User login
SABCS 2017: Top picks from Dr. William J. Gradishar
Oncology Practice Associate Editor William J. Gradishar, MD, reveals several anticipated studies from the 40th annual San Antonio Breast Cancer Symposium, set to begin on Wednesday, Dec. 6:
• GS1-01. Increasing the dose density of adjuvant chemotherapy by shortening intervals between courses or by sequential drug administration significantly reduces both disease recurrence and breast cancer mortality: an EBCTCG meta-analysis of 21,000 women in 16 randomized trials.
• GS4-02. Randomized comparison of adjuvant aromatase inhibitor exemestane plus ovarian function suppression vs. tamoxifen plus OFS in premenopausal women with hormone receptor–positive early breast cancer: Update of the combined TEXT and SOFT trials.
• GS1-03. Perioperative aromatase inhibitor treatment in determining or predicting long-term outcome in early breast cancer–the POETIC Trial.
• GS1-06. Extended adjuvant bisphosphonate treatment over 5 years in early breast cancer does not improve disease-free and overall survival, compared with 2 years of treatment: Phase III data from the SUCCESS A study.
• GS2-05. First-line ribociclib vs. placebo with goserelin and tamoxifen or a nonsteroidal aromatase inhibitor in premenopausal women with hormone receptor–positive, HER2-negative advanced breast cancer: Results from the randomized phase III MONALEESA-7 trial.
• GS2-06. Phase Ib/II study evaluating safety and efficacy of pembrolizumab and trastuzumab in patients with trastuzumab-resistant HER2-positive metastatic breast cancer: Results from the PANACEA (IBCSG 45-13/KEYNOTE-014) study.
• GS2-07. MANTA – A randomized phase 2 study of fulvestrant in combination with the dual mTOR inhibitor AZD2014 or everolimus or fulvestrant alone in estrogen receptor–positive advanced or metastatic breast cancer.
• GS3-01. A prospective, randomized, multicenter phase 3 trial of additional 2 versus additional 5 years of anastrozole after initial 5 years of adjuvant endocrine therapy – results from 3,484 postmenopausal women in the ABCSG-16 trial.
• GS3-02. Invasive disease-free survival and gene-expression signatures in CALGB (Alliance) 40601, a randomized phase III neoadjuvant trial of dual HER2 targeting with lapatinib added to chemotherapy plus trastuzumab.
• GS3-03. A phase III, multicenter, double-blind randomized trial of celecoxib versus placebo in primary breast cancer patients (REACT – Randomized European Celecoxib Trial).
• GS3-04. A randomized phase III study of adjuvant trastuzumab for a duration of 9 weeks versus 1 year, combined with adjuvant taxane-anthracycline chemotherapy, for early HER2-positive breast cancer (the SOLD study).
• GS3-06. Long-term follow-up of CALGB 40502/NCCTG N063H (Alliance): A randomized phase III trial of weekly paclitaxel, compared with weekly nanoparticle albumin-bound (nab)–paclitaxel or ixabepilone +/– bevacizumab as first-line therapy for locally recurrent or metastatic breast cancer.
• GS3-08. Pathological complete response predicts event-free and distant disease-free survival in the I-SPY2 TRIAL.
• GS4-01. Pooled analysis of five randomized trials investigating temporary ovarian suppression with gonadotropin-releasing hormone analogues during chemotherapy as a strategy to preserve ovarian function and fertility in premenopausal early breast cancer patients.
• GS4-02. Randomized comparison of adjuvant aromatase inhibitor exemestane plus ovarian function suppression vs. tamoxifen plus OFS in premenopausal women with hormone receptor–positive early breast cancer: Update of the combined TEXT and SOFT trials.
• GS4-03. Randomized comparison of adjuvant tamoxifen plus ovarian function suppression versus tamoxifen in premenopausal women with hormone receptor–positive early breast cancer: Update of the SOFT trial.
• GS4-04. Randomized blinded sham- and waitlist-controlled trial of acupuncture for joint symptoms related to aromatase inhibitors in women with early-stage breast cancer.
• GS5-03. Risk of arm morbidity after local therapy in young breast cancer survivors.
• GS5-05. Primary endocrine therapy for ER-positive ductal carcinoma in situ: CALGB 40903 (Alliance).
• GS6-01. Integration of clinical variables for the prediction of late distant recurrence in patients with estrogen receptor–positive breast cancer treated with 5 years of endocrine therapy.
• GS6-02. The benefit of abemaciclib in prognostic subgroups: An exploratory analysis of combined data from the MONARCH 2 and 3 studies.
• GS6-07. EMBRACA: A phase 3 trial comparing talazoparib, an oral PARP inhibitor, to physician’s choice of therapy in patients with advanced breast cancer and a germline BRCA mutation.
Dr. Gradishar is the Betsy Bramsen Professor of Breast Oncology, professor of medicine, and interim chief of the division of hematology/oncology, Northwestern University Feinberg School of Medicine, Chicago, and deputy director for the Clinical Network and director of the Maggie Daley Center for Women’s Cancer Care, Robert H. Lurie Comprehensive Cancer Center Network of Northwestern University, Chicago.
Oncology Practice Associate Editor William J. Gradishar, MD, reveals several anticipated studies from the 40th annual San Antonio Breast Cancer Symposium, set to begin on Wednesday, Dec. 6:
• GS1-01. Increasing the dose density of adjuvant chemotherapy by shortening intervals between courses or by sequential drug administration significantly reduces both disease recurrence and breast cancer mortality: an EBCTCG meta-analysis of 21,000 women in 16 randomized trials.
• GS4-02. Randomized comparison of adjuvant aromatase inhibitor exemestane plus ovarian function suppression vs. tamoxifen plus OFS in premenopausal women with hormone receptor–positive early breast cancer: Update of the combined TEXT and SOFT trials.
• GS1-03. Perioperative aromatase inhibitor treatment in determining or predicting long-term outcome in early breast cancer–the POETIC Trial.
• GS1-06. Extended adjuvant bisphosphonate treatment over 5 years in early breast cancer does not improve disease-free and overall survival, compared with 2 years of treatment: Phase III data from the SUCCESS A study.
• GS2-05. First-line ribociclib vs. placebo with goserelin and tamoxifen or a nonsteroidal aromatase inhibitor in premenopausal women with hormone receptor–positive, HER2-negative advanced breast cancer: Results from the randomized phase III MONALEESA-7 trial.
• GS2-06. Phase Ib/II study evaluating safety and efficacy of pembrolizumab and trastuzumab in patients with trastuzumab-resistant HER2-positive metastatic breast cancer: Results from the PANACEA (IBCSG 45-13/KEYNOTE-014) study.
• GS2-07. MANTA – A randomized phase 2 study of fulvestrant in combination with the dual mTOR inhibitor AZD2014 or everolimus or fulvestrant alone in estrogen receptor–positive advanced or metastatic breast cancer.
• GS3-01. A prospective, randomized, multicenter phase 3 trial of additional 2 versus additional 5 years of anastrozole after initial 5 years of adjuvant endocrine therapy – results from 3,484 postmenopausal women in the ABCSG-16 trial.
• GS3-02. Invasive disease-free survival and gene-expression signatures in CALGB (Alliance) 40601, a randomized phase III neoadjuvant trial of dual HER2 targeting with lapatinib added to chemotherapy plus trastuzumab.
• GS3-03. A phase III, multicenter, double-blind randomized trial of celecoxib versus placebo in primary breast cancer patients (REACT – Randomized European Celecoxib Trial).
• GS3-04. A randomized phase III study of adjuvant trastuzumab for a duration of 9 weeks versus 1 year, combined with adjuvant taxane-anthracycline chemotherapy, for early HER2-positive breast cancer (the SOLD study).
• GS3-06. Long-term follow-up of CALGB 40502/NCCTG N063H (Alliance): A randomized phase III trial of weekly paclitaxel, compared with weekly nanoparticle albumin-bound (nab)–paclitaxel or ixabepilone +/– bevacizumab as first-line therapy for locally recurrent or metastatic breast cancer.
• GS3-08. Pathological complete response predicts event-free and distant disease-free survival in the I-SPY2 TRIAL.
• GS4-01. Pooled analysis of five randomized trials investigating temporary ovarian suppression with gonadotropin-releasing hormone analogues during chemotherapy as a strategy to preserve ovarian function and fertility in premenopausal early breast cancer patients.
• GS4-02. Randomized comparison of adjuvant aromatase inhibitor exemestane plus ovarian function suppression vs. tamoxifen plus OFS in premenopausal women with hormone receptor–positive early breast cancer: Update of the combined TEXT and SOFT trials.
• GS4-03. Randomized comparison of adjuvant tamoxifen plus ovarian function suppression versus tamoxifen in premenopausal women with hormone receptor–positive early breast cancer: Update of the SOFT trial.
• GS4-04. Randomized blinded sham- and waitlist-controlled trial of acupuncture for joint symptoms related to aromatase inhibitors in women with early-stage breast cancer.
• GS5-03. Risk of arm morbidity after local therapy in young breast cancer survivors.
• GS5-05. Primary endocrine therapy for ER-positive ductal carcinoma in situ: CALGB 40903 (Alliance).
• GS6-01. Integration of clinical variables for the prediction of late distant recurrence in patients with estrogen receptor–positive breast cancer treated with 5 years of endocrine therapy.
• GS6-02. The benefit of abemaciclib in prognostic subgroups: An exploratory analysis of combined data from the MONARCH 2 and 3 studies.
• GS6-07. EMBRACA: A phase 3 trial comparing talazoparib, an oral PARP inhibitor, to physician’s choice of therapy in patients with advanced breast cancer and a germline BRCA mutation.
Dr. Gradishar is the Betsy Bramsen Professor of Breast Oncology, professor of medicine, and interim chief of the division of hematology/oncology, Northwestern University Feinberg School of Medicine, Chicago, and deputy director for the Clinical Network and director of the Maggie Daley Center for Women’s Cancer Care, Robert H. Lurie Comprehensive Cancer Center Network of Northwestern University, Chicago.
Oncology Practice Associate Editor William J. Gradishar, MD, reveals several anticipated studies from the 40th annual San Antonio Breast Cancer Symposium, set to begin on Wednesday, Dec. 6:
• GS1-01. Increasing the dose density of adjuvant chemotherapy by shortening intervals between courses or by sequential drug administration significantly reduces both disease recurrence and breast cancer mortality: an EBCTCG meta-analysis of 21,000 women in 16 randomized trials.
• GS4-02. Randomized comparison of adjuvant aromatase inhibitor exemestane plus ovarian function suppression vs. tamoxifen plus OFS in premenopausal women with hormone receptor–positive early breast cancer: Update of the combined TEXT and SOFT trials.
• GS1-03. Perioperative aromatase inhibitor treatment in determining or predicting long-term outcome in early breast cancer–the POETIC Trial.
• GS1-06. Extended adjuvant bisphosphonate treatment over 5 years in early breast cancer does not improve disease-free and overall survival, compared with 2 years of treatment: Phase III data from the SUCCESS A study.
• GS2-05. First-line ribociclib vs. placebo with goserelin and tamoxifen or a nonsteroidal aromatase inhibitor in premenopausal women with hormone receptor–positive, HER2-negative advanced breast cancer: Results from the randomized phase III MONALEESA-7 trial.
• GS2-06. Phase Ib/II study evaluating safety and efficacy of pembrolizumab and trastuzumab in patients with trastuzumab-resistant HER2-positive metastatic breast cancer: Results from the PANACEA (IBCSG 45-13/KEYNOTE-014) study.
• GS2-07. MANTA – A randomized phase 2 study of fulvestrant in combination with the dual mTOR inhibitor AZD2014 or everolimus or fulvestrant alone in estrogen receptor–positive advanced or metastatic breast cancer.
• GS3-01. A prospective, randomized, multicenter phase 3 trial of additional 2 versus additional 5 years of anastrozole after initial 5 years of adjuvant endocrine therapy – results from 3,484 postmenopausal women in the ABCSG-16 trial.
• GS3-02. Invasive disease-free survival and gene-expression signatures in CALGB (Alliance) 40601, a randomized phase III neoadjuvant trial of dual HER2 targeting with lapatinib added to chemotherapy plus trastuzumab.
• GS3-03. A phase III, multicenter, double-blind randomized trial of celecoxib versus placebo in primary breast cancer patients (REACT – Randomized European Celecoxib Trial).
• GS3-04. A randomized phase III study of adjuvant trastuzumab for a duration of 9 weeks versus 1 year, combined with adjuvant taxane-anthracycline chemotherapy, for early HER2-positive breast cancer (the SOLD study).
• GS3-06. Long-term follow-up of CALGB 40502/NCCTG N063H (Alliance): A randomized phase III trial of weekly paclitaxel, compared with weekly nanoparticle albumin-bound (nab)–paclitaxel or ixabepilone +/– bevacizumab as first-line therapy for locally recurrent or metastatic breast cancer.
• GS3-08. Pathological complete response predicts event-free and distant disease-free survival in the I-SPY2 TRIAL.
• GS4-01. Pooled analysis of five randomized trials investigating temporary ovarian suppression with gonadotropin-releasing hormone analogues during chemotherapy as a strategy to preserve ovarian function and fertility in premenopausal early breast cancer patients.
• GS4-02. Randomized comparison of adjuvant aromatase inhibitor exemestane plus ovarian function suppression vs. tamoxifen plus OFS in premenopausal women with hormone receptor–positive early breast cancer: Update of the combined TEXT and SOFT trials.
• GS4-03. Randomized comparison of adjuvant tamoxifen plus ovarian function suppression versus tamoxifen in premenopausal women with hormone receptor–positive early breast cancer: Update of the SOFT trial.
• GS4-04. Randomized blinded sham- and waitlist-controlled trial of acupuncture for joint symptoms related to aromatase inhibitors in women with early-stage breast cancer.
• GS5-03. Risk of arm morbidity after local therapy in young breast cancer survivors.
• GS5-05. Primary endocrine therapy for ER-positive ductal carcinoma in situ: CALGB 40903 (Alliance).
• GS6-01. Integration of clinical variables for the prediction of late distant recurrence in patients with estrogen receptor–positive breast cancer treated with 5 years of endocrine therapy.
• GS6-02. The benefit of abemaciclib in prognostic subgroups: An exploratory analysis of combined data from the MONARCH 2 and 3 studies.
• GS6-07. EMBRACA: A phase 3 trial comparing talazoparib, an oral PARP inhibitor, to physician’s choice of therapy in patients with advanced breast cancer and a germline BRCA mutation.
Dr. Gradishar is the Betsy Bramsen Professor of Breast Oncology, professor of medicine, and interim chief of the division of hematology/oncology, Northwestern University Feinberg School of Medicine, Chicago, and deputy director for the Clinical Network and director of the Maggie Daley Center for Women’s Cancer Care, Robert H. Lurie Comprehensive Cancer Center Network of Northwestern University, Chicago.
FROM SABCS 2017
Blisibimod shows mixed results for lupus in phase 3 trial
SAN DIEGO – While use of the investigational agent blisibimod did not meet the primary endpoint in a phase 3 trial in systemic lupus erythematosus (SLE), it was associated with steroid sparing, decreased urine protein, a trend toward decreased anti–double-stranded DNA antibodies, and significant decreases in anticardiolipin antibodies and immunoglobulin levels.
Those results were reported from CHABLIS-SC1 trial, a randomized (5:4), double-blind, placebo-controlled phase 3 study of 442 patients, and were presented by Joan T. Merrill, MD, at the annual meeting of the American College of Rheumatology. Blisibimod is a subcutaneously injected inhibitor of B-cell activating factor.
The SRI-6 primary endpoint at 52 weeks was met by 44% of patients on blisibimod, compared with 42% on placebo. But the SRI-6 endpoint widened when the most common features at entry were excluded (mucocutaneous and musculoskeletal), suggesting the possibility that blisibimod affects the more objective and potentially organ-threatening renal endpoint.
To be eligible for the trial, patients had to have a Safety of Estrogen in Lupus Erythematosus National Assessment Group–SLE Disease Activity Index (SELENA-SLEDAI) score of 10 or greater and be receiving steroids. The SRI-6 primary endpoint also required no worsening on the British Isles Lupus Assessment Group (BILAG) disease activity index or Physician Global Assessment. Key secondary endpoints included proteinuria and achievement of steroid taper.
Patients in the study were required to be receiving stable doses of prednisone or an equivalent steroid at less than or equal to 0.5 mg/kg or 40 mg daily for at least 28 days prior to randomization. Other permitted standard-of-care oral medications included methotrexate up to 25 mg weekly, azathioprine up to 300 mg daily, mycophenolate mofetil or sodium salt up to 3 mg daily, leflunomide up to 40 mg daily, hydroxychloroquine up to 400 mg daily, and nonsteroidal drugs within locally approved dose ranges.
Of the 442 patients enrolled, 245 received blisibimod, and 197 received placebo. They were well matched in demographics and baseline disease characteristics. “There were very few patients of African descent in this trial,” Dr. Merrill noted. “Also, about 30% of patients had some renal involvement. In fact, patients with stable, active renal disease were encouraged to participate in this trial.” The mean prednisone dose at entry was between 15 and 16 mg daily, and about 60% of patients were taking an antimalarial. An equal proportion of patients in both groups discontinued the study (22%). “A few more patients withdrew due to adverse events in the blisibimod group, and a few more patients withdrew due to lack of efficacy in the placebo group,” she said.
Of 135 patients with a baseline urine protein-to-creatinine ratio equal to or greater than 0.5, blisibimod treatment led to significantly greater improvement in proteinuria at several time points than did treatment with placebo, which also showed improvement in proteinuria.
Treatment with blisibimod also was associated with a reduction in anti–double-stranded DNA antibodies, as well as significant reductions in peripheral B-cell lineages, anticardiolipin antibodies, and immunoglobulins, and with significant increases in complement C3 and C4. “The expected pharmacodynamic markers were [also] met ... and more patients treated with blisibimod were able to achieve a prednisone milestone of reduction to less than or equal to 7.5 mg/day, compared with those in the placebo group,” she said. “This was statistically significant over time at multiple time points.”
Adverse events were balanced between treatment arms except for injection site reactions, which occurred in 7.3% of blisibimod-treated patients versus 2.6% of placebo patients. There were no major safety issues in the study.
Dr. Merrill hypothesized that the higher mean doses of corticosteroid at baseline could have contributed to the higher-than-usual placebo response rates and failure to meet the primary SRI-6 endpoint.
The study was supported by Anthera. Dr. Merrill disclosed that she has received research support from Anthera, Amgen, EMD Serono, GlaxoSmithKline. and Novartis.
SAN DIEGO – While use of the investigational agent blisibimod did not meet the primary endpoint in a phase 3 trial in systemic lupus erythematosus (SLE), it was associated with steroid sparing, decreased urine protein, a trend toward decreased anti–double-stranded DNA antibodies, and significant decreases in anticardiolipin antibodies and immunoglobulin levels.
Those results were reported from CHABLIS-SC1 trial, a randomized (5:4), double-blind, placebo-controlled phase 3 study of 442 patients, and were presented by Joan T. Merrill, MD, at the annual meeting of the American College of Rheumatology. Blisibimod is a subcutaneously injected inhibitor of B-cell activating factor.
The SRI-6 primary endpoint at 52 weeks was met by 44% of patients on blisibimod, compared with 42% on placebo. But the SRI-6 endpoint widened when the most common features at entry were excluded (mucocutaneous and musculoskeletal), suggesting the possibility that blisibimod affects the more objective and potentially organ-threatening renal endpoint.
To be eligible for the trial, patients had to have a Safety of Estrogen in Lupus Erythematosus National Assessment Group–SLE Disease Activity Index (SELENA-SLEDAI) score of 10 or greater and be receiving steroids. The SRI-6 primary endpoint also required no worsening on the British Isles Lupus Assessment Group (BILAG) disease activity index or Physician Global Assessment. Key secondary endpoints included proteinuria and achievement of steroid taper.
Patients in the study were required to be receiving stable doses of prednisone or an equivalent steroid at less than or equal to 0.5 mg/kg or 40 mg daily for at least 28 days prior to randomization. Other permitted standard-of-care oral medications included methotrexate up to 25 mg weekly, azathioprine up to 300 mg daily, mycophenolate mofetil or sodium salt up to 3 mg daily, leflunomide up to 40 mg daily, hydroxychloroquine up to 400 mg daily, and nonsteroidal drugs within locally approved dose ranges.
Of the 442 patients enrolled, 245 received blisibimod, and 197 received placebo. They were well matched in demographics and baseline disease characteristics. “There were very few patients of African descent in this trial,” Dr. Merrill noted. “Also, about 30% of patients had some renal involvement. In fact, patients with stable, active renal disease were encouraged to participate in this trial.” The mean prednisone dose at entry was between 15 and 16 mg daily, and about 60% of patients were taking an antimalarial. An equal proportion of patients in both groups discontinued the study (22%). “A few more patients withdrew due to adverse events in the blisibimod group, and a few more patients withdrew due to lack of efficacy in the placebo group,” she said.
Of 135 patients with a baseline urine protein-to-creatinine ratio equal to or greater than 0.5, blisibimod treatment led to significantly greater improvement in proteinuria at several time points than did treatment with placebo, which also showed improvement in proteinuria.
Treatment with blisibimod also was associated with a reduction in anti–double-stranded DNA antibodies, as well as significant reductions in peripheral B-cell lineages, anticardiolipin antibodies, and immunoglobulins, and with significant increases in complement C3 and C4. “The expected pharmacodynamic markers were [also] met ... and more patients treated with blisibimod were able to achieve a prednisone milestone of reduction to less than or equal to 7.5 mg/day, compared with those in the placebo group,” she said. “This was statistically significant over time at multiple time points.”
Adverse events were balanced between treatment arms except for injection site reactions, which occurred in 7.3% of blisibimod-treated patients versus 2.6% of placebo patients. There were no major safety issues in the study.
Dr. Merrill hypothesized that the higher mean doses of corticosteroid at baseline could have contributed to the higher-than-usual placebo response rates and failure to meet the primary SRI-6 endpoint.
The study was supported by Anthera. Dr. Merrill disclosed that she has received research support from Anthera, Amgen, EMD Serono, GlaxoSmithKline. and Novartis.
SAN DIEGO – While use of the investigational agent blisibimod did not meet the primary endpoint in a phase 3 trial in systemic lupus erythematosus (SLE), it was associated with steroid sparing, decreased urine protein, a trend toward decreased anti–double-stranded DNA antibodies, and significant decreases in anticardiolipin antibodies and immunoglobulin levels.
Those results were reported from CHABLIS-SC1 trial, a randomized (5:4), double-blind, placebo-controlled phase 3 study of 442 patients, and were presented by Joan T. Merrill, MD, at the annual meeting of the American College of Rheumatology. Blisibimod is a subcutaneously injected inhibitor of B-cell activating factor.
The SRI-6 primary endpoint at 52 weeks was met by 44% of patients on blisibimod, compared with 42% on placebo. But the SRI-6 endpoint widened when the most common features at entry were excluded (mucocutaneous and musculoskeletal), suggesting the possibility that blisibimod affects the more objective and potentially organ-threatening renal endpoint.
To be eligible for the trial, patients had to have a Safety of Estrogen in Lupus Erythematosus National Assessment Group–SLE Disease Activity Index (SELENA-SLEDAI) score of 10 or greater and be receiving steroids. The SRI-6 primary endpoint also required no worsening on the British Isles Lupus Assessment Group (BILAG) disease activity index or Physician Global Assessment. Key secondary endpoints included proteinuria and achievement of steroid taper.
Patients in the study were required to be receiving stable doses of prednisone or an equivalent steroid at less than or equal to 0.5 mg/kg or 40 mg daily for at least 28 days prior to randomization. Other permitted standard-of-care oral medications included methotrexate up to 25 mg weekly, azathioprine up to 300 mg daily, mycophenolate mofetil or sodium salt up to 3 mg daily, leflunomide up to 40 mg daily, hydroxychloroquine up to 400 mg daily, and nonsteroidal drugs within locally approved dose ranges.
Of the 442 patients enrolled, 245 received blisibimod, and 197 received placebo. They were well matched in demographics and baseline disease characteristics. “There were very few patients of African descent in this trial,” Dr. Merrill noted. “Also, about 30% of patients had some renal involvement. In fact, patients with stable, active renal disease were encouraged to participate in this trial.” The mean prednisone dose at entry was between 15 and 16 mg daily, and about 60% of patients were taking an antimalarial. An equal proportion of patients in both groups discontinued the study (22%). “A few more patients withdrew due to adverse events in the blisibimod group, and a few more patients withdrew due to lack of efficacy in the placebo group,” she said.
Of 135 patients with a baseline urine protein-to-creatinine ratio equal to or greater than 0.5, blisibimod treatment led to significantly greater improvement in proteinuria at several time points than did treatment with placebo, which also showed improvement in proteinuria.
Treatment with blisibimod also was associated with a reduction in anti–double-stranded DNA antibodies, as well as significant reductions in peripheral B-cell lineages, anticardiolipin antibodies, and immunoglobulins, and with significant increases in complement C3 and C4. “The expected pharmacodynamic markers were [also] met ... and more patients treated with blisibimod were able to achieve a prednisone milestone of reduction to less than or equal to 7.5 mg/day, compared with those in the placebo group,” she said. “This was statistically significant over time at multiple time points.”
Adverse events were balanced between treatment arms except for injection site reactions, which occurred in 7.3% of blisibimod-treated patients versus 2.6% of placebo patients. There were no major safety issues in the study.
Dr. Merrill hypothesized that the higher mean doses of corticosteroid at baseline could have contributed to the higher-than-usual placebo response rates and failure to meet the primary SRI-6 endpoint.
The study was supported by Anthera. Dr. Merrill disclosed that she has received research support from Anthera, Amgen, EMD Serono, GlaxoSmithKline. and Novartis.
AT ACR 2017
Key clinical point:
Major finding: The primary endpoint of CHABLIS-SC1 was not met, but expected pharmacodynamic markers were.
Study details: A phase 3 study in which 245 SLE patients received blisibimod and 197 received placebo.
Disclosures: The study was supported by Anthera Pharmaceuticals. Dr. Merrill disclosed that she has received research support from Anthera, Amgen, EMD Serono, GlaxoSmithKline, and Novartis.
Topical 5-Fluorouracil Made Easy?
What is the recent research behind 5-fluorouracil cream 5% combined with calcipotriol ointment 0.005% for actinic keratoses?
Cunningham et al published a randomized double-blind study in which 131 patients with actinic keratoses (AKs) were assigned to either 5-fluorouracil (5-FU) cream 5% combined with calcipotriol (calcipotriene) ointment 0.005% twice daily to the face, scalp, and arms for 4 days, or 5-FU 5% combined with petrolatum applied in the same fashion. There was an 87.8% versus 26.3% mean reduction in the number of AKs and less severe pain, crusting, and ulceration in the study cohort compared to the 5-FU plus petrolatum group.
The same study also investigated immune parameters in these patients and found that the study group preferentially displayed activated thymic stromal lymphopoietin and a CD4 T cell-mediated reaction, among other effects. In prior studies, thymic stromal lymphopoietin has been shown to be upregulated in barrier-defective skin, displays antitumor activity, and is enhanced by topical calcipotriol application based on its original indication for psoriasis.
How do these study results impact patient care?
In a perfect world, every patient could tolerate and afford chemopreventative measures such as 5-FU cream, apply it diffusely to sun-exposed skin, and experience no severe irritant reactions and/or social pariah status. We all know that this product is effective, and we all overprepare patients to use it, knowing that they will call our offices panicked and fearful that they are allergic to or are becoming infected by this cream.
Although further study clearly is needed to determine the optimal application amount, duration of use, and vehicle mix, this new compound utilizing 2 topicals that are familiar to us--5-FU cream approved for AKs and early squamous cell skin cancers and calcipotriol ointment (though available only in cream in the United States currently) for psoriasis--is an encouraging step. Home therapy for AKs and possibly early nonmelanoma skin cancers that is more tolerable, of shorter duration, and in turn more effective than the current options would lessen the burden of treating these lesions surgically or rescheduling 5-FU patients often for irritation reaction education.
How do patients respond to this regimen?
In my own anecdotal experience, this regimen has been well received by patients and often is covered by most insurances when written as 2 separate prescriptions (both in cream vehicle). They still report some irritation, but I prefer to utilize it segmentally instead of treating all sun-exposed areas at once (ie, treat one side of the face/scalp twice daily for 4 days, then the other, or even divide it into smaller segments once the prior segment has healed). This combination, in addition to, for example, adding nicotinamide 500 mg twice daily to a patient's skin cancer chemopreventative sequence, is in my opinion a novel but safe, effective, and well-tolerated field therapy recommendation.
American Academy of Dermatology Actinic Keratosis Overview
American Academy of Family Physicians Actinic Keratoses Information
Suggested Readings
- Cunningham TJ, Tabacchi M, Eliane JP, et al. Randomized trial of calcipotriol combined with 5-fluorouracil for skin cancer precursor immunotherapy. J Clin Invest. 2017;127:106-116.
- Demehri S, Turkoz A, Manivasagam S, et al. Elevated epidermal thymic stromal lymphopoietin levels establish an antitumor environment in the skin. Cancer Cell. 2012;22:494-505.
- Rosamilia LL. Three Cheers for B3? Cutis. July 7, 2015. http://www.mdedge.com/cutis/article/101102/nonmelanoma-skin-cancer/three-cheers-b3. Accessed November 20, 2017.
- Sato-Deguchi E, Imafuku S, Chou B, et al. Topical vitamin D(3) analogues induce thymic stromal lymphopoietin and cathelicidin in psoriatic skin lesions. Br J Dermatol. 2012;167:77-84.
What is the recent research behind 5-fluorouracil cream 5% combined with calcipotriol ointment 0.005% for actinic keratoses?
Cunningham et al published a randomized double-blind study in which 131 patients with actinic keratoses (AKs) were assigned to either 5-fluorouracil (5-FU) cream 5% combined with calcipotriol (calcipotriene) ointment 0.005% twice daily to the face, scalp, and arms for 4 days, or 5-FU 5% combined with petrolatum applied in the same fashion. There was an 87.8% versus 26.3% mean reduction in the number of AKs and less severe pain, crusting, and ulceration in the study cohort compared to the 5-FU plus petrolatum group.
The same study also investigated immune parameters in these patients and found that the study group preferentially displayed activated thymic stromal lymphopoietin and a CD4 T cell-mediated reaction, among other effects. In prior studies, thymic stromal lymphopoietin has been shown to be upregulated in barrier-defective skin, displays antitumor activity, and is enhanced by topical calcipotriol application based on its original indication for psoriasis.
How do these study results impact patient care?
In a perfect world, every patient could tolerate and afford chemopreventative measures such as 5-FU cream, apply it diffusely to sun-exposed skin, and experience no severe irritant reactions and/or social pariah status. We all know that this product is effective, and we all overprepare patients to use it, knowing that they will call our offices panicked and fearful that they are allergic to or are becoming infected by this cream.
Although further study clearly is needed to determine the optimal application amount, duration of use, and vehicle mix, this new compound utilizing 2 topicals that are familiar to us--5-FU cream approved for AKs and early squamous cell skin cancers and calcipotriol ointment (though available only in cream in the United States currently) for psoriasis--is an encouraging step. Home therapy for AKs and possibly early nonmelanoma skin cancers that is more tolerable, of shorter duration, and in turn more effective than the current options would lessen the burden of treating these lesions surgically or rescheduling 5-FU patients often for irritation reaction education.
How do patients respond to this regimen?
In my own anecdotal experience, this regimen has been well received by patients and often is covered by most insurances when written as 2 separate prescriptions (both in cream vehicle). They still report some irritation, but I prefer to utilize it segmentally instead of treating all sun-exposed areas at once (ie, treat one side of the face/scalp twice daily for 4 days, then the other, or even divide it into smaller segments once the prior segment has healed). This combination, in addition to, for example, adding nicotinamide 500 mg twice daily to a patient's skin cancer chemopreventative sequence, is in my opinion a novel but safe, effective, and well-tolerated field therapy recommendation.
American Academy of Dermatology Actinic Keratosis Overview
American Academy of Family Physicians Actinic Keratoses Information
Suggested Readings
- Cunningham TJ, Tabacchi M, Eliane JP, et al. Randomized trial of calcipotriol combined with 5-fluorouracil for skin cancer precursor immunotherapy. J Clin Invest. 2017;127:106-116.
- Demehri S, Turkoz A, Manivasagam S, et al. Elevated epidermal thymic stromal lymphopoietin levels establish an antitumor environment in the skin. Cancer Cell. 2012;22:494-505.
- Rosamilia LL. Three Cheers for B3? Cutis. July 7, 2015. http://www.mdedge.com/cutis/article/101102/nonmelanoma-skin-cancer/three-cheers-b3. Accessed November 20, 2017.
- Sato-Deguchi E, Imafuku S, Chou B, et al. Topical vitamin D(3) analogues induce thymic stromal lymphopoietin and cathelicidin in psoriatic skin lesions. Br J Dermatol. 2012;167:77-84.
What is the recent research behind 5-fluorouracil cream 5% combined with calcipotriol ointment 0.005% for actinic keratoses?
Cunningham et al published a randomized double-blind study in which 131 patients with actinic keratoses (AKs) were assigned to either 5-fluorouracil (5-FU) cream 5% combined with calcipotriol (calcipotriene) ointment 0.005% twice daily to the face, scalp, and arms for 4 days, or 5-FU 5% combined with petrolatum applied in the same fashion. There was an 87.8% versus 26.3% mean reduction in the number of AKs and less severe pain, crusting, and ulceration in the study cohort compared to the 5-FU plus petrolatum group.
The same study also investigated immune parameters in these patients and found that the study group preferentially displayed activated thymic stromal lymphopoietin and a CD4 T cell-mediated reaction, among other effects. In prior studies, thymic stromal lymphopoietin has been shown to be upregulated in barrier-defective skin, displays antitumor activity, and is enhanced by topical calcipotriol application based on its original indication for psoriasis.
How do these study results impact patient care?
In a perfect world, every patient could tolerate and afford chemopreventative measures such as 5-FU cream, apply it diffusely to sun-exposed skin, and experience no severe irritant reactions and/or social pariah status. We all know that this product is effective, and we all overprepare patients to use it, knowing that they will call our offices panicked and fearful that they are allergic to or are becoming infected by this cream.
Although further study clearly is needed to determine the optimal application amount, duration of use, and vehicle mix, this new compound utilizing 2 topicals that are familiar to us--5-FU cream approved for AKs and early squamous cell skin cancers and calcipotriol ointment (though available only in cream in the United States currently) for psoriasis--is an encouraging step. Home therapy for AKs and possibly early nonmelanoma skin cancers that is more tolerable, of shorter duration, and in turn more effective than the current options would lessen the burden of treating these lesions surgically or rescheduling 5-FU patients often for irritation reaction education.
How do patients respond to this regimen?
In my own anecdotal experience, this regimen has been well received by patients and often is covered by most insurances when written as 2 separate prescriptions (both in cream vehicle). They still report some irritation, but I prefer to utilize it segmentally instead of treating all sun-exposed areas at once (ie, treat one side of the face/scalp twice daily for 4 days, then the other, or even divide it into smaller segments once the prior segment has healed). This combination, in addition to, for example, adding nicotinamide 500 mg twice daily to a patient's skin cancer chemopreventative sequence, is in my opinion a novel but safe, effective, and well-tolerated field therapy recommendation.
American Academy of Dermatology Actinic Keratosis Overview
American Academy of Family Physicians Actinic Keratoses Information
Suggested Readings
- Cunningham TJ, Tabacchi M, Eliane JP, et al. Randomized trial of calcipotriol combined with 5-fluorouracil for skin cancer precursor immunotherapy. J Clin Invest. 2017;127:106-116.
- Demehri S, Turkoz A, Manivasagam S, et al. Elevated epidermal thymic stromal lymphopoietin levels establish an antitumor environment in the skin. Cancer Cell. 2012;22:494-505.
- Rosamilia LL. Three Cheers for B3? Cutis. July 7, 2015. http://www.mdedge.com/cutis/article/101102/nonmelanoma-skin-cancer/three-cheers-b3. Accessed November 20, 2017.
- Sato-Deguchi E, Imafuku S, Chou B, et al. Topical vitamin D(3) analogues induce thymic stromal lymphopoietin and cathelicidin in psoriatic skin lesions. Br J Dermatol. 2012;167:77-84.
Diversity in the Dermatology Workforce: 2017 Status Update
Physician diversity benefits patient care: Patients are more satisfied during race-concordant visits, report their physicians as more engaged and responsive to their needs, and experience notably longer visits.1,2 Nonwhite physicians (ie, races and ethnicities that are underrepresented in medicine [URM] with respect to the general population) are more likely to care for underserved communities. Furthermore, increased diversity in the learning environment supports preparedness of all trainees to serve diverse patients.3 For these reasons, a more diverse physician workforce can contribute to better access to care in all communities, thus addressing health disparities.1,4
Increasing diversity in the dermatology workforce has been identified as an emerging priority.5 Dermatology is one of the least diverse specialties,5 and the representation of URM dermatologists is lower compared to other medical specialties and the general US population. The proportion of specialty leaders from underrepresented backgrounds may be even smaller. The lack of diversity in academic dermatology has negative consequences for patients and communities. Increasing the diversity of resident trainees is the only way to improve the diversity gap within the dermatology workforce.6
Recent commentary on this topic has highlighted several priorities for addressing the dermatology diversity gap,6-11 including the following: (1) making diversity an explicit goal in dermatology; (2) ensuring early exposure to dermatology in medical school; (3) supporting mentorship programs for minority medical students; (4) increasing medical student diversity; (5) encouraging that all dermatology program directors and leaders train in implicit bias; and (6) reviewing residency admission criteria to ensure they are objective and equitable, not biased against any applicants.
The process of reviewing residency selection criteria has begun. In 2017, Chen and Shinkai7 called for our specialty to rethink the selection process. The authors argued that emphasis on test scores, grades, and publications systematically disadvantages underrepresented minorities and students from lower socioeconomic statuses. The authors proposed several solutions: (1) make diversity an explicit goal of the selection process, (2) shift away from test scores for all applicants, (3) change the interview format, (4) prioritize other competencies such as observation skills, and (5) recruit and retain faculty who support URM trainees.7
Several dermatology leadership groups have taken action to promote programs that aim to improve diversity within dermatology. The Dermatology Diversity Champions initiative includes 6 US dermatology residency programs that are committed to increasing diversity and collaborate to evaluate pilot approaches. The American Academy of Dermatology President’s Conference on Diversity in Dermatology in Chicago, Illinois, in August 2017, as well as the focus on diversity in residency training programs at the Annual Meeting of the Association of Professors of Dermatology in Chicago, Illinois, in October 2017, are strong indicators that our specialty as a whole is aware and eager to embrace diversity as a priority. The American Academy of Dermatology President’s Conference, which was comprised of representatives from many leadership organizations and interest groups within dermatology, identified 3 action items: (1) increase the pipeline of URM students into medical school, (2) increase interest in dermatology among URM medical students, and (3) increase URM representation in residency training programs.
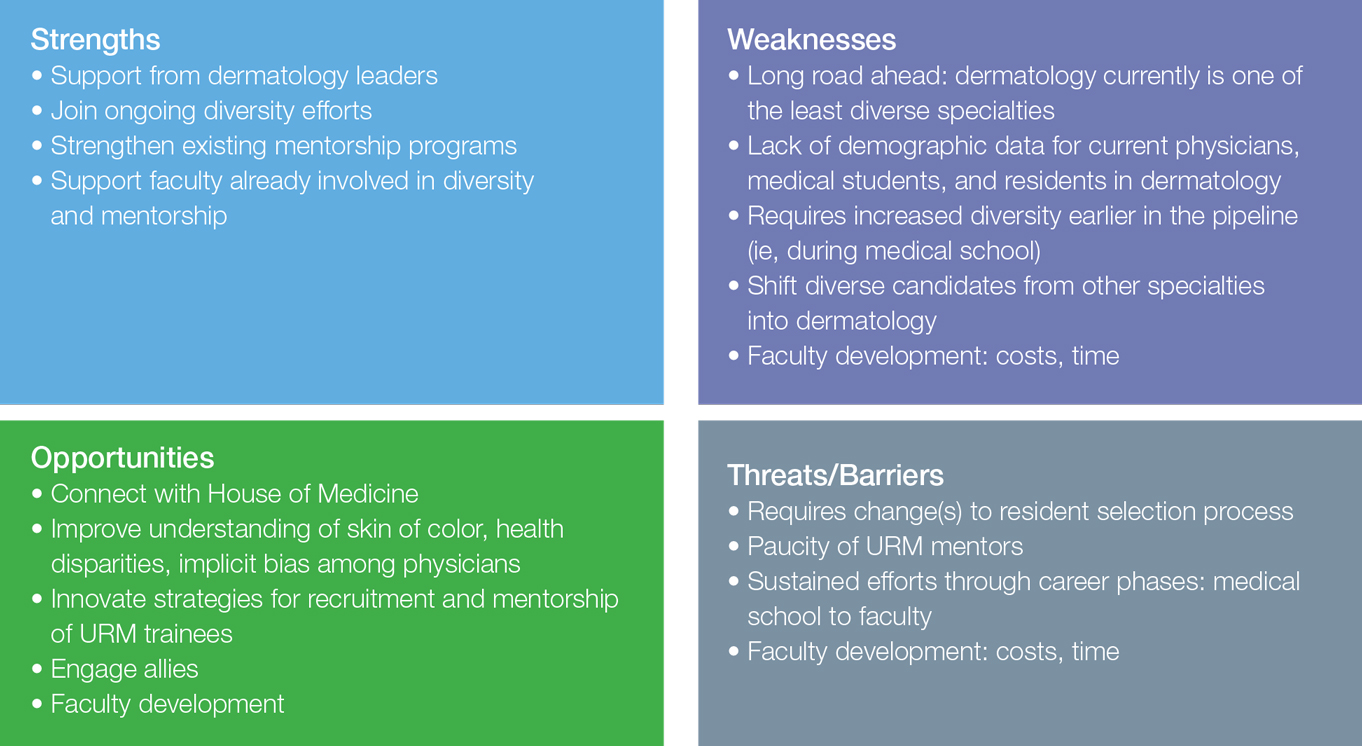
There are many strengths, weaknesses, opportunities, and threats/barriers (SWOT) to attaining this goal. Current strengths include strong support from dermatology leaders and activities that build on existing mentorship and diversity efforts by leaders within our specialty. SWOT analysis highlights several key opportunities of this mission, including connecting with the House of Medicine in shared efforts to improve diversity, as well as increased understanding of skin of color, health disparities, and implicit bias among physicians. Although faculty development will require time and financial investment, it will lead to tremendous benefits and opportunities for all dermatologists, including URM physicians. Other weaknesses and threats/barriers are outlined in the Figure.
Final Thoughts
We are far from reaching our goal of a diverse dermatology workforce, and the road ahead is long. We have a start and we have momentum. We can move forward by spreading the word that all types of diversity are a priority for our specialty. Making a true difference will require commitment and sustained efforts. Dermatology can lead the way as all of American medicine strives to attain workforce diversity.
- Saha S. Taking diversity seriously: the merits of increasing minority representation in medicine. JAMA Intern Med. 2014;174:291-292.
- Cooper LA, Roter DL, Johnson RL, et al. Patient-centered communication, ratings of care, and concordance of patient and physician race. Ann Intern Med. 2003;139:907-915.
- Saha S, Guiton G, Wimmers PF, et al. Student body racial and ethnic composition and diversity-related outcomes in US medical schools. JAMA. 2008;300:1135-1145.
- Marrast LM, Zallman L, Woolhandler S, et al. Minority physicians’ role in the care of underserved patients: diversifying the physician workforce may be key in addressing health disparities. JAMA Intern Med. 2014;174:289-291.
- Pandya AG, Alexis AF, Berger TG, et al. Increasing racial and ethnic diversity in dermatology: a call to action. J Am Acad Dermatol. 2016;74:584-587.
- Lester J, Wintroub B, Linos E. Disparities in academic dermatology. JAMA Dermatol. 2016;152:878-879.
- Chen A, Shinkai K. Rethinking how we select dermatology applicants—turning the tide. JAMA Dermatol. 2017;153:259-260.
- Granstein RD, Cornelius L, Shinkai K. Diversity in dermatology—a call for action. JAMA Dermatol. 2017;153:499-500.
- McKesey J, Berger TG, Lim HW, et al. Cultural competence for the 21st century dermatologist practicing in the United States. J Am Acad Dermatol. 2017;77:1159-1169.
- Van Voorhees AS, Enos CW. Diversity in dermatology residency programs. J Investig Dermatol Symp Proc. 2017;18:S46-S49.
- Imadojemu S, James WD. Increasing African American representation in dermatology. JAMA Dermatol. 2016;152:15-16.
Physician diversity benefits patient care: Patients are more satisfied during race-concordant visits, report their physicians as more engaged and responsive to their needs, and experience notably longer visits.1,2 Nonwhite physicians (ie, races and ethnicities that are underrepresented in medicine [URM] with respect to the general population) are more likely to care for underserved communities. Furthermore, increased diversity in the learning environment supports preparedness of all trainees to serve diverse patients.3 For these reasons, a more diverse physician workforce can contribute to better access to care in all communities, thus addressing health disparities.1,4
Increasing diversity in the dermatology workforce has been identified as an emerging priority.5 Dermatology is one of the least diverse specialties,5 and the representation of URM dermatologists is lower compared to other medical specialties and the general US population. The proportion of specialty leaders from underrepresented backgrounds may be even smaller. The lack of diversity in academic dermatology has negative consequences for patients and communities. Increasing the diversity of resident trainees is the only way to improve the diversity gap within the dermatology workforce.6
Recent commentary on this topic has highlighted several priorities for addressing the dermatology diversity gap,6-11 including the following: (1) making diversity an explicit goal in dermatology; (2) ensuring early exposure to dermatology in medical school; (3) supporting mentorship programs for minority medical students; (4) increasing medical student diversity; (5) encouraging that all dermatology program directors and leaders train in implicit bias; and (6) reviewing residency admission criteria to ensure they are objective and equitable, not biased against any applicants.
The process of reviewing residency selection criteria has begun. In 2017, Chen and Shinkai7 called for our specialty to rethink the selection process. The authors argued that emphasis on test scores, grades, and publications systematically disadvantages underrepresented minorities and students from lower socioeconomic statuses. The authors proposed several solutions: (1) make diversity an explicit goal of the selection process, (2) shift away from test scores for all applicants, (3) change the interview format, (4) prioritize other competencies such as observation skills, and (5) recruit and retain faculty who support URM trainees.7
Several dermatology leadership groups have taken action to promote programs that aim to improve diversity within dermatology. The Dermatology Diversity Champions initiative includes 6 US dermatology residency programs that are committed to increasing diversity and collaborate to evaluate pilot approaches. The American Academy of Dermatology President’s Conference on Diversity in Dermatology in Chicago, Illinois, in August 2017, as well as the focus on diversity in residency training programs at the Annual Meeting of the Association of Professors of Dermatology in Chicago, Illinois, in October 2017, are strong indicators that our specialty as a whole is aware and eager to embrace diversity as a priority. The American Academy of Dermatology President’s Conference, which was comprised of representatives from many leadership organizations and interest groups within dermatology, identified 3 action items: (1) increase the pipeline of URM students into medical school, (2) increase interest in dermatology among URM medical students, and (3) increase URM representation in residency training programs.
There are many strengths, weaknesses, opportunities, and threats/barriers (SWOT) to attaining this goal. Current strengths include strong support from dermatology leaders and activities that build on existing mentorship and diversity efforts by leaders within our specialty. SWOT analysis highlights several key opportunities of this mission, including connecting with the House of Medicine in shared efforts to improve diversity, as well as increased understanding of skin of color, health disparities, and implicit bias among physicians. Although faculty development will require time and financial investment, it will lead to tremendous benefits and opportunities for all dermatologists, including URM physicians. Other weaknesses and threats/barriers are outlined in the Figure.

Final Thoughts
We are far from reaching our goal of a diverse dermatology workforce, and the road ahead is long. We have a start and we have momentum. We can move forward by spreading the word that all types of diversity are a priority for our specialty. Making a true difference will require commitment and sustained efforts. Dermatology can lead the way as all of American medicine strives to attain workforce diversity.
Physician diversity benefits patient care: Patients are more satisfied during race-concordant visits, report their physicians as more engaged and responsive to their needs, and experience notably longer visits.1,2 Nonwhite physicians (ie, races and ethnicities that are underrepresented in medicine [URM] with respect to the general population) are more likely to care for underserved communities. Furthermore, increased diversity in the learning environment supports preparedness of all trainees to serve diverse patients.3 For these reasons, a more diverse physician workforce can contribute to better access to care in all communities, thus addressing health disparities.1,4
Increasing diversity in the dermatology workforce has been identified as an emerging priority.5 Dermatology is one of the least diverse specialties,5 and the representation of URM dermatologists is lower compared to other medical specialties and the general US population. The proportion of specialty leaders from underrepresented backgrounds may be even smaller. The lack of diversity in academic dermatology has negative consequences for patients and communities. Increasing the diversity of resident trainees is the only way to improve the diversity gap within the dermatology workforce.6
Recent commentary on this topic has highlighted several priorities for addressing the dermatology diversity gap,6-11 including the following: (1) making diversity an explicit goal in dermatology; (2) ensuring early exposure to dermatology in medical school; (3) supporting mentorship programs for minority medical students; (4) increasing medical student diversity; (5) encouraging that all dermatology program directors and leaders train in implicit bias; and (6) reviewing residency admission criteria to ensure they are objective and equitable, not biased against any applicants.
The process of reviewing residency selection criteria has begun. In 2017, Chen and Shinkai7 called for our specialty to rethink the selection process. The authors argued that emphasis on test scores, grades, and publications systematically disadvantages underrepresented minorities and students from lower socioeconomic statuses. The authors proposed several solutions: (1) make diversity an explicit goal of the selection process, (2) shift away from test scores for all applicants, (3) change the interview format, (4) prioritize other competencies such as observation skills, and (5) recruit and retain faculty who support URM trainees.7
Several dermatology leadership groups have taken action to promote programs that aim to improve diversity within dermatology. The Dermatology Diversity Champions initiative includes 6 US dermatology residency programs that are committed to increasing diversity and collaborate to evaluate pilot approaches. The American Academy of Dermatology President’s Conference on Diversity in Dermatology in Chicago, Illinois, in August 2017, as well as the focus on diversity in residency training programs at the Annual Meeting of the Association of Professors of Dermatology in Chicago, Illinois, in October 2017, are strong indicators that our specialty as a whole is aware and eager to embrace diversity as a priority. The American Academy of Dermatology President’s Conference, which was comprised of representatives from many leadership organizations and interest groups within dermatology, identified 3 action items: (1) increase the pipeline of URM students into medical school, (2) increase interest in dermatology among URM medical students, and (3) increase URM representation in residency training programs.
There are many strengths, weaknesses, opportunities, and threats/barriers (SWOT) to attaining this goal. Current strengths include strong support from dermatology leaders and activities that build on existing mentorship and diversity efforts by leaders within our specialty. SWOT analysis highlights several key opportunities of this mission, including connecting with the House of Medicine in shared efforts to improve diversity, as well as increased understanding of skin of color, health disparities, and implicit bias among physicians. Although faculty development will require time and financial investment, it will lead to tremendous benefits and opportunities for all dermatologists, including URM physicians. Other weaknesses and threats/barriers are outlined in the Figure.

Final Thoughts
We are far from reaching our goal of a diverse dermatology workforce, and the road ahead is long. We have a start and we have momentum. We can move forward by spreading the word that all types of diversity are a priority for our specialty. Making a true difference will require commitment and sustained efforts. Dermatology can lead the way as all of American medicine strives to attain workforce diversity.
- Saha S. Taking diversity seriously: the merits of increasing minority representation in medicine. JAMA Intern Med. 2014;174:291-292.
- Cooper LA, Roter DL, Johnson RL, et al. Patient-centered communication, ratings of care, and concordance of patient and physician race. Ann Intern Med. 2003;139:907-915.
- Saha S, Guiton G, Wimmers PF, et al. Student body racial and ethnic composition and diversity-related outcomes in US medical schools. JAMA. 2008;300:1135-1145.
- Marrast LM, Zallman L, Woolhandler S, et al. Minority physicians’ role in the care of underserved patients: diversifying the physician workforce may be key in addressing health disparities. JAMA Intern Med. 2014;174:289-291.
- Pandya AG, Alexis AF, Berger TG, et al. Increasing racial and ethnic diversity in dermatology: a call to action. J Am Acad Dermatol. 2016;74:584-587.
- Lester J, Wintroub B, Linos E. Disparities in academic dermatology. JAMA Dermatol. 2016;152:878-879.
- Chen A, Shinkai K. Rethinking how we select dermatology applicants—turning the tide. JAMA Dermatol. 2017;153:259-260.
- Granstein RD, Cornelius L, Shinkai K. Diversity in dermatology—a call for action. JAMA Dermatol. 2017;153:499-500.
- McKesey J, Berger TG, Lim HW, et al. Cultural competence for the 21st century dermatologist practicing in the United States. J Am Acad Dermatol. 2017;77:1159-1169.
- Van Voorhees AS, Enos CW. Diversity in dermatology residency programs. J Investig Dermatol Symp Proc. 2017;18:S46-S49.
- Imadojemu S, James WD. Increasing African American representation in dermatology. JAMA Dermatol. 2016;152:15-16.
- Saha S. Taking diversity seriously: the merits of increasing minority representation in medicine. JAMA Intern Med. 2014;174:291-292.
- Cooper LA, Roter DL, Johnson RL, et al. Patient-centered communication, ratings of care, and concordance of patient and physician race. Ann Intern Med. 2003;139:907-915.
- Saha S, Guiton G, Wimmers PF, et al. Student body racial and ethnic composition and diversity-related outcomes in US medical schools. JAMA. 2008;300:1135-1145.
- Marrast LM, Zallman L, Woolhandler S, et al. Minority physicians’ role in the care of underserved patients: diversifying the physician workforce may be key in addressing health disparities. JAMA Intern Med. 2014;174:289-291.
- Pandya AG, Alexis AF, Berger TG, et al. Increasing racial and ethnic diversity in dermatology: a call to action. J Am Acad Dermatol. 2016;74:584-587.
- Lester J, Wintroub B, Linos E. Disparities in academic dermatology. JAMA Dermatol. 2016;152:878-879.
- Chen A, Shinkai K. Rethinking how we select dermatology applicants—turning the tide. JAMA Dermatol. 2017;153:259-260.
- Granstein RD, Cornelius L, Shinkai K. Diversity in dermatology—a call for action. JAMA Dermatol. 2017;153:499-500.
- McKesey J, Berger TG, Lim HW, et al. Cultural competence for the 21st century dermatologist practicing in the United States. J Am Acad Dermatol. 2017;77:1159-1169.
- Van Voorhees AS, Enos CW. Diversity in dermatology residency programs. J Investig Dermatol Symp Proc. 2017;18:S46-S49.
- Imadojemu S, James WD. Increasing African American representation in dermatology. JAMA Dermatol. 2016;152:15-16.
The Effects of Sunscreen on Marine Environments
Coastal travel accounts for 80% of all tourism worldwide, a number that continues to grow. The number of travelers to the Mediterranean Sea alone is expected to rise to 350 million individuals per year within the next 20 years.1 As the number of tourists visiting the world’s oceans increases, the rate of sunscreen unintentionally washed into these marine environments also rises. One study estimated that approximately one-quarter of the sunscreen applied to the skin is washed off over a 20-minute period spent in the water.2 Four of the most common sunscreen agents—benzophenone-3 (BP-3),
Benzophenone-3
4-Methylbenzylidene Camphor
Environmental concerns have also been raised about another common chemical UV filter: 4-MBC, or enzacamene. In laboratory studies, 4-MBC has been shown to cause oxidative stress to Tetrahymena thermophila, an aquatic protozoan, which results in inhibited growth. At higher concentrations, damage to the cellular membrane was seen as soon as 4 hours after exposure.6 In embryonic zebrafish, elevated 4-MBC levels were correlated to improper nerve and muscular development, resulting in developmental defects.7 Another study demonstrated that 4-MBC was toxic to Mytilus galloprovincialis, known as the Mediterranean mussel, and Paracentrotus lividus, a species of sea urchin.8 Although these studies utilized highly controlled laboratory settings, further studies are needed to examine the effects of 4-MBC on these species at environmentally relevant concentrations.
Physical Sunscreens
Physical sunscreens, as compared to the chemical filters referenced above, use either zinc or titanium to protect the skin from the sun’s rays. Nanoparticles, in particular, are preferred because they do not leave a white film on the skin.9 Both titanium dioxide and zinc oxide nanoparticles have been found to inhibit the growth and photosynthesis of marine phytoplankton, the most abundant primary producers on Earth.10,11 These metal contaminants can be transferred to organisms of higher trophic levels, including zooplankton,12 and filter-feeding organisms, including marine abalone13 and the Mediterranean mussel.14 These nanoparticles have been shown to cause oxidative stress to these organisms, making them less fit to withstand environmental stressors. It is difficult to show their true impact, however, as it is challenging to accurately detect and quantify nanoparticle concentrations in vivo.15
Final Thoughts
- Marine problems: tourism & coastal development. World Wide Fund for Nature website. http://wwf.panda.org/about_our_earth/blue_planet/problems/tourism/. Published 2017. Accessed November 14, 2017.
- Danovaro R, Bongiorni L, Corinaldesi C, et al. Sunscreens cause coral bleaching by promoting viral infections. Environ Health Perspect. 2008;116:441-447.
- Downs C, Kramarsky-Winter E, Segal R, et al. Toxicopathological effects of the sunscreen UV filter, oxybenzone (benzophenone-3), on coral planulae and cultured primary cells and its environmental contamination in Hawaii and the US Virgin Islands. Arch Environ Contam Toxicol. 2016;70:265-288.
- Sánchez Rodríguez A, Rodrigo Sanz M, Betancort Rodríguez JR. Occurrence of eight UV filters in beaches of Gran Canaria (Canary Islands)[published online March 17, 2015]. Chemosphere. 2015;131:85-90.
- Bratkovics S, Sapozhnikova Y. Determination of seven commonly used organic UV filters in fresh and saline waters by liquid chromatography-tandem mass spectrometry. Analytical Methods. 2011;3:2943-2950.
- Gao L, Yuan T, Zhou C, et al. Effects of four commonly used UV filters on the growth, cell viability and oxidative stress responses of the Tetrahymena thermophila. Chemosphere. 2013;93:2507-2513.
- Li VW, Tsui MP, Chen X, et al. Effects of 4-methylbenzylidene camphor (4-MBC) on neuronal and muscular development in zebrafish (Danio rerio) embryos [published online February 18, 2016]. Environ Sci Pollut Res Int. 2016;23:8275-8285.
- Paredes E, Perez S, Rodil R, et al. Ecotoxicological evaluation of four UV filters using marine organisms from different trophic levels Isochrysis galbana, Mytilus galloprovincialis, Paracentrotus lividus, and Siriella armata. Chemosphere. 2014;104:44-50.
- Osterwalder U, Sohn M, Herzog B. Global state of sunscreens. Photodermatol Photoimmunol Photomed. 2014;30:62-80.
- Miller RJ, Bennett S, Keller AA, et al. TiO2 nanoparticles are phototoxic to marine phytoplankton. PloS One. 2012;7:E30321.
- Spisni E. Toxicity Assessment of Industrial- and Sunscreen-derived ZnO Nanoparticles [master’s thesis]. Coral Gables, FL: University of Miami Libraries Scholarly Repository; 2016. http://scholarlyrepository.miami.edu/cgi/viewcontent.cgi?article=1625&context=oa_theses. Accessed November 10, 2017.
- Jarvis TA, Miller RJ, Lenihan HS, et al. Toxicity of ZnO nanoparticles to the copepod Acartia tonsa, exposed through a phytoplankton diet [published online April 15, 2013]. Environ Toxicol Chem. 2013;32:1264-1269.
- Zhu X, Zhou J, Cai Z. The toxicity and oxidative stress of TiO2 nanoparticles in marine abalone (Haliotis diversicolor supertexta). Mar Pollut Bull. 2011;63:334-338.
- Barmo C, Ciacci C, Canonico B, et al. In vivo effects of n-TiO2 on digestive gland and immune function of the marine bivalve Mytilus galloprovincialis. Aquatic Toxicol. 2013;132:9-18.
- Sánchez-Quiles D, Tovar-Sánchez A. Are sunscreens a new environmental risk associated with coastal tourism? Environ Int. 2015;83:158-170.
- Xu S, Kwa M, Agarwal A, et al. Sunscreen product performance and other determinants of consumer preferences. JAMA Dermatol. 2016;152:920-927.
- Vesper I. Hawaii seeks to ban ‘reef-unfriendly’ sunscreen. Nature. February 3, 2017. https://www.nature.com/news/hawaii-seeks-to-ban-reef-unfriendly-sunscreen-1.21332. Accessed November 16, 2017.
Coastal travel accounts for 80% of all tourism worldwide, a number that continues to grow. The number of travelers to the Mediterranean Sea alone is expected to rise to 350 million individuals per year within the next 20 years.1 As the number of tourists visiting the world’s oceans increases, the rate of sunscreen unintentionally washed into these marine environments also rises. One study estimated that approximately one-quarter of the sunscreen applied to the skin is washed off over a 20-minute period spent in the water.2 Four of the most common sunscreen agents—benzophenone-3 (BP-3),
Benzophenone-3
4-Methylbenzylidene Camphor
Environmental concerns have also been raised about another common chemical UV filter: 4-MBC, or enzacamene. In laboratory studies, 4-MBC has been shown to cause oxidative stress to Tetrahymena thermophila, an aquatic protozoan, which results in inhibited growth. At higher concentrations, damage to the cellular membrane was seen as soon as 4 hours after exposure.6 In embryonic zebrafish, elevated 4-MBC levels were correlated to improper nerve and muscular development, resulting in developmental defects.7 Another study demonstrated that 4-MBC was toxic to Mytilus galloprovincialis, known as the Mediterranean mussel, and Paracentrotus lividus, a species of sea urchin.8 Although these studies utilized highly controlled laboratory settings, further studies are needed to examine the effects of 4-MBC on these species at environmentally relevant concentrations.
Physical Sunscreens
Physical sunscreens, as compared to the chemical filters referenced above, use either zinc or titanium to protect the skin from the sun’s rays. Nanoparticles, in particular, are preferred because they do not leave a white film on the skin.9 Both titanium dioxide and zinc oxide nanoparticles have been found to inhibit the growth and photosynthesis of marine phytoplankton, the most abundant primary producers on Earth.10,11 These metal contaminants can be transferred to organisms of higher trophic levels, including zooplankton,12 and filter-feeding organisms, including marine abalone13 and the Mediterranean mussel.14 These nanoparticles have been shown to cause oxidative stress to these organisms, making them less fit to withstand environmental stressors. It is difficult to show their true impact, however, as it is challenging to accurately detect and quantify nanoparticle concentrations in vivo.15
Final Thoughts
Coastal travel accounts for 80% of all tourism worldwide, a number that continues to grow. The number of travelers to the Mediterranean Sea alone is expected to rise to 350 million individuals per year within the next 20 years.1 As the number of tourists visiting the world’s oceans increases, the rate of sunscreen unintentionally washed into these marine environments also rises. One study estimated that approximately one-quarter of the sunscreen applied to the skin is washed off over a 20-minute period spent in the water.2 Four of the most common sunscreen agents—benzophenone-3 (BP-3),
Benzophenone-3
4-Methylbenzylidene Camphor
Environmental concerns have also been raised about another common chemical UV filter: 4-MBC, or enzacamene. In laboratory studies, 4-MBC has been shown to cause oxidative stress to Tetrahymena thermophila, an aquatic protozoan, which results in inhibited growth. At higher concentrations, damage to the cellular membrane was seen as soon as 4 hours after exposure.6 In embryonic zebrafish, elevated 4-MBC levels were correlated to improper nerve and muscular development, resulting in developmental defects.7 Another study demonstrated that 4-MBC was toxic to Mytilus galloprovincialis, known as the Mediterranean mussel, and Paracentrotus lividus, a species of sea urchin.8 Although these studies utilized highly controlled laboratory settings, further studies are needed to examine the effects of 4-MBC on these species at environmentally relevant concentrations.
Physical Sunscreens
Physical sunscreens, as compared to the chemical filters referenced above, use either zinc or titanium to protect the skin from the sun’s rays. Nanoparticles, in particular, are preferred because they do not leave a white film on the skin.9 Both titanium dioxide and zinc oxide nanoparticles have been found to inhibit the growth and photosynthesis of marine phytoplankton, the most abundant primary producers on Earth.10,11 These metal contaminants can be transferred to organisms of higher trophic levels, including zooplankton,12 and filter-feeding organisms, including marine abalone13 and the Mediterranean mussel.14 These nanoparticles have been shown to cause oxidative stress to these organisms, making them less fit to withstand environmental stressors. It is difficult to show their true impact, however, as it is challenging to accurately detect and quantify nanoparticle concentrations in vivo.15
Final Thoughts
- Marine problems: tourism & coastal development. World Wide Fund for Nature website. http://wwf.panda.org/about_our_earth/blue_planet/problems/tourism/. Published 2017. Accessed November 14, 2017.
- Danovaro R, Bongiorni L, Corinaldesi C, et al. Sunscreens cause coral bleaching by promoting viral infections. Environ Health Perspect. 2008;116:441-447.
- Downs C, Kramarsky-Winter E, Segal R, et al. Toxicopathological effects of the sunscreen UV filter, oxybenzone (benzophenone-3), on coral planulae and cultured primary cells and its environmental contamination in Hawaii and the US Virgin Islands. Arch Environ Contam Toxicol. 2016;70:265-288.
- Sánchez Rodríguez A, Rodrigo Sanz M, Betancort Rodríguez JR. Occurrence of eight UV filters in beaches of Gran Canaria (Canary Islands)[published online March 17, 2015]. Chemosphere. 2015;131:85-90.
- Bratkovics S, Sapozhnikova Y. Determination of seven commonly used organic UV filters in fresh and saline waters by liquid chromatography-tandem mass spectrometry. Analytical Methods. 2011;3:2943-2950.
- Gao L, Yuan T, Zhou C, et al. Effects of four commonly used UV filters on the growth, cell viability and oxidative stress responses of the Tetrahymena thermophila. Chemosphere. 2013;93:2507-2513.
- Li VW, Tsui MP, Chen X, et al. Effects of 4-methylbenzylidene camphor (4-MBC) on neuronal and muscular development in zebrafish (Danio rerio) embryos [published online February 18, 2016]. Environ Sci Pollut Res Int. 2016;23:8275-8285.
- Paredes E, Perez S, Rodil R, et al. Ecotoxicological evaluation of four UV filters using marine organisms from different trophic levels Isochrysis galbana, Mytilus galloprovincialis, Paracentrotus lividus, and Siriella armata. Chemosphere. 2014;104:44-50.
- Osterwalder U, Sohn M, Herzog B. Global state of sunscreens. Photodermatol Photoimmunol Photomed. 2014;30:62-80.
- Miller RJ, Bennett S, Keller AA, et al. TiO2 nanoparticles are phototoxic to marine phytoplankton. PloS One. 2012;7:E30321.
- Spisni E. Toxicity Assessment of Industrial- and Sunscreen-derived ZnO Nanoparticles [master’s thesis]. Coral Gables, FL: University of Miami Libraries Scholarly Repository; 2016. http://scholarlyrepository.miami.edu/cgi/viewcontent.cgi?article=1625&context=oa_theses. Accessed November 10, 2017.
- Jarvis TA, Miller RJ, Lenihan HS, et al. Toxicity of ZnO nanoparticles to the copepod Acartia tonsa, exposed through a phytoplankton diet [published online April 15, 2013]. Environ Toxicol Chem. 2013;32:1264-1269.
- Zhu X, Zhou J, Cai Z. The toxicity and oxidative stress of TiO2 nanoparticles in marine abalone (Haliotis diversicolor supertexta). Mar Pollut Bull. 2011;63:334-338.
- Barmo C, Ciacci C, Canonico B, et al. In vivo effects of n-TiO2 on digestive gland and immune function of the marine bivalve Mytilus galloprovincialis. Aquatic Toxicol. 2013;132:9-18.
- Sánchez-Quiles D, Tovar-Sánchez A. Are sunscreens a new environmental risk associated with coastal tourism? Environ Int. 2015;83:158-170.
- Xu S, Kwa M, Agarwal A, et al. Sunscreen product performance and other determinants of consumer preferences. JAMA Dermatol. 2016;152:920-927.
- Vesper I. Hawaii seeks to ban ‘reef-unfriendly’ sunscreen. Nature. February 3, 2017. https://www.nature.com/news/hawaii-seeks-to-ban-reef-unfriendly-sunscreen-1.21332. Accessed November 16, 2017.
- Marine problems: tourism & coastal development. World Wide Fund for Nature website. http://wwf.panda.org/about_our_earth/blue_planet/problems/tourism/. Published 2017. Accessed November 14, 2017.
- Danovaro R, Bongiorni L, Corinaldesi C, et al. Sunscreens cause coral bleaching by promoting viral infections. Environ Health Perspect. 2008;116:441-447.
- Downs C, Kramarsky-Winter E, Segal R, et al. Toxicopathological effects of the sunscreen UV filter, oxybenzone (benzophenone-3), on coral planulae and cultured primary cells and its environmental contamination in Hawaii and the US Virgin Islands. Arch Environ Contam Toxicol. 2016;70:265-288.
- Sánchez Rodríguez A, Rodrigo Sanz M, Betancort Rodríguez JR. Occurrence of eight UV filters in beaches of Gran Canaria (Canary Islands)[published online March 17, 2015]. Chemosphere. 2015;131:85-90.
- Bratkovics S, Sapozhnikova Y. Determination of seven commonly used organic UV filters in fresh and saline waters by liquid chromatography-tandem mass spectrometry. Analytical Methods. 2011;3:2943-2950.
- Gao L, Yuan T, Zhou C, et al. Effects of four commonly used UV filters on the growth, cell viability and oxidative stress responses of the Tetrahymena thermophila. Chemosphere. 2013;93:2507-2513.
- Li VW, Tsui MP, Chen X, et al. Effects of 4-methylbenzylidene camphor (4-MBC) on neuronal and muscular development in zebrafish (Danio rerio) embryos [published online February 18, 2016]. Environ Sci Pollut Res Int. 2016;23:8275-8285.
- Paredes E, Perez S, Rodil R, et al. Ecotoxicological evaluation of four UV filters using marine organisms from different trophic levels Isochrysis galbana, Mytilus galloprovincialis, Paracentrotus lividus, and Siriella armata. Chemosphere. 2014;104:44-50.
- Osterwalder U, Sohn M, Herzog B. Global state of sunscreens. Photodermatol Photoimmunol Photomed. 2014;30:62-80.
- Miller RJ, Bennett S, Keller AA, et al. TiO2 nanoparticles are phototoxic to marine phytoplankton. PloS One. 2012;7:E30321.
- Spisni E. Toxicity Assessment of Industrial- and Sunscreen-derived ZnO Nanoparticles [master’s thesis]. Coral Gables, FL: University of Miami Libraries Scholarly Repository; 2016. http://scholarlyrepository.miami.edu/cgi/viewcontent.cgi?article=1625&context=oa_theses. Accessed November 10, 2017.
- Jarvis TA, Miller RJ, Lenihan HS, et al. Toxicity of ZnO nanoparticles to the copepod Acartia tonsa, exposed through a phytoplankton diet [published online April 15, 2013]. Environ Toxicol Chem. 2013;32:1264-1269.
- Zhu X, Zhou J, Cai Z. The toxicity and oxidative stress of TiO2 nanoparticles in marine abalone (Haliotis diversicolor supertexta). Mar Pollut Bull. 2011;63:334-338.
- Barmo C, Ciacci C, Canonico B, et al. In vivo effects of n-TiO2 on digestive gland and immune function of the marine bivalve Mytilus galloprovincialis. Aquatic Toxicol. 2013;132:9-18.
- Sánchez-Quiles D, Tovar-Sánchez A. Are sunscreens a new environmental risk associated with coastal tourism? Environ Int. 2015;83:158-170.
- Xu S, Kwa M, Agarwal A, et al. Sunscreen product performance and other determinants of consumer preferences. JAMA Dermatol. 2016;152:920-927.
- Vesper I. Hawaii seeks to ban ‘reef-unfriendly’ sunscreen. Nature. February 3, 2017. https://www.nature.com/news/hawaii-seeks-to-ban-reef-unfriendly-sunscreen-1.21332. Accessed November 16, 2017.
Mississippi has highest varicella vaccination rate
Coverage for two doses of varicella vaccine among kindergarten students was highest in Mississippi and lowest in the District of Columbia, said Ranee Seither and associates at the National Center of Immunization and Respiratory Disease at the Centers for Disease Control and Prevention, Atlanta.
For the 2016-2017 school year, 99.4% of Mississippi children enrolled in kindergarten received the state-required two doses of varicella vaccine, compared with 84.6% in D.C. The median was 93.8% for the 42 states that require two doses and 96.5% for those 42 plus the 7 states that reported and only require one dose. Oklahoma and Wyoming “did not report data because of widespread problems with the quality of data reported by schools,” the CDC investigators wrote (MMWR 2017;66[40]:1073-80).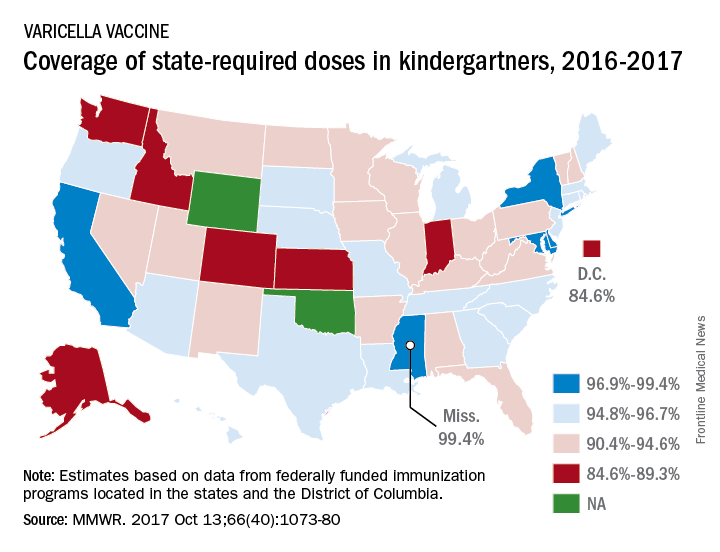
The data for the CDC analysis, which included 3,973,172 kindergartners for the 2016-2017 school year, were collected by federally funded immunization programs in the 50 states and D.C.
Coverage for two doses of varicella vaccine among kindergarten students was highest in Mississippi and lowest in the District of Columbia, said Ranee Seither and associates at the National Center of Immunization and Respiratory Disease at the Centers for Disease Control and Prevention, Atlanta.
For the 2016-2017 school year, 99.4% of Mississippi children enrolled in kindergarten received the state-required two doses of varicella vaccine, compared with 84.6% in D.C. The median was 93.8% for the 42 states that require two doses and 96.5% for those 42 plus the 7 states that reported and only require one dose. Oklahoma and Wyoming “did not report data because of widespread problems with the quality of data reported by schools,” the CDC investigators wrote (MMWR 2017;66[40]:1073-80).
The data for the CDC analysis, which included 3,973,172 kindergartners for the 2016-2017 school year, were collected by federally funded immunization programs in the 50 states and D.C.
Coverage for two doses of varicella vaccine among kindergarten students was highest in Mississippi and lowest in the District of Columbia, said Ranee Seither and associates at the National Center of Immunization and Respiratory Disease at the Centers for Disease Control and Prevention, Atlanta.
For the 2016-2017 school year, 99.4% of Mississippi children enrolled in kindergarten received the state-required two doses of varicella vaccine, compared with 84.6% in D.C. The median was 93.8% for the 42 states that require two doses and 96.5% for those 42 plus the 7 states that reported and only require one dose. Oklahoma and Wyoming “did not report data because of widespread problems with the quality of data reported by schools,” the CDC investigators wrote (MMWR 2017;66[40]:1073-80).
The data for the CDC analysis, which included 3,973,172 kindergartners for the 2016-2017 school year, were collected by federally funded immunization programs in the 50 states and D.C.
FROM MMWR
ENCORE 601 study: Entinostat shows promise in NSCLC
NATIONAL HARBOR, MD. – The oral, class I selective histone deacetylase (HDAC) inhibitor entinostat given in combination with pembrolizumab demonstrated antitumor activity and acceptable safety in patients with non–small cell lung cancer in the phase 1b/2 ENCORE 601 study.
Entinostat, which has been shown in preclinical models to enhance suppressor cells in the tumor microenvironment, was evaluated in ENCORE 601 as a treatment for non–small cell lung cancer (NSCLC), melanoma, and colorectal cancer. Previously reported phase 1 results showed that an oral dose of 5 mg weekly plus 200 mg of pembrolizumab given intravenously every 3 weeks deserved further exploration for these indications, according to Leena Gandhi, MD, who reported phase 2, stage 1 results from the lung cancer arm of the Simon two-stage study at the annual meeting of the Society for Immunotherapy of Cancer.
Treatment at that dose was studied in both anti-PD-L1–naive patients with advanced NSCLC, and in NSCLC patients who progressed on anti-PD-L1 treatment, said Dr. Gandhi of New York University Langone Medical Center.
The primary objective of stage 1 was objective response rate, and criteria for advancement were 4 or more responses out of 17 evaluable anti-PD-L1–naive patients (cohort 1), and at least 3 responses out of 31 patients who progressed on anti-PD-L1 therapy (cohort 2).
Both cohorts met the endpoint, with 4 of 17 evaluable cohort 1 patients (24%) achieving a partial response, and 3 of 31 evaluable cohort 2 patients (10%) achieving a partial response.
In cohort 1, two responses were confirmed and two were unconfirmed. One of the unconfirmed patients had malignant pericardial effusion, but remains on study with continued clinical benefit, Dr. Gandhi said, noting that three patients remain on study in all.
“The other notable thing I’d like to point out here … is that the majority of these were patients who did not have high levels of expression of PD-L1,” she said.
In cohort 2 patients, two responses were confirmed and one was unconfirmed. Three patients remain on study.
“In both of these cohorts there are a couple of patients who’ve had quite durable responses,” she said.
The best response to prior anti-PD-1therapy in the cohort 2 patients who had a response was stable disease (two patients). The response to prior therapy was unknown in one patient, she noted.
“All of them had clear regressions, after that initial PD-1 therapy, with this combination,” she said, noting that two had “essentially negative PD-L1 expression, and none had high levels of expression.”
Treatment was associated with grade 3/4 adverse events deemed drug related in 31% of patients; the most common of these events, occurring in at least 10% of patients in cohort 1, were hypophosphatemia and neutropenia, and in cohort 2 were fatigue, anemia, anorexia, and pneumonitis; 13% of patients discontinued treatment due to an adverse event, Dr. Gandhi said.
Of note, there were reductions in circulating myeloid derived suppressor cells in both cohorts following treatment.
Based on the responses seen in this first stage of the study, cohort 2 has advanced to stage 2 and has completed enrollment. Additional patients have not been enrolled in cohort 1, but that is still under consideration, she said.
Dr. Gandhi reported having no disclosures.
NATIONAL HARBOR, MD. – The oral, class I selective histone deacetylase (HDAC) inhibitor entinostat given in combination with pembrolizumab demonstrated antitumor activity and acceptable safety in patients with non–small cell lung cancer in the phase 1b/2 ENCORE 601 study.
Entinostat, which has been shown in preclinical models to enhance suppressor cells in the tumor microenvironment, was evaluated in ENCORE 601 as a treatment for non–small cell lung cancer (NSCLC), melanoma, and colorectal cancer. Previously reported phase 1 results showed that an oral dose of 5 mg weekly plus 200 mg of pembrolizumab given intravenously every 3 weeks deserved further exploration for these indications, according to Leena Gandhi, MD, who reported phase 2, stage 1 results from the lung cancer arm of the Simon two-stage study at the annual meeting of the Society for Immunotherapy of Cancer.
Treatment at that dose was studied in both anti-PD-L1–naive patients with advanced NSCLC, and in NSCLC patients who progressed on anti-PD-L1 treatment, said Dr. Gandhi of New York University Langone Medical Center.
The primary objective of stage 1 was objective response rate, and criteria for advancement were 4 or more responses out of 17 evaluable anti-PD-L1–naive patients (cohort 1), and at least 3 responses out of 31 patients who progressed on anti-PD-L1 therapy (cohort 2).
Both cohorts met the endpoint, with 4 of 17 evaluable cohort 1 patients (24%) achieving a partial response, and 3 of 31 evaluable cohort 2 patients (10%) achieving a partial response.
In cohort 1, two responses were confirmed and two were unconfirmed. One of the unconfirmed patients had malignant pericardial effusion, but remains on study with continued clinical benefit, Dr. Gandhi said, noting that three patients remain on study in all.
“The other notable thing I’d like to point out here … is that the majority of these were patients who did not have high levels of expression of PD-L1,” she said.
In cohort 2 patients, two responses were confirmed and one was unconfirmed. Three patients remain on study.
“In both of these cohorts there are a couple of patients who’ve had quite durable responses,” she said.
The best response to prior anti-PD-1therapy in the cohort 2 patients who had a response was stable disease (two patients). The response to prior therapy was unknown in one patient, she noted.
“All of them had clear regressions, after that initial PD-1 therapy, with this combination,” she said, noting that two had “essentially negative PD-L1 expression, and none had high levels of expression.”
Treatment was associated with grade 3/4 adverse events deemed drug related in 31% of patients; the most common of these events, occurring in at least 10% of patients in cohort 1, were hypophosphatemia and neutropenia, and in cohort 2 were fatigue, anemia, anorexia, and pneumonitis; 13% of patients discontinued treatment due to an adverse event, Dr. Gandhi said.
Of note, there were reductions in circulating myeloid derived suppressor cells in both cohorts following treatment.
Based on the responses seen in this first stage of the study, cohort 2 has advanced to stage 2 and has completed enrollment. Additional patients have not been enrolled in cohort 1, but that is still under consideration, she said.
Dr. Gandhi reported having no disclosures.
NATIONAL HARBOR, MD. – The oral, class I selective histone deacetylase (HDAC) inhibitor entinostat given in combination with pembrolizumab demonstrated antitumor activity and acceptable safety in patients with non–small cell lung cancer in the phase 1b/2 ENCORE 601 study.
Entinostat, which has been shown in preclinical models to enhance suppressor cells in the tumor microenvironment, was evaluated in ENCORE 601 as a treatment for non–small cell lung cancer (NSCLC), melanoma, and colorectal cancer. Previously reported phase 1 results showed that an oral dose of 5 mg weekly plus 200 mg of pembrolizumab given intravenously every 3 weeks deserved further exploration for these indications, according to Leena Gandhi, MD, who reported phase 2, stage 1 results from the lung cancer arm of the Simon two-stage study at the annual meeting of the Society for Immunotherapy of Cancer.
Treatment at that dose was studied in both anti-PD-L1–naive patients with advanced NSCLC, and in NSCLC patients who progressed on anti-PD-L1 treatment, said Dr. Gandhi of New York University Langone Medical Center.
The primary objective of stage 1 was objective response rate, and criteria for advancement were 4 or more responses out of 17 evaluable anti-PD-L1–naive patients (cohort 1), and at least 3 responses out of 31 patients who progressed on anti-PD-L1 therapy (cohort 2).
Both cohorts met the endpoint, with 4 of 17 evaluable cohort 1 patients (24%) achieving a partial response, and 3 of 31 evaluable cohort 2 patients (10%) achieving a partial response.
In cohort 1, two responses were confirmed and two were unconfirmed. One of the unconfirmed patients had malignant pericardial effusion, but remains on study with continued clinical benefit, Dr. Gandhi said, noting that three patients remain on study in all.
“The other notable thing I’d like to point out here … is that the majority of these were patients who did not have high levels of expression of PD-L1,” she said.
In cohort 2 patients, two responses were confirmed and one was unconfirmed. Three patients remain on study.
“In both of these cohorts there are a couple of patients who’ve had quite durable responses,” she said.
The best response to prior anti-PD-1therapy in the cohort 2 patients who had a response was stable disease (two patients). The response to prior therapy was unknown in one patient, she noted.
“All of them had clear regressions, after that initial PD-1 therapy, with this combination,” she said, noting that two had “essentially negative PD-L1 expression, and none had high levels of expression.”
Treatment was associated with grade 3/4 adverse events deemed drug related in 31% of patients; the most common of these events, occurring in at least 10% of patients in cohort 1, were hypophosphatemia and neutropenia, and in cohort 2 were fatigue, anemia, anorexia, and pneumonitis; 13% of patients discontinued treatment due to an adverse event, Dr. Gandhi said.
Of note, there were reductions in circulating myeloid derived suppressor cells in both cohorts following treatment.
Based on the responses seen in this first stage of the study, cohort 2 has advanced to stage 2 and has completed enrollment. Additional patients have not been enrolled in cohort 1, but that is still under consideration, she said.
Dr. Gandhi reported having no disclosures.
AT SITC 2017
Key clinical point:
Major finding: Partial responses were seen in 24% of cohort 1 patients and 10% of cohort 2 patients.
Data source: Stage 1 of a phase 2 Simon two-stage study (48 evaluable patients).
Disclosures: Dr. Gandhi reported having no disclosures.
Pediatric acute appendicitis: Is it time for nonoperative treatment (NOT)?
Clinical question
What are the differences in rates of treatment failure, duration of hospitalization, and cost between nonoperative treatment (NOT) for acute uncomplicated appendicitis versus urgent appendectomy?
Background
Acute appendicitis is found in around 5% of children presenting for urgent or emergent evaluation of abdominal pain. It is the most common illness prompting emergency abdominal surgery in children.
Several trials in adults have shown that urgent surgery may not be necessary, and NOT of uncomplicated appendicitis may be both effective and safe. NOT involves a course of IV antibiotics and careful clinical monitoring while hospitalized, then a course of oral antibiotics after discharge. Regimens vary but include coverage for aerobic and anaerobic gut flora, such as piperacillin-tazobactam followed by amoxicillin. Little is known about the safety and efficacy of NOT in children.
Study design
Meta-analysis.
Search strategy
PubMed, MEDLINE, EMBASE, and Cochrane Library were searched for relevant studies. This search identified 527 potential articles, of which the authors examined the full text of 68 and ultimately identified 5 single-center trials for analysis (4 prospective cohort trials and 1 randomized, controlled trial).
Synopsis
A total of 404 patients with uncomplicated appendicitis were seen in all trials: 168 received NOT and 236 received standard surgical care (urgent appendectomy). In the single randomized, controlled trial, patients were assigned NOT or surgical care randomly. In the other trials parental preference directed therapy.
The heterogeneity of the design, populations, definitions of illness, duration of follow-up, and NOT treatment regimens made the meta-analysis challenging. Antibiotic options for NOT varied by center but included a course of IV antibiotics followed by 7-10 days of oral antibiotics. NOT success was defined as no need for surgery within 48 hours and no recurrence of appendicitis within 1 month. Of the 236 patients who received standard surgical care, all had appendicitis and 1 had a complication requiring repeat operation. Of the NOT group, 16 (9.5%) had treatment failures, including 3 with perforated appendicitis, and 45 (27%) went on to have an appendectomy within the following year, yielding a risk ratio of failure versus standard treatment of 8.9 (95% confidence interval, 2.7-29.8). A subgroup analysis of patients with appendicoliths who received NOT found that these patients experienced a substantially increased risk of treatment failures and recurrent appendicitis with the risk ratio versus NOT without appendicolith of 10.4 (95% CI, 1.5-74). Of the 30 patients who experienced treatment failure with NOT, 15 had appendicoliths. NOT lengthened hospital stays by 14.3 hours (95% CI, 7.5-21.1) but led to lower total costs by $1,310 (95% CI, $920-$1,690).
Bottom line
NOT may be a reasonable alternative to standard surgical management for acute uncomplicated appendicitis without appendicolith in children, with a success rate of greater than 90%. Further larger, randomized prospective studies are required to establish its safety and efficacy.
Citation
Huang L et al. Comparison of antibiotic therapy and appendectomy for acute uncomplicated appendicitis in children: A meta-analysis. JAMA Pediatr. 2017;171(5):426-34.
Dr. Stubblefield is a pediatric hospitalist at Nemours/Alfred I. duPont Hospital for Children in Wilmington, Del., and a clinical assistant professor of pediatrics at Jefferson Medical College in Philadelphia.
Clinical question
What are the differences in rates of treatment failure, duration of hospitalization, and cost between nonoperative treatment (NOT) for acute uncomplicated appendicitis versus urgent appendectomy?
Background
Acute appendicitis is found in around 5% of children presenting for urgent or emergent evaluation of abdominal pain. It is the most common illness prompting emergency abdominal surgery in children.
Several trials in adults have shown that urgent surgery may not be necessary, and NOT of uncomplicated appendicitis may be both effective and safe. NOT involves a course of IV antibiotics and careful clinical monitoring while hospitalized, then a course of oral antibiotics after discharge. Regimens vary but include coverage for aerobic and anaerobic gut flora, such as piperacillin-tazobactam followed by amoxicillin. Little is known about the safety and efficacy of NOT in children.
Study design
Meta-analysis.
Search strategy
PubMed, MEDLINE, EMBASE, and Cochrane Library were searched for relevant studies. This search identified 527 potential articles, of which the authors examined the full text of 68 and ultimately identified 5 single-center trials for analysis (4 prospective cohort trials and 1 randomized, controlled trial).
Synopsis
A total of 404 patients with uncomplicated appendicitis were seen in all trials: 168 received NOT and 236 received standard surgical care (urgent appendectomy). In the single randomized, controlled trial, patients were assigned NOT or surgical care randomly. In the other trials parental preference directed therapy.
The heterogeneity of the design, populations, definitions of illness, duration of follow-up, and NOT treatment regimens made the meta-analysis challenging. Antibiotic options for NOT varied by center but included a course of IV antibiotics followed by 7-10 days of oral antibiotics. NOT success was defined as no need for surgery within 48 hours and no recurrence of appendicitis within 1 month. Of the 236 patients who received standard surgical care, all had appendicitis and 1 had a complication requiring repeat operation. Of the NOT group, 16 (9.5%) had treatment failures, including 3 with perforated appendicitis, and 45 (27%) went on to have an appendectomy within the following year, yielding a risk ratio of failure versus standard treatment of 8.9 (95% confidence interval, 2.7-29.8). A subgroup analysis of patients with appendicoliths who received NOT found that these patients experienced a substantially increased risk of treatment failures and recurrent appendicitis with the risk ratio versus NOT without appendicolith of 10.4 (95% CI, 1.5-74). Of the 30 patients who experienced treatment failure with NOT, 15 had appendicoliths. NOT lengthened hospital stays by 14.3 hours (95% CI, 7.5-21.1) but led to lower total costs by $1,310 (95% CI, $920-$1,690).
Bottom line
NOT may be a reasonable alternative to standard surgical management for acute uncomplicated appendicitis without appendicolith in children, with a success rate of greater than 90%. Further larger, randomized prospective studies are required to establish its safety and efficacy.
Citation
Huang L et al. Comparison of antibiotic therapy and appendectomy for acute uncomplicated appendicitis in children: A meta-analysis. JAMA Pediatr. 2017;171(5):426-34.
Dr. Stubblefield is a pediatric hospitalist at Nemours/Alfred I. duPont Hospital for Children in Wilmington, Del., and a clinical assistant professor of pediatrics at Jefferson Medical College in Philadelphia.
Clinical question
What are the differences in rates of treatment failure, duration of hospitalization, and cost between nonoperative treatment (NOT) for acute uncomplicated appendicitis versus urgent appendectomy?
Background
Acute appendicitis is found in around 5% of children presenting for urgent or emergent evaluation of abdominal pain. It is the most common illness prompting emergency abdominal surgery in children.
Several trials in adults have shown that urgent surgery may not be necessary, and NOT of uncomplicated appendicitis may be both effective and safe. NOT involves a course of IV antibiotics and careful clinical monitoring while hospitalized, then a course of oral antibiotics after discharge. Regimens vary but include coverage for aerobic and anaerobic gut flora, such as piperacillin-tazobactam followed by amoxicillin. Little is known about the safety and efficacy of NOT in children.
Study design
Meta-analysis.
Search strategy
PubMed, MEDLINE, EMBASE, and Cochrane Library were searched for relevant studies. This search identified 527 potential articles, of which the authors examined the full text of 68 and ultimately identified 5 single-center trials for analysis (4 prospective cohort trials and 1 randomized, controlled trial).
Synopsis
A total of 404 patients with uncomplicated appendicitis were seen in all trials: 168 received NOT and 236 received standard surgical care (urgent appendectomy). In the single randomized, controlled trial, patients were assigned NOT or surgical care randomly. In the other trials parental preference directed therapy.
The heterogeneity of the design, populations, definitions of illness, duration of follow-up, and NOT treatment regimens made the meta-analysis challenging. Antibiotic options for NOT varied by center but included a course of IV antibiotics followed by 7-10 days of oral antibiotics. NOT success was defined as no need for surgery within 48 hours and no recurrence of appendicitis within 1 month. Of the 236 patients who received standard surgical care, all had appendicitis and 1 had a complication requiring repeat operation. Of the NOT group, 16 (9.5%) had treatment failures, including 3 with perforated appendicitis, and 45 (27%) went on to have an appendectomy within the following year, yielding a risk ratio of failure versus standard treatment of 8.9 (95% confidence interval, 2.7-29.8). A subgroup analysis of patients with appendicoliths who received NOT found that these patients experienced a substantially increased risk of treatment failures and recurrent appendicitis with the risk ratio versus NOT without appendicolith of 10.4 (95% CI, 1.5-74). Of the 30 patients who experienced treatment failure with NOT, 15 had appendicoliths. NOT lengthened hospital stays by 14.3 hours (95% CI, 7.5-21.1) but led to lower total costs by $1,310 (95% CI, $920-$1,690).
Bottom line
NOT may be a reasonable alternative to standard surgical management for acute uncomplicated appendicitis without appendicolith in children, with a success rate of greater than 90%. Further larger, randomized prospective studies are required to establish its safety and efficacy.
Citation
Huang L et al. Comparison of antibiotic therapy and appendectomy for acute uncomplicated appendicitis in children: A meta-analysis. JAMA Pediatr. 2017;171(5):426-34.
Dr. Stubblefield is a pediatric hospitalist at Nemours/Alfred I. duPont Hospital for Children in Wilmington, Del., and a clinical assistant professor of pediatrics at Jefferson Medical College in Philadelphia.
Emerging treatments tackling hair loss challenges include light therapies
according to Maria Hordinsky, MD.
“Although the precise mechanism remains unclear, it has been postulated that photobiomodulation acts through oxidative metabolism and transcription factor stimulation,” Dr. Hordinsky said in a presentation at Skin Disease Education Foundation’s Women’s & Pediatric Dermatology Seminar.
She referred to one trial, which found that men with androgenetic alopecia who used the HairMax Lasercomb showed an increase in mean hair density at 26 weeks of daily use, compared with a group that used a sham device.
Photobiomodulation devices use either laser light or light-emitting diodes. Comparing the two types is a challenge, and the question of which is more effective remains unanswered, Dr. Hordinsky said.
Other issues to be addressed in future research include finding the optimal wavelength to use for different indications for light-based treatments, determining whether pulse or continuous wave is more effective, and evaluating the potential for systemic side effects of these therapies, she noted.
No treatment for alopecia areata is currently approved by the Food and Drug Administration, but factors to consider when choosing a treatment include the patient’s age, location and extent of hair loss, and the presence of other medical problems, as well as a scalp biopsy report with information on the hair cycle and inflammation. Patients and/or their parents should understand the risks and benefits associated with various treatments to make an informed decision, Dr. Hordinsky said.
Patients and their families “have heard the ‘buzz’ about potential new treatments for alopecia areata, and the discussion needs to include a conversation about ongoing and future clinical research opportunities, as well as off-label use of Janus kinase inhibitors,” particularly oral tofacitinib, she said.
Approximately two-thirds of patients in recent studies of oral tofacitinib have had clinically acceptable hair regrowth after 6 months, Dr. Hordinsky said. Ruxolitinib is also being studied. However, “until clinical research studies are completed, there will be ongoing debate regarding the risks and benefits, cost, and sustainability” of JAK inhibitors or other new treatments, she said.
Dr. Hordinsky disclosed that she is a consultant for companies including Procter & Gamble and Concert, and has received grant/research support from Incyte, Allergan, and the National Alopecia Areata Foundation.
SDEF and this news organization are owned by Frontline Medical Communications.
according to Maria Hordinsky, MD.
“Although the precise mechanism remains unclear, it has been postulated that photobiomodulation acts through oxidative metabolism and transcription factor stimulation,” Dr. Hordinsky said in a presentation at Skin Disease Education Foundation’s Women’s & Pediatric Dermatology Seminar.
She referred to one trial, which found that men with androgenetic alopecia who used the HairMax Lasercomb showed an increase in mean hair density at 26 weeks of daily use, compared with a group that used a sham device.
Photobiomodulation devices use either laser light or light-emitting diodes. Comparing the two types is a challenge, and the question of which is more effective remains unanswered, Dr. Hordinsky said.
Other issues to be addressed in future research include finding the optimal wavelength to use for different indications for light-based treatments, determining whether pulse or continuous wave is more effective, and evaluating the potential for systemic side effects of these therapies, she noted.
No treatment for alopecia areata is currently approved by the Food and Drug Administration, but factors to consider when choosing a treatment include the patient’s age, location and extent of hair loss, and the presence of other medical problems, as well as a scalp biopsy report with information on the hair cycle and inflammation. Patients and/or their parents should understand the risks and benefits associated with various treatments to make an informed decision, Dr. Hordinsky said.
Patients and their families “have heard the ‘buzz’ about potential new treatments for alopecia areata, and the discussion needs to include a conversation about ongoing and future clinical research opportunities, as well as off-label use of Janus kinase inhibitors,” particularly oral tofacitinib, she said.
Approximately two-thirds of patients in recent studies of oral tofacitinib have had clinically acceptable hair regrowth after 6 months, Dr. Hordinsky said. Ruxolitinib is also being studied. However, “until clinical research studies are completed, there will be ongoing debate regarding the risks and benefits, cost, and sustainability” of JAK inhibitors or other new treatments, she said.
Dr. Hordinsky disclosed that she is a consultant for companies including Procter & Gamble and Concert, and has received grant/research support from Incyte, Allergan, and the National Alopecia Areata Foundation.
SDEF and this news organization are owned by Frontline Medical Communications.
according to Maria Hordinsky, MD.
“Although the precise mechanism remains unclear, it has been postulated that photobiomodulation acts through oxidative metabolism and transcription factor stimulation,” Dr. Hordinsky said in a presentation at Skin Disease Education Foundation’s Women’s & Pediatric Dermatology Seminar.
She referred to one trial, which found that men with androgenetic alopecia who used the HairMax Lasercomb showed an increase in mean hair density at 26 weeks of daily use, compared with a group that used a sham device.
Photobiomodulation devices use either laser light or light-emitting diodes. Comparing the two types is a challenge, and the question of which is more effective remains unanswered, Dr. Hordinsky said.
Other issues to be addressed in future research include finding the optimal wavelength to use for different indications for light-based treatments, determining whether pulse or continuous wave is more effective, and evaluating the potential for systemic side effects of these therapies, she noted.
No treatment for alopecia areata is currently approved by the Food and Drug Administration, but factors to consider when choosing a treatment include the patient’s age, location and extent of hair loss, and the presence of other medical problems, as well as a scalp biopsy report with information on the hair cycle and inflammation. Patients and/or their parents should understand the risks and benefits associated with various treatments to make an informed decision, Dr. Hordinsky said.
Patients and their families “have heard the ‘buzz’ about potential new treatments for alopecia areata, and the discussion needs to include a conversation about ongoing and future clinical research opportunities, as well as off-label use of Janus kinase inhibitors,” particularly oral tofacitinib, she said.
Approximately two-thirds of patients in recent studies of oral tofacitinib have had clinically acceptable hair regrowth after 6 months, Dr. Hordinsky said. Ruxolitinib is also being studied. However, “until clinical research studies are completed, there will be ongoing debate regarding the risks and benefits, cost, and sustainability” of JAK inhibitors or other new treatments, she said.
Dr. Hordinsky disclosed that she is a consultant for companies including Procter & Gamble and Concert, and has received grant/research support from Incyte, Allergan, and the National Alopecia Areata Foundation.
SDEF and this news organization are owned by Frontline Medical Communications.
FROM SDEF WOMEN’S & PEDIATRIC DERMATOLOGY SEMINAR
FDA: Puerto Rico’s medical supply shortage issues continue
“In addition to our ongoing concerns related to IV saline products, we also are particularly focused on the shortage of amino acids for injection,” FDA Commissioner Scott Gottlieb, MD, said in a statement.
The shortage of amino acids for injection predates Hurricane Maria, but the hurricane’s impact worsened the situation by disrupting Baxter’s ability to manufacture amino acids in Puerto Rico. The FDA has worked with Baxter to temporarily import pediatric and adult amino acid injections from the United Kingdom and Italy, as well as working with other amino acid manufacturers to increase supplies.
“We continue to work closely with federal and Puerto Rican authorities to address the needs of manufacturers on the island for power and other resources. These efforts have been focused on the needs of patients – to prevent potential shortages of medically important products where possible, and help ensure that any shortages that do occur are mitigated as quickly as possible,” Dr. Gottlieb said.
Read the full statement on the FDA website.
“In addition to our ongoing concerns related to IV saline products, we also are particularly focused on the shortage of amino acids for injection,” FDA Commissioner Scott Gottlieb, MD, said in a statement.
The shortage of amino acids for injection predates Hurricane Maria, but the hurricane’s impact worsened the situation by disrupting Baxter’s ability to manufacture amino acids in Puerto Rico. The FDA has worked with Baxter to temporarily import pediatric and adult amino acid injections from the United Kingdom and Italy, as well as working with other amino acid manufacturers to increase supplies.
“We continue to work closely with federal and Puerto Rican authorities to address the needs of manufacturers on the island for power and other resources. These efforts have been focused on the needs of patients – to prevent potential shortages of medically important products where possible, and help ensure that any shortages that do occur are mitigated as quickly as possible,” Dr. Gottlieb said.
Read the full statement on the FDA website.
“In addition to our ongoing concerns related to IV saline products, we also are particularly focused on the shortage of amino acids for injection,” FDA Commissioner Scott Gottlieb, MD, said in a statement.
The shortage of amino acids for injection predates Hurricane Maria, but the hurricane’s impact worsened the situation by disrupting Baxter’s ability to manufacture amino acids in Puerto Rico. The FDA has worked with Baxter to temporarily import pediatric and adult amino acid injections from the United Kingdom and Italy, as well as working with other amino acid manufacturers to increase supplies.
“We continue to work closely with federal and Puerto Rican authorities to address the needs of manufacturers on the island for power and other resources. These efforts have been focused on the needs of patients – to prevent potential shortages of medically important products where possible, and help ensure that any shortages that do occur are mitigated as quickly as possible,” Dr. Gottlieb said.
Read the full statement on the FDA website.










