User login
Bringing you the latest news, research and reviews, exclusive interviews, podcasts, quizzes, and more.
div[contains(@class, 'header__large-screen')]
div[contains(@class, 'read-next-article')]
div[contains(@class, 'nav-primary')]
nav[contains(@class, 'nav-primary')]
section[contains(@class, 'footer-nav-section-wrapper')]
footer[@id='footer']
div[contains(@class, 'main-prefix')]
section[contains(@class, 'nav-hidden')]
div[contains(@class, 'ce-card-content')]
nav[contains(@class, 'nav-ce-stack')]
Study finds systemic AD treatment relieves depressive symptoms along with skin symptoms
MONTREAL – presented at the annual meeting of the International Society of Atopic Dermatitis.
“Randomized, controlled, phase 3 studies have shown that systemic treatment of AD reduces depressive symptoms, but whether this holds true in real-world cohorts remains to be shown,” said study investigator Lina Ivert, MD, PhD, of the dermatology and venereology unit in the department of medicine at the Karolinska Institutet, Stockholm.
The study used data from SwedAD, a newly launched web-based Swedish national registry of patients with AD on systemic treatment between June 2017 and August 2021. Participants were followed at 6 and 12 months for the primary outcome of depressive symptoms using the Montgomery–Åsberg Depression Rating Scale–self-report (MADRS-S). Secondary outcomes included the Eczema Area and Severity Index (EASI) score, Patient-Oriented Eczema Measure (POEM), the Dermatology Life Quality Index (DLQI), and pruritus visual analog scale/numeric rating scale (VAS/NRS).
At baseline, 120 patients (median age, 39 years; 57.5% men) were started on dupilumab (n = 91), methotrexate (26), or cyclosporin (3). Although almost half had no depression at baseline, mild depression was present in 29.2%, with moderate and severe depression in 20% and 4.2%, respectively.
Among 59 patients with 6-month follow-up data (48 on dupilumab, 10 on methotrexate, 1 on cyclosporin), all nine depressive symptoms in MADRS-S improved significantly, with reduced sleep improving the most (from a median of 3 points to a median of 1 point). Similarly, overall MADRS-S scores improved (from a median of 14 points to a median of 5; P < .001), as did EASI scores (from a median of 20.5 to 2), POEM scores (from a median of 22 to 6), DLQI (from a median of 15 to 3), and pruritus scores (from a median of 7.1 to 1.8; all P < .001).
The analysis also found a strong correlation between the MADRS-S score and all of the secondary outcomes (P < .001 for all). All these improvements remained significant among the 36 patients with 12-month follow-up data.
“The median MADRS-S reduction also remained when we excluded eight patients who were on antidepressants during the study period, so these results cannot be explained by psychiatric medication,” noted Dr. Ivert, adding that three patients with severe suicide ideation at baseline improved their MADRS-S suicide item to less than 2 points. “So, this study taught us to look at the suicide item score and not only the total MADRS-S score,” she commented.
Comparing patients treated with dupilumab with those treated with methotrexate, the analysis showed that though baseline median MADRS-S scores did not differ significantly between them, there was a significant 6-month reduction in the dupilumab group but not in the methotrexate group.
Asked to comment on the findings, moderator Marissa Joseph, MD, a pediatric dermatologist at the University of Toronto, said that “the mental health effects of inflammatory skin conditions like atopic dermatitis are well known, but whether or not they are well explored in the patient-physician interaction is a whole other scenario.” There are time constraints, she said, adding, “it sometimes takes some deep-diving ... but exploring those types of symptoms is something we need to do more of, and the severity of the disease and reasons for treatment are not just what you can see.”
Dr. Joseph pointed out that taking the deep dive also involves being prepared for what comes up. “Once you’ve established there’s a mental health issue, what do you do then?” she said. “If you are a dermatologist, is that in your wheelhouse to address? There’s the education and connection piece for the physician, creating networks where – if you identify a patient who has an issue – who is a person I can send them to? We have these types of connections with infectious disease or with ophthalmologists if there are ocular symptoms, but mental health is one area where there may not be as much support for dermatologists.”
She noted that though all doctors learn how to screen for depression, “there’s the formulaic, yes/no answers, and then there’s the nuanced history-taking, creating a safe space, where the patient is going to answer you fulsomely ... and feel heard. Many of us know how to do that. The question is time.”
Dr. Ivert had no disclosures connected to this study. Dr. Joseph had no disclosures.
A version of this article first appeared on Medscape.com.
MONTREAL – presented at the annual meeting of the International Society of Atopic Dermatitis.
“Randomized, controlled, phase 3 studies have shown that systemic treatment of AD reduces depressive symptoms, but whether this holds true in real-world cohorts remains to be shown,” said study investigator Lina Ivert, MD, PhD, of the dermatology and venereology unit in the department of medicine at the Karolinska Institutet, Stockholm.
The study used data from SwedAD, a newly launched web-based Swedish national registry of patients with AD on systemic treatment between June 2017 and August 2021. Participants were followed at 6 and 12 months for the primary outcome of depressive symptoms using the Montgomery–Åsberg Depression Rating Scale–self-report (MADRS-S). Secondary outcomes included the Eczema Area and Severity Index (EASI) score, Patient-Oriented Eczema Measure (POEM), the Dermatology Life Quality Index (DLQI), and pruritus visual analog scale/numeric rating scale (VAS/NRS).
At baseline, 120 patients (median age, 39 years; 57.5% men) were started on dupilumab (n = 91), methotrexate (26), or cyclosporin (3). Although almost half had no depression at baseline, mild depression was present in 29.2%, with moderate and severe depression in 20% and 4.2%, respectively.
Among 59 patients with 6-month follow-up data (48 on dupilumab, 10 on methotrexate, 1 on cyclosporin), all nine depressive symptoms in MADRS-S improved significantly, with reduced sleep improving the most (from a median of 3 points to a median of 1 point). Similarly, overall MADRS-S scores improved (from a median of 14 points to a median of 5; P < .001), as did EASI scores (from a median of 20.5 to 2), POEM scores (from a median of 22 to 6), DLQI (from a median of 15 to 3), and pruritus scores (from a median of 7.1 to 1.8; all P < .001).
The analysis also found a strong correlation between the MADRS-S score and all of the secondary outcomes (P < .001 for all). All these improvements remained significant among the 36 patients with 12-month follow-up data.
“The median MADRS-S reduction also remained when we excluded eight patients who were on antidepressants during the study period, so these results cannot be explained by psychiatric medication,” noted Dr. Ivert, adding that three patients with severe suicide ideation at baseline improved their MADRS-S suicide item to less than 2 points. “So, this study taught us to look at the suicide item score and not only the total MADRS-S score,” she commented.
Comparing patients treated with dupilumab with those treated with methotrexate, the analysis showed that though baseline median MADRS-S scores did not differ significantly between them, there was a significant 6-month reduction in the dupilumab group but not in the methotrexate group.
Asked to comment on the findings, moderator Marissa Joseph, MD, a pediatric dermatologist at the University of Toronto, said that “the mental health effects of inflammatory skin conditions like atopic dermatitis are well known, but whether or not they are well explored in the patient-physician interaction is a whole other scenario.” There are time constraints, she said, adding, “it sometimes takes some deep-diving ... but exploring those types of symptoms is something we need to do more of, and the severity of the disease and reasons for treatment are not just what you can see.”
Dr. Joseph pointed out that taking the deep dive also involves being prepared for what comes up. “Once you’ve established there’s a mental health issue, what do you do then?” she said. “If you are a dermatologist, is that in your wheelhouse to address? There’s the education and connection piece for the physician, creating networks where – if you identify a patient who has an issue – who is a person I can send them to? We have these types of connections with infectious disease or with ophthalmologists if there are ocular symptoms, but mental health is one area where there may not be as much support for dermatologists.”
She noted that though all doctors learn how to screen for depression, “there’s the formulaic, yes/no answers, and then there’s the nuanced history-taking, creating a safe space, where the patient is going to answer you fulsomely ... and feel heard. Many of us know how to do that. The question is time.”
Dr. Ivert had no disclosures connected to this study. Dr. Joseph had no disclosures.
A version of this article first appeared on Medscape.com.
MONTREAL – presented at the annual meeting of the International Society of Atopic Dermatitis.
“Randomized, controlled, phase 3 studies have shown that systemic treatment of AD reduces depressive symptoms, but whether this holds true in real-world cohorts remains to be shown,” said study investigator Lina Ivert, MD, PhD, of the dermatology and venereology unit in the department of medicine at the Karolinska Institutet, Stockholm.
The study used data from SwedAD, a newly launched web-based Swedish national registry of patients with AD on systemic treatment between June 2017 and August 2021. Participants were followed at 6 and 12 months for the primary outcome of depressive symptoms using the Montgomery–Åsberg Depression Rating Scale–self-report (MADRS-S). Secondary outcomes included the Eczema Area and Severity Index (EASI) score, Patient-Oriented Eczema Measure (POEM), the Dermatology Life Quality Index (DLQI), and pruritus visual analog scale/numeric rating scale (VAS/NRS).
At baseline, 120 patients (median age, 39 years; 57.5% men) were started on dupilumab (n = 91), methotrexate (26), or cyclosporin (3). Although almost half had no depression at baseline, mild depression was present in 29.2%, with moderate and severe depression in 20% and 4.2%, respectively.
Among 59 patients with 6-month follow-up data (48 on dupilumab, 10 on methotrexate, 1 on cyclosporin), all nine depressive symptoms in MADRS-S improved significantly, with reduced sleep improving the most (from a median of 3 points to a median of 1 point). Similarly, overall MADRS-S scores improved (from a median of 14 points to a median of 5; P < .001), as did EASI scores (from a median of 20.5 to 2), POEM scores (from a median of 22 to 6), DLQI (from a median of 15 to 3), and pruritus scores (from a median of 7.1 to 1.8; all P < .001).
The analysis also found a strong correlation between the MADRS-S score and all of the secondary outcomes (P < .001 for all). All these improvements remained significant among the 36 patients with 12-month follow-up data.
“The median MADRS-S reduction also remained when we excluded eight patients who were on antidepressants during the study period, so these results cannot be explained by psychiatric medication,” noted Dr. Ivert, adding that three patients with severe suicide ideation at baseline improved their MADRS-S suicide item to less than 2 points. “So, this study taught us to look at the suicide item score and not only the total MADRS-S score,” she commented.
Comparing patients treated with dupilumab with those treated with methotrexate, the analysis showed that though baseline median MADRS-S scores did not differ significantly between them, there was a significant 6-month reduction in the dupilumab group but not in the methotrexate group.
Asked to comment on the findings, moderator Marissa Joseph, MD, a pediatric dermatologist at the University of Toronto, said that “the mental health effects of inflammatory skin conditions like atopic dermatitis are well known, but whether or not they are well explored in the patient-physician interaction is a whole other scenario.” There are time constraints, she said, adding, “it sometimes takes some deep-diving ... but exploring those types of symptoms is something we need to do more of, and the severity of the disease and reasons for treatment are not just what you can see.”
Dr. Joseph pointed out that taking the deep dive also involves being prepared for what comes up. “Once you’ve established there’s a mental health issue, what do you do then?” she said. “If you are a dermatologist, is that in your wheelhouse to address? There’s the education and connection piece for the physician, creating networks where – if you identify a patient who has an issue – who is a person I can send them to? We have these types of connections with infectious disease or with ophthalmologists if there are ocular symptoms, but mental health is one area where there may not be as much support for dermatologists.”
She noted that though all doctors learn how to screen for depression, “there’s the formulaic, yes/no answers, and then there’s the nuanced history-taking, creating a safe space, where the patient is going to answer you fulsomely ... and feel heard. Many of us know how to do that. The question is time.”
Dr. Ivert had no disclosures connected to this study. Dr. Joseph had no disclosures.
A version of this article first appeared on Medscape.com.
FROM ISAD 2022
Hair straighteners’ risk too small for docs to advise against their use
Clarissa Ghazi gets lye relaxers, which contain the chemical sodium hydroxide, applied to her hair two to three times a year.
A recent study that made headlines over a potential link between hair straighteners and uterine cancer is not going to make her stop.
“This study is not enough to cause me to say I’ll stay away from this because [the researchers] don’t prove that using relaxers causes cancer,” Ms. Ghazi said.
Indeed, primary care doctors are unlikely to address the increased risk of uterine cancer in women who frequently use hair straighteners that the study reported.
In the recently published paper on this research, the authors said that they found an 80% higher adjusted risk of uterine cancer among women who had ever “straightened,” “relaxed,” or used “hair pressing products” in the 12 months before enrolling in their study.
This finding is “real, but small,” says internist Douglas S. Paauw, MD, professor of medicine at the University of Washington in Seattle.
Dr. Paauw is among several primary care doctors interviewed for this story who expressed little concern about the implications of this research for their patients.
“Since we have hundreds of things we are supposed to discuss at our 20-minute clinic visits, this would not make the cut,” Dr. Paauw said.
While it’s good to be able to answer questions a patient might ask about this new research, the study does not prove anything, he said.
Alan Nelson, MD, an internist-endocrinologist and former special adviser to the CEO of the American College of Physicians, said while the study is well done, the number of actual cases of uterine cancer found was small.
One of the reasons he would not recommend discussing the study with patients is that the brands of hair products used to straighten hair in the study were not identified.
Alexandra White, PhD, lead author of the study, said participants were simply asked, “In the past 12 months, how frequently have you or someone else straightened or relaxed your hair, or used hair pressing products?”
The terms “straightened,” “relaxed,” and “hair pressing products” were not defined, and “some women may have interpreted the term ‘pressing products’ to mean nonchemical products” such as flat irons, Dr. White, head of the National Institute of Environmental Health Sciences’ Environment and Cancer Epidemiology group, said in an email.
Dermatologist Crystal Aguh, MD, associate professor of dermatology at Johns Hopkins University, Baltimore, tweeted the following advice in light of the new findings: “The overall risk of uterine cancer is quite low so it’s important to remember that. For now, if you want to change your routine, there’s no downside to decreasing your frequency of hair straightening to every 12 weeks or more, as that may lessen your risk.”
She also noted that “styles like relaxer, silk pressing, and keratin treatments should only be done by a professional, as this will decrease the likelihood of hair damage and scalp irritation.
“I also encourage women to look for hair products free of parabens and phthalates (which are generically listed as “fragrance”) on products to minimize exposure to hormone disrupting chemicals.”
Not ready to go curly
Ms. Ghazi said she decided to stop using keratin straighteners years ago after she learned they are made with several added ingredients. That includes the chemical formaldehyde, a known carcinogen, according to the American Cancer Society.
“People have been relaxing their hair for a very long time, and I feel more comfortable using [a relaxer] to straighten my hair than any of the others out there,” Ms. Ghazi said.
Janaki Ram, who has had her hair chemically straightened several times, said the findings have not made her worried that straightening will cause her to get uterine cancer specifically, but that they are a reminder that the chemicals in these products could harm her in some other way.
She said the new study findings, her knowledge of the damage straightening causes to hair, and the lengthy amount of time receiving a keratin treatment takes will lead her to reduce the frequency with which she gets her hair straightened.
“Going forward, I will have this done once a year instead of twice a year,” she said.
Dr. White, the author of the paper, said in an interview that the takeaway for consumers is that women who reported frequent use of hair straighteners/relaxers and pressing products were more than twice as likely to go on to develop uterine cancer compared to women who reported no use of these products in the previous year.
“However, uterine cancer is relatively rare, so these increases in risks are small,” she said. “Less frequent use of these products was not as strongly associated with risk, suggesting that decreasing use may be an option to reduce harmful exposure. Black women were the most frequent users of these products and therefore these findings are more relevant for Black women.”
In a statement, Dr. White noted, “We estimated that 1.64% of women who never used hair straighteners would go on to develop uterine cancer by the age of 70; but for frequent users, that risk goes up to 4.05%.”
The findings were based on the Sister Study, which enrolled women living in the United States, including Puerto Rico, between 2003 and 2009. Participants needed to have at least one sister who had been diagnosed with breast cancer, been breast cancer-free themselves, and aged 35-74 years. Women who reported a diagnosis of uterine cancer before enrollment, had an uncertain uterine cancer history, or had a hysterectomy were excluded from the study.
The researchers examined hair product usage and uterine cancer incidence during an 11-year period among 33 ,947 women. The analysis controlled for variables such as age, race, and risk factors. At baseline, participants were asked to complete a questionnaire on hair products use in the previous 12 months.
“One of the original aims of the study was to better understand the environmental and genetic causes of breast cancer, but we are also interested in studying ovarian cancer, uterine cancer, and many other cancers and chronic diseases,” Dr. White said.
A version of this article first appeared on WebMD.com.
Clarissa Ghazi gets lye relaxers, which contain the chemical sodium hydroxide, applied to her hair two to three times a year.
A recent study that made headlines over a potential link between hair straighteners and uterine cancer is not going to make her stop.
“This study is not enough to cause me to say I’ll stay away from this because [the researchers] don’t prove that using relaxers causes cancer,” Ms. Ghazi said.
Indeed, primary care doctors are unlikely to address the increased risk of uterine cancer in women who frequently use hair straighteners that the study reported.
In the recently published paper on this research, the authors said that they found an 80% higher adjusted risk of uterine cancer among women who had ever “straightened,” “relaxed,” or used “hair pressing products” in the 12 months before enrolling in their study.
This finding is “real, but small,” says internist Douglas S. Paauw, MD, professor of medicine at the University of Washington in Seattle.
Dr. Paauw is among several primary care doctors interviewed for this story who expressed little concern about the implications of this research for their patients.
“Since we have hundreds of things we are supposed to discuss at our 20-minute clinic visits, this would not make the cut,” Dr. Paauw said.
While it’s good to be able to answer questions a patient might ask about this new research, the study does not prove anything, he said.
Alan Nelson, MD, an internist-endocrinologist and former special adviser to the CEO of the American College of Physicians, said while the study is well done, the number of actual cases of uterine cancer found was small.
One of the reasons he would not recommend discussing the study with patients is that the brands of hair products used to straighten hair in the study were not identified.
Alexandra White, PhD, lead author of the study, said participants were simply asked, “In the past 12 months, how frequently have you or someone else straightened or relaxed your hair, or used hair pressing products?”
The terms “straightened,” “relaxed,” and “hair pressing products” were not defined, and “some women may have interpreted the term ‘pressing products’ to mean nonchemical products” such as flat irons, Dr. White, head of the National Institute of Environmental Health Sciences’ Environment and Cancer Epidemiology group, said in an email.
Dermatologist Crystal Aguh, MD, associate professor of dermatology at Johns Hopkins University, Baltimore, tweeted the following advice in light of the new findings: “The overall risk of uterine cancer is quite low so it’s important to remember that. For now, if you want to change your routine, there’s no downside to decreasing your frequency of hair straightening to every 12 weeks or more, as that may lessen your risk.”
She also noted that “styles like relaxer, silk pressing, and keratin treatments should only be done by a professional, as this will decrease the likelihood of hair damage and scalp irritation.
“I also encourage women to look for hair products free of parabens and phthalates (which are generically listed as “fragrance”) on products to minimize exposure to hormone disrupting chemicals.”
Not ready to go curly
Ms. Ghazi said she decided to stop using keratin straighteners years ago after she learned they are made with several added ingredients. That includes the chemical formaldehyde, a known carcinogen, according to the American Cancer Society.
“People have been relaxing their hair for a very long time, and I feel more comfortable using [a relaxer] to straighten my hair than any of the others out there,” Ms. Ghazi said.
Janaki Ram, who has had her hair chemically straightened several times, said the findings have not made her worried that straightening will cause her to get uterine cancer specifically, but that they are a reminder that the chemicals in these products could harm her in some other way.
She said the new study findings, her knowledge of the damage straightening causes to hair, and the lengthy amount of time receiving a keratin treatment takes will lead her to reduce the frequency with which she gets her hair straightened.
“Going forward, I will have this done once a year instead of twice a year,” she said.
Dr. White, the author of the paper, said in an interview that the takeaway for consumers is that women who reported frequent use of hair straighteners/relaxers and pressing products were more than twice as likely to go on to develop uterine cancer compared to women who reported no use of these products in the previous year.
“However, uterine cancer is relatively rare, so these increases in risks are small,” she said. “Less frequent use of these products was not as strongly associated with risk, suggesting that decreasing use may be an option to reduce harmful exposure. Black women were the most frequent users of these products and therefore these findings are more relevant for Black women.”
In a statement, Dr. White noted, “We estimated that 1.64% of women who never used hair straighteners would go on to develop uterine cancer by the age of 70; but for frequent users, that risk goes up to 4.05%.”
The findings were based on the Sister Study, which enrolled women living in the United States, including Puerto Rico, between 2003 and 2009. Participants needed to have at least one sister who had been diagnosed with breast cancer, been breast cancer-free themselves, and aged 35-74 years. Women who reported a diagnosis of uterine cancer before enrollment, had an uncertain uterine cancer history, or had a hysterectomy were excluded from the study.
The researchers examined hair product usage and uterine cancer incidence during an 11-year period among 33 ,947 women. The analysis controlled for variables such as age, race, and risk factors. At baseline, participants were asked to complete a questionnaire on hair products use in the previous 12 months.
“One of the original aims of the study was to better understand the environmental and genetic causes of breast cancer, but we are also interested in studying ovarian cancer, uterine cancer, and many other cancers and chronic diseases,” Dr. White said.
A version of this article first appeared on WebMD.com.
Clarissa Ghazi gets lye relaxers, which contain the chemical sodium hydroxide, applied to her hair two to three times a year.
A recent study that made headlines over a potential link between hair straighteners and uterine cancer is not going to make her stop.
“This study is not enough to cause me to say I’ll stay away from this because [the researchers] don’t prove that using relaxers causes cancer,” Ms. Ghazi said.
Indeed, primary care doctors are unlikely to address the increased risk of uterine cancer in women who frequently use hair straighteners that the study reported.
In the recently published paper on this research, the authors said that they found an 80% higher adjusted risk of uterine cancer among women who had ever “straightened,” “relaxed,” or used “hair pressing products” in the 12 months before enrolling in their study.
This finding is “real, but small,” says internist Douglas S. Paauw, MD, professor of medicine at the University of Washington in Seattle.
Dr. Paauw is among several primary care doctors interviewed for this story who expressed little concern about the implications of this research for their patients.
“Since we have hundreds of things we are supposed to discuss at our 20-minute clinic visits, this would not make the cut,” Dr. Paauw said.
While it’s good to be able to answer questions a patient might ask about this new research, the study does not prove anything, he said.
Alan Nelson, MD, an internist-endocrinologist and former special adviser to the CEO of the American College of Physicians, said while the study is well done, the number of actual cases of uterine cancer found was small.
One of the reasons he would not recommend discussing the study with patients is that the brands of hair products used to straighten hair in the study were not identified.
Alexandra White, PhD, lead author of the study, said participants were simply asked, “In the past 12 months, how frequently have you or someone else straightened or relaxed your hair, or used hair pressing products?”
The terms “straightened,” “relaxed,” and “hair pressing products” were not defined, and “some women may have interpreted the term ‘pressing products’ to mean nonchemical products” such as flat irons, Dr. White, head of the National Institute of Environmental Health Sciences’ Environment and Cancer Epidemiology group, said in an email.
Dermatologist Crystal Aguh, MD, associate professor of dermatology at Johns Hopkins University, Baltimore, tweeted the following advice in light of the new findings: “The overall risk of uterine cancer is quite low so it’s important to remember that. For now, if you want to change your routine, there’s no downside to decreasing your frequency of hair straightening to every 12 weeks or more, as that may lessen your risk.”
She also noted that “styles like relaxer, silk pressing, and keratin treatments should only be done by a professional, as this will decrease the likelihood of hair damage and scalp irritation.
“I also encourage women to look for hair products free of parabens and phthalates (which are generically listed as “fragrance”) on products to minimize exposure to hormone disrupting chemicals.”
Not ready to go curly
Ms. Ghazi said she decided to stop using keratin straighteners years ago after she learned they are made with several added ingredients. That includes the chemical formaldehyde, a known carcinogen, according to the American Cancer Society.
“People have been relaxing their hair for a very long time, and I feel more comfortable using [a relaxer] to straighten my hair than any of the others out there,” Ms. Ghazi said.
Janaki Ram, who has had her hair chemically straightened several times, said the findings have not made her worried that straightening will cause her to get uterine cancer specifically, but that they are a reminder that the chemicals in these products could harm her in some other way.
She said the new study findings, her knowledge of the damage straightening causes to hair, and the lengthy amount of time receiving a keratin treatment takes will lead her to reduce the frequency with which she gets her hair straightened.
“Going forward, I will have this done once a year instead of twice a year,” she said.
Dr. White, the author of the paper, said in an interview that the takeaway for consumers is that women who reported frequent use of hair straighteners/relaxers and pressing products were more than twice as likely to go on to develop uterine cancer compared to women who reported no use of these products in the previous year.
“However, uterine cancer is relatively rare, so these increases in risks are small,” she said. “Less frequent use of these products was not as strongly associated with risk, suggesting that decreasing use may be an option to reduce harmful exposure. Black women were the most frequent users of these products and therefore these findings are more relevant for Black women.”
In a statement, Dr. White noted, “We estimated that 1.64% of women who never used hair straighteners would go on to develop uterine cancer by the age of 70; but for frequent users, that risk goes up to 4.05%.”
The findings were based on the Sister Study, which enrolled women living in the United States, including Puerto Rico, between 2003 and 2009. Participants needed to have at least one sister who had been diagnosed with breast cancer, been breast cancer-free themselves, and aged 35-74 years. Women who reported a diagnosis of uterine cancer before enrollment, had an uncertain uterine cancer history, or had a hysterectomy were excluded from the study.
The researchers examined hair product usage and uterine cancer incidence during an 11-year period among 33 ,947 women. The analysis controlled for variables such as age, race, and risk factors. At baseline, participants were asked to complete a questionnaire on hair products use in the previous 12 months.
“One of the original aims of the study was to better understand the environmental and genetic causes of breast cancer, but we are also interested in studying ovarian cancer, uterine cancer, and many other cancers and chronic diseases,” Dr. White said.
A version of this article first appeared on WebMD.com.
JAK inhibitors show no excess cardiovascular safety signal in French nationwide cohort
Janus kinase inhibitors tofacitinib (Xeljanz) and baricitinib (Olumiant) may pose no greater risk than does adalimumab (Humira and biosimilars) for major adverse cardiovascular events (MACEs) or venous thromboembolism (VTE) on the basis of a nationwide cohort study.
The French data, which included almost 16,000 patients with rheumatoid arthritis, revealed similar safety across subgroups, including older patients with at least one preexisting cardiovascular risk factor, reported lead author Léa Hoisnard, MD, of Henri Mondor Hospital, Paris, and colleagues.
These findings arrive 1 year after the U.S. Food and Drug Administration imposed class-wide boxed warnings on three Janus kinase (JAK) inhibitors, citing increased risks for both cancer and serious cardiac events detected by the open-label, randomized ORAL Surveillance postmarketing trial, which compared tofacitinib against adalimumab and etanercept.
More recently, the observational STAR-RA study, relying upon private insurance and Medicare claims in the United States, found no significant increase in cardiovascular events among patients taking tofacitinib, adding some uncertainty to the conversation.
“In this context, observational studies of unselected populations outside of North America are still needed to assess other JAK inhibitor agents,” Dr. Hoisnard and colleagues write in Annals of the Rheumatic Diseases.
Their retrospective study included 8,481 patients who received baricitinib or tofacitinib, and 7,354 patients who received adalimumab. Almost all patients in the tofacitinib group received 5 mg twice daily instead of 10 mg twice daily (99.4% vs. 0.6%), so cardiovascular safety was assessed only for the 5-mg dose. Baricitinib was prescribed at 4-mg and 2-mg doses (79.5% vs. 20.5%), allowing inclusion of both dose levels. The investigators accounted for a range of covariates, including concurrent therapy, comorbidities, and other patient characteristics.
Median follow-up durations were 440 days in the JAK inhibitor group and 344 days in the adalimumab group. The JAK inhibitor group had numerically more MACEs than did the adalimumab group, but the difference in risk was not statistically significant (54 vs. 35 MACEs; weighted hazard ratio, 1.0; 95% confidence interval, 0.7-1.5; P = .99). Similarly, more patients taking JAK inhibitors had VTEs, but relative risk was, again, not significant (75 vs. 32 VTEs; HRw, 1.1; 95% CI, 0.7-1.6; P = .63).
These findings were consistent for all subgroups, including patients aged 50 years or older and patients aged 65 years or older, although the investigators noted that statistical power was lacking for subgroup analyses.
Findings from Echo ORAL Surveillance
“I think the baricitinib data are important,” Kevin Winthrop, MD, MPH, professor of infectious diseases and epidemiology at Oregon Health & Science University, Portland, told this news organization. “There’s no difference between 2 mg and 4 mg [dose levels] in this analysis. And there doesn’t really seem to be a difference between baricitinib and tofacitinib. Most of the results are pretty consistent with ORAL Surveillance, which was a randomized, controlled trial.”
Dr. Winthrop, who has been active in JAK inhibitor clinical trials, recently coauthored an article in Nature Reviews Rheumatology encouraging clinicians to remember that the cardiovascular risks of JAK inhibitors are relative to adalimumab, and safety should be framed within the context of risk-to-benefit ratios.
He and his coauthor also called into question the FDA’s “better to be safe than sorry” approach, which resulted in boxed warnings across all JAK inhibitors, despite differences in target specificity.
“There are pros and cons of taking that approach,” Dr. Winthrop said in an interview. “The FDA might ultimately be right. Certainly, these drugs appear similar for some types of events, like herpes zoster, for example. But whether they’re similar with regard to malignancy or cardiovascular events, I don’t think we know.”
Dr. Winthrop noted that deucravacitinib was recently approved for psoriasis sans boxed warning, suggesting inconsistency in the FDA’s approach. The agent headlines as a “TYK2 inhibitor,” but TYK2 is a member of the JAK family.
“I don’t know why the FDA decided to treat them differently,” Dr. Winthrop said.
Boxed warnings encourage caution, lock treatment sequence
Michael Thakor, MD, of Arthritis & Rheumatology Clinic of Northern Colorado, Fort Collins, supports the boxed warnings because they encourage caution and transparency.
“It forces you to have that discussion with your patient, which may take some time, but it’s actually a very good thing,” Dr. Thakor said in an interview. “Some patients will say, ‘Oh my gosh, I don’t want to take that drug.’ But most patients, considering the level of risk that you’re talking about, are actually okay going ahead with the medication.”
If these risks aren’t discussed, he noted, patient trust may falter.
“They’re going to go online, and they’re going to be reading about it,” Dr. Thakor said. “And then they tend to get more spooked. They also may question your advice from then on, if you’re not telling them the possible risk.”
Reflecting on the present study, Dr. Thakor said that the findings initially appeared reassuring, but he became concerned about the lack of power and how adverse events trended higher in the JAK inhibitor group, particularly for VTEs, most of which occurred with baricitinib. This latter finding is challenging to interpret, however, because the 4-mg dose is not used in the United States, he added.
Dr. Thakor described how JAK inhibitors once seemed poised to assume a frontline role in RA until the boxed warnings came out. These safety concerns don’t take JAK inhibitors off the table, he said, but they do keep the class further down the treatment sequence, and the present data don’t alter this picture in daily practice.
“If I had a patient who was over the age of 50 with at least one cardiovascular risk factor, I might have a little bit of concern, but if they need their RA treated, I would definitely discuss the possibility of using a JAK inhibitor,” Dr. Thakor said. “If the patient is comfortable with it, then I would feel comfortable going ahead.”
The investigators disclosed no outside funding or conflicts of interest. Dr. Winthrop disclosed relationships with AbbVie, AstraZeneca, Bristol-Myers Squibb, and others. Dr. Thakor disclosed no conflicts of interest.
A version of this article first appeared on Medscape.com.
Janus kinase inhibitors tofacitinib (Xeljanz) and baricitinib (Olumiant) may pose no greater risk than does adalimumab (Humira and biosimilars) for major adverse cardiovascular events (MACEs) or venous thromboembolism (VTE) on the basis of a nationwide cohort study.
The French data, which included almost 16,000 patients with rheumatoid arthritis, revealed similar safety across subgroups, including older patients with at least one preexisting cardiovascular risk factor, reported lead author Léa Hoisnard, MD, of Henri Mondor Hospital, Paris, and colleagues.
These findings arrive 1 year after the U.S. Food and Drug Administration imposed class-wide boxed warnings on three Janus kinase (JAK) inhibitors, citing increased risks for both cancer and serious cardiac events detected by the open-label, randomized ORAL Surveillance postmarketing trial, which compared tofacitinib against adalimumab and etanercept.
More recently, the observational STAR-RA study, relying upon private insurance and Medicare claims in the United States, found no significant increase in cardiovascular events among patients taking tofacitinib, adding some uncertainty to the conversation.
“In this context, observational studies of unselected populations outside of North America are still needed to assess other JAK inhibitor agents,” Dr. Hoisnard and colleagues write in Annals of the Rheumatic Diseases.
Their retrospective study included 8,481 patients who received baricitinib or tofacitinib, and 7,354 patients who received adalimumab. Almost all patients in the tofacitinib group received 5 mg twice daily instead of 10 mg twice daily (99.4% vs. 0.6%), so cardiovascular safety was assessed only for the 5-mg dose. Baricitinib was prescribed at 4-mg and 2-mg doses (79.5% vs. 20.5%), allowing inclusion of both dose levels. The investigators accounted for a range of covariates, including concurrent therapy, comorbidities, and other patient characteristics.
Median follow-up durations were 440 days in the JAK inhibitor group and 344 days in the adalimumab group. The JAK inhibitor group had numerically more MACEs than did the adalimumab group, but the difference in risk was not statistically significant (54 vs. 35 MACEs; weighted hazard ratio, 1.0; 95% confidence interval, 0.7-1.5; P = .99). Similarly, more patients taking JAK inhibitors had VTEs, but relative risk was, again, not significant (75 vs. 32 VTEs; HRw, 1.1; 95% CI, 0.7-1.6; P = .63).
These findings were consistent for all subgroups, including patients aged 50 years or older and patients aged 65 years or older, although the investigators noted that statistical power was lacking for subgroup analyses.
Findings from Echo ORAL Surveillance
“I think the baricitinib data are important,” Kevin Winthrop, MD, MPH, professor of infectious diseases and epidemiology at Oregon Health & Science University, Portland, told this news organization. “There’s no difference between 2 mg and 4 mg [dose levels] in this analysis. And there doesn’t really seem to be a difference between baricitinib and tofacitinib. Most of the results are pretty consistent with ORAL Surveillance, which was a randomized, controlled trial.”
Dr. Winthrop, who has been active in JAK inhibitor clinical trials, recently coauthored an article in Nature Reviews Rheumatology encouraging clinicians to remember that the cardiovascular risks of JAK inhibitors are relative to adalimumab, and safety should be framed within the context of risk-to-benefit ratios.
He and his coauthor also called into question the FDA’s “better to be safe than sorry” approach, which resulted in boxed warnings across all JAK inhibitors, despite differences in target specificity.
“There are pros and cons of taking that approach,” Dr. Winthrop said in an interview. “The FDA might ultimately be right. Certainly, these drugs appear similar for some types of events, like herpes zoster, for example. But whether they’re similar with regard to malignancy or cardiovascular events, I don’t think we know.”
Dr. Winthrop noted that deucravacitinib was recently approved for psoriasis sans boxed warning, suggesting inconsistency in the FDA’s approach. The agent headlines as a “TYK2 inhibitor,” but TYK2 is a member of the JAK family.
“I don’t know why the FDA decided to treat them differently,” Dr. Winthrop said.
Boxed warnings encourage caution, lock treatment sequence
Michael Thakor, MD, of Arthritis & Rheumatology Clinic of Northern Colorado, Fort Collins, supports the boxed warnings because they encourage caution and transparency.
“It forces you to have that discussion with your patient, which may take some time, but it’s actually a very good thing,” Dr. Thakor said in an interview. “Some patients will say, ‘Oh my gosh, I don’t want to take that drug.’ But most patients, considering the level of risk that you’re talking about, are actually okay going ahead with the medication.”
If these risks aren’t discussed, he noted, patient trust may falter.
“They’re going to go online, and they’re going to be reading about it,” Dr. Thakor said. “And then they tend to get more spooked. They also may question your advice from then on, if you’re not telling them the possible risk.”
Reflecting on the present study, Dr. Thakor said that the findings initially appeared reassuring, but he became concerned about the lack of power and how adverse events trended higher in the JAK inhibitor group, particularly for VTEs, most of which occurred with baricitinib. This latter finding is challenging to interpret, however, because the 4-mg dose is not used in the United States, he added.
Dr. Thakor described how JAK inhibitors once seemed poised to assume a frontline role in RA until the boxed warnings came out. These safety concerns don’t take JAK inhibitors off the table, he said, but they do keep the class further down the treatment sequence, and the present data don’t alter this picture in daily practice.
“If I had a patient who was over the age of 50 with at least one cardiovascular risk factor, I might have a little bit of concern, but if they need their RA treated, I would definitely discuss the possibility of using a JAK inhibitor,” Dr. Thakor said. “If the patient is comfortable with it, then I would feel comfortable going ahead.”
The investigators disclosed no outside funding or conflicts of interest. Dr. Winthrop disclosed relationships with AbbVie, AstraZeneca, Bristol-Myers Squibb, and others. Dr. Thakor disclosed no conflicts of interest.
A version of this article first appeared on Medscape.com.
Janus kinase inhibitors tofacitinib (Xeljanz) and baricitinib (Olumiant) may pose no greater risk than does adalimumab (Humira and biosimilars) for major adverse cardiovascular events (MACEs) or venous thromboembolism (VTE) on the basis of a nationwide cohort study.
The French data, which included almost 16,000 patients with rheumatoid arthritis, revealed similar safety across subgroups, including older patients with at least one preexisting cardiovascular risk factor, reported lead author Léa Hoisnard, MD, of Henri Mondor Hospital, Paris, and colleagues.
These findings arrive 1 year after the U.S. Food and Drug Administration imposed class-wide boxed warnings on three Janus kinase (JAK) inhibitors, citing increased risks for both cancer and serious cardiac events detected by the open-label, randomized ORAL Surveillance postmarketing trial, which compared tofacitinib against adalimumab and etanercept.
More recently, the observational STAR-RA study, relying upon private insurance and Medicare claims in the United States, found no significant increase in cardiovascular events among patients taking tofacitinib, adding some uncertainty to the conversation.
“In this context, observational studies of unselected populations outside of North America are still needed to assess other JAK inhibitor agents,” Dr. Hoisnard and colleagues write in Annals of the Rheumatic Diseases.
Their retrospective study included 8,481 patients who received baricitinib or tofacitinib, and 7,354 patients who received adalimumab. Almost all patients in the tofacitinib group received 5 mg twice daily instead of 10 mg twice daily (99.4% vs. 0.6%), so cardiovascular safety was assessed only for the 5-mg dose. Baricitinib was prescribed at 4-mg and 2-mg doses (79.5% vs. 20.5%), allowing inclusion of both dose levels. The investigators accounted for a range of covariates, including concurrent therapy, comorbidities, and other patient characteristics.
Median follow-up durations were 440 days in the JAK inhibitor group and 344 days in the adalimumab group. The JAK inhibitor group had numerically more MACEs than did the adalimumab group, but the difference in risk was not statistically significant (54 vs. 35 MACEs; weighted hazard ratio, 1.0; 95% confidence interval, 0.7-1.5; P = .99). Similarly, more patients taking JAK inhibitors had VTEs, but relative risk was, again, not significant (75 vs. 32 VTEs; HRw, 1.1; 95% CI, 0.7-1.6; P = .63).
These findings were consistent for all subgroups, including patients aged 50 years or older and patients aged 65 years or older, although the investigators noted that statistical power was lacking for subgroup analyses.
Findings from Echo ORAL Surveillance
“I think the baricitinib data are important,” Kevin Winthrop, MD, MPH, professor of infectious diseases and epidemiology at Oregon Health & Science University, Portland, told this news organization. “There’s no difference between 2 mg and 4 mg [dose levels] in this analysis. And there doesn’t really seem to be a difference between baricitinib and tofacitinib. Most of the results are pretty consistent with ORAL Surveillance, which was a randomized, controlled trial.”
Dr. Winthrop, who has been active in JAK inhibitor clinical trials, recently coauthored an article in Nature Reviews Rheumatology encouraging clinicians to remember that the cardiovascular risks of JAK inhibitors are relative to adalimumab, and safety should be framed within the context of risk-to-benefit ratios.
He and his coauthor also called into question the FDA’s “better to be safe than sorry” approach, which resulted in boxed warnings across all JAK inhibitors, despite differences in target specificity.
“There are pros and cons of taking that approach,” Dr. Winthrop said in an interview. “The FDA might ultimately be right. Certainly, these drugs appear similar for some types of events, like herpes zoster, for example. But whether they’re similar with regard to malignancy or cardiovascular events, I don’t think we know.”
Dr. Winthrop noted that deucravacitinib was recently approved for psoriasis sans boxed warning, suggesting inconsistency in the FDA’s approach. The agent headlines as a “TYK2 inhibitor,” but TYK2 is a member of the JAK family.
“I don’t know why the FDA decided to treat them differently,” Dr. Winthrop said.
Boxed warnings encourage caution, lock treatment sequence
Michael Thakor, MD, of Arthritis & Rheumatology Clinic of Northern Colorado, Fort Collins, supports the boxed warnings because they encourage caution and transparency.
“It forces you to have that discussion with your patient, which may take some time, but it’s actually a very good thing,” Dr. Thakor said in an interview. “Some patients will say, ‘Oh my gosh, I don’t want to take that drug.’ But most patients, considering the level of risk that you’re talking about, are actually okay going ahead with the medication.”
If these risks aren’t discussed, he noted, patient trust may falter.
“They’re going to go online, and they’re going to be reading about it,” Dr. Thakor said. “And then they tend to get more spooked. They also may question your advice from then on, if you’re not telling them the possible risk.”
Reflecting on the present study, Dr. Thakor said that the findings initially appeared reassuring, but he became concerned about the lack of power and how adverse events trended higher in the JAK inhibitor group, particularly for VTEs, most of which occurred with baricitinib. This latter finding is challenging to interpret, however, because the 4-mg dose is not used in the United States, he added.
Dr. Thakor described how JAK inhibitors once seemed poised to assume a frontline role in RA until the boxed warnings came out. These safety concerns don’t take JAK inhibitors off the table, he said, but they do keep the class further down the treatment sequence, and the present data don’t alter this picture in daily practice.
“If I had a patient who was over the age of 50 with at least one cardiovascular risk factor, I might have a little bit of concern, but if they need their RA treated, I would definitely discuss the possibility of using a JAK inhibitor,” Dr. Thakor said. “If the patient is comfortable with it, then I would feel comfortable going ahead.”
The investigators disclosed no outside funding or conflicts of interest. Dr. Winthrop disclosed relationships with AbbVie, AstraZeneca, Bristol-Myers Squibb, and others. Dr. Thakor disclosed no conflicts of interest.
A version of this article first appeared on Medscape.com.
FROM ANNALS OF THE RHEUMATIC DISEASES
Reminder that COVID-19 and cancer can be a deadly combo
A new study underscores the importance of COVID-19 and regular COVID-19 testing among adults with a recent cancer diagnosis.
The Indiana statewide study, conducted at the beginning of the pandemic, found that
“This analysis provides additional empirical evidence on the magnitude of risk to patients with cancer whose immune systems are often weakened either by the disease or treatment,” the study team wrote.
The study was published online in JMIR Cancer.
Although evidence has consistently revealed similar findings, the risk of death among unvaccinated people with cancer and COVID-19 has not been nearly as high in previous studies, lead author Brian E. Dixon, PhD, MBA, with Indiana University Richard M. Fairbanks School of Public Health, Indianapolis, said in a statement. Previous studies from China, for instance, reported a two- to threefold greater risk of all-cause mortality among unvaccinated adults with cancer and COVID-19.
A potential reason for this discrepancy, Dr. Dixon noted, is that earlier studies were “generally smaller and made calculations based on data from a single cancer center or health system.”
Another reason is testing for COVID-19 early in the pandemic was limited to symptomatic individuals who may have had more severe infections, possibly leading to an overestimate of the association between SARS-CoV-2 infection, cancer, and all-cause mortality.
In the current analysis, researchers used electronic health records linked to Indiana’s statewide SARS-CoV-2 testing database and state vital records to evaluate the association between SARS-CoV-2 infection and all-cause mortality among 41,924 adults newly diagnosed with cancer between Jan. 1, 2019, and Dec. 31, 2020.
Most people with cancer were White (78.4%) and about half were male. At the time of diagnosis, 17% had one comorbid condition and about 10% had two or more. Most patients had breast cancer (14%), prostate cancer (13%), or melanoma (13%).
During the study period, 2,894 patients (7%) tested positive for SARS-CoV-2.
In multivariate adjusted analysis, the risk of death among those newly diagnosed with cancer increased by 91% (adjusted hazard ratio, 1.91) during the first year of the pandemic before vaccines were available, compared with the year before (January 2019 to Jan. 14, 2020).
During the pandemic period, the risk of death was roughly threefold higher among adults 65 years old and older, compared with adults 18-44 years old (aHR, 3.35).
When looking at the time from a cancer diagnosis to SARS-CoV-2 infection, infection was associated with an almost sevenfold increase in all-cause mortality (aHR, 6.91). Adults 65 years old and older had an almost threefold increased risk of dying, compared with their younger peers (aHR, 2.74).
Dr. Dixon and colleagues also observed an increased risk of death in men with cancer and COVID, compared with women (aHR, 1.23) and those with at least two comorbid conditions versus none (aHR, 2.12). In addition, the risk of dying was 9% higher among Indiana’s rural population than urban dwellers.
Compared with other cancer types, individuals with lung cancer and other digestive cancers had the highest risk of death after SARS-CoV-2 infection (aHR, 1.45 and 1.80, respectively).
“Our findings highlight the increased risk of death for adult cancer patients who test positive for COVID and underscore the importance to cancer patients – including those in remission – of vaccinations, boosters, and regular COVID testing,” Dr. Dixon commented.
“Our results should encourage individuals diagnosed with cancer not only to take preventive action, but also to expeditiously seek out treatments available in the marketplace should they test positive for COVID,” he added.
Support for the study was provided by Indiana University Simon Cancer Center and the Centers for Disease Control and Prevention. The authors have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
A new study underscores the importance of COVID-19 and regular COVID-19 testing among adults with a recent cancer diagnosis.
The Indiana statewide study, conducted at the beginning of the pandemic, found that
“This analysis provides additional empirical evidence on the magnitude of risk to patients with cancer whose immune systems are often weakened either by the disease or treatment,” the study team wrote.
The study was published online in JMIR Cancer.
Although evidence has consistently revealed similar findings, the risk of death among unvaccinated people with cancer and COVID-19 has not been nearly as high in previous studies, lead author Brian E. Dixon, PhD, MBA, with Indiana University Richard M. Fairbanks School of Public Health, Indianapolis, said in a statement. Previous studies from China, for instance, reported a two- to threefold greater risk of all-cause mortality among unvaccinated adults with cancer and COVID-19.
A potential reason for this discrepancy, Dr. Dixon noted, is that earlier studies were “generally smaller and made calculations based on data from a single cancer center or health system.”
Another reason is testing for COVID-19 early in the pandemic was limited to symptomatic individuals who may have had more severe infections, possibly leading to an overestimate of the association between SARS-CoV-2 infection, cancer, and all-cause mortality.
In the current analysis, researchers used electronic health records linked to Indiana’s statewide SARS-CoV-2 testing database and state vital records to evaluate the association between SARS-CoV-2 infection and all-cause mortality among 41,924 adults newly diagnosed with cancer between Jan. 1, 2019, and Dec. 31, 2020.
Most people with cancer were White (78.4%) and about half were male. At the time of diagnosis, 17% had one comorbid condition and about 10% had two or more. Most patients had breast cancer (14%), prostate cancer (13%), or melanoma (13%).
During the study period, 2,894 patients (7%) tested positive for SARS-CoV-2.
In multivariate adjusted analysis, the risk of death among those newly diagnosed with cancer increased by 91% (adjusted hazard ratio, 1.91) during the first year of the pandemic before vaccines were available, compared with the year before (January 2019 to Jan. 14, 2020).
During the pandemic period, the risk of death was roughly threefold higher among adults 65 years old and older, compared with adults 18-44 years old (aHR, 3.35).
When looking at the time from a cancer diagnosis to SARS-CoV-2 infection, infection was associated with an almost sevenfold increase in all-cause mortality (aHR, 6.91). Adults 65 years old and older had an almost threefold increased risk of dying, compared with their younger peers (aHR, 2.74).
Dr. Dixon and colleagues also observed an increased risk of death in men with cancer and COVID, compared with women (aHR, 1.23) and those with at least two comorbid conditions versus none (aHR, 2.12). In addition, the risk of dying was 9% higher among Indiana’s rural population than urban dwellers.
Compared with other cancer types, individuals with lung cancer and other digestive cancers had the highest risk of death after SARS-CoV-2 infection (aHR, 1.45 and 1.80, respectively).
“Our findings highlight the increased risk of death for adult cancer patients who test positive for COVID and underscore the importance to cancer patients – including those in remission – of vaccinations, boosters, and regular COVID testing,” Dr. Dixon commented.
“Our results should encourage individuals diagnosed with cancer not only to take preventive action, but also to expeditiously seek out treatments available in the marketplace should they test positive for COVID,” he added.
Support for the study was provided by Indiana University Simon Cancer Center and the Centers for Disease Control and Prevention. The authors have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
A new study underscores the importance of COVID-19 and regular COVID-19 testing among adults with a recent cancer diagnosis.
The Indiana statewide study, conducted at the beginning of the pandemic, found that
“This analysis provides additional empirical evidence on the magnitude of risk to patients with cancer whose immune systems are often weakened either by the disease or treatment,” the study team wrote.
The study was published online in JMIR Cancer.
Although evidence has consistently revealed similar findings, the risk of death among unvaccinated people with cancer and COVID-19 has not been nearly as high in previous studies, lead author Brian E. Dixon, PhD, MBA, with Indiana University Richard M. Fairbanks School of Public Health, Indianapolis, said in a statement. Previous studies from China, for instance, reported a two- to threefold greater risk of all-cause mortality among unvaccinated adults with cancer and COVID-19.
A potential reason for this discrepancy, Dr. Dixon noted, is that earlier studies were “generally smaller and made calculations based on data from a single cancer center or health system.”
Another reason is testing for COVID-19 early in the pandemic was limited to symptomatic individuals who may have had more severe infections, possibly leading to an overestimate of the association between SARS-CoV-2 infection, cancer, and all-cause mortality.
In the current analysis, researchers used electronic health records linked to Indiana’s statewide SARS-CoV-2 testing database and state vital records to evaluate the association between SARS-CoV-2 infection and all-cause mortality among 41,924 adults newly diagnosed with cancer between Jan. 1, 2019, and Dec. 31, 2020.
Most people with cancer were White (78.4%) and about half were male. At the time of diagnosis, 17% had one comorbid condition and about 10% had two or more. Most patients had breast cancer (14%), prostate cancer (13%), or melanoma (13%).
During the study period, 2,894 patients (7%) tested positive for SARS-CoV-2.
In multivariate adjusted analysis, the risk of death among those newly diagnosed with cancer increased by 91% (adjusted hazard ratio, 1.91) during the first year of the pandemic before vaccines were available, compared with the year before (January 2019 to Jan. 14, 2020).
During the pandemic period, the risk of death was roughly threefold higher among adults 65 years old and older, compared with adults 18-44 years old (aHR, 3.35).
When looking at the time from a cancer diagnosis to SARS-CoV-2 infection, infection was associated with an almost sevenfold increase in all-cause mortality (aHR, 6.91). Adults 65 years old and older had an almost threefold increased risk of dying, compared with their younger peers (aHR, 2.74).
Dr. Dixon and colleagues also observed an increased risk of death in men with cancer and COVID, compared with women (aHR, 1.23) and those with at least two comorbid conditions versus none (aHR, 2.12). In addition, the risk of dying was 9% higher among Indiana’s rural population than urban dwellers.
Compared with other cancer types, individuals with lung cancer and other digestive cancers had the highest risk of death after SARS-CoV-2 infection (aHR, 1.45 and 1.80, respectively).
“Our findings highlight the increased risk of death for adult cancer patients who test positive for COVID and underscore the importance to cancer patients – including those in remission – of vaccinations, boosters, and regular COVID testing,” Dr. Dixon commented.
“Our results should encourage individuals diagnosed with cancer not only to take preventive action, but also to expeditiously seek out treatments available in the marketplace should they test positive for COVID,” he added.
Support for the study was provided by Indiana University Simon Cancer Center and the Centers for Disease Control and Prevention. The authors have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FROM JMIR CANCER
This brain surgery was BYOS: Bring your own saxophone
Tumor vs. saxophone: The surgical grudge match
Brain surgery is a notoriously difficult task. There’s a reason we say, “Well, at least it’s not brain surgery” when we’re trying to convince someone that a task isn’t that tough. Make one wrong incision, cut the wrong neuron, and it’s goodbye higher cognitive function. And most people appreciate thinking. Crazy, right?
One would imagine that the act of brain surgery would become even more difficult when the patient brings his saxophone and plays it randomly throughout the operation. It’s a hospital, after all, not a jazz club. Patients don’t get to play musical instruments during other surgeries. Why should brain surgery patients get special treatment?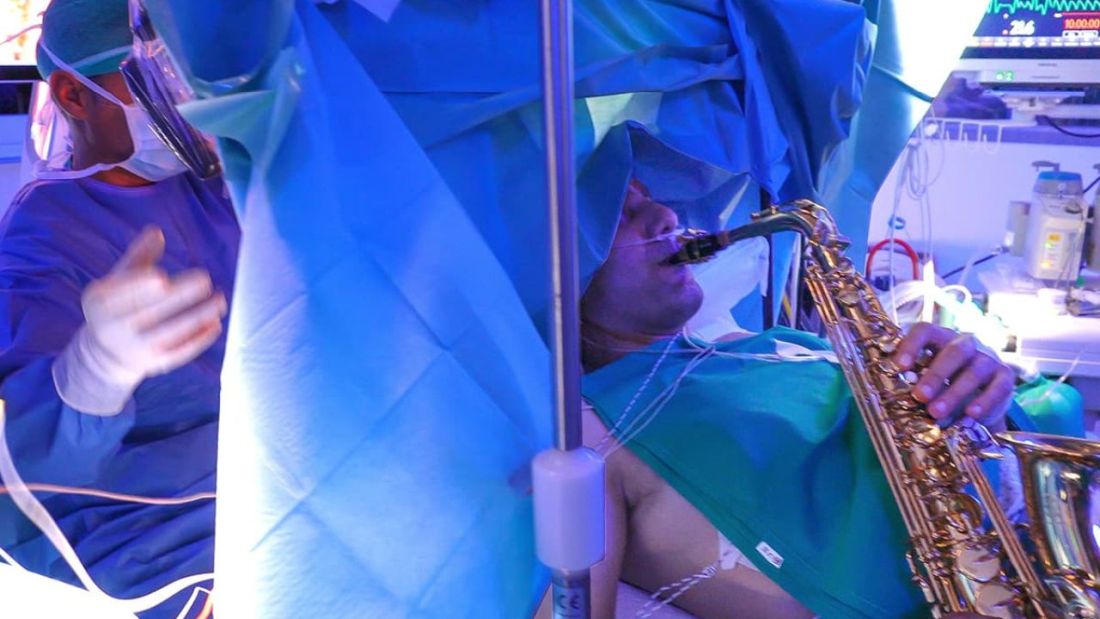
As it turns out, the musical performance was actually quite helpful. A man in Italy had a brain tumor in a particularly complex area, and he’s left-handed, which apparently makes the brain’s neural pathways much more complicated. Plus, he insisted that he retain his musical ability after the surgery. So he and his medical team had a crazy thought: Why not play the saxophone throughout the surgery? After all, according to head surgeon Christian Brogna, MD, playing an instrument means you understand music, which tests many higher cognitive functions such as coordination, mathematics, and memory.
And so, at various points throughout the 9-hour surgery, the patient played his saxophone for his doctors. Doing so allowed the surgeons to map the patient’s brain in a more complete and personalized fashion. With that extra knowledge, they were able to successfully remove the tumor while maintaining the patient’s musical ability, and the patient was discharged on Oct. 13, just 3 days after his operation.
While we’re happy the patient recovered, we do have to question his choice of music. During the surgery, he played the theme to the 1970 movie “Love Story” and the Italian national anthem. Perfectly fine pieces, no doubt, but the saxophone solo in “Jungleland” exists. And we could listen to that for 9 hours straight. In fact, we do that every Friday in the LOTME office.
Basketball has the Big Dance. Mosquitoes get the Big Sniff
In this week’s installment of our seemingly never-ending series, “Mosquitoes and the scientists who love them,” we visit The Rockefeller University in New York, where the olfactory capabilities of Aedes Aegypti – the primary vector species for Zika, dengue, yellow fever, and chikungunya – became the subject of a round robin–style tournament.
First things first, though. If you’re going to test mosquito noses, you have to give them something to smell. The researchers enrolled eight humans who were willing to wear nylon stockings on their forearms for 6 hours a day for multiple days. “Over the next few years, the researchers tested the nylons against each other in all possible pairings,” Leslie B. Vosshall, PhD, and associates said in a statement from the university. In other words, mosquito March Madness.
Nylons from different participants were hooked up in pairs to an olfactometer assay consisting of a plexiglass chamber divided into two tubes, each ending in a box that held a stocking. The mosquitoes were placed in the main chamber and observed as they flew down the tubes toward one stocking or the other.
Eventually, the “winner” of the “tournament” was Subject 33. And no, we don’t know why there was a Subject 33 since the study involved only eight participants. We do know that the nylons worn by Subject 33 were “four times more attractive to the mosquitoes than the next most-attractive study participant, and an astonishing 100 times more appealing than the least attractive, Subject 19,” according to the written statement.
Chemical analysis identified 50 molecular compounds that were elevated in the sebum of the high-attracting participants, and eventually the investigators discovered that mosquito magnets produced carboxylic acids at much higher levels than the less-attractive volunteers.
We could go on about the research team genetically engineering mosquitoes without odor receptors, but we have to save something for later. Tune in again next week for another exciting episode of “Mosquitoes and the scientists who love them.”
Are women better with words?
Men vs. Women is probably the oldest argument in the book, but there may now be movement. Researchers have been able not only to shift the advantage toward women, but also to use that knowledge to medical advantage.
When it comes to the matter of words and remembering them, women apparently have men beat. The margin is small, said lead author Marco Hirnstein, PhD, of the University of Bergen, Norway, but, after performing a meta-analysis of 168 published studies and PhD theses involving more than 350,000 participants, it’s pretty clear. The research supports women’s advantage over men in recall, verbal fluency (categorical and phonemic), and recognition.
So how is this information useful from a medical standpoint?
Dr. Hirnstein and colleagues suggested that this information can help in interpreting diagnostic assessment results. The example given was dementia diagnosis. Since women are underdiagnosed because their baseline exceeds average while men are overdiagnosed, taking gender and performance into account could clear up or catch cases that might otherwise slip through the cracks.
Now, let’s just put this part of the debate to rest and take this not only as a win for women but for science as well.
Tumor vs. saxophone: The surgical grudge match
Brain surgery is a notoriously difficult task. There’s a reason we say, “Well, at least it’s not brain surgery” when we’re trying to convince someone that a task isn’t that tough. Make one wrong incision, cut the wrong neuron, and it’s goodbye higher cognitive function. And most people appreciate thinking. Crazy, right?
One would imagine that the act of brain surgery would become even more difficult when the patient brings his saxophone and plays it randomly throughout the operation. It’s a hospital, after all, not a jazz club. Patients don’t get to play musical instruments during other surgeries. Why should brain surgery patients get special treatment?
As it turns out, the musical performance was actually quite helpful. A man in Italy had a brain tumor in a particularly complex area, and he’s left-handed, which apparently makes the brain’s neural pathways much more complicated. Plus, he insisted that he retain his musical ability after the surgery. So he and his medical team had a crazy thought: Why not play the saxophone throughout the surgery? After all, according to head surgeon Christian Brogna, MD, playing an instrument means you understand music, which tests many higher cognitive functions such as coordination, mathematics, and memory.
And so, at various points throughout the 9-hour surgery, the patient played his saxophone for his doctors. Doing so allowed the surgeons to map the patient’s brain in a more complete and personalized fashion. With that extra knowledge, they were able to successfully remove the tumor while maintaining the patient’s musical ability, and the patient was discharged on Oct. 13, just 3 days after his operation.
While we’re happy the patient recovered, we do have to question his choice of music. During the surgery, he played the theme to the 1970 movie “Love Story” and the Italian national anthem. Perfectly fine pieces, no doubt, but the saxophone solo in “Jungleland” exists. And we could listen to that for 9 hours straight. In fact, we do that every Friday in the LOTME office.
Basketball has the Big Dance. Mosquitoes get the Big Sniff
In this week’s installment of our seemingly never-ending series, “Mosquitoes and the scientists who love them,” we visit The Rockefeller University in New York, where the olfactory capabilities of Aedes Aegypti – the primary vector species for Zika, dengue, yellow fever, and chikungunya – became the subject of a round robin–style tournament.
First things first, though. If you’re going to test mosquito noses, you have to give them something to smell. The researchers enrolled eight humans who were willing to wear nylon stockings on their forearms for 6 hours a day for multiple days. “Over the next few years, the researchers tested the nylons against each other in all possible pairings,” Leslie B. Vosshall, PhD, and associates said in a statement from the university. In other words, mosquito March Madness.
Nylons from different participants were hooked up in pairs to an olfactometer assay consisting of a plexiglass chamber divided into two tubes, each ending in a box that held a stocking. The mosquitoes were placed in the main chamber and observed as they flew down the tubes toward one stocking or the other.
Eventually, the “winner” of the “tournament” was Subject 33. And no, we don’t know why there was a Subject 33 since the study involved only eight participants. We do know that the nylons worn by Subject 33 were “four times more attractive to the mosquitoes than the next most-attractive study participant, and an astonishing 100 times more appealing than the least attractive, Subject 19,” according to the written statement.
Chemical analysis identified 50 molecular compounds that were elevated in the sebum of the high-attracting participants, and eventually the investigators discovered that mosquito magnets produced carboxylic acids at much higher levels than the less-attractive volunteers.
We could go on about the research team genetically engineering mosquitoes without odor receptors, but we have to save something for later. Tune in again next week for another exciting episode of “Mosquitoes and the scientists who love them.”
Are women better with words?
Men vs. Women is probably the oldest argument in the book, but there may now be movement. Researchers have been able not only to shift the advantage toward women, but also to use that knowledge to medical advantage.
When it comes to the matter of words and remembering them, women apparently have men beat. The margin is small, said lead author Marco Hirnstein, PhD, of the University of Bergen, Norway, but, after performing a meta-analysis of 168 published studies and PhD theses involving more than 350,000 participants, it’s pretty clear. The research supports women’s advantage over men in recall, verbal fluency (categorical and phonemic), and recognition.
So how is this information useful from a medical standpoint?
Dr. Hirnstein and colleagues suggested that this information can help in interpreting diagnostic assessment results. The example given was dementia diagnosis. Since women are underdiagnosed because their baseline exceeds average while men are overdiagnosed, taking gender and performance into account could clear up or catch cases that might otherwise slip through the cracks.
Now, let’s just put this part of the debate to rest and take this not only as a win for women but for science as well.
Tumor vs. saxophone: The surgical grudge match
Brain surgery is a notoriously difficult task. There’s a reason we say, “Well, at least it’s not brain surgery” when we’re trying to convince someone that a task isn’t that tough. Make one wrong incision, cut the wrong neuron, and it’s goodbye higher cognitive function. And most people appreciate thinking. Crazy, right?
One would imagine that the act of brain surgery would become even more difficult when the patient brings his saxophone and plays it randomly throughout the operation. It’s a hospital, after all, not a jazz club. Patients don’t get to play musical instruments during other surgeries. Why should brain surgery patients get special treatment?
As it turns out, the musical performance was actually quite helpful. A man in Italy had a brain tumor in a particularly complex area, and he’s left-handed, which apparently makes the brain’s neural pathways much more complicated. Plus, he insisted that he retain his musical ability after the surgery. So he and his medical team had a crazy thought: Why not play the saxophone throughout the surgery? After all, according to head surgeon Christian Brogna, MD, playing an instrument means you understand music, which tests many higher cognitive functions such as coordination, mathematics, and memory.
And so, at various points throughout the 9-hour surgery, the patient played his saxophone for his doctors. Doing so allowed the surgeons to map the patient’s brain in a more complete and personalized fashion. With that extra knowledge, they were able to successfully remove the tumor while maintaining the patient’s musical ability, and the patient was discharged on Oct. 13, just 3 days after his operation.
While we’re happy the patient recovered, we do have to question his choice of music. During the surgery, he played the theme to the 1970 movie “Love Story” and the Italian national anthem. Perfectly fine pieces, no doubt, but the saxophone solo in “Jungleland” exists. And we could listen to that for 9 hours straight. In fact, we do that every Friday in the LOTME office.
Basketball has the Big Dance. Mosquitoes get the Big Sniff
In this week’s installment of our seemingly never-ending series, “Mosquitoes and the scientists who love them,” we visit The Rockefeller University in New York, where the olfactory capabilities of Aedes Aegypti – the primary vector species for Zika, dengue, yellow fever, and chikungunya – became the subject of a round robin–style tournament.
First things first, though. If you’re going to test mosquito noses, you have to give them something to smell. The researchers enrolled eight humans who were willing to wear nylon stockings on their forearms for 6 hours a day for multiple days. “Over the next few years, the researchers tested the nylons against each other in all possible pairings,” Leslie B. Vosshall, PhD, and associates said in a statement from the university. In other words, mosquito March Madness.
Nylons from different participants were hooked up in pairs to an olfactometer assay consisting of a plexiglass chamber divided into two tubes, each ending in a box that held a stocking. The mosquitoes were placed in the main chamber and observed as they flew down the tubes toward one stocking or the other.
Eventually, the “winner” of the “tournament” was Subject 33. And no, we don’t know why there was a Subject 33 since the study involved only eight participants. We do know that the nylons worn by Subject 33 were “four times more attractive to the mosquitoes than the next most-attractive study participant, and an astonishing 100 times more appealing than the least attractive, Subject 19,” according to the written statement.
Chemical analysis identified 50 molecular compounds that were elevated in the sebum of the high-attracting participants, and eventually the investigators discovered that mosquito magnets produced carboxylic acids at much higher levels than the less-attractive volunteers.
We could go on about the research team genetically engineering mosquitoes without odor receptors, but we have to save something for later. Tune in again next week for another exciting episode of “Mosquitoes and the scientists who love them.”
Are women better with words?
Men vs. Women is probably the oldest argument in the book, but there may now be movement. Researchers have been able not only to shift the advantage toward women, but also to use that knowledge to medical advantage.
When it comes to the matter of words and remembering them, women apparently have men beat. The margin is small, said lead author Marco Hirnstein, PhD, of the University of Bergen, Norway, but, after performing a meta-analysis of 168 published studies and PhD theses involving more than 350,000 participants, it’s pretty clear. The research supports women’s advantage over men in recall, verbal fluency (categorical and phonemic), and recognition.
So how is this information useful from a medical standpoint?
Dr. Hirnstein and colleagues suggested that this information can help in interpreting diagnostic assessment results. The example given was dementia diagnosis. Since women are underdiagnosed because their baseline exceeds average while men are overdiagnosed, taking gender and performance into account could clear up or catch cases that might otherwise slip through the cracks.
Now, let’s just put this part of the debate to rest and take this not only as a win for women but for science as well.
Vision loss may be a risk with PRP facial injections
A systematic review was recently conducted by Wu and colleagues examining the risk of blindness associated with platelet-rich plasma (PRP) injection. In dermatology, PRP is used more commonly now than 5 years ago to promote hair growth with injections on the scalp, as an adjunct to microneedling procedures, and sometimes – in a similar way to facial fillers – to improve volume loss, and skin tone and texture (particularly to the tear trough region).
Total unilateral blindness occurred in all cases. In one of the seven reported cases, the patient experienced recovery of vision after 3 months, but with some residual deficits noted on the ophthalmologist examination. In this case, the patient was evaluated and treated by an ophthalmologist within 3 hours of symptom onset.
In addition, four cases were reported from Venezuela, one from the United States, one from the United Kingdom, and one from Malaysia. Similar to reports of blindness with facial fillers, the most common injection site reported with this adverse effect was the glabella (five cases);
Other reports involved injections of the forehead (two), followed by the nasolabial fold (one), lateral canthus (one), and temporomandibular joint (one). Two of the seven patients received injections at more than one site, resulting in the total number of injections reported (10) being higher than the number of patients.
The risk of blindness is inherent with deep injection into a vessel that anastomoses with the blood supply to the eye. No mention was made as to whether PRP or platelet-rich fibrin was used. Other details are lacking from the original articles as to injection technique and whether or not cannula injection was used. No treatment was attempted in four of seven cases.
As plasma is native to the arteries and dissolves in the blood stream naturally, the mechanism as to why retinal artery occlusion or blindness would occur is not completely clear. One theory is that it is volume related and results from the speed of injection, causing a large rapid bolus that temporarily occludes or compresses an involved vessel.
Another theory is that damage to the vessel results from the injection itself or injection technique, leading to a clotting cascade and clot of the involved vessel with subsequent retrograde flow or blockade of the retinal artery. But if this were the case, we would expect to hear about more cases of clots leading to vascular occlusion or skin necrosis, which does not typically occur or we do not hear about.
Details about proper collection materials and technique or mixing with some other materials are also unknown in these cases, thus leaving the possibility that a more occlusive material may have been injected, as opposed to the fluid-like composition of the typical PRP preparation.With regards to risk with scalp PRP injection, the frontal scalp does receive blood supply from the supratrochlear artery that anastomoses with the angular artery of the face – both of which anastomose with the retinal artery (where occlusion would occur via back flow). The scalp tributaries are small and far enough away from the retina at that point that risk of back flow the to retinal artery should be minimal. Additionally, no reports of vascular occlusion from PRP scalp injection leading to skin necrosis have ever been reported. Of note, this is also not a risk that has been reported with the use of PRP with microneedling procedures, where PRP is placed on top of the skin before, during and after microneedling.
Anything that occludes the blood supply to the eye, whether it be fat, filler, or PRP, has an inherent risk of blindness. As there is no reversal agent or designated treatment for PRP occlusion, care must be taken to minimize risk, including awareness of anatomy and avoidance of injection into high risk areas, and cannula use where appropriate. Gentle, slow, low-volume administration, and when possible, use of a retrograde injection technique, may also be helpful.
Dr. Wesley and Lily Talakoub, MD, are cocontributors to this column. Dr. Wesley practices dermatology in Beverly Hills, Calif. Dr. Talakoub is in private practice in McLean, Va. This month’s column is by Dr. Wesley. Write to them at [email protected]. They had no relevant disclosures.
A systematic review was recently conducted by Wu and colleagues examining the risk of blindness associated with platelet-rich plasma (PRP) injection. In dermatology, PRP is used more commonly now than 5 years ago to promote hair growth with injections on the scalp, as an adjunct to microneedling procedures, and sometimes – in a similar way to facial fillers – to improve volume loss, and skin tone and texture (particularly to the tear trough region).
Total unilateral blindness occurred in all cases. In one of the seven reported cases, the patient experienced recovery of vision after 3 months, but with some residual deficits noted on the ophthalmologist examination. In this case, the patient was evaluated and treated by an ophthalmologist within 3 hours of symptom onset.
In addition, four cases were reported from Venezuela, one from the United States, one from the United Kingdom, and one from Malaysia. Similar to reports of blindness with facial fillers, the most common injection site reported with this adverse effect was the glabella (five cases);
Other reports involved injections of the forehead (two), followed by the nasolabial fold (one), lateral canthus (one), and temporomandibular joint (one). Two of the seven patients received injections at more than one site, resulting in the total number of injections reported (10) being higher than the number of patients.
The risk of blindness is inherent with deep injection into a vessel that anastomoses with the blood supply to the eye. No mention was made as to whether PRP or platelet-rich fibrin was used. Other details are lacking from the original articles as to injection technique and whether or not cannula injection was used. No treatment was attempted in four of seven cases.
As plasma is native to the arteries and dissolves in the blood stream naturally, the mechanism as to why retinal artery occlusion or blindness would occur is not completely clear. One theory is that it is volume related and results from the speed of injection, causing a large rapid bolus that temporarily occludes or compresses an involved vessel.
Another theory is that damage to the vessel results from the injection itself or injection technique, leading to a clotting cascade and clot of the involved vessel with subsequent retrograde flow or blockade of the retinal artery. But if this were the case, we would expect to hear about more cases of clots leading to vascular occlusion or skin necrosis, which does not typically occur or we do not hear about.
Details about proper collection materials and technique or mixing with some other materials are also unknown in these cases, thus leaving the possibility that a more occlusive material may have been injected, as opposed to the fluid-like composition of the typical PRP preparation.With regards to risk with scalp PRP injection, the frontal scalp does receive blood supply from the supratrochlear artery that anastomoses with the angular artery of the face – both of which anastomose with the retinal artery (where occlusion would occur via back flow). The scalp tributaries are small and far enough away from the retina at that point that risk of back flow the to retinal artery should be minimal. Additionally, no reports of vascular occlusion from PRP scalp injection leading to skin necrosis have ever been reported. Of note, this is also not a risk that has been reported with the use of PRP with microneedling procedures, where PRP is placed on top of the skin before, during and after microneedling.
Anything that occludes the blood supply to the eye, whether it be fat, filler, or PRP, has an inherent risk of blindness. As there is no reversal agent or designated treatment for PRP occlusion, care must be taken to minimize risk, including awareness of anatomy and avoidance of injection into high risk areas, and cannula use where appropriate. Gentle, slow, low-volume administration, and when possible, use of a retrograde injection technique, may also be helpful.
Dr. Wesley and Lily Talakoub, MD, are cocontributors to this column. Dr. Wesley practices dermatology in Beverly Hills, Calif. Dr. Talakoub is in private practice in McLean, Va. This month’s column is by Dr. Wesley. Write to them at [email protected]. They had no relevant disclosures.
A systematic review was recently conducted by Wu and colleagues examining the risk of blindness associated with platelet-rich plasma (PRP) injection. In dermatology, PRP is used more commonly now than 5 years ago to promote hair growth with injections on the scalp, as an adjunct to microneedling procedures, and sometimes – in a similar way to facial fillers – to improve volume loss, and skin tone and texture (particularly to the tear trough region).
Total unilateral blindness occurred in all cases. In one of the seven reported cases, the patient experienced recovery of vision after 3 months, but with some residual deficits noted on the ophthalmologist examination. In this case, the patient was evaluated and treated by an ophthalmologist within 3 hours of symptom onset.
In addition, four cases were reported from Venezuela, one from the United States, one from the United Kingdom, and one from Malaysia. Similar to reports of blindness with facial fillers, the most common injection site reported with this adverse effect was the glabella (five cases);
Other reports involved injections of the forehead (two), followed by the nasolabial fold (one), lateral canthus (one), and temporomandibular joint (one). Two of the seven patients received injections at more than one site, resulting in the total number of injections reported (10) being higher than the number of patients.
The risk of blindness is inherent with deep injection into a vessel that anastomoses with the blood supply to the eye. No mention was made as to whether PRP or platelet-rich fibrin was used. Other details are lacking from the original articles as to injection technique and whether or not cannula injection was used. No treatment was attempted in four of seven cases.
As plasma is native to the arteries and dissolves in the blood stream naturally, the mechanism as to why retinal artery occlusion or blindness would occur is not completely clear. One theory is that it is volume related and results from the speed of injection, causing a large rapid bolus that temporarily occludes or compresses an involved vessel.
Another theory is that damage to the vessel results from the injection itself or injection technique, leading to a clotting cascade and clot of the involved vessel with subsequent retrograde flow or blockade of the retinal artery. But if this were the case, we would expect to hear about more cases of clots leading to vascular occlusion or skin necrosis, which does not typically occur or we do not hear about.
Details about proper collection materials and technique or mixing with some other materials are also unknown in these cases, thus leaving the possibility that a more occlusive material may have been injected, as opposed to the fluid-like composition of the typical PRP preparation.With regards to risk with scalp PRP injection, the frontal scalp does receive blood supply from the supratrochlear artery that anastomoses with the angular artery of the face – both of which anastomose with the retinal artery (where occlusion would occur via back flow). The scalp tributaries are small and far enough away from the retina at that point that risk of back flow the to retinal artery should be minimal. Additionally, no reports of vascular occlusion from PRP scalp injection leading to skin necrosis have ever been reported. Of note, this is also not a risk that has been reported with the use of PRP with microneedling procedures, where PRP is placed on top of the skin before, during and after microneedling.
Anything that occludes the blood supply to the eye, whether it be fat, filler, or PRP, has an inherent risk of blindness. As there is no reversal agent or designated treatment for PRP occlusion, care must be taken to minimize risk, including awareness of anatomy and avoidance of injection into high risk areas, and cannula use where appropriate. Gentle, slow, low-volume administration, and when possible, use of a retrograde injection technique, may also be helpful.
Dr. Wesley and Lily Talakoub, MD, are cocontributors to this column. Dr. Wesley practices dermatology in Beverly Hills, Calif. Dr. Talakoub is in private practice in McLean, Va. This month’s column is by Dr. Wesley. Write to them at [email protected]. They had no relevant disclosures.
Atypical Localized Scleroderma Development During Nivolumab Therapy for Metastatic Lung Adenocarcinoma
To the Editor:
Immune checkpoint inhibitors such as anti–programmed cell death protein 1 (anti–PD-1) and anticytotoxic T lymphocyte–associated protein 4 therapies are a promising class of cancer therapeutics. However, they are associated with a variety of immune-related adverse events (irAEs), including cutaneous toxicity.1 The PD-1/programmed death ligand 1 (PD-L1) pathway is important for the maintenance of immune tolerance, and a blockade has been shown to lead to development of various autoimmune diseases.2 We present the case of a patient who developed new-onset localized scleroderma during treatment with the PD-1 inhibitor nivolumab.
A 65-year-old woman presented with a rash on the left thigh that was associated with pruritus, pain, and a pulling sensation. She had a history of stage IV lung adenocarcinoma, with a mass in the right upper lobe with metastatic foci to the left femur, right humerus, right hilar, and pretracheal lymph nodes. She received palliative radiation to the left femur and was started on carboplatin and pemetrexed. Metastasis to the liver was noted after completion of 6 cycles of therapy, and the patient’s treatment was changed to nivolumab. After 17 months on nivolumab therapy (2 years after initial diagnosis and 20 months after radiation therapy), she presented to our dermatology clinic with a cutaneous eruption on the buttocks that spread to the left thigh. The rash failed to improve after 1 month of treatment with emollients and triamcinolone cream 0.1%.
At the current presentation, which was 2 months after she initially presented to our clinic, dermatologic examination revealed erythematous and sclerotic plaques on the left lateral thigh (Figure 1A). Betamethasone cream 0.05% was prescribed, and nivolumab was discontinued due to progression of cutaneous symptoms. A punch biopsy from the left thigh demonstrated superficial dermal sclerosis that was suggestive of chronic radiation dermatitis; direct immunofluorescence testing was negative. The patient was started on prednisone 50 mg daily, which resulted in mild improvement in symptoms.
Within 6 months, new sclerotic plaques developed on the patient’s back and right thigh (Figure 1B). Because the lesions were located outside the radiation field of the left femur, a second biopsy was obtained from the right thigh. Histopathology revealed extensive dermal sclerosis and a perivascular lymphoplasmacytic infiltrate (Figure 2). An antinuclear antibody test was weakly positive (1:40, nucleolar pattern) with a negative extractable nuclear antigen panel result. Anti–double-stranded DNA, anti–topoisomerase 1, anti-Smith, antiribonucleoprotein, anti–Sjögren syndrome type A, anti–Sjögren syndrome type B, and anticentromere serology test results were negative. The patient denied decreased oral aperture, difficulty swallowing, or Raynaud phenomenon. Due to the atypical clinical presentation in the setting of PD-1 inhibitor therapy, the etiology of the eruption was potentially attributable to nivolumab. She was started on treatment with methotrexate 20 mg weekly and clobetasol cream 0.05% twice daily; she continued taking prednisone 5 mg daily. The cutaneous manifestations on the patient’s back completely resolved, and the legs continued to gradually improve on this regimen. Immunotherapy continued to be held due to skin toxicity.
Localized scleroderma is an autoimmune disorder characterized by inflammation and skin thickening. Overactive fibroblasts produce excess collagen, leading to the clinical symptoms of skin thickening, hardening, and discoloration.3 Lesions frequently develop on the arms, face, or legs and can present as patches or linear bands. Unlike systemic sclerosis, the internal organs typically are uninvolved; however, sclerotic lesions can be disfiguring and cause notable disability if they impede joint movement.
The PD-1/PD-L1 pathway is a negative regulator of the immune response that inactivates T cells and helps maintain self-tolerance. Modulation of the PD-1/PD-L1 pathway and overexpression of PD-L1 are seen in various cancers as a mechanism to help malignant cells avoid immune destruction.4 Conversely, inhibition of this pathway can be used to stimulate an antitumor immune response. This checkpoint inhibition strategy has been highly successful for the treatment of various cancers including melanoma and non–small cell lung carcinoma. There are several checkpoint inhibitors approved in the United States that are used for cancer therapy and target the PD-1/PD-L1 pathway, such as nivolumab, pembrolizumab, atezolizumab, durvalumab, and avelumab.4 A downside of checkpoint inhibitor treatment is that uncontrolled T-cell activation can lead to irAEs, including cutaneous eruptions, pruritus, diarrhea, colitis, hepatitis, endocrinopathies, pneumonitis, and renal insufficiency.5 These toxicities are reversible if treated appropriately but can cause notable morbidity and mortality if left unrecognized. Cutaneous eruption is one of the most common irAEs associated with anti–PD-1 and anti–PD-L1 therapies and can limit therapeutic efficacy, as the drug may need to be held or discontinued due to the severity of the eruption.6 Mid-potency to high-potency topical corticosteroids and systemic antihistamines are first-line treatments of grades 1 and 2 skin toxicities associated with PD-1 inhibitor therapy. For eruptions classified as grades 3 or 4 or refractory grade 2, discontinuation of the drug and systemic corticosteroids is recommended.7
The cutaneous eruption in immunotherapy-mediated dermatitis is thought to be largely mediated by activated T cells infiltrating the dermis.8 In localized scleroderma, increased tumor necrosis factor α, IFN-γ, IFN-γ–induced protein 10, and granulocyte macrophage colony stimulating factor activity have been shown to correlate with disease activity.9,10 Interestingly, increased tumor necrosis factor α and IFN-γ correlate with better response and increased overall survival in PD-1 inhibition therapy, suggesting a correlation between PD-1 inhibition and T helper activation as noted by the etiology of sclerosis in our patient.11 Additionally, history of radiation was a confounding factor in the diagnosis of our patient, as both sclerodermoid reactions and chronic radiation dermatitis can present with dermal sclerosis. However, the progression of disease outside of the radiation field excluded this etiology. Although new-onset sclerodermoid reactions have been reported with PD-1 inhibitors, they have been described secondary to sclerodermoid reactions from treatment with pembrolizumab.12,13 One case series reported a case of diffuse sclerodermoid reaction and a limited reaction in response to pembrolizumab treatment, while another case report described a relapse of generalized morphea in response to pembrolizumab treatment.12,13 One case of relapsing morphea in response to nivolumab treatment for stage IV lung adenocarcinoma also has been reported.14
Cutaneous toxicities are one of the most common irAEs associated with checkpoint inhibitors and are seen in more than one-third of treated patients. Most frequently, these irAEs manifest as spongiotic dermatitis on histopathology, but a broad spectrum of cutaneous reactions have been observed.15 Although sclerodermoid reactions have been reported with PD-1 inhibitors, most are described secondary to sclerodermoid reactions with pembrolizumab and involve relapse of previously diagnosed morphea rather than new-onset disease.12-14
Our case highlights new-onset localized scleroderma in the setting of nivolumab therapy that showed clinical improvement with methotrexate and topical and systemic steroids. This reaction pattern should be considered in all patients who develop cutaneous eruptions when treated with a PD-1 inhibitor. There should be a high index of suspicion for the potential occurrence of irAEs to ensure early recognition and treatment to minimize morbidity and maximize adherence to therapy for the underlying malignancy.
- Baxi S, Yang A, Gennarelli RL, et al. Immune-related adverse events for anti-PD-1 and anti-PD-L1 drugs: systematic review and meta-analysis. BMJ. 2018;360:k793.
- Dai S, Jia R, Zhang X, et al. The PD-1/PD-Ls pathway and autoimmune diseases. Cell Immunol. 2014;290:72-79.
- Badea I, Taylor M, Rosenberg A, et al. Pathogenesis and therapeutic approaches for improved topical treatment in localized scleroderma and systemic sclerosis. Rheumatology (Oxford). 2009;48:213-221.
- Constantinidou A, Alifieris C, Trafalis DT. Targeting programmed cell death-1 (PD-1) and ligand (PD-L1): a new era in cancer active immunotherapy. Pharmacol Ther. 2019;194:84-106.
- Villadolid J, Asim A. Immune checkpoint inhibitors in clinical practice: update on management of immune-related toxicities. Transl Lung Cancer Res. 2015;4:560-575.
- Naidoo J, Page DB, Li BT, et al. Toxicities of the anti-PD-1 and anti-PD-L1 immune checkpoint antibodies. Ann Oncol. 2016;27:1362.
- O’Kane GM, Labbé C, Doherty MK, et al. Monitoring and management of immune-related adverse events associated with programmed cell death protein-1 axis inhibitors in lung cancer. Oncologist. 2017;22:70-80.
- Shi VJ, Rodic N, Gettinger S, et al. Clinical and histologic features of lichenoid mucocutaneous eruptions due to anti-programmed celldeath 1 and anti-programmed cell death ligand 1 immunotherapy. JAMA Dermatol. 2016;152:1128-1136.
- Torok KS, Kurzinski K, Kelsey C, et al. Peripheral blood cytokine and chemokine profiles in juvenile localized scleroderma: T-helper cell-associated cytokine profiles. Semin Arthritis Rheum. 2015;45:284-293.
- Guo X, Higgs BW, Bay-Jensen AC, et al. Suppression of T cell activation and collagen accumulation by an anti-IFNAR1 mAb, anifrolumab, in adult patients with systemic sclerosis. J Invest Dermatol. 2015;135:2402-2409.
- Boutsikou E, Domvri K, Hardavella G, et al. Tumor necrosis factor, interferon-gamma and interleukins as predictive markers of antiprogrammed cell-death protein-1 treatment in advanced non-small cell lung cancer: a pragmatic approach in clinical practice. Ther Adv Med Oncol. 2018;10:1758835918768238.
- Barbosa NS, Wetter DA, Wieland CN, et al. Scleroderma induced by pembrolizumab: a case series. Mayo Clin Proc. 2017;92:1158-1163.
- Cheng MW, Hisaw LD, Bernet L. Generalized morphea in the setting of pembrolizumab. Int J Dermatol. 2019;58:736-738.
- Alegre-Sánchez A, Fonda-Pascual P, Saceda-Corralo D, et al. Relapse of morphea during nivolumab therapy for lung adenocarcinoma. Actas Dermosifiliogr. 2017;108:69-70.
- Sibaud V. Dermatologic reactions to immune checkpoint inhibitors: skin toxicities and immunotherapy. Am J Clin Dermatol. 2018;19:345-361.
To the Editor:
Immune checkpoint inhibitors such as anti–programmed cell death protein 1 (anti–PD-1) and anticytotoxic T lymphocyte–associated protein 4 therapies are a promising class of cancer therapeutics. However, they are associated with a variety of immune-related adverse events (irAEs), including cutaneous toxicity.1 The PD-1/programmed death ligand 1 (PD-L1) pathway is important for the maintenance of immune tolerance, and a blockade has been shown to lead to development of various autoimmune diseases.2 We present the case of a patient who developed new-onset localized scleroderma during treatment with the PD-1 inhibitor nivolumab.
A 65-year-old woman presented with a rash on the left thigh that was associated with pruritus, pain, and a pulling sensation. She had a history of stage IV lung adenocarcinoma, with a mass in the right upper lobe with metastatic foci to the left femur, right humerus, right hilar, and pretracheal lymph nodes. She received palliative radiation to the left femur and was started on carboplatin and pemetrexed. Metastasis to the liver was noted after completion of 6 cycles of therapy, and the patient’s treatment was changed to nivolumab. After 17 months on nivolumab therapy (2 years after initial diagnosis and 20 months after radiation therapy), she presented to our dermatology clinic with a cutaneous eruption on the buttocks that spread to the left thigh. The rash failed to improve after 1 month of treatment with emollients and triamcinolone cream 0.1%.
At the current presentation, which was 2 months after she initially presented to our clinic, dermatologic examination revealed erythematous and sclerotic plaques on the left lateral thigh (Figure 1A). Betamethasone cream 0.05% was prescribed, and nivolumab was discontinued due to progression of cutaneous symptoms. A punch biopsy from the left thigh demonstrated superficial dermal sclerosis that was suggestive of chronic radiation dermatitis; direct immunofluorescence testing was negative. The patient was started on prednisone 50 mg daily, which resulted in mild improvement in symptoms.

Within 6 months, new sclerotic plaques developed on the patient’s back and right thigh (Figure 1B). Because the lesions were located outside the radiation field of the left femur, a second biopsy was obtained from the right thigh. Histopathology revealed extensive dermal sclerosis and a perivascular lymphoplasmacytic infiltrate (Figure 2). An antinuclear antibody test was weakly positive (1:40, nucleolar pattern) with a negative extractable nuclear antigen panel result. Anti–double-stranded DNA, anti–topoisomerase 1, anti-Smith, antiribonucleoprotein, anti–Sjögren syndrome type A, anti–Sjögren syndrome type B, and anticentromere serology test results were negative. The patient denied decreased oral aperture, difficulty swallowing, or Raynaud phenomenon. Due to the atypical clinical presentation in the setting of PD-1 inhibitor therapy, the etiology of the eruption was potentially attributable to nivolumab. She was started on treatment with methotrexate 20 mg weekly and clobetasol cream 0.05% twice daily; she continued taking prednisone 5 mg daily. The cutaneous manifestations on the patient’s back completely resolved, and the legs continued to gradually improve on this regimen. Immunotherapy continued to be held due to skin toxicity.

Localized scleroderma is an autoimmune disorder characterized by inflammation and skin thickening. Overactive fibroblasts produce excess collagen, leading to the clinical symptoms of skin thickening, hardening, and discoloration.3 Lesions frequently develop on the arms, face, or legs and can present as patches or linear bands. Unlike systemic sclerosis, the internal organs typically are uninvolved; however, sclerotic lesions can be disfiguring and cause notable disability if they impede joint movement.
The PD-1/PD-L1 pathway is a negative regulator of the immune response that inactivates T cells and helps maintain self-tolerance. Modulation of the PD-1/PD-L1 pathway and overexpression of PD-L1 are seen in various cancers as a mechanism to help malignant cells avoid immune destruction.4 Conversely, inhibition of this pathway can be used to stimulate an antitumor immune response. This checkpoint inhibition strategy has been highly successful for the treatment of various cancers including melanoma and non–small cell lung carcinoma. There are several checkpoint inhibitors approved in the United States that are used for cancer therapy and target the PD-1/PD-L1 pathway, such as nivolumab, pembrolizumab, atezolizumab, durvalumab, and avelumab.4 A downside of checkpoint inhibitor treatment is that uncontrolled T-cell activation can lead to irAEs, including cutaneous eruptions, pruritus, diarrhea, colitis, hepatitis, endocrinopathies, pneumonitis, and renal insufficiency.5 These toxicities are reversible if treated appropriately but can cause notable morbidity and mortality if left unrecognized. Cutaneous eruption is one of the most common irAEs associated with anti–PD-1 and anti–PD-L1 therapies and can limit therapeutic efficacy, as the drug may need to be held or discontinued due to the severity of the eruption.6 Mid-potency to high-potency topical corticosteroids and systemic antihistamines are first-line treatments of grades 1 and 2 skin toxicities associated with PD-1 inhibitor therapy. For eruptions classified as grades 3 or 4 or refractory grade 2, discontinuation of the drug and systemic corticosteroids is recommended.7
The cutaneous eruption in immunotherapy-mediated dermatitis is thought to be largely mediated by activated T cells infiltrating the dermis.8 In localized scleroderma, increased tumor necrosis factor α, IFN-γ, IFN-γ–induced protein 10, and granulocyte macrophage colony stimulating factor activity have been shown to correlate with disease activity.9,10 Interestingly, increased tumor necrosis factor α and IFN-γ correlate with better response and increased overall survival in PD-1 inhibition therapy, suggesting a correlation between PD-1 inhibition and T helper activation as noted by the etiology of sclerosis in our patient.11 Additionally, history of radiation was a confounding factor in the diagnosis of our patient, as both sclerodermoid reactions and chronic radiation dermatitis can present with dermal sclerosis. However, the progression of disease outside of the radiation field excluded this etiology. Although new-onset sclerodermoid reactions have been reported with PD-1 inhibitors, they have been described secondary to sclerodermoid reactions from treatment with pembrolizumab.12,13 One case series reported a case of diffuse sclerodermoid reaction and a limited reaction in response to pembrolizumab treatment, while another case report described a relapse of generalized morphea in response to pembrolizumab treatment.12,13 One case of relapsing morphea in response to nivolumab treatment for stage IV lung adenocarcinoma also has been reported.14
Cutaneous toxicities are one of the most common irAEs associated with checkpoint inhibitors and are seen in more than one-third of treated patients. Most frequently, these irAEs manifest as spongiotic dermatitis on histopathology, but a broad spectrum of cutaneous reactions have been observed.15 Although sclerodermoid reactions have been reported with PD-1 inhibitors, most are described secondary to sclerodermoid reactions with pembrolizumab and involve relapse of previously diagnosed morphea rather than new-onset disease.12-14
Our case highlights new-onset localized scleroderma in the setting of nivolumab therapy that showed clinical improvement with methotrexate and topical and systemic steroids. This reaction pattern should be considered in all patients who develop cutaneous eruptions when treated with a PD-1 inhibitor. There should be a high index of suspicion for the potential occurrence of irAEs to ensure early recognition and treatment to minimize morbidity and maximize adherence to therapy for the underlying malignancy.
To the Editor:
Immune checkpoint inhibitors such as anti–programmed cell death protein 1 (anti–PD-1) and anticytotoxic T lymphocyte–associated protein 4 therapies are a promising class of cancer therapeutics. However, they are associated with a variety of immune-related adverse events (irAEs), including cutaneous toxicity.1 The PD-1/programmed death ligand 1 (PD-L1) pathway is important for the maintenance of immune tolerance, and a blockade has been shown to lead to development of various autoimmune diseases.2 We present the case of a patient who developed new-onset localized scleroderma during treatment with the PD-1 inhibitor nivolumab.
A 65-year-old woman presented with a rash on the left thigh that was associated with pruritus, pain, and a pulling sensation. She had a history of stage IV lung adenocarcinoma, with a mass in the right upper lobe with metastatic foci to the left femur, right humerus, right hilar, and pretracheal lymph nodes. She received palliative radiation to the left femur and was started on carboplatin and pemetrexed. Metastasis to the liver was noted after completion of 6 cycles of therapy, and the patient’s treatment was changed to nivolumab. After 17 months on nivolumab therapy (2 years after initial diagnosis and 20 months after radiation therapy), she presented to our dermatology clinic with a cutaneous eruption on the buttocks that spread to the left thigh. The rash failed to improve after 1 month of treatment with emollients and triamcinolone cream 0.1%.
At the current presentation, which was 2 months after she initially presented to our clinic, dermatologic examination revealed erythematous and sclerotic plaques on the left lateral thigh (Figure 1A). Betamethasone cream 0.05% was prescribed, and nivolumab was discontinued due to progression of cutaneous symptoms. A punch biopsy from the left thigh demonstrated superficial dermal sclerosis that was suggestive of chronic radiation dermatitis; direct immunofluorescence testing was negative. The patient was started on prednisone 50 mg daily, which resulted in mild improvement in symptoms.

Within 6 months, new sclerotic plaques developed on the patient’s back and right thigh (Figure 1B). Because the lesions were located outside the radiation field of the left femur, a second biopsy was obtained from the right thigh. Histopathology revealed extensive dermal sclerosis and a perivascular lymphoplasmacytic infiltrate (Figure 2). An antinuclear antibody test was weakly positive (1:40, nucleolar pattern) with a negative extractable nuclear antigen panel result. Anti–double-stranded DNA, anti–topoisomerase 1, anti-Smith, antiribonucleoprotein, anti–Sjögren syndrome type A, anti–Sjögren syndrome type B, and anticentromere serology test results were negative. The patient denied decreased oral aperture, difficulty swallowing, or Raynaud phenomenon. Due to the atypical clinical presentation in the setting of PD-1 inhibitor therapy, the etiology of the eruption was potentially attributable to nivolumab. She was started on treatment with methotrexate 20 mg weekly and clobetasol cream 0.05% twice daily; she continued taking prednisone 5 mg daily. The cutaneous manifestations on the patient’s back completely resolved, and the legs continued to gradually improve on this regimen. Immunotherapy continued to be held due to skin toxicity.

Localized scleroderma is an autoimmune disorder characterized by inflammation and skin thickening. Overactive fibroblasts produce excess collagen, leading to the clinical symptoms of skin thickening, hardening, and discoloration.3 Lesions frequently develop on the arms, face, or legs and can present as patches or linear bands. Unlike systemic sclerosis, the internal organs typically are uninvolved; however, sclerotic lesions can be disfiguring and cause notable disability if they impede joint movement.
The PD-1/PD-L1 pathway is a negative regulator of the immune response that inactivates T cells and helps maintain self-tolerance. Modulation of the PD-1/PD-L1 pathway and overexpression of PD-L1 are seen in various cancers as a mechanism to help malignant cells avoid immune destruction.4 Conversely, inhibition of this pathway can be used to stimulate an antitumor immune response. This checkpoint inhibition strategy has been highly successful for the treatment of various cancers including melanoma and non–small cell lung carcinoma. There are several checkpoint inhibitors approved in the United States that are used for cancer therapy and target the PD-1/PD-L1 pathway, such as nivolumab, pembrolizumab, atezolizumab, durvalumab, and avelumab.4 A downside of checkpoint inhibitor treatment is that uncontrolled T-cell activation can lead to irAEs, including cutaneous eruptions, pruritus, diarrhea, colitis, hepatitis, endocrinopathies, pneumonitis, and renal insufficiency.5 These toxicities are reversible if treated appropriately but can cause notable morbidity and mortality if left unrecognized. Cutaneous eruption is one of the most common irAEs associated with anti–PD-1 and anti–PD-L1 therapies and can limit therapeutic efficacy, as the drug may need to be held or discontinued due to the severity of the eruption.6 Mid-potency to high-potency topical corticosteroids and systemic antihistamines are first-line treatments of grades 1 and 2 skin toxicities associated with PD-1 inhibitor therapy. For eruptions classified as grades 3 or 4 or refractory grade 2, discontinuation of the drug and systemic corticosteroids is recommended.7
The cutaneous eruption in immunotherapy-mediated dermatitis is thought to be largely mediated by activated T cells infiltrating the dermis.8 In localized scleroderma, increased tumor necrosis factor α, IFN-γ, IFN-γ–induced protein 10, and granulocyte macrophage colony stimulating factor activity have been shown to correlate with disease activity.9,10 Interestingly, increased tumor necrosis factor α and IFN-γ correlate with better response and increased overall survival in PD-1 inhibition therapy, suggesting a correlation between PD-1 inhibition and T helper activation as noted by the etiology of sclerosis in our patient.11 Additionally, history of radiation was a confounding factor in the diagnosis of our patient, as both sclerodermoid reactions and chronic radiation dermatitis can present with dermal sclerosis. However, the progression of disease outside of the radiation field excluded this etiology. Although new-onset sclerodermoid reactions have been reported with PD-1 inhibitors, they have been described secondary to sclerodermoid reactions from treatment with pembrolizumab.12,13 One case series reported a case of diffuse sclerodermoid reaction and a limited reaction in response to pembrolizumab treatment, while another case report described a relapse of generalized morphea in response to pembrolizumab treatment.12,13 One case of relapsing morphea in response to nivolumab treatment for stage IV lung adenocarcinoma also has been reported.14
Cutaneous toxicities are one of the most common irAEs associated with checkpoint inhibitors and are seen in more than one-third of treated patients. Most frequently, these irAEs manifest as spongiotic dermatitis on histopathology, but a broad spectrum of cutaneous reactions have been observed.15 Although sclerodermoid reactions have been reported with PD-1 inhibitors, most are described secondary to sclerodermoid reactions with pembrolizumab and involve relapse of previously diagnosed morphea rather than new-onset disease.12-14
Our case highlights new-onset localized scleroderma in the setting of nivolumab therapy that showed clinical improvement with methotrexate and topical and systemic steroids. This reaction pattern should be considered in all patients who develop cutaneous eruptions when treated with a PD-1 inhibitor. There should be a high index of suspicion for the potential occurrence of irAEs to ensure early recognition and treatment to minimize morbidity and maximize adherence to therapy for the underlying malignancy.
- Baxi S, Yang A, Gennarelli RL, et al. Immune-related adverse events for anti-PD-1 and anti-PD-L1 drugs: systematic review and meta-analysis. BMJ. 2018;360:k793.
- Dai S, Jia R, Zhang X, et al. The PD-1/PD-Ls pathway and autoimmune diseases. Cell Immunol. 2014;290:72-79.
- Badea I, Taylor M, Rosenberg A, et al. Pathogenesis and therapeutic approaches for improved topical treatment in localized scleroderma and systemic sclerosis. Rheumatology (Oxford). 2009;48:213-221.
- Constantinidou A, Alifieris C, Trafalis DT. Targeting programmed cell death-1 (PD-1) and ligand (PD-L1): a new era in cancer active immunotherapy. Pharmacol Ther. 2019;194:84-106.
- Villadolid J, Asim A. Immune checkpoint inhibitors in clinical practice: update on management of immune-related toxicities. Transl Lung Cancer Res. 2015;4:560-575.
- Naidoo J, Page DB, Li BT, et al. Toxicities of the anti-PD-1 and anti-PD-L1 immune checkpoint antibodies. Ann Oncol. 2016;27:1362.
- O’Kane GM, Labbé C, Doherty MK, et al. Monitoring and management of immune-related adverse events associated with programmed cell death protein-1 axis inhibitors in lung cancer. Oncologist. 2017;22:70-80.
- Shi VJ, Rodic N, Gettinger S, et al. Clinical and histologic features of lichenoid mucocutaneous eruptions due to anti-programmed celldeath 1 and anti-programmed cell death ligand 1 immunotherapy. JAMA Dermatol. 2016;152:1128-1136.
- Torok KS, Kurzinski K, Kelsey C, et al. Peripheral blood cytokine and chemokine profiles in juvenile localized scleroderma: T-helper cell-associated cytokine profiles. Semin Arthritis Rheum. 2015;45:284-293.
- Guo X, Higgs BW, Bay-Jensen AC, et al. Suppression of T cell activation and collagen accumulation by an anti-IFNAR1 mAb, anifrolumab, in adult patients with systemic sclerosis. J Invest Dermatol. 2015;135:2402-2409.
- Boutsikou E, Domvri K, Hardavella G, et al. Tumor necrosis factor, interferon-gamma and interleukins as predictive markers of antiprogrammed cell-death protein-1 treatment in advanced non-small cell lung cancer: a pragmatic approach in clinical practice. Ther Adv Med Oncol. 2018;10:1758835918768238.
- Barbosa NS, Wetter DA, Wieland CN, et al. Scleroderma induced by pembrolizumab: a case series. Mayo Clin Proc. 2017;92:1158-1163.
- Cheng MW, Hisaw LD, Bernet L. Generalized morphea in the setting of pembrolizumab. Int J Dermatol. 2019;58:736-738.
- Alegre-Sánchez A, Fonda-Pascual P, Saceda-Corralo D, et al. Relapse of morphea during nivolumab therapy for lung adenocarcinoma. Actas Dermosifiliogr. 2017;108:69-70.
- Sibaud V. Dermatologic reactions to immune checkpoint inhibitors: skin toxicities and immunotherapy. Am J Clin Dermatol. 2018;19:345-361.
- Baxi S, Yang A, Gennarelli RL, et al. Immune-related adverse events for anti-PD-1 and anti-PD-L1 drugs: systematic review and meta-analysis. BMJ. 2018;360:k793.
- Dai S, Jia R, Zhang X, et al. The PD-1/PD-Ls pathway and autoimmune diseases. Cell Immunol. 2014;290:72-79.
- Badea I, Taylor M, Rosenberg A, et al. Pathogenesis and therapeutic approaches for improved topical treatment in localized scleroderma and systemic sclerosis. Rheumatology (Oxford). 2009;48:213-221.
- Constantinidou A, Alifieris C, Trafalis DT. Targeting programmed cell death-1 (PD-1) and ligand (PD-L1): a new era in cancer active immunotherapy. Pharmacol Ther. 2019;194:84-106.
- Villadolid J, Asim A. Immune checkpoint inhibitors in clinical practice: update on management of immune-related toxicities. Transl Lung Cancer Res. 2015;4:560-575.
- Naidoo J, Page DB, Li BT, et al. Toxicities of the anti-PD-1 and anti-PD-L1 immune checkpoint antibodies. Ann Oncol. 2016;27:1362.
- O’Kane GM, Labbé C, Doherty MK, et al. Monitoring and management of immune-related adverse events associated with programmed cell death protein-1 axis inhibitors in lung cancer. Oncologist. 2017;22:70-80.
- Shi VJ, Rodic N, Gettinger S, et al. Clinical and histologic features of lichenoid mucocutaneous eruptions due to anti-programmed celldeath 1 and anti-programmed cell death ligand 1 immunotherapy. JAMA Dermatol. 2016;152:1128-1136.
- Torok KS, Kurzinski K, Kelsey C, et al. Peripheral blood cytokine and chemokine profiles in juvenile localized scleroderma: T-helper cell-associated cytokine profiles. Semin Arthritis Rheum. 2015;45:284-293.
- Guo X, Higgs BW, Bay-Jensen AC, et al. Suppression of T cell activation and collagen accumulation by an anti-IFNAR1 mAb, anifrolumab, in adult patients with systemic sclerosis. J Invest Dermatol. 2015;135:2402-2409.
- Boutsikou E, Domvri K, Hardavella G, et al. Tumor necrosis factor, interferon-gamma and interleukins as predictive markers of antiprogrammed cell-death protein-1 treatment in advanced non-small cell lung cancer: a pragmatic approach in clinical practice. Ther Adv Med Oncol. 2018;10:1758835918768238.
- Barbosa NS, Wetter DA, Wieland CN, et al. Scleroderma induced by pembrolizumab: a case series. Mayo Clin Proc. 2017;92:1158-1163.
- Cheng MW, Hisaw LD, Bernet L. Generalized morphea in the setting of pembrolizumab. Int J Dermatol. 2019;58:736-738.
- Alegre-Sánchez A, Fonda-Pascual P, Saceda-Corralo D, et al. Relapse of morphea during nivolumab therapy for lung adenocarcinoma. Actas Dermosifiliogr. 2017;108:69-70.
- Sibaud V. Dermatologic reactions to immune checkpoint inhibitors: skin toxicities and immunotherapy. Am J Clin Dermatol. 2018;19:345-361.
Practice Points
- Immune checkpoint inhibitors such as nivolumab, a programmed cell death protein 1 (PD-1) inhibitor, are associated with immune-related adverse events (irAEs) such as skin toxicity.
- Scleroderma should be considered in the differential diagnosis of patients who develop cutaneous eruptions during treatment with PD-1 inhibitors.
- To ensure prompt recognition and treatment, health care providers should maintain a high index of suspicion for development of cutaneous irAEs in patients using checkpoint inhibitors.
Transitioning From an Intern to a Dermatology Resident
The transition from medical school to residency is a rewarding milestone but involves a steep learning curve wrought with new responsibilities, new colleagues, and a new schedule, often all within a new setting. This transition period has been a longstanding focus of graduate medical education research, and a recent study identified 6 key areas that residency programs need to address to better facilitate this transition: (1) a sense of community within the residency program, (2) relocation resources, (3) residency preparation courses in medical school, (4) readiness to address racism and bias, (5) connecting with peers, and (6) open communication with program leadership.1 There is considerable interest in ensuring that this transition is smooth for all graduates, as nearly all US medical schools feature some variety of a residency preparation course during the fourth year of medical school, which, alongside the subinternships, serves to better prepare their graduates for the healthcare workforce.2
What about the transition from intern to dermatology resident? Near the end of intern year, my categorical medicine colleagues experienced a crescendo of responsibilities, all in preparation for junior year. The senior medicine residents, themselves having previously experienced the graduated responsibilities, knew to ease their grip on the reins and provide the late spring interns an opportunity to lead rounds or run a code. This was not the case for the preliminary interns for whom there was no preview available for what was to come; little guidance exists on how to best transform from a preliminary or transitional postgraduate year (PGY) 1 to a dermatology PGY-2. A survey of 44 dermatology residents and 33 dermatology program directors found electives such as rheumatology, infectious diseases, and allergy and immunology to be helpful for this transition, and residents most often cited friendly and supportive senior and fellow residents as the factor that eased their transition to PGY-2.3 Notably, less than half of the residents (40%) surveyed stated that team-building exercises and dedicated time to meet colleagues were helpful for this transition. They identified studying principles of dermatologic disease, learning new clinical duties, and adjusting to new coworkers and supervisors as the greatest work-related stressors during entry to PGY-2.3
My transition from intern year to dermatology was shrouded in uncertainty, and I was fortunate to have supportive seniors and co-residents to ease the process. There is much about starting dermatology residency that cannot be prepared for by reading a book, and a natural metamorphosis into the new role is hard to articulate. Still, the following are pieces of information I wish I knew as a graduating intern, which I hope will prove useful for those graduating to their PGY-2 dermatology year.
The Pace of Outpatient Dermatology
If the preliminary or transitional year did not have an ambulatory component, the switch from wards to clinic can be jarring. An outpatient encounter can be as short as 10 to 15 minutes, necessitating an efficient interview and examination to avoid a backup of patients. Unlike a hospital admission where the history of present illness can expound on multiple concerns and organ systems, the general dermatology visit must focus on the chief concern, with priority given to the clinical examination of the skin. For total-body skin examinations, a formulaic approach to assessing all areas of the body, with fluent transitions and minimal repositioning of the patient, is critical for patient comfort and to save time. Of course, accuracy and thoroughness are paramount, but the constant mindfulness of time and efficiency is uniquely emphasized in the outpatient setting.
Continuity of Care
On the wards, patients are admitted with an acute problem and discharged with the aim to prevent re-admission. However, in the dermatology clinic, the conditions encountered often are chronic, requiring repeated follow-ups that involve dosage tapers, laboratory monitoring, and trial and error. Unlike the rigid algorithm-based treatments utilized in the inpatient setting, the management of the same chronic disease can vary, as it is tailored to the patient based on their comorbidities and response. This longitudinal relationship with patients, whereby many disorders are managed rather than treated, stands in stark contrast to inpatient medicine, and learning to value symptom management rather than focusing on a cure is critical in a largely outpatient specialty such as dermatology.
Consulter to Consultant
Calling a consultation as an intern is challenging and requires succinct delivery of pertinent information while fearing pushback from the consultant. In a survey of 50 hospitalist attendings, only 11% responded that interns could be entrusted to call an effective consultation without supervision.4 When undertaking the role of a consultant, the goals should be to identify the team’s main question and to obtain key information necessary to formulate a differential diagnosis. The quality of the consultation will inevitably fluctuate; try to remember what it was like for you as a member of the primary team and remain patient and courteous during the exchange.5 In 1983, Goldman et al6 published a guideline on effective consultations that often is cited to this day, dubbed the “Ten Commandments for Effective Consultations,” which consists of the following: (1) determine the question that is being asked, (2) establish the urgency of the consultation, (3) gather primary data, (4) communicate as briefly as appropriate, (5) make specific recommendations, (6) provide contingency plans, (7) understand your own role in the process, (8) offer educational information, (9) communicate recommendations directly to the requesting physician, and (10) provide appropriate follow-up.
Consider Your Future
Frequently reflect on what you most enjoy about your job. Although it can be easy to passively engage with intern year as a mere stepping-stone to dermatology residency, the years in PGY-2 and onward require active introspection to find a future niche. What made you gravitate to the specialty of dermatology? Try to identify your predilections for dermatopathology, pediatric dermatology, dermatologic surgery, cosmetic dermatology, and academia. Be consistently cognizant of your life after residency, as some fellowships such as dermatopathology require applications to be submitted at the conclusion of the PGY-2 year. Seek out faculty mentors or alumni who are walking a path similar to the one you want to embark on, as the next stop after graduation may be your forever job.
Depth, Not Breadth
The practice of medicine changes when narrowing the focus to one organ system. In both medical school and intern year, my study habits and history-taking of patients cast a wide net across multiple organ systems, aiming to know just enough about any one specialty to address all chief concerns and to know when it was appropriate to consult a specialist. This paradigm inevitably shifts in dermatology residency, as residents are tasked with memorizing the endless number of diagnoses of the skin alone, comprehending the many shades of “erythematous,” including pink, salmon, red, and purple. Both on the wards and in clinics, I had to grow comfortable with telling patients that I did not have an answer for many of their nondermatologic concerns and directing them to the right specialist. As medicine continues trending to specialization, subspecialization, and sub-subspecialization, the scope of any given physician likely will continue to narrow,7 as evidenced by specialty clinics within dermatology such as those focusing on hair loss or immunobullous disease. In this health care system, it is imperative to remember that you are only one physician within a team of care providers—understand your own role in the process and become comfortable with not having the answer to all the questions.
Final Thoughts
In a study of 44 dermatology residents, 35 (83%) indicated zero to less than 1 hour per week of independent preparation for dermatology residency during PGY-1.3 Although the usefulness of preparing is debatable, this figure likely reflects the absence of any insight on how to best prepare for the transition. Recognizing the many contrasts between internal medicine and dermatology and embracing the changes will enable a seamless promotion from a medicine PGY-1 to a dermatology PGY-2.
- Staples H, Frank S, Mullen M, et al. Improving the medical school to residency transition: narrative experiences from first-year residents.J Surg Educ. 2022;S1931-7204(22)00146-5. doi:10.1016/j.jsurg.2022.06.001
- Heidemann LA, Walford E, Mack J, et al. Is there a role for internal medicine residency preparation courses in the fourth year curriculum? a single-center experience. J Gen Intern Med. 2018;33:2048-2050.
- Hopkins C, Jalali O, Guffey D, et al. A survey of dermatology residents and program directors assessing the transition to dermatology residency. Proc (Bayl Univ Med Cent). 2020;34:59-62.
- Marcus CH, Winn AS, Sectish TC, et al. How much supervision is required is the beginning of intern year? Acad Pediatr. 2016;16:E3-E4.
- Bly RA, Bly EG. Consult courtesy. J Grad Med Educ. 2013;5:533-534.
- Goldman L, Lee T, Rudd P. Ten commandments for effective consultations. Arch Intern Med. 1983;143:1753-1755.
- Oren O, Gersh BJ, Bhatt DL. On the pearls and perils of sub-subspecialization. Am J Med. 2020;133:158-159.
The transition from medical school to residency is a rewarding milestone but involves a steep learning curve wrought with new responsibilities, new colleagues, and a new schedule, often all within a new setting. This transition period has been a longstanding focus of graduate medical education research, and a recent study identified 6 key areas that residency programs need to address to better facilitate this transition: (1) a sense of community within the residency program, (2) relocation resources, (3) residency preparation courses in medical school, (4) readiness to address racism and bias, (5) connecting with peers, and (6) open communication with program leadership.1 There is considerable interest in ensuring that this transition is smooth for all graduates, as nearly all US medical schools feature some variety of a residency preparation course during the fourth year of medical school, which, alongside the subinternships, serves to better prepare their graduates for the healthcare workforce.2
What about the transition from intern to dermatology resident? Near the end of intern year, my categorical medicine colleagues experienced a crescendo of responsibilities, all in preparation for junior year. The senior medicine residents, themselves having previously experienced the graduated responsibilities, knew to ease their grip on the reins and provide the late spring interns an opportunity to lead rounds or run a code. This was not the case for the preliminary interns for whom there was no preview available for what was to come; little guidance exists on how to best transform from a preliminary or transitional postgraduate year (PGY) 1 to a dermatology PGY-2. A survey of 44 dermatology residents and 33 dermatology program directors found electives such as rheumatology, infectious diseases, and allergy and immunology to be helpful for this transition, and residents most often cited friendly and supportive senior and fellow residents as the factor that eased their transition to PGY-2.3 Notably, less than half of the residents (40%) surveyed stated that team-building exercises and dedicated time to meet colleagues were helpful for this transition. They identified studying principles of dermatologic disease, learning new clinical duties, and adjusting to new coworkers and supervisors as the greatest work-related stressors during entry to PGY-2.3
My transition from intern year to dermatology was shrouded in uncertainty, and I was fortunate to have supportive seniors and co-residents to ease the process. There is much about starting dermatology residency that cannot be prepared for by reading a book, and a natural metamorphosis into the new role is hard to articulate. Still, the following are pieces of information I wish I knew as a graduating intern, which I hope will prove useful for those graduating to their PGY-2 dermatology year.
The Pace of Outpatient Dermatology
If the preliminary or transitional year did not have an ambulatory component, the switch from wards to clinic can be jarring. An outpatient encounter can be as short as 10 to 15 minutes, necessitating an efficient interview and examination to avoid a backup of patients. Unlike a hospital admission where the history of present illness can expound on multiple concerns and organ systems, the general dermatology visit must focus on the chief concern, with priority given to the clinical examination of the skin. For total-body skin examinations, a formulaic approach to assessing all areas of the body, with fluent transitions and minimal repositioning of the patient, is critical for patient comfort and to save time. Of course, accuracy and thoroughness are paramount, but the constant mindfulness of time and efficiency is uniquely emphasized in the outpatient setting.
Continuity of Care
On the wards, patients are admitted with an acute problem and discharged with the aim to prevent re-admission. However, in the dermatology clinic, the conditions encountered often are chronic, requiring repeated follow-ups that involve dosage tapers, laboratory monitoring, and trial and error. Unlike the rigid algorithm-based treatments utilized in the inpatient setting, the management of the same chronic disease can vary, as it is tailored to the patient based on their comorbidities and response. This longitudinal relationship with patients, whereby many disorders are managed rather than treated, stands in stark contrast to inpatient medicine, and learning to value symptom management rather than focusing on a cure is critical in a largely outpatient specialty such as dermatology.
Consulter to Consultant
Calling a consultation as an intern is challenging and requires succinct delivery of pertinent information while fearing pushback from the consultant. In a survey of 50 hospitalist attendings, only 11% responded that interns could be entrusted to call an effective consultation without supervision.4 When undertaking the role of a consultant, the goals should be to identify the team’s main question and to obtain key information necessary to formulate a differential diagnosis. The quality of the consultation will inevitably fluctuate; try to remember what it was like for you as a member of the primary team and remain patient and courteous during the exchange.5 In 1983, Goldman et al6 published a guideline on effective consultations that often is cited to this day, dubbed the “Ten Commandments for Effective Consultations,” which consists of the following: (1) determine the question that is being asked, (2) establish the urgency of the consultation, (3) gather primary data, (4) communicate as briefly as appropriate, (5) make specific recommendations, (6) provide contingency plans, (7) understand your own role in the process, (8) offer educational information, (9) communicate recommendations directly to the requesting physician, and (10) provide appropriate follow-up.
Consider Your Future
Frequently reflect on what you most enjoy about your job. Although it can be easy to passively engage with intern year as a mere stepping-stone to dermatology residency, the years in PGY-2 and onward require active introspection to find a future niche. What made you gravitate to the specialty of dermatology? Try to identify your predilections for dermatopathology, pediatric dermatology, dermatologic surgery, cosmetic dermatology, and academia. Be consistently cognizant of your life after residency, as some fellowships such as dermatopathology require applications to be submitted at the conclusion of the PGY-2 year. Seek out faculty mentors or alumni who are walking a path similar to the one you want to embark on, as the next stop after graduation may be your forever job.
Depth, Not Breadth
The practice of medicine changes when narrowing the focus to one organ system. In both medical school and intern year, my study habits and history-taking of patients cast a wide net across multiple organ systems, aiming to know just enough about any one specialty to address all chief concerns and to know when it was appropriate to consult a specialist. This paradigm inevitably shifts in dermatology residency, as residents are tasked with memorizing the endless number of diagnoses of the skin alone, comprehending the many shades of “erythematous,” including pink, salmon, red, and purple. Both on the wards and in clinics, I had to grow comfortable with telling patients that I did not have an answer for many of their nondermatologic concerns and directing them to the right specialist. As medicine continues trending to specialization, subspecialization, and sub-subspecialization, the scope of any given physician likely will continue to narrow,7 as evidenced by specialty clinics within dermatology such as those focusing on hair loss or immunobullous disease. In this health care system, it is imperative to remember that you are only one physician within a team of care providers—understand your own role in the process and become comfortable with not having the answer to all the questions.
Final Thoughts
In a study of 44 dermatology residents, 35 (83%) indicated zero to less than 1 hour per week of independent preparation for dermatology residency during PGY-1.3 Although the usefulness of preparing is debatable, this figure likely reflects the absence of any insight on how to best prepare for the transition. Recognizing the many contrasts between internal medicine and dermatology and embracing the changes will enable a seamless promotion from a medicine PGY-1 to a dermatology PGY-2.
The transition from medical school to residency is a rewarding milestone but involves a steep learning curve wrought with new responsibilities, new colleagues, and a new schedule, often all within a new setting. This transition period has been a longstanding focus of graduate medical education research, and a recent study identified 6 key areas that residency programs need to address to better facilitate this transition: (1) a sense of community within the residency program, (2) relocation resources, (3) residency preparation courses in medical school, (4) readiness to address racism and bias, (5) connecting with peers, and (6) open communication with program leadership.1 There is considerable interest in ensuring that this transition is smooth for all graduates, as nearly all US medical schools feature some variety of a residency preparation course during the fourth year of medical school, which, alongside the subinternships, serves to better prepare their graduates for the healthcare workforce.2
What about the transition from intern to dermatology resident? Near the end of intern year, my categorical medicine colleagues experienced a crescendo of responsibilities, all in preparation for junior year. The senior medicine residents, themselves having previously experienced the graduated responsibilities, knew to ease their grip on the reins and provide the late spring interns an opportunity to lead rounds or run a code. This was not the case for the preliminary interns for whom there was no preview available for what was to come; little guidance exists on how to best transform from a preliminary or transitional postgraduate year (PGY) 1 to a dermatology PGY-2. A survey of 44 dermatology residents and 33 dermatology program directors found electives such as rheumatology, infectious diseases, and allergy and immunology to be helpful for this transition, and residents most often cited friendly and supportive senior and fellow residents as the factor that eased their transition to PGY-2.3 Notably, less than half of the residents (40%) surveyed stated that team-building exercises and dedicated time to meet colleagues were helpful for this transition. They identified studying principles of dermatologic disease, learning new clinical duties, and adjusting to new coworkers and supervisors as the greatest work-related stressors during entry to PGY-2.3
My transition from intern year to dermatology was shrouded in uncertainty, and I was fortunate to have supportive seniors and co-residents to ease the process. There is much about starting dermatology residency that cannot be prepared for by reading a book, and a natural metamorphosis into the new role is hard to articulate. Still, the following are pieces of information I wish I knew as a graduating intern, which I hope will prove useful for those graduating to their PGY-2 dermatology year.
The Pace of Outpatient Dermatology
If the preliminary or transitional year did not have an ambulatory component, the switch from wards to clinic can be jarring. An outpatient encounter can be as short as 10 to 15 minutes, necessitating an efficient interview and examination to avoid a backup of patients. Unlike a hospital admission where the history of present illness can expound on multiple concerns and organ systems, the general dermatology visit must focus on the chief concern, with priority given to the clinical examination of the skin. For total-body skin examinations, a formulaic approach to assessing all areas of the body, with fluent transitions and minimal repositioning of the patient, is critical for patient comfort and to save time. Of course, accuracy and thoroughness are paramount, but the constant mindfulness of time and efficiency is uniquely emphasized in the outpatient setting.
Continuity of Care
On the wards, patients are admitted with an acute problem and discharged with the aim to prevent re-admission. However, in the dermatology clinic, the conditions encountered often are chronic, requiring repeated follow-ups that involve dosage tapers, laboratory monitoring, and trial and error. Unlike the rigid algorithm-based treatments utilized in the inpatient setting, the management of the same chronic disease can vary, as it is tailored to the patient based on their comorbidities and response. This longitudinal relationship with patients, whereby many disorders are managed rather than treated, stands in stark contrast to inpatient medicine, and learning to value symptom management rather than focusing on a cure is critical in a largely outpatient specialty such as dermatology.
Consulter to Consultant
Calling a consultation as an intern is challenging and requires succinct delivery of pertinent information while fearing pushback from the consultant. In a survey of 50 hospitalist attendings, only 11% responded that interns could be entrusted to call an effective consultation without supervision.4 When undertaking the role of a consultant, the goals should be to identify the team’s main question and to obtain key information necessary to formulate a differential diagnosis. The quality of the consultation will inevitably fluctuate; try to remember what it was like for you as a member of the primary team and remain patient and courteous during the exchange.5 In 1983, Goldman et al6 published a guideline on effective consultations that often is cited to this day, dubbed the “Ten Commandments for Effective Consultations,” which consists of the following: (1) determine the question that is being asked, (2) establish the urgency of the consultation, (3) gather primary data, (4) communicate as briefly as appropriate, (5) make specific recommendations, (6) provide contingency plans, (7) understand your own role in the process, (8) offer educational information, (9) communicate recommendations directly to the requesting physician, and (10) provide appropriate follow-up.
Consider Your Future
Frequently reflect on what you most enjoy about your job. Although it can be easy to passively engage with intern year as a mere stepping-stone to dermatology residency, the years in PGY-2 and onward require active introspection to find a future niche. What made you gravitate to the specialty of dermatology? Try to identify your predilections for dermatopathology, pediatric dermatology, dermatologic surgery, cosmetic dermatology, and academia. Be consistently cognizant of your life after residency, as some fellowships such as dermatopathology require applications to be submitted at the conclusion of the PGY-2 year. Seek out faculty mentors or alumni who are walking a path similar to the one you want to embark on, as the next stop after graduation may be your forever job.
Depth, Not Breadth
The practice of medicine changes when narrowing the focus to one organ system. In both medical school and intern year, my study habits and history-taking of patients cast a wide net across multiple organ systems, aiming to know just enough about any one specialty to address all chief concerns and to know when it was appropriate to consult a specialist. This paradigm inevitably shifts in dermatology residency, as residents are tasked with memorizing the endless number of diagnoses of the skin alone, comprehending the many shades of “erythematous,” including pink, salmon, red, and purple. Both on the wards and in clinics, I had to grow comfortable with telling patients that I did not have an answer for many of their nondermatologic concerns and directing them to the right specialist. As medicine continues trending to specialization, subspecialization, and sub-subspecialization, the scope of any given physician likely will continue to narrow,7 as evidenced by specialty clinics within dermatology such as those focusing on hair loss or immunobullous disease. In this health care system, it is imperative to remember that you are only one physician within a team of care providers—understand your own role in the process and become comfortable with not having the answer to all the questions.
Final Thoughts
In a study of 44 dermatology residents, 35 (83%) indicated zero to less than 1 hour per week of independent preparation for dermatology residency during PGY-1.3 Although the usefulness of preparing is debatable, this figure likely reflects the absence of any insight on how to best prepare for the transition. Recognizing the many contrasts between internal medicine and dermatology and embracing the changes will enable a seamless promotion from a medicine PGY-1 to a dermatology PGY-2.
- Staples H, Frank S, Mullen M, et al. Improving the medical school to residency transition: narrative experiences from first-year residents.J Surg Educ. 2022;S1931-7204(22)00146-5. doi:10.1016/j.jsurg.2022.06.001
- Heidemann LA, Walford E, Mack J, et al. Is there a role for internal medicine residency preparation courses in the fourth year curriculum? a single-center experience. J Gen Intern Med. 2018;33:2048-2050.
- Hopkins C, Jalali O, Guffey D, et al. A survey of dermatology residents and program directors assessing the transition to dermatology residency. Proc (Bayl Univ Med Cent). 2020;34:59-62.
- Marcus CH, Winn AS, Sectish TC, et al. How much supervision is required is the beginning of intern year? Acad Pediatr. 2016;16:E3-E4.
- Bly RA, Bly EG. Consult courtesy. J Grad Med Educ. 2013;5:533-534.
- Goldman L, Lee T, Rudd P. Ten commandments for effective consultations. Arch Intern Med. 1983;143:1753-1755.
- Oren O, Gersh BJ, Bhatt DL. On the pearls and perils of sub-subspecialization. Am J Med. 2020;133:158-159.
- Staples H, Frank S, Mullen M, et al. Improving the medical school to residency transition: narrative experiences from first-year residents.J Surg Educ. 2022;S1931-7204(22)00146-5. doi:10.1016/j.jsurg.2022.06.001
- Heidemann LA, Walford E, Mack J, et al. Is there a role for internal medicine residency preparation courses in the fourth year curriculum? a single-center experience. J Gen Intern Med. 2018;33:2048-2050.
- Hopkins C, Jalali O, Guffey D, et al. A survey of dermatology residents and program directors assessing the transition to dermatology residency. Proc (Bayl Univ Med Cent). 2020;34:59-62.
- Marcus CH, Winn AS, Sectish TC, et al. How much supervision is required is the beginning of intern year? Acad Pediatr. 2016;16:E3-E4.
- Bly RA, Bly EG. Consult courtesy. J Grad Med Educ. 2013;5:533-534.
- Goldman L, Lee T, Rudd P. Ten commandments for effective consultations. Arch Intern Med. 1983;143:1753-1755.
- Oren O, Gersh BJ, Bhatt DL. On the pearls and perils of sub-subspecialization. Am J Med. 2020;133:158-159.
Resident Pearl
- There is surprisingly little information on what to expect when transitioning from intern year to dermatology residency. Recognizing the unique aspects of a largely outpatient specialty and embracing the role of a specialist will help facilitate this transition.
Ossification and Migration of a Nodule Following Calcium Hydroxylapatite Injection
To the Editor:
Calcium hydroxylapatite is an injectable filler approved by the US Food and Drug Administration for moderate to severe rhytides of the face and the treatment of facial lipodystrophy in patients with HIV.1 This long-lasting filler generally is well tolerated with minimal side effects; however, there have been reports of nodules or granulomatous formation following injection.2 We present a case of a migrating nodule following injection of a calcium hydroxylapatite filler that appeared ossified on radiographic imaging. We highlight this rarely reported phenomenon to increase awareness of this complication.
A 72-year-old woman presented to our clinic with a mass on the left cheek. The patient had a history of treatment with facial fillers but no notable medical conditions. She initially received hyaluronic acid injectable gel dermal filler twice—3 years apart—before switching to calcium hydroxylapatite injections twice—4 months apart—from an outside provider. One month after the second treatment, she noticed a mass on the left cheek and promptly returned to the provider who performed the calcium hydroxylapatite injections. The provider, who had originally injected in the infraorbital area, stated it was unlikely that the filler would have migrated to the mid cheek and referred the patient to a general dentist who suspected salivary gland pathology. The patient was referred to an oral and maxillofacial surgeon who suspected the mass was related to the parotid gland. Maxillofacial computed tomography (CT) revealed heterotopic ossification vs myositis ossificans, possibly related to the recent injection. The patient was eventually referred to the Division of Plastic Surgery, Department of Surgery, at the University of Texas Medical Branch (Galveston, Texas) for further evaluation. Physical examination revealed a 2×1-cm firm, mobile, nontender mass in the left cheek in the area of the buccinator muscles. The mass did not express any fluid and was most easily palpable from the oral cavity. Radiography findings showed that the calcium hydroxylapatite filler had migrated to this location and formed a nodule (Figure). Because calcium hydroxylapatite fillers generally last 12 to 18 months, we opted to observe the lesion for spontaneous resolution. Four months later, the patient presented to our clinic for follow-up and the mass had reduced in size and appeared to be spontaneously resolving.
We present a unique case of a migrating nodule that occurred after injection with calcium hydroxylapatite, which led to concern for neoplastic tumor formation. This complication is rare, and it is important for practitioners who inject calcium hydroxylapatite as well as those who these patients may be referred to for evaluation to be aware that migrating nodules can occur. This awareness can help reduce unnecessary referrals, medical procedures, and anxiety.
Calcium hydroxylapatite filler is composed of 30% calcium hydroxylapatite microspheres suspended in a 70% sodium carboxymethylcellulose gel. The water-soluble gel rapidly becomes absorbed upon injection; however, the microspheres form a scaffold for the production of newly synthesized collagen. The filling effect generally lasts 12 to 18 months.1
Calcium hydroxylapatite, similar to most fillers, generally is well tolerated with a low complication rate of 3%.1 Although nodule formation with calcium hydroxylapatite is rare, it is the most common adverse event and encompasses 96% of complications. The remaining 4% of complications include persistent inflammation, swelling, erythema, and technical mistakes leading to overcorrection.1 Migrating nodules are rare; however, Beer3 reported a similar case.
Treatment of calcium hydroxylapatite nodules depends on differentiating a cause based on the time of onset. Early nodules that occur within 1 to 2 weeks of the injection usually represent incorrect positioning of the filler and can be treated by massaging the nodule. Other more invasive techniques involve aspiration or injection of sterile water. Late-onset nodules have shown response to corticosteroid injections. For inflammatory nodules of infectious origin, antibiotics can be useful. Surgical excision of the nodule rarely is required, as most nodules will resolve spontaneously, even without intervention.1,2
Radiologic findings of calcium hydroxylapatite appear as high-attenuation linear streaks or masses on CT (280–700 HU) and as low to intermediate signal intensity on T1- or T2-weighted sequences on magnetic resonance imaging. Oftentimes, calcium hydroxylapatite has a similar radiographic appearance to bone and can persist for 2 years or more on radiographic imaging, longer than they are clinically visible.4 The nodule formation from injection with calcium hydroxylapatite can mimic pathologic conditions such as miliary osteomas, myositis ossificans, heterotrophic/dystrophic calcifications, and foreign bodies on CT. Our patient’s CT findings of high attenuation linear streaks and nodules of similar signal intensity to bone were consistent with those previously described in the radiographic literature.
Calcium hydroxylapatite fillers have a good safety profile, but it is important to recognize that nodule formation is a common adverse event and that migration of nodules can occur. Practitioners should recognize this possibility in patients presenting with new masses after filler injection before advocating for potentially invasive and costly procedures and diagnostic modalities.
- Kadouch JA. Calcium hydroxylapatite: a review on safety and complications. J Cosmet Dermatol. 2017;16:152-161.
- Moulinets I, Arnaud E, Bui P, et al. Foreign body reaction to Radiesse: 2 cases. Am J Dermatopathol. 2013;35:e37-40.
- Beer KR. Radiesse nodule of the lips from a distant injection site: report of a case and consideration of etiology and management. J Drugs Dermatol. 2007;6:846-847.
- Ginat DT, Schatz CJ. Imaging features of midface injectable fillers and associated complications. AJNR Am J Neuroradiol. 2013;34:1488-1495.
To the Editor:
Calcium hydroxylapatite is an injectable filler approved by the US Food and Drug Administration for moderate to severe rhytides of the face and the treatment of facial lipodystrophy in patients with HIV.1 This long-lasting filler generally is well tolerated with minimal side effects; however, there have been reports of nodules or granulomatous formation following injection.2 We present a case of a migrating nodule following injection of a calcium hydroxylapatite filler that appeared ossified on radiographic imaging. We highlight this rarely reported phenomenon to increase awareness of this complication.
A 72-year-old woman presented to our clinic with a mass on the left cheek. The patient had a history of treatment with facial fillers but no notable medical conditions. She initially received hyaluronic acid injectable gel dermal filler twice—3 years apart—before switching to calcium hydroxylapatite injections twice—4 months apart—from an outside provider. One month after the second treatment, she noticed a mass on the left cheek and promptly returned to the provider who performed the calcium hydroxylapatite injections. The provider, who had originally injected in the infraorbital area, stated it was unlikely that the filler would have migrated to the mid cheek and referred the patient to a general dentist who suspected salivary gland pathology. The patient was referred to an oral and maxillofacial surgeon who suspected the mass was related to the parotid gland. Maxillofacial computed tomography (CT) revealed heterotopic ossification vs myositis ossificans, possibly related to the recent injection. The patient was eventually referred to the Division of Plastic Surgery, Department of Surgery, at the University of Texas Medical Branch (Galveston, Texas) for further evaluation. Physical examination revealed a 2×1-cm firm, mobile, nontender mass in the left cheek in the area of the buccinator muscles. The mass did not express any fluid and was most easily palpable from the oral cavity. Radiography findings showed that the calcium hydroxylapatite filler had migrated to this location and formed a nodule (Figure). Because calcium hydroxylapatite fillers generally last 12 to 18 months, we opted to observe the lesion for spontaneous resolution. Four months later, the patient presented to our clinic for follow-up and the mass had reduced in size and appeared to be spontaneously resolving.

We present a unique case of a migrating nodule that occurred after injection with calcium hydroxylapatite, which led to concern for neoplastic tumor formation. This complication is rare, and it is important for practitioners who inject calcium hydroxylapatite as well as those who these patients may be referred to for evaluation to be aware that migrating nodules can occur. This awareness can help reduce unnecessary referrals, medical procedures, and anxiety.
Calcium hydroxylapatite filler is composed of 30% calcium hydroxylapatite microspheres suspended in a 70% sodium carboxymethylcellulose gel. The water-soluble gel rapidly becomes absorbed upon injection; however, the microspheres form a scaffold for the production of newly synthesized collagen. The filling effect generally lasts 12 to 18 months.1
Calcium hydroxylapatite, similar to most fillers, generally is well tolerated with a low complication rate of 3%.1 Although nodule formation with calcium hydroxylapatite is rare, it is the most common adverse event and encompasses 96% of complications. The remaining 4% of complications include persistent inflammation, swelling, erythema, and technical mistakes leading to overcorrection.1 Migrating nodules are rare; however, Beer3 reported a similar case.
Treatment of calcium hydroxylapatite nodules depends on differentiating a cause based on the time of onset. Early nodules that occur within 1 to 2 weeks of the injection usually represent incorrect positioning of the filler and can be treated by massaging the nodule. Other more invasive techniques involve aspiration or injection of sterile water. Late-onset nodules have shown response to corticosteroid injections. For inflammatory nodules of infectious origin, antibiotics can be useful. Surgical excision of the nodule rarely is required, as most nodules will resolve spontaneously, even without intervention.1,2
Radiologic findings of calcium hydroxylapatite appear as high-attenuation linear streaks or masses on CT (280–700 HU) and as low to intermediate signal intensity on T1- or T2-weighted sequences on magnetic resonance imaging. Oftentimes, calcium hydroxylapatite has a similar radiographic appearance to bone and can persist for 2 years or more on radiographic imaging, longer than they are clinically visible.4 The nodule formation from injection with calcium hydroxylapatite can mimic pathologic conditions such as miliary osteomas, myositis ossificans, heterotrophic/dystrophic calcifications, and foreign bodies on CT. Our patient’s CT findings of high attenuation linear streaks and nodules of similar signal intensity to bone were consistent with those previously described in the radiographic literature.
Calcium hydroxylapatite fillers have a good safety profile, but it is important to recognize that nodule formation is a common adverse event and that migration of nodules can occur. Practitioners should recognize this possibility in patients presenting with new masses after filler injection before advocating for potentially invasive and costly procedures and diagnostic modalities.
To the Editor:
Calcium hydroxylapatite is an injectable filler approved by the US Food and Drug Administration for moderate to severe rhytides of the face and the treatment of facial lipodystrophy in patients with HIV.1 This long-lasting filler generally is well tolerated with minimal side effects; however, there have been reports of nodules or granulomatous formation following injection.2 We present a case of a migrating nodule following injection of a calcium hydroxylapatite filler that appeared ossified on radiographic imaging. We highlight this rarely reported phenomenon to increase awareness of this complication.
A 72-year-old woman presented to our clinic with a mass on the left cheek. The patient had a history of treatment with facial fillers but no notable medical conditions. She initially received hyaluronic acid injectable gel dermal filler twice—3 years apart—before switching to calcium hydroxylapatite injections twice—4 months apart—from an outside provider. One month after the second treatment, she noticed a mass on the left cheek and promptly returned to the provider who performed the calcium hydroxylapatite injections. The provider, who had originally injected in the infraorbital area, stated it was unlikely that the filler would have migrated to the mid cheek and referred the patient to a general dentist who suspected salivary gland pathology. The patient was referred to an oral and maxillofacial surgeon who suspected the mass was related to the parotid gland. Maxillofacial computed tomography (CT) revealed heterotopic ossification vs myositis ossificans, possibly related to the recent injection. The patient was eventually referred to the Division of Plastic Surgery, Department of Surgery, at the University of Texas Medical Branch (Galveston, Texas) for further evaluation. Physical examination revealed a 2×1-cm firm, mobile, nontender mass in the left cheek in the area of the buccinator muscles. The mass did not express any fluid and was most easily palpable from the oral cavity. Radiography findings showed that the calcium hydroxylapatite filler had migrated to this location and formed a nodule (Figure). Because calcium hydroxylapatite fillers generally last 12 to 18 months, we opted to observe the lesion for spontaneous resolution. Four months later, the patient presented to our clinic for follow-up and the mass had reduced in size and appeared to be spontaneously resolving.

We present a unique case of a migrating nodule that occurred after injection with calcium hydroxylapatite, which led to concern for neoplastic tumor formation. This complication is rare, and it is important for practitioners who inject calcium hydroxylapatite as well as those who these patients may be referred to for evaluation to be aware that migrating nodules can occur. This awareness can help reduce unnecessary referrals, medical procedures, and anxiety.
Calcium hydroxylapatite filler is composed of 30% calcium hydroxylapatite microspheres suspended in a 70% sodium carboxymethylcellulose gel. The water-soluble gel rapidly becomes absorbed upon injection; however, the microspheres form a scaffold for the production of newly synthesized collagen. The filling effect generally lasts 12 to 18 months.1
Calcium hydroxylapatite, similar to most fillers, generally is well tolerated with a low complication rate of 3%.1 Although nodule formation with calcium hydroxylapatite is rare, it is the most common adverse event and encompasses 96% of complications. The remaining 4% of complications include persistent inflammation, swelling, erythema, and technical mistakes leading to overcorrection.1 Migrating nodules are rare; however, Beer3 reported a similar case.
Treatment of calcium hydroxylapatite nodules depends on differentiating a cause based on the time of onset. Early nodules that occur within 1 to 2 weeks of the injection usually represent incorrect positioning of the filler and can be treated by massaging the nodule. Other more invasive techniques involve aspiration or injection of sterile water. Late-onset nodules have shown response to corticosteroid injections. For inflammatory nodules of infectious origin, antibiotics can be useful. Surgical excision of the nodule rarely is required, as most nodules will resolve spontaneously, even without intervention.1,2
Radiologic findings of calcium hydroxylapatite appear as high-attenuation linear streaks or masses on CT (280–700 HU) and as low to intermediate signal intensity on T1- or T2-weighted sequences on magnetic resonance imaging. Oftentimes, calcium hydroxylapatite has a similar radiographic appearance to bone and can persist for 2 years or more on radiographic imaging, longer than they are clinically visible.4 The nodule formation from injection with calcium hydroxylapatite can mimic pathologic conditions such as miliary osteomas, myositis ossificans, heterotrophic/dystrophic calcifications, and foreign bodies on CT. Our patient’s CT findings of high attenuation linear streaks and nodules of similar signal intensity to bone were consistent with those previously described in the radiographic literature.
Calcium hydroxylapatite fillers have a good safety profile, but it is important to recognize that nodule formation is a common adverse event and that migration of nodules can occur. Practitioners should recognize this possibility in patients presenting with new masses after filler injection before advocating for potentially invasive and costly procedures and diagnostic modalities.
- Kadouch JA. Calcium hydroxylapatite: a review on safety and complications. J Cosmet Dermatol. 2017;16:152-161.
- Moulinets I, Arnaud E, Bui P, et al. Foreign body reaction to Radiesse: 2 cases. Am J Dermatopathol. 2013;35:e37-40.
- Beer KR. Radiesse nodule of the lips from a distant injection site: report of a case and consideration of etiology and management. J Drugs Dermatol. 2007;6:846-847.
- Ginat DT, Schatz CJ. Imaging features of midface injectable fillers and associated complications. AJNR Am J Neuroradiol. 2013;34:1488-1495.
- Kadouch JA. Calcium hydroxylapatite: a review on safety and complications. J Cosmet Dermatol. 2017;16:152-161.
- Moulinets I, Arnaud E, Bui P, et al. Foreign body reaction to Radiesse: 2 cases. Am J Dermatopathol. 2013;35:e37-40.
- Beer KR. Radiesse nodule of the lips from a distant injection site: report of a case and consideration of etiology and management. J Drugs Dermatol. 2007;6:846-847.
- Ginat DT, Schatz CJ. Imaging features of midface injectable fillers and associated complications. AJNR Am J Neuroradiol. 2013;34:1488-1495.
Practice Points
- Calcium hydroxylapatite filler can migrate and form nodules in distant locations from the original injection site.
- Practitioners of calcium hydroxylapatite fillers should be aware of the potential for nodule migration to avoid costly, time-consuming, and invasive referrals and procedures.
An infant with a tender bump on her ear
A biopsy of the lesion was performed that showed a well-defined nodulocystic tumor composed of nests of basaloid cells that are undergoing trichilemmal keratinization. Shadow cells are seen as well as small areas of calcification. There is also a histiocytic infiltrate with multinucleated giant cells. The histologic diagnosis is of a pilomatrixoma.
Pilomatrixoma, also known as calcifying epithelioma of Malherbe, was first described in 1880, as a tumor of sebaceous gland origin. Later, in 1961, Robert Forbis Jr, MD, and Elson B. Helwig, MD, coined the term pilomatrixoma to describe the hair follicle matrix as the source of the tumor. Pilomatrixomas are commonly seen in the pediatric population, usually in children between 8 and 13 years of age. Our patient is one of the youngest described. The lesions are commonly seen on the face and neck in about 70% of the cases followed by the upper extremities, back, and legs. Clinically, the lesions appear as a firm dermal papule or nodule, which moves freely and may have associated erythema on the skin surface or a blueish gray hue on the underlying skin.
Most pilomatrixomas that have been studied have shown a mutation in Exon 3 of the beta-catenin gene (CTNNB1). The beta-catenin molecule is a subunit of the cadherin protein, which is part of an important pathway in the terminal hair follicle differentiation. Beta-catenin also plays an important role in the Wnt pathway, which regulates cell fate as well as early embryonic patterning. Beta-catenin is responsible for forming adhesion junctions among cells. There have also been immunohistochemical studies that have shown a BCL2 proto-oncogene overexpression to pilomatrixoma.
There are several genetic syndromes that have been associated with the presence of pilomatrixomas: Turner syndrome (XO chromosome abnormality associated with short stature and cardiac defects), Gardner syndrome (polyposis coli and colon and rectal cancer), myotonic dystrophy, Rubinstein-Taybi syndrome (characterized by broad thumbs and toes, short stature, distinctive facial features, and varying degrees of intellectual disability), and trisomy 9. On physical examination our patient didn’t present with any of the typical features or history that could suggest any of these syndromes. A close follow-up and evaluation by a geneticist was recommended because after the initial visit she developed a second lesion on the forehead.
The differential diagnosis for this lesion includes other cysts that may occur on the ear such as epidermal inclusion cyst or dermoid cysts, though these lesions do not tend to be as firm as pilomatrixomas are, which can help with the diagnosis. Dermoid cysts are made of dermal and epidermal components. They are usually present at birth and are commonly seen on the scalp and the periorbital face.
Keloids are rubbery nodules of scar tissue that can form on sites of trauma, and although the lesion occurred after she had her ears pierced, the consistency and rapid growth of the lesion as well as the pathological description made this benign fibrous growth less likely.
When pilomatrixomas are inflamed they can be confused with vascular growths: in this particular case, a hemangioma or another vascular tumor such as a tufted angioma or kaposiform hemangioendothelioma. An ultrasound of the lesion could have helped in the differential diagnosis of the lesion.
Pilomatrixomas can grow significantly and in some cases get inflamed or infected. Surgical management of pilomatrixomas is often required because the lesions do not regress spontaneously.
Dr. Matiz is a pediatric dermatologist at Southern California Permanente Medical Group, San Diego.
References
Forbis R Jr and Helwig EB. Arch Dermatol 1961;83:606-18.
Schwarz Y et al. Int J Pediatr Otorhinolaryngol. 2016 Jun;85:148-53.
A biopsy of the lesion was performed that showed a well-defined nodulocystic tumor composed of nests of basaloid cells that are undergoing trichilemmal keratinization. Shadow cells are seen as well as small areas of calcification. There is also a histiocytic infiltrate with multinucleated giant cells. The histologic diagnosis is of a pilomatrixoma.
Pilomatrixoma, also known as calcifying epithelioma of Malherbe, was first described in 1880, as a tumor of sebaceous gland origin. Later, in 1961, Robert Forbis Jr, MD, and Elson B. Helwig, MD, coined the term pilomatrixoma to describe the hair follicle matrix as the source of the tumor. Pilomatrixomas are commonly seen in the pediatric population, usually in children between 8 and 13 years of age. Our patient is one of the youngest described. The lesions are commonly seen on the face and neck in about 70% of the cases followed by the upper extremities, back, and legs. Clinically, the lesions appear as a firm dermal papule or nodule, which moves freely and may have associated erythema on the skin surface or a blueish gray hue on the underlying skin.
Most pilomatrixomas that have been studied have shown a mutation in Exon 3 of the beta-catenin gene (CTNNB1). The beta-catenin molecule is a subunit of the cadherin protein, which is part of an important pathway in the terminal hair follicle differentiation. Beta-catenin also plays an important role in the Wnt pathway, which regulates cell fate as well as early embryonic patterning. Beta-catenin is responsible for forming adhesion junctions among cells. There have also been immunohistochemical studies that have shown a BCL2 proto-oncogene overexpression to pilomatrixoma.
There are several genetic syndromes that have been associated with the presence of pilomatrixomas: Turner syndrome (XO chromosome abnormality associated with short stature and cardiac defects), Gardner syndrome (polyposis coli and colon and rectal cancer), myotonic dystrophy, Rubinstein-Taybi syndrome (characterized by broad thumbs and toes, short stature, distinctive facial features, and varying degrees of intellectual disability), and trisomy 9. On physical examination our patient didn’t present with any of the typical features or history that could suggest any of these syndromes. A close follow-up and evaluation by a geneticist was recommended because after the initial visit she developed a second lesion on the forehead.
The differential diagnosis for this lesion includes other cysts that may occur on the ear such as epidermal inclusion cyst or dermoid cysts, though these lesions do not tend to be as firm as pilomatrixomas are, which can help with the diagnosis. Dermoid cysts are made of dermal and epidermal components. They are usually present at birth and are commonly seen on the scalp and the periorbital face.
Keloids are rubbery nodules of scar tissue that can form on sites of trauma, and although the lesion occurred after she had her ears pierced, the consistency and rapid growth of the lesion as well as the pathological description made this benign fibrous growth less likely.
When pilomatrixomas are inflamed they can be confused with vascular growths: in this particular case, a hemangioma or another vascular tumor such as a tufted angioma or kaposiform hemangioendothelioma. An ultrasound of the lesion could have helped in the differential diagnosis of the lesion.
Pilomatrixomas can grow significantly and in some cases get inflamed or infected. Surgical management of pilomatrixomas is often required because the lesions do not regress spontaneously.
Dr. Matiz is a pediatric dermatologist at Southern California Permanente Medical Group, San Diego.
References
Forbis R Jr and Helwig EB. Arch Dermatol 1961;83:606-18.
Schwarz Y et al. Int J Pediatr Otorhinolaryngol. 2016 Jun;85:148-53.
A biopsy of the lesion was performed that showed a well-defined nodulocystic tumor composed of nests of basaloid cells that are undergoing trichilemmal keratinization. Shadow cells are seen as well as small areas of calcification. There is also a histiocytic infiltrate with multinucleated giant cells. The histologic diagnosis is of a pilomatrixoma.
Pilomatrixoma, also known as calcifying epithelioma of Malherbe, was first described in 1880, as a tumor of sebaceous gland origin. Later, in 1961, Robert Forbis Jr, MD, and Elson B. Helwig, MD, coined the term pilomatrixoma to describe the hair follicle matrix as the source of the tumor. Pilomatrixomas are commonly seen in the pediatric population, usually in children between 8 and 13 years of age. Our patient is one of the youngest described. The lesions are commonly seen on the face and neck in about 70% of the cases followed by the upper extremities, back, and legs. Clinically, the lesions appear as a firm dermal papule or nodule, which moves freely and may have associated erythema on the skin surface or a blueish gray hue on the underlying skin.
Most pilomatrixomas that have been studied have shown a mutation in Exon 3 of the beta-catenin gene (CTNNB1). The beta-catenin molecule is a subunit of the cadherin protein, which is part of an important pathway in the terminal hair follicle differentiation. Beta-catenin also plays an important role in the Wnt pathway, which regulates cell fate as well as early embryonic patterning. Beta-catenin is responsible for forming adhesion junctions among cells. There have also been immunohistochemical studies that have shown a BCL2 proto-oncogene overexpression to pilomatrixoma.
There are several genetic syndromes that have been associated with the presence of pilomatrixomas: Turner syndrome (XO chromosome abnormality associated with short stature and cardiac defects), Gardner syndrome (polyposis coli and colon and rectal cancer), myotonic dystrophy, Rubinstein-Taybi syndrome (characterized by broad thumbs and toes, short stature, distinctive facial features, and varying degrees of intellectual disability), and trisomy 9. On physical examination our patient didn’t present with any of the typical features or history that could suggest any of these syndromes. A close follow-up and evaluation by a geneticist was recommended because after the initial visit she developed a second lesion on the forehead.
The differential diagnosis for this lesion includes other cysts that may occur on the ear such as epidermal inclusion cyst or dermoid cysts, though these lesions do not tend to be as firm as pilomatrixomas are, which can help with the diagnosis. Dermoid cysts are made of dermal and epidermal components. They are usually present at birth and are commonly seen on the scalp and the periorbital face.
Keloids are rubbery nodules of scar tissue that can form on sites of trauma, and although the lesion occurred after she had her ears pierced, the consistency and rapid growth of the lesion as well as the pathological description made this benign fibrous growth less likely.
When pilomatrixomas are inflamed they can be confused with vascular growths: in this particular case, a hemangioma or another vascular tumor such as a tufted angioma or kaposiform hemangioendothelioma. An ultrasound of the lesion could have helped in the differential diagnosis of the lesion.
Pilomatrixomas can grow significantly and in some cases get inflamed or infected. Surgical management of pilomatrixomas is often required because the lesions do not regress spontaneously.
Dr. Matiz is a pediatric dermatologist at Southern California Permanente Medical Group, San Diego.
References
Forbis R Jr and Helwig EB. Arch Dermatol 1961;83:606-18.
Schwarz Y et al. Int J Pediatr Otorhinolaryngol. 2016 Jun;85:148-53.
A 4-month-old female was referred to our clinic for evaluation of a bump on the right ear. The lesion was first noted at 2 months of age as a little pimple. She was evaluated by her pediatrician and was treated with topical and oral antibiotics without resolution of the lesion. The bump continued to grow and seemed tender to palpation, so she was referred to dermatology for evaluation.
She was born via normal vaginal delivery at 40 weeks. Her mother has no medical conditions and the pregnancy was uneventful. She has been growing and developing well. She takes vitamin D and is currently breast fed.
There have been no other family members with similar lesions. She had her ears pierced at a month of age without any complications.
On skin examination she has a firm red nodule on the right ear that appears slightly tender to touch. She has no other skin lesions of concern. She has normal muscle tone and there are no other abnormalities noted on the physical exam. She has no hepatomegaly, splenomegaly, or lymphadenopathy.