User login
Life-threatening paradoxical bronchospasm may escape recognition in patients with COPD or asthma
according to a researcher who reviewed spirometry test results from U.S. military veterans.
Nearly 1.5% of the tests met the criteria for paradoxical bronchospasm, which refers to airway constriction that may rapidly occur after inhalation of a short-acting beta2 agonist (SABA) such as albuterol.
However, none of those reports alluded to paradoxical bronchospasm, said investigator Malvika Kaul, MD, fellow in the department of pulmonary and critical care at the University of Illinois at Chicago and the Jesse Brown Veterans Affairs Medical Center, also in Chicago.
“Paradoxical bronchospasm was neither recognized nor reported in any spirometry test results,” Dr. Kaul said in an online poster presentation at the annual meeting of the American College of Chest Physicians, held virtually this year.
By recognizing paradoxical bronchospasm, health care providers could address its clinical implications and identify potential alternative management options, according to Dr. Kaul.
“We hope in the future, education of clinicians about this phenomena is emphasized,” Dr. Kaul said in her presentation.
Recognizing paradoxical bronchospasm
In an interview, Dr. Kaul said she began researching paradoxical bronchospasm after encountering a patient who had an acute reaction to albuterol during a pulmonary function test.
“I was not taught about it, and I wasn’t recognizing that pattern very frequently in my patients,” she said.
Prescribing information for Food and Drug Administration–approved SABAs include a warning that life-threatening paradoxical bronchospasm may occur, said Dr. Kaul.
If paradoxical bronchospasm occurs, the patient should discontinue the medication immediately and start on alternative therapy, according to the available prescribing information for albuterol sulfate.
Paradoxical bronchospasm has been linked to worsened respiratory outcomes, including more frequent exacerbations, in patients with obstructive lung diseases, according to Dr. Kaul.
Two previous large studies pegged the prevalence of paradoxical bronchospasm at around 4.5% in patients with COPD or asthma, but “it has not been reported or addressed in high-risk population, such as veterans who have high prevalence of obstructive lung diseases like COPD,” Dr. Kaul said.
Latest study results
Dr. Kaul described a retrospective analysis of 1,150 pre- and postbronchodilator spirometry tests conducted in patients with COPD or asthma at the Jesse Brown VA Medical Center between 2017 and 2020.
A positive paradoxical bronchodilator response was defined as a decrease of least 12% and 200 mL in forced expiratory volume in 1 second and forced vital capacity from baseline after four puffs of albuterol were inhaled, Dr. Kaul said.
Out of 18 reviewed spirometry results that met the criteria, none of the test results reported or recognized paradoxical bronchospasm, according to Dr. Kaul.
Those meeting the criteria were predominantly COPD patients, according to Dr. Kaul, who said 12 had an underlying diagnosis COPD, 4 had asthma, and 2 had COPD and asthma.
Of the 18 patients, 13 were African American, and all but 1 of the 18 patients had a current or past smoking history, according to reported data.
A history of obstructive sleep apnea was reported in nine patients, and history of gastroesophageal reflux disease was also reported in nine patients. Eleven patients had emphysema.
Greater awareness needed
Results of this study emphasize the need to recognize potential cases paradoxical bronchospasm in clinical practice, as well as a need for more research, according to Allen J. Blaivas, DO, FCCP, chair of the CHEST Airway Disorders NetWork.
“It’s something to be on the alert for, and certainly be aware that, if your patient is telling you that they feel worse, we shouldn’t just pooh-pooh it,” said Dr. Blaivas, who is medical director of the intensive care unit at the East Orange campus of the VA New Jersey Health Care System.
Further research could focus on breaking down whether patients with suspected paradoxical bronchospasm are using metered-dose inhalers or nebulizers, whether or not they are also taking inhaled corticosteroids, and whether prospective testing can confirm paradoxical bronchospasm in patients who report tightness after using a SABA, he said in an interview.
Dr. Kaul and coauthor Israel Rubinstein, MD had no relevant relationships to disclose. Dr. Blaivas had no relevant relationships to disclose.
according to a researcher who reviewed spirometry test results from U.S. military veterans.
Nearly 1.5% of the tests met the criteria for paradoxical bronchospasm, which refers to airway constriction that may rapidly occur after inhalation of a short-acting beta2 agonist (SABA) such as albuterol.
However, none of those reports alluded to paradoxical bronchospasm, said investigator Malvika Kaul, MD, fellow in the department of pulmonary and critical care at the University of Illinois at Chicago and the Jesse Brown Veterans Affairs Medical Center, also in Chicago.
“Paradoxical bronchospasm was neither recognized nor reported in any spirometry test results,” Dr. Kaul said in an online poster presentation at the annual meeting of the American College of Chest Physicians, held virtually this year.
By recognizing paradoxical bronchospasm, health care providers could address its clinical implications and identify potential alternative management options, according to Dr. Kaul.
“We hope in the future, education of clinicians about this phenomena is emphasized,” Dr. Kaul said in her presentation.
Recognizing paradoxical bronchospasm
In an interview, Dr. Kaul said she began researching paradoxical bronchospasm after encountering a patient who had an acute reaction to albuterol during a pulmonary function test.
“I was not taught about it, and I wasn’t recognizing that pattern very frequently in my patients,” she said.
Prescribing information for Food and Drug Administration–approved SABAs include a warning that life-threatening paradoxical bronchospasm may occur, said Dr. Kaul.
If paradoxical bronchospasm occurs, the patient should discontinue the medication immediately and start on alternative therapy, according to the available prescribing information for albuterol sulfate.
Paradoxical bronchospasm has been linked to worsened respiratory outcomes, including more frequent exacerbations, in patients with obstructive lung diseases, according to Dr. Kaul.
Two previous large studies pegged the prevalence of paradoxical bronchospasm at around 4.5% in patients with COPD or asthma, but “it has not been reported or addressed in high-risk population, such as veterans who have high prevalence of obstructive lung diseases like COPD,” Dr. Kaul said.
Latest study results
Dr. Kaul described a retrospective analysis of 1,150 pre- and postbronchodilator spirometry tests conducted in patients with COPD or asthma at the Jesse Brown VA Medical Center between 2017 and 2020.
A positive paradoxical bronchodilator response was defined as a decrease of least 12% and 200 mL in forced expiratory volume in 1 second and forced vital capacity from baseline after four puffs of albuterol were inhaled, Dr. Kaul said.
Out of 18 reviewed spirometry results that met the criteria, none of the test results reported or recognized paradoxical bronchospasm, according to Dr. Kaul.
Those meeting the criteria were predominantly COPD patients, according to Dr. Kaul, who said 12 had an underlying diagnosis COPD, 4 had asthma, and 2 had COPD and asthma.
Of the 18 patients, 13 were African American, and all but 1 of the 18 patients had a current or past smoking history, according to reported data.
A history of obstructive sleep apnea was reported in nine patients, and history of gastroesophageal reflux disease was also reported in nine patients. Eleven patients had emphysema.
Greater awareness needed
Results of this study emphasize the need to recognize potential cases paradoxical bronchospasm in clinical practice, as well as a need for more research, according to Allen J. Blaivas, DO, FCCP, chair of the CHEST Airway Disorders NetWork.
“It’s something to be on the alert for, and certainly be aware that, if your patient is telling you that they feel worse, we shouldn’t just pooh-pooh it,” said Dr. Blaivas, who is medical director of the intensive care unit at the East Orange campus of the VA New Jersey Health Care System.
Further research could focus on breaking down whether patients with suspected paradoxical bronchospasm are using metered-dose inhalers or nebulizers, whether or not they are also taking inhaled corticosteroids, and whether prospective testing can confirm paradoxical bronchospasm in patients who report tightness after using a SABA, he said in an interview.
Dr. Kaul and coauthor Israel Rubinstein, MD had no relevant relationships to disclose. Dr. Blaivas had no relevant relationships to disclose.
according to a researcher who reviewed spirometry test results from U.S. military veterans.
Nearly 1.5% of the tests met the criteria for paradoxical bronchospasm, which refers to airway constriction that may rapidly occur after inhalation of a short-acting beta2 agonist (SABA) such as albuterol.
However, none of those reports alluded to paradoxical bronchospasm, said investigator Malvika Kaul, MD, fellow in the department of pulmonary and critical care at the University of Illinois at Chicago and the Jesse Brown Veterans Affairs Medical Center, also in Chicago.
“Paradoxical bronchospasm was neither recognized nor reported in any spirometry test results,” Dr. Kaul said in an online poster presentation at the annual meeting of the American College of Chest Physicians, held virtually this year.
By recognizing paradoxical bronchospasm, health care providers could address its clinical implications and identify potential alternative management options, according to Dr. Kaul.
“We hope in the future, education of clinicians about this phenomena is emphasized,” Dr. Kaul said in her presentation.
Recognizing paradoxical bronchospasm
In an interview, Dr. Kaul said she began researching paradoxical bronchospasm after encountering a patient who had an acute reaction to albuterol during a pulmonary function test.
“I was not taught about it, and I wasn’t recognizing that pattern very frequently in my patients,” she said.
Prescribing information for Food and Drug Administration–approved SABAs include a warning that life-threatening paradoxical bronchospasm may occur, said Dr. Kaul.
If paradoxical bronchospasm occurs, the patient should discontinue the medication immediately and start on alternative therapy, according to the available prescribing information for albuterol sulfate.
Paradoxical bronchospasm has been linked to worsened respiratory outcomes, including more frequent exacerbations, in patients with obstructive lung diseases, according to Dr. Kaul.
Two previous large studies pegged the prevalence of paradoxical bronchospasm at around 4.5% in patients with COPD or asthma, but “it has not been reported or addressed in high-risk population, such as veterans who have high prevalence of obstructive lung diseases like COPD,” Dr. Kaul said.
Latest study results
Dr. Kaul described a retrospective analysis of 1,150 pre- and postbronchodilator spirometry tests conducted in patients with COPD or asthma at the Jesse Brown VA Medical Center between 2017 and 2020.
A positive paradoxical bronchodilator response was defined as a decrease of least 12% and 200 mL in forced expiratory volume in 1 second and forced vital capacity from baseline after four puffs of albuterol were inhaled, Dr. Kaul said.
Out of 18 reviewed spirometry results that met the criteria, none of the test results reported or recognized paradoxical bronchospasm, according to Dr. Kaul.
Those meeting the criteria were predominantly COPD patients, according to Dr. Kaul, who said 12 had an underlying diagnosis COPD, 4 had asthma, and 2 had COPD and asthma.
Of the 18 patients, 13 were African American, and all but 1 of the 18 patients had a current or past smoking history, according to reported data.
A history of obstructive sleep apnea was reported in nine patients, and history of gastroesophageal reflux disease was also reported in nine patients. Eleven patients had emphysema.
Greater awareness needed
Results of this study emphasize the need to recognize potential cases paradoxical bronchospasm in clinical practice, as well as a need for more research, according to Allen J. Blaivas, DO, FCCP, chair of the CHEST Airway Disorders NetWork.
“It’s something to be on the alert for, and certainly be aware that, if your patient is telling you that they feel worse, we shouldn’t just pooh-pooh it,” said Dr. Blaivas, who is medical director of the intensive care unit at the East Orange campus of the VA New Jersey Health Care System.
Further research could focus on breaking down whether patients with suspected paradoxical bronchospasm are using metered-dose inhalers or nebulizers, whether or not they are also taking inhaled corticosteroids, and whether prospective testing can confirm paradoxical bronchospasm in patients who report tightness after using a SABA, he said in an interview.
Dr. Kaul and coauthor Israel Rubinstein, MD had no relevant relationships to disclose. Dr. Blaivas had no relevant relationships to disclose.
FROM CHEST 2021
Generalized Pustular Psoriasis: An Uncommon Diagnosis Carrying an Outsize Burden of Disease
In this supplement to Dermatology News, Alan Menter, MD discusses best practices for the rare, severe, and chronic autoinflammatory disease known as Generalized Pustular Psoriasis (GPP).
Read More
In this supplement to Dermatology News, Alan Menter, MD discusses best practices for the rare, severe, and chronic autoinflammatory disease known as Generalized Pustular Psoriasis (GPP).
Read More
In this supplement to Dermatology News, Alan Menter, MD discusses best practices for the rare, severe, and chronic autoinflammatory disease known as Generalized Pustular Psoriasis (GPP).
Read More
MS and (non-COVID) vaccinations: consensus recommendations
by the European Committee for Treatment and Research in Multiple Sclerosis (ECTRIMS) and the European Academy of Neurology (EAN).
The document, announced at the annual ECTRIMS meeting, proposes a standard for vaccination in patients with MS, including a global vaccination strategy for the general MS patient population and selected subpopulations.
The document does not include any recommendations regarding vaccination against COVID-19, which is the subject of a separate report, announced at the annual meeting.
The main conclusions in the new report are as follows:
- Vaccinations in general are considered safe for patients with MS and do not modify disease activity/progression.
- Live attenuated vaccines, however, are contraindicated with immunosuppressants.
- Inactivated vaccines can be used safely, but their efficacy may be decreased with immunosuppressants.
- Vaccinations should be considered early in MS management before using immunosuppressants whenever possible.
Presenting the vaccination consensus document, Susana Otero-Romero, MD, from the Multiple Sclerosis Center of Catalonia, Spain, explained that vaccination has become an important part of the risk management strategy in patients with MS treated with highly active drugs but that questions remain as to when and whether to introduce a particular vaccine and which disease-modifying treatments affect vaccine response.
“The current reference tool has been developed to help professionals to decide on the best vaccination strategy for their patients,” she said.
The consensus document recommends that, in general, vaccination should be performed at the time of diagnosis of MS or in the early stages of the disease to prevent future delays in starting therapies.
“Ideally, vaccination should take place before the onset of disease-modifying treatment,” Dr. Otero-Romero said. The consensus document recommends inactivated vaccines to be given 2-3 weeks before immunosuppressive therapy is started, and live attenuated vaccines at least 4 weeks beforehand.
In the case of relapse, vaccination should be delayed until clinical resolution or stabilization if possible, the consensus statement recommends.
Serological testing for vaccine-induced antibody titers can be performed 1-2 months after the last dose of the vaccine (suggested for hepatitis B, measles, mumps, and varicella). For attenuated live vaccines, serological tests should be done before starting immunosuppressive therapy. In the case of insufficient response, consideration should be given to administering a booster dose of the vaccine, except for hepatitis B, in which a complete revaccination is recommended, according to the document.
As for vaccination during immunosuppressive therapy, this is considered safe for patients on interferon or glatiramer acetate when indicated, the report says.
Vaccination should ideally be avoided in patients on dimethyl fumarate, teriflunomide (Aubagio) or natalizumab (Tysabri), although it can be considered in exceptional cases when the potential risk of acquiring the infection is greater than the risk of developing vaccine-related infections (unless the absolute lymphocyte count is below 800/mm3), it adds.
Vaccination should be avoided in patients on S1P modulators (for example, fingolimod [Gilenya]), anti-CD20 therapies, and before immune restoration for cladribine (Leustatin) and alemtuzumab (Lemtrada).
In the case of patients stopping immunosuppressive therapy, inactivated vaccines can be given any time after the discontinuation of therapy but preferably after immune restoration. Live attenuated vaccines should only be administered after a safety interval ensures immune restoration has been met.
Which vaccines?
On which vaccines are needed in patients with MS, the consensus document recommends the same routine vaccination schedule as for the general population. In addition, it advises influenza and pneumococcal vaccination if patients are immunosuppressed or have significant disability.
It also recommends human papillomavirus vaccine in women and men independent of their age if they are to be treated with alemtuzumab, fingolimod, cladribine, or anti-CD20 drugs. Hepatitis B vaccination is also advised in patients treated with anti-CD20 drugs.
Special populations: pregnancy/elderly
In patients with MS who are pregnant, inactivated flu vaccine can be given in any trimester at the start of the flu season, and vaccination against diphtheria, tetanus, and pertussis can be given during the third trimester, the report says. Live attenuated vaccines should be completed at least 1 month before pregnancy or after delivery and 4-6 weeks prior to the initiation of immunosuppressive therapy.
Elderly patients with MS should receive flu and pneumococcal vaccines annually and would also benefit from the inactivated herpes zoster vaccine.
Travel vaccines
On vaccinations needed for travel, the report recommends that patients with MS consult a specialized travel clinic or vaccination expert and start immunizations 2-3 months before departure. Patients with MS with or without immunosuppressive therapy can receive hepatitis A, rabies, Japanese encephalitis, tic-borne encephalitis, polio, and inactivated typhoid vaccine. But yellow fever and oral typhoid are contraindicated in patients on immunosuppressive therapies.
A version of this article first appeared on Medscape.com.
by the European Committee for Treatment and Research in Multiple Sclerosis (ECTRIMS) and the European Academy of Neurology (EAN).
The document, announced at the annual ECTRIMS meeting, proposes a standard for vaccination in patients with MS, including a global vaccination strategy for the general MS patient population and selected subpopulations.
The document does not include any recommendations regarding vaccination against COVID-19, which is the subject of a separate report, announced at the annual meeting.
The main conclusions in the new report are as follows:
- Vaccinations in general are considered safe for patients with MS and do not modify disease activity/progression.
- Live attenuated vaccines, however, are contraindicated with immunosuppressants.
- Inactivated vaccines can be used safely, but their efficacy may be decreased with immunosuppressants.
- Vaccinations should be considered early in MS management before using immunosuppressants whenever possible.
Presenting the vaccination consensus document, Susana Otero-Romero, MD, from the Multiple Sclerosis Center of Catalonia, Spain, explained that vaccination has become an important part of the risk management strategy in patients with MS treated with highly active drugs but that questions remain as to when and whether to introduce a particular vaccine and which disease-modifying treatments affect vaccine response.
“The current reference tool has been developed to help professionals to decide on the best vaccination strategy for their patients,” she said.
The consensus document recommends that, in general, vaccination should be performed at the time of diagnosis of MS or in the early stages of the disease to prevent future delays in starting therapies.
“Ideally, vaccination should take place before the onset of disease-modifying treatment,” Dr. Otero-Romero said. The consensus document recommends inactivated vaccines to be given 2-3 weeks before immunosuppressive therapy is started, and live attenuated vaccines at least 4 weeks beforehand.
In the case of relapse, vaccination should be delayed until clinical resolution or stabilization if possible, the consensus statement recommends.
Serological testing for vaccine-induced antibody titers can be performed 1-2 months after the last dose of the vaccine (suggested for hepatitis B, measles, mumps, and varicella). For attenuated live vaccines, serological tests should be done before starting immunosuppressive therapy. In the case of insufficient response, consideration should be given to administering a booster dose of the vaccine, except for hepatitis B, in which a complete revaccination is recommended, according to the document.
As for vaccination during immunosuppressive therapy, this is considered safe for patients on interferon or glatiramer acetate when indicated, the report says.
Vaccination should ideally be avoided in patients on dimethyl fumarate, teriflunomide (Aubagio) or natalizumab (Tysabri), although it can be considered in exceptional cases when the potential risk of acquiring the infection is greater than the risk of developing vaccine-related infections (unless the absolute lymphocyte count is below 800/mm3), it adds.
Vaccination should be avoided in patients on S1P modulators (for example, fingolimod [Gilenya]), anti-CD20 therapies, and before immune restoration for cladribine (Leustatin) and alemtuzumab (Lemtrada).
In the case of patients stopping immunosuppressive therapy, inactivated vaccines can be given any time after the discontinuation of therapy but preferably after immune restoration. Live attenuated vaccines should only be administered after a safety interval ensures immune restoration has been met.
Which vaccines?
On which vaccines are needed in patients with MS, the consensus document recommends the same routine vaccination schedule as for the general population. In addition, it advises influenza and pneumococcal vaccination if patients are immunosuppressed or have significant disability.
It also recommends human papillomavirus vaccine in women and men independent of their age if they are to be treated with alemtuzumab, fingolimod, cladribine, or anti-CD20 drugs. Hepatitis B vaccination is also advised in patients treated with anti-CD20 drugs.
Special populations: pregnancy/elderly
In patients with MS who are pregnant, inactivated flu vaccine can be given in any trimester at the start of the flu season, and vaccination against diphtheria, tetanus, and pertussis can be given during the third trimester, the report says. Live attenuated vaccines should be completed at least 1 month before pregnancy or after delivery and 4-6 weeks prior to the initiation of immunosuppressive therapy.
Elderly patients with MS should receive flu and pneumococcal vaccines annually and would also benefit from the inactivated herpes zoster vaccine.
Travel vaccines
On vaccinations needed for travel, the report recommends that patients with MS consult a specialized travel clinic or vaccination expert and start immunizations 2-3 months before departure. Patients with MS with or without immunosuppressive therapy can receive hepatitis A, rabies, Japanese encephalitis, tic-borne encephalitis, polio, and inactivated typhoid vaccine. But yellow fever and oral typhoid are contraindicated in patients on immunosuppressive therapies.
A version of this article first appeared on Medscape.com.
by the European Committee for Treatment and Research in Multiple Sclerosis (ECTRIMS) and the European Academy of Neurology (EAN).
The document, announced at the annual ECTRIMS meeting, proposes a standard for vaccination in patients with MS, including a global vaccination strategy for the general MS patient population and selected subpopulations.
The document does not include any recommendations regarding vaccination against COVID-19, which is the subject of a separate report, announced at the annual meeting.
The main conclusions in the new report are as follows:
- Vaccinations in general are considered safe for patients with MS and do not modify disease activity/progression.
- Live attenuated vaccines, however, are contraindicated with immunosuppressants.
- Inactivated vaccines can be used safely, but their efficacy may be decreased with immunosuppressants.
- Vaccinations should be considered early in MS management before using immunosuppressants whenever possible.
Presenting the vaccination consensus document, Susana Otero-Romero, MD, from the Multiple Sclerosis Center of Catalonia, Spain, explained that vaccination has become an important part of the risk management strategy in patients with MS treated with highly active drugs but that questions remain as to when and whether to introduce a particular vaccine and which disease-modifying treatments affect vaccine response.
“The current reference tool has been developed to help professionals to decide on the best vaccination strategy for their patients,” she said.
The consensus document recommends that, in general, vaccination should be performed at the time of diagnosis of MS or in the early stages of the disease to prevent future delays in starting therapies.
“Ideally, vaccination should take place before the onset of disease-modifying treatment,” Dr. Otero-Romero said. The consensus document recommends inactivated vaccines to be given 2-3 weeks before immunosuppressive therapy is started, and live attenuated vaccines at least 4 weeks beforehand.
In the case of relapse, vaccination should be delayed until clinical resolution or stabilization if possible, the consensus statement recommends.
Serological testing for vaccine-induced antibody titers can be performed 1-2 months after the last dose of the vaccine (suggested for hepatitis B, measles, mumps, and varicella). For attenuated live vaccines, serological tests should be done before starting immunosuppressive therapy. In the case of insufficient response, consideration should be given to administering a booster dose of the vaccine, except for hepatitis B, in which a complete revaccination is recommended, according to the document.
As for vaccination during immunosuppressive therapy, this is considered safe for patients on interferon or glatiramer acetate when indicated, the report says.
Vaccination should ideally be avoided in patients on dimethyl fumarate, teriflunomide (Aubagio) or natalizumab (Tysabri), although it can be considered in exceptional cases when the potential risk of acquiring the infection is greater than the risk of developing vaccine-related infections (unless the absolute lymphocyte count is below 800/mm3), it adds.
Vaccination should be avoided in patients on S1P modulators (for example, fingolimod [Gilenya]), anti-CD20 therapies, and before immune restoration for cladribine (Leustatin) and alemtuzumab (Lemtrada).
In the case of patients stopping immunosuppressive therapy, inactivated vaccines can be given any time after the discontinuation of therapy but preferably after immune restoration. Live attenuated vaccines should only be administered after a safety interval ensures immune restoration has been met.
Which vaccines?
On which vaccines are needed in patients with MS, the consensus document recommends the same routine vaccination schedule as for the general population. In addition, it advises influenza and pneumococcal vaccination if patients are immunosuppressed or have significant disability.
It also recommends human papillomavirus vaccine in women and men independent of their age if they are to be treated with alemtuzumab, fingolimod, cladribine, or anti-CD20 drugs. Hepatitis B vaccination is also advised in patients treated with anti-CD20 drugs.
Special populations: pregnancy/elderly
In patients with MS who are pregnant, inactivated flu vaccine can be given in any trimester at the start of the flu season, and vaccination against diphtheria, tetanus, and pertussis can be given during the third trimester, the report says. Live attenuated vaccines should be completed at least 1 month before pregnancy or after delivery and 4-6 weeks prior to the initiation of immunosuppressive therapy.
Elderly patients with MS should receive flu and pneumococcal vaccines annually and would also benefit from the inactivated herpes zoster vaccine.
Travel vaccines
On vaccinations needed for travel, the report recommends that patients with MS consult a specialized travel clinic or vaccination expert and start immunizations 2-3 months before departure. Patients with MS with or without immunosuppressive therapy can receive hepatitis A, rabies, Japanese encephalitis, tic-borne encephalitis, polio, and inactivated typhoid vaccine. But yellow fever and oral typhoid are contraindicated in patients on immunosuppressive therapies.
A version of this article first appeared on Medscape.com.
From ECTRIMS 2021
Facing Up to the Diagnosis
ANSWER
All of these items were rightly considered to be in the differential for this lesion, so the answer is choice “f.”
DISCUSSION
Individuals with type IV skin are less likely than those with types II and III to develop skin cancer. So, although cancer was definitely in the differential, the other items were considered just as, if not more, likely in this case.
The only way to sort through these diagnostic possibilities was to perform a biopsy. In this case, the entire lesion was removed by saucerization technique, under local anesthesia. The specimen provided would be adequate to detect any cancer, which a smaller specimen could easily miss.
The pathology results showed pigmented basal cell carcinoma. Given the patient’s extensive history of sun exposure, and the steady growth of the lesion, this was hardly a surprise. But prior to the biopsy, one could just as easily imagine the lesion to be, for example, a wart.
The take-home message is obvious: Nothing can take the place of biopsy in establishing a precise diagnosis. With that information in hand, the patient was referred for consultation with a Mohs surgeon. Surgical removal and closure would likely set him back several thousand dollars and leave a considerable scar.
ANSWER
All of these items were rightly considered to be in the differential for this lesion, so the answer is choice “f.”
DISCUSSION
Individuals with type IV skin are less likely than those with types II and III to develop skin cancer. So, although cancer was definitely in the differential, the other items were considered just as, if not more, likely in this case.
The only way to sort through these diagnostic possibilities was to perform a biopsy. In this case, the entire lesion was removed by saucerization technique, under local anesthesia. The specimen provided would be adequate to detect any cancer, which a smaller specimen could easily miss.
The pathology results showed pigmented basal cell carcinoma. Given the patient’s extensive history of sun exposure, and the steady growth of the lesion, this was hardly a surprise. But prior to the biopsy, one could just as easily imagine the lesion to be, for example, a wart.
The take-home message is obvious: Nothing can take the place of biopsy in establishing a precise diagnosis. With that information in hand, the patient was referred for consultation with a Mohs surgeon. Surgical removal and closure would likely set him back several thousand dollars and leave a considerable scar.
ANSWER
All of these items were rightly considered to be in the differential for this lesion, so the answer is choice “f.”
DISCUSSION
Individuals with type IV skin are less likely than those with types II and III to develop skin cancer. So, although cancer was definitely in the differential, the other items were considered just as, if not more, likely in this case.
The only way to sort through these diagnostic possibilities was to perform a biopsy. In this case, the entire lesion was removed by saucerization technique, under local anesthesia. The specimen provided would be adequate to detect any cancer, which a smaller specimen could easily miss.
The pathology results showed pigmented basal cell carcinoma. Given the patient’s extensive history of sun exposure, and the steady growth of the lesion, this was hardly a surprise. But prior to the biopsy, one could just as easily imagine the lesion to be, for example, a wart.
The take-home message is obvious: Nothing can take the place of biopsy in establishing a precise diagnosis. With that information in hand, the patient was referred for consultation with a Mohs surgeon. Surgical removal and closure would likely set him back several thousand dollars and leave a considerable scar.
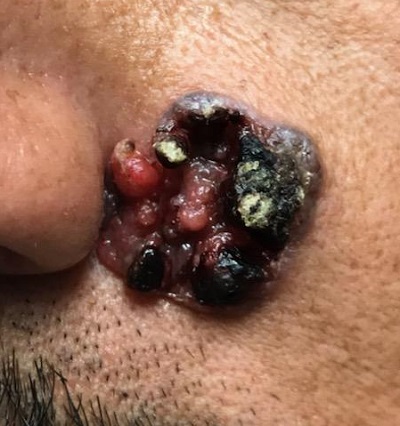
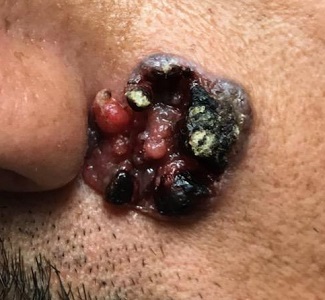
“Like a berry,” was how a 38-year-old Hispanic man described the lesion that had been slowly growing on his face for 4 years. His family was alarmed by it, but he reasoned that since it didn’t hurt and the surrounding area wasn’t especially red, it likely wasn’t much of a problem.
The patient worked as roofer up to 7 days per week when the weather was good and had been doing so since he was old enough to work. He had no insurance and was not inclined to spend money on a health care visit. When his wife finally convinced him to go to the urgent care clinic, he paid $100 just to be told he needed to see a dermatologist. He was so disgusted he almost refused to wait the 6 weeks it took to get into the dermatology office.
The patient, who had type IV skin with little evidence of sun damage, had an obvious, large, coarsely mamillated nodule on the left upper nasolabial area. The lesion measured 1.8 cm and was reddish blue. It was moderately firm, but no increased warmth could be detected. No nodes could be felt in the area. His skin elsewhere was free of any notable changes.
Mortality in 2nd wave higher with ECMO for COVID-ARDS
For patients with refractory acute respiratory distress syndrome (ARDS) caused by COVID-19 infections, extracorporeal membrane oxygenation (ECMO) may be the treatment of last resort.
But for reasons that aren’t clear, in the second wave of the COVID-19 pandemic at a major teaching hospital, the mortality rate of patients on ECMO for COVID-induced ARDS was significantly higher than it was during the first wave, despite changes in drug therapy and clinical management, reported Rohit Reddy, BS, a second-year medical student, and colleagues at Thomas Jefferson University Hospital in Philadelphia.
During the first wave, from April to September 2020, the survival rate of patients while on ECMO in their ICUs was 67%. In contrast, for patients treated during the second wave, from November 2020 to March 2021, the ECMO survival rate was 31% (P = .003).
The 30-day survival rates were also higher in the first wave compared with the second, at 54% versus 31%, but this difference was not statistically significant.
“More research is required to develop stricter inclusion/exclusion criteria and to improve pre-ECMO management in order to improve outcomes,” Mr. Reddy said in a narrated poster presented at the annual meeting of the American College of Chest Physicians, held virtually this year.
ARDS severity higher
ARDS is a major complication of COVID-19 infections, and there is evidence to suggest that COVID-associated ARDS is more severe than ARDS caused by other causes, the investigators noted.
“ECMO, which has been used as a rescue therapy in prior viral outbreaks, has been used to support certain patients with refractory ARDS due to COVID-19, but evidence for its efficacy is limited. Respiratory failure remained a highly concerning complication in the second wave of the COVID-19 pandemic, but it is unclear how the evolution of the disease and pharmacologic utility has affected the clinical utility of ECMO,” Mr. Reddy said.
To see whether changes in disease course or in treatment could explain changes in outcomes for patients with COVID-related ARDS, the investigators compared characteristics and outcomes for patients treated in the first versus second waves of the pandemic. Their study did not include data from patients infected with the Delta variant of the SARS-CoV-2 virus, which became the predominant viral strain later in 2021.
The study included data on 28 patients treated during the first wave, and 13 during the second. The sample included 28 men and 13 women with a mean age of 51 years.
All patients had venovenous ECMO, with cannulation in the femoral or internal jugular veins; some patients received ECMO via a single double-lumen cannula.
There were no significant differences between the two time periods in patient comorbidities prior to initiation of ECMO.
Patients in the second wave were significantly more likely to receive steroids (54% vs. 100%; P = .003) and remdesivir (39% vs. 85%; P = .007). Prone positioning before ECMO was also significantly more frequent in the second wave (11% vs. 85%; P < .001).
Patients in the second wave stayed on ECMO longer – median 20 days versus 14 days for first-wave patients – but as noted before, ECMO mortality rates were significantly higher during the second wave. During the first wave, 33% of patients died while on ECMO, compared with 69% in the second wave (P = .03). Respective 30-day mortality rates were 46% versus 69% (ns).
Rates of complications during ECMO were generally comparable between the groups, including acute renal failure (39% in the first wave vs 38% in the second), sepsis (32% vs. 23%), bacterial pneumonia (11% vs. 8%), and gastrointestinal bleeding (21% vs. 15%). However, significantly more patients in the second wave had cerebral vascular accidents (4% vs. 23%; P = .050).
Senior author Hitoshi Hirose, MD, PhD, professor of surgery at Thomas Jefferson University, said in an interview that the difference in outcomes was likely caused by changes in pre-ECMO therapy between the first and second waves.
“Our study showed the incidence of sepsis had a large impact on the patient outcomes,” he wrote. “We speculate that sepsis was attributed to use of immune modulation therapy. The prevention of the sepsis would be key to improve survival of ECMO for COVID 19.”
“It’s possible that the explanation for this is that patients in the second wave were sicker in a way that wasn’t adequately measured in the first wave,” CHEST 2021 program cochair Christopher Carroll, MD, FCCP, from Connecticut Children’s Medical Center in Hartford, said in an interview.
The differences may also have been attributable to changes in virulence, or to clinical decisions to put sicker patients on ECMO, he said.
Casey Cable, MD, MSc, a pulmonary disease and critical care specialist at Virginia Commonwealth Medical Center in Richmond, also speculated in an interview that second-wave patients may have been sicker.
“One interesting piece of this story is that we now know a lot more – we know about the use of steroids plus or minus remdesivir and proning, and patients received a large majority of those treatments but still got put on ECMO,” she said. “I wonder if there is a subset of really sick patients, and no matter what we treat with – steroids, proning – whatever we do they’re just not going to do well.”
Both Dr. Carroll and Dr. Cable emphasized the importance of ECMO as a rescue therapy for patients with severe, refractory ARDS associated with COVID-19 or other diseases.
Neither Dr. Carroll nor Dr. Cable were involved in the study.
No study funding was reported. Mr. Reddy, Dr. Hirose, Dr. Carroll, and Dr. Cable disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
For patients with refractory acute respiratory distress syndrome (ARDS) caused by COVID-19 infections, extracorporeal membrane oxygenation (ECMO) may be the treatment of last resort.
But for reasons that aren’t clear, in the second wave of the COVID-19 pandemic at a major teaching hospital, the mortality rate of patients on ECMO for COVID-induced ARDS was significantly higher than it was during the first wave, despite changes in drug therapy and clinical management, reported Rohit Reddy, BS, a second-year medical student, and colleagues at Thomas Jefferson University Hospital in Philadelphia.
During the first wave, from April to September 2020, the survival rate of patients while on ECMO in their ICUs was 67%. In contrast, for patients treated during the second wave, from November 2020 to March 2021, the ECMO survival rate was 31% (P = .003).
The 30-day survival rates were also higher in the first wave compared with the second, at 54% versus 31%, but this difference was not statistically significant.
“More research is required to develop stricter inclusion/exclusion criteria and to improve pre-ECMO management in order to improve outcomes,” Mr. Reddy said in a narrated poster presented at the annual meeting of the American College of Chest Physicians, held virtually this year.
ARDS severity higher
ARDS is a major complication of COVID-19 infections, and there is evidence to suggest that COVID-associated ARDS is more severe than ARDS caused by other causes, the investigators noted.
“ECMO, which has been used as a rescue therapy in prior viral outbreaks, has been used to support certain patients with refractory ARDS due to COVID-19, but evidence for its efficacy is limited. Respiratory failure remained a highly concerning complication in the second wave of the COVID-19 pandemic, but it is unclear how the evolution of the disease and pharmacologic utility has affected the clinical utility of ECMO,” Mr. Reddy said.
To see whether changes in disease course or in treatment could explain changes in outcomes for patients with COVID-related ARDS, the investigators compared characteristics and outcomes for patients treated in the first versus second waves of the pandemic. Their study did not include data from patients infected with the Delta variant of the SARS-CoV-2 virus, which became the predominant viral strain later in 2021.
The study included data on 28 patients treated during the first wave, and 13 during the second. The sample included 28 men and 13 women with a mean age of 51 years.
All patients had venovenous ECMO, with cannulation in the femoral or internal jugular veins; some patients received ECMO via a single double-lumen cannula.
There were no significant differences between the two time periods in patient comorbidities prior to initiation of ECMO.
Patients in the second wave were significantly more likely to receive steroids (54% vs. 100%; P = .003) and remdesivir (39% vs. 85%; P = .007). Prone positioning before ECMO was also significantly more frequent in the second wave (11% vs. 85%; P < .001).
Patients in the second wave stayed on ECMO longer – median 20 days versus 14 days for first-wave patients – but as noted before, ECMO mortality rates were significantly higher during the second wave. During the first wave, 33% of patients died while on ECMO, compared with 69% in the second wave (P = .03). Respective 30-day mortality rates were 46% versus 69% (ns).
Rates of complications during ECMO were generally comparable between the groups, including acute renal failure (39% in the first wave vs 38% in the second), sepsis (32% vs. 23%), bacterial pneumonia (11% vs. 8%), and gastrointestinal bleeding (21% vs. 15%). However, significantly more patients in the second wave had cerebral vascular accidents (4% vs. 23%; P = .050).
Senior author Hitoshi Hirose, MD, PhD, professor of surgery at Thomas Jefferson University, said in an interview that the difference in outcomes was likely caused by changes in pre-ECMO therapy between the first and second waves.
“Our study showed the incidence of sepsis had a large impact on the patient outcomes,” he wrote. “We speculate that sepsis was attributed to use of immune modulation therapy. The prevention of the sepsis would be key to improve survival of ECMO for COVID 19.”
“It’s possible that the explanation for this is that patients in the second wave were sicker in a way that wasn’t adequately measured in the first wave,” CHEST 2021 program cochair Christopher Carroll, MD, FCCP, from Connecticut Children’s Medical Center in Hartford, said in an interview.
The differences may also have been attributable to changes in virulence, or to clinical decisions to put sicker patients on ECMO, he said.
Casey Cable, MD, MSc, a pulmonary disease and critical care specialist at Virginia Commonwealth Medical Center in Richmond, also speculated in an interview that second-wave patients may have been sicker.
“One interesting piece of this story is that we now know a lot more – we know about the use of steroids plus or minus remdesivir and proning, and patients received a large majority of those treatments but still got put on ECMO,” she said. “I wonder if there is a subset of really sick patients, and no matter what we treat with – steroids, proning – whatever we do they’re just not going to do well.”
Both Dr. Carroll and Dr. Cable emphasized the importance of ECMO as a rescue therapy for patients with severe, refractory ARDS associated with COVID-19 or other diseases.
Neither Dr. Carroll nor Dr. Cable were involved in the study.
No study funding was reported. Mr. Reddy, Dr. Hirose, Dr. Carroll, and Dr. Cable disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
For patients with refractory acute respiratory distress syndrome (ARDS) caused by COVID-19 infections, extracorporeal membrane oxygenation (ECMO) may be the treatment of last resort.
But for reasons that aren’t clear, in the second wave of the COVID-19 pandemic at a major teaching hospital, the mortality rate of patients on ECMO for COVID-induced ARDS was significantly higher than it was during the first wave, despite changes in drug therapy and clinical management, reported Rohit Reddy, BS, a second-year medical student, and colleagues at Thomas Jefferson University Hospital in Philadelphia.
During the first wave, from April to September 2020, the survival rate of patients while on ECMO in their ICUs was 67%. In contrast, for patients treated during the second wave, from November 2020 to March 2021, the ECMO survival rate was 31% (P = .003).
The 30-day survival rates were also higher in the first wave compared with the second, at 54% versus 31%, but this difference was not statistically significant.
“More research is required to develop stricter inclusion/exclusion criteria and to improve pre-ECMO management in order to improve outcomes,” Mr. Reddy said in a narrated poster presented at the annual meeting of the American College of Chest Physicians, held virtually this year.
ARDS severity higher
ARDS is a major complication of COVID-19 infections, and there is evidence to suggest that COVID-associated ARDS is more severe than ARDS caused by other causes, the investigators noted.
“ECMO, which has been used as a rescue therapy in prior viral outbreaks, has been used to support certain patients with refractory ARDS due to COVID-19, but evidence for its efficacy is limited. Respiratory failure remained a highly concerning complication in the second wave of the COVID-19 pandemic, but it is unclear how the evolution of the disease and pharmacologic utility has affected the clinical utility of ECMO,” Mr. Reddy said.
To see whether changes in disease course or in treatment could explain changes in outcomes for patients with COVID-related ARDS, the investigators compared characteristics and outcomes for patients treated in the first versus second waves of the pandemic. Their study did not include data from patients infected with the Delta variant of the SARS-CoV-2 virus, which became the predominant viral strain later in 2021.
The study included data on 28 patients treated during the first wave, and 13 during the second. The sample included 28 men and 13 women with a mean age of 51 years.
All patients had venovenous ECMO, with cannulation in the femoral or internal jugular veins; some patients received ECMO via a single double-lumen cannula.
There were no significant differences between the two time periods in patient comorbidities prior to initiation of ECMO.
Patients in the second wave were significantly more likely to receive steroids (54% vs. 100%; P = .003) and remdesivir (39% vs. 85%; P = .007). Prone positioning before ECMO was also significantly more frequent in the second wave (11% vs. 85%; P < .001).
Patients in the second wave stayed on ECMO longer – median 20 days versus 14 days for first-wave patients – but as noted before, ECMO mortality rates were significantly higher during the second wave. During the first wave, 33% of patients died while on ECMO, compared with 69% in the second wave (P = .03). Respective 30-day mortality rates were 46% versus 69% (ns).
Rates of complications during ECMO were generally comparable between the groups, including acute renal failure (39% in the first wave vs 38% in the second), sepsis (32% vs. 23%), bacterial pneumonia (11% vs. 8%), and gastrointestinal bleeding (21% vs. 15%). However, significantly more patients in the second wave had cerebral vascular accidents (4% vs. 23%; P = .050).
Senior author Hitoshi Hirose, MD, PhD, professor of surgery at Thomas Jefferson University, said in an interview that the difference in outcomes was likely caused by changes in pre-ECMO therapy between the first and second waves.
“Our study showed the incidence of sepsis had a large impact on the patient outcomes,” he wrote. “We speculate that sepsis was attributed to use of immune modulation therapy. The prevention of the sepsis would be key to improve survival of ECMO for COVID 19.”
“It’s possible that the explanation for this is that patients in the second wave were sicker in a way that wasn’t adequately measured in the first wave,” CHEST 2021 program cochair Christopher Carroll, MD, FCCP, from Connecticut Children’s Medical Center in Hartford, said in an interview.
The differences may also have been attributable to changes in virulence, or to clinical decisions to put sicker patients on ECMO, he said.
Casey Cable, MD, MSc, a pulmonary disease and critical care specialist at Virginia Commonwealth Medical Center in Richmond, also speculated in an interview that second-wave patients may have been sicker.
“One interesting piece of this story is that we now know a lot more – we know about the use of steroids plus or minus remdesivir and proning, and patients received a large majority of those treatments but still got put on ECMO,” she said. “I wonder if there is a subset of really sick patients, and no matter what we treat with – steroids, proning – whatever we do they’re just not going to do well.”
Both Dr. Carroll and Dr. Cable emphasized the importance of ECMO as a rescue therapy for patients with severe, refractory ARDS associated with COVID-19 or other diseases.
Neither Dr. Carroll nor Dr. Cable were involved in the study.
No study funding was reported. Mr. Reddy, Dr. Hirose, Dr. Carroll, and Dr. Cable disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Rituximab more effective than other MS treatments?
, according to new research.
The risk for a first relapse was 6 times higher in patients receiving interferon beta or glatiramer acetate, compared with those receiving rituximab. But the level of disability at 3 years was only marginally different between the drugs studied.
The small differences in Expanded Disability Status Scale (EDSS) score are surprising, said investigator Peter Alping, a clinical assistant and doctoral student in the Department of Clinical Neuroscience at the Karolinska Institutet, Stockholm, as he presented the data. “It could be that we have too-short follow-up, so that EDSS doesn’t have time to diverge between therapies.”
He presented the findings at the annual meeting of the European Committee for Treatment and Research in Multiple Sclerosis (ECTRIMS).
COMBAT-MS study
Direct comparisons of disease-modifying therapies (DMTs) for MS can help neurologists choose the most appropriate treatment for a given patient. To compare the effectiveness of the most common initial DMTs administered in Sweden, the researchers examined data from the COMBAT-MS study.
They identified all patients who initiated an injectable therapy (interferon beta or glatiramer acetate), dimethyl fumarate, natalizumab, or rituximab as a first treatment between Jan. 1, 2011, and Dec. 14, 2020. Eligible participants had prospectively recorded outcome data in the Swedish MS Register. Follow-up for a participant continued even if the participant stopped receiving therapy.
The investigators replaced missing data using multiple imputation. They adjusted for potential confounders using stabilized inverse probability of treatment weighting with baseline variables. These variables included age, sex, disease duration, geographical region, EDSS score, and relapses.
Rituximab reduced relapses
The researchers included 1,938 first-ever treatment episodes in their analysis. Of this group, 858 were associated with injectables, 339 with dimethyl fumarate, 269 with natalizumab, and 472 with rituximab.
Participants’ baseline characteristics differed by the DMT that they used. Patients who initiated natalizumab were the youngest, had the shortest disease duration, and had the most previous relapses.
For each outcome, the investigators compared all other therapies with rituximab. After they adjusted the data, they found that the hazard ratio for first relapse was 6.0 for injectables, 2.9 for dimethyl fumarate, and 1.8 for natalizumab.
In the adjusted model, the MRI lesion rate ratio for injectables, compared with rituximab, was 4.5. The rate ratio was 4.8 for dimethyl fumarate and 1.9 for natalizumab.
But differences in EDSS score at 3 years from treatment initiation were small. EDSS score in patients who received injectables was 0.24 points higher, compared with those receiving rituximab. EDSS score was 0.05 points higher in patients receiving dimethyl fumarate and 0.01 points lower in patients receiving natalizumab.
The risk for treatment discontinuation, however, differed significantly between therapies. The HR for treatment discontinuation was 32.7 for injectables, 20.3 for dimethyl fumarate, and 16.3 for natalizumab, compared with rituximab.
Among patients receiving dimethyl fumarate and injectables, the main reasons for discontinuing therapy were inadequate effect and adverse events. The main reason for discontinuation among patients receiving natalizumab was categorized as “other reason,” which mostly reflected John Cunningham virus positivity and concern for developing progressive multifocal leukoencephalopathy.
‘The uncertainty continues’
“These differences that we see in the effectiveness can be somewhat surprising, especially when it comes to natalizumab,” which is considered very effective, said Mr. Alping. The vulnerable period that occurs after switching from natalizumab may partly explain the difference. “This is something to keep in mind when starting patients on natalizumab treatment in the clinic,” Mr. Alping added.
Although rituximab is not indicated for MS, many clinics are using it in this population, said Robert Fox, MD, staff neurologist at the Mellen Center for MS and vice chair for research at the Neurological Institute of Cleveland Clinic, both in Cleveland, Ohio. Dr. Fox was not involved in the study.
“Assessing the generalizability of the study outside Sweden will be important,” he added, “but I would be surprised if their findings did not hold up to external validation.”
The way that the researchers addressed missing data could affect the interpretation of the findings. “Depending upon how much data was missing, their imputation methods may have a high level of uncertainty,” said Dr. Fox.
The researchers’ adjustments for baseline differences also raise questions. “Even though MRI was an outcome, it doesn’t appear they adjusted for baseline differences in MRI between the groups,” Dr. Fox observed.
Moreover, the study was conducted over a long period of time. “We know there are time effects in MS, with a very different disease activity expected from patients over time,” said Dr. Fox. For example, relapse rates in placebo groups of MS trials tend to decline over time. “This time effect likely impacted their results.”
But the disability findings may be the most important part of the study, according to Dr. Fox. The lack of significant difference in disability progression between therapies “highlights that a couple relapses or lesions on MRI may be too small to translate into long-term differences in disability progression,” he said.
“The long-term implications of small differences in relapse and MRI outcomes may be very small,” Dr. Fox went on. “Thus, the uncertainty continues around escalation treatment versus initial highly effective treatment paradigms.”
The Patient-Centered Outcomes Research Institute, the Swedish Research Council, and NEURO Sweden funded this study. Mr. Alping disclosed no relevant financial relationships. Dr. Fox receives consulting fees from the companies that manufacture all the therapies analyzed in the study.
A version of this article first appeared on Medscape.com.
, according to new research.
The risk for a first relapse was 6 times higher in patients receiving interferon beta or glatiramer acetate, compared with those receiving rituximab. But the level of disability at 3 years was only marginally different between the drugs studied.
The small differences in Expanded Disability Status Scale (EDSS) score are surprising, said investigator Peter Alping, a clinical assistant and doctoral student in the Department of Clinical Neuroscience at the Karolinska Institutet, Stockholm, as he presented the data. “It could be that we have too-short follow-up, so that EDSS doesn’t have time to diverge between therapies.”
He presented the findings at the annual meeting of the European Committee for Treatment and Research in Multiple Sclerosis (ECTRIMS).
COMBAT-MS study
Direct comparisons of disease-modifying therapies (DMTs) for MS can help neurologists choose the most appropriate treatment for a given patient. To compare the effectiveness of the most common initial DMTs administered in Sweden, the researchers examined data from the COMBAT-MS study.
They identified all patients who initiated an injectable therapy (interferon beta or glatiramer acetate), dimethyl fumarate, natalizumab, or rituximab as a first treatment between Jan. 1, 2011, and Dec. 14, 2020. Eligible participants had prospectively recorded outcome data in the Swedish MS Register. Follow-up for a participant continued even if the participant stopped receiving therapy.
The investigators replaced missing data using multiple imputation. They adjusted for potential confounders using stabilized inverse probability of treatment weighting with baseline variables. These variables included age, sex, disease duration, geographical region, EDSS score, and relapses.
Rituximab reduced relapses
The researchers included 1,938 first-ever treatment episodes in their analysis. Of this group, 858 were associated with injectables, 339 with dimethyl fumarate, 269 with natalizumab, and 472 with rituximab.
Participants’ baseline characteristics differed by the DMT that they used. Patients who initiated natalizumab were the youngest, had the shortest disease duration, and had the most previous relapses.
For each outcome, the investigators compared all other therapies with rituximab. After they adjusted the data, they found that the hazard ratio for first relapse was 6.0 for injectables, 2.9 for dimethyl fumarate, and 1.8 for natalizumab.
In the adjusted model, the MRI lesion rate ratio for injectables, compared with rituximab, was 4.5. The rate ratio was 4.8 for dimethyl fumarate and 1.9 for natalizumab.
But differences in EDSS score at 3 years from treatment initiation were small. EDSS score in patients who received injectables was 0.24 points higher, compared with those receiving rituximab. EDSS score was 0.05 points higher in patients receiving dimethyl fumarate and 0.01 points lower in patients receiving natalizumab.
The risk for treatment discontinuation, however, differed significantly between therapies. The HR for treatment discontinuation was 32.7 for injectables, 20.3 for dimethyl fumarate, and 16.3 for natalizumab, compared with rituximab.
Among patients receiving dimethyl fumarate and injectables, the main reasons for discontinuing therapy were inadequate effect and adverse events. The main reason for discontinuation among patients receiving natalizumab was categorized as “other reason,” which mostly reflected John Cunningham virus positivity and concern for developing progressive multifocal leukoencephalopathy.
‘The uncertainty continues’
“These differences that we see in the effectiveness can be somewhat surprising, especially when it comes to natalizumab,” which is considered very effective, said Mr. Alping. The vulnerable period that occurs after switching from natalizumab may partly explain the difference. “This is something to keep in mind when starting patients on natalizumab treatment in the clinic,” Mr. Alping added.
Although rituximab is not indicated for MS, many clinics are using it in this population, said Robert Fox, MD, staff neurologist at the Mellen Center for MS and vice chair for research at the Neurological Institute of Cleveland Clinic, both in Cleveland, Ohio. Dr. Fox was not involved in the study.
“Assessing the generalizability of the study outside Sweden will be important,” he added, “but I would be surprised if their findings did not hold up to external validation.”
The way that the researchers addressed missing data could affect the interpretation of the findings. “Depending upon how much data was missing, their imputation methods may have a high level of uncertainty,” said Dr. Fox.
The researchers’ adjustments for baseline differences also raise questions. “Even though MRI was an outcome, it doesn’t appear they adjusted for baseline differences in MRI between the groups,” Dr. Fox observed.
Moreover, the study was conducted over a long period of time. “We know there are time effects in MS, with a very different disease activity expected from patients over time,” said Dr. Fox. For example, relapse rates in placebo groups of MS trials tend to decline over time. “This time effect likely impacted their results.”
But the disability findings may be the most important part of the study, according to Dr. Fox. The lack of significant difference in disability progression between therapies “highlights that a couple relapses or lesions on MRI may be too small to translate into long-term differences in disability progression,” he said.
“The long-term implications of small differences in relapse and MRI outcomes may be very small,” Dr. Fox went on. “Thus, the uncertainty continues around escalation treatment versus initial highly effective treatment paradigms.”
The Patient-Centered Outcomes Research Institute, the Swedish Research Council, and NEURO Sweden funded this study. Mr. Alping disclosed no relevant financial relationships. Dr. Fox receives consulting fees from the companies that manufacture all the therapies analyzed in the study.
A version of this article first appeared on Medscape.com.
, according to new research.
The risk for a first relapse was 6 times higher in patients receiving interferon beta or glatiramer acetate, compared with those receiving rituximab. But the level of disability at 3 years was only marginally different between the drugs studied.
The small differences in Expanded Disability Status Scale (EDSS) score are surprising, said investigator Peter Alping, a clinical assistant and doctoral student in the Department of Clinical Neuroscience at the Karolinska Institutet, Stockholm, as he presented the data. “It could be that we have too-short follow-up, so that EDSS doesn’t have time to diverge between therapies.”
He presented the findings at the annual meeting of the European Committee for Treatment and Research in Multiple Sclerosis (ECTRIMS).
COMBAT-MS study
Direct comparisons of disease-modifying therapies (DMTs) for MS can help neurologists choose the most appropriate treatment for a given patient. To compare the effectiveness of the most common initial DMTs administered in Sweden, the researchers examined data from the COMBAT-MS study.
They identified all patients who initiated an injectable therapy (interferon beta or glatiramer acetate), dimethyl fumarate, natalizumab, or rituximab as a first treatment between Jan. 1, 2011, and Dec. 14, 2020. Eligible participants had prospectively recorded outcome data in the Swedish MS Register. Follow-up for a participant continued even if the participant stopped receiving therapy.
The investigators replaced missing data using multiple imputation. They adjusted for potential confounders using stabilized inverse probability of treatment weighting with baseline variables. These variables included age, sex, disease duration, geographical region, EDSS score, and relapses.
Rituximab reduced relapses
The researchers included 1,938 first-ever treatment episodes in their analysis. Of this group, 858 were associated with injectables, 339 with dimethyl fumarate, 269 with natalizumab, and 472 with rituximab.
Participants’ baseline characteristics differed by the DMT that they used. Patients who initiated natalizumab were the youngest, had the shortest disease duration, and had the most previous relapses.
For each outcome, the investigators compared all other therapies with rituximab. After they adjusted the data, they found that the hazard ratio for first relapse was 6.0 for injectables, 2.9 for dimethyl fumarate, and 1.8 for natalizumab.
In the adjusted model, the MRI lesion rate ratio for injectables, compared with rituximab, was 4.5. The rate ratio was 4.8 for dimethyl fumarate and 1.9 for natalizumab.
But differences in EDSS score at 3 years from treatment initiation were small. EDSS score in patients who received injectables was 0.24 points higher, compared with those receiving rituximab. EDSS score was 0.05 points higher in patients receiving dimethyl fumarate and 0.01 points lower in patients receiving natalizumab.
The risk for treatment discontinuation, however, differed significantly between therapies. The HR for treatment discontinuation was 32.7 for injectables, 20.3 for dimethyl fumarate, and 16.3 for natalizumab, compared with rituximab.
Among patients receiving dimethyl fumarate and injectables, the main reasons for discontinuing therapy were inadequate effect and adverse events. The main reason for discontinuation among patients receiving natalizumab was categorized as “other reason,” which mostly reflected John Cunningham virus positivity and concern for developing progressive multifocal leukoencephalopathy.
‘The uncertainty continues’
“These differences that we see in the effectiveness can be somewhat surprising, especially when it comes to natalizumab,” which is considered very effective, said Mr. Alping. The vulnerable period that occurs after switching from natalizumab may partly explain the difference. “This is something to keep in mind when starting patients on natalizumab treatment in the clinic,” Mr. Alping added.
Although rituximab is not indicated for MS, many clinics are using it in this population, said Robert Fox, MD, staff neurologist at the Mellen Center for MS and vice chair for research at the Neurological Institute of Cleveland Clinic, both in Cleveland, Ohio. Dr. Fox was not involved in the study.
“Assessing the generalizability of the study outside Sweden will be important,” he added, “but I would be surprised if their findings did not hold up to external validation.”
The way that the researchers addressed missing data could affect the interpretation of the findings. “Depending upon how much data was missing, their imputation methods may have a high level of uncertainty,” said Dr. Fox.
The researchers’ adjustments for baseline differences also raise questions. “Even though MRI was an outcome, it doesn’t appear they adjusted for baseline differences in MRI between the groups,” Dr. Fox observed.
Moreover, the study was conducted over a long period of time. “We know there are time effects in MS, with a very different disease activity expected from patients over time,” said Dr. Fox. For example, relapse rates in placebo groups of MS trials tend to decline over time. “This time effect likely impacted their results.”
But the disability findings may be the most important part of the study, according to Dr. Fox. The lack of significant difference in disability progression between therapies “highlights that a couple relapses or lesions on MRI may be too small to translate into long-term differences in disability progression,” he said.
“The long-term implications of small differences in relapse and MRI outcomes may be very small,” Dr. Fox went on. “Thus, the uncertainty continues around escalation treatment versus initial highly effective treatment paradigms.”
The Patient-Centered Outcomes Research Institute, the Swedish Research Council, and NEURO Sweden funded this study. Mr. Alping disclosed no relevant financial relationships. Dr. Fox receives consulting fees from the companies that manufacture all the therapies analyzed in the study.
A version of this article first appeared on Medscape.com.
From ECTRIMS 2021
States can reserve COVID shots for kids 5-11 this week
States can preorder COVID-19 vaccine doses for younger children this week as they begin to set up vaccination campaigns for ages 5-11.
Vaccine advisory groups for the FDA and CDC are scheduled to discuss and approve the Pfizer shot for kids in the next three weeks. To help states and cities prepare for the rollout, the CDC issued guidance on how to set up expanded vaccination programs.
Immunization program managers can begin ordering doses on Wednesday, according to the guidance. The vials won’t be delivered until the FDA and CDC authorize the shot, but registering now will help federal officials ship doses quickly once they’re available.
Pharmacies in every state will be able to give COVID-19 shots to children, but they can only use doses that are prepared specifically for children. Ages 5-11 will need a 10-microgram dose, which is one-third of the dose administered to ages 12 and older. The guidance warns that doctors should not try to split up or fraction the adult doses.
The CDC guidance also recommends that pediatricians and family practice doctors should serve as primary places to give shots to kids. The document mentions other options, such as vaccination clinics at schools, but doesn’t endorse them as the first choice for vaccinating kids.
The CDC hasn’t yet addressed questions around whether kids should be required to get vaccinated to attend school. The decision will likely be left to state and city officials.
Federal health officials aren’t yet sure how many parents and guardians will seek shots for their younger kids right away, the AP reported. Demand may be high at first for some families, but it may not be as high as when shots first became available for adults, Marcus Plescia, MD, chief medical officer of the Association of State and Territorial Health Officials, told The Associated Press.
“We’re going to have potentially a very busy, and perhaps modestly chaotic time,” he said.
When vaccines were first authorized for adults, hospitals and pharmacies received priority for ordering shots. Some doctors felt left out. This time, however, the CDC has said that pediatricians will receive higher priority and be able to receive shipments quickly.
As the vaccine rollout begins, health officials should consider logistical concerns to address racial and economic disparities for younger kids, Richard Besser, MD, president and CEO of the Robert Wood Johnson Foundation and a former acting director of the CDC, told the AP.
If parents or guardians can’t leave work to take their kids to a pharmacy or doctor’s office, for instance, their kids may not receive a shot quickly – or at all.
“It’s really important that we recognize the barriers to vaccinations,” he said.
A version of this article first appeared on WebMD.com.
States can preorder COVID-19 vaccine doses for younger children this week as they begin to set up vaccination campaigns for ages 5-11.
Vaccine advisory groups for the FDA and CDC are scheduled to discuss and approve the Pfizer shot for kids in the next three weeks. To help states and cities prepare for the rollout, the CDC issued guidance on how to set up expanded vaccination programs.
Immunization program managers can begin ordering doses on Wednesday, according to the guidance. The vials won’t be delivered until the FDA and CDC authorize the shot, but registering now will help federal officials ship doses quickly once they’re available.
Pharmacies in every state will be able to give COVID-19 shots to children, but they can only use doses that are prepared specifically for children. Ages 5-11 will need a 10-microgram dose, which is one-third of the dose administered to ages 12 and older. The guidance warns that doctors should not try to split up or fraction the adult doses.
The CDC guidance also recommends that pediatricians and family practice doctors should serve as primary places to give shots to kids. The document mentions other options, such as vaccination clinics at schools, but doesn’t endorse them as the first choice for vaccinating kids.
The CDC hasn’t yet addressed questions around whether kids should be required to get vaccinated to attend school. The decision will likely be left to state and city officials.
Federal health officials aren’t yet sure how many parents and guardians will seek shots for their younger kids right away, the AP reported. Demand may be high at first for some families, but it may not be as high as when shots first became available for adults, Marcus Plescia, MD, chief medical officer of the Association of State and Territorial Health Officials, told The Associated Press.
“We’re going to have potentially a very busy, and perhaps modestly chaotic time,” he said.
When vaccines were first authorized for adults, hospitals and pharmacies received priority for ordering shots. Some doctors felt left out. This time, however, the CDC has said that pediatricians will receive higher priority and be able to receive shipments quickly.
As the vaccine rollout begins, health officials should consider logistical concerns to address racial and economic disparities for younger kids, Richard Besser, MD, president and CEO of the Robert Wood Johnson Foundation and a former acting director of the CDC, told the AP.
If parents or guardians can’t leave work to take their kids to a pharmacy or doctor’s office, for instance, their kids may not receive a shot quickly – or at all.
“It’s really important that we recognize the barriers to vaccinations,” he said.
A version of this article first appeared on WebMD.com.
States can preorder COVID-19 vaccine doses for younger children this week as they begin to set up vaccination campaigns for ages 5-11.
Vaccine advisory groups for the FDA and CDC are scheduled to discuss and approve the Pfizer shot for kids in the next three weeks. To help states and cities prepare for the rollout, the CDC issued guidance on how to set up expanded vaccination programs.
Immunization program managers can begin ordering doses on Wednesday, according to the guidance. The vials won’t be delivered until the FDA and CDC authorize the shot, but registering now will help federal officials ship doses quickly once they’re available.
Pharmacies in every state will be able to give COVID-19 shots to children, but they can only use doses that are prepared specifically for children. Ages 5-11 will need a 10-microgram dose, which is one-third of the dose administered to ages 12 and older. The guidance warns that doctors should not try to split up or fraction the adult doses.
The CDC guidance also recommends that pediatricians and family practice doctors should serve as primary places to give shots to kids. The document mentions other options, such as vaccination clinics at schools, but doesn’t endorse them as the first choice for vaccinating kids.
The CDC hasn’t yet addressed questions around whether kids should be required to get vaccinated to attend school. The decision will likely be left to state and city officials.
Federal health officials aren’t yet sure how many parents and guardians will seek shots for their younger kids right away, the AP reported. Demand may be high at first for some families, but it may not be as high as when shots first became available for adults, Marcus Plescia, MD, chief medical officer of the Association of State and Territorial Health Officials, told The Associated Press.
“We’re going to have potentially a very busy, and perhaps modestly chaotic time,” he said.
When vaccines were first authorized for adults, hospitals and pharmacies received priority for ordering shots. Some doctors felt left out. This time, however, the CDC has said that pediatricians will receive higher priority and be able to receive shipments quickly.
As the vaccine rollout begins, health officials should consider logistical concerns to address racial and economic disparities for younger kids, Richard Besser, MD, president and CEO of the Robert Wood Johnson Foundation and a former acting director of the CDC, told the AP.
If parents or guardians can’t leave work to take their kids to a pharmacy or doctor’s office, for instance, their kids may not receive a shot quickly – or at all.
“It’s really important that we recognize the barriers to vaccinations,” he said.
A version of this article first appeared on WebMD.com.
Sleep problems in mental illness highly pervasive
An inpatient psychiatric diagnosis at some point over a lifetime is significantly associated with a range of sleep problems, results from the largest study of its kind show.
A prior diagnosis of major depression, schizophrenia, anxiety, or bipolar disorder was associated with a later bedtime, earlier waking time, and significantly poorer sleep quality that included frequent awakenings during the night and shorter sleep bouts.
“We were struck by the pervasiveness of sleep problems across all the diagnoses of mental illness and sleep parameters we looked at,” study investigator Michael Wainberg, PhD, a postdoctoral fellow at the Krembil Centre for Neuroinformatics at the Center for Addiction and Mental Health (CAMH), Toronto, told this news organization. “This suggests there may need to be even more of an emphasis on sleep in these patients than there already is.”
The study, which includes data from nearly 90,000 adults in the United Kingdom, was published online October 12 in PLoS Medicine.
Trove of data
Data for the analysis comes from the UK Biobank, a large-scale biomedical database launched in 2006 that has collected biological and medical data on more than 500,000 individuals who consented to provide blood, urine, and saliva samples and detailed lifestyle information that is matched to their medical records.
Between 2013 and 2015, more than 103,000 of these participants agreed to wear accelerometers on their wrists for 24 hours a day for 7 days, collecting a trove of data for researchers to mine.
“This allows us to get at objectively derived sleep measures and to measure them in greater numbers of people who have experienced mental illness,” said senior author Shreejoy Tripathy, PhD, assistant professor at the University of Toronto and independent scientist for CAMH. “You can study multiple disorders at once and the influence of other variables that might not be possible in the context of other studies.”
The research is the first known large-scale transdiagnostic study of objectively measured sleep and mental health. Insomnia and other sleep disorders are common among people with mental illness, as shown in prior research, including at least one study that used the same dataset the team employed for this project.
The new findings add to that body of work, Dr. Wainberg said, and look beyond just how long a person sleeps to the quality of the sleep they get.
“We found that the metrics of sleep quality seem to be affected more than mere sleep duration,” he said.
Unexpected finding
After excluding participants with faulty accelerometers and those who didn’t wear them for the entire 7-day study period, data from 89,205 participants (aged 43-79, 56% female, 97% self-reported White) was included. Lifetime inpatient psychiatric diagnoses were reported in 2.5% of the entire cohort.
Researchers looked at 10 sleep measures: bedtime, wake-up time, sleep duration, wake after sleep onset, sleep efficiency, number of awakenings, duration of longest sleep bout, number of naps, and variability in bedtime and sleep duration.
Although the effect sizes were small, having any psychiatric diagnosis was associated with significantly lower scores on every sleep measure except sleep duration.
Compared with those with no inpatient psychiatric diagnosis, those with any psychiatric diagnosis were significantly more likely to:
- have a later bedtime (beta = 0.07; 95% confidence interval, 0.06-0.09)
- have later wake-up time (beta = 0.10; 95% CI, 0.09-0.11)
- wake after sleep onset (beta = 0.10; 95% CI, 0.09-0.12)
- have poorer sleep efficiency (beta = –0.12; 95% CI, −0.14 to −0.11)
- have more awakenings (beta = 0.10; 95% CI, 0.09-0.11)
- have shorter duration of their longest sleep bout (beta = –0.09; 95% CI, −0.11 to −0.08)
- take more naps (beta = 0.11; 95% CI, 0.09-0.12)
- have greater variability in their bedtime (beta = 0.08; 95% CI, 0.06-0.09)
- have greater variability in their sleep duration (beta = 0.10; 95% CI, 0.09-0.12)
The only significant differences in sleep duration were found in those with lifetime major depressive disorder, who slept significantly less (beta = −0.02; P = .003), and in those with lifetime schizophrenia, who slept significantly longer (beta = 0.02; P = .0008).
Researchers found similar results when they examined patient-reported sleep measures collected when participants enrolled in the biobank, long before they agreed to wear an accelerometer.
“Everyone with a lifetime mental illness diagnosis trended toward worse sleep quality, regardless of their diagnosis,” Dr. Tripathy said. “We didn’t expect to see that.”
Limitations of the biobank data prohibited analysis by age and past or current use of psychiatric medications. In addition, investigators were unable to determine whether mental illness was active or controlled at the time of the study. Information on these, and other factors, is needed to truly begin to understand the real-world status of sleep patterns in people with mental illness, the researchers note.
However, the biobank data demonstrates how this type of information can be collected, helping Dr. Tripathy and others to design a new study that will launch next year with patients at CAMH. This effort is part of the BrainHealth Databank, a project that aims to develop a patient data bank similar to the one in the UK that was used for this study.
“We’ve shown that you can use wearable devices to measure correlates of sleep and derive insights about the objective measurements of sleep and associate them with mental illness diagnosis,” Dr. Tripathy said.
The study received no outside funding. Dr. Wainberg and Dr. Tripathy report receiving funding from Kavli Foundation, Krembil Foundation, CAMH Discovery Fund, the McLaughlin Foundation, NSERC, and CIHR. Disclosures for other authors are fully listed in the original article.
A version of this article first appeared on Medscape.com.
An inpatient psychiatric diagnosis at some point over a lifetime is significantly associated with a range of sleep problems, results from the largest study of its kind show.
A prior diagnosis of major depression, schizophrenia, anxiety, or bipolar disorder was associated with a later bedtime, earlier waking time, and significantly poorer sleep quality that included frequent awakenings during the night and shorter sleep bouts.
“We were struck by the pervasiveness of sleep problems across all the diagnoses of mental illness and sleep parameters we looked at,” study investigator Michael Wainberg, PhD, a postdoctoral fellow at the Krembil Centre for Neuroinformatics at the Center for Addiction and Mental Health (CAMH), Toronto, told this news organization. “This suggests there may need to be even more of an emphasis on sleep in these patients than there already is.”
The study, which includes data from nearly 90,000 adults in the United Kingdom, was published online October 12 in PLoS Medicine.
Trove of data
Data for the analysis comes from the UK Biobank, a large-scale biomedical database launched in 2006 that has collected biological and medical data on more than 500,000 individuals who consented to provide blood, urine, and saliva samples and detailed lifestyle information that is matched to their medical records.
Between 2013 and 2015, more than 103,000 of these participants agreed to wear accelerometers on their wrists for 24 hours a day for 7 days, collecting a trove of data for researchers to mine.
“This allows us to get at objectively derived sleep measures and to measure them in greater numbers of people who have experienced mental illness,” said senior author Shreejoy Tripathy, PhD, assistant professor at the University of Toronto and independent scientist for CAMH. “You can study multiple disorders at once and the influence of other variables that might not be possible in the context of other studies.”
The research is the first known large-scale transdiagnostic study of objectively measured sleep and mental health. Insomnia and other sleep disorders are common among people with mental illness, as shown in prior research, including at least one study that used the same dataset the team employed for this project.
The new findings add to that body of work, Dr. Wainberg said, and look beyond just how long a person sleeps to the quality of the sleep they get.
“We found that the metrics of sleep quality seem to be affected more than mere sleep duration,” he said.
Unexpected finding
After excluding participants with faulty accelerometers and those who didn’t wear them for the entire 7-day study period, data from 89,205 participants (aged 43-79, 56% female, 97% self-reported White) was included. Lifetime inpatient psychiatric diagnoses were reported in 2.5% of the entire cohort.
Researchers looked at 10 sleep measures: bedtime, wake-up time, sleep duration, wake after sleep onset, sleep efficiency, number of awakenings, duration of longest sleep bout, number of naps, and variability in bedtime and sleep duration.
Although the effect sizes were small, having any psychiatric diagnosis was associated with significantly lower scores on every sleep measure except sleep duration.
Compared with those with no inpatient psychiatric diagnosis, those with any psychiatric diagnosis were significantly more likely to:
- have a later bedtime (beta = 0.07; 95% confidence interval, 0.06-0.09)
- have later wake-up time (beta = 0.10; 95% CI, 0.09-0.11)
- wake after sleep onset (beta = 0.10; 95% CI, 0.09-0.12)
- have poorer sleep efficiency (beta = –0.12; 95% CI, −0.14 to −0.11)
- have more awakenings (beta = 0.10; 95% CI, 0.09-0.11)
- have shorter duration of their longest sleep bout (beta = –0.09; 95% CI, −0.11 to −0.08)
- take more naps (beta = 0.11; 95% CI, 0.09-0.12)
- have greater variability in their bedtime (beta = 0.08; 95% CI, 0.06-0.09)
- have greater variability in their sleep duration (beta = 0.10; 95% CI, 0.09-0.12)
The only significant differences in sleep duration were found in those with lifetime major depressive disorder, who slept significantly less (beta = −0.02; P = .003), and in those with lifetime schizophrenia, who slept significantly longer (beta = 0.02; P = .0008).
Researchers found similar results when they examined patient-reported sleep measures collected when participants enrolled in the biobank, long before they agreed to wear an accelerometer.
“Everyone with a lifetime mental illness diagnosis trended toward worse sleep quality, regardless of their diagnosis,” Dr. Tripathy said. “We didn’t expect to see that.”
Limitations of the biobank data prohibited analysis by age and past or current use of psychiatric medications. In addition, investigators were unable to determine whether mental illness was active or controlled at the time of the study. Information on these, and other factors, is needed to truly begin to understand the real-world status of sleep patterns in people with mental illness, the researchers note.
However, the biobank data demonstrates how this type of information can be collected, helping Dr. Tripathy and others to design a new study that will launch next year with patients at CAMH. This effort is part of the BrainHealth Databank, a project that aims to develop a patient data bank similar to the one in the UK that was used for this study.
“We’ve shown that you can use wearable devices to measure correlates of sleep and derive insights about the objective measurements of sleep and associate them with mental illness diagnosis,” Dr. Tripathy said.
The study received no outside funding. Dr. Wainberg and Dr. Tripathy report receiving funding from Kavli Foundation, Krembil Foundation, CAMH Discovery Fund, the McLaughlin Foundation, NSERC, and CIHR. Disclosures for other authors are fully listed in the original article.
A version of this article first appeared on Medscape.com.
An inpatient psychiatric diagnosis at some point over a lifetime is significantly associated with a range of sleep problems, results from the largest study of its kind show.
A prior diagnosis of major depression, schizophrenia, anxiety, or bipolar disorder was associated with a later bedtime, earlier waking time, and significantly poorer sleep quality that included frequent awakenings during the night and shorter sleep bouts.
“We were struck by the pervasiveness of sleep problems across all the diagnoses of mental illness and sleep parameters we looked at,” study investigator Michael Wainberg, PhD, a postdoctoral fellow at the Krembil Centre for Neuroinformatics at the Center for Addiction and Mental Health (CAMH), Toronto, told this news organization. “This suggests there may need to be even more of an emphasis on sleep in these patients than there already is.”
The study, which includes data from nearly 90,000 adults in the United Kingdom, was published online October 12 in PLoS Medicine.
Trove of data
Data for the analysis comes from the UK Biobank, a large-scale biomedical database launched in 2006 that has collected biological and medical data on more than 500,000 individuals who consented to provide blood, urine, and saliva samples and detailed lifestyle information that is matched to their medical records.
Between 2013 and 2015, more than 103,000 of these participants agreed to wear accelerometers on their wrists for 24 hours a day for 7 days, collecting a trove of data for researchers to mine.
“This allows us to get at objectively derived sleep measures and to measure them in greater numbers of people who have experienced mental illness,” said senior author Shreejoy Tripathy, PhD, assistant professor at the University of Toronto and independent scientist for CAMH. “You can study multiple disorders at once and the influence of other variables that might not be possible in the context of other studies.”
The research is the first known large-scale transdiagnostic study of objectively measured sleep and mental health. Insomnia and other sleep disorders are common among people with mental illness, as shown in prior research, including at least one study that used the same dataset the team employed for this project.
The new findings add to that body of work, Dr. Wainberg said, and look beyond just how long a person sleeps to the quality of the sleep they get.
“We found that the metrics of sleep quality seem to be affected more than mere sleep duration,” he said.
Unexpected finding
After excluding participants with faulty accelerometers and those who didn’t wear them for the entire 7-day study period, data from 89,205 participants (aged 43-79, 56% female, 97% self-reported White) was included. Lifetime inpatient psychiatric diagnoses were reported in 2.5% of the entire cohort.
Researchers looked at 10 sleep measures: bedtime, wake-up time, sleep duration, wake after sleep onset, sleep efficiency, number of awakenings, duration of longest sleep bout, number of naps, and variability in bedtime and sleep duration.
Although the effect sizes were small, having any psychiatric diagnosis was associated with significantly lower scores on every sleep measure except sleep duration.
Compared with those with no inpatient psychiatric diagnosis, those with any psychiatric diagnosis were significantly more likely to:
- have a later bedtime (beta = 0.07; 95% confidence interval, 0.06-0.09)
- have later wake-up time (beta = 0.10; 95% CI, 0.09-0.11)
- wake after sleep onset (beta = 0.10; 95% CI, 0.09-0.12)
- have poorer sleep efficiency (beta = –0.12; 95% CI, −0.14 to −0.11)
- have more awakenings (beta = 0.10; 95% CI, 0.09-0.11)
- have shorter duration of their longest sleep bout (beta = –0.09; 95% CI, −0.11 to −0.08)
- take more naps (beta = 0.11; 95% CI, 0.09-0.12)
- have greater variability in their bedtime (beta = 0.08; 95% CI, 0.06-0.09)
- have greater variability in their sleep duration (beta = 0.10; 95% CI, 0.09-0.12)
The only significant differences in sleep duration were found in those with lifetime major depressive disorder, who slept significantly less (beta = −0.02; P = .003), and in those with lifetime schizophrenia, who slept significantly longer (beta = 0.02; P = .0008).
Researchers found similar results when they examined patient-reported sleep measures collected when participants enrolled in the biobank, long before they agreed to wear an accelerometer.
“Everyone with a lifetime mental illness diagnosis trended toward worse sleep quality, regardless of their diagnosis,” Dr. Tripathy said. “We didn’t expect to see that.”
Limitations of the biobank data prohibited analysis by age and past or current use of psychiatric medications. In addition, investigators were unable to determine whether mental illness was active or controlled at the time of the study. Information on these, and other factors, is needed to truly begin to understand the real-world status of sleep patterns in people with mental illness, the researchers note.
However, the biobank data demonstrates how this type of information can be collected, helping Dr. Tripathy and others to design a new study that will launch next year with patients at CAMH. This effort is part of the BrainHealth Databank, a project that aims to develop a patient data bank similar to the one in the UK that was used for this study.
“We’ve shown that you can use wearable devices to measure correlates of sleep and derive insights about the objective measurements of sleep and associate them with mental illness diagnosis,” Dr. Tripathy said.
The study received no outside funding. Dr. Wainberg and Dr. Tripathy report receiving funding from Kavli Foundation, Krembil Foundation, CAMH Discovery Fund, the McLaughlin Foundation, NSERC, and CIHR. Disclosures for other authors are fully listed in the original article.
A version of this article first appeared on Medscape.com.
Art therapy linked to slowed Parkinson’s progression
Adding art therapy to standard drug treatment in Parkinson’s disease (PD) not only improves severity of both motor and nonmotor symptoms, but also slows rates of disease progression, new research suggests.
Fifty PD patients were randomly assigned to receive either art therapy, including sculpting and drawing, plus drug therapy or drug therapy alone, and followed up over 12 months.
Patients receiving combined therapy experienced improvements in symptoms, depression, and cognitive scores, and had reduced tremor and daytime sleepiness. They were also substantially less likely to experience disease progression.
“The use of art therapy can reduce the severity of motor and nonmotor manifestations of Parkinson’s disease,” said study investigator Iryna Khubetova, MD, PhD, head of the neurology department, Odessa (Ukraine) Regional Clinical Hospital.
she added.
The findings were presented at the virtual congress of the European College of Neuropsychopharmacology.
A promising approach
Dr. Khubetova told this news organization that offering art therapy to PD patients was “very affordable,” especially as professional artists “provided materials for painting and other art supplies free of charge.”
“We hope this approach is very promising and would be widely adopted.”
She suggested the positive effect of art therapy could be related to “activating the brain’s reward neural network.”
This may be via improved visual attention acting on visuospatial mechanisms and emotional drive, with “activation of the medial orbitofrontal cortex, ventral striatum, and other structures.”
The researchers note PD, a “multisystem progressive neurodegenerative disease,” is among the three most common neurological disorders, with an incidence of 100-150 cases per 100,000 people.
They also note that nonpharmacologic approaches are “widely used” as an adjunct to drug therapy and as part of an “integrated approach” to disease management.
To examine the clinical efficacy of art therapy, the team recruited patients with PD who had preserved facility for independent movement, defined as stages 1-2.5 on the Hoehn and Yahr scale.
Patients were randomly assigned to art therapy sessions alongside standard drug therapy or to standard drug therapy alone. The art therapy included sculpting, free drawing, and coloring patterns.
Multiple benefits
Participants were assessed at baseline and at 6 and 12 months with the Unified Parkinson Disease Rating Scale (UPDRS), the Beck Depression Inventory, the Montreal Cognitive Assessment, and the Pegboard Test of finger dexterity.
Fifty patients were included in the study, with 30 assigned to standard drug therapy alone and 20 to the combined intervention. Participants had a mean age of 57.8 years, and 46% were women.
Over the study period, investigators found patients assigned to art therapy plus drug treatment had improved mood, as well as decreased daytime sleeping, reduced tremor, and a decrease in anxiety and fear intensity.
Between baseline and the 6- and 12-month assessments, patients in the combined therapy group showed improvements in scores on all of the questionnaires, and on the Pegboard Test. In contrast, scores were either stable or worsened in the standard drug therapy–alone group.
The team notes that there was also a marked difference in rates of disease progression, defined as a change on the Hoehn and Yahr scale of at least 0.5 points, between the two groups.
Only two (10%) patients in the combined drug and art therapy progressed over the study period, compared with 10 (33%) in the control group (P = .05).
The findings complement those of a recent study conducted by Alberto Cucca, MD, of the Fresco Institute for Parkinson’s and Movement Disorders, New York University, and colleagues.
Eighteen patients took part in the prospective, open-label trial. They were assessed before and after 20 sessions of art therapy on a range of measures.
Results revealed that following the art therapy, patients had improvements in the Navon Test (which assesses visual neglect, eye tracking, and UPDRS scores), as well as significantly increased functional connectivity levels in the visual cortex on resting-state functional MRI.
Many benefits, no side effects
Rebecca Gilbert, MD, PhD, vice president and chief scientific officer of the American Parkinson Disease Association, who was not involved in either study, told this news organization that the idea of art therapy for patients with Parkinson’s is “very reasonable.”
She highlighted that “people with Parkinson’s have many issues with their visuospatial abilities,” as well as their depth and distance perception, and so “enhancing that aspect could potentially be very beneficial.”
“So I’m hopeful that it’s a really good avenue to explore, and the preliminary data are very exciting.”
Dr. Gilbert also highlighted that the “wonderful” aspect of art therapy is that there are “so many benefits and not really any side effects.” Patients can “take the meds … and then enhance that with various therapies, and this would be an additional option.”
Another notable aspect of art therapy is the “social element” and the sense of “camaraderie,” although that has “to be teased out from the benefits you would get from the actual art therapy.”
Finally, Dr. Gilbert pointed out that the difference between the current trial and Dr. Cucca’s trial is the presence of a control group.
“Of course, it’s not blinded, because you know whether you got therapy or not … but that extra element of being able to compare with a group that didn’t get the treatment gives it a little more weight in terms of the field.”
No funding was declared. The authors have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Adding art therapy to standard drug treatment in Parkinson’s disease (PD) not only improves severity of both motor and nonmotor symptoms, but also slows rates of disease progression, new research suggests.
Fifty PD patients were randomly assigned to receive either art therapy, including sculpting and drawing, plus drug therapy or drug therapy alone, and followed up over 12 months.
Patients receiving combined therapy experienced improvements in symptoms, depression, and cognitive scores, and had reduced tremor and daytime sleepiness. They were also substantially less likely to experience disease progression.
“The use of art therapy can reduce the severity of motor and nonmotor manifestations of Parkinson’s disease,” said study investigator Iryna Khubetova, MD, PhD, head of the neurology department, Odessa (Ukraine) Regional Clinical Hospital.
she added.
The findings were presented at the virtual congress of the European College of Neuropsychopharmacology.
A promising approach
Dr. Khubetova told this news organization that offering art therapy to PD patients was “very affordable,” especially as professional artists “provided materials for painting and other art supplies free of charge.”
“We hope this approach is very promising and would be widely adopted.”
She suggested the positive effect of art therapy could be related to “activating the brain’s reward neural network.”
This may be via improved visual attention acting on visuospatial mechanisms and emotional drive, with “activation of the medial orbitofrontal cortex, ventral striatum, and other structures.”
The researchers note PD, a “multisystem progressive neurodegenerative disease,” is among the three most common neurological disorders, with an incidence of 100-150 cases per 100,000 people.
They also note that nonpharmacologic approaches are “widely used” as an adjunct to drug therapy and as part of an “integrated approach” to disease management.
To examine the clinical efficacy of art therapy, the team recruited patients with PD who had preserved facility for independent movement, defined as stages 1-2.5 on the Hoehn and Yahr scale.
Patients were randomly assigned to art therapy sessions alongside standard drug therapy or to standard drug therapy alone. The art therapy included sculpting, free drawing, and coloring patterns.
Multiple benefits
Participants were assessed at baseline and at 6 and 12 months with the Unified Parkinson Disease Rating Scale (UPDRS), the Beck Depression Inventory, the Montreal Cognitive Assessment, and the Pegboard Test of finger dexterity.
Fifty patients were included in the study, with 30 assigned to standard drug therapy alone and 20 to the combined intervention. Participants had a mean age of 57.8 years, and 46% were women.
Over the study period, investigators found patients assigned to art therapy plus drug treatment had improved mood, as well as decreased daytime sleeping, reduced tremor, and a decrease in anxiety and fear intensity.
Between baseline and the 6- and 12-month assessments, patients in the combined therapy group showed improvements in scores on all of the questionnaires, and on the Pegboard Test. In contrast, scores were either stable or worsened in the standard drug therapy–alone group.
The team notes that there was also a marked difference in rates of disease progression, defined as a change on the Hoehn and Yahr scale of at least 0.5 points, between the two groups.
Only two (10%) patients in the combined drug and art therapy progressed over the study period, compared with 10 (33%) in the control group (P = .05).
The findings complement those of a recent study conducted by Alberto Cucca, MD, of the Fresco Institute for Parkinson’s and Movement Disorders, New York University, and colleagues.
Eighteen patients took part in the prospective, open-label trial. They were assessed before and after 20 sessions of art therapy on a range of measures.
Results revealed that following the art therapy, patients had improvements in the Navon Test (which assesses visual neglect, eye tracking, and UPDRS scores), as well as significantly increased functional connectivity levels in the visual cortex on resting-state functional MRI.
Many benefits, no side effects
Rebecca Gilbert, MD, PhD, vice president and chief scientific officer of the American Parkinson Disease Association, who was not involved in either study, told this news organization that the idea of art therapy for patients with Parkinson’s is “very reasonable.”
She highlighted that “people with Parkinson’s have many issues with their visuospatial abilities,” as well as their depth and distance perception, and so “enhancing that aspect could potentially be very beneficial.”
“So I’m hopeful that it’s a really good avenue to explore, and the preliminary data are very exciting.”
Dr. Gilbert also highlighted that the “wonderful” aspect of art therapy is that there are “so many benefits and not really any side effects.” Patients can “take the meds … and then enhance that with various therapies, and this would be an additional option.”
Another notable aspect of art therapy is the “social element” and the sense of “camaraderie,” although that has “to be teased out from the benefits you would get from the actual art therapy.”
Finally, Dr. Gilbert pointed out that the difference between the current trial and Dr. Cucca’s trial is the presence of a control group.
“Of course, it’s not blinded, because you know whether you got therapy or not … but that extra element of being able to compare with a group that didn’t get the treatment gives it a little more weight in terms of the field.”
No funding was declared. The authors have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Adding art therapy to standard drug treatment in Parkinson’s disease (PD) not only improves severity of both motor and nonmotor symptoms, but also slows rates of disease progression, new research suggests.
Fifty PD patients were randomly assigned to receive either art therapy, including sculpting and drawing, plus drug therapy or drug therapy alone, and followed up over 12 months.
Patients receiving combined therapy experienced improvements in symptoms, depression, and cognitive scores, and had reduced tremor and daytime sleepiness. They were also substantially less likely to experience disease progression.
“The use of art therapy can reduce the severity of motor and nonmotor manifestations of Parkinson’s disease,” said study investigator Iryna Khubetova, MD, PhD, head of the neurology department, Odessa (Ukraine) Regional Clinical Hospital.
she added.
The findings were presented at the virtual congress of the European College of Neuropsychopharmacology.
A promising approach
Dr. Khubetova told this news organization that offering art therapy to PD patients was “very affordable,” especially as professional artists “provided materials for painting and other art supplies free of charge.”
“We hope this approach is very promising and would be widely adopted.”
She suggested the positive effect of art therapy could be related to “activating the brain’s reward neural network.”
This may be via improved visual attention acting on visuospatial mechanisms and emotional drive, with “activation of the medial orbitofrontal cortex, ventral striatum, and other structures.”
The researchers note PD, a “multisystem progressive neurodegenerative disease,” is among the three most common neurological disorders, with an incidence of 100-150 cases per 100,000 people.
They also note that nonpharmacologic approaches are “widely used” as an adjunct to drug therapy and as part of an “integrated approach” to disease management.
To examine the clinical efficacy of art therapy, the team recruited patients with PD who had preserved facility for independent movement, defined as stages 1-2.5 on the Hoehn and Yahr scale.
Patients were randomly assigned to art therapy sessions alongside standard drug therapy or to standard drug therapy alone. The art therapy included sculpting, free drawing, and coloring patterns.
Multiple benefits
Participants were assessed at baseline and at 6 and 12 months with the Unified Parkinson Disease Rating Scale (UPDRS), the Beck Depression Inventory, the Montreal Cognitive Assessment, and the Pegboard Test of finger dexterity.
Fifty patients were included in the study, with 30 assigned to standard drug therapy alone and 20 to the combined intervention. Participants had a mean age of 57.8 years, and 46% were women.
Over the study period, investigators found patients assigned to art therapy plus drug treatment had improved mood, as well as decreased daytime sleeping, reduced tremor, and a decrease in anxiety and fear intensity.
Between baseline and the 6- and 12-month assessments, patients in the combined therapy group showed improvements in scores on all of the questionnaires, and on the Pegboard Test. In contrast, scores were either stable or worsened in the standard drug therapy–alone group.
The team notes that there was also a marked difference in rates of disease progression, defined as a change on the Hoehn and Yahr scale of at least 0.5 points, between the two groups.
Only two (10%) patients in the combined drug and art therapy progressed over the study period, compared with 10 (33%) in the control group (P = .05).
The findings complement those of a recent study conducted by Alberto Cucca, MD, of the Fresco Institute for Parkinson’s and Movement Disorders, New York University, and colleagues.
Eighteen patients took part in the prospective, open-label trial. They were assessed before and after 20 sessions of art therapy on a range of measures.
Results revealed that following the art therapy, patients had improvements in the Navon Test (which assesses visual neglect, eye tracking, and UPDRS scores), as well as significantly increased functional connectivity levels in the visual cortex on resting-state functional MRI.
Many benefits, no side effects
Rebecca Gilbert, MD, PhD, vice president and chief scientific officer of the American Parkinson Disease Association, who was not involved in either study, told this news organization that the idea of art therapy for patients with Parkinson’s is “very reasonable.”
She highlighted that “people with Parkinson’s have many issues with their visuospatial abilities,” as well as their depth and distance perception, and so “enhancing that aspect could potentially be very beneficial.”
“So I’m hopeful that it’s a really good avenue to explore, and the preliminary data are very exciting.”
Dr. Gilbert also highlighted that the “wonderful” aspect of art therapy is that there are “so many benefits and not really any side effects.” Patients can “take the meds … and then enhance that with various therapies, and this would be an additional option.”
Another notable aspect of art therapy is the “social element” and the sense of “camaraderie,” although that has “to be teased out from the benefits you would get from the actual art therapy.”
Finally, Dr. Gilbert pointed out that the difference between the current trial and Dr. Cucca’s trial is the presence of a control group.
“Of course, it’s not blinded, because you know whether you got therapy or not … but that extra element of being able to compare with a group that didn’t get the treatment gives it a little more weight in terms of the field.”
No funding was declared. The authors have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FROM ECNP 2021
Substance use or substance use disorder: A question of judgment
Substance use disorders can be a thorny topic in residency because of our role as gatekeepers of mental hospitals during our training. Intoxicated patients often get dismissed as a burden and distraction, malingering their way into a comfortable place to regain sobriety. This is extremely prevalent, often constituting the majority of patients seen during an emergency department call.
A typical interview may elicit any or all symptoms in the DSM yet be best explained by substance use intoxication or withdrawal. Alcohol and other CNS depressants commonly cause feelings of sadness and/or suicidality. Methamphetamine and other CNS stimulants commonly cause symptoms of psychosis or mania, followed by feelings of sadness and/or suicidality.
Different EDs have different degrees of patience for individuals in the process of becoming sober. Some departments will pressure clinicians into quickly discarding those patients and often frown upon any attempt at providing solace by raising the concern of reinforcing maladaptive behavior. A mystery-meat sandwich of admirable blandness may be the extent of help offered. Some more fortunate patients also receive a juice box or even a taxi voucher in an especially generous ED. This is always against our better judgment, of course, as we are told those gestures encourage abuse.
Other EDs will permit patients to remain until sober, allowing for another evaluation without the influence of controlled substances. We are reminded of many conversations with patients with substance use disorders, where topics discussed included: 1. Recommendation to seek substance use services, which are often nonexistent or with wait lists spanning months; 2. Education on the role of mental health hospitals and how patients’ despair in the context of intoxication does not meet some scriptural criteria; 3. Pep talks aided by such previously described sandwiches and juice boxes to encourage a sobering patient to leave the facility of their own will.
Methamphetamine, heroin, and alcohol are rarely one-and-done endeavors. We sparingly see our patients for their very first ED visit while intoxicated or crashing. They know how the system runs and which ED will more readily allow them an overnight stay. The number of times they have been recommended for substance use treatment is beyond counting – they may have been on a wait list a handful of times. They are aware of our reluctance to provide inpatient psychiatric treatment for substance use, but it is worth a shot trying, anyway – sometimes they get lucky. Usually it is the pep talk, relief from hunger pangs, and daylight that get them out the doors – until next time.
It is under this context that many trainees become psychiatrists, a process that solidifies the separation between drug use and mental illness. Many graduate from residency practically equating substance use disorder with malingering or futility. This can take on a surreal quality as many localities have recently adopted particular forms or requirements like the dispensation of naloxone syringes to all patients with substance use disorders. While the desire and effort are noble, it may suggest to a patient presenting for help that society’s main interest is to avoid seeing them die rather than help with available resources for maintaining sobriety.
Therein lies the conundrum, a conundrum that spans psychiatry to society. The conundrum is our ambivalence between punishing the choice of drug use or healing the substance use disorder. Should we discharge the intoxicated patient as soon as they are safe to walk out, or should we make every effort possible to find long-term solutions?
The calculation becomes more complex
A defining moment appears to have been society’s reconsideration of its stance on substance use disorders when affluent White teenagers started dying in the suburbs from pain pills overdoses. Suddenly, those children needed and deserved treatment, not punishment. We find ourselves far away from a time when the loudest societal commentary on substance use entailed mothers advocating for harsher sentences against drunk drivers.
More recently, as psychiatry and large contingents of society have decided to take up the mantle of equity and social justice, we have begun to make progress in decriminalizing substance use in an effort to reverse systemic discrimination toward minority groups. This has taken many shapes, including drug legalization, criminal justice reform, and even the provision of clean substance use paraphernalia for safer use of IV drugs. Police reform has led to reluctance to arrest or press charges for nonviolent crimes and reduced police presence in minority neighborhoods. The “rich White teenager” approach is now recommended in all neighborhoods.
Society’s attempt at decriminalizing drug use has run parallel with psychiatry’s recent attempts at reduced pathologizing of behaviors more prevalent in underprivileged groups and cultures. This runs the gamut, from avoiding the use of the term “agitated” because of its racial connotations, to advocating for reduced rates of schizophrenia diagnoses in Black males.1 A diagnosis of substance use disorder carries with it similar troublesome societal implications. Decriminalization, legalization, provision of substances to the population, normalization, and other societal reforms will likely have an impact on the prevalence of substance use disorder diagnoses, which involve many criteria dependent on societal context.
It would be expected that criteria such as hazardous use, social problems, and attempts to quit will decrease as social acceptance increases. How might this affect access to substance use treatment, an already extremely limited resource?
Now, as forensic psychiatrists, we find ourselves adjudicating on the role of drugs at a time when society is wrestling with its attitude on the breadth of responsibility possessed by people who use drugs. In California, as in many other states, insanity laws exclude those who were insane as a result of drug use, as a testament to or possibly a remnant of how society feels about the role of choice and responsibility in the use of drugs. Yet another defendant who admits to drug use may on the contrary receive a much more lenient plea deal if willing to commit to sobriety. But in a never-ending maze of differing judgments and opinions, a less understanding district attorney may argue that the additional risk posed by the use of drugs and resulting impulsivity may actually warrant a heavier sentence.
In a recent attempt at atonement for our past punitive stance on drug users, we have found a desire to protect those who use drugs by punishing those who sell, at times forgetting that these populations are deeply intertwined. A recent law permits the federal charge of distribution of fentanyl resulting in death, which carries the mandatory minimum of 20 years in prison. Yet, if the user whom we are trying to protect by this law is also the one selling, what are we left with?
Fentanyl has been a particularly tragic development in the history of mankind and drug use. Substance use has rarely been so easily linked to accidental death. While many physicians can easily explain the safety of fentanyl when used as prescribed and in controlled settings, this is certainly not the case in the community. Measuring micrograms of fentanyl is outside the knowledge and capabilities of most drug dealers, who are not equipped with pharmacy-grade scales. Yet, as a result, they sell and customers buy quantities of fentanyl that range from homeopathically low to lethally high because of a mixture of negligence and deliberate indifference.
Another effort at atonement has been attempts at decriminalizing drug use and releasing many nonviolent offenders. This can, however, encourage bystanders to report more acts as crime rather than public intoxication, to ensure a police response when confronted by intoxicated people. Whereas previously an inebriated person who is homeless may have been called for and asked to seek shelter, they now get called on, and subsequently charged for, allegedly mumbling a threat by a frustrated bystander.
The release of offenders has its limits. Many placements on probation require sobriety and result in longer sentences for the use of substances that are otherwise decriminalized. The decriminalization and reexamination of substance use by society should widen the scope from simply considering crime to examining the use of drugs throughout the legal system and even beyond.
The DSM and psychiatry are not intended or equipped to adjudicate disputes on where the lines should be drawn between determinism and free will. We are knowledgeable of patients with substance use disorders, the effect of intoxicating substances, and the capacity of patients with substance use disorders to act in law-abiding ways. Our field can inform without simply advocating whether our patients should be punished. While society is currently struggling with how to apportion blame, psychiatry should resist the urge to impose medical solutions to social problems. Our solutions would almost certainly be grossly limited as we are still struggling to repent for lobotomizing “uppity” young women2 and using electroshock therapy to disrupt perverse impulses in homosexual males.3 Social norms and political zeitgeists change over time while the psychological and physiological principles underlying our understanding of mental illness should, in theory, stay relatively constant. Psychiatry’s answers for societal ills do not usually improve with time but rather have a tendency to be humbling.
Dr. Badre is a clinical and forensic psychiatrist in San Diego. He holds teaching positions at the University of California, San Diego, and the University of San Diego. He teaches medical education, psychopharmacology, ethics in psychiatry, and correctional care. Dr. Badre can be reached at his website, BadreMD.com.
Dr. Compton is a psychiatry resident at University of California, San Diego. His background includes medical education, mental health advocacy, work with underserved populations, and brain cancer research.
References
1.Medlock MM et al., eds. “Racism and Psychiatry: Contemporary Issues and Interventions” (New York: Springer, 2018).
2. Tone A and Koziol M. CMAJ. 2018:190(20):e624-5.
3. McGuire RJ and Vallance M. BMJ. 1964;1(5376):151-3.
Substance use disorders can be a thorny topic in residency because of our role as gatekeepers of mental hospitals during our training. Intoxicated patients often get dismissed as a burden and distraction, malingering their way into a comfortable place to regain sobriety. This is extremely prevalent, often constituting the majority of patients seen during an emergency department call.
A typical interview may elicit any or all symptoms in the DSM yet be best explained by substance use intoxication or withdrawal. Alcohol and other CNS depressants commonly cause feelings of sadness and/or suicidality. Methamphetamine and other CNS stimulants commonly cause symptoms of psychosis or mania, followed by feelings of sadness and/or suicidality.
Different EDs have different degrees of patience for individuals in the process of becoming sober. Some departments will pressure clinicians into quickly discarding those patients and often frown upon any attempt at providing solace by raising the concern of reinforcing maladaptive behavior. A mystery-meat sandwich of admirable blandness may be the extent of help offered. Some more fortunate patients also receive a juice box or even a taxi voucher in an especially generous ED. This is always against our better judgment, of course, as we are told those gestures encourage abuse.
Other EDs will permit patients to remain until sober, allowing for another evaluation without the influence of controlled substances. We are reminded of many conversations with patients with substance use disorders, where topics discussed included: 1. Recommendation to seek substance use services, which are often nonexistent or with wait lists spanning months; 2. Education on the role of mental health hospitals and how patients’ despair in the context of intoxication does not meet some scriptural criteria; 3. Pep talks aided by such previously described sandwiches and juice boxes to encourage a sobering patient to leave the facility of their own will.
Methamphetamine, heroin, and alcohol are rarely one-and-done endeavors. We sparingly see our patients for their very first ED visit while intoxicated or crashing. They know how the system runs and which ED will more readily allow them an overnight stay. The number of times they have been recommended for substance use treatment is beyond counting – they may have been on a wait list a handful of times. They are aware of our reluctance to provide inpatient psychiatric treatment for substance use, but it is worth a shot trying, anyway – sometimes they get lucky. Usually it is the pep talk, relief from hunger pangs, and daylight that get them out the doors – until next time.
It is under this context that many trainees become psychiatrists, a process that solidifies the separation between drug use and mental illness. Many graduate from residency practically equating substance use disorder with malingering or futility. This can take on a surreal quality as many localities have recently adopted particular forms or requirements like the dispensation of naloxone syringes to all patients with substance use disorders. While the desire and effort are noble, it may suggest to a patient presenting for help that society’s main interest is to avoid seeing them die rather than help with available resources for maintaining sobriety.
Therein lies the conundrum, a conundrum that spans psychiatry to society. The conundrum is our ambivalence between punishing the choice of drug use or healing the substance use disorder. Should we discharge the intoxicated patient as soon as they are safe to walk out, or should we make every effort possible to find long-term solutions?
The calculation becomes more complex
A defining moment appears to have been society’s reconsideration of its stance on substance use disorders when affluent White teenagers started dying in the suburbs from pain pills overdoses. Suddenly, those children needed and deserved treatment, not punishment. We find ourselves far away from a time when the loudest societal commentary on substance use entailed mothers advocating for harsher sentences against drunk drivers.
More recently, as psychiatry and large contingents of society have decided to take up the mantle of equity and social justice, we have begun to make progress in decriminalizing substance use in an effort to reverse systemic discrimination toward minority groups. This has taken many shapes, including drug legalization, criminal justice reform, and even the provision of clean substance use paraphernalia for safer use of IV drugs. Police reform has led to reluctance to arrest or press charges for nonviolent crimes and reduced police presence in minority neighborhoods. The “rich White teenager” approach is now recommended in all neighborhoods.
Society’s attempt at decriminalizing drug use has run parallel with psychiatry’s recent attempts at reduced pathologizing of behaviors more prevalent in underprivileged groups and cultures. This runs the gamut, from avoiding the use of the term “agitated” because of its racial connotations, to advocating for reduced rates of schizophrenia diagnoses in Black males.1 A diagnosis of substance use disorder carries with it similar troublesome societal implications. Decriminalization, legalization, provision of substances to the population, normalization, and other societal reforms will likely have an impact on the prevalence of substance use disorder diagnoses, which involve many criteria dependent on societal context.
It would be expected that criteria such as hazardous use, social problems, and attempts to quit will decrease as social acceptance increases. How might this affect access to substance use treatment, an already extremely limited resource?
Now, as forensic psychiatrists, we find ourselves adjudicating on the role of drugs at a time when society is wrestling with its attitude on the breadth of responsibility possessed by people who use drugs. In California, as in many other states, insanity laws exclude those who were insane as a result of drug use, as a testament to or possibly a remnant of how society feels about the role of choice and responsibility in the use of drugs. Yet another defendant who admits to drug use may on the contrary receive a much more lenient plea deal if willing to commit to sobriety. But in a never-ending maze of differing judgments and opinions, a less understanding district attorney may argue that the additional risk posed by the use of drugs and resulting impulsivity may actually warrant a heavier sentence.
In a recent attempt at atonement for our past punitive stance on drug users, we have found a desire to protect those who use drugs by punishing those who sell, at times forgetting that these populations are deeply intertwined. A recent law permits the federal charge of distribution of fentanyl resulting in death, which carries the mandatory minimum of 20 years in prison. Yet, if the user whom we are trying to protect by this law is also the one selling, what are we left with?
Fentanyl has been a particularly tragic development in the history of mankind and drug use. Substance use has rarely been so easily linked to accidental death. While many physicians can easily explain the safety of fentanyl when used as prescribed and in controlled settings, this is certainly not the case in the community. Measuring micrograms of fentanyl is outside the knowledge and capabilities of most drug dealers, who are not equipped with pharmacy-grade scales. Yet, as a result, they sell and customers buy quantities of fentanyl that range from homeopathically low to lethally high because of a mixture of negligence and deliberate indifference.
Another effort at atonement has been attempts at decriminalizing drug use and releasing many nonviolent offenders. This can, however, encourage bystanders to report more acts as crime rather than public intoxication, to ensure a police response when confronted by intoxicated people. Whereas previously an inebriated person who is homeless may have been called for and asked to seek shelter, they now get called on, and subsequently charged for, allegedly mumbling a threat by a frustrated bystander.
The release of offenders has its limits. Many placements on probation require sobriety and result in longer sentences for the use of substances that are otherwise decriminalized. The decriminalization and reexamination of substance use by society should widen the scope from simply considering crime to examining the use of drugs throughout the legal system and even beyond.
The DSM and psychiatry are not intended or equipped to adjudicate disputes on where the lines should be drawn between determinism and free will. We are knowledgeable of patients with substance use disorders, the effect of intoxicating substances, and the capacity of patients with substance use disorders to act in law-abiding ways. Our field can inform without simply advocating whether our patients should be punished. While society is currently struggling with how to apportion blame, psychiatry should resist the urge to impose medical solutions to social problems. Our solutions would almost certainly be grossly limited as we are still struggling to repent for lobotomizing “uppity” young women2 and using electroshock therapy to disrupt perverse impulses in homosexual males.3 Social norms and political zeitgeists change over time while the psychological and physiological principles underlying our understanding of mental illness should, in theory, stay relatively constant. Psychiatry’s answers for societal ills do not usually improve with time but rather have a tendency to be humbling.
Dr. Badre is a clinical and forensic psychiatrist in San Diego. He holds teaching positions at the University of California, San Diego, and the University of San Diego. He teaches medical education, psychopharmacology, ethics in psychiatry, and correctional care. Dr. Badre can be reached at his website, BadreMD.com.
Dr. Compton is a psychiatry resident at University of California, San Diego. His background includes medical education, mental health advocacy, work with underserved populations, and brain cancer research.
References
1.Medlock MM et al., eds. “Racism and Psychiatry: Contemporary Issues and Interventions” (New York: Springer, 2018).
2. Tone A and Koziol M. CMAJ. 2018:190(20):e624-5.
3. McGuire RJ and Vallance M. BMJ. 1964;1(5376):151-3.
Substance use disorders can be a thorny topic in residency because of our role as gatekeepers of mental hospitals during our training. Intoxicated patients often get dismissed as a burden and distraction, malingering their way into a comfortable place to regain sobriety. This is extremely prevalent, often constituting the majority of patients seen during an emergency department call.
A typical interview may elicit any or all symptoms in the DSM yet be best explained by substance use intoxication or withdrawal. Alcohol and other CNS depressants commonly cause feelings of sadness and/or suicidality. Methamphetamine and other CNS stimulants commonly cause symptoms of psychosis or mania, followed by feelings of sadness and/or suicidality.
Different EDs have different degrees of patience for individuals in the process of becoming sober. Some departments will pressure clinicians into quickly discarding those patients and often frown upon any attempt at providing solace by raising the concern of reinforcing maladaptive behavior. A mystery-meat sandwich of admirable blandness may be the extent of help offered. Some more fortunate patients also receive a juice box or even a taxi voucher in an especially generous ED. This is always against our better judgment, of course, as we are told those gestures encourage abuse.
Other EDs will permit patients to remain until sober, allowing for another evaluation without the influence of controlled substances. We are reminded of many conversations with patients with substance use disorders, where topics discussed included: 1. Recommendation to seek substance use services, which are often nonexistent or with wait lists spanning months; 2. Education on the role of mental health hospitals and how patients’ despair in the context of intoxication does not meet some scriptural criteria; 3. Pep talks aided by such previously described sandwiches and juice boxes to encourage a sobering patient to leave the facility of their own will.
Methamphetamine, heroin, and alcohol are rarely one-and-done endeavors. We sparingly see our patients for their very first ED visit while intoxicated or crashing. They know how the system runs and which ED will more readily allow them an overnight stay. The number of times they have been recommended for substance use treatment is beyond counting – they may have been on a wait list a handful of times. They are aware of our reluctance to provide inpatient psychiatric treatment for substance use, but it is worth a shot trying, anyway – sometimes they get lucky. Usually it is the pep talk, relief from hunger pangs, and daylight that get them out the doors – until next time.
It is under this context that many trainees become psychiatrists, a process that solidifies the separation between drug use and mental illness. Many graduate from residency practically equating substance use disorder with malingering or futility. This can take on a surreal quality as many localities have recently adopted particular forms or requirements like the dispensation of naloxone syringes to all patients with substance use disorders. While the desire and effort are noble, it may suggest to a patient presenting for help that society’s main interest is to avoid seeing them die rather than help with available resources for maintaining sobriety.
Therein lies the conundrum, a conundrum that spans psychiatry to society. The conundrum is our ambivalence between punishing the choice of drug use or healing the substance use disorder. Should we discharge the intoxicated patient as soon as they are safe to walk out, or should we make every effort possible to find long-term solutions?
The calculation becomes more complex
A defining moment appears to have been society’s reconsideration of its stance on substance use disorders when affluent White teenagers started dying in the suburbs from pain pills overdoses. Suddenly, those children needed and deserved treatment, not punishment. We find ourselves far away from a time when the loudest societal commentary on substance use entailed mothers advocating for harsher sentences against drunk drivers.
More recently, as psychiatry and large contingents of society have decided to take up the mantle of equity and social justice, we have begun to make progress in decriminalizing substance use in an effort to reverse systemic discrimination toward minority groups. This has taken many shapes, including drug legalization, criminal justice reform, and even the provision of clean substance use paraphernalia for safer use of IV drugs. Police reform has led to reluctance to arrest or press charges for nonviolent crimes and reduced police presence in minority neighborhoods. The “rich White teenager” approach is now recommended in all neighborhoods.
Society’s attempt at decriminalizing drug use has run parallel with psychiatry’s recent attempts at reduced pathologizing of behaviors more prevalent in underprivileged groups and cultures. This runs the gamut, from avoiding the use of the term “agitated” because of its racial connotations, to advocating for reduced rates of schizophrenia diagnoses in Black males.1 A diagnosis of substance use disorder carries with it similar troublesome societal implications. Decriminalization, legalization, provision of substances to the population, normalization, and other societal reforms will likely have an impact on the prevalence of substance use disorder diagnoses, which involve many criteria dependent on societal context.
It would be expected that criteria such as hazardous use, social problems, and attempts to quit will decrease as social acceptance increases. How might this affect access to substance use treatment, an already extremely limited resource?
Now, as forensic psychiatrists, we find ourselves adjudicating on the role of drugs at a time when society is wrestling with its attitude on the breadth of responsibility possessed by people who use drugs. In California, as in many other states, insanity laws exclude those who were insane as a result of drug use, as a testament to or possibly a remnant of how society feels about the role of choice and responsibility in the use of drugs. Yet another defendant who admits to drug use may on the contrary receive a much more lenient plea deal if willing to commit to sobriety. But in a never-ending maze of differing judgments and opinions, a less understanding district attorney may argue that the additional risk posed by the use of drugs and resulting impulsivity may actually warrant a heavier sentence.
In a recent attempt at atonement for our past punitive stance on drug users, we have found a desire to protect those who use drugs by punishing those who sell, at times forgetting that these populations are deeply intertwined. A recent law permits the federal charge of distribution of fentanyl resulting in death, which carries the mandatory minimum of 20 years in prison. Yet, if the user whom we are trying to protect by this law is also the one selling, what are we left with?
Fentanyl has been a particularly tragic development in the history of mankind and drug use. Substance use has rarely been so easily linked to accidental death. While many physicians can easily explain the safety of fentanyl when used as prescribed and in controlled settings, this is certainly not the case in the community. Measuring micrograms of fentanyl is outside the knowledge and capabilities of most drug dealers, who are not equipped with pharmacy-grade scales. Yet, as a result, they sell and customers buy quantities of fentanyl that range from homeopathically low to lethally high because of a mixture of negligence and deliberate indifference.
Another effort at atonement has been attempts at decriminalizing drug use and releasing many nonviolent offenders. This can, however, encourage bystanders to report more acts as crime rather than public intoxication, to ensure a police response when confronted by intoxicated people. Whereas previously an inebriated person who is homeless may have been called for and asked to seek shelter, they now get called on, and subsequently charged for, allegedly mumbling a threat by a frustrated bystander.
The release of offenders has its limits. Many placements on probation require sobriety and result in longer sentences for the use of substances that are otherwise decriminalized. The decriminalization and reexamination of substance use by society should widen the scope from simply considering crime to examining the use of drugs throughout the legal system and even beyond.
The DSM and psychiatry are not intended or equipped to adjudicate disputes on where the lines should be drawn between determinism and free will. We are knowledgeable of patients with substance use disorders, the effect of intoxicating substances, and the capacity of patients with substance use disorders to act in law-abiding ways. Our field can inform without simply advocating whether our patients should be punished. While society is currently struggling with how to apportion blame, psychiatry should resist the urge to impose medical solutions to social problems. Our solutions would almost certainly be grossly limited as we are still struggling to repent for lobotomizing “uppity” young women2 and using electroshock therapy to disrupt perverse impulses in homosexual males.3 Social norms and political zeitgeists change over time while the psychological and physiological principles underlying our understanding of mental illness should, in theory, stay relatively constant. Psychiatry’s answers for societal ills do not usually improve with time but rather have a tendency to be humbling.
Dr. Badre is a clinical and forensic psychiatrist in San Diego. He holds teaching positions at the University of California, San Diego, and the University of San Diego. He teaches medical education, psychopharmacology, ethics in psychiatry, and correctional care. Dr. Badre can be reached at his website, BadreMD.com.
Dr. Compton is a psychiatry resident at University of California, San Diego. His background includes medical education, mental health advocacy, work with underserved populations, and brain cancer research.
References
1.Medlock MM et al., eds. “Racism and Psychiatry: Contemporary Issues and Interventions” (New York: Springer, 2018).
2. Tone A and Koziol M. CMAJ. 2018:190(20):e624-5.
3. McGuire RJ and Vallance M. BMJ. 1964;1(5376):151-3.