User login
Bringing you the latest news, research and reviews, exclusive interviews, podcasts, quizzes, and more.
div[contains(@class, 'header__large-screen')]
div[contains(@class, 'read-next-article')]
div[contains(@class, 'nav-primary')]
nav[contains(@class, 'nav-primary')]
section[contains(@class, 'footer-nav-section-wrapper')]
footer[@id='footer']
div[contains(@class, 'main-prefix')]
section[contains(@class, 'nav-hidden')]
div[contains(@class, 'ce-card-content')]
nav[contains(@class, 'nav-ce-stack')]
NAFLD strongly correlated with psoriasis, PsA; risk linked to severity
NEW YORK – – and probably in those with psoriatic arthritis (PsA) as well, according to a systematic review and meta-analysis presented at the annual meeting of the Group for Research and Assessment of Psoriasis and Psoriatic Arthritis.
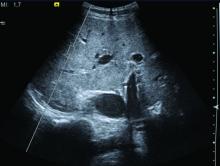
“Our findings imply that psoriatic patients should be screened with an ultrasonographic exam in cases where there are metabolic features that are associated with NAFLD,” reported Francesco Bellinato, MD, a researcher in the section of dermatology and venereology, University of Verona (Italy).
The data are strong. Of 76 nonduplicate publications found in the literature, the 11 observational studies included in the meta-analysis met stringent criteria, including a diagnosis of psoriasis and PsA based on objective criteria, NAFLD confirmed with liver biopsy or imaging, and odds rates calculated with 95% confidence intervals.
From these 11 studies, aggregate data were available for 249,333 psoriatic patients, of which 49% had NAFLD, and 1,491,402 were healthy controls. Among the controls, 36% had NAFLD. Four of the studies were from North America, four from Europe, and three from Asia.
In the pooled data, the risk of NAFLD among those with psoriasis relative to healthy controls fell just short of a twofold increase (odds ratio, 1.96; 95% CI, 1.70-2.26; P < .001). When stratified by studies that confirmed NAFLD by biopsy relative to ultrasonography, there was no significant heterogeneity.
Eight of the studies included an analysis of relative risk in the context of skin lesion severity defined by Psoriasis Area and Severity Index (PASI) score. Relative to those without NAFLD, psoriatic patients with NAFLD had a significant greater mean PASI score on a pooled weighted mean difference analysis (OR, 3.93; 95% CI, 2.01-5.84; P < .0001).
For PsA relative to no PsA in the five studies that compared risk between these two groups, the risk of NAFLD was again nearly twofold higher. This fell short of conventional definition of statistical significance, but it was associated with a strong trend (OR, 1.83; 95% CI, 0.98-3.43; P = .06).
The risk of NAFLD among patients with psoriasis was not found to vary significantly when assessed by univariable meta-regressions across numerous characteristics, such as sex and body mass index.
In one of the largest of the observational studies included in the meta-analysis by Alexis Ogdie, MD, associate professor of medicine and epidemiology at the University of Pennsylvania, Philadelphia, and colleagues, data were analyzed in more than 1.5 million patients, which included 54,251 patients with rheumatoid arthritis. While the hazard ratio of NAFLD was increased for both psoriasis (HR, 2.23) and PsA (HR, 2.11), it was not elevated in those with RA (HR, 0.96).
Risk by severity, possible mechanisms
This study also included an analysis of NAFLD risk according to psoriasis severity. While risk was still significant among those with mild disease (HR, 1.18; 95% CI, 1.07-1.30), it was almost twofold greater in those with moderate to severe psoriasis (HR, 2.23; 95% CI, 1.73-2.87).
Dr. Bellinato conceded that the mechanisms underlying the association between psoriasis and NAFLD are unknown, but he said “metaflammation” is suspected.
“The secretion of proinflammatory, prothrombotic, and oxidative stress mediators in both psoriatic skin and adipose tissue might act systemically and promote insulin resistance and other metabolic derangements that promote the development and progression of NAFLD,” Dr. Bellinato explained.
He thinks that noninvasive screening methods, such as currently used methods to calculate fibrosis score, might be useful for evaluating patients with psoriasis for NAFLD and referring them to a hepatologist when appropriate.
Given the strong association with NAFLD, Dr. Bellinato suggested that “the findings of this meta-analysis pave the way for novel, large, prospective, and histologically based studies.”
The association between psoriasis and NAFLD is clinically relevant, agreed Joel M. Gelfand, MD, vice-chair of clinical research and medical director of the clinical studies unit, department of dermatology, University of Pennsylvania, Philadelphia.
“It is not clear if psoriasis causes fatty liver disease or vice versa, but clinicians should be aware of this association,” he said in an interview. Dr. Gelfand was a coauthor of the study by Dr. Ogdie and colleagues and led another more recent population-based study that implicated methotrexate as a factor in psoriasis-related hepatotoxicity.
If NAFLD is identified in a patient with psoriasis, treatments are limited, but Dr. Gelfand suggested that patients should be made aware of the risk. “Clinicians should encourage patients with psoriasis to take measures to protect their liver, such as avoiding drinking alcohol to excess and trying to maintain a healthy body weight,” he said.
Dr. Bellinato reported no conflicts of interest. Dr. Gelfand has financial relationships with more than 10 pharmaceutical companies, including those that make therapies for psoriasis.
NEW YORK – – and probably in those with psoriatic arthritis (PsA) as well, according to a systematic review and meta-analysis presented at the annual meeting of the Group for Research and Assessment of Psoriasis and Psoriatic Arthritis.
“Our findings imply that psoriatic patients should be screened with an ultrasonographic exam in cases where there are metabolic features that are associated with NAFLD,” reported Francesco Bellinato, MD, a researcher in the section of dermatology and venereology, University of Verona (Italy).
The data are strong. Of 76 nonduplicate publications found in the literature, the 11 observational studies included in the meta-analysis met stringent criteria, including a diagnosis of psoriasis and PsA based on objective criteria, NAFLD confirmed with liver biopsy or imaging, and odds rates calculated with 95% confidence intervals.
From these 11 studies, aggregate data were available for 249,333 psoriatic patients, of which 49% had NAFLD, and 1,491,402 were healthy controls. Among the controls, 36% had NAFLD. Four of the studies were from North America, four from Europe, and three from Asia.
In the pooled data, the risk of NAFLD among those with psoriasis relative to healthy controls fell just short of a twofold increase (odds ratio, 1.96; 95% CI, 1.70-2.26; P < .001). When stratified by studies that confirmed NAFLD by biopsy relative to ultrasonography, there was no significant heterogeneity.
Eight of the studies included an analysis of relative risk in the context of skin lesion severity defined by Psoriasis Area and Severity Index (PASI) score. Relative to those without NAFLD, psoriatic patients with NAFLD had a significant greater mean PASI score on a pooled weighted mean difference analysis (OR, 3.93; 95% CI, 2.01-5.84; P < .0001).
For PsA relative to no PsA in the five studies that compared risk between these two groups, the risk of NAFLD was again nearly twofold higher. This fell short of conventional definition of statistical significance, but it was associated with a strong trend (OR, 1.83; 95% CI, 0.98-3.43; P = .06).
The risk of NAFLD among patients with psoriasis was not found to vary significantly when assessed by univariable meta-regressions across numerous characteristics, such as sex and body mass index.
In one of the largest of the observational studies included in the meta-analysis by Alexis Ogdie, MD, associate professor of medicine and epidemiology at the University of Pennsylvania, Philadelphia, and colleagues, data were analyzed in more than 1.5 million patients, which included 54,251 patients with rheumatoid arthritis. While the hazard ratio of NAFLD was increased for both psoriasis (HR, 2.23) and PsA (HR, 2.11), it was not elevated in those with RA (HR, 0.96).
Risk by severity, possible mechanisms
This study also included an analysis of NAFLD risk according to psoriasis severity. While risk was still significant among those with mild disease (HR, 1.18; 95% CI, 1.07-1.30), it was almost twofold greater in those with moderate to severe psoriasis (HR, 2.23; 95% CI, 1.73-2.87).
Dr. Bellinato conceded that the mechanisms underlying the association between psoriasis and NAFLD are unknown, but he said “metaflammation” is suspected.
“The secretion of proinflammatory, prothrombotic, and oxidative stress mediators in both psoriatic skin and adipose tissue might act systemically and promote insulin resistance and other metabolic derangements that promote the development and progression of NAFLD,” Dr. Bellinato explained.
He thinks that noninvasive screening methods, such as currently used methods to calculate fibrosis score, might be useful for evaluating patients with psoriasis for NAFLD and referring them to a hepatologist when appropriate.
Given the strong association with NAFLD, Dr. Bellinato suggested that “the findings of this meta-analysis pave the way for novel, large, prospective, and histologically based studies.”
The association between psoriasis and NAFLD is clinically relevant, agreed Joel M. Gelfand, MD, vice-chair of clinical research and medical director of the clinical studies unit, department of dermatology, University of Pennsylvania, Philadelphia.
“It is not clear if psoriasis causes fatty liver disease or vice versa, but clinicians should be aware of this association,” he said in an interview. Dr. Gelfand was a coauthor of the study by Dr. Ogdie and colleagues and led another more recent population-based study that implicated methotrexate as a factor in psoriasis-related hepatotoxicity.
If NAFLD is identified in a patient with psoriasis, treatments are limited, but Dr. Gelfand suggested that patients should be made aware of the risk. “Clinicians should encourage patients with psoriasis to take measures to protect their liver, such as avoiding drinking alcohol to excess and trying to maintain a healthy body weight,” he said.
Dr. Bellinato reported no conflicts of interest. Dr. Gelfand has financial relationships with more than 10 pharmaceutical companies, including those that make therapies for psoriasis.
NEW YORK – – and probably in those with psoriatic arthritis (PsA) as well, according to a systematic review and meta-analysis presented at the annual meeting of the Group for Research and Assessment of Psoriasis and Psoriatic Arthritis.
“Our findings imply that psoriatic patients should be screened with an ultrasonographic exam in cases where there are metabolic features that are associated with NAFLD,” reported Francesco Bellinato, MD, a researcher in the section of dermatology and venereology, University of Verona (Italy).
The data are strong. Of 76 nonduplicate publications found in the literature, the 11 observational studies included in the meta-analysis met stringent criteria, including a diagnosis of psoriasis and PsA based on objective criteria, NAFLD confirmed with liver biopsy or imaging, and odds rates calculated with 95% confidence intervals.
From these 11 studies, aggregate data were available for 249,333 psoriatic patients, of which 49% had NAFLD, and 1,491,402 were healthy controls. Among the controls, 36% had NAFLD. Four of the studies were from North America, four from Europe, and three from Asia.
In the pooled data, the risk of NAFLD among those with psoriasis relative to healthy controls fell just short of a twofold increase (odds ratio, 1.96; 95% CI, 1.70-2.26; P < .001). When stratified by studies that confirmed NAFLD by biopsy relative to ultrasonography, there was no significant heterogeneity.
Eight of the studies included an analysis of relative risk in the context of skin lesion severity defined by Psoriasis Area and Severity Index (PASI) score. Relative to those without NAFLD, psoriatic patients with NAFLD had a significant greater mean PASI score on a pooled weighted mean difference analysis (OR, 3.93; 95% CI, 2.01-5.84; P < .0001).
For PsA relative to no PsA in the five studies that compared risk between these two groups, the risk of NAFLD was again nearly twofold higher. This fell short of conventional definition of statistical significance, but it was associated with a strong trend (OR, 1.83; 95% CI, 0.98-3.43; P = .06).
The risk of NAFLD among patients with psoriasis was not found to vary significantly when assessed by univariable meta-regressions across numerous characteristics, such as sex and body mass index.
In one of the largest of the observational studies included in the meta-analysis by Alexis Ogdie, MD, associate professor of medicine and epidemiology at the University of Pennsylvania, Philadelphia, and colleagues, data were analyzed in more than 1.5 million patients, which included 54,251 patients with rheumatoid arthritis. While the hazard ratio of NAFLD was increased for both psoriasis (HR, 2.23) and PsA (HR, 2.11), it was not elevated in those with RA (HR, 0.96).
Risk by severity, possible mechanisms
This study also included an analysis of NAFLD risk according to psoriasis severity. While risk was still significant among those with mild disease (HR, 1.18; 95% CI, 1.07-1.30), it was almost twofold greater in those with moderate to severe psoriasis (HR, 2.23; 95% CI, 1.73-2.87).
Dr. Bellinato conceded that the mechanisms underlying the association between psoriasis and NAFLD are unknown, but he said “metaflammation” is suspected.
“The secretion of proinflammatory, prothrombotic, and oxidative stress mediators in both psoriatic skin and adipose tissue might act systemically and promote insulin resistance and other metabolic derangements that promote the development and progression of NAFLD,” Dr. Bellinato explained.
He thinks that noninvasive screening methods, such as currently used methods to calculate fibrosis score, might be useful for evaluating patients with psoriasis for NAFLD and referring them to a hepatologist when appropriate.
Given the strong association with NAFLD, Dr. Bellinato suggested that “the findings of this meta-analysis pave the way for novel, large, prospective, and histologically based studies.”
The association between psoriasis and NAFLD is clinically relevant, agreed Joel M. Gelfand, MD, vice-chair of clinical research and medical director of the clinical studies unit, department of dermatology, University of Pennsylvania, Philadelphia.
“It is not clear if psoriasis causes fatty liver disease or vice versa, but clinicians should be aware of this association,” he said in an interview. Dr. Gelfand was a coauthor of the study by Dr. Ogdie and colleagues and led another more recent population-based study that implicated methotrexate as a factor in psoriasis-related hepatotoxicity.
If NAFLD is identified in a patient with psoriasis, treatments are limited, but Dr. Gelfand suggested that patients should be made aware of the risk. “Clinicians should encourage patients with psoriasis to take measures to protect their liver, such as avoiding drinking alcohol to excess and trying to maintain a healthy body weight,” he said.
Dr. Bellinato reported no conflicts of interest. Dr. Gelfand has financial relationships with more than 10 pharmaceutical companies, including those that make therapies for psoriasis.
AT GRAPPA 2022
Focal Palmoplantar Keratoderma and Gingival Keratosis Caused by a KRT16 Mutation
To the Editor:
Focal palmoplantar keratoderma and gingival keratosis (FPGK)(Online Mendelian Inheritance in Man [OMIM] 148730) is a rare autosomal-dominant syndrome featuring focal, pressure-related, painful palmoplantar keratoderma and gingival hyperkeratosis presenting as leukokeratosis. Focal palmoplantar keratoderma and gingival keratosis was first defined by Gorlin1 in 1976. Since then, only a few cases have been reported, but no causative mutations have been identified.2
Focal pressure-related palmoplantar keratoderma (PPK) and oral hyperkeratosis also are seen in pachyonychia congenita (PC)(OMIM 167200, 615726, 615728, 167210), a rare autosomal-dominant disorder of keratinization characterized by PPK and nail dystrophy. Patients with PC often present with plantar pain; more variable features include oral leukokeratosis, follicular hyperkeratosis, pilosebaceous and epidermal inclusion cysts, hoarseness, hyperhidrosis, and natal teeth. Pachyonychia congenita is caused by mutation in keratin genes KRT6A, KRT6B, KRT16, or KRT17.
Focal palmoplantar keratoderma and gingival keratosis as well as PC are distinct from other forms of PPK with gingival involvement such as
Despite the common features of FPGK and PC, they are considered distinct disorders due to absence of nail changes in FPGK and no prior evidence of a common genetic cause. We present a patient with familial FPGK found by whole exome sequencing to be caused by a mutation in KRT16.
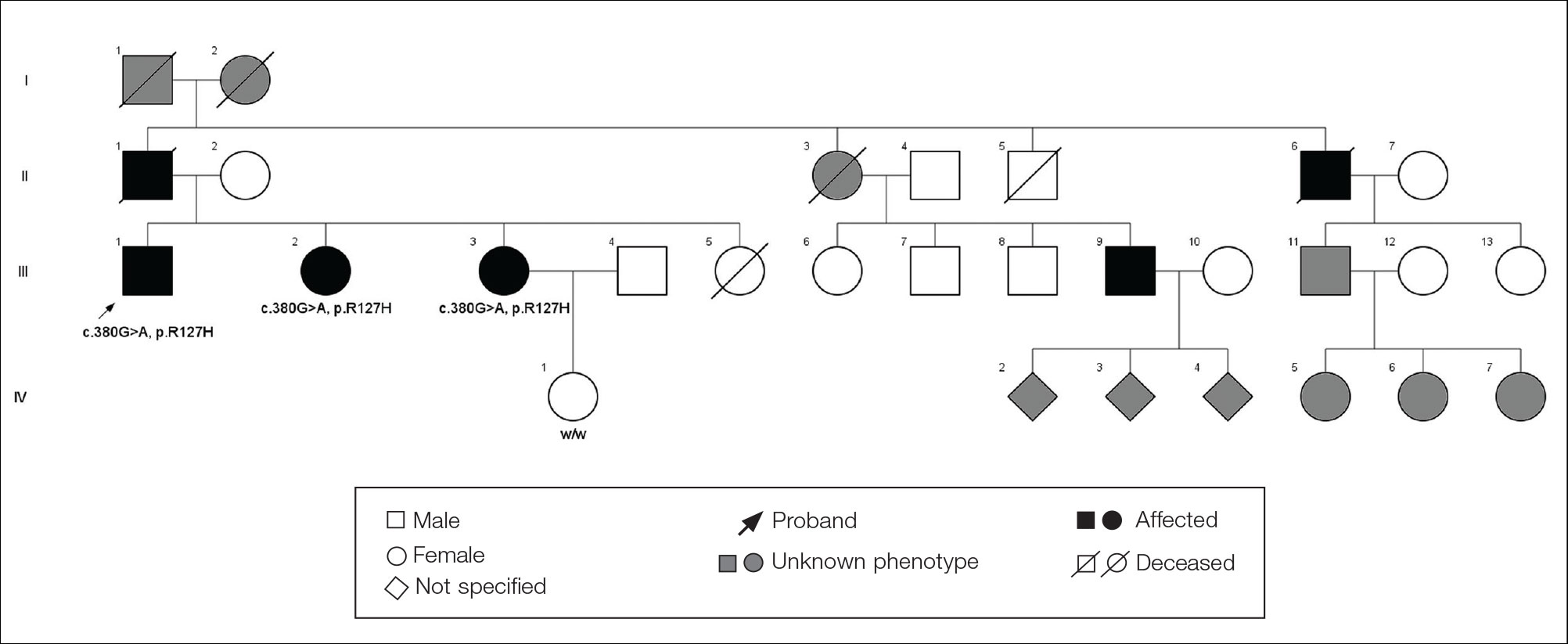
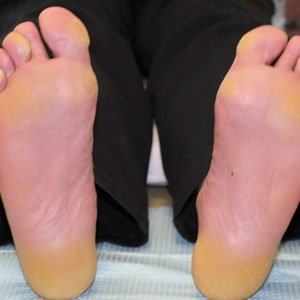
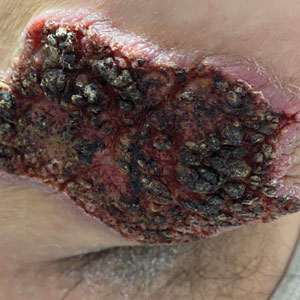
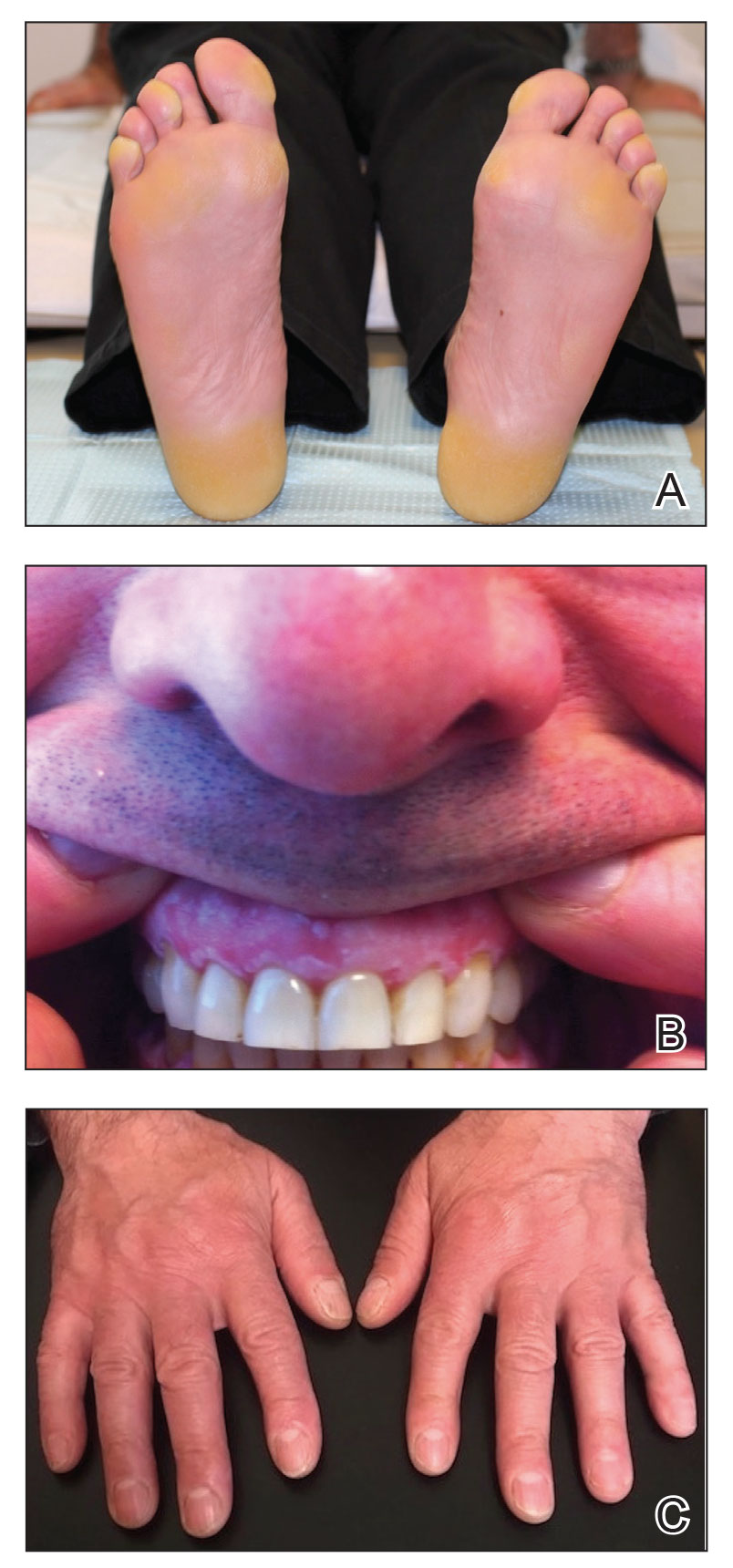
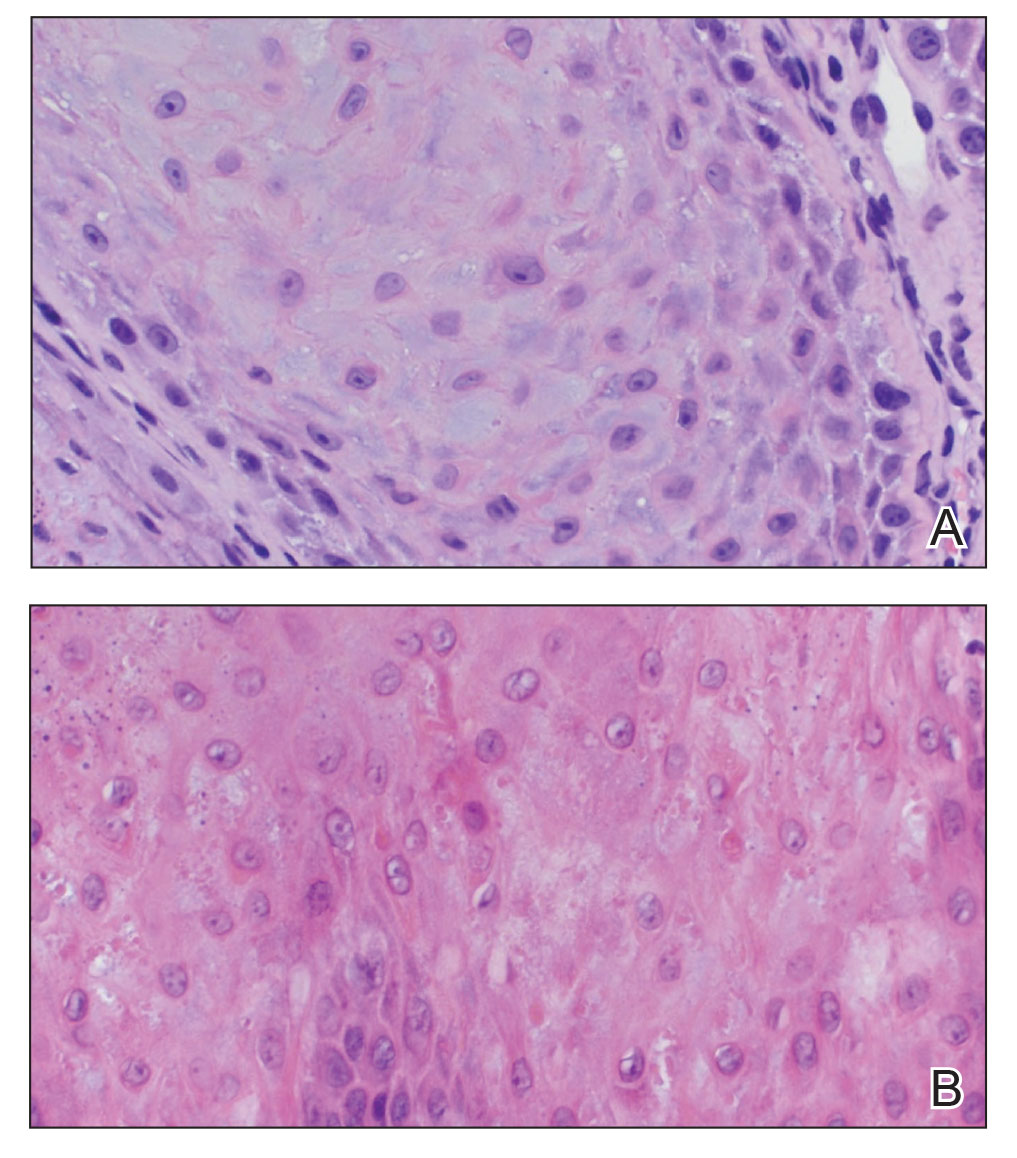
The proband was a 57-year-old man born to unrelated parents (Figure 1). He had no skin problems at birth, and his development was normal. He had painful focal keratoderma since childhood that were most prominent at pressure points on the soles and toes (Figure 2A), in addition to gingival hyperkeratosis and oral leukokeratosis (Figure 2B). He had no associated abnormalities of the skin, hair, or teeth and no nail findings (Figure 2C). He reported that his father and 2 of his 3 sisters were affected with similar symptoms. A punch biopsy of the right fifth toe was consistent with verrucous epidermal hyperplasia with perinuclear keratinization in the spinous layer (Figure 3A). A gingival biopsy showed perinuclear eosinophilic globules and basophilic stranding in the cytoplasm (Figure 3B). His older sister had more severe and painful focal keratoderma of the soles, punctate keratoderma of the palms, gingival hyperkeratosis, and leukokeratosis of the tongue.
Whole exome sequencing of the proband revealed a heterozygous missense mutation in KRT16 (c.380G>A, p.R127H, rs57424749). Sanger sequencing confirmed this mutation and showed that it was heterozygous in both of his affected sisters and absent in his unaffected niece (Figure 1). The patient was treated with topical and systemic retinoids, keratolytics, and mechanical removal to moderate effect, with noted improvement in the appearance and associated pain of the plantar keratoderma.
Phenotypic heterogeneity is common in PC, though PC due to KRT6A mutations demonstrates more severe nail disease with oral lesions, cysts, and follicular hyperkeratosis, while PC caused by KRT16 mutations generally presents with more extensive and painful PPK.4KRT16 mutations affecting p.R127 are frequent causes of PC, and genotype-phenotype correlations have been observed. Individuals with p.R127P mutations exhibit more severe disease with earlier age of onset, more extensive nail involvement and oral leukokeratosis, and greater impact on daily quality of life than in individuals with p.R127C mutations.5 Cases of PC with KRT16 p.R127S and p.R127G mutations also have been observed. The KRT16 c.380G>A, p.R127H mutation we documented has been reported in one kindred with PC who presented with PPK, oral leukokeratosis, toenail thickening, and pilosebaceous and follicular hyperkeratosis.6
Although patients with FPGK lack the thickening of fingernails and/or toenails considered a defining feature of PC, the disorders otherwise are phenotypically similar, suggesting the possibility of common pathogenesis. One linkage study of familial FPGK excluded genetic intervals containing type I and type II keratins but was limited to a single small kindred.2 This study and our data together suggest that, similar to PC, there are multiple genes in which mutations cause FPGK.
Murine Krt16 knockouts show distinct phenotypes depending on the mouse strain in which they are propagated, ranging from perinatal lethality to differences in the severity of oral and PPK lesions.7 These observations provide evidence that additional genetic variants contribute to Krt16 phenotypes in mice and suggest the same could be true for humans.
We propose that some cases of FPGK are due to mutations in KRT16 and thus share a genetic pathogenesis with PC, underscoring the utility of whole exome sequencing in providing genetic diagnoses for disorders that are genetically and clinically heterogeneous. Further biologic investigation of phenotypes caused by KRT16 mutation may reveal respective contributions of additional genetic variation and environmental effects to the variable clinical presentations.
- Gorlin RJ. Focal palmoplantar and marginal gingival hyperkeratosis—a syndrome. Birth Defects Orig Artic Ser. 1976;12:239-242.
- Kolde G, Hennies HC, Bethke G, et al. Focal palmoplantar and gingival keratosis: a distinct palmoplantar ectodermal dysplasia with epidermolytic alterations but lack of mutations in known keratins. J Am Acad Dermatol. 2005;52(3 pt 1):403-409.
- Duchatelet S, Hovnanian A. Olmsted syndrome: clinical, molecular and therapeutic aspects. Orphanet J Rare Dis. 2015;10:33.
- Spaunhurst KM, Hogendorf AM, Smith FJ, et al. Pachyonychia congenita patients with mutations in KRT6A have more extensive disease compared with patients who have mutations in KRT16. Br J Dermatol. 2012;166:875-878.
- Fu T, Leachman SA, Wilson NJ, et al. Genotype-phenotype correlations among pachyonychia congenita patients with K16 mutations. J Invest Dermatol. 2011;131:1025-1028.
- Wilson NJ, O’Toole EA, Milstone LM, et al. The molecular genetic analysis of the expanding pachyonychia congenita case collection. Br J Dermatol. 2014;171:343-355.
- Zieman A, Coulombe PA. The keratin 16 null phenotype is modestly impacted by genetic strain background in mice. Exp Dermatol. 2018;27:672-674.
To the Editor:
Focal palmoplantar keratoderma and gingival keratosis (FPGK)(Online Mendelian Inheritance in Man [OMIM] 148730) is a rare autosomal-dominant syndrome featuring focal, pressure-related, painful palmoplantar keratoderma and gingival hyperkeratosis presenting as leukokeratosis. Focal palmoplantar keratoderma and gingival keratosis was first defined by Gorlin1 in 1976. Since then, only a few cases have been reported, but no causative mutations have been identified.2
Focal pressure-related palmoplantar keratoderma (PPK) and oral hyperkeratosis also are seen in pachyonychia congenita (PC)(OMIM 167200, 615726, 615728, 167210), a rare autosomal-dominant disorder of keratinization characterized by PPK and nail dystrophy. Patients with PC often present with plantar pain; more variable features include oral leukokeratosis, follicular hyperkeratosis, pilosebaceous and epidermal inclusion cysts, hoarseness, hyperhidrosis, and natal teeth. Pachyonychia congenita is caused by mutation in keratin genes KRT6A, KRT6B, KRT16, or KRT17.
Focal palmoplantar keratoderma and gingival keratosis as well as PC are distinct from other forms of PPK with gingival involvement such as
Despite the common features of FPGK and PC, they are considered distinct disorders due to absence of nail changes in FPGK and no prior evidence of a common genetic cause. We present a patient with familial FPGK found by whole exome sequencing to be caused by a mutation in KRT16.

The proband was a 57-year-old man born to unrelated parents (Figure 1). He had no skin problems at birth, and his development was normal. He had painful focal keratoderma since childhood that were most prominent at pressure points on the soles and toes (Figure 2A), in addition to gingival hyperkeratosis and oral leukokeratosis (Figure 2B). He had no associated abnormalities of the skin, hair, or teeth and no nail findings (Figure 2C). He reported that his father and 2 of his 3 sisters were affected with similar symptoms. A punch biopsy of the right fifth toe was consistent with verrucous epidermal hyperplasia with perinuclear keratinization in the spinous layer (Figure 3A). A gingival biopsy showed perinuclear eosinophilic globules and basophilic stranding in the cytoplasm (Figure 3B). His older sister had more severe and painful focal keratoderma of the soles, punctate keratoderma of the palms, gingival hyperkeratosis, and leukokeratosis of the tongue.

Whole exome sequencing of the proband revealed a heterozygous missense mutation in KRT16 (c.380G>A, p.R127H, rs57424749). Sanger sequencing confirmed this mutation and showed that it was heterozygous in both of his affected sisters and absent in his unaffected niece (Figure 1). The patient was treated with topical and systemic retinoids, keratolytics, and mechanical removal to moderate effect, with noted improvement in the appearance and associated pain of the plantar keratoderma.

Phenotypic heterogeneity is common in PC, though PC due to KRT6A mutations demonstrates more severe nail disease with oral lesions, cysts, and follicular hyperkeratosis, while PC caused by KRT16 mutations generally presents with more extensive and painful PPK.4KRT16 mutations affecting p.R127 are frequent causes of PC, and genotype-phenotype correlations have been observed. Individuals with p.R127P mutations exhibit more severe disease with earlier age of onset, more extensive nail involvement and oral leukokeratosis, and greater impact on daily quality of life than in individuals with p.R127C mutations.5 Cases of PC with KRT16 p.R127S and p.R127G mutations also have been observed. The KRT16 c.380G>A, p.R127H mutation we documented has been reported in one kindred with PC who presented with PPK, oral leukokeratosis, toenail thickening, and pilosebaceous and follicular hyperkeratosis.6
Although patients with FPGK lack the thickening of fingernails and/or toenails considered a defining feature of PC, the disorders otherwise are phenotypically similar, suggesting the possibility of common pathogenesis. One linkage study of familial FPGK excluded genetic intervals containing type I and type II keratins but was limited to a single small kindred.2 This study and our data together suggest that, similar to PC, there are multiple genes in which mutations cause FPGK.
Murine Krt16 knockouts show distinct phenotypes depending on the mouse strain in which they are propagated, ranging from perinatal lethality to differences in the severity of oral and PPK lesions.7 These observations provide evidence that additional genetic variants contribute to Krt16 phenotypes in mice and suggest the same could be true for humans.
We propose that some cases of FPGK are due to mutations in KRT16 and thus share a genetic pathogenesis with PC, underscoring the utility of whole exome sequencing in providing genetic diagnoses for disorders that are genetically and clinically heterogeneous. Further biologic investigation of phenotypes caused by KRT16 mutation may reveal respective contributions of additional genetic variation and environmental effects to the variable clinical presentations.
To the Editor:
Focal palmoplantar keratoderma and gingival keratosis (FPGK)(Online Mendelian Inheritance in Man [OMIM] 148730) is a rare autosomal-dominant syndrome featuring focal, pressure-related, painful palmoplantar keratoderma and gingival hyperkeratosis presenting as leukokeratosis. Focal palmoplantar keratoderma and gingival keratosis was first defined by Gorlin1 in 1976. Since then, only a few cases have been reported, but no causative mutations have been identified.2
Focal pressure-related palmoplantar keratoderma (PPK) and oral hyperkeratosis also are seen in pachyonychia congenita (PC)(OMIM 167200, 615726, 615728, 167210), a rare autosomal-dominant disorder of keratinization characterized by PPK and nail dystrophy. Patients with PC often present with plantar pain; more variable features include oral leukokeratosis, follicular hyperkeratosis, pilosebaceous and epidermal inclusion cysts, hoarseness, hyperhidrosis, and natal teeth. Pachyonychia congenita is caused by mutation in keratin genes KRT6A, KRT6B, KRT16, or KRT17.
Focal palmoplantar keratoderma and gingival keratosis as well as PC are distinct from other forms of PPK with gingival involvement such as
Despite the common features of FPGK and PC, they are considered distinct disorders due to absence of nail changes in FPGK and no prior evidence of a common genetic cause. We present a patient with familial FPGK found by whole exome sequencing to be caused by a mutation in KRT16.

The proband was a 57-year-old man born to unrelated parents (Figure 1). He had no skin problems at birth, and his development was normal. He had painful focal keratoderma since childhood that were most prominent at pressure points on the soles and toes (Figure 2A), in addition to gingival hyperkeratosis and oral leukokeratosis (Figure 2B). He had no associated abnormalities of the skin, hair, or teeth and no nail findings (Figure 2C). He reported that his father and 2 of his 3 sisters were affected with similar symptoms. A punch biopsy of the right fifth toe was consistent with verrucous epidermal hyperplasia with perinuclear keratinization in the spinous layer (Figure 3A). A gingival biopsy showed perinuclear eosinophilic globules and basophilic stranding in the cytoplasm (Figure 3B). His older sister had more severe and painful focal keratoderma of the soles, punctate keratoderma of the palms, gingival hyperkeratosis, and leukokeratosis of the tongue.

Whole exome sequencing of the proband revealed a heterozygous missense mutation in KRT16 (c.380G>A, p.R127H, rs57424749). Sanger sequencing confirmed this mutation and showed that it was heterozygous in both of his affected sisters and absent in his unaffected niece (Figure 1). The patient was treated with topical and systemic retinoids, keratolytics, and mechanical removal to moderate effect, with noted improvement in the appearance and associated pain of the plantar keratoderma.

Phenotypic heterogeneity is common in PC, though PC due to KRT6A mutations demonstrates more severe nail disease with oral lesions, cysts, and follicular hyperkeratosis, while PC caused by KRT16 mutations generally presents with more extensive and painful PPK.4KRT16 mutations affecting p.R127 are frequent causes of PC, and genotype-phenotype correlations have been observed. Individuals with p.R127P mutations exhibit more severe disease with earlier age of onset, more extensive nail involvement and oral leukokeratosis, and greater impact on daily quality of life than in individuals with p.R127C mutations.5 Cases of PC with KRT16 p.R127S and p.R127G mutations also have been observed. The KRT16 c.380G>A, p.R127H mutation we documented has been reported in one kindred with PC who presented with PPK, oral leukokeratosis, toenail thickening, and pilosebaceous and follicular hyperkeratosis.6
Although patients with FPGK lack the thickening of fingernails and/or toenails considered a defining feature of PC, the disorders otherwise are phenotypically similar, suggesting the possibility of common pathogenesis. One linkage study of familial FPGK excluded genetic intervals containing type I and type II keratins but was limited to a single small kindred.2 This study and our data together suggest that, similar to PC, there are multiple genes in which mutations cause FPGK.
Murine Krt16 knockouts show distinct phenotypes depending on the mouse strain in which they are propagated, ranging from perinatal lethality to differences in the severity of oral and PPK lesions.7 These observations provide evidence that additional genetic variants contribute to Krt16 phenotypes in mice and suggest the same could be true for humans.
We propose that some cases of FPGK are due to mutations in KRT16 and thus share a genetic pathogenesis with PC, underscoring the utility of whole exome sequencing in providing genetic diagnoses for disorders that are genetically and clinically heterogeneous. Further biologic investigation of phenotypes caused by KRT16 mutation may reveal respective contributions of additional genetic variation and environmental effects to the variable clinical presentations.
- Gorlin RJ. Focal palmoplantar and marginal gingival hyperkeratosis—a syndrome. Birth Defects Orig Artic Ser. 1976;12:239-242.
- Kolde G, Hennies HC, Bethke G, et al. Focal palmoplantar and gingival keratosis: a distinct palmoplantar ectodermal dysplasia with epidermolytic alterations but lack of mutations in known keratins. J Am Acad Dermatol. 2005;52(3 pt 1):403-409.
- Duchatelet S, Hovnanian A. Olmsted syndrome: clinical, molecular and therapeutic aspects. Orphanet J Rare Dis. 2015;10:33.
- Spaunhurst KM, Hogendorf AM, Smith FJ, et al. Pachyonychia congenita patients with mutations in KRT6A have more extensive disease compared with patients who have mutations in KRT16. Br J Dermatol. 2012;166:875-878.
- Fu T, Leachman SA, Wilson NJ, et al. Genotype-phenotype correlations among pachyonychia congenita patients with K16 mutations. J Invest Dermatol. 2011;131:1025-1028.
- Wilson NJ, O’Toole EA, Milstone LM, et al. The molecular genetic analysis of the expanding pachyonychia congenita case collection. Br J Dermatol. 2014;171:343-355.
- Zieman A, Coulombe PA. The keratin 16 null phenotype is modestly impacted by genetic strain background in mice. Exp Dermatol. 2018;27:672-674.
- Gorlin RJ. Focal palmoplantar and marginal gingival hyperkeratosis—a syndrome. Birth Defects Orig Artic Ser. 1976;12:239-242.
- Kolde G, Hennies HC, Bethke G, et al. Focal palmoplantar and gingival keratosis: a distinct palmoplantar ectodermal dysplasia with epidermolytic alterations but lack of mutations in known keratins. J Am Acad Dermatol. 2005;52(3 pt 1):403-409.
- Duchatelet S, Hovnanian A. Olmsted syndrome: clinical, molecular and therapeutic aspects. Orphanet J Rare Dis. 2015;10:33.
- Spaunhurst KM, Hogendorf AM, Smith FJ, et al. Pachyonychia congenita patients with mutations in KRT6A have more extensive disease compared with patients who have mutations in KRT16. Br J Dermatol. 2012;166:875-878.
- Fu T, Leachman SA, Wilson NJ, et al. Genotype-phenotype correlations among pachyonychia congenita patients with K16 mutations. J Invest Dermatol. 2011;131:1025-1028.
- Wilson NJ, O’Toole EA, Milstone LM, et al. The molecular genetic analysis of the expanding pachyonychia congenita case collection. Br J Dermatol. 2014;171:343-355.
- Zieman A, Coulombe PA. The keratin 16 null phenotype is modestly impacted by genetic strain background in mice. Exp Dermatol. 2018;27:672-674.
Practice Points
- Focal palmoplantar keratoderma and gingival keratosis (FPGK) is a rare autosomal-dominant syndrome featuring focal, pressure-related, painful palmoplantar keratoderma (PPK) and gingival hyperkeratosis presenting as leukokeratosis.
- Focal pressure-related PPK and oral hyperkeratosis also are seen in pachyonychia congenita (PC), which is caused by mutations in keratin genes and is distinguished from FPGK by characteristic nail changes.
- A shared causative gene suggests that FPGK should be considered part of the PC spectrum.
Meet Argireline, the neurotoxinlike cosmeceutical
Acetyl hexapeptide-8 (or -3), better known by its brand name, Argireline (Lubrizol; Wickliffe, Ohio), is a synthetic peptide gaining popularity in cosmeceutical products for its antiaging benefits. Argireline was developed by the company Lipotec in 2001. Media, beauty bloggers, and product claims have likened this product to a “Botox [or other neurotoxin] alternative,” or “Botox mimicker.”
Mechanism of action
Understanding how Argireline works requires a brief refresher on the mechanism of action of botulinum neurotoxin (BoNT). BoNT relaxes facial muscles and smooths expression lines by inhibiting acetylcholine release at the neuromuscular junction.1 More specifically, the various serotypes of BoNT are single-chain polypeptides that target members of the SNARE complex: SNAP-25, syntaxin, and Vamp. The proteins within the SNARE complex are involved in the docking and fusion of presynaptic vesicles to the presynaptic membrane, necessary steps for acetylcholine release into the neuromuscular junction and muscle contraction. By blocking the action of the SNARE complex proteins, BoNT inhibits release of acetylcholine in the neuromuscular junction and prevents muscle contraction.
Argireline is a synthetic peptide with the sequence Ac-EEMQRR-NH2.2 It is patterned after the N-terminal domain of SNAP-25, one of the members of the SNARE complex targeted by BoNT, and functions to interfere with the assembly of the SNARE complex. In this manner, Argireline would theoretically inhibit fusion of presynaptic vesicles and release of acetylcholine into the neuromuscular junction, thus impeding muscle movement. For this reason, it has been likened to topical Botox. Unlike Botox and other neurotoxins, Argireline was developed for topical application rather than injection.
Preclinical studies
In vitro work done 20 years ago demonstrated that Argireline can prevent assembly of the SNARE complex and inhibit neurotransmitter release with a potency similar to that of BoNT A (Botox).2
In 2013, Wang et al. evaluated the histologic effects of Argireline in aged mouse skin induced by D-galactose. For 6 weeks, Argireline was applied twice daily, and histological changes were assessed using hematoxylin and eosin (H&E) and picrosirius–polarization (PSP) stains. The researchers found elevated levels of type I collagen (P < .01) and reduced type III collagen (P < .05) with the Argireline treatment. These results demonstrated that Argireline could histologically enhance collagen in a manner consistent with skin rejuvenation.3
Clinical studies
In 2002, Blanes et al. assessed the antiwrinkle activity of Argireline by measuring skin topography from silicone implants in the lateral periorbital region of an oil/water (O/W) emulsion containing 10% of the acetyl-hexapeptide in 10 healthy women volunteers. The hexapeptide emulsion was applied twice daily in one lateral periorbital area, and the emulsion vehicle alone was applied twice daily on the contralateral side. Over 30 days of treatment, wrinkle depth was found to have decreased by 30%. The investigators also found that Argireline significantly hindered neurotransmitter release in vitro as robustly as BoNT A, though with notably lower efficacy. No toxicity or irritation was associated with this treatment.2 However, it should be noted that this small study conducted 2 decades ago evaluated only silicone implants with confocal microscopy to evaluate wrinkle depth. There was no subjective clinical assessment of dynamic facial wrinkles. As such, their study is an insufficient basis for drawing conclusions that Argireline is a BoNT mimic. Botox and other types of BoNT affect dynamic facial wrinkles mostly (i.e., wrinkles created by moving muscles of facial expression). This study primarily considers static wrinkles on periorbital skin. While static wrinkles may result from longstanding dynamic wrinkles, BoNT mainly targets dynamic wrinkles, again not comparing apples to apples.
At the same time that Wang et al. conducted their experiment on the skin of aged mice as noted above, they performed a multicenter clinical trial in 60 human subjects who received a randomized treatment of Argireline or placebo in a ratio of 3:1 to assess its safety and efficacy. For 4 weeks, the test product or placebo was applied to periorbital wrinkles twice daily. The researchers found the total antiwrinkle efficacy in the Argireline group to be 48.9% based on the subjective evaluation, compared with 0% in the placebo group. The objective evaluation indicated that all parameters of roughness were diminished in the Argireline group (P < .01), with no reduction observed in the placebo group (P < .05).4 There was a little more to appreciate from this study compared with the one reported by Blanes et al., insofar as subjective evaluations and objective evaluations with silica replicas were done. However, this study was not blinded, so the 48.9% wrinkle reduction in the Argireline group vs. 0% in the control group seems suspicious. Additionally, there was a greater focus on static rather than dynamic wrinkles.
In 2017, Raikou et al. conducted a prospective, randomized controlled study to assess the effects of acetyl hexapeptide-3 (Argireline) and tripeptide-10 citrulline in 24 healthy female volunteers (aged 30-60 years) and determine if there was any synergistic action between the peptides. Subjects were randomized to receive a combination of the peptides, tripeptide-10 citrulline only, acetyl hexapeptide-3 only, or neither peptide for 60 days. The researchers found a significant reduction in transepidermal water loss (TEWL) in the Argireline group, compared with the placebo group.5 The result of this study makes me question if the decrease in depth of the wrinkles measured in the former studies is really just a measure of increased skin hydration from the Argireline, rather than a neurotoxic effect of Argireline.
Formulation and penetration: Can Argireline get through your skin?
One of the fundamental questions regarding Argireline is whether it can penetrate through the stratum corneum and find its target – the facial muscles – where it is intended to function. Argireline is a charged, hydrophilic, and large–molecular weight peptide, and each of these factors impairs penetration through the stratum corneum. Therefore, studies assessing penetration are particularly important.
In 2015, Kraeling et al. conducted an in vitro evaluation of the skin penetration of acetyl hexapeptide-8 in hairless guinea pig and human cadaver skin. An oil-in-water (O/W) emulsion containing 10% acetyl hexapeptide-8 was applied (2 mg/cm2) and penetration was quantified in skin layers via hydrophilic interaction liquid chromatography with tandem mass spectrometry. Most of the acetyl hexapeptide-8 was found to have been washed from human cadaver, as well as guinea pig, skin. Less than 1% of the peptide penetrated the guinea pig or human skin. Of this small amount that penetrated the skin, most stayed in the stratum corneum of guinea pigs (0.54%) and human cadavers (0.22%). The levels of acetyl hexapeptide-8 declined further with each layer of tape stripping removal. Epidermal levels of the peptide in tested skin were similar at 0.01%, and none of the peptide was found in the dermis.6 These results indicate negligible penetration by this highly touted peptide ingredient.
Some studies have shown that altering the formulation of acetyl hexapeptide-8 can enhance penetration. Hoppel et al. demonstrated that formulations of the peptide, especially in a water-oil-water (W/O/W emulsion [as compared with O/W and W/O emulsions] can increase penetration into the stratum corneum in porcine skin.7 Notably, this is still very superficial relative to the dermis and muscles. Irrespective of formulation, studies have shown that Argireline barely penetrates the stratum corneum, let alone the dermis. Therefore, I would give pause to attributing any clinical impact or benefit of Argireline to its neurotoxinlike effects measured in vitro.
Conclusion
Despite the growing popularity of this ingredient in cosmeceuticals and the praise it gets in media for acting as a topical neurotoxin, there are no rigorous clinical trials or data demonstrating its efficacy in suppressing dynamic facial wrinkles like BoNT does. Most importantly, without penetration into the stratum corneum and deeper layers of the skin, it seems unlikely that Argireline’s clinical benefit derives from a neurotoxiclike mechanism of action. It seems more likely that the Argireline-containing product enhances hydration or imparts some other quality to the skin surface. While there is certainly great appeal for a neurotoxinlike product without injections, I do not believe this ingredient will replace injections of BoNT in the foreseeable future, or at least until scientists can figure out how to enable these products to penetrate into the deeper layers of the skin.
Dr. Goldman is a dermatologist in private practice in Miami and specializes in cosmetic and general dermatology. She practices at Baumann Cosmetic & Research Institute and is also opening a general dermatology practice. Dr. Goldman has no relevant disclosures. Write to her at [email protected] or message her on Instagram @DrChloeGoldman.
References
1. Reddy BY et al. Exp Dermatol. 2012 Aug;21(8):569-75.
2. Blanes-Mira C et al. Int J Cosmet Sci. 2002 Oct;24(5):303-10.
3. Wang Y et al. J Cosmet Laser Ther. 2013 Aug;15(4):237-41.
4. Wang Y et al. J Cosmet Laser Ther. 2013;14(2):147-53.
5. Raikou V et al. J Cosmet Dermatol. 2017 Jun;16(2):271-8.
6. Kraeling ME et al. Cutan Ocul Toxicol. 2015 Mar;34(1):46-52.
7. Hoppel M et al. Eur J Pharm Sci. 2015 Feb 20;68:27-35.
Acetyl hexapeptide-8 (or -3), better known by its brand name, Argireline (Lubrizol; Wickliffe, Ohio), is a synthetic peptide gaining popularity in cosmeceutical products for its antiaging benefits. Argireline was developed by the company Lipotec in 2001. Media, beauty bloggers, and product claims have likened this product to a “Botox [or other neurotoxin] alternative,” or “Botox mimicker.”
Mechanism of action
Understanding how Argireline works requires a brief refresher on the mechanism of action of botulinum neurotoxin (BoNT). BoNT relaxes facial muscles and smooths expression lines by inhibiting acetylcholine release at the neuromuscular junction.1 More specifically, the various serotypes of BoNT are single-chain polypeptides that target members of the SNARE complex: SNAP-25, syntaxin, and Vamp. The proteins within the SNARE complex are involved in the docking and fusion of presynaptic vesicles to the presynaptic membrane, necessary steps for acetylcholine release into the neuromuscular junction and muscle contraction. By blocking the action of the SNARE complex proteins, BoNT inhibits release of acetylcholine in the neuromuscular junction and prevents muscle contraction.
Argireline is a synthetic peptide with the sequence Ac-EEMQRR-NH2.2 It is patterned after the N-terminal domain of SNAP-25, one of the members of the SNARE complex targeted by BoNT, and functions to interfere with the assembly of the SNARE complex. In this manner, Argireline would theoretically inhibit fusion of presynaptic vesicles and release of acetylcholine into the neuromuscular junction, thus impeding muscle movement. For this reason, it has been likened to topical Botox. Unlike Botox and other neurotoxins, Argireline was developed for topical application rather than injection.
Preclinical studies
In vitro work done 20 years ago demonstrated that Argireline can prevent assembly of the SNARE complex and inhibit neurotransmitter release with a potency similar to that of BoNT A (Botox).2
In 2013, Wang et al. evaluated the histologic effects of Argireline in aged mouse skin induced by D-galactose. For 6 weeks, Argireline was applied twice daily, and histological changes were assessed using hematoxylin and eosin (H&E) and picrosirius–polarization (PSP) stains. The researchers found elevated levels of type I collagen (P < .01) and reduced type III collagen (P < .05) with the Argireline treatment. These results demonstrated that Argireline could histologically enhance collagen in a manner consistent with skin rejuvenation.3
Clinical studies
In 2002, Blanes et al. assessed the antiwrinkle activity of Argireline by measuring skin topography from silicone implants in the lateral periorbital region of an oil/water (O/W) emulsion containing 10% of the acetyl-hexapeptide in 10 healthy women volunteers. The hexapeptide emulsion was applied twice daily in one lateral periorbital area, and the emulsion vehicle alone was applied twice daily on the contralateral side. Over 30 days of treatment, wrinkle depth was found to have decreased by 30%. The investigators also found that Argireline significantly hindered neurotransmitter release in vitro as robustly as BoNT A, though with notably lower efficacy. No toxicity or irritation was associated with this treatment.2 However, it should be noted that this small study conducted 2 decades ago evaluated only silicone implants with confocal microscopy to evaluate wrinkle depth. There was no subjective clinical assessment of dynamic facial wrinkles. As such, their study is an insufficient basis for drawing conclusions that Argireline is a BoNT mimic. Botox and other types of BoNT affect dynamic facial wrinkles mostly (i.e., wrinkles created by moving muscles of facial expression). This study primarily considers static wrinkles on periorbital skin. While static wrinkles may result from longstanding dynamic wrinkles, BoNT mainly targets dynamic wrinkles, again not comparing apples to apples.
At the same time that Wang et al. conducted their experiment on the skin of aged mice as noted above, they performed a multicenter clinical trial in 60 human subjects who received a randomized treatment of Argireline or placebo in a ratio of 3:1 to assess its safety and efficacy. For 4 weeks, the test product or placebo was applied to periorbital wrinkles twice daily. The researchers found the total antiwrinkle efficacy in the Argireline group to be 48.9% based on the subjective evaluation, compared with 0% in the placebo group. The objective evaluation indicated that all parameters of roughness were diminished in the Argireline group (P < .01), with no reduction observed in the placebo group (P < .05).4 There was a little more to appreciate from this study compared with the one reported by Blanes et al., insofar as subjective evaluations and objective evaluations with silica replicas were done. However, this study was not blinded, so the 48.9% wrinkle reduction in the Argireline group vs. 0% in the control group seems suspicious. Additionally, there was a greater focus on static rather than dynamic wrinkles.
In 2017, Raikou et al. conducted a prospective, randomized controlled study to assess the effects of acetyl hexapeptide-3 (Argireline) and tripeptide-10 citrulline in 24 healthy female volunteers (aged 30-60 years) and determine if there was any synergistic action between the peptides. Subjects were randomized to receive a combination of the peptides, tripeptide-10 citrulline only, acetyl hexapeptide-3 only, or neither peptide for 60 days. The researchers found a significant reduction in transepidermal water loss (TEWL) in the Argireline group, compared with the placebo group.5 The result of this study makes me question if the decrease in depth of the wrinkles measured in the former studies is really just a measure of increased skin hydration from the Argireline, rather than a neurotoxic effect of Argireline.
Formulation and penetration: Can Argireline get through your skin?
One of the fundamental questions regarding Argireline is whether it can penetrate through the stratum corneum and find its target – the facial muscles – where it is intended to function. Argireline is a charged, hydrophilic, and large–molecular weight peptide, and each of these factors impairs penetration through the stratum corneum. Therefore, studies assessing penetration are particularly important.
In 2015, Kraeling et al. conducted an in vitro evaluation of the skin penetration of acetyl hexapeptide-8 in hairless guinea pig and human cadaver skin. An oil-in-water (O/W) emulsion containing 10% acetyl hexapeptide-8 was applied (2 mg/cm2) and penetration was quantified in skin layers via hydrophilic interaction liquid chromatography with tandem mass spectrometry. Most of the acetyl hexapeptide-8 was found to have been washed from human cadaver, as well as guinea pig, skin. Less than 1% of the peptide penetrated the guinea pig or human skin. Of this small amount that penetrated the skin, most stayed in the stratum corneum of guinea pigs (0.54%) and human cadavers (0.22%). The levels of acetyl hexapeptide-8 declined further with each layer of tape stripping removal. Epidermal levels of the peptide in tested skin were similar at 0.01%, and none of the peptide was found in the dermis.6 These results indicate negligible penetration by this highly touted peptide ingredient.
Some studies have shown that altering the formulation of acetyl hexapeptide-8 can enhance penetration. Hoppel et al. demonstrated that formulations of the peptide, especially in a water-oil-water (W/O/W emulsion [as compared with O/W and W/O emulsions] can increase penetration into the stratum corneum in porcine skin.7 Notably, this is still very superficial relative to the dermis and muscles. Irrespective of formulation, studies have shown that Argireline barely penetrates the stratum corneum, let alone the dermis. Therefore, I would give pause to attributing any clinical impact or benefit of Argireline to its neurotoxinlike effects measured in vitro.
Conclusion
Despite the growing popularity of this ingredient in cosmeceuticals and the praise it gets in media for acting as a topical neurotoxin, there are no rigorous clinical trials or data demonstrating its efficacy in suppressing dynamic facial wrinkles like BoNT does. Most importantly, without penetration into the stratum corneum and deeper layers of the skin, it seems unlikely that Argireline’s clinical benefit derives from a neurotoxiclike mechanism of action. It seems more likely that the Argireline-containing product enhances hydration or imparts some other quality to the skin surface. While there is certainly great appeal for a neurotoxinlike product without injections, I do not believe this ingredient will replace injections of BoNT in the foreseeable future, or at least until scientists can figure out how to enable these products to penetrate into the deeper layers of the skin.
Dr. Goldman is a dermatologist in private practice in Miami and specializes in cosmetic and general dermatology. She practices at Baumann Cosmetic & Research Institute and is also opening a general dermatology practice. Dr. Goldman has no relevant disclosures. Write to her at [email protected] or message her on Instagram @DrChloeGoldman.
References
1. Reddy BY et al. Exp Dermatol. 2012 Aug;21(8):569-75.
2. Blanes-Mira C et al. Int J Cosmet Sci. 2002 Oct;24(5):303-10.
3. Wang Y et al. J Cosmet Laser Ther. 2013 Aug;15(4):237-41.
4. Wang Y et al. J Cosmet Laser Ther. 2013;14(2):147-53.
5. Raikou V et al. J Cosmet Dermatol. 2017 Jun;16(2):271-8.
6. Kraeling ME et al. Cutan Ocul Toxicol. 2015 Mar;34(1):46-52.
7. Hoppel M et al. Eur J Pharm Sci. 2015 Feb 20;68:27-35.
Acetyl hexapeptide-8 (or -3), better known by its brand name, Argireline (Lubrizol; Wickliffe, Ohio), is a synthetic peptide gaining popularity in cosmeceutical products for its antiaging benefits. Argireline was developed by the company Lipotec in 2001. Media, beauty bloggers, and product claims have likened this product to a “Botox [or other neurotoxin] alternative,” or “Botox mimicker.”
Mechanism of action
Understanding how Argireline works requires a brief refresher on the mechanism of action of botulinum neurotoxin (BoNT). BoNT relaxes facial muscles and smooths expression lines by inhibiting acetylcholine release at the neuromuscular junction.1 More specifically, the various serotypes of BoNT are single-chain polypeptides that target members of the SNARE complex: SNAP-25, syntaxin, and Vamp. The proteins within the SNARE complex are involved in the docking and fusion of presynaptic vesicles to the presynaptic membrane, necessary steps for acetylcholine release into the neuromuscular junction and muscle contraction. By blocking the action of the SNARE complex proteins, BoNT inhibits release of acetylcholine in the neuromuscular junction and prevents muscle contraction.
Argireline is a synthetic peptide with the sequence Ac-EEMQRR-NH2.2 It is patterned after the N-terminal domain of SNAP-25, one of the members of the SNARE complex targeted by BoNT, and functions to interfere with the assembly of the SNARE complex. In this manner, Argireline would theoretically inhibit fusion of presynaptic vesicles and release of acetylcholine into the neuromuscular junction, thus impeding muscle movement. For this reason, it has been likened to topical Botox. Unlike Botox and other neurotoxins, Argireline was developed for topical application rather than injection.
Preclinical studies
In vitro work done 20 years ago demonstrated that Argireline can prevent assembly of the SNARE complex and inhibit neurotransmitter release with a potency similar to that of BoNT A (Botox).2
In 2013, Wang et al. evaluated the histologic effects of Argireline in aged mouse skin induced by D-galactose. For 6 weeks, Argireline was applied twice daily, and histological changes were assessed using hematoxylin and eosin (H&E) and picrosirius–polarization (PSP) stains. The researchers found elevated levels of type I collagen (P < .01) and reduced type III collagen (P < .05) with the Argireline treatment. These results demonstrated that Argireline could histologically enhance collagen in a manner consistent with skin rejuvenation.3
Clinical studies
In 2002, Blanes et al. assessed the antiwrinkle activity of Argireline by measuring skin topography from silicone implants in the lateral periorbital region of an oil/water (O/W) emulsion containing 10% of the acetyl-hexapeptide in 10 healthy women volunteers. The hexapeptide emulsion was applied twice daily in one lateral periorbital area, and the emulsion vehicle alone was applied twice daily on the contralateral side. Over 30 days of treatment, wrinkle depth was found to have decreased by 30%. The investigators also found that Argireline significantly hindered neurotransmitter release in vitro as robustly as BoNT A, though with notably lower efficacy. No toxicity or irritation was associated with this treatment.2 However, it should be noted that this small study conducted 2 decades ago evaluated only silicone implants with confocal microscopy to evaluate wrinkle depth. There was no subjective clinical assessment of dynamic facial wrinkles. As such, their study is an insufficient basis for drawing conclusions that Argireline is a BoNT mimic. Botox and other types of BoNT affect dynamic facial wrinkles mostly (i.e., wrinkles created by moving muscles of facial expression). This study primarily considers static wrinkles on periorbital skin. While static wrinkles may result from longstanding dynamic wrinkles, BoNT mainly targets dynamic wrinkles, again not comparing apples to apples.
At the same time that Wang et al. conducted their experiment on the skin of aged mice as noted above, they performed a multicenter clinical trial in 60 human subjects who received a randomized treatment of Argireline or placebo in a ratio of 3:1 to assess its safety and efficacy. For 4 weeks, the test product or placebo was applied to periorbital wrinkles twice daily. The researchers found the total antiwrinkle efficacy in the Argireline group to be 48.9% based on the subjective evaluation, compared with 0% in the placebo group. The objective evaluation indicated that all parameters of roughness were diminished in the Argireline group (P < .01), with no reduction observed in the placebo group (P < .05).4 There was a little more to appreciate from this study compared with the one reported by Blanes et al., insofar as subjective evaluations and objective evaluations with silica replicas were done. However, this study was not blinded, so the 48.9% wrinkle reduction in the Argireline group vs. 0% in the control group seems suspicious. Additionally, there was a greater focus on static rather than dynamic wrinkles.
In 2017, Raikou et al. conducted a prospective, randomized controlled study to assess the effects of acetyl hexapeptide-3 (Argireline) and tripeptide-10 citrulline in 24 healthy female volunteers (aged 30-60 years) and determine if there was any synergistic action between the peptides. Subjects were randomized to receive a combination of the peptides, tripeptide-10 citrulline only, acetyl hexapeptide-3 only, or neither peptide for 60 days. The researchers found a significant reduction in transepidermal water loss (TEWL) in the Argireline group, compared with the placebo group.5 The result of this study makes me question if the decrease in depth of the wrinkles measured in the former studies is really just a measure of increased skin hydration from the Argireline, rather than a neurotoxic effect of Argireline.
Formulation and penetration: Can Argireline get through your skin?
One of the fundamental questions regarding Argireline is whether it can penetrate through the stratum corneum and find its target – the facial muscles – where it is intended to function. Argireline is a charged, hydrophilic, and large–molecular weight peptide, and each of these factors impairs penetration through the stratum corneum. Therefore, studies assessing penetration are particularly important.
In 2015, Kraeling et al. conducted an in vitro evaluation of the skin penetration of acetyl hexapeptide-8 in hairless guinea pig and human cadaver skin. An oil-in-water (O/W) emulsion containing 10% acetyl hexapeptide-8 was applied (2 mg/cm2) and penetration was quantified in skin layers via hydrophilic interaction liquid chromatography with tandem mass spectrometry. Most of the acetyl hexapeptide-8 was found to have been washed from human cadaver, as well as guinea pig, skin. Less than 1% of the peptide penetrated the guinea pig or human skin. Of this small amount that penetrated the skin, most stayed in the stratum corneum of guinea pigs (0.54%) and human cadavers (0.22%). The levels of acetyl hexapeptide-8 declined further with each layer of tape stripping removal. Epidermal levels of the peptide in tested skin were similar at 0.01%, and none of the peptide was found in the dermis.6 These results indicate negligible penetration by this highly touted peptide ingredient.
Some studies have shown that altering the formulation of acetyl hexapeptide-8 can enhance penetration. Hoppel et al. demonstrated that formulations of the peptide, especially in a water-oil-water (W/O/W emulsion [as compared with O/W and W/O emulsions] can increase penetration into the stratum corneum in porcine skin.7 Notably, this is still very superficial relative to the dermis and muscles. Irrespective of formulation, studies have shown that Argireline barely penetrates the stratum corneum, let alone the dermis. Therefore, I would give pause to attributing any clinical impact or benefit of Argireline to its neurotoxinlike effects measured in vitro.
Conclusion
Despite the growing popularity of this ingredient in cosmeceuticals and the praise it gets in media for acting as a topical neurotoxin, there are no rigorous clinical trials or data demonstrating its efficacy in suppressing dynamic facial wrinkles like BoNT does. Most importantly, without penetration into the stratum corneum and deeper layers of the skin, it seems unlikely that Argireline’s clinical benefit derives from a neurotoxiclike mechanism of action. It seems more likely that the Argireline-containing product enhances hydration or imparts some other quality to the skin surface. While there is certainly great appeal for a neurotoxinlike product without injections, I do not believe this ingredient will replace injections of BoNT in the foreseeable future, or at least until scientists can figure out how to enable these products to penetrate into the deeper layers of the skin.
Dr. Goldman is a dermatologist in private practice in Miami and specializes in cosmetic and general dermatology. She practices at Baumann Cosmetic & Research Institute and is also opening a general dermatology practice. Dr. Goldman has no relevant disclosures. Write to her at [email protected] or message her on Instagram @DrChloeGoldman.
References
1. Reddy BY et al. Exp Dermatol. 2012 Aug;21(8):569-75.
2. Blanes-Mira C et al. Int J Cosmet Sci. 2002 Oct;24(5):303-10.
3. Wang Y et al. J Cosmet Laser Ther. 2013 Aug;15(4):237-41.
4. Wang Y et al. J Cosmet Laser Ther. 2013;14(2):147-53.
5. Raikou V et al. J Cosmet Dermatol. 2017 Jun;16(2):271-8.
6. Kraeling ME et al. Cutan Ocul Toxicol. 2015 Mar;34(1):46-52.
7. Hoppel M et al. Eur J Pharm Sci. 2015 Feb 20;68:27-35.
FDA approves topical ruxolitinib for nonsegmental vitiligo
The on July 18. The treatment, which was approved for treating mild to moderate atopic dermatitis in September 2021, is a cream formulation of ruxolitinib, a Janus kinase 1 (JAK1)/JAK2 inhibitor.
Previously, no treatment was approved to repigment patients with vitiligo, says David Rosmarin, MD, vice chair for research and education in the department of dermatology at Tufts Medical Center, Boston. “It’s important to have options that we can give to patients that are both safe and effective to get them the desired results,” Dr. Rosmarin, the lead investigator of the phase 3 clinical trials of topical ruxolitinib, said in an interview. Vitiligo is “a disease that can really affect quality of life. Some people [with vitiligo] feel as if they’re being stared at or they’re being bullied; they don’t feel confident. It can affect relationships and intimacy.”
Approval was based on the results of two phase 3 trials (TruE-V1 and TruE-V2) in 674 patients with nonsegmental vitiligo aged 12 years or older. At 24 weeks, about 30% of the patients on treatment, applied twice a day, achieved at least a 75% improvement in the facial Vitiligo Area Scoring Index (F-VASI75), compared with about 8% and 13% among those in the vehicle groups in the two trials.
At 52 weeks, about 50% of the patients treated with topical ruxolitinib achieved F-VASI75.
Also, using self-reporting as measured by the Vitiligo Noticeability Scale, about 30%-40% of patients described their vitiligo as being “a lot less noticeable” or “no longer noticeable” at week 52. Dr. Rosmarin reported the 52-week results at the 2022 annual meeting of the American Academy of Dermatology.
The trial group used 1.5% ruxolitinib cream twice daily for the full year. The vehicle group began using ruxolitinib halfway through the trial. In this group, 26.8% and 29.6% achieved F-VASI 75 at 52 weeks in the two trials.
For treating vitiligo, patients are advised to apply a thin layer of topical ruxolitinib to affected areas twice a day, “up to 10% body surface area,” according to the prescribing information, which adds: “Satisfactory patient response may require treatment … for more than 24 weeks. If the patient does not find the repigmentation meaningful by 24 weeks, the patient should be reevaluated by the health care provider.”
The most common side effects during the vehicle-controlled part of the trials were development of acne and pruritus at the application site, headache, urinary tract infections, erythema at the application site, and pyrexia, according to the company.
The approved label for topical ruxolitinib includes a boxed warning about serious infections, mortality, cancer, major adverse cardiovascular events, and thrombosis – which, the warning notes, is based on reports in patients treated with oral JAK inhibitors for inflammatory conditions.
Dr. Rosmarin believes that using this drug with other therapies, like light treatment, might yield even better responses. The available data are in patients treated with ruxolitinib as monotherapy, without complementary therapies.
William Damsky, MD, PhD, professor of dermatology and dermatopathology at Yale University, New Haven, who was not involved in the trials, said what is most exciting about this drug is its novelty. Although some topical steroids are used off-label to treat vitiligo, their efficacy is far from what’s been observed in these trials of topical ruxolitinib, he told this news organization. “It’s huge for a number of reasons. … One very big reason is it just provides some hope” for the many patients with vitiligo who, over the years, have been told “that there’s nothing that could be done for their disease, and this really changes that.”
Dr. Rosmarin reports financial relationships with over 20 pharmaceutical companies. Dr. Damsky disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
The on July 18. The treatment, which was approved for treating mild to moderate atopic dermatitis in September 2021, is a cream formulation of ruxolitinib, a Janus kinase 1 (JAK1)/JAK2 inhibitor.
Previously, no treatment was approved to repigment patients with vitiligo, says David Rosmarin, MD, vice chair for research and education in the department of dermatology at Tufts Medical Center, Boston. “It’s important to have options that we can give to patients that are both safe and effective to get them the desired results,” Dr. Rosmarin, the lead investigator of the phase 3 clinical trials of topical ruxolitinib, said in an interview. Vitiligo is “a disease that can really affect quality of life. Some people [with vitiligo] feel as if they’re being stared at or they’re being bullied; they don’t feel confident. It can affect relationships and intimacy.”
Approval was based on the results of two phase 3 trials (TruE-V1 and TruE-V2) in 674 patients with nonsegmental vitiligo aged 12 years or older. At 24 weeks, about 30% of the patients on treatment, applied twice a day, achieved at least a 75% improvement in the facial Vitiligo Area Scoring Index (F-VASI75), compared with about 8% and 13% among those in the vehicle groups in the two trials.
At 52 weeks, about 50% of the patients treated with topical ruxolitinib achieved F-VASI75.
Also, using self-reporting as measured by the Vitiligo Noticeability Scale, about 30%-40% of patients described their vitiligo as being “a lot less noticeable” or “no longer noticeable” at week 52. Dr. Rosmarin reported the 52-week results at the 2022 annual meeting of the American Academy of Dermatology.
The trial group used 1.5% ruxolitinib cream twice daily for the full year. The vehicle group began using ruxolitinib halfway through the trial. In this group, 26.8% and 29.6% achieved F-VASI 75 at 52 weeks in the two trials.
For treating vitiligo, patients are advised to apply a thin layer of topical ruxolitinib to affected areas twice a day, “up to 10% body surface area,” according to the prescribing information, which adds: “Satisfactory patient response may require treatment … for more than 24 weeks. If the patient does not find the repigmentation meaningful by 24 weeks, the patient should be reevaluated by the health care provider.”
The most common side effects during the vehicle-controlled part of the trials were development of acne and pruritus at the application site, headache, urinary tract infections, erythema at the application site, and pyrexia, according to the company.
The approved label for topical ruxolitinib includes a boxed warning about serious infections, mortality, cancer, major adverse cardiovascular events, and thrombosis – which, the warning notes, is based on reports in patients treated with oral JAK inhibitors for inflammatory conditions.
Dr. Rosmarin believes that using this drug with other therapies, like light treatment, might yield even better responses. The available data are in patients treated with ruxolitinib as monotherapy, without complementary therapies.
William Damsky, MD, PhD, professor of dermatology and dermatopathology at Yale University, New Haven, who was not involved in the trials, said what is most exciting about this drug is its novelty. Although some topical steroids are used off-label to treat vitiligo, their efficacy is far from what’s been observed in these trials of topical ruxolitinib, he told this news organization. “It’s huge for a number of reasons. … One very big reason is it just provides some hope” for the many patients with vitiligo who, over the years, have been told “that there’s nothing that could be done for their disease, and this really changes that.”
Dr. Rosmarin reports financial relationships with over 20 pharmaceutical companies. Dr. Damsky disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
The on July 18. The treatment, which was approved for treating mild to moderate atopic dermatitis in September 2021, is a cream formulation of ruxolitinib, a Janus kinase 1 (JAK1)/JAK2 inhibitor.
Previously, no treatment was approved to repigment patients with vitiligo, says David Rosmarin, MD, vice chair for research and education in the department of dermatology at Tufts Medical Center, Boston. “It’s important to have options that we can give to patients that are both safe and effective to get them the desired results,” Dr. Rosmarin, the lead investigator of the phase 3 clinical trials of topical ruxolitinib, said in an interview. Vitiligo is “a disease that can really affect quality of life. Some people [with vitiligo] feel as if they’re being stared at or they’re being bullied; they don’t feel confident. It can affect relationships and intimacy.”
Approval was based on the results of two phase 3 trials (TruE-V1 and TruE-V2) in 674 patients with nonsegmental vitiligo aged 12 years or older. At 24 weeks, about 30% of the patients on treatment, applied twice a day, achieved at least a 75% improvement in the facial Vitiligo Area Scoring Index (F-VASI75), compared with about 8% and 13% among those in the vehicle groups in the two trials.
At 52 weeks, about 50% of the patients treated with topical ruxolitinib achieved F-VASI75.
Also, using self-reporting as measured by the Vitiligo Noticeability Scale, about 30%-40% of patients described their vitiligo as being “a lot less noticeable” or “no longer noticeable” at week 52. Dr. Rosmarin reported the 52-week results at the 2022 annual meeting of the American Academy of Dermatology.
The trial group used 1.5% ruxolitinib cream twice daily for the full year. The vehicle group began using ruxolitinib halfway through the trial. In this group, 26.8% and 29.6% achieved F-VASI 75 at 52 weeks in the two trials.
For treating vitiligo, patients are advised to apply a thin layer of topical ruxolitinib to affected areas twice a day, “up to 10% body surface area,” according to the prescribing information, which adds: “Satisfactory patient response may require treatment … for more than 24 weeks. If the patient does not find the repigmentation meaningful by 24 weeks, the patient should be reevaluated by the health care provider.”
The most common side effects during the vehicle-controlled part of the trials were development of acne and pruritus at the application site, headache, urinary tract infections, erythema at the application site, and pyrexia, according to the company.
The approved label for topical ruxolitinib includes a boxed warning about serious infections, mortality, cancer, major adverse cardiovascular events, and thrombosis – which, the warning notes, is based on reports in patients treated with oral JAK inhibitors for inflammatory conditions.
Dr. Rosmarin believes that using this drug with other therapies, like light treatment, might yield even better responses. The available data are in patients treated with ruxolitinib as monotherapy, without complementary therapies.
William Damsky, MD, PhD, professor of dermatology and dermatopathology at Yale University, New Haven, who was not involved in the trials, said what is most exciting about this drug is its novelty. Although some topical steroids are used off-label to treat vitiligo, their efficacy is far from what’s been observed in these trials of topical ruxolitinib, he told this news organization. “It’s huge for a number of reasons. … One very big reason is it just provides some hope” for the many patients with vitiligo who, over the years, have been told “that there’s nothing that could be done for their disease, and this really changes that.”
Dr. Rosmarin reports financial relationships with over 20 pharmaceutical companies. Dr. Damsky disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Methotrexate’s impact on COVID-19 vaccination: New insights made
Patients who take methotrexate for a variety of immune-mediated inflammatory diseases and pause taking the drug following receipt of a COVID-19 vaccine dose did not have a higher risk of disease flare and had higher antireceptor binding domain (anti-RBD) antibody titers and increased immunogenicity when compared with continuing the drug, three recent studies suggest.
In one study, British researchers examined the effects of a 2-week break in methotrexate therapy on anti-RBD titers following receipt of a third COVID-19 vaccine dose. In their paper published in The Lancet: Respiratory Medicine, they reported results from a randomized, open-label, superiority trial that suggested pausing the drug improved immunogenicity, compared with no break.
In two trials presented at the European Alliance of Associations for Rheumatology (EULAR) 2022 Congress, a team from India set out to determine whether holding methotrexate after receiving both doses of a COVID-19 vaccine, or holding it only after the second dose, was safe and effective. They found that pausing methotrexate only following the second dose contributed to a lower flare risk, and that patients had higher anti-RBD titers when holding methotrexate for 2 weeks following each dose.
Pausing methotrexate after booster
The 2-week methotrexate break and booster vaccine dose data in the Vaccine Response On Off Methotrexate (VROOM) trial showed that after a month, the geometric mean antispike 1 (S1)-RBD antibody titer was 10,798 U/mL (95% confidence interval [CI], 8,970-12,997) in the group that continued methotrexate and 22,750 U/mL (95% CI, 19,314-26,796) in the group that suspended methotrexate; the geometric mean ratio was 2.19 (P < .0001; mixed-effects model), reported Abhishek Abhishek, MD, PhD, professor of rheumatology at the University of Nottingham in Nottingham, England, and colleagues.
Prior research showed that stopping methotrexate therapy for 2 weeks following the seasonal influenza vaccine contributed to better vaccine immunity among patients with rheumatoid arthritis, but there was no impact of stopping the drug for up to 4 weeks before vaccination on vaccine-related immunity, the researchers noted.
It is crucial in maximizing long-lasting vaccine protection in people who are possibly susceptible through immune suppression at this point in the COVID-19 vaccination regimen, the study team noted.
“Evidence from this study will be useful for policymakers, national immunization advisory committees, and specialist societies formulating recommendations on the use of methotrexate around the time of COVID-19 vaccination. This evidence will help patients and clinicians make informed choices about the risks and benefits of interrupting methotrexate treatment around the time of COVID-19 vaccination, with implications for the potential to extend such approaches to other therapeutics,” they wrote.
In American College of Rheumatology (ACR) guidance for COVID-19 vaccination, the organization advised against using standard synthetic disease-modifying antirheumatic medicines such as methotrexate “for 1-2 weeks (as disease activity allows) after each COVID-19 vaccine dose,” given the at-risk population and public health concerns, Jeffrey A. Sparks, MD, MMSc, assistant professor of medicine and associate physician at Brigham and Women’s Hospital and Harvard Medical School, Boston, and Sara K. Tedeschi, MD, MPH, assistant professor of medicine at Harvard Medical School, noted in an accompanying editorial in The Lancet: Respiratory Medicine.
However, when the ACR developed this statement, there was only one trial involving patients with rheumatoid arthritis who paused methotrexate following seasonal influenza vaccination, the editorialists said.
“Although this finding adds to the evidence base to support interruption of methotrexate after vaccination, a shared decision process is needed to weigh the possible benefit of optimizing protection from COVID-19 and the possible risk of underlying disease flare,” they added.
Dr. Abhishek and colleagues assessed 254 patients with immune-mediated inflammatory disease from dermatology and rheumatology clinics across 26 hospitals in the United Kingdom. Participants had been diagnosed with systemic lupus erythematosus, rheumatoid arthritis, atopic dermatitis, polymyalgia rheumatica, axial spondyloarthritis, and psoriasis without or with arthritis. They had also been taking up to 25 mg of methotrexate per week for 3 months or longer and had received two doses of either the Pfizer/BioNTech BNT162b2 vaccine or AstraZeneca/Oxford viral vector vaccine. The booster dose was most often the Pfizer BNT162b2 vaccine (82%). The patients’ mean age was 59 years, with females comprising 61% of the cohort. Participants were randomly assigned 1:1 to either group.
Investigators performing laboratory analysis were masked to cohort assignment, and clinical research staff, data analysts, participants, and researchers were unmasked.
The elevated antibody response of patients who suspended methotrexate was the same across different kinds of immune-mediated inflammatory disease, primary vaccination platform, SARS-CoV-2 infection history, and age.
Notably, no intervention-associated adverse events were reported, the study team noted.
The conclusions that could be drawn from the booster-dose study were limited by the trial’s modest cohort size, the small number of patients in exploratory subgroup analyses, a lack of information about differences in prescription drug behavior, and early termination’s effect on the researchers’ ability to identify differences between subgroups and in secondary outcomes, the authors noted.
Other limitations included a lack of generalizability to patients with active disease who couldn’t stop therapy and were not included in the investigation, and participants were not blinded to what group they were in, the researchers said.
Expert commentary
This current study is consistent with other studies over the last several months showing that methotrexate harms both humoral and cell-mediated COVID-19 responses, noted Kevin Winthrop, MD, MPH, professor of infectious disease and public health at Oregon Health & Science University, Portland, who was not involved in the study. “And so now the new wave of studies are like this one, where they are holding methotrexate experimentally and seeing if it makes a difference,” he said.
“The one shortcoming of this study – and so far, the studies to date – is that no one has looked at whether the experimental hold has resulted in a change in T-cell responses, which ... we are [now] recognizing [the importance of] more and more in long-term protection, particularly in severe disease. Theoretically, holding [methotrexate] might help enhance T-cell responses, but that hasn’t been shown experimentally.”
Dr. Winthrop pointed out that one might get the same benefit from holding methotrexate for 1 week instead of 2 and that there likely is a reduced risk of flare-up from underlying autoimmune disease.
It is still not certain that this benefit extends to other vaccines, Dr. Winthrop noted. “It is probably true for most vaccines that if you hold methotrexate for 1 or 2 weeks, you might see some short-term benefit in responsiveness, but you don’t know that there is any clinical meaningfulness of this. That’s going to take other long-term studies. You don’t know how long this benefit lasts.”
Pausing methotrexate during initial COVID vaccine doses
Patients with either rheumatoid arthritis or psoriatic arthritis had higher anti-RBD antibody titers when methotrexate was stopped after both doses of the AstraZeneca vaccine, or simply after the second dose, than when methotrexate was continued, according to results from two single-center, randomized controlled trials called MIVAC I and II, Anu Sreekanth, MD, of Sree Sudheendra Medical Mission in Kochi, Kerala, India, and colleagues reported at EULAR 2022.
Results from MIVAC I indicated that there was a higher flare rate when methotrexate was stopped after both vaccine doses, but there was no difference in flare rate in MIVAC II when methotrexate was stopped only after the second dose as opposed to stopping it after both doses.
In the MIVAC I trial, 158 unvaccinated patients were randomized 1:1 to a cohort in which methotrexate was held for 2 weeks after both doses and a cohort in which methotrexate was continued despite the vaccine. In MIVAC II, 157 patients continued methotrexate while receiving the first vaccine dose. These patients were subsequently randomized either to continue or to stop methotrexate for 2 weeks following the second dose.
The findings from MIVAC I demonstrated the flare rate was lower in the methotrexate-continue group than in the methotrexate-pause group (8% vs. 25%; P = .005) and that the median anti-RBD titer was significantly higher for the methotrexate-pause group than the methotrexate-continue group (2,484 vs. 1,147; P = .001).
The results from MIVAC II trial indicated that there was no difference in flare rates between the two study groups (7.9% vs. 11.8%; P = .15). Yet, the median anti-RBD titer was significantly higher in the methotrexate-pause cohort than in the methotrexate-continue cohort (2,553 vs. 990; P = .001).
The report suggests there is a flare risk when methotrexate is stopped, Dr. Sreekanth noted. “It appears more logical to hold only after the second dose, as comparable anti-RBD titers are generated” with either approach, Dr. Sreekanth said.
Expert commentary: MIVAC I and II
Inés Colmegna, MD, associate professor at McGill University in Montreal, noted that it was intriguing that the risk of flares in MIVAC II is half of that reported after each of the doses of MIVAC I. “It is also worth emphasizing that despite the reported frequency of flares, the actual disease activity [as measured by the Disease Activity Score in 28 joints] in patients who did or did not withhold methotrexate was similar.
“MIVAC I and II have practical implications as they help to adequately inform patients about the risk and benefit trade of withholding methotrexate post–COVID-19 vaccination,” Dr. Colmegna told this news organization.
“Additional information would help to [further] interpret the findings of these studies, including whether any of the participants were taking any other DMARDs; data on the severity of the flares and functional impact; analysis of factors that predict the risk of flares, such as higher doses of methotrexate; [and change in] disease activity scores pre- and postvaccination,” Dr. Colmegna concluded.
Dr. Abhishek disclosed relationships with Springer, UpTodate, Oxford, Immunotec, AstraZeneca, Inflazome, NGM Biopharmaceuticals, Menarini Pharmaceuticals, and Cadila Pharmaceuticals. Dr. Abhishek is cochair of the ACR/EULAR CPPD Classification Criteria Working Group and the OMERACT CPPD Working Group. Dr. Sparks disclosed relationships with Gilead, Boehringer Ingelheim, Amgen, Bristol-Myers Squibb, and AbbVie, unrelated to this study. Dr. Tedeschi disclosed relationships with ModernaTx and NGM Biopharmaceuticals. Dr. Winthrop disclosed a research grant and serving as a scientific consultant for Pfizer. Dr. Sreekanth and Dr. Colmegna have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Patients who take methotrexate for a variety of immune-mediated inflammatory diseases and pause taking the drug following receipt of a COVID-19 vaccine dose did not have a higher risk of disease flare and had higher antireceptor binding domain (anti-RBD) antibody titers and increased immunogenicity when compared with continuing the drug, three recent studies suggest.
In one study, British researchers examined the effects of a 2-week break in methotrexate therapy on anti-RBD titers following receipt of a third COVID-19 vaccine dose. In their paper published in The Lancet: Respiratory Medicine, they reported results from a randomized, open-label, superiority trial that suggested pausing the drug improved immunogenicity, compared with no break.
In two trials presented at the European Alliance of Associations for Rheumatology (EULAR) 2022 Congress, a team from India set out to determine whether holding methotrexate after receiving both doses of a COVID-19 vaccine, or holding it only after the second dose, was safe and effective. They found that pausing methotrexate only following the second dose contributed to a lower flare risk, and that patients had higher anti-RBD titers when holding methotrexate for 2 weeks following each dose.
Pausing methotrexate after booster
The 2-week methotrexate break and booster vaccine dose data in the Vaccine Response On Off Methotrexate (VROOM) trial showed that after a month, the geometric mean antispike 1 (S1)-RBD antibody titer was 10,798 U/mL (95% confidence interval [CI], 8,970-12,997) in the group that continued methotrexate and 22,750 U/mL (95% CI, 19,314-26,796) in the group that suspended methotrexate; the geometric mean ratio was 2.19 (P < .0001; mixed-effects model), reported Abhishek Abhishek, MD, PhD, professor of rheumatology at the University of Nottingham in Nottingham, England, and colleagues.
Prior research showed that stopping methotrexate therapy for 2 weeks following the seasonal influenza vaccine contributed to better vaccine immunity among patients with rheumatoid arthritis, but there was no impact of stopping the drug for up to 4 weeks before vaccination on vaccine-related immunity, the researchers noted.
It is crucial in maximizing long-lasting vaccine protection in people who are possibly susceptible through immune suppression at this point in the COVID-19 vaccination regimen, the study team noted.
“Evidence from this study will be useful for policymakers, national immunization advisory committees, and specialist societies formulating recommendations on the use of methotrexate around the time of COVID-19 vaccination. This evidence will help patients and clinicians make informed choices about the risks and benefits of interrupting methotrexate treatment around the time of COVID-19 vaccination, with implications for the potential to extend such approaches to other therapeutics,” they wrote.
In American College of Rheumatology (ACR) guidance for COVID-19 vaccination, the organization advised against using standard synthetic disease-modifying antirheumatic medicines such as methotrexate “for 1-2 weeks (as disease activity allows) after each COVID-19 vaccine dose,” given the at-risk population and public health concerns, Jeffrey A. Sparks, MD, MMSc, assistant professor of medicine and associate physician at Brigham and Women’s Hospital and Harvard Medical School, Boston, and Sara K. Tedeschi, MD, MPH, assistant professor of medicine at Harvard Medical School, noted in an accompanying editorial in The Lancet: Respiratory Medicine.
However, when the ACR developed this statement, there was only one trial involving patients with rheumatoid arthritis who paused methotrexate following seasonal influenza vaccination, the editorialists said.
“Although this finding adds to the evidence base to support interruption of methotrexate after vaccination, a shared decision process is needed to weigh the possible benefit of optimizing protection from COVID-19 and the possible risk of underlying disease flare,” they added.
Dr. Abhishek and colleagues assessed 254 patients with immune-mediated inflammatory disease from dermatology and rheumatology clinics across 26 hospitals in the United Kingdom. Participants had been diagnosed with systemic lupus erythematosus, rheumatoid arthritis, atopic dermatitis, polymyalgia rheumatica, axial spondyloarthritis, and psoriasis without or with arthritis. They had also been taking up to 25 mg of methotrexate per week for 3 months or longer and had received two doses of either the Pfizer/BioNTech BNT162b2 vaccine or AstraZeneca/Oxford viral vector vaccine. The booster dose was most often the Pfizer BNT162b2 vaccine (82%). The patients’ mean age was 59 years, with females comprising 61% of the cohort. Participants were randomly assigned 1:1 to either group.
Investigators performing laboratory analysis were masked to cohort assignment, and clinical research staff, data analysts, participants, and researchers were unmasked.
The elevated antibody response of patients who suspended methotrexate was the same across different kinds of immune-mediated inflammatory disease, primary vaccination platform, SARS-CoV-2 infection history, and age.
Notably, no intervention-associated adverse events were reported, the study team noted.
The conclusions that could be drawn from the booster-dose study were limited by the trial’s modest cohort size, the small number of patients in exploratory subgroup analyses, a lack of information about differences in prescription drug behavior, and early termination’s effect on the researchers’ ability to identify differences between subgroups and in secondary outcomes, the authors noted.
Other limitations included a lack of generalizability to patients with active disease who couldn’t stop therapy and were not included in the investigation, and participants were not blinded to what group they were in, the researchers said.
Expert commentary
This current study is consistent with other studies over the last several months showing that methotrexate harms both humoral and cell-mediated COVID-19 responses, noted Kevin Winthrop, MD, MPH, professor of infectious disease and public health at Oregon Health & Science University, Portland, who was not involved in the study. “And so now the new wave of studies are like this one, where they are holding methotrexate experimentally and seeing if it makes a difference,” he said.
“The one shortcoming of this study – and so far, the studies to date – is that no one has looked at whether the experimental hold has resulted in a change in T-cell responses, which ... we are [now] recognizing [the importance of] more and more in long-term protection, particularly in severe disease. Theoretically, holding [methotrexate] might help enhance T-cell responses, but that hasn’t been shown experimentally.”
Dr. Winthrop pointed out that one might get the same benefit from holding methotrexate for 1 week instead of 2 and that there likely is a reduced risk of flare-up from underlying autoimmune disease.
It is still not certain that this benefit extends to other vaccines, Dr. Winthrop noted. “It is probably true for most vaccines that if you hold methotrexate for 1 or 2 weeks, you might see some short-term benefit in responsiveness, but you don’t know that there is any clinical meaningfulness of this. That’s going to take other long-term studies. You don’t know how long this benefit lasts.”
Pausing methotrexate during initial COVID vaccine doses
Patients with either rheumatoid arthritis or psoriatic arthritis had higher anti-RBD antibody titers when methotrexate was stopped after both doses of the AstraZeneca vaccine, or simply after the second dose, than when methotrexate was continued, according to results from two single-center, randomized controlled trials called MIVAC I and II, Anu Sreekanth, MD, of Sree Sudheendra Medical Mission in Kochi, Kerala, India, and colleagues reported at EULAR 2022.
Results from MIVAC I indicated that there was a higher flare rate when methotrexate was stopped after both vaccine doses, but there was no difference in flare rate in MIVAC II when methotrexate was stopped only after the second dose as opposed to stopping it after both doses.
In the MIVAC I trial, 158 unvaccinated patients were randomized 1:1 to a cohort in which methotrexate was held for 2 weeks after both doses and a cohort in which methotrexate was continued despite the vaccine. In MIVAC II, 157 patients continued methotrexate while receiving the first vaccine dose. These patients were subsequently randomized either to continue or to stop methotrexate for 2 weeks following the second dose.
The findings from MIVAC I demonstrated the flare rate was lower in the methotrexate-continue group than in the methotrexate-pause group (8% vs. 25%; P = .005) and that the median anti-RBD titer was significantly higher for the methotrexate-pause group than the methotrexate-continue group (2,484 vs. 1,147; P = .001).
The results from MIVAC II trial indicated that there was no difference in flare rates between the two study groups (7.9% vs. 11.8%; P = .15). Yet, the median anti-RBD titer was significantly higher in the methotrexate-pause cohort than in the methotrexate-continue cohort (2,553 vs. 990; P = .001).
The report suggests there is a flare risk when methotrexate is stopped, Dr. Sreekanth noted. “It appears more logical to hold only after the second dose, as comparable anti-RBD titers are generated” with either approach, Dr. Sreekanth said.
Expert commentary: MIVAC I and II
Inés Colmegna, MD, associate professor at McGill University in Montreal, noted that it was intriguing that the risk of flares in MIVAC II is half of that reported after each of the doses of MIVAC I. “It is also worth emphasizing that despite the reported frequency of flares, the actual disease activity [as measured by the Disease Activity Score in 28 joints] in patients who did or did not withhold methotrexate was similar.
“MIVAC I and II have practical implications as they help to adequately inform patients about the risk and benefit trade of withholding methotrexate post–COVID-19 vaccination,” Dr. Colmegna told this news organization.
“Additional information would help to [further] interpret the findings of these studies, including whether any of the participants were taking any other DMARDs; data on the severity of the flares and functional impact; analysis of factors that predict the risk of flares, such as higher doses of methotrexate; [and change in] disease activity scores pre- and postvaccination,” Dr. Colmegna concluded.
Dr. Abhishek disclosed relationships with Springer, UpTodate, Oxford, Immunotec, AstraZeneca, Inflazome, NGM Biopharmaceuticals, Menarini Pharmaceuticals, and Cadila Pharmaceuticals. Dr. Abhishek is cochair of the ACR/EULAR CPPD Classification Criteria Working Group and the OMERACT CPPD Working Group. Dr. Sparks disclosed relationships with Gilead, Boehringer Ingelheim, Amgen, Bristol-Myers Squibb, and AbbVie, unrelated to this study. Dr. Tedeschi disclosed relationships with ModernaTx and NGM Biopharmaceuticals. Dr. Winthrop disclosed a research grant and serving as a scientific consultant for Pfizer. Dr. Sreekanth and Dr. Colmegna have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Patients who take methotrexate for a variety of immune-mediated inflammatory diseases and pause taking the drug following receipt of a COVID-19 vaccine dose did not have a higher risk of disease flare and had higher antireceptor binding domain (anti-RBD) antibody titers and increased immunogenicity when compared with continuing the drug, three recent studies suggest.
In one study, British researchers examined the effects of a 2-week break in methotrexate therapy on anti-RBD titers following receipt of a third COVID-19 vaccine dose. In their paper published in The Lancet: Respiratory Medicine, they reported results from a randomized, open-label, superiority trial that suggested pausing the drug improved immunogenicity, compared with no break.
In two trials presented at the European Alliance of Associations for Rheumatology (EULAR) 2022 Congress, a team from India set out to determine whether holding methotrexate after receiving both doses of a COVID-19 vaccine, or holding it only after the second dose, was safe and effective. They found that pausing methotrexate only following the second dose contributed to a lower flare risk, and that patients had higher anti-RBD titers when holding methotrexate for 2 weeks following each dose.
Pausing methotrexate after booster
The 2-week methotrexate break and booster vaccine dose data in the Vaccine Response On Off Methotrexate (VROOM) trial showed that after a month, the geometric mean antispike 1 (S1)-RBD antibody titer was 10,798 U/mL (95% confidence interval [CI], 8,970-12,997) in the group that continued methotrexate and 22,750 U/mL (95% CI, 19,314-26,796) in the group that suspended methotrexate; the geometric mean ratio was 2.19 (P < .0001; mixed-effects model), reported Abhishek Abhishek, MD, PhD, professor of rheumatology at the University of Nottingham in Nottingham, England, and colleagues.
Prior research showed that stopping methotrexate therapy for 2 weeks following the seasonal influenza vaccine contributed to better vaccine immunity among patients with rheumatoid arthritis, but there was no impact of stopping the drug for up to 4 weeks before vaccination on vaccine-related immunity, the researchers noted.
It is crucial in maximizing long-lasting vaccine protection in people who are possibly susceptible through immune suppression at this point in the COVID-19 vaccination regimen, the study team noted.
“Evidence from this study will be useful for policymakers, national immunization advisory committees, and specialist societies formulating recommendations on the use of methotrexate around the time of COVID-19 vaccination. This evidence will help patients and clinicians make informed choices about the risks and benefits of interrupting methotrexate treatment around the time of COVID-19 vaccination, with implications for the potential to extend such approaches to other therapeutics,” they wrote.
In American College of Rheumatology (ACR) guidance for COVID-19 vaccination, the organization advised against using standard synthetic disease-modifying antirheumatic medicines such as methotrexate “for 1-2 weeks (as disease activity allows) after each COVID-19 vaccine dose,” given the at-risk population and public health concerns, Jeffrey A. Sparks, MD, MMSc, assistant professor of medicine and associate physician at Brigham and Women’s Hospital and Harvard Medical School, Boston, and Sara K. Tedeschi, MD, MPH, assistant professor of medicine at Harvard Medical School, noted in an accompanying editorial in The Lancet: Respiratory Medicine.
However, when the ACR developed this statement, there was only one trial involving patients with rheumatoid arthritis who paused methotrexate following seasonal influenza vaccination, the editorialists said.
“Although this finding adds to the evidence base to support interruption of methotrexate after vaccination, a shared decision process is needed to weigh the possible benefit of optimizing protection from COVID-19 and the possible risk of underlying disease flare,” they added.
Dr. Abhishek and colleagues assessed 254 patients with immune-mediated inflammatory disease from dermatology and rheumatology clinics across 26 hospitals in the United Kingdom. Participants had been diagnosed with systemic lupus erythematosus, rheumatoid arthritis, atopic dermatitis, polymyalgia rheumatica, axial spondyloarthritis, and psoriasis without or with arthritis. They had also been taking up to 25 mg of methotrexate per week for 3 months or longer and had received two doses of either the Pfizer/BioNTech BNT162b2 vaccine or AstraZeneca/Oxford viral vector vaccine. The booster dose was most often the Pfizer BNT162b2 vaccine (82%). The patients’ mean age was 59 years, with females comprising 61% of the cohort. Participants were randomly assigned 1:1 to either group.
Investigators performing laboratory analysis were masked to cohort assignment, and clinical research staff, data analysts, participants, and researchers were unmasked.
The elevated antibody response of patients who suspended methotrexate was the same across different kinds of immune-mediated inflammatory disease, primary vaccination platform, SARS-CoV-2 infection history, and age.
Notably, no intervention-associated adverse events were reported, the study team noted.
The conclusions that could be drawn from the booster-dose study were limited by the trial’s modest cohort size, the small number of patients in exploratory subgroup analyses, a lack of information about differences in prescription drug behavior, and early termination’s effect on the researchers’ ability to identify differences between subgroups and in secondary outcomes, the authors noted.
Other limitations included a lack of generalizability to patients with active disease who couldn’t stop therapy and were not included in the investigation, and participants were not blinded to what group they were in, the researchers said.
Expert commentary
This current study is consistent with other studies over the last several months showing that methotrexate harms both humoral and cell-mediated COVID-19 responses, noted Kevin Winthrop, MD, MPH, professor of infectious disease and public health at Oregon Health & Science University, Portland, who was not involved in the study. “And so now the new wave of studies are like this one, where they are holding methotrexate experimentally and seeing if it makes a difference,” he said.
“The one shortcoming of this study – and so far, the studies to date – is that no one has looked at whether the experimental hold has resulted in a change in T-cell responses, which ... we are [now] recognizing [the importance of] more and more in long-term protection, particularly in severe disease. Theoretically, holding [methotrexate] might help enhance T-cell responses, but that hasn’t been shown experimentally.”
Dr. Winthrop pointed out that one might get the same benefit from holding methotrexate for 1 week instead of 2 and that there likely is a reduced risk of flare-up from underlying autoimmune disease.
It is still not certain that this benefit extends to other vaccines, Dr. Winthrop noted. “It is probably true for most vaccines that if you hold methotrexate for 1 or 2 weeks, you might see some short-term benefit in responsiveness, but you don’t know that there is any clinical meaningfulness of this. That’s going to take other long-term studies. You don’t know how long this benefit lasts.”
Pausing methotrexate during initial COVID vaccine doses
Patients with either rheumatoid arthritis or psoriatic arthritis had higher anti-RBD antibody titers when methotrexate was stopped after both doses of the AstraZeneca vaccine, or simply after the second dose, than when methotrexate was continued, according to results from two single-center, randomized controlled trials called MIVAC I and II, Anu Sreekanth, MD, of Sree Sudheendra Medical Mission in Kochi, Kerala, India, and colleagues reported at EULAR 2022.
Results from MIVAC I indicated that there was a higher flare rate when methotrexate was stopped after both vaccine doses, but there was no difference in flare rate in MIVAC II when methotrexate was stopped only after the second dose as opposed to stopping it after both doses.
In the MIVAC I trial, 158 unvaccinated patients were randomized 1:1 to a cohort in which methotrexate was held for 2 weeks after both doses and a cohort in which methotrexate was continued despite the vaccine. In MIVAC II, 157 patients continued methotrexate while receiving the first vaccine dose. These patients were subsequently randomized either to continue or to stop methotrexate for 2 weeks following the second dose.
The findings from MIVAC I demonstrated the flare rate was lower in the methotrexate-continue group than in the methotrexate-pause group (8% vs. 25%; P = .005) and that the median anti-RBD titer was significantly higher for the methotrexate-pause group than the methotrexate-continue group (2,484 vs. 1,147; P = .001).
The results from MIVAC II trial indicated that there was no difference in flare rates between the two study groups (7.9% vs. 11.8%; P = .15). Yet, the median anti-RBD titer was significantly higher in the methotrexate-pause cohort than in the methotrexate-continue cohort (2,553 vs. 990; P = .001).
The report suggests there is a flare risk when methotrexate is stopped, Dr. Sreekanth noted. “It appears more logical to hold only after the second dose, as comparable anti-RBD titers are generated” with either approach, Dr. Sreekanth said.
Expert commentary: MIVAC I and II
Inés Colmegna, MD, associate professor at McGill University in Montreal, noted that it was intriguing that the risk of flares in MIVAC II is half of that reported after each of the doses of MIVAC I. “It is also worth emphasizing that despite the reported frequency of flares, the actual disease activity [as measured by the Disease Activity Score in 28 joints] in patients who did or did not withhold methotrexate was similar.
“MIVAC I and II have practical implications as they help to adequately inform patients about the risk and benefit trade of withholding methotrexate post–COVID-19 vaccination,” Dr. Colmegna told this news organization.
“Additional information would help to [further] interpret the findings of these studies, including whether any of the participants were taking any other DMARDs; data on the severity of the flares and functional impact; analysis of factors that predict the risk of flares, such as higher doses of methotrexate; [and change in] disease activity scores pre- and postvaccination,” Dr. Colmegna concluded.
Dr. Abhishek disclosed relationships with Springer, UpTodate, Oxford, Immunotec, AstraZeneca, Inflazome, NGM Biopharmaceuticals, Menarini Pharmaceuticals, and Cadila Pharmaceuticals. Dr. Abhishek is cochair of the ACR/EULAR CPPD Classification Criteria Working Group and the OMERACT CPPD Working Group. Dr. Sparks disclosed relationships with Gilead, Boehringer Ingelheim, Amgen, Bristol-Myers Squibb, and AbbVie, unrelated to this study. Dr. Tedeschi disclosed relationships with ModernaTx and NGM Biopharmaceuticals. Dr. Winthrop disclosed a research grant and serving as a scientific consultant for Pfizer. Dr. Sreekanth and Dr. Colmegna have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Think of pediatric morphea as a systemic, chronic disease, expert advises
INDIANAPOLIS – In the opinion of Elena Pope, MD, MSc,
“There is no correlation between the extent and activity of skin lesions and the presence, severity, and activity of extracutaneous manifestations,” Dr. Pope, professor of pediatrics at the University of Toronto and division head of pediatric dermatology at the Hospital for Sick Children in Toronto, said during the annual meeting of the Society for Pediatric Dermatology. “Treatment needs to be tailored to the extent of cutaneous manifestations, and I think we need to be aware of and address the impact on patients’ quality of life,” she added. There is also a need for more research “on targeted and better-tolerated therapies to put a stop to the progression of disease.”
Congenital morphea is a form of localized scleroderma that presents at birth but can be confused with port wine stain. Results from a multicenter retrospective review of 25 cases conducted by Dr. Pope and colleagues found that the median age at diagnosis was 2.9 years and 76% had linear-type lesions. In addition, 48% had extracutaneous involvement (all of these patients had linear morphea), most commonly of the central nervous system.
“It’s important to realize these lesions may become active over time,” Dr. Pope said. “In my experience, there are two different courses. Either you have innocuous lesions when the patients are born and they may become active around 3-4 years of age, or you have early intrauterine involvement, with lesions inactive at birth but with potential for significant damage in utero.”
She cautioned against treating a suspected port wine stain lesion with laser until congenital morphea is ruled out. “I’m aware of at least one lawsuit of a child where someone used a laser in a child who had progression with significant sclerosis,” she said. “The parents assumed it was the use of the laser that led to the progression, not the actual disease.”
Extracutaneous manifestations are common in morphea patients. A multicenter study of 750 patients with juvenile scleroderma found that 22% had extracutaneous manifestations. Almost half of patients (47%) had arthritis, but 17% had neurologic findings such as seizures and headaches, 9% had vascular manifestations, and 8% had uveitis. Subsequent studies found that neurological disease affects between 11% and 19% of cases, especially in those involving the head and neck.
“There is a wide range of manifestations from headache and neuropsychiatric changes to brain atrophy, seizures, and CNS cavernoma,” Dr. Pope said. “There also can be orthodental involvement such as malocclusion. It’s important to do a brain MRI, eye exam for uveitis, and don’t forget the orthodental assessment.”
She recalled a 10-year-old boy who presented to the Hospital for Sick Children with tissue loss on the forehead and eyebrow and eyelashes. He had no other congenital morphea symptoms and the MRI was normal, but the eye exam revealed uveitis. “It’s important to remember that uveitis is asymptomatic, so unless you look for it, you’re not going to find it,” she said.
According to unpublished data in 42 congenital morphea patients with lesions limited to the head and neck, who underwent MRI imaging at the Hospital for Sick Children, 57% had CNS changes that were ipsilateral in 68% of cases. “White matter changes were the most common, and to our surprise, there were patients who had progressive CNS disease, including CNS vasculitis, new lesions, and enhancement of prior stable lesions,” Dr. Pope said.
She recalled the case of an 8-year-old boy who presented to the hospital with intractable seizures. Upon completion of the MRI, one of the radiologists noted that the imaging showed subtle thinning of the forehead, and he was referred to Dr. Pope and colleagues for assessment. In the span of 4 years, despite aggressive treatment, the boy’s CNS disease progressed. “There was more enhancement, more tissue loss, his seizures are very hard to control, and he has many neurodevelopmental changes,” she recalled. “What I learned from this case is that skin activity does not correlate with imaging. Don’t assume that just because the skin is burnt out that the CNS will be the same. Also, the extent of skin disease does not predict involvement or progression of the CNS.”
Linear lesions on the lower extremities are a harbinger of orthopedic complications, which can occur in about half of patients. Joint contractures in this subset of patients are seen in about 81% of cases, while other sequelae can include arthritis, limb atrophy, leg-leg discrepancy, and angular deformity. “About 14% of patients require intervention,” Dr. Pope said. “In terms of working those patients up, you need to do an MRI and assess the extent of muscle and fascial involvement. Early physiotherapy and an orthopedic evaluation are also recommended.”
As for possible markers of morphea, antinuclear antibody is positive in 22%-68% of cases and correlates with disease severity, extracutaneous manifestations, and disease flare-up. Antihistone antibodies (AHA) are positive in about 47% of cases, “and that tends to correlate with the extent of skin and muscle involvement,” Dr. Pope said. “Anti–double-stranded DNA correlates with extent of disease, but the only known biomarker to date that correlates with disease activity is CXCL9/10. This has been documented in the skin as well as in the blood. So, this marker may help us determine if the patient needs to be treated or not.”
Treatments
For treatment of active localized disease, topical medications are helpful in some cases. Options include topical steroids, calcipotriol with or without betamethasone, imiquimod, and tacrolimus. “In my experience the combination of calcipotriol with betamethasone is best,” she said. “It really shuts down the activity fairly soon, and you can scale down to calcipotriol alone. I don’t find imiquimod very helpful for active lesions, although it has a role for inactive lesions.”
For patients with linear or generalized/mixed disease, “the combination of methotrexate and corticosteroids or methotrexate alone is probably the way to go,” Dr. Pope said. “The addition of steroids really depends on where the lesion is and how worried you are about other problems.”
According to the best available literature, 88% of patients should respond to treatment with methotrexate (MTX) and/or steroids within 3-6 months, and 74% within 3 months. “If they don’t, you have to wonder if the patient’s taking the medication, or you need to think about other alternative treatments,” she said. “Complete remission is possible in most of the patients, and the longer you treat the more you will see that. On average, most of us treat patients for about 3 years, but there are treatment failures as well. This can occur in up to 16% of patients.”
As for second-line treatment agents for congenital morphea, clinicians often turn to mycophenolate mofetil (MMF). Results from a retrospective longitudinal study of juvenile localized scleroderma patients found that after a mean of 9 years 91% of patients on MMF and 100% of patients on MTX had inactive disease. “There were no differences in relapse rates, although MMF seems to have a more sustained long-term effect and overall is better tolerated,” said Dr. Pope, who was not involved with the study. “However, it’s more immunosuppressive than MTX, which is important, especially in the era of COVID-19. You also need to think about the potential for more hematological suppression with MMF use.” If standard therapy fails, there is anecdotal data supporting the use of abatacept (which suppresses the T-cell activity in affected patients), tofacitinib (which inhibits transforming growth factor–beta), or dupilumab (which inhibits interleukin-4).
Dr. Pope emphasized the effect congenital morphea has on quality of life. Remarks from patients with facial morphea and their parents who participated in a focus group on the topic organized by the Hospital for Sick Children included, “You just want to stay inside because you are afraid of what people will say,” “They laugh at her. They make fun of her, and it’s terrible,” and “MTX makes me feel weird. I would throw up, feel dizzy.”
“You have to take that into consideration, because we cannot make the treatment worse than the disease,” Dr. Pope said. “There are many domains where patients could be affected, including skin symptoms, physical functioning, body image and social support, side effects of medication, and presence of extracutaneous manifestations. Predictors of poor quality of life include female sex and involvement of hands and feet.”
Dr. Pope disclosed that she has received grants/research support from AbbVie, Centocor, and Amgen. She has also received consulting fees from AbbVie, Sanofi, Novartis, Boehringer-Ingelheim, Phoenix, Amryt Pharma, and Timber Pharmaceuticals.
INDIANAPOLIS – In the opinion of Elena Pope, MD, MSc,
“There is no correlation between the extent and activity of skin lesions and the presence, severity, and activity of extracutaneous manifestations,” Dr. Pope, professor of pediatrics at the University of Toronto and division head of pediatric dermatology at the Hospital for Sick Children in Toronto, said during the annual meeting of the Society for Pediatric Dermatology. “Treatment needs to be tailored to the extent of cutaneous manifestations, and I think we need to be aware of and address the impact on patients’ quality of life,” she added. There is also a need for more research “on targeted and better-tolerated therapies to put a stop to the progression of disease.”
Congenital morphea is a form of localized scleroderma that presents at birth but can be confused with port wine stain. Results from a multicenter retrospective review of 25 cases conducted by Dr. Pope and colleagues found that the median age at diagnosis was 2.9 years and 76% had linear-type lesions. In addition, 48% had extracutaneous involvement (all of these patients had linear morphea), most commonly of the central nervous system.
“It’s important to realize these lesions may become active over time,” Dr. Pope said. “In my experience, there are two different courses. Either you have innocuous lesions when the patients are born and they may become active around 3-4 years of age, or you have early intrauterine involvement, with lesions inactive at birth but with potential for significant damage in utero.”
She cautioned against treating a suspected port wine stain lesion with laser until congenital morphea is ruled out. “I’m aware of at least one lawsuit of a child where someone used a laser in a child who had progression with significant sclerosis,” she said. “The parents assumed it was the use of the laser that led to the progression, not the actual disease.”
Extracutaneous manifestations are common in morphea patients. A multicenter study of 750 patients with juvenile scleroderma found that 22% had extracutaneous manifestations. Almost half of patients (47%) had arthritis, but 17% had neurologic findings such as seizures and headaches, 9% had vascular manifestations, and 8% had uveitis. Subsequent studies found that neurological disease affects between 11% and 19% of cases, especially in those involving the head and neck.
“There is a wide range of manifestations from headache and neuropsychiatric changes to brain atrophy, seizures, and CNS cavernoma,” Dr. Pope said. “There also can be orthodental involvement such as malocclusion. It’s important to do a brain MRI, eye exam for uveitis, and don’t forget the orthodental assessment.”
She recalled a 10-year-old boy who presented to the Hospital for Sick Children with tissue loss on the forehead and eyebrow and eyelashes. He had no other congenital morphea symptoms and the MRI was normal, but the eye exam revealed uveitis. “It’s important to remember that uveitis is asymptomatic, so unless you look for it, you’re not going to find it,” she said.
According to unpublished data in 42 congenital morphea patients with lesions limited to the head and neck, who underwent MRI imaging at the Hospital for Sick Children, 57% had CNS changes that were ipsilateral in 68% of cases. “White matter changes were the most common, and to our surprise, there were patients who had progressive CNS disease, including CNS vasculitis, new lesions, and enhancement of prior stable lesions,” Dr. Pope said.
She recalled the case of an 8-year-old boy who presented to the hospital with intractable seizures. Upon completion of the MRI, one of the radiologists noted that the imaging showed subtle thinning of the forehead, and he was referred to Dr. Pope and colleagues for assessment. In the span of 4 years, despite aggressive treatment, the boy’s CNS disease progressed. “There was more enhancement, more tissue loss, his seizures are very hard to control, and he has many neurodevelopmental changes,” she recalled. “What I learned from this case is that skin activity does not correlate with imaging. Don’t assume that just because the skin is burnt out that the CNS will be the same. Also, the extent of skin disease does not predict involvement or progression of the CNS.”
Linear lesions on the lower extremities are a harbinger of orthopedic complications, which can occur in about half of patients. Joint contractures in this subset of patients are seen in about 81% of cases, while other sequelae can include arthritis, limb atrophy, leg-leg discrepancy, and angular deformity. “About 14% of patients require intervention,” Dr. Pope said. “In terms of working those patients up, you need to do an MRI and assess the extent of muscle and fascial involvement. Early physiotherapy and an orthopedic evaluation are also recommended.”
As for possible markers of morphea, antinuclear antibody is positive in 22%-68% of cases and correlates with disease severity, extracutaneous manifestations, and disease flare-up. Antihistone antibodies (AHA) are positive in about 47% of cases, “and that tends to correlate with the extent of skin and muscle involvement,” Dr. Pope said. “Anti–double-stranded DNA correlates with extent of disease, but the only known biomarker to date that correlates with disease activity is CXCL9/10. This has been documented in the skin as well as in the blood. So, this marker may help us determine if the patient needs to be treated or not.”
Treatments
For treatment of active localized disease, topical medications are helpful in some cases. Options include topical steroids, calcipotriol with or without betamethasone, imiquimod, and tacrolimus. “In my experience the combination of calcipotriol with betamethasone is best,” she said. “It really shuts down the activity fairly soon, and you can scale down to calcipotriol alone. I don’t find imiquimod very helpful for active lesions, although it has a role for inactive lesions.”
For patients with linear or generalized/mixed disease, “the combination of methotrexate and corticosteroids or methotrexate alone is probably the way to go,” Dr. Pope said. “The addition of steroids really depends on where the lesion is and how worried you are about other problems.”
According to the best available literature, 88% of patients should respond to treatment with methotrexate (MTX) and/or steroids within 3-6 months, and 74% within 3 months. “If they don’t, you have to wonder if the patient’s taking the medication, or you need to think about other alternative treatments,” she said. “Complete remission is possible in most of the patients, and the longer you treat the more you will see that. On average, most of us treat patients for about 3 years, but there are treatment failures as well. This can occur in up to 16% of patients.”
As for second-line treatment agents for congenital morphea, clinicians often turn to mycophenolate mofetil (MMF). Results from a retrospective longitudinal study of juvenile localized scleroderma patients found that after a mean of 9 years 91% of patients on MMF and 100% of patients on MTX had inactive disease. “There were no differences in relapse rates, although MMF seems to have a more sustained long-term effect and overall is better tolerated,” said Dr. Pope, who was not involved with the study. “However, it’s more immunosuppressive than MTX, which is important, especially in the era of COVID-19. You also need to think about the potential for more hematological suppression with MMF use.” If standard therapy fails, there is anecdotal data supporting the use of abatacept (which suppresses the T-cell activity in affected patients), tofacitinib (which inhibits transforming growth factor–beta), or dupilumab (which inhibits interleukin-4).
Dr. Pope emphasized the effect congenital morphea has on quality of life. Remarks from patients with facial morphea and their parents who participated in a focus group on the topic organized by the Hospital for Sick Children included, “You just want to stay inside because you are afraid of what people will say,” “They laugh at her. They make fun of her, and it’s terrible,” and “MTX makes me feel weird. I would throw up, feel dizzy.”
“You have to take that into consideration, because we cannot make the treatment worse than the disease,” Dr. Pope said. “There are many domains where patients could be affected, including skin symptoms, physical functioning, body image and social support, side effects of medication, and presence of extracutaneous manifestations. Predictors of poor quality of life include female sex and involvement of hands and feet.”
Dr. Pope disclosed that she has received grants/research support from AbbVie, Centocor, and Amgen. She has also received consulting fees from AbbVie, Sanofi, Novartis, Boehringer-Ingelheim, Phoenix, Amryt Pharma, and Timber Pharmaceuticals.
INDIANAPOLIS – In the opinion of Elena Pope, MD, MSc,
“There is no correlation between the extent and activity of skin lesions and the presence, severity, and activity of extracutaneous manifestations,” Dr. Pope, professor of pediatrics at the University of Toronto and division head of pediatric dermatology at the Hospital for Sick Children in Toronto, said during the annual meeting of the Society for Pediatric Dermatology. “Treatment needs to be tailored to the extent of cutaneous manifestations, and I think we need to be aware of and address the impact on patients’ quality of life,” she added. There is also a need for more research “on targeted and better-tolerated therapies to put a stop to the progression of disease.”
Congenital morphea is a form of localized scleroderma that presents at birth but can be confused with port wine stain. Results from a multicenter retrospective review of 25 cases conducted by Dr. Pope and colleagues found that the median age at diagnosis was 2.9 years and 76% had linear-type lesions. In addition, 48% had extracutaneous involvement (all of these patients had linear morphea), most commonly of the central nervous system.
“It’s important to realize these lesions may become active over time,” Dr. Pope said. “In my experience, there are two different courses. Either you have innocuous lesions when the patients are born and they may become active around 3-4 years of age, or you have early intrauterine involvement, with lesions inactive at birth but with potential for significant damage in utero.”
She cautioned against treating a suspected port wine stain lesion with laser until congenital morphea is ruled out. “I’m aware of at least one lawsuit of a child where someone used a laser in a child who had progression with significant sclerosis,” she said. “The parents assumed it was the use of the laser that led to the progression, not the actual disease.”
Extracutaneous manifestations are common in morphea patients. A multicenter study of 750 patients with juvenile scleroderma found that 22% had extracutaneous manifestations. Almost half of patients (47%) had arthritis, but 17% had neurologic findings such as seizures and headaches, 9% had vascular manifestations, and 8% had uveitis. Subsequent studies found that neurological disease affects between 11% and 19% of cases, especially in those involving the head and neck.
“There is a wide range of manifestations from headache and neuropsychiatric changes to brain atrophy, seizures, and CNS cavernoma,” Dr. Pope said. “There also can be orthodental involvement such as malocclusion. It’s important to do a brain MRI, eye exam for uveitis, and don’t forget the orthodental assessment.”
She recalled a 10-year-old boy who presented to the Hospital for Sick Children with tissue loss on the forehead and eyebrow and eyelashes. He had no other congenital morphea symptoms and the MRI was normal, but the eye exam revealed uveitis. “It’s important to remember that uveitis is asymptomatic, so unless you look for it, you’re not going to find it,” she said.
According to unpublished data in 42 congenital morphea patients with lesions limited to the head and neck, who underwent MRI imaging at the Hospital for Sick Children, 57% had CNS changes that were ipsilateral in 68% of cases. “White matter changes were the most common, and to our surprise, there were patients who had progressive CNS disease, including CNS vasculitis, new lesions, and enhancement of prior stable lesions,” Dr. Pope said.
She recalled the case of an 8-year-old boy who presented to the hospital with intractable seizures. Upon completion of the MRI, one of the radiologists noted that the imaging showed subtle thinning of the forehead, and he was referred to Dr. Pope and colleagues for assessment. In the span of 4 years, despite aggressive treatment, the boy’s CNS disease progressed. “There was more enhancement, more tissue loss, his seizures are very hard to control, and he has many neurodevelopmental changes,” she recalled. “What I learned from this case is that skin activity does not correlate with imaging. Don’t assume that just because the skin is burnt out that the CNS will be the same. Also, the extent of skin disease does not predict involvement or progression of the CNS.”
Linear lesions on the lower extremities are a harbinger of orthopedic complications, which can occur in about half of patients. Joint contractures in this subset of patients are seen in about 81% of cases, while other sequelae can include arthritis, limb atrophy, leg-leg discrepancy, and angular deformity. “About 14% of patients require intervention,” Dr. Pope said. “In terms of working those patients up, you need to do an MRI and assess the extent of muscle and fascial involvement. Early physiotherapy and an orthopedic evaluation are also recommended.”
As for possible markers of morphea, antinuclear antibody is positive in 22%-68% of cases and correlates with disease severity, extracutaneous manifestations, and disease flare-up. Antihistone antibodies (AHA) are positive in about 47% of cases, “and that tends to correlate with the extent of skin and muscle involvement,” Dr. Pope said. “Anti–double-stranded DNA correlates with extent of disease, but the only known biomarker to date that correlates with disease activity is CXCL9/10. This has been documented in the skin as well as in the blood. So, this marker may help us determine if the patient needs to be treated or not.”
Treatments
For treatment of active localized disease, topical medications are helpful in some cases. Options include topical steroids, calcipotriol with or without betamethasone, imiquimod, and tacrolimus. “In my experience the combination of calcipotriol with betamethasone is best,” she said. “It really shuts down the activity fairly soon, and you can scale down to calcipotriol alone. I don’t find imiquimod very helpful for active lesions, although it has a role for inactive lesions.”
For patients with linear or generalized/mixed disease, “the combination of methotrexate and corticosteroids or methotrexate alone is probably the way to go,” Dr. Pope said. “The addition of steroids really depends on where the lesion is and how worried you are about other problems.”
According to the best available literature, 88% of patients should respond to treatment with methotrexate (MTX) and/or steroids within 3-6 months, and 74% within 3 months. “If they don’t, you have to wonder if the patient’s taking the medication, or you need to think about other alternative treatments,” she said. “Complete remission is possible in most of the patients, and the longer you treat the more you will see that. On average, most of us treat patients for about 3 years, but there are treatment failures as well. This can occur in up to 16% of patients.”
As for second-line treatment agents for congenital morphea, clinicians often turn to mycophenolate mofetil (MMF). Results from a retrospective longitudinal study of juvenile localized scleroderma patients found that after a mean of 9 years 91% of patients on MMF and 100% of patients on MTX had inactive disease. “There were no differences in relapse rates, although MMF seems to have a more sustained long-term effect and overall is better tolerated,” said Dr. Pope, who was not involved with the study. “However, it’s more immunosuppressive than MTX, which is important, especially in the era of COVID-19. You also need to think about the potential for more hematological suppression with MMF use.” If standard therapy fails, there is anecdotal data supporting the use of abatacept (which suppresses the T-cell activity in affected patients), tofacitinib (which inhibits transforming growth factor–beta), or dupilumab (which inhibits interleukin-4).
Dr. Pope emphasized the effect congenital morphea has on quality of life. Remarks from patients with facial morphea and their parents who participated in a focus group on the topic organized by the Hospital for Sick Children included, “You just want to stay inside because you are afraid of what people will say,” “They laugh at her. They make fun of her, and it’s terrible,” and “MTX makes me feel weird. I would throw up, feel dizzy.”
“You have to take that into consideration, because we cannot make the treatment worse than the disease,” Dr. Pope said. “There are many domains where patients could be affected, including skin symptoms, physical functioning, body image and social support, side effects of medication, and presence of extracutaneous manifestations. Predictors of poor quality of life include female sex and involvement of hands and feet.”
Dr. Pope disclosed that she has received grants/research support from AbbVie, Centocor, and Amgen. She has also received consulting fees from AbbVie, Sanofi, Novartis, Boehringer-Ingelheim, Phoenix, Amryt Pharma, and Timber Pharmaceuticals.
AT SPD 2022
Fungated Eroded Plaque on the Arm
The Diagnosis: Cutaneous Blastomycosis
A skin biopsy and fungal cultures confirmed the diagnosis of cutaneous blastomycosis. Grocott- Gomori methenamine-silver staining highlighted fungal organisms with refractile walls and broad-based budding consistent with cutaneous blastomycosis (Figure 1). The histopathologic specimen also demonstrated marked pseudoepitheliomatous hyperplasia (Figure 2A) with neutrophilic microabscesses (Figure 2B). Acid-fast bacillus and Fite staining were negative for bacterial organisms. A fungal culture was positive for Blastomyces dermatitidis. Urine and serum blastomycosis antigen were positive. Although Histoplasma serum antigen also was positive, this likely was from cross-reactivity. Chest radiography was negative for lung involvement, and the patient displayed no neurologic symptoms. He was started on oral itraconazole therapy for the treatment of cutaneous blastomycosis.
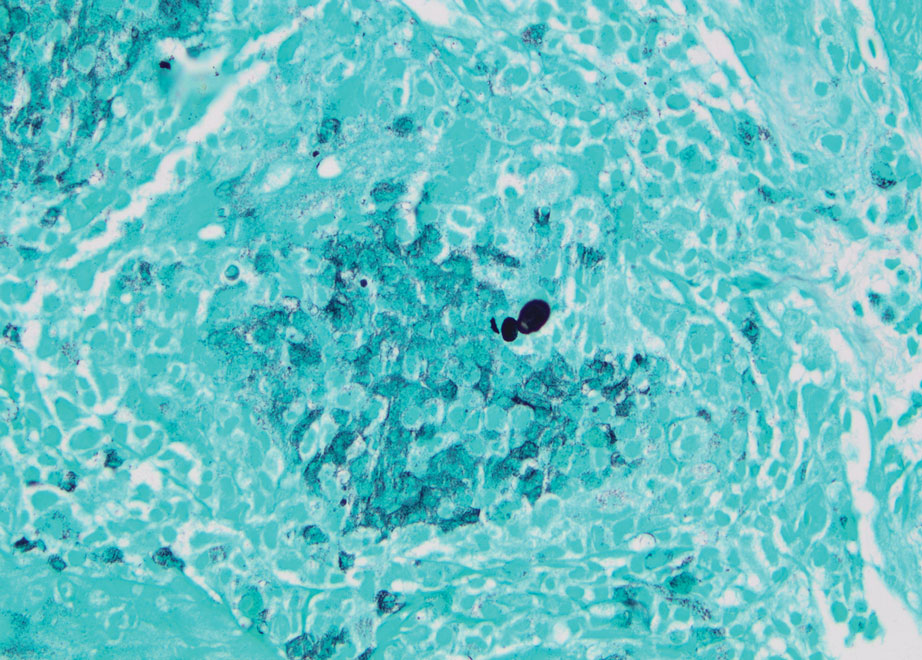
Blastomyces dermatitidis, the causative organism of blastomycosis, is endemic to the Ohio and Mississippi River valleys, Great Lakes region, and southeastern United States. It is a thermally dimorphic fungus found in soils that grows as a mold at 25 °C and yeast at 37 °C. Primary infection of the lungs—blastomycosis pneumonia—often is the only clinical manifestation1; however, subsequent hematogenous dissemination to extrapulmonary sites such as the skin, bones, and genitourinary system can occur. Cutaneous blastomycosis, the most common extrapulmonary manifestation, typically follows pulmonary infection. In rare cases, it can occur from direct inoculation.2,3 Skin lesions can occur anywhere but frequently are found on exposed surfaces of the head, neck, and extremities. Lesions classically present as verrucous crusting plaques with draining microabscesses. Violaceous nodules, ulcers, and pustules also may occur.1
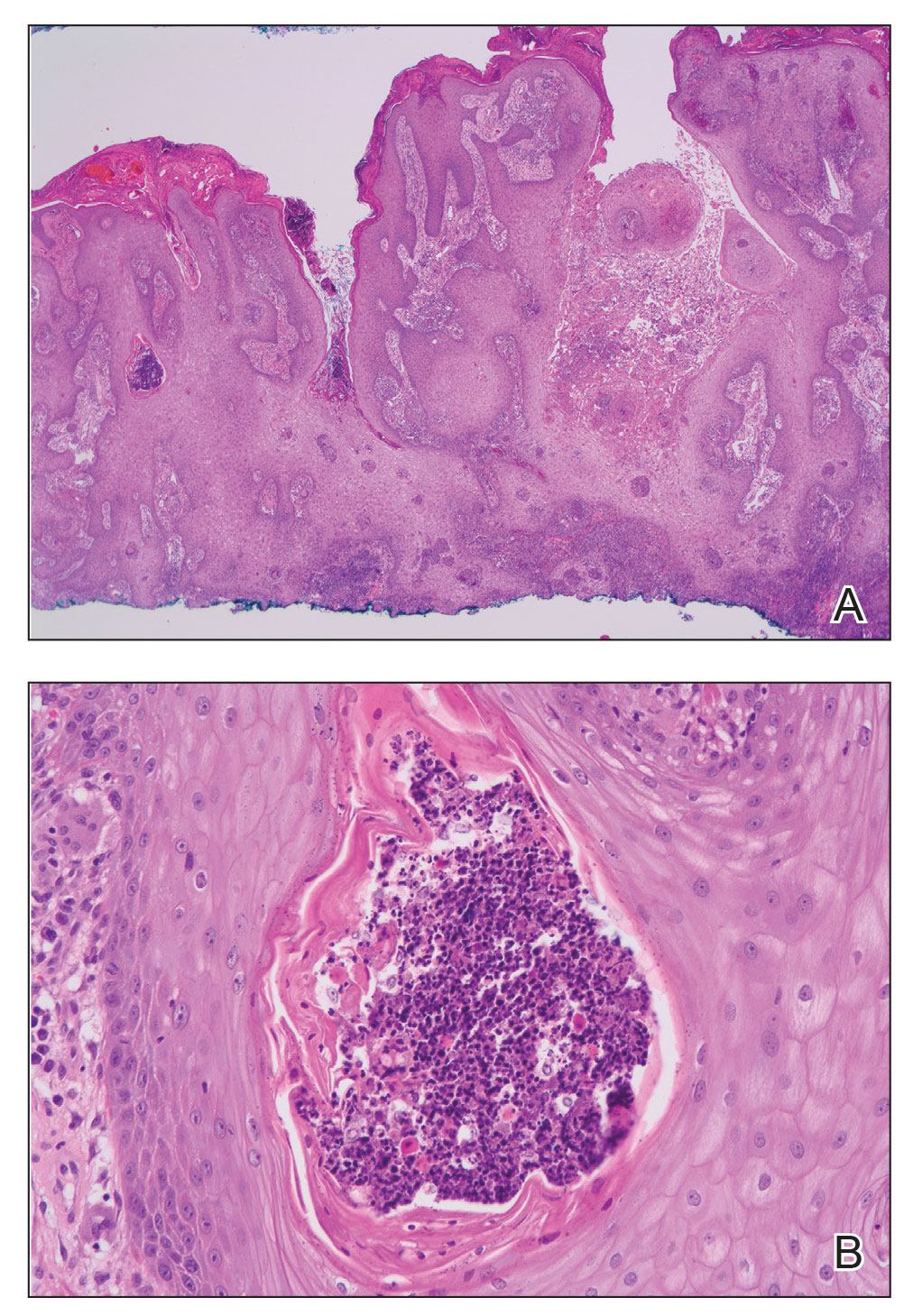
Diagnosis involves obtaining a thorough history of possible environmental exposures such as the patient’s geographic area of residence, occupation, and outdoor activities involving soil or decaying wood. Because blastomycosis can remain latent, remote exposures are relevant. Definitive diagnosis of cutaneous blastomycosis involves skin biopsy of the lesion with fungal culture, but the yeast’s distinctive thick wall and broad-based budding seen with periodic acid–Schiff or Grocott-Gomori methenamine-silver staining provides a rapid presumptive diagnosis.3 Pseudoepitheliomatous hyperplasia and microabscesses also are characteristic features.2 Urine antigen testing for a component of the polysaccharide cell wall has a sensitivity of 93% but a lower specificity of 79% due to cross-reactivity with histoplasmosis.4 Treatment consists of itraconazole for mild to moderate blastomycosis or amphotericin B for those with severe disease or central nervous system involvement or those who are immunosuppressed.1
The differential diagnosis for our patient’s lesion included infectious vs neoplastic etiologies. Histoplasma capsulatum, the dimorphic fungus that causes histoplasmosis, also is endemic to the Ohio and Mississippi River valleys. It is found in soil and droppings of some bats and birds such as chickens and pigeons. Similar to blastomycosis, the primary infection site most commonly is the lungs. It subsequently may disseminate to the skin or less commonly via direct inoculation of injured skin. It can present as papules, plaques, ulcers, purpura, or abscesses. Unlike blastomycosis, tissue biopsy of a cutaneous lesion reveals granuloma formation and distinctive oval, narrow-based budding yeast.5 Atypical mycobacteria are another source of infection to consider. For example, cutaneous Mycobacterium kansasii may present as papules and pustules forming verrucous or granulomatous plaques and ulceration. Histopathologic findings distinguishing mycobacterial infection from blastomycosis include granulomas and acid-fast bacilli in histiocytes.6
Noninfectious etiologies in the differential may include cutaneous squamous cell carcinoma or pemphigus vegetans. Squamous cell carcinoma may present with a broad range of clinical features—papules, plaques, or nodules with smooth, scaly, verrucous, or ulcerative secondary features all are possible presentations.7 Fairskinned individuals, such as our patient, would be at a higher risk in sun-damaged skin. Histologically, cutaneous squamous cell carcinoma is defined as an invasion of the dermis by neoplastic squamous epithelial cells in the form of cords, sheets, individual cells, nodules, or cystic structures.7 Pemphigus vegetans is the rarest variant of a group of autoimmune vesiculobullous diseases known as pemphigus. It can be differentiated from the most common variant—pemphigus vulgaris—by the presence of vegetative plaques in intertriginous areas. However, these verrucous vegetations can be misleading and make clinical diagnosis difficult. Histopathologic findings of hyperkeratosis, pseudoepitheliomatous hyperplasia, papillomatosis, and acantholysis with a suprabasal cleft would confirm the diagnosis.8
In summary, cutaneous blastomycosis classically presents as verrucous crusting plaques, as seen in our patient. It is important to conduct a thorough history for environmental exposures, but definitive diagnosis of cutaneous blastomycosis involves skin biopsy with fungal culture. Treatment depends on the severity of disease and organ involvement. Itraconazole would be appropriate for mild to moderate blastomycosis.
- Miceli A, Krishnamurthy K. Blastomycosis. StatPearls. StatPearls Publishing; 2022. Accessed June 21, 2022. https://www.ncbi.nlm.nih.gov/books/NBK441987/
- Gray NA, Baddour LM. Cutaneous inoculation blastomycosis. Clin Infect Dis. 2002;34:E44-E49.
- Schwartz IS, Kauffman CA. Blastomycosis. Semin Respir Crit Care Med. 2020;41:31-41. doi:10.1055/s-0039-3400281
- Castillo CG, Kauffman CA, Miceli MH. Blastomycosis. Infect Dis Clin North Am. 2016;30:247-264. doi:10.1016/j.idc.2015.10.002
- Raggio B. Primary cutaneous histoplasmosis. Ear Nose Throat J. 2018;97:346-348.
- Bhambri S, Bhambri A, Del Rosso JQ. Atypical mycobacterial cutaneous infections. Dermatol Clin. 2009;27:63-73. doi:10.1016/j.det.2008.07.009
- Parekh V, Seykora JT. Cutaneous squamous cell carcinoma. Clin Lab Med. 2017;37:503-525. doi:10.1016/j.cll.2017.06.003
- Messersmith L, Krauland K. Pemphigus vegetans. StatPearls. StatPearls Publishing; 2022. Accessed June 21, 2022. https://www.ncbi.nlm.nih.gov/books/NBK545229/
The Diagnosis: Cutaneous Blastomycosis
A skin biopsy and fungal cultures confirmed the diagnosis of cutaneous blastomycosis. Grocott- Gomori methenamine-silver staining highlighted fungal organisms with refractile walls and broad-based budding consistent with cutaneous blastomycosis (Figure 1). The histopathologic specimen also demonstrated marked pseudoepitheliomatous hyperplasia (Figure 2A) with neutrophilic microabscesses (Figure 2B). Acid-fast bacillus and Fite staining were negative for bacterial organisms. A fungal culture was positive for Blastomyces dermatitidis. Urine and serum blastomycosis antigen were positive. Although Histoplasma serum antigen also was positive, this likely was from cross-reactivity. Chest radiography was negative for lung involvement, and the patient displayed no neurologic symptoms. He was started on oral itraconazole therapy for the treatment of cutaneous blastomycosis.

Blastomyces dermatitidis, the causative organism of blastomycosis, is endemic to the Ohio and Mississippi River valleys, Great Lakes region, and southeastern United States. It is a thermally dimorphic fungus found in soils that grows as a mold at 25 °C and yeast at 37 °C. Primary infection of the lungs—blastomycosis pneumonia—often is the only clinical manifestation1; however, subsequent hematogenous dissemination to extrapulmonary sites such as the skin, bones, and genitourinary system can occur. Cutaneous blastomycosis, the most common extrapulmonary manifestation, typically follows pulmonary infection. In rare cases, it can occur from direct inoculation.2,3 Skin lesions can occur anywhere but frequently are found on exposed surfaces of the head, neck, and extremities. Lesions classically present as verrucous crusting plaques with draining microabscesses. Violaceous nodules, ulcers, and pustules also may occur.1

Diagnosis involves obtaining a thorough history of possible environmental exposures such as the patient’s geographic area of residence, occupation, and outdoor activities involving soil or decaying wood. Because blastomycosis can remain latent, remote exposures are relevant. Definitive diagnosis of cutaneous blastomycosis involves skin biopsy of the lesion with fungal culture, but the yeast’s distinctive thick wall and broad-based budding seen with periodic acid–Schiff or Grocott-Gomori methenamine-silver staining provides a rapid presumptive diagnosis.3 Pseudoepitheliomatous hyperplasia and microabscesses also are characteristic features.2 Urine antigen testing for a component of the polysaccharide cell wall has a sensitivity of 93% but a lower specificity of 79% due to cross-reactivity with histoplasmosis.4 Treatment consists of itraconazole for mild to moderate blastomycosis or amphotericin B for those with severe disease or central nervous system involvement or those who are immunosuppressed.1
The differential diagnosis for our patient’s lesion included infectious vs neoplastic etiologies. Histoplasma capsulatum, the dimorphic fungus that causes histoplasmosis, also is endemic to the Ohio and Mississippi River valleys. It is found in soil and droppings of some bats and birds such as chickens and pigeons. Similar to blastomycosis, the primary infection site most commonly is the lungs. It subsequently may disseminate to the skin or less commonly via direct inoculation of injured skin. It can present as papules, plaques, ulcers, purpura, or abscesses. Unlike blastomycosis, tissue biopsy of a cutaneous lesion reveals granuloma formation and distinctive oval, narrow-based budding yeast.5 Atypical mycobacteria are another source of infection to consider. For example, cutaneous Mycobacterium kansasii may present as papules and pustules forming verrucous or granulomatous plaques and ulceration. Histopathologic findings distinguishing mycobacterial infection from blastomycosis include granulomas and acid-fast bacilli in histiocytes.6
Noninfectious etiologies in the differential may include cutaneous squamous cell carcinoma or pemphigus vegetans. Squamous cell carcinoma may present with a broad range of clinical features—papules, plaques, or nodules with smooth, scaly, verrucous, or ulcerative secondary features all are possible presentations.7 Fairskinned individuals, such as our patient, would be at a higher risk in sun-damaged skin. Histologically, cutaneous squamous cell carcinoma is defined as an invasion of the dermis by neoplastic squamous epithelial cells in the form of cords, sheets, individual cells, nodules, or cystic structures.7 Pemphigus vegetans is the rarest variant of a group of autoimmune vesiculobullous diseases known as pemphigus. It can be differentiated from the most common variant—pemphigus vulgaris—by the presence of vegetative plaques in intertriginous areas. However, these verrucous vegetations can be misleading and make clinical diagnosis difficult. Histopathologic findings of hyperkeratosis, pseudoepitheliomatous hyperplasia, papillomatosis, and acantholysis with a suprabasal cleft would confirm the diagnosis.8
In summary, cutaneous blastomycosis classically presents as verrucous crusting plaques, as seen in our patient. It is important to conduct a thorough history for environmental exposures, but definitive diagnosis of cutaneous blastomycosis involves skin biopsy with fungal culture. Treatment depends on the severity of disease and organ involvement. Itraconazole would be appropriate for mild to moderate blastomycosis.
The Diagnosis: Cutaneous Blastomycosis
A skin biopsy and fungal cultures confirmed the diagnosis of cutaneous blastomycosis. Grocott- Gomori methenamine-silver staining highlighted fungal organisms with refractile walls and broad-based budding consistent with cutaneous blastomycosis (Figure 1). The histopathologic specimen also demonstrated marked pseudoepitheliomatous hyperplasia (Figure 2A) with neutrophilic microabscesses (Figure 2B). Acid-fast bacillus and Fite staining were negative for bacterial organisms. A fungal culture was positive for Blastomyces dermatitidis. Urine and serum blastomycosis antigen were positive. Although Histoplasma serum antigen also was positive, this likely was from cross-reactivity. Chest radiography was negative for lung involvement, and the patient displayed no neurologic symptoms. He was started on oral itraconazole therapy for the treatment of cutaneous blastomycosis.

Blastomyces dermatitidis, the causative organism of blastomycosis, is endemic to the Ohio and Mississippi River valleys, Great Lakes region, and southeastern United States. It is a thermally dimorphic fungus found in soils that grows as a mold at 25 °C and yeast at 37 °C. Primary infection of the lungs—blastomycosis pneumonia—often is the only clinical manifestation1; however, subsequent hematogenous dissemination to extrapulmonary sites such as the skin, bones, and genitourinary system can occur. Cutaneous blastomycosis, the most common extrapulmonary manifestation, typically follows pulmonary infection. In rare cases, it can occur from direct inoculation.2,3 Skin lesions can occur anywhere but frequently are found on exposed surfaces of the head, neck, and extremities. Lesions classically present as verrucous crusting plaques with draining microabscesses. Violaceous nodules, ulcers, and pustules also may occur.1

Diagnosis involves obtaining a thorough history of possible environmental exposures such as the patient’s geographic area of residence, occupation, and outdoor activities involving soil or decaying wood. Because blastomycosis can remain latent, remote exposures are relevant. Definitive diagnosis of cutaneous blastomycosis involves skin biopsy of the lesion with fungal culture, but the yeast’s distinctive thick wall and broad-based budding seen with periodic acid–Schiff or Grocott-Gomori methenamine-silver staining provides a rapid presumptive diagnosis.3 Pseudoepitheliomatous hyperplasia and microabscesses also are characteristic features.2 Urine antigen testing for a component of the polysaccharide cell wall has a sensitivity of 93% but a lower specificity of 79% due to cross-reactivity with histoplasmosis.4 Treatment consists of itraconazole for mild to moderate blastomycosis or amphotericin B for those with severe disease or central nervous system involvement or those who are immunosuppressed.1
The differential diagnosis for our patient’s lesion included infectious vs neoplastic etiologies. Histoplasma capsulatum, the dimorphic fungus that causes histoplasmosis, also is endemic to the Ohio and Mississippi River valleys. It is found in soil and droppings of some bats and birds such as chickens and pigeons. Similar to blastomycosis, the primary infection site most commonly is the lungs. It subsequently may disseminate to the skin or less commonly via direct inoculation of injured skin. It can present as papules, plaques, ulcers, purpura, or abscesses. Unlike blastomycosis, tissue biopsy of a cutaneous lesion reveals granuloma formation and distinctive oval, narrow-based budding yeast.5 Atypical mycobacteria are another source of infection to consider. For example, cutaneous Mycobacterium kansasii may present as papules and pustules forming verrucous or granulomatous plaques and ulceration. Histopathologic findings distinguishing mycobacterial infection from blastomycosis include granulomas and acid-fast bacilli in histiocytes.6
Noninfectious etiologies in the differential may include cutaneous squamous cell carcinoma or pemphigus vegetans. Squamous cell carcinoma may present with a broad range of clinical features—papules, plaques, or nodules with smooth, scaly, verrucous, or ulcerative secondary features all are possible presentations.7 Fairskinned individuals, such as our patient, would be at a higher risk in sun-damaged skin. Histologically, cutaneous squamous cell carcinoma is defined as an invasion of the dermis by neoplastic squamous epithelial cells in the form of cords, sheets, individual cells, nodules, or cystic structures.7 Pemphigus vegetans is the rarest variant of a group of autoimmune vesiculobullous diseases known as pemphigus. It can be differentiated from the most common variant—pemphigus vulgaris—by the presence of vegetative plaques in intertriginous areas. However, these verrucous vegetations can be misleading and make clinical diagnosis difficult. Histopathologic findings of hyperkeratosis, pseudoepitheliomatous hyperplasia, papillomatosis, and acantholysis with a suprabasal cleft would confirm the diagnosis.8
In summary, cutaneous blastomycosis classically presents as verrucous crusting plaques, as seen in our patient. It is important to conduct a thorough history for environmental exposures, but definitive diagnosis of cutaneous blastomycosis involves skin biopsy with fungal culture. Treatment depends on the severity of disease and organ involvement. Itraconazole would be appropriate for mild to moderate blastomycosis.
- Miceli A, Krishnamurthy K. Blastomycosis. StatPearls. StatPearls Publishing; 2022. Accessed June 21, 2022. https://www.ncbi.nlm.nih.gov/books/NBK441987/
- Gray NA, Baddour LM. Cutaneous inoculation blastomycosis. Clin Infect Dis. 2002;34:E44-E49.
- Schwartz IS, Kauffman CA. Blastomycosis. Semin Respir Crit Care Med. 2020;41:31-41. doi:10.1055/s-0039-3400281
- Castillo CG, Kauffman CA, Miceli MH. Blastomycosis. Infect Dis Clin North Am. 2016;30:247-264. doi:10.1016/j.idc.2015.10.002
- Raggio B. Primary cutaneous histoplasmosis. Ear Nose Throat J. 2018;97:346-348.
- Bhambri S, Bhambri A, Del Rosso JQ. Atypical mycobacterial cutaneous infections. Dermatol Clin. 2009;27:63-73. doi:10.1016/j.det.2008.07.009
- Parekh V, Seykora JT. Cutaneous squamous cell carcinoma. Clin Lab Med. 2017;37:503-525. doi:10.1016/j.cll.2017.06.003
- Messersmith L, Krauland K. Pemphigus vegetans. StatPearls. StatPearls Publishing; 2022. Accessed June 21, 2022. https://www.ncbi.nlm.nih.gov/books/NBK545229/
- Miceli A, Krishnamurthy K. Blastomycosis. StatPearls. StatPearls Publishing; 2022. Accessed June 21, 2022. https://www.ncbi.nlm.nih.gov/books/NBK441987/
- Gray NA, Baddour LM. Cutaneous inoculation blastomycosis. Clin Infect Dis. 2002;34:E44-E49.
- Schwartz IS, Kauffman CA. Blastomycosis. Semin Respir Crit Care Med. 2020;41:31-41. doi:10.1055/s-0039-3400281
- Castillo CG, Kauffman CA, Miceli MH. Blastomycosis. Infect Dis Clin North Am. 2016;30:247-264. doi:10.1016/j.idc.2015.10.002
- Raggio B. Primary cutaneous histoplasmosis. Ear Nose Throat J. 2018;97:346-348.
- Bhambri S, Bhambri A, Del Rosso JQ. Atypical mycobacterial cutaneous infections. Dermatol Clin. 2009;27:63-73. doi:10.1016/j.det.2008.07.009
- Parekh V, Seykora JT. Cutaneous squamous cell carcinoma. Clin Lab Med. 2017;37:503-525. doi:10.1016/j.cll.2017.06.003
- Messersmith L, Krauland K. Pemphigus vegetans. StatPearls. StatPearls Publishing; 2022. Accessed June 21, 2022. https://www.ncbi.nlm.nih.gov/books/NBK545229/
A 39-year-old man from Ohio presented with a tender, 10×6-cm, fungated, eroded plaque on the right medial upper arm that developed over the last 4 years. He initially noticed a firm lump under the right arm 4 years prior that was diagnosed as possible cellulitis at an outside clinic and treated with trimethoprim-sulfamethoxazole. The lesion then began to erode and became a chronic nonhealing wound. Approximately 1 year prior to the current presentation, the patient recalled unloading a truckload of soil around the same time the wound began to enlarge in diameter and depth. He denied any prior or current respiratory or systemic symptoms including fevers, chills, or weight loss.
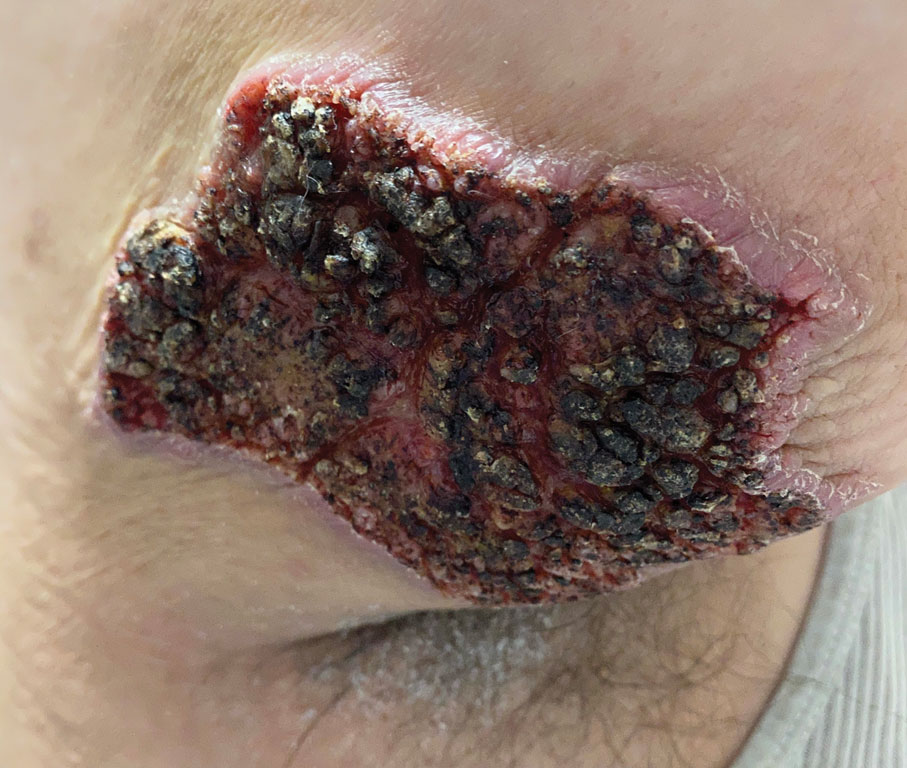
Medical assistants
When I began in private practice several eons ago, I employed only registered nurses (RNs) and licensed practical nurses (LPNs) in my office – as did, I think, most other physicians.
That is still the preferred way to go from an efficiency perspective, as well as the ability to delegate such tasks as blood collection and administering intramuscular injections. Unfortunately,
Given this reality, it makes sense to understand how the use of medical assistants has changed private medical practice, and how the most effective MAs manage their roles and maximize their efficiency in the office.
A recent article by two physicians at the University of Michigan, Ann Arbor, is one of the few published papers to address this issue. It presents the results of a cross-sectional study examining the MA’s experience and key factors that enhance or reduce efficiencies.
The authors sent an email survey to 86 MAs working in six clinics within the department of family medicine at the University of Michigan Medical Center, and received responses from 75 of them, including 61 who completed the entire survey. They then singled out 18 individuals deemed “most efficient” by their peers and conducted face-to-face interviews with them.
The surveys and interviews looked at how MAs identified personal strategies for efficiency, dealt with barriers to implementing those strategies, and navigated interoffice relationships, as well as how all of this affected overall job satisfaction.
All 61 respondents who completed the full survey agreed that the MA role was “very important to keep the clinic functioning” and nearly all said that working in health care was “a calling” for them. About half agreed that their work was very stressful, and about the same percentage reported that there was inadequate MA staffing at their clinic. Others complained of limited pay and promotion opportunities.
The surveyed MAs described important work values that increased their efficiency. These included good communication, strong teamwork, and workload sharing, as well as individual strategies such as multitasking, limiting patient conversations, and completing tasks in a consistent way to improve accuracy.
Other strategies identified as contributing to an efficient operation included preclinic huddles, reviews of patient records before the patient’s arrival, and completing routine office duties before the start of office hours.
Respondents were then asked to identify barriers to clinic efficiency, and most of them involved physicians who barked orders at them, did not complete paperwork or sign orders in a timely manner, and agreed to see late-arriving patients. Some MAs suggested that physicians refrain from “talking down” to them, and teach rather than criticize. They also faulted decisions affecting patient flow made by other staffers without soliciting the MAs’ input.
Despite these barriers, the authors found that most of the surveyed MAs agreed that their work was valued by doctors. “Proper training of managers to provide ... support and ensure equitable workloads may be one strategy to ensure that staff members feel the workplace is fair and collegial,” they said.
“Many described the working relationships with physicians as critical to their satisfaction at work and indicated that strong partnerships motivated them to do their best to make the physician’s day easier,” they added.
At the same time, the authors noted that most survey subjects reported that their jobs were “stressful,” and believed that their stress went underrecognized by physicians. They argued that “it’s important for physicians to be cognizant of these patterns and clinic culture, as reducing a hierarchy-based environment will be appreciated by MAs.”
Since this study involved only MAs in a family practice setting, further studies will be needed to determine whether these results translate to specialty offices – and whether the unique issues inherent in various specialty environments elicit different efficiency contributors and barriers.
Overall, though, “staff job satisfaction is linked to improved quality of care, so treating staff well contributes to high-value care for patients,” the authors wrote. “Disseminating practices that staff members themselves have identified as effective, and being attentive to how staff members are treated, may increase individual efficiency while improving staff retention and satisfaction.”
Dr. Eastern practices dermatology and dermatologic surgery in Belleville, N.J. He is the author of numerous articles and textbook chapters, and is a longtime monthly columnist for Dermatology News. Write to him at [email protected].
When I began in private practice several eons ago, I employed only registered nurses (RNs) and licensed practical nurses (LPNs) in my office – as did, I think, most other physicians.
That is still the preferred way to go from an efficiency perspective, as well as the ability to delegate such tasks as blood collection and administering intramuscular injections. Unfortunately,
Given this reality, it makes sense to understand how the use of medical assistants has changed private medical practice, and how the most effective MAs manage their roles and maximize their efficiency in the office.
A recent article by two physicians at the University of Michigan, Ann Arbor, is one of the few published papers to address this issue. It presents the results of a cross-sectional study examining the MA’s experience and key factors that enhance or reduce efficiencies.
The authors sent an email survey to 86 MAs working in six clinics within the department of family medicine at the University of Michigan Medical Center, and received responses from 75 of them, including 61 who completed the entire survey. They then singled out 18 individuals deemed “most efficient” by their peers and conducted face-to-face interviews with them.
The surveys and interviews looked at how MAs identified personal strategies for efficiency, dealt with barriers to implementing those strategies, and navigated interoffice relationships, as well as how all of this affected overall job satisfaction.
All 61 respondents who completed the full survey agreed that the MA role was “very important to keep the clinic functioning” and nearly all said that working in health care was “a calling” for them. About half agreed that their work was very stressful, and about the same percentage reported that there was inadequate MA staffing at their clinic. Others complained of limited pay and promotion opportunities.
The surveyed MAs described important work values that increased their efficiency. These included good communication, strong teamwork, and workload sharing, as well as individual strategies such as multitasking, limiting patient conversations, and completing tasks in a consistent way to improve accuracy.
Other strategies identified as contributing to an efficient operation included preclinic huddles, reviews of patient records before the patient’s arrival, and completing routine office duties before the start of office hours.
Respondents were then asked to identify barriers to clinic efficiency, and most of them involved physicians who barked orders at them, did not complete paperwork or sign orders in a timely manner, and agreed to see late-arriving patients. Some MAs suggested that physicians refrain from “talking down” to them, and teach rather than criticize. They also faulted decisions affecting patient flow made by other staffers without soliciting the MAs’ input.
Despite these barriers, the authors found that most of the surveyed MAs agreed that their work was valued by doctors. “Proper training of managers to provide ... support and ensure equitable workloads may be one strategy to ensure that staff members feel the workplace is fair and collegial,” they said.
“Many described the working relationships with physicians as critical to their satisfaction at work and indicated that strong partnerships motivated them to do their best to make the physician’s day easier,” they added.
At the same time, the authors noted that most survey subjects reported that their jobs were “stressful,” and believed that their stress went underrecognized by physicians. They argued that “it’s important for physicians to be cognizant of these patterns and clinic culture, as reducing a hierarchy-based environment will be appreciated by MAs.”
Since this study involved only MAs in a family practice setting, further studies will be needed to determine whether these results translate to specialty offices – and whether the unique issues inherent in various specialty environments elicit different efficiency contributors and barriers.
Overall, though, “staff job satisfaction is linked to improved quality of care, so treating staff well contributes to high-value care for patients,” the authors wrote. “Disseminating practices that staff members themselves have identified as effective, and being attentive to how staff members are treated, may increase individual efficiency while improving staff retention and satisfaction.”
Dr. Eastern practices dermatology and dermatologic surgery in Belleville, N.J. He is the author of numerous articles and textbook chapters, and is a longtime monthly columnist for Dermatology News. Write to him at [email protected].
When I began in private practice several eons ago, I employed only registered nurses (RNs) and licensed practical nurses (LPNs) in my office – as did, I think, most other physicians.
That is still the preferred way to go from an efficiency perspective, as well as the ability to delegate such tasks as blood collection and administering intramuscular injections. Unfortunately,
Given this reality, it makes sense to understand how the use of medical assistants has changed private medical practice, and how the most effective MAs manage their roles and maximize their efficiency in the office.
A recent article by two physicians at the University of Michigan, Ann Arbor, is one of the few published papers to address this issue. It presents the results of a cross-sectional study examining the MA’s experience and key factors that enhance or reduce efficiencies.
The authors sent an email survey to 86 MAs working in six clinics within the department of family medicine at the University of Michigan Medical Center, and received responses from 75 of them, including 61 who completed the entire survey. They then singled out 18 individuals deemed “most efficient” by their peers and conducted face-to-face interviews with them.
The surveys and interviews looked at how MAs identified personal strategies for efficiency, dealt with barriers to implementing those strategies, and navigated interoffice relationships, as well as how all of this affected overall job satisfaction.
All 61 respondents who completed the full survey agreed that the MA role was “very important to keep the clinic functioning” and nearly all said that working in health care was “a calling” for them. About half agreed that their work was very stressful, and about the same percentage reported that there was inadequate MA staffing at their clinic. Others complained of limited pay and promotion opportunities.
The surveyed MAs described important work values that increased their efficiency. These included good communication, strong teamwork, and workload sharing, as well as individual strategies such as multitasking, limiting patient conversations, and completing tasks in a consistent way to improve accuracy.
Other strategies identified as contributing to an efficient operation included preclinic huddles, reviews of patient records before the patient’s arrival, and completing routine office duties before the start of office hours.
Respondents were then asked to identify barriers to clinic efficiency, and most of them involved physicians who barked orders at them, did not complete paperwork or sign orders in a timely manner, and agreed to see late-arriving patients. Some MAs suggested that physicians refrain from “talking down” to them, and teach rather than criticize. They also faulted decisions affecting patient flow made by other staffers without soliciting the MAs’ input.
Despite these barriers, the authors found that most of the surveyed MAs agreed that their work was valued by doctors. “Proper training of managers to provide ... support and ensure equitable workloads may be one strategy to ensure that staff members feel the workplace is fair and collegial,” they said.
“Many described the working relationships with physicians as critical to their satisfaction at work and indicated that strong partnerships motivated them to do their best to make the physician’s day easier,” they added.
At the same time, the authors noted that most survey subjects reported that their jobs were “stressful,” and believed that their stress went underrecognized by physicians. They argued that “it’s important for physicians to be cognizant of these patterns and clinic culture, as reducing a hierarchy-based environment will be appreciated by MAs.”
Since this study involved only MAs in a family practice setting, further studies will be needed to determine whether these results translate to specialty offices – and whether the unique issues inherent in various specialty environments elicit different efficiency contributors and barriers.
Overall, though, “staff job satisfaction is linked to improved quality of care, so treating staff well contributes to high-value care for patients,” the authors wrote. “Disseminating practices that staff members themselves have identified as effective, and being attentive to how staff members are treated, may increase individual efficiency while improving staff retention and satisfaction.”
Dr. Eastern practices dermatology and dermatologic surgery in Belleville, N.J. He is the author of numerous articles and textbook chapters, and is a longtime monthly columnist for Dermatology News. Write to him at [email protected].
Best meds for insomnia identified?
In a comprehensive comparative-effectiveness analysis, lemborexant and eszopiclone showed the best efficacy, acceptability, and tolerability for acute and long-term insomnia treatment.
However, eszopiclone may cause substantial side effects – and safety data on lemborexant were inconclusive, the researchers note.
Not surprisingly, short-acting, intermediate-acting, and long-acting benzodiazepines were effective in the acute treatment of insomnia, but they have unfavorable tolerability and safety profiles, and there are no long-term data on these issues.
For many insomnia medications, there is a “striking” and “appalling” lack of long-term data, study investigator Andrea Cipriani, MD, PhD, professor of psychiatry, University of Oxford, United Kingdom, noted during a press briefing.
“This is a call for regulators to raise the bar and ask for long-term data when companies submit an application for licensing insomnia drugs,” Dr. Cipriani said.
The findings were published online in The Lancet.
Prevalent, debilitating
Insomnia is highly prevalent, affecting up to 1 in 5 adults, and can have a profound impact on health, well-being, and productivity.
Sleep hygiene and cognitive-behavioral therapy for insomnia (CBT-I) are recommended first-line treatments, but they are often unavailable, which often leads patients and clinicians to turn to medications.
However, “insomnia drugs are not all created equal. Even within the same drug class there are differences,” Dr. Cipriani said.
In a large-scale systematic review and network meta-analysis, the researchers analyzed data from 154 double-blind, randomized controlled trials of medications (licensed or not) used for acute and long-term treatment of insomnia in 44,089 adults (mean age, 51.7 years; 63% women).
Results showed, for the acute treatment of insomnia, benzodiazepines, doxylamine, eszopiclone, lemborexant, seltorexant, zolpidem, and zopiclone were more effective than placebo (standardized mean difference range, 0.36-0.83; high-to-moderate certainty of evidence).
In addition, benzodiazepines, eszopiclone, zolpidem, and zopiclone were more effective than melatonin, ramelteon, and zaleplon (SMD, 0.27-0.71; moderate-to-very low certainty of evidence).
“Our results show that the melatonergic drugs melatonin and ramelteon are not really effective. The data do not support the regular use of these drugs,” co-investigator Phil Cowen, PhD, professor of psychopharmacology, University of Oxford, said at the briefing.
Best available evidence
What little long-term data is available suggest eszopiclone and lemborexant are more effective than placebo. Plus, eszopiclone is more effective than ramelteon and zolpidem but with “very low” certainty of evidence, the researchers report.
“There was insufficient evidence to support the prescription of benzodiazepines and zolpidem in long-term treatment,” they write.
Another problem was lack of data on other important outcomes, they add.
“We wanted to look at hangover effects, daytime sleepiness, [and] rebound effect, but often there was no data reported in trials. We need to collect data about these outcomes because they matter to clinicians and patients,” Dr. Cipriani said.
Summing up, the researchers note the current findings represent the “best available evidence base to guide the choice about pharmacological treatment for insomnia disorder in adults and will assist in shared decisionmaking between patients, carers, and their clinicians, as well as policy makers.”
They caution, however, that all statements comparing the merits of one drug with another “should be tempered by the potential limitations of the current analysis, the quality of the available evidence, the characteristics of the patient populations, and the uncertainties that might result from choice of dose or treatment setting.”
In addition, it is important to also consider nonpharmacologic treatments for insomnia disorder, as they are supported by “high-quality evidence and recommended as first-line treatment by guidelines,” the investigator write.
Shared decisionmaking
In an accompanying editorial, Myrto Samara, MD, University of Thessaly, Larissa, Greece, agrees with the researchers that discussion with patients is key.
“For insomnia treatment, patient-physician shared decisionmaking is crucial to decide when a pharmacological intervention is deemed necessary and which drug [is] to be given by considering the trade-offs for efficacy and side effects,” Dr. Samara writes.
The study was funded by the UK National Institute for Health Research (NIHR) Oxford Health Biomedical Research Center. Dr. Cipriani has received research and consultancy fees from the Italian Network for Pediatric Trials, CARIPLO Foundation, and Angelini Pharma, and is the chief and principal investigator of two trials of seltorexant in depression that are sponsored by Janssen. Dr. Samara has reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
In a comprehensive comparative-effectiveness analysis, lemborexant and eszopiclone showed the best efficacy, acceptability, and tolerability for acute and long-term insomnia treatment.
However, eszopiclone may cause substantial side effects – and safety data on lemborexant were inconclusive, the researchers note.
Not surprisingly, short-acting, intermediate-acting, and long-acting benzodiazepines were effective in the acute treatment of insomnia, but they have unfavorable tolerability and safety profiles, and there are no long-term data on these issues.
For many insomnia medications, there is a “striking” and “appalling” lack of long-term data, study investigator Andrea Cipriani, MD, PhD, professor of psychiatry, University of Oxford, United Kingdom, noted during a press briefing.
“This is a call for regulators to raise the bar and ask for long-term data when companies submit an application for licensing insomnia drugs,” Dr. Cipriani said.
The findings were published online in The Lancet.
Prevalent, debilitating
Insomnia is highly prevalent, affecting up to 1 in 5 adults, and can have a profound impact on health, well-being, and productivity.
Sleep hygiene and cognitive-behavioral therapy for insomnia (CBT-I) are recommended first-line treatments, but they are often unavailable, which often leads patients and clinicians to turn to medications.
However, “insomnia drugs are not all created equal. Even within the same drug class there are differences,” Dr. Cipriani said.
In a large-scale systematic review and network meta-analysis, the researchers analyzed data from 154 double-blind, randomized controlled trials of medications (licensed or not) used for acute and long-term treatment of insomnia in 44,089 adults (mean age, 51.7 years; 63% women).
Results showed, for the acute treatment of insomnia, benzodiazepines, doxylamine, eszopiclone, lemborexant, seltorexant, zolpidem, and zopiclone were more effective than placebo (standardized mean difference range, 0.36-0.83; high-to-moderate certainty of evidence).
In addition, benzodiazepines, eszopiclone, zolpidem, and zopiclone were more effective than melatonin, ramelteon, and zaleplon (SMD, 0.27-0.71; moderate-to-very low certainty of evidence).
“Our results show that the melatonergic drugs melatonin and ramelteon are not really effective. The data do not support the regular use of these drugs,” co-investigator Phil Cowen, PhD, professor of psychopharmacology, University of Oxford, said at the briefing.
Best available evidence
What little long-term data is available suggest eszopiclone and lemborexant are more effective than placebo. Plus, eszopiclone is more effective than ramelteon and zolpidem but with “very low” certainty of evidence, the researchers report.
“There was insufficient evidence to support the prescription of benzodiazepines and zolpidem in long-term treatment,” they write.
Another problem was lack of data on other important outcomes, they add.
“We wanted to look at hangover effects, daytime sleepiness, [and] rebound effect, but often there was no data reported in trials. We need to collect data about these outcomes because they matter to clinicians and patients,” Dr. Cipriani said.
Summing up, the researchers note the current findings represent the “best available evidence base to guide the choice about pharmacological treatment for insomnia disorder in adults and will assist in shared decisionmaking between patients, carers, and their clinicians, as well as policy makers.”
They caution, however, that all statements comparing the merits of one drug with another “should be tempered by the potential limitations of the current analysis, the quality of the available evidence, the characteristics of the patient populations, and the uncertainties that might result from choice of dose or treatment setting.”
In addition, it is important to also consider nonpharmacologic treatments for insomnia disorder, as they are supported by “high-quality evidence and recommended as first-line treatment by guidelines,” the investigator write.
Shared decisionmaking
In an accompanying editorial, Myrto Samara, MD, University of Thessaly, Larissa, Greece, agrees with the researchers that discussion with patients is key.
“For insomnia treatment, patient-physician shared decisionmaking is crucial to decide when a pharmacological intervention is deemed necessary and which drug [is] to be given by considering the trade-offs for efficacy and side effects,” Dr. Samara writes.
The study was funded by the UK National Institute for Health Research (NIHR) Oxford Health Biomedical Research Center. Dr. Cipriani has received research and consultancy fees from the Italian Network for Pediatric Trials, CARIPLO Foundation, and Angelini Pharma, and is the chief and principal investigator of two trials of seltorexant in depression that are sponsored by Janssen. Dr. Samara has reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
In a comprehensive comparative-effectiveness analysis, lemborexant and eszopiclone showed the best efficacy, acceptability, and tolerability for acute and long-term insomnia treatment.
However, eszopiclone may cause substantial side effects – and safety data on lemborexant were inconclusive, the researchers note.
Not surprisingly, short-acting, intermediate-acting, and long-acting benzodiazepines were effective in the acute treatment of insomnia, but they have unfavorable tolerability and safety profiles, and there are no long-term data on these issues.
For many insomnia medications, there is a “striking” and “appalling” lack of long-term data, study investigator Andrea Cipriani, MD, PhD, professor of psychiatry, University of Oxford, United Kingdom, noted during a press briefing.
“This is a call for regulators to raise the bar and ask for long-term data when companies submit an application for licensing insomnia drugs,” Dr. Cipriani said.
The findings were published online in The Lancet.
Prevalent, debilitating
Insomnia is highly prevalent, affecting up to 1 in 5 adults, and can have a profound impact on health, well-being, and productivity.
Sleep hygiene and cognitive-behavioral therapy for insomnia (CBT-I) are recommended first-line treatments, but they are often unavailable, which often leads patients and clinicians to turn to medications.
However, “insomnia drugs are not all created equal. Even within the same drug class there are differences,” Dr. Cipriani said.
In a large-scale systematic review and network meta-analysis, the researchers analyzed data from 154 double-blind, randomized controlled trials of medications (licensed or not) used for acute and long-term treatment of insomnia in 44,089 adults (mean age, 51.7 years; 63% women).
Results showed, for the acute treatment of insomnia, benzodiazepines, doxylamine, eszopiclone, lemborexant, seltorexant, zolpidem, and zopiclone were more effective than placebo (standardized mean difference range, 0.36-0.83; high-to-moderate certainty of evidence).
In addition, benzodiazepines, eszopiclone, zolpidem, and zopiclone were more effective than melatonin, ramelteon, and zaleplon (SMD, 0.27-0.71; moderate-to-very low certainty of evidence).
“Our results show that the melatonergic drugs melatonin and ramelteon are not really effective. The data do not support the regular use of these drugs,” co-investigator Phil Cowen, PhD, professor of psychopharmacology, University of Oxford, said at the briefing.
Best available evidence
What little long-term data is available suggest eszopiclone and lemborexant are more effective than placebo. Plus, eszopiclone is more effective than ramelteon and zolpidem but with “very low” certainty of evidence, the researchers report.
“There was insufficient evidence to support the prescription of benzodiazepines and zolpidem in long-term treatment,” they write.
Another problem was lack of data on other important outcomes, they add.
“We wanted to look at hangover effects, daytime sleepiness, [and] rebound effect, but often there was no data reported in trials. We need to collect data about these outcomes because they matter to clinicians and patients,” Dr. Cipriani said.
Summing up, the researchers note the current findings represent the “best available evidence base to guide the choice about pharmacological treatment for insomnia disorder in adults and will assist in shared decisionmaking between patients, carers, and their clinicians, as well as policy makers.”
They caution, however, that all statements comparing the merits of one drug with another “should be tempered by the potential limitations of the current analysis, the quality of the available evidence, the characteristics of the patient populations, and the uncertainties that might result from choice of dose or treatment setting.”
In addition, it is important to also consider nonpharmacologic treatments for insomnia disorder, as they are supported by “high-quality evidence and recommended as first-line treatment by guidelines,” the investigator write.
Shared decisionmaking
In an accompanying editorial, Myrto Samara, MD, University of Thessaly, Larissa, Greece, agrees with the researchers that discussion with patients is key.
“For insomnia treatment, patient-physician shared decisionmaking is crucial to decide when a pharmacological intervention is deemed necessary and which drug [is] to be given by considering the trade-offs for efficacy and side effects,” Dr. Samara writes.
The study was funded by the UK National Institute for Health Research (NIHR) Oxford Health Biomedical Research Center. Dr. Cipriani has received research and consultancy fees from the Italian Network for Pediatric Trials, CARIPLO Foundation, and Angelini Pharma, and is the chief and principal investigator of two trials of seltorexant in depression that are sponsored by Janssen. Dr. Samara has reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FROM THE LANCET
Neural networks can distinguish PsA from rheumatoid arthritis on MRI
Hand images are sufficient
NEW YORK – On the basis of MRI images of the hand, a neural network has been trained to distinguish seronegative and seropositive rheumatoid arthritis (RA) from psoriatic arthritis (PsA) as well as from each other, according to a study that was presented at the annual meeting of the Group for Research and Assessment of Psoriasis and Psoriatic Arthritis.
In the work so far, the neural network was correct about 70% of the time in the absence of any further clinical analyses, according to David Simon, MD, a rheumatologist in the department of internal medicine at Friedrich-Alexander University, Erlangen, Germany.
Previous to this work, “there has been no study that has exclusively used hand MRI data and deep learning without requiring further expert input for the classification of arthritides,” Dr. Simon said.
In fact, when demographic and clinical data were added, there was no improvement in the performance of patient classification relative to the deep learning classification alone, according to the data presented by Dr. Simon.
The images were evaluated with residual neural networks (ResNet), which represents a sophisticated form of deep learning to facilitate the flow of information across the network layers as they form to improve accuracy in their ability to distinguish one form of disease from the other. The training was performed on images from the T1 coronal, T2 corona1, T1 coronal fat suppressed with contrast, T1 axial fat suppressed with contrast, and T2 fat suppressed axial sequences.
The study included hand MRI scans from 135 patients with seronegative RA, 190 with seropositive RA, 177 with PsA, and 147 with psoriasis. The performance was judged on the basis of area under the receiver operating characteristics curve (AUROC) with and without input of clinical characteristics. Patients who had psoriasis without clinical arthritis were included as a control population.
The AUROC for accuracy was 75% for seropositive RA relative to PsA, 74% for seronegative RA relative to PsA, and 67% for seropositive relative to seronegative RA. Of the patients who had psoriasis without arthritis, 98% were classified as PsA and 2% as RA.
Subsequent to the classification of the patients with psoriasis, 14 of the 147 (9.5%) have developed PsA so far over a relatively short follow-up. All of these were among those identified as PsA by neural network evaluation of the hand MRIs.
This suggests that “a PsA-like pattern may be present early in the course of psoriatic disease,” Dr. Simon said.
In the groups with joint disease, who had mean ages ranging from 56 to 65, the mean disease durations were 2.6 years for those with seropositive RA, 1.3 years for those with seronegative RA, and 0.8 years for those with PsA. The patients with psoriasis were younger (mean age, 40.5 years) but had a longer disease duration (mean 4.2 years).
All of the MRI sequences were relevant for classification, but contrast did not appear to help with accuracy.
“If the images with contrast enhancement were deleted, the loss of performance was only marginal,” Dr. Simon reported.
The accuracy of neural networks increases with data, making it likely that further refinements in methodology will lead to a greater degree of accuracy, according to Dr. Simon. While the methodology is not yet ready for routine use in the clinic, the study demonstrates that neural network analysis of hand MRI to distinguish forms of arthritis “is possible.” Further studies are planned toward the goal of creating a viable clinical tool.
“Of course, if we could create an accurate tool with ultrasound, this would be even more practical,” said Dr. Simon, recognizing the value of an office tool, but he cautioned that this would be far more challenging.
“The precision of MRI is an important factor for effective neural network training,” he said.
Utility: ‘In challenging cases if the accuracy improves’?
A viable method for objectively and rapidly distinguishing inflammatory joint diseases, particularly in patients with an ambiguous clinical presentation, is an unmet need, according to Philip J. Mease, MD, director of rheumatology research at Swedish Medical Center, Seattle.
Although the data presented are promising, Dr. Mease said in an interview that he believes there is a fair amount of work to be done before imaging analysis based on deep learning makes its way into routine clinical care. He is also hoping for methods to distinguish RA from PsA that are easier and less expensive, such as serum biomarkers. However, he agreed that a MRI-based tool could be useful when differentiating disease that is challenging.
“MRI is an expensive way for routine classification of disease, but this approach could be useful in challenging cases if the accuracy improves,” he said.
Meanwhile, other clinical researchers might want to test the principle. “You can try it,” said Dr. Simon, who reported that his team has made the methodology publicly available.
Dr. Simon reported no conflicts of interest. Dr. Mease reported financial relationships with more than 10 pharmaceutical companies, most of which make products used for the treatment of inflammatory joint diseases.
Hand images are sufficient
Hand images are sufficient
NEW YORK – On the basis of MRI images of the hand, a neural network has been trained to distinguish seronegative and seropositive rheumatoid arthritis (RA) from psoriatic arthritis (PsA) as well as from each other, according to a study that was presented at the annual meeting of the Group for Research and Assessment of Psoriasis and Psoriatic Arthritis.
In the work so far, the neural network was correct about 70% of the time in the absence of any further clinical analyses, according to David Simon, MD, a rheumatologist in the department of internal medicine at Friedrich-Alexander University, Erlangen, Germany.
Previous to this work, “there has been no study that has exclusively used hand MRI data and deep learning without requiring further expert input for the classification of arthritides,” Dr. Simon said.
In fact, when demographic and clinical data were added, there was no improvement in the performance of patient classification relative to the deep learning classification alone, according to the data presented by Dr. Simon.
The images were evaluated with residual neural networks (ResNet), which represents a sophisticated form of deep learning to facilitate the flow of information across the network layers as they form to improve accuracy in their ability to distinguish one form of disease from the other. The training was performed on images from the T1 coronal, T2 corona1, T1 coronal fat suppressed with contrast, T1 axial fat suppressed with contrast, and T2 fat suppressed axial sequences.
The study included hand MRI scans from 135 patients with seronegative RA, 190 with seropositive RA, 177 with PsA, and 147 with psoriasis. The performance was judged on the basis of area under the receiver operating characteristics curve (AUROC) with and without input of clinical characteristics. Patients who had psoriasis without clinical arthritis were included as a control population.
The AUROC for accuracy was 75% for seropositive RA relative to PsA, 74% for seronegative RA relative to PsA, and 67% for seropositive relative to seronegative RA. Of the patients who had psoriasis without arthritis, 98% were classified as PsA and 2% as RA.
Subsequent to the classification of the patients with psoriasis, 14 of the 147 (9.5%) have developed PsA so far over a relatively short follow-up. All of these were among those identified as PsA by neural network evaluation of the hand MRIs.
This suggests that “a PsA-like pattern may be present early in the course of psoriatic disease,” Dr. Simon said.
In the groups with joint disease, who had mean ages ranging from 56 to 65, the mean disease durations were 2.6 years for those with seropositive RA, 1.3 years for those with seronegative RA, and 0.8 years for those with PsA. The patients with psoriasis were younger (mean age, 40.5 years) but had a longer disease duration (mean 4.2 years).
All of the MRI sequences were relevant for classification, but contrast did not appear to help with accuracy.
“If the images with contrast enhancement were deleted, the loss of performance was only marginal,” Dr. Simon reported.
The accuracy of neural networks increases with data, making it likely that further refinements in methodology will lead to a greater degree of accuracy, according to Dr. Simon. While the methodology is not yet ready for routine use in the clinic, the study demonstrates that neural network analysis of hand MRI to distinguish forms of arthritis “is possible.” Further studies are planned toward the goal of creating a viable clinical tool.
“Of course, if we could create an accurate tool with ultrasound, this would be even more practical,” said Dr. Simon, recognizing the value of an office tool, but he cautioned that this would be far more challenging.
“The precision of MRI is an important factor for effective neural network training,” he said.
Utility: ‘In challenging cases if the accuracy improves’?
A viable method for objectively and rapidly distinguishing inflammatory joint diseases, particularly in patients with an ambiguous clinical presentation, is an unmet need, according to Philip J. Mease, MD, director of rheumatology research at Swedish Medical Center, Seattle.
Although the data presented are promising, Dr. Mease said in an interview that he believes there is a fair amount of work to be done before imaging analysis based on deep learning makes its way into routine clinical care. He is also hoping for methods to distinguish RA from PsA that are easier and less expensive, such as serum biomarkers. However, he agreed that a MRI-based tool could be useful when differentiating disease that is challenging.
“MRI is an expensive way for routine classification of disease, but this approach could be useful in challenging cases if the accuracy improves,” he said.
Meanwhile, other clinical researchers might want to test the principle. “You can try it,” said Dr. Simon, who reported that his team has made the methodology publicly available.
Dr. Simon reported no conflicts of interest. Dr. Mease reported financial relationships with more than 10 pharmaceutical companies, most of which make products used for the treatment of inflammatory joint diseases.
NEW YORK – On the basis of MRI images of the hand, a neural network has been trained to distinguish seronegative and seropositive rheumatoid arthritis (RA) from psoriatic arthritis (PsA) as well as from each other, according to a study that was presented at the annual meeting of the Group for Research and Assessment of Psoriasis and Psoriatic Arthritis.
In the work so far, the neural network was correct about 70% of the time in the absence of any further clinical analyses, according to David Simon, MD, a rheumatologist in the department of internal medicine at Friedrich-Alexander University, Erlangen, Germany.
Previous to this work, “there has been no study that has exclusively used hand MRI data and deep learning without requiring further expert input for the classification of arthritides,” Dr. Simon said.
In fact, when demographic and clinical data were added, there was no improvement in the performance of patient classification relative to the deep learning classification alone, according to the data presented by Dr. Simon.
The images were evaluated with residual neural networks (ResNet), which represents a sophisticated form of deep learning to facilitate the flow of information across the network layers as they form to improve accuracy in their ability to distinguish one form of disease from the other. The training was performed on images from the T1 coronal, T2 corona1, T1 coronal fat suppressed with contrast, T1 axial fat suppressed with contrast, and T2 fat suppressed axial sequences.
The study included hand MRI scans from 135 patients with seronegative RA, 190 with seropositive RA, 177 with PsA, and 147 with psoriasis. The performance was judged on the basis of area under the receiver operating characteristics curve (AUROC) with and without input of clinical characteristics. Patients who had psoriasis without clinical arthritis were included as a control population.
The AUROC for accuracy was 75% for seropositive RA relative to PsA, 74% for seronegative RA relative to PsA, and 67% for seropositive relative to seronegative RA. Of the patients who had psoriasis without arthritis, 98% were classified as PsA and 2% as RA.
Subsequent to the classification of the patients with psoriasis, 14 of the 147 (9.5%) have developed PsA so far over a relatively short follow-up. All of these were among those identified as PsA by neural network evaluation of the hand MRIs.
This suggests that “a PsA-like pattern may be present early in the course of psoriatic disease,” Dr. Simon said.
In the groups with joint disease, who had mean ages ranging from 56 to 65, the mean disease durations were 2.6 years for those with seropositive RA, 1.3 years for those with seronegative RA, and 0.8 years for those with PsA. The patients with psoriasis were younger (mean age, 40.5 years) but had a longer disease duration (mean 4.2 years).
All of the MRI sequences were relevant for classification, but contrast did not appear to help with accuracy.
“If the images with contrast enhancement were deleted, the loss of performance was only marginal,” Dr. Simon reported.
The accuracy of neural networks increases with data, making it likely that further refinements in methodology will lead to a greater degree of accuracy, according to Dr. Simon. While the methodology is not yet ready for routine use in the clinic, the study demonstrates that neural network analysis of hand MRI to distinguish forms of arthritis “is possible.” Further studies are planned toward the goal of creating a viable clinical tool.
“Of course, if we could create an accurate tool with ultrasound, this would be even more practical,” said Dr. Simon, recognizing the value of an office tool, but he cautioned that this would be far more challenging.
“The precision of MRI is an important factor for effective neural network training,” he said.
Utility: ‘In challenging cases if the accuracy improves’?
A viable method for objectively and rapidly distinguishing inflammatory joint diseases, particularly in patients with an ambiguous clinical presentation, is an unmet need, according to Philip J. Mease, MD, director of rheumatology research at Swedish Medical Center, Seattle.
Although the data presented are promising, Dr. Mease said in an interview that he believes there is a fair amount of work to be done before imaging analysis based on deep learning makes its way into routine clinical care. He is also hoping for methods to distinguish RA from PsA that are easier and less expensive, such as serum biomarkers. However, he agreed that a MRI-based tool could be useful when differentiating disease that is challenging.
“MRI is an expensive way for routine classification of disease, but this approach could be useful in challenging cases if the accuracy improves,” he said.
Meanwhile, other clinical researchers might want to test the principle. “You can try it,” said Dr. Simon, who reported that his team has made the methodology publicly available.
Dr. Simon reported no conflicts of interest. Dr. Mease reported financial relationships with more than 10 pharmaceutical companies, most of which make products used for the treatment of inflammatory joint diseases.
AT GRAPPA 2022