User login
Automated office BP measurement: The new standard in HTN screening
ILLUSTRATIVE CASE
A 45-year-old woman with no chronic medical illness presents to your office for her annual physical examination. After a medical assistant (MA) applies an automatic BP cuff to the patient’s left arm, the BP reading is 155/92 mm Hg. The MA then rechecks the BP, and this time it reads 160/98 mm Hg. The MA performs a manual BP reading, which is 158/90 mm Hg (left arm) and 162/100 mm Hg (right arm). The patient denies any headache, visual changes, chest pain, or difficulty breathing and tells the MA that her BP is always high during a doctor visit. You are wondering if she has hypertension or if is this the white-coat effect.
Depending on the definition of hypertension, its prevalence among US adults 18 years or older varies from 46%, based on the American College of Cardiology guideline (≥ 130/80 mm Hg), to 29%, based on the Eighth Joint National Committee (JNC-8) guideline (≥ 140/90 mm Hg for adults ages 18–59 years and ≥ 150/90 mm Hg for adults ≥ 60 years without diabetes and/or chronic kidney disease).2,3
According to JNC-8, the prevalence is similar among men (30.2%) and women (27.7%) and increases with age: 18 to 39 years, 7.5%; 40 to 59 years, 33.2%; and ≥ 60 years, 63.1%.3,4 When ranked by risk-attributable disability-adjusted life-years (DALYs), high systolic blood pressure (SBP) is the leading risk factor, accounting for 10.4 million deaths and 218 million DALYs globally in 2017.5 National medical costs associated with hypertension are estimated to account for about $131 billion in annual health care expenditures, averaged over 12 years from 2003 to 2014.6
When performed correctly, the auscultatory method using a mercury sphygmomanometer correlates well with simultaneous intra-arterial BP and was considered the gold standard for office-based measurements for many years.7,8 However, significant observer-related differences in auditory acuity and terminal digit rounding are sources of inaccurate measurement. White-coat hypertension cannot be detected with this method—another significant limitation. The inaccuracy of office-based BP readings leads to concerns about hypertension being inappropriately diagnosed in patients or delays in diagnosis occurring.9
A proposed solution to this problem is measurement using an oscillometric sphygmomanometer. This device uses a pressure transducer to assess the oscillations of pressure in a cuff during gradual deflation; it provides accurate BP measurements when fully automated and programmed to complete several BP measurements at appropriate intervals while the patient rests alone in a quiet room.10
The accuracy of this new method was tested in a 2009 cohort study of 309 patients referred to an ambulatory blood pressure (ABP) monitoring unit at an academic hospital for diagnosis or management of hypertension.11 The study compared mean awake
A 2019 meta-analysis that included 26 studies (N = 7116) comparing
Continue to: STUDY SUMMARY
STUDY SUMMARY
Automated office BP devices are just as accurate as more expensive ABP studies
This systematic review and meta-analysis (
The study also explored the protocol by which the best AOBP results could be obtained. For AOBP measurement, the included trials had no more than 2 minutes of elapsed time between individual AOBP measurements and had at least 3 AOBP readings to calculate the mean.
Compared with AOBP, in samples with an SBP of ≥ 130 mm Hg, SBP readings were significantly higher for both routine office visits (
Although there was statistical heterogeneity, the results were confirmed in the authors’ analysis of studies with high methodologic quality. In addition, researchers performed multiple
WHAT'S NEW
Study confirms unattended, automated office BP as preferred technique
This is the second recent comprehensive systematic review and meta-analysis to directly compare AOBP with other common techniques of BP measurement in screening for and diagnosing hypertension in the clinical setting. 9
Continue to: This meta-analysis...
This meta-analysis emphasized the technique (see below) by which to obtain the best AOBP vs ABP results, whereas the other meta-analysis9 did not. Thus the study provides practice-based settings with the information they need to more closely replicate the results of the studies included in the meta-analysis.
Also, the equivalency comparison with the more expensive and intrusive ABP monitoring may save money, improve patient adherence, and increase patient satisfaction. Given these advantages, along with its demonstrated accuracy, AOBP should be adopted in routine clinical practice to screen patients for hypertension.
CAVEATS
Close adherence to measurementprocedures is a necessity
Effective use of AOBP in clinical practice requires close adherence to the AOBP study procedures described in this meta-analysis. These include taking multiple (at least 3) BP readings, 1 to 2 minutes apart, recorded with a fully automated oscillometric sphygmomanometer while the patient rests alone in a quiet place.
CHALLENGES TO IMPLEMENTATION
Adjusting workflows, addressing cost
Physicians may be reluctant to adopt this technique because they may not be convinced of its advantages compared with the traditional methods of recording BP and because of difficulties with implementing new rooming workflows.12 The cost of AOBP devices used in this study (Omron 907 and BpTRU; BpTRU ceased operations in 2017) were not disclosed, which may be a hindrance, as devices may cost $1000 or more.
An online search for “automated oscillometric BP monitor” by one of the PURL authors (RCM) found oscillometric AOBP devices ranging from $150 to > $1000, depending on whether the device was medical grade; a search for “Omron 907” found devices for ≤ $599 on multiple sites. However, none of the lower-cost devices indicated the ability to take multiple, unattended BP readings.
ACKNOWLEDGEMENT
The PURLs Surveillance System was supported in part by Grant Number UL1RR024999 from the National Center for Research Resources, a Clinical Translational Science Award to the University of Chicago. The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Center for Research Resources or the National Institutes of Health.
1. Roerecke M, Kaczorowski J, Myers MG. Comparing automated office blood pressure readings with other methods of blood pressure measurement for identifying patients with possible hypertension: a systematic review and meta-analysis. JAMA Intern Med. 2019;179:351-362.
2. Whelton PK, Carey RM, Aronow WS, et al. 2017 ACC/AHA/AAPA/ABC/ACPM/AGS/APhA/ASH/ASPC/NMA/PCNA guideline for the prevention, detection, evaluation, and management of high blood pressure in adults: a report of the American College of Cardiology/American Heart Association Task Force on Clinical Practice Guidelines. Hypertension. 2018;71:e13-e115. Published correction appears in Hypertension. 2018;71:e140-e144.
3. James PA, Oparil S, Carter BL, et al. 2014 evidence-based guideline for the management of high blood pressure in adults: report from the panel members appointed to the Eighth Joint National Committee (JNC 8). JAMA. 2014;311:507-520. Published correction appears in JAMA. 2014;311:1809.
4. Fryar CD, Ostchega Y, Hales CM, et al. Hypertension prevalence and control among adults: United States, 2015-2016. NCHS Data Brief. 2017;(289):1-8.
5. GBD 2017 Risk Factor Collaborators. Global, regional, and national comparative risk assessment of 84 behavioural, environmental and occupational, and metabolic risks or clusters of risks for 195 countries and territories, 1990-2017: a systematic analysis for the Global Burden of Disease Study 2017. Lancet. 2018;392:1923-1994.
6. Kirkland EB, Heincelman M, Bishu KG, et al. Trends in healthcare expenditures among US adults with hypertension: national estimates, 2003-2014. J Am Heart Assoc. 2018;7:e008731.
7. Pickering TG, Hall JE, Appel LJ, et al. Recommendations for blood pressure measurement in humans and experimental animals: part 1: blood pressure measurement in humans: a statement for professionals from the Subcommittee of Professional and Public Education of the American Heart Association Council on High Blood Pressure Research. Circulation. 2005;111:697-716.
8. Ogedegbe G, Pickering T. Principles and techniques of blood pressure measurement. Cardiol Clin. 2010;28:571-586.
9. Pappaccogli M, Di Monaco S, Perlo E, et al. Comparison of automated office blood pressure with office and out-of-office measurement techniques. Hypertension. 2019;73:481-490.
10. Reeves RA. The rational clinical examination. Does this patient have hypertension? How to measure blood pressure. JAMA. 1995;273:1211-1218.
11. Myers MG, Valdivieso M, Kiss A. Use of automated office blood pressure measurement to reduce the white coat response. J Hypertens. 2009;27:280-286.
12. Centers for Medicare & Medicaid Services. Decision memo for ambulatory blood pressure monitoring (ABPM) (CAG-00067R2). July 2, 2019. Accessed September 29, 2020. www.cms.gov/medicare-coverage-database/details/nca-decision-memo.aspx?NCAId=294
ILLUSTRATIVE CASE
A 45-year-old woman with no chronic medical illness presents to your office for her annual physical examination. After a medical assistant (MA) applies an automatic BP cuff to the patient’s left arm, the BP reading is 155/92 mm Hg. The MA then rechecks the BP, and this time it reads 160/98 mm Hg. The MA performs a manual BP reading, which is 158/90 mm Hg (left arm) and 162/100 mm Hg (right arm). The patient denies any headache, visual changes, chest pain, or difficulty breathing and tells the MA that her BP is always high during a doctor visit. You are wondering if she has hypertension or if is this the white-coat effect.
Depending on the definition of hypertension, its prevalence among US adults 18 years or older varies from 46%, based on the American College of Cardiology guideline (≥ 130/80 mm Hg), to 29%, based on the Eighth Joint National Committee (JNC-8) guideline (≥ 140/90 mm Hg for adults ages 18–59 years and ≥ 150/90 mm Hg for adults ≥ 60 years without diabetes and/or chronic kidney disease).2,3
According to JNC-8, the prevalence is similar among men (30.2%) and women (27.7%) and increases with age: 18 to 39 years, 7.5%; 40 to 59 years, 33.2%; and ≥ 60 years, 63.1%.3,4 When ranked by risk-attributable disability-adjusted life-years (DALYs), high systolic blood pressure (SBP) is the leading risk factor, accounting for 10.4 million deaths and 218 million DALYs globally in 2017.5 National medical costs associated with hypertension are estimated to account for about $131 billion in annual health care expenditures, averaged over 12 years from 2003 to 2014.6
When performed correctly, the auscultatory method using a mercury sphygmomanometer correlates well with simultaneous intra-arterial BP and was considered the gold standard for office-based measurements for many years.7,8 However, significant observer-related differences in auditory acuity and terminal digit rounding are sources of inaccurate measurement. White-coat hypertension cannot be detected with this method—another significant limitation. The inaccuracy of office-based BP readings leads to concerns about hypertension being inappropriately diagnosed in patients or delays in diagnosis occurring.9
A proposed solution to this problem is measurement using an oscillometric sphygmomanometer. This device uses a pressure transducer to assess the oscillations of pressure in a cuff during gradual deflation; it provides accurate BP measurements when fully automated and programmed to complete several BP measurements at appropriate intervals while the patient rests alone in a quiet room.10
The accuracy of this new method was tested in a 2009 cohort study of 309 patients referred to an ambulatory blood pressure (ABP) monitoring unit at an academic hospital for diagnosis or management of hypertension.11 The study compared mean awake
A 2019 meta-analysis that included 26 studies (N = 7116) comparing
Continue to: STUDY SUMMARY
STUDY SUMMARY
Automated office BP devices are just as accurate as more expensive ABP studies
This systematic review and meta-analysis (
The study also explored the protocol by which the best AOBP results could be obtained. For AOBP measurement, the included trials had no more than 2 minutes of elapsed time between individual AOBP measurements and had at least 3 AOBP readings to calculate the mean.
Compared with AOBP, in samples with an SBP of ≥ 130 mm Hg, SBP readings were significantly higher for both routine office visits (
Although there was statistical heterogeneity, the results were confirmed in the authors’ analysis of studies with high methodologic quality. In addition, researchers performed multiple
WHAT'S NEW
Study confirms unattended, automated office BP as preferred technique
This is the second recent comprehensive systematic review and meta-analysis to directly compare AOBP with other common techniques of BP measurement in screening for and diagnosing hypertension in the clinical setting. 9
Continue to: This meta-analysis...
This meta-analysis emphasized the technique (see below) by which to obtain the best AOBP vs ABP results, whereas the other meta-analysis9 did not. Thus the study provides practice-based settings with the information they need to more closely replicate the results of the studies included in the meta-analysis.
Also, the equivalency comparison with the more expensive and intrusive ABP monitoring may save money, improve patient adherence, and increase patient satisfaction. Given these advantages, along with its demonstrated accuracy, AOBP should be adopted in routine clinical practice to screen patients for hypertension.
CAVEATS
Close adherence to measurementprocedures is a necessity
Effective use of AOBP in clinical practice requires close adherence to the AOBP study procedures described in this meta-analysis. These include taking multiple (at least 3) BP readings, 1 to 2 minutes apart, recorded with a fully automated oscillometric sphygmomanometer while the patient rests alone in a quiet place.
CHALLENGES TO IMPLEMENTATION
Adjusting workflows, addressing cost
Physicians may be reluctant to adopt this technique because they may not be convinced of its advantages compared with the traditional methods of recording BP and because of difficulties with implementing new rooming workflows.12 The cost of AOBP devices used in this study (Omron 907 and BpTRU; BpTRU ceased operations in 2017) were not disclosed, which may be a hindrance, as devices may cost $1000 or more.
An online search for “automated oscillometric BP monitor” by one of the PURL authors (RCM) found oscillometric AOBP devices ranging from $150 to > $1000, depending on whether the device was medical grade; a search for “Omron 907” found devices for ≤ $599 on multiple sites. However, none of the lower-cost devices indicated the ability to take multiple, unattended BP readings.
ACKNOWLEDGEMENT
The PURLs Surveillance System was supported in part by Grant Number UL1RR024999 from the National Center for Research Resources, a Clinical Translational Science Award to the University of Chicago. The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Center for Research Resources or the National Institutes of Health.
ILLUSTRATIVE CASE
A 45-year-old woman with no chronic medical illness presents to your office for her annual physical examination. After a medical assistant (MA) applies an automatic BP cuff to the patient’s left arm, the BP reading is 155/92 mm Hg. The MA then rechecks the BP, and this time it reads 160/98 mm Hg. The MA performs a manual BP reading, which is 158/90 mm Hg (left arm) and 162/100 mm Hg (right arm). The patient denies any headache, visual changes, chest pain, or difficulty breathing and tells the MA that her BP is always high during a doctor visit. You are wondering if she has hypertension or if is this the white-coat effect.
Depending on the definition of hypertension, its prevalence among US adults 18 years or older varies from 46%, based on the American College of Cardiology guideline (≥ 130/80 mm Hg), to 29%, based on the Eighth Joint National Committee (JNC-8) guideline (≥ 140/90 mm Hg for adults ages 18–59 years and ≥ 150/90 mm Hg for adults ≥ 60 years without diabetes and/or chronic kidney disease).2,3
According to JNC-8, the prevalence is similar among men (30.2%) and women (27.7%) and increases with age: 18 to 39 years, 7.5%; 40 to 59 years, 33.2%; and ≥ 60 years, 63.1%.3,4 When ranked by risk-attributable disability-adjusted life-years (DALYs), high systolic blood pressure (SBP) is the leading risk factor, accounting for 10.4 million deaths and 218 million DALYs globally in 2017.5 National medical costs associated with hypertension are estimated to account for about $131 billion in annual health care expenditures, averaged over 12 years from 2003 to 2014.6
When performed correctly, the auscultatory method using a mercury sphygmomanometer correlates well with simultaneous intra-arterial BP and was considered the gold standard for office-based measurements for many years.7,8 However, significant observer-related differences in auditory acuity and terminal digit rounding are sources of inaccurate measurement. White-coat hypertension cannot be detected with this method—another significant limitation. The inaccuracy of office-based BP readings leads to concerns about hypertension being inappropriately diagnosed in patients or delays in diagnosis occurring.9
A proposed solution to this problem is measurement using an oscillometric sphygmomanometer. This device uses a pressure transducer to assess the oscillations of pressure in a cuff during gradual deflation; it provides accurate BP measurements when fully automated and programmed to complete several BP measurements at appropriate intervals while the patient rests alone in a quiet room.10
The accuracy of this new method was tested in a 2009 cohort study of 309 patients referred to an ambulatory blood pressure (ABP) monitoring unit at an academic hospital for diagnosis or management of hypertension.11 The study compared mean awake
A 2019 meta-analysis that included 26 studies (N = 7116) comparing
Continue to: STUDY SUMMARY
STUDY SUMMARY
Automated office BP devices are just as accurate as more expensive ABP studies
This systematic review and meta-analysis (
The study also explored the protocol by which the best AOBP results could be obtained. For AOBP measurement, the included trials had no more than 2 minutes of elapsed time between individual AOBP measurements and had at least 3 AOBP readings to calculate the mean.
Compared with AOBP, in samples with an SBP of ≥ 130 mm Hg, SBP readings were significantly higher for both routine office visits (
Although there was statistical heterogeneity, the results were confirmed in the authors’ analysis of studies with high methodologic quality. In addition, researchers performed multiple
WHAT'S NEW
Study confirms unattended, automated office BP as preferred technique
This is the second recent comprehensive systematic review and meta-analysis to directly compare AOBP with other common techniques of BP measurement in screening for and diagnosing hypertension in the clinical setting. 9
Continue to: This meta-analysis...
This meta-analysis emphasized the technique (see below) by which to obtain the best AOBP vs ABP results, whereas the other meta-analysis9 did not. Thus the study provides practice-based settings with the information they need to more closely replicate the results of the studies included in the meta-analysis.
Also, the equivalency comparison with the more expensive and intrusive ABP monitoring may save money, improve patient adherence, and increase patient satisfaction. Given these advantages, along with its demonstrated accuracy, AOBP should be adopted in routine clinical practice to screen patients for hypertension.
CAVEATS
Close adherence to measurementprocedures is a necessity
Effective use of AOBP in clinical practice requires close adherence to the AOBP study procedures described in this meta-analysis. These include taking multiple (at least 3) BP readings, 1 to 2 minutes apart, recorded with a fully automated oscillometric sphygmomanometer while the patient rests alone in a quiet place.
CHALLENGES TO IMPLEMENTATION
Adjusting workflows, addressing cost
Physicians may be reluctant to adopt this technique because they may not be convinced of its advantages compared with the traditional methods of recording BP and because of difficulties with implementing new rooming workflows.12 The cost of AOBP devices used in this study (Omron 907 and BpTRU; BpTRU ceased operations in 2017) were not disclosed, which may be a hindrance, as devices may cost $1000 or more.
An online search for “automated oscillometric BP monitor” by one of the PURL authors (RCM) found oscillometric AOBP devices ranging from $150 to > $1000, depending on whether the device was medical grade; a search for “Omron 907” found devices for ≤ $599 on multiple sites. However, none of the lower-cost devices indicated the ability to take multiple, unattended BP readings.
ACKNOWLEDGEMENT
The PURLs Surveillance System was supported in part by Grant Number UL1RR024999 from the National Center for Research Resources, a Clinical Translational Science Award to the University of Chicago. The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Center for Research Resources or the National Institutes of Health.
1. Roerecke M, Kaczorowski J, Myers MG. Comparing automated office blood pressure readings with other methods of blood pressure measurement for identifying patients with possible hypertension: a systematic review and meta-analysis. JAMA Intern Med. 2019;179:351-362.
2. Whelton PK, Carey RM, Aronow WS, et al. 2017 ACC/AHA/AAPA/ABC/ACPM/AGS/APhA/ASH/ASPC/NMA/PCNA guideline for the prevention, detection, evaluation, and management of high blood pressure in adults: a report of the American College of Cardiology/American Heart Association Task Force on Clinical Practice Guidelines. Hypertension. 2018;71:e13-e115. Published correction appears in Hypertension. 2018;71:e140-e144.
3. James PA, Oparil S, Carter BL, et al. 2014 evidence-based guideline for the management of high blood pressure in adults: report from the panel members appointed to the Eighth Joint National Committee (JNC 8). JAMA. 2014;311:507-520. Published correction appears in JAMA. 2014;311:1809.
4. Fryar CD, Ostchega Y, Hales CM, et al. Hypertension prevalence and control among adults: United States, 2015-2016. NCHS Data Brief. 2017;(289):1-8.
5. GBD 2017 Risk Factor Collaborators. Global, regional, and national comparative risk assessment of 84 behavioural, environmental and occupational, and metabolic risks or clusters of risks for 195 countries and territories, 1990-2017: a systematic analysis for the Global Burden of Disease Study 2017. Lancet. 2018;392:1923-1994.
6. Kirkland EB, Heincelman M, Bishu KG, et al. Trends in healthcare expenditures among US adults with hypertension: national estimates, 2003-2014. J Am Heart Assoc. 2018;7:e008731.
7. Pickering TG, Hall JE, Appel LJ, et al. Recommendations for blood pressure measurement in humans and experimental animals: part 1: blood pressure measurement in humans: a statement for professionals from the Subcommittee of Professional and Public Education of the American Heart Association Council on High Blood Pressure Research. Circulation. 2005;111:697-716.
8. Ogedegbe G, Pickering T. Principles and techniques of blood pressure measurement. Cardiol Clin. 2010;28:571-586.
9. Pappaccogli M, Di Monaco S, Perlo E, et al. Comparison of automated office blood pressure with office and out-of-office measurement techniques. Hypertension. 2019;73:481-490.
10. Reeves RA. The rational clinical examination. Does this patient have hypertension? How to measure blood pressure. JAMA. 1995;273:1211-1218.
11. Myers MG, Valdivieso M, Kiss A. Use of automated office blood pressure measurement to reduce the white coat response. J Hypertens. 2009;27:280-286.
12. Centers for Medicare & Medicaid Services. Decision memo for ambulatory blood pressure monitoring (ABPM) (CAG-00067R2). July 2, 2019. Accessed September 29, 2020. www.cms.gov/medicare-coverage-database/details/nca-decision-memo.aspx?NCAId=294
1. Roerecke M, Kaczorowski J, Myers MG. Comparing automated office blood pressure readings with other methods of blood pressure measurement for identifying patients with possible hypertension: a systematic review and meta-analysis. JAMA Intern Med. 2019;179:351-362.
2. Whelton PK, Carey RM, Aronow WS, et al. 2017 ACC/AHA/AAPA/ABC/ACPM/AGS/APhA/ASH/ASPC/NMA/PCNA guideline for the prevention, detection, evaluation, and management of high blood pressure in adults: a report of the American College of Cardiology/American Heart Association Task Force on Clinical Practice Guidelines. Hypertension. 2018;71:e13-e115. Published correction appears in Hypertension. 2018;71:e140-e144.
3. James PA, Oparil S, Carter BL, et al. 2014 evidence-based guideline for the management of high blood pressure in adults: report from the panel members appointed to the Eighth Joint National Committee (JNC 8). JAMA. 2014;311:507-520. Published correction appears in JAMA. 2014;311:1809.
4. Fryar CD, Ostchega Y, Hales CM, et al. Hypertension prevalence and control among adults: United States, 2015-2016. NCHS Data Brief. 2017;(289):1-8.
5. GBD 2017 Risk Factor Collaborators. Global, regional, and national comparative risk assessment of 84 behavioural, environmental and occupational, and metabolic risks or clusters of risks for 195 countries and territories, 1990-2017: a systematic analysis for the Global Burden of Disease Study 2017. Lancet. 2018;392:1923-1994.
6. Kirkland EB, Heincelman M, Bishu KG, et al. Trends in healthcare expenditures among US adults with hypertension: national estimates, 2003-2014. J Am Heart Assoc. 2018;7:e008731.
7. Pickering TG, Hall JE, Appel LJ, et al. Recommendations for blood pressure measurement in humans and experimental animals: part 1: blood pressure measurement in humans: a statement for professionals from the Subcommittee of Professional and Public Education of the American Heart Association Council on High Blood Pressure Research. Circulation. 2005;111:697-716.
8. Ogedegbe G, Pickering T. Principles and techniques of blood pressure measurement. Cardiol Clin. 2010;28:571-586.
9. Pappaccogli M, Di Monaco S, Perlo E, et al. Comparison of automated office blood pressure with office and out-of-office measurement techniques. Hypertension. 2019;73:481-490.
10. Reeves RA. The rational clinical examination. Does this patient have hypertension? How to measure blood pressure. JAMA. 1995;273:1211-1218.
11. Myers MG, Valdivieso M, Kiss A. Use of automated office blood pressure measurement to reduce the white coat response. J Hypertens. 2009;27:280-286.
12. Centers for Medicare & Medicaid Services. Decision memo for ambulatory blood pressure monitoring (ABPM) (CAG-00067R2). July 2, 2019. Accessed September 29, 2020. www.cms.gov/medicare-coverage-database/details/nca-decision-memo.aspx?NCAId=294
PRACTICE CHANGER
Measure patients’ blood pressure (BP) using an oscillometric, fully automated office BP device, with the patient sitting alone in a quiet exam room, to accurately diagnose hypertension and eliminate the “white-coat” effect.
STRENGTH OF RECOMMENDATION
B: Based on a systematic review and meta-analysis of randomized controlled trials and cohort studies.1
Roerecke M, Kaczorowski J, Myers MG. Comparing automated office blood pressure readings with other methods of blood pressure measurement for identifying patients with possible hypertension: a systematic review and meta-analysis. JAMA Intern Med. 2019;179:351-362.
A review of the latest USPSTF recommendations
Since the last Practice Alert update on recommendations made by the US Preventive Services Task Force,1 the Task Force has completed work on 12 topics (TABLE 1).2-17 Five of these topics have been discussed in JFP audio recordings, and the links are provided in TABLE 1.
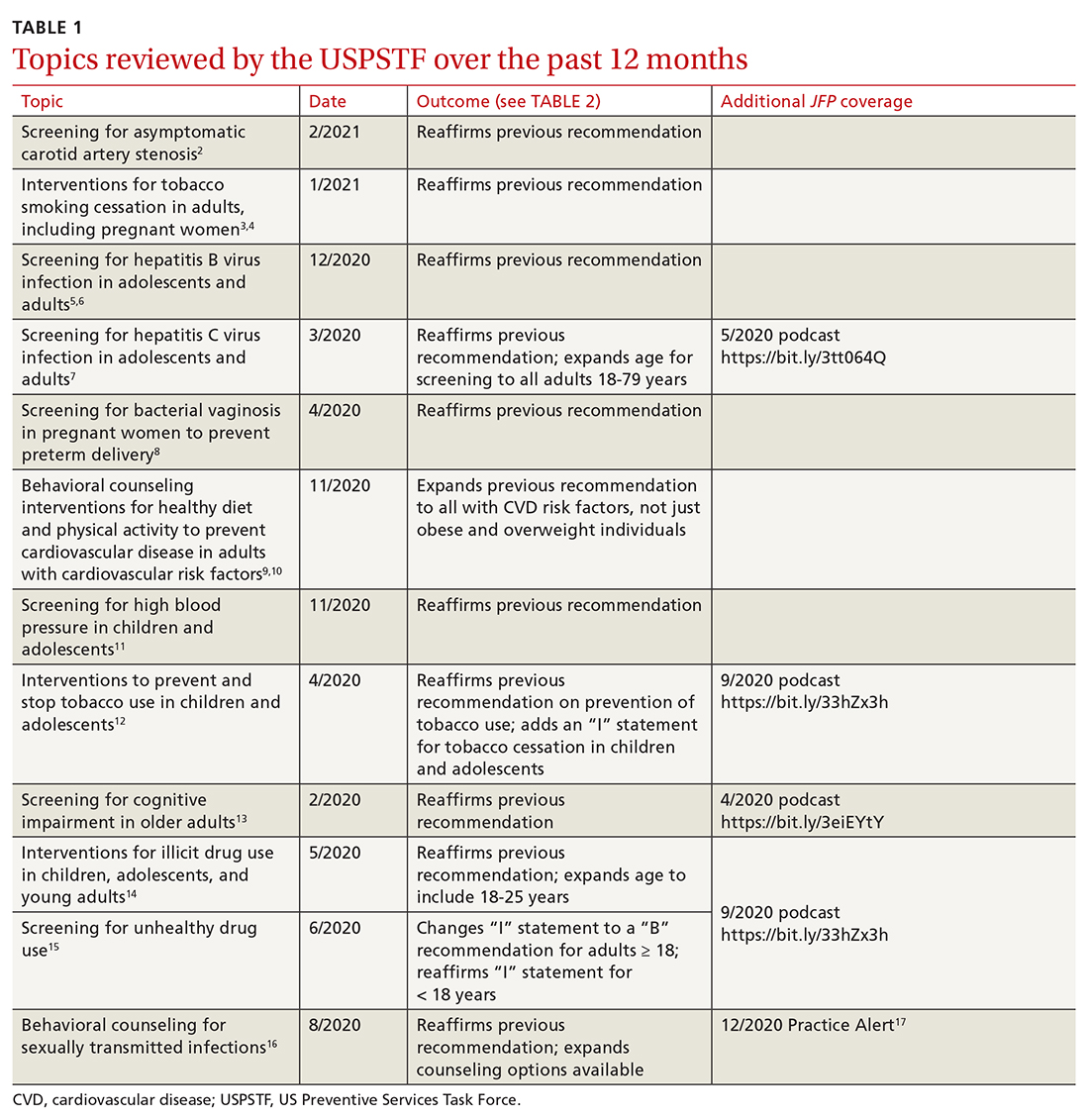
This latest Task Force endeavor resulted in 18 recommendations (TABLE 2), all of which reaffirm previous recommendations on these topics and expand the scope of 2. There were 2 “A” recommendations, 6 “B” recommendations, 2 “D” recommendations, and 8 “I” statements, indicating that there was insufficient evidence to assess effectiveness or harms. The willingness to make “I” statements when there is little or no evidence on the intervention being assessed distinguishes the USPSTF from other clinical guideline committees.

Screening for carotid artery stenosis
One of the “D” recommendations this past year reaffirms the prior recommendation against screening for carotid artery stenosis in asymptomatic adults—ie, those without a history of transient ischemic attack, stroke, or neurologic signs or symptoms that might be caused by carotid artery stenosis.2 The screening tests the Task Force researched included carotid duplex ultrasonography (DUS), magnetic resonance angiography, and computed tomography angiography. The Task Force did not look at the value of auscultation for carotid bruits because it has been proven to be inaccurate and they do not consider it to be a useful screening tool.
The Task Force based its “D” recommendation on a lack of evidence for any benefit in detecting asymptomatic carotid artery stenosis, and on evidence that screening can lead to harms through false-positive tests and potential complications from carotid endarterectomy and carotid artery angioplasty and stenting. In its clinical considerations, the Task Force emphasized the primary prevention of atherosclerotic disease by focusing on the following actions:
- screening for high blood pressure in adults
- encouraging tobacco smoking cessation in adults
- promoting a healthy diet and physical activity in adults with cardiovascular risk factors
- recommending aspirin use to prevent cardiovascular disease and colorectal cancer
- advising statin use for the primary prevention of cardiovascular disease in adults ages 45 to 75 years who have 1 or more risk factors (hyperlipidemia, diabetes, hypertension, smoking) and those with a 10-year risk of a cardiovascular event of 10% or greater.
This “D” recommendation differs from recommendations made by other professional organizations, some of which recommend testing with DUS for asymptomatic patients with a carotid bruit, and others that recommend DUS screening in patients with multiple risk factors for stroke and in those with known peripheral artery disease or other cardiovascular disease.18,19
Smoking cessation in adults
Smoking tobacco is the leading preventable cause of death in the United States, causing about 480,000 deaths annually.3 Smoking during pregnancy increases the risk of complications including miscarriage, congenital anomalies, stillbirth, fetal growth restriction, preterm birth, and placental abruption.
The Task Force published recommendations earlier this year advising all clinicians to ask all adult patients about tobacco use; and, for those who smoke, to provide (or refer them to) smoking cessation behavioral therapy. The Task Force also recommends prescribing pharmacotherapy approved by the Food and Drug Administration (FDA) for smoking cessation for nonpregnant adults. (There is a lack of information to assess the harms and benefits of smoking cessation pharmacotherapy during pregnancy.)
Continue to: FDA-approved medications...
FDA-approved medications for treating tobacco smoking dependence are nicotine replacement therapy (NRT), bupropion hydrochloride, and varenicline.3 NRT is available in transdermal patches, lozenges, gum, inhalers, and nasal sprays.
In addition, the Task Force indicates that there is insufficient evidence to assess the benefits and harms of e-cigarettes when used as a method of achieving smoking cessation: “Few randomized trials have evaluated the effectiveness of e-cigarettes to increase tobacco smoking cessation in nonpregnant adults, and no trials have evaluated e-cigarettes for tobacco smoking cessation in pregnant persons.”4
Hepatitis B infection screening
The Task Force reaffirmed a previous recommendation to screen for hepatitis B virus (HBV) infection only in adults who are at high risk,5 rather than universal screening that it recommends for hepatitis C virus infection (HCV).7 (See: https://bit.ly/3tt064Q). The Task Force has a separate recommendation to screen all pregnant women for hepatitis B at the first prenatal visit.6
Those at high risk for hepatitis B who should be screened include individuals born in countries or regions of the world with a hepatitis B surface antigen (HBsAg) prevalence ≥ 2% and individuals born in the United States who have not received HBV vaccine and whose parents were born in regions with an HBsAg prevalence ≥ 8%.5 (A table listing countries with HBsAg ≥ 8%—as well as those in lower prevalence categories—is included with the recommendation.5)
HBV screening should also be offered to other high-risk groups that have a prevalence of positive HBsAg ≥ 2%: those who have injected drugs in the past or are currently injecting drugs; men who have sex with men; individuals with HIV; and sex partners, needle-sharing contacts, and household contacts of people known to be HBsAg positive.5
Continue to: It is estimated that...
It is estimated that > 860,000 people in the United States have chronic HBV infection and that close to two-thirds of them are unaware of their infection.5 The screening test for HBV is highly accurate; sensitivity and specificity are both > 98%.5 While there is no direct evidence that screening, detecting, and treating asymptomatic HBV infection reduces morbidity and mortality, the Task Force felt that the evidence for improvement in multiple outcomes in those with HBV when treated with antiviral regimens was sufficient to support the recommendation.
Screening for bacterial vaginosis in pregnancy
While bacterial vaginosis (BV) is associated with a two-fold risk of preterm delivery, treating BV during pregnancy does not seem to reduce this risk, indicating that some other variable is involved.8 In addition, studies that looked at screening for, and treatment of, asymptomatic BV in pregnant women at high risk for preterm delivery (defined primarily as those with a previous preterm delivery) have shown inconsistent results. There is the potential for harm in treating BV in pregnancy, chiefly involving gastrointestinal upset caused by metronidazole or clindamycin.
Given that there are no benefits—and some harms—resulting from treatment, the Task Force recommends against screening for BV in non-high-risk pregnant women. A lack of sufficient information to assess any potential benefits to screening in high-risk pregnancies led the Task Force to an “I” statement on this question.8
Behavioral counseling on healthy diet, exercise for adults with CV risks
Cardiovascular disease (CVD) remains the number one cause of death in the United States. The major risk factors for CVD, which can be modified, are high blood pressure, hyperlipidemia, diabetes, smoking, obesity or overweight, and lack of physical activity.
The Task Force has previously recommended intensive behavioral interventions to improve nutrition and physical activity in those who are overweight/obese and in those with abnormal blood glucose levels,9 and has addressed smoking prevention and cessation.4 This new recommendation applies to those with other CVD risks such as high blood pressure and/or hyperlipidemia and those with an estimated 10-year CVD risk of ≥ 7.5%.10
Continue to: Behavioral interventions...
Behavioral interventions included in the Task Force analysis employed a median of 12 contacts and an estimated 6 hours of contact time over 6 to 18 months.10 Most interventions involved motivational interviewing and instruction on behavioral change methods. These interventions can be provided by primary care clinicians, as well as a wide range of other trained professionals. The Affordable Care Act dictates that all “A” and “B” recommendations must be provided by commercial health plans at no out-of-pocket expense for the patient.
Nutritional advice should include reductions in saturated fats, salt, and sugars and increases in fruits, vegetables, and whole grains. The Mediterranean diet and the Dietary Approaches to Stop Hypertension (DASH) diet are often recommended.10 Physical activity counseling should advocate for 90 to 180 minutes per week of moderate to vigorous activity.
This new recommendation, along with the previous ones pertaining to behavioral interventions for lifestyle changes, make it clear that intensive interventions are needed to achieve meaningful change. Simple advice from a clinician will have little to no effect.
Task Force reviews evidence on HTN, smoking cessation in young people
In 2020 the Task Force completed reviews of evidence relevant to screening for high blood pressure11 and
The 2 “I” statements are in disagreement with recommendations of other professional organizations. The American Academy of Pediatrics (AAP) and the American Heart Association recommend routine screening for high blood pressure starting at age 3 years. And the AAP recommends screening teenagers for tobacco use and offering tobacco dependence treatment, referral, or both (including pharmacotherapy) when indicated. E-cigarettes are not recommended as a treatment for tobacco dependence.20
Continue to: The difference between...
The difference between the methods used by the Task Force and other guideline-producing organizations becomes apparent when it comes to recommendations pertaining to children and adolescents, for whom long-term outcome-oriented studies on prevention issues are rare. The Task Force is unwilling to make recommendations when evidence does not exist. The AAP often makes recommendations based on expert opinion consensus in such situations. One notable part of each Task Force recommendation statement is a discussion of what other organizations recommend on the same topic so that these differences can be openly described.
Better Task Force funding could expand topic coverage
It is worth revisiting 2 issues that were pointed out in last year’s USPSTF summary in this column.1 First, the Task Force methods are robust and evidence based, and recommendations therefore are rarely changed once they are made at an “A”, “B”, or “D” level. Second, Task Force resources are finite, and thus, the group is currently unable to update previous recommendations with greater frequency or to consider many new topics. In the past 2 years, the Task Force has developed recommendations on only 2 completely new topics. Hopefully, its budget can be expanded so that new topics can be added in the future.
1. Campos-Outcalt D. USPSTF roundup. J Fam Pract. 2020;69:201-204.
2. USPSTF. Screening for asymptomatic carotid artery stenosis. Accessed April 30, 2021. https://uspreventiveservicestaskforce.org/uspstf/recommendation/carotid-artery-stenosis-screening
3. USPSTF. Interventions for tobacco smoking cessation in adults, including pregnant persons. Accessed April 30, 2021. www.uspreventiveservicestaskforce.org/uspstf/recommendation/tobacco-use-in-adults-and-pregnant-women-counseling-and-interventions
4. USPSTF. Interventions for tobacco smoking cessation in adults, including pregnant persons. JAMA. 2021;325:265-279.
5. USPSTF. Screening for Hepatitis B virus infection in adolescents and adults. Accessed April 30, 2021. https://uspreventiveservicestaskforce.org/uspstf/recommendation/hepatitis-b-virus-infection-screening
6. USPSTF. Hepatitis B virus infection in pregnant women: screening. Accessed April 30, 2021. https://uspreventiveservicestaskforce.org/uspstf/recommendation/hepatitis-b-virus-infection-in-pregnant-women-screening
7. USPSTF. Hepatitis C virus infection in adolescents and adults: screening. Accessed April 30, 2021. https://uspreventiveservicestaskforce.org/uspstf/recommendation/hepatitis-c-screening
8. USPSTF; Owens DK, Davidson KW, Krisk AH, et al. Screening for bacterial vaginosis in pregnant persons to prevent preterm delivery: US Preventive Services Task Force recommendation statement. JAMA. 2020;323:1286-1292.
9. Behavioral counseling to promote a healthful diet and physical activity for cardiovascular disease prevention in adults with cardiovascular risk factors: U.S. Preventive Services Task Force recommendation statement. Ann Intern Med. 2014;161:587-593.
10. USPSTF. Behavioral counseling interventions to promote a healthy and physical activity for cardiovascular disease prevention in adults with cardiovascular risk factors: US Preventive Services Task Force recommendation statement. JAMA. 2020;324:2069-2075.
11. USPSTF. High blood pressure in children and adolescents: screening. Accessed April 30, 2021. https://uspreventiveservicestaskforce.org/uspstf/recommendation/blood-pressure-in-children-and-adolescents-hypertension-screening
12. USPSTF. Prevention and cessation of tobacco use in children and adolescents: primary care interventions. Accessed April 30, 2021. https://uspreventiveservicestaskforce.org/uspstf/recommendation/tobacco-and-nicotine-use-prevention-in-children-and-adolescents-primary-care-interventions
13. USPSTF. Cognitive impairment in older adults: screening. Accessed March 26, 2021. https://uspreventiveservicestaskforce.org/uspstf/recommendation/cognitive-impairment-in-older-adults-screening
14. USPSTF. Illicit drug use in children, adolescents, and young adults: primary care-based interventions. Accessed April 30, 2021. https://uspreventiveservicestaskforce.org/uspstf/recommendation/drug-use-illicit-primary-care-interventions-for-children-and-adolescents
15. USPSTF. Unhealthy drug use: screening. Accessed April 30, 2021. https://uspreventiveservicestaskforce.org/uspstf/recommendation/drug-use-illicit-screening
16. USPSTF. Sexually transmitted infections: behavioral counseling. Accessed April 30, 2021. https://uspreventiveservicestaskforce.org/uspstf/recommendation/sexually-transmitted-infections-behavioral-counseling.
17. Campos-Outcalt D. USPSTF update on sexually transmitted infections. J Fam Pract. 2020;69:514-517.
18. Brott TG, Halperin JL, Abbara S, et al; ASA/ACCF/AHA/AANN/AANS/ACR/ASNR/CNS/SAIP/SCAI/SIR/SNIS/SVM/SVS guideline on the management of patients with extracranial carotid and vertebral artery disease. Catheter Cardiovasc Interv. 2013;81:E76-E123.
19. Ricotta JJ, Aburahma A, Ascher E, et al; Society for Vascular Surgery. Updated Society for Vascular Surgery guidelines for management of extracranial carotid disease. J Vasc Surg. 2011;54:e1-e31.
20. Farber HJ, Walley SC, Groner JA, et al; Section on Tobacco Control. Clinical practice policy to protect children from tobacco, nicotine, and tobacco smoke. Pediatrics. 2015;136:1008-1017.
Since the last Practice Alert update on recommendations made by the US Preventive Services Task Force,1 the Task Force has completed work on 12 topics (TABLE 1).2-17 Five of these topics have been discussed in JFP audio recordings, and the links are provided in TABLE 1.

This latest Task Force endeavor resulted in 18 recommendations (TABLE 2), all of which reaffirm previous recommendations on these topics and expand the scope of 2. There were 2 “A” recommendations, 6 “B” recommendations, 2 “D” recommendations, and 8 “I” statements, indicating that there was insufficient evidence to assess effectiveness or harms. The willingness to make “I” statements when there is little or no evidence on the intervention being assessed distinguishes the USPSTF from other clinical guideline committees.

Screening for carotid artery stenosis
One of the “D” recommendations this past year reaffirms the prior recommendation against screening for carotid artery stenosis in asymptomatic adults—ie, those without a history of transient ischemic attack, stroke, or neurologic signs or symptoms that might be caused by carotid artery stenosis.2 The screening tests the Task Force researched included carotid duplex ultrasonography (DUS), magnetic resonance angiography, and computed tomography angiography. The Task Force did not look at the value of auscultation for carotid bruits because it has been proven to be inaccurate and they do not consider it to be a useful screening tool.
The Task Force based its “D” recommendation on a lack of evidence for any benefit in detecting asymptomatic carotid artery stenosis, and on evidence that screening can lead to harms through false-positive tests and potential complications from carotid endarterectomy and carotid artery angioplasty and stenting. In its clinical considerations, the Task Force emphasized the primary prevention of atherosclerotic disease by focusing on the following actions:
- screening for high blood pressure in adults
- encouraging tobacco smoking cessation in adults
- promoting a healthy diet and physical activity in adults with cardiovascular risk factors
- recommending aspirin use to prevent cardiovascular disease and colorectal cancer
- advising statin use for the primary prevention of cardiovascular disease in adults ages 45 to 75 years who have 1 or more risk factors (hyperlipidemia, diabetes, hypertension, smoking) and those with a 10-year risk of a cardiovascular event of 10% or greater.
This “D” recommendation differs from recommendations made by other professional organizations, some of which recommend testing with DUS for asymptomatic patients with a carotid bruit, and others that recommend DUS screening in patients with multiple risk factors for stroke and in those with known peripheral artery disease or other cardiovascular disease.18,19
Smoking cessation in adults
Smoking tobacco is the leading preventable cause of death in the United States, causing about 480,000 deaths annually.3 Smoking during pregnancy increases the risk of complications including miscarriage, congenital anomalies, stillbirth, fetal growth restriction, preterm birth, and placental abruption.
The Task Force published recommendations earlier this year advising all clinicians to ask all adult patients about tobacco use; and, for those who smoke, to provide (or refer them to) smoking cessation behavioral therapy. The Task Force also recommends prescribing pharmacotherapy approved by the Food and Drug Administration (FDA) for smoking cessation for nonpregnant adults. (There is a lack of information to assess the harms and benefits of smoking cessation pharmacotherapy during pregnancy.)
Continue to: FDA-approved medications...
FDA-approved medications for treating tobacco smoking dependence are nicotine replacement therapy (NRT), bupropion hydrochloride, and varenicline.3 NRT is available in transdermal patches, lozenges, gum, inhalers, and nasal sprays.
In addition, the Task Force indicates that there is insufficient evidence to assess the benefits and harms of e-cigarettes when used as a method of achieving smoking cessation: “Few randomized trials have evaluated the effectiveness of e-cigarettes to increase tobacco smoking cessation in nonpregnant adults, and no trials have evaluated e-cigarettes for tobacco smoking cessation in pregnant persons.”4
Hepatitis B infection screening
The Task Force reaffirmed a previous recommendation to screen for hepatitis B virus (HBV) infection only in adults who are at high risk,5 rather than universal screening that it recommends for hepatitis C virus infection (HCV).7 (See: https://bit.ly/3tt064Q). The Task Force has a separate recommendation to screen all pregnant women for hepatitis B at the first prenatal visit.6
Those at high risk for hepatitis B who should be screened include individuals born in countries or regions of the world with a hepatitis B surface antigen (HBsAg) prevalence ≥ 2% and individuals born in the United States who have not received HBV vaccine and whose parents were born in regions with an HBsAg prevalence ≥ 8%.5 (A table listing countries with HBsAg ≥ 8%—as well as those in lower prevalence categories—is included with the recommendation.5)
HBV screening should also be offered to other high-risk groups that have a prevalence of positive HBsAg ≥ 2%: those who have injected drugs in the past or are currently injecting drugs; men who have sex with men; individuals with HIV; and sex partners, needle-sharing contacts, and household contacts of people known to be HBsAg positive.5
Continue to: It is estimated that...
It is estimated that > 860,000 people in the United States have chronic HBV infection and that close to two-thirds of them are unaware of their infection.5 The screening test for HBV is highly accurate; sensitivity and specificity are both > 98%.5 While there is no direct evidence that screening, detecting, and treating asymptomatic HBV infection reduces morbidity and mortality, the Task Force felt that the evidence for improvement in multiple outcomes in those with HBV when treated with antiviral regimens was sufficient to support the recommendation.
Screening for bacterial vaginosis in pregnancy
While bacterial vaginosis (BV) is associated with a two-fold risk of preterm delivery, treating BV during pregnancy does not seem to reduce this risk, indicating that some other variable is involved.8 In addition, studies that looked at screening for, and treatment of, asymptomatic BV in pregnant women at high risk for preterm delivery (defined primarily as those with a previous preterm delivery) have shown inconsistent results. There is the potential for harm in treating BV in pregnancy, chiefly involving gastrointestinal upset caused by metronidazole or clindamycin.
Given that there are no benefits—and some harms—resulting from treatment, the Task Force recommends against screening for BV in non-high-risk pregnant women. A lack of sufficient information to assess any potential benefits to screening in high-risk pregnancies led the Task Force to an “I” statement on this question.8
Behavioral counseling on healthy diet, exercise for adults with CV risks
Cardiovascular disease (CVD) remains the number one cause of death in the United States. The major risk factors for CVD, which can be modified, are high blood pressure, hyperlipidemia, diabetes, smoking, obesity or overweight, and lack of physical activity.
The Task Force has previously recommended intensive behavioral interventions to improve nutrition and physical activity in those who are overweight/obese and in those with abnormal blood glucose levels,9 and has addressed smoking prevention and cessation.4 This new recommendation applies to those with other CVD risks such as high blood pressure and/or hyperlipidemia and those with an estimated 10-year CVD risk of ≥ 7.5%.10
Continue to: Behavioral interventions...
Behavioral interventions included in the Task Force analysis employed a median of 12 contacts and an estimated 6 hours of contact time over 6 to 18 months.10 Most interventions involved motivational interviewing and instruction on behavioral change methods. These interventions can be provided by primary care clinicians, as well as a wide range of other trained professionals. The Affordable Care Act dictates that all “A” and “B” recommendations must be provided by commercial health plans at no out-of-pocket expense for the patient.
Nutritional advice should include reductions in saturated fats, salt, and sugars and increases in fruits, vegetables, and whole grains. The Mediterranean diet and the Dietary Approaches to Stop Hypertension (DASH) diet are often recommended.10 Physical activity counseling should advocate for 90 to 180 minutes per week of moderate to vigorous activity.
This new recommendation, along with the previous ones pertaining to behavioral interventions for lifestyle changes, make it clear that intensive interventions are needed to achieve meaningful change. Simple advice from a clinician will have little to no effect.
Task Force reviews evidence on HTN, smoking cessation in young people
In 2020 the Task Force completed reviews of evidence relevant to screening for high blood pressure11 and
The 2 “I” statements are in disagreement with recommendations of other professional organizations. The American Academy of Pediatrics (AAP) and the American Heart Association recommend routine screening for high blood pressure starting at age 3 years. And the AAP recommends screening teenagers for tobacco use and offering tobacco dependence treatment, referral, or both (including pharmacotherapy) when indicated. E-cigarettes are not recommended as a treatment for tobacco dependence.20
Continue to: The difference between...
The difference between the methods used by the Task Force and other guideline-producing organizations becomes apparent when it comes to recommendations pertaining to children and adolescents, for whom long-term outcome-oriented studies on prevention issues are rare. The Task Force is unwilling to make recommendations when evidence does not exist. The AAP often makes recommendations based on expert opinion consensus in such situations. One notable part of each Task Force recommendation statement is a discussion of what other organizations recommend on the same topic so that these differences can be openly described.
Better Task Force funding could expand topic coverage
It is worth revisiting 2 issues that were pointed out in last year’s USPSTF summary in this column.1 First, the Task Force methods are robust and evidence based, and recommendations therefore are rarely changed once they are made at an “A”, “B”, or “D” level. Second, Task Force resources are finite, and thus, the group is currently unable to update previous recommendations with greater frequency or to consider many new topics. In the past 2 years, the Task Force has developed recommendations on only 2 completely new topics. Hopefully, its budget can be expanded so that new topics can be added in the future.
Since the last Practice Alert update on recommendations made by the US Preventive Services Task Force,1 the Task Force has completed work on 12 topics (TABLE 1).2-17 Five of these topics have been discussed in JFP audio recordings, and the links are provided in TABLE 1.

This latest Task Force endeavor resulted in 18 recommendations (TABLE 2), all of which reaffirm previous recommendations on these topics and expand the scope of 2. There were 2 “A” recommendations, 6 “B” recommendations, 2 “D” recommendations, and 8 “I” statements, indicating that there was insufficient evidence to assess effectiveness or harms. The willingness to make “I” statements when there is little or no evidence on the intervention being assessed distinguishes the USPSTF from other clinical guideline committees.

Screening for carotid artery stenosis
One of the “D” recommendations this past year reaffirms the prior recommendation against screening for carotid artery stenosis in asymptomatic adults—ie, those without a history of transient ischemic attack, stroke, or neurologic signs or symptoms that might be caused by carotid artery stenosis.2 The screening tests the Task Force researched included carotid duplex ultrasonography (DUS), magnetic resonance angiography, and computed tomography angiography. The Task Force did not look at the value of auscultation for carotid bruits because it has been proven to be inaccurate and they do not consider it to be a useful screening tool.
The Task Force based its “D” recommendation on a lack of evidence for any benefit in detecting asymptomatic carotid artery stenosis, and on evidence that screening can lead to harms through false-positive tests and potential complications from carotid endarterectomy and carotid artery angioplasty and stenting. In its clinical considerations, the Task Force emphasized the primary prevention of atherosclerotic disease by focusing on the following actions:
- screening for high blood pressure in adults
- encouraging tobacco smoking cessation in adults
- promoting a healthy diet and physical activity in adults with cardiovascular risk factors
- recommending aspirin use to prevent cardiovascular disease and colorectal cancer
- advising statin use for the primary prevention of cardiovascular disease in adults ages 45 to 75 years who have 1 or more risk factors (hyperlipidemia, diabetes, hypertension, smoking) and those with a 10-year risk of a cardiovascular event of 10% or greater.
This “D” recommendation differs from recommendations made by other professional organizations, some of which recommend testing with DUS for asymptomatic patients with a carotid bruit, and others that recommend DUS screening in patients with multiple risk factors for stroke and in those with known peripheral artery disease or other cardiovascular disease.18,19
Smoking cessation in adults
Smoking tobacco is the leading preventable cause of death in the United States, causing about 480,000 deaths annually.3 Smoking during pregnancy increases the risk of complications including miscarriage, congenital anomalies, stillbirth, fetal growth restriction, preterm birth, and placental abruption.
The Task Force published recommendations earlier this year advising all clinicians to ask all adult patients about tobacco use; and, for those who smoke, to provide (or refer them to) smoking cessation behavioral therapy. The Task Force also recommends prescribing pharmacotherapy approved by the Food and Drug Administration (FDA) for smoking cessation for nonpregnant adults. (There is a lack of information to assess the harms and benefits of smoking cessation pharmacotherapy during pregnancy.)
Continue to: FDA-approved medications...
FDA-approved medications for treating tobacco smoking dependence are nicotine replacement therapy (NRT), bupropion hydrochloride, and varenicline.3 NRT is available in transdermal patches, lozenges, gum, inhalers, and nasal sprays.
In addition, the Task Force indicates that there is insufficient evidence to assess the benefits and harms of e-cigarettes when used as a method of achieving smoking cessation: “Few randomized trials have evaluated the effectiveness of e-cigarettes to increase tobacco smoking cessation in nonpregnant adults, and no trials have evaluated e-cigarettes for tobacco smoking cessation in pregnant persons.”4
Hepatitis B infection screening
The Task Force reaffirmed a previous recommendation to screen for hepatitis B virus (HBV) infection only in adults who are at high risk,5 rather than universal screening that it recommends for hepatitis C virus infection (HCV).7 (See: https://bit.ly/3tt064Q). The Task Force has a separate recommendation to screen all pregnant women for hepatitis B at the first prenatal visit.6
Those at high risk for hepatitis B who should be screened include individuals born in countries or regions of the world with a hepatitis B surface antigen (HBsAg) prevalence ≥ 2% and individuals born in the United States who have not received HBV vaccine and whose parents were born in regions with an HBsAg prevalence ≥ 8%.5 (A table listing countries with HBsAg ≥ 8%—as well as those in lower prevalence categories—is included with the recommendation.5)
HBV screening should also be offered to other high-risk groups that have a prevalence of positive HBsAg ≥ 2%: those who have injected drugs in the past or are currently injecting drugs; men who have sex with men; individuals with HIV; and sex partners, needle-sharing contacts, and household contacts of people known to be HBsAg positive.5
Continue to: It is estimated that...
It is estimated that > 860,000 people in the United States have chronic HBV infection and that close to two-thirds of them are unaware of their infection.5 The screening test for HBV is highly accurate; sensitivity and specificity are both > 98%.5 While there is no direct evidence that screening, detecting, and treating asymptomatic HBV infection reduces morbidity and mortality, the Task Force felt that the evidence for improvement in multiple outcomes in those with HBV when treated with antiviral regimens was sufficient to support the recommendation.
Screening for bacterial vaginosis in pregnancy
While bacterial vaginosis (BV) is associated with a two-fold risk of preterm delivery, treating BV during pregnancy does not seem to reduce this risk, indicating that some other variable is involved.8 In addition, studies that looked at screening for, and treatment of, asymptomatic BV in pregnant women at high risk for preterm delivery (defined primarily as those with a previous preterm delivery) have shown inconsistent results. There is the potential for harm in treating BV in pregnancy, chiefly involving gastrointestinal upset caused by metronidazole or clindamycin.
Given that there are no benefits—and some harms—resulting from treatment, the Task Force recommends against screening for BV in non-high-risk pregnant women. A lack of sufficient information to assess any potential benefits to screening in high-risk pregnancies led the Task Force to an “I” statement on this question.8
Behavioral counseling on healthy diet, exercise for adults with CV risks
Cardiovascular disease (CVD) remains the number one cause of death in the United States. The major risk factors for CVD, which can be modified, are high blood pressure, hyperlipidemia, diabetes, smoking, obesity or overweight, and lack of physical activity.
The Task Force has previously recommended intensive behavioral interventions to improve nutrition and physical activity in those who are overweight/obese and in those with abnormal blood glucose levels,9 and has addressed smoking prevention and cessation.4 This new recommendation applies to those with other CVD risks such as high blood pressure and/or hyperlipidemia and those with an estimated 10-year CVD risk of ≥ 7.5%.10
Continue to: Behavioral interventions...
Behavioral interventions included in the Task Force analysis employed a median of 12 contacts and an estimated 6 hours of contact time over 6 to 18 months.10 Most interventions involved motivational interviewing and instruction on behavioral change methods. These interventions can be provided by primary care clinicians, as well as a wide range of other trained professionals. The Affordable Care Act dictates that all “A” and “B” recommendations must be provided by commercial health plans at no out-of-pocket expense for the patient.
Nutritional advice should include reductions in saturated fats, salt, and sugars and increases in fruits, vegetables, and whole grains. The Mediterranean diet and the Dietary Approaches to Stop Hypertension (DASH) diet are often recommended.10 Physical activity counseling should advocate for 90 to 180 minutes per week of moderate to vigorous activity.
This new recommendation, along with the previous ones pertaining to behavioral interventions for lifestyle changes, make it clear that intensive interventions are needed to achieve meaningful change. Simple advice from a clinician will have little to no effect.
Task Force reviews evidence on HTN, smoking cessation in young people
In 2020 the Task Force completed reviews of evidence relevant to screening for high blood pressure11 and
The 2 “I” statements are in disagreement with recommendations of other professional organizations. The American Academy of Pediatrics (AAP) and the American Heart Association recommend routine screening for high blood pressure starting at age 3 years. And the AAP recommends screening teenagers for tobacco use and offering tobacco dependence treatment, referral, or both (including pharmacotherapy) when indicated. E-cigarettes are not recommended as a treatment for tobacco dependence.20
Continue to: The difference between...
The difference between the methods used by the Task Force and other guideline-producing organizations becomes apparent when it comes to recommendations pertaining to children and adolescents, for whom long-term outcome-oriented studies on prevention issues are rare. The Task Force is unwilling to make recommendations when evidence does not exist. The AAP often makes recommendations based on expert opinion consensus in such situations. One notable part of each Task Force recommendation statement is a discussion of what other organizations recommend on the same topic so that these differences can be openly described.
Better Task Force funding could expand topic coverage
It is worth revisiting 2 issues that were pointed out in last year’s USPSTF summary in this column.1 First, the Task Force methods are robust and evidence based, and recommendations therefore are rarely changed once they are made at an “A”, “B”, or “D” level. Second, Task Force resources are finite, and thus, the group is currently unable to update previous recommendations with greater frequency or to consider many new topics. In the past 2 years, the Task Force has developed recommendations on only 2 completely new topics. Hopefully, its budget can be expanded so that new topics can be added in the future.
1. Campos-Outcalt D. USPSTF roundup. J Fam Pract. 2020;69:201-204.
2. USPSTF. Screening for asymptomatic carotid artery stenosis. Accessed April 30, 2021. https://uspreventiveservicestaskforce.org/uspstf/recommendation/carotid-artery-stenosis-screening
3. USPSTF. Interventions for tobacco smoking cessation in adults, including pregnant persons. Accessed April 30, 2021. www.uspreventiveservicestaskforce.org/uspstf/recommendation/tobacco-use-in-adults-and-pregnant-women-counseling-and-interventions
4. USPSTF. Interventions for tobacco smoking cessation in adults, including pregnant persons. JAMA. 2021;325:265-279.
5. USPSTF. Screening for Hepatitis B virus infection in adolescents and adults. Accessed April 30, 2021. https://uspreventiveservicestaskforce.org/uspstf/recommendation/hepatitis-b-virus-infection-screening
6. USPSTF. Hepatitis B virus infection in pregnant women: screening. Accessed April 30, 2021. https://uspreventiveservicestaskforce.org/uspstf/recommendation/hepatitis-b-virus-infection-in-pregnant-women-screening
7. USPSTF. Hepatitis C virus infection in adolescents and adults: screening. Accessed April 30, 2021. https://uspreventiveservicestaskforce.org/uspstf/recommendation/hepatitis-c-screening
8. USPSTF; Owens DK, Davidson KW, Krisk AH, et al. Screening for bacterial vaginosis in pregnant persons to prevent preterm delivery: US Preventive Services Task Force recommendation statement. JAMA. 2020;323:1286-1292.
9. Behavioral counseling to promote a healthful diet and physical activity for cardiovascular disease prevention in adults with cardiovascular risk factors: U.S. Preventive Services Task Force recommendation statement. Ann Intern Med. 2014;161:587-593.
10. USPSTF. Behavioral counseling interventions to promote a healthy and physical activity for cardiovascular disease prevention in adults with cardiovascular risk factors: US Preventive Services Task Force recommendation statement. JAMA. 2020;324:2069-2075.
11. USPSTF. High blood pressure in children and adolescents: screening. Accessed April 30, 2021. https://uspreventiveservicestaskforce.org/uspstf/recommendation/blood-pressure-in-children-and-adolescents-hypertension-screening
12. USPSTF. Prevention and cessation of tobacco use in children and adolescents: primary care interventions. Accessed April 30, 2021. https://uspreventiveservicestaskforce.org/uspstf/recommendation/tobacco-and-nicotine-use-prevention-in-children-and-adolescents-primary-care-interventions
13. USPSTF. Cognitive impairment in older adults: screening. Accessed March 26, 2021. https://uspreventiveservicestaskforce.org/uspstf/recommendation/cognitive-impairment-in-older-adults-screening
14. USPSTF. Illicit drug use in children, adolescents, and young adults: primary care-based interventions. Accessed April 30, 2021. https://uspreventiveservicestaskforce.org/uspstf/recommendation/drug-use-illicit-primary-care-interventions-for-children-and-adolescents
15. USPSTF. Unhealthy drug use: screening. Accessed April 30, 2021. https://uspreventiveservicestaskforce.org/uspstf/recommendation/drug-use-illicit-screening
16. USPSTF. Sexually transmitted infections: behavioral counseling. Accessed April 30, 2021. https://uspreventiveservicestaskforce.org/uspstf/recommendation/sexually-transmitted-infections-behavioral-counseling.
17. Campos-Outcalt D. USPSTF update on sexually transmitted infections. J Fam Pract. 2020;69:514-517.
18. Brott TG, Halperin JL, Abbara S, et al; ASA/ACCF/AHA/AANN/AANS/ACR/ASNR/CNS/SAIP/SCAI/SIR/SNIS/SVM/SVS guideline on the management of patients with extracranial carotid and vertebral artery disease. Catheter Cardiovasc Interv. 2013;81:E76-E123.
19. Ricotta JJ, Aburahma A, Ascher E, et al; Society for Vascular Surgery. Updated Society for Vascular Surgery guidelines for management of extracranial carotid disease. J Vasc Surg. 2011;54:e1-e31.
20. Farber HJ, Walley SC, Groner JA, et al; Section on Tobacco Control. Clinical practice policy to protect children from tobacco, nicotine, and tobacco smoke. Pediatrics. 2015;136:1008-1017.
1. Campos-Outcalt D. USPSTF roundup. J Fam Pract. 2020;69:201-204.
2. USPSTF. Screening for asymptomatic carotid artery stenosis. Accessed April 30, 2021. https://uspreventiveservicestaskforce.org/uspstf/recommendation/carotid-artery-stenosis-screening
3. USPSTF. Interventions for tobacco smoking cessation in adults, including pregnant persons. Accessed April 30, 2021. www.uspreventiveservicestaskforce.org/uspstf/recommendation/tobacco-use-in-adults-and-pregnant-women-counseling-and-interventions
4. USPSTF. Interventions for tobacco smoking cessation in adults, including pregnant persons. JAMA. 2021;325:265-279.
5. USPSTF. Screening for Hepatitis B virus infection in adolescents and adults. Accessed April 30, 2021. https://uspreventiveservicestaskforce.org/uspstf/recommendation/hepatitis-b-virus-infection-screening
6. USPSTF. Hepatitis B virus infection in pregnant women: screening. Accessed April 30, 2021. https://uspreventiveservicestaskforce.org/uspstf/recommendation/hepatitis-b-virus-infection-in-pregnant-women-screening
7. USPSTF. Hepatitis C virus infection in adolescents and adults: screening. Accessed April 30, 2021. https://uspreventiveservicestaskforce.org/uspstf/recommendation/hepatitis-c-screening
8. USPSTF; Owens DK, Davidson KW, Krisk AH, et al. Screening for bacterial vaginosis in pregnant persons to prevent preterm delivery: US Preventive Services Task Force recommendation statement. JAMA. 2020;323:1286-1292.
9. Behavioral counseling to promote a healthful diet and physical activity for cardiovascular disease prevention in adults with cardiovascular risk factors: U.S. Preventive Services Task Force recommendation statement. Ann Intern Med. 2014;161:587-593.
10. USPSTF. Behavioral counseling interventions to promote a healthy and physical activity for cardiovascular disease prevention in adults with cardiovascular risk factors: US Preventive Services Task Force recommendation statement. JAMA. 2020;324:2069-2075.
11. USPSTF. High blood pressure in children and adolescents: screening. Accessed April 30, 2021. https://uspreventiveservicestaskforce.org/uspstf/recommendation/blood-pressure-in-children-and-adolescents-hypertension-screening
12. USPSTF. Prevention and cessation of tobacco use in children and adolescents: primary care interventions. Accessed April 30, 2021. https://uspreventiveservicestaskforce.org/uspstf/recommendation/tobacco-and-nicotine-use-prevention-in-children-and-adolescents-primary-care-interventions
13. USPSTF. Cognitive impairment in older adults: screening. Accessed March 26, 2021. https://uspreventiveservicestaskforce.org/uspstf/recommendation/cognitive-impairment-in-older-adults-screening
14. USPSTF. Illicit drug use in children, adolescents, and young adults: primary care-based interventions. Accessed April 30, 2021. https://uspreventiveservicestaskforce.org/uspstf/recommendation/drug-use-illicit-primary-care-interventions-for-children-and-adolescents
15. USPSTF. Unhealthy drug use: screening. Accessed April 30, 2021. https://uspreventiveservicestaskforce.org/uspstf/recommendation/drug-use-illicit-screening
16. USPSTF. Sexually transmitted infections: behavioral counseling. Accessed April 30, 2021. https://uspreventiveservicestaskforce.org/uspstf/recommendation/sexually-transmitted-infections-behavioral-counseling.
17. Campos-Outcalt D. USPSTF update on sexually transmitted infections. J Fam Pract. 2020;69:514-517.
18. Brott TG, Halperin JL, Abbara S, et al; ASA/ACCF/AHA/AANN/AANS/ACR/ASNR/CNS/SAIP/SCAI/SIR/SNIS/SVM/SVS guideline on the management of patients with extracranial carotid and vertebral artery disease. Catheter Cardiovasc Interv. 2013;81:E76-E123.
19. Ricotta JJ, Aburahma A, Ascher E, et al; Society for Vascular Surgery. Updated Society for Vascular Surgery guidelines for management of extracranial carotid disease. J Vasc Surg. 2011;54:e1-e31.
20. Farber HJ, Walley SC, Groner JA, et al; Section on Tobacco Control. Clinical practice policy to protect children from tobacco, nicotine, and tobacco smoke. Pediatrics. 2015;136:1008-1017.
Systemic racism is a cause of health disparities
I applaud the joint statement by the editors of the family medicine journals to commit to the eradication of systemic racism in medicine ( J Fam Pract . 2021;70:3 -4). These are crucial times in our history, where proactive change is necessary. The leadership they have shown is important.
No one wants health disparities. So, to eliminate them, we need to know what they are and where they came from. In my presentations on health disparities to students, residents, and health care providers, I use 3 definitions of health disparities. My definitions are slightly different from those proposed in the seminal report, Unequal Treatment: Confronting Racial and Ethnic Disparities in Health Care, from the National Academy of Medicine (then Institute of Medicine).1 I like to think that my definitions elicit the information needed to guide change.
The first definition focuses on health statistics. When there are different outcomes for different demographic groups for the same disease, that is a disparity. This could be Black vs white, male vs female, or 1 zip code vs another.2 We owe ourselves an explanation for these differences if we are to be able to propose solutions.
Second, there are disparities in the provision of health care. If there are 2 individuals who present with the exact same symptoms, we need to ask ourselves why they would be treated differently. Even in systems where insurance status is the same, there are documented differences in care. A well-studied example of this is pain. In 1 such study, a meta-analysis showed that Blacks were less likely than whites to receive medication for acute pain in the emergency department (OR = 0.60 [95% CI, 0.43-0.83]).3 Other examples of differences by race include cardiac services,4 lung cancer screening,5 and stroke interventions.6
The third definition of health disparities involves differences in health-seeking behavior. This is not to blame the “victim,” but to understand the reason why the difference exists so that adequate interventions can be designed to improve outcomes. Traditionally, the concept of access referenced whether or not the patient had health insurance. But the provision of health insurance is insufficient to explain issues of access.7
Extrinsic and intrinsic factors at work. Factors related to insurance are an example of the extrinsic factors related to access. However, there are intrinsic factors related to access, most of which involve health literacy. We must ask ourselves: What are the best practices to educate patients to get the care they need? I will take this 1 step further; it is the duty of all health care professionals to improve health literacy 1 patient, 1 community at a time.
The next point that I make in my presentations on health disparities is that if you control for socioeconomic status, some of the health disparities go away. However, they rarely disappear. We measure socioeconomic status in a variety of ways: education, insurance status, income, and wealth. And as would be expected, these variables are usually correlated. We also know that these variables are not distributed equally by race. This is by design. This has been intentional. This has been, in many cases, our country’s policy. This is the result of systemic racism.
Continue to: It is necessary...
It is necessary for us to be willing to accept the toxicity of racism. This we can assess in 2 major ways. First, if we apply the Koch postulates or the Bradford Hill criteria for causation to racism, we can assess the degree to which racism is an explanation for health disparities. These principles offer methods for determining the relationship between risk and outcome.
Second, when we analyze the historical antecedents of health disparities, we find that racism is directly responsible not only for the current toxicity that Black people face today, but for the socioeconomic disparities that continue to exist. Let me give just a few examples.
- The Farm Security Administration was created in 1937 to avoid the collapse of the farming industry. As a compromise to southern legislators, a model was approved to allow local administration of support to farmers that essentially condoned the discrimination that had been occurring and would continue to occur—especially in the South.
- The National Housing Act of 1934 was created to provide stability to the banking industry at a time of national crisis. It subsidized a massive building program, and many of the units had restrictive covenants that prevented the sale to Blacks. It also codified redlining that prevented insured mortgages from being provided to Black communities.
- The Social Security Act of 1935 was created to provide benefits for the elderly and disabled. All workers were included except domestic workers and farm workers—the majority of whom were Black. This was another compromise that was made with southern congressman to get this act passed.
- The Servicemen’s Readjustment Act of 1944 (also known as the GI Bill) was passed to support veterans returning from World War II. Two major functions of the bill were to support educational opportunities for veterans and their families and to support the purchase of homes. From 1945 to 1954, the US added 13 million new homes. In 1946 and 1947, the Veterans Administration financed 40% of all single-family houses in the United States. Additionally, there were educational benefits for veterans to go to college or to learn a trade. These provisions, education, and housing were not equally available to Blacks. Columbia University professor Ira Katznelson called this act and others “affirmative action for whites.” 8
In 2019, the median income in white households was $76,057 and in Black households it was $46,073. 9 So, when we look at disparities of income, we must acknowledge this difference within the context of the current environment and the historical conditions that created these disparities. If we go 1 step further and look at disparities of wealth, we find that in 2019, the median wealth for white families was $188,200 and the median wealth for Black families was $24,100. 10
When one considers that a major contributor of wealth is home ownership, these differences seem logical—particularly related to points 1, 2, and 4 that I've just described. These economic disparities would not be as great today if the 4 examples given here (not to mention numerous other examples) had been administered equitably. The same applies to disparities in housing, employment, and education. Systemic racism is the causative agent. Systemic racism must be neutralized if we are to obtain anything close to health equity. 11
The Centers for Disease Control and Prevention (CDC) has recently taken new steps to recognize the role of racism in health. 12 The CDC plans to use “science to investigate and better understand the intersection of racism and health, and then to take action.” 13
It is time for the entire nation to recognize the links between racism and health outcomes and examine how we can design, implement, and evaluate interventions that will permanently correct these inequities.
1. Institute of Medicine. Unequal Treatment: Confronting Racial and Ethnic Disparities in Health Care; 2003. Accessed April 22, 2021. https://doi.org/10.17226/10260
2. Life Expectancy: Could where you live influence how long you live? Robert Wood Johnson Foundation. Accessed April 22, 2021. www.rwjf.org/en/library/interactives/whereyouliveaffectshowlongyoulive.html
3. Lee P, Le Saux M, Siegel R, et al. Racial and ethnic disparities in the management of acute pain in US emergency departments: meta-analysis and systematic review. Am J Emerg Med. 2019;37:1770-1777. doi: 10.1016/j.ajem.2019.06.014
4. Youmans QR, Hastings-Spaine L, Princewill O, et al. Disparities in cardiovascular care: past, present, and solutions. Cleve Clin J Med. 2019;86:621-632. doi: 10.3949/ccjm.86a.18088
5. Rivera MP, Katki HA, Tanner NT, et al. Addressing disparities in lung cancer screening eligibility and healthcare access. An official American Thoracic Society statement. Am J Respir Crit Care Med. 2020;202: e95-e112. doi: 10.1164/rccm.202008-3053ST
6. Rinaldo L, Rabinstein AA, Cloft H, et al. Racial and ethnic disparities in the utilization of thrombectomy for acute stroke. Stroke. 2019;50:2428-2432. doi:10.1161/STROKEAHA.118.024651
7. Hall AG, Lemak CH, Steingraber H, et al. Expanding the definition of access: it isn’t just about health insurance. J Health Care Poor Underserved. 2008;19:625-638. doi: 10.1353/hpu.0.0011
8. Katznelson I. When Affirmative Action Was White: An Untold History of Racial Inequality in Twentieth Century America. W. W. Norton & Co; 2006.
9. US Census Bureau. Households by Total Money Income, Race, and Hispanic Origin of Householder: 1967 to 2019. Accessed April 26, 2021. www.census.gov/data/tables/2020/demo/income-poverty/p60-270.html
10. Robb G. Long-standing wealth gap between Black and white Americans remains substantial, Fed data for 2019 show. MarketWatch. September 29, 2020. Accessed April 26, 2021. www.marketwatch.com/story/long-standing-wealth-gap-between-blacks-and-whites-remains-substantial-new-fed-data-for-2019-show-2020-09-28
11. Jones CP. Levels of racism: a theoretic framework and a gardener’s tale. Am J Public Health. 2000;9:1212-1215. doi: 10.2105/ajph.90.8.1212
12. CDC. Health equity: Director’s commentary. April 8, 2021. Accessed April 23, 2021. www.cdc.gov/healthequity/racism-disparities/director-commentary.html
13. CDC. Health equity: CDC’s efforts. April 8, 2021. Accessed April 23, 2021. www.cdc.gov/healthequity/racism-disparities/cdc-efforts.html
I applaud the joint statement by the editors of the family medicine journals to commit to the eradication of systemic racism in medicine ( J Fam Pract . 2021;70:3 -4). These are crucial times in our history, where proactive change is necessary. The leadership they have shown is important.
No one wants health disparities. So, to eliminate them, we need to know what they are and where they came from. In my presentations on health disparities to students, residents, and health care providers, I use 3 definitions of health disparities. My definitions are slightly different from those proposed in the seminal report, Unequal Treatment: Confronting Racial and Ethnic Disparities in Health Care, from the National Academy of Medicine (then Institute of Medicine).1 I like to think that my definitions elicit the information needed to guide change.
The first definition focuses on health statistics. When there are different outcomes for different demographic groups for the same disease, that is a disparity. This could be Black vs white, male vs female, or 1 zip code vs another.2 We owe ourselves an explanation for these differences if we are to be able to propose solutions.
Second, there are disparities in the provision of health care. If there are 2 individuals who present with the exact same symptoms, we need to ask ourselves why they would be treated differently. Even in systems where insurance status is the same, there are documented differences in care. A well-studied example of this is pain. In 1 such study, a meta-analysis showed that Blacks were less likely than whites to receive medication for acute pain in the emergency department (OR = 0.60 [95% CI, 0.43-0.83]).3 Other examples of differences by race include cardiac services,4 lung cancer screening,5 and stroke interventions.6
The third definition of health disparities involves differences in health-seeking behavior. This is not to blame the “victim,” but to understand the reason why the difference exists so that adequate interventions can be designed to improve outcomes. Traditionally, the concept of access referenced whether or not the patient had health insurance. But the provision of health insurance is insufficient to explain issues of access.7
Extrinsic and intrinsic factors at work. Factors related to insurance are an example of the extrinsic factors related to access. However, there are intrinsic factors related to access, most of which involve health literacy. We must ask ourselves: What are the best practices to educate patients to get the care they need? I will take this 1 step further; it is the duty of all health care professionals to improve health literacy 1 patient, 1 community at a time.
The next point that I make in my presentations on health disparities is that if you control for socioeconomic status, some of the health disparities go away. However, they rarely disappear. We measure socioeconomic status in a variety of ways: education, insurance status, income, and wealth. And as would be expected, these variables are usually correlated. We also know that these variables are not distributed equally by race. This is by design. This has been intentional. This has been, in many cases, our country’s policy. This is the result of systemic racism.
Continue to: It is necessary...
It is necessary for us to be willing to accept the toxicity of racism. This we can assess in 2 major ways. First, if we apply the Koch postulates or the Bradford Hill criteria for causation to racism, we can assess the degree to which racism is an explanation for health disparities. These principles offer methods for determining the relationship between risk and outcome.
Second, when we analyze the historical antecedents of health disparities, we find that racism is directly responsible not only for the current toxicity that Black people face today, but for the socioeconomic disparities that continue to exist. Let me give just a few examples.
- The Farm Security Administration was created in 1937 to avoid the collapse of the farming industry. As a compromise to southern legislators, a model was approved to allow local administration of support to farmers that essentially condoned the discrimination that had been occurring and would continue to occur—especially in the South.
- The National Housing Act of 1934 was created to provide stability to the banking industry at a time of national crisis. It subsidized a massive building program, and many of the units had restrictive covenants that prevented the sale to Blacks. It also codified redlining that prevented insured mortgages from being provided to Black communities.
- The Social Security Act of 1935 was created to provide benefits for the elderly and disabled. All workers were included except domestic workers and farm workers—the majority of whom were Black. This was another compromise that was made with southern congressman to get this act passed.
- The Servicemen’s Readjustment Act of 1944 (also known as the GI Bill) was passed to support veterans returning from World War II. Two major functions of the bill were to support educational opportunities for veterans and their families and to support the purchase of homes. From 1945 to 1954, the US added 13 million new homes. In 1946 and 1947, the Veterans Administration financed 40% of all single-family houses in the United States. Additionally, there were educational benefits for veterans to go to college or to learn a trade. These provisions, education, and housing were not equally available to Blacks. Columbia University professor Ira Katznelson called this act and others “affirmative action for whites.” 8
In 2019, the median income in white households was $76,057 and in Black households it was $46,073. 9 So, when we look at disparities of income, we must acknowledge this difference within the context of the current environment and the historical conditions that created these disparities. If we go 1 step further and look at disparities of wealth, we find that in 2019, the median wealth for white families was $188,200 and the median wealth for Black families was $24,100. 10
When one considers that a major contributor of wealth is home ownership, these differences seem logical—particularly related to points 1, 2, and 4 that I've just described. These economic disparities would not be as great today if the 4 examples given here (not to mention numerous other examples) had been administered equitably. The same applies to disparities in housing, employment, and education. Systemic racism is the causative agent. Systemic racism must be neutralized if we are to obtain anything close to health equity. 11
The Centers for Disease Control and Prevention (CDC) has recently taken new steps to recognize the role of racism in health. 12 The CDC plans to use “science to investigate and better understand the intersection of racism and health, and then to take action.” 13
It is time for the entire nation to recognize the links between racism and health outcomes and examine how we can design, implement, and evaluate interventions that will permanently correct these inequities.
I applaud the joint statement by the editors of the family medicine journals to commit to the eradication of systemic racism in medicine ( J Fam Pract . 2021;70:3 -4). These are crucial times in our history, where proactive change is necessary. The leadership they have shown is important.
No one wants health disparities. So, to eliminate them, we need to know what they are and where they came from. In my presentations on health disparities to students, residents, and health care providers, I use 3 definitions of health disparities. My definitions are slightly different from those proposed in the seminal report, Unequal Treatment: Confronting Racial and Ethnic Disparities in Health Care, from the National Academy of Medicine (then Institute of Medicine).1 I like to think that my definitions elicit the information needed to guide change.
The first definition focuses on health statistics. When there are different outcomes for different demographic groups for the same disease, that is a disparity. This could be Black vs white, male vs female, or 1 zip code vs another.2 We owe ourselves an explanation for these differences if we are to be able to propose solutions.
Second, there are disparities in the provision of health care. If there are 2 individuals who present with the exact same symptoms, we need to ask ourselves why they would be treated differently. Even in systems where insurance status is the same, there are documented differences in care. A well-studied example of this is pain. In 1 such study, a meta-analysis showed that Blacks were less likely than whites to receive medication for acute pain in the emergency department (OR = 0.60 [95% CI, 0.43-0.83]).3 Other examples of differences by race include cardiac services,4 lung cancer screening,5 and stroke interventions.6
The third definition of health disparities involves differences in health-seeking behavior. This is not to blame the “victim,” but to understand the reason why the difference exists so that adequate interventions can be designed to improve outcomes. Traditionally, the concept of access referenced whether or not the patient had health insurance. But the provision of health insurance is insufficient to explain issues of access.7
Extrinsic and intrinsic factors at work. Factors related to insurance are an example of the extrinsic factors related to access. However, there are intrinsic factors related to access, most of which involve health literacy. We must ask ourselves: What are the best practices to educate patients to get the care they need? I will take this 1 step further; it is the duty of all health care professionals to improve health literacy 1 patient, 1 community at a time.
The next point that I make in my presentations on health disparities is that if you control for socioeconomic status, some of the health disparities go away. However, they rarely disappear. We measure socioeconomic status in a variety of ways: education, insurance status, income, and wealth. And as would be expected, these variables are usually correlated. We also know that these variables are not distributed equally by race. This is by design. This has been intentional. This has been, in many cases, our country’s policy. This is the result of systemic racism.
Continue to: It is necessary...
It is necessary for us to be willing to accept the toxicity of racism. This we can assess in 2 major ways. First, if we apply the Koch postulates or the Bradford Hill criteria for causation to racism, we can assess the degree to which racism is an explanation for health disparities. These principles offer methods for determining the relationship between risk and outcome.
Second, when we analyze the historical antecedents of health disparities, we find that racism is directly responsible not only for the current toxicity that Black people face today, but for the socioeconomic disparities that continue to exist. Let me give just a few examples.
- The Farm Security Administration was created in 1937 to avoid the collapse of the farming industry. As a compromise to southern legislators, a model was approved to allow local administration of support to farmers that essentially condoned the discrimination that had been occurring and would continue to occur—especially in the South.
- The National Housing Act of 1934 was created to provide stability to the banking industry at a time of national crisis. It subsidized a massive building program, and many of the units had restrictive covenants that prevented the sale to Blacks. It also codified redlining that prevented insured mortgages from being provided to Black communities.
- The Social Security Act of 1935 was created to provide benefits for the elderly and disabled. All workers were included except domestic workers and farm workers—the majority of whom were Black. This was another compromise that was made with southern congressman to get this act passed.
- The Servicemen’s Readjustment Act of 1944 (also known as the GI Bill) was passed to support veterans returning from World War II. Two major functions of the bill were to support educational opportunities for veterans and their families and to support the purchase of homes. From 1945 to 1954, the US added 13 million new homes. In 1946 and 1947, the Veterans Administration financed 40% of all single-family houses in the United States. Additionally, there were educational benefits for veterans to go to college or to learn a trade. These provisions, education, and housing were not equally available to Blacks. Columbia University professor Ira Katznelson called this act and others “affirmative action for whites.” 8
In 2019, the median income in white households was $76,057 and in Black households it was $46,073. 9 So, when we look at disparities of income, we must acknowledge this difference within the context of the current environment and the historical conditions that created these disparities. If we go 1 step further and look at disparities of wealth, we find that in 2019, the median wealth for white families was $188,200 and the median wealth for Black families was $24,100. 10
When one considers that a major contributor of wealth is home ownership, these differences seem logical—particularly related to points 1, 2, and 4 that I've just described. These economic disparities would not be as great today if the 4 examples given here (not to mention numerous other examples) had been administered equitably. The same applies to disparities in housing, employment, and education. Systemic racism is the causative agent. Systemic racism must be neutralized if we are to obtain anything close to health equity. 11
The Centers for Disease Control and Prevention (CDC) has recently taken new steps to recognize the role of racism in health. 12 The CDC plans to use “science to investigate and better understand the intersection of racism and health, and then to take action.” 13
It is time for the entire nation to recognize the links between racism and health outcomes and examine how we can design, implement, and evaluate interventions that will permanently correct these inequities.
1. Institute of Medicine. Unequal Treatment: Confronting Racial and Ethnic Disparities in Health Care; 2003. Accessed April 22, 2021. https://doi.org/10.17226/10260
2. Life Expectancy: Could where you live influence how long you live? Robert Wood Johnson Foundation. Accessed April 22, 2021. www.rwjf.org/en/library/interactives/whereyouliveaffectshowlongyoulive.html
3. Lee P, Le Saux M, Siegel R, et al. Racial and ethnic disparities in the management of acute pain in US emergency departments: meta-analysis and systematic review. Am J Emerg Med. 2019;37:1770-1777. doi: 10.1016/j.ajem.2019.06.014
4. Youmans QR, Hastings-Spaine L, Princewill O, et al. Disparities in cardiovascular care: past, present, and solutions. Cleve Clin J Med. 2019;86:621-632. doi: 10.3949/ccjm.86a.18088
5. Rivera MP, Katki HA, Tanner NT, et al. Addressing disparities in lung cancer screening eligibility and healthcare access. An official American Thoracic Society statement. Am J Respir Crit Care Med. 2020;202: e95-e112. doi: 10.1164/rccm.202008-3053ST
6. Rinaldo L, Rabinstein AA, Cloft H, et al. Racial and ethnic disparities in the utilization of thrombectomy for acute stroke. Stroke. 2019;50:2428-2432. doi:10.1161/STROKEAHA.118.024651
7. Hall AG, Lemak CH, Steingraber H, et al. Expanding the definition of access: it isn’t just about health insurance. J Health Care Poor Underserved. 2008;19:625-638. doi: 10.1353/hpu.0.0011
8. Katznelson I. When Affirmative Action Was White: An Untold History of Racial Inequality in Twentieth Century America. W. W. Norton & Co; 2006.
9. US Census Bureau. Households by Total Money Income, Race, and Hispanic Origin of Householder: 1967 to 2019. Accessed April 26, 2021. www.census.gov/data/tables/2020/demo/income-poverty/p60-270.html
10. Robb G. Long-standing wealth gap between Black and white Americans remains substantial, Fed data for 2019 show. MarketWatch. September 29, 2020. Accessed April 26, 2021. www.marketwatch.com/story/long-standing-wealth-gap-between-blacks-and-whites-remains-substantial-new-fed-data-for-2019-show-2020-09-28
11. Jones CP. Levels of racism: a theoretic framework and a gardener’s tale. Am J Public Health. 2000;9:1212-1215. doi: 10.2105/ajph.90.8.1212
12. CDC. Health equity: Director’s commentary. April 8, 2021. Accessed April 23, 2021. www.cdc.gov/healthequity/racism-disparities/director-commentary.html
13. CDC. Health equity: CDC’s efforts. April 8, 2021. Accessed April 23, 2021. www.cdc.gov/healthequity/racism-disparities/cdc-efforts.html
1. Institute of Medicine. Unequal Treatment: Confronting Racial and Ethnic Disparities in Health Care; 2003. Accessed April 22, 2021. https://doi.org/10.17226/10260
2. Life Expectancy: Could where you live influence how long you live? Robert Wood Johnson Foundation. Accessed April 22, 2021. www.rwjf.org/en/library/interactives/whereyouliveaffectshowlongyoulive.html
3. Lee P, Le Saux M, Siegel R, et al. Racial and ethnic disparities in the management of acute pain in US emergency departments: meta-analysis and systematic review. Am J Emerg Med. 2019;37:1770-1777. doi: 10.1016/j.ajem.2019.06.014
4. Youmans QR, Hastings-Spaine L, Princewill O, et al. Disparities in cardiovascular care: past, present, and solutions. Cleve Clin J Med. 2019;86:621-632. doi: 10.3949/ccjm.86a.18088
5. Rivera MP, Katki HA, Tanner NT, et al. Addressing disparities in lung cancer screening eligibility and healthcare access. An official American Thoracic Society statement. Am J Respir Crit Care Med. 2020;202: e95-e112. doi: 10.1164/rccm.202008-3053ST
6. Rinaldo L, Rabinstein AA, Cloft H, et al. Racial and ethnic disparities in the utilization of thrombectomy for acute stroke. Stroke. 2019;50:2428-2432. doi:10.1161/STROKEAHA.118.024651
7. Hall AG, Lemak CH, Steingraber H, et al. Expanding the definition of access: it isn’t just about health insurance. J Health Care Poor Underserved. 2008;19:625-638. doi: 10.1353/hpu.0.0011
8. Katznelson I. When Affirmative Action Was White: An Untold History of Racial Inequality in Twentieth Century America. W. W. Norton & Co; 2006.
9. US Census Bureau. Households by Total Money Income, Race, and Hispanic Origin of Householder: 1967 to 2019. Accessed April 26, 2021. www.census.gov/data/tables/2020/demo/income-poverty/p60-270.html
10. Robb G. Long-standing wealth gap between Black and white Americans remains substantial, Fed data for 2019 show. MarketWatch. September 29, 2020. Accessed April 26, 2021. www.marketwatch.com/story/long-standing-wealth-gap-between-blacks-and-whites-remains-substantial-new-fed-data-for-2019-show-2020-09-28
11. Jones CP. Levels of racism: a theoretic framework and a gardener’s tale. Am J Public Health. 2000;9:1212-1215. doi: 10.2105/ajph.90.8.1212
12. CDC. Health equity: Director’s commentary. April 8, 2021. Accessed April 23, 2021. www.cdc.gov/healthequity/racism-disparities/director-commentary.html
13. CDC. Health equity: CDC’s efforts. April 8, 2021. Accessed April 23, 2021. www.cdc.gov/healthequity/racism-disparities/cdc-efforts.html
How to help runners steer clear of injury
Approximately 60 million people in the United States run for exercise at least once a calendar year, with approximately 11 million of them running > 100 days a year.1,2 Running is an affordable, convenient, and efficient form of exercise, whose benefits include a decrease in the risk of all-cause early mortality, cancer, and diabetes; an improved lipid profile; and better mental health.3
However, running is also the cause of a significant percentage of exercise-associated injuries: More than 60% of runners report overuse injury annually.4 Given the high incidence of running-related injury, an important component of primary care is accurately diagnosing and managing such injuries and counseling patients about how to prevent them.

This article reviews risk factors for running-related injury and summarizes evidence-based recommendations for prevention.
CASE
During a health maintenance examination, Clara K, a 47-year-old woman who is obese (body mass index [BMI], 34) and has bilateral knee osteoarthritis (OA), inquires about establishing a weight-loss strategy. Ms. K is interested in starting an exercise regimen involving running but is worried about provoking a flare of OA pain.
Risk factors for running injuries
Several risk factors—some modifiable, others nonmodifiable—are associated with running-related injury (TABLE 14-16). In addition, research suggests that other variables once thought to be risk factors, such as running surface and the Q-angle (described later), are not associated with running-related injury.
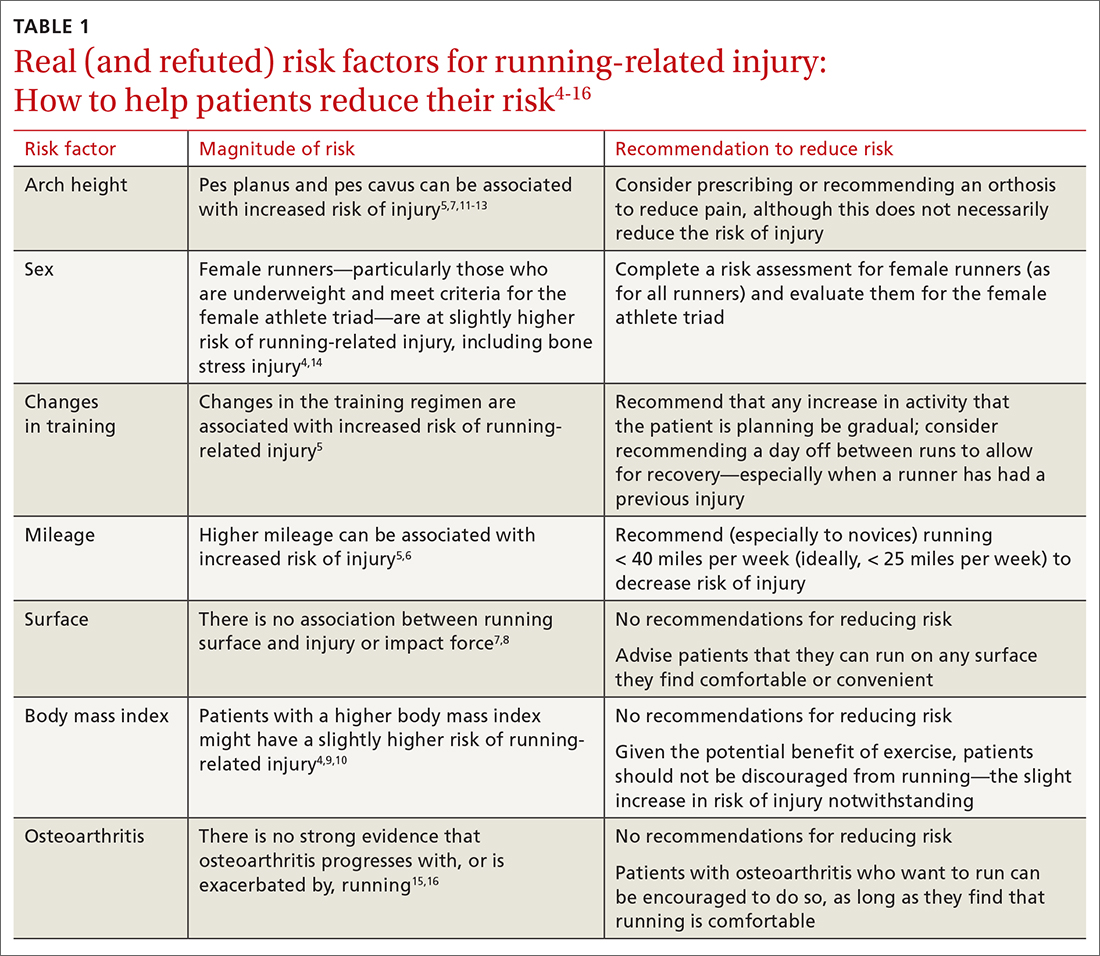
Modifiable risk factors
Changes in a training regimen or type of training. Many runners escalate training regimens as their fitness improves. Increasing mileage and changing the type of training (such as introducing hills or interval training) are independent risk factors for sustaining injury.5
The traditional recommendation has been for a runner to slowly increase or modify training with a 10% weekly increase in mileage or intensity.17 However, a randomized controlled trial failed to show a lower incidence of injury among amateur runners who adopted a graded exercise program.18 Regardless: It is still prudent to recommend a gradual increase in activity, such as taking ≥ 1 day off between running workouts or starting with a walking or jogging program, especially when there is a history of injury.19
Continue to: Excessive mileage
Excessive mileage. Many runners aspire to complete high-mileage runs. There is low-quality evidence demonstrating that high-mileage running, especially > 40 miles per week, is associated with increased risk of running-related injury.5 Injuries that occur with higher mileage are more often those of the hip and hamstring.5 A study noted that running ≤ 25 miles a week was protective against calf injury.6
Overall, there is little evidence to show that high-mileage running is associated with increased risk of running-related injury. However, this is still a risk factor that you should address with patients who have a running program—especially novices and those who ramp up mileage quickly.
Type of surface. Access to running surfaces—concrete, pavement, trails, treadmills, and athletic tracks—varies by time of day and season. Softer surfaces include treadmill, tracks, and trails; harder surfaces include asphalt and concrete.
There are limited data linking running surface with risk of injury.7 A study did not find an association between peak impact force based on running surface8; the authors hypothesized that runners compensate for a harder surface by making kinematic adjustments to minimize impact. With no strong evidence to link running-related injury to a particular running surface, patients should not be restricted to a softer running surface unless they notice a difference in comfort, because it is likely that they can compensate for a harder surface by adapting their gait.
Patients can therefore be counseled to run locally on sidewalks and neighborhood streets—if safe to do so—instead of obtaining a gym membership or driving to run on a trail. Such reassurance can increase a patient’s access to running and reduce barriers to exercise.
Continue to: BMI
BMI. Elevated BMI increases joint contact forces, which might increase risk of pain and injury.20 Results of studies investigating the link between BMI and running injury are mixed; some report that, in regard to bone stress injury, overweight BMI (> 25) is a risk factor for male runners and underweight BMI (< 18.5) is a risk factor for female runners.4,6 An observational study concluded that, among half-marathon and marathon runners, there was no significant increase in race-related injury, based on BMI.9 However, another study showed a higher rate of running-related injury in novice runners who had a higher BMI.10 A prospective cohort study found that runners with a higher BMI reported increased knee stiffness, which can place a runner at higher risk of overuse injury.4
Although these results conflict, there is consistency in the finding that obese novice runners are likely at increased risk of running-related injury; it is reasonable, therefore, for you to discuss strategies to reduce the risk of other modifiable factors, especially among obese novice runners. Patients with a higher BMI should not be discouraged from running, because exercise in combination with healthy eating habits is essential to decrease the myriad adverse health outcomes associated with obesity.
Female runners with a lower BMI, especially in the presence of other components of the female athlete triad (inadequate nutrition, amenorrhea, and low bone density), should be counseled about their increased risk of bone stress injury.21 Notably, a study of female US Navy recruits randomized to receive a trial of dietary supplementation of vitamin D plus calcium, or placebo, showed a 21% lower incidence of bone stress injury in the active-treatment group.22 To mitigate risk of injury associated with low BMI and the female athlete triad, therefore, a multidisciplinary approach of nutrition intervention, dietary optimization of vitamin D and calcium, and, possibly, activity modification should be implemented when appropriate.
Running gait. A study using 2-dimensional gait analysis to visualize biomechanical running patterns in injured and noninjured runners found that, in regard to mechanical variables, running-related injury was most strongly associated with contralateral pelvic drop.23 Gait retraining can be employed to help decrease contralateral pelvic drop.24 In addition, pelvic drop is often a result of weak gluteal muscles, and can be improved by doing strengthening exercises at home or with physical therapy.
Longer stride is also associated with running-related injury.25 A study showed improvement in patellofemoral pain by having runners increase stride rate by 10%, which reduces stride length to a significant degree.25,26 These improvements were maintained at 1-month and 3-month follow-up, and required only 1 gait retraining session.
Continue to: Get analysis is not feasible...
Gait analysis is not feasible in most primary care clinics. Instead, patients who run and (1) in whom pain persists despite more traditional treatments and (2) who have had recurring injury should be referred to a gait lab for analysis, usually by a physical therapist.
Nonmodifiable risk factors
Arch height. A high arch (pes cavus) is associated with increased risk of running-related injury, including bone stress injury, Achilles tendinopathy, plantar fasciitis, and patellofemoral pain syndrome.5 The mechanism of injury is thought to be increased forefoot loading forces.1
A review article showed that patients with pes cavus have reduced pain when using an orthosis, although there is no associated decrease in the risk of injury.5 To the contrary, a prospective study concluded that arch height was unrelated to increased risk of running-related injury.7
Evidence regarding flat feet (pes planus) and risk of injury is also mixed. Some studies show that pes planus is not associated with increased risk of injury in athletes.12 A cross-sectional study in older patients showed those with pes planus morphology had a higher rate of knee pain and wearing away of medial compartment cartilage.13 Because this study comprised only older adults, it is not generalizable to runners—nor can conclusions be drawn about causation, given the cross-sectional nature of the study.
Although a foot orthosis can correct mechanical differences caused by pes planus morphology, there is not enough evidence to conclude that correction results in a lower rate of injury. In sum, data are mixed with regard to arch height as a risk factor for running-related injury.
Continue to: Patients with...
Patients with pes planus or pes cavus should not be discouraged from running, however. If they experience pain with running, they might benefit from a trial of arch support inserts; or consider referral to an orthotist for evaluation for a custom orthosis.
Sex. Based on a prospective cohort study, female runners have a slightly higher rate of running injury than male counterparts.4 Similarly, a study showed that female military members generally had a higher incidence of stress fractures than male military members—specifically, femoral shaft and neck stress fractures.14 Runners who fall in the spectrum of the female athlete triad, as described earlier, are particularly vulnerable to bone stress injury. It is reasonable, therefore, to review risk factors for injury with female runners (as it is with all runners), especially those who have sustained a prior running-related injury.
Increased Q-angle (an obsolete risk factor). The Q-angle is approximated by drawing a line from the anterior superior iliac spine to the patella and a second line from the patella to the tibial tubercle. In males, a normal Q-angle is 14°; in females, 17° (SD = 4.5°). The Q-angle can be obtained by goniometric or radiographic measurement.
An increased Q-angle had been considered an intrinsic risk factor for running injury but has not been shown to be associated with increased risk of running-related injury or patellofemoral pain syndrome.27,28 Because the Q-angle is not a clinically relevant tool in assessing risk of injury, do not routinely measure it or include it in risk-factor counseling.
OA. Based on a systematic review of observational studies, data are inconclusive with regard to whether running contributes to, or is protective against, knee OA.15 In a large cohort study, running (1) was protective against development of hip OA and (2) decreased the risk of requiring hip replacement.29 This finding was supported by animal-model research that concluded that it is inactivity that results in thinning of articular cartilage.29 In addition, a systematic review of randomized controlled trials concluded that knee joint-loading exercises are not harmful to articular cartilage (this is low-quality evidence, however).16
Continue to: Given that there...
Given that there are no high-quality studies suggesting that running contributes to or exacerbates OA, patients with OA can be counseled to start or continue running as tolerated because the health benefit of running likely outweighs risk. Patients with pre-existing moderate-to-severe OA might report knee and hip pain that is already exacerbated by certain activities; if a high-impact activity, such as running, makes that pain worse, exercise counseling that you provide can be tailored to include lower-impact alternatives, such as swimming, cycling, or an elliptical workout.
CASE
In response to Ms. K’s interest in beginning an exercise regimen that includes running, you perform a complete routine pre-participation evaluation and appropriate cardiac screening. You discuss risk factors for running injury, focusing on modifiable risk factors.
Ms. K is perimenopausal but reports a history of regular menstrual cycles. She eats a relatively well-balanced diet. You advise that her BMI should not restrict her from incorporating running into her fitness regimen. Also, you reassure her that she should not restrict running based on a diagnosis of OA; instead, you advise her to monitor her symptoms and reconsider her program if running makes her knee pain worse.
At this point, Ms. K is ready to run. She tells you that, based on your guidance, she feels more comfortable and safe starting a running program.
Preventing injury
After reviewing risk factors for running-related injury with patients, encourage other evidence-based methods of reducing that risk.
Continue to: Shoes
Shoes
The running shoe industry offers a variety of running shoes, from minimalist shoes to cushioned stability shoes that vary based on the amount of cushioning, level of motion control, and amount of heel-to-toe drop. With so many options, new runners might wonder which shoes can reduce their risk of injury and how they should select a pair.
Stability. A characteristic of running shoes promoted by the industry is their stability: ie, their motion control. Stability shoes are marketed to runners who overpronate and therefore limit motion to prevent overpronation. The benefit of stability shoes, or stability insoles, is unclear.30 A randomized controlled trial showed that, in runners who overpronate, motion-control shoes reduced their risk of injury.31 However, another study assessed whether shoes that had been “prescribed” based on foot morphology and stride reduced the risk of injury (compared to neutral, cushioned shoes) and found no change in the incidence of soft-tissue injury.32 Given no strong evidence to suggest otherwise, runners can be advised to buy shoes based on comfort rather than on foot morphology or running stride.
Heel-to-toe drop. Another component of shoe variability is heel-to-toe drop (the height difference between heel and forefoot). A study suggests that moderate-to-high (8-12 mm) heel-to-toe drop is associated with a reduced risk of running injury.33 Barefoot running shoes, which, typically, have no heel-to-toe drop, are associated with increased risk of injury—specifically, foot stress fracture (especially in runners who are even moderately overweight).34,35
Shoe age and shoe wear can be modified to reduce injury. There is evidence that running shoes lose approximately 50% of cushioning after 300 to 500 miles of use.36 Another study found that rotating running shoes—ideally, different types or brands—can lead to fewer running-related injuries.37
In general, patients can be counseled to use shoes that feel comfortable, as long as they replace them regularly (TABLE 2). Runners can also consider alternating pairs of different running shoes between runs. Overweight runners should avoid minimally cushioned and low heel-to-toe drop running shoes.

Continue to: Cross-training
Cross-training
Cross-training exercises for runners include cycling, an elliptical workout, swimming, and weightlifting. Incorporating cross-training can be protective against running injury because cross-training requires different movement patterns, prevents overuse, and equalizes muscle imbalances that occur with running.7 In addition, replacing running with a cross-training activity can decrease weekly running time and mileage, which can further reduce risk of running-related injury.7 Runners—especially higher-mileage runners—should be encouraged to incorporate cross-training into their workout regimen to decrease their risk of injury.
Stretching. The authors of a Cochrane review concluded that there is no significant reduction in injury associated with hamstring or gastrocnemius stretching.32 A small randomized, controlled, crossover study concluded that participants subjectively felt their performance was better when warm-ups included stretching.38 This perceived improvement in performance was similar between groups who completed dynamic or static stretching. However, no difference was noted in flexibility or objective performance between groups who stretched or did not stretch before activity.
Although there is no supporting evidence that stretching reduces the risk of injury, stretching is a low-risk intervention. Because stretching might provide subjective benefit to runners, you need not discourage patients from including this activity in their running program.
CORRESPONDENCE
Kartik Sidhar, MD, 15370 Huff Way, Brookfield, WI, 53005; [email protected]
1. Brown CR Jr. Common injuries from running. In: Imboden JB, Hellerman, DB, Stone JH, eds. Current Diagnosis & Treatment: Rheumatology. 3rd ed. McGraw-Hill; 2013.
2. Lange D. Running & jogging - statistic and facts. Statista Web site. November 16, 2020. Accessed March 28, 2021. www.statista.com/topics/1743/running-and-jogging/
3. Warburton DER, Bredin SSD. Health benefits of physical activity: a systematic review of current systematic reviews. Curr Opin Cardiol. 2017;32:541-556. doi:10.1097/HCO.0000000000000437
4. Messier SP, Martin DF, Mihalko SL, et al. A 2-year prospective cohort study of overuse running injuries: The Runners and Injury Longitudinal Study (TRAILS). Am J Sports Med. 2018;46:2211-2221. doi:10.1177/0363546518773755
5. Fields KB, Sykes JC, Walker KM, et al. Prevention of running injuries. Curr Sports Med Rep. 2010;9:176-182. doi:10.1249/JSR.0b013e3181de7ec5
6. van der Worp MP, ten Haaf DSM, van Cingel R. Injuries in runners; a systematic review on risk factors and sex differences. PLoS One. 2015;10:1-18. doi:10.1371/journal.pone.0114937
7. Taunton JE, Ryan MB, Clement DB, et al. A prospective study of running injuries: the Vancouver Sun Run “In Training” clinics. Br J Sports Med. 2003;37:239-244. doi:10.1136/bjsm.37.3.239
8. Dixon SJ, Collop AC, Batt ME. Surface effects on ground reaction forces and lower extremity kinematics in running. Med Sci Sports Exerc. 2000;32:1919-1926. doi:10.1097/00005768-200011000-00016
9. Vadeboncoeur TF, Silvers SM, Taylor WC, et al. Impact of a high body mass index on lower extremity injury in marathon/half-marathon participants. J Phys Act Health. 2012;9:96-103. doi:10.1123/jpah.9.1.96
10. Buist I, Bredeweg SW. Higher risk of injury in overweight novice runners. Br J Sports Med. 2011;45:338. http://dx.doi.org/10.1136/bjsm.2011.084038.79
11. Cowan DN, Jones BH, Robinson JR. Foot morphologic characteristics and risk of Exercise-related injury. Arch Fam Med. 1993;2:773-777. doi:10.1001/archfami.2.7.773
12. Michelson JD, Durant DM, McFarland E. The injury risk associated with pes planus in athletes. Foot Ankle Int. 2002;23:629-633. doi: 10.1177/107110070202300708
13. Gross KD, Felson DT, Niu J, et al. Association of flat feet with knee pain and cartilage damage in older adults. Arthritis Care Res (Hoboken). 2011;63:937-944. doi:10.1002/acr.20431
14. Waterman BR, Gun B, Bader JO, et al. Epidemiology of lower extremity stress fractures in the United States military. Mil Med. 2016;181:1308-1313. doi:10.7205/MILMED-D-15-00571
15. Timmins KA, Leech RD, Batt ME, et al. Running and knee osteoarthritis: a systematic review and meta-analysis. Am J Sports Med. 2017;45:1447-1457. doi:10.1177/0363546516657531
16. Bricca A, Juhl CB, Steultjens M, et al. Impact of exercise on articular cartilage in people at risk of, or with established, knee osteoarthritis: a systematic review of randomised controlled trials. Br J Sports Med. 2019;53:940-947. doi:10.1136/bjsports-2017-098661
17. Johnston CAM, Taunton JE, Lloyd-Smith DR, et al. Preventing running injuries. Practical approach for family doctors. Can Fam Physician. 2003;49:1101-1109.
18. Buist I, Bredeweg SW, van Mechelen W, et al. No effect of a graded training program on the number of running-related injuries in novice runners: a randomized controlled trial. Am J Sports Med. 2008;36:33-39. doi:10.1177/0363546507307505
19. Warden SJ, Davis IS, Fredericson M. Management and prevention of bone stress injuries in long-distance runners. J Orthop Sports Phys Ther. 2014;44:749-765. doi:10.2519/jospt.2014.5334
20. Kim N, Browning RC, Lerner ZF. The effects of pediatric obesity on patellofemoral joint contact force during walking. Gait Posture. 2019;73:209-214. doi:10.1016/j.gaitpost.2019.07.307
21. Tenforde AS, Kraus E, Fredericson M. Bone stress injuries in runners. Phys Med Rehabil Clin N Am. 2016;27:139-149. doi:10.1016/j.pmr.2015.08.008
22. Lappe J, Cullen D, Haynatzki G, et al. Calcium and vitamin D supplementation decreases incidence of stress fractures in female navy recruits. J Bone Miner Res. 2008;23:741-749. doi:10.1359/jbmr.080102
23. Bramah C, Preece SJ, Gill N, et al. Is there a pathological gait associated with common soft tissue running injuries? Am J Sports Med. 2018;46:3023-3031. doi:10.1177/0363546518793657
24. Willy RW, Scholz PT, Davis IS. Mirror gait retraining for the treatment of patellofemoral pain in female runners. Clin Biomech (Bristol Avon). 2012;27:1045-1051. doi:10.1016/j.clinbiomech.2012.07.011
25. Schubert AG, Kempf J, Heiderscheit BC. Influence of stride frequency and length on running mechanics: a systematic review. Sports Health. 2014;6:210-217. doi:10.1177/1941738113508544
26. Bramah C, Preece SJ, Gill N et al. A 10% increase in step rate improves running kinematics and clinical outcomes in runners with patellofemoral pain at 4 weeks and 3 months. Am J Sports Med. 2019;47:3406-3413. doi: 10.1177/0363546519879693
27. Ramskov D, Jensen ML, Obling K, et al. No association between q-angle and foot posture with running-related injuries: a 10 week prospective follow-up study. Int J Sports Phys Ther. 2013;8:407-415.
28. Almeida GPL, Silva AP, França FJR, et al. Q-angle in patellofemoral pain: relationship with dynamic knee valgus, hip abductor torque, pain and function. Rev Bras Ortop. 2016;51:181-186. doi:10.1016/j.rboe.2016.01.010
29. Williams PT. Effects of running and walking on osteoarthritis and hip replacement risk. Med Sci Sports Exerc. 2013;45:1292-1297. doi:10.1249/MSS.0b013e3182885f26
30. Nigg BM, Baltich J, Hoerzer S, et al. Running shoes and running injuries: mythbusting and a proposal for two new paradigms: ‘Preferred movement path’ and ‘comfort filter.’ Br J Sports Med. 2015;49:1290-1294. doi:10.1136/bjsports-2015-095054
31. Malisoux L, Chambon N, Delattre N, et al. Injury risk in runners using standard or motion control shoes: a randomised controlled trial with participant and assessor blinding. Br J Sports Med. 2016;50:481-487. doi:10.1136/bjsports-2015-095031
32. Yeung SS, Yeung EW, Gillespie LD. Interventions for preventing lower limb soft-tissue running injuries. Cochrane Database Syst Rev. 2011(7):CD001256. doi:10.1002/14651858.cd001256.pub2
33. Malisoux L, Chambon N, Urhausen A, et al. Influence of the heel-to-toe drop of standard cushioned running shoes on injury risk in leisure-time runners: a randomized controlled trial with 6-month follow-up. Am J Sports Med. 2016;44:2933-2940. doi:10.1177/0363546516654690
34. Ryan M, Elashi M, Newsham-West R, et al. Examining injury risk and pain perception in runners using minimalist footwear. Br J Sports Med. 2014;48:1257-1262. doi:10.1136/bjsports-2012-092061
35. Fuller JT, Thewlis D, Buckley JD, et al.. Body mass and weekly training distance influence the pain and injuries experienced by runners using minimalist shoes: a randomized controlled trial. Am J Sports Med. 2017;45:1162-1170. doi:10.1177/0363546516682497
36. Cook SD, Kester MA, Brunet ME. Shock absorption characteristics of running shoes. Am J Sports Med. 1985;13:248-253. doi.org/10.1177/036354658501300406
37. Malisoux L, Ramesh J, Mann R, et al. Can parallel use of different running shoes decrease running-related injury risk? Scand J Med Sci Sport. 2015;25:110-115. doi:10.1111/sms.12154
38. Blazevich AJ, Gill ND, Kvorning T, et al. No effect of muscle stretching within a full, dynamic warm-up on athletic performance. Med Sci Sports Exerc. 2018;50:1258-1266. doi:10.1249/MSS.0000000000001539
Approximately 60 million people in the United States run for exercise at least once a calendar year, with approximately 11 million of them running > 100 days a year.1,2 Running is an affordable, convenient, and efficient form of exercise, whose benefits include a decrease in the risk of all-cause early mortality, cancer, and diabetes; an improved lipid profile; and better mental health.3
However, running is also the cause of a significant percentage of exercise-associated injuries: More than 60% of runners report overuse injury annually.4 Given the high incidence of running-related injury, an important component of primary care is accurately diagnosing and managing such injuries and counseling patients about how to prevent them.

This article reviews risk factors for running-related injury and summarizes evidence-based recommendations for prevention.
CASE
During a health maintenance examination, Clara K, a 47-year-old woman who is obese (body mass index [BMI], 34) and has bilateral knee osteoarthritis (OA), inquires about establishing a weight-loss strategy. Ms. K is interested in starting an exercise regimen involving running but is worried about provoking a flare of OA pain.
Risk factors for running injuries
Several risk factors—some modifiable, others nonmodifiable—are associated with running-related injury (TABLE 14-16). In addition, research suggests that other variables once thought to be risk factors, such as running surface and the Q-angle (described later), are not associated with running-related injury.

Modifiable risk factors
Changes in a training regimen or type of training. Many runners escalate training regimens as their fitness improves. Increasing mileage and changing the type of training (such as introducing hills or interval training) are independent risk factors for sustaining injury.5
The traditional recommendation has been for a runner to slowly increase or modify training with a 10% weekly increase in mileage or intensity.17 However, a randomized controlled trial failed to show a lower incidence of injury among amateur runners who adopted a graded exercise program.18 Regardless: It is still prudent to recommend a gradual increase in activity, such as taking ≥ 1 day off between running workouts or starting with a walking or jogging program, especially when there is a history of injury.19
Continue to: Excessive mileage
Excessive mileage. Many runners aspire to complete high-mileage runs. There is low-quality evidence demonstrating that high-mileage running, especially > 40 miles per week, is associated with increased risk of running-related injury.5 Injuries that occur with higher mileage are more often those of the hip and hamstring.5 A study noted that running ≤ 25 miles a week was protective against calf injury.6
Overall, there is little evidence to show that high-mileage running is associated with increased risk of running-related injury. However, this is still a risk factor that you should address with patients who have a running program—especially novices and those who ramp up mileage quickly.
Type of surface. Access to running surfaces—concrete, pavement, trails, treadmills, and athletic tracks—varies by time of day and season. Softer surfaces include treadmill, tracks, and trails; harder surfaces include asphalt and concrete.
There are limited data linking running surface with risk of injury.7 A study did not find an association between peak impact force based on running surface8; the authors hypothesized that runners compensate for a harder surface by making kinematic adjustments to minimize impact. With no strong evidence to link running-related injury to a particular running surface, patients should not be restricted to a softer running surface unless they notice a difference in comfort, because it is likely that they can compensate for a harder surface by adapting their gait.
Patients can therefore be counseled to run locally on sidewalks and neighborhood streets—if safe to do so—instead of obtaining a gym membership or driving to run on a trail. Such reassurance can increase a patient’s access to running and reduce barriers to exercise.
Continue to: BMI
BMI. Elevated BMI increases joint contact forces, which might increase risk of pain and injury.20 Results of studies investigating the link between BMI and running injury are mixed; some report that, in regard to bone stress injury, overweight BMI (> 25) is a risk factor for male runners and underweight BMI (< 18.5) is a risk factor for female runners.4,6 An observational study concluded that, among half-marathon and marathon runners, there was no significant increase in race-related injury, based on BMI.9 However, another study showed a higher rate of running-related injury in novice runners who had a higher BMI.10 A prospective cohort study found that runners with a higher BMI reported increased knee stiffness, which can place a runner at higher risk of overuse injury.4
Although these results conflict, there is consistency in the finding that obese novice runners are likely at increased risk of running-related injury; it is reasonable, therefore, for you to discuss strategies to reduce the risk of other modifiable factors, especially among obese novice runners. Patients with a higher BMI should not be discouraged from running, because exercise in combination with healthy eating habits is essential to decrease the myriad adverse health outcomes associated with obesity.
Female runners with a lower BMI, especially in the presence of other components of the female athlete triad (inadequate nutrition, amenorrhea, and low bone density), should be counseled about their increased risk of bone stress injury.21 Notably, a study of female US Navy recruits randomized to receive a trial of dietary supplementation of vitamin D plus calcium, or placebo, showed a 21% lower incidence of bone stress injury in the active-treatment group.22 To mitigate risk of injury associated with low BMI and the female athlete triad, therefore, a multidisciplinary approach of nutrition intervention, dietary optimization of vitamin D and calcium, and, possibly, activity modification should be implemented when appropriate.
Running gait. A study using 2-dimensional gait analysis to visualize biomechanical running patterns in injured and noninjured runners found that, in regard to mechanical variables, running-related injury was most strongly associated with contralateral pelvic drop.23 Gait retraining can be employed to help decrease contralateral pelvic drop.24 In addition, pelvic drop is often a result of weak gluteal muscles, and can be improved by doing strengthening exercises at home or with physical therapy.
Longer stride is also associated with running-related injury.25 A study showed improvement in patellofemoral pain by having runners increase stride rate by 10%, which reduces stride length to a significant degree.25,26 These improvements were maintained at 1-month and 3-month follow-up, and required only 1 gait retraining session.
Continue to: Get analysis is not feasible...
Gait analysis is not feasible in most primary care clinics. Instead, patients who run and (1) in whom pain persists despite more traditional treatments and (2) who have had recurring injury should be referred to a gait lab for analysis, usually by a physical therapist.
Nonmodifiable risk factors
Arch height. A high arch (pes cavus) is associated with increased risk of running-related injury, including bone stress injury, Achilles tendinopathy, plantar fasciitis, and patellofemoral pain syndrome.5 The mechanism of injury is thought to be increased forefoot loading forces.1
A review article showed that patients with pes cavus have reduced pain when using an orthosis, although there is no associated decrease in the risk of injury.5 To the contrary, a prospective study concluded that arch height was unrelated to increased risk of running-related injury.7
Evidence regarding flat feet (pes planus) and risk of injury is also mixed. Some studies show that pes planus is not associated with increased risk of injury in athletes.12 A cross-sectional study in older patients showed those with pes planus morphology had a higher rate of knee pain and wearing away of medial compartment cartilage.13 Because this study comprised only older adults, it is not generalizable to runners—nor can conclusions be drawn about causation, given the cross-sectional nature of the study.
Although a foot orthosis can correct mechanical differences caused by pes planus morphology, there is not enough evidence to conclude that correction results in a lower rate of injury. In sum, data are mixed with regard to arch height as a risk factor for running-related injury.
Continue to: Patients with...
Patients with pes planus or pes cavus should not be discouraged from running, however. If they experience pain with running, they might benefit from a trial of arch support inserts; or consider referral to an orthotist for evaluation for a custom orthosis.
Sex. Based on a prospective cohort study, female runners have a slightly higher rate of running injury than male counterparts.4 Similarly, a study showed that female military members generally had a higher incidence of stress fractures than male military members—specifically, femoral shaft and neck stress fractures.14 Runners who fall in the spectrum of the female athlete triad, as described earlier, are particularly vulnerable to bone stress injury. It is reasonable, therefore, to review risk factors for injury with female runners (as it is with all runners), especially those who have sustained a prior running-related injury.
Increased Q-angle (an obsolete risk factor). The Q-angle is approximated by drawing a line from the anterior superior iliac spine to the patella and a second line from the patella to the tibial tubercle. In males, a normal Q-angle is 14°; in females, 17° (SD = 4.5°). The Q-angle can be obtained by goniometric or radiographic measurement.
An increased Q-angle had been considered an intrinsic risk factor for running injury but has not been shown to be associated with increased risk of running-related injury or patellofemoral pain syndrome.27,28 Because the Q-angle is not a clinically relevant tool in assessing risk of injury, do not routinely measure it or include it in risk-factor counseling.
OA. Based on a systematic review of observational studies, data are inconclusive with regard to whether running contributes to, or is protective against, knee OA.15 In a large cohort study, running (1) was protective against development of hip OA and (2) decreased the risk of requiring hip replacement.29 This finding was supported by animal-model research that concluded that it is inactivity that results in thinning of articular cartilage.29 In addition, a systematic review of randomized controlled trials concluded that knee joint-loading exercises are not harmful to articular cartilage (this is low-quality evidence, however).16
Continue to: Given that there...
Given that there are no high-quality studies suggesting that running contributes to or exacerbates OA, patients with OA can be counseled to start or continue running as tolerated because the health benefit of running likely outweighs risk. Patients with pre-existing moderate-to-severe OA might report knee and hip pain that is already exacerbated by certain activities; if a high-impact activity, such as running, makes that pain worse, exercise counseling that you provide can be tailored to include lower-impact alternatives, such as swimming, cycling, or an elliptical workout.
CASE
In response to Ms. K’s interest in beginning an exercise regimen that includes running, you perform a complete routine pre-participation evaluation and appropriate cardiac screening. You discuss risk factors for running injury, focusing on modifiable risk factors.
Ms. K is perimenopausal but reports a history of regular menstrual cycles. She eats a relatively well-balanced diet. You advise that her BMI should not restrict her from incorporating running into her fitness regimen. Also, you reassure her that she should not restrict running based on a diagnosis of OA; instead, you advise her to monitor her symptoms and reconsider her program if running makes her knee pain worse.
At this point, Ms. K is ready to run. She tells you that, based on your guidance, she feels more comfortable and safe starting a running program.
Preventing injury
After reviewing risk factors for running-related injury with patients, encourage other evidence-based methods of reducing that risk.
Continue to: Shoes
Shoes
The running shoe industry offers a variety of running shoes, from minimalist shoes to cushioned stability shoes that vary based on the amount of cushioning, level of motion control, and amount of heel-to-toe drop. With so many options, new runners might wonder which shoes can reduce their risk of injury and how they should select a pair.
Stability. A characteristic of running shoes promoted by the industry is their stability: ie, their motion control. Stability shoes are marketed to runners who overpronate and therefore limit motion to prevent overpronation. The benefit of stability shoes, or stability insoles, is unclear.30 A randomized controlled trial showed that, in runners who overpronate, motion-control shoes reduced their risk of injury.31 However, another study assessed whether shoes that had been “prescribed” based on foot morphology and stride reduced the risk of injury (compared to neutral, cushioned shoes) and found no change in the incidence of soft-tissue injury.32 Given no strong evidence to suggest otherwise, runners can be advised to buy shoes based on comfort rather than on foot morphology or running stride.
Heel-to-toe drop. Another component of shoe variability is heel-to-toe drop (the height difference between heel and forefoot). A study suggests that moderate-to-high (8-12 mm) heel-to-toe drop is associated with a reduced risk of running injury.33 Barefoot running shoes, which, typically, have no heel-to-toe drop, are associated with increased risk of injury—specifically, foot stress fracture (especially in runners who are even moderately overweight).34,35
Shoe age and shoe wear can be modified to reduce injury. There is evidence that running shoes lose approximately 50% of cushioning after 300 to 500 miles of use.36 Another study found that rotating running shoes—ideally, different types or brands—can lead to fewer running-related injuries.37
In general, patients can be counseled to use shoes that feel comfortable, as long as they replace them regularly (TABLE 2). Runners can also consider alternating pairs of different running shoes between runs. Overweight runners should avoid minimally cushioned and low heel-to-toe drop running shoes.

Continue to: Cross-training
Cross-training
Cross-training exercises for runners include cycling, an elliptical workout, swimming, and weightlifting. Incorporating cross-training can be protective against running injury because cross-training requires different movement patterns, prevents overuse, and equalizes muscle imbalances that occur with running.7 In addition, replacing running with a cross-training activity can decrease weekly running time and mileage, which can further reduce risk of running-related injury.7 Runners—especially higher-mileage runners—should be encouraged to incorporate cross-training into their workout regimen to decrease their risk of injury.
Stretching. The authors of a Cochrane review concluded that there is no significant reduction in injury associated with hamstring or gastrocnemius stretching.32 A small randomized, controlled, crossover study concluded that participants subjectively felt their performance was better when warm-ups included stretching.38 This perceived improvement in performance was similar between groups who completed dynamic or static stretching. However, no difference was noted in flexibility or objective performance between groups who stretched or did not stretch before activity.
Although there is no supporting evidence that stretching reduces the risk of injury, stretching is a low-risk intervention. Because stretching might provide subjective benefit to runners, you need not discourage patients from including this activity in their running program.
CORRESPONDENCE
Kartik Sidhar, MD, 15370 Huff Way, Brookfield, WI, 53005; [email protected]
Approximately 60 million people in the United States run for exercise at least once a calendar year, with approximately 11 million of them running > 100 days a year.1,2 Running is an affordable, convenient, and efficient form of exercise, whose benefits include a decrease in the risk of all-cause early mortality, cancer, and diabetes; an improved lipid profile; and better mental health.3
However, running is also the cause of a significant percentage of exercise-associated injuries: More than 60% of runners report overuse injury annually.4 Given the high incidence of running-related injury, an important component of primary care is accurately diagnosing and managing such injuries and counseling patients about how to prevent them.

This article reviews risk factors for running-related injury and summarizes evidence-based recommendations for prevention.
CASE
During a health maintenance examination, Clara K, a 47-year-old woman who is obese (body mass index [BMI], 34) and has bilateral knee osteoarthritis (OA), inquires about establishing a weight-loss strategy. Ms. K is interested in starting an exercise regimen involving running but is worried about provoking a flare of OA pain.
Risk factors for running injuries
Several risk factors—some modifiable, others nonmodifiable—are associated with running-related injury (TABLE 14-16). In addition, research suggests that other variables once thought to be risk factors, such as running surface and the Q-angle (described later), are not associated with running-related injury.

Modifiable risk factors
Changes in a training regimen or type of training. Many runners escalate training regimens as their fitness improves. Increasing mileage and changing the type of training (such as introducing hills or interval training) are independent risk factors for sustaining injury.5
The traditional recommendation has been for a runner to slowly increase or modify training with a 10% weekly increase in mileage or intensity.17 However, a randomized controlled trial failed to show a lower incidence of injury among amateur runners who adopted a graded exercise program.18 Regardless: It is still prudent to recommend a gradual increase in activity, such as taking ≥ 1 day off between running workouts or starting with a walking or jogging program, especially when there is a history of injury.19
Continue to: Excessive mileage
Excessive mileage. Many runners aspire to complete high-mileage runs. There is low-quality evidence demonstrating that high-mileage running, especially > 40 miles per week, is associated with increased risk of running-related injury.5 Injuries that occur with higher mileage are more often those of the hip and hamstring.5 A study noted that running ≤ 25 miles a week was protective against calf injury.6
Overall, there is little evidence to show that high-mileage running is associated with increased risk of running-related injury. However, this is still a risk factor that you should address with patients who have a running program—especially novices and those who ramp up mileage quickly.
Type of surface. Access to running surfaces—concrete, pavement, trails, treadmills, and athletic tracks—varies by time of day and season. Softer surfaces include treadmill, tracks, and trails; harder surfaces include asphalt and concrete.
There are limited data linking running surface with risk of injury.7 A study did not find an association between peak impact force based on running surface8; the authors hypothesized that runners compensate for a harder surface by making kinematic adjustments to minimize impact. With no strong evidence to link running-related injury to a particular running surface, patients should not be restricted to a softer running surface unless they notice a difference in comfort, because it is likely that they can compensate for a harder surface by adapting their gait.
Patients can therefore be counseled to run locally on sidewalks and neighborhood streets—if safe to do so—instead of obtaining a gym membership or driving to run on a trail. Such reassurance can increase a patient’s access to running and reduce barriers to exercise.
Continue to: BMI
BMI. Elevated BMI increases joint contact forces, which might increase risk of pain and injury.20 Results of studies investigating the link between BMI and running injury are mixed; some report that, in regard to bone stress injury, overweight BMI (> 25) is a risk factor for male runners and underweight BMI (< 18.5) is a risk factor for female runners.4,6 An observational study concluded that, among half-marathon and marathon runners, there was no significant increase in race-related injury, based on BMI.9 However, another study showed a higher rate of running-related injury in novice runners who had a higher BMI.10 A prospective cohort study found that runners with a higher BMI reported increased knee stiffness, which can place a runner at higher risk of overuse injury.4
Although these results conflict, there is consistency in the finding that obese novice runners are likely at increased risk of running-related injury; it is reasonable, therefore, for you to discuss strategies to reduce the risk of other modifiable factors, especially among obese novice runners. Patients with a higher BMI should not be discouraged from running, because exercise in combination with healthy eating habits is essential to decrease the myriad adverse health outcomes associated with obesity.
Female runners with a lower BMI, especially in the presence of other components of the female athlete triad (inadequate nutrition, amenorrhea, and low bone density), should be counseled about their increased risk of bone stress injury.21 Notably, a study of female US Navy recruits randomized to receive a trial of dietary supplementation of vitamin D plus calcium, or placebo, showed a 21% lower incidence of bone stress injury in the active-treatment group.22 To mitigate risk of injury associated with low BMI and the female athlete triad, therefore, a multidisciplinary approach of nutrition intervention, dietary optimization of vitamin D and calcium, and, possibly, activity modification should be implemented when appropriate.
Running gait. A study using 2-dimensional gait analysis to visualize biomechanical running patterns in injured and noninjured runners found that, in regard to mechanical variables, running-related injury was most strongly associated with contralateral pelvic drop.23 Gait retraining can be employed to help decrease contralateral pelvic drop.24 In addition, pelvic drop is often a result of weak gluteal muscles, and can be improved by doing strengthening exercises at home or with physical therapy.
Longer stride is also associated with running-related injury.25 A study showed improvement in patellofemoral pain by having runners increase stride rate by 10%, which reduces stride length to a significant degree.25,26 These improvements were maintained at 1-month and 3-month follow-up, and required only 1 gait retraining session.
Continue to: Get analysis is not feasible...
Gait analysis is not feasible in most primary care clinics. Instead, patients who run and (1) in whom pain persists despite more traditional treatments and (2) who have had recurring injury should be referred to a gait lab for analysis, usually by a physical therapist.
Nonmodifiable risk factors
Arch height. A high arch (pes cavus) is associated with increased risk of running-related injury, including bone stress injury, Achilles tendinopathy, plantar fasciitis, and patellofemoral pain syndrome.5 The mechanism of injury is thought to be increased forefoot loading forces.1
A review article showed that patients with pes cavus have reduced pain when using an orthosis, although there is no associated decrease in the risk of injury.5 To the contrary, a prospective study concluded that arch height was unrelated to increased risk of running-related injury.7
Evidence regarding flat feet (pes planus) and risk of injury is also mixed. Some studies show that pes planus is not associated with increased risk of injury in athletes.12 A cross-sectional study in older patients showed those with pes planus morphology had a higher rate of knee pain and wearing away of medial compartment cartilage.13 Because this study comprised only older adults, it is not generalizable to runners—nor can conclusions be drawn about causation, given the cross-sectional nature of the study.
Although a foot orthosis can correct mechanical differences caused by pes planus morphology, there is not enough evidence to conclude that correction results in a lower rate of injury. In sum, data are mixed with regard to arch height as a risk factor for running-related injury.
Continue to: Patients with...
Patients with pes planus or pes cavus should not be discouraged from running, however. If they experience pain with running, they might benefit from a trial of arch support inserts; or consider referral to an orthotist for evaluation for a custom orthosis.
Sex. Based on a prospective cohort study, female runners have a slightly higher rate of running injury than male counterparts.4 Similarly, a study showed that female military members generally had a higher incidence of stress fractures than male military members—specifically, femoral shaft and neck stress fractures.14 Runners who fall in the spectrum of the female athlete triad, as described earlier, are particularly vulnerable to bone stress injury. It is reasonable, therefore, to review risk factors for injury with female runners (as it is with all runners), especially those who have sustained a prior running-related injury.
Increased Q-angle (an obsolete risk factor). The Q-angle is approximated by drawing a line from the anterior superior iliac spine to the patella and a second line from the patella to the tibial tubercle. In males, a normal Q-angle is 14°; in females, 17° (SD = 4.5°). The Q-angle can be obtained by goniometric or radiographic measurement.
An increased Q-angle had been considered an intrinsic risk factor for running injury but has not been shown to be associated with increased risk of running-related injury or patellofemoral pain syndrome.27,28 Because the Q-angle is not a clinically relevant tool in assessing risk of injury, do not routinely measure it or include it in risk-factor counseling.
OA. Based on a systematic review of observational studies, data are inconclusive with regard to whether running contributes to, or is protective against, knee OA.15 In a large cohort study, running (1) was protective against development of hip OA and (2) decreased the risk of requiring hip replacement.29 This finding was supported by animal-model research that concluded that it is inactivity that results in thinning of articular cartilage.29 In addition, a systematic review of randomized controlled trials concluded that knee joint-loading exercises are not harmful to articular cartilage (this is low-quality evidence, however).16
Continue to: Given that there...
Given that there are no high-quality studies suggesting that running contributes to or exacerbates OA, patients with OA can be counseled to start or continue running as tolerated because the health benefit of running likely outweighs risk. Patients with pre-existing moderate-to-severe OA might report knee and hip pain that is already exacerbated by certain activities; if a high-impact activity, such as running, makes that pain worse, exercise counseling that you provide can be tailored to include lower-impact alternatives, such as swimming, cycling, or an elliptical workout.
CASE
In response to Ms. K’s interest in beginning an exercise regimen that includes running, you perform a complete routine pre-participation evaluation and appropriate cardiac screening. You discuss risk factors for running injury, focusing on modifiable risk factors.
Ms. K is perimenopausal but reports a history of regular menstrual cycles. She eats a relatively well-balanced diet. You advise that her BMI should not restrict her from incorporating running into her fitness regimen. Also, you reassure her that she should not restrict running based on a diagnosis of OA; instead, you advise her to monitor her symptoms and reconsider her program if running makes her knee pain worse.
At this point, Ms. K is ready to run. She tells you that, based on your guidance, she feels more comfortable and safe starting a running program.
Preventing injury
After reviewing risk factors for running-related injury with patients, encourage other evidence-based methods of reducing that risk.
Continue to: Shoes
Shoes
The running shoe industry offers a variety of running shoes, from minimalist shoes to cushioned stability shoes that vary based on the amount of cushioning, level of motion control, and amount of heel-to-toe drop. With so many options, new runners might wonder which shoes can reduce their risk of injury and how they should select a pair.
Stability. A characteristic of running shoes promoted by the industry is their stability: ie, their motion control. Stability shoes are marketed to runners who overpronate and therefore limit motion to prevent overpronation. The benefit of stability shoes, or stability insoles, is unclear.30 A randomized controlled trial showed that, in runners who overpronate, motion-control shoes reduced their risk of injury.31 However, another study assessed whether shoes that had been “prescribed” based on foot morphology and stride reduced the risk of injury (compared to neutral, cushioned shoes) and found no change in the incidence of soft-tissue injury.32 Given no strong evidence to suggest otherwise, runners can be advised to buy shoes based on comfort rather than on foot morphology or running stride.
Heel-to-toe drop. Another component of shoe variability is heel-to-toe drop (the height difference between heel and forefoot). A study suggests that moderate-to-high (8-12 mm) heel-to-toe drop is associated with a reduced risk of running injury.33 Barefoot running shoes, which, typically, have no heel-to-toe drop, are associated with increased risk of injury—specifically, foot stress fracture (especially in runners who are even moderately overweight).34,35
Shoe age and shoe wear can be modified to reduce injury. There is evidence that running shoes lose approximately 50% of cushioning after 300 to 500 miles of use.36 Another study found that rotating running shoes—ideally, different types or brands—can lead to fewer running-related injuries.37
In general, patients can be counseled to use shoes that feel comfortable, as long as they replace them regularly (TABLE 2). Runners can also consider alternating pairs of different running shoes between runs. Overweight runners should avoid minimally cushioned and low heel-to-toe drop running shoes.

Continue to: Cross-training
Cross-training
Cross-training exercises for runners include cycling, an elliptical workout, swimming, and weightlifting. Incorporating cross-training can be protective against running injury because cross-training requires different movement patterns, prevents overuse, and equalizes muscle imbalances that occur with running.7 In addition, replacing running with a cross-training activity can decrease weekly running time and mileage, which can further reduce risk of running-related injury.7 Runners—especially higher-mileage runners—should be encouraged to incorporate cross-training into their workout regimen to decrease their risk of injury.
Stretching. The authors of a Cochrane review concluded that there is no significant reduction in injury associated with hamstring or gastrocnemius stretching.32 A small randomized, controlled, crossover study concluded that participants subjectively felt their performance was better when warm-ups included stretching.38 This perceived improvement in performance was similar between groups who completed dynamic or static stretching. However, no difference was noted in flexibility or objective performance between groups who stretched or did not stretch before activity.
Although there is no supporting evidence that stretching reduces the risk of injury, stretching is a low-risk intervention. Because stretching might provide subjective benefit to runners, you need not discourage patients from including this activity in their running program.
CORRESPONDENCE
Kartik Sidhar, MD, 15370 Huff Way, Brookfield, WI, 53005; [email protected]
1. Brown CR Jr. Common injuries from running. In: Imboden JB, Hellerman, DB, Stone JH, eds. Current Diagnosis & Treatment: Rheumatology. 3rd ed. McGraw-Hill; 2013.
2. Lange D. Running & jogging - statistic and facts. Statista Web site. November 16, 2020. Accessed March 28, 2021. www.statista.com/topics/1743/running-and-jogging/
3. Warburton DER, Bredin SSD. Health benefits of physical activity: a systematic review of current systematic reviews. Curr Opin Cardiol. 2017;32:541-556. doi:10.1097/HCO.0000000000000437
4. Messier SP, Martin DF, Mihalko SL, et al. A 2-year prospective cohort study of overuse running injuries: The Runners and Injury Longitudinal Study (TRAILS). Am J Sports Med. 2018;46:2211-2221. doi:10.1177/0363546518773755
5. Fields KB, Sykes JC, Walker KM, et al. Prevention of running injuries. Curr Sports Med Rep. 2010;9:176-182. doi:10.1249/JSR.0b013e3181de7ec5
6. van der Worp MP, ten Haaf DSM, van Cingel R. Injuries in runners; a systematic review on risk factors and sex differences. PLoS One. 2015;10:1-18. doi:10.1371/journal.pone.0114937
7. Taunton JE, Ryan MB, Clement DB, et al. A prospective study of running injuries: the Vancouver Sun Run “In Training” clinics. Br J Sports Med. 2003;37:239-244. doi:10.1136/bjsm.37.3.239
8. Dixon SJ, Collop AC, Batt ME. Surface effects on ground reaction forces and lower extremity kinematics in running. Med Sci Sports Exerc. 2000;32:1919-1926. doi:10.1097/00005768-200011000-00016
9. Vadeboncoeur TF, Silvers SM, Taylor WC, et al. Impact of a high body mass index on lower extremity injury in marathon/half-marathon participants. J Phys Act Health. 2012;9:96-103. doi:10.1123/jpah.9.1.96
10. Buist I, Bredeweg SW. Higher risk of injury in overweight novice runners. Br J Sports Med. 2011;45:338. http://dx.doi.org/10.1136/bjsm.2011.084038.79
11. Cowan DN, Jones BH, Robinson JR. Foot morphologic characteristics and risk of Exercise-related injury. Arch Fam Med. 1993;2:773-777. doi:10.1001/archfami.2.7.773
12. Michelson JD, Durant DM, McFarland E. The injury risk associated with pes planus in athletes. Foot Ankle Int. 2002;23:629-633. doi: 10.1177/107110070202300708
13. Gross KD, Felson DT, Niu J, et al. Association of flat feet with knee pain and cartilage damage in older adults. Arthritis Care Res (Hoboken). 2011;63:937-944. doi:10.1002/acr.20431
14. Waterman BR, Gun B, Bader JO, et al. Epidemiology of lower extremity stress fractures in the United States military. Mil Med. 2016;181:1308-1313. doi:10.7205/MILMED-D-15-00571
15. Timmins KA, Leech RD, Batt ME, et al. Running and knee osteoarthritis: a systematic review and meta-analysis. Am J Sports Med. 2017;45:1447-1457. doi:10.1177/0363546516657531
16. Bricca A, Juhl CB, Steultjens M, et al. Impact of exercise on articular cartilage in people at risk of, or with established, knee osteoarthritis: a systematic review of randomised controlled trials. Br J Sports Med. 2019;53:940-947. doi:10.1136/bjsports-2017-098661
17. Johnston CAM, Taunton JE, Lloyd-Smith DR, et al. Preventing running injuries. Practical approach for family doctors. Can Fam Physician. 2003;49:1101-1109.
18. Buist I, Bredeweg SW, van Mechelen W, et al. No effect of a graded training program on the number of running-related injuries in novice runners: a randomized controlled trial. Am J Sports Med. 2008;36:33-39. doi:10.1177/0363546507307505
19. Warden SJ, Davis IS, Fredericson M. Management and prevention of bone stress injuries in long-distance runners. J Orthop Sports Phys Ther. 2014;44:749-765. doi:10.2519/jospt.2014.5334
20. Kim N, Browning RC, Lerner ZF. The effects of pediatric obesity on patellofemoral joint contact force during walking. Gait Posture. 2019;73:209-214. doi:10.1016/j.gaitpost.2019.07.307
21. Tenforde AS, Kraus E, Fredericson M. Bone stress injuries in runners. Phys Med Rehabil Clin N Am. 2016;27:139-149. doi:10.1016/j.pmr.2015.08.008
22. Lappe J, Cullen D, Haynatzki G, et al. Calcium and vitamin D supplementation decreases incidence of stress fractures in female navy recruits. J Bone Miner Res. 2008;23:741-749. doi:10.1359/jbmr.080102
23. Bramah C, Preece SJ, Gill N, et al. Is there a pathological gait associated with common soft tissue running injuries? Am J Sports Med. 2018;46:3023-3031. doi:10.1177/0363546518793657
24. Willy RW, Scholz PT, Davis IS. Mirror gait retraining for the treatment of patellofemoral pain in female runners. Clin Biomech (Bristol Avon). 2012;27:1045-1051. doi:10.1016/j.clinbiomech.2012.07.011
25. Schubert AG, Kempf J, Heiderscheit BC. Influence of stride frequency and length on running mechanics: a systematic review. Sports Health. 2014;6:210-217. doi:10.1177/1941738113508544
26. Bramah C, Preece SJ, Gill N et al. A 10% increase in step rate improves running kinematics and clinical outcomes in runners with patellofemoral pain at 4 weeks and 3 months. Am J Sports Med. 2019;47:3406-3413. doi: 10.1177/0363546519879693
27. Ramskov D, Jensen ML, Obling K, et al. No association between q-angle and foot posture with running-related injuries: a 10 week prospective follow-up study. Int J Sports Phys Ther. 2013;8:407-415.
28. Almeida GPL, Silva AP, França FJR, et al. Q-angle in patellofemoral pain: relationship with dynamic knee valgus, hip abductor torque, pain and function. Rev Bras Ortop. 2016;51:181-186. doi:10.1016/j.rboe.2016.01.010
29. Williams PT. Effects of running and walking on osteoarthritis and hip replacement risk. Med Sci Sports Exerc. 2013;45:1292-1297. doi:10.1249/MSS.0b013e3182885f26
30. Nigg BM, Baltich J, Hoerzer S, et al. Running shoes and running injuries: mythbusting and a proposal for two new paradigms: ‘Preferred movement path’ and ‘comfort filter.’ Br J Sports Med. 2015;49:1290-1294. doi:10.1136/bjsports-2015-095054
31. Malisoux L, Chambon N, Delattre N, et al. Injury risk in runners using standard or motion control shoes: a randomised controlled trial with participant and assessor blinding. Br J Sports Med. 2016;50:481-487. doi:10.1136/bjsports-2015-095031
32. Yeung SS, Yeung EW, Gillespie LD. Interventions for preventing lower limb soft-tissue running injuries. Cochrane Database Syst Rev. 2011(7):CD001256. doi:10.1002/14651858.cd001256.pub2
33. Malisoux L, Chambon N, Urhausen A, et al. Influence of the heel-to-toe drop of standard cushioned running shoes on injury risk in leisure-time runners: a randomized controlled trial with 6-month follow-up. Am J Sports Med. 2016;44:2933-2940. doi:10.1177/0363546516654690
34. Ryan M, Elashi M, Newsham-West R, et al. Examining injury risk and pain perception in runners using minimalist footwear. Br J Sports Med. 2014;48:1257-1262. doi:10.1136/bjsports-2012-092061
35. Fuller JT, Thewlis D, Buckley JD, et al.. Body mass and weekly training distance influence the pain and injuries experienced by runners using minimalist shoes: a randomized controlled trial. Am J Sports Med. 2017;45:1162-1170. doi:10.1177/0363546516682497
36. Cook SD, Kester MA, Brunet ME. Shock absorption characteristics of running shoes. Am J Sports Med. 1985;13:248-253. doi.org/10.1177/036354658501300406
37. Malisoux L, Ramesh J, Mann R, et al. Can parallel use of different running shoes decrease running-related injury risk? Scand J Med Sci Sport. 2015;25:110-115. doi:10.1111/sms.12154
38. Blazevich AJ, Gill ND, Kvorning T, et al. No effect of muscle stretching within a full, dynamic warm-up on athletic performance. Med Sci Sports Exerc. 2018;50:1258-1266. doi:10.1249/MSS.0000000000001539
1. Brown CR Jr. Common injuries from running. In: Imboden JB, Hellerman, DB, Stone JH, eds. Current Diagnosis & Treatment: Rheumatology. 3rd ed. McGraw-Hill; 2013.
2. Lange D. Running & jogging - statistic and facts. Statista Web site. November 16, 2020. Accessed March 28, 2021. www.statista.com/topics/1743/running-and-jogging/
3. Warburton DER, Bredin SSD. Health benefits of physical activity: a systematic review of current systematic reviews. Curr Opin Cardiol. 2017;32:541-556. doi:10.1097/HCO.0000000000000437
4. Messier SP, Martin DF, Mihalko SL, et al. A 2-year prospective cohort study of overuse running injuries: The Runners and Injury Longitudinal Study (TRAILS). Am J Sports Med. 2018;46:2211-2221. doi:10.1177/0363546518773755
5. Fields KB, Sykes JC, Walker KM, et al. Prevention of running injuries. Curr Sports Med Rep. 2010;9:176-182. doi:10.1249/JSR.0b013e3181de7ec5
6. van der Worp MP, ten Haaf DSM, van Cingel R. Injuries in runners; a systematic review on risk factors and sex differences. PLoS One. 2015;10:1-18. doi:10.1371/journal.pone.0114937
7. Taunton JE, Ryan MB, Clement DB, et al. A prospective study of running injuries: the Vancouver Sun Run “In Training” clinics. Br J Sports Med. 2003;37:239-244. doi:10.1136/bjsm.37.3.239
8. Dixon SJ, Collop AC, Batt ME. Surface effects on ground reaction forces and lower extremity kinematics in running. Med Sci Sports Exerc. 2000;32:1919-1926. doi:10.1097/00005768-200011000-00016
9. Vadeboncoeur TF, Silvers SM, Taylor WC, et al. Impact of a high body mass index on lower extremity injury in marathon/half-marathon participants. J Phys Act Health. 2012;9:96-103. doi:10.1123/jpah.9.1.96
10. Buist I, Bredeweg SW. Higher risk of injury in overweight novice runners. Br J Sports Med. 2011;45:338. http://dx.doi.org/10.1136/bjsm.2011.084038.79
11. Cowan DN, Jones BH, Robinson JR. Foot morphologic characteristics and risk of Exercise-related injury. Arch Fam Med. 1993;2:773-777. doi:10.1001/archfami.2.7.773
12. Michelson JD, Durant DM, McFarland E. The injury risk associated with pes planus in athletes. Foot Ankle Int. 2002;23:629-633. doi: 10.1177/107110070202300708
13. Gross KD, Felson DT, Niu J, et al. Association of flat feet with knee pain and cartilage damage in older adults. Arthritis Care Res (Hoboken). 2011;63:937-944. doi:10.1002/acr.20431
14. Waterman BR, Gun B, Bader JO, et al. Epidemiology of lower extremity stress fractures in the United States military. Mil Med. 2016;181:1308-1313. doi:10.7205/MILMED-D-15-00571
15. Timmins KA, Leech RD, Batt ME, et al. Running and knee osteoarthritis: a systematic review and meta-analysis. Am J Sports Med. 2017;45:1447-1457. doi:10.1177/0363546516657531
16. Bricca A, Juhl CB, Steultjens M, et al. Impact of exercise on articular cartilage in people at risk of, or with established, knee osteoarthritis: a systematic review of randomised controlled trials. Br J Sports Med. 2019;53:940-947. doi:10.1136/bjsports-2017-098661
17. Johnston CAM, Taunton JE, Lloyd-Smith DR, et al. Preventing running injuries. Practical approach for family doctors. Can Fam Physician. 2003;49:1101-1109.
18. Buist I, Bredeweg SW, van Mechelen W, et al. No effect of a graded training program on the number of running-related injuries in novice runners: a randomized controlled trial. Am J Sports Med. 2008;36:33-39. doi:10.1177/0363546507307505
19. Warden SJ, Davis IS, Fredericson M. Management and prevention of bone stress injuries in long-distance runners. J Orthop Sports Phys Ther. 2014;44:749-765. doi:10.2519/jospt.2014.5334
20. Kim N, Browning RC, Lerner ZF. The effects of pediatric obesity on patellofemoral joint contact force during walking. Gait Posture. 2019;73:209-214. doi:10.1016/j.gaitpost.2019.07.307
21. Tenforde AS, Kraus E, Fredericson M. Bone stress injuries in runners. Phys Med Rehabil Clin N Am. 2016;27:139-149. doi:10.1016/j.pmr.2015.08.008
22. Lappe J, Cullen D, Haynatzki G, et al. Calcium and vitamin D supplementation decreases incidence of stress fractures in female navy recruits. J Bone Miner Res. 2008;23:741-749. doi:10.1359/jbmr.080102
23. Bramah C, Preece SJ, Gill N, et al. Is there a pathological gait associated with common soft tissue running injuries? Am J Sports Med. 2018;46:3023-3031. doi:10.1177/0363546518793657
24. Willy RW, Scholz PT, Davis IS. Mirror gait retraining for the treatment of patellofemoral pain in female runners. Clin Biomech (Bristol Avon). 2012;27:1045-1051. doi:10.1016/j.clinbiomech.2012.07.011
25. Schubert AG, Kempf J, Heiderscheit BC. Influence of stride frequency and length on running mechanics: a systematic review. Sports Health. 2014;6:210-217. doi:10.1177/1941738113508544
26. Bramah C, Preece SJ, Gill N et al. A 10% increase in step rate improves running kinematics and clinical outcomes in runners with patellofemoral pain at 4 weeks and 3 months. Am J Sports Med. 2019;47:3406-3413. doi: 10.1177/0363546519879693
27. Ramskov D, Jensen ML, Obling K, et al. No association between q-angle and foot posture with running-related injuries: a 10 week prospective follow-up study. Int J Sports Phys Ther. 2013;8:407-415.
28. Almeida GPL, Silva AP, França FJR, et al. Q-angle in patellofemoral pain: relationship with dynamic knee valgus, hip abductor torque, pain and function. Rev Bras Ortop. 2016;51:181-186. doi:10.1016/j.rboe.2016.01.010
29. Williams PT. Effects of running and walking on osteoarthritis and hip replacement risk. Med Sci Sports Exerc. 2013;45:1292-1297. doi:10.1249/MSS.0b013e3182885f26
30. Nigg BM, Baltich J, Hoerzer S, et al. Running shoes and running injuries: mythbusting and a proposal for two new paradigms: ‘Preferred movement path’ and ‘comfort filter.’ Br J Sports Med. 2015;49:1290-1294. doi:10.1136/bjsports-2015-095054
31. Malisoux L, Chambon N, Delattre N, et al. Injury risk in runners using standard or motion control shoes: a randomised controlled trial with participant and assessor blinding. Br J Sports Med. 2016;50:481-487. doi:10.1136/bjsports-2015-095031
32. Yeung SS, Yeung EW, Gillespie LD. Interventions for preventing lower limb soft-tissue running injuries. Cochrane Database Syst Rev. 2011(7):CD001256. doi:10.1002/14651858.cd001256.pub2
33. Malisoux L, Chambon N, Urhausen A, et al. Influence of the heel-to-toe drop of standard cushioned running shoes on injury risk in leisure-time runners: a randomized controlled trial with 6-month follow-up. Am J Sports Med. 2016;44:2933-2940. doi:10.1177/0363546516654690
34. Ryan M, Elashi M, Newsham-West R, et al. Examining injury risk and pain perception in runners using minimalist footwear. Br J Sports Med. 2014;48:1257-1262. doi:10.1136/bjsports-2012-092061
35. Fuller JT, Thewlis D, Buckley JD, et al.. Body mass and weekly training distance influence the pain and injuries experienced by runners using minimalist shoes: a randomized controlled trial. Am J Sports Med. 2017;45:1162-1170. doi:10.1177/0363546516682497
36. Cook SD, Kester MA, Brunet ME. Shock absorption characteristics of running shoes. Am J Sports Med. 1985;13:248-253. doi.org/10.1177/036354658501300406
37. Malisoux L, Ramesh J, Mann R, et al. Can parallel use of different running shoes decrease running-related injury risk? Scand J Med Sci Sport. 2015;25:110-115. doi:10.1111/sms.12154
38. Blazevich AJ, Gill ND, Kvorning T, et al. No effect of muscle stretching within a full, dynamic warm-up on athletic performance. Med Sci Sports Exerc. 2018;50:1258-1266. doi:10.1249/MSS.0000000000001539
PRACTICE RECOMMENDATIONS
› Counsel runners to cross-train, replace shoes regularly, and use shoes with moderate-to-high (8-12 mm) heel-to-toe drop. C
› Don’t discourage running for exercise, as long as it is tolerated, in patients who have osteoarthritis. C
› Encourage moderation in running distance and intensity, especially in novice runners. C
Strength of recommendation (SOR)
A Good-quality patient-oriented evidence
B Inconsistent or limited-quality patient-oriented evidence
C Consensus, usual practice, opinion, disease-oriented evidence, case series
High teen BMI linked to stroke risk in young adulthood
High and even high-normal body mass index (BMI) were linked to increased ischemic stroke risk, regardless of whether or not individuals had diabetes.
Overweight and obese adolescent groups in the study had a roughly two- to threefold increased risk of ischemic stroke, which was apparent even before age 30 years in the study that was based on records of Israeli adolescents evaluated prior to mandatory military service.
These findings highlight the importance of treating and preventing high BMI among adolescence, study coauthor Gilad Twig, MD, MPH, PhD, said in a press release.
“Adults who survive stroke earlier in life face poor functional outcomes, which can lead to unemployment, depression and anxiety,” said Dr. Twig, associate professor in the department of military medicine in The Hebrew University in Jerusalem.
The costs of stroke prevention and care, already high, are expected to become even higher as the adolescent obesity prevalence goes up, fueling further increases in stroke rate, Dr. Twig added.
This is believed to be the first study showing that stroke risk is associated with higher BMI values in both men and women, not just men, Dr. Twig and coauthors said in their article, published May 13, 2021 in the journal Stroke. Previous studies assessing the stroke-BMI relationship in adolescents were based on records of Swedish men evaluated during military conscription at age 18.
In the present study, Dr. Twig and coauthors assessed the linkage between adolescent BMI and first stroke event in 1.9 million male and female adolescents in Israel who were evaluated 1 year prior to mandatory military service, between the years of 1985 and 2013.
They cross-referenced that information with stroke events in a national registry to which all hospitals in Israel are required to report.
The adolescents were about 17 years of age on average at the time of evaluation, 58% were male, and 84% were born in Israel. The mean age at the beginning of follow-up for stroke was about 31 years.
Over the follow-up period, investigators identified 1,088 first stroke events, including 921 ischemic and 167 hemorrhagic strokes.
A gradual increase in stroke rate was seen across BMI categories for ischemic strokes, but not so much for hemorrhagic strokes, investigators found.
Hazard ratios for first ischemic stroke event were 1.4 (95% confidence interval, 1.2-1.6) for the high-normal BMI group, 2.0 (95% CI, 1.6-2.4) for the overweight group, and 3.5 (95% CI, 2.8-4.5) for the obese group after adjusting for age and sex at beginning of follow-up, investigators reported.
When the adjusted results were stratified by presence or absence of diabetes, estimates were similar to what was seen in the overall risk model, they added.
Among those young adults who developed ischemic stroke, 43% smoked, 29% had high blood pressure, 17% had diabetes, and 32% had abnormal lipids at the time of diagnosis, the reported data showed.
The clinical and public health implications of these findings could be substantial, since strokes are associated with worse medical and socioeconomic outcomes in younger as compared with older individuals, according to Dr. Twig and coauthors.
Younger individuals with stroke have a higher risk of recurrent stroke, heart attack, long-term care, or death, they said. Moreover, about half of young-adult stroke survivors have poor functional outcomes, and their risk of unemployment and depression/anxiety is higher than in young individuals without stroke.
One limitation of the study is that follow-up BMI data were not available for all participants. As a result, the contribution of obesity to stroke risk over time could not be assessed, and the independent risk of BMI during adolescence could not be determined. In addition, the authors said the study underrepresents orthodox and ultraorthodox Jewish women, as they are not obligated to serve in the Israeli military.
The study authors had no disclosures related to the study, which was supported by a medical corps Israel Defense Forces research grant.
High and even high-normal body mass index (BMI) were linked to increased ischemic stroke risk, regardless of whether or not individuals had diabetes.
Overweight and obese adolescent groups in the study had a roughly two- to threefold increased risk of ischemic stroke, which was apparent even before age 30 years in the study that was based on records of Israeli adolescents evaluated prior to mandatory military service.
These findings highlight the importance of treating and preventing high BMI among adolescence, study coauthor Gilad Twig, MD, MPH, PhD, said in a press release.
“Adults who survive stroke earlier in life face poor functional outcomes, which can lead to unemployment, depression and anxiety,” said Dr. Twig, associate professor in the department of military medicine in The Hebrew University in Jerusalem.
The costs of stroke prevention and care, already high, are expected to become even higher as the adolescent obesity prevalence goes up, fueling further increases in stroke rate, Dr. Twig added.
This is believed to be the first study showing that stroke risk is associated with higher BMI values in both men and women, not just men, Dr. Twig and coauthors said in their article, published May 13, 2021 in the journal Stroke. Previous studies assessing the stroke-BMI relationship in adolescents were based on records of Swedish men evaluated during military conscription at age 18.
In the present study, Dr. Twig and coauthors assessed the linkage between adolescent BMI and first stroke event in 1.9 million male and female adolescents in Israel who were evaluated 1 year prior to mandatory military service, between the years of 1985 and 2013.
They cross-referenced that information with stroke events in a national registry to which all hospitals in Israel are required to report.
The adolescents were about 17 years of age on average at the time of evaluation, 58% were male, and 84% were born in Israel. The mean age at the beginning of follow-up for stroke was about 31 years.
Over the follow-up period, investigators identified 1,088 first stroke events, including 921 ischemic and 167 hemorrhagic strokes.
A gradual increase in stroke rate was seen across BMI categories for ischemic strokes, but not so much for hemorrhagic strokes, investigators found.
Hazard ratios for first ischemic stroke event were 1.4 (95% confidence interval, 1.2-1.6) for the high-normal BMI group, 2.0 (95% CI, 1.6-2.4) for the overweight group, and 3.5 (95% CI, 2.8-4.5) for the obese group after adjusting for age and sex at beginning of follow-up, investigators reported.
When the adjusted results were stratified by presence or absence of diabetes, estimates were similar to what was seen in the overall risk model, they added.
Among those young adults who developed ischemic stroke, 43% smoked, 29% had high blood pressure, 17% had diabetes, and 32% had abnormal lipids at the time of diagnosis, the reported data showed.
The clinical and public health implications of these findings could be substantial, since strokes are associated with worse medical and socioeconomic outcomes in younger as compared with older individuals, according to Dr. Twig and coauthors.
Younger individuals with stroke have a higher risk of recurrent stroke, heart attack, long-term care, or death, they said. Moreover, about half of young-adult stroke survivors have poor functional outcomes, and their risk of unemployment and depression/anxiety is higher than in young individuals without stroke.
One limitation of the study is that follow-up BMI data were not available for all participants. As a result, the contribution of obesity to stroke risk over time could not be assessed, and the independent risk of BMI during adolescence could not be determined. In addition, the authors said the study underrepresents orthodox and ultraorthodox Jewish women, as they are not obligated to serve in the Israeli military.
The study authors had no disclosures related to the study, which was supported by a medical corps Israel Defense Forces research grant.
High and even high-normal body mass index (BMI) were linked to increased ischemic stroke risk, regardless of whether or not individuals had diabetes.
Overweight and obese adolescent groups in the study had a roughly two- to threefold increased risk of ischemic stroke, which was apparent even before age 30 years in the study that was based on records of Israeli adolescents evaluated prior to mandatory military service.
These findings highlight the importance of treating and preventing high BMI among adolescence, study coauthor Gilad Twig, MD, MPH, PhD, said in a press release.
“Adults who survive stroke earlier in life face poor functional outcomes, which can lead to unemployment, depression and anxiety,” said Dr. Twig, associate professor in the department of military medicine in The Hebrew University in Jerusalem.
The costs of stroke prevention and care, already high, are expected to become even higher as the adolescent obesity prevalence goes up, fueling further increases in stroke rate, Dr. Twig added.
This is believed to be the first study showing that stroke risk is associated with higher BMI values in both men and women, not just men, Dr. Twig and coauthors said in their article, published May 13, 2021 in the journal Stroke. Previous studies assessing the stroke-BMI relationship in adolescents were based on records of Swedish men evaluated during military conscription at age 18.
In the present study, Dr. Twig and coauthors assessed the linkage between adolescent BMI and first stroke event in 1.9 million male and female adolescents in Israel who were evaluated 1 year prior to mandatory military service, between the years of 1985 and 2013.
They cross-referenced that information with stroke events in a national registry to which all hospitals in Israel are required to report.
The adolescents were about 17 years of age on average at the time of evaluation, 58% were male, and 84% were born in Israel. The mean age at the beginning of follow-up for stroke was about 31 years.
Over the follow-up period, investigators identified 1,088 first stroke events, including 921 ischemic and 167 hemorrhagic strokes.
A gradual increase in stroke rate was seen across BMI categories for ischemic strokes, but not so much for hemorrhagic strokes, investigators found.
Hazard ratios for first ischemic stroke event were 1.4 (95% confidence interval, 1.2-1.6) for the high-normal BMI group, 2.0 (95% CI, 1.6-2.4) for the overweight group, and 3.5 (95% CI, 2.8-4.5) for the obese group after adjusting for age and sex at beginning of follow-up, investigators reported.
When the adjusted results were stratified by presence or absence of diabetes, estimates were similar to what was seen in the overall risk model, they added.
Among those young adults who developed ischemic stroke, 43% smoked, 29% had high blood pressure, 17% had diabetes, and 32% had abnormal lipids at the time of diagnosis, the reported data showed.
The clinical and public health implications of these findings could be substantial, since strokes are associated with worse medical and socioeconomic outcomes in younger as compared with older individuals, according to Dr. Twig and coauthors.
Younger individuals with stroke have a higher risk of recurrent stroke, heart attack, long-term care, or death, they said. Moreover, about half of young-adult stroke survivors have poor functional outcomes, and their risk of unemployment and depression/anxiety is higher than in young individuals without stroke.
One limitation of the study is that follow-up BMI data were not available for all participants. As a result, the contribution of obesity to stroke risk over time could not be assessed, and the independent risk of BMI during adolescence could not be determined. In addition, the authors said the study underrepresents orthodox and ultraorthodox Jewish women, as they are not obligated to serve in the Israeli military.
The study authors had no disclosures related to the study, which was supported by a medical corps Israel Defense Forces research grant.
FROM STROKE
CHEST Health Policy and Advocacy Conference
In keeping with CHEST’s commitment to advocating for our patients, we recently hosted a 2-day Health Policy and Advocacy Conference. This event aimed to carry on the tradition of the annual spring meeting held by the National Association for the Medical Direction of Respiratory Care (NAMDRC), which CHEST acquired last year.
In working with my Co-Chair, Katie Sarmiento, MD, MPH, we tried to stay true to what was so valuable from meetings past: convening stakeholders to discuss issues through their particular lens. While there were differences – this year, we gathered around a virtual table – the diversity of perspectives remained intact, bridging the landscape from clinical practice, the patients and caregivers we serve, the businesses that serve the field, and the decision-makers who must be swayed to create the change we desire.
At the same time, we wanted to take the opportunity to do what CHEST does best: provide best-in-class education. We tried to shape a program that would help the entirety of CHEST membership and our partner organizations understand the key components of why and how we advocate, and we dedicated a large portion of the program to exploring our priority issues, such as oxygen access and home mechanical ventilation. Finally, we aimed to address issues that simply cannot be ignored, including health care disparities and the impact of telemedicine on how we practice.
Today, you can access videos from the conference for free through the online CHEST store at Chestnet.org via the e-Learning Library. In the next few issues of CHEST Physician, you will find reporting and deep dives on some of the key sessions covered at the conference. Ahead at CHEST 2021 in October, there will be opportunities to join in the dialogue through formal sessions and networking opportunities. With thanks to my co-chair, all the faculty, and staff who supported this event, I hope you will listen, read along, and, most importantly, consider lending your lens and perspective to this continuing dialogue.
In keeping with CHEST’s commitment to advocating for our patients, we recently hosted a 2-day Health Policy and Advocacy Conference. This event aimed to carry on the tradition of the annual spring meeting held by the National Association for the Medical Direction of Respiratory Care (NAMDRC), which CHEST acquired last year.
In working with my Co-Chair, Katie Sarmiento, MD, MPH, we tried to stay true to what was so valuable from meetings past: convening stakeholders to discuss issues through their particular lens. While there were differences – this year, we gathered around a virtual table – the diversity of perspectives remained intact, bridging the landscape from clinical practice, the patients and caregivers we serve, the businesses that serve the field, and the decision-makers who must be swayed to create the change we desire.
At the same time, we wanted to take the opportunity to do what CHEST does best: provide best-in-class education. We tried to shape a program that would help the entirety of CHEST membership and our partner organizations understand the key components of why and how we advocate, and we dedicated a large portion of the program to exploring our priority issues, such as oxygen access and home mechanical ventilation. Finally, we aimed to address issues that simply cannot be ignored, including health care disparities and the impact of telemedicine on how we practice.
Today, you can access videos from the conference for free through the online CHEST store at Chestnet.org via the e-Learning Library. In the next few issues of CHEST Physician, you will find reporting and deep dives on some of the key sessions covered at the conference. Ahead at CHEST 2021 in October, there will be opportunities to join in the dialogue through formal sessions and networking opportunities. With thanks to my co-chair, all the faculty, and staff who supported this event, I hope you will listen, read along, and, most importantly, consider lending your lens and perspective to this continuing dialogue.
In keeping with CHEST’s commitment to advocating for our patients, we recently hosted a 2-day Health Policy and Advocacy Conference. This event aimed to carry on the tradition of the annual spring meeting held by the National Association for the Medical Direction of Respiratory Care (NAMDRC), which CHEST acquired last year.
In working with my Co-Chair, Katie Sarmiento, MD, MPH, we tried to stay true to what was so valuable from meetings past: convening stakeholders to discuss issues through their particular lens. While there were differences – this year, we gathered around a virtual table – the diversity of perspectives remained intact, bridging the landscape from clinical practice, the patients and caregivers we serve, the businesses that serve the field, and the decision-makers who must be swayed to create the change we desire.
At the same time, we wanted to take the opportunity to do what CHEST does best: provide best-in-class education. We tried to shape a program that would help the entirety of CHEST membership and our partner organizations understand the key components of why and how we advocate, and we dedicated a large portion of the program to exploring our priority issues, such as oxygen access and home mechanical ventilation. Finally, we aimed to address issues that simply cannot be ignored, including health care disparities and the impact of telemedicine on how we practice.
Today, you can access videos from the conference for free through the online CHEST store at Chestnet.org via the e-Learning Library. In the next few issues of CHEST Physician, you will find reporting and deep dives on some of the key sessions covered at the conference. Ahead at CHEST 2021 in October, there will be opportunities to join in the dialogue through formal sessions and networking opportunities. With thanks to my co-chair, all the faculty, and staff who supported this event, I hope you will listen, read along, and, most importantly, consider lending your lens and perspective to this continuing dialogue.
This month in the journal CHEST®
Editor’s picks
Clinical outcomes and healthcare resource utilization associated with reslizumab treatment in adults with severe eosinophilic asthma in real-world practice. By Dr. M. Wechsler et al.
Corticosteroid therapy is associated with improved outcome in critically ill COVID-19 patients with hyperinflammatory phenotype. By Dr. H. Qiu, et al.
Quantitative emphysema on low-dose computed tomography of the chest and risk of lung cancer and airflow obstruction: An analysis of the National Lung Screening Trial.By Dr. M. Han, et al.
How I Do It: Endobronchial valves for the treatment of advanced emphysema. By Dr. D-J. Slebos, et al.
Prolonged hospitalization following acute respiratory failure. By Dr. M. Marmor, et al.
How I Do It: Assessing patients for air travel. By Dr. J. Mandel, et al.
Development and validation of algorithms to identify pulmonary arterial hypertension in administrative data. By Dr. K. Gillmeyer, et al.
Sleep apnea and insomnia: Emerging evidence for effective clinical management. By Dr. J. Ong, et al.
Shades of gray: Subsolid nodule considerations and management. By Dr. L. Azour, et al.
Editor’s picks
Editor’s picks
Clinical outcomes and healthcare resource utilization associated with reslizumab treatment in adults with severe eosinophilic asthma in real-world practice. By Dr. M. Wechsler et al.
Corticosteroid therapy is associated with improved outcome in critically ill COVID-19 patients with hyperinflammatory phenotype. By Dr. H. Qiu, et al.
Quantitative emphysema on low-dose computed tomography of the chest and risk of lung cancer and airflow obstruction: An analysis of the National Lung Screening Trial.By Dr. M. Han, et al.
How I Do It: Endobronchial valves for the treatment of advanced emphysema. By Dr. D-J. Slebos, et al.
Prolonged hospitalization following acute respiratory failure. By Dr. M. Marmor, et al.
How I Do It: Assessing patients for air travel. By Dr. J. Mandel, et al.
Development and validation of algorithms to identify pulmonary arterial hypertension in administrative data. By Dr. K. Gillmeyer, et al.
Sleep apnea and insomnia: Emerging evidence for effective clinical management. By Dr. J. Ong, et al.
Shades of gray: Subsolid nodule considerations and management. By Dr. L. Azour, et al.
Clinical outcomes and healthcare resource utilization associated with reslizumab treatment in adults with severe eosinophilic asthma in real-world practice. By Dr. M. Wechsler et al.
Corticosteroid therapy is associated with improved outcome in critically ill COVID-19 patients with hyperinflammatory phenotype. By Dr. H. Qiu, et al.
Quantitative emphysema on low-dose computed tomography of the chest and risk of lung cancer and airflow obstruction: An analysis of the National Lung Screening Trial.By Dr. M. Han, et al.
How I Do It: Endobronchial valves for the treatment of advanced emphysema. By Dr. D-J. Slebos, et al.
Prolonged hospitalization following acute respiratory failure. By Dr. M. Marmor, et al.
How I Do It: Assessing patients for air travel. By Dr. J. Mandel, et al.
Development and validation of algorithms to identify pulmonary arterial hypertension in administrative data. By Dr. K. Gillmeyer, et al.
Sleep apnea and insomnia: Emerging evidence for effective clinical management. By Dr. J. Ong, et al.
Shades of gray: Subsolid nodule considerations and management. By Dr. L. Azour, et al.
: Management of pleural infections. Appendicitis and COVID-19. Screening for PAH. Lung function testing during the pandemic
Interventional chest and diagnostic procedures
Risk stratification and management of pleural infections
Pleural infection carries a significant health care burden with an estimated mortality rate between 10% and 20% in adults. Standard of care for pleural infections has traditionally included antibiotics and tube thoracostomy, with select patients requiring a surgical intervention. The landmark MIST II trial demonstrated that combination intrapleural fibrinolytic and DNase therapy led to reduced length of stay and lower surgical referral rates compared with placebo.1 While the use of combination intrapleural therapy has become common in the management of these patients, controversies still exist regarding nuances related to the various aspects of this therapy. A recent position paper published in Lancet Respiratory Medicine2 addresses these knowledge gaps and provides recommendations to offer guidance in decision-making. The consensus statement by the authors addresses the topics of intrapleural monotherapy, dosing regimen, sequence of dosing, and cost considerations amongst other things. The authors also summarize evidence and discuss a surgery first vs. intrapleural enzyme therapy first approach based on stage of empyema and presence of surgical expertise and surgical candidacy. However, the debate between early surgical intervention vs early intrapleural enzyme therapy has not been settled yet. A large prospective randomized control trial is currently ongoing to help answer this question [https://doi.org/10.1186/ISRCTN18192121].
Meanwhile, there has been a lack of robust validated prediction methods for selecting high-risk patients at presentation with pleural infection for an early aggressive intervention. Based on previous studies, Rahman et al.3 had described the RAPID (Renal[urea], Age, fluid Purulence, Infection Source, Dietary [albumin]) score for risk stratification of these patients. Corcoran et al.4 recently conducted a prospective, observational study and validated that the RAPID risk category (Low-risk [0-2], Medium-risk [3-4], and High-risk [5-7]) can help predict mortality at 3 months. This score may prove to be a useful tool for future research directed at improving outcomes in patients with pleural infections.
Abhinav Agrawal, MD
Samaan Rafeq , MD
NetWork Members
References
1. Rahman NM, et al. N Engl J Med. 2011 Aug 11;365(6):518.
doi: 10.1056/NEJMoa1012740.
2. Chaddha U, et al. Lancet Respir Med. 2021. S2213-2600(20)30533-6. doi: 10.1016/S2213-2600(20)30533-6.
3. Rahman NM, et al. Chest. 2014 Apr;145(4):848. doi: 10.1378/chest.13-1558.
4. Corcoran JP, et al. Eur Respir J. 2020 Nov 26;56(5):2000130. doi: 10.1183/13993003.00130-2020.
Pediatric chest medicine
Appendicitis and COVID-19
During the 2020-21 year, there was an unprecedent amount of literature and studies released to the scientific and general public about the severe, acute respiratory Coronavirus 2 (SARS-CoV-2) syndrome, commonly referred to as COVID-19. The impressive focus on SARS-CoV-2 appeared appropriately featured given the public health concerns with contraction of the disease.
While it is important to understand the potential presentations, complications, and treatments in the adult population, clinicians must be aware of the impact of this disease on children. Contrary to reports early in the pandemic, SARS-CoV-2 infection can lead to serious complications in the pediatric population. One complication is a condition called multisystem inflammation syndrome in children (MIS-C) that can mimic Kawasaki disease or toxic shock syndrome. In addition to the expected common clinical presentation of respiratory symptoms and fever, gastrointestinal complaints were reported in up to 84% of the infected children. Gastrointestinal symptoms may be the only complaint in this population, typically presenting with nausea, emesis, abdominal pain, and diarrhea. The Pediatric Chest NetWork intends to highlight these gastrointestinal complaints and make clinicians aware of an appendicitis-like syndrome or even true acute appendicitis that seems to occur in association with SARS-CoV-2 infection. There is a handful of case reports and case series that discussed this phenomenon. Due to the overlap of presenting symptoms in SARS-CoV-2 infection and acute appendicitis, clinicians must astutely evaluate patients to prevent worsening complications of a missed diagnosed appendicitis.
Eric Mull, DO
NetWork Fellow-in-Training
Pulmonary physiology, function, and rehabilitation
Lung function testing during the COVID-19 pandemic
The COVID-19 pandemic poses unique challenges to caring for patients with established lung disease or new onset respiratory complaints. Although maneuvers differ across individual tests, most involve forced expiration or high ventilatory rates. They also tend to generate cough. Because the SARS-CoV-2 virus is predominantly spread via respiratory droplets, coughing, forced expiration, and high ventilatory rates will increase the risk for transmission.
Respiratory societies across the world have developed recommendations for operating a pulmonary function lab during the pandemic (Pulmonology. 2020 Aug 5;S2531-0437[20]30175-6; Ann Am Thorac Soc. 2020;17[11]:1343). In general, deferring all non-ssential testing and adjusting precautions and testing volume by local infection rates is recommended. Using proper personal protective equipment (PPE), including N95 respirators for staff, enhanced cleaning of rooms and PFT equipment (per manufacturer recommendations), and allowing time for adequate air exchange between tests are recommended practices. Screening for symptoms prior to testing is mandatory, with the recognition that for pulmonary patients, the specificity for COVID-19 will be poor. Finally, testing for SARS-CoV-2, generally within 72 hours, and using negative pressure rooms, has been encouraged by all, though there is variation by institution and resources.
It remains imperative that lung function labs provide a safe environment for patients and staff. However, delays related to deferrals and the increased turnover time required for cleaning and air circulation grow worse over time. As the pandemic persists, the mounting toll on our pulmonary patients looms large – so please, get vaccinated and use proper precautions.
Thomas Decato, MD, FCCP
Vice-Chair
Aaron Holley, MD, FCCP
NetWork Member
Pulmonary vascular disease
I screen, you screen, we all screen for ... PAH
Although rare in the general population, pulmonary arterial hypertension (PAH) occurs more frequently in connective tissue disease, congenital heart disease, HIV, portal hypertension, and in carriers of gene mutations of heritable PAH. Given the high morbidity and mortality, and improved outcomes with earlier diagnosis and treatment, guidelines recommend aggressive assessment and screening for PAH in these high-risk groups (Frost A, et al. Eur Respir J. 2019; 53:1801904).
Effective PAH screening algorithms have been developed in systemic sclerosis. The best validated screening tool is the DETECT algorithm (Coghlan JG, et al. Ann Rheum Dis. 2014;73:1340), which uses clinical, laboratory, and pulmonary function test parameters in conjunction with echocardiographic findings to recommend right heart catheterization (RHC) for PH diagnosis. Multimodal assessments are more sensitive than echocardiography alone in diagnosing PAH in systemic sclerosis (Hao Y, et al. Arthritis Res Ther. 2015;17:7) and should be developed in other at-risk cohorts.
Recently, the DELPHI-2 study prospectively screened 55 asymptomatic adult carriers of a BMPR2 mutation- the most common genetic mutation in heritable PAH- for minimum of 2 years (Montani D, et al. Eur Respir J. 2020 Dec 30;2004229. doi: 10.1183/13993003.04229-2020). Using predefined symptomatic, echocardiographic, and cardiopulmonary exercise testing criteria for referral for RHC, the incidence of PAH was 2.3% per year. This study lays the foundation for a multimodal approach to screening carriers of BMPR2 mutations and emphasizes the importance of genetic counseling for idiopathic and familial PAH patients to identify mutation carriers who stand to benefit from appropriate PAH screening.
Christopher J. Mullin, MD, MHS
Steering Committee Member
Interventional chest and diagnostic procedures
Risk stratification and management of pleural infections
Pleural infection carries a significant health care burden with an estimated mortality rate between 10% and 20% in adults. Standard of care for pleural infections has traditionally included antibiotics and tube thoracostomy, with select patients requiring a surgical intervention. The landmark MIST II trial demonstrated that combination intrapleural fibrinolytic and DNase therapy led to reduced length of stay and lower surgical referral rates compared with placebo.1 While the use of combination intrapleural therapy has become common in the management of these patients, controversies still exist regarding nuances related to the various aspects of this therapy. A recent position paper published in Lancet Respiratory Medicine2 addresses these knowledge gaps and provides recommendations to offer guidance in decision-making. The consensus statement by the authors addresses the topics of intrapleural monotherapy, dosing regimen, sequence of dosing, and cost considerations amongst other things. The authors also summarize evidence and discuss a surgery first vs. intrapleural enzyme therapy first approach based on stage of empyema and presence of surgical expertise and surgical candidacy. However, the debate between early surgical intervention vs early intrapleural enzyme therapy has not been settled yet. A large prospective randomized control trial is currently ongoing to help answer this question [https://doi.org/10.1186/ISRCTN18192121].
Meanwhile, there has been a lack of robust validated prediction methods for selecting high-risk patients at presentation with pleural infection for an early aggressive intervention. Based on previous studies, Rahman et al.3 had described the RAPID (Renal[urea], Age, fluid Purulence, Infection Source, Dietary [albumin]) score for risk stratification of these patients. Corcoran et al.4 recently conducted a prospective, observational study and validated that the RAPID risk category (Low-risk [0-2], Medium-risk [3-4], and High-risk [5-7]) can help predict mortality at 3 months. This score may prove to be a useful tool for future research directed at improving outcomes in patients with pleural infections.
Abhinav Agrawal, MD
Samaan Rafeq , MD
NetWork Members
References
1. Rahman NM, et al. N Engl J Med. 2011 Aug 11;365(6):518.
doi: 10.1056/NEJMoa1012740.
2. Chaddha U, et al. Lancet Respir Med. 2021. S2213-2600(20)30533-6. doi: 10.1016/S2213-2600(20)30533-6.
3. Rahman NM, et al. Chest. 2014 Apr;145(4):848. doi: 10.1378/chest.13-1558.
4. Corcoran JP, et al. Eur Respir J. 2020 Nov 26;56(5):2000130. doi: 10.1183/13993003.00130-2020.
Pediatric chest medicine
Appendicitis and COVID-19
During the 2020-21 year, there was an unprecedent amount of literature and studies released to the scientific and general public about the severe, acute respiratory Coronavirus 2 (SARS-CoV-2) syndrome, commonly referred to as COVID-19. The impressive focus on SARS-CoV-2 appeared appropriately featured given the public health concerns with contraction of the disease.
While it is important to understand the potential presentations, complications, and treatments in the adult population, clinicians must be aware of the impact of this disease on children. Contrary to reports early in the pandemic, SARS-CoV-2 infection can lead to serious complications in the pediatric population. One complication is a condition called multisystem inflammation syndrome in children (MIS-C) that can mimic Kawasaki disease or toxic shock syndrome. In addition to the expected common clinical presentation of respiratory symptoms and fever, gastrointestinal complaints were reported in up to 84% of the infected children. Gastrointestinal symptoms may be the only complaint in this population, typically presenting with nausea, emesis, abdominal pain, and diarrhea. The Pediatric Chest NetWork intends to highlight these gastrointestinal complaints and make clinicians aware of an appendicitis-like syndrome or even true acute appendicitis that seems to occur in association with SARS-CoV-2 infection. There is a handful of case reports and case series that discussed this phenomenon. Due to the overlap of presenting symptoms in SARS-CoV-2 infection and acute appendicitis, clinicians must astutely evaluate patients to prevent worsening complications of a missed diagnosed appendicitis.
Eric Mull, DO
NetWork Fellow-in-Training
Pulmonary physiology, function, and rehabilitation
Lung function testing during the COVID-19 pandemic
The COVID-19 pandemic poses unique challenges to caring for patients with established lung disease or new onset respiratory complaints. Although maneuvers differ across individual tests, most involve forced expiration or high ventilatory rates. They also tend to generate cough. Because the SARS-CoV-2 virus is predominantly spread via respiratory droplets, coughing, forced expiration, and high ventilatory rates will increase the risk for transmission.
Respiratory societies across the world have developed recommendations for operating a pulmonary function lab during the pandemic (Pulmonology. 2020 Aug 5;S2531-0437[20]30175-6; Ann Am Thorac Soc. 2020;17[11]:1343). In general, deferring all non-ssential testing and adjusting precautions and testing volume by local infection rates is recommended. Using proper personal protective equipment (PPE), including N95 respirators for staff, enhanced cleaning of rooms and PFT equipment (per manufacturer recommendations), and allowing time for adequate air exchange between tests are recommended practices. Screening for symptoms prior to testing is mandatory, with the recognition that for pulmonary patients, the specificity for COVID-19 will be poor. Finally, testing for SARS-CoV-2, generally within 72 hours, and using negative pressure rooms, has been encouraged by all, though there is variation by institution and resources.
It remains imperative that lung function labs provide a safe environment for patients and staff. However, delays related to deferrals and the increased turnover time required for cleaning and air circulation grow worse over time. As the pandemic persists, the mounting toll on our pulmonary patients looms large – so please, get vaccinated and use proper precautions.
Thomas Decato, MD, FCCP
Vice-Chair
Aaron Holley, MD, FCCP
NetWork Member
Pulmonary vascular disease
I screen, you screen, we all screen for ... PAH
Although rare in the general population, pulmonary arterial hypertension (PAH) occurs more frequently in connective tissue disease, congenital heart disease, HIV, portal hypertension, and in carriers of gene mutations of heritable PAH. Given the high morbidity and mortality, and improved outcomes with earlier diagnosis and treatment, guidelines recommend aggressive assessment and screening for PAH in these high-risk groups (Frost A, et al. Eur Respir J. 2019; 53:1801904).
Effective PAH screening algorithms have been developed in systemic sclerosis. The best validated screening tool is the DETECT algorithm (Coghlan JG, et al. Ann Rheum Dis. 2014;73:1340), which uses clinical, laboratory, and pulmonary function test parameters in conjunction with echocardiographic findings to recommend right heart catheterization (RHC) for PH diagnosis. Multimodal assessments are more sensitive than echocardiography alone in diagnosing PAH in systemic sclerosis (Hao Y, et al. Arthritis Res Ther. 2015;17:7) and should be developed in other at-risk cohorts.
Recently, the DELPHI-2 study prospectively screened 55 asymptomatic adult carriers of a BMPR2 mutation- the most common genetic mutation in heritable PAH- for minimum of 2 years (Montani D, et al. Eur Respir J. 2020 Dec 30;2004229. doi: 10.1183/13993003.04229-2020). Using predefined symptomatic, echocardiographic, and cardiopulmonary exercise testing criteria for referral for RHC, the incidence of PAH was 2.3% per year. This study lays the foundation for a multimodal approach to screening carriers of BMPR2 mutations and emphasizes the importance of genetic counseling for idiopathic and familial PAH patients to identify mutation carriers who stand to benefit from appropriate PAH screening.
Christopher J. Mullin, MD, MHS
Steering Committee Member
Interventional chest and diagnostic procedures
Risk stratification and management of pleural infections
Pleural infection carries a significant health care burden with an estimated mortality rate between 10% and 20% in adults. Standard of care for pleural infections has traditionally included antibiotics and tube thoracostomy, with select patients requiring a surgical intervention. The landmark MIST II trial demonstrated that combination intrapleural fibrinolytic and DNase therapy led to reduced length of stay and lower surgical referral rates compared with placebo.1 While the use of combination intrapleural therapy has become common in the management of these patients, controversies still exist regarding nuances related to the various aspects of this therapy. A recent position paper published in Lancet Respiratory Medicine2 addresses these knowledge gaps and provides recommendations to offer guidance in decision-making. The consensus statement by the authors addresses the topics of intrapleural monotherapy, dosing regimen, sequence of dosing, and cost considerations amongst other things. The authors also summarize evidence and discuss a surgery first vs. intrapleural enzyme therapy first approach based on stage of empyema and presence of surgical expertise and surgical candidacy. However, the debate between early surgical intervention vs early intrapleural enzyme therapy has not been settled yet. A large prospective randomized control trial is currently ongoing to help answer this question [https://doi.org/10.1186/ISRCTN18192121].
Meanwhile, there has been a lack of robust validated prediction methods for selecting high-risk patients at presentation with pleural infection for an early aggressive intervention. Based on previous studies, Rahman et al.3 had described the RAPID (Renal[urea], Age, fluid Purulence, Infection Source, Dietary [albumin]) score for risk stratification of these patients. Corcoran et al.4 recently conducted a prospective, observational study and validated that the RAPID risk category (Low-risk [0-2], Medium-risk [3-4], and High-risk [5-7]) can help predict mortality at 3 months. This score may prove to be a useful tool for future research directed at improving outcomes in patients with pleural infections.
Abhinav Agrawal, MD
Samaan Rafeq , MD
NetWork Members
References
1. Rahman NM, et al. N Engl J Med. 2011 Aug 11;365(6):518.
doi: 10.1056/NEJMoa1012740.
2. Chaddha U, et al. Lancet Respir Med. 2021. S2213-2600(20)30533-6. doi: 10.1016/S2213-2600(20)30533-6.
3. Rahman NM, et al. Chest. 2014 Apr;145(4):848. doi: 10.1378/chest.13-1558.
4. Corcoran JP, et al. Eur Respir J. 2020 Nov 26;56(5):2000130. doi: 10.1183/13993003.00130-2020.
Pediatric chest medicine
Appendicitis and COVID-19
During the 2020-21 year, there was an unprecedent amount of literature and studies released to the scientific and general public about the severe, acute respiratory Coronavirus 2 (SARS-CoV-2) syndrome, commonly referred to as COVID-19. The impressive focus on SARS-CoV-2 appeared appropriately featured given the public health concerns with contraction of the disease.
While it is important to understand the potential presentations, complications, and treatments in the adult population, clinicians must be aware of the impact of this disease on children. Contrary to reports early in the pandemic, SARS-CoV-2 infection can lead to serious complications in the pediatric population. One complication is a condition called multisystem inflammation syndrome in children (MIS-C) that can mimic Kawasaki disease or toxic shock syndrome. In addition to the expected common clinical presentation of respiratory symptoms and fever, gastrointestinal complaints were reported in up to 84% of the infected children. Gastrointestinal symptoms may be the only complaint in this population, typically presenting with nausea, emesis, abdominal pain, and diarrhea. The Pediatric Chest NetWork intends to highlight these gastrointestinal complaints and make clinicians aware of an appendicitis-like syndrome or even true acute appendicitis that seems to occur in association with SARS-CoV-2 infection. There is a handful of case reports and case series that discussed this phenomenon. Due to the overlap of presenting symptoms in SARS-CoV-2 infection and acute appendicitis, clinicians must astutely evaluate patients to prevent worsening complications of a missed diagnosed appendicitis.
Eric Mull, DO
NetWork Fellow-in-Training
Pulmonary physiology, function, and rehabilitation
Lung function testing during the COVID-19 pandemic
The COVID-19 pandemic poses unique challenges to caring for patients with established lung disease or new onset respiratory complaints. Although maneuvers differ across individual tests, most involve forced expiration or high ventilatory rates. They also tend to generate cough. Because the SARS-CoV-2 virus is predominantly spread via respiratory droplets, coughing, forced expiration, and high ventilatory rates will increase the risk for transmission.
Respiratory societies across the world have developed recommendations for operating a pulmonary function lab during the pandemic (Pulmonology. 2020 Aug 5;S2531-0437[20]30175-6; Ann Am Thorac Soc. 2020;17[11]:1343). In general, deferring all non-ssential testing and adjusting precautions and testing volume by local infection rates is recommended. Using proper personal protective equipment (PPE), including N95 respirators for staff, enhanced cleaning of rooms and PFT equipment (per manufacturer recommendations), and allowing time for adequate air exchange between tests are recommended practices. Screening for symptoms prior to testing is mandatory, with the recognition that for pulmonary patients, the specificity for COVID-19 will be poor. Finally, testing for SARS-CoV-2, generally within 72 hours, and using negative pressure rooms, has been encouraged by all, though there is variation by institution and resources.
It remains imperative that lung function labs provide a safe environment for patients and staff. However, delays related to deferrals and the increased turnover time required for cleaning and air circulation grow worse over time. As the pandemic persists, the mounting toll on our pulmonary patients looms large – so please, get vaccinated and use proper precautions.
Thomas Decato, MD, FCCP
Vice-Chair
Aaron Holley, MD, FCCP
NetWork Member
Pulmonary vascular disease
I screen, you screen, we all screen for ... PAH
Although rare in the general population, pulmonary arterial hypertension (PAH) occurs more frequently in connective tissue disease, congenital heart disease, HIV, portal hypertension, and in carriers of gene mutations of heritable PAH. Given the high morbidity and mortality, and improved outcomes with earlier diagnosis and treatment, guidelines recommend aggressive assessment and screening for PAH in these high-risk groups (Frost A, et al. Eur Respir J. 2019; 53:1801904).
Effective PAH screening algorithms have been developed in systemic sclerosis. The best validated screening tool is the DETECT algorithm (Coghlan JG, et al. Ann Rheum Dis. 2014;73:1340), which uses clinical, laboratory, and pulmonary function test parameters in conjunction with echocardiographic findings to recommend right heart catheterization (RHC) for PH diagnosis. Multimodal assessments are more sensitive than echocardiography alone in diagnosing PAH in systemic sclerosis (Hao Y, et al. Arthritis Res Ther. 2015;17:7) and should be developed in other at-risk cohorts.
Recently, the DELPHI-2 study prospectively screened 55 asymptomatic adult carriers of a BMPR2 mutation- the most common genetic mutation in heritable PAH- for minimum of 2 years (Montani D, et al. Eur Respir J. 2020 Dec 30;2004229. doi: 10.1183/13993003.04229-2020). Using predefined symptomatic, echocardiographic, and cardiopulmonary exercise testing criteria for referral for RHC, the incidence of PAH was 2.3% per year. This study lays the foundation for a multimodal approach to screening carriers of BMPR2 mutations and emphasizes the importance of genetic counseling for idiopathic and familial PAH patients to identify mutation carriers who stand to benefit from appropriate PAH screening.
Christopher J. Mullin, MD, MHS
Steering Committee Member
In memoriam
CHEST has been informed of the following deaths of CHEST members.
We extend our sincere condolences.
Noe Zamel, MD (2020)
Stuart Craig Lennox, MD (2018)
Teruo Hirose, MD, PhD, FCCP
Priscilla S. A Sarinas, MD, FCCP
Stephen Jenkinson, MD, FCCP (2021)
CHEST has been informed of the following deaths of CHEST members.
We extend our sincere condolences.
Noe Zamel, MD (2020)
Stuart Craig Lennox, MD (2018)
Teruo Hirose, MD, PhD, FCCP
Priscilla S. A Sarinas, MD, FCCP
Stephen Jenkinson, MD, FCCP (2021)
CHEST has been informed of the following deaths of CHEST members.
We extend our sincere condolences.
Noe Zamel, MD (2020)
Stuart Craig Lennox, MD (2018)
Teruo Hirose, MD, PhD, FCCP
Priscilla S. A Sarinas, MD, FCCP
Stephen Jenkinson, MD, FCCP (2021)
Obstructive sleep apnea and COVID-19
Severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) caused by the novel coronavirus of the year 2019 (COVID-19) has had a major impact on global health and economy. United States reported a total caseload of 28,998,834 patients and total mortality of 525,031 as of March 2021 (NPR.org; worldometer. Accessed March 8, 2021). The beginning of 2021 ushered positivity with the development of multiple highly effective SARS-CoV-2 vaccines. Although the medical world has gained much knowledge about this deadly disease, there are many unknowns and still much to be learned.
Two early landmark studies from Italy (Lombardy) and United States (New York City area) provided initial insight on comorbid conditions associated with increased risk of severe COVID-19 infection (Richardson S, et al. JAMA. 2020;323[20]:2052; Grasselli G, et al. JAMA Intern Med. 2020;180[10]:1345). In the United States cohort, hypertension (HTN), obesity, and diabetes (DM) were independent risk factors for severe disease, while in the Italy cohort, older age, male, COPD, hypercholesterolemia, and diabetes were independent risk factors for increased mortality. Obstructive sleep apnea (OSA) was not mentioned as a comorbid risk factor.
There is much speculation regarding OSA as an independent risk factor for severe COVID-19 infection. OSA is a common sleep-related breathing disorder with increased prevalence in men, older age, and higher body mass index (BMI); and OSA is associated with hypertension, obesity, and diabetes, all of which are risk factors for severe COVID-19. Because of the shared similarities in pathophysiology between OSA and COVID-19 (Tufik S, et al. J Clin Sleep Med. 2020;16[8]:1425), and shared comorbid conditions associated with increased risk of severe COVID-19 disease, OSA has been suggested as an independent risk factor for unfavorable COVID-19-related outcomes.
SARS-CoV-2 triggers a severe inflammatory response involving type-II pneumocytes and angiotensin-converting enzyme 2 pathway. OSA is characterized by intermittent hypoxia and sleep fragmentation, leading to a cascade of systemic inflammatory response involving oxidative stress, pro-inflammatory cytokines, endothelial dysfunction, and consequent cardiovascular injury (Jose RJ, et al. Lancet Respir Med. 2020;8[6]:e46; Saxena K, et al. Sleep Medicine. 2021;79:223). In this regard, OSA may contribute to COVID-19 “cytokine storm” by causing or exacerbating endothelial dysfunction, inflammation, and oxidative stress.
Multiple studies have recently been published on the impact of OSA on COVID-19 outcomes. The Coronavirus SARS-CoV-2 and Diabetes Outcomes (CORONADO) study was one of the initial studies that analyzed the relationship between OSA and COVID-19-related outcomes. This was a multicenter observational study involving diabetic patients hospitalized with COVID-19. The primary outcome was mechanical ventilation and/or death within 7 days of admission. Multivariate adjustment showed that age, BMI, and OSA, among other factors, were independently associated with risk of death on day 7 (Cariou B, et al. Diabetologia. 2020;63[8]:1500). Strausz and colleagues also evaluated OSA as an independent risk factor for severe COVID-19 in a large registry of hospital discharge patients (FinnGen study). The authors reported that although the risk of contracting COVID-19 was the same for patients with or without OSA, after adjusting for age, sex, and BMI, OSA was associated with higher risk of hospitalization (Strausz S, et al. BMJ Open Resp Res. 2021;8:e000845). Similar findings were confirmed by the Maas et al. study, which utilized a large socioeconomically diverse database composed of 10 hospital systems. Diagnoses and outcomes were identified by ICD-10 coding and medical record data. After adjustments for diabetes, HTN, and BMI, OSA conferred an eight-fold risk for COVID-19 infection, was associated with increased risk of hospitalization, and doubled the risk of developing respiratory failure (Maas MB, et al. Sleep Breath. 2020 Sep; 29:1-3. doi: 10.1007/s11325-020-02203-0).
Peker and colleagues conducted a prospective multicenter observational study comparing clinical outcomes of severe COVID-19 infection in patients with low vs high pretest probability of having OSA based on the Berlin questionnaire. The authors reported a clinically significant risk of poorer clinical outcomes in the high pretest probability OSA group after adjustments for age, sex, and comorbidities (Peker Y, et al. Ann Am Thorac Soc. 2021. Feb 17. doi: 10.1513/AnnalsATS.202011-1409OC). A timely meta-analysis including 21 studies (19 with retrospective design) with 54,276 COVID-19 patients and 4,640 OSA patients concluded poor composite outcomes including severe COVID-19, intensive care unit admission, mechanical ventilatory support, and death in association with OSA (OR – 1.72 95% CI 1.55-1.91, P< .00001). In patients with obesity, OSA is a highly prevalent co-morbid condition. BMI, however, was not adjusted in this model (Hariyanto TI, et al. Sleep Med. 2021. doi: 10.1016/j.sleep.2021.03.029).
Other studies have concluded the opposite with OSA not being an independent risk factor for severe COVID-19 infection. Cade and colleagues conducted a retrospective analysis from a comprehensive electronic health dataset using ICD codes to identify OSA patients with severe COVID-19 infection. A significant association between OSA and COVID-19 death was noted after adjustment for demographics (ethnicity, age, sex). However, when fully adjusted for demographics, BMI, asthma, COPD, HTN, or DM, OSA was not an independent risk factor for COVID-19-related mortality and hospitalization (Cade BE, et al. Am J Respir Crit Care Med. 2020;202[10]:1462). The FinnGen study (Strausz et al.) was part of a meta-analysis examining the association between OSA and severe COVID-19 with and without adjustments for BMI. This meta-analysis consisted of 15,835 COVID-19 patients including 1,294 with OSA. The authors found that OSA was a risk factor with a two-fold increased risk of severe COVID-19 infection (OR = 2.37, P = .021). However, after adjustments were made for BMI, this finding lost statistical significance (OR=1.55, P=.13) (Strausz S, et al. BMJ Open Resp Res. 2021;8:e000845).
It is worth noting that a majority of studies identified OSA by indirect and imperfect methods through chart review, ICD codes, and databases. Confirmed OSA based on formal testing with a sleep study in COVID-19 patients remains a challenge. Perhaps well performed screening questionnaires, such as STOP-Bang, Berlin, or NoSAS, can be utilized as was the case in one study. It is also unclear if outcomes of COVID-19 infection differ in patients with treated or untreated OSA, as raised by the CORONADO study. A recent cross-sectional telephone interview survey of patients with confirmed OSA in Iran alluded to higher prevalence of COVID-19 in patients with severe OSA with suggestion of lower prevalence in patients who were currently receiving OSA treatment with positive airway pressure (PAP) therapy (Najafi A, et al. Sleep Health. 2021 Feb;7[1]:14). This is a crucial question as PAP therapy is considered an aerosol-generating procedure (Lance CG. Cleve Clin J Med. 2020 May 5. doi: 10.3949/ccjm.87a.ccc003). Studies have suggested continued use of PAP therapy with additional measures to mitigate the spread of virus, since failure to use PAP could be deleterious to the patient’s quality of life. Interestingly, PAP adherence seemed to have improved during the pandemic as evidenced by a telephonic survey done in New York City that showed 88% of patients with OSA used a PAP device consistently (Attias D, et al. Eur Respir J. 2020 Jul 30;56[1]:2001607. doi: 10.1183/13993003.01607-2020).
In summary, the jury is still out on whether OSA is a facilitator for viral replication, or an independent risk factor for poor prognosis related to COVID-19 infection, or has no clinical relevance to COVID-19. COVID-19 and OSA share comorbidities and pathways leading to a systemic inflammatory cascade. Theoretically, it would make sense that OSA is a risk factor for severe COVID-19 infection; however, it remains to be proven. The recent studies are limited by retrospective and observational nature, imprecise OSA classification/diagnostic criteria, and confounded by difficult to control variables. Further research is needed to expand our understanding of OSA -induced intermittent hypoxemia, inflammation, and endothelial dysfunction that may play a role in COVID-19 morbidity and mortality. Until we have more clarity, close monitoring of OSA patients infected with COVID-19 is recommended along with implementation of safe protocols for continuation of PAP usage during the infectious phase. Identifying underlying comorbid conditions that contribute to worsening of a COVID-19 infectious course is a crucial step in improving clinical outcomes.
Dr. Sahni is Assistant Professor of Clinical Medicine, Division of Pulmonary, Critical Care, Sleep and Allergy, Department of Medicine, University of Illinois at Chicago. Dr. Cao is Clinical Associate Professor, Division of Sleep Medicine and Division of Neuromuscular Medicine, Department of Psychiatry and Department of Neurology, Stanford (Calif.) University.
Severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) caused by the novel coronavirus of the year 2019 (COVID-19) has had a major impact on global health and economy. United States reported a total caseload of 28,998,834 patients and total mortality of 525,031 as of March 2021 (NPR.org; worldometer. Accessed March 8, 2021). The beginning of 2021 ushered positivity with the development of multiple highly effective SARS-CoV-2 vaccines. Although the medical world has gained much knowledge about this deadly disease, there are many unknowns and still much to be learned.
Two early landmark studies from Italy (Lombardy) and United States (New York City area) provided initial insight on comorbid conditions associated with increased risk of severe COVID-19 infection (Richardson S, et al. JAMA. 2020;323[20]:2052; Grasselli G, et al. JAMA Intern Med. 2020;180[10]:1345). In the United States cohort, hypertension (HTN), obesity, and diabetes (DM) were independent risk factors for severe disease, while in the Italy cohort, older age, male, COPD, hypercholesterolemia, and diabetes were independent risk factors for increased mortality. Obstructive sleep apnea (OSA) was not mentioned as a comorbid risk factor.
There is much speculation regarding OSA as an independent risk factor for severe COVID-19 infection. OSA is a common sleep-related breathing disorder with increased prevalence in men, older age, and higher body mass index (BMI); and OSA is associated with hypertension, obesity, and diabetes, all of which are risk factors for severe COVID-19. Because of the shared similarities in pathophysiology between OSA and COVID-19 (Tufik S, et al. J Clin Sleep Med. 2020;16[8]:1425), and shared comorbid conditions associated with increased risk of severe COVID-19 disease, OSA has been suggested as an independent risk factor for unfavorable COVID-19-related outcomes.
SARS-CoV-2 triggers a severe inflammatory response involving type-II pneumocytes and angiotensin-converting enzyme 2 pathway. OSA is characterized by intermittent hypoxia and sleep fragmentation, leading to a cascade of systemic inflammatory response involving oxidative stress, pro-inflammatory cytokines, endothelial dysfunction, and consequent cardiovascular injury (Jose RJ, et al. Lancet Respir Med. 2020;8[6]:e46; Saxena K, et al. Sleep Medicine. 2021;79:223). In this regard, OSA may contribute to COVID-19 “cytokine storm” by causing or exacerbating endothelial dysfunction, inflammation, and oxidative stress.
Multiple studies have recently been published on the impact of OSA on COVID-19 outcomes. The Coronavirus SARS-CoV-2 and Diabetes Outcomes (CORONADO) study was one of the initial studies that analyzed the relationship between OSA and COVID-19-related outcomes. This was a multicenter observational study involving diabetic patients hospitalized with COVID-19. The primary outcome was mechanical ventilation and/or death within 7 days of admission. Multivariate adjustment showed that age, BMI, and OSA, among other factors, were independently associated with risk of death on day 7 (Cariou B, et al. Diabetologia. 2020;63[8]:1500). Strausz and colleagues also evaluated OSA as an independent risk factor for severe COVID-19 in a large registry of hospital discharge patients (FinnGen study). The authors reported that although the risk of contracting COVID-19 was the same for patients with or without OSA, after adjusting for age, sex, and BMI, OSA was associated with higher risk of hospitalization (Strausz S, et al. BMJ Open Resp Res. 2021;8:e000845). Similar findings were confirmed by the Maas et al. study, which utilized a large socioeconomically diverse database composed of 10 hospital systems. Diagnoses and outcomes were identified by ICD-10 coding and medical record data. After adjustments for diabetes, HTN, and BMI, OSA conferred an eight-fold risk for COVID-19 infection, was associated with increased risk of hospitalization, and doubled the risk of developing respiratory failure (Maas MB, et al. Sleep Breath. 2020 Sep; 29:1-3. doi: 10.1007/s11325-020-02203-0).
Peker and colleagues conducted a prospective multicenter observational study comparing clinical outcomes of severe COVID-19 infection in patients with low vs high pretest probability of having OSA based on the Berlin questionnaire. The authors reported a clinically significant risk of poorer clinical outcomes in the high pretest probability OSA group after adjustments for age, sex, and comorbidities (Peker Y, et al. Ann Am Thorac Soc. 2021. Feb 17. doi: 10.1513/AnnalsATS.202011-1409OC). A timely meta-analysis including 21 studies (19 with retrospective design) with 54,276 COVID-19 patients and 4,640 OSA patients concluded poor composite outcomes including severe COVID-19, intensive care unit admission, mechanical ventilatory support, and death in association with OSA (OR – 1.72 95% CI 1.55-1.91, P< .00001). In patients with obesity, OSA is a highly prevalent co-morbid condition. BMI, however, was not adjusted in this model (Hariyanto TI, et al. Sleep Med. 2021. doi: 10.1016/j.sleep.2021.03.029).
Other studies have concluded the opposite with OSA not being an independent risk factor for severe COVID-19 infection. Cade and colleagues conducted a retrospective analysis from a comprehensive electronic health dataset using ICD codes to identify OSA patients with severe COVID-19 infection. A significant association between OSA and COVID-19 death was noted after adjustment for demographics (ethnicity, age, sex). However, when fully adjusted for demographics, BMI, asthma, COPD, HTN, or DM, OSA was not an independent risk factor for COVID-19-related mortality and hospitalization (Cade BE, et al. Am J Respir Crit Care Med. 2020;202[10]:1462). The FinnGen study (Strausz et al.) was part of a meta-analysis examining the association between OSA and severe COVID-19 with and without adjustments for BMI. This meta-analysis consisted of 15,835 COVID-19 patients including 1,294 with OSA. The authors found that OSA was a risk factor with a two-fold increased risk of severe COVID-19 infection (OR = 2.37, P = .021). However, after adjustments were made for BMI, this finding lost statistical significance (OR=1.55, P=.13) (Strausz S, et al. BMJ Open Resp Res. 2021;8:e000845).
It is worth noting that a majority of studies identified OSA by indirect and imperfect methods through chart review, ICD codes, and databases. Confirmed OSA based on formal testing with a sleep study in COVID-19 patients remains a challenge. Perhaps well performed screening questionnaires, such as STOP-Bang, Berlin, or NoSAS, can be utilized as was the case in one study. It is also unclear if outcomes of COVID-19 infection differ in patients with treated or untreated OSA, as raised by the CORONADO study. A recent cross-sectional telephone interview survey of patients with confirmed OSA in Iran alluded to higher prevalence of COVID-19 in patients with severe OSA with suggestion of lower prevalence in patients who were currently receiving OSA treatment with positive airway pressure (PAP) therapy (Najafi A, et al. Sleep Health. 2021 Feb;7[1]:14). This is a crucial question as PAP therapy is considered an aerosol-generating procedure (Lance CG. Cleve Clin J Med. 2020 May 5. doi: 10.3949/ccjm.87a.ccc003). Studies have suggested continued use of PAP therapy with additional measures to mitigate the spread of virus, since failure to use PAP could be deleterious to the patient’s quality of life. Interestingly, PAP adherence seemed to have improved during the pandemic as evidenced by a telephonic survey done in New York City that showed 88% of patients with OSA used a PAP device consistently (Attias D, et al. Eur Respir J. 2020 Jul 30;56[1]:2001607. doi: 10.1183/13993003.01607-2020).
In summary, the jury is still out on whether OSA is a facilitator for viral replication, or an independent risk factor for poor prognosis related to COVID-19 infection, or has no clinical relevance to COVID-19. COVID-19 and OSA share comorbidities and pathways leading to a systemic inflammatory cascade. Theoretically, it would make sense that OSA is a risk factor for severe COVID-19 infection; however, it remains to be proven. The recent studies are limited by retrospective and observational nature, imprecise OSA classification/diagnostic criteria, and confounded by difficult to control variables. Further research is needed to expand our understanding of OSA -induced intermittent hypoxemia, inflammation, and endothelial dysfunction that may play a role in COVID-19 morbidity and mortality. Until we have more clarity, close monitoring of OSA patients infected with COVID-19 is recommended along with implementation of safe protocols for continuation of PAP usage during the infectious phase. Identifying underlying comorbid conditions that contribute to worsening of a COVID-19 infectious course is a crucial step in improving clinical outcomes.
Dr. Sahni is Assistant Professor of Clinical Medicine, Division of Pulmonary, Critical Care, Sleep and Allergy, Department of Medicine, University of Illinois at Chicago. Dr. Cao is Clinical Associate Professor, Division of Sleep Medicine and Division of Neuromuscular Medicine, Department of Psychiatry and Department of Neurology, Stanford (Calif.) University.
Severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) caused by the novel coronavirus of the year 2019 (COVID-19) has had a major impact on global health and economy. United States reported a total caseload of 28,998,834 patients and total mortality of 525,031 as of March 2021 (NPR.org; worldometer. Accessed March 8, 2021). The beginning of 2021 ushered positivity with the development of multiple highly effective SARS-CoV-2 vaccines. Although the medical world has gained much knowledge about this deadly disease, there are many unknowns and still much to be learned.
Two early landmark studies from Italy (Lombardy) and United States (New York City area) provided initial insight on comorbid conditions associated with increased risk of severe COVID-19 infection (Richardson S, et al. JAMA. 2020;323[20]:2052; Grasselli G, et al. JAMA Intern Med. 2020;180[10]:1345). In the United States cohort, hypertension (HTN), obesity, and diabetes (DM) were independent risk factors for severe disease, while in the Italy cohort, older age, male, COPD, hypercholesterolemia, and diabetes were independent risk factors for increased mortality. Obstructive sleep apnea (OSA) was not mentioned as a comorbid risk factor.
There is much speculation regarding OSA as an independent risk factor for severe COVID-19 infection. OSA is a common sleep-related breathing disorder with increased prevalence in men, older age, and higher body mass index (BMI); and OSA is associated with hypertension, obesity, and diabetes, all of which are risk factors for severe COVID-19. Because of the shared similarities in pathophysiology between OSA and COVID-19 (Tufik S, et al. J Clin Sleep Med. 2020;16[8]:1425), and shared comorbid conditions associated with increased risk of severe COVID-19 disease, OSA has been suggested as an independent risk factor for unfavorable COVID-19-related outcomes.
SARS-CoV-2 triggers a severe inflammatory response involving type-II pneumocytes and angiotensin-converting enzyme 2 pathway. OSA is characterized by intermittent hypoxia and sleep fragmentation, leading to a cascade of systemic inflammatory response involving oxidative stress, pro-inflammatory cytokines, endothelial dysfunction, and consequent cardiovascular injury (Jose RJ, et al. Lancet Respir Med. 2020;8[6]:e46; Saxena K, et al. Sleep Medicine. 2021;79:223). In this regard, OSA may contribute to COVID-19 “cytokine storm” by causing or exacerbating endothelial dysfunction, inflammation, and oxidative stress.
Multiple studies have recently been published on the impact of OSA on COVID-19 outcomes. The Coronavirus SARS-CoV-2 and Diabetes Outcomes (CORONADO) study was one of the initial studies that analyzed the relationship between OSA and COVID-19-related outcomes. This was a multicenter observational study involving diabetic patients hospitalized with COVID-19. The primary outcome was mechanical ventilation and/or death within 7 days of admission. Multivariate adjustment showed that age, BMI, and OSA, among other factors, were independently associated with risk of death on day 7 (Cariou B, et al. Diabetologia. 2020;63[8]:1500). Strausz and colleagues also evaluated OSA as an independent risk factor for severe COVID-19 in a large registry of hospital discharge patients (FinnGen study). The authors reported that although the risk of contracting COVID-19 was the same for patients with or without OSA, after adjusting for age, sex, and BMI, OSA was associated with higher risk of hospitalization (Strausz S, et al. BMJ Open Resp Res. 2021;8:e000845). Similar findings were confirmed by the Maas et al. study, which utilized a large socioeconomically diverse database composed of 10 hospital systems. Diagnoses and outcomes were identified by ICD-10 coding and medical record data. After adjustments for diabetes, HTN, and BMI, OSA conferred an eight-fold risk for COVID-19 infection, was associated with increased risk of hospitalization, and doubled the risk of developing respiratory failure (Maas MB, et al. Sleep Breath. 2020 Sep; 29:1-3. doi: 10.1007/s11325-020-02203-0).
Peker and colleagues conducted a prospective multicenter observational study comparing clinical outcomes of severe COVID-19 infection in patients with low vs high pretest probability of having OSA based on the Berlin questionnaire. The authors reported a clinically significant risk of poorer clinical outcomes in the high pretest probability OSA group after adjustments for age, sex, and comorbidities (Peker Y, et al. Ann Am Thorac Soc. 2021. Feb 17. doi: 10.1513/AnnalsATS.202011-1409OC). A timely meta-analysis including 21 studies (19 with retrospective design) with 54,276 COVID-19 patients and 4,640 OSA patients concluded poor composite outcomes including severe COVID-19, intensive care unit admission, mechanical ventilatory support, and death in association with OSA (OR – 1.72 95% CI 1.55-1.91, P< .00001). In patients with obesity, OSA is a highly prevalent co-morbid condition. BMI, however, was not adjusted in this model (Hariyanto TI, et al. Sleep Med. 2021. doi: 10.1016/j.sleep.2021.03.029).
Other studies have concluded the opposite with OSA not being an independent risk factor for severe COVID-19 infection. Cade and colleagues conducted a retrospective analysis from a comprehensive electronic health dataset using ICD codes to identify OSA patients with severe COVID-19 infection. A significant association between OSA and COVID-19 death was noted after adjustment for demographics (ethnicity, age, sex). However, when fully adjusted for demographics, BMI, asthma, COPD, HTN, or DM, OSA was not an independent risk factor for COVID-19-related mortality and hospitalization (Cade BE, et al. Am J Respir Crit Care Med. 2020;202[10]:1462). The FinnGen study (Strausz et al.) was part of a meta-analysis examining the association between OSA and severe COVID-19 with and without adjustments for BMI. This meta-analysis consisted of 15,835 COVID-19 patients including 1,294 with OSA. The authors found that OSA was a risk factor with a two-fold increased risk of severe COVID-19 infection (OR = 2.37, P = .021). However, after adjustments were made for BMI, this finding lost statistical significance (OR=1.55, P=.13) (Strausz S, et al. BMJ Open Resp Res. 2021;8:e000845).
It is worth noting that a majority of studies identified OSA by indirect and imperfect methods through chart review, ICD codes, and databases. Confirmed OSA based on formal testing with a sleep study in COVID-19 patients remains a challenge. Perhaps well performed screening questionnaires, such as STOP-Bang, Berlin, or NoSAS, can be utilized as was the case in one study. It is also unclear if outcomes of COVID-19 infection differ in patients with treated or untreated OSA, as raised by the CORONADO study. A recent cross-sectional telephone interview survey of patients with confirmed OSA in Iran alluded to higher prevalence of COVID-19 in patients with severe OSA with suggestion of lower prevalence in patients who were currently receiving OSA treatment with positive airway pressure (PAP) therapy (Najafi A, et al. Sleep Health. 2021 Feb;7[1]:14). This is a crucial question as PAP therapy is considered an aerosol-generating procedure (Lance CG. Cleve Clin J Med. 2020 May 5. doi: 10.3949/ccjm.87a.ccc003). Studies have suggested continued use of PAP therapy with additional measures to mitigate the spread of virus, since failure to use PAP could be deleterious to the patient’s quality of life. Interestingly, PAP adherence seemed to have improved during the pandemic as evidenced by a telephonic survey done in New York City that showed 88% of patients with OSA used a PAP device consistently (Attias D, et al. Eur Respir J. 2020 Jul 30;56[1]:2001607. doi: 10.1183/13993003.01607-2020).
In summary, the jury is still out on whether OSA is a facilitator for viral replication, or an independent risk factor for poor prognosis related to COVID-19 infection, or has no clinical relevance to COVID-19. COVID-19 and OSA share comorbidities and pathways leading to a systemic inflammatory cascade. Theoretically, it would make sense that OSA is a risk factor for severe COVID-19 infection; however, it remains to be proven. The recent studies are limited by retrospective and observational nature, imprecise OSA classification/diagnostic criteria, and confounded by difficult to control variables. Further research is needed to expand our understanding of OSA -induced intermittent hypoxemia, inflammation, and endothelial dysfunction that may play a role in COVID-19 morbidity and mortality. Until we have more clarity, close monitoring of OSA patients infected with COVID-19 is recommended along with implementation of safe protocols for continuation of PAP usage during the infectious phase. Identifying underlying comorbid conditions that contribute to worsening of a COVID-19 infectious course is a crucial step in improving clinical outcomes.
Dr. Sahni is Assistant Professor of Clinical Medicine, Division of Pulmonary, Critical Care, Sleep and Allergy, Department of Medicine, University of Illinois at Chicago. Dr. Cao is Clinical Associate Professor, Division of Sleep Medicine and Division of Neuromuscular Medicine, Department of Psychiatry and Department of Neurology, Stanford (Calif.) University.