User login
HIV increases risk for severe COVID-19
according to a report from the World Health Organization on COVID-19 outcomes among people living with HIV. The study primarily included people from South Africa but also some data from other parts of the world, including the United States.
However, the report, presented at the 11th IAS Conference on HIV Science (IAS 2021), couldn’t answer some crucial questions clinicians have been wondering about since the COVID-19 pandemic began. For example, was the increase in COVID risk a result of the presence of HIV or because of the immune compromise caused by untreated HIV?
The report didn’t include data on viral load or CD counts, both used to evaluate the health of a person’s immune system. On effective treatment, people living with HIV have a lifespan close to their HIV-negative peers. And effective treatment causes undetectable viral loads which, when maintained for 6 months or more, eliminates transmission of HIV to sexual partners.
What’s clear is that in people with HIV, as in people without HIV, older people, men, and people with diabetes, hypertension, or obesity had the worst outcomes and were most likely to die from COVID-19.
For David Malebranche, MD, MPH, an internal medicine doctor who provides primary care for people in Atlanta, and who was not involved in the study, the WHO study didn’t add anything new. He already recommends the COVID-19 vaccine for all of his patients, HIV-positive or not.
“We don’t have any information from this about the T-cell counts [or] the rates of viral suppression, which I think is tremendously important,” he told this news organization. “To bypass that and not include that in any of the discussion puts the results in a questionable place for me.”
The results come from the WHO Clinical Platform, which culls data from WHO member country surveillance as well as manual case reports from all over the world. By April 29, data on 268,412 people hospitalized with COVID-19 from 37 countries were reported to the platform. Of those, 22,640 people are from the U.S.
A total of 15,522 participants worldwide were living with HIV, 664 in the United States. All U.S. cases were reported from the New York City Health and Hospitals system, Henry Ford Hospital in Detroit, and BronxCare Health System in New York City. Almost all of the remaining participants lived in South Africa – 14,682 of the 15,522, or 94.5%.
Of the 15,522 people living with HIV in the overall group, 37.1% of participants were male, and their median age was 45 years. More than 1 in 3 (36.2%) were admitted with severe or critical COVID-19, and nearly one quarter – 23.1% – with a known outcome died. More than half had one or more chronic conditions, including those that themselves are associated with worse COVID-19 outcomes, such as hypertension (in 33.2% of the participants), diabetes (22.7%), and BMIs above 30 (16.9%). In addition, 8.9% were smokers, 6.6% had chronic pulmonary disease, and 4.3% had chronic heart disease.
After adjusting for those chronic conditions, age, and sex, people living with HIV had a 6% higher rate of severe or critical COVID-19 illness. When investigators adjusted the analysis additionally to differentiate outcomes based on not just the presence of comorbid conditions but the number of them a person had, that increased risk rose to 13%. HIV itself is a comorbid condition, though it wasn’t counted as one in this adjusted analysis.
It didn’t matter whether researchers looked at risk for severe outcomes or deaths after removing the significant co-occurring conditions or if they looked at number of chronic illnesses (aside from HIV), said Silvia Bertagnolio, MD, medical officer at the World Health Organization and co-author of the analysis.
“Both models show almost identical [adjusted odds ratios], meaning that HIV was independently significantly associated with severe/critical presentation,” she told this news organization.
As for death, the analysis showed that, overall, people living with HIV were 30% more likely to die of COVID-19 compared with those not living with HIV. And while this held true even when they adjusted the data for comorbidities, people with HIV were more likely to die if they were over age 65 (risk increased by 82%), male (risk increased by 21%), had diabetes (risk increased by 50%), or had hypertension (risk increased by 26%).
When they broke down the data by WHO region – Africa, Europe, the Americas – investigators found that the increased risk for death held true in Africa. But there were not enough data from the other regions to model mortality risk. What’s more, when they broke the data down by country and excluded South Africa, they found that the elevated risk for death in people living with HIV did not reach statistical significance. Dr. Bertagnolio said she suspects that the small sample sizes from other regions made it impossible to detect a difference, but one could still be present.
One thing conspicuously absent from the analysis was information on viral load, CD4 T-cell count, progression of HIV to AIDS, and whether individuals were in HIV care. The first three factors were not reported in the platform, and the fourth was available for 60% of participants but was not included in the analysis. Dr. Bertagnolio pointed out that, for those 60% of participants, 91.8% were on antiretroviral treatment (ART).
“The majority of patients come from South Africa, and we know that in South Africa, over 90% of people receiving ART are virologically suppressed,” she told this news organization. “So we could speculate that this effect persists despite the use of ART, in a population likely to be virally suppressed, although we cannot assess this with certainty through the data set we had.”
A much smaller study of 749 people living with HIV and diagnosed with SARS-CoV-2, also presented at the conference, found that detectable HIV viral load was significantly associated with a slightly higher risk of severe outcomes (P < .039), but CD4 counts less than 200 cells/mm3 was not (P = .15).
And although both Dr. Bertagnolio and conference organizers presented this data as proof that HIV increases the risk for poor COVID-19 outcomes, Dr. Malebranche isn’t so sure. He estimates that only about half his patients have received the COVID-19 vaccine. But this study is unlikely to make him forcefully recommend a COVID-19 vaccination with young, otherwise healthy, and undetectable people in his care who express particular concern about long-term effects of the vaccine. He also manages a lot of people with HIV who have undetectable viral loads and CD4 counts of up to 1,200 but are older, with diabetes, obesity, and high blood pressure. Those are the people he will target with stronger messages regarding the vaccine.
“The young patients who are healthy, virally suppressed, and doing well may very much argue with me, ‘I’m not going to push it,’ but I will bring it up on the next visit,” he said. The analysis “just helps reinforce in me that I need to have these conversations and be a little bit more persuasive to my older patients with comorbid conditions.”
Dr. Bertagnolio has disclosed no relevant financial relationships. Dr. Malebranche serves on the pre-exposure prophylaxis (PrEP) speakers bureau for Gilead Sciences and has consulted and advised for ViiV Healthcare. This study was funded by the World Health Organization.
A version of this article first appeared on Medscape.com.
according to a report from the World Health Organization on COVID-19 outcomes among people living with HIV. The study primarily included people from South Africa but also some data from other parts of the world, including the United States.
However, the report, presented at the 11th IAS Conference on HIV Science (IAS 2021), couldn’t answer some crucial questions clinicians have been wondering about since the COVID-19 pandemic began. For example, was the increase in COVID risk a result of the presence of HIV or because of the immune compromise caused by untreated HIV?
The report didn’t include data on viral load or CD counts, both used to evaluate the health of a person’s immune system. On effective treatment, people living with HIV have a lifespan close to their HIV-negative peers. And effective treatment causes undetectable viral loads which, when maintained for 6 months or more, eliminates transmission of HIV to sexual partners.
What’s clear is that in people with HIV, as in people without HIV, older people, men, and people with diabetes, hypertension, or obesity had the worst outcomes and were most likely to die from COVID-19.
For David Malebranche, MD, MPH, an internal medicine doctor who provides primary care for people in Atlanta, and who was not involved in the study, the WHO study didn’t add anything new. He already recommends the COVID-19 vaccine for all of his patients, HIV-positive or not.
“We don’t have any information from this about the T-cell counts [or] the rates of viral suppression, which I think is tremendously important,” he told this news organization. “To bypass that and not include that in any of the discussion puts the results in a questionable place for me.”
The results come from the WHO Clinical Platform, which culls data from WHO member country surveillance as well as manual case reports from all over the world. By April 29, data on 268,412 people hospitalized with COVID-19 from 37 countries were reported to the platform. Of those, 22,640 people are from the U.S.
A total of 15,522 participants worldwide were living with HIV, 664 in the United States. All U.S. cases were reported from the New York City Health and Hospitals system, Henry Ford Hospital in Detroit, and BronxCare Health System in New York City. Almost all of the remaining participants lived in South Africa – 14,682 of the 15,522, or 94.5%.
Of the 15,522 people living with HIV in the overall group, 37.1% of participants were male, and their median age was 45 years. More than 1 in 3 (36.2%) were admitted with severe or critical COVID-19, and nearly one quarter – 23.1% – with a known outcome died. More than half had one or more chronic conditions, including those that themselves are associated with worse COVID-19 outcomes, such as hypertension (in 33.2% of the participants), diabetes (22.7%), and BMIs above 30 (16.9%). In addition, 8.9% were smokers, 6.6% had chronic pulmonary disease, and 4.3% had chronic heart disease.
After adjusting for those chronic conditions, age, and sex, people living with HIV had a 6% higher rate of severe or critical COVID-19 illness. When investigators adjusted the analysis additionally to differentiate outcomes based on not just the presence of comorbid conditions but the number of them a person had, that increased risk rose to 13%. HIV itself is a comorbid condition, though it wasn’t counted as one in this adjusted analysis.
It didn’t matter whether researchers looked at risk for severe outcomes or deaths after removing the significant co-occurring conditions or if they looked at number of chronic illnesses (aside from HIV), said Silvia Bertagnolio, MD, medical officer at the World Health Organization and co-author of the analysis.
“Both models show almost identical [adjusted odds ratios], meaning that HIV was independently significantly associated with severe/critical presentation,” she told this news organization.
As for death, the analysis showed that, overall, people living with HIV were 30% more likely to die of COVID-19 compared with those not living with HIV. And while this held true even when they adjusted the data for comorbidities, people with HIV were more likely to die if they were over age 65 (risk increased by 82%), male (risk increased by 21%), had diabetes (risk increased by 50%), or had hypertension (risk increased by 26%).
When they broke down the data by WHO region – Africa, Europe, the Americas – investigators found that the increased risk for death held true in Africa. But there were not enough data from the other regions to model mortality risk. What’s more, when they broke the data down by country and excluded South Africa, they found that the elevated risk for death in people living with HIV did not reach statistical significance. Dr. Bertagnolio said she suspects that the small sample sizes from other regions made it impossible to detect a difference, but one could still be present.
One thing conspicuously absent from the analysis was information on viral load, CD4 T-cell count, progression of HIV to AIDS, and whether individuals were in HIV care. The first three factors were not reported in the platform, and the fourth was available for 60% of participants but was not included in the analysis. Dr. Bertagnolio pointed out that, for those 60% of participants, 91.8% were on antiretroviral treatment (ART).
“The majority of patients come from South Africa, and we know that in South Africa, over 90% of people receiving ART are virologically suppressed,” she told this news organization. “So we could speculate that this effect persists despite the use of ART, in a population likely to be virally suppressed, although we cannot assess this with certainty through the data set we had.”
A much smaller study of 749 people living with HIV and diagnosed with SARS-CoV-2, also presented at the conference, found that detectable HIV viral load was significantly associated with a slightly higher risk of severe outcomes (P < .039), but CD4 counts less than 200 cells/mm3 was not (P = .15).
And although both Dr. Bertagnolio and conference organizers presented this data as proof that HIV increases the risk for poor COVID-19 outcomes, Dr. Malebranche isn’t so sure. He estimates that only about half his patients have received the COVID-19 vaccine. But this study is unlikely to make him forcefully recommend a COVID-19 vaccination with young, otherwise healthy, and undetectable people in his care who express particular concern about long-term effects of the vaccine. He also manages a lot of people with HIV who have undetectable viral loads and CD4 counts of up to 1,200 but are older, with diabetes, obesity, and high blood pressure. Those are the people he will target with stronger messages regarding the vaccine.
“The young patients who are healthy, virally suppressed, and doing well may very much argue with me, ‘I’m not going to push it,’ but I will bring it up on the next visit,” he said. The analysis “just helps reinforce in me that I need to have these conversations and be a little bit more persuasive to my older patients with comorbid conditions.”
Dr. Bertagnolio has disclosed no relevant financial relationships. Dr. Malebranche serves on the pre-exposure prophylaxis (PrEP) speakers bureau for Gilead Sciences and has consulted and advised for ViiV Healthcare. This study was funded by the World Health Organization.
A version of this article first appeared on Medscape.com.
according to a report from the World Health Organization on COVID-19 outcomes among people living with HIV. The study primarily included people from South Africa but also some data from other parts of the world, including the United States.
However, the report, presented at the 11th IAS Conference on HIV Science (IAS 2021), couldn’t answer some crucial questions clinicians have been wondering about since the COVID-19 pandemic began. For example, was the increase in COVID risk a result of the presence of HIV or because of the immune compromise caused by untreated HIV?
The report didn’t include data on viral load or CD counts, both used to evaluate the health of a person’s immune system. On effective treatment, people living with HIV have a lifespan close to their HIV-negative peers. And effective treatment causes undetectable viral loads which, when maintained for 6 months or more, eliminates transmission of HIV to sexual partners.
What’s clear is that in people with HIV, as in people without HIV, older people, men, and people with diabetes, hypertension, or obesity had the worst outcomes and were most likely to die from COVID-19.
For David Malebranche, MD, MPH, an internal medicine doctor who provides primary care for people in Atlanta, and who was not involved in the study, the WHO study didn’t add anything new. He already recommends the COVID-19 vaccine for all of his patients, HIV-positive or not.
“We don’t have any information from this about the T-cell counts [or] the rates of viral suppression, which I think is tremendously important,” he told this news organization. “To bypass that and not include that in any of the discussion puts the results in a questionable place for me.”
The results come from the WHO Clinical Platform, which culls data from WHO member country surveillance as well as manual case reports from all over the world. By April 29, data on 268,412 people hospitalized with COVID-19 from 37 countries were reported to the platform. Of those, 22,640 people are from the U.S.
A total of 15,522 participants worldwide were living with HIV, 664 in the United States. All U.S. cases were reported from the New York City Health and Hospitals system, Henry Ford Hospital in Detroit, and BronxCare Health System in New York City. Almost all of the remaining participants lived in South Africa – 14,682 of the 15,522, or 94.5%.
Of the 15,522 people living with HIV in the overall group, 37.1% of participants were male, and their median age was 45 years. More than 1 in 3 (36.2%) were admitted with severe or critical COVID-19, and nearly one quarter – 23.1% – with a known outcome died. More than half had one or more chronic conditions, including those that themselves are associated with worse COVID-19 outcomes, such as hypertension (in 33.2% of the participants), diabetes (22.7%), and BMIs above 30 (16.9%). In addition, 8.9% were smokers, 6.6% had chronic pulmonary disease, and 4.3% had chronic heart disease.
After adjusting for those chronic conditions, age, and sex, people living with HIV had a 6% higher rate of severe or critical COVID-19 illness. When investigators adjusted the analysis additionally to differentiate outcomes based on not just the presence of comorbid conditions but the number of them a person had, that increased risk rose to 13%. HIV itself is a comorbid condition, though it wasn’t counted as one in this adjusted analysis.
It didn’t matter whether researchers looked at risk for severe outcomes or deaths after removing the significant co-occurring conditions or if they looked at number of chronic illnesses (aside from HIV), said Silvia Bertagnolio, MD, medical officer at the World Health Organization and co-author of the analysis.
“Both models show almost identical [adjusted odds ratios], meaning that HIV was independently significantly associated with severe/critical presentation,” she told this news organization.
As for death, the analysis showed that, overall, people living with HIV were 30% more likely to die of COVID-19 compared with those not living with HIV. And while this held true even when they adjusted the data for comorbidities, people with HIV were more likely to die if they were over age 65 (risk increased by 82%), male (risk increased by 21%), had diabetes (risk increased by 50%), or had hypertension (risk increased by 26%).
When they broke down the data by WHO region – Africa, Europe, the Americas – investigators found that the increased risk for death held true in Africa. But there were not enough data from the other regions to model mortality risk. What’s more, when they broke the data down by country and excluded South Africa, they found that the elevated risk for death in people living with HIV did not reach statistical significance. Dr. Bertagnolio said she suspects that the small sample sizes from other regions made it impossible to detect a difference, but one could still be present.
One thing conspicuously absent from the analysis was information on viral load, CD4 T-cell count, progression of HIV to AIDS, and whether individuals were in HIV care. The first three factors were not reported in the platform, and the fourth was available for 60% of participants but was not included in the analysis. Dr. Bertagnolio pointed out that, for those 60% of participants, 91.8% were on antiretroviral treatment (ART).
“The majority of patients come from South Africa, and we know that in South Africa, over 90% of people receiving ART are virologically suppressed,” she told this news organization. “So we could speculate that this effect persists despite the use of ART, in a population likely to be virally suppressed, although we cannot assess this with certainty through the data set we had.”
A much smaller study of 749 people living with HIV and diagnosed with SARS-CoV-2, also presented at the conference, found that detectable HIV viral load was significantly associated with a slightly higher risk of severe outcomes (P < .039), but CD4 counts less than 200 cells/mm3 was not (P = .15).
And although both Dr. Bertagnolio and conference organizers presented this data as proof that HIV increases the risk for poor COVID-19 outcomes, Dr. Malebranche isn’t so sure. He estimates that only about half his patients have received the COVID-19 vaccine. But this study is unlikely to make him forcefully recommend a COVID-19 vaccination with young, otherwise healthy, and undetectable people in his care who express particular concern about long-term effects of the vaccine. He also manages a lot of people with HIV who have undetectable viral loads and CD4 counts of up to 1,200 but are older, with diabetes, obesity, and high blood pressure. Those are the people he will target with stronger messages regarding the vaccine.
“The young patients who are healthy, virally suppressed, and doing well may very much argue with me, ‘I’m not going to push it,’ but I will bring it up on the next visit,” he said. The analysis “just helps reinforce in me that I need to have these conversations and be a little bit more persuasive to my older patients with comorbid conditions.”
Dr. Bertagnolio has disclosed no relevant financial relationships. Dr. Malebranche serves on the pre-exposure prophylaxis (PrEP) speakers bureau for Gilead Sciences and has consulted and advised for ViiV Healthcare. This study was funded by the World Health Organization.
A version of this article first appeared on Medscape.com.
Pharmacologic and electrical cardioversion of acute Afib reduces hospital admissions
Background: Atrial fibrillation (Afib) is the most common arrhythmia requiring treatment in the ED. There is a paucity of literature regarding the management of acute (onset < 48 h) atrial fibrillation in this setting and no conclusive evidence exists regarding the superiority of pharmacologic vs. electrical cardioversion.
Study design: Multicenter, single-blind, randomized, placebo-controlled trial.
Setting: 11 Canadian academic medical centers.
Synopsis: In this trial of 396 patients with acute Afib, half were randomly assigned to pharmacologic cardioversion with procainamide infusion (followed by DC cardioversion, if unsuccessful), while half were given a placebo infusion then DC cardioversion. The primary outcome was conversion to sinus rhythm, with maintenance of sinus rhythm at 30 minutes. A secondary protocol evaluated the difference in efficacy between anterolateral (AL) and anteroposterior (AP) pad placement
The “drug-shock” group achieved and maintained sinus rhythm in 96% of cases, compared to 92% in the “placebo-shock” group (statistically insignificant difference). The procainamide infusion alone achieved and maintained sinus rhythm in 52% of recipients, who thereby avoided the need for procedural sedation and monitoring. Notably, only 2% of patients in the study required admission to the hospital. Pad placement was equally efficacious in the AL or AP positions. The most common adverse event observed was transient hypotension during infusion of procainamide. No strokes were observed in either arm. Follow-up ECGs obtained 14 days later showed that 95% of patients remained in sinus rhythm.
Bottom line: Pharmacologic cardioversion with procainamide infusion and/or electrical cardioversion is a safe and efficacious initial management strategy for acute atrial fibrillation, and all but eliminates the need for hospital admission.
Citation: Stiell IG et al. Electrical versus pharmacological cardioversion for emergency department patients with acute atrial fibrillation (RAFF2): a partial factorial randomized trial. Lancet. 2020 Feb 1;395(10221):339-49.
Dr. Lawson is assistant professor of medicine, section of hospital medicine, at the University of Virginia School of Medicine, Charlottesville.
Background: Atrial fibrillation (Afib) is the most common arrhythmia requiring treatment in the ED. There is a paucity of literature regarding the management of acute (onset < 48 h) atrial fibrillation in this setting and no conclusive evidence exists regarding the superiority of pharmacologic vs. electrical cardioversion.
Study design: Multicenter, single-blind, randomized, placebo-controlled trial.
Setting: 11 Canadian academic medical centers.
Synopsis: In this trial of 396 patients with acute Afib, half were randomly assigned to pharmacologic cardioversion with procainamide infusion (followed by DC cardioversion, if unsuccessful), while half were given a placebo infusion then DC cardioversion. The primary outcome was conversion to sinus rhythm, with maintenance of sinus rhythm at 30 minutes. A secondary protocol evaluated the difference in efficacy between anterolateral (AL) and anteroposterior (AP) pad placement
The “drug-shock” group achieved and maintained sinus rhythm in 96% of cases, compared to 92% in the “placebo-shock” group (statistically insignificant difference). The procainamide infusion alone achieved and maintained sinus rhythm in 52% of recipients, who thereby avoided the need for procedural sedation and monitoring. Notably, only 2% of patients in the study required admission to the hospital. Pad placement was equally efficacious in the AL or AP positions. The most common adverse event observed was transient hypotension during infusion of procainamide. No strokes were observed in either arm. Follow-up ECGs obtained 14 days later showed that 95% of patients remained in sinus rhythm.
Bottom line: Pharmacologic cardioversion with procainamide infusion and/or electrical cardioversion is a safe and efficacious initial management strategy for acute atrial fibrillation, and all but eliminates the need for hospital admission.
Citation: Stiell IG et al. Electrical versus pharmacological cardioversion for emergency department patients with acute atrial fibrillation (RAFF2): a partial factorial randomized trial. Lancet. 2020 Feb 1;395(10221):339-49.
Dr. Lawson is assistant professor of medicine, section of hospital medicine, at the University of Virginia School of Medicine, Charlottesville.
Background: Atrial fibrillation (Afib) is the most common arrhythmia requiring treatment in the ED. There is a paucity of literature regarding the management of acute (onset < 48 h) atrial fibrillation in this setting and no conclusive evidence exists regarding the superiority of pharmacologic vs. electrical cardioversion.
Study design: Multicenter, single-blind, randomized, placebo-controlled trial.
Setting: 11 Canadian academic medical centers.
Synopsis: In this trial of 396 patients with acute Afib, half were randomly assigned to pharmacologic cardioversion with procainamide infusion (followed by DC cardioversion, if unsuccessful), while half were given a placebo infusion then DC cardioversion. The primary outcome was conversion to sinus rhythm, with maintenance of sinus rhythm at 30 minutes. A secondary protocol evaluated the difference in efficacy between anterolateral (AL) and anteroposterior (AP) pad placement
The “drug-shock” group achieved and maintained sinus rhythm in 96% of cases, compared to 92% in the “placebo-shock” group (statistically insignificant difference). The procainamide infusion alone achieved and maintained sinus rhythm in 52% of recipients, who thereby avoided the need for procedural sedation and monitoring. Notably, only 2% of patients in the study required admission to the hospital. Pad placement was equally efficacious in the AL or AP positions. The most common adverse event observed was transient hypotension during infusion of procainamide. No strokes were observed in either arm. Follow-up ECGs obtained 14 days later showed that 95% of patients remained in sinus rhythm.
Bottom line: Pharmacologic cardioversion with procainamide infusion and/or electrical cardioversion is a safe and efficacious initial management strategy for acute atrial fibrillation, and all but eliminates the need for hospital admission.
Citation: Stiell IG et al. Electrical versus pharmacological cardioversion for emergency department patients with acute atrial fibrillation (RAFF2): a partial factorial randomized trial. Lancet. 2020 Feb 1;395(10221):339-49.
Dr. Lawson is assistant professor of medicine, section of hospital medicine, at the University of Virginia School of Medicine, Charlottesville.
Long COVID seen in patients with severe and mild disease
Findings from the cohort, composed of 113 COVID-19 survivors who developed ARDS after admission to a single center before to April 16, 2020, were presented online at the 31st European Congress of Clinical Microbiology & Infectious Diseases by Judit Aranda, MD, from Complex Hospitalari Moisés Broggi in Barcelona.
Median age of the participants was 64 years, and 70% were male. At least one persistent symptom was experienced during follow-up by 81% of the cohort, with 45% reporting shortness of breath, 50% reporting muscle pain, 43% reporting memory impairment, and 46% reporting physical weakness of at least 5 on a 10-point scale.
Of the 104 participants who completed a 6-minute walk test, 30% had a decrease in oxygen saturation level of at least 4%, and 5% had an initial or final level below 88%. Of the 46 participants who underwent a pulmonary function test, 15% had a forced expiratory volume in 1 second below 70%.
And of the 49% of participants with pathologic findings on chest x-ray, most were bilateral interstitial infiltrates (88%).
In addition, more than 90% of participants developed depression, anxiety, or PTSD, Dr. Aranda reported.
Not the whole picture
This study shows that sicker people – “those in intensive care units with acute respiratory distress syndrome” – are “more likely to be struggling with more severe symptoms,” said Christopher Terndrup, MD, from the division of general internal medicine and geriatrics at Oregon Health & Science University, Portland.
But a Swiss study, also presented at the meeting, “shows how even mild COVID cases can lead to debilitating symptoms,” Dr. Terndrup said in an interview.
The investigation of long-term COVID symptoms in outpatients was presented online by Florian Desgranges, MD, from Lausanne (Switzerland) University Hospital. He and his colleagues found that more than half of those with a mild to moderate disease had persistent symptoms at least 3 months after diagnosis.
The prevalence of long COVID has varied in previous research, from 15% in a study of health care workers, to 46% in a study of patients with mild COVID, 52% in a study of young COVID outpatients, and 76% in a study of patients hospitalized with COVID.
Dr. Desgranges and colleagues evaluated patients seen in an ED or outpatient clinic from February to April 2020.
The 418 patients with a confirmed COVID-19 diagnosis were compared with a control group of 89 patients who presented to the same centers during the same time frame with similar symptoms – cough, shortness of breath, or fever – but had a negative SARS-CoV-2 test.
The number of patients with comorbidities was similar in the COVID and control groups (34% vs. 36%), as was median age (41 vs. 36 years) and the prevalence of women (62% vs 64%), but the proportion of health care workers was lower in the COVID group (64% vs 82%; P =.006).
Symptoms that persisted for at least 3 months were more common in the COVID than in the control group (53% vs. 37%). And patients in the COVID group reported more symptoms than those in the control group after adjustment for age, gender, smoking status, comorbidities, and timing of the survey phone call.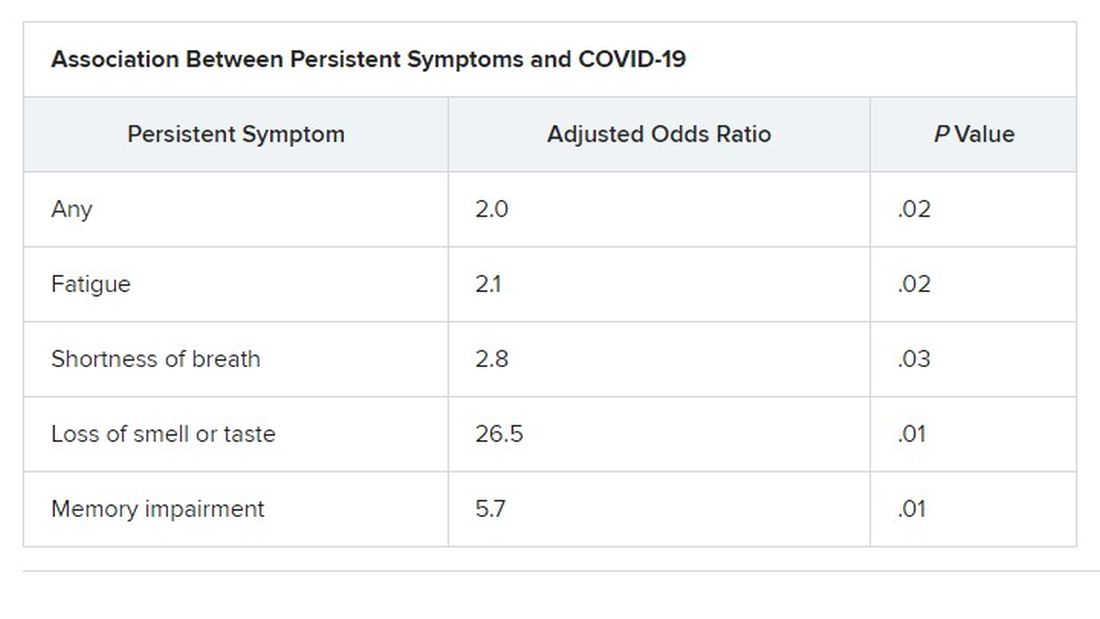
Levels of sleeping problems and headache were similar in the two groups.
“We have to remember that with COVID-19 came the psychosocial changes of the pandemic situation” Dr. Desgranges said.
This study suggests that some long-COVID symptoms – such as the fatigue, headache, and sleep disorders reported in the control group – could be related to the pandemic itself, which has caused psychosocial distress, Dr. Terndrup said.
Another study that looked at outpatients “has some fantastic long-term follow-up data, and shows that many patients are still engaging in rehabilitation programs nearly a year after their diagnosis,” he explained.
The COVID HOME study
That prospective longitudinal COVID HOME study, which assessed long-term symptoms in people who were never hospitalized for COVID, was presented online by Adriana Tami, MD, PhD, from the University Medical Center Groningen (the Netherlands).
The researchers visited the homes of patients to collect data, blood samples, and perform polymerase chain reaction (PCR) testing 1, 2, and 3 weeks after a diagnosis of COVID-19. If their PCR test was still positive, testing continued until week 6 or a negative test. In addition, participants completed questionnaires at week 2 and at months 3, 6 and 12 to assess fatigue, quality of life, and symptoms of depression and anxiety.
Three-month follow-up data were available for 134 of the 276 people initially enrolled in the study. Questionnaires were completed by 85 participants at 3 months, 62 participants at 6 months, and 10 participants at 12 months.
At least 40% of participants reported long-lasting symptoms at some point during follow-up, and at least 30% said they didn’t feel fully recovered at 12 months. The most common symptom was persistent fatigue, reported at 3, 6, and 12 months by at least 44% of participants. Other common symptoms – reported by at least 20% of respondents at 3, 6, and 12 months – were headache, mental or neurologic symptoms, and sleep disorders, shortness of breath, lack of smell or taste, and severe fatigue.
“We have a high proportion of nonhospitalized individuals who suffer from long COVID after more than 12 months,” Dr. Tami concluded, adding that the study is ongoing. “We have other variables that we want to look at, including duration viral shedding and serological results and variants.”
“These cohort studies are very helpful, but they can lead to inaccurate conclusions,” Dr. Terndrup cautioned.
They only provide pieces of the big picture, but they “do add to a growing body of knowledge about a significant portion of COVID patients still struggling with symptoms long after their initial infection. The symptoms can be quite variable but are dominated by both physical and mental fatigue, and tend to be worse in patients who were sicker at initial infection,” he said in an interview.
As a whole, these studies reinforce the need for treatment programs to help patients who suffer from long COVID, he added, but “I advise caution to folks suffering out there who seek ‘miracle cures’; across the world, we are collaborating to find solutions that are safe and effective.”
We are in desperate need of an equity lens in these studies.
“There is still a great deal to learn about long COVID,” said Dr. Terndrup. Data on underrepresented populations – such as Black, Indigenous, and people of color – are lacking from these and others studies, he explained. “We are in desperate need of an equity lens in these studies,” particularly in the United States, where there are “significant disparities” in the treatment of different populations.
However, “I do hope that this work can lead to a better understanding of how other viral infections can cause long-lasting symptoms,” said Dr. Terndrup.
“We have long proposed that after acute presentation, some microbes can cause chronic symptoms, like fatigue and widespread pain. Perhaps we can learn how to better care for these patients after learning from COVID’s significant impact on our societies across the globe.”
Dr. Aranda and Dr. Desgranges have disclosed no relevant financial relationships or study funding. The study by Dr. Tami’s team was funded by the University Medical Center Groningen Organization for Health Research and Development, and Connecting European Cohorts to Increase Common and Effective Response to SARS-CoV-2 Pandemic. Dr. Terndrup disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Findings from the cohort, composed of 113 COVID-19 survivors who developed ARDS after admission to a single center before to April 16, 2020, were presented online at the 31st European Congress of Clinical Microbiology & Infectious Diseases by Judit Aranda, MD, from Complex Hospitalari Moisés Broggi in Barcelona.
Median age of the participants was 64 years, and 70% were male. At least one persistent symptom was experienced during follow-up by 81% of the cohort, with 45% reporting shortness of breath, 50% reporting muscle pain, 43% reporting memory impairment, and 46% reporting physical weakness of at least 5 on a 10-point scale.
Of the 104 participants who completed a 6-minute walk test, 30% had a decrease in oxygen saturation level of at least 4%, and 5% had an initial or final level below 88%. Of the 46 participants who underwent a pulmonary function test, 15% had a forced expiratory volume in 1 second below 70%.
And of the 49% of participants with pathologic findings on chest x-ray, most were bilateral interstitial infiltrates (88%).
In addition, more than 90% of participants developed depression, anxiety, or PTSD, Dr. Aranda reported.
Not the whole picture
This study shows that sicker people – “those in intensive care units with acute respiratory distress syndrome” – are “more likely to be struggling with more severe symptoms,” said Christopher Terndrup, MD, from the division of general internal medicine and geriatrics at Oregon Health & Science University, Portland.
But a Swiss study, also presented at the meeting, “shows how even mild COVID cases can lead to debilitating symptoms,” Dr. Terndrup said in an interview.
The investigation of long-term COVID symptoms in outpatients was presented online by Florian Desgranges, MD, from Lausanne (Switzerland) University Hospital. He and his colleagues found that more than half of those with a mild to moderate disease had persistent symptoms at least 3 months after diagnosis.
The prevalence of long COVID has varied in previous research, from 15% in a study of health care workers, to 46% in a study of patients with mild COVID, 52% in a study of young COVID outpatients, and 76% in a study of patients hospitalized with COVID.
Dr. Desgranges and colleagues evaluated patients seen in an ED or outpatient clinic from February to April 2020.
The 418 patients with a confirmed COVID-19 diagnosis were compared with a control group of 89 patients who presented to the same centers during the same time frame with similar symptoms – cough, shortness of breath, or fever – but had a negative SARS-CoV-2 test.
The number of patients with comorbidities was similar in the COVID and control groups (34% vs. 36%), as was median age (41 vs. 36 years) and the prevalence of women (62% vs 64%), but the proportion of health care workers was lower in the COVID group (64% vs 82%; P =.006).
Symptoms that persisted for at least 3 months were more common in the COVID than in the control group (53% vs. 37%). And patients in the COVID group reported more symptoms than those in the control group after adjustment for age, gender, smoking status, comorbidities, and timing of the survey phone call.
Levels of sleeping problems and headache were similar in the two groups.
“We have to remember that with COVID-19 came the psychosocial changes of the pandemic situation” Dr. Desgranges said.
This study suggests that some long-COVID symptoms – such as the fatigue, headache, and sleep disorders reported in the control group – could be related to the pandemic itself, which has caused psychosocial distress, Dr. Terndrup said.
Another study that looked at outpatients “has some fantastic long-term follow-up data, and shows that many patients are still engaging in rehabilitation programs nearly a year after their diagnosis,” he explained.
The COVID HOME study
That prospective longitudinal COVID HOME study, which assessed long-term symptoms in people who were never hospitalized for COVID, was presented online by Adriana Tami, MD, PhD, from the University Medical Center Groningen (the Netherlands).
The researchers visited the homes of patients to collect data, blood samples, and perform polymerase chain reaction (PCR) testing 1, 2, and 3 weeks after a diagnosis of COVID-19. If their PCR test was still positive, testing continued until week 6 or a negative test. In addition, participants completed questionnaires at week 2 and at months 3, 6 and 12 to assess fatigue, quality of life, and symptoms of depression and anxiety.
Three-month follow-up data were available for 134 of the 276 people initially enrolled in the study. Questionnaires were completed by 85 participants at 3 months, 62 participants at 6 months, and 10 participants at 12 months.
At least 40% of participants reported long-lasting symptoms at some point during follow-up, and at least 30% said they didn’t feel fully recovered at 12 months. The most common symptom was persistent fatigue, reported at 3, 6, and 12 months by at least 44% of participants. Other common symptoms – reported by at least 20% of respondents at 3, 6, and 12 months – were headache, mental or neurologic symptoms, and sleep disorders, shortness of breath, lack of smell or taste, and severe fatigue.
“We have a high proportion of nonhospitalized individuals who suffer from long COVID after more than 12 months,” Dr. Tami concluded, adding that the study is ongoing. “We have other variables that we want to look at, including duration viral shedding and serological results and variants.”
“These cohort studies are very helpful, but they can lead to inaccurate conclusions,” Dr. Terndrup cautioned.
They only provide pieces of the big picture, but they “do add to a growing body of knowledge about a significant portion of COVID patients still struggling with symptoms long after their initial infection. The symptoms can be quite variable but are dominated by both physical and mental fatigue, and tend to be worse in patients who were sicker at initial infection,” he said in an interview.
As a whole, these studies reinforce the need for treatment programs to help patients who suffer from long COVID, he added, but “I advise caution to folks suffering out there who seek ‘miracle cures’; across the world, we are collaborating to find solutions that are safe and effective.”
We are in desperate need of an equity lens in these studies.
“There is still a great deal to learn about long COVID,” said Dr. Terndrup. Data on underrepresented populations – such as Black, Indigenous, and people of color – are lacking from these and others studies, he explained. “We are in desperate need of an equity lens in these studies,” particularly in the United States, where there are “significant disparities” in the treatment of different populations.
However, “I do hope that this work can lead to a better understanding of how other viral infections can cause long-lasting symptoms,” said Dr. Terndrup.
“We have long proposed that after acute presentation, some microbes can cause chronic symptoms, like fatigue and widespread pain. Perhaps we can learn how to better care for these patients after learning from COVID’s significant impact on our societies across the globe.”
Dr. Aranda and Dr. Desgranges have disclosed no relevant financial relationships or study funding. The study by Dr. Tami’s team was funded by the University Medical Center Groningen Organization for Health Research and Development, and Connecting European Cohorts to Increase Common and Effective Response to SARS-CoV-2 Pandemic. Dr. Terndrup disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Findings from the cohort, composed of 113 COVID-19 survivors who developed ARDS after admission to a single center before to April 16, 2020, were presented online at the 31st European Congress of Clinical Microbiology & Infectious Diseases by Judit Aranda, MD, from Complex Hospitalari Moisés Broggi in Barcelona.
Median age of the participants was 64 years, and 70% were male. At least one persistent symptom was experienced during follow-up by 81% of the cohort, with 45% reporting shortness of breath, 50% reporting muscle pain, 43% reporting memory impairment, and 46% reporting physical weakness of at least 5 on a 10-point scale.
Of the 104 participants who completed a 6-minute walk test, 30% had a decrease in oxygen saturation level of at least 4%, and 5% had an initial or final level below 88%. Of the 46 participants who underwent a pulmonary function test, 15% had a forced expiratory volume in 1 second below 70%.
And of the 49% of participants with pathologic findings on chest x-ray, most were bilateral interstitial infiltrates (88%).
In addition, more than 90% of participants developed depression, anxiety, or PTSD, Dr. Aranda reported.
Not the whole picture
This study shows that sicker people – “those in intensive care units with acute respiratory distress syndrome” – are “more likely to be struggling with more severe symptoms,” said Christopher Terndrup, MD, from the division of general internal medicine and geriatrics at Oregon Health & Science University, Portland.
But a Swiss study, also presented at the meeting, “shows how even mild COVID cases can lead to debilitating symptoms,” Dr. Terndrup said in an interview.
The investigation of long-term COVID symptoms in outpatients was presented online by Florian Desgranges, MD, from Lausanne (Switzerland) University Hospital. He and his colleagues found that more than half of those with a mild to moderate disease had persistent symptoms at least 3 months after diagnosis.
The prevalence of long COVID has varied in previous research, from 15% in a study of health care workers, to 46% in a study of patients with mild COVID, 52% in a study of young COVID outpatients, and 76% in a study of patients hospitalized with COVID.
Dr. Desgranges and colleagues evaluated patients seen in an ED or outpatient clinic from February to April 2020.
The 418 patients with a confirmed COVID-19 diagnosis were compared with a control group of 89 patients who presented to the same centers during the same time frame with similar symptoms – cough, shortness of breath, or fever – but had a negative SARS-CoV-2 test.
The number of patients with comorbidities was similar in the COVID and control groups (34% vs. 36%), as was median age (41 vs. 36 years) and the prevalence of women (62% vs 64%), but the proportion of health care workers was lower in the COVID group (64% vs 82%; P =.006).
Symptoms that persisted for at least 3 months were more common in the COVID than in the control group (53% vs. 37%). And patients in the COVID group reported more symptoms than those in the control group after adjustment for age, gender, smoking status, comorbidities, and timing of the survey phone call.
Levels of sleeping problems and headache were similar in the two groups.
“We have to remember that with COVID-19 came the psychosocial changes of the pandemic situation” Dr. Desgranges said.
This study suggests that some long-COVID symptoms – such as the fatigue, headache, and sleep disorders reported in the control group – could be related to the pandemic itself, which has caused psychosocial distress, Dr. Terndrup said.
Another study that looked at outpatients “has some fantastic long-term follow-up data, and shows that many patients are still engaging in rehabilitation programs nearly a year after their diagnosis,” he explained.
The COVID HOME study
That prospective longitudinal COVID HOME study, which assessed long-term symptoms in people who were never hospitalized for COVID, was presented online by Adriana Tami, MD, PhD, from the University Medical Center Groningen (the Netherlands).
The researchers visited the homes of patients to collect data, blood samples, and perform polymerase chain reaction (PCR) testing 1, 2, and 3 weeks after a diagnosis of COVID-19. If their PCR test was still positive, testing continued until week 6 or a negative test. In addition, participants completed questionnaires at week 2 and at months 3, 6 and 12 to assess fatigue, quality of life, and symptoms of depression and anxiety.
Three-month follow-up data were available for 134 of the 276 people initially enrolled in the study. Questionnaires were completed by 85 participants at 3 months, 62 participants at 6 months, and 10 participants at 12 months.
At least 40% of participants reported long-lasting symptoms at some point during follow-up, and at least 30% said they didn’t feel fully recovered at 12 months. The most common symptom was persistent fatigue, reported at 3, 6, and 12 months by at least 44% of participants. Other common symptoms – reported by at least 20% of respondents at 3, 6, and 12 months – were headache, mental or neurologic symptoms, and sleep disorders, shortness of breath, lack of smell or taste, and severe fatigue.
“We have a high proportion of nonhospitalized individuals who suffer from long COVID after more than 12 months,” Dr. Tami concluded, adding that the study is ongoing. “We have other variables that we want to look at, including duration viral shedding and serological results and variants.”
“These cohort studies are very helpful, but they can lead to inaccurate conclusions,” Dr. Terndrup cautioned.
They only provide pieces of the big picture, but they “do add to a growing body of knowledge about a significant portion of COVID patients still struggling with symptoms long after their initial infection. The symptoms can be quite variable but are dominated by both physical and mental fatigue, and tend to be worse in patients who were sicker at initial infection,” he said in an interview.
As a whole, these studies reinforce the need for treatment programs to help patients who suffer from long COVID, he added, but “I advise caution to folks suffering out there who seek ‘miracle cures’; across the world, we are collaborating to find solutions that are safe and effective.”
We are in desperate need of an equity lens in these studies.
“There is still a great deal to learn about long COVID,” said Dr. Terndrup. Data on underrepresented populations – such as Black, Indigenous, and people of color – are lacking from these and others studies, he explained. “We are in desperate need of an equity lens in these studies,” particularly in the United States, where there are “significant disparities” in the treatment of different populations.
However, “I do hope that this work can lead to a better understanding of how other viral infections can cause long-lasting symptoms,” said Dr. Terndrup.
“We have long proposed that after acute presentation, some microbes can cause chronic symptoms, like fatigue and widespread pain. Perhaps we can learn how to better care for these patients after learning from COVID’s significant impact on our societies across the globe.”
Dr. Aranda and Dr. Desgranges have disclosed no relevant financial relationships or study funding. The study by Dr. Tami’s team was funded by the University Medical Center Groningen Organization for Health Research and Development, and Connecting European Cohorts to Increase Common and Effective Response to SARS-CoV-2 Pandemic. Dr. Terndrup disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FROM EUROPEAN CONGRESS OF CLINICAL MICROBIOLOGY & INFECTIOUS DISEASES
The febrile infant: New AAP guidance for the first 2 months of life
Sixteen years in the making, the American Academy of Pediatrics just released a new clinical practice guideline (CPG), “Evaluation and Management of Well-Appearing Febrile Infants 8-60 Days Old”. The recommendations were derived from interpretations of sequential studies in young, febrile, but well-appearing infants that covered invasive bacterial infection (IBI) incidence, diagnostic modalities, and treatment during the first 2 months of life, further refining approaches to evaluation and empirical treatment.
Pediatricians have long had solid information to help assess the risk for IBI among febrile infants aged 0-3 months, but there has been an ongoing desire to further refine the suggested evaluation of these very young infants. A study of febrile infants from the Pediatric Research in Office Settings network along with subsequent evidence has identified the first 3 weeks of life as the period of highest risk for IBI, with risk declining in a graded fashion aged between 22 and 56 days.
Critical caveats
First, some caveats. Infants 0-7 days are not addressed in the CPG, and all should be treated as high risk and receive full IBI evaluation according to newborn protocols. Second, the recommendations apply only to “well-appearing” infants. Any ill-appearing infant should be treated as high risk and receive full IBI evaluation and begun on empirical antimicrobials. Third, even though the CPG deals with infants as young as 8-21 days old, the recommendations are to treat all infants in this age group as high risk, even if well-appearing, and complete full IBI evaluation and empirical therapy while awaiting results. Fourth, these guidelines apply only to infants born at 37 weeks’ gestation or more. Finally, the new CPG action statements are meant to be recommendations rather than a standard of medical care, leaving some leeway for clinician interpretation of individual patient scenarios. Where appropriate, parents’ values and preferences should be incorporated as part of shared decision-making.
The CPG divides young, febrile infants into three cohorts based on age:
- 8-21 days old
- 22-28 days old
- 29-60 days old
Age 8-21 days
For well-appearing febrile infants 8-21 days old, the CPG recommends a complete IBI evaluation that includes urine, blood, and cerebrospinal fluid (CSF) for culture, approaching all infants in this cohort as high risk. Inflammatory markers may be obtained, but the evidence is not comprehensive enough to evaluate their role in decision-making for this age group. A two-step urine evaluation method (urine analysis followed by culture if the urine analysis looks concerning) is not recommended for infants aged 8-21 days. Urine samples for culture from these young infants should be obtained by catheterization or suprapubic aspiration.
The CPG recommends drawing blood cultures and CSF by lumbar puncture from this cohort. These infants should be admitted to the hospital, treated empirically with antimicrobials, and actively monitored. However, if the cultures are negative at 24-36 hours, the clinician should discontinue antimicrobials and discharge the infant if there is no other reason for continued hospitalization.
Age 22-28 days
Well-appearing, febrile infants 22-28 days old are in an intermediate-risk zone. The recommendation for infants in this cohort is to obtain a urine specimen by catheterization or suprapubic aspiration for both urine analysis and culture. Clinicians may consider obtaining urine samples for analysis noninvasively (e.g., urine bag) in this cohort, but this is not the preferred method.
Blood culture should be obtained from all infants in this group. Inflammatory markers can help clinicians identify infants at greater risk for IBI, including meningitis. Previous data suggested that inflammatory markers such as serum white blood cell counts greater than 11,000/mcL, a serum absolute neutrophil count of greater than 4,000/mcL, and elevated C-reactive protein and procalcitonin levels could help providers identify febrile infants with true IBI. A 2008 study demonstrated that procalcitonin had the best receiver operating characteristic curve in regard to predicting IBI in young febrile infants. Other research backed up that finding and identified cutoff values for procalcitonin levels greater than 1.0 ng/mL. The CPG recommends considering a procalcitonin value of 0.5 ng/mL or higher as positive, indicating that the infant is at greater risk for IBI and potentially should undergo an expanded IBI workup. Therefore, in infants aged 22-28 days, inflammatory markers can play a role in deciding whether to perform a lumbar puncture.
Many more nuanced recommendations for whether to and how to empirically treat with antimicrobials in this cohort can be found in the CPG, including whether to manage in the hospital or at home. Treatment recommendations vary greatly for this cohort on the basis of the tests obtained and whether tests were positive or negative at the initial evaluation.
Age 29-60 days
The CPG will be most helpful when clinicians are faced with well-appearing, febrile infants in the 29- to 60-day age group. As with the other groups, a urine evaluation is recommended; however, the CPG suggests that the two-step approach – obtaining a urine analysis by a noninvasive method and only obtaining culture if the urine analysis is positive – is reasonable. This means that a bag or free-flowing urine specimen would be appropriate for urinalysis, followed by catheterization/suprapubic aspiration if a culture is necessary. This would save approximately 90% of infants from invasive urine collection. Regardless, only catheter or suprapubic specimens are appropriate for urine culture.
The CPG also recommends that clinicians obtain blood culture on all of these infants. Inflammatory markers should be assessed in this cohort because avoiding lumbar puncture for CSF culture would be appropriate in this cohort if the inflammatory markers are negative. If CSF is obtained in this age cohort, enterovirus testing should be added to the testing regimen. Again, for any infant considered at higher risk for IBI on the basis of screening tests, the CPG recommends a 24- to 36-hour rule-out period with empirical antimicrobial treatment and active monitoring in the hospital.
Summary
The recommended approach for febrile infants 8-21 days old is relatively aggressive, with urine, blood, and CSF evaluation for IBI. Clinicians gain some leeway for infants age 22-28 days, but the guidelines recommend a more flexible approach to evaluating well-appearing, febrile infants age 29-60 days, when a two-step urine evaluation and inflammatory marker assessment can help clinicians and parents have a better discussion about the risk-benefit trade-offs of more aggressive testing and empirical treatment.
The author would like to thank Ken Roberts, MD, for his review and helpful comments on this summary of the CPG highlights. Summary points of the CPG were presented by the writing group at the 2021 Pediatric Academic Societies meeting.
William T. Basco, Jr, MD, MS, is a professor of pediatrics at the Medical University of South Carolina, Charleston, and director of the division of general pediatrics. He is an active health services researcher and has published more than 60 manuscripts in the peer-reviewed literature.
A version of this article first appeared on Medscape.com.
Sixteen years in the making, the American Academy of Pediatrics just released a new clinical practice guideline (CPG), “Evaluation and Management of Well-Appearing Febrile Infants 8-60 Days Old”. The recommendations were derived from interpretations of sequential studies in young, febrile, but well-appearing infants that covered invasive bacterial infection (IBI) incidence, diagnostic modalities, and treatment during the first 2 months of life, further refining approaches to evaluation and empirical treatment.
Pediatricians have long had solid information to help assess the risk for IBI among febrile infants aged 0-3 months, but there has been an ongoing desire to further refine the suggested evaluation of these very young infants. A study of febrile infants from the Pediatric Research in Office Settings network along with subsequent evidence has identified the first 3 weeks of life as the period of highest risk for IBI, with risk declining in a graded fashion aged between 22 and 56 days.
Critical caveats
First, some caveats. Infants 0-7 days are not addressed in the CPG, and all should be treated as high risk and receive full IBI evaluation according to newborn protocols. Second, the recommendations apply only to “well-appearing” infants. Any ill-appearing infant should be treated as high risk and receive full IBI evaluation and begun on empirical antimicrobials. Third, even though the CPG deals with infants as young as 8-21 days old, the recommendations are to treat all infants in this age group as high risk, even if well-appearing, and complete full IBI evaluation and empirical therapy while awaiting results. Fourth, these guidelines apply only to infants born at 37 weeks’ gestation or more. Finally, the new CPG action statements are meant to be recommendations rather than a standard of medical care, leaving some leeway for clinician interpretation of individual patient scenarios. Where appropriate, parents’ values and preferences should be incorporated as part of shared decision-making.
The CPG divides young, febrile infants into three cohorts based on age:
- 8-21 days old
- 22-28 days old
- 29-60 days old
Age 8-21 days
For well-appearing febrile infants 8-21 days old, the CPG recommends a complete IBI evaluation that includes urine, blood, and cerebrospinal fluid (CSF) for culture, approaching all infants in this cohort as high risk. Inflammatory markers may be obtained, but the evidence is not comprehensive enough to evaluate their role in decision-making for this age group. A two-step urine evaluation method (urine analysis followed by culture if the urine analysis looks concerning) is not recommended for infants aged 8-21 days. Urine samples for culture from these young infants should be obtained by catheterization or suprapubic aspiration.
The CPG recommends drawing blood cultures and CSF by lumbar puncture from this cohort. These infants should be admitted to the hospital, treated empirically with antimicrobials, and actively monitored. However, if the cultures are negative at 24-36 hours, the clinician should discontinue antimicrobials and discharge the infant if there is no other reason for continued hospitalization.
Age 22-28 days
Well-appearing, febrile infants 22-28 days old are in an intermediate-risk zone. The recommendation for infants in this cohort is to obtain a urine specimen by catheterization or suprapubic aspiration for both urine analysis and culture. Clinicians may consider obtaining urine samples for analysis noninvasively (e.g., urine bag) in this cohort, but this is not the preferred method.
Blood culture should be obtained from all infants in this group. Inflammatory markers can help clinicians identify infants at greater risk for IBI, including meningitis. Previous data suggested that inflammatory markers such as serum white blood cell counts greater than 11,000/mcL, a serum absolute neutrophil count of greater than 4,000/mcL, and elevated C-reactive protein and procalcitonin levels could help providers identify febrile infants with true IBI. A 2008 study demonstrated that procalcitonin had the best receiver operating characteristic curve in regard to predicting IBI in young febrile infants. Other research backed up that finding and identified cutoff values for procalcitonin levels greater than 1.0 ng/mL. The CPG recommends considering a procalcitonin value of 0.5 ng/mL or higher as positive, indicating that the infant is at greater risk for IBI and potentially should undergo an expanded IBI workup. Therefore, in infants aged 22-28 days, inflammatory markers can play a role in deciding whether to perform a lumbar puncture.
Many more nuanced recommendations for whether to and how to empirically treat with antimicrobials in this cohort can be found in the CPG, including whether to manage in the hospital or at home. Treatment recommendations vary greatly for this cohort on the basis of the tests obtained and whether tests were positive or negative at the initial evaluation.
Age 29-60 days
The CPG will be most helpful when clinicians are faced with well-appearing, febrile infants in the 29- to 60-day age group. As with the other groups, a urine evaluation is recommended; however, the CPG suggests that the two-step approach – obtaining a urine analysis by a noninvasive method and only obtaining culture if the urine analysis is positive – is reasonable. This means that a bag or free-flowing urine specimen would be appropriate for urinalysis, followed by catheterization/suprapubic aspiration if a culture is necessary. This would save approximately 90% of infants from invasive urine collection. Regardless, only catheter or suprapubic specimens are appropriate for urine culture.
The CPG also recommends that clinicians obtain blood culture on all of these infants. Inflammatory markers should be assessed in this cohort because avoiding lumbar puncture for CSF culture would be appropriate in this cohort if the inflammatory markers are negative. If CSF is obtained in this age cohort, enterovirus testing should be added to the testing regimen. Again, for any infant considered at higher risk for IBI on the basis of screening tests, the CPG recommends a 24- to 36-hour rule-out period with empirical antimicrobial treatment and active monitoring in the hospital.
Summary
The recommended approach for febrile infants 8-21 days old is relatively aggressive, with urine, blood, and CSF evaluation for IBI. Clinicians gain some leeway for infants age 22-28 days, but the guidelines recommend a more flexible approach to evaluating well-appearing, febrile infants age 29-60 days, when a two-step urine evaluation and inflammatory marker assessment can help clinicians and parents have a better discussion about the risk-benefit trade-offs of more aggressive testing and empirical treatment.
The author would like to thank Ken Roberts, MD, for his review and helpful comments on this summary of the CPG highlights. Summary points of the CPG were presented by the writing group at the 2021 Pediatric Academic Societies meeting.
William T. Basco, Jr, MD, MS, is a professor of pediatrics at the Medical University of South Carolina, Charleston, and director of the division of general pediatrics. He is an active health services researcher and has published more than 60 manuscripts in the peer-reviewed literature.
A version of this article first appeared on Medscape.com.
Sixteen years in the making, the American Academy of Pediatrics just released a new clinical practice guideline (CPG), “Evaluation and Management of Well-Appearing Febrile Infants 8-60 Days Old”. The recommendations were derived from interpretations of sequential studies in young, febrile, but well-appearing infants that covered invasive bacterial infection (IBI) incidence, diagnostic modalities, and treatment during the first 2 months of life, further refining approaches to evaluation and empirical treatment.
Pediatricians have long had solid information to help assess the risk for IBI among febrile infants aged 0-3 months, but there has been an ongoing desire to further refine the suggested evaluation of these very young infants. A study of febrile infants from the Pediatric Research in Office Settings network along with subsequent evidence has identified the first 3 weeks of life as the period of highest risk for IBI, with risk declining in a graded fashion aged between 22 and 56 days.
Critical caveats
First, some caveats. Infants 0-7 days are not addressed in the CPG, and all should be treated as high risk and receive full IBI evaluation according to newborn protocols. Second, the recommendations apply only to “well-appearing” infants. Any ill-appearing infant should be treated as high risk and receive full IBI evaluation and begun on empirical antimicrobials. Third, even though the CPG deals with infants as young as 8-21 days old, the recommendations are to treat all infants in this age group as high risk, even if well-appearing, and complete full IBI evaluation and empirical therapy while awaiting results. Fourth, these guidelines apply only to infants born at 37 weeks’ gestation or more. Finally, the new CPG action statements are meant to be recommendations rather than a standard of medical care, leaving some leeway for clinician interpretation of individual patient scenarios. Where appropriate, parents’ values and preferences should be incorporated as part of shared decision-making.
The CPG divides young, febrile infants into three cohorts based on age:
- 8-21 days old
- 22-28 days old
- 29-60 days old
Age 8-21 days
For well-appearing febrile infants 8-21 days old, the CPG recommends a complete IBI evaluation that includes urine, blood, and cerebrospinal fluid (CSF) for culture, approaching all infants in this cohort as high risk. Inflammatory markers may be obtained, but the evidence is not comprehensive enough to evaluate their role in decision-making for this age group. A two-step urine evaluation method (urine analysis followed by culture if the urine analysis looks concerning) is not recommended for infants aged 8-21 days. Urine samples for culture from these young infants should be obtained by catheterization or suprapubic aspiration.
The CPG recommends drawing blood cultures and CSF by lumbar puncture from this cohort. These infants should be admitted to the hospital, treated empirically with antimicrobials, and actively monitored. However, if the cultures are negative at 24-36 hours, the clinician should discontinue antimicrobials and discharge the infant if there is no other reason for continued hospitalization.
Age 22-28 days
Well-appearing, febrile infants 22-28 days old are in an intermediate-risk zone. The recommendation for infants in this cohort is to obtain a urine specimen by catheterization or suprapubic aspiration for both urine analysis and culture. Clinicians may consider obtaining urine samples for analysis noninvasively (e.g., urine bag) in this cohort, but this is not the preferred method.
Blood culture should be obtained from all infants in this group. Inflammatory markers can help clinicians identify infants at greater risk for IBI, including meningitis. Previous data suggested that inflammatory markers such as serum white blood cell counts greater than 11,000/mcL, a serum absolute neutrophil count of greater than 4,000/mcL, and elevated C-reactive protein and procalcitonin levels could help providers identify febrile infants with true IBI. A 2008 study demonstrated that procalcitonin had the best receiver operating characteristic curve in regard to predicting IBI in young febrile infants. Other research backed up that finding and identified cutoff values for procalcitonin levels greater than 1.0 ng/mL. The CPG recommends considering a procalcitonin value of 0.5 ng/mL or higher as positive, indicating that the infant is at greater risk for IBI and potentially should undergo an expanded IBI workup. Therefore, in infants aged 22-28 days, inflammatory markers can play a role in deciding whether to perform a lumbar puncture.
Many more nuanced recommendations for whether to and how to empirically treat with antimicrobials in this cohort can be found in the CPG, including whether to manage in the hospital or at home. Treatment recommendations vary greatly for this cohort on the basis of the tests obtained and whether tests were positive or negative at the initial evaluation.
Age 29-60 days
The CPG will be most helpful when clinicians are faced with well-appearing, febrile infants in the 29- to 60-day age group. As with the other groups, a urine evaluation is recommended; however, the CPG suggests that the two-step approach – obtaining a urine analysis by a noninvasive method and only obtaining culture if the urine analysis is positive – is reasonable. This means that a bag or free-flowing urine specimen would be appropriate for urinalysis, followed by catheterization/suprapubic aspiration if a culture is necessary. This would save approximately 90% of infants from invasive urine collection. Regardless, only catheter or suprapubic specimens are appropriate for urine culture.
The CPG also recommends that clinicians obtain blood culture on all of these infants. Inflammatory markers should be assessed in this cohort because avoiding lumbar puncture for CSF culture would be appropriate in this cohort if the inflammatory markers are negative. If CSF is obtained in this age cohort, enterovirus testing should be added to the testing regimen. Again, for any infant considered at higher risk for IBI on the basis of screening tests, the CPG recommends a 24- to 36-hour rule-out period with empirical antimicrobial treatment and active monitoring in the hospital.
Summary
The recommended approach for febrile infants 8-21 days old is relatively aggressive, with urine, blood, and CSF evaluation for IBI. Clinicians gain some leeway for infants age 22-28 days, but the guidelines recommend a more flexible approach to evaluating well-appearing, febrile infants age 29-60 days, when a two-step urine evaluation and inflammatory marker assessment can help clinicians and parents have a better discussion about the risk-benefit trade-offs of more aggressive testing and empirical treatment.
The author would like to thank Ken Roberts, MD, for his review and helpful comments on this summary of the CPG highlights. Summary points of the CPG were presented by the writing group at the 2021 Pediatric Academic Societies meeting.
William T. Basco, Jr, MD, MS, is a professor of pediatrics at the Medical University of South Carolina, Charleston, and director of the division of general pediatrics. He is an active health services researcher and has published more than 60 manuscripts in the peer-reviewed literature.
A version of this article first appeared on Medscape.com.
Treatment of opioid use disorder with buprenorphine and methadone effective but underutilized
Background: Opioid use disorder (OUD) is a chronic disease with a high health care and societal burden from overdose and complications requiring hospitalization. Though clinical trials demonstrate effectiveness of methadone and buprenorphine, most patients do not have access to these medications.
Study design: Retrospective comparative effectiveness study.
Setting: Nationwide claims database of commercial and Medicare Advantage Enrollees.
Synopsis: A total of 40,885 individuals aged 16 years or older with OUD were studied in an intent-to-treat analysis of six unique treatment pathways. Though used in just 12.5% of patients, only treatment with buprenorphine or methadone was protective against overdose at 3 and 12 months, compared with no treatment. Additionally, these medications and nonintensive behavioral health counseling were associated with lower incidence of acute care episodes from complications of opioid use. Notably, those treated with buprenorphine or methadone for more than 6 months received the greatest benefit. With use of only health care encounters, the results may underestimate incidence of complications of ongoing opioid misuse.
Bottom line: Buprenorphine and methadone for OUD were associated with reduced overdose and opioid-related morbidity, compared with opioid antagonist therapy, inpatient treatment, or intensive outpatient behavioral interventions and should be considered a first-line treatment.
Citation: Wakeman SE et al. Comparative effectiveness of different treatment pathways for opioid use disorder. JAMA Netw Open. 2020 Feb 5;3(2):e1920622. doi: 10.1001/jamanetworkopen.2019.20622.
Dr. Inofuentes is assistant professor of medicine, section of hospital medicine, at the University of Virginia School of Medicine, Charlottesville.
Background: Opioid use disorder (OUD) is a chronic disease with a high health care and societal burden from overdose and complications requiring hospitalization. Though clinical trials demonstrate effectiveness of methadone and buprenorphine, most patients do not have access to these medications.
Study design: Retrospective comparative effectiveness study.
Setting: Nationwide claims database of commercial and Medicare Advantage Enrollees.
Synopsis: A total of 40,885 individuals aged 16 years or older with OUD were studied in an intent-to-treat analysis of six unique treatment pathways. Though used in just 12.5% of patients, only treatment with buprenorphine or methadone was protective against overdose at 3 and 12 months, compared with no treatment. Additionally, these medications and nonintensive behavioral health counseling were associated with lower incidence of acute care episodes from complications of opioid use. Notably, those treated with buprenorphine or methadone for more than 6 months received the greatest benefit. With use of only health care encounters, the results may underestimate incidence of complications of ongoing opioid misuse.
Bottom line: Buprenorphine and methadone for OUD were associated with reduced overdose and opioid-related morbidity, compared with opioid antagonist therapy, inpatient treatment, or intensive outpatient behavioral interventions and should be considered a first-line treatment.
Citation: Wakeman SE et al. Comparative effectiveness of different treatment pathways for opioid use disorder. JAMA Netw Open. 2020 Feb 5;3(2):e1920622. doi: 10.1001/jamanetworkopen.2019.20622.
Dr. Inofuentes is assistant professor of medicine, section of hospital medicine, at the University of Virginia School of Medicine, Charlottesville.
Background: Opioid use disorder (OUD) is a chronic disease with a high health care and societal burden from overdose and complications requiring hospitalization. Though clinical trials demonstrate effectiveness of methadone and buprenorphine, most patients do not have access to these medications.
Study design: Retrospective comparative effectiveness study.
Setting: Nationwide claims database of commercial and Medicare Advantage Enrollees.
Synopsis: A total of 40,885 individuals aged 16 years or older with OUD were studied in an intent-to-treat analysis of six unique treatment pathways. Though used in just 12.5% of patients, only treatment with buprenorphine or methadone was protective against overdose at 3 and 12 months, compared with no treatment. Additionally, these medications and nonintensive behavioral health counseling were associated with lower incidence of acute care episodes from complications of opioid use. Notably, those treated with buprenorphine or methadone for more than 6 months received the greatest benefit. With use of only health care encounters, the results may underestimate incidence of complications of ongoing opioid misuse.
Bottom line: Buprenorphine and methadone for OUD were associated with reduced overdose and opioid-related morbidity, compared with opioid antagonist therapy, inpatient treatment, or intensive outpatient behavioral interventions and should be considered a first-line treatment.
Citation: Wakeman SE et al. Comparative effectiveness of different treatment pathways for opioid use disorder. JAMA Netw Open. 2020 Feb 5;3(2):e1920622. doi: 10.1001/jamanetworkopen.2019.20622.
Dr. Inofuentes is assistant professor of medicine, section of hospital medicine, at the University of Virginia School of Medicine, Charlottesville.
Large remdesivir study finds no COVID-19 survival benefit
A lack of consensus in the evidence regarding the antiviral remdesivir (Veklury) to treat people with COVID-19 continues, leaving clinicians without clear direction on one of the few treatments for the illness approved under U.S. Food and Drug Administration emergency use authorization.
The latest research comes from Michael Ohl, MD, MSPH, and colleagues, who studied a large group of VA patients hospitalized with COVID-19. Compared with a matched group of veterans who did not receive the antiviral, remdesivir did not significantly improve survival.
The percentages were close: 12.2% of patients in the remdesivir group died within 30 days compared with 10.6% of those in the control group.
At the same time, the retrospective cohort study showed remdesivir was associated with more days in the hospital.
“There is still uncertainty about the role of remdesivir in treatment for people hospitalized with COVID-19,” Dr. Ohl told this news organization.
“It is reasonable to follow the CDC and Infectious Diseases Society of America guidelines for remdesivir use, “but clinicians should avoid admitting people or keeping people in the hospital solely to receive remdesivir if they do not meet other criteria for hospitalization,” said Dr. Ohl, lead author and an infectious disease specialist at the Center for Access & Delivery Research and Evaluation, Iowa City Veterans Affairs (VA) Health Care System.
The study was published online July 15 in JAMA Network Open.
Sticking with the official protocol?
The longer a hospital stays associated with remdesivir, a median 6 days versus 3 days, could be a result of treating people for 5 or 10 days with the antiviral agent. In other words, it is “possible that clinicians were not discharging patients who otherwise met the criteria for hospital discharge until the remdesivir course was completed,” Dr. Ohl and colleagues note.
Not doing so, they add, could have resulted in “increased used of scarce hospital beds during the pandemic.”
“The recommended remdesivir treatment course is a somewhat arbitrary 5 or 10 days depending on illness severity, and remdesivir is currently available only as an intravenous formulation for use in health care settings,” they add.
This is the “most likely explanation,” notes Gio J. Baracco, MD, in an invited commentary accompanying the study.
At the time of the study, use of remdesivir also required patient consent, close adverse event monitoring, and ongoing testing, Dr. Baracco notes.
He added that an option to discharge patients earlier if they responded to treatment might have been lost in translation from clinical trial protocol to real-world use in the VA system.
While a large clinical trial protocol called for the remdesivir infusions to be stopped early if the patient met the primary outcome and was ready to be discharged, “this detail was not adequately translated to the clinicians treating these patients,” added Dr. Baracco, who’s with the Division of Infectious Diseases at the University of Miami Miller School of Medicine and the Miami VA Healthcare System.
Conflicting evidence
Another large study, the World Health Organization Solidarity Trial, found remdesivir was not associated with shorter hospital stays or improved survival compared with standard of care. For this reason, the WHO recommends against use of remdesivir.
In contrast, the double-blind, randomized Adaptive COVID-19 Treatment Trial (ACTT-1) linked remdesivir treatment to shorter stays in the hospital, a median 10 days versus 15 days in a placebo group.
The FDA included the 2020 ACTT-1 in its consideration for remdesivir emergency use authorization. The FDA issued the EUA in May 2020, followed by full approval as the first treatment indicated for COVID-19 in October.
ACTT-1 lead author John H. Beigel, MD, and colleagues also looked at the death rates for remdesivir versus placebo.
By day 15, the proportion of people who died was 6.7% in the remdesivir group versus 11%. By day 29, the rate was 11.4% among those who received the antiviral versus 15.2% among those who did not.
When asked why the VA and ACTT-1 studies yielded different results, Dr. Beigel cited two reasons. The timing was different, with the VA study starting after the remdesivir EUA was issued, and ACTT-1 findings were announced.
“So at that point, clinicians understood those populations most likely to benefit from remdesivir. The use of remdesivir likely did not occur at random; it was likely to be more commonly used in those who were sicker or at higher risk for poor outcomes,” said Dr. Beigel, associate director for clinical research in the Division of Microbiology and Infectious Diseases at the National Institute of Allergy and Infectious Diseases (NIAID).
In addition, the studies evaluated very different populations, he said. The differences in median duration of hospitalization between the trials reflects this, Dr. Beigel added.
Furthermore, when asked if he thinks the new evidence should affect clinical use of remdesivir, Dr. Beigel replied, “No. Observational studies, even with adjustments such as propensity score matching, are not equivalent levels of proof compared to randomized trials.”
Study details
Dr. Ohl and colleagues identified patients admitted to one of 123 VA hospitals for the first time for COVID-19 from May 1 to Oct. 8, 2020. Each had a PCR-confirmed SARS-CoV-2 infection. The researchers then compared 1,172 patients receiving remdesivir to another 1,172 patients not receiving the agent.
Those receiving remdesivir were more likely to be older, White, have chronic obstructive pulmonary disease and have more severe COVID-19. A total 94% of the remdesivir group were men.
“Over 90% of the people included in VA study were men, mostly over the age of 60,” Dr. Ohl said when asked how generalizable the findings would be to a non-VA population.
“There is no obvious biological reason that remdesivir should have different effects in men and women, but we should be cautious about extrapolating study findings to women and younger individuals,” he added.
Limitations of the study include its observational design, which makes unadjusted confounding based on illness severity a possibility. In addition, the investigators were unable to identify specific subgroups that might benefit from remdesivir treatment.
The data did suggest that remdesivir was more effective earlier in the course of disease when patients required supplemental oxygen and before need for mechanical ventilation.
Dr. Baracco pointed out the contradictory findings in his commentary: “The real-life application of a drug promising to hasten discharge from the hospital as its primary beneficial outcome must include an assessment of how easy it is to do so and make it clear that once a patient reaches that point, they can discontinue the drug.”
“The paradoxical findings in the study by Dr. Ohl et al. compared with the study used for its authorization illustrate this point very clearly,” he adds.
Dr. Ohl reported receiving grants from Veterans Affairs Health Services Research and Development during the conduct of the study and consulting for Gilead Pharmaceuticals outside the submitted work. Dr. Baracco reported receiving salary support from the U.S. Department of Veterans Affairs. Dr. Beigel has disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
A lack of consensus in the evidence regarding the antiviral remdesivir (Veklury) to treat people with COVID-19 continues, leaving clinicians without clear direction on one of the few treatments for the illness approved under U.S. Food and Drug Administration emergency use authorization.
The latest research comes from Michael Ohl, MD, MSPH, and colleagues, who studied a large group of VA patients hospitalized with COVID-19. Compared with a matched group of veterans who did not receive the antiviral, remdesivir did not significantly improve survival.
The percentages were close: 12.2% of patients in the remdesivir group died within 30 days compared with 10.6% of those in the control group.
At the same time, the retrospective cohort study showed remdesivir was associated with more days in the hospital.
“There is still uncertainty about the role of remdesivir in treatment for people hospitalized with COVID-19,” Dr. Ohl told this news organization.
“It is reasonable to follow the CDC and Infectious Diseases Society of America guidelines for remdesivir use, “but clinicians should avoid admitting people or keeping people in the hospital solely to receive remdesivir if they do not meet other criteria for hospitalization,” said Dr. Ohl, lead author and an infectious disease specialist at the Center for Access & Delivery Research and Evaluation, Iowa City Veterans Affairs (VA) Health Care System.
The study was published online July 15 in JAMA Network Open.
Sticking with the official protocol?
The longer a hospital stays associated with remdesivir, a median 6 days versus 3 days, could be a result of treating people for 5 or 10 days with the antiviral agent. In other words, it is “possible that clinicians were not discharging patients who otherwise met the criteria for hospital discharge until the remdesivir course was completed,” Dr. Ohl and colleagues note.
Not doing so, they add, could have resulted in “increased used of scarce hospital beds during the pandemic.”
“The recommended remdesivir treatment course is a somewhat arbitrary 5 or 10 days depending on illness severity, and remdesivir is currently available only as an intravenous formulation for use in health care settings,” they add.
This is the “most likely explanation,” notes Gio J. Baracco, MD, in an invited commentary accompanying the study.
At the time of the study, use of remdesivir also required patient consent, close adverse event monitoring, and ongoing testing, Dr. Baracco notes.
He added that an option to discharge patients earlier if they responded to treatment might have been lost in translation from clinical trial protocol to real-world use in the VA system.
While a large clinical trial protocol called for the remdesivir infusions to be stopped early if the patient met the primary outcome and was ready to be discharged, “this detail was not adequately translated to the clinicians treating these patients,” added Dr. Baracco, who’s with the Division of Infectious Diseases at the University of Miami Miller School of Medicine and the Miami VA Healthcare System.
Conflicting evidence
Another large study, the World Health Organization Solidarity Trial, found remdesivir was not associated with shorter hospital stays or improved survival compared with standard of care. For this reason, the WHO recommends against use of remdesivir.
In contrast, the double-blind, randomized Adaptive COVID-19 Treatment Trial (ACTT-1) linked remdesivir treatment to shorter stays in the hospital, a median 10 days versus 15 days in a placebo group.
The FDA included the 2020 ACTT-1 in its consideration for remdesivir emergency use authorization. The FDA issued the EUA in May 2020, followed by full approval as the first treatment indicated for COVID-19 in October.
ACTT-1 lead author John H. Beigel, MD, and colleagues also looked at the death rates for remdesivir versus placebo.
By day 15, the proportion of people who died was 6.7% in the remdesivir group versus 11%. By day 29, the rate was 11.4% among those who received the antiviral versus 15.2% among those who did not.
When asked why the VA and ACTT-1 studies yielded different results, Dr. Beigel cited two reasons. The timing was different, with the VA study starting after the remdesivir EUA was issued, and ACTT-1 findings were announced.
“So at that point, clinicians understood those populations most likely to benefit from remdesivir. The use of remdesivir likely did not occur at random; it was likely to be more commonly used in those who were sicker or at higher risk for poor outcomes,” said Dr. Beigel, associate director for clinical research in the Division of Microbiology and Infectious Diseases at the National Institute of Allergy and Infectious Diseases (NIAID).
In addition, the studies evaluated very different populations, he said. The differences in median duration of hospitalization between the trials reflects this, Dr. Beigel added.
Furthermore, when asked if he thinks the new evidence should affect clinical use of remdesivir, Dr. Beigel replied, “No. Observational studies, even with adjustments such as propensity score matching, are not equivalent levels of proof compared to randomized trials.”
Study details
Dr. Ohl and colleagues identified patients admitted to one of 123 VA hospitals for the first time for COVID-19 from May 1 to Oct. 8, 2020. Each had a PCR-confirmed SARS-CoV-2 infection. The researchers then compared 1,172 patients receiving remdesivir to another 1,172 patients not receiving the agent.
Those receiving remdesivir were more likely to be older, White, have chronic obstructive pulmonary disease and have more severe COVID-19. A total 94% of the remdesivir group were men.
“Over 90% of the people included in VA study were men, mostly over the age of 60,” Dr. Ohl said when asked how generalizable the findings would be to a non-VA population.
“There is no obvious biological reason that remdesivir should have different effects in men and women, but we should be cautious about extrapolating study findings to women and younger individuals,” he added.
Limitations of the study include its observational design, which makes unadjusted confounding based on illness severity a possibility. In addition, the investigators were unable to identify specific subgroups that might benefit from remdesivir treatment.
The data did suggest that remdesivir was more effective earlier in the course of disease when patients required supplemental oxygen and before need for mechanical ventilation.
Dr. Baracco pointed out the contradictory findings in his commentary: “The real-life application of a drug promising to hasten discharge from the hospital as its primary beneficial outcome must include an assessment of how easy it is to do so and make it clear that once a patient reaches that point, they can discontinue the drug.”
“The paradoxical findings in the study by Dr. Ohl et al. compared with the study used for its authorization illustrate this point very clearly,” he adds.
Dr. Ohl reported receiving grants from Veterans Affairs Health Services Research and Development during the conduct of the study and consulting for Gilead Pharmaceuticals outside the submitted work. Dr. Baracco reported receiving salary support from the U.S. Department of Veterans Affairs. Dr. Beigel has disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
A lack of consensus in the evidence regarding the antiviral remdesivir (Veklury) to treat people with COVID-19 continues, leaving clinicians without clear direction on one of the few treatments for the illness approved under U.S. Food and Drug Administration emergency use authorization.
The latest research comes from Michael Ohl, MD, MSPH, and colleagues, who studied a large group of VA patients hospitalized with COVID-19. Compared with a matched group of veterans who did not receive the antiviral, remdesivir did not significantly improve survival.
The percentages were close: 12.2% of patients in the remdesivir group died within 30 days compared with 10.6% of those in the control group.
At the same time, the retrospective cohort study showed remdesivir was associated with more days in the hospital.
“There is still uncertainty about the role of remdesivir in treatment for people hospitalized with COVID-19,” Dr. Ohl told this news organization.
“It is reasonable to follow the CDC and Infectious Diseases Society of America guidelines for remdesivir use, “but clinicians should avoid admitting people or keeping people in the hospital solely to receive remdesivir if they do not meet other criteria for hospitalization,” said Dr. Ohl, lead author and an infectious disease specialist at the Center for Access & Delivery Research and Evaluation, Iowa City Veterans Affairs (VA) Health Care System.
The study was published online July 15 in JAMA Network Open.
Sticking with the official protocol?
The longer a hospital stays associated with remdesivir, a median 6 days versus 3 days, could be a result of treating people for 5 or 10 days with the antiviral agent. In other words, it is “possible that clinicians were not discharging patients who otherwise met the criteria for hospital discharge until the remdesivir course was completed,” Dr. Ohl and colleagues note.
Not doing so, they add, could have resulted in “increased used of scarce hospital beds during the pandemic.”
“The recommended remdesivir treatment course is a somewhat arbitrary 5 or 10 days depending on illness severity, and remdesivir is currently available only as an intravenous formulation for use in health care settings,” they add.
This is the “most likely explanation,” notes Gio J. Baracco, MD, in an invited commentary accompanying the study.
At the time of the study, use of remdesivir also required patient consent, close adverse event monitoring, and ongoing testing, Dr. Baracco notes.
He added that an option to discharge patients earlier if they responded to treatment might have been lost in translation from clinical trial protocol to real-world use in the VA system.
While a large clinical trial protocol called for the remdesivir infusions to be stopped early if the patient met the primary outcome and was ready to be discharged, “this detail was not adequately translated to the clinicians treating these patients,” added Dr. Baracco, who’s with the Division of Infectious Diseases at the University of Miami Miller School of Medicine and the Miami VA Healthcare System.
Conflicting evidence
Another large study, the World Health Organization Solidarity Trial, found remdesivir was not associated with shorter hospital stays or improved survival compared with standard of care. For this reason, the WHO recommends against use of remdesivir.
In contrast, the double-blind, randomized Adaptive COVID-19 Treatment Trial (ACTT-1) linked remdesivir treatment to shorter stays in the hospital, a median 10 days versus 15 days in a placebo group.
The FDA included the 2020 ACTT-1 in its consideration for remdesivir emergency use authorization. The FDA issued the EUA in May 2020, followed by full approval as the first treatment indicated for COVID-19 in October.
ACTT-1 lead author John H. Beigel, MD, and colleagues also looked at the death rates for remdesivir versus placebo.
By day 15, the proportion of people who died was 6.7% in the remdesivir group versus 11%. By day 29, the rate was 11.4% among those who received the antiviral versus 15.2% among those who did not.
When asked why the VA and ACTT-1 studies yielded different results, Dr. Beigel cited two reasons. The timing was different, with the VA study starting after the remdesivir EUA was issued, and ACTT-1 findings were announced.
“So at that point, clinicians understood those populations most likely to benefit from remdesivir. The use of remdesivir likely did not occur at random; it was likely to be more commonly used in those who were sicker or at higher risk for poor outcomes,” said Dr. Beigel, associate director for clinical research in the Division of Microbiology and Infectious Diseases at the National Institute of Allergy and Infectious Diseases (NIAID).
In addition, the studies evaluated very different populations, he said. The differences in median duration of hospitalization between the trials reflects this, Dr. Beigel added.
Furthermore, when asked if he thinks the new evidence should affect clinical use of remdesivir, Dr. Beigel replied, “No. Observational studies, even with adjustments such as propensity score matching, are not equivalent levels of proof compared to randomized trials.”
Study details
Dr. Ohl and colleagues identified patients admitted to one of 123 VA hospitals for the first time for COVID-19 from May 1 to Oct. 8, 2020. Each had a PCR-confirmed SARS-CoV-2 infection. The researchers then compared 1,172 patients receiving remdesivir to another 1,172 patients not receiving the agent.
Those receiving remdesivir were more likely to be older, White, have chronic obstructive pulmonary disease and have more severe COVID-19. A total 94% of the remdesivir group were men.
“Over 90% of the people included in VA study were men, mostly over the age of 60,” Dr. Ohl said when asked how generalizable the findings would be to a non-VA population.
“There is no obvious biological reason that remdesivir should have different effects in men and women, but we should be cautious about extrapolating study findings to women and younger individuals,” he added.
Limitations of the study include its observational design, which makes unadjusted confounding based on illness severity a possibility. In addition, the investigators were unable to identify specific subgroups that might benefit from remdesivir treatment.
The data did suggest that remdesivir was more effective earlier in the course of disease when patients required supplemental oxygen and before need for mechanical ventilation.
Dr. Baracco pointed out the contradictory findings in his commentary: “The real-life application of a drug promising to hasten discharge from the hospital as its primary beneficial outcome must include an assessment of how easy it is to do so and make it clear that once a patient reaches that point, they can discontinue the drug.”
“The paradoxical findings in the study by Dr. Ohl et al. compared with the study used for its authorization illustrate this point very clearly,” he adds.
Dr. Ohl reported receiving grants from Veterans Affairs Health Services Research and Development during the conduct of the study and consulting for Gilead Pharmaceuticals outside the submitted work. Dr. Baracco reported receiving salary support from the U.S. Department of Veterans Affairs. Dr. Beigel has disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
DOACs linked to lower mortality than vitamin K antagonist: 3-year TAVR registry
Following a transcatheter aortic valve replacement (TAVR), direct oral anticoagulants (DOACs) are preferable to vitamin K antagonists (VKAs) in patients who are candidates for oral anticoagulants, according to data drawn from a large multicenter French TAVR registry.
When oral anticoagulation is appropriate following TAVR, such as in patients with atrial fibrillation, “DOACs are associated with improved survival and lower incidence of bleeding, compared to VKA,” reported a team of investigators led by Martine Gilard, MD, PhD, director of interventional cardiology, Brest (France) University Hospital Center.
The comparison, using propensity score matching, is not definitive, but it might be the best data currently available to support DOACs over VKA until a randomized trial is completed, according to Dr. Gilard, senior author of the newly published study.
Asked in an interview if DOACs should now be used preferentially after TAVR when patients are indicated for oral anticoagulation, Dr. Gilard replied, “My answer is yes.”
Of more than 24,000 TAVR patients in the French TAVI and FRANCE2 multicenter registries, which are linked to the French single-payer claims database (SNDS), 8,962 (36.4%) received an oral anticoagulant following their procedure. Of these, 2,180 (24.3%) received a DOAC and the remaining received VKA.
By linking data from the registries to the SNDS, outcomes were tracked. Propensity matching was employed to control for differences in baseline characteristics, including age, body mass index, functional class, diabetes, comorbidities, and past medical history.
On the primary endpoint of mortality at the end of 3 years, the rates were 35.6% and 31.2% for VKA and DOACs, respectively. This translated in a 37% greater hazard ratio for death among those treated with VKA (P < .005).
The rate of major bleeding, a secondary endpoint, was also higher (12.3% vs. 8.4%) and significantly different (HR, 1.65; P < .005) for VKA versus DOACs. The rates of ischemic stroke, acute coronary syndrome, and hemorrhagic stroke were all numerically higher in patients treated with VKA than DOACs, although none of these differences reached statistical significance.
Residual confounding cannot be discounted
“The large number of events allowed for taking into account a higher number of potential confounders with appropriate statistical power,” according to the authors. However, they acknowledged that residual confounding cannot be eliminated by propensity matching and conceded that prospective data are needed for a definitive comparison.
In an accompanying editorial, Daniele Giacoppo, MD, a cardiologist at Alto Vicentino Hospital, Santorso, Italy, enlarged on this point . In addition to the inherent limitations of retrospective data, he also noted that data from other studies addressing the same question have been inconsistent.
Of these studies, he pointed to the ATLANTIS trial, presented 2 months ago at the annual meeting of the American College of Cardiology. This study failed to show an advantage for the DOAC apixaban over VKA in TAVR patients for the primary composite outcome of time to death, myocardial infarction, systemic emboli, valve thrombosis, or major bleeding. Although this study was not limited to patients with an indication for oral anticoagulants, Dr. Giacoppo pointed out that there was no advantage, even among the subgroup of patients who did have an indication.
Data are supportive in absence of trial results
In general, Dr. Giacoppo agreed that the French registry are generally supportive of DOACs over VKA in TAVR patients with an indication for oral anticoagulation, but he cautioned that blanket statements are difficult. He anticipates better information from a randomized trial called ENVISAGE-TAVI AF, which is comparing edoxaban with VKA following TAVR in atrial fibrillation patients who have an indication for oral anticoagulation, but he indicated that some individualization of choice will be needed among those high or low relative risks of thrombotic events or bleeding.
“The concerns related to DOACs after TAVR are most confined to patients without an indication for oral anticoagulation,” Dr. Giacoppo said in an interview. In patients with an indication, “oral anticoagulation alone without antithrombotic therapy significantly reduced the risk of bleeding” in several studies, he added, citing in particular the POPular TAVI trial.
Issues about when to employ – or not employ – both oral anticoagulation and antithrombotic therapy based on such factors as bleeding risk remain unresolved, but “in aggregate, waiting for additional high-quality data, the use of a DOAC in patients with an indication for oral anticoagulation who underwent TAVR seems to be safe,” Dr. Giacoppo said. He thinks that the “higher predictability of DOACS compared to vitamin K antagonists might translate into lower bleeding rates over time in a real-world, unselected population.”
Benefit-to-risk ratio requires attention
A similar concern about balancing risks and benefits of oral anticoagulation in TAVR patients with an indication for oral anticoagulation was emphasized by Ron Waksman, MD, associate director, division of cardiology, MedStar Washington (D.C.) Hospital Center.
“The TAVR population is elderly in general and so are at high risk for bleeding with any additional anticoagulation,” Dr. Waksman said. He cited data that bring into question the utility of using a DOAC in TAVR patients without an additional indication for anticoagulation, but he believes DOACs do make sense in those who were on and had an indication for a DOAC even before TAVR.
Patients who had atrial fibrillation or another indication “should continue to take the DOAC after TAVR. This population can be assumed to have less bleeding risk as they are vetted as safe for DOACs before their TAVR procedure,” he said.
Although mortality was the primary endpoint of the French registry evaluation, it is the bleeding risk that is a dominant concern, according to Romain Didier, MD, PhD, the first author of this study who performed this work in collaboration with Dr. Gilard.
“We really believe that VKA use in real life after TAVR, even with INR monitoring, is associated with a higher risk of bleeding as compared to DOACs,” he said. It is for this reason that “we currently use DOACs as a first choice in patients who require anticoagulant after TAVR.”
Dr. Gilard, Dr. Didier, and Dr. Giacoppo reported no potential conflicts of interest. Dr. Waksman reported financial relationships with Amgen, AstraZeneca, Boston Scientific, Cardioset, Cardiovascular Systems, Chiesi, MedAlliance, Medtronic, and Pi-Cardia.
Following a transcatheter aortic valve replacement (TAVR), direct oral anticoagulants (DOACs) are preferable to vitamin K antagonists (VKAs) in patients who are candidates for oral anticoagulants, according to data drawn from a large multicenter French TAVR registry.
When oral anticoagulation is appropriate following TAVR, such as in patients with atrial fibrillation, “DOACs are associated with improved survival and lower incidence of bleeding, compared to VKA,” reported a team of investigators led by Martine Gilard, MD, PhD, director of interventional cardiology, Brest (France) University Hospital Center.
The comparison, using propensity score matching, is not definitive, but it might be the best data currently available to support DOACs over VKA until a randomized trial is completed, according to Dr. Gilard, senior author of the newly published study.
Asked in an interview if DOACs should now be used preferentially after TAVR when patients are indicated for oral anticoagulation, Dr. Gilard replied, “My answer is yes.”
Of more than 24,000 TAVR patients in the French TAVI and FRANCE2 multicenter registries, which are linked to the French single-payer claims database (SNDS), 8,962 (36.4%) received an oral anticoagulant following their procedure. Of these, 2,180 (24.3%) received a DOAC and the remaining received VKA.
By linking data from the registries to the SNDS, outcomes were tracked. Propensity matching was employed to control for differences in baseline characteristics, including age, body mass index, functional class, diabetes, comorbidities, and past medical history.
On the primary endpoint of mortality at the end of 3 years, the rates were 35.6% and 31.2% for VKA and DOACs, respectively. This translated in a 37% greater hazard ratio for death among those treated with VKA (P < .005).
The rate of major bleeding, a secondary endpoint, was also higher (12.3% vs. 8.4%) and significantly different (HR, 1.65; P < .005) for VKA versus DOACs. The rates of ischemic stroke, acute coronary syndrome, and hemorrhagic stroke were all numerically higher in patients treated with VKA than DOACs, although none of these differences reached statistical significance.
Residual confounding cannot be discounted
“The large number of events allowed for taking into account a higher number of potential confounders with appropriate statistical power,” according to the authors. However, they acknowledged that residual confounding cannot be eliminated by propensity matching and conceded that prospective data are needed for a definitive comparison.
In an accompanying editorial, Daniele Giacoppo, MD, a cardiologist at Alto Vicentino Hospital, Santorso, Italy, enlarged on this point . In addition to the inherent limitations of retrospective data, he also noted that data from other studies addressing the same question have been inconsistent.
Of these studies, he pointed to the ATLANTIS trial, presented 2 months ago at the annual meeting of the American College of Cardiology. This study failed to show an advantage for the DOAC apixaban over VKA in TAVR patients for the primary composite outcome of time to death, myocardial infarction, systemic emboli, valve thrombosis, or major bleeding. Although this study was not limited to patients with an indication for oral anticoagulants, Dr. Giacoppo pointed out that there was no advantage, even among the subgroup of patients who did have an indication.
Data are supportive in absence of trial results
In general, Dr. Giacoppo agreed that the French registry are generally supportive of DOACs over VKA in TAVR patients with an indication for oral anticoagulation, but he cautioned that blanket statements are difficult. He anticipates better information from a randomized trial called ENVISAGE-TAVI AF, which is comparing edoxaban with VKA following TAVR in atrial fibrillation patients who have an indication for oral anticoagulation, but he indicated that some individualization of choice will be needed among those high or low relative risks of thrombotic events or bleeding.
“The concerns related to DOACs after TAVR are most confined to patients without an indication for oral anticoagulation,” Dr. Giacoppo said in an interview. In patients with an indication, “oral anticoagulation alone without antithrombotic therapy significantly reduced the risk of bleeding” in several studies, he added, citing in particular the POPular TAVI trial.
Issues about when to employ – or not employ – both oral anticoagulation and antithrombotic therapy based on such factors as bleeding risk remain unresolved, but “in aggregate, waiting for additional high-quality data, the use of a DOAC in patients with an indication for oral anticoagulation who underwent TAVR seems to be safe,” Dr. Giacoppo said. He thinks that the “higher predictability of DOACS compared to vitamin K antagonists might translate into lower bleeding rates over time in a real-world, unselected population.”
Benefit-to-risk ratio requires attention
A similar concern about balancing risks and benefits of oral anticoagulation in TAVR patients with an indication for oral anticoagulation was emphasized by Ron Waksman, MD, associate director, division of cardiology, MedStar Washington (D.C.) Hospital Center.
“The TAVR population is elderly in general and so are at high risk for bleeding with any additional anticoagulation,” Dr. Waksman said. He cited data that bring into question the utility of using a DOAC in TAVR patients without an additional indication for anticoagulation, but he believes DOACs do make sense in those who were on and had an indication for a DOAC even before TAVR.
Patients who had atrial fibrillation or another indication “should continue to take the DOAC after TAVR. This population can be assumed to have less bleeding risk as they are vetted as safe for DOACs before their TAVR procedure,” he said.
Although mortality was the primary endpoint of the French registry evaluation, it is the bleeding risk that is a dominant concern, according to Romain Didier, MD, PhD, the first author of this study who performed this work in collaboration with Dr. Gilard.
“We really believe that VKA use in real life after TAVR, even with INR monitoring, is associated with a higher risk of bleeding as compared to DOACs,” he said. It is for this reason that “we currently use DOACs as a first choice in patients who require anticoagulant after TAVR.”
Dr. Gilard, Dr. Didier, and Dr. Giacoppo reported no potential conflicts of interest. Dr. Waksman reported financial relationships with Amgen, AstraZeneca, Boston Scientific, Cardioset, Cardiovascular Systems, Chiesi, MedAlliance, Medtronic, and Pi-Cardia.
Following a transcatheter aortic valve replacement (TAVR), direct oral anticoagulants (DOACs) are preferable to vitamin K antagonists (VKAs) in patients who are candidates for oral anticoagulants, according to data drawn from a large multicenter French TAVR registry.
When oral anticoagulation is appropriate following TAVR, such as in patients with atrial fibrillation, “DOACs are associated with improved survival and lower incidence of bleeding, compared to VKA,” reported a team of investigators led by Martine Gilard, MD, PhD, director of interventional cardiology, Brest (France) University Hospital Center.
The comparison, using propensity score matching, is not definitive, but it might be the best data currently available to support DOACs over VKA until a randomized trial is completed, according to Dr. Gilard, senior author of the newly published study.
Asked in an interview if DOACs should now be used preferentially after TAVR when patients are indicated for oral anticoagulation, Dr. Gilard replied, “My answer is yes.”
Of more than 24,000 TAVR patients in the French TAVI and FRANCE2 multicenter registries, which are linked to the French single-payer claims database (SNDS), 8,962 (36.4%) received an oral anticoagulant following their procedure. Of these, 2,180 (24.3%) received a DOAC and the remaining received VKA.
By linking data from the registries to the SNDS, outcomes were tracked. Propensity matching was employed to control for differences in baseline characteristics, including age, body mass index, functional class, diabetes, comorbidities, and past medical history.
On the primary endpoint of mortality at the end of 3 years, the rates were 35.6% and 31.2% for VKA and DOACs, respectively. This translated in a 37% greater hazard ratio for death among those treated with VKA (P < .005).
The rate of major bleeding, a secondary endpoint, was also higher (12.3% vs. 8.4%) and significantly different (HR, 1.65; P < .005) for VKA versus DOACs. The rates of ischemic stroke, acute coronary syndrome, and hemorrhagic stroke were all numerically higher in patients treated with VKA than DOACs, although none of these differences reached statistical significance.
Residual confounding cannot be discounted
“The large number of events allowed for taking into account a higher number of potential confounders with appropriate statistical power,” according to the authors. However, they acknowledged that residual confounding cannot be eliminated by propensity matching and conceded that prospective data are needed for a definitive comparison.
In an accompanying editorial, Daniele Giacoppo, MD, a cardiologist at Alto Vicentino Hospital, Santorso, Italy, enlarged on this point . In addition to the inherent limitations of retrospective data, he also noted that data from other studies addressing the same question have been inconsistent.
Of these studies, he pointed to the ATLANTIS trial, presented 2 months ago at the annual meeting of the American College of Cardiology. This study failed to show an advantage for the DOAC apixaban over VKA in TAVR patients for the primary composite outcome of time to death, myocardial infarction, systemic emboli, valve thrombosis, or major bleeding. Although this study was not limited to patients with an indication for oral anticoagulants, Dr. Giacoppo pointed out that there was no advantage, even among the subgroup of patients who did have an indication.
Data are supportive in absence of trial results
In general, Dr. Giacoppo agreed that the French registry are generally supportive of DOACs over VKA in TAVR patients with an indication for oral anticoagulation, but he cautioned that blanket statements are difficult. He anticipates better information from a randomized trial called ENVISAGE-TAVI AF, which is comparing edoxaban with VKA following TAVR in atrial fibrillation patients who have an indication for oral anticoagulation, but he indicated that some individualization of choice will be needed among those high or low relative risks of thrombotic events or bleeding.
“The concerns related to DOACs after TAVR are most confined to patients without an indication for oral anticoagulation,” Dr. Giacoppo said in an interview. In patients with an indication, “oral anticoagulation alone without antithrombotic therapy significantly reduced the risk of bleeding” in several studies, he added, citing in particular the POPular TAVI trial.
Issues about when to employ – or not employ – both oral anticoagulation and antithrombotic therapy based on such factors as bleeding risk remain unresolved, but “in aggregate, waiting for additional high-quality data, the use of a DOAC in patients with an indication for oral anticoagulation who underwent TAVR seems to be safe,” Dr. Giacoppo said. He thinks that the “higher predictability of DOACS compared to vitamin K antagonists might translate into lower bleeding rates over time in a real-world, unselected population.”
Benefit-to-risk ratio requires attention
A similar concern about balancing risks and benefits of oral anticoagulation in TAVR patients with an indication for oral anticoagulation was emphasized by Ron Waksman, MD, associate director, division of cardiology, MedStar Washington (D.C.) Hospital Center.
“The TAVR population is elderly in general and so are at high risk for bleeding with any additional anticoagulation,” Dr. Waksman said. He cited data that bring into question the utility of using a DOAC in TAVR patients without an additional indication for anticoagulation, but he believes DOACs do make sense in those who were on and had an indication for a DOAC even before TAVR.
Patients who had atrial fibrillation or another indication “should continue to take the DOAC after TAVR. This population can be assumed to have less bleeding risk as they are vetted as safe for DOACs before their TAVR procedure,” he said.
Although mortality was the primary endpoint of the French registry evaluation, it is the bleeding risk that is a dominant concern, according to Romain Didier, MD, PhD, the first author of this study who performed this work in collaboration with Dr. Gilard.
“We really believe that VKA use in real life after TAVR, even with INR monitoring, is associated with a higher risk of bleeding as compared to DOACs,” he said. It is for this reason that “we currently use DOACs as a first choice in patients who require anticoagulant after TAVR.”
Dr. Gilard, Dr. Didier, and Dr. Giacoppo reported no potential conflicts of interest. Dr. Waksman reported financial relationships with Amgen, AstraZeneca, Boston Scientific, Cardioset, Cardiovascular Systems, Chiesi, MedAlliance, Medtronic, and Pi-Cardia.
FROM JACC: CARDIOVASCULAR INTERVENTIONS
Legalization of cannabis tied to drop in opioid-related ED visits
State laws permitting recreational marijuana use have not led to an increase in opioid-related emergency department visits, as many had feared.
On the contrary, states that legalize recreational marijuana may see a short-term decrease in opioid-related ED visits in the first 6 months, after which rates may return to prelegalization levels, new research suggests.
Previous research suggests that individuals may reduce the use of opioids when they have an alternative and that cannabis can provide pain relief.
“At the same time, we often hear claims from politicians that we should not legalize cannabis because it may act as a ‘gateway drug’ that leads to use of other drugs,” lead researcher Coleman Drake, PhD, Department of Health Policy and Management, University of Pittsburgh Graduate School of Public Health, told this news organization.
“Our findings indicate that cannabis legalization does not effect any increase in opioid-related ED visits, contradicting the gateway drug explanation,” Dr. Drake said.
The study was published online July 12 in Health Economics.
Significant reduction
So far, 19 states have legalized recreational cannabis, meaning that nearly half of the U.S. population lives in a state that allows recreational cannabis use.
The investigators analyzed data on opioid-related ED visits from 29 states between 2011 and 2017. Four states – California, Maine, Massachusetts, and Nevada – legalized recreational marijuana during the study period; the remaining 25 states did not.
The four states with recreational cannabis laws experienced a 7.6% reduction in opioid-related ED visits for 6 months after the law went into effect in comparison with the states that did not legalize recreational marijuana.
“This isn’t trivial – a decline in opioid-related emergency department visits, even if only for 6 months, is a welcome public health development,” Dr. Drake said in a statement.
Not surprisingly, these effects are driven by men and adults aged 25 to 44 years. “These are populations that are more likely to use cannabis, and the reduction in opioid-related ED visits that we find is concentrated among them,” Dr. Drake told this news organization.
However, the downturn in opioid-related ED visits after making marijuana legal was only temporary.
“
Encouragingly, he said, the data show that opioid-related ED visits don’t increase above baseline after recreational marijuana laws are adopted.
“We conclude that cannabis legalization likely is not a panacea for the opioid epidemic, but there are some helpful effects,” Dr. Drake said in an interview.
The study was supported by the National Institute on Drug Abuse. The authors have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
State laws permitting recreational marijuana use have not led to an increase in opioid-related emergency department visits, as many had feared.
On the contrary, states that legalize recreational marijuana may see a short-term decrease in opioid-related ED visits in the first 6 months, after which rates may return to prelegalization levels, new research suggests.
Previous research suggests that individuals may reduce the use of opioids when they have an alternative and that cannabis can provide pain relief.
“At the same time, we often hear claims from politicians that we should not legalize cannabis because it may act as a ‘gateway drug’ that leads to use of other drugs,” lead researcher Coleman Drake, PhD, Department of Health Policy and Management, University of Pittsburgh Graduate School of Public Health, told this news organization.
“Our findings indicate that cannabis legalization does not effect any increase in opioid-related ED visits, contradicting the gateway drug explanation,” Dr. Drake said.
The study was published online July 12 in Health Economics.
Significant reduction
So far, 19 states have legalized recreational cannabis, meaning that nearly half of the U.S. population lives in a state that allows recreational cannabis use.
The investigators analyzed data on opioid-related ED visits from 29 states between 2011 and 2017. Four states – California, Maine, Massachusetts, and Nevada – legalized recreational marijuana during the study period; the remaining 25 states did not.
The four states with recreational cannabis laws experienced a 7.6% reduction in opioid-related ED visits for 6 months after the law went into effect in comparison with the states that did not legalize recreational marijuana.
“This isn’t trivial – a decline in opioid-related emergency department visits, even if only for 6 months, is a welcome public health development,” Dr. Drake said in a statement.
Not surprisingly, these effects are driven by men and adults aged 25 to 44 years. “These are populations that are more likely to use cannabis, and the reduction in opioid-related ED visits that we find is concentrated among them,” Dr. Drake told this news organization.
However, the downturn in opioid-related ED visits after making marijuana legal was only temporary.
“
Encouragingly, he said, the data show that opioid-related ED visits don’t increase above baseline after recreational marijuana laws are adopted.
“We conclude that cannabis legalization likely is not a panacea for the opioid epidemic, but there are some helpful effects,” Dr. Drake said in an interview.
The study was supported by the National Institute on Drug Abuse. The authors have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
State laws permitting recreational marijuana use have not led to an increase in opioid-related emergency department visits, as many had feared.
On the contrary, states that legalize recreational marijuana may see a short-term decrease in opioid-related ED visits in the first 6 months, after which rates may return to prelegalization levels, new research suggests.
Previous research suggests that individuals may reduce the use of opioids when they have an alternative and that cannabis can provide pain relief.
“At the same time, we often hear claims from politicians that we should not legalize cannabis because it may act as a ‘gateway drug’ that leads to use of other drugs,” lead researcher Coleman Drake, PhD, Department of Health Policy and Management, University of Pittsburgh Graduate School of Public Health, told this news organization.
“Our findings indicate that cannabis legalization does not effect any increase in opioid-related ED visits, contradicting the gateway drug explanation,” Dr. Drake said.
The study was published online July 12 in Health Economics.
Significant reduction
So far, 19 states have legalized recreational cannabis, meaning that nearly half of the U.S. population lives in a state that allows recreational cannabis use.
The investigators analyzed data on opioid-related ED visits from 29 states between 2011 and 2017. Four states – California, Maine, Massachusetts, and Nevada – legalized recreational marijuana during the study period; the remaining 25 states did not.
The four states with recreational cannabis laws experienced a 7.6% reduction in opioid-related ED visits for 6 months after the law went into effect in comparison with the states that did not legalize recreational marijuana.
“This isn’t trivial – a decline in opioid-related emergency department visits, even if only for 6 months, is a welcome public health development,” Dr. Drake said in a statement.
Not surprisingly, these effects are driven by men and adults aged 25 to 44 years. “These are populations that are more likely to use cannabis, and the reduction in opioid-related ED visits that we find is concentrated among them,” Dr. Drake told this news organization.
However, the downturn in opioid-related ED visits after making marijuana legal was only temporary.
“
Encouragingly, he said, the data show that opioid-related ED visits don’t increase above baseline after recreational marijuana laws are adopted.
“We conclude that cannabis legalization likely is not a panacea for the opioid epidemic, but there are some helpful effects,” Dr. Drake said in an interview.
The study was supported by the National Institute on Drug Abuse. The authors have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Long-term outcome data suggest optimism for MIS-C patients
Only 1 child from a cohort of 45 children hospitalized with multisystem inflammatory syndrome following COVID-19 infection had persistent mild cardiac dysfunction after 9 months, according to data from patients younger than 21 years seen at a single center in 2020.
In a study published in Pediatrics, Kanwal M. Farooqi, MD, of Columbia University, New York, and colleagues provided the first report on longitudinal cardiac and immunologic outcomes in North American children hospitalized with multisystem inflammatory syndrome (MIS-C). In response to the COVID-19 pandemic, clinicians at New York–Presbyterian Hospital consolidated pediatric admissions and developed an interdisciplinary inpatient and outpatient MIS-C follow-up program to monitor cardiac and immunologic outcomes in their patients.
The study included all children younger than 21 years admitted to Columbia University Irving Medical Center/New York–Presbyterian Morgan Stanley Children’s Hospital for MIS-C in 2020. The median age of the patients was 9 years, and the median length of hospital stay was 5 days. Follow-up visits occurred at 1-4 weeks (average 2 weeks), 1-4 months (average 2 months), and 4-9 months (average 6 months) after hospital discharge. Follow-up visits included echocardiograms and measures of inflammatory markers.
Most of the children (84%) had no underlying medical conditions, but 24% presented with some level of respiratory distress or oxygen requirement, and 64% had vasodilatory shock. In addition, 80% had at least mild cardiac abnormalities and 66% had significant lymphopenia on admission.
Inflammatory profiles on admission showed elevation of C-reactive protein, ferritin, and D-dimer in 87%-98% of the patients. Consistent with cardiac involvement, 64% of the patients also had elevated troponin levels, and 91% had elevated N-terminal pro-brain natriuretic peptide (NT-proBNP) levels.
“These parameters peaked at or shortly after admission and then gradually normalized,” the researchers said. “By the first follow-up, [C-reactive protein], troponin, and NT-proBNP had normalized in nearly all tested patients (97%-100%),” they noted.
By the first follow-up period at 1-4 weeks, all patients had normal coronary arteries, and 18% (seven patients) had mild echocardiographic findings. However, approximately one-third (32%) of the patients had persistent lymphocytosis at 1-4 weeks, and 23 of the 24 patients assessed had elevated double-negative T cells, which persisted in 96% of the patients at 1-4 months’ follow-up. However, during the last follow-up of 4-9 months, only one patient had persistent mild biventricular dysfunction and a second patient had mild mitral and tricuspid valve regurgitation.
All patients were treated with steroids and immunoglobulins (2 g/kg), as well as enoxaparin prophylaxis or low-dose aspirin and GI prophylaxis. Treatment with methylprednisolone varied based on disease severity; patients with mild presentation received 2 mg/kg per day; those with moderate presentation received a methylprednisolone pulse of 10 mg/kg per day, followed by 2 mg/kg per day; those with severe disease received methylprednisolone at 20-30 mg/kg per day for 1-3 days, followed by 2 mg/kg per day.
“Aggressive use of steroids may also explain the lower incidence of coronary artery abnormalities in our cohort,” the researchers noted.
The study findings were limited by the observational design and inability to make definitive conclusions about treatment and outcomes, as well as the evolving case definitions for MIS-C, the researchers said.
The persistence of double-negative T cells was surprising, and “likely represent a prolonged postinflammatory recovery cell population, but further study is ongoing to better define this observation,” they noted.
“Our study reveals generally encouraging medium-term outcomes, including rapid normalization of inflammatory markers and significant cardiac abnormalities in the majority of patients with MIS-C,” the researchers said. “The exact nature and potential for long-term cardiac fibrosis, exercise intolerance, or other changes remain unknown,” and long-term caution and follow-up are recommended, they concluded.
Cautious optimism, long-term monitoring
The study is important to provide guidance for clinicians on how to manage their patients who have been hospitalized with MIS-C, said Susan Boulter, MD, of the Geisel School of Medicine at Dartmouth, Hanover, N.H.
“It was both surprising and reassuring to see that so many of the patients had positive outcomes in terms of cardiac function and that during the acute stage there were no deaths,” said Dr. Boulter. “Hospitalizations were brief, averaging just 5 days. The patients had many symptoms, but unlike adults, there was not a preponderance of underlying risk factors in this cohort of patients,” she said.
The results suggest optimism for MIS-C patients in that they generally recover, but the take-home message for clinicians is that these patients will require careful monitoring for long-term issues, Dr. Boulter said.
“These patients should be followed for years to assess long-term effects on morbidity and mortality,” Dr. Boulter emphasized.
The study was funded by Genentech. The researchers had no financial conflicts to disclose. Dr. Boulter had no financial conflicts to disclose, but serves on the Pediatric News Editorial Advisory Board.
Only 1 child from a cohort of 45 children hospitalized with multisystem inflammatory syndrome following COVID-19 infection had persistent mild cardiac dysfunction after 9 months, according to data from patients younger than 21 years seen at a single center in 2020.
In a study published in Pediatrics, Kanwal M. Farooqi, MD, of Columbia University, New York, and colleagues provided the first report on longitudinal cardiac and immunologic outcomes in North American children hospitalized with multisystem inflammatory syndrome (MIS-C). In response to the COVID-19 pandemic, clinicians at New York–Presbyterian Hospital consolidated pediatric admissions and developed an interdisciplinary inpatient and outpatient MIS-C follow-up program to monitor cardiac and immunologic outcomes in their patients.
The study included all children younger than 21 years admitted to Columbia University Irving Medical Center/New York–Presbyterian Morgan Stanley Children’s Hospital for MIS-C in 2020. The median age of the patients was 9 years, and the median length of hospital stay was 5 days. Follow-up visits occurred at 1-4 weeks (average 2 weeks), 1-4 months (average 2 months), and 4-9 months (average 6 months) after hospital discharge. Follow-up visits included echocardiograms and measures of inflammatory markers.
Most of the children (84%) had no underlying medical conditions, but 24% presented with some level of respiratory distress or oxygen requirement, and 64% had vasodilatory shock. In addition, 80% had at least mild cardiac abnormalities and 66% had significant lymphopenia on admission.
Inflammatory profiles on admission showed elevation of C-reactive protein, ferritin, and D-dimer in 87%-98% of the patients. Consistent with cardiac involvement, 64% of the patients also had elevated troponin levels, and 91% had elevated N-terminal pro-brain natriuretic peptide (NT-proBNP) levels.
“These parameters peaked at or shortly after admission and then gradually normalized,” the researchers said. “By the first follow-up, [C-reactive protein], troponin, and NT-proBNP had normalized in nearly all tested patients (97%-100%),” they noted.
By the first follow-up period at 1-4 weeks, all patients had normal coronary arteries, and 18% (seven patients) had mild echocardiographic findings. However, approximately one-third (32%) of the patients had persistent lymphocytosis at 1-4 weeks, and 23 of the 24 patients assessed had elevated double-negative T cells, which persisted in 96% of the patients at 1-4 months’ follow-up. However, during the last follow-up of 4-9 months, only one patient had persistent mild biventricular dysfunction and a second patient had mild mitral and tricuspid valve regurgitation.
All patients were treated with steroids and immunoglobulins (2 g/kg), as well as enoxaparin prophylaxis or low-dose aspirin and GI prophylaxis. Treatment with methylprednisolone varied based on disease severity; patients with mild presentation received 2 mg/kg per day; those with moderate presentation received a methylprednisolone pulse of 10 mg/kg per day, followed by 2 mg/kg per day; those with severe disease received methylprednisolone at 20-30 mg/kg per day for 1-3 days, followed by 2 mg/kg per day.
“Aggressive use of steroids may also explain the lower incidence of coronary artery abnormalities in our cohort,” the researchers noted.
The study findings were limited by the observational design and inability to make definitive conclusions about treatment and outcomes, as well as the evolving case definitions for MIS-C, the researchers said.
The persistence of double-negative T cells was surprising, and “likely represent a prolonged postinflammatory recovery cell population, but further study is ongoing to better define this observation,” they noted.
“Our study reveals generally encouraging medium-term outcomes, including rapid normalization of inflammatory markers and significant cardiac abnormalities in the majority of patients with MIS-C,” the researchers said. “The exact nature and potential for long-term cardiac fibrosis, exercise intolerance, or other changes remain unknown,” and long-term caution and follow-up are recommended, they concluded.
Cautious optimism, long-term monitoring
The study is important to provide guidance for clinicians on how to manage their patients who have been hospitalized with MIS-C, said Susan Boulter, MD, of the Geisel School of Medicine at Dartmouth, Hanover, N.H.
“It was both surprising and reassuring to see that so many of the patients had positive outcomes in terms of cardiac function and that during the acute stage there were no deaths,” said Dr. Boulter. “Hospitalizations were brief, averaging just 5 days. The patients had many symptoms, but unlike adults, there was not a preponderance of underlying risk factors in this cohort of patients,” she said.
The results suggest optimism for MIS-C patients in that they generally recover, but the take-home message for clinicians is that these patients will require careful monitoring for long-term issues, Dr. Boulter said.
“These patients should be followed for years to assess long-term effects on morbidity and mortality,” Dr. Boulter emphasized.
The study was funded by Genentech. The researchers had no financial conflicts to disclose. Dr. Boulter had no financial conflicts to disclose, but serves on the Pediatric News Editorial Advisory Board.
Only 1 child from a cohort of 45 children hospitalized with multisystem inflammatory syndrome following COVID-19 infection had persistent mild cardiac dysfunction after 9 months, according to data from patients younger than 21 years seen at a single center in 2020.
In a study published in Pediatrics, Kanwal M. Farooqi, MD, of Columbia University, New York, and colleagues provided the first report on longitudinal cardiac and immunologic outcomes in North American children hospitalized with multisystem inflammatory syndrome (MIS-C). In response to the COVID-19 pandemic, clinicians at New York–Presbyterian Hospital consolidated pediatric admissions and developed an interdisciplinary inpatient and outpatient MIS-C follow-up program to monitor cardiac and immunologic outcomes in their patients.
The study included all children younger than 21 years admitted to Columbia University Irving Medical Center/New York–Presbyterian Morgan Stanley Children’s Hospital for MIS-C in 2020. The median age of the patients was 9 years, and the median length of hospital stay was 5 days. Follow-up visits occurred at 1-4 weeks (average 2 weeks), 1-4 months (average 2 months), and 4-9 months (average 6 months) after hospital discharge. Follow-up visits included echocardiograms and measures of inflammatory markers.
Most of the children (84%) had no underlying medical conditions, but 24% presented with some level of respiratory distress or oxygen requirement, and 64% had vasodilatory shock. In addition, 80% had at least mild cardiac abnormalities and 66% had significant lymphopenia on admission.
Inflammatory profiles on admission showed elevation of C-reactive protein, ferritin, and D-dimer in 87%-98% of the patients. Consistent with cardiac involvement, 64% of the patients also had elevated troponin levels, and 91% had elevated N-terminal pro-brain natriuretic peptide (NT-proBNP) levels.
“These parameters peaked at or shortly after admission and then gradually normalized,” the researchers said. “By the first follow-up, [C-reactive protein], troponin, and NT-proBNP had normalized in nearly all tested patients (97%-100%),” they noted.
By the first follow-up period at 1-4 weeks, all patients had normal coronary arteries, and 18% (seven patients) had mild echocardiographic findings. However, approximately one-third (32%) of the patients had persistent lymphocytosis at 1-4 weeks, and 23 of the 24 patients assessed had elevated double-negative T cells, which persisted in 96% of the patients at 1-4 months’ follow-up. However, during the last follow-up of 4-9 months, only one patient had persistent mild biventricular dysfunction and a second patient had mild mitral and tricuspid valve regurgitation.
All patients were treated with steroids and immunoglobulins (2 g/kg), as well as enoxaparin prophylaxis or low-dose aspirin and GI prophylaxis. Treatment with methylprednisolone varied based on disease severity; patients with mild presentation received 2 mg/kg per day; those with moderate presentation received a methylprednisolone pulse of 10 mg/kg per day, followed by 2 mg/kg per day; those with severe disease received methylprednisolone at 20-30 mg/kg per day for 1-3 days, followed by 2 mg/kg per day.
“Aggressive use of steroids may also explain the lower incidence of coronary artery abnormalities in our cohort,” the researchers noted.
The study findings were limited by the observational design and inability to make definitive conclusions about treatment and outcomes, as well as the evolving case definitions for MIS-C, the researchers said.
The persistence of double-negative T cells was surprising, and “likely represent a prolonged postinflammatory recovery cell population, but further study is ongoing to better define this observation,” they noted.
“Our study reveals generally encouraging medium-term outcomes, including rapid normalization of inflammatory markers and significant cardiac abnormalities in the majority of patients with MIS-C,” the researchers said. “The exact nature and potential for long-term cardiac fibrosis, exercise intolerance, or other changes remain unknown,” and long-term caution and follow-up are recommended, they concluded.
Cautious optimism, long-term monitoring
The study is important to provide guidance for clinicians on how to manage their patients who have been hospitalized with MIS-C, said Susan Boulter, MD, of the Geisel School of Medicine at Dartmouth, Hanover, N.H.
“It was both surprising and reassuring to see that so many of the patients had positive outcomes in terms of cardiac function and that during the acute stage there were no deaths,” said Dr. Boulter. “Hospitalizations were brief, averaging just 5 days. The patients had many symptoms, but unlike adults, there was not a preponderance of underlying risk factors in this cohort of patients,” she said.
The results suggest optimism for MIS-C patients in that they generally recover, but the take-home message for clinicians is that these patients will require careful monitoring for long-term issues, Dr. Boulter said.
“These patients should be followed for years to assess long-term effects on morbidity and mortality,” Dr. Boulter emphasized.
The study was funded by Genentech. The researchers had no financial conflicts to disclose. Dr. Boulter had no financial conflicts to disclose, but serves on the Pediatric News Editorial Advisory Board.
FROM PEDIATRICS
Disconnect between POLST orders and end-of-life care
Background: In order to reduce the mismatch between patients’ desired and actual end-of-life care, the Physician Orders for Life-Sustaining Treatment (POLST) was created. POLST is a portable document delineating medical orders for emergency care treatment at the end of life including whether to attempt resuscitation and general level of medical interventions. For nursing home residents, an association between POLST creation and reduction of unwanted CPR has been substantiated. Outside of this population, the association is unknown.
Study design: Retrospective cohort study.
Setting: Two academic hospitals in Washington.
Synopsis: Patients older than age 18 years who had one of nine chronic health conditions associated with 90% of deaths among Medicare beneficiaries were identified using Washington state death certificates. Additional inclusion criteria included hospital admission in the last 6 months of life and creation of a POLST prior to this admission. This led to identification of 1,818 patients. Patients with full-treatment POLST orders were significantly more likely to be admitted to the ICU as well as receive life-sustaining treatments such as mechanical ventilation, vasoactive infusions, or CPR, compared with patients with limited interventions or comfort-only POLST orders (P < .001 for both). 38% of patients with treatment-limiting POLSTs received aggressive end-of-life care that was discordant with their previously documented wishes.
Bottom line: Completion of POLST was associated with a greater likelihood of receiving end-of-life care that was in line with patients’ previously documented wishes regarding admission to ICU and life-sustaining treatment. Washington was one of the first states to adopt POLST in 2005 and therefore these results may not be broadly applicable.
Citation: Lee RY et al. Association of physician orders for life-sustaining treatment with ICU admission among patients hospitalized hear the end of life. JAMA. 2020 Feb 16;323(10):950-60.
Dr. Dreicer is assistant professor of medicine, section of hospital medicine, at the University of Virginia School of Medicine, Charlottesville.
Background: In order to reduce the mismatch between patients’ desired and actual end-of-life care, the Physician Orders for Life-Sustaining Treatment (POLST) was created. POLST is a portable document delineating medical orders for emergency care treatment at the end of life including whether to attempt resuscitation and general level of medical interventions. For nursing home residents, an association between POLST creation and reduction of unwanted CPR has been substantiated. Outside of this population, the association is unknown.
Study design: Retrospective cohort study.
Setting: Two academic hospitals in Washington.
Synopsis: Patients older than age 18 years who had one of nine chronic health conditions associated with 90% of deaths among Medicare beneficiaries were identified using Washington state death certificates. Additional inclusion criteria included hospital admission in the last 6 months of life and creation of a POLST prior to this admission. This led to identification of 1,818 patients. Patients with full-treatment POLST orders were significantly more likely to be admitted to the ICU as well as receive life-sustaining treatments such as mechanical ventilation, vasoactive infusions, or CPR, compared with patients with limited interventions or comfort-only POLST orders (P < .001 for both). 38% of patients with treatment-limiting POLSTs received aggressive end-of-life care that was discordant with their previously documented wishes.
Bottom line: Completion of POLST was associated with a greater likelihood of receiving end-of-life care that was in line with patients’ previously documented wishes regarding admission to ICU and life-sustaining treatment. Washington was one of the first states to adopt POLST in 2005 and therefore these results may not be broadly applicable.
Citation: Lee RY et al. Association of physician orders for life-sustaining treatment with ICU admission among patients hospitalized hear the end of life. JAMA. 2020 Feb 16;323(10):950-60.
Dr. Dreicer is assistant professor of medicine, section of hospital medicine, at the University of Virginia School of Medicine, Charlottesville.
Background: In order to reduce the mismatch between patients’ desired and actual end-of-life care, the Physician Orders for Life-Sustaining Treatment (POLST) was created. POLST is a portable document delineating medical orders for emergency care treatment at the end of life including whether to attempt resuscitation and general level of medical interventions. For nursing home residents, an association between POLST creation and reduction of unwanted CPR has been substantiated. Outside of this population, the association is unknown.
Study design: Retrospective cohort study.
Setting: Two academic hospitals in Washington.
Synopsis: Patients older than age 18 years who had one of nine chronic health conditions associated with 90% of deaths among Medicare beneficiaries were identified using Washington state death certificates. Additional inclusion criteria included hospital admission in the last 6 months of life and creation of a POLST prior to this admission. This led to identification of 1,818 patients. Patients with full-treatment POLST orders were significantly more likely to be admitted to the ICU as well as receive life-sustaining treatments such as mechanical ventilation, vasoactive infusions, or CPR, compared with patients with limited interventions or comfort-only POLST orders (P < .001 for both). 38% of patients with treatment-limiting POLSTs received aggressive end-of-life care that was discordant with their previously documented wishes.
Bottom line: Completion of POLST was associated with a greater likelihood of receiving end-of-life care that was in line with patients’ previously documented wishes regarding admission to ICU and life-sustaining treatment. Washington was one of the first states to adopt POLST in 2005 and therefore these results may not be broadly applicable.
Citation: Lee RY et al. Association of physician orders for life-sustaining treatment with ICU admission among patients hospitalized hear the end of life. JAMA. 2020 Feb 16;323(10):950-60.
Dr. Dreicer is assistant professor of medicine, section of hospital medicine, at the University of Virginia School of Medicine, Charlottesville.






