User login
VIDEO: How to prepare PTCL patients for transplant
LA JOLLA, CALIF. – according to Steven M. Horwitz, MD, of Memorial Sloan Kettering Cancer Center, New York.
“If you’re really trying to go to transplant, you want a complete remission or close to it. So that’s often been combination chemotherapy. But I think what we’re learning is, when some of the newer agents are combined, we’re seeing higher complete response rates. And we’re doing a better job at picking subtype specific approaches,” Dr. Horwitz said in a video interview at the annual T-cell Lymphoma Forum.
Dr. Horwitz also explored the role for reduced-intensity regimens in older patients, the use of radiation conditioning, and which new agents look most promising in peripheral T-cell lymphoma.
Dr. Horwitz had previously disclosed financial relationships with Celgene, Forty Seven, Huya Bioscience International, Infinity, Kyowa Hakko Kirin, Millennium, Seattle Genetics, and Takeda. The T-Cell Lymphoma Forum is held by Jonathan Wood & Associates, which is owned by the same company as this news organization.
SOURCE: Horwitz SM. TCLF 2018.
LA JOLLA, CALIF. – according to Steven M. Horwitz, MD, of Memorial Sloan Kettering Cancer Center, New York.
“If you’re really trying to go to transplant, you want a complete remission or close to it. So that’s often been combination chemotherapy. But I think what we’re learning is, when some of the newer agents are combined, we’re seeing higher complete response rates. And we’re doing a better job at picking subtype specific approaches,” Dr. Horwitz said in a video interview at the annual T-cell Lymphoma Forum.
Dr. Horwitz also explored the role for reduced-intensity regimens in older patients, the use of radiation conditioning, and which new agents look most promising in peripheral T-cell lymphoma.
Dr. Horwitz had previously disclosed financial relationships with Celgene, Forty Seven, Huya Bioscience International, Infinity, Kyowa Hakko Kirin, Millennium, Seattle Genetics, and Takeda. The T-Cell Lymphoma Forum is held by Jonathan Wood & Associates, which is owned by the same company as this news organization.
SOURCE: Horwitz SM. TCLF 2018.
LA JOLLA, CALIF. – according to Steven M. Horwitz, MD, of Memorial Sloan Kettering Cancer Center, New York.
“If you’re really trying to go to transplant, you want a complete remission or close to it. So that’s often been combination chemotherapy. But I think what we’re learning is, when some of the newer agents are combined, we’re seeing higher complete response rates. And we’re doing a better job at picking subtype specific approaches,” Dr. Horwitz said in a video interview at the annual T-cell Lymphoma Forum.
Dr. Horwitz also explored the role for reduced-intensity regimens in older patients, the use of radiation conditioning, and which new agents look most promising in peripheral T-cell lymphoma.
Dr. Horwitz had previously disclosed financial relationships with Celgene, Forty Seven, Huya Bioscience International, Infinity, Kyowa Hakko Kirin, Millennium, Seattle Genetics, and Takeda. The T-Cell Lymphoma Forum is held by Jonathan Wood & Associates, which is owned by the same company as this news organization.
SOURCE: Horwitz SM. TCLF 2018.
REPORTING FROM TCLF 2018
High adverse events with TB prevention in HIV-infected pregnant women
BOSTON – World Health Organization guidelines on the use of isoniazid to prevent tuberculosis in HIV-infected women during pregnancy may need to be reconsidered in light of new evidence that isoniazid preventive therapy (IPT) is associated with a high risk for adverse pregnancy events, investigators say.
Among 156 HIV-infected pregnant women, the rate of adverse pregnancy outcomes was 23% for those randomly assigned to immediate IPT during pregnancy versus 17% for women randomized to IPT that was delayed until 12 weeks postpartum, a significant difference (P = .009), reported Amita Gupta, MD, from Johns Hopkins University in Baltimore.
There were six maternal deaths: two in the immediate therapy arm and four in the delayed arm. Two of the deaths were related to isoniazid-induced liver failure, and two other instances of liver failure were from other causes. The two remaining deaths were deemed unrelated to isoniazid: One woman died from bacterial sepsis, and the other from pneumonia.
The incidence of TB infections was low in both study arms, Dr. Gupta noted.
“So should we really be prioritizing IPT in pregnancy or give women the choice to know what the facts are and to select if they feel that they want to take on IPT during pregnancy or defer it until after they deliver?” she said at a briefing following her presentation of the data in an oral abstract session.
WHO guidelines for management of latent tuberculosis infections state that “[a]s isoniazid and rifampicin, the drugs commonly used in preventive treatment, are safe for use in pregnant women, pregnancy should not disqualify women living with HIV from receiving preventive treatment. Nevertheless, sound clinical judgment is required to determine the best time to provide it.”
However, the quality of evidence to support that statement is weak, primarily because pregnant women were typically excluded from IPT or other tuberculosis-prevention trials. In addition, isoniazid has been associated in retrospective studies with increased hepatotoxicity in women both during pregnancy and in the postpartum period, Dr. Gupta pointed out.
She and her colleagues conducted the phase 4 IMPAACT P1078 trial to test the hypothesis that IPT can be initiated safely during pregnancy. The study was conducted at centers in Botswana, Haiti, India, South Africa, Tanzania, Thailand, Uganda, and Zimbabwe.
HIV-infected pregnant women from 14 through 34 weeks of gestation who live in a high TB burden area (prevalence of 60 cases or more per 100,000 population) but had no evidence of TB infection were randomly assigned on a 1:1 basis to receive either immediate therapy with isoniazid 300 mg daily for 28 weeks, followed by placebo, or to the same dose of isoniazid started 12 weeks postpartum for 28 weeks. All patients also received vitamin B6 and a prenatal multivitamin until 40 weeks postpartum.
The participants were stratified by gestational age (14 to less than 24 weeks and 24 through 34 weeks). Women were excluded if they were suspected of having active TB, reported recent exposure, or had received TB treatment for more than 30 days in the past year. Women with recent acute hepatitis or peripheral neuropathy were excluded.
All women and their infants received the local standard of care for HIV. The investigators performed intensified TB case finding by using the WHO symptoms screening and exam, monitoring of signs and symptoms, conducting liver function tests, and screening for peripheral neuropathy.
“We had higher than expected adverse events in the study, but there was no statistical difference between arms,” she said.
In an intention-to-treat analysis, the rate of first maternal treatment-related grade 3 or greater adverse event or permanent drug discontinuation caused by toxicity (the primary endpoint) was 15.5% in the immediate IPT arm and 15.2% in the delayed IPT arm, a nonsignificant difference. The immediate therapy arm approached but did not quite reach the prespecified boundary of noninferiority, Dr. Gupta noted.
The per-protocol analysis of the primary endpoint was similar, at 17.6% vs. 17.8%, respectively.
In the intention-to-treat analysis, any-cause grade 3 or greater maternal adverse events were seen in 30.5% of women in the immediate arm versus 28.4% in the delayed arm, with an incidence-rate difference of 4.2 per 100 person-years, which did not reach the noninferiority boundary.
The respective rates in the per-protocol analysis were 33% vs. 30.4%, for an incidence-rate difference of 4.3 per 100 person-years.
In both groups, elevated liver enzymes and weight loss were the most common maternal adverse events.
All cause hepatotoxicity occurred in 6% of participants in the immediate arm and 7% in the deferred. Rates of permanent discontinuation because of toxicity were 4% and 6%, respectively, along with the two women in the immediate IPT arm and four in the delayed arm who died during the study. There were no significant differences in these outcomes between the groups.
Dr. Gupta noted that higher liver function test results were seen after delivery, regardless of whether the women were on IPT or what type of antiretroviral therapy they were receiving.
There were no significant differences seen in infant safety by treatment arm, a secondary endpoint.
“What did we learn? We learned that we had higher than expected adverse events that were at least possibly attributed to IPT in both arms. We did not reach our noninferiority margin partly because of the high rates, but we also did not find any major significant differences between the immediate and the deferred arm in terms of maternal safety when it was looked [at] by itself,” she said.
Although there were no significant differences in any maternal hepatotoxicity, grade 3 or greater infant adverse events, or maternal or infant death by treatment arm, “we did find an important distinction in terms of difference by adverse pregnancy outcomes, where immediate IPT was associated with more adverse pregnancy outcomes,” she said,
“I think we now need to reweigh the evidence for pros and cons for IPT in pregnancy,” Dr. Gupta concluded.
The study was sponsored the National Institutes of Health. Dr. Gupta reported having nothing to disclose.
SOURCE: Gupta A et al. CROI 2018, Abstract Number 142LB.
BOSTON – World Health Organization guidelines on the use of isoniazid to prevent tuberculosis in HIV-infected women during pregnancy may need to be reconsidered in light of new evidence that isoniazid preventive therapy (IPT) is associated with a high risk for adverse pregnancy events, investigators say.
Among 156 HIV-infected pregnant women, the rate of adverse pregnancy outcomes was 23% for those randomly assigned to immediate IPT during pregnancy versus 17% for women randomized to IPT that was delayed until 12 weeks postpartum, a significant difference (P = .009), reported Amita Gupta, MD, from Johns Hopkins University in Baltimore.
There were six maternal deaths: two in the immediate therapy arm and four in the delayed arm. Two of the deaths were related to isoniazid-induced liver failure, and two other instances of liver failure were from other causes. The two remaining deaths were deemed unrelated to isoniazid: One woman died from bacterial sepsis, and the other from pneumonia.
The incidence of TB infections was low in both study arms, Dr. Gupta noted.
“So should we really be prioritizing IPT in pregnancy or give women the choice to know what the facts are and to select if they feel that they want to take on IPT during pregnancy or defer it until after they deliver?” she said at a briefing following her presentation of the data in an oral abstract session.
WHO guidelines for management of latent tuberculosis infections state that “[a]s isoniazid and rifampicin, the drugs commonly used in preventive treatment, are safe for use in pregnant women, pregnancy should not disqualify women living with HIV from receiving preventive treatment. Nevertheless, sound clinical judgment is required to determine the best time to provide it.”
However, the quality of evidence to support that statement is weak, primarily because pregnant women were typically excluded from IPT or other tuberculosis-prevention trials. In addition, isoniazid has been associated in retrospective studies with increased hepatotoxicity in women both during pregnancy and in the postpartum period, Dr. Gupta pointed out.
She and her colleagues conducted the phase 4 IMPAACT P1078 trial to test the hypothesis that IPT can be initiated safely during pregnancy. The study was conducted at centers in Botswana, Haiti, India, South Africa, Tanzania, Thailand, Uganda, and Zimbabwe.
HIV-infected pregnant women from 14 through 34 weeks of gestation who live in a high TB burden area (prevalence of 60 cases or more per 100,000 population) but had no evidence of TB infection were randomly assigned on a 1:1 basis to receive either immediate therapy with isoniazid 300 mg daily for 28 weeks, followed by placebo, or to the same dose of isoniazid started 12 weeks postpartum for 28 weeks. All patients also received vitamin B6 and a prenatal multivitamin until 40 weeks postpartum.
The participants were stratified by gestational age (14 to less than 24 weeks and 24 through 34 weeks). Women were excluded if they were suspected of having active TB, reported recent exposure, or had received TB treatment for more than 30 days in the past year. Women with recent acute hepatitis or peripheral neuropathy were excluded.
All women and their infants received the local standard of care for HIV. The investigators performed intensified TB case finding by using the WHO symptoms screening and exam, monitoring of signs and symptoms, conducting liver function tests, and screening for peripheral neuropathy.
“We had higher than expected adverse events in the study, but there was no statistical difference between arms,” she said.
In an intention-to-treat analysis, the rate of first maternal treatment-related grade 3 or greater adverse event or permanent drug discontinuation caused by toxicity (the primary endpoint) was 15.5% in the immediate IPT arm and 15.2% in the delayed IPT arm, a nonsignificant difference. The immediate therapy arm approached but did not quite reach the prespecified boundary of noninferiority, Dr. Gupta noted.
The per-protocol analysis of the primary endpoint was similar, at 17.6% vs. 17.8%, respectively.
In the intention-to-treat analysis, any-cause grade 3 or greater maternal adverse events were seen in 30.5% of women in the immediate arm versus 28.4% in the delayed arm, with an incidence-rate difference of 4.2 per 100 person-years, which did not reach the noninferiority boundary.
The respective rates in the per-protocol analysis were 33% vs. 30.4%, for an incidence-rate difference of 4.3 per 100 person-years.
In both groups, elevated liver enzymes and weight loss were the most common maternal adverse events.
All cause hepatotoxicity occurred in 6% of participants in the immediate arm and 7% in the deferred. Rates of permanent discontinuation because of toxicity were 4% and 6%, respectively, along with the two women in the immediate IPT arm and four in the delayed arm who died during the study. There were no significant differences in these outcomes between the groups.
Dr. Gupta noted that higher liver function test results were seen after delivery, regardless of whether the women were on IPT or what type of antiretroviral therapy they were receiving.
There were no significant differences seen in infant safety by treatment arm, a secondary endpoint.
“What did we learn? We learned that we had higher than expected adverse events that were at least possibly attributed to IPT in both arms. We did not reach our noninferiority margin partly because of the high rates, but we also did not find any major significant differences between the immediate and the deferred arm in terms of maternal safety when it was looked [at] by itself,” she said.
Although there were no significant differences in any maternal hepatotoxicity, grade 3 or greater infant adverse events, or maternal or infant death by treatment arm, “we did find an important distinction in terms of difference by adverse pregnancy outcomes, where immediate IPT was associated with more adverse pregnancy outcomes,” she said,
“I think we now need to reweigh the evidence for pros and cons for IPT in pregnancy,” Dr. Gupta concluded.
The study was sponsored the National Institutes of Health. Dr. Gupta reported having nothing to disclose.
SOURCE: Gupta A et al. CROI 2018, Abstract Number 142LB.
BOSTON – World Health Organization guidelines on the use of isoniazid to prevent tuberculosis in HIV-infected women during pregnancy may need to be reconsidered in light of new evidence that isoniazid preventive therapy (IPT) is associated with a high risk for adverse pregnancy events, investigators say.
Among 156 HIV-infected pregnant women, the rate of adverse pregnancy outcomes was 23% for those randomly assigned to immediate IPT during pregnancy versus 17% for women randomized to IPT that was delayed until 12 weeks postpartum, a significant difference (P = .009), reported Amita Gupta, MD, from Johns Hopkins University in Baltimore.
There were six maternal deaths: two in the immediate therapy arm and four in the delayed arm. Two of the deaths were related to isoniazid-induced liver failure, and two other instances of liver failure were from other causes. The two remaining deaths were deemed unrelated to isoniazid: One woman died from bacterial sepsis, and the other from pneumonia.
The incidence of TB infections was low in both study arms, Dr. Gupta noted.
“So should we really be prioritizing IPT in pregnancy or give women the choice to know what the facts are and to select if they feel that they want to take on IPT during pregnancy or defer it until after they deliver?” she said at a briefing following her presentation of the data in an oral abstract session.
WHO guidelines for management of latent tuberculosis infections state that “[a]s isoniazid and rifampicin, the drugs commonly used in preventive treatment, are safe for use in pregnant women, pregnancy should not disqualify women living with HIV from receiving preventive treatment. Nevertheless, sound clinical judgment is required to determine the best time to provide it.”
However, the quality of evidence to support that statement is weak, primarily because pregnant women were typically excluded from IPT or other tuberculosis-prevention trials. In addition, isoniazid has been associated in retrospective studies with increased hepatotoxicity in women both during pregnancy and in the postpartum period, Dr. Gupta pointed out.
She and her colleagues conducted the phase 4 IMPAACT P1078 trial to test the hypothesis that IPT can be initiated safely during pregnancy. The study was conducted at centers in Botswana, Haiti, India, South Africa, Tanzania, Thailand, Uganda, and Zimbabwe.
HIV-infected pregnant women from 14 through 34 weeks of gestation who live in a high TB burden area (prevalence of 60 cases or more per 100,000 population) but had no evidence of TB infection were randomly assigned on a 1:1 basis to receive either immediate therapy with isoniazid 300 mg daily for 28 weeks, followed by placebo, or to the same dose of isoniazid started 12 weeks postpartum for 28 weeks. All patients also received vitamin B6 and a prenatal multivitamin until 40 weeks postpartum.
The participants were stratified by gestational age (14 to less than 24 weeks and 24 through 34 weeks). Women were excluded if they were suspected of having active TB, reported recent exposure, or had received TB treatment for more than 30 days in the past year. Women with recent acute hepatitis or peripheral neuropathy were excluded.
All women and their infants received the local standard of care for HIV. The investigators performed intensified TB case finding by using the WHO symptoms screening and exam, monitoring of signs and symptoms, conducting liver function tests, and screening for peripheral neuropathy.
“We had higher than expected adverse events in the study, but there was no statistical difference between arms,” she said.
In an intention-to-treat analysis, the rate of first maternal treatment-related grade 3 or greater adverse event or permanent drug discontinuation caused by toxicity (the primary endpoint) was 15.5% in the immediate IPT arm and 15.2% in the delayed IPT arm, a nonsignificant difference. The immediate therapy arm approached but did not quite reach the prespecified boundary of noninferiority, Dr. Gupta noted.
The per-protocol analysis of the primary endpoint was similar, at 17.6% vs. 17.8%, respectively.
In the intention-to-treat analysis, any-cause grade 3 or greater maternal adverse events were seen in 30.5% of women in the immediate arm versus 28.4% in the delayed arm, with an incidence-rate difference of 4.2 per 100 person-years, which did not reach the noninferiority boundary.
The respective rates in the per-protocol analysis were 33% vs. 30.4%, for an incidence-rate difference of 4.3 per 100 person-years.
In both groups, elevated liver enzymes and weight loss were the most common maternal adverse events.
All cause hepatotoxicity occurred in 6% of participants in the immediate arm and 7% in the deferred. Rates of permanent discontinuation because of toxicity were 4% and 6%, respectively, along with the two women in the immediate IPT arm and four in the delayed arm who died during the study. There were no significant differences in these outcomes between the groups.
Dr. Gupta noted that higher liver function test results were seen after delivery, regardless of whether the women were on IPT or what type of antiretroviral therapy they were receiving.
There were no significant differences seen in infant safety by treatment arm, a secondary endpoint.
“What did we learn? We learned that we had higher than expected adverse events that were at least possibly attributed to IPT in both arms. We did not reach our noninferiority margin partly because of the high rates, but we also did not find any major significant differences between the immediate and the deferred arm in terms of maternal safety when it was looked [at] by itself,” she said.
Although there were no significant differences in any maternal hepatotoxicity, grade 3 or greater infant adverse events, or maternal or infant death by treatment arm, “we did find an important distinction in terms of difference by adverse pregnancy outcomes, where immediate IPT was associated with more adverse pregnancy outcomes,” she said,
“I think we now need to reweigh the evidence for pros and cons for IPT in pregnancy,” Dr. Gupta concluded.
The study was sponsored the National Institutes of Health. Dr. Gupta reported having nothing to disclose.
SOURCE: Gupta A et al. CROI 2018, Abstract Number 142LB.
FROM CROI 2018
Key clinical point: Immediate tuberculosis prevention therapy with isoniazid (IPT) during pregnancy was associated with more adverse pregnancy outcomes than delayed IPT.
Major finding: The rate of adverse pregnancy outcomes was 23% with immediate IPT during pregnancy versus 17% for IPT delayed until 12 weeks postpartum (P =. 009).
Data source: Randomized trial in 156 HIV-infected pregnant women in seven countries that have high TB prevalence rates.
Disclosures: The study was sponsored by the U.S. National Institutes of Health. Dr. Gupta reported having nothing to disclose.
Source: Gupta A et al. CROI 2018, Abstract Number 142LB.
Why isn’t smart gun technology on Parkland activists’ agenda?
The evening before the March For Our Lives rally in Washington, I was hard at work on my sign. Making a statement is important, but at the Women’s March last January, I discovered that a sign was invaluable for keeping friends together in a crowd. Since my Women’s March sign read “Make America Kind Again,” I opted to keep with the theme, and I created a “Make America Safe Again” sign for gun control. My daughter looked at my sign skeptically. “It’s too vague and nondirective,” she declared. Now challenged, I added the following directives: “Universal Background Checks,” “Ban Assault Weapons,” and “Smart Guns.” My sign was now complete.
I was surprised when my daughter – our family Jeopardy! whiz – asked, “What’s a smart gun?” When we arrived to join friends, their daughter – a doctoral student – also looked at the sign and asked me what a smart gun is. I explained to both young women that a smart gun – like an iPhone – relies on a biometric such as a fingerprint so that it can be used only by authorized users. This technology would reduce the flow of firearms to illegal owners, prevent accidental discharge by children, protect rightful owners and law enforcement officers from having their own weapons used against them by criminals, and decrease the number of suicides by family members of gun owners. In my state of Maryland, there have been two school shootings, one as recently as March 20, and both were committed by boys who took a parent’s firearm to school.
We arrived at the rally and found a spot in front of the Newseum, with two jumbotrons in sight, right in the middle of the crowd. The young people who spoke were inspirational. Regardless of one’s political views or thoughts on gun control, they were bright, articulate, fearless, passionate, and determined. The show was stolen by Naomi Wadler, a precocious 11-year-old girl who is destined to be a member of Congress (if not president) one day, and by Dr. Martin Luther King Jr.’s 9-year-old granddaughter, Yolanda Renee King.
The rest of the speakers were teenagers, all of whom had lost someone to gunfire. There were speakers from Parkland, Fla., who talked about the raw losses they were feeling, and the student who wasn’t celebrating his 18th birthday on March 24. Matthew Soto from Newtown, Conn., talked about losing his sister, a first-grade teacher, in the Sandy Hook shooting.
But the rally wasn’t just about mass murders and school shootings: Zion Kelly talked about the death of his twin, a teen who was killed when he stopped at a convenience store on the way home from school in Washington, only to have his promising life snuffed out during a holdup. We heard about shootings in the bullet-riddled streets of Chicago and Los Angeles and the toll they have taken. The losses to gun violence have been felt most strongly in communities of color. While it was mentioned that the majority of gun deaths are suicides, there were no speakers from families of people who died from suicide. The final speaker was Emma Gonzalez, a young woman from Parkland who took the stage for 6 minutes and 20 seconds - the duration of the shooter’s rampage - and closed with a powerful moment of silence.* The rally closed with a performance of Bob Dylan’s “The Times They Are a-Changin’ ” by Jennifer Hudson, whose mother, brother, and nephew were murdered by a gunman.
The teens talked about what changes they wanted to see enacted. They talked about closing loopholes to background checks, and I was pleased that one young man specifically mentioned keeping guns from those who are violent, but did not mention mental illness as a reason to block gun ownership. A call to resume a ban on assault weapons was made repeatedly as was a call to raise the age (to 21) at which an individual can purchase a gun. Military-style weapons are not necessary for hunting or self-defense, and they enable the rapid-fire assassinations we have seen in mass shootings, so they remain an easy target of gun control advocacy. But in terms of numbers, these firearms are responsible for a small fraction of gun deaths. I was surprised, but the teens did not mention “smart gun” legislation as a way to reduce gun deaths.
Guns, and gun deaths, are a part of American society. While the majority of Americans favor stronger regulation of gun ownership, legislation that would end gun ownership is not likely to go anywhere. Smart guns, however, are different. Forty percent of polled gun owners have said they would swap their firearm for a smart firearm. So given the appeal of a firearm that can’t be diverted or stolen, used against the owner, discharged accidentally by a child, or used for suicide or homicide by a distressed family member, why don’t these weapons exist for use by the American people? . People want these guns and the protections they offer, yet they have never been produced and made available to either the American public or to our law enforcement officers.
So where are these firearms, and why aren’t our Parkland teens asking for them? The answer to the first part of that question lies with the National Rifle Association (NRA) and the state of New Jersey. In 2003, New Jersey passed the Childproof Handgun bill, which requires that all guns sold in the state be smart guns within 3 years of their availability. The NRA has vigorously opposed any legislation that would require all guns to be smart guns. Because the availability of these weapons would trigger the New Jersey bill, California and Maryland have been prevented from importing smart firearms from a German company. Perhaps, however, New Jersey does not need to bear all the blame; in 1999, 4 years before the passage of the bill, the NRA and its members boycotted Smith & Wesson when the gun manufacturer revealed plans to develop a smart gun for the government. The NRA’s public stance is that it does not oppose smart guns for those who want them, but it opposes legislation that would eliminate the sale of conventional firearms. The organization has voiced concerns that technology fails and that it potentially slows down firing the weapon. It doesn’t talk about dead toddlers, or about police officers who’ve been killed when their weapons were taken from them.
As for the Parkland students, I don’t know why they aren’t asking for smart gun technology while they have the attention of the country. Perhaps they, like the young women I was with, don’t know it’s an option. Perhaps it’s too removed from the issue of mass murders and an assault weapon ban feels more attainable. Or perhaps the NRA’s mission has too much of a stronghold in Florida. I don’t know why they aren’t asking for smart gun production, but I know they should be.
*Correction, 3/27/2018: An earlier version of this story misstated the duration of Emma Gonzalez's moment of silence.
Dr. Miller is coauthor with Annette Hanson, MD, of “Committed: The Battle Over Involuntary Psychiatric Care” (Baltimore: Johns Hopkins University Press), 2016. She practices in Baltimore.
The evening before the March For Our Lives rally in Washington, I was hard at work on my sign. Making a statement is important, but at the Women’s March last January, I discovered that a sign was invaluable for keeping friends together in a crowd. Since my Women’s March sign read “Make America Kind Again,” I opted to keep with the theme, and I created a “Make America Safe Again” sign for gun control. My daughter looked at my sign skeptically. “It’s too vague and nondirective,” she declared. Now challenged, I added the following directives: “Universal Background Checks,” “Ban Assault Weapons,” and “Smart Guns.” My sign was now complete.
I was surprised when my daughter – our family Jeopardy! whiz – asked, “What’s a smart gun?” When we arrived to join friends, their daughter – a doctoral student – also looked at the sign and asked me what a smart gun is. I explained to both young women that a smart gun – like an iPhone – relies on a biometric such as a fingerprint so that it can be used only by authorized users. This technology would reduce the flow of firearms to illegal owners, prevent accidental discharge by children, protect rightful owners and law enforcement officers from having their own weapons used against them by criminals, and decrease the number of suicides by family members of gun owners. In my state of Maryland, there have been two school shootings, one as recently as March 20, and both were committed by boys who took a parent’s firearm to school.
We arrived at the rally and found a spot in front of the Newseum, with two jumbotrons in sight, right in the middle of the crowd. The young people who spoke were inspirational. Regardless of one’s political views or thoughts on gun control, they were bright, articulate, fearless, passionate, and determined. The show was stolen by Naomi Wadler, a precocious 11-year-old girl who is destined to be a member of Congress (if not president) one day, and by Dr. Martin Luther King Jr.’s 9-year-old granddaughter, Yolanda Renee King.
The rest of the speakers were teenagers, all of whom had lost someone to gunfire. There were speakers from Parkland, Fla., who talked about the raw losses they were feeling, and the student who wasn’t celebrating his 18th birthday on March 24. Matthew Soto from Newtown, Conn., talked about losing his sister, a first-grade teacher, in the Sandy Hook shooting.
But the rally wasn’t just about mass murders and school shootings: Zion Kelly talked about the death of his twin, a teen who was killed when he stopped at a convenience store on the way home from school in Washington, only to have his promising life snuffed out during a holdup. We heard about shootings in the bullet-riddled streets of Chicago and Los Angeles and the toll they have taken. The losses to gun violence have been felt most strongly in communities of color. While it was mentioned that the majority of gun deaths are suicides, there were no speakers from families of people who died from suicide. The final speaker was Emma Gonzalez, a young woman from Parkland who took the stage for 6 minutes and 20 seconds - the duration of the shooter’s rampage - and closed with a powerful moment of silence.* The rally closed with a performance of Bob Dylan’s “The Times They Are a-Changin’ ” by Jennifer Hudson, whose mother, brother, and nephew were murdered by a gunman.
The teens talked about what changes they wanted to see enacted. They talked about closing loopholes to background checks, and I was pleased that one young man specifically mentioned keeping guns from those who are violent, but did not mention mental illness as a reason to block gun ownership. A call to resume a ban on assault weapons was made repeatedly as was a call to raise the age (to 21) at which an individual can purchase a gun. Military-style weapons are not necessary for hunting or self-defense, and they enable the rapid-fire assassinations we have seen in mass shootings, so they remain an easy target of gun control advocacy. But in terms of numbers, these firearms are responsible for a small fraction of gun deaths. I was surprised, but the teens did not mention “smart gun” legislation as a way to reduce gun deaths.
Guns, and gun deaths, are a part of American society. While the majority of Americans favor stronger regulation of gun ownership, legislation that would end gun ownership is not likely to go anywhere. Smart guns, however, are different. Forty percent of polled gun owners have said they would swap their firearm for a smart firearm. So given the appeal of a firearm that can’t be diverted or stolen, used against the owner, discharged accidentally by a child, or used for suicide or homicide by a distressed family member, why don’t these weapons exist for use by the American people? . People want these guns and the protections they offer, yet they have never been produced and made available to either the American public or to our law enforcement officers.
So where are these firearms, and why aren’t our Parkland teens asking for them? The answer to the first part of that question lies with the National Rifle Association (NRA) and the state of New Jersey. In 2003, New Jersey passed the Childproof Handgun bill, which requires that all guns sold in the state be smart guns within 3 years of their availability. The NRA has vigorously opposed any legislation that would require all guns to be smart guns. Because the availability of these weapons would trigger the New Jersey bill, California and Maryland have been prevented from importing smart firearms from a German company. Perhaps, however, New Jersey does not need to bear all the blame; in 1999, 4 years before the passage of the bill, the NRA and its members boycotted Smith & Wesson when the gun manufacturer revealed plans to develop a smart gun for the government. The NRA’s public stance is that it does not oppose smart guns for those who want them, but it opposes legislation that would eliminate the sale of conventional firearms. The organization has voiced concerns that technology fails and that it potentially slows down firing the weapon. It doesn’t talk about dead toddlers, or about police officers who’ve been killed when their weapons were taken from them.
As for the Parkland students, I don’t know why they aren’t asking for smart gun technology while they have the attention of the country. Perhaps they, like the young women I was with, don’t know it’s an option. Perhaps it’s too removed from the issue of mass murders and an assault weapon ban feels more attainable. Or perhaps the NRA’s mission has too much of a stronghold in Florida. I don’t know why they aren’t asking for smart gun production, but I know they should be.
*Correction, 3/27/2018: An earlier version of this story misstated the duration of Emma Gonzalez's moment of silence.
Dr. Miller is coauthor with Annette Hanson, MD, of “Committed: The Battle Over Involuntary Psychiatric Care” (Baltimore: Johns Hopkins University Press), 2016. She practices in Baltimore.
The evening before the March For Our Lives rally in Washington, I was hard at work on my sign. Making a statement is important, but at the Women’s March last January, I discovered that a sign was invaluable for keeping friends together in a crowd. Since my Women’s March sign read “Make America Kind Again,” I opted to keep with the theme, and I created a “Make America Safe Again” sign for gun control. My daughter looked at my sign skeptically. “It’s too vague and nondirective,” she declared. Now challenged, I added the following directives: “Universal Background Checks,” “Ban Assault Weapons,” and “Smart Guns.” My sign was now complete.
I was surprised when my daughter – our family Jeopardy! whiz – asked, “What’s a smart gun?” When we arrived to join friends, their daughter – a doctoral student – also looked at the sign and asked me what a smart gun is. I explained to both young women that a smart gun – like an iPhone – relies on a biometric such as a fingerprint so that it can be used only by authorized users. This technology would reduce the flow of firearms to illegal owners, prevent accidental discharge by children, protect rightful owners and law enforcement officers from having their own weapons used against them by criminals, and decrease the number of suicides by family members of gun owners. In my state of Maryland, there have been two school shootings, one as recently as March 20, and both were committed by boys who took a parent’s firearm to school.
We arrived at the rally and found a spot in front of the Newseum, with two jumbotrons in sight, right in the middle of the crowd. The young people who spoke were inspirational. Regardless of one’s political views or thoughts on gun control, they were bright, articulate, fearless, passionate, and determined. The show was stolen by Naomi Wadler, a precocious 11-year-old girl who is destined to be a member of Congress (if not president) one day, and by Dr. Martin Luther King Jr.’s 9-year-old granddaughter, Yolanda Renee King.
The rest of the speakers were teenagers, all of whom had lost someone to gunfire. There were speakers from Parkland, Fla., who talked about the raw losses they were feeling, and the student who wasn’t celebrating his 18th birthday on March 24. Matthew Soto from Newtown, Conn., talked about losing his sister, a first-grade teacher, in the Sandy Hook shooting.
But the rally wasn’t just about mass murders and school shootings: Zion Kelly talked about the death of his twin, a teen who was killed when he stopped at a convenience store on the way home from school in Washington, only to have his promising life snuffed out during a holdup. We heard about shootings in the bullet-riddled streets of Chicago and Los Angeles and the toll they have taken. The losses to gun violence have been felt most strongly in communities of color. While it was mentioned that the majority of gun deaths are suicides, there were no speakers from families of people who died from suicide. The final speaker was Emma Gonzalez, a young woman from Parkland who took the stage for 6 minutes and 20 seconds - the duration of the shooter’s rampage - and closed with a powerful moment of silence.* The rally closed with a performance of Bob Dylan’s “The Times They Are a-Changin’ ” by Jennifer Hudson, whose mother, brother, and nephew were murdered by a gunman.
The teens talked about what changes they wanted to see enacted. They talked about closing loopholes to background checks, and I was pleased that one young man specifically mentioned keeping guns from those who are violent, but did not mention mental illness as a reason to block gun ownership. A call to resume a ban on assault weapons was made repeatedly as was a call to raise the age (to 21) at which an individual can purchase a gun. Military-style weapons are not necessary for hunting or self-defense, and they enable the rapid-fire assassinations we have seen in mass shootings, so they remain an easy target of gun control advocacy. But in terms of numbers, these firearms are responsible for a small fraction of gun deaths. I was surprised, but the teens did not mention “smart gun” legislation as a way to reduce gun deaths.
Guns, and gun deaths, are a part of American society. While the majority of Americans favor stronger regulation of gun ownership, legislation that would end gun ownership is not likely to go anywhere. Smart guns, however, are different. Forty percent of polled gun owners have said they would swap their firearm for a smart firearm. So given the appeal of a firearm that can’t be diverted or stolen, used against the owner, discharged accidentally by a child, or used for suicide or homicide by a distressed family member, why don’t these weapons exist for use by the American people? . People want these guns and the protections they offer, yet they have never been produced and made available to either the American public or to our law enforcement officers.
So where are these firearms, and why aren’t our Parkland teens asking for them? The answer to the first part of that question lies with the National Rifle Association (NRA) and the state of New Jersey. In 2003, New Jersey passed the Childproof Handgun bill, which requires that all guns sold in the state be smart guns within 3 years of their availability. The NRA has vigorously opposed any legislation that would require all guns to be smart guns. Because the availability of these weapons would trigger the New Jersey bill, California and Maryland have been prevented from importing smart firearms from a German company. Perhaps, however, New Jersey does not need to bear all the blame; in 1999, 4 years before the passage of the bill, the NRA and its members boycotted Smith & Wesson when the gun manufacturer revealed plans to develop a smart gun for the government. The NRA’s public stance is that it does not oppose smart guns for those who want them, but it opposes legislation that would eliminate the sale of conventional firearms. The organization has voiced concerns that technology fails and that it potentially slows down firing the weapon. It doesn’t talk about dead toddlers, or about police officers who’ve been killed when their weapons were taken from them.
As for the Parkland students, I don’t know why they aren’t asking for smart gun technology while they have the attention of the country. Perhaps they, like the young women I was with, don’t know it’s an option. Perhaps it’s too removed from the issue of mass murders and an assault weapon ban feels more attainable. Or perhaps the NRA’s mission has too much of a stronghold in Florida. I don’t know why they aren’t asking for smart gun production, but I know they should be.
*Correction, 3/27/2018: An earlier version of this story misstated the duration of Emma Gonzalez's moment of silence.
Dr. Miller is coauthor with Annette Hanson, MD, of “Committed: The Battle Over Involuntary Psychiatric Care” (Baltimore: Johns Hopkins University Press), 2016. She practices in Baltimore.
Cancer groups offer guidance on musculoskeletal adverse events related to checkpoint inhibitors
Recently released guidelines from two major cancer organizations have provided some of the most comprehensive guidance to date on management of musculoskeletal side effects associated with cancer immunotherapy.
The guidelines, published in February, are a “sorely needed” reference point for the rheumatology community and others who will be encountering patients who experience immune-related adverse events (irAEs), according to Leonard H. Calabrese, DO, the R.J. Fasenmyer Chair of Clinical Immunology at the Cleveland Clinic in Ohio.
“They’re a good first start, given the fact that up until 4 or 5 months ago, there were no endorsed guidelines that included oncologists and rheumatologists,” Dr. Calabrese said of the guidelines, which were collaboratively developed and recently released by both the American Society of Clinical Oncology (ASCO) and the National Comprehensive Cancer Network (NCCN).
Rheumatologists can add value
“We talk a lot about rheumatologists being aware of these diseases, but it’s been pointed out by some oncologists that unless they’re really knowledgeable and can add considerably to the management, it doesn’t do any good just to be aware of it,” Dr. Calabrese explained. “You need to actually have some procedural knowledge.”
ASCO guidelines also describe a polymyalgia-like syndrome seen in some patients on immune checkpoint inhibitors that according to the guideline authors is characterized by pain, but not true muscle weakness.
In general, the guidelines endorse a stepwise approach, in which milder irAEs can be managed with conservative treatments and without the need to stop the immune checkpoint inhibitor therapy. In contrast, more serious side effects may require more intensive management and either temporary or permanent discontinuation of cancer immunotherapy.
One good example is NCCN’s take on managing inflammatory arthritis.
Mild cases of inflammatory arthritis can be treated with NSAIDs, low-dose prednisone, or intra-articular steroids with no need to stop immune checkpoint inhibitor treatment, according to NCCN guideline authors.
Moderate cases, by contrast, may require holding immunotherapy and treating with prednisone. A rheumatology consult by week 4 is “strongly recommended” if the immune arthritis doesn’t improve, the authors added.
Severe cases may warrant permanent discontinuation of immunotherapy and treatment with methylprednisolone/prednisone, infliximab, or tocilizumab, they added. If the irAE doesn’t improve after 2 weeks, a rheumatology consult should be considered for additional disease-modifying antirheumatic drugs, including methotrexate, leflunomide, or sulfasalazine.
irAEs on the rise
These guidelines are particularly useful for rheumatologists to familiarize themselves with the six Food and Drug Administration–approved immune checkpoint inhibitors, their spectrum of side effects, and how oncologists use the severity of presentation to guide therapy, according to Laura Cappelli, MD.
Understanding irAEs will be increasingly important for rheumatologist as the use of immune checkpoint inhibitors continues to increase, said Dr. Cappelli, who has started a research program at Johns Hopkins to evaluate the rheumatologic adverse effects of these therapies.
Dr. Cappelli said her division sees at least one suspected irAE case per week, most commonly the immune arthritis associated with checkpoint inhibitor therapy.
Likewise, Dr. Calabrese said he is already seeing approximately two new referrals per week for adverse events related to these relatively new therapies.
“It’s far outstripped our notion of what we thought we would be doing,” he said in an interview.
Systems have been set up to encourage interprofessional collaborations, he added, including a “virtual referral clinic” where advanced practitioners working with oncologists communicate with advanced practitioners in each of the specialty areas that are most frequently consulted in order to help facilitate care and triage patients.
In addition, a monthly irAE tumor board was set up to include only cases that have manifest autoimmune or autoinflammatory complications.
“We have a growing group of people who come to this from each area involved, whether it be nephrology, neurology, ophthalmology, gastroenterology, rheumatology, or something else,” Dr. Calabrese said. “That’s been a great learning experience for all of us to talk about these adverse events in real time.”
Critical need for guidance
These guidelines meet a growing need to help practicing clinicians identify and best manage immune-related adverse events, according to Bryan J. Schneider, MD, of the University of Michigan Comprehensive Cancer Center, and vice chair of the NCCN Panel on Management of Immunotherapy-Related Toxicities.
“We’re experienced with chemotherapy, and we are very comfortable with the side effects,” he said. “The immunotherapy story is just an entirely different world because, as I tell patients, the therapies aren’t directly damaging cancer cells like chemotherapy. Instead, they are helping the immune system to identify the cancer cells as abnormal and mount an assault. Proteins on cancer cells may suppress the immune response and these therapies effectively ‘release these brakes’ so the immune system can attack.”
Rheumatologists and those with particular expertise in rheumatologic side effects participated in the development of the ASCO and NCCN guidelines. They include Maria E. Suarez-Almazor, MD, PhD, chief of rheumatology and clinical immunology at the University of Texas MD Anderson Cancer Center, Houston, who served on the ASCO expert panel; Jarushka Naidoo, MBBCh, from the Sidney Kimmel Comprehensive Cancer Center at Johns Hopkins, provided expertise in the rheumatologic side effects that were considered by the NCCN panel, according to Dr. Schneider.
Rheumatologist input also informed another set of recommendations on immune checkpoint inhibitor toxicities published several months before the ASCO and NCCN guidelines. The working group for the September 2017 guidelines from the Society for Immunotherapy of Cancer (doi: 10.1186/s40425-017-0300-z) included Dr. Suarez-Almazor, as well as Clifton O. Bingham III, MD, director of the Johns Hopkins Arthritis Center.
Vigilance required
Checkpoint inhibitors have been approved by the FDA to treat a variety of cancers, including melanoma, lung cancer, and Hodgkin lymphoma, as well as lung, liver, kidney, and bladder cancers.
Clinicians managing patients on checkpoint inhibitors should always be vigilant because irAE symptoms can be subtle, according to Julie Brahmer, MD, of the Sidney Kimmel Comprehensive Cancer Center at Johns Hopkins in Baltimore.
“Everyone has to work as a team, which includes being educated on possible side effects to immunotherapy prior to prescribing it,” said Dr. Brahmer, chair of the ASCO panel and vice chair of the NCCN panel that developed the guidelines.
The guidelines were published Feb. 14 in two documents that are similar in content, but different in format. The ASCO guideline was published in the Journal of Clinical Oncology (doi: 10.1200/JCO.2017.77.6385) and the NCCN Clinical Practice Guidelines in Oncology were posted on the NCCN website.
While the first edition of the guidelines focus specifically on immune checkpoint inhibitors, an update anticipated for 2019 will include guidance on chimeric antigen receptor (CAR) T cell therapy, which is associated with several important side effects, notably cytokine release syndrome.
Recently released guidelines from two major cancer organizations have provided some of the most comprehensive guidance to date on management of musculoskeletal side effects associated with cancer immunotherapy.
The guidelines, published in February, are a “sorely needed” reference point for the rheumatology community and others who will be encountering patients who experience immune-related adverse events (irAEs), according to Leonard H. Calabrese, DO, the R.J. Fasenmyer Chair of Clinical Immunology at the Cleveland Clinic in Ohio.
“They’re a good first start, given the fact that up until 4 or 5 months ago, there were no endorsed guidelines that included oncologists and rheumatologists,” Dr. Calabrese said of the guidelines, which were collaboratively developed and recently released by both the American Society of Clinical Oncology (ASCO) and the National Comprehensive Cancer Network (NCCN).
Rheumatologists can add value
“We talk a lot about rheumatologists being aware of these diseases, but it’s been pointed out by some oncologists that unless they’re really knowledgeable and can add considerably to the management, it doesn’t do any good just to be aware of it,” Dr. Calabrese explained. “You need to actually have some procedural knowledge.”
ASCO guidelines also describe a polymyalgia-like syndrome seen in some patients on immune checkpoint inhibitors that according to the guideline authors is characterized by pain, but not true muscle weakness.
In general, the guidelines endorse a stepwise approach, in which milder irAEs can be managed with conservative treatments and without the need to stop the immune checkpoint inhibitor therapy. In contrast, more serious side effects may require more intensive management and either temporary or permanent discontinuation of cancer immunotherapy.
One good example is NCCN’s take on managing inflammatory arthritis.
Mild cases of inflammatory arthritis can be treated with NSAIDs, low-dose prednisone, or intra-articular steroids with no need to stop immune checkpoint inhibitor treatment, according to NCCN guideline authors.
Moderate cases, by contrast, may require holding immunotherapy and treating with prednisone. A rheumatology consult by week 4 is “strongly recommended” if the immune arthritis doesn’t improve, the authors added.
Severe cases may warrant permanent discontinuation of immunotherapy and treatment with methylprednisolone/prednisone, infliximab, or tocilizumab, they added. If the irAE doesn’t improve after 2 weeks, a rheumatology consult should be considered for additional disease-modifying antirheumatic drugs, including methotrexate, leflunomide, or sulfasalazine.
irAEs on the rise
These guidelines are particularly useful for rheumatologists to familiarize themselves with the six Food and Drug Administration–approved immune checkpoint inhibitors, their spectrum of side effects, and how oncologists use the severity of presentation to guide therapy, according to Laura Cappelli, MD.
Understanding irAEs will be increasingly important for rheumatologist as the use of immune checkpoint inhibitors continues to increase, said Dr. Cappelli, who has started a research program at Johns Hopkins to evaluate the rheumatologic adverse effects of these therapies.
Dr. Cappelli said her division sees at least one suspected irAE case per week, most commonly the immune arthritis associated with checkpoint inhibitor therapy.
Likewise, Dr. Calabrese said he is already seeing approximately two new referrals per week for adverse events related to these relatively new therapies.
“It’s far outstripped our notion of what we thought we would be doing,” he said in an interview.
Systems have been set up to encourage interprofessional collaborations, he added, including a “virtual referral clinic” where advanced practitioners working with oncologists communicate with advanced practitioners in each of the specialty areas that are most frequently consulted in order to help facilitate care and triage patients.
In addition, a monthly irAE tumor board was set up to include only cases that have manifest autoimmune or autoinflammatory complications.
“We have a growing group of people who come to this from each area involved, whether it be nephrology, neurology, ophthalmology, gastroenterology, rheumatology, or something else,” Dr. Calabrese said. “That’s been a great learning experience for all of us to talk about these adverse events in real time.”
Critical need for guidance
These guidelines meet a growing need to help practicing clinicians identify and best manage immune-related adverse events, according to Bryan J. Schneider, MD, of the University of Michigan Comprehensive Cancer Center, and vice chair of the NCCN Panel on Management of Immunotherapy-Related Toxicities.
“We’re experienced with chemotherapy, and we are very comfortable with the side effects,” he said. “The immunotherapy story is just an entirely different world because, as I tell patients, the therapies aren’t directly damaging cancer cells like chemotherapy. Instead, they are helping the immune system to identify the cancer cells as abnormal and mount an assault. Proteins on cancer cells may suppress the immune response and these therapies effectively ‘release these brakes’ so the immune system can attack.”
Rheumatologists and those with particular expertise in rheumatologic side effects participated in the development of the ASCO and NCCN guidelines. They include Maria E. Suarez-Almazor, MD, PhD, chief of rheumatology and clinical immunology at the University of Texas MD Anderson Cancer Center, Houston, who served on the ASCO expert panel; Jarushka Naidoo, MBBCh, from the Sidney Kimmel Comprehensive Cancer Center at Johns Hopkins, provided expertise in the rheumatologic side effects that were considered by the NCCN panel, according to Dr. Schneider.
Rheumatologist input also informed another set of recommendations on immune checkpoint inhibitor toxicities published several months before the ASCO and NCCN guidelines. The working group for the September 2017 guidelines from the Society for Immunotherapy of Cancer (doi: 10.1186/s40425-017-0300-z) included Dr. Suarez-Almazor, as well as Clifton O. Bingham III, MD, director of the Johns Hopkins Arthritis Center.
Vigilance required
Checkpoint inhibitors have been approved by the FDA to treat a variety of cancers, including melanoma, lung cancer, and Hodgkin lymphoma, as well as lung, liver, kidney, and bladder cancers.
Clinicians managing patients on checkpoint inhibitors should always be vigilant because irAE symptoms can be subtle, according to Julie Brahmer, MD, of the Sidney Kimmel Comprehensive Cancer Center at Johns Hopkins in Baltimore.
“Everyone has to work as a team, which includes being educated on possible side effects to immunotherapy prior to prescribing it,” said Dr. Brahmer, chair of the ASCO panel and vice chair of the NCCN panel that developed the guidelines.
The guidelines were published Feb. 14 in two documents that are similar in content, but different in format. The ASCO guideline was published in the Journal of Clinical Oncology (doi: 10.1200/JCO.2017.77.6385) and the NCCN Clinical Practice Guidelines in Oncology were posted on the NCCN website.
While the first edition of the guidelines focus specifically on immune checkpoint inhibitors, an update anticipated for 2019 will include guidance on chimeric antigen receptor (CAR) T cell therapy, which is associated with several important side effects, notably cytokine release syndrome.
Recently released guidelines from two major cancer organizations have provided some of the most comprehensive guidance to date on management of musculoskeletal side effects associated with cancer immunotherapy.
The guidelines, published in February, are a “sorely needed” reference point for the rheumatology community and others who will be encountering patients who experience immune-related adverse events (irAEs), according to Leonard H. Calabrese, DO, the R.J. Fasenmyer Chair of Clinical Immunology at the Cleveland Clinic in Ohio.
“They’re a good first start, given the fact that up until 4 or 5 months ago, there were no endorsed guidelines that included oncologists and rheumatologists,” Dr. Calabrese said of the guidelines, which were collaboratively developed and recently released by both the American Society of Clinical Oncology (ASCO) and the National Comprehensive Cancer Network (NCCN).
Rheumatologists can add value
“We talk a lot about rheumatologists being aware of these diseases, but it’s been pointed out by some oncologists that unless they’re really knowledgeable and can add considerably to the management, it doesn’t do any good just to be aware of it,” Dr. Calabrese explained. “You need to actually have some procedural knowledge.”
ASCO guidelines also describe a polymyalgia-like syndrome seen in some patients on immune checkpoint inhibitors that according to the guideline authors is characterized by pain, but not true muscle weakness.
In general, the guidelines endorse a stepwise approach, in which milder irAEs can be managed with conservative treatments and without the need to stop the immune checkpoint inhibitor therapy. In contrast, more serious side effects may require more intensive management and either temporary or permanent discontinuation of cancer immunotherapy.
One good example is NCCN’s take on managing inflammatory arthritis.
Mild cases of inflammatory arthritis can be treated with NSAIDs, low-dose prednisone, or intra-articular steroids with no need to stop immune checkpoint inhibitor treatment, according to NCCN guideline authors.
Moderate cases, by contrast, may require holding immunotherapy and treating with prednisone. A rheumatology consult by week 4 is “strongly recommended” if the immune arthritis doesn’t improve, the authors added.
Severe cases may warrant permanent discontinuation of immunotherapy and treatment with methylprednisolone/prednisone, infliximab, or tocilizumab, they added. If the irAE doesn’t improve after 2 weeks, a rheumatology consult should be considered for additional disease-modifying antirheumatic drugs, including methotrexate, leflunomide, or sulfasalazine.
irAEs on the rise
These guidelines are particularly useful for rheumatologists to familiarize themselves with the six Food and Drug Administration–approved immune checkpoint inhibitors, their spectrum of side effects, and how oncologists use the severity of presentation to guide therapy, according to Laura Cappelli, MD.
Understanding irAEs will be increasingly important for rheumatologist as the use of immune checkpoint inhibitors continues to increase, said Dr. Cappelli, who has started a research program at Johns Hopkins to evaluate the rheumatologic adverse effects of these therapies.
Dr. Cappelli said her division sees at least one suspected irAE case per week, most commonly the immune arthritis associated with checkpoint inhibitor therapy.
Likewise, Dr. Calabrese said he is already seeing approximately two new referrals per week for adverse events related to these relatively new therapies.
“It’s far outstripped our notion of what we thought we would be doing,” he said in an interview.
Systems have been set up to encourage interprofessional collaborations, he added, including a “virtual referral clinic” where advanced practitioners working with oncologists communicate with advanced practitioners in each of the specialty areas that are most frequently consulted in order to help facilitate care and triage patients.
In addition, a monthly irAE tumor board was set up to include only cases that have manifest autoimmune or autoinflammatory complications.
“We have a growing group of people who come to this from each area involved, whether it be nephrology, neurology, ophthalmology, gastroenterology, rheumatology, or something else,” Dr. Calabrese said. “That’s been a great learning experience for all of us to talk about these adverse events in real time.”
Critical need for guidance
These guidelines meet a growing need to help practicing clinicians identify and best manage immune-related adverse events, according to Bryan J. Schneider, MD, of the University of Michigan Comprehensive Cancer Center, and vice chair of the NCCN Panel on Management of Immunotherapy-Related Toxicities.
“We’re experienced with chemotherapy, and we are very comfortable with the side effects,” he said. “The immunotherapy story is just an entirely different world because, as I tell patients, the therapies aren’t directly damaging cancer cells like chemotherapy. Instead, they are helping the immune system to identify the cancer cells as abnormal and mount an assault. Proteins on cancer cells may suppress the immune response and these therapies effectively ‘release these brakes’ so the immune system can attack.”
Rheumatologists and those with particular expertise in rheumatologic side effects participated in the development of the ASCO and NCCN guidelines. They include Maria E. Suarez-Almazor, MD, PhD, chief of rheumatology and clinical immunology at the University of Texas MD Anderson Cancer Center, Houston, who served on the ASCO expert panel; Jarushka Naidoo, MBBCh, from the Sidney Kimmel Comprehensive Cancer Center at Johns Hopkins, provided expertise in the rheumatologic side effects that were considered by the NCCN panel, according to Dr. Schneider.
Rheumatologist input also informed another set of recommendations on immune checkpoint inhibitor toxicities published several months before the ASCO and NCCN guidelines. The working group for the September 2017 guidelines from the Society for Immunotherapy of Cancer (doi: 10.1186/s40425-017-0300-z) included Dr. Suarez-Almazor, as well as Clifton O. Bingham III, MD, director of the Johns Hopkins Arthritis Center.
Vigilance required
Checkpoint inhibitors have been approved by the FDA to treat a variety of cancers, including melanoma, lung cancer, and Hodgkin lymphoma, as well as lung, liver, kidney, and bladder cancers.
Clinicians managing patients on checkpoint inhibitors should always be vigilant because irAE symptoms can be subtle, according to Julie Brahmer, MD, of the Sidney Kimmel Comprehensive Cancer Center at Johns Hopkins in Baltimore.
“Everyone has to work as a team, which includes being educated on possible side effects to immunotherapy prior to prescribing it,” said Dr. Brahmer, chair of the ASCO panel and vice chair of the NCCN panel that developed the guidelines.
The guidelines were published Feb. 14 in two documents that are similar in content, but different in format. The ASCO guideline was published in the Journal of Clinical Oncology (doi: 10.1200/JCO.2017.77.6385) and the NCCN Clinical Practice Guidelines in Oncology were posted on the NCCN website.
While the first edition of the guidelines focus specifically on immune checkpoint inhibitors, an update anticipated for 2019 will include guidance on chimeric antigen receptor (CAR) T cell therapy, which is associated with several important side effects, notably cytokine release syndrome.
Better manage acute pancreatitis to improve patient outcomes
AGA has a new clinical guideline on the initial management of acute pancreatitis, published in Gastroenterology. In the U.S., acute pancreatitis (AP) is a leading cause of inpatient care among gastrointestinal conditions with more than 275,000 patients hospitalized annually, at an aggregate cost of over $2.6 billion per year. The guideline focuses on patient care within the first 48-72 hours of admission when management decisions can alter the course of disease and duration of hospitalization.
Guideline recommendations
AGA’s new guideline aims to reduce practice variation and promote high-quality and high-value care for patients suffering from acute pancreatitis. It addresses questions on the benefits of goal-directed fluid resuscitation, early oral feeding, enteral vs. parenteral nutrition, the routine use of prophylactic antibiotics, and routine ERCP in all patients with AP.
The guideline is accompanied by a technical review, a new spotlight (infographic) and a patient companion infographic, which provides key points and important information directly to patients.
AGA has a new clinical guideline on the initial management of acute pancreatitis, published in Gastroenterology. In the U.S., acute pancreatitis (AP) is a leading cause of inpatient care among gastrointestinal conditions with more than 275,000 patients hospitalized annually, at an aggregate cost of over $2.6 billion per year. The guideline focuses on patient care within the first 48-72 hours of admission when management decisions can alter the course of disease and duration of hospitalization.
Guideline recommendations
AGA’s new guideline aims to reduce practice variation and promote high-quality and high-value care for patients suffering from acute pancreatitis. It addresses questions on the benefits of goal-directed fluid resuscitation, early oral feeding, enteral vs. parenteral nutrition, the routine use of prophylactic antibiotics, and routine ERCP in all patients with AP.
The guideline is accompanied by a technical review, a new spotlight (infographic) and a patient companion infographic, which provides key points and important information directly to patients.
AGA has a new clinical guideline on the initial management of acute pancreatitis, published in Gastroenterology. In the U.S., acute pancreatitis (AP) is a leading cause of inpatient care among gastrointestinal conditions with more than 275,000 patients hospitalized annually, at an aggregate cost of over $2.6 billion per year. The guideline focuses on patient care within the first 48-72 hours of admission when management decisions can alter the course of disease and duration of hospitalization.
Guideline recommendations
AGA’s new guideline aims to reduce practice variation and promote high-quality and high-value care for patients suffering from acute pancreatitis. It addresses questions on the benefits of goal-directed fluid resuscitation, early oral feeding, enteral vs. parenteral nutrition, the routine use of prophylactic antibiotics, and routine ERCP in all patients with AP.
The guideline is accompanied by a technical review, a new spotlight (infographic) and a patient companion infographic, which provides key points and important information directly to patients.
How to talk with your patients about PPIs and cognitive decline
A 2018 study published in Clinical Gastroenterology and Hepatology, “Lack of association between proton pump inhibitor use and cognitive decline,” found no association between PPI use and cognitive decline in analyzing data from two large population-based studies in Denmark. While this data is reassuring, clinicians should continue to anticipate questions from their patients about the risks associated with PPI therapy.
- Reassure patients that you prescribed a PPI for a clear-cut indication, in the lowest possible dose, and for an appropriate period of time (lowest dose, shortest time). This advice echoes that offered by AGA and ABIM in the Choosing Wisely campaign.
- Educate patients not to ask “what side effects do PPIs have?” but rather “is it really indicated?” Reassure patients that, when PPIs are indicated, benefits outweigh risks.
- Keep conversation channels open with patients. When patients require long-term use of PPIs, the medication should not be stopped without a discussion with you about the risks and benefits.
- Recommend that patients also consider life-style modifications that may reduce or eliminate the need for PPIs for long-term use.
A 2018 study published in Clinical Gastroenterology and Hepatology, “Lack of association between proton pump inhibitor use and cognitive decline,” found no association between PPI use and cognitive decline in analyzing data from two large population-based studies in Denmark. While this data is reassuring, clinicians should continue to anticipate questions from their patients about the risks associated with PPI therapy.
- Reassure patients that you prescribed a PPI for a clear-cut indication, in the lowest possible dose, and for an appropriate period of time (lowest dose, shortest time). This advice echoes that offered by AGA and ABIM in the Choosing Wisely campaign.
- Educate patients not to ask “what side effects do PPIs have?” but rather “is it really indicated?” Reassure patients that, when PPIs are indicated, benefits outweigh risks.
- Keep conversation channels open with patients. When patients require long-term use of PPIs, the medication should not be stopped without a discussion with you about the risks and benefits.
- Recommend that patients also consider life-style modifications that may reduce or eliminate the need for PPIs for long-term use.
A 2018 study published in Clinical Gastroenterology and Hepatology, “Lack of association between proton pump inhibitor use and cognitive decline,” found no association between PPI use and cognitive decline in analyzing data from two large population-based studies in Denmark. While this data is reassuring, clinicians should continue to anticipate questions from their patients about the risks associated with PPI therapy.
- Reassure patients that you prescribed a PPI for a clear-cut indication, in the lowest possible dose, and for an appropriate period of time (lowest dose, shortest time). This advice echoes that offered by AGA and ABIM in the Choosing Wisely campaign.
- Educate patients not to ask “what side effects do PPIs have?” but rather “is it really indicated?” Reassure patients that, when PPIs are indicated, benefits outweigh risks.
- Keep conversation channels open with patients. When patients require long-term use of PPIs, the medication should not be stopped without a discussion with you about the risks and benefits.
- Recommend that patients also consider life-style modifications that may reduce or eliminate the need for PPIs for long-term use.
Four new and noteworthy IBD drug studies
Inflammatory bowel disease (IBD) is a vibrant area of clinical research. Many of the 250+ abstracts presented at the inaugural Crohn’s & Colitis Congress — a partnership of the Crohn’s & Colitis Foundation and AGA — looked at the efficacy and safety of IBD therapies. You can review all abstracts presented at the Crohn’s & Colitis Congress in Gastroenterology.
Double-blind, randomized, placebo-controlled, crossover trial to evaluate induction of clinical response in patients with moderate-severe Crohn’s disease treated with rifaximin
By Scott D. Lee, University of Washington Medicine, et al.
Significance: It is now known that the intestinal microbiome is integral to the pathogenesis of IBD. However, antibiotic treatments for IBD have previously shown limited effectiveness. In this 8-week clinical trial, there was a fourfold greater response to the antibiotic rifaximin in Crohn’s disease treatment, compared with placebo. The positive impact on clinical disease activity was seen even in patients with a significant disease burden and prior exposure to one or more biologic therapies. Quality of life and laboratory measurements were numerically improved. No new safety concerns were identified. These results offer renewed hope for the use of antibiotics in treating Crohn’s disease.
Post-hoc analysis of tofacitinib Crohn’s disease phase 2 induction efficacy in subgroups with baseline endoscopic or biomarker evidence of inflammation
By Bruce E. Sands, Icahn School of Medicine at Mount Sinai, et al.
Significance: Tofacitinib, a Janus kinase (JAK) inhibitor, is under investigation for treatment of ulcerative colitis and Crohn’s disease. To date, response rates in ulcerative colitis have been higher than for Crohn’s disease. In this report, investigators performed post-hoc analysis studies using objective baseline criteria of disease activity. Their findings showed a greater proportion of patients with moderate to severe Crohn’s disease were in remission with tofacitinib compared to placebo. These results provide evidence of JAK inhibition for the treatment of Crohn’s disease and support further investigation.
Refined population pharmacokinetic model for infliximab precision dosing in pediatric inflammatory bowel disease
By Laura E. Bauman, Cincinnati Children’s Hospital Medical Center, et al.
Significance: Long-term clinical remission from IBD with anti-TNF therapies has generally been limited to less than half of the treated patients. Improved outcomes are seen with optimal pre-infusion trough drug levels, a measurement of the level of drugs in the patient’s bloodstream. However, standard weight-based dosing for pediatric patients has provided widely varying trough drug levels. The investigators report the development of a multifactorial pharmacokinetic model for predicting infliximab trough levels during maintenance therapy for IBD. Such dynamic approaches to treatment address a specific gap in pediatric IBD therapeutic strategies.
Primary nonresponse to tumor necrosis factor antagonists is associated with inferior response to second-line biologics in patients with inflammatory bowel diseases: A systematic review and meta-analysis
By Siddharth Singh, University of California San Diego Health, et al.
Significance: Primary nonresponse to anti-TNF therapy is seen in 35%-65% of IBD patients and another 40%-60% lose responsiveness during the first year of treatment. Physicians struggle with what treatments to recommend for these patients. The investigators in this study performed a literature search and identified eight randomized controlled trials of biologics in patients with prior exposure to anti-TNF and compared outcomes based on their prior responses to anti-TNF. The analysis reveals a 24% decrease in likelihood to achieve remission in patients who changed medications because of immediate nonresponse compared to loss of responsiveness or intolerance during the treatment. These findings raise important questions about the biology of IBD, including the pharmacology of anti-TNF in a subset of patients.
Inflammatory bowel disease (IBD) is a vibrant area of clinical research. Many of the 250+ abstracts presented at the inaugural Crohn’s & Colitis Congress — a partnership of the Crohn’s & Colitis Foundation and AGA — looked at the efficacy and safety of IBD therapies. You can review all abstracts presented at the Crohn’s & Colitis Congress in Gastroenterology.
Double-blind, randomized, placebo-controlled, crossover trial to evaluate induction of clinical response in patients with moderate-severe Crohn’s disease treated with rifaximin
By Scott D. Lee, University of Washington Medicine, et al.
Significance: It is now known that the intestinal microbiome is integral to the pathogenesis of IBD. However, antibiotic treatments for IBD have previously shown limited effectiveness. In this 8-week clinical trial, there was a fourfold greater response to the antibiotic rifaximin in Crohn’s disease treatment, compared with placebo. The positive impact on clinical disease activity was seen even in patients with a significant disease burden and prior exposure to one or more biologic therapies. Quality of life and laboratory measurements were numerically improved. No new safety concerns were identified. These results offer renewed hope for the use of antibiotics in treating Crohn’s disease.
Post-hoc analysis of tofacitinib Crohn’s disease phase 2 induction efficacy in subgroups with baseline endoscopic or biomarker evidence of inflammation
By Bruce E. Sands, Icahn School of Medicine at Mount Sinai, et al.
Significance: Tofacitinib, a Janus kinase (JAK) inhibitor, is under investigation for treatment of ulcerative colitis and Crohn’s disease. To date, response rates in ulcerative colitis have been higher than for Crohn’s disease. In this report, investigators performed post-hoc analysis studies using objective baseline criteria of disease activity. Their findings showed a greater proportion of patients with moderate to severe Crohn’s disease were in remission with tofacitinib compared to placebo. These results provide evidence of JAK inhibition for the treatment of Crohn’s disease and support further investigation.
Refined population pharmacokinetic model for infliximab precision dosing in pediatric inflammatory bowel disease
By Laura E. Bauman, Cincinnati Children’s Hospital Medical Center, et al.
Significance: Long-term clinical remission from IBD with anti-TNF therapies has generally been limited to less than half of the treated patients. Improved outcomes are seen with optimal pre-infusion trough drug levels, a measurement of the level of drugs in the patient’s bloodstream. However, standard weight-based dosing for pediatric patients has provided widely varying trough drug levels. The investigators report the development of a multifactorial pharmacokinetic model for predicting infliximab trough levels during maintenance therapy for IBD. Such dynamic approaches to treatment address a specific gap in pediatric IBD therapeutic strategies.
Primary nonresponse to tumor necrosis factor antagonists is associated with inferior response to second-line biologics in patients with inflammatory bowel diseases: A systematic review and meta-analysis
By Siddharth Singh, University of California San Diego Health, et al.
Significance: Primary nonresponse to anti-TNF therapy is seen in 35%-65% of IBD patients and another 40%-60% lose responsiveness during the first year of treatment. Physicians struggle with what treatments to recommend for these patients. The investigators in this study performed a literature search and identified eight randomized controlled trials of biologics in patients with prior exposure to anti-TNF and compared outcomes based on their prior responses to anti-TNF. The analysis reveals a 24% decrease in likelihood to achieve remission in patients who changed medications because of immediate nonresponse compared to loss of responsiveness or intolerance during the treatment. These findings raise important questions about the biology of IBD, including the pharmacology of anti-TNF in a subset of patients.
Inflammatory bowel disease (IBD) is a vibrant area of clinical research. Many of the 250+ abstracts presented at the inaugural Crohn’s & Colitis Congress — a partnership of the Crohn’s & Colitis Foundation and AGA — looked at the efficacy and safety of IBD therapies. You can review all abstracts presented at the Crohn’s & Colitis Congress in Gastroenterology.
Double-blind, randomized, placebo-controlled, crossover trial to evaluate induction of clinical response in patients with moderate-severe Crohn’s disease treated with rifaximin
By Scott D. Lee, University of Washington Medicine, et al.
Significance: It is now known that the intestinal microbiome is integral to the pathogenesis of IBD. However, antibiotic treatments for IBD have previously shown limited effectiveness. In this 8-week clinical trial, there was a fourfold greater response to the antibiotic rifaximin in Crohn’s disease treatment, compared with placebo. The positive impact on clinical disease activity was seen even in patients with a significant disease burden and prior exposure to one or more biologic therapies. Quality of life and laboratory measurements were numerically improved. No new safety concerns were identified. These results offer renewed hope for the use of antibiotics in treating Crohn’s disease.
Post-hoc analysis of tofacitinib Crohn’s disease phase 2 induction efficacy in subgroups with baseline endoscopic or biomarker evidence of inflammation
By Bruce E. Sands, Icahn School of Medicine at Mount Sinai, et al.
Significance: Tofacitinib, a Janus kinase (JAK) inhibitor, is under investigation for treatment of ulcerative colitis and Crohn’s disease. To date, response rates in ulcerative colitis have been higher than for Crohn’s disease. In this report, investigators performed post-hoc analysis studies using objective baseline criteria of disease activity. Their findings showed a greater proportion of patients with moderate to severe Crohn’s disease were in remission with tofacitinib compared to placebo. These results provide evidence of JAK inhibition for the treatment of Crohn’s disease and support further investigation.
Refined population pharmacokinetic model for infliximab precision dosing in pediatric inflammatory bowel disease
By Laura E. Bauman, Cincinnati Children’s Hospital Medical Center, et al.
Significance: Long-term clinical remission from IBD with anti-TNF therapies has generally been limited to less than half of the treated patients. Improved outcomes are seen with optimal pre-infusion trough drug levels, a measurement of the level of drugs in the patient’s bloodstream. However, standard weight-based dosing for pediatric patients has provided widely varying trough drug levels. The investigators report the development of a multifactorial pharmacokinetic model for predicting infliximab trough levels during maintenance therapy for IBD. Such dynamic approaches to treatment address a specific gap in pediatric IBD therapeutic strategies.
Primary nonresponse to tumor necrosis factor antagonists is associated with inferior response to second-line biologics in patients with inflammatory bowel diseases: A systematic review and meta-analysis
By Siddharth Singh, University of California San Diego Health, et al.
Significance: Primary nonresponse to anti-TNF therapy is seen in 35%-65% of IBD patients and another 40%-60% lose responsiveness during the first year of treatment. Physicians struggle with what treatments to recommend for these patients. The investigators in this study performed a literature search and identified eight randomized controlled trials of biologics in patients with prior exposure to anti-TNF and compared outcomes based on their prior responses to anti-TNF. The analysis reveals a 24% decrease in likelihood to achieve remission in patients who changed medications because of immediate nonresponse compared to loss of responsiveness or intolerance during the treatment. These findings raise important questions about the biology of IBD, including the pharmacology of anti-TNF in a subset of patients.
Remember the AGA Research Foundation in your will or living trust
What if all you had to do to ensure that the AGA Research Foundation can have an impact for years to come is to write a simple sentence? Sound impossible?
The AGA Research Foundation provides a key source of funding at a critical juncture in a young investigators’ career. Securing the future of the talented investigators we serve really is as simple as one sentence.
Including the AGA Research Foundation in your will is a popular gift to give because it is:
- • Affordable. The actual giving of your gift occurs after your lifetime, so your current income is not affected.
- • Flexible. Until your will goes into effect, you are free to alter your plans or change your mind.
- • Versatile. You can give a specific item, a set amount of money, or a percentage of your estate. You can also make your gift contingent upon certain events.
We hope you’ll consider including a gift to the AGA Research Foundation in your will or living trust. It’s simple – just a few sentences in your will or trust are all that is needed. The official bequest language for the AGA Research Foundation is: “I, [name], of [city, state, ZIP], give, devise and bequeath to the AGA Research Foundation [written amount or percentage of the estate or description of property] for its unrestricted use and purpose.”
Join others in donating to the AGA Research Foundation and help fill the funding gap and protect the next generation of investigators.
Please contact us for more information at [email protected] or visit http://gastro.planmylegacy.org/.
What if all you had to do to ensure that the AGA Research Foundation can have an impact for years to come is to write a simple sentence? Sound impossible?
The AGA Research Foundation provides a key source of funding at a critical juncture in a young investigators’ career. Securing the future of the talented investigators we serve really is as simple as one sentence.
Including the AGA Research Foundation in your will is a popular gift to give because it is:
- • Affordable. The actual giving of your gift occurs after your lifetime, so your current income is not affected.
- • Flexible. Until your will goes into effect, you are free to alter your plans or change your mind.
- • Versatile. You can give a specific item, a set amount of money, or a percentage of your estate. You can also make your gift contingent upon certain events.
We hope you’ll consider including a gift to the AGA Research Foundation in your will or living trust. It’s simple – just a few sentences in your will or trust are all that is needed. The official bequest language for the AGA Research Foundation is: “I, [name], of [city, state, ZIP], give, devise and bequeath to the AGA Research Foundation [written amount or percentage of the estate or description of property] for its unrestricted use and purpose.”
Join others in donating to the AGA Research Foundation and help fill the funding gap and protect the next generation of investigators.
Please contact us for more information at [email protected] or visit http://gastro.planmylegacy.org/.
What if all you had to do to ensure that the AGA Research Foundation can have an impact for years to come is to write a simple sentence? Sound impossible?
The AGA Research Foundation provides a key source of funding at a critical juncture in a young investigators’ career. Securing the future of the talented investigators we serve really is as simple as one sentence.
Including the AGA Research Foundation in your will is a popular gift to give because it is:
- • Affordable. The actual giving of your gift occurs after your lifetime, so your current income is not affected.
- • Flexible. Until your will goes into effect, you are free to alter your plans or change your mind.
- • Versatile. You can give a specific item, a set amount of money, or a percentage of your estate. You can also make your gift contingent upon certain events.
We hope you’ll consider including a gift to the AGA Research Foundation in your will or living trust. It’s simple – just a few sentences in your will or trust are all that is needed. The official bequest language for the AGA Research Foundation is: “I, [name], of [city, state, ZIP], give, devise and bequeath to the AGA Research Foundation [written amount or percentage of the estate or description of property] for its unrestricted use and purpose.”
Join others in donating to the AGA Research Foundation and help fill the funding gap and protect the next generation of investigators.
Please contact us for more information at [email protected] or visit http://gastro.planmylegacy.org/.
TB in 2017: Good news and bad news
according to the Centers for Disease Control and Prevention.
Those new lows – TB incidence of 2.8 per 100,000 persons and 9,093 new cases – continue a downward trend that started in 1993, but the current rate of decline is much lower than the threshold needed to eliminate TB by the year 2100, Rebekah J. Stewart and her associates at the CDC’s Division of Tuberculosis Elimination, Atlanta, wrote in the Morbidity and Mortality Weekly Report.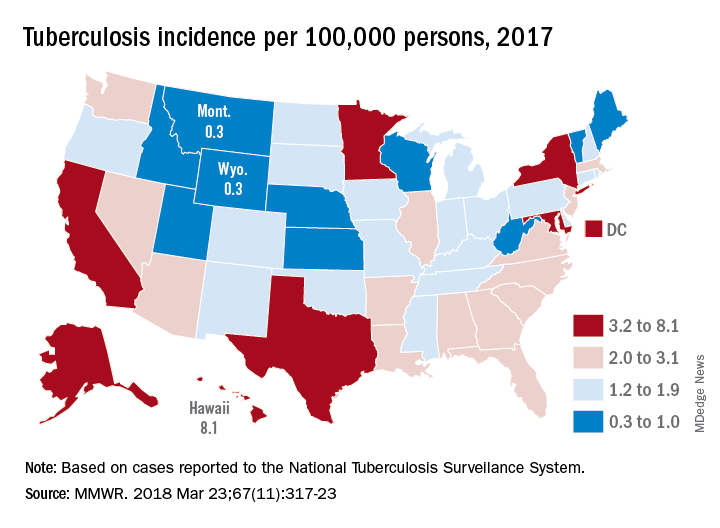
Geographically, at least, the states with populations at the highest risk are Hawaii, which had a TB incidence of 8.1 per 100,000 persons in 2017, and Alaska, with an incidence of 7.0 per 100,000. California and the District of Columbia were next, each with an incidence of 5.2. The states with the lowest rates were Montana and Wyoming at 0.3 per 100,000, the investigators reported, based on data from the National Tuberculosis Surveillance System as of Feb. 12, 2018.
Groups most affected by TB include persons housed in congregate settings – homeless shelters, long-term care facilities, and correctional facilities – and those from countries that have high TB prevalence. Overall incidence for non–U.S. born residents was 14.6 per 100,000 in 2017, compared with 1.0 for the native born, with large discrepancies seen between U.S. and non–U.S. born blacks (2.8 vs. 22.0), native Hawaiian/Pacific Islanders (6.5 vs. 21.0), and Asians (2.0 vs. 27.0), Ms. Stewart and her associates said.
“Increased support of global TB elimination efforts would help to reduce global … prevalence, thereby indirectly reducing the incidence of reactivation TB in the United States among non–U.S. born persons from higher-prevalence countries,” they wrote.
The issue of global action on TB was addressed by the Forum of International Respiratory Societies in a statement recognizing World TB Day (March 24). “TB is the world’s most common infectious disease killer, yet is identifiable, treatable and preventable; what is missing is the political will to dedicate the resources necessary to eradicate it, once and for all,” said Dean E. Schraufnagel, MD, the organization’s executive director.
SOURCE: Stewart RJ et al. MMWR 2018 Mar 23;67(11):317-23.
according to the Centers for Disease Control and Prevention.
Those new lows – TB incidence of 2.8 per 100,000 persons and 9,093 new cases – continue a downward trend that started in 1993, but the current rate of decline is much lower than the threshold needed to eliminate TB by the year 2100, Rebekah J. Stewart and her associates at the CDC’s Division of Tuberculosis Elimination, Atlanta, wrote in the Morbidity and Mortality Weekly Report.
Geographically, at least, the states with populations at the highest risk are Hawaii, which had a TB incidence of 8.1 per 100,000 persons in 2017, and Alaska, with an incidence of 7.0 per 100,000. California and the District of Columbia were next, each with an incidence of 5.2. The states with the lowest rates were Montana and Wyoming at 0.3 per 100,000, the investigators reported, based on data from the National Tuberculosis Surveillance System as of Feb. 12, 2018.
Groups most affected by TB include persons housed in congregate settings – homeless shelters, long-term care facilities, and correctional facilities – and those from countries that have high TB prevalence. Overall incidence for non–U.S. born residents was 14.6 per 100,000 in 2017, compared with 1.0 for the native born, with large discrepancies seen between U.S. and non–U.S. born blacks (2.8 vs. 22.0), native Hawaiian/Pacific Islanders (6.5 vs. 21.0), and Asians (2.0 vs. 27.0), Ms. Stewart and her associates said.
“Increased support of global TB elimination efforts would help to reduce global … prevalence, thereby indirectly reducing the incidence of reactivation TB in the United States among non–U.S. born persons from higher-prevalence countries,” they wrote.
The issue of global action on TB was addressed by the Forum of International Respiratory Societies in a statement recognizing World TB Day (March 24). “TB is the world’s most common infectious disease killer, yet is identifiable, treatable and preventable; what is missing is the political will to dedicate the resources necessary to eradicate it, once and for all,” said Dean E. Schraufnagel, MD, the organization’s executive director.
SOURCE: Stewart RJ et al. MMWR 2018 Mar 23;67(11):317-23.
according to the Centers for Disease Control and Prevention.
Those new lows – TB incidence of 2.8 per 100,000 persons and 9,093 new cases – continue a downward trend that started in 1993, but the current rate of decline is much lower than the threshold needed to eliminate TB by the year 2100, Rebekah J. Stewart and her associates at the CDC’s Division of Tuberculosis Elimination, Atlanta, wrote in the Morbidity and Mortality Weekly Report.
Geographically, at least, the states with populations at the highest risk are Hawaii, which had a TB incidence of 8.1 per 100,000 persons in 2017, and Alaska, with an incidence of 7.0 per 100,000. California and the District of Columbia were next, each with an incidence of 5.2. The states with the lowest rates were Montana and Wyoming at 0.3 per 100,000, the investigators reported, based on data from the National Tuberculosis Surveillance System as of Feb. 12, 2018.
Groups most affected by TB include persons housed in congregate settings – homeless shelters, long-term care facilities, and correctional facilities – and those from countries that have high TB prevalence. Overall incidence for non–U.S. born residents was 14.6 per 100,000 in 2017, compared with 1.0 for the native born, with large discrepancies seen between U.S. and non–U.S. born blacks (2.8 vs. 22.0), native Hawaiian/Pacific Islanders (6.5 vs. 21.0), and Asians (2.0 vs. 27.0), Ms. Stewart and her associates said.
“Increased support of global TB elimination efforts would help to reduce global … prevalence, thereby indirectly reducing the incidence of reactivation TB in the United States among non–U.S. born persons from higher-prevalence countries,” they wrote.
The issue of global action on TB was addressed by the Forum of International Respiratory Societies in a statement recognizing World TB Day (March 24). “TB is the world’s most common infectious disease killer, yet is identifiable, treatable and preventable; what is missing is the political will to dedicate the resources necessary to eradicate it, once and for all,” said Dean E. Schraufnagel, MD, the organization’s executive director.
SOURCE: Stewart RJ et al. MMWR 2018 Mar 23;67(11):317-23.
FROM MMWR
MRI matches standard biopsy at detecting significant prostate cancers
Men who dread the prospect of multicore prostate biopsies can take heart in the news that multiparametric MRI with or without targeted biopsy was noninferior to transrectal ultrasound at detecting clinically significant cancers, results of a multicenter randomized trial indicate.
The rate of clinically significant cancers detected in men with clinical suspicion of prostate cancer who were randomly assigned to undergo MRI was 38%, compared with 26% (P = .005) for men assigned to standard transrectal ultrasound guided biopsy with 10 or 12 biopsy cores, reported Veeru Kasivisvanathan, MRCS, of University College London, and colleagues in the PRECISION trial (Prostate Evaluation for Clinically Important Disease: Sampling Using Image-guidance or Not?).
Significantly fewer men assigned to MRI-targeted biopsy were diagnosed with clinically insignificant cancers, suggesting that MRI could help to reduce the number of invasive biopsies and the associated pain, discomfort, and infection risks, the investigators stated in the New England Journal of Medicine.
“MRI, with or without targeted biopsy, led to fewer men undergoing biopsy, more clinically significant cancers being identified, less overdetection of clinically insignificant cancer, and fewer biopsy cores being obtained than did standard transrectal ultrasonography-guided biopsy,” they wrote.
Multiparametric MRI combines several different imaging modalities, including standard T1- and T2-weighted scans with dynamic contrast–enhanced and/or diffusion-weighted imaging to provide a wealth of information to aid in diagnosis. The technique has been shown in single-center studies to be similar or superior to ultrasound guided biopsy at detecting clinically significant cancers and minimizing detection of cancers that turn out to be clinically insignificant, the investigators said.
To add to the body of evidence, investigators from 25 centers in 11 countries randomized a total of 500 men with clinical suspicion of prostate cancer and no history of prostate biopsy to undergo either MRI plus targeted biopsy (not 10- or 12-core biopsy) if the scans indicated suspicion of malignancy, or standard transrectal ultrasound-guided biopsy with 10 or 12 core samples.
The investigators defined clinically significant cancer as the presence of a single biopsy core indicating disease of Gleason score 3 plus 4 (Gleason sum of 7), or greater.
Of the 252 men assigned to MRI, 71 (28%) had results that did not suggest prostate cancer, and these men did not undergo biopsy.
As noted before, MRI was noninferior to standard ultrasound-guided biopsy. In the MRI group, 95 men (38%) were determined to have clinically significant tumors, compared with 64 of 248 men (26%) in the standard biopsy group.
Getting down into the statistical weeds, the lower boundary of the 95% confidence interval for the difference was greater than −5%, showing that MRI with or without targeted biopsy was noninferior to standard transrectal ultrasonography-guided biopsy for the detection of clinically significant cancers. In fact, the 95% confidence interval for the 12-point difference between the two techniques (95% confidence interval, 4-20) showed that MRI was superior to standard biopsy, the authors stated.
There were no health-related quality of life differences at either 24 hours or 30 days after the procedure, and immediate postintervention discomfort and pain were also similar between the groups. However, patient-reported complications were lower in patients assigned to MRI, including blood in urine (30% vs. 63% for standard biopsy), blood in semen (32% vs. 60%), procedural-site pain (13% vs. 23%), rectal bleeding (14% vs. 22%), and erectile dysfunction (11% vs. 16%).
“We found that a diagnostic pathway including risk assessment with MRI before biopsy and MRI-targeted biopsy in the presence of a lesion suggestive of cancer was superior to the diagnostic pathway of standard transrectal ultrasonography-guided biopsy,” Dr. Kasivisvanathan and his associates concluded.
The study was supported by awards to Dr. Kasivisvanathan from the U.K. National Institute for Health Research and by the European Association of Urology Research Foundation. He reported no other significant conflicts of interest to disclose.
SOURCE: Kasivisvanathan V et al. N Engl J Med. 2018 Mar 19. doi: 10.1056/NEJMoa1801993.
Men who dread the prospect of multicore prostate biopsies can take heart in the news that multiparametric MRI with or without targeted biopsy was noninferior to transrectal ultrasound at detecting clinically significant cancers, results of a multicenter randomized trial indicate.
The rate of clinically significant cancers detected in men with clinical suspicion of prostate cancer who were randomly assigned to undergo MRI was 38%, compared with 26% (P = .005) for men assigned to standard transrectal ultrasound guided biopsy with 10 or 12 biopsy cores, reported Veeru Kasivisvanathan, MRCS, of University College London, and colleagues in the PRECISION trial (Prostate Evaluation for Clinically Important Disease: Sampling Using Image-guidance or Not?).
Significantly fewer men assigned to MRI-targeted biopsy were diagnosed with clinically insignificant cancers, suggesting that MRI could help to reduce the number of invasive biopsies and the associated pain, discomfort, and infection risks, the investigators stated in the New England Journal of Medicine.
“MRI, with or without targeted biopsy, led to fewer men undergoing biopsy, more clinically significant cancers being identified, less overdetection of clinically insignificant cancer, and fewer biopsy cores being obtained than did standard transrectal ultrasonography-guided biopsy,” they wrote.
Multiparametric MRI combines several different imaging modalities, including standard T1- and T2-weighted scans with dynamic contrast–enhanced and/or diffusion-weighted imaging to provide a wealth of information to aid in diagnosis. The technique has been shown in single-center studies to be similar or superior to ultrasound guided biopsy at detecting clinically significant cancers and minimizing detection of cancers that turn out to be clinically insignificant, the investigators said.
To add to the body of evidence, investigators from 25 centers in 11 countries randomized a total of 500 men with clinical suspicion of prostate cancer and no history of prostate biopsy to undergo either MRI plus targeted biopsy (not 10- or 12-core biopsy) if the scans indicated suspicion of malignancy, or standard transrectal ultrasound-guided biopsy with 10 or 12 core samples.
The investigators defined clinically significant cancer as the presence of a single biopsy core indicating disease of Gleason score 3 plus 4 (Gleason sum of 7), or greater.
Of the 252 men assigned to MRI, 71 (28%) had results that did not suggest prostate cancer, and these men did not undergo biopsy.
As noted before, MRI was noninferior to standard ultrasound-guided biopsy. In the MRI group, 95 men (38%) were determined to have clinically significant tumors, compared with 64 of 248 men (26%) in the standard biopsy group.
Getting down into the statistical weeds, the lower boundary of the 95% confidence interval for the difference was greater than −5%, showing that MRI with or without targeted biopsy was noninferior to standard transrectal ultrasonography-guided biopsy for the detection of clinically significant cancers. In fact, the 95% confidence interval for the 12-point difference between the two techniques (95% confidence interval, 4-20) showed that MRI was superior to standard biopsy, the authors stated.
There were no health-related quality of life differences at either 24 hours or 30 days after the procedure, and immediate postintervention discomfort and pain were also similar between the groups. However, patient-reported complications were lower in patients assigned to MRI, including blood in urine (30% vs. 63% for standard biopsy), blood in semen (32% vs. 60%), procedural-site pain (13% vs. 23%), rectal bleeding (14% vs. 22%), and erectile dysfunction (11% vs. 16%).
“We found that a diagnostic pathway including risk assessment with MRI before biopsy and MRI-targeted biopsy in the presence of a lesion suggestive of cancer was superior to the diagnostic pathway of standard transrectal ultrasonography-guided biopsy,” Dr. Kasivisvanathan and his associates concluded.
The study was supported by awards to Dr. Kasivisvanathan from the U.K. National Institute for Health Research and by the European Association of Urology Research Foundation. He reported no other significant conflicts of interest to disclose.
SOURCE: Kasivisvanathan V et al. N Engl J Med. 2018 Mar 19. doi: 10.1056/NEJMoa1801993.
Men who dread the prospect of multicore prostate biopsies can take heart in the news that multiparametric MRI with or without targeted biopsy was noninferior to transrectal ultrasound at detecting clinically significant cancers, results of a multicenter randomized trial indicate.
The rate of clinically significant cancers detected in men with clinical suspicion of prostate cancer who were randomly assigned to undergo MRI was 38%, compared with 26% (P = .005) for men assigned to standard transrectal ultrasound guided biopsy with 10 or 12 biopsy cores, reported Veeru Kasivisvanathan, MRCS, of University College London, and colleagues in the PRECISION trial (Prostate Evaluation for Clinically Important Disease: Sampling Using Image-guidance or Not?).
Significantly fewer men assigned to MRI-targeted biopsy were diagnosed with clinically insignificant cancers, suggesting that MRI could help to reduce the number of invasive biopsies and the associated pain, discomfort, and infection risks, the investigators stated in the New England Journal of Medicine.
“MRI, with or without targeted biopsy, led to fewer men undergoing biopsy, more clinically significant cancers being identified, less overdetection of clinically insignificant cancer, and fewer biopsy cores being obtained than did standard transrectal ultrasonography-guided biopsy,” they wrote.
Multiparametric MRI combines several different imaging modalities, including standard T1- and T2-weighted scans with dynamic contrast–enhanced and/or diffusion-weighted imaging to provide a wealth of information to aid in diagnosis. The technique has been shown in single-center studies to be similar or superior to ultrasound guided biopsy at detecting clinically significant cancers and minimizing detection of cancers that turn out to be clinically insignificant, the investigators said.
To add to the body of evidence, investigators from 25 centers in 11 countries randomized a total of 500 men with clinical suspicion of prostate cancer and no history of prostate biopsy to undergo either MRI plus targeted biopsy (not 10- or 12-core biopsy) if the scans indicated suspicion of malignancy, or standard transrectal ultrasound-guided biopsy with 10 or 12 core samples.
The investigators defined clinically significant cancer as the presence of a single biopsy core indicating disease of Gleason score 3 plus 4 (Gleason sum of 7), or greater.
Of the 252 men assigned to MRI, 71 (28%) had results that did not suggest prostate cancer, and these men did not undergo biopsy.
As noted before, MRI was noninferior to standard ultrasound-guided biopsy. In the MRI group, 95 men (38%) were determined to have clinically significant tumors, compared with 64 of 248 men (26%) in the standard biopsy group.
Getting down into the statistical weeds, the lower boundary of the 95% confidence interval for the difference was greater than −5%, showing that MRI with or without targeted biopsy was noninferior to standard transrectal ultrasonography-guided biopsy for the detection of clinically significant cancers. In fact, the 95% confidence interval for the 12-point difference between the two techniques (95% confidence interval, 4-20) showed that MRI was superior to standard biopsy, the authors stated.
There were no health-related quality of life differences at either 24 hours or 30 days after the procedure, and immediate postintervention discomfort and pain were also similar between the groups. However, patient-reported complications were lower in patients assigned to MRI, including blood in urine (30% vs. 63% for standard biopsy), blood in semen (32% vs. 60%), procedural-site pain (13% vs. 23%), rectal bleeding (14% vs. 22%), and erectile dysfunction (11% vs. 16%).
“We found that a diagnostic pathway including risk assessment with MRI before biopsy and MRI-targeted biopsy in the presence of a lesion suggestive of cancer was superior to the diagnostic pathway of standard transrectal ultrasonography-guided biopsy,” Dr. Kasivisvanathan and his associates concluded.
The study was supported by awards to Dr. Kasivisvanathan from the U.K. National Institute for Health Research and by the European Association of Urology Research Foundation. He reported no other significant conflicts of interest to disclose.
SOURCE: Kasivisvanathan V et al. N Engl J Med. 2018 Mar 19. doi: 10.1056/NEJMoa1801993.
FROM THE NEW ENGLAND JOURNAL OF MEDICINE
Key clinical point:
Major finding: Clinically significant cancers were detected in 38% of men on MRI vs. 26% on standard biopsy (P = .005).
Study details: A randomized noninferiority trial in 500 men with clinical suspicion of prostate cancer.
Disclosures: The study was supported by awards to Dr. Kasivisvanathan from the U.K. National Institute for Health Research and by the European Association of Urology Research Foundation. He reported no other significant conflicts of interest to disclose.
Source: Kasivisvanathan V et al. N Engl J Med. 2018 Mar 19. doi: 10.1056/NEJMoa1801993.









