User login
Bringing you the latest news, research and reviews, exclusive interviews, podcasts, quizzes, and more.
div[contains(@class, 'header__large-screen')]
div[contains(@class, 'read-next-article')]
div[contains(@class, 'main-prefix')]
div[contains(@class, 'nav-primary')]
nav[contains(@class, 'nav-primary')]
section[contains(@class, 'footer-nav-section-wrapper')]
footer[@id='footer']
section[contains(@class, 'nav-hidden')]
div[contains(@class, 'ce-card-content')]
nav[contains(@class, 'nav-ce-stack')]
div[contains(@class, 'view-medstat-quiz-listing-panes')]
div[contains(@class, 'pane-article-sidebar-latest-news')]
div[contains(@class, 'medstat-accordion-set article-series')]
Washington medical board charges doctor with spreading COVID misinformation
Doctors and professional organizations are standing guard, hoping to protect patients from any harm that results from mistruths spread by colleagues.
Case in point: Several physicians and the American Board of Pathology filed complaints with Washington and Idaho medical boards alleging that Ryan Cole, MD, a board-certified pathologist who practices in Boise, Idaho, but who also holds a license in Washington, has spread antivaccine and pro-ivermectin statements on social media. Dr. Cole is one of the founders of America’s Frontline Doctors, a right-wing political organization. Dr. Cole did not respond to a request for comment.
Gary W. Procop, MD, CEO, American Board of Pathology, told this news organization that “as physicians and board-certified pathologists, we have a public trust, and we must be accountable to patients, society, and the profession. Misinformation can cause real harm to patients, which may include death. Misinformation diverts patients away from lifesaving vaccination and other preventive measures, promotes viral transmission, and recommends ineffective therapies that may be toxic instead of evidence-based medical care.”
Cavalcade of complaints
Several doctors also chimed in with formal complaints alleging that Cole is spreading unreliable information, according to a report from KTVB News. For example, a Boise doctor wrote in his complaint that Dr. Cole is “a major purveyor of misinformation” and called it “amazing” that the physician was continuing to publicly support debunked information about COVID-19 more than a year into the pandemic. The doctor also stated, “Cole is a health menace, abusing his status as a physician to mislead the public.”
As a result of such complaints, the Washington medical board has charged Cole with COVID-19–related violations. It is unclear whether or not the Idaho medical board will sanction the doctor. At least 12 medical boards have sanctioned doctors for similar violations since the start of the pandemic.
The statement of charges from the Washington medical board contends that since March 2021, Dr. Cole has made numerous misleading statements regarding the COVID-19 pandemic, vaccines, the use of ivermectin to treat COVID-19, and the effectiveness of masks.
In addition, the statement alleges that Dr. Cole treated several COVID-19 patients via telemedicine. During these sessions, he prescribed ivermectin, an antiparasite drug that has not been found to have any effectiveness in treating, curing, or preventing COVID-19. One of the patients died after receiving this treatment, according to the complaint.
Citing a study published in the New England Journal of Medicine, Dr. Procop pointed out that use of ivermectin, which is not approved by the U.S. Food and Drug Administration to treat COVID-19, is particularly troubling.
“There is a concern whenever an ineffective treatment is prescribed when more effective and scientifically proven therapies are available. Therapeutics have potential side effects, and toxicities have been associated with the use of ivermectin,” Dr. Procop said. “The benefits of therapy should always outweigh the risks of treatment.”
If the Washington medical board finds that Dr. Cole has engaged in unprofessional conduct, possible sanctions include revocation or suspension of his license. Washington state law also provides for a range of other possible sanctions, including restriction or limitation of his practice, requiring that he complete a specific program of remedial education or treatment, monitoring of his practice, censure or reprimand, probation, a fine of up to $5,000 for each violation, or refunding fees that his practice has billed to and collected from patients. Dr. Cole had until January 30 to respond to the medical board’s statement.
“The American Board of Pathology supports the actions of the Washington State Medical Board regarding their inquiries into any physician that holds license in their state who makes false and misleading medical claims, or provides medical care beyond their scope of practice, as indicated by their training,” Dr. Procop said.
Law in limbo
While medical boards are seeking to sanction professionals who spread falsehoods, the pause button has been hit on the California law that allows regulators to punish doctors for spreading false information about COVID-19 vaccinations and treatments.
The law went into effect Jan. 1 but was temporarily halted when U.S. District Judge William B. Shubb of the Eastern District of California granted a preliminary injunction against the law on Jan. 25, according to a report in the Sacramento Bee.
Mr. Shubb said the measure’s definition of “misinformation” was “unconstitutionally vague” under the due process clause of the 14th Amendment. He also criticized the law’s definition of “misinformation” as being “grammatically incoherent.”
A version of this article first appeared on Medscape.com.
Doctors and professional organizations are standing guard, hoping to protect patients from any harm that results from mistruths spread by colleagues.
Case in point: Several physicians and the American Board of Pathology filed complaints with Washington and Idaho medical boards alleging that Ryan Cole, MD, a board-certified pathologist who practices in Boise, Idaho, but who also holds a license in Washington, has spread antivaccine and pro-ivermectin statements on social media. Dr. Cole is one of the founders of America’s Frontline Doctors, a right-wing political organization. Dr. Cole did not respond to a request for comment.
Gary W. Procop, MD, CEO, American Board of Pathology, told this news organization that “as physicians and board-certified pathologists, we have a public trust, and we must be accountable to patients, society, and the profession. Misinformation can cause real harm to patients, which may include death. Misinformation diverts patients away from lifesaving vaccination and other preventive measures, promotes viral transmission, and recommends ineffective therapies that may be toxic instead of evidence-based medical care.”
Cavalcade of complaints
Several doctors also chimed in with formal complaints alleging that Cole is spreading unreliable information, according to a report from KTVB News. For example, a Boise doctor wrote in his complaint that Dr. Cole is “a major purveyor of misinformation” and called it “amazing” that the physician was continuing to publicly support debunked information about COVID-19 more than a year into the pandemic. The doctor also stated, “Cole is a health menace, abusing his status as a physician to mislead the public.”
As a result of such complaints, the Washington medical board has charged Cole with COVID-19–related violations. It is unclear whether or not the Idaho medical board will sanction the doctor. At least 12 medical boards have sanctioned doctors for similar violations since the start of the pandemic.
The statement of charges from the Washington medical board contends that since March 2021, Dr. Cole has made numerous misleading statements regarding the COVID-19 pandemic, vaccines, the use of ivermectin to treat COVID-19, and the effectiveness of masks.
In addition, the statement alleges that Dr. Cole treated several COVID-19 patients via telemedicine. During these sessions, he prescribed ivermectin, an antiparasite drug that has not been found to have any effectiveness in treating, curing, or preventing COVID-19. One of the patients died after receiving this treatment, according to the complaint.
Citing a study published in the New England Journal of Medicine, Dr. Procop pointed out that use of ivermectin, which is not approved by the U.S. Food and Drug Administration to treat COVID-19, is particularly troubling.
“There is a concern whenever an ineffective treatment is prescribed when more effective and scientifically proven therapies are available. Therapeutics have potential side effects, and toxicities have been associated with the use of ivermectin,” Dr. Procop said. “The benefits of therapy should always outweigh the risks of treatment.”
If the Washington medical board finds that Dr. Cole has engaged in unprofessional conduct, possible sanctions include revocation or suspension of his license. Washington state law also provides for a range of other possible sanctions, including restriction or limitation of his practice, requiring that he complete a specific program of remedial education or treatment, monitoring of his practice, censure or reprimand, probation, a fine of up to $5,000 for each violation, or refunding fees that his practice has billed to and collected from patients. Dr. Cole had until January 30 to respond to the medical board’s statement.
“The American Board of Pathology supports the actions of the Washington State Medical Board regarding their inquiries into any physician that holds license in their state who makes false and misleading medical claims, or provides medical care beyond their scope of practice, as indicated by their training,” Dr. Procop said.
Law in limbo
While medical boards are seeking to sanction professionals who spread falsehoods, the pause button has been hit on the California law that allows regulators to punish doctors for spreading false information about COVID-19 vaccinations and treatments.
The law went into effect Jan. 1 but was temporarily halted when U.S. District Judge William B. Shubb of the Eastern District of California granted a preliminary injunction against the law on Jan. 25, according to a report in the Sacramento Bee.
Mr. Shubb said the measure’s definition of “misinformation” was “unconstitutionally vague” under the due process clause of the 14th Amendment. He also criticized the law’s definition of “misinformation” as being “grammatically incoherent.”
A version of this article first appeared on Medscape.com.
Doctors and professional organizations are standing guard, hoping to protect patients from any harm that results from mistruths spread by colleagues.
Case in point: Several physicians and the American Board of Pathology filed complaints with Washington and Idaho medical boards alleging that Ryan Cole, MD, a board-certified pathologist who practices in Boise, Idaho, but who also holds a license in Washington, has spread antivaccine and pro-ivermectin statements on social media. Dr. Cole is one of the founders of America’s Frontline Doctors, a right-wing political organization. Dr. Cole did not respond to a request for comment.
Gary W. Procop, MD, CEO, American Board of Pathology, told this news organization that “as physicians and board-certified pathologists, we have a public trust, and we must be accountable to patients, society, and the profession. Misinformation can cause real harm to patients, which may include death. Misinformation diverts patients away from lifesaving vaccination and other preventive measures, promotes viral transmission, and recommends ineffective therapies that may be toxic instead of evidence-based medical care.”
Cavalcade of complaints
Several doctors also chimed in with formal complaints alleging that Cole is spreading unreliable information, according to a report from KTVB News. For example, a Boise doctor wrote in his complaint that Dr. Cole is “a major purveyor of misinformation” and called it “amazing” that the physician was continuing to publicly support debunked information about COVID-19 more than a year into the pandemic. The doctor also stated, “Cole is a health menace, abusing his status as a physician to mislead the public.”
As a result of such complaints, the Washington medical board has charged Cole with COVID-19–related violations. It is unclear whether or not the Idaho medical board will sanction the doctor. At least 12 medical boards have sanctioned doctors for similar violations since the start of the pandemic.
The statement of charges from the Washington medical board contends that since March 2021, Dr. Cole has made numerous misleading statements regarding the COVID-19 pandemic, vaccines, the use of ivermectin to treat COVID-19, and the effectiveness of masks.
In addition, the statement alleges that Dr. Cole treated several COVID-19 patients via telemedicine. During these sessions, he prescribed ivermectin, an antiparasite drug that has not been found to have any effectiveness in treating, curing, or preventing COVID-19. One of the patients died after receiving this treatment, according to the complaint.
Citing a study published in the New England Journal of Medicine, Dr. Procop pointed out that use of ivermectin, which is not approved by the U.S. Food and Drug Administration to treat COVID-19, is particularly troubling.
“There is a concern whenever an ineffective treatment is prescribed when more effective and scientifically proven therapies are available. Therapeutics have potential side effects, and toxicities have been associated with the use of ivermectin,” Dr. Procop said. “The benefits of therapy should always outweigh the risks of treatment.”
If the Washington medical board finds that Dr. Cole has engaged in unprofessional conduct, possible sanctions include revocation or suspension of his license. Washington state law also provides for a range of other possible sanctions, including restriction or limitation of his practice, requiring that he complete a specific program of remedial education or treatment, monitoring of his practice, censure or reprimand, probation, a fine of up to $5,000 for each violation, or refunding fees that his practice has billed to and collected from patients. Dr. Cole had until January 30 to respond to the medical board’s statement.
“The American Board of Pathology supports the actions of the Washington State Medical Board regarding their inquiries into any physician that holds license in their state who makes false and misleading medical claims, or provides medical care beyond their scope of practice, as indicated by their training,” Dr. Procop said.
Law in limbo
While medical boards are seeking to sanction professionals who spread falsehoods, the pause button has been hit on the California law that allows regulators to punish doctors for spreading false information about COVID-19 vaccinations and treatments.
The law went into effect Jan. 1 but was temporarily halted when U.S. District Judge William B. Shubb of the Eastern District of California granted a preliminary injunction against the law on Jan. 25, according to a report in the Sacramento Bee.
Mr. Shubb said the measure’s definition of “misinformation” was “unconstitutionally vague” under the due process clause of the 14th Amendment. He also criticized the law’s definition of “misinformation” as being “grammatically incoherent.”
A version of this article first appeared on Medscape.com.
Commentary: Glucocorticoid use and progression in RA, February 2023
Several recent studies have assessed the use of glucocorticoids, a frequent companion to disease-modifying antirheumatic drug (DMARD) and biologic therapy. Many patients are treated with glucocorticoids early in their disease course as a bridging therapy to long-term treatment, and others receive glucocorticoid therapy chronically or intermittently for flares. Van Ouwerkerk and colleagues performed a combined analysis of seven clinical trials, identified in a systematic literature review, that included a glucocorticoid taper protocol for the treatment of newly diagnosed rheumatoid arthritis (RA), undifferentiated arthritis, or "high-risk profile for persistent arthritis." These studies encompassed intravenous, intramuscular, and oral glucocorticoid regimens, and the continued use of glucocorticoids after bridging. These regimens, including cumulative doses, were examined and found to result in a low probability of ongoing use, especially in patients with lower initial doses and shorter bridging schedules. However, though reassuring as to the early use of glucocorticoids in clinical practice, this finding can be affected by patient characteristics not examined in detail in the aggregated results, including whether the patients were classified as having RA, undifferentiated arthritis, or a "high-risk profile."
Adami and colleagues also looked at tapering of glucocorticoids in patients with RA (though not necessarily early RA) in order to determine risk for flare associated with different tapering schedules. They examined the characteristics of patients with RA experiencing a flare (defined as an increase in Disease Activity Score 28 for Rheumatoid Arthritis with C-reactive protein [DAS28-CRP] > 1.2) and their glucocorticoid therapy in the preceding 6 months and found that tapering to a prednisone equivalent ≤ 2.5 mg daily was associated with a higher risk for flare but that doses > 2.5 mg daily were not. Though this finding is perhaps expected, it does not provide further insight into a strategy to minimize the associated adverse effects of glucocorticoid therapy.
Adding further weight to this point is a study performed in Denmark by Dieperink and colleagues examining risk for Staphylococcus aureus bacteremia (SAB) using a nation-wide registry of over 30,000 patients with RA. They found 180 cases of SAB and examined the patient characteristics. Patients who were currently using or previously used a biologic DMARD had an increased risk for SAB as well as those with moderate to high RA disease activity. Study participants who were currently using a prednisone-equivalent of ≤ 7.5 mg daily had an adjusted odds ratio (aOR) of 2.2 and those using > 7.5 mg daily had an aOR of 9.5 for SAB. This concerning finding suggests that even a relatively "low" dose of prednisone use is not benign for patients with RA, and these studies bring to light the need to research optimal strategies for disease control and balancing immunosuppression with the risk for infection and other adverse events.
Heckert and colleagues looked at another aspect of RA disease control, namely, local progression in a single affected joint. Their prior work has suggested that patients with RA may be prone to recurrent inflammation in a single joint despite systemic treatment, a finding that aligns with common clinical observations. This study evaluates radiographic progression in susceptible joints via post hoc analysis using data from the BeSt study including tender and swollen joints, hand and foot radiographs, and disease activity scores. Despite systemic treatment to a target low disease activity or remission state (as per the BeSt protocol), the study found an association between recurrent joint inflammation and radiographic progression (ie, erosions). However, because they only looked at hand and foot joints, the strength of this association in other joints is unknown, as is the use of local treatment, such as steroid injection to minimize inflammation, though both questions may be difficult to evaluate in a small prospective study.
Several recent studies have assessed the use of glucocorticoids, a frequent companion to disease-modifying antirheumatic drug (DMARD) and biologic therapy. Many patients are treated with glucocorticoids early in their disease course as a bridging therapy to long-term treatment, and others receive glucocorticoid therapy chronically or intermittently for flares. Van Ouwerkerk and colleagues performed a combined analysis of seven clinical trials, identified in a systematic literature review, that included a glucocorticoid taper protocol for the treatment of newly diagnosed rheumatoid arthritis (RA), undifferentiated arthritis, or "high-risk profile for persistent arthritis." These studies encompassed intravenous, intramuscular, and oral glucocorticoid regimens, and the continued use of glucocorticoids after bridging. These regimens, including cumulative doses, were examined and found to result in a low probability of ongoing use, especially in patients with lower initial doses and shorter bridging schedules. However, though reassuring as to the early use of glucocorticoids in clinical practice, this finding can be affected by patient characteristics not examined in detail in the aggregated results, including whether the patients were classified as having RA, undifferentiated arthritis, or a "high-risk profile."
Adami and colleagues also looked at tapering of glucocorticoids in patients with RA (though not necessarily early RA) in order to determine risk for flare associated with different tapering schedules. They examined the characteristics of patients with RA experiencing a flare (defined as an increase in Disease Activity Score 28 for Rheumatoid Arthritis with C-reactive protein [DAS28-CRP] > 1.2) and their glucocorticoid therapy in the preceding 6 months and found that tapering to a prednisone equivalent ≤ 2.5 mg daily was associated with a higher risk for flare but that doses > 2.5 mg daily were not. Though this finding is perhaps expected, it does not provide further insight into a strategy to minimize the associated adverse effects of glucocorticoid therapy.
Adding further weight to this point is a study performed in Denmark by Dieperink and colleagues examining risk for Staphylococcus aureus bacteremia (SAB) using a nation-wide registry of over 30,000 patients with RA. They found 180 cases of SAB and examined the patient characteristics. Patients who were currently using or previously used a biologic DMARD had an increased risk for SAB as well as those with moderate to high RA disease activity. Study participants who were currently using a prednisone-equivalent of ≤ 7.5 mg daily had an adjusted odds ratio (aOR) of 2.2 and those using > 7.5 mg daily had an aOR of 9.5 for SAB. This concerning finding suggests that even a relatively "low" dose of prednisone use is not benign for patients with RA, and these studies bring to light the need to research optimal strategies for disease control and balancing immunosuppression with the risk for infection and other adverse events.
Heckert and colleagues looked at another aspect of RA disease control, namely, local progression in a single affected joint. Their prior work has suggested that patients with RA may be prone to recurrent inflammation in a single joint despite systemic treatment, a finding that aligns with common clinical observations. This study evaluates radiographic progression in susceptible joints via post hoc analysis using data from the BeSt study including tender and swollen joints, hand and foot radiographs, and disease activity scores. Despite systemic treatment to a target low disease activity or remission state (as per the BeSt protocol), the study found an association between recurrent joint inflammation and radiographic progression (ie, erosions). However, because they only looked at hand and foot joints, the strength of this association in other joints is unknown, as is the use of local treatment, such as steroid injection to minimize inflammation, though both questions may be difficult to evaluate in a small prospective study.
Several recent studies have assessed the use of glucocorticoids, a frequent companion to disease-modifying antirheumatic drug (DMARD) and biologic therapy. Many patients are treated with glucocorticoids early in their disease course as a bridging therapy to long-term treatment, and others receive glucocorticoid therapy chronically or intermittently for flares. Van Ouwerkerk and colleagues performed a combined analysis of seven clinical trials, identified in a systematic literature review, that included a glucocorticoid taper protocol for the treatment of newly diagnosed rheumatoid arthritis (RA), undifferentiated arthritis, or "high-risk profile for persistent arthritis." These studies encompassed intravenous, intramuscular, and oral glucocorticoid regimens, and the continued use of glucocorticoids after bridging. These regimens, including cumulative doses, were examined and found to result in a low probability of ongoing use, especially in patients with lower initial doses and shorter bridging schedules. However, though reassuring as to the early use of glucocorticoids in clinical practice, this finding can be affected by patient characteristics not examined in detail in the aggregated results, including whether the patients were classified as having RA, undifferentiated arthritis, or a "high-risk profile."
Adami and colleagues also looked at tapering of glucocorticoids in patients with RA (though not necessarily early RA) in order to determine risk for flare associated with different tapering schedules. They examined the characteristics of patients with RA experiencing a flare (defined as an increase in Disease Activity Score 28 for Rheumatoid Arthritis with C-reactive protein [DAS28-CRP] > 1.2) and their glucocorticoid therapy in the preceding 6 months and found that tapering to a prednisone equivalent ≤ 2.5 mg daily was associated with a higher risk for flare but that doses > 2.5 mg daily were not. Though this finding is perhaps expected, it does not provide further insight into a strategy to minimize the associated adverse effects of glucocorticoid therapy.
Adding further weight to this point is a study performed in Denmark by Dieperink and colleagues examining risk for Staphylococcus aureus bacteremia (SAB) using a nation-wide registry of over 30,000 patients with RA. They found 180 cases of SAB and examined the patient characteristics. Patients who were currently using or previously used a biologic DMARD had an increased risk for SAB as well as those with moderate to high RA disease activity. Study participants who were currently using a prednisone-equivalent of ≤ 7.5 mg daily had an adjusted odds ratio (aOR) of 2.2 and those using > 7.5 mg daily had an aOR of 9.5 for SAB. This concerning finding suggests that even a relatively "low" dose of prednisone use is not benign for patients with RA, and these studies bring to light the need to research optimal strategies for disease control and balancing immunosuppression with the risk for infection and other adverse events.
Heckert and colleagues looked at another aspect of RA disease control, namely, local progression in a single affected joint. Their prior work has suggested that patients with RA may be prone to recurrent inflammation in a single joint despite systemic treatment, a finding that aligns with common clinical observations. This study evaluates radiographic progression in susceptible joints via post hoc analysis using data from the BeSt study including tender and swollen joints, hand and foot radiographs, and disease activity scores. Despite systemic treatment to a target low disease activity or remission state (as per the BeSt protocol), the study found an association between recurrent joint inflammation and radiographic progression (ie, erosions). However, because they only looked at hand and foot joints, the strength of this association in other joints is unknown, as is the use of local treatment, such as steroid injection to minimize inflammation, though both questions may be difficult to evaluate in a small prospective study.
First Humira biosimilar launches in U.S.
The first biosimilar for Humira, adalimumab-atto (Amjevita), is now available in the United States, according to an announcement on Jan. 31 by the manufacturer, Amgen. At least seven other U.S. Food and Drug Administration–approved Humira biosimilars are expected to become available later in 2023.
Amjevita was approved by the FDA in September 2016 for multiple inflammatory diseases, including rheumatoid arthritis, psoriatic arthritis, ankylosing spondylitis, Crohn’s disease, ulcerative colitis, and plaque psoriasis. The delayed launch was part of a global settlement with Humira’s manufacturer, AbbVie.
Humira (adalimumab) has been available since 2002 and is consistently one of the top-selling drugs in the United States. A single 40-mg Amjevita pen device will be available at two prices: a list price (wholesale acquisition cost) of $1,557.59, 55% below the current Humira list price, and a list price of $3,288.24, 5% below the current Humira list price, according to Amgen.
“Amgen’s goal is to provide broad access for patients by offering two options to health plans and pharmacy benefit managers,” the company said in the press release.
Patients are less likely to benefit from the more significant discount, said Marta Wosinska, PhD, a health care economist at the Brookings Institute in Washington, DC. It's expected that insurance companies will use the higher list price for Amjevita, she said, as this higher price will also likely have higher rebates. Rebates are payments to health insurance payers provided by drug manufacturers to promote use of an expensive drug. Some pharmacy benefit managers have already said that they plan to charge patients the same amount for Humira as its biosimilars, Dr. Wosinska said.
"For an existing patient, there's really no incentive for them to switch," she said in an interview.
So far only one insurance company, Kaiser Permanente, has plans to switch patients over to biosimilars, according to the health policy podcast Tradeoffs, and the insurer will stop covering Humira by the end of this year.
A version of this article first appeared on Medscape.com.
*This story was updated 2/1/2023.
The first biosimilar for Humira, adalimumab-atto (Amjevita), is now available in the United States, according to an announcement on Jan. 31 by the manufacturer, Amgen. At least seven other U.S. Food and Drug Administration–approved Humira biosimilars are expected to become available later in 2023.
Amjevita was approved by the FDA in September 2016 for multiple inflammatory diseases, including rheumatoid arthritis, psoriatic arthritis, ankylosing spondylitis, Crohn’s disease, ulcerative colitis, and plaque psoriasis. The delayed launch was part of a global settlement with Humira’s manufacturer, AbbVie.
Humira (adalimumab) has been available since 2002 and is consistently one of the top-selling drugs in the United States. A single 40-mg Amjevita pen device will be available at two prices: a list price (wholesale acquisition cost) of $1,557.59, 55% below the current Humira list price, and a list price of $3,288.24, 5% below the current Humira list price, according to Amgen.
“Amgen’s goal is to provide broad access for patients by offering two options to health plans and pharmacy benefit managers,” the company said in the press release.
Patients are less likely to benefit from the more significant discount, said Marta Wosinska, PhD, a health care economist at the Brookings Institute in Washington, DC. It's expected that insurance companies will use the higher list price for Amjevita, she said, as this higher price will also likely have higher rebates. Rebates are payments to health insurance payers provided by drug manufacturers to promote use of an expensive drug. Some pharmacy benefit managers have already said that they plan to charge patients the same amount for Humira as its biosimilars, Dr. Wosinska said.
"For an existing patient, there's really no incentive for them to switch," she said in an interview.
So far only one insurance company, Kaiser Permanente, has plans to switch patients over to biosimilars, according to the health policy podcast Tradeoffs, and the insurer will stop covering Humira by the end of this year.
A version of this article first appeared on Medscape.com.
*This story was updated 2/1/2023.
The first biosimilar for Humira, adalimumab-atto (Amjevita), is now available in the United States, according to an announcement on Jan. 31 by the manufacturer, Amgen. At least seven other U.S. Food and Drug Administration–approved Humira biosimilars are expected to become available later in 2023.
Amjevita was approved by the FDA in September 2016 for multiple inflammatory diseases, including rheumatoid arthritis, psoriatic arthritis, ankylosing spondylitis, Crohn’s disease, ulcerative colitis, and plaque psoriasis. The delayed launch was part of a global settlement with Humira’s manufacturer, AbbVie.
Humira (adalimumab) has been available since 2002 and is consistently one of the top-selling drugs in the United States. A single 40-mg Amjevita pen device will be available at two prices: a list price (wholesale acquisition cost) of $1,557.59, 55% below the current Humira list price, and a list price of $3,288.24, 5% below the current Humira list price, according to Amgen.
“Amgen’s goal is to provide broad access for patients by offering two options to health plans and pharmacy benefit managers,” the company said in the press release.
Patients are less likely to benefit from the more significant discount, said Marta Wosinska, PhD, a health care economist at the Brookings Institute in Washington, DC. It's expected that insurance companies will use the higher list price for Amjevita, she said, as this higher price will also likely have higher rebates. Rebates are payments to health insurance payers provided by drug manufacturers to promote use of an expensive drug. Some pharmacy benefit managers have already said that they plan to charge patients the same amount for Humira as its biosimilars, Dr. Wosinska said.
"For an existing patient, there's really no incentive for them to switch," she said in an interview.
So far only one insurance company, Kaiser Permanente, has plans to switch patients over to biosimilars, according to the health policy podcast Tradeoffs, and the insurer will stop covering Humira by the end of this year.
A version of this article first appeared on Medscape.com.
*This story was updated 2/1/2023.
Citing workplace violence, one-fourth of critical care workers are ready to quit
A surgeon in Tulsa shot by a disgruntled patient. A doctor in India beaten by a group of bereaved family members. A general practitioner in the United Kingdom threatened with stabbing. A new study identifies this trend and finds that 25% of health care workers polled were willing to quit because of such violence.
“That was pretty appalling,” Rahul Kashyap, MD, MBA, MBBS, recalls. Dr. Kashyap is one of the leaders of the Violence Study of Healthcare Workers and Systems (ViSHWaS), which polled an international sample of physicians, nurses, and hospital staff. This study has worrying implications, Dr. Kashyap says. In a time when hospital staff are reporting burnout in record numbers, further deterrents may be the last thing our health care system needs. But Dr. Kashyap hopes that bringing awareness to these trends may allow physicians, policymakers, and the public to mobilize and intervene before it’s too late.
Previous studies have revealed similar trends. The rate of workplace violence directed at U.S. health care workers is five times that of workers in any other industry, according to the Bureau of Labor Statistics. The same study found that attacks had increased 63% from 2011 to 2018. Other polls that focus on the pandemic show that nearly half of U.S. nurses believe that violence increased since the world shut down. Well before the pandemic, however, a study from the Indian Medical Association found that 75% of doctors experienced workplace violence.
With this history in mind, perhaps it’s not surprising that the idea for the study came from the authors’ personal experiences. They had seen coworkers go through attacks, or they had endured attacks themselves, Dr. Kashyap says. But they couldn’t find any global data to back up these experiences. So Dr. Kashyap and his colleagues formed a web of volunteers dedicated to creating a cross-sectional study.
They got in touch with researchers from countries across Asia, the Middle East, South America, North America, and Africa. The initial group agreed to reach out to their contacts, casting a wide net. Researchers used WhatsApp, LinkedIn, and text messages to distribute the survey. Health care workers in each country completed the brief questionnaire, recalling their prepandemic world and evaluating their current one.
Within 2 months, they had reached health care workers in more than 100 countries. They concluded the study when they received about 5,000 results, according to Dr. Kashyap, and then began the process of stratifying the data. For this report, they focused on critical care, emergency medicine, and anesthesiology, which resulted in 598 responses from 69 countries. Of these, India and the United States had the highest number of participants.
In all, 73% of participants reported facing physical or verbal violence while in the hospital; 48% said they felt less motivated to work because of that violence; 39% of respondents believed that the amount of violence they experienced was the same as before the COVID-19 pandemic; and 36% of respondents believed that violence had increased. Even though they were trained on guidelines from the Occupational Safety and Health Administration, 20% of participants felt unprepared to face violence.
Although the study didn’t analyze the reasons workers felt this way, Dr. Kashyap speculates that it could be related to the medical distrust that grew during the pandemic or the stress patients and health care professionals experienced during its peak.
Regardless, the researchers say their study is a starting point. Now that the trend has been highlighted, it may be acted on.
Moving forward, Dr. Kashyap believes that controlling for different variables could determine whether factors like gender or shift time put a worker at higher risk for violence. He hopes it’s possible to interrupt these patterns and reestablish trust in the hospital environment. “It’s aspirational, but you’re hoping that through studies like ViSHWaS, which means trust in Hindi ... [we could restore] the trust and confidence among health care providers for the patients and family members.”
A version of this article first appeared on Medscape.com.
A surgeon in Tulsa shot by a disgruntled patient. A doctor in India beaten by a group of bereaved family members. A general practitioner in the United Kingdom threatened with stabbing. A new study identifies this trend and finds that 25% of health care workers polled were willing to quit because of such violence.
“That was pretty appalling,” Rahul Kashyap, MD, MBA, MBBS, recalls. Dr. Kashyap is one of the leaders of the Violence Study of Healthcare Workers and Systems (ViSHWaS), which polled an international sample of physicians, nurses, and hospital staff. This study has worrying implications, Dr. Kashyap says. In a time when hospital staff are reporting burnout in record numbers, further deterrents may be the last thing our health care system needs. But Dr. Kashyap hopes that bringing awareness to these trends may allow physicians, policymakers, and the public to mobilize and intervene before it’s too late.
Previous studies have revealed similar trends. The rate of workplace violence directed at U.S. health care workers is five times that of workers in any other industry, according to the Bureau of Labor Statistics. The same study found that attacks had increased 63% from 2011 to 2018. Other polls that focus on the pandemic show that nearly half of U.S. nurses believe that violence increased since the world shut down. Well before the pandemic, however, a study from the Indian Medical Association found that 75% of doctors experienced workplace violence.
With this history in mind, perhaps it’s not surprising that the idea for the study came from the authors’ personal experiences. They had seen coworkers go through attacks, or they had endured attacks themselves, Dr. Kashyap says. But they couldn’t find any global data to back up these experiences. So Dr. Kashyap and his colleagues formed a web of volunteers dedicated to creating a cross-sectional study.
They got in touch with researchers from countries across Asia, the Middle East, South America, North America, and Africa. The initial group agreed to reach out to their contacts, casting a wide net. Researchers used WhatsApp, LinkedIn, and text messages to distribute the survey. Health care workers in each country completed the brief questionnaire, recalling their prepandemic world and evaluating their current one.
Within 2 months, they had reached health care workers in more than 100 countries. They concluded the study when they received about 5,000 results, according to Dr. Kashyap, and then began the process of stratifying the data. For this report, they focused on critical care, emergency medicine, and anesthesiology, which resulted in 598 responses from 69 countries. Of these, India and the United States had the highest number of participants.
In all, 73% of participants reported facing physical or verbal violence while in the hospital; 48% said they felt less motivated to work because of that violence; 39% of respondents believed that the amount of violence they experienced was the same as before the COVID-19 pandemic; and 36% of respondents believed that violence had increased. Even though they were trained on guidelines from the Occupational Safety and Health Administration, 20% of participants felt unprepared to face violence.
Although the study didn’t analyze the reasons workers felt this way, Dr. Kashyap speculates that it could be related to the medical distrust that grew during the pandemic or the stress patients and health care professionals experienced during its peak.
Regardless, the researchers say their study is a starting point. Now that the trend has been highlighted, it may be acted on.
Moving forward, Dr. Kashyap believes that controlling for different variables could determine whether factors like gender or shift time put a worker at higher risk for violence. He hopes it’s possible to interrupt these patterns and reestablish trust in the hospital environment. “It’s aspirational, but you’re hoping that through studies like ViSHWaS, which means trust in Hindi ... [we could restore] the trust and confidence among health care providers for the patients and family members.”
A version of this article first appeared on Medscape.com.
A surgeon in Tulsa shot by a disgruntled patient. A doctor in India beaten by a group of bereaved family members. A general practitioner in the United Kingdom threatened with stabbing. A new study identifies this trend and finds that 25% of health care workers polled were willing to quit because of such violence.
“That was pretty appalling,” Rahul Kashyap, MD, MBA, MBBS, recalls. Dr. Kashyap is one of the leaders of the Violence Study of Healthcare Workers and Systems (ViSHWaS), which polled an international sample of physicians, nurses, and hospital staff. This study has worrying implications, Dr. Kashyap says. In a time when hospital staff are reporting burnout in record numbers, further deterrents may be the last thing our health care system needs. But Dr. Kashyap hopes that bringing awareness to these trends may allow physicians, policymakers, and the public to mobilize and intervene before it’s too late.
Previous studies have revealed similar trends. The rate of workplace violence directed at U.S. health care workers is five times that of workers in any other industry, according to the Bureau of Labor Statistics. The same study found that attacks had increased 63% from 2011 to 2018. Other polls that focus on the pandemic show that nearly half of U.S. nurses believe that violence increased since the world shut down. Well before the pandemic, however, a study from the Indian Medical Association found that 75% of doctors experienced workplace violence.
With this history in mind, perhaps it’s not surprising that the idea for the study came from the authors’ personal experiences. They had seen coworkers go through attacks, or they had endured attacks themselves, Dr. Kashyap says. But they couldn’t find any global data to back up these experiences. So Dr. Kashyap and his colleagues formed a web of volunteers dedicated to creating a cross-sectional study.
They got in touch with researchers from countries across Asia, the Middle East, South America, North America, and Africa. The initial group agreed to reach out to their contacts, casting a wide net. Researchers used WhatsApp, LinkedIn, and text messages to distribute the survey. Health care workers in each country completed the brief questionnaire, recalling their prepandemic world and evaluating their current one.
Within 2 months, they had reached health care workers in more than 100 countries. They concluded the study when they received about 5,000 results, according to Dr. Kashyap, and then began the process of stratifying the data. For this report, they focused on critical care, emergency medicine, and anesthesiology, which resulted in 598 responses from 69 countries. Of these, India and the United States had the highest number of participants.
In all, 73% of participants reported facing physical or verbal violence while in the hospital; 48% said they felt less motivated to work because of that violence; 39% of respondents believed that the amount of violence they experienced was the same as before the COVID-19 pandemic; and 36% of respondents believed that violence had increased. Even though they were trained on guidelines from the Occupational Safety and Health Administration, 20% of participants felt unprepared to face violence.
Although the study didn’t analyze the reasons workers felt this way, Dr. Kashyap speculates that it could be related to the medical distrust that grew during the pandemic or the stress patients and health care professionals experienced during its peak.
Regardless, the researchers say their study is a starting point. Now that the trend has been highlighted, it may be acted on.
Moving forward, Dr. Kashyap believes that controlling for different variables could determine whether factors like gender or shift time put a worker at higher risk for violence. He hopes it’s possible to interrupt these patterns and reestablish trust in the hospital environment. “It’s aspirational, but you’re hoping that through studies like ViSHWaS, which means trust in Hindi ... [we could restore] the trust and confidence among health care providers for the patients and family members.”
A version of this article first appeared on Medscape.com.
Feds charge 25 nursing school execs, staff in fake diploma scheme
The U.S. Department of Justice recently announced charges against 25 owners, operators, and employees of three Florida nursing schools in a fraud scheme in which they sold as many as 7,600 fake nursing degrees.
The purchasers in the diploma scheme paid $10,000 to $15,000 for degrees and transcripts and some 2,800 of the buyers passed the national nursing licensing exam to become registered nurses (RNs) and licensed practice nurses/vocational nurses (LPN/VNs) around the country, according to The New York Times.
Many of the degree recipients went on to work at hospitals, nursing homes, and Veterans Affairs medical centers, according to the U.S. Attorney’s Office for the Southern District of Florida.
Several national nursing organizations cooperated with the investigation, and the Delaware Division of Professional Regulation already annulled 26 licenses, according to the Delaware Nurses Association. Fake licenses were issued in five states, according to federal reports.
“We are deeply unsettled by this egregious act,” DNA President Stephanie McClellan, MSN, RN, CMSRN, said in the group’s press statement. “We want all Delaware nurses to be aware of this active issue and to speak up if there is a concern regarding capacity to practice safely by a colleague/peer,” she said.
The Oregon State Board of Nursing is also investigating at least a dozen nurses who may have paid for their degrees, according to a Portland CBS affiliate.
The National Council of State Boards of Nursing said in a statement that it had helped authorities identify and monitor the individuals who allegedly provided the false degrees.
Nursing community reacts
News of the fraud scheme spread through the nursing community, including social media. “The recent report on falsified nursing school degrees is both heartbreaking and serves as an eye-opener,” tweeted Usha Menon, PhD, RN, FAAN, dean and health professor of the University of South Florida Health College of Nursing. “There was enough of a need that prompted these bad actors to develop a scheme that could’ve endangered dozens of lives.”
Jennifer Mensik Kennedy, PhD, MBA, RN, the new president of the American Nurses Association, also weighed in. “The accusation that personnel at once-accredited nursing schools allegedly participated in this scheme is simply deplorable. These unlawful and unethical acts disparage the reputation of actual nurses everywhere who have rightfully earned [their titles] through their education, hard work, dedication, and time.”
The false degrees and transcripts were issued by three once-accredited and now-shuttered nursing schools in South Florida: Palm Beach School of Nursing, Sacred Heart International Institute, and Sienna College.
The alleged co-conspirators reportedly made $114 million from the scheme, which dates back to 2016, according to several news reports. Each defendant faces up to 20 years in prison.
Most LPN programs charge $10,000 to $15,000 to complete a program, Robert Rosseter, a spokesperson for the American Association of Colleges of Nursing (AACN), told this news organization.
None were AACN members, and none were accredited by the Commission on Collegiate Nursing Education, which is AACN’s autonomous accrediting agency, Mr. Rosseter said. AACN membership is voluntary and is open to schools offering baccalaureate or higher degrees, he explained.
“What is disturbing about this investigation is that there are over 7,600 people around the country with fraudulent nursing credentials who are potentially in critical health care roles treating patients,” Chad Yarbrough, acting special agent in charge for the FBI in Miami, said in the federal justice department release.
‘Operation Nightingale’ based on tip
The federal action, dubbed “Operation Nightingale” after the nursing pioneer Florence Nightingale, began in 2019. It was based on a tip related to a case in Maryland, according to Nurse.org.
That case ensnared Palm Beach School of Nursing owner Johanah Napoleon, who reportedly was selling fake degrees for $6,000 to $18,000 each to two individuals in Maryland and Virginia. Ms. Napoleon was charged in 2021 and eventually pled guilty. The Florida Board of Nursing shut down the Palm Beach school in 2017 owing to its students’ low passing rate on the national licensing exam.
Two participants in the bigger scheme who had also worked with Ms. Napoleon – Geralda Adrien and Woosvelt Predestin – were indicted in 2021. Ms. Adrien owned private education companies for people who at aspired to be nurses, and Mr. Predestin was an employee. They were sentenced to 27 months in prison last year and helped the federal officials build the larger case.
The 25 individuals who were charged Jan. 25 operated in Delaware, New York, New Jersey, Texas, and Florida.
Schemes lured immigrants
In the scheme involving Siena College, some of the individuals acted as recruiters to direct nurses who were looking for employment to the school, where they allegedly would then pay for an RN or LPN/VN degree. The recipients of the false documents then used them to obtain jobs, including at a hospital in Georgia and a Veterans Affairs medical center in Maryland, according to one indictment. The president of Siena and her co-conspirators sold more than 2,000 fake diplomas, according to charging documents.
At the Palm Beach College of Nursing, individuals at various nursing prep and education programs allegedly helped others obtain fake degrees and transcripts, which were then used to pass RN and LPN/VN licensing exams in states that included Massachusetts, New Jersey, New York, and Ohio, according to the indictment.
Some individuals then secured employment with a nursing home in Ohio, a home health agency for pediatric patients in Massachusetts, and skilled nursing facilities in New York and New Jersey.
Prosecutors allege that the president of Sacred Heart International Institute and two other co-conspirators sold 588 fake diplomas.
The FBI said that some of the aspiring nurses who were talked into buying the degrees were LPNs who wanted to become RNs and that most of those lured into the scheme were from South Florida’s Haitian American immigrant community, Nurse.org reported.
A version of this article first appeared on Medscape.com.
The U.S. Department of Justice recently announced charges against 25 owners, operators, and employees of three Florida nursing schools in a fraud scheme in which they sold as many as 7,600 fake nursing degrees.
The purchasers in the diploma scheme paid $10,000 to $15,000 for degrees and transcripts and some 2,800 of the buyers passed the national nursing licensing exam to become registered nurses (RNs) and licensed practice nurses/vocational nurses (LPN/VNs) around the country, according to The New York Times.
Many of the degree recipients went on to work at hospitals, nursing homes, and Veterans Affairs medical centers, according to the U.S. Attorney’s Office for the Southern District of Florida.
Several national nursing organizations cooperated with the investigation, and the Delaware Division of Professional Regulation already annulled 26 licenses, according to the Delaware Nurses Association. Fake licenses were issued in five states, according to federal reports.
“We are deeply unsettled by this egregious act,” DNA President Stephanie McClellan, MSN, RN, CMSRN, said in the group’s press statement. “We want all Delaware nurses to be aware of this active issue and to speak up if there is a concern regarding capacity to practice safely by a colleague/peer,” she said.
The Oregon State Board of Nursing is also investigating at least a dozen nurses who may have paid for their degrees, according to a Portland CBS affiliate.
The National Council of State Boards of Nursing said in a statement that it had helped authorities identify and monitor the individuals who allegedly provided the false degrees.
Nursing community reacts
News of the fraud scheme spread through the nursing community, including social media. “The recent report on falsified nursing school degrees is both heartbreaking and serves as an eye-opener,” tweeted Usha Menon, PhD, RN, FAAN, dean and health professor of the University of South Florida Health College of Nursing. “There was enough of a need that prompted these bad actors to develop a scheme that could’ve endangered dozens of lives.”
Jennifer Mensik Kennedy, PhD, MBA, RN, the new president of the American Nurses Association, also weighed in. “The accusation that personnel at once-accredited nursing schools allegedly participated in this scheme is simply deplorable. These unlawful and unethical acts disparage the reputation of actual nurses everywhere who have rightfully earned [their titles] through their education, hard work, dedication, and time.”
The false degrees and transcripts were issued by three once-accredited and now-shuttered nursing schools in South Florida: Palm Beach School of Nursing, Sacred Heart International Institute, and Sienna College.
The alleged co-conspirators reportedly made $114 million from the scheme, which dates back to 2016, according to several news reports. Each defendant faces up to 20 years in prison.
Most LPN programs charge $10,000 to $15,000 to complete a program, Robert Rosseter, a spokesperson for the American Association of Colleges of Nursing (AACN), told this news organization.
None were AACN members, and none were accredited by the Commission on Collegiate Nursing Education, which is AACN’s autonomous accrediting agency, Mr. Rosseter said. AACN membership is voluntary and is open to schools offering baccalaureate or higher degrees, he explained.
“What is disturbing about this investigation is that there are over 7,600 people around the country with fraudulent nursing credentials who are potentially in critical health care roles treating patients,” Chad Yarbrough, acting special agent in charge for the FBI in Miami, said in the federal justice department release.
‘Operation Nightingale’ based on tip
The federal action, dubbed “Operation Nightingale” after the nursing pioneer Florence Nightingale, began in 2019. It was based on a tip related to a case in Maryland, according to Nurse.org.
That case ensnared Palm Beach School of Nursing owner Johanah Napoleon, who reportedly was selling fake degrees for $6,000 to $18,000 each to two individuals in Maryland and Virginia. Ms. Napoleon was charged in 2021 and eventually pled guilty. The Florida Board of Nursing shut down the Palm Beach school in 2017 owing to its students’ low passing rate on the national licensing exam.
Two participants in the bigger scheme who had also worked with Ms. Napoleon – Geralda Adrien and Woosvelt Predestin – were indicted in 2021. Ms. Adrien owned private education companies for people who at aspired to be nurses, and Mr. Predestin was an employee. They were sentenced to 27 months in prison last year and helped the federal officials build the larger case.
The 25 individuals who were charged Jan. 25 operated in Delaware, New York, New Jersey, Texas, and Florida.
Schemes lured immigrants
In the scheme involving Siena College, some of the individuals acted as recruiters to direct nurses who were looking for employment to the school, where they allegedly would then pay for an RN or LPN/VN degree. The recipients of the false documents then used them to obtain jobs, including at a hospital in Georgia and a Veterans Affairs medical center in Maryland, according to one indictment. The president of Siena and her co-conspirators sold more than 2,000 fake diplomas, according to charging documents.
At the Palm Beach College of Nursing, individuals at various nursing prep and education programs allegedly helped others obtain fake degrees and transcripts, which were then used to pass RN and LPN/VN licensing exams in states that included Massachusetts, New Jersey, New York, and Ohio, according to the indictment.
Some individuals then secured employment with a nursing home in Ohio, a home health agency for pediatric patients in Massachusetts, and skilled nursing facilities in New York and New Jersey.
Prosecutors allege that the president of Sacred Heart International Institute and two other co-conspirators sold 588 fake diplomas.
The FBI said that some of the aspiring nurses who were talked into buying the degrees were LPNs who wanted to become RNs and that most of those lured into the scheme were from South Florida’s Haitian American immigrant community, Nurse.org reported.
A version of this article first appeared on Medscape.com.
The U.S. Department of Justice recently announced charges against 25 owners, operators, and employees of three Florida nursing schools in a fraud scheme in which they sold as many as 7,600 fake nursing degrees.
The purchasers in the diploma scheme paid $10,000 to $15,000 for degrees and transcripts and some 2,800 of the buyers passed the national nursing licensing exam to become registered nurses (RNs) and licensed practice nurses/vocational nurses (LPN/VNs) around the country, according to The New York Times.
Many of the degree recipients went on to work at hospitals, nursing homes, and Veterans Affairs medical centers, according to the U.S. Attorney’s Office for the Southern District of Florida.
Several national nursing organizations cooperated with the investigation, and the Delaware Division of Professional Regulation already annulled 26 licenses, according to the Delaware Nurses Association. Fake licenses were issued in five states, according to federal reports.
“We are deeply unsettled by this egregious act,” DNA President Stephanie McClellan, MSN, RN, CMSRN, said in the group’s press statement. “We want all Delaware nurses to be aware of this active issue and to speak up if there is a concern regarding capacity to practice safely by a colleague/peer,” she said.
The Oregon State Board of Nursing is also investigating at least a dozen nurses who may have paid for their degrees, according to a Portland CBS affiliate.
The National Council of State Boards of Nursing said in a statement that it had helped authorities identify and monitor the individuals who allegedly provided the false degrees.
Nursing community reacts
News of the fraud scheme spread through the nursing community, including social media. “The recent report on falsified nursing school degrees is both heartbreaking and serves as an eye-opener,” tweeted Usha Menon, PhD, RN, FAAN, dean and health professor of the University of South Florida Health College of Nursing. “There was enough of a need that prompted these bad actors to develop a scheme that could’ve endangered dozens of lives.”
Jennifer Mensik Kennedy, PhD, MBA, RN, the new president of the American Nurses Association, also weighed in. “The accusation that personnel at once-accredited nursing schools allegedly participated in this scheme is simply deplorable. These unlawful and unethical acts disparage the reputation of actual nurses everywhere who have rightfully earned [their titles] through their education, hard work, dedication, and time.”
The false degrees and transcripts were issued by three once-accredited and now-shuttered nursing schools in South Florida: Palm Beach School of Nursing, Sacred Heart International Institute, and Sienna College.
The alleged co-conspirators reportedly made $114 million from the scheme, which dates back to 2016, according to several news reports. Each defendant faces up to 20 years in prison.
Most LPN programs charge $10,000 to $15,000 to complete a program, Robert Rosseter, a spokesperson for the American Association of Colleges of Nursing (AACN), told this news organization.
None were AACN members, and none were accredited by the Commission on Collegiate Nursing Education, which is AACN’s autonomous accrediting agency, Mr. Rosseter said. AACN membership is voluntary and is open to schools offering baccalaureate or higher degrees, he explained.
“What is disturbing about this investigation is that there are over 7,600 people around the country with fraudulent nursing credentials who are potentially in critical health care roles treating patients,” Chad Yarbrough, acting special agent in charge for the FBI in Miami, said in the federal justice department release.
‘Operation Nightingale’ based on tip
The federal action, dubbed “Operation Nightingale” after the nursing pioneer Florence Nightingale, began in 2019. It was based on a tip related to a case in Maryland, according to Nurse.org.
That case ensnared Palm Beach School of Nursing owner Johanah Napoleon, who reportedly was selling fake degrees for $6,000 to $18,000 each to two individuals in Maryland and Virginia. Ms. Napoleon was charged in 2021 and eventually pled guilty. The Florida Board of Nursing shut down the Palm Beach school in 2017 owing to its students’ low passing rate on the national licensing exam.
Two participants in the bigger scheme who had also worked with Ms. Napoleon – Geralda Adrien and Woosvelt Predestin – were indicted in 2021. Ms. Adrien owned private education companies for people who at aspired to be nurses, and Mr. Predestin was an employee. They were sentenced to 27 months in prison last year and helped the federal officials build the larger case.
The 25 individuals who were charged Jan. 25 operated in Delaware, New York, New Jersey, Texas, and Florida.
Schemes lured immigrants
In the scheme involving Siena College, some of the individuals acted as recruiters to direct nurses who were looking for employment to the school, where they allegedly would then pay for an RN or LPN/VN degree. The recipients of the false documents then used them to obtain jobs, including at a hospital in Georgia and a Veterans Affairs medical center in Maryland, according to one indictment. The president of Siena and her co-conspirators sold more than 2,000 fake diplomas, according to charging documents.
At the Palm Beach College of Nursing, individuals at various nursing prep and education programs allegedly helped others obtain fake degrees and transcripts, which were then used to pass RN and LPN/VN licensing exams in states that included Massachusetts, New Jersey, New York, and Ohio, according to the indictment.
Some individuals then secured employment with a nursing home in Ohio, a home health agency for pediatric patients in Massachusetts, and skilled nursing facilities in New York and New Jersey.
Prosecutors allege that the president of Sacred Heart International Institute and two other co-conspirators sold 588 fake diplomas.
The FBI said that some of the aspiring nurses who were talked into buying the degrees were LPNs who wanted to become RNs and that most of those lured into the scheme were from South Florida’s Haitian American immigrant community, Nurse.org reported.
A version of this article first appeared on Medscape.com.
Biden to end COVID emergencies in May
Doing so will have many effects, including the end of free vaccines and health services to fight the pandemic. The public health emergency has been renewed every 90 days since it was declared by the Trump administration in January 2020.
The declaration allowed major changes throughout the health care system to deal with the pandemic, including the free distribution of vaccines, testing, and treatments. In addition, telehealth services were expanded, and Medicaid and the Children’s Health Insurance Program were extended to millions more Americans.
Biden said the COVID-19 national emergency is set to expire March 1 while the declared public health emergency would currently expire on April 11. The president said both will be extended to end May 11.
There were nearly 300,000 newly reported COVID-19 cases in the United States for the week ending Jan. 25, according to CDC data, as well as more than 3,750 deaths.
A version of this article first appeared on WebMD.com.
Doing so will have many effects, including the end of free vaccines and health services to fight the pandemic. The public health emergency has been renewed every 90 days since it was declared by the Trump administration in January 2020.
The declaration allowed major changes throughout the health care system to deal with the pandemic, including the free distribution of vaccines, testing, and treatments. In addition, telehealth services were expanded, and Medicaid and the Children’s Health Insurance Program were extended to millions more Americans.
Biden said the COVID-19 national emergency is set to expire March 1 while the declared public health emergency would currently expire on April 11. The president said both will be extended to end May 11.
There were nearly 300,000 newly reported COVID-19 cases in the United States for the week ending Jan. 25, according to CDC data, as well as more than 3,750 deaths.
A version of this article first appeared on WebMD.com.
Doing so will have many effects, including the end of free vaccines and health services to fight the pandemic. The public health emergency has been renewed every 90 days since it was declared by the Trump administration in January 2020.
The declaration allowed major changes throughout the health care system to deal with the pandemic, including the free distribution of vaccines, testing, and treatments. In addition, telehealth services were expanded, and Medicaid and the Children’s Health Insurance Program were extended to millions more Americans.
Biden said the COVID-19 national emergency is set to expire March 1 while the declared public health emergency would currently expire on April 11. The president said both will be extended to end May 11.
There were nearly 300,000 newly reported COVID-19 cases in the United States for the week ending Jan. 25, according to CDC data, as well as more than 3,750 deaths.
A version of this article first appeared on WebMD.com.
Six healthy lifestyle habits linked to slowed memory decline
Investigators found that a healthy diet, cognitive activity, regular physical exercise, not smoking, and abstaining from alcohol were significantly linked to slowed cognitive decline irrespective of APOE4 status.
After adjusting for health and socioeconomic factors, investigators found that each individual healthy behavior was associated with a slower-than-average decline in memory over a decade. A healthy diet emerged as the strongest deterrent, followed by cognitive activity and physical exercise.
“A healthy lifestyle is associated with slower memory decline, even in the presence of the APOE4 allele,” study investigators led by Jianping Jia, MD, PhD, of the Innovation Center for Neurological Disorders and the department of neurology, Xuan Wu Hospital, Capital Medical University, Beijing, write.
“This study might offer important information to protect older adults against memory decline,” they add.
The study was published online in the BMJ.
Preventing memory decline
Memory “continuously declines as people age,” but age-related memory decline is not necessarily a prodrome of dementia and can “merely be senescent forgetfulness,” the investigators note. This can be “reversed or [can] become stable,” instead of progressing to a pathologic state.
Factors affecting memory include aging, APOE4 genotype, chronic diseases, and lifestyle patterns, with lifestyle “receiving increasing attention as a modifiable behavior.”
Nevertheless, few studies have focused on the impact of lifestyle on memory, and those that have are mostly cross-sectional and also “did not consider the interaction between a healthy lifestyle and genetic risk,” the researchers note.
To investigate, the researchers conducted a longitudinal study, known as the China Cognition and Aging Study, that considered genetic risk as well as lifestyle factors.
The study began in 2009 and concluded in 2019. Participants were evaluated and underwent neuropsychological testing in 2012, 2014, 2016, and at the study’s conclusion.
Participants (n = 29,072; mean [SD] age, 72.23 [6.61] years; 48.54% women; 20.43% APOE4 carriers) were required to have normal cognitive function at baseline. Data on those whose condition progressed to mild cognitive impairment (MCI) or dementia during the follow-up period were excluded after their diagnosis.
The Mini–Mental State Examination was used to assess global cognitive function. Memory function was assessed using the World Health Organization/University of California, Los Angeles Auditory Verbal Learning Test.
“Lifestyle” consisted of six modifiable factors: physical exercise (weekly frequency and total time), smoking (current, former, or never-smokers), alcohol consumption (never drank, drank occasionally, low to excess drinking, and heavy drinking), diet (daily intake of 12 food items: fruits, vegetables, fish, meat, dairy products, salt, oil, eggs, cereals, legumes, nuts, tea), cognitive activity (writing, reading, playing cards, mahjong, other games), and social contact (participating in meetings, attending parties, visiting friends/relatives, traveling, chatting online).
Participants’ lifestyles were scored on the basis of the number of healthy factors they engaged in.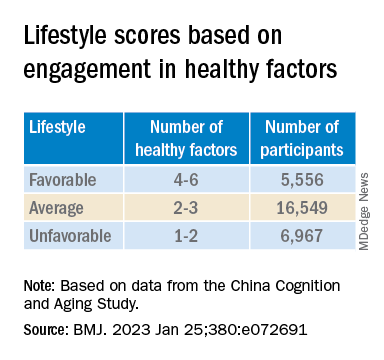
Participants were also stratified by APOE genotype into APOE4 carriers and noncarriers.
Demographic and other items of health information, including the presence of medical illness, were used as covariates. The researchers also included the “learning effect of each participant as a covariate, due to repeated cognitive assessments.”
Important for public health
During the 10-year period, 7,164 participants died, and 3,567 stopped participating.
Participants in the favorable and average groups showed slower memory decline per increased year of age (0.007 [0.005-0.009], P < .001; and 0.002 [0 .000-0.003], P = .033 points higher, respectively), compared with those in the unfavorable group.
Healthy diet had the strongest protective effect on memory.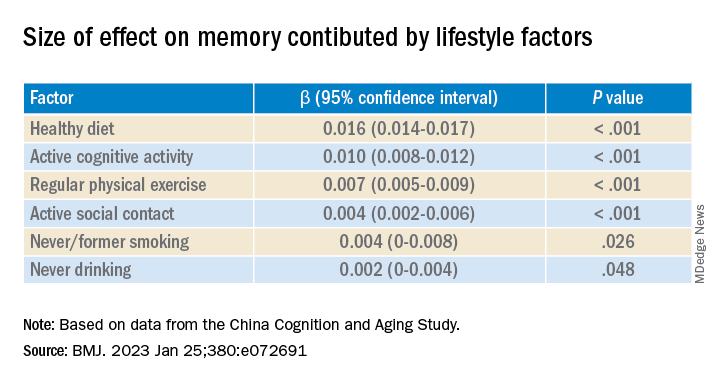
Memory decline occurred faster in APOE4 vesus non-APOE4 carriers (0.002 points/year [95% confidence interval, 0.001-0.003]; P = .007).
But APOE4 carriers with favorable and average lifestyles showed slower memory decline (0.027 [0.023-0.031] and 0.014 [0.010-0.019], respectively), compared with those with unfavorable lifestyles. Similar findings were obtained in non-APOE4 carriers.
Those with favorable or average lifestyle were respectively almost 90% and 30% less likely to develop dementia or MCI, compared with those with an unfavorable lifestyle.
The authors acknowledge the study’s limitations, including its observational design and the potential for measurement errors, owing to self-reporting of lifestyle factors. Additionally, some participants did not return for follow-up evaluations, leading to potential selection bias.
Nevertheless, the findings “might offer important information for public health to protect older [people] against memory decline,” they note – especially since the study “provides evidence that these effects also include individuals with the APOE4 allele.”
‘Important, encouraging’ research
In a comment, Severine Sabia, PhD, a senior researcher at the Université Paris Cité, INSERM Institut National de la Santé et de la Recherche Medicalé, France, called the findings “important and encouraging.”
However, said Dr. Sabia, who was not involved with the study, “there remain important research questions that need to be investigated in order to identify key behaviors: which combination, the cutoff of risk, and when to intervene.”
Future research on prevention “should examine a wider range of possible risk factors” and should also “identify specific exposures associated with the greatest risk, while also considering the risk threshold and age at exposure for each one.”
In an accompanying editorial, Dr. Sabia and co-author Archana Singh-Manoux, PhD, note that the risk of cognitive decline and dementia are probably determined by multiple factors.
They liken it to the “multifactorial risk paradigm introduced by the Framingham study,” which has “led to a substantial reduction in cardiovascular disease.” A similar approach could be used with dementia prevention, they suggest.
The authors received support from the Xuanwu Hospital of Capital Medical University for the submitted work. One of the authors received a grant from the French National Research Agency. The other authors have disclosed no relevant financial relationships. Dr. Sabia received grant funding from the French National Research Agency. Dr. Singh-Manoux received grants from the National Institute on Aging of the National Institutes of Health.
A version of this article first appeared on Medscape.com.
Investigators found that a healthy diet, cognitive activity, regular physical exercise, not smoking, and abstaining from alcohol were significantly linked to slowed cognitive decline irrespective of APOE4 status.
After adjusting for health and socioeconomic factors, investigators found that each individual healthy behavior was associated with a slower-than-average decline in memory over a decade. A healthy diet emerged as the strongest deterrent, followed by cognitive activity and physical exercise.
“A healthy lifestyle is associated with slower memory decline, even in the presence of the APOE4 allele,” study investigators led by Jianping Jia, MD, PhD, of the Innovation Center for Neurological Disorders and the department of neurology, Xuan Wu Hospital, Capital Medical University, Beijing, write.
“This study might offer important information to protect older adults against memory decline,” they add.
The study was published online in the BMJ.
Preventing memory decline
Memory “continuously declines as people age,” but age-related memory decline is not necessarily a prodrome of dementia and can “merely be senescent forgetfulness,” the investigators note. This can be “reversed or [can] become stable,” instead of progressing to a pathologic state.
Factors affecting memory include aging, APOE4 genotype, chronic diseases, and lifestyle patterns, with lifestyle “receiving increasing attention as a modifiable behavior.”
Nevertheless, few studies have focused on the impact of lifestyle on memory, and those that have are mostly cross-sectional and also “did not consider the interaction between a healthy lifestyle and genetic risk,” the researchers note.
To investigate, the researchers conducted a longitudinal study, known as the China Cognition and Aging Study, that considered genetic risk as well as lifestyle factors.
The study began in 2009 and concluded in 2019. Participants were evaluated and underwent neuropsychological testing in 2012, 2014, 2016, and at the study’s conclusion.
Participants (n = 29,072; mean [SD] age, 72.23 [6.61] years; 48.54% women; 20.43% APOE4 carriers) were required to have normal cognitive function at baseline. Data on those whose condition progressed to mild cognitive impairment (MCI) or dementia during the follow-up period were excluded after their diagnosis.
The Mini–Mental State Examination was used to assess global cognitive function. Memory function was assessed using the World Health Organization/University of California, Los Angeles Auditory Verbal Learning Test.
“Lifestyle” consisted of six modifiable factors: physical exercise (weekly frequency and total time), smoking (current, former, or never-smokers), alcohol consumption (never drank, drank occasionally, low to excess drinking, and heavy drinking), diet (daily intake of 12 food items: fruits, vegetables, fish, meat, dairy products, salt, oil, eggs, cereals, legumes, nuts, tea), cognitive activity (writing, reading, playing cards, mahjong, other games), and social contact (participating in meetings, attending parties, visiting friends/relatives, traveling, chatting online).
Participants’ lifestyles were scored on the basis of the number of healthy factors they engaged in.
Participants were also stratified by APOE genotype into APOE4 carriers and noncarriers.
Demographic and other items of health information, including the presence of medical illness, were used as covariates. The researchers also included the “learning effect of each participant as a covariate, due to repeated cognitive assessments.”
Important for public health
During the 10-year period, 7,164 participants died, and 3,567 stopped participating.
Participants in the favorable and average groups showed slower memory decline per increased year of age (0.007 [0.005-0.009], P < .001; and 0.002 [0 .000-0.003], P = .033 points higher, respectively), compared with those in the unfavorable group.
Healthy diet had the strongest protective effect on memory.
Memory decline occurred faster in APOE4 vesus non-APOE4 carriers (0.002 points/year [95% confidence interval, 0.001-0.003]; P = .007).
But APOE4 carriers with favorable and average lifestyles showed slower memory decline (0.027 [0.023-0.031] and 0.014 [0.010-0.019], respectively), compared with those with unfavorable lifestyles. Similar findings were obtained in non-APOE4 carriers.
Those with favorable or average lifestyle were respectively almost 90% and 30% less likely to develop dementia or MCI, compared with those with an unfavorable lifestyle.
The authors acknowledge the study’s limitations, including its observational design and the potential for measurement errors, owing to self-reporting of lifestyle factors. Additionally, some participants did not return for follow-up evaluations, leading to potential selection bias.
Nevertheless, the findings “might offer important information for public health to protect older [people] against memory decline,” they note – especially since the study “provides evidence that these effects also include individuals with the APOE4 allele.”
‘Important, encouraging’ research
In a comment, Severine Sabia, PhD, a senior researcher at the Université Paris Cité, INSERM Institut National de la Santé et de la Recherche Medicalé, France, called the findings “important and encouraging.”
However, said Dr. Sabia, who was not involved with the study, “there remain important research questions that need to be investigated in order to identify key behaviors: which combination, the cutoff of risk, and when to intervene.”
Future research on prevention “should examine a wider range of possible risk factors” and should also “identify specific exposures associated with the greatest risk, while also considering the risk threshold and age at exposure for each one.”
In an accompanying editorial, Dr. Sabia and co-author Archana Singh-Manoux, PhD, note that the risk of cognitive decline and dementia are probably determined by multiple factors.
They liken it to the “multifactorial risk paradigm introduced by the Framingham study,” which has “led to a substantial reduction in cardiovascular disease.” A similar approach could be used with dementia prevention, they suggest.
The authors received support from the Xuanwu Hospital of Capital Medical University for the submitted work. One of the authors received a grant from the French National Research Agency. The other authors have disclosed no relevant financial relationships. Dr. Sabia received grant funding from the French National Research Agency. Dr. Singh-Manoux received grants from the National Institute on Aging of the National Institutes of Health.
A version of this article first appeared on Medscape.com.
Investigators found that a healthy diet, cognitive activity, regular physical exercise, not smoking, and abstaining from alcohol were significantly linked to slowed cognitive decline irrespective of APOE4 status.
After adjusting for health and socioeconomic factors, investigators found that each individual healthy behavior was associated with a slower-than-average decline in memory over a decade. A healthy diet emerged as the strongest deterrent, followed by cognitive activity and physical exercise.
“A healthy lifestyle is associated with slower memory decline, even in the presence of the APOE4 allele,” study investigators led by Jianping Jia, MD, PhD, of the Innovation Center for Neurological Disorders and the department of neurology, Xuan Wu Hospital, Capital Medical University, Beijing, write.
“This study might offer important information to protect older adults against memory decline,” they add.
The study was published online in the BMJ.
Preventing memory decline
Memory “continuously declines as people age,” but age-related memory decline is not necessarily a prodrome of dementia and can “merely be senescent forgetfulness,” the investigators note. This can be “reversed or [can] become stable,” instead of progressing to a pathologic state.
Factors affecting memory include aging, APOE4 genotype, chronic diseases, and lifestyle patterns, with lifestyle “receiving increasing attention as a modifiable behavior.”
Nevertheless, few studies have focused on the impact of lifestyle on memory, and those that have are mostly cross-sectional and also “did not consider the interaction between a healthy lifestyle and genetic risk,” the researchers note.
To investigate, the researchers conducted a longitudinal study, known as the China Cognition and Aging Study, that considered genetic risk as well as lifestyle factors.
The study began in 2009 and concluded in 2019. Participants were evaluated and underwent neuropsychological testing in 2012, 2014, 2016, and at the study’s conclusion.
Participants (n = 29,072; mean [SD] age, 72.23 [6.61] years; 48.54% women; 20.43% APOE4 carriers) were required to have normal cognitive function at baseline. Data on those whose condition progressed to mild cognitive impairment (MCI) or dementia during the follow-up period were excluded after their diagnosis.
The Mini–Mental State Examination was used to assess global cognitive function. Memory function was assessed using the World Health Organization/University of California, Los Angeles Auditory Verbal Learning Test.
“Lifestyle” consisted of six modifiable factors: physical exercise (weekly frequency and total time), smoking (current, former, or never-smokers), alcohol consumption (never drank, drank occasionally, low to excess drinking, and heavy drinking), diet (daily intake of 12 food items: fruits, vegetables, fish, meat, dairy products, salt, oil, eggs, cereals, legumes, nuts, tea), cognitive activity (writing, reading, playing cards, mahjong, other games), and social contact (participating in meetings, attending parties, visiting friends/relatives, traveling, chatting online).
Participants’ lifestyles were scored on the basis of the number of healthy factors they engaged in.
Participants were also stratified by APOE genotype into APOE4 carriers and noncarriers.
Demographic and other items of health information, including the presence of medical illness, were used as covariates. The researchers also included the “learning effect of each participant as a covariate, due to repeated cognitive assessments.”
Important for public health
During the 10-year period, 7,164 participants died, and 3,567 stopped participating.
Participants in the favorable and average groups showed slower memory decline per increased year of age (0.007 [0.005-0.009], P < .001; and 0.002 [0 .000-0.003], P = .033 points higher, respectively), compared with those in the unfavorable group.
Healthy diet had the strongest protective effect on memory.
Memory decline occurred faster in APOE4 vesus non-APOE4 carriers (0.002 points/year [95% confidence interval, 0.001-0.003]; P = .007).
But APOE4 carriers with favorable and average lifestyles showed slower memory decline (0.027 [0.023-0.031] and 0.014 [0.010-0.019], respectively), compared with those with unfavorable lifestyles. Similar findings were obtained in non-APOE4 carriers.
Those with favorable or average lifestyle were respectively almost 90% and 30% less likely to develop dementia or MCI, compared with those with an unfavorable lifestyle.
The authors acknowledge the study’s limitations, including its observational design and the potential for measurement errors, owing to self-reporting of lifestyle factors. Additionally, some participants did not return for follow-up evaluations, leading to potential selection bias.
Nevertheless, the findings “might offer important information for public health to protect older [people] against memory decline,” they note – especially since the study “provides evidence that these effects also include individuals with the APOE4 allele.”
‘Important, encouraging’ research
In a comment, Severine Sabia, PhD, a senior researcher at the Université Paris Cité, INSERM Institut National de la Santé et de la Recherche Medicalé, France, called the findings “important and encouraging.”
However, said Dr. Sabia, who was not involved with the study, “there remain important research questions that need to be investigated in order to identify key behaviors: which combination, the cutoff of risk, and when to intervene.”
Future research on prevention “should examine a wider range of possible risk factors” and should also “identify specific exposures associated with the greatest risk, while also considering the risk threshold and age at exposure for each one.”
In an accompanying editorial, Dr. Sabia and co-author Archana Singh-Manoux, PhD, note that the risk of cognitive decline and dementia are probably determined by multiple factors.
They liken it to the “multifactorial risk paradigm introduced by the Framingham study,” which has “led to a substantial reduction in cardiovascular disease.” A similar approach could be used with dementia prevention, they suggest.
The authors received support from the Xuanwu Hospital of Capital Medical University for the submitted work. One of the authors received a grant from the French National Research Agency. The other authors have disclosed no relevant financial relationships. Dr. Sabia received grant funding from the French National Research Agency. Dr. Singh-Manoux received grants from the National Institute on Aging of the National Institutes of Health.
A version of this article first appeared on Medscape.com.
FROM THE BMJ
New cancer data spark outcry from patient advocates
The American Cancer Society on Jan. 13 revealed what it called “alarming” news about prostate cancer: After 2 decades of decline, the number of men diagnosed with the disease in the United States rose by 15% from 2014 to 2019.
“Most concerning,” according to the group’s CEO Karen Knudsen, PhD, MBA, is that the increase is being driven by diagnoses of advanced disease.
“Since 2011, the diagnosis of advanced-stage (regional- or distant-stage) prostate cancer has increased by 4%-5% annually and the proportion of men diagnosed with distant-stage disease has doubled,” said Dr. Knudsen at a press conference concerning the figures. “These findings underscore the importance of understanding and reducing this trend.”
The increase, which works out to be an additional 99,000 cases of prostate cancer, did not take the ACS by surprise; the group has been predicting a jump in diagnoses of the disease, which is the most common cancer in men after skin cancer, and the second most common cause of cancer death for that group.
The ACS announced a new action plan, “Improving Mortality from Prostate Cancer Together” – or IMPACT – to address the rise, especially in Black men, and to curb the increasing rate of advanced, difficult-to-treat cases.
“We must address these shifts in prostate cancer, especially in the Black community, since the incidence of prostate cancer in Black men is 70% higher than in White men and prostate cancer mortality rates in Black men are approximately two to four times higher than those in every other racial and ethnic group,” William Dahut, MD, PhD, chief scientific officer for the ACS, said at the press conference.
A study published in JAMA Network Open challenged that claim, finding that, after controlling for socioeconomic factors, race does not appear to be a significant predictor of mortality for prostate cancer.
Dr. Dahut said in an interview that IMPACT “is still [in the] early days for this initiative and more details will be coming out soon.”
Charles Ryan, MD, CEO of the Prostate Cancer Foundation, the world’s largest prostate cancer research charity, called IMPACT “extremely important work. Highlighting the disparities can only serve to benefit all men with prostate cancer, especially Black men.”
Bold action ... or passivity?
Overall cancer mortality has dropped 33% since 1991, averting an estimated 3.8 million deaths, according to ACS. But the story for prostate cancer is different.
The society and advocates had warned as recently as 2 years ago that prostate cancer was poised to rise again, especially advanced cases that may be too late to treat.
Leaders in the prostate cancer advocacy community praised the ACS plan for IMPACT, but some expressed frustration over what they said was ACS’ passivity in the face of long-anticipated increases in cases of the disease.
“I think prostate cancer was not high on their agenda,” said Rick Davis, founder of AnCan, which offers several support groups for patients with prostate cancer. “It’s good to see ACS get back into the prostate cancer game.”
Mr. Davis and patient advocate Darryl Mitteldorf, LCSW, founder of Malecare, another prostate support organization, said ACS dropped patient services for prostate cancer patients a decade ago and has not been a vocal supporter of screening for levels of prostate-specific antigen (PSA) to detect prostate cancer early.
“Early detection is supposed to be their goal,” Mr. Davis said.
In 2012, the U.S. Preventive Services Task Force recommended against PSA screening, giving it a D-rating. The move prompted attacks on the task force from most advocates and many urologists.
Following this criticism, the task force recommended shared decision-making between patient and doctor, while giving PSA screening a C-rating. Now, the ACS recommends men in general at age 50 discuss prostate cancer screening with their doctor and that Black men do the same at age 45.
Mr. Mitteldorf said ACS “owes prostate cancer patients an explanation and analysis of its response to the USPTF’s downgrade of PSA testing and how that response might be related to death and instance rates.”
Mr. Mitteldorf added that male patients lost key support from ACS when the group dismantled its Man to Man group for prostate cancer patients and its Brother to Brother group for Blacks in particular.
Dr. Dahut said Man to Man “sunsetted” and was turned over to any local organization that chose to offer it. He said longtime staff didn’t have “a lot of information about [the demise of] Brother to Brother.”
For Mr. Davis, those smaller cuts add up to a much larger insult.
“Today, in 2023, ACS continues to poke a finger in the eyes of prostate cancer patients,” he said. “Since 2010, they have not given us any respect. ACS dumped its support.”
He pointed to the group’s funding priorities, noting that outlays for prostate cancer have consistently lagged behind those for breast cancer.
The ACS spent $25.3 million on breast cancer research and $6.7 million for prostate cancer in 2018, and in 2023 will designate $126.5 for breast cancer research and $43.9 million for prostate cancer.
ACS has earmarked $62 million this year for lung cancer programs and $61 million for colorectal cancer.
“Parity between breast cancer and prostate cancer would be a good start in sizing the IMPACT program,” Mr. Davis said. “After all, breast cancer and prostate cancer are hardly different in numbers today.”
Dr. Dahut denied any gender bias in research funding. He said the group makes funding decisions “based on finding the most impactful science regardless of tumor type. Our mission includes funding every cancer, every day; thus, we generally do not go into our funding cycle with any set-asides for a particular cancer.”
Mr. Davis also said the ACS data suggest the growing number of prostate cancer cases is even worse than the group has said. Although the society cites a 3% annual increase in prostate cancer diagnoses from 2014 to 2019, since 2019 the annual increase is a much more dramatic 16%. Meanwhile, the number of new cases of the disease is projected to rise from 175,000 per year in 2019 to 288,000 this year.
Dr. Dahut said the society used the 2014-2019 time frame for technical reasons, separating confirmed cases in the earlier period from estimated cases in recent years.
“We discourage comparing projected cases over time because these cases are model-based and subject to fluctuations,” Dr. Dahut said.
A version of this article originally appeared on Medscape.com.
The American Cancer Society on Jan. 13 revealed what it called “alarming” news about prostate cancer: After 2 decades of decline, the number of men diagnosed with the disease in the United States rose by 15% from 2014 to 2019.
“Most concerning,” according to the group’s CEO Karen Knudsen, PhD, MBA, is that the increase is being driven by diagnoses of advanced disease.
“Since 2011, the diagnosis of advanced-stage (regional- or distant-stage) prostate cancer has increased by 4%-5% annually and the proportion of men diagnosed with distant-stage disease has doubled,” said Dr. Knudsen at a press conference concerning the figures. “These findings underscore the importance of understanding and reducing this trend.”
The increase, which works out to be an additional 99,000 cases of prostate cancer, did not take the ACS by surprise; the group has been predicting a jump in diagnoses of the disease, which is the most common cancer in men after skin cancer, and the second most common cause of cancer death for that group.
The ACS announced a new action plan, “Improving Mortality from Prostate Cancer Together” – or IMPACT – to address the rise, especially in Black men, and to curb the increasing rate of advanced, difficult-to-treat cases.
“We must address these shifts in prostate cancer, especially in the Black community, since the incidence of prostate cancer in Black men is 70% higher than in White men and prostate cancer mortality rates in Black men are approximately two to four times higher than those in every other racial and ethnic group,” William Dahut, MD, PhD, chief scientific officer for the ACS, said at the press conference.
A study published in JAMA Network Open challenged that claim, finding that, after controlling for socioeconomic factors, race does not appear to be a significant predictor of mortality for prostate cancer.
Dr. Dahut said in an interview that IMPACT “is still [in the] early days for this initiative and more details will be coming out soon.”
Charles Ryan, MD, CEO of the Prostate Cancer Foundation, the world’s largest prostate cancer research charity, called IMPACT “extremely important work. Highlighting the disparities can only serve to benefit all men with prostate cancer, especially Black men.”
Bold action ... or passivity?
Overall cancer mortality has dropped 33% since 1991, averting an estimated 3.8 million deaths, according to ACS. But the story for prostate cancer is different.
The society and advocates had warned as recently as 2 years ago that prostate cancer was poised to rise again, especially advanced cases that may be too late to treat.
Leaders in the prostate cancer advocacy community praised the ACS plan for IMPACT, but some expressed frustration over what they said was ACS’ passivity in the face of long-anticipated increases in cases of the disease.
“I think prostate cancer was not high on their agenda,” said Rick Davis, founder of AnCan, which offers several support groups for patients with prostate cancer. “It’s good to see ACS get back into the prostate cancer game.”
Mr. Davis and patient advocate Darryl Mitteldorf, LCSW, founder of Malecare, another prostate support organization, said ACS dropped patient services for prostate cancer patients a decade ago and has not been a vocal supporter of screening for levels of prostate-specific antigen (PSA) to detect prostate cancer early.
“Early detection is supposed to be their goal,” Mr. Davis said.
In 2012, the U.S. Preventive Services Task Force recommended against PSA screening, giving it a D-rating. The move prompted attacks on the task force from most advocates and many urologists.
Following this criticism, the task force recommended shared decision-making between patient and doctor, while giving PSA screening a C-rating. Now, the ACS recommends men in general at age 50 discuss prostate cancer screening with their doctor and that Black men do the same at age 45.
Mr. Mitteldorf said ACS “owes prostate cancer patients an explanation and analysis of its response to the USPTF’s downgrade of PSA testing and how that response might be related to death and instance rates.”
Mr. Mitteldorf added that male patients lost key support from ACS when the group dismantled its Man to Man group for prostate cancer patients and its Brother to Brother group for Blacks in particular.
Dr. Dahut said Man to Man “sunsetted” and was turned over to any local organization that chose to offer it. He said longtime staff didn’t have “a lot of information about [the demise of] Brother to Brother.”
For Mr. Davis, those smaller cuts add up to a much larger insult.
“Today, in 2023, ACS continues to poke a finger in the eyes of prostate cancer patients,” he said. “Since 2010, they have not given us any respect. ACS dumped its support.”
He pointed to the group’s funding priorities, noting that outlays for prostate cancer have consistently lagged behind those for breast cancer.
The ACS spent $25.3 million on breast cancer research and $6.7 million for prostate cancer in 2018, and in 2023 will designate $126.5 for breast cancer research and $43.9 million for prostate cancer.
ACS has earmarked $62 million this year for lung cancer programs and $61 million for colorectal cancer.
“Parity between breast cancer and prostate cancer would be a good start in sizing the IMPACT program,” Mr. Davis said. “After all, breast cancer and prostate cancer are hardly different in numbers today.”
Dr. Dahut denied any gender bias in research funding. He said the group makes funding decisions “based on finding the most impactful science regardless of tumor type. Our mission includes funding every cancer, every day; thus, we generally do not go into our funding cycle with any set-asides for a particular cancer.”
Mr. Davis also said the ACS data suggest the growing number of prostate cancer cases is even worse than the group has said. Although the society cites a 3% annual increase in prostate cancer diagnoses from 2014 to 2019, since 2019 the annual increase is a much more dramatic 16%. Meanwhile, the number of new cases of the disease is projected to rise from 175,000 per year in 2019 to 288,000 this year.
Dr. Dahut said the society used the 2014-2019 time frame for technical reasons, separating confirmed cases in the earlier period from estimated cases in recent years.
“We discourage comparing projected cases over time because these cases are model-based and subject to fluctuations,” Dr. Dahut said.
A version of this article originally appeared on Medscape.com.
The American Cancer Society on Jan. 13 revealed what it called “alarming” news about prostate cancer: After 2 decades of decline, the number of men diagnosed with the disease in the United States rose by 15% from 2014 to 2019.
“Most concerning,” according to the group’s CEO Karen Knudsen, PhD, MBA, is that the increase is being driven by diagnoses of advanced disease.
“Since 2011, the diagnosis of advanced-stage (regional- or distant-stage) prostate cancer has increased by 4%-5% annually and the proportion of men diagnosed with distant-stage disease has doubled,” said Dr. Knudsen at a press conference concerning the figures. “These findings underscore the importance of understanding and reducing this trend.”
The increase, which works out to be an additional 99,000 cases of prostate cancer, did not take the ACS by surprise; the group has been predicting a jump in diagnoses of the disease, which is the most common cancer in men after skin cancer, and the second most common cause of cancer death for that group.
The ACS announced a new action plan, “Improving Mortality from Prostate Cancer Together” – or IMPACT – to address the rise, especially in Black men, and to curb the increasing rate of advanced, difficult-to-treat cases.
“We must address these shifts in prostate cancer, especially in the Black community, since the incidence of prostate cancer in Black men is 70% higher than in White men and prostate cancer mortality rates in Black men are approximately two to four times higher than those in every other racial and ethnic group,” William Dahut, MD, PhD, chief scientific officer for the ACS, said at the press conference.
A study published in JAMA Network Open challenged that claim, finding that, after controlling for socioeconomic factors, race does not appear to be a significant predictor of mortality for prostate cancer.
Dr. Dahut said in an interview that IMPACT “is still [in the] early days for this initiative and more details will be coming out soon.”
Charles Ryan, MD, CEO of the Prostate Cancer Foundation, the world’s largest prostate cancer research charity, called IMPACT “extremely important work. Highlighting the disparities can only serve to benefit all men with prostate cancer, especially Black men.”
Bold action ... or passivity?
Overall cancer mortality has dropped 33% since 1991, averting an estimated 3.8 million deaths, according to ACS. But the story for prostate cancer is different.
The society and advocates had warned as recently as 2 years ago that prostate cancer was poised to rise again, especially advanced cases that may be too late to treat.
Leaders in the prostate cancer advocacy community praised the ACS plan for IMPACT, but some expressed frustration over what they said was ACS’ passivity in the face of long-anticipated increases in cases of the disease.
“I think prostate cancer was not high on their agenda,” said Rick Davis, founder of AnCan, which offers several support groups for patients with prostate cancer. “It’s good to see ACS get back into the prostate cancer game.”
Mr. Davis and patient advocate Darryl Mitteldorf, LCSW, founder of Malecare, another prostate support organization, said ACS dropped patient services for prostate cancer patients a decade ago and has not been a vocal supporter of screening for levels of prostate-specific antigen (PSA) to detect prostate cancer early.
“Early detection is supposed to be their goal,” Mr. Davis said.
In 2012, the U.S. Preventive Services Task Force recommended against PSA screening, giving it a D-rating. The move prompted attacks on the task force from most advocates and many urologists.
Following this criticism, the task force recommended shared decision-making between patient and doctor, while giving PSA screening a C-rating. Now, the ACS recommends men in general at age 50 discuss prostate cancer screening with their doctor and that Black men do the same at age 45.
Mr. Mitteldorf said ACS “owes prostate cancer patients an explanation and analysis of its response to the USPTF’s downgrade of PSA testing and how that response might be related to death and instance rates.”
Mr. Mitteldorf added that male patients lost key support from ACS when the group dismantled its Man to Man group for prostate cancer patients and its Brother to Brother group for Blacks in particular.
Dr. Dahut said Man to Man “sunsetted” and was turned over to any local organization that chose to offer it. He said longtime staff didn’t have “a lot of information about [the demise of] Brother to Brother.”
For Mr. Davis, those smaller cuts add up to a much larger insult.
“Today, in 2023, ACS continues to poke a finger in the eyes of prostate cancer patients,” he said. “Since 2010, they have not given us any respect. ACS dumped its support.”
He pointed to the group’s funding priorities, noting that outlays for prostate cancer have consistently lagged behind those for breast cancer.
The ACS spent $25.3 million on breast cancer research and $6.7 million for prostate cancer in 2018, and in 2023 will designate $126.5 for breast cancer research and $43.9 million for prostate cancer.
ACS has earmarked $62 million this year for lung cancer programs and $61 million for colorectal cancer.
“Parity between breast cancer and prostate cancer would be a good start in sizing the IMPACT program,” Mr. Davis said. “After all, breast cancer and prostate cancer are hardly different in numbers today.”
Dr. Dahut denied any gender bias in research funding. He said the group makes funding decisions “based on finding the most impactful science regardless of tumor type. Our mission includes funding every cancer, every day; thus, we generally do not go into our funding cycle with any set-asides for a particular cancer.”
Mr. Davis also said the ACS data suggest the growing number of prostate cancer cases is even worse than the group has said. Although the society cites a 3% annual increase in prostate cancer diagnoses from 2014 to 2019, since 2019 the annual increase is a much more dramatic 16%. Meanwhile, the number of new cases of the disease is projected to rise from 175,000 per year in 2019 to 288,000 this year.
Dr. Dahut said the society used the 2014-2019 time frame for technical reasons, separating confirmed cases in the earlier period from estimated cases in recent years.
“We discourage comparing projected cases over time because these cases are model-based and subject to fluctuations,” Dr. Dahut said.
A version of this article originally appeared on Medscape.com.
Commentary: Early Diagnosis of PsA, February 2023
Appropriate assessment of MSK symptoms and signs by dermatologists may lead to more appropriate referral to rheumatologists. MSUS is being increasingly explored for early identification of PsA. A handheld, chip-based ultrasound device (HHUD) is a novel promising instrument that can be easily implemented in clinical practice. In a prospective study including 140 patients with psoriasis who presented to dermatologists with arthralgia. Grobelski and colleagues screened for PsA using medical history, CE, and the German Psoriasis Arthritis Diagnostic PsA screening questionnaire (GEPARD) paired with MSUS examination of up to three painful joints by trained dermatologists. Nineteen patients (13.6%) were diagnosed with PsA by rheumatologists. Interestingly, in 45 of the 46 patients the preliminary diagnosis of PsA was revised to "no PsA" after MSUS. The addition of MSUS changed the sensitivity and specificity of early PsA screening strategy from 88.2% and 54.4% to 70.6% and 90.4%, respectively. The positive predictive value increased to 56.5% from 25.4% after MSUS. Thus, the use of a quick MSUS using HHUD may lead to more accurate referral to rheumatologists. The challenge is seamless integration of MSUS into busy dermatology practices.
The goal of PsA treatment is to achieve a state of remission or low disease activity. Criteria for minimal disease activity (MDA) have been established. Achieving MDA leads to better health-related quality of life (HRQOL), as well as less joint damage. In a prospective cohort study that included 240 patients with newly diagnosed disease-modifying antirheumatic drug-naive PsA, Snoeck Henkemans and colleagues demonstrate that failure to achieve MDA in the first year after the diagnosis of PsA was associated with worse HRQOL and health status, functional impairment, fatigue, pain, and higher anxiety and depression. Compared with patients who achieved sustained MDA in the first year after diagnosis, those who did not achieve MDA had higher scores for pain, fatigue, and functional ability and higher anxiety and depression during follow-up, which persisted despite treatment intensification. Thus, implementation of treat-to-target strategies with the aim of achieving sustained MDA within 1 year of diagnosis is likely to have better long-term benefits in this lifelong disease.
Another study emphasized the need for early treatment to improve long-term outcomes. In a post hoc analysis of two phase 3 trials including 1554 patients with PsA who received 300-mg or 150-mg secukinumab with or without a loading dose, Mease and colleagues showed that high baseline radiographic damage reduced the likelihood of achieving MDA.
Overall, these studies indicate that early diagnosis and treatment prior to developing joint damage with the aim to achieve sustained MDA within a year will lead to better long-term outcome for patients with PsA.
Appropriate assessment of MSK symptoms and signs by dermatologists may lead to more appropriate referral to rheumatologists. MSUS is being increasingly explored for early identification of PsA. A handheld, chip-based ultrasound device (HHUD) is a novel promising instrument that can be easily implemented in clinical practice. In a prospective study including 140 patients with psoriasis who presented to dermatologists with arthralgia. Grobelski and colleagues screened for PsA using medical history, CE, and the German Psoriasis Arthritis Diagnostic PsA screening questionnaire (GEPARD) paired with MSUS examination of up to three painful joints by trained dermatologists. Nineteen patients (13.6%) were diagnosed with PsA by rheumatologists. Interestingly, in 45 of the 46 patients the preliminary diagnosis of PsA was revised to "no PsA" after MSUS. The addition of MSUS changed the sensitivity and specificity of early PsA screening strategy from 88.2% and 54.4% to 70.6% and 90.4%, respectively. The positive predictive value increased to 56.5% from 25.4% after MSUS. Thus, the use of a quick MSUS using HHUD may lead to more accurate referral to rheumatologists. The challenge is seamless integration of MSUS into busy dermatology practices.
The goal of PsA treatment is to achieve a state of remission or low disease activity. Criteria for minimal disease activity (MDA) have been established. Achieving MDA leads to better health-related quality of life (HRQOL), as well as less joint damage. In a prospective cohort study that included 240 patients with newly diagnosed disease-modifying antirheumatic drug-naive PsA, Snoeck Henkemans and colleagues demonstrate that failure to achieve MDA in the first year after the diagnosis of PsA was associated with worse HRQOL and health status, functional impairment, fatigue, pain, and higher anxiety and depression. Compared with patients who achieved sustained MDA in the first year after diagnosis, those who did not achieve MDA had higher scores for pain, fatigue, and functional ability and higher anxiety and depression during follow-up, which persisted despite treatment intensification. Thus, implementation of treat-to-target strategies with the aim of achieving sustained MDA within 1 year of diagnosis is likely to have better long-term benefits in this lifelong disease.
Another study emphasized the need for early treatment to improve long-term outcomes. In a post hoc analysis of two phase 3 trials including 1554 patients with PsA who received 300-mg or 150-mg secukinumab with or without a loading dose, Mease and colleagues showed that high baseline radiographic damage reduced the likelihood of achieving MDA.
Overall, these studies indicate that early diagnosis and treatment prior to developing joint damage with the aim to achieve sustained MDA within a year will lead to better long-term outcome for patients with PsA.
Appropriate assessment of MSK symptoms and signs by dermatologists may lead to more appropriate referral to rheumatologists. MSUS is being increasingly explored for early identification of PsA. A handheld, chip-based ultrasound device (HHUD) is a novel promising instrument that can be easily implemented in clinical practice. In a prospective study including 140 patients with psoriasis who presented to dermatologists with arthralgia. Grobelski and colleagues screened for PsA using medical history, CE, and the German Psoriasis Arthritis Diagnostic PsA screening questionnaire (GEPARD) paired with MSUS examination of up to three painful joints by trained dermatologists. Nineteen patients (13.6%) were diagnosed with PsA by rheumatologists. Interestingly, in 45 of the 46 patients the preliminary diagnosis of PsA was revised to "no PsA" after MSUS. The addition of MSUS changed the sensitivity and specificity of early PsA screening strategy from 88.2% and 54.4% to 70.6% and 90.4%, respectively. The positive predictive value increased to 56.5% from 25.4% after MSUS. Thus, the use of a quick MSUS using HHUD may lead to more accurate referral to rheumatologists. The challenge is seamless integration of MSUS into busy dermatology practices.
The goal of PsA treatment is to achieve a state of remission or low disease activity. Criteria for minimal disease activity (MDA) have been established. Achieving MDA leads to better health-related quality of life (HRQOL), as well as less joint damage. In a prospective cohort study that included 240 patients with newly diagnosed disease-modifying antirheumatic drug-naive PsA, Snoeck Henkemans and colleagues demonstrate that failure to achieve MDA in the first year after the diagnosis of PsA was associated with worse HRQOL and health status, functional impairment, fatigue, pain, and higher anxiety and depression. Compared with patients who achieved sustained MDA in the first year after diagnosis, those who did not achieve MDA had higher scores for pain, fatigue, and functional ability and higher anxiety and depression during follow-up, which persisted despite treatment intensification. Thus, implementation of treat-to-target strategies with the aim of achieving sustained MDA within 1 year of diagnosis is likely to have better long-term benefits in this lifelong disease.
Another study emphasized the need for early treatment to improve long-term outcomes. In a post hoc analysis of two phase 3 trials including 1554 patients with PsA who received 300-mg or 150-mg secukinumab with or without a loading dose, Mease and colleagues showed that high baseline radiographic damage reduced the likelihood of achieving MDA.
Overall, these studies indicate that early diagnosis and treatment prior to developing joint damage with the aim to achieve sustained MDA within a year will lead to better long-term outcome for patients with PsA.
Fluorescence-optical imaging may detect preclinical PsA
Fluorescence-optical imaging (FOI) identified early signs of psoriatic arthritis, based on data from 2 years of follow-up of a cohort of 389 adults at 14 rheumatology centers.
Approximately 25% of individuals with psoriasis go on to develop psoriatic arthritis (PsA), but there are no validated biomarkers to identify patients at risk for progression to PsA, Michaela Koehm, MD, of Goethe University, Frankfurt am Main, Germany, and colleagues wrote in RMD Open.
FOI is a technique that allows assessment of changes in microvascularization and subdermal skin inflammation, and because individuals with psoriasis who develop PsA have shown changes in blood vessel formation in the early stages of disease, the researchers sought to determine if FOI could be used to predict early PsA.
The researchers conducted a multicenter, two-part observational cohort study. The two parts, known as XCITING and XTEND, included 389 adults aged 18-75 years with plaque psoriasis deemed at increased risk for PsA. The patients were seen at rheumatology sites in Germany between Jan. 28, 2014, and March 16, 2017. The XTEND study included clinic visits 18-24 months after the XCITING study.
Participants underwent a complete clinical examination, with musculoskeletal ultrasound (MSUS) and FOI on both hands at a single visit. Those with positive FOI findings not seen with clinical exam or MSUS underwent MRI within 7 days. Patients with positive FOI but negative findings on clinical exam, MSUS, and MRI were followed for 2 years in the XTEND study.
The primary outcome was the ability of FOI to detect musculoskeletal inflammation, compared with clinical examination and MSUS.
Overall, 50% of the patients were diagnosed with PsA. A total of 116 (30%) had positive FOI findings; complete MRI data were available for 108 of these patients, including 68 negative MRIs and 40 positive MRIs.
In the XTEND study, another 12% of patients who were positive on FOI but not on MRI also developed PsA by the end of the 2-year follow-up. In comparison, the researchers noted that “literature data on yearly incidence rates [of PsA] in different national cohorts indicate an incidence rate of approximately 4.3% per year.”
A total of 149 of the 196 patients with PsA confirmed by either clinical exam or MSUS were also positive on FOI, yielding a sensitivity of 76.0%. The specificity of FOI was 39.5%.
The sensitive visualization of musculoskeletal inflammation possible with FOI “may exceed its ability to detect clinically manifest PsA at high sensitivity or specificity, but early visualization is arguably of greater value as other imaging methods are currently available for detection of later stages of PsA,” the researchers wrote in their discussion. “A technique allowing early identification of PsA may be especially valuable for nonrheumatologists, including dermatologists and general practitioners, and help expedite more efficient referral to specialists.”
The findings were limited by several factors, including the nonrandomized design and small subgroup numbers, the researchers noted. Other limitations include the presence of alternative conditions such as osteoarthritis that might have complicated the imaging; the focus only on the hands; and potential variation in FOI assessment related to technical standards such as temperature and positioning.
However, the results support FOI as a safe and effective method of detecting early signs of joint inflammation that could predict increased risk for PsA in psoriasis patients, the researchers said.
The researchers added that more work is needed to evaluate FOI in clinical practice, but FOI has the potential to identify vascularization changes earlier than other imaging modalities and in advance of clinical symptoms.
“Accordingly, FOI may have the potential to improve patient outcomes in PsA by reducing the time to initiation of early treatment,” they concluded.
The study was supported by Fraunhofer ITMP, a nonprofit organization, and a research grant from Pfizer Germany. Some of the researchers disclosed financial relationships with many pharmaceutical companies, including Pfizer.
Fluorescence-optical imaging (FOI) identified early signs of psoriatic arthritis, based on data from 2 years of follow-up of a cohort of 389 adults at 14 rheumatology centers.
Approximately 25% of individuals with psoriasis go on to develop psoriatic arthritis (PsA), but there are no validated biomarkers to identify patients at risk for progression to PsA, Michaela Koehm, MD, of Goethe University, Frankfurt am Main, Germany, and colleagues wrote in RMD Open.
FOI is a technique that allows assessment of changes in microvascularization and subdermal skin inflammation, and because individuals with psoriasis who develop PsA have shown changes in blood vessel formation in the early stages of disease, the researchers sought to determine if FOI could be used to predict early PsA.
The researchers conducted a multicenter, two-part observational cohort study. The two parts, known as XCITING and XTEND, included 389 adults aged 18-75 years with plaque psoriasis deemed at increased risk for PsA. The patients were seen at rheumatology sites in Germany between Jan. 28, 2014, and March 16, 2017. The XTEND study included clinic visits 18-24 months after the XCITING study.
Participants underwent a complete clinical examination, with musculoskeletal ultrasound (MSUS) and FOI on both hands at a single visit. Those with positive FOI findings not seen with clinical exam or MSUS underwent MRI within 7 days. Patients with positive FOI but negative findings on clinical exam, MSUS, and MRI were followed for 2 years in the XTEND study.
The primary outcome was the ability of FOI to detect musculoskeletal inflammation, compared with clinical examination and MSUS.
Overall, 50% of the patients were diagnosed with PsA. A total of 116 (30%) had positive FOI findings; complete MRI data were available for 108 of these patients, including 68 negative MRIs and 40 positive MRIs.
In the XTEND study, another 12% of patients who were positive on FOI but not on MRI also developed PsA by the end of the 2-year follow-up. In comparison, the researchers noted that “literature data on yearly incidence rates [of PsA] in different national cohorts indicate an incidence rate of approximately 4.3% per year.”
A total of 149 of the 196 patients with PsA confirmed by either clinical exam or MSUS were also positive on FOI, yielding a sensitivity of 76.0%. The specificity of FOI was 39.5%.
The sensitive visualization of musculoskeletal inflammation possible with FOI “may exceed its ability to detect clinically manifest PsA at high sensitivity or specificity, but early visualization is arguably of greater value as other imaging methods are currently available for detection of later stages of PsA,” the researchers wrote in their discussion. “A technique allowing early identification of PsA may be especially valuable for nonrheumatologists, including dermatologists and general practitioners, and help expedite more efficient referral to specialists.”
The findings were limited by several factors, including the nonrandomized design and small subgroup numbers, the researchers noted. Other limitations include the presence of alternative conditions such as osteoarthritis that might have complicated the imaging; the focus only on the hands; and potential variation in FOI assessment related to technical standards such as temperature and positioning.
However, the results support FOI as a safe and effective method of detecting early signs of joint inflammation that could predict increased risk for PsA in psoriasis patients, the researchers said.
The researchers added that more work is needed to evaluate FOI in clinical practice, but FOI has the potential to identify vascularization changes earlier than other imaging modalities and in advance of clinical symptoms.
“Accordingly, FOI may have the potential to improve patient outcomes in PsA by reducing the time to initiation of early treatment,” they concluded.
The study was supported by Fraunhofer ITMP, a nonprofit organization, and a research grant from Pfizer Germany. Some of the researchers disclosed financial relationships with many pharmaceutical companies, including Pfizer.
Fluorescence-optical imaging (FOI) identified early signs of psoriatic arthritis, based on data from 2 years of follow-up of a cohort of 389 adults at 14 rheumatology centers.
Approximately 25% of individuals with psoriasis go on to develop psoriatic arthritis (PsA), but there are no validated biomarkers to identify patients at risk for progression to PsA, Michaela Koehm, MD, of Goethe University, Frankfurt am Main, Germany, and colleagues wrote in RMD Open.
FOI is a technique that allows assessment of changes in microvascularization and subdermal skin inflammation, and because individuals with psoriasis who develop PsA have shown changes in blood vessel formation in the early stages of disease, the researchers sought to determine if FOI could be used to predict early PsA.
The researchers conducted a multicenter, two-part observational cohort study. The two parts, known as XCITING and XTEND, included 389 adults aged 18-75 years with plaque psoriasis deemed at increased risk for PsA. The patients were seen at rheumatology sites in Germany between Jan. 28, 2014, and March 16, 2017. The XTEND study included clinic visits 18-24 months after the XCITING study.
Participants underwent a complete clinical examination, with musculoskeletal ultrasound (MSUS) and FOI on both hands at a single visit. Those with positive FOI findings not seen with clinical exam or MSUS underwent MRI within 7 days. Patients with positive FOI but negative findings on clinical exam, MSUS, and MRI were followed for 2 years in the XTEND study.
The primary outcome was the ability of FOI to detect musculoskeletal inflammation, compared with clinical examination and MSUS.
Overall, 50% of the patients were diagnosed with PsA. A total of 116 (30%) had positive FOI findings; complete MRI data were available for 108 of these patients, including 68 negative MRIs and 40 positive MRIs.
In the XTEND study, another 12% of patients who were positive on FOI but not on MRI also developed PsA by the end of the 2-year follow-up. In comparison, the researchers noted that “literature data on yearly incidence rates [of PsA] in different national cohorts indicate an incidence rate of approximately 4.3% per year.”
A total of 149 of the 196 patients with PsA confirmed by either clinical exam or MSUS were also positive on FOI, yielding a sensitivity of 76.0%. The specificity of FOI was 39.5%.
The sensitive visualization of musculoskeletal inflammation possible with FOI “may exceed its ability to detect clinically manifest PsA at high sensitivity or specificity, but early visualization is arguably of greater value as other imaging methods are currently available for detection of later stages of PsA,” the researchers wrote in their discussion. “A technique allowing early identification of PsA may be especially valuable for nonrheumatologists, including dermatologists and general practitioners, and help expedite more efficient referral to specialists.”
The findings were limited by several factors, including the nonrandomized design and small subgroup numbers, the researchers noted. Other limitations include the presence of alternative conditions such as osteoarthritis that might have complicated the imaging; the focus only on the hands; and potential variation in FOI assessment related to technical standards such as temperature and positioning.
However, the results support FOI as a safe and effective method of detecting early signs of joint inflammation that could predict increased risk for PsA in psoriasis patients, the researchers said.
The researchers added that more work is needed to evaluate FOI in clinical practice, but FOI has the potential to identify vascularization changes earlier than other imaging modalities and in advance of clinical symptoms.
“Accordingly, FOI may have the potential to improve patient outcomes in PsA by reducing the time to initiation of early treatment,” they concluded.
The study was supported by Fraunhofer ITMP, a nonprofit organization, and a research grant from Pfizer Germany. Some of the researchers disclosed financial relationships with many pharmaceutical companies, including Pfizer.
FROM RMD OPEN

