User login
Formerly Skin & Allergy News
ass lick
assault rifle
balls
ballsac
black jack
bleach
Boko Haram
bondage
causas
cheap
child abuse
cocaine
compulsive behaviors
cost of miracles
cunt
Daech
display network stats
drug paraphernalia
explosion
fart
fda and death
fda AND warn
fda AND warning
fda AND warns
feom
fuck
gambling
gfc
gun
human trafficking
humira AND expensive
illegal
ISIL
ISIS
Islamic caliphate
Islamic state
madvocate
masturbation
mixed martial arts
MMA
molestation
national rifle association
NRA
nsfw
nuccitelli
pedophile
pedophilia
poker
porn
porn
pornography
psychedelic drug
recreational drug
sex slave rings
shit
slot machine
snort
substance abuse
terrorism
terrorist
texarkana
Texas hold 'em
UFC
section[contains(@class, 'nav-hidden')]
section[contains(@class, 'nav-hidden active')]
The leading independent newspaper covering dermatology news and commentary.
First Results From Laser-Related Adverse Events Registry Reported
BALTIMORE — A relatively . But the process of reporting AEs to the registry needs to be made easier to attract more cases and provide a more complete picture of complications after dermatologic procedures, a researcher and observer said.
The Cutaneous Procedures Adverse Events Reporting Registry (CAPER) was established in 2021 to track AEs from dermatologic procedures. Since then, it has logged a total of 81 cases and 147 AEs from 27 unique procedures, Eric Koza, MD, a postdoctoral research fellow in the Department of Dermatology at Northwestern University, Chicago, reported at the annual conference of the American Society for Laser Medicine and Surgery.
“The takeaways from this project is that 20 laser and energy device treatments have been reported to the registry, half of which were nonablative laser treatments,” Dr. Koza said in presenting the results. “Of the adverse events reported, nonphysicians and non-dermatologic physicians were more likely to be associated with severe or persistent adverse events.”
The American Society for Dermatologic Surgery Association and the Northwestern University Department of Dermatology launched CAPER. Previously, Dr. Koza said, AEs were typically reported only through the Food and Drug Administration’s AE reporting system. He noted that CAPER is the only voluntary national reporting registry for AEs from dermatologic procedures.
What the Registry Shows So Far
The registry matched 72 of the 81 cases with type of provider, with dermatologist-conducted procedures (51, 70.8%) comprising the majority, followed by nonphysician-conducted procedures (14, 19.4%) and nondermatologist physician–conducted procedures (7, 9.7%).
Of the 81 total cases, the following reports were related to laser and energy device treatments: 12 (14.3%) from nonablative laser treatments, five (6%) from light treatments, and three (3.6%) from ablative laser treatments, Dr. Koza said.
Among nonablative laser treatments, the most common AE was blistering (six reports, 50%). Scar, pain, and hypopigmentation accounted for two cases each (16.67%). Dermatologists performed seven of these cases (58.3%); nonphysicians, four (33.3%); and a non-dermatologist physician, one (8.3%).
For intense pulsed-light treatments, burns were the most common AEs (three reports, 60%), with swelling and inflammation each accounting for one case (20%). Three of these cases (75%) were confirmed to have been performed by nonphysicians.
The ablative laser treatment AEs included one case each of hypopigmentation, scar, and erythema. Two of the three cases were confirmed to have been performed by dermatologists.
Dr. Koza acknowledged the low number of cases is a limitation of this analysis of registry reports. A future goal for CAPER is to publicize it more, he said. “The registry is only 3 years old,” he told this news organization. “Hopefully, we can get more data as time goes on. We’ve been getting more and more each year.” CAPER adapted data entry forms used in other registries.
Submitting a case to the registry takes about 15 minutes of the provider’s time, Dr. Koza said. “We can streamline that to make it easier for people to submit their adverse events,” he said in an interview.
Only registry staff have access to the reports, and when reported, the data “is de-identified and any identifying information pertaining to the patient or reporter is removed,” according to a statement on the CAPER website.
‘Needs a Little Help’
Jennifer Lin, MD, a dermatologist at Brigham and Women’s Hospital and Dana-Farber Cancer Institute in Boston, who was at the meeting, commented on the onerous reporting process and the “low” enrollment. “It’s such an important initiative and with everyone over-logging e-mails, a 15-minute entry just is not going to cut it,” she told this news organization.
For providers, reporting AEs is stressful, she said. “As it is, it’s hard to voluntarily submit an adverse event,” Dr. Lin continued. “There’s a feeling of shame. Hospitals require it in order to monitor adverse events, but there’s no monitoring when you’re out in your own private practice.”
“The idea is excellent, but I think to facilitate better enrollment, the word has to get out at all these meetings” and make it easier to submit cases, Dr. Lin added. “It’s a good idea, but it needs a little help.”
Information on submitting AE reports to CAPER is available on the CAPER website.
Dr. Koza and Dr. Lin had no relevant relationships to disclose.
A version of this article first appeared on Medscape.com.
BALTIMORE — A relatively . But the process of reporting AEs to the registry needs to be made easier to attract more cases and provide a more complete picture of complications after dermatologic procedures, a researcher and observer said.
The Cutaneous Procedures Adverse Events Reporting Registry (CAPER) was established in 2021 to track AEs from dermatologic procedures. Since then, it has logged a total of 81 cases and 147 AEs from 27 unique procedures, Eric Koza, MD, a postdoctoral research fellow in the Department of Dermatology at Northwestern University, Chicago, reported at the annual conference of the American Society for Laser Medicine and Surgery.
“The takeaways from this project is that 20 laser and energy device treatments have been reported to the registry, half of which were nonablative laser treatments,” Dr. Koza said in presenting the results. “Of the adverse events reported, nonphysicians and non-dermatologic physicians were more likely to be associated with severe or persistent adverse events.”
The American Society for Dermatologic Surgery Association and the Northwestern University Department of Dermatology launched CAPER. Previously, Dr. Koza said, AEs were typically reported only through the Food and Drug Administration’s AE reporting system. He noted that CAPER is the only voluntary national reporting registry for AEs from dermatologic procedures.
What the Registry Shows So Far
The registry matched 72 of the 81 cases with type of provider, with dermatologist-conducted procedures (51, 70.8%) comprising the majority, followed by nonphysician-conducted procedures (14, 19.4%) and nondermatologist physician–conducted procedures (7, 9.7%).
Of the 81 total cases, the following reports were related to laser and energy device treatments: 12 (14.3%) from nonablative laser treatments, five (6%) from light treatments, and three (3.6%) from ablative laser treatments, Dr. Koza said.
Among nonablative laser treatments, the most common AE was blistering (six reports, 50%). Scar, pain, and hypopigmentation accounted for two cases each (16.67%). Dermatologists performed seven of these cases (58.3%); nonphysicians, four (33.3%); and a non-dermatologist physician, one (8.3%).
For intense pulsed-light treatments, burns were the most common AEs (three reports, 60%), with swelling and inflammation each accounting for one case (20%). Three of these cases (75%) were confirmed to have been performed by nonphysicians.
The ablative laser treatment AEs included one case each of hypopigmentation, scar, and erythema. Two of the three cases were confirmed to have been performed by dermatologists.
Dr. Koza acknowledged the low number of cases is a limitation of this analysis of registry reports. A future goal for CAPER is to publicize it more, he said. “The registry is only 3 years old,” he told this news organization. “Hopefully, we can get more data as time goes on. We’ve been getting more and more each year.” CAPER adapted data entry forms used in other registries.
Submitting a case to the registry takes about 15 minutes of the provider’s time, Dr. Koza said. “We can streamline that to make it easier for people to submit their adverse events,” he said in an interview.
Only registry staff have access to the reports, and when reported, the data “is de-identified and any identifying information pertaining to the patient or reporter is removed,” according to a statement on the CAPER website.
‘Needs a Little Help’
Jennifer Lin, MD, a dermatologist at Brigham and Women’s Hospital and Dana-Farber Cancer Institute in Boston, who was at the meeting, commented on the onerous reporting process and the “low” enrollment. “It’s such an important initiative and with everyone over-logging e-mails, a 15-minute entry just is not going to cut it,” she told this news organization.
For providers, reporting AEs is stressful, she said. “As it is, it’s hard to voluntarily submit an adverse event,” Dr. Lin continued. “There’s a feeling of shame. Hospitals require it in order to monitor adverse events, but there’s no monitoring when you’re out in your own private practice.”
“The idea is excellent, but I think to facilitate better enrollment, the word has to get out at all these meetings” and make it easier to submit cases, Dr. Lin added. “It’s a good idea, but it needs a little help.”
Information on submitting AE reports to CAPER is available on the CAPER website.
Dr. Koza and Dr. Lin had no relevant relationships to disclose.
A version of this article first appeared on Medscape.com.
BALTIMORE — A relatively . But the process of reporting AEs to the registry needs to be made easier to attract more cases and provide a more complete picture of complications after dermatologic procedures, a researcher and observer said.
The Cutaneous Procedures Adverse Events Reporting Registry (CAPER) was established in 2021 to track AEs from dermatologic procedures. Since then, it has logged a total of 81 cases and 147 AEs from 27 unique procedures, Eric Koza, MD, a postdoctoral research fellow in the Department of Dermatology at Northwestern University, Chicago, reported at the annual conference of the American Society for Laser Medicine and Surgery.
“The takeaways from this project is that 20 laser and energy device treatments have been reported to the registry, half of which were nonablative laser treatments,” Dr. Koza said in presenting the results. “Of the adverse events reported, nonphysicians and non-dermatologic physicians were more likely to be associated with severe or persistent adverse events.”
The American Society for Dermatologic Surgery Association and the Northwestern University Department of Dermatology launched CAPER. Previously, Dr. Koza said, AEs were typically reported only through the Food and Drug Administration’s AE reporting system. He noted that CAPER is the only voluntary national reporting registry for AEs from dermatologic procedures.
What the Registry Shows So Far
The registry matched 72 of the 81 cases with type of provider, with dermatologist-conducted procedures (51, 70.8%) comprising the majority, followed by nonphysician-conducted procedures (14, 19.4%) and nondermatologist physician–conducted procedures (7, 9.7%).
Of the 81 total cases, the following reports were related to laser and energy device treatments: 12 (14.3%) from nonablative laser treatments, five (6%) from light treatments, and three (3.6%) from ablative laser treatments, Dr. Koza said.
Among nonablative laser treatments, the most common AE was blistering (six reports, 50%). Scar, pain, and hypopigmentation accounted for two cases each (16.67%). Dermatologists performed seven of these cases (58.3%); nonphysicians, four (33.3%); and a non-dermatologist physician, one (8.3%).
For intense pulsed-light treatments, burns were the most common AEs (three reports, 60%), with swelling and inflammation each accounting for one case (20%). Three of these cases (75%) were confirmed to have been performed by nonphysicians.
The ablative laser treatment AEs included one case each of hypopigmentation, scar, and erythema. Two of the three cases were confirmed to have been performed by dermatologists.
Dr. Koza acknowledged the low number of cases is a limitation of this analysis of registry reports. A future goal for CAPER is to publicize it more, he said. “The registry is only 3 years old,” he told this news organization. “Hopefully, we can get more data as time goes on. We’ve been getting more and more each year.” CAPER adapted data entry forms used in other registries.
Submitting a case to the registry takes about 15 minutes of the provider’s time, Dr. Koza said. “We can streamline that to make it easier for people to submit their adverse events,” he said in an interview.
Only registry staff have access to the reports, and when reported, the data “is de-identified and any identifying information pertaining to the patient or reporter is removed,” according to a statement on the CAPER website.
‘Needs a Little Help’
Jennifer Lin, MD, a dermatologist at Brigham and Women’s Hospital and Dana-Farber Cancer Institute in Boston, who was at the meeting, commented on the onerous reporting process and the “low” enrollment. “It’s such an important initiative and with everyone over-logging e-mails, a 15-minute entry just is not going to cut it,” she told this news organization.
For providers, reporting AEs is stressful, she said. “As it is, it’s hard to voluntarily submit an adverse event,” Dr. Lin continued. “There’s a feeling of shame. Hospitals require it in order to monitor adverse events, but there’s no monitoring when you’re out in your own private practice.”
“The idea is excellent, but I think to facilitate better enrollment, the word has to get out at all these meetings” and make it easier to submit cases, Dr. Lin added. “It’s a good idea, but it needs a little help.”
Information on submitting AE reports to CAPER is available on the CAPER website.
Dr. Koza and Dr. Lin had no relevant relationships to disclose.
A version of this article first appeared on Medscape.com.
FROM ASLMS 2024
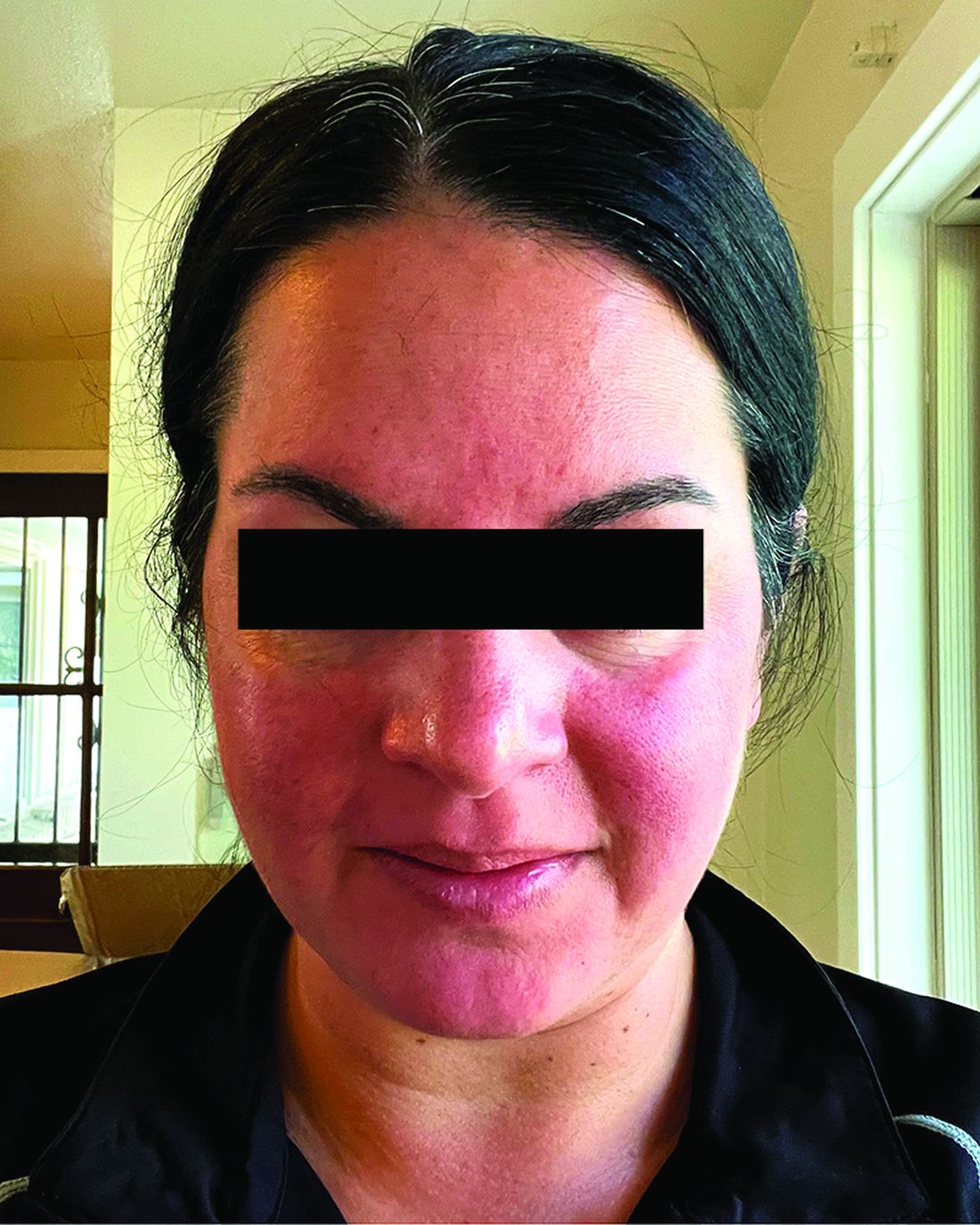
Migraine Drug Reduces Rosacea Flushing, Erythema in Small Study
In. Skin-related quality-of-life (QOL) measures also improved, albeit modestly.
The study was published in JAMA Dermatology.
“The transient erythema of rosacea is one of the most challenging rosacea symptoms to treat,” Emmy Graber, MD, MBA, who was not involved with the study, said in an interview. “As flushing can adversely impact quality of life in our rosacea patients, it is important to find therapeutic options for our patients. This study is exciting, not only because the treatment was successful for a notable number of patients, but also because it involved a drug with a novel mode of action in rosacea.” Dr. Graber practices in Boston and is an affiliate clinical instructor at Northeastern University, Boston.
Guy F. Webster, MD, PhD, clinical professor of dermatology, Sidney Kimmel Medical College, Thomas Jefferson University, Philadelphia, added, “The interesting thing about this study is that it gives us a new target to think about for therapy. But it’s a long way from saying we can use it tomorrow.” He was not involved with the study but was also asked to comment on the findings.
Spotlight on CGRP
Rosacea’s pathophysiology remains incompletely understood, wrote Nita K.F. Wienholtz, MD, PhD, Department of Dermatology, Bispebjerg and Frederiksberg Hospital, University of Copenhagen, Denmark, and coinvestigators. However, they added, mounting evidence suggests a possible role for CGRP. For example, a study published in JAMA Dermatology in 2015 revealed elevated CGRP levels in facial skin biopsies from patients with rosacea.
For the present study, the investigators enrolled 30 adults (including 23 women) with rosacea who experienced at least 15 days of moderate to severe erythema or extreme flushing during a 4-week, treatment-free run-in period. Most participants (87%) had previously failed one or more rosacea treatments because of a lack of efficacy or adverse reactions, and 43% had failed three or more treatments.
Participants received 3-monthly 140-mg doses of erenumab, which is approved by the Food and Drug Administration for migraine prevention. Patients recorded scores on the Patient Self-Assessment (PSA) and item 2 of the Flushing Assessment Tool online daily and made a final follow-up visit 12 weeks after the third dose.
Among the 27 patients who completed the study, the mean number of days with moderate to severe flushing from week 9 to week 12 fell by 6.9 from 23.6 days over 4 weeks at baseline (P < .001). Patients most severely affected by flushing at baseline experienced an 81% decline in days with severe to extreme flushing. Overall, 26% of patients experienced at least 50% reductions in moderate to extreme flushing days. The number of days with moderate to severe erythema as measured by PSA fell by 8.1 (mean) from baseline, and 56% of patients experienced at least 50% reductions in PSA scores. No unexpected safety signals emerged.
Questions Over QOL Data
“Although there were significant decreases in flushing and erythema,” wrote John S. Barbieri, MD, MBA, in an accompanying Editor’s Note, “the present study had relatively modest improvements in quality of life.” He is director of the Advanced Acne Therapeutics Clinic, Brigham and Women’s Hospital, Boston, and associate editor and evidence-based practice editor of JAMA Dermatology.
Compared with baseline (6.22), mean Dermatology Life Quality Index scores fell 2.08 points and 2.73 points at weeks 8 and 20, respectively (P = .004 and .003). At the same intervals, the mean baseline Rosacea Quality of Life score (48.22) decreased by 2.58 points and 4.14 points, respectively (P = .04 and .02).
No significant changes appeared in gauges of anxiety and depression. These findings, authors wrote, could stem from their decision to omit a follow-up visit at week 12 — where they may have seen mental-health effects which disappeared by week 20 — in response to patients’ logistical concerns.
However, Dr. Webster questioned the value of QOL measurements in rosacea. “Quality-of-life measures are blunt instruments,” he explained, and reducing severe itching or chronic pain improves the lives of affected patients. “But what question are you going to ask to tease out whether being less red-cheeked has made someone’s life easier? It’s not a problem that lends itself to quality-of-life assessments.” Moreover, he said, regulators who increasingly require such measures in clinical trials ignore this point, creating challenges for drug developers and researchers.
Because the study was neither blinded nor controlled, Dr. Webster suggested considering it a tantalizing proof of concept. “If I were putting money into a CGRP inhibitor, I’d want at least a small, placebo-controlled, double-blinded study.”
Study authors and Dr. Barbieri recommended larger randomized studies involving different populations and erenumab doses. For now, Dr. Barbieri wrote, CGRP inhibition represents a promising potential strategy for patients who have rosacea with comorbid migraine or recalcitrant flushing and erythema.
Dr. Wienholtz reported no relevant financial interests. Dr. Barbieri had no related disclosures. Dr. Webster reported no relevant financial interests. Dr. Graber reported no conflicts related to erenumab but consults for other companies with rosacea-related products including Galderma. The study was supported by and conducted in collaboration with Novartis Pharma AG. Additional funding came from the Novo Nordisk Foundation and the Lundbeck Foundation.
A version of this article appeared on Medscape.com.
In. Skin-related quality-of-life (QOL) measures also improved, albeit modestly.
The study was published in JAMA Dermatology.
“The transient erythema of rosacea is one of the most challenging rosacea symptoms to treat,” Emmy Graber, MD, MBA, who was not involved with the study, said in an interview. “As flushing can adversely impact quality of life in our rosacea patients, it is important to find therapeutic options for our patients. This study is exciting, not only because the treatment was successful for a notable number of patients, but also because it involved a drug with a novel mode of action in rosacea.” Dr. Graber practices in Boston and is an affiliate clinical instructor at Northeastern University, Boston.
Guy F. Webster, MD, PhD, clinical professor of dermatology, Sidney Kimmel Medical College, Thomas Jefferson University, Philadelphia, added, “The interesting thing about this study is that it gives us a new target to think about for therapy. But it’s a long way from saying we can use it tomorrow.” He was not involved with the study but was also asked to comment on the findings.
Spotlight on CGRP
Rosacea’s pathophysiology remains incompletely understood, wrote Nita K.F. Wienholtz, MD, PhD, Department of Dermatology, Bispebjerg and Frederiksberg Hospital, University of Copenhagen, Denmark, and coinvestigators. However, they added, mounting evidence suggests a possible role for CGRP. For example, a study published in JAMA Dermatology in 2015 revealed elevated CGRP levels in facial skin biopsies from patients with rosacea.
For the present study, the investigators enrolled 30 adults (including 23 women) with rosacea who experienced at least 15 days of moderate to severe erythema or extreme flushing during a 4-week, treatment-free run-in period. Most participants (87%) had previously failed one or more rosacea treatments because of a lack of efficacy or adverse reactions, and 43% had failed three or more treatments.
Participants received 3-monthly 140-mg doses of erenumab, which is approved by the Food and Drug Administration for migraine prevention. Patients recorded scores on the Patient Self-Assessment (PSA) and item 2 of the Flushing Assessment Tool online daily and made a final follow-up visit 12 weeks after the third dose.
Among the 27 patients who completed the study, the mean number of days with moderate to severe flushing from week 9 to week 12 fell by 6.9 from 23.6 days over 4 weeks at baseline (P < .001). Patients most severely affected by flushing at baseline experienced an 81% decline in days with severe to extreme flushing. Overall, 26% of patients experienced at least 50% reductions in moderate to extreme flushing days. The number of days with moderate to severe erythema as measured by PSA fell by 8.1 (mean) from baseline, and 56% of patients experienced at least 50% reductions in PSA scores. No unexpected safety signals emerged.
Questions Over QOL Data
“Although there were significant decreases in flushing and erythema,” wrote John S. Barbieri, MD, MBA, in an accompanying Editor’s Note, “the present study had relatively modest improvements in quality of life.” He is director of the Advanced Acne Therapeutics Clinic, Brigham and Women’s Hospital, Boston, and associate editor and evidence-based practice editor of JAMA Dermatology.
Compared with baseline (6.22), mean Dermatology Life Quality Index scores fell 2.08 points and 2.73 points at weeks 8 and 20, respectively (P = .004 and .003). At the same intervals, the mean baseline Rosacea Quality of Life score (48.22) decreased by 2.58 points and 4.14 points, respectively (P = .04 and .02).
No significant changes appeared in gauges of anxiety and depression. These findings, authors wrote, could stem from their decision to omit a follow-up visit at week 12 — where they may have seen mental-health effects which disappeared by week 20 — in response to patients’ logistical concerns.
However, Dr. Webster questioned the value of QOL measurements in rosacea. “Quality-of-life measures are blunt instruments,” he explained, and reducing severe itching or chronic pain improves the lives of affected patients. “But what question are you going to ask to tease out whether being less red-cheeked has made someone’s life easier? It’s not a problem that lends itself to quality-of-life assessments.” Moreover, he said, regulators who increasingly require such measures in clinical trials ignore this point, creating challenges for drug developers and researchers.
Because the study was neither blinded nor controlled, Dr. Webster suggested considering it a tantalizing proof of concept. “If I were putting money into a CGRP inhibitor, I’d want at least a small, placebo-controlled, double-blinded study.”
Study authors and Dr. Barbieri recommended larger randomized studies involving different populations and erenumab doses. For now, Dr. Barbieri wrote, CGRP inhibition represents a promising potential strategy for patients who have rosacea with comorbid migraine or recalcitrant flushing and erythema.
Dr. Wienholtz reported no relevant financial interests. Dr. Barbieri had no related disclosures. Dr. Webster reported no relevant financial interests. Dr. Graber reported no conflicts related to erenumab but consults for other companies with rosacea-related products including Galderma. The study was supported by and conducted in collaboration with Novartis Pharma AG. Additional funding came from the Novo Nordisk Foundation and the Lundbeck Foundation.
A version of this article appeared on Medscape.com.
In. Skin-related quality-of-life (QOL) measures also improved, albeit modestly.
The study was published in JAMA Dermatology.
“The transient erythema of rosacea is one of the most challenging rosacea symptoms to treat,” Emmy Graber, MD, MBA, who was not involved with the study, said in an interview. “As flushing can adversely impact quality of life in our rosacea patients, it is important to find therapeutic options for our patients. This study is exciting, not only because the treatment was successful for a notable number of patients, but also because it involved a drug with a novel mode of action in rosacea.” Dr. Graber practices in Boston and is an affiliate clinical instructor at Northeastern University, Boston.
Guy F. Webster, MD, PhD, clinical professor of dermatology, Sidney Kimmel Medical College, Thomas Jefferson University, Philadelphia, added, “The interesting thing about this study is that it gives us a new target to think about for therapy. But it’s a long way from saying we can use it tomorrow.” He was not involved with the study but was also asked to comment on the findings.
Spotlight on CGRP
Rosacea’s pathophysiology remains incompletely understood, wrote Nita K.F. Wienholtz, MD, PhD, Department of Dermatology, Bispebjerg and Frederiksberg Hospital, University of Copenhagen, Denmark, and coinvestigators. However, they added, mounting evidence suggests a possible role for CGRP. For example, a study published in JAMA Dermatology in 2015 revealed elevated CGRP levels in facial skin biopsies from patients with rosacea.
For the present study, the investigators enrolled 30 adults (including 23 women) with rosacea who experienced at least 15 days of moderate to severe erythema or extreme flushing during a 4-week, treatment-free run-in period. Most participants (87%) had previously failed one or more rosacea treatments because of a lack of efficacy or adverse reactions, and 43% had failed three or more treatments.
Participants received 3-monthly 140-mg doses of erenumab, which is approved by the Food and Drug Administration for migraine prevention. Patients recorded scores on the Patient Self-Assessment (PSA) and item 2 of the Flushing Assessment Tool online daily and made a final follow-up visit 12 weeks after the third dose.
Among the 27 patients who completed the study, the mean number of days with moderate to severe flushing from week 9 to week 12 fell by 6.9 from 23.6 days over 4 weeks at baseline (P < .001). Patients most severely affected by flushing at baseline experienced an 81% decline in days with severe to extreme flushing. Overall, 26% of patients experienced at least 50% reductions in moderate to extreme flushing days. The number of days with moderate to severe erythema as measured by PSA fell by 8.1 (mean) from baseline, and 56% of patients experienced at least 50% reductions in PSA scores. No unexpected safety signals emerged.
Questions Over QOL Data
“Although there were significant decreases in flushing and erythema,” wrote John S. Barbieri, MD, MBA, in an accompanying Editor’s Note, “the present study had relatively modest improvements in quality of life.” He is director of the Advanced Acne Therapeutics Clinic, Brigham and Women’s Hospital, Boston, and associate editor and evidence-based practice editor of JAMA Dermatology.
Compared with baseline (6.22), mean Dermatology Life Quality Index scores fell 2.08 points and 2.73 points at weeks 8 and 20, respectively (P = .004 and .003). At the same intervals, the mean baseline Rosacea Quality of Life score (48.22) decreased by 2.58 points and 4.14 points, respectively (P = .04 and .02).
No significant changes appeared in gauges of anxiety and depression. These findings, authors wrote, could stem from their decision to omit a follow-up visit at week 12 — where they may have seen mental-health effects which disappeared by week 20 — in response to patients’ logistical concerns.
However, Dr. Webster questioned the value of QOL measurements in rosacea. “Quality-of-life measures are blunt instruments,” he explained, and reducing severe itching or chronic pain improves the lives of affected patients. “But what question are you going to ask to tease out whether being less red-cheeked has made someone’s life easier? It’s not a problem that lends itself to quality-of-life assessments.” Moreover, he said, regulators who increasingly require such measures in clinical trials ignore this point, creating challenges for drug developers and researchers.
Because the study was neither blinded nor controlled, Dr. Webster suggested considering it a tantalizing proof of concept. “If I were putting money into a CGRP inhibitor, I’d want at least a small, placebo-controlled, double-blinded study.”
Study authors and Dr. Barbieri recommended larger randomized studies involving different populations and erenumab doses. For now, Dr. Barbieri wrote, CGRP inhibition represents a promising potential strategy for patients who have rosacea with comorbid migraine or recalcitrant flushing and erythema.
Dr. Wienholtz reported no relevant financial interests. Dr. Barbieri had no related disclosures. Dr. Webster reported no relevant financial interests. Dr. Graber reported no conflicts related to erenumab but consults for other companies with rosacea-related products including Galderma. The study was supported by and conducted in collaboration with Novartis Pharma AG. Additional funding came from the Novo Nordisk Foundation and the Lundbeck Foundation.
A version of this article appeared on Medscape.com.
FROM JAMA DERMATOLOGY
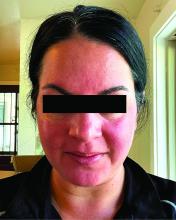
Lentigines: Study Finds Less PIH With Modified Laser Treatment
BALTIMORE — Laser treatment for solar lentigines in individuals with darker skin types has long been associated with a higher risk of postinflammatory hyperpigmentation (PIH), but.
The study enrolled 27 patients with solar lentigines and Fitzpatrick skin types (FSTs) III-IV, Woraphong Manuskiatti, MD, professor of dermatology at Siriraj Hospital, Mahidol University, Bangkok, reported at the annual meeting of the American Society for Laser Medicine and Surgery. They received the fractional beam treatment on one side of the face and the full-beam on the other side. At 6 months, the incidence of PIH was about 81% lower on the fractional-beam side, Dr. Manuskiatti said.
“In the past, when we used laser to treat pigmented lesions, we used the so-called full-beam technique on the pigmented area,” Dr. Manuskiatti told this news organization. “From the study, we found that you don’t need to treat it at 100%. You can fractionally treat the pigmented lesion and get a really comparable treatment outcome and, at that reduced beam, less incidence of postinflammatory hyperpigmentation.”
Study Design and Results
Of the 27 patients in the study, 12 were FST III (44%), 14 were FST IV (52%), and one was FST V (4%). On the fractional-beam side, the laser was delivered through a 9-mm spot size with an average fluence of 0.47 J/cm² at a frequency of 2 Hz for a total of two passes without pulse overlapping. On the full-beam side, the laser was operated with a 4.5-mm handpiece, with fluence ranging from 0.3 to 0.7 J/cm² (using an endpoint of slight darkening of the pigmented lesion) at 2 Hz.
The patients received a single treatment and had a clinical evaluation and color reading assessments at 2 weeks, 1 month, 3 months, and 6 months after the treatment. Twenty-five patients completed the study.
The researchers found no statistically significant differences in lesional clearance between the two techniques at any of the follow-up assessments, Dr. Manuskiatti said. “This might be one of the alternative treatments of treating solar lentigines in dark-skinned patients,” he said when presenting the study results.
He reported the rates of PIH on the full-beam and fractional-beam sides, respectively, at the following intervals were: 64% and 8% at 2 weeks, 80% and 32% at 1 month, 96% and 36% at 3 months, and 88% and 16% at 6 months.
“The incidence of PIH on the full-beam side was statistically higher than that on the fractional-beam side throughout the follow-up period,” he said. Transient and mild hypopigmentation was observed in one patient (4%) on the fractional-beam side and in five (20%) on the full-beam side. Dr. Manuskiatti added that no other adverse effects were documented during the study.
“ Normally when you use laser to treat skin type I or II, you don’t have … PIH or darkening of the skin,” Dr. Manuskiatti told this news organization, “but when you have skin type III and above, you run into a really high incidence of postinflammatory hyperpigmentation — and treating that with fractional beam can lead to a reduced incidence of darkening of the skin afterward.”
A Lower-Cost Option
This study showed that the 532-nm picosecond laser with fractional beam MLA is a useful option for patients with darker skin types, Kelly Stankiewicz, MD, a dermatologist who practices in Park City, Utah, and moderated the session where these results were presented, told this news organization.
“The most challenging thing about treating lentigines in darker skin types is preventing potential side effects, mainly dyspigmentation,” she said after the meeting. “These side effects are, for the most part, temporary, but they can take 6-18 months to resolve, so it’s important to prevent them in the first place.”
She noted that the 532-nm and 1064-nm wavelengths are the most commonly available for picosecond lasers and that they’re easier to produce and less expensive. “There are picosecond lasers with middle wavelengths in the red light to near-infrared range (650-785 nm) that are better for darker skin types because they are more gentle yet still effective at targeting pigment, but these lasers are more expensive and less widely available,” Dr. Stankiewicz said.
“The microlens array, used in this study with the 532-nm wavelength, is an inexpensive piece that fits at the end of the laser,” she added. “So, to have an option that turns a 532-nm laser into a safer device for the treatment of lentigines in darker skin types is very helpful.”
Dr. Manuskiatti and Dr. Stankiewicz had no relevant disclosures to report.
A version of this article first appeared on Medscape.com.
BALTIMORE — Laser treatment for solar lentigines in individuals with darker skin types has long been associated with a higher risk of postinflammatory hyperpigmentation (PIH), but.
The study enrolled 27 patients with solar lentigines and Fitzpatrick skin types (FSTs) III-IV, Woraphong Manuskiatti, MD, professor of dermatology at Siriraj Hospital, Mahidol University, Bangkok, reported at the annual meeting of the American Society for Laser Medicine and Surgery. They received the fractional beam treatment on one side of the face and the full-beam on the other side. At 6 months, the incidence of PIH was about 81% lower on the fractional-beam side, Dr. Manuskiatti said.
“In the past, when we used laser to treat pigmented lesions, we used the so-called full-beam technique on the pigmented area,” Dr. Manuskiatti told this news organization. “From the study, we found that you don’t need to treat it at 100%. You can fractionally treat the pigmented lesion and get a really comparable treatment outcome and, at that reduced beam, less incidence of postinflammatory hyperpigmentation.”
Study Design and Results
Of the 27 patients in the study, 12 were FST III (44%), 14 were FST IV (52%), and one was FST V (4%). On the fractional-beam side, the laser was delivered through a 9-mm spot size with an average fluence of 0.47 J/cm² at a frequency of 2 Hz for a total of two passes without pulse overlapping. On the full-beam side, the laser was operated with a 4.5-mm handpiece, with fluence ranging from 0.3 to 0.7 J/cm² (using an endpoint of slight darkening of the pigmented lesion) at 2 Hz.
The patients received a single treatment and had a clinical evaluation and color reading assessments at 2 weeks, 1 month, 3 months, and 6 months after the treatment. Twenty-five patients completed the study.
The researchers found no statistically significant differences in lesional clearance between the two techniques at any of the follow-up assessments, Dr. Manuskiatti said. “This might be one of the alternative treatments of treating solar lentigines in dark-skinned patients,” he said when presenting the study results.
He reported the rates of PIH on the full-beam and fractional-beam sides, respectively, at the following intervals were: 64% and 8% at 2 weeks, 80% and 32% at 1 month, 96% and 36% at 3 months, and 88% and 16% at 6 months.
“The incidence of PIH on the full-beam side was statistically higher than that on the fractional-beam side throughout the follow-up period,” he said. Transient and mild hypopigmentation was observed in one patient (4%) on the fractional-beam side and in five (20%) on the full-beam side. Dr. Manuskiatti added that no other adverse effects were documented during the study.
“ Normally when you use laser to treat skin type I or II, you don’t have … PIH or darkening of the skin,” Dr. Manuskiatti told this news organization, “but when you have skin type III and above, you run into a really high incidence of postinflammatory hyperpigmentation — and treating that with fractional beam can lead to a reduced incidence of darkening of the skin afterward.”
A Lower-Cost Option
This study showed that the 532-nm picosecond laser with fractional beam MLA is a useful option for patients with darker skin types, Kelly Stankiewicz, MD, a dermatologist who practices in Park City, Utah, and moderated the session where these results were presented, told this news organization.
“The most challenging thing about treating lentigines in darker skin types is preventing potential side effects, mainly dyspigmentation,” she said after the meeting. “These side effects are, for the most part, temporary, but they can take 6-18 months to resolve, so it’s important to prevent them in the first place.”
She noted that the 532-nm and 1064-nm wavelengths are the most commonly available for picosecond lasers and that they’re easier to produce and less expensive. “There are picosecond lasers with middle wavelengths in the red light to near-infrared range (650-785 nm) that are better for darker skin types because they are more gentle yet still effective at targeting pigment, but these lasers are more expensive and less widely available,” Dr. Stankiewicz said.
“The microlens array, used in this study with the 532-nm wavelength, is an inexpensive piece that fits at the end of the laser,” she added. “So, to have an option that turns a 532-nm laser into a safer device for the treatment of lentigines in darker skin types is very helpful.”
Dr. Manuskiatti and Dr. Stankiewicz had no relevant disclosures to report.
A version of this article first appeared on Medscape.com.
BALTIMORE — Laser treatment for solar lentigines in individuals with darker skin types has long been associated with a higher risk of postinflammatory hyperpigmentation (PIH), but.
The study enrolled 27 patients with solar lentigines and Fitzpatrick skin types (FSTs) III-IV, Woraphong Manuskiatti, MD, professor of dermatology at Siriraj Hospital, Mahidol University, Bangkok, reported at the annual meeting of the American Society for Laser Medicine and Surgery. They received the fractional beam treatment on one side of the face and the full-beam on the other side. At 6 months, the incidence of PIH was about 81% lower on the fractional-beam side, Dr. Manuskiatti said.
“In the past, when we used laser to treat pigmented lesions, we used the so-called full-beam technique on the pigmented area,” Dr. Manuskiatti told this news organization. “From the study, we found that you don’t need to treat it at 100%. You can fractionally treat the pigmented lesion and get a really comparable treatment outcome and, at that reduced beam, less incidence of postinflammatory hyperpigmentation.”
Study Design and Results
Of the 27 patients in the study, 12 were FST III (44%), 14 were FST IV (52%), and one was FST V (4%). On the fractional-beam side, the laser was delivered through a 9-mm spot size with an average fluence of 0.47 J/cm² at a frequency of 2 Hz for a total of two passes without pulse overlapping. On the full-beam side, the laser was operated with a 4.5-mm handpiece, with fluence ranging from 0.3 to 0.7 J/cm² (using an endpoint of slight darkening of the pigmented lesion) at 2 Hz.
The patients received a single treatment and had a clinical evaluation and color reading assessments at 2 weeks, 1 month, 3 months, and 6 months after the treatment. Twenty-five patients completed the study.
The researchers found no statistically significant differences in lesional clearance between the two techniques at any of the follow-up assessments, Dr. Manuskiatti said. “This might be one of the alternative treatments of treating solar lentigines in dark-skinned patients,” he said when presenting the study results.
He reported the rates of PIH on the full-beam and fractional-beam sides, respectively, at the following intervals were: 64% and 8% at 2 weeks, 80% and 32% at 1 month, 96% and 36% at 3 months, and 88% and 16% at 6 months.
“The incidence of PIH on the full-beam side was statistically higher than that on the fractional-beam side throughout the follow-up period,” he said. Transient and mild hypopigmentation was observed in one patient (4%) on the fractional-beam side and in five (20%) on the full-beam side. Dr. Manuskiatti added that no other adverse effects were documented during the study.
“ Normally when you use laser to treat skin type I or II, you don’t have … PIH or darkening of the skin,” Dr. Manuskiatti told this news organization, “but when you have skin type III and above, you run into a really high incidence of postinflammatory hyperpigmentation — and treating that with fractional beam can lead to a reduced incidence of darkening of the skin afterward.”
A Lower-Cost Option
This study showed that the 532-nm picosecond laser with fractional beam MLA is a useful option for patients with darker skin types, Kelly Stankiewicz, MD, a dermatologist who practices in Park City, Utah, and moderated the session where these results were presented, told this news organization.
“The most challenging thing about treating lentigines in darker skin types is preventing potential side effects, mainly dyspigmentation,” she said after the meeting. “These side effects are, for the most part, temporary, but they can take 6-18 months to resolve, so it’s important to prevent them in the first place.”
She noted that the 532-nm and 1064-nm wavelengths are the most commonly available for picosecond lasers and that they’re easier to produce and less expensive. “There are picosecond lasers with middle wavelengths in the red light to near-infrared range (650-785 nm) that are better for darker skin types because they are more gentle yet still effective at targeting pigment, but these lasers are more expensive and less widely available,” Dr. Stankiewicz said.
“The microlens array, used in this study with the 532-nm wavelength, is an inexpensive piece that fits at the end of the laser,” she added. “So, to have an option that turns a 532-nm laser into a safer device for the treatment of lentigines in darker skin types is very helpful.”
Dr. Manuskiatti and Dr. Stankiewicz had no relevant disclosures to report.
A version of this article first appeared on Medscape.com.
FROM ASLMS 2024
How These Young MDs Impressed the Hell Out of Their Bosses
Safe to say that anyone undertaking the physician journey does so with intense motivation and book smarts. Still, it can be incredibly hard to stand out. Everyone’s a go-getter, but what’s the X factor?
Here’s what they said ...
Lesson #1: Never Be Scared to Ask
Brien Barnewolt, MD, chairman and chief of the Department of Emergency Medicine at Tufts Medical Center, was very much surprised when a resident named Scott G. Weiner did something unexpected: Go after a job in the fall of his junior year residency instead of following the typical senior year trajectory.
“It’s very unusual for a trainee to apply for a job virtually a year ahead of schedule. But he knew what he wanted,” said Dr. Barnewolt. “I’d never had anybody come to me in that same scenario, and I’ve been doing this a long time.”
Under normal circumstances it would’ve been easy for Dr. Barnewolt to say no. But the unexpected request made him and his colleagues take a closer look, and they were impressed with Dr. Weiner’s skills. That, paired with his ambition and demeanor, compelled them to offer him an early job. But there’s more.
As the next year approached, Dr. Weiner explained he had an opportunity to work in emergency medicine in Tuscany and asked if he could take a 1-year delayed start for the position he applied a year early for.
The department held his position, and upon his return, Dr. Weiner made a lasting impact at Tufts before eventually moving on. “He outgrew us, which is nice to see,” Dr. Barnewolt said. (Dr. Weiner is currently McGraw Distinguished Chair in Emergency Medicine at Brigham and Women’s Hospital and associate professor at Harvard Medical School.)
Bottom line: Why did Dr. Barnewolt and his colleagues do so much to accommodate a young candidate? Yes, Dr. Weiner was talented, but he was also up-front about his ambitions from the get-go. Dr. Barnewolt said that kind of initiative can only be looked at positively.
“My advice would be, if you see an opportunity or a potential place where you might want to work, put out those feelers, start those conversations,” he said. “It’s not too early, especially in certain specialties, where the job market is very tight. Then, when circumstances change, be open about it and have that conversation. The worst that somebody can say is no, so it never hurts to be honest and open about where you want to go and what you want to be.”
Lesson #2: Chase Your Passion ‘Relentlessly’
Vance G. Fowler, MD, MHS, an infectious disease specialist at Duke University School of Medicine, runs a laboratory that researches methicillin-resistant Staphylococcus aureus (MRSA). Over the years, he’s mentored many doctors but understands the ambitions of young trainees don’t always align with the little free time that they have. “Many of them drop away when you give them a [side] project,” he said.
So when Tori Kinamon asked him to work on an MRSA project — in her first year — he gave her one that focused on researching vertebral osteomyelitis, a bone infection that can coincide with S aureus. What Dr. Fowler didn’t know: Kinamon (now MD) had been a competitive gymnast at Brown and battled her own life-threatening infection with MRSA.
“To my absolute astonishment, not only did she stick to it, but she was able to compile a presentation on the science and gave an oral presentation within a year of walking in the door,” said Dr. Fowler.
She went on to lead an initiative between the National Institutes of Health and US Food and Drug Administration to create endpoints for clinical drug trials, all of which occurred before starting her residency, which she’s about to embark upon.
Dr. Kinamon’s a good example, he said, of what happens when you add genuine passion to book smarts. Those who do always stand out because you can’t fake that. “Find your passion, and then chase it down relentlessly,” he said. “Once you’ve found your passion, things get easy because it stops being work and it starts being something else.”
If you haven’t identified a focus area, Dr. Fowler said to “be agnostic and observant. Keep your eyes open and your options open because you may surprise yourself. It may turn out that you end up liking something a whole lot more than you thought you did.”
Lesson #3: When You Say You’ve Always Wanted to Do Something, Do Something
As the chief of pulmonary and critical care medicine at the Northwestern Medicine Canning Thoracic Institute, Scott Budinger, MD, often hears lip service from doctors who want to put their skills to use in their local communities. One of his students actually did it.
Justin Fiala, MD, a pulmonary, critical care, and sleep specialist at Northwestern Medicine, joined Northwestern as a pulmonary fellow with a big interest in addressing health equity issues.
Dr. Fiala began volunteering with CommunityHealth during his fellowship and saw that many patients of the free Chicago-area clinic needed help with sleep disorders. He launched the organization’s first sleep clinic and its Patient-Centered Apnea Protocols Initiative.
“He developed a plan with some of the partners of the sleep apnea equipment to do home sleep testing for these patients that’s free of cost,” said Dr. Budinger.
Dr. Fiala goes in on Saturdays and runs a free clinic conducting sleep studies for patients and outfits them with devices that they need to improve their conditions, said Dr. Budinger.
“And these patients are the severest of the severe patients,” he said. “These are people that have severe sleep apnea that are driving around the roads, oftentimes don’t have insurance because they’re also precluded from having auto insurance. So, this is really something that not just benefits these patients but benefits our whole community.”
The fact that Dr. Fiala followed through on something that all doctors aspire to do — and in the middle of a very busy training program — is something that Dr. Budinger said makes him stand out in a big way.
“If you talk to any of our trainees or young faculty, everybody’s interested in addressing the issue of health disparities,” said Dr. Budinger. “Justin looked at that and said, ‘Well, you know, I’m not interested in talking about it. What can I do about this problem? And how can I actually get boots on the ground and help?’ That requires a big activation energy that many people don’t have.”
Lesson #4: Be a People-Person and a Patient-Person
When hiring employees at American Family Care in Portland, Oregon, Andrew Miller, MD, director of provider training, is always on the lookout for young MDs with emotional intelligence and a good bedside manner. He has been recently blown away, however, by a young physician’s assistant named Joseph Van Bindsbergen, PA-C, who was described as “all-around wonderful” during his reference check.
“Having less than 6 months of experience out of school, he is our highest ranked provider, whether it’s a nurse practitioner, PA, or doctor, in terms of patient satisfaction,” said Dr. Miller. The young PA has an “unprecedented perfect score” on his NPS rating.
Why? Patients said they’ve never felt as heard as they felt with Van Bindsbergen.
“That’s the thing I think that the up-and-coming providers should be focusing on is making your patients feel heard,” explained Dr. Miller. Van Bindsbergen is great at building rapport with a patient, whether they are 6 or 96. “He doesn’t just ask about sore throat symptoms. He asks, ‘what is the impact on your life of the sore throat? How does it affect your family or your work? What do you think this could be besides just strep? What are your concerns?’ ”
Dr. Miller said the magic of Van Bindsbergen is that he has an innate ability to look at patients “not just as a diagnosis but as a person, which they love.”
Lesson #5: Remember to Make That Difference With Each Patient
Doctors are used to swooping in and seeing a patient, ordering further testing if needed, and then moving on to the next patient. But one young intern at the start of his medical career broke this mold by giving a very anxious patient some much-needed support.
“There was a resident who was working overnight, and this poor young woman came in who had a new diagnosis of an advanced illness and a lot of anxiety around her condition, the newness of it, and the impact this is going to have on her family and her life,” said Elizabeth Horn Prsic, MD, assistant professor at Yale School of Medicine and firm chief for medical oncology and the director of Adult Inpatient Palliative Care.
Dr. Prsic found out the next morning that this trainee accompanied the patient to the MRI and held her hand as much as he was allowed to throughout the entire experience. “I was like, ‘wait you went down with her to radiology?’ And he’s like, ‘Yes, I was there the whole time,’ ” she recalled.
This gesture not only helped the patient feel calmer after receiving a potentially life-altering diagnosis but also helped ensure the test results were as clear as possible.
“If the study is not done well and a patient is moving or uncomfortable, it has to be stopped early or paused,” said Dr. Prsic. “Then the study is not very useful. In situations like these, medical decisions may be made based on imperfect data. The fact that we had this full complete good quality scan helped us get the care that she needed in a much timelier manner to help her and to move along the care that she that was medically appropriate for her.”
Dr. Prsic got emotional reflecting on the experience. Working at Yale, she saw a ton of intelligent doctors come through the ranks. But this gesture, she said, should serve as a reminder that “you don’t need to be the smartest person in the room to just be there for a patient. It was pure empathic presence and human connection. It gave me hope in the next generation of physicians.”
A version of this article appeared on Medscape.com.
Safe to say that anyone undertaking the physician journey does so with intense motivation and book smarts. Still, it can be incredibly hard to stand out. Everyone’s a go-getter, but what’s the X factor?
Here’s what they said ...
Lesson #1: Never Be Scared to Ask
Brien Barnewolt, MD, chairman and chief of the Department of Emergency Medicine at Tufts Medical Center, was very much surprised when a resident named Scott G. Weiner did something unexpected: Go after a job in the fall of his junior year residency instead of following the typical senior year trajectory.
“It’s very unusual for a trainee to apply for a job virtually a year ahead of schedule. But he knew what he wanted,” said Dr. Barnewolt. “I’d never had anybody come to me in that same scenario, and I’ve been doing this a long time.”
Under normal circumstances it would’ve been easy for Dr. Barnewolt to say no. But the unexpected request made him and his colleagues take a closer look, and they were impressed with Dr. Weiner’s skills. That, paired with his ambition and demeanor, compelled them to offer him an early job. But there’s more.
As the next year approached, Dr. Weiner explained he had an opportunity to work in emergency medicine in Tuscany and asked if he could take a 1-year delayed start for the position he applied a year early for.
The department held his position, and upon his return, Dr. Weiner made a lasting impact at Tufts before eventually moving on. “He outgrew us, which is nice to see,” Dr. Barnewolt said. (Dr. Weiner is currently McGraw Distinguished Chair in Emergency Medicine at Brigham and Women’s Hospital and associate professor at Harvard Medical School.)
Bottom line: Why did Dr. Barnewolt and his colleagues do so much to accommodate a young candidate? Yes, Dr. Weiner was talented, but he was also up-front about his ambitions from the get-go. Dr. Barnewolt said that kind of initiative can only be looked at positively.
“My advice would be, if you see an opportunity or a potential place where you might want to work, put out those feelers, start those conversations,” he said. “It’s not too early, especially in certain specialties, where the job market is very tight. Then, when circumstances change, be open about it and have that conversation. The worst that somebody can say is no, so it never hurts to be honest and open about where you want to go and what you want to be.”
Lesson #2: Chase Your Passion ‘Relentlessly’
Vance G. Fowler, MD, MHS, an infectious disease specialist at Duke University School of Medicine, runs a laboratory that researches methicillin-resistant Staphylococcus aureus (MRSA). Over the years, he’s mentored many doctors but understands the ambitions of young trainees don’t always align with the little free time that they have. “Many of them drop away when you give them a [side] project,” he said.
So when Tori Kinamon asked him to work on an MRSA project — in her first year — he gave her one that focused on researching vertebral osteomyelitis, a bone infection that can coincide with S aureus. What Dr. Fowler didn’t know: Kinamon (now MD) had been a competitive gymnast at Brown and battled her own life-threatening infection with MRSA.
“To my absolute astonishment, not only did she stick to it, but she was able to compile a presentation on the science and gave an oral presentation within a year of walking in the door,” said Dr. Fowler.
She went on to lead an initiative between the National Institutes of Health and US Food and Drug Administration to create endpoints for clinical drug trials, all of which occurred before starting her residency, which she’s about to embark upon.
Dr. Kinamon’s a good example, he said, of what happens when you add genuine passion to book smarts. Those who do always stand out because you can’t fake that. “Find your passion, and then chase it down relentlessly,” he said. “Once you’ve found your passion, things get easy because it stops being work and it starts being something else.”
If you haven’t identified a focus area, Dr. Fowler said to “be agnostic and observant. Keep your eyes open and your options open because you may surprise yourself. It may turn out that you end up liking something a whole lot more than you thought you did.”
Lesson #3: When You Say You’ve Always Wanted to Do Something, Do Something
As the chief of pulmonary and critical care medicine at the Northwestern Medicine Canning Thoracic Institute, Scott Budinger, MD, often hears lip service from doctors who want to put their skills to use in their local communities. One of his students actually did it.
Justin Fiala, MD, a pulmonary, critical care, and sleep specialist at Northwestern Medicine, joined Northwestern as a pulmonary fellow with a big interest in addressing health equity issues.
Dr. Fiala began volunteering with CommunityHealth during his fellowship and saw that many patients of the free Chicago-area clinic needed help with sleep disorders. He launched the organization’s first sleep clinic and its Patient-Centered Apnea Protocols Initiative.
“He developed a plan with some of the partners of the sleep apnea equipment to do home sleep testing for these patients that’s free of cost,” said Dr. Budinger.
Dr. Fiala goes in on Saturdays and runs a free clinic conducting sleep studies for patients and outfits them with devices that they need to improve their conditions, said Dr. Budinger.
“And these patients are the severest of the severe patients,” he said. “These are people that have severe sleep apnea that are driving around the roads, oftentimes don’t have insurance because they’re also precluded from having auto insurance. So, this is really something that not just benefits these patients but benefits our whole community.”
The fact that Dr. Fiala followed through on something that all doctors aspire to do — and in the middle of a very busy training program — is something that Dr. Budinger said makes him stand out in a big way.
“If you talk to any of our trainees or young faculty, everybody’s interested in addressing the issue of health disparities,” said Dr. Budinger. “Justin looked at that and said, ‘Well, you know, I’m not interested in talking about it. What can I do about this problem? And how can I actually get boots on the ground and help?’ That requires a big activation energy that many people don’t have.”
Lesson #4: Be a People-Person and a Patient-Person
When hiring employees at American Family Care in Portland, Oregon, Andrew Miller, MD, director of provider training, is always on the lookout for young MDs with emotional intelligence and a good bedside manner. He has been recently blown away, however, by a young physician’s assistant named Joseph Van Bindsbergen, PA-C, who was described as “all-around wonderful” during his reference check.
“Having less than 6 months of experience out of school, he is our highest ranked provider, whether it’s a nurse practitioner, PA, or doctor, in terms of patient satisfaction,” said Dr. Miller. The young PA has an “unprecedented perfect score” on his NPS rating.
Why? Patients said they’ve never felt as heard as they felt with Van Bindsbergen.
“That’s the thing I think that the up-and-coming providers should be focusing on is making your patients feel heard,” explained Dr. Miller. Van Bindsbergen is great at building rapport with a patient, whether they are 6 or 96. “He doesn’t just ask about sore throat symptoms. He asks, ‘what is the impact on your life of the sore throat? How does it affect your family or your work? What do you think this could be besides just strep? What are your concerns?’ ”
Dr. Miller said the magic of Van Bindsbergen is that he has an innate ability to look at patients “not just as a diagnosis but as a person, which they love.”
Lesson #5: Remember to Make That Difference With Each Patient
Doctors are used to swooping in and seeing a patient, ordering further testing if needed, and then moving on to the next patient. But one young intern at the start of his medical career broke this mold by giving a very anxious patient some much-needed support.
“There was a resident who was working overnight, and this poor young woman came in who had a new diagnosis of an advanced illness and a lot of anxiety around her condition, the newness of it, and the impact this is going to have on her family and her life,” said Elizabeth Horn Prsic, MD, assistant professor at Yale School of Medicine and firm chief for medical oncology and the director of Adult Inpatient Palliative Care.
Dr. Prsic found out the next morning that this trainee accompanied the patient to the MRI and held her hand as much as he was allowed to throughout the entire experience. “I was like, ‘wait you went down with her to radiology?’ And he’s like, ‘Yes, I was there the whole time,’ ” she recalled.
This gesture not only helped the patient feel calmer after receiving a potentially life-altering diagnosis but also helped ensure the test results were as clear as possible.
“If the study is not done well and a patient is moving or uncomfortable, it has to be stopped early or paused,” said Dr. Prsic. “Then the study is not very useful. In situations like these, medical decisions may be made based on imperfect data. The fact that we had this full complete good quality scan helped us get the care that she needed in a much timelier manner to help her and to move along the care that she that was medically appropriate for her.”
Dr. Prsic got emotional reflecting on the experience. Working at Yale, she saw a ton of intelligent doctors come through the ranks. But this gesture, she said, should serve as a reminder that “you don’t need to be the smartest person in the room to just be there for a patient. It was pure empathic presence and human connection. It gave me hope in the next generation of physicians.”
A version of this article appeared on Medscape.com.
Safe to say that anyone undertaking the physician journey does so with intense motivation and book smarts. Still, it can be incredibly hard to stand out. Everyone’s a go-getter, but what’s the X factor?
Here’s what they said ...
Lesson #1: Never Be Scared to Ask
Brien Barnewolt, MD, chairman and chief of the Department of Emergency Medicine at Tufts Medical Center, was very much surprised when a resident named Scott G. Weiner did something unexpected: Go after a job in the fall of his junior year residency instead of following the typical senior year trajectory.
“It’s very unusual for a trainee to apply for a job virtually a year ahead of schedule. But he knew what he wanted,” said Dr. Barnewolt. “I’d never had anybody come to me in that same scenario, and I’ve been doing this a long time.”
Under normal circumstances it would’ve been easy for Dr. Barnewolt to say no. But the unexpected request made him and his colleagues take a closer look, and they were impressed with Dr. Weiner’s skills. That, paired with his ambition and demeanor, compelled them to offer him an early job. But there’s more.
As the next year approached, Dr. Weiner explained he had an opportunity to work in emergency medicine in Tuscany and asked if he could take a 1-year delayed start for the position he applied a year early for.
The department held his position, and upon his return, Dr. Weiner made a lasting impact at Tufts before eventually moving on. “He outgrew us, which is nice to see,” Dr. Barnewolt said. (Dr. Weiner is currently McGraw Distinguished Chair in Emergency Medicine at Brigham and Women’s Hospital and associate professor at Harvard Medical School.)
Bottom line: Why did Dr. Barnewolt and his colleagues do so much to accommodate a young candidate? Yes, Dr. Weiner was talented, but he was also up-front about his ambitions from the get-go. Dr. Barnewolt said that kind of initiative can only be looked at positively.
“My advice would be, if you see an opportunity or a potential place where you might want to work, put out those feelers, start those conversations,” he said. “It’s not too early, especially in certain specialties, where the job market is very tight. Then, when circumstances change, be open about it and have that conversation. The worst that somebody can say is no, so it never hurts to be honest and open about where you want to go and what you want to be.”
Lesson #2: Chase Your Passion ‘Relentlessly’
Vance G. Fowler, MD, MHS, an infectious disease specialist at Duke University School of Medicine, runs a laboratory that researches methicillin-resistant Staphylococcus aureus (MRSA). Over the years, he’s mentored many doctors but understands the ambitions of young trainees don’t always align with the little free time that they have. “Many of them drop away when you give them a [side] project,” he said.
So when Tori Kinamon asked him to work on an MRSA project — in her first year — he gave her one that focused on researching vertebral osteomyelitis, a bone infection that can coincide with S aureus. What Dr. Fowler didn’t know: Kinamon (now MD) had been a competitive gymnast at Brown and battled her own life-threatening infection with MRSA.
“To my absolute astonishment, not only did she stick to it, but she was able to compile a presentation on the science and gave an oral presentation within a year of walking in the door,” said Dr. Fowler.
She went on to lead an initiative between the National Institutes of Health and US Food and Drug Administration to create endpoints for clinical drug trials, all of which occurred before starting her residency, which she’s about to embark upon.
Dr. Kinamon’s a good example, he said, of what happens when you add genuine passion to book smarts. Those who do always stand out because you can’t fake that. “Find your passion, and then chase it down relentlessly,” he said. “Once you’ve found your passion, things get easy because it stops being work and it starts being something else.”
If you haven’t identified a focus area, Dr. Fowler said to “be agnostic and observant. Keep your eyes open and your options open because you may surprise yourself. It may turn out that you end up liking something a whole lot more than you thought you did.”
Lesson #3: When You Say You’ve Always Wanted to Do Something, Do Something
As the chief of pulmonary and critical care medicine at the Northwestern Medicine Canning Thoracic Institute, Scott Budinger, MD, often hears lip service from doctors who want to put their skills to use in their local communities. One of his students actually did it.
Justin Fiala, MD, a pulmonary, critical care, and sleep specialist at Northwestern Medicine, joined Northwestern as a pulmonary fellow with a big interest in addressing health equity issues.
Dr. Fiala began volunteering with CommunityHealth during his fellowship and saw that many patients of the free Chicago-area clinic needed help with sleep disorders. He launched the organization’s first sleep clinic and its Patient-Centered Apnea Protocols Initiative.
“He developed a plan with some of the partners of the sleep apnea equipment to do home sleep testing for these patients that’s free of cost,” said Dr. Budinger.
Dr. Fiala goes in on Saturdays and runs a free clinic conducting sleep studies for patients and outfits them with devices that they need to improve their conditions, said Dr. Budinger.
“And these patients are the severest of the severe patients,” he said. “These are people that have severe sleep apnea that are driving around the roads, oftentimes don’t have insurance because they’re also precluded from having auto insurance. So, this is really something that not just benefits these patients but benefits our whole community.”
The fact that Dr. Fiala followed through on something that all doctors aspire to do — and in the middle of a very busy training program — is something that Dr. Budinger said makes him stand out in a big way.
“If you talk to any of our trainees or young faculty, everybody’s interested in addressing the issue of health disparities,” said Dr. Budinger. “Justin looked at that and said, ‘Well, you know, I’m not interested in talking about it. What can I do about this problem? And how can I actually get boots on the ground and help?’ That requires a big activation energy that many people don’t have.”
Lesson #4: Be a People-Person and a Patient-Person
When hiring employees at American Family Care in Portland, Oregon, Andrew Miller, MD, director of provider training, is always on the lookout for young MDs with emotional intelligence and a good bedside manner. He has been recently blown away, however, by a young physician’s assistant named Joseph Van Bindsbergen, PA-C, who was described as “all-around wonderful” during his reference check.
“Having less than 6 months of experience out of school, he is our highest ranked provider, whether it’s a nurse practitioner, PA, or doctor, in terms of patient satisfaction,” said Dr. Miller. The young PA has an “unprecedented perfect score” on his NPS rating.
Why? Patients said they’ve never felt as heard as they felt with Van Bindsbergen.
“That’s the thing I think that the up-and-coming providers should be focusing on is making your patients feel heard,” explained Dr. Miller. Van Bindsbergen is great at building rapport with a patient, whether they are 6 or 96. “He doesn’t just ask about sore throat symptoms. He asks, ‘what is the impact on your life of the sore throat? How does it affect your family or your work? What do you think this could be besides just strep? What are your concerns?’ ”
Dr. Miller said the magic of Van Bindsbergen is that he has an innate ability to look at patients “not just as a diagnosis but as a person, which they love.”
Lesson #5: Remember to Make That Difference With Each Patient
Doctors are used to swooping in and seeing a patient, ordering further testing if needed, and then moving on to the next patient. But one young intern at the start of his medical career broke this mold by giving a very anxious patient some much-needed support.
“There was a resident who was working overnight, and this poor young woman came in who had a new diagnosis of an advanced illness and a lot of anxiety around her condition, the newness of it, and the impact this is going to have on her family and her life,” said Elizabeth Horn Prsic, MD, assistant professor at Yale School of Medicine and firm chief for medical oncology and the director of Adult Inpatient Palliative Care.
Dr. Prsic found out the next morning that this trainee accompanied the patient to the MRI and held her hand as much as he was allowed to throughout the entire experience. “I was like, ‘wait you went down with her to radiology?’ And he’s like, ‘Yes, I was there the whole time,’ ” she recalled.
This gesture not only helped the patient feel calmer after receiving a potentially life-altering diagnosis but also helped ensure the test results were as clear as possible.
“If the study is not done well and a patient is moving or uncomfortable, it has to be stopped early or paused,” said Dr. Prsic. “Then the study is not very useful. In situations like these, medical decisions may be made based on imperfect data. The fact that we had this full complete good quality scan helped us get the care that she needed in a much timelier manner to help her and to move along the care that she that was medically appropriate for her.”
Dr. Prsic got emotional reflecting on the experience. Working at Yale, she saw a ton of intelligent doctors come through the ranks. But this gesture, she said, should serve as a reminder that “you don’t need to be the smartest person in the room to just be there for a patient. It was pure empathic presence and human connection. It gave me hope in the next generation of physicians.”
A version of this article appeared on Medscape.com.
Federal Trade Commission Bans Noncompete Agreements, Urges More Protections for Healthcare Workers
But business groups have vowed to challenge the decision in court.
The proposed final rule passed on a 3-2 vote, with the dissenting commissioners disputing the FTC’s authority to broadly ban noncompetes.
Tensions around noncompetes have been building for years. In 2021, President Biden issued an executive order supporting measures to improve economic competition, in which he urged the FTC to consider its rulemaking authority to address noncompete clauses that unfairly limit workers’ mobility. In January 2023, per that directive, the agency proposed ending the restrictive covenants.
While the FTC estimates that the final rule will reduce healthcare costs by up to $194 billion over the next decade and increase worker earnings by $300 million annually, the ruling faces legal hurdles.
US Chamber of Commerce president and CEO Suzanne P. Clark said in a statement that the move is a “blatant power grab” that will undermine competitive business practices, adding that the Chamber will sue to block the measure.
The FTC received more than 26,000 comments on noncompetes during the public feedback period, with about 25,000 supporting the measure, said Benjamin Cady, JD, an FTC attorney.
Mr. Cady called the feedback “compelling,” citing instances of workers who were forced to commute long distances, uproot their families, or risk expensive litigation for wanting to pursue job opportunities.
For example, a comment from a physician working in Appalachia highlights the potential real-life implications of the agreements. “With hospital systems merging, providers with aggressive noncompetes must abandon the community that they serve if they [choose] to leave their employer. Healthcare providers feel trapped in their current employment situation, leading to significant burnout that can shorten their [career] longevity.”
Commissioner Alvaro Bedoya said physicians have had their lives upended by cumbersome noncompetes, often having to move out of state to practice. “A pandemic killed a million people in this country, and there are doctors who cannot work because of a noncompete,” he said.
It’s unclear whether physicians and others who work for nonprofit healthcare groups or hospitals will be covered by the new ban. FTC Commissioner Rebecca Slaughter acknowledged that the agency’s jurisdictional limitations mean that employees of “certain nonprofit organizations” may not benefit from the rule.
“We want to be transparent about the limitation and recognize there are workers, especially healthcare workers, who are bound by anticompetitive and unfair noncompete clauses, that our rule will struggle to reach,” she said. To cover nonprofit healthcare employees, Ms. Slaughter urged Congress to pass legislation banning noncompetes, such as the Workforce Mobility Act of 2021 and the Freedom to Compete Act of 2023.
The FTC final rule will take effect 120 days after it is published in the federal register, and new noncompete agreements will be banned as of this date. However, existing contracts for senior executives will remain in effect because these individuals are less likely to experience “acute harm” due to their ability to negotiate accordingly, said Mr. Cady.
States, AMA Take Aim at Noncompetes
Before the federal ban, several states had already passed legislation limiting the reach of noncompetes. According to a recent article in the Journal of the American College of Cardiology, 12 states prohibit noncompete clauses for physicians: Alabama, California, Colorado, Delaware, Massachusetts, Montana, New Hampshire, New Mexico, North Dakota, Oklahoma, Rhode Island, and South Dakota.
The remaining states allow noncompetes in some form, often excluding them for employees earning below a certain threshold. For example, in Oregon, noncompete agreements may apply to employees earning more than $113,241. Most states have provisions to adjust the threshold annually. The District of Columbia permits 2-year noncompetes for “medical specialists” earning over $250,000 annually.
Indiana employers can no longer enter into noncompete agreements with primary care providers. Other specialties may be subject to the clauses, except when the physician terminates the contract for cause or when an employer terminates the contract without cause.
Rachel Marcus, MD, a cardiologist in Washington, DC, found out how limiting her employment contract’s noncompete clause was when she wanted to leave a former position. Due to the restrictions, she told this news organization that she couldn’t work locally for a competitor for 2 years. The closest location she could seek employment without violating the agreement was Baltimore, approximately 40 miles away.
Dr. Marcus ultimately moved to another position within the same organization because of the company’s reputation for being “aggressive” in their enforcement actions.
Although the American Medical Association (AMA) does not support a total ban, its House of Delegates adopted policies last year to support the prohibition of noncompete contracts for physicians employed by for-profit or nonprofit hospitals, hospital systems, or staffing companies.
Challenges Await
The American Hospital Association, which opposed the proposed rule, called it “bad policy.” The decision “will likely be short-lived, with courts almost certain to stop it before it can do damage to hospitals’ ability to care for their patients and communities,” the association said in a statement.
To ease the transition to the new rule, the FTC also released a model language for employers to use when discussing the changes with their employees. “All employers need to do to comply with the rule is to stop enforcing existing noncompetes with workers other than senior executives and provide notice to such workers,” he said.
Dr. Marcus hopes the ban improves doctors’ lives. “Your employer is going to have to treat you better because they know that you can easily go across town to a place that has a higher salary, and your patient can go with you.”
A version of this article appeared on Medscape.com.
But business groups have vowed to challenge the decision in court.
The proposed final rule passed on a 3-2 vote, with the dissenting commissioners disputing the FTC’s authority to broadly ban noncompetes.
Tensions around noncompetes have been building for years. In 2021, President Biden issued an executive order supporting measures to improve economic competition, in which he urged the FTC to consider its rulemaking authority to address noncompete clauses that unfairly limit workers’ mobility. In January 2023, per that directive, the agency proposed ending the restrictive covenants.
While the FTC estimates that the final rule will reduce healthcare costs by up to $194 billion over the next decade and increase worker earnings by $300 million annually, the ruling faces legal hurdles.
US Chamber of Commerce president and CEO Suzanne P. Clark said in a statement that the move is a “blatant power grab” that will undermine competitive business practices, adding that the Chamber will sue to block the measure.
The FTC received more than 26,000 comments on noncompetes during the public feedback period, with about 25,000 supporting the measure, said Benjamin Cady, JD, an FTC attorney.
Mr. Cady called the feedback “compelling,” citing instances of workers who were forced to commute long distances, uproot their families, or risk expensive litigation for wanting to pursue job opportunities.
For example, a comment from a physician working in Appalachia highlights the potential real-life implications of the agreements. “With hospital systems merging, providers with aggressive noncompetes must abandon the community that they serve if they [choose] to leave their employer. Healthcare providers feel trapped in their current employment situation, leading to significant burnout that can shorten their [career] longevity.”
Commissioner Alvaro Bedoya said physicians have had their lives upended by cumbersome noncompetes, often having to move out of state to practice. “A pandemic killed a million people in this country, and there are doctors who cannot work because of a noncompete,” he said.
It’s unclear whether physicians and others who work for nonprofit healthcare groups or hospitals will be covered by the new ban. FTC Commissioner Rebecca Slaughter acknowledged that the agency’s jurisdictional limitations mean that employees of “certain nonprofit organizations” may not benefit from the rule.
“We want to be transparent about the limitation and recognize there are workers, especially healthcare workers, who are bound by anticompetitive and unfair noncompete clauses, that our rule will struggle to reach,” she said. To cover nonprofit healthcare employees, Ms. Slaughter urged Congress to pass legislation banning noncompetes, such as the Workforce Mobility Act of 2021 and the Freedom to Compete Act of 2023.
The FTC final rule will take effect 120 days after it is published in the federal register, and new noncompete agreements will be banned as of this date. However, existing contracts for senior executives will remain in effect because these individuals are less likely to experience “acute harm” due to their ability to negotiate accordingly, said Mr. Cady.
States, AMA Take Aim at Noncompetes
Before the federal ban, several states had already passed legislation limiting the reach of noncompetes. According to a recent article in the Journal of the American College of Cardiology, 12 states prohibit noncompete clauses for physicians: Alabama, California, Colorado, Delaware, Massachusetts, Montana, New Hampshire, New Mexico, North Dakota, Oklahoma, Rhode Island, and South Dakota.
The remaining states allow noncompetes in some form, often excluding them for employees earning below a certain threshold. For example, in Oregon, noncompete agreements may apply to employees earning more than $113,241. Most states have provisions to adjust the threshold annually. The District of Columbia permits 2-year noncompetes for “medical specialists” earning over $250,000 annually.
Indiana employers can no longer enter into noncompete agreements with primary care providers. Other specialties may be subject to the clauses, except when the physician terminates the contract for cause or when an employer terminates the contract without cause.
Rachel Marcus, MD, a cardiologist in Washington, DC, found out how limiting her employment contract’s noncompete clause was when she wanted to leave a former position. Due to the restrictions, she told this news organization that she couldn’t work locally for a competitor for 2 years. The closest location she could seek employment without violating the agreement was Baltimore, approximately 40 miles away.
Dr. Marcus ultimately moved to another position within the same organization because of the company’s reputation for being “aggressive” in their enforcement actions.
Although the American Medical Association (AMA) does not support a total ban, its House of Delegates adopted policies last year to support the prohibition of noncompete contracts for physicians employed by for-profit or nonprofit hospitals, hospital systems, or staffing companies.
Challenges Await
The American Hospital Association, which opposed the proposed rule, called it “bad policy.” The decision “will likely be short-lived, with courts almost certain to stop it before it can do damage to hospitals’ ability to care for their patients and communities,” the association said in a statement.
To ease the transition to the new rule, the FTC also released a model language for employers to use when discussing the changes with their employees. “All employers need to do to comply with the rule is to stop enforcing existing noncompetes with workers other than senior executives and provide notice to such workers,” he said.
Dr. Marcus hopes the ban improves doctors’ lives. “Your employer is going to have to treat you better because they know that you can easily go across town to a place that has a higher salary, and your patient can go with you.”
A version of this article appeared on Medscape.com.
But business groups have vowed to challenge the decision in court.
The proposed final rule passed on a 3-2 vote, with the dissenting commissioners disputing the FTC’s authority to broadly ban noncompetes.
Tensions around noncompetes have been building for years. In 2021, President Biden issued an executive order supporting measures to improve economic competition, in which he urged the FTC to consider its rulemaking authority to address noncompete clauses that unfairly limit workers’ mobility. In January 2023, per that directive, the agency proposed ending the restrictive covenants.
While the FTC estimates that the final rule will reduce healthcare costs by up to $194 billion over the next decade and increase worker earnings by $300 million annually, the ruling faces legal hurdles.
US Chamber of Commerce president and CEO Suzanne P. Clark said in a statement that the move is a “blatant power grab” that will undermine competitive business practices, adding that the Chamber will sue to block the measure.
The FTC received more than 26,000 comments on noncompetes during the public feedback period, with about 25,000 supporting the measure, said Benjamin Cady, JD, an FTC attorney.
Mr. Cady called the feedback “compelling,” citing instances of workers who were forced to commute long distances, uproot their families, or risk expensive litigation for wanting to pursue job opportunities.
For example, a comment from a physician working in Appalachia highlights the potential real-life implications of the agreements. “With hospital systems merging, providers with aggressive noncompetes must abandon the community that they serve if they [choose] to leave their employer. Healthcare providers feel trapped in their current employment situation, leading to significant burnout that can shorten their [career] longevity.”
Commissioner Alvaro Bedoya said physicians have had their lives upended by cumbersome noncompetes, often having to move out of state to practice. “A pandemic killed a million people in this country, and there are doctors who cannot work because of a noncompete,” he said.
It’s unclear whether physicians and others who work for nonprofit healthcare groups or hospitals will be covered by the new ban. FTC Commissioner Rebecca Slaughter acknowledged that the agency’s jurisdictional limitations mean that employees of “certain nonprofit organizations” may not benefit from the rule.
“We want to be transparent about the limitation and recognize there are workers, especially healthcare workers, who are bound by anticompetitive and unfair noncompete clauses, that our rule will struggle to reach,” she said. To cover nonprofit healthcare employees, Ms. Slaughter urged Congress to pass legislation banning noncompetes, such as the Workforce Mobility Act of 2021 and the Freedom to Compete Act of 2023.
The FTC final rule will take effect 120 days after it is published in the federal register, and new noncompete agreements will be banned as of this date. However, existing contracts for senior executives will remain in effect because these individuals are less likely to experience “acute harm” due to their ability to negotiate accordingly, said Mr. Cady.
States, AMA Take Aim at Noncompetes
Before the federal ban, several states had already passed legislation limiting the reach of noncompetes. According to a recent article in the Journal of the American College of Cardiology, 12 states prohibit noncompete clauses for physicians: Alabama, California, Colorado, Delaware, Massachusetts, Montana, New Hampshire, New Mexico, North Dakota, Oklahoma, Rhode Island, and South Dakota.
The remaining states allow noncompetes in some form, often excluding them for employees earning below a certain threshold. For example, in Oregon, noncompete agreements may apply to employees earning more than $113,241. Most states have provisions to adjust the threshold annually. The District of Columbia permits 2-year noncompetes for “medical specialists” earning over $250,000 annually.
Indiana employers can no longer enter into noncompete agreements with primary care providers. Other specialties may be subject to the clauses, except when the physician terminates the contract for cause or when an employer terminates the contract without cause.
Rachel Marcus, MD, a cardiologist in Washington, DC, found out how limiting her employment contract’s noncompete clause was when she wanted to leave a former position. Due to the restrictions, she told this news organization that she couldn’t work locally for a competitor for 2 years. The closest location she could seek employment without violating the agreement was Baltimore, approximately 40 miles away.
Dr. Marcus ultimately moved to another position within the same organization because of the company’s reputation for being “aggressive” in their enforcement actions.
Although the American Medical Association (AMA) does not support a total ban, its House of Delegates adopted policies last year to support the prohibition of noncompete contracts for physicians employed by for-profit or nonprofit hospitals, hospital systems, or staffing companies.
Challenges Await
The American Hospital Association, which opposed the proposed rule, called it “bad policy.” The decision “will likely be short-lived, with courts almost certain to stop it before it can do damage to hospitals’ ability to care for their patients and communities,” the association said in a statement.
To ease the transition to the new rule, the FTC also released a model language for employers to use when discussing the changes with their employees. “All employers need to do to comply with the rule is to stop enforcing existing noncompetes with workers other than senior executives and provide notice to such workers,” he said.
Dr. Marcus hopes the ban improves doctors’ lives. “Your employer is going to have to treat you better because they know that you can easily go across town to a place that has a higher salary, and your patient can go with you.”
A version of this article appeared on Medscape.com.
Are Women Better Doctors Than Men?
This transcript has been edited for clarity.
It’s a battle of the sexes today as we dive into a paper that makes you say, “Wow, what an interesting study” and also “Boy, am I glad I didn’t do that study.” That’s because studies like this are always somewhat fraught; they say something about medicine but also something about society — and that makes this a bit precarious. But that’s never stopped us before. So, let’s go ahead and try to answer the question: Do women make better doctors than men?
On the surface, this question seems nearly impossible to answer. It’s too broad; what does it mean to be a “better” doctor? At first blush it seems that there are just too many variables to control for here: the type of doctor, the type of patient, the clinical scenario, and so on.
But this study, “Comparison of hospital mortality and readmission rates by physician and patient sex,” which appears in Annals of Internal Medicine, uses a fairly ingenious method to cut through all the bias by leveraging two simple facts: First, hospital medicine is largely conducted by hospitalists these days; second, due to the shift-based nature of hospitalist work, the hospitalist you get when you are admitted to the hospital is pretty much random.
In other words, if you are admitted to the hospital for an acute illness and get a hospitalist as your attending, you have no control over whether it is a man or a woman. Is this a randomized trial? No, but it’s not bad.
Researchers used Medicare claims data to identify adults over age 65 who had nonelective hospital admissions throughout the United States. The claims revealed the sex of the patient and the name of the attending physician. By linking to a medical provider database, they could determine the sex of the provider.
The goal was to look at outcomes across four dyads:
- Male patient – male doctor
- Male patient – female doctor
- Female patient – male doctor
- Female patient – female doctor
The primary outcome was 30-day mortality.
I told you that focusing on hospitalists produces some pseudorandomization, but let’s look at the data to be sure. Just under a million patients were treated by approximately 50,000 physicians, 30% of whom were female. And, though female patients and male patients differed, they did not differ with respect to the sex of their hospitalist. So, by physician sex, patients were similar in mean age, race, ethnicity, household income, eligibility for Medicaid, and comorbid conditions. The authors even created a “predicted mortality” score which was similar across the groups as well.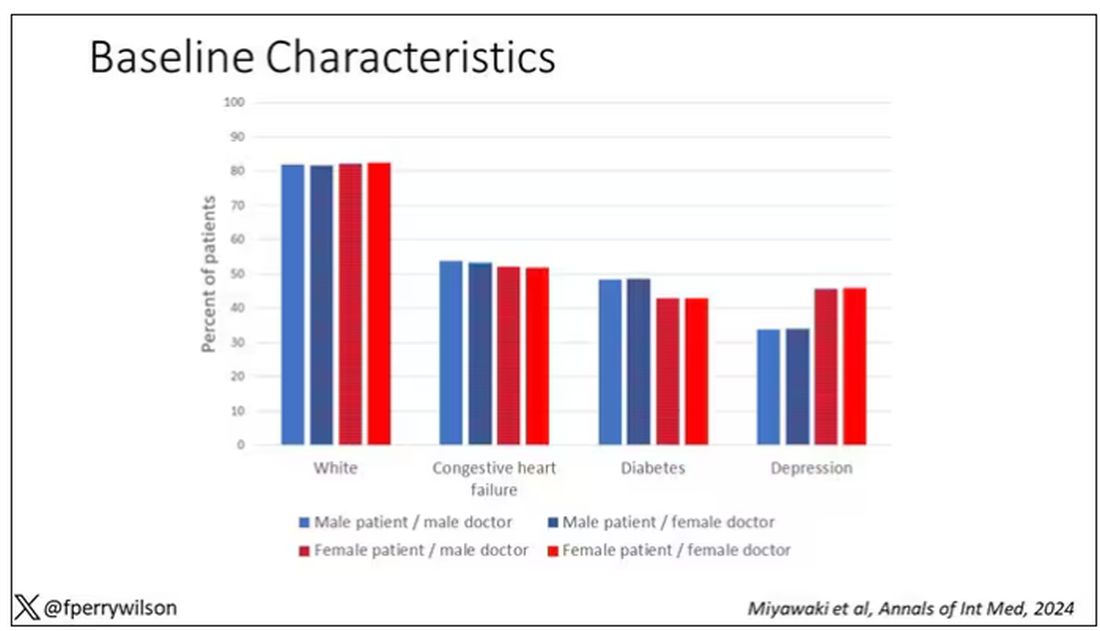
Now, the female physicians were a bit different from the male physicians. The female hospitalists were slightly more likely to have an osteopathic degree, had slightly fewer admissions per year, and were a bit younger.
So, we have broadly similar patients regardless of who their hospitalist was, but hospitalists differ by factors other than their sex. Fine.
I’ve graphed the results here. 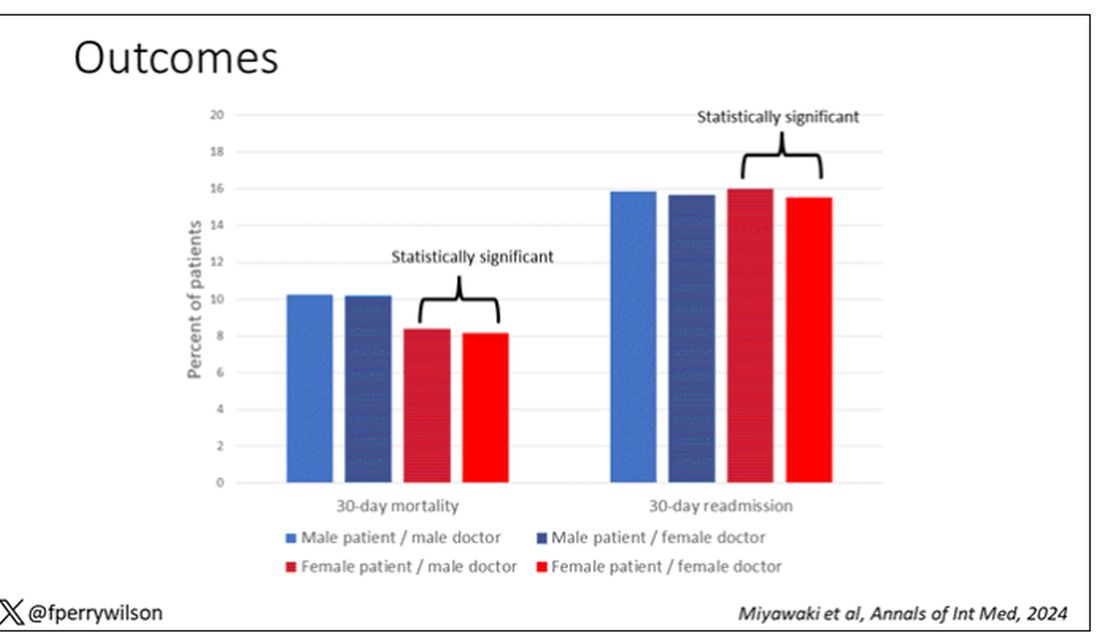
This is a relatively small effect, to be sure, but if you multiply it across the millions of hospitalist admissions per year, you can start to put up some real numbers.
So, what is going on here? I see four broad buckets of possibilities.
Let’s start with the obvious explanation: Women, on average, are better doctors than men. I am married to a woman doctor, and based on my personal experience, this explanation is undoubtedly true. But why would that be?
The authors cite data that suggest that female physicians are less likely than male physicians to dismiss patient concerns — and in particular, the concerns of female patients — perhaps leading to fewer missed diagnoses. But this is impossible to measure with administrative data, so this study can no more tell us whether these female hospitalists are more attentive than their male counterparts than it can suggest that the benefit is mediated by the shorter average height of female physicians. Perhaps the key is being closer to the patient?
The second possibility here is that this has nothing to do with the sex of the physician at all; it has to do with those other things that associate with the sex of the physician. We know, for example, that the female physicians saw fewer patients per year than the male physicians, but the study authors adjusted for this in the statistical models. Still, other unmeasured factors (confounders) could be present. By the way, confounders wouldn’t necessarily change the primary finding — you are better off being cared for by female physicians. It’s just not because they are female; it’s a convenient marker for some other quality, such as age.
The third possibility is that the study represents a phenomenon called collider bias. The idea here is that physicians only get into the study if they are hospitalists, and the quality of physicians who choose to become a hospitalist may differ by sex. When deciding on a specialty, a talented resident considering certain lifestyle issues may find hospital medicine particularly attractive — and that draw toward a more lifestyle-friendly specialty may differ by sex, as some prior studies have shown. If true, the pool of women hospitalists may be better than their male counterparts because male physicians of that caliber don’t become hospitalists.
Okay, don’t write in. I’m just trying to cite examples of how to think about collider bias. I can’t prove that this is the case, and in fact the authors do a sensitivity analysis of all physicians, not just hospitalists, and show the same thing. So this is probably not true, but epidemiology is fun, right?
And the fourth possibility: This is nothing but statistical noise. The effect size is incredibly small and just on the border of statistical significance. Especially when you’re working with very large datasets like this, you’ve got to be really careful about overinterpreting statistically significant findings that are nevertheless of small magnitude.
Regardless, it’s an interesting study, one that made me think and, of course, worry a bit about how I would present it. Forgive me if I’ve been indelicate in handling the complex issues of sex, gender, and society here. But I’m not sure what you expect; after all, I’m only a male doctor.
Dr. Wilson is associate professor of medicine and public health and director of the Clinical and Translational Research Accelerator at Yale University, New Haven, Conn. He has disclosed no relevant financial relationships.
A version of this article appeared on Medscape.com.
This transcript has been edited for clarity.
It’s a battle of the sexes today as we dive into a paper that makes you say, “Wow, what an interesting study” and also “Boy, am I glad I didn’t do that study.” That’s because studies like this are always somewhat fraught; they say something about medicine but also something about society — and that makes this a bit precarious. But that’s never stopped us before. So, let’s go ahead and try to answer the question: Do women make better doctors than men?
On the surface, this question seems nearly impossible to answer. It’s too broad; what does it mean to be a “better” doctor? At first blush it seems that there are just too many variables to control for here: the type of doctor, the type of patient, the clinical scenario, and so on.
But this study, “Comparison of hospital mortality and readmission rates by physician and patient sex,” which appears in Annals of Internal Medicine, uses a fairly ingenious method to cut through all the bias by leveraging two simple facts: First, hospital medicine is largely conducted by hospitalists these days; second, due to the shift-based nature of hospitalist work, the hospitalist you get when you are admitted to the hospital is pretty much random.
In other words, if you are admitted to the hospital for an acute illness and get a hospitalist as your attending, you have no control over whether it is a man or a woman. Is this a randomized trial? No, but it’s not bad.
Researchers used Medicare claims data to identify adults over age 65 who had nonelective hospital admissions throughout the United States. The claims revealed the sex of the patient and the name of the attending physician. By linking to a medical provider database, they could determine the sex of the provider.
The goal was to look at outcomes across four dyads:
- Male patient – male doctor
- Male patient – female doctor
- Female patient – male doctor
- Female patient – female doctor
The primary outcome was 30-day mortality.
I told you that focusing on hospitalists produces some pseudorandomization, but let’s look at the data to be sure. Just under a million patients were treated by approximately 50,000 physicians, 30% of whom were female. And, though female patients and male patients differed, they did not differ with respect to the sex of their hospitalist. So, by physician sex, patients were similar in mean age, race, ethnicity, household income, eligibility for Medicaid, and comorbid conditions. The authors even created a “predicted mortality” score which was similar across the groups as well.
Now, the female physicians were a bit different from the male physicians. The female hospitalists were slightly more likely to have an osteopathic degree, had slightly fewer admissions per year, and were a bit younger.
So, we have broadly similar patients regardless of who their hospitalist was, but hospitalists differ by factors other than their sex. Fine.
I’ve graphed the results here. 
This is a relatively small effect, to be sure, but if you multiply it across the millions of hospitalist admissions per year, you can start to put up some real numbers.
So, what is going on here? I see four broad buckets of possibilities.
Let’s start with the obvious explanation: Women, on average, are better doctors than men. I am married to a woman doctor, and based on my personal experience, this explanation is undoubtedly true. But why would that be?
The authors cite data that suggest that female physicians are less likely than male physicians to dismiss patient concerns — and in particular, the concerns of female patients — perhaps leading to fewer missed diagnoses. But this is impossible to measure with administrative data, so this study can no more tell us whether these female hospitalists are more attentive than their male counterparts than it can suggest that the benefit is mediated by the shorter average height of female physicians. Perhaps the key is being closer to the patient?
The second possibility here is that this has nothing to do with the sex of the physician at all; it has to do with those other things that associate with the sex of the physician. We know, for example, that the female physicians saw fewer patients per year than the male physicians, but the study authors adjusted for this in the statistical models. Still, other unmeasured factors (confounders) could be present. By the way, confounders wouldn’t necessarily change the primary finding — you are better off being cared for by female physicians. It’s just not because they are female; it’s a convenient marker for some other quality, such as age.
The third possibility is that the study represents a phenomenon called collider bias. The idea here is that physicians only get into the study if they are hospitalists, and the quality of physicians who choose to become a hospitalist may differ by sex. When deciding on a specialty, a talented resident considering certain lifestyle issues may find hospital medicine particularly attractive — and that draw toward a more lifestyle-friendly specialty may differ by sex, as some prior studies have shown. If true, the pool of women hospitalists may be better than their male counterparts because male physicians of that caliber don’t become hospitalists.
Okay, don’t write in. I’m just trying to cite examples of how to think about collider bias. I can’t prove that this is the case, and in fact the authors do a sensitivity analysis of all physicians, not just hospitalists, and show the same thing. So this is probably not true, but epidemiology is fun, right?
And the fourth possibility: This is nothing but statistical noise. The effect size is incredibly small and just on the border of statistical significance. Especially when you’re working with very large datasets like this, you’ve got to be really careful about overinterpreting statistically significant findings that are nevertheless of small magnitude.
Regardless, it’s an interesting study, one that made me think and, of course, worry a bit about how I would present it. Forgive me if I’ve been indelicate in handling the complex issues of sex, gender, and society here. But I’m not sure what you expect; after all, I’m only a male doctor.
Dr. Wilson is associate professor of medicine and public health and director of the Clinical and Translational Research Accelerator at Yale University, New Haven, Conn. He has disclosed no relevant financial relationships.
A version of this article appeared on Medscape.com.
This transcript has been edited for clarity.
It’s a battle of the sexes today as we dive into a paper that makes you say, “Wow, what an interesting study” and also “Boy, am I glad I didn’t do that study.” That’s because studies like this are always somewhat fraught; they say something about medicine but also something about society — and that makes this a bit precarious. But that’s never stopped us before. So, let’s go ahead and try to answer the question: Do women make better doctors than men?
On the surface, this question seems nearly impossible to answer. It’s too broad; what does it mean to be a “better” doctor? At first blush it seems that there are just too many variables to control for here: the type of doctor, the type of patient, the clinical scenario, and so on.
But this study, “Comparison of hospital mortality and readmission rates by physician and patient sex,” which appears in Annals of Internal Medicine, uses a fairly ingenious method to cut through all the bias by leveraging two simple facts: First, hospital medicine is largely conducted by hospitalists these days; second, due to the shift-based nature of hospitalist work, the hospitalist you get when you are admitted to the hospital is pretty much random.
In other words, if you are admitted to the hospital for an acute illness and get a hospitalist as your attending, you have no control over whether it is a man or a woman. Is this a randomized trial? No, but it’s not bad.
Researchers used Medicare claims data to identify adults over age 65 who had nonelective hospital admissions throughout the United States. The claims revealed the sex of the patient and the name of the attending physician. By linking to a medical provider database, they could determine the sex of the provider.
The goal was to look at outcomes across four dyads:
- Male patient – male doctor
- Male patient – female doctor
- Female patient – male doctor
- Female patient – female doctor
The primary outcome was 30-day mortality.
I told you that focusing on hospitalists produces some pseudorandomization, but let’s look at the data to be sure. Just under a million patients were treated by approximately 50,000 physicians, 30% of whom were female. And, though female patients and male patients differed, they did not differ with respect to the sex of their hospitalist. So, by physician sex, patients were similar in mean age, race, ethnicity, household income, eligibility for Medicaid, and comorbid conditions. The authors even created a “predicted mortality” score which was similar across the groups as well.
Now, the female physicians were a bit different from the male physicians. The female hospitalists were slightly more likely to have an osteopathic degree, had slightly fewer admissions per year, and were a bit younger.
So, we have broadly similar patients regardless of who their hospitalist was, but hospitalists differ by factors other than their sex. Fine.
I’ve graphed the results here. 
This is a relatively small effect, to be sure, but if you multiply it across the millions of hospitalist admissions per year, you can start to put up some real numbers.
So, what is going on here? I see four broad buckets of possibilities.
Let’s start with the obvious explanation: Women, on average, are better doctors than men. I am married to a woman doctor, and based on my personal experience, this explanation is undoubtedly true. But why would that be?
The authors cite data that suggest that female physicians are less likely than male physicians to dismiss patient concerns — and in particular, the concerns of female patients — perhaps leading to fewer missed diagnoses. But this is impossible to measure with administrative data, so this study can no more tell us whether these female hospitalists are more attentive than their male counterparts than it can suggest that the benefit is mediated by the shorter average height of female physicians. Perhaps the key is being closer to the patient?
The second possibility here is that this has nothing to do with the sex of the physician at all; it has to do with those other things that associate with the sex of the physician. We know, for example, that the female physicians saw fewer patients per year than the male physicians, but the study authors adjusted for this in the statistical models. Still, other unmeasured factors (confounders) could be present. By the way, confounders wouldn’t necessarily change the primary finding — you are better off being cared for by female physicians. It’s just not because they are female; it’s a convenient marker for some other quality, such as age.
The third possibility is that the study represents a phenomenon called collider bias. The idea here is that physicians only get into the study if they are hospitalists, and the quality of physicians who choose to become a hospitalist may differ by sex. When deciding on a specialty, a talented resident considering certain lifestyle issues may find hospital medicine particularly attractive — and that draw toward a more lifestyle-friendly specialty may differ by sex, as some prior studies have shown. If true, the pool of women hospitalists may be better than their male counterparts because male physicians of that caliber don’t become hospitalists.
Okay, don’t write in. I’m just trying to cite examples of how to think about collider bias. I can’t prove that this is the case, and in fact the authors do a sensitivity analysis of all physicians, not just hospitalists, and show the same thing. So this is probably not true, but epidemiology is fun, right?
And the fourth possibility: This is nothing but statistical noise. The effect size is incredibly small and just on the border of statistical significance. Especially when you’re working with very large datasets like this, you’ve got to be really careful about overinterpreting statistically significant findings that are nevertheless of small magnitude.
Regardless, it’s an interesting study, one that made me think and, of course, worry a bit about how I would present it. Forgive me if I’ve been indelicate in handling the complex issues of sex, gender, and society here. But I’m not sure what you expect; after all, I’m only a male doctor.
Dr. Wilson is associate professor of medicine and public health and director of the Clinical and Translational Research Accelerator at Yale University, New Haven, Conn. He has disclosed no relevant financial relationships.
A version of this article appeared on Medscape.com.
How to Play Like a Masters Champ
You know what the happiest animal in the world is? A goldfish. You know why? It’s got a 10-second memory. Be a goldfish. — Ted Lasso
I don’t play much golf. When I do, it’s when my dad is in town. He shoots his age (78). I shoot double mine (52). He was recently here. We played and watched the Masters where he pointed out how I looked a lot like Scottie Scheffler, the now two-time Masters champion. On the 10th hole of his third round, you could see the resemblance. Scheffler’s third shot flew past the hole into the galley. He rifled the fourth past the hole on its way back toward the fairway. It was now a good distance further from the cup than a minute ago. He proceeded to misread his bogey putt, ending his misery with a double bogey. Scheffler went on to bogey the next hole and dropped from first on the leaderboard to fifth. Yes, I looked just like that on my last round. But here is where Scheffler and I differ. After a hole like that, I’d have been apoplectic, seething with self loathing. Scheffler was not. He kept moving. Head up, he sauntered to the next hole as if he had no awareness of what just transpired.
The ability to compartmentalize is useful not only to become the Masters champion, but also to become master of your day. In this way, golf is a nice approximation for life. The best golfers in the world will always have horrible shots and dreadful holes. The winning ones are often those who recover rather than continue in a downward spiral of one bad shot after another.
It’s easy to think of regular days that went just like Scheffler’s atrocious 10th hole. Getting pimped in front of distinguished faculty at Grand Rounds and whiffing (it was Sweet Syndrome). Calling a patient to let him know that his syphilis test did in fact come back positive (it was his father on the phone, also Mr. Rodham). Arguing with a patient that a biopsy was not needed for me to diagnose her with zoster (you’ve lost once, you’ve lost your temper). Each of these made me feel like slamming my club down, quitting the round right then and there. Losing control though, leads to flubbing the next question or arguing with the following patient. The masters let it go. Like goldfish, they live in the present without any thought of what happened 10 seconds ago.
We don’t have to take advice just from Ted Lasso here; there is plenty of research to support this concept of the critical relationship between resilience and psychological flexibility. Specifically, flexible cognitive control allows us to guide attention and to choose appropriate appraisal and good coping strategies. Ultimately, this leads to better performance. You might be a skilled athlete or presenter, but if you can’t regulate your emotions and something goes wrong, then you’ll perform as poorly as an amateur.
Scheffler went on to eagle the 13th hole on that round. He eventually won the 2024 Masters Tournament. Remember that the next time you find yourself in a day that feels like it is spiraling toward disaster. Close the door on the compartment that was the last miserable hole and saunter to the next patient like it never happened.
And maybe close the clubface a bit on address for your next drive.
Dr. Benabio is director of Healthcare Transformation and chief of dermatology at Kaiser Permanente San Diego. The opinions expressed in this column are his own and do not represent those of Kaiser Permanente. Dr. Benabio is @Dermdoc on X. Write to him at [email protected].
You know what the happiest animal in the world is? A goldfish. You know why? It’s got a 10-second memory. Be a goldfish. — Ted Lasso
I don’t play much golf. When I do, it’s when my dad is in town. He shoots his age (78). I shoot double mine (52). He was recently here. We played and watched the Masters where he pointed out how I looked a lot like Scottie Scheffler, the now two-time Masters champion. On the 10th hole of his third round, you could see the resemblance. Scheffler’s third shot flew past the hole into the galley. He rifled the fourth past the hole on its way back toward the fairway. It was now a good distance further from the cup than a minute ago. He proceeded to misread his bogey putt, ending his misery with a double bogey. Scheffler went on to bogey the next hole and dropped from first on the leaderboard to fifth. Yes, I looked just like that on my last round. But here is where Scheffler and I differ. After a hole like that, I’d have been apoplectic, seething with self loathing. Scheffler was not. He kept moving. Head up, he sauntered to the next hole as if he had no awareness of what just transpired.
The ability to compartmentalize is useful not only to become the Masters champion, but also to become master of your day. In this way, golf is a nice approximation for life. The best golfers in the world will always have horrible shots and dreadful holes. The winning ones are often those who recover rather than continue in a downward spiral of one bad shot after another.
It’s easy to think of regular days that went just like Scheffler’s atrocious 10th hole. Getting pimped in front of distinguished faculty at Grand Rounds and whiffing (it was Sweet Syndrome). Calling a patient to let him know that his syphilis test did in fact come back positive (it was his father on the phone, also Mr. Rodham). Arguing with a patient that a biopsy was not needed for me to diagnose her with zoster (you’ve lost once, you’ve lost your temper). Each of these made me feel like slamming my club down, quitting the round right then and there. Losing control though, leads to flubbing the next question or arguing with the following patient. The masters let it go. Like goldfish, they live in the present without any thought of what happened 10 seconds ago.
We don’t have to take advice just from Ted Lasso here; there is plenty of research to support this concept of the critical relationship between resilience and psychological flexibility. Specifically, flexible cognitive control allows us to guide attention and to choose appropriate appraisal and good coping strategies. Ultimately, this leads to better performance. You might be a skilled athlete or presenter, but if you can’t regulate your emotions and something goes wrong, then you’ll perform as poorly as an amateur.
Scheffler went on to eagle the 13th hole on that round. He eventually won the 2024 Masters Tournament. Remember that the next time you find yourself in a day that feels like it is spiraling toward disaster. Close the door on the compartment that was the last miserable hole and saunter to the next patient like it never happened.
And maybe close the clubface a bit on address for your next drive.
Dr. Benabio is director of Healthcare Transformation and chief of dermatology at Kaiser Permanente San Diego. The opinions expressed in this column are his own and do not represent those of Kaiser Permanente. Dr. Benabio is @Dermdoc on X. Write to him at [email protected].
You know what the happiest animal in the world is? A goldfish. You know why? It’s got a 10-second memory. Be a goldfish. — Ted Lasso
I don’t play much golf. When I do, it’s when my dad is in town. He shoots his age (78). I shoot double mine (52). He was recently here. We played and watched the Masters where he pointed out how I looked a lot like Scottie Scheffler, the now two-time Masters champion. On the 10th hole of his third round, you could see the resemblance. Scheffler’s third shot flew past the hole into the galley. He rifled the fourth past the hole on its way back toward the fairway. It was now a good distance further from the cup than a minute ago. He proceeded to misread his bogey putt, ending his misery with a double bogey. Scheffler went on to bogey the next hole and dropped from first on the leaderboard to fifth. Yes, I looked just like that on my last round. But here is where Scheffler and I differ. After a hole like that, I’d have been apoplectic, seething with self loathing. Scheffler was not. He kept moving. Head up, he sauntered to the next hole as if he had no awareness of what just transpired.
The ability to compartmentalize is useful not only to become the Masters champion, but also to become master of your day. In this way, golf is a nice approximation for life. The best golfers in the world will always have horrible shots and dreadful holes. The winning ones are often those who recover rather than continue in a downward spiral of one bad shot after another.
It’s easy to think of regular days that went just like Scheffler’s atrocious 10th hole. Getting pimped in front of distinguished faculty at Grand Rounds and whiffing (it was Sweet Syndrome). Calling a patient to let him know that his syphilis test did in fact come back positive (it was his father on the phone, also Mr. Rodham). Arguing with a patient that a biopsy was not needed for me to diagnose her with zoster (you’ve lost once, you’ve lost your temper). Each of these made me feel like slamming my club down, quitting the round right then and there. Losing control though, leads to flubbing the next question or arguing with the following patient. The masters let it go. Like goldfish, they live in the present without any thought of what happened 10 seconds ago.
We don’t have to take advice just from Ted Lasso here; there is plenty of research to support this concept of the critical relationship between resilience and psychological flexibility. Specifically, flexible cognitive control allows us to guide attention and to choose appropriate appraisal and good coping strategies. Ultimately, this leads to better performance. You might be a skilled athlete or presenter, but if you can’t regulate your emotions and something goes wrong, then you’ll perform as poorly as an amateur.
Scheffler went on to eagle the 13th hole on that round. He eventually won the 2024 Masters Tournament. Remember that the next time you find yourself in a day that feels like it is spiraling toward disaster. Close the door on the compartment that was the last miserable hole and saunter to the next patient like it never happened.
And maybe close the clubface a bit on address for your next drive.
Dr. Benabio is director of Healthcare Transformation and chief of dermatology at Kaiser Permanente San Diego. The opinions expressed in this column are his own and do not represent those of Kaiser Permanente. Dr. Benabio is @Dermdoc on X. Write to him at [email protected].
Weighing the Benefits of Integrating AI-based Clinical Notes Into Your Practice
Picture a healthcare system where physicians aren’t bogged down by excessive charting but are instead fully present with their patients, offering undivided attention and personalized care. In a recent X post, Stuart Blitz, COO and co-founder of Hone Health, sparked a thought-provoking conversation. “The problem with US healthcare is physicians are burned out since they spend way too much time charting, not enough with patients,” he wrote. “If you created a health system that did zero charting, you’d attract the best physicians and all patients would go there. Who is working on this?”
This resonates with many in the medical community, myself included, because the strain of extensive documentation detracts from patient care. Having worked in both large and small healthcare systems, I know the burden of extensive charting is a palpable challenge, often detracting from the time we can devote to our patients.
The first part of this two-part series examines the overarching benefits of artificial intelligence (AI)–based clinical documentation in modern healthcare, a field witnessing a paradigm shift thanks to advancements in AI.
Transformative Evolution of Clinical Documentation
The transition from manual documentation to AI-driven solutions marks a significant shift in the field, with a number of products in development including Nuance, Abridge, Ambience, ScribeAmerica, 3M, and DeepScribe. These tools use ambient clinical intelligence (ACI) to automate documentation, capturing patient conversations and translating them into structured clinical summaries. This innovation aligns with the vision of reducing charting burdens and enhancing patient-physician interactions.
How does it work? ACI refers to a sophisticated form of AI applied in healthcare settings, particularly focusing on enhancing the clinical documentation process without disrupting the natural flow of the consultation. Here’s a technical yet practical breakdown of ACI and the algorithms it typically employs:
Data capture and processing: ACI systems employ various sensors and processing units, typically integrated into clinical settings. These sensors, like microphones and cameras, gather diverse data such as audio from patient-doctor dialogues and visual cues. This information is then processed in real-time or near–real-time.
Natural language processing (NLP): A core component of ACI is advanced NLP algorithms. These algorithms analyze the captured audio data, transcribing spoken words into text. NLP goes beyond mere transcription; it involves understanding context, extracting relevant medical information (like symptoms, diagnoses, and treatment plans), and interpreting the nuances of human language.
Deep learning: Machine learning, particularly deep-learning techniques, are employed to improve the accuracy of ACI systems continually. These algorithms can learn from vast datasets of clinical interactions, enhancing their ability to transcribe and interpret future conversations accurately. As they learn, they become better at understanding different accents, complex medical terms, and variations in speech patterns.
Integration with electronic health records (EHRs): ACI systems are often designed to integrate seamlessly with existing EHR systems. They can automatically populate patient records with information from patient-clinician interactions, reducing manual entry and potential errors.
Customization and personalization: Many ACI systems offer customizable templates or allow clinicians to tailor documentation workflows. This flexibility ensures that the output aligns with the specific needs and preferences of healthcare providers.
Ethical and privacy considerations: ACI systems must navigate significant ethical and privacy concerns, especially related to patient consent and data security. These systems need to comply with healthcare privacy regulations such as HIPAA. They need to securely manage sensitive patient data and restrict access to authorized personnel only.
Broad-Spectrum Benefits of AI in Documentation
- Reducing clinician burnout: By automating the documentation process, AI tools like DAX Copilot alleviate a significant contributor to physician burnout, enabling clinicians to focus more on patient care.
- Enhanced patient care: With AI handling documentation, clinicians can engage more with their patients, leading to improved care quality and patient satisfaction.
- Data accuracy and quality: AI-driven documentation captures detailed patient encounters accurately, ensuring high-quality and comprehensive medical records.
- Response to the growing need for efficient healthcare: AI-based documentation is a direct response to the growing call for more efficient healthcare practices, where clinicians spend less time on paperwork and more with patients.
The shift toward AI-based clinical documentation represents a critical step in addressing the inefficiencies in healthcare systems. It’s a move towards a more patient-centered approach, where clinicians can focus more on patient care by reducing the time spent on excessive charting. Hopefully, we can integrate these solutions into our clinics at a large enough scale to make such an impact.
In the next column, we will explore in-depth insights from Kenneth Harper at Nuance on the technical implementation of these tools, with DAX as an example.
I would love to read your comments on AI in clinical trials as well as other AI-related topics. Write me at [email protected] or find me on X @DrBonillaOnc.
Dr. Loaiza-Bonilla is the co-founder and chief medical officer at Massive Bio, a company connecting patients to clinical trials using artificial intelligence. His research and professional interests focus on precision medicine, clinical trial design, digital health, entrepreneurship, and patient advocacy. Dr Loaiza-Bonilla serves as medical director of oncology research at Capital Health in New Jersey, where he maintains a connection to patient care by attending to patients 2 days a week. He has served as a consultant for Verify, PSI CRO, Bayer, AstraZeneca, Cardinal Health, BrightInsight, The Lynx Group, Fresenius, Pfizer, Ipsen, and Guardant; served as a speaker or a member of a speakers bureau for Amgen, Guardant, Eisai, Ipsen, Natera, Merck, Bristol-Myers Squibb, and AstraZeneca. He holds a 5% or greater equity interest in Massive Bio.
A version of this article appeared on Medscape.com.
Picture a healthcare system where physicians aren’t bogged down by excessive charting but are instead fully present with their patients, offering undivided attention and personalized care. In a recent X post, Stuart Blitz, COO and co-founder of Hone Health, sparked a thought-provoking conversation. “The problem with US healthcare is physicians are burned out since they spend way too much time charting, not enough with patients,” he wrote. “If you created a health system that did zero charting, you’d attract the best physicians and all patients would go there. Who is working on this?”
This resonates with many in the medical community, myself included, because the strain of extensive documentation detracts from patient care. Having worked in both large and small healthcare systems, I know the burden of extensive charting is a palpable challenge, often detracting from the time we can devote to our patients.
The first part of this two-part series examines the overarching benefits of artificial intelligence (AI)–based clinical documentation in modern healthcare, a field witnessing a paradigm shift thanks to advancements in AI.
Transformative Evolution of Clinical Documentation
The transition from manual documentation to AI-driven solutions marks a significant shift in the field, with a number of products in development including Nuance, Abridge, Ambience, ScribeAmerica, 3M, and DeepScribe. These tools use ambient clinical intelligence (ACI) to automate documentation, capturing patient conversations and translating them into structured clinical summaries. This innovation aligns with the vision of reducing charting burdens and enhancing patient-physician interactions.
How does it work? ACI refers to a sophisticated form of AI applied in healthcare settings, particularly focusing on enhancing the clinical documentation process without disrupting the natural flow of the consultation. Here’s a technical yet practical breakdown of ACI and the algorithms it typically employs:
Data capture and processing: ACI systems employ various sensors and processing units, typically integrated into clinical settings. These sensors, like microphones and cameras, gather diverse data such as audio from patient-doctor dialogues and visual cues. This information is then processed in real-time or near–real-time.
Natural language processing (NLP): A core component of ACI is advanced NLP algorithms. These algorithms analyze the captured audio data, transcribing spoken words into text. NLP goes beyond mere transcription; it involves understanding context, extracting relevant medical information (like symptoms, diagnoses, and treatment plans), and interpreting the nuances of human language.
Deep learning: Machine learning, particularly deep-learning techniques, are employed to improve the accuracy of ACI systems continually. These algorithms can learn from vast datasets of clinical interactions, enhancing their ability to transcribe and interpret future conversations accurately. As they learn, they become better at understanding different accents, complex medical terms, and variations in speech patterns.
Integration with electronic health records (EHRs): ACI systems are often designed to integrate seamlessly with existing EHR systems. They can automatically populate patient records with information from patient-clinician interactions, reducing manual entry and potential errors.
Customization and personalization: Many ACI systems offer customizable templates or allow clinicians to tailor documentation workflows. This flexibility ensures that the output aligns with the specific needs and preferences of healthcare providers.
Ethical and privacy considerations: ACI systems must navigate significant ethical and privacy concerns, especially related to patient consent and data security. These systems need to comply with healthcare privacy regulations such as HIPAA. They need to securely manage sensitive patient data and restrict access to authorized personnel only.
Broad-Spectrum Benefits of AI in Documentation
- Reducing clinician burnout: By automating the documentation process, AI tools like DAX Copilot alleviate a significant contributor to physician burnout, enabling clinicians to focus more on patient care.
- Enhanced patient care: With AI handling documentation, clinicians can engage more with their patients, leading to improved care quality and patient satisfaction.
- Data accuracy and quality: AI-driven documentation captures detailed patient encounters accurately, ensuring high-quality and comprehensive medical records.
- Response to the growing need for efficient healthcare: AI-based documentation is a direct response to the growing call for more efficient healthcare practices, where clinicians spend less time on paperwork and more with patients.
The shift toward AI-based clinical documentation represents a critical step in addressing the inefficiencies in healthcare systems. It’s a move towards a more patient-centered approach, where clinicians can focus more on patient care by reducing the time spent on excessive charting. Hopefully, we can integrate these solutions into our clinics at a large enough scale to make such an impact.
In the next column, we will explore in-depth insights from Kenneth Harper at Nuance on the technical implementation of these tools, with DAX as an example.
I would love to read your comments on AI in clinical trials as well as other AI-related topics. Write me at [email protected] or find me on X @DrBonillaOnc.
Dr. Loaiza-Bonilla is the co-founder and chief medical officer at Massive Bio, a company connecting patients to clinical trials using artificial intelligence. His research and professional interests focus on precision medicine, clinical trial design, digital health, entrepreneurship, and patient advocacy. Dr Loaiza-Bonilla serves as medical director of oncology research at Capital Health in New Jersey, where he maintains a connection to patient care by attending to patients 2 days a week. He has served as a consultant for Verify, PSI CRO, Bayer, AstraZeneca, Cardinal Health, BrightInsight, The Lynx Group, Fresenius, Pfizer, Ipsen, and Guardant; served as a speaker or a member of a speakers bureau for Amgen, Guardant, Eisai, Ipsen, Natera, Merck, Bristol-Myers Squibb, and AstraZeneca. He holds a 5% or greater equity interest in Massive Bio.
A version of this article appeared on Medscape.com.
Picture a healthcare system where physicians aren’t bogged down by excessive charting but are instead fully present with their patients, offering undivided attention and personalized care. In a recent X post, Stuart Blitz, COO and co-founder of Hone Health, sparked a thought-provoking conversation. “The problem with US healthcare is physicians are burned out since they spend way too much time charting, not enough with patients,” he wrote. “If you created a health system that did zero charting, you’d attract the best physicians and all patients would go there. Who is working on this?”
This resonates with many in the medical community, myself included, because the strain of extensive documentation detracts from patient care. Having worked in both large and small healthcare systems, I know the burden of extensive charting is a palpable challenge, often detracting from the time we can devote to our patients.
The first part of this two-part series examines the overarching benefits of artificial intelligence (AI)–based clinical documentation in modern healthcare, a field witnessing a paradigm shift thanks to advancements in AI.
Transformative Evolution of Clinical Documentation
The transition from manual documentation to AI-driven solutions marks a significant shift in the field, with a number of products in development including Nuance, Abridge, Ambience, ScribeAmerica, 3M, and DeepScribe. These tools use ambient clinical intelligence (ACI) to automate documentation, capturing patient conversations and translating them into structured clinical summaries. This innovation aligns with the vision of reducing charting burdens and enhancing patient-physician interactions.
How does it work? ACI refers to a sophisticated form of AI applied in healthcare settings, particularly focusing on enhancing the clinical documentation process without disrupting the natural flow of the consultation. Here’s a technical yet practical breakdown of ACI and the algorithms it typically employs:
Data capture and processing: ACI systems employ various sensors and processing units, typically integrated into clinical settings. These sensors, like microphones and cameras, gather diverse data such as audio from patient-doctor dialogues and visual cues. This information is then processed in real-time or near–real-time.
Natural language processing (NLP): A core component of ACI is advanced NLP algorithms. These algorithms analyze the captured audio data, transcribing spoken words into text. NLP goes beyond mere transcription; it involves understanding context, extracting relevant medical information (like symptoms, diagnoses, and treatment plans), and interpreting the nuances of human language.
Deep learning: Machine learning, particularly deep-learning techniques, are employed to improve the accuracy of ACI systems continually. These algorithms can learn from vast datasets of clinical interactions, enhancing their ability to transcribe and interpret future conversations accurately. As they learn, they become better at understanding different accents, complex medical terms, and variations in speech patterns.
Integration with electronic health records (EHRs): ACI systems are often designed to integrate seamlessly with existing EHR systems. They can automatically populate patient records with information from patient-clinician interactions, reducing manual entry and potential errors.
Customization and personalization: Many ACI systems offer customizable templates or allow clinicians to tailor documentation workflows. This flexibility ensures that the output aligns with the specific needs and preferences of healthcare providers.
Ethical and privacy considerations: ACI systems must navigate significant ethical and privacy concerns, especially related to patient consent and data security. These systems need to comply with healthcare privacy regulations such as HIPAA. They need to securely manage sensitive patient data and restrict access to authorized personnel only.
Broad-Spectrum Benefits of AI in Documentation
- Reducing clinician burnout: By automating the documentation process, AI tools like DAX Copilot alleviate a significant contributor to physician burnout, enabling clinicians to focus more on patient care.
- Enhanced patient care: With AI handling documentation, clinicians can engage more with their patients, leading to improved care quality and patient satisfaction.
- Data accuracy and quality: AI-driven documentation captures detailed patient encounters accurately, ensuring high-quality and comprehensive medical records.
- Response to the growing need for efficient healthcare: AI-based documentation is a direct response to the growing call for more efficient healthcare practices, where clinicians spend less time on paperwork and more with patients.
The shift toward AI-based clinical documentation represents a critical step in addressing the inefficiencies in healthcare systems. It’s a move towards a more patient-centered approach, where clinicians can focus more on patient care by reducing the time spent on excessive charting. Hopefully, we can integrate these solutions into our clinics at a large enough scale to make such an impact.
In the next column, we will explore in-depth insights from Kenneth Harper at Nuance on the technical implementation of these tools, with DAX as an example.
I would love to read your comments on AI in clinical trials as well as other AI-related topics. Write me at [email protected] or find me on X @DrBonillaOnc.
Dr. Loaiza-Bonilla is the co-founder and chief medical officer at Massive Bio, a company connecting patients to clinical trials using artificial intelligence. His research and professional interests focus on precision medicine, clinical trial design, digital health, entrepreneurship, and patient advocacy. Dr Loaiza-Bonilla serves as medical director of oncology research at Capital Health in New Jersey, where he maintains a connection to patient care by attending to patients 2 days a week. He has served as a consultant for Verify, PSI CRO, Bayer, AstraZeneca, Cardinal Health, BrightInsight, The Lynx Group, Fresenius, Pfizer, Ipsen, and Guardant; served as a speaker or a member of a speakers bureau for Amgen, Guardant, Eisai, Ipsen, Natera, Merck, Bristol-Myers Squibb, and AstraZeneca. He holds a 5% or greater equity interest in Massive Bio.
A version of this article appeared on Medscape.com.
New Federal Rule Delivers Workplace Support, Time Off for Pregnant Docs
Pregnant physicians may receive more workplace accommodations and protection against discrimination thanks to an updated rule from the US Equal Employment Opportunity Commission (EEOC). The guidelines could prevent women from losing critical career momentum.
The Pregnant Workers Fairness Act (PWFA) aims to help workers balance professional demands with healthy pregnancies. It requires employers to provide reasonable accommodations for a “worker’s known limitations,” including physical or mental conditions associated with “pregnancy, childbirth, or related medical conditions.”
Reasonable accommodations vary but may involve time off to attend healthcare appointments or recover from childbirth, extra breaks during a shift, shorter work hours, or the ability to sit instead of stand. Private and public sector employers, including state and local governments, federal agencies, and employment agencies, must abide by the new guidelines unless they can provide evidence that doing so will cause undue hardship.
Female doctors have historically encountered significant barriers to family planning. Years of training cause them to delay having children, often leading to higher rates of infertility, miscarriage, and pregnancy complications than in the general population.
Some specialties, like surgeons, are particularly at risk, with 42% reporting at least one pregnancy loss. Most surgeons work their regular schedules until delivery despite desiring workload reductions, commonly citing unsupportive workplaces as a reason for not seeking accommodations.
Trauma surgeon Qaali Hussein, MD, became pregnant with her first child during her intern year in 2008. She told this news organization that her residency program didn’t even have a maternity policy at the time, and her male supervisor was certain that motherhood would end her surgical career.
She shared how “women usually waited until the end of their training to get pregnant. No one had ever gotten pregnant during the program and returned from maternity leave. I was the first to do so, so there wasn’t a policy or any program support to say, ‘What can we do to help?’ ”
Dr. Hussein used her vacation and sick time, returning to work 4 weeks after delivery. She had five more children, including twins her chief year and another baby during fellowship training in 2014.
Each subsequent pregnancy was met with the same response from program leadership, she recalled. “They’d say, ‘This is it. You may have been able to do the first and second child, but this one will be impossible.’ ”
After the PWFA regulations first became enforceable in June, the EEOC accepted public feedback. The guidelines received nearly 100,000 comments, spurred mainly by the inclusion of abortion care as a qualifying condition for which an employee could receive accommodations. About 54,000 comments called for abortion to be excluded from the final rule, and 40,000 supported keeping the clause.
The EEOC issued the final rule on April 15. It includes abortion care. However, the updated rule “does not require any employee to have — or not to have — an abortion, does not require taxpayers to pay for any abortions, and does not compel health care providers to provide any abortions,” the unpublished version of the final rule said. It is scheduled to take effect 60 days after its publication in the Federal Register on April 19.
Increasing Support for Doctor-Moms
The PWFA supplements other EEOC protections, such as pregnancy discrimination under Title VII of the Civil Rights Act of 1964 and access to reasonable accommodations under the Americans with Disabilities Act. In addition, it builds upon Department of Labor regulations, like the PUMP Act for breastfeeding employees and the Family and Medical Leave Act, which provides 12 weeks of unpaid, job-protected leave for the arrival of a child or certain medical conditions.
FMLA applies only to employees who have worked full-time for at least 12 months for an employer with 50 or more employees. Meanwhile, the unpaid, job-protected leave under the PWFA has no waiting period, lowers the required number of employees to 15, and permits accommodations for up to 40 weeks.
Employers are encouraged to honor “common and simple” requests, like using a closer parking space or pumping or nursing at work, without requiring a doctor’s note, the rule said.
Efforts to improve family leave policies for physicians and residents have been gaining traction. In 2021, the American Board of Medical Specialties began requiring its member boards with training programs lasting 2 or more years to allow at least 6 weeks off for parental, caregiver, and medical leave. This time can be taken without exhausting vacation or sick leave or requiring an extension in training. Over half of the 24 member boards permit leave beyond 6 weeks, including the American Boards of Allergy and Immunology, Emergency Medicine, Family Medicine, Radiology, and Surgery.
Estefania Oliveros, MD, MSc, cardiologist and assistant professor at the Lewis Katz School of Medicine at Temple University, Philadelphia, told this news organization that the Accreditation Council for Graduate Medical Education also requires that residents and fellows receive 6 weeks of paid leave.
“We add to that vacation time, so it gives them at least 8 weeks,” she said. The school has created spaces for nursing mothers — something neither she nor Dr. Hussein had access to when breastfeeding — and encourages the attendings to be proactive in excusing pregnant fellows for appointments.
This differs significantly from her fellowship training experience 6 years ago at another institution, where she worked without accommodations until the day before her cesarean delivery. Dr. Oliveros had to use all her vacation time for recovery, returning to the program after 4 weeks instead of the recommended 6.
“And that’s the story you hear all the time. Not because people are ill-intended; I just don’t think the system is designed to accommodate women, so we lose a lot of talent that way,” said Dr. Oliveros, whose 2019 survey in the Journal of the American College of Cardiology called for more support and protections for pregnant doctors.
Both doctors believe the PWFA will be beneficial but only if leadership in the field takes up the cause.
“The cultures of these institutions determine whether women feel safe or even confident enough to have children in medical school or residency,” said Dr. Hussein.
A version of this article appeared on Medscape.com.
Pregnant physicians may receive more workplace accommodations and protection against discrimination thanks to an updated rule from the US Equal Employment Opportunity Commission (EEOC). The guidelines could prevent women from losing critical career momentum.
The Pregnant Workers Fairness Act (PWFA) aims to help workers balance professional demands with healthy pregnancies. It requires employers to provide reasonable accommodations for a “worker’s known limitations,” including physical or mental conditions associated with “pregnancy, childbirth, or related medical conditions.”
Reasonable accommodations vary but may involve time off to attend healthcare appointments or recover from childbirth, extra breaks during a shift, shorter work hours, or the ability to sit instead of stand. Private and public sector employers, including state and local governments, federal agencies, and employment agencies, must abide by the new guidelines unless they can provide evidence that doing so will cause undue hardship.
Female doctors have historically encountered significant barriers to family planning. Years of training cause them to delay having children, often leading to higher rates of infertility, miscarriage, and pregnancy complications than in the general population.
Some specialties, like surgeons, are particularly at risk, with 42% reporting at least one pregnancy loss. Most surgeons work their regular schedules until delivery despite desiring workload reductions, commonly citing unsupportive workplaces as a reason for not seeking accommodations.
Trauma surgeon Qaali Hussein, MD, became pregnant with her first child during her intern year in 2008. She told this news organization that her residency program didn’t even have a maternity policy at the time, and her male supervisor was certain that motherhood would end her surgical career.
She shared how “women usually waited until the end of their training to get pregnant. No one had ever gotten pregnant during the program and returned from maternity leave. I was the first to do so, so there wasn’t a policy or any program support to say, ‘What can we do to help?’ ”
Dr. Hussein used her vacation and sick time, returning to work 4 weeks after delivery. She had five more children, including twins her chief year and another baby during fellowship training in 2014.
Each subsequent pregnancy was met with the same response from program leadership, she recalled. “They’d say, ‘This is it. You may have been able to do the first and second child, but this one will be impossible.’ ”
After the PWFA regulations first became enforceable in June, the EEOC accepted public feedback. The guidelines received nearly 100,000 comments, spurred mainly by the inclusion of abortion care as a qualifying condition for which an employee could receive accommodations. About 54,000 comments called for abortion to be excluded from the final rule, and 40,000 supported keeping the clause.
The EEOC issued the final rule on April 15. It includes abortion care. However, the updated rule “does not require any employee to have — or not to have — an abortion, does not require taxpayers to pay for any abortions, and does not compel health care providers to provide any abortions,” the unpublished version of the final rule said. It is scheduled to take effect 60 days after its publication in the Federal Register on April 19.
Increasing Support for Doctor-Moms
The PWFA supplements other EEOC protections, such as pregnancy discrimination under Title VII of the Civil Rights Act of 1964 and access to reasonable accommodations under the Americans with Disabilities Act. In addition, it builds upon Department of Labor regulations, like the PUMP Act for breastfeeding employees and the Family and Medical Leave Act, which provides 12 weeks of unpaid, job-protected leave for the arrival of a child or certain medical conditions.
FMLA applies only to employees who have worked full-time for at least 12 months for an employer with 50 or more employees. Meanwhile, the unpaid, job-protected leave under the PWFA has no waiting period, lowers the required number of employees to 15, and permits accommodations for up to 40 weeks.
Employers are encouraged to honor “common and simple” requests, like using a closer parking space or pumping or nursing at work, without requiring a doctor’s note, the rule said.
Efforts to improve family leave policies for physicians and residents have been gaining traction. In 2021, the American Board of Medical Specialties began requiring its member boards with training programs lasting 2 or more years to allow at least 6 weeks off for parental, caregiver, and medical leave. This time can be taken without exhausting vacation or sick leave or requiring an extension in training. Over half of the 24 member boards permit leave beyond 6 weeks, including the American Boards of Allergy and Immunology, Emergency Medicine, Family Medicine, Radiology, and Surgery.
Estefania Oliveros, MD, MSc, cardiologist and assistant professor at the Lewis Katz School of Medicine at Temple University, Philadelphia, told this news organization that the Accreditation Council for Graduate Medical Education also requires that residents and fellows receive 6 weeks of paid leave.
“We add to that vacation time, so it gives them at least 8 weeks,” she said. The school has created spaces for nursing mothers — something neither she nor Dr. Hussein had access to when breastfeeding — and encourages the attendings to be proactive in excusing pregnant fellows for appointments.
This differs significantly from her fellowship training experience 6 years ago at another institution, where she worked without accommodations until the day before her cesarean delivery. Dr. Oliveros had to use all her vacation time for recovery, returning to the program after 4 weeks instead of the recommended 6.
“And that’s the story you hear all the time. Not because people are ill-intended; I just don’t think the system is designed to accommodate women, so we lose a lot of talent that way,” said Dr. Oliveros, whose 2019 survey in the Journal of the American College of Cardiology called for more support and protections for pregnant doctors.
Both doctors believe the PWFA will be beneficial but only if leadership in the field takes up the cause.
“The cultures of these institutions determine whether women feel safe or even confident enough to have children in medical school or residency,” said Dr. Hussein.
A version of this article appeared on Medscape.com.
Pregnant physicians may receive more workplace accommodations and protection against discrimination thanks to an updated rule from the US Equal Employment Opportunity Commission (EEOC). The guidelines could prevent women from losing critical career momentum.
The Pregnant Workers Fairness Act (PWFA) aims to help workers balance professional demands with healthy pregnancies. It requires employers to provide reasonable accommodations for a “worker’s known limitations,” including physical or mental conditions associated with “pregnancy, childbirth, or related medical conditions.”
Reasonable accommodations vary but may involve time off to attend healthcare appointments or recover from childbirth, extra breaks during a shift, shorter work hours, or the ability to sit instead of stand. Private and public sector employers, including state and local governments, federal agencies, and employment agencies, must abide by the new guidelines unless they can provide evidence that doing so will cause undue hardship.
Female doctors have historically encountered significant barriers to family planning. Years of training cause them to delay having children, often leading to higher rates of infertility, miscarriage, and pregnancy complications than in the general population.
Some specialties, like surgeons, are particularly at risk, with 42% reporting at least one pregnancy loss. Most surgeons work their regular schedules until delivery despite desiring workload reductions, commonly citing unsupportive workplaces as a reason for not seeking accommodations.
Trauma surgeon Qaali Hussein, MD, became pregnant with her first child during her intern year in 2008. She told this news organization that her residency program didn’t even have a maternity policy at the time, and her male supervisor was certain that motherhood would end her surgical career.
She shared how “women usually waited until the end of their training to get pregnant. No one had ever gotten pregnant during the program and returned from maternity leave. I was the first to do so, so there wasn’t a policy or any program support to say, ‘What can we do to help?’ ”
Dr. Hussein used her vacation and sick time, returning to work 4 weeks after delivery. She had five more children, including twins her chief year and another baby during fellowship training in 2014.
Each subsequent pregnancy was met with the same response from program leadership, she recalled. “They’d say, ‘This is it. You may have been able to do the first and second child, but this one will be impossible.’ ”
After the PWFA regulations first became enforceable in June, the EEOC accepted public feedback. The guidelines received nearly 100,000 comments, spurred mainly by the inclusion of abortion care as a qualifying condition for which an employee could receive accommodations. About 54,000 comments called for abortion to be excluded from the final rule, and 40,000 supported keeping the clause.
The EEOC issued the final rule on April 15. It includes abortion care. However, the updated rule “does not require any employee to have — or not to have — an abortion, does not require taxpayers to pay for any abortions, and does not compel health care providers to provide any abortions,” the unpublished version of the final rule said. It is scheduled to take effect 60 days after its publication in the Federal Register on April 19.
Increasing Support for Doctor-Moms
The PWFA supplements other EEOC protections, such as pregnancy discrimination under Title VII of the Civil Rights Act of 1964 and access to reasonable accommodations under the Americans with Disabilities Act. In addition, it builds upon Department of Labor regulations, like the PUMP Act for breastfeeding employees and the Family and Medical Leave Act, which provides 12 weeks of unpaid, job-protected leave for the arrival of a child or certain medical conditions.
FMLA applies only to employees who have worked full-time for at least 12 months for an employer with 50 or more employees. Meanwhile, the unpaid, job-protected leave under the PWFA has no waiting period, lowers the required number of employees to 15, and permits accommodations for up to 40 weeks.
Employers are encouraged to honor “common and simple” requests, like using a closer parking space or pumping or nursing at work, without requiring a doctor’s note, the rule said.
Efforts to improve family leave policies for physicians and residents have been gaining traction. In 2021, the American Board of Medical Specialties began requiring its member boards with training programs lasting 2 or more years to allow at least 6 weeks off for parental, caregiver, and medical leave. This time can be taken without exhausting vacation or sick leave or requiring an extension in training. Over half of the 24 member boards permit leave beyond 6 weeks, including the American Boards of Allergy and Immunology, Emergency Medicine, Family Medicine, Radiology, and Surgery.
Estefania Oliveros, MD, MSc, cardiologist and assistant professor at the Lewis Katz School of Medicine at Temple University, Philadelphia, told this news organization that the Accreditation Council for Graduate Medical Education also requires that residents and fellows receive 6 weeks of paid leave.
“We add to that vacation time, so it gives them at least 8 weeks,” she said. The school has created spaces for nursing mothers — something neither she nor Dr. Hussein had access to when breastfeeding — and encourages the attendings to be proactive in excusing pregnant fellows for appointments.
This differs significantly from her fellowship training experience 6 years ago at another institution, where she worked without accommodations until the day before her cesarean delivery. Dr. Oliveros had to use all her vacation time for recovery, returning to the program after 4 weeks instead of the recommended 6.
“And that’s the story you hear all the time. Not because people are ill-intended; I just don’t think the system is designed to accommodate women, so we lose a lot of talent that way,” said Dr. Oliveros, whose 2019 survey in the Journal of the American College of Cardiology called for more support and protections for pregnant doctors.
Both doctors believe the PWFA will be beneficial but only if leadership in the field takes up the cause.
“The cultures of these institutions determine whether women feel safe or even confident enough to have children in medical school or residency,” said Dr. Hussein.
A version of this article appeared on Medscape.com.
AI Surpasses Harvard Docs on Clinical Reasoning Test
TOPLINE:
The AI had more instances of incorrect reasoning than the doctors did but scored better overall.
METHODOLOGY:
- The study involved 39 physicians from two academic medical centers in Boston and the generative AI model GPT-4.
- Participants were presented with 20 simulated clinical cases involving common problems such as pharyngitis, headache, abdominal pain, cough, and chest pain. Each case included sections describing the triage presentation, review of systems, physical examination, and diagnostic testing.
- The primary outcome was the Revised-IDEA (R-IDEA) score, a 10-point scale evaluating clinical reasoning documentation across four domains: Interpretive summary, differential diagnosis, explanation of the lead diagnosis, and alternative diagnoses.
TAKEAWAY:
- AI achieved a median R-IDEA score of 10, higher than attending physicians (median score, 9) and residents (8).
- The chatbot had a significantly higher estimated probability of achieving a high R-IDEA score of 8-10 (0.99) compared with attendings (0.76) and residents (0.56).
- AI provided more responses that contained instances of incorrect clinical reasoning (13.8%) than residents (2.8%) and attending physicians (12.5%). It performed similarly to physicians in diagnostic accuracy and inclusion of cannot-miss diagnoses.
IN PRACTICE:
“Future research should assess clinical reasoning of the LLM-physician interaction, as LLMs will more likely augment, not replace, the human reasoning process,” the authors of the study wrote.
SOURCE:
Adam Rodman, MD, MPH, with Beth Israel Deaconess Medical Center, Boston, was the corresponding author on the paper. The research was published online in JAMA Internal Medicine.
LIMITATIONS:
Simulated clinical cases may not replicate performance in real-world scenarios. Further training could enhance the performance of the AI, so the study may underestimate its capabilities, the researchers noted.
DISCLOSURES:
The study was supported by the Harvard Clinical and Translational Science Center and Harvard University. Authors disclosed financial ties to publishing companies and Solera Health. Dr. Rodman received funding from the Gordon and Betty Moore Foundation.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article appeared on Medscape.com.
TOPLINE:
The AI had more instances of incorrect reasoning than the doctors did but scored better overall.
METHODOLOGY:
- The study involved 39 physicians from two academic medical centers in Boston and the generative AI model GPT-4.
- Participants were presented with 20 simulated clinical cases involving common problems such as pharyngitis, headache, abdominal pain, cough, and chest pain. Each case included sections describing the triage presentation, review of systems, physical examination, and diagnostic testing.
- The primary outcome was the Revised-IDEA (R-IDEA) score, a 10-point scale evaluating clinical reasoning documentation across four domains: Interpretive summary, differential diagnosis, explanation of the lead diagnosis, and alternative diagnoses.
TAKEAWAY:
- AI achieved a median R-IDEA score of 10, higher than attending physicians (median score, 9) and residents (8).
- The chatbot had a significantly higher estimated probability of achieving a high R-IDEA score of 8-10 (0.99) compared with attendings (0.76) and residents (0.56).
- AI provided more responses that contained instances of incorrect clinical reasoning (13.8%) than residents (2.8%) and attending physicians (12.5%). It performed similarly to physicians in diagnostic accuracy and inclusion of cannot-miss diagnoses.
IN PRACTICE:
“Future research should assess clinical reasoning of the LLM-physician interaction, as LLMs will more likely augment, not replace, the human reasoning process,” the authors of the study wrote.
SOURCE:
Adam Rodman, MD, MPH, with Beth Israel Deaconess Medical Center, Boston, was the corresponding author on the paper. The research was published online in JAMA Internal Medicine.
LIMITATIONS:
Simulated clinical cases may not replicate performance in real-world scenarios. Further training could enhance the performance of the AI, so the study may underestimate its capabilities, the researchers noted.
DISCLOSURES:
The study was supported by the Harvard Clinical and Translational Science Center and Harvard University. Authors disclosed financial ties to publishing companies and Solera Health. Dr. Rodman received funding from the Gordon and Betty Moore Foundation.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article appeared on Medscape.com.
TOPLINE:
The AI had more instances of incorrect reasoning than the doctors did but scored better overall.
METHODOLOGY:
- The study involved 39 physicians from two academic medical centers in Boston and the generative AI model GPT-4.
- Participants were presented with 20 simulated clinical cases involving common problems such as pharyngitis, headache, abdominal pain, cough, and chest pain. Each case included sections describing the triage presentation, review of systems, physical examination, and diagnostic testing.
- The primary outcome was the Revised-IDEA (R-IDEA) score, a 10-point scale evaluating clinical reasoning documentation across four domains: Interpretive summary, differential diagnosis, explanation of the lead diagnosis, and alternative diagnoses.
TAKEAWAY:
- AI achieved a median R-IDEA score of 10, higher than attending physicians (median score, 9) and residents (8).
- The chatbot had a significantly higher estimated probability of achieving a high R-IDEA score of 8-10 (0.99) compared with attendings (0.76) and residents (0.56).
- AI provided more responses that contained instances of incorrect clinical reasoning (13.8%) than residents (2.8%) and attending physicians (12.5%). It performed similarly to physicians in diagnostic accuracy and inclusion of cannot-miss diagnoses.
IN PRACTICE:
“Future research should assess clinical reasoning of the LLM-physician interaction, as LLMs will more likely augment, not replace, the human reasoning process,” the authors of the study wrote.
SOURCE:
Adam Rodman, MD, MPH, with Beth Israel Deaconess Medical Center, Boston, was the corresponding author on the paper. The research was published online in JAMA Internal Medicine.
LIMITATIONS:
Simulated clinical cases may not replicate performance in real-world scenarios. Further training could enhance the performance of the AI, so the study may underestimate its capabilities, the researchers noted.
DISCLOSURES:
The study was supported by the Harvard Clinical and Translational Science Center and Harvard University. Authors disclosed financial ties to publishing companies and Solera Health. Dr. Rodman received funding from the Gordon and Betty Moore Foundation.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article appeared on Medscape.com.