User login
Bringing you the latest news, research and reviews, exclusive interviews, podcasts, quizzes, and more.
div[contains(@class, 'read-next-article')]
div[contains(@class, 'nav-primary')]
nav[contains(@class, 'nav-primary')]
section[contains(@class, 'footer-nav-section-wrapper')]
nav[contains(@class, 'nav-ce-stack nav-ce-stack__large-screen')]
header[@id='header']
div[contains(@class, 'header__large-screen')]
div[contains(@class, 'read-next-article')]
div[contains(@class, 'main-prefix')]
div[contains(@class, 'nav-primary')]
nav[contains(@class, 'nav-primary')]
section[contains(@class, 'footer-nav-section-wrapper')]
footer[@id='footer']
section[contains(@class, 'nav-hidden')]
div[contains(@class, 'ce-card-content')]
nav[contains(@class, 'nav-ce-stack')]
div[contains(@class, 'view-medstat-quiz-listing-panes')]
div[contains(@class, 'pane-article-sidebar-latest-news')]
Mastering mask communicating
. For those specialties not accustomed to wearing a mask all day, it’s frustrating: How many times have you had to repeat yourself today? Or ask your patient to say something again? (Ain’t no one got time to repeat a third time how to do that prednisone taper). Worse, we’re losing important nonverbal cues that help us connect with our patients. How can we be understood when our faces are covered and 6 feet away?
Masks muffle both verbal and nonverbal communication. For soft-spoken or high-pitched speakers, the verbal effect is significant. In particular, masks make hearing consonants more difficult. They can make the “sh,” “th,” “f,” and “s” sounds difficult to distinguish. Typically, we’d use context and lip reading to boost the signal, but this fix is blocked (and the clear mouth-window masks are kinda creepy).
Masks also prevent us from seeing facial microexpressions, critical information when you are trying to connect with someone or to build trust. A randomized controlled trial published in 2013 indeed showed that doctors wearing a mask were perceived as less empathetic and had diminished relational continuity with patients as compared to doctors not wearing a mask. There are a few things we can do to help.
Speak more loudly is obvious advice. Loud talking has limitations though, as it can feel rude, and it blunts inflections, which add richness and emotion. (Shouting “THIS WILL ONLY HURT A LITTLE” seems a mixed message). More important than the volume is your choice of words. Try to use simple terms and short sentences. Pause between points. Hit your consonants harder.
It’s also important that you have their full attention and are giving yours. As much as possible, try to align squared up with patients. Facing your computer exacerbates the problem. Look them in their eyes and be sure they are connected with you before any complex or difficult conversations. Hearing-impaired patients are now sometimes leaving out their aids because it’s too uncomfortable to wear them with their mask. You might ask them to put them back in. Check in with patients and repeat back what you heard them say. This can help with clarity and with connecting. Use your face more: if you’ve ever acted on stage, this would be your on-stage face. Exaggerate your expressions so it’s a little easier for them to read you.
Lastly, there are apps such as Ava or Google Live Translator, which can transcribe your speech real time. You could then share your screen with the patient so they can read exactly what you’ve said.
Some of us are natural communicators. Even if you are not, you can mitigate some of our current challenges. I’ll admit, it’s been a bit easier for me than for others. Between my prominent eyebrows and Italian-American upbringing, I can express my way through pretty much any face covering. If you’d like to learn how to use your hands better, then just watch this little girl: https://youtu.be/Z5wAWyqDrnc.
Dr. Benabio is director of Healthcare Transformation and chief of dermatology at Kaiser Permanente San Diego. The opinions expressed in this column are his own and do not represent those of Kaiser Permanente. Dr. Benabio is @Dermdoc on Twitter. Write to him at [email protected].
. For those specialties not accustomed to wearing a mask all day, it’s frustrating: How many times have you had to repeat yourself today? Or ask your patient to say something again? (Ain’t no one got time to repeat a third time how to do that prednisone taper). Worse, we’re losing important nonverbal cues that help us connect with our patients. How can we be understood when our faces are covered and 6 feet away?
Masks muffle both verbal and nonverbal communication. For soft-spoken or high-pitched speakers, the verbal effect is significant. In particular, masks make hearing consonants more difficult. They can make the “sh,” “th,” “f,” and “s” sounds difficult to distinguish. Typically, we’d use context and lip reading to boost the signal, but this fix is blocked (and the clear mouth-window masks are kinda creepy).
Masks also prevent us from seeing facial microexpressions, critical information when you are trying to connect with someone or to build trust. A randomized controlled trial published in 2013 indeed showed that doctors wearing a mask were perceived as less empathetic and had diminished relational continuity with patients as compared to doctors not wearing a mask. There are a few things we can do to help.
Speak more loudly is obvious advice. Loud talking has limitations though, as it can feel rude, and it blunts inflections, which add richness and emotion. (Shouting “THIS WILL ONLY HURT A LITTLE” seems a mixed message). More important than the volume is your choice of words. Try to use simple terms and short sentences. Pause between points. Hit your consonants harder.
It’s also important that you have their full attention and are giving yours. As much as possible, try to align squared up with patients. Facing your computer exacerbates the problem. Look them in their eyes and be sure they are connected with you before any complex or difficult conversations. Hearing-impaired patients are now sometimes leaving out their aids because it’s too uncomfortable to wear them with their mask. You might ask them to put them back in. Check in with patients and repeat back what you heard them say. This can help with clarity and with connecting. Use your face more: if you’ve ever acted on stage, this would be your on-stage face. Exaggerate your expressions so it’s a little easier for them to read you.
Lastly, there are apps such as Ava or Google Live Translator, which can transcribe your speech real time. You could then share your screen with the patient so they can read exactly what you’ve said.
Some of us are natural communicators. Even if you are not, you can mitigate some of our current challenges. I’ll admit, it’s been a bit easier for me than for others. Between my prominent eyebrows and Italian-American upbringing, I can express my way through pretty much any face covering. If you’d like to learn how to use your hands better, then just watch this little girl: https://youtu.be/Z5wAWyqDrnc.
Dr. Benabio is director of Healthcare Transformation and chief of dermatology at Kaiser Permanente San Diego. The opinions expressed in this column are his own and do not represent those of Kaiser Permanente. Dr. Benabio is @Dermdoc on Twitter. Write to him at [email protected].
. For those specialties not accustomed to wearing a mask all day, it’s frustrating: How many times have you had to repeat yourself today? Or ask your patient to say something again? (Ain’t no one got time to repeat a third time how to do that prednisone taper). Worse, we’re losing important nonverbal cues that help us connect with our patients. How can we be understood when our faces are covered and 6 feet away?
Masks muffle both verbal and nonverbal communication. For soft-spoken or high-pitched speakers, the verbal effect is significant. In particular, masks make hearing consonants more difficult. They can make the “sh,” “th,” “f,” and “s” sounds difficult to distinguish. Typically, we’d use context and lip reading to boost the signal, but this fix is blocked (and the clear mouth-window masks are kinda creepy).
Masks also prevent us from seeing facial microexpressions, critical information when you are trying to connect with someone or to build trust. A randomized controlled trial published in 2013 indeed showed that doctors wearing a mask were perceived as less empathetic and had diminished relational continuity with patients as compared to doctors not wearing a mask. There are a few things we can do to help.
Speak more loudly is obvious advice. Loud talking has limitations though, as it can feel rude, and it blunts inflections, which add richness and emotion. (Shouting “THIS WILL ONLY HURT A LITTLE” seems a mixed message). More important than the volume is your choice of words. Try to use simple terms and short sentences. Pause between points. Hit your consonants harder.
It’s also important that you have their full attention and are giving yours. As much as possible, try to align squared up with patients. Facing your computer exacerbates the problem. Look them in their eyes and be sure they are connected with you before any complex or difficult conversations. Hearing-impaired patients are now sometimes leaving out their aids because it’s too uncomfortable to wear them with their mask. You might ask them to put them back in. Check in with patients and repeat back what you heard them say. This can help with clarity and with connecting. Use your face more: if you’ve ever acted on stage, this would be your on-stage face. Exaggerate your expressions so it’s a little easier for them to read you.
Lastly, there are apps such as Ava or Google Live Translator, which can transcribe your speech real time. You could then share your screen with the patient so they can read exactly what you’ve said.
Some of us are natural communicators. Even if you are not, you can mitigate some of our current challenges. I’ll admit, it’s been a bit easier for me than for others. Between my prominent eyebrows and Italian-American upbringing, I can express my way through pretty much any face covering. If you’d like to learn how to use your hands better, then just watch this little girl: https://youtu.be/Z5wAWyqDrnc.
Dr. Benabio is director of Healthcare Transformation and chief of dermatology at Kaiser Permanente San Diego. The opinions expressed in this column are his own and do not represent those of Kaiser Permanente. Dr. Benabio is @Dermdoc on Twitter. Write to him at [email protected].
Entresto halves renal events in preserved EF heart failure patients
Patients with heart failure with preserved ejection fraction (HFpEF) who received sacubitril/valsartan in the PARAGON-HF trial had significant protection against progression of renal dysfunction in a prespecified secondary analysis.
The 2,419 patients with HFpEF who received sacubitril/valsartan (Entresto) had half the rate of the primary adverse renal outcome, compared with the 2,403 patients randomized to valsartan alone in the comparator group, a significant difference, according to the results published online Sept. 29 in Circulation by Finnian R. McCausland, MBBCh, and colleagues.
In absolute terms, sacubitril/valsartan treatment, an angiotensin-receptor/neprilysin inhibitor (ARNI), cut the incidence of the combined renal endpoint – renal death, end-stage renal disease, or at least a 50% drop in estimated glomerular filtration rate (eGFR) – from 2.7% in the control group to 1.4% in the sacubitril/valsartan group during a median follow-up of 35 months.
The absolute difference of 1.3% equated to a number needed to treat of 51 to prevent one of these events.
Also notable was that renal protection from sacubitril/valsartan was equally robust across the range of baseline kidney function.
‘An important therapeutic option’
The efficacy “across the spectrum of baseline renal function” indicates treatment with sacubitril/valsartan is “an important therapeutic option to slow renal-function decline in patients with heart failure,” wrote Dr. McCausland, a nephrologist at Brigham and Women’s Hospital in Boston, and colleagues.
The authors’ conclusion is striking because currently no drug class has produced clear evidence for efficacy in HFpEF.
On the other hand, the PARAGON-HF trial that provided the data for this new analysis was statistically neutral for its primary endpoint – a reduction in the combined rate of cardiovascular death and hospitalizations for heart failure – with a P value of .06 and 95% confidence interval of 0.75-1.01.
“Because this difference [in the primary endpoint incidence between the two study group] did not meet the predetermined level of statistical significance, subsequent analyses were considered to be exploratory,” noted the authors of the primary analysis of PARAGON-HF, as reported by Medscape Medical News.
Despite this limitation in interpreting secondary outcomes from the trial, the new report of a significant renal benefit “opens the potential to provide evidence-based treatment for patients with HFpEF,” commented Sheldon W. Tobe, MD, and Stephanie Poon, MD, in an editorial accompanying the latest analysis.
“At the very least, these results are certainly intriguing and suggest that there may be important patient subgroups with HFpEF who might benefit from using sacubitril/valsartan,” they emphasized.
First large trial to show renal improvement in HFpEF
The editorialists’ enthusiasm for the implications of the new findings relate in part to the fact that “PARAGON-HF is the first large trial to demonstrate improvement in renal parameters in HFpEF,” they noted.
“The finding that the composite renal outcome did not differ according to baseline eGFR is significant and suggests that the beneficial effect on renal function was indirect, possibly linked to improved cardiac function,” say Dr. Tobe, a nephrologist, and Dr. Poon, a cardiologist, both at Sunnybrook Health Sciences Centre in Toronto.
PARAGON-HF enrolled 4,822 HFpEF patients at 848 centers in 43 countries, and the efficacy analysis included 4,796 patients.
The composite renal outcome was mainly driven by the incidence of a 50% or greater drop from baseline in eGFR, which occurred in 27 patients (1.1%) in the sacubitril/valsartan group and 60 patients (2.5%) who received valsartan alone.
The annual average drop in eGFR during the study was 2.0 mL/min per 1.73m2 in the sacubitril/valsartan group and 2.7 mL/min per 1.73m2 in the control group.
Although the heart failure community was disappointed that sacubitril/valsartan failed to show a significant benefit for the study’s primary outcome in HFpEF, the combination has become a mainstay of treatment for patients with HFpEF based on its performance in the PARADIGM-HF trial.
And despite the unqualified support sacubitril/valsartan now receives in guidelines and its label as a foundational treatment for HFpEF, the formulation has had a hard time gaining traction in U.S. practice, often because of barriers placed by third-party payers.
PARAGON-HF was sponsored by Novartis, which markets sacubitril/valsartan (Entresto). Dr. McCausland has reported no relevant financial relationships. Dr. Tobe has reported participating on a steering committee for Bayer Fidelio/Figaro studies and being a speaker on behalf of Pfizer and Servier. Dr. Poon has reported being an adviser to Novartis, Boehringer Ingelheim, and Servier.
A version of this article originally appeared on Medscape.com.
Patients with heart failure with preserved ejection fraction (HFpEF) who received sacubitril/valsartan in the PARAGON-HF trial had significant protection against progression of renal dysfunction in a prespecified secondary analysis.
The 2,419 patients with HFpEF who received sacubitril/valsartan (Entresto) had half the rate of the primary adverse renal outcome, compared with the 2,403 patients randomized to valsartan alone in the comparator group, a significant difference, according to the results published online Sept. 29 in Circulation by Finnian R. McCausland, MBBCh, and colleagues.
In absolute terms, sacubitril/valsartan treatment, an angiotensin-receptor/neprilysin inhibitor (ARNI), cut the incidence of the combined renal endpoint – renal death, end-stage renal disease, or at least a 50% drop in estimated glomerular filtration rate (eGFR) – from 2.7% in the control group to 1.4% in the sacubitril/valsartan group during a median follow-up of 35 months.
The absolute difference of 1.3% equated to a number needed to treat of 51 to prevent one of these events.
Also notable was that renal protection from sacubitril/valsartan was equally robust across the range of baseline kidney function.
‘An important therapeutic option’
The efficacy “across the spectrum of baseline renal function” indicates treatment with sacubitril/valsartan is “an important therapeutic option to slow renal-function decline in patients with heart failure,” wrote Dr. McCausland, a nephrologist at Brigham and Women’s Hospital in Boston, and colleagues.
The authors’ conclusion is striking because currently no drug class has produced clear evidence for efficacy in HFpEF.
On the other hand, the PARAGON-HF trial that provided the data for this new analysis was statistically neutral for its primary endpoint – a reduction in the combined rate of cardiovascular death and hospitalizations for heart failure – with a P value of .06 and 95% confidence interval of 0.75-1.01.
“Because this difference [in the primary endpoint incidence between the two study group] did not meet the predetermined level of statistical significance, subsequent analyses were considered to be exploratory,” noted the authors of the primary analysis of PARAGON-HF, as reported by Medscape Medical News.
Despite this limitation in interpreting secondary outcomes from the trial, the new report of a significant renal benefit “opens the potential to provide evidence-based treatment for patients with HFpEF,” commented Sheldon W. Tobe, MD, and Stephanie Poon, MD, in an editorial accompanying the latest analysis.
“At the very least, these results are certainly intriguing and suggest that there may be important patient subgroups with HFpEF who might benefit from using sacubitril/valsartan,” they emphasized.
First large trial to show renal improvement in HFpEF
The editorialists’ enthusiasm for the implications of the new findings relate in part to the fact that “PARAGON-HF is the first large trial to demonstrate improvement in renal parameters in HFpEF,” they noted.
“The finding that the composite renal outcome did not differ according to baseline eGFR is significant and suggests that the beneficial effect on renal function was indirect, possibly linked to improved cardiac function,” say Dr. Tobe, a nephrologist, and Dr. Poon, a cardiologist, both at Sunnybrook Health Sciences Centre in Toronto.
PARAGON-HF enrolled 4,822 HFpEF patients at 848 centers in 43 countries, and the efficacy analysis included 4,796 patients.
The composite renal outcome was mainly driven by the incidence of a 50% or greater drop from baseline in eGFR, which occurred in 27 patients (1.1%) in the sacubitril/valsartan group and 60 patients (2.5%) who received valsartan alone.
The annual average drop in eGFR during the study was 2.0 mL/min per 1.73m2 in the sacubitril/valsartan group and 2.7 mL/min per 1.73m2 in the control group.
Although the heart failure community was disappointed that sacubitril/valsartan failed to show a significant benefit for the study’s primary outcome in HFpEF, the combination has become a mainstay of treatment for patients with HFpEF based on its performance in the PARADIGM-HF trial.
And despite the unqualified support sacubitril/valsartan now receives in guidelines and its label as a foundational treatment for HFpEF, the formulation has had a hard time gaining traction in U.S. practice, often because of barriers placed by third-party payers.
PARAGON-HF was sponsored by Novartis, which markets sacubitril/valsartan (Entresto). Dr. McCausland has reported no relevant financial relationships. Dr. Tobe has reported participating on a steering committee for Bayer Fidelio/Figaro studies and being a speaker on behalf of Pfizer and Servier. Dr. Poon has reported being an adviser to Novartis, Boehringer Ingelheim, and Servier.
A version of this article originally appeared on Medscape.com.
Patients with heart failure with preserved ejection fraction (HFpEF) who received sacubitril/valsartan in the PARAGON-HF trial had significant protection against progression of renal dysfunction in a prespecified secondary analysis.
The 2,419 patients with HFpEF who received sacubitril/valsartan (Entresto) had half the rate of the primary adverse renal outcome, compared with the 2,403 patients randomized to valsartan alone in the comparator group, a significant difference, according to the results published online Sept. 29 in Circulation by Finnian R. McCausland, MBBCh, and colleagues.
In absolute terms, sacubitril/valsartan treatment, an angiotensin-receptor/neprilysin inhibitor (ARNI), cut the incidence of the combined renal endpoint – renal death, end-stage renal disease, or at least a 50% drop in estimated glomerular filtration rate (eGFR) – from 2.7% in the control group to 1.4% in the sacubitril/valsartan group during a median follow-up of 35 months.
The absolute difference of 1.3% equated to a number needed to treat of 51 to prevent one of these events.
Also notable was that renal protection from sacubitril/valsartan was equally robust across the range of baseline kidney function.
‘An important therapeutic option’
The efficacy “across the spectrum of baseline renal function” indicates treatment with sacubitril/valsartan is “an important therapeutic option to slow renal-function decline in patients with heart failure,” wrote Dr. McCausland, a nephrologist at Brigham and Women’s Hospital in Boston, and colleagues.
The authors’ conclusion is striking because currently no drug class has produced clear evidence for efficacy in HFpEF.
On the other hand, the PARAGON-HF trial that provided the data for this new analysis was statistically neutral for its primary endpoint – a reduction in the combined rate of cardiovascular death and hospitalizations for heart failure – with a P value of .06 and 95% confidence interval of 0.75-1.01.
“Because this difference [in the primary endpoint incidence between the two study group] did not meet the predetermined level of statistical significance, subsequent analyses were considered to be exploratory,” noted the authors of the primary analysis of PARAGON-HF, as reported by Medscape Medical News.
Despite this limitation in interpreting secondary outcomes from the trial, the new report of a significant renal benefit “opens the potential to provide evidence-based treatment for patients with HFpEF,” commented Sheldon W. Tobe, MD, and Stephanie Poon, MD, in an editorial accompanying the latest analysis.
“At the very least, these results are certainly intriguing and suggest that there may be important patient subgroups with HFpEF who might benefit from using sacubitril/valsartan,” they emphasized.
First large trial to show renal improvement in HFpEF
The editorialists’ enthusiasm for the implications of the new findings relate in part to the fact that “PARAGON-HF is the first large trial to demonstrate improvement in renal parameters in HFpEF,” they noted.
“The finding that the composite renal outcome did not differ according to baseline eGFR is significant and suggests that the beneficial effect on renal function was indirect, possibly linked to improved cardiac function,” say Dr. Tobe, a nephrologist, and Dr. Poon, a cardiologist, both at Sunnybrook Health Sciences Centre in Toronto.
PARAGON-HF enrolled 4,822 HFpEF patients at 848 centers in 43 countries, and the efficacy analysis included 4,796 patients.
The composite renal outcome was mainly driven by the incidence of a 50% or greater drop from baseline in eGFR, which occurred in 27 patients (1.1%) in the sacubitril/valsartan group and 60 patients (2.5%) who received valsartan alone.
The annual average drop in eGFR during the study was 2.0 mL/min per 1.73m2 in the sacubitril/valsartan group and 2.7 mL/min per 1.73m2 in the control group.
Although the heart failure community was disappointed that sacubitril/valsartan failed to show a significant benefit for the study’s primary outcome in HFpEF, the combination has become a mainstay of treatment for patients with HFpEF based on its performance in the PARADIGM-HF trial.
And despite the unqualified support sacubitril/valsartan now receives in guidelines and its label as a foundational treatment for HFpEF, the formulation has had a hard time gaining traction in U.S. practice, often because of barriers placed by third-party payers.
PARAGON-HF was sponsored by Novartis, which markets sacubitril/valsartan (Entresto). Dr. McCausland has reported no relevant financial relationships. Dr. Tobe has reported participating on a steering committee for Bayer Fidelio/Figaro studies and being a speaker on behalf of Pfizer and Servier. Dr. Poon has reported being an adviser to Novartis, Boehringer Ingelheim, and Servier.
A version of this article originally appeared on Medscape.com.
Decline in febuxostat use trends with cardiovascular concerns
Use of febuxostat (Uloric) decreased among patients with gout in the United States following a Food and Drug Administration–mandated black-box warning that cited cardiovascular concerns, but overall use of urate-lowering therapy remained stable, according to data from a study of commercial insurance enrollees in the United States between 2009 and 2019.
Initiation of urate-lowering therapy (ULT) is recommended for gout patients, and allopurinol remains the first-line treatment, but it is not effective in all patients, and febuxostat was developed as an alternative, wrote Seoyoung C. Kim, MD, ScD, of Brigham and Women’s Hospital and Harvard Medical School, Boston, and colleagues.
However, based on data from a postmarketing safety trial (the CARES trial) mandated by the FDA, the agency first issued a safety announcement about an increased risk of cardiovascular mortality with febuxostat in November 2017, followed in February 2019 with a black-box warning after full CARES trial results were published in March 2018 showing a greater risk of cardiovascular and all-cause mortality in febuxostat versus allopurinol.
In a study published in Arthritis & Rheumatology, the researchers examined trends in the use of ULT before and after the FDA warning. They analyzed claims data from a national commercial health database that included 838,432 adult ULT users and 633,229 gout patients.
Overall, allopurinol accounted for the majority of ULT use between 2009 (95% in the first quarter) and 2019 (92% in the fourth quarter).
Febuxostat use peaked at 10% of all ULT use in 2013 and 2014, after a gradual increase following its introduction into the market in 2009, the researchers noted, but decreased to 6% of all ULT use in the fourth quarter of 2019. Other medications, including probenecid, lesinurad (Zurampic), and pegloticase (Krystexxa), accounted for no more than 5% of ULT use.
When the researchers examined gout patients in particular, they found a slight increase in any ULT use from 567 per 1,000 patients in the first quarter of 2009 to 656 per 1,000 patients in the fourth quarter of 2019.
The study findings were limited by several factors, including potential lack of generalizability to other health plans and lack of adjustment for comorbid conditions, the researchers noted.
However, the results highlight the suboptimal use of ULT as a class and the need to address the treatment gap in gout patients “with appropriate ULT prescribing and monitoring,” they said. “While the decrease in febuxostat use was accompanied by a compensatory increase in allopurinol use, the proportion of patients with gout without any ULT remained high throughout the study period,” they concluded.
The study was supported by the division of pharmacoepidemiology and pharmacoeconomics at Brigham and Women’s Hospital. Dr. Kim disclosed receiving research grants to Brigham and Women’s Hospital from Pfizer, AbbVie, Roche, and Bristol-Myers Squibb for research unrelated to the current study.
SOURCE: Kim SC et al. Arthritis Rheumatol. 2020 Oct 7. doi: 10.1002/art.41550.
Use of febuxostat (Uloric) decreased among patients with gout in the United States following a Food and Drug Administration–mandated black-box warning that cited cardiovascular concerns, but overall use of urate-lowering therapy remained stable, according to data from a study of commercial insurance enrollees in the United States between 2009 and 2019.
Initiation of urate-lowering therapy (ULT) is recommended for gout patients, and allopurinol remains the first-line treatment, but it is not effective in all patients, and febuxostat was developed as an alternative, wrote Seoyoung C. Kim, MD, ScD, of Brigham and Women’s Hospital and Harvard Medical School, Boston, and colleagues.
However, based on data from a postmarketing safety trial (the CARES trial) mandated by the FDA, the agency first issued a safety announcement about an increased risk of cardiovascular mortality with febuxostat in November 2017, followed in February 2019 with a black-box warning after full CARES trial results were published in March 2018 showing a greater risk of cardiovascular and all-cause mortality in febuxostat versus allopurinol.
In a study published in Arthritis & Rheumatology, the researchers examined trends in the use of ULT before and after the FDA warning. They analyzed claims data from a national commercial health database that included 838,432 adult ULT users and 633,229 gout patients.
Overall, allopurinol accounted for the majority of ULT use between 2009 (95% in the first quarter) and 2019 (92% in the fourth quarter).
Febuxostat use peaked at 10% of all ULT use in 2013 and 2014, after a gradual increase following its introduction into the market in 2009, the researchers noted, but decreased to 6% of all ULT use in the fourth quarter of 2019. Other medications, including probenecid, lesinurad (Zurampic), and pegloticase (Krystexxa), accounted for no more than 5% of ULT use.
When the researchers examined gout patients in particular, they found a slight increase in any ULT use from 567 per 1,000 patients in the first quarter of 2009 to 656 per 1,000 patients in the fourth quarter of 2019.
The study findings were limited by several factors, including potential lack of generalizability to other health plans and lack of adjustment for comorbid conditions, the researchers noted.
However, the results highlight the suboptimal use of ULT as a class and the need to address the treatment gap in gout patients “with appropriate ULT prescribing and monitoring,” they said. “While the decrease in febuxostat use was accompanied by a compensatory increase in allopurinol use, the proportion of patients with gout without any ULT remained high throughout the study period,” they concluded.
The study was supported by the division of pharmacoepidemiology and pharmacoeconomics at Brigham and Women’s Hospital. Dr. Kim disclosed receiving research grants to Brigham and Women’s Hospital from Pfizer, AbbVie, Roche, and Bristol-Myers Squibb for research unrelated to the current study.
SOURCE: Kim SC et al. Arthritis Rheumatol. 2020 Oct 7. doi: 10.1002/art.41550.
Use of febuxostat (Uloric) decreased among patients with gout in the United States following a Food and Drug Administration–mandated black-box warning that cited cardiovascular concerns, but overall use of urate-lowering therapy remained stable, according to data from a study of commercial insurance enrollees in the United States between 2009 and 2019.
Initiation of urate-lowering therapy (ULT) is recommended for gout patients, and allopurinol remains the first-line treatment, but it is not effective in all patients, and febuxostat was developed as an alternative, wrote Seoyoung C. Kim, MD, ScD, of Brigham and Women’s Hospital and Harvard Medical School, Boston, and colleagues.
However, based on data from a postmarketing safety trial (the CARES trial) mandated by the FDA, the agency first issued a safety announcement about an increased risk of cardiovascular mortality with febuxostat in November 2017, followed in February 2019 with a black-box warning after full CARES trial results were published in March 2018 showing a greater risk of cardiovascular and all-cause mortality in febuxostat versus allopurinol.
In a study published in Arthritis & Rheumatology, the researchers examined trends in the use of ULT before and after the FDA warning. They analyzed claims data from a national commercial health database that included 838,432 adult ULT users and 633,229 gout patients.
Overall, allopurinol accounted for the majority of ULT use between 2009 (95% in the first quarter) and 2019 (92% in the fourth quarter).
Febuxostat use peaked at 10% of all ULT use in 2013 and 2014, after a gradual increase following its introduction into the market in 2009, the researchers noted, but decreased to 6% of all ULT use in the fourth quarter of 2019. Other medications, including probenecid, lesinurad (Zurampic), and pegloticase (Krystexxa), accounted for no more than 5% of ULT use.
When the researchers examined gout patients in particular, they found a slight increase in any ULT use from 567 per 1,000 patients in the first quarter of 2009 to 656 per 1,000 patients in the fourth quarter of 2019.
The study findings were limited by several factors, including potential lack of generalizability to other health plans and lack of adjustment for comorbid conditions, the researchers noted.
However, the results highlight the suboptimal use of ULT as a class and the need to address the treatment gap in gout patients “with appropriate ULT prescribing and monitoring,” they said. “While the decrease in febuxostat use was accompanied by a compensatory increase in allopurinol use, the proportion of patients with gout without any ULT remained high throughout the study period,” they concluded.
The study was supported by the division of pharmacoepidemiology and pharmacoeconomics at Brigham and Women’s Hospital. Dr. Kim disclosed receiving research grants to Brigham and Women’s Hospital from Pfizer, AbbVie, Roche, and Bristol-Myers Squibb for research unrelated to the current study.
SOURCE: Kim SC et al. Arthritis Rheumatol. 2020 Oct 7. doi: 10.1002/art.41550.
FROM ARTHRITIS & RHEUMATOLOGY
Blood group O linked to decreased risk of SARS-CoV-2 infection
Blood group O was associated with a decreased risk for contracting SARS-CoV-2 infection, according to the results of large retrospective analysis of the Danish population.
Researchers Mike Bogetofte Barnkob, MD, of the Department of Clinical Immunology, Odense (Denmark) University Hospital, and colleagues performed a retrospective cohort analysis of all Danish individuals with a known ABO blood group who were tested for SARS-CoV-2 between Feb. 27, 2020, and July 30, 2020.
Of the 841,327 people tested, ABO and RhD blood groups could be identified for 473,654 individuals. ABO and RhD data from 2,204,742 (38% of the entire Danish population) were used as a reference, according to the online report in Blood Advances.
The primary outcome was status of ABO and RhD blood groups and test results for SARS-CoV-2. The secondary outcomes followed were hospitalization and death from COVID-19.
Reduced prevalence
The study found that ABO blood groups varied significantly between patients and the reference group, with only 38.41% (95% confidence interval, 37.30%-39.50%) of the patients belonging to blood group O, compared with 41.70% (95% CI, 41.60%-41.80%) in the controls, corresponding to a relative risk of 0.87 (95% CI, 0.83-0.91) for acquiring COVID-19.
There was a slight, but statistically significant, difference in blood group distribution between the SARS-CoV-22 individuals and the reference population (P < .001), according to the authors.
Among the SARS-CoV-2 individuals, fewer group O individuals were found (P < .001); while more A, B, and AB individuals were seen (P < .001, P = .011, and P = .091, respectively). There was no significant difference seen among A, B, and AB blood groups (P = .30). The RR for contracting SARS-CoV-2 were 1.09 (95% CI, 1.04-1.14) for A group individuals; 1.06 (95% CI, 0.99-1.14) for B group; and 1.15 (95% CI, 1.03-1.27) for AB group, respectively.
There was no difference found in the RhD group between positive test cases and the reference population (P = .15). In addition, there was no statistical difference (all P > .40) between ABO blood groups and clinical severity of COVID-19 for nonhospitalized patients versus hospitalized patients or for deceased patients versus living patients, the researchers added.
Possible causes
The authors speculated on two possible causes of the lower prevalence of SARS-CoV-2 infection in the blood group O population. The first is that anti-A and anti-B antibodies may have an effect on neutralizing SARS-CoV viruses and that anti-A and anti-B are present on mucosal surfaces in some individuals lacking the corresponding ABO blood group. The second is that the association between ABO blood groups and levels of von Willebrand factor, which is higher in non-O individuals and is tied to an increased likelihood of arterial and venous thrombosis, could have an indirect or unknown impact on susceptibility to infection, according to the authors.
“Given the known increased risk of thrombosis in non-O individuals and the evolving central role for thrombosis in the pathogenesis of COVID-19, it is important to explore this aspect more closely in larger patient cohorts (e.g., by examining ABO blood type and viral load, the severity of symptoms, and the long-term effects following COVID-19),” the researchers concluded.
One author reported receiving fees from Bristol Myers Squibb, Novartis, and Roche. The remaining authors reported they had no competing financial interests.
SOURCE: Barnkob MB et al. Blood Adv. 2020 Oct 14. doi: 10.1182/bloodadvances.2020002657.
Blood group O was associated with a decreased risk for contracting SARS-CoV-2 infection, according to the results of large retrospective analysis of the Danish population.
Researchers Mike Bogetofte Barnkob, MD, of the Department of Clinical Immunology, Odense (Denmark) University Hospital, and colleagues performed a retrospective cohort analysis of all Danish individuals with a known ABO blood group who were tested for SARS-CoV-2 between Feb. 27, 2020, and July 30, 2020.
Of the 841,327 people tested, ABO and RhD blood groups could be identified for 473,654 individuals. ABO and RhD data from 2,204,742 (38% of the entire Danish population) were used as a reference, according to the online report in Blood Advances.
The primary outcome was status of ABO and RhD blood groups and test results for SARS-CoV-2. The secondary outcomes followed were hospitalization and death from COVID-19.
Reduced prevalence
The study found that ABO blood groups varied significantly between patients and the reference group, with only 38.41% (95% confidence interval, 37.30%-39.50%) of the patients belonging to blood group O, compared with 41.70% (95% CI, 41.60%-41.80%) in the controls, corresponding to a relative risk of 0.87 (95% CI, 0.83-0.91) for acquiring COVID-19.
There was a slight, but statistically significant, difference in blood group distribution between the SARS-CoV-22 individuals and the reference population (P < .001), according to the authors.
Among the SARS-CoV-2 individuals, fewer group O individuals were found (P < .001); while more A, B, and AB individuals were seen (P < .001, P = .011, and P = .091, respectively). There was no significant difference seen among A, B, and AB blood groups (P = .30). The RR for contracting SARS-CoV-2 were 1.09 (95% CI, 1.04-1.14) for A group individuals; 1.06 (95% CI, 0.99-1.14) for B group; and 1.15 (95% CI, 1.03-1.27) for AB group, respectively.
There was no difference found in the RhD group between positive test cases and the reference population (P = .15). In addition, there was no statistical difference (all P > .40) between ABO blood groups and clinical severity of COVID-19 for nonhospitalized patients versus hospitalized patients or for deceased patients versus living patients, the researchers added.
Possible causes
The authors speculated on two possible causes of the lower prevalence of SARS-CoV-2 infection in the blood group O population. The first is that anti-A and anti-B antibodies may have an effect on neutralizing SARS-CoV viruses and that anti-A and anti-B are present on mucosal surfaces in some individuals lacking the corresponding ABO blood group. The second is that the association between ABO blood groups and levels of von Willebrand factor, which is higher in non-O individuals and is tied to an increased likelihood of arterial and venous thrombosis, could have an indirect or unknown impact on susceptibility to infection, according to the authors.
“Given the known increased risk of thrombosis in non-O individuals and the evolving central role for thrombosis in the pathogenesis of COVID-19, it is important to explore this aspect more closely in larger patient cohorts (e.g., by examining ABO blood type and viral load, the severity of symptoms, and the long-term effects following COVID-19),” the researchers concluded.
One author reported receiving fees from Bristol Myers Squibb, Novartis, and Roche. The remaining authors reported they had no competing financial interests.
SOURCE: Barnkob MB et al. Blood Adv. 2020 Oct 14. doi: 10.1182/bloodadvances.2020002657.
Blood group O was associated with a decreased risk for contracting SARS-CoV-2 infection, according to the results of large retrospective analysis of the Danish population.
Researchers Mike Bogetofte Barnkob, MD, of the Department of Clinical Immunology, Odense (Denmark) University Hospital, and colleagues performed a retrospective cohort analysis of all Danish individuals with a known ABO blood group who were tested for SARS-CoV-2 between Feb. 27, 2020, and July 30, 2020.
Of the 841,327 people tested, ABO and RhD blood groups could be identified for 473,654 individuals. ABO and RhD data from 2,204,742 (38% of the entire Danish population) were used as a reference, according to the online report in Blood Advances.
The primary outcome was status of ABO and RhD blood groups and test results for SARS-CoV-2. The secondary outcomes followed were hospitalization and death from COVID-19.
Reduced prevalence
The study found that ABO blood groups varied significantly between patients and the reference group, with only 38.41% (95% confidence interval, 37.30%-39.50%) of the patients belonging to blood group O, compared with 41.70% (95% CI, 41.60%-41.80%) in the controls, corresponding to a relative risk of 0.87 (95% CI, 0.83-0.91) for acquiring COVID-19.
There was a slight, but statistically significant, difference in blood group distribution between the SARS-CoV-22 individuals and the reference population (P < .001), according to the authors.
Among the SARS-CoV-2 individuals, fewer group O individuals were found (P < .001); while more A, B, and AB individuals were seen (P < .001, P = .011, and P = .091, respectively). There was no significant difference seen among A, B, and AB blood groups (P = .30). The RR for contracting SARS-CoV-2 were 1.09 (95% CI, 1.04-1.14) for A group individuals; 1.06 (95% CI, 0.99-1.14) for B group; and 1.15 (95% CI, 1.03-1.27) for AB group, respectively.
There was no difference found in the RhD group between positive test cases and the reference population (P = .15). In addition, there was no statistical difference (all P > .40) between ABO blood groups and clinical severity of COVID-19 for nonhospitalized patients versus hospitalized patients or for deceased patients versus living patients, the researchers added.
Possible causes
The authors speculated on two possible causes of the lower prevalence of SARS-CoV-2 infection in the blood group O population. The first is that anti-A and anti-B antibodies may have an effect on neutralizing SARS-CoV viruses and that anti-A and anti-B are present on mucosal surfaces in some individuals lacking the corresponding ABO blood group. The second is that the association between ABO blood groups and levels of von Willebrand factor, which is higher in non-O individuals and is tied to an increased likelihood of arterial and venous thrombosis, could have an indirect or unknown impact on susceptibility to infection, according to the authors.
“Given the known increased risk of thrombosis in non-O individuals and the evolving central role for thrombosis in the pathogenesis of COVID-19, it is important to explore this aspect more closely in larger patient cohorts (e.g., by examining ABO blood type and viral load, the severity of symptoms, and the long-term effects following COVID-19),” the researchers concluded.
One author reported receiving fees from Bristol Myers Squibb, Novartis, and Roche. The remaining authors reported they had no competing financial interests.
SOURCE: Barnkob MB et al. Blood Adv. 2020 Oct 14. doi: 10.1182/bloodadvances.2020002657.
FROM BLOOD ADVANCES
Rapidly developing vesicular eruption
A 23-month-old girl with a history of well-controlled atopic dermatitis was admitted to the hospital with fever and a widespread vesicular eruption of 2 days’ duration. Two days prior to admission, the patient had 3 episodes of nonbloody diarrhea and redness in the diaper area. The child’s parents reported that the red areas spread to her arms and legs later that day, and that she subsequently developed a fever, cough, and rhinorrhea. She was taken to an urgent care facility where she was diagnosed with vulvovaginitis and an upper respiratory infection; amoxicillin was prescribed. Shortly thereafter, the patient developed more lesions in and around the mouth, as well as on the trunk, prompting the parents to bring her to the emergency department.
The history revealed that the patient had spent time with her aunt and cousins who had “red spots” on their palms and soles. The patient’s sister had a flare of “cold sores,” about 2 weeks prior to the current presentation. The patient had received a varicella zoster virus (VZV) vaccine several months earlier.
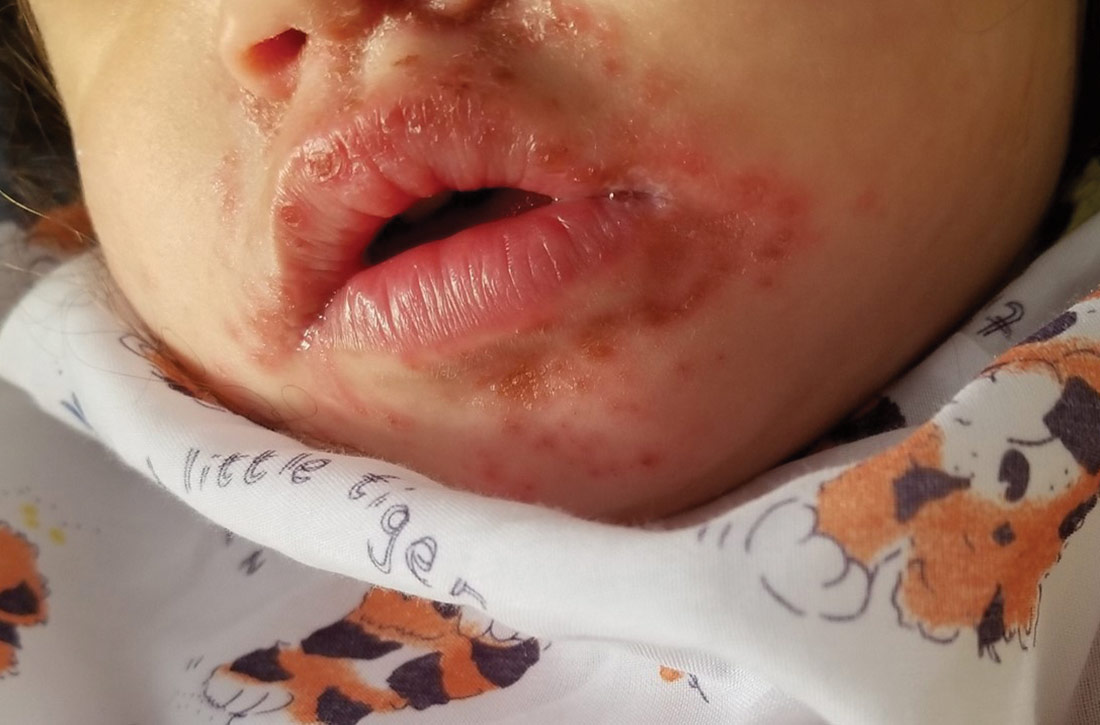
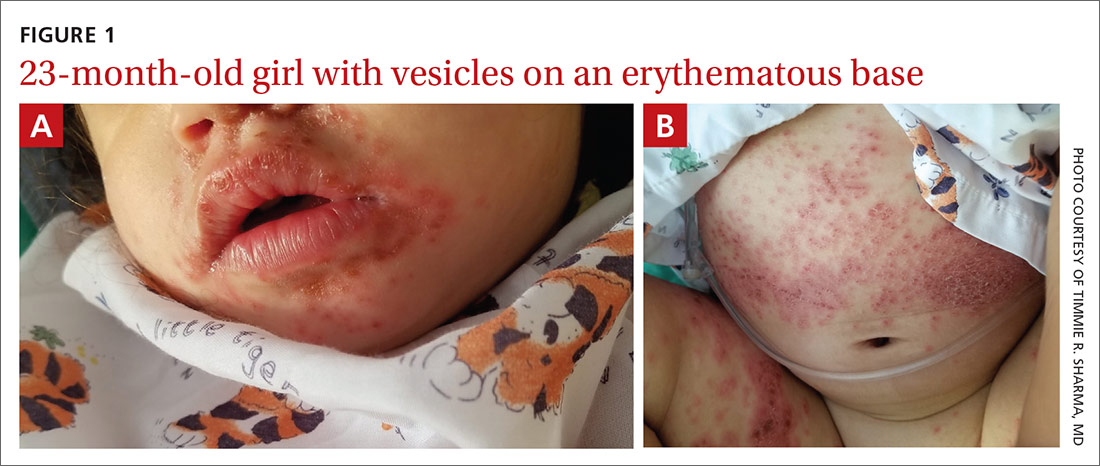
Physical examination was notable for an uncomfortable infant with erythematous macules on the bilateral palms and soles and an erythematous hard palate. The child also had scattered vesicles on an erythematous base with confluent crusted plaques on her lips, perioral skin (FIGURE 1A), abdomen, back, buttocks, arms, legs (FIGURE 1B), and dorsal aspects of her hands and feet.
WHAT IS YOUR DIAGNOSIS?
HOW WOULD YOU TREAT THIS PATIENT?
Diagnosis: Eczema coxsackium
Given the history of atopic dermatitis; prodromal diarrhea/rhinorrhea; papulovesicular eruption involving areas of prior dermatitis as well as the palms, soles, and mouth; recent contacts with suspected hand-foot-mouth disease (HFMD); and history of VZV vaccination, the favored diagnosis was eczema coxsackium.
Eczema coxsackium is an atypical form of HFMD that occurs in patients with a history of eczema. Classic HFMD usually is caused by coxsackievirus A16 or enterovirus 71, while atypical HFMD often is caused by coxsackievirus A6.1,2,3 Patients with HFMD present with painful oral vesicles and ulcers and a papulovesicular eruption on the palms, soles, and sometimes the buttocks and genitalia. Patients may have prodromal fever, fussiness, and diarrhea. Painful oral lesions may result in poor oral intake.1,2
Differential includes viral eruptions
Other conditions may manifest similarly to eczema coxsackium and must be ruled out before initiating proper treatment.
Eczema herpeticum (EH). In atypical HFMD, the virus can show tropism for active or previously inflamed areas of eczematous skin, leading to a widespread vesicular eruption, which can be difficult to distinguish from EH.1 Similar to EH, eczema coxsackium does not exclusively affect children with atopic dermatitis. It also has been described in adults and patients with Darier disease, incontinentia pigmenti, and epidermolytic ichthyosis.4-6
In cases of vesicular eruptions in eczema patients, it is imperative to rule out EH. One prospective study of atypical HFMD compared similarities of the conditions. Both have a predilection for mucosa during primary infection and develop vesicular eruptions on cutaneous eczematous skin.1 One key difference between eczema coxsackium and EH is that EH tends to produce intraoral vesicles beyond simple erythema; it also tends to predominate in the area of the head and neck.7
Continue to: Eczema varicellicum
Eczema varicellicum has been reported, and it has been suggested that some cases of EH may actually be caused by VZV as the 2 are clinically indistinguishable and less than half of EH cases are diagnosed with laboratory confirmation.8
Confirm Dx before you treat
To guide management, cases of suspected eczema coxsackium should be confirmed, and HSV/VZV should be ruled out.9 Testing modalities include swabbing vesicular fluid for enterovirus polymerase chain reaction (PCR) analysis (preferred modality), oropharyngeal swab up to 2 weeks after infection, or viral isolate from stool samples up to 3 months after infection.2,3
Treatment for eczema coxsackium involves supportive care such as intravenous (IV) hydration and antipyretics. Some studies show potential benefit with IV immunoglobulin in treating severe HFMD, while other studies show the exacerbation of widespread HFMD with this treatment.7,10
Prompt diagnosis and treatment for eczema coxsackium is critical to prevent unnecessary antiviral therapy and to help guide monitoring for associated morbidities including Gianotti-Crosti syndrome–like eruptions, purpuric eruptions, and onychomadesis.
Our patient. Because EH was in the differential, our patient was started on empiric IV acyclovir 10 mg/kg every 8 hours while test results were pending. In addition, she received acetaminophen, IV fluids, gentle sponge baths, and diligent emollient application. Scraping from a vesicle revealed negative herpes simplex virus 1/2 PCR, negative VZV direct fluorescent antibody, and a positive enterovirus PCR—confirming the diagnosis of eczema coxsackium. Interestingly, a viral culture was negative in our patient, consistent with prior reports of enterovirus being difficult to culture.11
With confirmation of the diagnosis of eczema coxsackium, the IV acyclovir was discontinued, and symptoms resolved after 7 days.
CORRESPONDENCE
Shane M. Swink, DO, MS, Division of Dermatology, 1200 South Cedar Crest Boulevard, Allentown, PA 18103; [email protected]
1. Neri I, Dondi A, Wollenberg A, et al. Atypical forms of hand, foot, and mouth disease: a prospective study of 47 Italian children. Pediatr Dermatol. 2016;33:429-437.
2. Nassef C, Ziemer C, Morrell DS. Hand-foot-and-mouth disease: a new look at a classic viral rash. Curr Opin Pediatr. 2015;27:486-491.
3. Horsten H, Fisker N, Bygu, A. Eczema coxsackium caused by coxsackievirus A6. Pediatr Dermatol. 2016;33:230-231.
4. Jefferson J, Grossberg A. Incontinentia pigmenti coxsackium. Pediatr Dermatol. 2016;33:E280-E281.
5. Ganguly S, Kuruvila S. Eczema coxsackium. Indian J Dermatol. 2016;61:682-683.
6. Harris P, Wang AD, Yin M, et al. Atypical hand, foot, and mouth disease: eczema coxsackium can also occur in adults. Lancet Infect Dis. 2014;14:1043.
7. Wollenberg A, Zoch C, Wetzel S, et al. Predisposing factors and clinical features of eczema herpeticum: a retrospective analysis of 100 cases. J Am Acad Dermatol. 2003;49:198-205.
8. Austin TA, Steele RW. Eczema varicella/zoster (varicellicum). Clin Pediatr. 2017;56:579-581.
9. Leung DYM. Why is eczema herpeticum unexpectedly rare? Antiviral Res. 2013;98:153-157.
10. Cao RY, Dong DY, Liu RJ, et al. Human IgG subclasses against enterovirus type 71: neutralization versus antibody dependent enhancement of infection. PLoS One. 2013;8:E64024.
11. Mathes EF, Oza V, Frieden IJ, et al. Eczema coxsackium and unusual cutaneous findings in an enterovirus outbreak. Pediatrics. 2013;132:149-157.
A 23-month-old girl with a history of well-controlled atopic dermatitis was admitted to the hospital with fever and a widespread vesicular eruption of 2 days’ duration. Two days prior to admission, the patient had 3 episodes of nonbloody diarrhea and redness in the diaper area. The child’s parents reported that the red areas spread to her arms and legs later that day, and that she subsequently developed a fever, cough, and rhinorrhea. She was taken to an urgent care facility where she was diagnosed with vulvovaginitis and an upper respiratory infection; amoxicillin was prescribed. Shortly thereafter, the patient developed more lesions in and around the mouth, as well as on the trunk, prompting the parents to bring her to the emergency department.
The history revealed that the patient had spent time with her aunt and cousins who had “red spots” on their palms and soles. The patient’s sister had a flare of “cold sores,” about 2 weeks prior to the current presentation. The patient had received a varicella zoster virus (VZV) vaccine several months earlier.
Physical examination was notable for an uncomfortable infant with erythematous macules on the bilateral palms and soles and an erythematous hard palate. The child also had scattered vesicles on an erythematous base with confluent crusted plaques on her lips, perioral skin (FIGURE 1A), abdomen, back, buttocks, arms, legs (FIGURE 1B), and dorsal aspects of her hands and feet.

WHAT IS YOUR DIAGNOSIS?
HOW WOULD YOU TREAT THIS PATIENT?
Diagnosis: Eczema coxsackium
Given the history of atopic dermatitis; prodromal diarrhea/rhinorrhea; papulovesicular eruption involving areas of prior dermatitis as well as the palms, soles, and mouth; recent contacts with suspected hand-foot-mouth disease (HFMD); and history of VZV vaccination, the favored diagnosis was eczema coxsackium.
Eczema coxsackium is an atypical form of HFMD that occurs in patients with a history of eczema. Classic HFMD usually is caused by coxsackievirus A16 or enterovirus 71, while atypical HFMD often is caused by coxsackievirus A6.1,2,3 Patients with HFMD present with painful oral vesicles and ulcers and a papulovesicular eruption on the palms, soles, and sometimes the buttocks and genitalia. Patients may have prodromal fever, fussiness, and diarrhea. Painful oral lesions may result in poor oral intake.1,2
Differential includes viral eruptions
Other conditions may manifest similarly to eczema coxsackium and must be ruled out before initiating proper treatment.
Eczema herpeticum (EH). In atypical HFMD, the virus can show tropism for active or previously inflamed areas of eczematous skin, leading to a widespread vesicular eruption, which can be difficult to distinguish from EH.1 Similar to EH, eczema coxsackium does not exclusively affect children with atopic dermatitis. It also has been described in adults and patients with Darier disease, incontinentia pigmenti, and epidermolytic ichthyosis.4-6
In cases of vesicular eruptions in eczema patients, it is imperative to rule out EH. One prospective study of atypical HFMD compared similarities of the conditions. Both have a predilection for mucosa during primary infection and develop vesicular eruptions on cutaneous eczematous skin.1 One key difference between eczema coxsackium and EH is that EH tends to produce intraoral vesicles beyond simple erythema; it also tends to predominate in the area of the head and neck.7
Continue to: Eczema varicellicum
Eczema varicellicum has been reported, and it has been suggested that some cases of EH may actually be caused by VZV as the 2 are clinically indistinguishable and less than half of EH cases are diagnosed with laboratory confirmation.8
Confirm Dx before you treat
To guide management, cases of suspected eczema coxsackium should be confirmed, and HSV/VZV should be ruled out.9 Testing modalities include swabbing vesicular fluid for enterovirus polymerase chain reaction (PCR) analysis (preferred modality), oropharyngeal swab up to 2 weeks after infection, or viral isolate from stool samples up to 3 months after infection.2,3
Treatment for eczema coxsackium involves supportive care such as intravenous (IV) hydration and antipyretics. Some studies show potential benefit with IV immunoglobulin in treating severe HFMD, while other studies show the exacerbation of widespread HFMD with this treatment.7,10
Prompt diagnosis and treatment for eczema coxsackium is critical to prevent unnecessary antiviral therapy and to help guide monitoring for associated morbidities including Gianotti-Crosti syndrome–like eruptions, purpuric eruptions, and onychomadesis.
Our patient. Because EH was in the differential, our patient was started on empiric IV acyclovir 10 mg/kg every 8 hours while test results were pending. In addition, she received acetaminophen, IV fluids, gentle sponge baths, and diligent emollient application. Scraping from a vesicle revealed negative herpes simplex virus 1/2 PCR, negative VZV direct fluorescent antibody, and a positive enterovirus PCR—confirming the diagnosis of eczema coxsackium. Interestingly, a viral culture was negative in our patient, consistent with prior reports of enterovirus being difficult to culture.11
With confirmation of the diagnosis of eczema coxsackium, the IV acyclovir was discontinued, and symptoms resolved after 7 days.
CORRESPONDENCE
Shane M. Swink, DO, MS, Division of Dermatology, 1200 South Cedar Crest Boulevard, Allentown, PA 18103; [email protected]
A 23-month-old girl with a history of well-controlled atopic dermatitis was admitted to the hospital with fever and a widespread vesicular eruption of 2 days’ duration. Two days prior to admission, the patient had 3 episodes of nonbloody diarrhea and redness in the diaper area. The child’s parents reported that the red areas spread to her arms and legs later that day, and that she subsequently developed a fever, cough, and rhinorrhea. She was taken to an urgent care facility where she was diagnosed with vulvovaginitis and an upper respiratory infection; amoxicillin was prescribed. Shortly thereafter, the patient developed more lesions in and around the mouth, as well as on the trunk, prompting the parents to bring her to the emergency department.
The history revealed that the patient had spent time with her aunt and cousins who had “red spots” on their palms and soles. The patient’s sister had a flare of “cold sores,” about 2 weeks prior to the current presentation. The patient had received a varicella zoster virus (VZV) vaccine several months earlier.
Physical examination was notable for an uncomfortable infant with erythematous macules on the bilateral palms and soles and an erythematous hard palate. The child also had scattered vesicles on an erythematous base with confluent crusted plaques on her lips, perioral skin (FIGURE 1A), abdomen, back, buttocks, arms, legs (FIGURE 1B), and dorsal aspects of her hands and feet.

WHAT IS YOUR DIAGNOSIS?
HOW WOULD YOU TREAT THIS PATIENT?
Diagnosis: Eczema coxsackium
Given the history of atopic dermatitis; prodromal diarrhea/rhinorrhea; papulovesicular eruption involving areas of prior dermatitis as well as the palms, soles, and mouth; recent contacts with suspected hand-foot-mouth disease (HFMD); and history of VZV vaccination, the favored diagnosis was eczema coxsackium.
Eczema coxsackium is an atypical form of HFMD that occurs in patients with a history of eczema. Classic HFMD usually is caused by coxsackievirus A16 or enterovirus 71, while atypical HFMD often is caused by coxsackievirus A6.1,2,3 Patients with HFMD present with painful oral vesicles and ulcers and a papulovesicular eruption on the palms, soles, and sometimes the buttocks and genitalia. Patients may have prodromal fever, fussiness, and diarrhea. Painful oral lesions may result in poor oral intake.1,2
Differential includes viral eruptions
Other conditions may manifest similarly to eczema coxsackium and must be ruled out before initiating proper treatment.
Eczema herpeticum (EH). In atypical HFMD, the virus can show tropism for active or previously inflamed areas of eczematous skin, leading to a widespread vesicular eruption, which can be difficult to distinguish from EH.1 Similar to EH, eczema coxsackium does not exclusively affect children with atopic dermatitis. It also has been described in adults and patients with Darier disease, incontinentia pigmenti, and epidermolytic ichthyosis.4-6
In cases of vesicular eruptions in eczema patients, it is imperative to rule out EH. One prospective study of atypical HFMD compared similarities of the conditions. Both have a predilection for mucosa during primary infection and develop vesicular eruptions on cutaneous eczematous skin.1 One key difference between eczema coxsackium and EH is that EH tends to produce intraoral vesicles beyond simple erythema; it also tends to predominate in the area of the head and neck.7
Continue to: Eczema varicellicum
Eczema varicellicum has been reported, and it has been suggested that some cases of EH may actually be caused by VZV as the 2 are clinically indistinguishable and less than half of EH cases are diagnosed with laboratory confirmation.8
Confirm Dx before you treat
To guide management, cases of suspected eczema coxsackium should be confirmed, and HSV/VZV should be ruled out.9 Testing modalities include swabbing vesicular fluid for enterovirus polymerase chain reaction (PCR) analysis (preferred modality), oropharyngeal swab up to 2 weeks after infection, or viral isolate from stool samples up to 3 months after infection.2,3
Treatment for eczema coxsackium involves supportive care such as intravenous (IV) hydration and antipyretics. Some studies show potential benefit with IV immunoglobulin in treating severe HFMD, while other studies show the exacerbation of widespread HFMD with this treatment.7,10
Prompt diagnosis and treatment for eczema coxsackium is critical to prevent unnecessary antiviral therapy and to help guide monitoring for associated morbidities including Gianotti-Crosti syndrome–like eruptions, purpuric eruptions, and onychomadesis.
Our patient. Because EH was in the differential, our patient was started on empiric IV acyclovir 10 mg/kg every 8 hours while test results were pending. In addition, she received acetaminophen, IV fluids, gentle sponge baths, and diligent emollient application. Scraping from a vesicle revealed negative herpes simplex virus 1/2 PCR, negative VZV direct fluorescent antibody, and a positive enterovirus PCR—confirming the diagnosis of eczema coxsackium. Interestingly, a viral culture was negative in our patient, consistent with prior reports of enterovirus being difficult to culture.11
With confirmation of the diagnosis of eczema coxsackium, the IV acyclovir was discontinued, and symptoms resolved after 7 days.
CORRESPONDENCE
Shane M. Swink, DO, MS, Division of Dermatology, 1200 South Cedar Crest Boulevard, Allentown, PA 18103; [email protected]
1. Neri I, Dondi A, Wollenberg A, et al. Atypical forms of hand, foot, and mouth disease: a prospective study of 47 Italian children. Pediatr Dermatol. 2016;33:429-437.
2. Nassef C, Ziemer C, Morrell DS. Hand-foot-and-mouth disease: a new look at a classic viral rash. Curr Opin Pediatr. 2015;27:486-491.
3. Horsten H, Fisker N, Bygu, A. Eczema coxsackium caused by coxsackievirus A6. Pediatr Dermatol. 2016;33:230-231.
4. Jefferson J, Grossberg A. Incontinentia pigmenti coxsackium. Pediatr Dermatol. 2016;33:E280-E281.
5. Ganguly S, Kuruvila S. Eczema coxsackium. Indian J Dermatol. 2016;61:682-683.
6. Harris P, Wang AD, Yin M, et al. Atypical hand, foot, and mouth disease: eczema coxsackium can also occur in adults. Lancet Infect Dis. 2014;14:1043.
7. Wollenberg A, Zoch C, Wetzel S, et al. Predisposing factors and clinical features of eczema herpeticum: a retrospective analysis of 100 cases. J Am Acad Dermatol. 2003;49:198-205.
8. Austin TA, Steele RW. Eczema varicella/zoster (varicellicum). Clin Pediatr. 2017;56:579-581.
9. Leung DYM. Why is eczema herpeticum unexpectedly rare? Antiviral Res. 2013;98:153-157.
10. Cao RY, Dong DY, Liu RJ, et al. Human IgG subclasses against enterovirus type 71: neutralization versus antibody dependent enhancement of infection. PLoS One. 2013;8:E64024.
11. Mathes EF, Oza V, Frieden IJ, et al. Eczema coxsackium and unusual cutaneous findings in an enterovirus outbreak. Pediatrics. 2013;132:149-157.
1. Neri I, Dondi A, Wollenberg A, et al. Atypical forms of hand, foot, and mouth disease: a prospective study of 47 Italian children. Pediatr Dermatol. 2016;33:429-437.
2. Nassef C, Ziemer C, Morrell DS. Hand-foot-and-mouth disease: a new look at a classic viral rash. Curr Opin Pediatr. 2015;27:486-491.
3. Horsten H, Fisker N, Bygu, A. Eczema coxsackium caused by coxsackievirus A6. Pediatr Dermatol. 2016;33:230-231.
4. Jefferson J, Grossberg A. Incontinentia pigmenti coxsackium. Pediatr Dermatol. 2016;33:E280-E281.
5. Ganguly S, Kuruvila S. Eczema coxsackium. Indian J Dermatol. 2016;61:682-683.
6. Harris P, Wang AD, Yin M, et al. Atypical hand, foot, and mouth disease: eczema coxsackium can also occur in adults. Lancet Infect Dis. 2014;14:1043.
7. Wollenberg A, Zoch C, Wetzel S, et al. Predisposing factors and clinical features of eczema herpeticum: a retrospective analysis of 100 cases. J Am Acad Dermatol. 2003;49:198-205.
8. Austin TA, Steele RW. Eczema varicella/zoster (varicellicum). Clin Pediatr. 2017;56:579-581.
9. Leung DYM. Why is eczema herpeticum unexpectedly rare? Antiviral Res. 2013;98:153-157.
10. Cao RY, Dong DY, Liu RJ, et al. Human IgG subclasses against enterovirus type 71: neutralization versus antibody dependent enhancement of infection. PLoS One. 2013;8:E64024.
11. Mathes EF, Oza V, Frieden IJ, et al. Eczema coxsackium and unusual cutaneous findings in an enterovirus outbreak. Pediatrics. 2013;132:149-157.
Dual therapy serves as well as triple for most HIV patients
based on a meta-analysis including data from more than 5,000 patients.
Although triple therapy remains the standard of care, the availability of more potent drugs has revived interest in dual and mono therapies, wrote Pisaturo Mariantonietta, MD, of the University of Campania Luigi Vanvitelli, Naples, Italy, and colleagues.
In a study published in Clinical Microbiology and Infection, the researchers identified 14 articles including 5,205 treatment-naive HIV adults. The studies were published between 2008 and 2020; 13 were randomized, controlled trials.
The dual therapies used in the studies included atazanavir/r plus maraviroc; lopinavir/r plus lamivudine; raltegravir plus darunavir/r; lopinavir/r plus tenofovir, raltegravir, efavirenz, or maraviroc; atazanavir/r plus raltegravir and darunavir/r plus maraviroc; and dolutegravir plus lamivudine.
Overall, no significant differences occurred in the primary endpoint of treatment failure across 10 studies between dual therapy and triple therapy patients based on data at 48 weeks (relative risk 1.20). “The rate of treatment failure did not differ among the two groups when stratifying the patients according to the drug used in the dual regimen,” the researchers said.
Low viral load’s link to treatment failure
Among 2,398 patients with a low HIV viral load (less than 100,000 copies/mL), dual therapy patients were significantly more likely to experience treatment failure than were triple therapy patients (RR, 1.47, P = .007). No differences were noted between dual and triple therapy failure among patients with high HIV viral loads at baseline. Patterns were similar at 96 weeks, but only three studies included 96-week data, the researchers said.
The rate of discontinuation because of adverse events was not significantly different between the groups at 48 weeks.
The study findings were limited by several factors, including the use of different regimens in the dual strategies, some of which are no longer in use, as well as there being insufficient data to fully compare outcomes at 96 weeks, and lack of information on cerebrospinal fluid viral load, the researchers noted.
However, the results suggest that dual therapy might be considered for HIV-naive patients with a low viral load, they said.
“Further RCTs that will evaluate the efficacy of antiretroviral regimens in use today among difficult-to-treat populations, such as patients with high viral load, including both intention-to-treat and per-protocol analysis, are needed to address this topic,” they concluded.
Consider range of patient factors when choosing therapies
Conducting the study at this time was important because of the expanding options for treating HIV patients, Donna E. Sweet, MD, an HIV specialist and professor of medicine at the University of Kansas, Wichita, said in an interview.
“We now have two single tablet formulations that are dual rather than triple therapy, and as treaters we are all trying to know when to use them,” she explained.
Dr. Sweet said she was not surprised by the study findings, given that well-conducted, randomized, controlled trials allowed the combination therapies to be approved.
Some of the key challenges to identifying the optimal treatment for HIV patients include factoring in the use of concomitant medications that could lead to drug-drug interactions, noted Dr. Sweet, who serves an editorial advisory board member of Internal Medicine News.
The take-home message for clinicians, in her opinion, is that “less drugs may mean less toxicity, but we don’t want to sacrifice efficacy,” she said. “There may be patients who are better suited than others for two vs. three drugs,” Dr. Sweet emphasized.
The next steps for research on the value of dual vs. triple therapy should include longer term efficacy studies, especially in those with lower CD4 counts and higher viral loads, said Dr. Sweet. In addition to factors such as CD4 counts and viral load, the food requirements of certain ART regimens could affect adherence and therefore a clinician decision to use two drugs rather than three, she noted.
Dr. Sweet disclosed past relationships with ViiV, Gilead, Merck, and Janssen on their speakers bureaus, and current advisory roles with Gilead and ViiV.
The study received no outside funding. Lead author Dr. Mariantonietta and several coauthors disclosed relationships with companies including ViiV Healthcare, AbbVie, Janssen-Cilag and Gilead Science, and Merck Sharp & Dohme, but no conflicts in connection with this study.
SOURCE: Mariantonietta P et al. Clin Microbiol Infect. 2020 Oct 5. doi: 10.1016/j.cmi.2020.09.048.
based on a meta-analysis including data from more than 5,000 patients.
Although triple therapy remains the standard of care, the availability of more potent drugs has revived interest in dual and mono therapies, wrote Pisaturo Mariantonietta, MD, of the University of Campania Luigi Vanvitelli, Naples, Italy, and colleagues.
In a study published in Clinical Microbiology and Infection, the researchers identified 14 articles including 5,205 treatment-naive HIV adults. The studies were published between 2008 and 2020; 13 were randomized, controlled trials.
The dual therapies used in the studies included atazanavir/r plus maraviroc; lopinavir/r plus lamivudine; raltegravir plus darunavir/r; lopinavir/r plus tenofovir, raltegravir, efavirenz, or maraviroc; atazanavir/r plus raltegravir and darunavir/r plus maraviroc; and dolutegravir plus lamivudine.
Overall, no significant differences occurred in the primary endpoint of treatment failure across 10 studies between dual therapy and triple therapy patients based on data at 48 weeks (relative risk 1.20). “The rate of treatment failure did not differ among the two groups when stratifying the patients according to the drug used in the dual regimen,” the researchers said.
Low viral load’s link to treatment failure
Among 2,398 patients with a low HIV viral load (less than 100,000 copies/mL), dual therapy patients were significantly more likely to experience treatment failure than were triple therapy patients (RR, 1.47, P = .007). No differences were noted between dual and triple therapy failure among patients with high HIV viral loads at baseline. Patterns were similar at 96 weeks, but only three studies included 96-week data, the researchers said.
The rate of discontinuation because of adverse events was not significantly different between the groups at 48 weeks.
The study findings were limited by several factors, including the use of different regimens in the dual strategies, some of which are no longer in use, as well as there being insufficient data to fully compare outcomes at 96 weeks, and lack of information on cerebrospinal fluid viral load, the researchers noted.
However, the results suggest that dual therapy might be considered for HIV-naive patients with a low viral load, they said.
“Further RCTs that will evaluate the efficacy of antiretroviral regimens in use today among difficult-to-treat populations, such as patients with high viral load, including both intention-to-treat and per-protocol analysis, are needed to address this topic,” they concluded.
Consider range of patient factors when choosing therapies
Conducting the study at this time was important because of the expanding options for treating HIV patients, Donna E. Sweet, MD, an HIV specialist and professor of medicine at the University of Kansas, Wichita, said in an interview.
“We now have two single tablet formulations that are dual rather than triple therapy, and as treaters we are all trying to know when to use them,” she explained.
Dr. Sweet said she was not surprised by the study findings, given that well-conducted, randomized, controlled trials allowed the combination therapies to be approved.
Some of the key challenges to identifying the optimal treatment for HIV patients include factoring in the use of concomitant medications that could lead to drug-drug interactions, noted Dr. Sweet, who serves an editorial advisory board member of Internal Medicine News.
The take-home message for clinicians, in her opinion, is that “less drugs may mean less toxicity, but we don’t want to sacrifice efficacy,” she said. “There may be patients who are better suited than others for two vs. three drugs,” Dr. Sweet emphasized.
The next steps for research on the value of dual vs. triple therapy should include longer term efficacy studies, especially in those with lower CD4 counts and higher viral loads, said Dr. Sweet. In addition to factors such as CD4 counts and viral load, the food requirements of certain ART regimens could affect adherence and therefore a clinician decision to use two drugs rather than three, she noted.
Dr. Sweet disclosed past relationships with ViiV, Gilead, Merck, and Janssen on their speakers bureaus, and current advisory roles with Gilead and ViiV.
The study received no outside funding. Lead author Dr. Mariantonietta and several coauthors disclosed relationships with companies including ViiV Healthcare, AbbVie, Janssen-Cilag and Gilead Science, and Merck Sharp & Dohme, but no conflicts in connection with this study.
SOURCE: Mariantonietta P et al. Clin Microbiol Infect. 2020 Oct 5. doi: 10.1016/j.cmi.2020.09.048.
based on a meta-analysis including data from more than 5,000 patients.
Although triple therapy remains the standard of care, the availability of more potent drugs has revived interest in dual and mono therapies, wrote Pisaturo Mariantonietta, MD, of the University of Campania Luigi Vanvitelli, Naples, Italy, and colleagues.
In a study published in Clinical Microbiology and Infection, the researchers identified 14 articles including 5,205 treatment-naive HIV adults. The studies were published between 2008 and 2020; 13 were randomized, controlled trials.
The dual therapies used in the studies included atazanavir/r plus maraviroc; lopinavir/r plus lamivudine; raltegravir plus darunavir/r; lopinavir/r plus tenofovir, raltegravir, efavirenz, or maraviroc; atazanavir/r plus raltegravir and darunavir/r plus maraviroc; and dolutegravir plus lamivudine.
Overall, no significant differences occurred in the primary endpoint of treatment failure across 10 studies between dual therapy and triple therapy patients based on data at 48 weeks (relative risk 1.20). “The rate of treatment failure did not differ among the two groups when stratifying the patients according to the drug used in the dual regimen,” the researchers said.
Low viral load’s link to treatment failure
Among 2,398 patients with a low HIV viral load (less than 100,000 copies/mL), dual therapy patients were significantly more likely to experience treatment failure than were triple therapy patients (RR, 1.47, P = .007). No differences were noted between dual and triple therapy failure among patients with high HIV viral loads at baseline. Patterns were similar at 96 weeks, but only three studies included 96-week data, the researchers said.
The rate of discontinuation because of adverse events was not significantly different between the groups at 48 weeks.
The study findings were limited by several factors, including the use of different regimens in the dual strategies, some of which are no longer in use, as well as there being insufficient data to fully compare outcomes at 96 weeks, and lack of information on cerebrospinal fluid viral load, the researchers noted.
However, the results suggest that dual therapy might be considered for HIV-naive patients with a low viral load, they said.
“Further RCTs that will evaluate the efficacy of antiretroviral regimens in use today among difficult-to-treat populations, such as patients with high viral load, including both intention-to-treat and per-protocol analysis, are needed to address this topic,” they concluded.
Consider range of patient factors when choosing therapies
Conducting the study at this time was important because of the expanding options for treating HIV patients, Donna E. Sweet, MD, an HIV specialist and professor of medicine at the University of Kansas, Wichita, said in an interview.
“We now have two single tablet formulations that are dual rather than triple therapy, and as treaters we are all trying to know when to use them,” she explained.
Dr. Sweet said she was not surprised by the study findings, given that well-conducted, randomized, controlled trials allowed the combination therapies to be approved.
Some of the key challenges to identifying the optimal treatment for HIV patients include factoring in the use of concomitant medications that could lead to drug-drug interactions, noted Dr. Sweet, who serves an editorial advisory board member of Internal Medicine News.
The take-home message for clinicians, in her opinion, is that “less drugs may mean less toxicity, but we don’t want to sacrifice efficacy,” she said. “There may be patients who are better suited than others for two vs. three drugs,” Dr. Sweet emphasized.
The next steps for research on the value of dual vs. triple therapy should include longer term efficacy studies, especially in those with lower CD4 counts and higher viral loads, said Dr. Sweet. In addition to factors such as CD4 counts and viral load, the food requirements of certain ART regimens could affect adherence and therefore a clinician decision to use two drugs rather than three, she noted.
Dr. Sweet disclosed past relationships with ViiV, Gilead, Merck, and Janssen on their speakers bureaus, and current advisory roles with Gilead and ViiV.
The study received no outside funding. Lead author Dr. Mariantonietta and several coauthors disclosed relationships with companies including ViiV Healthcare, AbbVie, Janssen-Cilag and Gilead Science, and Merck Sharp & Dohme, but no conflicts in connection with this study.
SOURCE: Mariantonietta P et al. Clin Microbiol Infect. 2020 Oct 5. doi: 10.1016/j.cmi.2020.09.048.
FROM CLINICAL MICROBIOLOGY AND INFECTION
Early hearing impairment interventions key to kindergarten readiness
Starting early intervention (EI) enrollment before age 6 months in children who are deaf or hard of hearing may have a lasting influence on ensuring kindergarten readiness, Jareen Meinzen-Derr, PhD, MPH of Cincinnati Children’s Hospital Medical Center and colleagues reported in Pediatrics.
The researchers created a comprehensive, longitudinal, population-based database, which linked hearing screening and diagnostic data to that of early intervention data and educational records for 1,746 infants identified with permanent hearing loss who were born between Jan. 1, 2008 and Dec. 31, 2014 The database was established in partnership with the Ohio Departments of Health, Developmental Disabilities and Education, and with the support of the Centers for Disease Control and Prevention and the National Center on Birth Defects and Developmental Disabilities.
Of those, 784 children ranging from preschool to fourth grade were evaluated based on education data available for the 2017 and 2018 school year that had been linked by way of an identifier that flagged students enrolled in EI.
All together, 417 students had kindergarten assessment records, and of those, 385 had Kindergarten Readiness Assessments (KRAs) between 2014 and 2018; 222 (58%) had been enrolled in EI before the age of 6 months. Of those who were enrolled early, the median age of EI enrollment was 3.4 months (2.4-4.3 months) and in those enrolled later, the median age was 9.2 months (7.5-15.4 months).
The importance of EI prior to 6 months
A total of 109 children (28%) receiving services as part of Ohio’s early intervention programs demonstrated kindergarten readiness on their overall KRA scores. The scores revealed that children receiving EI early (34%, n = 75) were more likely to be ready for kindergarten than were those who entered later (21%, n = 34; P = .005). They also were more likely to have on track language and literacy scores (60% vs. 42%, respectively; P = .0006).
Dr. Meinzen-Derr and colleagues noted that factors identified with “an increased odds of being on track included having private insurance and some college education for the mother.” Conversely, factors identified with a decreased likelihood included having a diagnosed disability and bilateral hearing loss.
The researchers cautioned that children transitioned from EI to academic settings will face challenges that may go underrecognized because a school’s focus often is largely on social and academic performance. Thus, working with linked data systems can provide the data to track outcomes that might otherwise be missed, the researchers noted.
Furthermore, they cautioned that even though kindergarten readiness offers some glimpse into future academic success, these measures alone may not be sufficient predictors for children who are deaf or hard of hearing. Risk for communication, social, and academic delays persist throughout school so it is important to employ alternative methods of reading instruction in order to “achieve more complex skills (e.g., complex syntax and advanced vocabulary) necessary for reading proficiency,” the researchers said.
Collecting data from public health and education systems posed limitations for the study. In addition, the absence of kindergarten language assessments prevented Dr. Meinzen-Derr and colleagues from better elucidating reasons for kindergarten readiness. Also beyond the scope of the study was the ability to evaluate the effect service types may have had on outcomes.
The next step in the research process is to evaluate the link between outcomes and specific EI parameters, they said. “Our study demonstrates that an integrated data system can address relevant and important topics regarding early academic outcomes (kindergarten readiness and reading levels) among children who received EI. The current findings provide a new context by evaluating later outcomes among children who are deaf or hard of hearing,” they added, noting that more research is needed to grasp how various EI services impact outcomes since enrollment age is a marker of EI exposure.
Early intervention is everyone’s business
In a separate interview, Amy Hardy M.S. CCC-SLP, speech language pathologist and clinical professor at Idaho State University, emphasized the importance of early intervention, citing reports from the National Center for Hearing Assessment and Management, which credits detection and treatment of hearing loss at birth per child to saving $400,000 in special education costs by the time they graduate from high school (https://www.ncsl.org/research/health/newborn-hearing-screening-state-laws.aspx).
Earliest possible hearing detection is and should be a standard of care for infants and children, and the importance of follow up appointments also cannot be understated,” Ms. Hardy said. Perhaps the biggest challenge for professionals involved with early learning is that many children are delayed in receiving follow up appointments for hearing detection, she added. When families fail to receive a follow-up notice or opt not attend the follow-up appointment, this leaves infants that may be deaf or hard of hearing unidentified, she explained, noting that in some states, lack of consistent and stable state funding needed for effective follow-up with these children and families is a factor.
Ms. Hardy urged that anyone who knows an expectant family can tout the importance of early screenings. Even daycare workers have a responsibility to play a role in early hearing detection, she noted.
Although speech language pathologists routinely advocate for early intervention, “it is never too late to work on skills that will assist children in their everyday lives,” she advised.
The authors had no relevant financial disclosures. The study was funded in part by the Disability Research and Dissemination Center via cooperative agreements with the Centers for Disease Control and Prevention.
SOURCE: Meinzen-Derr J et al. Pediatrics. 2020 October. doi: 10.1542/peds.2020-0557.
Starting early intervention (EI) enrollment before age 6 months in children who are deaf or hard of hearing may have a lasting influence on ensuring kindergarten readiness, Jareen Meinzen-Derr, PhD, MPH of Cincinnati Children’s Hospital Medical Center and colleagues reported in Pediatrics.
The researchers created a comprehensive, longitudinal, population-based database, which linked hearing screening and diagnostic data to that of early intervention data and educational records for 1,746 infants identified with permanent hearing loss who were born between Jan. 1, 2008 and Dec. 31, 2014 The database was established in partnership with the Ohio Departments of Health, Developmental Disabilities and Education, and with the support of the Centers for Disease Control and Prevention and the National Center on Birth Defects and Developmental Disabilities.
Of those, 784 children ranging from preschool to fourth grade were evaluated based on education data available for the 2017 and 2018 school year that had been linked by way of an identifier that flagged students enrolled in EI.
All together, 417 students had kindergarten assessment records, and of those, 385 had Kindergarten Readiness Assessments (KRAs) between 2014 and 2018; 222 (58%) had been enrolled in EI before the age of 6 months. Of those who were enrolled early, the median age of EI enrollment was 3.4 months (2.4-4.3 months) and in those enrolled later, the median age was 9.2 months (7.5-15.4 months).
The importance of EI prior to 6 months
A total of 109 children (28%) receiving services as part of Ohio’s early intervention programs demonstrated kindergarten readiness on their overall KRA scores. The scores revealed that children receiving EI early (34%, n = 75) were more likely to be ready for kindergarten than were those who entered later (21%, n = 34; P = .005). They also were more likely to have on track language and literacy scores (60% vs. 42%, respectively; P = .0006).
Dr. Meinzen-Derr and colleagues noted that factors identified with “an increased odds of being on track included having private insurance and some college education for the mother.” Conversely, factors identified with a decreased likelihood included having a diagnosed disability and bilateral hearing loss.
The researchers cautioned that children transitioned from EI to academic settings will face challenges that may go underrecognized because a school’s focus often is largely on social and academic performance. Thus, working with linked data systems can provide the data to track outcomes that might otherwise be missed, the researchers noted.
Furthermore, they cautioned that even though kindergarten readiness offers some glimpse into future academic success, these measures alone may not be sufficient predictors for children who are deaf or hard of hearing. Risk for communication, social, and academic delays persist throughout school so it is important to employ alternative methods of reading instruction in order to “achieve more complex skills (e.g., complex syntax and advanced vocabulary) necessary for reading proficiency,” the researchers said.
Collecting data from public health and education systems posed limitations for the study. In addition, the absence of kindergarten language assessments prevented Dr. Meinzen-Derr and colleagues from better elucidating reasons for kindergarten readiness. Also beyond the scope of the study was the ability to evaluate the effect service types may have had on outcomes.
The next step in the research process is to evaluate the link between outcomes and specific EI parameters, they said. “Our study demonstrates that an integrated data system can address relevant and important topics regarding early academic outcomes (kindergarten readiness and reading levels) among children who received EI. The current findings provide a new context by evaluating later outcomes among children who are deaf or hard of hearing,” they added, noting that more research is needed to grasp how various EI services impact outcomes since enrollment age is a marker of EI exposure.
Early intervention is everyone’s business
In a separate interview, Amy Hardy M.S. CCC-SLP, speech language pathologist and clinical professor at Idaho State University, emphasized the importance of early intervention, citing reports from the National Center for Hearing Assessment and Management, which credits detection and treatment of hearing loss at birth per child to saving $400,000 in special education costs by the time they graduate from high school (https://www.ncsl.org/research/health/newborn-hearing-screening-state-laws.aspx).
Earliest possible hearing detection is and should be a standard of care for infants and children, and the importance of follow up appointments also cannot be understated,” Ms. Hardy said. Perhaps the biggest challenge for professionals involved with early learning is that many children are delayed in receiving follow up appointments for hearing detection, she added. When families fail to receive a follow-up notice or opt not attend the follow-up appointment, this leaves infants that may be deaf or hard of hearing unidentified, she explained, noting that in some states, lack of consistent and stable state funding needed for effective follow-up with these children and families is a factor.
Ms. Hardy urged that anyone who knows an expectant family can tout the importance of early screenings. Even daycare workers have a responsibility to play a role in early hearing detection, she noted.
Although speech language pathologists routinely advocate for early intervention, “it is never too late to work on skills that will assist children in their everyday lives,” she advised.
The authors had no relevant financial disclosures. The study was funded in part by the Disability Research and Dissemination Center via cooperative agreements with the Centers for Disease Control and Prevention.
SOURCE: Meinzen-Derr J et al. Pediatrics. 2020 October. doi: 10.1542/peds.2020-0557.
Starting early intervention (EI) enrollment before age 6 months in children who are deaf or hard of hearing may have a lasting influence on ensuring kindergarten readiness, Jareen Meinzen-Derr, PhD, MPH of Cincinnati Children’s Hospital Medical Center and colleagues reported in Pediatrics.
The researchers created a comprehensive, longitudinal, population-based database, which linked hearing screening and diagnostic data to that of early intervention data and educational records for 1,746 infants identified with permanent hearing loss who were born between Jan. 1, 2008 and Dec. 31, 2014 The database was established in partnership with the Ohio Departments of Health, Developmental Disabilities and Education, and with the support of the Centers for Disease Control and Prevention and the National Center on Birth Defects and Developmental Disabilities.
Of those, 784 children ranging from preschool to fourth grade were evaluated based on education data available for the 2017 and 2018 school year that had been linked by way of an identifier that flagged students enrolled in EI.
All together, 417 students had kindergarten assessment records, and of those, 385 had Kindergarten Readiness Assessments (KRAs) between 2014 and 2018; 222 (58%) had been enrolled in EI before the age of 6 months. Of those who were enrolled early, the median age of EI enrollment was 3.4 months (2.4-4.3 months) and in those enrolled later, the median age was 9.2 months (7.5-15.4 months).
The importance of EI prior to 6 months
A total of 109 children (28%) receiving services as part of Ohio’s early intervention programs demonstrated kindergarten readiness on their overall KRA scores. The scores revealed that children receiving EI early (34%, n = 75) were more likely to be ready for kindergarten than were those who entered later (21%, n = 34; P = .005). They also were more likely to have on track language and literacy scores (60% vs. 42%, respectively; P = .0006).
Dr. Meinzen-Derr and colleagues noted that factors identified with “an increased odds of being on track included having private insurance and some college education for the mother.” Conversely, factors identified with a decreased likelihood included having a diagnosed disability and bilateral hearing loss.
The researchers cautioned that children transitioned from EI to academic settings will face challenges that may go underrecognized because a school’s focus often is largely on social and academic performance. Thus, working with linked data systems can provide the data to track outcomes that might otherwise be missed, the researchers noted.
Furthermore, they cautioned that even though kindergarten readiness offers some glimpse into future academic success, these measures alone may not be sufficient predictors for children who are deaf or hard of hearing. Risk for communication, social, and academic delays persist throughout school so it is important to employ alternative methods of reading instruction in order to “achieve more complex skills (e.g., complex syntax and advanced vocabulary) necessary for reading proficiency,” the researchers said.
Collecting data from public health and education systems posed limitations for the study. In addition, the absence of kindergarten language assessments prevented Dr. Meinzen-Derr and colleagues from better elucidating reasons for kindergarten readiness. Also beyond the scope of the study was the ability to evaluate the effect service types may have had on outcomes.
The next step in the research process is to evaluate the link between outcomes and specific EI parameters, they said. “Our study demonstrates that an integrated data system can address relevant and important topics regarding early academic outcomes (kindergarten readiness and reading levels) among children who received EI. The current findings provide a new context by evaluating later outcomes among children who are deaf or hard of hearing,” they added, noting that more research is needed to grasp how various EI services impact outcomes since enrollment age is a marker of EI exposure.
Early intervention is everyone’s business
In a separate interview, Amy Hardy M.S. CCC-SLP, speech language pathologist and clinical professor at Idaho State University, emphasized the importance of early intervention, citing reports from the National Center for Hearing Assessment and Management, which credits detection and treatment of hearing loss at birth per child to saving $400,000 in special education costs by the time they graduate from high school (https://www.ncsl.org/research/health/newborn-hearing-screening-state-laws.aspx).
Earliest possible hearing detection is and should be a standard of care for infants and children, and the importance of follow up appointments also cannot be understated,” Ms. Hardy said. Perhaps the biggest challenge for professionals involved with early learning is that many children are delayed in receiving follow up appointments for hearing detection, she added. When families fail to receive a follow-up notice or opt not attend the follow-up appointment, this leaves infants that may be deaf or hard of hearing unidentified, she explained, noting that in some states, lack of consistent and stable state funding needed for effective follow-up with these children and families is a factor.
Ms. Hardy urged that anyone who knows an expectant family can tout the importance of early screenings. Even daycare workers have a responsibility to play a role in early hearing detection, she noted.
Although speech language pathologists routinely advocate for early intervention, “it is never too late to work on skills that will assist children in their everyday lives,” she advised.
The authors had no relevant financial disclosures. The study was funded in part by the Disability Research and Dissemination Center via cooperative agreements with the Centers for Disease Control and Prevention.
SOURCE: Meinzen-Derr J et al. Pediatrics. 2020 October. doi: 10.1542/peds.2020-0557.
FROM PEDIATRICS
Being HIV positive increases risk of death from COVID-19
compared with people without HIV.
A comparison of outcomes of people with HIV to people without HIV who were hospitalized in the United Kingdom with COVID-19 from Jan. 17 to June 4 showed that HIV-positive status was associated with a 63% increased risk of day 28 mortality.
This was especially true for HIV+ patients younger than 70 years of age, said Anna Maria Geretti, MD, PhD, professor of virology and infectious diseases, University of Liverpool, England.
The results are from an analysis of data from the ISARIC World Health Organization (WHO) Clinical Characterisation Protocol (UK) study, and were presented at the HIV Glasgow annual meeting, held virtually this year because of the pandemic.
“We investigated whether HIV status could be important in COVID-19 outcomes because there was anxiety on the part of our patients, and we wanted to gather some evidence-based information in order to help guide them,” Dr. Geretti said in an interview.
“ISARIC is an international protocol and the UK is one of the nations participating. We applied for access to its very large database, which connects data from all patients who are hospitalized with either known or suspected COVID-19. We wanted to see specifically how the presentation and outcomes of patients with HIV compared with the rest of the population without HIV. It afforded us an ideal opportunity to start to answer this question, and this is our first analysis in what will be an ongoing process. Importantly, we showed that there is a need to really look more carefully at the population with HIV,” she said.
Out of a total of 47,539 patients in the database, 115 (0.24%) had confirmed HIV-positive status, and 103 of those 115, or 89.6%, had a record of being on antiretroviral therapy.
On admission, the patients with HIV were younger, with a median age of 55 compared with 74 for patients without HIV (P < .001). They also had a higher prevalence of obesity, moderate to severe liver disease, higher lymphocyte counts and C-reactive protein, as well as more systemic symptoms.
There were no differences in respiratory rate, need for oxygen, or prevalence of chest infiltrates.
The cumulative incidence of mortality at day 28 was 25.2% in HIV-positive patients compared with 32.1% in HIV-negative patients (P = .12).
But when the researchers looked more closely, they noticed that the mortality rate was actually higher in younger HIV+ patients compared with HIV-negative patients.
Stratified by age, 28-day mortality was significantly higher in HIV+ patients aged <50 years (P =.004); and those aged 50 to 59 years (P = .05).
“So below the age of 70, the risk of mortality was double in people with HIV. The people with HIV who died often had diabetes with complications and also more frequent obesity, but this was not the only explanation,” Dr. Geretti said. “There is something to do with the HIV status per se.”
Next steps will be to expand the data set and repeat the analysis with an additional 100 patients “at least” she said.
The researchers also hope to zero in on what about being HIV+ is increasing the mortality risk from COVID-19.
“Right now we need greater numbers and we hope that the research community will be stimulated to take a closer look at this information, and merge other data so that we can strengthen confidence in the data and tease out what factors are causing this increased risk for mortality,” Dr. Geretti said.
She also emphasized that all patients admitted to hospital with COVID-19 should be asked about their HIV status.
“It is important that the HIV status be recorded if we want to increase our ability to understand how HIV impacts survival,” she stressed. “In our experience we found that most of the hospital records were not doing that. Since HIV+ patients seem to be at increased risk, HIV status should be factored into the clinical management. Ask patients if they are HIV+, and if it is not known, then do a test. That would be good practice.”
Dr. Geretti reported no relevant financial relationships. The work was supported by grants from the National Institute of Health Research, the Medical Research Council, the Wellcome Trust, the Department for International Development, and the Bill and Melinda Gates Foundation.
A version of this article originally appeared on Medscape.com.
compared with people without HIV.
A comparison of outcomes of people with HIV to people without HIV who were hospitalized in the United Kingdom with COVID-19 from Jan. 17 to June 4 showed that HIV-positive status was associated with a 63% increased risk of day 28 mortality.
This was especially true for HIV+ patients younger than 70 years of age, said Anna Maria Geretti, MD, PhD, professor of virology and infectious diseases, University of Liverpool, England.
The results are from an analysis of data from the ISARIC World Health Organization (WHO) Clinical Characterisation Protocol (UK) study, and were presented at the HIV Glasgow annual meeting, held virtually this year because of the pandemic.
“We investigated whether HIV status could be important in COVID-19 outcomes because there was anxiety on the part of our patients, and we wanted to gather some evidence-based information in order to help guide them,” Dr. Geretti said in an interview.
“ISARIC is an international protocol and the UK is one of the nations participating. We applied for access to its very large database, which connects data from all patients who are hospitalized with either known or suspected COVID-19. We wanted to see specifically how the presentation and outcomes of patients with HIV compared with the rest of the population without HIV. It afforded us an ideal opportunity to start to answer this question, and this is our first analysis in what will be an ongoing process. Importantly, we showed that there is a need to really look more carefully at the population with HIV,” she said.
Out of a total of 47,539 patients in the database, 115 (0.24%) had confirmed HIV-positive status, and 103 of those 115, or 89.6%, had a record of being on antiretroviral therapy.
On admission, the patients with HIV were younger, with a median age of 55 compared with 74 for patients without HIV (P < .001). They also had a higher prevalence of obesity, moderate to severe liver disease, higher lymphocyte counts and C-reactive protein, as well as more systemic symptoms.
There were no differences in respiratory rate, need for oxygen, or prevalence of chest infiltrates.
The cumulative incidence of mortality at day 28 was 25.2% in HIV-positive patients compared with 32.1% in HIV-negative patients (P = .12).
But when the researchers looked more closely, they noticed that the mortality rate was actually higher in younger HIV+ patients compared with HIV-negative patients.
Stratified by age, 28-day mortality was significantly higher in HIV+ patients aged <50 years (P =.004); and those aged 50 to 59 years (P = .05).
“So below the age of 70, the risk of mortality was double in people with HIV. The people with HIV who died often had diabetes with complications and also more frequent obesity, but this was not the only explanation,” Dr. Geretti said. “There is something to do with the HIV status per se.”
Next steps will be to expand the data set and repeat the analysis with an additional 100 patients “at least” she said.
The researchers also hope to zero in on what about being HIV+ is increasing the mortality risk from COVID-19.
“Right now we need greater numbers and we hope that the research community will be stimulated to take a closer look at this information, and merge other data so that we can strengthen confidence in the data and tease out what factors are causing this increased risk for mortality,” Dr. Geretti said.
She also emphasized that all patients admitted to hospital with COVID-19 should be asked about their HIV status.
“It is important that the HIV status be recorded if we want to increase our ability to understand how HIV impacts survival,” she stressed. “In our experience we found that most of the hospital records were not doing that. Since HIV+ patients seem to be at increased risk, HIV status should be factored into the clinical management. Ask patients if they are HIV+, and if it is not known, then do a test. That would be good practice.”
Dr. Geretti reported no relevant financial relationships. The work was supported by grants from the National Institute of Health Research, the Medical Research Council, the Wellcome Trust, the Department for International Development, and the Bill and Melinda Gates Foundation.
A version of this article originally appeared on Medscape.com.
compared with people without HIV.
A comparison of outcomes of people with HIV to people without HIV who were hospitalized in the United Kingdom with COVID-19 from Jan. 17 to June 4 showed that HIV-positive status was associated with a 63% increased risk of day 28 mortality.
This was especially true for HIV+ patients younger than 70 years of age, said Anna Maria Geretti, MD, PhD, professor of virology and infectious diseases, University of Liverpool, England.
The results are from an analysis of data from the ISARIC World Health Organization (WHO) Clinical Characterisation Protocol (UK) study, and were presented at the HIV Glasgow annual meeting, held virtually this year because of the pandemic.
“We investigated whether HIV status could be important in COVID-19 outcomes because there was anxiety on the part of our patients, and we wanted to gather some evidence-based information in order to help guide them,” Dr. Geretti said in an interview.
“ISARIC is an international protocol and the UK is one of the nations participating. We applied for access to its very large database, which connects data from all patients who are hospitalized with either known or suspected COVID-19. We wanted to see specifically how the presentation and outcomes of patients with HIV compared with the rest of the population without HIV. It afforded us an ideal opportunity to start to answer this question, and this is our first analysis in what will be an ongoing process. Importantly, we showed that there is a need to really look more carefully at the population with HIV,” she said.
Out of a total of 47,539 patients in the database, 115 (0.24%) had confirmed HIV-positive status, and 103 of those 115, or 89.6%, had a record of being on antiretroviral therapy.
On admission, the patients with HIV were younger, with a median age of 55 compared with 74 for patients without HIV (P < .001). They also had a higher prevalence of obesity, moderate to severe liver disease, higher lymphocyte counts and C-reactive protein, as well as more systemic symptoms.
There were no differences in respiratory rate, need for oxygen, or prevalence of chest infiltrates.
The cumulative incidence of mortality at day 28 was 25.2% in HIV-positive patients compared with 32.1% in HIV-negative patients (P = .12).
But when the researchers looked more closely, they noticed that the mortality rate was actually higher in younger HIV+ patients compared with HIV-negative patients.
Stratified by age, 28-day mortality was significantly higher in HIV+ patients aged <50 years (P =.004); and those aged 50 to 59 years (P = .05).
“So below the age of 70, the risk of mortality was double in people with HIV. The people with HIV who died often had diabetes with complications and also more frequent obesity, but this was not the only explanation,” Dr. Geretti said. “There is something to do with the HIV status per se.”
Next steps will be to expand the data set and repeat the analysis with an additional 100 patients “at least” she said.
The researchers also hope to zero in on what about being HIV+ is increasing the mortality risk from COVID-19.
“Right now we need greater numbers and we hope that the research community will be stimulated to take a closer look at this information, and merge other data so that we can strengthen confidence in the data and tease out what factors are causing this increased risk for mortality,” Dr. Geretti said.
She also emphasized that all patients admitted to hospital with COVID-19 should be asked about their HIV status.
“It is important that the HIV status be recorded if we want to increase our ability to understand how HIV impacts survival,” she stressed. “In our experience we found that most of the hospital records were not doing that. Since HIV+ patients seem to be at increased risk, HIV status should be factored into the clinical management. Ask patients if they are HIV+, and if it is not known, then do a test. That would be good practice.”
Dr. Geretti reported no relevant financial relationships. The work was supported by grants from the National Institute of Health Research, the Medical Research Council, the Wellcome Trust, the Department for International Development, and the Bill and Melinda Gates Foundation.
A version of this article originally appeared on Medscape.com.
Switch to integrase inhibitor regimen safe and effective
data from a randomized trial indicate.
Among 212 women with successful HIV virologic suppression following 48 weeks of treatment with ritonavir-boosted atazanavir plus tenofovir disoproxil fumarate (ATV/r +TDF), among those who were switched to continued therapy with an integrase inhibitor–based regimen of elvitegravir/cobicistat/emtricitabine and tenofovir alafenamide (E/C/F/TAF), mean increases in lumbar spine bone mineral density (BMD) were greater and renal function was improved compared with patients who were maintained with ATV/r + TDF, reported Monica Thormann, MD, from Salvador B. Gautier Hospital in Santo Domingo, Dominican Republic, and colleagues at the HIV Glasgow drug therapy meeting, which was held online in 2020.
Although the E/C/F/TAF regimen was associated with a significantly greater increase in lipids, there was no significant change in the total cholesterol to high-density lipoprotein (HDL) cholesterol ratio.
The patients in the study had previously participated in a blinded randomized trial comparing the integrase inhibitor combination plus TDF with ATV/r + TDF in treatment-naive women.
In the current study, patients were randomly assigned in a 3:1 ratio to maintenance with either E/C/F/TAF (159 patients) or ATV/r + TDF (53 patients).
Forty-eight weeks after the switch, virologic suppression (to fewer than 50 copies/mL) was maintained among 94.3% of those on the integrase inhibitor–based regimen, compared with 86.8% of those on the protease inhibitor–based regimen. Virologic failure was seen in 1.9% of those on the integrase inhibitor–based regimen and in 3.8% of those on the protease inhibitor–based regimen.
In addition, virologic suppression below 20 c/mL at week 48 was more common among women maintained on E/C/F/TAF, at 84.9% vs 71.7% (P = .041). No treatment-emergent resistance was seen with either regimen.
As noted, there were higher mean percentage increases in BMD in the E/C/F/TAF group for both total hip and lumbar spine, but only the latter measure improved significantly in comparison with patients treated with ATV/r + TDF (2.82% vs 0%, P < .001).
Markers of renal tubule damage, including the beta-2 microglobulin to creatinine ratio and the rentinol-binding protein to creatinine ratio, were significantly improved with the integrase inhibitor regimen.
Increases in total cholesterol, LDL cholesterol, and HDL cholesterol were 27 vs 5 mg/dL, 16 vs 8 mg/dL, and 5 vs 0 mg/dL in each case comparing the integrase inhibitor–based regimen to the protease inhibitor–based regimen. All of those comparisons were statistically significant.
As noted, however, the total cholesterol to HDL cholesterol ratio was not significantly different between the treatment arms. The rate or initiation of lipid-modifying agents was 1.3% in the E/C/F/TAF group vs 0 in the ATV/r + TDF group, but this difference was not statistically significant.
“These data demonstrate that women who switch to an integrase inhibitor + TAF‐based regimen maintain high levels of virologic suppression with improvement in BMD and renal function biomarkers, as compared with those remaining on their ritonavir boosted atazanavir + TDF‐based regimen,” the authors wrote.
This article first appeared on Medscape.com.
data from a randomized trial indicate.
Among 212 women with successful HIV virologic suppression following 48 weeks of treatment with ritonavir-boosted atazanavir plus tenofovir disoproxil fumarate (ATV/r +TDF), among those who were switched to continued therapy with an integrase inhibitor–based regimen of elvitegravir/cobicistat/emtricitabine and tenofovir alafenamide (E/C/F/TAF), mean increases in lumbar spine bone mineral density (BMD) were greater and renal function was improved compared with patients who were maintained with ATV/r + TDF, reported Monica Thormann, MD, from Salvador B. Gautier Hospital in Santo Domingo, Dominican Republic, and colleagues at the HIV Glasgow drug therapy meeting, which was held online in 2020.
Although the E/C/F/TAF regimen was associated with a significantly greater increase in lipids, there was no significant change in the total cholesterol to high-density lipoprotein (HDL) cholesterol ratio.
The patients in the study had previously participated in a blinded randomized trial comparing the integrase inhibitor combination plus TDF with ATV/r + TDF in treatment-naive women.
In the current study, patients were randomly assigned in a 3:1 ratio to maintenance with either E/C/F/TAF (159 patients) or ATV/r + TDF (53 patients).
Forty-eight weeks after the switch, virologic suppression (to fewer than 50 copies/mL) was maintained among 94.3% of those on the integrase inhibitor–based regimen, compared with 86.8% of those on the protease inhibitor–based regimen. Virologic failure was seen in 1.9% of those on the integrase inhibitor–based regimen and in 3.8% of those on the protease inhibitor–based regimen.
In addition, virologic suppression below 20 c/mL at week 48 was more common among women maintained on E/C/F/TAF, at 84.9% vs 71.7% (P = .041). No treatment-emergent resistance was seen with either regimen.
As noted, there were higher mean percentage increases in BMD in the E/C/F/TAF group for both total hip and lumbar spine, but only the latter measure improved significantly in comparison with patients treated with ATV/r + TDF (2.82% vs 0%, P < .001).
Markers of renal tubule damage, including the beta-2 microglobulin to creatinine ratio and the rentinol-binding protein to creatinine ratio, were significantly improved with the integrase inhibitor regimen.
Increases in total cholesterol, LDL cholesterol, and HDL cholesterol were 27 vs 5 mg/dL, 16 vs 8 mg/dL, and 5 vs 0 mg/dL in each case comparing the integrase inhibitor–based regimen to the protease inhibitor–based regimen. All of those comparisons were statistically significant.
As noted, however, the total cholesterol to HDL cholesterol ratio was not significantly different between the treatment arms. The rate or initiation of lipid-modifying agents was 1.3% in the E/C/F/TAF group vs 0 in the ATV/r + TDF group, but this difference was not statistically significant.
“These data demonstrate that women who switch to an integrase inhibitor + TAF‐based regimen maintain high levels of virologic suppression with improvement in BMD and renal function biomarkers, as compared with those remaining on their ritonavir boosted atazanavir + TDF‐based regimen,” the authors wrote.
This article first appeared on Medscape.com.
data from a randomized trial indicate.
Among 212 women with successful HIV virologic suppression following 48 weeks of treatment with ritonavir-boosted atazanavir plus tenofovir disoproxil fumarate (ATV/r +TDF), among those who were switched to continued therapy with an integrase inhibitor–based regimen of elvitegravir/cobicistat/emtricitabine and tenofovir alafenamide (E/C/F/TAF), mean increases in lumbar spine bone mineral density (BMD) were greater and renal function was improved compared with patients who were maintained with ATV/r + TDF, reported Monica Thormann, MD, from Salvador B. Gautier Hospital in Santo Domingo, Dominican Republic, and colleagues at the HIV Glasgow drug therapy meeting, which was held online in 2020.
Although the E/C/F/TAF regimen was associated with a significantly greater increase in lipids, there was no significant change in the total cholesterol to high-density lipoprotein (HDL) cholesterol ratio.
The patients in the study had previously participated in a blinded randomized trial comparing the integrase inhibitor combination plus TDF with ATV/r + TDF in treatment-naive women.
In the current study, patients were randomly assigned in a 3:1 ratio to maintenance with either E/C/F/TAF (159 patients) or ATV/r + TDF (53 patients).
Forty-eight weeks after the switch, virologic suppression (to fewer than 50 copies/mL) was maintained among 94.3% of those on the integrase inhibitor–based regimen, compared with 86.8% of those on the protease inhibitor–based regimen. Virologic failure was seen in 1.9% of those on the integrase inhibitor–based regimen and in 3.8% of those on the protease inhibitor–based regimen.
In addition, virologic suppression below 20 c/mL at week 48 was more common among women maintained on E/C/F/TAF, at 84.9% vs 71.7% (P = .041). No treatment-emergent resistance was seen with either regimen.
As noted, there were higher mean percentage increases in BMD in the E/C/F/TAF group for both total hip and lumbar spine, but only the latter measure improved significantly in comparison with patients treated with ATV/r + TDF (2.82% vs 0%, P < .001).
Markers of renal tubule damage, including the beta-2 microglobulin to creatinine ratio and the rentinol-binding protein to creatinine ratio, were significantly improved with the integrase inhibitor regimen.
Increases in total cholesterol, LDL cholesterol, and HDL cholesterol were 27 vs 5 mg/dL, 16 vs 8 mg/dL, and 5 vs 0 mg/dL in each case comparing the integrase inhibitor–based regimen to the protease inhibitor–based regimen. All of those comparisons were statistically significant.
As noted, however, the total cholesterol to HDL cholesterol ratio was not significantly different between the treatment arms. The rate or initiation of lipid-modifying agents was 1.3% in the E/C/F/TAF group vs 0 in the ATV/r + TDF group, but this difference was not statistically significant.
“These data demonstrate that women who switch to an integrase inhibitor + TAF‐based regimen maintain high levels of virologic suppression with improvement in BMD and renal function biomarkers, as compared with those remaining on their ritonavir boosted atazanavir + TDF‐based regimen,” the authors wrote.
This article first appeared on Medscape.com.
Delayed cancer screening could cause increase in deaths, study says
Delays in colorectal cancer screening due to the COVID-19 pandemic could lead to higher rates of advanced-stage cancer and death, according to a new study.
When compared with a delay of less than three months, the longer delay seen this year may result in an 11.9% increase in death rates.
“Across the globe, health care systems are facing serious difficulties while dealing with COVID-19, and it is imperative that support is given to the public and patients throughout the crisis, including for high-impact diseases such as colorectal cancer,” Luigi Ricciardiello, the lead study author and a professor at the University of Bologna in Italy, said in a statement.
Ricciardiello and colleagues presented their research on Monday at UEG Week Virtual 2020, an international conference for gastroenterologists. The study will be published in the UEG Journal .
The researchers created a model to forecast the effects of delayed cancer screening during 2020. A “moderate” delay of 7-12 months caused a 3% increase in advanced-stage colon cancer, and a long delay of more than 12 months caused a 7% increase in advanced cancer.
Based on a survival rate of 5 years for stage 3 or stage 4 colorectal cancer, the death rate would increase nearly 12% when screening is delayed for more than a year, as compared with less than three months of delay.
The research team found similar results when forecasting advanced-stage cancer and deaths earlier this year. In a paper published in Clinical Gastroenterology and Hepatology in early September, they projected that deaths could increase 12% if screening is delayed for more than a year.
Throughout the pandemic, screening programs have been delayed in many countries, particularly across Europe.
“Healthcare authorities need to act urgently on how they reorganise activities during COVID-19, without compromising the diagnosis of other high-impact diseases like this research shows,” Ricciardiello said.
United European Gastroenterology, a professional medical organization for digestive health specialists, has called for policymakers to implement colon cancer screening programs across the European Union. Annually, more than 375,000 new cases are diagnosed across the EU, and more than 170,000 people die from colorectal cancer, according to a UEG report.
“Early-stage diagnosis of colorectal cancer is crucial — it’s far easier to treat and enhances optimal patient outcomes,” Ricciardiello said. “It is therefore essential that vital diagnosis tools, like screening programmes, continue and help to prevent mortality rates from rising even further.”
This article first appeared on Medscape.com.
Delays in colorectal cancer screening due to the COVID-19 pandemic could lead to higher rates of advanced-stage cancer and death, according to a new study.
When compared with a delay of less than three months, the longer delay seen this year may result in an 11.9% increase in death rates.
“Across the globe, health care systems are facing serious difficulties while dealing with COVID-19, and it is imperative that support is given to the public and patients throughout the crisis, including for high-impact diseases such as colorectal cancer,” Luigi Ricciardiello, the lead study author and a professor at the University of Bologna in Italy, said in a statement.
Ricciardiello and colleagues presented their research on Monday at UEG Week Virtual 2020, an international conference for gastroenterologists. The study will be published in the UEG Journal .
The researchers created a model to forecast the effects of delayed cancer screening during 2020. A “moderate” delay of 7-12 months caused a 3% increase in advanced-stage colon cancer, and a long delay of more than 12 months caused a 7% increase in advanced cancer.
Based on a survival rate of 5 years for stage 3 or stage 4 colorectal cancer, the death rate would increase nearly 12% when screening is delayed for more than a year, as compared with less than three months of delay.
The research team found similar results when forecasting advanced-stage cancer and deaths earlier this year. In a paper published in Clinical Gastroenterology and Hepatology in early September, they projected that deaths could increase 12% if screening is delayed for more than a year.
Throughout the pandemic, screening programs have been delayed in many countries, particularly across Europe.
“Healthcare authorities need to act urgently on how they reorganise activities during COVID-19, without compromising the diagnosis of other high-impact diseases like this research shows,” Ricciardiello said.
United European Gastroenterology, a professional medical organization for digestive health specialists, has called for policymakers to implement colon cancer screening programs across the European Union. Annually, more than 375,000 new cases are diagnosed across the EU, and more than 170,000 people die from colorectal cancer, according to a UEG report.
“Early-stage diagnosis of colorectal cancer is crucial — it’s far easier to treat and enhances optimal patient outcomes,” Ricciardiello said. “It is therefore essential that vital diagnosis tools, like screening programmes, continue and help to prevent mortality rates from rising even further.”
This article first appeared on Medscape.com.
Delays in colorectal cancer screening due to the COVID-19 pandemic could lead to higher rates of advanced-stage cancer and death, according to a new study.
When compared with a delay of less than three months, the longer delay seen this year may result in an 11.9% increase in death rates.
“Across the globe, health care systems are facing serious difficulties while dealing with COVID-19, and it is imperative that support is given to the public and patients throughout the crisis, including for high-impact diseases such as colorectal cancer,” Luigi Ricciardiello, the lead study author and a professor at the University of Bologna in Italy, said in a statement.
Ricciardiello and colleagues presented their research on Monday at UEG Week Virtual 2020, an international conference for gastroenterologists. The study will be published in the UEG Journal .
The researchers created a model to forecast the effects of delayed cancer screening during 2020. A “moderate” delay of 7-12 months caused a 3% increase in advanced-stage colon cancer, and a long delay of more than 12 months caused a 7% increase in advanced cancer.
Based on a survival rate of 5 years for stage 3 or stage 4 colorectal cancer, the death rate would increase nearly 12% when screening is delayed for more than a year, as compared with less than three months of delay.
The research team found similar results when forecasting advanced-stage cancer and deaths earlier this year. In a paper published in Clinical Gastroenterology and Hepatology in early September, they projected that deaths could increase 12% if screening is delayed for more than a year.
Throughout the pandemic, screening programs have been delayed in many countries, particularly across Europe.
“Healthcare authorities need to act urgently on how they reorganise activities during COVID-19, without compromising the diagnosis of other high-impact diseases like this research shows,” Ricciardiello said.
United European Gastroenterology, a professional medical organization for digestive health specialists, has called for policymakers to implement colon cancer screening programs across the European Union. Annually, more than 375,000 new cases are diagnosed across the EU, and more than 170,000 people die from colorectal cancer, according to a UEG report.
“Early-stage diagnosis of colorectal cancer is crucial — it’s far easier to treat and enhances optimal patient outcomes,” Ricciardiello said. “It is therefore essential that vital diagnosis tools, like screening programmes, continue and help to prevent mortality rates from rising even further.”
This article first appeared on Medscape.com.