User login
Bringing you the latest news, research and reviews, exclusive interviews, podcasts, quizzes, and more.
div[contains(@class, 'read-next-article')]
div[contains(@class, 'nav-primary')]
nav[contains(@class, 'nav-primary')]
section[contains(@class, 'footer-nav-section-wrapper')]
nav[contains(@class, 'nav-ce-stack nav-ce-stack__large-screen')]
header[@id='header']
div[contains(@class, 'header__large-screen')]
div[contains(@class, 'read-next-article')]
div[contains(@class, 'main-prefix')]
div[contains(@class, 'nav-primary')]
nav[contains(@class, 'nav-primary')]
section[contains(@class, 'footer-nav-section-wrapper')]
footer[@id='footer']
section[contains(@class, 'nav-hidden')]
div[contains(@class, 'ce-card-content')]
nav[contains(@class, 'nav-ce-stack')]
div[contains(@class, 'view-medstat-quiz-listing-panes')]
div[contains(@class, 'pane-article-sidebar-latest-news')]
Water birth may have benefits for healthy women: Meta-analysis suggests
Water immersion during labor and birth significantly reduced use of medications, maternal pain, and postpartum hemorrhage, compared with standard care with no water immersion, based on data from 36 studies including more than 150,000 women.
“Resting and laboring in water can reduce fear, anxiety, and pain perception; it helps optimize the physiology of childbirth through the release of endogenous endorphins and oxytocin,” and data from randomized, controlled trials have shown a reduced need for epidural analgesia with water immersion, Ethel Burns, PhD, of Oxford (England) Brookes University Faculty of Health and Life Sciences, and colleagues wrote.
Although previous studies have not shown an increased risk for adverse events for newborns following water birth, “There is a need to understand which clinical practices, when performed as part of water immersion care, result in the optimum outcomes for mother and newborn,” the researchers said.
In a systematic review and meta-analysis published in BMJ Open, the researchers identified studies published since 2000 that examined maternal or neonatal interventions and/or outcomes when birthing pools were used for labor and/or birth.
The primary objective was to compare intrapartum interventions and outcomes for water immersion during labor with standard care with no water immersion.
Water immersion generally involves the use of a birth pool for relaxation and pain relief in early labor, and some women proceed with immersion through the second stage of labor and delivery. Of the 36 included studies, 31 took place in a hospital setting, 4 in a midwife-led setting, and 1 in a mixed setting. Most of the studies (25) involved women who planned to have/had a water birth, and these studies included 151,742 women. Another seven studies including 1,901 women involved in water immersion for labor only, three studies including 3,688 women involved in water immersion during labor and water birth; the timing of water immersion was unclear in the remaining study of 215 women.
Overall, water immersion significantly reduced the use of epidurals (odds ratio, 0.17), injected opioids (OR, 0.22), and episiotomy (OR, 0.16). Maternal pain and postpartum hemorrhage also were significantly reduced with water immersion (OR, 0.24 and OR, 0.69, respectively).
Maternal satisfaction was significantly increased with water immersion, and the odds of an intact perineum increased as well (OR, 1.95 and OR, 1.48).
The overall odds of cord avulsion increased with water immersion (OR, 1.94), but the absolute risk was low, compared with births without water immersion (4.3 vs. 1.3 per 1,000). No significant differences in other identified neonatal outcomes were observed across the studies.
The study findings were limited by several factors including the inconsistency of reporting on birth setting, care practices, interventions, and outcomes, and the inclusion of only three outcomes for meta-regression analysis, the researchers noted. In addition, only four studies were conducted in midwifery-led settings.
“This is important because birth pool use is most prevalent in midwifery-led settings,” the researchers wrote.” Evidence-based practice of water immersion requires research that reflects the context of care provision.
“We suggest that studies incorporate the following fundamentals to advance the evidence: birth pool description, clearly described maternal and obstetric characteristics, the birth setting, the care model and use of standardized definitions.”
Despite the limitations and need for additional research, the data overall support the potential benefits from water immersion births for healthy women and newborns, the researchers concluded.
A Clinical Report issued by the American Academy of Pediatrics in January 2022 advised against water immersion during the second stage of labor and delivery. According to the report, the potential for neonatal infections from organisms such as Legionella and Pseudomonas species, is low, but does exist, and could result in serious complications.
Education is essential
Increasing numbers of women are seeking home births and water births, Marissa Platner, MD, a maternal-fetal medicine specialist at Emory University, Atlanta, said in an interview.
“Given the conflicting data and lack of data, it is important to be able to educate birthing mothers based on best available evidence,” said Dr. Platner, who was not involved in the study.
“I was not surprised by the findings, because the adverse outcomes that are of concern, such as neonatal sepsis, were not clearly addressed,” Dr. Platner said. Given that sepsis “is a rare outcome in the population of low-risk individuals, the study may not have been powered to assess for this. The findings of maternal pain and satisfaction being improved with water immersion are well known. ACOG [American College of Obstetricians and Gynecologists] has also stated that water immersion during the first stage of labor is safe and can help with pain control.”
On a practical level, “I think clinicians can use this guidance to discuss the potential benefits of water immersion in the first stages of labor, but would caution women regarding the unknown but possible risks of the water birth, given these findings are less clear,” Dr. Platner said.
“I think the findings regarding maternal outcomes are valid and consistent with the AAP/ACOG recommendations in terms of improving maternal pain control; however, more research is needed to determine the safety of the second stage of labor occurring in the water, given the potential for neonatal infection and respiratory distress, which could not be adequately addressed in this study,” Dr. Platner emphasized.
The study was supported by Oxford Brookes University. The researchers had no financial conflicts to disclose. Dr. Platner had no financial conflicts to disclose.
Water immersion during labor and birth significantly reduced use of medications, maternal pain, and postpartum hemorrhage, compared with standard care with no water immersion, based on data from 36 studies including more than 150,000 women.
“Resting and laboring in water can reduce fear, anxiety, and pain perception; it helps optimize the physiology of childbirth through the release of endogenous endorphins and oxytocin,” and data from randomized, controlled trials have shown a reduced need for epidural analgesia with water immersion, Ethel Burns, PhD, of Oxford (England) Brookes University Faculty of Health and Life Sciences, and colleagues wrote.
Although previous studies have not shown an increased risk for adverse events for newborns following water birth, “There is a need to understand which clinical practices, when performed as part of water immersion care, result in the optimum outcomes for mother and newborn,” the researchers said.
In a systematic review and meta-analysis published in BMJ Open, the researchers identified studies published since 2000 that examined maternal or neonatal interventions and/or outcomes when birthing pools were used for labor and/or birth.
The primary objective was to compare intrapartum interventions and outcomes for water immersion during labor with standard care with no water immersion.
Water immersion generally involves the use of a birth pool for relaxation and pain relief in early labor, and some women proceed with immersion through the second stage of labor and delivery. Of the 36 included studies, 31 took place in a hospital setting, 4 in a midwife-led setting, and 1 in a mixed setting. Most of the studies (25) involved women who planned to have/had a water birth, and these studies included 151,742 women. Another seven studies including 1,901 women involved in water immersion for labor only, three studies including 3,688 women involved in water immersion during labor and water birth; the timing of water immersion was unclear in the remaining study of 215 women.
Overall, water immersion significantly reduced the use of epidurals (odds ratio, 0.17), injected opioids (OR, 0.22), and episiotomy (OR, 0.16). Maternal pain and postpartum hemorrhage also were significantly reduced with water immersion (OR, 0.24 and OR, 0.69, respectively).
Maternal satisfaction was significantly increased with water immersion, and the odds of an intact perineum increased as well (OR, 1.95 and OR, 1.48).
The overall odds of cord avulsion increased with water immersion (OR, 1.94), but the absolute risk was low, compared with births without water immersion (4.3 vs. 1.3 per 1,000). No significant differences in other identified neonatal outcomes were observed across the studies.
The study findings were limited by several factors including the inconsistency of reporting on birth setting, care practices, interventions, and outcomes, and the inclusion of only three outcomes for meta-regression analysis, the researchers noted. In addition, only four studies were conducted in midwifery-led settings.
“This is important because birth pool use is most prevalent in midwifery-led settings,” the researchers wrote.” Evidence-based practice of water immersion requires research that reflects the context of care provision.
“We suggest that studies incorporate the following fundamentals to advance the evidence: birth pool description, clearly described maternal and obstetric characteristics, the birth setting, the care model and use of standardized definitions.”
Despite the limitations and need for additional research, the data overall support the potential benefits from water immersion births for healthy women and newborns, the researchers concluded.
A Clinical Report issued by the American Academy of Pediatrics in January 2022 advised against water immersion during the second stage of labor and delivery. According to the report, the potential for neonatal infections from organisms such as Legionella and Pseudomonas species, is low, but does exist, and could result in serious complications.
Education is essential
Increasing numbers of women are seeking home births and water births, Marissa Platner, MD, a maternal-fetal medicine specialist at Emory University, Atlanta, said in an interview.
“Given the conflicting data and lack of data, it is important to be able to educate birthing mothers based on best available evidence,” said Dr. Platner, who was not involved in the study.
“I was not surprised by the findings, because the adverse outcomes that are of concern, such as neonatal sepsis, were not clearly addressed,” Dr. Platner said. Given that sepsis “is a rare outcome in the population of low-risk individuals, the study may not have been powered to assess for this. The findings of maternal pain and satisfaction being improved with water immersion are well known. ACOG [American College of Obstetricians and Gynecologists] has also stated that water immersion during the first stage of labor is safe and can help with pain control.”
On a practical level, “I think clinicians can use this guidance to discuss the potential benefits of water immersion in the first stages of labor, but would caution women regarding the unknown but possible risks of the water birth, given these findings are less clear,” Dr. Platner said.
“I think the findings regarding maternal outcomes are valid and consistent with the AAP/ACOG recommendations in terms of improving maternal pain control; however, more research is needed to determine the safety of the second stage of labor occurring in the water, given the potential for neonatal infection and respiratory distress, which could not be adequately addressed in this study,” Dr. Platner emphasized.
The study was supported by Oxford Brookes University. The researchers had no financial conflicts to disclose. Dr. Platner had no financial conflicts to disclose.
Water immersion during labor and birth significantly reduced use of medications, maternal pain, and postpartum hemorrhage, compared with standard care with no water immersion, based on data from 36 studies including more than 150,000 women.
“Resting and laboring in water can reduce fear, anxiety, and pain perception; it helps optimize the physiology of childbirth through the release of endogenous endorphins and oxytocin,” and data from randomized, controlled trials have shown a reduced need for epidural analgesia with water immersion, Ethel Burns, PhD, of Oxford (England) Brookes University Faculty of Health and Life Sciences, and colleagues wrote.
Although previous studies have not shown an increased risk for adverse events for newborns following water birth, “There is a need to understand which clinical practices, when performed as part of water immersion care, result in the optimum outcomes for mother and newborn,” the researchers said.
In a systematic review and meta-analysis published in BMJ Open, the researchers identified studies published since 2000 that examined maternal or neonatal interventions and/or outcomes when birthing pools were used for labor and/or birth.
The primary objective was to compare intrapartum interventions and outcomes for water immersion during labor with standard care with no water immersion.
Water immersion generally involves the use of a birth pool for relaxation and pain relief in early labor, and some women proceed with immersion through the second stage of labor and delivery. Of the 36 included studies, 31 took place in a hospital setting, 4 in a midwife-led setting, and 1 in a mixed setting. Most of the studies (25) involved women who planned to have/had a water birth, and these studies included 151,742 women. Another seven studies including 1,901 women involved in water immersion for labor only, three studies including 3,688 women involved in water immersion during labor and water birth; the timing of water immersion was unclear in the remaining study of 215 women.
Overall, water immersion significantly reduced the use of epidurals (odds ratio, 0.17), injected opioids (OR, 0.22), and episiotomy (OR, 0.16). Maternal pain and postpartum hemorrhage also were significantly reduced with water immersion (OR, 0.24 and OR, 0.69, respectively).
Maternal satisfaction was significantly increased with water immersion, and the odds of an intact perineum increased as well (OR, 1.95 and OR, 1.48).
The overall odds of cord avulsion increased with water immersion (OR, 1.94), but the absolute risk was low, compared with births without water immersion (4.3 vs. 1.3 per 1,000). No significant differences in other identified neonatal outcomes were observed across the studies.
The study findings were limited by several factors including the inconsistency of reporting on birth setting, care practices, interventions, and outcomes, and the inclusion of only three outcomes for meta-regression analysis, the researchers noted. In addition, only four studies were conducted in midwifery-led settings.
“This is important because birth pool use is most prevalent in midwifery-led settings,” the researchers wrote.” Evidence-based practice of water immersion requires research that reflects the context of care provision.
“We suggest that studies incorporate the following fundamentals to advance the evidence: birth pool description, clearly described maternal and obstetric characteristics, the birth setting, the care model and use of standardized definitions.”
Despite the limitations and need for additional research, the data overall support the potential benefits from water immersion births for healthy women and newborns, the researchers concluded.
A Clinical Report issued by the American Academy of Pediatrics in January 2022 advised against water immersion during the second stage of labor and delivery. According to the report, the potential for neonatal infections from organisms such as Legionella and Pseudomonas species, is low, but does exist, and could result in serious complications.
Education is essential
Increasing numbers of women are seeking home births and water births, Marissa Platner, MD, a maternal-fetal medicine specialist at Emory University, Atlanta, said in an interview.
“Given the conflicting data and lack of data, it is important to be able to educate birthing mothers based on best available evidence,” said Dr. Platner, who was not involved in the study.
“I was not surprised by the findings, because the adverse outcomes that are of concern, such as neonatal sepsis, were not clearly addressed,” Dr. Platner said. Given that sepsis “is a rare outcome in the population of low-risk individuals, the study may not have been powered to assess for this. The findings of maternal pain and satisfaction being improved with water immersion are well known. ACOG [American College of Obstetricians and Gynecologists] has also stated that water immersion during the first stage of labor is safe and can help with pain control.”
On a practical level, “I think clinicians can use this guidance to discuss the potential benefits of water immersion in the first stages of labor, but would caution women regarding the unknown but possible risks of the water birth, given these findings are less clear,” Dr. Platner said.
“I think the findings regarding maternal outcomes are valid and consistent with the AAP/ACOG recommendations in terms of improving maternal pain control; however, more research is needed to determine the safety of the second stage of labor occurring in the water, given the potential for neonatal infection and respiratory distress, which could not be adequately addressed in this study,” Dr. Platner emphasized.
The study was supported by Oxford Brookes University. The researchers had no financial conflicts to disclose. Dr. Platner had no financial conflicts to disclose.
FROM BMJ OPEN
Study confirms BRCA1 and BRCA2 linked to seven cancers
from prior analyses showing associations with breast, ovarian, prostate, and pancreatic cancers. The finding, published in JAMA Oncology suggests a possible broader clinical relevance for BRCA1 and BRCA2 genetic testing.
Pathogenic variants in BRCA1 were found to be associated with biliary tract cancer, in BRCA2 with esophageal cancer, and in BRCA1/2 with gastric cancer.
“The results suggest the range of cancer types associated with pathogenic variants in BRCA1 and BRCA2 is likely broader than that determined from previous analysis of largely European ancestry cohorts,” wrote authors who were led by Yukihide Momozawa, DVM, PhD, RIKEN Center for Integrative Medical Sciences, Japan.
“These risk association findings, together with our analysis of an association with family history of cancer and clinical phenotypes, are relevant for developing and adapting guidelines about genetic testing, treatment options, and treatability with PARP [poly adenosine diphosphate-ribose polymerase] inhibitors for each cancer type,” the authors wrote.
Dr. Momozawa and associates conducted a large-scale sequencing study across 14 common cancer types in 63,828 patients (mean age 64 years, 42% female) and 37,086 controls on data drawn from a Japanese nationwide biobank between April 2003 and March 2018. They estimated the risk of each cancer type and determined clinical characteristics associated with pathogenic variant carrier status, while also investigating the utility of family history in detecting patients with pathogenic variants.
Three hundred fifteen pathogenic variants were identified. An odds ratios of greater than 4.0 (with P < 1 × 10−4 as the threshold of significance) for the pathogenic variants were found for biliary tract cancer (OR, 17.4; 95% confidence interval, 5.8-51.9) in BRCA1, esophageal cancer (OR, 5.6; 95% CI, 2.9-11.0) in BRCA2, and gastric cancer (OR, 5.2; 95% CI, 2.6-10.5) in BRCA1, and (OR, 4.7; 95% CI, 3.1-7.1) in BRCA2. Two other cancer types were found to be associated with BRCA1, and four other cancer types with BRCA2. Enrichment of carrier patients was shown in biliary tract, female breast, ovarian, and prostate cancers in accordance with increased numbers of reported cancer types in relatives.
Male patients with breast cancer had a very high carrier frequency of pathogenic variants in BRCA2 (18.9%), but not BRCA1 (1.89%). Patients with ovarian cancer showed the next highest proportion (BRCA1, 4.86%; BRCA2, 3.42%). Frequency exceeding 1% was seen for several other cancer types (two cancer types for BRCA1, four cancer types for BRCA2). More than one cancer types was identified in 4,128 patients (6.3%). Carrier frequency of pathogenic variants in BRCA1 was 0.44% with one cancer type, 0.85% with two cancer types, and 0.69% with three cancer types. It was 0.97%, 1.40%, and 1.74%, respectively, in BRCA2.
“The results of this large-scale registry-based case-control study suggest that pathogenic variants in BRCA1 and BRCA2 were associated with the risk of seven cancer types. These results indicate broader clinical relevance of BRCA1 and BRCA2 genetic testing,” the authors wrote.
PARP inhibitors were developed based on the mechanism in BRCA1 and BRCA2 of homologous recombination repair defects associated with pathogenic variants. PARP inhibitors have been found to have therapeutic efficacy also in pathogenic variants found to be enriched in prostate and pancreatic cancers. While risk for additional cancer types (for example, biliary tract cancer, cervical cancer, colorectal cancer, endometrial cancer, esophageal cancer, and stomach cancer) has been reported after analyzing family members for the presence of pathogenic variants and performing case-control analyses, evidence for an association with these cancer types has not been considered sufficient for them to be adopted into clinical management guidelines, the authors wrote.
In an interview, Dr. Momozawa said that BRCA1 and BRCA2 genetic testing should be expanded in Japan. “But further studies are needed to reveal how much. If a clinical trial of a PARP inhibitor for these three cancer types reveals its clinical utility, the importance of this expansion will increase.”
Dr. Momozawa and associates state that while their selection of controls without a family history of cancer affects the generalizability of the study results, the estimated cumulative risks were comparable with those based on prospective cohorts, suggesting the study design did not greatly affect the results.
from prior analyses showing associations with breast, ovarian, prostate, and pancreatic cancers. The finding, published in JAMA Oncology suggests a possible broader clinical relevance for BRCA1 and BRCA2 genetic testing.
Pathogenic variants in BRCA1 were found to be associated with biliary tract cancer, in BRCA2 with esophageal cancer, and in BRCA1/2 with gastric cancer.
“The results suggest the range of cancer types associated with pathogenic variants in BRCA1 and BRCA2 is likely broader than that determined from previous analysis of largely European ancestry cohorts,” wrote authors who were led by Yukihide Momozawa, DVM, PhD, RIKEN Center for Integrative Medical Sciences, Japan.
“These risk association findings, together with our analysis of an association with family history of cancer and clinical phenotypes, are relevant for developing and adapting guidelines about genetic testing, treatment options, and treatability with PARP [poly adenosine diphosphate-ribose polymerase] inhibitors for each cancer type,” the authors wrote.
Dr. Momozawa and associates conducted a large-scale sequencing study across 14 common cancer types in 63,828 patients (mean age 64 years, 42% female) and 37,086 controls on data drawn from a Japanese nationwide biobank between April 2003 and March 2018. They estimated the risk of each cancer type and determined clinical characteristics associated with pathogenic variant carrier status, while also investigating the utility of family history in detecting patients with pathogenic variants.
Three hundred fifteen pathogenic variants were identified. An odds ratios of greater than 4.0 (with P < 1 × 10−4 as the threshold of significance) for the pathogenic variants were found for biliary tract cancer (OR, 17.4; 95% confidence interval, 5.8-51.9) in BRCA1, esophageal cancer (OR, 5.6; 95% CI, 2.9-11.0) in BRCA2, and gastric cancer (OR, 5.2; 95% CI, 2.6-10.5) in BRCA1, and (OR, 4.7; 95% CI, 3.1-7.1) in BRCA2. Two other cancer types were found to be associated with BRCA1, and four other cancer types with BRCA2. Enrichment of carrier patients was shown in biliary tract, female breast, ovarian, and prostate cancers in accordance with increased numbers of reported cancer types in relatives.
Male patients with breast cancer had a very high carrier frequency of pathogenic variants in BRCA2 (18.9%), but not BRCA1 (1.89%). Patients with ovarian cancer showed the next highest proportion (BRCA1, 4.86%; BRCA2, 3.42%). Frequency exceeding 1% was seen for several other cancer types (two cancer types for BRCA1, four cancer types for BRCA2). More than one cancer types was identified in 4,128 patients (6.3%). Carrier frequency of pathogenic variants in BRCA1 was 0.44% with one cancer type, 0.85% with two cancer types, and 0.69% with three cancer types. It was 0.97%, 1.40%, and 1.74%, respectively, in BRCA2.
“The results of this large-scale registry-based case-control study suggest that pathogenic variants in BRCA1 and BRCA2 were associated with the risk of seven cancer types. These results indicate broader clinical relevance of BRCA1 and BRCA2 genetic testing,” the authors wrote.
PARP inhibitors were developed based on the mechanism in BRCA1 and BRCA2 of homologous recombination repair defects associated with pathogenic variants. PARP inhibitors have been found to have therapeutic efficacy also in pathogenic variants found to be enriched in prostate and pancreatic cancers. While risk for additional cancer types (for example, biliary tract cancer, cervical cancer, colorectal cancer, endometrial cancer, esophageal cancer, and stomach cancer) has been reported after analyzing family members for the presence of pathogenic variants and performing case-control analyses, evidence for an association with these cancer types has not been considered sufficient for them to be adopted into clinical management guidelines, the authors wrote.
In an interview, Dr. Momozawa said that BRCA1 and BRCA2 genetic testing should be expanded in Japan. “But further studies are needed to reveal how much. If a clinical trial of a PARP inhibitor for these three cancer types reveals its clinical utility, the importance of this expansion will increase.”
Dr. Momozawa and associates state that while their selection of controls without a family history of cancer affects the generalizability of the study results, the estimated cumulative risks were comparable with those based on prospective cohorts, suggesting the study design did not greatly affect the results.
from prior analyses showing associations with breast, ovarian, prostate, and pancreatic cancers. The finding, published in JAMA Oncology suggests a possible broader clinical relevance for BRCA1 and BRCA2 genetic testing.
Pathogenic variants in BRCA1 were found to be associated with biliary tract cancer, in BRCA2 with esophageal cancer, and in BRCA1/2 with gastric cancer.
“The results suggest the range of cancer types associated with pathogenic variants in BRCA1 and BRCA2 is likely broader than that determined from previous analysis of largely European ancestry cohorts,” wrote authors who were led by Yukihide Momozawa, DVM, PhD, RIKEN Center for Integrative Medical Sciences, Japan.
“These risk association findings, together with our analysis of an association with family history of cancer and clinical phenotypes, are relevant for developing and adapting guidelines about genetic testing, treatment options, and treatability with PARP [poly adenosine diphosphate-ribose polymerase] inhibitors for each cancer type,” the authors wrote.
Dr. Momozawa and associates conducted a large-scale sequencing study across 14 common cancer types in 63,828 patients (mean age 64 years, 42% female) and 37,086 controls on data drawn from a Japanese nationwide biobank between April 2003 and March 2018. They estimated the risk of each cancer type and determined clinical characteristics associated with pathogenic variant carrier status, while also investigating the utility of family history in detecting patients with pathogenic variants.
Three hundred fifteen pathogenic variants were identified. An odds ratios of greater than 4.0 (with P < 1 × 10−4 as the threshold of significance) for the pathogenic variants were found for biliary tract cancer (OR, 17.4; 95% confidence interval, 5.8-51.9) in BRCA1, esophageal cancer (OR, 5.6; 95% CI, 2.9-11.0) in BRCA2, and gastric cancer (OR, 5.2; 95% CI, 2.6-10.5) in BRCA1, and (OR, 4.7; 95% CI, 3.1-7.1) in BRCA2. Two other cancer types were found to be associated with BRCA1, and four other cancer types with BRCA2. Enrichment of carrier patients was shown in biliary tract, female breast, ovarian, and prostate cancers in accordance with increased numbers of reported cancer types in relatives.
Male patients with breast cancer had a very high carrier frequency of pathogenic variants in BRCA2 (18.9%), but not BRCA1 (1.89%). Patients with ovarian cancer showed the next highest proportion (BRCA1, 4.86%; BRCA2, 3.42%). Frequency exceeding 1% was seen for several other cancer types (two cancer types for BRCA1, four cancer types for BRCA2). More than one cancer types was identified in 4,128 patients (6.3%). Carrier frequency of pathogenic variants in BRCA1 was 0.44% with one cancer type, 0.85% with two cancer types, and 0.69% with three cancer types. It was 0.97%, 1.40%, and 1.74%, respectively, in BRCA2.
“The results of this large-scale registry-based case-control study suggest that pathogenic variants in BRCA1 and BRCA2 were associated with the risk of seven cancer types. These results indicate broader clinical relevance of BRCA1 and BRCA2 genetic testing,” the authors wrote.
PARP inhibitors were developed based on the mechanism in BRCA1 and BRCA2 of homologous recombination repair defects associated with pathogenic variants. PARP inhibitors have been found to have therapeutic efficacy also in pathogenic variants found to be enriched in prostate and pancreatic cancers. While risk for additional cancer types (for example, biliary tract cancer, cervical cancer, colorectal cancer, endometrial cancer, esophageal cancer, and stomach cancer) has been reported after analyzing family members for the presence of pathogenic variants and performing case-control analyses, evidence for an association with these cancer types has not been considered sufficient for them to be adopted into clinical management guidelines, the authors wrote.
In an interview, Dr. Momozawa said that BRCA1 and BRCA2 genetic testing should be expanded in Japan. “But further studies are needed to reveal how much. If a clinical trial of a PARP inhibitor for these three cancer types reveals its clinical utility, the importance of this expansion will increase.”
Dr. Momozawa and associates state that while their selection of controls without a family history of cancer affects the generalizability of the study results, the estimated cumulative risks were comparable with those based on prospective cohorts, suggesting the study design did not greatly affect the results.
FROM JAMA ONCOLOGY
Sociogenomics may explain race disparities in breast cancer mortality
Racial differences in cancer outcomes are widespread. Studies indicate that Black people face higher rates of mortality for most cancers than their White counterparts. To bridge this racial gap, researchers need to investigate the biological effects of structural racism and discrimination on cancer outcomes, experts say.
“As a physician, I always like to think that I can influence care in that if I just find the right drugs, help patients understand what their options are, it will help them,” said Ruth Carlos, MD, a radiologist with the University of Michigan Hospital, Ann Arbor. But these things alone are often not enough, because a large proportion of the variation in cancer outcomes is attributable to neighborhood social conditions and the physical environment. “It is incredibly important for us to start to understand just how the neighborhood exerts this effect.”
In a commentary published in the Journal of Clinical Oncology, Dr. Carlos and colleagues highlighted the limitations of previous studies aimed at identifying the causes of racial differences in cancer outcomes. They call upon researchers to turn instead to the long-underexamined biological effects of structural racism and discrimination that contribute to these differences.
In the past, studies on the role of race in health outcomes largely looked at race as a proxy for genetic predisposition. But such an interpretation is flawed, because no genes are specific for a racial or ethnic group, Dr. Carlos and coauthors wrote. Researchers have shown that the vast majority of genetic variation occurs within, rather than between groups.
In an analysis published in Science, researchers reported that within-group differences account for more than 90% of genetic variation.
“Using race in these analyses was not necessarily wrong, but the inferences may have been flawed or incomplete,” Dr. Carlos said. On one hand, looking at genetic predisposition has led to important insights, such as the link between mutations in the BRCA gene and increased risk for breast and ovarian cancer.
However, genetic variation alone is not enough to explain the disparities in cancer outcomes between racial and ethnic groups. The fact that breast cancer can be more aggressive in Black women raises several questions, Dr. Carlos said. Is the cancer worse because Black women have a specific genetic predisposition? Is it worse because Black women exist in a society that marginalizes them and exposes them to increased stress, which in turn produces bad outcomes? Or, could it be both?
Despite progress in the screening, diagnosis and treatment of breast cancer, Black women are 40% more likely to die from the disease than White women. At the time of diagnosis, Black women are more likely to have high-grade, more aggressive breast cancer molecular subtypes, and to have had their cancer spread to the lymph nodes. They also tend to be diagnosed at more advanced stages of breast cancer while at the same time, experience higher rates of false-positive screening results.
Although researchers have hypothesized that genetic differences related to African or European ancestry might contribute, studies have not turned up any differences in cancer susceptibility genes by race. Other factors, such as racial differences in the stage of presentation, molecular subtypes, and disparities in treatment, have also emerged as potential culprits.
In her commentary, Dr. Carlos and colleagues wrote that disparities in breast cancer outcomes previously attributed to race need to be examined from multiple angles. This means looking at both the complex interactions between social conditions and policies, which encompass racism both at the individual and structural level, and stressors such as the experience of discrimination in addition to potential biological and genetic contributions.
Many studies now provide evidence for the harmful effects of racism on health. For breast cancer, specifically, studies also suggest that factors such as racial segregation can influence the stage at which Black women get diagnosed and their likelihood of dying from the disease.
However, an important question that remains is what biological changes occur in women exposed to the kind of persistent low-level stress that is associated with structural racism and discrimination, Dr. Carlos said. “We don’t know what stress pathways actually manifest in the body and how they eventually produce the disease.” Studies to address this issue are important, “especially if you would like to develop interventions to prevent or mitigate disease.”
To address this issue, Dr. Carlos and colleagues called upon the research community to conduct both studies that delineate the underlying biology as well as those that test potential interventions – particularly those associated with breast cancer screening outcomes – to try to shed light on why Black women receive more false positives and diagnoses of more aggressive cancer.
Interventions that can target these specific biological pathways could potentially reduce the negative effects of structural racism and discrimination as well as the effects of other social factors that contribute to breast cancer outcomes, “to ultimately help enhance clinical outcomes and close persistent disparities gaps,” the authors wrote.
Racial differences in cancer outcomes are widespread. Studies indicate that Black people face higher rates of mortality for most cancers than their White counterparts. To bridge this racial gap, researchers need to investigate the biological effects of structural racism and discrimination on cancer outcomes, experts say.
“As a physician, I always like to think that I can influence care in that if I just find the right drugs, help patients understand what their options are, it will help them,” said Ruth Carlos, MD, a radiologist with the University of Michigan Hospital, Ann Arbor. But these things alone are often not enough, because a large proportion of the variation in cancer outcomes is attributable to neighborhood social conditions and the physical environment. “It is incredibly important for us to start to understand just how the neighborhood exerts this effect.”
In a commentary published in the Journal of Clinical Oncology, Dr. Carlos and colleagues highlighted the limitations of previous studies aimed at identifying the causes of racial differences in cancer outcomes. They call upon researchers to turn instead to the long-underexamined biological effects of structural racism and discrimination that contribute to these differences.
In the past, studies on the role of race in health outcomes largely looked at race as a proxy for genetic predisposition. But such an interpretation is flawed, because no genes are specific for a racial or ethnic group, Dr. Carlos and coauthors wrote. Researchers have shown that the vast majority of genetic variation occurs within, rather than between groups.
In an analysis published in Science, researchers reported that within-group differences account for more than 90% of genetic variation.
“Using race in these analyses was not necessarily wrong, but the inferences may have been flawed or incomplete,” Dr. Carlos said. On one hand, looking at genetic predisposition has led to important insights, such as the link between mutations in the BRCA gene and increased risk for breast and ovarian cancer.
However, genetic variation alone is not enough to explain the disparities in cancer outcomes between racial and ethnic groups. The fact that breast cancer can be more aggressive in Black women raises several questions, Dr. Carlos said. Is the cancer worse because Black women have a specific genetic predisposition? Is it worse because Black women exist in a society that marginalizes them and exposes them to increased stress, which in turn produces bad outcomes? Or, could it be both?
Despite progress in the screening, diagnosis and treatment of breast cancer, Black women are 40% more likely to die from the disease than White women. At the time of diagnosis, Black women are more likely to have high-grade, more aggressive breast cancer molecular subtypes, and to have had their cancer spread to the lymph nodes. They also tend to be diagnosed at more advanced stages of breast cancer while at the same time, experience higher rates of false-positive screening results.
Although researchers have hypothesized that genetic differences related to African or European ancestry might contribute, studies have not turned up any differences in cancer susceptibility genes by race. Other factors, such as racial differences in the stage of presentation, molecular subtypes, and disparities in treatment, have also emerged as potential culprits.
In her commentary, Dr. Carlos and colleagues wrote that disparities in breast cancer outcomes previously attributed to race need to be examined from multiple angles. This means looking at both the complex interactions between social conditions and policies, which encompass racism both at the individual and structural level, and stressors such as the experience of discrimination in addition to potential biological and genetic contributions.
Many studies now provide evidence for the harmful effects of racism on health. For breast cancer, specifically, studies also suggest that factors such as racial segregation can influence the stage at which Black women get diagnosed and their likelihood of dying from the disease.
However, an important question that remains is what biological changes occur in women exposed to the kind of persistent low-level stress that is associated with structural racism and discrimination, Dr. Carlos said. “We don’t know what stress pathways actually manifest in the body and how they eventually produce the disease.” Studies to address this issue are important, “especially if you would like to develop interventions to prevent or mitigate disease.”
To address this issue, Dr. Carlos and colleagues called upon the research community to conduct both studies that delineate the underlying biology as well as those that test potential interventions – particularly those associated with breast cancer screening outcomes – to try to shed light on why Black women receive more false positives and diagnoses of more aggressive cancer.
Interventions that can target these specific biological pathways could potentially reduce the negative effects of structural racism and discrimination as well as the effects of other social factors that contribute to breast cancer outcomes, “to ultimately help enhance clinical outcomes and close persistent disparities gaps,” the authors wrote.
Racial differences in cancer outcomes are widespread. Studies indicate that Black people face higher rates of mortality for most cancers than their White counterparts. To bridge this racial gap, researchers need to investigate the biological effects of structural racism and discrimination on cancer outcomes, experts say.
“As a physician, I always like to think that I can influence care in that if I just find the right drugs, help patients understand what their options are, it will help them,” said Ruth Carlos, MD, a radiologist with the University of Michigan Hospital, Ann Arbor. But these things alone are often not enough, because a large proportion of the variation in cancer outcomes is attributable to neighborhood social conditions and the physical environment. “It is incredibly important for us to start to understand just how the neighborhood exerts this effect.”
In a commentary published in the Journal of Clinical Oncology, Dr. Carlos and colleagues highlighted the limitations of previous studies aimed at identifying the causes of racial differences in cancer outcomes. They call upon researchers to turn instead to the long-underexamined biological effects of structural racism and discrimination that contribute to these differences.
In the past, studies on the role of race in health outcomes largely looked at race as a proxy for genetic predisposition. But such an interpretation is flawed, because no genes are specific for a racial or ethnic group, Dr. Carlos and coauthors wrote. Researchers have shown that the vast majority of genetic variation occurs within, rather than between groups.
In an analysis published in Science, researchers reported that within-group differences account for more than 90% of genetic variation.
“Using race in these analyses was not necessarily wrong, but the inferences may have been flawed or incomplete,” Dr. Carlos said. On one hand, looking at genetic predisposition has led to important insights, such as the link between mutations in the BRCA gene and increased risk for breast and ovarian cancer.
However, genetic variation alone is not enough to explain the disparities in cancer outcomes between racial and ethnic groups. The fact that breast cancer can be more aggressive in Black women raises several questions, Dr. Carlos said. Is the cancer worse because Black women have a specific genetic predisposition? Is it worse because Black women exist in a society that marginalizes them and exposes them to increased stress, which in turn produces bad outcomes? Or, could it be both?
Despite progress in the screening, diagnosis and treatment of breast cancer, Black women are 40% more likely to die from the disease than White women. At the time of diagnosis, Black women are more likely to have high-grade, more aggressive breast cancer molecular subtypes, and to have had their cancer spread to the lymph nodes. They also tend to be diagnosed at more advanced stages of breast cancer while at the same time, experience higher rates of false-positive screening results.
Although researchers have hypothesized that genetic differences related to African or European ancestry might contribute, studies have not turned up any differences in cancer susceptibility genes by race. Other factors, such as racial differences in the stage of presentation, molecular subtypes, and disparities in treatment, have also emerged as potential culprits.
In her commentary, Dr. Carlos and colleagues wrote that disparities in breast cancer outcomes previously attributed to race need to be examined from multiple angles. This means looking at both the complex interactions between social conditions and policies, which encompass racism both at the individual and structural level, and stressors such as the experience of discrimination in addition to potential biological and genetic contributions.
Many studies now provide evidence for the harmful effects of racism on health. For breast cancer, specifically, studies also suggest that factors such as racial segregation can influence the stage at which Black women get diagnosed and their likelihood of dying from the disease.
However, an important question that remains is what biological changes occur in women exposed to the kind of persistent low-level stress that is associated with structural racism and discrimination, Dr. Carlos said. “We don’t know what stress pathways actually manifest in the body and how they eventually produce the disease.” Studies to address this issue are important, “especially if you would like to develop interventions to prevent or mitigate disease.”
To address this issue, Dr. Carlos and colleagues called upon the research community to conduct both studies that delineate the underlying biology as well as those that test potential interventions – particularly those associated with breast cancer screening outcomes – to try to shed light on why Black women receive more false positives and diagnoses of more aggressive cancer.
Interventions that can target these specific biological pathways could potentially reduce the negative effects of structural racism and discrimination as well as the effects of other social factors that contribute to breast cancer outcomes, “to ultimately help enhance clinical outcomes and close persistent disparities gaps,” the authors wrote.
FROM THE JOURNAL OF CLINICAL ONCOLOGY
U.S. allows pharmacists to prescribe Paxlovid directly
The Food and Drug Administration revised the drug’s emergency use authorization on July 6, letting state-licensed pharmacists screen patients and determine if they are eligible for Paxlovid, according to The Associated Press.
Previously, only doctors could prescribe the antiviral drug, the AP reported. With some limits, pharmacists can now prescribe the medication for patients who face high risks for severe COVID-19.
“The FDA recognizes the important role pharmacists have played and continue to play in combating this pandemic,” Patrizia Cavazzoni, MD, director of the FDA’s Center for Drug Evaluation and Research, said in a statement.
“Since Paxlovid must be taken within 5 days after symptoms begin, authorizing state-licensed pharmacists to prescribe Paxlovid could expand access to timely treatment for some patients who are eligible to receive this drug for the treatment of COVID-19,” she said.
Tom Kraus, the vice president of government relations at the American Society of Health-System Pharmacists, said in a statement that the organization was “pleased to see the FDA remove this barrier to patients’ access to this critical treatment.”
“Pharmacists have played a vital role in our pandemic response efforts and are well-positioned to help patients, particularly those in rural and underserved communities, benefit from this medication,” he said.
But some doctor’s groups questioned the FDA’s move. Jack Resneck Jr., MD, the president of the American Medical Association, said in a statement that prescribing Paxlovid “requires knowledge of a patient’s medical history, as well as clinical monitoring for side effects and follow-up care to determine whether a patient is improving” – requirements that are “far beyond a pharmacist’s scope and training.”
“In the fight against a virus that has killed more than a million people in the United States and is still extremely present and transmissible, patients will get the best, most comprehensive care from physician-led teams – teams that include pharmacists. But, whenever possible, prescribing decisions should be made by a physician with knowledge of a patient’s medical history and the ability to follow up. To ensure the best possible care for COVID-19 patients, we urge people who test positive to discuss treatment options with their physician, if they have one,” he said.
After testing positive for COVID-19, patients should first consider seeking care from their regular health care provider or locating a Test-to-Treat site in their area, the FDA said. Although the latest update allows pharmacists to prescribe Paxlovid, community pharmacies that don’t yet take part in the Test-to-Treat program can decide if they will offer the prescription service to patients.
Paxlovid is authorized to treat mild to moderate COVID-19 in adults and in kids ages 12 and older who weigh at least 88 pounds. Patients who report a positive at-home test are eligible for Paxlovid under the FDA authorization.
If patients want to seek a prescription directly from a pharmacist, they should bring electronic or printed health records from the past year, including their most recent reports of blood work, so the pharmacist can review for kidney or liver problems. Pharmacists can also get this information from the patient’s health care provider.
In addition, patients should bring a list of all medications they are taking, including over-the-counter medications, so the pharmacist can screen for drugs that can have serious interactions with Paxlovid.
Under the limits in the updated FDA authorization, pharmacists should refer patients for more screening if Paxlovid isn’t a good option or if there’s not enough information to find out how well their kidneys or liver works, as well as potential drug interactions.
Paxlovid is intended for people with COVID-19 who face the highest risks for serious disease, the AP reported, including older adults and those with health conditions such as heart disease, obesity, cancer, or diabetes. It isn’t recommended for people with severe kidney or liver problems. A course of treatment requires three pills twice a day for 5 days.
A version of this article first appeared on WebMD.com.
The Food and Drug Administration revised the drug’s emergency use authorization on July 6, letting state-licensed pharmacists screen patients and determine if they are eligible for Paxlovid, according to The Associated Press.
Previously, only doctors could prescribe the antiviral drug, the AP reported. With some limits, pharmacists can now prescribe the medication for patients who face high risks for severe COVID-19.
“The FDA recognizes the important role pharmacists have played and continue to play in combating this pandemic,” Patrizia Cavazzoni, MD, director of the FDA’s Center for Drug Evaluation and Research, said in a statement.
“Since Paxlovid must be taken within 5 days after symptoms begin, authorizing state-licensed pharmacists to prescribe Paxlovid could expand access to timely treatment for some patients who are eligible to receive this drug for the treatment of COVID-19,” she said.
Tom Kraus, the vice president of government relations at the American Society of Health-System Pharmacists, said in a statement that the organization was “pleased to see the FDA remove this barrier to patients’ access to this critical treatment.”
“Pharmacists have played a vital role in our pandemic response efforts and are well-positioned to help patients, particularly those in rural and underserved communities, benefit from this medication,” he said.
But some doctor’s groups questioned the FDA’s move. Jack Resneck Jr., MD, the president of the American Medical Association, said in a statement that prescribing Paxlovid “requires knowledge of a patient’s medical history, as well as clinical monitoring for side effects and follow-up care to determine whether a patient is improving” – requirements that are “far beyond a pharmacist’s scope and training.”
“In the fight against a virus that has killed more than a million people in the United States and is still extremely present and transmissible, patients will get the best, most comprehensive care from physician-led teams – teams that include pharmacists. But, whenever possible, prescribing decisions should be made by a physician with knowledge of a patient’s medical history and the ability to follow up. To ensure the best possible care for COVID-19 patients, we urge people who test positive to discuss treatment options with their physician, if they have one,” he said.
After testing positive for COVID-19, patients should first consider seeking care from their regular health care provider or locating a Test-to-Treat site in their area, the FDA said. Although the latest update allows pharmacists to prescribe Paxlovid, community pharmacies that don’t yet take part in the Test-to-Treat program can decide if they will offer the prescription service to patients.
Paxlovid is authorized to treat mild to moderate COVID-19 in adults and in kids ages 12 and older who weigh at least 88 pounds. Patients who report a positive at-home test are eligible for Paxlovid under the FDA authorization.
If patients want to seek a prescription directly from a pharmacist, they should bring electronic or printed health records from the past year, including their most recent reports of blood work, so the pharmacist can review for kidney or liver problems. Pharmacists can also get this information from the patient’s health care provider.
In addition, patients should bring a list of all medications they are taking, including over-the-counter medications, so the pharmacist can screen for drugs that can have serious interactions with Paxlovid.
Under the limits in the updated FDA authorization, pharmacists should refer patients for more screening if Paxlovid isn’t a good option or if there’s not enough information to find out how well their kidneys or liver works, as well as potential drug interactions.
Paxlovid is intended for people with COVID-19 who face the highest risks for serious disease, the AP reported, including older adults and those with health conditions such as heart disease, obesity, cancer, or diabetes. It isn’t recommended for people with severe kidney or liver problems. A course of treatment requires three pills twice a day for 5 days.
A version of this article first appeared on WebMD.com.
The Food and Drug Administration revised the drug’s emergency use authorization on July 6, letting state-licensed pharmacists screen patients and determine if they are eligible for Paxlovid, according to The Associated Press.
Previously, only doctors could prescribe the antiviral drug, the AP reported. With some limits, pharmacists can now prescribe the medication for patients who face high risks for severe COVID-19.
“The FDA recognizes the important role pharmacists have played and continue to play in combating this pandemic,” Patrizia Cavazzoni, MD, director of the FDA’s Center for Drug Evaluation and Research, said in a statement.
“Since Paxlovid must be taken within 5 days after symptoms begin, authorizing state-licensed pharmacists to prescribe Paxlovid could expand access to timely treatment for some patients who are eligible to receive this drug for the treatment of COVID-19,” she said.
Tom Kraus, the vice president of government relations at the American Society of Health-System Pharmacists, said in a statement that the organization was “pleased to see the FDA remove this barrier to patients’ access to this critical treatment.”
“Pharmacists have played a vital role in our pandemic response efforts and are well-positioned to help patients, particularly those in rural and underserved communities, benefit from this medication,” he said.
But some doctor’s groups questioned the FDA’s move. Jack Resneck Jr., MD, the president of the American Medical Association, said in a statement that prescribing Paxlovid “requires knowledge of a patient’s medical history, as well as clinical monitoring for side effects and follow-up care to determine whether a patient is improving” – requirements that are “far beyond a pharmacist’s scope and training.”
“In the fight against a virus that has killed more than a million people in the United States and is still extremely present and transmissible, patients will get the best, most comprehensive care from physician-led teams – teams that include pharmacists. But, whenever possible, prescribing decisions should be made by a physician with knowledge of a patient’s medical history and the ability to follow up. To ensure the best possible care for COVID-19 patients, we urge people who test positive to discuss treatment options with their physician, if they have one,” he said.
After testing positive for COVID-19, patients should first consider seeking care from their regular health care provider or locating a Test-to-Treat site in their area, the FDA said. Although the latest update allows pharmacists to prescribe Paxlovid, community pharmacies that don’t yet take part in the Test-to-Treat program can decide if they will offer the prescription service to patients.
Paxlovid is authorized to treat mild to moderate COVID-19 in adults and in kids ages 12 and older who weigh at least 88 pounds. Patients who report a positive at-home test are eligible for Paxlovid under the FDA authorization.
If patients want to seek a prescription directly from a pharmacist, they should bring electronic or printed health records from the past year, including their most recent reports of blood work, so the pharmacist can review for kidney or liver problems. Pharmacists can also get this information from the patient’s health care provider.
In addition, patients should bring a list of all medications they are taking, including over-the-counter medications, so the pharmacist can screen for drugs that can have serious interactions with Paxlovid.
Under the limits in the updated FDA authorization, pharmacists should refer patients for more screening if Paxlovid isn’t a good option or if there’s not enough information to find out how well their kidneys or liver works, as well as potential drug interactions.
Paxlovid is intended for people with COVID-19 who face the highest risks for serious disease, the AP reported, including older adults and those with health conditions such as heart disease, obesity, cancer, or diabetes. It isn’t recommended for people with severe kidney or liver problems. A course of treatment requires three pills twice a day for 5 days.
A version of this article first appeared on WebMD.com.
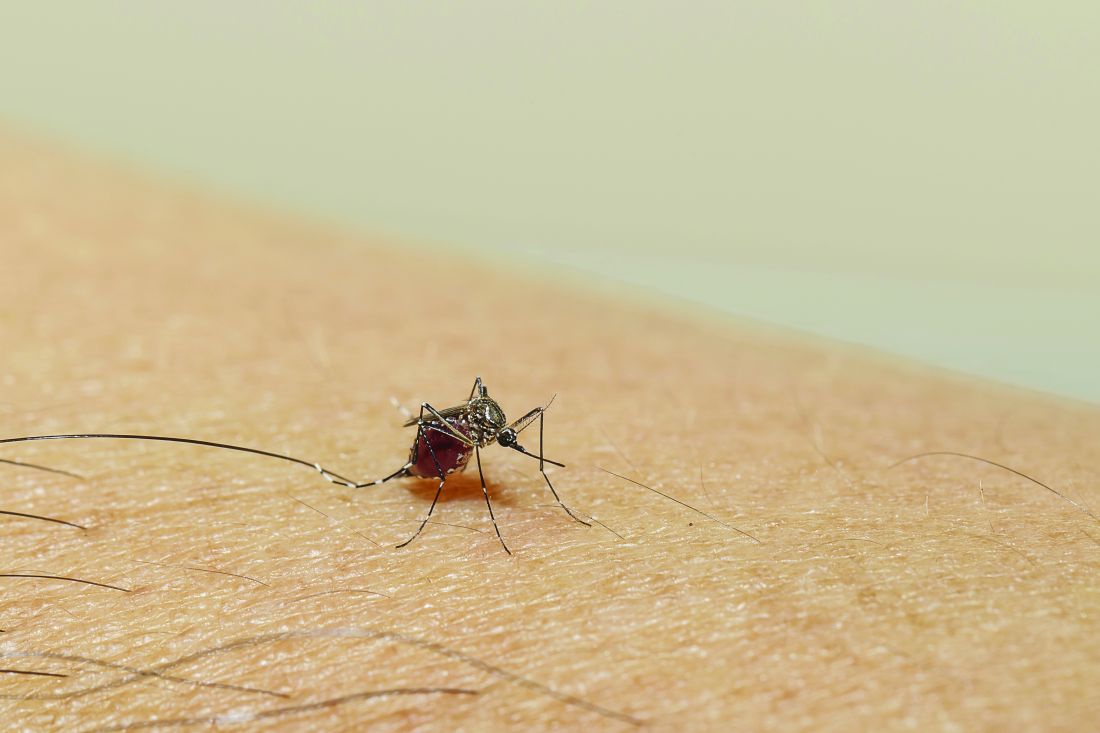
WHO tracking new Omicron subvariant in India
The subvariant, a sublineage of BA.2 being called BA.2.75, has been reported in eight countries and hasn’t yet been declared a variant of concern.
“There’s been an emergence of a ‘could be’ subvariant. It’s been not yet officially called, but some people are referring to it as BA.2.75,” Soumya Swaminathan, MD, the WHO’s chief scientist, said in a video posted on Twitter.
The subvariant appears to have mutations similar to other contagious strains, she said, though there are a limited number of sequences available to analyze. How transmissible and severe it is, and how well it can evade our immunity, aren’t yet known.
“We have to wait and see, and of course, we are tracking it,” Dr. Swaminathan said.
The WHO committee responsible for analyzing global coronavirus data will label the subvariant officially and release more information as the situation warrants it, she said.
Public health experts around the world are also talking about the subvariant, which has been nicknamed Centaurus. BA.2.75 was first found in India in May and is now competing with BA.5, which has become dominant in the United States.
BA.2.75 has eight mutations beyond those seen in BA.5, which “could make immune escape worse than what we’re seeing now,” Eric Topol, MD, founder and director of the Scripps Research Translational Institute and editor-in-chief at Medscape, wrote in a Twitter post.
Individually, the extra mutations aren’t too concerning, “but all appearing together at once is another matter,” Tom Peacock, PhD, a virologist at Imperial College London, wrote in a Twitter post.
The “apparent rapid growth and wide geographical spread” are “worth keeping a close eye on,” he said.
BA.2.75 has been found in a handful of cases in the United States, Australia, Canada, Germany, Japan, New Zealand, and the United Kingdom. In India, the sequence accounts for about 23% of recent samples.
“It is really too early to know if BA.2.75 will take over relative to BA.2 or even relative to BA.5,” Ulrich Elling, PhD, a researcher at Australia’s Institute of Molecular Biotechnology, wrote in a Twitter post.
“Just to emphasize it again: While the distribution across Indian regions as well as internationally and the very rapid appearance makes it likely we are dealing with a variant spreading fast and spread widely already, the absolute data points are few,” he said.
Globally, coronavirus cases have increased nearly 30% during the past 2 weeks, the WHO said July 6. Four out of six of the WHO subregions reported an increase in the last week, with BA.4 and BA.5 driving waves in the United States and Europe.
A version of this article first appeared on WebMD.com.
The subvariant, a sublineage of BA.2 being called BA.2.75, has been reported in eight countries and hasn’t yet been declared a variant of concern.
“There’s been an emergence of a ‘could be’ subvariant. It’s been not yet officially called, but some people are referring to it as BA.2.75,” Soumya Swaminathan, MD, the WHO’s chief scientist, said in a video posted on Twitter.
The subvariant appears to have mutations similar to other contagious strains, she said, though there are a limited number of sequences available to analyze. How transmissible and severe it is, and how well it can evade our immunity, aren’t yet known.
“We have to wait and see, and of course, we are tracking it,” Dr. Swaminathan said.
The WHO committee responsible for analyzing global coronavirus data will label the subvariant officially and release more information as the situation warrants it, she said.
Public health experts around the world are also talking about the subvariant, which has been nicknamed Centaurus. BA.2.75 was first found in India in May and is now competing with BA.5, which has become dominant in the United States.
BA.2.75 has eight mutations beyond those seen in BA.5, which “could make immune escape worse than what we’re seeing now,” Eric Topol, MD, founder and director of the Scripps Research Translational Institute and editor-in-chief at Medscape, wrote in a Twitter post.
Individually, the extra mutations aren’t too concerning, “but all appearing together at once is another matter,” Tom Peacock, PhD, a virologist at Imperial College London, wrote in a Twitter post.
The “apparent rapid growth and wide geographical spread” are “worth keeping a close eye on,” he said.
BA.2.75 has been found in a handful of cases in the United States, Australia, Canada, Germany, Japan, New Zealand, and the United Kingdom. In India, the sequence accounts for about 23% of recent samples.
“It is really too early to know if BA.2.75 will take over relative to BA.2 or even relative to BA.5,” Ulrich Elling, PhD, a researcher at Australia’s Institute of Molecular Biotechnology, wrote in a Twitter post.
“Just to emphasize it again: While the distribution across Indian regions as well as internationally and the very rapid appearance makes it likely we are dealing with a variant spreading fast and spread widely already, the absolute data points are few,” he said.
Globally, coronavirus cases have increased nearly 30% during the past 2 weeks, the WHO said July 6. Four out of six of the WHO subregions reported an increase in the last week, with BA.4 and BA.5 driving waves in the United States and Europe.
A version of this article first appeared on WebMD.com.
The subvariant, a sublineage of BA.2 being called BA.2.75, has been reported in eight countries and hasn’t yet been declared a variant of concern.
“There’s been an emergence of a ‘could be’ subvariant. It’s been not yet officially called, but some people are referring to it as BA.2.75,” Soumya Swaminathan, MD, the WHO’s chief scientist, said in a video posted on Twitter.
The subvariant appears to have mutations similar to other contagious strains, she said, though there are a limited number of sequences available to analyze. How transmissible and severe it is, and how well it can evade our immunity, aren’t yet known.
“We have to wait and see, and of course, we are tracking it,” Dr. Swaminathan said.
The WHO committee responsible for analyzing global coronavirus data will label the subvariant officially and release more information as the situation warrants it, she said.
Public health experts around the world are also talking about the subvariant, which has been nicknamed Centaurus. BA.2.75 was first found in India in May and is now competing with BA.5, which has become dominant in the United States.
BA.2.75 has eight mutations beyond those seen in BA.5, which “could make immune escape worse than what we’re seeing now,” Eric Topol, MD, founder and director of the Scripps Research Translational Institute and editor-in-chief at Medscape, wrote in a Twitter post.
Individually, the extra mutations aren’t too concerning, “but all appearing together at once is another matter,” Tom Peacock, PhD, a virologist at Imperial College London, wrote in a Twitter post.
The “apparent rapid growth and wide geographical spread” are “worth keeping a close eye on,” he said.
BA.2.75 has been found in a handful of cases in the United States, Australia, Canada, Germany, Japan, New Zealand, and the United Kingdom. In India, the sequence accounts for about 23% of recent samples.
“It is really too early to know if BA.2.75 will take over relative to BA.2 or even relative to BA.5,” Ulrich Elling, PhD, a researcher at Australia’s Institute of Molecular Biotechnology, wrote in a Twitter post.
“Just to emphasize it again: While the distribution across Indian regions as well as internationally and the very rapid appearance makes it likely we are dealing with a variant spreading fast and spread widely already, the absolute data points are few,” he said.
Globally, coronavirus cases have increased nearly 30% during the past 2 weeks, the WHO said July 6. Four out of six of the WHO subregions reported an increase in the last week, with BA.4 and BA.5 driving waves in the United States and Europe.
A version of this article first appeared on WebMD.com.
When too much treatment creates more harm than good
Ann Marco, 73, who was diagnosed with ovarian cancer in late 2018, credits her oncology team for saving her life. They treated her with chemotherapy, debulking surgery, and more chemotherapy. But it is her second and current care team that helped restore Ms. Marco’s quality of life, directing her toward such resources as palliative care, physical therapy and counseling for her and her husband.
“I can’t say enough about my palliative care doctor. She helped me manage pain, and the fatigue associated with chemotherapy. When she noticed that my leg was swollen she suspected a blood clot and sent me for an ultrasound,” Ms. Marco said.
The ultrasound revealed that she did indeed have a blood clot, for which she received, and continues to receive, medication. “Because with ovarian cancer, you always have blood clots. So little things like that, though they’re not that little, have really helped me in my journey with this cancer,” Ms. Marco said.
That journey has had its ups and downs. One chemotherapy regimen was so intolerable she decided to discontinue it, with full support of her oncologist. I told her, I just want to live my life, whether that’s only 6 more months or 3 years, but I don’t want to live it like this. And she said, ‘Ann, we’re going to do what you want to do.’”
Nine months later, when her cancer started growing again, Ms. Marco returned to chemotherapy. But this regimen has been much more tolerable, and it also appears to be doing its job. A recent CT scan showed that the tumors are shrinking.
“They’ll never go away. I have metastatic cancer. But they’re smaller, and I was really thrilled about that. It’s the best news I’ve had in more than 3 years,” Ms. Marco said.
End-of-life aggressive care still common
, shows a study published in JCO Oncology Practice.
“We have good evidence that the types of aggressive end-of-life care we looked at in this paper are generally related to a lower quality of life for patients, poorer bereavement outcomes for their families, and even shorter duration survivals,” said lead author Megan A. Mullins, PhD, MPH, a postdoctoral research fellow at the University of Michigan in Ann Arbor. “This suggests there’s a disconnect between what people think aggressive care might do and what it’s doing.”
In their evaluation of variation in end-of-life care, Dr. Mullins and her colleagues analyzed SEER-Medicare data on 6,288 women with ovarian cancer who died between 2016 and 2020. They found that 51% of those women received some form of aggressive cancer care. The most common forms were not being admitted to hospice (28.9%), receiving an invasive procedure (20.7%) and being admitted to an intensive care unit (18.6%).
Dr. Mullins noted that since palliative care was officially recognized as a specialty in 2006, there has been increasing guidance for earlier integration of palliative care and reducing the aggressiveness of end-of-life care; both ASCO and the National Quality Form have standards advising against aggressive end-of-life care.
“But there are a lot of complicated factors that I think make it hard to move the needle in this area,” she said. “For one thing, particularly with ovarian cancer, women tend to have recurrences. I’ve spoken with physicians who got their patients through a difficult patch; they rebounded and they did fine. You don’t know for sure if that’s going to happen again if you try something else. Prognostication is not an exact science.”
Also, end-of-life discussions can be challenging conversations. “Nobody wants to take hope away from their patients. But there’s evidence to show that these conversations don’t actually reduce patients’ hopes – that’s a misconception,” Dr. Mullins said.
“It’s challenging. In the United States, we don’t like to talk about death and dying. But I think having these conversations earlier and more often can help make them a more regular part of care,” she said.
Brittany A. Davidson, MD, a gynecologic oncologist with Duke Health in Durham, N.C., who wrote an accompanying editorial, acknowledges that end-of-life can be fraught with fear, anxiety, and a lot of emotion. But she finds helping patients and their families navigate the ups and downs of their cancer one of the most rewarding aspects of her career as a physician.
“We want to help patients and their family members make these transitions as smoothly as possible,” she said.
A proponent of communications skills training for physicians in general, Dr. Brittany said doctors can learn to identify cues that patients are ready to have conversations about their end-of-life care.
“Those cues will help us facilitate conversations sooner rather than later so we’re not waiting until the very end,” she said.
What these conversations consist of varies depending on where the patient is in her cancer trajectory. In a patient with recurrent ovarian or recurrent uterine cancer, this might start with making sure the patient understands that while their cancer is treatable, it is very unlikely to be curable.
“I have often had patients who have been treated for cancer for several years and didn’t know their cancer wasn’t curable. How many missed opportunities have we overlooked?” Dr. Davidson said.
Then the conversation can turn to the goals of treatment. What’s important to the patient? “Are there events they want to be around for? Symptoms they want to avoid? Some patients really want to know what it’s going to be like to die. I try to take the lead from the patient. Ask what kind of information is helpful to them. Is it numbers? Is it symptoms? It’s really different for everybody,” Dr. Davidson said.
Although Dr. Mullins’s research and Dr. Davidson’s editorial suggest there’s room for improvement toward achieving goal-concordant care in gynecological cancers, Dr. Davidson suspects these patients might be faring a bit better than patients with other types of cancer based on her own anecdotal observations.
“One of the unique things about gynecologic oncology is that we have an amazing longitudinal relationship with our patients – we are not only their surgeons, we’re their oncologists. In other solid tumors, care is fractionated.
“That’s one of the reasons I love gynecologic oncology. I have the opportunity to know my patients through all the stages they experience as part of their cancer. I’d like to think that allows me a better opportunity to get to know them and help them recognize the value of palliative care,” Dr. Mullins said.
Ann Marco, 73, who was diagnosed with ovarian cancer in late 2018, credits her oncology team for saving her life. They treated her with chemotherapy, debulking surgery, and more chemotherapy. But it is her second and current care team that helped restore Ms. Marco’s quality of life, directing her toward such resources as palliative care, physical therapy and counseling for her and her husband.
“I can’t say enough about my palliative care doctor. She helped me manage pain, and the fatigue associated with chemotherapy. When she noticed that my leg was swollen she suspected a blood clot and sent me for an ultrasound,” Ms. Marco said.
The ultrasound revealed that she did indeed have a blood clot, for which she received, and continues to receive, medication. “Because with ovarian cancer, you always have blood clots. So little things like that, though they’re not that little, have really helped me in my journey with this cancer,” Ms. Marco said.
That journey has had its ups and downs. One chemotherapy regimen was so intolerable she decided to discontinue it, with full support of her oncologist. I told her, I just want to live my life, whether that’s only 6 more months or 3 years, but I don’t want to live it like this. And she said, ‘Ann, we’re going to do what you want to do.’”
Nine months later, when her cancer started growing again, Ms. Marco returned to chemotherapy. But this regimen has been much more tolerable, and it also appears to be doing its job. A recent CT scan showed that the tumors are shrinking.
“They’ll never go away. I have metastatic cancer. But they’re smaller, and I was really thrilled about that. It’s the best news I’ve had in more than 3 years,” Ms. Marco said.
End-of-life aggressive care still common
, shows a study published in JCO Oncology Practice.
“We have good evidence that the types of aggressive end-of-life care we looked at in this paper are generally related to a lower quality of life for patients, poorer bereavement outcomes for their families, and even shorter duration survivals,” said lead author Megan A. Mullins, PhD, MPH, a postdoctoral research fellow at the University of Michigan in Ann Arbor. “This suggests there’s a disconnect between what people think aggressive care might do and what it’s doing.”
In their evaluation of variation in end-of-life care, Dr. Mullins and her colleagues analyzed SEER-Medicare data on 6,288 women with ovarian cancer who died between 2016 and 2020. They found that 51% of those women received some form of aggressive cancer care. The most common forms were not being admitted to hospice (28.9%), receiving an invasive procedure (20.7%) and being admitted to an intensive care unit (18.6%).
Dr. Mullins noted that since palliative care was officially recognized as a specialty in 2006, there has been increasing guidance for earlier integration of palliative care and reducing the aggressiveness of end-of-life care; both ASCO and the National Quality Form have standards advising against aggressive end-of-life care.
“But there are a lot of complicated factors that I think make it hard to move the needle in this area,” she said. “For one thing, particularly with ovarian cancer, women tend to have recurrences. I’ve spoken with physicians who got their patients through a difficult patch; they rebounded and they did fine. You don’t know for sure if that’s going to happen again if you try something else. Prognostication is not an exact science.”
Also, end-of-life discussions can be challenging conversations. “Nobody wants to take hope away from their patients. But there’s evidence to show that these conversations don’t actually reduce patients’ hopes – that’s a misconception,” Dr. Mullins said.
“It’s challenging. In the United States, we don’t like to talk about death and dying. But I think having these conversations earlier and more often can help make them a more regular part of care,” she said.
Brittany A. Davidson, MD, a gynecologic oncologist with Duke Health in Durham, N.C., who wrote an accompanying editorial, acknowledges that end-of-life can be fraught with fear, anxiety, and a lot of emotion. But she finds helping patients and their families navigate the ups and downs of their cancer one of the most rewarding aspects of her career as a physician.
“We want to help patients and their family members make these transitions as smoothly as possible,” she said.
A proponent of communications skills training for physicians in general, Dr. Brittany said doctors can learn to identify cues that patients are ready to have conversations about their end-of-life care.
“Those cues will help us facilitate conversations sooner rather than later so we’re not waiting until the very end,” she said.
What these conversations consist of varies depending on where the patient is in her cancer trajectory. In a patient with recurrent ovarian or recurrent uterine cancer, this might start with making sure the patient understands that while their cancer is treatable, it is very unlikely to be curable.
“I have often had patients who have been treated for cancer for several years and didn’t know their cancer wasn’t curable. How many missed opportunities have we overlooked?” Dr. Davidson said.
Then the conversation can turn to the goals of treatment. What’s important to the patient? “Are there events they want to be around for? Symptoms they want to avoid? Some patients really want to know what it’s going to be like to die. I try to take the lead from the patient. Ask what kind of information is helpful to them. Is it numbers? Is it symptoms? It’s really different for everybody,” Dr. Davidson said.
Although Dr. Mullins’s research and Dr. Davidson’s editorial suggest there’s room for improvement toward achieving goal-concordant care in gynecological cancers, Dr. Davidson suspects these patients might be faring a bit better than patients with other types of cancer based on her own anecdotal observations.
“One of the unique things about gynecologic oncology is that we have an amazing longitudinal relationship with our patients – we are not only their surgeons, we’re their oncologists. In other solid tumors, care is fractionated.
“That’s one of the reasons I love gynecologic oncology. I have the opportunity to know my patients through all the stages they experience as part of their cancer. I’d like to think that allows me a better opportunity to get to know them and help them recognize the value of palliative care,” Dr. Mullins said.
Ann Marco, 73, who was diagnosed with ovarian cancer in late 2018, credits her oncology team for saving her life. They treated her with chemotherapy, debulking surgery, and more chemotherapy. But it is her second and current care team that helped restore Ms. Marco’s quality of life, directing her toward such resources as palliative care, physical therapy and counseling for her and her husband.
“I can’t say enough about my palliative care doctor. She helped me manage pain, and the fatigue associated with chemotherapy. When she noticed that my leg was swollen she suspected a blood clot and sent me for an ultrasound,” Ms. Marco said.
The ultrasound revealed that she did indeed have a blood clot, for which she received, and continues to receive, medication. “Because with ovarian cancer, you always have blood clots. So little things like that, though they’re not that little, have really helped me in my journey with this cancer,” Ms. Marco said.
That journey has had its ups and downs. One chemotherapy regimen was so intolerable she decided to discontinue it, with full support of her oncologist. I told her, I just want to live my life, whether that’s only 6 more months or 3 years, but I don’t want to live it like this. And she said, ‘Ann, we’re going to do what you want to do.’”
Nine months later, when her cancer started growing again, Ms. Marco returned to chemotherapy. But this regimen has been much more tolerable, and it also appears to be doing its job. A recent CT scan showed that the tumors are shrinking.
“They’ll never go away. I have metastatic cancer. But they’re smaller, and I was really thrilled about that. It’s the best news I’ve had in more than 3 years,” Ms. Marco said.
End-of-life aggressive care still common
, shows a study published in JCO Oncology Practice.
“We have good evidence that the types of aggressive end-of-life care we looked at in this paper are generally related to a lower quality of life for patients, poorer bereavement outcomes for their families, and even shorter duration survivals,” said lead author Megan A. Mullins, PhD, MPH, a postdoctoral research fellow at the University of Michigan in Ann Arbor. “This suggests there’s a disconnect between what people think aggressive care might do and what it’s doing.”
In their evaluation of variation in end-of-life care, Dr. Mullins and her colleagues analyzed SEER-Medicare data on 6,288 women with ovarian cancer who died between 2016 and 2020. They found that 51% of those women received some form of aggressive cancer care. The most common forms were not being admitted to hospice (28.9%), receiving an invasive procedure (20.7%) and being admitted to an intensive care unit (18.6%).
Dr. Mullins noted that since palliative care was officially recognized as a specialty in 2006, there has been increasing guidance for earlier integration of palliative care and reducing the aggressiveness of end-of-life care; both ASCO and the National Quality Form have standards advising against aggressive end-of-life care.
“But there are a lot of complicated factors that I think make it hard to move the needle in this area,” she said. “For one thing, particularly with ovarian cancer, women tend to have recurrences. I’ve spoken with physicians who got their patients through a difficult patch; they rebounded and they did fine. You don’t know for sure if that’s going to happen again if you try something else. Prognostication is not an exact science.”
Also, end-of-life discussions can be challenging conversations. “Nobody wants to take hope away from their patients. But there’s evidence to show that these conversations don’t actually reduce patients’ hopes – that’s a misconception,” Dr. Mullins said.
“It’s challenging. In the United States, we don’t like to talk about death and dying. But I think having these conversations earlier and more often can help make them a more regular part of care,” she said.
Brittany A. Davidson, MD, a gynecologic oncologist with Duke Health in Durham, N.C., who wrote an accompanying editorial, acknowledges that end-of-life can be fraught with fear, anxiety, and a lot of emotion. But she finds helping patients and their families navigate the ups and downs of their cancer one of the most rewarding aspects of her career as a physician.
“We want to help patients and their family members make these transitions as smoothly as possible,” she said.
A proponent of communications skills training for physicians in general, Dr. Brittany said doctors can learn to identify cues that patients are ready to have conversations about their end-of-life care.
“Those cues will help us facilitate conversations sooner rather than later so we’re not waiting until the very end,” she said.
What these conversations consist of varies depending on where the patient is in her cancer trajectory. In a patient with recurrent ovarian or recurrent uterine cancer, this might start with making sure the patient understands that while their cancer is treatable, it is very unlikely to be curable.
“I have often had patients who have been treated for cancer for several years and didn’t know their cancer wasn’t curable. How many missed opportunities have we overlooked?” Dr. Davidson said.
Then the conversation can turn to the goals of treatment. What’s important to the patient? “Are there events they want to be around for? Symptoms they want to avoid? Some patients really want to know what it’s going to be like to die. I try to take the lead from the patient. Ask what kind of information is helpful to them. Is it numbers? Is it symptoms? It’s really different for everybody,” Dr. Davidson said.
Although Dr. Mullins’s research and Dr. Davidson’s editorial suggest there’s room for improvement toward achieving goal-concordant care in gynecological cancers, Dr. Davidson suspects these patients might be faring a bit better than patients with other types of cancer based on her own anecdotal observations.
“One of the unique things about gynecologic oncology is that we have an amazing longitudinal relationship with our patients – we are not only their surgeons, we’re their oncologists. In other solid tumors, care is fractionated.
“That’s one of the reasons I love gynecologic oncology. I have the opportunity to know my patients through all the stages they experience as part of their cancer. I’d like to think that allows me a better opportunity to get to know them and help them recognize the value of palliative care,” Dr. Mullins said.
Precocious puberty – how early is too soon?
A 6-year-old girl presents with breast development. Her medical history is unremarkable. The parents are of average height, and the mother reports her thelarche was age 11 years. The girl is at the 97th percentile for her height and 90th percentile for her weight. She has Tanner stage 3 breast development and Tanner stage 2 pubic hair development. She has grown slightly more than 3 inches over the past year. How should she be evaluated and managed (N Engl J Med. 2008;358:2366-77)?
The premature onset of puberty, i.e., precocious puberty (PP), can be an emotionally traumatic event for the child and parents. Over the past century, improvements in public health and nutrition, and, more recently, increased obesity, have been associated with earlier puberty and the dominant factor has been attributed to genetics (Curr Opin Endocrinol Diabetes Obes. 2018;25[1]:49-54). This month’s article will focus on understanding what is considered “early” puberty, evaluating for causes, and managing precocious puberty.
More commonly seen in girls than boys, PP is defined as the onset of secondary sexual characteristics before age 7.5 years in Black and Hispanic girls, and prior to 8 years in White girls, which is 2-2.5 standard deviations below the average age of pubertal onset in healthy children (J Pediatr Adolesc Gynecol. 2019;32:455-9). As a comparison, PP is diagnosed with onset before age 9 years in boys. For White compared with Black girls, the average timing of thelarche is age 10 vs. 9.5 years, peak growth velocity is age 11.5, menarche is age 12.5 vs. 12, while completion of puberty is near age 14.5 vs. 13.5, respectively (J Pediatr. 1985;107:317). Fortunately, most girls with PP have common variants rather than serious pathology.
Classification: Central (CPP) vs. peripheral (PPP)
CPP is gonadotropin dependent, meaning the hypothalamic-pituitary-ovarian axis (HPO) is prematurely activated resulting in the normal progression of puberty.
PPP is gonadotropin independent, caused by sex steroid secretion from any source – ovaries, adrenal gland, exogenous or ectopic production, e.g., germ-cell tumor. This results in a disordered progression of pubertal milestones.
Whereas CPP is typically isosexual development, i.e., consistent with the child’s gender, PPP can be isosexual or contrasexual, e.g., virilization of girls. A third classification is “benign or nonprogressive pubertal variants” manifesting as isolated premature thelarche or adrenarche.
Causes (see table)
CPP. Idiopathic causes account for 80%-90% of presentations in girls and 25%-80% in boys. Remarkably, international and domestic adoption, as well as a family history of PP increases the likelihood of CPP in girls. Other etiologies include CNS lesions, e.g., hamartomas, which are the most common cause of PP in young children. MRI with contrast has been the traditional mode of diagnosis for CNS tumors, yet the yield is dubious in girls above age 6. Genetic causes are found in only a small percentage of PP cases. Rarely, CPP can result from gonadotropin-secreting tumors because of elevated luteinizing hormone levels.
PPP. As a result of sex steroid secretion, peripheral causes of PPP include ovarian cysts and ovarian tumors that increase circulating estradiol, such as granulosa cell tumors, which would cause isosexual PPP and Sertoli-Leydig cell tumors that secrete testosterone, which can result in contrasexual PPP. Mild congenital adrenal hyperplasia can result in PPP with virilization (contrasexual) and markedly advanced bone age.
McCune-Albright syndrome is rare and presents with the classic triad of PPP, skin pigmentation called café-au-lait, and fibrous dysplasia of bone. The pathophysiology of McCune-Albright syndrome is autoactivation of the G-protein leading to activation of ovarian tissue that results in formation of large ovarian cysts and extreme elevations in serum estradiol as well as the potential production of other hormones, e.g., thyrotoxicosis, excess growth hormone (acromegaly), and Cushing syndrome.
Premature thelarche. Premature thelarche typically occurs in girls between the ages of 1 and 3 years and is limited to breast enlargement. While no cause has been determined, the plausible explanations include partial activation of the HPO axis, endocrine-disrupting chemicals (EDCs), or a genetic origin. A small percentage of these girls progress to CPP.
EDCs have been considered as potential influencers of early puberty, but no consensus has been established. (Examples of EDCs in the environment include air, soil, or water supply along with food sources, personal care products, and manufactured products that can affect the endocrine system.)
Premature adenarche. Premature adrenarche presents with adult body odor and/or body hair (pubic and/or axillary) in girls who have an elevated body mass index, most commonly at the ages of 6-7 years. The presumed mechanism is normal maturation of the adrenal gland with resultant elevation of circulating androgens. Bone age may be mildly accelerated and DHEAS is prematurely elevated for age. These girls appear to be at increased risk for polycystic ovary syndrome.
Evaluation
The initial step in the evaluation of PP is to determine whether the cause is CPP or PPP; the latter includes distinguishing isosexual from contrasexual development. A thorough history (growth, headaches, behavior or visual change, seizures, abdominal pain), physical exam, including Tanner staging, and bone age is required. However, with isolated premature thelarche or adrenarche, a bone age may not be necessary, as initial close clinical observation for pubertal progression is likely sufficient.
For CPP, the diagnosis is based on serum LH, whether random levels or elevations follow GnRH stimulation. Puberty milestones progress normally although adrenarche is not consistently apparent. For girls younger than age 6, a brain MRI is recommended but not in asymptomatic older girls with CPP. LH and FSH along with estradiol or testosterone, the latter especially in boys, are the first line of serum testing. Serum TSH is recommended for suspicion of primary hypothyroidism. In girls with premature adrenarche, a bone age, testosterone, DHEAS, and 17-OHP to rule out adrenal hyperplasia should be obtained. Pelvic ultrasound may be a useful adjunct to assess uterine volume and/or ovarian cysts/tumors.
Rapidity of onset can also lead the evaluation since a normal growth chart and skeletal maturation suggests a benign pubertal variant whereas a more rapid rate can signal CPP or PPP. Of note, health care providers should ensure prescription, over-the-counter oral or topical sources of hormones, and EDCs are ruled out.
Consequences
An association between childhood sexual abuse and earlier pubertal onset has been cited. These girls may be at increased risk for psychosocial difficulties, menstrual and fertility problems, and even reproductive cancers because of prolonged exposure to sex hormones (J Adolesc Health. 2016;60[1]:65-71).
Treatment
The mainstay of CPP treatment is maximizing adult height, typically through the use of a GnRH agonist for HPO suppression from pituitary downregulation. For girls above age 8 years, attempts at improving adult height have not shown a benefit.
In girls with PPP, treatment is directed at the prevailing pathology. Interestingly, early PPP can activate the HPO axis thereby converting to “secondary” CPP. In PPP, McCune-Albright syndrome treatment targets reducing circulating estrogens through letrozole or tamoxifen as well as addressing other autoactivated hormone production. Ovarian and adrenal tumors, albeit rare, can cause PP; therefore, surgical excision is the goal of treatment.
PP should be approached with equal concerns about the physical and emotional effects while including the family to help them understand the pathophysiology and psychosocial risks.
Dr. Mark P. Trolice is director of The IVF Center in Winter Park, Fla., and professor of obstetrics and gynecology at the University of Central Florida, Orlando.
A 6-year-old girl presents with breast development. Her medical history is unremarkable. The parents are of average height, and the mother reports her thelarche was age 11 years. The girl is at the 97th percentile for her height and 90th percentile for her weight. She has Tanner stage 3 breast development and Tanner stage 2 pubic hair development. She has grown slightly more than 3 inches over the past year. How should she be evaluated and managed (N Engl J Med. 2008;358:2366-77)?
The premature onset of puberty, i.e., precocious puberty (PP), can be an emotionally traumatic event for the child and parents. Over the past century, improvements in public health and nutrition, and, more recently, increased obesity, have been associated with earlier puberty and the dominant factor has been attributed to genetics (Curr Opin Endocrinol Diabetes Obes. 2018;25[1]:49-54). This month’s article will focus on understanding what is considered “early” puberty, evaluating for causes, and managing precocious puberty.
More commonly seen in girls than boys, PP is defined as the onset of secondary sexual characteristics before age 7.5 years in Black and Hispanic girls, and prior to 8 years in White girls, which is 2-2.5 standard deviations below the average age of pubertal onset in healthy children (J Pediatr Adolesc Gynecol. 2019;32:455-9). As a comparison, PP is diagnosed with onset before age 9 years in boys. For White compared with Black girls, the average timing of thelarche is age 10 vs. 9.5 years, peak growth velocity is age 11.5, menarche is age 12.5 vs. 12, while completion of puberty is near age 14.5 vs. 13.5, respectively (J Pediatr. 1985;107:317). Fortunately, most girls with PP have common variants rather than serious pathology.
Classification: Central (CPP) vs. peripheral (PPP)
CPP is gonadotropin dependent, meaning the hypothalamic-pituitary-ovarian axis (HPO) is prematurely activated resulting in the normal progression of puberty.
PPP is gonadotropin independent, caused by sex steroid secretion from any source – ovaries, adrenal gland, exogenous or ectopic production, e.g., germ-cell tumor. This results in a disordered progression of pubertal milestones.
Whereas CPP is typically isosexual development, i.e., consistent with the child’s gender, PPP can be isosexual or contrasexual, e.g., virilization of girls. A third classification is “benign or nonprogressive pubertal variants” manifesting as isolated premature thelarche or adrenarche.
Causes (see table)
CPP. Idiopathic causes account for 80%-90% of presentations in girls and 25%-80% in boys. Remarkably, international and domestic adoption, as well as a family history of PP increases the likelihood of CPP in girls. Other etiologies include CNS lesions, e.g., hamartomas, which are the most common cause of PP in young children. MRI with contrast has been the traditional mode of diagnosis for CNS tumors, yet the yield is dubious in girls above age 6. Genetic causes are found in only a small percentage of PP cases. Rarely, CPP can result from gonadotropin-secreting tumors because of elevated luteinizing hormone levels.
PPP. As a result of sex steroid secretion, peripheral causes of PPP include ovarian cysts and ovarian tumors that increase circulating estradiol, such as granulosa cell tumors, which would cause isosexual PPP and Sertoli-Leydig cell tumors that secrete testosterone, which can result in contrasexual PPP. Mild congenital adrenal hyperplasia can result in PPP with virilization (contrasexual) and markedly advanced bone age.
McCune-Albright syndrome is rare and presents with the classic triad of PPP, skin pigmentation called café-au-lait, and fibrous dysplasia of bone. The pathophysiology of McCune-Albright syndrome is autoactivation of the G-protein leading to activation of ovarian tissue that results in formation of large ovarian cysts and extreme elevations in serum estradiol as well as the potential production of other hormones, e.g., thyrotoxicosis, excess growth hormone (acromegaly), and Cushing syndrome.
Premature thelarche. Premature thelarche typically occurs in girls between the ages of 1 and 3 years and is limited to breast enlargement. While no cause has been determined, the plausible explanations include partial activation of the HPO axis, endocrine-disrupting chemicals (EDCs), or a genetic origin. A small percentage of these girls progress to CPP.
EDCs have been considered as potential influencers of early puberty, but no consensus has been established. (Examples of EDCs in the environment include air, soil, or water supply along with food sources, personal care products, and manufactured products that can affect the endocrine system.)
Premature adenarche. Premature adrenarche presents with adult body odor and/or body hair (pubic and/or axillary) in girls who have an elevated body mass index, most commonly at the ages of 6-7 years. The presumed mechanism is normal maturation of the adrenal gland with resultant elevation of circulating androgens. Bone age may be mildly accelerated and DHEAS is prematurely elevated for age. These girls appear to be at increased risk for polycystic ovary syndrome.
Evaluation
The initial step in the evaluation of PP is to determine whether the cause is CPP or PPP; the latter includes distinguishing isosexual from contrasexual development. A thorough history (growth, headaches, behavior or visual change, seizures, abdominal pain), physical exam, including Tanner staging, and bone age is required. However, with isolated premature thelarche or adrenarche, a bone age may not be necessary, as initial close clinical observation for pubertal progression is likely sufficient.
For CPP, the diagnosis is based on serum LH, whether random levels or elevations follow GnRH stimulation. Puberty milestones progress normally although adrenarche is not consistently apparent. For girls younger than age 6, a brain MRI is recommended but not in asymptomatic older girls with CPP. LH and FSH along with estradiol or testosterone, the latter especially in boys, are the first line of serum testing. Serum TSH is recommended for suspicion of primary hypothyroidism. In girls with premature adrenarche, a bone age, testosterone, DHEAS, and 17-OHP to rule out adrenal hyperplasia should be obtained. Pelvic ultrasound may be a useful adjunct to assess uterine volume and/or ovarian cysts/tumors.
Rapidity of onset can also lead the evaluation since a normal growth chart and skeletal maturation suggests a benign pubertal variant whereas a more rapid rate can signal CPP or PPP. Of note, health care providers should ensure prescription, over-the-counter oral or topical sources of hormones, and EDCs are ruled out.
Consequences
An association between childhood sexual abuse and earlier pubertal onset has been cited. These girls may be at increased risk for psychosocial difficulties, menstrual and fertility problems, and even reproductive cancers because of prolonged exposure to sex hormones (J Adolesc Health. 2016;60[1]:65-71).
Treatment
The mainstay of CPP treatment is maximizing adult height, typically through the use of a GnRH agonist for HPO suppression from pituitary downregulation. For girls above age 8 years, attempts at improving adult height have not shown a benefit.
In girls with PPP, treatment is directed at the prevailing pathology. Interestingly, early PPP can activate the HPO axis thereby converting to “secondary” CPP. In PPP, McCune-Albright syndrome treatment targets reducing circulating estrogens through letrozole or tamoxifen as well as addressing other autoactivated hormone production. Ovarian and adrenal tumors, albeit rare, can cause PP; therefore, surgical excision is the goal of treatment.
PP should be approached with equal concerns about the physical and emotional effects while including the family to help them understand the pathophysiology and psychosocial risks.
Dr. Mark P. Trolice is director of The IVF Center in Winter Park, Fla., and professor of obstetrics and gynecology at the University of Central Florida, Orlando.
A 6-year-old girl presents with breast development. Her medical history is unremarkable. The parents are of average height, and the mother reports her thelarche was age 11 years. The girl is at the 97th percentile for her height and 90th percentile for her weight. She has Tanner stage 3 breast development and Tanner stage 2 pubic hair development. She has grown slightly more than 3 inches over the past year. How should she be evaluated and managed (N Engl J Med. 2008;358:2366-77)?
The premature onset of puberty, i.e., precocious puberty (PP), can be an emotionally traumatic event for the child and parents. Over the past century, improvements in public health and nutrition, and, more recently, increased obesity, have been associated with earlier puberty and the dominant factor has been attributed to genetics (Curr Opin Endocrinol Diabetes Obes. 2018;25[1]:49-54). This month’s article will focus on understanding what is considered “early” puberty, evaluating for causes, and managing precocious puberty.
More commonly seen in girls than boys, PP is defined as the onset of secondary sexual characteristics before age 7.5 years in Black and Hispanic girls, and prior to 8 years in White girls, which is 2-2.5 standard deviations below the average age of pubertal onset in healthy children (J Pediatr Adolesc Gynecol. 2019;32:455-9). As a comparison, PP is diagnosed with onset before age 9 years in boys. For White compared with Black girls, the average timing of thelarche is age 10 vs. 9.5 years, peak growth velocity is age 11.5, menarche is age 12.5 vs. 12, while completion of puberty is near age 14.5 vs. 13.5, respectively (J Pediatr. 1985;107:317). Fortunately, most girls with PP have common variants rather than serious pathology.
Classification: Central (CPP) vs. peripheral (PPP)
CPP is gonadotropin dependent, meaning the hypothalamic-pituitary-ovarian axis (HPO) is prematurely activated resulting in the normal progression of puberty.
PPP is gonadotropin independent, caused by sex steroid secretion from any source – ovaries, adrenal gland, exogenous or ectopic production, e.g., germ-cell tumor. This results in a disordered progression of pubertal milestones.
Whereas CPP is typically isosexual development, i.e., consistent with the child’s gender, PPP can be isosexual or contrasexual, e.g., virilization of girls. A third classification is “benign or nonprogressive pubertal variants” manifesting as isolated premature thelarche or adrenarche.
Causes (see table)
CPP. Idiopathic causes account for 80%-90% of presentations in girls and 25%-80% in boys. Remarkably, international and domestic adoption, as well as a family history of PP increases the likelihood of CPP in girls. Other etiologies include CNS lesions, e.g., hamartomas, which are the most common cause of PP in young children. MRI with contrast has been the traditional mode of diagnosis for CNS tumors, yet the yield is dubious in girls above age 6. Genetic causes are found in only a small percentage of PP cases. Rarely, CPP can result from gonadotropin-secreting tumors because of elevated luteinizing hormone levels.
PPP. As a result of sex steroid secretion, peripheral causes of PPP include ovarian cysts and ovarian tumors that increase circulating estradiol, such as granulosa cell tumors, which would cause isosexual PPP and Sertoli-Leydig cell tumors that secrete testosterone, which can result in contrasexual PPP. Mild congenital adrenal hyperplasia can result in PPP with virilization (contrasexual) and markedly advanced bone age.
McCune-Albright syndrome is rare and presents with the classic triad of PPP, skin pigmentation called café-au-lait, and fibrous dysplasia of bone. The pathophysiology of McCune-Albright syndrome is autoactivation of the G-protein leading to activation of ovarian tissue that results in formation of large ovarian cysts and extreme elevations in serum estradiol as well as the potential production of other hormones, e.g., thyrotoxicosis, excess growth hormone (acromegaly), and Cushing syndrome.
Premature thelarche. Premature thelarche typically occurs in girls between the ages of 1 and 3 years and is limited to breast enlargement. While no cause has been determined, the plausible explanations include partial activation of the HPO axis, endocrine-disrupting chemicals (EDCs), or a genetic origin. A small percentage of these girls progress to CPP.
EDCs have been considered as potential influencers of early puberty, but no consensus has been established. (Examples of EDCs in the environment include air, soil, or water supply along with food sources, personal care products, and manufactured products that can affect the endocrine system.)
Premature adenarche. Premature adrenarche presents with adult body odor and/or body hair (pubic and/or axillary) in girls who have an elevated body mass index, most commonly at the ages of 6-7 years. The presumed mechanism is normal maturation of the adrenal gland with resultant elevation of circulating androgens. Bone age may be mildly accelerated and DHEAS is prematurely elevated for age. These girls appear to be at increased risk for polycystic ovary syndrome.
Evaluation
The initial step in the evaluation of PP is to determine whether the cause is CPP or PPP; the latter includes distinguishing isosexual from contrasexual development. A thorough history (growth, headaches, behavior or visual change, seizures, abdominal pain), physical exam, including Tanner staging, and bone age is required. However, with isolated premature thelarche or adrenarche, a bone age may not be necessary, as initial close clinical observation for pubertal progression is likely sufficient.
For CPP, the diagnosis is based on serum LH, whether random levels or elevations follow GnRH stimulation. Puberty milestones progress normally although adrenarche is not consistently apparent. For girls younger than age 6, a brain MRI is recommended but not in asymptomatic older girls with CPP. LH and FSH along with estradiol or testosterone, the latter especially in boys, are the first line of serum testing. Serum TSH is recommended for suspicion of primary hypothyroidism. In girls with premature adrenarche, a bone age, testosterone, DHEAS, and 17-OHP to rule out adrenal hyperplasia should be obtained. Pelvic ultrasound may be a useful adjunct to assess uterine volume and/or ovarian cysts/tumors.
Rapidity of onset can also lead the evaluation since a normal growth chart and skeletal maturation suggests a benign pubertal variant whereas a more rapid rate can signal CPP or PPP. Of note, health care providers should ensure prescription, over-the-counter oral or topical sources of hormones, and EDCs are ruled out.
Consequences
An association between childhood sexual abuse and earlier pubertal onset has been cited. These girls may be at increased risk for psychosocial difficulties, menstrual and fertility problems, and even reproductive cancers because of prolonged exposure to sex hormones (J Adolesc Health. 2016;60[1]:65-71).
Treatment
The mainstay of CPP treatment is maximizing adult height, typically through the use of a GnRH agonist for HPO suppression from pituitary downregulation. For girls above age 8 years, attempts at improving adult height have not shown a benefit.
In girls with PPP, treatment is directed at the prevailing pathology. Interestingly, early PPP can activate the HPO axis thereby converting to “secondary” CPP. In PPP, McCune-Albright syndrome treatment targets reducing circulating estrogens through letrozole or tamoxifen as well as addressing other autoactivated hormone production. Ovarian and adrenal tumors, albeit rare, can cause PP; therefore, surgical excision is the goal of treatment.
PP should be approached with equal concerns about the physical and emotional effects while including the family to help them understand the pathophysiology and psychosocial risks.
Dr. Mark P. Trolice is director of The IVF Center in Winter Park, Fla., and professor of obstetrics and gynecology at the University of Central Florida, Orlando.
Doc releases song after racist massacre in Buffalo
Physician-musician Cleveland Francis, MD, responded to the recent mass shooting in Buffalo, New York, which left 10 dead, in the only way he knew how. He wrote and recorded a song to honor the victims as “a plea to the other side to recognize us as people,” the Black cardiologist told this news organization.
He couldn’t sleep after the shooting, and “this song was just in my head.” In the 1990s, Dr. Francis took a 3-year sabbatical from medicine to perform and tour as a country singer. He leveraged his Nashville connections to get “Buffalo” produced and recorded.
Acclaimed artist James Threalkill created the accompanying art, titled “The Heavenly Escort of the Buffalo 10,” after listening to a scratch demo.
Dr. Francis doesn’t want people to overlook the massacre as just another gun violence incident because this was “overt hate-crime racism,” he said.
According to the affidavit submitted by FBI agent Christopher J. Dlugokinski, the suspect’s “motive for the mass shooting was to prevent Black people from replacing White people and eliminating the White race, and to inspire others to commit similar attacks.”
Dr. Francis views the Buffalo shooting as distinct from cases like the murder of George Floyd that involved crime or police. It immediately made him think of the Mother Emanuel Church shooting in Charleston, South Carolina. “Having a black skin is now a death warrant,” he said.
The song is also an appeal for White people to fight racism. Dr. Francis is concerned about young men caught up in white supremacy and suggests that we be more alert to children or grandchildren who disconnect from their families, spend time on the dark web, and access guns. The lyrics deliberately don’t mention guns because Dr. Francis wanted to stay out of that debate. “I just sang: ‘What else do I have to do to prove to you that I’m human too?’ ”
Despite his country credentials, Dr. Francis wrote “Buffalo” as a Gospel song because that genre “connects with Black people more and because that civil rights movement was through the church with Dr. Martin Luther King,” he explained. Although he sings all styles of music, the song is performed by Nashville-based singer Michael Lusk so that it’s not a “Cleve Francis thing,” he said, referring to his stage name.
Songwriter Norman Kerner collaborated on the song. The music was produced and recorded by David Thein and mixed by Bob Bullock of Nashville, who Dr. Francis had worked with when he was an artist on Capitol Records.
They sent the video and artwork to the Mayor of Buffalo, Byron Brown, but have yet to hear back. Dr. Francis hopes it could be part of their healing, noting that some people used the song in their Juneteenth celebrations.
The Louisiana native grew up during segregation and was one of two Black students in the Medical College of Virginia class of 1973. After completing his cardiology fellowship, no one would hire him, so Dr. Francis set up his own practice in Northern Virginia. He now works at Inova Heart and Vascular Institute in Alexandria, Va. He remains optimistic about race relations in America and would love a Black pop or Gospel star to record “Buffalo” and bring it to a wider audience.
Dr. Francis is a regular blogger for Medscape. His contribution to country music is recognized in the National Museum of African American History and Culture in Washington, DC. You can find more of his music on YouTube.
A version of this article first appeared on Medscape.com.
Physician-musician Cleveland Francis, MD, responded to the recent mass shooting in Buffalo, New York, which left 10 dead, in the only way he knew how. He wrote and recorded a song to honor the victims as “a plea to the other side to recognize us as people,” the Black cardiologist told this news organization.
He couldn’t sleep after the shooting, and “this song was just in my head.” In the 1990s, Dr. Francis took a 3-year sabbatical from medicine to perform and tour as a country singer. He leveraged his Nashville connections to get “Buffalo” produced and recorded.
Acclaimed artist James Threalkill created the accompanying art, titled “The Heavenly Escort of the Buffalo 10,” after listening to a scratch demo.
Dr. Francis doesn’t want people to overlook the massacre as just another gun violence incident because this was “overt hate-crime racism,” he said.
According to the affidavit submitted by FBI agent Christopher J. Dlugokinski, the suspect’s “motive for the mass shooting was to prevent Black people from replacing White people and eliminating the White race, and to inspire others to commit similar attacks.”
Dr. Francis views the Buffalo shooting as distinct from cases like the murder of George Floyd that involved crime or police. It immediately made him think of the Mother Emanuel Church shooting in Charleston, South Carolina. “Having a black skin is now a death warrant,” he said.
The song is also an appeal for White people to fight racism. Dr. Francis is concerned about young men caught up in white supremacy and suggests that we be more alert to children or grandchildren who disconnect from their families, spend time on the dark web, and access guns. The lyrics deliberately don’t mention guns because Dr. Francis wanted to stay out of that debate. “I just sang: ‘What else do I have to do to prove to you that I’m human too?’ ”
Despite his country credentials, Dr. Francis wrote “Buffalo” as a Gospel song because that genre “connects with Black people more and because that civil rights movement was through the church with Dr. Martin Luther King,” he explained. Although he sings all styles of music, the song is performed by Nashville-based singer Michael Lusk so that it’s not a “Cleve Francis thing,” he said, referring to his stage name.
Songwriter Norman Kerner collaborated on the song. The music was produced and recorded by David Thein and mixed by Bob Bullock of Nashville, who Dr. Francis had worked with when he was an artist on Capitol Records.
They sent the video and artwork to the Mayor of Buffalo, Byron Brown, but have yet to hear back. Dr. Francis hopes it could be part of their healing, noting that some people used the song in their Juneteenth celebrations.
The Louisiana native grew up during segregation and was one of two Black students in the Medical College of Virginia class of 1973. After completing his cardiology fellowship, no one would hire him, so Dr. Francis set up his own practice in Northern Virginia. He now works at Inova Heart and Vascular Institute in Alexandria, Va. He remains optimistic about race relations in America and would love a Black pop or Gospel star to record “Buffalo” and bring it to a wider audience.
Dr. Francis is a regular blogger for Medscape. His contribution to country music is recognized in the National Museum of African American History and Culture in Washington, DC. You can find more of his music on YouTube.
A version of this article first appeared on Medscape.com.
Physician-musician Cleveland Francis, MD, responded to the recent mass shooting in Buffalo, New York, which left 10 dead, in the only way he knew how. He wrote and recorded a song to honor the victims as “a plea to the other side to recognize us as people,” the Black cardiologist told this news organization.
He couldn’t sleep after the shooting, and “this song was just in my head.” In the 1990s, Dr. Francis took a 3-year sabbatical from medicine to perform and tour as a country singer. He leveraged his Nashville connections to get “Buffalo” produced and recorded.
Acclaimed artist James Threalkill created the accompanying art, titled “The Heavenly Escort of the Buffalo 10,” after listening to a scratch demo.
Dr. Francis doesn’t want people to overlook the massacre as just another gun violence incident because this was “overt hate-crime racism,” he said.
According to the affidavit submitted by FBI agent Christopher J. Dlugokinski, the suspect’s “motive for the mass shooting was to prevent Black people from replacing White people and eliminating the White race, and to inspire others to commit similar attacks.”
Dr. Francis views the Buffalo shooting as distinct from cases like the murder of George Floyd that involved crime or police. It immediately made him think of the Mother Emanuel Church shooting in Charleston, South Carolina. “Having a black skin is now a death warrant,” he said.
The song is also an appeal for White people to fight racism. Dr. Francis is concerned about young men caught up in white supremacy and suggests that we be more alert to children or grandchildren who disconnect from their families, spend time on the dark web, and access guns. The lyrics deliberately don’t mention guns because Dr. Francis wanted to stay out of that debate. “I just sang: ‘What else do I have to do to prove to you that I’m human too?’ ”
Despite his country credentials, Dr. Francis wrote “Buffalo” as a Gospel song because that genre “connects with Black people more and because that civil rights movement was through the church with Dr. Martin Luther King,” he explained. Although he sings all styles of music, the song is performed by Nashville-based singer Michael Lusk so that it’s not a “Cleve Francis thing,” he said, referring to his stage name.
Songwriter Norman Kerner collaborated on the song. The music was produced and recorded by David Thein and mixed by Bob Bullock of Nashville, who Dr. Francis had worked with when he was an artist on Capitol Records.
They sent the video and artwork to the Mayor of Buffalo, Byron Brown, but have yet to hear back. Dr. Francis hopes it could be part of their healing, noting that some people used the song in their Juneteenth celebrations.
The Louisiana native grew up during segregation and was one of two Black students in the Medical College of Virginia class of 1973. After completing his cardiology fellowship, no one would hire him, so Dr. Francis set up his own practice in Northern Virginia. He now works at Inova Heart and Vascular Institute in Alexandria, Va. He remains optimistic about race relations in America and would love a Black pop or Gospel star to record “Buffalo” and bring it to a wider audience.
Dr. Francis is a regular blogger for Medscape. His contribution to country music is recognized in the National Museum of African American History and Culture in Washington, DC. You can find more of his music on YouTube.
A version of this article first appeared on Medscape.com.
Nevada sees increase in out-of-state abortion patients
Nevada is already seeing more out-of-state patients seeking an abortion, which state officials expected after the Supreme Court overturned Roe v. Wade.
Las Vegas has seen a 200% increase in patients traveling from Texas, compared with the same time last year, according to the Las Vegas Review-Journal.
Patients are also expected from Arizona, Idaho, Oklahoma, and Utah after the ruling. Abortion providers are preparing for a ripple effect as abortion bans begin across the country.
“We haven’t seen the peak yet,” Kristina Tocce, MD, medical director for Planned Parenthood of the Rocky Mountains and an obstetrician-gynecologist in Colorado, told the newspaper.
“I don’t think we’re going to see any decrease anytime in the near future,” she said.
Nevada made the right to abortion part of state law more than 3 decades ago, in 1990, which protects abortions up to 24 weeks. Colorado passed a similar law this year.
In June, before Roe was overturned, Dr. Tocce said the organization expected abortions to rise by 80% – or about 10,000 patients – in the Rocky Mountain region, which includes southern Nevada, Colorado, and New Mexico.
Even before the ruling took place, Planned Parenthood saw higher numbers of patients as abortion bans took effect in Texas and Oklahoma, she said. After the 6-week ban took place in Texas, about 45% of traveling patients went to Oklahoma. Now that a ban is in place in Oklahoma, patients are going elsewhere.
Las Vegas providers have asked patients why they decided to travel to southern Nevada for services rather than Colorado or New Mexico, which are closer to Texas, Dr. Tocce said. Patients cited several reasons, including direct flight paths, cheaper plane tickets, and the presence of family or friends who could support them.
“We’re going to see such a demand on abortion in any state that has secure access,” Dr. Tocce said. “Patients may be forced to travel further away.”
After Roe was overturned, Nevada Gov. Steve Sisolak held an emergency news conference to reaffirm the state’s commitment to protecting abortion rights. He also said he wasn’t sure about Nevada’s capacity to support out-of-state patients but providers were researching and preparing.
Two Planned Parenthood centers in southern Nevada are adding staff and increasing their hours, Dr. Tocce told the newspaper last month, though there weren’t immediate plans to increase the number of locations or add centers near state borders.
Last week, Governor Sisolak signed an executive order that stops Nevada agencies from helping other states investigate patients seeking an abortion in Nevada. The order also protects patients from extradition and health care providers from losing their license for providing abortion services.
As abortion bans continue to roll out across the U.S., patients will likely consider traveling to states that have certain protections and accessible appointments, Dr. Tocce said.
“We’re in such an ambiguous time right now, we just don’t know what each state is going to attempt to enact,” she said. “My head just swims with all of the possibilities. If that’s challenging for me, I can’t even imagine what it’s going to be like for a patient to navigate.”
A version of this article first appeared on WebMD.com.
Nevada is already seeing more out-of-state patients seeking an abortion, which state officials expected after the Supreme Court overturned Roe v. Wade.
Las Vegas has seen a 200% increase in patients traveling from Texas, compared with the same time last year, according to the Las Vegas Review-Journal.
Patients are also expected from Arizona, Idaho, Oklahoma, and Utah after the ruling. Abortion providers are preparing for a ripple effect as abortion bans begin across the country.
“We haven’t seen the peak yet,” Kristina Tocce, MD, medical director for Planned Parenthood of the Rocky Mountains and an obstetrician-gynecologist in Colorado, told the newspaper.
“I don’t think we’re going to see any decrease anytime in the near future,” she said.
Nevada made the right to abortion part of state law more than 3 decades ago, in 1990, which protects abortions up to 24 weeks. Colorado passed a similar law this year.
In June, before Roe was overturned, Dr. Tocce said the organization expected abortions to rise by 80% – or about 10,000 patients – in the Rocky Mountain region, which includes southern Nevada, Colorado, and New Mexico.
Even before the ruling took place, Planned Parenthood saw higher numbers of patients as abortion bans took effect in Texas and Oklahoma, she said. After the 6-week ban took place in Texas, about 45% of traveling patients went to Oklahoma. Now that a ban is in place in Oklahoma, patients are going elsewhere.
Las Vegas providers have asked patients why they decided to travel to southern Nevada for services rather than Colorado or New Mexico, which are closer to Texas, Dr. Tocce said. Patients cited several reasons, including direct flight paths, cheaper plane tickets, and the presence of family or friends who could support them.
“We’re going to see such a demand on abortion in any state that has secure access,” Dr. Tocce said. “Patients may be forced to travel further away.”
After Roe was overturned, Nevada Gov. Steve Sisolak held an emergency news conference to reaffirm the state’s commitment to protecting abortion rights. He also said he wasn’t sure about Nevada’s capacity to support out-of-state patients but providers were researching and preparing.
Two Planned Parenthood centers in southern Nevada are adding staff and increasing their hours, Dr. Tocce told the newspaper last month, though there weren’t immediate plans to increase the number of locations or add centers near state borders.
Last week, Governor Sisolak signed an executive order that stops Nevada agencies from helping other states investigate patients seeking an abortion in Nevada. The order also protects patients from extradition and health care providers from losing their license for providing abortion services.
As abortion bans continue to roll out across the U.S., patients will likely consider traveling to states that have certain protections and accessible appointments, Dr. Tocce said.
“We’re in such an ambiguous time right now, we just don’t know what each state is going to attempt to enact,” she said. “My head just swims with all of the possibilities. If that’s challenging for me, I can’t even imagine what it’s going to be like for a patient to navigate.”
A version of this article first appeared on WebMD.com.
Nevada is already seeing more out-of-state patients seeking an abortion, which state officials expected after the Supreme Court overturned Roe v. Wade.
Las Vegas has seen a 200% increase in patients traveling from Texas, compared with the same time last year, according to the Las Vegas Review-Journal.
Patients are also expected from Arizona, Idaho, Oklahoma, and Utah after the ruling. Abortion providers are preparing for a ripple effect as abortion bans begin across the country.
“We haven’t seen the peak yet,” Kristina Tocce, MD, medical director for Planned Parenthood of the Rocky Mountains and an obstetrician-gynecologist in Colorado, told the newspaper.
“I don’t think we’re going to see any decrease anytime in the near future,” she said.
Nevada made the right to abortion part of state law more than 3 decades ago, in 1990, which protects abortions up to 24 weeks. Colorado passed a similar law this year.
In June, before Roe was overturned, Dr. Tocce said the organization expected abortions to rise by 80% – or about 10,000 patients – in the Rocky Mountain region, which includes southern Nevada, Colorado, and New Mexico.
Even before the ruling took place, Planned Parenthood saw higher numbers of patients as abortion bans took effect in Texas and Oklahoma, she said. After the 6-week ban took place in Texas, about 45% of traveling patients went to Oklahoma. Now that a ban is in place in Oklahoma, patients are going elsewhere.
Las Vegas providers have asked patients why they decided to travel to southern Nevada for services rather than Colorado or New Mexico, which are closer to Texas, Dr. Tocce said. Patients cited several reasons, including direct flight paths, cheaper plane tickets, and the presence of family or friends who could support them.
“We’re going to see such a demand on abortion in any state that has secure access,” Dr. Tocce said. “Patients may be forced to travel further away.”
After Roe was overturned, Nevada Gov. Steve Sisolak held an emergency news conference to reaffirm the state’s commitment to protecting abortion rights. He also said he wasn’t sure about Nevada’s capacity to support out-of-state patients but providers were researching and preparing.
Two Planned Parenthood centers in southern Nevada are adding staff and increasing their hours, Dr. Tocce told the newspaper last month, though there weren’t immediate plans to increase the number of locations or add centers near state borders.
Last week, Governor Sisolak signed an executive order that stops Nevada agencies from helping other states investigate patients seeking an abortion in Nevada. The order also protects patients from extradition and health care providers from losing their license for providing abortion services.
As abortion bans continue to roll out across the U.S., patients will likely consider traveling to states that have certain protections and accessible appointments, Dr. Tocce said.
“We’re in such an ambiguous time right now, we just don’t know what each state is going to attempt to enact,” she said. “My head just swims with all of the possibilities. If that’s challenging for me, I can’t even imagine what it’s going to be like for a patient to navigate.”
A version of this article first appeared on WebMD.com.
Mosquitoes and the vicious circle that’s gone viral
These viruses want mosquitoes with good taste
Taste can be a pretty subjective sense. Not everyone agrees on what tastes good and what tastes bad. Most people would agree that freshly baked cookies taste good, but what about lima beans? And what about mosquitoes? What tastes good to a mosquito?
The answer? Blood. Blood tastes good to a mosquito. That really wasn’t a very hard question, was it? You did know the answer, didn’t you? They don’t care about cookies, and they certainly don’t care about lima beans. It’s blood that they love.
That brings us back to subjectivity, because it is possible for blood to taste even better. The secret ingredient is dengue … and Zika.
A study just published in Cell demonstrates that mice infected with dengue and Zika viruses release a volatile compound called acetophenone. “We found that flavivirus [like dengue and Zika] can utilize the increased release of acetophenone to help itself achieve its lifecycles more effectively by making their hosts more attractive to mosquito vectors,” senior author Gong Cheng of Tsinghua University, Beijing, said in a written statement.
How do they do it? The viruses, he explained, promote the proliferation of acetophenone-producing skin bacteria. “As a result, some bacteria overreplicate and produce more acetophenone. Suddenly, these sick individuals smell as delicious to mosquitoes as a tray of freshly baked cookies to a group of five-year-old children,” the statement said.
And how do you stop a group of tiny, flying 5-year-olds? That’s right, with acne medication. Really? You knew that one but not the blood one before? The investigators fed isotretinoin to the infected mice, which led to reduced acetophenone release from skin bacteria and made the animals no more attractive to the mosquitoes than their uninfected counterparts.
The investigators are planning to take the next step – feeding isotretinoin to people with dengue and Zika – having gotten the official fictional taste-test approval of celebrity chef Gordon Ramsay, who said, “You’re going to feed this #$^% to sick people? ARE YOU &%*$@#& KIDDING ME?”
Okay, so maybe approval isn’t quite the right word.
Welcome to bladders of the rich and famous!
Don’t you hate it when you’re driving out to your multimillion-dollar second home in the Hamptons and traffic is so bad you absolutely have to find a place to “rest” along the way? But wouldn’t you know it, there just isn’t anywhere to stop! Geez, how do we live?
That’s where David Shusterman, MD, a urologist in New York City and a true American hero, comes in. He’s identified a market and positioned himself as the king of both bladder surgery and “bladder Botox” for the wealthy New Yorkers who regularly make long journeys from the city out to their second homes in the Hamptons. Traffic has increased dramatically on Long Island roads in recent years, and the journey can now taking upward of 4 hours. Some people just can’t make it that long without a bathroom break, and there are very few places to stop along the way.
Dr. Shusterman understands the plight of the Hamptons vacationer, as he told Insider.com: “I can’t tell you how many arguments I personally get into – I’ve lost three friends because I’m the driver and refuse to stop for them.” A tragedy worthy of Shakespeare himself.
During the summer season, Dr. Shusterman performs about 10 prostate artery embolizations a week, an hour-long procedure that shrinks the prostate, which is great for 50- to 60-year-old men with enlarged prostates that cause more frequent bathroom trips. He also performs Botox injections into the bladder once or twice a week for women, which reduces the need to urinate for roughly 6 months. The perfect amount of time to get them through the summer season.
These procedures are sometimes covered by insurance but can cost as much as $20,000 if paid out of pocket. That’s a lot of money to us, but if you’re the sort of person who has a second home in the Hamptons, $20,000 is chump change, especially if it means you won’t have to go 2 entire minutes out of your way to use a gas-station bathroom. Then again, having seen a more than a few gas-station bathrooms in our time, maybe they have a point.
Ditch the apples. Go for the avocados
We’ve all heard about “an apple a day,” but instead of apples you might want to go with avocados.
Avocados are generally thought to be a healthy fat. A study just published in the Journal of the American Heart Association proves that they actually don’t do anything for your waistline but will work wonders on your cholesterol level. The study involved 923 participants who were considered overweight/obese split into two groups: One was asked to consume an avocado a day, and the other continued their usual diets and were asked to consume fewer than two avocados a month.
At the end of the 6 months, the researchers found total cholesterol decreased by an additional 2.9 mg/dL and LDL cholesterol by 2.5 mg/dL in those who ate one avocado every day, compared with the usual-diet group. And even though avocados have a lot of calories, there was no clinical evidence that it impacted weight gain or any cardiometabolic risk factors, according to a statement from Penn State University.
Avocados, then, can be considered a guilt-free food. The findings from this study suggest it can give a substantial boost to your overall quality of diet, in turn lessening your risk of developing type 2 diabetes and some cancers, Kristina Peterson, PhD, assistant professor of nutritional sciences at Texas Tech University, said in the statement.
So get creative with your avocado recipes. You can only eat so much guacamole.
Your nose knows a good friend for you
You’ve probably noticed how dogs sniff other dogs and people before becoming friends. It would be pretty comical if people did the same thing, right? Just walked up to strangers and started sniffing them like dogs?
Well, apparently humans do go by smell when it comes to making friends, and they prefer people who smell like them. Maybe you’ve noticed that your friends look like you, share your values, and think the same way as you. You’re probably right, seeing as previous research has pointed to this.
For the current study, done to show how smell affects human behavior, researchers recruited people who befriended each other quickly, before knowing much about each other. They assumed that the relationships between these same-sex, nonromantic “click friends” relied more on physiological traits, including smell. After collecting samples from the click friends, researchers used an eNose to scan chemical signatures. In another experiment, human volunteers sniffed samples to determine if any were similar. Both experiments showed that click friends had more similar smells than pairs of random people.
“This is not to say that we act like goats or shrews – humans likely rely on other, far more dominant cues in their social decision-making. Nevertheless, our study’s results do suggest that our nose plays a bigger role than previously thought in our choice of friends,” said senior author Noam Sobel, PhD, of the Weizmann Institute of Science in Rehovot, Israel.
Lead author Inbal Ravreby, a graduate student at the institute, put it this way: “These results imply that, as the saying goes, there is chemistry in social chemistry.”
These viruses want mosquitoes with good taste
Taste can be a pretty subjective sense. Not everyone agrees on what tastes good and what tastes bad. Most people would agree that freshly baked cookies taste good, but what about lima beans? And what about mosquitoes? What tastes good to a mosquito?
The answer? Blood. Blood tastes good to a mosquito. That really wasn’t a very hard question, was it? You did know the answer, didn’t you? They don’t care about cookies, and they certainly don’t care about lima beans. It’s blood that they love.
That brings us back to subjectivity, because it is possible for blood to taste even better. The secret ingredient is dengue … and Zika.
A study just published in Cell demonstrates that mice infected with dengue and Zika viruses release a volatile compound called acetophenone. “We found that flavivirus [like dengue and Zika] can utilize the increased release of acetophenone to help itself achieve its lifecycles more effectively by making their hosts more attractive to mosquito vectors,” senior author Gong Cheng of Tsinghua University, Beijing, said in a written statement.
How do they do it? The viruses, he explained, promote the proliferation of acetophenone-producing skin bacteria. “As a result, some bacteria overreplicate and produce more acetophenone. Suddenly, these sick individuals smell as delicious to mosquitoes as a tray of freshly baked cookies to a group of five-year-old children,” the statement said.
And how do you stop a group of tiny, flying 5-year-olds? That’s right, with acne medication. Really? You knew that one but not the blood one before? The investigators fed isotretinoin to the infected mice, which led to reduced acetophenone release from skin bacteria and made the animals no more attractive to the mosquitoes than their uninfected counterparts.
The investigators are planning to take the next step – feeding isotretinoin to people with dengue and Zika – having gotten the official fictional taste-test approval of celebrity chef Gordon Ramsay, who said, “You’re going to feed this #$^% to sick people? ARE YOU &%*$@#& KIDDING ME?”
Okay, so maybe approval isn’t quite the right word.
Welcome to bladders of the rich and famous!
Don’t you hate it when you’re driving out to your multimillion-dollar second home in the Hamptons and traffic is so bad you absolutely have to find a place to “rest” along the way? But wouldn’t you know it, there just isn’t anywhere to stop! Geez, how do we live?
That’s where David Shusterman, MD, a urologist in New York City and a true American hero, comes in. He’s identified a market and positioned himself as the king of both bladder surgery and “bladder Botox” for the wealthy New Yorkers who regularly make long journeys from the city out to their second homes in the Hamptons. Traffic has increased dramatically on Long Island roads in recent years, and the journey can now taking upward of 4 hours. Some people just can’t make it that long without a bathroom break, and there are very few places to stop along the way.
Dr. Shusterman understands the plight of the Hamptons vacationer, as he told Insider.com: “I can’t tell you how many arguments I personally get into – I’ve lost three friends because I’m the driver and refuse to stop for them.” A tragedy worthy of Shakespeare himself.
During the summer season, Dr. Shusterman performs about 10 prostate artery embolizations a week, an hour-long procedure that shrinks the prostate, which is great for 50- to 60-year-old men with enlarged prostates that cause more frequent bathroom trips. He also performs Botox injections into the bladder once or twice a week for women, which reduces the need to urinate for roughly 6 months. The perfect amount of time to get them through the summer season.
These procedures are sometimes covered by insurance but can cost as much as $20,000 if paid out of pocket. That’s a lot of money to us, but if you’re the sort of person who has a second home in the Hamptons, $20,000 is chump change, especially if it means you won’t have to go 2 entire minutes out of your way to use a gas-station bathroom. Then again, having seen a more than a few gas-station bathrooms in our time, maybe they have a point.
Ditch the apples. Go for the avocados
We’ve all heard about “an apple a day,” but instead of apples you might want to go with avocados.
Avocados are generally thought to be a healthy fat. A study just published in the Journal of the American Heart Association proves that they actually don’t do anything for your waistline but will work wonders on your cholesterol level. The study involved 923 participants who were considered overweight/obese split into two groups: One was asked to consume an avocado a day, and the other continued their usual diets and were asked to consume fewer than two avocados a month.
At the end of the 6 months, the researchers found total cholesterol decreased by an additional 2.9 mg/dL and LDL cholesterol by 2.5 mg/dL in those who ate one avocado every day, compared with the usual-diet group. And even though avocados have a lot of calories, there was no clinical evidence that it impacted weight gain or any cardiometabolic risk factors, according to a statement from Penn State University.
Avocados, then, can be considered a guilt-free food. The findings from this study suggest it can give a substantial boost to your overall quality of diet, in turn lessening your risk of developing type 2 diabetes and some cancers, Kristina Peterson, PhD, assistant professor of nutritional sciences at Texas Tech University, said in the statement.
So get creative with your avocado recipes. You can only eat so much guacamole.
Your nose knows a good friend for you
You’ve probably noticed how dogs sniff other dogs and people before becoming friends. It would be pretty comical if people did the same thing, right? Just walked up to strangers and started sniffing them like dogs?
Well, apparently humans do go by smell when it comes to making friends, and they prefer people who smell like them. Maybe you’ve noticed that your friends look like you, share your values, and think the same way as you. You’re probably right, seeing as previous research has pointed to this.
For the current study, done to show how smell affects human behavior, researchers recruited people who befriended each other quickly, before knowing much about each other. They assumed that the relationships between these same-sex, nonromantic “click friends” relied more on physiological traits, including smell. After collecting samples from the click friends, researchers used an eNose to scan chemical signatures. In another experiment, human volunteers sniffed samples to determine if any were similar. Both experiments showed that click friends had more similar smells than pairs of random people.
“This is not to say that we act like goats or shrews – humans likely rely on other, far more dominant cues in their social decision-making. Nevertheless, our study’s results do suggest that our nose plays a bigger role than previously thought in our choice of friends,” said senior author Noam Sobel, PhD, of the Weizmann Institute of Science in Rehovot, Israel.
Lead author Inbal Ravreby, a graduate student at the institute, put it this way: “These results imply that, as the saying goes, there is chemistry in social chemistry.”
These viruses want mosquitoes with good taste
Taste can be a pretty subjective sense. Not everyone agrees on what tastes good and what tastes bad. Most people would agree that freshly baked cookies taste good, but what about lima beans? And what about mosquitoes? What tastes good to a mosquito?
The answer? Blood. Blood tastes good to a mosquito. That really wasn’t a very hard question, was it? You did know the answer, didn’t you? They don’t care about cookies, and they certainly don’t care about lima beans. It’s blood that they love.
That brings us back to subjectivity, because it is possible for blood to taste even better. The secret ingredient is dengue … and Zika.
A study just published in Cell demonstrates that mice infected with dengue and Zika viruses release a volatile compound called acetophenone. “We found that flavivirus [like dengue and Zika] can utilize the increased release of acetophenone to help itself achieve its lifecycles more effectively by making their hosts more attractive to mosquito vectors,” senior author Gong Cheng of Tsinghua University, Beijing, said in a written statement.
How do they do it? The viruses, he explained, promote the proliferation of acetophenone-producing skin bacteria. “As a result, some bacteria overreplicate and produce more acetophenone. Suddenly, these sick individuals smell as delicious to mosquitoes as a tray of freshly baked cookies to a group of five-year-old children,” the statement said.
And how do you stop a group of tiny, flying 5-year-olds? That’s right, with acne medication. Really? You knew that one but not the blood one before? The investigators fed isotretinoin to the infected mice, which led to reduced acetophenone release from skin bacteria and made the animals no more attractive to the mosquitoes than their uninfected counterparts.
The investigators are planning to take the next step – feeding isotretinoin to people with dengue and Zika – having gotten the official fictional taste-test approval of celebrity chef Gordon Ramsay, who said, “You’re going to feed this #$^% to sick people? ARE YOU &%*$@#& KIDDING ME?”
Okay, so maybe approval isn’t quite the right word.
Welcome to bladders of the rich and famous!
Don’t you hate it when you’re driving out to your multimillion-dollar second home in the Hamptons and traffic is so bad you absolutely have to find a place to “rest” along the way? But wouldn’t you know it, there just isn’t anywhere to stop! Geez, how do we live?
That’s where David Shusterman, MD, a urologist in New York City and a true American hero, comes in. He’s identified a market and positioned himself as the king of both bladder surgery and “bladder Botox” for the wealthy New Yorkers who regularly make long journeys from the city out to their second homes in the Hamptons. Traffic has increased dramatically on Long Island roads in recent years, and the journey can now taking upward of 4 hours. Some people just can’t make it that long without a bathroom break, and there are very few places to stop along the way.
Dr. Shusterman understands the plight of the Hamptons vacationer, as he told Insider.com: “I can’t tell you how many arguments I personally get into – I’ve lost three friends because I’m the driver and refuse to stop for them.” A tragedy worthy of Shakespeare himself.
During the summer season, Dr. Shusterman performs about 10 prostate artery embolizations a week, an hour-long procedure that shrinks the prostate, which is great for 50- to 60-year-old men with enlarged prostates that cause more frequent bathroom trips. He also performs Botox injections into the bladder once or twice a week for women, which reduces the need to urinate for roughly 6 months. The perfect amount of time to get them through the summer season.
These procedures are sometimes covered by insurance but can cost as much as $20,000 if paid out of pocket. That’s a lot of money to us, but if you’re the sort of person who has a second home in the Hamptons, $20,000 is chump change, especially if it means you won’t have to go 2 entire minutes out of your way to use a gas-station bathroom. Then again, having seen a more than a few gas-station bathrooms in our time, maybe they have a point.
Ditch the apples. Go for the avocados
We’ve all heard about “an apple a day,” but instead of apples you might want to go with avocados.
Avocados are generally thought to be a healthy fat. A study just published in the Journal of the American Heart Association proves that they actually don’t do anything for your waistline but will work wonders on your cholesterol level. The study involved 923 participants who were considered overweight/obese split into two groups: One was asked to consume an avocado a day, and the other continued their usual diets and were asked to consume fewer than two avocados a month.
At the end of the 6 months, the researchers found total cholesterol decreased by an additional 2.9 mg/dL and LDL cholesterol by 2.5 mg/dL in those who ate one avocado every day, compared with the usual-diet group. And even though avocados have a lot of calories, there was no clinical evidence that it impacted weight gain or any cardiometabolic risk factors, according to a statement from Penn State University.
Avocados, then, can be considered a guilt-free food. The findings from this study suggest it can give a substantial boost to your overall quality of diet, in turn lessening your risk of developing type 2 diabetes and some cancers, Kristina Peterson, PhD, assistant professor of nutritional sciences at Texas Tech University, said in the statement.
So get creative with your avocado recipes. You can only eat so much guacamole.
Your nose knows a good friend for you
You’ve probably noticed how dogs sniff other dogs and people before becoming friends. It would be pretty comical if people did the same thing, right? Just walked up to strangers and started sniffing them like dogs?
Well, apparently humans do go by smell when it comes to making friends, and they prefer people who smell like them. Maybe you’ve noticed that your friends look like you, share your values, and think the same way as you. You’re probably right, seeing as previous research has pointed to this.
For the current study, done to show how smell affects human behavior, researchers recruited people who befriended each other quickly, before knowing much about each other. They assumed that the relationships between these same-sex, nonromantic “click friends” relied more on physiological traits, including smell. After collecting samples from the click friends, researchers used an eNose to scan chemical signatures. In another experiment, human volunteers sniffed samples to determine if any were similar. Both experiments showed that click friends had more similar smells than pairs of random people.
“This is not to say that we act like goats or shrews – humans likely rely on other, far more dominant cues in their social decision-making. Nevertheless, our study’s results do suggest that our nose plays a bigger role than previously thought in our choice of friends,” said senior author Noam Sobel, PhD, of the Weizmann Institute of Science in Rehovot, Israel.
Lead author Inbal Ravreby, a graduate student at the institute, put it this way: “These results imply that, as the saying goes, there is chemistry in social chemistry.”