User login
Do Cannabis Users Need More Anesthesia During Surgery?
TOPLINE:
However, the clinical relevance of this difference remains unclear.
METHODOLOGY:
- To assess if cannabis use leads to higher doses of inhalational anesthesia during surgery, the researchers conducted a retrospective cohort study comparing the average intraoperative minimum alveolar concentrations of volatile anesthetics (isoflurane and sevoflurane) between older adults who used cannabis products and those who did not.
- The researchers reviewed electronic health records of 22,476 patients aged 65 years or older who underwent surgery at the University of Florida Health System between 2018 and 2020.
- Overall, 268 patients who reported using cannabis within 60 days of surgery (median age, 69 years; 35% women) were matched to 1072 nonusers.
- The median duration of anesthesia was 175 minutes.
- The primary outcome was the intraoperative time-weighted average of isoflurane or sevoflurane minimum alveolar concentration equivalents.
TAKEAWAY:
- Cannabis users had significantly higher average minimum alveolar concentrations of isoflurane or sevoflurane than nonusers (mean, 0.58 vs 0.54; mean difference, 0.04; P = .021).
- The findings were confirmed in a sensitivity analysis that revealed higher mean average minimum alveolar concentrations of anesthesia in cannabis users than in nonusers (0.57 vs 0.53; P = .029).
- Although the 0.04 difference in minimum alveolar concentration between cannabis users and nonusers was statistically significant, its clinical importance is unclear.
IN PRACTICE:
“While recent guidelines underscore the importance of universal screening for cannabinoids before surgery, caution is paramount to prevent clinical bias leading to the administration of unnecessary higher doses of inhalational anesthesia, especially as robust evidence supporting such practices remains lacking,” the authors of the study wrote.
SOURCE:
This study was led by Ruba Sajdeya, MD, PhD, of the Department of Epidemiology at the University of Florida, Gainesville, and was published online in August 2024 in Anesthesiology.
LIMITATIONS:
This study lacked access to prescription or dispensed medications, including opioids, which may have introduced residual confounding. Potential underdocumentation of cannabis use in medical records could have led to exposure misclassification. The causality between cannabis usage and increased anesthetic dosing could not be established due to the observational nature of this study.
DISCLOSURES:
This study was supported by the National Institute on Aging, the National Institutes of Health, and in part by the University of Florida Clinical and Translational Science Institute. Some authors declared receiving research support, consulting fees, and honoraria and having other ties with pharmaceutical companies and various other sources.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article first appeared on Medscape.com.
TOPLINE:
However, the clinical relevance of this difference remains unclear.
METHODOLOGY:
- To assess if cannabis use leads to higher doses of inhalational anesthesia during surgery, the researchers conducted a retrospective cohort study comparing the average intraoperative minimum alveolar concentrations of volatile anesthetics (isoflurane and sevoflurane) between older adults who used cannabis products and those who did not.
- The researchers reviewed electronic health records of 22,476 patients aged 65 years or older who underwent surgery at the University of Florida Health System between 2018 and 2020.
- Overall, 268 patients who reported using cannabis within 60 days of surgery (median age, 69 years; 35% women) were matched to 1072 nonusers.
- The median duration of anesthesia was 175 minutes.
- The primary outcome was the intraoperative time-weighted average of isoflurane or sevoflurane minimum alveolar concentration equivalents.
TAKEAWAY:
- Cannabis users had significantly higher average minimum alveolar concentrations of isoflurane or sevoflurane than nonusers (mean, 0.58 vs 0.54; mean difference, 0.04; P = .021).
- The findings were confirmed in a sensitivity analysis that revealed higher mean average minimum alveolar concentrations of anesthesia in cannabis users than in nonusers (0.57 vs 0.53; P = .029).
- Although the 0.04 difference in minimum alveolar concentration between cannabis users and nonusers was statistically significant, its clinical importance is unclear.
IN PRACTICE:
“While recent guidelines underscore the importance of universal screening for cannabinoids before surgery, caution is paramount to prevent clinical bias leading to the administration of unnecessary higher doses of inhalational anesthesia, especially as robust evidence supporting such practices remains lacking,” the authors of the study wrote.
SOURCE:
This study was led by Ruba Sajdeya, MD, PhD, of the Department of Epidemiology at the University of Florida, Gainesville, and was published online in August 2024 in Anesthesiology.
LIMITATIONS:
This study lacked access to prescription or dispensed medications, including opioids, which may have introduced residual confounding. Potential underdocumentation of cannabis use in medical records could have led to exposure misclassification. The causality between cannabis usage and increased anesthetic dosing could not be established due to the observational nature of this study.
DISCLOSURES:
This study was supported by the National Institute on Aging, the National Institutes of Health, and in part by the University of Florida Clinical and Translational Science Institute. Some authors declared receiving research support, consulting fees, and honoraria and having other ties with pharmaceutical companies and various other sources.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article first appeared on Medscape.com.
TOPLINE:
However, the clinical relevance of this difference remains unclear.
METHODOLOGY:
- To assess if cannabis use leads to higher doses of inhalational anesthesia during surgery, the researchers conducted a retrospective cohort study comparing the average intraoperative minimum alveolar concentrations of volatile anesthetics (isoflurane and sevoflurane) between older adults who used cannabis products and those who did not.
- The researchers reviewed electronic health records of 22,476 patients aged 65 years or older who underwent surgery at the University of Florida Health System between 2018 and 2020.
- Overall, 268 patients who reported using cannabis within 60 days of surgery (median age, 69 years; 35% women) were matched to 1072 nonusers.
- The median duration of anesthesia was 175 minutes.
- The primary outcome was the intraoperative time-weighted average of isoflurane or sevoflurane minimum alveolar concentration equivalents.
TAKEAWAY:
- Cannabis users had significantly higher average minimum alveolar concentrations of isoflurane or sevoflurane than nonusers (mean, 0.58 vs 0.54; mean difference, 0.04; P = .021).
- The findings were confirmed in a sensitivity analysis that revealed higher mean average minimum alveolar concentrations of anesthesia in cannabis users than in nonusers (0.57 vs 0.53; P = .029).
- Although the 0.04 difference in minimum alveolar concentration between cannabis users and nonusers was statistically significant, its clinical importance is unclear.
IN PRACTICE:
“While recent guidelines underscore the importance of universal screening for cannabinoids before surgery, caution is paramount to prevent clinical bias leading to the administration of unnecessary higher doses of inhalational anesthesia, especially as robust evidence supporting such practices remains lacking,” the authors of the study wrote.
SOURCE:
This study was led by Ruba Sajdeya, MD, PhD, of the Department of Epidemiology at the University of Florida, Gainesville, and was published online in August 2024 in Anesthesiology.
LIMITATIONS:
This study lacked access to prescription or dispensed medications, including opioids, which may have introduced residual confounding. Potential underdocumentation of cannabis use in medical records could have led to exposure misclassification. The causality between cannabis usage and increased anesthetic dosing could not be established due to the observational nature of this study.
DISCLOSURES:
This study was supported by the National Institute on Aging, the National Institutes of Health, and in part by the University of Florida Clinical and Translational Science Institute. Some authors declared receiving research support, consulting fees, and honoraria and having other ties with pharmaceutical companies and various other sources.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article first appeared on Medscape.com.
Enhanced Care for Pediatric Patients With Generalized Lichen Planus: Diagnosis and Treatment Tips
Practice Gap
Lichen planus (LP) is an inflammatory cutaneous disorder. Although it often is characterized by the 6 Ps—pruritic, polygonal, planar, purple, papules, and plaques with a predilection for the wrists and ankles—the presentation can vary in morphology and distribution.1-5 With an incidence of approximately 1% in the general population, LP is undoubtedly uncommon.1 Its prevalence in the pediatric population is especially low, with only 2% to 3% of cases manifesting in individuals younger than 20 years.2
Generalized LP (also referred to as eruptive or exanthematous LP) is a rarely reported clinical subtype in which lesions are disseminated or spread rapidly.5 The rarity of generalized LP in children often leads to misdiagnosis or delayed treatment, impacting the patient’s quality of life. Thus, there is a need for heightened awareness among clinicians on the variable presentation of LP in the pediatric population. Incorporating a punch biopsy for the diagnosis of LP when lesions manifest as widespread, erythematous to violaceous, flat-topped papules or plaques, along with the addition of an intramuscular (IM) injection in the treatment plan, improves overall patient outcomes.
Tools and Techniques
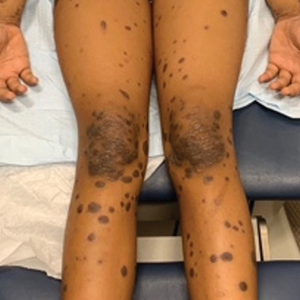
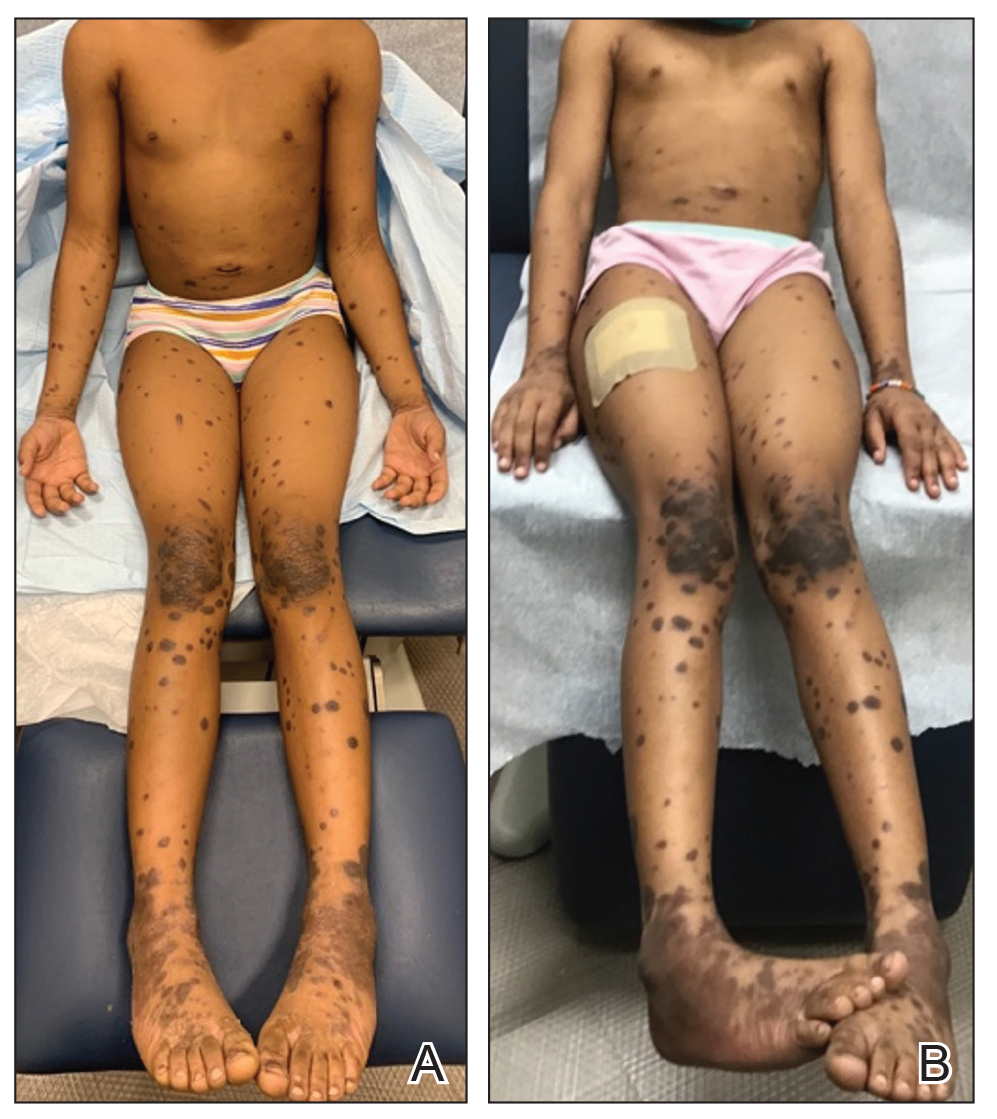
A detailed physical examination followed by a punch biopsy was critical for the diagnosis of generalized LP in a 7-year-old Black girl. The examination revealed a widespread distribution of dark, violaceous, polygonal, shiny, flat-topped, firm papules coalescing into plaques across the entire body, with a greater predilection for the legs and overlying joints (Figure, A). Some lesions exhibited fine, silver-white, reticular patterns consistent with Wickham striae. Notably, there was no involvement of the scalp, nails, or mucosal surfaces.
The patient had no relevant medical or family history of skin disease and no recent history of illness. She previously was treated by a pediatrician with triamcinolone cream 0.1%, a course of oral cephalexin, and oral cetirizine 10 mg once daily without relief of symptoms.
Although the clinical presentation was consistent with LP, the differential diagnosis included lichen simplex chronicus, atopic dermatitis, psoriasis, and generalized granuloma annulare. To address the need for early recognition of LP in pediatric patients, a punch biopsy of a lesion on the left anterior thigh was performed and showed lichenoid interface dermatitis—a pivotal finding in distinguishing LP from other conditions in the differential.
Given the patient’s age and severity of the LP, a combination of topical and systemic therapies was prescribed—clobetasol cream 0.025% twice daily and 1 injection of 0.5 cc of IM triamcinolone acetonide 40 mg/mL. This regimen was guided by the efficacy of IM injections in providing prompt symptomatic relief, particularly for patients with extensive disease or for those whose condition is refractory to topical treatments.6 Our patient achieved remarkable improvement at 2-week follow-up (Figure, B), without any observed adverse effects. At that time, the patient’s mother refused further systemic treatment and opted for only the topical therapy as well as natural light therapy.
Practice Implications
Timely and accurate diagnosis of LP in pediatric patients, especially those with skin of color, is crucial. Early intervention is especially important in mitigating the risk for chronic symptoms and preventing potential scarring, which tends to be more pronounced and challenging to treat in individuals with darker skin tones.7 Although not present in our patient, it is important to note that LP can affect the face (including the eyelids) as well as the palms and soles in pediatric patients with skin of color.
The most common approach to management of pediatric LP involves the use of a topical corticosteroid and an oral antihistamine, but the recalcitrant and generalized distribution of lesions warrants the administration of a systemic corticosteroid regardless of the patient’s age.6 In our patient, prompt administration of low-dose IM triamcinolone was both crucial and beneficial. Although an underutilized approach, IM triamcinolone helps to prevent the progression of lesions to the scalp, nails, and mucosa while also reducing inflammation and pruritus in glabrous skin.8
Triamcinolone acetonide injections—administered at concentrations of 5 to 40 mg/mL—directly into the lesion (0.5–1 cc per 2 cm2) are highly effective in managing recalcitrant thickened lesions such as those seen in hypertrophic LP and palmoplantar LP.6 This treatment is particularly beneficial when lesions are unresponsive to topical therapies. Administered every 3 to 6 weeks, these injections provide rapid symptom relief, typically within 72 hours,6 while also contributing to the reduction of lesion size and thickness over time. The concentration of triamcinolone acetonide should be selected based on the lesion’s severity, with higher concentrations reserved for thicker, more resistant lesions. More frequent injections may be warranted in cases in which rapid lesion reduction is necessary, while less frequent sessions may suffice for maintenance therapy. It is important to follow patients closely for adverse effects, such as signs of local skin atrophy or hypopigmentation, and to adjust the dose or frequency accordingly. To mitigate these risks, consider using the lowest effective concentration and rotating injection sites if treating multiple lesions. Additionally, combining intralesional corticosteroids with topical therapies can enhance outcomes, particularly in cases in which monotherapy is insufficient.
Patients should be monitored vigilantly for complications of LP. The risk for postinflammatory hyperpigmentation is a particular concern for patients with skin of color. Other complications of untreated LP include nail deformities and scarring alopecia.9 Regular and thorough follow-ups every few months to monitor scalp, mucosal, and genital involvement are essential to manage this risk effectively.
Furthermore, patient education is key. Informing patients and their caregivers about the nature of LP, the available treatment options, and the importance of ongoing follow-up can help to enhance treatment adherence and improve overall outcomes.
- Le Cleach L, Chosidow O. Clinical practice. Lichen planus. N Engl J Med. 2012;366:723-732. doi:10.1056/NEJMcp1103641
- Handa S, Sahoo B. Childhood lichen planus: a study of 87 cases. Int J Dermatol. 2002;41:423-427. doi:10.1046/j.1365-4362.2002.01522.x
- George J, Murray T, Bain M. Generalized, eruptive lichen planus in a pediatric patient. Contemp Pediatr. 2022;39:32-34.
- Arnold DL, Krishnamurthy K. Lichen planus. StatPearls [Internet]. Updated June 1, 2023. Accessed August 12, 2024. https://www.ncbi.nlm.nih.gov/books/NBK526126/
- Weston G, Payette M. Update on lichen planus and its clinical variants. Int J Womens Dermatol. 2015;1:140-149. doi:10.1016/j.ijwd.2015.04.001
- Mutalik SD, Belgaumkar VA, Rasal YD. Current perspectives in the treatment of childhood lichen planus. Indian J Paediatr Dermatol. 2021;22:316-325. doi:10.4103/ijpd.ijpd_165_20
- Usatine RP, Tinitigan M. Diagnosis and treatment of lichen planus. Am Fam Physician. 2011;84:53-60.
- Thomas LW, Elsensohn A, Bergheim T, et al. Intramuscular steroids in the treatment of dermatologic disease: a systematic review. J Drugs Dermatol. 2018;17:323-329.
- Gorouhi F, Davari P, Fazel N. Cutaneous and mucosal lichen planus: a comprehensive review of clinical subtypes, risk factors, diagnosis, and prognosis. ScientificWorldJournal. 2014;2014:742826. doi:10.1155/2014/742826
Practice Gap
Lichen planus (LP) is an inflammatory cutaneous disorder. Although it often is characterized by the 6 Ps—pruritic, polygonal, planar, purple, papules, and plaques with a predilection for the wrists and ankles—the presentation can vary in morphology and distribution.1-5 With an incidence of approximately 1% in the general population, LP is undoubtedly uncommon.1 Its prevalence in the pediatric population is especially low, with only 2% to 3% of cases manifesting in individuals younger than 20 years.2
Generalized LP (also referred to as eruptive or exanthematous LP) is a rarely reported clinical subtype in which lesions are disseminated or spread rapidly.5 The rarity of generalized LP in children often leads to misdiagnosis or delayed treatment, impacting the patient’s quality of life. Thus, there is a need for heightened awareness among clinicians on the variable presentation of LP in the pediatric population. Incorporating a punch biopsy for the diagnosis of LP when lesions manifest as widespread, erythematous to violaceous, flat-topped papules or plaques, along with the addition of an intramuscular (IM) injection in the treatment plan, improves overall patient outcomes.
Tools and Techniques
A detailed physical examination followed by a punch biopsy was critical for the diagnosis of generalized LP in a 7-year-old Black girl. The examination revealed a widespread distribution of dark, violaceous, polygonal, shiny, flat-topped, firm papules coalescing into plaques across the entire body, with a greater predilection for the legs and overlying joints (Figure, A). Some lesions exhibited fine, silver-white, reticular patterns consistent with Wickham striae. Notably, there was no involvement of the scalp, nails, or mucosal surfaces.

The patient had no relevant medical or family history of skin disease and no recent history of illness. She previously was treated by a pediatrician with triamcinolone cream 0.1%, a course of oral cephalexin, and oral cetirizine 10 mg once daily without relief of symptoms.
Although the clinical presentation was consistent with LP, the differential diagnosis included lichen simplex chronicus, atopic dermatitis, psoriasis, and generalized granuloma annulare. To address the need for early recognition of LP in pediatric patients, a punch biopsy of a lesion on the left anterior thigh was performed and showed lichenoid interface dermatitis—a pivotal finding in distinguishing LP from other conditions in the differential.
Given the patient’s age and severity of the LP, a combination of topical and systemic therapies was prescribed—clobetasol cream 0.025% twice daily and 1 injection of 0.5 cc of IM triamcinolone acetonide 40 mg/mL. This regimen was guided by the efficacy of IM injections in providing prompt symptomatic relief, particularly for patients with extensive disease or for those whose condition is refractory to topical treatments.6 Our patient achieved remarkable improvement at 2-week follow-up (Figure, B), without any observed adverse effects. At that time, the patient’s mother refused further systemic treatment and opted for only the topical therapy as well as natural light therapy.
Practice Implications
Timely and accurate diagnosis of LP in pediatric patients, especially those with skin of color, is crucial. Early intervention is especially important in mitigating the risk for chronic symptoms and preventing potential scarring, which tends to be more pronounced and challenging to treat in individuals with darker skin tones.7 Although not present in our patient, it is important to note that LP can affect the face (including the eyelids) as well as the palms and soles in pediatric patients with skin of color.
The most common approach to management of pediatric LP involves the use of a topical corticosteroid and an oral antihistamine, but the recalcitrant and generalized distribution of lesions warrants the administration of a systemic corticosteroid regardless of the patient’s age.6 In our patient, prompt administration of low-dose IM triamcinolone was both crucial and beneficial. Although an underutilized approach, IM triamcinolone helps to prevent the progression of lesions to the scalp, nails, and mucosa while also reducing inflammation and pruritus in glabrous skin.8
Triamcinolone acetonide injections—administered at concentrations of 5 to 40 mg/mL—directly into the lesion (0.5–1 cc per 2 cm2) are highly effective in managing recalcitrant thickened lesions such as those seen in hypertrophic LP and palmoplantar LP.6 This treatment is particularly beneficial when lesions are unresponsive to topical therapies. Administered every 3 to 6 weeks, these injections provide rapid symptom relief, typically within 72 hours,6 while also contributing to the reduction of lesion size and thickness over time. The concentration of triamcinolone acetonide should be selected based on the lesion’s severity, with higher concentrations reserved for thicker, more resistant lesions. More frequent injections may be warranted in cases in which rapid lesion reduction is necessary, while less frequent sessions may suffice for maintenance therapy. It is important to follow patients closely for adverse effects, such as signs of local skin atrophy or hypopigmentation, and to adjust the dose or frequency accordingly. To mitigate these risks, consider using the lowest effective concentration and rotating injection sites if treating multiple lesions. Additionally, combining intralesional corticosteroids with topical therapies can enhance outcomes, particularly in cases in which monotherapy is insufficient.
Patients should be monitored vigilantly for complications of LP. The risk for postinflammatory hyperpigmentation is a particular concern for patients with skin of color. Other complications of untreated LP include nail deformities and scarring alopecia.9 Regular and thorough follow-ups every few months to monitor scalp, mucosal, and genital involvement are essential to manage this risk effectively.
Furthermore, patient education is key. Informing patients and their caregivers about the nature of LP, the available treatment options, and the importance of ongoing follow-up can help to enhance treatment adherence and improve overall outcomes.
Practice Gap
Lichen planus (LP) is an inflammatory cutaneous disorder. Although it often is characterized by the 6 Ps—pruritic, polygonal, planar, purple, papules, and plaques with a predilection for the wrists and ankles—the presentation can vary in morphology and distribution.1-5 With an incidence of approximately 1% in the general population, LP is undoubtedly uncommon.1 Its prevalence in the pediatric population is especially low, with only 2% to 3% of cases manifesting in individuals younger than 20 years.2
Generalized LP (also referred to as eruptive or exanthematous LP) is a rarely reported clinical subtype in which lesions are disseminated or spread rapidly.5 The rarity of generalized LP in children often leads to misdiagnosis or delayed treatment, impacting the patient’s quality of life. Thus, there is a need for heightened awareness among clinicians on the variable presentation of LP in the pediatric population. Incorporating a punch biopsy for the diagnosis of LP when lesions manifest as widespread, erythematous to violaceous, flat-topped papules or plaques, along with the addition of an intramuscular (IM) injection in the treatment plan, improves overall patient outcomes.
Tools and Techniques
A detailed physical examination followed by a punch biopsy was critical for the diagnosis of generalized LP in a 7-year-old Black girl. The examination revealed a widespread distribution of dark, violaceous, polygonal, shiny, flat-topped, firm papules coalescing into plaques across the entire body, with a greater predilection for the legs and overlying joints (Figure, A). Some lesions exhibited fine, silver-white, reticular patterns consistent with Wickham striae. Notably, there was no involvement of the scalp, nails, or mucosal surfaces.

The patient had no relevant medical or family history of skin disease and no recent history of illness. She previously was treated by a pediatrician with triamcinolone cream 0.1%, a course of oral cephalexin, and oral cetirizine 10 mg once daily without relief of symptoms.
Although the clinical presentation was consistent with LP, the differential diagnosis included lichen simplex chronicus, atopic dermatitis, psoriasis, and generalized granuloma annulare. To address the need for early recognition of LP in pediatric patients, a punch biopsy of a lesion on the left anterior thigh was performed and showed lichenoid interface dermatitis—a pivotal finding in distinguishing LP from other conditions in the differential.
Given the patient’s age and severity of the LP, a combination of topical and systemic therapies was prescribed—clobetasol cream 0.025% twice daily and 1 injection of 0.5 cc of IM triamcinolone acetonide 40 mg/mL. This regimen was guided by the efficacy of IM injections in providing prompt symptomatic relief, particularly for patients with extensive disease or for those whose condition is refractory to topical treatments.6 Our patient achieved remarkable improvement at 2-week follow-up (Figure, B), without any observed adverse effects. At that time, the patient’s mother refused further systemic treatment and opted for only the topical therapy as well as natural light therapy.
Practice Implications
Timely and accurate diagnosis of LP in pediatric patients, especially those with skin of color, is crucial. Early intervention is especially important in mitigating the risk for chronic symptoms and preventing potential scarring, which tends to be more pronounced and challenging to treat in individuals with darker skin tones.7 Although not present in our patient, it is important to note that LP can affect the face (including the eyelids) as well as the palms and soles in pediatric patients with skin of color.
The most common approach to management of pediatric LP involves the use of a topical corticosteroid and an oral antihistamine, but the recalcitrant and generalized distribution of lesions warrants the administration of a systemic corticosteroid regardless of the patient’s age.6 In our patient, prompt administration of low-dose IM triamcinolone was both crucial and beneficial. Although an underutilized approach, IM triamcinolone helps to prevent the progression of lesions to the scalp, nails, and mucosa while also reducing inflammation and pruritus in glabrous skin.8
Triamcinolone acetonide injections—administered at concentrations of 5 to 40 mg/mL—directly into the lesion (0.5–1 cc per 2 cm2) are highly effective in managing recalcitrant thickened lesions such as those seen in hypertrophic LP and palmoplantar LP.6 This treatment is particularly beneficial when lesions are unresponsive to topical therapies. Administered every 3 to 6 weeks, these injections provide rapid symptom relief, typically within 72 hours,6 while also contributing to the reduction of lesion size and thickness over time. The concentration of triamcinolone acetonide should be selected based on the lesion’s severity, with higher concentrations reserved for thicker, more resistant lesions. More frequent injections may be warranted in cases in which rapid lesion reduction is necessary, while less frequent sessions may suffice for maintenance therapy. It is important to follow patients closely for adverse effects, such as signs of local skin atrophy or hypopigmentation, and to adjust the dose or frequency accordingly. To mitigate these risks, consider using the lowest effective concentration and rotating injection sites if treating multiple lesions. Additionally, combining intralesional corticosteroids with topical therapies can enhance outcomes, particularly in cases in which monotherapy is insufficient.
Patients should be monitored vigilantly for complications of LP. The risk for postinflammatory hyperpigmentation is a particular concern for patients with skin of color. Other complications of untreated LP include nail deformities and scarring alopecia.9 Regular and thorough follow-ups every few months to monitor scalp, mucosal, and genital involvement are essential to manage this risk effectively.
Furthermore, patient education is key. Informing patients and their caregivers about the nature of LP, the available treatment options, and the importance of ongoing follow-up can help to enhance treatment adherence and improve overall outcomes.
- Le Cleach L, Chosidow O. Clinical practice. Lichen planus. N Engl J Med. 2012;366:723-732. doi:10.1056/NEJMcp1103641
- Handa S, Sahoo B. Childhood lichen planus: a study of 87 cases. Int J Dermatol. 2002;41:423-427. doi:10.1046/j.1365-4362.2002.01522.x
- George J, Murray T, Bain M. Generalized, eruptive lichen planus in a pediatric patient. Contemp Pediatr. 2022;39:32-34.
- Arnold DL, Krishnamurthy K. Lichen planus. StatPearls [Internet]. Updated June 1, 2023. Accessed August 12, 2024. https://www.ncbi.nlm.nih.gov/books/NBK526126/
- Weston G, Payette M. Update on lichen planus and its clinical variants. Int J Womens Dermatol. 2015;1:140-149. doi:10.1016/j.ijwd.2015.04.001
- Mutalik SD, Belgaumkar VA, Rasal YD. Current perspectives in the treatment of childhood lichen planus. Indian J Paediatr Dermatol. 2021;22:316-325. doi:10.4103/ijpd.ijpd_165_20
- Usatine RP, Tinitigan M. Diagnosis and treatment of lichen planus. Am Fam Physician. 2011;84:53-60.
- Thomas LW, Elsensohn A, Bergheim T, et al. Intramuscular steroids in the treatment of dermatologic disease: a systematic review. J Drugs Dermatol. 2018;17:323-329.
- Gorouhi F, Davari P, Fazel N. Cutaneous and mucosal lichen planus: a comprehensive review of clinical subtypes, risk factors, diagnosis, and prognosis. ScientificWorldJournal. 2014;2014:742826. doi:10.1155/2014/742826
- Le Cleach L, Chosidow O. Clinical practice. Lichen planus. N Engl J Med. 2012;366:723-732. doi:10.1056/NEJMcp1103641
- Handa S, Sahoo B. Childhood lichen planus: a study of 87 cases. Int J Dermatol. 2002;41:423-427. doi:10.1046/j.1365-4362.2002.01522.x
- George J, Murray T, Bain M. Generalized, eruptive lichen planus in a pediatric patient. Contemp Pediatr. 2022;39:32-34.
- Arnold DL, Krishnamurthy K. Lichen planus. StatPearls [Internet]. Updated June 1, 2023. Accessed August 12, 2024. https://www.ncbi.nlm.nih.gov/books/NBK526126/
- Weston G, Payette M. Update on lichen planus and its clinical variants. Int J Womens Dermatol. 2015;1:140-149. doi:10.1016/j.ijwd.2015.04.001
- Mutalik SD, Belgaumkar VA, Rasal YD. Current perspectives in the treatment of childhood lichen planus. Indian J Paediatr Dermatol. 2021;22:316-325. doi:10.4103/ijpd.ijpd_165_20
- Usatine RP, Tinitigan M. Diagnosis and treatment of lichen planus. Am Fam Physician. 2011;84:53-60.
- Thomas LW, Elsensohn A, Bergheim T, et al. Intramuscular steroids in the treatment of dermatologic disease: a systematic review. J Drugs Dermatol. 2018;17:323-329.
- Gorouhi F, Davari P, Fazel N. Cutaneous and mucosal lichen planus: a comprehensive review of clinical subtypes, risk factors, diagnosis, and prognosis. ScientificWorldJournal. 2014;2014:742826. doi:10.1155/2014/742826
Nonhormonal Treatment May Ease Menopausal Symptoms
Elinzanetant, the selective antagonist of neurokinin 1 and 3 receptors, led to rapid improvement in the frequency of vasomotor symptoms and significant improvements in the severity of symptoms, sleep disturbances, and menopause-related quality of life in two phase 3 studies. Researchers led by JoAnn V. Pinkerton, MD, from the University of Virginia Health in Charlottesville, reported their findings, which resulted from the randomized OASIS 1 and 2 studies, in JAMA.
“Women experience a variety of symptoms during their menopausal transition, including vasomotor symptoms ... and sleep disturbances, reported by up to 80% and 60%, respectively,” wrote the researchers. “Menopausal symptoms can negatively impact quality of life, reducing the capacity for daily activities and work productivity, and may be associated with long-term negative health outcomes such as cardiovascular events, depressive symptoms, cognitive decline, and other adverse brain outcomes.” The researchers also noted that some therapeutic options are available, including hormone replacement therapy and, in some countries, paroxetine, a selective serotonin reuptake inhibitor.
The Italian Ministry of Health’s menopause website points out that the transition generally occurs between ages 45 and 55 years. This huge hormonal change has consequences for women’s health. Ministry experts explain that diet and hormone replacement therapy (which should be taken under medical supervision) can prevent or counteract these consequences.
“Many women have contraindications, have tolerability issues leading to discontinuation, or prefer not to take these treatments,” wrote Dr. Pinkerton and colleagues, who evaluated the efficacy and tolerability of elinzanetant, a nonhormonal alternative treatment in development. The two double-blind, randomized, phase 3 studies (OASIS 1 and 2) included postmenopausal participants between ages 40 and 65 years with moderate to severe vasomotor symptoms who were treated with elinzanetant (OASIS 1, n = 199; OASIS 2, n = 200) or placebo (OASIS 1, n = 197; OASIS 2, n = 200).
After 4 weeks of treatment, 62.8% of participants in the OASIS 1 study and 62.2% in the OASIS 2 study reported at least a 50% reduction in the frequency of vasomotor symptoms (29.2% and 32.3% in the respective placebo groups). Improvements increased by week 12, with 71.4% and 74.7% of women in the elinzanetant group achieving this reduction (42.0% and 48.3% in the respective placebo groups). Women who took the medication also reported a reduction in the severity of vasomotor symptoms and improvements in sleep and menopause-related quality of life, with no significant tolerability and safety issues. “Elinzanetant has the potential to provide a well-tolerated and efficacious nonhormonal treatment option to address the unmet health needs of many menopausal individuals with moderate to severe vasomotor symptoms,” the authors concluded.
“With the discovery of nonhormonal treatment options targeting the neurons responsible for vasomotor symptoms, menopause care should advance on this solid scientific footing to benefit affected individuals,” wrote Stephanie S. Faubion, MD, and Chrisandra L. Shufelt, MD, who are affiliated with the Mayo Clinic in Rochester, Minnesota, and Jacksonville, Florida, in an accompanying editorial.
This story was translated from Univadis Italy using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article appeared on Medscape.com.
Elinzanetant, the selective antagonist of neurokinin 1 and 3 receptors, led to rapid improvement in the frequency of vasomotor symptoms and significant improvements in the severity of symptoms, sleep disturbances, and menopause-related quality of life in two phase 3 studies. Researchers led by JoAnn V. Pinkerton, MD, from the University of Virginia Health in Charlottesville, reported their findings, which resulted from the randomized OASIS 1 and 2 studies, in JAMA.
“Women experience a variety of symptoms during their menopausal transition, including vasomotor symptoms ... and sleep disturbances, reported by up to 80% and 60%, respectively,” wrote the researchers. “Menopausal symptoms can negatively impact quality of life, reducing the capacity for daily activities and work productivity, and may be associated with long-term negative health outcomes such as cardiovascular events, depressive symptoms, cognitive decline, and other adverse brain outcomes.” The researchers also noted that some therapeutic options are available, including hormone replacement therapy and, in some countries, paroxetine, a selective serotonin reuptake inhibitor.
The Italian Ministry of Health’s menopause website points out that the transition generally occurs between ages 45 and 55 years. This huge hormonal change has consequences for women’s health. Ministry experts explain that diet and hormone replacement therapy (which should be taken under medical supervision) can prevent or counteract these consequences.
“Many women have contraindications, have tolerability issues leading to discontinuation, or prefer not to take these treatments,” wrote Dr. Pinkerton and colleagues, who evaluated the efficacy and tolerability of elinzanetant, a nonhormonal alternative treatment in development. The two double-blind, randomized, phase 3 studies (OASIS 1 and 2) included postmenopausal participants between ages 40 and 65 years with moderate to severe vasomotor symptoms who were treated with elinzanetant (OASIS 1, n = 199; OASIS 2, n = 200) or placebo (OASIS 1, n = 197; OASIS 2, n = 200).
After 4 weeks of treatment, 62.8% of participants in the OASIS 1 study and 62.2% in the OASIS 2 study reported at least a 50% reduction in the frequency of vasomotor symptoms (29.2% and 32.3% in the respective placebo groups). Improvements increased by week 12, with 71.4% and 74.7% of women in the elinzanetant group achieving this reduction (42.0% and 48.3% in the respective placebo groups). Women who took the medication also reported a reduction in the severity of vasomotor symptoms and improvements in sleep and menopause-related quality of life, with no significant tolerability and safety issues. “Elinzanetant has the potential to provide a well-tolerated and efficacious nonhormonal treatment option to address the unmet health needs of many menopausal individuals with moderate to severe vasomotor symptoms,” the authors concluded.
“With the discovery of nonhormonal treatment options targeting the neurons responsible for vasomotor symptoms, menopause care should advance on this solid scientific footing to benefit affected individuals,” wrote Stephanie S. Faubion, MD, and Chrisandra L. Shufelt, MD, who are affiliated with the Mayo Clinic in Rochester, Minnesota, and Jacksonville, Florida, in an accompanying editorial.
This story was translated from Univadis Italy using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article appeared on Medscape.com.
Elinzanetant, the selective antagonist of neurokinin 1 and 3 receptors, led to rapid improvement in the frequency of vasomotor symptoms and significant improvements in the severity of symptoms, sleep disturbances, and menopause-related quality of life in two phase 3 studies. Researchers led by JoAnn V. Pinkerton, MD, from the University of Virginia Health in Charlottesville, reported their findings, which resulted from the randomized OASIS 1 and 2 studies, in JAMA.
“Women experience a variety of symptoms during their menopausal transition, including vasomotor symptoms ... and sleep disturbances, reported by up to 80% and 60%, respectively,” wrote the researchers. “Menopausal symptoms can negatively impact quality of life, reducing the capacity for daily activities and work productivity, and may be associated with long-term negative health outcomes such as cardiovascular events, depressive symptoms, cognitive decline, and other adverse brain outcomes.” The researchers also noted that some therapeutic options are available, including hormone replacement therapy and, in some countries, paroxetine, a selective serotonin reuptake inhibitor.
The Italian Ministry of Health’s menopause website points out that the transition generally occurs between ages 45 and 55 years. This huge hormonal change has consequences for women’s health. Ministry experts explain that diet and hormone replacement therapy (which should be taken under medical supervision) can prevent or counteract these consequences.
“Many women have contraindications, have tolerability issues leading to discontinuation, or prefer not to take these treatments,” wrote Dr. Pinkerton and colleagues, who evaluated the efficacy and tolerability of elinzanetant, a nonhormonal alternative treatment in development. The two double-blind, randomized, phase 3 studies (OASIS 1 and 2) included postmenopausal participants between ages 40 and 65 years with moderate to severe vasomotor symptoms who were treated with elinzanetant (OASIS 1, n = 199; OASIS 2, n = 200) or placebo (OASIS 1, n = 197; OASIS 2, n = 200).
After 4 weeks of treatment, 62.8% of participants in the OASIS 1 study and 62.2% in the OASIS 2 study reported at least a 50% reduction in the frequency of vasomotor symptoms (29.2% and 32.3% in the respective placebo groups). Improvements increased by week 12, with 71.4% and 74.7% of women in the elinzanetant group achieving this reduction (42.0% and 48.3% in the respective placebo groups). Women who took the medication also reported a reduction in the severity of vasomotor symptoms and improvements in sleep and menopause-related quality of life, with no significant tolerability and safety issues. “Elinzanetant has the potential to provide a well-tolerated and efficacious nonhormonal treatment option to address the unmet health needs of many menopausal individuals with moderate to severe vasomotor symptoms,” the authors concluded.
“With the discovery of nonhormonal treatment options targeting the neurons responsible for vasomotor symptoms, menopause care should advance on this solid scientific footing to benefit affected individuals,” wrote Stephanie S. Faubion, MD, and Chrisandra L. Shufelt, MD, who are affiliated with the Mayo Clinic in Rochester, Minnesota, and Jacksonville, Florida, in an accompanying editorial.
This story was translated from Univadis Italy using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article appeared on Medscape.com.
FROM JAMA
Monitor Asthma Patients on Biologics for Remission, Potential EGPA Symptoms During Steroid Tapering
VIENNA — , according to pulmonary experts presenting at the European Respiratory Society (ERS) 2024 International Congress.
Biologics have revolutionized the treatment of severe asthma, significantly improving patient outcomes. However, the focus has recently shifted toward achieving more comprehensive disease control. Remission, already a well-established goal in conditions like rheumatoid arthritis and inflammatory bowel disease, is now being explored in patients with asthma receiving biologics.
Peter Howarth, medical director at Global Medical, Specialty Medicine, GSK, in Brentford, England, said that new clinical remission criteria in asthma may be overly rigid and of little use. He said that more attainable limits must be created. Meanwhile, clinicians should collect clinical data more thoroughly.
In parallel, studies have also raised questions about the role of biologics in the emergence of EGPA.
Defining Clinical Remission in Asthma
Last year, a working group, including members from the American Thoracic Society and the American College and Academy of Allergy, Asthma, and Immunology, proposed new guidelines to define clinical remission in asthma. These guidelines extended beyond the typical outcomes of no severe exacerbations, no maintenance oral corticosteroid use, good asthma control, and stable lung function. The additional recommendations included no missed work or school due to asthma, limited use of rescue medication (no more than once a month), and reduced inhaled corticosteroid use to low or medium doses.
To explore the feasibility of achieving these clinical remission outcomes, GSK partnered with the Mayo Clinic for a retrospective analysis of the medical records of 700 patients with asthma undergoing various biologic therapies. The study revealed that essential data for determining clinical remission, such as asthma control and exacerbation records, were inconsistently documented. While some data were recorded, such as maintenance corticosteroid use in 50%-60% of cases, other key measures, like asthma control, were recorded in less than a quarter of the patients.
GSK researchers analyzed available data and found that around 30% of patients on any biologic therapy met three components of remission. Mepolizumab performed better than other corticosteroids, with over 40% of those receiving the drug meeting these criteria. However, when stricter definitions were applied, such as requiring four or more remission components, fewer patients achieved remission — less than 10% for four components, with no patients meeting the full seven-point criteria proposed by the working group.
An ongoing ERS Task Force is now exploring what clinical remission outcomes are practical to achieve, as the current definitions may be too aspirational, said Mr. Howarth. “It’s a matter of defying what is practical to achieve because if you can’t achieve it, then it won’t be valuable.”
He also pointed out that biologics are often used for the most severe cases of asthma after other treatments have failed. Evidence suggests that introducing biologics earlier in the disease, before chronic damage occurs, may result in better patient outcomes.
Biologics and EGPA
In a retrospective study, clinical details of 27 patients with adult-onset asthma from 28 countries, all on biologic therapy, were analyzed. The study, a multicounty collaboration, was led by ERS Severe Heterogeneous Asthma Research Collaboration, Patient-centred (SHARP), and aimed to understand the role of biologics in the emergence of EGPA.
The most significant finding presented at the ERS 2024 International Congress was that EGPA was not associated with maintenance corticosteroids; instead, it often emerged when corticosteroid doses were reduced or tapered off. “This might suggest that steroid withdrawal may unmask the underlying disease,” said Hitasha Rupani, MD, a consultant respiratory physician at the University Hospital Southampton, in Southampton, England. Importantly, the rate at which steroids were tapered did not influence the onset of EGPA, indicating that the tapering process, rather than its speed, may be the critical factor. However, due to the small sample size, this remains a hypothesis, Dr. Rupani explained.
The study also found that when clinicians had a clinical suspicion of EGPA before starting biologic therapy, the diagnosis was made earlier than in cases without such suspicion. Dr. Rupani concluded that this underscores the importance of clinical vigilance and the need to monitor patients closely for EGPA symptoms, especially during corticosteroid tapering.
The study was funded by GSK. Mr. Howarth is an employee at GSK. Dr. Rupani reports no relevant financial relationships.
A version of this article appeared on Medscape.com.
VIENNA — , according to pulmonary experts presenting at the European Respiratory Society (ERS) 2024 International Congress.
Biologics have revolutionized the treatment of severe asthma, significantly improving patient outcomes. However, the focus has recently shifted toward achieving more comprehensive disease control. Remission, already a well-established goal in conditions like rheumatoid arthritis and inflammatory bowel disease, is now being explored in patients with asthma receiving biologics.
Peter Howarth, medical director at Global Medical, Specialty Medicine, GSK, in Brentford, England, said that new clinical remission criteria in asthma may be overly rigid and of little use. He said that more attainable limits must be created. Meanwhile, clinicians should collect clinical data more thoroughly.
In parallel, studies have also raised questions about the role of biologics in the emergence of EGPA.
Defining Clinical Remission in Asthma
Last year, a working group, including members from the American Thoracic Society and the American College and Academy of Allergy, Asthma, and Immunology, proposed new guidelines to define clinical remission in asthma. These guidelines extended beyond the typical outcomes of no severe exacerbations, no maintenance oral corticosteroid use, good asthma control, and stable lung function. The additional recommendations included no missed work or school due to asthma, limited use of rescue medication (no more than once a month), and reduced inhaled corticosteroid use to low or medium doses.
To explore the feasibility of achieving these clinical remission outcomes, GSK partnered with the Mayo Clinic for a retrospective analysis of the medical records of 700 patients with asthma undergoing various biologic therapies. The study revealed that essential data for determining clinical remission, such as asthma control and exacerbation records, were inconsistently documented. While some data were recorded, such as maintenance corticosteroid use in 50%-60% of cases, other key measures, like asthma control, were recorded in less than a quarter of the patients.
GSK researchers analyzed available data and found that around 30% of patients on any biologic therapy met three components of remission. Mepolizumab performed better than other corticosteroids, with over 40% of those receiving the drug meeting these criteria. However, when stricter definitions were applied, such as requiring four or more remission components, fewer patients achieved remission — less than 10% for four components, with no patients meeting the full seven-point criteria proposed by the working group.
An ongoing ERS Task Force is now exploring what clinical remission outcomes are practical to achieve, as the current definitions may be too aspirational, said Mr. Howarth. “It’s a matter of defying what is practical to achieve because if you can’t achieve it, then it won’t be valuable.”
He also pointed out that biologics are often used for the most severe cases of asthma after other treatments have failed. Evidence suggests that introducing biologics earlier in the disease, before chronic damage occurs, may result in better patient outcomes.
Biologics and EGPA
In a retrospective study, clinical details of 27 patients with adult-onset asthma from 28 countries, all on biologic therapy, were analyzed. The study, a multicounty collaboration, was led by ERS Severe Heterogeneous Asthma Research Collaboration, Patient-centred (SHARP), and aimed to understand the role of biologics in the emergence of EGPA.
The most significant finding presented at the ERS 2024 International Congress was that EGPA was not associated with maintenance corticosteroids; instead, it often emerged when corticosteroid doses were reduced or tapered off. “This might suggest that steroid withdrawal may unmask the underlying disease,” said Hitasha Rupani, MD, a consultant respiratory physician at the University Hospital Southampton, in Southampton, England. Importantly, the rate at which steroids were tapered did not influence the onset of EGPA, indicating that the tapering process, rather than its speed, may be the critical factor. However, due to the small sample size, this remains a hypothesis, Dr. Rupani explained.
The study also found that when clinicians had a clinical suspicion of EGPA before starting biologic therapy, the diagnosis was made earlier than in cases without such suspicion. Dr. Rupani concluded that this underscores the importance of clinical vigilance and the need to monitor patients closely for EGPA symptoms, especially during corticosteroid tapering.
The study was funded by GSK. Mr. Howarth is an employee at GSK. Dr. Rupani reports no relevant financial relationships.
A version of this article appeared on Medscape.com.
VIENNA — , according to pulmonary experts presenting at the European Respiratory Society (ERS) 2024 International Congress.
Biologics have revolutionized the treatment of severe asthma, significantly improving patient outcomes. However, the focus has recently shifted toward achieving more comprehensive disease control. Remission, already a well-established goal in conditions like rheumatoid arthritis and inflammatory bowel disease, is now being explored in patients with asthma receiving biologics.
Peter Howarth, medical director at Global Medical, Specialty Medicine, GSK, in Brentford, England, said that new clinical remission criteria in asthma may be overly rigid and of little use. He said that more attainable limits must be created. Meanwhile, clinicians should collect clinical data more thoroughly.
In parallel, studies have also raised questions about the role of biologics in the emergence of EGPA.
Defining Clinical Remission in Asthma
Last year, a working group, including members from the American Thoracic Society and the American College and Academy of Allergy, Asthma, and Immunology, proposed new guidelines to define clinical remission in asthma. These guidelines extended beyond the typical outcomes of no severe exacerbations, no maintenance oral corticosteroid use, good asthma control, and stable lung function. The additional recommendations included no missed work or school due to asthma, limited use of rescue medication (no more than once a month), and reduced inhaled corticosteroid use to low or medium doses.
To explore the feasibility of achieving these clinical remission outcomes, GSK partnered with the Mayo Clinic for a retrospective analysis of the medical records of 700 patients with asthma undergoing various biologic therapies. The study revealed that essential data for determining clinical remission, such as asthma control and exacerbation records, were inconsistently documented. While some data were recorded, such as maintenance corticosteroid use in 50%-60% of cases, other key measures, like asthma control, were recorded in less than a quarter of the patients.
GSK researchers analyzed available data and found that around 30% of patients on any biologic therapy met three components of remission. Mepolizumab performed better than other corticosteroids, with over 40% of those receiving the drug meeting these criteria. However, when stricter definitions were applied, such as requiring four or more remission components, fewer patients achieved remission — less than 10% for four components, with no patients meeting the full seven-point criteria proposed by the working group.
An ongoing ERS Task Force is now exploring what clinical remission outcomes are practical to achieve, as the current definitions may be too aspirational, said Mr. Howarth. “It’s a matter of defying what is practical to achieve because if you can’t achieve it, then it won’t be valuable.”
He also pointed out that biologics are often used for the most severe cases of asthma after other treatments have failed. Evidence suggests that introducing biologics earlier in the disease, before chronic damage occurs, may result in better patient outcomes.
Biologics and EGPA
In a retrospective study, clinical details of 27 patients with adult-onset asthma from 28 countries, all on biologic therapy, were analyzed. The study, a multicounty collaboration, was led by ERS Severe Heterogeneous Asthma Research Collaboration, Patient-centred (SHARP), and aimed to understand the role of biologics in the emergence of EGPA.
The most significant finding presented at the ERS 2024 International Congress was that EGPA was not associated with maintenance corticosteroids; instead, it often emerged when corticosteroid doses were reduced or tapered off. “This might suggest that steroid withdrawal may unmask the underlying disease,” said Hitasha Rupani, MD, a consultant respiratory physician at the University Hospital Southampton, in Southampton, England. Importantly, the rate at which steroids were tapered did not influence the onset of EGPA, indicating that the tapering process, rather than its speed, may be the critical factor. However, due to the small sample size, this remains a hypothesis, Dr. Rupani explained.
The study also found that when clinicians had a clinical suspicion of EGPA before starting biologic therapy, the diagnosis was made earlier than in cases without such suspicion. Dr. Rupani concluded that this underscores the importance of clinical vigilance and the need to monitor patients closely for EGPA symptoms, especially during corticosteroid tapering.
The study was funded by GSK. Mr. Howarth is an employee at GSK. Dr. Rupani reports no relevant financial relationships.
A version of this article appeared on Medscape.com.
Night Owls May Be at Greater Risk for T2D, Beyond Lifestyle
MADRID — research presented at the annual meeting of the European Association for the Study of Diabetes suggested.
In the study, night owls were almost 50% more likely to develop T2D than those who went to sleep earlier.
“The magnitude of this risk was more than I expected, [although] residual confounding may have occurred,” said Jeroen van der Velde, PhD, Leiden University Medical Center in the Netherlands, who presented the study.
“Late chronotype has previously been associated with unhealthy lifestyle and overweight or obesity and, subsequently, cardiometabolic diseases,” he said in an interview. However, although the current study found that individuals with late chronotypes did indeed have larger waists and more visceral fat, “we (and others) believe that lifestyle cannot fully explain the relation between late chronotype and metabolic disorders.”
“In addition,” he noted, “previous studies that observed that late chronotype is associated with overweight or obesity mainly focused on body mass index [BMI]. However, BMI alone does not provide accurate information regarding fat distribution in the body. People with similar BMI may have different underlying fat distribution, and this may be more relevant than BMI for metabolic risk.”
The researchers examined associations between chronotype and BMI, waist circumference, visceral fat, liver fat, and the risk for T2D in a middle-aged population from the Netherlands Epidemiology of Obesity study. Among the 5026 participants, the mean age was 56 years, 54% were women, and mean BMI was 30.
Using data from the study, the study investigators calculated the midpoint of sleep (MPS) and divided participants into three chronotypes: Early MPS < 2:30 PM (20% of participants); intermediate MPS 2:30–4:00 PM (reference category; 60% of participants); and late MPS ≥ 4:00 PM (20% of participants). BMI and waist circumference were measured in all participants, and visceral fat and liver fat were measured in 1576 participants using MRI scans and MR spectroscopy, respectively.
During a median follow-up of 6.6 years, 225 participants were diagnosed with T2D. After adjustment for age, sex, education, physical activity, smoking, alcohol intake, diet quality, sleep quality and duration, and total body fat, participants with a late chronotype had a 46% increased risk for T2D.
Further, those with a late chronotype had 0.7 higher BMI, 1.9-cm larger waist circumference, 7 cm2 more visceral fat, and 14% more liver fat.
Body Clock Out of Sync?
“Late chronotype was associated with increased ectopic body fat and with an increased risk of T2D independent of lifestyle factors and is an emerging risk factor for metabolic diseases,” the researchers concluded.
“A likely explanation is that the circadian rhythm or body clock in late chronotypes is out of sync with the work and social schedules followed by society,” Dr. van der Velde suggested. “This can lead to circadian misalignment, which we know can lead to metabolic disturbances and ultimately type 2 diabetes.”
Might trying to adjust chronotype earlier in life have an effect on risk?
“Chronotype, as measured via midpoint of sleep, does change a lot in the first 30 years or so in life,” he said. “After that it seems to stabilize. I suppose that if you adapt an intermediate or early chronotype around the age of 30 years, this will help to maintain an earlier chronotype later in life, although we cannot answer this from our study.”
Nevertheless, with respect to T2D risk, “chronotype is likely only part of the puzzle,” he noted.
“People with late chronotypes typically eat late in the evening, and this has also been associated with adverse metabolic effects. At this stage, we do not know if a person changes his/her chronotype that this will also lead to metabolic improvements. More research is needed before we can make recommendations regarding chronotype and timing of other lifestyle behaviors.”
Commenting on the study, Gianluca Iacobellis, MD, PhD, director of the University of Miami Hospital Diabetes Service, Coral Gables, Florida, said: “Interesting data. Altering the physiological circadian rhythm can affect the complex hormonal system — including cortisol, ghrelin, leptin, and serotonin — that regulates insulin sensitivity, glucose, and blood pressure control. The night owl may become more insulin resistant and therefore at higher risk of developing diabetes.”
Like Dr. van der Velde, he noted that “late sleep may be associated with night binging that can cause weight gain and ultimately obesity, further increasing the risk of diabetes.”
Dr. Iacobellis’s group recently showed that vital exhaustion, which is characterized by fatigue and loss of vigor, is associated with a higher cardiovascular risk for and markers of visceral adiposity.
“Abnormal circadian rhythms can be easily associated with vital exhaustion,” he said. Therefore, night owls with more visceral than peripheral fat accumulation might also be at higher cardiometabolic risk through that mechanism.
“However environmental factors and family history can play an important role too,” he added.
Regardless of the mechanisms involved, “preventive actions should be taken to educate teenagers and individuals at higher risk to have healthy sleep habits,” Dr. Iacobellis concluded.
No information regarding funding was provided; Dr. van der Velde and Dr. Iacobellis reported no conflicts of interest.
A version of this article first appeared on Medscape.com.
MADRID — research presented at the annual meeting of the European Association for the Study of Diabetes suggested.
In the study, night owls were almost 50% more likely to develop T2D than those who went to sleep earlier.
“The magnitude of this risk was more than I expected, [although] residual confounding may have occurred,” said Jeroen van der Velde, PhD, Leiden University Medical Center in the Netherlands, who presented the study.
“Late chronotype has previously been associated with unhealthy lifestyle and overweight or obesity and, subsequently, cardiometabolic diseases,” he said in an interview. However, although the current study found that individuals with late chronotypes did indeed have larger waists and more visceral fat, “we (and others) believe that lifestyle cannot fully explain the relation between late chronotype and metabolic disorders.”
“In addition,” he noted, “previous studies that observed that late chronotype is associated with overweight or obesity mainly focused on body mass index [BMI]. However, BMI alone does not provide accurate information regarding fat distribution in the body. People with similar BMI may have different underlying fat distribution, and this may be more relevant than BMI for metabolic risk.”
The researchers examined associations between chronotype and BMI, waist circumference, visceral fat, liver fat, and the risk for T2D in a middle-aged population from the Netherlands Epidemiology of Obesity study. Among the 5026 participants, the mean age was 56 years, 54% were women, and mean BMI was 30.
Using data from the study, the study investigators calculated the midpoint of sleep (MPS) and divided participants into three chronotypes: Early MPS < 2:30 PM (20% of participants); intermediate MPS 2:30–4:00 PM (reference category; 60% of participants); and late MPS ≥ 4:00 PM (20% of participants). BMI and waist circumference were measured in all participants, and visceral fat and liver fat were measured in 1576 participants using MRI scans and MR spectroscopy, respectively.
During a median follow-up of 6.6 years, 225 participants were diagnosed with T2D. After adjustment for age, sex, education, physical activity, smoking, alcohol intake, diet quality, sleep quality and duration, and total body fat, participants with a late chronotype had a 46% increased risk for T2D.
Further, those with a late chronotype had 0.7 higher BMI, 1.9-cm larger waist circumference, 7 cm2 more visceral fat, and 14% more liver fat.
Body Clock Out of Sync?
“Late chronotype was associated with increased ectopic body fat and with an increased risk of T2D independent of lifestyle factors and is an emerging risk factor for metabolic diseases,” the researchers concluded.
“A likely explanation is that the circadian rhythm or body clock in late chronotypes is out of sync with the work and social schedules followed by society,” Dr. van der Velde suggested. “This can lead to circadian misalignment, which we know can lead to metabolic disturbances and ultimately type 2 diabetes.”
Might trying to adjust chronotype earlier in life have an effect on risk?
“Chronotype, as measured via midpoint of sleep, does change a lot in the first 30 years or so in life,” he said. “After that it seems to stabilize. I suppose that if you adapt an intermediate or early chronotype around the age of 30 years, this will help to maintain an earlier chronotype later in life, although we cannot answer this from our study.”
Nevertheless, with respect to T2D risk, “chronotype is likely only part of the puzzle,” he noted.
“People with late chronotypes typically eat late in the evening, and this has also been associated with adverse metabolic effects. At this stage, we do not know if a person changes his/her chronotype that this will also lead to metabolic improvements. More research is needed before we can make recommendations regarding chronotype and timing of other lifestyle behaviors.”
Commenting on the study, Gianluca Iacobellis, MD, PhD, director of the University of Miami Hospital Diabetes Service, Coral Gables, Florida, said: “Interesting data. Altering the physiological circadian rhythm can affect the complex hormonal system — including cortisol, ghrelin, leptin, and serotonin — that regulates insulin sensitivity, glucose, and blood pressure control. The night owl may become more insulin resistant and therefore at higher risk of developing diabetes.”
Like Dr. van der Velde, he noted that “late sleep may be associated with night binging that can cause weight gain and ultimately obesity, further increasing the risk of diabetes.”
Dr. Iacobellis’s group recently showed that vital exhaustion, which is characterized by fatigue and loss of vigor, is associated with a higher cardiovascular risk for and markers of visceral adiposity.
“Abnormal circadian rhythms can be easily associated with vital exhaustion,” he said. Therefore, night owls with more visceral than peripheral fat accumulation might also be at higher cardiometabolic risk through that mechanism.
“However environmental factors and family history can play an important role too,” he added.
Regardless of the mechanisms involved, “preventive actions should be taken to educate teenagers and individuals at higher risk to have healthy sleep habits,” Dr. Iacobellis concluded.
No information regarding funding was provided; Dr. van der Velde and Dr. Iacobellis reported no conflicts of interest.
A version of this article first appeared on Medscape.com.
MADRID — research presented at the annual meeting of the European Association for the Study of Diabetes suggested.
In the study, night owls were almost 50% more likely to develop T2D than those who went to sleep earlier.
“The magnitude of this risk was more than I expected, [although] residual confounding may have occurred,” said Jeroen van der Velde, PhD, Leiden University Medical Center in the Netherlands, who presented the study.
“Late chronotype has previously been associated with unhealthy lifestyle and overweight or obesity and, subsequently, cardiometabolic diseases,” he said in an interview. However, although the current study found that individuals with late chronotypes did indeed have larger waists and more visceral fat, “we (and others) believe that lifestyle cannot fully explain the relation between late chronotype and metabolic disorders.”
“In addition,” he noted, “previous studies that observed that late chronotype is associated with overweight or obesity mainly focused on body mass index [BMI]. However, BMI alone does not provide accurate information regarding fat distribution in the body. People with similar BMI may have different underlying fat distribution, and this may be more relevant than BMI for metabolic risk.”
The researchers examined associations between chronotype and BMI, waist circumference, visceral fat, liver fat, and the risk for T2D in a middle-aged population from the Netherlands Epidemiology of Obesity study. Among the 5026 participants, the mean age was 56 years, 54% were women, and mean BMI was 30.
Using data from the study, the study investigators calculated the midpoint of sleep (MPS) and divided participants into three chronotypes: Early MPS < 2:30 PM (20% of participants); intermediate MPS 2:30–4:00 PM (reference category; 60% of participants); and late MPS ≥ 4:00 PM (20% of participants). BMI and waist circumference were measured in all participants, and visceral fat and liver fat were measured in 1576 participants using MRI scans and MR spectroscopy, respectively.
During a median follow-up of 6.6 years, 225 participants were diagnosed with T2D. After adjustment for age, sex, education, physical activity, smoking, alcohol intake, diet quality, sleep quality and duration, and total body fat, participants with a late chronotype had a 46% increased risk for T2D.
Further, those with a late chronotype had 0.7 higher BMI, 1.9-cm larger waist circumference, 7 cm2 more visceral fat, and 14% more liver fat.
Body Clock Out of Sync?
“Late chronotype was associated with increased ectopic body fat and with an increased risk of T2D independent of lifestyle factors and is an emerging risk factor for metabolic diseases,” the researchers concluded.
“A likely explanation is that the circadian rhythm or body clock in late chronotypes is out of sync with the work and social schedules followed by society,” Dr. van der Velde suggested. “This can lead to circadian misalignment, which we know can lead to metabolic disturbances and ultimately type 2 diabetes.”
Might trying to adjust chronotype earlier in life have an effect on risk?
“Chronotype, as measured via midpoint of sleep, does change a lot in the first 30 years or so in life,” he said. “After that it seems to stabilize. I suppose that if you adapt an intermediate or early chronotype around the age of 30 years, this will help to maintain an earlier chronotype later in life, although we cannot answer this from our study.”
Nevertheless, with respect to T2D risk, “chronotype is likely only part of the puzzle,” he noted.
“People with late chronotypes typically eat late in the evening, and this has also been associated with adverse metabolic effects. At this stage, we do not know if a person changes his/her chronotype that this will also lead to metabolic improvements. More research is needed before we can make recommendations regarding chronotype and timing of other lifestyle behaviors.”
Commenting on the study, Gianluca Iacobellis, MD, PhD, director of the University of Miami Hospital Diabetes Service, Coral Gables, Florida, said: “Interesting data. Altering the physiological circadian rhythm can affect the complex hormonal system — including cortisol, ghrelin, leptin, and serotonin — that regulates insulin sensitivity, glucose, and blood pressure control. The night owl may become more insulin resistant and therefore at higher risk of developing diabetes.”
Like Dr. van der Velde, he noted that “late sleep may be associated with night binging that can cause weight gain and ultimately obesity, further increasing the risk of diabetes.”
Dr. Iacobellis’s group recently showed that vital exhaustion, which is characterized by fatigue and loss of vigor, is associated with a higher cardiovascular risk for and markers of visceral adiposity.
“Abnormal circadian rhythms can be easily associated with vital exhaustion,” he said. Therefore, night owls with more visceral than peripheral fat accumulation might also be at higher cardiometabolic risk through that mechanism.
“However environmental factors and family history can play an important role too,” he added.
Regardless of the mechanisms involved, “preventive actions should be taken to educate teenagers and individuals at higher risk to have healthy sleep habits,” Dr. Iacobellis concluded.
No information regarding funding was provided; Dr. van der Velde and Dr. Iacobellis reported no conflicts of interest.
A version of this article first appeared on Medscape.com.
FROM EASD 2024
Debate: Should Patients With CLL Take Breaks From Targeted Therapies?
At the annual meeting of the Society of Hematologic Oncology, two hematologist-oncologists — Inhye Ahn, MD, of Dana-Farber Cancer Institute in Boston, Massachusetts, and Kerry A. Rogers, MD, of Ohio State University in Columbus — faced off in a debate. Ahn said the drugs can indeed be discontinued, while Rogers argued against stopping the medications.
“When I talk to my own patient about standard of care options in CLL, I use the analogy of a marathon and a sprint,” Dr. Ahn said. A marathon refers to continuous treatment with Bruton’s kinase inhibitors given daily for years, while the sprint refers to the combination of venetoclax with an anti-CD20 monoclonal antibody given over 12 cycles for the frontline regimen and 2 years for refractory CLL.
“I tell them these are both considered very efficacious regimens and well tolerated, one is by IV [the venetoclax regimen] and the other isn’t [Bruton’s kinase inhibitors], and they have different toxicity profile. I ask them what would you do? The most common question that I get from my patient is, ‘why would anyone do a marathon?’ ”
It’s not solely the length of treatment that’s important, Dr. Ahn said, as toxicities from the long-term use of Bruton’s kinase inhibitors build up over time and can lead to hypertension, arrhythmia, and sudden cardiac death.
In addition, she said, infections can occur, as well as hampered vaccine response, an important risk in the era of the COVID-19 pandemic. The cost of the drugs is high and adds up over time, and continuous use can boost resistance.
Is there a way to turn the marathon of Bruton’s kinase inhibitor use into a sprint without hurting patients? The answer is yes, through temporary discontinuation, Dr. Ahn said, although she cautioned that early cessation could lead to disease flare. “We dipped into our own database of 84 CLL patients treated with ibrutinib, and our conclusion was that temporary dose interruption or dose reduction did not impact progression-free survival”
Moving forward, she said, “more research is needed to define the optimal regimen that would lead to treatment cessation, the optimal patient population, who would benefit most from the cessation strategy, treatment duration, and how we define success.” For her part, Dr. Rogers argued that the continuous use of Bruton’s kinase inhibitors is “really the most effective treatment we have in CLL.”
It’s clear that “responses deepen with continued treatment,” Dr. Rogers said, noting that remission times grow over years of treatment. She highlighted a 2022 study of patients with CLL who took ibrutinib that found complete remission or complete remission with incomplete hematologic recovery was 7% at 12 months and 34% at 7 years. When patients quit taking the drugs, “you don’t get to maximize your patient’s response to this treatment.”
Dr. Rogers also noted that the RESONATE-2 trial found that ibrutinib is linked to the longest median progression-free survival of any CLL treatment at 8.9 years. “That really struck me a very effective initial therapy.”
Indeed, “when you’re offering someone initial therapy with a Bruton’s kinase inhibitor as a continuous treatment strategy, you can tell people that they can expect a normal lifespan with this approach. That’s extremely important when you’re talking to patients about whether they might want to alter their leukemia treatment.”
Finally, she noted that discontinuation of ibrutinib was linked to shorter survival in early research. “This was the first suggestion that discontinuation is not good.”
Dr. Rogers said that discontinuing the drugs is sometimes necessary because of adverse events, but patients can “certainly switch to a more tolerable Bruton’s kinase inhibitor. With the options available today, that should be a strategy that’s considered.”
Audience members at the debate were invited to respond to a live online survey about whether Bruton’s kinase inhibitors can be discontinued. Among 49 respondents, most (52.3%) said no, 42.8% said yes, and the rest were undecided/other.
Disclosures for the speakers were not provided. Dr. Ahn disclosed consulting for BeiGene and AstraZeneca. Dr. Rogers disclosed receiving research funding from Genentech, AbbVie, Janssen, and Novartis; consulting for AstraZeneca, BeiGene, Janssen, Pharmacyclics, AbbVie, Genentech, and LOXO@Lilly; and receiving travel funding from AstraZeneca.
A version of this article appeared on Medscape.com.
At the annual meeting of the Society of Hematologic Oncology, two hematologist-oncologists — Inhye Ahn, MD, of Dana-Farber Cancer Institute in Boston, Massachusetts, and Kerry A. Rogers, MD, of Ohio State University in Columbus — faced off in a debate. Ahn said the drugs can indeed be discontinued, while Rogers argued against stopping the medications.
“When I talk to my own patient about standard of care options in CLL, I use the analogy of a marathon and a sprint,” Dr. Ahn said. A marathon refers to continuous treatment with Bruton’s kinase inhibitors given daily for years, while the sprint refers to the combination of venetoclax with an anti-CD20 monoclonal antibody given over 12 cycles for the frontline regimen and 2 years for refractory CLL.
“I tell them these are both considered very efficacious regimens and well tolerated, one is by IV [the venetoclax regimen] and the other isn’t [Bruton’s kinase inhibitors], and they have different toxicity profile. I ask them what would you do? The most common question that I get from my patient is, ‘why would anyone do a marathon?’ ”
It’s not solely the length of treatment that’s important, Dr. Ahn said, as toxicities from the long-term use of Bruton’s kinase inhibitors build up over time and can lead to hypertension, arrhythmia, and sudden cardiac death.
In addition, she said, infections can occur, as well as hampered vaccine response, an important risk in the era of the COVID-19 pandemic. The cost of the drugs is high and adds up over time, and continuous use can boost resistance.
Is there a way to turn the marathon of Bruton’s kinase inhibitor use into a sprint without hurting patients? The answer is yes, through temporary discontinuation, Dr. Ahn said, although she cautioned that early cessation could lead to disease flare. “We dipped into our own database of 84 CLL patients treated with ibrutinib, and our conclusion was that temporary dose interruption or dose reduction did not impact progression-free survival”
Moving forward, she said, “more research is needed to define the optimal regimen that would lead to treatment cessation, the optimal patient population, who would benefit most from the cessation strategy, treatment duration, and how we define success.” For her part, Dr. Rogers argued that the continuous use of Bruton’s kinase inhibitors is “really the most effective treatment we have in CLL.”
It’s clear that “responses deepen with continued treatment,” Dr. Rogers said, noting that remission times grow over years of treatment. She highlighted a 2022 study of patients with CLL who took ibrutinib that found complete remission or complete remission with incomplete hematologic recovery was 7% at 12 months and 34% at 7 years. When patients quit taking the drugs, “you don’t get to maximize your patient’s response to this treatment.”
Dr. Rogers also noted that the RESONATE-2 trial found that ibrutinib is linked to the longest median progression-free survival of any CLL treatment at 8.9 years. “That really struck me a very effective initial therapy.”
Indeed, “when you’re offering someone initial therapy with a Bruton’s kinase inhibitor as a continuous treatment strategy, you can tell people that they can expect a normal lifespan with this approach. That’s extremely important when you’re talking to patients about whether they might want to alter their leukemia treatment.”
Finally, she noted that discontinuation of ibrutinib was linked to shorter survival in early research. “This was the first suggestion that discontinuation is not good.”
Dr. Rogers said that discontinuing the drugs is sometimes necessary because of adverse events, but patients can “certainly switch to a more tolerable Bruton’s kinase inhibitor. With the options available today, that should be a strategy that’s considered.”
Audience members at the debate were invited to respond to a live online survey about whether Bruton’s kinase inhibitors can be discontinued. Among 49 respondents, most (52.3%) said no, 42.8% said yes, and the rest were undecided/other.
Disclosures for the speakers were not provided. Dr. Ahn disclosed consulting for BeiGene and AstraZeneca. Dr. Rogers disclosed receiving research funding from Genentech, AbbVie, Janssen, and Novartis; consulting for AstraZeneca, BeiGene, Janssen, Pharmacyclics, AbbVie, Genentech, and LOXO@Lilly; and receiving travel funding from AstraZeneca.
A version of this article appeared on Medscape.com.
At the annual meeting of the Society of Hematologic Oncology, two hematologist-oncologists — Inhye Ahn, MD, of Dana-Farber Cancer Institute in Boston, Massachusetts, and Kerry A. Rogers, MD, of Ohio State University in Columbus — faced off in a debate. Ahn said the drugs can indeed be discontinued, while Rogers argued against stopping the medications.
“When I talk to my own patient about standard of care options in CLL, I use the analogy of a marathon and a sprint,” Dr. Ahn said. A marathon refers to continuous treatment with Bruton’s kinase inhibitors given daily for years, while the sprint refers to the combination of venetoclax with an anti-CD20 monoclonal antibody given over 12 cycles for the frontline regimen and 2 years for refractory CLL.
“I tell them these are both considered very efficacious regimens and well tolerated, one is by IV [the venetoclax regimen] and the other isn’t [Bruton’s kinase inhibitors], and they have different toxicity profile. I ask them what would you do? The most common question that I get from my patient is, ‘why would anyone do a marathon?’ ”
It’s not solely the length of treatment that’s important, Dr. Ahn said, as toxicities from the long-term use of Bruton’s kinase inhibitors build up over time and can lead to hypertension, arrhythmia, and sudden cardiac death.
In addition, she said, infections can occur, as well as hampered vaccine response, an important risk in the era of the COVID-19 pandemic. The cost of the drugs is high and adds up over time, and continuous use can boost resistance.
Is there a way to turn the marathon of Bruton’s kinase inhibitor use into a sprint without hurting patients? The answer is yes, through temporary discontinuation, Dr. Ahn said, although she cautioned that early cessation could lead to disease flare. “We dipped into our own database of 84 CLL patients treated with ibrutinib, and our conclusion was that temporary dose interruption or dose reduction did not impact progression-free survival”
Moving forward, she said, “more research is needed to define the optimal regimen that would lead to treatment cessation, the optimal patient population, who would benefit most from the cessation strategy, treatment duration, and how we define success.” For her part, Dr. Rogers argued that the continuous use of Bruton’s kinase inhibitors is “really the most effective treatment we have in CLL.”
It’s clear that “responses deepen with continued treatment,” Dr. Rogers said, noting that remission times grow over years of treatment. She highlighted a 2022 study of patients with CLL who took ibrutinib that found complete remission or complete remission with incomplete hematologic recovery was 7% at 12 months and 34% at 7 years. When patients quit taking the drugs, “you don’t get to maximize your patient’s response to this treatment.”
Dr. Rogers also noted that the RESONATE-2 trial found that ibrutinib is linked to the longest median progression-free survival of any CLL treatment at 8.9 years. “That really struck me a very effective initial therapy.”
Indeed, “when you’re offering someone initial therapy with a Bruton’s kinase inhibitor as a continuous treatment strategy, you can tell people that they can expect a normal lifespan with this approach. That’s extremely important when you’re talking to patients about whether they might want to alter their leukemia treatment.”
Finally, she noted that discontinuation of ibrutinib was linked to shorter survival in early research. “This was the first suggestion that discontinuation is not good.”
Dr. Rogers said that discontinuing the drugs is sometimes necessary because of adverse events, but patients can “certainly switch to a more tolerable Bruton’s kinase inhibitor. With the options available today, that should be a strategy that’s considered.”
Audience members at the debate were invited to respond to a live online survey about whether Bruton’s kinase inhibitors can be discontinued. Among 49 respondents, most (52.3%) said no, 42.8% said yes, and the rest were undecided/other.
Disclosures for the speakers were not provided. Dr. Ahn disclosed consulting for BeiGene and AstraZeneca. Dr. Rogers disclosed receiving research funding from Genentech, AbbVie, Janssen, and Novartis; consulting for AstraZeneca, BeiGene, Janssen, Pharmacyclics, AbbVie, Genentech, and LOXO@Lilly; and receiving travel funding from AstraZeneca.
A version of this article appeared on Medscape.com.
FROM SOHO 2024
Top DEI Topics to Incorporate Into Dermatology Residency Training: An Electronic Delphi Consensus Study
Diversity, equity, and inclusion (DEI) programs seek to improve dermatologic education and clinical care for an increasingly diverse patient population as well as to recruit and sustain a physician workforce that reflects the diversity of the patients they serve.1,2 In dermatology, only 4.2% and 3.0% of practicing dermatologists self-identify as being of Hispanic and African American ethnicity, respectively, compared with 18.5% and 13.4% of the general population, respectively.3 Creating an educational system that works to meet the goals of DEI is essential to improve health outcomes and address disparities. The lack of robust DEI-related curricula during residency training may limit the ability of practicing dermatologists to provide comprehensive and culturally sensitive care. It has been shown that racial concordance between patients and physicians has a positive impact on patient satisfaction by fostering a trusting patient-physician relationship.4
It is the responsibility of all dermatologists to create an environment where patients from any background can feel comfortable, which can be cultivated by establishing patient-centered communication and cultural humility.5 These skills can be strengthened via the implementation of DEI-related curricula during residency training. Augmenting exposure of these topics during training can optimize the delivery of dermatologic care by providing residents with the tools and confidence needed to care for patients of culturally diverse backgrounds. Enhancing DEI education is crucial to not only improve the recognition and treatment of dermatologic conditions in all skin and hair types but also to minimize misconceptions, stigma, health disparities, and discrimination faced by historically marginalized communities. Creating a culture of inclusion is of paramount importance to build successful relationships with patients and colleagues of culturally diverse backgrounds.6
There are multiple efforts underway to increase DEI education across the field of dermatology, including the development of DEI task forces in professional organizations and societies that serve to expand DEI-related research, mentorship, and education. The American Academy of Dermatology has been leading efforts to create a curriculum focused on skin of color, particularly addressing inadequate educational training on how dermatologic conditions manifest in this population.7 The Skin of Color Society has similar efforts underway and is developing a speakers bureau to give leading experts a platform to lecture dermatology trainees as well as patient and community audiences on various topics in skin of color.8 These are just 2 of many professional dermatology organizations that are advocating for expanded education on DEI; however, consistently integrating DEI-related topics into dermatology residency training curricula remains a gap in pedagogy. To identify the DEI-related topics of greatest relevance to the dermatology resident curricula, we implemented a modified electronic Delphi (e-Delphi) consensus process to provide standardized recommendations.
Methods
A 2-round modified e-Delphi method was utilized (Figure). An initial list of potential curricular topics was formulated by an expert panel consisting of 5 dermatologists from the Association of Professors of Dermatology DEI subcommittee and the American Academy of Dermatology Diversity Task Force (A.M.A., S.B., R.V., S.D.W., J.I.S.). Initial topics were selected via several meetings among the panel members to discuss existing DEI concerns and issues that were deemed relevant due to education gaps in residency training. The list of topics was further expanded with recommendations obtained via an email sent to dermatology program directors on the Association of Professors of Dermatology listserve, which solicited voluntary participation of academic dermatologists, including program directors and dermatology residents.
There were 2 voting rounds, with each round consisting of questions scored on a Likert scale ranging from 1 to 5 (1=not essential, 2=probably not essential, 3=neutral, 4=probably essential, 5=definitely essential). The inclusion criteria to classify a topic as necessary for integration into the dermatology residency curriculum included 95% (18/19) or more of respondents rating the topic as probably essential or definitely essential; if more than 90% (17/19) of respondents rated the topic as probably essential or definitely essential and less than 10% (2/19) rated it as not essential or probably not essential, the topic was still included as part of the suggested curriculum. Topics that received ratings of probably essential or definitely essential by less than 80% (15/19) of respondents were removed from consideration. The topics that did not meet inclusion or exclusion criteria during the first round of voting were refined by the e-Delphi steering committee (V.S.E-C. and F-A.R.) based on open-ended feedback from the voting group provided at the end of the survey and subsequently passed to the second round of voting.
Results
Participants—A total of 19 respondents participated in both voting rounds, the majority (80% [15/19]) of whom were program directors or dermatologists affiliated with academia or development of DEI education; the remaining 20% [4/19]) were dermatology residents.
Open-Ended Feedback—Voting group members were able to provide open-ended feedback for each of the sets of topics after the survey, which the steering committee utilized to modify the topics as needed for the final voting round. For example, “structural racism/discrimination” was originally mentioned as a topic, but several participants suggested including specific types of racism; therefore, the wording was changed to “racism: types, definitions” to encompass broader definitions and types of racism.
Survey Results—Two genres of topics were surveyed in each voting round: clinical and nonclinical. Participants voted on a total of 61 topics, with 23 ultimately selected in the final list of consensus curricular topics. Of those, 9 were clinical and 14 nonclinical. All topics deemed necessary for inclusion in residency curricula are presented in eTables 1 and 2.
During the first round of voting, the e-Delphi panel reached a consensus to include the following 17 topics as essential to dermatology residency training (along with the percentage of voters who classified them as probably essential or definitely essential): how to mitigate bias in clinical and workplace settings (100% [40/40]); social determinants of health-related disparities in dermatology (100% [40/40]); hairstyling practices across different hair textures (100% [40/40]); definitions and examples of microaggressions (97.50% [39/40]); definition, background, and types of bias (97.50% [39/40]); manifestations of bias in the clinical setting (97.44% [38/39]); racial and ethnic disparities in dermatology (97.44% [38/39]); keloids (97.37% [37/38]); differences in dermoscopic presentations in skin of color (97.30% [36/37]); skin cancer in patients with skin of color (97.30% [36/37]); disparities due to bias (95.00% [38/40]); how to apply cultural humility and safety to patients of different cultural backgrounds (94.87% [37/40]); best practices in providing care to patients with limited English proficiency (94.87% [37/40]); hair loss in patients with textured hair (94.74% [36/38]); pseudofolliculitis barbae and acne keloidalis nuchae (94.60% [35/37]); disparities regarding people experiencing homelessness (92.31% [36/39]); and definitions and types of racism and other forms of discrimination (92.31% [36/39]). eTable 1 provides a list of suggested resources to incorporate these topics into the educational components of residency curricula. The resources provided were not part of the voting process, and they were not considered in the consensus analysis; they are included here as suggested educational catalysts.
During the second round of voting, 25 topics were evaluated. Of those, the following 6 topics were proposed to be included as essential in residency training: differences in prevalence and presentation of common inflammatory disorders (100% [29/29]); manifestations of bias in the learning environment (96.55%); antiracist action and how to decrease the effects of structural racism in clinical and educational settings (96.55% [28/29]); diversity of images in dermatology education (96.55% [28/29]); pigmentary disorders and their psychological effects (96.55% [28/29]); and LGBTQ (lesbian, gay, bisexual, transgender, and queer) dermatologic health care (96.55% [28/29]). eTable 2 includes these topics as well as suggested resources to help incorporate them into training.
Comment
This study utilized a modified e-Delphi technique to identify relevant clinical and nonclinical DEI topics that should be incorporated into dermatology residency curricula. The panel members reached a consensus for 9 clinical DEI-related topics. The respondents agreed that the topics related to skin and hair conditions in patients with skin of color as well as textured hair were crucial to residency education. Skin cancer, hair loss, pseudofolliculitis barbae, acne keloidalis nuchae, keloids, pigmentary disorders, and their varying presentations in patients with skin of color were among the recommended topics. The panel also recommended educating residents on the variable visual presentations of inflammatory conditions in skin of color. Addressing the needs of diverse patients—for example, those belonging to the LGBTQ community—also was deemed important for inclusion.
The remaining 14 chosen topics were nonclinical items addressing concepts such as bias and health care disparities as well as cultural humility and safety.9 Cultural humility and safety focus on developing cultural awareness by creating a safe setting for patients rather than encouraging power relationships between them and their physicians. Various topics related to racism also were recommended to be included in residency curricula, including education on implementation of antiracist action in the workplace.
Many of the nonclinical topics are intertwined; for instance, learning about health care disparities in patients with limited English proficiency allows for improved best practices in delivering care to patients from this population. The first step in overcoming bias and subsequent disparities is acknowledging how the perpetuation of bias leads to disparities after being taught tools to recognize it.
Our group’s guidance on DEI topics should help dermatology residency program leaders as they design and refine program curricula. There are multiple avenues for incorporating education on these topics, including lectures, interactive workshops, role-playing sessions, book or journal clubs, and discussion circles. Many of these topics/programs may already be included in programs’ didactic curricula, which would minimize the burden of finding space to educate on these topics. Institutional cultural change is key to ensuring truly diverse, equitable, and inclusive workplaces. Educating tomorrow’s dermatologists on these topics is a first step toward achieving that cultural change.
Limitations—A limitation of this e-Delphi survey is that only a selection of experts in this field was included. Additionally, we were concerned that the Likert scale format and the bar we set for inclusion and exclusion may have failed to adequately capture participants’ nuanced opinions. As such, participants were able to provide open-ended feedback, and suggestions for alternate wording or other changes were considered by the steering committee. Finally, inclusion recommendations identified in this survey were developed specifically for US dermatology residents.
Conclusion
In this e-Delphi consensus assessment of DEI-related topics, we recommend the inclusion of 23 topics into dermatology residency program curricula to improve medical training and the patient-physician relationship as well as to create better health outcomes. We also provide specific sample resource recommendations in eTables 1 and 2 to facilitate inclusion of these topics into residency curricula across the country.
- US Census Bureau projections show a slower growing, older, more diverse nation a half century from now. News release. US Census Bureau. December 12, 2012. Accessed August 14, 2024. https://www.census.gov/newsroom/releases/archives/population/cb12243.html#:~:text=12%2C%202012,U.S.%20Census%20Bureau%20Projections%20Show%20a%20Slower%20Growing%2C%20Older%2C%20More,by%20the%20U.S.%20Census%20Bureau
- Lopez S, Lourido JO, Lim HW, et al. The call to action to increase racial and ethnic diversity in dermatology: a retrospective, cross-sectional study to monitor progress. J Am Acad Dermatol. 2020;86:E121-E123. doi:10.1016/j.jaad.2021.10.011
- El-Kashlan N, Alexis A. Disparities in dermatology: a reflection. J Clin Aesthet Dermatol. 2022;15:27-29.
- Laveist TA, Nuru-Jeter A. Is doctor-patient race concordance associated with greater satisfaction with care? J Health Soc Behav. 2002;43:296-306.
- Street RL Jr, O’Malley KJ, Cooper LA, et al. Understanding concordance in patient-physician relationships: personal and ethnic dimensions of shared identity. Ann Fam Med. 2008;6:198-205. doi:10.1370/afm.821
- Dadrass F, Bowers S, Shinkai K, et al. Diversity, equity, and inclusion in dermatology residency. Dermatol Clin. 2023;41:257-263. doi:10.1016/j.det.2022.10.006
- Diversity and the Academy. American Academy of Dermatology website. Accessed August 22, 2024. https://www.aad.org/member/career/diversity
- SOCS speaks. Skin of Color Society website. Accessed August 22, 2024. https://skinofcolorsociety.org/news-media/socs-speaks
- Solchanyk D, Ekeh O, Saffran L, et al. Integrating cultural humility into the medical education curriculum: strategies for educators. Teach Learn Med. 2021;33:554-560. doi:10.1080/10401334.2021.1877711
Diversity, equity, and inclusion (DEI) programs seek to improve dermatologic education and clinical care for an increasingly diverse patient population as well as to recruit and sustain a physician workforce that reflects the diversity of the patients they serve.1,2 In dermatology, only 4.2% and 3.0% of practicing dermatologists self-identify as being of Hispanic and African American ethnicity, respectively, compared with 18.5% and 13.4% of the general population, respectively.3 Creating an educational system that works to meet the goals of DEI is essential to improve health outcomes and address disparities. The lack of robust DEI-related curricula during residency training may limit the ability of practicing dermatologists to provide comprehensive and culturally sensitive care. It has been shown that racial concordance between patients and physicians has a positive impact on patient satisfaction by fostering a trusting patient-physician relationship.4
It is the responsibility of all dermatologists to create an environment where patients from any background can feel comfortable, which can be cultivated by establishing patient-centered communication and cultural humility.5 These skills can be strengthened via the implementation of DEI-related curricula during residency training. Augmenting exposure of these topics during training can optimize the delivery of dermatologic care by providing residents with the tools and confidence needed to care for patients of culturally diverse backgrounds. Enhancing DEI education is crucial to not only improve the recognition and treatment of dermatologic conditions in all skin and hair types but also to minimize misconceptions, stigma, health disparities, and discrimination faced by historically marginalized communities. Creating a culture of inclusion is of paramount importance to build successful relationships with patients and colleagues of culturally diverse backgrounds.6
There are multiple efforts underway to increase DEI education across the field of dermatology, including the development of DEI task forces in professional organizations and societies that serve to expand DEI-related research, mentorship, and education. The American Academy of Dermatology has been leading efforts to create a curriculum focused on skin of color, particularly addressing inadequate educational training on how dermatologic conditions manifest in this population.7 The Skin of Color Society has similar efforts underway and is developing a speakers bureau to give leading experts a platform to lecture dermatology trainees as well as patient and community audiences on various topics in skin of color.8 These are just 2 of many professional dermatology organizations that are advocating for expanded education on DEI; however, consistently integrating DEI-related topics into dermatology residency training curricula remains a gap in pedagogy. To identify the DEI-related topics of greatest relevance to the dermatology resident curricula, we implemented a modified electronic Delphi (e-Delphi) consensus process to provide standardized recommendations.
Methods
A 2-round modified e-Delphi method was utilized (Figure). An initial list of potential curricular topics was formulated by an expert panel consisting of 5 dermatologists from the Association of Professors of Dermatology DEI subcommittee and the American Academy of Dermatology Diversity Task Force (A.M.A., S.B., R.V., S.D.W., J.I.S.). Initial topics were selected via several meetings among the panel members to discuss existing DEI concerns and issues that were deemed relevant due to education gaps in residency training. The list of topics was further expanded with recommendations obtained via an email sent to dermatology program directors on the Association of Professors of Dermatology listserve, which solicited voluntary participation of academic dermatologists, including program directors and dermatology residents.

There were 2 voting rounds, with each round consisting of questions scored on a Likert scale ranging from 1 to 5 (1=not essential, 2=probably not essential, 3=neutral, 4=probably essential, 5=definitely essential). The inclusion criteria to classify a topic as necessary for integration into the dermatology residency curriculum included 95% (18/19) or more of respondents rating the topic as probably essential or definitely essential; if more than 90% (17/19) of respondents rated the topic as probably essential or definitely essential and less than 10% (2/19) rated it as not essential or probably not essential, the topic was still included as part of the suggested curriculum. Topics that received ratings of probably essential or definitely essential by less than 80% (15/19) of respondents were removed from consideration. The topics that did not meet inclusion or exclusion criteria during the first round of voting were refined by the e-Delphi steering committee (V.S.E-C. and F-A.R.) based on open-ended feedback from the voting group provided at the end of the survey and subsequently passed to the second round of voting.
Results
Participants—A total of 19 respondents participated in both voting rounds, the majority (80% [15/19]) of whom were program directors or dermatologists affiliated with academia or development of DEI education; the remaining 20% [4/19]) were dermatology residents.
Open-Ended Feedback—Voting group members were able to provide open-ended feedback for each of the sets of topics after the survey, which the steering committee utilized to modify the topics as needed for the final voting round. For example, “structural racism/discrimination” was originally mentioned as a topic, but several participants suggested including specific types of racism; therefore, the wording was changed to “racism: types, definitions” to encompass broader definitions and types of racism.
Survey Results—Two genres of topics were surveyed in each voting round: clinical and nonclinical. Participants voted on a total of 61 topics, with 23 ultimately selected in the final list of consensus curricular topics. Of those, 9 were clinical and 14 nonclinical. All topics deemed necessary for inclusion in residency curricula are presented in eTables 1 and 2.
During the first round of voting, the e-Delphi panel reached a consensus to include the following 17 topics as essential to dermatology residency training (along with the percentage of voters who classified them as probably essential or definitely essential): how to mitigate bias in clinical and workplace settings (100% [40/40]); social determinants of health-related disparities in dermatology (100% [40/40]); hairstyling practices across different hair textures (100% [40/40]); definitions and examples of microaggressions (97.50% [39/40]); definition, background, and types of bias (97.50% [39/40]); manifestations of bias in the clinical setting (97.44% [38/39]); racial and ethnic disparities in dermatology (97.44% [38/39]); keloids (97.37% [37/38]); differences in dermoscopic presentations in skin of color (97.30% [36/37]); skin cancer in patients with skin of color (97.30% [36/37]); disparities due to bias (95.00% [38/40]); how to apply cultural humility and safety to patients of different cultural backgrounds (94.87% [37/40]); best practices in providing care to patients with limited English proficiency (94.87% [37/40]); hair loss in patients with textured hair (94.74% [36/38]); pseudofolliculitis barbae and acne keloidalis nuchae (94.60% [35/37]); disparities regarding people experiencing homelessness (92.31% [36/39]); and definitions and types of racism and other forms of discrimination (92.31% [36/39]). eTable 1 provides a list of suggested resources to incorporate these topics into the educational components of residency curricula. The resources provided were not part of the voting process, and they were not considered in the consensus analysis; they are included here as suggested educational catalysts.
During the second round of voting, 25 topics were evaluated. Of those, the following 6 topics were proposed to be included as essential in residency training: differences in prevalence and presentation of common inflammatory disorders (100% [29/29]); manifestations of bias in the learning environment (96.55%); antiracist action and how to decrease the effects of structural racism in clinical and educational settings (96.55% [28/29]); diversity of images in dermatology education (96.55% [28/29]); pigmentary disorders and their psychological effects (96.55% [28/29]); and LGBTQ (lesbian, gay, bisexual, transgender, and queer) dermatologic health care (96.55% [28/29]). eTable 2 includes these topics as well as suggested resources to help incorporate them into training.
Comment
This study utilized a modified e-Delphi technique to identify relevant clinical and nonclinical DEI topics that should be incorporated into dermatology residency curricula. The panel members reached a consensus for 9 clinical DEI-related topics. The respondents agreed that the topics related to skin and hair conditions in patients with skin of color as well as textured hair were crucial to residency education. Skin cancer, hair loss, pseudofolliculitis barbae, acne keloidalis nuchae, keloids, pigmentary disorders, and their varying presentations in patients with skin of color were among the recommended topics. The panel also recommended educating residents on the variable visual presentations of inflammatory conditions in skin of color. Addressing the needs of diverse patients—for example, those belonging to the LGBTQ community—also was deemed important for inclusion.
The remaining 14 chosen topics were nonclinical items addressing concepts such as bias and health care disparities as well as cultural humility and safety.9 Cultural humility and safety focus on developing cultural awareness by creating a safe setting for patients rather than encouraging power relationships between them and their physicians. Various topics related to racism also were recommended to be included in residency curricula, including education on implementation of antiracist action in the workplace.
Many of the nonclinical topics are intertwined; for instance, learning about health care disparities in patients with limited English proficiency allows for improved best practices in delivering care to patients from this population. The first step in overcoming bias and subsequent disparities is acknowledging how the perpetuation of bias leads to disparities after being taught tools to recognize it.
Our group’s guidance on DEI topics should help dermatology residency program leaders as they design and refine program curricula. There are multiple avenues for incorporating education on these topics, including lectures, interactive workshops, role-playing sessions, book or journal clubs, and discussion circles. Many of these topics/programs may already be included in programs’ didactic curricula, which would minimize the burden of finding space to educate on these topics. Institutional cultural change is key to ensuring truly diverse, equitable, and inclusive workplaces. Educating tomorrow’s dermatologists on these topics is a first step toward achieving that cultural change.
Limitations—A limitation of this e-Delphi survey is that only a selection of experts in this field was included. Additionally, we were concerned that the Likert scale format and the bar we set for inclusion and exclusion may have failed to adequately capture participants’ nuanced opinions. As such, participants were able to provide open-ended feedback, and suggestions for alternate wording or other changes were considered by the steering committee. Finally, inclusion recommendations identified in this survey were developed specifically for US dermatology residents.
Conclusion
In this e-Delphi consensus assessment of DEI-related topics, we recommend the inclusion of 23 topics into dermatology residency program curricula to improve medical training and the patient-physician relationship as well as to create better health outcomes. We also provide specific sample resource recommendations in eTables 1 and 2 to facilitate inclusion of these topics into residency curricula across the country.
Diversity, equity, and inclusion (DEI) programs seek to improve dermatologic education and clinical care for an increasingly diverse patient population as well as to recruit and sustain a physician workforce that reflects the diversity of the patients they serve.1,2 In dermatology, only 4.2% and 3.0% of practicing dermatologists self-identify as being of Hispanic and African American ethnicity, respectively, compared with 18.5% and 13.4% of the general population, respectively.3 Creating an educational system that works to meet the goals of DEI is essential to improve health outcomes and address disparities. The lack of robust DEI-related curricula during residency training may limit the ability of practicing dermatologists to provide comprehensive and culturally sensitive care. It has been shown that racial concordance between patients and physicians has a positive impact on patient satisfaction by fostering a trusting patient-physician relationship.4
It is the responsibility of all dermatologists to create an environment where patients from any background can feel comfortable, which can be cultivated by establishing patient-centered communication and cultural humility.5 These skills can be strengthened via the implementation of DEI-related curricula during residency training. Augmenting exposure of these topics during training can optimize the delivery of dermatologic care by providing residents with the tools and confidence needed to care for patients of culturally diverse backgrounds. Enhancing DEI education is crucial to not only improve the recognition and treatment of dermatologic conditions in all skin and hair types but also to minimize misconceptions, stigma, health disparities, and discrimination faced by historically marginalized communities. Creating a culture of inclusion is of paramount importance to build successful relationships with patients and colleagues of culturally diverse backgrounds.6
There are multiple efforts underway to increase DEI education across the field of dermatology, including the development of DEI task forces in professional organizations and societies that serve to expand DEI-related research, mentorship, and education. The American Academy of Dermatology has been leading efforts to create a curriculum focused on skin of color, particularly addressing inadequate educational training on how dermatologic conditions manifest in this population.7 The Skin of Color Society has similar efforts underway and is developing a speakers bureau to give leading experts a platform to lecture dermatology trainees as well as patient and community audiences on various topics in skin of color.8 These are just 2 of many professional dermatology organizations that are advocating for expanded education on DEI; however, consistently integrating DEI-related topics into dermatology residency training curricula remains a gap in pedagogy. To identify the DEI-related topics of greatest relevance to the dermatology resident curricula, we implemented a modified electronic Delphi (e-Delphi) consensus process to provide standardized recommendations.
Methods
A 2-round modified e-Delphi method was utilized (Figure). An initial list of potential curricular topics was formulated by an expert panel consisting of 5 dermatologists from the Association of Professors of Dermatology DEI subcommittee and the American Academy of Dermatology Diversity Task Force (A.M.A., S.B., R.V., S.D.W., J.I.S.). Initial topics were selected via several meetings among the panel members to discuss existing DEI concerns and issues that were deemed relevant due to education gaps in residency training. The list of topics was further expanded with recommendations obtained via an email sent to dermatology program directors on the Association of Professors of Dermatology listserve, which solicited voluntary participation of academic dermatologists, including program directors and dermatology residents.

There were 2 voting rounds, with each round consisting of questions scored on a Likert scale ranging from 1 to 5 (1=not essential, 2=probably not essential, 3=neutral, 4=probably essential, 5=definitely essential). The inclusion criteria to classify a topic as necessary for integration into the dermatology residency curriculum included 95% (18/19) or more of respondents rating the topic as probably essential or definitely essential; if more than 90% (17/19) of respondents rated the topic as probably essential or definitely essential and less than 10% (2/19) rated it as not essential or probably not essential, the topic was still included as part of the suggested curriculum. Topics that received ratings of probably essential or definitely essential by less than 80% (15/19) of respondents were removed from consideration. The topics that did not meet inclusion or exclusion criteria during the first round of voting were refined by the e-Delphi steering committee (V.S.E-C. and F-A.R.) based on open-ended feedback from the voting group provided at the end of the survey and subsequently passed to the second round of voting.
Results
Participants—A total of 19 respondents participated in both voting rounds, the majority (80% [15/19]) of whom were program directors or dermatologists affiliated with academia or development of DEI education; the remaining 20% [4/19]) were dermatology residents.
Open-Ended Feedback—Voting group members were able to provide open-ended feedback for each of the sets of topics after the survey, which the steering committee utilized to modify the topics as needed for the final voting round. For example, “structural racism/discrimination” was originally mentioned as a topic, but several participants suggested including specific types of racism; therefore, the wording was changed to “racism: types, definitions” to encompass broader definitions and types of racism.
Survey Results—Two genres of topics were surveyed in each voting round: clinical and nonclinical. Participants voted on a total of 61 topics, with 23 ultimately selected in the final list of consensus curricular topics. Of those, 9 were clinical and 14 nonclinical. All topics deemed necessary for inclusion in residency curricula are presented in eTables 1 and 2.
During the first round of voting, the e-Delphi panel reached a consensus to include the following 17 topics as essential to dermatology residency training (along with the percentage of voters who classified them as probably essential or definitely essential): how to mitigate bias in clinical and workplace settings (100% [40/40]); social determinants of health-related disparities in dermatology (100% [40/40]); hairstyling practices across different hair textures (100% [40/40]); definitions and examples of microaggressions (97.50% [39/40]); definition, background, and types of bias (97.50% [39/40]); manifestations of bias in the clinical setting (97.44% [38/39]); racial and ethnic disparities in dermatology (97.44% [38/39]); keloids (97.37% [37/38]); differences in dermoscopic presentations in skin of color (97.30% [36/37]); skin cancer in patients with skin of color (97.30% [36/37]); disparities due to bias (95.00% [38/40]); how to apply cultural humility and safety to patients of different cultural backgrounds (94.87% [37/40]); best practices in providing care to patients with limited English proficiency (94.87% [37/40]); hair loss in patients with textured hair (94.74% [36/38]); pseudofolliculitis barbae and acne keloidalis nuchae (94.60% [35/37]); disparities regarding people experiencing homelessness (92.31% [36/39]); and definitions and types of racism and other forms of discrimination (92.31% [36/39]). eTable 1 provides a list of suggested resources to incorporate these topics into the educational components of residency curricula. The resources provided were not part of the voting process, and they were not considered in the consensus analysis; they are included here as suggested educational catalysts.
During the second round of voting, 25 topics were evaluated. Of those, the following 6 topics were proposed to be included as essential in residency training: differences in prevalence and presentation of common inflammatory disorders (100% [29/29]); manifestations of bias in the learning environment (96.55%); antiracist action and how to decrease the effects of structural racism in clinical and educational settings (96.55% [28/29]); diversity of images in dermatology education (96.55% [28/29]); pigmentary disorders and their psychological effects (96.55% [28/29]); and LGBTQ (lesbian, gay, bisexual, transgender, and queer) dermatologic health care (96.55% [28/29]). eTable 2 includes these topics as well as suggested resources to help incorporate them into training.
Comment
This study utilized a modified e-Delphi technique to identify relevant clinical and nonclinical DEI topics that should be incorporated into dermatology residency curricula. The panel members reached a consensus for 9 clinical DEI-related topics. The respondents agreed that the topics related to skin and hair conditions in patients with skin of color as well as textured hair were crucial to residency education. Skin cancer, hair loss, pseudofolliculitis barbae, acne keloidalis nuchae, keloids, pigmentary disorders, and their varying presentations in patients with skin of color were among the recommended topics. The panel also recommended educating residents on the variable visual presentations of inflammatory conditions in skin of color. Addressing the needs of diverse patients—for example, those belonging to the LGBTQ community—also was deemed important for inclusion.
The remaining 14 chosen topics were nonclinical items addressing concepts such as bias and health care disparities as well as cultural humility and safety.9 Cultural humility and safety focus on developing cultural awareness by creating a safe setting for patients rather than encouraging power relationships between them and their physicians. Various topics related to racism also were recommended to be included in residency curricula, including education on implementation of antiracist action in the workplace.
Many of the nonclinical topics are intertwined; for instance, learning about health care disparities in patients with limited English proficiency allows for improved best practices in delivering care to patients from this population. The first step in overcoming bias and subsequent disparities is acknowledging how the perpetuation of bias leads to disparities after being taught tools to recognize it.
Our group’s guidance on DEI topics should help dermatology residency program leaders as they design and refine program curricula. There are multiple avenues for incorporating education on these topics, including lectures, interactive workshops, role-playing sessions, book or journal clubs, and discussion circles. Many of these topics/programs may already be included in programs’ didactic curricula, which would minimize the burden of finding space to educate on these topics. Institutional cultural change is key to ensuring truly diverse, equitable, and inclusive workplaces. Educating tomorrow’s dermatologists on these topics is a first step toward achieving that cultural change.
Limitations—A limitation of this e-Delphi survey is that only a selection of experts in this field was included. Additionally, we were concerned that the Likert scale format and the bar we set for inclusion and exclusion may have failed to adequately capture participants’ nuanced opinions. As such, participants were able to provide open-ended feedback, and suggestions for alternate wording or other changes were considered by the steering committee. Finally, inclusion recommendations identified in this survey were developed specifically for US dermatology residents.
Conclusion
In this e-Delphi consensus assessment of DEI-related topics, we recommend the inclusion of 23 topics into dermatology residency program curricula to improve medical training and the patient-physician relationship as well as to create better health outcomes. We also provide specific sample resource recommendations in eTables 1 and 2 to facilitate inclusion of these topics into residency curricula across the country.
- US Census Bureau projections show a slower growing, older, more diverse nation a half century from now. News release. US Census Bureau. December 12, 2012. Accessed August 14, 2024. https://www.census.gov/newsroom/releases/archives/population/cb12243.html#:~:text=12%2C%202012,U.S.%20Census%20Bureau%20Projections%20Show%20a%20Slower%20Growing%2C%20Older%2C%20More,by%20the%20U.S.%20Census%20Bureau
- Lopez S, Lourido JO, Lim HW, et al. The call to action to increase racial and ethnic diversity in dermatology: a retrospective, cross-sectional study to monitor progress. J Am Acad Dermatol. 2020;86:E121-E123. doi:10.1016/j.jaad.2021.10.011
- El-Kashlan N, Alexis A. Disparities in dermatology: a reflection. J Clin Aesthet Dermatol. 2022;15:27-29.
- Laveist TA, Nuru-Jeter A. Is doctor-patient race concordance associated with greater satisfaction with care? J Health Soc Behav. 2002;43:296-306.
- Street RL Jr, O’Malley KJ, Cooper LA, et al. Understanding concordance in patient-physician relationships: personal and ethnic dimensions of shared identity. Ann Fam Med. 2008;6:198-205. doi:10.1370/afm.821
- Dadrass F, Bowers S, Shinkai K, et al. Diversity, equity, and inclusion in dermatology residency. Dermatol Clin. 2023;41:257-263. doi:10.1016/j.det.2022.10.006
- Diversity and the Academy. American Academy of Dermatology website. Accessed August 22, 2024. https://www.aad.org/member/career/diversity
- SOCS speaks. Skin of Color Society website. Accessed August 22, 2024. https://skinofcolorsociety.org/news-media/socs-speaks
- Solchanyk D, Ekeh O, Saffran L, et al. Integrating cultural humility into the medical education curriculum: strategies for educators. Teach Learn Med. 2021;33:554-560. doi:10.1080/10401334.2021.1877711
- US Census Bureau projections show a slower growing, older, more diverse nation a half century from now. News release. US Census Bureau. December 12, 2012. Accessed August 14, 2024. https://www.census.gov/newsroom/releases/archives/population/cb12243.html#:~:text=12%2C%202012,U.S.%20Census%20Bureau%20Projections%20Show%20a%20Slower%20Growing%2C%20Older%2C%20More,by%20the%20U.S.%20Census%20Bureau
- Lopez S, Lourido JO, Lim HW, et al. The call to action to increase racial and ethnic diversity in dermatology: a retrospective, cross-sectional study to monitor progress. J Am Acad Dermatol. 2020;86:E121-E123. doi:10.1016/j.jaad.2021.10.011
- El-Kashlan N, Alexis A. Disparities in dermatology: a reflection. J Clin Aesthet Dermatol. 2022;15:27-29.
- Laveist TA, Nuru-Jeter A. Is doctor-patient race concordance associated with greater satisfaction with care? J Health Soc Behav. 2002;43:296-306.
- Street RL Jr, O’Malley KJ, Cooper LA, et al. Understanding concordance in patient-physician relationships: personal and ethnic dimensions of shared identity. Ann Fam Med. 2008;6:198-205. doi:10.1370/afm.821
- Dadrass F, Bowers S, Shinkai K, et al. Diversity, equity, and inclusion in dermatology residency. Dermatol Clin. 2023;41:257-263. doi:10.1016/j.det.2022.10.006
- Diversity and the Academy. American Academy of Dermatology website. Accessed August 22, 2024. https://www.aad.org/member/career/diversity
- SOCS speaks. Skin of Color Society website. Accessed August 22, 2024. https://skinofcolorsociety.org/news-media/socs-speaks
- Solchanyk D, Ekeh O, Saffran L, et al. Integrating cultural humility into the medical education curriculum: strategies for educators. Teach Learn Med. 2021;33:554-560. doi:10.1080/10401334.2021.1877711
PRACTICE POINTS
- Advancing curricula related to diversity, equity, and inclusion in dermatology training can improve health outcomes, address health care workforce disparities, and enhance clinical care for diverse patient populations.
- Education on patient-centered communication, cultural humility, and the impact of social determinants of health results in dermatology residents who are better equipped with the necessary tools to effectively care for patients from diverse backgrounds.
Five Key Measures to Ensure a Quality Colonoscopy
, a list that, for the first time, includes adequate bowel preparation and sessile serrated lesion detection rate (SSLDR).
“Endoscopy teams now have an updated set of guidelines which can be used to enhance the quality of their colonoscopies and should certainly use these current quality measures to ‘raise the bar’ on behalf of their patients,” task force member Nicholas J. Shaheen, MD, MPH, Division of Gastroenterology and Hepatology, The University of North Carolina at Chapel Hill, said in a statement.
The task force published the recommendations online August 21 in The American Journal of Gastroenterology and in Gastrointestinal Endoscopy. It represents the third iteration of the ACG/ASGE quality indicators on colonoscopy recommendations and incorporates new evidence published since 2015.
“The last set of quality indicators from this group was 9 years ago. Since then, there has been a tremendous amount of new data published in colonoscopy quality,” Ziad F. Gellad, MD, MPH, professor of medicine, Duke University Medical Center, Durham, North Carolina, said in an interview.
“Keeping up with that data is a challenge, and so guidelines such as these are important in helping clinicians synthesize data on quality of care and implement best practices,” said Dr. Gellad, who was not involved with the task force.
Two New Priority Indicators
The task force identified 15 quality indicators, divided into preprocedure, intraprocedure, and postprocedure. It includes five “priority” indicators — two of which are new.
One is the rate of adequate bowel preparation, preferably defined as a Boston Bowel Preparation Scale score ≥ 2 in each of three colon segments or by description of the preparation as excellent, good, or adequate. It has a performance target > 90%.
“Inadequate bowel preparation substantially increases the cost of colonoscopy delivery and creates risk and inconvenience for patients, thus warranting a ranking as a priority indicator,” the task force wrote.
Dr. Gellad explained that the addition of this priority indicator is “notable because it highlights the importance of bowel prep in high-quality colonoscopy. It also shifts more of the responsibility of bowel prep from the patient to the practice.”
The second new quality indicator is the SSLDR, which was selected due to its ability to contribute to cancer prevention.
Based on available evidence, the task force recommends a current minimum threshold for the SSLDR of 6%. “This is expected to be revised upward as evidence of increasing detection occurs,” they wrote.
Dr. Gellad said the addition of SSLDR is “an important advance in these recommendations. We know that serrated adenomas are a precursor for colorectal cancer and that the detection of these subtle lesions is variable.
“Providing a benchmark encourages practices to measure the detection of serrated adenomas and intervene when rates are below benchmarks. Prior to these benchmarks, it was difficult to know where to peg our expectations,” Dr. Gellad added.
Changes to the Adenoma Detection Rate (ADR)
The ADR remains a priority indicator in the update, albeit with changes.
To keep the ADR measurement consistent with current screening guidelines, the task force now recommends that the ADR be measured starting at age 45 rather than 50 years.
“ADR plays a critical role in evaluating the performance of the colonoscopists,” task force lead Douglas K. Rex, MD, a gastroenterologist at Indiana University School of Medicine in Indianapolis, said in the statement.
“It is recommended that ADR calculations include screening, surveillance, and diagnostic colonoscopy but exclude indications of a positive noncolonoscopy screening test and therapeutic procedures for resection or treatment of known neoplasia, genetic cancer syndromes, and inflammatory bowel disease,” Dr. Rex explained.
The task force recommends a minimum ADR threshold of 35% (40% in men and 30% in women) and that colonoscopists with ADRs below 35% “undertake remedial measures to improve and to achieve acceptable performance.”
Additional Priorities
The cecal intubation rate (CIR) — the percentage of patients undergoing colonoscopy with intact colons who have full intubation of the cecum with photo documentation of cecal landmarks — remains a priority quality indicator and has a performance target ≥ 95%.
“A trained colonoscopist should achieve a high CIR with a very high level of safety,” the task force wrote. “Low CIRs have been associated with higher PCCRC [postcolonoscopy colorectal cancer] rates.”
The final priority indicator is the rate of using recommended screening and surveillance intervals, which carries a performance target ≥ 90%.
“We recommend that quality improvement efforts initially focus on high-priority indicators and then progress to other indicators once it is ascertained that endoscopists are performing above recommended thresholds, either at baseline or after corrective interventions,” the task force wrote.
“The priority indicators are absolutely important for practices to implement,” Dr. Gellad said.
“There is compelling evidence that these measures are correlated with clinically important outcomes, particularly ADR,” he added. “Many practices already capture this data, and the changes in ADR calculation make measurement less burdensome. Hopefully, this will encourage more practices to collect and report these measures.”
Dr. Rex is a consultant for Olympus, Boston Scientific, Braintree Laboratories, Norgine, GI Supply, Medtronic, and Acacia Pharmaceuticals; receives research support from Olympus, Medivators, Erbe USA, and Braintree Laboratories; and is a shareholder in Satisfai Health. Dr. Shaheen had no relevant disclosures. Dr. Gellad has consulted for Merck & Co. and Novo Nordisk and is a cofounder of Higgs Boson.
A version of this article first appeared on Medscape.com.
, a list that, for the first time, includes adequate bowel preparation and sessile serrated lesion detection rate (SSLDR).
“Endoscopy teams now have an updated set of guidelines which can be used to enhance the quality of their colonoscopies and should certainly use these current quality measures to ‘raise the bar’ on behalf of their patients,” task force member Nicholas J. Shaheen, MD, MPH, Division of Gastroenterology and Hepatology, The University of North Carolina at Chapel Hill, said in a statement.
The task force published the recommendations online August 21 in The American Journal of Gastroenterology and in Gastrointestinal Endoscopy. It represents the third iteration of the ACG/ASGE quality indicators on colonoscopy recommendations and incorporates new evidence published since 2015.
“The last set of quality indicators from this group was 9 years ago. Since then, there has been a tremendous amount of new data published in colonoscopy quality,” Ziad F. Gellad, MD, MPH, professor of medicine, Duke University Medical Center, Durham, North Carolina, said in an interview.
“Keeping up with that data is a challenge, and so guidelines such as these are important in helping clinicians synthesize data on quality of care and implement best practices,” said Dr. Gellad, who was not involved with the task force.
Two New Priority Indicators
The task force identified 15 quality indicators, divided into preprocedure, intraprocedure, and postprocedure. It includes five “priority” indicators — two of which are new.
One is the rate of adequate bowel preparation, preferably defined as a Boston Bowel Preparation Scale score ≥ 2 in each of three colon segments or by description of the preparation as excellent, good, or adequate. It has a performance target > 90%.
“Inadequate bowel preparation substantially increases the cost of colonoscopy delivery and creates risk and inconvenience for patients, thus warranting a ranking as a priority indicator,” the task force wrote.
Dr. Gellad explained that the addition of this priority indicator is “notable because it highlights the importance of bowel prep in high-quality colonoscopy. It also shifts more of the responsibility of bowel prep from the patient to the practice.”
The second new quality indicator is the SSLDR, which was selected due to its ability to contribute to cancer prevention.
Based on available evidence, the task force recommends a current minimum threshold for the SSLDR of 6%. “This is expected to be revised upward as evidence of increasing detection occurs,” they wrote.
Dr. Gellad said the addition of SSLDR is “an important advance in these recommendations. We know that serrated adenomas are a precursor for colorectal cancer and that the detection of these subtle lesions is variable.
“Providing a benchmark encourages practices to measure the detection of serrated adenomas and intervene when rates are below benchmarks. Prior to these benchmarks, it was difficult to know where to peg our expectations,” Dr. Gellad added.
Changes to the Adenoma Detection Rate (ADR)
The ADR remains a priority indicator in the update, albeit with changes.
To keep the ADR measurement consistent with current screening guidelines, the task force now recommends that the ADR be measured starting at age 45 rather than 50 years.
“ADR plays a critical role in evaluating the performance of the colonoscopists,” task force lead Douglas K. Rex, MD, a gastroenterologist at Indiana University School of Medicine in Indianapolis, said in the statement.
“It is recommended that ADR calculations include screening, surveillance, and diagnostic colonoscopy but exclude indications of a positive noncolonoscopy screening test and therapeutic procedures for resection or treatment of known neoplasia, genetic cancer syndromes, and inflammatory bowel disease,” Dr. Rex explained.
The task force recommends a minimum ADR threshold of 35% (40% in men and 30% in women) and that colonoscopists with ADRs below 35% “undertake remedial measures to improve and to achieve acceptable performance.”
Additional Priorities
The cecal intubation rate (CIR) — the percentage of patients undergoing colonoscopy with intact colons who have full intubation of the cecum with photo documentation of cecal landmarks — remains a priority quality indicator and has a performance target ≥ 95%.
“A trained colonoscopist should achieve a high CIR with a very high level of safety,” the task force wrote. “Low CIRs have been associated with higher PCCRC [postcolonoscopy colorectal cancer] rates.”
The final priority indicator is the rate of using recommended screening and surveillance intervals, which carries a performance target ≥ 90%.
“We recommend that quality improvement efforts initially focus on high-priority indicators and then progress to other indicators once it is ascertained that endoscopists are performing above recommended thresholds, either at baseline or after corrective interventions,” the task force wrote.
“The priority indicators are absolutely important for practices to implement,” Dr. Gellad said.
“There is compelling evidence that these measures are correlated with clinically important outcomes, particularly ADR,” he added. “Many practices already capture this data, and the changes in ADR calculation make measurement less burdensome. Hopefully, this will encourage more practices to collect and report these measures.”
Dr. Rex is a consultant for Olympus, Boston Scientific, Braintree Laboratories, Norgine, GI Supply, Medtronic, and Acacia Pharmaceuticals; receives research support from Olympus, Medivators, Erbe USA, and Braintree Laboratories; and is a shareholder in Satisfai Health. Dr. Shaheen had no relevant disclosures. Dr. Gellad has consulted for Merck & Co. and Novo Nordisk and is a cofounder of Higgs Boson.
A version of this article first appeared on Medscape.com.
, a list that, for the first time, includes adequate bowel preparation and sessile serrated lesion detection rate (SSLDR).
“Endoscopy teams now have an updated set of guidelines which can be used to enhance the quality of their colonoscopies and should certainly use these current quality measures to ‘raise the bar’ on behalf of their patients,” task force member Nicholas J. Shaheen, MD, MPH, Division of Gastroenterology and Hepatology, The University of North Carolina at Chapel Hill, said in a statement.
The task force published the recommendations online August 21 in The American Journal of Gastroenterology and in Gastrointestinal Endoscopy. It represents the third iteration of the ACG/ASGE quality indicators on colonoscopy recommendations and incorporates new evidence published since 2015.
“The last set of quality indicators from this group was 9 years ago. Since then, there has been a tremendous amount of new data published in colonoscopy quality,” Ziad F. Gellad, MD, MPH, professor of medicine, Duke University Medical Center, Durham, North Carolina, said in an interview.
“Keeping up with that data is a challenge, and so guidelines such as these are important in helping clinicians synthesize data on quality of care and implement best practices,” said Dr. Gellad, who was not involved with the task force.
Two New Priority Indicators
The task force identified 15 quality indicators, divided into preprocedure, intraprocedure, and postprocedure. It includes five “priority” indicators — two of which are new.
One is the rate of adequate bowel preparation, preferably defined as a Boston Bowel Preparation Scale score ≥ 2 in each of three colon segments or by description of the preparation as excellent, good, or adequate. It has a performance target > 90%.
“Inadequate bowel preparation substantially increases the cost of colonoscopy delivery and creates risk and inconvenience for patients, thus warranting a ranking as a priority indicator,” the task force wrote.
Dr. Gellad explained that the addition of this priority indicator is “notable because it highlights the importance of bowel prep in high-quality colonoscopy. It also shifts more of the responsibility of bowel prep from the patient to the practice.”
The second new quality indicator is the SSLDR, which was selected due to its ability to contribute to cancer prevention.
Based on available evidence, the task force recommends a current minimum threshold for the SSLDR of 6%. “This is expected to be revised upward as evidence of increasing detection occurs,” they wrote.
Dr. Gellad said the addition of SSLDR is “an important advance in these recommendations. We know that serrated adenomas are a precursor for colorectal cancer and that the detection of these subtle lesions is variable.
“Providing a benchmark encourages practices to measure the detection of serrated adenomas and intervene when rates are below benchmarks. Prior to these benchmarks, it was difficult to know where to peg our expectations,” Dr. Gellad added.
Changes to the Adenoma Detection Rate (ADR)
The ADR remains a priority indicator in the update, albeit with changes.
To keep the ADR measurement consistent with current screening guidelines, the task force now recommends that the ADR be measured starting at age 45 rather than 50 years.
“ADR plays a critical role in evaluating the performance of the colonoscopists,” task force lead Douglas K. Rex, MD, a gastroenterologist at Indiana University School of Medicine in Indianapolis, said in the statement.
“It is recommended that ADR calculations include screening, surveillance, and diagnostic colonoscopy but exclude indications of a positive noncolonoscopy screening test and therapeutic procedures for resection or treatment of known neoplasia, genetic cancer syndromes, and inflammatory bowel disease,” Dr. Rex explained.
The task force recommends a minimum ADR threshold of 35% (40% in men and 30% in women) and that colonoscopists with ADRs below 35% “undertake remedial measures to improve and to achieve acceptable performance.”
Additional Priorities
The cecal intubation rate (CIR) — the percentage of patients undergoing colonoscopy with intact colons who have full intubation of the cecum with photo documentation of cecal landmarks — remains a priority quality indicator and has a performance target ≥ 95%.
“A trained colonoscopist should achieve a high CIR with a very high level of safety,” the task force wrote. “Low CIRs have been associated with higher PCCRC [postcolonoscopy colorectal cancer] rates.”
The final priority indicator is the rate of using recommended screening and surveillance intervals, which carries a performance target ≥ 90%.
“We recommend that quality improvement efforts initially focus on high-priority indicators and then progress to other indicators once it is ascertained that endoscopists are performing above recommended thresholds, either at baseline or after corrective interventions,” the task force wrote.
“The priority indicators are absolutely important for practices to implement,” Dr. Gellad said.
“There is compelling evidence that these measures are correlated with clinically important outcomes, particularly ADR,” he added. “Many practices already capture this data, and the changes in ADR calculation make measurement less burdensome. Hopefully, this will encourage more practices to collect and report these measures.”
Dr. Rex is a consultant for Olympus, Boston Scientific, Braintree Laboratories, Norgine, GI Supply, Medtronic, and Acacia Pharmaceuticals; receives research support from Olympus, Medivators, Erbe USA, and Braintree Laboratories; and is a shareholder in Satisfai Health. Dr. Shaheen had no relevant disclosures. Dr. Gellad has consulted for Merck & Co. and Novo Nordisk and is a cofounder of Higgs Boson.
A version of this article first appeared on Medscape.com.
Hormone Therapy Can Benefit Women into Their 80s
Hormone therapy (HT) can help women manage menopause symptoms into their 80s and the reasons are varied, according to a retrospective analysis being presented at the annual meeting of The Menopause Society.
“It’s important to know that this is a preselected group of women who had no contraindications to continuing their hormone therapy,” senior author Wendy Wolfman, MD, director of the Menopause Clinic and The Premature Ovarian Insufficiency Clinic at Mount Sinai Hospital in Toronto, Ontario, Canada, said in an interview. “They had the initiation of hormone therapy closer to menopause and carried on their hormones. We followed them for a long time and basically saw no real concerns about taking the hormones and the patients did very well. It’s important to emphasize this was not the new initiation of hormone therapy in elderly women.”
She said that, in her large tertiary referral center, “I still see patients who are referred who are told that they have to stop their hormones after 5 years based on a false assumption. Everybody ages at different rates and everybody has different risk factors.”
About 70%-80% of women experience menopause symptoms that restrict quality of life and productivity, the authors noted. HT has consistently been the most effective means for managing many of the side effects, especially hot flashes.
Hot flashes last on average 7-11 years. But they continue in up to 40% of women in their 60s and 10%-15% in their 70s, the authors wrote.
The analysis included more than 100 women in Canada older than 65 who continue to use HT and explored the motivations of the women to use them.
The average age of the women was 71 and nearly 8% were age 80 or older. The mean age for starting HT was 52 years and the women continued HT for an average 18 years, though 42% used it regularly for more than 20 years. Most of the women (nearly 88%) used a transdermal form of estrogen; only 12% used oral estrogen pills. Fewer than 5% of participants used synthetic progestins.
Controlling hot flashes was the No. 1 reason the women continued HT beyond age 65 (55%), followed by a desire for a better quality of life (29%), and to reduce chronic pain and arthritis symptoms (7%).
Some adverse effects were reported – postmenopausal bleeding was the most common – but no strokes, myocardial infarctions, or uterine cancers were documented.
More than one fourth (26.4%) of the women tried stopping HT once, but 87% reported that the return of hot flashes was the main reason to restart HT.
In addition, “many women choose to continue hormone therapy long term for relief of nonvasomotor symptoms, preservation of bone density, and a desire to benefit from potential long-term cardiovascular protection,” said Lauren F. Streicher, MD, Professor of Obstetrics and Gynecology at Feinberg School of Medicine at Northwestern University in Chicago, who was not part of the research.
In 2022, The Menopause Society position statement on hormone therapy acknowledged that, on an individual basis, it is appropriate for women to continue hormone therapy long term with counseling on benefits and risks.
“However, few studies have evaluated the outcomes of using hormone therapy for more than 10 years, and individual motivation for doing so,” Dr. Streicher said. She pointed to a study that analyzed the insurance records of more than 10 million women who continued their HT past the age of 65 and reassuringly found that there were significant risk reductions in all-cause mortality, breast cancer, lung cancer, colorectal cancer, heart failure, venous thromboembolism, atrial fibrillation, acute myocardial infarction, and dementia. In that study, however, the reasons women chose to continue hormone therapy were not specified.
“In this retrospective Canadian study,” she noted, “the outcomes were again reassuring, with no increase in strokes, myocardial infarctions, or uterine cancers. The reasons cited for continuing hormone therapy were not just to treat ongoing vasomotor symptoms, but also other menopause symptoms such as musculoskeletal aches and pains, and overall quality of life.
Dr. Streicher said that, while long-term longitudinal studies are needed to make definitive recommendations, “It is reassuring that women who choose to extend hormone therapy can safely do so. It is irresponsible, cruel, and nonsensical to continue to make blanket statements that hormone therapy should be discontinued based on age or years of use and commit women to enduring symptoms and depriving them of possible long-term benefits.”
Dr. Streicher gives lectures for Midi Health and owns Sermonix stock. Dr. Wolfman has been on the advisory boards for many pharmaceutical companies. She is the past president of the Canadian Menopause Society and is on the board of the International Menopause Society.
Hormone therapy (HT) can help women manage menopause symptoms into their 80s and the reasons are varied, according to a retrospective analysis being presented at the annual meeting of The Menopause Society.
“It’s important to know that this is a preselected group of women who had no contraindications to continuing their hormone therapy,” senior author Wendy Wolfman, MD, director of the Menopause Clinic and The Premature Ovarian Insufficiency Clinic at Mount Sinai Hospital in Toronto, Ontario, Canada, said in an interview. “They had the initiation of hormone therapy closer to menopause and carried on their hormones. We followed them for a long time and basically saw no real concerns about taking the hormones and the patients did very well. It’s important to emphasize this was not the new initiation of hormone therapy in elderly women.”
She said that, in her large tertiary referral center, “I still see patients who are referred who are told that they have to stop their hormones after 5 years based on a false assumption. Everybody ages at different rates and everybody has different risk factors.”
About 70%-80% of women experience menopause symptoms that restrict quality of life and productivity, the authors noted. HT has consistently been the most effective means for managing many of the side effects, especially hot flashes.
Hot flashes last on average 7-11 years. But they continue in up to 40% of women in their 60s and 10%-15% in their 70s, the authors wrote.
The analysis included more than 100 women in Canada older than 65 who continue to use HT and explored the motivations of the women to use them.
The average age of the women was 71 and nearly 8% were age 80 or older. The mean age for starting HT was 52 years and the women continued HT for an average 18 years, though 42% used it regularly for more than 20 years. Most of the women (nearly 88%) used a transdermal form of estrogen; only 12% used oral estrogen pills. Fewer than 5% of participants used synthetic progestins.
Controlling hot flashes was the No. 1 reason the women continued HT beyond age 65 (55%), followed by a desire for a better quality of life (29%), and to reduce chronic pain and arthritis symptoms (7%).
Some adverse effects were reported – postmenopausal bleeding was the most common – but no strokes, myocardial infarctions, or uterine cancers were documented.
More than one fourth (26.4%) of the women tried stopping HT once, but 87% reported that the return of hot flashes was the main reason to restart HT.
In addition, “many women choose to continue hormone therapy long term for relief of nonvasomotor symptoms, preservation of bone density, and a desire to benefit from potential long-term cardiovascular protection,” said Lauren F. Streicher, MD, Professor of Obstetrics and Gynecology at Feinberg School of Medicine at Northwestern University in Chicago, who was not part of the research.
In 2022, The Menopause Society position statement on hormone therapy acknowledged that, on an individual basis, it is appropriate for women to continue hormone therapy long term with counseling on benefits and risks.
“However, few studies have evaluated the outcomes of using hormone therapy for more than 10 years, and individual motivation for doing so,” Dr. Streicher said. She pointed to a study that analyzed the insurance records of more than 10 million women who continued their HT past the age of 65 and reassuringly found that there were significant risk reductions in all-cause mortality, breast cancer, lung cancer, colorectal cancer, heart failure, venous thromboembolism, atrial fibrillation, acute myocardial infarction, and dementia. In that study, however, the reasons women chose to continue hormone therapy were not specified.
“In this retrospective Canadian study,” she noted, “the outcomes were again reassuring, with no increase in strokes, myocardial infarctions, or uterine cancers. The reasons cited for continuing hormone therapy were not just to treat ongoing vasomotor symptoms, but also other menopause symptoms such as musculoskeletal aches and pains, and overall quality of life.
Dr. Streicher said that, while long-term longitudinal studies are needed to make definitive recommendations, “It is reassuring that women who choose to extend hormone therapy can safely do so. It is irresponsible, cruel, and nonsensical to continue to make blanket statements that hormone therapy should be discontinued based on age or years of use and commit women to enduring symptoms and depriving them of possible long-term benefits.”
Dr. Streicher gives lectures for Midi Health and owns Sermonix stock. Dr. Wolfman has been on the advisory boards for many pharmaceutical companies. She is the past president of the Canadian Menopause Society and is on the board of the International Menopause Society.
Hormone therapy (HT) can help women manage menopause symptoms into their 80s and the reasons are varied, according to a retrospective analysis being presented at the annual meeting of The Menopause Society.
“It’s important to know that this is a preselected group of women who had no contraindications to continuing their hormone therapy,” senior author Wendy Wolfman, MD, director of the Menopause Clinic and The Premature Ovarian Insufficiency Clinic at Mount Sinai Hospital in Toronto, Ontario, Canada, said in an interview. “They had the initiation of hormone therapy closer to menopause and carried on their hormones. We followed them for a long time and basically saw no real concerns about taking the hormones and the patients did very well. It’s important to emphasize this was not the new initiation of hormone therapy in elderly women.”
She said that, in her large tertiary referral center, “I still see patients who are referred who are told that they have to stop their hormones after 5 years based on a false assumption. Everybody ages at different rates and everybody has different risk factors.”
About 70%-80% of women experience menopause symptoms that restrict quality of life and productivity, the authors noted. HT has consistently been the most effective means for managing many of the side effects, especially hot flashes.
Hot flashes last on average 7-11 years. But they continue in up to 40% of women in their 60s and 10%-15% in their 70s, the authors wrote.
The analysis included more than 100 women in Canada older than 65 who continue to use HT and explored the motivations of the women to use them.
The average age of the women was 71 and nearly 8% were age 80 or older. The mean age for starting HT was 52 years and the women continued HT for an average 18 years, though 42% used it regularly for more than 20 years. Most of the women (nearly 88%) used a transdermal form of estrogen; only 12% used oral estrogen pills. Fewer than 5% of participants used synthetic progestins.
Controlling hot flashes was the No. 1 reason the women continued HT beyond age 65 (55%), followed by a desire for a better quality of life (29%), and to reduce chronic pain and arthritis symptoms (7%).
Some adverse effects were reported – postmenopausal bleeding was the most common – but no strokes, myocardial infarctions, or uterine cancers were documented.
More than one fourth (26.4%) of the women tried stopping HT once, but 87% reported that the return of hot flashes was the main reason to restart HT.
In addition, “many women choose to continue hormone therapy long term for relief of nonvasomotor symptoms, preservation of bone density, and a desire to benefit from potential long-term cardiovascular protection,” said Lauren F. Streicher, MD, Professor of Obstetrics and Gynecology at Feinberg School of Medicine at Northwestern University in Chicago, who was not part of the research.
In 2022, The Menopause Society position statement on hormone therapy acknowledged that, on an individual basis, it is appropriate for women to continue hormone therapy long term with counseling on benefits and risks.
“However, few studies have evaluated the outcomes of using hormone therapy for more than 10 years, and individual motivation for doing so,” Dr. Streicher said. She pointed to a study that analyzed the insurance records of more than 10 million women who continued their HT past the age of 65 and reassuringly found that there were significant risk reductions in all-cause mortality, breast cancer, lung cancer, colorectal cancer, heart failure, venous thromboembolism, atrial fibrillation, acute myocardial infarction, and dementia. In that study, however, the reasons women chose to continue hormone therapy were not specified.
“In this retrospective Canadian study,” she noted, “the outcomes were again reassuring, with no increase in strokes, myocardial infarctions, or uterine cancers. The reasons cited for continuing hormone therapy were not just to treat ongoing vasomotor symptoms, but also other menopause symptoms such as musculoskeletal aches and pains, and overall quality of life.
Dr. Streicher said that, while long-term longitudinal studies are needed to make definitive recommendations, “It is reassuring that women who choose to extend hormone therapy can safely do so. It is irresponsible, cruel, and nonsensical to continue to make blanket statements that hormone therapy should be discontinued based on age or years of use and commit women to enduring symptoms and depriving them of possible long-term benefits.”
Dr. Streicher gives lectures for Midi Health and owns Sermonix stock. Dr. Wolfman has been on the advisory boards for many pharmaceutical companies. She is the past president of the Canadian Menopause Society and is on the board of the International Menopause Society.
FROM THE MENOPAUSE SOCIETY 2024
Melasma Risk Factors: A Matched Cohort Study Using Data From the All of Us Research Program
To the Editor:
Melasma (also known as chloasma) is characterized by symmetric hyperpigmented patches affecting sun-exposed areas. Women commonly develop this condition during pregnancy, suggesting a connection between melasma and increased female sex hormone levels.1 Other hypothesized risk factors include sun exposure, genetic susceptibility, estrogen and/or progesterone therapy, and thyroid abnormalities but have not been corroborated.2 Treatment options are limited because the pathogenesis is poorly understood; thus, we aimed to analyze melasma risk factors using a national database with a nested case-control approach.
We conducted a matched case-control study using the Registered Tier dataset (version 7) from the National Institute of Health’s All of Us Research Program (https://allofus.nih.gov/), which is available to authorized users through the program’s Researcher Workbench and includes more than 413,000 total participants enrolled from May 1, 2018, through July 1, 2022. Cases included patients 18 years and older with a diagnosis of melasma (International Classification of Diseases, Tenth Revision, Clinical Modification code L81.1 [Chloasma]; concept ID 4264234 [Chloasma]; and Systematized Nomenclature of Medicine [SNOMED] code 36209000 [Chloasma]), and controls without a diagnosis of melasma were matched in a 1:10 ratio based on age, sex, and self-reported race. Concept IDs and SNOMED codes were used to identify individuals in each cohort with a diagnosis of alcohol dependence (concept IDs 433753, 435243, 4218106; SNOMED codes 15167005, 66590003, 7200002), depression (concept ID 440383; SNOMED code 35489007), hypothyroidism (concept ID 140673; SNOMED code 40930008), hyperthyroidism (concept ID 4142479; SNOMED code 34486009), anxiety (concept IDs 441542, 442077, 434613; SNOMED codes 48694002, 197480006, 21897009), tobacco dependence (concept IDs 37109023, 437264, 4099811; SNOMED codes 16077091000119107, 89765005, 191887008), or obesity (concept IDs 433736 and 434005; SNOMED codes 414916001 and 238136002), or with a history of radiation therapy (concept IDs 4085340, 4311117, 4061844, 4029715; SNOMED codes 24803000, 85983004, 200861004, 108290001) or hormonal medications containing estrogen and/or progesterone, including oral medications and implants (concept IDs 21602445, 40254009, 21602514, 21603814, 19049228, 21602529, 1549080, 1551673, 1549254, 21602472, 21602446, 21602450, 21602515, 21602566, 21602473, 21602567, 21602488, 21602585, 1596779, 1586808, 21602524). In our case cohort, diagnoses and exposures to treatments were only considered for analysis if they occurred prior to melasma diagnosis.
Multivariate logistic regression was performed to calculate odds ratios and P values between melasma and each comorbidity or exposure to the treatments specified. Statistical significance was set at P<.05.
We identified 744 melasma cases (mean age, 55.20 years; 95.43% female; 12.10% Black) and 7440 controls with similar demographics (ie, age, sex, race/ethnicity) between groups (all P>.05 [Table 1]). Patients with a melasma diagnosis were more likely to have a pre-existing diagnosis of depression (OR, 1.87; 95% CI, 1.51-2.31 [P<.001]) or hypothyroidism (OR, 1.31; 95% CI, 1.04-1.65 [P<.05]), or a history of radiation therapy (OR, 19.08; 95% CI, 10.20-35.69 [P<.001]) and/or estrogen and/or progesterone therapy (OR, 2.01; 95% CI, 1.69-2.40 [P<.001]) prior to melasma diagnosis. A diagnosis of anxiety prior to melasma diagnosis trended toward an association with melasma (P=.067). Pre-existing alcohol dependence, obesity, and hyperthyroidism were not associated with melasma (P=.98, P=.28, and P=.29, respectively). A diagnosis of tobacco dependence was associated with a decreased melasma risk (OR, 0.53, 95% CI, 0.37-0.76)[P<.001])(Table 2).
Our study results suggest that pre-existing depression was a risk factor for subsequent melasma diagnosis. Depression may exacerbate stress, leading to increased activation of the hypothalamic-pituitary-adrenal axis as well as increased levels of cortisol and adrenocorticotropic hormone, which subsequently act on melanocytes to increase melanogenesis.3 A retrospective study of 254 participants, including 127 with melasma, showed that increased melasma severity was associated with higher rates of depression (P=.002)2; however, the risk for melasma following a depression diagnosis has not been reported.
Our results also showed that hypothyroidism was associated with an increased risk for melasma. On a cellular level, hypothyroidism can cause systemic inflammation, potentailly leading to increased stress and melanogenesis via activation of the hypothalamic-pituitary-adrenal axis.4 These findings are similar to a systematic review and meta-analysis reporting increased thyroid-stimulating hormone, anti–thyroid peroxidase, and antithyroglobulin antibody levels associated with increased melasma risk (mean difference between cases and controls, 0.33 [95% CI, 0.18-0.47]; pooled association, P=.020; mean difference between cases and controls, 0.28 [95% CI, 0.01-0.55], respectively).5
Patients in our cohort who had a history of radiation therapy were 19 times more likely to develop melasma, similar to findings of a survey-based study of 421 breast cancer survivors in which 336 (79.81%) reported hyperpigmentation in irradiated areas.6 Patients in our cohort who had a history of estrogen and/or progesterone therapy were 2 times more likely to develop melasma, similar to a case-control study of 207 patients with melasma and 207 controls that showed combined oral contraceptives increased risk for melasma (OR, 1.23 [95% CI, 1.08-1.41; P<.01).3
Tobacco use is not a well-known protective factor against melasma. Prior studies have indicated that tobacco smoking activates melanocytes via the Wnt/β-Catenin pathway, leading to hyperpigmentation.7 Although exposure to cigarette smoke decreases angiogenesis and would more likely lead to hyperpigmentation, nicotine exposure has been shown to increase angiogenesis, which could lead to increased blood flow and partially explain the protection against melasma demonstrated in our cohort.8 Future studies are needed to explore this relationship.
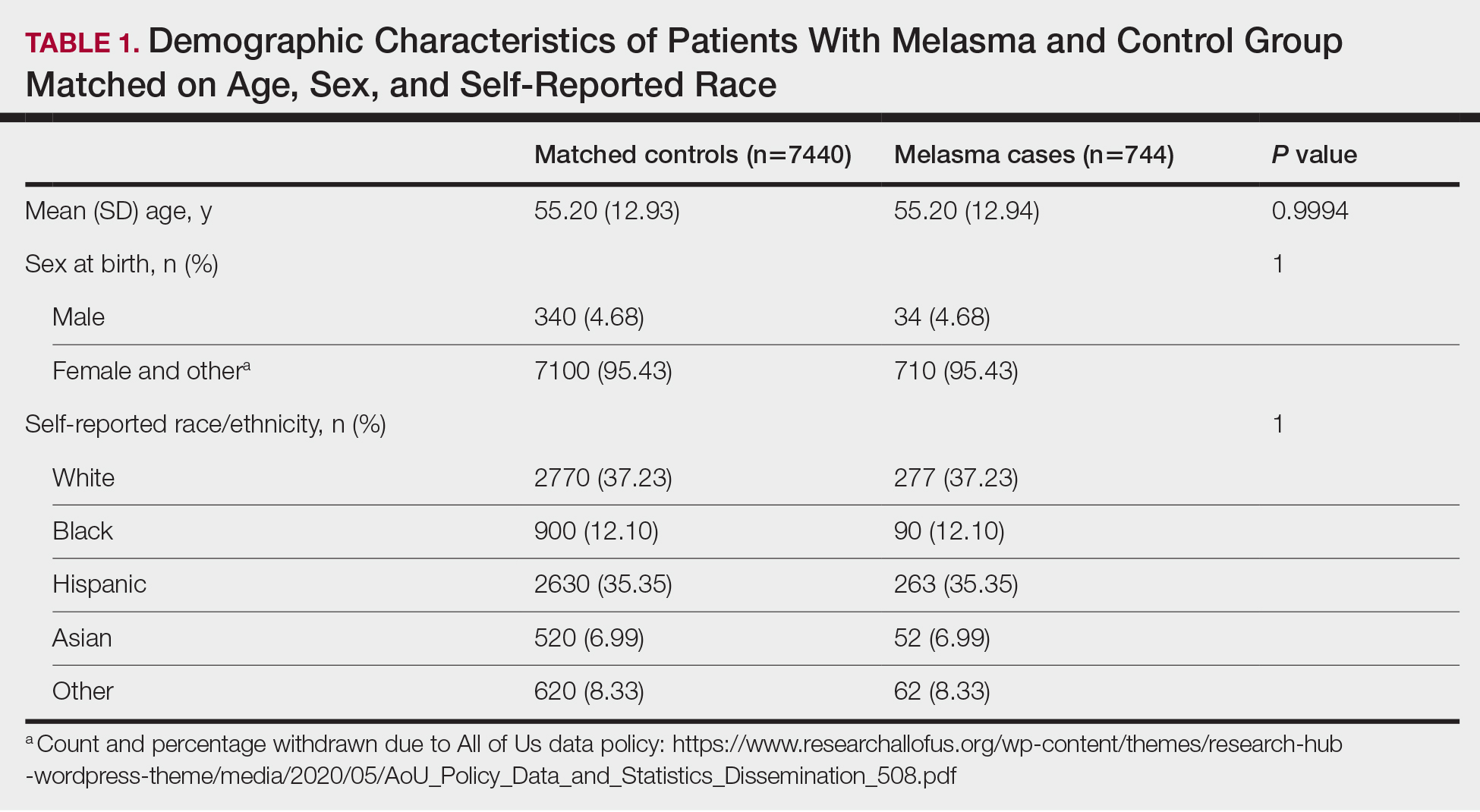
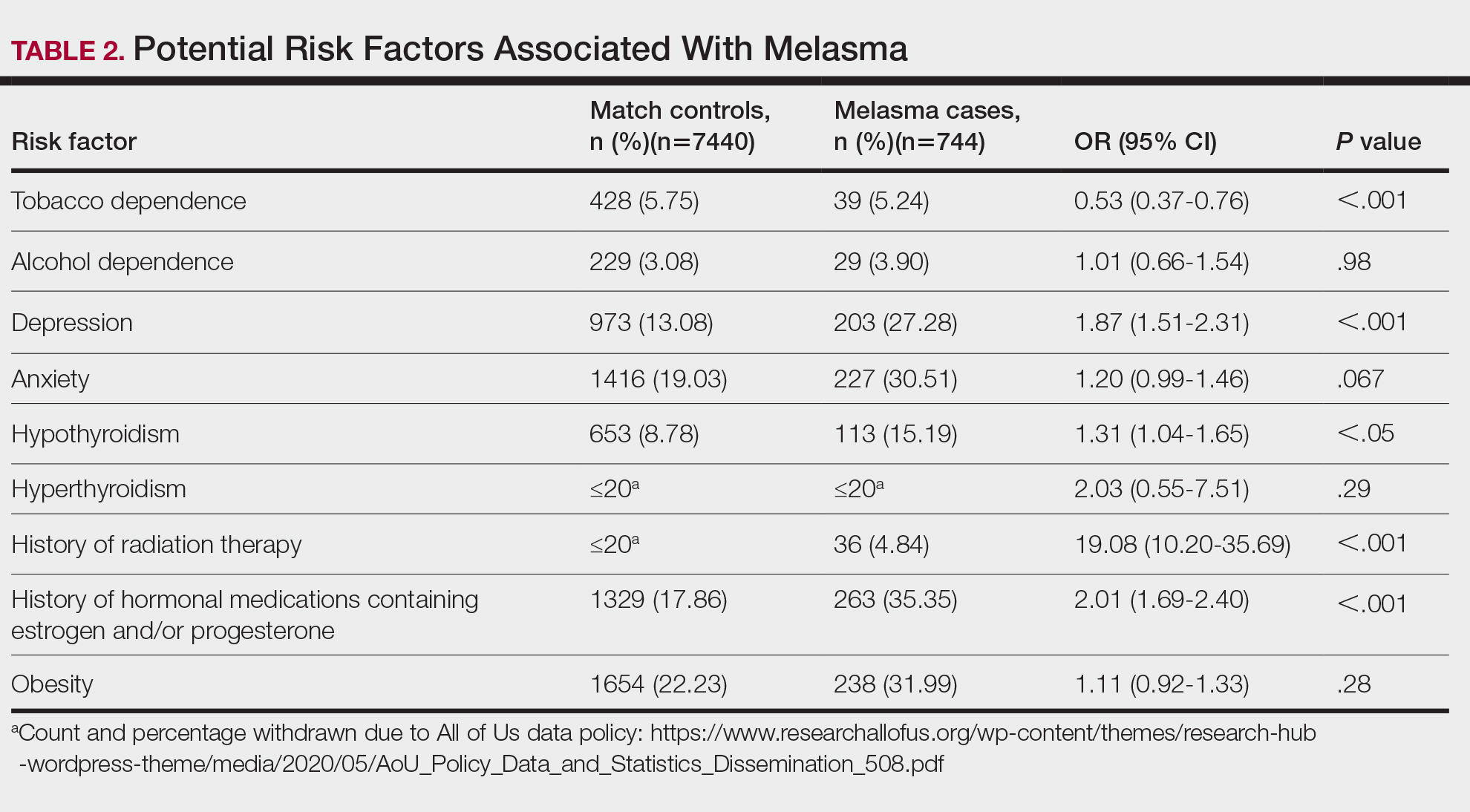
Limitations of our study include lack of information about melasma severity and information about prior melasma treatment in our cohort as well as possible misdiagnosis reported in the dataset.
Our results demonstrated that pre-existing depression and hypothyroidism as well as a history of radiation or estrogen and/or progesterone therapies are potential risk factors for melasma. Therefore, we recommend that patients with melasma be screened for depression and thyroid dysfunction, and patients undergoing radiation therapy or starting estrogen and/or progesterone therapy should be counseled on their increased risk for melasma. Future studies are needed to determine whether treatment of comorbidities such as hypothyroidism and depression improve melasma severity. The decreased risk for melasma associated with tobacco use also requires further investigation.
Acknowledgments—The All of Us Research Program is supported by the National Institutes of Health, Office of the Director: Regional Medical Centers: 1 OT2 OD026549; 1 OT2 OD026554; 1 OT2 OD026557; 1 OT2 OD026556; 1 OT2 OD026550; 1 OT2 OD 026552; 1 OT2 OD026553; 1 OT2 OD026548; 1 OT2 OD026551; 1 OT2 OD026555; IAA #: AOD 16037; Federally Qualified Health Centers: HHSN 263201600085U; Data and Research Center: 5 U2C OD023196; Biobank: 1 U24 OD023121; The Participant Center: U24 OD023176; Participant Technology Systems Center: 1 U24 OD023163; Communications and Engagement: 3 OT2 OD023205; 3 OT2 OD023206; and Community Partners: 1 OT2 OD025277; 3 OT2 OD025315; 1 OT2 OD025337; 1 OT2 OD025276.
In addition, the All of Us Research Program would not be possible without the partnership of its participants, who we gratefully acknowledge for their contributions and without whom this research would not have been possible. We also thank the All of Us Research Program for making the participant data examined in this study available to us.
- Filoni A, Mariano M, Cameli N. Melasma: how hormones can modulate skin pigmentation. J Cosmet Dermatol. 2019;18:458-463. doi:10.1111/jocd.12877
- Platsidaki E, Efstathiou V, Markantoni V, et al. Self-esteem, depression, anxiety and quality of life in patients with melasma living in a sunny mediterranean area: results from a prospective cross-sectional study. Dermatol Ther (Heidelb). 2023;13:1127-1136. doi:10.1007/s13555-023-00915-1
- Handel AC, Lima PB, Tonolli VM, et al. Risk factors for facial melasma in women: a case-control study. Br J Dermatol. 2014;171:588-594. doi:10.1111/bjd.13059
- Erge E, Kiziltunc C, Balci SB, et al. A novel inflammatory marker for the diagnosis of Hashimoto’s thyroiditis: platelet-count-to-lymphocyte-count ratio (published January 22, 2023). Diseases. 2023;11:15. doi:10.3390/diseases11010015
- Kheradmand M, Afshari M, Damiani G, et al. Melasma and thyroid disorders: a systematic review and meta-analysis. Int J Dermatol. 2019;58:1231-1238. doi:10.1111/ijd.14497
- Chu CN, Hu KC, Wu RS, et al. Radiation-irritated skin and hyperpigmentation may impact the quality of life of breast cancer patients after whole breast radiotherapy (published March 31, 2021). BMC Cancer. 2021;21:330. doi:10.1186/s12885-021-08047-5
- Nakamura M, Ueda Y, Hayashi M, et al. Tobacco smoke-induced skin pigmentation is mediated by the aryl hydrocarbon receptor. Exp Dermatol. 2013;22:556-558. doi:10.1111/exd.12170
- Ejaz S, Lim CW. Toxicological overview of cigarette smoking on angiogenesis. Environ Toxicol Pharmacol. 2005;20:335-344. doi:10.1016/j.etap.2005.03.011
To the Editor:
Melasma (also known as chloasma) is characterized by symmetric hyperpigmented patches affecting sun-exposed areas. Women commonly develop this condition during pregnancy, suggesting a connection between melasma and increased female sex hormone levels.1 Other hypothesized risk factors include sun exposure, genetic susceptibility, estrogen and/or progesterone therapy, and thyroid abnormalities but have not been corroborated.2 Treatment options are limited because the pathogenesis is poorly understood; thus, we aimed to analyze melasma risk factors using a national database with a nested case-control approach.
We conducted a matched case-control study using the Registered Tier dataset (version 7) from the National Institute of Health’s All of Us Research Program (https://allofus.nih.gov/), which is available to authorized users through the program’s Researcher Workbench and includes more than 413,000 total participants enrolled from May 1, 2018, through July 1, 2022. Cases included patients 18 years and older with a diagnosis of melasma (International Classification of Diseases, Tenth Revision, Clinical Modification code L81.1 [Chloasma]; concept ID 4264234 [Chloasma]; and Systematized Nomenclature of Medicine [SNOMED] code 36209000 [Chloasma]), and controls without a diagnosis of melasma were matched in a 1:10 ratio based on age, sex, and self-reported race. Concept IDs and SNOMED codes were used to identify individuals in each cohort with a diagnosis of alcohol dependence (concept IDs 433753, 435243, 4218106; SNOMED codes 15167005, 66590003, 7200002), depression (concept ID 440383; SNOMED code 35489007), hypothyroidism (concept ID 140673; SNOMED code 40930008), hyperthyroidism (concept ID 4142479; SNOMED code 34486009), anxiety (concept IDs 441542, 442077, 434613; SNOMED codes 48694002, 197480006, 21897009), tobacco dependence (concept IDs 37109023, 437264, 4099811; SNOMED codes 16077091000119107, 89765005, 191887008), or obesity (concept IDs 433736 and 434005; SNOMED codes 414916001 and 238136002), or with a history of radiation therapy (concept IDs 4085340, 4311117, 4061844, 4029715; SNOMED codes 24803000, 85983004, 200861004, 108290001) or hormonal medications containing estrogen and/or progesterone, including oral medications and implants (concept IDs 21602445, 40254009, 21602514, 21603814, 19049228, 21602529, 1549080, 1551673, 1549254, 21602472, 21602446, 21602450, 21602515, 21602566, 21602473, 21602567, 21602488, 21602585, 1596779, 1586808, 21602524). In our case cohort, diagnoses and exposures to treatments were only considered for analysis if they occurred prior to melasma diagnosis.
Multivariate logistic regression was performed to calculate odds ratios and P values between melasma and each comorbidity or exposure to the treatments specified. Statistical significance was set at P<.05.
We identified 744 melasma cases (mean age, 55.20 years; 95.43% female; 12.10% Black) and 7440 controls with similar demographics (ie, age, sex, race/ethnicity) between groups (all P>.05 [Table 1]). Patients with a melasma diagnosis were more likely to have a pre-existing diagnosis of depression (OR, 1.87; 95% CI, 1.51-2.31 [P<.001]) or hypothyroidism (OR, 1.31; 95% CI, 1.04-1.65 [P<.05]), or a history of radiation therapy (OR, 19.08; 95% CI, 10.20-35.69 [P<.001]) and/or estrogen and/or progesterone therapy (OR, 2.01; 95% CI, 1.69-2.40 [P<.001]) prior to melasma diagnosis. A diagnosis of anxiety prior to melasma diagnosis trended toward an association with melasma (P=.067). Pre-existing alcohol dependence, obesity, and hyperthyroidism were not associated with melasma (P=.98, P=.28, and P=.29, respectively). A diagnosis of tobacco dependence was associated with a decreased melasma risk (OR, 0.53, 95% CI, 0.37-0.76)[P<.001])(Table 2).
Our study results suggest that pre-existing depression was a risk factor for subsequent melasma diagnosis. Depression may exacerbate stress, leading to increased activation of the hypothalamic-pituitary-adrenal axis as well as increased levels of cortisol and adrenocorticotropic hormone, which subsequently act on melanocytes to increase melanogenesis.3 A retrospective study of 254 participants, including 127 with melasma, showed that increased melasma severity was associated with higher rates of depression (P=.002)2; however, the risk for melasma following a depression diagnosis has not been reported.
Our results also showed that hypothyroidism was associated with an increased risk for melasma. On a cellular level, hypothyroidism can cause systemic inflammation, potentailly leading to increased stress and melanogenesis via activation of the hypothalamic-pituitary-adrenal axis.4 These findings are similar to a systematic review and meta-analysis reporting increased thyroid-stimulating hormone, anti–thyroid peroxidase, and antithyroglobulin antibody levels associated with increased melasma risk (mean difference between cases and controls, 0.33 [95% CI, 0.18-0.47]; pooled association, P=.020; mean difference between cases and controls, 0.28 [95% CI, 0.01-0.55], respectively).5
Patients in our cohort who had a history of radiation therapy were 19 times more likely to develop melasma, similar to findings of a survey-based study of 421 breast cancer survivors in which 336 (79.81%) reported hyperpigmentation in irradiated areas.6 Patients in our cohort who had a history of estrogen and/or progesterone therapy were 2 times more likely to develop melasma, similar to a case-control study of 207 patients with melasma and 207 controls that showed combined oral contraceptives increased risk for melasma (OR, 1.23 [95% CI, 1.08-1.41; P<.01).3
Tobacco use is not a well-known protective factor against melasma. Prior studies have indicated that tobacco smoking activates melanocytes via the Wnt/β-Catenin pathway, leading to hyperpigmentation.7 Although exposure to cigarette smoke decreases angiogenesis and would more likely lead to hyperpigmentation, nicotine exposure has been shown to increase angiogenesis, which could lead to increased blood flow and partially explain the protection against melasma demonstrated in our cohort.8 Future studies are needed to explore this relationship.


Limitations of our study include lack of information about melasma severity and information about prior melasma treatment in our cohort as well as possible misdiagnosis reported in the dataset.
Our results demonstrated that pre-existing depression and hypothyroidism as well as a history of radiation or estrogen and/or progesterone therapies are potential risk factors for melasma. Therefore, we recommend that patients with melasma be screened for depression and thyroid dysfunction, and patients undergoing radiation therapy or starting estrogen and/or progesterone therapy should be counseled on their increased risk for melasma. Future studies are needed to determine whether treatment of comorbidities such as hypothyroidism and depression improve melasma severity. The decreased risk for melasma associated with tobacco use also requires further investigation.
Acknowledgments—The All of Us Research Program is supported by the National Institutes of Health, Office of the Director: Regional Medical Centers: 1 OT2 OD026549; 1 OT2 OD026554; 1 OT2 OD026557; 1 OT2 OD026556; 1 OT2 OD026550; 1 OT2 OD 026552; 1 OT2 OD026553; 1 OT2 OD026548; 1 OT2 OD026551; 1 OT2 OD026555; IAA #: AOD 16037; Federally Qualified Health Centers: HHSN 263201600085U; Data and Research Center: 5 U2C OD023196; Biobank: 1 U24 OD023121; The Participant Center: U24 OD023176; Participant Technology Systems Center: 1 U24 OD023163; Communications and Engagement: 3 OT2 OD023205; 3 OT2 OD023206; and Community Partners: 1 OT2 OD025277; 3 OT2 OD025315; 1 OT2 OD025337; 1 OT2 OD025276.
In addition, the All of Us Research Program would not be possible without the partnership of its participants, who we gratefully acknowledge for their contributions and without whom this research would not have been possible. We also thank the All of Us Research Program for making the participant data examined in this study available to us.
To the Editor:
Melasma (also known as chloasma) is characterized by symmetric hyperpigmented patches affecting sun-exposed areas. Women commonly develop this condition during pregnancy, suggesting a connection between melasma and increased female sex hormone levels.1 Other hypothesized risk factors include sun exposure, genetic susceptibility, estrogen and/or progesterone therapy, and thyroid abnormalities but have not been corroborated.2 Treatment options are limited because the pathogenesis is poorly understood; thus, we aimed to analyze melasma risk factors using a national database with a nested case-control approach.
We conducted a matched case-control study using the Registered Tier dataset (version 7) from the National Institute of Health’s All of Us Research Program (https://allofus.nih.gov/), which is available to authorized users through the program’s Researcher Workbench and includes more than 413,000 total participants enrolled from May 1, 2018, through July 1, 2022. Cases included patients 18 years and older with a diagnosis of melasma (International Classification of Diseases, Tenth Revision, Clinical Modification code L81.1 [Chloasma]; concept ID 4264234 [Chloasma]; and Systematized Nomenclature of Medicine [SNOMED] code 36209000 [Chloasma]), and controls without a diagnosis of melasma were matched in a 1:10 ratio based on age, sex, and self-reported race. Concept IDs and SNOMED codes were used to identify individuals in each cohort with a diagnosis of alcohol dependence (concept IDs 433753, 435243, 4218106; SNOMED codes 15167005, 66590003, 7200002), depression (concept ID 440383; SNOMED code 35489007), hypothyroidism (concept ID 140673; SNOMED code 40930008), hyperthyroidism (concept ID 4142479; SNOMED code 34486009), anxiety (concept IDs 441542, 442077, 434613; SNOMED codes 48694002, 197480006, 21897009), tobacco dependence (concept IDs 37109023, 437264, 4099811; SNOMED codes 16077091000119107, 89765005, 191887008), or obesity (concept IDs 433736 and 434005; SNOMED codes 414916001 and 238136002), or with a history of radiation therapy (concept IDs 4085340, 4311117, 4061844, 4029715; SNOMED codes 24803000, 85983004, 200861004, 108290001) or hormonal medications containing estrogen and/or progesterone, including oral medications and implants (concept IDs 21602445, 40254009, 21602514, 21603814, 19049228, 21602529, 1549080, 1551673, 1549254, 21602472, 21602446, 21602450, 21602515, 21602566, 21602473, 21602567, 21602488, 21602585, 1596779, 1586808, 21602524). In our case cohort, diagnoses and exposures to treatments were only considered for analysis if they occurred prior to melasma diagnosis.
Multivariate logistic regression was performed to calculate odds ratios and P values between melasma and each comorbidity or exposure to the treatments specified. Statistical significance was set at P<.05.
We identified 744 melasma cases (mean age, 55.20 years; 95.43% female; 12.10% Black) and 7440 controls with similar demographics (ie, age, sex, race/ethnicity) between groups (all P>.05 [Table 1]). Patients with a melasma diagnosis were more likely to have a pre-existing diagnosis of depression (OR, 1.87; 95% CI, 1.51-2.31 [P<.001]) or hypothyroidism (OR, 1.31; 95% CI, 1.04-1.65 [P<.05]), or a history of radiation therapy (OR, 19.08; 95% CI, 10.20-35.69 [P<.001]) and/or estrogen and/or progesterone therapy (OR, 2.01; 95% CI, 1.69-2.40 [P<.001]) prior to melasma diagnosis. A diagnosis of anxiety prior to melasma diagnosis trended toward an association with melasma (P=.067). Pre-existing alcohol dependence, obesity, and hyperthyroidism were not associated with melasma (P=.98, P=.28, and P=.29, respectively). A diagnosis of tobacco dependence was associated with a decreased melasma risk (OR, 0.53, 95% CI, 0.37-0.76)[P<.001])(Table 2).
Our study results suggest that pre-existing depression was a risk factor for subsequent melasma diagnosis. Depression may exacerbate stress, leading to increased activation of the hypothalamic-pituitary-adrenal axis as well as increased levels of cortisol and adrenocorticotropic hormone, which subsequently act on melanocytes to increase melanogenesis.3 A retrospective study of 254 participants, including 127 with melasma, showed that increased melasma severity was associated with higher rates of depression (P=.002)2; however, the risk for melasma following a depression diagnosis has not been reported.
Our results also showed that hypothyroidism was associated with an increased risk for melasma. On a cellular level, hypothyroidism can cause systemic inflammation, potentailly leading to increased stress and melanogenesis via activation of the hypothalamic-pituitary-adrenal axis.4 These findings are similar to a systematic review and meta-analysis reporting increased thyroid-stimulating hormone, anti–thyroid peroxidase, and antithyroglobulin antibody levels associated with increased melasma risk (mean difference between cases and controls, 0.33 [95% CI, 0.18-0.47]; pooled association, P=.020; mean difference between cases and controls, 0.28 [95% CI, 0.01-0.55], respectively).5
Patients in our cohort who had a history of radiation therapy were 19 times more likely to develop melasma, similar to findings of a survey-based study of 421 breast cancer survivors in which 336 (79.81%) reported hyperpigmentation in irradiated areas.6 Patients in our cohort who had a history of estrogen and/or progesterone therapy were 2 times more likely to develop melasma, similar to a case-control study of 207 patients with melasma and 207 controls that showed combined oral contraceptives increased risk for melasma (OR, 1.23 [95% CI, 1.08-1.41; P<.01).3
Tobacco use is not a well-known protective factor against melasma. Prior studies have indicated that tobacco smoking activates melanocytes via the Wnt/β-Catenin pathway, leading to hyperpigmentation.7 Although exposure to cigarette smoke decreases angiogenesis and would more likely lead to hyperpigmentation, nicotine exposure has been shown to increase angiogenesis, which could lead to increased blood flow and partially explain the protection against melasma demonstrated in our cohort.8 Future studies are needed to explore this relationship.


Limitations of our study include lack of information about melasma severity and information about prior melasma treatment in our cohort as well as possible misdiagnosis reported in the dataset.
Our results demonstrated that pre-existing depression and hypothyroidism as well as a history of radiation or estrogen and/or progesterone therapies are potential risk factors for melasma. Therefore, we recommend that patients with melasma be screened for depression and thyroid dysfunction, and patients undergoing radiation therapy or starting estrogen and/or progesterone therapy should be counseled on their increased risk for melasma. Future studies are needed to determine whether treatment of comorbidities such as hypothyroidism and depression improve melasma severity. The decreased risk for melasma associated with tobacco use also requires further investigation.
Acknowledgments—The All of Us Research Program is supported by the National Institutes of Health, Office of the Director: Regional Medical Centers: 1 OT2 OD026549; 1 OT2 OD026554; 1 OT2 OD026557; 1 OT2 OD026556; 1 OT2 OD026550; 1 OT2 OD 026552; 1 OT2 OD026553; 1 OT2 OD026548; 1 OT2 OD026551; 1 OT2 OD026555; IAA #: AOD 16037; Federally Qualified Health Centers: HHSN 263201600085U; Data and Research Center: 5 U2C OD023196; Biobank: 1 U24 OD023121; The Participant Center: U24 OD023176; Participant Technology Systems Center: 1 U24 OD023163; Communications and Engagement: 3 OT2 OD023205; 3 OT2 OD023206; and Community Partners: 1 OT2 OD025277; 3 OT2 OD025315; 1 OT2 OD025337; 1 OT2 OD025276.
In addition, the All of Us Research Program would not be possible without the partnership of its participants, who we gratefully acknowledge for their contributions and without whom this research would not have been possible. We also thank the All of Us Research Program for making the participant data examined in this study available to us.
- Filoni A, Mariano M, Cameli N. Melasma: how hormones can modulate skin pigmentation. J Cosmet Dermatol. 2019;18:458-463. doi:10.1111/jocd.12877
- Platsidaki E, Efstathiou V, Markantoni V, et al. Self-esteem, depression, anxiety and quality of life in patients with melasma living in a sunny mediterranean area: results from a prospective cross-sectional study. Dermatol Ther (Heidelb). 2023;13:1127-1136. doi:10.1007/s13555-023-00915-1
- Handel AC, Lima PB, Tonolli VM, et al. Risk factors for facial melasma in women: a case-control study. Br J Dermatol. 2014;171:588-594. doi:10.1111/bjd.13059
- Erge E, Kiziltunc C, Balci SB, et al. A novel inflammatory marker for the diagnosis of Hashimoto’s thyroiditis: platelet-count-to-lymphocyte-count ratio (published January 22, 2023). Diseases. 2023;11:15. doi:10.3390/diseases11010015
- Kheradmand M, Afshari M, Damiani G, et al. Melasma and thyroid disorders: a systematic review and meta-analysis. Int J Dermatol. 2019;58:1231-1238. doi:10.1111/ijd.14497
- Chu CN, Hu KC, Wu RS, et al. Radiation-irritated skin and hyperpigmentation may impact the quality of life of breast cancer patients after whole breast radiotherapy (published March 31, 2021). BMC Cancer. 2021;21:330. doi:10.1186/s12885-021-08047-5
- Nakamura M, Ueda Y, Hayashi M, et al. Tobacco smoke-induced skin pigmentation is mediated by the aryl hydrocarbon receptor. Exp Dermatol. 2013;22:556-558. doi:10.1111/exd.12170
- Ejaz S, Lim CW. Toxicological overview of cigarette smoking on angiogenesis. Environ Toxicol Pharmacol. 2005;20:335-344. doi:10.1016/j.etap.2005.03.011
- Filoni A, Mariano M, Cameli N. Melasma: how hormones can modulate skin pigmentation. J Cosmet Dermatol. 2019;18:458-463. doi:10.1111/jocd.12877
- Platsidaki E, Efstathiou V, Markantoni V, et al. Self-esteem, depression, anxiety and quality of life in patients with melasma living in a sunny mediterranean area: results from a prospective cross-sectional study. Dermatol Ther (Heidelb). 2023;13:1127-1136. doi:10.1007/s13555-023-00915-1
- Handel AC, Lima PB, Tonolli VM, et al. Risk factors for facial melasma in women: a case-control study. Br J Dermatol. 2014;171:588-594. doi:10.1111/bjd.13059
- Erge E, Kiziltunc C, Balci SB, et al. A novel inflammatory marker for the diagnosis of Hashimoto’s thyroiditis: platelet-count-to-lymphocyte-count ratio (published January 22, 2023). Diseases. 2023;11:15. doi:10.3390/diseases11010015
- Kheradmand M, Afshari M, Damiani G, et al. Melasma and thyroid disorders: a systematic review and meta-analysis. Int J Dermatol. 2019;58:1231-1238. doi:10.1111/ijd.14497
- Chu CN, Hu KC, Wu RS, et al. Radiation-irritated skin and hyperpigmentation may impact the quality of life of breast cancer patients after whole breast radiotherapy (published March 31, 2021). BMC Cancer. 2021;21:330. doi:10.1186/s12885-021-08047-5
- Nakamura M, Ueda Y, Hayashi M, et al. Tobacco smoke-induced skin pigmentation is mediated by the aryl hydrocarbon receptor. Exp Dermatol. 2013;22:556-558. doi:10.1111/exd.12170
- Ejaz S, Lim CW. Toxicological overview of cigarette smoking on angiogenesis. Environ Toxicol Pharmacol. 2005;20:335-344. doi:10.1016/j.etap.2005.03.011
Practice Points
- Treatment options for melasma are limited due to its poorly understood pathogenesis.
- Depression and hypothyroidism and/or history of exposure to radiation and hormonal therapies may increase melasma risk.
- We recommend that patients with melasma be screened for depression and thyroid dysfunction. Patients undergoing radiation therapy or starting estrogen and/ or progesterone therapy should be counseled on the increased risk for melasma.