User login
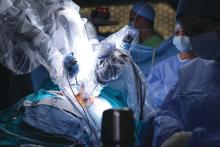
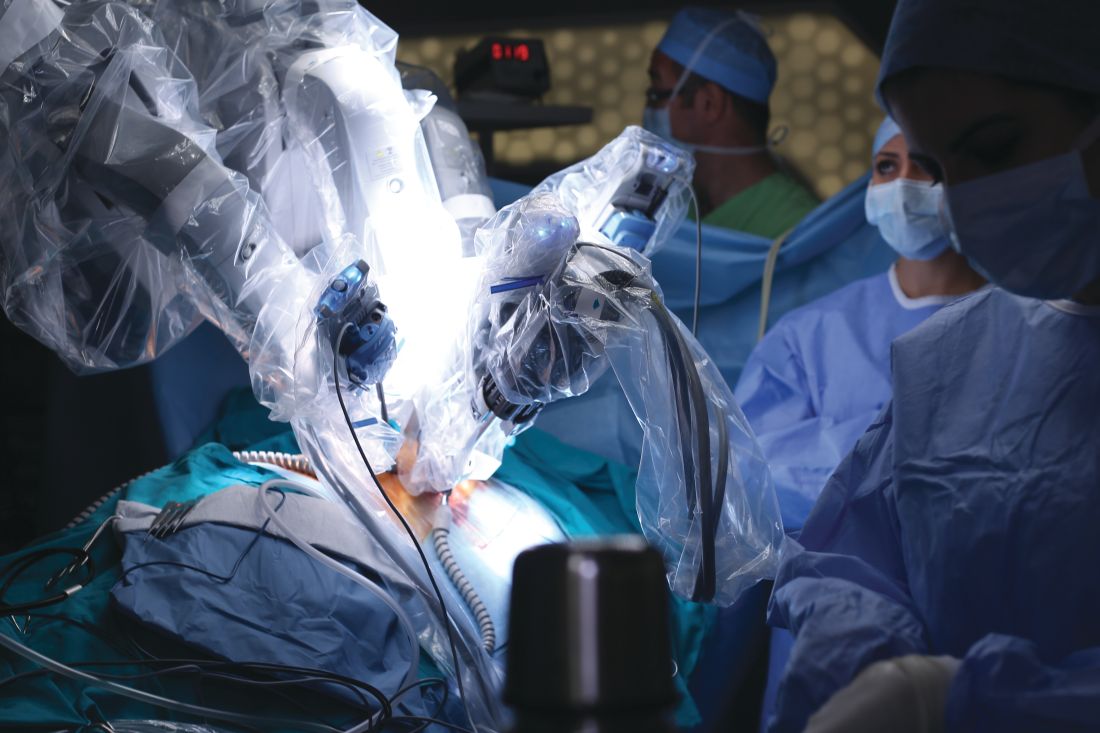
Practice makes better: Robotic lobectomy can shorten OR times
A new single-surgeon study suggests that and shave about 90 minutes off adjusted operating time.
The findings provide “further support to the adaptation of formalized robotic training and credentialing procedures,” wrote the authors of the retrospective, single-center study, which was presented at the 2018 Academic Surgical Congress and published in Surgery.
According to the study authors, advantages of robotic surgery, compared with thoracoscopic surgery, include “3-dimensional visualization, enhanced maneuverability in small spaces, and the ease of the hilar and mediastinal dissection. Disadvantages include the lack of haptic feedback, increased cost, and increased operative time.”
In the new study, the authors, led by thoracic surgeon Brian N. Arnold, MD, of Yale University, New Haven, Conn., attempted to quantify the learning curve in RATS pulmonary lobectomies by using a more “statistically rigorous” technique than previous studies.
The study tracked 101 of 116 consecutive patients who underwent RATS pulmonary lobectomy at a single unnamed center from 2010 to 2016. Some patients, such as those who underwent a right middle lobectomy that is considered an easier procedure, were excluded. All patients were treated by the same unidentified surgeon.
Researchers identified three phases of the RATS learning curve: cases 1-22, cases 23-63, and cases 64-101.
On average, the patients were aged 69 years; 52% were female. Overall, a third of the patients developed complications.
After controlling for various factors, the researchers found that adjusted operating time and estimated blood loss were statistically different between the first and second phases (P less than .05 and P = .016, respectively). They were also different between the first and third phases (P less than .05 and P = .006, respectively).
Specifically, operating time in the first phase was a mean of 256 minutes versus 195 minutes in the second phase (P = .0002) and 168 minutes in the third phase (P less than .0001). Blood loss was 200 mL (interquartile range, 150-300 mL) in the first phase versus 150 mL (IQR, 75-200 mL; P = .0219) in the second phase and 150 mL (IQR, 100-150 mL; P = .0096) in the third phase.
The researchers found no statistically significant evidence that the surgeon’s growing experience affected length of stay, postoperative complications, chest tube duration, or conversion rate. No patients died within 30 or 90 days.
The researchers also compared operating time, length of stay, and complication rate in the RATS procedures with those in video-assisted thoracoscopic (VATS) lobectomies performed at the same institution from 2008 to 2014. There was only a statistically significant difference in mean operating time (RATS, 319 minutes; VATS, 253 minutes; P less than .001)
The study authors noted that the surgeon had extensive previous experience with VATS procedures. “Therefore, for better or for worse, the results may not apply to surgeons without this experience who move from open surgery to robotic surgery.”
Study funding and disclosures were not reported.
SOURCE: Arnold BN et al. Surgery. 2019 Feb;165(2):450-4.
A new single-surgeon study suggests that and shave about 90 minutes off adjusted operating time.
The findings provide “further support to the adaptation of formalized robotic training and credentialing procedures,” wrote the authors of the retrospective, single-center study, which was presented at the 2018 Academic Surgical Congress and published in Surgery.
According to the study authors, advantages of robotic surgery, compared with thoracoscopic surgery, include “3-dimensional visualization, enhanced maneuverability in small spaces, and the ease of the hilar and mediastinal dissection. Disadvantages include the lack of haptic feedback, increased cost, and increased operative time.”
In the new study, the authors, led by thoracic surgeon Brian N. Arnold, MD, of Yale University, New Haven, Conn., attempted to quantify the learning curve in RATS pulmonary lobectomies by using a more “statistically rigorous” technique than previous studies.
The study tracked 101 of 116 consecutive patients who underwent RATS pulmonary lobectomy at a single unnamed center from 2010 to 2016. Some patients, such as those who underwent a right middle lobectomy that is considered an easier procedure, were excluded. All patients were treated by the same unidentified surgeon.
Researchers identified three phases of the RATS learning curve: cases 1-22, cases 23-63, and cases 64-101.
On average, the patients were aged 69 years; 52% were female. Overall, a third of the patients developed complications.
After controlling for various factors, the researchers found that adjusted operating time and estimated blood loss were statistically different between the first and second phases (P less than .05 and P = .016, respectively). They were also different between the first and third phases (P less than .05 and P = .006, respectively).
Specifically, operating time in the first phase was a mean of 256 minutes versus 195 minutes in the second phase (P = .0002) and 168 minutes in the third phase (P less than .0001). Blood loss was 200 mL (interquartile range, 150-300 mL) in the first phase versus 150 mL (IQR, 75-200 mL; P = .0219) in the second phase and 150 mL (IQR, 100-150 mL; P = .0096) in the third phase.
The researchers found no statistically significant evidence that the surgeon’s growing experience affected length of stay, postoperative complications, chest tube duration, or conversion rate. No patients died within 30 or 90 days.
The researchers also compared operating time, length of stay, and complication rate in the RATS procedures with those in video-assisted thoracoscopic (VATS) lobectomies performed at the same institution from 2008 to 2014. There was only a statistically significant difference in mean operating time (RATS, 319 minutes; VATS, 253 minutes; P less than .001)
The study authors noted that the surgeon had extensive previous experience with VATS procedures. “Therefore, for better or for worse, the results may not apply to surgeons without this experience who move from open surgery to robotic surgery.”
Study funding and disclosures were not reported.
SOURCE: Arnold BN et al. Surgery. 2019 Feb;165(2):450-4.
A new single-surgeon study suggests that and shave about 90 minutes off adjusted operating time.
The findings provide “further support to the adaptation of formalized robotic training and credentialing procedures,” wrote the authors of the retrospective, single-center study, which was presented at the 2018 Academic Surgical Congress and published in Surgery.
According to the study authors, advantages of robotic surgery, compared with thoracoscopic surgery, include “3-dimensional visualization, enhanced maneuverability in small spaces, and the ease of the hilar and mediastinal dissection. Disadvantages include the lack of haptic feedback, increased cost, and increased operative time.”
In the new study, the authors, led by thoracic surgeon Brian N. Arnold, MD, of Yale University, New Haven, Conn., attempted to quantify the learning curve in RATS pulmonary lobectomies by using a more “statistically rigorous” technique than previous studies.
The study tracked 101 of 116 consecutive patients who underwent RATS pulmonary lobectomy at a single unnamed center from 2010 to 2016. Some patients, such as those who underwent a right middle lobectomy that is considered an easier procedure, were excluded. All patients were treated by the same unidentified surgeon.
Researchers identified three phases of the RATS learning curve: cases 1-22, cases 23-63, and cases 64-101.
On average, the patients were aged 69 years; 52% were female. Overall, a third of the patients developed complications.
After controlling for various factors, the researchers found that adjusted operating time and estimated blood loss were statistically different between the first and second phases (P less than .05 and P = .016, respectively). They were also different between the first and third phases (P less than .05 and P = .006, respectively).
Specifically, operating time in the first phase was a mean of 256 minutes versus 195 minutes in the second phase (P = .0002) and 168 minutes in the third phase (P less than .0001). Blood loss was 200 mL (interquartile range, 150-300 mL) in the first phase versus 150 mL (IQR, 75-200 mL; P = .0219) in the second phase and 150 mL (IQR, 100-150 mL; P = .0096) in the third phase.
The researchers found no statistically significant evidence that the surgeon’s growing experience affected length of stay, postoperative complications, chest tube duration, or conversion rate. No patients died within 30 or 90 days.
The researchers also compared operating time, length of stay, and complication rate in the RATS procedures with those in video-assisted thoracoscopic (VATS) lobectomies performed at the same institution from 2008 to 2014. There was only a statistically significant difference in mean operating time (RATS, 319 minutes; VATS, 253 minutes; P less than .001)
The study authors noted that the surgeon had extensive previous experience with VATS procedures. “Therefore, for better or for worse, the results may not apply to surgeons without this experience who move from open surgery to robotic surgery.”
Study funding and disclosures were not reported.
SOURCE: Arnold BN et al. Surgery. 2019 Feb;165(2):450-4.
FROM SURGERY
Key clinical point: Extensive experience in robot-assisted thoracoscopic (RATS) pulmonary lobectomies could lead to dramatically shorter adjusted operating time.
Major finding: From a surgeon’s first 22 surgeries to cases 64-101, mean operating time fell from 256 minutes to 168 minutes, (P less than .05).
Study details: A retrospective, single-center, single-surgeon study of 101 patients who underwent robot-assisted thoracoscopic pulmonary lobectomies from 2010 to 2016.
Disclosures: Study funding and disclosures were not reported.
Source: Arnold BN et al. Surgery. 2019 Feb;165(2):450-4.
Impella RP shows higher mortality in postapproval study
The Food and Drug Administration issued a letter on Feb. 4, 2019, to health care providers regarding interim results from a postapproval study for Abiomed’s Impella RP System because these results appear to have a higher mortality rate than was seen in premarket clinical studies.
As a condition of its approval, the FDA mandated Abiomed to perform a postapproval study (PAS); this study reflects use in a broader population than the premarket studies, which adhered to stricter inclusion and exclusion criteria.
Earlier in January, Abiomed submitted data to the FDA suggesting that differences in preimplant characteristics between patients in the PAS and those in the premarket clinical studies may explain the difference in mortality. Specifically, 16 of the 23 patients enrolled in the PAS would not have met the enrollment criteria for the premarket clinical studies because they were in cardiogenic shock for longer than 48 hours, experienced an in-hospital cardiac arrest, were treated with an intra-aortic balloon pump, or suffered a preimplant hypoxic or ischemic neurologic event.
“Although the FDA is concerned about the high mortality rate from the interim PAS results,” they wrote in the letter, which is available on the FDA website, “we believe that, when the device is used for the currently approved indication in appropriately selected patients, the benefits of the Impella RP system continue to outweigh the risks.”
The Food and Drug Administration issued a letter on Feb. 4, 2019, to health care providers regarding interim results from a postapproval study for Abiomed’s Impella RP System because these results appear to have a higher mortality rate than was seen in premarket clinical studies.
As a condition of its approval, the FDA mandated Abiomed to perform a postapproval study (PAS); this study reflects use in a broader population than the premarket studies, which adhered to stricter inclusion and exclusion criteria.
Earlier in January, Abiomed submitted data to the FDA suggesting that differences in preimplant characteristics between patients in the PAS and those in the premarket clinical studies may explain the difference in mortality. Specifically, 16 of the 23 patients enrolled in the PAS would not have met the enrollment criteria for the premarket clinical studies because they were in cardiogenic shock for longer than 48 hours, experienced an in-hospital cardiac arrest, were treated with an intra-aortic balloon pump, or suffered a preimplant hypoxic or ischemic neurologic event.
“Although the FDA is concerned about the high mortality rate from the interim PAS results,” they wrote in the letter, which is available on the FDA website, “we believe that, when the device is used for the currently approved indication in appropriately selected patients, the benefits of the Impella RP system continue to outweigh the risks.”
The Food and Drug Administration issued a letter on Feb. 4, 2019, to health care providers regarding interim results from a postapproval study for Abiomed’s Impella RP System because these results appear to have a higher mortality rate than was seen in premarket clinical studies.
As a condition of its approval, the FDA mandated Abiomed to perform a postapproval study (PAS); this study reflects use in a broader population than the premarket studies, which adhered to stricter inclusion and exclusion criteria.
Earlier in January, Abiomed submitted data to the FDA suggesting that differences in preimplant characteristics between patients in the PAS and those in the premarket clinical studies may explain the difference in mortality. Specifically, 16 of the 23 patients enrolled in the PAS would not have met the enrollment criteria for the premarket clinical studies because they were in cardiogenic shock for longer than 48 hours, experienced an in-hospital cardiac arrest, were treated with an intra-aortic balloon pump, or suffered a preimplant hypoxic or ischemic neurologic event.
“Although the FDA is concerned about the high mortality rate from the interim PAS results,” they wrote in the letter, which is available on the FDA website, “we believe that, when the device is used for the currently approved indication in appropriately selected patients, the benefits of the Impella RP system continue to outweigh the risks.”
Different disease features found with family history of psoriasis versus PsA
the results of a retrospective cohort study suggest.
A family history of psoriasis was associated with younger onset of psoriatic disease and the presence of enthesitis, while by contrast, a family history of psoriatic arthritis (PsA) was associated with lower risk of plaque psoriasis and higher risk of deformities, according to Dilek Solmaz, MD, of the University of Ottawa and her coauthors, who reported their findings in Arthritis Care & Research.
“The link between family history of psoriasis/psoriatic arthritis and pustular/plaque phenotypes may point to a different genetic background and pathogenic mechanisms in these subsets,” the investigators wrote.
Most, if not all, previous studies evaluating family history have grouped psoriasis and PsA together, according to Dr. Solmaz and her colleagues, rather than looking at the individual effects of psoriasis or PsA family history that may lead to unique disease phenotypes, as was done in the present study.
The investigators based their retrospective analysis on patients recruited in a longitudinal, multicenter database in Turkey and Canada. The mean age of patients in the study was 48 years; nearly 65% were female.
Out of 1,393 patients in the database, 444 had a family history of psoriasis or PsA. That included 335 patients with a psoriasis-only family history and 74 with a family history of PsA; another 35 patients weren’t sure about having a family history of PsA or psoriasis and were left out of the analysis.
Plaque psoriasis was more common in individuals with a family history of only psoriasis, while pustular psoriasis was more common in those with a PsA family history, the investigators reported.
In multivariate analyses, having a family member with psoriasis was a risk factor for younger age of psoriasis onset (odds ratio, 0.976; 95% confidence interval, 0.964-0.989; P less than .001) as well as a higher risk for enthesitis (OR, 1.931; 95% CI, 1.276-2.922; P = .002) when compared against patients without a family history of psoriasis.
Patients with a family history of PsA were more likely to have deformities (OR, 2.557; 95% CI, 1.250-5.234; P less than .010) and lower risk of plaque-type psoriasis (OR, 0.417; 95% CI, 0.213-0.816; P less than .011) than patients without a family history of PsA.
Disease onset was earlier among patients with a family history of psoriasis at a mean of 28.1 years versus 31.9 years for those with a family history of PsA (P less than .001).
Dr. Solmaz and her colleagues reported no conflicts of interest related to the research, which was supported in part by the Turkish Society for Rheumatology, the Scientific and Technological Research Council of Turkey, and Union Chimique Belge.
SOURCE: Solmaz D et al. Arthritis Care Res (Hoboken). 2019 Jan 25. doi: 10.1002/acr.23836.
the results of a retrospective cohort study suggest.
A family history of psoriasis was associated with younger onset of psoriatic disease and the presence of enthesitis, while by contrast, a family history of psoriatic arthritis (PsA) was associated with lower risk of plaque psoriasis and higher risk of deformities, according to Dilek Solmaz, MD, of the University of Ottawa and her coauthors, who reported their findings in Arthritis Care & Research.
“The link between family history of psoriasis/psoriatic arthritis and pustular/plaque phenotypes may point to a different genetic background and pathogenic mechanisms in these subsets,” the investigators wrote.
Most, if not all, previous studies evaluating family history have grouped psoriasis and PsA together, according to Dr. Solmaz and her colleagues, rather than looking at the individual effects of psoriasis or PsA family history that may lead to unique disease phenotypes, as was done in the present study.
The investigators based their retrospective analysis on patients recruited in a longitudinal, multicenter database in Turkey and Canada. The mean age of patients in the study was 48 years; nearly 65% were female.
Out of 1,393 patients in the database, 444 had a family history of psoriasis or PsA. That included 335 patients with a psoriasis-only family history and 74 with a family history of PsA; another 35 patients weren’t sure about having a family history of PsA or psoriasis and were left out of the analysis.
Plaque psoriasis was more common in individuals with a family history of only psoriasis, while pustular psoriasis was more common in those with a PsA family history, the investigators reported.
In multivariate analyses, having a family member with psoriasis was a risk factor for younger age of psoriasis onset (odds ratio, 0.976; 95% confidence interval, 0.964-0.989; P less than .001) as well as a higher risk for enthesitis (OR, 1.931; 95% CI, 1.276-2.922; P = .002) when compared against patients without a family history of psoriasis.
Patients with a family history of PsA were more likely to have deformities (OR, 2.557; 95% CI, 1.250-5.234; P less than .010) and lower risk of plaque-type psoriasis (OR, 0.417; 95% CI, 0.213-0.816; P less than .011) than patients without a family history of PsA.
Disease onset was earlier among patients with a family history of psoriasis at a mean of 28.1 years versus 31.9 years for those with a family history of PsA (P less than .001).
Dr. Solmaz and her colleagues reported no conflicts of interest related to the research, which was supported in part by the Turkish Society for Rheumatology, the Scientific and Technological Research Council of Turkey, and Union Chimique Belge.
SOURCE: Solmaz D et al. Arthritis Care Res (Hoboken). 2019 Jan 25. doi: 10.1002/acr.23836.
the results of a retrospective cohort study suggest.
A family history of psoriasis was associated with younger onset of psoriatic disease and the presence of enthesitis, while by contrast, a family history of psoriatic arthritis (PsA) was associated with lower risk of plaque psoriasis and higher risk of deformities, according to Dilek Solmaz, MD, of the University of Ottawa and her coauthors, who reported their findings in Arthritis Care & Research.
“The link between family history of psoriasis/psoriatic arthritis and pustular/plaque phenotypes may point to a different genetic background and pathogenic mechanisms in these subsets,” the investigators wrote.
Most, if not all, previous studies evaluating family history have grouped psoriasis and PsA together, according to Dr. Solmaz and her colleagues, rather than looking at the individual effects of psoriasis or PsA family history that may lead to unique disease phenotypes, as was done in the present study.
The investigators based their retrospective analysis on patients recruited in a longitudinal, multicenter database in Turkey and Canada. The mean age of patients in the study was 48 years; nearly 65% were female.
Out of 1,393 patients in the database, 444 had a family history of psoriasis or PsA. That included 335 patients with a psoriasis-only family history and 74 with a family history of PsA; another 35 patients weren’t sure about having a family history of PsA or psoriasis and were left out of the analysis.
Plaque psoriasis was more common in individuals with a family history of only psoriasis, while pustular psoriasis was more common in those with a PsA family history, the investigators reported.
In multivariate analyses, having a family member with psoriasis was a risk factor for younger age of psoriasis onset (odds ratio, 0.976; 95% confidence interval, 0.964-0.989; P less than .001) as well as a higher risk for enthesitis (OR, 1.931; 95% CI, 1.276-2.922; P = .002) when compared against patients without a family history of psoriasis.
Patients with a family history of PsA were more likely to have deformities (OR, 2.557; 95% CI, 1.250-5.234; P less than .010) and lower risk of plaque-type psoriasis (OR, 0.417; 95% CI, 0.213-0.816; P less than .011) than patients without a family history of PsA.
Disease onset was earlier among patients with a family history of psoriasis at a mean of 28.1 years versus 31.9 years for those with a family history of PsA (P less than .001).
Dr. Solmaz and her colleagues reported no conflicts of interest related to the research, which was supported in part by the Turkish Society for Rheumatology, the Scientific and Technological Research Council of Turkey, and Union Chimique Belge.
SOURCE: Solmaz D et al. Arthritis Care Res (Hoboken). 2019 Jan 25. doi: 10.1002/acr.23836.
FROM ARTHRITIS CARE & RESEARCH
Key clinical point: Family histories of psoriasis and psoriatic arthritis were linked to different skin phenotypes, disease severity, and musculoskeletal features.
Major finding: Compared with no family history, psoriasis family history was a risk factor for enthesitis (odds ratio, 1.931) and younger age of onset (OR, 0.976) while psoriatic arthritis family history was linked to higher risk of deformities (OR, 2.557) and lower risk of plaque-type psoriasis (OR, 0.417).
Study details: A retrospective analysis including 1,393 Turkish or Canadian patients enrolled in a psoriatic arthritis database.
Disclosures: The study authors reported no conflicts of interest related to the research, which was supported in part by the Turkish Society for Rheumatology, the Scientific and Technological Research Council of Turkey, and Union Chimique Belge.
Source: Solmaz D et al. Arthritis Care Res (Hoboken). 2019 Jan 25. doi: 10.1002/acr.23836.
Biosimilars: Gary H. Lyman
In this episode, Gary H. Lyman, MD (https://bitly.is/2UJzUly) joins David Henry, MD, (http://bit.ly/2MFDfzm) to talk about biosimilars.
Dr. Lyman talks about the definition of biosimilars, how they are made, which are approved, and which ones are on the market.
He also talks about extrapolation and interchangeability as well as where some biosimilars stand in both ASCO and NCCN guidelines for patients who are going into supportive care.
And Ilana Yurkiewicz, MD (https://stanford.io/2RXPixR), talks about what the word "cure" means to you compared to what it means to patients in a world with OS, PFS, DFS, CR, etc. (26:35).
Subscribe here:
Show Notes
By Hitomi Hosoya, MD, PhD
- Biosimilars are biologic products that are highly similar to the reference products with no clinically meaningful difference in terms of safety, efficacy, purity, and potency.
- Unlike “generic” products, biosimilars are produced in living systems, therefore they cannot be replicated identically.
- The Food and Drug Administration encourages companies to produce biosimilars as a patent expires on the original product.
- For approval, the FDA requires largely preclinical data; amino acid sequence and molecular structures, pharmacodynamics and pharmacokinetic data in animal models and humans. Also, no greater immunogenicity should be demonstrated.
- Large randomized trials are not usually required for approval of biosimilars as there is already data on the original product. Post-marketing surveillance is important.
- 14 biosimilars have already been approved by the FDA.
- Interchangeable designation of biosimilars is yet to come.
- At this early stage of biosimilar marketing, we see a 10%-11% cost reduction. This is expected to increase as the market expands.
References:
JAMA Oncol.2018 Feb 1;4(2):241-247
Contact us: [email protected]
MDedge Hematology/Oncology Twitter: @MDedgeHemOnc
Ilana Yurkiewicz Twitter: @ilanayurkiewicz
In this episode, Gary H. Lyman, MD (https://bitly.is/2UJzUly) joins David Henry, MD, (http://bit.ly/2MFDfzm) to talk about biosimilars.
Dr. Lyman talks about the definition of biosimilars, how they are made, which are approved, and which ones are on the market.
He also talks about extrapolation and interchangeability as well as where some biosimilars stand in both ASCO and NCCN guidelines for patients who are going into supportive care.
And Ilana Yurkiewicz, MD (https://stanford.io/2RXPixR), talks about what the word "cure" means to you compared to what it means to patients in a world with OS, PFS, DFS, CR, etc. (26:35).
Subscribe here:
Show Notes
By Hitomi Hosoya, MD, PhD
- Biosimilars are biologic products that are highly similar to the reference products with no clinically meaningful difference in terms of safety, efficacy, purity, and potency.
- Unlike “generic” products, biosimilars are produced in living systems, therefore they cannot be replicated identically.
- The Food and Drug Administration encourages companies to produce biosimilars as a patent expires on the original product.
- For approval, the FDA requires largely preclinical data; amino acid sequence and molecular structures, pharmacodynamics and pharmacokinetic data in animal models and humans. Also, no greater immunogenicity should be demonstrated.
- Large randomized trials are not usually required for approval of biosimilars as there is already data on the original product. Post-marketing surveillance is important.
- 14 biosimilars have already been approved by the FDA.
- Interchangeable designation of biosimilars is yet to come.
- At this early stage of biosimilar marketing, we see a 10%-11% cost reduction. This is expected to increase as the market expands.
References:
JAMA Oncol.2018 Feb 1;4(2):241-247
Contact us: [email protected]
MDedge Hematology/Oncology Twitter: @MDedgeHemOnc
Ilana Yurkiewicz Twitter: @ilanayurkiewicz
In this episode, Gary H. Lyman, MD (https://bitly.is/2UJzUly) joins David Henry, MD, (http://bit.ly/2MFDfzm) to talk about biosimilars.
Dr. Lyman talks about the definition of biosimilars, how they are made, which are approved, and which ones are on the market.
He also talks about extrapolation and interchangeability as well as where some biosimilars stand in both ASCO and NCCN guidelines for patients who are going into supportive care.
And Ilana Yurkiewicz, MD (https://stanford.io/2RXPixR), talks about what the word "cure" means to you compared to what it means to patients in a world with OS, PFS, DFS, CR, etc. (26:35).
Subscribe here:
Show Notes
By Hitomi Hosoya, MD, PhD
- Biosimilars are biologic products that are highly similar to the reference products with no clinically meaningful difference in terms of safety, efficacy, purity, and potency.
- Unlike “generic” products, biosimilars are produced in living systems, therefore they cannot be replicated identically.
- The Food and Drug Administration encourages companies to produce biosimilars as a patent expires on the original product.
- For approval, the FDA requires largely preclinical data; amino acid sequence and molecular structures, pharmacodynamics and pharmacokinetic data in animal models and humans. Also, no greater immunogenicity should be demonstrated.
- Large randomized trials are not usually required for approval of biosimilars as there is already data on the original product. Post-marketing surveillance is important.
- 14 biosimilars have already been approved by the FDA.
- Interchangeable designation of biosimilars is yet to come.
- At this early stage of biosimilar marketing, we see a 10%-11% cost reduction. This is expected to increase as the market expands.
References:
JAMA Oncol.2018 Feb 1;4(2):241-247
Contact us: [email protected]
MDedge Hematology/Oncology Twitter: @MDedgeHemOnc
Ilana Yurkiewicz Twitter: @ilanayurkiewicz
Rise in HCV linked to OxyContin reformulation
Also today, public health experts have attributed to the opioid epidemic the recent alarming rise in the rate of hepatitis C virus infection. medical advice prompts unneeded emergency visits by patients with atrial fibrillation, and legal marijuana may complicate substance use disorder treatment in adolescents.
Amazon Alexa
Apple Podcasts
Google Podcasts
Spotify
Also today, public health experts have attributed to the opioid epidemic the recent alarming rise in the rate of hepatitis C virus infection. medical advice prompts unneeded emergency visits by patients with atrial fibrillation, and legal marijuana may complicate substance use disorder treatment in adolescents.
Amazon Alexa
Apple Podcasts
Google Podcasts
Spotify
Also today, public health experts have attributed to the opioid epidemic the recent alarming rise in the rate of hepatitis C virus infection. medical advice prompts unneeded emergency visits by patients with atrial fibrillation, and legal marijuana may complicate substance use disorder treatment in adolescents.
Amazon Alexa
Apple Podcasts
Google Podcasts
Spotify
Loan Repayment Plan for Substance Use Clinicians
The opioid emergency claims > 130 lives every day, says Health Resources and Services Administration (HRSA) Administrator George Sigounas, MS, PhD. By strengthening the health workforce, HRSA hopes to ensure that there are enough clinicians to cope with the growing epidemic.
That is why, in December 2018, HRSA launched a program that Sigounas says is “critical to HHS’ response to the opioid crisis.” The new National Health Service Corps (NHSC) Substance Use Disorder (SUD) Workforce Loan Repayment Program (LRP) will provide eligible health care clinicians with student loan repayment assistance in exchange for service in underserved communities.
A clinician may be awarded up to $75,000 for 3 years of full-time service at an NHSC-approved SUD site and $37,500 for part-time. Eligible providers use evidence-based treatment models to treat SUDs and must be trained and licensed to provide SUD treatment at NHSC-approved facilities. Qualification criteria are available at https://nhsc.hrsa.gov/loan-repayment/nhsc-sud-workforce-loan-repayment-program.html.
Clinicians also can apply to the NHSC Loan Repayment Program for primary care, dental, and behavioral health professionals. If accepted, they may receive up to $50,000 for 2 years of full-time service, $25,000 for part-time.
Military reservists also are eligible to participate in either the NHSC LRP or the NHSC Students to Service Loan Repayment Program. (Military training or service will not satisfy the NHSC service commitment.) More information is available at https://nhsc.hrsa.gov/loan-repayment/military-reservists.html.
Clinicians can only apply for 1 program. Sigounas says, “I am grateful to the clinicians who will apply and are looking to make a positive impact on patients, caregivers, and hard-hit communities throughout the country.”
The opioid emergency claims > 130 lives every day, says Health Resources and Services Administration (HRSA) Administrator George Sigounas, MS, PhD. By strengthening the health workforce, HRSA hopes to ensure that there are enough clinicians to cope with the growing epidemic.
That is why, in December 2018, HRSA launched a program that Sigounas says is “critical to HHS’ response to the opioid crisis.” The new National Health Service Corps (NHSC) Substance Use Disorder (SUD) Workforce Loan Repayment Program (LRP) will provide eligible health care clinicians with student loan repayment assistance in exchange for service in underserved communities.
A clinician may be awarded up to $75,000 for 3 years of full-time service at an NHSC-approved SUD site and $37,500 for part-time. Eligible providers use evidence-based treatment models to treat SUDs and must be trained and licensed to provide SUD treatment at NHSC-approved facilities. Qualification criteria are available at https://nhsc.hrsa.gov/loan-repayment/nhsc-sud-workforce-loan-repayment-program.html.
Clinicians also can apply to the NHSC Loan Repayment Program for primary care, dental, and behavioral health professionals. If accepted, they may receive up to $50,000 for 2 years of full-time service, $25,000 for part-time.
Military reservists also are eligible to participate in either the NHSC LRP or the NHSC Students to Service Loan Repayment Program. (Military training or service will not satisfy the NHSC service commitment.) More information is available at https://nhsc.hrsa.gov/loan-repayment/military-reservists.html.
Clinicians can only apply for 1 program. Sigounas says, “I am grateful to the clinicians who will apply and are looking to make a positive impact on patients, caregivers, and hard-hit communities throughout the country.”
The opioid emergency claims > 130 lives every day, says Health Resources and Services Administration (HRSA) Administrator George Sigounas, MS, PhD. By strengthening the health workforce, HRSA hopes to ensure that there are enough clinicians to cope with the growing epidemic.
That is why, in December 2018, HRSA launched a program that Sigounas says is “critical to HHS’ response to the opioid crisis.” The new National Health Service Corps (NHSC) Substance Use Disorder (SUD) Workforce Loan Repayment Program (LRP) will provide eligible health care clinicians with student loan repayment assistance in exchange for service in underserved communities.
A clinician may be awarded up to $75,000 for 3 years of full-time service at an NHSC-approved SUD site and $37,500 for part-time. Eligible providers use evidence-based treatment models to treat SUDs and must be trained and licensed to provide SUD treatment at NHSC-approved facilities. Qualification criteria are available at https://nhsc.hrsa.gov/loan-repayment/nhsc-sud-workforce-loan-repayment-program.html.
Clinicians also can apply to the NHSC Loan Repayment Program for primary care, dental, and behavioral health professionals. If accepted, they may receive up to $50,000 for 2 years of full-time service, $25,000 for part-time.
Military reservists also are eligible to participate in either the NHSC LRP or the NHSC Students to Service Loan Repayment Program. (Military training or service will not satisfy the NHSC service commitment.) More information is available at https://nhsc.hrsa.gov/loan-repayment/military-reservists.html.
Clinicians can only apply for 1 program. Sigounas says, “I am grateful to the clinicians who will apply and are looking to make a positive impact on patients, caregivers, and hard-hit communities throughout the country.”
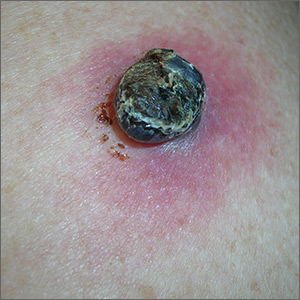
Black lesion on arm
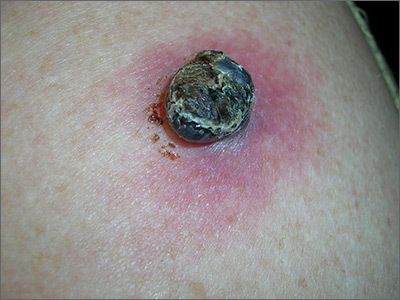
Due to the dark and rapidly growing nodule, the FP immediately worried about melanoma.
He thought that he should biopsy the entire lesion with an elliptical excision, so he scheduled the patient for a biopsy during some protected surgical time later that week. The patient did not show up for this appointment. Several calls were placed, and she returned for the biopsy the following week. The FP performed a narrow margin (2 mm) elliptical excision oriented to match the lymphatic drainage of the arm. He closed the excision with a 2-layer closure. (See the Watch & Learn video on elliptical excision.) The pathology report confirmed that it was a nodular melanoma that was 8 mm in depth. This was clearly an aggressive tumor, so the patient was referred to Surgical Oncology for sentinel lymph node biopsy. One node was positive for metastasis.
After a wide excision with 2 cm margins by Surgical Oncology, the patient underwent a course of chemotherapy and remained disease free 2 years later. She was carefully monitored for metastasis and new primary lesions by a multidisciplinary team that included family medicine, dermatology, and oncology.
While this FP handled the case in an excellent matter, he was fortunate to have the skills and time to be able to perform a full elliptical excision. It’s important to note that a 6 mm punch biopsy or a deep shave biopsy (saucerization) at the base of the thickest portion of this tumor would almost certainly have provided the same diagnosis of melanoma and at least showed that the tumor was thicker than 4 mm (an important cut-off for management). This could have been done on the day of original presentation and might have avoided the problem of the patient not showing up for the next appointment or a long delay to see a dermatologist.
FPs should be empowered to perform biopsies on the most worrisome of lesions as these biopsies can save lives. While incomplete sampling can result in false negative results and misdiagnosis, the protection against this is to not accept a benign pathology report in what appears to be an obvious malignancy. If this occurs, the next step is always complete excision. Having options and understanding potential sampling errors can help FPs diagnose patients more rapidly. This is essential when cancers are rapidly growing and delays of months for surgical appointments or referrals to specialists can worsen a prognosis.
Photos and text for Photo Rounds Friday courtesy of Richard P. Usatine, MD. This case was adapted from: Karnes J, Usatine R. Melanoma. In: Usatine R, Smith M, Mayeaux EJ, et al. Color Atlas and Synopsis of Family Medicine. 3rd ed. New York, NY: McGraw-Hill; 2019:1112-1123.
To learn more about the newest 3rd edition of the Color Atlas and Synopsis of Family Medicine, see: https://www.amazon.com/Color-Atlas-Synopsis-Family-Medicine/dp/1259862046/
You can get the Color Atlas of Family Medicine app by clicking on this link: usatinemedia.com

Due to the dark and rapidly growing nodule, the FP immediately worried about melanoma.
He thought that he should biopsy the entire lesion with an elliptical excision, so he scheduled the patient for a biopsy during some protected surgical time later that week. The patient did not show up for this appointment. Several calls were placed, and she returned for the biopsy the following week. The FP performed a narrow margin (2 mm) elliptical excision oriented to match the lymphatic drainage of the arm. He closed the excision with a 2-layer closure. (See the Watch & Learn video on elliptical excision.) The pathology report confirmed that it was a nodular melanoma that was 8 mm in depth. This was clearly an aggressive tumor, so the patient was referred to Surgical Oncology for sentinel lymph node biopsy. One node was positive for metastasis.
After a wide excision with 2 cm margins by Surgical Oncology, the patient underwent a course of chemotherapy and remained disease free 2 years later. She was carefully monitored for metastasis and new primary lesions by a multidisciplinary team that included family medicine, dermatology, and oncology.
While this FP handled the case in an excellent matter, he was fortunate to have the skills and time to be able to perform a full elliptical excision. It’s important to note that a 6 mm punch biopsy or a deep shave biopsy (saucerization) at the base of the thickest portion of this tumor would almost certainly have provided the same diagnosis of melanoma and at least showed that the tumor was thicker than 4 mm (an important cut-off for management). This could have been done on the day of original presentation and might have avoided the problem of the patient not showing up for the next appointment or a long delay to see a dermatologist.
FPs should be empowered to perform biopsies on the most worrisome of lesions as these biopsies can save lives. While incomplete sampling can result in false negative results and misdiagnosis, the protection against this is to not accept a benign pathology report in what appears to be an obvious malignancy. If this occurs, the next step is always complete excision. Having options and understanding potential sampling errors can help FPs diagnose patients more rapidly. This is essential when cancers are rapidly growing and delays of months for surgical appointments or referrals to specialists can worsen a prognosis.
Photos and text for Photo Rounds Friday courtesy of Richard P. Usatine, MD. This case was adapted from: Karnes J, Usatine R. Melanoma. In: Usatine R, Smith M, Mayeaux EJ, et al. Color Atlas and Synopsis of Family Medicine. 3rd ed. New York, NY: McGraw-Hill; 2019:1112-1123.
To learn more about the newest 3rd edition of the Color Atlas and Synopsis of Family Medicine, see: https://www.amazon.com/Color-Atlas-Synopsis-Family-Medicine/dp/1259862046/
You can get the Color Atlas of Family Medicine app by clicking on this link: usatinemedia.com

Due to the dark and rapidly growing nodule, the FP immediately worried about melanoma.
He thought that he should biopsy the entire lesion with an elliptical excision, so he scheduled the patient for a biopsy during some protected surgical time later that week. The patient did not show up for this appointment. Several calls were placed, and she returned for the biopsy the following week. The FP performed a narrow margin (2 mm) elliptical excision oriented to match the lymphatic drainage of the arm. He closed the excision with a 2-layer closure. (See the Watch & Learn video on elliptical excision.) The pathology report confirmed that it was a nodular melanoma that was 8 mm in depth. This was clearly an aggressive tumor, so the patient was referred to Surgical Oncology for sentinel lymph node biopsy. One node was positive for metastasis.
After a wide excision with 2 cm margins by Surgical Oncology, the patient underwent a course of chemotherapy and remained disease free 2 years later. She was carefully monitored for metastasis and new primary lesions by a multidisciplinary team that included family medicine, dermatology, and oncology.
While this FP handled the case in an excellent matter, he was fortunate to have the skills and time to be able to perform a full elliptical excision. It’s important to note that a 6 mm punch biopsy or a deep shave biopsy (saucerization) at the base of the thickest portion of this tumor would almost certainly have provided the same diagnosis of melanoma and at least showed that the tumor was thicker than 4 mm (an important cut-off for management). This could have been done on the day of original presentation and might have avoided the problem of the patient not showing up for the next appointment or a long delay to see a dermatologist.
FPs should be empowered to perform biopsies on the most worrisome of lesions as these biopsies can save lives. While incomplete sampling can result in false negative results and misdiagnosis, the protection against this is to not accept a benign pathology report in what appears to be an obvious malignancy. If this occurs, the next step is always complete excision. Having options and understanding potential sampling errors can help FPs diagnose patients more rapidly. This is essential when cancers are rapidly growing and delays of months for surgical appointments or referrals to specialists can worsen a prognosis.
Photos and text for Photo Rounds Friday courtesy of Richard P. Usatine, MD. This case was adapted from: Karnes J, Usatine R. Melanoma. In: Usatine R, Smith M, Mayeaux EJ, et al. Color Atlas and Synopsis of Family Medicine. 3rd ed. New York, NY: McGraw-Hill; 2019:1112-1123.
To learn more about the newest 3rd edition of the Color Atlas and Synopsis of Family Medicine, see: https://www.amazon.com/Color-Atlas-Synopsis-Family-Medicine/dp/1259862046/
You can get the Color Atlas of Family Medicine app by clicking on this link: usatinemedia.com
Roux-en-Y achieves diabetes remission in majority of patients
Around three-quarters of people with type 2 diabetes mellitus (T2DM) who undergo Roux-en-Y gastric bypass experience remission of their disease within a year of the surgery, according to published findings from a population-based observational study. However, one in four of those people will have relapsed by 5 years, the authors noted.
Researchers looked at the effect of Roux-en-Y gastric bypass (RYGB) in 1,111 individuals with T2DM, compared with 1,074 controls who also had T2DM but did not undergo gastric bypass.
By 6 months after surgery, 65% of those who had undergone RYGB met the criteria for remission – defined as no use of glucose-lowering drugs and an HbA1c below 48 mmol/mol (less than 6.5%) or metformin monotherapy with HbA1c below 42 mmol/mol (less than 6.0%).
By 1 year, 74% of those who had surgery had achieved remission, and 73% of those remained in remission 5 years after surgery. However, at 2 years, 6% of those who had achieved remission in the first year had already relapsed; by 3 years, 12% had relapsed; and by 4 years, 18% had relapsed. By 5 years after surgery, a total of 27% of those who originally achieved remission in the first year had relapsed.
The overall prevalence of remission remained at 70% for every 6-month period during the duration of the study, which suggests that, although some achieved remission early and then relapsed, others achieved remission later.
Individuals who were aged 50-60 years were 12% less likely to achieve remission, compared with those who were younger than 40 years, whereas those aged 60 years or more were 17% less likely to achieve remission.
A longer duration of diabetes was also associated with a lower likelihood of achieving remission after RYGB; individuals who had had diabetes for 8 years or more had a 27% lower likelihood of remission, compared with those who had had the disease for less than 2 years.
A higher HbA1c (greater than 53 mmol/mol) was associated with a 19% lower likelihood of remission, and individuals using insulin had a 43% lower likelihood of remission.
“Overall, our findings add evidence to the importance of regular check-ups following RYGB, despite initial diabetes remission, and also suggest that timing of RYGB is important (i.e., consider RYGB while there are still functional pancreatic beta cells),” wrote Lene R. Madsen, MD, from the department of endocrinology and internal medicine at Aarhus (Denmark) University Hospital and her colleagues.
The study also examined the effect of RYGB on microvascular and macrovascular diabetes complications. This revealed that the incidence of diabetic retinopathy was nearly halved among individuals who had undergone gastric bypass, the incidence of hospital-coded diabetic kidney disease was 46% lower, and the incidence of diabetic neuropathy was 16% lower.
In particular, individuals who achieved remission in the first year after surgery had a 57% lower incidence of microvascular events, compared with those who did not have surgery.
The authors noted that individuals who did not reach the threshold for diabetes remission after surgery still showed signs of better glycemic control, compared with individuals who had not undergone surgery.
“This aligns with the theory of ‘metabolic memory’ introduced by Coleman et al. [Diabetes Care. 2016;39(8):1400-07], suggesting that time spent in diabetes remission after RYGB is not spent in vain when it comes to reducing the risk of subsequent microvascular complications,” they wrote.
The surgery was also associated with a 46% reduction in the incidence of ischemic heart disease. In the first 30 days after surgery, 7.5% of patients were readmitted to hospital for any surgical complication, but the 90-day mortality rate after surgery was less than 0.5%.
The study was supported by the Health Research Fund of Central Denmark, the Novo Nordisk Foundation, and the A.P. Møller Foundation. The authors reported no conflicts of interest.
SOURCE: Madsen LR et al. Diabetologia. 2019, Feb 6. doi: 10.1007/s00125-019-4816-2.
Around three-quarters of people with type 2 diabetes mellitus (T2DM) who undergo Roux-en-Y gastric bypass experience remission of their disease within a year of the surgery, according to published findings from a population-based observational study. However, one in four of those people will have relapsed by 5 years, the authors noted.
Researchers looked at the effect of Roux-en-Y gastric bypass (RYGB) in 1,111 individuals with T2DM, compared with 1,074 controls who also had T2DM but did not undergo gastric bypass.
By 6 months after surgery, 65% of those who had undergone RYGB met the criteria for remission – defined as no use of glucose-lowering drugs and an HbA1c below 48 mmol/mol (less than 6.5%) or metformin monotherapy with HbA1c below 42 mmol/mol (less than 6.0%).
By 1 year, 74% of those who had surgery had achieved remission, and 73% of those remained in remission 5 years after surgery. However, at 2 years, 6% of those who had achieved remission in the first year had already relapsed; by 3 years, 12% had relapsed; and by 4 years, 18% had relapsed. By 5 years after surgery, a total of 27% of those who originally achieved remission in the first year had relapsed.
The overall prevalence of remission remained at 70% for every 6-month period during the duration of the study, which suggests that, although some achieved remission early and then relapsed, others achieved remission later.
Individuals who were aged 50-60 years were 12% less likely to achieve remission, compared with those who were younger than 40 years, whereas those aged 60 years or more were 17% less likely to achieve remission.
A longer duration of diabetes was also associated with a lower likelihood of achieving remission after RYGB; individuals who had had diabetes for 8 years or more had a 27% lower likelihood of remission, compared with those who had had the disease for less than 2 years.
A higher HbA1c (greater than 53 mmol/mol) was associated with a 19% lower likelihood of remission, and individuals using insulin had a 43% lower likelihood of remission.
“Overall, our findings add evidence to the importance of regular check-ups following RYGB, despite initial diabetes remission, and also suggest that timing of RYGB is important (i.e., consider RYGB while there are still functional pancreatic beta cells),” wrote Lene R. Madsen, MD, from the department of endocrinology and internal medicine at Aarhus (Denmark) University Hospital and her colleagues.
The study also examined the effect of RYGB on microvascular and macrovascular diabetes complications. This revealed that the incidence of diabetic retinopathy was nearly halved among individuals who had undergone gastric bypass, the incidence of hospital-coded diabetic kidney disease was 46% lower, and the incidence of diabetic neuropathy was 16% lower.
In particular, individuals who achieved remission in the first year after surgery had a 57% lower incidence of microvascular events, compared with those who did not have surgery.
The authors noted that individuals who did not reach the threshold for diabetes remission after surgery still showed signs of better glycemic control, compared with individuals who had not undergone surgery.
“This aligns with the theory of ‘metabolic memory’ introduced by Coleman et al. [Diabetes Care. 2016;39(8):1400-07], suggesting that time spent in diabetes remission after RYGB is not spent in vain when it comes to reducing the risk of subsequent microvascular complications,” they wrote.
The surgery was also associated with a 46% reduction in the incidence of ischemic heart disease. In the first 30 days after surgery, 7.5% of patients were readmitted to hospital for any surgical complication, but the 90-day mortality rate after surgery was less than 0.5%.
The study was supported by the Health Research Fund of Central Denmark, the Novo Nordisk Foundation, and the A.P. Møller Foundation. The authors reported no conflicts of interest.
SOURCE: Madsen LR et al. Diabetologia. 2019, Feb 6. doi: 10.1007/s00125-019-4816-2.
Around three-quarters of people with type 2 diabetes mellitus (T2DM) who undergo Roux-en-Y gastric bypass experience remission of their disease within a year of the surgery, according to published findings from a population-based observational study. However, one in four of those people will have relapsed by 5 years, the authors noted.
Researchers looked at the effect of Roux-en-Y gastric bypass (RYGB) in 1,111 individuals with T2DM, compared with 1,074 controls who also had T2DM but did not undergo gastric bypass.
By 6 months after surgery, 65% of those who had undergone RYGB met the criteria for remission – defined as no use of glucose-lowering drugs and an HbA1c below 48 mmol/mol (less than 6.5%) or metformin monotherapy with HbA1c below 42 mmol/mol (less than 6.0%).
By 1 year, 74% of those who had surgery had achieved remission, and 73% of those remained in remission 5 years after surgery. However, at 2 years, 6% of those who had achieved remission in the first year had already relapsed; by 3 years, 12% had relapsed; and by 4 years, 18% had relapsed. By 5 years after surgery, a total of 27% of those who originally achieved remission in the first year had relapsed.
The overall prevalence of remission remained at 70% for every 6-month period during the duration of the study, which suggests that, although some achieved remission early and then relapsed, others achieved remission later.
Individuals who were aged 50-60 years were 12% less likely to achieve remission, compared with those who were younger than 40 years, whereas those aged 60 years or more were 17% less likely to achieve remission.
A longer duration of diabetes was also associated with a lower likelihood of achieving remission after RYGB; individuals who had had diabetes for 8 years or more had a 27% lower likelihood of remission, compared with those who had had the disease for less than 2 years.
A higher HbA1c (greater than 53 mmol/mol) was associated with a 19% lower likelihood of remission, and individuals using insulin had a 43% lower likelihood of remission.
“Overall, our findings add evidence to the importance of regular check-ups following RYGB, despite initial diabetes remission, and also suggest that timing of RYGB is important (i.e., consider RYGB while there are still functional pancreatic beta cells),” wrote Lene R. Madsen, MD, from the department of endocrinology and internal medicine at Aarhus (Denmark) University Hospital and her colleagues.
The study also examined the effect of RYGB on microvascular and macrovascular diabetes complications. This revealed that the incidence of diabetic retinopathy was nearly halved among individuals who had undergone gastric bypass, the incidence of hospital-coded diabetic kidney disease was 46% lower, and the incidence of diabetic neuropathy was 16% lower.
In particular, individuals who achieved remission in the first year after surgery had a 57% lower incidence of microvascular events, compared with those who did not have surgery.
The authors noted that individuals who did not reach the threshold for diabetes remission after surgery still showed signs of better glycemic control, compared with individuals who had not undergone surgery.
“This aligns with the theory of ‘metabolic memory’ introduced by Coleman et al. [Diabetes Care. 2016;39(8):1400-07], suggesting that time spent in diabetes remission after RYGB is not spent in vain when it comes to reducing the risk of subsequent microvascular complications,” they wrote.
The surgery was also associated with a 46% reduction in the incidence of ischemic heart disease. In the first 30 days after surgery, 7.5% of patients were readmitted to hospital for any surgical complication, but the 90-day mortality rate after surgery was less than 0.5%.
The study was supported by the Health Research Fund of Central Denmark, the Novo Nordisk Foundation, and the A.P. Møller Foundation. The authors reported no conflicts of interest.
SOURCE: Madsen LR et al. Diabetologia. 2019, Feb 6. doi: 10.1007/s00125-019-4816-2.
FROM DIABETOLOGIA
Key clinical point: Diabetes remission was achieved in three-quarters of Roux-en-Y surgical patients.
Major finding: The incidence of diabetes remission 1 year after Roux-en-Y gastric bypass was 74%.
Study details: A population-based cohort study in 1,111 individuals with type 2 diabetes mellitus who underwent Roux-en-Y gastric bypass, compared with 1,074 nonsurgical controls with diabetes.
Disclosures: The study was supported by the Health Research Fund of Central Denmark, the Novo Nordisk Foundation, and the A.P. Møller Foundation. The authors reported no conflicts of interest.
Source: Madsen LR et al. Diabetologia. 2019, Feb 6. doi: 10.1007/s00125-019-4816-2.
Clown-tox, tattooed immunity, and cingulum-bundle comedy
Quit clowning around
Would you like a balloon giraffe, elephant, or hypodermic needle? A recently published study examined how the use of “medical clowns” eased the anxiety and pain of children during botulinum toxin injections. These injections are used to treat spasticity in children, and researchers hypothesized that a clown might be an effective distraction.
As anyone who has been to a circus can surmise, the clowns did not perform better than the control distractions.
Researchers concluded that the clowns were appreciated by the parents but not particularly effective on the children. Maybe that’s because the parents weren’t the ones being stuck with needles while some crazy person in clown makeup attempted to distract them. Or maybe they were all just big fans of Stephen King’s “It.”
Inked immunity
Starting to feel a bit under the weather? Head to the local tattoo artist for the cure! Research from the University of Alabama at Birmingham found evidence that the immune systems of heavily tattooed people are stronger than those without, proving once and for all that tats = toughness. Hell's Angels were on to something all along.
While your immune system can actually grow temporarily weaker after one tattoo, multiple tattoos create a stronger immunological response. Researchers tested the immunoglobulin A levels in those getting a first tattoo and those with many tattoos. They found that the latter group had higher IgA levels.
Maybe that’s why Adam Levine showed off his ink at the Super Bowl halftime show – he was just signaling his strong immune system to the rest of us.
Brain surgery is a laughing matter
This certainly came as a surprise to us, but as it turns out, undergoing brain surgery while conscious and awake can be extremely stressful and panic inducing to the patient. We’re sure most people (including us) would prefer to be asleep for their surgery, but sometimes when dealing with the brain, the surgeon needs to be able to talk to the patient to accurately assess their faculties in case they damage something important.
So the question is: How do you keep brain surgery patients from panicking? Why, with the power of laughter, of course! Specifically, a group at Emory University, Atlanta, published a case study in the Journal of Clinical Investigation about their treatment of a patient with moderate anxiety. When the patient woke up from initial anesthesia, she began to panic. However, after electrical stimulation of the cingulum bundle, the patient immediately turned her frown upside down and began laughing and joking with the surgeons.
Sadly, while the team did not report on the quality of the jokes being told, we can only assume the phrase “this isn’t brain surgery” was thrown around multiple times.
A male brain is a terrible thing to waste
In the future, comedy may mean pressing a button to stimulate your cingulum bundle, but for now we still have jokes. One old joke goes like this: Some aliens land on earth and want to learn about humans, so they go into a store to buy some brains. “Why does the male brain cost twice as much as the female brain?” one asks the store owner, who replies, “It’s hardly been used.”
There may be another explanation: Womens’ brains appear to age more slowly than mens’, investigators at Washington University, St. Louis, said in the Proceedings of the National Academy of Sciences.
They performed PET scans on 121 women and 84 men aged 20-82 years to determine the fraction of sugar committed to aerobic glycolysis in various regions of the brain, and then a machine-learning algorithm used those data to calculate metabolic ages.
The womens’ brains were younger than the mens’ brains, with various calculations producing average differences of 2.7-5.3 years, they reported.
The male brain, it seems, is used for something, and after one LOTME staffer spent 5 minutes explaining total quarterback rating (QBR) to his wife, we think we’ve figured out what it is: sports trivia.

Quit clowning around
Would you like a balloon giraffe, elephant, or hypodermic needle? A recently published study examined how the use of “medical clowns” eased the anxiety and pain of children during botulinum toxin injections. These injections are used to treat spasticity in children, and researchers hypothesized that a clown might be an effective distraction.
As anyone who has been to a circus can surmise, the clowns did not perform better than the control distractions.
Researchers concluded that the clowns were appreciated by the parents but not particularly effective on the children. Maybe that’s because the parents weren’t the ones being stuck with needles while some crazy person in clown makeup attempted to distract them. Or maybe they were all just big fans of Stephen King’s “It.”
Inked immunity
Starting to feel a bit under the weather? Head to the local tattoo artist for the cure! Research from the University of Alabama at Birmingham found evidence that the immune systems of heavily tattooed people are stronger than those without, proving once and for all that tats = toughness. Hell's Angels were on to something all along.
While your immune system can actually grow temporarily weaker after one tattoo, multiple tattoos create a stronger immunological response. Researchers tested the immunoglobulin A levels in those getting a first tattoo and those with many tattoos. They found that the latter group had higher IgA levels.
Maybe that’s why Adam Levine showed off his ink at the Super Bowl halftime show – he was just signaling his strong immune system to the rest of us.
Brain surgery is a laughing matter
This certainly came as a surprise to us, but as it turns out, undergoing brain surgery while conscious and awake can be extremely stressful and panic inducing to the patient. We’re sure most people (including us) would prefer to be asleep for their surgery, but sometimes when dealing with the brain, the surgeon needs to be able to talk to the patient to accurately assess their faculties in case they damage something important.
So the question is: How do you keep brain surgery patients from panicking? Why, with the power of laughter, of course! Specifically, a group at Emory University, Atlanta, published a case study in the Journal of Clinical Investigation about their treatment of a patient with moderate anxiety. When the patient woke up from initial anesthesia, she began to panic. However, after electrical stimulation of the cingulum bundle, the patient immediately turned her frown upside down and began laughing and joking with the surgeons.
Sadly, while the team did not report on the quality of the jokes being told, we can only assume the phrase “this isn’t brain surgery” was thrown around multiple times.
A male brain is a terrible thing to waste
In the future, comedy may mean pressing a button to stimulate your cingulum bundle, but for now we still have jokes. One old joke goes like this: Some aliens land on earth and want to learn about humans, so they go into a store to buy some brains. “Why does the male brain cost twice as much as the female brain?” one asks the store owner, who replies, “It’s hardly been used.”
There may be another explanation: Womens’ brains appear to age more slowly than mens’, investigators at Washington University, St. Louis, said in the Proceedings of the National Academy of Sciences.
They performed PET scans on 121 women and 84 men aged 20-82 years to determine the fraction of sugar committed to aerobic glycolysis in various regions of the brain, and then a machine-learning algorithm used those data to calculate metabolic ages.
The womens’ brains were younger than the mens’ brains, with various calculations producing average differences of 2.7-5.3 years, they reported.
The male brain, it seems, is used for something, and after one LOTME staffer spent 5 minutes explaining total quarterback rating (QBR) to his wife, we think we’ve figured out what it is: sports trivia.

Quit clowning around
Would you like a balloon giraffe, elephant, or hypodermic needle? A recently published study examined how the use of “medical clowns” eased the anxiety and pain of children during botulinum toxin injections. These injections are used to treat spasticity in children, and researchers hypothesized that a clown might be an effective distraction.
As anyone who has been to a circus can surmise, the clowns did not perform better than the control distractions.
Researchers concluded that the clowns were appreciated by the parents but not particularly effective on the children. Maybe that’s because the parents weren’t the ones being stuck with needles while some crazy person in clown makeup attempted to distract them. Or maybe they were all just big fans of Stephen King’s “It.”
Inked immunity
Starting to feel a bit under the weather? Head to the local tattoo artist for the cure! Research from the University of Alabama at Birmingham found evidence that the immune systems of heavily tattooed people are stronger than those without, proving once and for all that tats = toughness. Hell's Angels were on to something all along.
While your immune system can actually grow temporarily weaker after one tattoo, multiple tattoos create a stronger immunological response. Researchers tested the immunoglobulin A levels in those getting a first tattoo and those with many tattoos. They found that the latter group had higher IgA levels.
Maybe that’s why Adam Levine showed off his ink at the Super Bowl halftime show – he was just signaling his strong immune system to the rest of us.
Brain surgery is a laughing matter
This certainly came as a surprise to us, but as it turns out, undergoing brain surgery while conscious and awake can be extremely stressful and panic inducing to the patient. We’re sure most people (including us) would prefer to be asleep for their surgery, but sometimes when dealing with the brain, the surgeon needs to be able to talk to the patient to accurately assess their faculties in case they damage something important.
So the question is: How do you keep brain surgery patients from panicking? Why, with the power of laughter, of course! Specifically, a group at Emory University, Atlanta, published a case study in the Journal of Clinical Investigation about their treatment of a patient with moderate anxiety. When the patient woke up from initial anesthesia, she began to panic. However, after electrical stimulation of the cingulum bundle, the patient immediately turned her frown upside down and began laughing and joking with the surgeons.
Sadly, while the team did not report on the quality of the jokes being told, we can only assume the phrase “this isn’t brain surgery” was thrown around multiple times.
A male brain is a terrible thing to waste
In the future, comedy may mean pressing a button to stimulate your cingulum bundle, but for now we still have jokes. One old joke goes like this: Some aliens land on earth and want to learn about humans, so they go into a store to buy some brains. “Why does the male brain cost twice as much as the female brain?” one asks the store owner, who replies, “It’s hardly been used.”
There may be another explanation: Womens’ brains appear to age more slowly than mens’, investigators at Washington University, St. Louis, said in the Proceedings of the National Academy of Sciences.
They performed PET scans on 121 women and 84 men aged 20-82 years to determine the fraction of sugar committed to aerobic glycolysis in various regions of the brain, and then a machine-learning algorithm used those data to calculate metabolic ages.
The womens’ brains were younger than the mens’ brains, with various calculations producing average differences of 2.7-5.3 years, they reported.
The male brain, it seems, is used for something, and after one LOTME staffer spent 5 minutes explaining total quarterback rating (QBR) to his wife, we think we’ve figured out what it is: sports trivia.

Cloud of inconsistency hangs over cannabis data
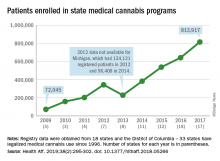
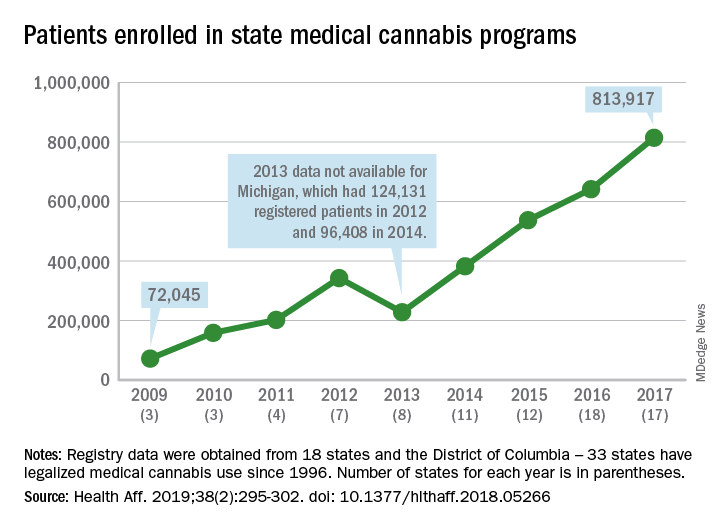
More people are using medical cannabis as it becomes legal in more states, but the lack of standardization in states’ data collection hindered investigators’ efforts to track that use.
Legalized medical cannabis is now available in 33 states and the District of Columbia, and the number of users has risen from just over 72,000 in 2009 to almost 814,000 in 2017. That 814,000, however, covers only 16 states and D.C., since 1 state (Connecticut) does not publish reports on medical cannabis use, 12 did not have statistics available, 2 (New York and Vermont) didn’t report data for 2017, and 2 (California and Maine) have voluntary registries that are unlikely to be accurate, according to Kevin F. Boehnke, PhD, of the University of Michigan, Ann Arbor, and his associates.
Michigan had the largest reported number of patients enrolled in its medical cannabis program in 2017, almost 270,000. California – the state with the oldest medical cannabis legislation (passed in 1996) and the largest overall population but a voluntary cannabis registry – reported its highest number of enrollees, 12,659, in 2009-2010, the investigators said. Colorado had more than 116,000 patients in its medical cannabis program in 2010 (Health Aff. 2019;38[2]:295-302).
The “many inconsistencies in data quality across states [suggest] the need for further standardization of data collection. Such standardization would add transparency to understanding how medical cannabis programs are used, which would help guide both research and policy needs,” Dr. Boehnke and his associates wrote.
More consistency was seen in the reasons for using medical cannabis. Chronic pain made up 62.2% of all qualifying conditions reported by patients during 1999-2016, with the annual average varying between 33.3% and 73%. Multiple sclerosis spasticity symptoms had the second-highest number of reports over the study period, followed by chemotherapy-induced nausea and vomiting, posttraumatic stress disorder, and cancer, they reported.
The investigators also looked at the appropriateness of cannabis and determined that its use in 85.5% of patient-reported conditions was “supported by conclusive or substantial evidence of therapeutic effectiveness, according to the 2017 National Academies report” on the health effects of cannabis.
“We believe not only that it is inappropriate for cannabis to remain a Schedule I substance, but also that state and federal policy makers should begin evaluating evidence-based ways for safely integrating cannabis research and products into the health care system,” they concluded.
SOURCE: Boehnke KF et al. Health Aff. 2019;38(2):295-302.
More people are using medical cannabis as it becomes legal in more states, but the lack of standardization in states’ data collection hindered investigators’ efforts to track that use.
Legalized medical cannabis is now available in 33 states and the District of Columbia, and the number of users has risen from just over 72,000 in 2009 to almost 814,000 in 2017. That 814,000, however, covers only 16 states and D.C., since 1 state (Connecticut) does not publish reports on medical cannabis use, 12 did not have statistics available, 2 (New York and Vermont) didn’t report data for 2017, and 2 (California and Maine) have voluntary registries that are unlikely to be accurate, according to Kevin F. Boehnke, PhD, of the University of Michigan, Ann Arbor, and his associates.
Michigan had the largest reported number of patients enrolled in its medical cannabis program in 2017, almost 270,000. California – the state with the oldest medical cannabis legislation (passed in 1996) and the largest overall population but a voluntary cannabis registry – reported its highest number of enrollees, 12,659, in 2009-2010, the investigators said. Colorado had more than 116,000 patients in its medical cannabis program in 2010 (Health Aff. 2019;38[2]:295-302).
The “many inconsistencies in data quality across states [suggest] the need for further standardization of data collection. Such standardization would add transparency to understanding how medical cannabis programs are used, which would help guide both research and policy needs,” Dr. Boehnke and his associates wrote.
More consistency was seen in the reasons for using medical cannabis. Chronic pain made up 62.2% of all qualifying conditions reported by patients during 1999-2016, with the annual average varying between 33.3% and 73%. Multiple sclerosis spasticity symptoms had the second-highest number of reports over the study period, followed by chemotherapy-induced nausea and vomiting, posttraumatic stress disorder, and cancer, they reported.
The investigators also looked at the appropriateness of cannabis and determined that its use in 85.5% of patient-reported conditions was “supported by conclusive or substantial evidence of therapeutic effectiveness, according to the 2017 National Academies report” on the health effects of cannabis.
“We believe not only that it is inappropriate for cannabis to remain a Schedule I substance, but also that state and federal policy makers should begin evaluating evidence-based ways for safely integrating cannabis research and products into the health care system,” they concluded.
SOURCE: Boehnke KF et al. Health Aff. 2019;38(2):295-302.
More people are using medical cannabis as it becomes legal in more states, but the lack of standardization in states’ data collection hindered investigators’ efforts to track that use.
Legalized medical cannabis is now available in 33 states and the District of Columbia, and the number of users has risen from just over 72,000 in 2009 to almost 814,000 in 2017. That 814,000, however, covers only 16 states and D.C., since 1 state (Connecticut) does not publish reports on medical cannabis use, 12 did not have statistics available, 2 (New York and Vermont) didn’t report data for 2017, and 2 (California and Maine) have voluntary registries that are unlikely to be accurate, according to Kevin F. Boehnke, PhD, of the University of Michigan, Ann Arbor, and his associates.
Michigan had the largest reported number of patients enrolled in its medical cannabis program in 2017, almost 270,000. California – the state with the oldest medical cannabis legislation (passed in 1996) and the largest overall population but a voluntary cannabis registry – reported its highest number of enrollees, 12,659, in 2009-2010, the investigators said. Colorado had more than 116,000 patients in its medical cannabis program in 2010 (Health Aff. 2019;38[2]:295-302).
The “many inconsistencies in data quality across states [suggest] the need for further standardization of data collection. Such standardization would add transparency to understanding how medical cannabis programs are used, which would help guide both research and policy needs,” Dr. Boehnke and his associates wrote.
More consistency was seen in the reasons for using medical cannabis. Chronic pain made up 62.2% of all qualifying conditions reported by patients during 1999-2016, with the annual average varying between 33.3% and 73%. Multiple sclerosis spasticity symptoms had the second-highest number of reports over the study period, followed by chemotherapy-induced nausea and vomiting, posttraumatic stress disorder, and cancer, they reported.
The investigators also looked at the appropriateness of cannabis and determined that its use in 85.5% of patient-reported conditions was “supported by conclusive or substantial evidence of therapeutic effectiveness, according to the 2017 National Academies report” on the health effects of cannabis.
“We believe not only that it is inappropriate for cannabis to remain a Schedule I substance, but also that state and federal policy makers should begin evaluating evidence-based ways for safely integrating cannabis research and products into the health care system,” they concluded.
SOURCE: Boehnke KF et al. Health Aff. 2019;38(2):295-302.
FROM HEALTH AFFAIRS