in patients with PsA and that it is a known proinflammatory condition. As such, obesity could potentially affect immunologic processes, the pharmacokinetics of treatments, and ultimately patient outcomes.
Since TNF-alpha inhibitor (TNFi) treatment fails in around half of all patients with PsA treated in routine care, Ms. Højgaard noted that the aim of the cohort study was to investigate whether obesity could be having any influence on this.
Data on baseline characteristics, EULAR response rates, and drug adherence were obtained for 1,943 patients with PsA prescribed their anti-TNF therapy from two nationwide registries of disease-modifying therapies being used to treat rheumatic conditions in Denmark and Iceland, DANBIO (Rheumatology. 2011;50:69–77) and ICEBIO, respectively.
At baseline, body mass index (BMI) data were available for 1,271 patients and 408 (32%) of these had a BMI of 30 kg/m2 or more and were classed as being obese. The majority (39%) had received a first prescription for adalimumab, with around a quarter each prescribed etanercept (26%) or infliximab (24%), and the remainder prescribed golimumab (7%) and certolizumab (4%).
Compared to the 863 (68%) nonobese individuals, the obese patients were older (47 vs. 49 years, P = .01), less likely to smoke (30% vs. 23%, P = .01), and had higher disease activity measured on the Disease Activity Score 28 (DAS28) (4.4 vs. 4.6, P = .01). Health Assessment Questionnaire scores were also higher in obese than in nonobese individuals (1.1 vs. 0.9, P less than .01), and there were higher tender joint counts (6 vs. 5, P = .01), and higher pain levels assessed on a visual analog scale (VAS). Obese patients also had higher scores on a VAS patient global scale. The median follow-up time was 1.5 years.
Patients who were obese were found to adhere to TNFi treatment for shorter periods of time than nonobese patients, with median durations of 1.76 and 3.08 years, respectively (P less than .001). This discrepancy was most pronounced among men, a finding that may account for the fact that they were less likely to achieve a good EULAR response than their nonobese counterparts (OR = 0.5).
Being obese versus not being obese independently predicted TNFi withdrawal overall (hazard ratio, 1.6), especially in men (HR, 1.8; HR, 1.5 in women). TNFi withdrawal was more likely in obese than in nonobese patients even when individual treatments were considered; adalimumab: HR, 1.6; etanercept: HR, 2.0; infliximab: HR, 1.6.
An association between obesity and reduced response to anti-TNF therapy has also been observed in patients with rheumatoid arthritis, Ms. Højgaard acknowledged. There have also been a few studies of PsA and psoriasis “but to my knowledge, I think in the field of psoriatic arthritis, we are one of the few that have been looking at long-time drug survival,” she said. “We also include quite a lot of patients.”
“Of course this is not a randomized clinical study, so there could be residual confounding factors,” Ms. Højgaard cautioned. “It is always a bit difficult to say something about causality when it is a database study,” she added. “I think what we can see here is that there is an association, but in order to recommend weight loss we need some prospective studies.”
She noted that there was one published clinical study (Ann Rheum Dis. 2014;73:1157–62) that had looked at the benefit of a weight reduction program started at the same time as TNFi initiation in patients with PsA. This found there was a benefit of weight loss on response to TNFis, regardless of the type of diet.
DANBIO is supported by unrestricted grants from Abbott, Pfizer, MSD, Bristol-Myers Squibb, Roche, and UCB-Nordic. The sponsors have had no influence on data collection, analysis, or publication. ICEBIO is part of the electronic medical record system held by the University of Reykjavik and receives no industrial funding. Ms. Højgaard has received speaking fees from Celgene and UCB not related to this work.
LONDON – Patients with psoriatic arthritis appear less likely to achieve a good response to their first anti–tumor necrosis factor (anti-TNF) therapy if they are obese, according to data taken from two Nordic registries.
In a large observational cohort study, obese individuals with psoriatic arthritis (PsA) were significantly less likely than their nonobese counterparts to achieve a European League Against Rheumatism (EULAR) good or moderate response at 6 months (55% vs. 65%, P = .02). The overall odds ratio for achieving a good or moderate response was 0.47 when comparing obese with nonobese individuals.
The findings are potentially important because, with the exception of infliximab, anti-TNF therapy is not currently adjusted according to body weight, said presenting study author Pil Højgaard in an interview at the European Congress of Rheumatology.
Ms. Højgaard, who is an M.D. Ph.D. student at the department of rheumatology, Copenhagen University Hospital Gentofte, Rigshospitalet and the Parker Institute in Copenhagen, noted that obesity was a frequent comorbid condition in patients with PsA and that it is a known proinflammatory condition. As such, obesity could potentially affect immunologic processes, the pharmacokinetics of treatments, and ultimately patient outcomes.
Since TNF-alpha inhibitor (TNFi) treatment fails in around half of all patients with PsA treated in routine care, Ms. Højgaard noted that the aim of the cohort study was to investigate whether obesity could be having any influence on this.
Data on baseline characteristics, EULAR response rates, and drug adherence were obtained for 1,943 patients with PsA prescribed their anti-TNF therapy from two nationwide registries of disease-modifying therapies being used to treat rheumatic conditions in Denmark and Iceland, DANBIO (Rheumatology. 2011;50:69–77) and ICEBIO, respectively.
At baseline, body mass index (BMI) data were available for 1,271 patients and 408 (32%) of these had a BMI of 30 kg/m2 or more and were classed as being obese. The majority (39%) had received a first prescription for adalimumab, with around a quarter each prescribed etanercept (26%) or infliximab (24%), and the remainder prescribed golimumab (7%) and certolizumab (4%).
Compared to the 863 (68%) nonobese individuals, the obese patients were older (47 vs. 49 years, P = .01), less likely to smoke (30% vs. 23%, P = .01), and had higher disease activity measured on the Disease Activity Score 28 (DAS28) (4.4 vs. 4.6, P = .01). Health Assessment Questionnaire scores were also higher in obese than in nonobese individuals (1.1 vs. 0.9, P less than .01), and there were higher tender joint counts (6 vs. 5, P = .01), and higher pain levels assessed on a visual analog scale (VAS). Obese patients also had higher scores on a VAS patient global scale. The median follow-up time was 1.5 years.
Patients who were obese were found to adhere to TNFi treatment for shorter periods of time than nonobese patients, with median durations of 1.76 and 3.08 years, respectively (P less than .001). This discrepancy was most pronounced among men, a finding that may account for the fact that they were less likely to achieve a good EULAR response than their nonobese counterparts (OR = 0.5).
Being obese versus not being obese independently predicted TNFi withdrawal overall (hazard ratio, 1.6), especially in men (HR, 1.8; HR, 1.5 in women). TNFi withdrawal was more likely in obese than in nonobese patients even when individual treatments were considered; adalimumab: HR, 1.6; etanercept: HR, 2.0; infliximab: HR, 1.6.
An association between obesity and reduced response to anti-TNF therapy has also been observed in patients with rheumatoid arthritis, Ms. Højgaard acknowledged. There have also been a few studies of PsA and psoriasis “but to my knowledge, I think in the field of psoriatic arthritis, we are one of the few that have been looking at long-time drug survival,” she said. “We also include quite a lot of patients.”
“Of course this is not a randomized clinical study, so there could be residual confounding factors,” Ms. Højgaard cautioned. “It is always a bit difficult to say something about causality when it is a database study,” she added. “I think what we can see here is that there is an association, but in order to recommend weight loss we need some prospective studies.”
She noted that there was one published clinical study (Ann Rheum Dis. 2014;73:1157–62) that had looked at the benefit of a weight reduction program started at the same time as TNFi initiation in patients with PsA. This found there was a benefit of weight loss on response to TNFis, regardless of the type of diet.
DANBIO is supported by unrestricted grants from Abbott, Pfizer, MSD, Bristol-Myers Squibb, Roche, and UCB-Nordic. The sponsors have had no influence on data collection, analysis, or publication. ICEBIO is part of the electronic medical record system held by the University of Reykjavik and receives no industrial funding. Ms. Højgaard has received speaking fees from Celgene and UCB not related to this work.
Key clinical point: Obesity may reduce response and adherence to tumor necrosis factor–inhibitor therapy in patients with psoriatic arthritis.
Major finding: The EULAR good or moderate response rate at 6 months was 55% in obese vs. 65% in nonobese patients (P = .02).
Data source: Observational cohort study based on two Nordic registries of 1,943 patients with PsA prescribed their first TNFi.
Disclosures: DANBIO is supported by unrestricted grants from Abbott, Pfizer, MSD, Bristol-Myers Squibb, Roche, and UCB-Nordic. The sponsors have had no influence on data collection, analysis, or publication. ICEBIO is part of the electronic medical record system held by the University of Reykjavik and receives no industrial funding. Ms. Højgaard has received speaking fees from Celgene and UCB not related to this work.
European ANCA-associated vasculitis guidance gets first makeover since 2009
LONDON – Updated management recommendations for patients with antineutrophil cytoplasmic antibody (ANCA)–associated vasculitis from the European League Against Rheumatism and the European Renal Association-European Dialysis and Transplant Association aim to provide clinicians with reliable guidance on the best approach to treatment.
The update, presented at the European Congress of Rheumatology and recently published online in Annals of the Rheumatic Diseases (Ann Rheum Dis. 2016 Jun 23. doi:10.1136/annrheumdis-2016-209133), reassessed items in the 2009 recommendations for the management of primary systemic vasculitis and focused only on the management of ANCA-associated vasculitis (AAV), according to recommendations task force member Dr. Max Yates.
“In the past 5 years, 1,691 papers have been published on primary systemic vasculitis in internal medicine, rheumatology, and nephrology journals. Together with the licensing of rituximab for AAV, it was an opportune time to update the recommendations with an AAV focus,” Dr. Yates explained. The revised guidance is based on a systematic literature review from January 2007 to February 2015, focusing in particular on specific items that needed updating, such as the importance of ANCA testing and biopsy in diagnosis and follow-up, disease staging at diagnosis, the choices for remission-induction and remission-maintenance therapies, and the drug choices for relapsing and refractory disease. The task force considered for the first time the choice of immunosuppressive drugs and biologic agents (principally rituximab) and immunologic monitoring. They identified patient education as another priority.
“These updated recommendations provide a framework of practice and should apply to the majority of patients with AAV,” added Dr. Yates, who is a clinical fellow at Norwich Medical School at the University of East Anglia and works in the department of rheumatology at the Norfolk and Norwich (England) University Hospital.
The 22-member task force included rheumatologists, internists, nephrologists, a clinical immunologist, an otorhinolaryngologist, a chest physician, an ophthalmologist, a vasculitis nurse, and a patient with vasculitis from 11 countries in Europe and the United States. The task force was convened by rheumatologist Dr. Chetan Mukhtyar of the Norfolk and Norwich University Hospital on behalf of EULAR and by vasculitis and renal specialist Dr. David Jayne of Addenbrooke’s Hospital in Cambridge (England) on behalf of the European Renal Association-European Dialysis and Transplant Association.
The recommendations now contain one single, simple overarching principle, Dr. Mukhtyar said at the congress. That is, the need for shared decision making between the patient and the clinician. This principle is also included as the first point in many of the other recently updated EULAR recommendations on the management of rheumatic diseases.
Both previous and updated versions of the vasculitis recommendations contain 15 recommendations, with some changed and others combined. One key recommendation is about who should treat patients with AAV; it states that patients “should be managed in close collaboration with, or at, centers of expertise,” Dr. Mukhtyar said.“Patients with ANCA-associated vasculitis have often very complex presentations that involve several different specialties, and it is always worthwhile that these patients are looked after by people who commonly see them, because these are rare conditions,” he observed.
Deciding when to perform a biopsy is also covered, with the recommendation being that it can be used to establish a new diagnosis and to further evaluate cases of suspected relapsing vasculitis. “When do you do a biopsy?” Dr. Mukhtyar asked. “Well, every time you can, every time it is clinically feasible,” he suggested.
As for treatment, there are different recommendations depending on whether the aim is to induce or maintain remission and whether there has been a major relapse. In patients with organ- or life-threatening disease, for example, the advice is to use glucocorticoids and either cyclophosphamide or rituximab to induce remission, Dr. Mukhtyar said. The specific dosing or administration of glucocorticoids is not specified as this will depend on the clinical situation, but the advice is to taper down when possible, somewhere between month 3 and 5.
For remission induction in less severe (non–organ threatening) disease, the recommendation is to use glucocorticoids plus either methotrexate or mycophenolate mofetil. Situations when methotrexate or mycophenolate mofetil should and should not be used are specified, notably when cyclophosphamide or rituximab are not available or are contraindicated.
For maintenance of remission, the task force advised using low-dose glucocorticoids plus azathioprine, rituximab, methotrexate, or mycophenolate mofetil.
Guidance on when to use plasma exchange is given for patients with severe disease and options following failure of remission-induction therapy, and when to switch therapy is also covered.
There are also several follow-up recommendations, such as the periodic assessment of cardiovascular risk, and patient-focused recommendations on awareness of the nature, benefits, and risks of therapy.
The recommendations should provide clinicians with reliable guidance on the best approach to treating AAV, according to Dr. Yates. “From the patients’ point of view, these recommendations should provide useful insight into which treatments they are likely to be offered and when. They also emphasize that as a patient, you should have a voice in your treatment and if you have any questions or concerns, be sure to speak with your specialist.”
Dr. Yates and Dr. Mukhtyar did not report having any relevant disclosures.
The prior 2009 EULAR recommendations were very much in need of updating given the plethora of studies in the past 7 years addressing ANCA-associated vasculitis (AAV). The emergence of rituximab as an effective therapy in AAV had to be considered and included in these newer guidelines. Its potential role in both remission induction, as well as remission maintenance of AAV, is addressed.
The recommendations are somewhat complicated, particularly as eosinophilic granulomatosis with polyangiitis (EGPA, previously referred to as Churg-Strauss syndrome) has been included, but most of the well-done prospective clinical trials addressing remission induction and remission maintenance in AAV were limited to patients with granulomatosis with polyangiitis or microscopic polyangiitis and did not include patients with EGPA. The role of plasma exchange is also discussed, but the results of the PEXIVAS trial, which will address that more definitively, are not yet forthcoming. Those results are anticipated in the not too distant future and will much better define that component of management in those most severely ill patients with AAV.

|
| Dr. Robert Spiera |
These recommendations serve as a framework for helping clinicians understand what is widely accepted as standard of care for these diseases but in no way can define individual treatment decisions as the authors acknowledge. Such decisions must become very personalized in relation to details of the patient’s individual comorbidities and other features of their medical and even socioeconomic status. For example, when choosing between rituximab and cyclophosphamide for remission induction in a young woman (or man, for that matter), future fertility concerns (which cyclophosphamide could potentially compromise) are very relevant. Moreover, the costs of rituximab are substantial, and the lack of superiority of rituximab over cyclophosphamide in many situations, particularly in patients with new severe disease, could be an important factor to consider when choosing which immunosuppressive will be used.
Many of the unanswered questions await results of ongoing or upcoming trials, including some addressing the relative efficacy of various remission maintenance regimens (rituximab vs. azathioprine) or the role of plasmapheresis. Many questions in AAV are not easily addressable in clinical trials, such as whether there are some groups of patients in whom remission maintenance therapy should never be withdrawn. However, such questions may be addressed through observational studies of the well-defined patient cohorts and registries that have been developed in the United States and Europe.
Robert F. Spiera, MD, is director of the Scleroderma, Vasculitis, & Myositis Center at the Hospital for Special Surgery, N.Y. He is also professor of clinical medicine at Cornell University, N.Y. He has received research funding and consulting fees from Roche/Genentech, which markets rituximab.
The prior 2009 EULAR recommendations were very much in need of updating given the plethora of studies in the past 7 years addressing ANCA-associated vasculitis (AAV). The emergence of rituximab as an effective therapy in AAV had to be considered and included in these newer guidelines. Its potential role in both remission induction, as well as remission maintenance of AAV, is addressed.
The recommendations are somewhat complicated, particularly as eosinophilic granulomatosis with polyangiitis (EGPA, previously referred to as Churg-Strauss syndrome) has been included, but most of the well-done prospective clinical trials addressing remission induction and remission maintenance in AAV were limited to patients with granulomatosis with polyangiitis or microscopic polyangiitis and did not include patients with EGPA. The role of plasma exchange is also discussed, but the results of the PEXIVAS trial, which will address that more definitively, are not yet forthcoming. Those results are anticipated in the not too distant future and will much better define that component of management in those most severely ill patients with AAV.

|
| Dr. Robert Spiera |
These recommendations serve as a framework for helping clinicians understand what is widely accepted as standard of care for these diseases but in no way can define individual treatment decisions as the authors acknowledge. Such decisions must become very personalized in relation to details of the patient’s individual comorbidities and other features of their medical and even socioeconomic status. For example, when choosing between rituximab and cyclophosphamide for remission induction in a young woman (or man, for that matter), future fertility concerns (which cyclophosphamide could potentially compromise) are very relevant. Moreover, the costs of rituximab are substantial, and the lack of superiority of rituximab over cyclophosphamide in many situations, particularly in patients with new severe disease, could be an important factor to consider when choosing which immunosuppressive will be used.
Many of the unanswered questions await results of ongoing or upcoming trials, including some addressing the relative efficacy of various remission maintenance regimens (rituximab vs. azathioprine) or the role of plasmapheresis. Many questions in AAV are not easily addressable in clinical trials, such as whether there are some groups of patients in whom remission maintenance therapy should never be withdrawn. However, such questions may be addressed through observational studies of the well-defined patient cohorts and registries that have been developed in the United States and Europe.
Robert F. Spiera, MD, is director of the Scleroderma, Vasculitis, & Myositis Center at the Hospital for Special Surgery, N.Y. He is also professor of clinical medicine at Cornell University, N.Y. He has received research funding and consulting fees from Roche/Genentech, which markets rituximab.
The prior 2009 EULAR recommendations were very much in need of updating given the plethora of studies in the past 7 years addressing ANCA-associated vasculitis (AAV). The emergence of rituximab as an effective therapy in AAV had to be considered and included in these newer guidelines. Its potential role in both remission induction, as well as remission maintenance of AAV, is addressed.
The recommendations are somewhat complicated, particularly as eosinophilic granulomatosis with polyangiitis (EGPA, previously referred to as Churg-Strauss syndrome) has been included, but most of the well-done prospective clinical trials addressing remission induction and remission maintenance in AAV were limited to patients with granulomatosis with polyangiitis or microscopic polyangiitis and did not include patients with EGPA. The role of plasma exchange is also discussed, but the results of the PEXIVAS trial, which will address that more definitively, are not yet forthcoming. Those results are anticipated in the not too distant future and will much better define that component of management in those most severely ill patients with AAV.

|
| Dr. Robert Spiera |
These recommendations serve as a framework for helping clinicians understand what is widely accepted as standard of care for these diseases but in no way can define individual treatment decisions as the authors acknowledge. Such decisions must become very personalized in relation to details of the patient’s individual comorbidities and other features of their medical and even socioeconomic status. For example, when choosing between rituximab and cyclophosphamide for remission induction in a young woman (or man, for that matter), future fertility concerns (which cyclophosphamide could potentially compromise) are very relevant. Moreover, the costs of rituximab are substantial, and the lack of superiority of rituximab over cyclophosphamide in many situations, particularly in patients with new severe disease, could be an important factor to consider when choosing which immunosuppressive will be used.
Many of the unanswered questions await results of ongoing or upcoming trials, including some addressing the relative efficacy of various remission maintenance regimens (rituximab vs. azathioprine) or the role of plasmapheresis. Many questions in AAV are not easily addressable in clinical trials, such as whether there are some groups of patients in whom remission maintenance therapy should never be withdrawn. However, such questions may be addressed through observational studies of the well-defined patient cohorts and registries that have been developed in the United States and Europe.
Robert F. Spiera, MD, is director of the Scleroderma, Vasculitis, & Myositis Center at the Hospital for Special Surgery, N.Y. He is also professor of clinical medicine at Cornell University, N.Y. He has received research funding and consulting fees from Roche/Genentech, which markets rituximab.
LONDON – Updated management recommendations for patients with antineutrophil cytoplasmic antibody (ANCA)–associated vasculitis from the European League Against Rheumatism and the European Renal Association-European Dialysis and Transplant Association aim to provide clinicians with reliable guidance on the best approach to treatment.
The update, presented at the European Congress of Rheumatology and recently published online in Annals of the Rheumatic Diseases (Ann Rheum Dis. 2016 Jun 23. doi:10.1136/annrheumdis-2016-209133), reassessed items in the 2009 recommendations for the management of primary systemic vasculitis and focused only on the management of ANCA-associated vasculitis (AAV), according to recommendations task force member Dr. Max Yates.
“In the past 5 years, 1,691 papers have been published on primary systemic vasculitis in internal medicine, rheumatology, and nephrology journals. Together with the licensing of rituximab for AAV, it was an opportune time to update the recommendations with an AAV focus,” Dr. Yates explained. The revised guidance is based on a systematic literature review from January 2007 to February 2015, focusing in particular on specific items that needed updating, such as the importance of ANCA testing and biopsy in diagnosis and follow-up, disease staging at diagnosis, the choices for remission-induction and remission-maintenance therapies, and the drug choices for relapsing and refractory disease. The task force considered for the first time the choice of immunosuppressive drugs and biologic agents (principally rituximab) and immunologic monitoring. They identified patient education as another priority.
“These updated recommendations provide a framework of practice and should apply to the majority of patients with AAV,” added Dr. Yates, who is a clinical fellow at Norwich Medical School at the University of East Anglia and works in the department of rheumatology at the Norfolk and Norwich (England) University Hospital.
The 22-member task force included rheumatologists, internists, nephrologists, a clinical immunologist, an otorhinolaryngologist, a chest physician, an ophthalmologist, a vasculitis nurse, and a patient with vasculitis from 11 countries in Europe and the United States. The task force was convened by rheumatologist Dr. Chetan Mukhtyar of the Norfolk and Norwich University Hospital on behalf of EULAR and by vasculitis and renal specialist Dr. David Jayne of Addenbrooke’s Hospital in Cambridge (England) on behalf of the European Renal Association-European Dialysis and Transplant Association.
The recommendations now contain one single, simple overarching principle, Dr. Mukhtyar said at the congress. That is, the need for shared decision making between the patient and the clinician. This principle is also included as the first point in many of the other recently updated EULAR recommendations on the management of rheumatic diseases.
Both previous and updated versions of the vasculitis recommendations contain 15 recommendations, with some changed and others combined. One key recommendation is about who should treat patients with AAV; it states that patients “should be managed in close collaboration with, or at, centers of expertise,” Dr. Mukhtyar said.“Patients with ANCA-associated vasculitis have often very complex presentations that involve several different specialties, and it is always worthwhile that these patients are looked after by people who commonly see them, because these are rare conditions,” he observed.
Deciding when to perform a biopsy is also covered, with the recommendation being that it can be used to establish a new diagnosis and to further evaluate cases of suspected relapsing vasculitis. “When do you do a biopsy?” Dr. Mukhtyar asked. “Well, every time you can, every time it is clinically feasible,” he suggested.
As for treatment, there are different recommendations depending on whether the aim is to induce or maintain remission and whether there has been a major relapse. In patients with organ- or life-threatening disease, for example, the advice is to use glucocorticoids and either cyclophosphamide or rituximab to induce remission, Dr. Mukhtyar said. The specific dosing or administration of glucocorticoids is not specified as this will depend on the clinical situation, but the advice is to taper down when possible, somewhere between month 3 and 5.
For remission induction in less severe (non–organ threatening) disease, the recommendation is to use glucocorticoids plus either methotrexate or mycophenolate mofetil. Situations when methotrexate or mycophenolate mofetil should and should not be used are specified, notably when cyclophosphamide or rituximab are not available or are contraindicated.
For maintenance of remission, the task force advised using low-dose glucocorticoids plus azathioprine, rituximab, methotrexate, or mycophenolate mofetil.
Guidance on when to use plasma exchange is given for patients with severe disease and options following failure of remission-induction therapy, and when to switch therapy is also covered.
There are also several follow-up recommendations, such as the periodic assessment of cardiovascular risk, and patient-focused recommendations on awareness of the nature, benefits, and risks of therapy.
The recommendations should provide clinicians with reliable guidance on the best approach to treating AAV, according to Dr. Yates. “From the patients’ point of view, these recommendations should provide useful insight into which treatments they are likely to be offered and when. They also emphasize that as a patient, you should have a voice in your treatment and if you have any questions or concerns, be sure to speak with your specialist.”
Dr. Yates and Dr. Mukhtyar did not report having any relevant disclosures.
LONDON – Updated management recommendations for patients with antineutrophil cytoplasmic antibody (ANCA)–associated vasculitis from the European League Against Rheumatism and the European Renal Association-European Dialysis and Transplant Association aim to provide clinicians with reliable guidance on the best approach to treatment.
The update, presented at the European Congress of Rheumatology and recently published online in Annals of the Rheumatic Diseases (Ann Rheum Dis. 2016 Jun 23. doi:10.1136/annrheumdis-2016-209133), reassessed items in the 2009 recommendations for the management of primary systemic vasculitis and focused only on the management of ANCA-associated vasculitis (AAV), according to recommendations task force member Dr. Max Yates.
“In the past 5 years, 1,691 papers have been published on primary systemic vasculitis in internal medicine, rheumatology, and nephrology journals. Together with the licensing of rituximab for AAV, it was an opportune time to update the recommendations with an AAV focus,” Dr. Yates explained. The revised guidance is based on a systematic literature review from January 2007 to February 2015, focusing in particular on specific items that needed updating, such as the importance of ANCA testing and biopsy in diagnosis and follow-up, disease staging at diagnosis, the choices for remission-induction and remission-maintenance therapies, and the drug choices for relapsing and refractory disease. The task force considered for the first time the choice of immunosuppressive drugs and biologic agents (principally rituximab) and immunologic monitoring. They identified patient education as another priority.
“These updated recommendations provide a framework of practice and should apply to the majority of patients with AAV,” added Dr. Yates, who is a clinical fellow at Norwich Medical School at the University of East Anglia and works in the department of rheumatology at the Norfolk and Norwich (England) University Hospital.
The 22-member task force included rheumatologists, internists, nephrologists, a clinical immunologist, an otorhinolaryngologist, a chest physician, an ophthalmologist, a vasculitis nurse, and a patient with vasculitis from 11 countries in Europe and the United States. The task force was convened by rheumatologist Dr. Chetan Mukhtyar of the Norfolk and Norwich University Hospital on behalf of EULAR and by vasculitis and renal specialist Dr. David Jayne of Addenbrooke’s Hospital in Cambridge (England) on behalf of the European Renal Association-European Dialysis and Transplant Association.
The recommendations now contain one single, simple overarching principle, Dr. Mukhtyar said at the congress. That is, the need for shared decision making between the patient and the clinician. This principle is also included as the first point in many of the other recently updated EULAR recommendations on the management of rheumatic diseases.
Both previous and updated versions of the vasculitis recommendations contain 15 recommendations, with some changed and others combined. One key recommendation is about who should treat patients with AAV; it states that patients “should be managed in close collaboration with, or at, centers of expertise,” Dr. Mukhtyar said.“Patients with ANCA-associated vasculitis have often very complex presentations that involve several different specialties, and it is always worthwhile that these patients are looked after by people who commonly see them, because these are rare conditions,” he observed.
Deciding when to perform a biopsy is also covered, with the recommendation being that it can be used to establish a new diagnosis and to further evaluate cases of suspected relapsing vasculitis. “When do you do a biopsy?” Dr. Mukhtyar asked. “Well, every time you can, every time it is clinically feasible,” he suggested.
As for treatment, there are different recommendations depending on whether the aim is to induce or maintain remission and whether there has been a major relapse. In patients with organ- or life-threatening disease, for example, the advice is to use glucocorticoids and either cyclophosphamide or rituximab to induce remission, Dr. Mukhtyar said. The specific dosing or administration of glucocorticoids is not specified as this will depend on the clinical situation, but the advice is to taper down when possible, somewhere between month 3 and 5.
For remission induction in less severe (non–organ threatening) disease, the recommendation is to use glucocorticoids plus either methotrexate or mycophenolate mofetil. Situations when methotrexate or mycophenolate mofetil should and should not be used are specified, notably when cyclophosphamide or rituximab are not available or are contraindicated.
For maintenance of remission, the task force advised using low-dose glucocorticoids plus azathioprine, rituximab, methotrexate, or mycophenolate mofetil.
Guidance on when to use plasma exchange is given for patients with severe disease and options following failure of remission-induction therapy, and when to switch therapy is also covered.
There are also several follow-up recommendations, such as the periodic assessment of cardiovascular risk, and patient-focused recommendations on awareness of the nature, benefits, and risks of therapy.
The recommendations should provide clinicians with reliable guidance on the best approach to treating AAV, according to Dr. Yates. “From the patients’ point of view, these recommendations should provide useful insight into which treatments they are likely to be offered and when. They also emphasize that as a patient, you should have a voice in your treatment and if you have any questions or concerns, be sure to speak with your specialist.”
Dr. Yates and Dr. Mukhtyar did not report having any relevant disclosures.
AT THE EULAR 2016 CONGRESS
Investigational Wnt inhibitor shows promise in knee osteoarthritis
LONDON – Early clinical data show that a novel injectable drug holds promise for becoming the first disease-modifying osteoarthritis drug.
The results of a randomized, placebo-controlled, double-blind phase I trial involving 61 patients showed that a single intra-articular injection of SM04690 was associated with improved Western Ontario and McMaster Universities Arthritis Index (WOMAC) function and pain scores. The investigational drug also seemed to slow joint-space narrowing, compared with baseline values, with the suggestion that it may even increase joint space width.
However, those were exploratory efficacy analyses because the primary objective of the trial was to examine the safety of SM04690, a small molecule that inhibits the Wnt signaling pathway.
“The Wnt pathway has been implicated in the development of osteoarthritis [OA],” said Dr. Yusuf Yazici during a poster presentation at the European Congress of Rheumatology.
“Overactivity of Wnt signaling leads to stem cells constantly differentiating into osteoblasts, leading to osteophyte formation,” he explained, noting that Wnt signaling also stimulates the secretion of cartilage-destroying metalloproteases (Osteoarthritis Cartilage. 2012;20:162-71). “It has been very well established in the literature that if you could somehow turn that off you, could maybe improve some of the things that are happening in osteoarthritis.”
SM04690 works by “pushing the lineage fate of progenitor stem cells in the knee towards chondrocyte formation and away from osteoblast formation,” said Dr. Yazici of New York University Langone Medical Center, New York, and the chief medical officer of Samumed, the San Diego–based company developing the novel Wnt inhibitor.
He noted preclinical data had been presented orally at the EULAR congress showing that there was cartilage growth, suppressed protease production, and reduced proinflammatory cytokine (interleukin-6 and tumor necrosis factor–alpha) production.
The phase I data represent the first in-human results, with three doses of SM04690 evaluated (0.03 mg, 0.07 mg, and 0.23 mg) versus placebo in patients with moderate to severe symptomatic OA. For inclusion, patients had to have a WOMAC total score of between 36 and 72 and Kellgren-Lawrence (KL) grade 2 or 3 knee OA, and be willing to forgo pain medication for 24 hours prior to pain assessments being performed.
At baseline, the mean age of patients ranged from 60 to 64 years, their body mass index ranged from 28.7 kg/m2 to 31.4 kg/m2, and 41%-69% had KL grade 3 knee OA.
In terms of safety, the primary objective of the trial, there were no reports of serious adverse events related to the study drug. One patient who had reported increased knee pain and paroxysmal tachycardia 2 months after the injection was found to have a history of the condition, and after unblinding, none of the patients had detectable drug levels outside of the knee.
Overall, the number of adverse events was low and no different from placebo, Dr. Yazici said. The percentage of patients reporting an adverse event with the three rising doses of SM04690 were 53%, 35%, and 44%, respectively, compared with 55% of those given placebo.
WOMAC function scores for the 0.03-mg dose declined by a mean of –18.4 at week 12 and by –20.1 at week 24 from a baseline of 39.1; for 0.07 mg, by –19.5 at week 12 and by –18.9 at 24 weeks from 37.5; for 0.23 mg, by –17.8 at week 12 and by –12.4 at week 24 from 40.4; and for placebo, by –14.9 at week 12 and by –16.0 at week 24 from 34.4.
WOMAC pain scores at baseline were a respective 10.8, 10.8, 11.4, and 9.9, and the mean changes at week 12 were –4.4, –5.8, –5.7, and –4.2. At week 24, the mean declines were –5.6, –5.3, –4.3, and –4.8.
Medial joint space width was a mean of 4.5, 3.72, 3.62, and 3.74 mm at baseline in the four treatment groups, with mean changes from baseline to 24 weeks of 0.00, 0.49, –0.15, and –0.33 for the 0.03-mg, 0.07-mg, and 0.23-mg SM04690 and placebo groups, respectively.
Although the trial was not powered to detect any statistically significant differences between the active treatment dose and placebo, there was an indication that more patients treated with SM04690 than with placebo were likely to achieve an OMERACT-OARSI strict response.
These data support the ongoing phase II trial that is being conducted in 455 patients, Dr. Yazici said. The results of that trial are expected around October 2016, which should be in time for their presentation at the annual meeting of the American College of Rheumatology.
LONDON – Early clinical data show that a novel injectable drug holds promise for becoming the first disease-modifying osteoarthritis drug.
The results of a randomized, placebo-controlled, double-blind phase I trial involving 61 patients showed that a single intra-articular injection of SM04690 was associated with improved Western Ontario and McMaster Universities Arthritis Index (WOMAC) function and pain scores. The investigational drug also seemed to slow joint-space narrowing, compared with baseline values, with the suggestion that it may even increase joint space width.
However, those were exploratory efficacy analyses because the primary objective of the trial was to examine the safety of SM04690, a small molecule that inhibits the Wnt signaling pathway.
“The Wnt pathway has been implicated in the development of osteoarthritis [OA],” said Dr. Yusuf Yazici during a poster presentation at the European Congress of Rheumatology.
“Overactivity of Wnt signaling leads to stem cells constantly differentiating into osteoblasts, leading to osteophyte formation,” he explained, noting that Wnt signaling also stimulates the secretion of cartilage-destroying metalloproteases (Osteoarthritis Cartilage. 2012;20:162-71). “It has been very well established in the literature that if you could somehow turn that off you, could maybe improve some of the things that are happening in osteoarthritis.”
SM04690 works by “pushing the lineage fate of progenitor stem cells in the knee towards chondrocyte formation and away from osteoblast formation,” said Dr. Yazici of New York University Langone Medical Center, New York, and the chief medical officer of Samumed, the San Diego–based company developing the novel Wnt inhibitor.
He noted preclinical data had been presented orally at the EULAR congress showing that there was cartilage growth, suppressed protease production, and reduced proinflammatory cytokine (interleukin-6 and tumor necrosis factor–alpha) production.
The phase I data represent the first in-human results, with three doses of SM04690 evaluated (0.03 mg, 0.07 mg, and 0.23 mg) versus placebo in patients with moderate to severe symptomatic OA. For inclusion, patients had to have a WOMAC total score of between 36 and 72 and Kellgren-Lawrence (KL) grade 2 or 3 knee OA, and be willing to forgo pain medication for 24 hours prior to pain assessments being performed.
At baseline, the mean age of patients ranged from 60 to 64 years, their body mass index ranged from 28.7 kg/m2 to 31.4 kg/m2, and 41%-69% had KL grade 3 knee OA.
In terms of safety, the primary objective of the trial, there were no reports of serious adverse events related to the study drug. One patient who had reported increased knee pain and paroxysmal tachycardia 2 months after the injection was found to have a history of the condition, and after unblinding, none of the patients had detectable drug levels outside of the knee.
Overall, the number of adverse events was low and no different from placebo, Dr. Yazici said. The percentage of patients reporting an adverse event with the three rising doses of SM04690 were 53%, 35%, and 44%, respectively, compared with 55% of those given placebo.
WOMAC function scores for the 0.03-mg dose declined by a mean of –18.4 at week 12 and by –20.1 at week 24 from a baseline of 39.1; for 0.07 mg, by –19.5 at week 12 and by –18.9 at 24 weeks from 37.5; for 0.23 mg, by –17.8 at week 12 and by –12.4 at week 24 from 40.4; and for placebo, by –14.9 at week 12 and by –16.0 at week 24 from 34.4.
WOMAC pain scores at baseline were a respective 10.8, 10.8, 11.4, and 9.9, and the mean changes at week 12 were –4.4, –5.8, –5.7, and –4.2. At week 24, the mean declines were –5.6, –5.3, –4.3, and –4.8.
Medial joint space width was a mean of 4.5, 3.72, 3.62, and 3.74 mm at baseline in the four treatment groups, with mean changes from baseline to 24 weeks of 0.00, 0.49, –0.15, and –0.33 for the 0.03-mg, 0.07-mg, and 0.23-mg SM04690 and placebo groups, respectively.
Although the trial was not powered to detect any statistically significant differences between the active treatment dose and placebo, there was an indication that more patients treated with SM04690 than with placebo were likely to achieve an OMERACT-OARSI strict response.
These data support the ongoing phase II trial that is being conducted in 455 patients, Dr. Yazici said. The results of that trial are expected around October 2016, which should be in time for their presentation at the annual meeting of the American College of Rheumatology.
LONDON – Early clinical data show that a novel injectable drug holds promise for becoming the first disease-modifying osteoarthritis drug.
The results of a randomized, placebo-controlled, double-blind phase I trial involving 61 patients showed that a single intra-articular injection of SM04690 was associated with improved Western Ontario and McMaster Universities Arthritis Index (WOMAC) function and pain scores. The investigational drug also seemed to slow joint-space narrowing, compared with baseline values, with the suggestion that it may even increase joint space width.
However, those were exploratory efficacy analyses because the primary objective of the trial was to examine the safety of SM04690, a small molecule that inhibits the Wnt signaling pathway.
“The Wnt pathway has been implicated in the development of osteoarthritis [OA],” said Dr. Yusuf Yazici during a poster presentation at the European Congress of Rheumatology.
“Overactivity of Wnt signaling leads to stem cells constantly differentiating into osteoblasts, leading to osteophyte formation,” he explained, noting that Wnt signaling also stimulates the secretion of cartilage-destroying metalloproteases (Osteoarthritis Cartilage. 2012;20:162-71). “It has been very well established in the literature that if you could somehow turn that off you, could maybe improve some of the things that are happening in osteoarthritis.”
SM04690 works by “pushing the lineage fate of progenitor stem cells in the knee towards chondrocyte formation and away from osteoblast formation,” said Dr. Yazici of New York University Langone Medical Center, New York, and the chief medical officer of Samumed, the San Diego–based company developing the novel Wnt inhibitor.
He noted preclinical data had been presented orally at the EULAR congress showing that there was cartilage growth, suppressed protease production, and reduced proinflammatory cytokine (interleukin-6 and tumor necrosis factor–alpha) production.
The phase I data represent the first in-human results, with three doses of SM04690 evaluated (0.03 mg, 0.07 mg, and 0.23 mg) versus placebo in patients with moderate to severe symptomatic OA. For inclusion, patients had to have a WOMAC total score of between 36 and 72 and Kellgren-Lawrence (KL) grade 2 or 3 knee OA, and be willing to forgo pain medication for 24 hours prior to pain assessments being performed.
At baseline, the mean age of patients ranged from 60 to 64 years, their body mass index ranged from 28.7 kg/m2 to 31.4 kg/m2, and 41%-69% had KL grade 3 knee OA.
In terms of safety, the primary objective of the trial, there were no reports of serious adverse events related to the study drug. One patient who had reported increased knee pain and paroxysmal tachycardia 2 months after the injection was found to have a history of the condition, and after unblinding, none of the patients had detectable drug levels outside of the knee.
Overall, the number of adverse events was low and no different from placebo, Dr. Yazici said. The percentage of patients reporting an adverse event with the three rising doses of SM04690 were 53%, 35%, and 44%, respectively, compared with 55% of those given placebo.
WOMAC function scores for the 0.03-mg dose declined by a mean of –18.4 at week 12 and by –20.1 at week 24 from a baseline of 39.1; for 0.07 mg, by –19.5 at week 12 and by –18.9 at 24 weeks from 37.5; for 0.23 mg, by –17.8 at week 12 and by –12.4 at week 24 from 40.4; and for placebo, by –14.9 at week 12 and by –16.0 at week 24 from 34.4.
WOMAC pain scores at baseline were a respective 10.8, 10.8, 11.4, and 9.9, and the mean changes at week 12 were –4.4, –5.8, –5.7, and –4.2. At week 24, the mean declines were –5.6, –5.3, –4.3, and –4.8.
Medial joint space width was a mean of 4.5, 3.72, 3.62, and 3.74 mm at baseline in the four treatment groups, with mean changes from baseline to 24 weeks of 0.00, 0.49, –0.15, and –0.33 for the 0.03-mg, 0.07-mg, and 0.23-mg SM04690 and placebo groups, respectively.
Although the trial was not powered to detect any statistically significant differences between the active treatment dose and placebo, there was an indication that more patients treated with SM04690 than with placebo were likely to achieve an OMERACT-OARSI strict response.
These data support the ongoing phase II trial that is being conducted in 455 patients, Dr. Yazici said. The results of that trial are expected around October 2016, which should be in time for their presentation at the annual meeting of the American College of Rheumatology.
AT THE EULAR 2016 CONGRESS
Key clinical point: Early clinical data show that a novel injectable drug holds promise for becoming the first disease-modifying osteoarthritis drug.
Major finding: SM04690 was well tolerated, and exploratory efficacy analyses showed improved function, pain, and joint space width.
Data source: A multicenter, randomized, placebo-controlled, double-blind phase I trial involving 61 patients with knee osteoarthritis.
Disclosures: Dr. Yazici is chief medical officer of Samumed, the company that funded the study.
European initiative unveils pediatric care recommendations
LONDON – Recommendations on managing juvenile idiopathic arthritis and connective tissue disorders in children and young people across Europe were unveiled at the European Congress of Rheumatology.
The recommendations, which come from the SHARE (Single Hub and Access Point for Paediatric Rheumatology in Europe) project, cover best practices and provide guidance based on current evidence and expert opinion for the optimal diagnosis and treatment of these rare rheumatic diseases that affect the pediatric population.
It is hoped that the recommendations will be used to improve access to treatment and care within individual countries such that a child in one country will be able to receive the same standard of care as a child in another, Dr. Nico Wulffraat of University Medical Center Utrecht (the Netherlands) said in an interview.
Dr. Wulffraat, one of the driving forces behind the project, noted that the SHARE project was set up to look at making the management of rare pediatric rheumatic diseases more uniform across Europe. It addressed conditions such as juvenile idiopathic arthritis (JIA), childhood-onset systemic lupus erythematosus (cSLE), childhood antiphospholipid syndrome (APS), childhood vasculitis, juvenile dermatomyositis, and pediatric scleroderma. In addition, recommendations on diagnosis and treatment of periodic fever syndromes have been developed in collaboration with experts from the Eurofever Project.
“Our evidence- and consensus-based recommendations will hopefully drive access to uniform and optimal care throughout Europe, including off-label therapy when appropriate according to international consensus–derived expert advice,” Dr. Sebastiaan Vastert, SHARE project co-coordinator, said in an interview. He added: “The SHARE network will be invaluable for further international collaboration, both for optimization of care and for international collaboration in research as well.”
Dr. Wulffraat observed that while the recommendations are primarily directed at health care professionals, they also are of use for other stakeholders such as health authorities and insurance companies, and of course patients themselves to ensure the best level of care is being achieved throughout Europe.
The process for developing the guidelines was perhaps as important as the recommendations themselves, said Dr. Vastert, also of University Medical Center Utrecht. The process helped to build a network of international experts who could work together to develop future recommendations for improving patient care.
The recommendations for JIA and other pediatric rheumatic diseases included 51 “cross-cutting” statements, Dr. Vastert said. One of these statements was that a pediatric rheumatologist should manage children with signs of rheumatic disease. Another highlighted the members of a multidisciplinary team who should be involved as appropriate, such as a nurse specializing in pediatric rheumatic disease, a physiotherapist or occupational therapist, and a psychologist or psychosocial worker. Dr. Vastert also noted that good communication between team members is essential. In addition, there needs to be clear guidance on when to refer to a pediatric rheumatologist.
The SHARE project JIA recommendations include 10 evidence-based statements on diagnosis, 31 evidence-based statements on treatment, and 17 general statements on specific care for JIA, Dr. Vastert said. A few examples of the latter are that new patients should be seen in a specialist center within 4 weeks of referral; new patients and those starting a new therapy should be reviewed within 2-3 months to check on adherence, tolerance, and disease progression; and monitoring response to ongoing treatment should be every 3-6 months, preferably using existing standardized disease activity tools.
EULAR standard operating procedures were followed when developing the various SHARE recommendations, said Dr. Michael Beresford of the University of Liverpool (England) and the lead for the recommendations on childhood connective tissue disorders. Dr. Beresford noted that the latter were a rare, and in some cases extremely rare, complex group of pediatric rheumatic diseases that could lead to significant morbidity and mortality.
“Evidence-based guidelines have been lacking, and management is based mainly on physician experience. Consequently, treatment regimens vary widely throughout Europe,” Dr. Beresford observed. “These [recommendations] provide evidence-based, internationally agreed-upon standards of optimal care for pediatric connective tissue disorders.”
Specifically, the connective tissue disorder recommendations cover when to refer and how to diagnose, treat, and monitor cSLE (including neuropsychiatric SLE), childhood APS, and juvenile vasculitides, including rare pediatric vasculitides such as Takayasu arteritis. The SHARE recommendations for the management of juvenile dermatomyositis are currently in press in Annals of the Rheumatic Diseases, Dr. Beresford said.
Giving a few examples of recommendations for cSLE, Dr. Beresford noted that one of the challenges is to try to prevent delay in diagnosis. The expert panel decided that the 2012 Systemic Lupus International Collaborating Clinics (SLICC) criteria could be used for diagnosis. A referral to a pediatric rheumatologist is warranted, they determined, when a child has a positive antinuclear antibody (ANA) test and meets two clinical SLICC criteria. Dr. Beresford conceded that antibody testing might not be available because of cost in all countries, but they “decided to draw a line in the sand” to say that it is important that it is routinely done in order to come closer to a definitive diagnosis.
The aim of treatment for cSLE, the recommendations advise, is to optimize control and prevent damage caused by both the disease and by its treatment. For example, all children should be on hydroxychloroquine, and if tapering of prednisone is not possible, a disease-modifying antirheumatic drug should be added. It’s also important to actively check compliance with therapy, Dr. Beresford said.
The SHARE project was initially funded by a grant from the European Agency for Health and Consumers between 2012 and 2015 and now continues under the auspices of the Paediatric Rheumatology European Society. All speakers reported having no relevant disclosures.
LONDON – Recommendations on managing juvenile idiopathic arthritis and connective tissue disorders in children and young people across Europe were unveiled at the European Congress of Rheumatology.
The recommendations, which come from the SHARE (Single Hub and Access Point for Paediatric Rheumatology in Europe) project, cover best practices and provide guidance based on current evidence and expert opinion for the optimal diagnosis and treatment of these rare rheumatic diseases that affect the pediatric population.
It is hoped that the recommendations will be used to improve access to treatment and care within individual countries such that a child in one country will be able to receive the same standard of care as a child in another, Dr. Nico Wulffraat of University Medical Center Utrecht (the Netherlands) said in an interview.
Dr. Wulffraat, one of the driving forces behind the project, noted that the SHARE project was set up to look at making the management of rare pediatric rheumatic diseases more uniform across Europe. It addressed conditions such as juvenile idiopathic arthritis (JIA), childhood-onset systemic lupus erythematosus (cSLE), childhood antiphospholipid syndrome (APS), childhood vasculitis, juvenile dermatomyositis, and pediatric scleroderma. In addition, recommendations on diagnosis and treatment of periodic fever syndromes have been developed in collaboration with experts from the Eurofever Project.
“Our evidence- and consensus-based recommendations will hopefully drive access to uniform and optimal care throughout Europe, including off-label therapy when appropriate according to international consensus–derived expert advice,” Dr. Sebastiaan Vastert, SHARE project co-coordinator, said in an interview. He added: “The SHARE network will be invaluable for further international collaboration, both for optimization of care and for international collaboration in research as well.”
Dr. Wulffraat observed that while the recommendations are primarily directed at health care professionals, they also are of use for other stakeholders such as health authorities and insurance companies, and of course patients themselves to ensure the best level of care is being achieved throughout Europe.
The process for developing the guidelines was perhaps as important as the recommendations themselves, said Dr. Vastert, also of University Medical Center Utrecht. The process helped to build a network of international experts who could work together to develop future recommendations for improving patient care.
The recommendations for JIA and other pediatric rheumatic diseases included 51 “cross-cutting” statements, Dr. Vastert said. One of these statements was that a pediatric rheumatologist should manage children with signs of rheumatic disease. Another highlighted the members of a multidisciplinary team who should be involved as appropriate, such as a nurse specializing in pediatric rheumatic disease, a physiotherapist or occupational therapist, and a psychologist or psychosocial worker. Dr. Vastert also noted that good communication between team members is essential. In addition, there needs to be clear guidance on when to refer to a pediatric rheumatologist.
The SHARE project JIA recommendations include 10 evidence-based statements on diagnosis, 31 evidence-based statements on treatment, and 17 general statements on specific care for JIA, Dr. Vastert said. A few examples of the latter are that new patients should be seen in a specialist center within 4 weeks of referral; new patients and those starting a new therapy should be reviewed within 2-3 months to check on adherence, tolerance, and disease progression; and monitoring response to ongoing treatment should be every 3-6 months, preferably using existing standardized disease activity tools.
EULAR standard operating procedures were followed when developing the various SHARE recommendations, said Dr. Michael Beresford of the University of Liverpool (England) and the lead for the recommendations on childhood connective tissue disorders. Dr. Beresford noted that the latter were a rare, and in some cases extremely rare, complex group of pediatric rheumatic diseases that could lead to significant morbidity and mortality.
“Evidence-based guidelines have been lacking, and management is based mainly on physician experience. Consequently, treatment regimens vary widely throughout Europe,” Dr. Beresford observed. “These [recommendations] provide evidence-based, internationally agreed-upon standards of optimal care for pediatric connective tissue disorders.”
Specifically, the connective tissue disorder recommendations cover when to refer and how to diagnose, treat, and monitor cSLE (including neuropsychiatric SLE), childhood APS, and juvenile vasculitides, including rare pediatric vasculitides such as Takayasu arteritis. The SHARE recommendations for the management of juvenile dermatomyositis are currently in press in Annals of the Rheumatic Diseases, Dr. Beresford said.
Giving a few examples of recommendations for cSLE, Dr. Beresford noted that one of the challenges is to try to prevent delay in diagnosis. The expert panel decided that the 2012 Systemic Lupus International Collaborating Clinics (SLICC) criteria could be used for diagnosis. A referral to a pediatric rheumatologist is warranted, they determined, when a child has a positive antinuclear antibody (ANA) test and meets two clinical SLICC criteria. Dr. Beresford conceded that antibody testing might not be available because of cost in all countries, but they “decided to draw a line in the sand” to say that it is important that it is routinely done in order to come closer to a definitive diagnosis.
The aim of treatment for cSLE, the recommendations advise, is to optimize control and prevent damage caused by both the disease and by its treatment. For example, all children should be on hydroxychloroquine, and if tapering of prednisone is not possible, a disease-modifying antirheumatic drug should be added. It’s also important to actively check compliance with therapy, Dr. Beresford said.
The SHARE project was initially funded by a grant from the European Agency for Health and Consumers between 2012 and 2015 and now continues under the auspices of the Paediatric Rheumatology European Society. All speakers reported having no relevant disclosures.
LONDON – Recommendations on managing juvenile idiopathic arthritis and connective tissue disorders in children and young people across Europe were unveiled at the European Congress of Rheumatology.
The recommendations, which come from the SHARE (Single Hub and Access Point for Paediatric Rheumatology in Europe) project, cover best practices and provide guidance based on current evidence and expert opinion for the optimal diagnosis and treatment of these rare rheumatic diseases that affect the pediatric population.
It is hoped that the recommendations will be used to improve access to treatment and care within individual countries such that a child in one country will be able to receive the same standard of care as a child in another, Dr. Nico Wulffraat of University Medical Center Utrecht (the Netherlands) said in an interview.
Dr. Wulffraat, one of the driving forces behind the project, noted that the SHARE project was set up to look at making the management of rare pediatric rheumatic diseases more uniform across Europe. It addressed conditions such as juvenile idiopathic arthritis (JIA), childhood-onset systemic lupus erythematosus (cSLE), childhood antiphospholipid syndrome (APS), childhood vasculitis, juvenile dermatomyositis, and pediatric scleroderma. In addition, recommendations on diagnosis and treatment of periodic fever syndromes have been developed in collaboration with experts from the Eurofever Project.
“Our evidence- and consensus-based recommendations will hopefully drive access to uniform and optimal care throughout Europe, including off-label therapy when appropriate according to international consensus–derived expert advice,” Dr. Sebastiaan Vastert, SHARE project co-coordinator, said in an interview. He added: “The SHARE network will be invaluable for further international collaboration, both for optimization of care and for international collaboration in research as well.”
Dr. Wulffraat observed that while the recommendations are primarily directed at health care professionals, they also are of use for other stakeholders such as health authorities and insurance companies, and of course patients themselves to ensure the best level of care is being achieved throughout Europe.
The process for developing the guidelines was perhaps as important as the recommendations themselves, said Dr. Vastert, also of University Medical Center Utrecht. The process helped to build a network of international experts who could work together to develop future recommendations for improving patient care.
The recommendations for JIA and other pediatric rheumatic diseases included 51 “cross-cutting” statements, Dr. Vastert said. One of these statements was that a pediatric rheumatologist should manage children with signs of rheumatic disease. Another highlighted the members of a multidisciplinary team who should be involved as appropriate, such as a nurse specializing in pediatric rheumatic disease, a physiotherapist or occupational therapist, and a psychologist or psychosocial worker. Dr. Vastert also noted that good communication between team members is essential. In addition, there needs to be clear guidance on when to refer to a pediatric rheumatologist.
The SHARE project JIA recommendations include 10 evidence-based statements on diagnosis, 31 evidence-based statements on treatment, and 17 general statements on specific care for JIA, Dr. Vastert said. A few examples of the latter are that new patients should be seen in a specialist center within 4 weeks of referral; new patients and those starting a new therapy should be reviewed within 2-3 months to check on adherence, tolerance, and disease progression; and monitoring response to ongoing treatment should be every 3-6 months, preferably using existing standardized disease activity tools.
EULAR standard operating procedures were followed when developing the various SHARE recommendations, said Dr. Michael Beresford of the University of Liverpool (England) and the lead for the recommendations on childhood connective tissue disorders. Dr. Beresford noted that the latter were a rare, and in some cases extremely rare, complex group of pediatric rheumatic diseases that could lead to significant morbidity and mortality.
“Evidence-based guidelines have been lacking, and management is based mainly on physician experience. Consequently, treatment regimens vary widely throughout Europe,” Dr. Beresford observed. “These [recommendations] provide evidence-based, internationally agreed-upon standards of optimal care for pediatric connective tissue disorders.”
Specifically, the connective tissue disorder recommendations cover when to refer and how to diagnose, treat, and monitor cSLE (including neuropsychiatric SLE), childhood APS, and juvenile vasculitides, including rare pediatric vasculitides such as Takayasu arteritis. The SHARE recommendations for the management of juvenile dermatomyositis are currently in press in Annals of the Rheumatic Diseases, Dr. Beresford said.
Giving a few examples of recommendations for cSLE, Dr. Beresford noted that one of the challenges is to try to prevent delay in diagnosis. The expert panel decided that the 2012 Systemic Lupus International Collaborating Clinics (SLICC) criteria could be used for diagnosis. A referral to a pediatric rheumatologist is warranted, they determined, when a child has a positive antinuclear antibody (ANA) test and meets two clinical SLICC criteria. Dr. Beresford conceded that antibody testing might not be available because of cost in all countries, but they “decided to draw a line in the sand” to say that it is important that it is routinely done in order to come closer to a definitive diagnosis.
The aim of treatment for cSLE, the recommendations advise, is to optimize control and prevent damage caused by both the disease and by its treatment. For example, all children should be on hydroxychloroquine, and if tapering of prednisone is not possible, a disease-modifying antirheumatic drug should be added. It’s also important to actively check compliance with therapy, Dr. Beresford said.
The SHARE project was initially funded by a grant from the European Agency for Health and Consumers between 2012 and 2015 and now continues under the auspices of the Paediatric Rheumatology European Society. All speakers reported having no relevant disclosures.
EXPERT ANALYSIS FROM THE EULAR 2016 CONGRESS