User login
Bringing you the latest news, research and reviews, exclusive interviews, podcasts, quizzes, and more.
div[contains(@class, 'header__large-screen')]
div[contains(@class, 'read-next-article')]
div[contains(@class, 'main-prefix')]
div[contains(@class, 'nav-primary')]
nav[contains(@class, 'nav-primary')]
section[contains(@class, 'footer-nav-section-wrapper')]
footer[@id='footer']
section[contains(@class, 'nav-hidden')]
div[contains(@class, 'ce-card-content')]
nav[contains(@class, 'nav-ce-stack')]
div[contains(@class, 'view-medstat-quiz-listing-panes')]
div[contains(@class, 'pane-article-sidebar-latest-news')]
div[contains(@class, 'medstat-accordion-set article-series')]
Certolizumab pegol and abatacept show superiority over conventional therapy in early RA
Key clinical point: First-line therapy with biologics, including abatacept and certolizumab pegol but not tocilizumab, all in combination with methotrexate, was clinically superior to conventional therapy with bridging glucocorticoids in patients with moderate-to-severe early rheumatoid arthritis (RA).
Major finding: Compared with active conventional therapy, the Clinical Disease Activity Index-based remission rates at week 48 were significantly higher with abatacept (adjusted difference +20.1%; P < .001) and certolizumab pegol (adjusted difference +13.1%; P = .021) but not with tocilizumab, whereas the radiographic progression was low and lacked between-group differences.
Study details: Findings are from the NORD-STAR trial including 812 treatment-naive patients with moderate-to-severe early RA who were randomly assigned to receive methotrexate combined with active conventional therapy, certolizumab pegol, abatacept, or tocilizumab.
Disclosures: This study was funded by the Academy of Finland, Finska Läkaresällskapet, and other sources. Several authors declared chairing the committees of or receiving travel support, research grants, or fees for lectures, speaking, consultancy, advisory roles, or other services from various sources.
Source: Østergaard M et al, on behalf of the NORD-STAR study group. Certolizumab pegol, abatacept, tocilizumab or active conventional treatment in early rheumatoid arthritis: 48-week clinical and radiographic results of the investigator-initiated randomised controlled NORD-STAR trial. Ann Rheum Dis. 2023 (Jul 9). Doi: 10.1136/ard-2023-224116
Key clinical point: First-line therapy with biologics, including abatacept and certolizumab pegol but not tocilizumab, all in combination with methotrexate, was clinically superior to conventional therapy with bridging glucocorticoids in patients with moderate-to-severe early rheumatoid arthritis (RA).
Major finding: Compared with active conventional therapy, the Clinical Disease Activity Index-based remission rates at week 48 were significantly higher with abatacept (adjusted difference +20.1%; P < .001) and certolizumab pegol (adjusted difference +13.1%; P = .021) but not with tocilizumab, whereas the radiographic progression was low and lacked between-group differences.
Study details: Findings are from the NORD-STAR trial including 812 treatment-naive patients with moderate-to-severe early RA who were randomly assigned to receive methotrexate combined with active conventional therapy, certolizumab pegol, abatacept, or tocilizumab.
Disclosures: This study was funded by the Academy of Finland, Finska Läkaresällskapet, and other sources. Several authors declared chairing the committees of or receiving travel support, research grants, or fees for lectures, speaking, consultancy, advisory roles, or other services from various sources.
Source: Østergaard M et al, on behalf of the NORD-STAR study group. Certolizumab pegol, abatacept, tocilizumab or active conventional treatment in early rheumatoid arthritis: 48-week clinical and radiographic results of the investigator-initiated randomised controlled NORD-STAR trial. Ann Rheum Dis. 2023 (Jul 9). Doi: 10.1136/ard-2023-224116
Key clinical point: First-line therapy with biologics, including abatacept and certolizumab pegol but not tocilizumab, all in combination with methotrexate, was clinically superior to conventional therapy with bridging glucocorticoids in patients with moderate-to-severe early rheumatoid arthritis (RA).
Major finding: Compared with active conventional therapy, the Clinical Disease Activity Index-based remission rates at week 48 were significantly higher with abatacept (adjusted difference +20.1%; P < .001) and certolizumab pegol (adjusted difference +13.1%; P = .021) but not with tocilizumab, whereas the radiographic progression was low and lacked between-group differences.
Study details: Findings are from the NORD-STAR trial including 812 treatment-naive patients with moderate-to-severe early RA who were randomly assigned to receive methotrexate combined with active conventional therapy, certolizumab pegol, abatacept, or tocilizumab.
Disclosures: This study was funded by the Academy of Finland, Finska Läkaresällskapet, and other sources. Several authors declared chairing the committees of or receiving travel support, research grants, or fees for lectures, speaking, consultancy, advisory roles, or other services from various sources.
Source: Østergaard M et al, on behalf of the NORD-STAR study group. Certolizumab pegol, abatacept, tocilizumab or active conventional treatment in early rheumatoid arthritis: 48-week clinical and radiographic results of the investigator-initiated randomised controlled NORD-STAR trial. Ann Rheum Dis. 2023 (Jul 9). Doi: 10.1136/ard-2023-224116
Tofacitinib shows marginal edge over adalimumab in real-world patients with RA
Key clinical point: In patients with rheumatoid arthritis (RA), tofacitinib led to a modest yet statistically significant reduction in disease activity at 3 months compared with adalimumab; however, the reduction in disease activity was not significantly different between the treatment groups at 9 months.
Major finding: The difference in the mean Disease Activity Score in 28 Joints using C-reactive protein between patients treated with tofacitinib vs adalimumab was modest yet statistically significant at 3 months (average treatment effect [ATE] −0.2; P = .02), whereas there was no significant difference at 9 months (ATE −0.03; P = .60).
Study details: This observational study emulated a randomized controlled trial using the data of 842 biologic or targeted synthetic disease-modifying antirheumatic drug-naïve patients with RA from the OPAL dataset who initiated adalimumab (n = 569) or tofacitinib (n = 273).
Disclosures: This study did not declare any specific funding source. Four authors declared being a director of, serving on advisory boards or speakers’ bureaus for, or receiving personal fees for consultancy from various sources.
Source: Deakin CT et al, for the OPAL Rheumatology Network. Comparative effectiveness of adalimumab vs tofacitinib in patients with rheumatoid arthritis in Australia. JAMA Netw Open. 2023;6(6):e2320851 (Jun 29). Doi: 10.1001/jamanetworkopen.2023.20851
Key clinical point: In patients with rheumatoid arthritis (RA), tofacitinib led to a modest yet statistically significant reduction in disease activity at 3 months compared with adalimumab; however, the reduction in disease activity was not significantly different between the treatment groups at 9 months.
Major finding: The difference in the mean Disease Activity Score in 28 Joints using C-reactive protein between patients treated with tofacitinib vs adalimumab was modest yet statistically significant at 3 months (average treatment effect [ATE] −0.2; P = .02), whereas there was no significant difference at 9 months (ATE −0.03; P = .60).
Study details: This observational study emulated a randomized controlled trial using the data of 842 biologic or targeted synthetic disease-modifying antirheumatic drug-naïve patients with RA from the OPAL dataset who initiated adalimumab (n = 569) or tofacitinib (n = 273).
Disclosures: This study did not declare any specific funding source. Four authors declared being a director of, serving on advisory boards or speakers’ bureaus for, or receiving personal fees for consultancy from various sources.
Source: Deakin CT et al, for the OPAL Rheumatology Network. Comparative effectiveness of adalimumab vs tofacitinib in patients with rheumatoid arthritis in Australia. JAMA Netw Open. 2023;6(6):e2320851 (Jun 29). Doi: 10.1001/jamanetworkopen.2023.20851
Key clinical point: In patients with rheumatoid arthritis (RA), tofacitinib led to a modest yet statistically significant reduction in disease activity at 3 months compared with adalimumab; however, the reduction in disease activity was not significantly different between the treatment groups at 9 months.
Major finding: The difference in the mean Disease Activity Score in 28 Joints using C-reactive protein between patients treated with tofacitinib vs adalimumab was modest yet statistically significant at 3 months (average treatment effect [ATE] −0.2; P = .02), whereas there was no significant difference at 9 months (ATE −0.03; P = .60).
Study details: This observational study emulated a randomized controlled trial using the data of 842 biologic or targeted synthetic disease-modifying antirheumatic drug-naïve patients with RA from the OPAL dataset who initiated adalimumab (n = 569) or tofacitinib (n = 273).
Disclosures: This study did not declare any specific funding source. Four authors declared being a director of, serving on advisory boards or speakers’ bureaus for, or receiving personal fees for consultancy from various sources.
Source: Deakin CT et al, for the OPAL Rheumatology Network. Comparative effectiveness of adalimumab vs tofacitinib in patients with rheumatoid arthritis in Australia. JAMA Netw Open. 2023;6(6):e2320851 (Jun 29). Doi: 10.1001/jamanetworkopen.2023.20851
What makes teens choose to use sunscreen?
a cornerstone of skin cancer prevention, according to results from a systematic review.
“We know that skin cancer is one of the most common malignancies in the world, and sun protection methods such as sunscreen make it highly preventable,” first author Carly R. Stevens, a student at Tulane University, New Orleans, said in an interview. “This study demonstrates the adolescent populations that are most vulnerable to sun damage and how we can help mitigate their risk of developing skin cancer through education methods, such as Sun Protection Outreach Teaching by Students.”
Ms. Stevens and coauthors presented the findings during a poster session at the annual meeting of the Society for Pediatric Dermatology.
To investigate predictors of sunscreen use among high school students, they searched PubMed, Embase, and Web of Science using the terms (“sunscreen” or “SPF” or “sun protection”) and (“high school” or “teen” or “teenager” or “adolescent”) and limited the analysis to English studies reporting data on sunscreen use in U.S. high school students up to November 2021.
A total of 20 studies were included in the final review. The study populations ranged in number from 208 to 24,645. Of 11 studies that examined gender, all showed increased sunscreen use in females compared with males. Of five studies that examined age, all showed increased sunscreen use in younger adolescents, compared with their older counterparts.
Of four studies that examined the role of ethnicity on sunscreen use, White students were more likely to use sunscreen, compared with their peers of other ethnicities. “This may be due to perceived sun sensitivity, as [these four studies] also showed increased sunscreen use in populations that believed were more susceptible to sun damage,” the researchers wrote in their abstract.
In other findings, two studies that examined perceived self-efficacy concluded that higher levels of sunscreen use correlated with higher self-efficacy, while four studies concluded that high school students were more likely to use sunscreen if their parents encouraged them the wear it or if the parent used it themselves.
“With 40%-50% of ultraviolet damage being done before the age of 20, it’s crucial that we find ways to educate adolescents on the importance of sunscreen use and target those populations who were found to rarely use sunscreen in our study,” Ms. Stevens said.
In one outreach program, Sun Protection Outreach Teaching by Students (SPOTS), medical students visit middle and high schools to educate them about the importance of practicing sun protection. The program began as a collaboration between Saint Louis University and Washington University in St. Louis, but has expanded nationwide. Ms. Stevens described SPOTS as “a great way for medical students to present the information to middle and high school students in a way that is engaging and interactive.”
The researchers reported having no disclosures.
a cornerstone of skin cancer prevention, according to results from a systematic review.
“We know that skin cancer is one of the most common malignancies in the world, and sun protection methods such as sunscreen make it highly preventable,” first author Carly R. Stevens, a student at Tulane University, New Orleans, said in an interview. “This study demonstrates the adolescent populations that are most vulnerable to sun damage and how we can help mitigate their risk of developing skin cancer through education methods, such as Sun Protection Outreach Teaching by Students.”
Ms. Stevens and coauthors presented the findings during a poster session at the annual meeting of the Society for Pediatric Dermatology.
To investigate predictors of sunscreen use among high school students, they searched PubMed, Embase, and Web of Science using the terms (“sunscreen” or “SPF” or “sun protection”) and (“high school” or “teen” or “teenager” or “adolescent”) and limited the analysis to English studies reporting data on sunscreen use in U.S. high school students up to November 2021.
A total of 20 studies were included in the final review. The study populations ranged in number from 208 to 24,645. Of 11 studies that examined gender, all showed increased sunscreen use in females compared with males. Of five studies that examined age, all showed increased sunscreen use in younger adolescents, compared with their older counterparts.
Of four studies that examined the role of ethnicity on sunscreen use, White students were more likely to use sunscreen, compared with their peers of other ethnicities. “This may be due to perceived sun sensitivity, as [these four studies] also showed increased sunscreen use in populations that believed were more susceptible to sun damage,” the researchers wrote in their abstract.
In other findings, two studies that examined perceived self-efficacy concluded that higher levels of sunscreen use correlated with higher self-efficacy, while four studies concluded that high school students were more likely to use sunscreen if their parents encouraged them the wear it or if the parent used it themselves.
“With 40%-50% of ultraviolet damage being done before the age of 20, it’s crucial that we find ways to educate adolescents on the importance of sunscreen use and target those populations who were found to rarely use sunscreen in our study,” Ms. Stevens said.
In one outreach program, Sun Protection Outreach Teaching by Students (SPOTS), medical students visit middle and high schools to educate them about the importance of practicing sun protection. The program began as a collaboration between Saint Louis University and Washington University in St. Louis, but has expanded nationwide. Ms. Stevens described SPOTS as “a great way for medical students to present the information to middle and high school students in a way that is engaging and interactive.”
The researchers reported having no disclosures.
a cornerstone of skin cancer prevention, according to results from a systematic review.
“We know that skin cancer is one of the most common malignancies in the world, and sun protection methods such as sunscreen make it highly preventable,” first author Carly R. Stevens, a student at Tulane University, New Orleans, said in an interview. “This study demonstrates the adolescent populations that are most vulnerable to sun damage and how we can help mitigate their risk of developing skin cancer through education methods, such as Sun Protection Outreach Teaching by Students.”
Ms. Stevens and coauthors presented the findings during a poster session at the annual meeting of the Society for Pediatric Dermatology.
To investigate predictors of sunscreen use among high school students, they searched PubMed, Embase, and Web of Science using the terms (“sunscreen” or “SPF” or “sun protection”) and (“high school” or “teen” or “teenager” or “adolescent”) and limited the analysis to English studies reporting data on sunscreen use in U.S. high school students up to November 2021.
A total of 20 studies were included in the final review. The study populations ranged in number from 208 to 24,645. Of 11 studies that examined gender, all showed increased sunscreen use in females compared with males. Of five studies that examined age, all showed increased sunscreen use in younger adolescents, compared with their older counterparts.
Of four studies that examined the role of ethnicity on sunscreen use, White students were more likely to use sunscreen, compared with their peers of other ethnicities. “This may be due to perceived sun sensitivity, as [these four studies] also showed increased sunscreen use in populations that believed were more susceptible to sun damage,” the researchers wrote in their abstract.
In other findings, two studies that examined perceived self-efficacy concluded that higher levels of sunscreen use correlated with higher self-efficacy, while four studies concluded that high school students were more likely to use sunscreen if their parents encouraged them the wear it or if the parent used it themselves.
“With 40%-50% of ultraviolet damage being done before the age of 20, it’s crucial that we find ways to educate adolescents on the importance of sunscreen use and target those populations who were found to rarely use sunscreen in our study,” Ms. Stevens said.
In one outreach program, Sun Protection Outreach Teaching by Students (SPOTS), medical students visit middle and high schools to educate them about the importance of practicing sun protection. The program began as a collaboration between Saint Louis University and Washington University in St. Louis, but has expanded nationwide. Ms. Stevens described SPOTS as “a great way for medical students to present the information to middle and high school students in a way that is engaging and interactive.”
The researchers reported having no disclosures.
FROM SPD 2023
Infection-related chronic illness: A new paradigm for research and treatment
Experience with long COVID has shone a spotlight on persistent Lyme disease and other often debilitating chronic illnesses that follow known or suspected infections – and on the urgent need for a common and well-funded research agenda, education of physicians, growth of multidisciplinary clinics, and financially supported clinical care.
“We critically need to understand the epidemiology and pathogenesis of chronic symptoms, and identify more effective ways to manage, treat, and potentially cure these illnesses,” Lyle Petersen, MD, MPH, director of the division of vector-borne diseases at the Centers for Disease Control and Prevention, said at the start of a 2-day National Academies of Science, Engineering, and Medicine (NASEM) workshop, “Toward a Common Research Agenda in Infection-Associated Chronic Illnesses.”
Thinking about infection-associated chronic illnesses as an entity – one predicated on commonalities in chronic symptoms and in leading hypotheses for causes – represents a paradigm shift that researchers and patient advocates said can avoid research redundancies and is essential to address what the NASEM calls an overlooked, growing public health problem.
An estimated 2 million people in the United States are living with what’s called posttreatment Lyme disease (PTLD) – a subset of patients with persistent or chronic Lyme disease – and an estimated 1.7-3.3 million people in the United States have diagnoses of myalgic encephalomyelitis/chronic fatigue syndrome (ME/CFS). More than 700,000 people are living with multiple sclerosis. And as of January 2023, 11% of people in the United States reported having long COVID symptoms; the incidence of long COVID is currently estimated at 10%-30% of nonhospitalized cases of COVID-19.
These illnesses “have come under one umbrella,” said Avindra Nath, MD, clinical director of the National Institute of Neurologic Disorders and Stroke (NINDS), Bethesda, Md.
To date, common ground in the literature has grown largely around long COVID and ME/CFS, the latter of which is often associated with a prior, often unidentified infection.
Symptoms of both have been “rigorously” studied and shown to have overlaps, and the illnesses appear to share underlying biologic abnormalities in metabolism and the gut microbiome, as well as viral reactivation and abnormalities in the immune system, central and autonomic nervous systems, and the cardiovascular and pulmonary systems, said Anthony L. Komaroff, MD, professor of medicine at Harvard Medical School and a senior physician at Brigham & Women’s Hospital, both in Boston. (An estimated half of patients with long COVID meet the diagnostic criteria for ME/CFS.)
Although less thoroughly researched, similar symptoms are experienced by a subset of people following a variety of viral, bacterial, and protozoal infections, Dr. Komaroff said. To be determined, he said, is whether the pathophysiology believed to be shared by long COVID and ME/CFS is also shared with other postinfectious syndromes following acute illness with Ebola, West Nile, dengue, mycoplasma pneumonia, enteroviruses, and other pathogens, he said.
Persistent infection, viral reactivation
RNA viral infections can lead to persistent inflammation and dysregulated immunity, with or without viral persistence over time, Timothy J. Henrich, MD, MMSc, associate professor of medicine at the University of California, San Francisco, said in a keynote address.
Research on Ebola survivors has documented long-lasting inflammation and severe immune dysfunction 2 years after infection, for instance. And it’s well known that HIV-1 leads to aberrant immune responses, inflammation, and organ damage despite antiretroviral therapy, said Dr. Henrich, who leads a laboratory/research group that studies approaches to HIV-1 cure and PET-based imaging approaches to characterize viral reservoirs and immune sequelae.
Viral persistence, which can be difficult to measure, has also been documented in Ebola survivors. And in patients living with HIV-1, HIV-1 RNA and protein expression have been shown to persist, again despite antiretroviral therapy. The UCSF Long-Term Immunological Impact of Novel Coronavirus (LIINC) study, for which Dr. Henrich is the principal investigator, found spike RNA in colorectal tissue more than 22 months post COVID, and other research documented viral protein in gut tissue for up to 6 months, he said.
“I think we’re appreciating now, in at least the scientific and treatment community, that there’s a potential for ‘acute’ infections to exhibit some degree of persistence leading to clinical morbidity,” said Dr. Henrich, one of several speakers to describe reports of pathogen persistence. Regarding long COVID, its “etiology is likely heterogeneous,” he said, but persistence of SARS-CoV-2 “may lay behind” other described mechanisms, from clotting/microvascular dysfunction to inflammation and tissue damage to immune dysregulation.
Reactivation of existing latent viral infections in the setting of new acute microbial illness may also play an etiologic role in chronic illnesses, Dr. Henrich said. Epstein-Barr virus (EBV) reactivation has been shown in some studies, including their UCSF COVID-19 cohort, to be associated with long COVID.
“Physicians have been trained to be skeptical about the role [of latent viral infections],” Michael Peluso, MD, an infectious disease physician and assistant professor at UCSF, said during a talk on viral reactivation. This skepticism needs to be “reexamined and overcome,” he said.
Herpesviruses have frequently been associated with ME/CFS, he noted. And evidence of a strong association between EBV and multiple sclerosis came recently from a prospective study of 10 million military recruits that found a 32-fold increased risk of MS after EBV infection but no increase after infection with other viruses, Dr. Peluso and Dr. Henrich both noted.
Research needs, treatment trials
Research needs are vast: The need to learn more about the mechanisms of pathogen persistence and immune evasion, for instance, and the need for more biomarker studies, more imaging studies and tissue analyses, more study of microbiome composition and activity, and continued development and application of metagenomic next-generation sequencing.
Workshop participants also spoke of the need to better understand the molecular mimicry that can occur between pathogen-produced proteins and self-antigens, for instance, and the effects of inflammation and infection-related immune changes on neuronal and microglial function in the brain.
“We should perform similar forms of analysis [across] patients with different infection-associated chronic conditions,” said Amy Proal, PhD, president of the PolyBio Research Foundation, which funds research on infection-associated chronic infections. And within individual conditions and well-characterized study groups “we should perform many different forms of analysis … so we can define endotypes and get more solid biomarkers so that industry [will have more confidence] to run clinical trials.”
In the meantime, patients need fast-moving treatment trials for long COVID, long Lyme, and other infection-associated chronic illnesses, speakers emphasized. “We all agree that treatment trials are overdue,” said the NINDS’ Dr. Nath. “We can’t afford to wait for another decade until we understand all the mechanisms, but rather we can do clinical trials based on what we understand now and study the pathophysiology in the context of the clinical trials.”
Just as was done with HIV, said Steven G. Deeks, MD, professor of medicine at UCSF, researchers must “practice experimental medicine” and select pathways and mechanisms of interest, interrupt those pathways in a controlled manner, and assess impact. “Much of this can be done by repurposing existing drugs,” he said, like antivirals for persistent viral infection, EBV-directed therapies for EBV reactivation, anti-inflammatory drugs for inflammation, B–cell-directed therapies for autoantibodies, and antiplatelet drugs for microvascular disease.
When done correctly, he said, such “probe” studies can deepen mechanistic understandings, lead to biomarkers, and provide proof-of-concept that “will encourage massive investment in developing new therapies” for long COVID and other infection-associated chronic illnesses.
Trials of treatments for long COVID “are starting, so I’m optimistic,” said Dr. Deeks, an expert on HIV pathogenesis and treatment and a principal investigator of the Researching COVID to Enhance Recovery (RECOVER) study. Among the trials: A study of intravenous immunoglobulin (IVIG) for neurologic long COVID; a study of an anti-SARS-CoV-2 monoclonal antibody that can deplete tissue/cellular reservoirs of viral particles (replicating or not); and a study evaluating baricitinib (Olumiant), a Janus kinase inhibitor, for neurocognitive impairment and cardiopulmonary symptoms of long COVID.
Alessio Fasano, MD, professor of pediatrics at Harvard Medical School and professor of nutrition at the Harvard T.H. Chan School of Public Health, Boston, described at the workshop how he began investigating the use of larazotide acetate – an inhibitor of the protein zonulin, which increases intestinal permeability – in children with COVID-19 Multisystem Inflammatory Syndrome (MIS-C) after learning that SARS-CoV-2 viral particles persist in the gastrointestinal tract, causing dysbiosis and zonulin upregulation.
In an ongoing phase 2, double-blind, placebo-controlled trial, the agent thus far has expedited the resolution of gastrointestinal symptoms and clearance of spike protein from the circulation, he said. A phase 2 trial of the agent for pediatric patients with long COVID and SARS-CoV-2 antigenemia is underway. “What if we were to stop [chains of events] by stopping the passage of elements from the virus into circulation?’ he said.
In the realm of Lyme disease, a recently launched Clinical Trials Network for Lyme and Other Tick-Borne Diseases has awarded pilot study grants to evaluate treatments aimed at a variety of possible disease mechanisms that, notably, are similar to those of other chronic illnesses: persistence of infection or remnants of infection, immune dysregulation and autoimmune reactions, neural dysfunction, and gut microbiome changes. (Microclots and mitochondrial dysfunction have not been as well studied in Lyme.)
Current and upcoming studies include evaluations of transcutaneous auricular vagus nerve stimulation for those with persistent Lyme fatigue, transcranial direct current stimulation with cognitive retraining for Lyme brain fog, and tetracycline for PTLD, said Brian Fallon, MD, MPH, professor of clinical psychiatry at Columbia University, New York, who directs the Lyme & Tick-Borne Diseases Research Center and the coordinating center of the new network.
Moving forward, he said, it is important to loosen exclusion criteria and include patients with “probable or possible” Lyme and those with suspected infections with other tick-borne pathogens. All told, these patients comprise a large portion of those with chronic symptoms and have been neglected in an already thin research space, Dr. Fallon said, noting that “there haven’t been any clinical trials of posttreatment Lyme disease in ages – in 10-15 years.”
(PTLD refers to symptoms lasting for more than 6 months after the completion of standard Infectious Diseases Society of America–recommended antibiotic protocols. It occurs in about 15% of patients, said John Aucott, MD, director of the Johns Hopkins Lyme Disease Research Center, Baltimore, a member of the new clinical trials network.)
Calls for a new NIH center and patient involvement
Patients and patient advocacy organizations have played a vital role in research thus far: They’ve documented post-COVID symptoms that academic researchers said they would not otherwise have known of. Leaders of the Patient-Led Research Collaborative have coauthored published reviews with leading long COVID experts. And patients with tick-borne illnesses have enrolled in the MyLymeData patient registry run by LymeDisease.org, which has documented patient-experienced efficacy of alternative treatments and described antibiotic responders and nonresponders.
At the workshop, they shared findings alongside academic experts, and researchers called for their continued involvement. “Patient engagement at every step of the research process is critical,” Dr. Nath said.
“We need to ensure that research is reflective of lived experiences … and [that we’re] accelerating clinical trials of therapeutics that are of priority to the patient community,” said Lisa McCorkell, cofounder of the long COVID-focused Patient-Led Research Collaborative.
Ms. McCorkell also called for the creation of an office for infection-associated chronic illnesses in the NIH director’s office. Others voiced their support. “I think it’s a great idea to have an NIH center for infection-associated chronic illnesses,” said Dr. Fallon. “I think it would have a profound impact.”
The other great need, of course, is funding. “We have ideas, we have drugs that can be repurposed, we have a highly informed and engaged community that will enroll in and be retained in studies, and we have outcomes we can measure,” Dr. Deeks said. “What we’re missing is industry engagement and funding. We need massive engagement from the NIH.”
Real-world treatment needs
In the meantime, patients are seeking treatment, and “clinicians need to have uncertainty tolerance” and try multiple treatments simultaneously, said David Putrino, PT, PhD, director of rehabilitation innovation for the Mount Sinai Health System and professor of rehabilitation and human performance at the Icahn School of Medicine at Mount Sinai, New York. He oversees a multidisciplinary hybrid clinical care research center that has seen over 1,500 patients with long COVID and is beginning to see patients with other infection-associated chronic illnesses.
It’s a model that should be replicated to help fill the “enormous unmet clinical need” of patients with infection-associated chronic illness, said Peter Rowe, MD, professor of pediatrics at the Johns Hopkins School of Medicine and an expert on ME/CFS. And “as we request [more research funding], we will also need [financial] support for clinical care,” he emphasized, to provide equitable access for patients and to attract treating physicians.
Moreover, said Linda Geng, MD, PhD, the culture of stigma needs to change. Right now, patients with long COVID often feel dismissed not only by friends, families, and coworkers, but by clinicians who find it find it hard “to grasp that this is real and a biological condition.”
And it’s not just conditions such as long COVID that are stigmatized, but treatments as well, she said. For instance, some clinicians view low-dose naltrexone, a treatment increasingly being used for inflammation, with suspicion because it is used for opioid use disorder and alcohol use disorder – or because the “low-dose” label summons mistrust of homeopathy. “Even with therapies, there are preconceived notions and biases,” said Dr. Geng, cofounder and codirector of the Stanford (Calif.) Long COVID program.
“What almost killed me,” said Meghan O’Rourke, who has ongoing effects from long-undiagnosed tick-borne illness, “was the invisibility of the illness.” Ms. O’Rourke teaches at Yale University and is the author of “The Invisible Kingdom: Reimagining Chronic Illness.”
Teaching young physicians about these illnesses would help, she and others said. During a question and answer session, Dr. Putrino shared that the Icahn School of Medicine has recently committed to “create a complex chronic illness medical curriculum” that will impact medical education from the first year of medical school through residencies. Dr. Putrino said his team is also working on materials to help other clinics develop care models similar to those at his Mount Sinai clinic.
The NASEM workshop did not collect or require disclosures of its participants.
Experience with long COVID has shone a spotlight on persistent Lyme disease and other often debilitating chronic illnesses that follow known or suspected infections – and on the urgent need for a common and well-funded research agenda, education of physicians, growth of multidisciplinary clinics, and financially supported clinical care.
“We critically need to understand the epidemiology and pathogenesis of chronic symptoms, and identify more effective ways to manage, treat, and potentially cure these illnesses,” Lyle Petersen, MD, MPH, director of the division of vector-borne diseases at the Centers for Disease Control and Prevention, said at the start of a 2-day National Academies of Science, Engineering, and Medicine (NASEM) workshop, “Toward a Common Research Agenda in Infection-Associated Chronic Illnesses.”
Thinking about infection-associated chronic illnesses as an entity – one predicated on commonalities in chronic symptoms and in leading hypotheses for causes – represents a paradigm shift that researchers and patient advocates said can avoid research redundancies and is essential to address what the NASEM calls an overlooked, growing public health problem.
An estimated 2 million people in the United States are living with what’s called posttreatment Lyme disease (PTLD) – a subset of patients with persistent or chronic Lyme disease – and an estimated 1.7-3.3 million people in the United States have diagnoses of myalgic encephalomyelitis/chronic fatigue syndrome (ME/CFS). More than 700,000 people are living with multiple sclerosis. And as of January 2023, 11% of people in the United States reported having long COVID symptoms; the incidence of long COVID is currently estimated at 10%-30% of nonhospitalized cases of COVID-19.
These illnesses “have come under one umbrella,” said Avindra Nath, MD, clinical director of the National Institute of Neurologic Disorders and Stroke (NINDS), Bethesda, Md.
To date, common ground in the literature has grown largely around long COVID and ME/CFS, the latter of which is often associated with a prior, often unidentified infection.
Symptoms of both have been “rigorously” studied and shown to have overlaps, and the illnesses appear to share underlying biologic abnormalities in metabolism and the gut microbiome, as well as viral reactivation and abnormalities in the immune system, central and autonomic nervous systems, and the cardiovascular and pulmonary systems, said Anthony L. Komaroff, MD, professor of medicine at Harvard Medical School and a senior physician at Brigham & Women’s Hospital, both in Boston. (An estimated half of patients with long COVID meet the diagnostic criteria for ME/CFS.)
Although less thoroughly researched, similar symptoms are experienced by a subset of people following a variety of viral, bacterial, and protozoal infections, Dr. Komaroff said. To be determined, he said, is whether the pathophysiology believed to be shared by long COVID and ME/CFS is also shared with other postinfectious syndromes following acute illness with Ebola, West Nile, dengue, mycoplasma pneumonia, enteroviruses, and other pathogens, he said.
Persistent infection, viral reactivation
RNA viral infections can lead to persistent inflammation and dysregulated immunity, with or without viral persistence over time, Timothy J. Henrich, MD, MMSc, associate professor of medicine at the University of California, San Francisco, said in a keynote address.
Research on Ebola survivors has documented long-lasting inflammation and severe immune dysfunction 2 years after infection, for instance. And it’s well known that HIV-1 leads to aberrant immune responses, inflammation, and organ damage despite antiretroviral therapy, said Dr. Henrich, who leads a laboratory/research group that studies approaches to HIV-1 cure and PET-based imaging approaches to characterize viral reservoirs and immune sequelae.
Viral persistence, which can be difficult to measure, has also been documented in Ebola survivors. And in patients living with HIV-1, HIV-1 RNA and protein expression have been shown to persist, again despite antiretroviral therapy. The UCSF Long-Term Immunological Impact of Novel Coronavirus (LIINC) study, for which Dr. Henrich is the principal investigator, found spike RNA in colorectal tissue more than 22 months post COVID, and other research documented viral protein in gut tissue for up to 6 months, he said.
“I think we’re appreciating now, in at least the scientific and treatment community, that there’s a potential for ‘acute’ infections to exhibit some degree of persistence leading to clinical morbidity,” said Dr. Henrich, one of several speakers to describe reports of pathogen persistence. Regarding long COVID, its “etiology is likely heterogeneous,” he said, but persistence of SARS-CoV-2 “may lay behind” other described mechanisms, from clotting/microvascular dysfunction to inflammation and tissue damage to immune dysregulation.
Reactivation of existing latent viral infections in the setting of new acute microbial illness may also play an etiologic role in chronic illnesses, Dr. Henrich said. Epstein-Barr virus (EBV) reactivation has been shown in some studies, including their UCSF COVID-19 cohort, to be associated with long COVID.
“Physicians have been trained to be skeptical about the role [of latent viral infections],” Michael Peluso, MD, an infectious disease physician and assistant professor at UCSF, said during a talk on viral reactivation. This skepticism needs to be “reexamined and overcome,” he said.
Herpesviruses have frequently been associated with ME/CFS, he noted. And evidence of a strong association between EBV and multiple sclerosis came recently from a prospective study of 10 million military recruits that found a 32-fold increased risk of MS after EBV infection but no increase after infection with other viruses, Dr. Peluso and Dr. Henrich both noted.
Research needs, treatment trials
Research needs are vast: The need to learn more about the mechanisms of pathogen persistence and immune evasion, for instance, and the need for more biomarker studies, more imaging studies and tissue analyses, more study of microbiome composition and activity, and continued development and application of metagenomic next-generation sequencing.
Workshop participants also spoke of the need to better understand the molecular mimicry that can occur between pathogen-produced proteins and self-antigens, for instance, and the effects of inflammation and infection-related immune changes on neuronal and microglial function in the brain.
“We should perform similar forms of analysis [across] patients with different infection-associated chronic conditions,” said Amy Proal, PhD, president of the PolyBio Research Foundation, which funds research on infection-associated chronic infections. And within individual conditions and well-characterized study groups “we should perform many different forms of analysis … so we can define endotypes and get more solid biomarkers so that industry [will have more confidence] to run clinical trials.”
In the meantime, patients need fast-moving treatment trials for long COVID, long Lyme, and other infection-associated chronic illnesses, speakers emphasized. “We all agree that treatment trials are overdue,” said the NINDS’ Dr. Nath. “We can’t afford to wait for another decade until we understand all the mechanisms, but rather we can do clinical trials based on what we understand now and study the pathophysiology in the context of the clinical trials.”
Just as was done with HIV, said Steven G. Deeks, MD, professor of medicine at UCSF, researchers must “practice experimental medicine” and select pathways and mechanisms of interest, interrupt those pathways in a controlled manner, and assess impact. “Much of this can be done by repurposing existing drugs,” he said, like antivirals for persistent viral infection, EBV-directed therapies for EBV reactivation, anti-inflammatory drugs for inflammation, B–cell-directed therapies for autoantibodies, and antiplatelet drugs for microvascular disease.
When done correctly, he said, such “probe” studies can deepen mechanistic understandings, lead to biomarkers, and provide proof-of-concept that “will encourage massive investment in developing new therapies” for long COVID and other infection-associated chronic illnesses.
Trials of treatments for long COVID “are starting, so I’m optimistic,” said Dr. Deeks, an expert on HIV pathogenesis and treatment and a principal investigator of the Researching COVID to Enhance Recovery (RECOVER) study. Among the trials: A study of intravenous immunoglobulin (IVIG) for neurologic long COVID; a study of an anti-SARS-CoV-2 monoclonal antibody that can deplete tissue/cellular reservoirs of viral particles (replicating or not); and a study evaluating baricitinib (Olumiant), a Janus kinase inhibitor, for neurocognitive impairment and cardiopulmonary symptoms of long COVID.
Alessio Fasano, MD, professor of pediatrics at Harvard Medical School and professor of nutrition at the Harvard T.H. Chan School of Public Health, Boston, described at the workshop how he began investigating the use of larazotide acetate – an inhibitor of the protein zonulin, which increases intestinal permeability – in children with COVID-19 Multisystem Inflammatory Syndrome (MIS-C) after learning that SARS-CoV-2 viral particles persist in the gastrointestinal tract, causing dysbiosis and zonulin upregulation.
In an ongoing phase 2, double-blind, placebo-controlled trial, the agent thus far has expedited the resolution of gastrointestinal symptoms and clearance of spike protein from the circulation, he said. A phase 2 trial of the agent for pediatric patients with long COVID and SARS-CoV-2 antigenemia is underway. “What if we were to stop [chains of events] by stopping the passage of elements from the virus into circulation?’ he said.
In the realm of Lyme disease, a recently launched Clinical Trials Network for Lyme and Other Tick-Borne Diseases has awarded pilot study grants to evaluate treatments aimed at a variety of possible disease mechanisms that, notably, are similar to those of other chronic illnesses: persistence of infection or remnants of infection, immune dysregulation and autoimmune reactions, neural dysfunction, and gut microbiome changes. (Microclots and mitochondrial dysfunction have not been as well studied in Lyme.)
Current and upcoming studies include evaluations of transcutaneous auricular vagus nerve stimulation for those with persistent Lyme fatigue, transcranial direct current stimulation with cognitive retraining for Lyme brain fog, and tetracycline for PTLD, said Brian Fallon, MD, MPH, professor of clinical psychiatry at Columbia University, New York, who directs the Lyme & Tick-Borne Diseases Research Center and the coordinating center of the new network.
Moving forward, he said, it is important to loosen exclusion criteria and include patients with “probable or possible” Lyme and those with suspected infections with other tick-borne pathogens. All told, these patients comprise a large portion of those with chronic symptoms and have been neglected in an already thin research space, Dr. Fallon said, noting that “there haven’t been any clinical trials of posttreatment Lyme disease in ages – in 10-15 years.”
(PTLD refers to symptoms lasting for more than 6 months after the completion of standard Infectious Diseases Society of America–recommended antibiotic protocols. It occurs in about 15% of patients, said John Aucott, MD, director of the Johns Hopkins Lyme Disease Research Center, Baltimore, a member of the new clinical trials network.)
Calls for a new NIH center and patient involvement
Patients and patient advocacy organizations have played a vital role in research thus far: They’ve documented post-COVID symptoms that academic researchers said they would not otherwise have known of. Leaders of the Patient-Led Research Collaborative have coauthored published reviews with leading long COVID experts. And patients with tick-borne illnesses have enrolled in the MyLymeData patient registry run by LymeDisease.org, which has documented patient-experienced efficacy of alternative treatments and described antibiotic responders and nonresponders.
At the workshop, they shared findings alongside academic experts, and researchers called for their continued involvement. “Patient engagement at every step of the research process is critical,” Dr. Nath said.
“We need to ensure that research is reflective of lived experiences … and [that we’re] accelerating clinical trials of therapeutics that are of priority to the patient community,” said Lisa McCorkell, cofounder of the long COVID-focused Patient-Led Research Collaborative.
Ms. McCorkell also called for the creation of an office for infection-associated chronic illnesses in the NIH director’s office. Others voiced their support. “I think it’s a great idea to have an NIH center for infection-associated chronic illnesses,” said Dr. Fallon. “I think it would have a profound impact.”
The other great need, of course, is funding. “We have ideas, we have drugs that can be repurposed, we have a highly informed and engaged community that will enroll in and be retained in studies, and we have outcomes we can measure,” Dr. Deeks said. “What we’re missing is industry engagement and funding. We need massive engagement from the NIH.”
Real-world treatment needs
In the meantime, patients are seeking treatment, and “clinicians need to have uncertainty tolerance” and try multiple treatments simultaneously, said David Putrino, PT, PhD, director of rehabilitation innovation for the Mount Sinai Health System and professor of rehabilitation and human performance at the Icahn School of Medicine at Mount Sinai, New York. He oversees a multidisciplinary hybrid clinical care research center that has seen over 1,500 patients with long COVID and is beginning to see patients with other infection-associated chronic illnesses.
It’s a model that should be replicated to help fill the “enormous unmet clinical need” of patients with infection-associated chronic illness, said Peter Rowe, MD, professor of pediatrics at the Johns Hopkins School of Medicine and an expert on ME/CFS. And “as we request [more research funding], we will also need [financial] support for clinical care,” he emphasized, to provide equitable access for patients and to attract treating physicians.
Moreover, said Linda Geng, MD, PhD, the culture of stigma needs to change. Right now, patients with long COVID often feel dismissed not only by friends, families, and coworkers, but by clinicians who find it find it hard “to grasp that this is real and a biological condition.”
And it’s not just conditions such as long COVID that are stigmatized, but treatments as well, she said. For instance, some clinicians view low-dose naltrexone, a treatment increasingly being used for inflammation, with suspicion because it is used for opioid use disorder and alcohol use disorder – or because the “low-dose” label summons mistrust of homeopathy. “Even with therapies, there are preconceived notions and biases,” said Dr. Geng, cofounder and codirector of the Stanford (Calif.) Long COVID program.
“What almost killed me,” said Meghan O’Rourke, who has ongoing effects from long-undiagnosed tick-borne illness, “was the invisibility of the illness.” Ms. O’Rourke teaches at Yale University and is the author of “The Invisible Kingdom: Reimagining Chronic Illness.”
Teaching young physicians about these illnesses would help, she and others said. During a question and answer session, Dr. Putrino shared that the Icahn School of Medicine has recently committed to “create a complex chronic illness medical curriculum” that will impact medical education from the first year of medical school through residencies. Dr. Putrino said his team is also working on materials to help other clinics develop care models similar to those at his Mount Sinai clinic.
The NASEM workshop did not collect or require disclosures of its participants.
Experience with long COVID has shone a spotlight on persistent Lyme disease and other often debilitating chronic illnesses that follow known or suspected infections – and on the urgent need for a common and well-funded research agenda, education of physicians, growth of multidisciplinary clinics, and financially supported clinical care.
“We critically need to understand the epidemiology and pathogenesis of chronic symptoms, and identify more effective ways to manage, treat, and potentially cure these illnesses,” Lyle Petersen, MD, MPH, director of the division of vector-borne diseases at the Centers for Disease Control and Prevention, said at the start of a 2-day National Academies of Science, Engineering, and Medicine (NASEM) workshop, “Toward a Common Research Agenda in Infection-Associated Chronic Illnesses.”
Thinking about infection-associated chronic illnesses as an entity – one predicated on commonalities in chronic symptoms and in leading hypotheses for causes – represents a paradigm shift that researchers and patient advocates said can avoid research redundancies and is essential to address what the NASEM calls an overlooked, growing public health problem.
An estimated 2 million people in the United States are living with what’s called posttreatment Lyme disease (PTLD) – a subset of patients with persistent or chronic Lyme disease – and an estimated 1.7-3.3 million people in the United States have diagnoses of myalgic encephalomyelitis/chronic fatigue syndrome (ME/CFS). More than 700,000 people are living with multiple sclerosis. And as of January 2023, 11% of people in the United States reported having long COVID symptoms; the incidence of long COVID is currently estimated at 10%-30% of nonhospitalized cases of COVID-19.
These illnesses “have come under one umbrella,” said Avindra Nath, MD, clinical director of the National Institute of Neurologic Disorders and Stroke (NINDS), Bethesda, Md.
To date, common ground in the literature has grown largely around long COVID and ME/CFS, the latter of which is often associated with a prior, often unidentified infection.
Symptoms of both have been “rigorously” studied and shown to have overlaps, and the illnesses appear to share underlying biologic abnormalities in metabolism and the gut microbiome, as well as viral reactivation and abnormalities in the immune system, central and autonomic nervous systems, and the cardiovascular and pulmonary systems, said Anthony L. Komaroff, MD, professor of medicine at Harvard Medical School and a senior physician at Brigham & Women’s Hospital, both in Boston. (An estimated half of patients with long COVID meet the diagnostic criteria for ME/CFS.)
Although less thoroughly researched, similar symptoms are experienced by a subset of people following a variety of viral, bacterial, and protozoal infections, Dr. Komaroff said. To be determined, he said, is whether the pathophysiology believed to be shared by long COVID and ME/CFS is also shared with other postinfectious syndromes following acute illness with Ebola, West Nile, dengue, mycoplasma pneumonia, enteroviruses, and other pathogens, he said.
Persistent infection, viral reactivation
RNA viral infections can lead to persistent inflammation and dysregulated immunity, with or without viral persistence over time, Timothy J. Henrich, MD, MMSc, associate professor of medicine at the University of California, San Francisco, said in a keynote address.
Research on Ebola survivors has documented long-lasting inflammation and severe immune dysfunction 2 years after infection, for instance. And it’s well known that HIV-1 leads to aberrant immune responses, inflammation, and organ damage despite antiretroviral therapy, said Dr. Henrich, who leads a laboratory/research group that studies approaches to HIV-1 cure and PET-based imaging approaches to characterize viral reservoirs and immune sequelae.
Viral persistence, which can be difficult to measure, has also been documented in Ebola survivors. And in patients living with HIV-1, HIV-1 RNA and protein expression have been shown to persist, again despite antiretroviral therapy. The UCSF Long-Term Immunological Impact of Novel Coronavirus (LIINC) study, for which Dr. Henrich is the principal investigator, found spike RNA in colorectal tissue more than 22 months post COVID, and other research documented viral protein in gut tissue for up to 6 months, he said.
“I think we’re appreciating now, in at least the scientific and treatment community, that there’s a potential for ‘acute’ infections to exhibit some degree of persistence leading to clinical morbidity,” said Dr. Henrich, one of several speakers to describe reports of pathogen persistence. Regarding long COVID, its “etiology is likely heterogeneous,” he said, but persistence of SARS-CoV-2 “may lay behind” other described mechanisms, from clotting/microvascular dysfunction to inflammation and tissue damage to immune dysregulation.
Reactivation of existing latent viral infections in the setting of new acute microbial illness may also play an etiologic role in chronic illnesses, Dr. Henrich said. Epstein-Barr virus (EBV) reactivation has been shown in some studies, including their UCSF COVID-19 cohort, to be associated with long COVID.
“Physicians have been trained to be skeptical about the role [of latent viral infections],” Michael Peluso, MD, an infectious disease physician and assistant professor at UCSF, said during a talk on viral reactivation. This skepticism needs to be “reexamined and overcome,” he said.
Herpesviruses have frequently been associated with ME/CFS, he noted. And evidence of a strong association between EBV and multiple sclerosis came recently from a prospective study of 10 million military recruits that found a 32-fold increased risk of MS after EBV infection but no increase after infection with other viruses, Dr. Peluso and Dr. Henrich both noted.
Research needs, treatment trials
Research needs are vast: The need to learn more about the mechanisms of pathogen persistence and immune evasion, for instance, and the need for more biomarker studies, more imaging studies and tissue analyses, more study of microbiome composition and activity, and continued development and application of metagenomic next-generation sequencing.
Workshop participants also spoke of the need to better understand the molecular mimicry that can occur between pathogen-produced proteins and self-antigens, for instance, and the effects of inflammation and infection-related immune changes on neuronal and microglial function in the brain.
“We should perform similar forms of analysis [across] patients with different infection-associated chronic conditions,” said Amy Proal, PhD, president of the PolyBio Research Foundation, which funds research on infection-associated chronic infections. And within individual conditions and well-characterized study groups “we should perform many different forms of analysis … so we can define endotypes and get more solid biomarkers so that industry [will have more confidence] to run clinical trials.”
In the meantime, patients need fast-moving treatment trials for long COVID, long Lyme, and other infection-associated chronic illnesses, speakers emphasized. “We all agree that treatment trials are overdue,” said the NINDS’ Dr. Nath. “We can’t afford to wait for another decade until we understand all the mechanisms, but rather we can do clinical trials based on what we understand now and study the pathophysiology in the context of the clinical trials.”
Just as was done with HIV, said Steven G. Deeks, MD, professor of medicine at UCSF, researchers must “practice experimental medicine” and select pathways and mechanisms of interest, interrupt those pathways in a controlled manner, and assess impact. “Much of this can be done by repurposing existing drugs,” he said, like antivirals for persistent viral infection, EBV-directed therapies for EBV reactivation, anti-inflammatory drugs for inflammation, B–cell-directed therapies for autoantibodies, and antiplatelet drugs for microvascular disease.
When done correctly, he said, such “probe” studies can deepen mechanistic understandings, lead to biomarkers, and provide proof-of-concept that “will encourage massive investment in developing new therapies” for long COVID and other infection-associated chronic illnesses.
Trials of treatments for long COVID “are starting, so I’m optimistic,” said Dr. Deeks, an expert on HIV pathogenesis and treatment and a principal investigator of the Researching COVID to Enhance Recovery (RECOVER) study. Among the trials: A study of intravenous immunoglobulin (IVIG) for neurologic long COVID; a study of an anti-SARS-CoV-2 monoclonal antibody that can deplete tissue/cellular reservoirs of viral particles (replicating or not); and a study evaluating baricitinib (Olumiant), a Janus kinase inhibitor, for neurocognitive impairment and cardiopulmonary symptoms of long COVID.
Alessio Fasano, MD, professor of pediatrics at Harvard Medical School and professor of nutrition at the Harvard T.H. Chan School of Public Health, Boston, described at the workshop how he began investigating the use of larazotide acetate – an inhibitor of the protein zonulin, which increases intestinal permeability – in children with COVID-19 Multisystem Inflammatory Syndrome (MIS-C) after learning that SARS-CoV-2 viral particles persist in the gastrointestinal tract, causing dysbiosis and zonulin upregulation.
In an ongoing phase 2, double-blind, placebo-controlled trial, the agent thus far has expedited the resolution of gastrointestinal symptoms and clearance of spike protein from the circulation, he said. A phase 2 trial of the agent for pediatric patients with long COVID and SARS-CoV-2 antigenemia is underway. “What if we were to stop [chains of events] by stopping the passage of elements from the virus into circulation?’ he said.
In the realm of Lyme disease, a recently launched Clinical Trials Network for Lyme and Other Tick-Borne Diseases has awarded pilot study grants to evaluate treatments aimed at a variety of possible disease mechanisms that, notably, are similar to those of other chronic illnesses: persistence of infection or remnants of infection, immune dysregulation and autoimmune reactions, neural dysfunction, and gut microbiome changes. (Microclots and mitochondrial dysfunction have not been as well studied in Lyme.)
Current and upcoming studies include evaluations of transcutaneous auricular vagus nerve stimulation for those with persistent Lyme fatigue, transcranial direct current stimulation with cognitive retraining for Lyme brain fog, and tetracycline for PTLD, said Brian Fallon, MD, MPH, professor of clinical psychiatry at Columbia University, New York, who directs the Lyme & Tick-Borne Diseases Research Center and the coordinating center of the new network.
Moving forward, he said, it is important to loosen exclusion criteria and include patients with “probable or possible” Lyme and those with suspected infections with other tick-borne pathogens. All told, these patients comprise a large portion of those with chronic symptoms and have been neglected in an already thin research space, Dr. Fallon said, noting that “there haven’t been any clinical trials of posttreatment Lyme disease in ages – in 10-15 years.”
(PTLD refers to symptoms lasting for more than 6 months after the completion of standard Infectious Diseases Society of America–recommended antibiotic protocols. It occurs in about 15% of patients, said John Aucott, MD, director of the Johns Hopkins Lyme Disease Research Center, Baltimore, a member of the new clinical trials network.)
Calls for a new NIH center and patient involvement
Patients and patient advocacy organizations have played a vital role in research thus far: They’ve documented post-COVID symptoms that academic researchers said they would not otherwise have known of. Leaders of the Patient-Led Research Collaborative have coauthored published reviews with leading long COVID experts. And patients with tick-borne illnesses have enrolled in the MyLymeData patient registry run by LymeDisease.org, which has documented patient-experienced efficacy of alternative treatments and described antibiotic responders and nonresponders.
At the workshop, they shared findings alongside academic experts, and researchers called for their continued involvement. “Patient engagement at every step of the research process is critical,” Dr. Nath said.
“We need to ensure that research is reflective of lived experiences … and [that we’re] accelerating clinical trials of therapeutics that are of priority to the patient community,” said Lisa McCorkell, cofounder of the long COVID-focused Patient-Led Research Collaborative.
Ms. McCorkell also called for the creation of an office for infection-associated chronic illnesses in the NIH director’s office. Others voiced their support. “I think it’s a great idea to have an NIH center for infection-associated chronic illnesses,” said Dr. Fallon. “I think it would have a profound impact.”
The other great need, of course, is funding. “We have ideas, we have drugs that can be repurposed, we have a highly informed and engaged community that will enroll in and be retained in studies, and we have outcomes we can measure,” Dr. Deeks said. “What we’re missing is industry engagement and funding. We need massive engagement from the NIH.”
Real-world treatment needs
In the meantime, patients are seeking treatment, and “clinicians need to have uncertainty tolerance” and try multiple treatments simultaneously, said David Putrino, PT, PhD, director of rehabilitation innovation for the Mount Sinai Health System and professor of rehabilitation and human performance at the Icahn School of Medicine at Mount Sinai, New York. He oversees a multidisciplinary hybrid clinical care research center that has seen over 1,500 patients with long COVID and is beginning to see patients with other infection-associated chronic illnesses.
It’s a model that should be replicated to help fill the “enormous unmet clinical need” of patients with infection-associated chronic illness, said Peter Rowe, MD, professor of pediatrics at the Johns Hopkins School of Medicine and an expert on ME/CFS. And “as we request [more research funding], we will also need [financial] support for clinical care,” he emphasized, to provide equitable access for patients and to attract treating physicians.
Moreover, said Linda Geng, MD, PhD, the culture of stigma needs to change. Right now, patients with long COVID often feel dismissed not only by friends, families, and coworkers, but by clinicians who find it find it hard “to grasp that this is real and a biological condition.”
And it’s not just conditions such as long COVID that are stigmatized, but treatments as well, she said. For instance, some clinicians view low-dose naltrexone, a treatment increasingly being used for inflammation, with suspicion because it is used for opioid use disorder and alcohol use disorder – or because the “low-dose” label summons mistrust of homeopathy. “Even with therapies, there are preconceived notions and biases,” said Dr. Geng, cofounder and codirector of the Stanford (Calif.) Long COVID program.
“What almost killed me,” said Meghan O’Rourke, who has ongoing effects from long-undiagnosed tick-borne illness, “was the invisibility of the illness.” Ms. O’Rourke teaches at Yale University and is the author of “The Invisible Kingdom: Reimagining Chronic Illness.”
Teaching young physicians about these illnesses would help, she and others said. During a question and answer session, Dr. Putrino shared that the Icahn School of Medicine has recently committed to “create a complex chronic illness medical curriculum” that will impact medical education from the first year of medical school through residencies. Dr. Putrino said his team is also working on materials to help other clinics develop care models similar to those at his Mount Sinai clinic.
The NASEM workshop did not collect or require disclosures of its participants.
FROM A NATIONAL ACADEMIES OF SCIENCE, ENGINEERING, AND MEDICINE WORKSHOP
Nurse practitioners sue state over right to use ‘doctor’ title
, saying it violates their first amendment right to use the honorific title without fear of regulatory repercussions.
The case highlights ongoing scope-creep battles as the American Medical Association tries to preserve the physician-led team model and nursing organizations and some lawmakers push for greater autonomy for allied professionals.
In the complaint filed in district court in June, plaintiffs Jacqueline Palmer, DNP, Heather Lewis, DNP, and Rodolfo Jaravata-Hanson, DNP, say they fear the state will sanction them. They note that “Doctor Sarah,” another DNP, was fined nearly $20,000 by the state last November for false advertising and fraud after using the moniker in her online advertising and social media accounts.
The fine was part of a settlement that the DNP, Sarah Erny, reached with the state to resolve allegations that she failed to identify her supervising physician and inform the public that she was not a medical doctor.
Under California’s Medical Practice Act, individuals cannot refer to themselves as “doctor, physician, or any other terms or letters indicating or implying that he or she is a physician and surgeon ... without having ... a certificate as a physician and surgeon.”
Instead, nurse practitioners certified by the California Board of Registered Nursing may use titles like “Certified Nurse Practitioner” and “Advanced Practice Registered Nurse,” corresponding letters such as APRN-CNP, RN, and NP, and phrases like pediatric nurse practitioner to identify specialization.
Individuals who misrepresent themselves are subject to misdemeanor charges and civil penalties.
The nonprofit Pacific Legal Foundation represents the plaintiffs. In court records, its attorneys argue that after “years earning their advanced degrees and qualifications ... they should be able to speak truthfully about them in their workplaces, on their business cards, the Internet, and social media, so long as they clarify that they are nurse practitioners.”
State lawmakers’ attempts to clarify the roles of physicians and nurse practitioners have seen mixed results. Florida legislators recently passed a bill to prevent advanced practice nurses from using the honorific title, reserving it only for MDs and DOs. Gov. Ron DeSantis vetoed it last month.
In May, Georgia lawmakers passed the Health Care Practitioners Truth and Transparency Act. It requires advanced practice nurses and physician assistants with doctoral degrees who refer to themselves as doctors in a clinical setting to state they are not medical doctors or physicians.
Still, some health professionals say that the designation should only be used in academic settings or among peers, and that all doctoral degree holders should ditch the moniker at the bedside to ease patient communications.
Named as defendants in the suit are three state officials: California Attorney General Rob Bonta, state Medical Board President Kristina Lawson, and California Board of Registered Nursing Executive Officer Loretta Melby.
A version of this article first appeared on Medscape.com.
, saying it violates their first amendment right to use the honorific title without fear of regulatory repercussions.
The case highlights ongoing scope-creep battles as the American Medical Association tries to preserve the physician-led team model and nursing organizations and some lawmakers push for greater autonomy for allied professionals.
In the complaint filed in district court in June, plaintiffs Jacqueline Palmer, DNP, Heather Lewis, DNP, and Rodolfo Jaravata-Hanson, DNP, say they fear the state will sanction them. They note that “Doctor Sarah,” another DNP, was fined nearly $20,000 by the state last November for false advertising and fraud after using the moniker in her online advertising and social media accounts.
The fine was part of a settlement that the DNP, Sarah Erny, reached with the state to resolve allegations that she failed to identify her supervising physician and inform the public that she was not a medical doctor.
Under California’s Medical Practice Act, individuals cannot refer to themselves as “doctor, physician, or any other terms or letters indicating or implying that he or she is a physician and surgeon ... without having ... a certificate as a physician and surgeon.”
Instead, nurse practitioners certified by the California Board of Registered Nursing may use titles like “Certified Nurse Practitioner” and “Advanced Practice Registered Nurse,” corresponding letters such as APRN-CNP, RN, and NP, and phrases like pediatric nurse practitioner to identify specialization.
Individuals who misrepresent themselves are subject to misdemeanor charges and civil penalties.
The nonprofit Pacific Legal Foundation represents the plaintiffs. In court records, its attorneys argue that after “years earning their advanced degrees and qualifications ... they should be able to speak truthfully about them in their workplaces, on their business cards, the Internet, and social media, so long as they clarify that they are nurse practitioners.”
State lawmakers’ attempts to clarify the roles of physicians and nurse practitioners have seen mixed results. Florida legislators recently passed a bill to prevent advanced practice nurses from using the honorific title, reserving it only for MDs and DOs. Gov. Ron DeSantis vetoed it last month.
In May, Georgia lawmakers passed the Health Care Practitioners Truth and Transparency Act. It requires advanced practice nurses and physician assistants with doctoral degrees who refer to themselves as doctors in a clinical setting to state they are not medical doctors or physicians.
Still, some health professionals say that the designation should only be used in academic settings or among peers, and that all doctoral degree holders should ditch the moniker at the bedside to ease patient communications.
Named as defendants in the suit are three state officials: California Attorney General Rob Bonta, state Medical Board President Kristina Lawson, and California Board of Registered Nursing Executive Officer Loretta Melby.
A version of this article first appeared on Medscape.com.
, saying it violates their first amendment right to use the honorific title without fear of regulatory repercussions.
The case highlights ongoing scope-creep battles as the American Medical Association tries to preserve the physician-led team model and nursing organizations and some lawmakers push for greater autonomy for allied professionals.
In the complaint filed in district court in June, plaintiffs Jacqueline Palmer, DNP, Heather Lewis, DNP, and Rodolfo Jaravata-Hanson, DNP, say they fear the state will sanction them. They note that “Doctor Sarah,” another DNP, was fined nearly $20,000 by the state last November for false advertising and fraud after using the moniker in her online advertising and social media accounts.
The fine was part of a settlement that the DNP, Sarah Erny, reached with the state to resolve allegations that she failed to identify her supervising physician and inform the public that she was not a medical doctor.
Under California’s Medical Practice Act, individuals cannot refer to themselves as “doctor, physician, or any other terms or letters indicating or implying that he or she is a physician and surgeon ... without having ... a certificate as a physician and surgeon.”
Instead, nurse practitioners certified by the California Board of Registered Nursing may use titles like “Certified Nurse Practitioner” and “Advanced Practice Registered Nurse,” corresponding letters such as APRN-CNP, RN, and NP, and phrases like pediatric nurse practitioner to identify specialization.
Individuals who misrepresent themselves are subject to misdemeanor charges and civil penalties.
The nonprofit Pacific Legal Foundation represents the plaintiffs. In court records, its attorneys argue that after “years earning their advanced degrees and qualifications ... they should be able to speak truthfully about them in their workplaces, on their business cards, the Internet, and social media, so long as they clarify that they are nurse practitioners.”
State lawmakers’ attempts to clarify the roles of physicians and nurse practitioners have seen mixed results. Florida legislators recently passed a bill to prevent advanced practice nurses from using the honorific title, reserving it only for MDs and DOs. Gov. Ron DeSantis vetoed it last month.
In May, Georgia lawmakers passed the Health Care Practitioners Truth and Transparency Act. It requires advanced practice nurses and physician assistants with doctoral degrees who refer to themselves as doctors in a clinical setting to state they are not medical doctors or physicians.
Still, some health professionals say that the designation should only be used in academic settings or among peers, and that all doctoral degree holders should ditch the moniker at the bedside to ease patient communications.
Named as defendants in the suit are three state officials: California Attorney General Rob Bonta, state Medical Board President Kristina Lawson, and California Board of Registered Nursing Executive Officer Loretta Melby.
A version of this article first appeared on Medscape.com.
Research points toward combination therapy for Lyme and improved diagnostics
Several recent developments in Lyme disease treatment and diagnosis may pave the way forward for combating disease that persists following missed or delayed diagnoses or remains following standard treatment. These include combination therapy to address “persister” bacteria and diagnostic tests that test directly for the pathogen and/or indirectly test for host response, according to experts who presented at a 2-day National Academies of Science, Engineering and Medicine workshop on infection-associated chronic illnesses.
Research has shown that 60% of people who are infected and not treated during the early or early disseminated stages of Lyme disease go on to develop late Lyme arthritis, said John Aucott, MD, director of the Johns Hopkins Lyme Disease Clinical Research Center in Baltimore. And in the real world, there’s an additional category of patients: Those who are misdiagnosed and develop infection-related persistent symptoms – such as fatigue, brain fog/cognitive dysfunction, and musculoskeletal problems – that don’t match the “textbook schematic” involving late Lyme arthritis and late neurologic disease.
Moreover, of patients who are treated with protocols recommended by the Infectious Diseases Society of America (IDSA), about 15% go on to develop persistent symptoms at 6 months – again, symptoms that don’t match textbook manifestations and do match symptoms of other infection-associated chronic illnesses. As a “research construct,” this has been coined posttreatment Lyme disease (PTLD), he said at the workshop, “Toward a Common Research Agenda in Infection-Associated Chronic Illnesses.”
(On a practical level, it is hard to know clinically who has early disseminated disease unless they have multiple erythema migrans rashes or neurologic or cardiac involvement, he said after the meeting.)
All this points to the need for tests that are sensitive and specific for diagnosis at all stages of infection and disease, he said in a talk on diagnostics. Currently available tests – those that fit into the widely used two-tiered enzyme-linked immunosorbent assay, Western Blot serology testing – have significant limitations in sensitivity and specificity, including for acute infection when the body has not generated enough antibodies, yet treatment is most likely to succeed.
Move toward combination therapy research
Lyme disease is most commonly treated with doxycycline, and that’s problematic because the antibiotic is a microstatic whose efficacy relies on immune clearance of static bacteria, said Monica E. Embers, PhD, director of vector-borne disease at the Tulane National Primate Research Center and associate professor of immunology at Tulane University, New Orleans.
“But we know that Borrelia burgdorferi has the capability to evade the host immune response in almost every way possible. Persistence is the norm in an immunocompetent host ... [and] dormant bacteria/persisters are more tolerant of microstatic antibiotics,” she said.
Other considerations for antibiotic efficacy include the fact that B. burgdorferi survives for many months inside ticks without nutrient replenishment or replication, “so dormancy is part of their life cycle,” she said. Moreover, the bacteria can be found deep in connective tissues and joints.
The efficacy of accepted regimens of antibiotic treatment has been “a very contentious issue,” she said, noting that guidelines from the International Lyme and Associated Diseases Society “leave open the possibility for antibiotic retreatment when a chronic infection is judged to be a possible cause [of ongoing symptoms].”
The development of persister B. burgdorferi in the presence of antibiotics has been well studied in vitro, which has limitations, Dr. Embers said. But her group specializes in animal models and has shown persistence of antimicrobial-tolerant B. burgdorferi in tick-inoculated rhesus macaques 8-9 months after treatment with oral doxycycline.
“We [also] saw persistence of mild-moderate inflammation in the brain, peripheral nerves, spinal cord, joints and skeletal muscle, and in the heart,” Dr. Embers said, who coauthored a 2022 review of B. burgdorferi antimicrobial-tolerant persistence in Lyme disease and PTLD.
Her work has also shown that ceftriaxone, which is recommended by IDSA for patients with clinically evident neurological and/or cardiac involvement, does not clear infection in mice. “In general, single drugs have not been capable of clearing the infection, yet combinations show promise,” she said.
Dr. Embers has combed large drug libraries looking for combinations of antibiotics that employ different mechanisms of action in hopes of eliminating persister spirochetes. Certain combinations have shown promise in mice and have been tested in her rhesus macaque model; data analyses are underway.
Other research teams, such as that of Ying Zhang, MD, PhD, at Johns Hopkins, have similarly been screening combinations of antibiotics and other compounds, identifying candidates for further testing.
During a question and answer period, Dr. Embers said her team is also investigating the pathophysiology and long-term effects of tick-borne coinfections, including Bartonella, and is pursuing a hypothesis that infection with Borrelia allows Bartonella to cause more extensive disease and persist longer. “I think Lyme is at the core because of its ability to evade and suppress the immune response so effectively.”
Diagnostic possibilities, biomarkers for PTLD
Direct diagnostic tests for microbial nucleic acid and proteins “are promising alternatives for indirect serologic tests,” Dr. Aucott said. For instance, in addition to polymerase chain reaction tests, which “are making advances,” it may be possible to target the B. burgdorferi peptidoglycan for antigen detection.
Researchers have shown that peptidoglycan, a component of the B. burgdorferi cell envelope, is a persistent antigen in the synovial fluid of patients with Lyme arthritis who have been treated with oral and intravenous antibiotics, and that it likely contributes to inflammation.
“Maybe the infection is gone but parts of the bacteria are still there that are driving inflammation,” said Dr. Aucott, also associate professor of medicine at John Hopkins.
Researchers have also been looking at the host response to B. burgdorferi – including cytokines, chemokines, and autoantibiodies – to identify biomarkers for PTLD and to identify patients during posttreatment follow-up who are at increased risk of developing PTLD, with the hope of someday intervening. Persistently high levels of interleukin-23, CCL19, and interferon-alpha have each been associated in different studies with persistent symptoms after treatment, Dr. Aucott said.
In addition, metabolomics research is showing that patients with PTLD have metabolic fingerprints that are different from those who return to good health after treatment, and it may be possible to identify an epigenetic signature for Lyme disease. A project sponsored by the Defense Advanced Research Projects Agency called ECHO (Epigenetic Characterization and Observation) aims to identify epigenetic signatures of exposures to various threats, including B. burgdorferi.
“At the very proximal end of [indirectly testing for host response], there are modifications of the DNA that can occur in response to infectious insults ... and that changed DNA changes RNA expression and protein synthesis,” Dr. Aucott explained. DARPA’s project is “exciting because their goal [at DARPA] is to have a diagnostic test quickly as a result of this epigenetics work.”
Imaging research is also fast offering diagnostic opportunities, Dr. Aucott said. Levels of microglial activation on brain PET imaging have been found to correlate with PTLD, and a study at Johns Hopkins of multimodal neuroimaging with functional MRI and diffusion tensor imaging has shown distinct changes to white matter activation within the frontal lobe of patients with PTLD, compared with controls.
The NASEM workshop did not collect or require disclosures of its participants.
Several recent developments in Lyme disease treatment and diagnosis may pave the way forward for combating disease that persists following missed or delayed diagnoses or remains following standard treatment. These include combination therapy to address “persister” bacteria and diagnostic tests that test directly for the pathogen and/or indirectly test for host response, according to experts who presented at a 2-day National Academies of Science, Engineering and Medicine workshop on infection-associated chronic illnesses.
Research has shown that 60% of people who are infected and not treated during the early or early disseminated stages of Lyme disease go on to develop late Lyme arthritis, said John Aucott, MD, director of the Johns Hopkins Lyme Disease Clinical Research Center in Baltimore. And in the real world, there’s an additional category of patients: Those who are misdiagnosed and develop infection-related persistent symptoms – such as fatigue, brain fog/cognitive dysfunction, and musculoskeletal problems – that don’t match the “textbook schematic” involving late Lyme arthritis and late neurologic disease.
Moreover, of patients who are treated with protocols recommended by the Infectious Diseases Society of America (IDSA), about 15% go on to develop persistent symptoms at 6 months – again, symptoms that don’t match textbook manifestations and do match symptoms of other infection-associated chronic illnesses. As a “research construct,” this has been coined posttreatment Lyme disease (PTLD), he said at the workshop, “Toward a Common Research Agenda in Infection-Associated Chronic Illnesses.”
(On a practical level, it is hard to know clinically who has early disseminated disease unless they have multiple erythema migrans rashes or neurologic or cardiac involvement, he said after the meeting.)
All this points to the need for tests that are sensitive and specific for diagnosis at all stages of infection and disease, he said in a talk on diagnostics. Currently available tests – those that fit into the widely used two-tiered enzyme-linked immunosorbent assay, Western Blot serology testing – have significant limitations in sensitivity and specificity, including for acute infection when the body has not generated enough antibodies, yet treatment is most likely to succeed.
Move toward combination therapy research
Lyme disease is most commonly treated with doxycycline, and that’s problematic because the antibiotic is a microstatic whose efficacy relies on immune clearance of static bacteria, said Monica E. Embers, PhD, director of vector-borne disease at the Tulane National Primate Research Center and associate professor of immunology at Tulane University, New Orleans.
“But we know that Borrelia burgdorferi has the capability to evade the host immune response in almost every way possible. Persistence is the norm in an immunocompetent host ... [and] dormant bacteria/persisters are more tolerant of microstatic antibiotics,” she said.
Other considerations for antibiotic efficacy include the fact that B. burgdorferi survives for many months inside ticks without nutrient replenishment or replication, “so dormancy is part of their life cycle,” she said. Moreover, the bacteria can be found deep in connective tissues and joints.
The efficacy of accepted regimens of antibiotic treatment has been “a very contentious issue,” she said, noting that guidelines from the International Lyme and Associated Diseases Society “leave open the possibility for antibiotic retreatment when a chronic infection is judged to be a possible cause [of ongoing symptoms].”
The development of persister B. burgdorferi in the presence of antibiotics has been well studied in vitro, which has limitations, Dr. Embers said. But her group specializes in animal models and has shown persistence of antimicrobial-tolerant B. burgdorferi in tick-inoculated rhesus macaques 8-9 months after treatment with oral doxycycline.
“We [also] saw persistence of mild-moderate inflammation in the brain, peripheral nerves, spinal cord, joints and skeletal muscle, and in the heart,” Dr. Embers said, who coauthored a 2022 review of B. burgdorferi antimicrobial-tolerant persistence in Lyme disease and PTLD.
Her work has also shown that ceftriaxone, which is recommended by IDSA for patients with clinically evident neurological and/or cardiac involvement, does not clear infection in mice. “In general, single drugs have not been capable of clearing the infection, yet combinations show promise,” she said.
Dr. Embers has combed large drug libraries looking for combinations of antibiotics that employ different mechanisms of action in hopes of eliminating persister spirochetes. Certain combinations have shown promise in mice and have been tested in her rhesus macaque model; data analyses are underway.
Other research teams, such as that of Ying Zhang, MD, PhD, at Johns Hopkins, have similarly been screening combinations of antibiotics and other compounds, identifying candidates for further testing.
During a question and answer period, Dr. Embers said her team is also investigating the pathophysiology and long-term effects of tick-borne coinfections, including Bartonella, and is pursuing a hypothesis that infection with Borrelia allows Bartonella to cause more extensive disease and persist longer. “I think Lyme is at the core because of its ability to evade and suppress the immune response so effectively.”
Diagnostic possibilities, biomarkers for PTLD
Direct diagnostic tests for microbial nucleic acid and proteins “are promising alternatives for indirect serologic tests,” Dr. Aucott said. For instance, in addition to polymerase chain reaction tests, which “are making advances,” it may be possible to target the B. burgdorferi peptidoglycan for antigen detection.
Researchers have shown that peptidoglycan, a component of the B. burgdorferi cell envelope, is a persistent antigen in the synovial fluid of patients with Lyme arthritis who have been treated with oral and intravenous antibiotics, and that it likely contributes to inflammation.
“Maybe the infection is gone but parts of the bacteria are still there that are driving inflammation,” said Dr. Aucott, also associate professor of medicine at John Hopkins.
Researchers have also been looking at the host response to B. burgdorferi – including cytokines, chemokines, and autoantibiodies – to identify biomarkers for PTLD and to identify patients during posttreatment follow-up who are at increased risk of developing PTLD, with the hope of someday intervening. Persistently high levels of interleukin-23, CCL19, and interferon-alpha have each been associated in different studies with persistent symptoms after treatment, Dr. Aucott said.
In addition, metabolomics research is showing that patients with PTLD have metabolic fingerprints that are different from those who return to good health after treatment, and it may be possible to identify an epigenetic signature for Lyme disease. A project sponsored by the Defense Advanced Research Projects Agency called ECHO (Epigenetic Characterization and Observation) aims to identify epigenetic signatures of exposures to various threats, including B. burgdorferi.
“At the very proximal end of [indirectly testing for host response], there are modifications of the DNA that can occur in response to infectious insults ... and that changed DNA changes RNA expression and protein synthesis,” Dr. Aucott explained. DARPA’s project is “exciting because their goal [at DARPA] is to have a diagnostic test quickly as a result of this epigenetics work.”
Imaging research is also fast offering diagnostic opportunities, Dr. Aucott said. Levels of microglial activation on brain PET imaging have been found to correlate with PTLD, and a study at Johns Hopkins of multimodal neuroimaging with functional MRI and diffusion tensor imaging has shown distinct changes to white matter activation within the frontal lobe of patients with PTLD, compared with controls.
The NASEM workshop did not collect or require disclosures of its participants.
Several recent developments in Lyme disease treatment and diagnosis may pave the way forward for combating disease that persists following missed or delayed diagnoses or remains following standard treatment. These include combination therapy to address “persister” bacteria and diagnostic tests that test directly for the pathogen and/or indirectly test for host response, according to experts who presented at a 2-day National Academies of Science, Engineering and Medicine workshop on infection-associated chronic illnesses.
Research has shown that 60% of people who are infected and not treated during the early or early disseminated stages of Lyme disease go on to develop late Lyme arthritis, said John Aucott, MD, director of the Johns Hopkins Lyme Disease Clinical Research Center in Baltimore. And in the real world, there’s an additional category of patients: Those who are misdiagnosed and develop infection-related persistent symptoms – such as fatigue, brain fog/cognitive dysfunction, and musculoskeletal problems – that don’t match the “textbook schematic” involving late Lyme arthritis and late neurologic disease.
Moreover, of patients who are treated with protocols recommended by the Infectious Diseases Society of America (IDSA), about 15% go on to develop persistent symptoms at 6 months – again, symptoms that don’t match textbook manifestations and do match symptoms of other infection-associated chronic illnesses. As a “research construct,” this has been coined posttreatment Lyme disease (PTLD), he said at the workshop, “Toward a Common Research Agenda in Infection-Associated Chronic Illnesses.”
(On a practical level, it is hard to know clinically who has early disseminated disease unless they have multiple erythema migrans rashes or neurologic or cardiac involvement, he said after the meeting.)
All this points to the need for tests that are sensitive and specific for diagnosis at all stages of infection and disease, he said in a talk on diagnostics. Currently available tests – those that fit into the widely used two-tiered enzyme-linked immunosorbent assay, Western Blot serology testing – have significant limitations in sensitivity and specificity, including for acute infection when the body has not generated enough antibodies, yet treatment is most likely to succeed.
Move toward combination therapy research
Lyme disease is most commonly treated with doxycycline, and that’s problematic because the antibiotic is a microstatic whose efficacy relies on immune clearance of static bacteria, said Monica E. Embers, PhD, director of vector-borne disease at the Tulane National Primate Research Center and associate professor of immunology at Tulane University, New Orleans.
“But we know that Borrelia burgdorferi has the capability to evade the host immune response in almost every way possible. Persistence is the norm in an immunocompetent host ... [and] dormant bacteria/persisters are more tolerant of microstatic antibiotics,” she said.
Other considerations for antibiotic efficacy include the fact that B. burgdorferi survives for many months inside ticks without nutrient replenishment or replication, “so dormancy is part of their life cycle,” she said. Moreover, the bacteria can be found deep in connective tissues and joints.
The efficacy of accepted regimens of antibiotic treatment has been “a very contentious issue,” she said, noting that guidelines from the International Lyme and Associated Diseases Society “leave open the possibility for antibiotic retreatment when a chronic infection is judged to be a possible cause [of ongoing symptoms].”
The development of persister B. burgdorferi in the presence of antibiotics has been well studied in vitro, which has limitations, Dr. Embers said. But her group specializes in animal models and has shown persistence of antimicrobial-tolerant B. burgdorferi in tick-inoculated rhesus macaques 8-9 months after treatment with oral doxycycline.
“We [also] saw persistence of mild-moderate inflammation in the brain, peripheral nerves, spinal cord, joints and skeletal muscle, and in the heart,” Dr. Embers said, who coauthored a 2022 review of B. burgdorferi antimicrobial-tolerant persistence in Lyme disease and PTLD.
Her work has also shown that ceftriaxone, which is recommended by IDSA for patients with clinically evident neurological and/or cardiac involvement, does not clear infection in mice. “In general, single drugs have not been capable of clearing the infection, yet combinations show promise,” she said.
Dr. Embers has combed large drug libraries looking for combinations of antibiotics that employ different mechanisms of action in hopes of eliminating persister spirochetes. Certain combinations have shown promise in mice and have been tested in her rhesus macaque model; data analyses are underway.
Other research teams, such as that of Ying Zhang, MD, PhD, at Johns Hopkins, have similarly been screening combinations of antibiotics and other compounds, identifying candidates for further testing.
During a question and answer period, Dr. Embers said her team is also investigating the pathophysiology and long-term effects of tick-borne coinfections, including Bartonella, and is pursuing a hypothesis that infection with Borrelia allows Bartonella to cause more extensive disease and persist longer. “I think Lyme is at the core because of its ability to evade and suppress the immune response so effectively.”
Diagnostic possibilities, biomarkers for PTLD
Direct diagnostic tests for microbial nucleic acid and proteins “are promising alternatives for indirect serologic tests,” Dr. Aucott said. For instance, in addition to polymerase chain reaction tests, which “are making advances,” it may be possible to target the B. burgdorferi peptidoglycan for antigen detection.
Researchers have shown that peptidoglycan, a component of the B. burgdorferi cell envelope, is a persistent antigen in the synovial fluid of patients with Lyme arthritis who have been treated with oral and intravenous antibiotics, and that it likely contributes to inflammation.
“Maybe the infection is gone but parts of the bacteria are still there that are driving inflammation,” said Dr. Aucott, also associate professor of medicine at John Hopkins.
Researchers have also been looking at the host response to B. burgdorferi – including cytokines, chemokines, and autoantibiodies – to identify biomarkers for PTLD and to identify patients during posttreatment follow-up who are at increased risk of developing PTLD, with the hope of someday intervening. Persistently high levels of interleukin-23, CCL19, and interferon-alpha have each been associated in different studies with persistent symptoms after treatment, Dr. Aucott said.
In addition, metabolomics research is showing that patients with PTLD have metabolic fingerprints that are different from those who return to good health after treatment, and it may be possible to identify an epigenetic signature for Lyme disease. A project sponsored by the Defense Advanced Research Projects Agency called ECHO (Epigenetic Characterization and Observation) aims to identify epigenetic signatures of exposures to various threats, including B. burgdorferi.
“At the very proximal end of [indirectly testing for host response], there are modifications of the DNA that can occur in response to infectious insults ... and that changed DNA changes RNA expression and protein synthesis,” Dr. Aucott explained. DARPA’s project is “exciting because their goal [at DARPA] is to have a diagnostic test quickly as a result of this epigenetics work.”
Imaging research is also fast offering diagnostic opportunities, Dr. Aucott said. Levels of microglial activation on brain PET imaging have been found to correlate with PTLD, and a study at Johns Hopkins of multimodal neuroimaging with functional MRI and diffusion tensor imaging has shown distinct changes to white matter activation within the frontal lobe of patients with PTLD, compared with controls.
The NASEM workshop did not collect or require disclosures of its participants.
FROM A NATIONAL ACADEMIES OF SCIENCE, ENGINEERING AND MEDICINE WORKSHOP
Functional MRI shows that empathetic remarks reduce pain
These are the results of a study, recently published in the Proceedings of the National Academy of Sciences, that was conducted by a team led by neuroscientist Dan-Mikael Ellingsen, PhD, from Oslo University Hospital.
The researchers used functional MRI to scan the brains of 20 patients with chronic pain to investigate how a physician’s demeanor may affect patients’ sensitivity to pain, including effects in the central nervous system. During the scans, which were conducted in two sessions, the patients’ legs were exposed to stimuli that ranged from painless to moderately painful. The patients recorded perceived pain intensity using a scale. The physicians also underwent fMRI.
Half of the patients were subjected to the pain stimuli while alone; the other half were subjected to pain while in the presence of a physician. The latter group of patients was divided into two subgroups. Half of the patients had spoken to the accompanying physician before the examination. They discussed the history of the patient’s condition to date, among other things. The other half underwent the brain scans without any prior interaction with a physician.
Worse when alone
Dr. Ellingsen and his colleagues found that patients who were alone during the examination reported greater pain than those who were in the presence of a physician, even though they were subjected to stimuli of the same intensity. In instances in which the physician and patient had already spoken before the brain scan, patients additionally felt that the physician was empathetic and understood their pain. Furthermore, the physicians were better able to estimate the pain that their patients experienced.
The patients who had a physician by their side consistently experienced pain that was milder than the pain experienced by those who were alone. For pairs that had spoken beforehand, the patients considered their physician to be better able to understand their pain, and the physicians estimated the perceived pain intensity of their patients more accurately.
Evidence of trust
There was greater activity in the dorsolateral and ventrolateral prefrontal cortex, as well as in the primary and secondary somatosensory areas, in patients in the subgroup that had spoken to a physician. For the physicians, compared with the comparison group, there was an increase in correspondence between activity in the dorsolateral prefrontal cortex and activity in the secondary somatosensory areas of patients, which is a brain region that is known to react to pain. The brain activity correlation increased in line with the self-reported mutual trust between the physician and patient.
“These results prove that empathy and support can decrease pain intensity,” the investigators write. The data shed light on the brain processes behind the social modulation of pain during the interaction between the physician and the patient. Concordances in the brain are increased by greater therapeutic alliance.
Beyond medication
Winfried Meissner, MD, head of the pain clinic at the department of anesthesiology and intensive care medicine at Jena University Hospital, Germany, and former president of the German Pain Society, said in an interview: “I view this as a vital study that impressively demonstrates that effective, intensive pain therapy is not just a case of administering the correct analgesic.”
“Instead, a focus should be placed on what common sense tells us, which is just how crucial an empathetic attitude from physicians and good communication with patients are when it comes to the success of any therapy,” Dr. Meissner added. Unfortunately, such an attitude and such communication often are not provided in clinical practice because of limitations on time.
“Now, with objectively collected data from patients and physicians, [Dr.] Ellingsen’s team has been able to demonstrate that human interaction has a decisive impact on the treatment of patients experiencing pain,” said Dr. Meissner. “The study should encourage practitioners to treat communication just as seriously as the pharmacology of analgesics.”
Perception and attitude
“The study shows remarkably well that empathetic conversation between the physician and patient represents a valuable therapeutic method and should be recognized as such,” emphasized Dr. Meissner. Of course, conversation cannot replace pharmacologic treatment, but it can supplement and reinforce it. Furthermore, a physician’s empathy presumably has an effect that is at least as great as a suitable analgesic.
“Pain is more than just sensory perception,” explained Dr. Meissner. “We all know that it has a strong affective component, and perception is greatly determined by context.” This can be seen, for example, in athletes, who often attribute less importance to their pain and can successfully perform competitively despite a painful injury.
Positive expectations
Dr. Meissner advised all physicians to treat patients with pain empathetically. He encourages them to ask patients about their pain, accompanying symptoms, possible fears, and other mental stress and to take these factors seriously.
Moreover, the findings accentuate the effect of prescribed analgesics. “Numerous studies have meanwhile shown that the more positive a patient’s expectations, the better the effect of a medication,” said Dr. Meissner. “We physicians must exploit this effect, too.”
This article was translated from the Medscape German Edition and a version appeared on Medscape.com.
These are the results of a study, recently published in the Proceedings of the National Academy of Sciences, that was conducted by a team led by neuroscientist Dan-Mikael Ellingsen, PhD, from Oslo University Hospital.
The researchers used functional MRI to scan the brains of 20 patients with chronic pain to investigate how a physician’s demeanor may affect patients’ sensitivity to pain, including effects in the central nervous system. During the scans, which were conducted in two sessions, the patients’ legs were exposed to stimuli that ranged from painless to moderately painful. The patients recorded perceived pain intensity using a scale. The physicians also underwent fMRI.
Half of the patients were subjected to the pain stimuli while alone; the other half were subjected to pain while in the presence of a physician. The latter group of patients was divided into two subgroups. Half of the patients had spoken to the accompanying physician before the examination. They discussed the history of the patient’s condition to date, among other things. The other half underwent the brain scans without any prior interaction with a physician.
Worse when alone
Dr. Ellingsen and his colleagues found that patients who were alone during the examination reported greater pain than those who were in the presence of a physician, even though they were subjected to stimuli of the same intensity. In instances in which the physician and patient had already spoken before the brain scan, patients additionally felt that the physician was empathetic and understood their pain. Furthermore, the physicians were better able to estimate the pain that their patients experienced.
The patients who had a physician by their side consistently experienced pain that was milder than the pain experienced by those who were alone. For pairs that had spoken beforehand, the patients considered their physician to be better able to understand their pain, and the physicians estimated the perceived pain intensity of their patients more accurately.
Evidence of trust
There was greater activity in the dorsolateral and ventrolateral prefrontal cortex, as well as in the primary and secondary somatosensory areas, in patients in the subgroup that had spoken to a physician. For the physicians, compared with the comparison group, there was an increase in correspondence between activity in the dorsolateral prefrontal cortex and activity in the secondary somatosensory areas of patients, which is a brain region that is known to react to pain. The brain activity correlation increased in line with the self-reported mutual trust between the physician and patient.
“These results prove that empathy and support can decrease pain intensity,” the investigators write. The data shed light on the brain processes behind the social modulation of pain during the interaction between the physician and the patient. Concordances in the brain are increased by greater therapeutic alliance.
Beyond medication
Winfried Meissner, MD, head of the pain clinic at the department of anesthesiology and intensive care medicine at Jena University Hospital, Germany, and former president of the German Pain Society, said in an interview: “I view this as a vital study that impressively demonstrates that effective, intensive pain therapy is not just a case of administering the correct analgesic.”
“Instead, a focus should be placed on what common sense tells us, which is just how crucial an empathetic attitude from physicians and good communication with patients are when it comes to the success of any therapy,” Dr. Meissner added. Unfortunately, such an attitude and such communication often are not provided in clinical practice because of limitations on time.
“Now, with objectively collected data from patients and physicians, [Dr.] Ellingsen’s team has been able to demonstrate that human interaction has a decisive impact on the treatment of patients experiencing pain,” said Dr. Meissner. “The study should encourage practitioners to treat communication just as seriously as the pharmacology of analgesics.”
Perception and attitude
“The study shows remarkably well that empathetic conversation between the physician and patient represents a valuable therapeutic method and should be recognized as such,” emphasized Dr. Meissner. Of course, conversation cannot replace pharmacologic treatment, but it can supplement and reinforce it. Furthermore, a physician’s empathy presumably has an effect that is at least as great as a suitable analgesic.
“Pain is more than just sensory perception,” explained Dr. Meissner. “We all know that it has a strong affective component, and perception is greatly determined by context.” This can be seen, for example, in athletes, who often attribute less importance to their pain and can successfully perform competitively despite a painful injury.
Positive expectations
Dr. Meissner advised all physicians to treat patients with pain empathetically. He encourages them to ask patients about their pain, accompanying symptoms, possible fears, and other mental stress and to take these factors seriously.
Moreover, the findings accentuate the effect of prescribed analgesics. “Numerous studies have meanwhile shown that the more positive a patient’s expectations, the better the effect of a medication,” said Dr. Meissner. “We physicians must exploit this effect, too.”
This article was translated from the Medscape German Edition and a version appeared on Medscape.com.
These are the results of a study, recently published in the Proceedings of the National Academy of Sciences, that was conducted by a team led by neuroscientist Dan-Mikael Ellingsen, PhD, from Oslo University Hospital.
The researchers used functional MRI to scan the brains of 20 patients with chronic pain to investigate how a physician’s demeanor may affect patients’ sensitivity to pain, including effects in the central nervous system. During the scans, which were conducted in two sessions, the patients’ legs were exposed to stimuli that ranged from painless to moderately painful. The patients recorded perceived pain intensity using a scale. The physicians also underwent fMRI.
Half of the patients were subjected to the pain stimuli while alone; the other half were subjected to pain while in the presence of a physician. The latter group of patients was divided into two subgroups. Half of the patients had spoken to the accompanying physician before the examination. They discussed the history of the patient’s condition to date, among other things. The other half underwent the brain scans without any prior interaction with a physician.
Worse when alone
Dr. Ellingsen and his colleagues found that patients who were alone during the examination reported greater pain than those who were in the presence of a physician, even though they were subjected to stimuli of the same intensity. In instances in which the physician and patient had already spoken before the brain scan, patients additionally felt that the physician was empathetic and understood their pain. Furthermore, the physicians were better able to estimate the pain that their patients experienced.
The patients who had a physician by their side consistently experienced pain that was milder than the pain experienced by those who were alone. For pairs that had spoken beforehand, the patients considered their physician to be better able to understand their pain, and the physicians estimated the perceived pain intensity of their patients more accurately.
Evidence of trust
There was greater activity in the dorsolateral and ventrolateral prefrontal cortex, as well as in the primary and secondary somatosensory areas, in patients in the subgroup that had spoken to a physician. For the physicians, compared with the comparison group, there was an increase in correspondence between activity in the dorsolateral prefrontal cortex and activity in the secondary somatosensory areas of patients, which is a brain region that is known to react to pain. The brain activity correlation increased in line with the self-reported mutual trust between the physician and patient.
“These results prove that empathy and support can decrease pain intensity,” the investigators write. The data shed light on the brain processes behind the social modulation of pain during the interaction between the physician and the patient. Concordances in the brain are increased by greater therapeutic alliance.
Beyond medication
Winfried Meissner, MD, head of the pain clinic at the department of anesthesiology and intensive care medicine at Jena University Hospital, Germany, and former president of the German Pain Society, said in an interview: “I view this as a vital study that impressively demonstrates that effective, intensive pain therapy is not just a case of administering the correct analgesic.”
“Instead, a focus should be placed on what common sense tells us, which is just how crucial an empathetic attitude from physicians and good communication with patients are when it comes to the success of any therapy,” Dr. Meissner added. Unfortunately, such an attitude and such communication often are not provided in clinical practice because of limitations on time.
“Now, with objectively collected data from patients and physicians, [Dr.] Ellingsen’s team has been able to demonstrate that human interaction has a decisive impact on the treatment of patients experiencing pain,” said Dr. Meissner. “The study should encourage practitioners to treat communication just as seriously as the pharmacology of analgesics.”
Perception and attitude
“The study shows remarkably well that empathetic conversation between the physician and patient represents a valuable therapeutic method and should be recognized as such,” emphasized Dr. Meissner. Of course, conversation cannot replace pharmacologic treatment, but it can supplement and reinforce it. Furthermore, a physician’s empathy presumably has an effect that is at least as great as a suitable analgesic.
“Pain is more than just sensory perception,” explained Dr. Meissner. “We all know that it has a strong affective component, and perception is greatly determined by context.” This can be seen, for example, in athletes, who often attribute less importance to their pain and can successfully perform competitively despite a painful injury.
Positive expectations
Dr. Meissner advised all physicians to treat patients with pain empathetically. He encourages them to ask patients about their pain, accompanying symptoms, possible fears, and other mental stress and to take these factors seriously.
Moreover, the findings accentuate the effect of prescribed analgesics. “Numerous studies have meanwhile shown that the more positive a patient’s expectations, the better the effect of a medication,” said Dr. Meissner. “We physicians must exploit this effect, too.”
This article was translated from the Medscape German Edition and a version appeared on Medscape.com.
FROM THE PROCEEDINGS OF THE NATIONAL ACADEMY OF SCIENCES
Humira biosimilars: Five things to know
The best-selling drug Humira (adalimumab) now faces competition in the United States after a 20-year monopoly. The first adalimumab biosimilar, Amjevita, launched in the United States on January 31, and in July, seven additional biosimilars became available. These drugs have the potential to lower prescription drug prices, but when and by how much remains to be seen.
Here’s what you need to know about adalimumab biosimilars.
What Humira biosimilars are now available?
Eight different biosimilars have launched in 2023 with discounts as large at 85% from Humira’s list price of $6,922. A few companies also offer two price points.
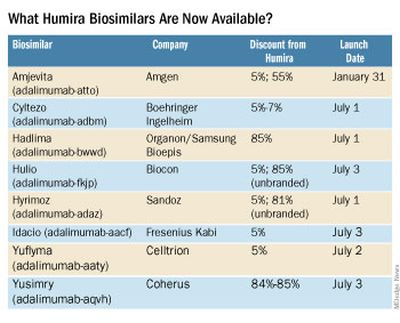
Three of these biosimilars – Hadlima, Hyrimoz, and Yuflyma – are available in high concentration formulations. This high concentration formulation makes up 85% of Humira prescriptions, according to a report from Goodroot, a collection of companies focused on lowering health care costs.
Cyltezo is currently the only adalimumab biosimilar with an interchangeability designation, meaning that a pharmacist can substitute the biosimilar for an equivalent Humira prescription without the intervention of a clinician. A total of 47 states allow for these substitutions without prior approval from a clinician, according to Goodroot, and the clinician must be notified of the switch within a certain time frame. A total of 40 states require that patients be notified of the switch before substitution.
However, it’s not clear if this interchangeability designation will prove an advantage for Cyltezo, as it is interchangeable with the lower concentration version of Humira that makes up just 15% of prescriptions.
Most of the companies behind these biosimilars are pursuing interchangeability designations for their drugs, except for Fresenius Kabi (Idacio) and Coherus (Yusimry).
A ninth biosimilar, Pfizer’s adalimumab-afzb (Abrilada), is not yet on the market and is currently awaiting an approval decision from the Food and Drug Administration to add an interchangeability designation to its prior approval for a low-concentration formulation.
Why are they priced differently?
The two price points offer different deals to payers. Pharmacy benefit managers make confidential agreements with drug manufacturers to get a discount – called a rebate – to get the drug on the PBM’s formulary. The PBM keeps a portion of that rebate, and the rest is passed on to the insurance company and patients. Biosimilars at a higher price point will likely offer larger rebates. Biosimilars offered at lower price points incorporate this discount up front in their list pricing and likely will not offer large rebates.
Will biosimilars be covered by payers?
Currently, biosimilars are being offered on formularies at parity with Humira, meaning they are on the same tier. The PBM companies OptumRx and Cigna Group’s Express Scripts will offer Amjevita (at both price points), Cyltezo, and Hyrimoz (at both price points).
“This decision allows our clients flexibility to provide access to the lower list price, so members in high-deductible plans and benefit designs with coinsurance can experience lower out-of-pocket costs,” said OptumRx spokesperson Isaac Sorensen in an email.
Mark Cuban Cost Plus Drug Company, which uses a direct-to-consumer model, will offer Yusimry for $567.27 on its website. SmithRx, a PBM based in San Francisco, announced it would partner with Cost Plus Drugs to offer Yusimry, adding that SmithRx members can use their insurance benefits to further reduce out-of-pocket costs. RxPreferred, another PBM, will also offer Yusimry through its partnership with Cuban’s company.
The news website Formulary Watch previously reported that CVS Caremark, another of the biggest PBMs, will be offering Amjevita, but as a nonpreferred brand, while Humira remains the preferred brand. CVS Caremark did not respond to a request for comment.
Will patients pay less?
Biosimilars have been touted as a potential solution to lower spending on biologic drugs, but it’s unknown if patients will ultimately benefit with lower out-of-pocket costs. It’s “impossible to predict” if the discount that third-party payers pay will be passed on to consumers, said Mark Fendrick, MD, who directs the University of Michigan Center for Value-based Insurance Design in Ann Arbor.
Generally, a consumer’s copay is a percentage of a drug’s list price, so it stands to reason that a low drug price would result in lower out-of-pocket payments. While this is mostly true, Humira has a successful copay assistance program to lower prescription costs for consumers. According to a 2022 IQVIA report, 82% of commercial prescriptions cost patients less than $10 for Humira because of this program.
To appeal to patients, biosimilar companies will need to offer similar savings, Dr. Fendrick added. “There will be some discontent if patients are actually asked to pay more out-of-pocket for a less expensive drug,” he said.
All eight companies behind these biosimilars are offering or will be launching copay saving programs, many which advertise copays as low as $0 per month for eligible patients.
How will Humira respond?
Marta Wosińska, PhD, a health care economist at the Brookings Institute, Washington, predicts payers will use these lower biosimilar prices to negotiate better deals with AbbVie, Humira’s manufacturer. “We have a lot of players coming into [the market] right now, so the competition is really fierce,” she said. In response, AbbVie will need to increase rebates on Humira and/or lower its price to compete with these biosimilars.
“The ball is in AbbVie’s court,” she said. “If [the company] is not willing to drop price sufficiently, then payers will start switching to biosimilars.”
Dr. Fendrick reported past financial relationships and consulting arrangements with AbbVie, Amgen, Arnold Ventures, Bayer, CareFirst, BlueCross BlueShield, and many other companies. Dr. Wosińska has received funding from Arnold Ventures and serves as an expert witness on antitrust cases involving generic medication.
A version of this article first appeared on Medscape.com.
The best-selling drug Humira (adalimumab) now faces competition in the United States after a 20-year monopoly. The first adalimumab biosimilar, Amjevita, launched in the United States on January 31, and in July, seven additional biosimilars became available. These drugs have the potential to lower prescription drug prices, but when and by how much remains to be seen.
Here’s what you need to know about adalimumab biosimilars.
What Humira biosimilars are now available?
Eight different biosimilars have launched in 2023 with discounts as large at 85% from Humira’s list price of $6,922. A few companies also offer two price points.

Three of these biosimilars – Hadlima, Hyrimoz, and Yuflyma – are available in high concentration formulations. This high concentration formulation makes up 85% of Humira prescriptions, according to a report from Goodroot, a collection of companies focused on lowering health care costs.
Cyltezo is currently the only adalimumab biosimilar with an interchangeability designation, meaning that a pharmacist can substitute the biosimilar for an equivalent Humira prescription without the intervention of a clinician. A total of 47 states allow for these substitutions without prior approval from a clinician, according to Goodroot, and the clinician must be notified of the switch within a certain time frame. A total of 40 states require that patients be notified of the switch before substitution.
However, it’s not clear if this interchangeability designation will prove an advantage for Cyltezo, as it is interchangeable with the lower concentration version of Humira that makes up just 15% of prescriptions.
Most of the companies behind these biosimilars are pursuing interchangeability designations for their drugs, except for Fresenius Kabi (Idacio) and Coherus (Yusimry).
A ninth biosimilar, Pfizer’s adalimumab-afzb (Abrilada), is not yet on the market and is currently awaiting an approval decision from the Food and Drug Administration to add an interchangeability designation to its prior approval for a low-concentration formulation.
Why are they priced differently?
The two price points offer different deals to payers. Pharmacy benefit managers make confidential agreements with drug manufacturers to get a discount – called a rebate – to get the drug on the PBM’s formulary. The PBM keeps a portion of that rebate, and the rest is passed on to the insurance company and patients. Biosimilars at a higher price point will likely offer larger rebates. Biosimilars offered at lower price points incorporate this discount up front in their list pricing and likely will not offer large rebates.
Will biosimilars be covered by payers?
Currently, biosimilars are being offered on formularies at parity with Humira, meaning they are on the same tier. The PBM companies OptumRx and Cigna Group’s Express Scripts will offer Amjevita (at both price points), Cyltezo, and Hyrimoz (at both price points).
“This decision allows our clients flexibility to provide access to the lower list price, so members in high-deductible plans and benefit designs with coinsurance can experience lower out-of-pocket costs,” said OptumRx spokesperson Isaac Sorensen in an email.
Mark Cuban Cost Plus Drug Company, which uses a direct-to-consumer model, will offer Yusimry for $567.27 on its website. SmithRx, a PBM based in San Francisco, announced it would partner with Cost Plus Drugs to offer Yusimry, adding that SmithRx members can use their insurance benefits to further reduce out-of-pocket costs. RxPreferred, another PBM, will also offer Yusimry through its partnership with Cuban’s company.
The news website Formulary Watch previously reported that CVS Caremark, another of the biggest PBMs, will be offering Amjevita, but as a nonpreferred brand, while Humira remains the preferred brand. CVS Caremark did not respond to a request for comment.
Will patients pay less?
Biosimilars have been touted as a potential solution to lower spending on biologic drugs, but it’s unknown if patients will ultimately benefit with lower out-of-pocket costs. It’s “impossible to predict” if the discount that third-party payers pay will be passed on to consumers, said Mark Fendrick, MD, who directs the University of Michigan Center for Value-based Insurance Design in Ann Arbor.
Generally, a consumer’s copay is a percentage of a drug’s list price, so it stands to reason that a low drug price would result in lower out-of-pocket payments. While this is mostly true, Humira has a successful copay assistance program to lower prescription costs for consumers. According to a 2022 IQVIA report, 82% of commercial prescriptions cost patients less than $10 for Humira because of this program.
To appeal to patients, biosimilar companies will need to offer similar savings, Dr. Fendrick added. “There will be some discontent if patients are actually asked to pay more out-of-pocket for a less expensive drug,” he said.
All eight companies behind these biosimilars are offering or will be launching copay saving programs, many which advertise copays as low as $0 per month for eligible patients.
How will Humira respond?
Marta Wosińska, PhD, a health care economist at the Brookings Institute, Washington, predicts payers will use these lower biosimilar prices to negotiate better deals with AbbVie, Humira’s manufacturer. “We have a lot of players coming into [the market] right now, so the competition is really fierce,” she said. In response, AbbVie will need to increase rebates on Humira and/or lower its price to compete with these biosimilars.
“The ball is in AbbVie’s court,” she said. “If [the company] is not willing to drop price sufficiently, then payers will start switching to biosimilars.”
Dr. Fendrick reported past financial relationships and consulting arrangements with AbbVie, Amgen, Arnold Ventures, Bayer, CareFirst, BlueCross BlueShield, and many other companies. Dr. Wosińska has received funding from Arnold Ventures and serves as an expert witness on antitrust cases involving generic medication.
A version of this article first appeared on Medscape.com.
The best-selling drug Humira (adalimumab) now faces competition in the United States after a 20-year monopoly. The first adalimumab biosimilar, Amjevita, launched in the United States on January 31, and in July, seven additional biosimilars became available. These drugs have the potential to lower prescription drug prices, but when and by how much remains to be seen.
Here’s what you need to know about adalimumab biosimilars.
What Humira biosimilars are now available?
Eight different biosimilars have launched in 2023 with discounts as large at 85% from Humira’s list price of $6,922. A few companies also offer two price points.

Three of these biosimilars – Hadlima, Hyrimoz, and Yuflyma – are available in high concentration formulations. This high concentration formulation makes up 85% of Humira prescriptions, according to a report from Goodroot, a collection of companies focused on lowering health care costs.
Cyltezo is currently the only adalimumab biosimilar with an interchangeability designation, meaning that a pharmacist can substitute the biosimilar for an equivalent Humira prescription without the intervention of a clinician. A total of 47 states allow for these substitutions without prior approval from a clinician, according to Goodroot, and the clinician must be notified of the switch within a certain time frame. A total of 40 states require that patients be notified of the switch before substitution.
However, it’s not clear if this interchangeability designation will prove an advantage for Cyltezo, as it is interchangeable with the lower concentration version of Humira that makes up just 15% of prescriptions.
Most of the companies behind these biosimilars are pursuing interchangeability designations for their drugs, except for Fresenius Kabi (Idacio) and Coherus (Yusimry).
A ninth biosimilar, Pfizer’s adalimumab-afzb (Abrilada), is not yet on the market and is currently awaiting an approval decision from the Food and Drug Administration to add an interchangeability designation to its prior approval for a low-concentration formulation.
Why are they priced differently?
The two price points offer different deals to payers. Pharmacy benefit managers make confidential agreements with drug manufacturers to get a discount – called a rebate – to get the drug on the PBM’s formulary. The PBM keeps a portion of that rebate, and the rest is passed on to the insurance company and patients. Biosimilars at a higher price point will likely offer larger rebates. Biosimilars offered at lower price points incorporate this discount up front in their list pricing and likely will not offer large rebates.
Will biosimilars be covered by payers?
Currently, biosimilars are being offered on formularies at parity with Humira, meaning they are on the same tier. The PBM companies OptumRx and Cigna Group’s Express Scripts will offer Amjevita (at both price points), Cyltezo, and Hyrimoz (at both price points).
“This decision allows our clients flexibility to provide access to the lower list price, so members in high-deductible plans and benefit designs with coinsurance can experience lower out-of-pocket costs,” said OptumRx spokesperson Isaac Sorensen in an email.
Mark Cuban Cost Plus Drug Company, which uses a direct-to-consumer model, will offer Yusimry for $567.27 on its website. SmithRx, a PBM based in San Francisco, announced it would partner with Cost Plus Drugs to offer Yusimry, adding that SmithRx members can use their insurance benefits to further reduce out-of-pocket costs. RxPreferred, another PBM, will also offer Yusimry through its partnership with Cuban’s company.
The news website Formulary Watch previously reported that CVS Caremark, another of the biggest PBMs, will be offering Amjevita, but as a nonpreferred brand, while Humira remains the preferred brand. CVS Caremark did not respond to a request for comment.
Will patients pay less?
Biosimilars have been touted as a potential solution to lower spending on biologic drugs, but it’s unknown if patients will ultimately benefit with lower out-of-pocket costs. It’s “impossible to predict” if the discount that third-party payers pay will be passed on to consumers, said Mark Fendrick, MD, who directs the University of Michigan Center for Value-based Insurance Design in Ann Arbor.
Generally, a consumer’s copay is a percentage of a drug’s list price, so it stands to reason that a low drug price would result in lower out-of-pocket payments. While this is mostly true, Humira has a successful copay assistance program to lower prescription costs for consumers. According to a 2022 IQVIA report, 82% of commercial prescriptions cost patients less than $10 for Humira because of this program.
To appeal to patients, biosimilar companies will need to offer similar savings, Dr. Fendrick added. “There will be some discontent if patients are actually asked to pay more out-of-pocket for a less expensive drug,” he said.
All eight companies behind these biosimilars are offering or will be launching copay saving programs, many which advertise copays as low as $0 per month for eligible patients.
How will Humira respond?
Marta Wosińska, PhD, a health care economist at the Brookings Institute, Washington, predicts payers will use these lower biosimilar prices to negotiate better deals with AbbVie, Humira’s manufacturer. “We have a lot of players coming into [the market] right now, so the competition is really fierce,” she said. In response, AbbVie will need to increase rebates on Humira and/or lower its price to compete with these biosimilars.
“The ball is in AbbVie’s court,” she said. “If [the company] is not willing to drop price sufficiently, then payers will start switching to biosimilars.”
Dr. Fendrick reported past financial relationships and consulting arrangements with AbbVie, Amgen, Arnold Ventures, Bayer, CareFirst, BlueCross BlueShield, and many other companies. Dr. Wosińska has received funding from Arnold Ventures and serves as an expert witness on antitrust cases involving generic medication.
A version of this article first appeared on Medscape.com.
The sacred office space
Church architecture describes visually the idea of the sacred, which is a fundamental need of man.
– Mario Botta, Swiss architect
My parents are visiting the Holy See today – prima volta in Italia! My mom waited years for this. She isn’t meeting the Pope or attending Mass. Yet, in the Whatsapp pics they sent me, you can see tears well up as she experiences St. Peter’s Basilica. It’s a visceral response to what is just a building and a poignant example of the significance of spaces.
More than just appreciating an edifice’s grandeur or exquisiteness, we are wired to connect with spaces emotionally. Beautiful or significant buildings move us, they make us feel something. Churches, synagogues, or mosques are good examples. They combine spiritual and aesthetic allure. But so too do gorgeous hotels, Apple stores, and posh restaurants. We crave the richness of an environment experienced through our five senses. The glory of sunlight through stained glass, the smell of luxurious scent pumped into a lobby, the weight of a silky new iPhone in your hand. We also have a sixth sense, that feeling we get from knowing that we are standing in a sacred place. A physical space that connects us with something wider and deeper than ourselves.
Virtual may be the peak of convenience, but in-real-life is the pinnacle of experience. Patients will be inconvenienced and pay higher costs to experience their appointment in person. This should not be surprising. Contemplate this: Every year, millions of people will travel across the globe to stand before a wall or walk seven times around a stone building. And millions everyday will perambulate around an Apple Store, willingly paying a higher price for the same product they can buy for less elsewhere. The willingness to pay for certain experiences is remarkably high.
Every day when I cover patient messages, I offer some patients an immediate, free solution to their problem. Just today I exchanged emails with a patient thinking I had addressed her concern by reassuring her that it was a benign seborrheic keratosis. Done. She then replied, “Thanks so much, Dr. Benabio! I still would like to schedule an appointment to come in person.” So much for the efficiency of digital medicine.
Before dismissing these patients as Luddites, understand what they want is the doctor’s office experience. The sights, the smells, the sacredness of what happens here. It is no coincidence that the first clinics were temples. In ancient Greece and Rome, the sick and the gashed made pilgrimages to one of at least 300 Asclepieia, temples of healing. During the medieval period, monasteries doubled as housing for the sick until the church began constructing stand-alone hospitals, often in cross-shaped design with an altar in the middle (eventually that became the nurses station, but without the wine).
Patients entrust us with their lives and their loved ones’ lives and a visit takes on far more significance than a simple service transaction. Forty years on, I can recall visits to Dr. Bellin’s office. He saw pediatric patients out of his Victorian home office with broad, creaky hardwood floors, stained glass, and cast iron radiators. The scent of isopropyl soaked cotton balls and typewriter ink is unforgettable. Far from sterile, it was warm, safe. It was a sacred place, one for which we still sometimes drive by when doing the tour of where I grew up.
We shall forge ahead and continue to offer virtual channels to serve our patients just as any service industry. But don’t force them there. At the same time Starbucks has been building its digital app, it is also building Starbucks Reserve Roasteries. Immense cathedral edifices with warm woods and luxurious brass, the smell of roasting coffee and warm leather perfuming the air. It is where patrons will travel long distances and endure long waits to pay a lot more for a cup of coffee.
Dr. Benabio is director of Healthcare Transformation and chief of dermatology at Kaiser Permanente San Diego. The opinions expressed in this column are his own and do not represent those of Kaiser Permanente. Dr. Benabio is @Dermdoc on Twitter. Write to him at [email protected].
Church architecture describes visually the idea of the sacred, which is a fundamental need of man.
– Mario Botta, Swiss architect
My parents are visiting the Holy See today – prima volta in Italia! My mom waited years for this. She isn’t meeting the Pope or attending Mass. Yet, in the Whatsapp pics they sent me, you can see tears well up as she experiences St. Peter’s Basilica. It’s a visceral response to what is just a building and a poignant example of the significance of spaces.
More than just appreciating an edifice’s grandeur or exquisiteness, we are wired to connect with spaces emotionally. Beautiful or significant buildings move us, they make us feel something. Churches, synagogues, or mosques are good examples. They combine spiritual and aesthetic allure. But so too do gorgeous hotels, Apple stores, and posh restaurants. We crave the richness of an environment experienced through our five senses. The glory of sunlight through stained glass, the smell of luxurious scent pumped into a lobby, the weight of a silky new iPhone in your hand. We also have a sixth sense, that feeling we get from knowing that we are standing in a sacred place. A physical space that connects us with something wider and deeper than ourselves.
Virtual may be the peak of convenience, but in-real-life is the pinnacle of experience. Patients will be inconvenienced and pay higher costs to experience their appointment in person. This should not be surprising. Contemplate this: Every year, millions of people will travel across the globe to stand before a wall or walk seven times around a stone building. And millions everyday will perambulate around an Apple Store, willingly paying a higher price for the same product they can buy for less elsewhere. The willingness to pay for certain experiences is remarkably high.
Every day when I cover patient messages, I offer some patients an immediate, free solution to their problem. Just today I exchanged emails with a patient thinking I had addressed her concern by reassuring her that it was a benign seborrheic keratosis. Done. She then replied, “Thanks so much, Dr. Benabio! I still would like to schedule an appointment to come in person.” So much for the efficiency of digital medicine.
Before dismissing these patients as Luddites, understand what they want is the doctor’s office experience. The sights, the smells, the sacredness of what happens here. It is no coincidence that the first clinics were temples. In ancient Greece and Rome, the sick and the gashed made pilgrimages to one of at least 300 Asclepieia, temples of healing. During the medieval period, monasteries doubled as housing for the sick until the church began constructing stand-alone hospitals, often in cross-shaped design with an altar in the middle (eventually that became the nurses station, but without the wine).
Patients entrust us with their lives and their loved ones’ lives and a visit takes on far more significance than a simple service transaction. Forty years on, I can recall visits to Dr. Bellin’s office. He saw pediatric patients out of his Victorian home office with broad, creaky hardwood floors, stained glass, and cast iron radiators. The scent of isopropyl soaked cotton balls and typewriter ink is unforgettable. Far from sterile, it was warm, safe. It was a sacred place, one for which we still sometimes drive by when doing the tour of where I grew up.
We shall forge ahead and continue to offer virtual channels to serve our patients just as any service industry. But don’t force them there. At the same time Starbucks has been building its digital app, it is also building Starbucks Reserve Roasteries. Immense cathedral edifices with warm woods and luxurious brass, the smell of roasting coffee and warm leather perfuming the air. It is where patrons will travel long distances and endure long waits to pay a lot more for a cup of coffee.
Dr. Benabio is director of Healthcare Transformation and chief of dermatology at Kaiser Permanente San Diego. The opinions expressed in this column are his own and do not represent those of Kaiser Permanente. Dr. Benabio is @Dermdoc on Twitter. Write to him at [email protected].
Church architecture describes visually the idea of the sacred, which is a fundamental need of man.
– Mario Botta, Swiss architect
My parents are visiting the Holy See today – prima volta in Italia! My mom waited years for this. She isn’t meeting the Pope or attending Mass. Yet, in the Whatsapp pics they sent me, you can see tears well up as she experiences St. Peter’s Basilica. It’s a visceral response to what is just a building and a poignant example of the significance of spaces.
More than just appreciating an edifice’s grandeur or exquisiteness, we are wired to connect with spaces emotionally. Beautiful or significant buildings move us, they make us feel something. Churches, synagogues, or mosques are good examples. They combine spiritual and aesthetic allure. But so too do gorgeous hotels, Apple stores, and posh restaurants. We crave the richness of an environment experienced through our five senses. The glory of sunlight through stained glass, the smell of luxurious scent pumped into a lobby, the weight of a silky new iPhone in your hand. We also have a sixth sense, that feeling we get from knowing that we are standing in a sacred place. A physical space that connects us with something wider and deeper than ourselves.
Virtual may be the peak of convenience, but in-real-life is the pinnacle of experience. Patients will be inconvenienced and pay higher costs to experience their appointment in person. This should not be surprising. Contemplate this: Every year, millions of people will travel across the globe to stand before a wall or walk seven times around a stone building. And millions everyday will perambulate around an Apple Store, willingly paying a higher price for the same product they can buy for less elsewhere. The willingness to pay for certain experiences is remarkably high.
Every day when I cover patient messages, I offer some patients an immediate, free solution to their problem. Just today I exchanged emails with a patient thinking I had addressed her concern by reassuring her that it was a benign seborrheic keratosis. Done. She then replied, “Thanks so much, Dr. Benabio! I still would like to schedule an appointment to come in person.” So much for the efficiency of digital medicine.
Before dismissing these patients as Luddites, understand what they want is the doctor’s office experience. The sights, the smells, the sacredness of what happens here. It is no coincidence that the first clinics were temples. In ancient Greece and Rome, the sick and the gashed made pilgrimages to one of at least 300 Asclepieia, temples of healing. During the medieval period, monasteries doubled as housing for the sick until the church began constructing stand-alone hospitals, often in cross-shaped design with an altar in the middle (eventually that became the nurses station, but without the wine).
Patients entrust us with their lives and their loved ones’ lives and a visit takes on far more significance than a simple service transaction. Forty years on, I can recall visits to Dr. Bellin’s office. He saw pediatric patients out of his Victorian home office with broad, creaky hardwood floors, stained glass, and cast iron radiators. The scent of isopropyl soaked cotton balls and typewriter ink is unforgettable. Far from sterile, it was warm, safe. It was a sacred place, one for which we still sometimes drive by when doing the tour of where I grew up.
We shall forge ahead and continue to offer virtual channels to serve our patients just as any service industry. But don’t force them there. At the same time Starbucks has been building its digital app, it is also building Starbucks Reserve Roasteries. Immense cathedral edifices with warm woods and luxurious brass, the smell of roasting coffee and warm leather perfuming the air. It is where patrons will travel long distances and endure long waits to pay a lot more for a cup of coffee.
Dr. Benabio is director of Healthcare Transformation and chief of dermatology at Kaiser Permanente San Diego. The opinions expressed in this column are his own and do not represent those of Kaiser Permanente. Dr. Benabio is @Dermdoc on Twitter. Write to him at [email protected].
Want to add a new partner to your practice? Here’s what to consider
When the match is right, the benefits can be significant: more hands to share the load of running a medical practice, and increased revenue and expanded patient population. A partner can bring in new, complementary strengths and skills. Adding a partner is also a way to prepare for the future by setting your practice up for a smooth transition if you or another partner is looking toward retirement.
But a mismatched partnership can cost you time and money, not to mention endless amount of conflict, dysfunction, and liability. Mutual trust and a long-term commitment on both sides are critical.
“Just like with marriage, it can be very difficult, traumatic, and expensive to break up with a partner,” said Clifton Straughn, MD, partner at Direct Access MD, a concierge-service model family practice in Anderson, S.C. “So, do your due diligence and take your time.” Picking the right partner is essential.
The basics
Before you begin the process of partnership with a physician, be sure you know what you need, the skill sets you’re looking for to complement your practice, and the personality characteristics and values that are important to you so the person you choose can check all the boxes and not just add a name to the letterhead.
“A lot of times, doctors go into this with just a general idea that they need more doctors or that they would like to be bigger or have more clout,” said Tim Boden, a certified medical practice executive with over 40 years of experience. “But you have to understand that to a certain degree, if you’re bringing somebody in who has basically an identical clinical profile to yours, you’re going to be sacrificing a bit of your lunch for a while until that person builds a name for himself or herself. A new partner’s skill set should match the need that you’re trying to fill.”
Figure out and discuss with your current partners how much it will cost to bring in a partner between their compensation and additional practice expenses. How much revenue will you expect the partner to generate? Will your practice break even the first year or the second? And how will you cover any shortfall?
It’s also essential to understand how the day-to-day operation of your practice will change after you add another partner.
- Will the new partner’s percentage of ownership be the same as that of the other partners?
- Will their ownership include a percentage of the facility, equipment, supplies, and accounts receivable?
- How will you split call and work hours?
- How will decision-making work?
- How would buyout work if a partner were to leave the practice, and is there a minimum obligation, such as a 5-year commitment?
As a team, you may also want to discuss “soft skills,” or the way you’d hope a partner would represent your practice to patients and the community.
“These can be harder to quantify,” said Dr. Straughn. “Evaluating them can take artful questions and simple observation over time.”
It’s a slow process
Many practices offer paths to partnership rather than bringing in a partner straight away. With this process, an incoming physician works toward that goal. If you’re going this route, discuss this during the hiring process, so that both sides are clear about the process. Rule No. 1 is to make sure that new hires understand that partnership is possible, although it’s not a given. The typical partnership track is 2-3 years, but you can set the timeline that works best for your practice.
Mr. Boden recommends at least a year for this period so as to allow you the opportunity to evaluate the new member, how they work, and how they fit with your team. The partnership track method is typically for young or fairly new physicians.
“I would avoid ever promising an ownership position to a recruit,” said Mr. Boden. “I would only show them how it can happen and what it would look like if they qualify.”
Consider professional help
If you want to be sure you weigh all the pros and cons of your new partner, a medical practice consultant may be the way to go. A consultant can identify many situations that you might overlook.
Some services offer a medical practice assessment to help you see where you need the most help and what skills might be best to bring to the table. They might also be able to take over some of the administrative work of a new hire if you like, so you and the other partners can focus solely on interacting with and observing the clinical abilities of a potential partner.
A health care attorney can help you build a sound agreement regarding decision-making and how the fees/costs will be divided and can put legal protections in place for everyone involved.
You’ll need a buy-sell agreement (also called a partnership or shareholder agreement) that spells out the terms and conditions, including buying into and selling out of the practice. A fair agreement respects all parties, while a poor one that offers the new partner a minority share or lessor profit may favor the practice’s current partners but could breed resentment, undermining the practice’s culture and morale.
Takeaway
Ideally, you’ll select someone with excellent credentials and experience with similar goals for the practice who blends well with your staff. It’s best to find someone who fits well culturally with your office and who practices medicine with a similar patient philosophy.
To that end, Mr. Boden encourages out-of-the-box questions for interviews, such as what a potential partner wants to make sure they have room for in their life, or what their ideal work and family life looks like. The more you can assess components such as emotional intelligence, =the fuller picture you’ll get.
“You’re going to be spending major hours every week with this person, and your destiny is going to be tied up with theirs to some degree,” said Mr. Boden. You can teach somebody the job, but if you don’t genuinely like and respect them and want to work with them daily, it may not be the right fit.
A version of this article first appeared on Medscape.com.
When the match is right, the benefits can be significant: more hands to share the load of running a medical practice, and increased revenue and expanded patient population. A partner can bring in new, complementary strengths and skills. Adding a partner is also a way to prepare for the future by setting your practice up for a smooth transition if you or another partner is looking toward retirement.
But a mismatched partnership can cost you time and money, not to mention endless amount of conflict, dysfunction, and liability. Mutual trust and a long-term commitment on both sides are critical.
“Just like with marriage, it can be very difficult, traumatic, and expensive to break up with a partner,” said Clifton Straughn, MD, partner at Direct Access MD, a concierge-service model family practice in Anderson, S.C. “So, do your due diligence and take your time.” Picking the right partner is essential.
The basics
Before you begin the process of partnership with a physician, be sure you know what you need, the skill sets you’re looking for to complement your practice, and the personality characteristics and values that are important to you so the person you choose can check all the boxes and not just add a name to the letterhead.
“A lot of times, doctors go into this with just a general idea that they need more doctors or that they would like to be bigger or have more clout,” said Tim Boden, a certified medical practice executive with over 40 years of experience. “But you have to understand that to a certain degree, if you’re bringing somebody in who has basically an identical clinical profile to yours, you’re going to be sacrificing a bit of your lunch for a while until that person builds a name for himself or herself. A new partner’s skill set should match the need that you’re trying to fill.”
Figure out and discuss with your current partners how much it will cost to bring in a partner between their compensation and additional practice expenses. How much revenue will you expect the partner to generate? Will your practice break even the first year or the second? And how will you cover any shortfall?
It’s also essential to understand how the day-to-day operation of your practice will change after you add another partner.
- Will the new partner’s percentage of ownership be the same as that of the other partners?
- Will their ownership include a percentage of the facility, equipment, supplies, and accounts receivable?
- How will you split call and work hours?
- How will decision-making work?
- How would buyout work if a partner were to leave the practice, and is there a minimum obligation, such as a 5-year commitment?
As a team, you may also want to discuss “soft skills,” or the way you’d hope a partner would represent your practice to patients and the community.
“These can be harder to quantify,” said Dr. Straughn. “Evaluating them can take artful questions and simple observation over time.”
It’s a slow process
Many practices offer paths to partnership rather than bringing in a partner straight away. With this process, an incoming physician works toward that goal. If you’re going this route, discuss this during the hiring process, so that both sides are clear about the process. Rule No. 1 is to make sure that new hires understand that partnership is possible, although it’s not a given. The typical partnership track is 2-3 years, but you can set the timeline that works best for your practice.
Mr. Boden recommends at least a year for this period so as to allow you the opportunity to evaluate the new member, how they work, and how they fit with your team. The partnership track method is typically for young or fairly new physicians.
“I would avoid ever promising an ownership position to a recruit,” said Mr. Boden. “I would only show them how it can happen and what it would look like if they qualify.”
Consider professional help
If you want to be sure you weigh all the pros and cons of your new partner, a medical practice consultant may be the way to go. A consultant can identify many situations that you might overlook.
Some services offer a medical practice assessment to help you see where you need the most help and what skills might be best to bring to the table. They might also be able to take over some of the administrative work of a new hire if you like, so you and the other partners can focus solely on interacting with and observing the clinical abilities of a potential partner.
A health care attorney can help you build a sound agreement regarding decision-making and how the fees/costs will be divided and can put legal protections in place for everyone involved.
You’ll need a buy-sell agreement (also called a partnership or shareholder agreement) that spells out the terms and conditions, including buying into and selling out of the practice. A fair agreement respects all parties, while a poor one that offers the new partner a minority share or lessor profit may favor the practice’s current partners but could breed resentment, undermining the practice’s culture and morale.
Takeaway
Ideally, you’ll select someone with excellent credentials and experience with similar goals for the practice who blends well with your staff. It’s best to find someone who fits well culturally with your office and who practices medicine with a similar patient philosophy.
To that end, Mr. Boden encourages out-of-the-box questions for interviews, such as what a potential partner wants to make sure they have room for in their life, or what their ideal work and family life looks like. The more you can assess components such as emotional intelligence, =the fuller picture you’ll get.
“You’re going to be spending major hours every week with this person, and your destiny is going to be tied up with theirs to some degree,” said Mr. Boden. You can teach somebody the job, but if you don’t genuinely like and respect them and want to work with them daily, it may not be the right fit.
A version of this article first appeared on Medscape.com.
When the match is right, the benefits can be significant: more hands to share the load of running a medical practice, and increased revenue and expanded patient population. A partner can bring in new, complementary strengths and skills. Adding a partner is also a way to prepare for the future by setting your practice up for a smooth transition if you or another partner is looking toward retirement.
But a mismatched partnership can cost you time and money, not to mention endless amount of conflict, dysfunction, and liability. Mutual trust and a long-term commitment on both sides are critical.
“Just like with marriage, it can be very difficult, traumatic, and expensive to break up with a partner,” said Clifton Straughn, MD, partner at Direct Access MD, a concierge-service model family practice in Anderson, S.C. “So, do your due diligence and take your time.” Picking the right partner is essential.
The basics
Before you begin the process of partnership with a physician, be sure you know what you need, the skill sets you’re looking for to complement your practice, and the personality characteristics and values that are important to you so the person you choose can check all the boxes and not just add a name to the letterhead.
“A lot of times, doctors go into this with just a general idea that they need more doctors or that they would like to be bigger or have more clout,” said Tim Boden, a certified medical practice executive with over 40 years of experience. “But you have to understand that to a certain degree, if you’re bringing somebody in who has basically an identical clinical profile to yours, you’re going to be sacrificing a bit of your lunch for a while until that person builds a name for himself or herself. A new partner’s skill set should match the need that you’re trying to fill.”
Figure out and discuss with your current partners how much it will cost to bring in a partner between their compensation and additional practice expenses. How much revenue will you expect the partner to generate? Will your practice break even the first year or the second? And how will you cover any shortfall?
It’s also essential to understand how the day-to-day operation of your practice will change after you add another partner.
- Will the new partner’s percentage of ownership be the same as that of the other partners?
- Will their ownership include a percentage of the facility, equipment, supplies, and accounts receivable?
- How will you split call and work hours?
- How will decision-making work?
- How would buyout work if a partner were to leave the practice, and is there a minimum obligation, such as a 5-year commitment?
As a team, you may also want to discuss “soft skills,” or the way you’d hope a partner would represent your practice to patients and the community.
“These can be harder to quantify,” said Dr. Straughn. “Evaluating them can take artful questions and simple observation over time.”
It’s a slow process
Many practices offer paths to partnership rather than bringing in a partner straight away. With this process, an incoming physician works toward that goal. If you’re going this route, discuss this during the hiring process, so that both sides are clear about the process. Rule No. 1 is to make sure that new hires understand that partnership is possible, although it’s not a given. The typical partnership track is 2-3 years, but you can set the timeline that works best for your practice.
Mr. Boden recommends at least a year for this period so as to allow you the opportunity to evaluate the new member, how they work, and how they fit with your team. The partnership track method is typically for young or fairly new physicians.
“I would avoid ever promising an ownership position to a recruit,” said Mr. Boden. “I would only show them how it can happen and what it would look like if they qualify.”
Consider professional help
If you want to be sure you weigh all the pros and cons of your new partner, a medical practice consultant may be the way to go. A consultant can identify many situations that you might overlook.
Some services offer a medical practice assessment to help you see where you need the most help and what skills might be best to bring to the table. They might also be able to take over some of the administrative work of a new hire if you like, so you and the other partners can focus solely on interacting with and observing the clinical abilities of a potential partner.
A health care attorney can help you build a sound agreement regarding decision-making and how the fees/costs will be divided and can put legal protections in place for everyone involved.
You’ll need a buy-sell agreement (also called a partnership or shareholder agreement) that spells out the terms and conditions, including buying into and selling out of the practice. A fair agreement respects all parties, while a poor one that offers the new partner a minority share or lessor profit may favor the practice’s current partners but could breed resentment, undermining the practice’s culture and morale.
Takeaway
Ideally, you’ll select someone with excellent credentials and experience with similar goals for the practice who blends well with your staff. It’s best to find someone who fits well culturally with your office and who practices medicine with a similar patient philosophy.
To that end, Mr. Boden encourages out-of-the-box questions for interviews, such as what a potential partner wants to make sure they have room for in their life, or what their ideal work and family life looks like. The more you can assess components such as emotional intelligence, =the fuller picture you’ll get.
“You’re going to be spending major hours every week with this person, and your destiny is going to be tied up with theirs to some degree,” said Mr. Boden. You can teach somebody the job, but if you don’t genuinely like and respect them and want to work with them daily, it may not be the right fit.
A version of this article first appeared on Medscape.com.














