User login
Bringing you the latest news, research and reviews, exclusive interviews, podcasts, quizzes, and more.
div[contains(@class, 'header__large-screen')]
div[contains(@class, 'read-next-article')]
div[contains(@class, 'nav-primary')]
nav[contains(@class, 'nav-primary')]
section[contains(@class, 'footer-nav-section-wrapper')]
footer[@id='footer']
div[contains(@class, 'main-prefix')]
section[contains(@class, 'nav-hidden')]
div[contains(@class, 'ce-card-content')]
nav[contains(@class, 'nav-ce-stack')]
Study addresses whether cosmetic treatments make patients happier
DENVER – compared with the general population, according to a study of 42 individuals. However, these treatments did not improve their baseline happiness or life satisfaction scores at follow-up.
Those are key findings from the study that lead author Rishi Chopra, MD, MS, presented during an oral abstract session at the annual meeting of the American Society for Dermatologic Surgery.
“These are interesting and surprising results,” said Dr. Chopra, a dermatologist and laser and cosmetic dermatologic surgery fellow at Harvard Medical School and Massachusetts General Hospital in Boston. “Patients are seeking consultations with us with the hope that the treatments we offer may potentially help them feel happier, but are we really delivering on that?”
In a pivotal 2018 study that examined patient motivations for undergoing cosmetic dermatology procedures, investigators found that 67.2% did so to “feel happier and more confident or improve total quality of life”. Moreover, 38.5% cited the desire to “feel happier, better overall, or improve total quality of life” as the key reason for pursuing cosmetic procedures.
Prior published evidence validates this benefit of procedures, as neuromodulators have repeatedly demonstrated to improve mood and depression, including a 2020 randomized, single-blind crossover study that examined the impact of neuromodulators on mood and appearance during the COVID-19 pandemic. It found that patients who received treatment with neuromodulators prior to the pandemic, stopped during the pandemic, and restarted again, reported increased happiness, self-satisfaction with appearance, and overall treatment satisfaction.
“However, studies evaluating the effect of filler on happiness have failed to demonstrate an impact,” Dr. Chopra said. “Thus, the jury is still out.”
Study evaluated 42 patients
In what he said is the first study of its kind, he and his colleagues evaluated the impact of minimally invasive cosmetic procedures on the happiness of 42 treatment non-naive patients (those who regularly undergo cosmetic procedures) with a mean age of 47 years who were surveyed in November and December of 2021 during the COVID-19 Omicron subvariant outbreak at the cosmetic dermatology practices of Sabrina G. Fabi, MD, in San Diego, and Nicole Kanaris, MBBCh, in Johannesburg, South Africa.
“On average, these patients were undergoing six treatments per year during four visits per year, so these were frequent flyers,” Dr. Chopra said. “We set out to assess: Are patients who seek cosmetic procedures happy at baseline? And, do cosmetic procedures make us happier or more satisfied with life?”
Prior to treatment, patients completed the Subjective Happiness Scale (SHS) and Satisfaction With Life Scale (SWLS). Three weeks later, patients completed the SHS, SWLS, the Global Aesthetic Improvement Scale (GAIS) and a 5-point satisfaction score. The researchers used paired and unpaired t-tests, independent sample t-tests, and Spearman rank correlations to conduct statistical analyses.
The baseline SHS score of study participants was an average of 5.87, which Dr. Chopra said is higher than the worldwide population range between 4.57 and 5.33, and 5.05 in the U.S. population. “The patients in our study were very happy to begin with,” an important point to consider, he said. Following their treatments, respondents felt “improved” or “much improved” on the GAIS (a mean score of 3.64) and “somewhat satisfied” or “very satisfied” based on the SWLS (a mean score of 4.4). “So overall, they viewed their treatments as a success,” Dr. Chopra said.
In terms of happiness, however, the researchers observed no significant differences between pre- and posttreatment scores on the SHS (a mean of 5.87 vs. 6.61, respectively; P = .634) nor on the SWLS (a mean of 29.62 vs. 29.1; P = .709). On stratified analysis, no significant differences in the SHS, SWLS, and the GAIS were observed when the researchers accounted for the aggressiveness of the procedure, the number of treatments, the number of sites treated, the type of treatment, and whether the respondents were happier or sadder at baseline. “Surprisingly, this had no effect whatsoever on happiness,” he said. “Not only that, these factors didn’t improve a patient’s perception of the efficacy or satisfaction with a treatment either.”
According to Dr. Chopra, this is the first study to evaluate the impact of a broad spectrum of minimally invasive cosmetic procedures, including injectables and lasers, on the happiness and life satisfaction of treatment non-naive patients.
“Surprisingly, we found these patients were no happier after treatment,” he told this news organization. “However, before rushing to declare that cosmetic procedures don’t make us happier, it is critical to evaluate these results in the context of our study population. We believe there to be a distinction between treatment naive and non-naive patients. All the patients in our study were treatment non-naive, routinely and frequently undergoing cosmetic procedures. Moreover, our treatment non-naive patients were very happy at baseline prior to treatment.”
He and his colleagues hypothesize that there is a “ceiling effect” to the happiness one can attain via these procedures. “Our treatment non-naive patients had already reached this ceiling-peak happiness of their treatment journey, and at this point were only pursuing procedures to maintain their results and happiness,” he said. “Thus, we were unable to measure any effect this late in the ‘maintenance-phase’ of their journey via our study. On the other hand, treatment naive patients (those who have never undergone a cosmetic procedure) were not included. We hypothesize that evaluating patients at the start of their journey after their first round of treatments will demonstrate an impact on happiness, prior to reaching the ceiling and subsequent ‘maintenance phase.’ ”
Lawrence J. Green, MD, clinical professor of dermatology at George Washington University, Washington, who was asked to comment on the study results, said that it was not clear which specific cosmetic treatments the study participants received. “I would like to see if different injectable or device treatments would give different happiness scale results,” Dr. Green said.
“In addition, only two locations were surveyed, so the results could have location bias. I think it would be a great idea to replicate this survey of experienced cosmetic treatment patients with many locations and to include survey responses based on the procedure that was done. That said, it is interesting that overall, investigator satisfaction did not correlate with patient happiness from the treatments.”
Dr. Chopra reported having no financial disclosures. Dr. Green disclosed that he is a speaker, consultant, or investigator for numerous pharmaceutical companies.
DENVER – compared with the general population, according to a study of 42 individuals. However, these treatments did not improve their baseline happiness or life satisfaction scores at follow-up.
Those are key findings from the study that lead author Rishi Chopra, MD, MS, presented during an oral abstract session at the annual meeting of the American Society for Dermatologic Surgery.
“These are interesting and surprising results,” said Dr. Chopra, a dermatologist and laser and cosmetic dermatologic surgery fellow at Harvard Medical School and Massachusetts General Hospital in Boston. “Patients are seeking consultations with us with the hope that the treatments we offer may potentially help them feel happier, but are we really delivering on that?”
In a pivotal 2018 study that examined patient motivations for undergoing cosmetic dermatology procedures, investigators found that 67.2% did so to “feel happier and more confident or improve total quality of life”. Moreover, 38.5% cited the desire to “feel happier, better overall, or improve total quality of life” as the key reason for pursuing cosmetic procedures.
Prior published evidence validates this benefit of procedures, as neuromodulators have repeatedly demonstrated to improve mood and depression, including a 2020 randomized, single-blind crossover study that examined the impact of neuromodulators on mood and appearance during the COVID-19 pandemic. It found that patients who received treatment with neuromodulators prior to the pandemic, stopped during the pandemic, and restarted again, reported increased happiness, self-satisfaction with appearance, and overall treatment satisfaction.
“However, studies evaluating the effect of filler on happiness have failed to demonstrate an impact,” Dr. Chopra said. “Thus, the jury is still out.”
Study evaluated 42 patients
In what he said is the first study of its kind, he and his colleagues evaluated the impact of minimally invasive cosmetic procedures on the happiness of 42 treatment non-naive patients (those who regularly undergo cosmetic procedures) with a mean age of 47 years who were surveyed in November and December of 2021 during the COVID-19 Omicron subvariant outbreak at the cosmetic dermatology practices of Sabrina G. Fabi, MD, in San Diego, and Nicole Kanaris, MBBCh, in Johannesburg, South Africa.
“On average, these patients were undergoing six treatments per year during four visits per year, so these were frequent flyers,” Dr. Chopra said. “We set out to assess: Are patients who seek cosmetic procedures happy at baseline? And, do cosmetic procedures make us happier or more satisfied with life?”
Prior to treatment, patients completed the Subjective Happiness Scale (SHS) and Satisfaction With Life Scale (SWLS). Three weeks later, patients completed the SHS, SWLS, the Global Aesthetic Improvement Scale (GAIS) and a 5-point satisfaction score. The researchers used paired and unpaired t-tests, independent sample t-tests, and Spearman rank correlations to conduct statistical analyses.
The baseline SHS score of study participants was an average of 5.87, which Dr. Chopra said is higher than the worldwide population range between 4.57 and 5.33, and 5.05 in the U.S. population. “The patients in our study were very happy to begin with,” an important point to consider, he said. Following their treatments, respondents felt “improved” or “much improved” on the GAIS (a mean score of 3.64) and “somewhat satisfied” or “very satisfied” based on the SWLS (a mean score of 4.4). “So overall, they viewed their treatments as a success,” Dr. Chopra said.
In terms of happiness, however, the researchers observed no significant differences between pre- and posttreatment scores on the SHS (a mean of 5.87 vs. 6.61, respectively; P = .634) nor on the SWLS (a mean of 29.62 vs. 29.1; P = .709). On stratified analysis, no significant differences in the SHS, SWLS, and the GAIS were observed when the researchers accounted for the aggressiveness of the procedure, the number of treatments, the number of sites treated, the type of treatment, and whether the respondents were happier or sadder at baseline. “Surprisingly, this had no effect whatsoever on happiness,” he said. “Not only that, these factors didn’t improve a patient’s perception of the efficacy or satisfaction with a treatment either.”
According to Dr. Chopra, this is the first study to evaluate the impact of a broad spectrum of minimally invasive cosmetic procedures, including injectables and lasers, on the happiness and life satisfaction of treatment non-naive patients.
“Surprisingly, we found these patients were no happier after treatment,” he told this news organization. “However, before rushing to declare that cosmetic procedures don’t make us happier, it is critical to evaluate these results in the context of our study population. We believe there to be a distinction between treatment naive and non-naive patients. All the patients in our study were treatment non-naive, routinely and frequently undergoing cosmetic procedures. Moreover, our treatment non-naive patients were very happy at baseline prior to treatment.”
He and his colleagues hypothesize that there is a “ceiling effect” to the happiness one can attain via these procedures. “Our treatment non-naive patients had already reached this ceiling-peak happiness of their treatment journey, and at this point were only pursuing procedures to maintain their results and happiness,” he said. “Thus, we were unable to measure any effect this late in the ‘maintenance-phase’ of their journey via our study. On the other hand, treatment naive patients (those who have never undergone a cosmetic procedure) were not included. We hypothesize that evaluating patients at the start of their journey after their first round of treatments will demonstrate an impact on happiness, prior to reaching the ceiling and subsequent ‘maintenance phase.’ ”
Lawrence J. Green, MD, clinical professor of dermatology at George Washington University, Washington, who was asked to comment on the study results, said that it was not clear which specific cosmetic treatments the study participants received. “I would like to see if different injectable or device treatments would give different happiness scale results,” Dr. Green said.
“In addition, only two locations were surveyed, so the results could have location bias. I think it would be a great idea to replicate this survey of experienced cosmetic treatment patients with many locations and to include survey responses based on the procedure that was done. That said, it is interesting that overall, investigator satisfaction did not correlate with patient happiness from the treatments.”
Dr. Chopra reported having no financial disclosures. Dr. Green disclosed that he is a speaker, consultant, or investigator for numerous pharmaceutical companies.
DENVER – compared with the general population, according to a study of 42 individuals. However, these treatments did not improve their baseline happiness or life satisfaction scores at follow-up.
Those are key findings from the study that lead author Rishi Chopra, MD, MS, presented during an oral abstract session at the annual meeting of the American Society for Dermatologic Surgery.
“These are interesting and surprising results,” said Dr. Chopra, a dermatologist and laser and cosmetic dermatologic surgery fellow at Harvard Medical School and Massachusetts General Hospital in Boston. “Patients are seeking consultations with us with the hope that the treatments we offer may potentially help them feel happier, but are we really delivering on that?”
In a pivotal 2018 study that examined patient motivations for undergoing cosmetic dermatology procedures, investigators found that 67.2% did so to “feel happier and more confident or improve total quality of life”. Moreover, 38.5% cited the desire to “feel happier, better overall, or improve total quality of life” as the key reason for pursuing cosmetic procedures.
Prior published evidence validates this benefit of procedures, as neuromodulators have repeatedly demonstrated to improve mood and depression, including a 2020 randomized, single-blind crossover study that examined the impact of neuromodulators on mood and appearance during the COVID-19 pandemic. It found that patients who received treatment with neuromodulators prior to the pandemic, stopped during the pandemic, and restarted again, reported increased happiness, self-satisfaction with appearance, and overall treatment satisfaction.
“However, studies evaluating the effect of filler on happiness have failed to demonstrate an impact,” Dr. Chopra said. “Thus, the jury is still out.”
Study evaluated 42 patients
In what he said is the first study of its kind, he and his colleagues evaluated the impact of minimally invasive cosmetic procedures on the happiness of 42 treatment non-naive patients (those who regularly undergo cosmetic procedures) with a mean age of 47 years who were surveyed in November and December of 2021 during the COVID-19 Omicron subvariant outbreak at the cosmetic dermatology practices of Sabrina G. Fabi, MD, in San Diego, and Nicole Kanaris, MBBCh, in Johannesburg, South Africa.
“On average, these patients were undergoing six treatments per year during four visits per year, so these were frequent flyers,” Dr. Chopra said. “We set out to assess: Are patients who seek cosmetic procedures happy at baseline? And, do cosmetic procedures make us happier or more satisfied with life?”
Prior to treatment, patients completed the Subjective Happiness Scale (SHS) and Satisfaction With Life Scale (SWLS). Three weeks later, patients completed the SHS, SWLS, the Global Aesthetic Improvement Scale (GAIS) and a 5-point satisfaction score. The researchers used paired and unpaired t-tests, independent sample t-tests, and Spearman rank correlations to conduct statistical analyses.
The baseline SHS score of study participants was an average of 5.87, which Dr. Chopra said is higher than the worldwide population range between 4.57 and 5.33, and 5.05 in the U.S. population. “The patients in our study were very happy to begin with,” an important point to consider, he said. Following their treatments, respondents felt “improved” or “much improved” on the GAIS (a mean score of 3.64) and “somewhat satisfied” or “very satisfied” based on the SWLS (a mean score of 4.4). “So overall, they viewed their treatments as a success,” Dr. Chopra said.
In terms of happiness, however, the researchers observed no significant differences between pre- and posttreatment scores on the SHS (a mean of 5.87 vs. 6.61, respectively; P = .634) nor on the SWLS (a mean of 29.62 vs. 29.1; P = .709). On stratified analysis, no significant differences in the SHS, SWLS, and the GAIS were observed when the researchers accounted for the aggressiveness of the procedure, the number of treatments, the number of sites treated, the type of treatment, and whether the respondents were happier or sadder at baseline. “Surprisingly, this had no effect whatsoever on happiness,” he said. “Not only that, these factors didn’t improve a patient’s perception of the efficacy or satisfaction with a treatment either.”
According to Dr. Chopra, this is the first study to evaluate the impact of a broad spectrum of minimally invasive cosmetic procedures, including injectables and lasers, on the happiness and life satisfaction of treatment non-naive patients.
“Surprisingly, we found these patients were no happier after treatment,” he told this news organization. “However, before rushing to declare that cosmetic procedures don’t make us happier, it is critical to evaluate these results in the context of our study population. We believe there to be a distinction between treatment naive and non-naive patients. All the patients in our study were treatment non-naive, routinely and frequently undergoing cosmetic procedures. Moreover, our treatment non-naive patients were very happy at baseline prior to treatment.”
He and his colleagues hypothesize that there is a “ceiling effect” to the happiness one can attain via these procedures. “Our treatment non-naive patients had already reached this ceiling-peak happiness of their treatment journey, and at this point were only pursuing procedures to maintain their results and happiness,” he said. “Thus, we were unable to measure any effect this late in the ‘maintenance-phase’ of their journey via our study. On the other hand, treatment naive patients (those who have never undergone a cosmetic procedure) were not included. We hypothesize that evaluating patients at the start of their journey after their first round of treatments will demonstrate an impact on happiness, prior to reaching the ceiling and subsequent ‘maintenance phase.’ ”
Lawrence J. Green, MD, clinical professor of dermatology at George Washington University, Washington, who was asked to comment on the study results, said that it was not clear which specific cosmetic treatments the study participants received. “I would like to see if different injectable or device treatments would give different happiness scale results,” Dr. Green said.
“In addition, only two locations were surveyed, so the results could have location bias. I think it would be a great idea to replicate this survey of experienced cosmetic treatment patients with many locations and to include survey responses based on the procedure that was done. That said, it is interesting that overall, investigator satisfaction did not correlate with patient happiness from the treatments.”
Dr. Chopra reported having no financial disclosures. Dr. Green disclosed that he is a speaker, consultant, or investigator for numerous pharmaceutical companies.
AT ASDS 2022
Would a national provider directory save docs’ time, help patients?
When a consumer uses a health plan provider directory to look up a physician, there’s a high probability that the entry for that doctor is incomplete or inaccurate. The Centers for Medicare & Medicaid Services would like to change that by creating a National Directory of Healthcare Providers and Services, which the agency believes would be more valuable to consumers.
In asking for public comments on whether and how it should establish the directory, CMS argues that this data repository would help patients locate physicians and could help with care coordination, health information exchange, and public health data reporting.
However, it’s not clear that such a directory would be any better than current insurance company listings or that people would use it. But a national directory could benefit physician practices by reducing their administrative work, according to observers.
In requesting public comment on the proposed national directory, CMS explains that provider organizations face “redundant and burdensome reporting requirements to multiple databases.” The directory could greatly reduce this challenge by requiring health care organizations to report provider information to a single database. Currently, physician practices have to submit these data to an average of 20 payers each, according to CMS.
“Right now, [physicians are] inundated with requests, and it takes a lot of time to update this stuff,” said David Zetter, a practice management consultant in Mechanicsburg, Pa.. “If there were one national repository of this information, that would be a good move.”
CMS envisions the National Directory as a central hub from which payers could obtain the latest provider data, which would be updated through a standardized application programming interface (API). Consequently, the insurers would no longer need to have providers submit this information to them separately.
CMS is soliciting input on what should be included in the directory. It notes that in addition to contact information, insurer directories also include a physicians’ specialties, health plan affiliations, and whether they accept new patients.
CMS’ 60-day public comment period ends Dec. 6. After that, the agency will decide what steps to take if it is decided that CMS has the legal authority to create the directory.
Terrible track record
In its annual reviews of health plan directories, CMS found that, from 2017 to 2022, only 47% of provider entries were complete. Only 73% of the providers could be matched to published directories. And only 28% of the provider names, addresses, and specialties in the directories matched those in the National Provider Identifier (NPI) registry.
Many of the mistakes in provider directories stem from errors made by practice staff, who have many other duties besides updating directory data. Yet an astonishing amount of time and effort is devoted to this task. A 2019 survey found that physician practices spend $2.76 billion annually on directory maintenance, or nearly $1000 per month per practice, on average.
The Council for Affordable Quality Healthcare, which conducted the survey, estimated that placing all directory data collection on a single platform could save the average practice $4,746 per year. For all practices in the United States, that works out to about $1.1 billion annually, CAQH said.
Pros and cons of national directory
For all the money spent on maintaining provider directories, consumers don’t use them very much. According to a 2021 Press Ganey survey, fewer than 5% of consumers seeking a primary care doctor get their information from an insurer or a benefits manager. About half search the internet first, and 24% seek a referral from a physician.
A national provider directory would be useful only if it were done right, Mr. Zetter said. Citing the inaccuracy and incompleteness of health plan directories, he said it was likely that a national directory would have similar problems. Data entered by practice staff would have to be automatically validated, perhaps through use of some kind of AI algorithm.
Effect on coordination of care
Mr. Zetter doubts the directory could improve care coordination, because primary care doctors usually refer patients to specialists they already know.
But Julia Adler-Milstein, PhD, professor of medicine and director of the Center for Clinical Informatics at the University of California, San Francisco, said that a national directory could improve communications among providers when patients select specialists outside of their primary care physician’s referral network.
“Especially if it’s not an established referral relationship, that’s where a national directory would be helpful, not only to locate the physicians but also to understand their preferences in how they’d like to receive information,” she said in an interview.
Dr. Adler-Milstein worries less than Mr. Zetter does about the challenge of ensuring the accuracy of data in the directory. She pointed out that the National Plan and Provider Enumeration System, which includes the NPI registry, has done a good job of validating provider name, address, and specialty information.
Dr. Adler-Milstein is more concerned about whether the proposed directory would address physician preferences as to how they wish to receive information. For example, while some physicians may prefer to be contacted directly, others may prefer or are required to communicate through their practices or health systems.
Efficiency in data exchange
The API used by the proposed directory would be based on the Fast Health Interoperability Resources standard that all electronic health record vendors must now include in their products. That raises the question of whether communications using contact information from the directory would be sent through a secure email system or through integrated EHR systems, Dr. Adler-Milstein said.
“I’m not sure whether the directory could support that [integration],” she said. “If it focuses on the concept of secure email exchange, that’s a relatively inefficient way of doing it,” because providers want clinical messages to pop up in their EHR workflow rather than their inboxes.
Nevertheless, Dr. Milstein-Adler added, the directory “would clearly take a lot of today’s manual work out of the system. I think organizations like UCSF would be very motivated to support the directory, knowing that people were going to a single source to find the updated information, including preferences in how we’d like people to communicate with us. There would be a lot of efficiency reasons for organizations to use this national directory.”
A version of this article first appeared on Medscape.com.
When a consumer uses a health plan provider directory to look up a physician, there’s a high probability that the entry for that doctor is incomplete or inaccurate. The Centers for Medicare & Medicaid Services would like to change that by creating a National Directory of Healthcare Providers and Services, which the agency believes would be more valuable to consumers.
In asking for public comments on whether and how it should establish the directory, CMS argues that this data repository would help patients locate physicians and could help with care coordination, health information exchange, and public health data reporting.
However, it’s not clear that such a directory would be any better than current insurance company listings or that people would use it. But a national directory could benefit physician practices by reducing their administrative work, according to observers.
In requesting public comment on the proposed national directory, CMS explains that provider organizations face “redundant and burdensome reporting requirements to multiple databases.” The directory could greatly reduce this challenge by requiring health care organizations to report provider information to a single database. Currently, physician practices have to submit these data to an average of 20 payers each, according to CMS.
“Right now, [physicians are] inundated with requests, and it takes a lot of time to update this stuff,” said David Zetter, a practice management consultant in Mechanicsburg, Pa.. “If there were one national repository of this information, that would be a good move.”
CMS envisions the National Directory as a central hub from which payers could obtain the latest provider data, which would be updated through a standardized application programming interface (API). Consequently, the insurers would no longer need to have providers submit this information to them separately.
CMS is soliciting input on what should be included in the directory. It notes that in addition to contact information, insurer directories also include a physicians’ specialties, health plan affiliations, and whether they accept new patients.
CMS’ 60-day public comment period ends Dec. 6. After that, the agency will decide what steps to take if it is decided that CMS has the legal authority to create the directory.
Terrible track record
In its annual reviews of health plan directories, CMS found that, from 2017 to 2022, only 47% of provider entries were complete. Only 73% of the providers could be matched to published directories. And only 28% of the provider names, addresses, and specialties in the directories matched those in the National Provider Identifier (NPI) registry.
Many of the mistakes in provider directories stem from errors made by practice staff, who have many other duties besides updating directory data. Yet an astonishing amount of time and effort is devoted to this task. A 2019 survey found that physician practices spend $2.76 billion annually on directory maintenance, or nearly $1000 per month per practice, on average.
The Council for Affordable Quality Healthcare, which conducted the survey, estimated that placing all directory data collection on a single platform could save the average practice $4,746 per year. For all practices in the United States, that works out to about $1.1 billion annually, CAQH said.
Pros and cons of national directory
For all the money spent on maintaining provider directories, consumers don’t use them very much. According to a 2021 Press Ganey survey, fewer than 5% of consumers seeking a primary care doctor get their information from an insurer or a benefits manager. About half search the internet first, and 24% seek a referral from a physician.
A national provider directory would be useful only if it were done right, Mr. Zetter said. Citing the inaccuracy and incompleteness of health plan directories, he said it was likely that a national directory would have similar problems. Data entered by practice staff would have to be automatically validated, perhaps through use of some kind of AI algorithm.
Effect on coordination of care
Mr. Zetter doubts the directory could improve care coordination, because primary care doctors usually refer patients to specialists they already know.
But Julia Adler-Milstein, PhD, professor of medicine and director of the Center for Clinical Informatics at the University of California, San Francisco, said that a national directory could improve communications among providers when patients select specialists outside of their primary care physician’s referral network.
“Especially if it’s not an established referral relationship, that’s where a national directory would be helpful, not only to locate the physicians but also to understand their preferences in how they’d like to receive information,” she said in an interview.
Dr. Adler-Milstein worries less than Mr. Zetter does about the challenge of ensuring the accuracy of data in the directory. She pointed out that the National Plan and Provider Enumeration System, which includes the NPI registry, has done a good job of validating provider name, address, and specialty information.
Dr. Adler-Milstein is more concerned about whether the proposed directory would address physician preferences as to how they wish to receive information. For example, while some physicians may prefer to be contacted directly, others may prefer or are required to communicate through their practices or health systems.
Efficiency in data exchange
The API used by the proposed directory would be based on the Fast Health Interoperability Resources standard that all electronic health record vendors must now include in their products. That raises the question of whether communications using contact information from the directory would be sent through a secure email system or through integrated EHR systems, Dr. Adler-Milstein said.
“I’m not sure whether the directory could support that [integration],” she said. “If it focuses on the concept of secure email exchange, that’s a relatively inefficient way of doing it,” because providers want clinical messages to pop up in their EHR workflow rather than their inboxes.
Nevertheless, Dr. Milstein-Adler added, the directory “would clearly take a lot of today’s manual work out of the system. I think organizations like UCSF would be very motivated to support the directory, knowing that people were going to a single source to find the updated information, including preferences in how we’d like people to communicate with us. There would be a lot of efficiency reasons for organizations to use this national directory.”
A version of this article first appeared on Medscape.com.
When a consumer uses a health plan provider directory to look up a physician, there’s a high probability that the entry for that doctor is incomplete or inaccurate. The Centers for Medicare & Medicaid Services would like to change that by creating a National Directory of Healthcare Providers and Services, which the agency believes would be more valuable to consumers.
In asking for public comments on whether and how it should establish the directory, CMS argues that this data repository would help patients locate physicians and could help with care coordination, health information exchange, and public health data reporting.
However, it’s not clear that such a directory would be any better than current insurance company listings or that people would use it. But a national directory could benefit physician practices by reducing their administrative work, according to observers.
In requesting public comment on the proposed national directory, CMS explains that provider organizations face “redundant and burdensome reporting requirements to multiple databases.” The directory could greatly reduce this challenge by requiring health care organizations to report provider information to a single database. Currently, physician practices have to submit these data to an average of 20 payers each, according to CMS.
“Right now, [physicians are] inundated with requests, and it takes a lot of time to update this stuff,” said David Zetter, a practice management consultant in Mechanicsburg, Pa.. “If there were one national repository of this information, that would be a good move.”
CMS envisions the National Directory as a central hub from which payers could obtain the latest provider data, which would be updated through a standardized application programming interface (API). Consequently, the insurers would no longer need to have providers submit this information to them separately.
CMS is soliciting input on what should be included in the directory. It notes that in addition to contact information, insurer directories also include a physicians’ specialties, health plan affiliations, and whether they accept new patients.
CMS’ 60-day public comment period ends Dec. 6. After that, the agency will decide what steps to take if it is decided that CMS has the legal authority to create the directory.
Terrible track record
In its annual reviews of health plan directories, CMS found that, from 2017 to 2022, only 47% of provider entries were complete. Only 73% of the providers could be matched to published directories. And only 28% of the provider names, addresses, and specialties in the directories matched those in the National Provider Identifier (NPI) registry.
Many of the mistakes in provider directories stem from errors made by practice staff, who have many other duties besides updating directory data. Yet an astonishing amount of time and effort is devoted to this task. A 2019 survey found that physician practices spend $2.76 billion annually on directory maintenance, or nearly $1000 per month per practice, on average.
The Council for Affordable Quality Healthcare, which conducted the survey, estimated that placing all directory data collection on a single platform could save the average practice $4,746 per year. For all practices in the United States, that works out to about $1.1 billion annually, CAQH said.
Pros and cons of national directory
For all the money spent on maintaining provider directories, consumers don’t use them very much. According to a 2021 Press Ganey survey, fewer than 5% of consumers seeking a primary care doctor get their information from an insurer or a benefits manager. About half search the internet first, and 24% seek a referral from a physician.
A national provider directory would be useful only if it were done right, Mr. Zetter said. Citing the inaccuracy and incompleteness of health plan directories, he said it was likely that a national directory would have similar problems. Data entered by practice staff would have to be automatically validated, perhaps through use of some kind of AI algorithm.
Effect on coordination of care
Mr. Zetter doubts the directory could improve care coordination, because primary care doctors usually refer patients to specialists they already know.
But Julia Adler-Milstein, PhD, professor of medicine and director of the Center for Clinical Informatics at the University of California, San Francisco, said that a national directory could improve communications among providers when patients select specialists outside of their primary care physician’s referral network.
“Especially if it’s not an established referral relationship, that’s where a national directory would be helpful, not only to locate the physicians but also to understand their preferences in how they’d like to receive information,” she said in an interview.
Dr. Adler-Milstein worries less than Mr. Zetter does about the challenge of ensuring the accuracy of data in the directory. She pointed out that the National Plan and Provider Enumeration System, which includes the NPI registry, has done a good job of validating provider name, address, and specialty information.
Dr. Adler-Milstein is more concerned about whether the proposed directory would address physician preferences as to how they wish to receive information. For example, while some physicians may prefer to be contacted directly, others may prefer or are required to communicate through their practices or health systems.
Efficiency in data exchange
The API used by the proposed directory would be based on the Fast Health Interoperability Resources standard that all electronic health record vendors must now include in their products. That raises the question of whether communications using contact information from the directory would be sent through a secure email system or through integrated EHR systems, Dr. Adler-Milstein said.
“I’m not sure whether the directory could support that [integration],” she said. “If it focuses on the concept of secure email exchange, that’s a relatively inefficient way of doing it,” because providers want clinical messages to pop up in their EHR workflow rather than their inboxes.
Nevertheless, Dr. Milstein-Adler added, the directory “would clearly take a lot of today’s manual work out of the system. I think organizations like UCSF would be very motivated to support the directory, knowing that people were going to a single source to find the updated information, including preferences in how we’d like people to communicate with us. There would be a lot of efficiency reasons for organizations to use this national directory.”
A version of this article first appeared on Medscape.com.
From Frankenstein to Lecter: Hollywood’s baddest docs
Masks can be scary on Halloween, but more so when they come with scrubs, scalpels, and God complexes. In March, Medscape readers chose their favorite characters and performers in the Hollywood health care system. from a deep bench (and no, Dr Evil didn’t go to medical school; neither did Dr No, for that matter). Before you see these folks who’d rather haunt than heal, we urge you to seek a second opinion.
George Harris (Richard Widmark, “Coma,” 1978)
“Medicine is now a great social force,” says Dr. George Harris (Richard Widmark), chief of surgery at Boston Memorial. Because the public trusts doctors, “we’ll make the hard decisions” – like choosing which young, healthy patients to put into an irreversible coma to harvest their organs. Harris’ audience of one here is Dr. Susan Wheeler (Genevieve Bujold), the upstart who has uncovered his plot, and whom Harris has just drugged to prepare her as his next unintentional donor. “Coma” was based on a bestseller by Robin Cook and directed by Michael Crichton, who left Harvard Medical School for a career in popular books and films, including “The Andromeda Strain” and “Jurassic Park.” Although Dr. Harris starts out as a reassuring friend and mentor to Dr. Wheeler, older moviegoers won’t forget that he launched to stardom by tossing a woman in a wheelchair down the stairs in 1947’s “Kiss of Death.”
Christian Szell (Laurence Olivier, “Marathon Man,” 1976)
He may look harmless, but Christian Szell (Laurence Olivier) is a sadist with a secret, a stash, and throat-slitting skills. Szell, a dentist known as the White Angel of Auschwitz for his war crimes, stops at nothing to protect the diamonds he stole from his victims in the camps. In one of Hollywood’s most infamous torture scenes, Szell tries to extract information from Babe Levy (Dustin Hoffman), an innocent grad student, plying the tools of his trade. When Szell asks, “Is it safe?” he’s not curious about whether Babe’s insurance covers anesthesia.
Orin Scrivello (Steve Martin, “Little Shop of Horrors,” 1986)
Sticking with deranged dentists, Orin Scrivello, DDS, (Steve Martin) sings and dances his way into your nightmares buoyed by copious helpings of nitrous oxide. Orin’s too-encouraging momma told him to parlay his sadistic tendencies into a career “where people will pay you to be inhumane.” Sonny listened. Moviegoers were treated to screeching sound effects of a tooth getting yanked during an Elvis-like musical number shot in part from inside a patient’s mouth. Martin makes a creepy scene more fun than a long, slow root canal.
Henry Frankenstein (Colin Clive, “Frankenstein,” 1931)
His alarming need for fresh corpses forced Henry Frankenstein (Colin Clive) to leave medical school and experiment solo in a castle. He insists to his betrothed that he hasn’t gone mad when she arrives as he is bringing a dead body back to life during a raging lightning storm. When she and Henry’s mentor, Dr Waldman, witness him succeed, Waldman warns Henry that the former owner of the purloined brain was a notorious criminal. When Henry exclaims: “It’s alive, it’s alive !” little did he know that he created the face (Boris Karloff) that would launch a thousand sequels, a spectacular satire, and untold Halloween masks.
Dr. Gogol (Peter Lorre, “Mad Love,” 1935)
A few years after playing doctor Frankenstein, Colin Clive became the patient of a mad medic himself. A concert pianist whose hands have been mangled in a train wreck, Clive’s wife turns to Dr. Gogol (Peter Lorre, in his Hollywood debut), who promises to surgically reattach the musician’s hands. Unfortunately, Gogol is so obsessed with the wife, a star of gory stage shows, that he has created a wax figure of her. He schemes to win her in the flesh by attaching a murderer’s hands to Clive, then frame him for committing murder with those hands. Gogol utters the madman’s lament: “I have conquered science. Why can’t I conquer love?” A modern remake would surely have him asking, “Why do they swipe left?
Hannibal Lecter (Anthony Hopkins, “Silence of the Lambs,” 1991)
The FBI, hunting for a serial killer, sends trainee Clarice Starling (Jodie Foster) to seek insight into the murderer from the imprisoned Dr. Hannibal Lecter (Anthony Hopkins), a brilliant psychiatrist with a penchant for murder — and a taste for the flesh of his victims. Lecter proves to be a menace from their first meeting; the bars and glass surrounding his cell offer Clarice no protection from his gaze and ability to read her mind. In his own way, the urbane, pathologically charming Lecter takes a shine to Clarice, helping with the case while embarking on another murderous spree against men who recently wronged her. When he escapes, his plans do not include dinner with – or of – Clarice, but others, well, they’re not so lucky.
Henry Jekyll (Fredric March, “Dr. Jekyll and Mr. Hyde,” 1931)
Henry Jekyll (Fredric March) is a jumble of personalities. By day, he’s a kindly doctor in Victorian London with an American accent. But he is so determined to split good and evil personalities that he devises a potion to outsource his id. As he watches himself morph into Mr. Hyde – a hairy, cone-headed dude in serious need of an orthodontist – he exclaims, “Free! Free at last!” Free, that is, for his simian side to engage in debauchery, abuse, self-hatred, intimations of rape, and ultimately murder – all of which are explored in this pre-Code film, the first talkie version of Robert Louis Stevenson’s story.
Dr. Moreau (Charles Laughton, “Island of Lost Souls,” 1932)
“Strange-looking natives you have here,” shipwreck victim Edward Parker (Richard Arlen) tells his host, the white-suited, whip-wielding Dr Moreau. Before long, we learn that Moreau’s evil veterinary talents have created an island population of human/beast hybrids who are forced to follow his laws – especially one forbidding them from eating meat or walking on all fours. Lawbreakers get taken to the House of Pain, a medical setting which, as its name suggests, lacks adequate analgesia. Burt Lancaster and Marlon Brando took on the Moreau role in later versions, but Laughton is the creepiest when he asks, “Do you know what it means to feel like God?” The film was banned for years in Britain, and H.G. Wells despised this take on his antivivisection tale.
Charles Nichols (Jeroen Krabbé, “The Fugitive,” 1993)
Richard Kimble, a Chicago vascular surgeon, arrives home to find that a man just brutally murdered his loving wife. The killer escapes, and Kimble falls into the frame-up. Convicted for the murder and headed to prison, Kimble breaks free in an epic escape scene. He spends the rest of the movie all but giving his right arm to find the murderer, while being pursued by a dogged U.S. Marshal played with gusto by Tommy Lee Jones. Kimble eventually discovers that his colleague, Dr. Charles Nichols (Jeroen Krabbé), is not quite the best friend a man could have – or the most ethical of clinical investigators.
Elliot and Beverly Mantle (Jeremy Irons, “Dead Ringers,” 1988)
“You’ve got to try the movie star,” fertility specialist Elliot Mantle (Jeremy Irons) implores to his identical but meek twin brother, Beverly (also Jeremy Irons), talking about an actress-patient (Genevieve Bujold) as if she were a menu item. Beverly shares a practice with Elliot, along with a soul and an easily satisfied drug addiction. Beverly is unaware that Elliot seduces patients before passing them off to his brother, including the actress. Beverly is in love with the actress, which upsets the equilibrium of their shared soul. He aims to fix this, but not without some trauma involving freakish and unsanitary operating implements.
Dean Armitage (Bradley Whitford, “Get Out,” 2017)
Neurosurgeon Dean Armitage (Bradley Whitford) was such a fan of President Obama that he would have voted for him a third time if he could. At least, that’s how he portrays himself to Chris (Daniel Kaluuya), an African American photographer and the new boyfriend of Armitage’s White daughter. The Armitage estate has plenty of people of color – on staff, anyway – but Chris finds them odd and distant. It turns out that a gathering of rich White people is in fact an auction for his eyesight. Horror ensues. The main message from this film is not unlike that of Russian operatives who fall out of favor with the Kremlin: Don’t drink the tea.
A version of this article first appeared on Medscape.com.
Masks can be scary on Halloween, but more so when they come with scrubs, scalpels, and God complexes. In March, Medscape readers chose their favorite characters and performers in the Hollywood health care system. from a deep bench (and no, Dr Evil didn’t go to medical school; neither did Dr No, for that matter). Before you see these folks who’d rather haunt than heal, we urge you to seek a second opinion.
George Harris (Richard Widmark, “Coma,” 1978)
“Medicine is now a great social force,” says Dr. George Harris (Richard Widmark), chief of surgery at Boston Memorial. Because the public trusts doctors, “we’ll make the hard decisions” – like choosing which young, healthy patients to put into an irreversible coma to harvest their organs. Harris’ audience of one here is Dr. Susan Wheeler (Genevieve Bujold), the upstart who has uncovered his plot, and whom Harris has just drugged to prepare her as his next unintentional donor. “Coma” was based on a bestseller by Robin Cook and directed by Michael Crichton, who left Harvard Medical School for a career in popular books and films, including “The Andromeda Strain” and “Jurassic Park.” Although Dr. Harris starts out as a reassuring friend and mentor to Dr. Wheeler, older moviegoers won’t forget that he launched to stardom by tossing a woman in a wheelchair down the stairs in 1947’s “Kiss of Death.”
Christian Szell (Laurence Olivier, “Marathon Man,” 1976)
He may look harmless, but Christian Szell (Laurence Olivier) is a sadist with a secret, a stash, and throat-slitting skills. Szell, a dentist known as the White Angel of Auschwitz for his war crimes, stops at nothing to protect the diamonds he stole from his victims in the camps. In one of Hollywood’s most infamous torture scenes, Szell tries to extract information from Babe Levy (Dustin Hoffman), an innocent grad student, plying the tools of his trade. When Szell asks, “Is it safe?” he’s not curious about whether Babe’s insurance covers anesthesia.
Orin Scrivello (Steve Martin, “Little Shop of Horrors,” 1986)
Sticking with deranged dentists, Orin Scrivello, DDS, (Steve Martin) sings and dances his way into your nightmares buoyed by copious helpings of nitrous oxide. Orin’s too-encouraging momma told him to parlay his sadistic tendencies into a career “where people will pay you to be inhumane.” Sonny listened. Moviegoers were treated to screeching sound effects of a tooth getting yanked during an Elvis-like musical number shot in part from inside a patient’s mouth. Martin makes a creepy scene more fun than a long, slow root canal.
Henry Frankenstein (Colin Clive, “Frankenstein,” 1931)
His alarming need for fresh corpses forced Henry Frankenstein (Colin Clive) to leave medical school and experiment solo in a castle. He insists to his betrothed that he hasn’t gone mad when she arrives as he is bringing a dead body back to life during a raging lightning storm. When she and Henry’s mentor, Dr Waldman, witness him succeed, Waldman warns Henry that the former owner of the purloined brain was a notorious criminal. When Henry exclaims: “It’s alive, it’s alive !” little did he know that he created the face (Boris Karloff) that would launch a thousand sequels, a spectacular satire, and untold Halloween masks.
Dr. Gogol (Peter Lorre, “Mad Love,” 1935)
A few years after playing doctor Frankenstein, Colin Clive became the patient of a mad medic himself. A concert pianist whose hands have been mangled in a train wreck, Clive’s wife turns to Dr. Gogol (Peter Lorre, in his Hollywood debut), who promises to surgically reattach the musician’s hands. Unfortunately, Gogol is so obsessed with the wife, a star of gory stage shows, that he has created a wax figure of her. He schemes to win her in the flesh by attaching a murderer’s hands to Clive, then frame him for committing murder with those hands. Gogol utters the madman’s lament: “I have conquered science. Why can’t I conquer love?” A modern remake would surely have him asking, “Why do they swipe left?
Hannibal Lecter (Anthony Hopkins, “Silence of the Lambs,” 1991)
The FBI, hunting for a serial killer, sends trainee Clarice Starling (Jodie Foster) to seek insight into the murderer from the imprisoned Dr. Hannibal Lecter (Anthony Hopkins), a brilliant psychiatrist with a penchant for murder — and a taste for the flesh of his victims. Lecter proves to be a menace from their first meeting; the bars and glass surrounding his cell offer Clarice no protection from his gaze and ability to read her mind. In his own way, the urbane, pathologically charming Lecter takes a shine to Clarice, helping with the case while embarking on another murderous spree against men who recently wronged her. When he escapes, his plans do not include dinner with – or of – Clarice, but others, well, they’re not so lucky.
Henry Jekyll (Fredric March, “Dr. Jekyll and Mr. Hyde,” 1931)
Henry Jekyll (Fredric March) is a jumble of personalities. By day, he’s a kindly doctor in Victorian London with an American accent. But he is so determined to split good and evil personalities that he devises a potion to outsource his id. As he watches himself morph into Mr. Hyde – a hairy, cone-headed dude in serious need of an orthodontist – he exclaims, “Free! Free at last!” Free, that is, for his simian side to engage in debauchery, abuse, self-hatred, intimations of rape, and ultimately murder – all of which are explored in this pre-Code film, the first talkie version of Robert Louis Stevenson’s story.
Dr. Moreau (Charles Laughton, “Island of Lost Souls,” 1932)
“Strange-looking natives you have here,” shipwreck victim Edward Parker (Richard Arlen) tells his host, the white-suited, whip-wielding Dr Moreau. Before long, we learn that Moreau’s evil veterinary talents have created an island population of human/beast hybrids who are forced to follow his laws – especially one forbidding them from eating meat or walking on all fours. Lawbreakers get taken to the House of Pain, a medical setting which, as its name suggests, lacks adequate analgesia. Burt Lancaster and Marlon Brando took on the Moreau role in later versions, but Laughton is the creepiest when he asks, “Do you know what it means to feel like God?” The film was banned for years in Britain, and H.G. Wells despised this take on his antivivisection tale.
Charles Nichols (Jeroen Krabbé, “The Fugitive,” 1993)
Richard Kimble, a Chicago vascular surgeon, arrives home to find that a man just brutally murdered his loving wife. The killer escapes, and Kimble falls into the frame-up. Convicted for the murder and headed to prison, Kimble breaks free in an epic escape scene. He spends the rest of the movie all but giving his right arm to find the murderer, while being pursued by a dogged U.S. Marshal played with gusto by Tommy Lee Jones. Kimble eventually discovers that his colleague, Dr. Charles Nichols (Jeroen Krabbé), is not quite the best friend a man could have – or the most ethical of clinical investigators.
Elliot and Beverly Mantle (Jeremy Irons, “Dead Ringers,” 1988)
“You’ve got to try the movie star,” fertility specialist Elliot Mantle (Jeremy Irons) implores to his identical but meek twin brother, Beverly (also Jeremy Irons), talking about an actress-patient (Genevieve Bujold) as if she were a menu item. Beverly shares a practice with Elliot, along with a soul and an easily satisfied drug addiction. Beverly is unaware that Elliot seduces patients before passing them off to his brother, including the actress. Beverly is in love with the actress, which upsets the equilibrium of their shared soul. He aims to fix this, but not without some trauma involving freakish and unsanitary operating implements.
Dean Armitage (Bradley Whitford, “Get Out,” 2017)
Neurosurgeon Dean Armitage (Bradley Whitford) was such a fan of President Obama that he would have voted for him a third time if he could. At least, that’s how he portrays himself to Chris (Daniel Kaluuya), an African American photographer and the new boyfriend of Armitage’s White daughter. The Armitage estate has plenty of people of color – on staff, anyway – but Chris finds them odd and distant. It turns out that a gathering of rich White people is in fact an auction for his eyesight. Horror ensues. The main message from this film is not unlike that of Russian operatives who fall out of favor with the Kremlin: Don’t drink the tea.
A version of this article first appeared on Medscape.com.
Masks can be scary on Halloween, but more so when they come with scrubs, scalpels, and God complexes. In March, Medscape readers chose their favorite characters and performers in the Hollywood health care system. from a deep bench (and no, Dr Evil didn’t go to medical school; neither did Dr No, for that matter). Before you see these folks who’d rather haunt than heal, we urge you to seek a second opinion.
George Harris (Richard Widmark, “Coma,” 1978)
“Medicine is now a great social force,” says Dr. George Harris (Richard Widmark), chief of surgery at Boston Memorial. Because the public trusts doctors, “we’ll make the hard decisions” – like choosing which young, healthy patients to put into an irreversible coma to harvest their organs. Harris’ audience of one here is Dr. Susan Wheeler (Genevieve Bujold), the upstart who has uncovered his plot, and whom Harris has just drugged to prepare her as his next unintentional donor. “Coma” was based on a bestseller by Robin Cook and directed by Michael Crichton, who left Harvard Medical School for a career in popular books and films, including “The Andromeda Strain” and “Jurassic Park.” Although Dr. Harris starts out as a reassuring friend and mentor to Dr. Wheeler, older moviegoers won’t forget that he launched to stardom by tossing a woman in a wheelchair down the stairs in 1947’s “Kiss of Death.”
Christian Szell (Laurence Olivier, “Marathon Man,” 1976)
He may look harmless, but Christian Szell (Laurence Olivier) is a sadist with a secret, a stash, and throat-slitting skills. Szell, a dentist known as the White Angel of Auschwitz for his war crimes, stops at nothing to protect the diamonds he stole from his victims in the camps. In one of Hollywood’s most infamous torture scenes, Szell tries to extract information from Babe Levy (Dustin Hoffman), an innocent grad student, plying the tools of his trade. When Szell asks, “Is it safe?” he’s not curious about whether Babe’s insurance covers anesthesia.
Orin Scrivello (Steve Martin, “Little Shop of Horrors,” 1986)
Sticking with deranged dentists, Orin Scrivello, DDS, (Steve Martin) sings and dances his way into your nightmares buoyed by copious helpings of nitrous oxide. Orin’s too-encouraging momma told him to parlay his sadistic tendencies into a career “where people will pay you to be inhumane.” Sonny listened. Moviegoers were treated to screeching sound effects of a tooth getting yanked during an Elvis-like musical number shot in part from inside a patient’s mouth. Martin makes a creepy scene more fun than a long, slow root canal.
Henry Frankenstein (Colin Clive, “Frankenstein,” 1931)
His alarming need for fresh corpses forced Henry Frankenstein (Colin Clive) to leave medical school and experiment solo in a castle. He insists to his betrothed that he hasn’t gone mad when she arrives as he is bringing a dead body back to life during a raging lightning storm. When she and Henry’s mentor, Dr Waldman, witness him succeed, Waldman warns Henry that the former owner of the purloined brain was a notorious criminal. When Henry exclaims: “It’s alive, it’s alive !” little did he know that he created the face (Boris Karloff) that would launch a thousand sequels, a spectacular satire, and untold Halloween masks.
Dr. Gogol (Peter Lorre, “Mad Love,” 1935)
A few years after playing doctor Frankenstein, Colin Clive became the patient of a mad medic himself. A concert pianist whose hands have been mangled in a train wreck, Clive’s wife turns to Dr. Gogol (Peter Lorre, in his Hollywood debut), who promises to surgically reattach the musician’s hands. Unfortunately, Gogol is so obsessed with the wife, a star of gory stage shows, that he has created a wax figure of her. He schemes to win her in the flesh by attaching a murderer’s hands to Clive, then frame him for committing murder with those hands. Gogol utters the madman’s lament: “I have conquered science. Why can’t I conquer love?” A modern remake would surely have him asking, “Why do they swipe left?
Hannibal Lecter (Anthony Hopkins, “Silence of the Lambs,” 1991)
The FBI, hunting for a serial killer, sends trainee Clarice Starling (Jodie Foster) to seek insight into the murderer from the imprisoned Dr. Hannibal Lecter (Anthony Hopkins), a brilliant psychiatrist with a penchant for murder — and a taste for the flesh of his victims. Lecter proves to be a menace from their first meeting; the bars and glass surrounding his cell offer Clarice no protection from his gaze and ability to read her mind. In his own way, the urbane, pathologically charming Lecter takes a shine to Clarice, helping with the case while embarking on another murderous spree against men who recently wronged her. When he escapes, his plans do not include dinner with – or of – Clarice, but others, well, they’re not so lucky.
Henry Jekyll (Fredric March, “Dr. Jekyll and Mr. Hyde,” 1931)
Henry Jekyll (Fredric March) is a jumble of personalities. By day, he’s a kindly doctor in Victorian London with an American accent. But he is so determined to split good and evil personalities that he devises a potion to outsource his id. As he watches himself morph into Mr. Hyde – a hairy, cone-headed dude in serious need of an orthodontist – he exclaims, “Free! Free at last!” Free, that is, for his simian side to engage in debauchery, abuse, self-hatred, intimations of rape, and ultimately murder – all of which are explored in this pre-Code film, the first talkie version of Robert Louis Stevenson’s story.
Dr. Moreau (Charles Laughton, “Island of Lost Souls,” 1932)
“Strange-looking natives you have here,” shipwreck victim Edward Parker (Richard Arlen) tells his host, the white-suited, whip-wielding Dr Moreau. Before long, we learn that Moreau’s evil veterinary talents have created an island population of human/beast hybrids who are forced to follow his laws – especially one forbidding them from eating meat or walking on all fours. Lawbreakers get taken to the House of Pain, a medical setting which, as its name suggests, lacks adequate analgesia. Burt Lancaster and Marlon Brando took on the Moreau role in later versions, but Laughton is the creepiest when he asks, “Do you know what it means to feel like God?” The film was banned for years in Britain, and H.G. Wells despised this take on his antivivisection tale.
Charles Nichols (Jeroen Krabbé, “The Fugitive,” 1993)
Richard Kimble, a Chicago vascular surgeon, arrives home to find that a man just brutally murdered his loving wife. The killer escapes, and Kimble falls into the frame-up. Convicted for the murder and headed to prison, Kimble breaks free in an epic escape scene. He spends the rest of the movie all but giving his right arm to find the murderer, while being pursued by a dogged U.S. Marshal played with gusto by Tommy Lee Jones. Kimble eventually discovers that his colleague, Dr. Charles Nichols (Jeroen Krabbé), is not quite the best friend a man could have – or the most ethical of clinical investigators.
Elliot and Beverly Mantle (Jeremy Irons, “Dead Ringers,” 1988)
“You’ve got to try the movie star,” fertility specialist Elliot Mantle (Jeremy Irons) implores to his identical but meek twin brother, Beverly (also Jeremy Irons), talking about an actress-patient (Genevieve Bujold) as if she were a menu item. Beverly shares a practice with Elliot, along with a soul and an easily satisfied drug addiction. Beverly is unaware that Elliot seduces patients before passing them off to his brother, including the actress. Beverly is in love with the actress, which upsets the equilibrium of their shared soul. He aims to fix this, but not without some trauma involving freakish and unsanitary operating implements.
Dean Armitage (Bradley Whitford, “Get Out,” 2017)
Neurosurgeon Dean Armitage (Bradley Whitford) was such a fan of President Obama that he would have voted for him a third time if he could. At least, that’s how he portrays himself to Chris (Daniel Kaluuya), an African American photographer and the new boyfriend of Armitage’s White daughter. The Armitage estate has plenty of people of color – on staff, anyway – but Chris finds them odd and distant. It turns out that a gathering of rich White people is in fact an auction for his eyesight. Horror ensues. The main message from this film is not unlike that of Russian operatives who fall out of favor with the Kremlin: Don’t drink the tea.
A version of this article first appeared on Medscape.com.
Is it flu, RSV, or COVID? Experts fear the ‘tripledemic’
Just when we thought this holiday season, finally, would be the back-to-normal one, some infectious disease experts are warning that a so-called “tripledemic” – influenza, COVID-19, and RSV – may be in the forecast.
The warning isn’t without basis.
The flu season has gotten an early start. As of Oct. 21, early increases in seasonal flu activity have been reported in most of the country, the Centers for Disease Control and Prevention said, with the southeast and south-central areas having the highest activity levels.
Children’s hospitals and EDs are seeing a surge in children with RSV.
COVID-19 cases are trending down, according to the CDC, but epidemiologists – scientists who study disease outbreaks – always have their eyes on emerging variants.
said Justin Lessler, PhD, a professor of epidemiology at the University of North Carolina at Chapel Hill. Dr. Lessler is on the coordinating team for the COVID-19 Scenario Modeling Hub, which aims to predict the course COVID-19, and the Flu Scenario Modeling Hub, which does the same for influenza.
For COVID-19, some models are predicting some spikes before Christmas, he said, and others see a new wave in 2023. For the flu, the model is predicting an earlier-than-usual start, as the CDC has reported.
While flu activity is relatively low, the CDC said, the season is off to an early start. For the week ending Oct. 21, 1,674 patients were hospitalized for flu, higher than in the summer months but fewer than the 2,675 hospitalizations for the week of May 15, 2022.
As of Oct. 20, COVID-19 cases have declined 12% over the last 2 weeks, nationwide. But hospitalizations are up 10% in much of the Northeast, The New York Times reports, and the improvement in cases and deaths has been slowing down.
As of Oct. 15, 15% of RSV tests reported nationwide were positive, compared with about 11% at that time in 2021, the CDC said. The surveillance collects information from 75 counties in 12 states.
Experts point out that the viruses – all three are respiratory viruses – are simply playing catchup.
“They spread the same way and along with lots of other viruses, and you tend to see an increase in them during the cold months,” said Timothy Brewer, MD, professor of medicine and epidemiology at UCLA.
The increase in all three viruses “is almost predictable at this point in the pandemic,” said Dean Blumberg, MD, a professor and chief of pediatric infectious diseases at the University of California Davis Health. “All the respiratory viruses are out of whack.”
Last year, RSV cases were up, too, and began to appear very early, he said, in the summer instead of in the cooler months. Flu also appeared early in 2021, as it has in 2022.
That contrasts with the flu season of 2020-2021, when COVID precautions were nearly universal, and cases were down. At UC Davis, “we didn’t have one pediatric admission due to influenza in the 2020-2021 [flu] season,” Dr. Blumberg said.
The number of pediatric flu deaths usually range from 37 to 199 per year, according to CDC records. But in the 2020-2021 season, the CDC recorded one pediatric flu death in the U.S.
Both children and adults have had less contact with others the past two seasons, Dr. Blumberg said, “and they don’t get the immunity they got with those infections [previously]. That’s why we are seeing out-of-season, early season [viruses].”
Eventually, he said, the cases of flu and RSV will return to previous levels. “It could be as soon as next year,” Dr. Blumberg said. And COVID-19, hopefully, will become like influenza, he said.
“RSV has always come around in the fall and winter,” said Elizabeth Murray, DO, a pediatric emergency medicine doctor at the University of Rochester (N.Y.) Medical Center and a spokesperson for the American Academy of Pediatrics. In 2022, children are back in school and for the most part not masking. “It’s a perfect storm for all the germs to spread now. They’ve just been waiting for their opportunity to come back.”
Self-care vs. not
RSV can pose a risk for anyone, but most at risk are children under age 5, especially infants under age 1, and adults over age 65. There is no vaccine for it. Symptoms include a runny nose, decreased appetite, coughing, sneezing, fever, and wheezing. But in young infants, there may only be decreased activity, crankiness, and breathing issues, the CDC said.
Keep an eye on the breathing if RSV is suspected, Dr. Murray tells parents. If your child can’t breathe easily, is unable to lie down comfortably, can’t speak clearly, or is sucking in the chest muscles to breathe, get medical help. Most kids with RSV can stay home and recover, she said, but often will need to be checked by a medical professional.
She advises against getting an oximeter to measure oxygen levels for home use. “They are often not accurate,” she said. If in doubt about how serious your child’s symptoms are, “don’t wait it out,” and don’t hesitate to call 911.
Symptoms of flu, COVID, and RSV can overlap. But each can involve breathing problems, which can be an emergency.
“It’s important to seek medical attention for any concerning symptoms, but especially severe shortness of breath or difficulty breathing, as these could signal the need for supplemental oxygen or other emergency interventions,” said Mandy De Vries, a respiratory therapist and director of education at the American Association for Respiratory Care. Inhalation treatment or mechanical ventilation may be needed for severe respiratory issues.
Precautions
To avoid the tripledemic – or any single infection – Timothy Brewer, MD, a professor of medicine and epidemiology at the University of California, Los Angeles, suggests some familiar measures: “Stay home if you’re feeling sick. Make sure you are up to date on your vaccinations. Wear a mask indoors.”
A version of this article first appeared on Medscape.com.
Just when we thought this holiday season, finally, would be the back-to-normal one, some infectious disease experts are warning that a so-called “tripledemic” – influenza, COVID-19, and RSV – may be in the forecast.
The warning isn’t without basis.
The flu season has gotten an early start. As of Oct. 21, early increases in seasonal flu activity have been reported in most of the country, the Centers for Disease Control and Prevention said, with the southeast and south-central areas having the highest activity levels.
Children’s hospitals and EDs are seeing a surge in children with RSV.
COVID-19 cases are trending down, according to the CDC, but epidemiologists – scientists who study disease outbreaks – always have their eyes on emerging variants.
said Justin Lessler, PhD, a professor of epidemiology at the University of North Carolina at Chapel Hill. Dr. Lessler is on the coordinating team for the COVID-19 Scenario Modeling Hub, which aims to predict the course COVID-19, and the Flu Scenario Modeling Hub, which does the same for influenza.
For COVID-19, some models are predicting some spikes before Christmas, he said, and others see a new wave in 2023. For the flu, the model is predicting an earlier-than-usual start, as the CDC has reported.
While flu activity is relatively low, the CDC said, the season is off to an early start. For the week ending Oct. 21, 1,674 patients were hospitalized for flu, higher than in the summer months but fewer than the 2,675 hospitalizations for the week of May 15, 2022.
As of Oct. 20, COVID-19 cases have declined 12% over the last 2 weeks, nationwide. But hospitalizations are up 10% in much of the Northeast, The New York Times reports, and the improvement in cases and deaths has been slowing down.
As of Oct. 15, 15% of RSV tests reported nationwide were positive, compared with about 11% at that time in 2021, the CDC said. The surveillance collects information from 75 counties in 12 states.
Experts point out that the viruses – all three are respiratory viruses – are simply playing catchup.
“They spread the same way and along with lots of other viruses, and you tend to see an increase in them during the cold months,” said Timothy Brewer, MD, professor of medicine and epidemiology at UCLA.
The increase in all three viruses “is almost predictable at this point in the pandemic,” said Dean Blumberg, MD, a professor and chief of pediatric infectious diseases at the University of California Davis Health. “All the respiratory viruses are out of whack.”
Last year, RSV cases were up, too, and began to appear very early, he said, in the summer instead of in the cooler months. Flu also appeared early in 2021, as it has in 2022.
That contrasts with the flu season of 2020-2021, when COVID precautions were nearly universal, and cases were down. At UC Davis, “we didn’t have one pediatric admission due to influenza in the 2020-2021 [flu] season,” Dr. Blumberg said.
The number of pediatric flu deaths usually range from 37 to 199 per year, according to CDC records. But in the 2020-2021 season, the CDC recorded one pediatric flu death in the U.S.
Both children and adults have had less contact with others the past two seasons, Dr. Blumberg said, “and they don’t get the immunity they got with those infections [previously]. That’s why we are seeing out-of-season, early season [viruses].”
Eventually, he said, the cases of flu and RSV will return to previous levels. “It could be as soon as next year,” Dr. Blumberg said. And COVID-19, hopefully, will become like influenza, he said.
“RSV has always come around in the fall and winter,” said Elizabeth Murray, DO, a pediatric emergency medicine doctor at the University of Rochester (N.Y.) Medical Center and a spokesperson for the American Academy of Pediatrics. In 2022, children are back in school and for the most part not masking. “It’s a perfect storm for all the germs to spread now. They’ve just been waiting for their opportunity to come back.”
Self-care vs. not
RSV can pose a risk for anyone, but most at risk are children under age 5, especially infants under age 1, and adults over age 65. There is no vaccine for it. Symptoms include a runny nose, decreased appetite, coughing, sneezing, fever, and wheezing. But in young infants, there may only be decreased activity, crankiness, and breathing issues, the CDC said.
Keep an eye on the breathing if RSV is suspected, Dr. Murray tells parents. If your child can’t breathe easily, is unable to lie down comfortably, can’t speak clearly, or is sucking in the chest muscles to breathe, get medical help. Most kids with RSV can stay home and recover, she said, but often will need to be checked by a medical professional.
She advises against getting an oximeter to measure oxygen levels for home use. “They are often not accurate,” she said. If in doubt about how serious your child’s symptoms are, “don’t wait it out,” and don’t hesitate to call 911.
Symptoms of flu, COVID, and RSV can overlap. But each can involve breathing problems, which can be an emergency.
“It’s important to seek medical attention for any concerning symptoms, but especially severe shortness of breath or difficulty breathing, as these could signal the need for supplemental oxygen or other emergency interventions,” said Mandy De Vries, a respiratory therapist and director of education at the American Association for Respiratory Care. Inhalation treatment or mechanical ventilation may be needed for severe respiratory issues.
Precautions
To avoid the tripledemic – or any single infection – Timothy Brewer, MD, a professor of medicine and epidemiology at the University of California, Los Angeles, suggests some familiar measures: “Stay home if you’re feeling sick. Make sure you are up to date on your vaccinations. Wear a mask indoors.”
A version of this article first appeared on Medscape.com.
Just when we thought this holiday season, finally, would be the back-to-normal one, some infectious disease experts are warning that a so-called “tripledemic” – influenza, COVID-19, and RSV – may be in the forecast.
The warning isn’t without basis.
The flu season has gotten an early start. As of Oct. 21, early increases in seasonal flu activity have been reported in most of the country, the Centers for Disease Control and Prevention said, with the southeast and south-central areas having the highest activity levels.
Children’s hospitals and EDs are seeing a surge in children with RSV.
COVID-19 cases are trending down, according to the CDC, but epidemiologists – scientists who study disease outbreaks – always have their eyes on emerging variants.
said Justin Lessler, PhD, a professor of epidemiology at the University of North Carolina at Chapel Hill. Dr. Lessler is on the coordinating team for the COVID-19 Scenario Modeling Hub, which aims to predict the course COVID-19, and the Flu Scenario Modeling Hub, which does the same for influenza.
For COVID-19, some models are predicting some spikes before Christmas, he said, and others see a new wave in 2023. For the flu, the model is predicting an earlier-than-usual start, as the CDC has reported.
While flu activity is relatively low, the CDC said, the season is off to an early start. For the week ending Oct. 21, 1,674 patients were hospitalized for flu, higher than in the summer months but fewer than the 2,675 hospitalizations for the week of May 15, 2022.
As of Oct. 20, COVID-19 cases have declined 12% over the last 2 weeks, nationwide. But hospitalizations are up 10% in much of the Northeast, The New York Times reports, and the improvement in cases and deaths has been slowing down.
As of Oct. 15, 15% of RSV tests reported nationwide were positive, compared with about 11% at that time in 2021, the CDC said. The surveillance collects information from 75 counties in 12 states.
Experts point out that the viruses – all three are respiratory viruses – are simply playing catchup.
“They spread the same way and along with lots of other viruses, and you tend to see an increase in them during the cold months,” said Timothy Brewer, MD, professor of medicine and epidemiology at UCLA.
The increase in all three viruses “is almost predictable at this point in the pandemic,” said Dean Blumberg, MD, a professor and chief of pediatric infectious diseases at the University of California Davis Health. “All the respiratory viruses are out of whack.”
Last year, RSV cases were up, too, and began to appear very early, he said, in the summer instead of in the cooler months. Flu also appeared early in 2021, as it has in 2022.
That contrasts with the flu season of 2020-2021, when COVID precautions were nearly universal, and cases were down. At UC Davis, “we didn’t have one pediatric admission due to influenza in the 2020-2021 [flu] season,” Dr. Blumberg said.
The number of pediatric flu deaths usually range from 37 to 199 per year, according to CDC records. But in the 2020-2021 season, the CDC recorded one pediatric flu death in the U.S.
Both children and adults have had less contact with others the past two seasons, Dr. Blumberg said, “and they don’t get the immunity they got with those infections [previously]. That’s why we are seeing out-of-season, early season [viruses].”
Eventually, he said, the cases of flu and RSV will return to previous levels. “It could be as soon as next year,” Dr. Blumberg said. And COVID-19, hopefully, will become like influenza, he said.
“RSV has always come around in the fall and winter,” said Elizabeth Murray, DO, a pediatric emergency medicine doctor at the University of Rochester (N.Y.) Medical Center and a spokesperson for the American Academy of Pediatrics. In 2022, children are back in school and for the most part not masking. “It’s a perfect storm for all the germs to spread now. They’ve just been waiting for their opportunity to come back.”
Self-care vs. not
RSV can pose a risk for anyone, but most at risk are children under age 5, especially infants under age 1, and adults over age 65. There is no vaccine for it. Symptoms include a runny nose, decreased appetite, coughing, sneezing, fever, and wheezing. But in young infants, there may only be decreased activity, crankiness, and breathing issues, the CDC said.
Keep an eye on the breathing if RSV is suspected, Dr. Murray tells parents. If your child can’t breathe easily, is unable to lie down comfortably, can’t speak clearly, or is sucking in the chest muscles to breathe, get medical help. Most kids with RSV can stay home and recover, she said, but often will need to be checked by a medical professional.
She advises against getting an oximeter to measure oxygen levels for home use. “They are often not accurate,” she said. If in doubt about how serious your child’s symptoms are, “don’t wait it out,” and don’t hesitate to call 911.
Symptoms of flu, COVID, and RSV can overlap. But each can involve breathing problems, which can be an emergency.
“It’s important to seek medical attention for any concerning symptoms, but especially severe shortness of breath or difficulty breathing, as these could signal the need for supplemental oxygen or other emergency interventions,” said Mandy De Vries, a respiratory therapist and director of education at the American Association for Respiratory Care. Inhalation treatment or mechanical ventilation may be needed for severe respiratory issues.
Precautions
To avoid the tripledemic – or any single infection – Timothy Brewer, MD, a professor of medicine and epidemiology at the University of California, Los Angeles, suggests some familiar measures: “Stay home if you’re feeling sick. Make sure you are up to date on your vaccinations. Wear a mask indoors.”
A version of this article first appeared on Medscape.com.
Two biologics equally effective for extraintestinal manifestations of IBD
Vedolizumab (Entyvio) and ustekinumab (Stelara) appear to be equally effective for extraintestinal manifestation (EIM) of inflammatory bowel disease (IBD), according to results of a retrospective study published online in Digestive and Liver Disease.
Between 25% and 40% of patients with IBD experience EIM, which reduces quality of life, according to the Crohn’s & Colitis Foundation. EIM commonly involves the joints, skin, bones, eyes, kidney, and liver. Anemia is another extraintestinal complication.
Until now, it’s been unclear whether vedolizumab and ustekinumab are equally effective for treating EIM.
Vedolizumab specifically targets the gastrointestinal tract, a potential disadvantage in reducing EIM, while ustekinumab is thought to have a systemic effect, a potential treatment advantage, Moran Livne-Margolin, MD, and colleagues, Chaim Sheba Medical Center, Ramat Gan, Israel, point out.
To investigate, they included 111 adults with IBD who were treated at the medical center between 2015 and 2021 – 53 with vedolizumab and 58 with ustekinumab. Before starting treatment, all of them had active EIM, most commonly arthralgia (84%).
After 6 weeks of treatment, 66% of patients in both groups had a clinical response to their intestinal disease.
After 14 and 26 weeks of treatment, clinical response rates were 59% and 50%, respectively, with vedolizumab, and 48% and 41%, respectively, with ustekinumab.
Over 52 weeks, both biologics were equally effective against the intestinal disease, with clinical response rates of 42% with vedolizumab and 44% with ustekinumab.
A similar pattern emerged when looking at improvement in EIM.
At week 6, 44% of patients taking vedolizumab and 35% taking ustekinumab had improvement in EIM, with no significant difference between the two biologics (P = .4).
At week 14, rates of improvement in EIM were 43% for vedolizumab and 33% for ustekinumab (P = .39); at 26 weeks, rates were 39% and 33%, respectively (P = .6); and at 52 weeks, rates were 34% and 36% (P = .9).
Researchers also found a significant positive correlation between improvement of the intestinal disease and clinical improvement of EIM at each time point.
Ustekinumab is usually preferred in patients with EIM, Dr. Livne-Margolin and colleagues note. But their findings “may raise some questions whether ustekinumab is, in fact, a better choice in those specific patients.”
Limitations of the study include its retrospective design and small cohort size.
Additionally, vedolizumab is given intravenously in the clinic and mandates patients to have a routine checkup every 1-2 months, whereas ustekinumab can be given at home. As a result, data were missing on some of the patients treated with ustekinumab during the follow-up.
Another limitation is that most of the patients had articular complaints with a small presentation of other EIM.
Also, most of the patients had Crohn’s disease, with only one patient with ulcerative colitis in the ustekinumab group, compared with 12 in the vedolizumab group.
Finally, patients treated with ustekinumab had more experience with anti-TNF treatment, compared with the vedolizumab group, which might have influenced the results with a negative bias toward ustekinumab.
The study had no specific funding. Three authors have disclosed relationships with Janssen, which makes ustekinumab.
A version of this article first appeared on Medscape.com.
Vedolizumab (Entyvio) and ustekinumab (Stelara) appear to be equally effective for extraintestinal manifestation (EIM) of inflammatory bowel disease (IBD), according to results of a retrospective study published online in Digestive and Liver Disease.
Between 25% and 40% of patients with IBD experience EIM, which reduces quality of life, according to the Crohn’s & Colitis Foundation. EIM commonly involves the joints, skin, bones, eyes, kidney, and liver. Anemia is another extraintestinal complication.
Until now, it’s been unclear whether vedolizumab and ustekinumab are equally effective for treating EIM.
Vedolizumab specifically targets the gastrointestinal tract, a potential disadvantage in reducing EIM, while ustekinumab is thought to have a systemic effect, a potential treatment advantage, Moran Livne-Margolin, MD, and colleagues, Chaim Sheba Medical Center, Ramat Gan, Israel, point out.
To investigate, they included 111 adults with IBD who were treated at the medical center between 2015 and 2021 – 53 with vedolizumab and 58 with ustekinumab. Before starting treatment, all of them had active EIM, most commonly arthralgia (84%).
After 6 weeks of treatment, 66% of patients in both groups had a clinical response to their intestinal disease.
After 14 and 26 weeks of treatment, clinical response rates were 59% and 50%, respectively, with vedolizumab, and 48% and 41%, respectively, with ustekinumab.
Over 52 weeks, both biologics were equally effective against the intestinal disease, with clinical response rates of 42% with vedolizumab and 44% with ustekinumab.
A similar pattern emerged when looking at improvement in EIM.
At week 6, 44% of patients taking vedolizumab and 35% taking ustekinumab had improvement in EIM, with no significant difference between the two biologics (P = .4).
At week 14, rates of improvement in EIM were 43% for vedolizumab and 33% for ustekinumab (P = .39); at 26 weeks, rates were 39% and 33%, respectively (P = .6); and at 52 weeks, rates were 34% and 36% (P = .9).
Researchers also found a significant positive correlation between improvement of the intestinal disease and clinical improvement of EIM at each time point.
Ustekinumab is usually preferred in patients with EIM, Dr. Livne-Margolin and colleagues note. But their findings “may raise some questions whether ustekinumab is, in fact, a better choice in those specific patients.”
Limitations of the study include its retrospective design and small cohort size.
Additionally, vedolizumab is given intravenously in the clinic and mandates patients to have a routine checkup every 1-2 months, whereas ustekinumab can be given at home. As a result, data were missing on some of the patients treated with ustekinumab during the follow-up.
Another limitation is that most of the patients had articular complaints with a small presentation of other EIM.
Also, most of the patients had Crohn’s disease, with only one patient with ulcerative colitis in the ustekinumab group, compared with 12 in the vedolizumab group.
Finally, patients treated with ustekinumab had more experience with anti-TNF treatment, compared with the vedolizumab group, which might have influenced the results with a negative bias toward ustekinumab.
The study had no specific funding. Three authors have disclosed relationships with Janssen, which makes ustekinumab.
A version of this article first appeared on Medscape.com.
Vedolizumab (Entyvio) and ustekinumab (Stelara) appear to be equally effective for extraintestinal manifestation (EIM) of inflammatory bowel disease (IBD), according to results of a retrospective study published online in Digestive and Liver Disease.
Between 25% and 40% of patients with IBD experience EIM, which reduces quality of life, according to the Crohn’s & Colitis Foundation. EIM commonly involves the joints, skin, bones, eyes, kidney, and liver. Anemia is another extraintestinal complication.
Until now, it’s been unclear whether vedolizumab and ustekinumab are equally effective for treating EIM.
Vedolizumab specifically targets the gastrointestinal tract, a potential disadvantage in reducing EIM, while ustekinumab is thought to have a systemic effect, a potential treatment advantage, Moran Livne-Margolin, MD, and colleagues, Chaim Sheba Medical Center, Ramat Gan, Israel, point out.
To investigate, they included 111 adults with IBD who were treated at the medical center between 2015 and 2021 – 53 with vedolizumab and 58 with ustekinumab. Before starting treatment, all of them had active EIM, most commonly arthralgia (84%).
After 6 weeks of treatment, 66% of patients in both groups had a clinical response to their intestinal disease.
After 14 and 26 weeks of treatment, clinical response rates were 59% and 50%, respectively, with vedolizumab, and 48% and 41%, respectively, with ustekinumab.
Over 52 weeks, both biologics were equally effective against the intestinal disease, with clinical response rates of 42% with vedolizumab and 44% with ustekinumab.
A similar pattern emerged when looking at improvement in EIM.
At week 6, 44% of patients taking vedolizumab and 35% taking ustekinumab had improvement in EIM, with no significant difference between the two biologics (P = .4).
At week 14, rates of improvement in EIM were 43% for vedolizumab and 33% for ustekinumab (P = .39); at 26 weeks, rates were 39% and 33%, respectively (P = .6); and at 52 weeks, rates were 34% and 36% (P = .9).
Researchers also found a significant positive correlation between improvement of the intestinal disease and clinical improvement of EIM at each time point.
Ustekinumab is usually preferred in patients with EIM, Dr. Livne-Margolin and colleagues note. But their findings “may raise some questions whether ustekinumab is, in fact, a better choice in those specific patients.”
Limitations of the study include its retrospective design and small cohort size.
Additionally, vedolizumab is given intravenously in the clinic and mandates patients to have a routine checkup every 1-2 months, whereas ustekinumab can be given at home. As a result, data were missing on some of the patients treated with ustekinumab during the follow-up.
Another limitation is that most of the patients had articular complaints with a small presentation of other EIM.
Also, most of the patients had Crohn’s disease, with only one patient with ulcerative colitis in the ustekinumab group, compared with 12 in the vedolizumab group.
Finally, patients treated with ustekinumab had more experience with anti-TNF treatment, compared with the vedolizumab group, which might have influenced the results with a negative bias toward ustekinumab.
The study had no specific funding. Three authors have disclosed relationships with Janssen, which makes ustekinumab.
A version of this article first appeared on Medscape.com.
FROM DIGESTIVE AND LIVER DISEASE
Anatomic site influences ropivacaine duration during dermatologic surgery
DENVER – , results from a single-center study showed.
Ropivacaine is a long-acting anesthetic that may be used as a substitute for the more commonly local anesthetics such as lidocaine or bupivacaine in dermatologic surgery, lead study author Kira Minkis, MD, PhD, told this news organization following the annual meeting of the American Society for Dermatologic Surgery, where the study results were presented during an oral abstract session. By comparison, ropivacaine has been reported to have a faster onset, similar duration in the range of 6-14 hours, less pain upon injection, and inherent vasoconstrictive properties.
“With tumescent anesthesia, studies have previously shown that the rate and absorption of anesthetics is influenced by the site of administration,” said Dr. Minkis, director of Mohs and dermatologic surgery at Weill Cornell Medicine, New York. “In studies comparing absorption of local anesthetics in tumescent anesthesia by regions that differ in vascularity, peak serum concentrations are greater and rise more rapidly after use in the head and neck compared to the trunk and extremities. However, no studies to date have compared the duration of ropivacaine in highly vascularized tissue or compared duration between regions that differ in vascularity.” The aim of the study, she noted, was to characterize the difference in duration of ropivacaine’s effects between anatomic regions of rich and comparably poor vascularity, such as the face and extremities, respectively.
Dr. Minkis and her colleagues recruited 17 women and 12 men with a mean age of 72 years who underwent Mohs surgery on the nose or the shin at Weill Cornell Medicine. Patients were anesthetized at each site with a subcutaneous injection of 0.5 mL of ropivacaine, 0.2%. Sensation was determined by pinprick prior to injection, at baseline, and every 15 minutes until sensation returned or surgery concluded. The primary endpoint was time to return of pinprick sensation.
The researchers found that the duration of ropivacaine was significantly shorter on the nose (a median of 60 minutes) than on the shin (a median of 210 minutes). In fact, the upper limit of the range of duration at the shin was not determinable because 22 of the 29 (76%) of participants did not regain sensation on the shin prior to leaving the surgical suite and concluding the study. The proportion of study participants who regained sensation within 1 hour was 76% among those who were treated on the nose vs. 3% of those who were treated on the shin (P < .0001).
“With durations of up to 6-14 hours reported, our results indicate a strikingly shorter duration of local anesthesia in highly vascularized tissue,” Dr. Minkis said. “The brevity of local anesthesia is even more surprising given the intrinsic vasoconstrictive properties of ropivacaine. Often, we co-administer epinephrine to achieve vasoconstriction and reduce local blood flow, thus prolonging local concentrations of the anesthetic with the added benefit of reducing bleeding during surgery. The short duration we’ve observed in our study is emphasized in using a potent, long-acting local anesthetic with vasoconstrictive properties that otherwise should attenuate the effects of high local vascularity.”
In other findings, patients with history of hypertension were more likely to regain sensation on the nose by 60 minutes but this did not reach statistical significance (P = .079). Other comorbidities including underlying anxiety/depression, diabetes, and kidney disease did not significantly impact duration of ropivacaine action on the nose. The same held true for patients who were treated on the shin.
“We highlight an inconsistency between the reported duration of a long-lasting local anesthetic and the short-lived anesthesia experienced by our patients in a highly vascularized region,” Dr. Minkis said. “In practice, adjunctive use of a long-acting anesthetic to prolong anesthesia is common, which may provide relief from multiple injections of shorter-acting lidocaine. However, the duration of Mohs surgery can be unpredictable. Extended wait times between stages may exceed the duration we’ve observed in this study.”
In addition, she continued, “pain is frequently reported on postoperative days 0 to 3, leading some to recommend the use of long-acting local anesthetics to prevent overprescription or a gap in pain coverage. This emphasizes a gap in effective pain control, but also an opportunity to improve our patients’ surgical and recovery experiences.”
Impact on practice
Keith L. Duffy, MD, associate professor of dermatology at the University of Utah, Salt Lake City, who was asked to comment on the study, said that in light of current local anesthetic shortages and back orders, “we dermatologic surgeons have been experimenting with different anesthetics and concentrations that we can use in our patients. Ropivacaine may become the anesthetic of choice for many of our practices given its inherent properties.”
The duration of anesthetic effects by anatomic location in this study is “actually more impressive than I would have suspected as a practicing Mohs surgeon. The results of this study will immediately impact my Mohs surgery clinic,” he said, adding that he hoped that Dr. Minkis and others “will expand on this study to include more patients, different anesthetics, and more anatomic locations.”
Dr. Minkis acknowledged certain limitations of the study, including its single-center design and the fact that there were too few observations of medical and clinical characteristics for subgroup analysis.
She and Dr. Duffy reported having no financial disclosures.
DENVER – , results from a single-center study showed.
Ropivacaine is a long-acting anesthetic that may be used as a substitute for the more commonly local anesthetics such as lidocaine or bupivacaine in dermatologic surgery, lead study author Kira Minkis, MD, PhD, told this news organization following the annual meeting of the American Society for Dermatologic Surgery, where the study results were presented during an oral abstract session. By comparison, ropivacaine has been reported to have a faster onset, similar duration in the range of 6-14 hours, less pain upon injection, and inherent vasoconstrictive properties.
“With tumescent anesthesia, studies have previously shown that the rate and absorption of anesthetics is influenced by the site of administration,” said Dr. Minkis, director of Mohs and dermatologic surgery at Weill Cornell Medicine, New York. “In studies comparing absorption of local anesthetics in tumescent anesthesia by regions that differ in vascularity, peak serum concentrations are greater and rise more rapidly after use in the head and neck compared to the trunk and extremities. However, no studies to date have compared the duration of ropivacaine in highly vascularized tissue or compared duration between regions that differ in vascularity.” The aim of the study, she noted, was to characterize the difference in duration of ropivacaine’s effects between anatomic regions of rich and comparably poor vascularity, such as the face and extremities, respectively.
Dr. Minkis and her colleagues recruited 17 women and 12 men with a mean age of 72 years who underwent Mohs surgery on the nose or the shin at Weill Cornell Medicine. Patients were anesthetized at each site with a subcutaneous injection of 0.5 mL of ropivacaine, 0.2%. Sensation was determined by pinprick prior to injection, at baseline, and every 15 minutes until sensation returned or surgery concluded. The primary endpoint was time to return of pinprick sensation.
The researchers found that the duration of ropivacaine was significantly shorter on the nose (a median of 60 minutes) than on the shin (a median of 210 minutes). In fact, the upper limit of the range of duration at the shin was not determinable because 22 of the 29 (76%) of participants did not regain sensation on the shin prior to leaving the surgical suite and concluding the study. The proportion of study participants who regained sensation within 1 hour was 76% among those who were treated on the nose vs. 3% of those who were treated on the shin (P < .0001).
“With durations of up to 6-14 hours reported, our results indicate a strikingly shorter duration of local anesthesia in highly vascularized tissue,” Dr. Minkis said. “The brevity of local anesthesia is even more surprising given the intrinsic vasoconstrictive properties of ropivacaine. Often, we co-administer epinephrine to achieve vasoconstriction and reduce local blood flow, thus prolonging local concentrations of the anesthetic with the added benefit of reducing bleeding during surgery. The short duration we’ve observed in our study is emphasized in using a potent, long-acting local anesthetic with vasoconstrictive properties that otherwise should attenuate the effects of high local vascularity.”
In other findings, patients with history of hypertension were more likely to regain sensation on the nose by 60 minutes but this did not reach statistical significance (P = .079). Other comorbidities including underlying anxiety/depression, diabetes, and kidney disease did not significantly impact duration of ropivacaine action on the nose. The same held true for patients who were treated on the shin.
“We highlight an inconsistency between the reported duration of a long-lasting local anesthetic and the short-lived anesthesia experienced by our patients in a highly vascularized region,” Dr. Minkis said. “In practice, adjunctive use of a long-acting anesthetic to prolong anesthesia is common, which may provide relief from multiple injections of shorter-acting lidocaine. However, the duration of Mohs surgery can be unpredictable. Extended wait times between stages may exceed the duration we’ve observed in this study.”
In addition, she continued, “pain is frequently reported on postoperative days 0 to 3, leading some to recommend the use of long-acting local anesthetics to prevent overprescription or a gap in pain coverage. This emphasizes a gap in effective pain control, but also an opportunity to improve our patients’ surgical and recovery experiences.”
Impact on practice
Keith L. Duffy, MD, associate professor of dermatology at the University of Utah, Salt Lake City, who was asked to comment on the study, said that in light of current local anesthetic shortages and back orders, “we dermatologic surgeons have been experimenting with different anesthetics and concentrations that we can use in our patients. Ropivacaine may become the anesthetic of choice for many of our practices given its inherent properties.”
The duration of anesthetic effects by anatomic location in this study is “actually more impressive than I would have suspected as a practicing Mohs surgeon. The results of this study will immediately impact my Mohs surgery clinic,” he said, adding that he hoped that Dr. Minkis and others “will expand on this study to include more patients, different anesthetics, and more anatomic locations.”
Dr. Minkis acknowledged certain limitations of the study, including its single-center design and the fact that there were too few observations of medical and clinical characteristics for subgroup analysis.
She and Dr. Duffy reported having no financial disclosures.
DENVER – , results from a single-center study showed.
Ropivacaine is a long-acting anesthetic that may be used as a substitute for the more commonly local anesthetics such as lidocaine or bupivacaine in dermatologic surgery, lead study author Kira Minkis, MD, PhD, told this news organization following the annual meeting of the American Society for Dermatologic Surgery, where the study results were presented during an oral abstract session. By comparison, ropivacaine has been reported to have a faster onset, similar duration in the range of 6-14 hours, less pain upon injection, and inherent vasoconstrictive properties.
“With tumescent anesthesia, studies have previously shown that the rate and absorption of anesthetics is influenced by the site of administration,” said Dr. Minkis, director of Mohs and dermatologic surgery at Weill Cornell Medicine, New York. “In studies comparing absorption of local anesthetics in tumescent anesthesia by regions that differ in vascularity, peak serum concentrations are greater and rise more rapidly after use in the head and neck compared to the trunk and extremities. However, no studies to date have compared the duration of ropivacaine in highly vascularized tissue or compared duration between regions that differ in vascularity.” The aim of the study, she noted, was to characterize the difference in duration of ropivacaine’s effects between anatomic regions of rich and comparably poor vascularity, such as the face and extremities, respectively.
Dr. Minkis and her colleagues recruited 17 women and 12 men with a mean age of 72 years who underwent Mohs surgery on the nose or the shin at Weill Cornell Medicine. Patients were anesthetized at each site with a subcutaneous injection of 0.5 mL of ropivacaine, 0.2%. Sensation was determined by pinprick prior to injection, at baseline, and every 15 minutes until sensation returned or surgery concluded. The primary endpoint was time to return of pinprick sensation.
The researchers found that the duration of ropivacaine was significantly shorter on the nose (a median of 60 minutes) than on the shin (a median of 210 minutes). In fact, the upper limit of the range of duration at the shin was not determinable because 22 of the 29 (76%) of participants did not regain sensation on the shin prior to leaving the surgical suite and concluding the study. The proportion of study participants who regained sensation within 1 hour was 76% among those who were treated on the nose vs. 3% of those who were treated on the shin (P < .0001).
“With durations of up to 6-14 hours reported, our results indicate a strikingly shorter duration of local anesthesia in highly vascularized tissue,” Dr. Minkis said. “The brevity of local anesthesia is even more surprising given the intrinsic vasoconstrictive properties of ropivacaine. Often, we co-administer epinephrine to achieve vasoconstriction and reduce local blood flow, thus prolonging local concentrations of the anesthetic with the added benefit of reducing bleeding during surgery. The short duration we’ve observed in our study is emphasized in using a potent, long-acting local anesthetic with vasoconstrictive properties that otherwise should attenuate the effects of high local vascularity.”
In other findings, patients with history of hypertension were more likely to regain sensation on the nose by 60 minutes but this did not reach statistical significance (P = .079). Other comorbidities including underlying anxiety/depression, diabetes, and kidney disease did not significantly impact duration of ropivacaine action on the nose. The same held true for patients who were treated on the shin.
“We highlight an inconsistency between the reported duration of a long-lasting local anesthetic and the short-lived anesthesia experienced by our patients in a highly vascularized region,” Dr. Minkis said. “In practice, adjunctive use of a long-acting anesthetic to prolong anesthesia is common, which may provide relief from multiple injections of shorter-acting lidocaine. However, the duration of Mohs surgery can be unpredictable. Extended wait times between stages may exceed the duration we’ve observed in this study.”
In addition, she continued, “pain is frequently reported on postoperative days 0 to 3, leading some to recommend the use of long-acting local anesthetics to prevent overprescription or a gap in pain coverage. This emphasizes a gap in effective pain control, but also an opportunity to improve our patients’ surgical and recovery experiences.”
Impact on practice
Keith L. Duffy, MD, associate professor of dermatology at the University of Utah, Salt Lake City, who was asked to comment on the study, said that in light of current local anesthetic shortages and back orders, “we dermatologic surgeons have been experimenting with different anesthetics and concentrations that we can use in our patients. Ropivacaine may become the anesthetic of choice for many of our practices given its inherent properties.”
The duration of anesthetic effects by anatomic location in this study is “actually more impressive than I would have suspected as a practicing Mohs surgeon. The results of this study will immediately impact my Mohs surgery clinic,” he said, adding that he hoped that Dr. Minkis and others “will expand on this study to include more patients, different anesthetics, and more anatomic locations.”
Dr. Minkis acknowledged certain limitations of the study, including its single-center design and the fact that there were too few observations of medical and clinical characteristics for subgroup analysis.
She and Dr. Duffy reported having no financial disclosures.
AT ASDS 2022
First-in-class device for facial wrinkles, tightening hits the market
DENVER – .
“It’s early yet, but I have treated dozens of patients with this device, and they have been happy with the results,” Mathew M. Avram, MD, JD, said at the annual meeting of the American Society for Dermatologic Surgery. “This is a new technique that offers the ability to remove a significant amount of damaged, lax skin without concern for scarring,” he said.
A brainchild of dermatologists and plastic surgeons at Massachusetts General Hospital, Boston, the first-in-class device is cleared by the Food and Drug Administration for the treatment of moderate and severe wrinkles in the mid and lower face in adults aged 22 years or older with Fitzpatrick skin types I-IV. It features a proprietary needle design that makes a series of high throughput microexcisions in epidermal and dermal tissue, with minimal downtime and without using thermal energy.
“It doesn’t do anything equivalent to a facelift, but the concept is a facelift by thousands of micro-punch excisions,” said Dr. Avram, director of laser, cosmetics, and dermatologic surgery at Massachusetts General Hospital. “Rather than pulling up the skin and lifting it and cutting the excess skin like we do with a facelift, we are creating thousands of smaller-scale tissue removals with immediate closures to do the same thing. The micro-cores are about the size of a 22-gauge needle and there is no scarring due to the small size of these tissue extractions.”
The device features needle cartridges capable of excising up to 24,000 cores per treatment. According to data from Cytrellis, the manufacturer, the equivalent of about 2 inches of skin can be removed during the procedure, which typically takes fewer than 30 minutes to perform. “There is no heat whatsoever,” Dr. Avram said. “In my experience, it especially helps with jawline definition, the lower medial cheek excess skin, and accordion lines in that area.”
In a pivotal trial of the device, 51 patients with mid to lower face wrinkles (moderately deep or deep wrinkles with well-defined edges) were treated 2-3 times with 7%-8% skin removal and up to a 5-mm needle coring depth). The investigators found that 40% of study participants achieved an improvement of 2 grades on the Lemperle Wrinkle Severity Scale and that the rate of overall satisfaction (slightly, somewhat, and extremely satisfied) was 86%.
In addition, 90% showed improvement of treated sites on the Global Aesthetic Improvement Scale, and 70% were comfortable enough to go out in public or return to work 3 days after treatment. Common side effects that can occur immediately post treatment include redness, swelling, and pinpoint bleeding, which typically clear in a few days.
Dr. Avram, immediate past president of the ASDS, has posted videos to his Instagram feed that show him treating patients with the Ellacor device and he admits that the procedure looks painful. “There are all these tear emojis and people cursing me out,” he said, referring to responses from his Instagram followers.
Proper local anesthesia prior to treatment is key. “I perform nerve blocks and infiltrate the skin,” he said. “You have to cover the whole treatment area. If you don’t, then it’s going to hurt. The average pain score is 1.9 out of 10. The highest pain score I’ve gotten from a patient is a 3 out of 10.”
Dr. Avram disclosed that he has received consulting fees from Allergan, Merz, Sciton, and Soliton, and has ownership and/or shareholder interest in Cytrellis.
DENVER – .
“It’s early yet, but I have treated dozens of patients with this device, and they have been happy with the results,” Mathew M. Avram, MD, JD, said at the annual meeting of the American Society for Dermatologic Surgery. “This is a new technique that offers the ability to remove a significant amount of damaged, lax skin without concern for scarring,” he said.
A brainchild of dermatologists and plastic surgeons at Massachusetts General Hospital, Boston, the first-in-class device is cleared by the Food and Drug Administration for the treatment of moderate and severe wrinkles in the mid and lower face in adults aged 22 years or older with Fitzpatrick skin types I-IV. It features a proprietary needle design that makes a series of high throughput microexcisions in epidermal and dermal tissue, with minimal downtime and without using thermal energy.
“It doesn’t do anything equivalent to a facelift, but the concept is a facelift by thousands of micro-punch excisions,” said Dr. Avram, director of laser, cosmetics, and dermatologic surgery at Massachusetts General Hospital. “Rather than pulling up the skin and lifting it and cutting the excess skin like we do with a facelift, we are creating thousands of smaller-scale tissue removals with immediate closures to do the same thing. The micro-cores are about the size of a 22-gauge needle and there is no scarring due to the small size of these tissue extractions.”
The device features needle cartridges capable of excising up to 24,000 cores per treatment. According to data from Cytrellis, the manufacturer, the equivalent of about 2 inches of skin can be removed during the procedure, which typically takes fewer than 30 minutes to perform. “There is no heat whatsoever,” Dr. Avram said. “In my experience, it especially helps with jawline definition, the lower medial cheek excess skin, and accordion lines in that area.”
In a pivotal trial of the device, 51 patients with mid to lower face wrinkles (moderately deep or deep wrinkles with well-defined edges) were treated 2-3 times with 7%-8% skin removal and up to a 5-mm needle coring depth). The investigators found that 40% of study participants achieved an improvement of 2 grades on the Lemperle Wrinkle Severity Scale and that the rate of overall satisfaction (slightly, somewhat, and extremely satisfied) was 86%.
In addition, 90% showed improvement of treated sites on the Global Aesthetic Improvement Scale, and 70% were comfortable enough to go out in public or return to work 3 days after treatment. Common side effects that can occur immediately post treatment include redness, swelling, and pinpoint bleeding, which typically clear in a few days.
Dr. Avram, immediate past president of the ASDS, has posted videos to his Instagram feed that show him treating patients with the Ellacor device and he admits that the procedure looks painful. “There are all these tear emojis and people cursing me out,” he said, referring to responses from his Instagram followers.
Proper local anesthesia prior to treatment is key. “I perform nerve blocks and infiltrate the skin,” he said. “You have to cover the whole treatment area. If you don’t, then it’s going to hurt. The average pain score is 1.9 out of 10. The highest pain score I’ve gotten from a patient is a 3 out of 10.”
Dr. Avram disclosed that he has received consulting fees from Allergan, Merz, Sciton, and Soliton, and has ownership and/or shareholder interest in Cytrellis.
DENVER – .
“It’s early yet, but I have treated dozens of patients with this device, and they have been happy with the results,” Mathew M. Avram, MD, JD, said at the annual meeting of the American Society for Dermatologic Surgery. “This is a new technique that offers the ability to remove a significant amount of damaged, lax skin without concern for scarring,” he said.
A brainchild of dermatologists and plastic surgeons at Massachusetts General Hospital, Boston, the first-in-class device is cleared by the Food and Drug Administration for the treatment of moderate and severe wrinkles in the mid and lower face in adults aged 22 years or older with Fitzpatrick skin types I-IV. It features a proprietary needle design that makes a series of high throughput microexcisions in epidermal and dermal tissue, with minimal downtime and without using thermal energy.
“It doesn’t do anything equivalent to a facelift, but the concept is a facelift by thousands of micro-punch excisions,” said Dr. Avram, director of laser, cosmetics, and dermatologic surgery at Massachusetts General Hospital. “Rather than pulling up the skin and lifting it and cutting the excess skin like we do with a facelift, we are creating thousands of smaller-scale tissue removals with immediate closures to do the same thing. The micro-cores are about the size of a 22-gauge needle and there is no scarring due to the small size of these tissue extractions.”
The device features needle cartridges capable of excising up to 24,000 cores per treatment. According to data from Cytrellis, the manufacturer, the equivalent of about 2 inches of skin can be removed during the procedure, which typically takes fewer than 30 minutes to perform. “There is no heat whatsoever,” Dr. Avram said. “In my experience, it especially helps with jawline definition, the lower medial cheek excess skin, and accordion lines in that area.”
In a pivotal trial of the device, 51 patients with mid to lower face wrinkles (moderately deep or deep wrinkles with well-defined edges) were treated 2-3 times with 7%-8% skin removal and up to a 5-mm needle coring depth). The investigators found that 40% of study participants achieved an improvement of 2 grades on the Lemperle Wrinkle Severity Scale and that the rate of overall satisfaction (slightly, somewhat, and extremely satisfied) was 86%.
In addition, 90% showed improvement of treated sites on the Global Aesthetic Improvement Scale, and 70% were comfortable enough to go out in public or return to work 3 days after treatment. Common side effects that can occur immediately post treatment include redness, swelling, and pinpoint bleeding, which typically clear in a few days.
Dr. Avram, immediate past president of the ASDS, has posted videos to his Instagram feed that show him treating patients with the Ellacor device and he admits that the procedure looks painful. “There are all these tear emojis and people cursing me out,” he said, referring to responses from his Instagram followers.
Proper local anesthesia prior to treatment is key. “I perform nerve blocks and infiltrate the skin,” he said. “You have to cover the whole treatment area. If you don’t, then it’s going to hurt. The average pain score is 1.9 out of 10. The highest pain score I’ve gotten from a patient is a 3 out of 10.”
Dr. Avram disclosed that he has received consulting fees from Allergan, Merz, Sciton, and Soliton, and has ownership and/or shareholder interest in Cytrellis.
AT ASDS 2022
Many specialists are on the wrong side of the patient-jargon relationship
Doctor, doctor, gimme the news. I got a bad case of misidentifying you
There are a lot of medical specialties out there. A lot. Everything from allergists to urologists, with something like 150 subspecialties grouped in among the larger specialties. Can you name every one? Do you know what they do?
The point is, telling a patient or anyone in the general public that you’re an ophthalmologist may not be as helpful as you might think, if a recent study is to be believed. In a survey of 204 adults, conducted at the Minnesota State Fair of all places, researchers asked volunteers to define 14 different specialties, as well as five medical seniority titles.
The results were less than stellar. While more than 90% of people correctly defined what cardiologists and dermatologists do, 6 of the other 12 specialists were correctly identified by less than half of those surveyed. Nephrology was at the bottom, correctly identified by just 20% of the fair-attending public, followed by internists (21%), intensivists (29%), hospitalists (31%), pulmonologists (43%), and neonatologists at 48%. The hospitalists are particularly concerning. They’re doctors, but in hospitals. How hard is that? (Yes, it’s obviously more complicated than that, but still.)
The general public didn’t fare much better when it came to correctly lining up the order of progression from medical student to attending. Just 12% managed to place all five in the correct order of med student, intern, senior resident, fellow, then attending, with senior resident proving especially troublesome. More than 40% put senior resident at the end, compared with 27% for attending. Which does make a certain amount of sense, since it has senior in the name.
While the results speak for themselves – maybe elaborate on what the heck your fancy title actually means – it’s too bad the researchers didn’t throw in something really tricky. If two-thirds of the population can’t identify a hospitalist, just imagine how many people would misidentify an otolaryngologist.
Beach-to-table sand could fight obesity
People are always looking for the new weight loss solution. Whether it’s to just look good in a new pair of jeans or reduce the risk of cardiovascular disease, there are millions of diets and exercise routines out here. We’re here to tell you that the next new therapy to reduce fat comes from a very unsuspecting place: Sand.
Like sand from the beach and desert, sand? Well, yes and no.
The research involved engineered porous silica particles made from sand that are designed to have a high surface area. Investigators used a two-step GI model in which gastric digestion was modeled for 30 minutes, followed by a 60-minute intestinal phase, to show that the porous silica particles helped prevent fat and sugar adsorption within the GI tract.
By mimicking the gastrointestinal environment during digestion of a high-fat, high-carb meal, the researchers found that the porous silica created an “anti-obesity effect” by restricting the adsorption of those fats and carbohydrates.
Okay, but how is that on the tummy? Much gentler on the stomach than a drug such as orlistat, said senior researcher Paul Joyce, PhD, of the University of South Australia, Adelaide, who noted the lack of effective therapies without side effects, such as bloating, diarrhea, and abdominal pain, that deter people from treatment.
Obesity affects over 1.9 billion people worldwide, so the researchers think this could be a breakthrough. Reducing obesity may be one of the most preventable ways to reduce the risk of type 2 diabetes, heart disease, and other weight-related chronic conditions. A treatment solution this simple could be the answer to this global health crisis.
Who would have thought the solution would be as simple as sand? But how would the sand get in our stomachs? Do we sprinkle it on our food? Mix it in during cooking? Or will the sand come in pill form? We sure hope it’s that third one.
I am Reliebo. I am here to help you
Halloween is almost here, and the LOTME staff has been trying to make the office look as scary as possible: Headless vampires, ghost clowns, Ted Cruz, gray tombstones, pink hearts, green clovers, red balloons. Wait a second, those last three are Lucky Charms marshmallows, aren’t they? We’ll use those some other time.
What are we not using to decorate? Well, besides marshmallows from cereal, we’re not using Reliebo. That’s what we’re not using. Reliebo is a cute little fuzzy robot, and is not at all scary. Reliebo was designed to be the opposite of scary. Reliebo “may reduce fear as well as alleviate the perception of pain during medical treatments, including vaccinations,” senior author Fumihide Tanaka, PhD, of the University of Tsukuba (Japan) said in a written statement.
The soft, fur-covered robot contains small airbags that can inflate in response to hand movements. When study participants were subjected to a moderate heat stimulus on one arm, those who held the robot with the other arm experienced less pain than those who did not have a Reliebo.
The results also were encouraging when Dr. Tanaka and associates measured the levels of oxytocin and cortisol (biomarkers for stress) from the subjects’ saliva samples and evaluated their fear of injections and their psychological state before and after the experiments.
After looking at that photo of Reliebo for a while, though, we have to admit that we’re having a bit of a rethink about its cuteness. Is it cute, or weird-looking? An office full of fuzzy little inflating robots just could be seriously creepy. Please don’t tell the rest of the staff about this. We want to surprise them on Monday.
Doctor, doctor, gimme the news. I got a bad case of misidentifying you
There are a lot of medical specialties out there. A lot. Everything from allergists to urologists, with something like 150 subspecialties grouped in among the larger specialties. Can you name every one? Do you know what they do?
The point is, telling a patient or anyone in the general public that you’re an ophthalmologist may not be as helpful as you might think, if a recent study is to be believed. In a survey of 204 adults, conducted at the Minnesota State Fair of all places, researchers asked volunteers to define 14 different specialties, as well as five medical seniority titles.
The results were less than stellar. While more than 90% of people correctly defined what cardiologists and dermatologists do, 6 of the other 12 specialists were correctly identified by less than half of those surveyed. Nephrology was at the bottom, correctly identified by just 20% of the fair-attending public, followed by internists (21%), intensivists (29%), hospitalists (31%), pulmonologists (43%), and neonatologists at 48%. The hospitalists are particularly concerning. They’re doctors, but in hospitals. How hard is that? (Yes, it’s obviously more complicated than that, but still.)
The general public didn’t fare much better when it came to correctly lining up the order of progression from medical student to attending. Just 12% managed to place all five in the correct order of med student, intern, senior resident, fellow, then attending, with senior resident proving especially troublesome. More than 40% put senior resident at the end, compared with 27% for attending. Which does make a certain amount of sense, since it has senior in the name.
While the results speak for themselves – maybe elaborate on what the heck your fancy title actually means – it’s too bad the researchers didn’t throw in something really tricky. If two-thirds of the population can’t identify a hospitalist, just imagine how many people would misidentify an otolaryngologist.
Beach-to-table sand could fight obesity
People are always looking for the new weight loss solution. Whether it’s to just look good in a new pair of jeans or reduce the risk of cardiovascular disease, there are millions of diets and exercise routines out here. We’re here to tell you that the next new therapy to reduce fat comes from a very unsuspecting place: Sand.
Like sand from the beach and desert, sand? Well, yes and no.
The research involved engineered porous silica particles made from sand that are designed to have a high surface area. Investigators used a two-step GI model in which gastric digestion was modeled for 30 minutes, followed by a 60-minute intestinal phase, to show that the porous silica particles helped prevent fat and sugar adsorption within the GI tract.
By mimicking the gastrointestinal environment during digestion of a high-fat, high-carb meal, the researchers found that the porous silica created an “anti-obesity effect” by restricting the adsorption of those fats and carbohydrates.
Okay, but how is that on the tummy? Much gentler on the stomach than a drug such as orlistat, said senior researcher Paul Joyce, PhD, of the University of South Australia, Adelaide, who noted the lack of effective therapies without side effects, such as bloating, diarrhea, and abdominal pain, that deter people from treatment.
Obesity affects over 1.9 billion people worldwide, so the researchers think this could be a breakthrough. Reducing obesity may be one of the most preventable ways to reduce the risk of type 2 diabetes, heart disease, and other weight-related chronic conditions. A treatment solution this simple could be the answer to this global health crisis.
Who would have thought the solution would be as simple as sand? But how would the sand get in our stomachs? Do we sprinkle it on our food? Mix it in during cooking? Or will the sand come in pill form? We sure hope it’s that third one.
I am Reliebo. I am here to help you
Halloween is almost here, and the LOTME staff has been trying to make the office look as scary as possible: Headless vampires, ghost clowns, Ted Cruz, gray tombstones, pink hearts, green clovers, red balloons. Wait a second, those last three are Lucky Charms marshmallows, aren’t they? We’ll use those some other time.
What are we not using to decorate? Well, besides marshmallows from cereal, we’re not using Reliebo. That’s what we’re not using. Reliebo is a cute little fuzzy robot, and is not at all scary. Reliebo was designed to be the opposite of scary. Reliebo “may reduce fear as well as alleviate the perception of pain during medical treatments, including vaccinations,” senior author Fumihide Tanaka, PhD, of the University of Tsukuba (Japan) said in a written statement.
The soft, fur-covered robot contains small airbags that can inflate in response to hand movements. When study participants were subjected to a moderate heat stimulus on one arm, those who held the robot with the other arm experienced less pain than those who did not have a Reliebo.
The results also were encouraging when Dr. Tanaka and associates measured the levels of oxytocin and cortisol (biomarkers for stress) from the subjects’ saliva samples and evaluated their fear of injections and their psychological state before and after the experiments.
After looking at that photo of Reliebo for a while, though, we have to admit that we’re having a bit of a rethink about its cuteness. Is it cute, or weird-looking? An office full of fuzzy little inflating robots just could be seriously creepy. Please don’t tell the rest of the staff about this. We want to surprise them on Monday.
Doctor, doctor, gimme the news. I got a bad case of misidentifying you
There are a lot of medical specialties out there. A lot. Everything from allergists to urologists, with something like 150 subspecialties grouped in among the larger specialties. Can you name every one? Do you know what they do?
The point is, telling a patient or anyone in the general public that you’re an ophthalmologist may not be as helpful as you might think, if a recent study is to be believed. In a survey of 204 adults, conducted at the Minnesota State Fair of all places, researchers asked volunteers to define 14 different specialties, as well as five medical seniority titles.
The results were less than stellar. While more than 90% of people correctly defined what cardiologists and dermatologists do, 6 of the other 12 specialists were correctly identified by less than half of those surveyed. Nephrology was at the bottom, correctly identified by just 20% of the fair-attending public, followed by internists (21%), intensivists (29%), hospitalists (31%), pulmonologists (43%), and neonatologists at 48%. The hospitalists are particularly concerning. They’re doctors, but in hospitals. How hard is that? (Yes, it’s obviously more complicated than that, but still.)
The general public didn’t fare much better when it came to correctly lining up the order of progression from medical student to attending. Just 12% managed to place all five in the correct order of med student, intern, senior resident, fellow, then attending, with senior resident proving especially troublesome. More than 40% put senior resident at the end, compared with 27% for attending. Which does make a certain amount of sense, since it has senior in the name.
While the results speak for themselves – maybe elaborate on what the heck your fancy title actually means – it’s too bad the researchers didn’t throw in something really tricky. If two-thirds of the population can’t identify a hospitalist, just imagine how many people would misidentify an otolaryngologist.
Beach-to-table sand could fight obesity
People are always looking for the new weight loss solution. Whether it’s to just look good in a new pair of jeans or reduce the risk of cardiovascular disease, there are millions of diets and exercise routines out here. We’re here to tell you that the next new therapy to reduce fat comes from a very unsuspecting place: Sand.
Like sand from the beach and desert, sand? Well, yes and no.
The research involved engineered porous silica particles made from sand that are designed to have a high surface area. Investigators used a two-step GI model in which gastric digestion was modeled for 30 minutes, followed by a 60-minute intestinal phase, to show that the porous silica particles helped prevent fat and sugar adsorption within the GI tract.
By mimicking the gastrointestinal environment during digestion of a high-fat, high-carb meal, the researchers found that the porous silica created an “anti-obesity effect” by restricting the adsorption of those fats and carbohydrates.
Okay, but how is that on the tummy? Much gentler on the stomach than a drug such as orlistat, said senior researcher Paul Joyce, PhD, of the University of South Australia, Adelaide, who noted the lack of effective therapies without side effects, such as bloating, diarrhea, and abdominal pain, that deter people from treatment.
Obesity affects over 1.9 billion people worldwide, so the researchers think this could be a breakthrough. Reducing obesity may be one of the most preventable ways to reduce the risk of type 2 diabetes, heart disease, and other weight-related chronic conditions. A treatment solution this simple could be the answer to this global health crisis.
Who would have thought the solution would be as simple as sand? But how would the sand get in our stomachs? Do we sprinkle it on our food? Mix it in during cooking? Or will the sand come in pill form? We sure hope it’s that third one.
I am Reliebo. I am here to help you
Halloween is almost here, and the LOTME staff has been trying to make the office look as scary as possible: Headless vampires, ghost clowns, Ted Cruz, gray tombstones, pink hearts, green clovers, red balloons. Wait a second, those last three are Lucky Charms marshmallows, aren’t they? We’ll use those some other time.
What are we not using to decorate? Well, besides marshmallows from cereal, we’re not using Reliebo. That’s what we’re not using. Reliebo is a cute little fuzzy robot, and is not at all scary. Reliebo was designed to be the opposite of scary. Reliebo “may reduce fear as well as alleviate the perception of pain during medical treatments, including vaccinations,” senior author Fumihide Tanaka, PhD, of the University of Tsukuba (Japan) said in a written statement.
The soft, fur-covered robot contains small airbags that can inflate in response to hand movements. When study participants were subjected to a moderate heat stimulus on one arm, those who held the robot with the other arm experienced less pain than those who did not have a Reliebo.
The results also were encouraging when Dr. Tanaka and associates measured the levels of oxytocin and cortisol (biomarkers for stress) from the subjects’ saliva samples and evaluated their fear of injections and their psychological state before and after the experiments.
After looking at that photo of Reliebo for a while, though, we have to admit that we’re having a bit of a rethink about its cuteness. Is it cute, or weird-looking? An office full of fuzzy little inflating robots just could be seriously creepy. Please don’t tell the rest of the staff about this. We want to surprise them on Monday.
IgA Vasculitis in the Setting of Biologic Therapy for Psoriasis and Recurrent Cutaneous Methicillin-Resistant Staphylococcus aureus Colonization
Case Report
A 47-year-old man presented with a sudden-onset rash consisting of red bumps on the abdomen and legs that had been ongoing for several days. He had known psoriasis and psoriatic arthritis that had been well controlled with adalimumab for the last 18 months. He reported concurrent onset of nausea but denied fevers, chills, night sweats, unintentional weight loss, abdominal pain, and pruritus. He endorsed prior cutaneous infections of methicillin-resistant Staphylococcus aureus (MRSA). His medical history also included diabetes mellitus, hypertension, and obesity. His other medications included oral losartan-hydrochlorothiazide, amlodipine, naproxen, and atorvastatin.
Physical examination revealed numerous thin purpuric papules—some with adherent scale—distributed on the lower legs, extensor forearms, and abdomen. Abdominal lesions were confined to weight-related striae (Figure 1). The palms, soles, oral mucosa, and face were spared. Three punch biopsies were performed, including 1 for direct immunofluorescence (DIF), and the patient was instructed to apply clobetasol to the affected areas twice daily until further notice.
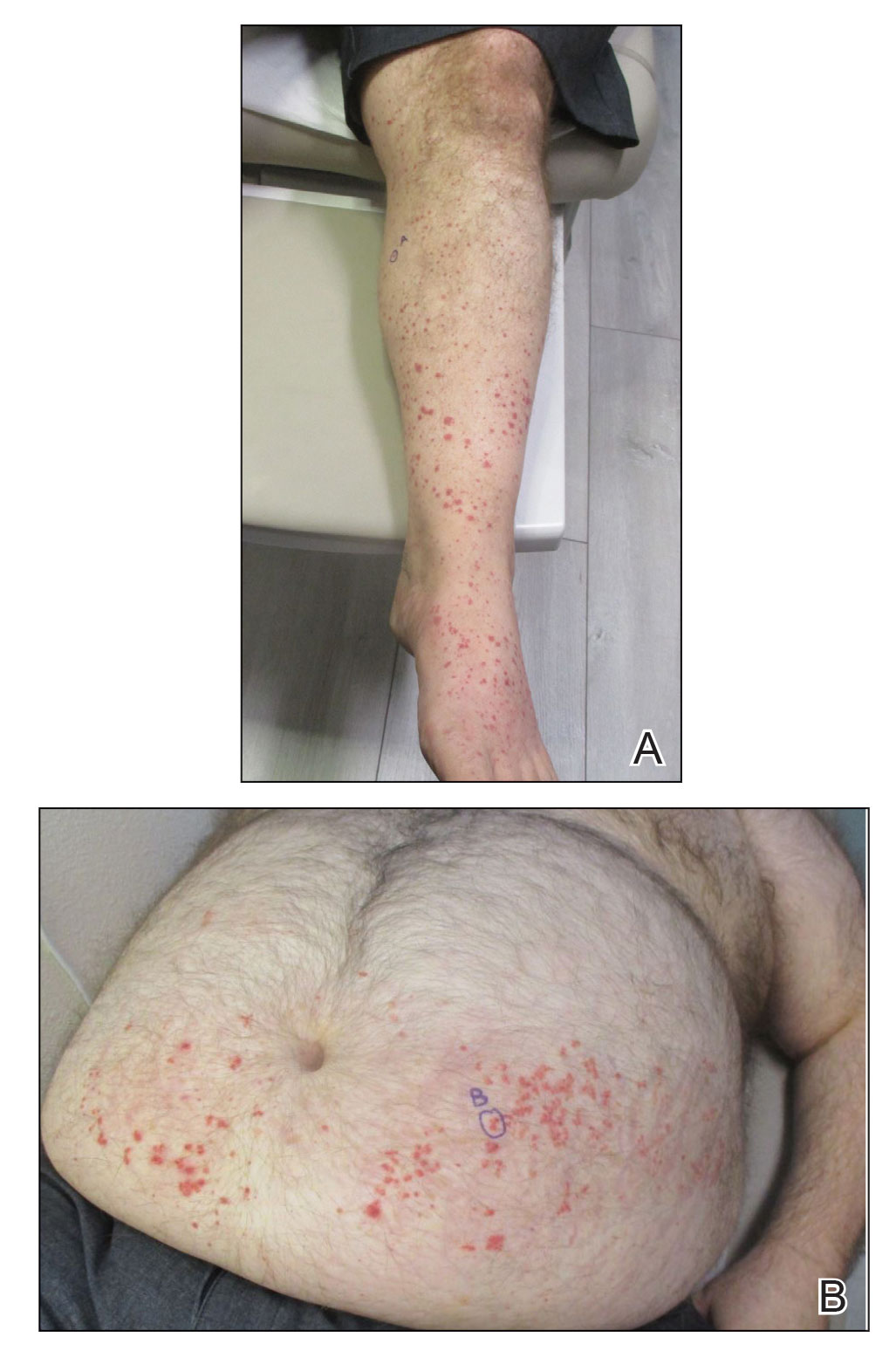
Pathology showed perivascular extravasation of erythrocytes, neutrophils, eosinophils, and leukocytoclasis surrounding blood vessels associated with fibrin (Figure 2). Direct immunofluorescence showed granular deposition of IgA, complement component 3, and fibrinogen in a superficial dermal vascular pattern (Figure 3). These results were consistent with IgA small-vessel vasculitis. One specimen was consistent with the patient’s known psoriasis.
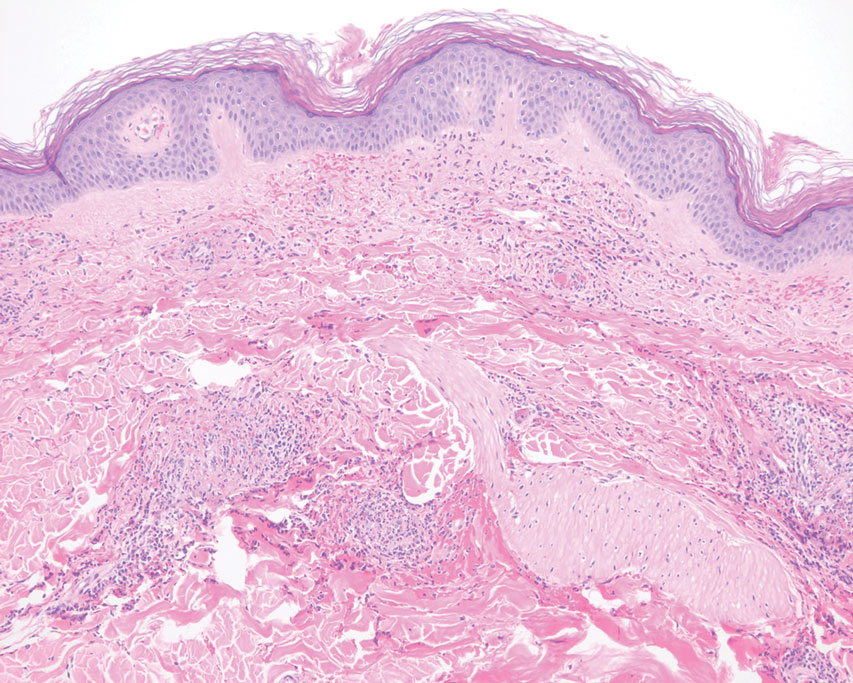
Urinalysis revealed moderate hemoglobinuria, and urine microscopy showed 174 red blood cells per high-power field. Creatinine was high at 1.87 mg/dL (reference range, <1.34 mg/dL; patient’s baseline, 0.81 mg/dL) and glomerular filtration rate was low (42 mL/min, patient’s baseline, >60 mL/min [reference range, 90–120 mL/min]). Erythrocyte sedimentation rate (21 mm/h [reference range, 0–22 mm/h]) and C-reactive protein were elevated (2.2 mg/dL [reference range, 0.3–1.0 mg/dL]). Given his history of cutaneous MRSA infections, a bacterial culture swab was collected from the skin surface to check for colonization, which showed moderate growth of MRSA. Naproxen was discontinued over concern of worsening the patient’s renal status. The patient was instructed to rest at home with his legs elevated, wear compression socks when ambulatory, use chlorhexidine antiseptic daily as a body wash when showering, and apply mupirocin three times daily to the biopsy sites. He was referred to urology for his microhematuria, where cystoscopy revealed no abnormalities.A month passed with no improvement of the patient’s cutaneous vasculitis, and his psoriatic arthritis worsened without his usual use of naproxen. He developed abdominal pain and loss of appetite. A prednisone taper was ordered starting at 40 mg/d (28.8 mg/kg), which provided relief of the skin and joint symptoms only until the course was completed 12 days later.
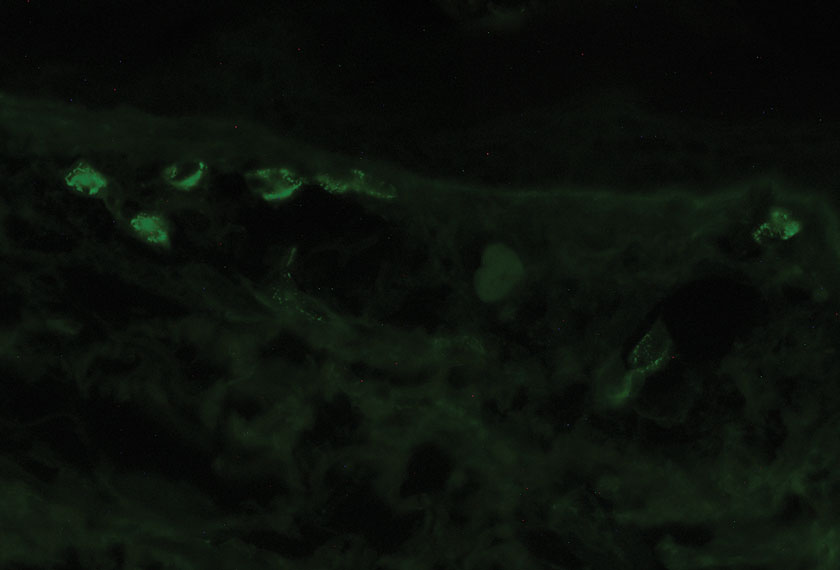
Five weeks after the initial presentation, the patient returned with a more severe eruption consisting of innumerable purpuric papules that coalesced in plaques on the abdomen, arms, and legs. He also had erythematous facial pustules and mild palmar petechiae (Figure 4). Three biopsies were performed, including 1 for DIF and 1 from a pustule on the forehead. Histology and DIF were again consistent with IgA small-vessel vasculitis. The forehead biopsy was compatible with steroid acne (attributed to recent prednisone use) and psoriasis.
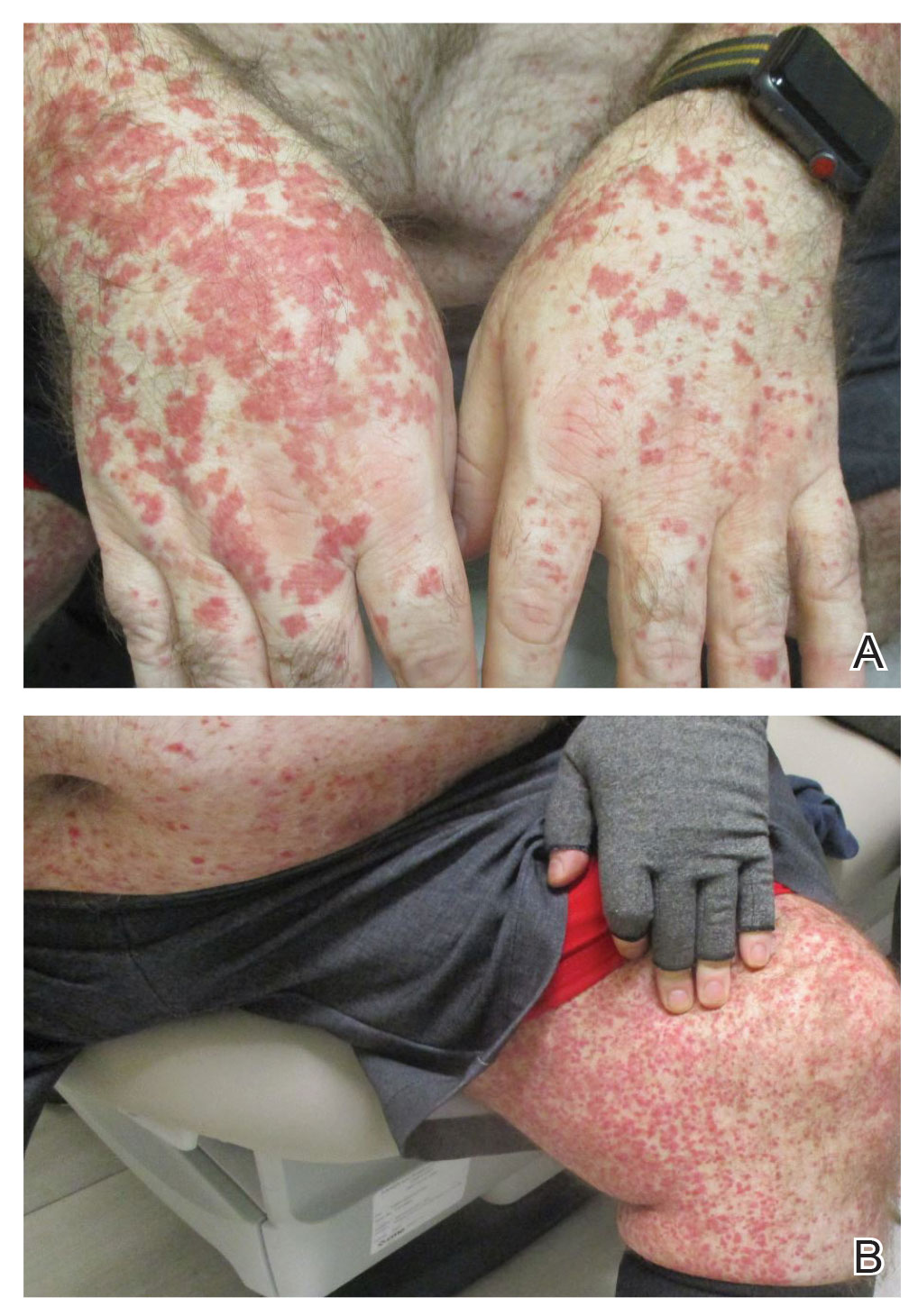
Rheumatology was consulted, and adalimumab was discontinued 6 weeks after the initial presentation out of concern for drug-induced cutaneous vasculitis. Vasculitis work-up was unremarkable, including antineutrophil cytoplasmic antibodies, rheumatoid factor, cyclic citrullinated peptide, and serum protein electrophoresis. Oral dapsone was started at 100 mg/d, with the tentative plan of starting secukinumab if cutaneous symptoms improved. For 3 weeks, the patient’s cutaneous symptoms steadily improved.
Nine weeks after initial presentation to dermatology (3 weeks after discontinuing adalimumab) the patient self-administered his first dose of secukinumab at home. Several hours later, he reported sudden reappearance of vasculitis. He denied diarrhea, abdominal pain, bowel movement urgency, fevers, fatigue, and unintentional weight loss. Antistreptolysin O and hepatitis A antibodies were negative. He was instructed to hold secukinumab indefinitely.
Four weeks after his only secukinumab injection, the patient reported another episode of acute worsening cutaneous symptoms. A 4-week prednisone taper starting at 40 mg/d was ordered. Computed tomography of the chest, abdomen, and pelvis to rule out internal malignancy was unremarkable. Around this time, the patient reported major emotional distress related to an unexpected death in his family, which added to a gradual increase in his stress level related to the COVID-19 pandemic.
Three weeks later, dapsone was increased to 100 mg twice daily on account of the patient’s adiposity and lack of cutaneous improvement on the lower dose. Subsequently, the vasculitis rapidly improved for 2 weeks. The patient then reported symptoms of headache, dizziness, and chills. He was tested for COVID-19 and was negative. Six weeks after increasing the dapsone dose (5 months after initial presentation), the skin was normalizing, showing only faintly hyperpigmented macules confined to areas of resolved vasculitis (forearms, abdomen, legs).
The patient had been on dapsone 100 mg twice daily for 3 months when he was started on ustekinumab (90 mg at weeks 0 and 4, with planned doses every 12 weeks) for psoriatic arthritis in hopes of withdrawing dapsone. His cutaneous symptoms have remained well controlled on this regimen for 18 months. Lowering of dapsone below 100 mg daily has resulted in recurrent mild vasculitis symptoms; he now maintains the once-daily dosing without negative side effects.
Comment
IgA vasculitis is a form of cutaneous small-vessel leukocytoclastic vasculitis (LCV) characterized by episodes of palpable purpura on the extensor surfaces of the arms and legs that may be associated with arthritis, abdominal pain, and/or hematuria. Although vasculitis is a known potential adverse effect of anti–tumor necrosis factor (TNF) α therapy, cases of adalimumab-induced IgA vasculitis are uncommon. As use of more targeted therapies for psoriasis and psoriatic arthritis, such as the IL-17 inhibitor secukinumab, increases so do reports of associated adverse events. Of 6 previously reported cases of secukinumab-associated vasculitis, at least 4 were IgA vasculitis (Table).1-6 Another case described one patient with rheumatoid arthritis undergoing secukinumab treatment who experienced necrotizing glomerulonephritis; however, the authors concluded secukinumab likely was not causative in that case, as serologies and urinalyses suggested gradual onset of the process prior to initiating the medication.7
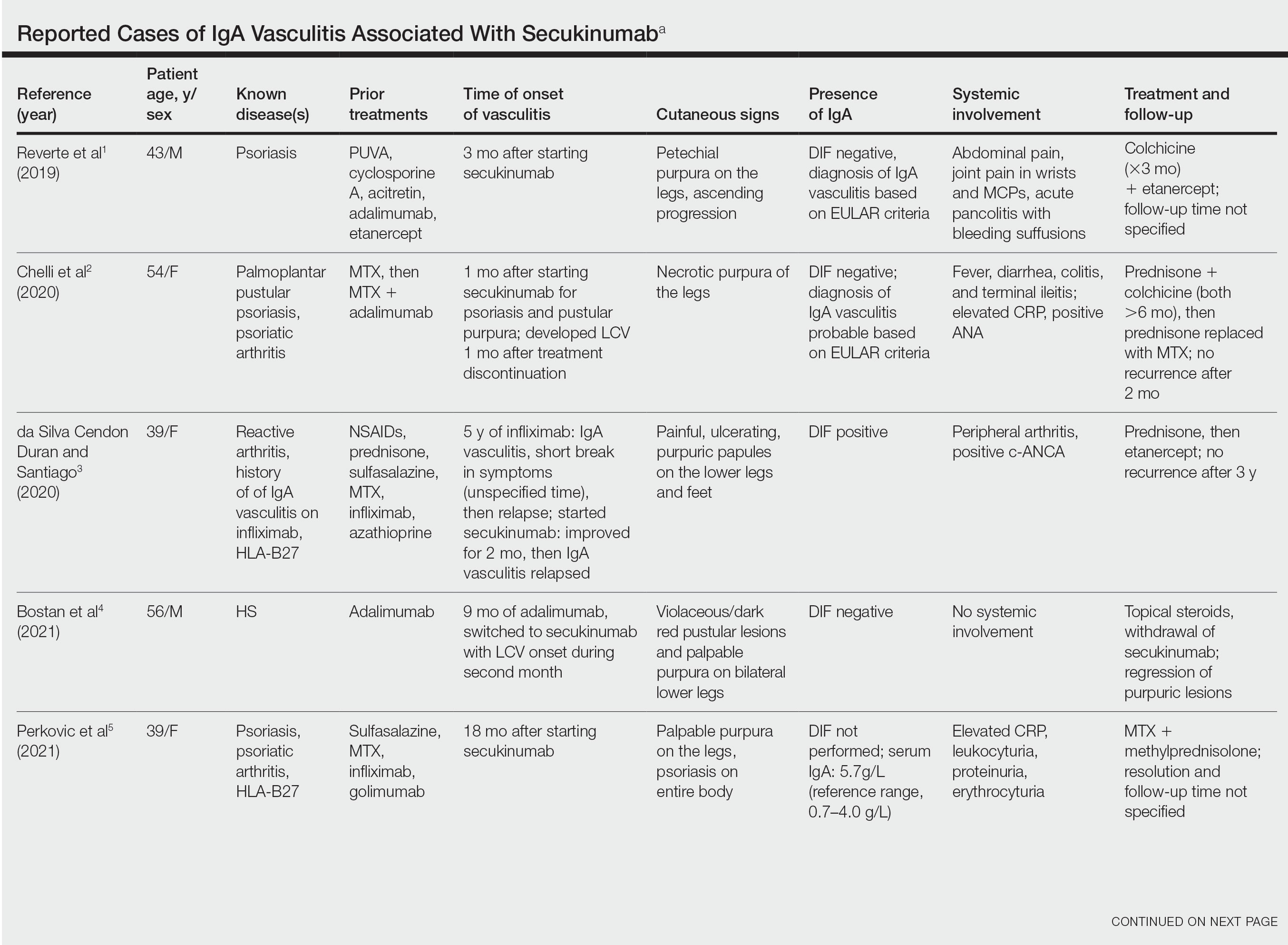
The exact pathogenesis of IgA vasculitis is unclear, but a prevailing theory involves the dysregulation of IgA synthesis and metabolism. Other than increased serum levels of transforming growth factor β, which is a major stimulating factor for IgA production, it also has been hypothesized that the presence of aberrantly hypoglycosylated IgA exposes an autoepitope for recognition by other pathogenic IgG and IgA, leading to the formation of large immune complexes that can readily deposit in postcapillary venules. The deposition of IgA immune complexes in postcapillary venules and the subsequent activation of the complement system causes direct damage to the endothelial cells of vessel walls. This complement activation is evidenced by vascular complement component 3 deposition on DIF (a nonspecific feature of LCV). Chemotaxis of neutrophils ensues, followed by their firm adherence and transendothelial migration (mediated by monocyte chemoattractant protein 1 [MCP-1]). Neutrophil degranulation releases reactive oxygen species and cytokines, which in turn recruit additional leukocytes to the area of inflammation, subsequently undergoing degeneration (leukocytoclasis). Microvascular permeability also is enhanced by MCP-1, allowing exudation of serum, erythrocytes, and fibrin. In the setting of elevated circulating TNF and IL-1, endothelium is stimulated to activate the intrinsic and extrinsic coagulation pathways. This decreases endothelial fibrinolytic activity, leading to thrombosis. The high venous pressure and low fibrinolytic activity in the lower legs explains why vasculitic lesions often are confined to or begin in this distribution.1,8-10
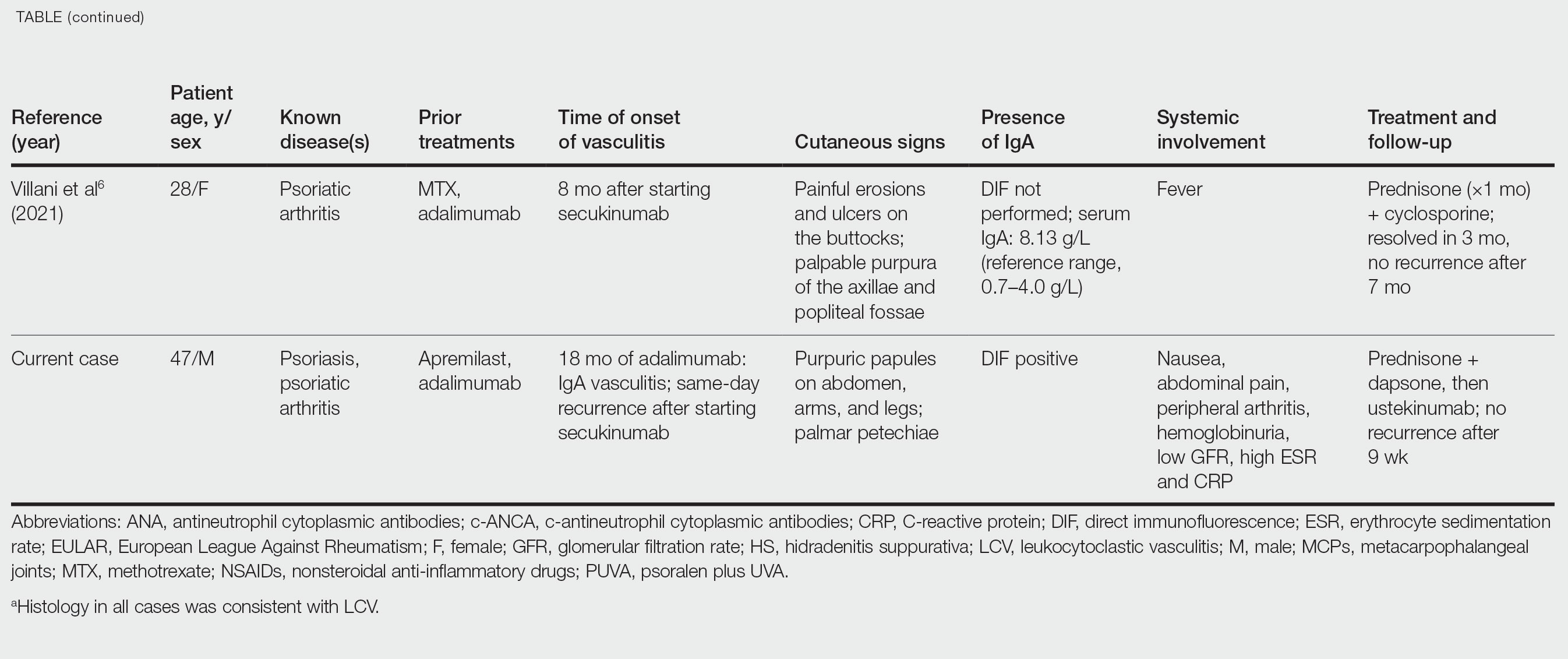
There also are noteworthy roles for cytokines in LCV. Circulating transforming growth factor β and IL-6—which are necessary for development of T helper 17 (TH17) cells and production of IL-17—are higher in patients with LCV compared to controls. Peripheral blood monocytes in patients with LCV demonstrate higher production of IL-17. Once TH17 cells develop, their survival and phenotype are maintained by IL-23 (considered the master regulator of TH17 differentiation). IL-17 is a potent chemoattractant of IL-8 (CXCL8) and MCP-1, both of which promote neutrophil-mediated perivascular inflammation. The IL-23 and IL-17 pathways implicated in the pathogenesis of psoriasis also cause neutrophil activation and upregulate transcription of proinflammatory cytokines (IL-1, IL-6, IL-8, and TNF-α), which overlap with those implicated in LCV. Autoimmune disease generally entails some positive feedback loop of progressively severe self-recognition and tissue destruction by the immune system. These shared cytokinetic processes may explain how the internal environment of psoriasis could perpetuate IgA vasculitis.1,2,8,10-12
The mechanisms underlying vasculitis associated with adalimumab are unclear, but hypotheses involve direct toxicity on vessels, capillary deposition of anti-TNF/TNF immune complexes, or an inflammatory process resulting in autoantibodies. Similar hypotheses are posited for secukinumab-associated vasculitis, including deposition of secukinumab–IL-17 complexes. Anti–TNF-α medications may increase TH17 cell numbers, leading to increased production of IL-22 and a resultant immunologic microenvironment conducive to vasculitis. All 6 published cases of secukinumab-associated vasculitis that we found had received prior treatment with a TNF-α blocker, but only 1 had occurrence of vasculitis during that treatment.1-6,10
In the 6 cases we reviewed, the time from starting secukinumab to onset of vasculitis ranged from 1 to 18 months. Our patient’s same-day re-emergence of vasculitis after his first secukinumab dose was so acute that we were skeptical of secukinumab as a potential trigger; this may simply have been coincident to the natural waxing and waning of the vasculitis (although onset of IgA vasculitis within 1 day of starting anti–TNF-α therapy has been reported).1-6,13
Specific associations of IgA vasculitis are many and can include bacterial organisms such as Helicobacter pylori, streptococci, and staphylococci. Although internal mucous membrane infections are considered more linked because of the surveillance role of IgA predominantly in mucosal tissues, it is possible that our patient with cutaneous MRSA harbored the same within the nasal mucosa. Our patient also received multiple vaccinations outside our department throughout his clinical course (2 hepatitis B and 1 pneumococcal conjugate), which are known potential triggers for vasculitis. Psychological stress is a known trigger for psoriasis, and given the cytokinetic relationship of psoriasis to vasculitis described previously, it may have indirectly contributed to vasculitis in our case. The anxiety associated with being immunosuppressed during the COVID-19 pandemic and bereavement of losing a family member may have contributed to the refractory nature of our patient’s condition. Renal involvement is relatively common in adults with IgA vasculitis and so should be ruled out, as should occult internal malignancy.8,10,14
It is unclear which of the above factors was causative in our case, but a multifactorial process is likely. Treatment of monoclonal antibody–associated vasculitis entails investigating for triggers and systemic involvement, removing the most likely culprit, quelling the vasculitis acutely, avoiding known potential exacerbators, and introducing an alternative long-term immunomodulant. In all 6 reported similar cases, discontinuation of secukinumab and initiation of prednisone or colchicine led to resolution.1-6 Dapsone also is acceptable for acute control of IgA vasculitis, although this medication is highly lipid soluble and penetrates well into various tissues.15 Thus, lower doses may prove ineffective for obese patients, as was demonstrated in our case. Given the known potential of vaccinations, infections, and other factors (eg, alcohol, penicillin) to trigger IgA vasculitis, these should be avoided.10
Blockade of IL-23 with ustekinumab has been suggested by other authors encountering secukinumab-associated vasculitis, as IL-23 is the main driver and sustainer of TH17 cell differentiation.8 Although 6 previously reported cases of secukinumab-associated vasculitis achieved resolution without long-term recurrence, none did so using an IL-23 inhibitor (nor had any of the described patients received IL-23 inhibitors previously).1-6 Given the established safety of IL-23 inhibitors and that they theoretically are well suited for this unique circumstance (by ceasing the main causative cytokine cascades “upstream”) and were efficacious in quickly resolving our patient’s vasculitis, we suggest that ustekinumab may represent
- Reverte M, Etienne M, Fouchard M, et al. Occurrence of Henoch-Schönlein purpura in a patient treated with secukinumab. J Eur Acad Dermatol Venereol. 2019;33:E455-E457.
- Chelli C, Loget J, Vanhaecke C, et al. Cutaneous vasculitis with gut involvement during secukinumab treatment for psoriatic arthritis. Acta Derm Venereol. 2020;100:adv00077.
- da Silva Cendon Duran C, Santiago MB. Cutaneous vasculitis during secukinumab treatment. Eur J Case Rep Intern Med. 2020;7:001815.
- Bostan E, Gulseren D, Yalici-Armagan B, et al. Vasculitis during certolizumab pegol and secukinumab treatment: report of two cases. Dermatol Ther. 2021;34:E15007.
- Perkovic D, Simac P, Katic J. IgA vasculitis during secukinumab therapy. Clin Rheumatol. 2021;40:2071-2073.
- Villani A, DE Fata Salvatores G, Nappa P, et al. Cutaneous leucocytoclastic vasculitis during secukinumab treatment. Ital J Dermatol Venerol. 2021;156(suppl 1 to no. 6):9-10.
- Góis M, Messias A, Carvalho D, et al. MPO-ANCA-associated necrotizing glomerulonephritis in rheumatoid arthritis; a case report and review of literature. J Nephropathol. 2017;6:58-62.
- Jen HY, Chuang YH, Lin SC, et al. Increased serum interleukin-17 and peripheral Th17 cells in children with acute Henoch-Schönlein purpura. Pediatr Allergy Immunol. 2011;22:862-868.
- Hetland LE, Susrud KS, Lindahl KH, et al. Henoch-Schönlein purpura: a literature review. Acta Derm Venereol 2017;97:1160-1166.
- Weedon D. The vasculopathic reaction pattern. In: Houston M, Davie B, eds. Weedon’s Skin Pathology. 3rd ed. Elsevier Limited; 2010:207-211.
- Puig L. Paradoxical reactions: anti-TNFα ants, ustekinumab, secukinumab, ixekizumab, and others. Curr Probl Dermatol. 2018;53:49-63.
- Nestle F, Kaplan D, Barker J. Psoriasis. N Engl J Med. 2009;361:496-509.
- Pinheiro RR, Lencastre A. Henoch-Schönlein purpura during anti-TNFα therapy: a fortuitous event or an indication to stop therapy? Eur J Dermatol. 2017;27:304-305.
- Hello CL, Cohen P, Bousser MG, et al. Suspected hepatitis B vaccination related vasculitis. J Rheumatol. 1999;26:191-194.
- Wolverton SE. Dapsone. In: Wolverton SE, Wu JJ, eds. Comprehensive Dermatologic Drug Therapy. 4th ed. Elsevier, Inc; 2021:222-231.
Case Report
A 47-year-old man presented with a sudden-onset rash consisting of red bumps on the abdomen and legs that had been ongoing for several days. He had known psoriasis and psoriatic arthritis that had been well controlled with adalimumab for the last 18 months. He reported concurrent onset of nausea but denied fevers, chills, night sweats, unintentional weight loss, abdominal pain, and pruritus. He endorsed prior cutaneous infections of methicillin-resistant Staphylococcus aureus (MRSA). His medical history also included diabetes mellitus, hypertension, and obesity. His other medications included oral losartan-hydrochlorothiazide, amlodipine, naproxen, and atorvastatin.
Physical examination revealed numerous thin purpuric papules—some with adherent scale—distributed on the lower legs, extensor forearms, and abdomen. Abdominal lesions were confined to weight-related striae (Figure 1). The palms, soles, oral mucosa, and face were spared. Three punch biopsies were performed, including 1 for direct immunofluorescence (DIF), and the patient was instructed to apply clobetasol to the affected areas twice daily until further notice.

Pathology showed perivascular extravasation of erythrocytes, neutrophils, eosinophils, and leukocytoclasis surrounding blood vessels associated with fibrin (Figure 2). Direct immunofluorescence showed granular deposition of IgA, complement component 3, and fibrinogen in a superficial dermal vascular pattern (Figure 3). These results were consistent with IgA small-vessel vasculitis. One specimen was consistent with the patient’s known psoriasis.

Urinalysis revealed moderate hemoglobinuria, and urine microscopy showed 174 red blood cells per high-power field. Creatinine was high at 1.87 mg/dL (reference range, <1.34 mg/dL; patient’s baseline, 0.81 mg/dL) and glomerular filtration rate was low (42 mL/min, patient’s baseline, >60 mL/min [reference range, 90–120 mL/min]). Erythrocyte sedimentation rate (21 mm/h [reference range, 0–22 mm/h]) and C-reactive protein were elevated (2.2 mg/dL [reference range, 0.3–1.0 mg/dL]). Given his history of cutaneous MRSA infections, a bacterial culture swab was collected from the skin surface to check for colonization, which showed moderate growth of MRSA. Naproxen was discontinued over concern of worsening the patient’s renal status. The patient was instructed to rest at home with his legs elevated, wear compression socks when ambulatory, use chlorhexidine antiseptic daily as a body wash when showering, and apply mupirocin three times daily to the biopsy sites. He was referred to urology for his microhematuria, where cystoscopy revealed no abnormalities.A month passed with no improvement of the patient’s cutaneous vasculitis, and his psoriatic arthritis worsened without his usual use of naproxen. He developed abdominal pain and loss of appetite. A prednisone taper was ordered starting at 40 mg/d (28.8 mg/kg), which provided relief of the skin and joint symptoms only until the course was completed 12 days later.

Five weeks after the initial presentation, the patient returned with a more severe eruption consisting of innumerable purpuric papules that coalesced in plaques on the abdomen, arms, and legs. He also had erythematous facial pustules and mild palmar petechiae (Figure 4). Three biopsies were performed, including 1 for DIF and 1 from a pustule on the forehead. Histology and DIF were again consistent with IgA small-vessel vasculitis. The forehead biopsy was compatible with steroid acne (attributed to recent prednisone use) and psoriasis.

Rheumatology was consulted, and adalimumab was discontinued 6 weeks after the initial presentation out of concern for drug-induced cutaneous vasculitis. Vasculitis work-up was unremarkable, including antineutrophil cytoplasmic antibodies, rheumatoid factor, cyclic citrullinated peptide, and serum protein electrophoresis. Oral dapsone was started at 100 mg/d, with the tentative plan of starting secukinumab if cutaneous symptoms improved. For 3 weeks, the patient’s cutaneous symptoms steadily improved.
Nine weeks after initial presentation to dermatology (3 weeks after discontinuing adalimumab) the patient self-administered his first dose of secukinumab at home. Several hours later, he reported sudden reappearance of vasculitis. He denied diarrhea, abdominal pain, bowel movement urgency, fevers, fatigue, and unintentional weight loss. Antistreptolysin O and hepatitis A antibodies were negative. He was instructed to hold secukinumab indefinitely.
Four weeks after his only secukinumab injection, the patient reported another episode of acute worsening cutaneous symptoms. A 4-week prednisone taper starting at 40 mg/d was ordered. Computed tomography of the chest, abdomen, and pelvis to rule out internal malignancy was unremarkable. Around this time, the patient reported major emotional distress related to an unexpected death in his family, which added to a gradual increase in his stress level related to the COVID-19 pandemic.
Three weeks later, dapsone was increased to 100 mg twice daily on account of the patient’s adiposity and lack of cutaneous improvement on the lower dose. Subsequently, the vasculitis rapidly improved for 2 weeks. The patient then reported symptoms of headache, dizziness, and chills. He was tested for COVID-19 and was negative. Six weeks after increasing the dapsone dose (5 months after initial presentation), the skin was normalizing, showing only faintly hyperpigmented macules confined to areas of resolved vasculitis (forearms, abdomen, legs).
The patient had been on dapsone 100 mg twice daily for 3 months when he was started on ustekinumab (90 mg at weeks 0 and 4, with planned doses every 12 weeks) for psoriatic arthritis in hopes of withdrawing dapsone. His cutaneous symptoms have remained well controlled on this regimen for 18 months. Lowering of dapsone below 100 mg daily has resulted in recurrent mild vasculitis symptoms; he now maintains the once-daily dosing without negative side effects.
Comment
IgA vasculitis is a form of cutaneous small-vessel leukocytoclastic vasculitis (LCV) characterized by episodes of palpable purpura on the extensor surfaces of the arms and legs that may be associated with arthritis, abdominal pain, and/or hematuria. Although vasculitis is a known potential adverse effect of anti–tumor necrosis factor (TNF) α therapy, cases of adalimumab-induced IgA vasculitis are uncommon. As use of more targeted therapies for psoriasis and psoriatic arthritis, such as the IL-17 inhibitor secukinumab, increases so do reports of associated adverse events. Of 6 previously reported cases of secukinumab-associated vasculitis, at least 4 were IgA vasculitis (Table).1-6 Another case described one patient with rheumatoid arthritis undergoing secukinumab treatment who experienced necrotizing glomerulonephritis; however, the authors concluded secukinumab likely was not causative in that case, as serologies and urinalyses suggested gradual onset of the process prior to initiating the medication.7

The exact pathogenesis of IgA vasculitis is unclear, but a prevailing theory involves the dysregulation of IgA synthesis and metabolism. Other than increased serum levels of transforming growth factor β, which is a major stimulating factor for IgA production, it also has been hypothesized that the presence of aberrantly hypoglycosylated IgA exposes an autoepitope for recognition by other pathogenic IgG and IgA, leading to the formation of large immune complexes that can readily deposit in postcapillary venules. The deposition of IgA immune complexes in postcapillary venules and the subsequent activation of the complement system causes direct damage to the endothelial cells of vessel walls. This complement activation is evidenced by vascular complement component 3 deposition on DIF (a nonspecific feature of LCV). Chemotaxis of neutrophils ensues, followed by their firm adherence and transendothelial migration (mediated by monocyte chemoattractant protein 1 [MCP-1]). Neutrophil degranulation releases reactive oxygen species and cytokines, which in turn recruit additional leukocytes to the area of inflammation, subsequently undergoing degeneration (leukocytoclasis). Microvascular permeability also is enhanced by MCP-1, allowing exudation of serum, erythrocytes, and fibrin. In the setting of elevated circulating TNF and IL-1, endothelium is stimulated to activate the intrinsic and extrinsic coagulation pathways. This decreases endothelial fibrinolytic activity, leading to thrombosis. The high venous pressure and low fibrinolytic activity in the lower legs explains why vasculitic lesions often are confined to or begin in this distribution.1,8-10

There also are noteworthy roles for cytokines in LCV. Circulating transforming growth factor β and IL-6—which are necessary for development of T helper 17 (TH17) cells and production of IL-17—are higher in patients with LCV compared to controls. Peripheral blood monocytes in patients with LCV demonstrate higher production of IL-17. Once TH17 cells develop, their survival and phenotype are maintained by IL-23 (considered the master regulator of TH17 differentiation). IL-17 is a potent chemoattractant of IL-8 (CXCL8) and MCP-1, both of which promote neutrophil-mediated perivascular inflammation. The IL-23 and IL-17 pathways implicated in the pathogenesis of psoriasis also cause neutrophil activation and upregulate transcription of proinflammatory cytokines (IL-1, IL-6, IL-8, and TNF-α), which overlap with those implicated in LCV. Autoimmune disease generally entails some positive feedback loop of progressively severe self-recognition and tissue destruction by the immune system. These shared cytokinetic processes may explain how the internal environment of psoriasis could perpetuate IgA vasculitis.1,2,8,10-12
The mechanisms underlying vasculitis associated with adalimumab are unclear, but hypotheses involve direct toxicity on vessels, capillary deposition of anti-TNF/TNF immune complexes, or an inflammatory process resulting in autoantibodies. Similar hypotheses are posited for secukinumab-associated vasculitis, including deposition of secukinumab–IL-17 complexes. Anti–TNF-α medications may increase TH17 cell numbers, leading to increased production of IL-22 and a resultant immunologic microenvironment conducive to vasculitis. All 6 published cases of secukinumab-associated vasculitis that we found had received prior treatment with a TNF-α blocker, but only 1 had occurrence of vasculitis during that treatment.1-6,10
In the 6 cases we reviewed, the time from starting secukinumab to onset of vasculitis ranged from 1 to 18 months. Our patient’s same-day re-emergence of vasculitis after his first secukinumab dose was so acute that we were skeptical of secukinumab as a potential trigger; this may simply have been coincident to the natural waxing and waning of the vasculitis (although onset of IgA vasculitis within 1 day of starting anti–TNF-α therapy has been reported).1-6,13
Specific associations of IgA vasculitis are many and can include bacterial organisms such as Helicobacter pylori, streptococci, and staphylococci. Although internal mucous membrane infections are considered more linked because of the surveillance role of IgA predominantly in mucosal tissues, it is possible that our patient with cutaneous MRSA harbored the same within the nasal mucosa. Our patient also received multiple vaccinations outside our department throughout his clinical course (2 hepatitis B and 1 pneumococcal conjugate), which are known potential triggers for vasculitis. Psychological stress is a known trigger for psoriasis, and given the cytokinetic relationship of psoriasis to vasculitis described previously, it may have indirectly contributed to vasculitis in our case. The anxiety associated with being immunosuppressed during the COVID-19 pandemic and bereavement of losing a family member may have contributed to the refractory nature of our patient’s condition. Renal involvement is relatively common in adults with IgA vasculitis and so should be ruled out, as should occult internal malignancy.8,10,14
It is unclear which of the above factors was causative in our case, but a multifactorial process is likely. Treatment of monoclonal antibody–associated vasculitis entails investigating for triggers and systemic involvement, removing the most likely culprit, quelling the vasculitis acutely, avoiding known potential exacerbators, and introducing an alternative long-term immunomodulant. In all 6 reported similar cases, discontinuation of secukinumab and initiation of prednisone or colchicine led to resolution.1-6 Dapsone also is acceptable for acute control of IgA vasculitis, although this medication is highly lipid soluble and penetrates well into various tissues.15 Thus, lower doses may prove ineffective for obese patients, as was demonstrated in our case. Given the known potential of vaccinations, infections, and other factors (eg, alcohol, penicillin) to trigger IgA vasculitis, these should be avoided.10
Blockade of IL-23 with ustekinumab has been suggested by other authors encountering secukinumab-associated vasculitis, as IL-23 is the main driver and sustainer of TH17 cell differentiation.8 Although 6 previously reported cases of secukinumab-associated vasculitis achieved resolution without long-term recurrence, none did so using an IL-23 inhibitor (nor had any of the described patients received IL-23 inhibitors previously).1-6 Given the established safety of IL-23 inhibitors and that they theoretically are well suited for this unique circumstance (by ceasing the main causative cytokine cascades “upstream”) and were efficacious in quickly resolving our patient’s vasculitis, we suggest that ustekinumab may represent
Case Report
A 47-year-old man presented with a sudden-onset rash consisting of red bumps on the abdomen and legs that had been ongoing for several days. He had known psoriasis and psoriatic arthritis that had been well controlled with adalimumab for the last 18 months. He reported concurrent onset of nausea but denied fevers, chills, night sweats, unintentional weight loss, abdominal pain, and pruritus. He endorsed prior cutaneous infections of methicillin-resistant Staphylococcus aureus (MRSA). His medical history also included diabetes mellitus, hypertension, and obesity. His other medications included oral losartan-hydrochlorothiazide, amlodipine, naproxen, and atorvastatin.
Physical examination revealed numerous thin purpuric papules—some with adherent scale—distributed on the lower legs, extensor forearms, and abdomen. Abdominal lesions were confined to weight-related striae (Figure 1). The palms, soles, oral mucosa, and face were spared. Three punch biopsies were performed, including 1 for direct immunofluorescence (DIF), and the patient was instructed to apply clobetasol to the affected areas twice daily until further notice.

Pathology showed perivascular extravasation of erythrocytes, neutrophils, eosinophils, and leukocytoclasis surrounding blood vessels associated with fibrin (Figure 2). Direct immunofluorescence showed granular deposition of IgA, complement component 3, and fibrinogen in a superficial dermal vascular pattern (Figure 3). These results were consistent with IgA small-vessel vasculitis. One specimen was consistent with the patient’s known psoriasis.

Urinalysis revealed moderate hemoglobinuria, and urine microscopy showed 174 red blood cells per high-power field. Creatinine was high at 1.87 mg/dL (reference range, <1.34 mg/dL; patient’s baseline, 0.81 mg/dL) and glomerular filtration rate was low (42 mL/min, patient’s baseline, >60 mL/min [reference range, 90–120 mL/min]). Erythrocyte sedimentation rate (21 mm/h [reference range, 0–22 mm/h]) and C-reactive protein were elevated (2.2 mg/dL [reference range, 0.3–1.0 mg/dL]). Given his history of cutaneous MRSA infections, a bacterial culture swab was collected from the skin surface to check for colonization, which showed moderate growth of MRSA. Naproxen was discontinued over concern of worsening the patient’s renal status. The patient was instructed to rest at home with his legs elevated, wear compression socks when ambulatory, use chlorhexidine antiseptic daily as a body wash when showering, and apply mupirocin three times daily to the biopsy sites. He was referred to urology for his microhematuria, where cystoscopy revealed no abnormalities.A month passed with no improvement of the patient’s cutaneous vasculitis, and his psoriatic arthritis worsened without his usual use of naproxen. He developed abdominal pain and loss of appetite. A prednisone taper was ordered starting at 40 mg/d (28.8 mg/kg), which provided relief of the skin and joint symptoms only until the course was completed 12 days later.

Five weeks after the initial presentation, the patient returned with a more severe eruption consisting of innumerable purpuric papules that coalesced in plaques on the abdomen, arms, and legs. He also had erythematous facial pustules and mild palmar petechiae (Figure 4). Three biopsies were performed, including 1 for DIF and 1 from a pustule on the forehead. Histology and DIF were again consistent with IgA small-vessel vasculitis. The forehead biopsy was compatible with steroid acne (attributed to recent prednisone use) and psoriasis.

Rheumatology was consulted, and adalimumab was discontinued 6 weeks after the initial presentation out of concern for drug-induced cutaneous vasculitis. Vasculitis work-up was unremarkable, including antineutrophil cytoplasmic antibodies, rheumatoid factor, cyclic citrullinated peptide, and serum protein electrophoresis. Oral dapsone was started at 100 mg/d, with the tentative plan of starting secukinumab if cutaneous symptoms improved. For 3 weeks, the patient’s cutaneous symptoms steadily improved.
Nine weeks after initial presentation to dermatology (3 weeks after discontinuing adalimumab) the patient self-administered his first dose of secukinumab at home. Several hours later, he reported sudden reappearance of vasculitis. He denied diarrhea, abdominal pain, bowel movement urgency, fevers, fatigue, and unintentional weight loss. Antistreptolysin O and hepatitis A antibodies were negative. He was instructed to hold secukinumab indefinitely.
Four weeks after his only secukinumab injection, the patient reported another episode of acute worsening cutaneous symptoms. A 4-week prednisone taper starting at 40 mg/d was ordered. Computed tomography of the chest, abdomen, and pelvis to rule out internal malignancy was unremarkable. Around this time, the patient reported major emotional distress related to an unexpected death in his family, which added to a gradual increase in his stress level related to the COVID-19 pandemic.
Three weeks later, dapsone was increased to 100 mg twice daily on account of the patient’s adiposity and lack of cutaneous improvement on the lower dose. Subsequently, the vasculitis rapidly improved for 2 weeks. The patient then reported symptoms of headache, dizziness, and chills. He was tested for COVID-19 and was negative. Six weeks after increasing the dapsone dose (5 months after initial presentation), the skin was normalizing, showing only faintly hyperpigmented macules confined to areas of resolved vasculitis (forearms, abdomen, legs).
The patient had been on dapsone 100 mg twice daily for 3 months when he was started on ustekinumab (90 mg at weeks 0 and 4, with planned doses every 12 weeks) for psoriatic arthritis in hopes of withdrawing dapsone. His cutaneous symptoms have remained well controlled on this regimen for 18 months. Lowering of dapsone below 100 mg daily has resulted in recurrent mild vasculitis symptoms; he now maintains the once-daily dosing without negative side effects.
Comment
IgA vasculitis is a form of cutaneous small-vessel leukocytoclastic vasculitis (LCV) characterized by episodes of palpable purpura on the extensor surfaces of the arms and legs that may be associated with arthritis, abdominal pain, and/or hematuria. Although vasculitis is a known potential adverse effect of anti–tumor necrosis factor (TNF) α therapy, cases of adalimumab-induced IgA vasculitis are uncommon. As use of more targeted therapies for psoriasis and psoriatic arthritis, such as the IL-17 inhibitor secukinumab, increases so do reports of associated adverse events. Of 6 previously reported cases of secukinumab-associated vasculitis, at least 4 were IgA vasculitis (Table).1-6 Another case described one patient with rheumatoid arthritis undergoing secukinumab treatment who experienced necrotizing glomerulonephritis; however, the authors concluded secukinumab likely was not causative in that case, as serologies and urinalyses suggested gradual onset of the process prior to initiating the medication.7

The exact pathogenesis of IgA vasculitis is unclear, but a prevailing theory involves the dysregulation of IgA synthesis and metabolism. Other than increased serum levels of transforming growth factor β, which is a major stimulating factor for IgA production, it also has been hypothesized that the presence of aberrantly hypoglycosylated IgA exposes an autoepitope for recognition by other pathogenic IgG and IgA, leading to the formation of large immune complexes that can readily deposit in postcapillary venules. The deposition of IgA immune complexes in postcapillary venules and the subsequent activation of the complement system causes direct damage to the endothelial cells of vessel walls. This complement activation is evidenced by vascular complement component 3 deposition on DIF (a nonspecific feature of LCV). Chemotaxis of neutrophils ensues, followed by their firm adherence and transendothelial migration (mediated by monocyte chemoattractant protein 1 [MCP-1]). Neutrophil degranulation releases reactive oxygen species and cytokines, which in turn recruit additional leukocytes to the area of inflammation, subsequently undergoing degeneration (leukocytoclasis). Microvascular permeability also is enhanced by MCP-1, allowing exudation of serum, erythrocytes, and fibrin. In the setting of elevated circulating TNF and IL-1, endothelium is stimulated to activate the intrinsic and extrinsic coagulation pathways. This decreases endothelial fibrinolytic activity, leading to thrombosis. The high venous pressure and low fibrinolytic activity in the lower legs explains why vasculitic lesions often are confined to or begin in this distribution.1,8-10

There also are noteworthy roles for cytokines in LCV. Circulating transforming growth factor β and IL-6—which are necessary for development of T helper 17 (TH17) cells and production of IL-17—are higher in patients with LCV compared to controls. Peripheral blood monocytes in patients with LCV demonstrate higher production of IL-17. Once TH17 cells develop, their survival and phenotype are maintained by IL-23 (considered the master regulator of TH17 differentiation). IL-17 is a potent chemoattractant of IL-8 (CXCL8) and MCP-1, both of which promote neutrophil-mediated perivascular inflammation. The IL-23 and IL-17 pathways implicated in the pathogenesis of psoriasis also cause neutrophil activation and upregulate transcription of proinflammatory cytokines (IL-1, IL-6, IL-8, and TNF-α), which overlap with those implicated in LCV. Autoimmune disease generally entails some positive feedback loop of progressively severe self-recognition and tissue destruction by the immune system. These shared cytokinetic processes may explain how the internal environment of psoriasis could perpetuate IgA vasculitis.1,2,8,10-12
The mechanisms underlying vasculitis associated with adalimumab are unclear, but hypotheses involve direct toxicity on vessels, capillary deposition of anti-TNF/TNF immune complexes, or an inflammatory process resulting in autoantibodies. Similar hypotheses are posited for secukinumab-associated vasculitis, including deposition of secukinumab–IL-17 complexes. Anti–TNF-α medications may increase TH17 cell numbers, leading to increased production of IL-22 and a resultant immunologic microenvironment conducive to vasculitis. All 6 published cases of secukinumab-associated vasculitis that we found had received prior treatment with a TNF-α blocker, but only 1 had occurrence of vasculitis during that treatment.1-6,10
In the 6 cases we reviewed, the time from starting secukinumab to onset of vasculitis ranged from 1 to 18 months. Our patient’s same-day re-emergence of vasculitis after his first secukinumab dose was so acute that we were skeptical of secukinumab as a potential trigger; this may simply have been coincident to the natural waxing and waning of the vasculitis (although onset of IgA vasculitis within 1 day of starting anti–TNF-α therapy has been reported).1-6,13
Specific associations of IgA vasculitis are many and can include bacterial organisms such as Helicobacter pylori, streptococci, and staphylococci. Although internal mucous membrane infections are considered more linked because of the surveillance role of IgA predominantly in mucosal tissues, it is possible that our patient with cutaneous MRSA harbored the same within the nasal mucosa. Our patient also received multiple vaccinations outside our department throughout his clinical course (2 hepatitis B and 1 pneumococcal conjugate), which are known potential triggers for vasculitis. Psychological stress is a known trigger for psoriasis, and given the cytokinetic relationship of psoriasis to vasculitis described previously, it may have indirectly contributed to vasculitis in our case. The anxiety associated with being immunosuppressed during the COVID-19 pandemic and bereavement of losing a family member may have contributed to the refractory nature of our patient’s condition. Renal involvement is relatively common in adults with IgA vasculitis and so should be ruled out, as should occult internal malignancy.8,10,14
It is unclear which of the above factors was causative in our case, but a multifactorial process is likely. Treatment of monoclonal antibody–associated vasculitis entails investigating for triggers and systemic involvement, removing the most likely culprit, quelling the vasculitis acutely, avoiding known potential exacerbators, and introducing an alternative long-term immunomodulant. In all 6 reported similar cases, discontinuation of secukinumab and initiation of prednisone or colchicine led to resolution.1-6 Dapsone also is acceptable for acute control of IgA vasculitis, although this medication is highly lipid soluble and penetrates well into various tissues.15 Thus, lower doses may prove ineffective for obese patients, as was demonstrated in our case. Given the known potential of vaccinations, infections, and other factors (eg, alcohol, penicillin) to trigger IgA vasculitis, these should be avoided.10
Blockade of IL-23 with ustekinumab has been suggested by other authors encountering secukinumab-associated vasculitis, as IL-23 is the main driver and sustainer of TH17 cell differentiation.8 Although 6 previously reported cases of secukinumab-associated vasculitis achieved resolution without long-term recurrence, none did so using an IL-23 inhibitor (nor had any of the described patients received IL-23 inhibitors previously).1-6 Given the established safety of IL-23 inhibitors and that they theoretically are well suited for this unique circumstance (by ceasing the main causative cytokine cascades “upstream”) and were efficacious in quickly resolving our patient’s vasculitis, we suggest that ustekinumab may represent
- Reverte M, Etienne M, Fouchard M, et al. Occurrence of Henoch-Schönlein purpura in a patient treated with secukinumab. J Eur Acad Dermatol Venereol. 2019;33:E455-E457.
- Chelli C, Loget J, Vanhaecke C, et al. Cutaneous vasculitis with gut involvement during secukinumab treatment for psoriatic arthritis. Acta Derm Venereol. 2020;100:adv00077.
- da Silva Cendon Duran C, Santiago MB. Cutaneous vasculitis during secukinumab treatment. Eur J Case Rep Intern Med. 2020;7:001815.
- Bostan E, Gulseren D, Yalici-Armagan B, et al. Vasculitis during certolizumab pegol and secukinumab treatment: report of two cases. Dermatol Ther. 2021;34:E15007.
- Perkovic D, Simac P, Katic J. IgA vasculitis during secukinumab therapy. Clin Rheumatol. 2021;40:2071-2073.
- Villani A, DE Fata Salvatores G, Nappa P, et al. Cutaneous leucocytoclastic vasculitis during secukinumab treatment. Ital J Dermatol Venerol. 2021;156(suppl 1 to no. 6):9-10.
- Góis M, Messias A, Carvalho D, et al. MPO-ANCA-associated necrotizing glomerulonephritis in rheumatoid arthritis; a case report and review of literature. J Nephropathol. 2017;6:58-62.
- Jen HY, Chuang YH, Lin SC, et al. Increased serum interleukin-17 and peripheral Th17 cells in children with acute Henoch-Schönlein purpura. Pediatr Allergy Immunol. 2011;22:862-868.
- Hetland LE, Susrud KS, Lindahl KH, et al. Henoch-Schönlein purpura: a literature review. Acta Derm Venereol 2017;97:1160-1166.
- Weedon D. The vasculopathic reaction pattern. In: Houston M, Davie B, eds. Weedon’s Skin Pathology. 3rd ed. Elsevier Limited; 2010:207-211.
- Puig L. Paradoxical reactions: anti-TNFα ants, ustekinumab, secukinumab, ixekizumab, and others. Curr Probl Dermatol. 2018;53:49-63.
- Nestle F, Kaplan D, Barker J. Psoriasis. N Engl J Med. 2009;361:496-509.
- Pinheiro RR, Lencastre A. Henoch-Schönlein purpura during anti-TNFα therapy: a fortuitous event or an indication to stop therapy? Eur J Dermatol. 2017;27:304-305.
- Hello CL, Cohen P, Bousser MG, et al. Suspected hepatitis B vaccination related vasculitis. J Rheumatol. 1999;26:191-194.
- Wolverton SE. Dapsone. In: Wolverton SE, Wu JJ, eds. Comprehensive Dermatologic Drug Therapy. 4th ed. Elsevier, Inc; 2021:222-231.
- Reverte M, Etienne M, Fouchard M, et al. Occurrence of Henoch-Schönlein purpura in a patient treated with secukinumab. J Eur Acad Dermatol Venereol. 2019;33:E455-E457.
- Chelli C, Loget J, Vanhaecke C, et al. Cutaneous vasculitis with gut involvement during secukinumab treatment for psoriatic arthritis. Acta Derm Venereol. 2020;100:adv00077.
- da Silva Cendon Duran C, Santiago MB. Cutaneous vasculitis during secukinumab treatment. Eur J Case Rep Intern Med. 2020;7:001815.
- Bostan E, Gulseren D, Yalici-Armagan B, et al. Vasculitis during certolizumab pegol and secukinumab treatment: report of two cases. Dermatol Ther. 2021;34:E15007.
- Perkovic D, Simac P, Katic J. IgA vasculitis during secukinumab therapy. Clin Rheumatol. 2021;40:2071-2073.
- Villani A, DE Fata Salvatores G, Nappa P, et al. Cutaneous leucocytoclastic vasculitis during secukinumab treatment. Ital J Dermatol Venerol. 2021;156(suppl 1 to no. 6):9-10.
- Góis M, Messias A, Carvalho D, et al. MPO-ANCA-associated necrotizing glomerulonephritis in rheumatoid arthritis; a case report and review of literature. J Nephropathol. 2017;6:58-62.
- Jen HY, Chuang YH, Lin SC, et al. Increased serum interleukin-17 and peripheral Th17 cells in children with acute Henoch-Schönlein purpura. Pediatr Allergy Immunol. 2011;22:862-868.
- Hetland LE, Susrud KS, Lindahl KH, et al. Henoch-Schönlein purpura: a literature review. Acta Derm Venereol 2017;97:1160-1166.
- Weedon D. The vasculopathic reaction pattern. In: Houston M, Davie B, eds. Weedon’s Skin Pathology. 3rd ed. Elsevier Limited; 2010:207-211.
- Puig L. Paradoxical reactions: anti-TNFα ants, ustekinumab, secukinumab, ixekizumab, and others. Curr Probl Dermatol. 2018;53:49-63.
- Nestle F, Kaplan D, Barker J. Psoriasis. N Engl J Med. 2009;361:496-509.
- Pinheiro RR, Lencastre A. Henoch-Schönlein purpura during anti-TNFα therapy: a fortuitous event or an indication to stop therapy? Eur J Dermatol. 2017;27:304-305.
- Hello CL, Cohen P, Bousser MG, et al. Suspected hepatitis B vaccination related vasculitis. J Rheumatol. 1999;26:191-194.
- Wolverton SE. Dapsone. In: Wolverton SE, Wu JJ, eds. Comprehensive Dermatologic Drug Therapy. 4th ed. Elsevier, Inc; 2021:222-231.
Practice Points
- Biologic medications including adalimumab and more rarely secukinumab may be associated with leukocytoclastic vasculitis; a smaller subset of patients may experience IgA vasculitis.
- The IL-23 blocker ustekinumab may represent an ideal therapeutic agent when secukinumabassociated vasculitis is suspected. Because IL-23 is the main driver and sustainer of TH17 cell differentiation, it may cease the main causative cytokine cascades “upstream.”
Remote assessment of atopic dermatitis is feasible with patient-provided images: Study
MONTREAL – , as well as the possibility of conducting remote clinical trials that would be less expensive and less burdensome for participants, according to investigators, who presented the study at the annual meeting of the International Society of Atopic Dermatitis.
Still, practical barriers need to be addressed, particularly the problem of image quality, noted study investigator Aviël Ragamin, MD, from the department of dermatology, Erasmus MC, University Medical Center, Rotterdam, the Netherlands.
“Good-quality images are crucial, [and] in our study, patients didn’t have any incentive to provide images because they had already received their medical consultation,” he explained. He suggested that this problem could be overcome by providing technical support for patients and compensation for trial participants.
The study included 87 children (median age, 7 years), who were assessed for AD severity at an academic outpatient clinic. The in-person visit included assessment with the Eczema Area and Severity Index (EASI) score, as well as the collection of whole-body clinical images. Parents were then asked to return home and to provide their own clinical images and self-administered EASI assessments of their child for comparison. Four raters were asked to rate all images twice and to compare in-clinic and self-administered EASI scores based on the images.
At the in-clinic visit, the median EASI score of the group was 8.8. The majority of patients had moderate (46.6%) or severe (14.8%) AD. Roughly 40% of the patients had darker skin (Fitzpatrick skin types IV–VI).
Using Spearman rank correlation of 1,534 in-clinic and 425 patient-provided images, the study found good inter- and intra-rater reliability for clinical image assessment and strong agreement between images and the in-clinic EASI scores. The top outliers in the assessment were individuals with either darker skin or significant postinflammatory hyperpigmentation, which are “the most difficult cases to rate, based on images,” Dr. Ragamin noted.
There was only moderate correlation between the in-clinic and self-administered EASI scores, with a significant number of patients either underestimating or overestimating their AD severity, he added.
Overall, the main problem with remote assessment seems to be the feasibility of patients providing images, said Dr. Ragamin. Only 36.8% of parents provided any images at all, and of these, 1 of 5 were deemed too blurry, leaving just 13 for final assessment, he explained.
“Pragmatically, it’s tricky,” said Aaron Drucker, MD, a dermatologist at Women’s College Hospital and associate professor at the University of Toronto, who was asked to comment on the study. “It takes long enough to do an EASI score in person, let alone looking through blurry pictures that take too long to load into your electronic medical record. We know it works, but when our hospital went virtual [during the COVID pandemic] ... most of my patients with chronic eczema weren’t even sending me pictures.”
Regarding the utility of remote, full-body photography in clinical practice, he said, “There’s too many feasibility hoops to jump through at this point. The most promise I see is for clinical trials, where it’s hard to get people to come in.”
Dr. Ragamin and Dr. Drucker have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
MONTREAL – , as well as the possibility of conducting remote clinical trials that would be less expensive and less burdensome for participants, according to investigators, who presented the study at the annual meeting of the International Society of Atopic Dermatitis.
Still, practical barriers need to be addressed, particularly the problem of image quality, noted study investigator Aviël Ragamin, MD, from the department of dermatology, Erasmus MC, University Medical Center, Rotterdam, the Netherlands.
“Good-quality images are crucial, [and] in our study, patients didn’t have any incentive to provide images because they had already received their medical consultation,” he explained. He suggested that this problem could be overcome by providing technical support for patients and compensation for trial participants.
The study included 87 children (median age, 7 years), who were assessed for AD severity at an academic outpatient clinic. The in-person visit included assessment with the Eczema Area and Severity Index (EASI) score, as well as the collection of whole-body clinical images. Parents were then asked to return home and to provide their own clinical images and self-administered EASI assessments of their child for comparison. Four raters were asked to rate all images twice and to compare in-clinic and self-administered EASI scores based on the images.
At the in-clinic visit, the median EASI score of the group was 8.8. The majority of patients had moderate (46.6%) or severe (14.8%) AD. Roughly 40% of the patients had darker skin (Fitzpatrick skin types IV–VI).
Using Spearman rank correlation of 1,534 in-clinic and 425 patient-provided images, the study found good inter- and intra-rater reliability for clinical image assessment and strong agreement between images and the in-clinic EASI scores. The top outliers in the assessment were individuals with either darker skin or significant postinflammatory hyperpigmentation, which are “the most difficult cases to rate, based on images,” Dr. Ragamin noted.
There was only moderate correlation between the in-clinic and self-administered EASI scores, with a significant number of patients either underestimating or overestimating their AD severity, he added.
Overall, the main problem with remote assessment seems to be the feasibility of patients providing images, said Dr. Ragamin. Only 36.8% of parents provided any images at all, and of these, 1 of 5 were deemed too blurry, leaving just 13 for final assessment, he explained.
“Pragmatically, it’s tricky,” said Aaron Drucker, MD, a dermatologist at Women’s College Hospital and associate professor at the University of Toronto, who was asked to comment on the study. “It takes long enough to do an EASI score in person, let alone looking through blurry pictures that take too long to load into your electronic medical record. We know it works, but when our hospital went virtual [during the COVID pandemic] ... most of my patients with chronic eczema weren’t even sending me pictures.”
Regarding the utility of remote, full-body photography in clinical practice, he said, “There’s too many feasibility hoops to jump through at this point. The most promise I see is for clinical trials, where it’s hard to get people to come in.”
Dr. Ragamin and Dr. Drucker have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
MONTREAL – , as well as the possibility of conducting remote clinical trials that would be less expensive and less burdensome for participants, according to investigators, who presented the study at the annual meeting of the International Society of Atopic Dermatitis.
Still, practical barriers need to be addressed, particularly the problem of image quality, noted study investigator Aviël Ragamin, MD, from the department of dermatology, Erasmus MC, University Medical Center, Rotterdam, the Netherlands.
“Good-quality images are crucial, [and] in our study, patients didn’t have any incentive to provide images because they had already received their medical consultation,” he explained. He suggested that this problem could be overcome by providing technical support for patients and compensation for trial participants.
The study included 87 children (median age, 7 years), who were assessed for AD severity at an academic outpatient clinic. The in-person visit included assessment with the Eczema Area and Severity Index (EASI) score, as well as the collection of whole-body clinical images. Parents were then asked to return home and to provide their own clinical images and self-administered EASI assessments of their child for comparison. Four raters were asked to rate all images twice and to compare in-clinic and self-administered EASI scores based on the images.
At the in-clinic visit, the median EASI score of the group was 8.8. The majority of patients had moderate (46.6%) or severe (14.8%) AD. Roughly 40% of the patients had darker skin (Fitzpatrick skin types IV–VI).
Using Spearman rank correlation of 1,534 in-clinic and 425 patient-provided images, the study found good inter- and intra-rater reliability for clinical image assessment and strong agreement between images and the in-clinic EASI scores. The top outliers in the assessment were individuals with either darker skin or significant postinflammatory hyperpigmentation, which are “the most difficult cases to rate, based on images,” Dr. Ragamin noted.
There was only moderate correlation between the in-clinic and self-administered EASI scores, with a significant number of patients either underestimating or overestimating their AD severity, he added.
Overall, the main problem with remote assessment seems to be the feasibility of patients providing images, said Dr. Ragamin. Only 36.8% of parents provided any images at all, and of these, 1 of 5 were deemed too blurry, leaving just 13 for final assessment, he explained.
“Pragmatically, it’s tricky,” said Aaron Drucker, MD, a dermatologist at Women’s College Hospital and associate professor at the University of Toronto, who was asked to comment on the study. “It takes long enough to do an EASI score in person, let alone looking through blurry pictures that take too long to load into your electronic medical record. We know it works, but when our hospital went virtual [during the COVID pandemic] ... most of my patients with chronic eczema weren’t even sending me pictures.”
Regarding the utility of remote, full-body photography in clinical practice, he said, “There’s too many feasibility hoops to jump through at this point. The most promise I see is for clinical trials, where it’s hard to get people to come in.”
Dr. Ragamin and Dr. Drucker have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.